User login
AHA: Poor real-world adherence to NOACs
ORLANDO – Adherence to the novel oral anticoagulants (NOACs) is surprisingly poor in clinical practice, Xiaoxi Yao, Ph.D., reported at the American Heart Association scientific sessions.
Her retrospective study of nearly 65,000 patients with atrial fibrillation who initiated therapy with apixaban, dabigatran, rivaroxaban, or warfarin showed that during a median 1.1 years of follow-up fewer than half of all patients were treatment adherent, with adherence defined as possession of sufficient medication to cover at least 80% of days.
Adherence rates, while uniformly suboptimal, nevertheless varied considerably: lowest at 38.5% for dabigatran, followed by 40.2% for warfarin, 50.5% for rivaroxaban, and 61.9% for apixaban.
This poor adherence to NOACs in real-world clinical practice is surprising in light of the drugs’ greater convenience, with fewer drug interactions than warfarin and no need for laboratory monitoring, observed Dr. Yao of the Mayo Clinic in Rochester, Minn.
It’s possible, although speculative, that the NOACs’ greater convenience paradoxically contributes to the low adherence rates, since unlike warfarin, NOACs don’t require regular interactions with the health care system for INR monitoring. And then there is the hefty cost of the novel agents, she added.
The study population consisted of 3,900 patients with atrial fibrillation who initiated oral anticoagulation with apixaban (Eliquis), 10,235 who started on dabigatran (Pradaxa), 12,366 on rivaroxaban (Xarelto), and 38,190 on warfarin. The analysis utilized claims data from a large U.S. commercial insurance database.
Adherence rates were better among patients with greater stroke risk as reflected by their CHA2DS2-VASc scores. For example, at the high end of the adherence spectrum, the adherence rate for apixaban was 50% in patients with a CHA2DS2-VASc score of 0-1, rising to 62% with a score of 2-3 and 64% with a score of 4 or more. The corresponding adherence rates for dabigatran were 25% in patients with a CHA2DS2-VASc of 0-1, 40% among those with a score of 2-3, and 42% in patients with a score of 4 or higher.
Dr. Yao and coinvestigators were interested in whether lower adherence to oral anticoagulation was associated with worse outcomes. This proved to be the case with regard to stroke rate for patients with a CHA2DS2-VASc score of 2 or more, where a clear dose-response relationship was evident between the event rate and cumulative time off oral anticoagulation during follow-up.
Among patients with a CHA2DS2-VASc of 2 or 3, the stroke rate was nearly twice as high among those off oral anticoagulation for a total of 3-6 months and three times greater if off therapy for more than 6 months than in those with a total time off of less than 1 week. The stroke rate was even higher in patients with a CHA2DS2-VASc of 4 or more who had suboptimal adherence.
An unexpected finding, she continued, was that among patients with a CHA2DS2-VASc score of 2 or more there was no significant relationship between cumulative time off oral anticoagulation and the risk of major bleeding unless they were off treatment for a total of 6 months or more; only then was the major bleeding risk lower than in patients whose total time off therapy was less than a week. Also, one would expect that when patients are off oral anticoagulation they should be at significantly lower risk of intracranial hemorrhage than when on-therapy, but this proved not to be the case.
For patients at substantial stroke risk as indicated by a CHA2DS2-VASc score of at least 2, this finding about off-treatment bleeding risk actually constitutes a good argument for sticking to their medication, in Dr. Yao’s view.
“Physicians and patients often fear bleeding, especially intracranial hemorrhage, but we found that for patients at higher risk for stroke there is little difference in intracranial hemorrhage risk whether you’re on or off of oral anticoagulation. So higher-risk patients should definitely adhere to their medication because of the stroke prevention benefit. However, in low-risk patients with a CHA2DS2-VASc of 0-1, the benefits of oral anticoagulation may not always outweigh the harm,” she said.
Dr. Yao reported having no financial conflicts of interest regarding her study.
ORLANDO – Adherence to the novel oral anticoagulants (NOACs) is surprisingly poor in clinical practice, Xiaoxi Yao, Ph.D., reported at the American Heart Association scientific sessions.
Her retrospective study of nearly 65,000 patients with atrial fibrillation who initiated therapy with apixaban, dabigatran, rivaroxaban, or warfarin showed that during a median 1.1 years of follow-up fewer than half of all patients were treatment adherent, with adherence defined as possession of sufficient medication to cover at least 80% of days.
Adherence rates, while uniformly suboptimal, nevertheless varied considerably: lowest at 38.5% for dabigatran, followed by 40.2% for warfarin, 50.5% for rivaroxaban, and 61.9% for apixaban.
This poor adherence to NOACs in real-world clinical practice is surprising in light of the drugs’ greater convenience, with fewer drug interactions than warfarin and no need for laboratory monitoring, observed Dr. Yao of the Mayo Clinic in Rochester, Minn.
It’s possible, although speculative, that the NOACs’ greater convenience paradoxically contributes to the low adherence rates, since unlike warfarin, NOACs don’t require regular interactions with the health care system for INR monitoring. And then there is the hefty cost of the novel agents, she added.
The study population consisted of 3,900 patients with atrial fibrillation who initiated oral anticoagulation with apixaban (Eliquis), 10,235 who started on dabigatran (Pradaxa), 12,366 on rivaroxaban (Xarelto), and 38,190 on warfarin. The analysis utilized claims data from a large U.S. commercial insurance database.
Adherence rates were better among patients with greater stroke risk as reflected by their CHA2DS2-VASc scores. For example, at the high end of the adherence spectrum, the adherence rate for apixaban was 50% in patients with a CHA2DS2-VASc score of 0-1, rising to 62% with a score of 2-3 and 64% with a score of 4 or more. The corresponding adherence rates for dabigatran were 25% in patients with a CHA2DS2-VASc of 0-1, 40% among those with a score of 2-3, and 42% in patients with a score of 4 or higher.
Dr. Yao and coinvestigators were interested in whether lower adherence to oral anticoagulation was associated with worse outcomes. This proved to be the case with regard to stroke rate for patients with a CHA2DS2-VASc score of 2 or more, where a clear dose-response relationship was evident between the event rate and cumulative time off oral anticoagulation during follow-up.
Among patients with a CHA2DS2-VASc of 2 or 3, the stroke rate was nearly twice as high among those off oral anticoagulation for a total of 3-6 months and three times greater if off therapy for more than 6 months than in those with a total time off of less than 1 week. The stroke rate was even higher in patients with a CHA2DS2-VASc of 4 or more who had suboptimal adherence.
An unexpected finding, she continued, was that among patients with a CHA2DS2-VASc score of 2 or more there was no significant relationship between cumulative time off oral anticoagulation and the risk of major bleeding unless they were off treatment for a total of 6 months or more; only then was the major bleeding risk lower than in patients whose total time off therapy was less than a week. Also, one would expect that when patients are off oral anticoagulation they should be at significantly lower risk of intracranial hemorrhage than when on-therapy, but this proved not to be the case.
For patients at substantial stroke risk as indicated by a CHA2DS2-VASc score of at least 2, this finding about off-treatment bleeding risk actually constitutes a good argument for sticking to their medication, in Dr. Yao’s view.
“Physicians and patients often fear bleeding, especially intracranial hemorrhage, but we found that for patients at higher risk for stroke there is little difference in intracranial hemorrhage risk whether you’re on or off of oral anticoagulation. So higher-risk patients should definitely adhere to their medication because of the stroke prevention benefit. However, in low-risk patients with a CHA2DS2-VASc of 0-1, the benefits of oral anticoagulation may not always outweigh the harm,” she said.
Dr. Yao reported having no financial conflicts of interest regarding her study.
ORLANDO – Adherence to the novel oral anticoagulants (NOACs) is surprisingly poor in clinical practice, Xiaoxi Yao, Ph.D., reported at the American Heart Association scientific sessions.
Her retrospective study of nearly 65,000 patients with atrial fibrillation who initiated therapy with apixaban, dabigatran, rivaroxaban, or warfarin showed that during a median 1.1 years of follow-up fewer than half of all patients were treatment adherent, with adherence defined as possession of sufficient medication to cover at least 80% of days.
Adherence rates, while uniformly suboptimal, nevertheless varied considerably: lowest at 38.5% for dabigatran, followed by 40.2% for warfarin, 50.5% for rivaroxaban, and 61.9% for apixaban.
This poor adherence to NOACs in real-world clinical practice is surprising in light of the drugs’ greater convenience, with fewer drug interactions than warfarin and no need for laboratory monitoring, observed Dr. Yao of the Mayo Clinic in Rochester, Minn.
It’s possible, although speculative, that the NOACs’ greater convenience paradoxically contributes to the low adherence rates, since unlike warfarin, NOACs don’t require regular interactions with the health care system for INR monitoring. And then there is the hefty cost of the novel agents, she added.
The study population consisted of 3,900 patients with atrial fibrillation who initiated oral anticoagulation with apixaban (Eliquis), 10,235 who started on dabigatran (Pradaxa), 12,366 on rivaroxaban (Xarelto), and 38,190 on warfarin. The analysis utilized claims data from a large U.S. commercial insurance database.
Adherence rates were better among patients with greater stroke risk as reflected by their CHA2DS2-VASc scores. For example, at the high end of the adherence spectrum, the adherence rate for apixaban was 50% in patients with a CHA2DS2-VASc score of 0-1, rising to 62% with a score of 2-3 and 64% with a score of 4 or more. The corresponding adherence rates for dabigatran were 25% in patients with a CHA2DS2-VASc of 0-1, 40% among those with a score of 2-3, and 42% in patients with a score of 4 or higher.
Dr. Yao and coinvestigators were interested in whether lower adherence to oral anticoagulation was associated with worse outcomes. This proved to be the case with regard to stroke rate for patients with a CHA2DS2-VASc score of 2 or more, where a clear dose-response relationship was evident between the event rate and cumulative time off oral anticoagulation during follow-up.
Among patients with a CHA2DS2-VASc of 2 or 3, the stroke rate was nearly twice as high among those off oral anticoagulation for a total of 3-6 months and three times greater if off therapy for more than 6 months than in those with a total time off of less than 1 week. The stroke rate was even higher in patients with a CHA2DS2-VASc of 4 or more who had suboptimal adherence.
An unexpected finding, she continued, was that among patients with a CHA2DS2-VASc score of 2 or more there was no significant relationship between cumulative time off oral anticoagulation and the risk of major bleeding unless they were off treatment for a total of 6 months or more; only then was the major bleeding risk lower than in patients whose total time off therapy was less than a week. Also, one would expect that when patients are off oral anticoagulation they should be at significantly lower risk of intracranial hemorrhage than when on-therapy, but this proved not to be the case.
For patients at substantial stroke risk as indicated by a CHA2DS2-VASc score of at least 2, this finding about off-treatment bleeding risk actually constitutes a good argument for sticking to their medication, in Dr. Yao’s view.
“Physicians and patients often fear bleeding, especially intracranial hemorrhage, but we found that for patients at higher risk for stroke there is little difference in intracranial hemorrhage risk whether you’re on or off of oral anticoagulation. So higher-risk patients should definitely adhere to their medication because of the stroke prevention benefit. However, in low-risk patients with a CHA2DS2-VASc of 0-1, the benefits of oral anticoagulation may not always outweigh the harm,” she said.
Dr. Yao reported having no financial conflicts of interest regarding her study.
AT THE AHA SCIENTIFIC SESSIONS
Key clinical point: Adherence to the novel oral anticoagulants by patients with atrial fibrillation is surprisingly poor outside the clinical trial setting.
Major finding: More than half of patients with atrial fibrillation who started on a novel oral anticoagulant were medication adherent less than 80% of the time.
Data source: This was a retrospective study of nearly 65,000 patients with atrial fibrillation who initiated oral anticoagulant therapy and were then followed for a median of 1.1 years.
Disclosures: The presenter reported having no financial conflicts of interest regarding the study.,
EADV: DLX105 is a novel treatment strategy for Behçet’s flares
COPENHAGEN – An ultrasmall yet highly potent single-chain antibody fragment directed against tumor necrosis factor–alpha showed promise for the treatment of Behçet’s disease flares in a pilot phase II study presented at the annual congress of the European Academy of Dermatology and Venereology.
“We believe that we have something in our hands that may make a difference to these patients. Further development with follow-up studies is planned,” said Dr. Thomas Jung, chief medical officer at Delenex Therapeutics.
The agent, known for now as DLX105, utilizes the company’s proprietary PentraBody platform. DLX105 inhibits soluble as well as membrane-bound TNF-alpha. Because the protein antibody is so small, it has the capacity to penetrate into inflamed tissue, be it skin or cartilage. DLX105 is also being explored as a potential therapy for other flaring inflammatory skin and autoimmune disorders, he added.
“The antibody also leaves the tissue very rapidly. It doesn’t stick around as long as an IgG antibody. The typical half-life of this molecule is about 1 day. We believe this is actually an advantage when we talk about treating a flaring disease such as Behçet’s, where we want exposure for a certain time frame, but we don’t want to overexpose the patient over weeks and months when it is not really necessary,” Dr. Jung explained.
Behçet’s disease is a chronic autoimmune vasculitic disease which presents most often as oral ulcers, papulopustular skin lesions, genital ulcers, uveitis, and/or arthritis. Cardiac, gastrointestinal, and CNS involvement occurs less frequently. The pathogenesis of the disease is unknown; no specific cause or triggers have been identified. Behçet’s disease affects an estimated 20,000 people in the United States, but is far more common in Turkey, the Middle East, and Asia. All treatment is off-label; there is no approved therapy for Behçet’s disease. The most widely used agents are corticosteroids, colchicine, and cyclosporine, with the biologic TNF inhibitors often being utilized in an effort to prevent blindness when uveitis occurs.
Dr. Jung presented results of the small phase II open-label study, which involved six patients with Behçet’s disease for a mean of 10 years. All presented with a disease flare. All six had oral aphthous ulcers, four had skin lesions, three had joint pain, two had erythema nodosum, and one had genital ulcers. All participants received a single intravenous infusion of DLX105 at 10 mg/kg.
All of the oral ulcers healed within 1 week following the single dose of DLX105. Patients with joint pain reported it was substantially improved within 1-2 days after treatment. Genital lesions healed completely within 2 weeks. The skin lesions were also markedly improved. The clinical improvement was maintained up to 4 weeks post treatment.
The improvement in joint symptoms was not unexpected. DLX105 has been shown to inhibit human TNF-alpha–induced joint swelling in rats with an efficacy comparable to infliximab (Remicade), according to Dr. Jung.
Perhaps most impressively, Dr. Jung observed, erythema nodosum healed completely in both affected patients. Erythema nodosum can be notoriously difficult to treat. Indeed, one of the patients had erythema nodosum for 5 years during which multiple systemic therapies were employed without benefit.
COPENHAGEN – An ultrasmall yet highly potent single-chain antibody fragment directed against tumor necrosis factor–alpha showed promise for the treatment of Behçet’s disease flares in a pilot phase II study presented at the annual congress of the European Academy of Dermatology and Venereology.
“We believe that we have something in our hands that may make a difference to these patients. Further development with follow-up studies is planned,” said Dr. Thomas Jung, chief medical officer at Delenex Therapeutics.
The agent, known for now as DLX105, utilizes the company’s proprietary PentraBody platform. DLX105 inhibits soluble as well as membrane-bound TNF-alpha. Because the protein antibody is so small, it has the capacity to penetrate into inflamed tissue, be it skin or cartilage. DLX105 is also being explored as a potential therapy for other flaring inflammatory skin and autoimmune disorders, he added.
“The antibody also leaves the tissue very rapidly. It doesn’t stick around as long as an IgG antibody. The typical half-life of this molecule is about 1 day. We believe this is actually an advantage when we talk about treating a flaring disease such as Behçet’s, where we want exposure for a certain time frame, but we don’t want to overexpose the patient over weeks and months when it is not really necessary,” Dr. Jung explained.
Behçet’s disease is a chronic autoimmune vasculitic disease which presents most often as oral ulcers, papulopustular skin lesions, genital ulcers, uveitis, and/or arthritis. Cardiac, gastrointestinal, and CNS involvement occurs less frequently. The pathogenesis of the disease is unknown; no specific cause or triggers have been identified. Behçet’s disease affects an estimated 20,000 people in the United States, but is far more common in Turkey, the Middle East, and Asia. All treatment is off-label; there is no approved therapy for Behçet’s disease. The most widely used agents are corticosteroids, colchicine, and cyclosporine, with the biologic TNF inhibitors often being utilized in an effort to prevent blindness when uveitis occurs.
Dr. Jung presented results of the small phase II open-label study, which involved six patients with Behçet’s disease for a mean of 10 years. All presented with a disease flare. All six had oral aphthous ulcers, four had skin lesions, three had joint pain, two had erythema nodosum, and one had genital ulcers. All participants received a single intravenous infusion of DLX105 at 10 mg/kg.
All of the oral ulcers healed within 1 week following the single dose of DLX105. Patients with joint pain reported it was substantially improved within 1-2 days after treatment. Genital lesions healed completely within 2 weeks. The skin lesions were also markedly improved. The clinical improvement was maintained up to 4 weeks post treatment.
The improvement in joint symptoms was not unexpected. DLX105 has been shown to inhibit human TNF-alpha–induced joint swelling in rats with an efficacy comparable to infliximab (Remicade), according to Dr. Jung.
Perhaps most impressively, Dr. Jung observed, erythema nodosum healed completely in both affected patients. Erythema nodosum can be notoriously difficult to treat. Indeed, one of the patients had erythema nodosum for 5 years during which multiple systemic therapies were employed without benefit.
COPENHAGEN – An ultrasmall yet highly potent single-chain antibody fragment directed against tumor necrosis factor–alpha showed promise for the treatment of Behçet’s disease flares in a pilot phase II study presented at the annual congress of the European Academy of Dermatology and Venereology.
“We believe that we have something in our hands that may make a difference to these patients. Further development with follow-up studies is planned,” said Dr. Thomas Jung, chief medical officer at Delenex Therapeutics.
The agent, known for now as DLX105, utilizes the company’s proprietary PentraBody platform. DLX105 inhibits soluble as well as membrane-bound TNF-alpha. Because the protein antibody is so small, it has the capacity to penetrate into inflamed tissue, be it skin or cartilage. DLX105 is also being explored as a potential therapy for other flaring inflammatory skin and autoimmune disorders, he added.
“The antibody also leaves the tissue very rapidly. It doesn’t stick around as long as an IgG antibody. The typical half-life of this molecule is about 1 day. We believe this is actually an advantage when we talk about treating a flaring disease such as Behçet’s, where we want exposure for a certain time frame, but we don’t want to overexpose the patient over weeks and months when it is not really necessary,” Dr. Jung explained.
Behçet’s disease is a chronic autoimmune vasculitic disease which presents most often as oral ulcers, papulopustular skin lesions, genital ulcers, uveitis, and/or arthritis. Cardiac, gastrointestinal, and CNS involvement occurs less frequently. The pathogenesis of the disease is unknown; no specific cause or triggers have been identified. Behçet’s disease affects an estimated 20,000 people in the United States, but is far more common in Turkey, the Middle East, and Asia. All treatment is off-label; there is no approved therapy for Behçet’s disease. The most widely used agents are corticosteroids, colchicine, and cyclosporine, with the biologic TNF inhibitors often being utilized in an effort to prevent blindness when uveitis occurs.
Dr. Jung presented results of the small phase II open-label study, which involved six patients with Behçet’s disease for a mean of 10 years. All presented with a disease flare. All six had oral aphthous ulcers, four had skin lesions, three had joint pain, two had erythema nodosum, and one had genital ulcers. All participants received a single intravenous infusion of DLX105 at 10 mg/kg.
All of the oral ulcers healed within 1 week following the single dose of DLX105. Patients with joint pain reported it was substantially improved within 1-2 days after treatment. Genital lesions healed completely within 2 weeks. The skin lesions were also markedly improved. The clinical improvement was maintained up to 4 weeks post treatment.
The improvement in joint symptoms was not unexpected. DLX105 has been shown to inhibit human TNF-alpha–induced joint swelling in rats with an efficacy comparable to infliximab (Remicade), according to Dr. Jung.
Perhaps most impressively, Dr. Jung observed, erythema nodosum healed completely in both affected patients. Erythema nodosum can be notoriously difficult to treat. Indeed, one of the patients had erythema nodosum for 5 years during which multiple systemic therapies were employed without benefit.
AT THE EADV CONGRESS
Key clinical point: DLX105, a highly potent single-chain antibody fragment, shows early promise as a fast-acting treatment of flares of Behçet’s disease.
Major finding: Cutaneous, mucosal, and joint flares of Behçet’s disease resolved almost completely within several days following a single intravenous dose of DLX105 at 10 mg/kg.
Data source: This was a phase II open-label study of the effects of a single intravenous dose of DLX105 in 6 patients with flares of Behçet’s disease.
Disclosures: The study was sponsored by Delenex Therapeutics and presented by the Swiss company’s chief medical officer.
Short sleep duration in hypertensives ups mortality
ORLANDO – Hypertensive persons who sleep 5 hours or less per night have a significantly higher all-cause mortality rate than those who get more shut-eye, according to an analysis from the Penn State Adult Cohort Study.
“We found that the odds of all-cause mortality associated with hypertension increased in a dose-response manner as a function of the degree of objective short sleep duration, even after adjusting for a multitude of factors,” Julio Fernandez-Mendoza, Ph.D., reported at the American Heart Association scientific sessions.
The Penn State Adult Cohort consists of a random, general population sample of 1,741 men and women who enrolled in the study back in the 1990s, at a mean age of 48.7 years. As part of their comprehensive evaluation they were studied in the overnight sleep laboratory. The cohort has been followed for 15.5 years, during which 20% of subjects died.
As expected, hypertension was associated with increased risk of all-cause mortality in the Penn State Adult Cohort. But Dr. Fernandez-Mendoza and coinvestigators further dissected this association by incorporating the subjects’ objective sleep lab data, something that hadn’t been done in other studies. They found that while as a group the roughly 35% of study participants with hypertension had an adjusted 2.54-fold increased risk of all-cause mortality, compared with normotensive subjects, those who slept 6 or more hours at night – placing them at or above the 50th percentile for sleep duration – had a 1.75-fold increased risk, which just barely reached statistical significance.
In contrast, those who slept 5-6 hours per night were at 2.36-fold increased risk of all-cause mortality, while hypertensives in the bottom quartile for sleep duration with 5 hours or less of sleep had an even more robust 4.04-fold increased risk. All risk figures were determined in a multivariate logistic regression analysis extensively adjusted for age, gender, race, diabetes, obesity, smoking, depression, insomnia, sleep apnea, and history of heart disease or stroke.
This finding of an inverse association between sleep duration and all-cause mortality was consistent with the investigators’ study hypothesis that short sleep duration in hypertensive patients may be a marker of the severity of autonomic dysfunction. After all, it is known that the autonomic nervous system not only controls cardiovascular function, it also regulates sleep, explained Dr. Fernandez-Mendoza, a behavioral psychologist at Pennsylvania State University in Hershey.
Other possible explanations for the findings are that short sleep duration in hypertensive patients might be genetically driven or behaviorally induced, but he considers these less plausible.
In an interview, Dr. Fernandez-Mendoza said he and his coinvestigators have found the same relationship between short sleep duration and increased all-cause mortality in Penn State Adult Cohort members with diabetes or dyslipidemia, although he didn’t present those data at the AHA meeting.
If indeed short sleep duration is a marker of autonomic dysfunction, it would have important clinical implications: “Objective sleep duration may allow for refinement of estimates of mortality risk. I predict that someday cardiovascular risk calculators will incorporate sleep duration,” he said.
The Penn State Adult Cohort findings bring a measure of clarity to what has been a somewhat cloudy area, Dr. Fernandez-Mendoza said. Most prior epidemiologic studies of sleep’s impact on health have relied upon self-reported sleep duration, which is considerably less reliable than objectively measured sleep lab data. And many studies have looked at sleep duration as an isolated variable in relation to morbidity and mortality risk. This, he said, has contributed to public misunderstanding.
“We have people coming into the sleep lab thinking, ‘If I don’t get 7 hours of sleep I’m going to die,’ ” according to the sleep scientist. “But the paradigm we’ve developed, tied to what we know about autonomic control, is that the cardiovascular system and the sleep system are connected to each other. It doesn’t mean that short sleep kills you, it’s that the combination of the traditional cardiometabolic risk factors and short sleep increases risk of morbidity and mortality.”
Dr. Fernandez-Mendoza’s study was funded by an AHA Scientist Development Grant. He reported having no financial conflicts of interest.
ORLANDO – Hypertensive persons who sleep 5 hours or less per night have a significantly higher all-cause mortality rate than those who get more shut-eye, according to an analysis from the Penn State Adult Cohort Study.
“We found that the odds of all-cause mortality associated with hypertension increased in a dose-response manner as a function of the degree of objective short sleep duration, even after adjusting for a multitude of factors,” Julio Fernandez-Mendoza, Ph.D., reported at the American Heart Association scientific sessions.
The Penn State Adult Cohort consists of a random, general population sample of 1,741 men and women who enrolled in the study back in the 1990s, at a mean age of 48.7 years. As part of their comprehensive evaluation they were studied in the overnight sleep laboratory. The cohort has been followed for 15.5 years, during which 20% of subjects died.
As expected, hypertension was associated with increased risk of all-cause mortality in the Penn State Adult Cohort. But Dr. Fernandez-Mendoza and coinvestigators further dissected this association by incorporating the subjects’ objective sleep lab data, something that hadn’t been done in other studies. They found that while as a group the roughly 35% of study participants with hypertension had an adjusted 2.54-fold increased risk of all-cause mortality, compared with normotensive subjects, those who slept 6 or more hours at night – placing them at or above the 50th percentile for sleep duration – had a 1.75-fold increased risk, which just barely reached statistical significance.
In contrast, those who slept 5-6 hours per night were at 2.36-fold increased risk of all-cause mortality, while hypertensives in the bottom quartile for sleep duration with 5 hours or less of sleep had an even more robust 4.04-fold increased risk. All risk figures were determined in a multivariate logistic regression analysis extensively adjusted for age, gender, race, diabetes, obesity, smoking, depression, insomnia, sleep apnea, and history of heart disease or stroke.
This finding of an inverse association between sleep duration and all-cause mortality was consistent with the investigators’ study hypothesis that short sleep duration in hypertensive patients may be a marker of the severity of autonomic dysfunction. After all, it is known that the autonomic nervous system not only controls cardiovascular function, it also regulates sleep, explained Dr. Fernandez-Mendoza, a behavioral psychologist at Pennsylvania State University in Hershey.
Other possible explanations for the findings are that short sleep duration in hypertensive patients might be genetically driven or behaviorally induced, but he considers these less plausible.
In an interview, Dr. Fernandez-Mendoza said he and his coinvestigators have found the same relationship between short sleep duration and increased all-cause mortality in Penn State Adult Cohort members with diabetes or dyslipidemia, although he didn’t present those data at the AHA meeting.
If indeed short sleep duration is a marker of autonomic dysfunction, it would have important clinical implications: “Objective sleep duration may allow for refinement of estimates of mortality risk. I predict that someday cardiovascular risk calculators will incorporate sleep duration,” he said.
The Penn State Adult Cohort findings bring a measure of clarity to what has been a somewhat cloudy area, Dr. Fernandez-Mendoza said. Most prior epidemiologic studies of sleep’s impact on health have relied upon self-reported sleep duration, which is considerably less reliable than objectively measured sleep lab data. And many studies have looked at sleep duration as an isolated variable in relation to morbidity and mortality risk. This, he said, has contributed to public misunderstanding.
“We have people coming into the sleep lab thinking, ‘If I don’t get 7 hours of sleep I’m going to die,’ ” according to the sleep scientist. “But the paradigm we’ve developed, tied to what we know about autonomic control, is that the cardiovascular system and the sleep system are connected to each other. It doesn’t mean that short sleep kills you, it’s that the combination of the traditional cardiometabolic risk factors and short sleep increases risk of morbidity and mortality.”
Dr. Fernandez-Mendoza’s study was funded by an AHA Scientist Development Grant. He reported having no financial conflicts of interest.
ORLANDO – Hypertensive persons who sleep 5 hours or less per night have a significantly higher all-cause mortality rate than those who get more shut-eye, according to an analysis from the Penn State Adult Cohort Study.
“We found that the odds of all-cause mortality associated with hypertension increased in a dose-response manner as a function of the degree of objective short sleep duration, even after adjusting for a multitude of factors,” Julio Fernandez-Mendoza, Ph.D., reported at the American Heart Association scientific sessions.
The Penn State Adult Cohort consists of a random, general population sample of 1,741 men and women who enrolled in the study back in the 1990s, at a mean age of 48.7 years. As part of their comprehensive evaluation they were studied in the overnight sleep laboratory. The cohort has been followed for 15.5 years, during which 20% of subjects died.
As expected, hypertension was associated with increased risk of all-cause mortality in the Penn State Adult Cohort. But Dr. Fernandez-Mendoza and coinvestigators further dissected this association by incorporating the subjects’ objective sleep lab data, something that hadn’t been done in other studies. They found that while as a group the roughly 35% of study participants with hypertension had an adjusted 2.54-fold increased risk of all-cause mortality, compared with normotensive subjects, those who slept 6 or more hours at night – placing them at or above the 50th percentile for sleep duration – had a 1.75-fold increased risk, which just barely reached statistical significance.
In contrast, those who slept 5-6 hours per night were at 2.36-fold increased risk of all-cause mortality, while hypertensives in the bottom quartile for sleep duration with 5 hours or less of sleep had an even more robust 4.04-fold increased risk. All risk figures were determined in a multivariate logistic regression analysis extensively adjusted for age, gender, race, diabetes, obesity, smoking, depression, insomnia, sleep apnea, and history of heart disease or stroke.
This finding of an inverse association between sleep duration and all-cause mortality was consistent with the investigators’ study hypothesis that short sleep duration in hypertensive patients may be a marker of the severity of autonomic dysfunction. After all, it is known that the autonomic nervous system not only controls cardiovascular function, it also regulates sleep, explained Dr. Fernandez-Mendoza, a behavioral psychologist at Pennsylvania State University in Hershey.
Other possible explanations for the findings are that short sleep duration in hypertensive patients might be genetically driven or behaviorally induced, but he considers these less plausible.
In an interview, Dr. Fernandez-Mendoza said he and his coinvestigators have found the same relationship between short sleep duration and increased all-cause mortality in Penn State Adult Cohort members with diabetes or dyslipidemia, although he didn’t present those data at the AHA meeting.
If indeed short sleep duration is a marker of autonomic dysfunction, it would have important clinical implications: “Objective sleep duration may allow for refinement of estimates of mortality risk. I predict that someday cardiovascular risk calculators will incorporate sleep duration,” he said.
The Penn State Adult Cohort findings bring a measure of clarity to what has been a somewhat cloudy area, Dr. Fernandez-Mendoza said. Most prior epidemiologic studies of sleep’s impact on health have relied upon self-reported sleep duration, which is considerably less reliable than objectively measured sleep lab data. And many studies have looked at sleep duration as an isolated variable in relation to morbidity and mortality risk. This, he said, has contributed to public misunderstanding.
“We have people coming into the sleep lab thinking, ‘If I don’t get 7 hours of sleep I’m going to die,’ ” according to the sleep scientist. “But the paradigm we’ve developed, tied to what we know about autonomic control, is that the cardiovascular system and the sleep system are connected to each other. It doesn’t mean that short sleep kills you, it’s that the combination of the traditional cardiometabolic risk factors and short sleep increases risk of morbidity and mortality.”
Dr. Fernandez-Mendoza’s study was funded by an AHA Scientist Development Grant. He reported having no financial conflicts of interest.
AT THE AHA SCIENTIFIC SESSIONS
Key clinical point: The shorter a hypertensive patient’s objectively measured sleep duration, the greater the all-cause mortality risk, compared with normotensives.
Major finding: Hypertensive persons with 5 hours of sleep or less were at 4.04-fold increased risk of all-cause mortality, compared with normotensives. Those with a sleep duration of 5-6 hours were at 2.36-fold increased risk, while hypertensives with a sleep duration of 6 hours or more were at 1.75-fold increased risk.
Data source: This study involved 1,741 participants in the Penn State Adult Cohort followed prospectively for 15.5 years.
Disclosures: The presenter’s study was funded by an AHA Scientist Development Grant. He reported having no financial conflicts of interest.
EADV: Vismodegib treatment breaks don’t hurt efficacy
COPENHAGEN – Treatment breaks due to adverse events in patients taking vismodegib for advanced basal cell carcinoma don’t appear to compromise the oral hedgehog pathway inhibitor’s efficacy; in fact, they might even enhance it, according to a prespecified interim analysis of the STEVIE trial.
STEVIE is an ongoing phase II, long-term, open-label international study designed primarily to assess the safety of vismodegib (Erivedge) in a situation similar to routine clinical practice. Efficacy and impact on quality of life are secondary endpoints. Although STEVIE has enrolled 1,227 patients, a prespecified interim analysis was conducted in the first 499 followed for at least 12 months, of whom 468 had locally advanced basal cell carcinoma (BCC) and 31 had metastatic BCC, explained Dr. Johan Hansson, an oncologist at the Karolinska Institute in Stockholm.
The drug was dosed at 150 mg once daily continuously in 28-day cycles until disease progression, intolerable toxicity, or study withdrawal. Safety follow-up was conducted at 1, 3, 5, 9, and 12 months. In an earlier report, the complete and partial response rates were 34% and 33%, respectively, in patients with locally advanced BCC, and 7% and 31% in those with metastatic disease (Lancet Oncol. 2015 Jun;16[6]:729-36).
Dr. Hansson presented new data on efficacy outcomes broken down according to treatment breaks, as well as quality of life results, at the annual congress of the European Academy of Dermatology and Venereology.
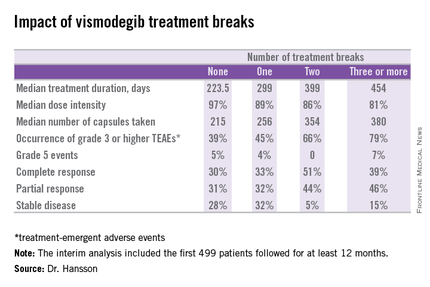
Twenty-six percent of patients had one or more treatment breaks. Seventy-six patients had one, 41 had two, and 14 had three or more. The median duration of the breaks was 22 days. The two most frequent reasons for treatment breaks were intolerable adverse events in 53% of cases, and lesser adverse events in 23%.
Close to 100% of STEVIE participants had treatment-emergent adverse events. The most common were muscle spasms, alopecia, altered sense of smell, and weight loss.
Although the number of patients with treatment breaks was relatively small, the response rates were higher in patients with more treatment breaks. So was median treatment duration as well as the median number of capsules taken.
Median progression-free survival was 19.8 months in patients with no treatment breaks, was 19.0 months in those with one, and hasn’t yet been reached in patients with two or more breaks.
In interpreting these findings, Dr. Hansson said, “We have to remember that although intriguing, these are tentative results from an exploratory analysis of subgroups in an ongoing study and should be interpreted with caution.”
The oncologist added, however, based upon these promising results he and his coinvestigators plan to look further into the concept of deliberate intermittent dosing of vismodegib.
Quality of life was assessed using the Skindex-16 questionnaire at baseline, again after two and seven 28-day cycles of vismodegib, and at 12 months. Three domains were examined: emotion, function, and symptoms.
A clinically meaningful improvement – defined as a 10-point or greater reduction from baseline – was seen in the emotion domain at all time points in patients with locally advanced BCC, with median improvements of 14.3 points after two cycles and 23.8 points after seven cycles and at the 12-month mark. Clinically meaningful improvement in symptom scores on the Skindex-16 were noted in patients aged 65 and older, in women, and in those with BCCs in locations other than the head or neck. However, no clinically meaningful improvement in the domain of function was seen at any time in patients with locally advanced BCC.
Patients with metastatic BCC didn’t show significant improvement in any of the three quality of life domains at any time point, added Dr. Hansson.
The STEVIE trial is sponsored by F. Hoffmann–La Roche/Genentech. Dr. Hansson reported receiving research grants from and serving as a consultant to Bristol-Myers Squibb, GlaxoSmithKline, Merck, Novartis, and Roche.
COPENHAGEN – Treatment breaks due to adverse events in patients taking vismodegib for advanced basal cell carcinoma don’t appear to compromise the oral hedgehog pathway inhibitor’s efficacy; in fact, they might even enhance it, according to a prespecified interim analysis of the STEVIE trial.
STEVIE is an ongoing phase II, long-term, open-label international study designed primarily to assess the safety of vismodegib (Erivedge) in a situation similar to routine clinical practice. Efficacy and impact on quality of life are secondary endpoints. Although STEVIE has enrolled 1,227 patients, a prespecified interim analysis was conducted in the first 499 followed for at least 12 months, of whom 468 had locally advanced basal cell carcinoma (BCC) and 31 had metastatic BCC, explained Dr. Johan Hansson, an oncologist at the Karolinska Institute in Stockholm.
The drug was dosed at 150 mg once daily continuously in 28-day cycles until disease progression, intolerable toxicity, or study withdrawal. Safety follow-up was conducted at 1, 3, 5, 9, and 12 months. In an earlier report, the complete and partial response rates were 34% and 33%, respectively, in patients with locally advanced BCC, and 7% and 31% in those with metastatic disease (Lancet Oncol. 2015 Jun;16[6]:729-36).
Dr. Hansson presented new data on efficacy outcomes broken down according to treatment breaks, as well as quality of life results, at the annual congress of the European Academy of Dermatology and Venereology.
Twenty-six percent of patients had one or more treatment breaks. Seventy-six patients had one, 41 had two, and 14 had three or more. The median duration of the breaks was 22 days. The two most frequent reasons for treatment breaks were intolerable adverse events in 53% of cases, and lesser adverse events in 23%.
Close to 100% of STEVIE participants had treatment-emergent adverse events. The most common were muscle spasms, alopecia, altered sense of smell, and weight loss.
Although the number of patients with treatment breaks was relatively small, the response rates were higher in patients with more treatment breaks. So was median treatment duration as well as the median number of capsules taken.
Median progression-free survival was 19.8 months in patients with no treatment breaks, was 19.0 months in those with one, and hasn’t yet been reached in patients with two or more breaks.
In interpreting these findings, Dr. Hansson said, “We have to remember that although intriguing, these are tentative results from an exploratory analysis of subgroups in an ongoing study and should be interpreted with caution.”
The oncologist added, however, based upon these promising results he and his coinvestigators plan to look further into the concept of deliberate intermittent dosing of vismodegib.
Quality of life was assessed using the Skindex-16 questionnaire at baseline, again after two and seven 28-day cycles of vismodegib, and at 12 months. Three domains were examined: emotion, function, and symptoms.
A clinically meaningful improvement – defined as a 10-point or greater reduction from baseline – was seen in the emotion domain at all time points in patients with locally advanced BCC, with median improvements of 14.3 points after two cycles and 23.8 points after seven cycles and at the 12-month mark. Clinically meaningful improvement in symptom scores on the Skindex-16 were noted in patients aged 65 and older, in women, and in those with BCCs in locations other than the head or neck. However, no clinically meaningful improvement in the domain of function was seen at any time in patients with locally advanced BCC.
Patients with metastatic BCC didn’t show significant improvement in any of the three quality of life domains at any time point, added Dr. Hansson.
The STEVIE trial is sponsored by F. Hoffmann–La Roche/Genentech. Dr. Hansson reported receiving research grants from and serving as a consultant to Bristol-Myers Squibb, GlaxoSmithKline, Merck, Novartis, and Roche.

COPENHAGEN – Treatment breaks due to adverse events in patients taking vismodegib for advanced basal cell carcinoma don’t appear to compromise the oral hedgehog pathway inhibitor’s efficacy; in fact, they might even enhance it, according to a prespecified interim analysis of the STEVIE trial.
STEVIE is an ongoing phase II, long-term, open-label international study designed primarily to assess the safety of vismodegib (Erivedge) in a situation similar to routine clinical practice. Efficacy and impact on quality of life are secondary endpoints. Although STEVIE has enrolled 1,227 patients, a prespecified interim analysis was conducted in the first 499 followed for at least 12 months, of whom 468 had locally advanced basal cell carcinoma (BCC) and 31 had metastatic BCC, explained Dr. Johan Hansson, an oncologist at the Karolinska Institute in Stockholm.
The drug was dosed at 150 mg once daily continuously in 28-day cycles until disease progression, intolerable toxicity, or study withdrawal. Safety follow-up was conducted at 1, 3, 5, 9, and 12 months. In an earlier report, the complete and partial response rates were 34% and 33%, respectively, in patients with locally advanced BCC, and 7% and 31% in those with metastatic disease (Lancet Oncol. 2015 Jun;16[6]:729-36).
Dr. Hansson presented new data on efficacy outcomes broken down according to treatment breaks, as well as quality of life results, at the annual congress of the European Academy of Dermatology and Venereology.
Twenty-six percent of patients had one or more treatment breaks. Seventy-six patients had one, 41 had two, and 14 had three or more. The median duration of the breaks was 22 days. The two most frequent reasons for treatment breaks were intolerable adverse events in 53% of cases, and lesser adverse events in 23%.
Close to 100% of STEVIE participants had treatment-emergent adverse events. The most common were muscle spasms, alopecia, altered sense of smell, and weight loss.
Although the number of patients with treatment breaks was relatively small, the response rates were higher in patients with more treatment breaks. So was median treatment duration as well as the median number of capsules taken.
Median progression-free survival was 19.8 months in patients with no treatment breaks, was 19.0 months in those with one, and hasn’t yet been reached in patients with two or more breaks.
In interpreting these findings, Dr. Hansson said, “We have to remember that although intriguing, these are tentative results from an exploratory analysis of subgroups in an ongoing study and should be interpreted with caution.”
The oncologist added, however, based upon these promising results he and his coinvestigators plan to look further into the concept of deliberate intermittent dosing of vismodegib.
Quality of life was assessed using the Skindex-16 questionnaire at baseline, again after two and seven 28-day cycles of vismodegib, and at 12 months. Three domains were examined: emotion, function, and symptoms.
A clinically meaningful improvement – defined as a 10-point or greater reduction from baseline – was seen in the emotion domain at all time points in patients with locally advanced BCC, with median improvements of 14.3 points after two cycles and 23.8 points after seven cycles and at the 12-month mark. Clinically meaningful improvement in symptom scores on the Skindex-16 were noted in patients aged 65 and older, in women, and in those with BCCs in locations other than the head or neck. However, no clinically meaningful improvement in the domain of function was seen at any time in patients with locally advanced BCC.
Patients with metastatic BCC didn’t show significant improvement in any of the three quality of life domains at any time point, added Dr. Hansson.
The STEVIE trial is sponsored by F. Hoffmann–La Roche/Genentech. Dr. Hansson reported receiving research grants from and serving as a consultant to Bristol-Myers Squibb, GlaxoSmithKline, Merck, Novartis, and Roche.

AT THE EADV CONGRESS
Key clinical point: Treatment breaks due to adverse events in patients taking vismodegib for advanced basal cell carcinoma don’t compromise efficacy.
Major finding: The complete response rate to vismodegib in patients with advanced BCC was intriguingly higher in those with more treatment breaks due to adverse events.
Data source: A prespecified interim analysis of the first 499 patients with advanced BCC enrolled in STEVIE, a large ongoing phase II, long-term, open-label international safety study of vismodegib.
Disclosures: The STEVIE trial is sponsored by F. Hoffmann–La Roche/Genentech. The presenter reported receiving research grants from and serving as a consultant to Bristol-Myers Squibb, GlaxoSmithKline, Merck, Novartis, and Roche.
Family History of Cardiovascular Disease Is Key in Psoriasis Patients
COPENHAGEN – The increased risk of MI and stroke in patients who develop psoriasis as young adults is essentially confined to those having a positive family history of cardiovascular disease, according to a Danish national study presented at the annual congress of the European Academy of Dermatology and Venereology.
“We found a significantly increased risk of MACE [major adverse cardiovascular events] in patients with psoriasis only when a family history of cardiovascular disease was present. This just highlights why it’s important that future studies of cardiovascular risk in psoriasis should include family history. Also, an increased focus on cardiovascular disease in relatives may be appropriate in the cardiovascular risk assessment of patients with psoriasis,” said Dr. Alexander Egeberg of the University of Copenhagen.
He presented a population-based study involving 15 years of follow-up of 30,278 Danes diagnosed with psoriasis in their 20s and a control group consisting of nearly 2.7 million of their Danish contemporaries who were not. None had personal history of acute MI or stroke at baseline. Family medical history, including whether cardiovascular disease occurred in first-degree relatives, was available for all subjects.
Dr. Egeberg and coinvestigators mapped the incidence of acute MI, ischemic stroke, or cardiovascular death in psoriasis patients and the general population controls during follow-up.
“When you look at the patients who developed psoriasis and didn’t have a positive family history of cardiovascular disease, there are almost no cardiovascular events for the entire country,” Dr. Egeberg observed.
In contrast, in a multivariate analysis adjusted for age, gender, socioeconomic status, comorbid cardiovascular disease, smoking, and the use of cardiovascular medications, patients with mild psoriasis and a positive family history for cardiovascular disease had a 28% greater risk of a premature cardiovascular event than the general population during follow-up out to roughly age 40. Those with a positive family history and severe psoriasis as defined by the use of systemic therapies had a 62% increase in risk. Both of these elevated risks were statistically significant.
Among young adult Danes with a positive family history for cardiovascular disease, there were 222 MACE events during 62,225 person-years of follow-up in the mild psoriasis group and 31 events during 6,848 person-years in the 4,504 subjects with severe psoriasis. The resultant incidence rates in both groups were significantly higher than in the control group, who experienced 28,846 MACE events during 16.1 million person-years of follow-up.
In contrast, fewer than 10 MACE events occurred in Danish psoriasis patients without a family history of cardiovascular disease.
A positive family history was also associated with increased MACE in the nonpsoriatic general population, although it didn’t confer as great a risk as in the Danes with psoriasis.
A point worthy of consideration, Dr. Egeberg noted, is that the epidemiology of psoriasis in Denmark apparently differs in several important ways from psoriasis in the United States and some other countries. For one, the prevalence is higher in Scandinavian countries – 7.1% in a Danish national cross-sectional study (Int J Dermatol. 2013 Jun;52[6]:681-3) and 8% in neighboring Norway – as compared with 2%-3% in much of the rest of the world.
Moreover, according to the same cross-sectional study, the prevalence of traditional cardiovascular risk factors, such as smoking and the components of the metabolic syndrome, isn’t higher in Danish psoriasis patients than in the country’s general population. That’s in contrast to the situation in the United Kingdom, where Dr. Joel M. Gelfand of the University of Pennsylvania and associates reported a decade ago in a landmark study that the prevalence of hypertension, obesity, hyperlipidemia, diabetes, and smoking were all higher in persons with psoriasis than in the general population (J Am Acad Dermatol. 2006 Nov;55[5]:829-35). Similar findings were subsequently reported in U.S. psoriasis patients.
Despite their absence of elevated levels of the standard cardiovascular risk factors, Danish psoriasis patients as a group do face a clinically significant increase in cardiovascular risk, compared with the general population, as shown in yet another Danish national cohort study in which the rate ratios for cardiovascular death for mild and severe psoriasis were 1.14 and 1.57, respectively, compared with controls (J Intern Med. 2011 Aug;270[2]:147-57).
In an even more recent Danish nationwide study, the overall death rate was found to be 25.4 per 1,000 person-years in patients with severe psoriasis, 17.0 in those with mild psoriasis, and 13.8 per 1,000 person-years in the general population (J Eur Acad Dermatol Venereol. 2015 May;29[5]:1002-5).
Dr. Egeberg said his new Danish findings suggest that even in psoriasis patients with a greater burden of systemic inflammation as expressed in severe disease, that burden alone doesn’t translate into increased cardiovascular risk. Rather, elevated cardiovascular risk appears to be a consequence of heritable factors, Dr. Egeberg said.
An important caveat regarding this study, he continued, is that the mean age at which participants were diagnosed with psoriasis was 26.6 years. It’s unclear whether the study findings extend to individuals who develop the dermatologic disease later in life.
Dr. Egeberg reported having no financial conflicts regarding this study, supported by Danish national research funding.
COPENHAGEN – The increased risk of MI and stroke in patients who develop psoriasis as young adults is essentially confined to those having a positive family history of cardiovascular disease, according to a Danish national study presented at the annual congress of the European Academy of Dermatology and Venereology.
“We found a significantly increased risk of MACE [major adverse cardiovascular events] in patients with psoriasis only when a family history of cardiovascular disease was present. This just highlights why it’s important that future studies of cardiovascular risk in psoriasis should include family history. Also, an increased focus on cardiovascular disease in relatives may be appropriate in the cardiovascular risk assessment of patients with psoriasis,” said Dr. Alexander Egeberg of the University of Copenhagen.
He presented a population-based study involving 15 years of follow-up of 30,278 Danes diagnosed with psoriasis in their 20s and a control group consisting of nearly 2.7 million of their Danish contemporaries who were not. None had personal history of acute MI or stroke at baseline. Family medical history, including whether cardiovascular disease occurred in first-degree relatives, was available for all subjects.
Dr. Egeberg and coinvestigators mapped the incidence of acute MI, ischemic stroke, or cardiovascular death in psoriasis patients and the general population controls during follow-up.
“When you look at the patients who developed psoriasis and didn’t have a positive family history of cardiovascular disease, there are almost no cardiovascular events for the entire country,” Dr. Egeberg observed.
In contrast, in a multivariate analysis adjusted for age, gender, socioeconomic status, comorbid cardiovascular disease, smoking, and the use of cardiovascular medications, patients with mild psoriasis and a positive family history for cardiovascular disease had a 28% greater risk of a premature cardiovascular event than the general population during follow-up out to roughly age 40. Those with a positive family history and severe psoriasis as defined by the use of systemic therapies had a 62% increase in risk. Both of these elevated risks were statistically significant.
Among young adult Danes with a positive family history for cardiovascular disease, there were 222 MACE events during 62,225 person-years of follow-up in the mild psoriasis group and 31 events during 6,848 person-years in the 4,504 subjects with severe psoriasis. The resultant incidence rates in both groups were significantly higher than in the control group, who experienced 28,846 MACE events during 16.1 million person-years of follow-up.
In contrast, fewer than 10 MACE events occurred in Danish psoriasis patients without a family history of cardiovascular disease.
A positive family history was also associated with increased MACE in the nonpsoriatic general population, although it didn’t confer as great a risk as in the Danes with psoriasis.
A point worthy of consideration, Dr. Egeberg noted, is that the epidemiology of psoriasis in Denmark apparently differs in several important ways from psoriasis in the United States and some other countries. For one, the prevalence is higher in Scandinavian countries – 7.1% in a Danish national cross-sectional study (Int J Dermatol. 2013 Jun;52[6]:681-3) and 8% in neighboring Norway – as compared with 2%-3% in much of the rest of the world.
Moreover, according to the same cross-sectional study, the prevalence of traditional cardiovascular risk factors, such as smoking and the components of the metabolic syndrome, isn’t higher in Danish psoriasis patients than in the country’s general population. That’s in contrast to the situation in the United Kingdom, where Dr. Joel M. Gelfand of the University of Pennsylvania and associates reported a decade ago in a landmark study that the prevalence of hypertension, obesity, hyperlipidemia, diabetes, and smoking were all higher in persons with psoriasis than in the general population (J Am Acad Dermatol. 2006 Nov;55[5]:829-35). Similar findings were subsequently reported in U.S. psoriasis patients.
Despite their absence of elevated levels of the standard cardiovascular risk factors, Danish psoriasis patients as a group do face a clinically significant increase in cardiovascular risk, compared with the general population, as shown in yet another Danish national cohort study in which the rate ratios for cardiovascular death for mild and severe psoriasis were 1.14 and 1.57, respectively, compared with controls (J Intern Med. 2011 Aug;270[2]:147-57).
In an even more recent Danish nationwide study, the overall death rate was found to be 25.4 per 1,000 person-years in patients with severe psoriasis, 17.0 in those with mild psoriasis, and 13.8 per 1,000 person-years in the general population (J Eur Acad Dermatol Venereol. 2015 May;29[5]:1002-5).
Dr. Egeberg said his new Danish findings suggest that even in psoriasis patients with a greater burden of systemic inflammation as expressed in severe disease, that burden alone doesn’t translate into increased cardiovascular risk. Rather, elevated cardiovascular risk appears to be a consequence of heritable factors, Dr. Egeberg said.
An important caveat regarding this study, he continued, is that the mean age at which participants were diagnosed with psoriasis was 26.6 years. It’s unclear whether the study findings extend to individuals who develop the dermatologic disease later in life.
Dr. Egeberg reported having no financial conflicts regarding this study, supported by Danish national research funding.
COPENHAGEN – The increased risk of MI and stroke in patients who develop psoriasis as young adults is essentially confined to those having a positive family history of cardiovascular disease, according to a Danish national study presented at the annual congress of the European Academy of Dermatology and Venereology.
“We found a significantly increased risk of MACE [major adverse cardiovascular events] in patients with psoriasis only when a family history of cardiovascular disease was present. This just highlights why it’s important that future studies of cardiovascular risk in psoriasis should include family history. Also, an increased focus on cardiovascular disease in relatives may be appropriate in the cardiovascular risk assessment of patients with psoriasis,” said Dr. Alexander Egeberg of the University of Copenhagen.
He presented a population-based study involving 15 years of follow-up of 30,278 Danes diagnosed with psoriasis in their 20s and a control group consisting of nearly 2.7 million of their Danish contemporaries who were not. None had personal history of acute MI or stroke at baseline. Family medical history, including whether cardiovascular disease occurred in first-degree relatives, was available for all subjects.
Dr. Egeberg and coinvestigators mapped the incidence of acute MI, ischemic stroke, or cardiovascular death in psoriasis patients and the general population controls during follow-up.
“When you look at the patients who developed psoriasis and didn’t have a positive family history of cardiovascular disease, there are almost no cardiovascular events for the entire country,” Dr. Egeberg observed.
In contrast, in a multivariate analysis adjusted for age, gender, socioeconomic status, comorbid cardiovascular disease, smoking, and the use of cardiovascular medications, patients with mild psoriasis and a positive family history for cardiovascular disease had a 28% greater risk of a premature cardiovascular event than the general population during follow-up out to roughly age 40. Those with a positive family history and severe psoriasis as defined by the use of systemic therapies had a 62% increase in risk. Both of these elevated risks were statistically significant.
Among young adult Danes with a positive family history for cardiovascular disease, there were 222 MACE events during 62,225 person-years of follow-up in the mild psoriasis group and 31 events during 6,848 person-years in the 4,504 subjects with severe psoriasis. The resultant incidence rates in both groups were significantly higher than in the control group, who experienced 28,846 MACE events during 16.1 million person-years of follow-up.
In contrast, fewer than 10 MACE events occurred in Danish psoriasis patients without a family history of cardiovascular disease.
A positive family history was also associated with increased MACE in the nonpsoriatic general population, although it didn’t confer as great a risk as in the Danes with psoriasis.
A point worthy of consideration, Dr. Egeberg noted, is that the epidemiology of psoriasis in Denmark apparently differs in several important ways from psoriasis in the United States and some other countries. For one, the prevalence is higher in Scandinavian countries – 7.1% in a Danish national cross-sectional study (Int J Dermatol. 2013 Jun;52[6]:681-3) and 8% in neighboring Norway – as compared with 2%-3% in much of the rest of the world.
Moreover, according to the same cross-sectional study, the prevalence of traditional cardiovascular risk factors, such as smoking and the components of the metabolic syndrome, isn’t higher in Danish psoriasis patients than in the country’s general population. That’s in contrast to the situation in the United Kingdom, where Dr. Joel M. Gelfand of the University of Pennsylvania and associates reported a decade ago in a landmark study that the prevalence of hypertension, obesity, hyperlipidemia, diabetes, and smoking were all higher in persons with psoriasis than in the general population (J Am Acad Dermatol. 2006 Nov;55[5]:829-35). Similar findings were subsequently reported in U.S. psoriasis patients.
Despite their absence of elevated levels of the standard cardiovascular risk factors, Danish psoriasis patients as a group do face a clinically significant increase in cardiovascular risk, compared with the general population, as shown in yet another Danish national cohort study in which the rate ratios for cardiovascular death for mild and severe psoriasis were 1.14 and 1.57, respectively, compared with controls (J Intern Med. 2011 Aug;270[2]:147-57).
In an even more recent Danish nationwide study, the overall death rate was found to be 25.4 per 1,000 person-years in patients with severe psoriasis, 17.0 in those with mild psoriasis, and 13.8 per 1,000 person-years in the general population (J Eur Acad Dermatol Venereol. 2015 May;29[5]:1002-5).
Dr. Egeberg said his new Danish findings suggest that even in psoriasis patients with a greater burden of systemic inflammation as expressed in severe disease, that burden alone doesn’t translate into increased cardiovascular risk. Rather, elevated cardiovascular risk appears to be a consequence of heritable factors, Dr. Egeberg said.
An important caveat regarding this study, he continued, is that the mean age at which participants were diagnosed with psoriasis was 26.6 years. It’s unclear whether the study findings extend to individuals who develop the dermatologic disease later in life.
Dr. Egeberg reported having no financial conflicts regarding this study, supported by Danish national research funding.
AT THE EADV CONGRESS
EADV: Family history of cardiovascular disease is key in psoriasis patients
COPENHAGEN – The increased risk of MI and stroke in patients who develop psoriasis as young adults is essentially confined to those having a positive family history of cardiovascular disease, according to a Danish national study presented at the annual congress of the European Academy of Dermatology and Venereology.
“We found a significantly increased risk of MACE [major adverse cardiovascular events] in patients with psoriasis only when a family history of cardiovascular disease was present. This just highlights why it’s important that future studies of cardiovascular risk in psoriasis should include family history. Also, an increased focus on cardiovascular disease in relatives may be appropriate in the cardiovascular risk assessment of patients with psoriasis,” said Dr. Alexander Egeberg of the University of Copenhagen.
He presented a population-based study involving 15 years of follow-up of 30,278 Danes diagnosed with psoriasis in their 20s and a control group consisting of nearly 2.7 million of their Danish contemporaries who were not. None had personal history of acute MI or stroke at baseline. Family medical history, including whether cardiovascular disease occurred in first-degree relatives, was available for all subjects.
Dr. Egeberg and coinvestigators mapped the incidence of acute MI, ischemic stroke, or cardiovascular death in psoriasis patients and the general population controls during follow-up.
“When you look at the patients who developed psoriasis and didn’t have a positive family history of cardiovascular disease, there are almost no cardiovascular events for the entire country,” Dr. Egeberg observed.
In contrast, in a multivariate analysis adjusted for age, gender, socioeconomic status, comorbid cardiovascular disease, smoking, and the use of cardiovascular medications, patients with mild psoriasis and a positive family history for cardiovascular disease had a 28% greater risk of a premature cardiovascular event than the general population during follow-up out to roughly age 40. Those with a positive family history and severe psoriasis as defined by the use of systemic therapies had a 62% increase in risk. Both of these elevated risks were statistically significant.
Among young adult Danes with a positive family history for cardiovascular disease, there were 222 MACE events during 62,225 person-years of follow-up in the mild psoriasis group and 31 events during 6,848 person-years in the 4,504 subjects with severe psoriasis. The resultant incidence rates in both groups were significantly higher than in the control group, who experienced 28,846 MACE events during 16.1 million person-years of follow-up.
In contrast, fewer than 10 MACE events occurred in Danish psoriasis patients without a family history of cardiovascular disease.
A positive family history was also associated with increased MACE in the nonpsoriatic general population, although it didn’t confer as great a risk as in the Danes with psoriasis.
A point worthy of consideration, Dr. Egeberg noted, is that the epidemiology of psoriasis in Denmark apparently differs in several important ways from psoriasis in the United States and some other countries. For one, the prevalence is higher in Scandinavian countries – 7.1% in a Danish national cross-sectional study (Int J Dermatol. 2013 Jun;52[6]:681-3) and 8% in neighboring Norway – as compared with 2%-3% in much of the rest of the world.
Moreover, according to the same cross-sectional study, the prevalence of traditional cardiovascular risk factors, such as smoking and the components of the metabolic syndrome, isn’t higher in Danish psoriasis patients than in the country’s general population. That’s in contrast to the situation in the United Kingdom, where Dr. Joel M. Gelfand of the University of Pennsylvania and associates reported a decade ago in a landmark study that the prevalence of hypertension, obesity, hyperlipidemia, diabetes, and smoking were all higher in persons with psoriasis than in the general population (J Am Acad Dermatol. 2006 Nov;55[5]:829-35). Similar findings were subsequently reported in U.S. psoriasis patients.
Despite their absence of elevated levels of the standard cardiovascular risk factors, Danish psoriasis patients as a group do face a clinically significant increase in cardiovascular risk, compared with the general population, as shown in yet another Danish national cohort study in which the rate ratios for cardiovascular death for mild and severe psoriasis were 1.14 and 1.57, respectively, compared with controls (J Intern Med. 2011 Aug;270[2]:147-57).
In an even more recent Danish nationwide study, the overall death rate was found to be 25.4 per 1,000 person-years in patients with severe psoriasis, 17.0 in those with mild psoriasis, and 13.8 per 1,000 person-years in the general population (J Eur Acad Dermatol Venereol. 2015 May;29[5]:1002-5).
Dr. Egeberg said his new Danish findings suggest that even in psoriasis patients with a greater burden of systemic inflammation as expressed in severe disease, that burden alone doesn’t translate into increased cardiovascular risk. Rather, elevated cardiovascular risk appears to be a consequence of heritable factors, Dr. Egeberg said.
An important caveat regarding this study, he continued, is that the mean age at which participants were diagnosed with psoriasis was 26.6 years. It’s unclear whether the study findings extend to individuals who develop the dermatologic disease later in life.
Dr. Egeberg reported having no financial conflicts regarding this study, supported by Danish national research funding.
COPENHAGEN – The increased risk of MI and stroke in patients who develop psoriasis as young adults is essentially confined to those having a positive family history of cardiovascular disease, according to a Danish national study presented at the annual congress of the European Academy of Dermatology and Venereology.
“We found a significantly increased risk of MACE [major adverse cardiovascular events] in patients with psoriasis only when a family history of cardiovascular disease was present. This just highlights why it’s important that future studies of cardiovascular risk in psoriasis should include family history. Also, an increased focus on cardiovascular disease in relatives may be appropriate in the cardiovascular risk assessment of patients with psoriasis,” said Dr. Alexander Egeberg of the University of Copenhagen.
He presented a population-based study involving 15 years of follow-up of 30,278 Danes diagnosed with psoriasis in their 20s and a control group consisting of nearly 2.7 million of their Danish contemporaries who were not. None had personal history of acute MI or stroke at baseline. Family medical history, including whether cardiovascular disease occurred in first-degree relatives, was available for all subjects.
Dr. Egeberg and coinvestigators mapped the incidence of acute MI, ischemic stroke, or cardiovascular death in psoriasis patients and the general population controls during follow-up.
“When you look at the patients who developed psoriasis and didn’t have a positive family history of cardiovascular disease, there are almost no cardiovascular events for the entire country,” Dr. Egeberg observed.
In contrast, in a multivariate analysis adjusted for age, gender, socioeconomic status, comorbid cardiovascular disease, smoking, and the use of cardiovascular medications, patients with mild psoriasis and a positive family history for cardiovascular disease had a 28% greater risk of a premature cardiovascular event than the general population during follow-up out to roughly age 40. Those with a positive family history and severe psoriasis as defined by the use of systemic therapies had a 62% increase in risk. Both of these elevated risks were statistically significant.
Among young adult Danes with a positive family history for cardiovascular disease, there were 222 MACE events during 62,225 person-years of follow-up in the mild psoriasis group and 31 events during 6,848 person-years in the 4,504 subjects with severe psoriasis. The resultant incidence rates in both groups were significantly higher than in the control group, who experienced 28,846 MACE events during 16.1 million person-years of follow-up.
In contrast, fewer than 10 MACE events occurred in Danish psoriasis patients without a family history of cardiovascular disease.
A positive family history was also associated with increased MACE in the nonpsoriatic general population, although it didn’t confer as great a risk as in the Danes with psoriasis.
A point worthy of consideration, Dr. Egeberg noted, is that the epidemiology of psoriasis in Denmark apparently differs in several important ways from psoriasis in the United States and some other countries. For one, the prevalence is higher in Scandinavian countries – 7.1% in a Danish national cross-sectional study (Int J Dermatol. 2013 Jun;52[6]:681-3) and 8% in neighboring Norway – as compared with 2%-3% in much of the rest of the world.
Moreover, according to the same cross-sectional study, the prevalence of traditional cardiovascular risk factors, such as smoking and the components of the metabolic syndrome, isn’t higher in Danish psoriasis patients than in the country’s general population. That’s in contrast to the situation in the United Kingdom, where Dr. Joel M. Gelfand of the University of Pennsylvania and associates reported a decade ago in a landmark study that the prevalence of hypertension, obesity, hyperlipidemia, diabetes, and smoking were all higher in persons with psoriasis than in the general population (J Am Acad Dermatol. 2006 Nov;55[5]:829-35). Similar findings were subsequently reported in U.S. psoriasis patients.
Despite their absence of elevated levels of the standard cardiovascular risk factors, Danish psoriasis patients as a group do face a clinically significant increase in cardiovascular risk, compared with the general population, as shown in yet another Danish national cohort study in which the rate ratios for cardiovascular death for mild and severe psoriasis were 1.14 and 1.57, respectively, compared with controls (J Intern Med. 2011 Aug;270[2]:147-57).
In an even more recent Danish nationwide study, the overall death rate was found to be 25.4 per 1,000 person-years in patients with severe psoriasis, 17.0 in those with mild psoriasis, and 13.8 per 1,000 person-years in the general population (J Eur Acad Dermatol Venereol. 2015 May;29[5]:1002-5).
Dr. Egeberg said his new Danish findings suggest that even in psoriasis patients with a greater burden of systemic inflammation as expressed in severe disease, that burden alone doesn’t translate into increased cardiovascular risk. Rather, elevated cardiovascular risk appears to be a consequence of heritable factors, Dr. Egeberg said.
An important caveat regarding this study, he continued, is that the mean age at which participants were diagnosed with psoriasis was 26.6 years. It’s unclear whether the study findings extend to individuals who develop the dermatologic disease later in life.
Dr. Egeberg reported having no financial conflicts regarding this study, supported by Danish national research funding.
COPENHAGEN – The increased risk of MI and stroke in patients who develop psoriasis as young adults is essentially confined to those having a positive family history of cardiovascular disease, according to a Danish national study presented at the annual congress of the European Academy of Dermatology and Venereology.
“We found a significantly increased risk of MACE [major adverse cardiovascular events] in patients with psoriasis only when a family history of cardiovascular disease was present. This just highlights why it’s important that future studies of cardiovascular risk in psoriasis should include family history. Also, an increased focus on cardiovascular disease in relatives may be appropriate in the cardiovascular risk assessment of patients with psoriasis,” said Dr. Alexander Egeberg of the University of Copenhagen.
He presented a population-based study involving 15 years of follow-up of 30,278 Danes diagnosed with psoriasis in their 20s and a control group consisting of nearly 2.7 million of their Danish contemporaries who were not. None had personal history of acute MI or stroke at baseline. Family medical history, including whether cardiovascular disease occurred in first-degree relatives, was available for all subjects.
Dr. Egeberg and coinvestigators mapped the incidence of acute MI, ischemic stroke, or cardiovascular death in psoriasis patients and the general population controls during follow-up.
“When you look at the patients who developed psoriasis and didn’t have a positive family history of cardiovascular disease, there are almost no cardiovascular events for the entire country,” Dr. Egeberg observed.
In contrast, in a multivariate analysis adjusted for age, gender, socioeconomic status, comorbid cardiovascular disease, smoking, and the use of cardiovascular medications, patients with mild psoriasis and a positive family history for cardiovascular disease had a 28% greater risk of a premature cardiovascular event than the general population during follow-up out to roughly age 40. Those with a positive family history and severe psoriasis as defined by the use of systemic therapies had a 62% increase in risk. Both of these elevated risks were statistically significant.
Among young adult Danes with a positive family history for cardiovascular disease, there were 222 MACE events during 62,225 person-years of follow-up in the mild psoriasis group and 31 events during 6,848 person-years in the 4,504 subjects with severe psoriasis. The resultant incidence rates in both groups were significantly higher than in the control group, who experienced 28,846 MACE events during 16.1 million person-years of follow-up.
In contrast, fewer than 10 MACE events occurred in Danish psoriasis patients without a family history of cardiovascular disease.
A positive family history was also associated with increased MACE in the nonpsoriatic general population, although it didn’t confer as great a risk as in the Danes with psoriasis.
A point worthy of consideration, Dr. Egeberg noted, is that the epidemiology of psoriasis in Denmark apparently differs in several important ways from psoriasis in the United States and some other countries. For one, the prevalence is higher in Scandinavian countries – 7.1% in a Danish national cross-sectional study (Int J Dermatol. 2013 Jun;52[6]:681-3) and 8% in neighboring Norway – as compared with 2%-3% in much of the rest of the world.
Moreover, according to the same cross-sectional study, the prevalence of traditional cardiovascular risk factors, such as smoking and the components of the metabolic syndrome, isn’t higher in Danish psoriasis patients than in the country’s general population. That’s in contrast to the situation in the United Kingdom, where Dr. Joel M. Gelfand of the University of Pennsylvania and associates reported a decade ago in a landmark study that the prevalence of hypertension, obesity, hyperlipidemia, diabetes, and smoking were all higher in persons with psoriasis than in the general population (J Am Acad Dermatol. 2006 Nov;55[5]:829-35). Similar findings were subsequently reported in U.S. psoriasis patients.
Despite their absence of elevated levels of the standard cardiovascular risk factors, Danish psoriasis patients as a group do face a clinically significant increase in cardiovascular risk, compared with the general population, as shown in yet another Danish national cohort study in which the rate ratios for cardiovascular death for mild and severe psoriasis were 1.14 and 1.57, respectively, compared with controls (J Intern Med. 2011 Aug;270[2]:147-57).
In an even more recent Danish nationwide study, the overall death rate was found to be 25.4 per 1,000 person-years in patients with severe psoriasis, 17.0 in those with mild psoriasis, and 13.8 per 1,000 person-years in the general population (J Eur Acad Dermatol Venereol. 2015 May;29[5]:1002-5).
Dr. Egeberg said his new Danish findings suggest that even in psoriasis patients with a greater burden of systemic inflammation as expressed in severe disease, that burden alone doesn’t translate into increased cardiovascular risk. Rather, elevated cardiovascular risk appears to be a consequence of heritable factors, Dr. Egeberg said.
An important caveat regarding this study, he continued, is that the mean age at which participants were diagnosed with psoriasis was 26.6 years. It’s unclear whether the study findings extend to individuals who develop the dermatologic disease later in life.
Dr. Egeberg reported having no financial conflicts regarding this study, supported by Danish national research funding.
AT THE EADV CONGRESS
Key clinical point: A family history of cardiovascular disease takes on extra importance in assessing cardiovascular risk in young adult psoriasis patients.
Major finding: Danes with mild or severe psoriasis plus a family history of cardiovascular disease were respectively 28% and 62% more likely to have an early cardiovascular event than the general population. In contrast, Danish psoriasis patients without a positive family history were not at increased risk of a cardiovascular event.
Data source: A population-based study of 2.7 million Danish young adults, including more than 30,000 diagnosed with psoriasis in their 20s, who were followed for 15 years.
Disclosures: The presenter reported having no financial conflicts regarding this study, which was supported by Danish national research funding.
EADV: New oral psoriasis drug shows excellent safety
COPENHAGEN – An oral small molecule with a novel mechanism of action for treatment of moderate to severe plaque psoriasis is being developed as a potential first-line systemic treatment in view of its highly favorable safety profile.
The investigational drug, known for now as CF101, is a first-in-class agonist of the A3 adenosine receptor. This cell surface receptor is upregulated in the pathologic cells of certain inflammatory diseases, but has little or no expression in normal cells. This high degree of specificity accounts for its safety, which in a recent phase II/III trial was essentially indistinguishable from placebo, making it an attractive potential alternative to methotrexate or biologics as a starting point in systemic therapy, Pnina Fishman, Ph.D., said at the annual congress of the European Academy of Dermatology and Venereology.

Planning is underway for a pivotal phase III trial of CF101 in psoriasis, which is also being organized for CF101 in rheumatoid arthritis on the strength of favorable phase II findings, according to Dr. Fishman, CEO of Can-Fite BioPharma, an Israeli biotech company that is developing the drug.
The phase II/III psoriasis trial was a 326-patient, double-blind, placebo-controlled study. It showed efficacy comparable to and in some respects better than that reported in the phase III ESTEEM-1 study of the oral phosphodiesterase 4 inhibitor apremilast (J Am Acad Dermatol. 2015 Jul;73:37-49).
Moreover, it appears that the twice-daily 2-mg dosing of CF101 studied in the phase II/III trial was suboptimal in light of the observed linear increase in Psoriasis Area and Severity Index (PASI)-75, -90, and -100 response rates over time. Those response rates rose steadily until the study’s end at week 32 with no evidence of a plateau. In contrast, in ESTEEM-1, the improvement with apremilast (Otezla) leveled off starting at about week 20, she observed.
In the recent phase II/III trial, the PASI-75 rate for CF101 at week 12 – the prespecified primary endpoint – was not significantly better than placebo was. However, the PASI-75 response rate continued to climb such that by week 32, it was 35.3%, similar to the 33.1% PASI-75 response seen at 16 weeks in ESTEEM-1. PASI improved by an average of 57% from baseline to week 32 with CF101 with no plateau in sight, and by 55% with apremilast at week 16, with a leveling off at week 20. This is why the upcoming phase III trial will employ a higher dose of CF101 than twice-daily 2-mg dose used in the phase II/III study and will run longer. The goal is to achieve higher PASI response rates faster than obtainable with 2 mg BID, Dr. Fishman explained.
She added that the PASI-90 response data in the phase II/III trial bode particularly well for the future of CF101 as a first-line systemic agent. At week 32, this stringent outcome measure was achieved by 26.9% of participants who hadn’t previously been on methotrexate or a biologic and by 13.7% of those who had. And as was the case for the PASI-75 results, the PASI-90 response increased in linear fashion out to 32 weeks with no plateau. In ESTEEM-1, the PASI-90 rate was 9.8% at week 16.
No treatment-related adverse events were seen in the phase II/III CF101 study, Dr. Fishman reported.
The study was sponsored by Can-Fite BioPharma and presented by Dr. Fishman, who is the company’s CEO.
COPENHAGEN – An oral small molecule with a novel mechanism of action for treatment of moderate to severe plaque psoriasis is being developed as a potential first-line systemic treatment in view of its highly favorable safety profile.
The investigational drug, known for now as CF101, is a first-in-class agonist of the A3 adenosine receptor. This cell surface receptor is upregulated in the pathologic cells of certain inflammatory diseases, but has little or no expression in normal cells. This high degree of specificity accounts for its safety, which in a recent phase II/III trial was essentially indistinguishable from placebo, making it an attractive potential alternative to methotrexate or biologics as a starting point in systemic therapy, Pnina Fishman, Ph.D., said at the annual congress of the European Academy of Dermatology and Venereology.

Planning is underway for a pivotal phase III trial of CF101 in psoriasis, which is also being organized for CF101 in rheumatoid arthritis on the strength of favorable phase II findings, according to Dr. Fishman, CEO of Can-Fite BioPharma, an Israeli biotech company that is developing the drug.
The phase II/III psoriasis trial was a 326-patient, double-blind, placebo-controlled study. It showed efficacy comparable to and in some respects better than that reported in the phase III ESTEEM-1 study of the oral phosphodiesterase 4 inhibitor apremilast (J Am Acad Dermatol. 2015 Jul;73:37-49).
Moreover, it appears that the twice-daily 2-mg dosing of CF101 studied in the phase II/III trial was suboptimal in light of the observed linear increase in Psoriasis Area and Severity Index (PASI)-75, -90, and -100 response rates over time. Those response rates rose steadily until the study’s end at week 32 with no evidence of a plateau. In contrast, in ESTEEM-1, the improvement with apremilast (Otezla) leveled off starting at about week 20, she observed.
In the recent phase II/III trial, the PASI-75 rate for CF101 at week 12 – the prespecified primary endpoint – was not significantly better than placebo was. However, the PASI-75 response rate continued to climb such that by week 32, it was 35.3%, similar to the 33.1% PASI-75 response seen at 16 weeks in ESTEEM-1. PASI improved by an average of 57% from baseline to week 32 with CF101 with no plateau in sight, and by 55% with apremilast at week 16, with a leveling off at week 20. This is why the upcoming phase III trial will employ a higher dose of CF101 than twice-daily 2-mg dose used in the phase II/III study and will run longer. The goal is to achieve higher PASI response rates faster than obtainable with 2 mg BID, Dr. Fishman explained.
She added that the PASI-90 response data in the phase II/III trial bode particularly well for the future of CF101 as a first-line systemic agent. At week 32, this stringent outcome measure was achieved by 26.9% of participants who hadn’t previously been on methotrexate or a biologic and by 13.7% of those who had. And as was the case for the PASI-75 results, the PASI-90 response increased in linear fashion out to 32 weeks with no plateau. In ESTEEM-1, the PASI-90 rate was 9.8% at week 16.
No treatment-related adverse events were seen in the phase II/III CF101 study, Dr. Fishman reported.
The study was sponsored by Can-Fite BioPharma and presented by Dr. Fishman, who is the company’s CEO.
COPENHAGEN – An oral small molecule with a novel mechanism of action for treatment of moderate to severe plaque psoriasis is being developed as a potential first-line systemic treatment in view of its highly favorable safety profile.
The investigational drug, known for now as CF101, is a first-in-class agonist of the A3 adenosine receptor. This cell surface receptor is upregulated in the pathologic cells of certain inflammatory diseases, but has little or no expression in normal cells. This high degree of specificity accounts for its safety, which in a recent phase II/III trial was essentially indistinguishable from placebo, making it an attractive potential alternative to methotrexate or biologics as a starting point in systemic therapy, Pnina Fishman, Ph.D., said at the annual congress of the European Academy of Dermatology and Venereology.

Planning is underway for a pivotal phase III trial of CF101 in psoriasis, which is also being organized for CF101 in rheumatoid arthritis on the strength of favorable phase II findings, according to Dr. Fishman, CEO of Can-Fite BioPharma, an Israeli biotech company that is developing the drug.
The phase II/III psoriasis trial was a 326-patient, double-blind, placebo-controlled study. It showed efficacy comparable to and in some respects better than that reported in the phase III ESTEEM-1 study of the oral phosphodiesterase 4 inhibitor apremilast (J Am Acad Dermatol. 2015 Jul;73:37-49).
Moreover, it appears that the twice-daily 2-mg dosing of CF101 studied in the phase II/III trial was suboptimal in light of the observed linear increase in Psoriasis Area and Severity Index (PASI)-75, -90, and -100 response rates over time. Those response rates rose steadily until the study’s end at week 32 with no evidence of a plateau. In contrast, in ESTEEM-1, the improvement with apremilast (Otezla) leveled off starting at about week 20, she observed.
In the recent phase II/III trial, the PASI-75 rate for CF101 at week 12 – the prespecified primary endpoint – was not significantly better than placebo was. However, the PASI-75 response rate continued to climb such that by week 32, it was 35.3%, similar to the 33.1% PASI-75 response seen at 16 weeks in ESTEEM-1. PASI improved by an average of 57% from baseline to week 32 with CF101 with no plateau in sight, and by 55% with apremilast at week 16, with a leveling off at week 20. This is why the upcoming phase III trial will employ a higher dose of CF101 than twice-daily 2-mg dose used in the phase II/III study and will run longer. The goal is to achieve higher PASI response rates faster than obtainable with 2 mg BID, Dr. Fishman explained.
She added that the PASI-90 response data in the phase II/III trial bode particularly well for the future of CF101 as a first-line systemic agent. At week 32, this stringent outcome measure was achieved by 26.9% of participants who hadn’t previously been on methotrexate or a biologic and by 13.7% of those who had. And as was the case for the PASI-75 results, the PASI-90 response increased in linear fashion out to 32 weeks with no plateau. In ESTEEM-1, the PASI-90 rate was 9.8% at week 16.
No treatment-related adverse events were seen in the phase II/III CF101 study, Dr. Fishman reported.
The study was sponsored by Can-Fite BioPharma and presented by Dr. Fishman, who is the company’s CEO.
AT THE EADV CONGRESS
Key clinical point: The oral A3 adenosine receptor agonist CF1-1 shows promise as a potential first-line systemic therapy for moderate to severe plaque psoriasis.
Major finding: The PASI-75 response rate with the twice-daily 2-mg dose of oral CF101 was 35.3% at 32 weeks in a phase II/III study.
Data source: This was a randomized, double-blind, placebo-controlled, 32-week phase II/III study including 326 patients with moderate-to-severe plaque psoriasis.
Disclosures: The study was sponsored by Can-Fite BioPharma of Israel and presented by the Dr. Fishman, the company’s CEO.
Moderate, Intensive Exercise Cut Triglycerides Equally in NAFLD
ORLANDO – Moderate aerobic exercise and a more intensive regimen proved equally effective in reducing intrahepatic triglyceride content in centrally obese patients with nonalcoholic fatty liver disease in a 12-month randomized, controlled trial.
However, the more intensive exercise program had the edge in terms of favorable impact on metabolic risk factors. While both exercise prescriptions reduced body weight and blood pressure to a similar extent over the course of 12 months, the intensive-exercise group showed bigger reductions in body fat mass, visceral fat, and subcutaneous fat (see graphic), Dr. Hui-Jie Zhang of Xiamen (China) University said at the American Heart Association scientific sessions.
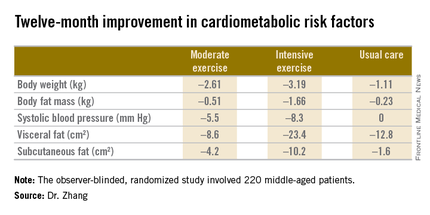
The observer-blinded study included 220 middle-age Chinese patients with central obesity and confirmed nonalcoholic fatty liver disease (NAFLD), a common risk factor for cardiovascular disease. They were randomized to three groups: a moderate exercise program that consisted of brisk walking for 150 minutes per week for 12 months; an intensive exercise regimen involving 30 minutes of treadmill running at 65%-80% of maximum oxygen consumption 5 days per week for 6 months, followed by 6 months of brisk walking on the same schedule as the moderate exercise group; or a usual-care control group that received lifestyle counseling.
The primary endpoint was change in intrahepatic triglyceride content as measured by proton magnetic resonance spectroscopy at 12 months. It was reduced from baseline by 6.45% in the intensive exercise group and similarly by 6.19% with moderate exercise, both significantly greater effects than the 2.85% decrease in the control group.
Dr. Zhang reported having no financial conflicts of interest regarding this study.
ORLANDO – Moderate aerobic exercise and a more intensive regimen proved equally effective in reducing intrahepatic triglyceride content in centrally obese patients with nonalcoholic fatty liver disease in a 12-month randomized, controlled trial.
However, the more intensive exercise program had the edge in terms of favorable impact on metabolic risk factors. While both exercise prescriptions reduced body weight and blood pressure to a similar extent over the course of 12 months, the intensive-exercise group showed bigger reductions in body fat mass, visceral fat, and subcutaneous fat (see graphic), Dr. Hui-Jie Zhang of Xiamen (China) University said at the American Heart Association scientific sessions.

The observer-blinded study included 220 middle-age Chinese patients with central obesity and confirmed nonalcoholic fatty liver disease (NAFLD), a common risk factor for cardiovascular disease. They were randomized to three groups: a moderate exercise program that consisted of brisk walking for 150 minutes per week for 12 months; an intensive exercise regimen involving 30 minutes of treadmill running at 65%-80% of maximum oxygen consumption 5 days per week for 6 months, followed by 6 months of brisk walking on the same schedule as the moderate exercise group; or a usual-care control group that received lifestyle counseling.
The primary endpoint was change in intrahepatic triglyceride content as measured by proton magnetic resonance spectroscopy at 12 months. It was reduced from baseline by 6.45% in the intensive exercise group and similarly by 6.19% with moderate exercise, both significantly greater effects than the 2.85% decrease in the control group.
Dr. Zhang reported having no financial conflicts of interest regarding this study.
ORLANDO – Moderate aerobic exercise and a more intensive regimen proved equally effective in reducing intrahepatic triglyceride content in centrally obese patients with nonalcoholic fatty liver disease in a 12-month randomized, controlled trial.
However, the more intensive exercise program had the edge in terms of favorable impact on metabolic risk factors. While both exercise prescriptions reduced body weight and blood pressure to a similar extent over the course of 12 months, the intensive-exercise group showed bigger reductions in body fat mass, visceral fat, and subcutaneous fat (see graphic), Dr. Hui-Jie Zhang of Xiamen (China) University said at the American Heart Association scientific sessions.

The observer-blinded study included 220 middle-age Chinese patients with central obesity and confirmed nonalcoholic fatty liver disease (NAFLD), a common risk factor for cardiovascular disease. They were randomized to three groups: a moderate exercise program that consisted of brisk walking for 150 minutes per week for 12 months; an intensive exercise regimen involving 30 minutes of treadmill running at 65%-80% of maximum oxygen consumption 5 days per week for 6 months, followed by 6 months of brisk walking on the same schedule as the moderate exercise group; or a usual-care control group that received lifestyle counseling.
The primary endpoint was change in intrahepatic triglyceride content as measured by proton magnetic resonance spectroscopy at 12 months. It was reduced from baseline by 6.45% in the intensive exercise group and similarly by 6.19% with moderate exercise, both significantly greater effects than the 2.85% decrease in the control group.
Dr. Zhang reported having no financial conflicts of interest regarding this study.
AT THE AHA SCIENTIFIC SESSIONS
Moderate, intensive exercise cut triglycerides equally in NAFLD
ORLANDO – Moderate aerobic exercise and a more intensive regimen proved equally effective in reducing intrahepatic triglyceride content in centrally obese patients with nonalcoholic fatty liver disease in a 12-month randomized, controlled trial.
However, the more intensive exercise program had the edge in terms of favorable impact on metabolic risk factors. While both exercise prescriptions reduced body weight and blood pressure to a similar extent over the course of 12 months, the intensive-exercise group showed bigger reductions in body fat mass, visceral fat, and subcutaneous fat (see graphic), Dr. Hui-Jie Zhang of Xiamen (China) University said at the American Heart Association scientific sessions.
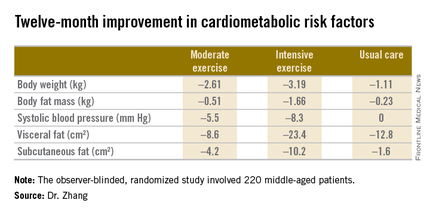
The observer-blinded study included 220 middle-age Chinese patients with central obesity and confirmed nonalcoholic fatty liver disease (NAFLD), a common risk factor for cardiovascular disease. They were randomized to three groups: a moderate exercise program that consisted of brisk walking for 150 minutes per week for 12 months; an intensive exercise regimen involving 30 minutes of treadmill running at 65%-80% of maximum oxygen consumption 5 days per week for 6 months, followed by 6 months of brisk walking on the same schedule as the moderate exercise group; or a usual-care control group that received lifestyle counseling.
The primary endpoint was change in intrahepatic triglyceride content as measured by proton magnetic resonance spectroscopy at 12 months. It was reduced from baseline by 6.45% in the intensive exercise group and similarly by 6.19% with moderate exercise, both significantly greater effects than the 2.85% decrease in the control group.
Dr. Zhang reported having no financial conflicts of interest regarding this study.
ORLANDO – Moderate aerobic exercise and a more intensive regimen proved equally effective in reducing intrahepatic triglyceride content in centrally obese patients with nonalcoholic fatty liver disease in a 12-month randomized, controlled trial.
However, the more intensive exercise program had the edge in terms of favorable impact on metabolic risk factors. While both exercise prescriptions reduced body weight and blood pressure to a similar extent over the course of 12 months, the intensive-exercise group showed bigger reductions in body fat mass, visceral fat, and subcutaneous fat (see graphic), Dr. Hui-Jie Zhang of Xiamen (China) University said at the American Heart Association scientific sessions.

The observer-blinded study included 220 middle-age Chinese patients with central obesity and confirmed nonalcoholic fatty liver disease (NAFLD), a common risk factor for cardiovascular disease. They were randomized to three groups: a moderate exercise program that consisted of brisk walking for 150 minutes per week for 12 months; an intensive exercise regimen involving 30 minutes of treadmill running at 65%-80% of maximum oxygen consumption 5 days per week for 6 months, followed by 6 months of brisk walking on the same schedule as the moderate exercise group; or a usual-care control group that received lifestyle counseling.
The primary endpoint was change in intrahepatic triglyceride content as measured by proton magnetic resonance spectroscopy at 12 months. It was reduced from baseline by 6.45% in the intensive exercise group and similarly by 6.19% with moderate exercise, both significantly greater effects than the 2.85% decrease in the control group.
Dr. Zhang reported having no financial conflicts of interest regarding this study.
ORLANDO – Moderate aerobic exercise and a more intensive regimen proved equally effective in reducing intrahepatic triglyceride content in centrally obese patients with nonalcoholic fatty liver disease in a 12-month randomized, controlled trial.
However, the more intensive exercise program had the edge in terms of favorable impact on metabolic risk factors. While both exercise prescriptions reduced body weight and blood pressure to a similar extent over the course of 12 months, the intensive-exercise group showed bigger reductions in body fat mass, visceral fat, and subcutaneous fat (see graphic), Dr. Hui-Jie Zhang of Xiamen (China) University said at the American Heart Association scientific sessions.

The observer-blinded study included 220 middle-age Chinese patients with central obesity and confirmed nonalcoholic fatty liver disease (NAFLD), a common risk factor for cardiovascular disease. They were randomized to three groups: a moderate exercise program that consisted of brisk walking for 150 minutes per week for 12 months; an intensive exercise regimen involving 30 minutes of treadmill running at 65%-80% of maximum oxygen consumption 5 days per week for 6 months, followed by 6 months of brisk walking on the same schedule as the moderate exercise group; or a usual-care control group that received lifestyle counseling.
The primary endpoint was change in intrahepatic triglyceride content as measured by proton magnetic resonance spectroscopy at 12 months. It was reduced from baseline by 6.45% in the intensive exercise group and similarly by 6.19% with moderate exercise, both significantly greater effects than the 2.85% decrease in the control group.
Dr. Zhang reported having no financial conflicts of interest regarding this study.
AT THE AHA SCIENTIFIC SESSIONS
Key clinical point: Moderate and intensive aerobic exercise, done regularly, are equally effective in reducing the intrahepatic triglyceride level in patients with nonalcoholic fatty liver disease.
Major finding: Intrahepatic triglyceride concentration in patients with NAFLD was reduced by a mean of 6.19% after 12 months on a moderate exercise program and 6.45% with a more intensive exercise regimen, compared with 2.85% in usual-care controls.
Data source: A 12-month, observer-blinded randomized trial in which 220 middle-age Chinese patients with NAFLD and central obesity were randomized to a program of moderate aerobic exercise, a more intensive regimen, or a usual-care control group.
Disclosures: The presenter reported having no financial conflicts of interest regarding this study.
Subtly elevated maternal glucose linked to increased risk of tetralogy of Fallot
ORLANDO – Subclinical elevated maternal blood glucose levels during the second trimester are strongly associated with increased risk of tetralogy of Fallot, Dr. James R. Priest reported at the American Heart Association scientific sessions.
It’s well established that maternal type 2 diabetes and gestational diabetes are strong maternal risk factors for structural congenital heart disease in their offspring. But the women in this study didn’t meet criteria for those diagnoses. They were asymptomatic, and their elevations in blood glucose in random nonfasting measurements obtained in the second trimester were below the 200 mg/dL threshold.
Thus, this study provides the first evidence that maternal blood glucose as a continuous variable is associated with increased risk of specific congenital heart defects in offspring, according to Dr. Priest, a pediatric cardiologist at Stanford (Calif.) University.
Of note, in this study of nondiabetic women, an elevated blood glucose was not associated with increased risk of d-transposition of the great arteries (dTGA), which, like tetralogy of Fallot, has been associated with maternal diabetes.
Dr. Priest presented an observational study of 277 pregnant, nondiabetic women, of whom 55 gave birth to a baby with tetralogy of Fallot, 42 had a baby with dTGA, and 180 gave birth to a baby without congenital heart disease or other malformations. Random nonfasting blood glucose and serum insulin levels were obtained from all subjects during weeks 15-18 of pregnancy.
The median maternal blood glucose level was 91.5 mg/dL in controls, similar at 90 mg/dL in the mothers of infants with dTGA, and significantly increased at 97 mg/dL in the mothers of offspring with tetralogy of Fallot. In a multivariate analysis adjusted for maternal age and ethnicity, elevated nonfasting second-trimester maternal glucose was associated with a 7.54-fold increased risk of having an infant with tetralogy of Fallot.
Maternal serum insulin level wasn’t associated with the risk of congenital heart disease.
Dr. Priest emphasized that while this study breaks new ground, the findings must be considered preliminary.
“Maternal blood glucose levels are influenced by diet, exercise, beta-cell function, and insulin resistance. Thus, blood glucose levels may simply be a marker of risk conferred by another physiologic process. The question remains whether elevated blood glucose or a variety of correlated but independent traits is behind the observed association,” he noted.
He plans to answer this key question by studying the relationship between these physiologic traits and blood glucose levels earlier in pregnancy.
This study was funded by the Stanford Cardiovascular Institute and the National Institutes of Health. Dr. Priest reported having no financial conflicts of interest.
ORLANDO – Subclinical elevated maternal blood glucose levels during the second trimester are strongly associated with increased risk of tetralogy of Fallot, Dr. James R. Priest reported at the American Heart Association scientific sessions.
It’s well established that maternal type 2 diabetes and gestational diabetes are strong maternal risk factors for structural congenital heart disease in their offspring. But the women in this study didn’t meet criteria for those diagnoses. They were asymptomatic, and their elevations in blood glucose in random nonfasting measurements obtained in the second trimester were below the 200 mg/dL threshold.
Thus, this study provides the first evidence that maternal blood glucose as a continuous variable is associated with increased risk of specific congenital heart defects in offspring, according to Dr. Priest, a pediatric cardiologist at Stanford (Calif.) University.
Of note, in this study of nondiabetic women, an elevated blood glucose was not associated with increased risk of d-transposition of the great arteries (dTGA), which, like tetralogy of Fallot, has been associated with maternal diabetes.
Dr. Priest presented an observational study of 277 pregnant, nondiabetic women, of whom 55 gave birth to a baby with tetralogy of Fallot, 42 had a baby with dTGA, and 180 gave birth to a baby without congenital heart disease or other malformations. Random nonfasting blood glucose and serum insulin levels were obtained from all subjects during weeks 15-18 of pregnancy.
The median maternal blood glucose level was 91.5 mg/dL in controls, similar at 90 mg/dL in the mothers of infants with dTGA, and significantly increased at 97 mg/dL in the mothers of offspring with tetralogy of Fallot. In a multivariate analysis adjusted for maternal age and ethnicity, elevated nonfasting second-trimester maternal glucose was associated with a 7.54-fold increased risk of having an infant with tetralogy of Fallot.
Maternal serum insulin level wasn’t associated with the risk of congenital heart disease.
Dr. Priest emphasized that while this study breaks new ground, the findings must be considered preliminary.
“Maternal blood glucose levels are influenced by diet, exercise, beta-cell function, and insulin resistance. Thus, blood glucose levels may simply be a marker of risk conferred by another physiologic process. The question remains whether elevated blood glucose or a variety of correlated but independent traits is behind the observed association,” he noted.
He plans to answer this key question by studying the relationship between these physiologic traits and blood glucose levels earlier in pregnancy.
This study was funded by the Stanford Cardiovascular Institute and the National Institutes of Health. Dr. Priest reported having no financial conflicts of interest.
ORLANDO – Subclinical elevated maternal blood glucose levels during the second trimester are strongly associated with increased risk of tetralogy of Fallot, Dr. James R. Priest reported at the American Heart Association scientific sessions.
It’s well established that maternal type 2 diabetes and gestational diabetes are strong maternal risk factors for structural congenital heart disease in their offspring. But the women in this study didn’t meet criteria for those diagnoses. They were asymptomatic, and their elevations in blood glucose in random nonfasting measurements obtained in the second trimester were below the 200 mg/dL threshold.
Thus, this study provides the first evidence that maternal blood glucose as a continuous variable is associated with increased risk of specific congenital heart defects in offspring, according to Dr. Priest, a pediatric cardiologist at Stanford (Calif.) University.
Of note, in this study of nondiabetic women, an elevated blood glucose was not associated with increased risk of d-transposition of the great arteries (dTGA), which, like tetralogy of Fallot, has been associated with maternal diabetes.
Dr. Priest presented an observational study of 277 pregnant, nondiabetic women, of whom 55 gave birth to a baby with tetralogy of Fallot, 42 had a baby with dTGA, and 180 gave birth to a baby without congenital heart disease or other malformations. Random nonfasting blood glucose and serum insulin levels were obtained from all subjects during weeks 15-18 of pregnancy.
The median maternal blood glucose level was 91.5 mg/dL in controls, similar at 90 mg/dL in the mothers of infants with dTGA, and significantly increased at 97 mg/dL in the mothers of offspring with tetralogy of Fallot. In a multivariate analysis adjusted for maternal age and ethnicity, elevated nonfasting second-trimester maternal glucose was associated with a 7.54-fold increased risk of having an infant with tetralogy of Fallot.
Maternal serum insulin level wasn’t associated with the risk of congenital heart disease.
Dr. Priest emphasized that while this study breaks new ground, the findings must be considered preliminary.
“Maternal blood glucose levels are influenced by diet, exercise, beta-cell function, and insulin resistance. Thus, blood glucose levels may simply be a marker of risk conferred by another physiologic process. The question remains whether elevated blood glucose or a variety of correlated but independent traits is behind the observed association,” he noted.
He plans to answer this key question by studying the relationship between these physiologic traits and blood glucose levels earlier in pregnancy.
This study was funded by the Stanford Cardiovascular Institute and the National Institutes of Health. Dr. Priest reported having no financial conflicts of interest.
AT THE AHA SCIENTIFIC SESSIONS
Key clinical point: Second-trimester subclinical elevated maternal blood glucose in nondiabetic women is associated with sharply increased risk of tetralogy of Fallot.
Major finding: Subtly elevated maternal blood glucose in nondiabetic women in their second trimester is associated with a 7.54-fold increased risk of tetralogy of Fallot in the baby.
Data source: This observational study involved prospectively collected data on 277 pregnant women included in California’s birth defects screening program.
Disclosures: This study was funded by the Stanford Cardiovascular Institute and the National Institutes of Health. The presenter reported having no financial conflicts of interest.