User login
Small fiber neuropathy common, vexing in sarcoidosis
MAUI, HAWAII – Small fiber neuropathy is a common and underappreciated expression of systemic sarcoidosis, Dr. Alvin F. Wells observed at the 2016 Rheumatology Winter Clinical Symposium.
Small fiber neuropathy (SFN) occurs in roughly 40% of patients with sarcoidosis. Affected patients present with painful neuropathic symptoms and/or dysautonomia. SFN is often mistaken for fibromyalgia syndrome. And it poses a special therapeutic challenge.
“It can be very, very difficult to treat. It’s generally resistant to methotrexate, even to corticosteroids, our treatment mainstay in sarcoidosis,” said Dr. Wells, director of the Rheumatology and Immunotherapy Center in Franklin, Wisc.
He credited researchers at the Cleveland Clinic with bringing intravenous immunoglobulin (IVIG) to wider attention as an effective treatment for refractory SFN associated with sarcoidosis (Respir Med. 2011 Jan;105[1]:101-5).
“Here you’re treating it more like an immune-mediated neuropathy,” Dr. Wells noted.”It’s expensive therapy, but when these patients have a positive biopsy and they’ve failed other types of treatment, these data show IVIG can achieve a good response.”
Indeed, the Cleveland Clinic physicians reported excellent success with IVIG dosed at 2 g/kg initially, then 1 g/kg in 2 weeks, followed by maintenance dosing at 1 g/kg every 4 weeks.
This sarcoidosis-associated neuropathy involves both myelinated and nonmyelinated small nerve fibers. The diagnosis is established by epidermal nerve fiber density testing. This entails taking three 3-mm skin punch biopsies, one each at the lateral proximal and distal thigh, the third 10 cm proximal to the lateral malleolus. Specimens obtained from these sites in normal individuals feature a rich density of small nerve fibers; in patients with SNF-associated sarcoidosis, there is a notable paucity of the fibers, the rheumatologist explained.
Treating organ involvement besides SNF
Studies have consistently shown that the lungs and thoracic lymph nodes are the organs most commonly involved in sarcoidosis, affecting more than 90% of patients. Indeed, respiratory symptoms are most often the complaint that brings patients in seeking medical attention. The skin is involved in about 30% of cases, the eyes in 20%-25%, and the liver or heart in roughly 20% each.
Osteoarticular involvement is uncommon. Moroccan investigators for a study presented at the 2015 European League Against Rheumatism (EULAR) meeting in Rome concluded that when osteoarticular sarcoidosis occurs, it most often takes the form of an inflammatory chronic polyarthritis (Ann Rheum Dis. 2015;74[Suppl2]:786). Bone involvement is rare but damaging and mostly affects small distal bones, Dr. Wells said.
With the exception of sarcoidosis-associated SNF, the other types of organ involvement typically respond well to corticosteroids.
“Dosing depends upon disease severity. Most of us use 1 mg/kg to get ocular disease under control,” according to the rheumatologist. “The disease is very organ-specific. So if someone has eye disease, we throw everything at them, including the kitchen sink, to make sure they don’t go blind.”
“Steroids are the mainstay,” Dr. Wells emphasized. “The question to ask is, ‘What can I find that’s steroid-sparing and yet keeps the same kind of clinical response?’ ”
It’s virtually all off-label therapy. There is a dearth of randomized, blinded, placebo-controlled treatment trials in sarcoidosis. Many, many agents have been tried as second-, third-, and fourth-line therapy, including various disease-modifying antirheumatic drugs, tumor necrosis factor inhibitors, and phosphodiesterase-4 inhibitors. Dr. Wells’ favorites are methotrexate, azathioprine, and mycophenolate mofetil. However, hydroxychloroquine works well for skin disease, chloroquine helps combat hypercalcemia, and pentoxifylline and thalidomide can be helpful in cases of treatment-resistant lupus pernio.
A study presented at the 2015 EULAR meeting in Rome showed high-dose methotrexate in the 25-30 mg/week range was significantly more effective than mycophenolate mofetil in preventing relapses of neurosarcoidosis (Ann Rheum Dis. 2015;74[Suppl2]:859). This study has caused Dr. Wells to change his own practice.
He noted that the oral phosphodiesterase-4 inhibitor apremilast (Otezla) at 20 mg/day added to background therapy achieved “really dramatic results” for refractory chronic cutaneous sarcoidosis in a 15-patient, open-label, phase II study (Arch Dermatol. 2012 Feb;148[2]:262-4). A definitive randomized, placebo-controlled trial is certainly warranted, he added.
A question often asked by patients with extrathoracic sarcoid is, “Am I at greater risk of mortality than if I just had pulmonary disease?” The answer is no, Dr. Wells said, citing a study presented by investigators from Barcelona at the 2015 EULAR meeting (Ann Rheum Dis. 2015;74[Suppl2]:404). Mortality during a mean follow-up of 107 months was 11% in patients with extrapulmonary involvement and similar at 14% in patients whose sarcoidosis was limited to the lungs.
He reported serving as a consultant to roughly a dozen pharmaceutical companies.
MAUI, HAWAII – Small fiber neuropathy is a common and underappreciated expression of systemic sarcoidosis, Dr. Alvin F. Wells observed at the 2016 Rheumatology Winter Clinical Symposium.
Small fiber neuropathy (SFN) occurs in roughly 40% of patients with sarcoidosis. Affected patients present with painful neuropathic symptoms and/or dysautonomia. SFN is often mistaken for fibromyalgia syndrome. And it poses a special therapeutic challenge.
“It can be very, very difficult to treat. It’s generally resistant to methotrexate, even to corticosteroids, our treatment mainstay in sarcoidosis,” said Dr. Wells, director of the Rheumatology and Immunotherapy Center in Franklin, Wisc.
He credited researchers at the Cleveland Clinic with bringing intravenous immunoglobulin (IVIG) to wider attention as an effective treatment for refractory SFN associated with sarcoidosis (Respir Med. 2011 Jan;105[1]:101-5).
“Here you’re treating it more like an immune-mediated neuropathy,” Dr. Wells noted.”It’s expensive therapy, but when these patients have a positive biopsy and they’ve failed other types of treatment, these data show IVIG can achieve a good response.”
Indeed, the Cleveland Clinic physicians reported excellent success with IVIG dosed at 2 g/kg initially, then 1 g/kg in 2 weeks, followed by maintenance dosing at 1 g/kg every 4 weeks.
This sarcoidosis-associated neuropathy involves both myelinated and nonmyelinated small nerve fibers. The diagnosis is established by epidermal nerve fiber density testing. This entails taking three 3-mm skin punch biopsies, one each at the lateral proximal and distal thigh, the third 10 cm proximal to the lateral malleolus. Specimens obtained from these sites in normal individuals feature a rich density of small nerve fibers; in patients with SNF-associated sarcoidosis, there is a notable paucity of the fibers, the rheumatologist explained.
Treating organ involvement besides SNF
Studies have consistently shown that the lungs and thoracic lymph nodes are the organs most commonly involved in sarcoidosis, affecting more than 90% of patients. Indeed, respiratory symptoms are most often the complaint that brings patients in seeking medical attention. The skin is involved in about 30% of cases, the eyes in 20%-25%, and the liver or heart in roughly 20% each.
Osteoarticular involvement is uncommon. Moroccan investigators for a study presented at the 2015 European League Against Rheumatism (EULAR) meeting in Rome concluded that when osteoarticular sarcoidosis occurs, it most often takes the form of an inflammatory chronic polyarthritis (Ann Rheum Dis. 2015;74[Suppl2]:786). Bone involvement is rare but damaging and mostly affects small distal bones, Dr. Wells said.
With the exception of sarcoidosis-associated SNF, the other types of organ involvement typically respond well to corticosteroids.
“Dosing depends upon disease severity. Most of us use 1 mg/kg to get ocular disease under control,” according to the rheumatologist. “The disease is very organ-specific. So if someone has eye disease, we throw everything at them, including the kitchen sink, to make sure they don’t go blind.”
“Steroids are the mainstay,” Dr. Wells emphasized. “The question to ask is, ‘What can I find that’s steroid-sparing and yet keeps the same kind of clinical response?’ ”
It’s virtually all off-label therapy. There is a dearth of randomized, blinded, placebo-controlled treatment trials in sarcoidosis. Many, many agents have been tried as second-, third-, and fourth-line therapy, including various disease-modifying antirheumatic drugs, tumor necrosis factor inhibitors, and phosphodiesterase-4 inhibitors. Dr. Wells’ favorites are methotrexate, azathioprine, and mycophenolate mofetil. However, hydroxychloroquine works well for skin disease, chloroquine helps combat hypercalcemia, and pentoxifylline and thalidomide can be helpful in cases of treatment-resistant lupus pernio.
A study presented at the 2015 EULAR meeting in Rome showed high-dose methotrexate in the 25-30 mg/week range was significantly more effective than mycophenolate mofetil in preventing relapses of neurosarcoidosis (Ann Rheum Dis. 2015;74[Suppl2]:859). This study has caused Dr. Wells to change his own practice.
He noted that the oral phosphodiesterase-4 inhibitor apremilast (Otezla) at 20 mg/day added to background therapy achieved “really dramatic results” for refractory chronic cutaneous sarcoidosis in a 15-patient, open-label, phase II study (Arch Dermatol. 2012 Feb;148[2]:262-4). A definitive randomized, placebo-controlled trial is certainly warranted, he added.
A question often asked by patients with extrathoracic sarcoid is, “Am I at greater risk of mortality than if I just had pulmonary disease?” The answer is no, Dr. Wells said, citing a study presented by investigators from Barcelona at the 2015 EULAR meeting (Ann Rheum Dis. 2015;74[Suppl2]:404). Mortality during a mean follow-up of 107 months was 11% in patients with extrapulmonary involvement and similar at 14% in patients whose sarcoidosis was limited to the lungs.
He reported serving as a consultant to roughly a dozen pharmaceutical companies.
MAUI, HAWAII – Small fiber neuropathy is a common and underappreciated expression of systemic sarcoidosis, Dr. Alvin F. Wells observed at the 2016 Rheumatology Winter Clinical Symposium.
Small fiber neuropathy (SFN) occurs in roughly 40% of patients with sarcoidosis. Affected patients present with painful neuropathic symptoms and/or dysautonomia. SFN is often mistaken for fibromyalgia syndrome. And it poses a special therapeutic challenge.
“It can be very, very difficult to treat. It’s generally resistant to methotrexate, even to corticosteroids, our treatment mainstay in sarcoidosis,” said Dr. Wells, director of the Rheumatology and Immunotherapy Center in Franklin, Wisc.
He credited researchers at the Cleveland Clinic with bringing intravenous immunoglobulin (IVIG) to wider attention as an effective treatment for refractory SFN associated with sarcoidosis (Respir Med. 2011 Jan;105[1]:101-5).
“Here you’re treating it more like an immune-mediated neuropathy,” Dr. Wells noted.”It’s expensive therapy, but when these patients have a positive biopsy and they’ve failed other types of treatment, these data show IVIG can achieve a good response.”
Indeed, the Cleveland Clinic physicians reported excellent success with IVIG dosed at 2 g/kg initially, then 1 g/kg in 2 weeks, followed by maintenance dosing at 1 g/kg every 4 weeks.
This sarcoidosis-associated neuropathy involves both myelinated and nonmyelinated small nerve fibers. The diagnosis is established by epidermal nerve fiber density testing. This entails taking three 3-mm skin punch biopsies, one each at the lateral proximal and distal thigh, the third 10 cm proximal to the lateral malleolus. Specimens obtained from these sites in normal individuals feature a rich density of small nerve fibers; in patients with SNF-associated sarcoidosis, there is a notable paucity of the fibers, the rheumatologist explained.
Treating organ involvement besides SNF
Studies have consistently shown that the lungs and thoracic lymph nodes are the organs most commonly involved in sarcoidosis, affecting more than 90% of patients. Indeed, respiratory symptoms are most often the complaint that brings patients in seeking medical attention. The skin is involved in about 30% of cases, the eyes in 20%-25%, and the liver or heart in roughly 20% each.
Osteoarticular involvement is uncommon. Moroccan investigators for a study presented at the 2015 European League Against Rheumatism (EULAR) meeting in Rome concluded that when osteoarticular sarcoidosis occurs, it most often takes the form of an inflammatory chronic polyarthritis (Ann Rheum Dis. 2015;74[Suppl2]:786). Bone involvement is rare but damaging and mostly affects small distal bones, Dr. Wells said.
With the exception of sarcoidosis-associated SNF, the other types of organ involvement typically respond well to corticosteroids.
“Dosing depends upon disease severity. Most of us use 1 mg/kg to get ocular disease under control,” according to the rheumatologist. “The disease is very organ-specific. So if someone has eye disease, we throw everything at them, including the kitchen sink, to make sure they don’t go blind.”
“Steroids are the mainstay,” Dr. Wells emphasized. “The question to ask is, ‘What can I find that’s steroid-sparing and yet keeps the same kind of clinical response?’ ”
It’s virtually all off-label therapy. There is a dearth of randomized, blinded, placebo-controlled treatment trials in sarcoidosis. Many, many agents have been tried as second-, third-, and fourth-line therapy, including various disease-modifying antirheumatic drugs, tumor necrosis factor inhibitors, and phosphodiesterase-4 inhibitors. Dr. Wells’ favorites are methotrexate, azathioprine, and mycophenolate mofetil. However, hydroxychloroquine works well for skin disease, chloroquine helps combat hypercalcemia, and pentoxifylline and thalidomide can be helpful in cases of treatment-resistant lupus pernio.
A study presented at the 2015 EULAR meeting in Rome showed high-dose methotrexate in the 25-30 mg/week range was significantly more effective than mycophenolate mofetil in preventing relapses of neurosarcoidosis (Ann Rheum Dis. 2015;74[Suppl2]:859). This study has caused Dr. Wells to change his own practice.
He noted that the oral phosphodiesterase-4 inhibitor apremilast (Otezla) at 20 mg/day added to background therapy achieved “really dramatic results” for refractory chronic cutaneous sarcoidosis in a 15-patient, open-label, phase II study (Arch Dermatol. 2012 Feb;148[2]:262-4). A definitive randomized, placebo-controlled trial is certainly warranted, he added.
A question often asked by patients with extrathoracic sarcoid is, “Am I at greater risk of mortality than if I just had pulmonary disease?” The answer is no, Dr. Wells said, citing a study presented by investigators from Barcelona at the 2015 EULAR meeting (Ann Rheum Dis. 2015;74[Suppl2]:404). Mortality during a mean follow-up of 107 months was 11% in patients with extrapulmonary involvement and similar at 14% in patients whose sarcoidosis was limited to the lungs.
He reported serving as a consultant to roughly a dozen pharmaceutical companies.
EXPERT ANALYSIS FROM RWCS 2016
Drug-eluting Stent Recipients Can Safely Have Surgery Sooner
CHICAGO – Current U.S. and European guidelines recommending postponement of noncardiac surgery for 6-12 months after drug-eluting stent implantation appear to be excessive, Dr. Gro Egholm reported at the annual meeting of the American College of Cardiology.
She presented a large retrospective observational study of outcomes in patients undergoing various types of noncardiac surgery in western Denmark during 2005-2012. Among 4,303 patients who had noncardiac surgery within 12 months after receiving a drug-eluting stent (DES), only those whose operations took place during the first month post stenting had increased risks of acute MI and cardiac death within 30 days post surgery.
Risks of major adverse cardiac events among the DES recipients who had noncardiac surgery within that first month post–percutaneous coronary intervention were increased roughly 7.5-fold compared with controls, but for surgery performed after that the risks of MI and cardiac death dropped off abruptly and were no different from rates in 20,232 controls without ischemic heart disease or stents who were matched for age, gender, surgical procedure, and Charlson Comorbidity Index, according to Dr. Egholm of Aarhus (Denmark) University.
Moreover, even in DES recipients undergoing noncardiac surgery during the first month post stenting, all-cause mortality was no greater than in controls.
“Surgery could be performed much earlier than recommended,” she concluded.
Her study was carried out by linking data from comprehensive regional and national Danish health care registries. Most patients with DES remained on dual antiplatelet therapy periprocedurally. The exceptions were neurosurgical operations and others where it’s standard that dual antiplatelet therapy must be stopped.
“If you can continue only one antiplatelet agent, aspirin would be the most appealing,” she said.
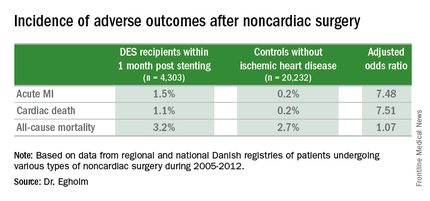
Of the DES participants, 56% received their device as treatment for an acute coronary syndrome. The average time from stent placement to noncardiac surgery in this large series was 147 days.
Session co-chair Dr. Sunil V. Rao of Duke University in Durham, N.C., called this work “a very important study that’s relevant to daily practice.” However, he found the 23% incidence of noncardiac surgery within 12 months following DES implantation reported in Dr. Egholm’s study to be “shockingly high.” She agreed, noting that rates in some non-Danish registries she’s looked at are more in the 8%-15% range. But Denmark’s health care registries are known for rigorous accuracy and completeness.
Dr. Egholm reported having no financial conflicts regarding her study.
CHICAGO – Current U.S. and European guidelines recommending postponement of noncardiac surgery for 6-12 months after drug-eluting stent implantation appear to be excessive, Dr. Gro Egholm reported at the annual meeting of the American College of Cardiology.
She presented a large retrospective observational study of outcomes in patients undergoing various types of noncardiac surgery in western Denmark during 2005-2012. Among 4,303 patients who had noncardiac surgery within 12 months after receiving a drug-eluting stent (DES), only those whose operations took place during the first month post stenting had increased risks of acute MI and cardiac death within 30 days post surgery.
Risks of major adverse cardiac events among the DES recipients who had noncardiac surgery within that first month post–percutaneous coronary intervention were increased roughly 7.5-fold compared with controls, but for surgery performed after that the risks of MI and cardiac death dropped off abruptly and were no different from rates in 20,232 controls without ischemic heart disease or stents who were matched for age, gender, surgical procedure, and Charlson Comorbidity Index, according to Dr. Egholm of Aarhus (Denmark) University.
Moreover, even in DES recipients undergoing noncardiac surgery during the first month post stenting, all-cause mortality was no greater than in controls.
“Surgery could be performed much earlier than recommended,” she concluded.
Her study was carried out by linking data from comprehensive regional and national Danish health care registries. Most patients with DES remained on dual antiplatelet therapy periprocedurally. The exceptions were neurosurgical operations and others where it’s standard that dual antiplatelet therapy must be stopped.
“If you can continue only one antiplatelet agent, aspirin would be the most appealing,” she said.

Of the DES participants, 56% received their device as treatment for an acute coronary syndrome. The average time from stent placement to noncardiac surgery in this large series was 147 days.
Session co-chair Dr. Sunil V. Rao of Duke University in Durham, N.C., called this work “a very important study that’s relevant to daily practice.” However, he found the 23% incidence of noncardiac surgery within 12 months following DES implantation reported in Dr. Egholm’s study to be “shockingly high.” She agreed, noting that rates in some non-Danish registries she’s looked at are more in the 8%-15% range. But Denmark’s health care registries are known for rigorous accuracy and completeness.
Dr. Egholm reported having no financial conflicts regarding her study.
CHICAGO – Current U.S. and European guidelines recommending postponement of noncardiac surgery for 6-12 months after drug-eluting stent implantation appear to be excessive, Dr. Gro Egholm reported at the annual meeting of the American College of Cardiology.
She presented a large retrospective observational study of outcomes in patients undergoing various types of noncardiac surgery in western Denmark during 2005-2012. Among 4,303 patients who had noncardiac surgery within 12 months after receiving a drug-eluting stent (DES), only those whose operations took place during the first month post stenting had increased risks of acute MI and cardiac death within 30 days post surgery.
Risks of major adverse cardiac events among the DES recipients who had noncardiac surgery within that first month post–percutaneous coronary intervention were increased roughly 7.5-fold compared with controls, but for surgery performed after that the risks of MI and cardiac death dropped off abruptly and were no different from rates in 20,232 controls without ischemic heart disease or stents who were matched for age, gender, surgical procedure, and Charlson Comorbidity Index, according to Dr. Egholm of Aarhus (Denmark) University.
Moreover, even in DES recipients undergoing noncardiac surgery during the first month post stenting, all-cause mortality was no greater than in controls.
“Surgery could be performed much earlier than recommended,” she concluded.
Her study was carried out by linking data from comprehensive regional and national Danish health care registries. Most patients with DES remained on dual antiplatelet therapy periprocedurally. The exceptions were neurosurgical operations and others where it’s standard that dual antiplatelet therapy must be stopped.
“If you can continue only one antiplatelet agent, aspirin would be the most appealing,” she said.

Of the DES participants, 56% received their device as treatment for an acute coronary syndrome. The average time from stent placement to noncardiac surgery in this large series was 147 days.
Session co-chair Dr. Sunil V. Rao of Duke University in Durham, N.C., called this work “a very important study that’s relevant to daily practice.” However, he found the 23% incidence of noncardiac surgery within 12 months following DES implantation reported in Dr. Egholm’s study to be “shockingly high.” She agreed, noting that rates in some non-Danish registries she’s looked at are more in the 8%-15% range. But Denmark’s health care registries are known for rigorous accuracy and completeness.
Dr. Egholm reported having no financial conflicts regarding her study.
AT ACC 16
Drug-eluting stent recipients can safely have surgery sooner
CHICAGO – Current U.S. and European guidelines recommending postponement of noncardiac surgery for 6-12 months after drug-eluting stent implantation appear to be excessive, Dr. Gro Egholm reported at the annual meeting of the American College of Cardiology.
She presented a large retrospective observational study of outcomes in patients undergoing various types of noncardiac surgery in western Denmark during 2005-2012. Among 4,303 patients who had noncardiac surgery within 12 months after receiving a drug-eluting stent (DES), only those whose operations took place during the first month post stenting had increased risks of acute MI and cardiac death within 30 days post surgery.
Risks of major adverse cardiac events among the DES recipients who had noncardiac surgery within that first month post–percutaneous coronary intervention were increased roughly 7.5-fold compared with controls, but for surgery performed after that the risks of MI and cardiac death dropped off abruptly and were no different from rates in 20,232 controls without ischemic heart disease or stents who were matched for age, gender, surgical procedure, and Charlson Comorbidity Index, according to Dr. Egholm of Aarhus (Denmark) University.
Moreover, even in DES recipients undergoing noncardiac surgery during the first month post stenting, all-cause mortality was no greater than in controls.
“Surgery could be performed much earlier than recommended,” she concluded.
Her study was carried out by linking data from comprehensive regional and national Danish health care registries. Most patients with DES remained on dual antiplatelet therapy periprocedurally. The exceptions were neurosurgical operations and others where it’s standard that dual antiplatelet therapy must be stopped.
“If you can continue only one antiplatelet agent, aspirin would be the most appealing,” she said.
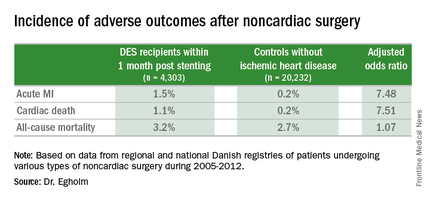
Of the DES participants, 56% received their device as treatment for an acute coronary syndrome. The average time from stent placement to noncardiac surgery in this large series was 147 days.
Session co-chair Dr. Sunil V. Rao of Duke University in Durham, N.C., called this work “a very important study that’s relevant to daily practice.” However, he found the 23% incidence of noncardiac surgery within 12 months following DES implantation reported in Dr. Egholm’s study to be “shockingly high.” She agreed, noting that rates in some non-Danish registries she’s looked at are more in the 8%-15% range. But Denmark’s health care registries are known for rigorous accuracy and completeness.
Dr. Egholm reported having no financial conflicts regarding her study.
CHICAGO – Current U.S. and European guidelines recommending postponement of noncardiac surgery for 6-12 months after drug-eluting stent implantation appear to be excessive, Dr. Gro Egholm reported at the annual meeting of the American College of Cardiology.
She presented a large retrospective observational study of outcomes in patients undergoing various types of noncardiac surgery in western Denmark during 2005-2012. Among 4,303 patients who had noncardiac surgery within 12 months after receiving a drug-eluting stent (DES), only those whose operations took place during the first month post stenting had increased risks of acute MI and cardiac death within 30 days post surgery.
Risks of major adverse cardiac events among the DES recipients who had noncardiac surgery within that first month post–percutaneous coronary intervention were increased roughly 7.5-fold compared with controls, but for surgery performed after that the risks of MI and cardiac death dropped off abruptly and were no different from rates in 20,232 controls without ischemic heart disease or stents who were matched for age, gender, surgical procedure, and Charlson Comorbidity Index, according to Dr. Egholm of Aarhus (Denmark) University.
Moreover, even in DES recipients undergoing noncardiac surgery during the first month post stenting, all-cause mortality was no greater than in controls.
“Surgery could be performed much earlier than recommended,” she concluded.
Her study was carried out by linking data from comprehensive regional and national Danish health care registries. Most patients with DES remained on dual antiplatelet therapy periprocedurally. The exceptions were neurosurgical operations and others where it’s standard that dual antiplatelet therapy must be stopped.
“If you can continue only one antiplatelet agent, aspirin would be the most appealing,” she said.

Of the DES participants, 56% received their device as treatment for an acute coronary syndrome. The average time from stent placement to noncardiac surgery in this large series was 147 days.
Session co-chair Dr. Sunil V. Rao of Duke University in Durham, N.C., called this work “a very important study that’s relevant to daily practice.” However, he found the 23% incidence of noncardiac surgery within 12 months following DES implantation reported in Dr. Egholm’s study to be “shockingly high.” She agreed, noting that rates in some non-Danish registries she’s looked at are more in the 8%-15% range. But Denmark’s health care registries are known for rigorous accuracy and completeness.
Dr. Egholm reported having no financial conflicts regarding her study.
CHICAGO – Current U.S. and European guidelines recommending postponement of noncardiac surgery for 6-12 months after drug-eluting stent implantation appear to be excessive, Dr. Gro Egholm reported at the annual meeting of the American College of Cardiology.
She presented a large retrospective observational study of outcomes in patients undergoing various types of noncardiac surgery in western Denmark during 2005-2012. Among 4,303 patients who had noncardiac surgery within 12 months after receiving a drug-eluting stent (DES), only those whose operations took place during the first month post stenting had increased risks of acute MI and cardiac death within 30 days post surgery.
Risks of major adverse cardiac events among the DES recipients who had noncardiac surgery within that first month post–percutaneous coronary intervention were increased roughly 7.5-fold compared with controls, but for surgery performed after that the risks of MI and cardiac death dropped off abruptly and were no different from rates in 20,232 controls without ischemic heart disease or stents who were matched for age, gender, surgical procedure, and Charlson Comorbidity Index, according to Dr. Egholm of Aarhus (Denmark) University.
Moreover, even in DES recipients undergoing noncardiac surgery during the first month post stenting, all-cause mortality was no greater than in controls.
“Surgery could be performed much earlier than recommended,” she concluded.
Her study was carried out by linking data from comprehensive regional and national Danish health care registries. Most patients with DES remained on dual antiplatelet therapy periprocedurally. The exceptions were neurosurgical operations and others where it’s standard that dual antiplatelet therapy must be stopped.
“If you can continue only one antiplatelet agent, aspirin would be the most appealing,” she said.

Of the DES participants, 56% received their device as treatment for an acute coronary syndrome. The average time from stent placement to noncardiac surgery in this large series was 147 days.
Session co-chair Dr. Sunil V. Rao of Duke University in Durham, N.C., called this work “a very important study that’s relevant to daily practice.” However, he found the 23% incidence of noncardiac surgery within 12 months following DES implantation reported in Dr. Egholm’s study to be “shockingly high.” She agreed, noting that rates in some non-Danish registries she’s looked at are more in the 8%-15% range. But Denmark’s health care registries are known for rigorous accuracy and completeness.
Dr. Egholm reported having no financial conflicts regarding her study.
AT ACC 16
Key clinical point: The risk of noncardiac surgery is elevated only when the operation occurs during the first month after stenting.
Major finding: Danish drug-eluting stent recipients who underwent noncardiac surgery within 1 month after stent placement were at 7.5-fold increased risks of acute MI and cardiac death, but surgery performed 2-12 months post stenting carried no increased risks.
Data source: This retrospective observational study based upon large Danish patient registries compared outcomes of noncardiac surgery performed within 12 months after drug-eluting stent placement in 4,303 patients with 20,232 matched controls without ischemic heart disease who underwent the same operations.
Disclosures: The study was supported by Danish research funds. The presenter reported having no financial conflicts of interest.
Novel drug fails to prevent contrast-induced nephropathy
CHICAGO – CMX-2043, a novel agent intended for prevention of contrast-induced nephropathy, failed in the phase II, double-blind, placebo-controlled CARIN clinical trial presented at the annual meeting of the American College of Cardiology.
The drug had also shown promise in small preliminary studies for the prevention of periprocedural myocardial infarction in patients undergoing coronary stenting. There again, however, CMX-2043 – a derivative of alpha lipoic acid with antioxidant and cell membrane–stabilizing properties – proved ineffective in the 361-patient, 31-center phase II trial, reported Dr. Deepak L. Bhatt, professor of medicine at Harvard Medical School and executive director of interventional cardiovascular programs at Brigham and Women’s Hospital, both in Boston.
All participants in CARIN had baseline severe impairment of kidney function or mild to moderate renal impairment plus another risk factor, such as diabetes or age greater than 75 years. One hour prior to coronary angiography, they received various doses of CMX-2043 or placebo.
Unfortunately, no difference between the four treatment arms was present in terms of the primary study endpoint: the incidence of acute kidney injury as defined by at least a 0.3 mg/dL rise in serum creatinine from baseline on day 4. No dose response to CMX-2043 was evident, nor did the investigational agent have any impact on the risk of major adverse cardiovascular events.
Immediately prior to Dr. Bhatt’s presentation, Dr. Michelle L. O’Donoghue of Brigham and Women’s Hospital presented the equally negative results of the LATITUDE-TIMI 60 trial, a phase III trial of the investigational mitogen-activated protein kinase inhibitor losmapimod, a drug developed to improve outcomes in patients with an acute coronary syndrome.
“It’s a bit distressing” to witness back to back presentations of clinical trials that proved resoundingly negative despite very strong-looking preliminary data, commented discussant Dr. Anthony N. DeMaria, professor of medicine at the University of California, San Diego. What’s going on here? he asked.
“I think it’s a fundamental truth that a lot of things that look good in preclinical work, even when backed up by a lot of solid science, don’t pan out in human studies,” Dr. Bhatt replied. “That’s a challenge, and probably in no other arena more so than in tackling inflammation and antioxidant therapy.
“There’s a graveyard of compounds that have not worked, and now we’ve perhaps added another one,” Dr. Bhatt continued. “But it doesn’t mean that scientific inquiry isn’t important, because I think eventually we’ll have drugs for these problems, whether it’s reperfusion injury or contrast-induced nephropathy. It’ll probably just take a lot more time and effort.”
The one solace regarding the CARIN trial, in Dr. Bhatt’s view, is that it highlighted the advantages of what is known as an adaptive trial design. Instead of jumping from positive early-phase results straight to a definitive 10,000-patient phase III clinical trial, investigators were able to obtain answers regarding the drug’s ability to prevent two major problems in patients undergoing coronary angiography – contrast-induced nephropathy and major adverse cardiac events – by means of a single 361-patient trial that was comparatively inexpensive.
Acute kidney injury secondary to exposure to contrast agents remains a significant problem, with an incidence of 20%-25% in high-risk patients. Numerous proposed prophylactic agents have ultimately proved not useful, including sodium bicarbonate, N-acetylcysteine, and intravenous fenoldopam.
Indeed, the only preventive measures of proven effectiveness are hydration with saline for 12 hours preangioplasty, and limiting the volume of contrast agent used. In real-world clinical practice, however, it’s often impractical to administer the optimal 12 hours of saline because of hospital pressure to get patients out quickly, Dr. Bhatt observed.
“There remains an important unmet clinical need to find agents that reduce the occurrence of contrast nephropathy,” he stressed.
Ischemix funded the CARIN trial. Dr. Bhatt reported receiving a research grant from the company that was directed to Brigham and Women’s Hospital.
CHICAGO – CMX-2043, a novel agent intended for prevention of contrast-induced nephropathy, failed in the phase II, double-blind, placebo-controlled CARIN clinical trial presented at the annual meeting of the American College of Cardiology.
The drug had also shown promise in small preliminary studies for the prevention of periprocedural myocardial infarction in patients undergoing coronary stenting. There again, however, CMX-2043 – a derivative of alpha lipoic acid with antioxidant and cell membrane–stabilizing properties – proved ineffective in the 361-patient, 31-center phase II trial, reported Dr. Deepak L. Bhatt, professor of medicine at Harvard Medical School and executive director of interventional cardiovascular programs at Brigham and Women’s Hospital, both in Boston.
All participants in CARIN had baseline severe impairment of kidney function or mild to moderate renal impairment plus another risk factor, such as diabetes or age greater than 75 years. One hour prior to coronary angiography, they received various doses of CMX-2043 or placebo.
Unfortunately, no difference between the four treatment arms was present in terms of the primary study endpoint: the incidence of acute kidney injury as defined by at least a 0.3 mg/dL rise in serum creatinine from baseline on day 4. No dose response to CMX-2043 was evident, nor did the investigational agent have any impact on the risk of major adverse cardiovascular events.
Immediately prior to Dr. Bhatt’s presentation, Dr. Michelle L. O’Donoghue of Brigham and Women’s Hospital presented the equally negative results of the LATITUDE-TIMI 60 trial, a phase III trial of the investigational mitogen-activated protein kinase inhibitor losmapimod, a drug developed to improve outcomes in patients with an acute coronary syndrome.
“It’s a bit distressing” to witness back to back presentations of clinical trials that proved resoundingly negative despite very strong-looking preliminary data, commented discussant Dr. Anthony N. DeMaria, professor of medicine at the University of California, San Diego. What’s going on here? he asked.
“I think it’s a fundamental truth that a lot of things that look good in preclinical work, even when backed up by a lot of solid science, don’t pan out in human studies,” Dr. Bhatt replied. “That’s a challenge, and probably in no other arena more so than in tackling inflammation and antioxidant therapy.
“There’s a graveyard of compounds that have not worked, and now we’ve perhaps added another one,” Dr. Bhatt continued. “But it doesn’t mean that scientific inquiry isn’t important, because I think eventually we’ll have drugs for these problems, whether it’s reperfusion injury or contrast-induced nephropathy. It’ll probably just take a lot more time and effort.”
The one solace regarding the CARIN trial, in Dr. Bhatt’s view, is that it highlighted the advantages of what is known as an adaptive trial design. Instead of jumping from positive early-phase results straight to a definitive 10,000-patient phase III clinical trial, investigators were able to obtain answers regarding the drug’s ability to prevent two major problems in patients undergoing coronary angiography – contrast-induced nephropathy and major adverse cardiac events – by means of a single 361-patient trial that was comparatively inexpensive.
Acute kidney injury secondary to exposure to contrast agents remains a significant problem, with an incidence of 20%-25% in high-risk patients. Numerous proposed prophylactic agents have ultimately proved not useful, including sodium bicarbonate, N-acetylcysteine, and intravenous fenoldopam.
Indeed, the only preventive measures of proven effectiveness are hydration with saline for 12 hours preangioplasty, and limiting the volume of contrast agent used. In real-world clinical practice, however, it’s often impractical to administer the optimal 12 hours of saline because of hospital pressure to get patients out quickly, Dr. Bhatt observed.
“There remains an important unmet clinical need to find agents that reduce the occurrence of contrast nephropathy,” he stressed.
Ischemix funded the CARIN trial. Dr. Bhatt reported receiving a research grant from the company that was directed to Brigham and Women’s Hospital.
CHICAGO – CMX-2043, a novel agent intended for prevention of contrast-induced nephropathy, failed in the phase II, double-blind, placebo-controlled CARIN clinical trial presented at the annual meeting of the American College of Cardiology.
The drug had also shown promise in small preliminary studies for the prevention of periprocedural myocardial infarction in patients undergoing coronary stenting. There again, however, CMX-2043 – a derivative of alpha lipoic acid with antioxidant and cell membrane–stabilizing properties – proved ineffective in the 361-patient, 31-center phase II trial, reported Dr. Deepak L. Bhatt, professor of medicine at Harvard Medical School and executive director of interventional cardiovascular programs at Brigham and Women’s Hospital, both in Boston.
All participants in CARIN had baseline severe impairment of kidney function or mild to moderate renal impairment plus another risk factor, such as diabetes or age greater than 75 years. One hour prior to coronary angiography, they received various doses of CMX-2043 or placebo.
Unfortunately, no difference between the four treatment arms was present in terms of the primary study endpoint: the incidence of acute kidney injury as defined by at least a 0.3 mg/dL rise in serum creatinine from baseline on day 4. No dose response to CMX-2043 was evident, nor did the investigational agent have any impact on the risk of major adverse cardiovascular events.
Immediately prior to Dr. Bhatt’s presentation, Dr. Michelle L. O’Donoghue of Brigham and Women’s Hospital presented the equally negative results of the LATITUDE-TIMI 60 trial, a phase III trial of the investigational mitogen-activated protein kinase inhibitor losmapimod, a drug developed to improve outcomes in patients with an acute coronary syndrome.
“It’s a bit distressing” to witness back to back presentations of clinical trials that proved resoundingly negative despite very strong-looking preliminary data, commented discussant Dr. Anthony N. DeMaria, professor of medicine at the University of California, San Diego. What’s going on here? he asked.
“I think it’s a fundamental truth that a lot of things that look good in preclinical work, even when backed up by a lot of solid science, don’t pan out in human studies,” Dr. Bhatt replied. “That’s a challenge, and probably in no other arena more so than in tackling inflammation and antioxidant therapy.
“There’s a graveyard of compounds that have not worked, and now we’ve perhaps added another one,” Dr. Bhatt continued. “But it doesn’t mean that scientific inquiry isn’t important, because I think eventually we’ll have drugs for these problems, whether it’s reperfusion injury or contrast-induced nephropathy. It’ll probably just take a lot more time and effort.”
The one solace regarding the CARIN trial, in Dr. Bhatt’s view, is that it highlighted the advantages of what is known as an adaptive trial design. Instead of jumping from positive early-phase results straight to a definitive 10,000-patient phase III clinical trial, investigators were able to obtain answers regarding the drug’s ability to prevent two major problems in patients undergoing coronary angiography – contrast-induced nephropathy and major adverse cardiac events – by means of a single 361-patient trial that was comparatively inexpensive.
Acute kidney injury secondary to exposure to contrast agents remains a significant problem, with an incidence of 20%-25% in high-risk patients. Numerous proposed prophylactic agents have ultimately proved not useful, including sodium bicarbonate, N-acetylcysteine, and intravenous fenoldopam.
Indeed, the only preventive measures of proven effectiveness are hydration with saline for 12 hours preangioplasty, and limiting the volume of contrast agent used. In real-world clinical practice, however, it’s often impractical to administer the optimal 12 hours of saline because of hospital pressure to get patients out quickly, Dr. Bhatt observed.
“There remains an important unmet clinical need to find agents that reduce the occurrence of contrast nephropathy,” he stressed.
Ischemix funded the CARIN trial. Dr. Bhatt reported receiving a research grant from the company that was directed to Brigham and Women’s Hospital.
AT ACC 16
Key clinical point: There continues to be a major unmet need for agents that reduce the risk of contrast-induced nephropathy.
Major finding: The once-promising investigational antioxidant and cell membrane stabilizer CMX-2043 proved ineffective for prevention of renal or cardiac injuries in patients undergoing coronary angiography.
Data source: This randomized, double-blind, placebo-controlled, 31-center, phase II study involved 361 patients with baseline renal impairment, all of whom were scheduled for coronary angiography.
Disclosures: Ischemix funded the study. Dr. Bhatt reported receiving a research grant from the company that was directed to Brigham and Women’s Hospital.
Early antiarrhythmic drugs boost survival in shock-refractory cardiac arrest
CHICAGO – Paramedic-administered amiodarone or lidocaine during resuscitation of patients with shock-refractory ventricular fibrillation or ventricular tachycardia of out-of-hospital cardiac arrest significantly improves survival, according to the findings of the largest-ever clinical trial of out-of-hospital cardiac arrest.
However, the survival advantage was limited to bystander-witnessed arrest. The reason for this difference in the Amiodarone, Lidocaine or Placebo Study (ALPS) is that observed arrest is a good surrogate for earlier recognition and treatment of an out-of-hospital cardiac arrest (OHCA), Dr. Peter J. Kudenchuk explained in presenting the study findings at the annual meeting of the American College of Cardiology.
“The message from this trial is that, if you give these drugs to everyone across the board with out-of-hospital shock-refractory VF/VT cardiac arrest, you’ll help those who can be helped and you won’t hurt those who are beyond help,” said Dr. Kudenchuk, professor of medicine at the University of Washington, Seattle.
ALPS was a randomized, blinded, placebo-controlled clinical trial of 3,026 patients with shock-refractory VF/VT OHCA at 10 U.S. and Canadian sites participating in the Resuscitation Outcomes Consortium. Subjects were randomized to paramedic-administered treatment with prefilled syringes of amiodarone, lidocaine, or placebo. Time to treatment averaged 19 minutes from the initial call made to emergency services.
The primary endpoint in ALPS was survival to hospital discharge. Rates were 24.4% in the amiodarone group, 23.7% with lidocaine, and 21% with placebo. Differences in survival rates between the antiarrhythmic drug and placebo groups approached but did not achieve statistical significance.
Survival to hospital discharge in the 1,934 participants with bystander-witnessed arrest was a prespecified secondary endpoint. That outcome was achieved in 27.7% of the amiodarone group and 27.8% who got lidocaine, compared with 22.7% of placebo-treated patients. Those differences were statistically significant and clinically meaningful, Dr. Kudenchuk asserted.
“Though these differences – an absolute 5% improvement over placebo – may seem small, were we to implement this as policy, upwards of 1,800 more lives could potentially be saved each year in the United States alone,” said Dr. Kudenchuk, an electrophysiologist and cardiologist.
Bystander-witnessed OHCA was 2.3-fold more common than unwitnessed arrest. In the unwitnessed arrest subgroup there was no hint of benefit for either amiodarone or lidocaine.
“Many patients with unwitnessed arrest have already sustained mortal ischemic damage by the time they’re found,” he observed. “If you go into a morgue and give the best drug in the world, you’re not going to save anybody.”
Moreover, among the roughly 5% of patients whose OHCA was witnessed by EMS personnel, survival to hospital discharge was a whopping absolute 22% greater with antiarrhythmic drug therapy than with placebo.
“Taken together, these findings suggest that treatment sooner after heart collapse may be a critical determinant of drug effect,” Dr. Kudenchuk continued.
He said the ALPS findings are generalizable to all communities across North America where the local EMS system follows the Resuscitation Outcomes Consortium philosophy that early defibrillation and good CPR are the cornerstones of effective management of OHCA, without which no treatment can be effective.
Current use of these drugs across the United States is not standardized. “It is really a free-for-all,” according to Dr. Kudenchuk. “Some agencies strictly use lidocaine, others may use amiodarone. Some use both. And some use neither. I think in part that’s because current guideline recommendations give these drugs a class IIb recommendation – meaning they’re optional – because up until this point there have been no data to support their effectiveness in changing outcome.”
In his view, the ALPS data clearly warrant upgrading the strength of the recommendation for antiarrhythmic drug therapy in the next iteration of the guidelines. Although he is on the guideline committee, Dr. Kudenchuk added, he cannot predict what the committee as a whole will decide.
ALPS will not lead to a change in practice such that paramedic-administered antiarrhythmic agents are given only to patients with witnessed arrest, Dr. Kudenchuk said. It’s not practical for rescue personnel in the midst of the fray to try to figure out whether an OHCA was witnessed or not. Plus, there’s an ethical issue involved.
“If we’d wanted to hit the headlines with a major trial with a positive outcome we would have selected only people with witnessed cardiac arrest from the get-go to do this trial, since we guessed that’s where the money was going to be. The reality is you can’t treat people that way. Everyone has to have a chance,” he said.
Asked which antiarrhythmic drug the next edition of the resuscitation guidelines should recommend preferentially, he said ALPS wasn’t powered to distinguish between amiodarone and lidocaine. “If I were writing the guidelines, I would simply say either or both happens to be okay.”
An important footnote is that ALPS utilized a new, Food and Drug Administration–approved formulation of amiodarone, known as Nexterone, designed to reduce hypotensive effects. Had investigators employed the more familiar version of the drug, the safety results wouldn’t have been as good.
Out-of-hospital cardiac arrest accounts for roughly 350,000 deaths per year in the United States
Simultaneously with Dr. Kudenchuk’s presentation of the ALPS findings at ACC 16 in Chicago, the results were published online (N Engl J Med. 2016 Apr 4; doi: 10.1056/NEJMoa1514204).
He reported having no financial conflicts regarding the ALPS study, which was funded by the National Heart, Lung, and Blood Institute, the Canadian Institutes of Health Research, the American Heart Association, the U.S. Army, and Defense Research and Development Canada.
CHICAGO – Paramedic-administered amiodarone or lidocaine during resuscitation of patients with shock-refractory ventricular fibrillation or ventricular tachycardia of out-of-hospital cardiac arrest significantly improves survival, according to the findings of the largest-ever clinical trial of out-of-hospital cardiac arrest.
However, the survival advantage was limited to bystander-witnessed arrest. The reason for this difference in the Amiodarone, Lidocaine or Placebo Study (ALPS) is that observed arrest is a good surrogate for earlier recognition and treatment of an out-of-hospital cardiac arrest (OHCA), Dr. Peter J. Kudenchuk explained in presenting the study findings at the annual meeting of the American College of Cardiology.
“The message from this trial is that, if you give these drugs to everyone across the board with out-of-hospital shock-refractory VF/VT cardiac arrest, you’ll help those who can be helped and you won’t hurt those who are beyond help,” said Dr. Kudenchuk, professor of medicine at the University of Washington, Seattle.
ALPS was a randomized, blinded, placebo-controlled clinical trial of 3,026 patients with shock-refractory VF/VT OHCA at 10 U.S. and Canadian sites participating in the Resuscitation Outcomes Consortium. Subjects were randomized to paramedic-administered treatment with prefilled syringes of amiodarone, lidocaine, or placebo. Time to treatment averaged 19 minutes from the initial call made to emergency services.
The primary endpoint in ALPS was survival to hospital discharge. Rates were 24.4% in the amiodarone group, 23.7% with lidocaine, and 21% with placebo. Differences in survival rates between the antiarrhythmic drug and placebo groups approached but did not achieve statistical significance.
Survival to hospital discharge in the 1,934 participants with bystander-witnessed arrest was a prespecified secondary endpoint. That outcome was achieved in 27.7% of the amiodarone group and 27.8% who got lidocaine, compared with 22.7% of placebo-treated patients. Those differences were statistically significant and clinically meaningful, Dr. Kudenchuk asserted.
“Though these differences – an absolute 5% improvement over placebo – may seem small, were we to implement this as policy, upwards of 1,800 more lives could potentially be saved each year in the United States alone,” said Dr. Kudenchuk, an electrophysiologist and cardiologist.
Bystander-witnessed OHCA was 2.3-fold more common than unwitnessed arrest. In the unwitnessed arrest subgroup there was no hint of benefit for either amiodarone or lidocaine.
“Many patients with unwitnessed arrest have already sustained mortal ischemic damage by the time they’re found,” he observed. “If you go into a morgue and give the best drug in the world, you’re not going to save anybody.”
Moreover, among the roughly 5% of patients whose OHCA was witnessed by EMS personnel, survival to hospital discharge was a whopping absolute 22% greater with antiarrhythmic drug therapy than with placebo.
“Taken together, these findings suggest that treatment sooner after heart collapse may be a critical determinant of drug effect,” Dr. Kudenchuk continued.
He said the ALPS findings are generalizable to all communities across North America where the local EMS system follows the Resuscitation Outcomes Consortium philosophy that early defibrillation and good CPR are the cornerstones of effective management of OHCA, without which no treatment can be effective.
Current use of these drugs across the United States is not standardized. “It is really a free-for-all,” according to Dr. Kudenchuk. “Some agencies strictly use lidocaine, others may use amiodarone. Some use both. And some use neither. I think in part that’s because current guideline recommendations give these drugs a class IIb recommendation – meaning they’re optional – because up until this point there have been no data to support their effectiveness in changing outcome.”
In his view, the ALPS data clearly warrant upgrading the strength of the recommendation for antiarrhythmic drug therapy in the next iteration of the guidelines. Although he is on the guideline committee, Dr. Kudenchuk added, he cannot predict what the committee as a whole will decide.
ALPS will not lead to a change in practice such that paramedic-administered antiarrhythmic agents are given only to patients with witnessed arrest, Dr. Kudenchuk said. It’s not practical for rescue personnel in the midst of the fray to try to figure out whether an OHCA was witnessed or not. Plus, there’s an ethical issue involved.
“If we’d wanted to hit the headlines with a major trial with a positive outcome we would have selected only people with witnessed cardiac arrest from the get-go to do this trial, since we guessed that’s where the money was going to be. The reality is you can’t treat people that way. Everyone has to have a chance,” he said.
Asked which antiarrhythmic drug the next edition of the resuscitation guidelines should recommend preferentially, he said ALPS wasn’t powered to distinguish between amiodarone and lidocaine. “If I were writing the guidelines, I would simply say either or both happens to be okay.”
An important footnote is that ALPS utilized a new, Food and Drug Administration–approved formulation of amiodarone, known as Nexterone, designed to reduce hypotensive effects. Had investigators employed the more familiar version of the drug, the safety results wouldn’t have been as good.
Out-of-hospital cardiac arrest accounts for roughly 350,000 deaths per year in the United States
Simultaneously with Dr. Kudenchuk’s presentation of the ALPS findings at ACC 16 in Chicago, the results were published online (N Engl J Med. 2016 Apr 4; doi: 10.1056/NEJMoa1514204).
He reported having no financial conflicts regarding the ALPS study, which was funded by the National Heart, Lung, and Blood Institute, the Canadian Institutes of Health Research, the American Heart Association, the U.S. Army, and Defense Research and Development Canada.
CHICAGO – Paramedic-administered amiodarone or lidocaine during resuscitation of patients with shock-refractory ventricular fibrillation or ventricular tachycardia of out-of-hospital cardiac arrest significantly improves survival, according to the findings of the largest-ever clinical trial of out-of-hospital cardiac arrest.
However, the survival advantage was limited to bystander-witnessed arrest. The reason for this difference in the Amiodarone, Lidocaine or Placebo Study (ALPS) is that observed arrest is a good surrogate for earlier recognition and treatment of an out-of-hospital cardiac arrest (OHCA), Dr. Peter J. Kudenchuk explained in presenting the study findings at the annual meeting of the American College of Cardiology.
“The message from this trial is that, if you give these drugs to everyone across the board with out-of-hospital shock-refractory VF/VT cardiac arrest, you’ll help those who can be helped and you won’t hurt those who are beyond help,” said Dr. Kudenchuk, professor of medicine at the University of Washington, Seattle.
ALPS was a randomized, blinded, placebo-controlled clinical trial of 3,026 patients with shock-refractory VF/VT OHCA at 10 U.S. and Canadian sites participating in the Resuscitation Outcomes Consortium. Subjects were randomized to paramedic-administered treatment with prefilled syringes of amiodarone, lidocaine, or placebo. Time to treatment averaged 19 minutes from the initial call made to emergency services.
The primary endpoint in ALPS was survival to hospital discharge. Rates were 24.4% in the amiodarone group, 23.7% with lidocaine, and 21% with placebo. Differences in survival rates between the antiarrhythmic drug and placebo groups approached but did not achieve statistical significance.
Survival to hospital discharge in the 1,934 participants with bystander-witnessed arrest was a prespecified secondary endpoint. That outcome was achieved in 27.7% of the amiodarone group and 27.8% who got lidocaine, compared with 22.7% of placebo-treated patients. Those differences were statistically significant and clinically meaningful, Dr. Kudenchuk asserted.
“Though these differences – an absolute 5% improvement over placebo – may seem small, were we to implement this as policy, upwards of 1,800 more lives could potentially be saved each year in the United States alone,” said Dr. Kudenchuk, an electrophysiologist and cardiologist.
Bystander-witnessed OHCA was 2.3-fold more common than unwitnessed arrest. In the unwitnessed arrest subgroup there was no hint of benefit for either amiodarone or lidocaine.
“Many patients with unwitnessed arrest have already sustained mortal ischemic damage by the time they’re found,” he observed. “If you go into a morgue and give the best drug in the world, you’re not going to save anybody.”
Moreover, among the roughly 5% of patients whose OHCA was witnessed by EMS personnel, survival to hospital discharge was a whopping absolute 22% greater with antiarrhythmic drug therapy than with placebo.
“Taken together, these findings suggest that treatment sooner after heart collapse may be a critical determinant of drug effect,” Dr. Kudenchuk continued.
He said the ALPS findings are generalizable to all communities across North America where the local EMS system follows the Resuscitation Outcomes Consortium philosophy that early defibrillation and good CPR are the cornerstones of effective management of OHCA, without which no treatment can be effective.
Current use of these drugs across the United States is not standardized. “It is really a free-for-all,” according to Dr. Kudenchuk. “Some agencies strictly use lidocaine, others may use amiodarone. Some use both. And some use neither. I think in part that’s because current guideline recommendations give these drugs a class IIb recommendation – meaning they’re optional – because up until this point there have been no data to support their effectiveness in changing outcome.”
In his view, the ALPS data clearly warrant upgrading the strength of the recommendation for antiarrhythmic drug therapy in the next iteration of the guidelines. Although he is on the guideline committee, Dr. Kudenchuk added, he cannot predict what the committee as a whole will decide.
ALPS will not lead to a change in practice such that paramedic-administered antiarrhythmic agents are given only to patients with witnessed arrest, Dr. Kudenchuk said. It’s not practical for rescue personnel in the midst of the fray to try to figure out whether an OHCA was witnessed or not. Plus, there’s an ethical issue involved.
“If we’d wanted to hit the headlines with a major trial with a positive outcome we would have selected only people with witnessed cardiac arrest from the get-go to do this trial, since we guessed that’s where the money was going to be. The reality is you can’t treat people that way. Everyone has to have a chance,” he said.
Asked which antiarrhythmic drug the next edition of the resuscitation guidelines should recommend preferentially, he said ALPS wasn’t powered to distinguish between amiodarone and lidocaine. “If I were writing the guidelines, I would simply say either or both happens to be okay.”
An important footnote is that ALPS utilized a new, Food and Drug Administration–approved formulation of amiodarone, known as Nexterone, designed to reduce hypotensive effects. Had investigators employed the more familiar version of the drug, the safety results wouldn’t have been as good.
Out-of-hospital cardiac arrest accounts for roughly 350,000 deaths per year in the United States
Simultaneously with Dr. Kudenchuk’s presentation of the ALPS findings at ACC 16 in Chicago, the results were published online (N Engl J Med. 2016 Apr 4; doi: 10.1056/NEJMoa1514204).
He reported having no financial conflicts regarding the ALPS study, which was funded by the National Heart, Lung, and Blood Institute, the Canadian Institutes of Health Research, the American Heart Association, the U.S. Army, and Defense Research and Development Canada.
AT ACC 16
Key clinical point: Routine use of injectable amiodarone or lidocaine by paramedics in cases of shock-refractory VF/VT out-of-hospital cardiac arrest would save at least 1,800 lives per year in the U.S.
Major finding: Survival to hospital discharge was an absolute 5% greater when such patients got the antiarrhythmic compared with placebo.
Data source: A blinded, randomized, placebo-controlled clinical trial of 3,026 subjects at 10 sites.
Disclosures: The National Heart, Lung, and Blood Institute, the Canadian Institutes of Health Research, the American Heart Association, the U.S. Army, and Defense Research and Development Canada funded the ALPS trial. The presenter reported having no financial conflicts of interest.
Beware HIPAA pitfalls in emailing patients
WAIKOLOA, HAWAII – Emailing patients must be done with care in order to avoid potentially treacherous medicolegal problems under the Health Insurance Portability and Accountability Act, Dr. Whitney A. High said at the Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
And as for exchanging conventional texts with patients, colleagues, or staff, forget about it. Texting is a flat out terrible idea if the message includes anything defined under HIPAA as protected health information – and HIPAA’s definition is surprisingly inclusive, according to Dr. High, director of dermatopathology at the University of Colorado and a nonpracticing attorney who follows medicolegal issues closely. HIPAA is no longer the toothless paper tiger it was in the early years after the legislation’s passage. The HITECH Act of 2009 and the HIPAA Final Omnibus Rule of 2013 beefed up the financial penalties for HIPAA violations and allowed state attorneys general to enforce the law, not just federal officials. The revisions also reversed the burden of proof for breaches of HIPAA such that when protected health information gets mishandled, it’s automatically assumed that harm resulted. It’s up to the physician or health plan to prove otherwise.
For example, when an employee at a New England dermatology practice lost a flash drive containing before-and-after images from several thousand Mohs micrographic surgery procedures, the practice was fined $250,000 under HIPAA, even though there was no indication that the missing information had ever been published or misused in any way, Dr. High noted.
Under HIPAA, health information is protected if it’s identifiable. And the patient’s name needn’t be mentioned for that to be the case. A birth date, Social Security number, email address, medical record number, account number, website address, vehicle identifier, full-face photo, image of an unusual tattoo or birthmark – that’s all potentially identifiable and therefore protected information.
Dr. High recalled that while in law school, he had a professor who used to say that if you want your pants to stay up, the surest way is to use both a belt and suspenders.
“I’m a belt and suspenders guy. I practice dermatology that way,” he said by way of explaining his own cautious, multisafeguard approach to medicolegal self-protection in the Internet age.
“Unencrypted email is a morass,” he cautioned. It’s vulnerable to compromise at multiple points during a message’s transmission, receipt, and storage on a server. For that reason, a physician who emails patients should use email that’s encrypted “at rest”, meaning the server is encrypted and a Business Associate Agreement exists with the email provider. Even so, if the patient’s email is not encrypted there is the possibility of unauthorized access by a third party, so it’s advisable to obtain a written acceptance of this risk from the patient before responding to any medical questions the patient might pose electronically.
Dr. High and other providers in the University of Colorado system utilize a “patient portal” built into the group’s website. They can send encrypted emails to patients there. An alert about the message is sent to the patient’s unencrypted email, so the patient can go to the portal and read the secure email after creating an account and logging on.
Other secure options for email include RelayHealth (www.relayhealth.com), Pretty Good Privacy (www.pgp.com), Neocertified (www.neocertified.com), and Zixmail (www.zixcorp.com), although Dr. High noted he has no personal experience with any of those companies.
Sending out appointment reminder emails to patients without their permission in an effort to cut down on no-shows is a HIPAA violation. The correct, HIPAA-approved way to do this is to proactively get the patients’ written permission to receive such reminders, along with the email address where they’d like them to be sent and their acknowledgment that the reminder message could be intercepted if their email isn’t encrypted. That’s the belt-and-suspenders approach, Dr. High noted.
Most text messaging systems do not provide encryption. Thus, it’s not possible to safely send protected health information by text message. The only way to do so is to purchase the use of a pseudo–text messaging service such as Tiger Text (www.tigertext.com), QliqSOFT (www.qliqsoft.com), or Spok (www.spok.com), the dermatologist continued.
It’s important to understand that patients aren’t covered by HIPAA. They can email their physician with photos of a skin lesion, information about a change in their symptoms, or anything else without consequences under HIPAA. But if the photos or other data provided electronically by the patient are used in medical decision making, then that email must get incorporated into the electronic health record. Like the rest of the EHR, that email needs to be kept for 10 years and must be available on demand. In the event of a malpractice allegation, a physician will be on shaky ground if he says he based his medical decision on information provided in a patient’s email which no longer exists, Dr. High said.
He reported having no financial conflicts of interest regarding his presentation.
SDEF and this news organization are owned by the same parent company.
WAIKOLOA, HAWAII – Emailing patients must be done with care in order to avoid potentially treacherous medicolegal problems under the Health Insurance Portability and Accountability Act, Dr. Whitney A. High said at the Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
And as for exchanging conventional texts with patients, colleagues, or staff, forget about it. Texting is a flat out terrible idea if the message includes anything defined under HIPAA as protected health information – and HIPAA’s definition is surprisingly inclusive, according to Dr. High, director of dermatopathology at the University of Colorado and a nonpracticing attorney who follows medicolegal issues closely. HIPAA is no longer the toothless paper tiger it was in the early years after the legislation’s passage. The HITECH Act of 2009 and the HIPAA Final Omnibus Rule of 2013 beefed up the financial penalties for HIPAA violations and allowed state attorneys general to enforce the law, not just federal officials. The revisions also reversed the burden of proof for breaches of HIPAA such that when protected health information gets mishandled, it’s automatically assumed that harm resulted. It’s up to the physician or health plan to prove otherwise.
For example, when an employee at a New England dermatology practice lost a flash drive containing before-and-after images from several thousand Mohs micrographic surgery procedures, the practice was fined $250,000 under HIPAA, even though there was no indication that the missing information had ever been published or misused in any way, Dr. High noted.
Under HIPAA, health information is protected if it’s identifiable. And the patient’s name needn’t be mentioned for that to be the case. A birth date, Social Security number, email address, medical record number, account number, website address, vehicle identifier, full-face photo, image of an unusual tattoo or birthmark – that’s all potentially identifiable and therefore protected information.
Dr. High recalled that while in law school, he had a professor who used to say that if you want your pants to stay up, the surest way is to use both a belt and suspenders.
“I’m a belt and suspenders guy. I practice dermatology that way,” he said by way of explaining his own cautious, multisafeguard approach to medicolegal self-protection in the Internet age.
“Unencrypted email is a morass,” he cautioned. It’s vulnerable to compromise at multiple points during a message’s transmission, receipt, and storage on a server. For that reason, a physician who emails patients should use email that’s encrypted “at rest”, meaning the server is encrypted and a Business Associate Agreement exists with the email provider. Even so, if the patient’s email is not encrypted there is the possibility of unauthorized access by a third party, so it’s advisable to obtain a written acceptance of this risk from the patient before responding to any medical questions the patient might pose electronically.
Dr. High and other providers in the University of Colorado system utilize a “patient portal” built into the group’s website. They can send encrypted emails to patients there. An alert about the message is sent to the patient’s unencrypted email, so the patient can go to the portal and read the secure email after creating an account and logging on.
Other secure options for email include RelayHealth (www.relayhealth.com), Pretty Good Privacy (www.pgp.com), Neocertified (www.neocertified.com), and Zixmail (www.zixcorp.com), although Dr. High noted he has no personal experience with any of those companies.
Sending out appointment reminder emails to patients without their permission in an effort to cut down on no-shows is a HIPAA violation. The correct, HIPAA-approved way to do this is to proactively get the patients’ written permission to receive such reminders, along with the email address where they’d like them to be sent and their acknowledgment that the reminder message could be intercepted if their email isn’t encrypted. That’s the belt-and-suspenders approach, Dr. High noted.
Most text messaging systems do not provide encryption. Thus, it’s not possible to safely send protected health information by text message. The only way to do so is to purchase the use of a pseudo–text messaging service such as Tiger Text (www.tigertext.com), QliqSOFT (www.qliqsoft.com), or Spok (www.spok.com), the dermatologist continued.
It’s important to understand that patients aren’t covered by HIPAA. They can email their physician with photos of a skin lesion, information about a change in their symptoms, or anything else without consequences under HIPAA. But if the photos or other data provided electronically by the patient are used in medical decision making, then that email must get incorporated into the electronic health record. Like the rest of the EHR, that email needs to be kept for 10 years and must be available on demand. In the event of a malpractice allegation, a physician will be on shaky ground if he says he based his medical decision on information provided in a patient’s email which no longer exists, Dr. High said.
He reported having no financial conflicts of interest regarding his presentation.
SDEF and this news organization are owned by the same parent company.
WAIKOLOA, HAWAII – Emailing patients must be done with care in order to avoid potentially treacherous medicolegal problems under the Health Insurance Portability and Accountability Act, Dr. Whitney A. High said at the Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
And as for exchanging conventional texts with patients, colleagues, or staff, forget about it. Texting is a flat out terrible idea if the message includes anything defined under HIPAA as protected health information – and HIPAA’s definition is surprisingly inclusive, according to Dr. High, director of dermatopathology at the University of Colorado and a nonpracticing attorney who follows medicolegal issues closely. HIPAA is no longer the toothless paper tiger it was in the early years after the legislation’s passage. The HITECH Act of 2009 and the HIPAA Final Omnibus Rule of 2013 beefed up the financial penalties for HIPAA violations and allowed state attorneys general to enforce the law, not just federal officials. The revisions also reversed the burden of proof for breaches of HIPAA such that when protected health information gets mishandled, it’s automatically assumed that harm resulted. It’s up to the physician or health plan to prove otherwise.
For example, when an employee at a New England dermatology practice lost a flash drive containing before-and-after images from several thousand Mohs micrographic surgery procedures, the practice was fined $250,000 under HIPAA, even though there was no indication that the missing information had ever been published or misused in any way, Dr. High noted.
Under HIPAA, health information is protected if it’s identifiable. And the patient’s name needn’t be mentioned for that to be the case. A birth date, Social Security number, email address, medical record number, account number, website address, vehicle identifier, full-face photo, image of an unusual tattoo or birthmark – that’s all potentially identifiable and therefore protected information.
Dr. High recalled that while in law school, he had a professor who used to say that if you want your pants to stay up, the surest way is to use both a belt and suspenders.
“I’m a belt and suspenders guy. I practice dermatology that way,” he said by way of explaining his own cautious, multisafeguard approach to medicolegal self-protection in the Internet age.
“Unencrypted email is a morass,” he cautioned. It’s vulnerable to compromise at multiple points during a message’s transmission, receipt, and storage on a server. For that reason, a physician who emails patients should use email that’s encrypted “at rest”, meaning the server is encrypted and a Business Associate Agreement exists with the email provider. Even so, if the patient’s email is not encrypted there is the possibility of unauthorized access by a third party, so it’s advisable to obtain a written acceptance of this risk from the patient before responding to any medical questions the patient might pose electronically.
Dr. High and other providers in the University of Colorado system utilize a “patient portal” built into the group’s website. They can send encrypted emails to patients there. An alert about the message is sent to the patient’s unencrypted email, so the patient can go to the portal and read the secure email after creating an account and logging on.
Other secure options for email include RelayHealth (www.relayhealth.com), Pretty Good Privacy (www.pgp.com), Neocertified (www.neocertified.com), and Zixmail (www.zixcorp.com), although Dr. High noted he has no personal experience with any of those companies.
Sending out appointment reminder emails to patients without their permission in an effort to cut down on no-shows is a HIPAA violation. The correct, HIPAA-approved way to do this is to proactively get the patients’ written permission to receive such reminders, along with the email address where they’d like them to be sent and their acknowledgment that the reminder message could be intercepted if their email isn’t encrypted. That’s the belt-and-suspenders approach, Dr. High noted.
Most text messaging systems do not provide encryption. Thus, it’s not possible to safely send protected health information by text message. The only way to do so is to purchase the use of a pseudo–text messaging service such as Tiger Text (www.tigertext.com), QliqSOFT (www.qliqsoft.com), or Spok (www.spok.com), the dermatologist continued.
It’s important to understand that patients aren’t covered by HIPAA. They can email their physician with photos of a skin lesion, information about a change in their symptoms, or anything else without consequences under HIPAA. But if the photos or other data provided electronically by the patient are used in medical decision making, then that email must get incorporated into the electronic health record. Like the rest of the EHR, that email needs to be kept for 10 years and must be available on demand. In the event of a malpractice allegation, a physician will be on shaky ground if he says he based his medical decision on information provided in a patient’s email which no longer exists, Dr. High said.
He reported having no financial conflicts of interest regarding his presentation.
SDEF and this news organization are owned by the same parent company.
EXPERT ANALYSIS FROM SDEF HAWAII DERMATOLOGY SEMINAR
FIRE AND ICE trial called a win for cryoablation of AF
CHICAGO – The largest-ever randomized trial of catheter ablation of atrial fibrillation has ended in a draw between radiofrequency and cryoballoon ablation in safety and efficacy – and that actually represents a win for cryoablation, a simpler and far more easily mastered procedure, Dr. Karl-Heinz Kuck said at the annual meeting of the American College of Cardiology.
“We can teach physicians how to do cryoablation much more easily. That will allow more patients with atrial fibrillation to get access to catheter ablation, which is what we really need,” according to Dr. Kuck, principal investigator in the poetically named FIRE AND ICE trial and head of cardiology at St. Georg Hospital in Hamburg (Germany).
FIRE AND ICE included 769 patients in eight European countries. The participants, all of whom had antiarrhythmic drug–refractory paroxysmal atrial fibrillation (AF), were randomized to radiofrequency ablation – the long-time standard – or to cryoablation, a newer technology. Radiofrequency ablation was guided by three-dimensional electroanatomic mapping, while cryoablation utilized fluoroscopic guidance.
The primary efficacy endpoint was the 1-year rate of clinical failure, defined as an occurrence of AF, atrial flutter, or atrial tachycardia lasting for at least 30 seconds, or repeat ablation or the use of antiarrhythmic drugs following a 90-day postprocedural blanking period. The clinical failure rate was 34.6% in the cryoballoon group and similar at 35.9% in the radiofrequency group.
Serious treatment-related adverse events occurred in 10.2% of the cryoballoon group and 12.8% of the radiofrequency group, a nonsignificant difference. No procedural deaths occurred in the study.
There were, however, several significant procedural differences. Procedure time averaged 124 minutes in the cryoablation group, nearly 20 minutes less than the 142 minutes for radiofrequency ablation. However, the 17-minute fluoroscopy time in the radiofrequency group was 5 minutes shorter than for cryoablation.
Dr. Kuck said the study underestimates the true procedural differences because FIRE AND ICE was carried out by extremely experienced operators. In routine clinical practice involving non-elite operators, it’s not unusual for radiofrequency ablation fluoroscopy times to be two or even three times longer than the 17 minutes seen in the study. Plus, FIRE AND ICE was conducted when the procedure entailed two applications of the cryoballoon. Now only one application is recommended, cutting an additional 12 minutes off the total procedure time, he added.
Radiofrequency ablation takes longer because it entails creating a series of point-to-point lesions in a circle to isolate the pulmonary veins. With cryoablation, the balloon is moved into position, inflated, and a 3-minute-freeze is administered to create a circle of necrotic tissue in a single-step procedure.
Discussant Dr. Hugh G. Calkins praised the FIRE AND ICE investigators’ use of a rigorous definition of recurrence that required as little as a 30-second episode of atrial arrhythmia.
“That’s a very high bar, so I think the results are very impressive,” said Dr. Calkins, professor of medicine and of pediatrics and director of the cardiac arrhythmia service at Johns Hopkins University, Baltimore.
He commented that “this study is a clear reminder that 90% success rates just don’t happen in this field,” despite what some practitioners have claimed.
Asked how he predicts the study results will influence the field of AF ablation, Dr. Kuck replied that he foresees much wider adoption of cryoablation and a stronger endorsement of the technology in updated guideline recommendations.
“I personally believe this will be the most important development in our field in the next several years,” he added.
The electrophysiologist noted that even though current guidelines give a class Ia recommendation to catheter ablation of paroxysmal AF that’s refractory to at least one antiarrhythmic drug, at present only 4% of such patients actually undergo the procedure.
“Having just 4% of patients with AF undergo catheter ablation cannot be what we are looking for as physicians,” Dr. Kuck said. “I believe if we want to roll out catheter ablation for AF, we need simple and safe tools. This trial elegantly shows that with a simpler device that allows single-shot isolation of the pulmonary veins, we can get the same safety and efficacy as with radiofrequency ablation. I often tell people that radiofrequency ablation of atrial fibrillation is the most challenging procedure in all cardiology. We do this procedure from the groin in a moving heart. It’s a very complex technology.”
His dream, he continued, is that cryoablation will eventually enable patients with atrial fibrillation to be managed the same way electrophysiologists treat patients with Wolff-Parkinson-White syndrome; with the first episode, the patient goes to the electrophysiology catheterization lab for an ablation procedure.
“I think there’s a great message here: The cryoballoon will move catheter ablation from a niche procedure performed in specialized centers by the few guys in the world who can do it really well out into the broader world. To do that you need a tool that is safe, simple, and can be handled by the average doctor,” Dr. Kuck said.
Discussant Dr. Anthony DeMaria commented that it would be premature at this point to start thinking about cryoablation as a first approach to new-onset AF, given the roughly 35% clinical failure rate at 1 year seen in FIRE AND ICE. That rate doubtless would have been even higher had patients been equipped with implantable loop recorders, added Dr. DeMaria, professor of medicine at the University of California, San Diego.
Dr. Kuck conceded that the high recurrence rate is one of the great unsolved limitations of catheter ablation of AF.
“We don’t know how to get the pulmonary veins permanently isolated,” he said. “We can create acute lesions, but over time what we’ve seen is recovery of tissue and then reconduction by the pulmonary veins. I believe that 20% of the 40% recurrence rate is due to reconduction from the pulmonary veins, and the rest is probably due to triggers coming from other sites.”
The FIRE AND ICE trial was funded in part by Medtronic, which markets the Arctic Front Advance cryoablation catheter used in the study. Dr. Kuck reported serving on a speakers’ bureau for Medtronic and acting as a consultant to Biosense Webster, Edwards, and St. Jude.
Simultaneous with Dr. Kuck’s presentation at ACC 16, the results of FIRE AND ICE were published online (N Engl J Med. 2016 Apr 4. doi: 10.1056/NEJMoa1602014).
CHICAGO – The largest-ever randomized trial of catheter ablation of atrial fibrillation has ended in a draw between radiofrequency and cryoballoon ablation in safety and efficacy – and that actually represents a win for cryoablation, a simpler and far more easily mastered procedure, Dr. Karl-Heinz Kuck said at the annual meeting of the American College of Cardiology.
“We can teach physicians how to do cryoablation much more easily. That will allow more patients with atrial fibrillation to get access to catheter ablation, which is what we really need,” according to Dr. Kuck, principal investigator in the poetically named FIRE AND ICE trial and head of cardiology at St. Georg Hospital in Hamburg (Germany).
FIRE AND ICE included 769 patients in eight European countries. The participants, all of whom had antiarrhythmic drug–refractory paroxysmal atrial fibrillation (AF), were randomized to radiofrequency ablation – the long-time standard – or to cryoablation, a newer technology. Radiofrequency ablation was guided by three-dimensional electroanatomic mapping, while cryoablation utilized fluoroscopic guidance.
The primary efficacy endpoint was the 1-year rate of clinical failure, defined as an occurrence of AF, atrial flutter, or atrial tachycardia lasting for at least 30 seconds, or repeat ablation or the use of antiarrhythmic drugs following a 90-day postprocedural blanking period. The clinical failure rate was 34.6% in the cryoballoon group and similar at 35.9% in the radiofrequency group.
Serious treatment-related adverse events occurred in 10.2% of the cryoballoon group and 12.8% of the radiofrequency group, a nonsignificant difference. No procedural deaths occurred in the study.
There were, however, several significant procedural differences. Procedure time averaged 124 minutes in the cryoablation group, nearly 20 minutes less than the 142 minutes for radiofrequency ablation. However, the 17-minute fluoroscopy time in the radiofrequency group was 5 minutes shorter than for cryoablation.
Dr. Kuck said the study underestimates the true procedural differences because FIRE AND ICE was carried out by extremely experienced operators. In routine clinical practice involving non-elite operators, it’s not unusual for radiofrequency ablation fluoroscopy times to be two or even three times longer than the 17 minutes seen in the study. Plus, FIRE AND ICE was conducted when the procedure entailed two applications of the cryoballoon. Now only one application is recommended, cutting an additional 12 minutes off the total procedure time, he added.
Radiofrequency ablation takes longer because it entails creating a series of point-to-point lesions in a circle to isolate the pulmonary veins. With cryoablation, the balloon is moved into position, inflated, and a 3-minute-freeze is administered to create a circle of necrotic tissue in a single-step procedure.
Discussant Dr. Hugh G. Calkins praised the FIRE AND ICE investigators’ use of a rigorous definition of recurrence that required as little as a 30-second episode of atrial arrhythmia.
“That’s a very high bar, so I think the results are very impressive,” said Dr. Calkins, professor of medicine and of pediatrics and director of the cardiac arrhythmia service at Johns Hopkins University, Baltimore.
He commented that “this study is a clear reminder that 90% success rates just don’t happen in this field,” despite what some practitioners have claimed.
Asked how he predicts the study results will influence the field of AF ablation, Dr. Kuck replied that he foresees much wider adoption of cryoablation and a stronger endorsement of the technology in updated guideline recommendations.
“I personally believe this will be the most important development in our field in the next several years,” he added.
The electrophysiologist noted that even though current guidelines give a class Ia recommendation to catheter ablation of paroxysmal AF that’s refractory to at least one antiarrhythmic drug, at present only 4% of such patients actually undergo the procedure.
“Having just 4% of patients with AF undergo catheter ablation cannot be what we are looking for as physicians,” Dr. Kuck said. “I believe if we want to roll out catheter ablation for AF, we need simple and safe tools. This trial elegantly shows that with a simpler device that allows single-shot isolation of the pulmonary veins, we can get the same safety and efficacy as with radiofrequency ablation. I often tell people that radiofrequency ablation of atrial fibrillation is the most challenging procedure in all cardiology. We do this procedure from the groin in a moving heart. It’s a very complex technology.”
His dream, he continued, is that cryoablation will eventually enable patients with atrial fibrillation to be managed the same way electrophysiologists treat patients with Wolff-Parkinson-White syndrome; with the first episode, the patient goes to the electrophysiology catheterization lab for an ablation procedure.
“I think there’s a great message here: The cryoballoon will move catheter ablation from a niche procedure performed in specialized centers by the few guys in the world who can do it really well out into the broader world. To do that you need a tool that is safe, simple, and can be handled by the average doctor,” Dr. Kuck said.
Discussant Dr. Anthony DeMaria commented that it would be premature at this point to start thinking about cryoablation as a first approach to new-onset AF, given the roughly 35% clinical failure rate at 1 year seen in FIRE AND ICE. That rate doubtless would have been even higher had patients been equipped with implantable loop recorders, added Dr. DeMaria, professor of medicine at the University of California, San Diego.
Dr. Kuck conceded that the high recurrence rate is one of the great unsolved limitations of catheter ablation of AF.
“We don’t know how to get the pulmonary veins permanently isolated,” he said. “We can create acute lesions, but over time what we’ve seen is recovery of tissue and then reconduction by the pulmonary veins. I believe that 20% of the 40% recurrence rate is due to reconduction from the pulmonary veins, and the rest is probably due to triggers coming from other sites.”
The FIRE AND ICE trial was funded in part by Medtronic, which markets the Arctic Front Advance cryoablation catheter used in the study. Dr. Kuck reported serving on a speakers’ bureau for Medtronic and acting as a consultant to Biosense Webster, Edwards, and St. Jude.
Simultaneous with Dr. Kuck’s presentation at ACC 16, the results of FIRE AND ICE were published online (N Engl J Med. 2016 Apr 4. doi: 10.1056/NEJMoa1602014).
CHICAGO – The largest-ever randomized trial of catheter ablation of atrial fibrillation has ended in a draw between radiofrequency and cryoballoon ablation in safety and efficacy – and that actually represents a win for cryoablation, a simpler and far more easily mastered procedure, Dr. Karl-Heinz Kuck said at the annual meeting of the American College of Cardiology.
“We can teach physicians how to do cryoablation much more easily. That will allow more patients with atrial fibrillation to get access to catheter ablation, which is what we really need,” according to Dr. Kuck, principal investigator in the poetically named FIRE AND ICE trial and head of cardiology at St. Georg Hospital in Hamburg (Germany).
FIRE AND ICE included 769 patients in eight European countries. The participants, all of whom had antiarrhythmic drug–refractory paroxysmal atrial fibrillation (AF), were randomized to radiofrequency ablation – the long-time standard – or to cryoablation, a newer technology. Radiofrequency ablation was guided by three-dimensional electroanatomic mapping, while cryoablation utilized fluoroscopic guidance.
The primary efficacy endpoint was the 1-year rate of clinical failure, defined as an occurrence of AF, atrial flutter, or atrial tachycardia lasting for at least 30 seconds, or repeat ablation or the use of antiarrhythmic drugs following a 90-day postprocedural blanking period. The clinical failure rate was 34.6% in the cryoballoon group and similar at 35.9% in the radiofrequency group.
Serious treatment-related adverse events occurred in 10.2% of the cryoballoon group and 12.8% of the radiofrequency group, a nonsignificant difference. No procedural deaths occurred in the study.
There were, however, several significant procedural differences. Procedure time averaged 124 minutes in the cryoablation group, nearly 20 minutes less than the 142 minutes for radiofrequency ablation. However, the 17-minute fluoroscopy time in the radiofrequency group was 5 minutes shorter than for cryoablation.
Dr. Kuck said the study underestimates the true procedural differences because FIRE AND ICE was carried out by extremely experienced operators. In routine clinical practice involving non-elite operators, it’s not unusual for radiofrequency ablation fluoroscopy times to be two or even three times longer than the 17 minutes seen in the study. Plus, FIRE AND ICE was conducted when the procedure entailed two applications of the cryoballoon. Now only one application is recommended, cutting an additional 12 minutes off the total procedure time, he added.
Radiofrequency ablation takes longer because it entails creating a series of point-to-point lesions in a circle to isolate the pulmonary veins. With cryoablation, the balloon is moved into position, inflated, and a 3-minute-freeze is administered to create a circle of necrotic tissue in a single-step procedure.
Discussant Dr. Hugh G. Calkins praised the FIRE AND ICE investigators’ use of a rigorous definition of recurrence that required as little as a 30-second episode of atrial arrhythmia.
“That’s a very high bar, so I think the results are very impressive,” said Dr. Calkins, professor of medicine and of pediatrics and director of the cardiac arrhythmia service at Johns Hopkins University, Baltimore.
He commented that “this study is a clear reminder that 90% success rates just don’t happen in this field,” despite what some practitioners have claimed.
Asked how he predicts the study results will influence the field of AF ablation, Dr. Kuck replied that he foresees much wider adoption of cryoablation and a stronger endorsement of the technology in updated guideline recommendations.
“I personally believe this will be the most important development in our field in the next several years,” he added.
The electrophysiologist noted that even though current guidelines give a class Ia recommendation to catheter ablation of paroxysmal AF that’s refractory to at least one antiarrhythmic drug, at present only 4% of such patients actually undergo the procedure.
“Having just 4% of patients with AF undergo catheter ablation cannot be what we are looking for as physicians,” Dr. Kuck said. “I believe if we want to roll out catheter ablation for AF, we need simple and safe tools. This trial elegantly shows that with a simpler device that allows single-shot isolation of the pulmonary veins, we can get the same safety and efficacy as with radiofrequency ablation. I often tell people that radiofrequency ablation of atrial fibrillation is the most challenging procedure in all cardiology. We do this procedure from the groin in a moving heart. It’s a very complex technology.”
His dream, he continued, is that cryoablation will eventually enable patients with atrial fibrillation to be managed the same way electrophysiologists treat patients with Wolff-Parkinson-White syndrome; with the first episode, the patient goes to the electrophysiology catheterization lab for an ablation procedure.
“I think there’s a great message here: The cryoballoon will move catheter ablation from a niche procedure performed in specialized centers by the few guys in the world who can do it really well out into the broader world. To do that you need a tool that is safe, simple, and can be handled by the average doctor,” Dr. Kuck said.
Discussant Dr. Anthony DeMaria commented that it would be premature at this point to start thinking about cryoablation as a first approach to new-onset AF, given the roughly 35% clinical failure rate at 1 year seen in FIRE AND ICE. That rate doubtless would have been even higher had patients been equipped with implantable loop recorders, added Dr. DeMaria, professor of medicine at the University of California, San Diego.
Dr. Kuck conceded that the high recurrence rate is one of the great unsolved limitations of catheter ablation of AF.
“We don’t know how to get the pulmonary veins permanently isolated,” he said. “We can create acute lesions, but over time what we’ve seen is recovery of tissue and then reconduction by the pulmonary veins. I believe that 20% of the 40% recurrence rate is due to reconduction from the pulmonary veins, and the rest is probably due to triggers coming from other sites.”
The FIRE AND ICE trial was funded in part by Medtronic, which markets the Arctic Front Advance cryoablation catheter used in the study. Dr. Kuck reported serving on a speakers’ bureau for Medtronic and acting as a consultant to Biosense Webster, Edwards, and St. Jude.
Simultaneous with Dr. Kuck’s presentation at ACC 16, the results of FIRE AND ICE were published online (N Engl J Med. 2016 Apr 4. doi: 10.1056/NEJMoa1602014).
AT ACC 16
Key clinical point: Cryoablation of atrial fibrillation offers significant advantages over radiofrequency ablation.
Major finding: In a rigorous randomized trial, cryoablation of atrial fibrillation had a 34.6% clinical failure rate at 1 year, similar to the 35.9% rate for radiofrequency ablation.
Data source: The FIRE AND ICE trial, which randomized 769 patients with paroxysmal atrial fibrillation in eight European countries.
Disclosures: FIRE AND ICE was funded in part by Medtronic. The presenter reported serving on a speakers’ bureau for the company.
Similarities seen in rate and rhythm control for postsurgical AF
CHICAGO – Rate and rhythm control proved equally effective for treatment of new-onset post–cardiac surgery atrial fibrillation in a randomized trial that was far and away the largest ever to examine the best way to address this common and costly arrhythmia, Dr. A. Marc Gillinov said at the annual meeting of the American College of Cardiology.
Thus, either strategy is acceptable. That being said, rate control gets the edge as the initial treatment strategy because it avoids the considerable toxicities accompanying amiodarone for rhythm control, most of which arise only after patients have been discharged from the hospital. In contrast, when rate control doesn’t work, it becomes evident while the patient is still in the hospital, according to Dr. Gillinov, a cardiothoracic surgeon at the Cleveland Clinic .
Atrial fibrillation (AF) is the most common complication of cardiac surgery, with an incidence variously reported at 20%-50%. It results in lengthier hospital stays, greater cost of care, and increased risks of mortality, stroke, heart failure, and infection. Postoperative AF adds an estimated $1 billion per year to health care costs in the United States.
While current ACC/AHA/Heart Rhythm Society joint guidelines recommend rate control with a beta-blocker as first-line therapy for patients with this postoperative complication, with a class I, level-of-evidence A rating, upon closer inspection the evidence cited mainly involves extrapolation from studies looking at how to prevent postoperative AF. Because no persuasive evidence existed as to how best to treat this common and economically and medically costly condition, Dr. Gillinov and his coinvestigators in the National Institutes of Health–funded Cardiothoracic Surgical Trials Network carried out a randomized trial 10-fold larger than anything prior.
The 23-site study included 2,109 patients enrolled prior to cardiac surgery, of whom 40% underwent isolated coronary artery bypass grafting (CABG) while the other 60% had valve surgery, either alone or with CABG. These proportions reflect current cardiac surgery treatment patterns nationally. Overall, 33% of the cardiac surgery patients experienced postoperative AF. The incidence was 28% in patients who underwent isolated CABG but rose with increasing surgical complexity to nearly 50% in patients who had combined CABG and valve operations. The average time to onset of postoperative AF was 2.4 days.
A total of 523 patients with postoperative AF were randomized to rate or rhythm control. Rate control most often entailed use of a beta-blocker, while amiodarone was prescribed for rhythm control.
The primary endpoint in the trial was a measure of health care resource utilization: total days in hospital during a 60-day period starting from the time of randomization. This endpoint was a draw: a median of 5.1 days with rate control and 5.0 days with rhythm control.
At hospital discharge, 89.9% of patients in the rate control group and 93.5% in the rhythm control group had a stable heart rhythm without AF. From discharge to 60 days, 84.2% of patients in the rate control group and a similar 86.9% of the rhythm control group remained free of AF.
Rates of serious adverse events were similar in the two groups: 24.8 per 100 patient-months in the rate control arm and 26.4 per 100 patient-months in the rhythm control arm. Three patients in the rate control arm died during the 60-day study period, and two died in the rhythm control group.
Of note, roughly one-quarter of patients in each study arm crossed over to the other arm. In the rate control group, this was typically due to drug ineffectiveness, while in the rhythm control arm the switch was most often made in response to amiodarone side effects.
Roughly 43% of patients in each group were placed on anticoagulation with warfarin for 60 days according to study protocol, which called for such action if a patient remained in AF 48 hours after randomization.
There were five strokes, one case of transient ischemic attack, and four noncerebral thromboembolisms. Also, 21 bleeding events occurred, 17 of which were classified as serious; 90% of the bleeding events happened in patients on warfarin.
“I found the results very striking and very reassuring,” said discussant Hugh G. Calkins. “To me, the clinical message is clearly that rate control is the preference.”
It was troubling, however, to see that 10 thromboembolic events occurred in 523 patients over the course of just 60 days. “Should we be anticoagulating these postsurgical atrial fibrillation patients a lot more frequently?” asked Dr. Calkins, professor of medicine and of pediatrics and director of the cardiac arrhythmia service at Johns Hopkins University, Baltimore.
Dr. Gillinov replied that he and his colleagues in the Cardiothoracic Surgical Trials Network consider that to be the key remaining question regarding postoperative AF. They are now planning a clinical trial aimed at finding the optimal balance between stroke protection via anticoagulation and bleeding risk.
The National Institutes of Health and the Canadian Institutes of Health Research funded the work. Dr. Gillinov reported serving as a consultant to five surgical device companies, none of which played any role in the study.
Simultaneously with Dr. Gillinov’s presentation at ACC 16, the study results were published in the New England Journal of Medicine (doi: 10.1056/NEJMoa1602002).
CHICAGO – Rate and rhythm control proved equally effective for treatment of new-onset post–cardiac surgery atrial fibrillation in a randomized trial that was far and away the largest ever to examine the best way to address this common and costly arrhythmia, Dr. A. Marc Gillinov said at the annual meeting of the American College of Cardiology.
Thus, either strategy is acceptable. That being said, rate control gets the edge as the initial treatment strategy because it avoids the considerable toxicities accompanying amiodarone for rhythm control, most of which arise only after patients have been discharged from the hospital. In contrast, when rate control doesn’t work, it becomes evident while the patient is still in the hospital, according to Dr. Gillinov, a cardiothoracic surgeon at the Cleveland Clinic .
Atrial fibrillation (AF) is the most common complication of cardiac surgery, with an incidence variously reported at 20%-50%. It results in lengthier hospital stays, greater cost of care, and increased risks of mortality, stroke, heart failure, and infection. Postoperative AF adds an estimated $1 billion per year to health care costs in the United States.
While current ACC/AHA/Heart Rhythm Society joint guidelines recommend rate control with a beta-blocker as first-line therapy for patients with this postoperative complication, with a class I, level-of-evidence A rating, upon closer inspection the evidence cited mainly involves extrapolation from studies looking at how to prevent postoperative AF. Because no persuasive evidence existed as to how best to treat this common and economically and medically costly condition, Dr. Gillinov and his coinvestigators in the National Institutes of Health–funded Cardiothoracic Surgical Trials Network carried out a randomized trial 10-fold larger than anything prior.
The 23-site study included 2,109 patients enrolled prior to cardiac surgery, of whom 40% underwent isolated coronary artery bypass grafting (CABG) while the other 60% had valve surgery, either alone or with CABG. These proportions reflect current cardiac surgery treatment patterns nationally. Overall, 33% of the cardiac surgery patients experienced postoperative AF. The incidence was 28% in patients who underwent isolated CABG but rose with increasing surgical complexity to nearly 50% in patients who had combined CABG and valve operations. The average time to onset of postoperative AF was 2.4 days.
A total of 523 patients with postoperative AF were randomized to rate or rhythm control. Rate control most often entailed use of a beta-blocker, while amiodarone was prescribed for rhythm control.
The primary endpoint in the trial was a measure of health care resource utilization: total days in hospital during a 60-day period starting from the time of randomization. This endpoint was a draw: a median of 5.1 days with rate control and 5.0 days with rhythm control.
At hospital discharge, 89.9% of patients in the rate control group and 93.5% in the rhythm control group had a stable heart rhythm without AF. From discharge to 60 days, 84.2% of patients in the rate control group and a similar 86.9% of the rhythm control group remained free of AF.
Rates of serious adverse events were similar in the two groups: 24.8 per 100 patient-months in the rate control arm and 26.4 per 100 patient-months in the rhythm control arm. Three patients in the rate control arm died during the 60-day study period, and two died in the rhythm control group.
Of note, roughly one-quarter of patients in each study arm crossed over to the other arm. In the rate control group, this was typically due to drug ineffectiveness, while in the rhythm control arm the switch was most often made in response to amiodarone side effects.
Roughly 43% of patients in each group were placed on anticoagulation with warfarin for 60 days according to study protocol, which called for such action if a patient remained in AF 48 hours after randomization.
There were five strokes, one case of transient ischemic attack, and four noncerebral thromboembolisms. Also, 21 bleeding events occurred, 17 of which were classified as serious; 90% of the bleeding events happened in patients on warfarin.
“I found the results very striking and very reassuring,” said discussant Hugh G. Calkins. “To me, the clinical message is clearly that rate control is the preference.”
It was troubling, however, to see that 10 thromboembolic events occurred in 523 patients over the course of just 60 days. “Should we be anticoagulating these postsurgical atrial fibrillation patients a lot more frequently?” asked Dr. Calkins, professor of medicine and of pediatrics and director of the cardiac arrhythmia service at Johns Hopkins University, Baltimore.
Dr. Gillinov replied that he and his colleagues in the Cardiothoracic Surgical Trials Network consider that to be the key remaining question regarding postoperative AF. They are now planning a clinical trial aimed at finding the optimal balance between stroke protection via anticoagulation and bleeding risk.
The National Institutes of Health and the Canadian Institutes of Health Research funded the work. Dr. Gillinov reported serving as a consultant to five surgical device companies, none of which played any role in the study.
Simultaneously with Dr. Gillinov’s presentation at ACC 16, the study results were published in the New England Journal of Medicine (doi: 10.1056/NEJMoa1602002).
CHICAGO – Rate and rhythm control proved equally effective for treatment of new-onset post–cardiac surgery atrial fibrillation in a randomized trial that was far and away the largest ever to examine the best way to address this common and costly arrhythmia, Dr. A. Marc Gillinov said at the annual meeting of the American College of Cardiology.
Thus, either strategy is acceptable. That being said, rate control gets the edge as the initial treatment strategy because it avoids the considerable toxicities accompanying amiodarone for rhythm control, most of which arise only after patients have been discharged from the hospital. In contrast, when rate control doesn’t work, it becomes evident while the patient is still in the hospital, according to Dr. Gillinov, a cardiothoracic surgeon at the Cleveland Clinic .
Atrial fibrillation (AF) is the most common complication of cardiac surgery, with an incidence variously reported at 20%-50%. It results in lengthier hospital stays, greater cost of care, and increased risks of mortality, stroke, heart failure, and infection. Postoperative AF adds an estimated $1 billion per year to health care costs in the United States.
While current ACC/AHA/Heart Rhythm Society joint guidelines recommend rate control with a beta-blocker as first-line therapy for patients with this postoperative complication, with a class I, level-of-evidence A rating, upon closer inspection the evidence cited mainly involves extrapolation from studies looking at how to prevent postoperative AF. Because no persuasive evidence existed as to how best to treat this common and economically and medically costly condition, Dr. Gillinov and his coinvestigators in the National Institutes of Health–funded Cardiothoracic Surgical Trials Network carried out a randomized trial 10-fold larger than anything prior.
The 23-site study included 2,109 patients enrolled prior to cardiac surgery, of whom 40% underwent isolated coronary artery bypass grafting (CABG) while the other 60% had valve surgery, either alone or with CABG. These proportions reflect current cardiac surgery treatment patterns nationally. Overall, 33% of the cardiac surgery patients experienced postoperative AF. The incidence was 28% in patients who underwent isolated CABG but rose with increasing surgical complexity to nearly 50% in patients who had combined CABG and valve operations. The average time to onset of postoperative AF was 2.4 days.
A total of 523 patients with postoperative AF were randomized to rate or rhythm control. Rate control most often entailed use of a beta-blocker, while amiodarone was prescribed for rhythm control.
The primary endpoint in the trial was a measure of health care resource utilization: total days in hospital during a 60-day period starting from the time of randomization. This endpoint was a draw: a median of 5.1 days with rate control and 5.0 days with rhythm control.
At hospital discharge, 89.9% of patients in the rate control group and 93.5% in the rhythm control group had a stable heart rhythm without AF. From discharge to 60 days, 84.2% of patients in the rate control group and a similar 86.9% of the rhythm control group remained free of AF.
Rates of serious adverse events were similar in the two groups: 24.8 per 100 patient-months in the rate control arm and 26.4 per 100 patient-months in the rhythm control arm. Three patients in the rate control arm died during the 60-day study period, and two died in the rhythm control group.
Of note, roughly one-quarter of patients in each study arm crossed over to the other arm. In the rate control group, this was typically due to drug ineffectiveness, while in the rhythm control arm the switch was most often made in response to amiodarone side effects.
Roughly 43% of patients in each group were placed on anticoagulation with warfarin for 60 days according to study protocol, which called for such action if a patient remained in AF 48 hours after randomization.
There were five strokes, one case of transient ischemic attack, and four noncerebral thromboembolisms. Also, 21 bleeding events occurred, 17 of which were classified as serious; 90% of the bleeding events happened in patients on warfarin.
“I found the results very striking and very reassuring,” said discussant Hugh G. Calkins. “To me, the clinical message is clearly that rate control is the preference.”
It was troubling, however, to see that 10 thromboembolic events occurred in 523 patients over the course of just 60 days. “Should we be anticoagulating these postsurgical atrial fibrillation patients a lot more frequently?” asked Dr. Calkins, professor of medicine and of pediatrics and director of the cardiac arrhythmia service at Johns Hopkins University, Baltimore.
Dr. Gillinov replied that he and his colleagues in the Cardiothoracic Surgical Trials Network consider that to be the key remaining question regarding postoperative AF. They are now planning a clinical trial aimed at finding the optimal balance between stroke protection via anticoagulation and bleeding risk.
The National Institutes of Health and the Canadian Institutes of Health Research funded the work. Dr. Gillinov reported serving as a consultant to five surgical device companies, none of which played any role in the study.
Simultaneously with Dr. Gillinov’s presentation at ACC 16, the study results were published in the New England Journal of Medicine (doi: 10.1056/NEJMoa1602002).
AT ACC 16
Key clinical point: Rate control offers the advantage of simplicity over a rhythm control strategy in new-onset atrial fibrillation after cardiac surgery.
Major finding: Rate and rhythm control strategies for treatment of new-onset atrial fibrillation after cardiac surgery resulted in equal numbers of hospital days, similar serious complication rates, and low rates of persistent atrial fibrillation at 60 days of follow-up.
Data source: A randomized clinical trial of 523 patients with new-onset atrial fibrillation following cardiac surgery at 23 U.S. and Canadian academic medical centers.
Disclosures: The study was funded by the National Institutes of Health and the Canadian Institutes of Health Research and carried out through the Cardiothoracic Surgical Trials Network. The presenter reported having no relevant financial interests.
Recently Identified Eczema Comorbidities Include Anemia, Obesity
WAIKOLOA, HAWAII – The list of nonatopic comorbid conditions associated with atopic dermatitis is rapidly expanding.
Just in the past year, published studies have linked pediatric atopic dermatitis to increased risks of obesity, high blood pressure, headaches, anemia, and speech disorders. Meanwhile, adult atopic dermatitis was reported to be associated with increased rates of fracture and cardiovascular disease. And in the dermatologic arena, a link between atopic dermatitis, vitiligo, and alopecia areata was identified, Dr. Lawrence F. Eichenfield noted at the Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
Actually, these reported associations published in 2015-2016 might best be termed “emerging comorbidities,” as they are first reports and thus need confirmation, said Dr. Eichenfield, professor of dermatology and pediatrics at the University of California, San Diego, and chief of pediatric and adolescent dermatology at Rady Children’s Hospital San Diego.
Much of this work on emerging comorbidities has been done by dermatologist Dr. Jonathan I. Silverberg of Northwestern University, Chicago, and various coinvestigators. They have been prolific.
For example, in a multivariate logistic regression analysis of 207,007 children and adolescents included in the cross-sectional 1997-2013 U.S. National Health Interview Survey, Dr. Silverberg and coinvestigators found that eczema was independently associated with a 1.83-fold increased odds of anemia. They bolstered this observation with an analysis of more than 30,000 children and adolescents in the 1992-2012 National Health and Nutrition Examination Survey (NHANES) in which they found that current eczema was associated with a 1.93-fold increased odds of anemia, particularly microcytic anemia. The underlying mechanism is unknown; however, the investigators noted that chronic inflammation and systemic immunosuppressant drugs have been shown to be associated with anemia (JAMA Pediatr. 2016;170[1]:29-34).
Dr. Silverberg also found in a multivariate logistic regression analysis of data on more than 400,000 pediatric participants in the National Survey of Children’s Health and the National Health Interview Survey that mild and severe eczema were independently associated with 1.79-fold and 2.72-fold, respectively, increased odds of headaches (J Allergy Clin Immunol. 2016 Feb;137[2]:492-9).
Using these same two data sources, with multivariate analysis adjusted for potential confounders, the investigators found that eczema was associated with a 1.81-fold increased risk of speech disorder (J Pediatr. 2016 Jan;168:185-92).
In a case-control study involving 132 children and adolescents with current moderate to severe atopic dermatitis and 143 healthy controls, Dr. Silverberg and coinvestigators found in a logistic regression analysis that atopic dermatitis was independently associated with a doubled risk of having a systolic blood pressure in the 90th percentile or higher – as well as 3.92-fold increased odds of central obesity, as defined by a waist circumference in the 85th percentile or higher (JAMA Dermatol. 2015 Feb;151[2]:144-52).
In a systematic review and meta-analysis of 16 published vitiligo studies and 17 published studies of alopecia areata, Dr. Silverberg and medical student Girish C. Mohan found that patients with vitiligo or alopecia areata were respectively 7.8 and 2.6 times more likely to have atopic dermatitis than controls without those disorders (JAMA Dermatol. 2015 May;151[5]:522-8).
In a logistic regression analysis of data on 34,500 adults with a history of eczema within the prior year who participated in the 2012 National Health Interview Survey, Drs. Nitin Garg and Dr. Silverberg concluded that the results suggested that adult atopic dermatitis is a previously unrecognized risk factor for fracture and other bone or joint injuries causing limitation. Adults with atopic dermatitis were at a 1.67-fold increased risk for such injuries in an analysis controlling for sociodemographics, other forms of atopic disease, and psychiatric and behavioral disorders (JAMA Dermatol. 2015 Jan;151[1]:33-41).
In another large cross-sectional study, Dr. Silverberg found that adults with atopic dermatitis had significantly higher odds of a history of acute MI, coronary artery disease, heart failure, and stroke (Allergy. 2015 Oct;70[10]:1300-8).
Dr. Eichenfield said that while he would like to see these hot-off-the-presses 2015-2016 findings on nonatopic comorbidities backed up by confirmatory studies in other populations, the evidence is stronger for an association between pediatric atopic dermatitis and several mental health disorders. The initial reports came mainly from Europe, but were then supported by a large study by Dr. Eric L. Simpson and coinvestigators at Oregon Health and Science University, Portland.
In their analysis of data on nearly 93,000 noninstitutionalized children and adolescents included in the 2007 National Survey of Children’s Health, the investigators found after controlling for potential confounders that atopic dermatitis was associated with a 1.87-fold increased risk of having attention-deficit/hyperactivity disorder, a 3-fold increased risk of diagnosed autism, an adjusted 1.81-fold increase in depression, and 1.87-fold increased odds of conduct disorder. The Oregon group found a clear dose-dependent relationship between the reported severity of the skin disease and the likelihood of those mental health disorders (J Allergy Clin Immunol. 2013 Feb;131[2]:428-33).
The hope is that emerging strategies to prevent atopic dermatitis or aggressively treat it early on will reduce the risk of many of these comorbid conditions, Dr. Eichenfield said.
SDEF and this news organization are owned by the same parent company.
WAIKOLOA, HAWAII – The list of nonatopic comorbid conditions associated with atopic dermatitis is rapidly expanding.
Just in the past year, published studies have linked pediatric atopic dermatitis to increased risks of obesity, high blood pressure, headaches, anemia, and speech disorders. Meanwhile, adult atopic dermatitis was reported to be associated with increased rates of fracture and cardiovascular disease. And in the dermatologic arena, a link between atopic dermatitis, vitiligo, and alopecia areata was identified, Dr. Lawrence F. Eichenfield noted at the Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
Actually, these reported associations published in 2015-2016 might best be termed “emerging comorbidities,” as they are first reports and thus need confirmation, said Dr. Eichenfield, professor of dermatology and pediatrics at the University of California, San Diego, and chief of pediatric and adolescent dermatology at Rady Children’s Hospital San Diego.
Much of this work on emerging comorbidities has been done by dermatologist Dr. Jonathan I. Silverberg of Northwestern University, Chicago, and various coinvestigators. They have been prolific.
For example, in a multivariate logistic regression analysis of 207,007 children and adolescents included in the cross-sectional 1997-2013 U.S. National Health Interview Survey, Dr. Silverberg and coinvestigators found that eczema was independently associated with a 1.83-fold increased odds of anemia. They bolstered this observation with an analysis of more than 30,000 children and adolescents in the 1992-2012 National Health and Nutrition Examination Survey (NHANES) in which they found that current eczema was associated with a 1.93-fold increased odds of anemia, particularly microcytic anemia. The underlying mechanism is unknown; however, the investigators noted that chronic inflammation and systemic immunosuppressant drugs have been shown to be associated with anemia (JAMA Pediatr. 2016;170[1]:29-34).
Dr. Silverberg also found in a multivariate logistic regression analysis of data on more than 400,000 pediatric participants in the National Survey of Children’s Health and the National Health Interview Survey that mild and severe eczema were independently associated with 1.79-fold and 2.72-fold, respectively, increased odds of headaches (J Allergy Clin Immunol. 2016 Feb;137[2]:492-9).
Using these same two data sources, with multivariate analysis adjusted for potential confounders, the investigators found that eczema was associated with a 1.81-fold increased risk of speech disorder (J Pediatr. 2016 Jan;168:185-92).
In a case-control study involving 132 children and adolescents with current moderate to severe atopic dermatitis and 143 healthy controls, Dr. Silverberg and coinvestigators found in a logistic regression analysis that atopic dermatitis was independently associated with a doubled risk of having a systolic blood pressure in the 90th percentile or higher – as well as 3.92-fold increased odds of central obesity, as defined by a waist circumference in the 85th percentile or higher (JAMA Dermatol. 2015 Feb;151[2]:144-52).
In a systematic review and meta-analysis of 16 published vitiligo studies and 17 published studies of alopecia areata, Dr. Silverberg and medical student Girish C. Mohan found that patients with vitiligo or alopecia areata were respectively 7.8 and 2.6 times more likely to have atopic dermatitis than controls without those disorders (JAMA Dermatol. 2015 May;151[5]:522-8).
In a logistic regression analysis of data on 34,500 adults with a history of eczema within the prior year who participated in the 2012 National Health Interview Survey, Drs. Nitin Garg and Dr. Silverberg concluded that the results suggested that adult atopic dermatitis is a previously unrecognized risk factor for fracture and other bone or joint injuries causing limitation. Adults with atopic dermatitis were at a 1.67-fold increased risk for such injuries in an analysis controlling for sociodemographics, other forms of atopic disease, and psychiatric and behavioral disorders (JAMA Dermatol. 2015 Jan;151[1]:33-41).
In another large cross-sectional study, Dr. Silverberg found that adults with atopic dermatitis had significantly higher odds of a history of acute MI, coronary artery disease, heart failure, and stroke (Allergy. 2015 Oct;70[10]:1300-8).
Dr. Eichenfield said that while he would like to see these hot-off-the-presses 2015-2016 findings on nonatopic comorbidities backed up by confirmatory studies in other populations, the evidence is stronger for an association between pediatric atopic dermatitis and several mental health disorders. The initial reports came mainly from Europe, but were then supported by a large study by Dr. Eric L. Simpson and coinvestigators at Oregon Health and Science University, Portland.
In their analysis of data on nearly 93,000 noninstitutionalized children and adolescents included in the 2007 National Survey of Children’s Health, the investigators found after controlling for potential confounders that atopic dermatitis was associated with a 1.87-fold increased risk of having attention-deficit/hyperactivity disorder, a 3-fold increased risk of diagnosed autism, an adjusted 1.81-fold increase in depression, and 1.87-fold increased odds of conduct disorder. The Oregon group found a clear dose-dependent relationship between the reported severity of the skin disease and the likelihood of those mental health disorders (J Allergy Clin Immunol. 2013 Feb;131[2]:428-33).
The hope is that emerging strategies to prevent atopic dermatitis or aggressively treat it early on will reduce the risk of many of these comorbid conditions, Dr. Eichenfield said.
SDEF and this news organization are owned by the same parent company.
WAIKOLOA, HAWAII – The list of nonatopic comorbid conditions associated with atopic dermatitis is rapidly expanding.
Just in the past year, published studies have linked pediatric atopic dermatitis to increased risks of obesity, high blood pressure, headaches, anemia, and speech disorders. Meanwhile, adult atopic dermatitis was reported to be associated with increased rates of fracture and cardiovascular disease. And in the dermatologic arena, a link between atopic dermatitis, vitiligo, and alopecia areata was identified, Dr. Lawrence F. Eichenfield noted at the Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
Actually, these reported associations published in 2015-2016 might best be termed “emerging comorbidities,” as they are first reports and thus need confirmation, said Dr. Eichenfield, professor of dermatology and pediatrics at the University of California, San Diego, and chief of pediatric and adolescent dermatology at Rady Children’s Hospital San Diego.
Much of this work on emerging comorbidities has been done by dermatologist Dr. Jonathan I. Silverberg of Northwestern University, Chicago, and various coinvestigators. They have been prolific.
For example, in a multivariate logistic regression analysis of 207,007 children and adolescents included in the cross-sectional 1997-2013 U.S. National Health Interview Survey, Dr. Silverberg and coinvestigators found that eczema was independently associated with a 1.83-fold increased odds of anemia. They bolstered this observation with an analysis of more than 30,000 children and adolescents in the 1992-2012 National Health and Nutrition Examination Survey (NHANES) in which they found that current eczema was associated with a 1.93-fold increased odds of anemia, particularly microcytic anemia. The underlying mechanism is unknown; however, the investigators noted that chronic inflammation and systemic immunosuppressant drugs have been shown to be associated with anemia (JAMA Pediatr. 2016;170[1]:29-34).
Dr. Silverberg also found in a multivariate logistic regression analysis of data on more than 400,000 pediatric participants in the National Survey of Children’s Health and the National Health Interview Survey that mild and severe eczema were independently associated with 1.79-fold and 2.72-fold, respectively, increased odds of headaches (J Allergy Clin Immunol. 2016 Feb;137[2]:492-9).
Using these same two data sources, with multivariate analysis adjusted for potential confounders, the investigators found that eczema was associated with a 1.81-fold increased risk of speech disorder (J Pediatr. 2016 Jan;168:185-92).
In a case-control study involving 132 children and adolescents with current moderate to severe atopic dermatitis and 143 healthy controls, Dr. Silverberg and coinvestigators found in a logistic regression analysis that atopic dermatitis was independently associated with a doubled risk of having a systolic blood pressure in the 90th percentile or higher – as well as 3.92-fold increased odds of central obesity, as defined by a waist circumference in the 85th percentile or higher (JAMA Dermatol. 2015 Feb;151[2]:144-52).
In a systematic review and meta-analysis of 16 published vitiligo studies and 17 published studies of alopecia areata, Dr. Silverberg and medical student Girish C. Mohan found that patients with vitiligo or alopecia areata were respectively 7.8 and 2.6 times more likely to have atopic dermatitis than controls without those disorders (JAMA Dermatol. 2015 May;151[5]:522-8).
In a logistic regression analysis of data on 34,500 adults with a history of eczema within the prior year who participated in the 2012 National Health Interview Survey, Drs. Nitin Garg and Dr. Silverberg concluded that the results suggested that adult atopic dermatitis is a previously unrecognized risk factor for fracture and other bone or joint injuries causing limitation. Adults with atopic dermatitis were at a 1.67-fold increased risk for such injuries in an analysis controlling for sociodemographics, other forms of atopic disease, and psychiatric and behavioral disorders (JAMA Dermatol. 2015 Jan;151[1]:33-41).
In another large cross-sectional study, Dr. Silverberg found that adults with atopic dermatitis had significantly higher odds of a history of acute MI, coronary artery disease, heart failure, and stroke (Allergy. 2015 Oct;70[10]:1300-8).
Dr. Eichenfield said that while he would like to see these hot-off-the-presses 2015-2016 findings on nonatopic comorbidities backed up by confirmatory studies in other populations, the evidence is stronger for an association between pediatric atopic dermatitis and several mental health disorders. The initial reports came mainly from Europe, but were then supported by a large study by Dr. Eric L. Simpson and coinvestigators at Oregon Health and Science University, Portland.
In their analysis of data on nearly 93,000 noninstitutionalized children and adolescents included in the 2007 National Survey of Children’s Health, the investigators found after controlling for potential confounders that atopic dermatitis was associated with a 1.87-fold increased risk of having attention-deficit/hyperactivity disorder, a 3-fold increased risk of diagnosed autism, an adjusted 1.81-fold increase in depression, and 1.87-fold increased odds of conduct disorder. The Oregon group found a clear dose-dependent relationship between the reported severity of the skin disease and the likelihood of those mental health disorders (J Allergy Clin Immunol. 2013 Feb;131[2]:428-33).
The hope is that emerging strategies to prevent atopic dermatitis or aggressively treat it early on will reduce the risk of many of these comorbid conditions, Dr. Eichenfield said.
SDEF and this news organization are owned by the same parent company.
EXPERT ANALYSIS FROM THE SDEF HAWAII DERMATOLOGY SEMINAR
Recently identified eczema comorbidities include anemia, obesity
WAIKOLOA, HAWAII – The list of nonatopic comorbid conditions associated with atopic dermatitis is rapidly expanding.
Just in the past year, published studies have linked pediatric atopic dermatitis to increased risks of obesity, high blood pressure, headaches, anemia, and speech disorders. Meanwhile, adult atopic dermatitis was reported to be associated with increased rates of fracture and cardiovascular disease. And in the dermatologic arena, a link between atopic dermatitis, vitiligo, and alopecia areata was identified, Dr. Lawrence F. Eichenfield noted at the Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
Actually, these reported associations published in 2015-2016 might best be termed “emerging comorbidities,” as they are first reports and thus need confirmation, said Dr. Eichenfield, professor of dermatology and pediatrics at the University of California, San Diego, and chief of pediatric and adolescent dermatology at Rady Children’s Hospital San Diego.
Much of this work on emerging comorbidities has been done by dermatologist Dr. Jonathan I. Silverberg of Northwestern University, Chicago, and various coinvestigators. They have been prolific.
For example, in a multivariate logistic regression analysis of 207,007 children and adolescents included in the cross-sectional 1997-2013 U.S. National Health Interview Survey, Dr. Silverberg and coinvestigators found that eczema was independently associated with a 1.83-fold increased odds of anemia. They bolstered this observation with an analysis of more than 30,000 children and adolescents in the 1992-2012 National Health and Nutrition Examination Survey (NHANES) in which they found that current eczema was associated with a 1.93-fold increased odds of anemia, particularly microcytic anemia. The underlying mechanism is unknown; however, the investigators noted that chronic inflammation and systemic immunosuppressant drugs have been shown to be associated with anemia (JAMA Pediatr. 2016;170[1]:29-34).
Dr. Silverberg also found in a multivariate logistic regression analysis of data on more than 400,000 pediatric participants in the National Survey of Children’s Health and the National Health Interview Survey that mild and severe eczema were independently associated with 1.79-fold and 2.72-fold, respectively, increased odds of headaches (J Allergy Clin Immunol. 2016 Feb;137[2]:492-9).
Using these same two data sources, with multivariate analysis adjusted for potential confounders, the investigators found that eczema was associated with a 1.81-fold increased risk of speech disorder (J Pediatr. 2016 Jan;168:185-92).
In a case-control study involving 132 children and adolescents with current moderate to severe atopic dermatitis and 143 healthy controls, Dr. Silverberg and coinvestigators found in a logistic regression analysis that atopic dermatitis was independently associated with a doubled risk of having a systolic blood pressure in the 90th percentile or higher – as well as 3.92-fold increased odds of central obesity, as defined by a waist circumference in the 85th percentile or higher (JAMA Dermatol. 2015 Feb;151[2]:144-52).
In a systematic review and meta-analysis of 16 published vitiligo studies and 17 published studies of alopecia areata, Dr. Silverberg and medical student Girish C. Mohan found that patients with vitiligo or alopecia areata were respectively 7.8 and 2.6 times more likely to have atopic dermatitis than controls without those disorders (JAMA Dermatol. 2015 May;151[5]:522-8).
In a logistic regression analysis of data on 34,500 adults with a history of eczema within the prior year who participated in the 2012 National Health Interview Survey, Drs. Nitin Garg and Dr. Silverberg concluded that the results suggested that adult atopic dermatitis is a previously unrecognized risk factor for fracture and other bone or joint injuries causing limitation. Adults with atopic dermatitis were at a 1.67-fold increased risk for such injuries in an analysis controlling for sociodemographics, other forms of atopic disease, and psychiatric and behavioral disorders (JAMA Dermatol. 2015 Jan;151[1]:33-41).
In another large cross-sectional study, Dr. Silverberg found that adults with atopic dermatitis had significantly higher odds of a history of acute MI, coronary artery disease, heart failure, and stroke (Allergy. 2015 Oct;70[10]:1300-8).
Dr. Eichenfield said that while he would like to see these hot-off-the-presses 2015-2016 findings on nonatopic comorbidities backed up by confirmatory studies in other populations, the evidence is stronger for an association between pediatric atopic dermatitis and several mental health disorders. The initial reports came mainly from Europe, but were then supported by a large study by Dr. Eric L. Simpson and coinvestigators at Oregon Health and Science University, Portland.
In their analysis of data on nearly 93,000 noninstitutionalized children and adolescents included in the 2007 National Survey of Children’s Health, the investigators found after controlling for potential confounders that atopic dermatitis was associated with a 1.87-fold increased risk of having attention-deficit/hyperactivity disorder, a 3-fold increased risk of diagnosed autism, an adjusted 1.81-fold increase in depression, and 1.87-fold increased odds of conduct disorder. The Oregon group found a clear dose-dependent relationship between the reported severity of the skin disease and the likelihood of those mental health disorders (J Allergy Clin Immunol. 2013 Feb;131[2]:428-33).
The hope is that emerging strategies to prevent atopic dermatitis or aggressively treat it early on will reduce the risk of many of these comorbid conditions, Dr. Eichenfield said.
SDEF and this news organization are owned by the same parent company.
WAIKOLOA, HAWAII – The list of nonatopic comorbid conditions associated with atopic dermatitis is rapidly expanding.
Just in the past year, published studies have linked pediatric atopic dermatitis to increased risks of obesity, high blood pressure, headaches, anemia, and speech disorders. Meanwhile, adult atopic dermatitis was reported to be associated with increased rates of fracture and cardiovascular disease. And in the dermatologic arena, a link between atopic dermatitis, vitiligo, and alopecia areata was identified, Dr. Lawrence F. Eichenfield noted at the Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
Actually, these reported associations published in 2015-2016 might best be termed “emerging comorbidities,” as they are first reports and thus need confirmation, said Dr. Eichenfield, professor of dermatology and pediatrics at the University of California, San Diego, and chief of pediatric and adolescent dermatology at Rady Children’s Hospital San Diego.
Much of this work on emerging comorbidities has been done by dermatologist Dr. Jonathan I. Silverberg of Northwestern University, Chicago, and various coinvestigators. They have been prolific.
For example, in a multivariate logistic regression analysis of 207,007 children and adolescents included in the cross-sectional 1997-2013 U.S. National Health Interview Survey, Dr. Silverberg and coinvestigators found that eczema was independently associated with a 1.83-fold increased odds of anemia. They bolstered this observation with an analysis of more than 30,000 children and adolescents in the 1992-2012 National Health and Nutrition Examination Survey (NHANES) in which they found that current eczema was associated with a 1.93-fold increased odds of anemia, particularly microcytic anemia. The underlying mechanism is unknown; however, the investigators noted that chronic inflammation and systemic immunosuppressant drugs have been shown to be associated with anemia (JAMA Pediatr. 2016;170[1]:29-34).
Dr. Silverberg also found in a multivariate logistic regression analysis of data on more than 400,000 pediatric participants in the National Survey of Children’s Health and the National Health Interview Survey that mild and severe eczema were independently associated with 1.79-fold and 2.72-fold, respectively, increased odds of headaches (J Allergy Clin Immunol. 2016 Feb;137[2]:492-9).
Using these same two data sources, with multivariate analysis adjusted for potential confounders, the investigators found that eczema was associated with a 1.81-fold increased risk of speech disorder (J Pediatr. 2016 Jan;168:185-92).
In a case-control study involving 132 children and adolescents with current moderate to severe atopic dermatitis and 143 healthy controls, Dr. Silverberg and coinvestigators found in a logistic regression analysis that atopic dermatitis was independently associated with a doubled risk of having a systolic blood pressure in the 90th percentile or higher – as well as 3.92-fold increased odds of central obesity, as defined by a waist circumference in the 85th percentile or higher (JAMA Dermatol. 2015 Feb;151[2]:144-52).
In a systematic review and meta-analysis of 16 published vitiligo studies and 17 published studies of alopecia areata, Dr. Silverberg and medical student Girish C. Mohan found that patients with vitiligo or alopecia areata were respectively 7.8 and 2.6 times more likely to have atopic dermatitis than controls without those disorders (JAMA Dermatol. 2015 May;151[5]:522-8).
In a logistic regression analysis of data on 34,500 adults with a history of eczema within the prior year who participated in the 2012 National Health Interview Survey, Drs. Nitin Garg and Dr. Silverberg concluded that the results suggested that adult atopic dermatitis is a previously unrecognized risk factor for fracture and other bone or joint injuries causing limitation. Adults with atopic dermatitis were at a 1.67-fold increased risk for such injuries in an analysis controlling for sociodemographics, other forms of atopic disease, and psychiatric and behavioral disorders (JAMA Dermatol. 2015 Jan;151[1]:33-41).
In another large cross-sectional study, Dr. Silverberg found that adults with atopic dermatitis had significantly higher odds of a history of acute MI, coronary artery disease, heart failure, and stroke (Allergy. 2015 Oct;70[10]:1300-8).
Dr. Eichenfield said that while he would like to see these hot-off-the-presses 2015-2016 findings on nonatopic comorbidities backed up by confirmatory studies in other populations, the evidence is stronger for an association between pediatric atopic dermatitis and several mental health disorders. The initial reports came mainly from Europe, but were then supported by a large study by Dr. Eric L. Simpson and coinvestigators at Oregon Health and Science University, Portland.
In their analysis of data on nearly 93,000 noninstitutionalized children and adolescents included in the 2007 National Survey of Children’s Health, the investigators found after controlling for potential confounders that atopic dermatitis was associated with a 1.87-fold increased risk of having attention-deficit/hyperactivity disorder, a 3-fold increased risk of diagnosed autism, an adjusted 1.81-fold increase in depression, and 1.87-fold increased odds of conduct disorder. The Oregon group found a clear dose-dependent relationship between the reported severity of the skin disease and the likelihood of those mental health disorders (J Allergy Clin Immunol. 2013 Feb;131[2]:428-33).
The hope is that emerging strategies to prevent atopic dermatitis or aggressively treat it early on will reduce the risk of many of these comorbid conditions, Dr. Eichenfield said.
SDEF and this news organization are owned by the same parent company.
WAIKOLOA, HAWAII – The list of nonatopic comorbid conditions associated with atopic dermatitis is rapidly expanding.
Just in the past year, published studies have linked pediatric atopic dermatitis to increased risks of obesity, high blood pressure, headaches, anemia, and speech disorders. Meanwhile, adult atopic dermatitis was reported to be associated with increased rates of fracture and cardiovascular disease. And in the dermatologic arena, a link between atopic dermatitis, vitiligo, and alopecia areata was identified, Dr. Lawrence F. Eichenfield noted at the Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
Actually, these reported associations published in 2015-2016 might best be termed “emerging comorbidities,” as they are first reports and thus need confirmation, said Dr. Eichenfield, professor of dermatology and pediatrics at the University of California, San Diego, and chief of pediatric and adolescent dermatology at Rady Children’s Hospital San Diego.
Much of this work on emerging comorbidities has been done by dermatologist Dr. Jonathan I. Silverberg of Northwestern University, Chicago, and various coinvestigators. They have been prolific.
For example, in a multivariate logistic regression analysis of 207,007 children and adolescents included in the cross-sectional 1997-2013 U.S. National Health Interview Survey, Dr. Silverberg and coinvestigators found that eczema was independently associated with a 1.83-fold increased odds of anemia. They bolstered this observation with an analysis of more than 30,000 children and adolescents in the 1992-2012 National Health and Nutrition Examination Survey (NHANES) in which they found that current eczema was associated with a 1.93-fold increased odds of anemia, particularly microcytic anemia. The underlying mechanism is unknown; however, the investigators noted that chronic inflammation and systemic immunosuppressant drugs have been shown to be associated with anemia (JAMA Pediatr. 2016;170[1]:29-34).
Dr. Silverberg also found in a multivariate logistic regression analysis of data on more than 400,000 pediatric participants in the National Survey of Children’s Health and the National Health Interview Survey that mild and severe eczema were independently associated with 1.79-fold and 2.72-fold, respectively, increased odds of headaches (J Allergy Clin Immunol. 2016 Feb;137[2]:492-9).
Using these same two data sources, with multivariate analysis adjusted for potential confounders, the investigators found that eczema was associated with a 1.81-fold increased risk of speech disorder (J Pediatr. 2016 Jan;168:185-92).
In a case-control study involving 132 children and adolescents with current moderate to severe atopic dermatitis and 143 healthy controls, Dr. Silverberg and coinvestigators found in a logistic regression analysis that atopic dermatitis was independently associated with a doubled risk of having a systolic blood pressure in the 90th percentile or higher – as well as 3.92-fold increased odds of central obesity, as defined by a waist circumference in the 85th percentile or higher (JAMA Dermatol. 2015 Feb;151[2]:144-52).
In a systematic review and meta-analysis of 16 published vitiligo studies and 17 published studies of alopecia areata, Dr. Silverberg and medical student Girish C. Mohan found that patients with vitiligo or alopecia areata were respectively 7.8 and 2.6 times more likely to have atopic dermatitis than controls without those disorders (JAMA Dermatol. 2015 May;151[5]:522-8).
In a logistic regression analysis of data on 34,500 adults with a history of eczema within the prior year who participated in the 2012 National Health Interview Survey, Drs. Nitin Garg and Dr. Silverberg concluded that the results suggested that adult atopic dermatitis is a previously unrecognized risk factor for fracture and other bone or joint injuries causing limitation. Adults with atopic dermatitis were at a 1.67-fold increased risk for such injuries in an analysis controlling for sociodemographics, other forms of atopic disease, and psychiatric and behavioral disorders (JAMA Dermatol. 2015 Jan;151[1]:33-41).
In another large cross-sectional study, Dr. Silverberg found that adults with atopic dermatitis had significantly higher odds of a history of acute MI, coronary artery disease, heart failure, and stroke (Allergy. 2015 Oct;70[10]:1300-8).
Dr. Eichenfield said that while he would like to see these hot-off-the-presses 2015-2016 findings on nonatopic comorbidities backed up by confirmatory studies in other populations, the evidence is stronger for an association between pediatric atopic dermatitis and several mental health disorders. The initial reports came mainly from Europe, but were then supported by a large study by Dr. Eric L. Simpson and coinvestigators at Oregon Health and Science University, Portland.
In their analysis of data on nearly 93,000 noninstitutionalized children and adolescents included in the 2007 National Survey of Children’s Health, the investigators found after controlling for potential confounders that atopic dermatitis was associated with a 1.87-fold increased risk of having attention-deficit/hyperactivity disorder, a 3-fold increased risk of diagnosed autism, an adjusted 1.81-fold increase in depression, and 1.87-fold increased odds of conduct disorder. The Oregon group found a clear dose-dependent relationship between the reported severity of the skin disease and the likelihood of those mental health disorders (J Allergy Clin Immunol. 2013 Feb;131[2]:428-33).
The hope is that emerging strategies to prevent atopic dermatitis or aggressively treat it early on will reduce the risk of many of these comorbid conditions, Dr. Eichenfield said.
SDEF and this news organization are owned by the same parent company.
EXPERT ANALYSIS FROM THE SDEF HAWAII DERMATOLOGY SEMINAR




















