User login
Mitchel is a reporter for MDedge based in the Philadelphia area. He started with the company in 1992, when it was International Medical News Group (IMNG), and has since covered a range of medical specialties. Mitchel trained as a virologist at Roswell Park Memorial Institute in Buffalo, and then worked briefly as a researcher at Boston Children's Hospital before pivoting to journalism as a AAAS Mass Media Fellow in 1980. His first reporting job was with Science Digest magazine, and from the mid-1980s to early-1990s he was a reporter with Medical World News. @mitchelzoler
Registry renal denervation skews from resistant hypertension
WASHINGTON – Renal denervation may have struck out as antihypertensive therapy in the sham-controlled SYMPLICITY HTN-3 trial reported in March at the American College of Cardiology’s annual meeting, but an independent report at the same meeting from a worldwide renal denervation registry showed that the vast majority of patients who’ve undergone with renal denervation recently were nothing like the patients enrolled in the failed trial.
In fact, patients who met enrollment criteria for the SYMPLICITY HTN-3 trial constituted a bare 8% of "real world" patients treated with renal denervation, based on results from the first 1,000 patients enrolled in the Global SYMPLICITY Registry, said Dr. Michael Böhm, professor and chairman of cardiology at Saarland University Hospital in Homburg/Saar, Germany.
Out of the 751 patients of the first 1,000 in the registry with data available on their office systolic blood pressure 6 months after treatment, 62 patients (8%) fulfilled the entry criteria of the SYMPLICITY HTN-3 (Renal Denervation in Patients With Uncontrolled Hypertension) trial by having an office systolic pressure of at least 160 mm Hg, a systolic pressure of at least 135 mm Hg by ambulatory blood pressure monitoring, and on treatment at maximally tolerated dosages with at least three different classes of antihypertensive medications (N. Engl. J. Med. 2014;370:1393-1401).
This low level of patients with clinically confirmed, resistant hypertension occurred against a backdrop in which a scant majority of registry patients even had severe hypertension, let alone a medically uncontrolled form. Among the 751 patients with 6-month follow-up blood pressure data, 433 (58%) received renal denervation with an office systolic blood pressure at baseline of at least 160 mm Hg, said Dr. Böhm.
"SYMPLICITY HTN-3 covered only a few percent of patients" who undergo renal denervation, he said. "The goal of the registry is to include a broad population, including patients without hypertension but other conditions associated with overactivity of the sympathetic nervous system" such as arrhythmia or heart failure.
Among the 433 patients with an office systolic blood pressure of at least 160 mm Hg – the minimum level of hypertension to warrant routine treatment by renal denervation according to several current policies – 244 (56%) were at this high pressure despite treatment with at least three classes of antihypertensive drugs. In this more clearly drug-resistant group, office systolic pressure fell by an average 20.2 mm Hg at 6 months after renal denervation. But only 62 patients on at least three types of drugs were also at maximally tolerated dosages of these drugs, another key element in defining drug resistance. Patients on maximally tolerated dosages represented only a quarter of all patients on multiple drugs in the Registry, and just 14% of the 433 patients with severe hypertension.
Among the 62 patients who matched the SYMPLICITY HTN-3 enrollment criteria, the average drop in office, systolic blood pressure at 6 months after treatment was 17.3 mm Hg, not too different from the average drop of 14.1 mm Hg seen in the renal denervation arm of SYMPLICITY HTN-3, Dr. Böhm noted.
The registry results showed that in patients with an office systolic pressure of less than 160 mm Hg, renal denervation was lousy at blood pressure reduction. Among the 222 patients who had pretreatment systolic pressures of 140-159 mm Hg and 6-month follow-up, office systolic blood pressure dropped by an average of 4.6 mm Hg. The procedure was even less effective in the 96 registry patients with an office systolic pressure of less than 140 mm Hg prior to treatment. In this subgroup systolic pressures averaged a 14.2-mm Hg increase 6 months after treatment.
"The only significant interaction between blood pressure lowering and no lowering was blood pressure at baseline," Dr. Böhm said. He gave no details on how many patients in the registry underwent renal denervation to treat hypertension and how many for other reasons.
The Global SYMPLICITY Register involves more than 200 centers in many European countries as well as in Canada, Australia, Korea, and elsewhere. A large number of registry centers are in Germany, and many centers there and elsewhere participated in the first two SYMPLICITY HTN trials. Participating centers entered the registry based on their experience with renal denervation and had to have performed at least 30 procedures prior to entry into the registry. The 6-month data also showed that renal denervation was "very safe," with "very low" rates of adverse events, Dr. Böhm said.
The Global SYMPLICITY Registry is sponsored by Medtronic, the company that markets Symplicity renal denervation devices. Dr, Böhm said that he has served on an advisory board to, been a speaker for, and received research support from Medtronic as well as from several other drug and device companies.
On Twitter @mitchelzoler
WASHINGTON – Renal denervation may have struck out as antihypertensive therapy in the sham-controlled SYMPLICITY HTN-3 trial reported in March at the American College of Cardiology’s annual meeting, but an independent report at the same meeting from a worldwide renal denervation registry showed that the vast majority of patients who’ve undergone with renal denervation recently were nothing like the patients enrolled in the failed trial.
In fact, patients who met enrollment criteria for the SYMPLICITY HTN-3 trial constituted a bare 8% of "real world" patients treated with renal denervation, based on results from the first 1,000 patients enrolled in the Global SYMPLICITY Registry, said Dr. Michael Böhm, professor and chairman of cardiology at Saarland University Hospital in Homburg/Saar, Germany.
Out of the 751 patients of the first 1,000 in the registry with data available on their office systolic blood pressure 6 months after treatment, 62 patients (8%) fulfilled the entry criteria of the SYMPLICITY HTN-3 (Renal Denervation in Patients With Uncontrolled Hypertension) trial by having an office systolic pressure of at least 160 mm Hg, a systolic pressure of at least 135 mm Hg by ambulatory blood pressure monitoring, and on treatment at maximally tolerated dosages with at least three different classes of antihypertensive medications (N. Engl. J. Med. 2014;370:1393-1401).
This low level of patients with clinically confirmed, resistant hypertension occurred against a backdrop in which a scant majority of registry patients even had severe hypertension, let alone a medically uncontrolled form. Among the 751 patients with 6-month follow-up blood pressure data, 433 (58%) received renal denervation with an office systolic blood pressure at baseline of at least 160 mm Hg, said Dr. Böhm.
"SYMPLICITY HTN-3 covered only a few percent of patients" who undergo renal denervation, he said. "The goal of the registry is to include a broad population, including patients without hypertension but other conditions associated with overactivity of the sympathetic nervous system" such as arrhythmia or heart failure.
Among the 433 patients with an office systolic blood pressure of at least 160 mm Hg – the minimum level of hypertension to warrant routine treatment by renal denervation according to several current policies – 244 (56%) were at this high pressure despite treatment with at least three classes of antihypertensive drugs. In this more clearly drug-resistant group, office systolic pressure fell by an average 20.2 mm Hg at 6 months after renal denervation. But only 62 patients on at least three types of drugs were also at maximally tolerated dosages of these drugs, another key element in defining drug resistance. Patients on maximally tolerated dosages represented only a quarter of all patients on multiple drugs in the Registry, and just 14% of the 433 patients with severe hypertension.
Among the 62 patients who matched the SYMPLICITY HTN-3 enrollment criteria, the average drop in office, systolic blood pressure at 6 months after treatment was 17.3 mm Hg, not too different from the average drop of 14.1 mm Hg seen in the renal denervation arm of SYMPLICITY HTN-3, Dr. Böhm noted.
The registry results showed that in patients with an office systolic pressure of less than 160 mm Hg, renal denervation was lousy at blood pressure reduction. Among the 222 patients who had pretreatment systolic pressures of 140-159 mm Hg and 6-month follow-up, office systolic blood pressure dropped by an average of 4.6 mm Hg. The procedure was even less effective in the 96 registry patients with an office systolic pressure of less than 140 mm Hg prior to treatment. In this subgroup systolic pressures averaged a 14.2-mm Hg increase 6 months after treatment.
"The only significant interaction between blood pressure lowering and no lowering was blood pressure at baseline," Dr. Böhm said. He gave no details on how many patients in the registry underwent renal denervation to treat hypertension and how many for other reasons.
The Global SYMPLICITY Register involves more than 200 centers in many European countries as well as in Canada, Australia, Korea, and elsewhere. A large number of registry centers are in Germany, and many centers there and elsewhere participated in the first two SYMPLICITY HTN trials. Participating centers entered the registry based on their experience with renal denervation and had to have performed at least 30 procedures prior to entry into the registry. The 6-month data also showed that renal denervation was "very safe," with "very low" rates of adverse events, Dr. Böhm said.
The Global SYMPLICITY Registry is sponsored by Medtronic, the company that markets Symplicity renal denervation devices. Dr, Böhm said that he has served on an advisory board to, been a speaker for, and received research support from Medtronic as well as from several other drug and device companies.
On Twitter @mitchelzoler
WASHINGTON – Renal denervation may have struck out as antihypertensive therapy in the sham-controlled SYMPLICITY HTN-3 trial reported in March at the American College of Cardiology’s annual meeting, but an independent report at the same meeting from a worldwide renal denervation registry showed that the vast majority of patients who’ve undergone with renal denervation recently were nothing like the patients enrolled in the failed trial.
In fact, patients who met enrollment criteria for the SYMPLICITY HTN-3 trial constituted a bare 8% of "real world" patients treated with renal denervation, based on results from the first 1,000 patients enrolled in the Global SYMPLICITY Registry, said Dr. Michael Böhm, professor and chairman of cardiology at Saarland University Hospital in Homburg/Saar, Germany.
Out of the 751 patients of the first 1,000 in the registry with data available on their office systolic blood pressure 6 months after treatment, 62 patients (8%) fulfilled the entry criteria of the SYMPLICITY HTN-3 (Renal Denervation in Patients With Uncontrolled Hypertension) trial by having an office systolic pressure of at least 160 mm Hg, a systolic pressure of at least 135 mm Hg by ambulatory blood pressure monitoring, and on treatment at maximally tolerated dosages with at least three different classes of antihypertensive medications (N. Engl. J. Med. 2014;370:1393-1401).
This low level of patients with clinically confirmed, resistant hypertension occurred against a backdrop in which a scant majority of registry patients even had severe hypertension, let alone a medically uncontrolled form. Among the 751 patients with 6-month follow-up blood pressure data, 433 (58%) received renal denervation with an office systolic blood pressure at baseline of at least 160 mm Hg, said Dr. Böhm.
"SYMPLICITY HTN-3 covered only a few percent of patients" who undergo renal denervation, he said. "The goal of the registry is to include a broad population, including patients without hypertension but other conditions associated with overactivity of the sympathetic nervous system" such as arrhythmia or heart failure.
Among the 433 patients with an office systolic blood pressure of at least 160 mm Hg – the minimum level of hypertension to warrant routine treatment by renal denervation according to several current policies – 244 (56%) were at this high pressure despite treatment with at least three classes of antihypertensive drugs. In this more clearly drug-resistant group, office systolic pressure fell by an average 20.2 mm Hg at 6 months after renal denervation. But only 62 patients on at least three types of drugs were also at maximally tolerated dosages of these drugs, another key element in defining drug resistance. Patients on maximally tolerated dosages represented only a quarter of all patients on multiple drugs in the Registry, and just 14% of the 433 patients with severe hypertension.
Among the 62 patients who matched the SYMPLICITY HTN-3 enrollment criteria, the average drop in office, systolic blood pressure at 6 months after treatment was 17.3 mm Hg, not too different from the average drop of 14.1 mm Hg seen in the renal denervation arm of SYMPLICITY HTN-3, Dr. Böhm noted.
The registry results showed that in patients with an office systolic pressure of less than 160 mm Hg, renal denervation was lousy at blood pressure reduction. Among the 222 patients who had pretreatment systolic pressures of 140-159 mm Hg and 6-month follow-up, office systolic blood pressure dropped by an average of 4.6 mm Hg. The procedure was even less effective in the 96 registry patients with an office systolic pressure of less than 140 mm Hg prior to treatment. In this subgroup systolic pressures averaged a 14.2-mm Hg increase 6 months after treatment.
"The only significant interaction between blood pressure lowering and no lowering was blood pressure at baseline," Dr. Böhm said. He gave no details on how many patients in the registry underwent renal denervation to treat hypertension and how many for other reasons.
The Global SYMPLICITY Register involves more than 200 centers in many European countries as well as in Canada, Australia, Korea, and elsewhere. A large number of registry centers are in Germany, and many centers there and elsewhere participated in the first two SYMPLICITY HTN trials. Participating centers entered the registry based on their experience with renal denervation and had to have performed at least 30 procedures prior to entry into the registry. The 6-month data also showed that renal denervation was "very safe," with "very low" rates of adverse events, Dr. Böhm said.
The Global SYMPLICITY Registry is sponsored by Medtronic, the company that markets Symplicity renal denervation devices. Dr, Böhm said that he has served on an advisory board to, been a speaker for, and received research support from Medtronic as well as from several other drug and device companies.
On Twitter @mitchelzoler
AT ACC 2014
Key clinical point: Real-world use of renal denervation has not targeted patients with drug-resistant hypertension.
Major finding: Only 8% of patient in the Global SYMPLICITY Registry for renal denervation matched the patients enrolled in the SYMPLICITY HTN-3 trial.
Data source: The Global SYMPLICITY Registry, which enrolled 1,000 patients who underwent renal denervation at more than 200 centers worldwide.
Disclosures: The registry is sponsored by Medtronic, the company that markets Symplicity renal denervation devices. Dr, Böhm said that he has served on an advisory board to, been a speaker for, and received research support from Medtronic as well as from several other drug and device companies.
New Scourge Poses Old ID Challenge
There are hardly any infectious diseases that a little soap and water can’t help.
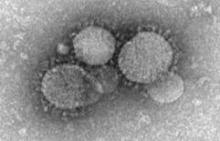
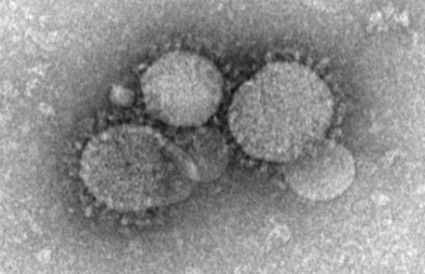
That old chestnut resurfaced when Dr. Keiji Fukuda of the World Health Organization announced on May 14 that while a special WHO panel had determined that the worldwide spread of Middle East Respiratory Syndrome Coronavirus (MERS-CoV) did not yet constitute an international public health emergency, its spread right now seems to be driven largely by poor infection-control measures.
"Hospital outbreaks have been a key feature of this virus," which has produced nearly 600 confirmed cases worldwide, mostly in Saudi Arabia and the United Arab Emirates, but also with at least two confirmed cases in the United States, Dr. Fukuda said.
The recent uptick in cases – half of all cases to date were diagnosed in April – as well as their spread to new countries led the WHO special panel on MERS-CoV to meet May 13-14.
The panel concluded that no emergency exists yet. And they urged hospitals to put a much greater emphasis on the principles of Infection Fighting 101: handwashing, using masks and gloves, and changing gloves between patients.
In late April and early May, WHO staffers inspected several Saudi Arabian hospitals where MERS-CoV clusters had appeared and found "infection control practices were not up to standards," and that issues such as overcrowded emergency departments "amplified" the spread of MERS-CoV, Dr. Fukuda, WHO assistant director general, said during a press conference in Geneva.
MERS-CoV cases in the community also increased in recent weeks, which may reflect a seasonal rise in infections, improved surveillance and case recognition, or increasing person-to-person transmission. But Dr. Fukuda stressed that for now there is "no convincing evidence" of any change in the transmissibility of MERS-CoV or of its genetic makeup, and no evidence that MERS-CoV is cutting infectious swaths through communities.
The WHO committee put all the MERS-CoV evidence "on the table to see how it adds up," to produce "a very sober and critical assessment," a process Dr. Fukuda said could not have happened even 10 years ago.
On Twitter @mitchelzoler
There are hardly any infectious diseases that a little soap and water can’t help.
That old chestnut resurfaced when Dr. Keiji Fukuda of the World Health Organization announced on May 14 that while a special WHO panel had determined that the worldwide spread of Middle East Respiratory Syndrome Coronavirus (MERS-CoV) did not yet constitute an international public health emergency, its spread right now seems to be driven largely by poor infection-control measures.
"Hospital outbreaks have been a key feature of this virus," which has produced nearly 600 confirmed cases worldwide, mostly in Saudi Arabia and the United Arab Emirates, but also with at least two confirmed cases in the United States, Dr. Fukuda said.
The recent uptick in cases – half of all cases to date were diagnosed in April – as well as their spread to new countries led the WHO special panel on MERS-CoV to meet May 13-14.
The panel concluded that no emergency exists yet. And they urged hospitals to put a much greater emphasis on the principles of Infection Fighting 101: handwashing, using masks and gloves, and changing gloves between patients.
In late April and early May, WHO staffers inspected several Saudi Arabian hospitals where MERS-CoV clusters had appeared and found "infection control practices were not up to standards," and that issues such as overcrowded emergency departments "amplified" the spread of MERS-CoV, Dr. Fukuda, WHO assistant director general, said during a press conference in Geneva.
MERS-CoV cases in the community also increased in recent weeks, which may reflect a seasonal rise in infections, improved surveillance and case recognition, or increasing person-to-person transmission. But Dr. Fukuda stressed that for now there is "no convincing evidence" of any change in the transmissibility of MERS-CoV or of its genetic makeup, and no evidence that MERS-CoV is cutting infectious swaths through communities.
The WHO committee put all the MERS-CoV evidence "on the table to see how it adds up," to produce "a very sober and critical assessment," a process Dr. Fukuda said could not have happened even 10 years ago.
On Twitter @mitchelzoler
There are hardly any infectious diseases that a little soap and water can’t help.
That old chestnut resurfaced when Dr. Keiji Fukuda of the World Health Organization announced on May 14 that while a special WHO panel had determined that the worldwide spread of Middle East Respiratory Syndrome Coronavirus (MERS-CoV) did not yet constitute an international public health emergency, its spread right now seems to be driven largely by poor infection-control measures.
"Hospital outbreaks have been a key feature of this virus," which has produced nearly 600 confirmed cases worldwide, mostly in Saudi Arabia and the United Arab Emirates, but also with at least two confirmed cases in the United States, Dr. Fukuda said.
The recent uptick in cases – half of all cases to date were diagnosed in April – as well as their spread to new countries led the WHO special panel on MERS-CoV to meet May 13-14.
The panel concluded that no emergency exists yet. And they urged hospitals to put a much greater emphasis on the principles of Infection Fighting 101: handwashing, using masks and gloves, and changing gloves between patients.
In late April and early May, WHO staffers inspected several Saudi Arabian hospitals where MERS-CoV clusters had appeared and found "infection control practices were not up to standards," and that issues such as overcrowded emergency departments "amplified" the spread of MERS-CoV, Dr. Fukuda, WHO assistant director general, said during a press conference in Geneva.
MERS-CoV cases in the community also increased in recent weeks, which may reflect a seasonal rise in infections, improved surveillance and case recognition, or increasing person-to-person transmission. But Dr. Fukuda stressed that for now there is "no convincing evidence" of any change in the transmissibility of MERS-CoV or of its genetic makeup, and no evidence that MERS-CoV is cutting infectious swaths through communities.
The WHO committee put all the MERS-CoV evidence "on the table to see how it adds up," to produce "a very sober and critical assessment," a process Dr. Fukuda said could not have happened even 10 years ago.
On Twitter @mitchelzoler
TAVR quickly penetrates to low-risk patients
Have too many low-risk U.S. patients undergone transcatheter aortic-valve replacement since the procedure became available in October 2012 to U.S. patients who are also judged eligible for surgical aortic-valve replacement?
Furthermore, regardless of the answer, are the tools currently available to cardiologists and cardiac surgeons to estimate a patient’s risk for undergoing aortic-valve surgery too limited and flawed to even allow clinicians to reasonably judge who is at high risk for surgical valve replacement and who isn’t?
And finally, have the benefits of transcatheter aortic valve replacement (TAVR) as an alternative to surgery become so compelling that patients, cardiologists, and surgeons are all now willing to ignore the possible downside that still remains to TAVR and the risk-level ground rules that the field set up just a few years ago?
Answering the third question is probably the easiest, and the answer seems to be yes, at least based on U.S. use of TAVR since the first valve system came onto the U.S. market for inoperable patients in November 2011, as well as on what happened in the latest big TAVR trial. Last November, researchers published a report on the first 7,710 U.S. TAVR patients, while results from the latest big trial, the CoreValve pivotal trial, came out in March.
A key finding in the JAMA report last November on nearly 8,000 TAVR recipients, most of whom were operable patients once this indication received U.S. approval in 2012, was that the median Predicted Risk of Operative Mortality score by the formula crafted by the Society of Thoracic Surgeons (the STS PROM score) was 7%, and a quarter of all U.S. patients had a score of 5% or less. Those risk levels are quite low relative to the levels in the first TAVR pivotal trial, the PARTNER I trial, and relative to how TAVR developers viewed the role for this technology when it first entered the U.S. market a couple of years ago.
In the first U.S. pivotal trial for TAVR in patients judged operable, a head-to-head comparison of TAVR and surgical aortic valve replacement (SAVR), all enrolled patients had to have an STS PROM score of at least 10%, and the average score of enrolled patients was 11.8%. Labeling for the first U.S. approved TAVR system was for patients with an STS PROM score of at least 8%. The follow-up trial designed to test a second-generation TAVR device, PARTNER II, launched about 3 years ago and not scheduled to finish until the end of 2015, specifically targeted "intermediate-risk" patients with aortic stenosis, those with an STS PROM of 4%-8%. Even this next-generation-device trial, PARTNER II, wasn’t designed to target patients with risk levels of less than 4%, yet patients of that very sort have already received treatment with the first-generation device based on the registry results.
It’s not just the registry that shows a shift toward lower-risk patients. The CoreValve pivotal trial that pitted a different TAVR system head to head against SAVR showed more of the same. The trial was designed to enroll operable patients with a predicted 30-day mortality risk after SAVR of at least 15%, though the study left it up to clinicians to decide how to measure risk and gave them free rein to use parameters in addition to the STS PROM score. The result was that the average STS PROM score of enrolled patients in the CoreValve trial was 7%, and roughly 10% of enrolled patients had a score of less than 4%. The temptation to use TAVR on lower-risk patients seems to have been inescapable, happening in both the CoreValve trial as well as in the registry’s Sapien experience.
Of course, the CoreValve results also showed significant survival benefit from TAVR using the CoreValve system, a game-changing result.
Part of what has been going on with risk assessment is that in the CoreValve study as well as in routine practice, clinicians have been fudging their use of the STS PROM score when sizing up patients for TAVR. I asked several interventional cardiologists about this at the ACC meeting in March, and their answers were all variants of what Dr. James Hermiller told me: "It’s frailty that often gets a patient to TAVR, and frailty is hard to quantify. Frailty can exist even when the STS score is not high." And even though labeling for the first-generation TAVR system that all 7,710 of the first U.S. patients received specified an STS PROM score of at least 8%, Dr. David Holmes told me that for Medicare reimbursement, all that’s needed is for two cardiac surgeons to sign off on saying that the patient’s status warrants TAVR. "That’s what carries the day," he said.
Data from the new CoreValve study underscore how limited the STS PROM score is right now. The average score of the patients enrolled in the surgical arm of this study was 7.5%, which means that 7.5 % of the patients who underwent SAVR were predicted by the scoring system to die during the first 30 days after surgery. But the actual rate was 4.5%, "substantially lower," said the CoreValve report. STS PROM scoring resulted in a substantial overcall on predicted risk.
When TAVR was first introduced, experts had two caveats about its potential to completely replace SAVR. The first was uncertainty about the long-term durability (think 10 or more years) of TAVR. The second was uncertainty about the short- and intermediate-term safety and efficacy of TAVR, especially for the patients for whom conventional SAVR was a reasonable option.
Doubts about short- and intermediate-term efficacy arose with the first-generation TAVR device, Sapien, because of the issue of paravalvular leak and the inability of TAVR to surpass SAVR outcomes in the PARTNER I results, but those doubts have now been mostly swept away from by the CoreValve results, which established CoreValve as superior to SAVR and made it the current standard for essentially all patients who need their aortic valve replaced. Even if paravalvular leak is still an issue for some patients, patients treated with CoreValve, TAVR overall did significantly better after 1 year than SAVR in the CoreValve trial, which means that TAVR was best regardless of whether paravalvular leak was an issue for some patients. And this was in patients who represented a wide range of STS PROM risk, with close to 10% of enrolled patients having a score of less than 4%. A subanalysis showed that the low-risk patients derived as much benefit from CoreValve TAVR, compared with SAVR, as did higher-risk patients.
The long-term durability question still remains for now, but the substantial mortality benefit in the CoreValve trial seen after 1 year probably trumps that.
Researchers designed the TAVR trials to methodically progress through a spectrum of patient risk levels. As recently as a year ago, several experts told me that no way in the near future could TAVR be an option for low-risk patients with STS PROM scores of less than 4%. But that is not how it has worked out. Patients, cardiac surgeons, and cardiologists embraced TAVR way faster and tighter than anyone expected just a few years ago.
On Twitter @mitchelzoler
Have too many low-risk U.S. patients undergone transcatheter aortic-valve replacement since the procedure became available in October 2012 to U.S. patients who are also judged eligible for surgical aortic-valve replacement?
Furthermore, regardless of the answer, are the tools currently available to cardiologists and cardiac surgeons to estimate a patient’s risk for undergoing aortic-valve surgery too limited and flawed to even allow clinicians to reasonably judge who is at high risk for surgical valve replacement and who isn’t?
And finally, have the benefits of transcatheter aortic valve replacement (TAVR) as an alternative to surgery become so compelling that patients, cardiologists, and surgeons are all now willing to ignore the possible downside that still remains to TAVR and the risk-level ground rules that the field set up just a few years ago?
Answering the third question is probably the easiest, and the answer seems to be yes, at least based on U.S. use of TAVR since the first valve system came onto the U.S. market for inoperable patients in November 2011, as well as on what happened in the latest big TAVR trial. Last November, researchers published a report on the first 7,710 U.S. TAVR patients, while results from the latest big trial, the CoreValve pivotal trial, came out in March.
A key finding in the JAMA report last November on nearly 8,000 TAVR recipients, most of whom were operable patients once this indication received U.S. approval in 2012, was that the median Predicted Risk of Operative Mortality score by the formula crafted by the Society of Thoracic Surgeons (the STS PROM score) was 7%, and a quarter of all U.S. patients had a score of 5% or less. Those risk levels are quite low relative to the levels in the first TAVR pivotal trial, the PARTNER I trial, and relative to how TAVR developers viewed the role for this technology when it first entered the U.S. market a couple of years ago.
In the first U.S. pivotal trial for TAVR in patients judged operable, a head-to-head comparison of TAVR and surgical aortic valve replacement (SAVR), all enrolled patients had to have an STS PROM score of at least 10%, and the average score of enrolled patients was 11.8%. Labeling for the first U.S. approved TAVR system was for patients with an STS PROM score of at least 8%. The follow-up trial designed to test a second-generation TAVR device, PARTNER II, launched about 3 years ago and not scheduled to finish until the end of 2015, specifically targeted "intermediate-risk" patients with aortic stenosis, those with an STS PROM of 4%-8%. Even this next-generation-device trial, PARTNER II, wasn’t designed to target patients with risk levels of less than 4%, yet patients of that very sort have already received treatment with the first-generation device based on the registry results.
It’s not just the registry that shows a shift toward lower-risk patients. The CoreValve pivotal trial that pitted a different TAVR system head to head against SAVR showed more of the same. The trial was designed to enroll operable patients with a predicted 30-day mortality risk after SAVR of at least 15%, though the study left it up to clinicians to decide how to measure risk and gave them free rein to use parameters in addition to the STS PROM score. The result was that the average STS PROM score of enrolled patients in the CoreValve trial was 7%, and roughly 10% of enrolled patients had a score of less than 4%. The temptation to use TAVR on lower-risk patients seems to have been inescapable, happening in both the CoreValve trial as well as in the registry’s Sapien experience.
Of course, the CoreValve results also showed significant survival benefit from TAVR using the CoreValve system, a game-changing result.
Part of what has been going on with risk assessment is that in the CoreValve study as well as in routine practice, clinicians have been fudging their use of the STS PROM score when sizing up patients for TAVR. I asked several interventional cardiologists about this at the ACC meeting in March, and their answers were all variants of what Dr. James Hermiller told me: "It’s frailty that often gets a patient to TAVR, and frailty is hard to quantify. Frailty can exist even when the STS score is not high." And even though labeling for the first-generation TAVR system that all 7,710 of the first U.S. patients received specified an STS PROM score of at least 8%, Dr. David Holmes told me that for Medicare reimbursement, all that’s needed is for two cardiac surgeons to sign off on saying that the patient’s status warrants TAVR. "That’s what carries the day," he said.
Data from the new CoreValve study underscore how limited the STS PROM score is right now. The average score of the patients enrolled in the surgical arm of this study was 7.5%, which means that 7.5 % of the patients who underwent SAVR were predicted by the scoring system to die during the first 30 days after surgery. But the actual rate was 4.5%, "substantially lower," said the CoreValve report. STS PROM scoring resulted in a substantial overcall on predicted risk.
When TAVR was first introduced, experts had two caveats about its potential to completely replace SAVR. The first was uncertainty about the long-term durability (think 10 or more years) of TAVR. The second was uncertainty about the short- and intermediate-term safety and efficacy of TAVR, especially for the patients for whom conventional SAVR was a reasonable option.
Doubts about short- and intermediate-term efficacy arose with the first-generation TAVR device, Sapien, because of the issue of paravalvular leak and the inability of TAVR to surpass SAVR outcomes in the PARTNER I results, but those doubts have now been mostly swept away from by the CoreValve results, which established CoreValve as superior to SAVR and made it the current standard for essentially all patients who need their aortic valve replaced. Even if paravalvular leak is still an issue for some patients, patients treated with CoreValve, TAVR overall did significantly better after 1 year than SAVR in the CoreValve trial, which means that TAVR was best regardless of whether paravalvular leak was an issue for some patients. And this was in patients who represented a wide range of STS PROM risk, with close to 10% of enrolled patients having a score of less than 4%. A subanalysis showed that the low-risk patients derived as much benefit from CoreValve TAVR, compared with SAVR, as did higher-risk patients.
The long-term durability question still remains for now, but the substantial mortality benefit in the CoreValve trial seen after 1 year probably trumps that.
Researchers designed the TAVR trials to methodically progress through a spectrum of patient risk levels. As recently as a year ago, several experts told me that no way in the near future could TAVR be an option for low-risk patients with STS PROM scores of less than 4%. But that is not how it has worked out. Patients, cardiac surgeons, and cardiologists embraced TAVR way faster and tighter than anyone expected just a few years ago.
On Twitter @mitchelzoler
Have too many low-risk U.S. patients undergone transcatheter aortic-valve replacement since the procedure became available in October 2012 to U.S. patients who are also judged eligible for surgical aortic-valve replacement?
Furthermore, regardless of the answer, are the tools currently available to cardiologists and cardiac surgeons to estimate a patient’s risk for undergoing aortic-valve surgery too limited and flawed to even allow clinicians to reasonably judge who is at high risk for surgical valve replacement and who isn’t?
And finally, have the benefits of transcatheter aortic valve replacement (TAVR) as an alternative to surgery become so compelling that patients, cardiologists, and surgeons are all now willing to ignore the possible downside that still remains to TAVR and the risk-level ground rules that the field set up just a few years ago?
Answering the third question is probably the easiest, and the answer seems to be yes, at least based on U.S. use of TAVR since the first valve system came onto the U.S. market for inoperable patients in November 2011, as well as on what happened in the latest big TAVR trial. Last November, researchers published a report on the first 7,710 U.S. TAVR patients, while results from the latest big trial, the CoreValve pivotal trial, came out in March.
A key finding in the JAMA report last November on nearly 8,000 TAVR recipients, most of whom were operable patients once this indication received U.S. approval in 2012, was that the median Predicted Risk of Operative Mortality score by the formula crafted by the Society of Thoracic Surgeons (the STS PROM score) was 7%, and a quarter of all U.S. patients had a score of 5% or less. Those risk levels are quite low relative to the levels in the first TAVR pivotal trial, the PARTNER I trial, and relative to how TAVR developers viewed the role for this technology when it first entered the U.S. market a couple of years ago.
In the first U.S. pivotal trial for TAVR in patients judged operable, a head-to-head comparison of TAVR and surgical aortic valve replacement (SAVR), all enrolled patients had to have an STS PROM score of at least 10%, and the average score of enrolled patients was 11.8%. Labeling for the first U.S. approved TAVR system was for patients with an STS PROM score of at least 8%. The follow-up trial designed to test a second-generation TAVR device, PARTNER II, launched about 3 years ago and not scheduled to finish until the end of 2015, specifically targeted "intermediate-risk" patients with aortic stenosis, those with an STS PROM of 4%-8%. Even this next-generation-device trial, PARTNER II, wasn’t designed to target patients with risk levels of less than 4%, yet patients of that very sort have already received treatment with the first-generation device based on the registry results.
It’s not just the registry that shows a shift toward lower-risk patients. The CoreValve pivotal trial that pitted a different TAVR system head to head against SAVR showed more of the same. The trial was designed to enroll operable patients with a predicted 30-day mortality risk after SAVR of at least 15%, though the study left it up to clinicians to decide how to measure risk and gave them free rein to use parameters in addition to the STS PROM score. The result was that the average STS PROM score of enrolled patients in the CoreValve trial was 7%, and roughly 10% of enrolled patients had a score of less than 4%. The temptation to use TAVR on lower-risk patients seems to have been inescapable, happening in both the CoreValve trial as well as in the registry’s Sapien experience.
Of course, the CoreValve results also showed significant survival benefit from TAVR using the CoreValve system, a game-changing result.
Part of what has been going on with risk assessment is that in the CoreValve study as well as in routine practice, clinicians have been fudging their use of the STS PROM score when sizing up patients for TAVR. I asked several interventional cardiologists about this at the ACC meeting in March, and their answers were all variants of what Dr. James Hermiller told me: "It’s frailty that often gets a patient to TAVR, and frailty is hard to quantify. Frailty can exist even when the STS score is not high." And even though labeling for the first-generation TAVR system that all 7,710 of the first U.S. patients received specified an STS PROM score of at least 8%, Dr. David Holmes told me that for Medicare reimbursement, all that’s needed is for two cardiac surgeons to sign off on saying that the patient’s status warrants TAVR. "That’s what carries the day," he said.
Data from the new CoreValve study underscore how limited the STS PROM score is right now. The average score of the patients enrolled in the surgical arm of this study was 7.5%, which means that 7.5 % of the patients who underwent SAVR were predicted by the scoring system to die during the first 30 days after surgery. But the actual rate was 4.5%, "substantially lower," said the CoreValve report. STS PROM scoring resulted in a substantial overcall on predicted risk.
When TAVR was first introduced, experts had two caveats about its potential to completely replace SAVR. The first was uncertainty about the long-term durability (think 10 or more years) of TAVR. The second was uncertainty about the short- and intermediate-term safety and efficacy of TAVR, especially for the patients for whom conventional SAVR was a reasonable option.
Doubts about short- and intermediate-term efficacy arose with the first-generation TAVR device, Sapien, because of the issue of paravalvular leak and the inability of TAVR to surpass SAVR outcomes in the PARTNER I results, but those doubts have now been mostly swept away from by the CoreValve results, which established CoreValve as superior to SAVR and made it the current standard for essentially all patients who need their aortic valve replaced. Even if paravalvular leak is still an issue for some patients, patients treated with CoreValve, TAVR overall did significantly better after 1 year than SAVR in the CoreValve trial, which means that TAVR was best regardless of whether paravalvular leak was an issue for some patients. And this was in patients who represented a wide range of STS PROM risk, with close to 10% of enrolled patients having a score of less than 4%. A subanalysis showed that the low-risk patients derived as much benefit from CoreValve TAVR, compared with SAVR, as did higher-risk patients.
The long-term durability question still remains for now, but the substantial mortality benefit in the CoreValve trial seen after 1 year probably trumps that.
Researchers designed the TAVR trials to methodically progress through a spectrum of patient risk levels. As recently as a year ago, several experts told me that no way in the near future could TAVR be an option for low-risk patients with STS PROM scores of less than 4%. But that is not how it has worked out. Patients, cardiac surgeons, and cardiologists embraced TAVR way faster and tighter than anyone expected just a few years ago.
On Twitter @mitchelzoler
MERS: New scourge poses old ID challenge
There are hardly any infectious diseases that a little soap and water can’t help.
That old chestnut resurfaced when Dr. Keiji Fukuda of the World Health Organization announced on May 14 that while a special WHO panel had determined that the worldwide spread of Middle East Respiratory Syndrome Coronavirus (MERS-CoV) did not yet constitute an international public health emergency, its spread right now seems to be driven largely by poor infection-control measures.
"Hospital outbreaks have been a key feature of this virus," which has produced nearly 600 confirmed cases worldwide, mostly in Saudi Arabia and the United Arab Emirates, but also with at least two confirmed cases in the United States, Dr. Fukuda said.
The recent uptick in cases – half of all cases to date were diagnosed in April – as well as their spread to new countries led the WHO special panel on MERS-CoV to meet May 13-14.
The panel concluded that no emergency exists yet. And they urged hospitals to put a much greater emphasis on the principles of Infection Fighting 101: handwashing, using masks and gloves, and changing gloves between patients.
In late April and early May, WHO staffers inspected several Saudi Arabian hospitals where MERS-CoV clusters had appeared and found "infection control practices were not up to standards," and that issues such as overcrowded emergency departments "amplified" the spread of MERS-CoV, Dr. Fukuda, WHO assistant director general, said during a press conference in Geneva.
MERS-CoV cases in the community also increased in recent weeks, which may reflect a seasonal rise in infections, improved surveillance and case recognition, or increasing person-to-person transmission. But Dr. Fukuda stressed that for now there is "no convincing evidence" of any change in the transmissibility of MERS-CoV or of its genetic makeup, and no evidence that MERS-CoV is cutting infectious swaths through communities.
The WHO committee put all the MERS-CoV evidence "on the table to see how it adds up," to produce "a very sober and critical assessment," a process Dr. Fukuda said could not have happened even 10 years ago.
On Twitter @mitchelzoler
There are hardly any infectious diseases that a little soap and water can’t help.
That old chestnut resurfaced when Dr. Keiji Fukuda of the World Health Organization announced on May 14 that while a special WHO panel had determined that the worldwide spread of Middle East Respiratory Syndrome Coronavirus (MERS-CoV) did not yet constitute an international public health emergency, its spread right now seems to be driven largely by poor infection-control measures.
"Hospital outbreaks have been a key feature of this virus," which has produced nearly 600 confirmed cases worldwide, mostly in Saudi Arabia and the United Arab Emirates, but also with at least two confirmed cases in the United States, Dr. Fukuda said.
The recent uptick in cases – half of all cases to date were diagnosed in April – as well as their spread to new countries led the WHO special panel on MERS-CoV to meet May 13-14.
The panel concluded that no emergency exists yet. And they urged hospitals to put a much greater emphasis on the principles of Infection Fighting 101: handwashing, using masks and gloves, and changing gloves between patients.
In late April and early May, WHO staffers inspected several Saudi Arabian hospitals where MERS-CoV clusters had appeared and found "infection control practices were not up to standards," and that issues such as overcrowded emergency departments "amplified" the spread of MERS-CoV, Dr. Fukuda, WHO assistant director general, said during a press conference in Geneva.
MERS-CoV cases in the community also increased in recent weeks, which may reflect a seasonal rise in infections, improved surveillance and case recognition, or increasing person-to-person transmission. But Dr. Fukuda stressed that for now there is "no convincing evidence" of any change in the transmissibility of MERS-CoV or of its genetic makeup, and no evidence that MERS-CoV is cutting infectious swaths through communities.
The WHO committee put all the MERS-CoV evidence "on the table to see how it adds up," to produce "a very sober and critical assessment," a process Dr. Fukuda said could not have happened even 10 years ago.
On Twitter @mitchelzoler
There are hardly any infectious diseases that a little soap and water can’t help.
That old chestnut resurfaced when Dr. Keiji Fukuda of the World Health Organization announced on May 14 that while a special WHO panel had determined that the worldwide spread of Middle East Respiratory Syndrome Coronavirus (MERS-CoV) did not yet constitute an international public health emergency, its spread right now seems to be driven largely by poor infection-control measures.
"Hospital outbreaks have been a key feature of this virus," which has produced nearly 600 confirmed cases worldwide, mostly in Saudi Arabia and the United Arab Emirates, but also with at least two confirmed cases in the United States, Dr. Fukuda said.
The recent uptick in cases – half of all cases to date were diagnosed in April – as well as their spread to new countries led the WHO special panel on MERS-CoV to meet May 13-14.
The panel concluded that no emergency exists yet. And they urged hospitals to put a much greater emphasis on the principles of Infection Fighting 101: handwashing, using masks and gloves, and changing gloves between patients.
In late April and early May, WHO staffers inspected several Saudi Arabian hospitals where MERS-CoV clusters had appeared and found "infection control practices were not up to standards," and that issues such as overcrowded emergency departments "amplified" the spread of MERS-CoV, Dr. Fukuda, WHO assistant director general, said during a press conference in Geneva.
MERS-CoV cases in the community also increased in recent weeks, which may reflect a seasonal rise in infections, improved surveillance and case recognition, or increasing person-to-person transmission. But Dr. Fukuda stressed that for now there is "no convincing evidence" of any change in the transmissibility of MERS-CoV or of its genetic makeup, and no evidence that MERS-CoV is cutting infectious swaths through communities.
The WHO committee put all the MERS-CoV evidence "on the table to see how it adds up," to produce "a very sober and critical assessment," a process Dr. Fukuda said could not have happened even 10 years ago.
On Twitter @mitchelzoler
Six factors predict 1-year survival after TAVR
WASHINGTON – Patient gender was the only factor that significantly affected stroke risk during the first year following transcatheter aortic valve replacement, according to an analysis of data from nearly 6,000 patients in the U.S. registry for these procedures.
The 1-year outcome results also showed six factors that significantly affected 1-year mortality following transcatheter aortic valve replacement (TAVR): age, gender, severe chronic obstructive pulmonary disease (COPD), end-stage renal disease, TAVR access route, and risk score.
"Identification of these associations is essential for developing risk-prediction models, and will aid in patient-selection criteria for TAVR," said Dr. David R. Holmes Jr., who presented the results at the annual meeting of the American College of Cardiology. "The factors we have identified will be used to develop models that patients and physicians can use for deciding on appropriate treatment for aortic stenosis."
One-year outcomes for 5,980 of the first U.S. Medicare patients to undergo TAVR since it arrived on the U.S. market in November 2011 came from records of the Centers for Medicare & Medicaid Services (CMS) for patients enrolled during November 2011–July 2013 in the Society of Thoracic Surgeons (STS)/American College of Cardiology (ACC) Transcatheter Valve Therapy Registry.
The registry’s findings also continued to generate interest in the risk level of the first wave of U.S. patients undergoing routine TAVR. The 5,980 registry patients with 1-year outcome data had a median PROM (STS Predicted Risk of Operative Mortality) score of 7% prior to treatment, showing that a substantial number of U.S. TAVR patients had STS scores below the device’s labeled minimum risk score of 8%.
"Sometimes you look at a patient and say, ‘I don’t care what the STS score is, I think this patient will have problems’ " with surgical valve replacement, said Dr. Holmes, an interventional cardiologist and professor of medicine at the Mayo Clinic in Rochester, Minn. "You need two thoracic surgeons who say that the patient is high risk" and should undergo TAVR regardless of a low STS PROM score, "and that is what carries the day" in terms of securing Medicare coverage for the procedure, he said.
The STS and ACC began the registry at the request of the Food and Drug Administration and to satisfy a CMS mandate to track all Medicare TAVR patients. The first wave of patients entered the registry when the Sapien valve system marketed by Edwards became the first TAVR device approved for routine U.S. use in patients with severe aortic stenosis judged ineligible for surgical valve replacement. In October 2012, the FDA approved the same device as an alternative to surgery for "high-risk" but operable patients, generally defined as those with an STS PROM of at least 8%. The PARTNER trial that led to FDA approval of the Sapien system for operable patients had a minimum STS PROM score threshold for enrollment of 10%, and enrolled patients had an average score of 11.8% (N. Engl. J. Med. 2011;364:2187-98).
Last November, researchers running the registry reported in-hospital and 30-day outcomes for 7,710 of the first TAVR patients who entered during November 2011–May 2013 (JAMA 2013;310:2069-77). They collected 1-year outcomes by linking the registry to data from the CMS Administrative Claims Center.
The results showed a 1-year mortality of 26%, and a 1-year stroke rate of 4%.
"Mortality was fairly consistent with results from trials; however, the stroke rate was lower, which might result from how strokes are identified" in a registry compared with more structured follow-up in trials, commented Dr. David E. Kandzari, director of interventional cardiology and chief scientific officer at the Piedmont Heart Institute in Atlanta. "It’s also important that the mortality rate [in the registry] occurred with a high, 36% rate of nontransfemoral access. The higher mortality seen in the registry with nontransfemoral access cannot be discounted. This is probably the highest event rate we will see" with TAVR, Dr. Kandzari predicted. "We can expect outcomes will continue to improve" with advances in TAVR technique and technology.
In multivariate modeling, gender was the only analyzed factor that significantly linked with the 1-year stroke rate. Men had a relative 34% lower stroke rate than women. Women are known to have higher stroke rates than men following other catheter procedures, such as arrhythmia ablations, though the reasons why remain unclear, Dr. Holmes noted.
The relative rates for the six factors that each had a significant link to mortality were a 36% increased mortality for patients aged 85-94 years, compared with those younger than 75 years; a 19% increased rate in men, compared with women; a 41% increased rate in patients with severe COPD, compared with patients with no or mild COPD; an 81% increased rate in dialysis patients, compared with patients with normal creatinine levels; a 42% higher rate with nontransfemoral access, compared with transfemoral access; and a 44% higher mortality among patients with an STS PROM score of 8%-15%, compared with patients with a score of less than 8%.
The substantial number of U.S. patients who have undergone TAVR despite relatively low STS PROM scores, a pattern first seen in baseline data from the entire group of 7,710 patients with in-hospital data in the registry reported last November, also stuck out in the subgroup of 5,980 patients with 1-year data in the new report. Some experts at the meeting commented on the low PROM scores, a phenomenon that last year’s JAMA report dubbed "risk creep," as well as on the rapid uptake of TAVR in routine U.S. practice, with some 8,000 patients undergoing the procedure at about 230 U.S. sites during the first 21 months it was available.
"I’m amazed that we brought 200 centers online [performing TAVR] in just 20 months," commented Dr. David L. Brown, an interventional cardiologist and professor of medicine at Stony Brook (N.Y.) University.
"It’s very striking how quickly the technology was adopted in the United States; 5,980 patients is an enormous number compared with the numbers in the randomized trials," commented Dr. Cindy L. Grines, an interventional cardiologist and vice president at Detroit Medical Center.
"There was incredible, pent-up interest" in TAVR, Dr. Holmes explained.
Dr. Brown characterized the low STS PROM scores of many of the TAVR patients in the registry as "real-world risk creep." Dr. Grines provided a rationale for why patients with relatively low STS PROM scores may undergo TAVR. "There is something to be said for the eyeball estimate of how sick a patient is, and no registry can collect all that information," she said.
"It’s frailty that often gets a patient to TAVR, and frailty is hard to quantify. Frailty can exist even when the STS score is not high," commented Dr. James B. Hermiller, an interventional cardiologist at St. Vincent Heart Center in Indianapolis.
The registry does not receive commercial support. Dr. Holmes had no relevant disclosures. Dr. Kandzari has been a consultant to or received honoraria from Micell Technologies, Biotronik, and other companies. Dr. Brown had no disclosures. Dr. Grines has been a consultant to or received honoraria from Bristol-Meyers Squibb, Merck, and other companies. Dr. Hermiller has been a consultant to or received honoraria from St. Jude, Abbott Vascular, Boston Scientific, and Medtronic.
On Twitter @mitchelzoler
WASHINGTON – Patient gender was the only factor that significantly affected stroke risk during the first year following transcatheter aortic valve replacement, according to an analysis of data from nearly 6,000 patients in the U.S. registry for these procedures.
The 1-year outcome results also showed six factors that significantly affected 1-year mortality following transcatheter aortic valve replacement (TAVR): age, gender, severe chronic obstructive pulmonary disease (COPD), end-stage renal disease, TAVR access route, and risk score.
"Identification of these associations is essential for developing risk-prediction models, and will aid in patient-selection criteria for TAVR," said Dr. David R. Holmes Jr., who presented the results at the annual meeting of the American College of Cardiology. "The factors we have identified will be used to develop models that patients and physicians can use for deciding on appropriate treatment for aortic stenosis."
One-year outcomes for 5,980 of the first U.S. Medicare patients to undergo TAVR since it arrived on the U.S. market in November 2011 came from records of the Centers for Medicare & Medicaid Services (CMS) for patients enrolled during November 2011–July 2013 in the Society of Thoracic Surgeons (STS)/American College of Cardiology (ACC) Transcatheter Valve Therapy Registry.
The registry’s findings also continued to generate interest in the risk level of the first wave of U.S. patients undergoing routine TAVR. The 5,980 registry patients with 1-year outcome data had a median PROM (STS Predicted Risk of Operative Mortality) score of 7% prior to treatment, showing that a substantial number of U.S. TAVR patients had STS scores below the device’s labeled minimum risk score of 8%.
"Sometimes you look at a patient and say, ‘I don’t care what the STS score is, I think this patient will have problems’ " with surgical valve replacement, said Dr. Holmes, an interventional cardiologist and professor of medicine at the Mayo Clinic in Rochester, Minn. "You need two thoracic surgeons who say that the patient is high risk" and should undergo TAVR regardless of a low STS PROM score, "and that is what carries the day" in terms of securing Medicare coverage for the procedure, he said.
The STS and ACC began the registry at the request of the Food and Drug Administration and to satisfy a CMS mandate to track all Medicare TAVR patients. The first wave of patients entered the registry when the Sapien valve system marketed by Edwards became the first TAVR device approved for routine U.S. use in patients with severe aortic stenosis judged ineligible for surgical valve replacement. In October 2012, the FDA approved the same device as an alternative to surgery for "high-risk" but operable patients, generally defined as those with an STS PROM of at least 8%. The PARTNER trial that led to FDA approval of the Sapien system for operable patients had a minimum STS PROM score threshold for enrollment of 10%, and enrolled patients had an average score of 11.8% (N. Engl. J. Med. 2011;364:2187-98).
Last November, researchers running the registry reported in-hospital and 30-day outcomes for 7,710 of the first TAVR patients who entered during November 2011–May 2013 (JAMA 2013;310:2069-77). They collected 1-year outcomes by linking the registry to data from the CMS Administrative Claims Center.
The results showed a 1-year mortality of 26%, and a 1-year stroke rate of 4%.
"Mortality was fairly consistent with results from trials; however, the stroke rate was lower, which might result from how strokes are identified" in a registry compared with more structured follow-up in trials, commented Dr. David E. Kandzari, director of interventional cardiology and chief scientific officer at the Piedmont Heart Institute in Atlanta. "It’s also important that the mortality rate [in the registry] occurred with a high, 36% rate of nontransfemoral access. The higher mortality seen in the registry with nontransfemoral access cannot be discounted. This is probably the highest event rate we will see" with TAVR, Dr. Kandzari predicted. "We can expect outcomes will continue to improve" with advances in TAVR technique and technology.
In multivariate modeling, gender was the only analyzed factor that significantly linked with the 1-year stroke rate. Men had a relative 34% lower stroke rate than women. Women are known to have higher stroke rates than men following other catheter procedures, such as arrhythmia ablations, though the reasons why remain unclear, Dr. Holmes noted.
The relative rates for the six factors that each had a significant link to mortality were a 36% increased mortality for patients aged 85-94 years, compared with those younger than 75 years; a 19% increased rate in men, compared with women; a 41% increased rate in patients with severe COPD, compared with patients with no or mild COPD; an 81% increased rate in dialysis patients, compared with patients with normal creatinine levels; a 42% higher rate with nontransfemoral access, compared with transfemoral access; and a 44% higher mortality among patients with an STS PROM score of 8%-15%, compared with patients with a score of less than 8%.
The substantial number of U.S. patients who have undergone TAVR despite relatively low STS PROM scores, a pattern first seen in baseline data from the entire group of 7,710 patients with in-hospital data in the registry reported last November, also stuck out in the subgroup of 5,980 patients with 1-year data in the new report. Some experts at the meeting commented on the low PROM scores, a phenomenon that last year’s JAMA report dubbed "risk creep," as well as on the rapid uptake of TAVR in routine U.S. practice, with some 8,000 patients undergoing the procedure at about 230 U.S. sites during the first 21 months it was available.
"I’m amazed that we brought 200 centers online [performing TAVR] in just 20 months," commented Dr. David L. Brown, an interventional cardiologist and professor of medicine at Stony Brook (N.Y.) University.
"It’s very striking how quickly the technology was adopted in the United States; 5,980 patients is an enormous number compared with the numbers in the randomized trials," commented Dr. Cindy L. Grines, an interventional cardiologist and vice president at Detroit Medical Center.
"There was incredible, pent-up interest" in TAVR, Dr. Holmes explained.
Dr. Brown characterized the low STS PROM scores of many of the TAVR patients in the registry as "real-world risk creep." Dr. Grines provided a rationale for why patients with relatively low STS PROM scores may undergo TAVR. "There is something to be said for the eyeball estimate of how sick a patient is, and no registry can collect all that information," she said.
"It’s frailty that often gets a patient to TAVR, and frailty is hard to quantify. Frailty can exist even when the STS score is not high," commented Dr. James B. Hermiller, an interventional cardiologist at St. Vincent Heart Center in Indianapolis.
The registry does not receive commercial support. Dr. Holmes had no relevant disclosures. Dr. Kandzari has been a consultant to or received honoraria from Micell Technologies, Biotronik, and other companies. Dr. Brown had no disclosures. Dr. Grines has been a consultant to or received honoraria from Bristol-Meyers Squibb, Merck, and other companies. Dr. Hermiller has been a consultant to or received honoraria from St. Jude, Abbott Vascular, Boston Scientific, and Medtronic.
On Twitter @mitchelzoler
WASHINGTON – Patient gender was the only factor that significantly affected stroke risk during the first year following transcatheter aortic valve replacement, according to an analysis of data from nearly 6,000 patients in the U.S. registry for these procedures.
The 1-year outcome results also showed six factors that significantly affected 1-year mortality following transcatheter aortic valve replacement (TAVR): age, gender, severe chronic obstructive pulmonary disease (COPD), end-stage renal disease, TAVR access route, and risk score.
"Identification of these associations is essential for developing risk-prediction models, and will aid in patient-selection criteria for TAVR," said Dr. David R. Holmes Jr., who presented the results at the annual meeting of the American College of Cardiology. "The factors we have identified will be used to develop models that patients and physicians can use for deciding on appropriate treatment for aortic stenosis."
One-year outcomes for 5,980 of the first U.S. Medicare patients to undergo TAVR since it arrived on the U.S. market in November 2011 came from records of the Centers for Medicare & Medicaid Services (CMS) for patients enrolled during November 2011–July 2013 in the Society of Thoracic Surgeons (STS)/American College of Cardiology (ACC) Transcatheter Valve Therapy Registry.
The registry’s findings also continued to generate interest in the risk level of the first wave of U.S. patients undergoing routine TAVR. The 5,980 registry patients with 1-year outcome data had a median PROM (STS Predicted Risk of Operative Mortality) score of 7% prior to treatment, showing that a substantial number of U.S. TAVR patients had STS scores below the device’s labeled minimum risk score of 8%.
"Sometimes you look at a patient and say, ‘I don’t care what the STS score is, I think this patient will have problems’ " with surgical valve replacement, said Dr. Holmes, an interventional cardiologist and professor of medicine at the Mayo Clinic in Rochester, Minn. "You need two thoracic surgeons who say that the patient is high risk" and should undergo TAVR regardless of a low STS PROM score, "and that is what carries the day" in terms of securing Medicare coverage for the procedure, he said.
The STS and ACC began the registry at the request of the Food and Drug Administration and to satisfy a CMS mandate to track all Medicare TAVR patients. The first wave of patients entered the registry when the Sapien valve system marketed by Edwards became the first TAVR device approved for routine U.S. use in patients with severe aortic stenosis judged ineligible for surgical valve replacement. In October 2012, the FDA approved the same device as an alternative to surgery for "high-risk" but operable patients, generally defined as those with an STS PROM of at least 8%. The PARTNER trial that led to FDA approval of the Sapien system for operable patients had a minimum STS PROM score threshold for enrollment of 10%, and enrolled patients had an average score of 11.8% (N. Engl. J. Med. 2011;364:2187-98).
Last November, researchers running the registry reported in-hospital and 30-day outcomes for 7,710 of the first TAVR patients who entered during November 2011–May 2013 (JAMA 2013;310:2069-77). They collected 1-year outcomes by linking the registry to data from the CMS Administrative Claims Center.
The results showed a 1-year mortality of 26%, and a 1-year stroke rate of 4%.
"Mortality was fairly consistent with results from trials; however, the stroke rate was lower, which might result from how strokes are identified" in a registry compared with more structured follow-up in trials, commented Dr. David E. Kandzari, director of interventional cardiology and chief scientific officer at the Piedmont Heart Institute in Atlanta. "It’s also important that the mortality rate [in the registry] occurred with a high, 36% rate of nontransfemoral access. The higher mortality seen in the registry with nontransfemoral access cannot be discounted. This is probably the highest event rate we will see" with TAVR, Dr. Kandzari predicted. "We can expect outcomes will continue to improve" with advances in TAVR technique and technology.
In multivariate modeling, gender was the only analyzed factor that significantly linked with the 1-year stroke rate. Men had a relative 34% lower stroke rate than women. Women are known to have higher stroke rates than men following other catheter procedures, such as arrhythmia ablations, though the reasons why remain unclear, Dr. Holmes noted.
The relative rates for the six factors that each had a significant link to mortality were a 36% increased mortality for patients aged 85-94 years, compared with those younger than 75 years; a 19% increased rate in men, compared with women; a 41% increased rate in patients with severe COPD, compared with patients with no or mild COPD; an 81% increased rate in dialysis patients, compared with patients with normal creatinine levels; a 42% higher rate with nontransfemoral access, compared with transfemoral access; and a 44% higher mortality among patients with an STS PROM score of 8%-15%, compared with patients with a score of less than 8%.
The substantial number of U.S. patients who have undergone TAVR despite relatively low STS PROM scores, a pattern first seen in baseline data from the entire group of 7,710 patients with in-hospital data in the registry reported last November, also stuck out in the subgroup of 5,980 patients with 1-year data in the new report. Some experts at the meeting commented on the low PROM scores, a phenomenon that last year’s JAMA report dubbed "risk creep," as well as on the rapid uptake of TAVR in routine U.S. practice, with some 8,000 patients undergoing the procedure at about 230 U.S. sites during the first 21 months it was available.
"I’m amazed that we brought 200 centers online [performing TAVR] in just 20 months," commented Dr. David L. Brown, an interventional cardiologist and professor of medicine at Stony Brook (N.Y.) University.
"It’s very striking how quickly the technology was adopted in the United States; 5,980 patients is an enormous number compared with the numbers in the randomized trials," commented Dr. Cindy L. Grines, an interventional cardiologist and vice president at Detroit Medical Center.
"There was incredible, pent-up interest" in TAVR, Dr. Holmes explained.
Dr. Brown characterized the low STS PROM scores of many of the TAVR patients in the registry as "real-world risk creep." Dr. Grines provided a rationale for why patients with relatively low STS PROM scores may undergo TAVR. "There is something to be said for the eyeball estimate of how sick a patient is, and no registry can collect all that information," she said.
"It’s frailty that often gets a patient to TAVR, and frailty is hard to quantify. Frailty can exist even when the STS score is not high," commented Dr. James B. Hermiller, an interventional cardiologist at St. Vincent Heart Center in Indianapolis.
The registry does not receive commercial support. Dr. Holmes had no relevant disclosures. Dr. Kandzari has been a consultant to or received honoraria from Micell Technologies, Biotronik, and other companies. Dr. Brown had no disclosures. Dr. Grines has been a consultant to or received honoraria from Bristol-Meyers Squibb, Merck, and other companies. Dr. Hermiller has been a consultant to or received honoraria from St. Jude, Abbott Vascular, Boston Scientific, and Medtronic.
On Twitter @mitchelzoler
AT ACC 2014
Key clinical point: One-year outcome data from almost 6,000 U.S. TAVR patients give potential insight into the procedure’s best targets.
Major finding: Six factors significantly influenced 1-year survival following transcatheter aortic valve replacement.
Data source: The STS/ACC Transcatheter Valve Therapy Registry, which included 1-year outcomes data for the first 5,980 U.S. patients who underwent TAVR since November 2011.
Disclosures: The registry does not receive commercial support. Dr. Holmes had no relevant disclosures.
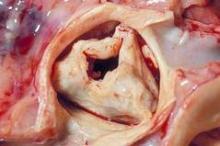
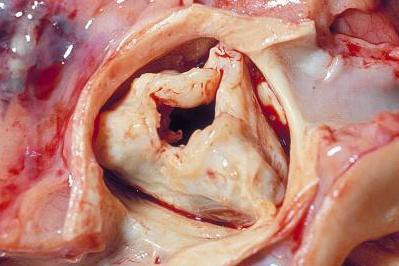
Efficacy, safety confirmed for transcatheter pulmonary valve
WASHINGTON – A transcatheter pulmonary valve system that provides a new right ventricle to pulmonary artery conduit to congenital heart disease patients without the need for open heart surgery performed a little better in a real world registry at 10 U.S. centers than it had in the pivotal trial that led to the system’s 2010 FDA approval.
The new results "confirm the strong performance of the Melody transcatheter pulmonary valve achieved by real world providers with results comparable to the U.S. investigational device exemption [IDE] trial," Dr. Aimee K. Armstrong said at the annual meeting of the American College of Cardiology. The "high level" of 97% freedom from transcatheter pulmonary valve (TPV) dysfunction at 1 year "was better than in the IDE trial," where the level reached 94%, noted Dr. Armstrong, an interventional cardiologist in the pediatric cardiac catheterization laboratory at the University of Michigan in Ann Arbor.
The registry study, which the Food and Drug Administration mandated when it approved the Melody valve in 2010, ran during July 2010 to July 2012 at 10 U.S. centers that had not participated in the pivotal trial. The 99 patients who received an implant that stayed in place for at least 1 day ranged from 5 to 45 years old, with an average age of 20 years.
Although patient follow-up averaged 22 months, the study’s primary endpoint was acceptable hemodynamic function within the conduit at 6 months, with a prespecified performance goal of 75% of patients achieving this outcome. The outcome actually occurred in 97% of the 90 evaluable patients at 6 months, and in 88% of all 99 patients who received a conduit. The difference between each of these rates and the performance goal was statistically significant, Dr. Armstrong said.
The transcatheter valve showed excellent performance by other criteria as well. Acceptable hemodynamic function continued through 1 year in 94% of the 87 implanted patients with evaluable data at 12 months, which translated to 83% of the entire 99 patients in the implanted group. Severe or moderate pulmonary valve regurgitation existed in 85% of the patients before treatment; after treatment no patient had severe or moderate regurgitation, and after 1 year 63% had no regurgitation, 24% had trace, and 12% had mild regurgitation (figures total 99% because of rounding). The 1-year rate of 97% of patients free from dysfunction of their implanted valve appeared to surpass the 94% rate seen in the pivotal trial (Circulation 2010;122:507-16). Dr. Armstrong suggested that if this represented truly improved performance, it may have resulted from lessons during the prior trial on how to best deploy the transcatheter pulmonary valve.
"We learned that the need for reinterventions in the prior trial was primarily due to stent fracture," she said. To reduce the fracture risk, she and her associates routinely began by placing stainless steel stents in the pulmonary artery, an approach known as "pre-stenting," which makes "the risk of fracture go down significantly," she said. Investigators in the new study used pre-stents in 76% of the cases, compared with 35% in the first study. "Our practice now is to pre-stent almost all patients," Dr. Armstrong said.
The results also showed that high right-ventricular pressure prior to valve placement was the only variable independently associated with subsequent valve dysfunction. "Patients who go into the procedure with a very stenotic conduit are probably at higher risk for transcatheter pulmonary valve dysfunction down the road," she said.
The study was sponsored by Medtronic, which markets the Melody transcatheter pulmonary valve. Dr. Armstrong said that she has received consultant fees and honoraria from Siemens Healthcare and St. Jude Medical, and has received research funding from Medtronic and Edwards Lifesciences.
On Twitter @mitchelzoler
The importance of this study cannot be overestimated. Congenital heart disease is the No. 1 birth defect worldwide, affecting nearly 1% of all live births. Our success in treating infants, children, and teenagers with congenital heart disease has produced an epidemic of young adults with congenital heart disease, an estimated 1.5-2 million Americans today
These young adults need to undergo multiple major heart surgeries during their life. The transcatheter pulmonary valve provides, for the first time, a technology to treat a common problem for patients with congenital heart disease in a way that produces excellent results without the need for surgery.
The most impressive part of the findings is that freedom from valve dysfunction after 1 year was even greater than in the initial trial cohort. These results show that this technology is both transferable and can improve. This was very important work for a population that desperately needs improved treatments.
Dr. Evan M. Zahn is director of the congenital heart program of the Heart Institute of Cedar’s Sinai Medical Center in Los Angeles. He said that he has received consulting fees and honoraria from Medtronic and research grants from Edwards Lifesciences. Dr. Zahn was a coinvestigator in the pivotal trial of the Melody transcatheter pulmonary valve system when he was on the staff at Miami Children’s Hospital He made these comments during a press conference.
The importance of this study cannot be overestimated. Congenital heart disease is the No. 1 birth defect worldwide, affecting nearly 1% of all live births. Our success in treating infants, children, and teenagers with congenital heart disease has produced an epidemic of young adults with congenital heart disease, an estimated 1.5-2 million Americans today
These young adults need to undergo multiple major heart surgeries during their life. The transcatheter pulmonary valve provides, for the first time, a technology to treat a common problem for patients with congenital heart disease in a way that produces excellent results without the need for surgery.
The most impressive part of the findings is that freedom from valve dysfunction after 1 year was even greater than in the initial trial cohort. These results show that this technology is both transferable and can improve. This was very important work for a population that desperately needs improved treatments.
Dr. Evan M. Zahn is director of the congenital heart program of the Heart Institute of Cedar’s Sinai Medical Center in Los Angeles. He said that he has received consulting fees and honoraria from Medtronic and research grants from Edwards Lifesciences. Dr. Zahn was a coinvestigator in the pivotal trial of the Melody transcatheter pulmonary valve system when he was on the staff at Miami Children’s Hospital He made these comments during a press conference.
The importance of this study cannot be overestimated. Congenital heart disease is the No. 1 birth defect worldwide, affecting nearly 1% of all live births. Our success in treating infants, children, and teenagers with congenital heart disease has produced an epidemic of young adults with congenital heart disease, an estimated 1.5-2 million Americans today
These young adults need to undergo multiple major heart surgeries during their life. The transcatheter pulmonary valve provides, for the first time, a technology to treat a common problem for patients with congenital heart disease in a way that produces excellent results without the need for surgery.
The most impressive part of the findings is that freedom from valve dysfunction after 1 year was even greater than in the initial trial cohort. These results show that this technology is both transferable and can improve. This was very important work for a population that desperately needs improved treatments.
Dr. Evan M. Zahn is director of the congenital heart program of the Heart Institute of Cedar’s Sinai Medical Center in Los Angeles. He said that he has received consulting fees and honoraria from Medtronic and research grants from Edwards Lifesciences. Dr. Zahn was a coinvestigator in the pivotal trial of the Melody transcatheter pulmonary valve system when he was on the staff at Miami Children’s Hospital He made these comments during a press conference.
WASHINGTON – A transcatheter pulmonary valve system that provides a new right ventricle to pulmonary artery conduit to congenital heart disease patients without the need for open heart surgery performed a little better in a real world registry at 10 U.S. centers than it had in the pivotal trial that led to the system’s 2010 FDA approval.
The new results "confirm the strong performance of the Melody transcatheter pulmonary valve achieved by real world providers with results comparable to the U.S. investigational device exemption [IDE] trial," Dr. Aimee K. Armstrong said at the annual meeting of the American College of Cardiology. The "high level" of 97% freedom from transcatheter pulmonary valve (TPV) dysfunction at 1 year "was better than in the IDE trial," where the level reached 94%, noted Dr. Armstrong, an interventional cardiologist in the pediatric cardiac catheterization laboratory at the University of Michigan in Ann Arbor.
The registry study, which the Food and Drug Administration mandated when it approved the Melody valve in 2010, ran during July 2010 to July 2012 at 10 U.S. centers that had not participated in the pivotal trial. The 99 patients who received an implant that stayed in place for at least 1 day ranged from 5 to 45 years old, with an average age of 20 years.
Although patient follow-up averaged 22 months, the study’s primary endpoint was acceptable hemodynamic function within the conduit at 6 months, with a prespecified performance goal of 75% of patients achieving this outcome. The outcome actually occurred in 97% of the 90 evaluable patients at 6 months, and in 88% of all 99 patients who received a conduit. The difference between each of these rates and the performance goal was statistically significant, Dr. Armstrong said.
The transcatheter valve showed excellent performance by other criteria as well. Acceptable hemodynamic function continued through 1 year in 94% of the 87 implanted patients with evaluable data at 12 months, which translated to 83% of the entire 99 patients in the implanted group. Severe or moderate pulmonary valve regurgitation existed in 85% of the patients before treatment; after treatment no patient had severe or moderate regurgitation, and after 1 year 63% had no regurgitation, 24% had trace, and 12% had mild regurgitation (figures total 99% because of rounding). The 1-year rate of 97% of patients free from dysfunction of their implanted valve appeared to surpass the 94% rate seen in the pivotal trial (Circulation 2010;122:507-16). Dr. Armstrong suggested that if this represented truly improved performance, it may have resulted from lessons during the prior trial on how to best deploy the transcatheter pulmonary valve.
"We learned that the need for reinterventions in the prior trial was primarily due to stent fracture," she said. To reduce the fracture risk, she and her associates routinely began by placing stainless steel stents in the pulmonary artery, an approach known as "pre-stenting," which makes "the risk of fracture go down significantly," she said. Investigators in the new study used pre-stents in 76% of the cases, compared with 35% in the first study. "Our practice now is to pre-stent almost all patients," Dr. Armstrong said.
The results also showed that high right-ventricular pressure prior to valve placement was the only variable independently associated with subsequent valve dysfunction. "Patients who go into the procedure with a very stenotic conduit are probably at higher risk for transcatheter pulmonary valve dysfunction down the road," she said.
The study was sponsored by Medtronic, which markets the Melody transcatheter pulmonary valve. Dr. Armstrong said that she has received consultant fees and honoraria from Siemens Healthcare and St. Jude Medical, and has received research funding from Medtronic and Edwards Lifesciences.
On Twitter @mitchelzoler
WASHINGTON – A transcatheter pulmonary valve system that provides a new right ventricle to pulmonary artery conduit to congenital heart disease patients without the need for open heart surgery performed a little better in a real world registry at 10 U.S. centers than it had in the pivotal trial that led to the system’s 2010 FDA approval.
The new results "confirm the strong performance of the Melody transcatheter pulmonary valve achieved by real world providers with results comparable to the U.S. investigational device exemption [IDE] trial," Dr. Aimee K. Armstrong said at the annual meeting of the American College of Cardiology. The "high level" of 97% freedom from transcatheter pulmonary valve (TPV) dysfunction at 1 year "was better than in the IDE trial," where the level reached 94%, noted Dr. Armstrong, an interventional cardiologist in the pediatric cardiac catheterization laboratory at the University of Michigan in Ann Arbor.
The registry study, which the Food and Drug Administration mandated when it approved the Melody valve in 2010, ran during July 2010 to July 2012 at 10 U.S. centers that had not participated in the pivotal trial. The 99 patients who received an implant that stayed in place for at least 1 day ranged from 5 to 45 years old, with an average age of 20 years.
Although patient follow-up averaged 22 months, the study’s primary endpoint was acceptable hemodynamic function within the conduit at 6 months, with a prespecified performance goal of 75% of patients achieving this outcome. The outcome actually occurred in 97% of the 90 evaluable patients at 6 months, and in 88% of all 99 patients who received a conduit. The difference between each of these rates and the performance goal was statistically significant, Dr. Armstrong said.
The transcatheter valve showed excellent performance by other criteria as well. Acceptable hemodynamic function continued through 1 year in 94% of the 87 implanted patients with evaluable data at 12 months, which translated to 83% of the entire 99 patients in the implanted group. Severe or moderate pulmonary valve regurgitation existed in 85% of the patients before treatment; after treatment no patient had severe or moderate regurgitation, and after 1 year 63% had no regurgitation, 24% had trace, and 12% had mild regurgitation (figures total 99% because of rounding). The 1-year rate of 97% of patients free from dysfunction of their implanted valve appeared to surpass the 94% rate seen in the pivotal trial (Circulation 2010;122:507-16). Dr. Armstrong suggested that if this represented truly improved performance, it may have resulted from lessons during the prior trial on how to best deploy the transcatheter pulmonary valve.
"We learned that the need for reinterventions in the prior trial was primarily due to stent fracture," she said. To reduce the fracture risk, she and her associates routinely began by placing stainless steel stents in the pulmonary artery, an approach known as "pre-stenting," which makes "the risk of fracture go down significantly," she said. Investigators in the new study used pre-stents in 76% of the cases, compared with 35% in the first study. "Our practice now is to pre-stent almost all patients," Dr. Armstrong said.
The results also showed that high right-ventricular pressure prior to valve placement was the only variable independently associated with subsequent valve dysfunction. "Patients who go into the procedure with a very stenotic conduit are probably at higher risk for transcatheter pulmonary valve dysfunction down the road," she said.
The study was sponsored by Medtronic, which markets the Melody transcatheter pulmonary valve. Dr. Armstrong said that she has received consultant fees and honoraria from Siemens Healthcare and St. Jude Medical, and has received research funding from Medtronic and Edwards Lifesciences.
On Twitter @mitchelzoler
AT ACC 2014
Key clinical point: The Melody transcatheter pulmonary valve system worked as well in a real world registry as it did in its pivotal trial as a conduit between the right ventricle and pulmonary artery.
Major finding: Acceptable hemodynamic function at 6 months occurred in 88% of implanted patients, significantly surpassing the 75% performance goal.
Data source: A series of 99 patients who received a transcatheter pulmonary valve at any of 10 participating U.S. centers.
Disclosures: The study was sponsored by Medtronic, which markets the Melody transcatheter pulmonary valve. Dr. Armstrong said that she has received consultant fees and honoraria from Siemens Healthcare and St. Jude Medical, and has received research funding from Medtronic and Edwards Lifesciences.
HS cardiac troponin T shows MI rule-out potential
WASHINGTON – Patients presenting with chest pain with a negative ECG and an undetectable blood level of cardiac troponin T on a high-sensitivity assay had virtually no risk of a myocardial infarction during the following 30 days, in a retrospective review of more than 8,900 patients at one Swedish medical center.
Applying a high-sensitivity cardiac troponin T screen to rule out a possible MI in patients who present to emergency departments with chest pain could substantially reduce the rate of unnecessary hospitalizations such patients, Dr. Martin Holzmann said at the annual meeting of the American College of Cardiology.
During December 2010–December 2012, a total of 8,907 patients older than 25 years who presented to the emergency department at one large European medical center with chest pain, no identifiable ST-segment elevation, or depression on their ECG, and a blood level of cardiac troponin T that was undetectable at less than 5 ng/L on a high-sensitivity assay. From this group of patients, 15 subsequently developed an MI during the next 30 days of follow-up.
This finding showed that an undetectable level of high-sensitivity cardiac troponin T had a negative predictive accuracy of 99.8%, said Dr. Holzmann, an emergency medicine physician at Karolinska University Hospital in Stockholm. The researchers used an Elecsys 2010 device marketed by Roche for high-sensitivity measurement of cardiac troponin T.
But this finding, while consistent with prior reports suggesting that high-sensitivity measurement of cardiac troponin T could detect or rule out MI, came from a study that was not rigorously enough designed to justify routine high-sensitivity screening for cardiac troponin T in chest-pain patients in emergency departments, commented Dr. Allan S. Jaffe, a cardiologist and professor of medicine and laboratory medicine at the Mayo Clinic in Rochester, Minn. He also noted that the device used in the study is not approved for U.S. marketing.
"This is a strategy that has been hinted at since a paper in 2009 by Dr. Tobias Reichlin (N. Engl. J. Med. 2009;361:858-67)," but Dr. Jaffe questioned whether all patients in the new, Swedish study underwent adequate assessment for new-onset MIs during follow-up.
"I agree that in the long-term this strategy will likely work for low-risk people, but I have concerns" whether adequate proof is now in hand. "I want to see it tested in a study with all the ‘I’s dotted and ‘T’s crossed," including two or three serial measures of high-sensitivity cardiac troponin T, Dr. Jaffe said in an interview.
The study had no commercial funding, and Dr. Holzmann and his associates had no disclosures. Dr. Jaffe said that he has received research support and had consulted for several companies developing devices to measure cardiac troponin T.
On Twitter @mitchelzoler
WASHINGTON – Patients presenting with chest pain with a negative ECG and an undetectable blood level of cardiac troponin T on a high-sensitivity assay had virtually no risk of a myocardial infarction during the following 30 days, in a retrospective review of more than 8,900 patients at one Swedish medical center.
Applying a high-sensitivity cardiac troponin T screen to rule out a possible MI in patients who present to emergency departments with chest pain could substantially reduce the rate of unnecessary hospitalizations such patients, Dr. Martin Holzmann said at the annual meeting of the American College of Cardiology.
During December 2010–December 2012, a total of 8,907 patients older than 25 years who presented to the emergency department at one large European medical center with chest pain, no identifiable ST-segment elevation, or depression on their ECG, and a blood level of cardiac troponin T that was undetectable at less than 5 ng/L on a high-sensitivity assay. From this group of patients, 15 subsequently developed an MI during the next 30 days of follow-up.
This finding showed that an undetectable level of high-sensitivity cardiac troponin T had a negative predictive accuracy of 99.8%, said Dr. Holzmann, an emergency medicine physician at Karolinska University Hospital in Stockholm. The researchers used an Elecsys 2010 device marketed by Roche for high-sensitivity measurement of cardiac troponin T.
But this finding, while consistent with prior reports suggesting that high-sensitivity measurement of cardiac troponin T could detect or rule out MI, came from a study that was not rigorously enough designed to justify routine high-sensitivity screening for cardiac troponin T in chest-pain patients in emergency departments, commented Dr. Allan S. Jaffe, a cardiologist and professor of medicine and laboratory medicine at the Mayo Clinic in Rochester, Minn. He also noted that the device used in the study is not approved for U.S. marketing.
"This is a strategy that has been hinted at since a paper in 2009 by Dr. Tobias Reichlin (N. Engl. J. Med. 2009;361:858-67)," but Dr. Jaffe questioned whether all patients in the new, Swedish study underwent adequate assessment for new-onset MIs during follow-up.
"I agree that in the long-term this strategy will likely work for low-risk people, but I have concerns" whether adequate proof is now in hand. "I want to see it tested in a study with all the ‘I’s dotted and ‘T’s crossed," including two or three serial measures of high-sensitivity cardiac troponin T, Dr. Jaffe said in an interview.
The study had no commercial funding, and Dr. Holzmann and his associates had no disclosures. Dr. Jaffe said that he has received research support and had consulted for several companies developing devices to measure cardiac troponin T.
On Twitter @mitchelzoler
WASHINGTON – Patients presenting with chest pain with a negative ECG and an undetectable blood level of cardiac troponin T on a high-sensitivity assay had virtually no risk of a myocardial infarction during the following 30 days, in a retrospective review of more than 8,900 patients at one Swedish medical center.
Applying a high-sensitivity cardiac troponin T screen to rule out a possible MI in patients who present to emergency departments with chest pain could substantially reduce the rate of unnecessary hospitalizations such patients, Dr. Martin Holzmann said at the annual meeting of the American College of Cardiology.
During December 2010–December 2012, a total of 8,907 patients older than 25 years who presented to the emergency department at one large European medical center with chest pain, no identifiable ST-segment elevation, or depression on their ECG, and a blood level of cardiac troponin T that was undetectable at less than 5 ng/L on a high-sensitivity assay. From this group of patients, 15 subsequently developed an MI during the next 30 days of follow-up.
This finding showed that an undetectable level of high-sensitivity cardiac troponin T had a negative predictive accuracy of 99.8%, said Dr. Holzmann, an emergency medicine physician at Karolinska University Hospital in Stockholm. The researchers used an Elecsys 2010 device marketed by Roche for high-sensitivity measurement of cardiac troponin T.
But this finding, while consistent with prior reports suggesting that high-sensitivity measurement of cardiac troponin T could detect or rule out MI, came from a study that was not rigorously enough designed to justify routine high-sensitivity screening for cardiac troponin T in chest-pain patients in emergency departments, commented Dr. Allan S. Jaffe, a cardiologist and professor of medicine and laboratory medicine at the Mayo Clinic in Rochester, Minn. He also noted that the device used in the study is not approved for U.S. marketing.
"This is a strategy that has been hinted at since a paper in 2009 by Dr. Tobias Reichlin (N. Engl. J. Med. 2009;361:858-67)," but Dr. Jaffe questioned whether all patients in the new, Swedish study underwent adequate assessment for new-onset MIs during follow-up.
"I agree that in the long-term this strategy will likely work for low-risk people, but I have concerns" whether adequate proof is now in hand. "I want to see it tested in a study with all the ‘I’s dotted and ‘T’s crossed," including two or three serial measures of high-sensitivity cardiac troponin T, Dr. Jaffe said in an interview.
The study had no commercial funding, and Dr. Holzmann and his associates had no disclosures. Dr. Jaffe said that he has received research support and had consulted for several companies developing devices to measure cardiac troponin T.
On Twitter @mitchelzoler
AT ACC 2014
Major finding: Undetectable cardiac troponin T and no ECG change ruled out MI in emergency chest-pain patients with 99.8% accuracy.
Data source: Retrospective, single-center review of 8,907 patients with undetectable cardiac troponin T during emergency department visit.
Disclosures: The study had no commercial funding. Dr. Holzmann and his associates had no disclosures.
Hepatitis C drugs pricey, but cure is priceless
It was almost as if someone decided April would be hepatitis C month.
In case you hadn’t notice, April saw publication of six separate articles in the New England Journal of Medicine on what were essentially two new, different, all-oral regimens for hepatitis C infection that both consistently produced better than 90% sustained viral response after 8-24 weeks of treatment with good tolerability.
In short, two different, effective cures for hepatitis C arrived this month with safe and easy-to-administer regimens.
(The articles were by Dr. Stefan Zeuzem (N. Engl. J. Med. 2014 April 24;370:1604-14), Dr. Jordan J. Feld (N. Engl. J. Med. 2014 April 24;370:1594-603), Dr. Fred Poordad (N. Engl. J. Med. 2014 April 12 [doi:10.1056/NEJMoa1402869]), Dr. Kris V. Kowdley (N. Engl. J. Med. 2014 April 11 [doi:10.1056/NEJMoa1402355]), and two articles by Dr. Nezam Afdhal (N. Engl. J. Med. 2014 April 12 [doi:10.1056/NEJMoa1402454]; N. Engl. J. Med. 2014 April 17;370:1483-93).
Heralding the good news were no less than three editorials and perspectives in the journal (by Dr. Raymond T. Chung (N. Engl. J. Med. 2014 April 24;370:1576-8), Dr. Channa R. Jayasekera (N. Engl. J. Med. 2014 April 10 [doi:10.1056/NEJMp1400160]), and Dr. Jay H. Hoofnagle (N. Engl. J. Med. 2014 April 17;370:1552-3), making the New England Journal of Medicine’s April hepatitis C total a whopping nine major items.
That wasn’t all, because not only did April’s hepatitis C tsunami feature major advances in treatment efficacy and safety, it also brought to the table unprecedented issues of drug cost. Unprecedented to the tune of sofosbuvir’s (Sovaldi) $1,000/pill price tag set by manufacturer Gilead. That adds up to a 12-week course of the drug costing $84,000 – and sofosbuvir is just part of one of the new hepatitis C wunderkind combinations.
As one of the commentaries put it, "the predicted costs of the new oral antiviral agents are as breathtaking as their effectiveness."
Breathtaking cost inevitably means media buzz, such as an April piece in the New Yorker, and another in the New York Times. Coming to terms with the cost was also a major focus in all three of the NEJM comment pieces.
Drug cost, especially $1,000/day cost, is nothing to minimize. But it also shouldn’t diminish the news that the era of hepatitis C cure has arrived, and at a speed that adds to the breathlessness. Less than a year ago, a review of hepatitis C treatment pegged the state of treatment as achieving 70%-80% sustained viral response (N. Engl. J. Med. 2013;368:1907-17). That’s some leap, to move from that to two different regimens scoring in the mid-90s in less than a year.
Also striking was how much credit experts gave the antiviral path carved out by HIV research that the hepatitis C work could follow, especially the concept of combining antiviral regimens to subvert emergence of drug resistance.
As experts from Stanford and the Mayo Clinic said in their commentary, the HIV model also carries the promise that drug costs can eventually be controlled, and that widespread administration of these effective new hepatitis C treatments will happen worldwide, just as they have for HIV (N. Engl. J. Med. 2014 April 10 [doi:10.1056/NEJMp1400160]).
"Annual costs for first-line ART [antiretroviral therapy, for HIV] have decreased from more than $20,000 in the mid-1990s to often less than $100/person," they noted. "The task of ART delivery has shifted from specialist physicians to primary care physicians, medical assistants, nurses, and public health officers, and nearly 10 million people in low- and middle-income countries are currently receiving treatment."
In fact, the authors of this commentary suggested that global hepatitis C treatment could piggyback onto the existing infrastructure of global HIV treatment. Another comment author, Dr. Raymond Chung from Massachusetts General, Boston, said "it is incumbent on countries, providers, and pharmaceutical companies to enact delivery strategies that mirror the delivery of antiretrovirals for HIV," in an audio interview with NEJM.
A regimen that costs close to $100,000/patient poses an economic hurdle, but the much greater challenge was creating the agents and finding combinations that safely worked at virus elimination. That achievement dwarfs issues of financial practicality. Competition among drugs will produce price reductions. Drugs eventually come off patent. Society will find a way to foot the bill to eradicate hepatitis C now that tools exist to do it.
On Twitter @mitchelzoler
It was almost as if someone decided April would be hepatitis C month.
In case you hadn’t notice, April saw publication of six separate articles in the New England Journal of Medicine on what were essentially two new, different, all-oral regimens for hepatitis C infection that both consistently produced better than 90% sustained viral response after 8-24 weeks of treatment with good tolerability.
In short, two different, effective cures for hepatitis C arrived this month with safe and easy-to-administer regimens.
(The articles were by Dr. Stefan Zeuzem (N. Engl. J. Med. 2014 April 24;370:1604-14), Dr. Jordan J. Feld (N. Engl. J. Med. 2014 April 24;370:1594-603), Dr. Fred Poordad (N. Engl. J. Med. 2014 April 12 [doi:10.1056/NEJMoa1402869]), Dr. Kris V. Kowdley (N. Engl. J. Med. 2014 April 11 [doi:10.1056/NEJMoa1402355]), and two articles by Dr. Nezam Afdhal (N. Engl. J. Med. 2014 April 12 [doi:10.1056/NEJMoa1402454]; N. Engl. J. Med. 2014 April 17;370:1483-93).
Heralding the good news were no less than three editorials and perspectives in the journal (by Dr. Raymond T. Chung (N. Engl. J. Med. 2014 April 24;370:1576-8), Dr. Channa R. Jayasekera (N. Engl. J. Med. 2014 April 10 [doi:10.1056/NEJMp1400160]), and Dr. Jay H. Hoofnagle (N. Engl. J. Med. 2014 April 17;370:1552-3), making the New England Journal of Medicine’s April hepatitis C total a whopping nine major items.
That wasn’t all, because not only did April’s hepatitis C tsunami feature major advances in treatment efficacy and safety, it also brought to the table unprecedented issues of drug cost. Unprecedented to the tune of sofosbuvir’s (Sovaldi) $1,000/pill price tag set by manufacturer Gilead. That adds up to a 12-week course of the drug costing $84,000 – and sofosbuvir is just part of one of the new hepatitis C wunderkind combinations.
As one of the commentaries put it, "the predicted costs of the new oral antiviral agents are as breathtaking as their effectiveness."
Breathtaking cost inevitably means media buzz, such as an April piece in the New Yorker, and another in the New York Times. Coming to terms with the cost was also a major focus in all three of the NEJM comment pieces.
Drug cost, especially $1,000/day cost, is nothing to minimize. But it also shouldn’t diminish the news that the era of hepatitis C cure has arrived, and at a speed that adds to the breathlessness. Less than a year ago, a review of hepatitis C treatment pegged the state of treatment as achieving 70%-80% sustained viral response (N. Engl. J. Med. 2013;368:1907-17). That’s some leap, to move from that to two different regimens scoring in the mid-90s in less than a year.
Also striking was how much credit experts gave the antiviral path carved out by HIV research that the hepatitis C work could follow, especially the concept of combining antiviral regimens to subvert emergence of drug resistance.
As experts from Stanford and the Mayo Clinic said in their commentary, the HIV model also carries the promise that drug costs can eventually be controlled, and that widespread administration of these effective new hepatitis C treatments will happen worldwide, just as they have for HIV (N. Engl. J. Med. 2014 April 10 [doi:10.1056/NEJMp1400160]).
"Annual costs for first-line ART [antiretroviral therapy, for HIV] have decreased from more than $20,000 in the mid-1990s to often less than $100/person," they noted. "The task of ART delivery has shifted from specialist physicians to primary care physicians, medical assistants, nurses, and public health officers, and nearly 10 million people in low- and middle-income countries are currently receiving treatment."
In fact, the authors of this commentary suggested that global hepatitis C treatment could piggyback onto the existing infrastructure of global HIV treatment. Another comment author, Dr. Raymond Chung from Massachusetts General, Boston, said "it is incumbent on countries, providers, and pharmaceutical companies to enact delivery strategies that mirror the delivery of antiretrovirals for HIV," in an audio interview with NEJM.
A regimen that costs close to $100,000/patient poses an economic hurdle, but the much greater challenge was creating the agents and finding combinations that safely worked at virus elimination. That achievement dwarfs issues of financial practicality. Competition among drugs will produce price reductions. Drugs eventually come off patent. Society will find a way to foot the bill to eradicate hepatitis C now that tools exist to do it.
On Twitter @mitchelzoler
It was almost as if someone decided April would be hepatitis C month.
In case you hadn’t notice, April saw publication of six separate articles in the New England Journal of Medicine on what were essentially two new, different, all-oral regimens for hepatitis C infection that both consistently produced better than 90% sustained viral response after 8-24 weeks of treatment with good tolerability.
In short, two different, effective cures for hepatitis C arrived this month with safe and easy-to-administer regimens.
(The articles were by Dr. Stefan Zeuzem (N. Engl. J. Med. 2014 April 24;370:1604-14), Dr. Jordan J. Feld (N. Engl. J. Med. 2014 April 24;370:1594-603), Dr. Fred Poordad (N. Engl. J. Med. 2014 April 12 [doi:10.1056/NEJMoa1402869]), Dr. Kris V. Kowdley (N. Engl. J. Med. 2014 April 11 [doi:10.1056/NEJMoa1402355]), and two articles by Dr. Nezam Afdhal (N. Engl. J. Med. 2014 April 12 [doi:10.1056/NEJMoa1402454]; N. Engl. J. Med. 2014 April 17;370:1483-93).
Heralding the good news were no less than three editorials and perspectives in the journal (by Dr. Raymond T. Chung (N. Engl. J. Med. 2014 April 24;370:1576-8), Dr. Channa R. Jayasekera (N. Engl. J. Med. 2014 April 10 [doi:10.1056/NEJMp1400160]), and Dr. Jay H. Hoofnagle (N. Engl. J. Med. 2014 April 17;370:1552-3), making the New England Journal of Medicine’s April hepatitis C total a whopping nine major items.
That wasn’t all, because not only did April’s hepatitis C tsunami feature major advances in treatment efficacy and safety, it also brought to the table unprecedented issues of drug cost. Unprecedented to the tune of sofosbuvir’s (Sovaldi) $1,000/pill price tag set by manufacturer Gilead. That adds up to a 12-week course of the drug costing $84,000 – and sofosbuvir is just part of one of the new hepatitis C wunderkind combinations.
As one of the commentaries put it, "the predicted costs of the new oral antiviral agents are as breathtaking as their effectiveness."
Breathtaking cost inevitably means media buzz, such as an April piece in the New Yorker, and another in the New York Times. Coming to terms with the cost was also a major focus in all three of the NEJM comment pieces.
Drug cost, especially $1,000/day cost, is nothing to minimize. But it also shouldn’t diminish the news that the era of hepatitis C cure has arrived, and at a speed that adds to the breathlessness. Less than a year ago, a review of hepatitis C treatment pegged the state of treatment as achieving 70%-80% sustained viral response (N. Engl. J. Med. 2013;368:1907-17). That’s some leap, to move from that to two different regimens scoring in the mid-90s in less than a year.
Also striking was how much credit experts gave the antiviral path carved out by HIV research that the hepatitis C work could follow, especially the concept of combining antiviral regimens to subvert emergence of drug resistance.
As experts from Stanford and the Mayo Clinic said in their commentary, the HIV model also carries the promise that drug costs can eventually be controlled, and that widespread administration of these effective new hepatitis C treatments will happen worldwide, just as they have for HIV (N. Engl. J. Med. 2014 April 10 [doi:10.1056/NEJMp1400160]).
"Annual costs for first-line ART [antiretroviral therapy, for HIV] have decreased from more than $20,000 in the mid-1990s to often less than $100/person," they noted. "The task of ART delivery has shifted from specialist physicians to primary care physicians, medical assistants, nurses, and public health officers, and nearly 10 million people in low- and middle-income countries are currently receiving treatment."
In fact, the authors of this commentary suggested that global hepatitis C treatment could piggyback onto the existing infrastructure of global HIV treatment. Another comment author, Dr. Raymond Chung from Massachusetts General, Boston, said "it is incumbent on countries, providers, and pharmaceutical companies to enact delivery strategies that mirror the delivery of antiretrovirals for HIV," in an audio interview with NEJM.
A regimen that costs close to $100,000/patient poses an economic hurdle, but the much greater challenge was creating the agents and finding combinations that safely worked at virus elimination. That achievement dwarfs issues of financial practicality. Competition among drugs will produce price reductions. Drugs eventually come off patent. Society will find a way to foot the bill to eradicate hepatitis C now that tools exist to do it.
On Twitter @mitchelzoler
Obstructive sleep apnea complicates atrial fibrillation
WASHINGTON – About 18% of U.S. patients with atrial fibrillation also have a diagnosis of obstructive sleep apnea, and the confluence of the two appeared linked to increased hospitalizations and further progression of atrial fibrillation, based on a registry of more than 10,000 U.S. atrial fibrillation patients.
In addition, patients who have AF and OSA and who are treated with continuous positive airway pressure (CPAP) have a reduced rate of AF progression, Dr. Jonathan P. Piccini Sr. said at the annual meeting of the American College of Cardiology.
"We probably are not screening for obstructive sleep apnea as aggressively as we should" in AF patients, said Dr. Piccini, an electrophysiologist at Duke University in Durham, N.C. "We know that if obstructive sleep apnea is treated [with CPAP,] the AF burden can be dramatically reduced."
The data Dr. Piccini and his associates analyzed came from the ORBIT-AF (Outcomes Registry for Better Informed Treatment of Atrial Fibrillation), which starting in 2010 enrolled more than 10,000 AF patients from 172 U.S. locations with a variety of practice settings and followed them for more than 2 years. Their medical records showed that at enrollment, 1,841 of the 10,132 enrolled AF patients (18%) had also been diagnosed with OSA. Patients with OSA averaged 69 years old, while those without the disorder averaged 76 years old. Those with OSA had an average body mass index of 34 kg/m2, compared with 28 kg/m2 among those without OSA. Patients with OSA also had higher prevalence rates of dyspnea and fatigue than the enrolled AF patients without OSA.
During 2 years of follow-up, the two subgroups had similar rates of all-cause death, cardiovascular events, major bleeding events, and AF progression, but after multivariate adjustment, patients with OSA had a 12% higher rate of hospitalizations than did patients without OSA, a statistically significant difference.
Further analysis of the 1,624 OSA patients showed that 937 (58%) used CPAP during follow-up as a treatment for OSA. Comparison of the CPAP users and nonusers showed no significant difference in outcomes during follow-up for all-cause death, hospitalizations, cardiovascular events, or major bleeding events, but there was a statistically significant, 34% relative drop in the rate of AF progression among CPAP users compared with nonusers.
The ORBIT-AF registry is sponsored by Johnson & Johnson. Dr. Piccini has received remuneration from Johnson & Johnson, Forest Laboratories, and other companies.
On Twitter @mitchelzoler
WASHINGTON – About 18% of U.S. patients with atrial fibrillation also have a diagnosis of obstructive sleep apnea, and the confluence of the two appeared linked to increased hospitalizations and further progression of atrial fibrillation, based on a registry of more than 10,000 U.S. atrial fibrillation patients.
In addition, patients who have AF and OSA and who are treated with continuous positive airway pressure (CPAP) have a reduced rate of AF progression, Dr. Jonathan P. Piccini Sr. said at the annual meeting of the American College of Cardiology.
"We probably are not screening for obstructive sleep apnea as aggressively as we should" in AF patients, said Dr. Piccini, an electrophysiologist at Duke University in Durham, N.C. "We know that if obstructive sleep apnea is treated [with CPAP,] the AF burden can be dramatically reduced."
The data Dr. Piccini and his associates analyzed came from the ORBIT-AF (Outcomes Registry for Better Informed Treatment of Atrial Fibrillation), which starting in 2010 enrolled more than 10,000 AF patients from 172 U.S. locations with a variety of practice settings and followed them for more than 2 years. Their medical records showed that at enrollment, 1,841 of the 10,132 enrolled AF patients (18%) had also been diagnosed with OSA. Patients with OSA averaged 69 years old, while those without the disorder averaged 76 years old. Those with OSA had an average body mass index of 34 kg/m2, compared with 28 kg/m2 among those without OSA. Patients with OSA also had higher prevalence rates of dyspnea and fatigue than the enrolled AF patients without OSA.
During 2 years of follow-up, the two subgroups had similar rates of all-cause death, cardiovascular events, major bleeding events, and AF progression, but after multivariate adjustment, patients with OSA had a 12% higher rate of hospitalizations than did patients without OSA, a statistically significant difference.
Further analysis of the 1,624 OSA patients showed that 937 (58%) used CPAP during follow-up as a treatment for OSA. Comparison of the CPAP users and nonusers showed no significant difference in outcomes during follow-up for all-cause death, hospitalizations, cardiovascular events, or major bleeding events, but there was a statistically significant, 34% relative drop in the rate of AF progression among CPAP users compared with nonusers.
The ORBIT-AF registry is sponsored by Johnson & Johnson. Dr. Piccini has received remuneration from Johnson & Johnson, Forest Laboratories, and other companies.
On Twitter @mitchelzoler
WASHINGTON – About 18% of U.S. patients with atrial fibrillation also have a diagnosis of obstructive sleep apnea, and the confluence of the two appeared linked to increased hospitalizations and further progression of atrial fibrillation, based on a registry of more than 10,000 U.S. atrial fibrillation patients.
In addition, patients who have AF and OSA and who are treated with continuous positive airway pressure (CPAP) have a reduced rate of AF progression, Dr. Jonathan P. Piccini Sr. said at the annual meeting of the American College of Cardiology.
"We probably are not screening for obstructive sleep apnea as aggressively as we should" in AF patients, said Dr. Piccini, an electrophysiologist at Duke University in Durham, N.C. "We know that if obstructive sleep apnea is treated [with CPAP,] the AF burden can be dramatically reduced."
The data Dr. Piccini and his associates analyzed came from the ORBIT-AF (Outcomes Registry for Better Informed Treatment of Atrial Fibrillation), which starting in 2010 enrolled more than 10,000 AF patients from 172 U.S. locations with a variety of practice settings and followed them for more than 2 years. Their medical records showed that at enrollment, 1,841 of the 10,132 enrolled AF patients (18%) had also been diagnosed with OSA. Patients with OSA averaged 69 years old, while those without the disorder averaged 76 years old. Those with OSA had an average body mass index of 34 kg/m2, compared with 28 kg/m2 among those without OSA. Patients with OSA also had higher prevalence rates of dyspnea and fatigue than the enrolled AF patients without OSA.
During 2 years of follow-up, the two subgroups had similar rates of all-cause death, cardiovascular events, major bleeding events, and AF progression, but after multivariate adjustment, patients with OSA had a 12% higher rate of hospitalizations than did patients without OSA, a statistically significant difference.
Further analysis of the 1,624 OSA patients showed that 937 (58%) used CPAP during follow-up as a treatment for OSA. Comparison of the CPAP users and nonusers showed no significant difference in outcomes during follow-up for all-cause death, hospitalizations, cardiovascular events, or major bleeding events, but there was a statistically significant, 34% relative drop in the rate of AF progression among CPAP users compared with nonusers.
The ORBIT-AF registry is sponsored by Johnson & Johnson. Dr. Piccini has received remuneration from Johnson & Johnson, Forest Laboratories, and other companies.
On Twitter @mitchelzoler
AT ACC 2014
Major finding: Atrial fibrillation patients with sleep apnea had a significant, 12% increased rate of hospitalizations, compared with atrial fibrillation patients without sleep apnea.
Data source: The ORBIT-AF registry, which enrolled 10,132 U.S. patients with atrial fibrillation starting in 2010.
Disclosures: The ORBIT-AF registry is sponsored by Johnson & Johnson. Dr. Piccini has received remuneration from Johnson & Johnson, Forest Laboratories, and other companies.
Rivaroxaban replacing warfarin for AF stroke protection
WASHINGTON – Use of the new oral anticoagulant rivaroxaban to prevent stroke in atrial fibrillation patients doubled and then redoubled during 2010-2013, based on a review of nearly 10,000 atrial fibrillation patients who received their care through a large health network in Wisconsin and Illinois.
Data from 9,652 atrial fibrillation (AF) patients who received care through Aurora Health Care during the 4-year period also showed that two new oral anticoagulants, rivaroxaban (Xarelto) and dabigatran (Pradaxa), seemed roughly comparable to warfarin for preventing strokes and not causing bleeding, including in patients aged 80 years or older, a patient group poorly represented in the pivotal trials of the new drugs, Dr. Anton V. Strunets said at the annual meeting of the American College of Cardiology.
"In community-based, real-world practice, use of novel anticoagulants in the older population was substantial and increasing," and the results were also "reassuring" about the efficacy and safety of these drugs during routine use, particularly in the elderly, said Dr. Strunets, a physician on the staff of Aurora Health Care in Milwaukee.
Since 2010, "the type of anticoagulant used has changed dramatically," he said, with the use of warfarin and dabigatran as a percentage of AF patients falling and the use of rivaroxaban rising sharply. After rivaroxaban received Food and Drug Administration approval in 2011 for use in preventing stroke in AF patients, it entered the Aurora practice with roughly 8% of patients on the drug by the end of the year, rising to about 16% in 2012, and up to about 30% by the end of last year. In contrast, about 8% of patients were on dabigatran in 2010, and the level then dropped steadily during the next 3 years, so that last year about 1% of AF patients were on dabigatran. While warfarin remained the most widely used anticoagulant throughout, its share of the treated AF population fell from more than 90% in 2010 to less than 70% in 2013.
These numbers from Aurora matched numbers reported last November from a U.S. registry maintained by the American College of Cardiology, which showed that during June-September 2013, 72% of U.S. AF patients on an anticoagulant received warfarin.
Dr. Strunets cited several factors likely driving shifting anticoagulant use in AF patients, though none were documented by the data he reported: the attraction of once-daily dosing with rivaroxaban to boost compliance, especially in older patients; the coverage allowed by insurers and health plans; and recent concern about dabigatran tied to a new study of the drug’s safety launched by the Food and Drug Administration.
Another facet of oral anticoagulant use at Aurora is that during the 4-year period, 38% of AF patients were 80 years or older, including about 30% of those who received rivaroxaban and 25% of those on dabigatran. The efficacy and safety of treatment, based on the incidence of strokes and of intracranial and gastrointestinal bleeds, were roughly similar in the dabigatran, rivaroxaban, and warfarin subgroups, and the rates for the new anticoagulants also roughly matched the rates seen in the pivotal trials with those drugs, suggesting that the new anticoagulants were performing well when used in octo- and nonagenarians, Dr. Strunets said. But he cautioned that the patients who received rivaroxaban or dabigatran were significantly younger and had significantly lower stroke and bleeding risk scores than patients who received warfarin, a selection bias that may have affected outcomes.
Dr. Strunets and his associates had no disclosures.
On Twitter @mitchelzoler
WASHINGTON – Use of the new oral anticoagulant rivaroxaban to prevent stroke in atrial fibrillation patients doubled and then redoubled during 2010-2013, based on a review of nearly 10,000 atrial fibrillation patients who received their care through a large health network in Wisconsin and Illinois.
Data from 9,652 atrial fibrillation (AF) patients who received care through Aurora Health Care during the 4-year period also showed that two new oral anticoagulants, rivaroxaban (Xarelto) and dabigatran (Pradaxa), seemed roughly comparable to warfarin for preventing strokes and not causing bleeding, including in patients aged 80 years or older, a patient group poorly represented in the pivotal trials of the new drugs, Dr. Anton V. Strunets said at the annual meeting of the American College of Cardiology.
"In community-based, real-world practice, use of novel anticoagulants in the older population was substantial and increasing," and the results were also "reassuring" about the efficacy and safety of these drugs during routine use, particularly in the elderly, said Dr. Strunets, a physician on the staff of Aurora Health Care in Milwaukee.
Since 2010, "the type of anticoagulant used has changed dramatically," he said, with the use of warfarin and dabigatran as a percentage of AF patients falling and the use of rivaroxaban rising sharply. After rivaroxaban received Food and Drug Administration approval in 2011 for use in preventing stroke in AF patients, it entered the Aurora practice with roughly 8% of patients on the drug by the end of the year, rising to about 16% in 2012, and up to about 30% by the end of last year. In contrast, about 8% of patients were on dabigatran in 2010, and the level then dropped steadily during the next 3 years, so that last year about 1% of AF patients were on dabigatran. While warfarin remained the most widely used anticoagulant throughout, its share of the treated AF population fell from more than 90% in 2010 to less than 70% in 2013.
These numbers from Aurora matched numbers reported last November from a U.S. registry maintained by the American College of Cardiology, which showed that during June-September 2013, 72% of U.S. AF patients on an anticoagulant received warfarin.
Dr. Strunets cited several factors likely driving shifting anticoagulant use in AF patients, though none were documented by the data he reported: the attraction of once-daily dosing with rivaroxaban to boost compliance, especially in older patients; the coverage allowed by insurers and health plans; and recent concern about dabigatran tied to a new study of the drug’s safety launched by the Food and Drug Administration.
Another facet of oral anticoagulant use at Aurora is that during the 4-year period, 38% of AF patients were 80 years or older, including about 30% of those who received rivaroxaban and 25% of those on dabigatran. The efficacy and safety of treatment, based on the incidence of strokes and of intracranial and gastrointestinal bleeds, were roughly similar in the dabigatran, rivaroxaban, and warfarin subgroups, and the rates for the new anticoagulants also roughly matched the rates seen in the pivotal trials with those drugs, suggesting that the new anticoagulants were performing well when used in octo- and nonagenarians, Dr. Strunets said. But he cautioned that the patients who received rivaroxaban or dabigatran were significantly younger and had significantly lower stroke and bleeding risk scores than patients who received warfarin, a selection bias that may have affected outcomes.
Dr. Strunets and his associates had no disclosures.
On Twitter @mitchelzoler
WASHINGTON – Use of the new oral anticoagulant rivaroxaban to prevent stroke in atrial fibrillation patients doubled and then redoubled during 2010-2013, based on a review of nearly 10,000 atrial fibrillation patients who received their care through a large health network in Wisconsin and Illinois.
Data from 9,652 atrial fibrillation (AF) patients who received care through Aurora Health Care during the 4-year period also showed that two new oral anticoagulants, rivaroxaban (Xarelto) and dabigatran (Pradaxa), seemed roughly comparable to warfarin for preventing strokes and not causing bleeding, including in patients aged 80 years or older, a patient group poorly represented in the pivotal trials of the new drugs, Dr. Anton V. Strunets said at the annual meeting of the American College of Cardiology.
"In community-based, real-world practice, use of novel anticoagulants in the older population was substantial and increasing," and the results were also "reassuring" about the efficacy and safety of these drugs during routine use, particularly in the elderly, said Dr. Strunets, a physician on the staff of Aurora Health Care in Milwaukee.
Since 2010, "the type of anticoagulant used has changed dramatically," he said, with the use of warfarin and dabigatran as a percentage of AF patients falling and the use of rivaroxaban rising sharply. After rivaroxaban received Food and Drug Administration approval in 2011 for use in preventing stroke in AF patients, it entered the Aurora practice with roughly 8% of patients on the drug by the end of the year, rising to about 16% in 2012, and up to about 30% by the end of last year. In contrast, about 8% of patients were on dabigatran in 2010, and the level then dropped steadily during the next 3 years, so that last year about 1% of AF patients were on dabigatran. While warfarin remained the most widely used anticoagulant throughout, its share of the treated AF population fell from more than 90% in 2010 to less than 70% in 2013.
These numbers from Aurora matched numbers reported last November from a U.S. registry maintained by the American College of Cardiology, which showed that during June-September 2013, 72% of U.S. AF patients on an anticoagulant received warfarin.
Dr. Strunets cited several factors likely driving shifting anticoagulant use in AF patients, though none were documented by the data he reported: the attraction of once-daily dosing with rivaroxaban to boost compliance, especially in older patients; the coverage allowed by insurers and health plans; and recent concern about dabigatran tied to a new study of the drug’s safety launched by the Food and Drug Administration.
Another facet of oral anticoagulant use at Aurora is that during the 4-year period, 38% of AF patients were 80 years or older, including about 30% of those who received rivaroxaban and 25% of those on dabigatran. The efficacy and safety of treatment, based on the incidence of strokes and of intracranial and gastrointestinal bleeds, were roughly similar in the dabigatran, rivaroxaban, and warfarin subgroups, and the rates for the new anticoagulants also roughly matched the rates seen in the pivotal trials with those drugs, suggesting that the new anticoagulants were performing well when used in octo- and nonagenarians, Dr. Strunets said. But he cautioned that the patients who received rivaroxaban or dabigatran were significantly younger and had significantly lower stroke and bleeding risk scores than patients who received warfarin, a selection bias that may have affected outcomes.
Dr. Strunets and his associates had no disclosures.
On Twitter @mitchelzoler
AT ACC 2014
Major finding: Rivaroxaban use grew from approximately 8% of AF patients in 2011 to 16% in 2012 and 30% in 2013.
Data source: Review of anticoagulant care received by 9,652 AF patients treated through Aurora Health Care during 2010-2013.
Disclosures: Dr. Strunets and his associates had no disclosures.