User login
Primary arrhythmia syndromes: Common cause of pediatric sudden cardiac death
CHICAGO – Just over one-half of all sudden deaths in a large pediatric case series were due to a primary arrhythmia syndrome, Dr. Grazia Delle Donne reported at the annual meeting of the American College of Cardiology.
She presented an analysis of all patients under the age of 18 years who were referred to London’s Royal Brompton Hospital for post mortem examination following presumed sudden cardiac death during 1991-2013. Royal Brompton is a national referral center for sudden cardiac death.
The review was undertaken because sudden cardiac death in the pediatric population occurs infrequently. Little is known about the prevalence of the various causes, noted Dr. Delle Donne of Royal Brompton.
Of the 398 subjects, 266 (67%) were female. The median age at death was 14 years. Twenty-two percent of the fatalities occurred during or immediately after exercise. Thirty-nine percent occurred while at rest.
Thirty-one percent of subjects had a family history of sudden cardiac death, another 14% had a family history of cardiomyopathy, and in 5% of cases there was a significant family history of arrhythmia.
Five percent of the children were known to have congenital heart disease. Eighteen percent of the children had a history of syncope.
Investigators determined that a primary arrhythmia syndrome such as long QT or Brugada syndrome was the cause of sudden death in 54% of cases. Death was attributed to cardiomyopathy in 15% cases, congenital heart disease in 8%, myocarditis in 6%, and coronary anomalies in 5%, with miscellaneous causes accounting for the remainder.
Dr. Delle Donne reported having no financial conflicts of interest regarding her presentation.
CHICAGO – Just over one-half of all sudden deaths in a large pediatric case series were due to a primary arrhythmia syndrome, Dr. Grazia Delle Donne reported at the annual meeting of the American College of Cardiology.
She presented an analysis of all patients under the age of 18 years who were referred to London’s Royal Brompton Hospital for post mortem examination following presumed sudden cardiac death during 1991-2013. Royal Brompton is a national referral center for sudden cardiac death.
The review was undertaken because sudden cardiac death in the pediatric population occurs infrequently. Little is known about the prevalence of the various causes, noted Dr. Delle Donne of Royal Brompton.
Of the 398 subjects, 266 (67%) were female. The median age at death was 14 years. Twenty-two percent of the fatalities occurred during or immediately after exercise. Thirty-nine percent occurred while at rest.
Thirty-one percent of subjects had a family history of sudden cardiac death, another 14% had a family history of cardiomyopathy, and in 5% of cases there was a significant family history of arrhythmia.
Five percent of the children were known to have congenital heart disease. Eighteen percent of the children had a history of syncope.
Investigators determined that a primary arrhythmia syndrome such as long QT or Brugada syndrome was the cause of sudden death in 54% of cases. Death was attributed to cardiomyopathy in 15% cases, congenital heart disease in 8%, myocarditis in 6%, and coronary anomalies in 5%, with miscellaneous causes accounting for the remainder.
Dr. Delle Donne reported having no financial conflicts of interest regarding her presentation.
CHICAGO – Just over one-half of all sudden deaths in a large pediatric case series were due to a primary arrhythmia syndrome, Dr. Grazia Delle Donne reported at the annual meeting of the American College of Cardiology.
She presented an analysis of all patients under the age of 18 years who were referred to London’s Royal Brompton Hospital for post mortem examination following presumed sudden cardiac death during 1991-2013. Royal Brompton is a national referral center for sudden cardiac death.
The review was undertaken because sudden cardiac death in the pediatric population occurs infrequently. Little is known about the prevalence of the various causes, noted Dr. Delle Donne of Royal Brompton.
Of the 398 subjects, 266 (67%) were female. The median age at death was 14 years. Twenty-two percent of the fatalities occurred during or immediately after exercise. Thirty-nine percent occurred while at rest.
Thirty-one percent of subjects had a family history of sudden cardiac death, another 14% had a family history of cardiomyopathy, and in 5% of cases there was a significant family history of arrhythmia.
Five percent of the children were known to have congenital heart disease. Eighteen percent of the children had a history of syncope.
Investigators determined that a primary arrhythmia syndrome such as long QT or Brugada syndrome was the cause of sudden death in 54% of cases. Death was attributed to cardiomyopathy in 15% cases, congenital heart disease in 8%, myocarditis in 6%, and coronary anomalies in 5%, with miscellaneous causes accounting for the remainder.
Dr. Delle Donne reported having no financial conflicts of interest regarding her presentation.
AT ACC 16
Key clinical point: Primary arrhythmia syndromes accounted for most cases of sudden cardiac death in a large pediatric case series.
Major finding: Family history of sudden cardiac death was present in 31% of 398 cases.
Data source: A retrospective review of all 398 cases of sudden cardiac death in childhood referred for post mortem examination at a British center during 1991-2013.
Disclosures: Dr. Delle Donne reported having no financial conflicts of interest.
Cardiovascular consequences of extreme prematurity persist into late adolescence
CHICAGO – The abnormal arterial hemodynamics identified in 11-year-olds with an extremely preterm birth persist at age 19, according to an update from the landmark longitudinal EPICure study.
“Given the implications of these significant findings, cardiovascular monitoring and risk prevention would be highly recommended for all individuals born extremely preterm,” Dr. Joanne Beckmann said in presenting the EPICure results on the long-term consequences of extreme prematurity at the annual meeting of the American College of Cardiology.
EPICure is a longitudinal study investigating health outcomes in a national cohort of babies born extremely preterm at 22-25 weeks’ gestation in the United Kingdom during 1995-1996. It is the longest such study conducted anywhere.
“Neonatal survival at the lowest gestations has improved significantly since the 1990s with the advancement in neonatal care treatments and the implementation of evidence-based practices. Therefore, long-term health outcomes following extremely preterm birth will have increasing relevance to adult physicians,” observed Dr. Beckmann of University College London.
She reported on the results of detailed cardiovascular assessments conducted in 130 extremely premature EPICure participants and 64 matched controls who made it to London for 2 days of health testing when they turned 19 years of age. The findings update the results of similar comprehensive examinations done at age 11 years.
The extremely premature birth (EP) subjects were shorter and weighed less than did the controls. The two groups had similar seated systolic and diastolic blood pressure, and cardiac index didn’t differ between the two groups. However, the EP group had significantly higher supine central systolic and diastolic blood pressure and a higher heart rate.
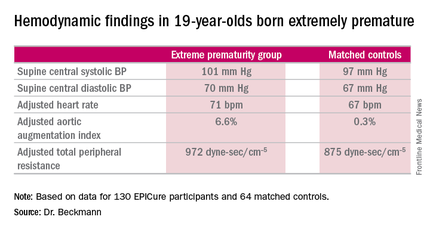
Moreover, the increases in aortic augmentation index – a composite of arterial stiffness and global wave reflections – and total peripheral resistance seen in the EP group at age 11 years persisted at the 19-year mark. It’s unclear whether the abnormal peripheral resistance in the EP group is structural or functional in nature. All hemodynamic differences between the two groups remained significant after adjustment for potential confounders.
Aortic pulse wave velocity was not significantly different between the two groups of 19-year-olds.
Data pertaining to other aspects of health in the 19-year-olds are now being analyzed. At the age-11 assessment, the EP group was found to have significantly impaired lung function (J Pediatr. 2012 Oct;161[4]:595-601.e2), high risk for neurodevelopmental disability (Pediatrics. 2009 Aug;124[2]:3249-57), a high rate of learning impairments, and an 18-fold increased risk of poor academic attainment compared to their matched peers (Arch Dis Child Fetal Neonatal Ed. 2009 Jul;94[4]:F283-9).
EPICure is funded by the Medical Research Council. Dr. Beckmann reported having no financial conflicts of interest.
CHICAGO – The abnormal arterial hemodynamics identified in 11-year-olds with an extremely preterm birth persist at age 19, according to an update from the landmark longitudinal EPICure study.
“Given the implications of these significant findings, cardiovascular monitoring and risk prevention would be highly recommended for all individuals born extremely preterm,” Dr. Joanne Beckmann said in presenting the EPICure results on the long-term consequences of extreme prematurity at the annual meeting of the American College of Cardiology.
EPICure is a longitudinal study investigating health outcomes in a national cohort of babies born extremely preterm at 22-25 weeks’ gestation in the United Kingdom during 1995-1996. It is the longest such study conducted anywhere.
“Neonatal survival at the lowest gestations has improved significantly since the 1990s with the advancement in neonatal care treatments and the implementation of evidence-based practices. Therefore, long-term health outcomes following extremely preterm birth will have increasing relevance to adult physicians,” observed Dr. Beckmann of University College London.
She reported on the results of detailed cardiovascular assessments conducted in 130 extremely premature EPICure participants and 64 matched controls who made it to London for 2 days of health testing when they turned 19 years of age. The findings update the results of similar comprehensive examinations done at age 11 years.
The extremely premature birth (EP) subjects were shorter and weighed less than did the controls. The two groups had similar seated systolic and diastolic blood pressure, and cardiac index didn’t differ between the two groups. However, the EP group had significantly higher supine central systolic and diastolic blood pressure and a higher heart rate.

Moreover, the increases in aortic augmentation index – a composite of arterial stiffness and global wave reflections – and total peripheral resistance seen in the EP group at age 11 years persisted at the 19-year mark. It’s unclear whether the abnormal peripheral resistance in the EP group is structural or functional in nature. All hemodynamic differences between the two groups remained significant after adjustment for potential confounders.
Aortic pulse wave velocity was not significantly different between the two groups of 19-year-olds.
Data pertaining to other aspects of health in the 19-year-olds are now being analyzed. At the age-11 assessment, the EP group was found to have significantly impaired lung function (J Pediatr. 2012 Oct;161[4]:595-601.e2), high risk for neurodevelopmental disability (Pediatrics. 2009 Aug;124[2]:3249-57), a high rate of learning impairments, and an 18-fold increased risk of poor academic attainment compared to their matched peers (Arch Dis Child Fetal Neonatal Ed. 2009 Jul;94[4]:F283-9).
EPICure is funded by the Medical Research Council. Dr. Beckmann reported having no financial conflicts of interest.
CHICAGO – The abnormal arterial hemodynamics identified in 11-year-olds with an extremely preterm birth persist at age 19, according to an update from the landmark longitudinal EPICure study.
“Given the implications of these significant findings, cardiovascular monitoring and risk prevention would be highly recommended for all individuals born extremely preterm,” Dr. Joanne Beckmann said in presenting the EPICure results on the long-term consequences of extreme prematurity at the annual meeting of the American College of Cardiology.
EPICure is a longitudinal study investigating health outcomes in a national cohort of babies born extremely preterm at 22-25 weeks’ gestation in the United Kingdom during 1995-1996. It is the longest such study conducted anywhere.
“Neonatal survival at the lowest gestations has improved significantly since the 1990s with the advancement in neonatal care treatments and the implementation of evidence-based practices. Therefore, long-term health outcomes following extremely preterm birth will have increasing relevance to adult physicians,” observed Dr. Beckmann of University College London.
She reported on the results of detailed cardiovascular assessments conducted in 130 extremely premature EPICure participants and 64 matched controls who made it to London for 2 days of health testing when they turned 19 years of age. The findings update the results of similar comprehensive examinations done at age 11 years.
The extremely premature birth (EP) subjects were shorter and weighed less than did the controls. The two groups had similar seated systolic and diastolic blood pressure, and cardiac index didn’t differ between the two groups. However, the EP group had significantly higher supine central systolic and diastolic blood pressure and a higher heart rate.

Moreover, the increases in aortic augmentation index – a composite of arterial stiffness and global wave reflections – and total peripheral resistance seen in the EP group at age 11 years persisted at the 19-year mark. It’s unclear whether the abnormal peripheral resistance in the EP group is structural or functional in nature. All hemodynamic differences between the two groups remained significant after adjustment for potential confounders.
Aortic pulse wave velocity was not significantly different between the two groups of 19-year-olds.
Data pertaining to other aspects of health in the 19-year-olds are now being analyzed. At the age-11 assessment, the EP group was found to have significantly impaired lung function (J Pediatr. 2012 Oct;161[4]:595-601.e2), high risk for neurodevelopmental disability (Pediatrics. 2009 Aug;124[2]:3249-57), a high rate of learning impairments, and an 18-fold increased risk of poor academic attainment compared to their matched peers (Arch Dis Child Fetal Neonatal Ed. 2009 Jul;94[4]:F283-9).
EPICure is funded by the Medical Research Council. Dr. Beckmann reported having no financial conflicts of interest.
AT ACC 16
Key clinical point: At age 19 years, persons born extremely premature still show significant abnormalities in arterial hemodynamics and peripheral resistance.
Major finding: The adjusted aortic augmentation index was 6.6% in 19-year-olds born at 22-25 weeks gestation compared with 0.3% in matched controls.
Data source: EPICure, a longitudinal study of health outcomes in a national cohort of babies born extremely preterm at 22-25 weeks gestation in the United Kingdom during 1995-1996.
Disclosures: EPICure is funded by the Medical Research Council. The presenter reported having no financial conflicts of interest.
Effective Psoriasis Therapy May Reduce Coronary Plaque Burden
CHICAGO – Improvement in psoriasis was associated with a significant reduction in coronary plaque burden within 1 year in a pilot study conducted at the National Heart, Lung, and Blood Institute, Joseph B. Lerman reported at the annual meeting of the American College of Cardiology.
“If you look at psoriatic plaque on the skin, it’s spewing out cytokines such as tumor necrosis factor–alpha and interleukin-17 which are highly linked to atherosclerosis. What we’ve found is that if you treat those plaques and reduce the severity of psoriasis, we’ve noticed small but statistically significant regression in the early noncalcified plaque. It’s a very exciting observation,” said Mr. Lerman, a medical student at Mount Sinai School of Medicine, New York.
He presented an observational study involving 50 consecutive patients with mild to moderate psoriasis of roughly 20 years duration and a median baseline Framingham Risk Score of 4. They underwent measurement of coronary plaque burden by coronary CT angiography at baseline and 1 year later.
During the study year, 33 patients showed significant improvement in their psoriasis as reflected in a decline in their mean Psoriasis Area and Severity Index score from 5.6 to 3.1. Those patients also showed significant improvement in their total and noncalcified plaque burden, with total plaque burden adjusted for luminal attenuation declining from 126 mm2 to 117 mm2. The association remained significant even after adjustment for traditional cardiovascular risk factors, the use of statin therapy, body mass index, and the use of systemic psoriasis therapies, including biologic agents.
Importantly, the reduction in plaque burden appeared to be largely concentrated in the subgroup of 31 patients on methotrexate or a biologic. And while this was a naturalistic observational study, the investigators have followed up with a prospective study of psoriasis patients placed on tumor necrosis factor inhibitors and confirmed that they, too, experienced a reduction in coronary plaque as measured by coronary CT angiography.
The investigators plan to expand the size of the study in order to confirm the findings. Mr. Lerman said the next question they would like to address is, how early does a measurable reduction in coronary plaque burden occur in response to clinical improvement in psoriasis? In order to explore this, the investigators will have to obtain institutional approval of a new investigative protocol which permits more frequent use of coronary CT angiography. At present the imaging study can be conducted only once per year due to the radiation exposure.
Mr. Lerman was involved in the psoriasis study while participating in the National Institutes of Health Medical Research Scholars Program. Senior investigator in the pilot study was Dr. Nehal Mehta, chief of the Section of Inflammation and Metabolic Disease at NHLBI in Bethesda, Md.
Mr. Lerman reported having no financial conflicts of interest.
CHICAGO – Improvement in psoriasis was associated with a significant reduction in coronary plaque burden within 1 year in a pilot study conducted at the National Heart, Lung, and Blood Institute, Joseph B. Lerman reported at the annual meeting of the American College of Cardiology.
“If you look at psoriatic plaque on the skin, it’s spewing out cytokines such as tumor necrosis factor–alpha and interleukin-17 which are highly linked to atherosclerosis. What we’ve found is that if you treat those plaques and reduce the severity of psoriasis, we’ve noticed small but statistically significant regression in the early noncalcified plaque. It’s a very exciting observation,” said Mr. Lerman, a medical student at Mount Sinai School of Medicine, New York.
He presented an observational study involving 50 consecutive patients with mild to moderate psoriasis of roughly 20 years duration and a median baseline Framingham Risk Score of 4. They underwent measurement of coronary plaque burden by coronary CT angiography at baseline and 1 year later.
During the study year, 33 patients showed significant improvement in their psoriasis as reflected in a decline in their mean Psoriasis Area and Severity Index score from 5.6 to 3.1. Those patients also showed significant improvement in their total and noncalcified plaque burden, with total plaque burden adjusted for luminal attenuation declining from 126 mm2 to 117 mm2. The association remained significant even after adjustment for traditional cardiovascular risk factors, the use of statin therapy, body mass index, and the use of systemic psoriasis therapies, including biologic agents.
Importantly, the reduction in plaque burden appeared to be largely concentrated in the subgroup of 31 patients on methotrexate or a biologic. And while this was a naturalistic observational study, the investigators have followed up with a prospective study of psoriasis patients placed on tumor necrosis factor inhibitors and confirmed that they, too, experienced a reduction in coronary plaque as measured by coronary CT angiography.
The investigators plan to expand the size of the study in order to confirm the findings. Mr. Lerman said the next question they would like to address is, how early does a measurable reduction in coronary plaque burden occur in response to clinical improvement in psoriasis? In order to explore this, the investigators will have to obtain institutional approval of a new investigative protocol which permits more frequent use of coronary CT angiography. At present the imaging study can be conducted only once per year due to the radiation exposure.
Mr. Lerman was involved in the psoriasis study while participating in the National Institutes of Health Medical Research Scholars Program. Senior investigator in the pilot study was Dr. Nehal Mehta, chief of the Section of Inflammation and Metabolic Disease at NHLBI in Bethesda, Md.
Mr. Lerman reported having no financial conflicts of interest.
CHICAGO – Improvement in psoriasis was associated with a significant reduction in coronary plaque burden within 1 year in a pilot study conducted at the National Heart, Lung, and Blood Institute, Joseph B. Lerman reported at the annual meeting of the American College of Cardiology.
“If you look at psoriatic plaque on the skin, it’s spewing out cytokines such as tumor necrosis factor–alpha and interleukin-17 which are highly linked to atherosclerosis. What we’ve found is that if you treat those plaques and reduce the severity of psoriasis, we’ve noticed small but statistically significant regression in the early noncalcified plaque. It’s a very exciting observation,” said Mr. Lerman, a medical student at Mount Sinai School of Medicine, New York.
He presented an observational study involving 50 consecutive patients with mild to moderate psoriasis of roughly 20 years duration and a median baseline Framingham Risk Score of 4. They underwent measurement of coronary plaque burden by coronary CT angiography at baseline and 1 year later.
During the study year, 33 patients showed significant improvement in their psoriasis as reflected in a decline in their mean Psoriasis Area and Severity Index score from 5.6 to 3.1. Those patients also showed significant improvement in their total and noncalcified plaque burden, with total plaque burden adjusted for luminal attenuation declining from 126 mm2 to 117 mm2. The association remained significant even after adjustment for traditional cardiovascular risk factors, the use of statin therapy, body mass index, and the use of systemic psoriasis therapies, including biologic agents.
Importantly, the reduction in plaque burden appeared to be largely concentrated in the subgroup of 31 patients on methotrexate or a biologic. And while this was a naturalistic observational study, the investigators have followed up with a prospective study of psoriasis patients placed on tumor necrosis factor inhibitors and confirmed that they, too, experienced a reduction in coronary plaque as measured by coronary CT angiography.
The investigators plan to expand the size of the study in order to confirm the findings. Mr. Lerman said the next question they would like to address is, how early does a measurable reduction in coronary plaque burden occur in response to clinical improvement in psoriasis? In order to explore this, the investigators will have to obtain institutional approval of a new investigative protocol which permits more frequent use of coronary CT angiography. At present the imaging study can be conducted only once per year due to the radiation exposure.
Mr. Lerman was involved in the psoriasis study while participating in the National Institutes of Health Medical Research Scholars Program. Senior investigator in the pilot study was Dr. Nehal Mehta, chief of the Section of Inflammation and Metabolic Disease at NHLBI in Bethesda, Md.
Mr. Lerman reported having no financial conflicts of interest.
AT ACC 16
Effective psoriasis therapy may reduce coronary plaque burden
CHICAGO – Improvement in psoriasis was associated with a significant reduction in coronary plaque burden within 1 year in a pilot study conducted at the National Heart, Lung, and Blood Institute, Joseph B. Lerman reported at the annual meeting of the American College of Cardiology.
“If you look at psoriatic plaque on the skin, it’s spewing out cytokines such as tumor necrosis factor–alpha and interleukin-17 which are highly linked to atherosclerosis. What we’ve found is that if you treat those plaques and reduce the severity of psoriasis, we’ve noticed small but statistically significant regression in the early noncalcified plaque. It’s a very exciting observation,” said Mr. Lerman, a medical student at Mount Sinai School of Medicine, New York.
He presented an observational study involving 50 consecutive patients with mild to moderate psoriasis of roughly 20 years duration and a median baseline Framingham Risk Score of 4. They underwent measurement of coronary plaque burden by coronary CT angiography at baseline and 1 year later.
During the study year, 33 patients showed significant improvement in their psoriasis as reflected in a decline in their mean Psoriasis Area and Severity Index score from 5.6 to 3.1. Those patients also showed significant improvement in their total and noncalcified plaque burden, with total plaque burden adjusted for luminal attenuation declining from 126 mm2 to 117 mm2. The association remained significant even after adjustment for traditional cardiovascular risk factors, the use of statin therapy, body mass index, and the use of systemic psoriasis therapies, including biologic agents.
Importantly, the reduction in plaque burden appeared to be largely concentrated in the subgroup of 31 patients on methotrexate or a biologic. And while this was a naturalistic observational study, the investigators have followed up with a prospective study of psoriasis patients placed on tumor necrosis factor inhibitors and confirmed that they, too, experienced a reduction in coronary plaque as measured by coronary CT angiography.
The investigators plan to expand the size of the study in order to confirm the findings. Mr. Lerman said the next question they would like to address is, how early does a measurable reduction in coronary plaque burden occur in response to clinical improvement in psoriasis? In order to explore this, the investigators will have to obtain institutional approval of a new investigative protocol which permits more frequent use of coronary CT angiography. At present the imaging study can be conducted only once per year due to the radiation exposure.
Mr. Lerman was involved in the psoriasis study while participating in the National Institutes of Health Medical Research Scholars Program. Senior investigator in the pilot study was Dr. Nehal Mehta, chief of the Section of Inflammation and Metabolic Disease at NHLBI in Bethesda, Md.
Mr. Lerman reported having no financial conflicts of interest.
CHICAGO – Improvement in psoriasis was associated with a significant reduction in coronary plaque burden within 1 year in a pilot study conducted at the National Heart, Lung, and Blood Institute, Joseph B. Lerman reported at the annual meeting of the American College of Cardiology.
“If you look at psoriatic plaque on the skin, it’s spewing out cytokines such as tumor necrosis factor–alpha and interleukin-17 which are highly linked to atherosclerosis. What we’ve found is that if you treat those plaques and reduce the severity of psoriasis, we’ve noticed small but statistically significant regression in the early noncalcified plaque. It’s a very exciting observation,” said Mr. Lerman, a medical student at Mount Sinai School of Medicine, New York.
He presented an observational study involving 50 consecutive patients with mild to moderate psoriasis of roughly 20 years duration and a median baseline Framingham Risk Score of 4. They underwent measurement of coronary plaque burden by coronary CT angiography at baseline and 1 year later.
During the study year, 33 patients showed significant improvement in their psoriasis as reflected in a decline in their mean Psoriasis Area and Severity Index score from 5.6 to 3.1. Those patients also showed significant improvement in their total and noncalcified plaque burden, with total plaque burden adjusted for luminal attenuation declining from 126 mm2 to 117 mm2. The association remained significant even after adjustment for traditional cardiovascular risk factors, the use of statin therapy, body mass index, and the use of systemic psoriasis therapies, including biologic agents.
Importantly, the reduction in plaque burden appeared to be largely concentrated in the subgroup of 31 patients on methotrexate or a biologic. And while this was a naturalistic observational study, the investigators have followed up with a prospective study of psoriasis patients placed on tumor necrosis factor inhibitors and confirmed that they, too, experienced a reduction in coronary plaque as measured by coronary CT angiography.
The investigators plan to expand the size of the study in order to confirm the findings. Mr. Lerman said the next question they would like to address is, how early does a measurable reduction in coronary plaque burden occur in response to clinical improvement in psoriasis? In order to explore this, the investigators will have to obtain institutional approval of a new investigative protocol which permits more frequent use of coronary CT angiography. At present the imaging study can be conducted only once per year due to the radiation exposure.
Mr. Lerman was involved in the psoriasis study while participating in the National Institutes of Health Medical Research Scholars Program. Senior investigator in the pilot study was Dr. Nehal Mehta, chief of the Section of Inflammation and Metabolic Disease at NHLBI in Bethesda, Md.
Mr. Lerman reported having no financial conflicts of interest.
CHICAGO – Improvement in psoriasis was associated with a significant reduction in coronary plaque burden within 1 year in a pilot study conducted at the National Heart, Lung, and Blood Institute, Joseph B. Lerman reported at the annual meeting of the American College of Cardiology.
“If you look at psoriatic plaque on the skin, it’s spewing out cytokines such as tumor necrosis factor–alpha and interleukin-17 which are highly linked to atherosclerosis. What we’ve found is that if you treat those plaques and reduce the severity of psoriasis, we’ve noticed small but statistically significant regression in the early noncalcified plaque. It’s a very exciting observation,” said Mr. Lerman, a medical student at Mount Sinai School of Medicine, New York.
He presented an observational study involving 50 consecutive patients with mild to moderate psoriasis of roughly 20 years duration and a median baseline Framingham Risk Score of 4. They underwent measurement of coronary plaque burden by coronary CT angiography at baseline and 1 year later.
During the study year, 33 patients showed significant improvement in their psoriasis as reflected in a decline in their mean Psoriasis Area and Severity Index score from 5.6 to 3.1. Those patients also showed significant improvement in their total and noncalcified plaque burden, with total plaque burden adjusted for luminal attenuation declining from 126 mm2 to 117 mm2. The association remained significant even after adjustment for traditional cardiovascular risk factors, the use of statin therapy, body mass index, and the use of systemic psoriasis therapies, including biologic agents.
Importantly, the reduction in plaque burden appeared to be largely concentrated in the subgroup of 31 patients on methotrexate or a biologic. And while this was a naturalistic observational study, the investigators have followed up with a prospective study of psoriasis patients placed on tumor necrosis factor inhibitors and confirmed that they, too, experienced a reduction in coronary plaque as measured by coronary CT angiography.
The investigators plan to expand the size of the study in order to confirm the findings. Mr. Lerman said the next question they would like to address is, how early does a measurable reduction in coronary plaque burden occur in response to clinical improvement in psoriasis? In order to explore this, the investigators will have to obtain institutional approval of a new investigative protocol which permits more frequent use of coronary CT angiography. At present the imaging study can be conducted only once per year due to the radiation exposure.
Mr. Lerman was involved in the psoriasis study while participating in the National Institutes of Health Medical Research Scholars Program. Senior investigator in the pilot study was Dr. Nehal Mehta, chief of the Section of Inflammation and Metabolic Disease at NHLBI in Bethesda, Md.
Mr. Lerman reported having no financial conflicts of interest.
AT ACC 16
Key clinical point: Improved PASI scores were linked to regression of early noncalcified coronary plaque.
Major finding: Reduction in skin inflammation in psoriasis patients may cause regression of coronary plaque.
Data source: This prospective study of 50 patients with mild to moderate psoriasis featured precise measurements of coronary plaque burden at baseline and 1 year later.
Disclosures: The study was sponsored by the National Heart, Lung, and Blood Institute. The presenter reported having no financial conflicts of interest.
Seven highly effective hospital strategies for reducing PCI mortality
CHICAGO – Researchers have identified seven specific hospital practices and procedures that are independently associated with very low 30-day risk-standardized mortality rates for percutaneous coronary interventions.
“Hospitals seeking to improve their outcomes may consider adopting one or more of these strategies if they haven’t done so already,” Dr. Jeptha P. Curtis said at the annual meeting of the American College of Cardiology.
He presented highlights of the Translating Outstanding Performance in Percutaneous Coronary Intervention (TOP PCI) study, a 5-year examination of hospital organizational strategies and enabling structures associated with lower 30-day risk-standardized rates for Medicare fee-for-service patients who underwent PCI in 2010-2012. The study included 398 randomly selected hospitals participating in the ACC’s National Cardiovascular Data Registry. The effort involved site visits and completion of a detailed 76-question survey by hospital officials.
The TOP PCI results have important practice implications.
“Despite improved technology and expansion of our evidence base, PCI carries a significant risk of adverse outcomes, including mortality. PCI outcomes are being considered for inclusion in public reporting and value-based purchasing programs, which reinforces the importance of trying to reduce mortality rates for hospitals,” observed Dr. Curtis of Yale University in New Haven, Conn.
Top among the findings that came as a surprise to him was that officials at 26% of hospitals reported that, at their institution, revenue and profits drove most decisions by senior management.
Another unexpected finding: “We asked if this hospital used data from the ACC CathPCI Registry or other sources to support specific quality improvement initiatives. I’m proud to say 84% did. I’m ashamed to say that 16% of hospitals that are making the investment to participate in this registry had not identified a single quality improvement project that was based off the data they were getting back,” Dr. Curtis said.
Many different hospital strategies and practices were looked at in TOP PCI, but in multivariate analysis, these seven stood out as having a significant association with lower 30-day risk-standardized mortality rates in PCI:
• Conducting regular reviews of PCI appropriateness by an interdisciplinary team including noninterventional cardiologists: 37% of hospitals reported doing so.
• Retaining and rewarding high-quality staff. Eight percent of hospitals were unable to do so, and they were at the high end of the 30-day mortality range.
• Using the radial approach to perform most or all PCIs. This was the practice at 22% of hospitals.
• Clinicians holding regular meetings with home health agencies to review the postdischarge care of cardiac patients. This was the practice at 24% of hospitals.
• Having PCI patients cared for by hospitalists only rarely or sometimes. At 22% of hospitals, hospitalists always cared for the PCI patients, and that practice was associated with higher 30-day mortality.
• Implementing hospital-initiated quality improvements aimed at reducing postdischarge mortality. Thirty percent of hospitals engaged in this approach.
• Use of the Plan-Do-Study-Act method of quality of improvement. This approach, widely accepted within the quality improvement field, was practiced routinely at 30% of hospitals.
Dr. Curtis noted that the nearly 400 hospitals participating in TOP PCI followed a bell-shaped curve in terms of the number of these successful strategies utilized. Some hospitals used none or one, a fewer number used five or six. None used all seven.
Discussant Spencer B. King III voiced skepticism about the TOP PCI effort.
“I know mortality is easy to measure, but in my view it does not assure quality of a PCI program by itself. I think one of the problems is that quality is kind of like pornography: Everybody knows it when they see it, but it’s hard to define. You’ve got to realize that the major mortality from interventions is patient related,” said Dr. King, who is president of the Heart and Vascular Institute at Saint Joseph’s Health System in Atlanta.
“This is a first step, it’s not the end of the story,” Dr. Curtis replied. “What this data set really represents is a unique snapshot of how at a very granular level hospitals are caring for their patients. That’s information we’ve really never had before. We can use it to look at differences in appropriateness, differences in discharge medications – any quality outcome you can come up with we can probably assess what strategies may or not be effective for those outcomes.”
Another audience critique was that, while each of the seven hospital strategies was associated with a statistically significant improvement in 30-day risk-stratified mortality, the absolute differences were quite small.
“Changing mortality is challenging. You’re really trying to change the direction of the Titanic,” Dr. Curtis responded. “It takes a long time, and it’s a matter of doing a bunch of small, subtle things. The issue is that we’ve never had any evidence to say what specific care and practices are effective. This is really the first toe in the water to say there are things you can do that actually make a difference at the end of the day.”
TOP PCI was funded by the National Heart, Lung, and Blood Institute. Dr. Curtis reported receiving salary support from the ACC’s National Cardiovascular Data Registry and the Centers for Medicare & Medicaid Services.
CHICAGO – Researchers have identified seven specific hospital practices and procedures that are independently associated with very low 30-day risk-standardized mortality rates for percutaneous coronary interventions.
“Hospitals seeking to improve their outcomes may consider adopting one or more of these strategies if they haven’t done so already,” Dr. Jeptha P. Curtis said at the annual meeting of the American College of Cardiology.
He presented highlights of the Translating Outstanding Performance in Percutaneous Coronary Intervention (TOP PCI) study, a 5-year examination of hospital organizational strategies and enabling structures associated with lower 30-day risk-standardized rates for Medicare fee-for-service patients who underwent PCI in 2010-2012. The study included 398 randomly selected hospitals participating in the ACC’s National Cardiovascular Data Registry. The effort involved site visits and completion of a detailed 76-question survey by hospital officials.
The TOP PCI results have important practice implications.
“Despite improved technology and expansion of our evidence base, PCI carries a significant risk of adverse outcomes, including mortality. PCI outcomes are being considered for inclusion in public reporting and value-based purchasing programs, which reinforces the importance of trying to reduce mortality rates for hospitals,” observed Dr. Curtis of Yale University in New Haven, Conn.
Top among the findings that came as a surprise to him was that officials at 26% of hospitals reported that, at their institution, revenue and profits drove most decisions by senior management.
Another unexpected finding: “We asked if this hospital used data from the ACC CathPCI Registry or other sources to support specific quality improvement initiatives. I’m proud to say 84% did. I’m ashamed to say that 16% of hospitals that are making the investment to participate in this registry had not identified a single quality improvement project that was based off the data they were getting back,” Dr. Curtis said.
Many different hospital strategies and practices were looked at in TOP PCI, but in multivariate analysis, these seven stood out as having a significant association with lower 30-day risk-standardized mortality rates in PCI:
• Conducting regular reviews of PCI appropriateness by an interdisciplinary team including noninterventional cardiologists: 37% of hospitals reported doing so.
• Retaining and rewarding high-quality staff. Eight percent of hospitals were unable to do so, and they were at the high end of the 30-day mortality range.
• Using the radial approach to perform most or all PCIs. This was the practice at 22% of hospitals.
• Clinicians holding regular meetings with home health agencies to review the postdischarge care of cardiac patients. This was the practice at 24% of hospitals.
• Having PCI patients cared for by hospitalists only rarely or sometimes. At 22% of hospitals, hospitalists always cared for the PCI patients, and that practice was associated with higher 30-day mortality.
• Implementing hospital-initiated quality improvements aimed at reducing postdischarge mortality. Thirty percent of hospitals engaged in this approach.
• Use of the Plan-Do-Study-Act method of quality of improvement. This approach, widely accepted within the quality improvement field, was practiced routinely at 30% of hospitals.
Dr. Curtis noted that the nearly 400 hospitals participating in TOP PCI followed a bell-shaped curve in terms of the number of these successful strategies utilized. Some hospitals used none or one, a fewer number used five or six. None used all seven.
Discussant Spencer B. King III voiced skepticism about the TOP PCI effort.
“I know mortality is easy to measure, but in my view it does not assure quality of a PCI program by itself. I think one of the problems is that quality is kind of like pornography: Everybody knows it when they see it, but it’s hard to define. You’ve got to realize that the major mortality from interventions is patient related,” said Dr. King, who is president of the Heart and Vascular Institute at Saint Joseph’s Health System in Atlanta.
“This is a first step, it’s not the end of the story,” Dr. Curtis replied. “What this data set really represents is a unique snapshot of how at a very granular level hospitals are caring for their patients. That’s information we’ve really never had before. We can use it to look at differences in appropriateness, differences in discharge medications – any quality outcome you can come up with we can probably assess what strategies may or not be effective for those outcomes.”
Another audience critique was that, while each of the seven hospital strategies was associated with a statistically significant improvement in 30-day risk-stratified mortality, the absolute differences were quite small.
“Changing mortality is challenging. You’re really trying to change the direction of the Titanic,” Dr. Curtis responded. “It takes a long time, and it’s a matter of doing a bunch of small, subtle things. The issue is that we’ve never had any evidence to say what specific care and practices are effective. This is really the first toe in the water to say there are things you can do that actually make a difference at the end of the day.”
TOP PCI was funded by the National Heart, Lung, and Blood Institute. Dr. Curtis reported receiving salary support from the ACC’s National Cardiovascular Data Registry and the Centers for Medicare & Medicaid Services.
CHICAGO – Researchers have identified seven specific hospital practices and procedures that are independently associated with very low 30-day risk-standardized mortality rates for percutaneous coronary interventions.
“Hospitals seeking to improve their outcomes may consider adopting one or more of these strategies if they haven’t done so already,” Dr. Jeptha P. Curtis said at the annual meeting of the American College of Cardiology.
He presented highlights of the Translating Outstanding Performance in Percutaneous Coronary Intervention (TOP PCI) study, a 5-year examination of hospital organizational strategies and enabling structures associated with lower 30-day risk-standardized rates for Medicare fee-for-service patients who underwent PCI in 2010-2012. The study included 398 randomly selected hospitals participating in the ACC’s National Cardiovascular Data Registry. The effort involved site visits and completion of a detailed 76-question survey by hospital officials.
The TOP PCI results have important practice implications.
“Despite improved technology and expansion of our evidence base, PCI carries a significant risk of adverse outcomes, including mortality. PCI outcomes are being considered for inclusion in public reporting and value-based purchasing programs, which reinforces the importance of trying to reduce mortality rates for hospitals,” observed Dr. Curtis of Yale University in New Haven, Conn.
Top among the findings that came as a surprise to him was that officials at 26% of hospitals reported that, at their institution, revenue and profits drove most decisions by senior management.
Another unexpected finding: “We asked if this hospital used data from the ACC CathPCI Registry or other sources to support specific quality improvement initiatives. I’m proud to say 84% did. I’m ashamed to say that 16% of hospitals that are making the investment to participate in this registry had not identified a single quality improvement project that was based off the data they were getting back,” Dr. Curtis said.
Many different hospital strategies and practices were looked at in TOP PCI, but in multivariate analysis, these seven stood out as having a significant association with lower 30-day risk-standardized mortality rates in PCI:
• Conducting regular reviews of PCI appropriateness by an interdisciplinary team including noninterventional cardiologists: 37% of hospitals reported doing so.
• Retaining and rewarding high-quality staff. Eight percent of hospitals were unable to do so, and they were at the high end of the 30-day mortality range.
• Using the radial approach to perform most or all PCIs. This was the practice at 22% of hospitals.
• Clinicians holding regular meetings with home health agencies to review the postdischarge care of cardiac patients. This was the practice at 24% of hospitals.
• Having PCI patients cared for by hospitalists only rarely or sometimes. At 22% of hospitals, hospitalists always cared for the PCI patients, and that practice was associated with higher 30-day mortality.
• Implementing hospital-initiated quality improvements aimed at reducing postdischarge mortality. Thirty percent of hospitals engaged in this approach.
• Use of the Plan-Do-Study-Act method of quality of improvement. This approach, widely accepted within the quality improvement field, was practiced routinely at 30% of hospitals.
Dr. Curtis noted that the nearly 400 hospitals participating in TOP PCI followed a bell-shaped curve in terms of the number of these successful strategies utilized. Some hospitals used none or one, a fewer number used five or six. None used all seven.
Discussant Spencer B. King III voiced skepticism about the TOP PCI effort.
“I know mortality is easy to measure, but in my view it does not assure quality of a PCI program by itself. I think one of the problems is that quality is kind of like pornography: Everybody knows it when they see it, but it’s hard to define. You’ve got to realize that the major mortality from interventions is patient related,” said Dr. King, who is president of the Heart and Vascular Institute at Saint Joseph’s Health System in Atlanta.
“This is a first step, it’s not the end of the story,” Dr. Curtis replied. “What this data set really represents is a unique snapshot of how at a very granular level hospitals are caring for their patients. That’s information we’ve really never had before. We can use it to look at differences in appropriateness, differences in discharge medications – any quality outcome you can come up with we can probably assess what strategies may or not be effective for those outcomes.”
Another audience critique was that, while each of the seven hospital strategies was associated with a statistically significant improvement in 30-day risk-stratified mortality, the absolute differences were quite small.
“Changing mortality is challenging. You’re really trying to change the direction of the Titanic,” Dr. Curtis responded. “It takes a long time, and it’s a matter of doing a bunch of small, subtle things. The issue is that we’ve never had any evidence to say what specific care and practices are effective. This is really the first toe in the water to say there are things you can do that actually make a difference at the end of the day.”
TOP PCI was funded by the National Heart, Lung, and Blood Institute. Dr. Curtis reported receiving salary support from the ACC’s National Cardiovascular Data Registry and the Centers for Medicare & Medicaid Services.
AT ACC 16
Key clinical point: Specific hospital strategies and practices are associated with differences in 30-day risk-standardized mortality rates in PCI.
Major finding: Seven specific strategies associated with significantly lower PCI mortality were identified.
Data source: The TOP PCI study was a detailed survey of 398 hospitals.
Disclosures: The study was funded by the National Heart, Lung, and Blood Institute. The presenter reported receiving salary support from the ACC’s National Cardiovascular Data Registry and the Centers for Medicare and Medicaid Services.
Superhigh ferritin spells trouble in macrophage activation syndrome
MAUI, HAWAII – The use of new classification criteria for macrophage activation syndrome specific to systemic juvenile idiopathic arthritis will help to avoid missing patients with the criteria that hematologists have used to diagnose the condition, which they call hemophagocytic lymphohistiocytosis, according to Dr. Anne M. Stevens, a pediatric rheumatologist at the University of Washington, Seattle.
“The problem is, if we try to use the HLH [hemophagocytic lymphohistiocytosis] criteria, we’re going to miss a lot of patients with systemic juvenile idiopathic arthritis–related macrophage activation syndrome, so we’re not going to treat them aggressively and many of them will die,” she said at the 2016 Rheumatology Winter Clinical Symposium.
Indeed, roughly one-third of patients with macrophage activation syndrome (MAS) arising as a complication of systemic juvenile idiopathic arthritis (SJIA) don’t meet the HLH diagnostic criteria. And the mortality of untreated MAS is up to 28%, Dr. Stevens noted.
MAS isn’t a new entity. It has become a more frequent topic of discussion lately because the pathogenesis of this cytokine storm is better understood, and biologic agents addressing the underlying disease mechanisms are in clinical trials. But MAS was first described nearly 40 years ago at a pediatric rheumatology conference in Park City, Utah.
Recently, an international consensus panel convened as a collaboration between the European League Against Rheumatism, the American College of Rheumatology, and the Paediatric Rheumatology International Trials Organisation has developed a new set of classification criteria for MAS complicating SJIA (Arthritis Rheumatol. 2016 Mar;68[3]:566-76).
In a validation study, the criteria had a sensitivity of 73% and a specificity of 99% for the diagnosis.
Under the 2016 criteria, a patient with known or suspected SJIA is classified as having MAS if he or she is febrile, has a serum ferritin level in excess of 684 ng/mL, and meets any two of the following four criteria: a platelet count of 181 x 109/L or less, triglycerides greater than 156 mg/dL, a serum aspartate aminotransferase level in excess of 48 U/L, or a fibrinogen level of 360 mg/dL or lower.
The specific cutoff threshold values for these laboratory measures are mainly of interest for research purposes in assuring appropriate patient enrollment in clinical trials. Practically speaking, it’s time to suspect MAS – a true medical emergency – in a child with SJIA whose fever has become persistent all day every day, rather than the quotidian fever that spikes in the evening characteristic of SJIA, along with hemodynamic instability, altered mental status, a rash, organomegaly, relative or progressive cytopenias, a very high ferritin level, and/or a relatively low or falling erythrocyte sedimentation rate.
A child with MAS superimposed on SJIA will look septic, according to Dr. Stevens. And if the ESR was 75 mm/hr yesterday and has “improved” to 45 mm/hr today, yet the child looks sicker or the other blood parameters are falling, that’s not an encouraging sign.
A very high serum ferritin level in patients with SJIA-related MAS is a red flag for increased mortality risk, as demonstrated in a study by physicians at Seattle Children’s Hospital. They retrospectively looked at 171 patients with a serum ferritin of 1,000 ng/dL and showed that the mortality risk climbed in parallel with the peak ferritin level. Children with a peak serum ferritin greater than 3,000 ng/mL had a 4.3-fold increased risk of mortality and a 2.5-fold greater risk of ICU admission than did those with a ferritin level of 1,000-3,000 ng/mL (Pediatr Crit Care Med. 2011 Nov;12[6]:e233-6).
Based upon these data, Dr. Stevens said she considers a serum ferritin of 1,000 ng/mL or more worthy of immediate attention.
The current standard of care in treating SJIA-related MAS is high-dose pulsed methylprednisolone at 30 mg up to 1 g/kg and IV cyclosporine at 5 mg/kg per day divided into two doses.
“There are good data to show that this will turn around half of affected kids,” according to the pediatric rheumatologist.
Second-line therapy targets interleukin-1: either IV anakinra (Kineret) at 2 mg/kg per day titrated up to 10 mg/kg, or canakinumab (Ilaris) at 4 mg/kg titrated up to 12.5 mg/kg every 4 weeks.
However, the current thinking is that what tips patients with SJIA into MAS is high levels of free interleukin-18. For that reason, several different anti–IL-18 agents are now under study, including tadekinig alfa, an IL-18 binding protein, and rituximab (Rituxan) for patients with Epstein-Barr virus–related MAS.
Dr. Stevens reported having no relevant financial conflicts.
MAUI, HAWAII – The use of new classification criteria for macrophage activation syndrome specific to systemic juvenile idiopathic arthritis will help to avoid missing patients with the criteria that hematologists have used to diagnose the condition, which they call hemophagocytic lymphohistiocytosis, according to Dr. Anne M. Stevens, a pediatric rheumatologist at the University of Washington, Seattle.
“The problem is, if we try to use the HLH [hemophagocytic lymphohistiocytosis] criteria, we’re going to miss a lot of patients with systemic juvenile idiopathic arthritis–related macrophage activation syndrome, so we’re not going to treat them aggressively and many of them will die,” she said at the 2016 Rheumatology Winter Clinical Symposium.
Indeed, roughly one-third of patients with macrophage activation syndrome (MAS) arising as a complication of systemic juvenile idiopathic arthritis (SJIA) don’t meet the HLH diagnostic criteria. And the mortality of untreated MAS is up to 28%, Dr. Stevens noted.
MAS isn’t a new entity. It has become a more frequent topic of discussion lately because the pathogenesis of this cytokine storm is better understood, and biologic agents addressing the underlying disease mechanisms are in clinical trials. But MAS was first described nearly 40 years ago at a pediatric rheumatology conference in Park City, Utah.
Recently, an international consensus panel convened as a collaboration between the European League Against Rheumatism, the American College of Rheumatology, and the Paediatric Rheumatology International Trials Organisation has developed a new set of classification criteria for MAS complicating SJIA (Arthritis Rheumatol. 2016 Mar;68[3]:566-76).
In a validation study, the criteria had a sensitivity of 73% and a specificity of 99% for the diagnosis.
Under the 2016 criteria, a patient with known or suspected SJIA is classified as having MAS if he or she is febrile, has a serum ferritin level in excess of 684 ng/mL, and meets any two of the following four criteria: a platelet count of 181 x 109/L or less, triglycerides greater than 156 mg/dL, a serum aspartate aminotransferase level in excess of 48 U/L, or a fibrinogen level of 360 mg/dL or lower.
The specific cutoff threshold values for these laboratory measures are mainly of interest for research purposes in assuring appropriate patient enrollment in clinical trials. Practically speaking, it’s time to suspect MAS – a true medical emergency – in a child with SJIA whose fever has become persistent all day every day, rather than the quotidian fever that spikes in the evening characteristic of SJIA, along with hemodynamic instability, altered mental status, a rash, organomegaly, relative or progressive cytopenias, a very high ferritin level, and/or a relatively low or falling erythrocyte sedimentation rate.
A child with MAS superimposed on SJIA will look septic, according to Dr. Stevens. And if the ESR was 75 mm/hr yesterday and has “improved” to 45 mm/hr today, yet the child looks sicker or the other blood parameters are falling, that’s not an encouraging sign.
A very high serum ferritin level in patients with SJIA-related MAS is a red flag for increased mortality risk, as demonstrated in a study by physicians at Seattle Children’s Hospital. They retrospectively looked at 171 patients with a serum ferritin of 1,000 ng/dL and showed that the mortality risk climbed in parallel with the peak ferritin level. Children with a peak serum ferritin greater than 3,000 ng/mL had a 4.3-fold increased risk of mortality and a 2.5-fold greater risk of ICU admission than did those with a ferritin level of 1,000-3,000 ng/mL (Pediatr Crit Care Med. 2011 Nov;12[6]:e233-6).
Based upon these data, Dr. Stevens said she considers a serum ferritin of 1,000 ng/mL or more worthy of immediate attention.
The current standard of care in treating SJIA-related MAS is high-dose pulsed methylprednisolone at 30 mg up to 1 g/kg and IV cyclosporine at 5 mg/kg per day divided into two doses.
“There are good data to show that this will turn around half of affected kids,” according to the pediatric rheumatologist.
Second-line therapy targets interleukin-1: either IV anakinra (Kineret) at 2 mg/kg per day titrated up to 10 mg/kg, or canakinumab (Ilaris) at 4 mg/kg titrated up to 12.5 mg/kg every 4 weeks.
However, the current thinking is that what tips patients with SJIA into MAS is high levels of free interleukin-18. For that reason, several different anti–IL-18 agents are now under study, including tadekinig alfa, an IL-18 binding protein, and rituximab (Rituxan) for patients with Epstein-Barr virus–related MAS.
Dr. Stevens reported having no relevant financial conflicts.
MAUI, HAWAII – The use of new classification criteria for macrophage activation syndrome specific to systemic juvenile idiopathic arthritis will help to avoid missing patients with the criteria that hematologists have used to diagnose the condition, which they call hemophagocytic lymphohistiocytosis, according to Dr. Anne M. Stevens, a pediatric rheumatologist at the University of Washington, Seattle.
“The problem is, if we try to use the HLH [hemophagocytic lymphohistiocytosis] criteria, we’re going to miss a lot of patients with systemic juvenile idiopathic arthritis–related macrophage activation syndrome, so we’re not going to treat them aggressively and many of them will die,” she said at the 2016 Rheumatology Winter Clinical Symposium.
Indeed, roughly one-third of patients with macrophage activation syndrome (MAS) arising as a complication of systemic juvenile idiopathic arthritis (SJIA) don’t meet the HLH diagnostic criteria. And the mortality of untreated MAS is up to 28%, Dr. Stevens noted.
MAS isn’t a new entity. It has become a more frequent topic of discussion lately because the pathogenesis of this cytokine storm is better understood, and biologic agents addressing the underlying disease mechanisms are in clinical trials. But MAS was first described nearly 40 years ago at a pediatric rheumatology conference in Park City, Utah.
Recently, an international consensus panel convened as a collaboration between the European League Against Rheumatism, the American College of Rheumatology, and the Paediatric Rheumatology International Trials Organisation has developed a new set of classification criteria for MAS complicating SJIA (Arthritis Rheumatol. 2016 Mar;68[3]:566-76).
In a validation study, the criteria had a sensitivity of 73% and a specificity of 99% for the diagnosis.
Under the 2016 criteria, a patient with known or suspected SJIA is classified as having MAS if he or she is febrile, has a serum ferritin level in excess of 684 ng/mL, and meets any two of the following four criteria: a platelet count of 181 x 109/L or less, triglycerides greater than 156 mg/dL, a serum aspartate aminotransferase level in excess of 48 U/L, or a fibrinogen level of 360 mg/dL or lower.
The specific cutoff threshold values for these laboratory measures are mainly of interest for research purposes in assuring appropriate patient enrollment in clinical trials. Practically speaking, it’s time to suspect MAS – a true medical emergency – in a child with SJIA whose fever has become persistent all day every day, rather than the quotidian fever that spikes in the evening characteristic of SJIA, along with hemodynamic instability, altered mental status, a rash, organomegaly, relative or progressive cytopenias, a very high ferritin level, and/or a relatively low or falling erythrocyte sedimentation rate.
A child with MAS superimposed on SJIA will look septic, according to Dr. Stevens. And if the ESR was 75 mm/hr yesterday and has “improved” to 45 mm/hr today, yet the child looks sicker or the other blood parameters are falling, that’s not an encouraging sign.
A very high serum ferritin level in patients with SJIA-related MAS is a red flag for increased mortality risk, as demonstrated in a study by physicians at Seattle Children’s Hospital. They retrospectively looked at 171 patients with a serum ferritin of 1,000 ng/dL and showed that the mortality risk climbed in parallel with the peak ferritin level. Children with a peak serum ferritin greater than 3,000 ng/mL had a 4.3-fold increased risk of mortality and a 2.5-fold greater risk of ICU admission than did those with a ferritin level of 1,000-3,000 ng/mL (Pediatr Crit Care Med. 2011 Nov;12[6]:e233-6).
Based upon these data, Dr. Stevens said she considers a serum ferritin of 1,000 ng/mL or more worthy of immediate attention.
The current standard of care in treating SJIA-related MAS is high-dose pulsed methylprednisolone at 30 mg up to 1 g/kg and IV cyclosporine at 5 mg/kg per day divided into two doses.
“There are good data to show that this will turn around half of affected kids,” according to the pediatric rheumatologist.
Second-line therapy targets interleukin-1: either IV anakinra (Kineret) at 2 mg/kg per day titrated up to 10 mg/kg, or canakinumab (Ilaris) at 4 mg/kg titrated up to 12.5 mg/kg every 4 weeks.
However, the current thinking is that what tips patients with SJIA into MAS is high levels of free interleukin-18. For that reason, several different anti–IL-18 agents are now under study, including tadekinig alfa, an IL-18 binding protein, and rituximab (Rituxan) for patients with Epstein-Barr virus–related MAS.
Dr. Stevens reported having no relevant financial conflicts.
EXPERT ANALYSIS FROM RWCS 2016
Cardioband scores hit for percutaneous direct mitral annuloplasty
CHICAGO – One-year results of a pivotal European trial of the percutaneous Cardioband mitral valve reconstruction system show a stable, consistent, and clinically meaningful reduction in mitral regurgitation coupled with significant quality of life improvements and a safety profile equivalent to that of other transcatheter valve procedures.
“The results are quite impressive. Ladies and gentlemen, I can tell you that I’ve used almost all the devices for direct and indirect annuloplasty, and this is the only device that works in a reproducible fashion,” Dr. Karl-Heinz Kuck said at the annual meeting of the American College of Cardiology.
He presented the results for the first 50 patients to reach 12 months of follow-up after undergoing the Cardioband procedure in a multicenter prospective study in which participants served as their own before-and-after controls. On the strength of these results, the Cardioband device has been approved by European Union regulatory authorities for the nonsurgical treatment of symptomatic moderate to severe secondary, or functional, mitral regurgitation. In Germany, the Cardioband procedure is now routinely reimbursed at a level similar to that of the MitraClip, according to Dr. Kuck, president of the German Cardiac Society and head of cardiology at St. Georg Hospital in Hamburg, Germany.
The Cardioband procedure essentially entails percutaneous implantation of an adjustable surgical ring designed to remodel a severely dysfunctional mitral valve by repairing the valve annulus. The implantation procedure features transfemoral venous access, which the TAVR experience has shown to be safer than transapical access. As in surgery, the percutaneous procedure utilizes supra-annular fixation. And it accomplishes a significant reduction in annular dimensions, comparable to what is achieved with a size 28 surgical ring.
“And the most important thing: Because we are not interfering with the leaflets or any other part of the mitral valve, the procedure leaves all options open for the future by preserving the native anatomy,” Dr. Kuck noted.
The procedure entails a transseptal puncture, insertion of the system, deployment of the implant, and adjustment of its size by cinching it down under echocardiographic guidance in order to reduce the septolateral valve dimension. The whole thing takes about 75 minutes.
The connection of the implant to the annulus is achieved in sutureless fashion using a series of screw-in anchors.
All 50 participants in the consecutive series were deemed by a heart team to be at unacceptably high surgical risk. They averaged 71 years of age, with an left ventricular ejection fraction of 33% and a left ventricular end diastolic diameter of 61 mm. Among them, 31 had ischemic heart disease, 11 had chronic obstructive pulmonary disease, 38 were in moderate or severe renal failure, 39 had atrial fibrillation, 12 had severe pulmonary hypertension, and 16 had previously undergone CABG surgery.
The 30-day safety adverse events consisted of one hemorrhagic stroke, a single major bleeding complication, two cases of acute renal failure, and one of cardiac tamponade. There were no MIs, and neither of the two deaths were related to the procedure.
In terms of efficacy, at baseline three-quarters of patients had grade 3-4 mitral regurgitation (MR). At discharge that was true for only 12%. At 1 year of follow-up, 90% of subjects had MR grade 2 or less, and roughly two-thirds of patients had MR grade 0-1.
The procedure did what it was designed to do: The mean valve septolateral dimension decreased by 30%, from 37 mm at baseline to 26 mm at discharge.
Dr. Kuck provided 6-month data on functional improvement. The mean 6-minute walk distance improved from 262 to 339 meters. At baseline, 87% of subjects were NYHA class III or IV; at 6 months, 77% were NYHA class I or II. Scores on the Minnesota Living with Heart Failure Questionnaire improved from a mean of 39 to 16 at follow-up.
A much larger European postmarketing commercial use study of the Cardioband system is now underway.
The Cardioband procedure addresses a major unmet need, Dr. Kuck observed. More than 4 million patients in the United States alone have mitral valve disease. When medically managed, patients with severe secondary mitral regurgitation have a poor prognosis, with 1- and 5-year mortality rates of 20% and 50%, and an extremely high rate of rehospitalization for heart failure. And yet multiple surveys have shown only a minority of these patients undergo surgery.
Discussant Dr. Spencer B. King III called the transcatheter mitral valve reconstruction system “quite fascinating.” He wondered what happens if the operator accidentally grabs the nearby circumflex artery with one of the device anchors. The answer, Dr. Kuck replied, is that the anchors can be unscrewed and repositioned at any point during the procedure.
Dr. King, president of the Heart and Vascular Institute at Saint Joseph’s Health System in Atlanta, has developed several devices widely used in interventional cardiology. He shook his head in amazement at the speed at which the European regulatory agency operated in this case, noting that EU marketing approval for the Valtech Cardioband device was granted and a payment structure was almost immediately established on the basis of a 50-patient, first-in-man study.
“The data are very consistent. I think that‘s what made the difference,” Dr. Kuck said.
The study was funded by Valtech. Dr. Kuck reported serving as a consultant to Biosense Webster, Edwards, and St. Jude, and on a speakers’ bureau for Medtronic.
CHICAGO – One-year results of a pivotal European trial of the percutaneous Cardioband mitral valve reconstruction system show a stable, consistent, and clinically meaningful reduction in mitral regurgitation coupled with significant quality of life improvements and a safety profile equivalent to that of other transcatheter valve procedures.
“The results are quite impressive. Ladies and gentlemen, I can tell you that I’ve used almost all the devices for direct and indirect annuloplasty, and this is the only device that works in a reproducible fashion,” Dr. Karl-Heinz Kuck said at the annual meeting of the American College of Cardiology.
He presented the results for the first 50 patients to reach 12 months of follow-up after undergoing the Cardioband procedure in a multicenter prospective study in which participants served as their own before-and-after controls. On the strength of these results, the Cardioband device has been approved by European Union regulatory authorities for the nonsurgical treatment of symptomatic moderate to severe secondary, or functional, mitral regurgitation. In Germany, the Cardioband procedure is now routinely reimbursed at a level similar to that of the MitraClip, according to Dr. Kuck, president of the German Cardiac Society and head of cardiology at St. Georg Hospital in Hamburg, Germany.
The Cardioband procedure essentially entails percutaneous implantation of an adjustable surgical ring designed to remodel a severely dysfunctional mitral valve by repairing the valve annulus. The implantation procedure features transfemoral venous access, which the TAVR experience has shown to be safer than transapical access. As in surgery, the percutaneous procedure utilizes supra-annular fixation. And it accomplishes a significant reduction in annular dimensions, comparable to what is achieved with a size 28 surgical ring.
“And the most important thing: Because we are not interfering with the leaflets or any other part of the mitral valve, the procedure leaves all options open for the future by preserving the native anatomy,” Dr. Kuck noted.
The procedure entails a transseptal puncture, insertion of the system, deployment of the implant, and adjustment of its size by cinching it down under echocardiographic guidance in order to reduce the septolateral valve dimension. The whole thing takes about 75 minutes.
The connection of the implant to the annulus is achieved in sutureless fashion using a series of screw-in anchors.
All 50 participants in the consecutive series were deemed by a heart team to be at unacceptably high surgical risk. They averaged 71 years of age, with an left ventricular ejection fraction of 33% and a left ventricular end diastolic diameter of 61 mm. Among them, 31 had ischemic heart disease, 11 had chronic obstructive pulmonary disease, 38 were in moderate or severe renal failure, 39 had atrial fibrillation, 12 had severe pulmonary hypertension, and 16 had previously undergone CABG surgery.
The 30-day safety adverse events consisted of one hemorrhagic stroke, a single major bleeding complication, two cases of acute renal failure, and one of cardiac tamponade. There were no MIs, and neither of the two deaths were related to the procedure.
In terms of efficacy, at baseline three-quarters of patients had grade 3-4 mitral regurgitation (MR). At discharge that was true for only 12%. At 1 year of follow-up, 90% of subjects had MR grade 2 or less, and roughly two-thirds of patients had MR grade 0-1.
The procedure did what it was designed to do: The mean valve septolateral dimension decreased by 30%, from 37 mm at baseline to 26 mm at discharge.
Dr. Kuck provided 6-month data on functional improvement. The mean 6-minute walk distance improved from 262 to 339 meters. At baseline, 87% of subjects were NYHA class III or IV; at 6 months, 77% were NYHA class I or II. Scores on the Minnesota Living with Heart Failure Questionnaire improved from a mean of 39 to 16 at follow-up.
A much larger European postmarketing commercial use study of the Cardioband system is now underway.
The Cardioband procedure addresses a major unmet need, Dr. Kuck observed. More than 4 million patients in the United States alone have mitral valve disease. When medically managed, patients with severe secondary mitral regurgitation have a poor prognosis, with 1- and 5-year mortality rates of 20% and 50%, and an extremely high rate of rehospitalization for heart failure. And yet multiple surveys have shown only a minority of these patients undergo surgery.
Discussant Dr. Spencer B. King III called the transcatheter mitral valve reconstruction system “quite fascinating.” He wondered what happens if the operator accidentally grabs the nearby circumflex artery with one of the device anchors. The answer, Dr. Kuck replied, is that the anchors can be unscrewed and repositioned at any point during the procedure.
Dr. King, president of the Heart and Vascular Institute at Saint Joseph’s Health System in Atlanta, has developed several devices widely used in interventional cardiology. He shook his head in amazement at the speed at which the European regulatory agency operated in this case, noting that EU marketing approval for the Valtech Cardioband device was granted and a payment structure was almost immediately established on the basis of a 50-patient, first-in-man study.
“The data are very consistent. I think that‘s what made the difference,” Dr. Kuck said.
The study was funded by Valtech. Dr. Kuck reported serving as a consultant to Biosense Webster, Edwards, and St. Jude, and on a speakers’ bureau for Medtronic.
CHICAGO – One-year results of a pivotal European trial of the percutaneous Cardioband mitral valve reconstruction system show a stable, consistent, and clinically meaningful reduction in mitral regurgitation coupled with significant quality of life improvements and a safety profile equivalent to that of other transcatheter valve procedures.
“The results are quite impressive. Ladies and gentlemen, I can tell you that I’ve used almost all the devices for direct and indirect annuloplasty, and this is the only device that works in a reproducible fashion,” Dr. Karl-Heinz Kuck said at the annual meeting of the American College of Cardiology.
He presented the results for the first 50 patients to reach 12 months of follow-up after undergoing the Cardioband procedure in a multicenter prospective study in which participants served as their own before-and-after controls. On the strength of these results, the Cardioband device has been approved by European Union regulatory authorities for the nonsurgical treatment of symptomatic moderate to severe secondary, or functional, mitral regurgitation. In Germany, the Cardioband procedure is now routinely reimbursed at a level similar to that of the MitraClip, according to Dr. Kuck, president of the German Cardiac Society and head of cardiology at St. Georg Hospital in Hamburg, Germany.
The Cardioband procedure essentially entails percutaneous implantation of an adjustable surgical ring designed to remodel a severely dysfunctional mitral valve by repairing the valve annulus. The implantation procedure features transfemoral venous access, which the TAVR experience has shown to be safer than transapical access. As in surgery, the percutaneous procedure utilizes supra-annular fixation. And it accomplishes a significant reduction in annular dimensions, comparable to what is achieved with a size 28 surgical ring.
“And the most important thing: Because we are not interfering with the leaflets or any other part of the mitral valve, the procedure leaves all options open for the future by preserving the native anatomy,” Dr. Kuck noted.
The procedure entails a transseptal puncture, insertion of the system, deployment of the implant, and adjustment of its size by cinching it down under echocardiographic guidance in order to reduce the septolateral valve dimension. The whole thing takes about 75 minutes.
The connection of the implant to the annulus is achieved in sutureless fashion using a series of screw-in anchors.
All 50 participants in the consecutive series were deemed by a heart team to be at unacceptably high surgical risk. They averaged 71 years of age, with an left ventricular ejection fraction of 33% and a left ventricular end diastolic diameter of 61 mm. Among them, 31 had ischemic heart disease, 11 had chronic obstructive pulmonary disease, 38 were in moderate or severe renal failure, 39 had atrial fibrillation, 12 had severe pulmonary hypertension, and 16 had previously undergone CABG surgery.
The 30-day safety adverse events consisted of one hemorrhagic stroke, a single major bleeding complication, two cases of acute renal failure, and one of cardiac tamponade. There were no MIs, and neither of the two deaths were related to the procedure.
In terms of efficacy, at baseline three-quarters of patients had grade 3-4 mitral regurgitation (MR). At discharge that was true for only 12%. At 1 year of follow-up, 90% of subjects had MR grade 2 or less, and roughly two-thirds of patients had MR grade 0-1.
The procedure did what it was designed to do: The mean valve septolateral dimension decreased by 30%, from 37 mm at baseline to 26 mm at discharge.
Dr. Kuck provided 6-month data on functional improvement. The mean 6-minute walk distance improved from 262 to 339 meters. At baseline, 87% of subjects were NYHA class III or IV; at 6 months, 77% were NYHA class I or II. Scores on the Minnesota Living with Heart Failure Questionnaire improved from a mean of 39 to 16 at follow-up.
A much larger European postmarketing commercial use study of the Cardioband system is now underway.
The Cardioband procedure addresses a major unmet need, Dr. Kuck observed. More than 4 million patients in the United States alone have mitral valve disease. When medically managed, patients with severe secondary mitral regurgitation have a poor prognosis, with 1- and 5-year mortality rates of 20% and 50%, and an extremely high rate of rehospitalization for heart failure. And yet multiple surveys have shown only a minority of these patients undergo surgery.
Discussant Dr. Spencer B. King III called the transcatheter mitral valve reconstruction system “quite fascinating.” He wondered what happens if the operator accidentally grabs the nearby circumflex artery with one of the device anchors. The answer, Dr. Kuck replied, is that the anchors can be unscrewed and repositioned at any point during the procedure.
Dr. King, president of the Heart and Vascular Institute at Saint Joseph’s Health System in Atlanta, has developed several devices widely used in interventional cardiology. He shook his head in amazement at the speed at which the European regulatory agency operated in this case, noting that EU marketing approval for the Valtech Cardioband device was granted and a payment structure was almost immediately established on the basis of a 50-patient, first-in-man study.
“The data are very consistent. I think that‘s what made the difference,” Dr. Kuck said.
The study was funded by Valtech. Dr. Kuck reported serving as a consultant to Biosense Webster, Edwards, and St. Jude, and on a speakers’ bureau for Medtronic.
AT ACC 16
Key clinical point: A new percutaneous repair option has been developed for patients with secondary mitral regurgitation.
Major finding: At 1 year of follow-up after receiving the Cardioband mitral reconstruction system, 90% of treated subjects with baseline severe mitral valve disease had mitral regurgitation of grade 2 or less.
Data source: A prospective, multicenter clinical trial featuring 12 months of follow-up of 50 treated patients with severe secondary mitral regurgitation.
Disclosures: The study was sponsored by Valtech, maker of the Cardioband system. The presenter reported serving as a consultant to Biosense Webster, Edwards, and St. Jude, and on a speakers bureau for Medtronic.
Imaging can predict who’ll progress from nonspecific symptoms to arthritis
MAUI, HAWAII – Recent evidence indicates that subclinical joint inflammation on ultrasound or MRI in patients having a positive cyclic citrullinated peptide antibody test but only nonspecific musculoskeletal symptoms predicts sharply increased risk of progression to full-blown diagnosable inflammatory arthritis within a matter of months. And this creates a new dilemma for rheumatologists.
The quandary is this: What are you going to do about it? After all, in the current era, the traditional treatment pyramid has been turned upside down, and early and aggressive therapy of arthritis is now recognized as best care.
“These are patients I have come back on a monthly basis instead of every 3 or 6 months. I’m hoping I’m going to be able to pick up diagnosable arthritis either by my physical exam or by the CDAI [Clinical Disease Activity Index score] or SDAI [Simplified Disease Activity Index score] so that I can initiate therapy as quickly as I possibly can to try to have a bigger impact. But I don’t initiate therapy just based on this imaging information,” Dr. Orrin M. Troum said at the 2016 Rheumatology Winter Clinical Symposium.
He highlighted two recent European studies that illustrate the prognostic power of contemporary joint imaging technologies performed at a point when patients don’t yet meet diagnostic criteria for arthritis. Both prospective studies were presented at the 2015 annual meeting of the American College of Rheumatology and have since been published.
Dr. Jackie L. Nam of the University of Leeds (England) reported on 136 consecutive anti–cyclic citrullinated peptide (anti-CCP) antibody–positive patients who presented with nonspecific musculoskeletal symptoms and no clinical synovitis. They underwent baseline ultrasound evaluation using power Doppler and grayscale imaging of 32 joints and were then prospectively followed for a median of 18.3 months.
At baseline ultrasound, 21% of patients had one or more erosions, 96% of patients had positive grayscale findings in one or more joints, and 30% were positive on power Doppler.
Forty-two percent of patients developed inflammatory arthritis after a median of 8.6 months. Patients with a baseline power Doppler score of 2 or more on a standard 0-3 scale in any joint had a 55% risk of developing inflammatory arthritis, compared with 4% if their power Doppler score was 0 or 1. A grayscale score of at least 2 was associated with a 26% likelihood of developing inflammatory arthritis, while a 0 or 1 on gray scale conferred a 3% risk. Progression to inflammatory arthritis occurred earlier in patients with a power Doppler score of 2 or 3, as well, at a median of 7.1 months (Ann Rheum Dis. 2016 Jan 22. doi: 10.1136/annrheumdis-2015-208235).
The other longitudinal study that grabbed Dr. Troum’s attention was a Dutch report on 150 patients who presented with nonspecific aches and pains. They underwent baseline serologic testing along with unilateral imaging of metacarpophalangeal, wrist, and metatarsophalangeal joints using 1.5-Tesla MRI. The imaging study was able to detect erosions as well as osteitis – that is, an inflammatory infiltrate in the bone marrow, as opposed to edema, which is watery fluid.
During a minimum of 6 months and median of 75 weeks of follow-up, 30 patients developed clinical arthritis. Twenty-six of the 30 did so before 20 weeks of follow-up had elapsed. The strongest risk factors for progression to arthritis were anti-CCP antibody positivity, with a hazard ratio of 6.4, and subclinical MRI inflammation, with a hazard ratio of 5.1.
The 1-year rate of progression to arthritis was 31% with MRI-detected subclinical inflammation alone, 71% in patients who were both MRI- and anti-CCP antibody–positive, and 3% in those who were both MRI- and anti-CCP antibody–negative (Ann Rheum Dis. 2015 Nov 27. doi: 10.1136/annrheumdis-2015-208138).
Dr. Roy Fleischmann rose from the audience to challenge Dr. Troum: Don’t these baseline imaging–positive, anti-CCP antibody–positive patients already have disease? Isn’t this the time you want to treat? he asked.
“It would ideally be the time to treat, yes,” replied Dr. Troum. “If it was me, I probably would treat myself.”
“With what, Xanax?” quipped Dr. Fleischmann of the University of Texas, Dallas.
“With methotrexate, probably,” Dr. Troum said.
He added that these were preliminary studies with relatively small numbers of patients. Before changing his practice regarding patients with these subclinical findings, he’d like to see more data and an estimate of the number needed to treat.
“It could be as many as two-thirds of the population that you’d be overtreating, people who were never going to develop anything,” Dr. Troum observed.
He reported having no financial conflicts of interest regarding his presentation.
MAUI, HAWAII – Recent evidence indicates that subclinical joint inflammation on ultrasound or MRI in patients having a positive cyclic citrullinated peptide antibody test but only nonspecific musculoskeletal symptoms predicts sharply increased risk of progression to full-blown diagnosable inflammatory arthritis within a matter of months. And this creates a new dilemma for rheumatologists.
The quandary is this: What are you going to do about it? After all, in the current era, the traditional treatment pyramid has been turned upside down, and early and aggressive therapy of arthritis is now recognized as best care.
“These are patients I have come back on a monthly basis instead of every 3 or 6 months. I’m hoping I’m going to be able to pick up diagnosable arthritis either by my physical exam or by the CDAI [Clinical Disease Activity Index score] or SDAI [Simplified Disease Activity Index score] so that I can initiate therapy as quickly as I possibly can to try to have a bigger impact. But I don’t initiate therapy just based on this imaging information,” Dr. Orrin M. Troum said at the 2016 Rheumatology Winter Clinical Symposium.
He highlighted two recent European studies that illustrate the prognostic power of contemporary joint imaging technologies performed at a point when patients don’t yet meet diagnostic criteria for arthritis. Both prospective studies were presented at the 2015 annual meeting of the American College of Rheumatology and have since been published.
Dr. Jackie L. Nam of the University of Leeds (England) reported on 136 consecutive anti–cyclic citrullinated peptide (anti-CCP) antibody–positive patients who presented with nonspecific musculoskeletal symptoms and no clinical synovitis. They underwent baseline ultrasound evaluation using power Doppler and grayscale imaging of 32 joints and were then prospectively followed for a median of 18.3 months.
At baseline ultrasound, 21% of patients had one or more erosions, 96% of patients had positive grayscale findings in one or more joints, and 30% were positive on power Doppler.
Forty-two percent of patients developed inflammatory arthritis after a median of 8.6 months. Patients with a baseline power Doppler score of 2 or more on a standard 0-3 scale in any joint had a 55% risk of developing inflammatory arthritis, compared with 4% if their power Doppler score was 0 or 1. A grayscale score of at least 2 was associated with a 26% likelihood of developing inflammatory arthritis, while a 0 or 1 on gray scale conferred a 3% risk. Progression to inflammatory arthritis occurred earlier in patients with a power Doppler score of 2 or 3, as well, at a median of 7.1 months (Ann Rheum Dis. 2016 Jan 22. doi: 10.1136/annrheumdis-2015-208235).
The other longitudinal study that grabbed Dr. Troum’s attention was a Dutch report on 150 patients who presented with nonspecific aches and pains. They underwent baseline serologic testing along with unilateral imaging of metacarpophalangeal, wrist, and metatarsophalangeal joints using 1.5-Tesla MRI. The imaging study was able to detect erosions as well as osteitis – that is, an inflammatory infiltrate in the bone marrow, as opposed to edema, which is watery fluid.
During a minimum of 6 months and median of 75 weeks of follow-up, 30 patients developed clinical arthritis. Twenty-six of the 30 did so before 20 weeks of follow-up had elapsed. The strongest risk factors for progression to arthritis were anti-CCP antibody positivity, with a hazard ratio of 6.4, and subclinical MRI inflammation, with a hazard ratio of 5.1.
The 1-year rate of progression to arthritis was 31% with MRI-detected subclinical inflammation alone, 71% in patients who were both MRI- and anti-CCP antibody–positive, and 3% in those who were both MRI- and anti-CCP antibody–negative (Ann Rheum Dis. 2015 Nov 27. doi: 10.1136/annrheumdis-2015-208138).
Dr. Roy Fleischmann rose from the audience to challenge Dr. Troum: Don’t these baseline imaging–positive, anti-CCP antibody–positive patients already have disease? Isn’t this the time you want to treat? he asked.
“It would ideally be the time to treat, yes,” replied Dr. Troum. “If it was me, I probably would treat myself.”
“With what, Xanax?” quipped Dr. Fleischmann of the University of Texas, Dallas.
“With methotrexate, probably,” Dr. Troum said.
He added that these were preliminary studies with relatively small numbers of patients. Before changing his practice regarding patients with these subclinical findings, he’d like to see more data and an estimate of the number needed to treat.
“It could be as many as two-thirds of the population that you’d be overtreating, people who were never going to develop anything,” Dr. Troum observed.
He reported having no financial conflicts of interest regarding his presentation.
MAUI, HAWAII – Recent evidence indicates that subclinical joint inflammation on ultrasound or MRI in patients having a positive cyclic citrullinated peptide antibody test but only nonspecific musculoskeletal symptoms predicts sharply increased risk of progression to full-blown diagnosable inflammatory arthritis within a matter of months. And this creates a new dilemma for rheumatologists.
The quandary is this: What are you going to do about it? After all, in the current era, the traditional treatment pyramid has been turned upside down, and early and aggressive therapy of arthritis is now recognized as best care.
“These are patients I have come back on a monthly basis instead of every 3 or 6 months. I’m hoping I’m going to be able to pick up diagnosable arthritis either by my physical exam or by the CDAI [Clinical Disease Activity Index score] or SDAI [Simplified Disease Activity Index score] so that I can initiate therapy as quickly as I possibly can to try to have a bigger impact. But I don’t initiate therapy just based on this imaging information,” Dr. Orrin M. Troum said at the 2016 Rheumatology Winter Clinical Symposium.
He highlighted two recent European studies that illustrate the prognostic power of contemporary joint imaging technologies performed at a point when patients don’t yet meet diagnostic criteria for arthritis. Both prospective studies were presented at the 2015 annual meeting of the American College of Rheumatology and have since been published.
Dr. Jackie L. Nam of the University of Leeds (England) reported on 136 consecutive anti–cyclic citrullinated peptide (anti-CCP) antibody–positive patients who presented with nonspecific musculoskeletal symptoms and no clinical synovitis. They underwent baseline ultrasound evaluation using power Doppler and grayscale imaging of 32 joints and were then prospectively followed for a median of 18.3 months.
At baseline ultrasound, 21% of patients had one or more erosions, 96% of patients had positive grayscale findings in one or more joints, and 30% were positive on power Doppler.
Forty-two percent of patients developed inflammatory arthritis after a median of 8.6 months. Patients with a baseline power Doppler score of 2 or more on a standard 0-3 scale in any joint had a 55% risk of developing inflammatory arthritis, compared with 4% if their power Doppler score was 0 or 1. A grayscale score of at least 2 was associated with a 26% likelihood of developing inflammatory arthritis, while a 0 or 1 on gray scale conferred a 3% risk. Progression to inflammatory arthritis occurred earlier in patients with a power Doppler score of 2 or 3, as well, at a median of 7.1 months (Ann Rheum Dis. 2016 Jan 22. doi: 10.1136/annrheumdis-2015-208235).
The other longitudinal study that grabbed Dr. Troum’s attention was a Dutch report on 150 patients who presented with nonspecific aches and pains. They underwent baseline serologic testing along with unilateral imaging of metacarpophalangeal, wrist, and metatarsophalangeal joints using 1.5-Tesla MRI. The imaging study was able to detect erosions as well as osteitis – that is, an inflammatory infiltrate in the bone marrow, as opposed to edema, which is watery fluid.
During a minimum of 6 months and median of 75 weeks of follow-up, 30 patients developed clinical arthritis. Twenty-six of the 30 did so before 20 weeks of follow-up had elapsed. The strongest risk factors for progression to arthritis were anti-CCP antibody positivity, with a hazard ratio of 6.4, and subclinical MRI inflammation, with a hazard ratio of 5.1.
The 1-year rate of progression to arthritis was 31% with MRI-detected subclinical inflammation alone, 71% in patients who were both MRI- and anti-CCP antibody–positive, and 3% in those who were both MRI- and anti-CCP antibody–negative (Ann Rheum Dis. 2015 Nov 27. doi: 10.1136/annrheumdis-2015-208138).
Dr. Roy Fleischmann rose from the audience to challenge Dr. Troum: Don’t these baseline imaging–positive, anti-CCP antibody–positive patients already have disease? Isn’t this the time you want to treat? he asked.
“It would ideally be the time to treat, yes,” replied Dr. Troum. “If it was me, I probably would treat myself.”
“With what, Xanax?” quipped Dr. Fleischmann of the University of Texas, Dallas.
“With methotrexate, probably,” Dr. Troum said.
He added that these were preliminary studies with relatively small numbers of patients. Before changing his practice regarding patients with these subclinical findings, he’d like to see more data and an estimate of the number needed to treat.
“It could be as many as two-thirds of the population that you’d be overtreating, people who were never going to develop anything,” Dr. Troum observed.
He reported having no financial conflicts of interest regarding his presentation.
EXPERT ANALYSIS FROM RWCS 2016
Detecting morphologically difficult-to-diagnose melanomas
WAIKOLOA, HAWAII – Nodular melanoma is one of a handful of morphologic subtypes of melanoma that are difficult to diagnose in a timely way because they lack the conventional features associated with melanoma. But certain clinical and dermoscopic clues are helpful in avoiding misdiagnosis, Dr. Ashfaq A. Marghoob said at the Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
He provided tips on how to recognize nodular melanoma and other difficult-to-diagnose morphologic subtypes, including desmoplastic, amelanotic, nevoid, and epidermotropic metastatic melanoma.
These diagnostically challenging melanoma subtypes have several things in common: clinically, they don’t look like melanomas, and dermoscopically, they display specific tell-tale features that speak of malignancy, including atypical polymorphous blood vessels and crystalline structures. Dermoscopy can be a big help in deciding whether to biopsy a clinically unremarkable-looking lesion, observed Dr. Marghoob of Memorial Sloan Kettering Cancer Center in New York.
Here’s the rundown on how to avoid delayed diagnosis of these melanomas:
Nodular melanoma: The big challenge posed by nodular melanoma is that it grows at a very high rate, a median of 0.5 mm per month, “so it’s really not a subtype of melanoma that’s amenable to screening,” the dermatologist said.
“Think about this: A patient comes in for screening today and you see nothing on their skin. Then a month from today they start a nodular melanoma. Their next appointment with you is a year from now. By the time they come back, that nodular melanoma is going to be a life-threatening cancer,” he explained. “It requires patient engagement in self-examination to bring these lesions to our attention, and we have to be perceptive enough to see these patients in a timely manner when they call. Otherwise these are melanomas that are really the killer tumors.”
Nodular melanomas often lack the “ABCD” features associated with most melanomas: asymmetry, border irregularity, color, and diameter greater than one-quarter of an inch. Instead, the lesions may exhibit the modified ABCDs described in pediatric melanoma: amelanotic, bump showing symmetry, color homogeneity in a shade of either pink or blue-black, and de novo, meaning the lesion didn’t arise in association with a nevus, Dr. Marghoob continued.
Roughly 60% of nodular melanomas are pigmented, the other 40% amelanotic. Dermoscopically, pigmented nodular melanomas are characterized by a greater degree of symmetry than that seen with other melanomas. Atypical vessels that exhibit tortuosity, a cork screw pattern, or an irregular hairpin shape are prominently visible. The lesions are multicolored, often blue-black, and may exhibit a blue-white veil. Notably absent are regression structures, streaks, and networks.
An amelanotic nodular melanoma also features prominent atypical vessels, symmetry, and sometimes crystalline structures, which appear as shiny white lines. Comma vessels and arborizing vessels are absent.
A more detailed description of the dermoscopic findings in nodular melanoma is available in a study coauthored by Dr. Marghoob (JAMA Dermatol. 2013;149[6]:699-709).
Desmoplastic melanoma: Seventy percent of desmoplastic melanomas are amelanotic. They often present as an unremarkable firm subcutaneous papule or nodule. Indeed, desmoplastic melanomas often have such a banal clinical appearance that in one-third of cases, melanoma is not even in the differential diagnosis.
“You’re thinking cyst, lipoma, dermatofibroma ... something other than a malignancy,” Dr. Marghoob said.
Suspicion is raised by a benign-looking lesion that’s symptomatic and/or exhibiting growth on chronically sun-damaged skin in an older patient. A scar with no history of trauma that’s located on chronically sun-damaged skin is another tipoff. Also, desmoplastic melanoma can arise in association with a lentigo maligna component.
“It is this knowledge of the association between lentigo maligna and desmoplastic melanoma that for the first time has enabled us to diagnose desmoplastic melanomas that are thin – not 5 or 10 mm, but more like 0.6 mm in thickness,” Dr. Marghoob said.
The main dermoscopic clue is the presence of atypical vascular structures.
For a fuller account of the dermoscopic characteristics of desmoplastic melanomas, Dr. Marghoob recommended a study he coauthored (JAMA Dermatol. 2013;149[4]:413-21).
Amelanotic melanoma: An indication of how difficult these cancers are to diagnose was provided by the international Genes, Environment, and Melanoma Study of nearly 3,500 invasive melanomas, 8% of which were amelanotic. They were at a more advanced tumor stage at diagnosis. As a result, the mortality risk during a median 7.6 years of follow-up was an adjusted twofold greater for patients with amelanotic as compared with pigmented melanomas (JAMA Dermatol. 2014;150[12]:1306-314).
Dr. Marghoob coauthored a retrospective review of 20 consecutively diagnosed amelanotic melanomas. The lesions lacked any of the classic ABCD features. Most were symmetric, oval to round in shape, had regular borders, and were pink in color. Dermoscopically, a consistent finding was a polymorphous vascular pattern (J Eur Acad Dermatol Venereol. 2012;26[5]:591-6).
In an earlier five-continent study comparing dermoscopic features of 222 amelanotic melanomas, 105 pigmented melanomas, and 170 benign melanocytic lesions, investigators came up with a simple model that distinguished melanomas from nonmelanomas with 70% sensitivity and 56% specificity. The take-home message: Amelanotic melanomas often feature more than one shade of pink upon dermoscopic examination, along with dotted and linear irregular vessels and predominant central vessels (Arch Dermatol. 2008;144[9]:1120-7).
Nevoid melanoma: Clinically they often resemble an intradermal nevus, and they’re often amelanotic. Dermoscopically, however, an intradermal nevus displays a mammillated surface, with each individual mammillated area being associated with an isolated comma or hairpin vessel. In contrast, the surface of a nevoid melanoma is more undulating in appearance and lacks the classic vascular pattern seen in intradermal nevi. If there’s pigmentation present in a nevoid melanoma it will be distributed heterogeneously. The dermoscopic finding of irregular blood vessels and/or crystalline structures tips the balance in favor of biopsy.
Dr. Marghoob referred dermatologists interested in the dermoscopic features of nevoid melanoma to a recent study by members of the International Dermoscopy Society; he was one of the authors (Br J Dermatol. 2015;172[4]:961-7).
Epidermotropic metastatic melanoma: These lesions lack the ABCDs, are often amelanotic, and look like nondescript papules. If such a lesion is new, especially in a patient with a history of melanoma, think epidermotropic metastatic melanoma.
Spanish dermatologists have described five different dermoscopic patterns of melanoma metastases in an informative retrospective study: blue nevus-like, angioma-like, nevus-like, vascular, and unspecific. The vascular type is the most common form, characterized by amelanotic papules 2-3 mm in diameter and tortuous corkscrew vessels within the lesions (Br J Dermatol. 2013;169[1]:91-9).
Dr. Marghoob reported having no financial conflicts of interest regarding his presentation. SDEF and this news organization are owned by the same parent company.
WAIKOLOA, HAWAII – Nodular melanoma is one of a handful of morphologic subtypes of melanoma that are difficult to diagnose in a timely way because they lack the conventional features associated with melanoma. But certain clinical and dermoscopic clues are helpful in avoiding misdiagnosis, Dr. Ashfaq A. Marghoob said at the Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
He provided tips on how to recognize nodular melanoma and other difficult-to-diagnose morphologic subtypes, including desmoplastic, amelanotic, nevoid, and epidermotropic metastatic melanoma.
These diagnostically challenging melanoma subtypes have several things in common: clinically, they don’t look like melanomas, and dermoscopically, they display specific tell-tale features that speak of malignancy, including atypical polymorphous blood vessels and crystalline structures. Dermoscopy can be a big help in deciding whether to biopsy a clinically unremarkable-looking lesion, observed Dr. Marghoob of Memorial Sloan Kettering Cancer Center in New York.
Here’s the rundown on how to avoid delayed diagnosis of these melanomas:
Nodular melanoma: The big challenge posed by nodular melanoma is that it grows at a very high rate, a median of 0.5 mm per month, “so it’s really not a subtype of melanoma that’s amenable to screening,” the dermatologist said.
“Think about this: A patient comes in for screening today and you see nothing on their skin. Then a month from today they start a nodular melanoma. Their next appointment with you is a year from now. By the time they come back, that nodular melanoma is going to be a life-threatening cancer,” he explained. “It requires patient engagement in self-examination to bring these lesions to our attention, and we have to be perceptive enough to see these patients in a timely manner when they call. Otherwise these are melanomas that are really the killer tumors.”
Nodular melanomas often lack the “ABCD” features associated with most melanomas: asymmetry, border irregularity, color, and diameter greater than one-quarter of an inch. Instead, the lesions may exhibit the modified ABCDs described in pediatric melanoma: amelanotic, bump showing symmetry, color homogeneity in a shade of either pink or blue-black, and de novo, meaning the lesion didn’t arise in association with a nevus, Dr. Marghoob continued.
Roughly 60% of nodular melanomas are pigmented, the other 40% amelanotic. Dermoscopically, pigmented nodular melanomas are characterized by a greater degree of symmetry than that seen with other melanomas. Atypical vessels that exhibit tortuosity, a cork screw pattern, or an irregular hairpin shape are prominently visible. The lesions are multicolored, often blue-black, and may exhibit a blue-white veil. Notably absent are regression structures, streaks, and networks.
An amelanotic nodular melanoma also features prominent atypical vessels, symmetry, and sometimes crystalline structures, which appear as shiny white lines. Comma vessels and arborizing vessels are absent.
A more detailed description of the dermoscopic findings in nodular melanoma is available in a study coauthored by Dr. Marghoob (JAMA Dermatol. 2013;149[6]:699-709).
Desmoplastic melanoma: Seventy percent of desmoplastic melanomas are amelanotic. They often present as an unremarkable firm subcutaneous papule or nodule. Indeed, desmoplastic melanomas often have such a banal clinical appearance that in one-third of cases, melanoma is not even in the differential diagnosis.
“You’re thinking cyst, lipoma, dermatofibroma ... something other than a malignancy,” Dr. Marghoob said.
Suspicion is raised by a benign-looking lesion that’s symptomatic and/or exhibiting growth on chronically sun-damaged skin in an older patient. A scar with no history of trauma that’s located on chronically sun-damaged skin is another tipoff. Also, desmoplastic melanoma can arise in association with a lentigo maligna component.
“It is this knowledge of the association between lentigo maligna and desmoplastic melanoma that for the first time has enabled us to diagnose desmoplastic melanomas that are thin – not 5 or 10 mm, but more like 0.6 mm in thickness,” Dr. Marghoob said.
The main dermoscopic clue is the presence of atypical vascular structures.
For a fuller account of the dermoscopic characteristics of desmoplastic melanomas, Dr. Marghoob recommended a study he coauthored (JAMA Dermatol. 2013;149[4]:413-21).
Amelanotic melanoma: An indication of how difficult these cancers are to diagnose was provided by the international Genes, Environment, and Melanoma Study of nearly 3,500 invasive melanomas, 8% of which were amelanotic. They were at a more advanced tumor stage at diagnosis. As a result, the mortality risk during a median 7.6 years of follow-up was an adjusted twofold greater for patients with amelanotic as compared with pigmented melanomas (JAMA Dermatol. 2014;150[12]:1306-314).
Dr. Marghoob coauthored a retrospective review of 20 consecutively diagnosed amelanotic melanomas. The lesions lacked any of the classic ABCD features. Most were symmetric, oval to round in shape, had regular borders, and were pink in color. Dermoscopically, a consistent finding was a polymorphous vascular pattern (J Eur Acad Dermatol Venereol. 2012;26[5]:591-6).
In an earlier five-continent study comparing dermoscopic features of 222 amelanotic melanomas, 105 pigmented melanomas, and 170 benign melanocytic lesions, investigators came up with a simple model that distinguished melanomas from nonmelanomas with 70% sensitivity and 56% specificity. The take-home message: Amelanotic melanomas often feature more than one shade of pink upon dermoscopic examination, along with dotted and linear irregular vessels and predominant central vessels (Arch Dermatol. 2008;144[9]:1120-7).
Nevoid melanoma: Clinically they often resemble an intradermal nevus, and they’re often amelanotic. Dermoscopically, however, an intradermal nevus displays a mammillated surface, with each individual mammillated area being associated with an isolated comma or hairpin vessel. In contrast, the surface of a nevoid melanoma is more undulating in appearance and lacks the classic vascular pattern seen in intradermal nevi. If there’s pigmentation present in a nevoid melanoma it will be distributed heterogeneously. The dermoscopic finding of irregular blood vessels and/or crystalline structures tips the balance in favor of biopsy.
Dr. Marghoob referred dermatologists interested in the dermoscopic features of nevoid melanoma to a recent study by members of the International Dermoscopy Society; he was one of the authors (Br J Dermatol. 2015;172[4]:961-7).
Epidermotropic metastatic melanoma: These lesions lack the ABCDs, are often amelanotic, and look like nondescript papules. If such a lesion is new, especially in a patient with a history of melanoma, think epidermotropic metastatic melanoma.
Spanish dermatologists have described five different dermoscopic patterns of melanoma metastases in an informative retrospective study: blue nevus-like, angioma-like, nevus-like, vascular, and unspecific. The vascular type is the most common form, characterized by amelanotic papules 2-3 mm in diameter and tortuous corkscrew vessels within the lesions (Br J Dermatol. 2013;169[1]:91-9).
Dr. Marghoob reported having no financial conflicts of interest regarding his presentation. SDEF and this news organization are owned by the same parent company.
WAIKOLOA, HAWAII – Nodular melanoma is one of a handful of morphologic subtypes of melanoma that are difficult to diagnose in a timely way because they lack the conventional features associated with melanoma. But certain clinical and dermoscopic clues are helpful in avoiding misdiagnosis, Dr. Ashfaq A. Marghoob said at the Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
He provided tips on how to recognize nodular melanoma and other difficult-to-diagnose morphologic subtypes, including desmoplastic, amelanotic, nevoid, and epidermotropic metastatic melanoma.
These diagnostically challenging melanoma subtypes have several things in common: clinically, they don’t look like melanomas, and dermoscopically, they display specific tell-tale features that speak of malignancy, including atypical polymorphous blood vessels and crystalline structures. Dermoscopy can be a big help in deciding whether to biopsy a clinically unremarkable-looking lesion, observed Dr. Marghoob of Memorial Sloan Kettering Cancer Center in New York.
Here’s the rundown on how to avoid delayed diagnosis of these melanomas:
Nodular melanoma: The big challenge posed by nodular melanoma is that it grows at a very high rate, a median of 0.5 mm per month, “so it’s really not a subtype of melanoma that’s amenable to screening,” the dermatologist said.
“Think about this: A patient comes in for screening today and you see nothing on their skin. Then a month from today they start a nodular melanoma. Their next appointment with you is a year from now. By the time they come back, that nodular melanoma is going to be a life-threatening cancer,” he explained. “It requires patient engagement in self-examination to bring these lesions to our attention, and we have to be perceptive enough to see these patients in a timely manner when they call. Otherwise these are melanomas that are really the killer tumors.”
Nodular melanomas often lack the “ABCD” features associated with most melanomas: asymmetry, border irregularity, color, and diameter greater than one-quarter of an inch. Instead, the lesions may exhibit the modified ABCDs described in pediatric melanoma: amelanotic, bump showing symmetry, color homogeneity in a shade of either pink or blue-black, and de novo, meaning the lesion didn’t arise in association with a nevus, Dr. Marghoob continued.
Roughly 60% of nodular melanomas are pigmented, the other 40% amelanotic. Dermoscopically, pigmented nodular melanomas are characterized by a greater degree of symmetry than that seen with other melanomas. Atypical vessels that exhibit tortuosity, a cork screw pattern, or an irregular hairpin shape are prominently visible. The lesions are multicolored, often blue-black, and may exhibit a blue-white veil. Notably absent are regression structures, streaks, and networks.
An amelanotic nodular melanoma also features prominent atypical vessels, symmetry, and sometimes crystalline structures, which appear as shiny white lines. Comma vessels and arborizing vessels are absent.
A more detailed description of the dermoscopic findings in nodular melanoma is available in a study coauthored by Dr. Marghoob (JAMA Dermatol. 2013;149[6]:699-709).
Desmoplastic melanoma: Seventy percent of desmoplastic melanomas are amelanotic. They often present as an unremarkable firm subcutaneous papule or nodule. Indeed, desmoplastic melanomas often have such a banal clinical appearance that in one-third of cases, melanoma is not even in the differential diagnosis.
“You’re thinking cyst, lipoma, dermatofibroma ... something other than a malignancy,” Dr. Marghoob said.
Suspicion is raised by a benign-looking lesion that’s symptomatic and/or exhibiting growth on chronically sun-damaged skin in an older patient. A scar with no history of trauma that’s located on chronically sun-damaged skin is another tipoff. Also, desmoplastic melanoma can arise in association with a lentigo maligna component.
“It is this knowledge of the association between lentigo maligna and desmoplastic melanoma that for the first time has enabled us to diagnose desmoplastic melanomas that are thin – not 5 or 10 mm, but more like 0.6 mm in thickness,” Dr. Marghoob said.
The main dermoscopic clue is the presence of atypical vascular structures.
For a fuller account of the dermoscopic characteristics of desmoplastic melanomas, Dr. Marghoob recommended a study he coauthored (JAMA Dermatol. 2013;149[4]:413-21).
Amelanotic melanoma: An indication of how difficult these cancers are to diagnose was provided by the international Genes, Environment, and Melanoma Study of nearly 3,500 invasive melanomas, 8% of which were amelanotic. They were at a more advanced tumor stage at diagnosis. As a result, the mortality risk during a median 7.6 years of follow-up was an adjusted twofold greater for patients with amelanotic as compared with pigmented melanomas (JAMA Dermatol. 2014;150[12]:1306-314).
Dr. Marghoob coauthored a retrospective review of 20 consecutively diagnosed amelanotic melanomas. The lesions lacked any of the classic ABCD features. Most were symmetric, oval to round in shape, had regular borders, and were pink in color. Dermoscopically, a consistent finding was a polymorphous vascular pattern (J Eur Acad Dermatol Venereol. 2012;26[5]:591-6).
In an earlier five-continent study comparing dermoscopic features of 222 amelanotic melanomas, 105 pigmented melanomas, and 170 benign melanocytic lesions, investigators came up with a simple model that distinguished melanomas from nonmelanomas with 70% sensitivity and 56% specificity. The take-home message: Amelanotic melanomas often feature more than one shade of pink upon dermoscopic examination, along with dotted and linear irregular vessels and predominant central vessels (Arch Dermatol. 2008;144[9]:1120-7).
Nevoid melanoma: Clinically they often resemble an intradermal nevus, and they’re often amelanotic. Dermoscopically, however, an intradermal nevus displays a mammillated surface, with each individual mammillated area being associated with an isolated comma or hairpin vessel. In contrast, the surface of a nevoid melanoma is more undulating in appearance and lacks the classic vascular pattern seen in intradermal nevi. If there’s pigmentation present in a nevoid melanoma it will be distributed heterogeneously. The dermoscopic finding of irregular blood vessels and/or crystalline structures tips the balance in favor of biopsy.
Dr. Marghoob referred dermatologists interested in the dermoscopic features of nevoid melanoma to a recent study by members of the International Dermoscopy Society; he was one of the authors (Br J Dermatol. 2015;172[4]:961-7).
Epidermotropic metastatic melanoma: These lesions lack the ABCDs, are often amelanotic, and look like nondescript papules. If such a lesion is new, especially in a patient with a history of melanoma, think epidermotropic metastatic melanoma.
Spanish dermatologists have described five different dermoscopic patterns of melanoma metastases in an informative retrospective study: blue nevus-like, angioma-like, nevus-like, vascular, and unspecific. The vascular type is the most common form, characterized by amelanotic papules 2-3 mm in diameter and tortuous corkscrew vessels within the lesions (Br J Dermatol. 2013;169[1]:91-9).
Dr. Marghoob reported having no financial conflicts of interest regarding his presentation. SDEF and this news organization are owned by the same parent company.
EXPERT ANALYSIS FROM SDEF HAWAII DERMATOLOGY SEMINAR
ACC survey: Women remain underrepresented in cardiology
CHICAGO – The latest American College of Cardiology Professional Life Survey paints a picture of a medical specialty with an aging workforce, low numbers of women, a growing preoccupation with career burnout, and large changes in practice settings in recent years.
“I’m very concerned that we haven’t seen much growth in the number of women in adult cardiology. Twenty years ago we acknowledged a need to increase the number of women in cardiology, and 10 years ago we saw an increase, but we’ve hit a wall,” Dr. Sandra J. Lewis said in presenting the survey results at the annual meeting of the American College of Cardiology.
Women comprise 50% of all medical school graduates, 47% of internal medicine residents, but only 11% of adult cardiologists, noted Dr. Lewis, chair of the ACC Women in Cardiology section, which conducted the survey in the fall of 2015.
It was completed by 1,349 male and 964 female cardiologists. This was the third such ACC survey; the others were done in 1996 and 2006.
The 2015 survey was the first to incorporate a section on burnout. Only 20% of female cardiologists and 26% of male cardiologists reported having no symptoms of burnout; the sex difference was statistically significant. Twenty-two percent of women and 17% of men characterized themselves as having “definite burnout,” noted Dr. Lewis, a cardiologist in group practice in Portland, Ore.
Recurring major sources of concern among both men and women were hassles related to electronic medical records, issues pertaining to Maintenance of Certification and the American Board of Internal Medicine, and the challenge of finding a suitable work-life balance.
On the other hand, 88% of female and 90% of male cardiologists pronounced themselves moderately or very satisfied with their work, and 63% of women and 65% of men indicated they were moderately or very satisfied with their compensation. In addition, 72% of women and 74% of men said they would recommend cardiology as a career.
About 65% of women reported experiencing past career discrimination, such as being passed over for a promotion, compared with just 23% of men. Furthermore, 26% of female cardiologists indicated their level of career advancement was slower than their peers, as did 8% of male cardiologists.
Other areas where significant sex-related differences were identified included subspecialty choice and practice pattern.
The shrinkage of private practice cardiology during the past 2 decades was impressive, Dr. Lewis said. In the 1996 survey, 53% of female cardiologists were in private practice. In 2006, it was down to 35%. And in the 2015 survey, only 17% of female cardiologists were in private practice. Similarly, 73% of male cardiologists were in private practice in 1996, 56% in 2006, and 23% in 2015.
In the latest survey, 18% of female and 17% of male respondents were aged 60 or older. In the 1996 survey, that was the case for 6% of women and 11% of men.
Dr. Lewis reported having no relevant financial conflicts.
CHICAGO – The latest American College of Cardiology Professional Life Survey paints a picture of a medical specialty with an aging workforce, low numbers of women, a growing preoccupation with career burnout, and large changes in practice settings in recent years.
“I’m very concerned that we haven’t seen much growth in the number of women in adult cardiology. Twenty years ago we acknowledged a need to increase the number of women in cardiology, and 10 years ago we saw an increase, but we’ve hit a wall,” Dr. Sandra J. Lewis said in presenting the survey results at the annual meeting of the American College of Cardiology.
Women comprise 50% of all medical school graduates, 47% of internal medicine residents, but only 11% of adult cardiologists, noted Dr. Lewis, chair of the ACC Women in Cardiology section, which conducted the survey in the fall of 2015.
It was completed by 1,349 male and 964 female cardiologists. This was the third such ACC survey; the others were done in 1996 and 2006.
The 2015 survey was the first to incorporate a section on burnout. Only 20% of female cardiologists and 26% of male cardiologists reported having no symptoms of burnout; the sex difference was statistically significant. Twenty-two percent of women and 17% of men characterized themselves as having “definite burnout,” noted Dr. Lewis, a cardiologist in group practice in Portland, Ore.
Recurring major sources of concern among both men and women were hassles related to electronic medical records, issues pertaining to Maintenance of Certification and the American Board of Internal Medicine, and the challenge of finding a suitable work-life balance.
On the other hand, 88% of female and 90% of male cardiologists pronounced themselves moderately or very satisfied with their work, and 63% of women and 65% of men indicated they were moderately or very satisfied with their compensation. In addition, 72% of women and 74% of men said they would recommend cardiology as a career.
About 65% of women reported experiencing past career discrimination, such as being passed over for a promotion, compared with just 23% of men. Furthermore, 26% of female cardiologists indicated their level of career advancement was slower than their peers, as did 8% of male cardiologists.
Other areas where significant sex-related differences were identified included subspecialty choice and practice pattern.
The shrinkage of private practice cardiology during the past 2 decades was impressive, Dr. Lewis said. In the 1996 survey, 53% of female cardiologists were in private practice. In 2006, it was down to 35%. And in the 2015 survey, only 17% of female cardiologists were in private practice. Similarly, 73% of male cardiologists were in private practice in 1996, 56% in 2006, and 23% in 2015.
In the latest survey, 18% of female and 17% of male respondents were aged 60 or older. In the 1996 survey, that was the case for 6% of women and 11% of men.
Dr. Lewis reported having no relevant financial conflicts.
CHICAGO – The latest American College of Cardiology Professional Life Survey paints a picture of a medical specialty with an aging workforce, low numbers of women, a growing preoccupation with career burnout, and large changes in practice settings in recent years.
“I’m very concerned that we haven’t seen much growth in the number of women in adult cardiology. Twenty years ago we acknowledged a need to increase the number of women in cardiology, and 10 years ago we saw an increase, but we’ve hit a wall,” Dr. Sandra J. Lewis said in presenting the survey results at the annual meeting of the American College of Cardiology.
Women comprise 50% of all medical school graduates, 47% of internal medicine residents, but only 11% of adult cardiologists, noted Dr. Lewis, chair of the ACC Women in Cardiology section, which conducted the survey in the fall of 2015.
It was completed by 1,349 male and 964 female cardiologists. This was the third such ACC survey; the others were done in 1996 and 2006.
The 2015 survey was the first to incorporate a section on burnout. Only 20% of female cardiologists and 26% of male cardiologists reported having no symptoms of burnout; the sex difference was statistically significant. Twenty-two percent of women and 17% of men characterized themselves as having “definite burnout,” noted Dr. Lewis, a cardiologist in group practice in Portland, Ore.
Recurring major sources of concern among both men and women were hassles related to electronic medical records, issues pertaining to Maintenance of Certification and the American Board of Internal Medicine, and the challenge of finding a suitable work-life balance.
On the other hand, 88% of female and 90% of male cardiologists pronounced themselves moderately or very satisfied with their work, and 63% of women and 65% of men indicated they were moderately or very satisfied with their compensation. In addition, 72% of women and 74% of men said they would recommend cardiology as a career.
About 65% of women reported experiencing past career discrimination, such as being passed over for a promotion, compared with just 23% of men. Furthermore, 26% of female cardiologists indicated their level of career advancement was slower than their peers, as did 8% of male cardiologists.
Other areas where significant sex-related differences were identified included subspecialty choice and practice pattern.
The shrinkage of private practice cardiology during the past 2 decades was impressive, Dr. Lewis said. In the 1996 survey, 53% of female cardiologists were in private practice. In 2006, it was down to 35%. And in the 2015 survey, only 17% of female cardiologists were in private practice. Similarly, 73% of male cardiologists were in private practice in 1996, 56% in 2006, and 23% in 2015.
In the latest survey, 18% of female and 17% of male respondents were aged 60 or older. In the 1996 survey, that was the case for 6% of women and 11% of men.
Dr. Lewis reported having no relevant financial conflicts.
AT ACC 16















