User login
First-in-man bioengineered graft proves enduring for vascular access
DALLAS – An investigational tissue-engineered vascular graft has enduring potential for vascular access for hemodialysis in patients with end-stage renal disease, based on early clinical results.
Moreover, other potential uses are on the horizon. The big picture involves subsequent extrapolation of this technology from the large-diameter, high-flow bioengineered vessels required for hemodialysis to the creation of small-diameter, low-flow vessels for coronary artery and peripheral arterial graft surgery, Dr. Jeffrey H. Lawson explained at the American Heart Association scientific sessions.
"Our goal is to make a tissue-engineered conduit that could be used widely throughout the body," said Dr. Lawson, professor of surgery and of pathology at Duke University Medical Center, Durham, N.C.
He presented the results from the first-in-man, ongoing phase I clinical experience with the Humacyte graft, which to date has been implanted to provide vascular access for hemodialysis in 28 patients, with 6-month patency as the primary study endpoint. This was a challenging study population, with an average of 4.1 previous access procedure failures per patient. The presentation at the AHA was the first public disclosure of the results of a project Dr. Lawson has been working on for more than 15 years. His surgical colleagues from Poland, who have done the implantations in patients with end-stage renal disease, were in attendance.
The overall 6-month patency was 100%, with no infections, no sign of an immune response, and no aneurysms or other indication of structural degeneration, he said.
Of the 28 patients, 20 had no further interventions, yielding a primary unassisted 6-month patency rate of 71%. Eight patients collectively underwent 10 interventions to maintain patency: eight had thrombectomies for graft- or surgically related thrombosis and two had venous anastomoses. Flow rates have remained suitable for dialysis in all patients, and the grafts are being used for dialysis three times per week. Dr. Lawson described the grafts as easy to cannulate via standard techniques.
He characterized these initial results as "quite remarkable" compared with the outcomes in two large studies of the current benchmark technologies, which are synthetic grafts made of PTFE (polytetrafluoroethyline). In those studies, the primary patency rate at 6 months was less than 50%, with a secondary patency rate of 77% and a 10% infection rate. In other studies, 30%-40% of PTFE grafts are abandoned within 12 months due to loss of patency.
The process of creating the bioengineered grafts begins with harvesting human aortic vascular smooth muscle cells, seeding them on a biodegradable matrix, then culturing them under pulsatile conditions. When the biodegradable matrix melts away, what remains is a tube comprised of vascular smooth muscle cells and extracellular matrix. This is then decellularized, yielding a tube of extracellular matrix that can be shipped off the shelf and around the world.
In primate models, the implanted bioengineered graft has been shown to repopulate with the host’s own vascular smooth muscle cells lined intimally by endothelium.
"Where we implanted an acellular structure, it appears to now be a living tissue, suggesting [the graft] has become their tissue, not ours," Dr. Lawson said.
To date, none of the bioengineered grafts implanted in patients has been explanted, so it’s unknown whether the favorable histologic changes seen in primates’ grafts also occur in humans. Larger clinical trials with longer follow-up are planned in order to assess the bioengineered graft’s durability.
Dr. Lawson’s study is funded by a Department of Defense research grant and by Humacyte. He serves as a consultant to the company.
This work is exciting. The early patency, thrombosis, and infection rates are encouraging.
The unmet clinical need for better ways to provide vascular access for hemodialysis is huge. There are 450,000 U.S. patients with end-stage renal disease on long-term hemodialysis. In this population, hemodialysis access morbidity costs more than $1 billion per year. Although the preferred means of vascular access is an arteriovenous fistula, many hemodialysis patients don’t have suitable veins. And 60% of fistulas become unusable within 6 months.

|
|
We’ve got a conundrum where PTFE grafts have their problems and fistulas have their own problems. We don’t have a good clinical armamentarium.
Synthetic grafts most often lose patency because of venous outflow tract stenosis due to intimal hyperplasia. Balloon angioplasty of the stenotic anastomosis has been the conventional treatment to restore patency, but a landmark randomized trial carried out several years ago (N. Engl. J. Med. 2010;362:494-503) showed the patency rate was a mere 23%, significantly worse than the 51% patency rate with a PTFE-covered stent graft – and even that 51% patency rate, is abysmal.
Dr. Sanjay Misra is professor of radiology at the Mayo Clinic in Rochester, Minn. He was the invited discussant of the paper at the meeting and declared having no relevant financial disclosures.
This work is exciting. The early patency, thrombosis, and infection rates are encouraging.
The unmet clinical need for better ways to provide vascular access for hemodialysis is huge. There are 450,000 U.S. patients with end-stage renal disease on long-term hemodialysis. In this population, hemodialysis access morbidity costs more than $1 billion per year. Although the preferred means of vascular access is an arteriovenous fistula, many hemodialysis patients don’t have suitable veins. And 60% of fistulas become unusable within 6 months.

|
|
We’ve got a conundrum where PTFE grafts have their problems and fistulas have their own problems. We don’t have a good clinical armamentarium.
Synthetic grafts most often lose patency because of venous outflow tract stenosis due to intimal hyperplasia. Balloon angioplasty of the stenotic anastomosis has been the conventional treatment to restore patency, but a landmark randomized trial carried out several years ago (N. Engl. J. Med. 2010;362:494-503) showed the patency rate was a mere 23%, significantly worse than the 51% patency rate with a PTFE-covered stent graft – and even that 51% patency rate, is abysmal.
Dr. Sanjay Misra is professor of radiology at the Mayo Clinic in Rochester, Minn. He was the invited discussant of the paper at the meeting and declared having no relevant financial disclosures.
This work is exciting. The early patency, thrombosis, and infection rates are encouraging.
The unmet clinical need for better ways to provide vascular access for hemodialysis is huge. There are 450,000 U.S. patients with end-stage renal disease on long-term hemodialysis. In this population, hemodialysis access morbidity costs more than $1 billion per year. Although the preferred means of vascular access is an arteriovenous fistula, many hemodialysis patients don’t have suitable veins. And 60% of fistulas become unusable within 6 months.

|
|
We’ve got a conundrum where PTFE grafts have their problems and fistulas have their own problems. We don’t have a good clinical armamentarium.
Synthetic grafts most often lose patency because of venous outflow tract stenosis due to intimal hyperplasia. Balloon angioplasty of the stenotic anastomosis has been the conventional treatment to restore patency, but a landmark randomized trial carried out several years ago (N. Engl. J. Med. 2010;362:494-503) showed the patency rate was a mere 23%, significantly worse than the 51% patency rate with a PTFE-covered stent graft – and even that 51% patency rate, is abysmal.
Dr. Sanjay Misra is professor of radiology at the Mayo Clinic in Rochester, Minn. He was the invited discussant of the paper at the meeting and declared having no relevant financial disclosures.
DALLAS – An investigational tissue-engineered vascular graft has enduring potential for vascular access for hemodialysis in patients with end-stage renal disease, based on early clinical results.
Moreover, other potential uses are on the horizon. The big picture involves subsequent extrapolation of this technology from the large-diameter, high-flow bioengineered vessels required for hemodialysis to the creation of small-diameter, low-flow vessels for coronary artery and peripheral arterial graft surgery, Dr. Jeffrey H. Lawson explained at the American Heart Association scientific sessions.
"Our goal is to make a tissue-engineered conduit that could be used widely throughout the body," said Dr. Lawson, professor of surgery and of pathology at Duke University Medical Center, Durham, N.C.
He presented the results from the first-in-man, ongoing phase I clinical experience with the Humacyte graft, which to date has been implanted to provide vascular access for hemodialysis in 28 patients, with 6-month patency as the primary study endpoint. This was a challenging study population, with an average of 4.1 previous access procedure failures per patient. The presentation at the AHA was the first public disclosure of the results of a project Dr. Lawson has been working on for more than 15 years. His surgical colleagues from Poland, who have done the implantations in patients with end-stage renal disease, were in attendance.
The overall 6-month patency was 100%, with no infections, no sign of an immune response, and no aneurysms or other indication of structural degeneration, he said.
Of the 28 patients, 20 had no further interventions, yielding a primary unassisted 6-month patency rate of 71%. Eight patients collectively underwent 10 interventions to maintain patency: eight had thrombectomies for graft- or surgically related thrombosis and two had venous anastomoses. Flow rates have remained suitable for dialysis in all patients, and the grafts are being used for dialysis three times per week. Dr. Lawson described the grafts as easy to cannulate via standard techniques.
He characterized these initial results as "quite remarkable" compared with the outcomes in two large studies of the current benchmark technologies, which are synthetic grafts made of PTFE (polytetrafluoroethyline). In those studies, the primary patency rate at 6 months was less than 50%, with a secondary patency rate of 77% and a 10% infection rate. In other studies, 30%-40% of PTFE grafts are abandoned within 12 months due to loss of patency.
The process of creating the bioengineered grafts begins with harvesting human aortic vascular smooth muscle cells, seeding them on a biodegradable matrix, then culturing them under pulsatile conditions. When the biodegradable matrix melts away, what remains is a tube comprised of vascular smooth muscle cells and extracellular matrix. This is then decellularized, yielding a tube of extracellular matrix that can be shipped off the shelf and around the world.
In primate models, the implanted bioengineered graft has been shown to repopulate with the host’s own vascular smooth muscle cells lined intimally by endothelium.
"Where we implanted an acellular structure, it appears to now be a living tissue, suggesting [the graft] has become their tissue, not ours," Dr. Lawson said.
To date, none of the bioengineered grafts implanted in patients has been explanted, so it’s unknown whether the favorable histologic changes seen in primates’ grafts also occur in humans. Larger clinical trials with longer follow-up are planned in order to assess the bioengineered graft’s durability.
Dr. Lawson’s study is funded by a Department of Defense research grant and by Humacyte. He serves as a consultant to the company.
DALLAS – An investigational tissue-engineered vascular graft has enduring potential for vascular access for hemodialysis in patients with end-stage renal disease, based on early clinical results.
Moreover, other potential uses are on the horizon. The big picture involves subsequent extrapolation of this technology from the large-diameter, high-flow bioengineered vessels required for hemodialysis to the creation of small-diameter, low-flow vessels for coronary artery and peripheral arterial graft surgery, Dr. Jeffrey H. Lawson explained at the American Heart Association scientific sessions.
"Our goal is to make a tissue-engineered conduit that could be used widely throughout the body," said Dr. Lawson, professor of surgery and of pathology at Duke University Medical Center, Durham, N.C.
He presented the results from the first-in-man, ongoing phase I clinical experience with the Humacyte graft, which to date has been implanted to provide vascular access for hemodialysis in 28 patients, with 6-month patency as the primary study endpoint. This was a challenging study population, with an average of 4.1 previous access procedure failures per patient. The presentation at the AHA was the first public disclosure of the results of a project Dr. Lawson has been working on for more than 15 years. His surgical colleagues from Poland, who have done the implantations in patients with end-stage renal disease, were in attendance.
The overall 6-month patency was 100%, with no infections, no sign of an immune response, and no aneurysms or other indication of structural degeneration, he said.
Of the 28 patients, 20 had no further interventions, yielding a primary unassisted 6-month patency rate of 71%. Eight patients collectively underwent 10 interventions to maintain patency: eight had thrombectomies for graft- or surgically related thrombosis and two had venous anastomoses. Flow rates have remained suitable for dialysis in all patients, and the grafts are being used for dialysis three times per week. Dr. Lawson described the grafts as easy to cannulate via standard techniques.
He characterized these initial results as "quite remarkable" compared with the outcomes in two large studies of the current benchmark technologies, which are synthetic grafts made of PTFE (polytetrafluoroethyline). In those studies, the primary patency rate at 6 months was less than 50%, with a secondary patency rate of 77% and a 10% infection rate. In other studies, 30%-40% of PTFE grafts are abandoned within 12 months due to loss of patency.
The process of creating the bioengineered grafts begins with harvesting human aortic vascular smooth muscle cells, seeding them on a biodegradable matrix, then culturing them under pulsatile conditions. When the biodegradable matrix melts away, what remains is a tube comprised of vascular smooth muscle cells and extracellular matrix. This is then decellularized, yielding a tube of extracellular matrix that can be shipped off the shelf and around the world.
In primate models, the implanted bioengineered graft has been shown to repopulate with the host’s own vascular smooth muscle cells lined intimally by endothelium.
"Where we implanted an acellular structure, it appears to now be a living tissue, suggesting [the graft] has become their tissue, not ours," Dr. Lawson said.
To date, none of the bioengineered grafts implanted in patients has been explanted, so it’s unknown whether the favorable histologic changes seen in primates’ grafts also occur in humans. Larger clinical trials with longer follow-up are planned in order to assess the bioengineered graft’s durability.
Dr. Lawson’s study is funded by a Department of Defense research grant and by Humacyte. He serves as a consultant to the company.
AT THE AHA SCIENTIFIC SESSIONS
Major finding: The 6-month enduring patency rate of an investigational tissue-engineered vascular graft for hemodialysis access was 100%, markedly better than rates achievable with synthetic PTFE grafts, the current benchmark technology.
Data source: An initial report from an ongoing prospective first-in-man study in which, to date, 28 patients with end-stage renal disease have been implanted with a novel tissue-engineered vascular graft for use as a hemodialysis access.
Disclosures: The study was funded by the Department of Defense and Humacyte. The presenter is a consultant to the company.
No overall benefit seen with bisphosphonate treatment in chemoresistant breast cancer
SAN ANTONIO – Adjuvant zoledronate failed to improve outcomes in breast cancer patients with residual tumor following neoadjuvant chemotherapy in a large, randomized, phase-3 trial, Dr. Gunter von Minckwitz reported at the San Antonio Breast Cancer Symposium.
There was, however, a bright spot buried within the results of the Neo-Adjuvant Trial Add-On (NATAN) trial: the subset of participants over age 55 years showed a 17% improvement in disease-free survival compared with controls. Although this difference didn’t reach statistical significance because of limited patient numbers, it was closely similar to the benefit seen for adjuvant bisphosphonate therapy in the Early Breast Cancer Trialists Collaborative Group’s practice-changing meta-analysis presented earlier at the San Antonio symposium.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
That meta-analysis, presented by Dr. Robert Coleman of the University of Sheffield (England), included more than 17,000 participants in randomized trials. The conclusion was that adjuvant bisphosphonate therapy was of significant benefit in postmenopausal breast cancer patients, with a 17% reduction in the risk of mortality and a 34% decrease in the risk of bone metastases, compared with controls. Premenopausal patients didn’t benefit from adjuvant bisphosphonate therapy.
"We had discussed doing another bisphosphonate trial, this one to be limited to postmenopausal patients without a pathologic complete response after neoadjuvant chemotherapy, but having seen Robert Coleman’s presentation yesterday we are not very much in favor of that any longer. We have our practice guideline meeting for Germany in January, and I expect that we will give a recommendation to use bisphosphonates in postmenopausal patients, so it makes no sense to do another prospective trial," said Dr. von Minckwitz, chairman of the German Breast Group, Neu-Isenburg, and a gynecologist at the University of Frankfurt.
NATAN was carried out because patients with residual disease after neoadjuvant chemotherapy have a worse prognosis than those with a pathologic complete response, and they have few adjuvant treatment options. The study included 654 patients with residual disease after at least four cycles of neoadjuvant anthracycline/taxane-based chemotherapy. Patients were randomized to a planned 5 years of postsurgical intravenous zoledronate or observation, plus adjuvant endocrine therapy and/or trastuzumab as indicated.
The study was halted early due to futility after a median follow-up of 48 months because of virtually identical event-free survival rates in the two study arms. Women over age 55 years, who comprised one-third of the study population, were the only subgroup with a strong, albeit nonsignificant, trend toward benefit for zoledronate.
There is a clear need for new treatment options for women with residual tumor after neoadjuvant chemotherapy, particularly those who aren’t postmenopausal. Several novel agents are now in clinical trials in patients with chemoresistant breast cancer, including rucaparib, an oral small-molecule inhibitor of PARP (poly ADP-ribose polymerase), for triple-negative breast cancer; palbociclib, an oral and selective inhibitor of cyclin dependent kinases 4 and 6, in women with hormone receptor–positive/HER2-negative disease; and trastuzumab emtansine, a conjugate of trastuzumab and the cytotoxic agent mertansine, in patients with chemoresistant HER2-positive breast cancer, Dr. von Minckwitz said.
NATAN was funded by Novartis. Dr. von Minckwitz reported having received research grants from, and serving as a speaker and consultant for, Novartis and Roche.
SAN ANTONIO – Adjuvant zoledronate failed to improve outcomes in breast cancer patients with residual tumor following neoadjuvant chemotherapy in a large, randomized, phase-3 trial, Dr. Gunter von Minckwitz reported at the San Antonio Breast Cancer Symposium.
There was, however, a bright spot buried within the results of the Neo-Adjuvant Trial Add-On (NATAN) trial: the subset of participants over age 55 years showed a 17% improvement in disease-free survival compared with controls. Although this difference didn’t reach statistical significance because of limited patient numbers, it was closely similar to the benefit seen for adjuvant bisphosphonate therapy in the Early Breast Cancer Trialists Collaborative Group’s practice-changing meta-analysis presented earlier at the San Antonio symposium.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
That meta-analysis, presented by Dr. Robert Coleman of the University of Sheffield (England), included more than 17,000 participants in randomized trials. The conclusion was that adjuvant bisphosphonate therapy was of significant benefit in postmenopausal breast cancer patients, with a 17% reduction in the risk of mortality and a 34% decrease in the risk of bone metastases, compared with controls. Premenopausal patients didn’t benefit from adjuvant bisphosphonate therapy.
"We had discussed doing another bisphosphonate trial, this one to be limited to postmenopausal patients without a pathologic complete response after neoadjuvant chemotherapy, but having seen Robert Coleman’s presentation yesterday we are not very much in favor of that any longer. We have our practice guideline meeting for Germany in January, and I expect that we will give a recommendation to use bisphosphonates in postmenopausal patients, so it makes no sense to do another prospective trial," said Dr. von Minckwitz, chairman of the German Breast Group, Neu-Isenburg, and a gynecologist at the University of Frankfurt.
NATAN was carried out because patients with residual disease after neoadjuvant chemotherapy have a worse prognosis than those with a pathologic complete response, and they have few adjuvant treatment options. The study included 654 patients with residual disease after at least four cycles of neoadjuvant anthracycline/taxane-based chemotherapy. Patients were randomized to a planned 5 years of postsurgical intravenous zoledronate or observation, plus adjuvant endocrine therapy and/or trastuzumab as indicated.
The study was halted early due to futility after a median follow-up of 48 months because of virtually identical event-free survival rates in the two study arms. Women over age 55 years, who comprised one-third of the study population, were the only subgroup with a strong, albeit nonsignificant, trend toward benefit for zoledronate.
There is a clear need for new treatment options for women with residual tumor after neoadjuvant chemotherapy, particularly those who aren’t postmenopausal. Several novel agents are now in clinical trials in patients with chemoresistant breast cancer, including rucaparib, an oral small-molecule inhibitor of PARP (poly ADP-ribose polymerase), for triple-negative breast cancer; palbociclib, an oral and selective inhibitor of cyclin dependent kinases 4 and 6, in women with hormone receptor–positive/HER2-negative disease; and trastuzumab emtansine, a conjugate of trastuzumab and the cytotoxic agent mertansine, in patients with chemoresistant HER2-positive breast cancer, Dr. von Minckwitz said.
NATAN was funded by Novartis. Dr. von Minckwitz reported having received research grants from, and serving as a speaker and consultant for, Novartis and Roche.
SAN ANTONIO – Adjuvant zoledronate failed to improve outcomes in breast cancer patients with residual tumor following neoadjuvant chemotherapy in a large, randomized, phase-3 trial, Dr. Gunter von Minckwitz reported at the San Antonio Breast Cancer Symposium.
There was, however, a bright spot buried within the results of the Neo-Adjuvant Trial Add-On (NATAN) trial: the subset of participants over age 55 years showed a 17% improvement in disease-free survival compared with controls. Although this difference didn’t reach statistical significance because of limited patient numbers, it was closely similar to the benefit seen for adjuvant bisphosphonate therapy in the Early Breast Cancer Trialists Collaborative Group’s practice-changing meta-analysis presented earlier at the San Antonio symposium.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
That meta-analysis, presented by Dr. Robert Coleman of the University of Sheffield (England), included more than 17,000 participants in randomized trials. The conclusion was that adjuvant bisphosphonate therapy was of significant benefit in postmenopausal breast cancer patients, with a 17% reduction in the risk of mortality and a 34% decrease in the risk of bone metastases, compared with controls. Premenopausal patients didn’t benefit from adjuvant bisphosphonate therapy.
"We had discussed doing another bisphosphonate trial, this one to be limited to postmenopausal patients without a pathologic complete response after neoadjuvant chemotherapy, but having seen Robert Coleman’s presentation yesterday we are not very much in favor of that any longer. We have our practice guideline meeting for Germany in January, and I expect that we will give a recommendation to use bisphosphonates in postmenopausal patients, so it makes no sense to do another prospective trial," said Dr. von Minckwitz, chairman of the German Breast Group, Neu-Isenburg, and a gynecologist at the University of Frankfurt.
NATAN was carried out because patients with residual disease after neoadjuvant chemotherapy have a worse prognosis than those with a pathologic complete response, and they have few adjuvant treatment options. The study included 654 patients with residual disease after at least four cycles of neoadjuvant anthracycline/taxane-based chemotherapy. Patients were randomized to a planned 5 years of postsurgical intravenous zoledronate or observation, plus adjuvant endocrine therapy and/or trastuzumab as indicated.
The study was halted early due to futility after a median follow-up of 48 months because of virtually identical event-free survival rates in the two study arms. Women over age 55 years, who comprised one-third of the study population, were the only subgroup with a strong, albeit nonsignificant, trend toward benefit for zoledronate.
There is a clear need for new treatment options for women with residual tumor after neoadjuvant chemotherapy, particularly those who aren’t postmenopausal. Several novel agents are now in clinical trials in patients with chemoresistant breast cancer, including rucaparib, an oral small-molecule inhibitor of PARP (poly ADP-ribose polymerase), for triple-negative breast cancer; palbociclib, an oral and selective inhibitor of cyclin dependent kinases 4 and 6, in women with hormone receptor–positive/HER2-negative disease; and trastuzumab emtansine, a conjugate of trastuzumab and the cytotoxic agent mertansine, in patients with chemoresistant HER2-positive breast cancer, Dr. von Minckwitz said.
NATAN was funded by Novartis. Dr. von Minckwitz reported having received research grants from, and serving as a speaker and consultant for, Novartis and Roche.
AT SABCS 2013
Major finding: Women with residual tumor after neoadjuvant chemotherapy who were randomized to adjuvant zoledronate didn’t have a lower risk of recurrent disease or death during a median 4 years of follow-up than those who didn’t get the bisphosphonate, although there was a strong trend for benefit in the subgroup over age 55.
Data source: NATAN was a randomized, phase-3 clinical trial including 654 patients with post-neoadjuvant chemoresistant breast cancer.
Disclosures: The trial was sponsored by Novartis. Dr. von Minckwitz reported having received research grants from, and serving as a speaker and consultant for, Novartis and Roche.
Novel treatment promising for chronic neuropathic postmastectomy pain
SAN ANTONIO – Perineural injection of bupivacaine and dexamethasone was a simple and effective treatment for chronic neuropathic pain following mastectomy, based on results of a pilot study.
The effectiveness of this novel therapy strongly suggests the source of this common pain syndrome is damage to the T4 and T5 sensory nerves during surgery, rather than damage to the intercostobrachial nerve, as traditionally thought, according to Dr. Cathy J. Tang of the University of California, San Francisco.
The T4 and T5 sensory nerves come off the chest wall and enter the breast accompanied by a blood vessel. When these nerves are cut and cauterized during mastectomy, the resultant nerve damage can manifest as neuroma formation and neuropathic pain along the two dermatomes, she said at the San Antonio Breast Cancer Symposium.
Chronic postmastectomy breast pain is commonly referred to as postmastectomy pain syndrome. Published estimates of its incidence after mastectomy range from 20% to 68%. The pain can start in the immediate postoperative period, or onset can be delayed up to 6 months or more post mastectomy. The pain is typically experienced as a shooting or burning pain, with point tenderness. It persists well after the expected healing period.
The intervention involves identifying a patient’s points of maximum pain or tenderness, usually located laterally along the midaxillary line or at the inframammary fold directly below the nipple. These points are injected at the level of the chest wall. Each injection consists of 2 mL of an equal ratio of 0.5% bupivacaine plus 4 mg/mL of dexamethasone followed by a minute or two of massage to enhance infiltration of the area.
Dr. Tang reported on 19 patients who developed postmastectomy pain syndrome after either partial mastectomy, total mastectomy with immediate reconstruction, or lateral core biopsy in one case. A total of 29 points of maximum tenderness were identified and treated. All patients had pain relief within minutes, with point pain scores on a 0-10 scale falling from 8-9 to 0-1. Long-term pain relief was experienced after 17 of the 29 initial injections (59%) in 11 patients. Pain was resolved at another nine sites after a second injection. A third injection at one recalcitrant site led to long-term pain relief. Thus, perineural injections alleviated pain at 27 of 29 treated sites, or 93%, at a mean of 10.7 months of follow-up.
In light of how simple and safe this treatment is, Dr. Tang urged routine inquiry about postmastectomy neuropathic pain. Patients with postmastectomy pain often report an inability to lie on the affected side or to wear a bra.
The study also indicates the importance of careful dissection of the T4 and T5 sensory nerves during mastectomy in order to minimize the risk of postoperative neuroma formation.
Dr. Tang reported having no financial conflicts regarding this unfunded study.
SAN ANTONIO – Perineural injection of bupivacaine and dexamethasone was a simple and effective treatment for chronic neuropathic pain following mastectomy, based on results of a pilot study.
The effectiveness of this novel therapy strongly suggests the source of this common pain syndrome is damage to the T4 and T5 sensory nerves during surgery, rather than damage to the intercostobrachial nerve, as traditionally thought, according to Dr. Cathy J. Tang of the University of California, San Francisco.
The T4 and T5 sensory nerves come off the chest wall and enter the breast accompanied by a blood vessel. When these nerves are cut and cauterized during mastectomy, the resultant nerve damage can manifest as neuroma formation and neuropathic pain along the two dermatomes, she said at the San Antonio Breast Cancer Symposium.
Chronic postmastectomy breast pain is commonly referred to as postmastectomy pain syndrome. Published estimates of its incidence after mastectomy range from 20% to 68%. The pain can start in the immediate postoperative period, or onset can be delayed up to 6 months or more post mastectomy. The pain is typically experienced as a shooting or burning pain, with point tenderness. It persists well after the expected healing period.
The intervention involves identifying a patient’s points of maximum pain or tenderness, usually located laterally along the midaxillary line or at the inframammary fold directly below the nipple. These points are injected at the level of the chest wall. Each injection consists of 2 mL of an equal ratio of 0.5% bupivacaine plus 4 mg/mL of dexamethasone followed by a minute or two of massage to enhance infiltration of the area.
Dr. Tang reported on 19 patients who developed postmastectomy pain syndrome after either partial mastectomy, total mastectomy with immediate reconstruction, or lateral core biopsy in one case. A total of 29 points of maximum tenderness were identified and treated. All patients had pain relief within minutes, with point pain scores on a 0-10 scale falling from 8-9 to 0-1. Long-term pain relief was experienced after 17 of the 29 initial injections (59%) in 11 patients. Pain was resolved at another nine sites after a second injection. A third injection at one recalcitrant site led to long-term pain relief. Thus, perineural injections alleviated pain at 27 of 29 treated sites, or 93%, at a mean of 10.7 months of follow-up.
In light of how simple and safe this treatment is, Dr. Tang urged routine inquiry about postmastectomy neuropathic pain. Patients with postmastectomy pain often report an inability to lie on the affected side or to wear a bra.
The study also indicates the importance of careful dissection of the T4 and T5 sensory nerves during mastectomy in order to minimize the risk of postoperative neuroma formation.
Dr. Tang reported having no financial conflicts regarding this unfunded study.
SAN ANTONIO – Perineural injection of bupivacaine and dexamethasone was a simple and effective treatment for chronic neuropathic pain following mastectomy, based on results of a pilot study.
The effectiveness of this novel therapy strongly suggests the source of this common pain syndrome is damage to the T4 and T5 sensory nerves during surgery, rather than damage to the intercostobrachial nerve, as traditionally thought, according to Dr. Cathy J. Tang of the University of California, San Francisco.
The T4 and T5 sensory nerves come off the chest wall and enter the breast accompanied by a blood vessel. When these nerves are cut and cauterized during mastectomy, the resultant nerve damage can manifest as neuroma formation and neuropathic pain along the two dermatomes, she said at the San Antonio Breast Cancer Symposium.
Chronic postmastectomy breast pain is commonly referred to as postmastectomy pain syndrome. Published estimates of its incidence after mastectomy range from 20% to 68%. The pain can start in the immediate postoperative period, or onset can be delayed up to 6 months or more post mastectomy. The pain is typically experienced as a shooting or burning pain, with point tenderness. It persists well after the expected healing period.
The intervention involves identifying a patient’s points of maximum pain or tenderness, usually located laterally along the midaxillary line or at the inframammary fold directly below the nipple. These points are injected at the level of the chest wall. Each injection consists of 2 mL of an equal ratio of 0.5% bupivacaine plus 4 mg/mL of dexamethasone followed by a minute or two of massage to enhance infiltration of the area.
Dr. Tang reported on 19 patients who developed postmastectomy pain syndrome after either partial mastectomy, total mastectomy with immediate reconstruction, or lateral core biopsy in one case. A total of 29 points of maximum tenderness were identified and treated. All patients had pain relief within minutes, with point pain scores on a 0-10 scale falling from 8-9 to 0-1. Long-term pain relief was experienced after 17 of the 29 initial injections (59%) in 11 patients. Pain was resolved at another nine sites after a second injection. A third injection at one recalcitrant site led to long-term pain relief. Thus, perineural injections alleviated pain at 27 of 29 treated sites, or 93%, at a mean of 10.7 months of follow-up.
In light of how simple and safe this treatment is, Dr. Tang urged routine inquiry about postmastectomy neuropathic pain. Patients with postmastectomy pain often report an inability to lie on the affected side or to wear a bra.
The study also indicates the importance of careful dissection of the T4 and T5 sensory nerves during mastectomy in order to minimize the risk of postoperative neuroma formation.
Dr. Tang reported having no financial conflicts regarding this unfunded study.
AT SABCS 2013
Major finding: Injection of a combination of bupivacaine and dexamethasone at well-defined sites of maximum pain and tenderness resolved pain at 93% of treated sites in patients with chronic neuropathic postmastectomy pain.
Data source: A prospective case series involving 19 patients with postmastectomy pain syndrome. A total of 29 sites of maximum pain and tenderness were treated.
Disclosures: The study was conducted free of commercial support. The presenter reported having no relevant financial conflicts.
Exercise protects black women against ER-negative breast cancer
SAN ANTONIO – African-American women had half the risk of developing estrogen receptor–negative breast cancer if they exercised vigorously 3 hours per week, according to data from the Black Women’s Health Study.
The findings are based on nearly 20 years of follow-up on 44,704 women in the prospective, observational Black Women's Health Study. During this period, 1,377 subjects were diagnosed with invasive breast cancer, including 327 with ER-negative disease.
Black women who reported having engaged in vigorous exercise from high school onwards for a lifetime average of 3 hours or more per week had a 47% reduction in the risk of ER-negative breast cancer, compared with women exercising vigorously for less than 1 hour per week after adjusting for age, education, parity, and dietary pattern, Lucile Adams-Campbell, Ph.D., said at the San Antonio Breast Cancer Symposium.
Exercise proved unrelated to the risk of developing the generally more indolent ER-positive breast cancers in this population, said Dr. Adams-Campbell, professor of oncology at the Georgetown University Lombardi Comprehensive Cancer Center in Washington, D.C.
The inverse relationship between vigorous exercise and ER-negative breast cancer risk was statistically significant only among postmenopausal black women. In that population, the adjusted risk was reduced by 59% in participants with a lifetime average of at least 3 hours of exercise per week, compared with less than 1 hour.
The Centers for Disease Control and Prevention defines vigorous exercise as more than 6.0 metabolic equivalents (more than 7 kcal/min). Examples of activities through which this is readily accomplished include jogging, bicycling at more than 10 mph, or briskly walking uphill.
Dr. Adams-Campbell is a coleader of the ongoing Black Women’s Health Study, which aims to shed light on the elevated risks of hypertension, stroke, diabetes, lupus, and other diseases present in African-American women. The study is funded by the National Cancer Institute. Dr. Adams-Campbell reported having no relevant financial interests.
SAN ANTONIO – African-American women had half the risk of developing estrogen receptor–negative breast cancer if they exercised vigorously 3 hours per week, according to data from the Black Women’s Health Study.
The findings are based on nearly 20 years of follow-up on 44,704 women in the prospective, observational Black Women's Health Study. During this period, 1,377 subjects were diagnosed with invasive breast cancer, including 327 with ER-negative disease.
Black women who reported having engaged in vigorous exercise from high school onwards for a lifetime average of 3 hours or more per week had a 47% reduction in the risk of ER-negative breast cancer, compared with women exercising vigorously for less than 1 hour per week after adjusting for age, education, parity, and dietary pattern, Lucile Adams-Campbell, Ph.D., said at the San Antonio Breast Cancer Symposium.
Exercise proved unrelated to the risk of developing the generally more indolent ER-positive breast cancers in this population, said Dr. Adams-Campbell, professor of oncology at the Georgetown University Lombardi Comprehensive Cancer Center in Washington, D.C.
The inverse relationship between vigorous exercise and ER-negative breast cancer risk was statistically significant only among postmenopausal black women. In that population, the adjusted risk was reduced by 59% in participants with a lifetime average of at least 3 hours of exercise per week, compared with less than 1 hour.
The Centers for Disease Control and Prevention defines vigorous exercise as more than 6.0 metabolic equivalents (more than 7 kcal/min). Examples of activities through which this is readily accomplished include jogging, bicycling at more than 10 mph, or briskly walking uphill.
Dr. Adams-Campbell is a coleader of the ongoing Black Women’s Health Study, which aims to shed light on the elevated risks of hypertension, stroke, diabetes, lupus, and other diseases present in African-American women. The study is funded by the National Cancer Institute. Dr. Adams-Campbell reported having no relevant financial interests.
SAN ANTONIO – African-American women had half the risk of developing estrogen receptor–negative breast cancer if they exercised vigorously 3 hours per week, according to data from the Black Women’s Health Study.
The findings are based on nearly 20 years of follow-up on 44,704 women in the prospective, observational Black Women's Health Study. During this period, 1,377 subjects were diagnosed with invasive breast cancer, including 327 with ER-negative disease.
Black women who reported having engaged in vigorous exercise from high school onwards for a lifetime average of 3 hours or more per week had a 47% reduction in the risk of ER-negative breast cancer, compared with women exercising vigorously for less than 1 hour per week after adjusting for age, education, parity, and dietary pattern, Lucile Adams-Campbell, Ph.D., said at the San Antonio Breast Cancer Symposium.
Exercise proved unrelated to the risk of developing the generally more indolent ER-positive breast cancers in this population, said Dr. Adams-Campbell, professor of oncology at the Georgetown University Lombardi Comprehensive Cancer Center in Washington, D.C.
The inverse relationship between vigorous exercise and ER-negative breast cancer risk was statistically significant only among postmenopausal black women. In that population, the adjusted risk was reduced by 59% in participants with a lifetime average of at least 3 hours of exercise per week, compared with less than 1 hour.
The Centers for Disease Control and Prevention defines vigorous exercise as more than 6.0 metabolic equivalents (more than 7 kcal/min). Examples of activities through which this is readily accomplished include jogging, bicycling at more than 10 mph, or briskly walking uphill.
Dr. Adams-Campbell is a coleader of the ongoing Black Women’s Health Study, which aims to shed light on the elevated risks of hypertension, stroke, diabetes, lupus, and other diseases present in African-American women. The study is funded by the National Cancer Institute. Dr. Adams-Campbell reported having no relevant financial interests.
AT SABCS 2013
Major finding: Black women who reported engaging in an average of least 3 hours of vigorous exercise per week over their lifetime had a 47% reduction in the risk of developing estrogen receptor–negative breast cancer, compared with those averaging less than 1 hour per week of vigorous exercise.
Data source: This analysis from the ongoing Black Women’s Health Study included nearly 45,000 women aged 30 years or older at enrollment, with close to 20 years of prospective follow-up.
Disclosures: The study is funded by the National Cancer Institute. The presenter reported having no financial conflicts.
Findings set stage for immunomodulatory approaches in breast cancer
SAN ANTONIO – Tumor-infiltrating lymphocyte levels at breast cancer diagnosis predict response to therapies for both HER2-positive and triple-negative breast cancers, according to studies presented at the San Antonio Breast Cancer Symposium.
Tumor-infiltrating lymphocytes (TILs) appeared to be a useful biomarker of response to trastuzumab (Herceptin) as well as to chemotherapy. By extension, breast cancers appear to be immunogenic, contrary to traditional thinking. Trastuzumab and chemotherapy appear to act not only on the tumor, but to enhance antitumor immunity as well.
The findings could result in a paradigm shift in treatment approaches. Clinical outcomes could potentially be improved in women with HER2-positive or triple-negative breast cancers by targeting the immune system with immunomodulatory agents in addition to providing standard therapies, according to Dr. Sherene Loi, a medical oncologist and head of the translational breast cancer genomics laboratory at the Peter MacCallum Cancer Center in Melbourne.
Dr. Loi presented an analysis of breast cancer samples from 156 women with operable or locally advanced HER2-positive breast cancer who participated in the previously reported GeparQuattro trial. All of the women received neoadjuvant trastuzumab and cytotoxic chemotherapy. In a multivariate analysis, for every 10% increase in the level of stromal TILs there was a 16% increase in the pathologic complete response rate, meaning no residual invasive cancer in the breast or lymph nodes at surgery.
"These data reinforced for us that there is a relationship between the immune system and responses to trastuzumab and chemotherapy," she said.
In another study presented at the meeting, Dr. Sylvia Adams demonstrated the prognostic value of TILs in patients with triple-negative breast cancer. Tumors were analyzed from 481 participants in Eastern Cooperative Oncology Group 2197 and 1199, two previously reported phase III randomized, prospective trials of adjuvant chemotherapy in triple-negative breast cancer.
At least 10% lymphocytic infiltration in the tumor stroma was seen in 80% of tumors, meaning that 10% or more of all cells in the stroma were TILs. For each 10% increment in stromal TILs at the time of breast cancer diagnosis, there was an 18% reduction in the risk of distant recurrence and a "very impressive" 19% reduction in the risk of mortality at a median followup of 10.6 years, reported Dr. Adams of New York University.
She noted that this new analysis confirms an earlier study led by Dr. Loi, the prospective, randomized BIG 02-98 trial. In that study, TILs were associated with improved prognosis in patients with operable triple-negative breast cancer. The new validation study, Dr. Adams noted, raises to Level I the evidence that supports measuring stromal TILs as a biomarker in triple-negative breast cancer.
"The data provide strong evidence for the incorporation of this feature into the AJCC (American Joint Committee on Cancer) staging for triple-negative breast cancer and for utilization of stromal TILs as a stratifier in clinical trials," Dr. Adams said. "And most important, I would like to say – because some may ask ‘Is this just another prognostic factor for breast cancer?’ – is that these data really help provide additional evidence that some breast cancers can be immunogenic and that the endogenous immune response to cancer is predictive of survival in triple-negative breast cancer. So this actually opens up the area for introducing immunotherapies in triple-negative breast cancer. There is a hope that by targeting and harnessing the immune system we can improve cure rates."
Dr. Loi provided a glimpse into the possible way forward. In an effort to learn how trastuzumab affects antitumor immunity, she and her colleagues performed gene expression analyses on HER2-positive breast tumor specimens from 202 participants in the phase III randomized FinHER clinical trial of adjuvant trastuzumab or no trastuzumab along with postoperative chemotherapy. The greatest clinical response to trastuzumab was seen in patients whose tumors highly expressed the immunosuppressive gene known as programmed cell death 1 (PD-1).
This led to studies conducted in a mouse model of HER2-positive breast cancer. Dr. Loi and her colleagues showed that combining trastuzumab with a T-cell checkpoint inhibitor – an anti-PD-1 agent resulted in greater reductions in tumor size than with trastuzumab alone.
More than half a dozen T-cell checkpoint inhibitors are in the developmental pipeline for treatment of a range of malignancies, including the anti-PD-1 agents nivolumab, lambrolizumab, and pidilizumab. Clinical trials testing the combination of trastuzumab and a PD-1 inhibitor in HER2-positive breast cancer patients are being planned.
Dr. Loi’s work was funded by the European Union RESPONSIFY project, the Breast Cancer Research Foundation, and the Fonds J.C. Heuson. She and Dr. Adams reported having no relevant financial conflicts of interest.
SAN ANTONIO – Tumor-infiltrating lymphocyte levels at breast cancer diagnosis predict response to therapies for both HER2-positive and triple-negative breast cancers, according to studies presented at the San Antonio Breast Cancer Symposium.
Tumor-infiltrating lymphocytes (TILs) appeared to be a useful biomarker of response to trastuzumab (Herceptin) as well as to chemotherapy. By extension, breast cancers appear to be immunogenic, contrary to traditional thinking. Trastuzumab and chemotherapy appear to act not only on the tumor, but to enhance antitumor immunity as well.
The findings could result in a paradigm shift in treatment approaches. Clinical outcomes could potentially be improved in women with HER2-positive or triple-negative breast cancers by targeting the immune system with immunomodulatory agents in addition to providing standard therapies, according to Dr. Sherene Loi, a medical oncologist and head of the translational breast cancer genomics laboratory at the Peter MacCallum Cancer Center in Melbourne.
Dr. Loi presented an analysis of breast cancer samples from 156 women with operable or locally advanced HER2-positive breast cancer who participated in the previously reported GeparQuattro trial. All of the women received neoadjuvant trastuzumab and cytotoxic chemotherapy. In a multivariate analysis, for every 10% increase in the level of stromal TILs there was a 16% increase in the pathologic complete response rate, meaning no residual invasive cancer in the breast or lymph nodes at surgery.
"These data reinforced for us that there is a relationship between the immune system and responses to trastuzumab and chemotherapy," she said.
In another study presented at the meeting, Dr. Sylvia Adams demonstrated the prognostic value of TILs in patients with triple-negative breast cancer. Tumors were analyzed from 481 participants in Eastern Cooperative Oncology Group 2197 and 1199, two previously reported phase III randomized, prospective trials of adjuvant chemotherapy in triple-negative breast cancer.
At least 10% lymphocytic infiltration in the tumor stroma was seen in 80% of tumors, meaning that 10% or more of all cells in the stroma were TILs. For each 10% increment in stromal TILs at the time of breast cancer diagnosis, there was an 18% reduction in the risk of distant recurrence and a "very impressive" 19% reduction in the risk of mortality at a median followup of 10.6 years, reported Dr. Adams of New York University.
She noted that this new analysis confirms an earlier study led by Dr. Loi, the prospective, randomized BIG 02-98 trial. In that study, TILs were associated with improved prognosis in patients with operable triple-negative breast cancer. The new validation study, Dr. Adams noted, raises to Level I the evidence that supports measuring stromal TILs as a biomarker in triple-negative breast cancer.
"The data provide strong evidence for the incorporation of this feature into the AJCC (American Joint Committee on Cancer) staging for triple-negative breast cancer and for utilization of stromal TILs as a stratifier in clinical trials," Dr. Adams said. "And most important, I would like to say – because some may ask ‘Is this just another prognostic factor for breast cancer?’ – is that these data really help provide additional evidence that some breast cancers can be immunogenic and that the endogenous immune response to cancer is predictive of survival in triple-negative breast cancer. So this actually opens up the area for introducing immunotherapies in triple-negative breast cancer. There is a hope that by targeting and harnessing the immune system we can improve cure rates."
Dr. Loi provided a glimpse into the possible way forward. In an effort to learn how trastuzumab affects antitumor immunity, she and her colleagues performed gene expression analyses on HER2-positive breast tumor specimens from 202 participants in the phase III randomized FinHER clinical trial of adjuvant trastuzumab or no trastuzumab along with postoperative chemotherapy. The greatest clinical response to trastuzumab was seen in patients whose tumors highly expressed the immunosuppressive gene known as programmed cell death 1 (PD-1).
This led to studies conducted in a mouse model of HER2-positive breast cancer. Dr. Loi and her colleagues showed that combining trastuzumab with a T-cell checkpoint inhibitor – an anti-PD-1 agent resulted in greater reductions in tumor size than with trastuzumab alone.
More than half a dozen T-cell checkpoint inhibitors are in the developmental pipeline for treatment of a range of malignancies, including the anti-PD-1 agents nivolumab, lambrolizumab, and pidilizumab. Clinical trials testing the combination of trastuzumab and a PD-1 inhibitor in HER2-positive breast cancer patients are being planned.
Dr. Loi’s work was funded by the European Union RESPONSIFY project, the Breast Cancer Research Foundation, and the Fonds J.C. Heuson. She and Dr. Adams reported having no relevant financial conflicts of interest.
SAN ANTONIO – Tumor-infiltrating lymphocyte levels at breast cancer diagnosis predict response to therapies for both HER2-positive and triple-negative breast cancers, according to studies presented at the San Antonio Breast Cancer Symposium.
Tumor-infiltrating lymphocytes (TILs) appeared to be a useful biomarker of response to trastuzumab (Herceptin) as well as to chemotherapy. By extension, breast cancers appear to be immunogenic, contrary to traditional thinking. Trastuzumab and chemotherapy appear to act not only on the tumor, but to enhance antitumor immunity as well.
The findings could result in a paradigm shift in treatment approaches. Clinical outcomes could potentially be improved in women with HER2-positive or triple-negative breast cancers by targeting the immune system with immunomodulatory agents in addition to providing standard therapies, according to Dr. Sherene Loi, a medical oncologist and head of the translational breast cancer genomics laboratory at the Peter MacCallum Cancer Center in Melbourne.
Dr. Loi presented an analysis of breast cancer samples from 156 women with operable or locally advanced HER2-positive breast cancer who participated in the previously reported GeparQuattro trial. All of the women received neoadjuvant trastuzumab and cytotoxic chemotherapy. In a multivariate analysis, for every 10% increase in the level of stromal TILs there was a 16% increase in the pathologic complete response rate, meaning no residual invasive cancer in the breast or lymph nodes at surgery.
"These data reinforced for us that there is a relationship between the immune system and responses to trastuzumab and chemotherapy," she said.
In another study presented at the meeting, Dr. Sylvia Adams demonstrated the prognostic value of TILs in patients with triple-negative breast cancer. Tumors were analyzed from 481 participants in Eastern Cooperative Oncology Group 2197 and 1199, two previously reported phase III randomized, prospective trials of adjuvant chemotherapy in triple-negative breast cancer.
At least 10% lymphocytic infiltration in the tumor stroma was seen in 80% of tumors, meaning that 10% or more of all cells in the stroma were TILs. For each 10% increment in stromal TILs at the time of breast cancer diagnosis, there was an 18% reduction in the risk of distant recurrence and a "very impressive" 19% reduction in the risk of mortality at a median followup of 10.6 years, reported Dr. Adams of New York University.
She noted that this new analysis confirms an earlier study led by Dr. Loi, the prospective, randomized BIG 02-98 trial. In that study, TILs were associated with improved prognosis in patients with operable triple-negative breast cancer. The new validation study, Dr. Adams noted, raises to Level I the evidence that supports measuring stromal TILs as a biomarker in triple-negative breast cancer.
"The data provide strong evidence for the incorporation of this feature into the AJCC (American Joint Committee on Cancer) staging for triple-negative breast cancer and for utilization of stromal TILs as a stratifier in clinical trials," Dr. Adams said. "And most important, I would like to say – because some may ask ‘Is this just another prognostic factor for breast cancer?’ – is that these data really help provide additional evidence that some breast cancers can be immunogenic and that the endogenous immune response to cancer is predictive of survival in triple-negative breast cancer. So this actually opens up the area for introducing immunotherapies in triple-negative breast cancer. There is a hope that by targeting and harnessing the immune system we can improve cure rates."
Dr. Loi provided a glimpse into the possible way forward. In an effort to learn how trastuzumab affects antitumor immunity, she and her colleagues performed gene expression analyses on HER2-positive breast tumor specimens from 202 participants in the phase III randomized FinHER clinical trial of adjuvant trastuzumab or no trastuzumab along with postoperative chemotherapy. The greatest clinical response to trastuzumab was seen in patients whose tumors highly expressed the immunosuppressive gene known as programmed cell death 1 (PD-1).
This led to studies conducted in a mouse model of HER2-positive breast cancer. Dr. Loi and her colleagues showed that combining trastuzumab with a T-cell checkpoint inhibitor – an anti-PD-1 agent resulted in greater reductions in tumor size than with trastuzumab alone.
More than half a dozen T-cell checkpoint inhibitors are in the developmental pipeline for treatment of a range of malignancies, including the anti-PD-1 agents nivolumab, lambrolizumab, and pidilizumab. Clinical trials testing the combination of trastuzumab and a PD-1 inhibitor in HER2-positive breast cancer patients are being planned.
Dr. Loi’s work was funded by the European Union RESPONSIFY project, the Breast Cancer Research Foundation, and the Fonds J.C. Heuson. She and Dr. Adams reported having no relevant financial conflicts of interest.
AT SABCS 2013
Major finding: For every 10% increment in stromal tumor-infiltrating lymphocytes present at the time of diagnosis of HER2-positive breast cancer, the likelihood of a pathologic complete response to neoadjuvant trastuzumab plus chemotherapy climbed by 16%. In a separate study, every 10% increment in stromal tumor-infiltrating lymphocytes present in triple-negative breast cancer specimens was associated with 19% reduction in the risk of mortality at almost 11 years of followup.
Data source: These translational studies entailed measurement of tumor-infiltrating lymphocyte levels in HER2-positive tumors from 156 participants in the randomized, prospective GeparQuattro trial and 481 triple-negative cancer specimens from participants in the phase III ECOG 2197 and 1199 randomized adjuvant chemotherapy trials.
Disclosures: The presenters reported having no financial conflicts of interest.
Breast MRI screening finds undetected cancers in 11 per 1,000 average-risk women
SAN ANTONIO – MRI screening of women who were at average risk of breast cancer and had a negative screening mammogram resulted in the diagnosis of 11 cases of cancer per 1,000 women screened, Dr. Simone Schrading said at the San Antonio Breast Cancer Symposium.
"The additional cancer detection rate is high, even in heavily prescreened women. In experienced hands, the positive predictive value of MRI screening in this average-risk cohort was comparable to that seen in mammographic screening programs or in MRI high-risk screening cohorts," said Dr. Schrading, of the University of Aachen, Germany.
While MRI is well established as a screening method for women at high familial risk of breast cancer, there have been no data to support its use in average-risk women.
Dr. Schrading reported on a prospective, single-center study evaluating the additional cancer yield and accuracy of breast MRI screening in 1,725 women at average risk for breast cancer. All had normal clinical breast examinations and digital screening mammograms. In addition, 89% of the women had undergone high-frequency breast ultrasound screening, which was normal in all cases. None of the subjects had a personal or family history of breast or ovarian cancer, and none had been diagnosed with breast proliferative changes or atypia. Their mean age was 55 years (range, 42-71 years).
MRI screening detected breast cancers in 18 of these patients, for a detection rate of 11 per 1,000 screened. Seven malignancies were ductal carcinoma in situ, and the other 11 were invasive breast cancer. The mean size of the invasive cancers was 11 mm. Five of the 7 ductal carcinomas in situ and 6 of 11 invasive cancers were high-grade cancers. On the other hand, the stage distribution of the invasive cancers was favorable: All were staged as pN0, M0.
Nearly 91% of screening MRIs were negative as defined by a BIRADS 1 or 2 rating. Another 5.9% were rated BIRADS 3; follow-up MRIs in this group of patients showed no breast cancers. Suspicious lesions – that is, BIRADS 4 or 5 – were noted in 54 patients, or 3.2% of the study group, and they were evaluated by MRI-guided biopsy. The lesions proved benign in 28 of 54 cases, high risk in 8, and malignant in 18.
Thus, the positive predictive value of breast MRI screening in this average-risk, extensively prescreened population was 33% if only BIRADS 4 and 5 cancers were defined as true positives, and 48% if BIRADS 4 and 5 high-risk lesions were also counted.
The age distribution of the patients with MRI-detected breast cancers was similar to that of the total study population. Mammographic breast density did not predict the likelihood of identifying breast cancer by MRI.
Dr. Laura J. Esserman rose from the audience to question the cost-benefit ratio of introducing MRI screening for the vast population of average-risk women.
The aggregate cost of breast cancer screening in the United States is already at least $8 billion annually. Utilizing MRI to screen average-risk women would push that figure up by an order of magnitude, observed Dr. Esserman, professor of surgery and of radiology and director of the Carol Franc Buck Breast Care Center at the University of California, San Francisco.
"MRI screening is still very expensive, it’s true," Dr. Schrading replied. "The main reason for the high cost of MRI is the long acquisition time and the long reading time. The long acquisition time is because the MRI protocols we have today are designed for diagnostic purposes, not for the purpose of screening. Our goal is to use a screening protocol for MRI, where the acquisition time is only 3 minutes and the reading time is less than 30 seconds. This might be a way to reduce the cost of MRI," she said.
The study was conducted free of commercial support. Dr. Schrading declared having no financial conflicts of interest.
SAN ANTONIO – MRI screening of women who were at average risk of breast cancer and had a negative screening mammogram resulted in the diagnosis of 11 cases of cancer per 1,000 women screened, Dr. Simone Schrading said at the San Antonio Breast Cancer Symposium.
"The additional cancer detection rate is high, even in heavily prescreened women. In experienced hands, the positive predictive value of MRI screening in this average-risk cohort was comparable to that seen in mammographic screening programs or in MRI high-risk screening cohorts," said Dr. Schrading, of the University of Aachen, Germany.
While MRI is well established as a screening method for women at high familial risk of breast cancer, there have been no data to support its use in average-risk women.
Dr. Schrading reported on a prospective, single-center study evaluating the additional cancer yield and accuracy of breast MRI screening in 1,725 women at average risk for breast cancer. All had normal clinical breast examinations and digital screening mammograms. In addition, 89% of the women had undergone high-frequency breast ultrasound screening, which was normal in all cases. None of the subjects had a personal or family history of breast or ovarian cancer, and none had been diagnosed with breast proliferative changes or atypia. Their mean age was 55 years (range, 42-71 years).
MRI screening detected breast cancers in 18 of these patients, for a detection rate of 11 per 1,000 screened. Seven malignancies were ductal carcinoma in situ, and the other 11 were invasive breast cancer. The mean size of the invasive cancers was 11 mm. Five of the 7 ductal carcinomas in situ and 6 of 11 invasive cancers were high-grade cancers. On the other hand, the stage distribution of the invasive cancers was favorable: All were staged as pN0, M0.
Nearly 91% of screening MRIs were negative as defined by a BIRADS 1 or 2 rating. Another 5.9% were rated BIRADS 3; follow-up MRIs in this group of patients showed no breast cancers. Suspicious lesions – that is, BIRADS 4 or 5 – were noted in 54 patients, or 3.2% of the study group, and they were evaluated by MRI-guided biopsy. The lesions proved benign in 28 of 54 cases, high risk in 8, and malignant in 18.
Thus, the positive predictive value of breast MRI screening in this average-risk, extensively prescreened population was 33% if only BIRADS 4 and 5 cancers were defined as true positives, and 48% if BIRADS 4 and 5 high-risk lesions were also counted.
The age distribution of the patients with MRI-detected breast cancers was similar to that of the total study population. Mammographic breast density did not predict the likelihood of identifying breast cancer by MRI.
Dr. Laura J. Esserman rose from the audience to question the cost-benefit ratio of introducing MRI screening for the vast population of average-risk women.
The aggregate cost of breast cancer screening in the United States is already at least $8 billion annually. Utilizing MRI to screen average-risk women would push that figure up by an order of magnitude, observed Dr. Esserman, professor of surgery and of radiology and director of the Carol Franc Buck Breast Care Center at the University of California, San Francisco.
"MRI screening is still very expensive, it’s true," Dr. Schrading replied. "The main reason for the high cost of MRI is the long acquisition time and the long reading time. The long acquisition time is because the MRI protocols we have today are designed for diagnostic purposes, not for the purpose of screening. Our goal is to use a screening protocol for MRI, where the acquisition time is only 3 minutes and the reading time is less than 30 seconds. This might be a way to reduce the cost of MRI," she said.
The study was conducted free of commercial support. Dr. Schrading declared having no financial conflicts of interest.
SAN ANTONIO – MRI screening of women who were at average risk of breast cancer and had a negative screening mammogram resulted in the diagnosis of 11 cases of cancer per 1,000 women screened, Dr. Simone Schrading said at the San Antonio Breast Cancer Symposium.
"The additional cancer detection rate is high, even in heavily prescreened women. In experienced hands, the positive predictive value of MRI screening in this average-risk cohort was comparable to that seen in mammographic screening programs or in MRI high-risk screening cohorts," said Dr. Schrading, of the University of Aachen, Germany.
While MRI is well established as a screening method for women at high familial risk of breast cancer, there have been no data to support its use in average-risk women.
Dr. Schrading reported on a prospective, single-center study evaluating the additional cancer yield and accuracy of breast MRI screening in 1,725 women at average risk for breast cancer. All had normal clinical breast examinations and digital screening mammograms. In addition, 89% of the women had undergone high-frequency breast ultrasound screening, which was normal in all cases. None of the subjects had a personal or family history of breast or ovarian cancer, and none had been diagnosed with breast proliferative changes or atypia. Their mean age was 55 years (range, 42-71 years).
MRI screening detected breast cancers in 18 of these patients, for a detection rate of 11 per 1,000 screened. Seven malignancies were ductal carcinoma in situ, and the other 11 were invasive breast cancer. The mean size of the invasive cancers was 11 mm. Five of the 7 ductal carcinomas in situ and 6 of 11 invasive cancers were high-grade cancers. On the other hand, the stage distribution of the invasive cancers was favorable: All were staged as pN0, M0.
Nearly 91% of screening MRIs were negative as defined by a BIRADS 1 or 2 rating. Another 5.9% were rated BIRADS 3; follow-up MRIs in this group of patients showed no breast cancers. Suspicious lesions – that is, BIRADS 4 or 5 – were noted in 54 patients, or 3.2% of the study group, and they were evaluated by MRI-guided biopsy. The lesions proved benign in 28 of 54 cases, high risk in 8, and malignant in 18.
Thus, the positive predictive value of breast MRI screening in this average-risk, extensively prescreened population was 33% if only BIRADS 4 and 5 cancers were defined as true positives, and 48% if BIRADS 4 and 5 high-risk lesions were also counted.
The age distribution of the patients with MRI-detected breast cancers was similar to that of the total study population. Mammographic breast density did not predict the likelihood of identifying breast cancer by MRI.
Dr. Laura J. Esserman rose from the audience to question the cost-benefit ratio of introducing MRI screening for the vast population of average-risk women.
The aggregate cost of breast cancer screening in the United States is already at least $8 billion annually. Utilizing MRI to screen average-risk women would push that figure up by an order of magnitude, observed Dr. Esserman, professor of surgery and of radiology and director of the Carol Franc Buck Breast Care Center at the University of California, San Francisco.
"MRI screening is still very expensive, it’s true," Dr. Schrading replied. "The main reason for the high cost of MRI is the long acquisition time and the long reading time. The long acquisition time is because the MRI protocols we have today are designed for diagnostic purposes, not for the purpose of screening. Our goal is to use a screening protocol for MRI, where the acquisition time is only 3 minutes and the reading time is less than 30 seconds. This might be a way to reduce the cost of MRI," she said.
The study was conducted free of commercial support. Dr. Schrading declared having no financial conflicts of interest.
AT SABCS 2013
Major finding: Breast MRI screening of women who were at average risk for breast cancer and already had a negative mammogram had a breast cancer detection rate of 11 cases per 1,000 women screened.
Data source: This was a prospective, uncontrolled, single-center study of breast MRI screening in 1,725 women with no personal or family history of breast or ovarian cancer, no diagnosis of proliferative changes, and a mean age of 55 years.
Disclosures: The study was conducted free of commercial support. Dr. Schrading declared having no financial conflicts of interest.
New Mega-review Underscores Mammography’s Benefits
SAN ANTONIO – The much-publicized wide disparities in the estimated value of mammographic screening for breast cancer reported in recent major reviews are overblown and largely an artifact of methodologic differences, according to a new examination of the evidence.
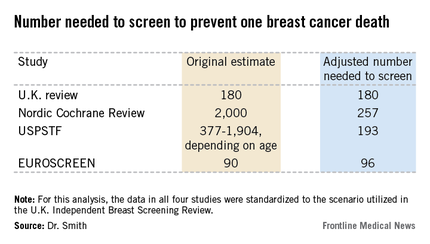
The four recent major reviews of the data regarding the absolute benefits of mammography came up with estimates ranging from 90-2,000 of the number of women who need to be screened (NNS) in order to prevent one death from breast cancer. That greater than 20-fold difference in estimated magnitude of benefit has done little to inspire public and physician confidence that mammography is a key tool in reducing cancer deaths.
But the two analyses with the least supportive outcomes – the Nordic Cochrane and U.S. Preventive Services Task Force (USPSTF-) analyses – used follow-up periods of 10 and 15 years, respectively. That follow-up is too short a time to assess the full value of mammographic screening, Robert A. Smith, Ph.D., asserted at the San Antonio Breast Cancer Symposium.
To illustrate: In a European mammographic screening study with a 30-year follow-up, the NNS after 10 years was 922 women. By 29 years of follow-up, the NNS had fallen to 414.
"At 10 years of follow-up, you haven’t even observed half of the deaths prevented. So follow-up of 20 years at a minimum is really critical to begin to see the full benefit of screening," according to Dr. Smith, senior director of cancer screening at the American Cancer Society in Atlanta.
Also, several of the major reviews estimated the absolute mortality benefit of screening by means of an intent-to-treat analysis based upon the number of women invited to screening in randomized trials. That approach, too, is highly problematic because commonly 30%-40% of women invited to breast cancer screening in randomized trials never actually present for mammography, he said.
"The difference between the number-needed-to-invite and number-needed-to-screen is quite a critical difference in these estimates of absolute benefit. If you want to measure the effectiveness, you have to appreciate that a letter of invitation doesn’t do anyone any good. You have to show up to get mammography in order to benefit from it," Dr. Smith observed.
All of the four recent major reviews – the Nordic Cochrane (Cochrane Database Syst. Rev. 2013;6:CD001877), the USPSTF (Ann. Intern. Med. 2009;151:727-37), the U.K. Independent Breast Screening Review (Br. J. Cancer 2013;108:2205-40), and the European Screening Network (EUROSCREEN) Review (J. Med. Screen. 2012;19 Suppl1:14-25) – painted different pictures of the benefits of mammographic screening because they focused on different age groups, with different screening and follow-up durations, and were inconsistent as to whether the appropriate yardstick was NNS or number-needed-to-invite.
Dr. Smith and his coinvestigators sought to level the playing field by reanalyzing each review, standardizing the data to the scenario utilized in the U.K. independent review. They picked the U.K. review as the reference because it was most recently published and it was led by renowned statistical experts who aren’t part of the debate over mammography’s value. The U.K. review scenario entailed screening every 3 years for 20 years starting at age 50 years, with a 20-year follow-up period and the endpoint being breast cancer mortality at ages 55-79 years. When the data were reanalyzed in this way, the magnitude of the difference between the high and low estimates of absolute benefit among the four major reviews dropped from more than 20-fold to less than 3-fold.
"The so-called controversy over the benefit of mammography screening as estimated from the trials is largely contrived," he declared. "In short, once you standardize the evidence to the same population, the same screening scenario, and the same duration of follow-up, then the differences in absolute benefit over 20 years in the reviews become really not so significant or important at all. They are hardly worth discussing, and are certainly not enough to question the value of mammography over a lifetime of screening."
The flip side of estimating the benefit of mammographic screening in terms of breast cancer deaths avoided is the harm from overdiagnosis of cancers that never would have been symptomatic during a woman’s lifetime and wouldn’t have been detected had screening mammography not been performed. Here again, the estimates reported in the four reviews differed widely because of the divergent analytic methods employed. The U.K. review concluded that for every death from breast cancer avoided via mammography, three people would be overdiagnosed, for an overdiagnosis rate of 19%. The Nordic Cochrane analysis estimated 10 cases of overdiagnosis for every breast cancer death avoided, for a 30% overdiagnosis rate. The USPSTF didn’t give an overdiagnosis estimate. The EUROSCREEN group calculated that for every two breast cancer deaths avoided there would be one case of overdiagnosis, for a 6.5% overdiagnosis rate.
Dr. Smith said the EUROSCREEN estimate of overdiagnosis is the one that rings true. The EUROSCREEN investigators have demonstrated that in estimating mammography overdiagnosis rates, it’s essential to adjust for trends over time in breast cancer incidence and for lead time bias. When that’s not done, estimated overdiagnosis rates run in the 30%-50% range. When adjustments are made, the overdiagnosis rates are in the 0%-10% range, with the EUROSCREEN estimate of 6.5% being representative (J. Med. Screen. 2012;19 Suppl 1:42-56).
The full details of the mega-review were recently published (Breast Cancer Management 2013;2:519-28 [doi:10.2217/bmt.13.53]). The mega-review was funded by the Center for Cancer Prevention, the Wolfson Institute of Preventive Medicine, Barts and the London School of Medicine and Dentistry, and Queen Mary University of London. Dr. Smith declared having no financial conflicts of interest.
I’m not a statistician, but the analysis presented by Dr. Smith makes common sense to me.
Basing benefit solely on those women invited to screen will clearly result in an underestimate of mammography’s true benefit if you’re going to count women who weren’t screened as being in the screened group. More and more, we are learning that breast cancer is a more indolent disease than we thought. Particularly in those with estrogen receptor–positive breast cancer, the most common subtype, many patients don’t recur and die for a decade or more. If you’re not following patients long enough to capture all of the recurrences and deaths from the disease, then you don’t get a full assessment of the value of screening earlier in the course of disease. Long-term follow-up is extremely important.
You’d think that point would be universally accepted in the world of breast cancer screening research, but it isn’t. Mammography is controversial. It takes on some of the same features as debates over global warming. Quite often we see that scientists are just as vulnerable to motivated reasoning as anybody else.
Dr. C. Kent Osborne is the SABCS codirector and director of the Dan L. Duncan Cancer Center and the Lester & Sue Smith Breast Center at Baylor College of Medicine, Houston. He made his comments during the post presentation discussion of the research.
I’m not a statistician, but the analysis presented by Dr. Smith makes common sense to me.
Basing benefit solely on those women invited to screen will clearly result in an underestimate of mammography’s true benefit if you’re going to count women who weren’t screened as being in the screened group. More and more, we are learning that breast cancer is a more indolent disease than we thought. Particularly in those with estrogen receptor–positive breast cancer, the most common subtype, many patients don’t recur and die for a decade or more. If you’re not following patients long enough to capture all of the recurrences and deaths from the disease, then you don’t get a full assessment of the value of screening earlier in the course of disease. Long-term follow-up is extremely important.
You’d think that point would be universally accepted in the world of breast cancer screening research, but it isn’t. Mammography is controversial. It takes on some of the same features as debates over global warming. Quite often we see that scientists are just as vulnerable to motivated reasoning as anybody else.
Dr. C. Kent Osborne is the SABCS codirector and director of the Dan L. Duncan Cancer Center and the Lester & Sue Smith Breast Center at Baylor College of Medicine, Houston. He made his comments during the post presentation discussion of the research.
I’m not a statistician, but the analysis presented by Dr. Smith makes common sense to me.
Basing benefit solely on those women invited to screen will clearly result in an underestimate of mammography’s true benefit if you’re going to count women who weren’t screened as being in the screened group. More and more, we are learning that breast cancer is a more indolent disease than we thought. Particularly in those with estrogen receptor–positive breast cancer, the most common subtype, many patients don’t recur and die for a decade or more. If you’re not following patients long enough to capture all of the recurrences and deaths from the disease, then you don’t get a full assessment of the value of screening earlier in the course of disease. Long-term follow-up is extremely important.
You’d think that point would be universally accepted in the world of breast cancer screening research, but it isn’t. Mammography is controversial. It takes on some of the same features as debates over global warming. Quite often we see that scientists are just as vulnerable to motivated reasoning as anybody else.
Dr. C. Kent Osborne is the SABCS codirector and director of the Dan L. Duncan Cancer Center and the Lester & Sue Smith Breast Center at Baylor College of Medicine, Houston. He made his comments during the post presentation discussion of the research.
SAN ANTONIO – The much-publicized wide disparities in the estimated value of mammographic screening for breast cancer reported in recent major reviews are overblown and largely an artifact of methodologic differences, according to a new examination of the evidence.
The four recent major reviews of the data regarding the absolute benefits of mammography came up with estimates ranging from 90-2,000 of the number of women who need to be screened (NNS) in order to prevent one death from breast cancer. That greater than 20-fold difference in estimated magnitude of benefit has done little to inspire public and physician confidence that mammography is a key tool in reducing cancer deaths.

But the two analyses with the least supportive outcomes – the Nordic Cochrane and U.S. Preventive Services Task Force (USPSTF-) analyses – used follow-up periods of 10 and 15 years, respectively. That follow-up is too short a time to assess the full value of mammographic screening, Robert A. Smith, Ph.D., asserted at the San Antonio Breast Cancer Symposium.
To illustrate: In a European mammographic screening study with a 30-year follow-up, the NNS after 10 years was 922 women. By 29 years of follow-up, the NNS had fallen to 414.
"At 10 years of follow-up, you haven’t even observed half of the deaths prevented. So follow-up of 20 years at a minimum is really critical to begin to see the full benefit of screening," according to Dr. Smith, senior director of cancer screening at the American Cancer Society in Atlanta.
Also, several of the major reviews estimated the absolute mortality benefit of screening by means of an intent-to-treat analysis based upon the number of women invited to screening in randomized trials. That approach, too, is highly problematic because commonly 30%-40% of women invited to breast cancer screening in randomized trials never actually present for mammography, he said.
"The difference between the number-needed-to-invite and number-needed-to-screen is quite a critical difference in these estimates of absolute benefit. If you want to measure the effectiveness, you have to appreciate that a letter of invitation doesn’t do anyone any good. You have to show up to get mammography in order to benefit from it," Dr. Smith observed.
All of the four recent major reviews – the Nordic Cochrane (Cochrane Database Syst. Rev. 2013;6:CD001877), the USPSTF (Ann. Intern. Med. 2009;151:727-37), the U.K. Independent Breast Screening Review (Br. J. Cancer 2013;108:2205-40), and the European Screening Network (EUROSCREEN) Review (J. Med. Screen. 2012;19 Suppl1:14-25) – painted different pictures of the benefits of mammographic screening because they focused on different age groups, with different screening and follow-up durations, and were inconsistent as to whether the appropriate yardstick was NNS or number-needed-to-invite.
Dr. Smith and his coinvestigators sought to level the playing field by reanalyzing each review, standardizing the data to the scenario utilized in the U.K. independent review. They picked the U.K. review as the reference because it was most recently published and it was led by renowned statistical experts who aren’t part of the debate over mammography’s value. The U.K. review scenario entailed screening every 3 years for 20 years starting at age 50 years, with a 20-year follow-up period and the endpoint being breast cancer mortality at ages 55-79 years. When the data were reanalyzed in this way, the magnitude of the difference between the high and low estimates of absolute benefit among the four major reviews dropped from more than 20-fold to less than 3-fold.
"The so-called controversy over the benefit of mammography screening as estimated from the trials is largely contrived," he declared. "In short, once you standardize the evidence to the same population, the same screening scenario, and the same duration of follow-up, then the differences in absolute benefit over 20 years in the reviews become really not so significant or important at all. They are hardly worth discussing, and are certainly not enough to question the value of mammography over a lifetime of screening."
The flip side of estimating the benefit of mammographic screening in terms of breast cancer deaths avoided is the harm from overdiagnosis of cancers that never would have been symptomatic during a woman’s lifetime and wouldn’t have been detected had screening mammography not been performed. Here again, the estimates reported in the four reviews differed widely because of the divergent analytic methods employed. The U.K. review concluded that for every death from breast cancer avoided via mammography, three people would be overdiagnosed, for an overdiagnosis rate of 19%. The Nordic Cochrane analysis estimated 10 cases of overdiagnosis for every breast cancer death avoided, for a 30% overdiagnosis rate. The USPSTF didn’t give an overdiagnosis estimate. The EUROSCREEN group calculated that for every two breast cancer deaths avoided there would be one case of overdiagnosis, for a 6.5% overdiagnosis rate.
Dr. Smith said the EUROSCREEN estimate of overdiagnosis is the one that rings true. The EUROSCREEN investigators have demonstrated that in estimating mammography overdiagnosis rates, it’s essential to adjust for trends over time in breast cancer incidence and for lead time bias. When that’s not done, estimated overdiagnosis rates run in the 30%-50% range. When adjustments are made, the overdiagnosis rates are in the 0%-10% range, with the EUROSCREEN estimate of 6.5% being representative (J. Med. Screen. 2012;19 Suppl 1:42-56).
The full details of the mega-review were recently published (Breast Cancer Management 2013;2:519-28 [doi:10.2217/bmt.13.53]). The mega-review was funded by the Center for Cancer Prevention, the Wolfson Institute of Preventive Medicine, Barts and the London School of Medicine and Dentistry, and Queen Mary University of London. Dr. Smith declared having no financial conflicts of interest.
SAN ANTONIO – The much-publicized wide disparities in the estimated value of mammographic screening for breast cancer reported in recent major reviews are overblown and largely an artifact of methodologic differences, according to a new examination of the evidence.
The four recent major reviews of the data regarding the absolute benefits of mammography came up with estimates ranging from 90-2,000 of the number of women who need to be screened (NNS) in order to prevent one death from breast cancer. That greater than 20-fold difference in estimated magnitude of benefit has done little to inspire public and physician confidence that mammography is a key tool in reducing cancer deaths.

But the two analyses with the least supportive outcomes – the Nordic Cochrane and U.S. Preventive Services Task Force (USPSTF-) analyses – used follow-up periods of 10 and 15 years, respectively. That follow-up is too short a time to assess the full value of mammographic screening, Robert A. Smith, Ph.D., asserted at the San Antonio Breast Cancer Symposium.
To illustrate: In a European mammographic screening study with a 30-year follow-up, the NNS after 10 years was 922 women. By 29 years of follow-up, the NNS had fallen to 414.
"At 10 years of follow-up, you haven’t even observed half of the deaths prevented. So follow-up of 20 years at a minimum is really critical to begin to see the full benefit of screening," according to Dr. Smith, senior director of cancer screening at the American Cancer Society in Atlanta.
Also, several of the major reviews estimated the absolute mortality benefit of screening by means of an intent-to-treat analysis based upon the number of women invited to screening in randomized trials. That approach, too, is highly problematic because commonly 30%-40% of women invited to breast cancer screening in randomized trials never actually present for mammography, he said.
"The difference between the number-needed-to-invite and number-needed-to-screen is quite a critical difference in these estimates of absolute benefit. If you want to measure the effectiveness, you have to appreciate that a letter of invitation doesn’t do anyone any good. You have to show up to get mammography in order to benefit from it," Dr. Smith observed.
All of the four recent major reviews – the Nordic Cochrane (Cochrane Database Syst. Rev. 2013;6:CD001877), the USPSTF (Ann. Intern. Med. 2009;151:727-37), the U.K. Independent Breast Screening Review (Br. J. Cancer 2013;108:2205-40), and the European Screening Network (EUROSCREEN) Review (J. Med. Screen. 2012;19 Suppl1:14-25) – painted different pictures of the benefits of mammographic screening because they focused on different age groups, with different screening and follow-up durations, and were inconsistent as to whether the appropriate yardstick was NNS or number-needed-to-invite.
Dr. Smith and his coinvestigators sought to level the playing field by reanalyzing each review, standardizing the data to the scenario utilized in the U.K. independent review. They picked the U.K. review as the reference because it was most recently published and it was led by renowned statistical experts who aren’t part of the debate over mammography’s value. The U.K. review scenario entailed screening every 3 years for 20 years starting at age 50 years, with a 20-year follow-up period and the endpoint being breast cancer mortality at ages 55-79 years. When the data were reanalyzed in this way, the magnitude of the difference between the high and low estimates of absolute benefit among the four major reviews dropped from more than 20-fold to less than 3-fold.
"The so-called controversy over the benefit of mammography screening as estimated from the trials is largely contrived," he declared. "In short, once you standardize the evidence to the same population, the same screening scenario, and the same duration of follow-up, then the differences in absolute benefit over 20 years in the reviews become really not so significant or important at all. They are hardly worth discussing, and are certainly not enough to question the value of mammography over a lifetime of screening."
The flip side of estimating the benefit of mammographic screening in terms of breast cancer deaths avoided is the harm from overdiagnosis of cancers that never would have been symptomatic during a woman’s lifetime and wouldn’t have been detected had screening mammography not been performed. Here again, the estimates reported in the four reviews differed widely because of the divergent analytic methods employed. The U.K. review concluded that for every death from breast cancer avoided via mammography, three people would be overdiagnosed, for an overdiagnosis rate of 19%. The Nordic Cochrane analysis estimated 10 cases of overdiagnosis for every breast cancer death avoided, for a 30% overdiagnosis rate. The USPSTF didn’t give an overdiagnosis estimate. The EUROSCREEN group calculated that for every two breast cancer deaths avoided there would be one case of overdiagnosis, for a 6.5% overdiagnosis rate.
Dr. Smith said the EUROSCREEN estimate of overdiagnosis is the one that rings true. The EUROSCREEN investigators have demonstrated that in estimating mammography overdiagnosis rates, it’s essential to adjust for trends over time in breast cancer incidence and for lead time bias. When that’s not done, estimated overdiagnosis rates run in the 30%-50% range. When adjustments are made, the overdiagnosis rates are in the 0%-10% range, with the EUROSCREEN estimate of 6.5% being representative (J. Med. Screen. 2012;19 Suppl 1:42-56).
The full details of the mega-review were recently published (Breast Cancer Management 2013;2:519-28 [doi:10.2217/bmt.13.53]). The mega-review was funded by the Center for Cancer Prevention, the Wolfson Institute of Preventive Medicine, Barts and the London School of Medicine and Dentistry, and Queen Mary University of London. Dr. Smith declared having no financial conflicts of interest.
EXPERT OPINION FROM SABCS 2013
New mega-review underscores mammography’s benefits
SAN ANTONIO – The much-publicized wide disparities in the estimated value of mammographic screening for breast cancer reported in recent major reviews are overblown and largely an artifact of methodologic differences, according to a new examination of the evidence.
The four recent major reviews of the data regarding the absolute benefits of mammography came up with estimates ranging from 90-2,000 of the number of women who need to be screened (NNS) in order to prevent one death from breast cancer. That greater than 20-fold difference in estimated magnitude of benefit has done little to inspire public and physician confidence that mammography is a key tool in reducing cancer deaths.
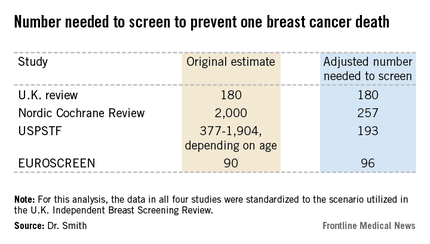
But the two analyses with the least supportive outcomes – the Nordic Cochrane and U.S. Preventive Services Task Force (USPSTF-) analyses – used follow-up periods of 10 and 15 years, respectively. That follow-up is too short a time to assess the full value of mammographic screening, Robert A. Smith, Ph.D., asserted at the San Antonio Breast Cancer Symposium.
To illustrate: In a European mammographic screening study with a 30-year follow-up, the NNS after 10 years was 922 women. By 29 years of follow-up, the NNS had fallen to 414.
"At 10 years of follow-up, you haven’t even observed half of the deaths prevented. So follow-up of 20 years at a minimum is really critical to begin to see the full benefit of screening," according to Dr. Smith, senior director of cancer screening at the American Cancer Society in Atlanta.
Also, several of the major reviews estimated the absolute mortality benefit of screening by means of an intent-to-treat analysis based upon the number of women invited to screening in randomized trials. That approach, too, is highly problematic because commonly 30%-40% of women invited to breast cancer screening in randomized trials never actually present for mammography, he said.
"The difference between the number-needed-to-invite and number-needed-to-screen is quite a critical difference in these estimates of absolute benefit. If you want to measure the effectiveness, you have to appreciate that a letter of invitation doesn’t do anyone any good. You have to show up to get mammography in order to benefit from it," Dr. Smith observed.
All of the four recent major reviews – the Nordic Cochrane (Cochrane Database Syst. Rev. 2013;6:CD001877), the USPSTF (Ann. Intern. Med. 2009;151:727-37), the U.K. Independent Breast Screening Review (Br. J. Cancer 2013;108:2205-40), and the European Screening Network (EUROSCREEN) Review (J. Med. Screen. 2012;19 Suppl1:14-25) – painted different pictures of the benefits of mammographic screening because they focused on different age groups, with different screening and follow-up durations, and were inconsistent as to whether the appropriate yardstick was NNS or number-needed-to-invite.
Dr. Smith and his coinvestigators sought to level the playing field by reanalyzing each review, standardizing the data to the scenario utilized in the U.K. independent review. They picked the U.K. review as the reference because it was most recently published and it was led by renowned statistical experts who aren’t part of the debate over mammography’s value. The U.K. review scenario entailed screening every 3 years for 20 years starting at age 50 years, with a 20-year follow-up period and the endpoint being breast cancer mortality at ages 55-79 years. When the data were reanalyzed in this way, the magnitude of the difference between the high and low estimates of absolute benefit among the four major reviews dropped from more than 20-fold to less than 3-fold.
"The so-called controversy over the benefit of mammography screening as estimated from the trials is largely contrived," he declared. "In short, once you standardize the evidence to the same population, the same screening scenario, and the same duration of follow-up, then the differences in absolute benefit over 20 years in the reviews become really not so significant or important at all. They are hardly worth discussing, and are certainly not enough to question the value of mammography over a lifetime of screening."
The flip side of estimating the benefit of mammographic screening in terms of breast cancer deaths avoided is the harm from overdiagnosis of cancers that never would have been symptomatic during a woman’s lifetime and wouldn’t have been detected had screening mammography not been performed. Here again, the estimates reported in the four reviews differed widely because of the divergent analytic methods employed. The U.K. review concluded that for every death from breast cancer avoided via mammography, three people would be overdiagnosed, for an overdiagnosis rate of 19%. The Nordic Cochrane analysis estimated 10 cases of overdiagnosis for every breast cancer death avoided, for a 30% overdiagnosis rate. The USPSTF didn’t give an overdiagnosis estimate. The EUROSCREEN group calculated that for every two breast cancer deaths avoided there would be one case of overdiagnosis, for a 6.5% overdiagnosis rate.
Dr. Smith said the EUROSCREEN estimate of overdiagnosis is the one that rings true. The EUROSCREEN investigators have demonstrated that in estimating mammography overdiagnosis rates, it’s essential to adjust for trends over time in breast cancer incidence and for lead time bias. When that’s not done, estimated overdiagnosis rates run in the 30%-50% range. When adjustments are made, the overdiagnosis rates are in the 0%-10% range, with the EUROSCREEN estimate of 6.5% being representative (J. Med. Screen. 2012;19 Suppl 1:42-56).
The full details of the mega-review were recently published (Breast Cancer Management 2013;2:519-28 [doi:10.2217/bmt.13.53]). The mega-review was funded by the Center for Cancer Prevention, the Wolfson Institute of Preventive Medicine, Barts and the London School of Medicine and Dentistry, and Queen Mary University of London. Dr. Smith declared having no financial conflicts of interest.
I’m not a statistician, but the analysis presented by Dr. Smith makes common sense to me.
Basing benefit solely on those women invited to screen will clearly result in an underestimate of mammography’s true benefit if you’re going to count women who weren’t screened as being in the screened group. More and more, we are learning that breast cancer is a more indolent disease than we thought. Particularly in those with estrogen receptor–positive breast cancer, the most common subtype, many patients don’t recur and die for a decade or more. If you’re not following patients long enough to capture all of the recurrences and deaths from the disease, then you don’t get a full assessment of the value of screening earlier in the course of disease. Long-term follow-up is extremely important.
You’d think that point would be universally accepted in the world of breast cancer screening research, but it isn’t. Mammography is controversial. It takes on some of the same features as debates over global warming. Quite often we see that scientists are just as vulnerable to motivated reasoning as anybody else.
Dr. C. Kent Osborne is the SABCS codirector and director of the Dan L. Duncan Cancer Center and the Lester & Sue Smith Breast Center at Baylor College of Medicine, Houston. He made his comments during the post presentation discussion of the research.
I’m not a statistician, but the analysis presented by Dr. Smith makes common sense to me.
Basing benefit solely on those women invited to screen will clearly result in an underestimate of mammography’s true benefit if you’re going to count women who weren’t screened as being in the screened group. More and more, we are learning that breast cancer is a more indolent disease than we thought. Particularly in those with estrogen receptor–positive breast cancer, the most common subtype, many patients don’t recur and die for a decade or more. If you’re not following patients long enough to capture all of the recurrences and deaths from the disease, then you don’t get a full assessment of the value of screening earlier in the course of disease. Long-term follow-up is extremely important.
You’d think that point would be universally accepted in the world of breast cancer screening research, but it isn’t. Mammography is controversial. It takes on some of the same features as debates over global warming. Quite often we see that scientists are just as vulnerable to motivated reasoning as anybody else.
Dr. C. Kent Osborne is the SABCS codirector and director of the Dan L. Duncan Cancer Center and the Lester & Sue Smith Breast Center at Baylor College of Medicine, Houston. He made his comments during the post presentation discussion of the research.
I’m not a statistician, but the analysis presented by Dr. Smith makes common sense to me.
Basing benefit solely on those women invited to screen will clearly result in an underestimate of mammography’s true benefit if you’re going to count women who weren’t screened as being in the screened group. More and more, we are learning that breast cancer is a more indolent disease than we thought. Particularly in those with estrogen receptor–positive breast cancer, the most common subtype, many patients don’t recur and die for a decade or more. If you’re not following patients long enough to capture all of the recurrences and deaths from the disease, then you don’t get a full assessment of the value of screening earlier in the course of disease. Long-term follow-up is extremely important.
You’d think that point would be universally accepted in the world of breast cancer screening research, but it isn’t. Mammography is controversial. It takes on some of the same features as debates over global warming. Quite often we see that scientists are just as vulnerable to motivated reasoning as anybody else.
Dr. C. Kent Osborne is the SABCS codirector and director of the Dan L. Duncan Cancer Center and the Lester & Sue Smith Breast Center at Baylor College of Medicine, Houston. He made his comments during the post presentation discussion of the research.
SAN ANTONIO – The much-publicized wide disparities in the estimated value of mammographic screening for breast cancer reported in recent major reviews are overblown and largely an artifact of methodologic differences, according to a new examination of the evidence.
The four recent major reviews of the data regarding the absolute benefits of mammography came up with estimates ranging from 90-2,000 of the number of women who need to be screened (NNS) in order to prevent one death from breast cancer. That greater than 20-fold difference in estimated magnitude of benefit has done little to inspire public and physician confidence that mammography is a key tool in reducing cancer deaths.

But the two analyses with the least supportive outcomes – the Nordic Cochrane and U.S. Preventive Services Task Force (USPSTF-) analyses – used follow-up periods of 10 and 15 years, respectively. That follow-up is too short a time to assess the full value of mammographic screening, Robert A. Smith, Ph.D., asserted at the San Antonio Breast Cancer Symposium.
To illustrate: In a European mammographic screening study with a 30-year follow-up, the NNS after 10 years was 922 women. By 29 years of follow-up, the NNS had fallen to 414.
"At 10 years of follow-up, you haven’t even observed half of the deaths prevented. So follow-up of 20 years at a minimum is really critical to begin to see the full benefit of screening," according to Dr. Smith, senior director of cancer screening at the American Cancer Society in Atlanta.
Also, several of the major reviews estimated the absolute mortality benefit of screening by means of an intent-to-treat analysis based upon the number of women invited to screening in randomized trials. That approach, too, is highly problematic because commonly 30%-40% of women invited to breast cancer screening in randomized trials never actually present for mammography, he said.
"The difference between the number-needed-to-invite and number-needed-to-screen is quite a critical difference in these estimates of absolute benefit. If you want to measure the effectiveness, you have to appreciate that a letter of invitation doesn’t do anyone any good. You have to show up to get mammography in order to benefit from it," Dr. Smith observed.
All of the four recent major reviews – the Nordic Cochrane (Cochrane Database Syst. Rev. 2013;6:CD001877), the USPSTF (Ann. Intern. Med. 2009;151:727-37), the U.K. Independent Breast Screening Review (Br. J. Cancer 2013;108:2205-40), and the European Screening Network (EUROSCREEN) Review (J. Med. Screen. 2012;19 Suppl1:14-25) – painted different pictures of the benefits of mammographic screening because they focused on different age groups, with different screening and follow-up durations, and were inconsistent as to whether the appropriate yardstick was NNS or number-needed-to-invite.
Dr. Smith and his coinvestigators sought to level the playing field by reanalyzing each review, standardizing the data to the scenario utilized in the U.K. independent review. They picked the U.K. review as the reference because it was most recently published and it was led by renowned statistical experts who aren’t part of the debate over mammography’s value. The U.K. review scenario entailed screening every 3 years for 20 years starting at age 50 years, with a 20-year follow-up period and the endpoint being breast cancer mortality at ages 55-79 years. When the data were reanalyzed in this way, the magnitude of the difference between the high and low estimates of absolute benefit among the four major reviews dropped from more than 20-fold to less than 3-fold.
"The so-called controversy over the benefit of mammography screening as estimated from the trials is largely contrived," he declared. "In short, once you standardize the evidence to the same population, the same screening scenario, and the same duration of follow-up, then the differences in absolute benefit over 20 years in the reviews become really not so significant or important at all. They are hardly worth discussing, and are certainly not enough to question the value of mammography over a lifetime of screening."
The flip side of estimating the benefit of mammographic screening in terms of breast cancer deaths avoided is the harm from overdiagnosis of cancers that never would have been symptomatic during a woman’s lifetime and wouldn’t have been detected had screening mammography not been performed. Here again, the estimates reported in the four reviews differed widely because of the divergent analytic methods employed. The U.K. review concluded that for every death from breast cancer avoided via mammography, three people would be overdiagnosed, for an overdiagnosis rate of 19%. The Nordic Cochrane analysis estimated 10 cases of overdiagnosis for every breast cancer death avoided, for a 30% overdiagnosis rate. The USPSTF didn’t give an overdiagnosis estimate. The EUROSCREEN group calculated that for every two breast cancer deaths avoided there would be one case of overdiagnosis, for a 6.5% overdiagnosis rate.
Dr. Smith said the EUROSCREEN estimate of overdiagnosis is the one that rings true. The EUROSCREEN investigators have demonstrated that in estimating mammography overdiagnosis rates, it’s essential to adjust for trends over time in breast cancer incidence and for lead time bias. When that’s not done, estimated overdiagnosis rates run in the 30%-50% range. When adjustments are made, the overdiagnosis rates are in the 0%-10% range, with the EUROSCREEN estimate of 6.5% being representative (J. Med. Screen. 2012;19 Suppl 1:42-56).
The full details of the mega-review were recently published (Breast Cancer Management 2013;2:519-28 [doi:10.2217/bmt.13.53]). The mega-review was funded by the Center for Cancer Prevention, the Wolfson Institute of Preventive Medicine, Barts and the London School of Medicine and Dentistry, and Queen Mary University of London. Dr. Smith declared having no financial conflicts of interest.
SAN ANTONIO – The much-publicized wide disparities in the estimated value of mammographic screening for breast cancer reported in recent major reviews are overblown and largely an artifact of methodologic differences, according to a new examination of the evidence.
The four recent major reviews of the data regarding the absolute benefits of mammography came up with estimates ranging from 90-2,000 of the number of women who need to be screened (NNS) in order to prevent one death from breast cancer. That greater than 20-fold difference in estimated magnitude of benefit has done little to inspire public and physician confidence that mammography is a key tool in reducing cancer deaths.

But the two analyses with the least supportive outcomes – the Nordic Cochrane and U.S. Preventive Services Task Force (USPSTF-) analyses – used follow-up periods of 10 and 15 years, respectively. That follow-up is too short a time to assess the full value of mammographic screening, Robert A. Smith, Ph.D., asserted at the San Antonio Breast Cancer Symposium.
To illustrate: In a European mammographic screening study with a 30-year follow-up, the NNS after 10 years was 922 women. By 29 years of follow-up, the NNS had fallen to 414.
"At 10 years of follow-up, you haven’t even observed half of the deaths prevented. So follow-up of 20 years at a minimum is really critical to begin to see the full benefit of screening," according to Dr. Smith, senior director of cancer screening at the American Cancer Society in Atlanta.
Also, several of the major reviews estimated the absolute mortality benefit of screening by means of an intent-to-treat analysis based upon the number of women invited to screening in randomized trials. That approach, too, is highly problematic because commonly 30%-40% of women invited to breast cancer screening in randomized trials never actually present for mammography, he said.
"The difference between the number-needed-to-invite and number-needed-to-screen is quite a critical difference in these estimates of absolute benefit. If you want to measure the effectiveness, you have to appreciate that a letter of invitation doesn’t do anyone any good. You have to show up to get mammography in order to benefit from it," Dr. Smith observed.
All of the four recent major reviews – the Nordic Cochrane (Cochrane Database Syst. Rev. 2013;6:CD001877), the USPSTF (Ann. Intern. Med. 2009;151:727-37), the U.K. Independent Breast Screening Review (Br. J. Cancer 2013;108:2205-40), and the European Screening Network (EUROSCREEN) Review (J. Med. Screen. 2012;19 Suppl1:14-25) – painted different pictures of the benefits of mammographic screening because they focused on different age groups, with different screening and follow-up durations, and were inconsistent as to whether the appropriate yardstick was NNS or number-needed-to-invite.
Dr. Smith and his coinvestigators sought to level the playing field by reanalyzing each review, standardizing the data to the scenario utilized in the U.K. independent review. They picked the U.K. review as the reference because it was most recently published and it was led by renowned statistical experts who aren’t part of the debate over mammography’s value. The U.K. review scenario entailed screening every 3 years for 20 years starting at age 50 years, with a 20-year follow-up period and the endpoint being breast cancer mortality at ages 55-79 years. When the data were reanalyzed in this way, the magnitude of the difference between the high and low estimates of absolute benefit among the four major reviews dropped from more than 20-fold to less than 3-fold.
"The so-called controversy over the benefit of mammography screening as estimated from the trials is largely contrived," he declared. "In short, once you standardize the evidence to the same population, the same screening scenario, and the same duration of follow-up, then the differences in absolute benefit over 20 years in the reviews become really not so significant or important at all. They are hardly worth discussing, and are certainly not enough to question the value of mammography over a lifetime of screening."
The flip side of estimating the benefit of mammographic screening in terms of breast cancer deaths avoided is the harm from overdiagnosis of cancers that never would have been symptomatic during a woman’s lifetime and wouldn’t have been detected had screening mammography not been performed. Here again, the estimates reported in the four reviews differed widely because of the divergent analytic methods employed. The U.K. review concluded that for every death from breast cancer avoided via mammography, three people would be overdiagnosed, for an overdiagnosis rate of 19%. The Nordic Cochrane analysis estimated 10 cases of overdiagnosis for every breast cancer death avoided, for a 30% overdiagnosis rate. The USPSTF didn’t give an overdiagnosis estimate. The EUROSCREEN group calculated that for every two breast cancer deaths avoided there would be one case of overdiagnosis, for a 6.5% overdiagnosis rate.
Dr. Smith said the EUROSCREEN estimate of overdiagnosis is the one that rings true. The EUROSCREEN investigators have demonstrated that in estimating mammography overdiagnosis rates, it’s essential to adjust for trends over time in breast cancer incidence and for lead time bias. When that’s not done, estimated overdiagnosis rates run in the 30%-50% range. When adjustments are made, the overdiagnosis rates are in the 0%-10% range, with the EUROSCREEN estimate of 6.5% being representative (J. Med. Screen. 2012;19 Suppl 1:42-56).
The full details of the mega-review were recently published (Breast Cancer Management 2013;2:519-28 [doi:10.2217/bmt.13.53]). The mega-review was funded by the Center for Cancer Prevention, the Wolfson Institute of Preventive Medicine, Barts and the London School of Medicine and Dentistry, and Queen Mary University of London. Dr. Smith declared having no financial conflicts of interest.
EXPERT OPINION FROM SABCS 2013
No survival benefit to bisphosphonate in chemoresistant breast cancer
The bisphosphonate zolendronate didn't improve survival in patients with chemoresistant breast cancer, according to results from the phase III NATAN trial.
In an interview at the San Antonio Breast Cancer Symposium, Dr. Gunter von Minckwitz discusses the trial's results and clinical implications, and whether a role remains for bisphosphonates in postmenopausal patients.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
The bisphosphonate zolendronate didn't improve survival in patients with chemoresistant breast cancer, according to results from the phase III NATAN trial.
In an interview at the San Antonio Breast Cancer Symposium, Dr. Gunter von Minckwitz discusses the trial's results and clinical implications, and whether a role remains for bisphosphonates in postmenopausal patients.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
The bisphosphonate zolendronate didn't improve survival in patients with chemoresistant breast cancer, according to results from the phase III NATAN trial.
In an interview at the San Antonio Breast Cancer Symposium, Dr. Gunter von Minckwitz discusses the trial's results and clinical implications, and whether a role remains for bisphosphonates in postmenopausal patients.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Exercise dampens aromatase inhibitor–related joint pain
SAN ANTONIO – Adopting a standard exercise program resulted in a clinically meaningful 30% reduction in aromatase inhibitor-associated joint pain in breast cancer patients who participated in a year-long randomized trial.
The exercise prescription utilized in the HOPE (Hormones and Physical Exercise) trial was what’s recommended in national guidelines both for cancer survivors and healthy adults: 150 minutes per week of at least moderate-intensity aerobic activity, such as brisk walking, along with two strength-training sessions per week, Melinda L. Irwin, Ph.D., explained at the San Antonio Breast Cancer Symposium.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
The HOPE studies enrolled 121 postmenopausal women who had stage 1-3, hormone receptor–positive breast cancers and were physically inactive and overweight yet physically able to exercise. At enrollment, they were experiencing moderate aromatase inhibitor (AI)–associated joint pain, defined as a score of 5-7 on the 0-10 Brief Pain Inventory (BPI), after about 18 months on the medication. Roughly two-thirds of participants had no history of joint pain prior to starting AI therapy; the rest reported the AI exacerbated their preexisting joint pain. Subjects were randomized to the exercise program or to usual care, which included written information about the importance of exercise.
The primary study endpoint was the 12-month change in BPI worst pain score, which dropped by an average of 30% among the exercise group. This translated to an improvement in pain level from moderate at baseline to mild at follow-up. In addition, BPI scores rating pain severity and pain interference improved by about 20%. In contrast, patients in the usual care control group experienced a slight increase in BPI scores in all three domains over time, added Dr. Irwin, co-leader of the cancer prevention and control research program at Yale University Cancer Center, New Haven, Conn.
This degree of improvement in joint pain is greater than reported in studies of glucosamine, acupuncture, or vitamin D supplementation, she noted.
The improvement in pain scores in the exercise group was greater at 12 months than at 3 or 6, suggesting that a year-long exercise program is probably necessary to see sustained reduction in joint pain.
At 12 months of follow-up, women in the exercise group averaged 159 minutes of physical activity per week, 110 minutes more than controls. Compliance with the supervised exercise program was notably good, with women attending an average of 70% of the twice-weekly small-group strength-training sessions.
In addition to the improvement in AI-related arthralgias, the exercise group experienced ancillary benefits: a mean 6.5% improvement in peak oxygen consumption, or VO2 max, compared with baseline, along with a 3% reduction in body weight.
HOPE was the first randomized trial to examine the effects of exercise on AI side effects in breast cancer patients. The impetus for the study was the recognition that arthralgias are the most common reason for poor adherence to and discontinuation of AI therapy. Up to 20% of breast cancer patients discontinue their AI within the first year. And both early discontinuation and poor adherence have been shown to be predictive of increased mortality risk.
The HOPE results received an enthusiastic audience reception. Physicians were particularly impressed with the 70% exercise adherence rate over the course of a year. They asked how they can keep their previously sedentary patients’ commitment to regular exercise from waning after an initial burst of enthusiasm, as so often happens.
Dr. Irwin replied that adherence to lifestyle change is always a challenge. Social support is quite helpful. The exercise group in HOPE received a paid gym membership and met in small groups with a personal trainer twice weekly.
"The women really bonded with each other. And there are now a growing number of free programs throughout the country, which give cancer survivors a start on an exercise program with a free gym membership for several months. For example, the Livestrong Foundation has partnered with the YMCA to offer free exercise programs for cancer survivors at local Ys," she said.
The HOPE trial was funded by the National Cancer Institute. Dr. Irwin reported having no financial conflicts of interest.
SAN ANTONIO – Adopting a standard exercise program resulted in a clinically meaningful 30% reduction in aromatase inhibitor-associated joint pain in breast cancer patients who participated in a year-long randomized trial.
The exercise prescription utilized in the HOPE (Hormones and Physical Exercise) trial was what’s recommended in national guidelines both for cancer survivors and healthy adults: 150 minutes per week of at least moderate-intensity aerobic activity, such as brisk walking, along with two strength-training sessions per week, Melinda L. Irwin, Ph.D., explained at the San Antonio Breast Cancer Symposium.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
The HOPE studies enrolled 121 postmenopausal women who had stage 1-3, hormone receptor–positive breast cancers and were physically inactive and overweight yet physically able to exercise. At enrollment, they were experiencing moderate aromatase inhibitor (AI)–associated joint pain, defined as a score of 5-7 on the 0-10 Brief Pain Inventory (BPI), after about 18 months on the medication. Roughly two-thirds of participants had no history of joint pain prior to starting AI therapy; the rest reported the AI exacerbated their preexisting joint pain. Subjects were randomized to the exercise program or to usual care, which included written information about the importance of exercise.
The primary study endpoint was the 12-month change in BPI worst pain score, which dropped by an average of 30% among the exercise group. This translated to an improvement in pain level from moderate at baseline to mild at follow-up. In addition, BPI scores rating pain severity and pain interference improved by about 20%. In contrast, patients in the usual care control group experienced a slight increase in BPI scores in all three domains over time, added Dr. Irwin, co-leader of the cancer prevention and control research program at Yale University Cancer Center, New Haven, Conn.
This degree of improvement in joint pain is greater than reported in studies of glucosamine, acupuncture, or vitamin D supplementation, she noted.
The improvement in pain scores in the exercise group was greater at 12 months than at 3 or 6, suggesting that a year-long exercise program is probably necessary to see sustained reduction in joint pain.
At 12 months of follow-up, women in the exercise group averaged 159 minutes of physical activity per week, 110 minutes more than controls. Compliance with the supervised exercise program was notably good, with women attending an average of 70% of the twice-weekly small-group strength-training sessions.
In addition to the improvement in AI-related arthralgias, the exercise group experienced ancillary benefits: a mean 6.5% improvement in peak oxygen consumption, or VO2 max, compared with baseline, along with a 3% reduction in body weight.
HOPE was the first randomized trial to examine the effects of exercise on AI side effects in breast cancer patients. The impetus for the study was the recognition that arthralgias are the most common reason for poor adherence to and discontinuation of AI therapy. Up to 20% of breast cancer patients discontinue their AI within the first year. And both early discontinuation and poor adherence have been shown to be predictive of increased mortality risk.
The HOPE results received an enthusiastic audience reception. Physicians were particularly impressed with the 70% exercise adherence rate over the course of a year. They asked how they can keep their previously sedentary patients’ commitment to regular exercise from waning after an initial burst of enthusiasm, as so often happens.
Dr. Irwin replied that adherence to lifestyle change is always a challenge. Social support is quite helpful. The exercise group in HOPE received a paid gym membership and met in small groups with a personal trainer twice weekly.
"The women really bonded with each other. And there are now a growing number of free programs throughout the country, which give cancer survivors a start on an exercise program with a free gym membership for several months. For example, the Livestrong Foundation has partnered with the YMCA to offer free exercise programs for cancer survivors at local Ys," she said.
The HOPE trial was funded by the National Cancer Institute. Dr. Irwin reported having no financial conflicts of interest.
SAN ANTONIO – Adopting a standard exercise program resulted in a clinically meaningful 30% reduction in aromatase inhibitor-associated joint pain in breast cancer patients who participated in a year-long randomized trial.
The exercise prescription utilized in the HOPE (Hormones and Physical Exercise) trial was what’s recommended in national guidelines both for cancer survivors and healthy adults: 150 minutes per week of at least moderate-intensity aerobic activity, such as brisk walking, along with two strength-training sessions per week, Melinda L. Irwin, Ph.D., explained at the San Antonio Breast Cancer Symposium.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
The HOPE studies enrolled 121 postmenopausal women who had stage 1-3, hormone receptor–positive breast cancers and were physically inactive and overweight yet physically able to exercise. At enrollment, they were experiencing moderate aromatase inhibitor (AI)–associated joint pain, defined as a score of 5-7 on the 0-10 Brief Pain Inventory (BPI), after about 18 months on the medication. Roughly two-thirds of participants had no history of joint pain prior to starting AI therapy; the rest reported the AI exacerbated their preexisting joint pain. Subjects were randomized to the exercise program or to usual care, which included written information about the importance of exercise.
The primary study endpoint was the 12-month change in BPI worst pain score, which dropped by an average of 30% among the exercise group. This translated to an improvement in pain level from moderate at baseline to mild at follow-up. In addition, BPI scores rating pain severity and pain interference improved by about 20%. In contrast, patients in the usual care control group experienced a slight increase in BPI scores in all three domains over time, added Dr. Irwin, co-leader of the cancer prevention and control research program at Yale University Cancer Center, New Haven, Conn.
This degree of improvement in joint pain is greater than reported in studies of glucosamine, acupuncture, or vitamin D supplementation, she noted.
The improvement in pain scores in the exercise group was greater at 12 months than at 3 or 6, suggesting that a year-long exercise program is probably necessary to see sustained reduction in joint pain.
At 12 months of follow-up, women in the exercise group averaged 159 minutes of physical activity per week, 110 minutes more than controls. Compliance with the supervised exercise program was notably good, with women attending an average of 70% of the twice-weekly small-group strength-training sessions.
In addition to the improvement in AI-related arthralgias, the exercise group experienced ancillary benefits: a mean 6.5% improvement in peak oxygen consumption, or VO2 max, compared with baseline, along with a 3% reduction in body weight.
HOPE was the first randomized trial to examine the effects of exercise on AI side effects in breast cancer patients. The impetus for the study was the recognition that arthralgias are the most common reason for poor adherence to and discontinuation of AI therapy. Up to 20% of breast cancer patients discontinue their AI within the first year. And both early discontinuation and poor adherence have been shown to be predictive of increased mortality risk.
The HOPE results received an enthusiastic audience reception. Physicians were particularly impressed with the 70% exercise adherence rate over the course of a year. They asked how they can keep their previously sedentary patients’ commitment to regular exercise from waning after an initial burst of enthusiasm, as so often happens.
Dr. Irwin replied that adherence to lifestyle change is always a challenge. Social support is quite helpful. The exercise group in HOPE received a paid gym membership and met in small groups with a personal trainer twice weekly.
"The women really bonded with each other. And there are now a growing number of free programs throughout the country, which give cancer survivors a start on an exercise program with a free gym membership for several months. For example, the Livestrong Foundation has partnered with the YMCA to offer free exercise programs for cancer survivors at local Ys," she said.
The HOPE trial was funded by the National Cancer Institute. Dr. Irwin reported having no financial conflicts of interest.
AT SABCS 20123
Major finding: Breast cancer patients experienced a 30% reduction in aromatase inhibitor–related joint pain, with pain scores going from moderate to mild, in response to a year-long exercise intervention.
Data source: The HOPE study was a 1-year randomized trial in which 121 postmenopausal breast cancer patients with significant pain from aromatase inhibitor–related arthralgias were randomized to a structured exercise program or to usual care.
Disclosures: The trial was funded by the National Cancer Institute. The presenter reported having no financial conflicts.















