User login
Reassurance on cardiovascular safety of Breo Ellipta for COPD
CHICAGO – Using an inhaled long-acting beta agonist and corticosteroid to treat patients with moderate chronic obstructive pulmonary disease who have known cardiovascular disease had no impact on the cardiovascular event rate in the landmark SUMMIT trial, Dr. David E. Newby reported at the annual meeting of the American College of Cardiology.
SUMMIT (the Study to Understand Mortality and Morbidity) was a randomized, double-blind, placebo-controlled, 43-country, four-arm clinical trial in which 16,485 patients with moderate COPD were placed on once-daily inhaled fluticasone furoate/vilanterol 100/25 mcg by dry powder inhaler (Breo Ellipta), either drug alone, or placebo for an average of 2 years. Two-thirds of participants had overt cardiovascular disease; the rest were at high risk as defined by age greater than 60 years plus the presence of two or more cardiovascular risk factors.
This was the first large prospective clinical trial to investigate the impact of respiratory therapy on survival in patients with two commonly comorbid conditions. The incidence of major adverse cardiovascular events – a prespecified secondary endpoint – was of major interest because some other long-acting beta agonists have been linked to increased cardiovascular risk, explained Dr. Newby of the University of Edinburgh.
The primary endpoint in SUMMIT was all-cause mortality. The 12.2% relative risk reduction in the fluticasone furoate/vilanterol group, compared with placebo, didn’t achieve statistical significance. Neither did the 7.4% reduction in the secondary composite endpoint of cardiovascular death, MI, stroke, unstable angina, or TIA seen in the corticosteroid/LABA group, which is reassuring from a safety standpoint, he noted.
A positive result was seen for another secondary endpoint: the rate of lung function decline as measured by forced expiratory volume in 1 second. The rate of decline was 8 mL/year less with fluticasone furoate/vilanterol, compared with placebo.
The Breo Ellipta group also had significantly lower rates of moderate or severe COPD exacerbations, hospitalization for exacerbations, and improved quality of life as measured by the St. George’s Respiratory Questionnaire – COPD total score at 12 months.
The SUMMIT trial was sponsored by GlaxoSmithKline. The presenter is a consultant to GSK and eight other pharmaceutical companies.
CHICAGO – Using an inhaled long-acting beta agonist and corticosteroid to treat patients with moderate chronic obstructive pulmonary disease who have known cardiovascular disease had no impact on the cardiovascular event rate in the landmark SUMMIT trial, Dr. David E. Newby reported at the annual meeting of the American College of Cardiology.
SUMMIT (the Study to Understand Mortality and Morbidity) was a randomized, double-blind, placebo-controlled, 43-country, four-arm clinical trial in which 16,485 patients with moderate COPD were placed on once-daily inhaled fluticasone furoate/vilanterol 100/25 mcg by dry powder inhaler (Breo Ellipta), either drug alone, or placebo for an average of 2 years. Two-thirds of participants had overt cardiovascular disease; the rest were at high risk as defined by age greater than 60 years plus the presence of two or more cardiovascular risk factors.
This was the first large prospective clinical trial to investigate the impact of respiratory therapy on survival in patients with two commonly comorbid conditions. The incidence of major adverse cardiovascular events – a prespecified secondary endpoint – was of major interest because some other long-acting beta agonists have been linked to increased cardiovascular risk, explained Dr. Newby of the University of Edinburgh.
The primary endpoint in SUMMIT was all-cause mortality. The 12.2% relative risk reduction in the fluticasone furoate/vilanterol group, compared with placebo, didn’t achieve statistical significance. Neither did the 7.4% reduction in the secondary composite endpoint of cardiovascular death, MI, stroke, unstable angina, or TIA seen in the corticosteroid/LABA group, which is reassuring from a safety standpoint, he noted.
A positive result was seen for another secondary endpoint: the rate of lung function decline as measured by forced expiratory volume in 1 second. The rate of decline was 8 mL/year less with fluticasone furoate/vilanterol, compared with placebo.
The Breo Ellipta group also had significantly lower rates of moderate or severe COPD exacerbations, hospitalization for exacerbations, and improved quality of life as measured by the St. George’s Respiratory Questionnaire – COPD total score at 12 months.
The SUMMIT trial was sponsored by GlaxoSmithKline. The presenter is a consultant to GSK and eight other pharmaceutical companies.
CHICAGO – Using an inhaled long-acting beta agonist and corticosteroid to treat patients with moderate chronic obstructive pulmonary disease who have known cardiovascular disease had no impact on the cardiovascular event rate in the landmark SUMMIT trial, Dr. David E. Newby reported at the annual meeting of the American College of Cardiology.
SUMMIT (the Study to Understand Mortality and Morbidity) was a randomized, double-blind, placebo-controlled, 43-country, four-arm clinical trial in which 16,485 patients with moderate COPD were placed on once-daily inhaled fluticasone furoate/vilanterol 100/25 mcg by dry powder inhaler (Breo Ellipta), either drug alone, or placebo for an average of 2 years. Two-thirds of participants had overt cardiovascular disease; the rest were at high risk as defined by age greater than 60 years plus the presence of two or more cardiovascular risk factors.
This was the first large prospective clinical trial to investigate the impact of respiratory therapy on survival in patients with two commonly comorbid conditions. The incidence of major adverse cardiovascular events – a prespecified secondary endpoint – was of major interest because some other long-acting beta agonists have been linked to increased cardiovascular risk, explained Dr. Newby of the University of Edinburgh.
The primary endpoint in SUMMIT was all-cause mortality. The 12.2% relative risk reduction in the fluticasone furoate/vilanterol group, compared with placebo, didn’t achieve statistical significance. Neither did the 7.4% reduction in the secondary composite endpoint of cardiovascular death, MI, stroke, unstable angina, or TIA seen in the corticosteroid/LABA group, which is reassuring from a safety standpoint, he noted.
A positive result was seen for another secondary endpoint: the rate of lung function decline as measured by forced expiratory volume in 1 second. The rate of decline was 8 mL/year less with fluticasone furoate/vilanterol, compared with placebo.
The Breo Ellipta group also had significantly lower rates of moderate or severe COPD exacerbations, hospitalization for exacerbations, and improved quality of life as measured by the St. George’s Respiratory Questionnaire – COPD total score at 12 months.
The SUMMIT trial was sponsored by GlaxoSmithKline. The presenter is a consultant to GSK and eight other pharmaceutical companies.
AT ACC 16
Key clinical point: Once-daily inhaled fluticasone furoate/vilanterol 100/25 mcg to treat moderate COPD in patients with comorbid cardiovascular disease did not increase their risk of cardiovascular events.
Major finding: The major adverse cardiovascular event rate in patients on once-daily inhaled fluticasone furoate/vilanterol 100/25 mcg was 7.4% less than with placebo, a nonsignificant difference.
Data source: This was a randomized, double-blind, placebo-controlled, 2-year clinical trial including 16,485 patients with moderate COPD and overt cardiovascular disease or at high risk for it.
Disclosures: The SUMMIT trial was sponsored by GlaxoSmithKline. The presenter is a consultant to GSK and eight other pharmaceutical companies.
Novel device therapy shows continued benefits in pediatric peanut allergy
LOS ANGELES – A peanut protein–bearing skin patch known as the Viaskin Peanut gave a continued strong performance for treatment of peanut allergy in children during the second year of an international study of this novel form of epicutaneous immunotherapy.
The clinical response rate in 6- to 11-year-olds after 1 year of treatment with the 250-mcg dose of peanut protein in the medical device was 57% in the phase IIb, double-blind, 22-site, international VIPES trial, as reported last year.
After an additional year of treatment with the 250-mcg Viaskin Peanut in the open-label extension study known as OLFUS-VIPES, this rate climbed to 80%. Safety and tolerability of the device therapy remained excellent, Dr. Hugh A. Sampson said at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
In adolescents and adults, however, the clinical response – while significantly better than placebo in VIPES – was less robust than in children, and it remained stable from year 1 to year 2. This is believed to be due to the greater plasticity of the immune system in children, observed Dr. Sampson, director of the Jaffe Food Allergy Institute at Kravis Children’s Hospital at Mount Sinai in New York and chief scientific officer at DBV Technologies, which is developing the Viaskin Peanut.
The ongoing phase III trial uses the 250-mcg dose of peanut protein – the highest of several doses studied in VIPES and OLFUS-VIPES – and is restricted to peanut-allergic children ages 4-11 years. Doses of peanut protein greater than 250 mcg will be explored in separate studies of adolescents and adults.
The clinical response rate in children on the 250-mcg Viaskin Peanut rose from 57% after 1 year to 80% – that is, 16 of 20 subjects – after 2 years. A clinical response in VIPES and OLFUS-VIPES was defined as nonreactivity to a dose of at least 1,000 mg of peanut protein – the equivalent of four peanuts – during a formal double-blind food challenge or at least a tenfold increase in the eliciting dose, compared to the original eliciting dose.
In VIPES, one-third of children on the 250-mcg device therapy for 1 year could tolerate at least 1,000 mg of peanut protein; after an additional year of open-label therapy, 60% of children were able to do so.
Among 6- to 11-year-olds, the median cumulative reactive dose of peanut protein was 44 mg at baseline, 444 mg after 12 months of using the 250-mcg Viaskin Peanut, and 1,444 mg at 2 years.
The children’s immunologic response to the Viaskin Peanut was impressive: A 40% reduction from baseline in peanut IgE at 2 years, along with a ninefold increase in protective peanut-specific IgG4.
The skin patch consists of a dried allergen – in this case, peanut protein – which is made electrostatically adherent to a membrane on a Band-Aid–like chamber. A set of patches is placed on noneczematous skin on a child’s back and on the inner upper arm of older patients. Moisture emitted from the skin gradually causes the protein allergen to solubilize and get absorbed into the outer layer of the skin. It is then picked up by antigen-presenting Langerhans cells and transported to regional lymph nodes for deactivation. The patches are changed daily.
“It appears that we need to look at the skin as a tolerogenic organ when it’s uninflamed,” Dr. Sampson observed.
Compliance with treatment was in excess of 96% in both VIPES and OLFUS-VIPES. There have been no serious treatment-related adverse events and no need for the use of epinephrine. Side effects have been limited to occasional mild to moderate application site reactions easily managed with antihistamines and/or topical steroids, according to Dr. Sampson.
The double-blind VIPES study included 207 subjects with documented peanut allergy. OLFUS-VIPES, which will continue for 1 additional year of open-label therapy, includes 171 of the original 207, including 97 children, 49 adolescents, and 25 adults up to age 55 years.
“We’ll see if there’s continued improvement in children through the third year or it levels off, but based upon the immunologic parameters I think it’s having continued effect,” the pediatric allergist said.
LOS ANGELES – A peanut protein–bearing skin patch known as the Viaskin Peanut gave a continued strong performance for treatment of peanut allergy in children during the second year of an international study of this novel form of epicutaneous immunotherapy.
The clinical response rate in 6- to 11-year-olds after 1 year of treatment with the 250-mcg dose of peanut protein in the medical device was 57% in the phase IIb, double-blind, 22-site, international VIPES trial, as reported last year.
After an additional year of treatment with the 250-mcg Viaskin Peanut in the open-label extension study known as OLFUS-VIPES, this rate climbed to 80%. Safety and tolerability of the device therapy remained excellent, Dr. Hugh A. Sampson said at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
In adolescents and adults, however, the clinical response – while significantly better than placebo in VIPES – was less robust than in children, and it remained stable from year 1 to year 2. This is believed to be due to the greater plasticity of the immune system in children, observed Dr. Sampson, director of the Jaffe Food Allergy Institute at Kravis Children’s Hospital at Mount Sinai in New York and chief scientific officer at DBV Technologies, which is developing the Viaskin Peanut.
The ongoing phase III trial uses the 250-mcg dose of peanut protein – the highest of several doses studied in VIPES and OLFUS-VIPES – and is restricted to peanut-allergic children ages 4-11 years. Doses of peanut protein greater than 250 mcg will be explored in separate studies of adolescents and adults.
The clinical response rate in children on the 250-mcg Viaskin Peanut rose from 57% after 1 year to 80% – that is, 16 of 20 subjects – after 2 years. A clinical response in VIPES and OLFUS-VIPES was defined as nonreactivity to a dose of at least 1,000 mg of peanut protein – the equivalent of four peanuts – during a formal double-blind food challenge or at least a tenfold increase in the eliciting dose, compared to the original eliciting dose.
In VIPES, one-third of children on the 250-mcg device therapy for 1 year could tolerate at least 1,000 mg of peanut protein; after an additional year of open-label therapy, 60% of children were able to do so.
Among 6- to 11-year-olds, the median cumulative reactive dose of peanut protein was 44 mg at baseline, 444 mg after 12 months of using the 250-mcg Viaskin Peanut, and 1,444 mg at 2 years.
The children’s immunologic response to the Viaskin Peanut was impressive: A 40% reduction from baseline in peanut IgE at 2 years, along with a ninefold increase in protective peanut-specific IgG4.
The skin patch consists of a dried allergen – in this case, peanut protein – which is made electrostatically adherent to a membrane on a Band-Aid–like chamber. A set of patches is placed on noneczematous skin on a child’s back and on the inner upper arm of older patients. Moisture emitted from the skin gradually causes the protein allergen to solubilize and get absorbed into the outer layer of the skin. It is then picked up by antigen-presenting Langerhans cells and transported to regional lymph nodes for deactivation. The patches are changed daily.
“It appears that we need to look at the skin as a tolerogenic organ when it’s uninflamed,” Dr. Sampson observed.
Compliance with treatment was in excess of 96% in both VIPES and OLFUS-VIPES. There have been no serious treatment-related adverse events and no need for the use of epinephrine. Side effects have been limited to occasional mild to moderate application site reactions easily managed with antihistamines and/or topical steroids, according to Dr. Sampson.
The double-blind VIPES study included 207 subjects with documented peanut allergy. OLFUS-VIPES, which will continue for 1 additional year of open-label therapy, includes 171 of the original 207, including 97 children, 49 adolescents, and 25 adults up to age 55 years.
“We’ll see if there’s continued improvement in children through the third year or it levels off, but based upon the immunologic parameters I think it’s having continued effect,” the pediatric allergist said.
LOS ANGELES – A peanut protein–bearing skin patch known as the Viaskin Peanut gave a continued strong performance for treatment of peanut allergy in children during the second year of an international study of this novel form of epicutaneous immunotherapy.
The clinical response rate in 6- to 11-year-olds after 1 year of treatment with the 250-mcg dose of peanut protein in the medical device was 57% in the phase IIb, double-blind, 22-site, international VIPES trial, as reported last year.
After an additional year of treatment with the 250-mcg Viaskin Peanut in the open-label extension study known as OLFUS-VIPES, this rate climbed to 80%. Safety and tolerability of the device therapy remained excellent, Dr. Hugh A. Sampson said at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
In adolescents and adults, however, the clinical response – while significantly better than placebo in VIPES – was less robust than in children, and it remained stable from year 1 to year 2. This is believed to be due to the greater plasticity of the immune system in children, observed Dr. Sampson, director of the Jaffe Food Allergy Institute at Kravis Children’s Hospital at Mount Sinai in New York and chief scientific officer at DBV Technologies, which is developing the Viaskin Peanut.
The ongoing phase III trial uses the 250-mcg dose of peanut protein – the highest of several doses studied in VIPES and OLFUS-VIPES – and is restricted to peanut-allergic children ages 4-11 years. Doses of peanut protein greater than 250 mcg will be explored in separate studies of adolescents and adults.
The clinical response rate in children on the 250-mcg Viaskin Peanut rose from 57% after 1 year to 80% – that is, 16 of 20 subjects – after 2 years. A clinical response in VIPES and OLFUS-VIPES was defined as nonreactivity to a dose of at least 1,000 mg of peanut protein – the equivalent of four peanuts – during a formal double-blind food challenge or at least a tenfold increase in the eliciting dose, compared to the original eliciting dose.
In VIPES, one-third of children on the 250-mcg device therapy for 1 year could tolerate at least 1,000 mg of peanut protein; after an additional year of open-label therapy, 60% of children were able to do so.
Among 6- to 11-year-olds, the median cumulative reactive dose of peanut protein was 44 mg at baseline, 444 mg after 12 months of using the 250-mcg Viaskin Peanut, and 1,444 mg at 2 years.
The children’s immunologic response to the Viaskin Peanut was impressive: A 40% reduction from baseline in peanut IgE at 2 years, along with a ninefold increase in protective peanut-specific IgG4.
The skin patch consists of a dried allergen – in this case, peanut protein – which is made electrostatically adherent to a membrane on a Band-Aid–like chamber. A set of patches is placed on noneczematous skin on a child’s back and on the inner upper arm of older patients. Moisture emitted from the skin gradually causes the protein allergen to solubilize and get absorbed into the outer layer of the skin. It is then picked up by antigen-presenting Langerhans cells and transported to regional lymph nodes for deactivation. The patches are changed daily.
“It appears that we need to look at the skin as a tolerogenic organ when it’s uninflamed,” Dr. Sampson observed.
Compliance with treatment was in excess of 96% in both VIPES and OLFUS-VIPES. There have been no serious treatment-related adverse events and no need for the use of epinephrine. Side effects have been limited to occasional mild to moderate application site reactions easily managed with antihistamines and/or topical steroids, according to Dr. Sampson.
The double-blind VIPES study included 207 subjects with documented peanut allergy. OLFUS-VIPES, which will continue for 1 additional year of open-label therapy, includes 171 of the original 207, including 97 children, 49 adolescents, and 25 adults up to age 55 years.
“We’ll see if there’s continued improvement in children through the third year or it levels off, but based upon the immunologic parameters I think it’s having continued effect,” the pediatric allergist said.
AT 2016 AAAAI ANNUAL MEETING
Key clinical point: A peanut protein–bearing skin patch shows favorable efficacy and safety as a treatment for peanut allergy, especially in children.
Major finding: After 1 year using the Viaskin Peanut device at the 250-mcg dose, one-third of formerly peanut-allergic children tolerated at least 1,000 mg of peanut protein in an oral food challenge; after 2 years using the patch, the rate increased to 60%.
Data source: Ongoing 2-year, open-label extension of the yearlong, double-blind, randomized VIPES trial of 171 peanut-allergic subjects aged 6-55 years.
Disclosures: The study was funded by DBV Technologies and presented by the company’s chief scientific officer.
AF and Stroke May Be Temporally Related
CHICAGO – One-third of a large cohort of patients with an implantable cardiac device in place at the time of an ischemic stroke had one or more episodes of atrial fibrillation within the prior 30 days, Dr. Rhea C. Pimentel reported at the annual meeting of the American College of Cardiology.
The in-hospital mortality rate of these atrial fibrillation–related strokes was high: 11 of 42 (26%) such patients died during their stroke hospitalization, compared with 6 of 83 (7%) whose strokes were not temporally related to atrial fibrillation (AF), noted Dr. Pimentel, an electrophysiologist at the University of Kansas Medical Center in Kansas City.
Data from the Framingham Heart Study and other sources suggest that stroke in patients who have AF carries about double the mortality rate of strokes in patients without AF. Mortality associated with AF-related stroke in the Kansas study was probably so much higher because the hospital serves as a comprehensive stroke center, drawing patients from considerable distances across the Midwest, she said.
Dr. Pimentel reported on 125 patients who presented with an ischemic stroke when a cardiac monitoring device was in place. This is believed to be the largest such patient series ever reported. Their mean age was 73 years, and 41% were women. The mean CHADS2 score was 3.96, with a CHA2DS2-VASc score of 5.28. Of the patients, 62% had a pacemaker; the rest had an implantable cardioverter-defibrillator or cardiac resynchronization device. One-quarter of the group had a prior history of AF, and a fifth were on an oral anticoagulant – warfarin, in 70% of cases – at the time of their stroke.
The investigators defined a stroke-related AF episode as a total of at least 1 hour spent in AF during the 30 days preceding the stroke. Eighty percent of affected patients had paroxysmal AF. They typically fulfilled the 1-hour AF requirement via multiple short, self-terminated episodes rather than in an hour-long episode.
Being on an oral anticoagulant had no impact on in-hospital mortality, which was 14.2% in patients on warfarin or a newer anticoagulant and 14.3% in those who were not. Dr. Pimentel noted that she was presenting the results of the investigators’ initial look at the data. They are in the process of obtaining the patients’ international normalized ratio data, which “should be very enlightening,” she said.
She and her coinvestigators also plan to subdivide their 30-day study period into 5-day segments to learn just how soon after an AF episode the strokes occurred. Researchers at Stanford (Calif.) University have reported that the greatest stroke risk in patients with AF is in the first 5 days after an AF episode, and the Kansas group would like to confirm that observation.
In addition, because it remains an unresolved question whether any amount of AF is safe, Dr. Pimentel and her coworkers are considering reanalyzing their data using a cutoff of 6 minutes of AF rather than 1 hour during the 30 days prior to stroke.
The study was conducted free of commercial support. Dr. Pimentel reported having no relevant financial conflicts.
CHICAGO – One-third of a large cohort of patients with an implantable cardiac device in place at the time of an ischemic stroke had one or more episodes of atrial fibrillation within the prior 30 days, Dr. Rhea C. Pimentel reported at the annual meeting of the American College of Cardiology.
The in-hospital mortality rate of these atrial fibrillation–related strokes was high: 11 of 42 (26%) such patients died during their stroke hospitalization, compared with 6 of 83 (7%) whose strokes were not temporally related to atrial fibrillation (AF), noted Dr. Pimentel, an electrophysiologist at the University of Kansas Medical Center in Kansas City.
Data from the Framingham Heart Study and other sources suggest that stroke in patients who have AF carries about double the mortality rate of strokes in patients without AF. Mortality associated with AF-related stroke in the Kansas study was probably so much higher because the hospital serves as a comprehensive stroke center, drawing patients from considerable distances across the Midwest, she said.
Dr. Pimentel reported on 125 patients who presented with an ischemic stroke when a cardiac monitoring device was in place. This is believed to be the largest such patient series ever reported. Their mean age was 73 years, and 41% were women. The mean CHADS2 score was 3.96, with a CHA2DS2-VASc score of 5.28. Of the patients, 62% had a pacemaker; the rest had an implantable cardioverter-defibrillator or cardiac resynchronization device. One-quarter of the group had a prior history of AF, and a fifth were on an oral anticoagulant – warfarin, in 70% of cases – at the time of their stroke.
The investigators defined a stroke-related AF episode as a total of at least 1 hour spent in AF during the 30 days preceding the stroke. Eighty percent of affected patients had paroxysmal AF. They typically fulfilled the 1-hour AF requirement via multiple short, self-terminated episodes rather than in an hour-long episode.
Being on an oral anticoagulant had no impact on in-hospital mortality, which was 14.2% in patients on warfarin or a newer anticoagulant and 14.3% in those who were not. Dr. Pimentel noted that she was presenting the results of the investigators’ initial look at the data. They are in the process of obtaining the patients’ international normalized ratio data, which “should be very enlightening,” she said.
She and her coinvestigators also plan to subdivide their 30-day study period into 5-day segments to learn just how soon after an AF episode the strokes occurred. Researchers at Stanford (Calif.) University have reported that the greatest stroke risk in patients with AF is in the first 5 days after an AF episode, and the Kansas group would like to confirm that observation.
In addition, because it remains an unresolved question whether any amount of AF is safe, Dr. Pimentel and her coworkers are considering reanalyzing their data using a cutoff of 6 minutes of AF rather than 1 hour during the 30 days prior to stroke.
The study was conducted free of commercial support. Dr. Pimentel reported having no relevant financial conflicts.
CHICAGO – One-third of a large cohort of patients with an implantable cardiac device in place at the time of an ischemic stroke had one or more episodes of atrial fibrillation within the prior 30 days, Dr. Rhea C. Pimentel reported at the annual meeting of the American College of Cardiology.
The in-hospital mortality rate of these atrial fibrillation–related strokes was high: 11 of 42 (26%) such patients died during their stroke hospitalization, compared with 6 of 83 (7%) whose strokes were not temporally related to atrial fibrillation (AF), noted Dr. Pimentel, an electrophysiologist at the University of Kansas Medical Center in Kansas City.
Data from the Framingham Heart Study and other sources suggest that stroke in patients who have AF carries about double the mortality rate of strokes in patients without AF. Mortality associated with AF-related stroke in the Kansas study was probably so much higher because the hospital serves as a comprehensive stroke center, drawing patients from considerable distances across the Midwest, she said.
Dr. Pimentel reported on 125 patients who presented with an ischemic stroke when a cardiac monitoring device was in place. This is believed to be the largest such patient series ever reported. Their mean age was 73 years, and 41% were women. The mean CHADS2 score was 3.96, with a CHA2DS2-VASc score of 5.28. Of the patients, 62% had a pacemaker; the rest had an implantable cardioverter-defibrillator or cardiac resynchronization device. One-quarter of the group had a prior history of AF, and a fifth were on an oral anticoagulant – warfarin, in 70% of cases – at the time of their stroke.
The investigators defined a stroke-related AF episode as a total of at least 1 hour spent in AF during the 30 days preceding the stroke. Eighty percent of affected patients had paroxysmal AF. They typically fulfilled the 1-hour AF requirement via multiple short, self-terminated episodes rather than in an hour-long episode.
Being on an oral anticoagulant had no impact on in-hospital mortality, which was 14.2% in patients on warfarin or a newer anticoagulant and 14.3% in those who were not. Dr. Pimentel noted that she was presenting the results of the investigators’ initial look at the data. They are in the process of obtaining the patients’ international normalized ratio data, which “should be very enlightening,” she said.
She and her coinvestigators also plan to subdivide their 30-day study period into 5-day segments to learn just how soon after an AF episode the strokes occurred. Researchers at Stanford (Calif.) University have reported that the greatest stroke risk in patients with AF is in the first 5 days after an AF episode, and the Kansas group would like to confirm that observation.
In addition, because it remains an unresolved question whether any amount of AF is safe, Dr. Pimentel and her coworkers are considering reanalyzing their data using a cutoff of 6 minutes of AF rather than 1 hour during the 30 days prior to stroke.
The study was conducted free of commercial support. Dr. Pimentel reported having no relevant financial conflicts.
AT ACC 16
AF and stroke may be temporally related
CHICAGO – One-third of a large cohort of patients with an implantable cardiac device in place at the time of an ischemic stroke had one or more episodes of atrial fibrillation within the prior 30 days, Dr. Rhea C. Pimentel reported at the annual meeting of the American College of Cardiology.
The in-hospital mortality rate of these atrial fibrillation–related strokes was high: 11 of 42 (26%) such patients died during their stroke hospitalization, compared with 6 of 83 (7%) whose strokes were not temporally related to atrial fibrillation (AF), noted Dr. Pimentel, an electrophysiologist at the University of Kansas Medical Center in Kansas City.
Data from the Framingham Heart Study and other sources suggest that stroke in patients who have AF carries about double the mortality rate of strokes in patients without AF. Mortality associated with AF-related stroke in the Kansas study was probably so much higher because the hospital serves as a comprehensive stroke center, drawing patients from considerable distances across the Midwest, she said.
Dr. Pimentel reported on 125 patients who presented with an ischemic stroke when a cardiac monitoring device was in place. This is believed to be the largest such patient series ever reported. Their mean age was 73 years, and 41% were women. The mean CHADS2 score was 3.96, with a CHA2DS2-VASc score of 5.28. Of the patients, 62% had a pacemaker; the rest had an implantable cardioverter-defibrillator or cardiac resynchronization device. One-quarter of the group had a prior history of AF, and a fifth were on an oral anticoagulant – warfarin, in 70% of cases – at the time of their stroke.
The investigators defined a stroke-related AF episode as a total of at least 1 hour spent in AF during the 30 days preceding the stroke. Eighty percent of affected patients had paroxysmal AF. They typically fulfilled the 1-hour AF requirement via multiple short, self-terminated episodes rather than in an hour-long episode.
Being on an oral anticoagulant had no impact on in-hospital mortality, which was 14.2% in patients on warfarin or a newer anticoagulant and 14.3% in those who were not. Dr. Pimentel noted that she was presenting the results of the investigators’ initial look at the data. They are in the process of obtaining the patients’ international normalized ratio data, which “should be very enlightening,” she said.
She and her coinvestigators also plan to subdivide their 30-day study period into 5-day segments to learn just how soon after an AF episode the strokes occurred. Researchers at Stanford (Calif.) University have reported that the greatest stroke risk in patients with AF is in the first 5 days after an AF episode, and the Kansas group would like to confirm that observation.
In addition, because it remains an unresolved question whether any amount of AF is safe, Dr. Pimentel and her coworkers are considering reanalyzing their data using a cutoff of 6 minutes of AF rather than 1 hour during the 30 days prior to stroke.
The study was conducted free of commercial support. Dr. Pimentel reported having no relevant financial conflicts.
CHICAGO – One-third of a large cohort of patients with an implantable cardiac device in place at the time of an ischemic stroke had one or more episodes of atrial fibrillation within the prior 30 days, Dr. Rhea C. Pimentel reported at the annual meeting of the American College of Cardiology.
The in-hospital mortality rate of these atrial fibrillation–related strokes was high: 11 of 42 (26%) such patients died during their stroke hospitalization, compared with 6 of 83 (7%) whose strokes were not temporally related to atrial fibrillation (AF), noted Dr. Pimentel, an electrophysiologist at the University of Kansas Medical Center in Kansas City.
Data from the Framingham Heart Study and other sources suggest that stroke in patients who have AF carries about double the mortality rate of strokes in patients without AF. Mortality associated with AF-related stroke in the Kansas study was probably so much higher because the hospital serves as a comprehensive stroke center, drawing patients from considerable distances across the Midwest, she said.
Dr. Pimentel reported on 125 patients who presented with an ischemic stroke when a cardiac monitoring device was in place. This is believed to be the largest such patient series ever reported. Their mean age was 73 years, and 41% were women. The mean CHADS2 score was 3.96, with a CHA2DS2-VASc score of 5.28. Of the patients, 62% had a pacemaker; the rest had an implantable cardioverter-defibrillator or cardiac resynchronization device. One-quarter of the group had a prior history of AF, and a fifth were on an oral anticoagulant – warfarin, in 70% of cases – at the time of their stroke.
The investigators defined a stroke-related AF episode as a total of at least 1 hour spent in AF during the 30 days preceding the stroke. Eighty percent of affected patients had paroxysmal AF. They typically fulfilled the 1-hour AF requirement via multiple short, self-terminated episodes rather than in an hour-long episode.
Being on an oral anticoagulant had no impact on in-hospital mortality, which was 14.2% in patients on warfarin or a newer anticoagulant and 14.3% in those who were not. Dr. Pimentel noted that she was presenting the results of the investigators’ initial look at the data. They are in the process of obtaining the patients’ international normalized ratio data, which “should be very enlightening,” she said.
She and her coinvestigators also plan to subdivide their 30-day study period into 5-day segments to learn just how soon after an AF episode the strokes occurred. Researchers at Stanford (Calif.) University have reported that the greatest stroke risk in patients with AF is in the first 5 days after an AF episode, and the Kansas group would like to confirm that observation.
In addition, because it remains an unresolved question whether any amount of AF is safe, Dr. Pimentel and her coworkers are considering reanalyzing their data using a cutoff of 6 minutes of AF rather than 1 hour during the 30 days prior to stroke.
The study was conducted free of commercial support. Dr. Pimentel reported having no relevant financial conflicts.
CHICAGO – One-third of a large cohort of patients with an implantable cardiac device in place at the time of an ischemic stroke had one or more episodes of atrial fibrillation within the prior 30 days, Dr. Rhea C. Pimentel reported at the annual meeting of the American College of Cardiology.
The in-hospital mortality rate of these atrial fibrillation–related strokes was high: 11 of 42 (26%) such patients died during their stroke hospitalization, compared with 6 of 83 (7%) whose strokes were not temporally related to atrial fibrillation (AF), noted Dr. Pimentel, an electrophysiologist at the University of Kansas Medical Center in Kansas City.
Data from the Framingham Heart Study and other sources suggest that stroke in patients who have AF carries about double the mortality rate of strokes in patients without AF. Mortality associated with AF-related stroke in the Kansas study was probably so much higher because the hospital serves as a comprehensive stroke center, drawing patients from considerable distances across the Midwest, she said.
Dr. Pimentel reported on 125 patients who presented with an ischemic stroke when a cardiac monitoring device was in place. This is believed to be the largest such patient series ever reported. Their mean age was 73 years, and 41% were women. The mean CHADS2 score was 3.96, with a CHA2DS2-VASc score of 5.28. Of the patients, 62% had a pacemaker; the rest had an implantable cardioverter-defibrillator or cardiac resynchronization device. One-quarter of the group had a prior history of AF, and a fifth were on an oral anticoagulant – warfarin, in 70% of cases – at the time of their stroke.
The investigators defined a stroke-related AF episode as a total of at least 1 hour spent in AF during the 30 days preceding the stroke. Eighty percent of affected patients had paroxysmal AF. They typically fulfilled the 1-hour AF requirement via multiple short, self-terminated episodes rather than in an hour-long episode.
Being on an oral anticoagulant had no impact on in-hospital mortality, which was 14.2% in patients on warfarin or a newer anticoagulant and 14.3% in those who were not. Dr. Pimentel noted that she was presenting the results of the investigators’ initial look at the data. They are in the process of obtaining the patients’ international normalized ratio data, which “should be very enlightening,” she said.
She and her coinvestigators also plan to subdivide their 30-day study period into 5-day segments to learn just how soon after an AF episode the strokes occurred. Researchers at Stanford (Calif.) University have reported that the greatest stroke risk in patients with AF is in the first 5 days after an AF episode, and the Kansas group would like to confirm that observation.
In addition, because it remains an unresolved question whether any amount of AF is safe, Dr. Pimentel and her coworkers are considering reanalyzing their data using a cutoff of 6 minutes of AF rather than 1 hour during the 30 days prior to stroke.
The study was conducted free of commercial support. Dr. Pimentel reported having no relevant financial conflicts.
AT ACC 16
Key clinical point: One-third of patients with an ischemic stroke while they had an implantable cardiac device had atrial fibrillation in the 30 days prior.
Major finding: The in-hospital mortality rate was 26% in patients with AF-related stroke, compared with 7% in those without AF.
Data source: A single-center, retrospective study of 125 patients who had an ischemic stroke while wearing a pacemaker or other implanted cardiac device.
Disclosures: The study was conducted free of commercial support. The presenter reported having no financial conflicts of interest.
Acute Heart Failure Mortality Climbs With Severity of Peripheral Edema
CHICAGO – Breathlessness typically results in hospital admission for patients with heart failure, but peripheral edema is what prolongs their stay, according to Dr. John G.F. Cleland.
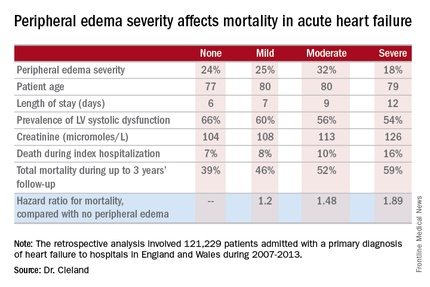
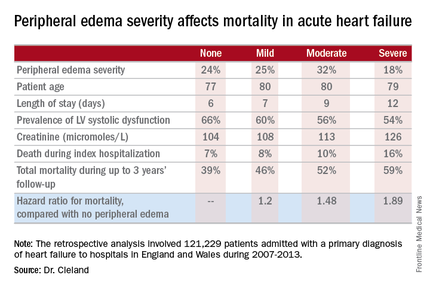
Moreover, it’s not only hospital length of stay that climbs with increasing severity of peripheral edema on admission. So does mortality, both during the index admission and long term, Dr. Cleland reported at the annual meeting of the American College of Cardiology.
He presented a retrospective analysis of 121,229 patients admitted with a primary diagnosis of heart failure to more than 90% of the hospitals in England and Wales during 2007-2013.
“It turns out that the majority of patients we’re seeing admitted with heart failure at U.K. hospitals are not admitted because of severe breathlessness at rest; they’re admitted for increasing fluid retention. They have lots of peripheral edema, which is actually associated with bad outcome. The patients who are very breathless tend to have the better outcome because they have left heart failure. The ones who are full of peripheral edema have more renal dysfunction and anemia, and they’re more likely to have right heart failure,” said Dr. Cleland, professor of cardiology at Imperial College London.
“I’m not saying the breathless group isn’t a target, but this group with peripheral edema is a bigger target – and we’re not designing trials to address their problems,” he added.
Compared with patients with no peripheral edema on hospital admission, the risk of mortality during that hospitalization and up to 3 years of subsequent follow-up was increased 1.2-fold in those with mild peripheral edema, 1.48-fold with moderate peripheral edema, and 1.89-fold in those with severe peripheral edema.
“We’re designing all the big clinical trials in acute heart failure to capture what I regard as neither fish nor fowl. They’re recruiting patients 6-12 hours after admission for acute heart failure. But by that point they’ve pretty well responded to their intravenous diuretics, and we’re just catching the tail end of their breathlessness. They’re not an emergency. The problem is really their peripheral edema, and that’s a day 2/day 3 problem. The first 6 hours of care really isn’t relevant to this group,” according to the internationally renowned heart failure researcher.
Indeed, he said he’d like to see the term “acute heart failure” laid to rest.
“If you think of acute decompensated heart failure, you think of patients wearing an oxygen mask coming in by an ambulance with blue lights flashing, and it’s an emergency. We need to move the mind-set. We shouldn’t call it acute heart failure at all, we should start talking about hospitalized heart failure, of which some is acute but much of it is subacute in people who have been deteriorating over several weeks and have just gotten to the point where they’re not coping at home anymore. They call a taxi or a friend who takes them to the hospital. Then they take a wheelchair from the taxi to the ER, but they don’t really need an ER at all,” Dr. Cleland said.
Many of these patients could be redirected to a different sort of facility at great cost savings, he added.
“In the United Kingdom and I think in the States, we’re now talking about furosemide lounges where, rather than admit the patient, you can bring them up as a day case, give them intravenous therapy, then [have them] go home at night, perhaps coming back for 3 or 4 days if needed. People are also now looking at home infusion services, and there’s a nice device for giving subcutaneous doses of furosemide as well,” the cardiologist said.
Apart from diuretics, there really aren’t any effective medications at present for peripheral edema in heart failure. But there are novel investigational agents worthy of evaluation, according to Dr. Cleland, including drugs aimed at improving mitochondrial function, agents that inhibit channels that allow edema to gather, and iron therapy.
The study was supported by the British Society for Heart Failure and the National Institute for Cardiovascular Outcomes Research. Dr. Cleland reported having no relevant financial conflicts.
CHICAGO – Breathlessness typically results in hospital admission for patients with heart failure, but peripheral edema is what prolongs their stay, according to Dr. John G.F. Cleland.
Moreover, it’s not only hospital length of stay that climbs with increasing severity of peripheral edema on admission. So does mortality, both during the index admission and long term, Dr. Cleland reported at the annual meeting of the American College of Cardiology.
He presented a retrospective analysis of 121,229 patients admitted with a primary diagnosis of heart failure to more than 90% of the hospitals in England and Wales during 2007-2013.
“It turns out that the majority of patients we’re seeing admitted with heart failure at U.K. hospitals are not admitted because of severe breathlessness at rest; they’re admitted for increasing fluid retention. They have lots of peripheral edema, which is actually associated with bad outcome. The patients who are very breathless tend to have the better outcome because they have left heart failure. The ones who are full of peripheral edema have more renal dysfunction and anemia, and they’re more likely to have right heart failure,” said Dr. Cleland, professor of cardiology at Imperial College London.
“I’m not saying the breathless group isn’t a target, but this group with peripheral edema is a bigger target – and we’re not designing trials to address their problems,” he added.
Compared with patients with no peripheral edema on hospital admission, the risk of mortality during that hospitalization and up to 3 years of subsequent follow-up was increased 1.2-fold in those with mild peripheral edema, 1.48-fold with moderate peripheral edema, and 1.89-fold in those with severe peripheral edema.

“We’re designing all the big clinical trials in acute heart failure to capture what I regard as neither fish nor fowl. They’re recruiting patients 6-12 hours after admission for acute heart failure. But by that point they’ve pretty well responded to their intravenous diuretics, and we’re just catching the tail end of their breathlessness. They’re not an emergency. The problem is really their peripheral edema, and that’s a day 2/day 3 problem. The first 6 hours of care really isn’t relevant to this group,” according to the internationally renowned heart failure researcher.
Indeed, he said he’d like to see the term “acute heart failure” laid to rest.
“If you think of acute decompensated heart failure, you think of patients wearing an oxygen mask coming in by an ambulance with blue lights flashing, and it’s an emergency. We need to move the mind-set. We shouldn’t call it acute heart failure at all, we should start talking about hospitalized heart failure, of which some is acute but much of it is subacute in people who have been deteriorating over several weeks and have just gotten to the point where they’re not coping at home anymore. They call a taxi or a friend who takes them to the hospital. Then they take a wheelchair from the taxi to the ER, but they don’t really need an ER at all,” Dr. Cleland said.
Many of these patients could be redirected to a different sort of facility at great cost savings, he added.
“In the United Kingdom and I think in the States, we’re now talking about furosemide lounges where, rather than admit the patient, you can bring them up as a day case, give them intravenous therapy, then [have them] go home at night, perhaps coming back for 3 or 4 days if needed. People are also now looking at home infusion services, and there’s a nice device for giving subcutaneous doses of furosemide as well,” the cardiologist said.
Apart from diuretics, there really aren’t any effective medications at present for peripheral edema in heart failure. But there are novel investigational agents worthy of evaluation, according to Dr. Cleland, including drugs aimed at improving mitochondrial function, agents that inhibit channels that allow edema to gather, and iron therapy.
The study was supported by the British Society for Heart Failure and the National Institute for Cardiovascular Outcomes Research. Dr. Cleland reported having no relevant financial conflicts.
CHICAGO – Breathlessness typically results in hospital admission for patients with heart failure, but peripheral edema is what prolongs their stay, according to Dr. John G.F. Cleland.
Moreover, it’s not only hospital length of stay that climbs with increasing severity of peripheral edema on admission. So does mortality, both during the index admission and long term, Dr. Cleland reported at the annual meeting of the American College of Cardiology.
He presented a retrospective analysis of 121,229 patients admitted with a primary diagnosis of heart failure to more than 90% of the hospitals in England and Wales during 2007-2013.
“It turns out that the majority of patients we’re seeing admitted with heart failure at U.K. hospitals are not admitted because of severe breathlessness at rest; they’re admitted for increasing fluid retention. They have lots of peripheral edema, which is actually associated with bad outcome. The patients who are very breathless tend to have the better outcome because they have left heart failure. The ones who are full of peripheral edema have more renal dysfunction and anemia, and they’re more likely to have right heart failure,” said Dr. Cleland, professor of cardiology at Imperial College London.
“I’m not saying the breathless group isn’t a target, but this group with peripheral edema is a bigger target – and we’re not designing trials to address their problems,” he added.
Compared with patients with no peripheral edema on hospital admission, the risk of mortality during that hospitalization and up to 3 years of subsequent follow-up was increased 1.2-fold in those with mild peripheral edema, 1.48-fold with moderate peripheral edema, and 1.89-fold in those with severe peripheral edema.

“We’re designing all the big clinical trials in acute heart failure to capture what I regard as neither fish nor fowl. They’re recruiting patients 6-12 hours after admission for acute heart failure. But by that point they’ve pretty well responded to their intravenous diuretics, and we’re just catching the tail end of their breathlessness. They’re not an emergency. The problem is really their peripheral edema, and that’s a day 2/day 3 problem. The first 6 hours of care really isn’t relevant to this group,” according to the internationally renowned heart failure researcher.
Indeed, he said he’d like to see the term “acute heart failure” laid to rest.
“If you think of acute decompensated heart failure, you think of patients wearing an oxygen mask coming in by an ambulance with blue lights flashing, and it’s an emergency. We need to move the mind-set. We shouldn’t call it acute heart failure at all, we should start talking about hospitalized heart failure, of which some is acute but much of it is subacute in people who have been deteriorating over several weeks and have just gotten to the point where they’re not coping at home anymore. They call a taxi or a friend who takes them to the hospital. Then they take a wheelchair from the taxi to the ER, but they don’t really need an ER at all,” Dr. Cleland said.
Many of these patients could be redirected to a different sort of facility at great cost savings, he added.
“In the United Kingdom and I think in the States, we’re now talking about furosemide lounges where, rather than admit the patient, you can bring them up as a day case, give them intravenous therapy, then [have them] go home at night, perhaps coming back for 3 or 4 days if needed. People are also now looking at home infusion services, and there’s a nice device for giving subcutaneous doses of furosemide as well,” the cardiologist said.
Apart from diuretics, there really aren’t any effective medications at present for peripheral edema in heart failure. But there are novel investigational agents worthy of evaluation, according to Dr. Cleland, including drugs aimed at improving mitochondrial function, agents that inhibit channels that allow edema to gather, and iron therapy.
The study was supported by the British Society for Heart Failure and the National Institute for Cardiovascular Outcomes Research. Dr. Cleland reported having no relevant financial conflicts.
AT ACC 16
Acute heart failure mortality climbs with severity of peripheral edema
CHICAGO – Breathlessness typically results in hospital admission for patients with heart failure, but peripheral edema is what prolongs their stay, according to Dr. John G.F. Cleland.
Moreover, it’s not only hospital length of stay that climbs with increasing severity of peripheral edema on admission. So does mortality, both during the index admission and long term, Dr. Cleland reported at the annual meeting of the American College of Cardiology.
He presented a retrospective analysis of 121,229 patients admitted with a primary diagnosis of heart failure to more than 90% of the hospitals in England and Wales during 2007-2013.
“It turns out that the majority of patients we’re seeing admitted with heart failure at U.K. hospitals are not admitted because of severe breathlessness at rest; they’re admitted for increasing fluid retention. They have lots of peripheral edema, which is actually associated with bad outcome. The patients who are very breathless tend to have the better outcome because they have left heart failure. The ones who are full of peripheral edema have more renal dysfunction and anemia, and they’re more likely to have right heart failure,” said Dr. Cleland, professor of cardiology at Imperial College London.
“I’m not saying the breathless group isn’t a target, but this group with peripheral edema is a bigger target – and we’re not designing trials to address their problems,” he added.
Compared with patients with no peripheral edema on hospital admission, the risk of mortality during that hospitalization and up to 3 years of subsequent follow-up was increased 1.2-fold in those with mild peripheral edema, 1.48-fold with moderate peripheral edema, and 1.89-fold in those with severe peripheral edema.
“We’re designing all the big clinical trials in acute heart failure to capture what I regard as neither fish nor fowl. They’re recruiting patients 6-12 hours after admission for acute heart failure. But by that point they’ve pretty well responded to their intravenous diuretics, and we’re just catching the tail end of their breathlessness. They’re not an emergency. The problem is really their peripheral edema, and that’s a day 2/day 3 problem. The first 6 hours of care really isn’t relevant to this group,” according to the internationally renowned heart failure researcher.
Indeed, he said he’d like to see the term “acute heart failure” laid to rest.
“If you think of acute decompensated heart failure, you think of patients wearing an oxygen mask coming in by an ambulance with blue lights flashing, and it’s an emergency. We need to move the mind-set. We shouldn’t call it acute heart failure at all, we should start talking about hospitalized heart failure, of which some is acute but much of it is subacute in people who have been deteriorating over several weeks and have just gotten to the point where they’re not coping at home anymore. They call a taxi or a friend who takes them to the hospital. Then they take a wheelchair from the taxi to the ER, but they don’t really need an ER at all,” Dr. Cleland said.
Many of these patients could be redirected to a different sort of facility at great cost savings, he added.
“In the United Kingdom and I think in the States, we’re now talking about furosemide lounges where, rather than admit the patient, you can bring them up as a day case, give them intravenous therapy, then [have them] go home at night, perhaps coming back for 3 or 4 days if needed. People are also now looking at home infusion services, and there’s a nice device for giving subcutaneous doses of furosemide as well,” the cardiologist said.
Apart from diuretics, there really aren’t any effective medications at present for peripheral edema in heart failure. But there are novel investigational agents worthy of evaluation, according to Dr. Cleland, including drugs aimed at improving mitochondrial function, agents that inhibit channels that allow edema to gather, and iron therapy.
The study was supported by the British Society for Heart Failure and the National Institute for Cardiovascular Outcomes Research. Dr. Cleland reported having no relevant financial conflicts.
CHICAGO – Breathlessness typically results in hospital admission for patients with heart failure, but peripheral edema is what prolongs their stay, according to Dr. John G.F. Cleland.
Moreover, it’s not only hospital length of stay that climbs with increasing severity of peripheral edema on admission. So does mortality, both during the index admission and long term, Dr. Cleland reported at the annual meeting of the American College of Cardiology.
He presented a retrospective analysis of 121,229 patients admitted with a primary diagnosis of heart failure to more than 90% of the hospitals in England and Wales during 2007-2013.
“It turns out that the majority of patients we’re seeing admitted with heart failure at U.K. hospitals are not admitted because of severe breathlessness at rest; they’re admitted for increasing fluid retention. They have lots of peripheral edema, which is actually associated with bad outcome. The patients who are very breathless tend to have the better outcome because they have left heart failure. The ones who are full of peripheral edema have more renal dysfunction and anemia, and they’re more likely to have right heart failure,” said Dr. Cleland, professor of cardiology at Imperial College London.
“I’m not saying the breathless group isn’t a target, but this group with peripheral edema is a bigger target – and we’re not designing trials to address their problems,” he added.
Compared with patients with no peripheral edema on hospital admission, the risk of mortality during that hospitalization and up to 3 years of subsequent follow-up was increased 1.2-fold in those with mild peripheral edema, 1.48-fold with moderate peripheral edema, and 1.89-fold in those with severe peripheral edema.

“We’re designing all the big clinical trials in acute heart failure to capture what I regard as neither fish nor fowl. They’re recruiting patients 6-12 hours after admission for acute heart failure. But by that point they’ve pretty well responded to their intravenous diuretics, and we’re just catching the tail end of their breathlessness. They’re not an emergency. The problem is really their peripheral edema, and that’s a day 2/day 3 problem. The first 6 hours of care really isn’t relevant to this group,” according to the internationally renowned heart failure researcher.
Indeed, he said he’d like to see the term “acute heart failure” laid to rest.
“If you think of acute decompensated heart failure, you think of patients wearing an oxygen mask coming in by an ambulance with blue lights flashing, and it’s an emergency. We need to move the mind-set. We shouldn’t call it acute heart failure at all, we should start talking about hospitalized heart failure, of which some is acute but much of it is subacute in people who have been deteriorating over several weeks and have just gotten to the point where they’re not coping at home anymore. They call a taxi or a friend who takes them to the hospital. Then they take a wheelchair from the taxi to the ER, but they don’t really need an ER at all,” Dr. Cleland said.
Many of these patients could be redirected to a different sort of facility at great cost savings, he added.
“In the United Kingdom and I think in the States, we’re now talking about furosemide lounges where, rather than admit the patient, you can bring them up as a day case, give them intravenous therapy, then [have them] go home at night, perhaps coming back for 3 or 4 days if needed. People are also now looking at home infusion services, and there’s a nice device for giving subcutaneous doses of furosemide as well,” the cardiologist said.
Apart from diuretics, there really aren’t any effective medications at present for peripheral edema in heart failure. But there are novel investigational agents worthy of evaluation, according to Dr. Cleland, including drugs aimed at improving mitochondrial function, agents that inhibit channels that allow edema to gather, and iron therapy.
The study was supported by the British Society for Heart Failure and the National Institute for Cardiovascular Outcomes Research. Dr. Cleland reported having no relevant financial conflicts.
CHICAGO – Breathlessness typically results in hospital admission for patients with heart failure, but peripheral edema is what prolongs their stay, according to Dr. John G.F. Cleland.
Moreover, it’s not only hospital length of stay that climbs with increasing severity of peripheral edema on admission. So does mortality, both during the index admission and long term, Dr. Cleland reported at the annual meeting of the American College of Cardiology.
He presented a retrospective analysis of 121,229 patients admitted with a primary diagnosis of heart failure to more than 90% of the hospitals in England and Wales during 2007-2013.
“It turns out that the majority of patients we’re seeing admitted with heart failure at U.K. hospitals are not admitted because of severe breathlessness at rest; they’re admitted for increasing fluid retention. They have lots of peripheral edema, which is actually associated with bad outcome. The patients who are very breathless tend to have the better outcome because they have left heart failure. The ones who are full of peripheral edema have more renal dysfunction and anemia, and they’re more likely to have right heart failure,” said Dr. Cleland, professor of cardiology at Imperial College London.
“I’m not saying the breathless group isn’t a target, but this group with peripheral edema is a bigger target – and we’re not designing trials to address their problems,” he added.
Compared with patients with no peripheral edema on hospital admission, the risk of mortality during that hospitalization and up to 3 years of subsequent follow-up was increased 1.2-fold in those with mild peripheral edema, 1.48-fold with moderate peripheral edema, and 1.89-fold in those with severe peripheral edema.

“We’re designing all the big clinical trials in acute heart failure to capture what I regard as neither fish nor fowl. They’re recruiting patients 6-12 hours after admission for acute heart failure. But by that point they’ve pretty well responded to their intravenous diuretics, and we’re just catching the tail end of their breathlessness. They’re not an emergency. The problem is really their peripheral edema, and that’s a day 2/day 3 problem. The first 6 hours of care really isn’t relevant to this group,” according to the internationally renowned heart failure researcher.
Indeed, he said he’d like to see the term “acute heart failure” laid to rest.
“If you think of acute decompensated heart failure, you think of patients wearing an oxygen mask coming in by an ambulance with blue lights flashing, and it’s an emergency. We need to move the mind-set. We shouldn’t call it acute heart failure at all, we should start talking about hospitalized heart failure, of which some is acute but much of it is subacute in people who have been deteriorating over several weeks and have just gotten to the point where they’re not coping at home anymore. They call a taxi or a friend who takes them to the hospital. Then they take a wheelchair from the taxi to the ER, but they don’t really need an ER at all,” Dr. Cleland said.
Many of these patients could be redirected to a different sort of facility at great cost savings, he added.
“In the United Kingdom and I think in the States, we’re now talking about furosemide lounges where, rather than admit the patient, you can bring them up as a day case, give them intravenous therapy, then [have them] go home at night, perhaps coming back for 3 or 4 days if needed. People are also now looking at home infusion services, and there’s a nice device for giving subcutaneous doses of furosemide as well,” the cardiologist said.
Apart from diuretics, there really aren’t any effective medications at present for peripheral edema in heart failure. But there are novel investigational agents worthy of evaluation, according to Dr. Cleland, including drugs aimed at improving mitochondrial function, agents that inhibit channels that allow edema to gather, and iron therapy.
The study was supported by the British Society for Heart Failure and the National Institute for Cardiovascular Outcomes Research. Dr. Cleland reported having no relevant financial conflicts.
AT ACC 16
Key clinical point: Leg swelling warrants greater attention in patients hospitalized for acute heart failure.
Major finding: In-hospital mortality was more than twice as great in patients admitted for acute heart failure with severe peripheral edema, compared with no leg swelling.
Data source: A retrospective study of more than 121,000 patients hospitalized for acute heart failure in England and Wales.
Disclosures: The study was supported by the British Society for Heart Failure and the National Institute for Cardiovascular Outcomes Research. The presenter reported having no relevant financial conflicts.
Valve hemodynamic deterioration 2.5% at 1 year
CHICAGO – The incidence of valve hemodynamic deterioration in the first year after transcatheter aortic valve replacement is about 2.5%, but this event wasn’t clearly associated with adverse clinical outcomes out to 18 months of follow-up in an analysis of the large U.S. registry collaboratively maintained by the Society of Thoracic Surgeons and the American College of Cardiology.
“These findings, especially the patient and procedural predictors of valve hemodynamic deterioration we identified, may help to inform TAVR care, including patient selection, surveillance, and preventive strategies,” Dr. Sreekanth Vemulapalli reported at the annual meeting of the American College of Cardiology.
Recent reports have linked TAVR to subsequent development of leaflet abnormalities and valve thrombosis, with widely ranging estimates of incidence. Definitive answers as to the true rate of these adverse events and the underlying mechanisms will come from ongoing prospective studies using advanced imaging via four-dimensional CT or transesophageal echocardiography, but those studies will take years to complete, noted Dr. Vemulapalli of the Duke Clinical Research Institute in Durham, N.C.
In the meantime, he continued, the STS/ACC Transcatheter Valve Therapy Registry provides a unique opportunity to shed light on the incidence and consequences of valve hemodynamic deterioration (VHD) in real-world clinical practice. The registry includes all commercial TAVR procedures performed in the United States, with transthoracic echocardiograms obtained pre- and post-TAVR, at 30 days, and at 1 year after the procedure.
To examine the short- and longer-term rates of VHD, which Dr. Vemulapalli and his coinvestigators defined as an increase in the mean aortic valve gradient of 10 mm or more, the researchers assembled two separate patient cohorts. They comprised a short-term–risk group of 10,095 patients who underwent TAVR at 334 sites, with an incidence of VHD of 2.1% during the first 30 days after the procedure, and 3,175 patients at 254 sites, whose incidence of VHD from day 30 through 1 year post TAVR was 2.5%.
The combined rate of VHD and all-cause mortality during the first 30 days was 7.1%. For the long-term cohort, the combined endpoint rate from day 30 to 1 year was 23.5%.
Importantly, the occurrence of VHD was not associated with an excess of the composite endpoint of mortality, stroke, heart failure hospitalization, or aortic valve reintervention at 1 year in either the short- or long-term cohort. The same held true in an analysis covering the period of 12-18 months post TAVR, according to Dr. Vemulapalli.
In a multivariate analysis, the significant predictors of VHD in the short-term cohort were male sex; increased body mass index, with the risk rising stepwise with every additional 5 kg/m above normal weight; baseline severe chronic lung disease; a valve-in-valve procedure; a larger baseline aortic valve gradient; a TAVR valve size of 23 mm or less; and severe patient/prosthesis mismatch.
In the long-term cohort, the risk factors for VHD were hospital discharge on a factor Xa inhibitor and a larger predischarge aortic valve gradient.
Change in left ventricular ejection fraction over the course of the study bore no relation to VHD risk. Neither did which of the two commercially available TAVR valves a patient received.
This study was funded by the American College of Cardiology’s National Cardiovascular Data Registry. Dr. Vemulapalli reported serving as a consultant to Novella and Premiere and receiving research grants from the Agency for Healthcare Research and Quality, Boston Scientific, Abbott Vascular, and the ACC.
CHICAGO – The incidence of valve hemodynamic deterioration in the first year after transcatheter aortic valve replacement is about 2.5%, but this event wasn’t clearly associated with adverse clinical outcomes out to 18 months of follow-up in an analysis of the large U.S. registry collaboratively maintained by the Society of Thoracic Surgeons and the American College of Cardiology.
“These findings, especially the patient and procedural predictors of valve hemodynamic deterioration we identified, may help to inform TAVR care, including patient selection, surveillance, and preventive strategies,” Dr. Sreekanth Vemulapalli reported at the annual meeting of the American College of Cardiology.
Recent reports have linked TAVR to subsequent development of leaflet abnormalities and valve thrombosis, with widely ranging estimates of incidence. Definitive answers as to the true rate of these adverse events and the underlying mechanisms will come from ongoing prospective studies using advanced imaging via four-dimensional CT or transesophageal echocardiography, but those studies will take years to complete, noted Dr. Vemulapalli of the Duke Clinical Research Institute in Durham, N.C.
In the meantime, he continued, the STS/ACC Transcatheter Valve Therapy Registry provides a unique opportunity to shed light on the incidence and consequences of valve hemodynamic deterioration (VHD) in real-world clinical practice. The registry includes all commercial TAVR procedures performed in the United States, with transthoracic echocardiograms obtained pre- and post-TAVR, at 30 days, and at 1 year after the procedure.
To examine the short- and longer-term rates of VHD, which Dr. Vemulapalli and his coinvestigators defined as an increase in the mean aortic valve gradient of 10 mm or more, the researchers assembled two separate patient cohorts. They comprised a short-term–risk group of 10,095 patients who underwent TAVR at 334 sites, with an incidence of VHD of 2.1% during the first 30 days after the procedure, and 3,175 patients at 254 sites, whose incidence of VHD from day 30 through 1 year post TAVR was 2.5%.
The combined rate of VHD and all-cause mortality during the first 30 days was 7.1%. For the long-term cohort, the combined endpoint rate from day 30 to 1 year was 23.5%.
Importantly, the occurrence of VHD was not associated with an excess of the composite endpoint of mortality, stroke, heart failure hospitalization, or aortic valve reintervention at 1 year in either the short- or long-term cohort. The same held true in an analysis covering the period of 12-18 months post TAVR, according to Dr. Vemulapalli.
In a multivariate analysis, the significant predictors of VHD in the short-term cohort were male sex; increased body mass index, with the risk rising stepwise with every additional 5 kg/m above normal weight; baseline severe chronic lung disease; a valve-in-valve procedure; a larger baseline aortic valve gradient; a TAVR valve size of 23 mm or less; and severe patient/prosthesis mismatch.
In the long-term cohort, the risk factors for VHD were hospital discharge on a factor Xa inhibitor and a larger predischarge aortic valve gradient.
Change in left ventricular ejection fraction over the course of the study bore no relation to VHD risk. Neither did which of the two commercially available TAVR valves a patient received.
This study was funded by the American College of Cardiology’s National Cardiovascular Data Registry. Dr. Vemulapalli reported serving as a consultant to Novella and Premiere and receiving research grants from the Agency for Healthcare Research and Quality, Boston Scientific, Abbott Vascular, and the ACC.
CHICAGO – The incidence of valve hemodynamic deterioration in the first year after transcatheter aortic valve replacement is about 2.5%, but this event wasn’t clearly associated with adverse clinical outcomes out to 18 months of follow-up in an analysis of the large U.S. registry collaboratively maintained by the Society of Thoracic Surgeons and the American College of Cardiology.
“These findings, especially the patient and procedural predictors of valve hemodynamic deterioration we identified, may help to inform TAVR care, including patient selection, surveillance, and preventive strategies,” Dr. Sreekanth Vemulapalli reported at the annual meeting of the American College of Cardiology.
Recent reports have linked TAVR to subsequent development of leaflet abnormalities and valve thrombosis, with widely ranging estimates of incidence. Definitive answers as to the true rate of these adverse events and the underlying mechanisms will come from ongoing prospective studies using advanced imaging via four-dimensional CT or transesophageal echocardiography, but those studies will take years to complete, noted Dr. Vemulapalli of the Duke Clinical Research Institute in Durham, N.C.
In the meantime, he continued, the STS/ACC Transcatheter Valve Therapy Registry provides a unique opportunity to shed light on the incidence and consequences of valve hemodynamic deterioration (VHD) in real-world clinical practice. The registry includes all commercial TAVR procedures performed in the United States, with transthoracic echocardiograms obtained pre- and post-TAVR, at 30 days, and at 1 year after the procedure.
To examine the short- and longer-term rates of VHD, which Dr. Vemulapalli and his coinvestigators defined as an increase in the mean aortic valve gradient of 10 mm or more, the researchers assembled two separate patient cohorts. They comprised a short-term–risk group of 10,095 patients who underwent TAVR at 334 sites, with an incidence of VHD of 2.1% during the first 30 days after the procedure, and 3,175 patients at 254 sites, whose incidence of VHD from day 30 through 1 year post TAVR was 2.5%.
The combined rate of VHD and all-cause mortality during the first 30 days was 7.1%. For the long-term cohort, the combined endpoint rate from day 30 to 1 year was 23.5%.
Importantly, the occurrence of VHD was not associated with an excess of the composite endpoint of mortality, stroke, heart failure hospitalization, or aortic valve reintervention at 1 year in either the short- or long-term cohort. The same held true in an analysis covering the period of 12-18 months post TAVR, according to Dr. Vemulapalli.
In a multivariate analysis, the significant predictors of VHD in the short-term cohort were male sex; increased body mass index, with the risk rising stepwise with every additional 5 kg/m above normal weight; baseline severe chronic lung disease; a valve-in-valve procedure; a larger baseline aortic valve gradient; a TAVR valve size of 23 mm or less; and severe patient/prosthesis mismatch.
In the long-term cohort, the risk factors for VHD were hospital discharge on a factor Xa inhibitor and a larger predischarge aortic valve gradient.
Change in left ventricular ejection fraction over the course of the study bore no relation to VHD risk. Neither did which of the two commercially available TAVR valves a patient received.
This study was funded by the American College of Cardiology’s National Cardiovascular Data Registry. Dr. Vemulapalli reported serving as a consultant to Novella and Premiere and receiving research grants from the Agency for Healthcare Research and Quality, Boston Scientific, Abbott Vascular, and the ACC.
AT ACC 16
Key clinical point: The incidence of valve hemodynamic deterioration after transaortic valve replacement is 2.5% from day 30 through 12 months post procedure.
Major finding: Patients who experienced valve hemodynamic deterioration had a rate of adverse clinical outcomes similar to those without valve deterioration.
Data source: This was a retrospective study of 18-month outcomes in the Society of Thoracic Surgeons/American College of Cardiology Transcatheter Valve Therapy Registry, which covers all commercial transcatheter valve replacements done in the United States.
Disclosures: This study was funded by the American College of Cardiology’s National Cardiovascular Data Registry. The presenter reported serving as a consultant to Novella and Premiere and receiving research grants from the Agency for Healthcare Research and Quality, Boston Scientific, Abbott Vascular, and the ACC.
Tiotropium inhalation spray effective for asthma regardless of allergic status
LOS ANGELES – Once-daily tiotropium bromide inhalation spray as long-term, add-on maintenance therapy in patients with poorly controlled symptomatic asthma is similarly effective in both allergic and nonallergic asthma, according to Dr. Donald P. Tashkin.
In a series of analyses presented at the annual meeting of the American Academy of Allergy, Asthma, and Immunology, he and his coinvestigators showed that tiotropium bromide inhalation spray (Spiriva Respimat) given as add-on maintenance therapy resulted in significantly improved lung function, enhanced asthma symptom control, and fewer asthma exacerbations. The important new finding in these post hoc analyses was that the medication was similarly effective across the full range of baseline serum IgE and blood eosinophil levels, said Dr. Tashkin, director of the pulmonary function laboratories at the University of California, Los Angeles.
Last fall, the Food and Drug Administration approved an expanded indication for tiotropium bromide inhalation spray as long-term, add-on maintenance therapy of asthma in patients aged 12 and up who remain symptomatic despite taking other maintenance therapies. The once-daily medication had already been approved in the fall of 2014 for maintenance therapy of chronic obstructive pulmonary disease. Spiriva Respimat delivers a once-daily 2.5-mcg dose of tiotropium bromide via two 1.25-mcg puffs in a slow-moving, propellant-free mist designed to get the drug into the distal lungs independent of a patient’s skill in using a conventional metered-dose inhaler.
Dr. Tashkin and his coinvestigators presented a series of post hoc analyses combining data on 3,012 participants in the two prospective PrimoTinA-asthma and two MezzoTinA-asthma clinical trials. These phase III, double-blind, placebo-controlled trials defined participants’ allergic phenotype on the basis of the conventional cut points of a serum IgE level above or below 430 mcg/mL or a blood eosinophil count above or below 600 cells/mcL.
“The question is, are these appropriate cut points? Can we be sure that somebody below those cut points doesn’t have atopy? To answer that question, we looked at the whole spectrum of eosinophils in the blood from 5/mcL to 2,000/mcL and serum IgE levels from 2 mcg/mL to 2,000 mcg/mL. We found that the efficacy was similar across the entire spectrum of these measures of allergy,” he said in an interview.
Thus, these new findings support the use of this novel maintenance therapy without any need for lab tests to determine whether an individual patient’s asthma is T helper 2–cell dominant or not, Dr. Tashkin added.
The PrimoTinA-asthma trials were 48-week studies of add-on Spiriva Respimat or placebo conducted in patients with symptomatic asthma despite treatment with an inhaled corticosteroid plus a long-acting beta-agonist. The MezzoTinA-asthma studies were 24 weeks long and focused on patients who remained symptomatic despite at least moderate-dose inhaled corticosteroid therapy. Key endpoints included improvement in asthma symptom control as measured by the seven-question Asthma Control Questionnaire, improved peak and trough forced expiratory volume in 1 second, and time to a first severe asthma exacerbation.
Dr. Tashkin reported serving as a paid speaker on behalf of Boehringer Ingelheim, which markets Spiriva Respimat and sponsored the studies.
LOS ANGELES – Once-daily tiotropium bromide inhalation spray as long-term, add-on maintenance therapy in patients with poorly controlled symptomatic asthma is similarly effective in both allergic and nonallergic asthma, according to Dr. Donald P. Tashkin.
In a series of analyses presented at the annual meeting of the American Academy of Allergy, Asthma, and Immunology, he and his coinvestigators showed that tiotropium bromide inhalation spray (Spiriva Respimat) given as add-on maintenance therapy resulted in significantly improved lung function, enhanced asthma symptom control, and fewer asthma exacerbations. The important new finding in these post hoc analyses was that the medication was similarly effective across the full range of baseline serum IgE and blood eosinophil levels, said Dr. Tashkin, director of the pulmonary function laboratories at the University of California, Los Angeles.
Last fall, the Food and Drug Administration approved an expanded indication for tiotropium bromide inhalation spray as long-term, add-on maintenance therapy of asthma in patients aged 12 and up who remain symptomatic despite taking other maintenance therapies. The once-daily medication had already been approved in the fall of 2014 for maintenance therapy of chronic obstructive pulmonary disease. Spiriva Respimat delivers a once-daily 2.5-mcg dose of tiotropium bromide via two 1.25-mcg puffs in a slow-moving, propellant-free mist designed to get the drug into the distal lungs independent of a patient’s skill in using a conventional metered-dose inhaler.
Dr. Tashkin and his coinvestigators presented a series of post hoc analyses combining data on 3,012 participants in the two prospective PrimoTinA-asthma and two MezzoTinA-asthma clinical trials. These phase III, double-blind, placebo-controlled trials defined participants’ allergic phenotype on the basis of the conventional cut points of a serum IgE level above or below 430 mcg/mL or a blood eosinophil count above or below 600 cells/mcL.
“The question is, are these appropriate cut points? Can we be sure that somebody below those cut points doesn’t have atopy? To answer that question, we looked at the whole spectrum of eosinophils in the blood from 5/mcL to 2,000/mcL and serum IgE levels from 2 mcg/mL to 2,000 mcg/mL. We found that the efficacy was similar across the entire spectrum of these measures of allergy,” he said in an interview.
Thus, these new findings support the use of this novel maintenance therapy without any need for lab tests to determine whether an individual patient’s asthma is T helper 2–cell dominant or not, Dr. Tashkin added.
The PrimoTinA-asthma trials were 48-week studies of add-on Spiriva Respimat or placebo conducted in patients with symptomatic asthma despite treatment with an inhaled corticosteroid plus a long-acting beta-agonist. The MezzoTinA-asthma studies were 24 weeks long and focused on patients who remained symptomatic despite at least moderate-dose inhaled corticosteroid therapy. Key endpoints included improvement in asthma symptom control as measured by the seven-question Asthma Control Questionnaire, improved peak and trough forced expiratory volume in 1 second, and time to a first severe asthma exacerbation.
Dr. Tashkin reported serving as a paid speaker on behalf of Boehringer Ingelheim, which markets Spiriva Respimat and sponsored the studies.
LOS ANGELES – Once-daily tiotropium bromide inhalation spray as long-term, add-on maintenance therapy in patients with poorly controlled symptomatic asthma is similarly effective in both allergic and nonallergic asthma, according to Dr. Donald P. Tashkin.
In a series of analyses presented at the annual meeting of the American Academy of Allergy, Asthma, and Immunology, he and his coinvestigators showed that tiotropium bromide inhalation spray (Spiriva Respimat) given as add-on maintenance therapy resulted in significantly improved lung function, enhanced asthma symptom control, and fewer asthma exacerbations. The important new finding in these post hoc analyses was that the medication was similarly effective across the full range of baseline serum IgE and blood eosinophil levels, said Dr. Tashkin, director of the pulmonary function laboratories at the University of California, Los Angeles.
Last fall, the Food and Drug Administration approved an expanded indication for tiotropium bromide inhalation spray as long-term, add-on maintenance therapy of asthma in patients aged 12 and up who remain symptomatic despite taking other maintenance therapies. The once-daily medication had already been approved in the fall of 2014 for maintenance therapy of chronic obstructive pulmonary disease. Spiriva Respimat delivers a once-daily 2.5-mcg dose of tiotropium bromide via two 1.25-mcg puffs in a slow-moving, propellant-free mist designed to get the drug into the distal lungs independent of a patient’s skill in using a conventional metered-dose inhaler.
Dr. Tashkin and his coinvestigators presented a series of post hoc analyses combining data on 3,012 participants in the two prospective PrimoTinA-asthma and two MezzoTinA-asthma clinical trials. These phase III, double-blind, placebo-controlled trials defined participants’ allergic phenotype on the basis of the conventional cut points of a serum IgE level above or below 430 mcg/mL or a blood eosinophil count above or below 600 cells/mcL.
“The question is, are these appropriate cut points? Can we be sure that somebody below those cut points doesn’t have atopy? To answer that question, we looked at the whole spectrum of eosinophils in the blood from 5/mcL to 2,000/mcL and serum IgE levels from 2 mcg/mL to 2,000 mcg/mL. We found that the efficacy was similar across the entire spectrum of these measures of allergy,” he said in an interview.
Thus, these new findings support the use of this novel maintenance therapy without any need for lab tests to determine whether an individual patient’s asthma is T helper 2–cell dominant or not, Dr. Tashkin added.
The PrimoTinA-asthma trials were 48-week studies of add-on Spiriva Respimat or placebo conducted in patients with symptomatic asthma despite treatment with an inhaled corticosteroid plus a long-acting beta-agonist. The MezzoTinA-asthma studies were 24 weeks long and focused on patients who remained symptomatic despite at least moderate-dose inhaled corticosteroid therapy. Key endpoints included improvement in asthma symptom control as measured by the seven-question Asthma Control Questionnaire, improved peak and trough forced expiratory volume in 1 second, and time to a first severe asthma exacerbation.
Dr. Tashkin reported serving as a paid speaker on behalf of Boehringer Ingelheim, which markets Spiriva Respimat and sponsored the studies.
EXPERT ANALYSIS FROM THE 2016 AAAAI ANNUAL MEETING
A dermatologist’s skin tips for rheumatologists
MAUI, HAWAII – Before concluding that a patient’s erythematous facial butterfly rash is an open and shut case of acute cutaneous lupus erythematosus, consider this, Dr. George M. Martin advised: 10-15 million Americans have acne rosacea.
“The 1% of those acne rosacea patients with the most severe disease are very often walking around misdiagnosed as having the butterfly rash of lupus,” Dr. Martin said at the 2016 Rheumatology Winter Clinical Symposium.
Indeed, rosacea – particularly the erythrotelangiectatic subtype – is the skin disease most often confused with acute cutaneous lupus erythematosus (ACLE), noted Dr. Martin, symposium codirector and a dermatologist practicing on Maui.
That’s a particularly common mistake in the event an individual with severe acne rosacea also happens to have the musculoskeletal symptoms of fibromyalgia, another very common disorder.
One clue useful in separating erythrotelangiectatic rosacea from ACLE is that “usually patients with the butterfly rash of acute lupus are sick. Rosacea patients are out there playing tennis and having a good time with a little bit of flush,” he said.
Other causes of a butterfly rash that get mistaken for ACLE are sebhorrheic dermatitis, contact dermatitis, and a photosensitizing drug eruption.
A biopsy may be necessary to establish a diagnosis, especially if the possibility of fibromyalgia is muddying the waters, according to Dr. Martin.
But cutaneous clues can also be helpful in deciding whether a butterfly rash is due to ACLE or cutaneous dermatomyositis. In lupus the rash typically spares the nasolabial folds, while the facial rash of dermatomyositis involves them. On the hands, the rash of ACLE spares the knuckles, whereas one-third of patients with dermatomyositis have the scaly red lesions known as Gottron’s papules over their knuckles. Cutaneous dermatomyositis has a predilection for the scalp, knees, and elbows.
A key distinction is that patients will report that the skin lesions of dermatomyositis are very itchy or have a burning sensation; that’s uncommon in cutaneous LE.
Also, prominent nailfold microhemorrhages at the distal cuticle are a telltale clue of dermatomyositis or possibly systemic sclerosis but aren’t part of the clinical picture in cutaneous LE. These microvascular changes are actually hemosiderin-containing deposits.
A skin biopsy is unhelpful in distinguishing cutaneous LE from cutaneous dermatomyositis. They basically have the same histopathologic findings, an interface dermatitis. Moreover, direct immunofluorescence and antinuclear antibody testing can be positive in both.
Dermatomyositis is more difficult to treat successfully than is cutaneous lupus. Antimalarials are often ineffective. But dapsone at 100-200 mg/day often works well in dermatomyositis, and it’s a relatively inexpensive drug, Dr. Martin said.
The hallmarks of subacute cutaneous lupus erythematosus (SCLE) are photosensitivity and small erythematous papules that expand into scaly patches and plaques that resemble psoriasis. SCLE is most common in women up to about age 40. It’s a disorder that predominates from the neck down. If more inflammation is present on the patient’s face than on the neck, shoulders, and upper torso, it’s not SCLE.
SCLE can be either drug induced or idiopathic; the two forms don’t differ clinically or histopathologically. In a patient older than age 50, SCLE is most commonly a drug-related eruption. In a 2011 review, Dr. Richard D. Sontheimer and his coinvestigators reported that antihypertensive agents accounted for one-third of cases of drug-induced SCLE, antifungals for one-quarter, chemotherapeutic agents for less than 9%, and antihistamines for 8% (Br J Dermatol. 2011 Mar;164[3]:465-72).
Dr. Sontheimer, professor of dermatology at the University of Utah in Salt Lake City, recently told Dr. Martin that his soon-to-be-published follow-up study of new cases since 2011 shows big changes in the culprit drugs. The proportion associated with chemotherapeutic agents, proton pump inhibitors, or biologics has jumped sharply. And with proton pump inhibitors now available over the counter, that class of medication is likely to continue to grow in importance as a source of drug-induced SCLE.
Dr. Martin reported serving on scientific advisory boards for and/or as a consultant to nine pharmaceutical companies.
MAUI, HAWAII – Before concluding that a patient’s erythematous facial butterfly rash is an open and shut case of acute cutaneous lupus erythematosus, consider this, Dr. George M. Martin advised: 10-15 million Americans have acne rosacea.
“The 1% of those acne rosacea patients with the most severe disease are very often walking around misdiagnosed as having the butterfly rash of lupus,” Dr. Martin said at the 2016 Rheumatology Winter Clinical Symposium.
Indeed, rosacea – particularly the erythrotelangiectatic subtype – is the skin disease most often confused with acute cutaneous lupus erythematosus (ACLE), noted Dr. Martin, symposium codirector and a dermatologist practicing on Maui.
That’s a particularly common mistake in the event an individual with severe acne rosacea also happens to have the musculoskeletal symptoms of fibromyalgia, another very common disorder.
One clue useful in separating erythrotelangiectatic rosacea from ACLE is that “usually patients with the butterfly rash of acute lupus are sick. Rosacea patients are out there playing tennis and having a good time with a little bit of flush,” he said.
Other causes of a butterfly rash that get mistaken for ACLE are sebhorrheic dermatitis, contact dermatitis, and a photosensitizing drug eruption.
A biopsy may be necessary to establish a diagnosis, especially if the possibility of fibromyalgia is muddying the waters, according to Dr. Martin.
But cutaneous clues can also be helpful in deciding whether a butterfly rash is due to ACLE or cutaneous dermatomyositis. In lupus the rash typically spares the nasolabial folds, while the facial rash of dermatomyositis involves them. On the hands, the rash of ACLE spares the knuckles, whereas one-third of patients with dermatomyositis have the scaly red lesions known as Gottron’s papules over their knuckles. Cutaneous dermatomyositis has a predilection for the scalp, knees, and elbows.
A key distinction is that patients will report that the skin lesions of dermatomyositis are very itchy or have a burning sensation; that’s uncommon in cutaneous LE.
Also, prominent nailfold microhemorrhages at the distal cuticle are a telltale clue of dermatomyositis or possibly systemic sclerosis but aren’t part of the clinical picture in cutaneous LE. These microvascular changes are actually hemosiderin-containing deposits.
A skin biopsy is unhelpful in distinguishing cutaneous LE from cutaneous dermatomyositis. They basically have the same histopathologic findings, an interface dermatitis. Moreover, direct immunofluorescence and antinuclear antibody testing can be positive in both.
Dermatomyositis is more difficult to treat successfully than is cutaneous lupus. Antimalarials are often ineffective. But dapsone at 100-200 mg/day often works well in dermatomyositis, and it’s a relatively inexpensive drug, Dr. Martin said.
The hallmarks of subacute cutaneous lupus erythematosus (SCLE) are photosensitivity and small erythematous papules that expand into scaly patches and plaques that resemble psoriasis. SCLE is most common in women up to about age 40. It’s a disorder that predominates from the neck down. If more inflammation is present on the patient’s face than on the neck, shoulders, and upper torso, it’s not SCLE.
SCLE can be either drug induced or idiopathic; the two forms don’t differ clinically or histopathologically. In a patient older than age 50, SCLE is most commonly a drug-related eruption. In a 2011 review, Dr. Richard D. Sontheimer and his coinvestigators reported that antihypertensive agents accounted for one-third of cases of drug-induced SCLE, antifungals for one-quarter, chemotherapeutic agents for less than 9%, and antihistamines for 8% (Br J Dermatol. 2011 Mar;164[3]:465-72).
Dr. Sontheimer, professor of dermatology at the University of Utah in Salt Lake City, recently told Dr. Martin that his soon-to-be-published follow-up study of new cases since 2011 shows big changes in the culprit drugs. The proportion associated with chemotherapeutic agents, proton pump inhibitors, or biologics has jumped sharply. And with proton pump inhibitors now available over the counter, that class of medication is likely to continue to grow in importance as a source of drug-induced SCLE.
Dr. Martin reported serving on scientific advisory boards for and/or as a consultant to nine pharmaceutical companies.
MAUI, HAWAII – Before concluding that a patient’s erythematous facial butterfly rash is an open and shut case of acute cutaneous lupus erythematosus, consider this, Dr. George M. Martin advised: 10-15 million Americans have acne rosacea.
“The 1% of those acne rosacea patients with the most severe disease are very often walking around misdiagnosed as having the butterfly rash of lupus,” Dr. Martin said at the 2016 Rheumatology Winter Clinical Symposium.
Indeed, rosacea – particularly the erythrotelangiectatic subtype – is the skin disease most often confused with acute cutaneous lupus erythematosus (ACLE), noted Dr. Martin, symposium codirector and a dermatologist practicing on Maui.
That’s a particularly common mistake in the event an individual with severe acne rosacea also happens to have the musculoskeletal symptoms of fibromyalgia, another very common disorder.
One clue useful in separating erythrotelangiectatic rosacea from ACLE is that “usually patients with the butterfly rash of acute lupus are sick. Rosacea patients are out there playing tennis and having a good time with a little bit of flush,” he said.
Other causes of a butterfly rash that get mistaken for ACLE are sebhorrheic dermatitis, contact dermatitis, and a photosensitizing drug eruption.
A biopsy may be necessary to establish a diagnosis, especially if the possibility of fibromyalgia is muddying the waters, according to Dr. Martin.
But cutaneous clues can also be helpful in deciding whether a butterfly rash is due to ACLE or cutaneous dermatomyositis. In lupus the rash typically spares the nasolabial folds, while the facial rash of dermatomyositis involves them. On the hands, the rash of ACLE spares the knuckles, whereas one-third of patients with dermatomyositis have the scaly red lesions known as Gottron’s papules over their knuckles. Cutaneous dermatomyositis has a predilection for the scalp, knees, and elbows.
A key distinction is that patients will report that the skin lesions of dermatomyositis are very itchy or have a burning sensation; that’s uncommon in cutaneous LE.
Also, prominent nailfold microhemorrhages at the distal cuticle are a telltale clue of dermatomyositis or possibly systemic sclerosis but aren’t part of the clinical picture in cutaneous LE. These microvascular changes are actually hemosiderin-containing deposits.
A skin biopsy is unhelpful in distinguishing cutaneous LE from cutaneous dermatomyositis. They basically have the same histopathologic findings, an interface dermatitis. Moreover, direct immunofluorescence and antinuclear antibody testing can be positive in both.
Dermatomyositis is more difficult to treat successfully than is cutaneous lupus. Antimalarials are often ineffective. But dapsone at 100-200 mg/day often works well in dermatomyositis, and it’s a relatively inexpensive drug, Dr. Martin said.
The hallmarks of subacute cutaneous lupus erythematosus (SCLE) are photosensitivity and small erythematous papules that expand into scaly patches and plaques that resemble psoriasis. SCLE is most common in women up to about age 40. It’s a disorder that predominates from the neck down. If more inflammation is present on the patient’s face than on the neck, shoulders, and upper torso, it’s not SCLE.
SCLE can be either drug induced or idiopathic; the two forms don’t differ clinically or histopathologically. In a patient older than age 50, SCLE is most commonly a drug-related eruption. In a 2011 review, Dr. Richard D. Sontheimer and his coinvestigators reported that antihypertensive agents accounted for one-third of cases of drug-induced SCLE, antifungals for one-quarter, chemotherapeutic agents for less than 9%, and antihistamines for 8% (Br J Dermatol. 2011 Mar;164[3]:465-72).
Dr. Sontheimer, professor of dermatology at the University of Utah in Salt Lake City, recently told Dr. Martin that his soon-to-be-published follow-up study of new cases since 2011 shows big changes in the culprit drugs. The proportion associated with chemotherapeutic agents, proton pump inhibitors, or biologics has jumped sharply. And with proton pump inhibitors now available over the counter, that class of medication is likely to continue to grow in importance as a source of drug-induced SCLE.
Dr. Martin reported serving on scientific advisory boards for and/or as a consultant to nine pharmaceutical companies.
EXPERT ANALYSIS FROM RWCS 2016
Kawasaki disease and infections aren’t mutually exclusive
CHICAGO – Kawasaki disease and concurrent bacterial or viral infection are by no means mutually exclusive, Dr. Cathie-Kim Le cautioned at the annual meeting of the American College of Cardiology.
“Recognizing that both can coexist will ensure timely IVIg [intravenous immunoglobulin] treatment and appropriate containment of coronary artery complications,” said Dr. Le of Sainte-Justine University Hospital in Montreal.
She presented a retrospective study of 128 patients, mean age 3.4 years, admitted to the pediatric academic tertiary care center with a discharge diagnosis of Kawasaki disease. During their hospitalization all of them underwent a work-up for bacterial and viral infectious diseases, which proved positive in 33% of cases. Roughly 40% of subjects had incomplete Kawasaki disease, meaning they lacked a sufficient number of signs of mucocutaneous inflammation to fulfill the epidemiologic case definition; however, their prevalence of concomitant infection was similar to that of the group with classic Kawasaki disease.
Among the most common types of infections in patients with Kawasaki disease were otitis media, which accounted for 17% of the infections; upper respiratory infections, 21%; and group A streptococcal pharyngitis and pneumonia, which accounted for 14% each.
There were no differences in clinical presentation or laboratory values between children with or without concomitant infection. Nor was myocardial profiling useful in differentiating patients with concomitant infection from those without: Ventricular shortening fraction scores and N-terminal probrain natriuretic peptide levels were similar in the two groups of Kawasaki disease patients.
Acute coronary dilatation occurred in 26% of Kawasaki disease patients with concomitant infection and similarly in 30% of those without infection. Coronary aneurysm, the most serious complication of Kawasaki disease, occurred in 14% of patients with infection and an identical proportion of the uninfected.
Almost all patients received IVIg. Resistance to the effects of IVIg occurred in 36% of patients with concomitant infection, a rate twice that seen in the group without infection. Coronary aneurysms and dilatations were significantly more common in IVIg-resistant patients, regardless of their infection status.
Dr. Le reported having no relevant financial conflicts.
CHICAGO – Kawasaki disease and concurrent bacterial or viral infection are by no means mutually exclusive, Dr. Cathie-Kim Le cautioned at the annual meeting of the American College of Cardiology.
“Recognizing that both can coexist will ensure timely IVIg [intravenous immunoglobulin] treatment and appropriate containment of coronary artery complications,” said Dr. Le of Sainte-Justine University Hospital in Montreal.
She presented a retrospective study of 128 patients, mean age 3.4 years, admitted to the pediatric academic tertiary care center with a discharge diagnosis of Kawasaki disease. During their hospitalization all of them underwent a work-up for bacterial and viral infectious diseases, which proved positive in 33% of cases. Roughly 40% of subjects had incomplete Kawasaki disease, meaning they lacked a sufficient number of signs of mucocutaneous inflammation to fulfill the epidemiologic case definition; however, their prevalence of concomitant infection was similar to that of the group with classic Kawasaki disease.
Among the most common types of infections in patients with Kawasaki disease were otitis media, which accounted for 17% of the infections; upper respiratory infections, 21%; and group A streptococcal pharyngitis and pneumonia, which accounted for 14% each.
There were no differences in clinical presentation or laboratory values between children with or without concomitant infection. Nor was myocardial profiling useful in differentiating patients with concomitant infection from those without: Ventricular shortening fraction scores and N-terminal probrain natriuretic peptide levels were similar in the two groups of Kawasaki disease patients.
Acute coronary dilatation occurred in 26% of Kawasaki disease patients with concomitant infection and similarly in 30% of those without infection. Coronary aneurysm, the most serious complication of Kawasaki disease, occurred in 14% of patients with infection and an identical proportion of the uninfected.
Almost all patients received IVIg. Resistance to the effects of IVIg occurred in 36% of patients with concomitant infection, a rate twice that seen in the group without infection. Coronary aneurysms and dilatations were significantly more common in IVIg-resistant patients, regardless of their infection status.
Dr. Le reported having no relevant financial conflicts.
CHICAGO – Kawasaki disease and concurrent bacterial or viral infection are by no means mutually exclusive, Dr. Cathie-Kim Le cautioned at the annual meeting of the American College of Cardiology.
“Recognizing that both can coexist will ensure timely IVIg [intravenous immunoglobulin] treatment and appropriate containment of coronary artery complications,” said Dr. Le of Sainte-Justine University Hospital in Montreal.
She presented a retrospective study of 128 patients, mean age 3.4 years, admitted to the pediatric academic tertiary care center with a discharge diagnosis of Kawasaki disease. During their hospitalization all of them underwent a work-up for bacterial and viral infectious diseases, which proved positive in 33% of cases. Roughly 40% of subjects had incomplete Kawasaki disease, meaning they lacked a sufficient number of signs of mucocutaneous inflammation to fulfill the epidemiologic case definition; however, their prevalence of concomitant infection was similar to that of the group with classic Kawasaki disease.
Among the most common types of infections in patients with Kawasaki disease were otitis media, which accounted for 17% of the infections; upper respiratory infections, 21%; and group A streptococcal pharyngitis and pneumonia, which accounted for 14% each.
There were no differences in clinical presentation or laboratory values between children with or without concomitant infection. Nor was myocardial profiling useful in differentiating patients with concomitant infection from those without: Ventricular shortening fraction scores and N-terminal probrain natriuretic peptide levels were similar in the two groups of Kawasaki disease patients.
Acute coronary dilatation occurred in 26% of Kawasaki disease patients with concomitant infection and similarly in 30% of those without infection. Coronary aneurysm, the most serious complication of Kawasaki disease, occurred in 14% of patients with infection and an identical proportion of the uninfected.
Almost all patients received IVIg. Resistance to the effects of IVIg occurred in 36% of patients with concomitant infection, a rate twice that seen in the group without infection. Coronary aneurysms and dilatations were significantly more common in IVIg-resistant patients, regardless of their infection status.
Dr. Le reported having no relevant financial conflicts.
AT ACC 16
Key clinical point: One-third of Kawasaki patients have a concomitant common infection that may delay timely diagnosis.
Major finding: Coronary aneurysm, the most serious complication of Kawasaki disease, occurred in 14% of patients with an infection and in 14% of the uninfected.
Data source: A retrospective study of 128 children with Kawasaki disease.
Disclosures: The study presenter reported having no relevant financial conflicts.