User login
High dietary phosphorus linked to increased mortality
DALLAS – More than one-third of Americans have a high dietary phosphorus intake and an all-cause mortality rate that is more than double that of Americans with lower dietary phosphorus consumption, based on a large observational study.
High dietary phosphorus density – a value of at least 35 mg/kcal for the product of dietary phosphorus intake divided by total energy intake – was associated with a 2.27-fold increased risk of all-cause mortality and a 3.39-fold elevation in cardiovascular mortality, Dr. Alex R. Chang reported at the American Heart Association scientific sessions.
The findings came from an analysis of a nationally representative cohort comprising 8,686 adult participants in the Third National Health and Nutrition Examination Survey (NHANES III). Since this was an observational study, the strong association between high phosphorus intake and increased mortality doesn’t prove causality and there is no evidence that reducing phosphorus consumption would reduce mortality. But the study findings do raise public health concerns, given that 35% of Americans consume daily more than 1,400 mg of phosphorus.
That intake level, twice the Recommended Daily Allowance of 700 mg, was associated with a 2.23-fold increased risk of all-cause mortality in an NHANES III multivariate analysis adjusted for demographics, traditional cardiovascular risk factors, estimated glomerular filtration rate, vitamin D status, and total energy intake. Consumption below the 1,400 mg/day threshold was not associated with any increased mortality risk, according to Dr. Chang, a nephrologist at the Geisinger Health System in Danville, Pa.
During a median 14.7 years of follow-up in the study, there were 1,129 deaths, including 384 cardiovascular deaths.
Inorganic phosphates added to processed foods as preservatives are the chief sources of phosphorus in the U.S. diet. A recent study by researchers at Case Western Reserve University, Cleveland, determined that fully 44% of the 2,394 top-selling branded grocery products contain phosphorus additives. The additives were present in 72% of prepared frozen foods, 70% of dry food mixes, 65% of packaged meats, 57% of baked goods, 54% of soups, and 51% of yogurts. Phosphorus-added foods were not only widespread, they cost less than foods free of phosphorus additives (J. Ren. Nutr. 2013;23:265-70).
It’s noteworthy that the inorganic phosphates used in food additives are more bioavailable than the organic phosphorus found naturally in eggs, nuts, and other foods, Dr. Chang said.
Animal studies point to several possible mechanisms that might account for the observed association between high dietary phosphorus and increased mortality in NHANES III, including promotion of vascular calcification, endothelial dysfunction, and increased levels of fibroblast growth factor 23, which is a phosphaturic hormone whose overexpression is linked to left ventricular hypertrophy, progression of chronic kidney disease, and cardiovascular events.
Dr. Chang’s study was supported by the National Institute of Diabetes and Digestive and Kidney Diseases. He reported having no financial conflicts.
DALLAS – More than one-third of Americans have a high dietary phosphorus intake and an all-cause mortality rate that is more than double that of Americans with lower dietary phosphorus consumption, based on a large observational study.
High dietary phosphorus density – a value of at least 35 mg/kcal for the product of dietary phosphorus intake divided by total energy intake – was associated with a 2.27-fold increased risk of all-cause mortality and a 3.39-fold elevation in cardiovascular mortality, Dr. Alex R. Chang reported at the American Heart Association scientific sessions.
The findings came from an analysis of a nationally representative cohort comprising 8,686 adult participants in the Third National Health and Nutrition Examination Survey (NHANES III). Since this was an observational study, the strong association between high phosphorus intake and increased mortality doesn’t prove causality and there is no evidence that reducing phosphorus consumption would reduce mortality. But the study findings do raise public health concerns, given that 35% of Americans consume daily more than 1,400 mg of phosphorus.
That intake level, twice the Recommended Daily Allowance of 700 mg, was associated with a 2.23-fold increased risk of all-cause mortality in an NHANES III multivariate analysis adjusted for demographics, traditional cardiovascular risk factors, estimated glomerular filtration rate, vitamin D status, and total energy intake. Consumption below the 1,400 mg/day threshold was not associated with any increased mortality risk, according to Dr. Chang, a nephrologist at the Geisinger Health System in Danville, Pa.
During a median 14.7 years of follow-up in the study, there were 1,129 deaths, including 384 cardiovascular deaths.
Inorganic phosphates added to processed foods as preservatives are the chief sources of phosphorus in the U.S. diet. A recent study by researchers at Case Western Reserve University, Cleveland, determined that fully 44% of the 2,394 top-selling branded grocery products contain phosphorus additives. The additives were present in 72% of prepared frozen foods, 70% of dry food mixes, 65% of packaged meats, 57% of baked goods, 54% of soups, and 51% of yogurts. Phosphorus-added foods were not only widespread, they cost less than foods free of phosphorus additives (J. Ren. Nutr. 2013;23:265-70).
It’s noteworthy that the inorganic phosphates used in food additives are more bioavailable than the organic phosphorus found naturally in eggs, nuts, and other foods, Dr. Chang said.
Animal studies point to several possible mechanisms that might account for the observed association between high dietary phosphorus and increased mortality in NHANES III, including promotion of vascular calcification, endothelial dysfunction, and increased levels of fibroblast growth factor 23, which is a phosphaturic hormone whose overexpression is linked to left ventricular hypertrophy, progression of chronic kidney disease, and cardiovascular events.
Dr. Chang’s study was supported by the National Institute of Diabetes and Digestive and Kidney Diseases. He reported having no financial conflicts.
DALLAS – More than one-third of Americans have a high dietary phosphorus intake and an all-cause mortality rate that is more than double that of Americans with lower dietary phosphorus consumption, based on a large observational study.
High dietary phosphorus density – a value of at least 35 mg/kcal for the product of dietary phosphorus intake divided by total energy intake – was associated with a 2.27-fold increased risk of all-cause mortality and a 3.39-fold elevation in cardiovascular mortality, Dr. Alex R. Chang reported at the American Heart Association scientific sessions.
The findings came from an analysis of a nationally representative cohort comprising 8,686 adult participants in the Third National Health and Nutrition Examination Survey (NHANES III). Since this was an observational study, the strong association between high phosphorus intake and increased mortality doesn’t prove causality and there is no evidence that reducing phosphorus consumption would reduce mortality. But the study findings do raise public health concerns, given that 35% of Americans consume daily more than 1,400 mg of phosphorus.
That intake level, twice the Recommended Daily Allowance of 700 mg, was associated with a 2.23-fold increased risk of all-cause mortality in an NHANES III multivariate analysis adjusted for demographics, traditional cardiovascular risk factors, estimated glomerular filtration rate, vitamin D status, and total energy intake. Consumption below the 1,400 mg/day threshold was not associated with any increased mortality risk, according to Dr. Chang, a nephrologist at the Geisinger Health System in Danville, Pa.
During a median 14.7 years of follow-up in the study, there were 1,129 deaths, including 384 cardiovascular deaths.
Inorganic phosphates added to processed foods as preservatives are the chief sources of phosphorus in the U.S. diet. A recent study by researchers at Case Western Reserve University, Cleveland, determined that fully 44% of the 2,394 top-selling branded grocery products contain phosphorus additives. The additives were present in 72% of prepared frozen foods, 70% of dry food mixes, 65% of packaged meats, 57% of baked goods, 54% of soups, and 51% of yogurts. Phosphorus-added foods were not only widespread, they cost less than foods free of phosphorus additives (J. Ren. Nutr. 2013;23:265-70).
It’s noteworthy that the inorganic phosphates used in food additives are more bioavailable than the organic phosphorus found naturally in eggs, nuts, and other foods, Dr. Chang said.
Animal studies point to several possible mechanisms that might account for the observed association between high dietary phosphorus and increased mortality in NHANES III, including promotion of vascular calcification, endothelial dysfunction, and increased levels of fibroblast growth factor 23, which is a phosphaturic hormone whose overexpression is linked to left ventricular hypertrophy, progression of chronic kidney disease, and cardiovascular events.
Dr. Chang’s study was supported by the National Institute of Diabetes and Digestive and Kidney Diseases. He reported having no financial conflicts.
AT THE AHA SCIENTIFIC SESSIONS
Major finding: Of American adults, 35% have a dietary phosphorus intake in excess of 1,400 mg/day, which is associated with a 2.23-fold increased risk of all-cause mortality.
Data source: This study included 8,686 participants in the Third National Health and Nutrition Examination Survey with an average of nearly 15 years of follow-up.
Disclosures: The study was funded by the NIDDKD. The presenter reported having no relevant financial interests.
New-onset hypertension in pregnancy boosts post-delivery risk
DALLAS – Previously normotensive women who develop a hypertensive disorder during pregnancy are at more than double the risk of being diagnosed with prehypertension or hypertension within the first year after delivery, according to a large California study.
"Early postpartum screening and improved follow-up of women with hypertensive disorders first identified during pregnancy may be necessary to identify those at risk for future hypertension or prehypertension," Mary Helen Black, Ph.D., concluded in presenting the results of her population-based study at the American Heart Association scientific sessions.
The study was a retrospective review of the comprehensive electronic health records of 5,960 women who received their prenatal care and subsequently delivered a live singleton neonate at 20 weeks’ gestation or more at a single Kaiser Permanente Southern California medical center during 2006-2010. All of these women were specifically identified as being normotensive prior to their pregnancy and 6% of them developed a hypertensive disorder in pregnancy; 215 of the 358 affected women had preeclampsia or eclampsia.
During the first year after delivery, 63 women, or 1.1% of the total study population, developed frank hypertension. Another 902, or 15%, developed prehypertension during that first year. Prehypertension was defined as a modestly elevated blood pressure of at least 120/80 mm Hg on two consecutive ambulatory visits.
After adjustment for potential confounders including age, race, parity, prepregnancy body mass index, and smoking status, previously normotensive women with hypertension in pregnancy had an adjusted 2.4-fold increased risk of developing hypertension or prehypertension in the year after delivery, compared with women without any hypertensive disorder in pregnancy. Women who developed preeclampsia or eclampsia had a 2.5-fold increased risk, according to Dr. Black, a research scientist at Kaiser Permanente Southern California in Pasadena.
Protocols exist for monitoring and treatment of pregnant women identified as having chronic hypertension prior to pregnancy. However, the new Kaiser study is one of the first to look at the downstream impact of hypertensive disorders in pregnancy arising among women with no preexisting indication of hypertension. The plan is to follow these women further to learn how their clinical outcomes progress. Long-term Scandinavian registry studies have linked hypertensive disorders in pregnancy to increased risk of eventual type 2 diabetes, hypertension, and cardiovascular disease.
Dr. Black and her coinvestigators are planning a postpartum intervention study aimed at preventing onset of type 2 diabetes and cardiovascular disease in previously normotensive women with hypertensive disorders in pregnancy. Serious consideration is being given to intervening with the DASH diet and other nonpharmacologic interventions, she said.
While women with chronic hypertension prior to pregnancy are known to be at increased risk for development of preeclampsia and other hypertensive disorders of pregnancy as well as for gestational diabetes mellitus, the situation regarding women with prepregnancy prehypertension has been less clear. Dr. Black and coworkers shed new light on this issue in a separate study presented at the AHA meeting. She reported that women with prepregnancy prehypertension are also at increased risk for these pregnancy complications.
This study involved 2,156 Kaiser Permanente patients with prepregnancy prehypertension and 5,646 who were normotensive. A total of 8.4% of the women developed a hypertensive disorder during pregnancy, including 373 with preeclampsia/eclampsia. In addition, 1,877 women developed gestational diabetes mellitus.
Women with prehypertension prior to pregnancy were an adjusted 2.7-fold more likely to develop a hypertensive disorder in pregnancy, 2.2-fold more likely to have preeclampsia/eclampsia, and 1.2-fold more likely to develop gestational diabetes than women who were normotensive before pregnancy.
The risk of preeclampsia/eclampsia among women with prepregnancy prehypertension was significantly greater if they were at least 30 years old than if they were younger.
Both studies were funded by Kaiser Permanente. Dr. Black is an employee of the company.
DALLAS – Previously normotensive women who develop a hypertensive disorder during pregnancy are at more than double the risk of being diagnosed with prehypertension or hypertension within the first year after delivery, according to a large California study.
"Early postpartum screening and improved follow-up of women with hypertensive disorders first identified during pregnancy may be necessary to identify those at risk for future hypertension or prehypertension," Mary Helen Black, Ph.D., concluded in presenting the results of her population-based study at the American Heart Association scientific sessions.
The study was a retrospective review of the comprehensive electronic health records of 5,960 women who received their prenatal care and subsequently delivered a live singleton neonate at 20 weeks’ gestation or more at a single Kaiser Permanente Southern California medical center during 2006-2010. All of these women were specifically identified as being normotensive prior to their pregnancy and 6% of them developed a hypertensive disorder in pregnancy; 215 of the 358 affected women had preeclampsia or eclampsia.
During the first year after delivery, 63 women, or 1.1% of the total study population, developed frank hypertension. Another 902, or 15%, developed prehypertension during that first year. Prehypertension was defined as a modestly elevated blood pressure of at least 120/80 mm Hg on two consecutive ambulatory visits.
After adjustment for potential confounders including age, race, parity, prepregnancy body mass index, and smoking status, previously normotensive women with hypertension in pregnancy had an adjusted 2.4-fold increased risk of developing hypertension or prehypertension in the year after delivery, compared with women without any hypertensive disorder in pregnancy. Women who developed preeclampsia or eclampsia had a 2.5-fold increased risk, according to Dr. Black, a research scientist at Kaiser Permanente Southern California in Pasadena.
Protocols exist for monitoring and treatment of pregnant women identified as having chronic hypertension prior to pregnancy. However, the new Kaiser study is one of the first to look at the downstream impact of hypertensive disorders in pregnancy arising among women with no preexisting indication of hypertension. The plan is to follow these women further to learn how their clinical outcomes progress. Long-term Scandinavian registry studies have linked hypertensive disorders in pregnancy to increased risk of eventual type 2 diabetes, hypertension, and cardiovascular disease.
Dr. Black and her coinvestigators are planning a postpartum intervention study aimed at preventing onset of type 2 diabetes and cardiovascular disease in previously normotensive women with hypertensive disorders in pregnancy. Serious consideration is being given to intervening with the DASH diet and other nonpharmacologic interventions, she said.
While women with chronic hypertension prior to pregnancy are known to be at increased risk for development of preeclampsia and other hypertensive disorders of pregnancy as well as for gestational diabetes mellitus, the situation regarding women with prepregnancy prehypertension has been less clear. Dr. Black and coworkers shed new light on this issue in a separate study presented at the AHA meeting. She reported that women with prepregnancy prehypertension are also at increased risk for these pregnancy complications.
This study involved 2,156 Kaiser Permanente patients with prepregnancy prehypertension and 5,646 who were normotensive. A total of 8.4% of the women developed a hypertensive disorder during pregnancy, including 373 with preeclampsia/eclampsia. In addition, 1,877 women developed gestational diabetes mellitus.
Women with prehypertension prior to pregnancy were an adjusted 2.7-fold more likely to develop a hypertensive disorder in pregnancy, 2.2-fold more likely to have preeclampsia/eclampsia, and 1.2-fold more likely to develop gestational diabetes than women who were normotensive before pregnancy.
The risk of preeclampsia/eclampsia among women with prepregnancy prehypertension was significantly greater if they were at least 30 years old than if they were younger.
Both studies were funded by Kaiser Permanente. Dr. Black is an employee of the company.
DALLAS – Previously normotensive women who develop a hypertensive disorder during pregnancy are at more than double the risk of being diagnosed with prehypertension or hypertension within the first year after delivery, according to a large California study.
"Early postpartum screening and improved follow-up of women with hypertensive disorders first identified during pregnancy may be necessary to identify those at risk for future hypertension or prehypertension," Mary Helen Black, Ph.D., concluded in presenting the results of her population-based study at the American Heart Association scientific sessions.
The study was a retrospective review of the comprehensive electronic health records of 5,960 women who received their prenatal care and subsequently delivered a live singleton neonate at 20 weeks’ gestation or more at a single Kaiser Permanente Southern California medical center during 2006-2010. All of these women were specifically identified as being normotensive prior to their pregnancy and 6% of them developed a hypertensive disorder in pregnancy; 215 of the 358 affected women had preeclampsia or eclampsia.
During the first year after delivery, 63 women, or 1.1% of the total study population, developed frank hypertension. Another 902, or 15%, developed prehypertension during that first year. Prehypertension was defined as a modestly elevated blood pressure of at least 120/80 mm Hg on two consecutive ambulatory visits.
After adjustment for potential confounders including age, race, parity, prepregnancy body mass index, and smoking status, previously normotensive women with hypertension in pregnancy had an adjusted 2.4-fold increased risk of developing hypertension or prehypertension in the year after delivery, compared with women without any hypertensive disorder in pregnancy. Women who developed preeclampsia or eclampsia had a 2.5-fold increased risk, according to Dr. Black, a research scientist at Kaiser Permanente Southern California in Pasadena.
Protocols exist for monitoring and treatment of pregnant women identified as having chronic hypertension prior to pregnancy. However, the new Kaiser study is one of the first to look at the downstream impact of hypertensive disorders in pregnancy arising among women with no preexisting indication of hypertension. The plan is to follow these women further to learn how their clinical outcomes progress. Long-term Scandinavian registry studies have linked hypertensive disorders in pregnancy to increased risk of eventual type 2 diabetes, hypertension, and cardiovascular disease.
Dr. Black and her coinvestigators are planning a postpartum intervention study aimed at preventing onset of type 2 diabetes and cardiovascular disease in previously normotensive women with hypertensive disorders in pregnancy. Serious consideration is being given to intervening with the DASH diet and other nonpharmacologic interventions, she said.
While women with chronic hypertension prior to pregnancy are known to be at increased risk for development of preeclampsia and other hypertensive disorders of pregnancy as well as for gestational diabetes mellitus, the situation regarding women with prepregnancy prehypertension has been less clear. Dr. Black and coworkers shed new light on this issue in a separate study presented at the AHA meeting. She reported that women with prepregnancy prehypertension are also at increased risk for these pregnancy complications.
This study involved 2,156 Kaiser Permanente patients with prepregnancy prehypertension and 5,646 who were normotensive. A total of 8.4% of the women developed a hypertensive disorder during pregnancy, including 373 with preeclampsia/eclampsia. In addition, 1,877 women developed gestational diabetes mellitus.
Women with prehypertension prior to pregnancy were an adjusted 2.7-fold more likely to develop a hypertensive disorder in pregnancy, 2.2-fold more likely to have preeclampsia/eclampsia, and 1.2-fold more likely to develop gestational diabetes than women who were normotensive before pregnancy.
The risk of preeclampsia/eclampsia among women with prepregnancy prehypertension was significantly greater if they were at least 30 years old than if they were younger.
Both studies were funded by Kaiser Permanente. Dr. Black is an employee of the company.
AT THE AHA SCIENTIFIC SESSIONS
Major finding: Women who were normotensive prepregnancy but developed a hypertensive disorder during pregnancy were at 2.4-fold increased risk of frank hypertension or prehypertension in their first year after delivery, compared with women who remained normotensive in pregnancy.
Data source: An observational study of 5,960 women who delivered a singleton neonate, 6% of whom developed a hypertensive disorder of pregnancy. All were known to be normotensive prior to pregnancy.
Disclosures: Dr. Black is an employee of Kaiser Permanente, which funded the study.
How to foil post-CABG aspirin resistance
AMSTERDAM (IMNG) – Giving low-dose aspirin four times per day in the first days after coronary artery bypass graft surgery suppresses serum thromboxane levels far more effectively than does conventional once-daily dosing at 325 mg, according to a randomized trial.
The clinical implication of this finding is that more frequent dosing of aspirin may prevent the serious problem of premature vein graft failure from the development of aspirin resistance in the postoperative period, although at this point this is a hypothesis that requires testing in a future study, Dr. Jeremy S. Paikin said at the annual congress of the European Society of Cardiology.
He reported on 110 on-pump coronary artery bypass graft (CABG) patients randomized on postoperative day 1 to aspirin either at 81 mg four times daily, the standard 325 mg once daily, or to 81 mg once daily.
The primary study endpoint was the serum thromboxane level on the morning of postoperative day 4. The median level was 13.3 ng/mL in the group on aspirin at 81 mg once daily, 3.4 ng/mL with 325 mg once daily, and significantly lower at 1.1 ng/mL in patients on 81 mg four times daily.
"With 81 mg QD [four times daily], there’s almost complete suppression of serum thromboxane throughout the course of the hospital stay," according to Dr. Paikin of McMaster University, Hamilton, Ont.
Aspirin is known to prevent CABG graft failure, but its effectiveness is limited by the not-infrequent development of aspirin hyporesponsiveness in the postoperative period. The underlying mechanism involved in this aspirin resistance was previously unknown; however, in their randomized trial Dr. Paikin and coinvestigators established that the hyporesponsiveness is caused at least in part by increased platelet turnover in the postoperative period. The investigators showed that platelet turnover per day was increased two- to threefold in the week after CABG, compared with presurgical levels, a finding Dr. Paikin termed "quite exciting."
Recognizing that administration of any drug four times daily raises formidable adherence obstacles, he and his coworkers are just about to start a clinical trial looking at twice-daily aspirin dosing post CABG. They’re also interested in drawing a firm evidentiary connection between serum thromboxane levels and risk of premature graft failure.
Dr. Paikin reported having no financial conflicts of interest.
AMSTERDAM (IMNG) – Giving low-dose aspirin four times per day in the first days after coronary artery bypass graft surgery suppresses serum thromboxane levels far more effectively than does conventional once-daily dosing at 325 mg, according to a randomized trial.
The clinical implication of this finding is that more frequent dosing of aspirin may prevent the serious problem of premature vein graft failure from the development of aspirin resistance in the postoperative period, although at this point this is a hypothesis that requires testing in a future study, Dr. Jeremy S. Paikin said at the annual congress of the European Society of Cardiology.
He reported on 110 on-pump coronary artery bypass graft (CABG) patients randomized on postoperative day 1 to aspirin either at 81 mg four times daily, the standard 325 mg once daily, or to 81 mg once daily.
The primary study endpoint was the serum thromboxane level on the morning of postoperative day 4. The median level was 13.3 ng/mL in the group on aspirin at 81 mg once daily, 3.4 ng/mL with 325 mg once daily, and significantly lower at 1.1 ng/mL in patients on 81 mg four times daily.
"With 81 mg QD [four times daily], there’s almost complete suppression of serum thromboxane throughout the course of the hospital stay," according to Dr. Paikin of McMaster University, Hamilton, Ont.
Aspirin is known to prevent CABG graft failure, but its effectiveness is limited by the not-infrequent development of aspirin hyporesponsiveness in the postoperative period. The underlying mechanism involved in this aspirin resistance was previously unknown; however, in their randomized trial Dr. Paikin and coinvestigators established that the hyporesponsiveness is caused at least in part by increased platelet turnover in the postoperative period. The investigators showed that platelet turnover per day was increased two- to threefold in the week after CABG, compared with presurgical levels, a finding Dr. Paikin termed "quite exciting."
Recognizing that administration of any drug four times daily raises formidable adherence obstacles, he and his coworkers are just about to start a clinical trial looking at twice-daily aspirin dosing post CABG. They’re also interested in drawing a firm evidentiary connection between serum thromboxane levels and risk of premature graft failure.
Dr. Paikin reported having no financial conflicts of interest.
AMSTERDAM (IMNG) – Giving low-dose aspirin four times per day in the first days after coronary artery bypass graft surgery suppresses serum thromboxane levels far more effectively than does conventional once-daily dosing at 325 mg, according to a randomized trial.
The clinical implication of this finding is that more frequent dosing of aspirin may prevent the serious problem of premature vein graft failure from the development of aspirin resistance in the postoperative period, although at this point this is a hypothesis that requires testing in a future study, Dr. Jeremy S. Paikin said at the annual congress of the European Society of Cardiology.
He reported on 110 on-pump coronary artery bypass graft (CABG) patients randomized on postoperative day 1 to aspirin either at 81 mg four times daily, the standard 325 mg once daily, or to 81 mg once daily.
The primary study endpoint was the serum thromboxane level on the morning of postoperative day 4. The median level was 13.3 ng/mL in the group on aspirin at 81 mg once daily, 3.4 ng/mL with 325 mg once daily, and significantly lower at 1.1 ng/mL in patients on 81 mg four times daily.
"With 81 mg QD [four times daily], there’s almost complete suppression of serum thromboxane throughout the course of the hospital stay," according to Dr. Paikin of McMaster University, Hamilton, Ont.
Aspirin is known to prevent CABG graft failure, but its effectiveness is limited by the not-infrequent development of aspirin hyporesponsiveness in the postoperative period. The underlying mechanism involved in this aspirin resistance was previously unknown; however, in their randomized trial Dr. Paikin and coinvestigators established that the hyporesponsiveness is caused at least in part by increased platelet turnover in the postoperative period. The investigators showed that platelet turnover per day was increased two- to threefold in the week after CABG, compared with presurgical levels, a finding Dr. Paikin termed "quite exciting."
Recognizing that administration of any drug four times daily raises formidable adherence obstacles, he and his coworkers are just about to start a clinical trial looking at twice-daily aspirin dosing post CABG. They’re also interested in drawing a firm evidentiary connection between serum thromboxane levels and risk of premature graft failure.
Dr. Paikin reported having no financial conflicts of interest.
AT THE ESC CONGRESS 2013
Major finding: The median serum thromboxane level on the morning of post CABG day 4 was 13.3 ng/mL in the group on aspirin at 81 mg once daily, 3.4 ng/mL with 325 mg once daily, and significantly lower at 1.1 ng/mL in patients on 81 mg four times daily.
Data source: A randomized clinical trial in which 110 patients who underwent on-pump CABG surgery were randomized on postoperative day 1 to aspirin at either 81 mg four times daily, 325 mg once daily, or 81 mg once daily.
Disclosures: The study presenter reported having no financial conflicts.
Dopamine, nesiritide ineffective in acute heart failure with renal dysfunction
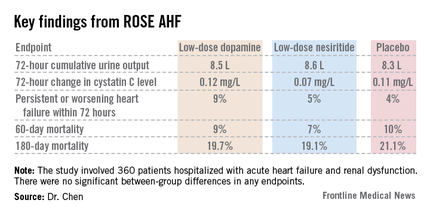
DALLAS – Neither low-dose dopamine nor low-dose nesiritide enhanced decongestion or improved renal function in patients with acute heart failure and kidney dysfunction in the randomized ROSE AHF trial.
Thus, the major unmet need for drugs that accomplish these benefits continues. There remains no Food and Drug Administration–approved therapy for enhancing renal function in acute heart failure, Dr. Horng H. Chen observed in presenting the ROSE AHF results at the American Heart Association scientific sessions.
The need for such therapies is great. Most patients with heart failure eventually develop renal dysfunction. If this dysfunction is moderate or severe, patients may experience inadequate decongestion in response to diuretic therapy along with worsening renal dysfunction, both linked to poor clinical outcomes.
Prior small, single-center studies with various methodologic shortcomings had suggested possible benefit for dopamine or nesiritide in low doses thought to be renal specific. The ROSE AHF (Renal Optimization Strategies Evaluation in Acute Heart Failure) trial was conducted to provide definitive evidence to guide practice, explained Dr. Chen, professor of medicine at the Mayo Clinic, Rochester, Minn.
ROSE AHF was a randomized, double-blind, placebo-controlled clinical trial involving 360 patients hospitalized with AHF and renal dysfunction at 26 U.S. and Canadian centers. The study was carried out by the National Heart, Lung, and Blood Institute (NHLBI) Heart Failure Clinical Research Network. Patients were randomized within 24 hours of admission to low-dose dopamine at 2 mcg/kg per minute, low-dose nesiritide at 0.005 mcg/kg per minute, or placebo.
The coprimary endpoints were 72-hour cumulative urine volume as a measure of decongestion and 72-hour change in serum cystatin C as an indicator of renal function.
Interestingly, the two drugs clearly were not truly renal specific, even in these low doses widely considered to be so, Dr. Chen noted. This lack of renal specificity was evident from the side effect profile. Significant tachycardia occurred in 7.2% of the low-dose dopamine group, none on nesiritide, and 10.4% of placebo-treated controls. In contrast, significant hypotension occurred in 0.9% of patients on dopamine, 18.8% on nesiritide, and 10.4% on placebo.
A trend toward a differential treatment response depending upon whether a patient had heart failure with a reduced versus preserved ejection fraction was evident. Patients with diastolic heart failure tended to do worse with dopamine than placebo in terms of 72-hour urine output, while those with systolic heart failure tended to do better with nesiritide than placebo. However, the trial was insufficiently powered to draw definitive conclusions on this score.
"One of our take-away messages when we looked at the data was that acute heart failure is broadly defined and includes a very diverse group of patients. With the suggestion that there may be a differential response based on ejection fraction, as we proceed with future heart failure studies we may consider targeting subsets of patients. Some therapies may benefit patients with reduced ejection fraction, others will benefit patients with preserved ejection fraction. We might want to power our studies to look at these subgroups," according to Dr. Chen.
Noting that current American Heart Association/American College of Cardiology guidelines for AHF management state that low-dose dopamine "may be considered" to improve diuresis and preserve renal function during diuretic therapy, the cardiologist said "the only conclusion we can make from our study is they should not be used routinely."
He added a caveat, however: "In my own practice, if I have a patient who is hypotensive and who hasn’t responded to increased doses of diuretics, I still may consider using low-dose dopamine at that stage" because of the reassuringly low 0.9% rate of hypotension in ROSE AHF.
Discussant Dr. Marco Metra of the University of Brescia (Italy) noted that low doses of dopamine and nesiritide are widely used in the United States and Europe. Recent data suggest nesiritide is used in 5%-10% of American patients with AHF and low-dose dopamine in 3%-5%.
"Surely this trial should reduce the use of these drugs in patients with acute heart failure. They are given routinely too often in some places," he said.
Discussant Marc Pfeffer called ROSE AHF "yet another example of shedding the light of the randomized controlled trial on things that we assume to be true but actually aren’t. And we probably spend more time on rounds talking about [improving decongestion in patients with AHF and renal dysfunction] than anything else," added Dr. Pfeffer, professor of medicine at Harvard University, Boston.
The ROSE AHF trial was sponsored by the NHLBI. Dr. Chen reported receiving research grants from Scios and royalties from Niles Therapeutics, Anexon, and UpToDate. Dr. Metra is a paid consultant to a handful of pharmaceutical companies.
DALLAS – Neither low-dose dopamine nor low-dose nesiritide enhanced decongestion or improved renal function in patients with acute heart failure and kidney dysfunction in the randomized ROSE AHF trial.
Thus, the major unmet need for drugs that accomplish these benefits continues. There remains no Food and Drug Administration–approved therapy for enhancing renal function in acute heart failure, Dr. Horng H. Chen observed in presenting the ROSE AHF results at the American Heart Association scientific sessions.

The need for such therapies is great. Most patients with heart failure eventually develop renal dysfunction. If this dysfunction is moderate or severe, patients may experience inadequate decongestion in response to diuretic therapy along with worsening renal dysfunction, both linked to poor clinical outcomes.
Prior small, single-center studies with various methodologic shortcomings had suggested possible benefit for dopamine or nesiritide in low doses thought to be renal specific. The ROSE AHF (Renal Optimization Strategies Evaluation in Acute Heart Failure) trial was conducted to provide definitive evidence to guide practice, explained Dr. Chen, professor of medicine at the Mayo Clinic, Rochester, Minn.
ROSE AHF was a randomized, double-blind, placebo-controlled clinical trial involving 360 patients hospitalized with AHF and renal dysfunction at 26 U.S. and Canadian centers. The study was carried out by the National Heart, Lung, and Blood Institute (NHLBI) Heart Failure Clinical Research Network. Patients were randomized within 24 hours of admission to low-dose dopamine at 2 mcg/kg per minute, low-dose nesiritide at 0.005 mcg/kg per minute, or placebo.
The coprimary endpoints were 72-hour cumulative urine volume as a measure of decongestion and 72-hour change in serum cystatin C as an indicator of renal function.
Interestingly, the two drugs clearly were not truly renal specific, even in these low doses widely considered to be so, Dr. Chen noted. This lack of renal specificity was evident from the side effect profile. Significant tachycardia occurred in 7.2% of the low-dose dopamine group, none on nesiritide, and 10.4% of placebo-treated controls. In contrast, significant hypotension occurred in 0.9% of patients on dopamine, 18.8% on nesiritide, and 10.4% on placebo.
A trend toward a differential treatment response depending upon whether a patient had heart failure with a reduced versus preserved ejection fraction was evident. Patients with diastolic heart failure tended to do worse with dopamine than placebo in terms of 72-hour urine output, while those with systolic heart failure tended to do better with nesiritide than placebo. However, the trial was insufficiently powered to draw definitive conclusions on this score.
"One of our take-away messages when we looked at the data was that acute heart failure is broadly defined and includes a very diverse group of patients. With the suggestion that there may be a differential response based on ejection fraction, as we proceed with future heart failure studies we may consider targeting subsets of patients. Some therapies may benefit patients with reduced ejection fraction, others will benefit patients with preserved ejection fraction. We might want to power our studies to look at these subgroups," according to Dr. Chen.
Noting that current American Heart Association/American College of Cardiology guidelines for AHF management state that low-dose dopamine "may be considered" to improve diuresis and preserve renal function during diuretic therapy, the cardiologist said "the only conclusion we can make from our study is they should not be used routinely."
He added a caveat, however: "In my own practice, if I have a patient who is hypotensive and who hasn’t responded to increased doses of diuretics, I still may consider using low-dose dopamine at that stage" because of the reassuringly low 0.9% rate of hypotension in ROSE AHF.
Discussant Dr. Marco Metra of the University of Brescia (Italy) noted that low doses of dopamine and nesiritide are widely used in the United States and Europe. Recent data suggest nesiritide is used in 5%-10% of American patients with AHF and low-dose dopamine in 3%-5%.
"Surely this trial should reduce the use of these drugs in patients with acute heart failure. They are given routinely too often in some places," he said.
Discussant Marc Pfeffer called ROSE AHF "yet another example of shedding the light of the randomized controlled trial on things that we assume to be true but actually aren’t. And we probably spend more time on rounds talking about [improving decongestion in patients with AHF and renal dysfunction] than anything else," added Dr. Pfeffer, professor of medicine at Harvard University, Boston.
The ROSE AHF trial was sponsored by the NHLBI. Dr. Chen reported receiving research grants from Scios and royalties from Niles Therapeutics, Anexon, and UpToDate. Dr. Metra is a paid consultant to a handful of pharmaceutical companies.
DALLAS – Neither low-dose dopamine nor low-dose nesiritide enhanced decongestion or improved renal function in patients with acute heart failure and kidney dysfunction in the randomized ROSE AHF trial.
Thus, the major unmet need for drugs that accomplish these benefits continues. There remains no Food and Drug Administration–approved therapy for enhancing renal function in acute heart failure, Dr. Horng H. Chen observed in presenting the ROSE AHF results at the American Heart Association scientific sessions.

The need for such therapies is great. Most patients with heart failure eventually develop renal dysfunction. If this dysfunction is moderate or severe, patients may experience inadequate decongestion in response to diuretic therapy along with worsening renal dysfunction, both linked to poor clinical outcomes.
Prior small, single-center studies with various methodologic shortcomings had suggested possible benefit for dopamine or nesiritide in low doses thought to be renal specific. The ROSE AHF (Renal Optimization Strategies Evaluation in Acute Heart Failure) trial was conducted to provide definitive evidence to guide practice, explained Dr. Chen, professor of medicine at the Mayo Clinic, Rochester, Minn.
ROSE AHF was a randomized, double-blind, placebo-controlled clinical trial involving 360 patients hospitalized with AHF and renal dysfunction at 26 U.S. and Canadian centers. The study was carried out by the National Heart, Lung, and Blood Institute (NHLBI) Heart Failure Clinical Research Network. Patients were randomized within 24 hours of admission to low-dose dopamine at 2 mcg/kg per minute, low-dose nesiritide at 0.005 mcg/kg per minute, or placebo.
The coprimary endpoints were 72-hour cumulative urine volume as a measure of decongestion and 72-hour change in serum cystatin C as an indicator of renal function.
Interestingly, the two drugs clearly were not truly renal specific, even in these low doses widely considered to be so, Dr. Chen noted. This lack of renal specificity was evident from the side effect profile. Significant tachycardia occurred in 7.2% of the low-dose dopamine group, none on nesiritide, and 10.4% of placebo-treated controls. In contrast, significant hypotension occurred in 0.9% of patients on dopamine, 18.8% on nesiritide, and 10.4% on placebo.
A trend toward a differential treatment response depending upon whether a patient had heart failure with a reduced versus preserved ejection fraction was evident. Patients with diastolic heart failure tended to do worse with dopamine than placebo in terms of 72-hour urine output, while those with systolic heart failure tended to do better with nesiritide than placebo. However, the trial was insufficiently powered to draw definitive conclusions on this score.
"One of our take-away messages when we looked at the data was that acute heart failure is broadly defined and includes a very diverse group of patients. With the suggestion that there may be a differential response based on ejection fraction, as we proceed with future heart failure studies we may consider targeting subsets of patients. Some therapies may benefit patients with reduced ejection fraction, others will benefit patients with preserved ejection fraction. We might want to power our studies to look at these subgroups," according to Dr. Chen.
Noting that current American Heart Association/American College of Cardiology guidelines for AHF management state that low-dose dopamine "may be considered" to improve diuresis and preserve renal function during diuretic therapy, the cardiologist said "the only conclusion we can make from our study is they should not be used routinely."
He added a caveat, however: "In my own practice, if I have a patient who is hypotensive and who hasn’t responded to increased doses of diuretics, I still may consider using low-dose dopamine at that stage" because of the reassuringly low 0.9% rate of hypotension in ROSE AHF.
Discussant Dr. Marco Metra of the University of Brescia (Italy) noted that low doses of dopamine and nesiritide are widely used in the United States and Europe. Recent data suggest nesiritide is used in 5%-10% of American patients with AHF and low-dose dopamine in 3%-5%.
"Surely this trial should reduce the use of these drugs in patients with acute heart failure. They are given routinely too often in some places," he said.
Discussant Marc Pfeffer called ROSE AHF "yet another example of shedding the light of the randomized controlled trial on things that we assume to be true but actually aren’t. And we probably spend more time on rounds talking about [improving decongestion in patients with AHF and renal dysfunction] than anything else," added Dr. Pfeffer, professor of medicine at Harvard University, Boston.
The ROSE AHF trial was sponsored by the NHLBI. Dr. Chen reported receiving research grants from Scios and royalties from Niles Therapeutics, Anexon, and UpToDate. Dr. Metra is a paid consultant to a handful of pharmaceutical companies.
AT THE AHA SCIENTIFIC SESSIONS
Major finding: Neither low-dose dopamine nor low-dose nesiritide improved decongestion or boosted renal function when added to diuretic therapy in patients hospitalized with acute heart failure and kidney dysfunction.
Data source: ROSE AHF, a multicenter, randomized, double-blind, placebo-controlled clinical trial of 360 patients with acute heart failure and renal dysfunction.
Disclosures. The trial was sponsored by the National Heart, Lung, and Blood Institute. Dr. Chen reported receiving research grants from Scios and royalties from Niles Therapeutics, Anexon, and UpToDate. Dr. Metra is a paid consultant to a handful of pharmaceutical companies.
Chelation plus vitamins halved cardiovascular events in diabetics
DALLAS – An EDTA-based chelation regimen for secondary prevention of cardiovascular events in patients with a history of myocardial infarction got a boost from a new TACT trial analysis showing additive benefit when chelation was accompanied by high-dose oral multivitamins.
"The benefit of chelation plus vitamins compared to placebo plus placebo is statistically significant and of a magnitude sufficient to be clinically important, with a number needed to treat of 12 to prevent one primary event over 5 years," Dr. Gervasio A. Lamas said in presenting the latest TACT (Trial to Assess Chelation Therapy) results at the American Heart Association scientific sessions.
Moreover, the benefit of chelation plus multivitamins was magnified in the more than 600 TACT participants with diabetes. The number needed to treat in that group was an impressively low 5.5, added Dr. Lamas, chairman of medicine at Mount Sinai Medical Center, Miami Beach, and chief of the Columbia University division of cardiology at Mount Sinai Medical Center.
Since 1956, EDTA (ethylenediaminetetraacetic acid) chelation has been utilized in complementary and alternative medical (CAM) practice to treat atherosclerotic disease, despite an absence of any supporting evidence. The TACT trial, sponsored by the National Institutes of Health, was conducted in order to put the CAM regimen to the test.
TACT was a rigorously conducted, randomized, double-blind, two-by-two factorial design trial in which 1,708 stable patients with a previous MI at 134 North American sites were placed on the intravenous chelation regimen or placebo and high-dose oral multivitamins or placebo. It was an arduous regimen designed to replicate what’s being used in CAM practice. The chelation regimen consisted of 30 weekly 500-cc intravenous infusions followed by another 10 infusions at 2- to 8-week intervals. Patients randomized to the multivitamin arm took 6 capsules per day.
"The capsules are large. They’re a bear to take," according to the cardiologist.
Nevertheless, 77% of patients completed 30 infusions and 65% completed all 40. And more than three-quarters of patients took the vitamins for at least a year, and half of participants did so for at least 3 years. All participants had high rates of guideline-recommended preventive medications usage.
The primary composite endpoint was the 5-year rate of all-cause mortality, MI, stroke, coronary revascularization, or hospitalization for angina. As previously reported, chelation therapy resulted in a statistically significant 18% reduction in the primary endpoint relative to placebo (JAMA 2013;309:1241-50), while high-dose multivitamin therapy led to a nonsignificant 11% reduction (Ann. Intern. Med., in press).
At the Dallas AHA meeting, Dr. Lamas focused on the results in the 421 subjects randomized to both chelation and multivitamins as compared to the 437 patients on double placebo. The primary composite endpoint occurred in 26% of patients on double active therapy and 32% on double placebo, representing a 26% reduction in relative risk. Thus, the addition of multivitamin therapy increased the magnitude of risk reduction in patients on chelation therapy from 18% to 26%.
The prespecified secondary ‘hard’ endpoint, a composite of cardiovascular death, recurrent MI, or stroke, occurred in 9% of participants on chelation plus multivitamins versus 13% of those on dual placebo, for a 34% reduction in risk.
The results of active therapy were even more impressive in patients with diabetes. They had a 41% reduction in risk of the primary composite endpoint with chelation alone compared with placebo, with a 5-year incidence of 25%, compared with 38% in controls. Notably, diabetic patients’ all-cause mortality with chelation therapy was 10%, compared with 16% with placebo, a 43% reduction in relative risk, while their rate of the secondary composite hard endpoint was 11%, versus 17% in controls.
The diabetic subgroup assigned to dual active therapy with both chelation and multivitamins fared even better, with a 51% decrease in the primary composite endpoint compared with controls on double placebo.
"A lot more work needs to be done on this before we can bring it to the bedside, but this is certainly suggestive data," Dr. Lamas said in summary.
Specific details on the chelation and multivitamin components of the regimen are available in an earlier publication (Am. Heart J. 2012;163:7-12).
Asked about the proposed mechanism of benefit of this CAM therapy, the cardiologist was quick to reply, "The simple answer is we do not really know what is happening."
Plausible hypotheses abound, though. One of the leading ones has to do with the fact that EDTA is a superb metal chelator.
"Heavy metals are associated very well in epidemiologic data with cardiovascular events; in particular, lead, cadmium, arsenic, sometimes mercury, and others that have less evidence, like tungsten and antimony. They’re all in our environment. Any of us who are of an age to have been exposed to leaded gasoline have lead in our bones. If we get an infusion of EDTA, we’ll have lead in our urine. It’s just the way it is. And as you get older and become osteoporotic, that lead starts getting released," Dr. Lamas explained.
In addition, in diabetic patients the formation of advanced glycation endpoints requires catalytic activity by metals in order to create oxygen species and cross linkage, he continued.
The TACT trial was sponsored by the National Center for Complementary and Alternative Medicine and the National Heart, Lung, and Blood Institute. Dr. Lamas reported having no relevant financial interests.
DALLAS – An EDTA-based chelation regimen for secondary prevention of cardiovascular events in patients with a history of myocardial infarction got a boost from a new TACT trial analysis showing additive benefit when chelation was accompanied by high-dose oral multivitamins.
"The benefit of chelation plus vitamins compared to placebo plus placebo is statistically significant and of a magnitude sufficient to be clinically important, with a number needed to treat of 12 to prevent one primary event over 5 years," Dr. Gervasio A. Lamas said in presenting the latest TACT (Trial to Assess Chelation Therapy) results at the American Heart Association scientific sessions.
Moreover, the benefit of chelation plus multivitamins was magnified in the more than 600 TACT participants with diabetes. The number needed to treat in that group was an impressively low 5.5, added Dr. Lamas, chairman of medicine at Mount Sinai Medical Center, Miami Beach, and chief of the Columbia University division of cardiology at Mount Sinai Medical Center.
Since 1956, EDTA (ethylenediaminetetraacetic acid) chelation has been utilized in complementary and alternative medical (CAM) practice to treat atherosclerotic disease, despite an absence of any supporting evidence. The TACT trial, sponsored by the National Institutes of Health, was conducted in order to put the CAM regimen to the test.
TACT was a rigorously conducted, randomized, double-blind, two-by-two factorial design trial in which 1,708 stable patients with a previous MI at 134 North American sites were placed on the intravenous chelation regimen or placebo and high-dose oral multivitamins or placebo. It was an arduous regimen designed to replicate what’s being used in CAM practice. The chelation regimen consisted of 30 weekly 500-cc intravenous infusions followed by another 10 infusions at 2- to 8-week intervals. Patients randomized to the multivitamin arm took 6 capsules per day.
"The capsules are large. They’re a bear to take," according to the cardiologist.
Nevertheless, 77% of patients completed 30 infusions and 65% completed all 40. And more than three-quarters of patients took the vitamins for at least a year, and half of participants did so for at least 3 years. All participants had high rates of guideline-recommended preventive medications usage.
The primary composite endpoint was the 5-year rate of all-cause mortality, MI, stroke, coronary revascularization, or hospitalization for angina. As previously reported, chelation therapy resulted in a statistically significant 18% reduction in the primary endpoint relative to placebo (JAMA 2013;309:1241-50), while high-dose multivitamin therapy led to a nonsignificant 11% reduction (Ann. Intern. Med., in press).
At the Dallas AHA meeting, Dr. Lamas focused on the results in the 421 subjects randomized to both chelation and multivitamins as compared to the 437 patients on double placebo. The primary composite endpoint occurred in 26% of patients on double active therapy and 32% on double placebo, representing a 26% reduction in relative risk. Thus, the addition of multivitamin therapy increased the magnitude of risk reduction in patients on chelation therapy from 18% to 26%.
The prespecified secondary ‘hard’ endpoint, a composite of cardiovascular death, recurrent MI, or stroke, occurred in 9% of participants on chelation plus multivitamins versus 13% of those on dual placebo, for a 34% reduction in risk.
The results of active therapy were even more impressive in patients with diabetes. They had a 41% reduction in risk of the primary composite endpoint with chelation alone compared with placebo, with a 5-year incidence of 25%, compared with 38% in controls. Notably, diabetic patients’ all-cause mortality with chelation therapy was 10%, compared with 16% with placebo, a 43% reduction in relative risk, while their rate of the secondary composite hard endpoint was 11%, versus 17% in controls.
The diabetic subgroup assigned to dual active therapy with both chelation and multivitamins fared even better, with a 51% decrease in the primary composite endpoint compared with controls on double placebo.
"A lot more work needs to be done on this before we can bring it to the bedside, but this is certainly suggestive data," Dr. Lamas said in summary.
Specific details on the chelation and multivitamin components of the regimen are available in an earlier publication (Am. Heart J. 2012;163:7-12).
Asked about the proposed mechanism of benefit of this CAM therapy, the cardiologist was quick to reply, "The simple answer is we do not really know what is happening."
Plausible hypotheses abound, though. One of the leading ones has to do with the fact that EDTA is a superb metal chelator.
"Heavy metals are associated very well in epidemiologic data with cardiovascular events; in particular, lead, cadmium, arsenic, sometimes mercury, and others that have less evidence, like tungsten and antimony. They’re all in our environment. Any of us who are of an age to have been exposed to leaded gasoline have lead in our bones. If we get an infusion of EDTA, we’ll have lead in our urine. It’s just the way it is. And as you get older and become osteoporotic, that lead starts getting released," Dr. Lamas explained.
In addition, in diabetic patients the formation of advanced glycation endpoints requires catalytic activity by metals in order to create oxygen species and cross linkage, he continued.
The TACT trial was sponsored by the National Center for Complementary and Alternative Medicine and the National Heart, Lung, and Blood Institute. Dr. Lamas reported having no relevant financial interests.
DALLAS – An EDTA-based chelation regimen for secondary prevention of cardiovascular events in patients with a history of myocardial infarction got a boost from a new TACT trial analysis showing additive benefit when chelation was accompanied by high-dose oral multivitamins.
"The benefit of chelation plus vitamins compared to placebo plus placebo is statistically significant and of a magnitude sufficient to be clinically important, with a number needed to treat of 12 to prevent one primary event over 5 years," Dr. Gervasio A. Lamas said in presenting the latest TACT (Trial to Assess Chelation Therapy) results at the American Heart Association scientific sessions.
Moreover, the benefit of chelation plus multivitamins was magnified in the more than 600 TACT participants with diabetes. The number needed to treat in that group was an impressively low 5.5, added Dr. Lamas, chairman of medicine at Mount Sinai Medical Center, Miami Beach, and chief of the Columbia University division of cardiology at Mount Sinai Medical Center.
Since 1956, EDTA (ethylenediaminetetraacetic acid) chelation has been utilized in complementary and alternative medical (CAM) practice to treat atherosclerotic disease, despite an absence of any supporting evidence. The TACT trial, sponsored by the National Institutes of Health, was conducted in order to put the CAM regimen to the test.
TACT was a rigorously conducted, randomized, double-blind, two-by-two factorial design trial in which 1,708 stable patients with a previous MI at 134 North American sites were placed on the intravenous chelation regimen or placebo and high-dose oral multivitamins or placebo. It was an arduous regimen designed to replicate what’s being used in CAM practice. The chelation regimen consisted of 30 weekly 500-cc intravenous infusions followed by another 10 infusions at 2- to 8-week intervals. Patients randomized to the multivitamin arm took 6 capsules per day.
"The capsules are large. They’re a bear to take," according to the cardiologist.
Nevertheless, 77% of patients completed 30 infusions and 65% completed all 40. And more than three-quarters of patients took the vitamins for at least a year, and half of participants did so for at least 3 years. All participants had high rates of guideline-recommended preventive medications usage.
The primary composite endpoint was the 5-year rate of all-cause mortality, MI, stroke, coronary revascularization, or hospitalization for angina. As previously reported, chelation therapy resulted in a statistically significant 18% reduction in the primary endpoint relative to placebo (JAMA 2013;309:1241-50), while high-dose multivitamin therapy led to a nonsignificant 11% reduction (Ann. Intern. Med., in press).
At the Dallas AHA meeting, Dr. Lamas focused on the results in the 421 subjects randomized to both chelation and multivitamins as compared to the 437 patients on double placebo. The primary composite endpoint occurred in 26% of patients on double active therapy and 32% on double placebo, representing a 26% reduction in relative risk. Thus, the addition of multivitamin therapy increased the magnitude of risk reduction in patients on chelation therapy from 18% to 26%.
The prespecified secondary ‘hard’ endpoint, a composite of cardiovascular death, recurrent MI, or stroke, occurred in 9% of participants on chelation plus multivitamins versus 13% of those on dual placebo, for a 34% reduction in risk.
The results of active therapy were even more impressive in patients with diabetes. They had a 41% reduction in risk of the primary composite endpoint with chelation alone compared with placebo, with a 5-year incidence of 25%, compared with 38% in controls. Notably, diabetic patients’ all-cause mortality with chelation therapy was 10%, compared with 16% with placebo, a 43% reduction in relative risk, while their rate of the secondary composite hard endpoint was 11%, versus 17% in controls.
The diabetic subgroup assigned to dual active therapy with both chelation and multivitamins fared even better, with a 51% decrease in the primary composite endpoint compared with controls on double placebo.
"A lot more work needs to be done on this before we can bring it to the bedside, but this is certainly suggestive data," Dr. Lamas said in summary.
Specific details on the chelation and multivitamin components of the regimen are available in an earlier publication (Am. Heart J. 2012;163:7-12).
Asked about the proposed mechanism of benefit of this CAM therapy, the cardiologist was quick to reply, "The simple answer is we do not really know what is happening."
Plausible hypotheses abound, though. One of the leading ones has to do with the fact that EDTA is a superb metal chelator.
"Heavy metals are associated very well in epidemiologic data with cardiovascular events; in particular, lead, cadmium, arsenic, sometimes mercury, and others that have less evidence, like tungsten and antimony. They’re all in our environment. Any of us who are of an age to have been exposed to leaded gasoline have lead in our bones. If we get an infusion of EDTA, we’ll have lead in our urine. It’s just the way it is. And as you get older and become osteoporotic, that lead starts getting released," Dr. Lamas explained.
In addition, in diabetic patients the formation of advanced glycation endpoints requires catalytic activity by metals in order to create oxygen species and cross linkage, he continued.
The TACT trial was sponsored by the National Center for Complementary and Alternative Medicine and the National Heart, Lung, and Blood Institute. Dr. Lamas reported having no relevant financial interests.
AT THE AHA SCIENTIFIC SESSIONS
Major finding: A mere 5.5 diabetic patients with a history of MI needed to be treated with a regimen of EDTA chelation plus high-dose multivitamins in order to prevent 1 additional cardiovascular event over 5 years in a large randomized trial.
Data source: The TACT trial was a double-blind, placebo-controlled, multicenter study with a two-by-two factorial design, involving 1,708 patients with a history of MI.
Disclosures. The TACT trial was sponsored by the National Center for Complementary and Alternative Medicine and the National Heart, Lung, and Blood Institute. Dr. Lamas reported having no relevant financial interests.
Naltrexone, hyperbaric oxygen show promise for fibromyalgia
SAN DIEGO – Low-dose naltrexone and repeated visits to the hyperbaric oxygen chamber are two novel potential therapies for fibromyalgia that showed promise in separate studies presented at the annual meeting of the American College of Rheumatology.
The impetus for investigating these unconventional treatments lies in recognition that fibromyalgia is challenging to treat. The Food and Drug Administration–approved medications for this common condition – duloxetine (Cymbalta), pregabalin (Lyrica), and milnacipran (Savella) – often provide inadequate results.
Naltrexone is an opioid receptor antagonist indicated for the treatment of alcohol or opioid dependence. Dr. Daniel G. Arkfeld explained the therapeutic rationale for its use in fibromyalgia thusly: Fibromyalgia has been classified as a CNS sensitization syndrome mediated by proinflammatory cytokines, and naltrexone has been shown to suppress such centrally acting cytokines.
He presented a prospective, open-label, single-center, uncontrolled pilot study involving 25 patients with fibromyalgia diagnosed by ACR criteria. They were started on oral naltrexone at 3 mg at night, with titration up to 4.5 mg. Patients were permitted to continue on their FDA-approved medications for fibromyalgia, and 18 of the 25 did so. The other 7 patients were on naltrexone monotherapy for the study duration.
Twenty-two patients completed the 3-month study. Two dropped out because they deemed naltrexone to be ineffective and one patient quit because of diarrhea.
The primary study endpoint was change in the Revised Fibromyalgia Impact Questionnaire (FIQR) after 90 days on naltrexone. Overall, study participants improved their FIQR scores by an average of 19.5%. However, 11 of the 22 study completers displayed a more robust response, with an average 41% improvement in the metric, according to Dr. Arkfeld, a rheumatologist at the University of Southern California, Los Angeles.
"Naltrexone is dosed once daily and is inexpensive, making it a viable treatment in fibromyalgia," he concluded.
Controlled clinical trials are planned.
Separately, Dr. Shai Efrati presented a randomized, prospective, controlled clinical trial of hyperbaric oxygen therapy in 60 women with fibromyalgia of at least 2 years duration. The therapeutic rationale lies in an earlier randomized trial by Dr. Efrati and coworkers demonstrating that hyperbaric oxygen therapy induced late neuroplasticity in post-stroke patients (PLoS One 2013; 8:e53716).
Thirty fibromyalgia patients commenced an 8-week course of hyperbaric oxygen therapy consisting of five 90-minute sessions per week involving 100% oxygen at 2 atmospheres absolute. After an 8-week hiatus with no treatment, the other 30 patients embarked on the same protocol.
The baseline tender joint count was 17 out of a possible 18 in the first 30 patients; after completion of the 40 treatment sessions, the tender joint count dropped to 9. Similarly, in the second 30-patient cohort, the baseline tender joint count was 17. It remained at 17 at the end of the no-treatment period and dropped to 5 after the course of hyperbaric oxygen therapy. Scores on the FIQR also dropped significantly from baseline to post treatment in both cohorts, reported Dr. Efrati of Assaf Harofeh Medical Center, Zerifin, Israel.
Dr. Arkfeld and Dr. Efrati reported having no financial conflicts of interest regarding their studies, which were free of commercial sponsorship.
SAN DIEGO – Low-dose naltrexone and repeated visits to the hyperbaric oxygen chamber are two novel potential therapies for fibromyalgia that showed promise in separate studies presented at the annual meeting of the American College of Rheumatology.
The impetus for investigating these unconventional treatments lies in recognition that fibromyalgia is challenging to treat. The Food and Drug Administration–approved medications for this common condition – duloxetine (Cymbalta), pregabalin (Lyrica), and milnacipran (Savella) – often provide inadequate results.
Naltrexone is an opioid receptor antagonist indicated for the treatment of alcohol or opioid dependence. Dr. Daniel G. Arkfeld explained the therapeutic rationale for its use in fibromyalgia thusly: Fibromyalgia has been classified as a CNS sensitization syndrome mediated by proinflammatory cytokines, and naltrexone has been shown to suppress such centrally acting cytokines.
He presented a prospective, open-label, single-center, uncontrolled pilot study involving 25 patients with fibromyalgia diagnosed by ACR criteria. They were started on oral naltrexone at 3 mg at night, with titration up to 4.5 mg. Patients were permitted to continue on their FDA-approved medications for fibromyalgia, and 18 of the 25 did so. The other 7 patients were on naltrexone monotherapy for the study duration.
Twenty-two patients completed the 3-month study. Two dropped out because they deemed naltrexone to be ineffective and one patient quit because of diarrhea.
The primary study endpoint was change in the Revised Fibromyalgia Impact Questionnaire (FIQR) after 90 days on naltrexone. Overall, study participants improved their FIQR scores by an average of 19.5%. However, 11 of the 22 study completers displayed a more robust response, with an average 41% improvement in the metric, according to Dr. Arkfeld, a rheumatologist at the University of Southern California, Los Angeles.
"Naltrexone is dosed once daily and is inexpensive, making it a viable treatment in fibromyalgia," he concluded.
Controlled clinical trials are planned.
Separately, Dr. Shai Efrati presented a randomized, prospective, controlled clinical trial of hyperbaric oxygen therapy in 60 women with fibromyalgia of at least 2 years duration. The therapeutic rationale lies in an earlier randomized trial by Dr. Efrati and coworkers demonstrating that hyperbaric oxygen therapy induced late neuroplasticity in post-stroke patients (PLoS One 2013; 8:e53716).
Thirty fibromyalgia patients commenced an 8-week course of hyperbaric oxygen therapy consisting of five 90-minute sessions per week involving 100% oxygen at 2 atmospheres absolute. After an 8-week hiatus with no treatment, the other 30 patients embarked on the same protocol.
The baseline tender joint count was 17 out of a possible 18 in the first 30 patients; after completion of the 40 treatment sessions, the tender joint count dropped to 9. Similarly, in the second 30-patient cohort, the baseline tender joint count was 17. It remained at 17 at the end of the no-treatment period and dropped to 5 after the course of hyperbaric oxygen therapy. Scores on the FIQR also dropped significantly from baseline to post treatment in both cohorts, reported Dr. Efrati of Assaf Harofeh Medical Center, Zerifin, Israel.
Dr. Arkfeld and Dr. Efrati reported having no financial conflicts of interest regarding their studies, which were free of commercial sponsorship.
SAN DIEGO – Low-dose naltrexone and repeated visits to the hyperbaric oxygen chamber are two novel potential therapies for fibromyalgia that showed promise in separate studies presented at the annual meeting of the American College of Rheumatology.
The impetus for investigating these unconventional treatments lies in recognition that fibromyalgia is challenging to treat. The Food and Drug Administration–approved medications for this common condition – duloxetine (Cymbalta), pregabalin (Lyrica), and milnacipran (Savella) – often provide inadequate results.
Naltrexone is an opioid receptor antagonist indicated for the treatment of alcohol or opioid dependence. Dr. Daniel G. Arkfeld explained the therapeutic rationale for its use in fibromyalgia thusly: Fibromyalgia has been classified as a CNS sensitization syndrome mediated by proinflammatory cytokines, and naltrexone has been shown to suppress such centrally acting cytokines.
He presented a prospective, open-label, single-center, uncontrolled pilot study involving 25 patients with fibromyalgia diagnosed by ACR criteria. They were started on oral naltrexone at 3 mg at night, with titration up to 4.5 mg. Patients were permitted to continue on their FDA-approved medications for fibromyalgia, and 18 of the 25 did so. The other 7 patients were on naltrexone monotherapy for the study duration.
Twenty-two patients completed the 3-month study. Two dropped out because they deemed naltrexone to be ineffective and one patient quit because of diarrhea.
The primary study endpoint was change in the Revised Fibromyalgia Impact Questionnaire (FIQR) after 90 days on naltrexone. Overall, study participants improved their FIQR scores by an average of 19.5%. However, 11 of the 22 study completers displayed a more robust response, with an average 41% improvement in the metric, according to Dr. Arkfeld, a rheumatologist at the University of Southern California, Los Angeles.
"Naltrexone is dosed once daily and is inexpensive, making it a viable treatment in fibromyalgia," he concluded.
Controlled clinical trials are planned.
Separately, Dr. Shai Efrati presented a randomized, prospective, controlled clinical trial of hyperbaric oxygen therapy in 60 women with fibromyalgia of at least 2 years duration. The therapeutic rationale lies in an earlier randomized trial by Dr. Efrati and coworkers demonstrating that hyperbaric oxygen therapy induced late neuroplasticity in post-stroke patients (PLoS One 2013; 8:e53716).
Thirty fibromyalgia patients commenced an 8-week course of hyperbaric oxygen therapy consisting of five 90-minute sessions per week involving 100% oxygen at 2 atmospheres absolute. After an 8-week hiatus with no treatment, the other 30 patients embarked on the same protocol.
The baseline tender joint count was 17 out of a possible 18 in the first 30 patients; after completion of the 40 treatment sessions, the tender joint count dropped to 9. Similarly, in the second 30-patient cohort, the baseline tender joint count was 17. It remained at 17 at the end of the no-treatment period and dropped to 5 after the course of hyperbaric oxygen therapy. Scores on the FIQR also dropped significantly from baseline to post treatment in both cohorts, reported Dr. Efrati of Assaf Harofeh Medical Center, Zerifin, Israel.
Dr. Arkfeld and Dr. Efrati reported having no financial conflicts of interest regarding their studies, which were free of commercial sponsorship.
AT THE ACR ANNUAL MEETING
Major finding: Fibromyalgia patients experienced an average 19.5% reduction in Revised Fibromyalgia Impact Questionnaire scores from baseline after 3 months on low-dose naltrexone. Half of the patients were high-end responders, with an average 41% reduction in scores.
Data source: A prospective, open-label, single-center study involving 25 patients with fibromyalgia.
Disclosures: The presenter reported having no financial conflicts with regard to the study, which was free of commercial support.
New international fibromyalgia guidelines indicate shifting focus
SAN DIEGO – The high degree of consistency among recent national fibromyalgia guidelines developed independently by multispecialty panels in Canada, Israel, and Germany suggests big changes are afoot in how this common and vexing syndrome is conceptualized and treated, according to Dr. Jacob N. Ablin.
"I hope to convey the feeling that there is somewhat of a paradigm change in the recommendations regarding treatment of fibromyalgia as expressed by these three guidelines. All three emphasize an individually tailored approach based upon the key symptoms and severity, with nonpharmacologic therapies as the major positive first choice for all. The emphasis is on the necessity of self-management strategies, which include aerobic exercise, cognitive-behavioral therapy, and multicomponent exercise and psychologic therapies," he said at the annual meeting of the American College of Rheumatology.
"Pharmacologic therapies were less enthusiastically recommended by all three groups. Contrary to popular perception, the drugs actually achieve only relatively modest effects. And all three groups caution about the side effects of drugs, which may mimic fibromyalgia symptoms," added Dr. Ablin of the Tel Aviv Sourasky Medical Center.
The three medications approved by the Food and Drug Administration for the treatment of fibromyalgia – pregabalin (Lyrica), duloxetine (Cymbalta), and milnacipran (Savella) – received only a weak grade C recommendation in the German guidelines (Schmerz 2012;26:287-90) because all three failed to achieve their primary endpoints in pivotal European clinical trials.
"While drug treatments absolutely continue to play a role in the management of fibromyalgia, the long-term safety and efficacy of nonpharmacologic treatments should be appreciated and stressed. Fibromyalgia is not rheumatoid arthritis: We don’t have true disease-modifying antirheumatic drugs for fibromyalgia. And until we do, pharmacologic treatment is a very useful adjunct, not an imperative. This is an important message for patients, who will probably need treatment for many years to come," Dr. Ablin explained.
The German and Israeli guidelines contain detailed recommendations for a variety of complementary and alternative medicine (CAM) practices, including Tai Chi, guided imagery, acupuncture, yoga, and spa therapy. In contrast, the Canadian guidelines (CMAJ 2013;185:E645-51) deem current evidence insufficient to support the use of CAM practices in fibromyalgia.
The German guidelines recommend a graded approach to treatment. Patients with mild fibromyalgia are to be managed by primary care physicians, with advice given to engage in physical exercise and social activities, with no additional treatment recommended and no specialist care. In moderate fibromyalgia, the treatment plan involves aerobic exercise, time-limited psychological therapy, and referral to a specialist, with drug therapy optional. Patients with severe fibromyalgia symptoms, as well as those with moderate fibromyalgia unresponsive to the earlier-stage interventions, are best managed in a specialized day clinic or inpatient service that emphasizes psychiatric treatment of mental comorbidities, according to the German guidelines. In Germany, insurance companies cover these more intensive services because of their proven track record in reducing occupational disability.
The Israeli approach is different in that it is not based upon the initial severity of fibromyalgia. In step 1, patients receive education about their disorder and the principles involved in its treatment. They also get an individualized aerobic exercise program and are referred for aquatic exercise. Amitriptyline at 10-25 mg at bedtime is prescribed, and a referral is to be made for cognitive-behavioral therapy.
Step 2 is based upon a reassessment 12 weeks after starting step 1. If the patient isn’t doing significantly better, consideration is given to substituting a serotonin-norepinephrine reuptake inhibitor for the amitriptyline, or adding a selective serotonin reuptake inhibitor to amitriptyline, along with prescribing pregabalin to improve sleep and reduce pain. Referral is made for spa therapy and yoga or another meditative movement practice.
As in the German guidelines, the Canadian guidelines also recommend that fibromyalgia diagnosis and care be centered in the primary care setting, with only selective referrals for specialist care.
Dr. Mary-Ann Fitzcharles, lead author of the new Canadian guidelines, said all three guidelines, developed independently on three continents, share in common the same broad clinical concept of fibromyalgia.
"We are all speaking with one voice with the same message: We accept that fibromyalgia is neither a distinct rheumatic nor mental disorder, but rather a cluster of symptoms spanning a broad range of medical disciplines. We’re saying that just focusing on pain is taking away from a large component of the suffering of many of these patients," according to Dr. Fitzcharles of McGill University, Montreal.
The Canadian and German guidelines advise dropping the tender point examination from the patient evaluation, replacing it with an examination for generalized soft tissue tenderness. The Israeli guidelines retain the tender point exam.
One audience member vigorously objected to eliminating the tender point examination.
"The trigger point exam has always been a way for physicians to assess whether the patient is believable. Without using a trigger point exam, I might as well just sign a blank check. How am I going to weed out those who have fibromyalgia from those who are faking and seeking disability status?" he asked.
Dr. Fitzcharles responded: "I think we all know that depending upon who is doing the trigger point exam and how hard you’re pressing, you can make positive trigger points or you can cool them down. So it really is a very inaccurate clinical assessment. However, I will concede that in taking away the security blanket of trigger points from this condition, we now have to think very hard about putting something back in its place for the average primary practitioner to use in the office," the rheumatologist said.
Just what that might be remains unclear, she said. "The conundrum of fibromyalgia is that we have no defining biomarker as yet," Dr. Fitzcharles noted.
She reported serving as a consultant to and/or receiving research funding from Purdue Pharma, Eli Lilly, Pfizer, and Valeant. Dr. Ablin is a consultant to Pfizer.
SAN DIEGO – The high degree of consistency among recent national fibromyalgia guidelines developed independently by multispecialty panels in Canada, Israel, and Germany suggests big changes are afoot in how this common and vexing syndrome is conceptualized and treated, according to Dr. Jacob N. Ablin.
"I hope to convey the feeling that there is somewhat of a paradigm change in the recommendations regarding treatment of fibromyalgia as expressed by these three guidelines. All three emphasize an individually tailored approach based upon the key symptoms and severity, with nonpharmacologic therapies as the major positive first choice for all. The emphasis is on the necessity of self-management strategies, which include aerobic exercise, cognitive-behavioral therapy, and multicomponent exercise and psychologic therapies," he said at the annual meeting of the American College of Rheumatology.
"Pharmacologic therapies were less enthusiastically recommended by all three groups. Contrary to popular perception, the drugs actually achieve only relatively modest effects. And all three groups caution about the side effects of drugs, which may mimic fibromyalgia symptoms," added Dr. Ablin of the Tel Aviv Sourasky Medical Center.
The three medications approved by the Food and Drug Administration for the treatment of fibromyalgia – pregabalin (Lyrica), duloxetine (Cymbalta), and milnacipran (Savella) – received only a weak grade C recommendation in the German guidelines (Schmerz 2012;26:287-90) because all three failed to achieve their primary endpoints in pivotal European clinical trials.
"While drug treatments absolutely continue to play a role in the management of fibromyalgia, the long-term safety and efficacy of nonpharmacologic treatments should be appreciated and stressed. Fibromyalgia is not rheumatoid arthritis: We don’t have true disease-modifying antirheumatic drugs for fibromyalgia. And until we do, pharmacologic treatment is a very useful adjunct, not an imperative. This is an important message for patients, who will probably need treatment for many years to come," Dr. Ablin explained.
The German and Israeli guidelines contain detailed recommendations for a variety of complementary and alternative medicine (CAM) practices, including Tai Chi, guided imagery, acupuncture, yoga, and spa therapy. In contrast, the Canadian guidelines (CMAJ 2013;185:E645-51) deem current evidence insufficient to support the use of CAM practices in fibromyalgia.
The German guidelines recommend a graded approach to treatment. Patients with mild fibromyalgia are to be managed by primary care physicians, with advice given to engage in physical exercise and social activities, with no additional treatment recommended and no specialist care. In moderate fibromyalgia, the treatment plan involves aerobic exercise, time-limited psychological therapy, and referral to a specialist, with drug therapy optional. Patients with severe fibromyalgia symptoms, as well as those with moderate fibromyalgia unresponsive to the earlier-stage interventions, are best managed in a specialized day clinic or inpatient service that emphasizes psychiatric treatment of mental comorbidities, according to the German guidelines. In Germany, insurance companies cover these more intensive services because of their proven track record in reducing occupational disability.
The Israeli approach is different in that it is not based upon the initial severity of fibromyalgia. In step 1, patients receive education about their disorder and the principles involved in its treatment. They also get an individualized aerobic exercise program and are referred for aquatic exercise. Amitriptyline at 10-25 mg at bedtime is prescribed, and a referral is to be made for cognitive-behavioral therapy.
Step 2 is based upon a reassessment 12 weeks after starting step 1. If the patient isn’t doing significantly better, consideration is given to substituting a serotonin-norepinephrine reuptake inhibitor for the amitriptyline, or adding a selective serotonin reuptake inhibitor to amitriptyline, along with prescribing pregabalin to improve sleep and reduce pain. Referral is made for spa therapy and yoga or another meditative movement practice.
As in the German guidelines, the Canadian guidelines also recommend that fibromyalgia diagnosis and care be centered in the primary care setting, with only selective referrals for specialist care.
Dr. Mary-Ann Fitzcharles, lead author of the new Canadian guidelines, said all three guidelines, developed independently on three continents, share in common the same broad clinical concept of fibromyalgia.
"We are all speaking with one voice with the same message: We accept that fibromyalgia is neither a distinct rheumatic nor mental disorder, but rather a cluster of symptoms spanning a broad range of medical disciplines. We’re saying that just focusing on pain is taking away from a large component of the suffering of many of these patients," according to Dr. Fitzcharles of McGill University, Montreal.
The Canadian and German guidelines advise dropping the tender point examination from the patient evaluation, replacing it with an examination for generalized soft tissue tenderness. The Israeli guidelines retain the tender point exam.
One audience member vigorously objected to eliminating the tender point examination.
"The trigger point exam has always been a way for physicians to assess whether the patient is believable. Without using a trigger point exam, I might as well just sign a blank check. How am I going to weed out those who have fibromyalgia from those who are faking and seeking disability status?" he asked.
Dr. Fitzcharles responded: "I think we all know that depending upon who is doing the trigger point exam and how hard you’re pressing, you can make positive trigger points or you can cool them down. So it really is a very inaccurate clinical assessment. However, I will concede that in taking away the security blanket of trigger points from this condition, we now have to think very hard about putting something back in its place for the average primary practitioner to use in the office," the rheumatologist said.
Just what that might be remains unclear, she said. "The conundrum of fibromyalgia is that we have no defining biomarker as yet," Dr. Fitzcharles noted.
She reported serving as a consultant to and/or receiving research funding from Purdue Pharma, Eli Lilly, Pfizer, and Valeant. Dr. Ablin is a consultant to Pfizer.
SAN DIEGO – The high degree of consistency among recent national fibromyalgia guidelines developed independently by multispecialty panels in Canada, Israel, and Germany suggests big changes are afoot in how this common and vexing syndrome is conceptualized and treated, according to Dr. Jacob N. Ablin.
"I hope to convey the feeling that there is somewhat of a paradigm change in the recommendations regarding treatment of fibromyalgia as expressed by these three guidelines. All three emphasize an individually tailored approach based upon the key symptoms and severity, with nonpharmacologic therapies as the major positive first choice for all. The emphasis is on the necessity of self-management strategies, which include aerobic exercise, cognitive-behavioral therapy, and multicomponent exercise and psychologic therapies," he said at the annual meeting of the American College of Rheumatology.
"Pharmacologic therapies were less enthusiastically recommended by all three groups. Contrary to popular perception, the drugs actually achieve only relatively modest effects. And all three groups caution about the side effects of drugs, which may mimic fibromyalgia symptoms," added Dr. Ablin of the Tel Aviv Sourasky Medical Center.
The three medications approved by the Food and Drug Administration for the treatment of fibromyalgia – pregabalin (Lyrica), duloxetine (Cymbalta), and milnacipran (Savella) – received only a weak grade C recommendation in the German guidelines (Schmerz 2012;26:287-90) because all three failed to achieve their primary endpoints in pivotal European clinical trials.
"While drug treatments absolutely continue to play a role in the management of fibromyalgia, the long-term safety and efficacy of nonpharmacologic treatments should be appreciated and stressed. Fibromyalgia is not rheumatoid arthritis: We don’t have true disease-modifying antirheumatic drugs for fibromyalgia. And until we do, pharmacologic treatment is a very useful adjunct, not an imperative. This is an important message for patients, who will probably need treatment for many years to come," Dr. Ablin explained.
The German and Israeli guidelines contain detailed recommendations for a variety of complementary and alternative medicine (CAM) practices, including Tai Chi, guided imagery, acupuncture, yoga, and spa therapy. In contrast, the Canadian guidelines (CMAJ 2013;185:E645-51) deem current evidence insufficient to support the use of CAM practices in fibromyalgia.
The German guidelines recommend a graded approach to treatment. Patients with mild fibromyalgia are to be managed by primary care physicians, with advice given to engage in physical exercise and social activities, with no additional treatment recommended and no specialist care. In moderate fibromyalgia, the treatment plan involves aerobic exercise, time-limited psychological therapy, and referral to a specialist, with drug therapy optional. Patients with severe fibromyalgia symptoms, as well as those with moderate fibromyalgia unresponsive to the earlier-stage interventions, are best managed in a specialized day clinic or inpatient service that emphasizes psychiatric treatment of mental comorbidities, according to the German guidelines. In Germany, insurance companies cover these more intensive services because of their proven track record in reducing occupational disability.
The Israeli approach is different in that it is not based upon the initial severity of fibromyalgia. In step 1, patients receive education about their disorder and the principles involved in its treatment. They also get an individualized aerobic exercise program and are referred for aquatic exercise. Amitriptyline at 10-25 mg at bedtime is prescribed, and a referral is to be made for cognitive-behavioral therapy.
Step 2 is based upon a reassessment 12 weeks after starting step 1. If the patient isn’t doing significantly better, consideration is given to substituting a serotonin-norepinephrine reuptake inhibitor for the amitriptyline, or adding a selective serotonin reuptake inhibitor to amitriptyline, along with prescribing pregabalin to improve sleep and reduce pain. Referral is made for spa therapy and yoga or another meditative movement practice.
As in the German guidelines, the Canadian guidelines also recommend that fibromyalgia diagnosis and care be centered in the primary care setting, with only selective referrals for specialist care.
Dr. Mary-Ann Fitzcharles, lead author of the new Canadian guidelines, said all three guidelines, developed independently on three continents, share in common the same broad clinical concept of fibromyalgia.
"We are all speaking with one voice with the same message: We accept that fibromyalgia is neither a distinct rheumatic nor mental disorder, but rather a cluster of symptoms spanning a broad range of medical disciplines. We’re saying that just focusing on pain is taking away from a large component of the suffering of many of these patients," according to Dr. Fitzcharles of McGill University, Montreal.
The Canadian and German guidelines advise dropping the tender point examination from the patient evaluation, replacing it with an examination for generalized soft tissue tenderness. The Israeli guidelines retain the tender point exam.
One audience member vigorously objected to eliminating the tender point examination.
"The trigger point exam has always been a way for physicians to assess whether the patient is believable. Without using a trigger point exam, I might as well just sign a blank check. How am I going to weed out those who have fibromyalgia from those who are faking and seeking disability status?" he asked.
Dr. Fitzcharles responded: "I think we all know that depending upon who is doing the trigger point exam and how hard you’re pressing, you can make positive trigger points or you can cool them down. So it really is a very inaccurate clinical assessment. However, I will concede that in taking away the security blanket of trigger points from this condition, we now have to think very hard about putting something back in its place for the average primary practitioner to use in the office," the rheumatologist said.
Just what that might be remains unclear, she said. "The conundrum of fibromyalgia is that we have no defining biomarker as yet," Dr. Fitzcharles noted.
She reported serving as a consultant to and/or receiving research funding from Purdue Pharma, Eli Lilly, Pfizer, and Valeant. Dr. Ablin is a consultant to Pfizer.
EXPERT ANALYSIS FROM THE ACR ANNUAL MEETING
Good news: ‘fibrofog’ doesn’t portend Alzheimer’s
SAN DIEGO – ‘Fibrofog’ – the cognitive dysfunction experienced by up to 80% of fibromyalgia patients – is not an early harbinger of Alzheimer’s disease, Dr. Robert S. Katz asserted at the annual meeting of the American College of Rheumatology.
The problems with memory, concentration, language, and thinking, collectively known as fibrofog, affect fibromyalgia patients in their 20s-50s. The onset is typically more sudden and dramatic than with classic forms of dementia. As fibrofog becomes chronic, many affected fibromyalgia patients – fearing the worst – worry they are on a road to Alzheimer’s disease in middle age.
Not so, according to Dr. Katz, professor of medicine at Rush Medical College, Chicago.
He presented a cross-sectional study involving two cohorts. One comprised 69 fibromyalgia patients with symptoms of cognitive dysfunction of 12 months duration or less. The other consisted of 39 fibromyalgia patients with fibrofog symptoms of 7-26 years duration. The long-duration group averaged 52.3 years of age, nearly 7 years older than the group with more recent onset of fibrofog. But the two groups were closely similar in terms of educational level, depression scores, and vocabulary scale scores.
The key study finding: No significant differences existed between the two groups on 13 of the 14 measures of neurocognitive function assessed in the study, including logical memory, paired associate, digit symbol, letter fluency, processing speed, and the Stroop word speed and color speed tests.
Indeed, the sole metric where the long-duration fibrofog patients fared significantly worse was the Trails A test, a measure of spatial scanning and cognitive sequencing. However, the two groups had closely similar scores on the Trails B.
Compared with standardized normative means that have been established for each of the 14 neurocognitive tests, the level of cognitive impairment in the fibromyalgia patients was markedly less than that seen in patients with Alzheimer’s disease. More specifically, measures of processing speed and episodic memory, which are significantly diminished in individuals with preclinical Alzheimer’s disease, were within normal range in both study groups. That’s reassuring. So is the fact that the cognitive deficits present in the shorter-duration group weren’t markedly more pronounced in the group troubled by cognitive problems for an additional 6-25 years. Thus, fibrofog does not appear to be a condition characterized by progressive cognitive decline, the rheumatologist observed.
Dr. Katz reported having no financial conflicts of interest with regard to this study.
SAN DIEGO – ‘Fibrofog’ – the cognitive dysfunction experienced by up to 80% of fibromyalgia patients – is not an early harbinger of Alzheimer’s disease, Dr. Robert S. Katz asserted at the annual meeting of the American College of Rheumatology.
The problems with memory, concentration, language, and thinking, collectively known as fibrofog, affect fibromyalgia patients in their 20s-50s. The onset is typically more sudden and dramatic than with classic forms of dementia. As fibrofog becomes chronic, many affected fibromyalgia patients – fearing the worst – worry they are on a road to Alzheimer’s disease in middle age.
Not so, according to Dr. Katz, professor of medicine at Rush Medical College, Chicago.
He presented a cross-sectional study involving two cohorts. One comprised 69 fibromyalgia patients with symptoms of cognitive dysfunction of 12 months duration or less. The other consisted of 39 fibromyalgia patients with fibrofog symptoms of 7-26 years duration. The long-duration group averaged 52.3 years of age, nearly 7 years older than the group with more recent onset of fibrofog. But the two groups were closely similar in terms of educational level, depression scores, and vocabulary scale scores.
The key study finding: No significant differences existed between the two groups on 13 of the 14 measures of neurocognitive function assessed in the study, including logical memory, paired associate, digit symbol, letter fluency, processing speed, and the Stroop word speed and color speed tests.
Indeed, the sole metric where the long-duration fibrofog patients fared significantly worse was the Trails A test, a measure of spatial scanning and cognitive sequencing. However, the two groups had closely similar scores on the Trails B.
Compared with standardized normative means that have been established for each of the 14 neurocognitive tests, the level of cognitive impairment in the fibromyalgia patients was markedly less than that seen in patients with Alzheimer’s disease. More specifically, measures of processing speed and episodic memory, which are significantly diminished in individuals with preclinical Alzheimer’s disease, were within normal range in both study groups. That’s reassuring. So is the fact that the cognitive deficits present in the shorter-duration group weren’t markedly more pronounced in the group troubled by cognitive problems for an additional 6-25 years. Thus, fibrofog does not appear to be a condition characterized by progressive cognitive decline, the rheumatologist observed.
Dr. Katz reported having no financial conflicts of interest with regard to this study.
SAN DIEGO – ‘Fibrofog’ – the cognitive dysfunction experienced by up to 80% of fibromyalgia patients – is not an early harbinger of Alzheimer’s disease, Dr. Robert S. Katz asserted at the annual meeting of the American College of Rheumatology.
The problems with memory, concentration, language, and thinking, collectively known as fibrofog, affect fibromyalgia patients in their 20s-50s. The onset is typically more sudden and dramatic than with classic forms of dementia. As fibrofog becomes chronic, many affected fibromyalgia patients – fearing the worst – worry they are on a road to Alzheimer’s disease in middle age.
Not so, according to Dr. Katz, professor of medicine at Rush Medical College, Chicago.
He presented a cross-sectional study involving two cohorts. One comprised 69 fibromyalgia patients with symptoms of cognitive dysfunction of 12 months duration or less. The other consisted of 39 fibromyalgia patients with fibrofog symptoms of 7-26 years duration. The long-duration group averaged 52.3 years of age, nearly 7 years older than the group with more recent onset of fibrofog. But the two groups were closely similar in terms of educational level, depression scores, and vocabulary scale scores.
The key study finding: No significant differences existed between the two groups on 13 of the 14 measures of neurocognitive function assessed in the study, including logical memory, paired associate, digit symbol, letter fluency, processing speed, and the Stroop word speed and color speed tests.
Indeed, the sole metric where the long-duration fibrofog patients fared significantly worse was the Trails A test, a measure of spatial scanning and cognitive sequencing. However, the two groups had closely similar scores on the Trails B.
Compared with standardized normative means that have been established for each of the 14 neurocognitive tests, the level of cognitive impairment in the fibromyalgia patients was markedly less than that seen in patients with Alzheimer’s disease. More specifically, measures of processing speed and episodic memory, which are significantly diminished in individuals with preclinical Alzheimer’s disease, were within normal range in both study groups. That’s reassuring. So is the fact that the cognitive deficits present in the shorter-duration group weren’t markedly more pronounced in the group troubled by cognitive problems for an additional 6-25 years. Thus, fibrofog does not appear to be a condition characterized by progressive cognitive decline, the rheumatologist observed.
Dr. Katz reported having no financial conflicts of interest with regard to this study.
AT THE ACR ANNUAL MEETING
Major finding: Fibromyalgia patients who had recent onset of the constellation of cognitive dysfunction complaints known as fibrofog did not score worse on 13 of 14 measures of neurocognitive function than did those with far longer duration of symptoms, suggesting this is not a progressive dementing process.
Data source: A cross-sectional study in which 69 fibromyalgia patients with fibrofog symptoms of 12 months or less and 39 others with cognitive symptoms of 7-26 years duration completed a battery of 14 measures of neurocognitive functioning.
Disclosures: The presenter reported having no financial conflicts of interest.
Advanced pacing slows AF progression in bradycardia
DALLAS – Enhanced pacemaker technology sharply reduced progression of nonpermanent atrial fibrillation to permanent AF in patients with bradycardia and sinus node disease in the large international, randomized MINERVA trial.
In the MINERVA (Minimize Right Ventricular Pacing to Prevent Atrial Fibrillation and Heart Failure) study, the use of dual-chamber pacemakers containing features for atrial preventive pacing and atrial antitachycardia pacing, called DDDRPs, plus managed ventricular pacing (MVP) resulted in a 61% reduction in the incidence of permanent AF during 2 years of prospective follow-up, compared with conventional dual-chamber pacing, Dr. Giuseppe Boriani reported at the American Heart Association scientific sessions.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
In addition, patients in the enhanced-pacing group experienced a 52% reduction in AF-related hospitalizations and emergency department visits, as well as a 49% decrease in atrial cardioversions relative to controls, added Dr. Boriani of the University of Bologna (Italy).
"This is the first study to show that using these enhanced pacing features in combination not only delays the AF disease progression, but also has an impact on health care utilization," he noted. "With this solid evidence of beneficial results, I think the practice guidelines probably should be updated to state this type of device should be implanted for sinus node disease in patients with a history of atrial fibrillation."
MINERVA included 1,166 patients at 63 European, Asian, and Middle Eastern centers; all had bradycardia, sinus node disease, and a history of nonpermanent AF. They received a commercially available Medtronic dual-chamber pacemaker that was randomized single-blind to operate using both the DDDRP and MVP features, the MVP alone, or conventional dual-chamber pacing with neither enhanced feature.
At 2 years, the primary composite endpoint of death, cardiovascular hospitalization, or permanent AF had occurred in 19.8% of the DDDRP plus MVP group. That amounted to a significant 26% reduction in the endpoint compared with the 26.5% rate in the controls on standard dual-chamber pacing. The 21.4% rate in patients whose device used only the MVP algorithm was not significantly different from the rate in controls.
Most strikingly, the 2-year incidence of progression to permanent AF was 3.8% in patients with the enhanced pacing features, compared with 9.2% in those assigned to conventional dual-chamber pacing, for a 61% relative risk reduction. This means 20 patients would need to be treated for 2 years with a pacemaker utilizing the advanced pacing features in order to prevent one additional case of permanent AF.
In addition, patients whose pacemakers utilized rate response and antitachycardia pacing plus MVP reported better quality of life and less fatigue than did those in the other two study arms.
Dr. Boriani estimated that of the 170,000 patients each year in the United States who undergo pacemaker implantation for bradycardia, roughly 25,000 have sinus node disease and a history of AF and would thus now be considered strong candidates for the enhanced pacing features.
Discussant Dr. Anthony Tang explained that it has been known for years that pacemaker algorithms designed to increase the atrial pacing rate above the intrinsic sinus rate can prevent events that trigger AF. The problem has been that this atrial pacing can have the side effect of also increasing ventricular pacing, which can initiate AF, thus canceling the advantage of the atrial preventive pacing. The MVP algorithm decreases this unnecessary pacing in the right ventricle, with the resultant benefits seen in MINERVA.
In his view, the enhanced pacing features used in MINERVA represent a very good option for patients with severe sinus node dysfunction but preserved atrioventricular node conduction, that is, patients who need atrial pacing for long sinus pauses but don’t need much ventricular pacing.
In contrast, for patients with an indication for a pacemaker but who only occasionally need pacing, such as those with a rare sinus pause or occasional AV block, a simple single-chamber ventricular pacemaker will generally do. For those with second- or third-degree AV block with left ventricular dysfunction and who need both atrial and ventricular pacing most of the time, a cardiac resynchronization therapy device plus either a pacemaker or a defibrillator is appropriate, depending upon the severity of the left ventricular dysfunction, according to Dr. Tang, professor of medicine and chair of cardiovascular population health at Western University, London, Ont.
Discussant Dr. Timothy J. Gardner wasn’t bowled over by the number-needed-to-treat (NNT) of 20 in MINERVA.
"That seems a very modest impact. I wonder from a health care/societal benefit standpoint whether an NNT of 20 for a more complex and expensive technology is appropriate," challenged Dr. Gardner, a heart surgeon and medical director of the Christiana Care Center for Heart and Vascular Health in Newark, Del.
Dr. Boriani replied that the benefits seen with advanced pacing in this 2-year study will only become more pronounced with longer follow-up.
"Permanent atrial fibrillation is the later stage of irreversible AF, when the arrhythmia becomes untreatable. And it is a marker for adverse events, including heart failure, stroke, and death," he said.
The MINERVA trial was sponsored by Medtronic, which markets the proprietary enhanced pacing technology. Dr. Boriani reported having no relevant financial interests.
DALLAS – Enhanced pacemaker technology sharply reduced progression of nonpermanent atrial fibrillation to permanent AF in patients with bradycardia and sinus node disease in the large international, randomized MINERVA trial.
In the MINERVA (Minimize Right Ventricular Pacing to Prevent Atrial Fibrillation and Heart Failure) study, the use of dual-chamber pacemakers containing features for atrial preventive pacing and atrial antitachycardia pacing, called DDDRPs, plus managed ventricular pacing (MVP) resulted in a 61% reduction in the incidence of permanent AF during 2 years of prospective follow-up, compared with conventional dual-chamber pacing, Dr. Giuseppe Boriani reported at the American Heart Association scientific sessions.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
In addition, patients in the enhanced-pacing group experienced a 52% reduction in AF-related hospitalizations and emergency department visits, as well as a 49% decrease in atrial cardioversions relative to controls, added Dr. Boriani of the University of Bologna (Italy).
"This is the first study to show that using these enhanced pacing features in combination not only delays the AF disease progression, but also has an impact on health care utilization," he noted. "With this solid evidence of beneficial results, I think the practice guidelines probably should be updated to state this type of device should be implanted for sinus node disease in patients with a history of atrial fibrillation."
MINERVA included 1,166 patients at 63 European, Asian, and Middle Eastern centers; all had bradycardia, sinus node disease, and a history of nonpermanent AF. They received a commercially available Medtronic dual-chamber pacemaker that was randomized single-blind to operate using both the DDDRP and MVP features, the MVP alone, or conventional dual-chamber pacing with neither enhanced feature.
At 2 years, the primary composite endpoint of death, cardiovascular hospitalization, or permanent AF had occurred in 19.8% of the DDDRP plus MVP group. That amounted to a significant 26% reduction in the endpoint compared with the 26.5% rate in the controls on standard dual-chamber pacing. The 21.4% rate in patients whose device used only the MVP algorithm was not significantly different from the rate in controls.
Most strikingly, the 2-year incidence of progression to permanent AF was 3.8% in patients with the enhanced pacing features, compared with 9.2% in those assigned to conventional dual-chamber pacing, for a 61% relative risk reduction. This means 20 patients would need to be treated for 2 years with a pacemaker utilizing the advanced pacing features in order to prevent one additional case of permanent AF.
In addition, patients whose pacemakers utilized rate response and antitachycardia pacing plus MVP reported better quality of life and less fatigue than did those in the other two study arms.
Dr. Boriani estimated that of the 170,000 patients each year in the United States who undergo pacemaker implantation for bradycardia, roughly 25,000 have sinus node disease and a history of AF and would thus now be considered strong candidates for the enhanced pacing features.
Discussant Dr. Anthony Tang explained that it has been known for years that pacemaker algorithms designed to increase the atrial pacing rate above the intrinsic sinus rate can prevent events that trigger AF. The problem has been that this atrial pacing can have the side effect of also increasing ventricular pacing, which can initiate AF, thus canceling the advantage of the atrial preventive pacing. The MVP algorithm decreases this unnecessary pacing in the right ventricle, with the resultant benefits seen in MINERVA.
In his view, the enhanced pacing features used in MINERVA represent a very good option for patients with severe sinus node dysfunction but preserved atrioventricular node conduction, that is, patients who need atrial pacing for long sinus pauses but don’t need much ventricular pacing.
In contrast, for patients with an indication for a pacemaker but who only occasionally need pacing, such as those with a rare sinus pause or occasional AV block, a simple single-chamber ventricular pacemaker will generally do. For those with second- or third-degree AV block with left ventricular dysfunction and who need both atrial and ventricular pacing most of the time, a cardiac resynchronization therapy device plus either a pacemaker or a defibrillator is appropriate, depending upon the severity of the left ventricular dysfunction, according to Dr. Tang, professor of medicine and chair of cardiovascular population health at Western University, London, Ont.
Discussant Dr. Timothy J. Gardner wasn’t bowled over by the number-needed-to-treat (NNT) of 20 in MINERVA.
"That seems a very modest impact. I wonder from a health care/societal benefit standpoint whether an NNT of 20 for a more complex and expensive technology is appropriate," challenged Dr. Gardner, a heart surgeon and medical director of the Christiana Care Center for Heart and Vascular Health in Newark, Del.
Dr. Boriani replied that the benefits seen with advanced pacing in this 2-year study will only become more pronounced with longer follow-up.
"Permanent atrial fibrillation is the later stage of irreversible AF, when the arrhythmia becomes untreatable. And it is a marker for adverse events, including heart failure, stroke, and death," he said.
The MINERVA trial was sponsored by Medtronic, which markets the proprietary enhanced pacing technology. Dr. Boriani reported having no relevant financial interests.
DALLAS – Enhanced pacemaker technology sharply reduced progression of nonpermanent atrial fibrillation to permanent AF in patients with bradycardia and sinus node disease in the large international, randomized MINERVA trial.
In the MINERVA (Minimize Right Ventricular Pacing to Prevent Atrial Fibrillation and Heart Failure) study, the use of dual-chamber pacemakers containing features for atrial preventive pacing and atrial antitachycardia pacing, called DDDRPs, plus managed ventricular pacing (MVP) resulted in a 61% reduction in the incidence of permanent AF during 2 years of prospective follow-up, compared with conventional dual-chamber pacing, Dr. Giuseppe Boriani reported at the American Heart Association scientific sessions.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
In addition, patients in the enhanced-pacing group experienced a 52% reduction in AF-related hospitalizations and emergency department visits, as well as a 49% decrease in atrial cardioversions relative to controls, added Dr. Boriani of the University of Bologna (Italy).
"This is the first study to show that using these enhanced pacing features in combination not only delays the AF disease progression, but also has an impact on health care utilization," he noted. "With this solid evidence of beneficial results, I think the practice guidelines probably should be updated to state this type of device should be implanted for sinus node disease in patients with a history of atrial fibrillation."
MINERVA included 1,166 patients at 63 European, Asian, and Middle Eastern centers; all had bradycardia, sinus node disease, and a history of nonpermanent AF. They received a commercially available Medtronic dual-chamber pacemaker that was randomized single-blind to operate using both the DDDRP and MVP features, the MVP alone, or conventional dual-chamber pacing with neither enhanced feature.
At 2 years, the primary composite endpoint of death, cardiovascular hospitalization, or permanent AF had occurred in 19.8% of the DDDRP plus MVP group. That amounted to a significant 26% reduction in the endpoint compared with the 26.5% rate in the controls on standard dual-chamber pacing. The 21.4% rate in patients whose device used only the MVP algorithm was not significantly different from the rate in controls.
Most strikingly, the 2-year incidence of progression to permanent AF was 3.8% in patients with the enhanced pacing features, compared with 9.2% in those assigned to conventional dual-chamber pacing, for a 61% relative risk reduction. This means 20 patients would need to be treated for 2 years with a pacemaker utilizing the advanced pacing features in order to prevent one additional case of permanent AF.
In addition, patients whose pacemakers utilized rate response and antitachycardia pacing plus MVP reported better quality of life and less fatigue than did those in the other two study arms.
Dr. Boriani estimated that of the 170,000 patients each year in the United States who undergo pacemaker implantation for bradycardia, roughly 25,000 have sinus node disease and a history of AF and would thus now be considered strong candidates for the enhanced pacing features.
Discussant Dr. Anthony Tang explained that it has been known for years that pacemaker algorithms designed to increase the atrial pacing rate above the intrinsic sinus rate can prevent events that trigger AF. The problem has been that this atrial pacing can have the side effect of also increasing ventricular pacing, which can initiate AF, thus canceling the advantage of the atrial preventive pacing. The MVP algorithm decreases this unnecessary pacing in the right ventricle, with the resultant benefits seen in MINERVA.
In his view, the enhanced pacing features used in MINERVA represent a very good option for patients with severe sinus node dysfunction but preserved atrioventricular node conduction, that is, patients who need atrial pacing for long sinus pauses but don’t need much ventricular pacing.
In contrast, for patients with an indication for a pacemaker but who only occasionally need pacing, such as those with a rare sinus pause or occasional AV block, a simple single-chamber ventricular pacemaker will generally do. For those with second- or third-degree AV block with left ventricular dysfunction and who need both atrial and ventricular pacing most of the time, a cardiac resynchronization therapy device plus either a pacemaker or a defibrillator is appropriate, depending upon the severity of the left ventricular dysfunction, according to Dr. Tang, professor of medicine and chair of cardiovascular population health at Western University, London, Ont.
Discussant Dr. Timothy J. Gardner wasn’t bowled over by the number-needed-to-treat (NNT) of 20 in MINERVA.
"That seems a very modest impact. I wonder from a health care/societal benefit standpoint whether an NNT of 20 for a more complex and expensive technology is appropriate," challenged Dr. Gardner, a heart surgeon and medical director of the Christiana Care Center for Heart and Vascular Health in Newark, Del.
Dr. Boriani replied that the benefits seen with advanced pacing in this 2-year study will only become more pronounced with longer follow-up.
"Permanent atrial fibrillation is the later stage of irreversible AF, when the arrhythmia becomes untreatable. And it is a marker for adverse events, including heart failure, stroke, and death," he said.
The MINERVA trial was sponsored by Medtronic, which markets the proprietary enhanced pacing technology. Dr. Boriani reported having no relevant financial interests.
AT THE AHA SCIENTIFIC SESSIONS
Major finding: Patients with bradycardia, sinus node disease, and a history of atrial fibrillation who were implanted with a dual-chamber pacemaker utilizing enhanced pacing features showed a 61% reduction in progression to permanent AF over 2 years, compared with those randomized to conventional dual-chamber pacing.
Data source: The MINERVA trial, a single-blind, randomized, international, prospective, 2-year trial conducted in 1,166 patients.
Disclosures: The MINERVA trial was sponsored by Medtronic, which markets the proprietary enhanced pacing technology. Dr. Boriani reported having no relevant financial interests.
Maternal lupus doubled autism risk
SAN DIEGO – The risk of autism spectrum disorders is more than doubled among children born to mothers with systemic lupus erythematosus, according to the first-ever controlled study to address the question.
That being said, women with SLE can be reassured that despite this elevated relative risk, the absolute risk that their child will be diagnosed with an autism spectrum disorder (ASD) is low – less than 1 in 50 – Dr. Evelyne Vinet said at the annual meeting of the American College of Rheumatology.
The increased risk of ASD in children born to women with SLE documented in this large study was not mediated by in utero exposure to medications for SLE, including antimalarials, immunosuppressive agents, corticosteroids, and antidepressants. Use of those drugs in pregnancy wasn’t associated with any increased risk. In light of this, further research is warranted into a highly promising alternative hypothesis: that in utero exposure to SLE-related autoantibodies, such as anti-DNA and antiphospholipid antibodies, may play a causative role, said Dr. Vinet, a rheumatologist at McGill University, Montreal.
Children born to mothers with SLE also had an increased likelihood of being diagnosed with attention-deficit/hyperactivity disorder in this study. However, in contrast to the situation with ASD, the increased risk of ADHD appeared to result from exposure to medications in utero – specifically, antidepressants and possibly immunosuppressives – rather than to maternal SLE per se, she added.
Dr. Vinet presented an analysis of the OSLER (Offspring of Systemic Lupus Erythematosus Mothers Registry) database, the world’s largest cohort of children born to mothers with SLE. OSLER includes all women with a diagnosis of SLE hospitalized for childbirth in the province of Quebec since 1989. This study included 509 women who had 719 children after they had been diagnosed with SLE, as well as 5,824 controls matched for age and year of delivery along with their 8,493 children. The mean maternal age was 30.3 years. The children were followed out to a mean age of 9.1 years.
ASD was diagnosed in 1.4% of children born to mothers with SLE, compared with 0.6% of control children. The mean age at diagnosis of ASD was noticeably lower in the offspring of mothers with SLE: 3.8 years, compared with 5.7 years in the control children.
Women with SLE had higher rates of hypertension, asthma, and diabetes than did controls at the time of delivery. They also had higher rates of obstetric complications, including preterm birth, small for gestational age, and gestational diabetes. In a multivariate analysis adjusted for these variables, maternal SLE remained independently associated with a 2.3-fold increased risk of diagnosis of an ASD in offspring.
Complete and reliable records of prescription drug use during pregnancy were available only for the roughly 20% of mothers belonging to the provincial medication public assistance program. Of the 18 cases of ASD diagnosed in the children of 1,925 mothers covered by the program, only one occurred among the 155 offspring of SLE mothers; that child had been exposed to corticosteroids in utero. Of the 17 cases of ASD diagnosed in the control group, 16 had no exposures to the medications under scrutiny, and one involved in utero exposure to an anticonvulsant.
The OSLER database doesn’t include information about maternal autoantibody levels, but Dr. Vinet and her coinvestigators have made the collection of such data a top priority in light of recent studies in animal models of SLE by other investigators. Those studies showed that SLE-related autoantibodies, including n-methyl-d-aspartate receptor antibodies and antiphospholipid antibodies, as well as interleukin-6 and other cytokines, alter fetal brain development and induce behavioral anomalies suggestive of autism, such as withdrawal, in the mouse offspring.
In addition, French investigators recently reported that 3 of 36 children born to mothers with antiphospholipid syndrome developed ASD, although Dr. Vinet noted that this small but intriguing study lacked a control group (Semin. Arthritis Rheum. 2013 Aug. 1 [doi: 10.1016/j.semarthrit.2013.07.001]).
ADHD was diagnosed in 9.9% of the children of mothers with SLE in the OSLER database, compared with 6.1% of controls. It was diagnosed when the children were older: at a mean age of 12.5 years, compared with 7.8 years in the controls. In a multivariate analysis adjusted for in utero drug exposures, the association between ADHD and maternal SLE was no longer significant. However, in utero exposure to antidepressant medication, regardless of whether or not a mother had SLE, was associated with a hefty 3.7-fold increase in ADHD in the offspring. Because of the relatively small number of mothers on antidepressant medication, Dr. Vinet is now collaborating with investigators in other Canadian provinces having more comprehensive maternal medication exposure data to take a closer look at this tentative link between in utero drug exposure and ADHD.
This study of the OSLER database was funded by the Canadian Institutes of Health Research. Dr. Vinet reported having no financial conflicts.
SAN DIEGO – The risk of autism spectrum disorders is more than doubled among children born to mothers with systemic lupus erythematosus, according to the first-ever controlled study to address the question.
That being said, women with SLE can be reassured that despite this elevated relative risk, the absolute risk that their child will be diagnosed with an autism spectrum disorder (ASD) is low – less than 1 in 50 – Dr. Evelyne Vinet said at the annual meeting of the American College of Rheumatology.
The increased risk of ASD in children born to women with SLE documented in this large study was not mediated by in utero exposure to medications for SLE, including antimalarials, immunosuppressive agents, corticosteroids, and antidepressants. Use of those drugs in pregnancy wasn’t associated with any increased risk. In light of this, further research is warranted into a highly promising alternative hypothesis: that in utero exposure to SLE-related autoantibodies, such as anti-DNA and antiphospholipid antibodies, may play a causative role, said Dr. Vinet, a rheumatologist at McGill University, Montreal.
Children born to mothers with SLE also had an increased likelihood of being diagnosed with attention-deficit/hyperactivity disorder in this study. However, in contrast to the situation with ASD, the increased risk of ADHD appeared to result from exposure to medications in utero – specifically, antidepressants and possibly immunosuppressives – rather than to maternal SLE per se, she added.
Dr. Vinet presented an analysis of the OSLER (Offspring of Systemic Lupus Erythematosus Mothers Registry) database, the world’s largest cohort of children born to mothers with SLE. OSLER includes all women with a diagnosis of SLE hospitalized for childbirth in the province of Quebec since 1989. This study included 509 women who had 719 children after they had been diagnosed with SLE, as well as 5,824 controls matched for age and year of delivery along with their 8,493 children. The mean maternal age was 30.3 years. The children were followed out to a mean age of 9.1 years.
ASD was diagnosed in 1.4% of children born to mothers with SLE, compared with 0.6% of control children. The mean age at diagnosis of ASD was noticeably lower in the offspring of mothers with SLE: 3.8 years, compared with 5.7 years in the control children.
Women with SLE had higher rates of hypertension, asthma, and diabetes than did controls at the time of delivery. They also had higher rates of obstetric complications, including preterm birth, small for gestational age, and gestational diabetes. In a multivariate analysis adjusted for these variables, maternal SLE remained independently associated with a 2.3-fold increased risk of diagnosis of an ASD in offspring.
Complete and reliable records of prescription drug use during pregnancy were available only for the roughly 20% of mothers belonging to the provincial medication public assistance program. Of the 18 cases of ASD diagnosed in the children of 1,925 mothers covered by the program, only one occurred among the 155 offspring of SLE mothers; that child had been exposed to corticosteroids in utero. Of the 17 cases of ASD diagnosed in the control group, 16 had no exposures to the medications under scrutiny, and one involved in utero exposure to an anticonvulsant.
The OSLER database doesn’t include information about maternal autoantibody levels, but Dr. Vinet and her coinvestigators have made the collection of such data a top priority in light of recent studies in animal models of SLE by other investigators. Those studies showed that SLE-related autoantibodies, including n-methyl-d-aspartate receptor antibodies and antiphospholipid antibodies, as well as interleukin-6 and other cytokines, alter fetal brain development and induce behavioral anomalies suggestive of autism, such as withdrawal, in the mouse offspring.
In addition, French investigators recently reported that 3 of 36 children born to mothers with antiphospholipid syndrome developed ASD, although Dr. Vinet noted that this small but intriguing study lacked a control group (Semin. Arthritis Rheum. 2013 Aug. 1 [doi: 10.1016/j.semarthrit.2013.07.001]).
ADHD was diagnosed in 9.9% of the children of mothers with SLE in the OSLER database, compared with 6.1% of controls. It was diagnosed when the children were older: at a mean age of 12.5 years, compared with 7.8 years in the controls. In a multivariate analysis adjusted for in utero drug exposures, the association between ADHD and maternal SLE was no longer significant. However, in utero exposure to antidepressant medication, regardless of whether or not a mother had SLE, was associated with a hefty 3.7-fold increase in ADHD in the offspring. Because of the relatively small number of mothers on antidepressant medication, Dr. Vinet is now collaborating with investigators in other Canadian provinces having more comprehensive maternal medication exposure data to take a closer look at this tentative link between in utero drug exposure and ADHD.
This study of the OSLER database was funded by the Canadian Institutes of Health Research. Dr. Vinet reported having no financial conflicts.
SAN DIEGO – The risk of autism spectrum disorders is more than doubled among children born to mothers with systemic lupus erythematosus, according to the first-ever controlled study to address the question.
That being said, women with SLE can be reassured that despite this elevated relative risk, the absolute risk that their child will be diagnosed with an autism spectrum disorder (ASD) is low – less than 1 in 50 – Dr. Evelyne Vinet said at the annual meeting of the American College of Rheumatology.
The increased risk of ASD in children born to women with SLE documented in this large study was not mediated by in utero exposure to medications for SLE, including antimalarials, immunosuppressive agents, corticosteroids, and antidepressants. Use of those drugs in pregnancy wasn’t associated with any increased risk. In light of this, further research is warranted into a highly promising alternative hypothesis: that in utero exposure to SLE-related autoantibodies, such as anti-DNA and antiphospholipid antibodies, may play a causative role, said Dr. Vinet, a rheumatologist at McGill University, Montreal.
Children born to mothers with SLE also had an increased likelihood of being diagnosed with attention-deficit/hyperactivity disorder in this study. However, in contrast to the situation with ASD, the increased risk of ADHD appeared to result from exposure to medications in utero – specifically, antidepressants and possibly immunosuppressives – rather than to maternal SLE per se, she added.
Dr. Vinet presented an analysis of the OSLER (Offspring of Systemic Lupus Erythematosus Mothers Registry) database, the world’s largest cohort of children born to mothers with SLE. OSLER includes all women with a diagnosis of SLE hospitalized for childbirth in the province of Quebec since 1989. This study included 509 women who had 719 children after they had been diagnosed with SLE, as well as 5,824 controls matched for age and year of delivery along with their 8,493 children. The mean maternal age was 30.3 years. The children were followed out to a mean age of 9.1 years.
ASD was diagnosed in 1.4% of children born to mothers with SLE, compared with 0.6% of control children. The mean age at diagnosis of ASD was noticeably lower in the offspring of mothers with SLE: 3.8 years, compared with 5.7 years in the control children.
Women with SLE had higher rates of hypertension, asthma, and diabetes than did controls at the time of delivery. They also had higher rates of obstetric complications, including preterm birth, small for gestational age, and gestational diabetes. In a multivariate analysis adjusted for these variables, maternal SLE remained independently associated with a 2.3-fold increased risk of diagnosis of an ASD in offspring.
Complete and reliable records of prescription drug use during pregnancy were available only for the roughly 20% of mothers belonging to the provincial medication public assistance program. Of the 18 cases of ASD diagnosed in the children of 1,925 mothers covered by the program, only one occurred among the 155 offspring of SLE mothers; that child had been exposed to corticosteroids in utero. Of the 17 cases of ASD diagnosed in the control group, 16 had no exposures to the medications under scrutiny, and one involved in utero exposure to an anticonvulsant.
The OSLER database doesn’t include information about maternal autoantibody levels, but Dr. Vinet and her coinvestigators have made the collection of such data a top priority in light of recent studies in animal models of SLE by other investigators. Those studies showed that SLE-related autoantibodies, including n-methyl-d-aspartate receptor antibodies and antiphospholipid antibodies, as well as interleukin-6 and other cytokines, alter fetal brain development and induce behavioral anomalies suggestive of autism, such as withdrawal, in the mouse offspring.
In addition, French investigators recently reported that 3 of 36 children born to mothers with antiphospholipid syndrome developed ASD, although Dr. Vinet noted that this small but intriguing study lacked a control group (Semin. Arthritis Rheum. 2013 Aug. 1 [doi: 10.1016/j.semarthrit.2013.07.001]).
ADHD was diagnosed in 9.9% of the children of mothers with SLE in the OSLER database, compared with 6.1% of controls. It was diagnosed when the children were older: at a mean age of 12.5 years, compared with 7.8 years in the controls. In a multivariate analysis adjusted for in utero drug exposures, the association between ADHD and maternal SLE was no longer significant. However, in utero exposure to antidepressant medication, regardless of whether or not a mother had SLE, was associated with a hefty 3.7-fold increase in ADHD in the offspring. Because of the relatively small number of mothers on antidepressant medication, Dr. Vinet is now collaborating with investigators in other Canadian provinces having more comprehensive maternal medication exposure data to take a closer look at this tentative link between in utero drug exposure and ADHD.
This study of the OSLER database was funded by the Canadian Institutes of Health Research. Dr. Vinet reported having no financial conflicts.
AT THE ACR ANNUAL MEETING
Major finding: Children born to mothers with systemic lupus erythematosus had an adjusted 2.3-fold increased risk of being diagnosed with an autism spectrum disorder, compared with controls.
Data source: This study from the world’s largest cohort of children born to mothers with SLE included 509 affected mothers and their 719 children, as well as 5,824 matched control mothers and their 8,493 children.
Disclosures: This study of the OSLER database was funded by the Canadian Institutes of Health Research. Dr. Vinet reported having no financial conflicts.