User login
Sharon Worcester is an award-winning medical journalist for MDedge News. She has been with the company since 1996, first as the Southeast Bureau Chief (1996-2009) when the company was known as International Medical News Group, then as a freelance writer (2010-2015) before returning as a reporter in 2015. She previously worked as a daily newspaper reporter covering health and local government. Sharon currently reports primarily on oncology and hematology. She has a BA from Eckerd College and an MA in Mass Communication/Print Journalism from the University of Florida. Connect with her via LinkedIn and follow her on twitter @SW_MedReporter.
IV fluid use in heart failure patients raises concerns
Intravenous fluids are frequently administered during early hospitalization in patients with acute heart failure who are receiving loop diuretics, and the practice is associated with worse outcomes, according to findings from a retrospective cohort study.
Of 131,430 hospitalizations for heart failure at 346 hospitals from 2009 to 2010, 13,806 (11%) involved administration of at least 500 mL of IV fluids during the first 2 days in patients on diuretics. Normal saline was used most often (80% of cases), followed by half-normal saline (12%), and the median administered volume was 1,000 mL.
Those who received fluids were significantly more likely than were those who did not to have subsequent critical care admission (odds ratio, 1.57), intubation (OR, 1.46), renal replacement therapy (OR, 2.04), and hospital death (OR, 2.02), reported Dr. Behnood Bikdeli of Yale New Haven (Conn.) Hospital, and colleagues. The report was published Feb. 2 in the Journal of the American College of Cardiology.
Wide variation was seen in the proportion of hospitalizations that used fluid treatment (range of 0% to 71%), the researchers noted (J. Am. Coll. Cardiol. HF 2015 Feb. 2 [doi:10.1016/j.jchf.2014.09.007]).
The use of IV fluids in heart failure patients on diuretics may be inadvertent given that “guidelines generally suggest fluid restriction for patients with heart failure and do not generally recommend intravenous fluid therapy,” the investigators noted, adding that the findings highlight an opportunity for improvement.
The study was funded by two of the National Institutes of Health as well as grants from the Patrick and Catherine Weldon Donaghue Medical Research Foundation in West Hartford, Ct. Dr. Bikdeli reported having no disclosures.
The finding by Bikdeli et al. that fluid administration is common in patients admitted to the hospital with heart failure points to a path forward with respect to improving acute care, according to Dr. Larry A. Allen.
“Now that this relatively common practice is revealed, it behooves us to better understand exactly why it is happening; this understanding can then guide efforts to extinguish truly inappropriate care,” he wrote (J. Am. Coll. Cardiol. HF 2015 Feb. 2 [doi:10.1016/j.jchf.2014.11.001]).
Particular attention should be paid to what matters most with respect to heart failure: a thoughtful approach to the control of fluid status, he said, adding that “good care is grounded in physiology, guided by evidence, and tailored to the patient.
“In the end, we deliver an incredible number and diversity of therapies well in the modern U.S. health care system. The pressing question is whether the totality of therapies makes sense in each individual,” he concluded.
Dr. Allen is with the University of Colorado, Aurora, and the Colorado Cardiovascular Outcomes Research Consortium, Denver. He disclosed funding from the National Heart, Lung, and Blood Institute.
The finding by Bikdeli et al. that fluid administration is common in patients admitted to the hospital with heart failure points to a path forward with respect to improving acute care, according to Dr. Larry A. Allen.
“Now that this relatively common practice is revealed, it behooves us to better understand exactly why it is happening; this understanding can then guide efforts to extinguish truly inappropriate care,” he wrote (J. Am. Coll. Cardiol. HF 2015 Feb. 2 [doi:10.1016/j.jchf.2014.11.001]).
Particular attention should be paid to what matters most with respect to heart failure: a thoughtful approach to the control of fluid status, he said, adding that “good care is grounded in physiology, guided by evidence, and tailored to the patient.
“In the end, we deliver an incredible number and diversity of therapies well in the modern U.S. health care system. The pressing question is whether the totality of therapies makes sense in each individual,” he concluded.
Dr. Allen is with the University of Colorado, Aurora, and the Colorado Cardiovascular Outcomes Research Consortium, Denver. He disclosed funding from the National Heart, Lung, and Blood Institute.
The finding by Bikdeli et al. that fluid administration is common in patients admitted to the hospital with heart failure points to a path forward with respect to improving acute care, according to Dr. Larry A. Allen.
“Now that this relatively common practice is revealed, it behooves us to better understand exactly why it is happening; this understanding can then guide efforts to extinguish truly inappropriate care,” he wrote (J. Am. Coll. Cardiol. HF 2015 Feb. 2 [doi:10.1016/j.jchf.2014.11.001]).
Particular attention should be paid to what matters most with respect to heart failure: a thoughtful approach to the control of fluid status, he said, adding that “good care is grounded in physiology, guided by evidence, and tailored to the patient.
“In the end, we deliver an incredible number and diversity of therapies well in the modern U.S. health care system. The pressing question is whether the totality of therapies makes sense in each individual,” he concluded.
Dr. Allen is with the University of Colorado, Aurora, and the Colorado Cardiovascular Outcomes Research Consortium, Denver. He disclosed funding from the National Heart, Lung, and Blood Institute.
Intravenous fluids are frequently administered during early hospitalization in patients with acute heart failure who are receiving loop diuretics, and the practice is associated with worse outcomes, according to findings from a retrospective cohort study.
Of 131,430 hospitalizations for heart failure at 346 hospitals from 2009 to 2010, 13,806 (11%) involved administration of at least 500 mL of IV fluids during the first 2 days in patients on diuretics. Normal saline was used most often (80% of cases), followed by half-normal saline (12%), and the median administered volume was 1,000 mL.
Those who received fluids were significantly more likely than were those who did not to have subsequent critical care admission (odds ratio, 1.57), intubation (OR, 1.46), renal replacement therapy (OR, 2.04), and hospital death (OR, 2.02), reported Dr. Behnood Bikdeli of Yale New Haven (Conn.) Hospital, and colleagues. The report was published Feb. 2 in the Journal of the American College of Cardiology.
Wide variation was seen in the proportion of hospitalizations that used fluid treatment (range of 0% to 71%), the researchers noted (J. Am. Coll. Cardiol. HF 2015 Feb. 2 [doi:10.1016/j.jchf.2014.09.007]).
The use of IV fluids in heart failure patients on diuretics may be inadvertent given that “guidelines generally suggest fluid restriction for patients with heart failure and do not generally recommend intravenous fluid therapy,” the investigators noted, adding that the findings highlight an opportunity for improvement.
The study was funded by two of the National Institutes of Health as well as grants from the Patrick and Catherine Weldon Donaghue Medical Research Foundation in West Hartford, Ct. Dr. Bikdeli reported having no disclosures.
Intravenous fluids are frequently administered during early hospitalization in patients with acute heart failure who are receiving loop diuretics, and the practice is associated with worse outcomes, according to findings from a retrospective cohort study.
Of 131,430 hospitalizations for heart failure at 346 hospitals from 2009 to 2010, 13,806 (11%) involved administration of at least 500 mL of IV fluids during the first 2 days in patients on diuretics. Normal saline was used most often (80% of cases), followed by half-normal saline (12%), and the median administered volume was 1,000 mL.
Those who received fluids were significantly more likely than were those who did not to have subsequent critical care admission (odds ratio, 1.57), intubation (OR, 1.46), renal replacement therapy (OR, 2.04), and hospital death (OR, 2.02), reported Dr. Behnood Bikdeli of Yale New Haven (Conn.) Hospital, and colleagues. The report was published Feb. 2 in the Journal of the American College of Cardiology.
Wide variation was seen in the proportion of hospitalizations that used fluid treatment (range of 0% to 71%), the researchers noted (J. Am. Coll. Cardiol. HF 2015 Feb. 2 [doi:10.1016/j.jchf.2014.09.007]).
The use of IV fluids in heart failure patients on diuretics may be inadvertent given that “guidelines generally suggest fluid restriction for patients with heart failure and do not generally recommend intravenous fluid therapy,” the investigators noted, adding that the findings highlight an opportunity for improvement.
The study was funded by two of the National Institutes of Health as well as grants from the Patrick and Catherine Weldon Donaghue Medical Research Foundation in West Hartford, Ct. Dr. Bikdeli reported having no disclosures.
Key clinical point: Patients hospitalized for heart failure may receive IV fluids inappropriately.
Major finding: Eleven percent of heart failure patients on diuretics received IV fluids during the first 2 inpatient days.
Data source: A retrospective cohort study of 131,430 hospitalizations.
Disclosures: The study was funded by two of the National Institutes of Health as well as grants from the Patrick and Catherine Weldon Donaghue Medical Research Foundation in West Hartford, Conn. Dr. Bikdeli reported having no disclosures.
Detailed history, nonsedating antihistamines improve management of pediatric urticaria
ORLANDO – Acute urticaria in children is most often caused by infection, food, or medication, and a detailed history is imperative for improving the likelihood of identifying the culprit, according to Dr. Adam Friedman.
Mycoplasma is a particularly common infectious cause in children, but adenovirus, enterovirus, rotavirus, respiratory syncytial virus, Epstein-Barr virus, and cytomegalovirus also have been implicated in urticaria cases, Dr. Friedman, director of dermatologic research at the Albert Einstein College of Medicine, New York, said at the Orlando Dermatology Aesthetic and Clinical Conference.
With respect to foods, ask about intake of milk, eggs, peanuts, wheat, and soy. When it comes to prescription medications, antibiotics are an especially common cause.
“Nonsteroidal anti-inflammatory drugs are also a very important one. If you have a patient with a history of urticaria or a pediatric patient who has a history of urticaria of the mucosa, definitely educate the parents not to give them NSAIDs,” Dr. Friedman said.
Nonimmunologic direct mast cell activation also can be a source of acute urticaria, resulting from exposure to numerous products. These include, but are not limited to, polymyxin B, radiocontrast media, opiates, muscle relaxants, salicylates, and NSAIDs, all of which can “potentially induce urticaria in almost anyone,” he said.
Identifying the cause is more likely in acute case than in chronic cases, for which the etiology is unknown about 70% of time. Regardless, the good news is that about two-thirds of cases are self-limiting; it’s the other third that poses the greatest challenge, he said.
The best bet for nailing down a diagnosis and identifying the cause is an extensive history and physical evaluation.
“Ask a million and one questions to really get to the root of it. In some cases, chronic urticaria is really a diagnosis of exclusion,” he said.
Asking the patient to keep a diary to help identify any unusual exposures just prior to the episode, and having the patient or parent take pictures of the skin are shortcuts that can help.
Extensive laboratory testing is rarely helpful, but certain tests may be warranted. New European guidelines call for erythrocyte sedimentation rate/C-reactive protein and blood differential testing for chronic spontaneous urticaria, but others – like liver function tests, hepatitis B, antinuclear antibody, stool, urinalysis, thyroid function, and antithyroid antibodies – should be directed by the history. A complement panel may be useful in cases involving angioedema, and allergy skin testing may be warranted if a specific trigger can be implicated, but “don’t just order for the sake of ordering,” he said.
Biopsies are not typically useful except in suspected neutrophilic urticaria, which may indicate an association with autoimmune disease, as well as offer some insight into whether dapsone treatment would be helpful over other third-line therapies. Persistent cases of urticaria (lasting over 24-48 hours in one location) may suggest urticarial vasculitis, which would warrant a biopsy.
The therapeutic approach to urticaria involves educating patients about avoiding triggers and identifying and addressing underlying conditions, and using medications that address the pathophysiology of the disease (mast cells, histamine, etc).
In children, the guidelines are generally similar to those in adults, but there is a real push to avoid systemic steroids, Dr. Friedman said, noting that the only time he uses systemic steroids is as a bridge to get to the point where other therapies are beginning to take effect.
Other key concepts for managing urticaria in children, as published in 2013 (Acta. Derm. Venereol. 2013;93:500-8), include using second-generation histamine1 antihistamines for symptom relief, avoiding first-generation H1 antihistamines (due mainly to sedation), and using other therapeutic interventions only after carefully weighing risks and benefits, as evidence in children is lacking.
Nonsedating antihistamines are preferable, as sending kids to school on sedating medications can impact learning as well as social interaction, ultimately resulting in developmental delay.
Keep in mind that standard doses of such medications often are inadequate, and it is acceptable to work up to four times the dose, even in children, he said.
Triple-drug therapy, including H1 and H2 antagonists plus leukotriene blockers may be necessary.
“This is a very complicated and still important disease. The key is history – sometimes – and climbing the therapeutic ladder. These patients are really uncomfortable, and they will love you if you get this disease under control,” Dr. Friedman said.
One thing that might provide some comfort to the patients and their parents is the fact that urticaria in children does seem to have a point of remission. Parents often fear that their children will be plagued with urticaria for life, but a recent study of 92 patients showed that the remission rates at 1, 3, and 5 years were 18.5%, 54%, and 67.7% (J. Am. Acad. Dermatol. 2014;71:663-8).
The median duration of chronic urticaria was 4.3 years.
“That doesn’t sound great, but in considering one’s entire lifetime, it keeps things in perspective,” he said.
Dr. Friedman reported having no relevant disclosures.
ORLANDO – Acute urticaria in children is most often caused by infection, food, or medication, and a detailed history is imperative for improving the likelihood of identifying the culprit, according to Dr. Adam Friedman.
Mycoplasma is a particularly common infectious cause in children, but adenovirus, enterovirus, rotavirus, respiratory syncytial virus, Epstein-Barr virus, and cytomegalovirus also have been implicated in urticaria cases, Dr. Friedman, director of dermatologic research at the Albert Einstein College of Medicine, New York, said at the Orlando Dermatology Aesthetic and Clinical Conference.
With respect to foods, ask about intake of milk, eggs, peanuts, wheat, and soy. When it comes to prescription medications, antibiotics are an especially common cause.
“Nonsteroidal anti-inflammatory drugs are also a very important one. If you have a patient with a history of urticaria or a pediatric patient who has a history of urticaria of the mucosa, definitely educate the parents not to give them NSAIDs,” Dr. Friedman said.
Nonimmunologic direct mast cell activation also can be a source of acute urticaria, resulting from exposure to numerous products. These include, but are not limited to, polymyxin B, radiocontrast media, opiates, muscle relaxants, salicylates, and NSAIDs, all of which can “potentially induce urticaria in almost anyone,” he said.
Identifying the cause is more likely in acute case than in chronic cases, for which the etiology is unknown about 70% of time. Regardless, the good news is that about two-thirds of cases are self-limiting; it’s the other third that poses the greatest challenge, he said.
The best bet for nailing down a diagnosis and identifying the cause is an extensive history and physical evaluation.
“Ask a million and one questions to really get to the root of it. In some cases, chronic urticaria is really a diagnosis of exclusion,” he said.
Asking the patient to keep a diary to help identify any unusual exposures just prior to the episode, and having the patient or parent take pictures of the skin are shortcuts that can help.
Extensive laboratory testing is rarely helpful, but certain tests may be warranted. New European guidelines call for erythrocyte sedimentation rate/C-reactive protein and blood differential testing for chronic spontaneous urticaria, but others – like liver function tests, hepatitis B, antinuclear antibody, stool, urinalysis, thyroid function, and antithyroid antibodies – should be directed by the history. A complement panel may be useful in cases involving angioedema, and allergy skin testing may be warranted if a specific trigger can be implicated, but “don’t just order for the sake of ordering,” he said.
Biopsies are not typically useful except in suspected neutrophilic urticaria, which may indicate an association with autoimmune disease, as well as offer some insight into whether dapsone treatment would be helpful over other third-line therapies. Persistent cases of urticaria (lasting over 24-48 hours in one location) may suggest urticarial vasculitis, which would warrant a biopsy.
The therapeutic approach to urticaria involves educating patients about avoiding triggers and identifying and addressing underlying conditions, and using medications that address the pathophysiology of the disease (mast cells, histamine, etc).
In children, the guidelines are generally similar to those in adults, but there is a real push to avoid systemic steroids, Dr. Friedman said, noting that the only time he uses systemic steroids is as a bridge to get to the point where other therapies are beginning to take effect.
Other key concepts for managing urticaria in children, as published in 2013 (Acta. Derm. Venereol. 2013;93:500-8), include using second-generation histamine1 antihistamines for symptom relief, avoiding first-generation H1 antihistamines (due mainly to sedation), and using other therapeutic interventions only after carefully weighing risks and benefits, as evidence in children is lacking.
Nonsedating antihistamines are preferable, as sending kids to school on sedating medications can impact learning as well as social interaction, ultimately resulting in developmental delay.
Keep in mind that standard doses of such medications often are inadequate, and it is acceptable to work up to four times the dose, even in children, he said.
Triple-drug therapy, including H1 and H2 antagonists plus leukotriene blockers may be necessary.
“This is a very complicated and still important disease. The key is history – sometimes – and climbing the therapeutic ladder. These patients are really uncomfortable, and they will love you if you get this disease under control,” Dr. Friedman said.
One thing that might provide some comfort to the patients and their parents is the fact that urticaria in children does seem to have a point of remission. Parents often fear that their children will be plagued with urticaria for life, but a recent study of 92 patients showed that the remission rates at 1, 3, and 5 years were 18.5%, 54%, and 67.7% (J. Am. Acad. Dermatol. 2014;71:663-8).
The median duration of chronic urticaria was 4.3 years.
“That doesn’t sound great, but in considering one’s entire lifetime, it keeps things in perspective,” he said.
Dr. Friedman reported having no relevant disclosures.
ORLANDO – Acute urticaria in children is most often caused by infection, food, or medication, and a detailed history is imperative for improving the likelihood of identifying the culprit, according to Dr. Adam Friedman.
Mycoplasma is a particularly common infectious cause in children, but adenovirus, enterovirus, rotavirus, respiratory syncytial virus, Epstein-Barr virus, and cytomegalovirus also have been implicated in urticaria cases, Dr. Friedman, director of dermatologic research at the Albert Einstein College of Medicine, New York, said at the Orlando Dermatology Aesthetic and Clinical Conference.
With respect to foods, ask about intake of milk, eggs, peanuts, wheat, and soy. When it comes to prescription medications, antibiotics are an especially common cause.
“Nonsteroidal anti-inflammatory drugs are also a very important one. If you have a patient with a history of urticaria or a pediatric patient who has a history of urticaria of the mucosa, definitely educate the parents not to give them NSAIDs,” Dr. Friedman said.
Nonimmunologic direct mast cell activation also can be a source of acute urticaria, resulting from exposure to numerous products. These include, but are not limited to, polymyxin B, radiocontrast media, opiates, muscle relaxants, salicylates, and NSAIDs, all of which can “potentially induce urticaria in almost anyone,” he said.
Identifying the cause is more likely in acute case than in chronic cases, for which the etiology is unknown about 70% of time. Regardless, the good news is that about two-thirds of cases are self-limiting; it’s the other third that poses the greatest challenge, he said.
The best bet for nailing down a diagnosis and identifying the cause is an extensive history and physical evaluation.
“Ask a million and one questions to really get to the root of it. In some cases, chronic urticaria is really a diagnosis of exclusion,” he said.
Asking the patient to keep a diary to help identify any unusual exposures just prior to the episode, and having the patient or parent take pictures of the skin are shortcuts that can help.
Extensive laboratory testing is rarely helpful, but certain tests may be warranted. New European guidelines call for erythrocyte sedimentation rate/C-reactive protein and blood differential testing for chronic spontaneous urticaria, but others – like liver function tests, hepatitis B, antinuclear antibody, stool, urinalysis, thyroid function, and antithyroid antibodies – should be directed by the history. A complement panel may be useful in cases involving angioedema, and allergy skin testing may be warranted if a specific trigger can be implicated, but “don’t just order for the sake of ordering,” he said.
Biopsies are not typically useful except in suspected neutrophilic urticaria, which may indicate an association with autoimmune disease, as well as offer some insight into whether dapsone treatment would be helpful over other third-line therapies. Persistent cases of urticaria (lasting over 24-48 hours in one location) may suggest urticarial vasculitis, which would warrant a biopsy.
The therapeutic approach to urticaria involves educating patients about avoiding triggers and identifying and addressing underlying conditions, and using medications that address the pathophysiology of the disease (mast cells, histamine, etc).
In children, the guidelines are generally similar to those in adults, but there is a real push to avoid systemic steroids, Dr. Friedman said, noting that the only time he uses systemic steroids is as a bridge to get to the point where other therapies are beginning to take effect.
Other key concepts for managing urticaria in children, as published in 2013 (Acta. Derm. Venereol. 2013;93:500-8), include using second-generation histamine1 antihistamines for symptom relief, avoiding first-generation H1 antihistamines (due mainly to sedation), and using other therapeutic interventions only after carefully weighing risks and benefits, as evidence in children is lacking.
Nonsedating antihistamines are preferable, as sending kids to school on sedating medications can impact learning as well as social interaction, ultimately resulting in developmental delay.
Keep in mind that standard doses of such medications often are inadequate, and it is acceptable to work up to four times the dose, even in children, he said.
Triple-drug therapy, including H1 and H2 antagonists plus leukotriene blockers may be necessary.
“This is a very complicated and still important disease. The key is history – sometimes – and climbing the therapeutic ladder. These patients are really uncomfortable, and they will love you if you get this disease under control,” Dr. Friedman said.
One thing that might provide some comfort to the patients and their parents is the fact that urticaria in children does seem to have a point of remission. Parents often fear that their children will be plagued with urticaria for life, but a recent study of 92 patients showed that the remission rates at 1, 3, and 5 years were 18.5%, 54%, and 67.7% (J. Am. Acad. Dermatol. 2014;71:663-8).
The median duration of chronic urticaria was 4.3 years.
“That doesn’t sound great, but in considering one’s entire lifetime, it keeps things in perspective,” he said.
Dr. Friedman reported having no relevant disclosures.
EXPERT ANALYSIS FROM THE ODAC CONFERENCE
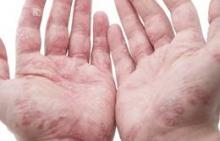
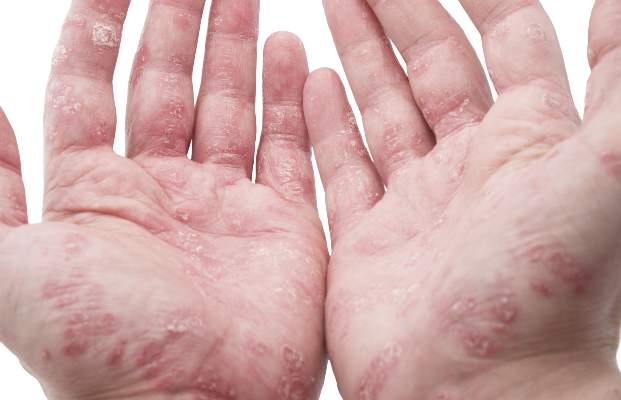
Survey: Psoriasis/Psoriatic Arthritis Undertreated
ORLANDO – Patients with psoriasis and psoriatic arthritis have a high level of dissatisfaction with current treatment options and have numerous unmet health care needs, according to the U.S. findings from the Multinational Assessment of Psoriasis and Psoriatic Arthritis (MAPP) survey.
Of 1,005 U.S. adults who participated in the population-based telephone survey, 735 had psoriasis diagnosed by a health care provider, and 270 had psoriatic arthritis diagnosed – with or without a diagnosis of psoriasis. Half of those surveyed reported dissatisfaction with available oral and biologic therapies, Dr. Mark Lebwohl of Mount Sinai Hospital, New York, and his colleagues reported in a poster at the Orlando Dermatology Aesthetic and Clinical Conference.
Most respondents with psoriasis reported moderate symptoms (40%), with about 30% each reporting mild or severe symptoms. Itching was the most bothersome symptom, occurring in about 42% of patients, followed by flaking (26%) and scales (22%). Itching was the greatest contributor to estimates of disease severity.
Psoriatic arthritis patients were more likely to report severe symptoms (56%), while 12% reported mild symptoms, and about a third reported moderate symptoms. Itching was the most bothersome symptom among these patients, followed by pain (21%) and flaking (19%). Pain and joint swelling were the greatest contributors to estimates of disease severity in this group, the investigators said.
The majority of respondents (57%) had seen a doctor in the past year. For psoriasis patients, that doctor was most often a dermatologist (57%), while psoriatic arthritis patients most often saw a rheumatologist (38%).
A third of patients most often saw a primary care physician.
The main reasons cited for not having seen a doctor in the past year were having mild or no symptoms, and a feeling that a health care provider would be unable to help.
Of concern, patterns reported by the patients indicated widespread undertreatment of psoriasis and psoriatic arthritis, they said.
More than one in five psoriasis patients (22%) reported no treatment at the time of the survey, and although most described their disease as moderate or severe, only 23% reported ever discussing the use of conventional oral or biologic therapies with their health care providers, and only 9% were currently receiving systemic therapy; most were using prescription topical therapy.
Psoriatic arthritis patients were much more likely than were psoriasis patients to have ever used a conventional oral or biologic therapy, but only 50% were receiving systemic therapy, while 26% reported use of biologic therapy and 24% reported use of an oral therapy.
Methotrexate was the conventional oral therapy used most often (68% of the time), followed by cyclosporine and acitretin (in 14% and 12% of patients, respectively), and etanercept was the biologic therapy used most often (56%) followed by adalimumab (used by 48%).
The most common reasons for discontinuing treatment were concerns about safety, issues with tolerability, and lack or loss of effectiveness. The greatest contributors to treatment burden among those using conventional oral therapies were side effects and the need for laboratory monitoring, and the greatest contributors among those using biologics were concerns about long-term safety, anxiety or fear, pain, and inconvenience associated with self injection.
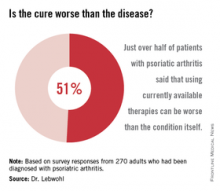
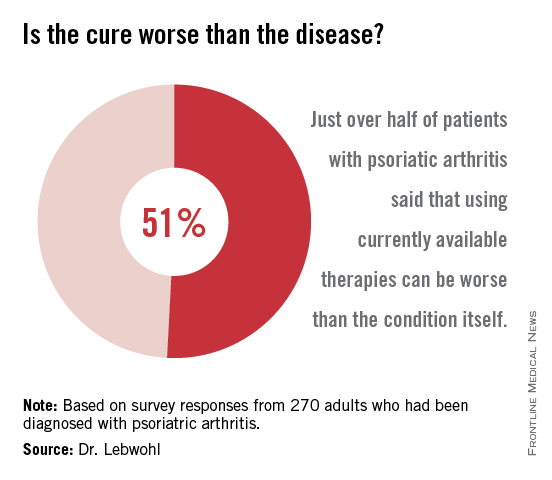
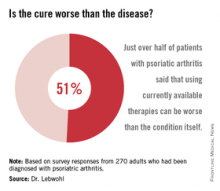
More than half of all respondents with psoriatic arthritis (51%) agreed that using currently available therapies can be worse than the condition itself, and 88% said better therapies are needed.
These results underscore the need for improved screening, as many patients with joint symptoms had not been diagnosed with psoriatic arthritis. They also demonstrate a need for improved assessment of disease severity; pruritus – which was reported as the most bothersome symptom – “is not included in most assessment tools,” the investigators noted.
Further, the results highlight the need for a higher level of treatment in many patients, they said, explaining that survey responses indicated substantial undertreatment in numerous cases. Many patients were receiving no treatment or only topical treatment, and many who were experiencing symptoms were not currently seeking care from a health care provider.
The MAPP survey is the first large-scale, multinational, population-based survey of psoriasis and psoriatic arthritis patients. The survey included 3,426 patients and 781 physicians in North America and Europe. The U.S. findings, which indicate little systematic implementation of treatment algorithms as well as widespread undertreatment, are particularly important given that psoriasis affects 3.2% of adults in the United States, and that 30% of those patients have psoriatic arthritis, the researchers noted.
Most of these patients report that their condition affects their emotional well-being, their quality of life, and their daily activities, they noted.
“These findings deserve attention and action to improve the care and lives of patients with psoriasis and psoriatic arthritis,” they concluded.
The survey was sponsored by Celgene, which markets apremilast (Otezla). Dr. Lebwohl disclosed ties to numerous pharmaceutical companies, including several that market drugs for psoriasis and psoriatic arthritis.
ORLANDO – Patients with psoriasis and psoriatic arthritis have a high level of dissatisfaction with current treatment options and have numerous unmet health care needs, according to the U.S. findings from the Multinational Assessment of Psoriasis and Psoriatic Arthritis (MAPP) survey.
Of 1,005 U.S. adults who participated in the population-based telephone survey, 735 had psoriasis diagnosed by a health care provider, and 270 had psoriatic arthritis diagnosed – with or without a diagnosis of psoriasis. Half of those surveyed reported dissatisfaction with available oral and biologic therapies, Dr. Mark Lebwohl of Mount Sinai Hospital, New York, and his colleagues reported in a poster at the Orlando Dermatology Aesthetic and Clinical Conference.
Most respondents with psoriasis reported moderate symptoms (40%), with about 30% each reporting mild or severe symptoms. Itching was the most bothersome symptom, occurring in about 42% of patients, followed by flaking (26%) and scales (22%). Itching was the greatest contributor to estimates of disease severity.
Psoriatic arthritis patients were more likely to report severe symptoms (56%), while 12% reported mild symptoms, and about a third reported moderate symptoms. Itching was the most bothersome symptom among these patients, followed by pain (21%) and flaking (19%). Pain and joint swelling were the greatest contributors to estimates of disease severity in this group, the investigators said.
The majority of respondents (57%) had seen a doctor in the past year. For psoriasis patients, that doctor was most often a dermatologist (57%), while psoriatic arthritis patients most often saw a rheumatologist (38%).
A third of patients most often saw a primary care physician.
The main reasons cited for not having seen a doctor in the past year were having mild or no symptoms, and a feeling that a health care provider would be unable to help.
Of concern, patterns reported by the patients indicated widespread undertreatment of psoriasis and psoriatic arthritis, they said.
More than one in five psoriasis patients (22%) reported no treatment at the time of the survey, and although most described their disease as moderate or severe, only 23% reported ever discussing the use of conventional oral or biologic therapies with their health care providers, and only 9% were currently receiving systemic therapy; most were using prescription topical therapy.
Psoriatic arthritis patients were much more likely than were psoriasis patients to have ever used a conventional oral or biologic therapy, but only 50% were receiving systemic therapy, while 26% reported use of biologic therapy and 24% reported use of an oral therapy.
Methotrexate was the conventional oral therapy used most often (68% of the time), followed by cyclosporine and acitretin (in 14% and 12% of patients, respectively), and etanercept was the biologic therapy used most often (56%) followed by adalimumab (used by 48%).
The most common reasons for discontinuing treatment were concerns about safety, issues with tolerability, and lack or loss of effectiveness. The greatest contributors to treatment burden among those using conventional oral therapies were side effects and the need for laboratory monitoring, and the greatest contributors among those using biologics were concerns about long-term safety, anxiety or fear, pain, and inconvenience associated with self injection.
More than half of all respondents with psoriatic arthritis (51%) agreed that using currently available therapies can be worse than the condition itself, and 88% said better therapies are needed.
These results underscore the need for improved screening, as many patients with joint symptoms had not been diagnosed with psoriatic arthritis. They also demonstrate a need for improved assessment of disease severity; pruritus – which was reported as the most bothersome symptom – “is not included in most assessment tools,” the investigators noted.
Further, the results highlight the need for a higher level of treatment in many patients, they said, explaining that survey responses indicated substantial undertreatment in numerous cases. Many patients were receiving no treatment or only topical treatment, and many who were experiencing symptoms were not currently seeking care from a health care provider.
The MAPP survey is the first large-scale, multinational, population-based survey of psoriasis and psoriatic arthritis patients. The survey included 3,426 patients and 781 physicians in North America and Europe. The U.S. findings, which indicate little systematic implementation of treatment algorithms as well as widespread undertreatment, are particularly important given that psoriasis affects 3.2% of adults in the United States, and that 30% of those patients have psoriatic arthritis, the researchers noted.
Most of these patients report that their condition affects their emotional well-being, their quality of life, and their daily activities, they noted.
“These findings deserve attention and action to improve the care and lives of patients with psoriasis and psoriatic arthritis,” they concluded.
The survey was sponsored by Celgene, which markets apremilast (Otezla). Dr. Lebwohl disclosed ties to numerous pharmaceutical companies, including several that market drugs for psoriasis and psoriatic arthritis.
ORLANDO – Patients with psoriasis and psoriatic arthritis have a high level of dissatisfaction with current treatment options and have numerous unmet health care needs, according to the U.S. findings from the Multinational Assessment of Psoriasis and Psoriatic Arthritis (MAPP) survey.
Of 1,005 U.S. adults who participated in the population-based telephone survey, 735 had psoriasis diagnosed by a health care provider, and 270 had psoriatic arthritis diagnosed – with or without a diagnosis of psoriasis. Half of those surveyed reported dissatisfaction with available oral and biologic therapies, Dr. Mark Lebwohl of Mount Sinai Hospital, New York, and his colleagues reported in a poster at the Orlando Dermatology Aesthetic and Clinical Conference.
Most respondents with psoriasis reported moderate symptoms (40%), with about 30% each reporting mild or severe symptoms. Itching was the most bothersome symptom, occurring in about 42% of patients, followed by flaking (26%) and scales (22%). Itching was the greatest contributor to estimates of disease severity.
Psoriatic arthritis patients were more likely to report severe symptoms (56%), while 12% reported mild symptoms, and about a third reported moderate symptoms. Itching was the most bothersome symptom among these patients, followed by pain (21%) and flaking (19%). Pain and joint swelling were the greatest contributors to estimates of disease severity in this group, the investigators said.
The majority of respondents (57%) had seen a doctor in the past year. For psoriasis patients, that doctor was most often a dermatologist (57%), while psoriatic arthritis patients most often saw a rheumatologist (38%).
A third of patients most often saw a primary care physician.
The main reasons cited for not having seen a doctor in the past year were having mild or no symptoms, and a feeling that a health care provider would be unable to help.
Of concern, patterns reported by the patients indicated widespread undertreatment of psoriasis and psoriatic arthritis, they said.
More than one in five psoriasis patients (22%) reported no treatment at the time of the survey, and although most described their disease as moderate or severe, only 23% reported ever discussing the use of conventional oral or biologic therapies with their health care providers, and only 9% were currently receiving systemic therapy; most were using prescription topical therapy.
Psoriatic arthritis patients were much more likely than were psoriasis patients to have ever used a conventional oral or biologic therapy, but only 50% were receiving systemic therapy, while 26% reported use of biologic therapy and 24% reported use of an oral therapy.
Methotrexate was the conventional oral therapy used most often (68% of the time), followed by cyclosporine and acitretin (in 14% and 12% of patients, respectively), and etanercept was the biologic therapy used most often (56%) followed by adalimumab (used by 48%).
The most common reasons for discontinuing treatment were concerns about safety, issues with tolerability, and lack or loss of effectiveness. The greatest contributors to treatment burden among those using conventional oral therapies were side effects and the need for laboratory monitoring, and the greatest contributors among those using biologics were concerns about long-term safety, anxiety or fear, pain, and inconvenience associated with self injection.
More than half of all respondents with psoriatic arthritis (51%) agreed that using currently available therapies can be worse than the condition itself, and 88% said better therapies are needed.
These results underscore the need for improved screening, as many patients with joint symptoms had not been diagnosed with psoriatic arthritis. They also demonstrate a need for improved assessment of disease severity; pruritus – which was reported as the most bothersome symptom – “is not included in most assessment tools,” the investigators noted.
Further, the results highlight the need for a higher level of treatment in many patients, they said, explaining that survey responses indicated substantial undertreatment in numerous cases. Many patients were receiving no treatment or only topical treatment, and many who were experiencing symptoms were not currently seeking care from a health care provider.
The MAPP survey is the first large-scale, multinational, population-based survey of psoriasis and psoriatic arthritis patients. The survey included 3,426 patients and 781 physicians in North America and Europe. The U.S. findings, which indicate little systematic implementation of treatment algorithms as well as widespread undertreatment, are particularly important given that psoriasis affects 3.2% of adults in the United States, and that 30% of those patients have psoriatic arthritis, the researchers noted.
Most of these patients report that their condition affects their emotional well-being, their quality of life, and their daily activities, they noted.
“These findings deserve attention and action to improve the care and lives of patients with psoriasis and psoriatic arthritis,” they concluded.
The survey was sponsored by Celgene, which markets apremilast (Otezla). Dr. Lebwohl disclosed ties to numerous pharmaceutical companies, including several that market drugs for psoriasis and psoriatic arthritis.
AT THE ODAC CONFERENCE
Survey: Psoriasis/psoriatic arthritis undertreated
ORLANDO – Patients with psoriasis and psoriatic arthritis have a high level of dissatisfaction with current treatment options and have numerous unmet health care needs, according to the U.S. findings from the Multinational Assessment of Psoriasis and Psoriatic Arthritis (MAPP) survey.
Of 1,005 U.S. adults who participated in the population-based telephone survey, 735 had psoriasis diagnosed by a health care provider, and 270 had psoriatic arthritis diagnosed – with or without a diagnosis of psoriasis. Half of those surveyed reported dissatisfaction with available oral and biologic therapies, Dr. Mark Lebwohl of Mount Sinai Hospital, New York, and his colleagues reported in a poster at the Orlando Dermatology Aesthetic and Clinical Conference.
Most respondents with psoriasis reported moderate symptoms (40%), with about 30% each reporting mild or severe symptoms. Itching was the most bothersome symptom, occurring in about 42% of patients, followed by flaking (26%) and scales (22%). Itching was the greatest contributor to estimates of disease severity.
Psoriatic arthritis patients were more likely to report severe symptoms (56%), while 12% reported mild symptoms, and about a third reported moderate symptoms. Itching was the most bothersome symptom among these patients, followed by pain (21%) and flaking (19%). Pain and joint swelling were the greatest contributors to estimates of disease severity in this group, the investigators said.
The majority of respondents (57%) had seen a doctor in the past year. For psoriasis patients, that doctor was most often a dermatologist (57%), while psoriatic arthritis patients most often saw a rheumatologist (38%).
A third of patients most often saw a primary care physician.
The main reasons cited for not having seen a doctor in the past year were having mild or no symptoms, and a feeling that a health care provider would be unable to help.
Of concern, patterns reported by the patients indicated widespread undertreatment of psoriasis and psoriatic arthritis, they said.
More than one in five psoriasis patients (22%) reported no treatment at the time of the survey, and although most described their disease as moderate or severe, only 23% reported ever discussing the use of conventional oral or biologic therapies with their health care providers, and only 9% were currently receiving systemic therapy; most were using prescription topical therapy.
Psoriatic arthritis patients were much more likely than were psoriasis patients to have ever used a conventional oral or biologic therapy, but only 50% were receiving systemic therapy, while 26% reported use of biologic therapy and 24% reported use of an oral therapy.
Methotrexate was the conventional oral therapy used most often (68% of the time), followed by cyclosporine and acitretin (in 14% and 12% of patients, respectively), and etanercept was the biologic therapy used most often (56%) followed by adalimumab (used by 48%).
The most common reasons for discontinuing treatment were concerns about safety, issues with tolerability, and lack or loss of effectiveness. The greatest contributors to treatment burden among those using conventional oral therapies were side effects and the need for laboratory monitoring, and the greatest contributors among those using biologics were concerns about long-term safety, anxiety or fear, pain, and inconvenience associated with self injection.
More than half of all respondents with psoriatic arthritis (51%) agreed that using currently available therapies can be worse than the condition itself, and 88% said better therapies are needed.
These results underscore the need for improved screening, as many patients with joint symptoms had not been diagnosed with psoriatic arthritis. They also demonstrate a need for improved assessment of disease severity; pruritus – which was reported as the most bothersome symptom – “is not included in most assessment tools,” the investigators noted.
Further, the results highlight the need for a higher level of treatment in many patients, they said, explaining that survey responses indicated substantial undertreatment in numerous cases. Many patients were receiving no treatment or only topical treatment, and many who were experiencing symptoms were not currently seeking care from a health care provider.
The MAPP survey is the first large-scale, multinational, population-based survey of psoriasis and psoriatic arthritis patients. The survey included 3,426 patients and 781 physicians in North America and Europe. The U.S. findings, which indicate little systematic implementation of treatment algorithms as well as widespread undertreatment, are particularly important given that psoriasis affects 3.2% of adults in the United States, and that 30% of those patients have psoriatic arthritis, the researchers noted.
Most of these patients report that their condition affects their emotional well-being, their quality of life, and their daily activities, they noted.
“These findings deserve attention and action to improve the care and lives of patients with psoriasis and psoriatic arthritis,” they concluded.
The survey was sponsored by Celgene, which markets apremilast (Otezla). Dr. Lebwohl disclosed ties to numerous pharmaceutical companies, including several that market drugs for psoriasis and psoriatic arthritis.
ORLANDO – Patients with psoriasis and psoriatic arthritis have a high level of dissatisfaction with current treatment options and have numerous unmet health care needs, according to the U.S. findings from the Multinational Assessment of Psoriasis and Psoriatic Arthritis (MAPP) survey.
Of 1,005 U.S. adults who participated in the population-based telephone survey, 735 had psoriasis diagnosed by a health care provider, and 270 had psoriatic arthritis diagnosed – with or without a diagnosis of psoriasis. Half of those surveyed reported dissatisfaction with available oral and biologic therapies, Dr. Mark Lebwohl of Mount Sinai Hospital, New York, and his colleagues reported in a poster at the Orlando Dermatology Aesthetic and Clinical Conference.
Most respondents with psoriasis reported moderate symptoms (40%), with about 30% each reporting mild or severe symptoms. Itching was the most bothersome symptom, occurring in about 42% of patients, followed by flaking (26%) and scales (22%). Itching was the greatest contributor to estimates of disease severity.
Psoriatic arthritis patients were more likely to report severe symptoms (56%), while 12% reported mild symptoms, and about a third reported moderate symptoms. Itching was the most bothersome symptom among these patients, followed by pain (21%) and flaking (19%). Pain and joint swelling were the greatest contributors to estimates of disease severity in this group, the investigators said.
The majority of respondents (57%) had seen a doctor in the past year. For psoriasis patients, that doctor was most often a dermatologist (57%), while psoriatic arthritis patients most often saw a rheumatologist (38%).
A third of patients most often saw a primary care physician.
The main reasons cited for not having seen a doctor in the past year were having mild or no symptoms, and a feeling that a health care provider would be unable to help.
Of concern, patterns reported by the patients indicated widespread undertreatment of psoriasis and psoriatic arthritis, they said.
More than one in five psoriasis patients (22%) reported no treatment at the time of the survey, and although most described their disease as moderate or severe, only 23% reported ever discussing the use of conventional oral or biologic therapies with their health care providers, and only 9% were currently receiving systemic therapy; most were using prescription topical therapy.
Psoriatic arthritis patients were much more likely than were psoriasis patients to have ever used a conventional oral or biologic therapy, but only 50% were receiving systemic therapy, while 26% reported use of biologic therapy and 24% reported use of an oral therapy.
Methotrexate was the conventional oral therapy used most often (68% of the time), followed by cyclosporine and acitretin (in 14% and 12% of patients, respectively), and etanercept was the biologic therapy used most often (56%) followed by adalimumab (used by 48%).
The most common reasons for discontinuing treatment were concerns about safety, issues with tolerability, and lack or loss of effectiveness. The greatest contributors to treatment burden among those using conventional oral therapies were side effects and the need for laboratory monitoring, and the greatest contributors among those using biologics were concerns about long-term safety, anxiety or fear, pain, and inconvenience associated with self injection.
More than half of all respondents with psoriatic arthritis (51%) agreed that using currently available therapies can be worse than the condition itself, and 88% said better therapies are needed.
These results underscore the need for improved screening, as many patients with joint symptoms had not been diagnosed with psoriatic arthritis. They also demonstrate a need for improved assessment of disease severity; pruritus – which was reported as the most bothersome symptom – “is not included in most assessment tools,” the investigators noted.
Further, the results highlight the need for a higher level of treatment in many patients, they said, explaining that survey responses indicated substantial undertreatment in numerous cases. Many patients were receiving no treatment or only topical treatment, and many who were experiencing symptoms were not currently seeking care from a health care provider.
The MAPP survey is the first large-scale, multinational, population-based survey of psoriasis and psoriatic arthritis patients. The survey included 3,426 patients and 781 physicians in North America and Europe. The U.S. findings, which indicate little systematic implementation of treatment algorithms as well as widespread undertreatment, are particularly important given that psoriasis affects 3.2% of adults in the United States, and that 30% of those patients have psoriatic arthritis, the researchers noted.
Most of these patients report that their condition affects their emotional well-being, their quality of life, and their daily activities, they noted.
“These findings deserve attention and action to improve the care and lives of patients with psoriasis and psoriatic arthritis,” they concluded.
The survey was sponsored by Celgene, which markets apremilast (Otezla). Dr. Lebwohl disclosed ties to numerous pharmaceutical companies, including several that market drugs for psoriasis and psoriatic arthritis.
ORLANDO – Patients with psoriasis and psoriatic arthritis have a high level of dissatisfaction with current treatment options and have numerous unmet health care needs, according to the U.S. findings from the Multinational Assessment of Psoriasis and Psoriatic Arthritis (MAPP) survey.
Of 1,005 U.S. adults who participated in the population-based telephone survey, 735 had psoriasis diagnosed by a health care provider, and 270 had psoriatic arthritis diagnosed – with or without a diagnosis of psoriasis. Half of those surveyed reported dissatisfaction with available oral and biologic therapies, Dr. Mark Lebwohl of Mount Sinai Hospital, New York, and his colleagues reported in a poster at the Orlando Dermatology Aesthetic and Clinical Conference.
Most respondents with psoriasis reported moderate symptoms (40%), with about 30% each reporting mild or severe symptoms. Itching was the most bothersome symptom, occurring in about 42% of patients, followed by flaking (26%) and scales (22%). Itching was the greatest contributor to estimates of disease severity.
Psoriatic arthritis patients were more likely to report severe symptoms (56%), while 12% reported mild symptoms, and about a third reported moderate symptoms. Itching was the most bothersome symptom among these patients, followed by pain (21%) and flaking (19%). Pain and joint swelling were the greatest contributors to estimates of disease severity in this group, the investigators said.
The majority of respondents (57%) had seen a doctor in the past year. For psoriasis patients, that doctor was most often a dermatologist (57%), while psoriatic arthritis patients most often saw a rheumatologist (38%).
A third of patients most often saw a primary care physician.
The main reasons cited for not having seen a doctor in the past year were having mild or no symptoms, and a feeling that a health care provider would be unable to help.
Of concern, patterns reported by the patients indicated widespread undertreatment of psoriasis and psoriatic arthritis, they said.
More than one in five psoriasis patients (22%) reported no treatment at the time of the survey, and although most described their disease as moderate or severe, only 23% reported ever discussing the use of conventional oral or biologic therapies with their health care providers, and only 9% were currently receiving systemic therapy; most were using prescription topical therapy.
Psoriatic arthritis patients were much more likely than were psoriasis patients to have ever used a conventional oral or biologic therapy, but only 50% were receiving systemic therapy, while 26% reported use of biologic therapy and 24% reported use of an oral therapy.
Methotrexate was the conventional oral therapy used most often (68% of the time), followed by cyclosporine and acitretin (in 14% and 12% of patients, respectively), and etanercept was the biologic therapy used most often (56%) followed by adalimumab (used by 48%).
The most common reasons for discontinuing treatment were concerns about safety, issues with tolerability, and lack or loss of effectiveness. The greatest contributors to treatment burden among those using conventional oral therapies were side effects and the need for laboratory monitoring, and the greatest contributors among those using biologics were concerns about long-term safety, anxiety or fear, pain, and inconvenience associated with self injection.
More than half of all respondents with psoriatic arthritis (51%) agreed that using currently available therapies can be worse than the condition itself, and 88% said better therapies are needed.
These results underscore the need for improved screening, as many patients with joint symptoms had not been diagnosed with psoriatic arthritis. They also demonstrate a need for improved assessment of disease severity; pruritus – which was reported as the most bothersome symptom – “is not included in most assessment tools,” the investigators noted.
Further, the results highlight the need for a higher level of treatment in many patients, they said, explaining that survey responses indicated substantial undertreatment in numerous cases. Many patients were receiving no treatment or only topical treatment, and many who were experiencing symptoms were not currently seeking care from a health care provider.
The MAPP survey is the first large-scale, multinational, population-based survey of psoriasis and psoriatic arthritis patients. The survey included 3,426 patients and 781 physicians in North America and Europe. The U.S. findings, which indicate little systematic implementation of treatment algorithms as well as widespread undertreatment, are particularly important given that psoriasis affects 3.2% of adults in the United States, and that 30% of those patients have psoriatic arthritis, the researchers noted.
Most of these patients report that their condition affects their emotional well-being, their quality of life, and their daily activities, they noted.
“These findings deserve attention and action to improve the care and lives of patients with psoriasis and psoriatic arthritis,” they concluded.
The survey was sponsored by Celgene, which markets apremilast (Otezla). Dr. Lebwohl disclosed ties to numerous pharmaceutical companies, including several that market drugs for psoriasis and psoriatic arthritis.
AT THE ODAC CONFERENCE
Key clinical point: Psoriasis and psoriatic arthritis treatment algorithms are underused, and patients are often undertreated.
Major finding: Among survey respondents, 22% were on no treatment, and 23% reported ever discussing conventional oral or biologic therapies with a health care provider.
Data source: A telephone survey (MAPP) of 735 psoriasis and 270 psoriatic arthritis patients.
Disclosures: The survey was sponsored by Celgene, which markets apremilast (Otezla). Dr. Lebwohl disclosed ties to numerous pharmaceutical companies, including several that market drugs for psoriasis and psoriatic arthritis.
Meta-analysis: Oseltamivir shortens time to flu relief
Oseltamivir treatment in adults with influenza shortens the time to clinical symptom alleviation by about 1 day and substantially reduces the risk of lower respiratory tract complications and hospitalization, according to findings from a meta-analysis of nine randomized, controlled trials involving 4,328 adult patients.
The findings of the study, which is the first to use individual patient data to evaluate the neuraminidase inhibitor, should put to rest persistent doubts its efficacy and safety, according to Dr. Arnold S. Monto of the University of Michigan School of Public Health, Ann Arbor, who was lead investigator, and his colleagues.
The intention-to-treat population of 1,591 patients with confirmed influenza had a significant 21% shorter time to clinical symptom alleviation, compared with the intention-to-treat infected population of 1,302 patients who received placebo (97.5 hours vs. 122.7 hours, difference of 25.2 hours; time ratio, 0.79), the investigators reported online Jan. 30 in the Lancet.
The effects were somewhat attenuated in the 2,402 treated patients in the overall intention-to-treat population – with a 15% reduction in time to symptom alleviation – compared with the 1,926 placebo patients in that population. But the difference remained significant (median of 17.8 hours; time ratio, 0.85), the investigators said (Lancet 2015 Jan. 30 [doi:10.106/S0140-6736(14)62449-1]).
“In the intention-to-treat noninfected population, the estimated time ratio was close to unity (time ratio, 0.99), so only participants identified as influenza infected benefited from oseltamivir,” the study authors wrote. That suggests efficacy is “confined to the antiviral activity of the drug.”
In addition, treatment significantly reduced the risk of lower respiratory tract complications occurring more than 48 hours after randomization by 44% in the intention-to-treat infected population. Only 4.2% of oseltamivir-treated patients requiring such treatment, compared with 8.7% of those who received placebo (risk ratio, 0.56). The risk of hospitalization for any cause was significantly reduced by 63% (0.6% vs. 1.7% in the groups, respectively; RR, 0.37).
The risk ratios for lower respiratory tract complications and hospitalization were 0.62 and 0.61, respectively, in the overall intention-to-treat population, but the difference in hospitalization between the groups was no longer statistically significant.
The benefits of treatment came at the cost of increased risk of nausea and vomiting (RR, 1.60 and 2.43, respectively), but no effect was seen with respect to neurologic or psychiatric disorders or serious adverse events in either of the groups.
To overcome previous concerns regarding potential publication bias, the investigators included all published and unpublished Roche-sponsored randomized, placebo-controlled, double-blind trials of the standard prescribed oseltamivir dose of 75 mg twice daily in adults, as well other applicable trials of the Roche drug for the treatment of naturally occurring influenzalike illness. The trials were conducted between 1997 and 2001, and included patients who were within 36 hours of feeling unwell, and who had a fever and at least one respiratory symptom and one additional constitutional symptom.
Treatment was administered at 12-hour intervals for 5 days, and patients were followed for 21 days, during which time virus cultures were performed in most cases.
“The safety and effectiveness of oseltamivir has been hotly debated, with some researchers claiming there is little evidence that oseltamivir works,” Dr. Monto said in a press statement. “Our meta-analysis provides compelling evidence that oseltamivir therapy reduces by 1 day the typical length of illness in adults infected with influenza and also prevents complications and reduces the number of people needing hospital treatment. Whether the magnitude of these benefits outweighs the harms of nausea and vomiting needs careful consideration.”
The Multiparty Group for Advice on Science funded the study and assembled a multidisciplinary team to examine available data. The group obtained an unrestricted grant from Roche to cover the costs.
Dr. Monto reported receiving fees from BioCryst and Roche outside of the submitted work. His coauthor, Dr. Richard J. Whitely, reported receiving fees as a board member of Gilead Sciences and travel funding from Roche. The remaining authors reported having no disclosures.
Oseltamivir treatment in adults with influenza shortens the time to clinical symptom alleviation by about 1 day and substantially reduces the risk of lower respiratory tract complications and hospitalization, according to findings from a meta-analysis of nine randomized, controlled trials involving 4,328 adult patients.
The findings of the study, which is the first to use individual patient data to evaluate the neuraminidase inhibitor, should put to rest persistent doubts its efficacy and safety, according to Dr. Arnold S. Monto of the University of Michigan School of Public Health, Ann Arbor, who was lead investigator, and his colleagues.
The intention-to-treat population of 1,591 patients with confirmed influenza had a significant 21% shorter time to clinical symptom alleviation, compared with the intention-to-treat infected population of 1,302 patients who received placebo (97.5 hours vs. 122.7 hours, difference of 25.2 hours; time ratio, 0.79), the investigators reported online Jan. 30 in the Lancet.
The effects were somewhat attenuated in the 2,402 treated patients in the overall intention-to-treat population – with a 15% reduction in time to symptom alleviation – compared with the 1,926 placebo patients in that population. But the difference remained significant (median of 17.8 hours; time ratio, 0.85), the investigators said (Lancet 2015 Jan. 30 [doi:10.106/S0140-6736(14)62449-1]).
“In the intention-to-treat noninfected population, the estimated time ratio was close to unity (time ratio, 0.99), so only participants identified as influenza infected benefited from oseltamivir,” the study authors wrote. That suggests efficacy is “confined to the antiviral activity of the drug.”
In addition, treatment significantly reduced the risk of lower respiratory tract complications occurring more than 48 hours after randomization by 44% in the intention-to-treat infected population. Only 4.2% of oseltamivir-treated patients requiring such treatment, compared with 8.7% of those who received placebo (risk ratio, 0.56). The risk of hospitalization for any cause was significantly reduced by 63% (0.6% vs. 1.7% in the groups, respectively; RR, 0.37).
The risk ratios for lower respiratory tract complications and hospitalization were 0.62 and 0.61, respectively, in the overall intention-to-treat population, but the difference in hospitalization between the groups was no longer statistically significant.
The benefits of treatment came at the cost of increased risk of nausea and vomiting (RR, 1.60 and 2.43, respectively), but no effect was seen with respect to neurologic or psychiatric disorders or serious adverse events in either of the groups.
To overcome previous concerns regarding potential publication bias, the investigators included all published and unpublished Roche-sponsored randomized, placebo-controlled, double-blind trials of the standard prescribed oseltamivir dose of 75 mg twice daily in adults, as well other applicable trials of the Roche drug for the treatment of naturally occurring influenzalike illness. The trials were conducted between 1997 and 2001, and included patients who were within 36 hours of feeling unwell, and who had a fever and at least one respiratory symptom and one additional constitutional symptom.
Treatment was administered at 12-hour intervals for 5 days, and patients were followed for 21 days, during which time virus cultures were performed in most cases.
“The safety and effectiveness of oseltamivir has been hotly debated, with some researchers claiming there is little evidence that oseltamivir works,” Dr. Monto said in a press statement. “Our meta-analysis provides compelling evidence that oseltamivir therapy reduces by 1 day the typical length of illness in adults infected with influenza and also prevents complications and reduces the number of people needing hospital treatment. Whether the magnitude of these benefits outweighs the harms of nausea and vomiting needs careful consideration.”
The Multiparty Group for Advice on Science funded the study and assembled a multidisciplinary team to examine available data. The group obtained an unrestricted grant from Roche to cover the costs.
Dr. Monto reported receiving fees from BioCryst and Roche outside of the submitted work. His coauthor, Dr. Richard J. Whitely, reported receiving fees as a board member of Gilead Sciences and travel funding from Roche. The remaining authors reported having no disclosures.
Oseltamivir treatment in adults with influenza shortens the time to clinical symptom alleviation by about 1 day and substantially reduces the risk of lower respiratory tract complications and hospitalization, according to findings from a meta-analysis of nine randomized, controlled trials involving 4,328 adult patients.
The findings of the study, which is the first to use individual patient data to evaluate the neuraminidase inhibitor, should put to rest persistent doubts its efficacy and safety, according to Dr. Arnold S. Monto of the University of Michigan School of Public Health, Ann Arbor, who was lead investigator, and his colleagues.
The intention-to-treat population of 1,591 patients with confirmed influenza had a significant 21% shorter time to clinical symptom alleviation, compared with the intention-to-treat infected population of 1,302 patients who received placebo (97.5 hours vs. 122.7 hours, difference of 25.2 hours; time ratio, 0.79), the investigators reported online Jan. 30 in the Lancet.
The effects were somewhat attenuated in the 2,402 treated patients in the overall intention-to-treat population – with a 15% reduction in time to symptom alleviation – compared with the 1,926 placebo patients in that population. But the difference remained significant (median of 17.8 hours; time ratio, 0.85), the investigators said (Lancet 2015 Jan. 30 [doi:10.106/S0140-6736(14)62449-1]).
“In the intention-to-treat noninfected population, the estimated time ratio was close to unity (time ratio, 0.99), so only participants identified as influenza infected benefited from oseltamivir,” the study authors wrote. That suggests efficacy is “confined to the antiviral activity of the drug.”
In addition, treatment significantly reduced the risk of lower respiratory tract complications occurring more than 48 hours after randomization by 44% in the intention-to-treat infected population. Only 4.2% of oseltamivir-treated patients requiring such treatment, compared with 8.7% of those who received placebo (risk ratio, 0.56). The risk of hospitalization for any cause was significantly reduced by 63% (0.6% vs. 1.7% in the groups, respectively; RR, 0.37).
The risk ratios for lower respiratory tract complications and hospitalization were 0.62 and 0.61, respectively, in the overall intention-to-treat population, but the difference in hospitalization between the groups was no longer statistically significant.
The benefits of treatment came at the cost of increased risk of nausea and vomiting (RR, 1.60 and 2.43, respectively), but no effect was seen with respect to neurologic or psychiatric disorders or serious adverse events in either of the groups.
To overcome previous concerns regarding potential publication bias, the investigators included all published and unpublished Roche-sponsored randomized, placebo-controlled, double-blind trials of the standard prescribed oseltamivir dose of 75 mg twice daily in adults, as well other applicable trials of the Roche drug for the treatment of naturally occurring influenzalike illness. The trials were conducted between 1997 and 2001, and included patients who were within 36 hours of feeling unwell, and who had a fever and at least one respiratory symptom and one additional constitutional symptom.
Treatment was administered at 12-hour intervals for 5 days, and patients were followed for 21 days, during which time virus cultures were performed in most cases.
“The safety and effectiveness of oseltamivir has been hotly debated, with some researchers claiming there is little evidence that oseltamivir works,” Dr. Monto said in a press statement. “Our meta-analysis provides compelling evidence that oseltamivir therapy reduces by 1 day the typical length of illness in adults infected with influenza and also prevents complications and reduces the number of people needing hospital treatment. Whether the magnitude of these benefits outweighs the harms of nausea and vomiting needs careful consideration.”
The Multiparty Group for Advice on Science funded the study and assembled a multidisciplinary team to examine available data. The group obtained an unrestricted grant from Roche to cover the costs.
Dr. Monto reported receiving fees from BioCryst and Roche outside of the submitted work. His coauthor, Dr. Richard J. Whitely, reported receiving fees as a board member of Gilead Sciences and travel funding from Roche. The remaining authors reported having no disclosures.
FROM THE LANCET
Key clinical point: Oseltamivir shortens the time to flu symptom alleviation, but nausea and vomiting are risks.
Major finding: The time to clinical symptom alleviation was about 25 hours shorter (21%) with oseltamivir vs. placebo.
Data source: An individual patient data meta-analysis encompassing nine studies involving 4,328 patients.
Disclosures: The Multiparty Group for Advice on Science funded the study and assembled a multidisciplinary team to examine available data. The group obtained an unrestricted grant from Roche to cover the costs. Dr. Monto reported receiving fees from Biocryst and Roche outside of the submitted work. His coauthor, Dr. Richard J. Whitely, reported receiving fees as a board member of Gilead Sciences and travel funding from Roche. The remaining authors reported having no disclosures.
rPFS may predict treatment response in mCRPC trials
A reproducible quantitative definition of radiographic progression-free survival in patients with metastatic castration-resistant prostate cancer was highly consistent and highly associated with overall survival in the Cougar-Abiraterone Acetate Study 302.
The findings could have implications for the interim measurement of treatment response in future studies, according to Dr. Michael J. Morris of Memorial Sloan Kettering Cancer Center, New York, and his colleagues.
Radiographic progression-free survival (rPFS) – defined in the Cougar-Abiraterone Acetate Study 302 (COU-AA-302) as the time from randomization to the first occurrence of progression by bone scan, progression by computed tomography or magnetic resonance imaging as defined by modified RECIST 1.0, or death resulting from any cause – was highly positively associated with overall survival in the randomized, placebo-controlled phase III study of 1,088 patients with metastatic castration-resistant prostate cancer (mCRPC) who were assigned to receive treatment with abiraterone plus prednisone or prednisone alone (Spearman’s correlation coefficient, 0.72), Dr. Morris and his colleagues reported online Jan. 26 in the Journal of Clinical Oncology.
Most men with mCRPC will succumb to the disease as a result of overwhelming osseous metastases; thus, approved treatments are prescribed to control or relieve pain and to delay or prevent skeletal-related events or death.
“There has long been a need to develop additional time-to-event endpoints short of [overall survival] to accelerate drug development,” the authors wrote, noting that the need is particularly urgent given the recent approval of numerous life-prolonging therapies for mCRPC (J. Clin. Oncol. 2015 Jan. 26 [doi:10.1200/JCO.2014.55.3875]).
A specific unmet need is a reproducible assay that can be interpreted and reported consistently and quantitatively as a biomarker in the assessment of bone disease with radionuclide bone scans, the investigators said.
A Prostate Cancer Working Group 2 (PCSG2) proposal to use a time-to-event progression endpoint for bone scan interpretation – with progression defined as two or more new lesions on an initial posttreatment bone scan, followed by two additional lesions on the subsequent scan – was evaluated along with a bone scan data capture assay developed through the Prostate Cancer Clinical Consortium in COU-AA-302.
Chemotherapy-naive patients were randomly assigned to receive 1,000 mg abiraterone plus prednisone daily or prednisone alone. At the first interim analysis for overall survival, independent review recorded 401 rPFS events. A 57% reduction in radiographic progression or death was seen in the abiraterone-treated group vs. the prednisone group (hazard ratio, 0.43); when rPFS was based on investigator-reviewed assessments of scans at that analysis, the decrease in the hazard of radiographic progression or death in the abiraterone group vs. the prednisone-only group was similar at 51% (hazard ratio, 0.49), the investigators reported.
At the second interim analysis for overall survival, 607 rPFS events were observed on investigator review. Treatment with abiraterone plus prednisone led to a 47% reduction in the risk of radiographic progression or death, compared with prednisone (hazard ratio, 0.53). This finding also closely matched the findings from the independent review at the first interim analysis.
Of note, the definition of radiographic progression used in the study prevented premature treatment discontinuation in 166 of 229 patients (72%) with two or more new lesions on their initial scan, because they did not develop two additional new lesions as of the subsequent scan, the investigators said.
The study – the first to use rPFS as a registration endpoint for evaluating chemotherapy-naive mCRPC patients per Food and Drug Administration approval of a supplemental new drug application – provides the highest level of evidence to date that rPFS is highly associated with overall survival in such patients, they said, noting that “the rigor of the independently validated data showing significant benefit in rPFS and a strong trend in [overall survival] as co-primary endpoints in combination with clinically relevant secondary endpoints” supported the regulatory approval of abiraterone acetate plus prednisone in chemotherapy-naive mCRPC patients.
“The results suggest that this objective, prospectively defined endpoint may serve as a response indicator biomarker that is evaluable in future studies,” they said, adding that while the findings demonstrate a highly positive association between rPFS and overall survival in mCRPC, they do not provide support for the use of rPFS as a substitute for overall survival.
This study was supported by Ortho Biotech Oncology Research and Development, the Prostate Cancer Clinical Trials Consortium, sponsored by the Department of Defense, and by Janssen Global Services, which provided writing assistance. Dr. Morris reported having no disclosures; his coauthors reported holding employment or leadership positions, serving in consultant or advisory roles, having stock ownership, and/or receiving honoraria or research funding from numerous pharmaceutical and other companies.
A reproducible quantitative definition of radiographic progression-free survival in patients with metastatic castration-resistant prostate cancer was highly consistent and highly associated with overall survival in the Cougar-Abiraterone Acetate Study 302.
The findings could have implications for the interim measurement of treatment response in future studies, according to Dr. Michael J. Morris of Memorial Sloan Kettering Cancer Center, New York, and his colleagues.
Radiographic progression-free survival (rPFS) – defined in the Cougar-Abiraterone Acetate Study 302 (COU-AA-302) as the time from randomization to the first occurrence of progression by bone scan, progression by computed tomography or magnetic resonance imaging as defined by modified RECIST 1.0, or death resulting from any cause – was highly positively associated with overall survival in the randomized, placebo-controlled phase III study of 1,088 patients with metastatic castration-resistant prostate cancer (mCRPC) who were assigned to receive treatment with abiraterone plus prednisone or prednisone alone (Spearman’s correlation coefficient, 0.72), Dr. Morris and his colleagues reported online Jan. 26 in the Journal of Clinical Oncology.
Most men with mCRPC will succumb to the disease as a result of overwhelming osseous metastases; thus, approved treatments are prescribed to control or relieve pain and to delay or prevent skeletal-related events or death.
“There has long been a need to develop additional time-to-event endpoints short of [overall survival] to accelerate drug development,” the authors wrote, noting that the need is particularly urgent given the recent approval of numerous life-prolonging therapies for mCRPC (J. Clin. Oncol. 2015 Jan. 26 [doi:10.1200/JCO.2014.55.3875]).
A specific unmet need is a reproducible assay that can be interpreted and reported consistently and quantitatively as a biomarker in the assessment of bone disease with radionuclide bone scans, the investigators said.
A Prostate Cancer Working Group 2 (PCSG2) proposal to use a time-to-event progression endpoint for bone scan interpretation – with progression defined as two or more new lesions on an initial posttreatment bone scan, followed by two additional lesions on the subsequent scan – was evaluated along with a bone scan data capture assay developed through the Prostate Cancer Clinical Consortium in COU-AA-302.
Chemotherapy-naive patients were randomly assigned to receive 1,000 mg abiraterone plus prednisone daily or prednisone alone. At the first interim analysis for overall survival, independent review recorded 401 rPFS events. A 57% reduction in radiographic progression or death was seen in the abiraterone-treated group vs. the prednisone group (hazard ratio, 0.43); when rPFS was based on investigator-reviewed assessments of scans at that analysis, the decrease in the hazard of radiographic progression or death in the abiraterone group vs. the prednisone-only group was similar at 51% (hazard ratio, 0.49), the investigators reported.
At the second interim analysis for overall survival, 607 rPFS events were observed on investigator review. Treatment with abiraterone plus prednisone led to a 47% reduction in the risk of radiographic progression or death, compared with prednisone (hazard ratio, 0.53). This finding also closely matched the findings from the independent review at the first interim analysis.
Of note, the definition of radiographic progression used in the study prevented premature treatment discontinuation in 166 of 229 patients (72%) with two or more new lesions on their initial scan, because they did not develop two additional new lesions as of the subsequent scan, the investigators said.
The study – the first to use rPFS as a registration endpoint for evaluating chemotherapy-naive mCRPC patients per Food and Drug Administration approval of a supplemental new drug application – provides the highest level of evidence to date that rPFS is highly associated with overall survival in such patients, they said, noting that “the rigor of the independently validated data showing significant benefit in rPFS and a strong trend in [overall survival] as co-primary endpoints in combination with clinically relevant secondary endpoints” supported the regulatory approval of abiraterone acetate plus prednisone in chemotherapy-naive mCRPC patients.
“The results suggest that this objective, prospectively defined endpoint may serve as a response indicator biomarker that is evaluable in future studies,” they said, adding that while the findings demonstrate a highly positive association between rPFS and overall survival in mCRPC, they do not provide support for the use of rPFS as a substitute for overall survival.
This study was supported by Ortho Biotech Oncology Research and Development, the Prostate Cancer Clinical Trials Consortium, sponsored by the Department of Defense, and by Janssen Global Services, which provided writing assistance. Dr. Morris reported having no disclosures; his coauthors reported holding employment or leadership positions, serving in consultant or advisory roles, having stock ownership, and/or receiving honoraria or research funding from numerous pharmaceutical and other companies.
A reproducible quantitative definition of radiographic progression-free survival in patients with metastatic castration-resistant prostate cancer was highly consistent and highly associated with overall survival in the Cougar-Abiraterone Acetate Study 302.
The findings could have implications for the interim measurement of treatment response in future studies, according to Dr. Michael J. Morris of Memorial Sloan Kettering Cancer Center, New York, and his colleagues.
Radiographic progression-free survival (rPFS) – defined in the Cougar-Abiraterone Acetate Study 302 (COU-AA-302) as the time from randomization to the first occurrence of progression by bone scan, progression by computed tomography or magnetic resonance imaging as defined by modified RECIST 1.0, or death resulting from any cause – was highly positively associated with overall survival in the randomized, placebo-controlled phase III study of 1,088 patients with metastatic castration-resistant prostate cancer (mCRPC) who were assigned to receive treatment with abiraterone plus prednisone or prednisone alone (Spearman’s correlation coefficient, 0.72), Dr. Morris and his colleagues reported online Jan. 26 in the Journal of Clinical Oncology.
Most men with mCRPC will succumb to the disease as a result of overwhelming osseous metastases; thus, approved treatments are prescribed to control or relieve pain and to delay or prevent skeletal-related events or death.
“There has long been a need to develop additional time-to-event endpoints short of [overall survival] to accelerate drug development,” the authors wrote, noting that the need is particularly urgent given the recent approval of numerous life-prolonging therapies for mCRPC (J. Clin. Oncol. 2015 Jan. 26 [doi:10.1200/JCO.2014.55.3875]).
A specific unmet need is a reproducible assay that can be interpreted and reported consistently and quantitatively as a biomarker in the assessment of bone disease with radionuclide bone scans, the investigators said.
A Prostate Cancer Working Group 2 (PCSG2) proposal to use a time-to-event progression endpoint for bone scan interpretation – with progression defined as two or more new lesions on an initial posttreatment bone scan, followed by two additional lesions on the subsequent scan – was evaluated along with a bone scan data capture assay developed through the Prostate Cancer Clinical Consortium in COU-AA-302.
Chemotherapy-naive patients were randomly assigned to receive 1,000 mg abiraterone plus prednisone daily or prednisone alone. At the first interim analysis for overall survival, independent review recorded 401 rPFS events. A 57% reduction in radiographic progression or death was seen in the abiraterone-treated group vs. the prednisone group (hazard ratio, 0.43); when rPFS was based on investigator-reviewed assessments of scans at that analysis, the decrease in the hazard of radiographic progression or death in the abiraterone group vs. the prednisone-only group was similar at 51% (hazard ratio, 0.49), the investigators reported.
At the second interim analysis for overall survival, 607 rPFS events were observed on investigator review. Treatment with abiraterone plus prednisone led to a 47% reduction in the risk of radiographic progression or death, compared with prednisone (hazard ratio, 0.53). This finding also closely matched the findings from the independent review at the first interim analysis.
Of note, the definition of radiographic progression used in the study prevented premature treatment discontinuation in 166 of 229 patients (72%) with two or more new lesions on their initial scan, because they did not develop two additional new lesions as of the subsequent scan, the investigators said.
The study – the first to use rPFS as a registration endpoint for evaluating chemotherapy-naive mCRPC patients per Food and Drug Administration approval of a supplemental new drug application – provides the highest level of evidence to date that rPFS is highly associated with overall survival in such patients, they said, noting that “the rigor of the independently validated data showing significant benefit in rPFS and a strong trend in [overall survival] as co-primary endpoints in combination with clinically relevant secondary endpoints” supported the regulatory approval of abiraterone acetate plus prednisone in chemotherapy-naive mCRPC patients.
“The results suggest that this objective, prospectively defined endpoint may serve as a response indicator biomarker that is evaluable in future studies,” they said, adding that while the findings demonstrate a highly positive association between rPFS and overall survival in mCRPC, they do not provide support for the use of rPFS as a substitute for overall survival.
This study was supported by Ortho Biotech Oncology Research and Development, the Prostate Cancer Clinical Trials Consortium, sponsored by the Department of Defense, and by Janssen Global Services, which provided writing assistance. Dr. Morris reported having no disclosures; his coauthors reported holding employment or leadership positions, serving in consultant or advisory roles, having stock ownership, and/or receiving honoraria or research funding from numerous pharmaceutical and other companies.
Key clinical point: Radiographic progression-free survival could serve as a valuable interim measure of treatment success in mCRPC trials.
Major finding: Radiographic progression-free survival was highly positively correlated with overall survival (Spearman’s correlation coefficient, 0.72).
Data source: A randomized, placebo-controlled phase III trial of 1,088 patients.
Disclosures: This study was supported by Ortho Biotech Oncology Research and Development, the Prostate Cancer Clinical Trials Consortium, sponsored by the Department of Defense, and by Janssen Global Services, which provided writing assistance. Dr. Morris reported having no disclosures; his coauthors reported holding employment or leadership positions, serving in consultant or advisory roles, having stock ownership, and/or receiving honoraria or research funding from numerous pharmaceutical and other companies.
Everolimus/letrozole combo provides clinical benefit in recurrent endometrial cancer
A treatment regimen designed to combat endocrine resistance in patients with recurrent endometrial cancer resulted in high clinical benefit and objective response rates in a phase II trial.
The clinical benefit rate in 35 women who were treated with the mammalian target of rapamycin (mTOR) inhibitor everolimus along with the aromatase inhibitor letrozole was 40% after a median of 15 cycles, and the confirmed objective response rate was 32%, Dr. Brian M. Slomovitz of the University of Texas MD Anderson Cancer Center, Houston, and his colleagues reported online Jan. 26 in the Journal of Clinical Oncology.
Most women with endometrial cancer (EC) are cured with surgery or with surgery and adjuvant therapy, but more than 8,000 die each year – largely as a result of resistance to conventional therapy, the investigators said.
Increased phosphoinositol-3 kinase (PI3K)/AKT/mTOR signaling has been linked with aggressive disease and poor prognosis, they noted.
Treatment with mTOR inhibitors such as everolimus has been shown in a number of studies to be of benefit for those with recurrent and/or metastatic endometrial cancer, and to have a favorable toxicity profile. Conversely, hormonal therapies in these patients have been associated with disappointing overall response rates.
However, given “the well-documented importance of estrogen receptor (ER) signaling as a driver of type I EC and cross-regulation between the ER and PI3K/AKT/mTOR pathways, synergistic antitumor effects might be achieved by combining PI3K/AKT/mTOR pathway inhibitors with agents that disrupt ER signaling,” the investigators wrote.
“We hypothesized that mTOR inhibition in combination with hormonal therapy may have an additive or synergistic effect and improve the overall response rates over either agent alone,” Dr. Slomovitz and his associates added, noting that success with everolimus and the aromatase inhibitor exemestane with respect to progression-free survival in patients with aromatase inhibitor–refractory breast cancer served as proof of concept.
Patients included in the study had a median age of 62 years and measurable disease that was considered incurable. They were enrolled between May 2010 and September 2011, and all had been treated with up to two prior cytotoxic regimens.
For the current study, they were treated with 4-week cycles of 10 mg oral everolimus and 2.5 mg oral letrozole daily until progression, dose-limiting toxicity, or complete response, and they were followed for a median of 14 months, the investigators said.
The clinical benefit rate was defined as complete response, partial response, or stable disease by RECIST 1.0 criteria: nine women achieved complete response and seven were taken off therapy at the discretion of their clinician after a prolonged complete response (J. Clin. Oncol. 2014 Jan. 26 [doi:10.1200/JCO.2014.58.3401]).
None of the patients discontinued treatment because of toxicity. The most common adverse events considered possibly, probably, or definitely associated with treatment were fatigue, hypertriglyceridemia, hypercholesterolemia, mucositis, anemia, hyperglycemia, and nausea, each occurring in 53%-74% of patients. Most were easily managed grade-1 or -2 toxicities.
Correlative studies, including mutational analysis, showed that the best predictor of lack of response was serous histology; endometrioid histology and CTNNB1 mutations appeared to confer a good response.
“Because of the different pathogenesis of serous cancers and differing serous-like molecular profile defined by the Cancer Genome Atlas uterine analysis, a different treatment response may not be surprising. Further analysis of patients with endometrioid tumors suggests heterogeneity even within one subtype. A single group of responders is difficult to define in patients with endometrioid EC in this study, but there are hints at multiple molecular subgroups that may benefit from everolimus/letrozole treatment,” Dr. Slomovitz and his coworkers wrote, noting that additional studies are essential to clearly define these subgroups, and that more comprehensive molecular profiling will be necessary to identify important drivers behind response and nonresponse.
Given the encouraging clinical benefit rate and the fact that the majority of complete responders in the current trial also had extended response duration similar to that seen in a Gynecologic Oncology Group trial of alternating courses of megestrol acetate and tamoxifen, a randomized comparison of that regimen and the current study regimen is planned.
This study was supported by grants from the Uterine Specialized Programs of Research Excellence, University of Texas MD Anderson Cancer, Novartis, and the Ann Rife Cox Chair in Gynecology. Dr. Slomovitz reported having no disclosures. His coauthors reported research funding from Amgen, Merck, Novartis, MedImmune, BiPar/sanofi-aventis, Abbott Laboratories, Johnson & Johnson, and Array BioPharma; patents/royalties/other intellectual property from Elsevier, Informa UK, and UpToDate; and consulting or advisory roles with Esperance Pharmaceuticals, Genentech/Roche, AstraZeneca, MedImmune, Merck, Johnson & Johnson, Clovis Oncology, Endocyte, and Astellas Pharma.
A treatment regimen designed to combat endocrine resistance in patients with recurrent endometrial cancer resulted in high clinical benefit and objective response rates in a phase II trial.
The clinical benefit rate in 35 women who were treated with the mammalian target of rapamycin (mTOR) inhibitor everolimus along with the aromatase inhibitor letrozole was 40% after a median of 15 cycles, and the confirmed objective response rate was 32%, Dr. Brian M. Slomovitz of the University of Texas MD Anderson Cancer Center, Houston, and his colleagues reported online Jan. 26 in the Journal of Clinical Oncology.
Most women with endometrial cancer (EC) are cured with surgery or with surgery and adjuvant therapy, but more than 8,000 die each year – largely as a result of resistance to conventional therapy, the investigators said.
Increased phosphoinositol-3 kinase (PI3K)/AKT/mTOR signaling has been linked with aggressive disease and poor prognosis, they noted.
Treatment with mTOR inhibitors such as everolimus has been shown in a number of studies to be of benefit for those with recurrent and/or metastatic endometrial cancer, and to have a favorable toxicity profile. Conversely, hormonal therapies in these patients have been associated with disappointing overall response rates.
However, given “the well-documented importance of estrogen receptor (ER) signaling as a driver of type I EC and cross-regulation between the ER and PI3K/AKT/mTOR pathways, synergistic antitumor effects might be achieved by combining PI3K/AKT/mTOR pathway inhibitors with agents that disrupt ER signaling,” the investigators wrote.
“We hypothesized that mTOR inhibition in combination with hormonal therapy may have an additive or synergistic effect and improve the overall response rates over either agent alone,” Dr. Slomovitz and his associates added, noting that success with everolimus and the aromatase inhibitor exemestane with respect to progression-free survival in patients with aromatase inhibitor–refractory breast cancer served as proof of concept.
Patients included in the study had a median age of 62 years and measurable disease that was considered incurable. They were enrolled between May 2010 and September 2011, and all had been treated with up to two prior cytotoxic regimens.
For the current study, they were treated with 4-week cycles of 10 mg oral everolimus and 2.5 mg oral letrozole daily until progression, dose-limiting toxicity, or complete response, and they were followed for a median of 14 months, the investigators said.
The clinical benefit rate was defined as complete response, partial response, or stable disease by RECIST 1.0 criteria: nine women achieved complete response and seven were taken off therapy at the discretion of their clinician after a prolonged complete response (J. Clin. Oncol. 2014 Jan. 26 [doi:10.1200/JCO.2014.58.3401]).
None of the patients discontinued treatment because of toxicity. The most common adverse events considered possibly, probably, or definitely associated with treatment were fatigue, hypertriglyceridemia, hypercholesterolemia, mucositis, anemia, hyperglycemia, and nausea, each occurring in 53%-74% of patients. Most were easily managed grade-1 or -2 toxicities.
Correlative studies, including mutational analysis, showed that the best predictor of lack of response was serous histology; endometrioid histology and CTNNB1 mutations appeared to confer a good response.
“Because of the different pathogenesis of serous cancers and differing serous-like molecular profile defined by the Cancer Genome Atlas uterine analysis, a different treatment response may not be surprising. Further analysis of patients with endometrioid tumors suggests heterogeneity even within one subtype. A single group of responders is difficult to define in patients with endometrioid EC in this study, but there are hints at multiple molecular subgroups that may benefit from everolimus/letrozole treatment,” Dr. Slomovitz and his coworkers wrote, noting that additional studies are essential to clearly define these subgroups, and that more comprehensive molecular profiling will be necessary to identify important drivers behind response and nonresponse.
Given the encouraging clinical benefit rate and the fact that the majority of complete responders in the current trial also had extended response duration similar to that seen in a Gynecologic Oncology Group trial of alternating courses of megestrol acetate and tamoxifen, a randomized comparison of that regimen and the current study regimen is planned.
This study was supported by grants from the Uterine Specialized Programs of Research Excellence, University of Texas MD Anderson Cancer, Novartis, and the Ann Rife Cox Chair in Gynecology. Dr. Slomovitz reported having no disclosures. His coauthors reported research funding from Amgen, Merck, Novartis, MedImmune, BiPar/sanofi-aventis, Abbott Laboratories, Johnson & Johnson, and Array BioPharma; patents/royalties/other intellectual property from Elsevier, Informa UK, and UpToDate; and consulting or advisory roles with Esperance Pharmaceuticals, Genentech/Roche, AstraZeneca, MedImmune, Merck, Johnson & Johnson, Clovis Oncology, Endocyte, and Astellas Pharma.
A treatment regimen designed to combat endocrine resistance in patients with recurrent endometrial cancer resulted in high clinical benefit and objective response rates in a phase II trial.
The clinical benefit rate in 35 women who were treated with the mammalian target of rapamycin (mTOR) inhibitor everolimus along with the aromatase inhibitor letrozole was 40% after a median of 15 cycles, and the confirmed objective response rate was 32%, Dr. Brian M. Slomovitz of the University of Texas MD Anderson Cancer Center, Houston, and his colleagues reported online Jan. 26 in the Journal of Clinical Oncology.
Most women with endometrial cancer (EC) are cured with surgery or with surgery and adjuvant therapy, but more than 8,000 die each year – largely as a result of resistance to conventional therapy, the investigators said.
Increased phosphoinositol-3 kinase (PI3K)/AKT/mTOR signaling has been linked with aggressive disease and poor prognosis, they noted.
Treatment with mTOR inhibitors such as everolimus has been shown in a number of studies to be of benefit for those with recurrent and/or metastatic endometrial cancer, and to have a favorable toxicity profile. Conversely, hormonal therapies in these patients have been associated with disappointing overall response rates.
However, given “the well-documented importance of estrogen receptor (ER) signaling as a driver of type I EC and cross-regulation between the ER and PI3K/AKT/mTOR pathways, synergistic antitumor effects might be achieved by combining PI3K/AKT/mTOR pathway inhibitors with agents that disrupt ER signaling,” the investigators wrote.
“We hypothesized that mTOR inhibition in combination with hormonal therapy may have an additive or synergistic effect and improve the overall response rates over either agent alone,” Dr. Slomovitz and his associates added, noting that success with everolimus and the aromatase inhibitor exemestane with respect to progression-free survival in patients with aromatase inhibitor–refractory breast cancer served as proof of concept.
Patients included in the study had a median age of 62 years and measurable disease that was considered incurable. They were enrolled between May 2010 and September 2011, and all had been treated with up to two prior cytotoxic regimens.
For the current study, they were treated with 4-week cycles of 10 mg oral everolimus and 2.5 mg oral letrozole daily until progression, dose-limiting toxicity, or complete response, and they were followed for a median of 14 months, the investigators said.
The clinical benefit rate was defined as complete response, partial response, or stable disease by RECIST 1.0 criteria: nine women achieved complete response and seven were taken off therapy at the discretion of their clinician after a prolonged complete response (J. Clin. Oncol. 2014 Jan. 26 [doi:10.1200/JCO.2014.58.3401]).
None of the patients discontinued treatment because of toxicity. The most common adverse events considered possibly, probably, or definitely associated with treatment were fatigue, hypertriglyceridemia, hypercholesterolemia, mucositis, anemia, hyperglycemia, and nausea, each occurring in 53%-74% of patients. Most were easily managed grade-1 or -2 toxicities.
Correlative studies, including mutational analysis, showed that the best predictor of lack of response was serous histology; endometrioid histology and CTNNB1 mutations appeared to confer a good response.
“Because of the different pathogenesis of serous cancers and differing serous-like molecular profile defined by the Cancer Genome Atlas uterine analysis, a different treatment response may not be surprising. Further analysis of patients with endometrioid tumors suggests heterogeneity even within one subtype. A single group of responders is difficult to define in patients with endometrioid EC in this study, but there are hints at multiple molecular subgroups that may benefit from everolimus/letrozole treatment,” Dr. Slomovitz and his coworkers wrote, noting that additional studies are essential to clearly define these subgroups, and that more comprehensive molecular profiling will be necessary to identify important drivers behind response and nonresponse.
Given the encouraging clinical benefit rate and the fact that the majority of complete responders in the current trial also had extended response duration similar to that seen in a Gynecologic Oncology Group trial of alternating courses of megestrol acetate and tamoxifen, a randomized comparison of that regimen and the current study regimen is planned.
This study was supported by grants from the Uterine Specialized Programs of Research Excellence, University of Texas MD Anderson Cancer, Novartis, and the Ann Rife Cox Chair in Gynecology. Dr. Slomovitz reported having no disclosures. His coauthors reported research funding from Amgen, Merck, Novartis, MedImmune, BiPar/sanofi-aventis, Abbott Laboratories, Johnson & Johnson, and Array BioPharma; patents/royalties/other intellectual property from Elsevier, Informa UK, and UpToDate; and consulting or advisory roles with Esperance Pharmaceuticals, Genentech/Roche, AstraZeneca, MedImmune, Merck, Johnson & Johnson, Clovis Oncology, Endocyte, and Astellas Pharma.
Key clinical point: Further development of an everolimus/letrozole combination is underway, given promising phase II trial results.
Major finding: The clinical benefit and objective response rates with everolimus plus letrozole were 40% and 32%, respectively.
Data source: A phase II study of 35 patients.
Disclosures: This study was supported by grants from the Uterine Specialized Programs of Research Excellence, University of Texas MD Anderson Cancer, Novartis, and the Ann Rife Cox Chair in Gynecology. Dr. Slomovitz reported having no disclosures. His coauthors reported research funding from Amgen, Merck, Novartis, MedImmune, BiPar/sanofi-aventis, Abbott Laboratories, Johnson & Johnson, and Array BioPharma; patents/royalties/other intellectual property from Elsevier, Informa UK, and UpToDate; and consulting or advisory roles with Esperance Pharmaceuticals, Genentech/Roche, AstraZeneca, MedImmune, Merck, Johnson & Johnson, Clovis Oncology, Endocyte, and Astellas Pharma.
Subungual Exostosis Masquerades as Nail Fungus
ORLANDO – In a patient presenting with a tender, firm lesion under the nail, think subungual exostosis, Dr. Phoebe Rich advised.
Subungual exostosis is a benign bony growth that can cause discomfort by pressing up from the nail bed. It can be mistaken for onychomycosis, but keep in mind that only 50% of those presenting with nail disorders will have onychomycosis, so it is important to consider other diagnoses, Dr. Rich, director of the Nail Disorder Center at Oregon Health and Science University, Portland, said at the Orlando Dermatology Aesthetic and Clinical Conference.
She described a teenager who presented with a growth under her nail, which the patient thought was nail fungus. The lesion developed after an injury experienced during a touch football game 2 months earlier.
An X-ray will aid in the diagnosis of such lesions, Dr. Rich said.
“You want to know that’s what it is … because you don’t want to just cut into it for a biopsy if you don’t know that it’s bone,” she said.
The diagnosis can be confirmed by removing the nail to take a look at the tumor.
“They are pretty characteristic. They are a bony growth. They’re actually composed of bone with a fibro-cartilaginous cap,” she said noting that tenderness is an important clue to the diagnosis.
Subungual exostosis occurs more often in toes than fingers, more often in girls than boys, and more often in children and young adults than older individuals, Dr. Rich noted.
Surgical removal is typically successful; the recurrence rate is about 10%, and recurrence is more common in children.
Presentation varies; some lesions can be quite obvious, with the growth protruding from under the nail, while others are more subtle. Dr. Rich described one young college student who presented with slight white spotting of the nail, and slight onycholysis. The patient had tenderness of the nail area when pressed, and the tumor peeked out from under the nail.
“If you see something like this, get an X-ray,” she said, adding: “You can be a hero and make the diagnosis. Don’t just treat it as though it’s a fungus.”
“If you’re very comfortable and expert at nails, great. Otherwise, make the diagnosis and send them to someone else, but you can really help these patients a lot,” she said.
Dr. Rich has participated in clinical trials with companies with antifungal/onychomycosis drugs, including Anacor, Merz, and Valeant.
ORLANDO – In a patient presenting with a tender, firm lesion under the nail, think subungual exostosis, Dr. Phoebe Rich advised.
Subungual exostosis is a benign bony growth that can cause discomfort by pressing up from the nail bed. It can be mistaken for onychomycosis, but keep in mind that only 50% of those presenting with nail disorders will have onychomycosis, so it is important to consider other diagnoses, Dr. Rich, director of the Nail Disorder Center at Oregon Health and Science University, Portland, said at the Orlando Dermatology Aesthetic and Clinical Conference.
She described a teenager who presented with a growth under her nail, which the patient thought was nail fungus. The lesion developed after an injury experienced during a touch football game 2 months earlier.
An X-ray will aid in the diagnosis of such lesions, Dr. Rich said.
“You want to know that’s what it is … because you don’t want to just cut into it for a biopsy if you don’t know that it’s bone,” she said.
The diagnosis can be confirmed by removing the nail to take a look at the tumor.
“They are pretty characteristic. They are a bony growth. They’re actually composed of bone with a fibro-cartilaginous cap,” she said noting that tenderness is an important clue to the diagnosis.
Subungual exostosis occurs more often in toes than fingers, more often in girls than boys, and more often in children and young adults than older individuals, Dr. Rich noted.
Surgical removal is typically successful; the recurrence rate is about 10%, and recurrence is more common in children.
Presentation varies; some lesions can be quite obvious, with the growth protruding from under the nail, while others are more subtle. Dr. Rich described one young college student who presented with slight white spotting of the nail, and slight onycholysis. The patient had tenderness of the nail area when pressed, and the tumor peeked out from under the nail.
“If you see something like this, get an X-ray,” she said, adding: “You can be a hero and make the diagnosis. Don’t just treat it as though it’s a fungus.”
“If you’re very comfortable and expert at nails, great. Otherwise, make the diagnosis and send them to someone else, but you can really help these patients a lot,” she said.
Dr. Rich has participated in clinical trials with companies with antifungal/onychomycosis drugs, including Anacor, Merz, and Valeant.
ORLANDO – In a patient presenting with a tender, firm lesion under the nail, think subungual exostosis, Dr. Phoebe Rich advised.
Subungual exostosis is a benign bony growth that can cause discomfort by pressing up from the nail bed. It can be mistaken for onychomycosis, but keep in mind that only 50% of those presenting with nail disorders will have onychomycosis, so it is important to consider other diagnoses, Dr. Rich, director of the Nail Disorder Center at Oregon Health and Science University, Portland, said at the Orlando Dermatology Aesthetic and Clinical Conference.
She described a teenager who presented with a growth under her nail, which the patient thought was nail fungus. The lesion developed after an injury experienced during a touch football game 2 months earlier.
An X-ray will aid in the diagnosis of such lesions, Dr. Rich said.
“You want to know that’s what it is … because you don’t want to just cut into it for a biopsy if you don’t know that it’s bone,” she said.
The diagnosis can be confirmed by removing the nail to take a look at the tumor.
“They are pretty characteristic. They are a bony growth. They’re actually composed of bone with a fibro-cartilaginous cap,” she said noting that tenderness is an important clue to the diagnosis.
Subungual exostosis occurs more often in toes than fingers, more often in girls than boys, and more often in children and young adults than older individuals, Dr. Rich noted.
Surgical removal is typically successful; the recurrence rate is about 10%, and recurrence is more common in children.
Presentation varies; some lesions can be quite obvious, with the growth protruding from under the nail, while others are more subtle. Dr. Rich described one young college student who presented with slight white spotting of the nail, and slight onycholysis. The patient had tenderness of the nail area when pressed, and the tumor peeked out from under the nail.
“If you see something like this, get an X-ray,” she said, adding: “You can be a hero and make the diagnosis. Don’t just treat it as though it’s a fungus.”
“If you’re very comfortable and expert at nails, great. Otherwise, make the diagnosis and send them to someone else, but you can really help these patients a lot,” she said.
Dr. Rich has participated in clinical trials with companies with antifungal/onychomycosis drugs, including Anacor, Merz, and Valeant.
EXPERT ANALYSIS AT THE ODAC CONFERENCE
Subungual exostosis masquerades as nail fungus
ORLANDO – In a patient presenting with a tender, firm lesion under the nail, think subungual exostosis, Dr. Phoebe Rich advised.
Subungual exostosis is a benign bony growth that can cause discomfort by pressing up from the nail bed. It can be mistaken for onychomycosis, but keep in mind that only 50% of those presenting with nail disorders will have onychomycosis, so it is important to consider other diagnoses, Dr. Rich, director of the Nail Disorder Center at Oregon Health and Science University, Portland, said at the Orlando Dermatology Aesthetic and Clinical Conference.
She described a teenager who presented with a growth under her nail, which the patient thought was nail fungus. The lesion developed after an injury experienced during a touch football game 2 months earlier.
An X-ray will aid in the diagnosis of such lesions, Dr. Rich said.
“You want to know that’s what it is … because you don’t want to just cut into it for a biopsy if you don’t know that it’s bone,” she said.
The diagnosis can be confirmed by removing the nail to take a look at the tumor.
“They are pretty characteristic. They are a bony growth. They’re actually composed of bone with a fibro-cartilaginous cap,” she said noting that tenderness is an important clue to the diagnosis.
Subungual exostosis occurs more often in toes than fingers, more often in girls than boys, and more often in children and young adults than older individuals, Dr. Rich noted.
Surgical removal is typically successful; the recurrence rate is about 10%, and recurrence is more common in children.
Presentation varies; some lesions can be quite obvious, with the growth protruding from under the nail, while others are more subtle. Dr. Rich described one young college student who presented with slight white spotting of the nail, and slight onycholysis. The patient had tenderness of the nail area when pressed, and the tumor peeked out from under the nail.
“If you see something like this, get an X-ray,” she said, adding: “You can be a hero and make the diagnosis. Don’t just treat it as though it’s a fungus.”
“If you’re very comfortable and expert at nails, great. Otherwise, make the diagnosis and send them to someone else, but you can really help these patients a lot,” she said.
Dr. Rich has participated in clinical trials with companies with antifungal/onychomycosis drugs, including Anacor, Merz, and Valeant.
ORLANDO – In a patient presenting with a tender, firm lesion under the nail, think subungual exostosis, Dr. Phoebe Rich advised.
Subungual exostosis is a benign bony growth that can cause discomfort by pressing up from the nail bed. It can be mistaken for onychomycosis, but keep in mind that only 50% of those presenting with nail disorders will have onychomycosis, so it is important to consider other diagnoses, Dr. Rich, director of the Nail Disorder Center at Oregon Health and Science University, Portland, said at the Orlando Dermatology Aesthetic and Clinical Conference.
She described a teenager who presented with a growth under her nail, which the patient thought was nail fungus. The lesion developed after an injury experienced during a touch football game 2 months earlier.
An X-ray will aid in the diagnosis of such lesions, Dr. Rich said.
“You want to know that’s what it is … because you don’t want to just cut into it for a biopsy if you don’t know that it’s bone,” she said.
The diagnosis can be confirmed by removing the nail to take a look at the tumor.
“They are pretty characteristic. They are a bony growth. They’re actually composed of bone with a fibro-cartilaginous cap,” she said noting that tenderness is an important clue to the diagnosis.
Subungual exostosis occurs more often in toes than fingers, more often in girls than boys, and more often in children and young adults than older individuals, Dr. Rich noted.
Surgical removal is typically successful; the recurrence rate is about 10%, and recurrence is more common in children.
Presentation varies; some lesions can be quite obvious, with the growth protruding from under the nail, while others are more subtle. Dr. Rich described one young college student who presented with slight white spotting of the nail, and slight onycholysis. The patient had tenderness of the nail area when pressed, and the tumor peeked out from under the nail.
“If you see something like this, get an X-ray,” she said, adding: “You can be a hero and make the diagnosis. Don’t just treat it as though it’s a fungus.”
“If you’re very comfortable and expert at nails, great. Otherwise, make the diagnosis and send them to someone else, but you can really help these patients a lot,” she said.
Dr. Rich has participated in clinical trials with companies with antifungal/onychomycosis drugs, including Anacor, Merz, and Valeant.
ORLANDO – In a patient presenting with a tender, firm lesion under the nail, think subungual exostosis, Dr. Phoebe Rich advised.
Subungual exostosis is a benign bony growth that can cause discomfort by pressing up from the nail bed. It can be mistaken for onychomycosis, but keep in mind that only 50% of those presenting with nail disorders will have onychomycosis, so it is important to consider other diagnoses, Dr. Rich, director of the Nail Disorder Center at Oregon Health and Science University, Portland, said at the Orlando Dermatology Aesthetic and Clinical Conference.
She described a teenager who presented with a growth under her nail, which the patient thought was nail fungus. The lesion developed after an injury experienced during a touch football game 2 months earlier.
An X-ray will aid in the diagnosis of such lesions, Dr. Rich said.
“You want to know that’s what it is … because you don’t want to just cut into it for a biopsy if you don’t know that it’s bone,” she said.
The diagnosis can be confirmed by removing the nail to take a look at the tumor.
“They are pretty characteristic. They are a bony growth. They’re actually composed of bone with a fibro-cartilaginous cap,” she said noting that tenderness is an important clue to the diagnosis.
Subungual exostosis occurs more often in toes than fingers, more often in girls than boys, and more often in children and young adults than older individuals, Dr. Rich noted.
Surgical removal is typically successful; the recurrence rate is about 10%, and recurrence is more common in children.
Presentation varies; some lesions can be quite obvious, with the growth protruding from under the nail, while others are more subtle. Dr. Rich described one young college student who presented with slight white spotting of the nail, and slight onycholysis. The patient had tenderness of the nail area when pressed, and the tumor peeked out from under the nail.
“If you see something like this, get an X-ray,” she said, adding: “You can be a hero and make the diagnosis. Don’t just treat it as though it’s a fungus.”
“If you’re very comfortable and expert at nails, great. Otherwise, make the diagnosis and send them to someone else, but you can really help these patients a lot,” she said.
Dr. Rich has participated in clinical trials with companies with antifungal/onychomycosis drugs, including Anacor, Merz, and Valeant.
EXPERT ANALYSIS AT THE ODAC CONFERENCE
Corticosteroid spray quickly, safely relieves psoriasis scaling
ORLANDO – A topical corticosteroid spray quickly and effectively reduced scaling in patients with plaque psoriasis, according to pooled phase III study results.
After 1 week of twice daily application of 0.25% Topicort spray (TaroPharma, Hawthorne, N.Y.), scaling was clear or almost clear in 29% of 120 treated patients, and was mild in an additional 40% of patients. At 4 weeks, scaling was clear or almost clear in 61% of patients, and mild in an additional 23%, Dr. Brian Keegan of East Windsor, N.J., reported in a poster at the Orlando Dermatology Aesthetic and Clinical Conference.
Additionally, 70% of patients experienced global improvement in psoriasis after 4 weeks of treatment; psoriasis was clear or almost clear in 42% of patients, and mild in 28%, Dr. Keegan said.
Study participants were adults with 10%-86% of body area affected by plaque psoriasis. Most of the patients (58%) were aged 41-to-64 years with a mean affected body surface area of 17%. Most had moderate scaling, but about a third of patients had severe or very severe scaling.
They applied Topicort 0.25% spray twice daily for 4 weeks.
The only adverse events that occurred in more than 1% of patients were dryness, irritation, and pruritus at the application site, which each occurred in 2%-3% of patients.
Treatment with Topicort – a class 1 super-potent corticosteroid indicated for up to 4 weeks of treatment of plaque psoriasis – quickly and safely relieved scaling in the majority of patients treated, Dr. Keegan concluded, noting that the findings are important given that scaling is very common among patients with plaque psoriasis, many of whom find it to be bothersome.
In fact, 94% of more than 17,000 participants in a National Psoriasis Foundation survey reported being bothered by scaling, he noted.
This study was sponsored by TaroPharma.
ORLANDO – A topical corticosteroid spray quickly and effectively reduced scaling in patients with plaque psoriasis, according to pooled phase III study results.
After 1 week of twice daily application of 0.25% Topicort spray (TaroPharma, Hawthorne, N.Y.), scaling was clear or almost clear in 29% of 120 treated patients, and was mild in an additional 40% of patients. At 4 weeks, scaling was clear or almost clear in 61% of patients, and mild in an additional 23%, Dr. Brian Keegan of East Windsor, N.J., reported in a poster at the Orlando Dermatology Aesthetic and Clinical Conference.
Additionally, 70% of patients experienced global improvement in psoriasis after 4 weeks of treatment; psoriasis was clear or almost clear in 42% of patients, and mild in 28%, Dr. Keegan said.
Study participants were adults with 10%-86% of body area affected by plaque psoriasis. Most of the patients (58%) were aged 41-to-64 years with a mean affected body surface area of 17%. Most had moderate scaling, but about a third of patients had severe or very severe scaling.
They applied Topicort 0.25% spray twice daily for 4 weeks.
The only adverse events that occurred in more than 1% of patients were dryness, irritation, and pruritus at the application site, which each occurred in 2%-3% of patients.
Treatment with Topicort – a class 1 super-potent corticosteroid indicated for up to 4 weeks of treatment of plaque psoriasis – quickly and safely relieved scaling in the majority of patients treated, Dr. Keegan concluded, noting that the findings are important given that scaling is very common among patients with plaque psoriasis, many of whom find it to be bothersome.
In fact, 94% of more than 17,000 participants in a National Psoriasis Foundation survey reported being bothered by scaling, he noted.
This study was sponsored by TaroPharma.
ORLANDO – A topical corticosteroid spray quickly and effectively reduced scaling in patients with plaque psoriasis, according to pooled phase III study results.
After 1 week of twice daily application of 0.25% Topicort spray (TaroPharma, Hawthorne, N.Y.), scaling was clear or almost clear in 29% of 120 treated patients, and was mild in an additional 40% of patients. At 4 weeks, scaling was clear or almost clear in 61% of patients, and mild in an additional 23%, Dr. Brian Keegan of East Windsor, N.J., reported in a poster at the Orlando Dermatology Aesthetic and Clinical Conference.
Additionally, 70% of patients experienced global improvement in psoriasis after 4 weeks of treatment; psoriasis was clear or almost clear in 42% of patients, and mild in 28%, Dr. Keegan said.
Study participants were adults with 10%-86% of body area affected by plaque psoriasis. Most of the patients (58%) were aged 41-to-64 years with a mean affected body surface area of 17%. Most had moderate scaling, but about a third of patients had severe or very severe scaling.
They applied Topicort 0.25% spray twice daily for 4 weeks.
The only adverse events that occurred in more than 1% of patients were dryness, irritation, and pruritus at the application site, which each occurred in 2%-3% of patients.
Treatment with Topicort – a class 1 super-potent corticosteroid indicated for up to 4 weeks of treatment of plaque psoriasis – quickly and safely relieved scaling in the majority of patients treated, Dr. Keegan concluded, noting that the findings are important given that scaling is very common among patients with plaque psoriasis, many of whom find it to be bothersome.
In fact, 94% of more than 17,000 participants in a National Psoriasis Foundation survey reported being bothered by scaling, he noted.
This study was sponsored by TaroPharma.
AT THE ODAC CONFERENCE
Key clinical point: Psoriasis patients with scaling are likely to respond to corticosteroid spray treatment.
Major finding: At 4 weeks, scaling was clear, almost clear, or mild in 84% of patients.
Data source: An analysis of pooled phase III trial data for 120 patients.
Disclosures: This study was sponsored by TaroPharma.