User login
IBIS-II: Anastrozole highly effective in preventing breast cancer
SAN ANTONIO – The aromatase inhibitor anastrozole has emerged as a major new agent for the primary prevention of breast cancer in high-risk postmenopausal women on the strength of a 53% reduction relative to placebo in the IBIS-II trial.
"Our results provide substantial support for the use of anastrozole as the treatment of first choice for prevention of breast cancer in high-risk postmenopausal women," Jack Cuzick, Ph.D., declared in presenting the study results at the San Antonio Breast Cancer Symposium.
IBIS-II (the International Breast Cancer Intervention Study II) was an 16-country, double-blind, randomized, placebo-controlled trial in which 3,864 postmenopausal women aged 40-70 years at high risk for breast cancer were randomized to 1 mg of oral anastrozole (Arimidex) or placebo daily for 5 years.
At 7 years of follow-up, the cumulative incidence of all breast cancers in an intent-to-treat analysis was 5.6% with placebo and 2.8% in the anastrozole group, for a highly significant 53% reduction in risk. This translates into a number needed to treat of 36; that is, it’s estimated that treating 36 women for 5 years will prevent one breast cancer in 7 years.
The incidence of estrogen receptor–positive invasive breast cancer was 3.3% in controls, compared with 1.4% with anastrozole, for a 58% relative risk reduction, added Dr. Cuzick, head of the Cancer Research U.K. Center for Cancer Prevention and director of the Wolfson Institute of Preventive Medicine at Queen Mary University of London.
The incidence of high-grade tumors was 35% lower in the anastrozole group. With additional years of follow-up, this will probably translate into a reduction in deaths from breast cancer in the anastrozole group. The plan is for at least 10 years of follow-up in IBIS-II.
Among women on placebo, 72% completed 5 years, as did 68% assigned to anastrozole.
"One of the most important findings of this trial is that the drug was very well tolerated, with only an absolute 4% difference in compliance. So drug side effects didn’t influence compliance in any major way," Dr. Cuzick observed.
The incidence of other cancers was 3.6% with placebo and 2.1% with anastrozole, for a significant 42% relative risk reduction. This was mostly driven by markedly fewer cases of skin cancer and colorectal cancer in patients on the aromatase inhibitor.
"We don’t really understand this. It’s another exciting possibility – that this drug may have an effect in reducing the risk of other cancers – which we will continue to explore," he continued.
Going into the trial, the anastrozole side effect of chief concern was fractures, but under the study protocol all participants had a baseline bone mineral density scan and were placed on a bisphosphonate if indicated. That’s presumably the explanation for the finding that the incidence of fractures didn’t differ significantly between the two study arms.
Musculoskeletal pain was reported by 64% of anastrozole-treated patients and 58% on placebo. The incidence of moderate musculoskeletal pain was 22% with anastrozole and 19% with placebo, while severe pain was reported by 8% of women in the anastrozole group, compared with 6% on placebo.
Pointing to the 58% incidence of musculoskeletal side effects in the placebo group, Dr. Cuzick said that "there’s a general perception that the aromatase inhibitors are very toxic and cause a lot of aches and pains. But 90% of the aches and pains in this study had nothing to do with the drug. They’re just a reflection of the fact that in the postmenopausal years women do get aches and pains."
Vasomotor symptoms occurred in 49% of the placebo group and 57% of women on anastrozole, a statistically significant difference.
Dr. Cuzick said that he believes the sharp reduction in breast cancer seen with anastrozole in IBIS-II is probably a class effect common to the other aromatase inhibitors. In the MAP.3 trial, an earlier study with shorter follow-up (N. Engl. J. Med. 2011;364:2381-91), exemestane showed results similar to those seen in IBIS-II. He added that the greater size and duration of IBIS-II, coupled with the fact that contralateral breast cancer rates continue to be lower at 10 years of follow-up in the anastrozole breast cancer treatment trials, makes him "quite confident" in declaring anastrozole to be the drug of first choice for prevention.
Audience member Dr. Pamela J. Goodwin rose to take issue with that assertion. She noted that there are two approved agents for this indication – tamoxifen and raloxifene – yet placebo was used as the comparator in IBIS-II.
"The evidence is indirect evidence," Dr. Cuzick replied. "Anastrozole and exemestane have shown very, very similar results. The aromatase inhibitors show larger risk reductions than tamoxifen or raloxifene, and overall serious side effects are substantially less. For that reason we think they are the appropriate first choice."
"Those are cross-trial comparisons. I’m not sure I would go that far," retorted Dr. Goodwin, professor of medicine at the University of Toronto.
In a press conference announcing the IBIS-II results, Dr. Cuzick noted that neither tamoxifen nor raloxifene is widely utilized for primary prevention of breast cancer. More public education is in order, he added.
"I think there’s a lot to be done to model what the cardiologists have done: They’ve convinced people that high cholesterol and high blood pressure are actually diseases. In fact, they’re only risk factors, but by taking drugs to reduce those risk factors there’s been a major effect. We need to make people aware there are effective ways of reducing the risk of breast cancer by more than 50%. The toxicities are limited, and if you have toxicity you simply stop treatment," he said.
The IBIS-II study was funded by Cancer Research U.K., the National Health and Medical Research Council of Australia, AstraZeneca, and Sanofi-Aventis. Dr. Cuzick is on the speaker’s bureau for AstraZeneca.
Simultaneous with Dr. Cuzick’s presentation in San Antonio, the IBIS-II results were published online in the Lancet (2013 Dec. 12 [doi:10.1016/S0140-6736(13)62292-8]).
SAN ANTONIO – The aromatase inhibitor anastrozole has emerged as a major new agent for the primary prevention of breast cancer in high-risk postmenopausal women on the strength of a 53% reduction relative to placebo in the IBIS-II trial.
"Our results provide substantial support for the use of anastrozole as the treatment of first choice for prevention of breast cancer in high-risk postmenopausal women," Jack Cuzick, Ph.D., declared in presenting the study results at the San Antonio Breast Cancer Symposium.
IBIS-II (the International Breast Cancer Intervention Study II) was an 16-country, double-blind, randomized, placebo-controlled trial in which 3,864 postmenopausal women aged 40-70 years at high risk for breast cancer were randomized to 1 mg of oral anastrozole (Arimidex) or placebo daily for 5 years.
At 7 years of follow-up, the cumulative incidence of all breast cancers in an intent-to-treat analysis was 5.6% with placebo and 2.8% in the anastrozole group, for a highly significant 53% reduction in risk. This translates into a number needed to treat of 36; that is, it’s estimated that treating 36 women for 5 years will prevent one breast cancer in 7 years.
The incidence of estrogen receptor–positive invasive breast cancer was 3.3% in controls, compared with 1.4% with anastrozole, for a 58% relative risk reduction, added Dr. Cuzick, head of the Cancer Research U.K. Center for Cancer Prevention and director of the Wolfson Institute of Preventive Medicine at Queen Mary University of London.
The incidence of high-grade tumors was 35% lower in the anastrozole group. With additional years of follow-up, this will probably translate into a reduction in deaths from breast cancer in the anastrozole group. The plan is for at least 10 years of follow-up in IBIS-II.
Among women on placebo, 72% completed 5 years, as did 68% assigned to anastrozole.
"One of the most important findings of this trial is that the drug was very well tolerated, with only an absolute 4% difference in compliance. So drug side effects didn’t influence compliance in any major way," Dr. Cuzick observed.
The incidence of other cancers was 3.6% with placebo and 2.1% with anastrozole, for a significant 42% relative risk reduction. This was mostly driven by markedly fewer cases of skin cancer and colorectal cancer in patients on the aromatase inhibitor.
"We don’t really understand this. It’s another exciting possibility – that this drug may have an effect in reducing the risk of other cancers – which we will continue to explore," he continued.
Going into the trial, the anastrozole side effect of chief concern was fractures, but under the study protocol all participants had a baseline bone mineral density scan and were placed on a bisphosphonate if indicated. That’s presumably the explanation for the finding that the incidence of fractures didn’t differ significantly between the two study arms.
Musculoskeletal pain was reported by 64% of anastrozole-treated patients and 58% on placebo. The incidence of moderate musculoskeletal pain was 22% with anastrozole and 19% with placebo, while severe pain was reported by 8% of women in the anastrozole group, compared with 6% on placebo.
Pointing to the 58% incidence of musculoskeletal side effects in the placebo group, Dr. Cuzick said that "there’s a general perception that the aromatase inhibitors are very toxic and cause a lot of aches and pains. But 90% of the aches and pains in this study had nothing to do with the drug. They’re just a reflection of the fact that in the postmenopausal years women do get aches and pains."
Vasomotor symptoms occurred in 49% of the placebo group and 57% of women on anastrozole, a statistically significant difference.
Dr. Cuzick said that he believes the sharp reduction in breast cancer seen with anastrozole in IBIS-II is probably a class effect common to the other aromatase inhibitors. In the MAP.3 trial, an earlier study with shorter follow-up (N. Engl. J. Med. 2011;364:2381-91), exemestane showed results similar to those seen in IBIS-II. He added that the greater size and duration of IBIS-II, coupled with the fact that contralateral breast cancer rates continue to be lower at 10 years of follow-up in the anastrozole breast cancer treatment trials, makes him "quite confident" in declaring anastrozole to be the drug of first choice for prevention.
Audience member Dr. Pamela J. Goodwin rose to take issue with that assertion. She noted that there are two approved agents for this indication – tamoxifen and raloxifene – yet placebo was used as the comparator in IBIS-II.
"The evidence is indirect evidence," Dr. Cuzick replied. "Anastrozole and exemestane have shown very, very similar results. The aromatase inhibitors show larger risk reductions than tamoxifen or raloxifene, and overall serious side effects are substantially less. For that reason we think they are the appropriate first choice."
"Those are cross-trial comparisons. I’m not sure I would go that far," retorted Dr. Goodwin, professor of medicine at the University of Toronto.
In a press conference announcing the IBIS-II results, Dr. Cuzick noted that neither tamoxifen nor raloxifene is widely utilized for primary prevention of breast cancer. More public education is in order, he added.
"I think there’s a lot to be done to model what the cardiologists have done: They’ve convinced people that high cholesterol and high blood pressure are actually diseases. In fact, they’re only risk factors, but by taking drugs to reduce those risk factors there’s been a major effect. We need to make people aware there are effective ways of reducing the risk of breast cancer by more than 50%. The toxicities are limited, and if you have toxicity you simply stop treatment," he said.
The IBIS-II study was funded by Cancer Research U.K., the National Health and Medical Research Council of Australia, AstraZeneca, and Sanofi-Aventis. Dr. Cuzick is on the speaker’s bureau for AstraZeneca.
Simultaneous with Dr. Cuzick’s presentation in San Antonio, the IBIS-II results were published online in the Lancet (2013 Dec. 12 [doi:10.1016/S0140-6736(13)62292-8]).
SAN ANTONIO – The aromatase inhibitor anastrozole has emerged as a major new agent for the primary prevention of breast cancer in high-risk postmenopausal women on the strength of a 53% reduction relative to placebo in the IBIS-II trial.
"Our results provide substantial support for the use of anastrozole as the treatment of first choice for prevention of breast cancer in high-risk postmenopausal women," Jack Cuzick, Ph.D., declared in presenting the study results at the San Antonio Breast Cancer Symposium.
IBIS-II (the International Breast Cancer Intervention Study II) was an 16-country, double-blind, randomized, placebo-controlled trial in which 3,864 postmenopausal women aged 40-70 years at high risk for breast cancer were randomized to 1 mg of oral anastrozole (Arimidex) or placebo daily for 5 years.
At 7 years of follow-up, the cumulative incidence of all breast cancers in an intent-to-treat analysis was 5.6% with placebo and 2.8% in the anastrozole group, for a highly significant 53% reduction in risk. This translates into a number needed to treat of 36; that is, it’s estimated that treating 36 women for 5 years will prevent one breast cancer in 7 years.
The incidence of estrogen receptor–positive invasive breast cancer was 3.3% in controls, compared with 1.4% with anastrozole, for a 58% relative risk reduction, added Dr. Cuzick, head of the Cancer Research U.K. Center for Cancer Prevention and director of the Wolfson Institute of Preventive Medicine at Queen Mary University of London.
The incidence of high-grade tumors was 35% lower in the anastrozole group. With additional years of follow-up, this will probably translate into a reduction in deaths from breast cancer in the anastrozole group. The plan is for at least 10 years of follow-up in IBIS-II.
Among women on placebo, 72% completed 5 years, as did 68% assigned to anastrozole.
"One of the most important findings of this trial is that the drug was very well tolerated, with only an absolute 4% difference in compliance. So drug side effects didn’t influence compliance in any major way," Dr. Cuzick observed.
The incidence of other cancers was 3.6% with placebo and 2.1% with anastrozole, for a significant 42% relative risk reduction. This was mostly driven by markedly fewer cases of skin cancer and colorectal cancer in patients on the aromatase inhibitor.
"We don’t really understand this. It’s another exciting possibility – that this drug may have an effect in reducing the risk of other cancers – which we will continue to explore," he continued.
Going into the trial, the anastrozole side effect of chief concern was fractures, but under the study protocol all participants had a baseline bone mineral density scan and were placed on a bisphosphonate if indicated. That’s presumably the explanation for the finding that the incidence of fractures didn’t differ significantly between the two study arms.
Musculoskeletal pain was reported by 64% of anastrozole-treated patients and 58% on placebo. The incidence of moderate musculoskeletal pain was 22% with anastrozole and 19% with placebo, while severe pain was reported by 8% of women in the anastrozole group, compared with 6% on placebo.
Pointing to the 58% incidence of musculoskeletal side effects in the placebo group, Dr. Cuzick said that "there’s a general perception that the aromatase inhibitors are very toxic and cause a lot of aches and pains. But 90% of the aches and pains in this study had nothing to do with the drug. They’re just a reflection of the fact that in the postmenopausal years women do get aches and pains."
Vasomotor symptoms occurred in 49% of the placebo group and 57% of women on anastrozole, a statistically significant difference.
Dr. Cuzick said that he believes the sharp reduction in breast cancer seen with anastrozole in IBIS-II is probably a class effect common to the other aromatase inhibitors. In the MAP.3 trial, an earlier study with shorter follow-up (N. Engl. J. Med. 2011;364:2381-91), exemestane showed results similar to those seen in IBIS-II. He added that the greater size and duration of IBIS-II, coupled with the fact that contralateral breast cancer rates continue to be lower at 10 years of follow-up in the anastrozole breast cancer treatment trials, makes him "quite confident" in declaring anastrozole to be the drug of first choice for prevention.
Audience member Dr. Pamela J. Goodwin rose to take issue with that assertion. She noted that there are two approved agents for this indication – tamoxifen and raloxifene – yet placebo was used as the comparator in IBIS-II.
"The evidence is indirect evidence," Dr. Cuzick replied. "Anastrozole and exemestane have shown very, very similar results. The aromatase inhibitors show larger risk reductions than tamoxifen or raloxifene, and overall serious side effects are substantially less. For that reason we think they are the appropriate first choice."
"Those are cross-trial comparisons. I’m not sure I would go that far," retorted Dr. Goodwin, professor of medicine at the University of Toronto.
In a press conference announcing the IBIS-II results, Dr. Cuzick noted that neither tamoxifen nor raloxifene is widely utilized for primary prevention of breast cancer. More public education is in order, he added.
"I think there’s a lot to be done to model what the cardiologists have done: They’ve convinced people that high cholesterol and high blood pressure are actually diseases. In fact, they’re only risk factors, but by taking drugs to reduce those risk factors there’s been a major effect. We need to make people aware there are effective ways of reducing the risk of breast cancer by more than 50%. The toxicities are limited, and if you have toxicity you simply stop treatment," he said.
The IBIS-II study was funded by Cancer Research U.K., the National Health and Medical Research Council of Australia, AstraZeneca, and Sanofi-Aventis. Dr. Cuzick is on the speaker’s bureau for AstraZeneca.
Simultaneous with Dr. Cuzick’s presentation in San Antonio, the IBIS-II results were published online in the Lancet (2013 Dec. 12 [doi:10.1016/S0140-6736(13)62292-8]).
AT SABCS 2013
Major finding: The 7-year cumulative incidence of all breast cancers in high-risk postmenopausal women who took anastrozole once daily for 5 years was 2.8%, representing a 53% reduction in risk compared with the 5.6% rate in controls. The number needed to treat for 5 years to prevent one breast cancer in 7 years was 36 women.
Data source: The IBIS-II trial was a randomized, double-blind, placebo-controlled study in which 3,864 postmenopausal women at high risk for breast cancer were assigned to 1 mg/day of anastrozole or placebo for 5 years.
Disclosures: The study was funded by Cancer Research U.K., the National Health and Medical Research Council of Australia, AstraZeneca, and Sanofi-Aventis. The presenter is on the speaker’s bureau for AstraZeneca.
VIDEO: Tumor-infiltrating lymphocytes may predict trastuzumab success
SAN ANTONIO – High levels of tumor-infiltrating lymphocytes in primary breast cancer are a good biomarker for favorable clinical response to trastuzumab, according to research presented at the San Antonio Breast Cancer Symposium.
In an exclusive interview with Frontline Medical News, Dr. Sherene Loi discusses the findings' clinical implications, including the potential that breast cancers could be amenable to immunotherapeutic approaches.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
SAN ANTONIO – High levels of tumor-infiltrating lymphocytes in primary breast cancer are a good biomarker for favorable clinical response to trastuzumab, according to research presented at the San Antonio Breast Cancer Symposium.
In an exclusive interview with Frontline Medical News, Dr. Sherene Loi discusses the findings' clinical implications, including the potential that breast cancers could be amenable to immunotherapeutic approaches.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
SAN ANTONIO – High levels of tumor-infiltrating lymphocytes in primary breast cancer are a good biomarker for favorable clinical response to trastuzumab, according to research presented at the San Antonio Breast Cancer Symposium.
In an exclusive interview with Frontline Medical News, Dr. Sherene Loi discusses the findings' clinical implications, including the potential that breast cancers could be amenable to immunotherapeutic approaches.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Meta-analysis: Statins beneficial, even after age 75
DALLAS – Reassurance regarding the cardiovascular benefits of statin therapy in the elderly, even in those above age 75, is provided by a new meta-analysis by the international Cholesterol Treatment Trialists’ Collaboration.
The meta-analysis, which included 174,099 participants in 27 major, published, randomized controlled trials with a median follow-up of 4.9 years, should go a long way toward banishing physician and patient uncertainty about the appropriateness of statin therapy in the elderly. It’s evident that such uncertainty is widespread from recent studies indicating only about half of patients over age 65 are on statin therapy post myocardial infarction (MI). Moreover, the controversial new prevention guidelines don’t address the use of statins in patients over age 75, citing a lack of persuasive evidence because such patients were often excluded from participation in the major statin trials (J. Am. Coll. Cardiol. 2013 [doi: 10.1016/j.jacc.2013.11.002]).
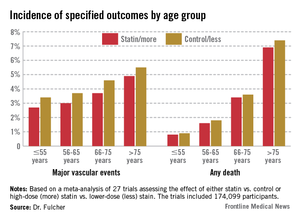
Yet in the new meta-analysis by the University of Oxford–based Cholesterol Treatment Trialist's Collaboration, 7% of all participants – that’s nearly 13,000 patients – were over age 75. That’s a large enough number to be able to draw tentative conclusions. In addition, another 33% of subjects in the meta-analysis were aged 66-75 years, Dr. Jordan Fulcher observed in presenting the results at the American Heart Association scientific sessions.
Dividing the nearly 175,000 subjects into four age groups – 55 and younger, 56-65, 66-75, and over 75 – it quickly became apparent to the investigators that while statin therapy significantly reduced nonfatal MI, cardiovascular death, all-cause mortality, and major vascular events in each of the four age groups, there was also a significant trend for smaller relative risk reductions with advancing age.
For example, the incidence of nonfatal MI or coronary heart disease (CHD) death in statin-treated patients aged 55 years or younger was 1.1%, compared with 1.5% in controls, for a 31% relative risk reduction per 39 mg/dL decrease in low-density lipoprotein (LDL) cholesterol, while in the over-75 group the rates were 2.8% versus 3.3%, for a less robust 24% relative risk reduction, reported Dr. Jordan Fulcher of the University of Sydney, Australia.
Looking at major vascular events, which is a broader composite outcome composed of nonfatal MI, CHD death, stroke, or coronary revascularization, patients aged 55 years or less who achieved a 39-mg/dL reduction in LDL had a 25% reduction compared with controls. This relative risk reduction was whittled down to 15% in the over-75s after adjustment for baseline differences in hypertension, gender, diabetes, prior cardiovascular disease, smoking, and creatinine clearance.
Nonetheless – and this is a key study finding – because of the increasing absolute risk of major events with advancing age, the number needed to treat in order to prevent one additional event is "almost identical" across all age groups, according to Dr. Fulcher.
"We therefore conclude that elderly patients at risk should be considered equally as younger patients for statin therapy," he said.
The meta-analysis showed no evidence of favorable or adverse effects of statin therapy on cancer incidence or mortality or on nonvascular mortality in any age group.
Session chair Dr. Lori Mosca said that "as a card-carrying epidemiologist," she thinks it’s important to emphasize for the benefit of nonepidemiologists the truism that relative risk reductions for effective therapies get smaller as people get older because the background rate of disease goes up.
"Relative risk is used for etiology. Absolute risk is used for treatment decisions. I don’t really care if the relative risk [reduction] gets lower as we get older. That has nothing to do with the importance of statins in older patients. It could potentially be more important to treat the elderly despite a lower relative risk" reduction, said Dr. Mosca, professor of medicine at Columbia University, New York, and director of preventive cardiology at New York–Presbyterian Hospital.
The Cholesterol Treatment Trialists’ Collaboration was established back in 1994 in early recognition that no single statin megatrial would have sufficient size and statistical power to answer all the key future clinical issues to arise. The trialists’ database includes virtually all the landmark statin trials whose acronyms are household names within medicine.
Funding for the trialists’ ongoing work is provided by the U.K. Medical Research Council and other national health research organizations. Dr. Fulcher reported having no financial conflicts of interest.
DALLAS – Reassurance regarding the cardiovascular benefits of statin therapy in the elderly, even in those above age 75, is provided by a new meta-analysis by the international Cholesterol Treatment Trialists’ Collaboration.
The meta-analysis, which included 174,099 participants in 27 major, published, randomized controlled trials with a median follow-up of 4.9 years, should go a long way toward banishing physician and patient uncertainty about the appropriateness of statin therapy in the elderly. It’s evident that such uncertainty is widespread from recent studies indicating only about half of patients over age 65 are on statin therapy post myocardial infarction (MI). Moreover, the controversial new prevention guidelines don’t address the use of statins in patients over age 75, citing a lack of persuasive evidence because such patients were often excluded from participation in the major statin trials (J. Am. Coll. Cardiol. 2013 [doi: 10.1016/j.jacc.2013.11.002]).

Yet in the new meta-analysis by the University of Oxford–based Cholesterol Treatment Trialist's Collaboration, 7% of all participants – that’s nearly 13,000 patients – were over age 75. That’s a large enough number to be able to draw tentative conclusions. In addition, another 33% of subjects in the meta-analysis were aged 66-75 years, Dr. Jordan Fulcher observed in presenting the results at the American Heart Association scientific sessions.
Dividing the nearly 175,000 subjects into four age groups – 55 and younger, 56-65, 66-75, and over 75 – it quickly became apparent to the investigators that while statin therapy significantly reduced nonfatal MI, cardiovascular death, all-cause mortality, and major vascular events in each of the four age groups, there was also a significant trend for smaller relative risk reductions with advancing age.
For example, the incidence of nonfatal MI or coronary heart disease (CHD) death in statin-treated patients aged 55 years or younger was 1.1%, compared with 1.5% in controls, for a 31% relative risk reduction per 39 mg/dL decrease in low-density lipoprotein (LDL) cholesterol, while in the over-75 group the rates were 2.8% versus 3.3%, for a less robust 24% relative risk reduction, reported Dr. Jordan Fulcher of the University of Sydney, Australia.
Looking at major vascular events, which is a broader composite outcome composed of nonfatal MI, CHD death, stroke, or coronary revascularization, patients aged 55 years or less who achieved a 39-mg/dL reduction in LDL had a 25% reduction compared with controls. This relative risk reduction was whittled down to 15% in the over-75s after adjustment for baseline differences in hypertension, gender, diabetes, prior cardiovascular disease, smoking, and creatinine clearance.
Nonetheless – and this is a key study finding – because of the increasing absolute risk of major events with advancing age, the number needed to treat in order to prevent one additional event is "almost identical" across all age groups, according to Dr. Fulcher.
"We therefore conclude that elderly patients at risk should be considered equally as younger patients for statin therapy," he said.
The meta-analysis showed no evidence of favorable or adverse effects of statin therapy on cancer incidence or mortality or on nonvascular mortality in any age group.
Session chair Dr. Lori Mosca said that "as a card-carrying epidemiologist," she thinks it’s important to emphasize for the benefit of nonepidemiologists the truism that relative risk reductions for effective therapies get smaller as people get older because the background rate of disease goes up.
"Relative risk is used for etiology. Absolute risk is used for treatment decisions. I don’t really care if the relative risk [reduction] gets lower as we get older. That has nothing to do with the importance of statins in older patients. It could potentially be more important to treat the elderly despite a lower relative risk" reduction, said Dr. Mosca, professor of medicine at Columbia University, New York, and director of preventive cardiology at New York–Presbyterian Hospital.
The Cholesterol Treatment Trialists’ Collaboration was established back in 1994 in early recognition that no single statin megatrial would have sufficient size and statistical power to answer all the key future clinical issues to arise. The trialists’ database includes virtually all the landmark statin trials whose acronyms are household names within medicine.
Funding for the trialists’ ongoing work is provided by the U.K. Medical Research Council and other national health research organizations. Dr. Fulcher reported having no financial conflicts of interest.
DALLAS – Reassurance regarding the cardiovascular benefits of statin therapy in the elderly, even in those above age 75, is provided by a new meta-analysis by the international Cholesterol Treatment Trialists’ Collaboration.
The meta-analysis, which included 174,099 participants in 27 major, published, randomized controlled trials with a median follow-up of 4.9 years, should go a long way toward banishing physician and patient uncertainty about the appropriateness of statin therapy in the elderly. It’s evident that such uncertainty is widespread from recent studies indicating only about half of patients over age 65 are on statin therapy post myocardial infarction (MI). Moreover, the controversial new prevention guidelines don’t address the use of statins in patients over age 75, citing a lack of persuasive evidence because such patients were often excluded from participation in the major statin trials (J. Am. Coll. Cardiol. 2013 [doi: 10.1016/j.jacc.2013.11.002]).

Yet in the new meta-analysis by the University of Oxford–based Cholesterol Treatment Trialist's Collaboration, 7% of all participants – that’s nearly 13,000 patients – were over age 75. That’s a large enough number to be able to draw tentative conclusions. In addition, another 33% of subjects in the meta-analysis were aged 66-75 years, Dr. Jordan Fulcher observed in presenting the results at the American Heart Association scientific sessions.
Dividing the nearly 175,000 subjects into four age groups – 55 and younger, 56-65, 66-75, and over 75 – it quickly became apparent to the investigators that while statin therapy significantly reduced nonfatal MI, cardiovascular death, all-cause mortality, and major vascular events in each of the four age groups, there was also a significant trend for smaller relative risk reductions with advancing age.
For example, the incidence of nonfatal MI or coronary heart disease (CHD) death in statin-treated patients aged 55 years or younger was 1.1%, compared with 1.5% in controls, for a 31% relative risk reduction per 39 mg/dL decrease in low-density lipoprotein (LDL) cholesterol, while in the over-75 group the rates were 2.8% versus 3.3%, for a less robust 24% relative risk reduction, reported Dr. Jordan Fulcher of the University of Sydney, Australia.
Looking at major vascular events, which is a broader composite outcome composed of nonfatal MI, CHD death, stroke, or coronary revascularization, patients aged 55 years or less who achieved a 39-mg/dL reduction in LDL had a 25% reduction compared with controls. This relative risk reduction was whittled down to 15% in the over-75s after adjustment for baseline differences in hypertension, gender, diabetes, prior cardiovascular disease, smoking, and creatinine clearance.
Nonetheless – and this is a key study finding – because of the increasing absolute risk of major events with advancing age, the number needed to treat in order to prevent one additional event is "almost identical" across all age groups, according to Dr. Fulcher.
"We therefore conclude that elderly patients at risk should be considered equally as younger patients for statin therapy," he said.
The meta-analysis showed no evidence of favorable or adverse effects of statin therapy on cancer incidence or mortality or on nonvascular mortality in any age group.
Session chair Dr. Lori Mosca said that "as a card-carrying epidemiologist," she thinks it’s important to emphasize for the benefit of nonepidemiologists the truism that relative risk reductions for effective therapies get smaller as people get older because the background rate of disease goes up.
"Relative risk is used for etiology. Absolute risk is used for treatment decisions. I don’t really care if the relative risk [reduction] gets lower as we get older. That has nothing to do with the importance of statins in older patients. It could potentially be more important to treat the elderly despite a lower relative risk" reduction, said Dr. Mosca, professor of medicine at Columbia University, New York, and director of preventive cardiology at New York–Presbyterian Hospital.
The Cholesterol Treatment Trialists’ Collaboration was established back in 1994 in early recognition that no single statin megatrial would have sufficient size and statistical power to answer all the key future clinical issues to arise. The trialists’ database includes virtually all the landmark statin trials whose acronyms are household names within medicine.
Funding for the trialists’ ongoing work is provided by the U.K. Medical Research Council and other national health research organizations. Dr. Fulcher reported having no financial conflicts of interest.
AT THE AHA SCIENTIFIC SESSIONS
Major finding: While the relative risk reduction provided by statin therapy in patients over age 75 is smaller than in younger age groups, the number needed to treat in order to prevent one major vascular event is virtually identical across the full age spectrum due to the increasing absolute risk of vascular disease with advancing age.
Data source: A meta-analysis of 27 major randomized controlled clinical trials of statin therapy with 174,099 participants.
Disclosures: Funding for the trialists’ ongoing work is provided by the U.K. Medical Research Council and other national health research organizations. Dr. Fulcher reported having no financial conflicts of interest.
Radiotherapy can be omitted for many older breast cancer patients
SAN ANTONIO – Avoiding whole-breast radiation therapy is a reasonable – and even attractive – option for many older women with early-stage breast cancer, according to the results of the Postoperative Radiotherapy in Minimum-Risk Elderly (PRIME II) trial.
The patient population identified in PRIME II as being suitable for omission of postoperative radiotherapy on the basis of a relatively benign natural history consists of women aged 65 or older who are on adjuvant hormone therapy after undergoing lumpectomy with clear margins for estrogen receptor–rich, axillary node–negative breast cancer.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
PRIME II was a six-country trial in which 1,326 patients 65 or older with hormone receptor–positive early breast cancer were randomized to radiotherapy or no radiotherapy following breast-conserving surgery and endocrine therapy. The 5-year actuarial rate of ipsilateral breast cancer recurrence – the primary study endpoint – was 1.3% in those who received radiotherapy and 4.1% in those who did not, Dr. Ian H. Kunkler reported at the San Antonio Breast Cancer Symposium.
The 5-year actuarial rate of overall survival was 94.2% in patients randomized to radiotherapy and closely similar at 93.8% in the no-radiotherapy group, added Dr. Kunkler, professor of clinical oncology at the University of Edinburgh.
The relative benefit of radiotherapy was even smaller in the 91% of subjects who had estrogen-rich tumors as defined by an ER score of at least 7. They had a local recurrence rate of 3.2% with radiotherapy and 0.8% without. While that absolute 2.4% difference was statistically significant, it is arguably not clinically meaningful. For every 100 women who fit the description carefully defined in PRIME II and who undergo radiotherapy, three will have a recurrence prevented, one will have a recurrence anyway, and 96 will have had treatment that was not beneficial, he said.
"I think we’re really at the cusp of overtreatment here. I think it’s a matter for discussion between the physician and patient as to whether that very modest benefit is worth the potential complications of radiotherapy and the burdens of ongoing treatment, as well as the costs to the health service. Older patients find radiotherapy very burdensome, the relative benefits are very small, and there is no compromise in terms of overall survival with its omission," Dr. Kunkler said.
An important caveat: Among the 9% of patients with low estrogen receptor status, the local recurrence rate was 11.1% with no radiotherapy compared to zero with radiation.
"This is a group for whom radiotherapy should not be omitted," Dr. Kunkler declared.
More than one-half of all early breast cancers present in women aged 65 or older. While postoperative radiotherapy after lumpectomy has been the standard of care regardless of age and other risk factors, there has been only sparse high-quality supporting evidence for this practice in older patients.
Dr. Kunkler estimated that the PRIME II findings are generalizable to 60%-70% of all breast cancer patients over age 65. He predicted that the PRIME II study will "very likely" alter practice in the United Kingdom, and symposium codirector Dr. C. Kent Osborne predicted that the study will be practice changing in the United States as well.
"When I was in training, everybody thought that more was better: more drug treatment, more radiation, more surgery, high-dose chemotherapy, and bone marrow transplant. As we’ve evolved over the last 3 decades, that’s turning out not to be the case. I think we’re gradually doing less and less treatment, either with radiotherapy or with surgery, to control the local disease in appropriate patients. And I think more and more people will begin to accept it," said Dr. Osborne, director of the Dan L. Duncan Cancer Center and the Lester and Sue Smith Breast Center at Baylor College of Medicine, Houston.
PRIME II was funded by the Chief Scientist Office for Scotland. Dr. Kunkler declared having no conflicts of interest.
SAN ANTONIO – Avoiding whole-breast radiation therapy is a reasonable – and even attractive – option for many older women with early-stage breast cancer, according to the results of the Postoperative Radiotherapy in Minimum-Risk Elderly (PRIME II) trial.
The patient population identified in PRIME II as being suitable for omission of postoperative radiotherapy on the basis of a relatively benign natural history consists of women aged 65 or older who are on adjuvant hormone therapy after undergoing lumpectomy with clear margins for estrogen receptor–rich, axillary node–negative breast cancer.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
PRIME II was a six-country trial in which 1,326 patients 65 or older with hormone receptor–positive early breast cancer were randomized to radiotherapy or no radiotherapy following breast-conserving surgery and endocrine therapy. The 5-year actuarial rate of ipsilateral breast cancer recurrence – the primary study endpoint – was 1.3% in those who received radiotherapy and 4.1% in those who did not, Dr. Ian H. Kunkler reported at the San Antonio Breast Cancer Symposium.
The 5-year actuarial rate of overall survival was 94.2% in patients randomized to radiotherapy and closely similar at 93.8% in the no-radiotherapy group, added Dr. Kunkler, professor of clinical oncology at the University of Edinburgh.
The relative benefit of radiotherapy was even smaller in the 91% of subjects who had estrogen-rich tumors as defined by an ER score of at least 7. They had a local recurrence rate of 3.2% with radiotherapy and 0.8% without. While that absolute 2.4% difference was statistically significant, it is arguably not clinically meaningful. For every 100 women who fit the description carefully defined in PRIME II and who undergo radiotherapy, three will have a recurrence prevented, one will have a recurrence anyway, and 96 will have had treatment that was not beneficial, he said.
"I think we’re really at the cusp of overtreatment here. I think it’s a matter for discussion between the physician and patient as to whether that very modest benefit is worth the potential complications of radiotherapy and the burdens of ongoing treatment, as well as the costs to the health service. Older patients find radiotherapy very burdensome, the relative benefits are very small, and there is no compromise in terms of overall survival with its omission," Dr. Kunkler said.
An important caveat: Among the 9% of patients with low estrogen receptor status, the local recurrence rate was 11.1% with no radiotherapy compared to zero with radiation.
"This is a group for whom radiotherapy should not be omitted," Dr. Kunkler declared.
More than one-half of all early breast cancers present in women aged 65 or older. While postoperative radiotherapy after lumpectomy has been the standard of care regardless of age and other risk factors, there has been only sparse high-quality supporting evidence for this practice in older patients.
Dr. Kunkler estimated that the PRIME II findings are generalizable to 60%-70% of all breast cancer patients over age 65. He predicted that the PRIME II study will "very likely" alter practice in the United Kingdom, and symposium codirector Dr. C. Kent Osborne predicted that the study will be practice changing in the United States as well.
"When I was in training, everybody thought that more was better: more drug treatment, more radiation, more surgery, high-dose chemotherapy, and bone marrow transplant. As we’ve evolved over the last 3 decades, that’s turning out not to be the case. I think we’re gradually doing less and less treatment, either with radiotherapy or with surgery, to control the local disease in appropriate patients. And I think more and more people will begin to accept it," said Dr. Osborne, director of the Dan L. Duncan Cancer Center and the Lester and Sue Smith Breast Center at Baylor College of Medicine, Houston.
PRIME II was funded by the Chief Scientist Office for Scotland. Dr. Kunkler declared having no conflicts of interest.
SAN ANTONIO – Avoiding whole-breast radiation therapy is a reasonable – and even attractive – option for many older women with early-stage breast cancer, according to the results of the Postoperative Radiotherapy in Minimum-Risk Elderly (PRIME II) trial.
The patient population identified in PRIME II as being suitable for omission of postoperative radiotherapy on the basis of a relatively benign natural history consists of women aged 65 or older who are on adjuvant hormone therapy after undergoing lumpectomy with clear margins for estrogen receptor–rich, axillary node–negative breast cancer.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
PRIME II was a six-country trial in which 1,326 patients 65 or older with hormone receptor–positive early breast cancer were randomized to radiotherapy or no radiotherapy following breast-conserving surgery and endocrine therapy. The 5-year actuarial rate of ipsilateral breast cancer recurrence – the primary study endpoint – was 1.3% in those who received radiotherapy and 4.1% in those who did not, Dr. Ian H. Kunkler reported at the San Antonio Breast Cancer Symposium.
The 5-year actuarial rate of overall survival was 94.2% in patients randomized to radiotherapy and closely similar at 93.8% in the no-radiotherapy group, added Dr. Kunkler, professor of clinical oncology at the University of Edinburgh.
The relative benefit of radiotherapy was even smaller in the 91% of subjects who had estrogen-rich tumors as defined by an ER score of at least 7. They had a local recurrence rate of 3.2% with radiotherapy and 0.8% without. While that absolute 2.4% difference was statistically significant, it is arguably not clinically meaningful. For every 100 women who fit the description carefully defined in PRIME II and who undergo radiotherapy, three will have a recurrence prevented, one will have a recurrence anyway, and 96 will have had treatment that was not beneficial, he said.
"I think we’re really at the cusp of overtreatment here. I think it’s a matter for discussion between the physician and patient as to whether that very modest benefit is worth the potential complications of radiotherapy and the burdens of ongoing treatment, as well as the costs to the health service. Older patients find radiotherapy very burdensome, the relative benefits are very small, and there is no compromise in terms of overall survival with its omission," Dr. Kunkler said.
An important caveat: Among the 9% of patients with low estrogen receptor status, the local recurrence rate was 11.1% with no radiotherapy compared to zero with radiation.
"This is a group for whom radiotherapy should not be omitted," Dr. Kunkler declared.
More than one-half of all early breast cancers present in women aged 65 or older. While postoperative radiotherapy after lumpectomy has been the standard of care regardless of age and other risk factors, there has been only sparse high-quality supporting evidence for this practice in older patients.
Dr. Kunkler estimated that the PRIME II findings are generalizable to 60%-70% of all breast cancer patients over age 65. He predicted that the PRIME II study will "very likely" alter practice in the United Kingdom, and symposium codirector Dr. C. Kent Osborne predicted that the study will be practice changing in the United States as well.
"When I was in training, everybody thought that more was better: more drug treatment, more radiation, more surgery, high-dose chemotherapy, and bone marrow transplant. As we’ve evolved over the last 3 decades, that’s turning out not to be the case. I think we’re gradually doing less and less treatment, either with radiotherapy or with surgery, to control the local disease in appropriate patients. And I think more and more people will begin to accept it," said Dr. Osborne, director of the Dan L. Duncan Cancer Center and the Lester and Sue Smith Breast Center at Baylor College of Medicine, Houston.
PRIME II was funded by the Chief Scientist Office for Scotland. Dr. Kunkler declared having no conflicts of interest.
AT SABCS 2013
Major finding: The 5-year ipsilateral breast cancer recurrence rate in a selected population of older women undergoing breast-conserving surgery and adjuvant hormone therapy was 1.3% with postoperative radiotherapy and 4.1% without it, a modest difference that did not impact overall survival.
Data source: A prospective randomized trial in six countries, involving 1,326 patients aged 65 or older who underwent lumpectomy with clear margins for hormone receptor–positive, axillary node–negative breast cancer and were on adjuvant endocrine therapy. They were randomized to postoperative radiotherapy or no radiotherapy.
Disclosures: The PRIME II study was funded by the Chief Scientist Office for Scotland. The presenter reported having no financial conflicts.
Statin in childhood reduces CHD risk in familial hypercholesterolemia
DALLAS – Roughly 10 years after going on statin therapy as children or adolescents, a group of Dutch patients with familial hypercholesterolemia already has a significantly lower cardiovascular event rate than their affected parents did at the same age.
"Statin therapy in childhood seems to be effective in reducing CHD [coronary heart disease] risk in individuals with FH [familial hypercholesterolemia], although longer follow-up is needed to confirm these results," Dr. Marjet J.A.M. Braamskamp declared at the American Heart Association scientific sessions.
The 214 Dutch youths with FH started on statin therapy at age 8-18 years as part of a randomized clinical trial. None has experienced a cardiovascular event by age 30. In contrast, their affected parents, for whom statins weren’t available until they were well into adulthood, already had a 7% CHD event rate by age 30, a statistically significant difference, reported Dr. Braamskamp of the Academic Medical Center, Amsterdam.
In an interview, she said the plan is to continue to follow this cohort of young adults who started statin therapy in childhood at least until age 50. By that age, 55% of their fathers with FH and 24% of their mothers with FH had known CHD. While none of the FH mothers died of CHD before age 60, 16% of the FH fathers did, at an average age of 35.9 years.
Current European and American guidelines recommend statin therapy starting at age 8 or 10 years in children with the molecular or clinical diagnosis of FH and elevated cholesterol levels. Those guidelines were issued based upon recognition that children with FH have elevated cholesterol levels from birth onward, coupled with evidence from short-term randomized trials demonstrating that statins are safe and effective for lipid-lowering in such patients. The Dutch follow-up study provides evidence to suggest this early-treatment strategy also pays off in terms of reduced CHD risk.
The study was supported by the Dutch Heart Foundation. Dr. Braamskamp reported having no financial conflicts.
DALLAS – Roughly 10 years after going on statin therapy as children or adolescents, a group of Dutch patients with familial hypercholesterolemia already has a significantly lower cardiovascular event rate than their affected parents did at the same age.
"Statin therapy in childhood seems to be effective in reducing CHD [coronary heart disease] risk in individuals with FH [familial hypercholesterolemia], although longer follow-up is needed to confirm these results," Dr. Marjet J.A.M. Braamskamp declared at the American Heart Association scientific sessions.
The 214 Dutch youths with FH started on statin therapy at age 8-18 years as part of a randomized clinical trial. None has experienced a cardiovascular event by age 30. In contrast, their affected parents, for whom statins weren’t available until they were well into adulthood, already had a 7% CHD event rate by age 30, a statistically significant difference, reported Dr. Braamskamp of the Academic Medical Center, Amsterdam.
In an interview, she said the plan is to continue to follow this cohort of young adults who started statin therapy in childhood at least until age 50. By that age, 55% of their fathers with FH and 24% of their mothers with FH had known CHD. While none of the FH mothers died of CHD before age 60, 16% of the FH fathers did, at an average age of 35.9 years.
Current European and American guidelines recommend statin therapy starting at age 8 or 10 years in children with the molecular or clinical diagnosis of FH and elevated cholesterol levels. Those guidelines were issued based upon recognition that children with FH have elevated cholesterol levels from birth onward, coupled with evidence from short-term randomized trials demonstrating that statins are safe and effective for lipid-lowering in such patients. The Dutch follow-up study provides evidence to suggest this early-treatment strategy also pays off in terms of reduced CHD risk.
The study was supported by the Dutch Heart Foundation. Dr. Braamskamp reported having no financial conflicts.
DALLAS – Roughly 10 years after going on statin therapy as children or adolescents, a group of Dutch patients with familial hypercholesterolemia already has a significantly lower cardiovascular event rate than their affected parents did at the same age.
"Statin therapy in childhood seems to be effective in reducing CHD [coronary heart disease] risk in individuals with FH [familial hypercholesterolemia], although longer follow-up is needed to confirm these results," Dr. Marjet J.A.M. Braamskamp declared at the American Heart Association scientific sessions.
The 214 Dutch youths with FH started on statin therapy at age 8-18 years as part of a randomized clinical trial. None has experienced a cardiovascular event by age 30. In contrast, their affected parents, for whom statins weren’t available until they were well into adulthood, already had a 7% CHD event rate by age 30, a statistically significant difference, reported Dr. Braamskamp of the Academic Medical Center, Amsterdam.
In an interview, she said the plan is to continue to follow this cohort of young adults who started statin therapy in childhood at least until age 50. By that age, 55% of their fathers with FH and 24% of their mothers with FH had known CHD. While none of the FH mothers died of CHD before age 60, 16% of the FH fathers did, at an average age of 35.9 years.
Current European and American guidelines recommend statin therapy starting at age 8 or 10 years in children with the molecular or clinical diagnosis of FH and elevated cholesterol levels. Those guidelines were issued based upon recognition that children with FH have elevated cholesterol levels from birth onward, coupled with evidence from short-term randomized trials demonstrating that statins are safe and effective for lipid-lowering in such patients. The Dutch follow-up study provides evidence to suggest this early-treatment strategy also pays off in terms of reduced CHD risk.
The study was supported by the Dutch Heart Foundation. Dr. Braamskamp reported having no financial conflicts.
AT THE AHA SCIENTIFIC SESSIONS
Major finding: At age 30, the cardiovascular event rate in 214 Dutch patients with FH who began statin therapy as children or adolescents was zero, significantly lower than the 7% rate in their affected parents at the same age, who didn’t have access to statin therapy.
Data source: This was a follow-up report on 214 young adults with familial hypercholesterolemia.
Disclosures: The study was funded by the Dutch Heart Foundation. Dr. Braamskamp reported having no financial conflicts.
Anakinra showed benefits in diastolic heart failure
DALLAS – Interleukin-1 blockade resulted in significantly improved exercise capacity and a corresponding reduction in systemic inflammation in patients with heart failure with preserved ejection fraction in the small, pilot D-HART study.
"These results confirm the role of IL-1 in regulating cardiovascular function and provide the first evidence that an IL-1-targeted therapeutic strategy may be valuable in HFpEF [heart failure with preserved ejection fraction]," Benjamin W. Van Tassell, Pharm.D., declared at the American Heart Association scientific sessions.
The findings are welcome given that there are no approved therapies for HFpEF, which accounts for at least half of all cases of heart failure, noted Dr. Van Tassell of Virginia Commonwealth University, Richmond.
D-HART was a randomized, double-blind, placebo-controlled study involving 12 patients with symptomatic HFpEF and high levels of systemic inflammation as reflected in their elevated baseline high-sensitivity C-reactive protein levels. Patients were placed on 14 days of daily subcutaneous injections of the IL-1 antagonist anakinra (Kineret) at 100 mg or placebo, then crossed over to the other study arm. Half of the subjects had New York Heart Association (NYHA) class II heart failure, the other half class III.
Since heart failure is a syndrome of exercise intolerance secondary to impaired cardiac function, Dr. Van Tassell and his coinvestigators chose change in peak oxygen consumption (VO2) as the primary study endpoint. The median peak VO2 during the anakinra phase of the study was 16.3 mL/kg per minute, which was a 1.2–mL/kg per minute or 8% improvement over the peak value on placebo.
The improvement in peak VO2 correlated with a sharp reduction in high-sensitivity C-reactive protein (hsCRP), a surrogate marker for IL-1 activity. From a baseline of 7.5 mg/L, hsCRP levels plunged by 84% after 14 days on anakinra compared to 10% with placebo.
Ventilatory efficiency (VE) improved with IL-1 blockade as evidenced by a significant increase in the oxygen uptake efficiency slope. However, the VE/VCO2 slope – another measure of ventilatory efficiency – improved significantly with anakinra only in the five patients with above-median values at baseline. Their 12% improvement in this metric during IL-1 inhibition implies that maximum benefit occurs in patients with impaired baseline function, according to Dr. Van Tassell.
He added that these encouraging findings from D-HART provide a sound basis for larger confirmatory randomized trials evaluating IL-1 inhibition in patients with HFpEF. Anakinra is approved for the treatment of rheumatoid arthritis.
D-HART was supported by the American Heart Association and university funds. Dr. Van Tassell reported having no financial conflicts of interest.
DALLAS – Interleukin-1 blockade resulted in significantly improved exercise capacity and a corresponding reduction in systemic inflammation in patients with heart failure with preserved ejection fraction in the small, pilot D-HART study.
"These results confirm the role of IL-1 in regulating cardiovascular function and provide the first evidence that an IL-1-targeted therapeutic strategy may be valuable in HFpEF [heart failure with preserved ejection fraction]," Benjamin W. Van Tassell, Pharm.D., declared at the American Heart Association scientific sessions.
The findings are welcome given that there are no approved therapies for HFpEF, which accounts for at least half of all cases of heart failure, noted Dr. Van Tassell of Virginia Commonwealth University, Richmond.
D-HART was a randomized, double-blind, placebo-controlled study involving 12 patients with symptomatic HFpEF and high levels of systemic inflammation as reflected in their elevated baseline high-sensitivity C-reactive protein levels. Patients were placed on 14 days of daily subcutaneous injections of the IL-1 antagonist anakinra (Kineret) at 100 mg or placebo, then crossed over to the other study arm. Half of the subjects had New York Heart Association (NYHA) class II heart failure, the other half class III.
Since heart failure is a syndrome of exercise intolerance secondary to impaired cardiac function, Dr. Van Tassell and his coinvestigators chose change in peak oxygen consumption (VO2) as the primary study endpoint. The median peak VO2 during the anakinra phase of the study was 16.3 mL/kg per minute, which was a 1.2–mL/kg per minute or 8% improvement over the peak value on placebo.
The improvement in peak VO2 correlated with a sharp reduction in high-sensitivity C-reactive protein (hsCRP), a surrogate marker for IL-1 activity. From a baseline of 7.5 mg/L, hsCRP levels plunged by 84% after 14 days on anakinra compared to 10% with placebo.
Ventilatory efficiency (VE) improved with IL-1 blockade as evidenced by a significant increase in the oxygen uptake efficiency slope. However, the VE/VCO2 slope – another measure of ventilatory efficiency – improved significantly with anakinra only in the five patients with above-median values at baseline. Their 12% improvement in this metric during IL-1 inhibition implies that maximum benefit occurs in patients with impaired baseline function, according to Dr. Van Tassell.
He added that these encouraging findings from D-HART provide a sound basis for larger confirmatory randomized trials evaluating IL-1 inhibition in patients with HFpEF. Anakinra is approved for the treatment of rheumatoid arthritis.
D-HART was supported by the American Heart Association and university funds. Dr. Van Tassell reported having no financial conflicts of interest.
DALLAS – Interleukin-1 blockade resulted in significantly improved exercise capacity and a corresponding reduction in systemic inflammation in patients with heart failure with preserved ejection fraction in the small, pilot D-HART study.
"These results confirm the role of IL-1 in regulating cardiovascular function and provide the first evidence that an IL-1-targeted therapeutic strategy may be valuable in HFpEF [heart failure with preserved ejection fraction]," Benjamin W. Van Tassell, Pharm.D., declared at the American Heart Association scientific sessions.
The findings are welcome given that there are no approved therapies for HFpEF, which accounts for at least half of all cases of heart failure, noted Dr. Van Tassell of Virginia Commonwealth University, Richmond.
D-HART was a randomized, double-blind, placebo-controlled study involving 12 patients with symptomatic HFpEF and high levels of systemic inflammation as reflected in their elevated baseline high-sensitivity C-reactive protein levels. Patients were placed on 14 days of daily subcutaneous injections of the IL-1 antagonist anakinra (Kineret) at 100 mg or placebo, then crossed over to the other study arm. Half of the subjects had New York Heart Association (NYHA) class II heart failure, the other half class III.
Since heart failure is a syndrome of exercise intolerance secondary to impaired cardiac function, Dr. Van Tassell and his coinvestigators chose change in peak oxygen consumption (VO2) as the primary study endpoint. The median peak VO2 during the anakinra phase of the study was 16.3 mL/kg per minute, which was a 1.2–mL/kg per minute or 8% improvement over the peak value on placebo.
The improvement in peak VO2 correlated with a sharp reduction in high-sensitivity C-reactive protein (hsCRP), a surrogate marker for IL-1 activity. From a baseline of 7.5 mg/L, hsCRP levels plunged by 84% after 14 days on anakinra compared to 10% with placebo.
Ventilatory efficiency (VE) improved with IL-1 blockade as evidenced by a significant increase in the oxygen uptake efficiency slope. However, the VE/VCO2 slope – another measure of ventilatory efficiency – improved significantly with anakinra only in the five patients with above-median values at baseline. Their 12% improvement in this metric during IL-1 inhibition implies that maximum benefit occurs in patients with impaired baseline function, according to Dr. Van Tassell.
He added that these encouraging findings from D-HART provide a sound basis for larger confirmatory randomized trials evaluating IL-1 inhibition in patients with HFpEF. Anakinra is approved for the treatment of rheumatoid arthritis.
D-HART was supported by the American Heart Association and university funds. Dr. Van Tassell reported having no financial conflicts of interest.
AT THE AHA SCIENTIFIC SESSIONS
Major finding: Patients with heart failure with preserved ejection fraction showed a significant 8% placebo-subtracted gain in peak oxygen consumption after 14 days of interleukin-1 inhibition via daily anakinra. This was accompanied by an 84% reduction in baseline elevated high-sensitivity C-reactive protein levels.
Data source: D-HART, a randomized, double-blind pilot study in which 12 patients with heart failure with preserved ejection fraction were placed on 14 days of daily subcutaneous injections of anakinra at 100 mg or placebo, then crossed to 14 days of the alternative.
Disclosures: D-HART was supported by the American Heart Association and university funds. Dr. Van Tassell reported having no financial conflicts of interest.
Healthy diet after diabetes diagnosis improves survival
DALLAS – Women who embraced a healthy diet after being diagnosed with type 2 diabetes had significantly lower rates of all-cause and cardiovascular mortality than did those with a bad diet, in a large observational study with up to 26 years of follow-up.
"It’s never too late to improve your diet," Dr. Hyun Joon Shin said in presenting his analysis of data from the Nurses’ Health Study at the American Heart Association scientific sessions.
The prospective observational Nurses’ Health Study began in 1976. It included 121,700 female American nurses aged 30-55 upon enrollment. Dr. Shin’s analysis included 8,354 women with no baseline cardiovascular disease, diabetes, or cancer who were diagnosed with type 2 diabetes during 1984-2006 and followed to June 2010.
During follow-up, there were 1,183 deaths in the cohort diagnosed with type 2 diabetes, including 491 deaths due to cardiovascular disease and 514 due to cancer.
Mortality risks were evaluated in relation to the extent to which the nurses adhered to a healthy, high-quality diet following their diagnosis of type 2 diabetes. The yardstick employed in evaluating eating patterns was the Alternative Healthy Eating Index–2010 (AHEI). The index, developed by researchers at Harvard University, scores patients’ consumption of various foods and nutrients known to be predictive of chronic disease risk, explained Dr. Shin of Baylor University Medical Center, Dallas.
There were 14.4 deaths per 1,000 persons per year in the top quintile in terms of adherence to the AHEI. A dose-response relationship was evident: The mortality rate was 28.8/1,000 persons per year among those in the bottom quintile, 23.1/1,000 in the second quintile, 19.3 in the third, and 20.7 in the fourth.
In a multivariate analysis adjusted for numerous potential confounders, including a participant’s prediabetic AHEI score, the diabetic nurses in the top quintile in terms of healthy eating had a 52% reduction in the risk of all-cause mortality compared with those in the lowest AHEI quintile. The healthiest eaters also had a 49% lower risk of cardiovascular mortality. However, their more modest 28% reduction in the risk of cancer-related death fell just short of statistical significance.
The AHEI, updated in 2010, awards points for increased consumption of whole grains, nuts, legumes, vegetables, fruits, fish or fish oil, and moderate alcohol intake. The index penalizes for consumption of red meat, processed meat products, trans fats, sodium, sugar-sweetened beverages, and fruit juices (J. Nutr. 2012;142:1009-18).
In examining the study findings more closely in terms of the impact of individual dietary components of the AHEI, Dr. Shin found nearly all of them had significant effects in multivariate analyses. For example, subjects in the top quintile for whole grain consumption had a 41% reduction in the risk of all-cause mortality compared with those in the bottom quintile, as well as a 33% reduction in the risk of cardiovascular mortality. Participants who drank the least amount of sugar-sweetened beverages had reductions in all-cause and cardiovascular mortality of 26% and 37%, compared with these rates for the quintile of biggest quaffers.
This was the first-ever postdiabetes dietary pattern analysis examining mortality in women with incident type 2 diabetes, according to Dr. Shin. The mechanism for the observed inverse relationship between diet quality and mortality is straightforward: A poor-quality diet is associated with increased risks of coronary heart disease, stroke, and weight gain and higher circulating levels of fasting insulin, inflammatory cytokines, and leptin, he said.
D. Shin’s study was supported by Baylor University research funding. He reported having no relevant financial conflicts.
DALLAS – Women who embraced a healthy diet after being diagnosed with type 2 diabetes had significantly lower rates of all-cause and cardiovascular mortality than did those with a bad diet, in a large observational study with up to 26 years of follow-up.
"It’s never too late to improve your diet," Dr. Hyun Joon Shin said in presenting his analysis of data from the Nurses’ Health Study at the American Heart Association scientific sessions.
The prospective observational Nurses’ Health Study began in 1976. It included 121,700 female American nurses aged 30-55 upon enrollment. Dr. Shin’s analysis included 8,354 women with no baseline cardiovascular disease, diabetes, or cancer who were diagnosed with type 2 diabetes during 1984-2006 and followed to June 2010.
During follow-up, there were 1,183 deaths in the cohort diagnosed with type 2 diabetes, including 491 deaths due to cardiovascular disease and 514 due to cancer.
Mortality risks were evaluated in relation to the extent to which the nurses adhered to a healthy, high-quality diet following their diagnosis of type 2 diabetes. The yardstick employed in evaluating eating patterns was the Alternative Healthy Eating Index–2010 (AHEI). The index, developed by researchers at Harvard University, scores patients’ consumption of various foods and nutrients known to be predictive of chronic disease risk, explained Dr. Shin of Baylor University Medical Center, Dallas.
There were 14.4 deaths per 1,000 persons per year in the top quintile in terms of adherence to the AHEI. A dose-response relationship was evident: The mortality rate was 28.8/1,000 persons per year among those in the bottom quintile, 23.1/1,000 in the second quintile, 19.3 in the third, and 20.7 in the fourth.
In a multivariate analysis adjusted for numerous potential confounders, including a participant’s prediabetic AHEI score, the diabetic nurses in the top quintile in terms of healthy eating had a 52% reduction in the risk of all-cause mortality compared with those in the lowest AHEI quintile. The healthiest eaters also had a 49% lower risk of cardiovascular mortality. However, their more modest 28% reduction in the risk of cancer-related death fell just short of statistical significance.
The AHEI, updated in 2010, awards points for increased consumption of whole grains, nuts, legumes, vegetables, fruits, fish or fish oil, and moderate alcohol intake. The index penalizes for consumption of red meat, processed meat products, trans fats, sodium, sugar-sweetened beverages, and fruit juices (J. Nutr. 2012;142:1009-18).
In examining the study findings more closely in terms of the impact of individual dietary components of the AHEI, Dr. Shin found nearly all of them had significant effects in multivariate analyses. For example, subjects in the top quintile for whole grain consumption had a 41% reduction in the risk of all-cause mortality compared with those in the bottom quintile, as well as a 33% reduction in the risk of cardiovascular mortality. Participants who drank the least amount of sugar-sweetened beverages had reductions in all-cause and cardiovascular mortality of 26% and 37%, compared with these rates for the quintile of biggest quaffers.
This was the first-ever postdiabetes dietary pattern analysis examining mortality in women with incident type 2 diabetes, according to Dr. Shin. The mechanism for the observed inverse relationship between diet quality and mortality is straightforward: A poor-quality diet is associated with increased risks of coronary heart disease, stroke, and weight gain and higher circulating levels of fasting insulin, inflammatory cytokines, and leptin, he said.
D. Shin’s study was supported by Baylor University research funding. He reported having no relevant financial conflicts.
DALLAS – Women who embraced a healthy diet after being diagnosed with type 2 diabetes had significantly lower rates of all-cause and cardiovascular mortality than did those with a bad diet, in a large observational study with up to 26 years of follow-up.
"It’s never too late to improve your diet," Dr. Hyun Joon Shin said in presenting his analysis of data from the Nurses’ Health Study at the American Heart Association scientific sessions.
The prospective observational Nurses’ Health Study began in 1976. It included 121,700 female American nurses aged 30-55 upon enrollment. Dr. Shin’s analysis included 8,354 women with no baseline cardiovascular disease, diabetes, or cancer who were diagnosed with type 2 diabetes during 1984-2006 and followed to June 2010.
During follow-up, there were 1,183 deaths in the cohort diagnosed with type 2 diabetes, including 491 deaths due to cardiovascular disease and 514 due to cancer.
Mortality risks were evaluated in relation to the extent to which the nurses adhered to a healthy, high-quality diet following their diagnosis of type 2 diabetes. The yardstick employed in evaluating eating patterns was the Alternative Healthy Eating Index–2010 (AHEI). The index, developed by researchers at Harvard University, scores patients’ consumption of various foods and nutrients known to be predictive of chronic disease risk, explained Dr. Shin of Baylor University Medical Center, Dallas.
There were 14.4 deaths per 1,000 persons per year in the top quintile in terms of adherence to the AHEI. A dose-response relationship was evident: The mortality rate was 28.8/1,000 persons per year among those in the bottom quintile, 23.1/1,000 in the second quintile, 19.3 in the third, and 20.7 in the fourth.
In a multivariate analysis adjusted for numerous potential confounders, including a participant’s prediabetic AHEI score, the diabetic nurses in the top quintile in terms of healthy eating had a 52% reduction in the risk of all-cause mortality compared with those in the lowest AHEI quintile. The healthiest eaters also had a 49% lower risk of cardiovascular mortality. However, their more modest 28% reduction in the risk of cancer-related death fell just short of statistical significance.
The AHEI, updated in 2010, awards points for increased consumption of whole grains, nuts, legumes, vegetables, fruits, fish or fish oil, and moderate alcohol intake. The index penalizes for consumption of red meat, processed meat products, trans fats, sodium, sugar-sweetened beverages, and fruit juices (J. Nutr. 2012;142:1009-18).
In examining the study findings more closely in terms of the impact of individual dietary components of the AHEI, Dr. Shin found nearly all of them had significant effects in multivariate analyses. For example, subjects in the top quintile for whole grain consumption had a 41% reduction in the risk of all-cause mortality compared with those in the bottom quintile, as well as a 33% reduction in the risk of cardiovascular mortality. Participants who drank the least amount of sugar-sweetened beverages had reductions in all-cause and cardiovascular mortality of 26% and 37%, compared with these rates for the quintile of biggest quaffers.
This was the first-ever postdiabetes dietary pattern analysis examining mortality in women with incident type 2 diabetes, according to Dr. Shin. The mechanism for the observed inverse relationship between diet quality and mortality is straightforward: A poor-quality diet is associated with increased risks of coronary heart disease, stroke, and weight gain and higher circulating levels of fasting insulin, inflammatory cytokines, and leptin, he said.
D. Shin’s study was supported by Baylor University research funding. He reported having no relevant financial conflicts.
AT THE AHA SCIENTIFIC SESSIONS
Major finding: Women who placed in the top quintile for a healthy eating pattern following their diagnosis of type 2 diabetes had all-cause and cardiovascular mortality rates 52% and 49% lower, respectively, than did women in the bottom quintile for healthy eating.
Data source: An analysis of long-term mortality trends in relation to eating habits in 8,354 women in the prospective Nurses’ Health Study who were diagnosed with type 2 diabetes and followed for a maximum of 26 years.
Disclosures: The study was supported by a Baylor University research grant. The presenter reported having no relevant financial conflicts.
Statin reduces MI risk in ischemic heart failure
DALLAS – Rosuvastatin appears to reduce the risk of acute myocardial infarction in patients with ischemic heart failure, according to a meta-analysis of two landmark statin trials.
This finding is at odds with current thinking as expressed in the new American Heart Association/American College of Cardiology (AHA/ACC) guidelines, which don’t specifically recommend statin therapy for heart failure patients because no large randomized trials have shown a significant reduction in cardiovascular events.
That’s probably because those studies relied on traditional statistical techniques of survival analysis rather than the less familiar method of competing risk analysis, which is better suited to detecting a statin benefit in heart failure patients who are simultaneously at risk for multiple types of events, including arrhythmias, cancer, infection, and renal failure, as well as ischemic events, Dr. Matthew Feinstein explained at the American Heart Association scientific sessions.
When he and his coinvestigators applied competing risk analysis in their meta-analysis of the randomized, placebo-controlled CORONA (N. Engl. J. Med. 2007; 357:2248-61) and GISSI-HF trials, they found after accounting for competing risks in close to 10,000 heart failure patients that those on 10 mg/day of rosuvastatin had a 17% reduction in the risk of myocardial infarction during follow-up, compared with those on placebo. This benefit fell just shy of statistical significance. However, when the researchers restricted the analysis to the nearly 7,100 subjects with ischemic heart failure, the resultant 19% reduction in MI risk with rosuvastatin did achieve significance.
While there was a consistent trend for a lower MI risk with rosuvastatin in all prespecified subgroups of patients with ischemic heart failure in the pooled analysis, this trend reached statistical significance in two subgroups: men, who were 22% less likely to have an MI on rosuvastatin than with placebo, and patients with a baseline low-density lipoprotein (LDL) cholesterol above 96 mg/dL, who had a 23% reduction in risk, reported Dr. Feinstein of Northwestern University, Chicago, and his colleagues.
"It’s likely that ischemic heart failure patients do benefit from statin therapy, contrary to current thinking, if they are likely to live long enough to derive benefit from statins in preventing atherothrombotic events," he concluded.
Session chair Dr. Lori Mosca was enthusiastic about the meta-analysis findings and their clinical implications. She also was strongly supportive of the application of competing risk analysis in this setting.
"That was actually a brilliant analysis. I really loved it. The lesson it teaches is that when we design a clinical trial where our nonischemic outcomes are more common than the ischemic outcomes that we’re trying to prevent, we really need to take a look at those competing risks. We might come to completely different conclusions if we do that," said Dr. Mosca, professor of medicine at Columbia University, New York, and director of preventive cardiology at New York-Presbyterian Hospital.
Other important take-home points from this pooled analysis of the CORONA and GISSI-HF trials are that despite all the confusion surrounding the new AHA/ACC prevention guidelines, LDL cholesterol levels still matter in terms of predicting outcomes, and that, as always, it’s important to look at gender interactions in looking at clinical trial outcomes, added Dr. Mosca, who is current chair of the AHA Expert Panel for the Effectiveness-Based Guidelines for the Prevention of Cardiovascular Disease in Women.
Noting that it’s not always possible for a physician to tell whether the cause of a patient’s heart failure is ischemic or nonischemic, she asked Dr. Feinstein if he’d prescribe a statin if he merely suspected that the heart failure was ischemic, perhaps because the patient had evidence of underlying coronary disease.
Yes, he replied, provided the patient had no contraindications to statin therapy, was able to tolerate it, and had a sufficiently long life expectancy to be able to reap the benefits. He added a caveat: Heart failure patients typically have multiple comorbid conditions that could place them at risk with higher doses of statins.
"Rosuvastatin tends to be a somewhat less toxic and better tolerated statin, and 10 mg is a relatively low dose," Dr. Feinstein noted.
The meta-analysis was conducted free of commercial sponsorship. Dr. Feinstein reported having no financial conflicts of interest.
DALLAS – Rosuvastatin appears to reduce the risk of acute myocardial infarction in patients with ischemic heart failure, according to a meta-analysis of two landmark statin trials.
This finding is at odds with current thinking as expressed in the new American Heart Association/American College of Cardiology (AHA/ACC) guidelines, which don’t specifically recommend statin therapy for heart failure patients because no large randomized trials have shown a significant reduction in cardiovascular events.
That’s probably because those studies relied on traditional statistical techniques of survival analysis rather than the less familiar method of competing risk analysis, which is better suited to detecting a statin benefit in heart failure patients who are simultaneously at risk for multiple types of events, including arrhythmias, cancer, infection, and renal failure, as well as ischemic events, Dr. Matthew Feinstein explained at the American Heart Association scientific sessions.
When he and his coinvestigators applied competing risk analysis in their meta-analysis of the randomized, placebo-controlled CORONA (N. Engl. J. Med. 2007; 357:2248-61) and GISSI-HF trials, they found after accounting for competing risks in close to 10,000 heart failure patients that those on 10 mg/day of rosuvastatin had a 17% reduction in the risk of myocardial infarction during follow-up, compared with those on placebo. This benefit fell just shy of statistical significance. However, when the researchers restricted the analysis to the nearly 7,100 subjects with ischemic heart failure, the resultant 19% reduction in MI risk with rosuvastatin did achieve significance.
While there was a consistent trend for a lower MI risk with rosuvastatin in all prespecified subgroups of patients with ischemic heart failure in the pooled analysis, this trend reached statistical significance in two subgroups: men, who were 22% less likely to have an MI on rosuvastatin than with placebo, and patients with a baseline low-density lipoprotein (LDL) cholesterol above 96 mg/dL, who had a 23% reduction in risk, reported Dr. Feinstein of Northwestern University, Chicago, and his colleagues.
"It’s likely that ischemic heart failure patients do benefit from statin therapy, contrary to current thinking, if they are likely to live long enough to derive benefit from statins in preventing atherothrombotic events," he concluded.
Session chair Dr. Lori Mosca was enthusiastic about the meta-analysis findings and their clinical implications. She also was strongly supportive of the application of competing risk analysis in this setting.
"That was actually a brilliant analysis. I really loved it. The lesson it teaches is that when we design a clinical trial where our nonischemic outcomes are more common than the ischemic outcomes that we’re trying to prevent, we really need to take a look at those competing risks. We might come to completely different conclusions if we do that," said Dr. Mosca, professor of medicine at Columbia University, New York, and director of preventive cardiology at New York-Presbyterian Hospital.
Other important take-home points from this pooled analysis of the CORONA and GISSI-HF trials are that despite all the confusion surrounding the new AHA/ACC prevention guidelines, LDL cholesterol levels still matter in terms of predicting outcomes, and that, as always, it’s important to look at gender interactions in looking at clinical trial outcomes, added Dr. Mosca, who is current chair of the AHA Expert Panel for the Effectiveness-Based Guidelines for the Prevention of Cardiovascular Disease in Women.
Noting that it’s not always possible for a physician to tell whether the cause of a patient’s heart failure is ischemic or nonischemic, she asked Dr. Feinstein if he’d prescribe a statin if he merely suspected that the heart failure was ischemic, perhaps because the patient had evidence of underlying coronary disease.
Yes, he replied, provided the patient had no contraindications to statin therapy, was able to tolerate it, and had a sufficiently long life expectancy to be able to reap the benefits. He added a caveat: Heart failure patients typically have multiple comorbid conditions that could place them at risk with higher doses of statins.
"Rosuvastatin tends to be a somewhat less toxic and better tolerated statin, and 10 mg is a relatively low dose," Dr. Feinstein noted.
The meta-analysis was conducted free of commercial sponsorship. Dr. Feinstein reported having no financial conflicts of interest.
DALLAS – Rosuvastatin appears to reduce the risk of acute myocardial infarction in patients with ischemic heart failure, according to a meta-analysis of two landmark statin trials.
This finding is at odds with current thinking as expressed in the new American Heart Association/American College of Cardiology (AHA/ACC) guidelines, which don’t specifically recommend statin therapy for heart failure patients because no large randomized trials have shown a significant reduction in cardiovascular events.
That’s probably because those studies relied on traditional statistical techniques of survival analysis rather than the less familiar method of competing risk analysis, which is better suited to detecting a statin benefit in heart failure patients who are simultaneously at risk for multiple types of events, including arrhythmias, cancer, infection, and renal failure, as well as ischemic events, Dr. Matthew Feinstein explained at the American Heart Association scientific sessions.
When he and his coinvestigators applied competing risk analysis in their meta-analysis of the randomized, placebo-controlled CORONA (N. Engl. J. Med. 2007; 357:2248-61) and GISSI-HF trials, they found after accounting for competing risks in close to 10,000 heart failure patients that those on 10 mg/day of rosuvastatin had a 17% reduction in the risk of myocardial infarction during follow-up, compared with those on placebo. This benefit fell just shy of statistical significance. However, when the researchers restricted the analysis to the nearly 7,100 subjects with ischemic heart failure, the resultant 19% reduction in MI risk with rosuvastatin did achieve significance.
While there was a consistent trend for a lower MI risk with rosuvastatin in all prespecified subgroups of patients with ischemic heart failure in the pooled analysis, this trend reached statistical significance in two subgroups: men, who were 22% less likely to have an MI on rosuvastatin than with placebo, and patients with a baseline low-density lipoprotein (LDL) cholesterol above 96 mg/dL, who had a 23% reduction in risk, reported Dr. Feinstein of Northwestern University, Chicago, and his colleagues.
"It’s likely that ischemic heart failure patients do benefit from statin therapy, contrary to current thinking, if they are likely to live long enough to derive benefit from statins in preventing atherothrombotic events," he concluded.
Session chair Dr. Lori Mosca was enthusiastic about the meta-analysis findings and their clinical implications. She also was strongly supportive of the application of competing risk analysis in this setting.
"That was actually a brilliant analysis. I really loved it. The lesson it teaches is that when we design a clinical trial where our nonischemic outcomes are more common than the ischemic outcomes that we’re trying to prevent, we really need to take a look at those competing risks. We might come to completely different conclusions if we do that," said Dr. Mosca, professor of medicine at Columbia University, New York, and director of preventive cardiology at New York-Presbyterian Hospital.
Other important take-home points from this pooled analysis of the CORONA and GISSI-HF trials are that despite all the confusion surrounding the new AHA/ACC prevention guidelines, LDL cholesterol levels still matter in terms of predicting outcomes, and that, as always, it’s important to look at gender interactions in looking at clinical trial outcomes, added Dr. Mosca, who is current chair of the AHA Expert Panel for the Effectiveness-Based Guidelines for the Prevention of Cardiovascular Disease in Women.
Noting that it’s not always possible for a physician to tell whether the cause of a patient’s heart failure is ischemic or nonischemic, she asked Dr. Feinstein if he’d prescribe a statin if he merely suspected that the heart failure was ischemic, perhaps because the patient had evidence of underlying coronary disease.
Yes, he replied, provided the patient had no contraindications to statin therapy, was able to tolerate it, and had a sufficiently long life expectancy to be able to reap the benefits. He added a caveat: Heart failure patients typically have multiple comorbid conditions that could place them at risk with higher doses of statins.
"Rosuvastatin tends to be a somewhat less toxic and better tolerated statin, and 10 mg is a relatively low dose," Dr. Feinstein noted.
The meta-analysis was conducted free of commercial sponsorship. Dr. Feinstein reported having no financial conflicts of interest.
AT THE AHA SCIENTIFIC SESSIONS
Major finding: Rosuvastatin at 10 mg/day resulted in a significant 19% reduction in the risk of MI, compared with placebo, in patients with ischemic heart failure.
Data source: A post hoc meta-analysis of two landmark, randomized, placebo-controlled clinical trials – the CORONA and GISSI-HF trials – totaling nearly 10,000 subjects with heart failure followed prospectively for a median of 33 and 47 months, respectively.
Disclosures: The meta-analysis was conducted free of commercial sponsorship. Dr. Feinstein reported having no financial conflicts of interest.
Body fat and cardiovascular risk: Location, location, location!
DALLAS – It’s not obesity per se that affects cardiovascular risk, it’s where that excess body fat is stored, according to the results of a novel adipose tissue–imaging study.
Excess visceral adipose tissue is independently associated with increased risk of developing cardiovascular disease. In contrast, increased lower body subcutaneous adipose tissue – that is, fat around the hips and a big butt – actually seems to protect against cardiovascular disease, Dr. Ian J. Neeland reported at the American Heart Association scientific sessions.
He presented an analysis of 972 obese participants in the Dallas Heart Study with a mean age of 44 years at enrollment and no baseline cardiovascular disease. All underwent dual-energy x-ray absorptiometry and MRI assessment of their body fat distribution, focusing on the visceral, abdominal subcutaneous, and lower body subcutaneous adipose tissue depots. Participants were then followed prospectively for a median 8.1 years, during which 91 cardiovascular events occurred in 68 subjects.
The impetus for the adipose tissue imaging study was the researchers’ recognition that obesity is a heterogeneous disorder.
"Currently, we know that obesity is associated with incident cardiovascular disease at a general population level. However, body mass index alone is really an inadequate marker of risk among the obese. Many individuals with even high BMIs do not develop cardiovascular disease. Marked abdominal obesity is a stronger predictor of cardiovascular disease but still lacks the necessary specificity. So there’s really a clinical need for tools to differentiate obese individuals who will develop cardiovascular disease from those who will be free of cardiovascular disease for their lifetime," explained Dr. Neeland, a fellow in cardiovascular medicine at the University of Texas Southwestern Medical Center, Dallas.
In a multivariate analysis adjusted for age, sex, race, and the conventional cardiovascular risk factors, each 1–standard deviation increase in visceral adipose tissue was independently associated with a 24% increase in the risk of developing cardiovascular disease during follow-up. Dividing the study population into quartiles on the basis of their extent of visceral adipose tissue, the cumulative incidence of cardiovascular disease rose in stepwise fashion, with subjects in the lowest quartile having the least cardiovascular events and those in the top quartile having the most.
Having more lower body subcutaneous fat had the opposite effect. For every 1–standard deviation increase in the amount of fat at that location, the cardiovascular event risk dropped by 27%.
Participants’ amount of abdominal subcutaneous fat didn’t affect their cardiovascular event risk one way or another. Nor did BMI, waist circumference, waist-hip ratio, or the amount of liver fat on MRI show any significant association with cardiovascular disease risk.
"These results really underscore the biologic importance of body fat distribution with regard to cardiovascular disease risk in obesity and suggest a possible prognostic role for imaging-based assessment of body fat distribution in high-risk obese patients," according to Dr. Neeland.
One intriguing clinical implication of this study is that preventing accumulation of visceral adipose tissue may have benefit in terms of cardiovascular disease prevention even in the absence of meaningful weight loss. It’s possible that new drugs could be developed that lower cardiovascular risk in obese patients by changing their body fat distribution profile rather than lopping off pounds.
In the Dallas study, increased visceral abdominal tissue was consistently associated with a higher risk of cardiovascular disease across subgroups based upon age, race, sex, and BMI.
"Interestingly, those with increased visceral abdominal fat who were less than 40 years of age had greater risk for cardiovascular disease than [did] those over 40. This could suggest that visceral abdominal tissue has a greater impact on the young, in whom other cardiovascular risk factors have not yet accumulated over time," Dr. Neeland observed.
The cardiovascular event endpoint in the study was a composite of cardiovascular death, acute MI, stroke, heart failure, atrial fibrillation, or event-driven coronary or peripheral artery revascularization.
The Dallas Heart Study is funded by the Donald W. Reynolds Foundation. Dr. Neeland reported having no financial conflicts.
DALLAS – It’s not obesity per se that affects cardiovascular risk, it’s where that excess body fat is stored, according to the results of a novel adipose tissue–imaging study.
Excess visceral adipose tissue is independently associated with increased risk of developing cardiovascular disease. In contrast, increased lower body subcutaneous adipose tissue – that is, fat around the hips and a big butt – actually seems to protect against cardiovascular disease, Dr. Ian J. Neeland reported at the American Heart Association scientific sessions.
He presented an analysis of 972 obese participants in the Dallas Heart Study with a mean age of 44 years at enrollment and no baseline cardiovascular disease. All underwent dual-energy x-ray absorptiometry and MRI assessment of their body fat distribution, focusing on the visceral, abdominal subcutaneous, and lower body subcutaneous adipose tissue depots. Participants were then followed prospectively for a median 8.1 years, during which 91 cardiovascular events occurred in 68 subjects.
The impetus for the adipose tissue imaging study was the researchers’ recognition that obesity is a heterogeneous disorder.
"Currently, we know that obesity is associated with incident cardiovascular disease at a general population level. However, body mass index alone is really an inadequate marker of risk among the obese. Many individuals with even high BMIs do not develop cardiovascular disease. Marked abdominal obesity is a stronger predictor of cardiovascular disease but still lacks the necessary specificity. So there’s really a clinical need for tools to differentiate obese individuals who will develop cardiovascular disease from those who will be free of cardiovascular disease for their lifetime," explained Dr. Neeland, a fellow in cardiovascular medicine at the University of Texas Southwestern Medical Center, Dallas.
In a multivariate analysis adjusted for age, sex, race, and the conventional cardiovascular risk factors, each 1–standard deviation increase in visceral adipose tissue was independently associated with a 24% increase in the risk of developing cardiovascular disease during follow-up. Dividing the study population into quartiles on the basis of their extent of visceral adipose tissue, the cumulative incidence of cardiovascular disease rose in stepwise fashion, with subjects in the lowest quartile having the least cardiovascular events and those in the top quartile having the most.
Having more lower body subcutaneous fat had the opposite effect. For every 1–standard deviation increase in the amount of fat at that location, the cardiovascular event risk dropped by 27%.
Participants’ amount of abdominal subcutaneous fat didn’t affect their cardiovascular event risk one way or another. Nor did BMI, waist circumference, waist-hip ratio, or the amount of liver fat on MRI show any significant association with cardiovascular disease risk.
"These results really underscore the biologic importance of body fat distribution with regard to cardiovascular disease risk in obesity and suggest a possible prognostic role for imaging-based assessment of body fat distribution in high-risk obese patients," according to Dr. Neeland.
One intriguing clinical implication of this study is that preventing accumulation of visceral adipose tissue may have benefit in terms of cardiovascular disease prevention even in the absence of meaningful weight loss. It’s possible that new drugs could be developed that lower cardiovascular risk in obese patients by changing their body fat distribution profile rather than lopping off pounds.
In the Dallas study, increased visceral abdominal tissue was consistently associated with a higher risk of cardiovascular disease across subgroups based upon age, race, sex, and BMI.
"Interestingly, those with increased visceral abdominal fat who were less than 40 years of age had greater risk for cardiovascular disease than [did] those over 40. This could suggest that visceral abdominal tissue has a greater impact on the young, in whom other cardiovascular risk factors have not yet accumulated over time," Dr. Neeland observed.
The cardiovascular event endpoint in the study was a composite of cardiovascular death, acute MI, stroke, heart failure, atrial fibrillation, or event-driven coronary or peripheral artery revascularization.
The Dallas Heart Study is funded by the Donald W. Reynolds Foundation. Dr. Neeland reported having no financial conflicts.
DALLAS – It’s not obesity per se that affects cardiovascular risk, it’s where that excess body fat is stored, according to the results of a novel adipose tissue–imaging study.
Excess visceral adipose tissue is independently associated with increased risk of developing cardiovascular disease. In contrast, increased lower body subcutaneous adipose tissue – that is, fat around the hips and a big butt – actually seems to protect against cardiovascular disease, Dr. Ian J. Neeland reported at the American Heart Association scientific sessions.
He presented an analysis of 972 obese participants in the Dallas Heart Study with a mean age of 44 years at enrollment and no baseline cardiovascular disease. All underwent dual-energy x-ray absorptiometry and MRI assessment of their body fat distribution, focusing on the visceral, abdominal subcutaneous, and lower body subcutaneous adipose tissue depots. Participants were then followed prospectively for a median 8.1 years, during which 91 cardiovascular events occurred in 68 subjects.
The impetus for the adipose tissue imaging study was the researchers’ recognition that obesity is a heterogeneous disorder.
"Currently, we know that obesity is associated with incident cardiovascular disease at a general population level. However, body mass index alone is really an inadequate marker of risk among the obese. Many individuals with even high BMIs do not develop cardiovascular disease. Marked abdominal obesity is a stronger predictor of cardiovascular disease but still lacks the necessary specificity. So there’s really a clinical need for tools to differentiate obese individuals who will develop cardiovascular disease from those who will be free of cardiovascular disease for their lifetime," explained Dr. Neeland, a fellow in cardiovascular medicine at the University of Texas Southwestern Medical Center, Dallas.
In a multivariate analysis adjusted for age, sex, race, and the conventional cardiovascular risk factors, each 1–standard deviation increase in visceral adipose tissue was independently associated with a 24% increase in the risk of developing cardiovascular disease during follow-up. Dividing the study population into quartiles on the basis of their extent of visceral adipose tissue, the cumulative incidence of cardiovascular disease rose in stepwise fashion, with subjects in the lowest quartile having the least cardiovascular events and those in the top quartile having the most.
Having more lower body subcutaneous fat had the opposite effect. For every 1–standard deviation increase in the amount of fat at that location, the cardiovascular event risk dropped by 27%.
Participants’ amount of abdominal subcutaneous fat didn’t affect their cardiovascular event risk one way or another. Nor did BMI, waist circumference, waist-hip ratio, or the amount of liver fat on MRI show any significant association with cardiovascular disease risk.
"These results really underscore the biologic importance of body fat distribution with regard to cardiovascular disease risk in obesity and suggest a possible prognostic role for imaging-based assessment of body fat distribution in high-risk obese patients," according to Dr. Neeland.
One intriguing clinical implication of this study is that preventing accumulation of visceral adipose tissue may have benefit in terms of cardiovascular disease prevention even in the absence of meaningful weight loss. It’s possible that new drugs could be developed that lower cardiovascular risk in obese patients by changing their body fat distribution profile rather than lopping off pounds.
In the Dallas study, increased visceral abdominal tissue was consistently associated with a higher risk of cardiovascular disease across subgroups based upon age, race, sex, and BMI.
"Interestingly, those with increased visceral abdominal fat who were less than 40 years of age had greater risk for cardiovascular disease than [did] those over 40. This could suggest that visceral abdominal tissue has a greater impact on the young, in whom other cardiovascular risk factors have not yet accumulated over time," Dr. Neeland observed.
The cardiovascular event endpoint in the study was a composite of cardiovascular death, acute MI, stroke, heart failure, atrial fibrillation, or event-driven coronary or peripheral artery revascularization.
The Dallas Heart Study is funded by the Donald W. Reynolds Foundation. Dr. Neeland reported having no financial conflicts.
AT THE AHA SCIENTIFIC SESSIONS
Major finding: Excess visceral adipose tissue in obese adults was independently associated with increased risk of subsequent cardiovascular disease, while excess lower body subcutaneous fat had a protective effect.
Data source: This report involved 972 obese participants in the prospective, single-center Dallas Heart Study, all without baseline cardiovascular disease who underwent baseline imaging of specific adipose tissue depots.
Disclosures: The Dallas Heart Study is funded by the Donald W. Reynolds Foundation. The presenter reported having no relevant financial interests.
Bromocriptine-QR slashes cardiovascular events in diabetics on metformin
DALLAS – The quick-release formulation of bromocriptine known as Cycloset shows strong potential as a novel approach for cardiovascular risk reduction in type 2 diabetic patients already on metformin, according to Dr. Bindu Chamarthi.
A post-hoc analysis of 1,791 such patients who participated in the 1-year double-blind Cycloset Safety Trial showed a 53% reduction in cardiovascular events in those randomized to bromocriptine-QR, compared with placebo-treated controls, Dr. Chamarthi reported at the American Heart Association scientific sessions.
The composite cardiovascular endpoint comprising a first MI, stroke, coronary revascularization, or hospitalization for either angina or heart failure occurred in 1.3% of 1,208 bromocriptine-QR–treated patients, compared with 3.1% of 583 placebo-treated controls. All subjects were on metformin at baseline, were generally in good metabolic control, and had high rates of utilization of guideline-recommended cardioprotective medications, including statins and antihypertensive agents, noted Dr. Chamarthi, an endocrinologist at Brigham and Women’s Hospital, Boston.
This sharp reduction in cardiovascular events is particularly impressive in light of the increased risk of such events faced by patients with type 2 diabetes, along with the lack of conclusive evidence of cardiovascular benefit for any of the approved therapies for the disease other than metformin, she added.
Bromocriptine-QR is approved by the Food and Drug Administration for the treatment of type 2 diabetes, so it’s not surprising that the group assigned to the drug evidenced improved glycemic control. At the end of 52 weeks of treatment, they were 1.75-fold more likely than controls to be in good glycemic control, as defined by a hemoglobin A1c level of 7.0% or less.
Current evidence suggests that the development of obesity, insulin resistance, and diabetes is linked to a central hypodopaminergic state.
Bromocriptine-QR is a sympatholytic dopamine agonist approved in 2009. Taken once daily with breakfast, it provides a circadian-timed brief period of increased central dopaminergic activity. The result is improved postprandial blood glucose control without raising the insulin concentration.
How bromocriptine-QR achieved the observed reduction in cardiovascular events seen in this study remains unclear. One possibility is that the mechanism involves a reduction in elevated sympathetic nervous system drive to the vasculature, liver, and adipose tissue along with a reduction in hypothalamic-pituitary-adrenal axis activity, with resultant diminution of endothelial dysfunction. These benefits might flow from the drug’s ability to restore the daily morning peak in central circadian dopaminergic neuroendocrine activities that are disrupted in patients with type 2 diabetes. Further studies are planned, according to Dr. Chamarthi.
This study was sponsored by VeroScience, which markets bromocriptine-QR. Dr. Chamarthi is a consultant for the company.
DALLAS – The quick-release formulation of bromocriptine known as Cycloset shows strong potential as a novel approach for cardiovascular risk reduction in type 2 diabetic patients already on metformin, according to Dr. Bindu Chamarthi.
A post-hoc analysis of 1,791 such patients who participated in the 1-year double-blind Cycloset Safety Trial showed a 53% reduction in cardiovascular events in those randomized to bromocriptine-QR, compared with placebo-treated controls, Dr. Chamarthi reported at the American Heart Association scientific sessions.
The composite cardiovascular endpoint comprising a first MI, stroke, coronary revascularization, or hospitalization for either angina or heart failure occurred in 1.3% of 1,208 bromocriptine-QR–treated patients, compared with 3.1% of 583 placebo-treated controls. All subjects were on metformin at baseline, were generally in good metabolic control, and had high rates of utilization of guideline-recommended cardioprotective medications, including statins and antihypertensive agents, noted Dr. Chamarthi, an endocrinologist at Brigham and Women’s Hospital, Boston.
This sharp reduction in cardiovascular events is particularly impressive in light of the increased risk of such events faced by patients with type 2 diabetes, along with the lack of conclusive evidence of cardiovascular benefit for any of the approved therapies for the disease other than metformin, she added.
Bromocriptine-QR is approved by the Food and Drug Administration for the treatment of type 2 diabetes, so it’s not surprising that the group assigned to the drug evidenced improved glycemic control. At the end of 52 weeks of treatment, they were 1.75-fold more likely than controls to be in good glycemic control, as defined by a hemoglobin A1c level of 7.0% or less.
Current evidence suggests that the development of obesity, insulin resistance, and diabetes is linked to a central hypodopaminergic state.
Bromocriptine-QR is a sympatholytic dopamine agonist approved in 2009. Taken once daily with breakfast, it provides a circadian-timed brief period of increased central dopaminergic activity. The result is improved postprandial blood glucose control without raising the insulin concentration.
How bromocriptine-QR achieved the observed reduction in cardiovascular events seen in this study remains unclear. One possibility is that the mechanism involves a reduction in elevated sympathetic nervous system drive to the vasculature, liver, and adipose tissue along with a reduction in hypothalamic-pituitary-adrenal axis activity, with resultant diminution of endothelial dysfunction. These benefits might flow from the drug’s ability to restore the daily morning peak in central circadian dopaminergic neuroendocrine activities that are disrupted in patients with type 2 diabetes. Further studies are planned, according to Dr. Chamarthi.
This study was sponsored by VeroScience, which markets bromocriptine-QR. Dr. Chamarthi is a consultant for the company.
DALLAS – The quick-release formulation of bromocriptine known as Cycloset shows strong potential as a novel approach for cardiovascular risk reduction in type 2 diabetic patients already on metformin, according to Dr. Bindu Chamarthi.
A post-hoc analysis of 1,791 such patients who participated in the 1-year double-blind Cycloset Safety Trial showed a 53% reduction in cardiovascular events in those randomized to bromocriptine-QR, compared with placebo-treated controls, Dr. Chamarthi reported at the American Heart Association scientific sessions.
The composite cardiovascular endpoint comprising a first MI, stroke, coronary revascularization, or hospitalization for either angina or heart failure occurred in 1.3% of 1,208 bromocriptine-QR–treated patients, compared with 3.1% of 583 placebo-treated controls. All subjects were on metformin at baseline, were generally in good metabolic control, and had high rates of utilization of guideline-recommended cardioprotective medications, including statins and antihypertensive agents, noted Dr. Chamarthi, an endocrinologist at Brigham and Women’s Hospital, Boston.
This sharp reduction in cardiovascular events is particularly impressive in light of the increased risk of such events faced by patients with type 2 diabetes, along with the lack of conclusive evidence of cardiovascular benefit for any of the approved therapies for the disease other than metformin, she added.
Bromocriptine-QR is approved by the Food and Drug Administration for the treatment of type 2 diabetes, so it’s not surprising that the group assigned to the drug evidenced improved glycemic control. At the end of 52 weeks of treatment, they were 1.75-fold more likely than controls to be in good glycemic control, as defined by a hemoglobin A1c level of 7.0% or less.
Current evidence suggests that the development of obesity, insulin resistance, and diabetes is linked to a central hypodopaminergic state.
Bromocriptine-QR is a sympatholytic dopamine agonist approved in 2009. Taken once daily with breakfast, it provides a circadian-timed brief period of increased central dopaminergic activity. The result is improved postprandial blood glucose control without raising the insulin concentration.
How bromocriptine-QR achieved the observed reduction in cardiovascular events seen in this study remains unclear. One possibility is that the mechanism involves a reduction in elevated sympathetic nervous system drive to the vasculature, liver, and adipose tissue along with a reduction in hypothalamic-pituitary-adrenal axis activity, with resultant diminution of endothelial dysfunction. These benefits might flow from the drug’s ability to restore the daily morning peak in central circadian dopaminergic neuroendocrine activities that are disrupted in patients with type 2 diabetes. Further studies are planned, according to Dr. Chamarthi.
This study was sponsored by VeroScience, which markets bromocriptine-QR. Dr. Chamarthi is a consultant for the company.
AT THE AHA SCIENTIFIC SESSIONS
Major finding: The 1-year composite cardiovascular event rate in type 2 diabetic patients on metformin at baseline was 1.3% in patients on quick-release bromocriptine and 3.1% in those taking placebo, for a 53% relative risk reduction.
Data source: A secondary post hoc analysis of the randomized, double-blind Cycloset Safety Trial, which was restricted to the 1,791-patient subgroup on metformin at enrollment.
Disclosures: The study was sponsored by VeroScience. The presenter is a consultant to the company, which markets bromocriptine-QR.
















