User login
Mitchel is a reporter for MDedge based in the Philadelphia area. He started with the company in 1992, when it was International Medical News Group (IMNG), and has since covered a range of medical specialties. Mitchel trained as a virologist at Roswell Park Memorial Institute in Buffalo, and then worked briefly as a researcher at Boston Children's Hospital before pivoting to journalism as a AAAS Mass Media Fellow in 1980. His first reporting job was with Science Digest magazine, and from the mid-1980s to early-1990s he was a reporter with Medical World News. @mitchelzoler
New drug boosts survival in metastatic CRC
MADRID – Treatment with a new nucleoside-analogue drug produced clinically meaningful improvements in survival and performance status in a phase III trial that enrolled 800 patients with metastatic colorectal cancer who had failed at least two prior chemotherapy regimens.
In addition to a statistically significant, 32% cut in mortality during a median follow-up of 8 months, treatment with TAS-102 was “well tolerated,” and extended the time to deterioration of performance status by more than 40% compared with patients randomized to placebo, Dr. Erik Van Cutsem said at the European Society for Medical Oncology Congress.
S-102’s ability to stabilize patient performance status, along with its benefit for the study’s primary endpoint of overall survival, is “an important clinical parameter, a real benefit” that shows the drug’s positive effect on quality of life, said Dr. Cutsem, professor and head of clinical digestive oncology at the University Hospital, Leuven, Belgium.
TAS-102 contains trifluridine, a nucleoside analogue that gets incorporated into tumor cell DNA, causing dysfunction and inhibited tumor growth. A second agent in the TAS-120 formulation, tipiracil hydrochloride, inhibits trifluridine breakdown.
The RECOURSE (Study of TAS-102 in Patients With Metastatic Colorectal Cancer Refractory to Standard Chemotherapies) trial randomized 800 patients with metastatic colorectal cancer who had failed at least two prior regimens at 114 sites in 13 countries from June 2012 to October 2013. More than half the patients had failed or were intolerant of at least four prior regimens, and more than three-quarters of enrolled patients had been diagnosed with metastatic disease 18 months prior to study entry. Patients’ median age was 63 years, and at entry they all had an Eastern Cooperative Oncology Group performance status rating of 0 or 1.
Patients received 35 mg/m2 oral TAS-102 b.i.d. on days 1-5 and 8-12 for one cycle, repeated every 4 weeks, or placebo in addition to best supportive care. Treatment with the active agent produced a hazard ratio of 0.68 (confidence interval, 0.58-0.81; P < .0001), Dr. Van Cutsem reported. After 1 year, 27% of the 534 patients on TAS-102 treatment remained alive, compared with 18% of the 266 patients randomized to placebo. Prespecified subgroup analyses showed a consistent benefit across all subgroups.
A multivariate analysis showed a similar impact of treatment on overall survival. Secondary analysis showed that TAS-102 treatment was linked to a significant improvement in progression-free survival (HR, 0.48; CI, 0.41-0.57; P < .0001). The median time to deterioration to a performance status rating of 2 was 5.7 months among patients who received TAS-102 and 4.0 months among placebo patients.
TAS-102 produced a low level of nonhematologic grade 3 or 4 adverse events. The most common, usually grade 1 or 2, were nausea, fatigue, diarrhea, and vomiting. It did not produce any excess cardiac or thromboembolic events, and had no effect on liver function or creatinine levels. The novel agent produced a 38% incidence of grade 3 or 4 neutropenia, but grade 3 or 4 febrile neutropenia was limited to 4% of patients, Dr. Van Cutsem noted. Grade 3 anemia occurred in 18% of patients on the combination agent, and 5% had grade 3 or 4 thrombocytopenia.
RECOURSE was sponsored by Taiho, which is developing TAS-102. Dr. Van Cutsem has received research funding from Amgen, Bayer, Boehringer, Lilly, Merck Serono, Novartis, Roche, and Sanofi.
On Twitter @mitchelzoler
The results of RECOURSE were robust and convincing and clearly in favor of TAS-102. This will be a very important drug in the future.
The efficacy of TAS-102 looks similar to what was recently reported for regorafenib in similar patients with refractory, metastatic colorectal cancer (Lancet 2013;381:303-12), which now makes these two agents competitors in the same situation. But TAS-102 and regorafenib have substantially different safety profiles. TAS-102 treatment produced many episodes of grade 3 or 4 neutropenia, but a much smaller number of patients developed febrile neutropenia. This toxicity is manageable. TAS-102 does not cause the hand-foot syndrome that regorafenib often triggers.

|
| Mitchel L. Zoler/Frontline Medical News Dr. Christophe Tournigand |
The efficacy of TAS-102 in the very advanced cases enrolled in the trial raises questions about how it will perform when used on less advanced patients, and whether it has a role as first- or second-line treatment. It will also be important to determine how well TAS-102 can be used in combination with other drugs; I suspect it may be easier to combine with other agents than regorafenib. TAS-102 has a mechanism of action very similar to that of fluorouracil, and it is intriguing to imagine that perhaps someday TAS-102 will replace fluorouracil as a mainstay of colorectal cancer treatment.
Dr. Christophe Tournigand is professor and head of medical oncology at Henri Mondor University Hospital in Créteil, France. He has been an adviser to Roche, Sanofi, Merck, Amgen, and Bayer. He made these comments as designated discussant for the RECOURSE trial and in an interview.
The results of RECOURSE were robust and convincing and clearly in favor of TAS-102. This will be a very important drug in the future.
The efficacy of TAS-102 looks similar to what was recently reported for regorafenib in similar patients with refractory, metastatic colorectal cancer (Lancet 2013;381:303-12), which now makes these two agents competitors in the same situation. But TAS-102 and regorafenib have substantially different safety profiles. TAS-102 treatment produced many episodes of grade 3 or 4 neutropenia, but a much smaller number of patients developed febrile neutropenia. This toxicity is manageable. TAS-102 does not cause the hand-foot syndrome that regorafenib often triggers.

|
| Mitchel L. Zoler/Frontline Medical News Dr. Christophe Tournigand |
The efficacy of TAS-102 in the very advanced cases enrolled in the trial raises questions about how it will perform when used on less advanced patients, and whether it has a role as first- or second-line treatment. It will also be important to determine how well TAS-102 can be used in combination with other drugs; I suspect it may be easier to combine with other agents than regorafenib. TAS-102 has a mechanism of action very similar to that of fluorouracil, and it is intriguing to imagine that perhaps someday TAS-102 will replace fluorouracil as a mainstay of colorectal cancer treatment.
Dr. Christophe Tournigand is professor and head of medical oncology at Henri Mondor University Hospital in Créteil, France. He has been an adviser to Roche, Sanofi, Merck, Amgen, and Bayer. He made these comments as designated discussant for the RECOURSE trial and in an interview.
The results of RECOURSE were robust and convincing and clearly in favor of TAS-102. This will be a very important drug in the future.
The efficacy of TAS-102 looks similar to what was recently reported for regorafenib in similar patients with refractory, metastatic colorectal cancer (Lancet 2013;381:303-12), which now makes these two agents competitors in the same situation. But TAS-102 and regorafenib have substantially different safety profiles. TAS-102 treatment produced many episodes of grade 3 or 4 neutropenia, but a much smaller number of patients developed febrile neutropenia. This toxicity is manageable. TAS-102 does not cause the hand-foot syndrome that regorafenib often triggers.

|
| Mitchel L. Zoler/Frontline Medical News Dr. Christophe Tournigand |
The efficacy of TAS-102 in the very advanced cases enrolled in the trial raises questions about how it will perform when used on less advanced patients, and whether it has a role as first- or second-line treatment. It will also be important to determine how well TAS-102 can be used in combination with other drugs; I suspect it may be easier to combine with other agents than regorafenib. TAS-102 has a mechanism of action very similar to that of fluorouracil, and it is intriguing to imagine that perhaps someday TAS-102 will replace fluorouracil as a mainstay of colorectal cancer treatment.
Dr. Christophe Tournigand is professor and head of medical oncology at Henri Mondor University Hospital in Créteil, France. He has been an adviser to Roche, Sanofi, Merck, Amgen, and Bayer. He made these comments as designated discussant for the RECOURSE trial and in an interview.
MADRID – Treatment with a new nucleoside-analogue drug produced clinically meaningful improvements in survival and performance status in a phase III trial that enrolled 800 patients with metastatic colorectal cancer who had failed at least two prior chemotherapy regimens.
In addition to a statistically significant, 32% cut in mortality during a median follow-up of 8 months, treatment with TAS-102 was “well tolerated,” and extended the time to deterioration of performance status by more than 40% compared with patients randomized to placebo, Dr. Erik Van Cutsem said at the European Society for Medical Oncology Congress.
S-102’s ability to stabilize patient performance status, along with its benefit for the study’s primary endpoint of overall survival, is “an important clinical parameter, a real benefit” that shows the drug’s positive effect on quality of life, said Dr. Cutsem, professor and head of clinical digestive oncology at the University Hospital, Leuven, Belgium.
TAS-102 contains trifluridine, a nucleoside analogue that gets incorporated into tumor cell DNA, causing dysfunction and inhibited tumor growth. A second agent in the TAS-120 formulation, tipiracil hydrochloride, inhibits trifluridine breakdown.
The RECOURSE (Study of TAS-102 in Patients With Metastatic Colorectal Cancer Refractory to Standard Chemotherapies) trial randomized 800 patients with metastatic colorectal cancer who had failed at least two prior regimens at 114 sites in 13 countries from June 2012 to October 2013. More than half the patients had failed or were intolerant of at least four prior regimens, and more than three-quarters of enrolled patients had been diagnosed with metastatic disease 18 months prior to study entry. Patients’ median age was 63 years, and at entry they all had an Eastern Cooperative Oncology Group performance status rating of 0 or 1.
Patients received 35 mg/m2 oral TAS-102 b.i.d. on days 1-5 and 8-12 for one cycle, repeated every 4 weeks, or placebo in addition to best supportive care. Treatment with the active agent produced a hazard ratio of 0.68 (confidence interval, 0.58-0.81; P < .0001), Dr. Van Cutsem reported. After 1 year, 27% of the 534 patients on TAS-102 treatment remained alive, compared with 18% of the 266 patients randomized to placebo. Prespecified subgroup analyses showed a consistent benefit across all subgroups.
A multivariate analysis showed a similar impact of treatment on overall survival. Secondary analysis showed that TAS-102 treatment was linked to a significant improvement in progression-free survival (HR, 0.48; CI, 0.41-0.57; P < .0001). The median time to deterioration to a performance status rating of 2 was 5.7 months among patients who received TAS-102 and 4.0 months among placebo patients.
TAS-102 produced a low level of nonhematologic grade 3 or 4 adverse events. The most common, usually grade 1 or 2, were nausea, fatigue, diarrhea, and vomiting. It did not produce any excess cardiac or thromboembolic events, and had no effect on liver function or creatinine levels. The novel agent produced a 38% incidence of grade 3 or 4 neutropenia, but grade 3 or 4 febrile neutropenia was limited to 4% of patients, Dr. Van Cutsem noted. Grade 3 anemia occurred in 18% of patients on the combination agent, and 5% had grade 3 or 4 thrombocytopenia.
RECOURSE was sponsored by Taiho, which is developing TAS-102. Dr. Van Cutsem has received research funding from Amgen, Bayer, Boehringer, Lilly, Merck Serono, Novartis, Roche, and Sanofi.
On Twitter @mitchelzoler
MADRID – Treatment with a new nucleoside-analogue drug produced clinically meaningful improvements in survival and performance status in a phase III trial that enrolled 800 patients with metastatic colorectal cancer who had failed at least two prior chemotherapy regimens.
In addition to a statistically significant, 32% cut in mortality during a median follow-up of 8 months, treatment with TAS-102 was “well tolerated,” and extended the time to deterioration of performance status by more than 40% compared with patients randomized to placebo, Dr. Erik Van Cutsem said at the European Society for Medical Oncology Congress.
S-102’s ability to stabilize patient performance status, along with its benefit for the study’s primary endpoint of overall survival, is “an important clinical parameter, a real benefit” that shows the drug’s positive effect on quality of life, said Dr. Cutsem, professor and head of clinical digestive oncology at the University Hospital, Leuven, Belgium.
TAS-102 contains trifluridine, a nucleoside analogue that gets incorporated into tumor cell DNA, causing dysfunction and inhibited tumor growth. A second agent in the TAS-120 formulation, tipiracil hydrochloride, inhibits trifluridine breakdown.
The RECOURSE (Study of TAS-102 in Patients With Metastatic Colorectal Cancer Refractory to Standard Chemotherapies) trial randomized 800 patients with metastatic colorectal cancer who had failed at least two prior regimens at 114 sites in 13 countries from June 2012 to October 2013. More than half the patients had failed or were intolerant of at least four prior regimens, and more than three-quarters of enrolled patients had been diagnosed with metastatic disease 18 months prior to study entry. Patients’ median age was 63 years, and at entry they all had an Eastern Cooperative Oncology Group performance status rating of 0 or 1.
Patients received 35 mg/m2 oral TAS-102 b.i.d. on days 1-5 and 8-12 for one cycle, repeated every 4 weeks, or placebo in addition to best supportive care. Treatment with the active agent produced a hazard ratio of 0.68 (confidence interval, 0.58-0.81; P < .0001), Dr. Van Cutsem reported. After 1 year, 27% of the 534 patients on TAS-102 treatment remained alive, compared with 18% of the 266 patients randomized to placebo. Prespecified subgroup analyses showed a consistent benefit across all subgroups.
A multivariate analysis showed a similar impact of treatment on overall survival. Secondary analysis showed that TAS-102 treatment was linked to a significant improvement in progression-free survival (HR, 0.48; CI, 0.41-0.57; P < .0001). The median time to deterioration to a performance status rating of 2 was 5.7 months among patients who received TAS-102 and 4.0 months among placebo patients.
TAS-102 produced a low level of nonhematologic grade 3 or 4 adverse events. The most common, usually grade 1 or 2, were nausea, fatigue, diarrhea, and vomiting. It did not produce any excess cardiac or thromboembolic events, and had no effect on liver function or creatinine levels. The novel agent produced a 38% incidence of grade 3 or 4 neutropenia, but grade 3 or 4 febrile neutropenia was limited to 4% of patients, Dr. Van Cutsem noted. Grade 3 anemia occurred in 18% of patients on the combination agent, and 5% had grade 3 or 4 thrombocytopenia.
RECOURSE was sponsored by Taiho, which is developing TAS-102. Dr. Van Cutsem has received research funding from Amgen, Bayer, Boehringer, Lilly, Merck Serono, Novartis, Roche, and Sanofi.
On Twitter @mitchelzoler
AT ESMO 2014
Key clinical point: TAS-102 treatment significantly boosted overall survival with good tolerability in patients with metastatic colorectal cancer refractory to at least two prior treatments in a phase III study.
Major finding: TAS-102 treatment improved overall survival by a hazard ratio of 0.68 compared with placebo.
Data source: RECOURSE, a randomized study with 800 patients at 114 centers in 13 countries.
Disclosures: RECOURSE was sponsored by Taiho, which is developing TAS-102. Dr. Van Cutsem has received research funding from Amgen, Bayer, Boehringer, Lilly, Merck Serono, Novartis, Roche, and Sanofi. Dr. Tournigand has been an adviser to Roche, Sanofi, Merck, Amgen, and Bayer.
Intensive glycemic control safely cut end-stage renal disease
VIENNA – Five years of intensive glycemic control in patients with type 2 diabetes safely halved long-term rate of end-stage renal disease, compared with placebo, in a multicenter study with about 8,000 patients, a finding that refutes prior suggestions that more intensive glycemic control can harm patients.
Intensive glucose control that produced an average hemoglobin A1c of 6.5% “is important for preventing serious renal complications and does not cause harm in patients with established type 2 diabetes,” Dr. Sophia Zoungas said at the annual meeting of the European Association for the Study of Diabetes.
Following the report of results from the ACCORD (Action to Control Cardiovascular Risk in Diabetes) trial in 2008 (N. Engl. J. Med. 2008;358;2545-59), which showed increased mortality with intensive glycemic control, compared with standard treatment, many physicians became leery of treating patients to a HbA1c level below 7%. But 5-year follow-up results of patients originally enrolled in the ADVANCE (Action in Diabetes and Vascular Disease: Preterax and Diamicron MR Controlled Evaluation) trial (N. Engl. J. Med. 2008;358:2560-72) has now shown that a prolonged period maintaining patients at a HbA1c of roughly 6.5% produced no excess rates of total mortality, major macrovascular events, or major clinical microvascular events when compared with patients on standard care with an average HbA1c of 7.2%, said Dr. Zoungas, an endocrinologist with the George Institute of the University of Sydney.
“Whether the excess mortality seen in ACCORD was a true effect or not, there has been concern [about tight glycemic control] that has translated into where HbA1c targets have been set,” Dr. Zoungas said in an interview. “We have been blinded by this signal of increased fatal myocardial infarctions [seen in ACCORD] that many have been a chance effect.”
She suggested that the safety that intense glycemic control showed during 10-year follow-up of patients in the ADVANCE posttrial Observational Study (ADVANCE ON) may be explained by the more gradual HbA1c reductions achieved in the ADVANCE patients, compared with patients in ACCORD. In ADVANCE, patients’ HbA1c levels came down to about 6.5% over the course of a year, compared with a 3-6 month period in ACCORD, Dr. Zoungas noted.
In addition to establishing safety, the new results reported by Dr. Zoungas showed a statistically significant, 46% reduction in the incidence of end-stage renal disease (defined as progression to dialysis or need for kidney transplant) during the entire, 10-year follow-up. Simultaneous with Dr. Zoungas’s report at the meeting the ADVANCE ON results were published online (N. Engl. J. Med. 2014 [doi:10.1056/NEJMoa1407963]).
“This is the first time study results have shown that maintaining a HbA1cof less than 7% is associated with a long-term reduction in end-stage kidney disease,” she said. “End-stage kidney disease has been hard to study [as an endpoint] because the overall incidence is low, so patients need to be followed for a long time to see enough events.”
ADVANCE ON initially included 8,494 of the roughly 10,000 patients who completed the 5-year ADVANCE intervention trial at 215 centers in 20 countries. ADVANCE tested the impact of an antihypertensive intervention as well as the effect of intensified glycemic control in patients aged 55 years or older with type 2 diabetes and at least one other cardiovascular risk factor. At the time ADVANCE ended, average HbA1c levels were 7.2% in the control patients and 6.5% in those who had been on a more intensive hypoglycemic regimen. However, at the time patients began ADVANCE ON, their average HbA1c was 7.5%, regardless of which arm of ADVANCE they had been in, and the average level remained there through a median of 5 more years of follow-up. A total of 5,131 patients remained under study for the entire 10 years of follow-up. Analysis of the ADVANCE ON findings also showed a significant long-term effect from 5 years of added antihypertensive treatment, results reported in early September at the annual congress of the European Society of Cardiology.
ADVANCE ON received partial funding from Servier. Dr. Zoungas has received honoraria from Servier as well as from Merck, Bristol-Myers Squibb/AstraZeneca, Sanofi-Aventis, Novo Nordisk, and Amgen.
On Twitter @mitchelzoler
The ADVANCE ON results change our perspective on the medical treatment of patients with type 2 diabetes.
The results provide more evidence for the safety of metformin and sulfonylureas, two of the major drugs used on patients in the study during the intensive-management phase. This is not level I evidence, but it is evidence of safety. The results from ADVANCE ON as well as from other trials also suggest that these drugs reduce microvascular and macrovascular complications. The long-term legacy effect of intensive glycemic control to reduce the incidence of end-stage kidney disease was impressive. The hemoglobin A1c target established by existing management guidelines can remain in place.

|
| Mitchel L. Zoler/Frontline Medical News Dr. Joachim Spranger |
The results also highlight our desperate need for glucose-lowering drugs that can substantially reduce fatal cardiovascular disease.
New drugs being considered for treatment of type 2 diabetes need to outperform metformin, sulfonylureas, and insulin for reducing clinically relevant endpoints. It is not enough for trials to show that new drugs can reduce glucose while causing fewer adverse effects than the older agents. Proven impact on clinical endpoints is also needed. We also must remember that the safety of treatment with three oral agents in a single regimen remains unproven.
Dr. Joachim Spranger is a professor of endocrinology at Charité Medical University in Berlin. He had no disclosures. He made these comments as the invited discussant for ADVANCE ON.
The ADVANCE ON results change our perspective on the medical treatment of patients with type 2 diabetes.
The results provide more evidence for the safety of metformin and sulfonylureas, two of the major drugs used on patients in the study during the intensive-management phase. This is not level I evidence, but it is evidence of safety. The results from ADVANCE ON as well as from other trials also suggest that these drugs reduce microvascular and macrovascular complications. The long-term legacy effect of intensive glycemic control to reduce the incidence of end-stage kidney disease was impressive. The hemoglobin A1c target established by existing management guidelines can remain in place.

|
| Mitchel L. Zoler/Frontline Medical News Dr. Joachim Spranger |
The results also highlight our desperate need for glucose-lowering drugs that can substantially reduce fatal cardiovascular disease.
New drugs being considered for treatment of type 2 diabetes need to outperform metformin, sulfonylureas, and insulin for reducing clinically relevant endpoints. It is not enough for trials to show that new drugs can reduce glucose while causing fewer adverse effects than the older agents. Proven impact on clinical endpoints is also needed. We also must remember that the safety of treatment with three oral agents in a single regimen remains unproven.
Dr. Joachim Spranger is a professor of endocrinology at Charité Medical University in Berlin. He had no disclosures. He made these comments as the invited discussant for ADVANCE ON.
The ADVANCE ON results change our perspective on the medical treatment of patients with type 2 diabetes.
The results provide more evidence for the safety of metformin and sulfonylureas, two of the major drugs used on patients in the study during the intensive-management phase. This is not level I evidence, but it is evidence of safety. The results from ADVANCE ON as well as from other trials also suggest that these drugs reduce microvascular and macrovascular complications. The long-term legacy effect of intensive glycemic control to reduce the incidence of end-stage kidney disease was impressive. The hemoglobin A1c target established by existing management guidelines can remain in place.

|
| Mitchel L. Zoler/Frontline Medical News Dr. Joachim Spranger |
The results also highlight our desperate need for glucose-lowering drugs that can substantially reduce fatal cardiovascular disease.
New drugs being considered for treatment of type 2 diabetes need to outperform metformin, sulfonylureas, and insulin for reducing clinically relevant endpoints. It is not enough for trials to show that new drugs can reduce glucose while causing fewer adverse effects than the older agents. Proven impact on clinical endpoints is also needed. We also must remember that the safety of treatment with three oral agents in a single regimen remains unproven.
Dr. Joachim Spranger is a professor of endocrinology at Charité Medical University in Berlin. He had no disclosures. He made these comments as the invited discussant for ADVANCE ON.
VIENNA – Five years of intensive glycemic control in patients with type 2 diabetes safely halved long-term rate of end-stage renal disease, compared with placebo, in a multicenter study with about 8,000 patients, a finding that refutes prior suggestions that more intensive glycemic control can harm patients.
Intensive glucose control that produced an average hemoglobin A1c of 6.5% “is important for preventing serious renal complications and does not cause harm in patients with established type 2 diabetes,” Dr. Sophia Zoungas said at the annual meeting of the European Association for the Study of Diabetes.
Following the report of results from the ACCORD (Action to Control Cardiovascular Risk in Diabetes) trial in 2008 (N. Engl. J. Med. 2008;358;2545-59), which showed increased mortality with intensive glycemic control, compared with standard treatment, many physicians became leery of treating patients to a HbA1c level below 7%. But 5-year follow-up results of patients originally enrolled in the ADVANCE (Action in Diabetes and Vascular Disease: Preterax and Diamicron MR Controlled Evaluation) trial (N. Engl. J. Med. 2008;358:2560-72) has now shown that a prolonged period maintaining patients at a HbA1c of roughly 6.5% produced no excess rates of total mortality, major macrovascular events, or major clinical microvascular events when compared with patients on standard care with an average HbA1c of 7.2%, said Dr. Zoungas, an endocrinologist with the George Institute of the University of Sydney.
“Whether the excess mortality seen in ACCORD was a true effect or not, there has been concern [about tight glycemic control] that has translated into where HbA1c targets have been set,” Dr. Zoungas said in an interview. “We have been blinded by this signal of increased fatal myocardial infarctions [seen in ACCORD] that many have been a chance effect.”
She suggested that the safety that intense glycemic control showed during 10-year follow-up of patients in the ADVANCE posttrial Observational Study (ADVANCE ON) may be explained by the more gradual HbA1c reductions achieved in the ADVANCE patients, compared with patients in ACCORD. In ADVANCE, patients’ HbA1c levels came down to about 6.5% over the course of a year, compared with a 3-6 month period in ACCORD, Dr. Zoungas noted.
In addition to establishing safety, the new results reported by Dr. Zoungas showed a statistically significant, 46% reduction in the incidence of end-stage renal disease (defined as progression to dialysis or need for kidney transplant) during the entire, 10-year follow-up. Simultaneous with Dr. Zoungas’s report at the meeting the ADVANCE ON results were published online (N. Engl. J. Med. 2014 [doi:10.1056/NEJMoa1407963]).
“This is the first time study results have shown that maintaining a HbA1cof less than 7% is associated with a long-term reduction in end-stage kidney disease,” she said. “End-stage kidney disease has been hard to study [as an endpoint] because the overall incidence is low, so patients need to be followed for a long time to see enough events.”
ADVANCE ON initially included 8,494 of the roughly 10,000 patients who completed the 5-year ADVANCE intervention trial at 215 centers in 20 countries. ADVANCE tested the impact of an antihypertensive intervention as well as the effect of intensified glycemic control in patients aged 55 years or older with type 2 diabetes and at least one other cardiovascular risk factor. At the time ADVANCE ended, average HbA1c levels were 7.2% in the control patients and 6.5% in those who had been on a more intensive hypoglycemic regimen. However, at the time patients began ADVANCE ON, their average HbA1c was 7.5%, regardless of which arm of ADVANCE they had been in, and the average level remained there through a median of 5 more years of follow-up. A total of 5,131 patients remained under study for the entire 10 years of follow-up. Analysis of the ADVANCE ON findings also showed a significant long-term effect from 5 years of added antihypertensive treatment, results reported in early September at the annual congress of the European Society of Cardiology.
ADVANCE ON received partial funding from Servier. Dr. Zoungas has received honoraria from Servier as well as from Merck, Bristol-Myers Squibb/AstraZeneca, Sanofi-Aventis, Novo Nordisk, and Amgen.
On Twitter @mitchelzoler
VIENNA – Five years of intensive glycemic control in patients with type 2 diabetes safely halved long-term rate of end-stage renal disease, compared with placebo, in a multicenter study with about 8,000 patients, a finding that refutes prior suggestions that more intensive glycemic control can harm patients.
Intensive glucose control that produced an average hemoglobin A1c of 6.5% “is important for preventing serious renal complications and does not cause harm in patients with established type 2 diabetes,” Dr. Sophia Zoungas said at the annual meeting of the European Association for the Study of Diabetes.
Following the report of results from the ACCORD (Action to Control Cardiovascular Risk in Diabetes) trial in 2008 (N. Engl. J. Med. 2008;358;2545-59), which showed increased mortality with intensive glycemic control, compared with standard treatment, many physicians became leery of treating patients to a HbA1c level below 7%. But 5-year follow-up results of patients originally enrolled in the ADVANCE (Action in Diabetes and Vascular Disease: Preterax and Diamicron MR Controlled Evaluation) trial (N. Engl. J. Med. 2008;358:2560-72) has now shown that a prolonged period maintaining patients at a HbA1c of roughly 6.5% produced no excess rates of total mortality, major macrovascular events, or major clinical microvascular events when compared with patients on standard care with an average HbA1c of 7.2%, said Dr. Zoungas, an endocrinologist with the George Institute of the University of Sydney.
“Whether the excess mortality seen in ACCORD was a true effect or not, there has been concern [about tight glycemic control] that has translated into where HbA1c targets have been set,” Dr. Zoungas said in an interview. “We have been blinded by this signal of increased fatal myocardial infarctions [seen in ACCORD] that many have been a chance effect.”
She suggested that the safety that intense glycemic control showed during 10-year follow-up of patients in the ADVANCE posttrial Observational Study (ADVANCE ON) may be explained by the more gradual HbA1c reductions achieved in the ADVANCE patients, compared with patients in ACCORD. In ADVANCE, patients’ HbA1c levels came down to about 6.5% over the course of a year, compared with a 3-6 month period in ACCORD, Dr. Zoungas noted.
In addition to establishing safety, the new results reported by Dr. Zoungas showed a statistically significant, 46% reduction in the incidence of end-stage renal disease (defined as progression to dialysis or need for kidney transplant) during the entire, 10-year follow-up. Simultaneous with Dr. Zoungas’s report at the meeting the ADVANCE ON results were published online (N. Engl. J. Med. 2014 [doi:10.1056/NEJMoa1407963]).
“This is the first time study results have shown that maintaining a HbA1cof less than 7% is associated with a long-term reduction in end-stage kidney disease,” she said. “End-stage kidney disease has been hard to study [as an endpoint] because the overall incidence is low, so patients need to be followed for a long time to see enough events.”
ADVANCE ON initially included 8,494 of the roughly 10,000 patients who completed the 5-year ADVANCE intervention trial at 215 centers in 20 countries. ADVANCE tested the impact of an antihypertensive intervention as well as the effect of intensified glycemic control in patients aged 55 years or older with type 2 diabetes and at least one other cardiovascular risk factor. At the time ADVANCE ended, average HbA1c levels were 7.2% in the control patients and 6.5% in those who had been on a more intensive hypoglycemic regimen. However, at the time patients began ADVANCE ON, their average HbA1c was 7.5%, regardless of which arm of ADVANCE they had been in, and the average level remained there through a median of 5 more years of follow-up. A total of 5,131 patients remained under study for the entire 10 years of follow-up. Analysis of the ADVANCE ON findings also showed a significant long-term effect from 5 years of added antihypertensive treatment, results reported in early September at the annual congress of the European Society of Cardiology.
ADVANCE ON received partial funding from Servier. Dr. Zoungas has received honoraria from Servier as well as from Merck, Bristol-Myers Squibb/AstraZeneca, Sanofi-Aventis, Novo Nordisk, and Amgen.
On Twitter @mitchelzoler
AT EASD 2014
Key clinical point: Intensive glycemic control in patients with type 2 diabetes safely led to a long-term reduction in end-stage kidney disease, compared with standard management.
Major finding: Intensive glycemic-control patients had a 46% reduced rate of end-stage kidney disease, compared with standard-care patients.
Data source: The ADVANCE ON study, which followed 8,494 patients after their participation in an international glycemia-control trial.
Disclosures: ADVANCE ON received partial funding from Servier. Dr. Zoungas has received honoraria from Servier as well as from Merck, Bristol-Myers Squibb/AstraZeneca, Sanofi-Aventis, Novo Nordisk, and Amgen.
Saxagliptin reverses proteinuria in type 2 diabetes
VIENNA – Patients with type 2 diabetes treated with the oral hypoglycemic saxagliptin (Onglyza) benefited from stabilization and, in some cases, reversal of urinary protein levels in a prespecified, secondary analysis of data collected from more than 16,000 patients in a randomized, international trial.
Progression of patients with diabetes from normoalbuminuria to microalbuminuria and then proteinuria tracks along with their deteriorating renal function. Until now, no agent has been identified that could stop or reverse this process, although a similar effect had been documented in results from a few prior, much smaller studies using other drugs from the class of dipeptidyl peptidase-4 (DPP-4) inhibitors, which includes saxagliptin.
“It’s a very important finding that needs further investigation in longer-term studies so we can see whether the improvement in the [albumin to creatinine ratio (ACR)] will also mean less deterioration of the eGFR [estimated glomerular filtration rate] and thereby prevent end-stage renal disease,” Dr. Ofri Mosenzon said following her report at the annual meeting of the European Association for the Study of Diabetes.
The new analysis also showed that saxagliptin did this safely, without causing any renal damage, and that the effect on microalbuminuria and proteinuria was completely independent of the drug’s glycemic control effect measured by hemoglobin A1c levels. Dr. Mosenzon had no simple explanation for the mechanism by which saxagliptin and other DPP-4 inhibitors might exert this effect, though she suggested that an anti-inflammatory pleiotropic effect of the drug class might be involved.
“We have this clinical result. Now we need to look for an explanation,” said Dr. Mosenzon, a diabetologist at Hadassah Hospital in Jerusalem.
Her analysis used data collected in the the Saxagliptin Assessment of Vascular Outcomes Recorded in Patients with Diabetes Mellitus (SAVOR) – Thrombolysis in Myocardial Infarction (TIMI) 53 trial, which had the primary purpose of assessing the cardiovascular safety of saxagliptin in 16,492 patients with type 2 diabetes and a high risk for cardiovascular events (N. Engl. J. Med. 2013;369:1317-26). The study randomized patients to treatment with either 5 mg saxagliptin daily (2.5 mg daily in patients with impaired renal function at entry) or placebo in addition to whatever other standard medications their physicians prescribed. The secondary analysis of the impact of saxagliptin on proteinuria and renal function used prespecified definitions for evaluating the renal safety and renal-protective efficacy of treatment.
On the safety side, treatment with saxagliptin, compared with placebo linked with no statistically significant differences in the rate or magnitude of serum creatinine increases or in the rates of progression to dialysis or renal transplant.
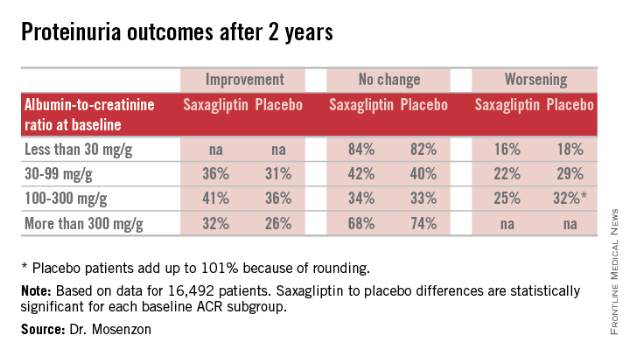
For efficacy, treatment with saxagliptin, compared with placebo led to a consistent pattern of stabilization and reversal of the severity of microalbuminuria and proteinuria that cut across patients at all baseline levels of urinary protein (see table). For example, among 1,638 categorized as having proteinuria at entry into the study, based on an ACR of more than 300 mg/g, 32% of the patients treated with saxagliptin had improved to daily urinary protein excretions of less than 300 mg/g after an average 2.1 years on treatment, compared with a 26% rate in the placebo-treated control patients, a statistically significant difference. The extent of reversals and progressions prevented were also statistically significant among the 9,696 patients who entered the study with normoalbuminuria and urinary protein levels of less than 30 mg/g and those who entered with microalbuminuria and daily urinary protein levels of 30-300 mg/g.
Expressed another way, when compared with placebo-treated patients those patients treated with saxagliptin for 2 years who entered the study with an eGFR of more than 50 mL/min/1.73 m2 had on average 19 mg less daily protein in their urine, those who entered with an eGFR of 30-50 mL/min/1.73 m2 averaged 105 mg less daily urine protein, and those who entered with an eGFR of less than 30 mL/min/1.73 m2 averaged 245 mg less daily urine protein.
SAVOR-TIMI 53 was sponsored by AstraZeneca and Bristol-Myers Squibb, the companies that market saxagliptin (Onglyza). Dr. Mosenzon has been a speaker for and advisor to those and to several other drug companies.
VIENNA – Patients with type 2 diabetes treated with the oral hypoglycemic saxagliptin (Onglyza) benefited from stabilization and, in some cases, reversal of urinary protein levels in a prespecified, secondary analysis of data collected from more than 16,000 patients in a randomized, international trial.
Progression of patients with diabetes from normoalbuminuria to microalbuminuria and then proteinuria tracks along with their deteriorating renal function. Until now, no agent has been identified that could stop or reverse this process, although a similar effect had been documented in results from a few prior, much smaller studies using other drugs from the class of dipeptidyl peptidase-4 (DPP-4) inhibitors, which includes saxagliptin.

“It’s a very important finding that needs further investigation in longer-term studies so we can see whether the improvement in the [albumin to creatinine ratio (ACR)] will also mean less deterioration of the eGFR [estimated glomerular filtration rate] and thereby prevent end-stage renal disease,” Dr. Ofri Mosenzon said following her report at the annual meeting of the European Association for the Study of Diabetes.
The new analysis also showed that saxagliptin did this safely, without causing any renal damage, and that the effect on microalbuminuria and proteinuria was completely independent of the drug’s glycemic control effect measured by hemoglobin A1c levels. Dr. Mosenzon had no simple explanation for the mechanism by which saxagliptin and other DPP-4 inhibitors might exert this effect, though she suggested that an anti-inflammatory pleiotropic effect of the drug class might be involved.
“We have this clinical result. Now we need to look for an explanation,” said Dr. Mosenzon, a diabetologist at Hadassah Hospital in Jerusalem.
Her analysis used data collected in the the Saxagliptin Assessment of Vascular Outcomes Recorded in Patients with Diabetes Mellitus (SAVOR) – Thrombolysis in Myocardial Infarction (TIMI) 53 trial, which had the primary purpose of assessing the cardiovascular safety of saxagliptin in 16,492 patients with type 2 diabetes and a high risk for cardiovascular events (N. Engl. J. Med. 2013;369:1317-26). The study randomized patients to treatment with either 5 mg saxagliptin daily (2.5 mg daily in patients with impaired renal function at entry) or placebo in addition to whatever other standard medications their physicians prescribed. The secondary analysis of the impact of saxagliptin on proteinuria and renal function used prespecified definitions for evaluating the renal safety and renal-protective efficacy of treatment.
On the safety side, treatment with saxagliptin, compared with placebo linked with no statistically significant differences in the rate or magnitude of serum creatinine increases or in the rates of progression to dialysis or renal transplant.
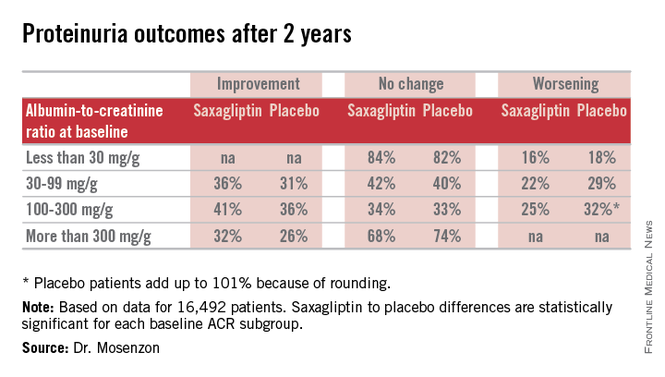
For efficacy, treatment with saxagliptin, compared with placebo led to a consistent pattern of stabilization and reversal of the severity of microalbuminuria and proteinuria that cut across patients at all baseline levels of urinary protein (see table). For example, among 1,638 categorized as having proteinuria at entry into the study, based on an ACR of more than 300 mg/g, 32% of the patients treated with saxagliptin had improved to daily urinary protein excretions of less than 300 mg/g after an average 2.1 years on treatment, compared with a 26% rate in the placebo-treated control patients, a statistically significant difference. The extent of reversals and progressions prevented were also statistically significant among the 9,696 patients who entered the study with normoalbuminuria and urinary protein levels of less than 30 mg/g and those who entered with microalbuminuria and daily urinary protein levels of 30-300 mg/g.
Expressed another way, when compared with placebo-treated patients those patients treated with saxagliptin for 2 years who entered the study with an eGFR of more than 50 mL/min/1.73 m2 had on average 19 mg less daily protein in their urine, those who entered with an eGFR of 30-50 mL/min/1.73 m2 averaged 105 mg less daily urine protein, and those who entered with an eGFR of less than 30 mL/min/1.73 m2 averaged 245 mg less daily urine protein.
SAVOR-TIMI 53 was sponsored by AstraZeneca and Bristol-Myers Squibb, the companies that market saxagliptin (Onglyza). Dr. Mosenzon has been a speaker for and advisor to those and to several other drug companies.
VIENNA – Patients with type 2 diabetes treated with the oral hypoglycemic saxagliptin (Onglyza) benefited from stabilization and, in some cases, reversal of urinary protein levels in a prespecified, secondary analysis of data collected from more than 16,000 patients in a randomized, international trial.
Progression of patients with diabetes from normoalbuminuria to microalbuminuria and then proteinuria tracks along with their deteriorating renal function. Until now, no agent has been identified that could stop or reverse this process, although a similar effect had been documented in results from a few prior, much smaller studies using other drugs from the class of dipeptidyl peptidase-4 (DPP-4) inhibitors, which includes saxagliptin.

“It’s a very important finding that needs further investigation in longer-term studies so we can see whether the improvement in the [albumin to creatinine ratio (ACR)] will also mean less deterioration of the eGFR [estimated glomerular filtration rate] and thereby prevent end-stage renal disease,” Dr. Ofri Mosenzon said following her report at the annual meeting of the European Association for the Study of Diabetes.
The new analysis also showed that saxagliptin did this safely, without causing any renal damage, and that the effect on microalbuminuria and proteinuria was completely independent of the drug’s glycemic control effect measured by hemoglobin A1c levels. Dr. Mosenzon had no simple explanation for the mechanism by which saxagliptin and other DPP-4 inhibitors might exert this effect, though she suggested that an anti-inflammatory pleiotropic effect of the drug class might be involved.
“We have this clinical result. Now we need to look for an explanation,” said Dr. Mosenzon, a diabetologist at Hadassah Hospital in Jerusalem.
Her analysis used data collected in the the Saxagliptin Assessment of Vascular Outcomes Recorded in Patients with Diabetes Mellitus (SAVOR) – Thrombolysis in Myocardial Infarction (TIMI) 53 trial, which had the primary purpose of assessing the cardiovascular safety of saxagliptin in 16,492 patients with type 2 diabetes and a high risk for cardiovascular events (N. Engl. J. Med. 2013;369:1317-26). The study randomized patients to treatment with either 5 mg saxagliptin daily (2.5 mg daily in patients with impaired renal function at entry) or placebo in addition to whatever other standard medications their physicians prescribed. The secondary analysis of the impact of saxagliptin on proteinuria and renal function used prespecified definitions for evaluating the renal safety and renal-protective efficacy of treatment.
On the safety side, treatment with saxagliptin, compared with placebo linked with no statistically significant differences in the rate or magnitude of serum creatinine increases or in the rates of progression to dialysis or renal transplant.
For efficacy, treatment with saxagliptin, compared with placebo led to a consistent pattern of stabilization and reversal of the severity of microalbuminuria and proteinuria that cut across patients at all baseline levels of urinary protein (see table). For example, among 1,638 categorized as having proteinuria at entry into the study, based on an ACR of more than 300 mg/g, 32% of the patients treated with saxagliptin had improved to daily urinary protein excretions of less than 300 mg/g after an average 2.1 years on treatment, compared with a 26% rate in the placebo-treated control patients, a statistically significant difference. The extent of reversals and progressions prevented were also statistically significant among the 9,696 patients who entered the study with normoalbuminuria and urinary protein levels of less than 30 mg/g and those who entered with microalbuminuria and daily urinary protein levels of 30-300 mg/g.
Expressed another way, when compared with placebo-treated patients those patients treated with saxagliptin for 2 years who entered the study with an eGFR of more than 50 mL/min/1.73 m2 had on average 19 mg less daily protein in their urine, those who entered with an eGFR of 30-50 mL/min/1.73 m2 averaged 105 mg less daily urine protein, and those who entered with an eGFR of less than 30 mL/min/1.73 m2 averaged 245 mg less daily urine protein.
SAVOR-TIMI 53 was sponsored by AstraZeneca and Bristol-Myers Squibb, the companies that market saxagliptin (Onglyza). Dr. Mosenzon has been a speaker for and advisor to those and to several other drug companies.
AT EASD 2014
Key clinical point: Treatment with saxagliptin led to stabilization and in some patients reversal of microalbuminuria and proteinuria in patients with type 2 diabetes.
Major finding: Proteinuria reversed in 32% of patients treated with saxagliptin during 2.1 years, compared with a 26% rate in controls.
Data source: Prespecified, secondary analysis of data from SAVOR-TIMI 53, an international, randomized trial with 16,492 patients with type 2 diabetes..
Disclosures: SAVOR-TIMI 53 was sponsored by AstraZeneca and Bristol-Myers Squibb, the companies that market saxagliptin (Onglyza). Dr. Mosenzon has been a speaker for and advisor to those and to several other drug companies.
VIDEO: Gradual HbA1c reduction safely benefits T2DM
VIENNA – A “pragmatic and simple” approach to gradually lower the hemoglobin A1c level in patients with type 2 diabetes to 6.5% and then maintain it for an average of 5 years caused no suggestion of harm and led to an important halving of end-stage renal disease during 10-year follow-up in a controlled study with more than 8,000 patients, Dr. Sophia Zoungas said in an interview at the annual meeting of the European Association for the Study of Diabetes.
Intensive glucose control did not increase mortality or the rate of major macrovascular events in 10-year results from the ADVANCE ON (Action in Diabetes and Vascular Disease: Preterax and Diamicron MR Controlled Evaluation Post Trial Observational Study) trial. The results also showed for the first time that intensive glucose control produced a significant and large reduction in end-stage kidney disease, said Dr. Zoungas, an endocrinologist with the George Institute of the University of Sydney.
ADVANCE ON received partial funding from Servier. Dr. Zoungas has received honoraria from Servier as well as from Merck Sharp & Dohme, Bristol-Myers Squibb/AstraZeneca, Sanofi-Aventis, Novo Nordisk, and Amgen.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
VIENNA – A “pragmatic and simple” approach to gradually lower the hemoglobin A1c level in patients with type 2 diabetes to 6.5% and then maintain it for an average of 5 years caused no suggestion of harm and led to an important halving of end-stage renal disease during 10-year follow-up in a controlled study with more than 8,000 patients, Dr. Sophia Zoungas said in an interview at the annual meeting of the European Association for the Study of Diabetes.
Intensive glucose control did not increase mortality or the rate of major macrovascular events in 10-year results from the ADVANCE ON (Action in Diabetes and Vascular Disease: Preterax and Diamicron MR Controlled Evaluation Post Trial Observational Study) trial. The results also showed for the first time that intensive glucose control produced a significant and large reduction in end-stage kidney disease, said Dr. Zoungas, an endocrinologist with the George Institute of the University of Sydney.
ADVANCE ON received partial funding from Servier. Dr. Zoungas has received honoraria from Servier as well as from Merck Sharp & Dohme, Bristol-Myers Squibb/AstraZeneca, Sanofi-Aventis, Novo Nordisk, and Amgen.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
VIENNA – A “pragmatic and simple” approach to gradually lower the hemoglobin A1c level in patients with type 2 diabetes to 6.5% and then maintain it for an average of 5 years caused no suggestion of harm and led to an important halving of end-stage renal disease during 10-year follow-up in a controlled study with more than 8,000 patients, Dr. Sophia Zoungas said in an interview at the annual meeting of the European Association for the Study of Diabetes.
Intensive glucose control did not increase mortality or the rate of major macrovascular events in 10-year results from the ADVANCE ON (Action in Diabetes and Vascular Disease: Preterax and Diamicron MR Controlled Evaluation Post Trial Observational Study) trial. The results also showed for the first time that intensive glucose control produced a significant and large reduction in end-stage kidney disease, said Dr. Zoungas, an endocrinologist with the George Institute of the University of Sydney.
ADVANCE ON received partial funding from Servier. Dr. Zoungas has received honoraria from Servier as well as from Merck Sharp & Dohme, Bristol-Myers Squibb/AstraZeneca, Sanofi-Aventis, Novo Nordisk, and Amgen.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
AT EASD 2014
VIDEO: Insulin still mainstay of T2DM treatment
VIENNA – Although several new oral hypoglycemic drug classes have entered formularies over the past 5 years or so, none has supplanted insulin as a linchpin for managing patients with type 2 diabetes who fail to achieve adequate glycemic control with diet, exercise, and metformin treatment, Dr. David M. Nathan said in an interview during the annual meeting of the European Association for the Study of Diabetes.
Physicians should keep in mind that insulin remains a “powerful and important drug” for type 2 patients, said Dr. Nathan, chief of diabetes at Massachusetts General Hospital and professor of medicine at Harvard Medical School, both in Boston. Even though insulin’s price has risen recently, it is still a bargain, compared with the new drugs, he added.
Dr. Nathan said he had no relevant financial disclosures.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
VIENNA – Although several new oral hypoglycemic drug classes have entered formularies over the past 5 years or so, none has supplanted insulin as a linchpin for managing patients with type 2 diabetes who fail to achieve adequate glycemic control with diet, exercise, and metformin treatment, Dr. David M. Nathan said in an interview during the annual meeting of the European Association for the Study of Diabetes.
Physicians should keep in mind that insulin remains a “powerful and important drug” for type 2 patients, said Dr. Nathan, chief of diabetes at Massachusetts General Hospital and professor of medicine at Harvard Medical School, both in Boston. Even though insulin’s price has risen recently, it is still a bargain, compared with the new drugs, he added.
Dr. Nathan said he had no relevant financial disclosures.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
VIENNA – Although several new oral hypoglycemic drug classes have entered formularies over the past 5 years or so, none has supplanted insulin as a linchpin for managing patients with type 2 diabetes who fail to achieve adequate glycemic control with diet, exercise, and metformin treatment, Dr. David M. Nathan said in an interview during the annual meeting of the European Association for the Study of Diabetes.
Physicians should keep in mind that insulin remains a “powerful and important drug” for type 2 patients, said Dr. Nathan, chief of diabetes at Massachusetts General Hospital and professor of medicine at Harvard Medical School, both in Boston. Even though insulin’s price has risen recently, it is still a bargain, compared with the new drugs, he added.
Dr. Nathan said he had no relevant financial disclosures.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
AT EASD 2014
Triple-drug success foreshadows T2DM combo pill
VIENNA – Adding agents from two relatively new oral hypoglycemic drug classes to metformin produced a rapid, incremental reduction in hemoglobin A1c of more than 1% in two separate, phase III randomized controlled trials of patients with type 2 diabetes.
The efficacy and safety shown in the studies raises the possibility of formulating such three-agent combinations into single-pills, said Dr. Ralph A. DeFronzo, lead investigator for one of the studies.
The results “open the way to thinking about triple combinations. The next step is to concoct a single tablet with all three drugs together,” said Dr. DeFronzo at the annual meeting of the European Association for the Study of Diabetes.
The concept of treating patients with type 2 diabetes who fail to receive adequate glycemic control from diet and metformin treatment with the simultaneous addition of a drug from the sodium-glucose co-transporter 2 (SGLT-2) inhibitors class and a drug from the dipeptidyl peptidase-4 (DPP-4) inhibitor class “represents a new, proactive treatment paradigm, and appears to be an attractive option to safely and effectively bring difficult-to-treat metformin-failure patients to individualized glycemic goals,” said Dr. Julio Rosenstock, lead investigator for the second reported study and an endocrinologist at the University of Texas Southwestern Medical Center.
The study results reported by Dr. DeFronzo came from 677 patients with a HbA1c level of 7%-10% despite treatment with a stable metformin regimen for at least 12 weeks. Patients averaged about 56 years of age, and about three-quarters had been diagnosed with diabetes for more than 5 years. The researchers maintained all patients on their metformin dosage and randomized them to any of five treatment arms: 10 mg daily of the SGLT2 inhibitor empagliflozin (Jardiance), 25 mg daily empagliflozin, 5 mg daily of the DPP-4 inhibitor linagliptin (Trajenta), and two combination regimens: 5 mg linagliptin and 10 mg empagliflozin, and 5 mg linagliptin and 25 mg empagliflozin. The study’s primary endpoint was change from baseline in HbA1c after 24 weeks on treatment.
The results showed and average 1.19% drop in HbA1c after 24 weeks in patients on triple treatment with the higher dosage of empagliflozin, a 1.08% reduction in patients on the lower-dose, triple-treatment combination, and reductions that ranged from 0.62%-0.70% among the patients who received just one drug added to metformin. The differences between patients on either triple regimen and those on the three different dual regimens were statistically significant.
“Clearly, the combinations [of empagliflozin and linagliptin] have a greater effect than either of the drugs alone, but the effect is not completely additive,” noted Dr. DeFronzo, professor and chief of diabetes at the University of Texas Health Science Center, San Antonio.
The results also showed an average reduction in systolic blood pressure of 3-4 mm Hg among patients on triple therapy, similar to the effect from adding empagliflozin alone. The triple combinations were well tolerated, with a safety profile similar to monotherapy with these approved drugs, and confirmed hypoglycemia rates of 2%-4%, also similar to the rates seen with these drugs when used singly in combination with metformin.
The second study randomized 534 patients with a HbA1c level of 8%-12% while on stable metformin treatment. They averaged 53 years of age and had an average HbA1c of 8.9%. In addition to remaining on metformin, the researchers assigned patients to receive 10 mg of dapagliflozin (Farxiga) daily, 5 mg saxagliptin (Onglyza) daily, or a regimen that included all three drugs.
After 24 weeks on treatment, patients on the triple-drug regimen had an average reduction in HbA1c of 1.5%, while those on dapagliflozin plus metformin averaged a 1.2% reduction, while those on saxagliptin plus metformin had an average 0.9% drop. The differences between the triple-drug group and each of the two-drug groups were statistically significant for the study’s primary endpoint. The percentage of patients who achieved a HbA1c of less than 7% was 41% for patients on all three drugs, 22% for those on dapagliflozin plus metformin, and 18% for those on saxagliptin and metformin.
Patients on all three drugs also showed weight loss and blood pressure reductions roughly similar to the higher rates seen in the two control arms. The triple regimen was also well tolerated, with one patient having a minor hypoglycemic episode.
The empagliflozin and linagliptin study was sponsored by Boehringer Ingelheim, which markets both drugs. The dapagliflozin plus saxagliptin study was sponsored by Bristol-Myers Squibb and AstraZeneca, which market both drugs. Dr. DeFronzo is an adviser to and speaker for Boehringer Ingelheim, Bristol-Myers Squibb, AstraZeneca, and other drug companies. Dr. Rosenstock is a consultant to Boehringer Ingelheim, Bristol-Myers Squibb, AstraZeneca, and other drug companies.
Reducing tablet counts for patients is a way to move forward in managing type 2 diabetes. We already have formulations that combine two oral hypoglycemic drugs, and developing a single pill that combines three drugs seems like a logical next step. But the logistics of this are not simple. I have concerns about drug tolerability, and we need a much better understanding of the potential complementary effects of simultaneous treatment with metformin, a sodium-glucose co-transporter 2 (SGLT2) inhibitor, and a dipeptidyl peptidase-4 (DPP-4) inhibitor. In addition, in some patients, moving from treatment with a single drug to treatment with three drugs may be too great a leap. Sequential progression from one to two and then a third drug if still needed may be best for some patients.

|
Dr. Melanie Davies |
Combination of metformin, an SGLT2 inhibitor, and a DPP-4 inhibitor has been suggested by many diabetologists as an attractive option, and I agree it has promise. These drugs are reasonably well tolerated and have good effects on hemoglobin A1c as well as blood pressure and weight loss, with a low risk of causing hypoglycemia.
Dr. Melanie J. Davies is professor of diabetes medicine at the University of Leicester, England. She has been a consultant to and speaker for Boehringer Ingelheim and several other drug companies. She made these comments in an interview.
Reducing tablet counts for patients is a way to move forward in managing type 2 diabetes. We already have formulations that combine two oral hypoglycemic drugs, and developing a single pill that combines three drugs seems like a logical next step. But the logistics of this are not simple. I have concerns about drug tolerability, and we need a much better understanding of the potential complementary effects of simultaneous treatment with metformin, a sodium-glucose co-transporter 2 (SGLT2) inhibitor, and a dipeptidyl peptidase-4 (DPP-4) inhibitor. In addition, in some patients, moving from treatment with a single drug to treatment with three drugs may be too great a leap. Sequential progression from one to two and then a third drug if still needed may be best for some patients.

|
Dr. Melanie Davies |
Combination of metformin, an SGLT2 inhibitor, and a DPP-4 inhibitor has been suggested by many diabetologists as an attractive option, and I agree it has promise. These drugs are reasonably well tolerated and have good effects on hemoglobin A1c as well as blood pressure and weight loss, with a low risk of causing hypoglycemia.
Dr. Melanie J. Davies is professor of diabetes medicine at the University of Leicester, England. She has been a consultant to and speaker for Boehringer Ingelheim and several other drug companies. She made these comments in an interview.
Reducing tablet counts for patients is a way to move forward in managing type 2 diabetes. We already have formulations that combine two oral hypoglycemic drugs, and developing a single pill that combines three drugs seems like a logical next step. But the logistics of this are not simple. I have concerns about drug tolerability, and we need a much better understanding of the potential complementary effects of simultaneous treatment with metformin, a sodium-glucose co-transporter 2 (SGLT2) inhibitor, and a dipeptidyl peptidase-4 (DPP-4) inhibitor. In addition, in some patients, moving from treatment with a single drug to treatment with three drugs may be too great a leap. Sequential progression from one to two and then a third drug if still needed may be best for some patients.

|
Dr. Melanie Davies |
Combination of metformin, an SGLT2 inhibitor, and a DPP-4 inhibitor has been suggested by many diabetologists as an attractive option, and I agree it has promise. These drugs are reasonably well tolerated and have good effects on hemoglobin A1c as well as blood pressure and weight loss, with a low risk of causing hypoglycemia.
Dr. Melanie J. Davies is professor of diabetes medicine at the University of Leicester, England. She has been a consultant to and speaker for Boehringer Ingelheim and several other drug companies. She made these comments in an interview.
VIENNA – Adding agents from two relatively new oral hypoglycemic drug classes to metformin produced a rapid, incremental reduction in hemoglobin A1c of more than 1% in two separate, phase III randomized controlled trials of patients with type 2 diabetes.
The efficacy and safety shown in the studies raises the possibility of formulating such three-agent combinations into single-pills, said Dr. Ralph A. DeFronzo, lead investigator for one of the studies.
The results “open the way to thinking about triple combinations. The next step is to concoct a single tablet with all three drugs together,” said Dr. DeFronzo at the annual meeting of the European Association for the Study of Diabetes.
The concept of treating patients with type 2 diabetes who fail to receive adequate glycemic control from diet and metformin treatment with the simultaneous addition of a drug from the sodium-glucose co-transporter 2 (SGLT-2) inhibitors class and a drug from the dipeptidyl peptidase-4 (DPP-4) inhibitor class “represents a new, proactive treatment paradigm, and appears to be an attractive option to safely and effectively bring difficult-to-treat metformin-failure patients to individualized glycemic goals,” said Dr. Julio Rosenstock, lead investigator for the second reported study and an endocrinologist at the University of Texas Southwestern Medical Center.
The study results reported by Dr. DeFronzo came from 677 patients with a HbA1c level of 7%-10% despite treatment with a stable metformin regimen for at least 12 weeks. Patients averaged about 56 years of age, and about three-quarters had been diagnosed with diabetes for more than 5 years. The researchers maintained all patients on their metformin dosage and randomized them to any of five treatment arms: 10 mg daily of the SGLT2 inhibitor empagliflozin (Jardiance), 25 mg daily empagliflozin, 5 mg daily of the DPP-4 inhibitor linagliptin (Trajenta), and two combination regimens: 5 mg linagliptin and 10 mg empagliflozin, and 5 mg linagliptin and 25 mg empagliflozin. The study’s primary endpoint was change from baseline in HbA1c after 24 weeks on treatment.
The results showed and average 1.19% drop in HbA1c after 24 weeks in patients on triple treatment with the higher dosage of empagliflozin, a 1.08% reduction in patients on the lower-dose, triple-treatment combination, and reductions that ranged from 0.62%-0.70% among the patients who received just one drug added to metformin. The differences between patients on either triple regimen and those on the three different dual regimens were statistically significant.
“Clearly, the combinations [of empagliflozin and linagliptin] have a greater effect than either of the drugs alone, but the effect is not completely additive,” noted Dr. DeFronzo, professor and chief of diabetes at the University of Texas Health Science Center, San Antonio.
The results also showed an average reduction in systolic blood pressure of 3-4 mm Hg among patients on triple therapy, similar to the effect from adding empagliflozin alone. The triple combinations were well tolerated, with a safety profile similar to monotherapy with these approved drugs, and confirmed hypoglycemia rates of 2%-4%, also similar to the rates seen with these drugs when used singly in combination with metformin.
The second study randomized 534 patients with a HbA1c level of 8%-12% while on stable metformin treatment. They averaged 53 years of age and had an average HbA1c of 8.9%. In addition to remaining on metformin, the researchers assigned patients to receive 10 mg of dapagliflozin (Farxiga) daily, 5 mg saxagliptin (Onglyza) daily, or a regimen that included all three drugs.
After 24 weeks on treatment, patients on the triple-drug regimen had an average reduction in HbA1c of 1.5%, while those on dapagliflozin plus metformin averaged a 1.2% reduction, while those on saxagliptin plus metformin had an average 0.9% drop. The differences between the triple-drug group and each of the two-drug groups were statistically significant for the study’s primary endpoint. The percentage of patients who achieved a HbA1c of less than 7% was 41% for patients on all three drugs, 22% for those on dapagliflozin plus metformin, and 18% for those on saxagliptin and metformin.
Patients on all three drugs also showed weight loss and blood pressure reductions roughly similar to the higher rates seen in the two control arms. The triple regimen was also well tolerated, with one patient having a minor hypoglycemic episode.
The empagliflozin and linagliptin study was sponsored by Boehringer Ingelheim, which markets both drugs. The dapagliflozin plus saxagliptin study was sponsored by Bristol-Myers Squibb and AstraZeneca, which market both drugs. Dr. DeFronzo is an adviser to and speaker for Boehringer Ingelheim, Bristol-Myers Squibb, AstraZeneca, and other drug companies. Dr. Rosenstock is a consultant to Boehringer Ingelheim, Bristol-Myers Squibb, AstraZeneca, and other drug companies.
VIENNA – Adding agents from two relatively new oral hypoglycemic drug classes to metformin produced a rapid, incremental reduction in hemoglobin A1c of more than 1% in two separate, phase III randomized controlled trials of patients with type 2 diabetes.
The efficacy and safety shown in the studies raises the possibility of formulating such three-agent combinations into single-pills, said Dr. Ralph A. DeFronzo, lead investigator for one of the studies.
The results “open the way to thinking about triple combinations. The next step is to concoct a single tablet with all three drugs together,” said Dr. DeFronzo at the annual meeting of the European Association for the Study of Diabetes.
The concept of treating patients with type 2 diabetes who fail to receive adequate glycemic control from diet and metformin treatment with the simultaneous addition of a drug from the sodium-glucose co-transporter 2 (SGLT-2) inhibitors class and a drug from the dipeptidyl peptidase-4 (DPP-4) inhibitor class “represents a new, proactive treatment paradigm, and appears to be an attractive option to safely and effectively bring difficult-to-treat metformin-failure patients to individualized glycemic goals,” said Dr. Julio Rosenstock, lead investigator for the second reported study and an endocrinologist at the University of Texas Southwestern Medical Center.
The study results reported by Dr. DeFronzo came from 677 patients with a HbA1c level of 7%-10% despite treatment with a stable metformin regimen for at least 12 weeks. Patients averaged about 56 years of age, and about three-quarters had been diagnosed with diabetes for more than 5 years. The researchers maintained all patients on their metformin dosage and randomized them to any of five treatment arms: 10 mg daily of the SGLT2 inhibitor empagliflozin (Jardiance), 25 mg daily empagliflozin, 5 mg daily of the DPP-4 inhibitor linagliptin (Trajenta), and two combination regimens: 5 mg linagliptin and 10 mg empagliflozin, and 5 mg linagliptin and 25 mg empagliflozin. The study’s primary endpoint was change from baseline in HbA1c after 24 weeks on treatment.
The results showed and average 1.19% drop in HbA1c after 24 weeks in patients on triple treatment with the higher dosage of empagliflozin, a 1.08% reduction in patients on the lower-dose, triple-treatment combination, and reductions that ranged from 0.62%-0.70% among the patients who received just one drug added to metformin. The differences between patients on either triple regimen and those on the three different dual regimens were statistically significant.
“Clearly, the combinations [of empagliflozin and linagliptin] have a greater effect than either of the drugs alone, but the effect is not completely additive,” noted Dr. DeFronzo, professor and chief of diabetes at the University of Texas Health Science Center, San Antonio.
The results also showed an average reduction in systolic blood pressure of 3-4 mm Hg among patients on triple therapy, similar to the effect from adding empagliflozin alone. The triple combinations were well tolerated, with a safety profile similar to monotherapy with these approved drugs, and confirmed hypoglycemia rates of 2%-4%, also similar to the rates seen with these drugs when used singly in combination with metformin.
The second study randomized 534 patients with a HbA1c level of 8%-12% while on stable metformin treatment. They averaged 53 years of age and had an average HbA1c of 8.9%. In addition to remaining on metformin, the researchers assigned patients to receive 10 mg of dapagliflozin (Farxiga) daily, 5 mg saxagliptin (Onglyza) daily, or a regimen that included all three drugs.
After 24 weeks on treatment, patients on the triple-drug regimen had an average reduction in HbA1c of 1.5%, while those on dapagliflozin plus metformin averaged a 1.2% reduction, while those on saxagliptin plus metformin had an average 0.9% drop. The differences between the triple-drug group and each of the two-drug groups were statistically significant for the study’s primary endpoint. The percentage of patients who achieved a HbA1c of less than 7% was 41% for patients on all three drugs, 22% for those on dapagliflozin plus metformin, and 18% for those on saxagliptin and metformin.
Patients on all three drugs also showed weight loss and blood pressure reductions roughly similar to the higher rates seen in the two control arms. The triple regimen was also well tolerated, with one patient having a minor hypoglycemic episode.
The empagliflozin and linagliptin study was sponsored by Boehringer Ingelheim, which markets both drugs. The dapagliflozin plus saxagliptin study was sponsored by Bristol-Myers Squibb and AstraZeneca, which market both drugs. Dr. DeFronzo is an adviser to and speaker for Boehringer Ingelheim, Bristol-Myers Squibb, AstraZeneca, and other drug companies. Dr. Rosenstock is a consultant to Boehringer Ingelheim, Bristol-Myers Squibb, AstraZeneca, and other drug companies.
AT EASD 2014
Key clinical point: Augmenting a metformin regimen with a combination of a SGLT2 inhibitor drug and a DPP-4 inhibitor produced greater glycemic control than adding one of these drugs alone in patients with type 2 diabetes.
Major finding: Treatment with 25 mg empagliflozin, linagliptin, and metformin produced an average 1.19% reduction in HbA1c after 24 weeks. Treatment with dapagliflozin, saxagliptin, and metformin produced an average 1.5% reduction in HbA1c after 24 weeks.
Data source: Two separate randomized controlled studies that enrolled 677 and 534 patients, respectively, with type 2 diabetes.
Disclosures: The empagliflozin and linagliptin study was sponsored by Boehringer Ingelheim, which markets both drugs. The dapagliflozin plus saxagliptin study was sponsored by Bristol-Myers Squibb and AstraZeneca, which market both drugs. Dr. DeFronzo is an adviser to and speaker for Boehringer Ingelheim, Bristol-Myers Squibb, AstraZeneca, and other drug companies. Dr. Rosenstock is a consultant to Boehringer Ingelheim, Bristol-Myers Squibb, AstraZeneca, and other drug companies.
Mechanical heart valves create high pregnancy risk
BARCELONA – Women with mechanical heart valves who become pregnant face a very-high-risk pregnancy, with a 58% rate of an uncomplicated pregnancy resulting in a live birth, according to international registry data collected since 2007.
Pregnant women at high risk because of a mechanical heart valve need management by a multidisciplinary team at a referral center that focuses on these cases, a type of care that many of these women do not receive today, Dr. Jolien W. Roos-Hesselink said at the annual congress of the European Society of Cardiology.
“We believe management of these women needs to be better structured and organized,” said Dr. Roos-Hesselink, professor and head of the department of congenital cardiology at Erasmus Medical Center in Rotterdam, the Netherlands. “Pregnancy is a time of risk for any woman with structural heart disease, but for those with a mechanical valve are really high-risk patients,” she said in an interview.
“Most of these women are now cared for by a general cardiologist. They need a specialist in obstetric cardiology,” as well as care from other experts with experience in the types of complications these woman develop, said Dr. Roger J.C. Hall, professor of cardiology at Norfolk and Norwich (U.K.) University Hospital.
The data also showed that physicians around the world used any one of seven different anticoagulant regimens during these pregnancies, a strikingly high number that highlights uncertainty about which regimen is best, although heparin use during the first trimester was linked with a higher rate of valve thrombosis. The various regimens use different combinations of periods of treatment with unfractionated heparin, low-molecular-weight heparin, or a vitamin K antagonist drug during the first trimester, during weeks 14-36, and during the last weeks of pregnancy.
“We found large differences in management among different countries, physicians, and among individual patients. All the regimens have advantages and disadvantages” and are based on expert opinion with no prospect for a randomized, controlled trial, said Dr. Roos-Hesselink. For now, the numbers of women receiving each of the seven regimens remains too small for statistical analysis, but the researchers hope that eventually larger numbers may start to reveal which regimens work best, said Dr. Hall.
However, the available data showed two clear trends: Treatment with vitamin K antagonists was tied to an increased rate of miscarriages, and treatment with heparin during the first trimester was associated with an increased rate of valve thrombosis, noted Dr. Roos-Hesselink.
One other notable finding was that the risks faced by women with mechanical heart valves far exceeded the risk seen in women with tissue valves, and in women with other forms of mechanical heart disease but no valve prostheses. Adolescents and young women who need a heart valve should be part of a shared decision process that reviews the pros and cons of a mechanical and tissue valve, said Dr. Roos-Hesselink and Dr. Hall. A tissue valve is less durable, and so typically requires replacement sooner than does a mechanical valve. But in the current study, the pregnancy loss and complication rates among the 134 women with a tissue valve roughly matched the rates among the 2,620 women with no valve prosthesis, while the rates among 212 women with a mechanical valve ran much higher.
The Registry of Pregnancy and Cardiac Diseases began in 2007 through an initiative of two interest groups of the European Society of Cardiology, the valve group and the congenital heart disease group (Eur. Heart J. 2013;34:657-65). By September 2014, the registry had enrolled more than 3,600 pregnancies. The current report focused on the first 2,966 women enrolled, with an average age of 29 years. Slightly more than half the enrolled women had congenital heart disease, slightly fewer than a third had valvular heart disease (usually because of rheumatic heart disease), 7% had cardiomyopathy, and smaller number of women had other etiologies.
Maternal mortality averaged 1.4% for women with mechanical valves, 1.5% for those with tissue valves, and 0.2% for everyone else. Miscarriage rates were 16% for mothers with mechanical valves and 2% for everyone else, including those with tissues valves. Fetal mortality was 3% among women with mechanical valves and less than 1% for everyone else. Thrombotic and hemorrhagic events occurred in about 29% of women with mechanical valves, compared with less than 6% in everyone else. Ten of the women with mechanical valves (5%) developed valve thrombosis. The live birth rate was 80% for women with mechanical valves and 95% or better for everyone else.
Dr. Roos-Hesselink and Dr. Hall had no disclosures.
On Twitter @mitchelzoler
Dr. Hossein Almassi, FCCP, comments: One of the toughest decisions in valvular heart surgery is the selection of a valve prosthesis for a young female. Historically, age has been the major deciding factor in selecting a mechanical valve over a tissue valve. The current report is a testament to this difficulty, showing a lack of a standardized anticoagulation regimen and a much higher rate of pregnancy loss and complications with mechanical heart valves. Availability of transcatheter valve technology may make the tissue valves more attractive for this group of patients. Clearly, the care for these women would be best provided by a multidisciplinary team of specialists.
Dr. Almassi specializes in cardiothoracic surgery at the Medical College of Wisconsin in Milwaukee, Wisconsin.
Dr. Hossein Almassi, FCCP, comments: One of the toughest decisions in valvular heart surgery is the selection of a valve prosthesis for a young female. Historically, age has been the major deciding factor in selecting a mechanical valve over a tissue valve. The current report is a testament to this difficulty, showing a lack of a standardized anticoagulation regimen and a much higher rate of pregnancy loss and complications with mechanical heart valves. Availability of transcatheter valve technology may make the tissue valves more attractive for this group of patients. Clearly, the care for these women would be best provided by a multidisciplinary team of specialists.
Dr. Almassi specializes in cardiothoracic surgery at the Medical College of Wisconsin in Milwaukee, Wisconsin.
Dr. Hossein Almassi, FCCP, comments: One of the toughest decisions in valvular heart surgery is the selection of a valve prosthesis for a young female. Historically, age has been the major deciding factor in selecting a mechanical valve over a tissue valve. The current report is a testament to this difficulty, showing a lack of a standardized anticoagulation regimen and a much higher rate of pregnancy loss and complications with mechanical heart valves. Availability of transcatheter valve technology may make the tissue valves more attractive for this group of patients. Clearly, the care for these women would be best provided by a multidisciplinary team of specialists.
Dr. Almassi specializes in cardiothoracic surgery at the Medical College of Wisconsin in Milwaukee, Wisconsin.
BARCELONA – Women with mechanical heart valves who become pregnant face a very-high-risk pregnancy, with a 58% rate of an uncomplicated pregnancy resulting in a live birth, according to international registry data collected since 2007.
Pregnant women at high risk because of a mechanical heart valve need management by a multidisciplinary team at a referral center that focuses on these cases, a type of care that many of these women do not receive today, Dr. Jolien W. Roos-Hesselink said at the annual congress of the European Society of Cardiology.
“We believe management of these women needs to be better structured and organized,” said Dr. Roos-Hesselink, professor and head of the department of congenital cardiology at Erasmus Medical Center in Rotterdam, the Netherlands. “Pregnancy is a time of risk for any woman with structural heart disease, but for those with a mechanical valve are really high-risk patients,” she said in an interview.
“Most of these women are now cared for by a general cardiologist. They need a specialist in obstetric cardiology,” as well as care from other experts with experience in the types of complications these woman develop, said Dr. Roger J.C. Hall, professor of cardiology at Norfolk and Norwich (U.K.) University Hospital.
The data also showed that physicians around the world used any one of seven different anticoagulant regimens during these pregnancies, a strikingly high number that highlights uncertainty about which regimen is best, although heparin use during the first trimester was linked with a higher rate of valve thrombosis. The various regimens use different combinations of periods of treatment with unfractionated heparin, low-molecular-weight heparin, or a vitamin K antagonist drug during the first trimester, during weeks 14-36, and during the last weeks of pregnancy.
“We found large differences in management among different countries, physicians, and among individual patients. All the regimens have advantages and disadvantages” and are based on expert opinion with no prospect for a randomized, controlled trial, said Dr. Roos-Hesselink. For now, the numbers of women receiving each of the seven regimens remains too small for statistical analysis, but the researchers hope that eventually larger numbers may start to reveal which regimens work best, said Dr. Hall.
However, the available data showed two clear trends: Treatment with vitamin K antagonists was tied to an increased rate of miscarriages, and treatment with heparin during the first trimester was associated with an increased rate of valve thrombosis, noted Dr. Roos-Hesselink.
One other notable finding was that the risks faced by women with mechanical heart valves far exceeded the risk seen in women with tissue valves, and in women with other forms of mechanical heart disease but no valve prostheses. Adolescents and young women who need a heart valve should be part of a shared decision process that reviews the pros and cons of a mechanical and tissue valve, said Dr. Roos-Hesselink and Dr. Hall. A tissue valve is less durable, and so typically requires replacement sooner than does a mechanical valve. But in the current study, the pregnancy loss and complication rates among the 134 women with a tissue valve roughly matched the rates among the 2,620 women with no valve prosthesis, while the rates among 212 women with a mechanical valve ran much higher.
The Registry of Pregnancy and Cardiac Diseases began in 2007 through an initiative of two interest groups of the European Society of Cardiology, the valve group and the congenital heart disease group (Eur. Heart J. 2013;34:657-65). By September 2014, the registry had enrolled more than 3,600 pregnancies. The current report focused on the first 2,966 women enrolled, with an average age of 29 years. Slightly more than half the enrolled women had congenital heart disease, slightly fewer than a third had valvular heart disease (usually because of rheumatic heart disease), 7% had cardiomyopathy, and smaller number of women had other etiologies.
Maternal mortality averaged 1.4% for women with mechanical valves, 1.5% for those with tissue valves, and 0.2% for everyone else. Miscarriage rates were 16% for mothers with mechanical valves and 2% for everyone else, including those with tissues valves. Fetal mortality was 3% among women with mechanical valves and less than 1% for everyone else. Thrombotic and hemorrhagic events occurred in about 29% of women with mechanical valves, compared with less than 6% in everyone else. Ten of the women with mechanical valves (5%) developed valve thrombosis. The live birth rate was 80% for women with mechanical valves and 95% or better for everyone else.
Dr. Roos-Hesselink and Dr. Hall had no disclosures.
On Twitter @mitchelzoler
BARCELONA – Women with mechanical heart valves who become pregnant face a very-high-risk pregnancy, with a 58% rate of an uncomplicated pregnancy resulting in a live birth, according to international registry data collected since 2007.
Pregnant women at high risk because of a mechanical heart valve need management by a multidisciplinary team at a referral center that focuses on these cases, a type of care that many of these women do not receive today, Dr. Jolien W. Roos-Hesselink said at the annual congress of the European Society of Cardiology.
“We believe management of these women needs to be better structured and organized,” said Dr. Roos-Hesselink, professor and head of the department of congenital cardiology at Erasmus Medical Center in Rotterdam, the Netherlands. “Pregnancy is a time of risk for any woman with structural heart disease, but for those with a mechanical valve are really high-risk patients,” she said in an interview.
“Most of these women are now cared for by a general cardiologist. They need a specialist in obstetric cardiology,” as well as care from other experts with experience in the types of complications these woman develop, said Dr. Roger J.C. Hall, professor of cardiology at Norfolk and Norwich (U.K.) University Hospital.
The data also showed that physicians around the world used any one of seven different anticoagulant regimens during these pregnancies, a strikingly high number that highlights uncertainty about which regimen is best, although heparin use during the first trimester was linked with a higher rate of valve thrombosis. The various regimens use different combinations of periods of treatment with unfractionated heparin, low-molecular-weight heparin, or a vitamin K antagonist drug during the first trimester, during weeks 14-36, and during the last weeks of pregnancy.
“We found large differences in management among different countries, physicians, and among individual patients. All the regimens have advantages and disadvantages” and are based on expert opinion with no prospect for a randomized, controlled trial, said Dr. Roos-Hesselink. For now, the numbers of women receiving each of the seven regimens remains too small for statistical analysis, but the researchers hope that eventually larger numbers may start to reveal which regimens work best, said Dr. Hall.
However, the available data showed two clear trends: Treatment with vitamin K antagonists was tied to an increased rate of miscarriages, and treatment with heparin during the first trimester was associated with an increased rate of valve thrombosis, noted Dr. Roos-Hesselink.
One other notable finding was that the risks faced by women with mechanical heart valves far exceeded the risk seen in women with tissue valves, and in women with other forms of mechanical heart disease but no valve prostheses. Adolescents and young women who need a heart valve should be part of a shared decision process that reviews the pros and cons of a mechanical and tissue valve, said Dr. Roos-Hesselink and Dr. Hall. A tissue valve is less durable, and so typically requires replacement sooner than does a mechanical valve. But in the current study, the pregnancy loss and complication rates among the 134 women with a tissue valve roughly matched the rates among the 2,620 women with no valve prosthesis, while the rates among 212 women with a mechanical valve ran much higher.
The Registry of Pregnancy and Cardiac Diseases began in 2007 through an initiative of two interest groups of the European Society of Cardiology, the valve group and the congenital heart disease group (Eur. Heart J. 2013;34:657-65). By September 2014, the registry had enrolled more than 3,600 pregnancies. The current report focused on the first 2,966 women enrolled, with an average age of 29 years. Slightly more than half the enrolled women had congenital heart disease, slightly fewer than a third had valvular heart disease (usually because of rheumatic heart disease), 7% had cardiomyopathy, and smaller number of women had other etiologies.
Maternal mortality averaged 1.4% for women with mechanical valves, 1.5% for those with tissue valves, and 0.2% for everyone else. Miscarriage rates were 16% for mothers with mechanical valves and 2% for everyone else, including those with tissues valves. Fetal mortality was 3% among women with mechanical valves and less than 1% for everyone else. Thrombotic and hemorrhagic events occurred in about 29% of women with mechanical valves, compared with less than 6% in everyone else. Ten of the women with mechanical valves (5%) developed valve thrombosis. The live birth rate was 80% for women with mechanical valves and 95% or better for everyone else.
Dr. Roos-Hesselink and Dr. Hall had no disclosures.
On Twitter @mitchelzoler
AT ESC 2014
Key clinical point: Pregnant women with mechanical heart valves have a very high rate of pregnancy complications and miscarriages.
Major finding: The rate of live births without complications was 58% in women with a mechanical heart valve.
Data source: ROPAC registry of 2,966 pregnant women with structural heart disease from 132 centers in 48 countries.
Disclosures: Dr. Roos-Hesselink and Dr. Hall had no disclosures.
VIDEO: A fib screening finds 5% of elderly undiagnosed
BARCELONA – Roughly 5% of people aged 75 years or older have undiagnosed atrial fibrillation, based on a population-based screening study in Sweden that has assessed nearly 7,000 people, Dr. Mårten Rosenqvist said during an interview at the annual congress of the European Society of Cardiology.
Once diagnosed with atrial fibrillation, all these people immediately qualified for anticoagulant treatment because of their age-related stroke risk. The StrokeStop study will follow all the screened people for 5 years, as well as a concurrently assembled cohort of unscreened controls, to determine the benefit from screening for preventing strokes. “If we can reduce the rate of stroke, it would be a reason to implement a national atrial fibrillation screening program” for all people aged 75 years and older, said Dr. Rosenqvist, professor of cardiology at the Karolinska Institute in Stockholm.
Dr. Rosenqvist said that he is a consultant to Zenicor, a company that markets an ECG-based device for diagnosing atrial fibrillation being used in the StrokeStop study. He also is a consultant to several drug companies.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter@mitchelzoler
BARCELONA – Roughly 5% of people aged 75 years or older have undiagnosed atrial fibrillation, based on a population-based screening study in Sweden that has assessed nearly 7,000 people, Dr. Mårten Rosenqvist said during an interview at the annual congress of the European Society of Cardiology.
Once diagnosed with atrial fibrillation, all these people immediately qualified for anticoagulant treatment because of their age-related stroke risk. The StrokeStop study will follow all the screened people for 5 years, as well as a concurrently assembled cohort of unscreened controls, to determine the benefit from screening for preventing strokes. “If we can reduce the rate of stroke, it would be a reason to implement a national atrial fibrillation screening program” for all people aged 75 years and older, said Dr. Rosenqvist, professor of cardiology at the Karolinska Institute in Stockholm.
Dr. Rosenqvist said that he is a consultant to Zenicor, a company that markets an ECG-based device for diagnosing atrial fibrillation being used in the StrokeStop study. He also is a consultant to several drug companies.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter@mitchelzoler
BARCELONA – Roughly 5% of people aged 75 years or older have undiagnosed atrial fibrillation, based on a population-based screening study in Sweden that has assessed nearly 7,000 people, Dr. Mårten Rosenqvist said during an interview at the annual congress of the European Society of Cardiology.
Once diagnosed with atrial fibrillation, all these people immediately qualified for anticoagulant treatment because of their age-related stroke risk. The StrokeStop study will follow all the screened people for 5 years, as well as a concurrently assembled cohort of unscreened controls, to determine the benefit from screening for preventing strokes. “If we can reduce the rate of stroke, it would be a reason to implement a national atrial fibrillation screening program” for all people aged 75 years and older, said Dr. Rosenqvist, professor of cardiology at the Karolinska Institute in Stockholm.
Dr. Rosenqvist said that he is a consultant to Zenicor, a company that markets an ECG-based device for diagnosing atrial fibrillation being used in the StrokeStop study. He also is a consultant to several drug companies.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter@mitchelzoler
AT THE ESC CONGRESS 2014
Thrombectomy during primary PCI lacks 1-year benefits
BARCELONA – Routine thrombus aspiration in patients with acute ST-elevation myocardial infarction failed to produce a 1-year survival benefit over conventional care in a multicenter, randomized trial with more than 7,000 patients, negating a previously reported 1-year survival boost from thrombus aspiration in a single-center study with about 1,000 patients.
"Our recommendation is that routine thrombus aspiration not be done" in patients with ST-elevation myocardial infarction [STEMI] undergoing percutaneous coronary intervention [PCI], Dr. Bo Lagerqvist said at the annual congress of the European Society of Cardiology. A year ago, he and his associates reported no 30-day mortality benefit from routine thrombus aspiration during PCI in the same TASTE (Thrombus Aspiration in ST-Elevation Myocardial Infarction in Scandinavia) study (N. Eng. J. Med. 2013;369:1587-97).
But Dr. Lagerqvist and other experts left the door open to thrombus aspiration for cause during PCI, a niche that likely will continue to fuel interest in a technique that has soared in popularity despite its unproven benefit.
"The abundance of devices [for performing thrombus aspiration] on the market contrasts with the paucity of data" showing benefit, commented Dr. Gilles Montalescot, professor of cardiology at University Hospital La Pitié Salpêtrière in Paris.
"I don’t expect a reduction in use of thrombus aspiration despite the results," commented Dr. Carlo Di Mario, an interventional cardiologist at Royal Brompton Hospital in London. "Cardiologists use thrombus aspiration to create a larger lumen" when an infarct-related artery is blocked with a large thrombus. "The alternative it to use a balloon, which can cause embolization and which just exchanges one device for another."
TASTE randomized 7,244 acute STEMI patients undergoing PCI at 29 centers in Sweden and 1 center each in Iceland and Denmark. They averaged about 66 years of age, and 78% of patients had either full occlusion or low coronary flow through the infarct-related artery at the time of hospitalization.
One year after intervention, the rates of all-cause death, repeat STEMI, and of stent thrombosis were each identical in the patients who received thrombus aspiration and those who did not, reported Dr Lagerqvist, a cardiologist at Uppsala (Sweden) University Hospital. In addition, the absence of a benefit was consistent across several subgroups included in the analysis, which meant the researchers found no type of acute STEMI patient who clearly benefited from thrombus aspiration. Concurrent with his report at the meeting the results also appeared online (N. Engl. J. Med. 2014 [doi: 10.1056/NEJMoa1405707]).
"We saw no danger from thrombus aspiration, and it is relatively inexpensive, so it is possible to use it, but we don’t know how to identify the patients who might benefit from it, even patients with a full coronary occlusion" from thrombus, he said during a press conference. Before the TASTE results were known, roughly 50% of Swedish primary PCI cases for acute STEMI also received thrombus aspiration; today, use is "very, very low," Dr. Lagerqvist said.
Dr. Lagerqvist and others acknowledged that even more definitive data on the role of thrombus aspiration during PCI for acute STEMI will come from the TOTAL (Trial of Routine Aspiration Thrombectomy With PCI vs. PCI Alone in Patients With STEMI Undergoing Primary PCI) study, which has randomized nearly 11,000 patients and is expected to report results next year. Dr. Sanjit Jolly, an interventional cardiologist at McMaster University in Hamilton, Ont., and lead investigator of TOTAL, made this written statement about the new TASTE report:
"Thrombectomy is a one-time intervention unlike an ongoing drug therapy (i.e., ticagrelor). Therefore, the hypothesis that late events may have been more responsive to thrombectomy than early events is likely unrealistic. While TASTE excludes a 50% reduction in mortality with thrombectomy, it was not powered for realistic 20%-25% reductions in outcomes. Therefore, clinicians should reserve judgment and await the results of the largest trial examining this question, the TOTAL trial. The TOTAL trial has randomized nearly 11,000 patients to thrombectomy vs. PCI alone in STEMI and uses a composite outcome with blinded adjudication of outcomes. The TOTAL trial is powered for a 20% relative risk reduction and is expected to be presented in early 2015."
TASTE received no commercial support. Dr. Lagerqvist and Dr. Di Marco had no disclosures. Dr. Montalescot has received honoraria and research support from several companies including Medtronic, which markets a thrombectomy catheter. Dr. Jolly has received research support from Medtronic.
On Twitter @mitchelzoler
Thrombus aspiration is easy to use, it can make percutaneous coronary interventions easier to perform, and is probably safe, but there is insufficient evidence to recommend its use. One-year results from a relatively small, single-center study had suggested that it produced beneficial effects. But the TASTE study is the largest test of thrombectomy to date, and we must accept its findings.
It is not good to have so many devices on the market for performing thrombus aspiration when no data show what they achieve. The results from the TOTAL trial next year should be definitive, but I think that TOTAL will bring an end to thrombus aspiration during primary PCI. Thrombectomy risks the danger of displaced thrombus. When a large thrombus totally occludes an artery, there is a need for thrombus aspiration, so I don’t think the technique will totally disappear. But routine use for all patients will die out. We also need to study the possible impact of thrombectomy on reducing the risk for heart failure following STEMI.
Dr. Gilles Montalescot is professor of cardiology at University Hospital La Pitié Salpêtrière in Paris. He has received honoraria and grant support from several drug and device companies, including Medtronic, which markets a thrombectomy device. He made these comments as designated discussant for the study and in an interview.
Thrombus aspiration is easy to use, it can make percutaneous coronary interventions easier to perform, and is probably safe, but there is insufficient evidence to recommend its use. One-year results from a relatively small, single-center study had suggested that it produced beneficial effects. But the TASTE study is the largest test of thrombectomy to date, and we must accept its findings.
It is not good to have so many devices on the market for performing thrombus aspiration when no data show what they achieve. The results from the TOTAL trial next year should be definitive, but I think that TOTAL will bring an end to thrombus aspiration during primary PCI. Thrombectomy risks the danger of displaced thrombus. When a large thrombus totally occludes an artery, there is a need for thrombus aspiration, so I don’t think the technique will totally disappear. But routine use for all patients will die out. We also need to study the possible impact of thrombectomy on reducing the risk for heart failure following STEMI.
Dr. Gilles Montalescot is professor of cardiology at University Hospital La Pitié Salpêtrière in Paris. He has received honoraria and grant support from several drug and device companies, including Medtronic, which markets a thrombectomy device. He made these comments as designated discussant for the study and in an interview.
Thrombus aspiration is easy to use, it can make percutaneous coronary interventions easier to perform, and is probably safe, but there is insufficient evidence to recommend its use. One-year results from a relatively small, single-center study had suggested that it produced beneficial effects. But the TASTE study is the largest test of thrombectomy to date, and we must accept its findings.
It is not good to have so many devices on the market for performing thrombus aspiration when no data show what they achieve. The results from the TOTAL trial next year should be definitive, but I think that TOTAL will bring an end to thrombus aspiration during primary PCI. Thrombectomy risks the danger of displaced thrombus. When a large thrombus totally occludes an artery, there is a need for thrombus aspiration, so I don’t think the technique will totally disappear. But routine use for all patients will die out. We also need to study the possible impact of thrombectomy on reducing the risk for heart failure following STEMI.
Dr. Gilles Montalescot is professor of cardiology at University Hospital La Pitié Salpêtrière in Paris. He has received honoraria and grant support from several drug and device companies, including Medtronic, which markets a thrombectomy device. He made these comments as designated discussant for the study and in an interview.
BARCELONA – Routine thrombus aspiration in patients with acute ST-elevation myocardial infarction failed to produce a 1-year survival benefit over conventional care in a multicenter, randomized trial with more than 7,000 patients, negating a previously reported 1-year survival boost from thrombus aspiration in a single-center study with about 1,000 patients.
"Our recommendation is that routine thrombus aspiration not be done" in patients with ST-elevation myocardial infarction [STEMI] undergoing percutaneous coronary intervention [PCI], Dr. Bo Lagerqvist said at the annual congress of the European Society of Cardiology. A year ago, he and his associates reported no 30-day mortality benefit from routine thrombus aspiration during PCI in the same TASTE (Thrombus Aspiration in ST-Elevation Myocardial Infarction in Scandinavia) study (N. Eng. J. Med. 2013;369:1587-97).
But Dr. Lagerqvist and other experts left the door open to thrombus aspiration for cause during PCI, a niche that likely will continue to fuel interest in a technique that has soared in popularity despite its unproven benefit.
"The abundance of devices [for performing thrombus aspiration] on the market contrasts with the paucity of data" showing benefit, commented Dr. Gilles Montalescot, professor of cardiology at University Hospital La Pitié Salpêtrière in Paris.
"I don’t expect a reduction in use of thrombus aspiration despite the results," commented Dr. Carlo Di Mario, an interventional cardiologist at Royal Brompton Hospital in London. "Cardiologists use thrombus aspiration to create a larger lumen" when an infarct-related artery is blocked with a large thrombus. "The alternative it to use a balloon, which can cause embolization and which just exchanges one device for another."
TASTE randomized 7,244 acute STEMI patients undergoing PCI at 29 centers in Sweden and 1 center each in Iceland and Denmark. They averaged about 66 years of age, and 78% of patients had either full occlusion or low coronary flow through the infarct-related artery at the time of hospitalization.
One year after intervention, the rates of all-cause death, repeat STEMI, and of stent thrombosis were each identical in the patients who received thrombus aspiration and those who did not, reported Dr Lagerqvist, a cardiologist at Uppsala (Sweden) University Hospital. In addition, the absence of a benefit was consistent across several subgroups included in the analysis, which meant the researchers found no type of acute STEMI patient who clearly benefited from thrombus aspiration. Concurrent with his report at the meeting the results also appeared online (N. Engl. J. Med. 2014 [doi: 10.1056/NEJMoa1405707]).
"We saw no danger from thrombus aspiration, and it is relatively inexpensive, so it is possible to use it, but we don’t know how to identify the patients who might benefit from it, even patients with a full coronary occlusion" from thrombus, he said during a press conference. Before the TASTE results were known, roughly 50% of Swedish primary PCI cases for acute STEMI also received thrombus aspiration; today, use is "very, very low," Dr. Lagerqvist said.
Dr. Lagerqvist and others acknowledged that even more definitive data on the role of thrombus aspiration during PCI for acute STEMI will come from the TOTAL (Trial of Routine Aspiration Thrombectomy With PCI vs. PCI Alone in Patients With STEMI Undergoing Primary PCI) study, which has randomized nearly 11,000 patients and is expected to report results next year. Dr. Sanjit Jolly, an interventional cardiologist at McMaster University in Hamilton, Ont., and lead investigator of TOTAL, made this written statement about the new TASTE report:
"Thrombectomy is a one-time intervention unlike an ongoing drug therapy (i.e., ticagrelor). Therefore, the hypothesis that late events may have been more responsive to thrombectomy than early events is likely unrealistic. While TASTE excludes a 50% reduction in mortality with thrombectomy, it was not powered for realistic 20%-25% reductions in outcomes. Therefore, clinicians should reserve judgment and await the results of the largest trial examining this question, the TOTAL trial. The TOTAL trial has randomized nearly 11,000 patients to thrombectomy vs. PCI alone in STEMI and uses a composite outcome with blinded adjudication of outcomes. The TOTAL trial is powered for a 20% relative risk reduction and is expected to be presented in early 2015."
TASTE received no commercial support. Dr. Lagerqvist and Dr. Di Marco had no disclosures. Dr. Montalescot has received honoraria and research support from several companies including Medtronic, which markets a thrombectomy catheter. Dr. Jolly has received research support from Medtronic.
On Twitter @mitchelzoler
BARCELONA – Routine thrombus aspiration in patients with acute ST-elevation myocardial infarction failed to produce a 1-year survival benefit over conventional care in a multicenter, randomized trial with more than 7,000 patients, negating a previously reported 1-year survival boost from thrombus aspiration in a single-center study with about 1,000 patients.
"Our recommendation is that routine thrombus aspiration not be done" in patients with ST-elevation myocardial infarction [STEMI] undergoing percutaneous coronary intervention [PCI], Dr. Bo Lagerqvist said at the annual congress of the European Society of Cardiology. A year ago, he and his associates reported no 30-day mortality benefit from routine thrombus aspiration during PCI in the same TASTE (Thrombus Aspiration in ST-Elevation Myocardial Infarction in Scandinavia) study (N. Eng. J. Med. 2013;369:1587-97).
But Dr. Lagerqvist and other experts left the door open to thrombus aspiration for cause during PCI, a niche that likely will continue to fuel interest in a technique that has soared in popularity despite its unproven benefit.
"The abundance of devices [for performing thrombus aspiration] on the market contrasts with the paucity of data" showing benefit, commented Dr. Gilles Montalescot, professor of cardiology at University Hospital La Pitié Salpêtrière in Paris.
"I don’t expect a reduction in use of thrombus aspiration despite the results," commented Dr. Carlo Di Mario, an interventional cardiologist at Royal Brompton Hospital in London. "Cardiologists use thrombus aspiration to create a larger lumen" when an infarct-related artery is blocked with a large thrombus. "The alternative it to use a balloon, which can cause embolization and which just exchanges one device for another."
TASTE randomized 7,244 acute STEMI patients undergoing PCI at 29 centers in Sweden and 1 center each in Iceland and Denmark. They averaged about 66 years of age, and 78% of patients had either full occlusion or low coronary flow through the infarct-related artery at the time of hospitalization.
One year after intervention, the rates of all-cause death, repeat STEMI, and of stent thrombosis were each identical in the patients who received thrombus aspiration and those who did not, reported Dr Lagerqvist, a cardiologist at Uppsala (Sweden) University Hospital. In addition, the absence of a benefit was consistent across several subgroups included in the analysis, which meant the researchers found no type of acute STEMI patient who clearly benefited from thrombus aspiration. Concurrent with his report at the meeting the results also appeared online (N. Engl. J. Med. 2014 [doi: 10.1056/NEJMoa1405707]).
"We saw no danger from thrombus aspiration, and it is relatively inexpensive, so it is possible to use it, but we don’t know how to identify the patients who might benefit from it, even patients with a full coronary occlusion" from thrombus, he said during a press conference. Before the TASTE results were known, roughly 50% of Swedish primary PCI cases for acute STEMI also received thrombus aspiration; today, use is "very, very low," Dr. Lagerqvist said.
Dr. Lagerqvist and others acknowledged that even more definitive data on the role of thrombus aspiration during PCI for acute STEMI will come from the TOTAL (Trial of Routine Aspiration Thrombectomy With PCI vs. PCI Alone in Patients With STEMI Undergoing Primary PCI) study, which has randomized nearly 11,000 patients and is expected to report results next year. Dr. Sanjit Jolly, an interventional cardiologist at McMaster University in Hamilton, Ont., and lead investigator of TOTAL, made this written statement about the new TASTE report:
"Thrombectomy is a one-time intervention unlike an ongoing drug therapy (i.e., ticagrelor). Therefore, the hypothesis that late events may have been more responsive to thrombectomy than early events is likely unrealistic. While TASTE excludes a 50% reduction in mortality with thrombectomy, it was not powered for realistic 20%-25% reductions in outcomes. Therefore, clinicians should reserve judgment and await the results of the largest trial examining this question, the TOTAL trial. The TOTAL trial has randomized nearly 11,000 patients to thrombectomy vs. PCI alone in STEMI and uses a composite outcome with blinded adjudication of outcomes. The TOTAL trial is powered for a 20% relative risk reduction and is expected to be presented in early 2015."
TASTE received no commercial support. Dr. Lagerqvist and Dr. Di Marco had no disclosures. Dr. Montalescot has received honoraria and research support from several companies including Medtronic, which markets a thrombectomy catheter. Dr. Jolly has received research support from Medtronic.
On Twitter @mitchelzoler
AT ESC 2014
Key clinical point: Thrombus aspiration during primary PCI in acute STEMI patients produced no survival benefit 1 year after treatment in a randomized trial with 7,000 patients.
Major finding: The 1-year all-cause death rate was 5.3% in patients who received thrombectomy and 5.6% in those who did not, a nonsignificant difference.
Data source: TASTE, a multicenter study with 7,244 acute STEMI patients who underwent primary PCI.
Disclosures: TASTE received no commercial support. Dr. Lagerqvist and Dr. Di Marco had no disclosures. Dr. Montalescot has received honoraria and research support from several companies including Medtronic, which markets a thrombectomy catheter. Dr. Jolly has received research support from Medtronic.
Modest hypertension control in diabetic patients boosts survival
BARCELONA – A modest amount of blood pressure control over an average of 4 years produced a significant, long-term survival benefit in patients with type 2 diabetes, based on a 10-year follow-up of more than 8,000 patients.
"It is critically important to maintain active blood-pressure lowering in both the short and long term in order to derive the greatest possible reductions in mortality and major cardiovascular events" in patients with type 2 diabetes, Dr. John Chalmers said at the annual congress of the European Society of Cardiology.
The results he reported showed that during 10 years of total follow-up, 4 years of concerted therapy with a dual antihypertensive-drug formulation on top of background therapy produced an average drop in blood pressure of 5.6/2.2 mm Hg, which was linked to a relative 9% decrease in mortality through the entire 10-year period.
Dr. Chalmers’ analysis used data from an average 6-year follow-up of patients after they completed a 4-year trial that had randomized them to treatment with either the combined formulation of the angiotensin-converting enzyme inhibitor perindopril and the diuretic indapamide, or to placebo. The 6-year assessment of these patients after they finished the closely regulated treatment phase while in the clinical trial is "the longest follow-up on the impact of blood pressure reduction in patients with diabetes," commented Dr. Lars Rydén, a cardiologist at the Karolinska Institute in Stockholm.
"The greatest part of the overall, cumulative benefit [over a 10-year period] was contributed by the carry forward of the in-trial benefits of active BP lowering," said Dr. Chalmers, senior director of the George Institute for Global Health in Sydney, Australia.
His study focused on data collected from patients enrolled in the ADVANCE (Action in Diabetes and Vascular Disease: Preterax and Diamicron-MR Controlled Evaluation) trial. This study randomized 11,140 patients with type 2 diabetes who were at least 55 years old and had at least one additional cardiovascular disease risk factor, to treatment with the combined formulation on top of their preexisting medications or to placebo plus existing medications (Lancet 2007;370:829-40). Patients in the placebo group were unable to receive an angiotensin-converting enzyme inhibitor during the study. The study was sponsored by Servier, the company that markets a combined perindopril and indapamide formulation (Preterax).
At enrollment, ADVANCE patients averaged 66 years old, had an average 8-year history of type 2 diabetes, and had an average blood pressure of 145/81 mm Hg. Three-quarters were on an antihypertensive regimen of some kind at entry into the study.
During the study’s 4 years of active treatment, patients randomized to receive the perindopril and indapamide combination had significantly reduced blood pressure, compared with the control patients. After 4 years, this linked to a 9% relative drop in the combined incidence of major macrovascular and microvascular events (including cardiovascular death, nonfatal stroke, and nonfatal myocardial infarction). Compared with the control group, this cut in events was statistically significant for the study’s primary endpoint. The antihypertensive intervention also was linked to a statistically significant, 14% relative drop in all-cause deaths.
To assess the longer-term impact of this 4-year intervention, Dr. Chalmers and his associates ran a post-trial observational study, ADVANCE-ON. Initially, 8,494 of the patients who completed ADVANCE (83% of surviving patients) agreed to participate in ADVANCE-ON; after another 6 years of follow-up, 5,131 patients remained under observation. During the extended 6 years of follow-up, patients from both the initial intervention arm and the initial control arm all maintained average blood pressures of roughly 137/75 mm Hg. The blood pressures of the original intervention and control patient arms "fully converged," Dr. Chalmers said.
During this 6-year period, patients received care from their personal physicians and could receive any antihypertensive regimen their physician prescribed. At their first follow-up medical examination after the end of the main ADVANCE study, about 40% of patients were on no antihypertensive drugs, 23% were on one drug, 21% were on two drugs, and the remainder were on higher numbers of antihypertensive drugs.
During the 6-year post-trial period, patients who had previously been in the active-treatment arm had a 6% relative reduction in all-cause mortality, compared with patients originally in the control arm, a difference that was not significant.
But when the researchers combined the 6-year post-trial follow-up with the in-trial results for a total median 10-year follow-up, they found an overall 9% relative cut in mortality in the intervention patients, compared with the controls, and a relative 12% drop in cardiovascular mortality.
The persistent, long-term benefits were "attenuated" compared with the shorter-term benefits seen during the active phase, but nonetheless they persisted and were consistent across all subgroups studied, Dr. Chalmers said.
The ADVANCE and ADVANCE-ON studies were partly funded by Servier. Dr. Chalmers has received research grants and honoraria from that company. Dr. Rydén has been an adviser to AstraZeneca, Roche, Bristol-Myers Squibb, and Sanofi, and received research grants from Roche.
On Twitter @mitchelzoler
The findings from ADVANCE-ON are important. Clinicians need clear evidence that controlling a patient’s blood pressure produces long-term benefits, and the results from ADVANCE-ON provide this. We need many more studies that track the long-term impact of blood pressure reduction. Currently, a large fraction of patients with diabetes are not maintained at the recommended blood pressure levels. This report presents the longest follow-up of any antihypertensive intervention in patients with diabetes.
The findings also show that a small blood pressure reduction can have an important and long-lasting effect. This highlights the value of blood pressure control, even when the reduction achieved in an individual patient seems small.

|
|
I believe that the effects linked with blood pressure reduction in ADVANCE-ON are likely generalizable to any antihypertensive regimen that produces a similar magnitude of effect. It is the blood pressure effect rather than the specific antihypertensive drugs used that is that critical, although drugs that affect the renin-angiotensin-aldosterone system are clearly the first choice for patients with diabetes and hypertension.
Dr. Lars Rydén is a cardiologist at the Karolinska Institute in Stockholm. He has been an adviser to AstraZeneca, Roche, Bristol-Myers Squibb, and Sanofi, and received research grants from Roche. He made these comments as designated discussant for the ADVANCE-ON report and in an interview.
The findings from ADVANCE-ON are important. Clinicians need clear evidence that controlling a patient’s blood pressure produces long-term benefits, and the results from ADVANCE-ON provide this. We need many more studies that track the long-term impact of blood pressure reduction. Currently, a large fraction of patients with diabetes are not maintained at the recommended blood pressure levels. This report presents the longest follow-up of any antihypertensive intervention in patients with diabetes.
The findings also show that a small blood pressure reduction can have an important and long-lasting effect. This highlights the value of blood pressure control, even when the reduction achieved in an individual patient seems small.

|
|
I believe that the effects linked with blood pressure reduction in ADVANCE-ON are likely generalizable to any antihypertensive regimen that produces a similar magnitude of effect. It is the blood pressure effect rather than the specific antihypertensive drugs used that is that critical, although drugs that affect the renin-angiotensin-aldosterone system are clearly the first choice for patients with diabetes and hypertension.
Dr. Lars Rydén is a cardiologist at the Karolinska Institute in Stockholm. He has been an adviser to AstraZeneca, Roche, Bristol-Myers Squibb, and Sanofi, and received research grants from Roche. He made these comments as designated discussant for the ADVANCE-ON report and in an interview.
The findings from ADVANCE-ON are important. Clinicians need clear evidence that controlling a patient’s blood pressure produces long-term benefits, and the results from ADVANCE-ON provide this. We need many more studies that track the long-term impact of blood pressure reduction. Currently, a large fraction of patients with diabetes are not maintained at the recommended blood pressure levels. This report presents the longest follow-up of any antihypertensive intervention in patients with diabetes.
The findings also show that a small blood pressure reduction can have an important and long-lasting effect. This highlights the value of blood pressure control, even when the reduction achieved in an individual patient seems small.

|
|
I believe that the effects linked with blood pressure reduction in ADVANCE-ON are likely generalizable to any antihypertensive regimen that produces a similar magnitude of effect. It is the blood pressure effect rather than the specific antihypertensive drugs used that is that critical, although drugs that affect the renin-angiotensin-aldosterone system are clearly the first choice for patients with diabetes and hypertension.
Dr. Lars Rydén is a cardiologist at the Karolinska Institute in Stockholm. He has been an adviser to AstraZeneca, Roche, Bristol-Myers Squibb, and Sanofi, and received research grants from Roche. He made these comments as designated discussant for the ADVANCE-ON report and in an interview.
BARCELONA – A modest amount of blood pressure control over an average of 4 years produced a significant, long-term survival benefit in patients with type 2 diabetes, based on a 10-year follow-up of more than 8,000 patients.
"It is critically important to maintain active blood-pressure lowering in both the short and long term in order to derive the greatest possible reductions in mortality and major cardiovascular events" in patients with type 2 diabetes, Dr. John Chalmers said at the annual congress of the European Society of Cardiology.
The results he reported showed that during 10 years of total follow-up, 4 years of concerted therapy with a dual antihypertensive-drug formulation on top of background therapy produced an average drop in blood pressure of 5.6/2.2 mm Hg, which was linked to a relative 9% decrease in mortality through the entire 10-year period.
Dr. Chalmers’ analysis used data from an average 6-year follow-up of patients after they completed a 4-year trial that had randomized them to treatment with either the combined formulation of the angiotensin-converting enzyme inhibitor perindopril and the diuretic indapamide, or to placebo. The 6-year assessment of these patients after they finished the closely regulated treatment phase while in the clinical trial is "the longest follow-up on the impact of blood pressure reduction in patients with diabetes," commented Dr. Lars Rydén, a cardiologist at the Karolinska Institute in Stockholm.
"The greatest part of the overall, cumulative benefit [over a 10-year period] was contributed by the carry forward of the in-trial benefits of active BP lowering," said Dr. Chalmers, senior director of the George Institute for Global Health in Sydney, Australia.
His study focused on data collected from patients enrolled in the ADVANCE (Action in Diabetes and Vascular Disease: Preterax and Diamicron-MR Controlled Evaluation) trial. This study randomized 11,140 patients with type 2 diabetes who were at least 55 years old and had at least one additional cardiovascular disease risk factor, to treatment with the combined formulation on top of their preexisting medications or to placebo plus existing medications (Lancet 2007;370:829-40). Patients in the placebo group were unable to receive an angiotensin-converting enzyme inhibitor during the study. The study was sponsored by Servier, the company that markets a combined perindopril and indapamide formulation (Preterax).
At enrollment, ADVANCE patients averaged 66 years old, had an average 8-year history of type 2 diabetes, and had an average blood pressure of 145/81 mm Hg. Three-quarters were on an antihypertensive regimen of some kind at entry into the study.
During the study’s 4 years of active treatment, patients randomized to receive the perindopril and indapamide combination had significantly reduced blood pressure, compared with the control patients. After 4 years, this linked to a 9% relative drop in the combined incidence of major macrovascular and microvascular events (including cardiovascular death, nonfatal stroke, and nonfatal myocardial infarction). Compared with the control group, this cut in events was statistically significant for the study’s primary endpoint. The antihypertensive intervention also was linked to a statistically significant, 14% relative drop in all-cause deaths.
To assess the longer-term impact of this 4-year intervention, Dr. Chalmers and his associates ran a post-trial observational study, ADVANCE-ON. Initially, 8,494 of the patients who completed ADVANCE (83% of surviving patients) agreed to participate in ADVANCE-ON; after another 6 years of follow-up, 5,131 patients remained under observation. During the extended 6 years of follow-up, patients from both the initial intervention arm and the initial control arm all maintained average blood pressures of roughly 137/75 mm Hg. The blood pressures of the original intervention and control patient arms "fully converged," Dr. Chalmers said.
During this 6-year period, patients received care from their personal physicians and could receive any antihypertensive regimen their physician prescribed. At their first follow-up medical examination after the end of the main ADVANCE study, about 40% of patients were on no antihypertensive drugs, 23% were on one drug, 21% were on two drugs, and the remainder were on higher numbers of antihypertensive drugs.
During the 6-year post-trial period, patients who had previously been in the active-treatment arm had a 6% relative reduction in all-cause mortality, compared with patients originally in the control arm, a difference that was not significant.
But when the researchers combined the 6-year post-trial follow-up with the in-trial results for a total median 10-year follow-up, they found an overall 9% relative cut in mortality in the intervention patients, compared with the controls, and a relative 12% drop in cardiovascular mortality.
The persistent, long-term benefits were "attenuated" compared with the shorter-term benefits seen during the active phase, but nonetheless they persisted and were consistent across all subgroups studied, Dr. Chalmers said.
The ADVANCE and ADVANCE-ON studies were partly funded by Servier. Dr. Chalmers has received research grants and honoraria from that company. Dr. Rydén has been an adviser to AstraZeneca, Roche, Bristol-Myers Squibb, and Sanofi, and received research grants from Roche.
On Twitter @mitchelzoler
BARCELONA – A modest amount of blood pressure control over an average of 4 years produced a significant, long-term survival benefit in patients with type 2 diabetes, based on a 10-year follow-up of more than 8,000 patients.
"It is critically important to maintain active blood-pressure lowering in both the short and long term in order to derive the greatest possible reductions in mortality and major cardiovascular events" in patients with type 2 diabetes, Dr. John Chalmers said at the annual congress of the European Society of Cardiology.
The results he reported showed that during 10 years of total follow-up, 4 years of concerted therapy with a dual antihypertensive-drug formulation on top of background therapy produced an average drop in blood pressure of 5.6/2.2 mm Hg, which was linked to a relative 9% decrease in mortality through the entire 10-year period.
Dr. Chalmers’ analysis used data from an average 6-year follow-up of patients after they completed a 4-year trial that had randomized them to treatment with either the combined formulation of the angiotensin-converting enzyme inhibitor perindopril and the diuretic indapamide, or to placebo. The 6-year assessment of these patients after they finished the closely regulated treatment phase while in the clinical trial is "the longest follow-up on the impact of blood pressure reduction in patients with diabetes," commented Dr. Lars Rydén, a cardiologist at the Karolinska Institute in Stockholm.
"The greatest part of the overall, cumulative benefit [over a 10-year period] was contributed by the carry forward of the in-trial benefits of active BP lowering," said Dr. Chalmers, senior director of the George Institute for Global Health in Sydney, Australia.
His study focused on data collected from patients enrolled in the ADVANCE (Action in Diabetes and Vascular Disease: Preterax and Diamicron-MR Controlled Evaluation) trial. This study randomized 11,140 patients with type 2 diabetes who were at least 55 years old and had at least one additional cardiovascular disease risk factor, to treatment with the combined formulation on top of their preexisting medications or to placebo plus existing medications (Lancet 2007;370:829-40). Patients in the placebo group were unable to receive an angiotensin-converting enzyme inhibitor during the study. The study was sponsored by Servier, the company that markets a combined perindopril and indapamide formulation (Preterax).
At enrollment, ADVANCE patients averaged 66 years old, had an average 8-year history of type 2 diabetes, and had an average blood pressure of 145/81 mm Hg. Three-quarters were on an antihypertensive regimen of some kind at entry into the study.
During the study’s 4 years of active treatment, patients randomized to receive the perindopril and indapamide combination had significantly reduced blood pressure, compared with the control patients. After 4 years, this linked to a 9% relative drop in the combined incidence of major macrovascular and microvascular events (including cardiovascular death, nonfatal stroke, and nonfatal myocardial infarction). Compared with the control group, this cut in events was statistically significant for the study’s primary endpoint. The antihypertensive intervention also was linked to a statistically significant, 14% relative drop in all-cause deaths.
To assess the longer-term impact of this 4-year intervention, Dr. Chalmers and his associates ran a post-trial observational study, ADVANCE-ON. Initially, 8,494 of the patients who completed ADVANCE (83% of surviving patients) agreed to participate in ADVANCE-ON; after another 6 years of follow-up, 5,131 patients remained under observation. During the extended 6 years of follow-up, patients from both the initial intervention arm and the initial control arm all maintained average blood pressures of roughly 137/75 mm Hg. The blood pressures of the original intervention and control patient arms "fully converged," Dr. Chalmers said.
During this 6-year period, patients received care from their personal physicians and could receive any antihypertensive regimen their physician prescribed. At their first follow-up medical examination after the end of the main ADVANCE study, about 40% of patients were on no antihypertensive drugs, 23% were on one drug, 21% were on two drugs, and the remainder were on higher numbers of antihypertensive drugs.
During the 6-year post-trial period, patients who had previously been in the active-treatment arm had a 6% relative reduction in all-cause mortality, compared with patients originally in the control arm, a difference that was not significant.
But when the researchers combined the 6-year post-trial follow-up with the in-trial results for a total median 10-year follow-up, they found an overall 9% relative cut in mortality in the intervention patients, compared with the controls, and a relative 12% drop in cardiovascular mortality.
The persistent, long-term benefits were "attenuated" compared with the shorter-term benefits seen during the active phase, but nonetheless they persisted and were consistent across all subgroups studied, Dr. Chalmers said.
The ADVANCE and ADVANCE-ON studies were partly funded by Servier. Dr. Chalmers has received research grants and honoraria from that company. Dr. Rydén has been an adviser to AstraZeneca, Roche, Bristol-Myers Squibb, and Sanofi, and received research grants from Roche.
On Twitter @mitchelzoler
AT THE ESC CONGRESS 2014
Key clinical point: The survival benefit from 4 years of concerted antihypertensive treatment in patients with type 2 diabetes persisted for at least another 6 years.
Major finding: During a median 10-year follow-up, 4 years of active antihypertensive treatment cut mortality by 9%, compared with controls in diabetic patients.
Data source: ADVANCE-ON, a 10-year follow-up of 8,494 patients initially enrolled in ADVANCE, a 4-year, controlled, randomized trial that began with 11,140 patients with type 2 diabetes and at least one other cardiovascular risk factor.
Disclosures:. The ADVANCE and ADVANCE-ON studies were partly funded by Servier. Dr. Chalmers has received research grants and honoraria from that company. Dr. Rydén has been an adviser to AstraZeneca, Roche, Bristol-Myers Squibb, and Sanofi, and received research grants from Roche.