User login
Mitchel is a reporter for MDedge based in the Philadelphia area. He started with the company in 1992, when it was International Medical News Group (IMNG), and has since covered a range of medical specialties. Mitchel trained as a virologist at Roswell Park Memorial Institute in Buffalo, and then worked briefly as a researcher at Boston Children's Hospital before pivoting to journalism as a AAAS Mass Media Fellow in 1980. His first reporting job was with Science Digest magazine, and from the mid-1980s to early-1990s he was a reporter with Medical World News. @mitchelzoler
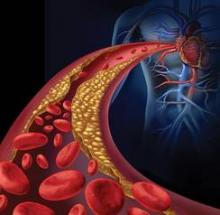
LCZ696 surpasses enalapril for heart failure
BARCELONA – A new, dual-agent formulation for treating heart failure patients with reduced ejection fraction showed a dramatic benefit for cutting the rate of cardiovascular death and hospitalizations in an international, controlled, pivotal trial with more than 8,000 patients.
The superiority of the new compound, which combines the novel neprilysin-inhibitor drug sacubitril with the established angiotensin receptor (ARB) blocking drug valsartan, was so strong that it immediately loomed as the new cornerstone-drug of choice for heart-failure patients with reduced ejection fraction pending regulatory approvals, said Dr. Milton Packer at the annual congress of the European Society of Cardiology.
Treatment with the new combination agent, currently known as LCZ696, doubled the cardiovascular-death benefit of the ACE inhibitor enalapril, while simultaneously edging out enalapril for safety and tolerability. Neprilysin is an endopeptidase that degrades several endogenous vasoactive peptides. Inhibiting this degradation process by treatment with LCZ696 boosts levels of these peptides and counters the neurohormonal overactivation that characterizes heart failure and leads to vasoconstriction, sodium retention, and maladaptive remodeling.
"This robust finding provides strong evidence supporting use of LCZ696 as the treatment of choice for heart failure instead of an ACE inhibitor or ARB" alone, said Dr. Packer, professor and chairman of clinical sciences at UT Southwestern Medical Center in Dallas. "The intent of this trial was to provide persuasive evidence that LCZ696 should replace current use of ACE inhibitors and ARBs as the cornerstone of heart failure treatment," said Dr. Packer, coprincipal investigator of the study.
Especially striking to several heart failure experts who heard the study results was the range of clinically important benefits seen when the new combination substituted for enalapril.
"This is the first study in a long time where we saw not only a reduction in the combined endpoint of cardiovascular death and hospitalization, but also significant effect on each of these two endpoints individually as well as on all-cause mortality," commented Dr. Mariell Jessup, professor of medicine at the University of Pennsylvania in Philadelphia. The new trial "was really trying to upset the foundation of heart failure treatment that we’ve used for the last 2 decades, and it succeeded. We were hoping for a positive outcome; what we got was remarkable. It very convincingly showed that this drug has incredible promise," she said in an interview.
"This will be an important change," commented Dr. John G.F. Cleland, professor of cardiology at the University of Hull, U.K. "The science and medicine will make it hard not to switch everyone with low ejection fraction heart failure [and other features that mirror the study’s enrollment criteria] from an ACE inhibitor to this the moment it becomes available," he said in an interview. "This is now the drug of choice for any heart failure patient with persistently elevated natriuretic peptide and left ventricular systolic dysfunction."
The Prospective Comparison of ARNI [angiotensin receptor–neprilysin inhibitor] with ACEI to Determine Impact on Global Mortality and Morbidity in Heart Failure (PARADIGM-HF) trial randomized 8,399 patients with heart failure and a left ventricular ejection fraction of 40% of less to treatment with either 200 mg b.i.d. of LCZ696 or 10 mg b.i.d. enalapril plus other standard heart failure medications, including a beta-blocker (in 93%), a diuretic (in 80%) and a mineralocorticoid antagonist such as spironolactone (in about 55%). About 70% of enrolled patients had New York Heart Association class II heart failure, and another 24% had class III heart failure.
After a median treatment duration of 27 months, the primary combined endpoint of cardiovascular death or first hospitalization for heart failure occurred in 22% of the patients treated with LCZ696 and in 27% of those treated with enalapril, a 20% relative risk reduction that was statistically significant, and which showed a number needed to treat of 21 to prevent one of these primary endpoints. LCZ696 treatment also linked with a significant reduction in all-cause death, and a statistically significant and clinically meaningful improvement in patient quality of life as measured using the Kansas City Cardiomyopathy Questionnaire after patients had been on treatment for 8 months. Online publication of the full results occurred concurrent with Dr. Packer’s report at the meeting (N.Engl. J. Med 2014;DOI: 10.1056/NEJMoa1409077).
While Dr. Packer highlighted the positive impact of LCZ696 on quality of life, he also stressed that the primary impact of treatment with LCZ696 is not to make patients feel better. "The major benefit of this drug is to change the natural course and rate of progression of heart failure. It should be used in patients with mild heart failure to reduce progression and death," he said.
"By switching patients from an ACE inhibitor to LCZ696 you can prolong patient survival. It is exciting to have a new drug" that can confer as many benefits as LCZ696, Dr. Jessup said.
PARADIGM-HF was sponsored by Novartis. Dr. Packer has been a consultant to Novartis as well as several other companies. Dr. Jessup had no disclosures. Dr. Cleland has received research support and honoraria from Sorin, St. Jude, and Medtronic.
Twitter: @mitchelzoler
PARADIGM-HF is the first contemporary trial to test the concept of substituting a new agent for an established heart-failure drug rather than adding a new compound to already established treatments. It was a large trial that enrolled patients with an elevated level of natriuretic peptide, used very relevant primary and secondary outcomes, and had a modest discontinuation rate during 27 months of treatment. The result showed a significant, 20% relative risk reduction in patients who were well treated for heart failure based on current guidelines, and the outcomes showed consistent results across all prespecified subgroups studied.
The investigational drug also showed good safety for renal function and potassium levels. Although patients who received LCZ696 had significantly more hypotensive episodes than patients in the enalapril comparator group, the investigational arm had no significant excess of treatment discontinuations. Patients who received LCZ696 also had a small increase in episodes of angioedema, but the difference did no reach statistical significance.
The trial used a clever run-in design that screened out patients who were intolerant of either enalapril or the study drug, a step that excluded 12% of the initial patient group.
The magnitude of the treatment effect and the number needed to treat suggests that LCZ696 will be a cost-effective treatment for the types of patients studied. Future studies should explore the efficacy of this drug combination in patients with heart failure with preserved ejection fraction, and in patients with acute decompensated heart failure, and a study should also compare LCZ696 with the safety and efficacy of a direct renin inhibitor such as aliskiren.
Michel Komajda, M.D., is professor of cardiology at University Pierre and Marie Curie in Paris, and is past president of the European Society of Cardiology. He has been a consultant to or speaker on behalf of Novartis and several other companies. He made these comments as the designated discussant for PARADIGM-HF.
PARADIGM-HF is the first contemporary trial to test the concept of substituting a new agent for an established heart-failure drug rather than adding a new compound to already established treatments. It was a large trial that enrolled patients with an elevated level of natriuretic peptide, used very relevant primary and secondary outcomes, and had a modest discontinuation rate during 27 months of treatment. The result showed a significant, 20% relative risk reduction in patients who were well treated for heart failure based on current guidelines, and the outcomes showed consistent results across all prespecified subgroups studied.
The investigational drug also showed good safety for renal function and potassium levels. Although patients who received LCZ696 had significantly more hypotensive episodes than patients in the enalapril comparator group, the investigational arm had no significant excess of treatment discontinuations. Patients who received LCZ696 also had a small increase in episodes of angioedema, but the difference did no reach statistical significance.
The trial used a clever run-in design that screened out patients who were intolerant of either enalapril or the study drug, a step that excluded 12% of the initial patient group.
The magnitude of the treatment effect and the number needed to treat suggests that LCZ696 will be a cost-effective treatment for the types of patients studied. Future studies should explore the efficacy of this drug combination in patients with heart failure with preserved ejection fraction, and in patients with acute decompensated heart failure, and a study should also compare LCZ696 with the safety and efficacy of a direct renin inhibitor such as aliskiren.
Michel Komajda, M.D., is professor of cardiology at University Pierre and Marie Curie in Paris, and is past president of the European Society of Cardiology. He has been a consultant to or speaker on behalf of Novartis and several other companies. He made these comments as the designated discussant for PARADIGM-HF.
PARADIGM-HF is the first contemporary trial to test the concept of substituting a new agent for an established heart-failure drug rather than adding a new compound to already established treatments. It was a large trial that enrolled patients with an elevated level of natriuretic peptide, used very relevant primary and secondary outcomes, and had a modest discontinuation rate during 27 months of treatment. The result showed a significant, 20% relative risk reduction in patients who were well treated for heart failure based on current guidelines, and the outcomes showed consistent results across all prespecified subgroups studied.
The investigational drug also showed good safety for renal function and potassium levels. Although patients who received LCZ696 had significantly more hypotensive episodes than patients in the enalapril comparator group, the investigational arm had no significant excess of treatment discontinuations. Patients who received LCZ696 also had a small increase in episodes of angioedema, but the difference did no reach statistical significance.
The trial used a clever run-in design that screened out patients who were intolerant of either enalapril or the study drug, a step that excluded 12% of the initial patient group.
The magnitude of the treatment effect and the number needed to treat suggests that LCZ696 will be a cost-effective treatment for the types of patients studied. Future studies should explore the efficacy of this drug combination in patients with heart failure with preserved ejection fraction, and in patients with acute decompensated heart failure, and a study should also compare LCZ696 with the safety and efficacy of a direct renin inhibitor such as aliskiren.
Michel Komajda, M.D., is professor of cardiology at University Pierre and Marie Curie in Paris, and is past president of the European Society of Cardiology. He has been a consultant to or speaker on behalf of Novartis and several other companies. He made these comments as the designated discussant for PARADIGM-HF.
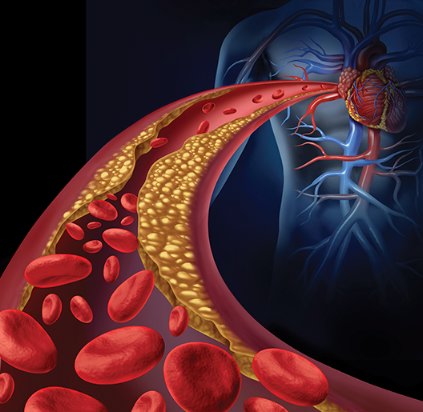
BARCELONA – A new, dual-agent formulation for treating heart failure patients with reduced ejection fraction showed a dramatic benefit for cutting the rate of cardiovascular death and hospitalizations in an international, controlled, pivotal trial with more than 8,000 patients.
The superiority of the new compound, which combines the novel neprilysin-inhibitor drug sacubitril with the established angiotensin receptor (ARB) blocking drug valsartan, was so strong that it immediately loomed as the new cornerstone-drug of choice for heart-failure patients with reduced ejection fraction pending regulatory approvals, said Dr. Milton Packer at the annual congress of the European Society of Cardiology.
Treatment with the new combination agent, currently known as LCZ696, doubled the cardiovascular-death benefit of the ACE inhibitor enalapril, while simultaneously edging out enalapril for safety and tolerability. Neprilysin is an endopeptidase that degrades several endogenous vasoactive peptides. Inhibiting this degradation process by treatment with LCZ696 boosts levels of these peptides and counters the neurohormonal overactivation that characterizes heart failure and leads to vasoconstriction, sodium retention, and maladaptive remodeling.
"This robust finding provides strong evidence supporting use of LCZ696 as the treatment of choice for heart failure instead of an ACE inhibitor or ARB" alone, said Dr. Packer, professor and chairman of clinical sciences at UT Southwestern Medical Center in Dallas. "The intent of this trial was to provide persuasive evidence that LCZ696 should replace current use of ACE inhibitors and ARBs as the cornerstone of heart failure treatment," said Dr. Packer, coprincipal investigator of the study.
Especially striking to several heart failure experts who heard the study results was the range of clinically important benefits seen when the new combination substituted for enalapril.
"This is the first study in a long time where we saw not only a reduction in the combined endpoint of cardiovascular death and hospitalization, but also significant effect on each of these two endpoints individually as well as on all-cause mortality," commented Dr. Mariell Jessup, professor of medicine at the University of Pennsylvania in Philadelphia. The new trial "was really trying to upset the foundation of heart failure treatment that we’ve used for the last 2 decades, and it succeeded. We were hoping for a positive outcome; what we got was remarkable. It very convincingly showed that this drug has incredible promise," she said in an interview.
"This will be an important change," commented Dr. John G.F. Cleland, professor of cardiology at the University of Hull, U.K. "The science and medicine will make it hard not to switch everyone with low ejection fraction heart failure [and other features that mirror the study’s enrollment criteria] from an ACE inhibitor to this the moment it becomes available," he said in an interview. "This is now the drug of choice for any heart failure patient with persistently elevated natriuretic peptide and left ventricular systolic dysfunction."
The Prospective Comparison of ARNI [angiotensin receptor–neprilysin inhibitor] with ACEI to Determine Impact on Global Mortality and Morbidity in Heart Failure (PARADIGM-HF) trial randomized 8,399 patients with heart failure and a left ventricular ejection fraction of 40% of less to treatment with either 200 mg b.i.d. of LCZ696 or 10 mg b.i.d. enalapril plus other standard heart failure medications, including a beta-blocker (in 93%), a diuretic (in 80%) and a mineralocorticoid antagonist such as spironolactone (in about 55%). About 70% of enrolled patients had New York Heart Association class II heart failure, and another 24% had class III heart failure.
After a median treatment duration of 27 months, the primary combined endpoint of cardiovascular death or first hospitalization for heart failure occurred in 22% of the patients treated with LCZ696 and in 27% of those treated with enalapril, a 20% relative risk reduction that was statistically significant, and which showed a number needed to treat of 21 to prevent one of these primary endpoints. LCZ696 treatment also linked with a significant reduction in all-cause death, and a statistically significant and clinically meaningful improvement in patient quality of life as measured using the Kansas City Cardiomyopathy Questionnaire after patients had been on treatment for 8 months. Online publication of the full results occurred concurrent with Dr. Packer’s report at the meeting (N.Engl. J. Med 2014;DOI: 10.1056/NEJMoa1409077).
While Dr. Packer highlighted the positive impact of LCZ696 on quality of life, he also stressed that the primary impact of treatment with LCZ696 is not to make patients feel better. "The major benefit of this drug is to change the natural course and rate of progression of heart failure. It should be used in patients with mild heart failure to reduce progression and death," he said.
"By switching patients from an ACE inhibitor to LCZ696 you can prolong patient survival. It is exciting to have a new drug" that can confer as many benefits as LCZ696, Dr. Jessup said.
PARADIGM-HF was sponsored by Novartis. Dr. Packer has been a consultant to Novartis as well as several other companies. Dr. Jessup had no disclosures. Dr. Cleland has received research support and honoraria from Sorin, St. Jude, and Medtronic.
Twitter: @mitchelzoler
BARCELONA – A new, dual-agent formulation for treating heart failure patients with reduced ejection fraction showed a dramatic benefit for cutting the rate of cardiovascular death and hospitalizations in an international, controlled, pivotal trial with more than 8,000 patients.
The superiority of the new compound, which combines the novel neprilysin-inhibitor drug sacubitril with the established angiotensin receptor (ARB) blocking drug valsartan, was so strong that it immediately loomed as the new cornerstone-drug of choice for heart-failure patients with reduced ejection fraction pending regulatory approvals, said Dr. Milton Packer at the annual congress of the European Society of Cardiology.
Treatment with the new combination agent, currently known as LCZ696, doubled the cardiovascular-death benefit of the ACE inhibitor enalapril, while simultaneously edging out enalapril for safety and tolerability. Neprilysin is an endopeptidase that degrades several endogenous vasoactive peptides. Inhibiting this degradation process by treatment with LCZ696 boosts levels of these peptides and counters the neurohormonal overactivation that characterizes heart failure and leads to vasoconstriction, sodium retention, and maladaptive remodeling.
"This robust finding provides strong evidence supporting use of LCZ696 as the treatment of choice for heart failure instead of an ACE inhibitor or ARB" alone, said Dr. Packer, professor and chairman of clinical sciences at UT Southwestern Medical Center in Dallas. "The intent of this trial was to provide persuasive evidence that LCZ696 should replace current use of ACE inhibitors and ARBs as the cornerstone of heart failure treatment," said Dr. Packer, coprincipal investigator of the study.
Especially striking to several heart failure experts who heard the study results was the range of clinically important benefits seen when the new combination substituted for enalapril.
"This is the first study in a long time where we saw not only a reduction in the combined endpoint of cardiovascular death and hospitalization, but also significant effect on each of these two endpoints individually as well as on all-cause mortality," commented Dr. Mariell Jessup, professor of medicine at the University of Pennsylvania in Philadelphia. The new trial "was really trying to upset the foundation of heart failure treatment that we’ve used for the last 2 decades, and it succeeded. We were hoping for a positive outcome; what we got was remarkable. It very convincingly showed that this drug has incredible promise," she said in an interview.
"This will be an important change," commented Dr. John G.F. Cleland, professor of cardiology at the University of Hull, U.K. "The science and medicine will make it hard not to switch everyone with low ejection fraction heart failure [and other features that mirror the study’s enrollment criteria] from an ACE inhibitor to this the moment it becomes available," he said in an interview. "This is now the drug of choice for any heart failure patient with persistently elevated natriuretic peptide and left ventricular systolic dysfunction."
The Prospective Comparison of ARNI [angiotensin receptor–neprilysin inhibitor] with ACEI to Determine Impact on Global Mortality and Morbidity in Heart Failure (PARADIGM-HF) trial randomized 8,399 patients with heart failure and a left ventricular ejection fraction of 40% of less to treatment with either 200 mg b.i.d. of LCZ696 or 10 mg b.i.d. enalapril plus other standard heart failure medications, including a beta-blocker (in 93%), a diuretic (in 80%) and a mineralocorticoid antagonist such as spironolactone (in about 55%). About 70% of enrolled patients had New York Heart Association class II heart failure, and another 24% had class III heart failure.
After a median treatment duration of 27 months, the primary combined endpoint of cardiovascular death or first hospitalization for heart failure occurred in 22% of the patients treated with LCZ696 and in 27% of those treated with enalapril, a 20% relative risk reduction that was statistically significant, and which showed a number needed to treat of 21 to prevent one of these primary endpoints. LCZ696 treatment also linked with a significant reduction in all-cause death, and a statistically significant and clinically meaningful improvement in patient quality of life as measured using the Kansas City Cardiomyopathy Questionnaire after patients had been on treatment for 8 months. Online publication of the full results occurred concurrent with Dr. Packer’s report at the meeting (N.Engl. J. Med 2014;DOI: 10.1056/NEJMoa1409077).
While Dr. Packer highlighted the positive impact of LCZ696 on quality of life, he also stressed that the primary impact of treatment with LCZ696 is not to make patients feel better. "The major benefit of this drug is to change the natural course and rate of progression of heart failure. It should be used in patients with mild heart failure to reduce progression and death," he said.
"By switching patients from an ACE inhibitor to LCZ696 you can prolong patient survival. It is exciting to have a new drug" that can confer as many benefits as LCZ696, Dr. Jessup said.
PARADIGM-HF was sponsored by Novartis. Dr. Packer has been a consultant to Novartis as well as several other companies. Dr. Jessup had no disclosures. Dr. Cleland has received research support and honoraria from Sorin, St. Jude, and Medtronic.
Twitter: @mitchelzoler
AT THE ESC CONGRESS 2014
Key clinical point: Substituting LCZ696 for enalapril in heart failure patients was linked with significant improvements in survival, heart failure hospitalization, and quality of life.
Major finding: LCZ696 was associated with a 20% relative risk reduction in cardiovascular death and heart-failure hospitalization compared with enalapril in heart failure patients.
Data source: PARADIGM-HF, which assessed 8,399 patients randomized at 1,043 centers in 47 countries.
Disclosures: PARADIGM-HF was sponsored by Novartis. Dr. Packer has been a consultant to Novartis as well as several other companies. Dr. Jessup had no disclosures. Dr. Cleland has received research support and honoraria from Sorin, St. Jude, and Medtronic.
VIDEO: New dual heart-failure formulation scores several benefits
BARCELONA – It’s been a while since physicians had a new treatment for heart failure patients with reduced ejection fraction that makes them feel better, stay out of hospital, and live longer. But the investigational drug LCZ696 did just that in PARADIGM-HF, a controlled, pivotal trial with more than 8,000 patients, Dr. John McMurray said at the annual congress of the European Society of Cardiology.
In addition to robustly beating the comparator drug enalapril with reduced rates of both cardiovascular deaths and the combined endpoint of cardiovascular deaths and heart-failure hospitalizations, the new LCZ696 combination drug boosted patients’ quality of life in several clinically meaningful ways. The magnitude of the quality of life effect matched that seen in prior, successful, heart-failure treatment trials, Dr. McMurray, a professor of medical cardiology at the University of Glasgow, said in an interview.
He stressed that the combination of sacubitril, a new drug that inhibits neprilysin and thereby boosts levels of endogenous vasoactive peptides, and the well-established angiotensin-receptor blocker valsartan helped patients with milder, New York Heart Association class II heart failure, roughly 70% of enrolled patients. Activity in these less severely ill patients means that treatment with LCZ696 can "keep these patients stable, feeling well, functional, and out of the hospital as well as improve their survival," he said.
PARADIGM-HF was sponsored by Novartis, the company developing LCZ696. Dr. McMurray said that he has received travel support from Novartis.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Twitter: @mitchelzoler
BARCELONA – It’s been a while since physicians had a new treatment for heart failure patients with reduced ejection fraction that makes them feel better, stay out of hospital, and live longer. But the investigational drug LCZ696 did just that in PARADIGM-HF, a controlled, pivotal trial with more than 8,000 patients, Dr. John McMurray said at the annual congress of the European Society of Cardiology.
In addition to robustly beating the comparator drug enalapril with reduced rates of both cardiovascular deaths and the combined endpoint of cardiovascular deaths and heart-failure hospitalizations, the new LCZ696 combination drug boosted patients’ quality of life in several clinically meaningful ways. The magnitude of the quality of life effect matched that seen in prior, successful, heart-failure treatment trials, Dr. McMurray, a professor of medical cardiology at the University of Glasgow, said in an interview.
He stressed that the combination of sacubitril, a new drug that inhibits neprilysin and thereby boosts levels of endogenous vasoactive peptides, and the well-established angiotensin-receptor blocker valsartan helped patients with milder, New York Heart Association class II heart failure, roughly 70% of enrolled patients. Activity in these less severely ill patients means that treatment with LCZ696 can "keep these patients stable, feeling well, functional, and out of the hospital as well as improve their survival," he said.
PARADIGM-HF was sponsored by Novartis, the company developing LCZ696. Dr. McMurray said that he has received travel support from Novartis.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Twitter: @mitchelzoler
BARCELONA – It’s been a while since physicians had a new treatment for heart failure patients with reduced ejection fraction that makes them feel better, stay out of hospital, and live longer. But the investigational drug LCZ696 did just that in PARADIGM-HF, a controlled, pivotal trial with more than 8,000 patients, Dr. John McMurray said at the annual congress of the European Society of Cardiology.
In addition to robustly beating the comparator drug enalapril with reduced rates of both cardiovascular deaths and the combined endpoint of cardiovascular deaths and heart-failure hospitalizations, the new LCZ696 combination drug boosted patients’ quality of life in several clinically meaningful ways. The magnitude of the quality of life effect matched that seen in prior, successful, heart-failure treatment trials, Dr. McMurray, a professor of medical cardiology at the University of Glasgow, said in an interview.
He stressed that the combination of sacubitril, a new drug that inhibits neprilysin and thereby boosts levels of endogenous vasoactive peptides, and the well-established angiotensin-receptor blocker valsartan helped patients with milder, New York Heart Association class II heart failure, roughly 70% of enrolled patients. Activity in these less severely ill patients means that treatment with LCZ696 can "keep these patients stable, feeling well, functional, and out of the hospital as well as improve their survival," he said.
PARADIGM-HF was sponsored by Novartis, the company developing LCZ696. Dr. McMurray said that he has received travel support from Novartis.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Twitter: @mitchelzoler
AT ESC 2014
Estrogen-containing OCs cloud measurement of ovarian reserve
MUNICH – Oral contraceptives do more than prevent unwanted pregnancy. They also make it hard to gauge a woman’s ovarian reserve, based on data from 833 women aged 19-46 years seen at a single Danish fertility clinic.
Study findings suggested that an accurate measure of a woman’s ovarian reserve can occur only after she has been off an estrogen-containing OC, probably for at least 3 months, Dr. Kathrine Birch Petersen reported at the annual meeting of the European Society of Human Reproduction and Embryology.
The impact of estrogen-containing OC use on reducing ovarian volume was especially pronounced in women under age 30, the reduction increased with longer durations of OC use, and the ability of OCs to mask a woman’s actual ovarian reserve was strong enough to potentially conceal a true case of premature ovarian insufficiency, said Dr. Birch Petersen, an ob.gyn. at the Fertility Assessment and Counseling Clinic at Rigshospitalet in Copenhagen.
"When we see a woman on an OC with impaired ovarian reserve, we would presume [based on these new findings] that her real ovarian reserve was about 30% higher than what we measure. We would advise her to be retested after she was off her OC for about 3 months," Dr. Birch Petersen said during a press conference before her presentation at the meeting.
The study included the first women seen at the clinic since it opened in 2011, excluding those who were pregnant or failed to supply adequate information. The cross-sectional cohort included 240 women on an estrogen-containing OC and 593 women with natural cycles.
The analysis focused on three parameters: blood level of anti-Müllerian hormone (AMH), antral follicle count (AFC), and ovarian volume. The multivariate, linear regression analysis adjusted for age, body mass index, smoking, age of maternal menopause, maternal smoking during pregnancy, preterm birth, and duration of OC use.
The analysis showed that compared with the women with natural cycles, those on an OC had a 19% relative reduction in their average blood level of AMH, a 16% relative reduction in average AFC, and a 47% relative reduction in average ovarian volume. The women on an OC also had smaller antral follicles. All three differences were statistically significant.
Seeing an effect from an estrogen-containing OC on all three measures makes sense because of their interrelatedness. The antral follicles produce AMH, and a reduction in antral follicle number as well as size would shrink the ovarian contents and result in reduced volume. These effects would not occur in women on a progestin-only OC, she said.
Dr. Birch Petersen had no disclosures.
On Twitter @mitchelzoler
MUNICH – Oral contraceptives do more than prevent unwanted pregnancy. They also make it hard to gauge a woman’s ovarian reserve, based on data from 833 women aged 19-46 years seen at a single Danish fertility clinic.
Study findings suggested that an accurate measure of a woman’s ovarian reserve can occur only after she has been off an estrogen-containing OC, probably for at least 3 months, Dr. Kathrine Birch Petersen reported at the annual meeting of the European Society of Human Reproduction and Embryology.
The impact of estrogen-containing OC use on reducing ovarian volume was especially pronounced in women under age 30, the reduction increased with longer durations of OC use, and the ability of OCs to mask a woman’s actual ovarian reserve was strong enough to potentially conceal a true case of premature ovarian insufficiency, said Dr. Birch Petersen, an ob.gyn. at the Fertility Assessment and Counseling Clinic at Rigshospitalet in Copenhagen.
"When we see a woman on an OC with impaired ovarian reserve, we would presume [based on these new findings] that her real ovarian reserve was about 30% higher than what we measure. We would advise her to be retested after she was off her OC for about 3 months," Dr. Birch Petersen said during a press conference before her presentation at the meeting.
The study included the first women seen at the clinic since it opened in 2011, excluding those who were pregnant or failed to supply adequate information. The cross-sectional cohort included 240 women on an estrogen-containing OC and 593 women with natural cycles.
The analysis focused on three parameters: blood level of anti-Müllerian hormone (AMH), antral follicle count (AFC), and ovarian volume. The multivariate, linear regression analysis adjusted for age, body mass index, smoking, age of maternal menopause, maternal smoking during pregnancy, preterm birth, and duration of OC use.
The analysis showed that compared with the women with natural cycles, those on an OC had a 19% relative reduction in their average blood level of AMH, a 16% relative reduction in average AFC, and a 47% relative reduction in average ovarian volume. The women on an OC also had smaller antral follicles. All three differences were statistically significant.
Seeing an effect from an estrogen-containing OC on all three measures makes sense because of their interrelatedness. The antral follicles produce AMH, and a reduction in antral follicle number as well as size would shrink the ovarian contents and result in reduced volume. These effects would not occur in women on a progestin-only OC, she said.
Dr. Birch Petersen had no disclosures.
On Twitter @mitchelzoler
MUNICH – Oral contraceptives do more than prevent unwanted pregnancy. They also make it hard to gauge a woman’s ovarian reserve, based on data from 833 women aged 19-46 years seen at a single Danish fertility clinic.
Study findings suggested that an accurate measure of a woman’s ovarian reserve can occur only after she has been off an estrogen-containing OC, probably for at least 3 months, Dr. Kathrine Birch Petersen reported at the annual meeting of the European Society of Human Reproduction and Embryology.
The impact of estrogen-containing OC use on reducing ovarian volume was especially pronounced in women under age 30, the reduction increased with longer durations of OC use, and the ability of OCs to mask a woman’s actual ovarian reserve was strong enough to potentially conceal a true case of premature ovarian insufficiency, said Dr. Birch Petersen, an ob.gyn. at the Fertility Assessment and Counseling Clinic at Rigshospitalet in Copenhagen.
"When we see a woman on an OC with impaired ovarian reserve, we would presume [based on these new findings] that her real ovarian reserve was about 30% higher than what we measure. We would advise her to be retested after she was off her OC for about 3 months," Dr. Birch Petersen said during a press conference before her presentation at the meeting.
The study included the first women seen at the clinic since it opened in 2011, excluding those who were pregnant or failed to supply adequate information. The cross-sectional cohort included 240 women on an estrogen-containing OC and 593 women with natural cycles.
The analysis focused on three parameters: blood level of anti-Müllerian hormone (AMH), antral follicle count (AFC), and ovarian volume. The multivariate, linear regression analysis adjusted for age, body mass index, smoking, age of maternal menopause, maternal smoking during pregnancy, preterm birth, and duration of OC use.
The analysis showed that compared with the women with natural cycles, those on an OC had a 19% relative reduction in their average blood level of AMH, a 16% relative reduction in average AFC, and a 47% relative reduction in average ovarian volume. The women on an OC also had smaller antral follicles. All three differences were statistically significant.
Seeing an effect from an estrogen-containing OC on all three measures makes sense because of their interrelatedness. The antral follicles produce AMH, and a reduction in antral follicle number as well as size would shrink the ovarian contents and result in reduced volume. These effects would not occur in women on a progestin-only OC, she said.
Dr. Birch Petersen had no disclosures.
On Twitter @mitchelzoler
AT ESHRE 2014
Key clinical point: Estrogen-containing oral contraceptive use is linked with significantly reduced levels of ovarian volume, antral follicle count, and anti-Müllerian hormone.
Major finding: Users of estrogen-containing OCs had an average 47% reduced ovarian volume, compared with similar women not taking an OC.
Data source: Review of 833 women aged 19-46 years seen at a Danish fertility clinic during 2011-2014.
Disclosures: Dr. Birch Petersen had no disclosures.
Hyperovulation before IVF linked to higher obstetric complication risk
MUNICH – Women who hyperrespond to ovarian stimulation have an increased risk for obstetric complication of a pregnancy started during the same oocyte retrieval cycle, based on data from more than 40,000 women who underwent assisted reproduction during 2001-2008.
A retrospective review of 28,275 singleton pregnancies that followed in vitro fertilization or intracytoplasmic sperm injection found that hyperresponsive women, defined as those producing more than 15 oocytes following ovarian stimulation, had a relative 9% increased risk for having a low-birth-weight baby and a 10% relative increased rate of preterm birth, compared with women who produced a normal oocyte number, 4-15, Dr. Sesh K. Sunkara said at the annual meeting of the European Society of Human Reproduction and Embryology. These differences were statistically significant.
Other significant correlates of an increased risk for low birth weight and preterm birth were age, presence of an ovulatory disorder, and presence of a tubular disorder, said Dr. Sunkara, an obstetrician at Royal Marsden Hospital in London. After establishing these risk factors in this derivation cohort of more than 28,000 women, Dr. Sunkara and her associates tested the relationships with data collected from an additional 12,118 women drawn from the same U.K. registry during 2001-2008 and found identical relationships.
The findings highlight the importance of using ovarian stimulation methods that avoid hyperstimulation and hyperresponse, Dr. Sunkara said. In addition, future research should explore the physiologic processes that underlie the increased obstetric risk seen in the oocyte hyperresponders.
The study used data collected by the U.K. Human Fertilization and Embryology Authority. The overall live birth rate among the 28,275 women in the derivation cohort was 23%. The rate of preterm birth (less than 37 weeks’ gestation) and low-birth-weight babies (less than 2500 g) was 10% for each category. The rate of extreme preterm birth (less than 32 weeks) and the rate of extreme low birth weight (less than 1500 g) was 2% for each category.
The rate of extreme low birth rate was increased by a relative 16% among the hyperresponsive women, compared with those who produced 4-15 oocytes. The rate of extreme preterm birth increased by a relative 32%, compared with normally responsive women. Women with a poor response to ovarian stimulation, producing three or fewer oocytes, had similar rates of obstetric complications as normally responding women.
The analysis showed the highest combined rate of low birth weight plus preterm birth as 19% of all births in women younger than age 34 with hyperresponse to stimulation and both ovulatory and tubular disorders. In contrast, women aged 38 or older with a poor response to ovarian stimulation and neither an ovulatory nor a tubal disorder had a 10% combined rate of low-birth-weight and preterm birth pregnancies.
Dr. Sunkara had no disclosures.
On Twitter @mitchelzoler
MUNICH – Women who hyperrespond to ovarian stimulation have an increased risk for obstetric complication of a pregnancy started during the same oocyte retrieval cycle, based on data from more than 40,000 women who underwent assisted reproduction during 2001-2008.
A retrospective review of 28,275 singleton pregnancies that followed in vitro fertilization or intracytoplasmic sperm injection found that hyperresponsive women, defined as those producing more than 15 oocytes following ovarian stimulation, had a relative 9% increased risk for having a low-birth-weight baby and a 10% relative increased rate of preterm birth, compared with women who produced a normal oocyte number, 4-15, Dr. Sesh K. Sunkara said at the annual meeting of the European Society of Human Reproduction and Embryology. These differences were statistically significant.
Other significant correlates of an increased risk for low birth weight and preterm birth were age, presence of an ovulatory disorder, and presence of a tubular disorder, said Dr. Sunkara, an obstetrician at Royal Marsden Hospital in London. After establishing these risk factors in this derivation cohort of more than 28,000 women, Dr. Sunkara and her associates tested the relationships with data collected from an additional 12,118 women drawn from the same U.K. registry during 2001-2008 and found identical relationships.
The findings highlight the importance of using ovarian stimulation methods that avoid hyperstimulation and hyperresponse, Dr. Sunkara said. In addition, future research should explore the physiologic processes that underlie the increased obstetric risk seen in the oocyte hyperresponders.
The study used data collected by the U.K. Human Fertilization and Embryology Authority. The overall live birth rate among the 28,275 women in the derivation cohort was 23%. The rate of preterm birth (less than 37 weeks’ gestation) and low-birth-weight babies (less than 2500 g) was 10% for each category. The rate of extreme preterm birth (less than 32 weeks) and the rate of extreme low birth weight (less than 1500 g) was 2% for each category.
The rate of extreme low birth rate was increased by a relative 16% among the hyperresponsive women, compared with those who produced 4-15 oocytes. The rate of extreme preterm birth increased by a relative 32%, compared with normally responsive women. Women with a poor response to ovarian stimulation, producing three or fewer oocytes, had similar rates of obstetric complications as normally responding women.
The analysis showed the highest combined rate of low birth weight plus preterm birth as 19% of all births in women younger than age 34 with hyperresponse to stimulation and both ovulatory and tubular disorders. In contrast, women aged 38 or older with a poor response to ovarian stimulation and neither an ovulatory nor a tubal disorder had a 10% combined rate of low-birth-weight and preterm birth pregnancies.
Dr. Sunkara had no disclosures.
On Twitter @mitchelzoler
MUNICH – Women who hyperrespond to ovarian stimulation have an increased risk for obstetric complication of a pregnancy started during the same oocyte retrieval cycle, based on data from more than 40,000 women who underwent assisted reproduction during 2001-2008.
A retrospective review of 28,275 singleton pregnancies that followed in vitro fertilization or intracytoplasmic sperm injection found that hyperresponsive women, defined as those producing more than 15 oocytes following ovarian stimulation, had a relative 9% increased risk for having a low-birth-weight baby and a 10% relative increased rate of preterm birth, compared with women who produced a normal oocyte number, 4-15, Dr. Sesh K. Sunkara said at the annual meeting of the European Society of Human Reproduction and Embryology. These differences were statistically significant.
Other significant correlates of an increased risk for low birth weight and preterm birth were age, presence of an ovulatory disorder, and presence of a tubular disorder, said Dr. Sunkara, an obstetrician at Royal Marsden Hospital in London. After establishing these risk factors in this derivation cohort of more than 28,000 women, Dr. Sunkara and her associates tested the relationships with data collected from an additional 12,118 women drawn from the same U.K. registry during 2001-2008 and found identical relationships.
The findings highlight the importance of using ovarian stimulation methods that avoid hyperstimulation and hyperresponse, Dr. Sunkara said. In addition, future research should explore the physiologic processes that underlie the increased obstetric risk seen in the oocyte hyperresponders.
The study used data collected by the U.K. Human Fertilization and Embryology Authority. The overall live birth rate among the 28,275 women in the derivation cohort was 23%. The rate of preterm birth (less than 37 weeks’ gestation) and low-birth-weight babies (less than 2500 g) was 10% for each category. The rate of extreme preterm birth (less than 32 weeks) and the rate of extreme low birth weight (less than 1500 g) was 2% for each category.
The rate of extreme low birth rate was increased by a relative 16% among the hyperresponsive women, compared with those who produced 4-15 oocytes. The rate of extreme preterm birth increased by a relative 32%, compared with normally responsive women. Women with a poor response to ovarian stimulation, producing three or fewer oocytes, had similar rates of obstetric complications as normally responding women.
The analysis showed the highest combined rate of low birth weight plus preterm birth as 19% of all births in women younger than age 34 with hyperresponse to stimulation and both ovulatory and tubular disorders. In contrast, women aged 38 or older with a poor response to ovarian stimulation and neither an ovulatory nor a tubal disorder had a 10% combined rate of low-birth-weight and preterm birth pregnancies.
Dr. Sunkara had no disclosures.
On Twitter @mitchelzoler
FROM ESHRE 2014
Key clinical point: Hyperresponders to ovarian stimulation are at increased risk for low-birth-weight and preterm birth deliveries.
Major finding: Hyperresponsive women had a 10% increase in relative risk of low birth weight and preterm birth as compared with normal responders.
Data source: Records review for 40,393 women pregnant after assisted reproduction during 2001-2008 collected by the U.K. Human Fertilization and Embryology Authority.
Disclosures: Dr. Sunkara had no disclosures.
High aortic aneurysm dissection risk in giant-cell arteritis patients
PARIS – The usual rule that the larger an aortic aneurysm grows the greater the risk it will undergo dissection or rupture doesn’t work in patients with giant-cell arteritis. Their aortic aneurysms appear liable to dissect or rupture at any size after the diagnosis of giant-cell arteritis occurs, based on a retrospective study of 195 patients followed at a single U.S. center.
"Aortic size at diagnosis or last follow-up did not predict aortic dissection or rupture," Dr. Ashima Makol reported at the annual European Congress of Rheumatology, nor were linear, serial measurements of aortic size able to reliably predict risk for these complications in patients with GCA. Without a reliable way to identify patients with GCA at risk for dissection or rupture, the only management advice remaining is to follow GCA patients annually with imaging, said Dr. Makol, a rheumatologist at the Mayo Clinic in Rochester, Minn.
Positron emission tomography, CT angiography, or MR angiography seem to be the best ways to follow these patients, but if those are too costly to do annually, then transesophageal echocardiography or a chest x-ray are other options, Dr. Makol said in an interview.
Although 30% of patients with GCA have a vasculitis that involves the aorta and its branches and an increased risk for developing aortic aneurysms, the way these aneurysms change over time and the relationship between aneurysm size and the risk for dissection or rupture in GCA patients were not previously reported. To address this, Dr. Makol and her associates reviewed 195 patients with GCA and an aortic aneurysm seen at the Mayo Clinic during 2000-2012.
The aneurysms occurred in the ascending thoracic aorta in 161 patients (83%), the descending thoracic aorta in 21 (11%), and the abdominal aorta in the remaining 13 patients (7%). (Percentages total 101% because of rounding.) The patients averaged 74 years old, 62% were women, and 49% had a history of smoking.
During follow-up, 14 patients (7%) had an aneurysm dissection, and 1 patient (1%) had an aneurysm rupture, the investigators reported. All of the dissections and the rupture occurred in thoracic aorta aneurysms.
At the time of GCA diagnosis, the average aneurysm size in the 15 patients who developed an aneurysm complication was 51 mm, which was very similar to the average size of 49 mm in the 180 patients who did not have an aneurysm dissection or rupture during follow-up.
Patients also showed no clear link between aneurysm size at the time of dissection or rupture and the aneurysm size during follow-up of patients without these complications. The average maximum aneurysm diameter among the 15 patients with a complication at the time of their event was 54 mm, while the average aneurysm size at last follow-up among those without a dissection or rupture during follow-up was 50 mm, a difference that was not statistically significant, Dr. Makol said.
The average rate of aneurysm growth during 3 years of follow-up for all the GCA patients in the analysis was 1.59 mm/year, a rate "somewhat higher" than the average annual growth rate of 1 mm/year reported for aortic aneurysms in patients without inflammatory disease. The 54-mm average aneurysm diameter at the time of dissection or rupture in the CGA patients was "somewhat lower" than the 65-mm average aneurysm diameter seen at the time of dissection or rupture in patients without inflammatory disease, she noted.
The study is the first reported to look at the pattern of aneurysm growth and complications in GCA patients, although it is limited to the retrospective experience at one tertiary referral center and so may reflect a referral bias, Dr. Makol said. But the inability of the analysis to identify aneurysm characteristics in GCA patients that can telegraph an increased risk for complications means that all GCA patients with an aortic aneurysm need careful surveillance by annual imaging, she advised.
Dr. Makol said that she had no disclosures.
The take home message from this article can be summed up in this manner: Aneurysms in patients with, or that are the result of, giant-cell arteritis (GCA) may be at increased risk for a catastrophic event at an earlier, or at least more unpredictable point, than aneurysms that are not associated with GCA. As the article states, "the usual rule that the larger an aortic aneurysm grows the greater the risk" may not hold in this small subset of patients with GCA. Further interpretation of this article leads me to conclude, however, that the discussion centers (nearly) exclusively on pathology involving the ascending aorta. Only a very small percentage of the cohort that was studied had aneurysms distal to the subclavian artery and none of the fifteen catastrophic events that were noted occurred below the diaphragm. While not clear from the article, it is quite likely all of the catastrophic events, rupture or dissection, occurred in association with disease involving the ascending aorta. As such, the article’s content may be more appropriate for those who routinely treat patients with ascending aortic pathology and not so much for vascular surgeons who traditionally treat aortic pathology from the transverse arch down. In addition, there seems to be the inference in the article that this data can be directly translated to disease affecting the infra-renal aorta which is a conclusion that I would hold, is premature, at best. I, for one, must admit that while I have seen a number of patients with GCA, I have never had the occasion to treat an aneurysm in one of them. While this may be true for many of us, the point of the article does have merit and the information should be stored somewhere in our memory banks. Who knows, perhaps someday we may see it pop up on some board question.
Dr. Mark D. Morasch is a vascular surgeon at St Vincent Healthcare Heart and Vascular, Billings, Montana, and an associate medical editor for Vascular Specialist.
The take home message from this article can be summed up in this manner: Aneurysms in patients with, or that are the result of, giant-cell arteritis (GCA) may be at increased risk for a catastrophic event at an earlier, or at least more unpredictable point, than aneurysms that are not associated with GCA. As the article states, "the usual rule that the larger an aortic aneurysm grows the greater the risk" may not hold in this small subset of patients with GCA. Further interpretation of this article leads me to conclude, however, that the discussion centers (nearly) exclusively on pathology involving the ascending aorta. Only a very small percentage of the cohort that was studied had aneurysms distal to the subclavian artery and none of the fifteen catastrophic events that were noted occurred below the diaphragm. While not clear from the article, it is quite likely all of the catastrophic events, rupture or dissection, occurred in association with disease involving the ascending aorta. As such, the article’s content may be more appropriate for those who routinely treat patients with ascending aortic pathology and not so much for vascular surgeons who traditionally treat aortic pathology from the transverse arch down. In addition, there seems to be the inference in the article that this data can be directly translated to disease affecting the infra-renal aorta which is a conclusion that I would hold, is premature, at best. I, for one, must admit that while I have seen a number of patients with GCA, I have never had the occasion to treat an aneurysm in one of them. While this may be true for many of us, the point of the article does have merit and the information should be stored somewhere in our memory banks. Who knows, perhaps someday we may see it pop up on some board question.
Dr. Mark D. Morasch is a vascular surgeon at St Vincent Healthcare Heart and Vascular, Billings, Montana, and an associate medical editor for Vascular Specialist.
The take home message from this article can be summed up in this manner: Aneurysms in patients with, or that are the result of, giant-cell arteritis (GCA) may be at increased risk for a catastrophic event at an earlier, or at least more unpredictable point, than aneurysms that are not associated with GCA. As the article states, "the usual rule that the larger an aortic aneurysm grows the greater the risk" may not hold in this small subset of patients with GCA. Further interpretation of this article leads me to conclude, however, that the discussion centers (nearly) exclusively on pathology involving the ascending aorta. Only a very small percentage of the cohort that was studied had aneurysms distal to the subclavian artery and none of the fifteen catastrophic events that were noted occurred below the diaphragm. While not clear from the article, it is quite likely all of the catastrophic events, rupture or dissection, occurred in association with disease involving the ascending aorta. As such, the article’s content may be more appropriate for those who routinely treat patients with ascending aortic pathology and not so much for vascular surgeons who traditionally treat aortic pathology from the transverse arch down. In addition, there seems to be the inference in the article that this data can be directly translated to disease affecting the infra-renal aorta which is a conclusion that I would hold, is premature, at best. I, for one, must admit that while I have seen a number of patients with GCA, I have never had the occasion to treat an aneurysm in one of them. While this may be true for many of us, the point of the article does have merit and the information should be stored somewhere in our memory banks. Who knows, perhaps someday we may see it pop up on some board question.
Dr. Mark D. Morasch is a vascular surgeon at St Vincent Healthcare Heart and Vascular, Billings, Montana, and an associate medical editor for Vascular Specialist.
PARIS – The usual rule that the larger an aortic aneurysm grows the greater the risk it will undergo dissection or rupture doesn’t work in patients with giant-cell arteritis. Their aortic aneurysms appear liable to dissect or rupture at any size after the diagnosis of giant-cell arteritis occurs, based on a retrospective study of 195 patients followed at a single U.S. center.
"Aortic size at diagnosis or last follow-up did not predict aortic dissection or rupture," Dr. Ashima Makol reported at the annual European Congress of Rheumatology, nor were linear, serial measurements of aortic size able to reliably predict risk for these complications in patients with GCA. Without a reliable way to identify patients with GCA at risk for dissection or rupture, the only management advice remaining is to follow GCA patients annually with imaging, said Dr. Makol, a rheumatologist at the Mayo Clinic in Rochester, Minn.
Positron emission tomography, CT angiography, or MR angiography seem to be the best ways to follow these patients, but if those are too costly to do annually, then transesophageal echocardiography or a chest x-ray are other options, Dr. Makol said in an interview.
Although 30% of patients with GCA have a vasculitis that involves the aorta and its branches and an increased risk for developing aortic aneurysms, the way these aneurysms change over time and the relationship between aneurysm size and the risk for dissection or rupture in GCA patients were not previously reported. To address this, Dr. Makol and her associates reviewed 195 patients with GCA and an aortic aneurysm seen at the Mayo Clinic during 2000-2012.
The aneurysms occurred in the ascending thoracic aorta in 161 patients (83%), the descending thoracic aorta in 21 (11%), and the abdominal aorta in the remaining 13 patients (7%). (Percentages total 101% because of rounding.) The patients averaged 74 years old, 62% were women, and 49% had a history of smoking.
During follow-up, 14 patients (7%) had an aneurysm dissection, and 1 patient (1%) had an aneurysm rupture, the investigators reported. All of the dissections and the rupture occurred in thoracic aorta aneurysms.
At the time of GCA diagnosis, the average aneurysm size in the 15 patients who developed an aneurysm complication was 51 mm, which was very similar to the average size of 49 mm in the 180 patients who did not have an aneurysm dissection or rupture during follow-up.
Patients also showed no clear link between aneurysm size at the time of dissection or rupture and the aneurysm size during follow-up of patients without these complications. The average maximum aneurysm diameter among the 15 patients with a complication at the time of their event was 54 mm, while the average aneurysm size at last follow-up among those without a dissection or rupture during follow-up was 50 mm, a difference that was not statistically significant, Dr. Makol said.
The average rate of aneurysm growth during 3 years of follow-up for all the GCA patients in the analysis was 1.59 mm/year, a rate "somewhat higher" than the average annual growth rate of 1 mm/year reported for aortic aneurysms in patients without inflammatory disease. The 54-mm average aneurysm diameter at the time of dissection or rupture in the CGA patients was "somewhat lower" than the 65-mm average aneurysm diameter seen at the time of dissection or rupture in patients without inflammatory disease, she noted.
The study is the first reported to look at the pattern of aneurysm growth and complications in GCA patients, although it is limited to the retrospective experience at one tertiary referral center and so may reflect a referral bias, Dr. Makol said. But the inability of the analysis to identify aneurysm characteristics in GCA patients that can telegraph an increased risk for complications means that all GCA patients with an aortic aneurysm need careful surveillance by annual imaging, she advised.
Dr. Makol said that she had no disclosures.
PARIS – The usual rule that the larger an aortic aneurysm grows the greater the risk it will undergo dissection or rupture doesn’t work in patients with giant-cell arteritis. Their aortic aneurysms appear liable to dissect or rupture at any size after the diagnosis of giant-cell arteritis occurs, based on a retrospective study of 195 patients followed at a single U.S. center.
"Aortic size at diagnosis or last follow-up did not predict aortic dissection or rupture," Dr. Ashima Makol reported at the annual European Congress of Rheumatology, nor were linear, serial measurements of aortic size able to reliably predict risk for these complications in patients with GCA. Without a reliable way to identify patients with GCA at risk for dissection or rupture, the only management advice remaining is to follow GCA patients annually with imaging, said Dr. Makol, a rheumatologist at the Mayo Clinic in Rochester, Minn.
Positron emission tomography, CT angiography, or MR angiography seem to be the best ways to follow these patients, but if those are too costly to do annually, then transesophageal echocardiography or a chest x-ray are other options, Dr. Makol said in an interview.
Although 30% of patients with GCA have a vasculitis that involves the aorta and its branches and an increased risk for developing aortic aneurysms, the way these aneurysms change over time and the relationship between aneurysm size and the risk for dissection or rupture in GCA patients were not previously reported. To address this, Dr. Makol and her associates reviewed 195 patients with GCA and an aortic aneurysm seen at the Mayo Clinic during 2000-2012.
The aneurysms occurred in the ascending thoracic aorta in 161 patients (83%), the descending thoracic aorta in 21 (11%), and the abdominal aorta in the remaining 13 patients (7%). (Percentages total 101% because of rounding.) The patients averaged 74 years old, 62% were women, and 49% had a history of smoking.
During follow-up, 14 patients (7%) had an aneurysm dissection, and 1 patient (1%) had an aneurysm rupture, the investigators reported. All of the dissections and the rupture occurred in thoracic aorta aneurysms.
At the time of GCA diagnosis, the average aneurysm size in the 15 patients who developed an aneurysm complication was 51 mm, which was very similar to the average size of 49 mm in the 180 patients who did not have an aneurysm dissection or rupture during follow-up.
Patients also showed no clear link between aneurysm size at the time of dissection or rupture and the aneurysm size during follow-up of patients without these complications. The average maximum aneurysm diameter among the 15 patients with a complication at the time of their event was 54 mm, while the average aneurysm size at last follow-up among those without a dissection or rupture during follow-up was 50 mm, a difference that was not statistically significant, Dr. Makol said.
The average rate of aneurysm growth during 3 years of follow-up for all the GCA patients in the analysis was 1.59 mm/year, a rate "somewhat higher" than the average annual growth rate of 1 mm/year reported for aortic aneurysms in patients without inflammatory disease. The 54-mm average aneurysm diameter at the time of dissection or rupture in the CGA patients was "somewhat lower" than the 65-mm average aneurysm diameter seen at the time of dissection or rupture in patients without inflammatory disease, she noted.
The study is the first reported to look at the pattern of aneurysm growth and complications in GCA patients, although it is limited to the retrospective experience at one tertiary referral center and so may reflect a referral bias, Dr. Makol said. But the inability of the analysis to identify aneurysm characteristics in GCA patients that can telegraph an increased risk for complications means that all GCA patients with an aortic aneurysm need careful surveillance by annual imaging, she advised.
Dr. Makol said that she had no disclosures.
AT THE EULAR CONGRESS 2014
Key clinical point: Risk for aortic aneurysm dissection in patients with giant-cell arteritis showed no link to aneurysm size.
Major finding: GCA patients with dissection or rupture of an aortic aneurysm had aneurysms that were similar in size to those of GCA patients without these complications.
Data source: Retrospective study of 195 patients with GCA at one U.S. center.
Disclosures: Dr. Makol said that she had no disclosures.
BVS and renal denervation fuel a continental divide
It’s no secret that the Food and Drug Administration and the European Commission take very different approaches to how they allow medical devices onto the market. Their different tacks have produced a profound disconnect in how cardiologists on the two continents use two of the newest technologies in their specialty: bioabsorbable vascular scaffolds for coronary disease and renal denervation for controlling treatment-resistant hypertension.
The European Commission granted a CE mark to the first bioabsorbable vascular stent (BVS), Abbott’s Absorb, in 2011, and Abbott announced the launch of international marketing in September 2012. By last May, an Absorb BVS had been placed in more than 50,000 patients worldwide, according to a company spokesperson.
In contrast, the U.S. total of patients who received a BVS is much lower, and is limited to those randomized to receive a BVS in one of the two ongoing U.S. pivotal trials comparing its performance with a second-generation, drug-eluting metallic stent. One of those trials, with 2,000 total patients, completed enrollment in April 2014, but a second slated to have 3,000 total continues to enroll, so the soonest a BVS might come onto the U.S. market is at least 3 years off.
Not only does Europe have tens of thousands of BVS-treated patients, but several established interventional programs there, such as the Thoraxcenter in Rotterdam (the Netherlands), and others in Italy, Spain, Poland, and the Czech Republic, have adopted the BVS as their default approach for treating patients with percutaneous coronary intervention (PCI). For now, a BVS-first approach does not translate into universal use because only about a fifth of all PCI patients have anatomy suitable for BVS placement. But these BVS-first centers are using them in any PCI patient with anatomy that’s appropriate regardless of the severity of their coronary disease or the complexity of their coronary lesion. This means that BVSs have been placed in growing numbers of European patients with myocardial infarctions, acute coronary syndrome, and bifurcations, as well as in other types of clinically advanced PCI patients.
For renal denervation the numbers are not as disparate, but the prevailing attitudes of many thought leaders are. In Europe, Asia, and a few other parts of the world, interventionalists have used the Symplicity catheter, the first renal denervation device on the world market starting in 2010, in more than 5,500 patients as of this August, according to a spokesperson for Medtronic, the company that sells this device.
The consensus among many European interventional cardiologists and hypertension specialists at the annual EuroPCR meeting was that renal denervation is an effective and valuable option for managing otherwise uncontrolled hypertension, when performed carefully and thoroughly in well-selected patients with true drug-resistant hypertension. They stuck with that opinion despite the report in March that renal denervation failed to produce a significant blood pressure reduction compared with a sham procedure in the pivotal U.S. trial, SYMPLICITY HTN 3 (New Engl. J. Med. 2014;370:1393-1401).
A statement from three leading European cardiologists issued under the auspices of the EuroPCR meeting last May spelled out that despite the negative trial findings, "renal denervation is an option in patients with difficult-to-control hypertension, in whom other treatments have failed."
In the United States, the failed pivotal trial seems to have temporarily put U.S. testing of renal denervation on hold as companies and their consultants scramble to figure out how to avoid the problems that doomed SYMPLICITY HTN 3.
U.S. and European cardiologists show agreement in several other new areas of practice, but for BVS and renal denervation the transatlantic divide is stark.
On Twitter @mitchelzoler
It’s no secret that the Food and Drug Administration and the European Commission take very different approaches to how they allow medical devices onto the market. Their different tacks have produced a profound disconnect in how cardiologists on the two continents use two of the newest technologies in their specialty: bioabsorbable vascular scaffolds for coronary disease and renal denervation for controlling treatment-resistant hypertension.
The European Commission granted a CE mark to the first bioabsorbable vascular stent (BVS), Abbott’s Absorb, in 2011, and Abbott announced the launch of international marketing in September 2012. By last May, an Absorb BVS had been placed in more than 50,000 patients worldwide, according to a company spokesperson.
In contrast, the U.S. total of patients who received a BVS is much lower, and is limited to those randomized to receive a BVS in one of the two ongoing U.S. pivotal trials comparing its performance with a second-generation, drug-eluting metallic stent. One of those trials, with 2,000 total patients, completed enrollment in April 2014, but a second slated to have 3,000 total continues to enroll, so the soonest a BVS might come onto the U.S. market is at least 3 years off.
Not only does Europe have tens of thousands of BVS-treated patients, but several established interventional programs there, such as the Thoraxcenter in Rotterdam (the Netherlands), and others in Italy, Spain, Poland, and the Czech Republic, have adopted the BVS as their default approach for treating patients with percutaneous coronary intervention (PCI). For now, a BVS-first approach does not translate into universal use because only about a fifth of all PCI patients have anatomy suitable for BVS placement. But these BVS-first centers are using them in any PCI patient with anatomy that’s appropriate regardless of the severity of their coronary disease or the complexity of their coronary lesion. This means that BVSs have been placed in growing numbers of European patients with myocardial infarctions, acute coronary syndrome, and bifurcations, as well as in other types of clinically advanced PCI patients.
For renal denervation the numbers are not as disparate, but the prevailing attitudes of many thought leaders are. In Europe, Asia, and a few other parts of the world, interventionalists have used the Symplicity catheter, the first renal denervation device on the world market starting in 2010, in more than 5,500 patients as of this August, according to a spokesperson for Medtronic, the company that sells this device.
The consensus among many European interventional cardiologists and hypertension specialists at the annual EuroPCR meeting was that renal denervation is an effective and valuable option for managing otherwise uncontrolled hypertension, when performed carefully and thoroughly in well-selected patients with true drug-resistant hypertension. They stuck with that opinion despite the report in March that renal denervation failed to produce a significant blood pressure reduction compared with a sham procedure in the pivotal U.S. trial, SYMPLICITY HTN 3 (New Engl. J. Med. 2014;370:1393-1401).
A statement from three leading European cardiologists issued under the auspices of the EuroPCR meeting last May spelled out that despite the negative trial findings, "renal denervation is an option in patients with difficult-to-control hypertension, in whom other treatments have failed."
In the United States, the failed pivotal trial seems to have temporarily put U.S. testing of renal denervation on hold as companies and their consultants scramble to figure out how to avoid the problems that doomed SYMPLICITY HTN 3.
U.S. and European cardiologists show agreement in several other new areas of practice, but for BVS and renal denervation the transatlantic divide is stark.
On Twitter @mitchelzoler
It’s no secret that the Food and Drug Administration and the European Commission take very different approaches to how they allow medical devices onto the market. Their different tacks have produced a profound disconnect in how cardiologists on the two continents use two of the newest technologies in their specialty: bioabsorbable vascular scaffolds for coronary disease and renal denervation for controlling treatment-resistant hypertension.
The European Commission granted a CE mark to the first bioabsorbable vascular stent (BVS), Abbott’s Absorb, in 2011, and Abbott announced the launch of international marketing in September 2012. By last May, an Absorb BVS had been placed in more than 50,000 patients worldwide, according to a company spokesperson.
In contrast, the U.S. total of patients who received a BVS is much lower, and is limited to those randomized to receive a BVS in one of the two ongoing U.S. pivotal trials comparing its performance with a second-generation, drug-eluting metallic stent. One of those trials, with 2,000 total patients, completed enrollment in April 2014, but a second slated to have 3,000 total continues to enroll, so the soonest a BVS might come onto the U.S. market is at least 3 years off.
Not only does Europe have tens of thousands of BVS-treated patients, but several established interventional programs there, such as the Thoraxcenter in Rotterdam (the Netherlands), and others in Italy, Spain, Poland, and the Czech Republic, have adopted the BVS as their default approach for treating patients with percutaneous coronary intervention (PCI). For now, a BVS-first approach does not translate into universal use because only about a fifth of all PCI patients have anatomy suitable for BVS placement. But these BVS-first centers are using them in any PCI patient with anatomy that’s appropriate regardless of the severity of their coronary disease or the complexity of their coronary lesion. This means that BVSs have been placed in growing numbers of European patients with myocardial infarctions, acute coronary syndrome, and bifurcations, as well as in other types of clinically advanced PCI patients.
For renal denervation the numbers are not as disparate, but the prevailing attitudes of many thought leaders are. In Europe, Asia, and a few other parts of the world, interventionalists have used the Symplicity catheter, the first renal denervation device on the world market starting in 2010, in more than 5,500 patients as of this August, according to a spokesperson for Medtronic, the company that sells this device.
The consensus among many European interventional cardiologists and hypertension specialists at the annual EuroPCR meeting was that renal denervation is an effective and valuable option for managing otherwise uncontrolled hypertension, when performed carefully and thoroughly in well-selected patients with true drug-resistant hypertension. They stuck with that opinion despite the report in March that renal denervation failed to produce a significant blood pressure reduction compared with a sham procedure in the pivotal U.S. trial, SYMPLICITY HTN 3 (New Engl. J. Med. 2014;370:1393-1401).
A statement from three leading European cardiologists issued under the auspices of the EuroPCR meeting last May spelled out that despite the negative trial findings, "renal denervation is an option in patients with difficult-to-control hypertension, in whom other treatments have failed."
In the United States, the failed pivotal trial seems to have temporarily put U.S. testing of renal denervation on hold as companies and their consultants scramble to figure out how to avoid the problems that doomed SYMPLICITY HTN 3.
U.S. and European cardiologists show agreement in several other new areas of practice, but for BVS and renal denervation the transatlantic divide is stark.
On Twitter @mitchelzoler
Elderly CAD patients benefit from a systolic value below 140 mm Hg
Elderly hypertensive patients who had coronary artery disease and a baseline systolic blood pressure greater than 150 mm Hg had significantly fewer cardiovascular deaths and strokes if they achieved a systolic pressure of less than 140 mm Hg, compared with similar patients who achieved a systolic pressure of 140-149 mm Hg.
The findings were noted in a post-hoc analysis of data collected from more than 8,000 patients in an international treatment study. The researchers compared outcomes for patients who achieved these two different levels of systolic pressure as a way to assess the new hypertension treatment goal of a systolic pressure less than 150 mm Hg in patients aged 60 years or older as recommended by the group originally appointed as the Eighth Joint National Committee (JNC 8) (JAMA 2014;311:507-20). Their finding that the patients who achieved the lower systolic pressure level of less than 140 mm Hg had a significantly lower rate of cardiovascular death and stroke as compared with patients whose systolic pressure remained in the 140-149 mm Hg range provided "important information to focus the risk-benefit discussion for patients who had on-treatment blood pressure in the 140-150 mm Hg range," wrote Dr. Sripal Bangalore, a cardiologist at New York University and his associates in an article published online on August 18 (J. Amer. Coll. Cardiol. 2014;311:784-93).
At least one expert questioned the ability of such a post-hoc analysis to address blood pressure goals in a meaningful way. "To adjudicate between a target of 140 mm Hg and 150 mm Hg it would be necessary to perform a prospective trial in which the population was randomized to these targets," commented Dr. Alan H. Gradman in an editorial that accompanied the new study (J. Amer. Coll. Cardiol. 2014;311:794-6). More aggressive blood pressure lowering could result in increased adverse events, he warned.
Despite these limitations, other experts hailed the new finding as important evidence against the recommendations of the former JNC 8 committee. "This study supports the concerns raised by many stakeholders, including the American College of Cardiology [ACC], the American Heart Association [AHA], and a number of the individual members of the JNC 8 panel (Ann. Intern. Med. 2014;160:499-503.), about the panel’s 2013 recommendations to raise blood pressure targets in older patients," said Dr. Patrick T. O’Gara, ACC president, and Dr. Elliott Antman, AHA president, in a joint written statement released to coincide with the new report’s publication.
"This new research suggests that raising the threshold for treatment of hypertension in patients 60 years of age or older with coronary artery disease may be detrimental to the best interest of patients and the public. It underscores ongoing concerns about adopting the unofficial 2013 targets as proposed by the panel originally appointed to write JNC 8," Dr. O’Gara and Dr. Antman said in their statement. Their statement added that "the ACC and AHA, working with the NHLBI [National Heart, Lung, and Blood Institute] are in the process of assembling the writing panel that will evaluate evidence from a variety of sources and provide a comprehensive update of the hypertension guideline."
The post-hoc analysis focused on a subgroup of 8,354 patients from among the 22,576 total patients enrolled in the (International Verapamil SR Trandolapril Study (INVEST), which enrolled patients aged 50 years or older with hypertension and pre-existing coronary artery disease at 862 centers in 14 countries (JAMA 2003;290:2805-16). Patients had been randomized to receive treatment with either a combination of verapamil sustained release plus trandolapril and hydrochlorothiazide if needed, or a combination of atenolol plus hydrochlorothiazide and trandolapril if needed, with a goal blood pressure on treatment of less than 140/90 mm Hg or less than 130/85 mmHg in patients with diabetes, renal impairment, or both. The primary, combined outcome of INVEST showed no significant difference in efficacy or safety between the two regimens compared in the study, and roughly 70% of patients in both treatment subgroups reached their goal blood pressures.
The new analysis selected the 8,354 patients who enrolled in INVEST at age 60 years or older and had a systolic blood pressure of at least 150 mm Hg at baseline. This subgroup included 4,787 patients (57%) who achieved a systolic pressure of less than 140 mm Hg after 24 months on study treatment, 1,747 patients who achieved a systolic pressure of 140-149 mm Hg after 24 months, and 1,820 patients whose systolic pressure failed to fall below 150 mm Hg.
In multiple propensity score – adjusted analyses, the rate of cardiovascular death was a relative 34% higher among patients with an achieved systolic pressure of 140-149 mm Hg, compared with those whose pressures fell below 140 mm Hg. Also, the total rate of stroke was 89% higher among patients whose pressures stayed in the 140-149 mm Hg range, compared with patients who achieved lower pressures. The rate of nonfatal stroke was 70% higher among the 140-149 mm Hg subgroup. All three of these differences were statistically significant.
By several other outcome measures, the event rates did not significantly differ between the two achieved pressure subgroups, including all-cause deaths, total myocardial infarctions, rate of revascularization, or total events. In contrast, for most outcome measures assessed, patients whose systolic pressures remained at 150 mm Hg or greater had significantly more events than patients whose pressures fell below 140 mm Hg. Similar rates of adverse events were seen in each of the three achieved–blood pressure subgroups.
Dr. Bangalore and his coauthors acknowledged several limitations of their analysis: The data were analyzed by achieved blood pressure, which is likely influenced by baseline characteristics. INVEST was not designed to compare different blood pressure targets. The results were specific for patients aged 60 or older with coronary artery disease and a baseline systolic pressure greater than 150 mm Hg. And the researchers did not design the new analysis to assess the benefit of treating patients with a systolic blood pressure of 140-150 mm Hg.
INVEST was sponsored by BASF Pharma and Abbott. Dr. Bangalore said that he has served on advisory boards for Abbott and other drug companies. Dr. Gradman, Dr. O’Gara, and Dr. Antman had no relevant disclosures.
On Twitter @mitchelzoler
The findings reported by Dr. Bangalore and his coauthors support a lower blood pressure target, specifically in patients with hypertension and coronary artery disease. The implication is that the appropriate systolic blood pressure target should be less than 140 mm Hg for patients aged 60 or older with coronary artery disease.
However, the evidence they present does not firmly refute a systolic pressure goal of less than 150 mm Hg for patients aged 60 or older, as recommended by the panel originally constituted as the Eighth Joint National Committee (JNC 8) (JAMA 2014;311:507-20). That’s because all patients included in the analysis – those patients who entered the INVEST trial with a systolic pressure greater than 150 mm Hg – would qualify for treatment based on the recommendations of the former JNC 8 panel. But little, if any, evidence from randomized clinical trials supports the idea that antihypertensive treatment reduces cardiovascular events in older adults who start treatment with a systolic blood pressure of 140-160 mm Hg.
Another limitation of the new analysis is that the INVEST design called on physicians to treat all patients to a goal pressure of less than 140/90 mm Hg or less than 130/85 mm Hg for patients with diabetes or renal impairment using either of two prespecified treatment regimens (JAMA 2003;290:2805-16). Although patients received either one or the other regimen based on the treatment arm to which they were randomized, the intensity of treatment that all patients in INVEST received was roughly the same. After 2 years, 57% of all patients in the subgroup studied in the post-hoc analysis responded with a systolic pressure that achieved the target. Although the 43% who did not meet the target had worse outcomes than the 57% who achieved a lower systolic pressure, it is not clear that the less responsive patients in the 43% group would have done better with intensified treatment that might have driven their systolic pressure down further, below 140 mm Hg.
In other words, the new analysis compared the outcomes of responders with nonresponders. To compare a target of less than 140 mm Hg with a target in the 140-150 mm Hg range among the nonresponder patients in INVEST would require a prospective trial in which the nonresponder patients are randomized to additional antihypertensive treatment or to placebo to test the benefit of intensified treatment to reach the sub–140 mm Hg goal. More aggressive treatment of these elderly nonresponders could increase the number of treatment-related adverse events. Without data like these, there is no right answer to the question of which systolic target to use.
I believe that the blood pressure target recommendations of the former JNC 8 panel are reasonable for patients with hypertension and coronary artery disease. Their recommendations continue to use a diastolic target pressure of less than 90 mm Hg, which helps insure that most patients with significant hypertension will receive appropriate treatment regardless of their systolic pressure. Clinicians need to use their clinical judgment about treatment for patients with isolated systolic hypertension in the 140-149 mm Hg range. Patients at increased risk for stroke should probably receive more intensive treatment. An important corollary recommendation from the former JNC-8 panel was that when treatment drops a patient’s systolic pressure below 140 mm Hg without causing adverse effects, the treatment can be maintained and the patient’s pressure kept at this lower level.
Dr. Alan H. Gradman is a cardiologist who practices in Pittsburgh. He had no relevant disclosures. He made these comments in an editorial that accompanied the report by Dr. Bangalore and coauthors (J. Amer. Coll. Cardiol. 2014;64:794-6).
The findings reported by Dr. Bangalore and his coauthors support a lower blood pressure target, specifically in patients with hypertension and coronary artery disease. The implication is that the appropriate systolic blood pressure target should be less than 140 mm Hg for patients aged 60 or older with coronary artery disease.
However, the evidence they present does not firmly refute a systolic pressure goal of less than 150 mm Hg for patients aged 60 or older, as recommended by the panel originally constituted as the Eighth Joint National Committee (JNC 8) (JAMA 2014;311:507-20). That’s because all patients included in the analysis – those patients who entered the INVEST trial with a systolic pressure greater than 150 mm Hg – would qualify for treatment based on the recommendations of the former JNC 8 panel. But little, if any, evidence from randomized clinical trials supports the idea that antihypertensive treatment reduces cardiovascular events in older adults who start treatment with a systolic blood pressure of 140-160 mm Hg.
Another limitation of the new analysis is that the INVEST design called on physicians to treat all patients to a goal pressure of less than 140/90 mm Hg or less than 130/85 mm Hg for patients with diabetes or renal impairment using either of two prespecified treatment regimens (JAMA 2003;290:2805-16). Although patients received either one or the other regimen based on the treatment arm to which they were randomized, the intensity of treatment that all patients in INVEST received was roughly the same. After 2 years, 57% of all patients in the subgroup studied in the post-hoc analysis responded with a systolic pressure that achieved the target. Although the 43% who did not meet the target had worse outcomes than the 57% who achieved a lower systolic pressure, it is not clear that the less responsive patients in the 43% group would have done better with intensified treatment that might have driven their systolic pressure down further, below 140 mm Hg.
In other words, the new analysis compared the outcomes of responders with nonresponders. To compare a target of less than 140 mm Hg with a target in the 140-150 mm Hg range among the nonresponder patients in INVEST would require a prospective trial in which the nonresponder patients are randomized to additional antihypertensive treatment or to placebo to test the benefit of intensified treatment to reach the sub–140 mm Hg goal. More aggressive treatment of these elderly nonresponders could increase the number of treatment-related adverse events. Without data like these, there is no right answer to the question of which systolic target to use.
I believe that the blood pressure target recommendations of the former JNC 8 panel are reasonable for patients with hypertension and coronary artery disease. Their recommendations continue to use a diastolic target pressure of less than 90 mm Hg, which helps insure that most patients with significant hypertension will receive appropriate treatment regardless of their systolic pressure. Clinicians need to use their clinical judgment about treatment for patients with isolated systolic hypertension in the 140-149 mm Hg range. Patients at increased risk for stroke should probably receive more intensive treatment. An important corollary recommendation from the former JNC-8 panel was that when treatment drops a patient’s systolic pressure below 140 mm Hg without causing adverse effects, the treatment can be maintained and the patient’s pressure kept at this lower level.
Dr. Alan H. Gradman is a cardiologist who practices in Pittsburgh. He had no relevant disclosures. He made these comments in an editorial that accompanied the report by Dr. Bangalore and coauthors (J. Amer. Coll. Cardiol. 2014;64:794-6).
The findings reported by Dr. Bangalore and his coauthors support a lower blood pressure target, specifically in patients with hypertension and coronary artery disease. The implication is that the appropriate systolic blood pressure target should be less than 140 mm Hg for patients aged 60 or older with coronary artery disease.
However, the evidence they present does not firmly refute a systolic pressure goal of less than 150 mm Hg for patients aged 60 or older, as recommended by the panel originally constituted as the Eighth Joint National Committee (JNC 8) (JAMA 2014;311:507-20). That’s because all patients included in the analysis – those patients who entered the INVEST trial with a systolic pressure greater than 150 mm Hg – would qualify for treatment based on the recommendations of the former JNC 8 panel. But little, if any, evidence from randomized clinical trials supports the idea that antihypertensive treatment reduces cardiovascular events in older adults who start treatment with a systolic blood pressure of 140-160 mm Hg.
Another limitation of the new analysis is that the INVEST design called on physicians to treat all patients to a goal pressure of less than 140/90 mm Hg or less than 130/85 mm Hg for patients with diabetes or renal impairment using either of two prespecified treatment regimens (JAMA 2003;290:2805-16). Although patients received either one or the other regimen based on the treatment arm to which they were randomized, the intensity of treatment that all patients in INVEST received was roughly the same. After 2 years, 57% of all patients in the subgroup studied in the post-hoc analysis responded with a systolic pressure that achieved the target. Although the 43% who did not meet the target had worse outcomes than the 57% who achieved a lower systolic pressure, it is not clear that the less responsive patients in the 43% group would have done better with intensified treatment that might have driven their systolic pressure down further, below 140 mm Hg.
In other words, the new analysis compared the outcomes of responders with nonresponders. To compare a target of less than 140 mm Hg with a target in the 140-150 mm Hg range among the nonresponder patients in INVEST would require a prospective trial in which the nonresponder patients are randomized to additional antihypertensive treatment or to placebo to test the benefit of intensified treatment to reach the sub–140 mm Hg goal. More aggressive treatment of these elderly nonresponders could increase the number of treatment-related adverse events. Without data like these, there is no right answer to the question of which systolic target to use.
I believe that the blood pressure target recommendations of the former JNC 8 panel are reasonable for patients with hypertension and coronary artery disease. Their recommendations continue to use a diastolic target pressure of less than 90 mm Hg, which helps insure that most patients with significant hypertension will receive appropriate treatment regardless of their systolic pressure. Clinicians need to use their clinical judgment about treatment for patients with isolated systolic hypertension in the 140-149 mm Hg range. Patients at increased risk for stroke should probably receive more intensive treatment. An important corollary recommendation from the former JNC-8 panel was that when treatment drops a patient’s systolic pressure below 140 mm Hg without causing adverse effects, the treatment can be maintained and the patient’s pressure kept at this lower level.
Dr. Alan H. Gradman is a cardiologist who practices in Pittsburgh. He had no relevant disclosures. He made these comments in an editorial that accompanied the report by Dr. Bangalore and coauthors (J. Amer. Coll. Cardiol. 2014;64:794-6).
Elderly hypertensive patients who had coronary artery disease and a baseline systolic blood pressure greater than 150 mm Hg had significantly fewer cardiovascular deaths and strokes if they achieved a systolic pressure of less than 140 mm Hg, compared with similar patients who achieved a systolic pressure of 140-149 mm Hg.
The findings were noted in a post-hoc analysis of data collected from more than 8,000 patients in an international treatment study. The researchers compared outcomes for patients who achieved these two different levels of systolic pressure as a way to assess the new hypertension treatment goal of a systolic pressure less than 150 mm Hg in patients aged 60 years or older as recommended by the group originally appointed as the Eighth Joint National Committee (JNC 8) (JAMA 2014;311:507-20). Their finding that the patients who achieved the lower systolic pressure level of less than 140 mm Hg had a significantly lower rate of cardiovascular death and stroke as compared with patients whose systolic pressure remained in the 140-149 mm Hg range provided "important information to focus the risk-benefit discussion for patients who had on-treatment blood pressure in the 140-150 mm Hg range," wrote Dr. Sripal Bangalore, a cardiologist at New York University and his associates in an article published online on August 18 (J. Amer. Coll. Cardiol. 2014;311:784-93).
At least one expert questioned the ability of such a post-hoc analysis to address blood pressure goals in a meaningful way. "To adjudicate between a target of 140 mm Hg and 150 mm Hg it would be necessary to perform a prospective trial in which the population was randomized to these targets," commented Dr. Alan H. Gradman in an editorial that accompanied the new study (J. Amer. Coll. Cardiol. 2014;311:794-6). More aggressive blood pressure lowering could result in increased adverse events, he warned.
Despite these limitations, other experts hailed the new finding as important evidence against the recommendations of the former JNC 8 committee. "This study supports the concerns raised by many stakeholders, including the American College of Cardiology [ACC], the American Heart Association [AHA], and a number of the individual members of the JNC 8 panel (Ann. Intern. Med. 2014;160:499-503.), about the panel’s 2013 recommendations to raise blood pressure targets in older patients," said Dr. Patrick T. O’Gara, ACC president, and Dr. Elliott Antman, AHA president, in a joint written statement released to coincide with the new report’s publication.
"This new research suggests that raising the threshold for treatment of hypertension in patients 60 years of age or older with coronary artery disease may be detrimental to the best interest of patients and the public. It underscores ongoing concerns about adopting the unofficial 2013 targets as proposed by the panel originally appointed to write JNC 8," Dr. O’Gara and Dr. Antman said in their statement. Their statement added that "the ACC and AHA, working with the NHLBI [National Heart, Lung, and Blood Institute] are in the process of assembling the writing panel that will evaluate evidence from a variety of sources and provide a comprehensive update of the hypertension guideline."
The post-hoc analysis focused on a subgroup of 8,354 patients from among the 22,576 total patients enrolled in the (International Verapamil SR Trandolapril Study (INVEST), which enrolled patients aged 50 years or older with hypertension and pre-existing coronary artery disease at 862 centers in 14 countries (JAMA 2003;290:2805-16). Patients had been randomized to receive treatment with either a combination of verapamil sustained release plus trandolapril and hydrochlorothiazide if needed, or a combination of atenolol plus hydrochlorothiazide and trandolapril if needed, with a goal blood pressure on treatment of less than 140/90 mm Hg or less than 130/85 mmHg in patients with diabetes, renal impairment, or both. The primary, combined outcome of INVEST showed no significant difference in efficacy or safety between the two regimens compared in the study, and roughly 70% of patients in both treatment subgroups reached their goal blood pressures.
The new analysis selected the 8,354 patients who enrolled in INVEST at age 60 years or older and had a systolic blood pressure of at least 150 mm Hg at baseline. This subgroup included 4,787 patients (57%) who achieved a systolic pressure of less than 140 mm Hg after 24 months on study treatment, 1,747 patients who achieved a systolic pressure of 140-149 mm Hg after 24 months, and 1,820 patients whose systolic pressure failed to fall below 150 mm Hg.
In multiple propensity score – adjusted analyses, the rate of cardiovascular death was a relative 34% higher among patients with an achieved systolic pressure of 140-149 mm Hg, compared with those whose pressures fell below 140 mm Hg. Also, the total rate of stroke was 89% higher among patients whose pressures stayed in the 140-149 mm Hg range, compared with patients who achieved lower pressures. The rate of nonfatal stroke was 70% higher among the 140-149 mm Hg subgroup. All three of these differences were statistically significant.
By several other outcome measures, the event rates did not significantly differ between the two achieved pressure subgroups, including all-cause deaths, total myocardial infarctions, rate of revascularization, or total events. In contrast, for most outcome measures assessed, patients whose systolic pressures remained at 150 mm Hg or greater had significantly more events than patients whose pressures fell below 140 mm Hg. Similar rates of adverse events were seen in each of the three achieved–blood pressure subgroups.
Dr. Bangalore and his coauthors acknowledged several limitations of their analysis: The data were analyzed by achieved blood pressure, which is likely influenced by baseline characteristics. INVEST was not designed to compare different blood pressure targets. The results were specific for patients aged 60 or older with coronary artery disease and a baseline systolic pressure greater than 150 mm Hg. And the researchers did not design the new analysis to assess the benefit of treating patients with a systolic blood pressure of 140-150 mm Hg.
INVEST was sponsored by BASF Pharma and Abbott. Dr. Bangalore said that he has served on advisory boards for Abbott and other drug companies. Dr. Gradman, Dr. O’Gara, and Dr. Antman had no relevant disclosures.
On Twitter @mitchelzoler
Elderly hypertensive patients who had coronary artery disease and a baseline systolic blood pressure greater than 150 mm Hg had significantly fewer cardiovascular deaths and strokes if they achieved a systolic pressure of less than 140 mm Hg, compared with similar patients who achieved a systolic pressure of 140-149 mm Hg.
The findings were noted in a post-hoc analysis of data collected from more than 8,000 patients in an international treatment study. The researchers compared outcomes for patients who achieved these two different levels of systolic pressure as a way to assess the new hypertension treatment goal of a systolic pressure less than 150 mm Hg in patients aged 60 years or older as recommended by the group originally appointed as the Eighth Joint National Committee (JNC 8) (JAMA 2014;311:507-20). Their finding that the patients who achieved the lower systolic pressure level of less than 140 mm Hg had a significantly lower rate of cardiovascular death and stroke as compared with patients whose systolic pressure remained in the 140-149 mm Hg range provided "important information to focus the risk-benefit discussion for patients who had on-treatment blood pressure in the 140-150 mm Hg range," wrote Dr. Sripal Bangalore, a cardiologist at New York University and his associates in an article published online on August 18 (J. Amer. Coll. Cardiol. 2014;311:784-93).
At least one expert questioned the ability of such a post-hoc analysis to address blood pressure goals in a meaningful way. "To adjudicate between a target of 140 mm Hg and 150 mm Hg it would be necessary to perform a prospective trial in which the population was randomized to these targets," commented Dr. Alan H. Gradman in an editorial that accompanied the new study (J. Amer. Coll. Cardiol. 2014;311:794-6). More aggressive blood pressure lowering could result in increased adverse events, he warned.
Despite these limitations, other experts hailed the new finding as important evidence against the recommendations of the former JNC 8 committee. "This study supports the concerns raised by many stakeholders, including the American College of Cardiology [ACC], the American Heart Association [AHA], and a number of the individual members of the JNC 8 panel (Ann. Intern. Med. 2014;160:499-503.), about the panel’s 2013 recommendations to raise blood pressure targets in older patients," said Dr. Patrick T. O’Gara, ACC president, and Dr. Elliott Antman, AHA president, in a joint written statement released to coincide with the new report’s publication.
"This new research suggests that raising the threshold for treatment of hypertension in patients 60 years of age or older with coronary artery disease may be detrimental to the best interest of patients and the public. It underscores ongoing concerns about adopting the unofficial 2013 targets as proposed by the panel originally appointed to write JNC 8," Dr. O’Gara and Dr. Antman said in their statement. Their statement added that "the ACC and AHA, working with the NHLBI [National Heart, Lung, and Blood Institute] are in the process of assembling the writing panel that will evaluate evidence from a variety of sources and provide a comprehensive update of the hypertension guideline."
The post-hoc analysis focused on a subgroup of 8,354 patients from among the 22,576 total patients enrolled in the (International Verapamil SR Trandolapril Study (INVEST), which enrolled patients aged 50 years or older with hypertension and pre-existing coronary artery disease at 862 centers in 14 countries (JAMA 2003;290:2805-16). Patients had been randomized to receive treatment with either a combination of verapamil sustained release plus trandolapril and hydrochlorothiazide if needed, or a combination of atenolol plus hydrochlorothiazide and trandolapril if needed, with a goal blood pressure on treatment of less than 140/90 mm Hg or less than 130/85 mmHg in patients with diabetes, renal impairment, or both. The primary, combined outcome of INVEST showed no significant difference in efficacy or safety between the two regimens compared in the study, and roughly 70% of patients in both treatment subgroups reached their goal blood pressures.
The new analysis selected the 8,354 patients who enrolled in INVEST at age 60 years or older and had a systolic blood pressure of at least 150 mm Hg at baseline. This subgroup included 4,787 patients (57%) who achieved a systolic pressure of less than 140 mm Hg after 24 months on study treatment, 1,747 patients who achieved a systolic pressure of 140-149 mm Hg after 24 months, and 1,820 patients whose systolic pressure failed to fall below 150 mm Hg.
In multiple propensity score – adjusted analyses, the rate of cardiovascular death was a relative 34% higher among patients with an achieved systolic pressure of 140-149 mm Hg, compared with those whose pressures fell below 140 mm Hg. Also, the total rate of stroke was 89% higher among patients whose pressures stayed in the 140-149 mm Hg range, compared with patients who achieved lower pressures. The rate of nonfatal stroke was 70% higher among the 140-149 mm Hg subgroup. All three of these differences were statistically significant.
By several other outcome measures, the event rates did not significantly differ between the two achieved pressure subgroups, including all-cause deaths, total myocardial infarctions, rate of revascularization, or total events. In contrast, for most outcome measures assessed, patients whose systolic pressures remained at 150 mm Hg or greater had significantly more events than patients whose pressures fell below 140 mm Hg. Similar rates of adverse events were seen in each of the three achieved–blood pressure subgroups.
Dr. Bangalore and his coauthors acknowledged several limitations of their analysis: The data were analyzed by achieved blood pressure, which is likely influenced by baseline characteristics. INVEST was not designed to compare different blood pressure targets. The results were specific for patients aged 60 or older with coronary artery disease and a baseline systolic pressure greater than 150 mm Hg. And the researchers did not design the new analysis to assess the benefit of treating patients with a systolic blood pressure of 140-150 mm Hg.
INVEST was sponsored by BASF Pharma and Abbott. Dr. Bangalore said that he has served on advisory boards for Abbott and other drug companies. Dr. Gradman, Dr. O’Gara, and Dr. Antman had no relevant disclosures.
On Twitter @mitchelzoler
FROM JACC
Key clinical point: When treatment drops systolic pressure below 140 mm Hg without causing adverse effects in patients over age 60 with coronary artery disease, the treatment can be maintained and the patient’s pressure kept at this lower level.
Major finding: The rate of cardiovascular death was a relative 34% higher among patients with an achieved systolic pressure of 140-149 mm Hg, compared with those whose pressures fell below 140 mm Hg.
Data source: Post-hoc analysis of data from 8,354 patients enrolled in INVEST, an international hypertension-treatment trial with 22,576 patients.
Disclosures: INVEST was sponsored by BASF Pharma and Abbott. Dr. Bangalore said that he has served on advisory boards for Abbott and several other drug companies. Dr. Gradman, Dr. O’Gara, and Dr. Antman had no relevant disclosures.
Bioabsorbable coronary scaffold use soars despite missing evidence
PARIS – Bioabsorbable vascular scaffolds have caught on in Europe and elsewhere in the world as an alternative to conventional metal stents for percutaneous coronary interventions in a wide spectrum of patient types.
By May 2014, more than 50,000 bioabsorbable vascular scaffolds (BVS) of the first brand to enter the European market – the Absorb stent made by Abbott Vascular – had gone into patients since the first placement in 2007, with the vast majority placed since this scaffold entered the European market in September 2012. Use of BVS in the United States remains investigational, while in the 20-plus months since BVS first became available in Europe other brands of BVS also have entered that market.
Despite growing BVS use in Europe, as well as in Asia and Central and South America, where they are also available, large-scale data on the consequences, especially long-term consequences, of BVS placement are mostly lacking, particularly data on how BVS perform when directly compared with drug-eluting metallic stents.
"Today we have uncontrolled dissemination of the technology without comparative data," Dr. Patrick W. Serruys, a pioneer in using BVS, said during a session at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
"We need evidence to know exactly what to do" with BVS, which patients to use them in, and when they are preferable to metallic stents, said Dr. Serruys, an interventional cardiologist and professor of medicine at the Thoraxcenter of Erasmus University in Rotterdam, the Netherlands.
"In the 7 years I’ve used them, in patients with 5-year follow-up, there have been no negative signals," he said in an interview, discussing his part of the sizeable anecdotal experience that BVS are safe and effective.
Several large trials comparing BVS with metal stents are in progress, including U.S. pivotal trials, but until these results are out, all that interventionalists have to gauge BVS performance are results from registries and uncontrolled trials. Given that limitation, so far the data look uniformly excellent.
"It is clear that emerging data from real-world, expanded-use registries suggest that use of the Absorb BVS is feasible and safe in a variety of patients, from low to high risk, and from simple to complex lesions," said Dr. Gregg W. Stone in a talk at the meeting. "But attention to procedural detail and technique is essential to optimize the outcomes. BVS are a little more complicated than metallic drug-eluting stents," said Dr. Stone, professor and director of cardiovascular research and education at Columbia University in New York.
"Data from large, randomized controlled trials are necessary to demonstrate whether BVS truly improve outcomes compared with best-in-class metallic stents. We are still early in the technology," he said. Dr. Stone is a lead investigator for one of the large U.S. randomized trials comparing the Absorb BVS to the current standard for percutaneous coronary interventions, a second-generation drug-eluting metallic stent.
Hints of what BVS may deliver
Hints of some of the potential benefits that BVS might provide appeared in results from a post-hoc, across-studies analysis that Dr. Stone presented in his talk. He compared the incidence of angina during the first year following PCI in 287 patients from the Absorb EXTEND study and 602 patients who received coronary stenting with the Xience second-generation everolimus-eluting metallic stent in the SPIRIT IV trial. The analysis matched patients in the Absorb arm with those in the Xience arm by propensity scoring.
The 1-year angina rates were 28% for the patients who received a metallic stent and 16% for those who received an absorbable scaffold, a 12% absolute difference, with a hazard ratio showing a 47% relative reduction in angina in patients who received the BVS, Dr. Stone reported. "There are a variety of mechanisms" by which BVS might reduce the rate of angina compared with metallic stents, including restoration of cyclic pulsatility in the treated arteries and improved vasomotion, he said.
The two outstanding clinical issues that dog PCI with second-generation drug-eluting metallic stents are the accrual of "very late" adverse events and the slow return of angina, with an average rate of target lesion failure with second-generation stents of about 1.8% year that continues out for at least 5 years, he said. "Can BVS flatten out the ongoing event rate" and improve measures like recurrent angina? Dr. Stone asked.
In another cross-study comparison, Dr. Serruys cited a recent, still unpublished analysis on which he collaborated that compared the 1-year outcomes of 102 patients with diabetes who received a BVS and 172 patients with diabetes who received a second-generation drug-eluting metallic stent. The 12-month cumulative rate of cardiovascular death, myocardial infarction, or ischemia-driven target lesion revascularization was 34.9% in the BVS recipients and 6.4% in patients who received state-of-the-art drug-eluting stents.
The working hypothesis behind BVS is that, by dissolving during a period of 12-24 months, the scaffold provides temporary bracing against restenosis but then disappears, allowing improved endovascular healing. During a separate session at EuroPCR, Dr. Charles A. Simonton, an interventional cardiologist and chief medical officer for Abbott Vascular, described the hypothesized impact that bioabsorbable coronary stents might have.
"Our vision is to convert interventional cardiology from mechanically oriented, plumbing-type interventions where you do mechanical things to open arteries" to a focus on tissue regeneration. "What we see when scaffolds resorb is that, at 3-5 years, the arteries look very different from what we see with a metal stent. The vessels show a highly functional neointimal layer that is functioning smooth muscle cells that protect against rupture in underlying plaque," he said.
Having BVS will allow interventional cardiologists to "transition from being mechanical plumbers to really being medical doctors. Not just opening lesions but putting in a treatment that can improve patients long term," Dr. Simonton said, presenting his best-case prediction of what BVS might achieve. "We need to generate a lot of data to make sure this is real," he added, in something of an understatement.
"There is late lumen enlargement with BVS. That is a clear fact," said Dr. Serruys.
The BVS experience
With data from studies that directly compared BVS and metallic stents still a few years off, the only indications of BVS performance now available come from either single- or multicenter registries. The thrust of much of these data presented at EuroPCR was that BVS have performed well as interventionalists have moved in the months since BVS became available for routine use to placing them in sicker patients and in patients with more complex coronary lesions.
The largest registry is ABSORB First, a voluntary-reporting registry that began in January 2013 and involves about 90 centers in countries where Absorb is commercially available in Europe, Asia, New Zealand, and Columbia. Dr. Eric Eeckhout, a cardiologist at Batiment Hospital in Lausanne, Switzerland, reported procedural and 30-day outcome data for the first 800 of the 1,305 patients enrolled in the registry as of May 2014.
Unlike previously reported multicenter series of patients getting BVS, these patients included 46% with at least two affected coronary arteries, 29% with stable angina, 15% with unstable angina, and 35% with an acute or recent MI. Significant calcification was present in 20% of the treated coronaries, 12% of the treated coronary stenoses were located at bifurcations, 12% of the coronaries featured extensive tortuosity, 11% had total occlusions, and 6% had ostial lesions. Lesion complexity scoring based on the American Heart Association and American College of Cardiology method rated 24% of the stenoses type C lesions and another 23% rated as B2 lesions. In addition, 38% of the lesions were at least 20 mm long.
Despite the challenging anatomy that many patients featured, device success occurred in 99% of the patients and procedure success in 98%, reported Dr. Eeckhout. Bailout treatment with a metal stent was needed in five patients (0.6%). During the 30 days following treatment the 800 patients had no deaths, and two cases (0.3%) of definite or probably stent thrombosis with both episodes occurring 1-30 days after scaffold placement – low rates given the complexity of the patients and lesions treated.
"These findings suggest the early safety and performance of Absorb in complex, real-world patients in daily PCI use," Dr. Eeckhout said. "Probably the operators [who treated these 800 patients] knew how to handle a BVS and they were able to expand to more complex pathology without an impact on safety or efficacy," he noted, adding that the "excellent results reflect the increasing experience of interventional cardiologists who use BVS; complexity did not impact efficacy or safety."
"These were "extraordinarily good outcomes in very complex patients," commented Dr. Stone, but he cautioned that the follow-up so far has been brief and that this was a "self-reported" registry, so it is possible that some operators failed to report some of their complications.
Other registry data reported at EuroPCR had longer follow-up, in some cases out to as long as 1 year, but in smaller patient sets and at fewer centers. These series represent "real world" use of BVS at these centers since Absorb came onto the European market in September 2012. Soon after BVS became commercially available, the interventionalists at these centers decided to use them in whichever patients they could regardless of their clinical status – in patients with acute coronary syndrome as well as stable patients, and regardless of lesion type or location. This contrasted with the initial, preapproval trials of BVS that excluded unstable patients and patients with complex coronary lesions or lesions in challenging sites, such as bifurcations or at an arterial ostium.
Findings from several ongoing registries were reported at EuroPCR:
• A group of six German centers had 1-year follow-up on 181 patients. Two thirds had class B2 or C lesions, 16% of lesions were calcified, and 14% involved side branches. These patients had a 5% rate of major adverse coronary events in the first year with no episodes of stent thrombosis, a result comparable with the pivotal trials for second-generation drug-eluting metallic stents as well as the early BVS studies that enrolled patients with simpler lesions, noted Dr. Thomas Schmitz, a cardiologist at the Contilla Heart and Vascular Center in Essen, Germany.
• Single-center experience at the Thoraxcenter in Rotterdam included 6-month follow-up on the first 180 of what are now more than 600 patients who have received BVS at the Thoraxcenter since it came onto the market. The series featured 48% of patients with calcified lesions, 41% with type B2 or C lesions, 38% of patients with multivessel disease, and lesion lengths up to 25 mm. Many patients received a BVS acutely after a ST-segment MI. Device success occurred in 98% of patients. The 6-month rate of major adverse coronary events was 3.3%, with definite or probable stent thrombosis in 2%, and 2% required target-lesion revascularization during 6 months of follow-up. "We had very good results in a complex group of patients that is getting closer to the real world by including patients with unstable and stable angina and ST-elevation myocardial infarction," said Dr. Robert-Jan van Geuns, a cardiologist at the Thoraxcenter. "We got stentlike results using a temporary scaffold. But we want to improve the flexibility of the BVS, reduce strut thickness, and make the procedures easier," he said.
• Interventionalists at Ferrarotto University Hospital in Catania, Italy, decided in March 2013 to start placing BVS when they could "regardless of lesion location or complexity." By this past May they had treated 367 lesions in 289 patients with BVS and had 6-month follow-up on 169 patients and 12-month follow-up on 48, said Dr. Corrado Tamburino, professor of cardiology and director of clinical cardiology at Ferrarotto. Half of their BVS patients had acute coronary syndrome, including 18% with ST-elevation MI, 17% with unstable angina, and 15% with non-STEMI. Half the treated lesions were class C or B2, 27% of patients had two or more lesions, 16% were at bifurcations, 9% were chronic total occlusions, and the mean lesion length was 21 mm. The operators successfully delivered BVS into 98% of patients. Among patients with 6-months’ follow-up, the incidence of major adverse coronary events was 2%, with no episodes of stent thrombosis after 30 days. The series included two deaths: one in a patient who stopped dual-antiplatelet therapy (DAPT) after 25 days and another in a patient who had an arterial dissection. All the BVS thromboses occurred within the first 30 days, "meaning it’s a technical issue. You need to always do lesion preparation, proper sizing [of the BVS for the artery it is entering], and post dilatation. If you do all that, you reduce the risk of thrombosis," he explained. The findings showed that placing "BVS is feasible and safe in the real world, and is effective at midterm follow-up. Stent thrombosis is not an issue if the procedural steps are respected. We expect similar results after 1 and 2 years, but we need to wait and be cautious," Dr. Tamburino said.
• Several centers in Poland began last year to enroll consecutive patients with acute coronary syndrome and culprit lesions that were amenable to BVS treatment. By May they had 1-year follow-up data on 94 BVS recipients, mostly patients with unstable angina or non–ST-elevation MI (NSTEMI) but also several with STEMI. The series had 100% success for BVS delivery and device success, with a 3% rate of major adverse coronary events while the patients remained initially hospitalized. After 12 months the patients had no deaths, three MIs, one target vessel revascularization, and one non–target vessel revascularization. Stent thrombosis occurred in one patient who prematurely stopped DAPT. The series so far has had no need for target-lesion revascularization. The 1-year results "show excellent outcomes. I’m amazed the results have been so good," said Dr. Dariusz Dudek, a cardiologist at University Hospital in Krakow, Poland.
• Interventionalists at two centers in Prague saw 311 consecutive patients with STEMI from December 2012 through the end of April 2014, and among these patients 79 (25%) met the group’s criteria for treatment with BVS. The operators excluded patients with a high likelihood of dying during the 2-3 year period for full BVS disappearance, with a Killip class of III or IV. They also excluded patients with an infarct-related artery less than 2.3 mm in diameter or more than 3.7 mm because of the limited size availability of BVS and because of the dangers from using incorrectly sized BVS. The 79 patients who received a BVS were a third of all patients in the series who received some form of PCI. An analysis presented by Dr. Petr Widimský, professor and head of the department of cardiology at Vinohrady Teaching Hospital in Prague, compared 9-month outcomes for these 79 patients who received BVS and 184 patients from the same series who also had Killip class I or II disease at the time of PCI. The remaining 48 patients from the series had Killip class III or IV disease and were excluded. After 9 months, event-free survival rate was 95% in the BVS recipients and 90% in the patients who received PCI with metallic stents, a nonsignificant difference. In the BVS group, 76 of the 79 patients successfully received a total of 85 BVS. Seventy-two of the 76 who received a BVS had an "ideal" procedural result, with no dissections and achievement of full blood flow through the treated lesion; the other four patients had slightly compromised blood flow after BVS placement. The 76 patients who successfully received a BVS had one death 4 hours after PCI as a result of septal rupture, and another BVS thrombosis 3 days after the patient stopped DAPT. A stroke or transient ischemic attack occurred in three patients. "We conclude that using BVS to treat acute STEMI is feasible and safe, and with currently available BVS sizes can be done in 25%-33% of STEMI patients. Having a 4.0-mm diameter BVS would substantially increase the number of patients we could treat," Dr. Widimský said.
• Cardiologists at one center in Barcelona treated 67 chronic total occlusions (CTO) since BVS became available, 49 of which could be crossed with a guidewire. Of these, the interventionalists could place a BVS in 38 patients; they excluded the other 11 patients because of excessive coronary-artery tortuosity, bifurcated lesions, or inappropriate vessel size. Among the 38 CTO patients who received BVS, three required bail-out PCI with a metallic stent because of a dissection. Average CTO length was 19 mm, and the average total lesion length was 36 mm. At 1-month follow-up, there were no deaths, no MI, no need for additional revascularization, and no BVS thrombosis. The same pattern held among the 33 patients from this group who had 6-months’ follow-up. "These surprising, extremely good results seem too good to be true," said Dr. Antonio Serra, an interventional cardiologist at Hospital Sant Pau in Barcelona. Results from the series so far "show that BVS is extremely feasible and safe for treating complex CTO. We report excellent patency and safety. The results at 6 months were outstanding."
Limits on BVS use
BVS are not for everyone. They get ruled out by substantial tortuosity of the route from the catheterization site to the affected coronary because BVS are stiffer than metallic stents and less amenable to pass through a winding arterial route. Patients need to be good candidates to remain on DAPT for at least 12 months, and the target-vessel diameter cannot veer too far from the currently available BVS diameters of 2.5, 3.0, or 3.5 mm. In most centers, these and other exclusion criteria usually rule out 75%-85% of patients, said several experienced BVS interventionalists at the meeting.
"I want patients who are healthy enough so they stay on DAPT. A BVS is not where you try 3 or 6 months of DAPT," said Dr. van Geuns. He also said that he would not use BVS for treating venous grafts, nor for in-stent restenosis because the existing metal likely would prevent late-lumen enlargement. And in some patients BVS cannot reach the lesion site despite aggressive predilation and vessel preparation.
"We’ve seen increasing breadth of use in the real world. It’s not yet true 100% all-comers, because appropriately people are learning where to use this technology," commented Dr. Stone.
"Our data represent a bias because it is not 100% of our patient population. They are the patients whom we think it is possible to implant a BVS," Dr. Tamburino said.
BVS "technology is not forgiving," Dr. van Guens pointed out. "If you leave a small lumen you will get a small lumen, and that can cause flow disturbances and perhaps a thrombus. Sometimes we are too eager with this technology. I think we can get to 100% in the future, but we should go slow because we are not there yet." He added that the number of BVS recipients and the rate of stent thrombosis were so far too small to allow meaningful analysis of which BVS recipients faced the greatest risk of stent thrombosis. Possible factors include inadequate post dilitation of the BVS, overexpansion of a BVS, and premature halt of DAPT.
"BVS is now feasible to use for every case" as long as the target vessel is no more than 10% smaller than the available BVS, Dr. Julinda Mehilli, an interventional cardiologist at the German Heart Center in Munich, said in a talk at the meeting. Preferred locations are long lesions in distal segments. Operators should be cautious using BVS to treat bifurcations, arteries with pronounced tortuosity, or arteries with moderate to severe calcification. When two BVS are needed in tandem, they should be placed as close to edge to edge as possible. She also stressed the need for systematic attention to every phase of BVS preparation and delivery, starting from selecting the appropriate BVS diameter to match the target vessel, predilation of the lesion and vessel and removal of calcium, gradual inflation of the BVS itself, and then followed by post dilitation of the BVS with a noncompliant balloon, ideally for at least 20 seconds.
"Now that we have BVS, it is the device of choice for chronic total occlusions because long term there is healing of the vessel walls when we use BVS. We often see long-term malopposition of stent struts in chronic total occlusions. With BVS the scaffold will disappear," eliminating the malopposition risk, Dr. Mehilli said.
Dr. Stone had no relevant disclosures, but he is lead investigator for an ongoing trial studying Abbot Vascular’s Absorb BVS. Dr. Serruys, Dr. Eeckhout, Dr. Schmitz, Dr. Dudek, and Dr. Serra had no disclosures. Dr. Tamburino is a consultant to Abbott Vascular and three other companies. Dr. Simonton is an employee of Abbott Vascular. Dr. van Geuns has received honoraria and research support from Abbott Vascular and other companies. Dr. Widimský has received honoraria from Abbott Vascular and other companies. Dr. Mehilli has received lecture fees from Abbott Vascular and other companies.
On Twitter @mitchelzoler
PARIS – Bioabsorbable vascular scaffolds have caught on in Europe and elsewhere in the world as an alternative to conventional metal stents for percutaneous coronary interventions in a wide spectrum of patient types.
By May 2014, more than 50,000 bioabsorbable vascular scaffolds (BVS) of the first brand to enter the European market – the Absorb stent made by Abbott Vascular – had gone into patients since the first placement in 2007, with the vast majority placed since this scaffold entered the European market in September 2012. Use of BVS in the United States remains investigational, while in the 20-plus months since BVS first became available in Europe other brands of BVS also have entered that market.
Despite growing BVS use in Europe, as well as in Asia and Central and South America, where they are also available, large-scale data on the consequences, especially long-term consequences, of BVS placement are mostly lacking, particularly data on how BVS perform when directly compared with drug-eluting metallic stents.
"Today we have uncontrolled dissemination of the technology without comparative data," Dr. Patrick W. Serruys, a pioneer in using BVS, said during a session at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
"We need evidence to know exactly what to do" with BVS, which patients to use them in, and when they are preferable to metallic stents, said Dr. Serruys, an interventional cardiologist and professor of medicine at the Thoraxcenter of Erasmus University in Rotterdam, the Netherlands.
"In the 7 years I’ve used them, in patients with 5-year follow-up, there have been no negative signals," he said in an interview, discussing his part of the sizeable anecdotal experience that BVS are safe and effective.
Several large trials comparing BVS with metal stents are in progress, including U.S. pivotal trials, but until these results are out, all that interventionalists have to gauge BVS performance are results from registries and uncontrolled trials. Given that limitation, so far the data look uniformly excellent.
"It is clear that emerging data from real-world, expanded-use registries suggest that use of the Absorb BVS is feasible and safe in a variety of patients, from low to high risk, and from simple to complex lesions," said Dr. Gregg W. Stone in a talk at the meeting. "But attention to procedural detail and technique is essential to optimize the outcomes. BVS are a little more complicated than metallic drug-eluting stents," said Dr. Stone, professor and director of cardiovascular research and education at Columbia University in New York.
"Data from large, randomized controlled trials are necessary to demonstrate whether BVS truly improve outcomes compared with best-in-class metallic stents. We are still early in the technology," he said. Dr. Stone is a lead investigator for one of the large U.S. randomized trials comparing the Absorb BVS to the current standard for percutaneous coronary interventions, a second-generation drug-eluting metallic stent.
Hints of what BVS may deliver
Hints of some of the potential benefits that BVS might provide appeared in results from a post-hoc, across-studies analysis that Dr. Stone presented in his talk. He compared the incidence of angina during the first year following PCI in 287 patients from the Absorb EXTEND study and 602 patients who received coronary stenting with the Xience second-generation everolimus-eluting metallic stent in the SPIRIT IV trial. The analysis matched patients in the Absorb arm with those in the Xience arm by propensity scoring.
The 1-year angina rates were 28% for the patients who received a metallic stent and 16% for those who received an absorbable scaffold, a 12% absolute difference, with a hazard ratio showing a 47% relative reduction in angina in patients who received the BVS, Dr. Stone reported. "There are a variety of mechanisms" by which BVS might reduce the rate of angina compared with metallic stents, including restoration of cyclic pulsatility in the treated arteries and improved vasomotion, he said.
The two outstanding clinical issues that dog PCI with second-generation drug-eluting metallic stents are the accrual of "very late" adverse events and the slow return of angina, with an average rate of target lesion failure with second-generation stents of about 1.8% year that continues out for at least 5 years, he said. "Can BVS flatten out the ongoing event rate" and improve measures like recurrent angina? Dr. Stone asked.
In another cross-study comparison, Dr. Serruys cited a recent, still unpublished analysis on which he collaborated that compared the 1-year outcomes of 102 patients with diabetes who received a BVS and 172 patients with diabetes who received a second-generation drug-eluting metallic stent. The 12-month cumulative rate of cardiovascular death, myocardial infarction, or ischemia-driven target lesion revascularization was 34.9% in the BVS recipients and 6.4% in patients who received state-of-the-art drug-eluting stents.
The working hypothesis behind BVS is that, by dissolving during a period of 12-24 months, the scaffold provides temporary bracing against restenosis but then disappears, allowing improved endovascular healing. During a separate session at EuroPCR, Dr. Charles A. Simonton, an interventional cardiologist and chief medical officer for Abbott Vascular, described the hypothesized impact that bioabsorbable coronary stents might have.
"Our vision is to convert interventional cardiology from mechanically oriented, plumbing-type interventions where you do mechanical things to open arteries" to a focus on tissue regeneration. "What we see when scaffolds resorb is that, at 3-5 years, the arteries look very different from what we see with a metal stent. The vessels show a highly functional neointimal layer that is functioning smooth muscle cells that protect against rupture in underlying plaque," he said.
Having BVS will allow interventional cardiologists to "transition from being mechanical plumbers to really being medical doctors. Not just opening lesions but putting in a treatment that can improve patients long term," Dr. Simonton said, presenting his best-case prediction of what BVS might achieve. "We need to generate a lot of data to make sure this is real," he added, in something of an understatement.
"There is late lumen enlargement with BVS. That is a clear fact," said Dr. Serruys.
The BVS experience
With data from studies that directly compared BVS and metallic stents still a few years off, the only indications of BVS performance now available come from either single- or multicenter registries. The thrust of much of these data presented at EuroPCR was that BVS have performed well as interventionalists have moved in the months since BVS became available for routine use to placing them in sicker patients and in patients with more complex coronary lesions.
The largest registry is ABSORB First, a voluntary-reporting registry that began in January 2013 and involves about 90 centers in countries where Absorb is commercially available in Europe, Asia, New Zealand, and Columbia. Dr. Eric Eeckhout, a cardiologist at Batiment Hospital in Lausanne, Switzerland, reported procedural and 30-day outcome data for the first 800 of the 1,305 patients enrolled in the registry as of May 2014.
Unlike previously reported multicenter series of patients getting BVS, these patients included 46% with at least two affected coronary arteries, 29% with stable angina, 15% with unstable angina, and 35% with an acute or recent MI. Significant calcification was present in 20% of the treated coronaries, 12% of the treated coronary stenoses were located at bifurcations, 12% of the coronaries featured extensive tortuosity, 11% had total occlusions, and 6% had ostial lesions. Lesion complexity scoring based on the American Heart Association and American College of Cardiology method rated 24% of the stenoses type C lesions and another 23% rated as B2 lesions. In addition, 38% of the lesions were at least 20 mm long.
Despite the challenging anatomy that many patients featured, device success occurred in 99% of the patients and procedure success in 98%, reported Dr. Eeckhout. Bailout treatment with a metal stent was needed in five patients (0.6%). During the 30 days following treatment the 800 patients had no deaths, and two cases (0.3%) of definite or probably stent thrombosis with both episodes occurring 1-30 days after scaffold placement – low rates given the complexity of the patients and lesions treated.
"These findings suggest the early safety and performance of Absorb in complex, real-world patients in daily PCI use," Dr. Eeckhout said. "Probably the operators [who treated these 800 patients] knew how to handle a BVS and they were able to expand to more complex pathology without an impact on safety or efficacy," he noted, adding that the "excellent results reflect the increasing experience of interventional cardiologists who use BVS; complexity did not impact efficacy or safety."
"These were "extraordinarily good outcomes in very complex patients," commented Dr. Stone, but he cautioned that the follow-up so far has been brief and that this was a "self-reported" registry, so it is possible that some operators failed to report some of their complications.
Other registry data reported at EuroPCR had longer follow-up, in some cases out to as long as 1 year, but in smaller patient sets and at fewer centers. These series represent "real world" use of BVS at these centers since Absorb came onto the European market in September 2012. Soon after BVS became commercially available, the interventionalists at these centers decided to use them in whichever patients they could regardless of their clinical status – in patients with acute coronary syndrome as well as stable patients, and regardless of lesion type or location. This contrasted with the initial, preapproval trials of BVS that excluded unstable patients and patients with complex coronary lesions or lesions in challenging sites, such as bifurcations or at an arterial ostium.
Findings from several ongoing registries were reported at EuroPCR:
• A group of six German centers had 1-year follow-up on 181 patients. Two thirds had class B2 or C lesions, 16% of lesions were calcified, and 14% involved side branches. These patients had a 5% rate of major adverse coronary events in the first year with no episodes of stent thrombosis, a result comparable with the pivotal trials for second-generation drug-eluting metallic stents as well as the early BVS studies that enrolled patients with simpler lesions, noted Dr. Thomas Schmitz, a cardiologist at the Contilla Heart and Vascular Center in Essen, Germany.
• Single-center experience at the Thoraxcenter in Rotterdam included 6-month follow-up on the first 180 of what are now more than 600 patients who have received BVS at the Thoraxcenter since it came onto the market. The series featured 48% of patients with calcified lesions, 41% with type B2 or C lesions, 38% of patients with multivessel disease, and lesion lengths up to 25 mm. Many patients received a BVS acutely after a ST-segment MI. Device success occurred in 98% of patients. The 6-month rate of major adverse coronary events was 3.3%, with definite or probable stent thrombosis in 2%, and 2% required target-lesion revascularization during 6 months of follow-up. "We had very good results in a complex group of patients that is getting closer to the real world by including patients with unstable and stable angina and ST-elevation myocardial infarction," said Dr. Robert-Jan van Geuns, a cardiologist at the Thoraxcenter. "We got stentlike results using a temporary scaffold. But we want to improve the flexibility of the BVS, reduce strut thickness, and make the procedures easier," he said.
• Interventionalists at Ferrarotto University Hospital in Catania, Italy, decided in March 2013 to start placing BVS when they could "regardless of lesion location or complexity." By this past May they had treated 367 lesions in 289 patients with BVS and had 6-month follow-up on 169 patients and 12-month follow-up on 48, said Dr. Corrado Tamburino, professor of cardiology and director of clinical cardiology at Ferrarotto. Half of their BVS patients had acute coronary syndrome, including 18% with ST-elevation MI, 17% with unstable angina, and 15% with non-STEMI. Half the treated lesions were class C or B2, 27% of patients had two or more lesions, 16% were at bifurcations, 9% were chronic total occlusions, and the mean lesion length was 21 mm. The operators successfully delivered BVS into 98% of patients. Among patients with 6-months’ follow-up, the incidence of major adverse coronary events was 2%, with no episodes of stent thrombosis after 30 days. The series included two deaths: one in a patient who stopped dual-antiplatelet therapy (DAPT) after 25 days and another in a patient who had an arterial dissection. All the BVS thromboses occurred within the first 30 days, "meaning it’s a technical issue. You need to always do lesion preparation, proper sizing [of the BVS for the artery it is entering], and post dilatation. If you do all that, you reduce the risk of thrombosis," he explained. The findings showed that placing "BVS is feasible and safe in the real world, and is effective at midterm follow-up. Stent thrombosis is not an issue if the procedural steps are respected. We expect similar results after 1 and 2 years, but we need to wait and be cautious," Dr. Tamburino said.
• Several centers in Poland began last year to enroll consecutive patients with acute coronary syndrome and culprit lesions that were amenable to BVS treatment. By May they had 1-year follow-up data on 94 BVS recipients, mostly patients with unstable angina or non–ST-elevation MI (NSTEMI) but also several with STEMI. The series had 100% success for BVS delivery and device success, with a 3% rate of major adverse coronary events while the patients remained initially hospitalized. After 12 months the patients had no deaths, three MIs, one target vessel revascularization, and one non–target vessel revascularization. Stent thrombosis occurred in one patient who prematurely stopped DAPT. The series so far has had no need for target-lesion revascularization. The 1-year results "show excellent outcomes. I’m amazed the results have been so good," said Dr. Dariusz Dudek, a cardiologist at University Hospital in Krakow, Poland.
• Interventionalists at two centers in Prague saw 311 consecutive patients with STEMI from December 2012 through the end of April 2014, and among these patients 79 (25%) met the group’s criteria for treatment with BVS. The operators excluded patients with a high likelihood of dying during the 2-3 year period for full BVS disappearance, with a Killip class of III or IV. They also excluded patients with an infarct-related artery less than 2.3 mm in diameter or more than 3.7 mm because of the limited size availability of BVS and because of the dangers from using incorrectly sized BVS. The 79 patients who received a BVS were a third of all patients in the series who received some form of PCI. An analysis presented by Dr. Petr Widimský, professor and head of the department of cardiology at Vinohrady Teaching Hospital in Prague, compared 9-month outcomes for these 79 patients who received BVS and 184 patients from the same series who also had Killip class I or II disease at the time of PCI. The remaining 48 patients from the series had Killip class III or IV disease and were excluded. After 9 months, event-free survival rate was 95% in the BVS recipients and 90% in the patients who received PCI with metallic stents, a nonsignificant difference. In the BVS group, 76 of the 79 patients successfully received a total of 85 BVS. Seventy-two of the 76 who received a BVS had an "ideal" procedural result, with no dissections and achievement of full blood flow through the treated lesion; the other four patients had slightly compromised blood flow after BVS placement. The 76 patients who successfully received a BVS had one death 4 hours after PCI as a result of septal rupture, and another BVS thrombosis 3 days after the patient stopped DAPT. A stroke or transient ischemic attack occurred in three patients. "We conclude that using BVS to treat acute STEMI is feasible and safe, and with currently available BVS sizes can be done in 25%-33% of STEMI patients. Having a 4.0-mm diameter BVS would substantially increase the number of patients we could treat," Dr. Widimský said.
• Cardiologists at one center in Barcelona treated 67 chronic total occlusions (CTO) since BVS became available, 49 of which could be crossed with a guidewire. Of these, the interventionalists could place a BVS in 38 patients; they excluded the other 11 patients because of excessive coronary-artery tortuosity, bifurcated lesions, or inappropriate vessel size. Among the 38 CTO patients who received BVS, three required bail-out PCI with a metallic stent because of a dissection. Average CTO length was 19 mm, and the average total lesion length was 36 mm. At 1-month follow-up, there were no deaths, no MI, no need for additional revascularization, and no BVS thrombosis. The same pattern held among the 33 patients from this group who had 6-months’ follow-up. "These surprising, extremely good results seem too good to be true," said Dr. Antonio Serra, an interventional cardiologist at Hospital Sant Pau in Barcelona. Results from the series so far "show that BVS is extremely feasible and safe for treating complex CTO. We report excellent patency and safety. The results at 6 months were outstanding."
Limits on BVS use
BVS are not for everyone. They get ruled out by substantial tortuosity of the route from the catheterization site to the affected coronary because BVS are stiffer than metallic stents and less amenable to pass through a winding arterial route. Patients need to be good candidates to remain on DAPT for at least 12 months, and the target-vessel diameter cannot veer too far from the currently available BVS diameters of 2.5, 3.0, or 3.5 mm. In most centers, these and other exclusion criteria usually rule out 75%-85% of patients, said several experienced BVS interventionalists at the meeting.
"I want patients who are healthy enough so they stay on DAPT. A BVS is not where you try 3 or 6 months of DAPT," said Dr. van Geuns. He also said that he would not use BVS for treating venous grafts, nor for in-stent restenosis because the existing metal likely would prevent late-lumen enlargement. And in some patients BVS cannot reach the lesion site despite aggressive predilation and vessel preparation.
"We’ve seen increasing breadth of use in the real world. It’s not yet true 100% all-comers, because appropriately people are learning where to use this technology," commented Dr. Stone.
"Our data represent a bias because it is not 100% of our patient population. They are the patients whom we think it is possible to implant a BVS," Dr. Tamburino said.
BVS "technology is not forgiving," Dr. van Guens pointed out. "If you leave a small lumen you will get a small lumen, and that can cause flow disturbances and perhaps a thrombus. Sometimes we are too eager with this technology. I think we can get to 100% in the future, but we should go slow because we are not there yet." He added that the number of BVS recipients and the rate of stent thrombosis were so far too small to allow meaningful analysis of which BVS recipients faced the greatest risk of stent thrombosis. Possible factors include inadequate post dilitation of the BVS, overexpansion of a BVS, and premature halt of DAPT.
"BVS is now feasible to use for every case" as long as the target vessel is no more than 10% smaller than the available BVS, Dr. Julinda Mehilli, an interventional cardiologist at the German Heart Center in Munich, said in a talk at the meeting. Preferred locations are long lesions in distal segments. Operators should be cautious using BVS to treat bifurcations, arteries with pronounced tortuosity, or arteries with moderate to severe calcification. When two BVS are needed in tandem, they should be placed as close to edge to edge as possible. She also stressed the need for systematic attention to every phase of BVS preparation and delivery, starting from selecting the appropriate BVS diameter to match the target vessel, predilation of the lesion and vessel and removal of calcium, gradual inflation of the BVS itself, and then followed by post dilitation of the BVS with a noncompliant balloon, ideally for at least 20 seconds.
"Now that we have BVS, it is the device of choice for chronic total occlusions because long term there is healing of the vessel walls when we use BVS. We often see long-term malopposition of stent struts in chronic total occlusions. With BVS the scaffold will disappear," eliminating the malopposition risk, Dr. Mehilli said.
Dr. Stone had no relevant disclosures, but he is lead investigator for an ongoing trial studying Abbot Vascular’s Absorb BVS. Dr. Serruys, Dr. Eeckhout, Dr. Schmitz, Dr. Dudek, and Dr. Serra had no disclosures. Dr. Tamburino is a consultant to Abbott Vascular and three other companies. Dr. Simonton is an employee of Abbott Vascular. Dr. van Geuns has received honoraria and research support from Abbott Vascular and other companies. Dr. Widimský has received honoraria from Abbott Vascular and other companies. Dr. Mehilli has received lecture fees from Abbott Vascular and other companies.
On Twitter @mitchelzoler
PARIS – Bioabsorbable vascular scaffolds have caught on in Europe and elsewhere in the world as an alternative to conventional metal stents for percutaneous coronary interventions in a wide spectrum of patient types.
By May 2014, more than 50,000 bioabsorbable vascular scaffolds (BVS) of the first brand to enter the European market – the Absorb stent made by Abbott Vascular – had gone into patients since the first placement in 2007, with the vast majority placed since this scaffold entered the European market in September 2012. Use of BVS in the United States remains investigational, while in the 20-plus months since BVS first became available in Europe other brands of BVS also have entered that market.
Despite growing BVS use in Europe, as well as in Asia and Central and South America, where they are also available, large-scale data on the consequences, especially long-term consequences, of BVS placement are mostly lacking, particularly data on how BVS perform when directly compared with drug-eluting metallic stents.
"Today we have uncontrolled dissemination of the technology without comparative data," Dr. Patrick W. Serruys, a pioneer in using BVS, said during a session at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
"We need evidence to know exactly what to do" with BVS, which patients to use them in, and when they are preferable to metallic stents, said Dr. Serruys, an interventional cardiologist and professor of medicine at the Thoraxcenter of Erasmus University in Rotterdam, the Netherlands.
"In the 7 years I’ve used them, in patients with 5-year follow-up, there have been no negative signals," he said in an interview, discussing his part of the sizeable anecdotal experience that BVS are safe and effective.
Several large trials comparing BVS with metal stents are in progress, including U.S. pivotal trials, but until these results are out, all that interventionalists have to gauge BVS performance are results from registries and uncontrolled trials. Given that limitation, so far the data look uniformly excellent.
"It is clear that emerging data from real-world, expanded-use registries suggest that use of the Absorb BVS is feasible and safe in a variety of patients, from low to high risk, and from simple to complex lesions," said Dr. Gregg W. Stone in a talk at the meeting. "But attention to procedural detail and technique is essential to optimize the outcomes. BVS are a little more complicated than metallic drug-eluting stents," said Dr. Stone, professor and director of cardiovascular research and education at Columbia University in New York.
"Data from large, randomized controlled trials are necessary to demonstrate whether BVS truly improve outcomes compared with best-in-class metallic stents. We are still early in the technology," he said. Dr. Stone is a lead investigator for one of the large U.S. randomized trials comparing the Absorb BVS to the current standard for percutaneous coronary interventions, a second-generation drug-eluting metallic stent.
Hints of what BVS may deliver
Hints of some of the potential benefits that BVS might provide appeared in results from a post-hoc, across-studies analysis that Dr. Stone presented in his talk. He compared the incidence of angina during the first year following PCI in 287 patients from the Absorb EXTEND study and 602 patients who received coronary stenting with the Xience second-generation everolimus-eluting metallic stent in the SPIRIT IV trial. The analysis matched patients in the Absorb arm with those in the Xience arm by propensity scoring.
The 1-year angina rates were 28% for the patients who received a metallic stent and 16% for those who received an absorbable scaffold, a 12% absolute difference, with a hazard ratio showing a 47% relative reduction in angina in patients who received the BVS, Dr. Stone reported. "There are a variety of mechanisms" by which BVS might reduce the rate of angina compared with metallic stents, including restoration of cyclic pulsatility in the treated arteries and improved vasomotion, he said.
The two outstanding clinical issues that dog PCI with second-generation drug-eluting metallic stents are the accrual of "very late" adverse events and the slow return of angina, with an average rate of target lesion failure with second-generation stents of about 1.8% year that continues out for at least 5 years, he said. "Can BVS flatten out the ongoing event rate" and improve measures like recurrent angina? Dr. Stone asked.
In another cross-study comparison, Dr. Serruys cited a recent, still unpublished analysis on which he collaborated that compared the 1-year outcomes of 102 patients with diabetes who received a BVS and 172 patients with diabetes who received a second-generation drug-eluting metallic stent. The 12-month cumulative rate of cardiovascular death, myocardial infarction, or ischemia-driven target lesion revascularization was 34.9% in the BVS recipients and 6.4% in patients who received state-of-the-art drug-eluting stents.
The working hypothesis behind BVS is that, by dissolving during a period of 12-24 months, the scaffold provides temporary bracing against restenosis but then disappears, allowing improved endovascular healing. During a separate session at EuroPCR, Dr. Charles A. Simonton, an interventional cardiologist and chief medical officer for Abbott Vascular, described the hypothesized impact that bioabsorbable coronary stents might have.
"Our vision is to convert interventional cardiology from mechanically oriented, plumbing-type interventions where you do mechanical things to open arteries" to a focus on tissue regeneration. "What we see when scaffolds resorb is that, at 3-5 years, the arteries look very different from what we see with a metal stent. The vessels show a highly functional neointimal layer that is functioning smooth muscle cells that protect against rupture in underlying plaque," he said.
Having BVS will allow interventional cardiologists to "transition from being mechanical plumbers to really being medical doctors. Not just opening lesions but putting in a treatment that can improve patients long term," Dr. Simonton said, presenting his best-case prediction of what BVS might achieve. "We need to generate a lot of data to make sure this is real," he added, in something of an understatement.
"There is late lumen enlargement with BVS. That is a clear fact," said Dr. Serruys.
The BVS experience
With data from studies that directly compared BVS and metallic stents still a few years off, the only indications of BVS performance now available come from either single- or multicenter registries. The thrust of much of these data presented at EuroPCR was that BVS have performed well as interventionalists have moved in the months since BVS became available for routine use to placing them in sicker patients and in patients with more complex coronary lesions.
The largest registry is ABSORB First, a voluntary-reporting registry that began in January 2013 and involves about 90 centers in countries where Absorb is commercially available in Europe, Asia, New Zealand, and Columbia. Dr. Eric Eeckhout, a cardiologist at Batiment Hospital in Lausanne, Switzerland, reported procedural and 30-day outcome data for the first 800 of the 1,305 patients enrolled in the registry as of May 2014.
Unlike previously reported multicenter series of patients getting BVS, these patients included 46% with at least two affected coronary arteries, 29% with stable angina, 15% with unstable angina, and 35% with an acute or recent MI. Significant calcification was present in 20% of the treated coronaries, 12% of the treated coronary stenoses were located at bifurcations, 12% of the coronaries featured extensive tortuosity, 11% had total occlusions, and 6% had ostial lesions. Lesion complexity scoring based on the American Heart Association and American College of Cardiology method rated 24% of the stenoses type C lesions and another 23% rated as B2 lesions. In addition, 38% of the lesions were at least 20 mm long.
Despite the challenging anatomy that many patients featured, device success occurred in 99% of the patients and procedure success in 98%, reported Dr. Eeckhout. Bailout treatment with a metal stent was needed in five patients (0.6%). During the 30 days following treatment the 800 patients had no deaths, and two cases (0.3%) of definite or probably stent thrombosis with both episodes occurring 1-30 days after scaffold placement – low rates given the complexity of the patients and lesions treated.
"These findings suggest the early safety and performance of Absorb in complex, real-world patients in daily PCI use," Dr. Eeckhout said. "Probably the operators [who treated these 800 patients] knew how to handle a BVS and they were able to expand to more complex pathology without an impact on safety or efficacy," he noted, adding that the "excellent results reflect the increasing experience of interventional cardiologists who use BVS; complexity did not impact efficacy or safety."
"These were "extraordinarily good outcomes in very complex patients," commented Dr. Stone, but he cautioned that the follow-up so far has been brief and that this was a "self-reported" registry, so it is possible that some operators failed to report some of their complications.
Other registry data reported at EuroPCR had longer follow-up, in some cases out to as long as 1 year, but in smaller patient sets and at fewer centers. These series represent "real world" use of BVS at these centers since Absorb came onto the European market in September 2012. Soon after BVS became commercially available, the interventionalists at these centers decided to use them in whichever patients they could regardless of their clinical status – in patients with acute coronary syndrome as well as stable patients, and regardless of lesion type or location. This contrasted with the initial, preapproval trials of BVS that excluded unstable patients and patients with complex coronary lesions or lesions in challenging sites, such as bifurcations or at an arterial ostium.
Findings from several ongoing registries were reported at EuroPCR:
• A group of six German centers had 1-year follow-up on 181 patients. Two thirds had class B2 or C lesions, 16% of lesions were calcified, and 14% involved side branches. These patients had a 5% rate of major adverse coronary events in the first year with no episodes of stent thrombosis, a result comparable with the pivotal trials for second-generation drug-eluting metallic stents as well as the early BVS studies that enrolled patients with simpler lesions, noted Dr. Thomas Schmitz, a cardiologist at the Contilla Heart and Vascular Center in Essen, Germany.
• Single-center experience at the Thoraxcenter in Rotterdam included 6-month follow-up on the first 180 of what are now more than 600 patients who have received BVS at the Thoraxcenter since it came onto the market. The series featured 48% of patients with calcified lesions, 41% with type B2 or C lesions, 38% of patients with multivessel disease, and lesion lengths up to 25 mm. Many patients received a BVS acutely after a ST-segment MI. Device success occurred in 98% of patients. The 6-month rate of major adverse coronary events was 3.3%, with definite or probable stent thrombosis in 2%, and 2% required target-lesion revascularization during 6 months of follow-up. "We had very good results in a complex group of patients that is getting closer to the real world by including patients with unstable and stable angina and ST-elevation myocardial infarction," said Dr. Robert-Jan van Geuns, a cardiologist at the Thoraxcenter. "We got stentlike results using a temporary scaffold. But we want to improve the flexibility of the BVS, reduce strut thickness, and make the procedures easier," he said.
• Interventionalists at Ferrarotto University Hospital in Catania, Italy, decided in March 2013 to start placing BVS when they could "regardless of lesion location or complexity." By this past May they had treated 367 lesions in 289 patients with BVS and had 6-month follow-up on 169 patients and 12-month follow-up on 48, said Dr. Corrado Tamburino, professor of cardiology and director of clinical cardiology at Ferrarotto. Half of their BVS patients had acute coronary syndrome, including 18% with ST-elevation MI, 17% with unstable angina, and 15% with non-STEMI. Half the treated lesions were class C or B2, 27% of patients had two or more lesions, 16% were at bifurcations, 9% were chronic total occlusions, and the mean lesion length was 21 mm. The operators successfully delivered BVS into 98% of patients. Among patients with 6-months’ follow-up, the incidence of major adverse coronary events was 2%, with no episodes of stent thrombosis after 30 days. The series included two deaths: one in a patient who stopped dual-antiplatelet therapy (DAPT) after 25 days and another in a patient who had an arterial dissection. All the BVS thromboses occurred within the first 30 days, "meaning it’s a technical issue. You need to always do lesion preparation, proper sizing [of the BVS for the artery it is entering], and post dilatation. If you do all that, you reduce the risk of thrombosis," he explained. The findings showed that placing "BVS is feasible and safe in the real world, and is effective at midterm follow-up. Stent thrombosis is not an issue if the procedural steps are respected. We expect similar results after 1 and 2 years, but we need to wait and be cautious," Dr. Tamburino said.
• Several centers in Poland began last year to enroll consecutive patients with acute coronary syndrome and culprit lesions that were amenable to BVS treatment. By May they had 1-year follow-up data on 94 BVS recipients, mostly patients with unstable angina or non–ST-elevation MI (NSTEMI) but also several with STEMI. The series had 100% success for BVS delivery and device success, with a 3% rate of major adverse coronary events while the patients remained initially hospitalized. After 12 months the patients had no deaths, three MIs, one target vessel revascularization, and one non–target vessel revascularization. Stent thrombosis occurred in one patient who prematurely stopped DAPT. The series so far has had no need for target-lesion revascularization. The 1-year results "show excellent outcomes. I’m amazed the results have been so good," said Dr. Dariusz Dudek, a cardiologist at University Hospital in Krakow, Poland.
• Interventionalists at two centers in Prague saw 311 consecutive patients with STEMI from December 2012 through the end of April 2014, and among these patients 79 (25%) met the group’s criteria for treatment with BVS. The operators excluded patients with a high likelihood of dying during the 2-3 year period for full BVS disappearance, with a Killip class of III or IV. They also excluded patients with an infarct-related artery less than 2.3 mm in diameter or more than 3.7 mm because of the limited size availability of BVS and because of the dangers from using incorrectly sized BVS. The 79 patients who received a BVS were a third of all patients in the series who received some form of PCI. An analysis presented by Dr. Petr Widimský, professor and head of the department of cardiology at Vinohrady Teaching Hospital in Prague, compared 9-month outcomes for these 79 patients who received BVS and 184 patients from the same series who also had Killip class I or II disease at the time of PCI. The remaining 48 patients from the series had Killip class III or IV disease and were excluded. After 9 months, event-free survival rate was 95% in the BVS recipients and 90% in the patients who received PCI with metallic stents, a nonsignificant difference. In the BVS group, 76 of the 79 patients successfully received a total of 85 BVS. Seventy-two of the 76 who received a BVS had an "ideal" procedural result, with no dissections and achievement of full blood flow through the treated lesion; the other four patients had slightly compromised blood flow after BVS placement. The 76 patients who successfully received a BVS had one death 4 hours after PCI as a result of septal rupture, and another BVS thrombosis 3 days after the patient stopped DAPT. A stroke or transient ischemic attack occurred in three patients. "We conclude that using BVS to treat acute STEMI is feasible and safe, and with currently available BVS sizes can be done in 25%-33% of STEMI patients. Having a 4.0-mm diameter BVS would substantially increase the number of patients we could treat," Dr. Widimský said.
• Cardiologists at one center in Barcelona treated 67 chronic total occlusions (CTO) since BVS became available, 49 of which could be crossed with a guidewire. Of these, the interventionalists could place a BVS in 38 patients; they excluded the other 11 patients because of excessive coronary-artery tortuosity, bifurcated lesions, or inappropriate vessel size. Among the 38 CTO patients who received BVS, three required bail-out PCI with a metallic stent because of a dissection. Average CTO length was 19 mm, and the average total lesion length was 36 mm. At 1-month follow-up, there were no deaths, no MI, no need for additional revascularization, and no BVS thrombosis. The same pattern held among the 33 patients from this group who had 6-months’ follow-up. "These surprising, extremely good results seem too good to be true," said Dr. Antonio Serra, an interventional cardiologist at Hospital Sant Pau in Barcelona. Results from the series so far "show that BVS is extremely feasible and safe for treating complex CTO. We report excellent patency and safety. The results at 6 months were outstanding."
Limits on BVS use
BVS are not for everyone. They get ruled out by substantial tortuosity of the route from the catheterization site to the affected coronary because BVS are stiffer than metallic stents and less amenable to pass through a winding arterial route. Patients need to be good candidates to remain on DAPT for at least 12 months, and the target-vessel diameter cannot veer too far from the currently available BVS diameters of 2.5, 3.0, or 3.5 mm. In most centers, these and other exclusion criteria usually rule out 75%-85% of patients, said several experienced BVS interventionalists at the meeting.
"I want patients who are healthy enough so they stay on DAPT. A BVS is not where you try 3 or 6 months of DAPT," said Dr. van Geuns. He also said that he would not use BVS for treating venous grafts, nor for in-stent restenosis because the existing metal likely would prevent late-lumen enlargement. And in some patients BVS cannot reach the lesion site despite aggressive predilation and vessel preparation.
"We’ve seen increasing breadth of use in the real world. It’s not yet true 100% all-comers, because appropriately people are learning where to use this technology," commented Dr. Stone.
"Our data represent a bias because it is not 100% of our patient population. They are the patients whom we think it is possible to implant a BVS," Dr. Tamburino said.
BVS "technology is not forgiving," Dr. van Guens pointed out. "If you leave a small lumen you will get a small lumen, and that can cause flow disturbances and perhaps a thrombus. Sometimes we are too eager with this technology. I think we can get to 100% in the future, but we should go slow because we are not there yet." He added that the number of BVS recipients and the rate of stent thrombosis were so far too small to allow meaningful analysis of which BVS recipients faced the greatest risk of stent thrombosis. Possible factors include inadequate post dilitation of the BVS, overexpansion of a BVS, and premature halt of DAPT.
"BVS is now feasible to use for every case" as long as the target vessel is no more than 10% smaller than the available BVS, Dr. Julinda Mehilli, an interventional cardiologist at the German Heart Center in Munich, said in a talk at the meeting. Preferred locations are long lesions in distal segments. Operators should be cautious using BVS to treat bifurcations, arteries with pronounced tortuosity, or arteries with moderate to severe calcification. When two BVS are needed in tandem, they should be placed as close to edge to edge as possible. She also stressed the need for systematic attention to every phase of BVS preparation and delivery, starting from selecting the appropriate BVS diameter to match the target vessel, predilation of the lesion and vessel and removal of calcium, gradual inflation of the BVS itself, and then followed by post dilitation of the BVS with a noncompliant balloon, ideally for at least 20 seconds.
"Now that we have BVS, it is the device of choice for chronic total occlusions because long term there is healing of the vessel walls when we use BVS. We often see long-term malopposition of stent struts in chronic total occlusions. With BVS the scaffold will disappear," eliminating the malopposition risk, Dr. Mehilli said.
Dr. Stone had no relevant disclosures, but he is lead investigator for an ongoing trial studying Abbot Vascular’s Absorb BVS. Dr. Serruys, Dr. Eeckhout, Dr. Schmitz, Dr. Dudek, and Dr. Serra had no disclosures. Dr. Tamburino is a consultant to Abbott Vascular and three other companies. Dr. Simonton is an employee of Abbott Vascular. Dr. van Geuns has received honoraria and research support from Abbott Vascular and other companies. Dr. Widimský has received honoraria from Abbott Vascular and other companies. Dr. Mehilli has received lecture fees from Abbott Vascular and other companies.
On Twitter @mitchelzoler
EXPERT ANALYSIS FROM EUROPCR 2014
Quick freezing oocytes and embryos transforms assisted reproduction
MUNICH – Vitrification, quick freezing oocytes or embryos with concentrated cyroprotectant and no ice crystal formation to better preserve viability, has been around clinically some 15 years, but now is gathering steam and may have already helped downshift the rate of triplets or higher pregnancies for women undergoing assisted reproduction.
While vitrification of all embryos fertilized in vitro has not proven superior to fresh embryo transfers and may even be inferior for older women whose eggs may not be as forgiving of cryopreservation, its use has grown. It offers the unique advantage over fresh embryos of easier coordination between the in vitro fertilization process and subsequent transfer to the woman who will carry the pregnancy. Vitrification has also made possible true oocyte banking, be it autologous for fertility preservation, or for oocyte donation.
"It is now possible to freeze embryos with a 90% survival rate; with slow freezing there was 50%-60% survival. Now we have a technique that is really good, and it may cause a paradigm shift toward elective freezing," said Dr. Anja Pinborg, a professor of ob.gyn. at Hvidovre (Denmark) Hospital.
Most clinicians still use the approach of placing an embryo during the same cycle when the oocyte was harvested, "the way it’s always been done, but at many clinics now they see that with vitrification they can get pregnancy rates that are similar to or even better than fresh embryo transfer. I think they will go more and more to frozen embryos," Dr. Pinborg said in an interview during the annual meeting of the European Society of Human Reproduction and Embryology.
For now the trend is specific for women younger than 35 years. Safety and efficacy of vitrification for embryos made from oocytes taken from older women has not yet been studied, cautioned Dr. Pinborg and others.
"All we know about cryopreservation is from young women with young oocytes. We don’t know what will happen with oocytes from women who are 41 or 42, who are the types of patients I see," said Dr. Richard J. Paulson, professor of ob.gyn. and chief of the division of reproductive endocrinology and infertility at the University of Southern California and director of USC Fertility in Los Angeles. "Vitrification is so good and embryos come out of it so well that the benefit from improved endometrial receptivity outweighs any change in embryo quality. The timing coordinates so much better."
The biggest impact of vitrification for embryo freezing may be its ability to successfully cryopreserve blastocysts. "We will see many more programs going to 5-day embryos" for implantation, and this will allow preimplantation genetic diagnosis (PGD), predicted Dr. Paulson during an interview at the meeting. "It looks like you can do pretty good trophectoderm biopsy on a vitrified blastocyst, so you can place only chromosomally normal embryos. That will reduce miscarriages." So far, only a handful of groups have presented case reports using this approach, with "very good numbers," he said.
Vitrification’s impact on embryo transfer number
Despite lingering questions about vitrification safety and limited knowledge of its efficacy for embryos begun from oocytes from older women, the most recent worldwide and European data on patterns of assisted reproductive technology show increases in frozen embryo use and decreases in triplets or greater multiples.
Worldwide, frozen embryo transfers grew from 10% of all in vitro fertilization or sperm injection procedures in 1991 to 28% in 2010, the most recent year with worldwide data available. Transfers of one or two embryos grew from 20% of all in vitro procedures done in North America in 1998 to 68% in 2010. In Australia in 2010, the rate of one- or two-embryo transfers stood at 98%.
"The trend is for more frozen transfers, and for reducing multiple rates. I think that is a very positive development," said Dr. David Adamson, a reproductive endocrinologist who practices in Palo Alto, Calif., and presented at the meeting worldwide data collected by the International Committee Monitoring Assisted Reproductive Technologies (ICMART). "The pregnancy rate is continuing to go up, but the multiple rate is dropping. We hope that will continue," he said in an interview.
"There is no question that because vitrification works so much better than slow freezing, clinics have more confidence to put in fewer embryos and still have viability," he said. "We’ll see more frozen embryo transfers in the next few years. When you decrease the number of embryos transferred, you increase the likelihood that you’ll need to follow with a frozen embryo transfer, but now you have higher-quality embryos."
A similar shift to fewer embryos transferred at a time appeared in the exclusively European registry with data derived from 33 countries that reported complete or partial data to the European IVF Monitoring Program, a project organized by ESHRE. The prevalence of triplet or greater deliveries from in vitro pregnancies fell in Europe from about 4% in 1999 to less than 1% in 2011. By 2011, roughly 28% of IVF or sperm-injected pregnancies involved placement of two embryos, and about 57% involved a single embryo, reported Dr. Markus S. Kupka, an ob.gyn. and reproductive medicine specialist who practices in Hamburg, Germany. Frozen embryos involved 31% of transfers in 2011 in Europe. European data for 2010 were included in the worldwide tallies reported by Dr. Adamson.
Vitrification’s safety
Despite growing use, some questions remain about vitrification’s impact on children, although the concern has not been strong enough to dim enthusiasm.
The most striking safety signal is a consistent, 50% increased rate of babies born following vitrification who are large for gestational age. Dr. Pinborg also cited a possibly increased rate of maternal preeclampsia.
"It’s only a 50% increased risk for large for gestational age with frozen embryo transfer. We should not pay too much attention to this, but it reminds us that when we freeze, it may have an effect on the children" Dr. Pinborg said in an interview.
"We can’t say that vitrified embryos do better than after fresh embryo transfer, but they have an altered risk profile." Frozen embryos have a reduced risk for preterm birth and small for gestational age, compared with fresh embryo transfers, but an increased risk of large for gestational age (LGA). "And the risk is small," stressed Dr. Pinborg. "When you stimulate ovulation, you harvest 10-fold as many oocytes than occur with a normal cycle, and the woman’s estrogen and progesterone levels are very high. So, you could ask: Why should we ever place an embryo during an oocyte harvesting cycle? We did that because we had poor freezing methods, but with vitrification we have a way to use preserved embryos and there is better cycle synchronization."
In addition, seeing increased rates of LGA babies following frozen embryo transfer may result from unadjusted confounding by, for example, the embryo’s age when cryopreservation occurred that could influence epigenetic changes that happen early after fertilization or maternal disorders such as polycystic ovary syndrome, Dr. Paulson said.
Safety aside, vitrification is very effective for assisted reproduction because any decrement in viability is counterbalanced by the advantage of placing embryos squarely during a natural cycle, these experts said.
Vitrification and oocytes
Vitrification does not stop with embryos. The impact it has had on oocyte banking has arguably been even greater.
Creating multiple embryos at one time is problematic, especially in the United States, Dr. Paulson said. "The obvious thing we should do is freeze oocytes. This approach would create another significant advantage. Routine vitrification at the oocyte stage rather than after embryos are made "would make donor oocytes a reality. You would be able to buy frozen oocytes the same way as sperm. Results from good randomized, controlled trials show that outcomes with cryopreserved oocytes are as good as with fresh oocytes, which was never possible with slow freezing."
"Vitrification created the ability to have oocyte banking. It has been a breakthrough in assisted reproductive technology," said Ana Cobo, Ph.D., director of the cryopreservation laboratory at the IVI Foundation in Valencia, Spain. Cryopreserving oocytes was essentially impossible before vitrification became available, she said in an interview. Studies run by her group showed that the vitrification process has no detectable effect on oocyte viability, embryo viability, or the health of the child. It also made oocyte donation much more feasible by eliminating the need for cycle synchrony between the donor and recipient.
The success rate of in vitro fertilization using a donated oocyte is generally much higher than for an autologous oocyte because "we use a highly selected population of young, healthy women with the best quality oocytes. In assisted reproductive technology, oocyte quality means a lot," Dr. Cobo said.
Dr. Pinborg said that she has received research grants from MSD and Ferring. Dr. Paulson said that he has been a speaker on behalf of Ferring and served on advisory boards for Origio and Cooper Surgical. Dr. Adamson said that he had received honoraria from or consulted for Bayer, Glycotype, and Ziva Medical, that he owns stock in Advanced Reproductive Care, and has received research support from Auxogyn and LabCorp. Dr. Kupka said he had no disclosures. Dr. Cobo had no disclosures.
On Twitter @mitchelzoler
MUNICH – Vitrification, quick freezing oocytes or embryos with concentrated cyroprotectant and no ice crystal formation to better preserve viability, has been around clinically some 15 years, but now is gathering steam and may have already helped downshift the rate of triplets or higher pregnancies for women undergoing assisted reproduction.
While vitrification of all embryos fertilized in vitro has not proven superior to fresh embryo transfers and may even be inferior for older women whose eggs may not be as forgiving of cryopreservation, its use has grown. It offers the unique advantage over fresh embryos of easier coordination between the in vitro fertilization process and subsequent transfer to the woman who will carry the pregnancy. Vitrification has also made possible true oocyte banking, be it autologous for fertility preservation, or for oocyte donation.
"It is now possible to freeze embryos with a 90% survival rate; with slow freezing there was 50%-60% survival. Now we have a technique that is really good, and it may cause a paradigm shift toward elective freezing," said Dr. Anja Pinborg, a professor of ob.gyn. at Hvidovre (Denmark) Hospital.
Most clinicians still use the approach of placing an embryo during the same cycle when the oocyte was harvested, "the way it’s always been done, but at many clinics now they see that with vitrification they can get pregnancy rates that are similar to or even better than fresh embryo transfer. I think they will go more and more to frozen embryos," Dr. Pinborg said in an interview during the annual meeting of the European Society of Human Reproduction and Embryology.
For now the trend is specific for women younger than 35 years. Safety and efficacy of vitrification for embryos made from oocytes taken from older women has not yet been studied, cautioned Dr. Pinborg and others.
"All we know about cryopreservation is from young women with young oocytes. We don’t know what will happen with oocytes from women who are 41 or 42, who are the types of patients I see," said Dr. Richard J. Paulson, professor of ob.gyn. and chief of the division of reproductive endocrinology and infertility at the University of Southern California and director of USC Fertility in Los Angeles. "Vitrification is so good and embryos come out of it so well that the benefit from improved endometrial receptivity outweighs any change in embryo quality. The timing coordinates so much better."
The biggest impact of vitrification for embryo freezing may be its ability to successfully cryopreserve blastocysts. "We will see many more programs going to 5-day embryos" for implantation, and this will allow preimplantation genetic diagnosis (PGD), predicted Dr. Paulson during an interview at the meeting. "It looks like you can do pretty good trophectoderm biopsy on a vitrified blastocyst, so you can place only chromosomally normal embryos. That will reduce miscarriages." So far, only a handful of groups have presented case reports using this approach, with "very good numbers," he said.
Vitrification’s impact on embryo transfer number
Despite lingering questions about vitrification safety and limited knowledge of its efficacy for embryos begun from oocytes from older women, the most recent worldwide and European data on patterns of assisted reproductive technology show increases in frozen embryo use and decreases in triplets or greater multiples.
Worldwide, frozen embryo transfers grew from 10% of all in vitro fertilization or sperm injection procedures in 1991 to 28% in 2010, the most recent year with worldwide data available. Transfers of one or two embryos grew from 20% of all in vitro procedures done in North America in 1998 to 68% in 2010. In Australia in 2010, the rate of one- or two-embryo transfers stood at 98%.
"The trend is for more frozen transfers, and for reducing multiple rates. I think that is a very positive development," said Dr. David Adamson, a reproductive endocrinologist who practices in Palo Alto, Calif., and presented at the meeting worldwide data collected by the International Committee Monitoring Assisted Reproductive Technologies (ICMART). "The pregnancy rate is continuing to go up, but the multiple rate is dropping. We hope that will continue," he said in an interview.
"There is no question that because vitrification works so much better than slow freezing, clinics have more confidence to put in fewer embryos and still have viability," he said. "We’ll see more frozen embryo transfers in the next few years. When you decrease the number of embryos transferred, you increase the likelihood that you’ll need to follow with a frozen embryo transfer, but now you have higher-quality embryos."
A similar shift to fewer embryos transferred at a time appeared in the exclusively European registry with data derived from 33 countries that reported complete or partial data to the European IVF Monitoring Program, a project organized by ESHRE. The prevalence of triplet or greater deliveries from in vitro pregnancies fell in Europe from about 4% in 1999 to less than 1% in 2011. By 2011, roughly 28% of IVF or sperm-injected pregnancies involved placement of two embryos, and about 57% involved a single embryo, reported Dr. Markus S. Kupka, an ob.gyn. and reproductive medicine specialist who practices in Hamburg, Germany. Frozen embryos involved 31% of transfers in 2011 in Europe. European data for 2010 were included in the worldwide tallies reported by Dr. Adamson.
Vitrification’s safety
Despite growing use, some questions remain about vitrification’s impact on children, although the concern has not been strong enough to dim enthusiasm.
The most striking safety signal is a consistent, 50% increased rate of babies born following vitrification who are large for gestational age. Dr. Pinborg also cited a possibly increased rate of maternal preeclampsia.
"It’s only a 50% increased risk for large for gestational age with frozen embryo transfer. We should not pay too much attention to this, but it reminds us that when we freeze, it may have an effect on the children" Dr. Pinborg said in an interview.
"We can’t say that vitrified embryos do better than after fresh embryo transfer, but they have an altered risk profile." Frozen embryos have a reduced risk for preterm birth and small for gestational age, compared with fresh embryo transfers, but an increased risk of large for gestational age (LGA). "And the risk is small," stressed Dr. Pinborg. "When you stimulate ovulation, you harvest 10-fold as many oocytes than occur with a normal cycle, and the woman’s estrogen and progesterone levels are very high. So, you could ask: Why should we ever place an embryo during an oocyte harvesting cycle? We did that because we had poor freezing methods, but with vitrification we have a way to use preserved embryos and there is better cycle synchronization."
In addition, seeing increased rates of LGA babies following frozen embryo transfer may result from unadjusted confounding by, for example, the embryo’s age when cryopreservation occurred that could influence epigenetic changes that happen early after fertilization or maternal disorders such as polycystic ovary syndrome, Dr. Paulson said.
Safety aside, vitrification is very effective for assisted reproduction because any decrement in viability is counterbalanced by the advantage of placing embryos squarely during a natural cycle, these experts said.
Vitrification and oocytes
Vitrification does not stop with embryos. The impact it has had on oocyte banking has arguably been even greater.
Creating multiple embryos at one time is problematic, especially in the United States, Dr. Paulson said. "The obvious thing we should do is freeze oocytes. This approach would create another significant advantage. Routine vitrification at the oocyte stage rather than after embryos are made "would make donor oocytes a reality. You would be able to buy frozen oocytes the same way as sperm. Results from good randomized, controlled trials show that outcomes with cryopreserved oocytes are as good as with fresh oocytes, which was never possible with slow freezing."
"Vitrification created the ability to have oocyte banking. It has been a breakthrough in assisted reproductive technology," said Ana Cobo, Ph.D., director of the cryopreservation laboratory at the IVI Foundation in Valencia, Spain. Cryopreserving oocytes was essentially impossible before vitrification became available, she said in an interview. Studies run by her group showed that the vitrification process has no detectable effect on oocyte viability, embryo viability, or the health of the child. It also made oocyte donation much more feasible by eliminating the need for cycle synchrony between the donor and recipient.
The success rate of in vitro fertilization using a donated oocyte is generally much higher than for an autologous oocyte because "we use a highly selected population of young, healthy women with the best quality oocytes. In assisted reproductive technology, oocyte quality means a lot," Dr. Cobo said.
Dr. Pinborg said that she has received research grants from MSD and Ferring. Dr. Paulson said that he has been a speaker on behalf of Ferring and served on advisory boards for Origio and Cooper Surgical. Dr. Adamson said that he had received honoraria from or consulted for Bayer, Glycotype, and Ziva Medical, that he owns stock in Advanced Reproductive Care, and has received research support from Auxogyn and LabCorp. Dr. Kupka said he had no disclosures. Dr. Cobo had no disclosures.
On Twitter @mitchelzoler
MUNICH – Vitrification, quick freezing oocytes or embryos with concentrated cyroprotectant and no ice crystal formation to better preserve viability, has been around clinically some 15 years, but now is gathering steam and may have already helped downshift the rate of triplets or higher pregnancies for women undergoing assisted reproduction.
While vitrification of all embryos fertilized in vitro has not proven superior to fresh embryo transfers and may even be inferior for older women whose eggs may not be as forgiving of cryopreservation, its use has grown. It offers the unique advantage over fresh embryos of easier coordination between the in vitro fertilization process and subsequent transfer to the woman who will carry the pregnancy. Vitrification has also made possible true oocyte banking, be it autologous for fertility preservation, or for oocyte donation.
"It is now possible to freeze embryos with a 90% survival rate; with slow freezing there was 50%-60% survival. Now we have a technique that is really good, and it may cause a paradigm shift toward elective freezing," said Dr. Anja Pinborg, a professor of ob.gyn. at Hvidovre (Denmark) Hospital.
Most clinicians still use the approach of placing an embryo during the same cycle when the oocyte was harvested, "the way it’s always been done, but at many clinics now they see that with vitrification they can get pregnancy rates that are similar to or even better than fresh embryo transfer. I think they will go more and more to frozen embryos," Dr. Pinborg said in an interview during the annual meeting of the European Society of Human Reproduction and Embryology.
For now the trend is specific for women younger than 35 years. Safety and efficacy of vitrification for embryos made from oocytes taken from older women has not yet been studied, cautioned Dr. Pinborg and others.
"All we know about cryopreservation is from young women with young oocytes. We don’t know what will happen with oocytes from women who are 41 or 42, who are the types of patients I see," said Dr. Richard J. Paulson, professor of ob.gyn. and chief of the division of reproductive endocrinology and infertility at the University of Southern California and director of USC Fertility in Los Angeles. "Vitrification is so good and embryos come out of it so well that the benefit from improved endometrial receptivity outweighs any change in embryo quality. The timing coordinates so much better."
The biggest impact of vitrification for embryo freezing may be its ability to successfully cryopreserve blastocysts. "We will see many more programs going to 5-day embryos" for implantation, and this will allow preimplantation genetic diagnosis (PGD), predicted Dr. Paulson during an interview at the meeting. "It looks like you can do pretty good trophectoderm biopsy on a vitrified blastocyst, so you can place only chromosomally normal embryos. That will reduce miscarriages." So far, only a handful of groups have presented case reports using this approach, with "very good numbers," he said.
Vitrification’s impact on embryo transfer number
Despite lingering questions about vitrification safety and limited knowledge of its efficacy for embryos begun from oocytes from older women, the most recent worldwide and European data on patterns of assisted reproductive technology show increases in frozen embryo use and decreases in triplets or greater multiples.
Worldwide, frozen embryo transfers grew from 10% of all in vitro fertilization or sperm injection procedures in 1991 to 28% in 2010, the most recent year with worldwide data available. Transfers of one or two embryos grew from 20% of all in vitro procedures done in North America in 1998 to 68% in 2010. In Australia in 2010, the rate of one- or two-embryo transfers stood at 98%.
"The trend is for more frozen transfers, and for reducing multiple rates. I think that is a very positive development," said Dr. David Adamson, a reproductive endocrinologist who practices in Palo Alto, Calif., and presented at the meeting worldwide data collected by the International Committee Monitoring Assisted Reproductive Technologies (ICMART). "The pregnancy rate is continuing to go up, but the multiple rate is dropping. We hope that will continue," he said in an interview.
"There is no question that because vitrification works so much better than slow freezing, clinics have more confidence to put in fewer embryos and still have viability," he said. "We’ll see more frozen embryo transfers in the next few years. When you decrease the number of embryos transferred, you increase the likelihood that you’ll need to follow with a frozen embryo transfer, but now you have higher-quality embryos."
A similar shift to fewer embryos transferred at a time appeared in the exclusively European registry with data derived from 33 countries that reported complete or partial data to the European IVF Monitoring Program, a project organized by ESHRE. The prevalence of triplet or greater deliveries from in vitro pregnancies fell in Europe from about 4% in 1999 to less than 1% in 2011. By 2011, roughly 28% of IVF or sperm-injected pregnancies involved placement of two embryos, and about 57% involved a single embryo, reported Dr. Markus S. Kupka, an ob.gyn. and reproductive medicine specialist who practices in Hamburg, Germany. Frozen embryos involved 31% of transfers in 2011 in Europe. European data for 2010 were included in the worldwide tallies reported by Dr. Adamson.
Vitrification’s safety
Despite growing use, some questions remain about vitrification’s impact on children, although the concern has not been strong enough to dim enthusiasm.
The most striking safety signal is a consistent, 50% increased rate of babies born following vitrification who are large for gestational age. Dr. Pinborg also cited a possibly increased rate of maternal preeclampsia.
"It’s only a 50% increased risk for large for gestational age with frozen embryo transfer. We should not pay too much attention to this, but it reminds us that when we freeze, it may have an effect on the children" Dr. Pinborg said in an interview.
"We can’t say that vitrified embryos do better than after fresh embryo transfer, but they have an altered risk profile." Frozen embryos have a reduced risk for preterm birth and small for gestational age, compared with fresh embryo transfers, but an increased risk of large for gestational age (LGA). "And the risk is small," stressed Dr. Pinborg. "When you stimulate ovulation, you harvest 10-fold as many oocytes than occur with a normal cycle, and the woman’s estrogen and progesterone levels are very high. So, you could ask: Why should we ever place an embryo during an oocyte harvesting cycle? We did that because we had poor freezing methods, but with vitrification we have a way to use preserved embryos and there is better cycle synchronization."
In addition, seeing increased rates of LGA babies following frozen embryo transfer may result from unadjusted confounding by, for example, the embryo’s age when cryopreservation occurred that could influence epigenetic changes that happen early after fertilization or maternal disorders such as polycystic ovary syndrome, Dr. Paulson said.
Safety aside, vitrification is very effective for assisted reproduction because any decrement in viability is counterbalanced by the advantage of placing embryos squarely during a natural cycle, these experts said.
Vitrification and oocytes
Vitrification does not stop with embryos. The impact it has had on oocyte banking has arguably been even greater.
Creating multiple embryos at one time is problematic, especially in the United States, Dr. Paulson said. "The obvious thing we should do is freeze oocytes. This approach would create another significant advantage. Routine vitrification at the oocyte stage rather than after embryos are made "would make donor oocytes a reality. You would be able to buy frozen oocytes the same way as sperm. Results from good randomized, controlled trials show that outcomes with cryopreserved oocytes are as good as with fresh oocytes, which was never possible with slow freezing."
"Vitrification created the ability to have oocyte banking. It has been a breakthrough in assisted reproductive technology," said Ana Cobo, Ph.D., director of the cryopreservation laboratory at the IVI Foundation in Valencia, Spain. Cryopreserving oocytes was essentially impossible before vitrification became available, she said in an interview. Studies run by her group showed that the vitrification process has no detectable effect on oocyte viability, embryo viability, or the health of the child. It also made oocyte donation much more feasible by eliminating the need for cycle synchrony between the donor and recipient.
The success rate of in vitro fertilization using a donated oocyte is generally much higher than for an autologous oocyte because "we use a highly selected population of young, healthy women with the best quality oocytes. In assisted reproductive technology, oocyte quality means a lot," Dr. Cobo said.
Dr. Pinborg said that she has received research grants from MSD and Ferring. Dr. Paulson said that he has been a speaker on behalf of Ferring and served on advisory boards for Origio and Cooper Surgical. Dr. Adamson said that he had received honoraria from or consulted for Bayer, Glycotype, and Ziva Medical, that he owns stock in Advanced Reproductive Care, and has received research support from Auxogyn and LabCorp. Dr. Kupka said he had no disclosures. Dr. Cobo had no disclosures.
On Twitter @mitchelzoler
EXPERT ANALYSIS FROM ESHRE 2014
ESHRE writes first premature ovarian insufficiency guidelines
MUNICH – A working group of the European Society of Human Reproduction and Embryology drafted the first international guidelines for managing premature ovarian insufficiency.
Once formally released, by late this year or soon after, the guidelines should "improve clinical practice and awareness of the condition," which affects roughly 1% of women, said Dr. Melanie Davies, a gynecologist at University College London Hospital and a working-group member who presented a few key items from the draft guidelines during a session at the society’s annual meeting.
Women with premature ovarian insufficiency (POI) sometimes are treated for one aspect of POI by a physician "without full realization of the condition and its implications," Dr. Davies said in an interview. For example, "there have been fatal cases of women with Turner syndrome who received donor oocytes," the disorder posing the greatest risk to women receiving donor eggs. "This is a good example of where the guidelines may increase awareness," she said.
One draft excerpt Dr. Davies presented advises physicians and patients that candidates for donor oocytes suspected of having POI should undergo prior to receiving eggs a full investigation for thyroid and adrenal function and karyotype. "Pregnancy in some women can be of such high risk that clinicians may consider egg donation life threatening and inappropriate," she said.
For women with Turner syndrome, the guidelines call for assessment by a cardiologist with a special interest in adult congenital heart disease, as well as a general medical and endocrine examination prior to pregnancy. During pregnancy, these women have an elevated risk for obstetric complications and should be managed in an obstetric unit designed for high-risk pregnancies with involvement of a cardiologist.
The draft defines POI as depletion of follicular activity before age 40 years with menstrual disturbance, raised gonadotropin levels, and low estradiol levels. These women should understand that no interventions are proven to reliably increase ovarian activity and their natural conception rate. But physicians also can reassure these women that spontaneous pregnancy after idiopathic POI or most forms of chemotherapy has no higher obstetric or neonatal risk than in the general population.
Pregnancy in women who received cancer treatment is increasingly common, Dr. Davies said in her talk at the meeting. Women who received ovarian irradiation face an increased risk of pregnancy complications and should be managed accordingly. Women who received treatment with an anthracycline, high-dose cyclophosphamide, or cardiac irradiation should undergo echocardiography before pregnancy, and prenatal care should include cardiology assessment.
The draft guidelines contain a total of 13 chapters addressing 31 key issues, ranging from symptoms and diagnosis, complications including bone and cardiovascular health, sexual function, and quality of life. The 17-member working group included a patient representative as well as experts from psychology, cardiology, endocrinology, internal medicine, and ob.gyn. In mid-August, the ESHRE website said that posting of the draft version should occur later in August. Once posted, ESHRE will open a 6-week comment period. The final version should appear by late 2014, Dr. Davies said.
She said she had no relevant financial disclosures.
On Twitter @mitchelzoler
MUNICH – A working group of the European Society of Human Reproduction and Embryology drafted the first international guidelines for managing premature ovarian insufficiency.
Once formally released, by late this year or soon after, the guidelines should "improve clinical practice and awareness of the condition," which affects roughly 1% of women, said Dr. Melanie Davies, a gynecologist at University College London Hospital and a working-group member who presented a few key items from the draft guidelines during a session at the society’s annual meeting.
Women with premature ovarian insufficiency (POI) sometimes are treated for one aspect of POI by a physician "without full realization of the condition and its implications," Dr. Davies said in an interview. For example, "there have been fatal cases of women with Turner syndrome who received donor oocytes," the disorder posing the greatest risk to women receiving donor eggs. "This is a good example of where the guidelines may increase awareness," she said.
One draft excerpt Dr. Davies presented advises physicians and patients that candidates for donor oocytes suspected of having POI should undergo prior to receiving eggs a full investigation for thyroid and adrenal function and karyotype. "Pregnancy in some women can be of such high risk that clinicians may consider egg donation life threatening and inappropriate," she said.
For women with Turner syndrome, the guidelines call for assessment by a cardiologist with a special interest in adult congenital heart disease, as well as a general medical and endocrine examination prior to pregnancy. During pregnancy, these women have an elevated risk for obstetric complications and should be managed in an obstetric unit designed for high-risk pregnancies with involvement of a cardiologist.
The draft defines POI as depletion of follicular activity before age 40 years with menstrual disturbance, raised gonadotropin levels, and low estradiol levels. These women should understand that no interventions are proven to reliably increase ovarian activity and their natural conception rate. But physicians also can reassure these women that spontaneous pregnancy after idiopathic POI or most forms of chemotherapy has no higher obstetric or neonatal risk than in the general population.
Pregnancy in women who received cancer treatment is increasingly common, Dr. Davies said in her talk at the meeting. Women who received ovarian irradiation face an increased risk of pregnancy complications and should be managed accordingly. Women who received treatment with an anthracycline, high-dose cyclophosphamide, or cardiac irradiation should undergo echocardiography before pregnancy, and prenatal care should include cardiology assessment.
The draft guidelines contain a total of 13 chapters addressing 31 key issues, ranging from symptoms and diagnosis, complications including bone and cardiovascular health, sexual function, and quality of life. The 17-member working group included a patient representative as well as experts from psychology, cardiology, endocrinology, internal medicine, and ob.gyn. In mid-August, the ESHRE website said that posting of the draft version should occur later in August. Once posted, ESHRE will open a 6-week comment period. The final version should appear by late 2014, Dr. Davies said.
She said she had no relevant financial disclosures.
On Twitter @mitchelzoler
MUNICH – A working group of the European Society of Human Reproduction and Embryology drafted the first international guidelines for managing premature ovarian insufficiency.
Once formally released, by late this year or soon after, the guidelines should "improve clinical practice and awareness of the condition," which affects roughly 1% of women, said Dr. Melanie Davies, a gynecologist at University College London Hospital and a working-group member who presented a few key items from the draft guidelines during a session at the society’s annual meeting.
Women with premature ovarian insufficiency (POI) sometimes are treated for one aspect of POI by a physician "without full realization of the condition and its implications," Dr. Davies said in an interview. For example, "there have been fatal cases of women with Turner syndrome who received donor oocytes," the disorder posing the greatest risk to women receiving donor eggs. "This is a good example of where the guidelines may increase awareness," she said.
One draft excerpt Dr. Davies presented advises physicians and patients that candidates for donor oocytes suspected of having POI should undergo prior to receiving eggs a full investigation for thyroid and adrenal function and karyotype. "Pregnancy in some women can be of such high risk that clinicians may consider egg donation life threatening and inappropriate," she said.
For women with Turner syndrome, the guidelines call for assessment by a cardiologist with a special interest in adult congenital heart disease, as well as a general medical and endocrine examination prior to pregnancy. During pregnancy, these women have an elevated risk for obstetric complications and should be managed in an obstetric unit designed for high-risk pregnancies with involvement of a cardiologist.
The draft defines POI as depletion of follicular activity before age 40 years with menstrual disturbance, raised gonadotropin levels, and low estradiol levels. These women should understand that no interventions are proven to reliably increase ovarian activity and their natural conception rate. But physicians also can reassure these women that spontaneous pregnancy after idiopathic POI or most forms of chemotherapy has no higher obstetric or neonatal risk than in the general population.
Pregnancy in women who received cancer treatment is increasingly common, Dr. Davies said in her talk at the meeting. Women who received ovarian irradiation face an increased risk of pregnancy complications and should be managed accordingly. Women who received treatment with an anthracycline, high-dose cyclophosphamide, or cardiac irradiation should undergo echocardiography before pregnancy, and prenatal care should include cardiology assessment.
The draft guidelines contain a total of 13 chapters addressing 31 key issues, ranging from symptoms and diagnosis, complications including bone and cardiovascular health, sexual function, and quality of life. The 17-member working group included a patient representative as well as experts from psychology, cardiology, endocrinology, internal medicine, and ob.gyn. In mid-August, the ESHRE website said that posting of the draft version should occur later in August. Once posted, ESHRE will open a 6-week comment period. The final version should appear by late 2014, Dr. Davies said.
She said she had no relevant financial disclosures.
On Twitter @mitchelzoler
EXPERT OPINION FROM ESHRE 2014