User login
AMA House Takes on Obesity
CHICAGO – Is obesity a chronic disease on par with diabetes and rheumatoid arthritis that warrants separate coding and reimbursement? That was one question the American Medical Association House of Delegates grappled with at its annual meeting.
The AMA’s policy-making body approached obesity from several different angles, from physical education to soda taxes to physician counseling.
The Illinois delegation put forward a resolution urging the AMA to "recognize obesity and overweight as a chronic medical condition (de facto disease state) and urgent public health problem." As such, the resolution said, the AMA should also recommend that insurers and government agencies pay appropriately for obesity intervention services and develop an ICD code for managing and treating obese and overweight patients.
In committee debates, most delegates agreed that physicians should be paid for managing obesity, but there was a difference of opinion as to whether it should be classified as a disease. Current AMA policy labels obesity a major health problem; the group previously has called for better coding and payment for obesity-related treatment.
When the measure came to the floor for a vote, the delegates voted to study further whether obesity should be classified as a disease.
The House also agreed to a report written by the AMA Council on Science and Public Health that found that taxes on sugar-sweetened beverages are a potentially effective way to reduce consumption. This would be a new policy for the AMA. But the report found that the beverages are a contributing factor to the obesity epidemic. The report also said that the tax revenue should be used to fund programs to prevent obesity.
The new policy was not without its detractors. Dr. Daniel Koretz, an alternate from New York, wanted to amend the report so that the word tax was removed.
"I would hate to see the headline that the AMA is in favor of increased taxes," he said. He also said he felt that taxes should not be used as a method to induce people to act in their best interests. "This is a question of individuals’ freedom to make their own choices about health free of government coercion," he said.
Dr. Russell Kridel, a member of the Council on Science and Public Health, noted that the report merely explored whether a tax would reduce consumption.
"Our council looked at the science. We don’t advocate yes or no on taxes," said Dr. Kridel, a Houston plastic surgeon.
In the end, the House agreed to the Council report as written.
The House also delved into whether enough was being done in the schools to combat obesity. It approved a resolution that would back legislation or new policies to have "meaningful yearly instruction in nutrition, including instruction in the causes, consequences, and prevention of obesity, in grades 1 through 12 in public schools."
The delegates also considered a measure urging the AMA to create a checklist or guidelines for patients who wanted to resume physical activity after an illness or surgery. Delegates agreed that physical activity was important for quality of life and to combat obesity, but some were concerned that physicians might face liability – or be challenged outside their scope of knowledge – when signing off on a return to physical activity.
The House referred the idea for further study.
CHICAGO – Is obesity a chronic disease on par with diabetes and rheumatoid arthritis that warrants separate coding and reimbursement? That was one question the American Medical Association House of Delegates grappled with at its annual meeting.
The AMA’s policy-making body approached obesity from several different angles, from physical education to soda taxes to physician counseling.
The Illinois delegation put forward a resolution urging the AMA to "recognize obesity and overweight as a chronic medical condition (de facto disease state) and urgent public health problem." As such, the resolution said, the AMA should also recommend that insurers and government agencies pay appropriately for obesity intervention services and develop an ICD code for managing and treating obese and overweight patients.
In committee debates, most delegates agreed that physicians should be paid for managing obesity, but there was a difference of opinion as to whether it should be classified as a disease. Current AMA policy labels obesity a major health problem; the group previously has called for better coding and payment for obesity-related treatment.
When the measure came to the floor for a vote, the delegates voted to study further whether obesity should be classified as a disease.
The House also agreed to a report written by the AMA Council on Science and Public Health that found that taxes on sugar-sweetened beverages are a potentially effective way to reduce consumption. This would be a new policy for the AMA. But the report found that the beverages are a contributing factor to the obesity epidemic. The report also said that the tax revenue should be used to fund programs to prevent obesity.
The new policy was not without its detractors. Dr. Daniel Koretz, an alternate from New York, wanted to amend the report so that the word tax was removed.
"I would hate to see the headline that the AMA is in favor of increased taxes," he said. He also said he felt that taxes should not be used as a method to induce people to act in their best interests. "This is a question of individuals’ freedom to make their own choices about health free of government coercion," he said.
Dr. Russell Kridel, a member of the Council on Science and Public Health, noted that the report merely explored whether a tax would reduce consumption.
"Our council looked at the science. We don’t advocate yes or no on taxes," said Dr. Kridel, a Houston plastic surgeon.
In the end, the House agreed to the Council report as written.
The House also delved into whether enough was being done in the schools to combat obesity. It approved a resolution that would back legislation or new policies to have "meaningful yearly instruction in nutrition, including instruction in the causes, consequences, and prevention of obesity, in grades 1 through 12 in public schools."
The delegates also considered a measure urging the AMA to create a checklist or guidelines for patients who wanted to resume physical activity after an illness or surgery. Delegates agreed that physical activity was important for quality of life and to combat obesity, but some were concerned that physicians might face liability – or be challenged outside their scope of knowledge – when signing off on a return to physical activity.
The House referred the idea for further study.
CHICAGO – Is obesity a chronic disease on par with diabetes and rheumatoid arthritis that warrants separate coding and reimbursement? That was one question the American Medical Association House of Delegates grappled with at its annual meeting.
The AMA’s policy-making body approached obesity from several different angles, from physical education to soda taxes to physician counseling.
The Illinois delegation put forward a resolution urging the AMA to "recognize obesity and overweight as a chronic medical condition (de facto disease state) and urgent public health problem." As such, the resolution said, the AMA should also recommend that insurers and government agencies pay appropriately for obesity intervention services and develop an ICD code for managing and treating obese and overweight patients.
In committee debates, most delegates agreed that physicians should be paid for managing obesity, but there was a difference of opinion as to whether it should be classified as a disease. Current AMA policy labels obesity a major health problem; the group previously has called for better coding and payment for obesity-related treatment.
When the measure came to the floor for a vote, the delegates voted to study further whether obesity should be classified as a disease.
The House also agreed to a report written by the AMA Council on Science and Public Health that found that taxes on sugar-sweetened beverages are a potentially effective way to reduce consumption. This would be a new policy for the AMA. But the report found that the beverages are a contributing factor to the obesity epidemic. The report also said that the tax revenue should be used to fund programs to prevent obesity.
The new policy was not without its detractors. Dr. Daniel Koretz, an alternate from New York, wanted to amend the report so that the word tax was removed.
"I would hate to see the headline that the AMA is in favor of increased taxes," he said. He also said he felt that taxes should not be used as a method to induce people to act in their best interests. "This is a question of individuals’ freedom to make their own choices about health free of government coercion," he said.
Dr. Russell Kridel, a member of the Council on Science and Public Health, noted that the report merely explored whether a tax would reduce consumption.
"Our council looked at the science. We don’t advocate yes or no on taxes," said Dr. Kridel, a Houston plastic surgeon.
In the end, the House agreed to the Council report as written.
The House also delved into whether enough was being done in the schools to combat obesity. It approved a resolution that would back legislation or new policies to have "meaningful yearly instruction in nutrition, including instruction in the causes, consequences, and prevention of obesity, in grades 1 through 12 in public schools."
The delegates also considered a measure urging the AMA to create a checklist or guidelines for patients who wanted to resume physical activity after an illness or surgery. Delegates agreed that physical activity was important for quality of life and to combat obesity, but some were concerned that physicians might face liability – or be challenged outside their scope of knowledge – when signing off on a return to physical activity.
The House referred the idea for further study.
AT THE AMERICAN MEDICAL ASSOCIATION ANNUAL HOUSE OF DELEGATES
AMA Steps Back From Premium Support Plan
CHICAGO – It may have been this year’s tempest in a teapot: After some premeeting scuffles and a marginally exercised debate in committee, the American Medical Association’s House of Delegates calmly voted to direct the organization to explore Medicare financing options, including a defined-contribution program.
The vote was a step back from the wholehearted endorsement of a so-called premium support plan that had circulated before the meeting.
The AMA’s Council on Medical Service had prepared a report for delegates to debate, on options for propping up Medicare over the long term. The report came out in favor of building "on existing policies to support making Medicare a defined contribution program," and crafted a resolution urging the AMA to support "transitioning Medicare to a premium support program."
But a few days before the House got underway, the council withdrew its report and recommendation. Dr. Tom Sullivan, the council chairman, said in a statement that it was withdrawn because the council "believes there is a need to put in additional work on a revised report that addresses a number of complicated policy issues."
The Louisiana delegation was not pleased. It sought – and eventually won – the ability to put the council’s resolution back on the table. That resolution was hotly debated during a committee hearing on the second day of the House of Delegates meeting.
Many delegates argued in favor of supporting a defined-contribution plan for Medicare, saying that the idea already was incorporated into existing AMA policy.
But others worried about what taking a stand on Medicare financing could do to the AMA’s credibility and standing in an election year.
"It’s a potentially politically polarizing stance we’re asking the AMA to take," said Dr. Charles Rothberg, an ophthalmologist with the New York delegation. He also asked why the House was being asked to take such urgent action.
Dr. Nancy Nielsen, the immediate past-president of the AMA, also cautioned against moving too quickly. She urged delegates to take more time to think about Medicare financing options, noting that a wrong move could raise the ire of senior citizens.
When the issue came to the floor for a vote, delegates voted to approve a weakened substitute resolution that directed the AMA to explore all options, including premium support, and "to consider mechanisms to adjust contributions in order to ensure that health insurance coverage remains affordable for all beneficiaries."
The council was directed to report back to the AMA at its interim meeting, which will be held after the November election.
After the vote, Dr. Donald Palmisano, a former AMA president and member of the Louisiana delegation, said in an interview that "it confirms long-standing policy use of a defined-contribution model for the purchase of insurance." Dr. Palmisano added, "We need a system that protects seniors and the disabled and also keeps doctors in the practice of medicine."
Dr. William E. Golden, head of the American College of Physicians’ delegation, said in an interview that the ACP was pleased with the notion that the AMA would further study premium support. The ACP has urged against a quick transformation of Medicare from a defined benefit to a defined contribution.
But in a white paper on Medicare reform released in April the ACP backed a deeper investigation of premium support.
CHICAGO – It may have been this year’s tempest in a teapot: After some premeeting scuffles and a marginally exercised debate in committee, the American Medical Association’s House of Delegates calmly voted to direct the organization to explore Medicare financing options, including a defined-contribution program.
The vote was a step back from the wholehearted endorsement of a so-called premium support plan that had circulated before the meeting.
The AMA’s Council on Medical Service had prepared a report for delegates to debate, on options for propping up Medicare over the long term. The report came out in favor of building "on existing policies to support making Medicare a defined contribution program," and crafted a resolution urging the AMA to support "transitioning Medicare to a premium support program."
But a few days before the House got underway, the council withdrew its report and recommendation. Dr. Tom Sullivan, the council chairman, said in a statement that it was withdrawn because the council "believes there is a need to put in additional work on a revised report that addresses a number of complicated policy issues."
The Louisiana delegation was not pleased. It sought – and eventually won – the ability to put the council’s resolution back on the table. That resolution was hotly debated during a committee hearing on the second day of the House of Delegates meeting.
Many delegates argued in favor of supporting a defined-contribution plan for Medicare, saying that the idea already was incorporated into existing AMA policy.
But others worried about what taking a stand on Medicare financing could do to the AMA’s credibility and standing in an election year.
"It’s a potentially politically polarizing stance we’re asking the AMA to take," said Dr. Charles Rothberg, an ophthalmologist with the New York delegation. He also asked why the House was being asked to take such urgent action.
Dr. Nancy Nielsen, the immediate past-president of the AMA, also cautioned against moving too quickly. She urged delegates to take more time to think about Medicare financing options, noting that a wrong move could raise the ire of senior citizens.
When the issue came to the floor for a vote, delegates voted to approve a weakened substitute resolution that directed the AMA to explore all options, including premium support, and "to consider mechanisms to adjust contributions in order to ensure that health insurance coverage remains affordable for all beneficiaries."
The council was directed to report back to the AMA at its interim meeting, which will be held after the November election.
After the vote, Dr. Donald Palmisano, a former AMA president and member of the Louisiana delegation, said in an interview that "it confirms long-standing policy use of a defined-contribution model for the purchase of insurance." Dr. Palmisano added, "We need a system that protects seniors and the disabled and also keeps doctors in the practice of medicine."
Dr. William E. Golden, head of the American College of Physicians’ delegation, said in an interview that the ACP was pleased with the notion that the AMA would further study premium support. The ACP has urged against a quick transformation of Medicare from a defined benefit to a defined contribution.
But in a white paper on Medicare reform released in April the ACP backed a deeper investigation of premium support.
CHICAGO – It may have been this year’s tempest in a teapot: After some premeeting scuffles and a marginally exercised debate in committee, the American Medical Association’s House of Delegates calmly voted to direct the organization to explore Medicare financing options, including a defined-contribution program.
The vote was a step back from the wholehearted endorsement of a so-called premium support plan that had circulated before the meeting.
The AMA’s Council on Medical Service had prepared a report for delegates to debate, on options for propping up Medicare over the long term. The report came out in favor of building "on existing policies to support making Medicare a defined contribution program," and crafted a resolution urging the AMA to support "transitioning Medicare to a premium support program."
But a few days before the House got underway, the council withdrew its report and recommendation. Dr. Tom Sullivan, the council chairman, said in a statement that it was withdrawn because the council "believes there is a need to put in additional work on a revised report that addresses a number of complicated policy issues."
The Louisiana delegation was not pleased. It sought – and eventually won – the ability to put the council’s resolution back on the table. That resolution was hotly debated during a committee hearing on the second day of the House of Delegates meeting.
Many delegates argued in favor of supporting a defined-contribution plan for Medicare, saying that the idea already was incorporated into existing AMA policy.
But others worried about what taking a stand on Medicare financing could do to the AMA’s credibility and standing in an election year.
"It’s a potentially politically polarizing stance we’re asking the AMA to take," said Dr. Charles Rothberg, an ophthalmologist with the New York delegation. He also asked why the House was being asked to take such urgent action.
Dr. Nancy Nielsen, the immediate past-president of the AMA, also cautioned against moving too quickly. She urged delegates to take more time to think about Medicare financing options, noting that a wrong move could raise the ire of senior citizens.
When the issue came to the floor for a vote, delegates voted to approve a weakened substitute resolution that directed the AMA to explore all options, including premium support, and "to consider mechanisms to adjust contributions in order to ensure that health insurance coverage remains affordable for all beneficiaries."
The council was directed to report back to the AMA at its interim meeting, which will be held after the November election.
After the vote, Dr. Donald Palmisano, a former AMA president and member of the Louisiana delegation, said in an interview that "it confirms long-standing policy use of a defined-contribution model for the purchase of insurance." Dr. Palmisano added, "We need a system that protects seniors and the disabled and also keeps doctors in the practice of medicine."
Dr. William E. Golden, head of the American College of Physicians’ delegation, said in an interview that the ACP was pleased with the notion that the AMA would further study premium support. The ACP has urged against a quick transformation of Medicare from a defined benefit to a defined contribution.
But in a white paper on Medicare reform released in April the ACP backed a deeper investigation of premium support.
AT THE AMERICAN MEDICAL ASSOCIATION ANNUAL HOUSE OF DELEGATES MEETING
AMA Delegates Slam PSA, Mammography Screening Recs
CHICAGO – Delegates to the American Medical Association’s legislative body are not happy with the U.S. Preventive Services Task Force.
They voted overwhelmingly (322 to 93) to express "concern" over the task force’s recommendations on screening mammography and prostate-specific antigen screening. Both recommendations attracted a firestorm of criticism when they were issued – the mammography recommendation in 2009 and those on PSA testing in May.
Many delegates also said they thought that the USPSTF, a quasi-governmental group operating under the auspices of the Agency for Healthcare Research and Quality, had reached its decisions without proper input from specialty societies or experts in each field.
In a second resolution, the delegates voted to encourage the USPSTF to "implement procedures that allow for meaningful input on recommendation development from specialists and stakeholders in the topic area under study."
Dr. Arl Van Moore Jr., a delegate from the American College of Radiology, said that neither the ACR nor any prominent breast imaging or surgical societies were contacted by the USPSTF in creating the screening mammography recommendations.
"None of the recognized experts in the field were contacted, to the best of our knowledge," said Dr. Moore, an interventional radiologist in Charlotte, N.C.
The American Urological Association delegate, Dr. William Gee, did not mince words when it came to the PSA screening guidelines.
"The [USPSTF] did not use an open process and ignored the public in reaching their conclusions," he said.
But Dr. Sally J. Trippel, a preventive medicine specialist at the Mayo Clinic and delegate from Minnesota, defended the USPSTF and its process. The task force is "about as politically independent as any national organization can get," and "about as free of conflict of interest as is possible in any organization developing guidelines for American clinicians," she said.
She quoted from task force documents showing that urologists provided peer review of the PSA evidence review. "So there were experts from urology involved in the development of that guideline," and also for the one on screening mammography.
To further show its consternation with the task force, AMA delegates also approved a report that stated that starting at age 40, women should be "eligible for screening mammography," and encouraging physicians "to regularly discuss with their individual patients the benefits and risks of screening mammography, and whether screening is appropriate for each clinical situation given that the balance of benefits and risks will be viewed differently by each patient."
Primary care delegates from the American Academy of Family Physicians and the American College of Physicians opposed the resolutions of concern against the USPSTF and the mammography report.
The AAFP supported the task force when it issued its mammography recommendations "because it was the most comprehensive pattern and set of preventive guidelines using current methodology with what was available in science at that time," said Dr. Roland Goertz, AAFP delegate.
The task force basically recommends what the AMA report urged: that physicians have discussions with their patients about risks and benefits, he said. The problem is not the USPSTF recommendations, but that they are being used to deny payment, he added.
Dr. Richard Reiling, a delegate from the American College of Surgeons, said that the task force had confused patients with its mammography recommendation, and called for the AMA to convene all interested parties to craft a single guideline. The USPSTF was "wrong in presenting this report without listening to the stakeholders in the past," he said, adding, "let’s get one guideline out there."
Dr. Goertz agreed that there needed to be a common guideline.
ACP Delegate Dr. William Golden expressed the ACP’s view that the House of Delegates was not the appropriate venue for voting on particular guidelines. "The House should not be in the position of voting on what guideline is best," he said.
CHICAGO – Delegates to the American Medical Association’s legislative body are not happy with the U.S. Preventive Services Task Force.
They voted overwhelmingly (322 to 93) to express "concern" over the task force’s recommendations on screening mammography and prostate-specific antigen screening. Both recommendations attracted a firestorm of criticism when they were issued – the mammography recommendation in 2009 and those on PSA testing in May.
Many delegates also said they thought that the USPSTF, a quasi-governmental group operating under the auspices of the Agency for Healthcare Research and Quality, had reached its decisions without proper input from specialty societies or experts in each field.
In a second resolution, the delegates voted to encourage the USPSTF to "implement procedures that allow for meaningful input on recommendation development from specialists and stakeholders in the topic area under study."
Dr. Arl Van Moore Jr., a delegate from the American College of Radiology, said that neither the ACR nor any prominent breast imaging or surgical societies were contacted by the USPSTF in creating the screening mammography recommendations.
"None of the recognized experts in the field were contacted, to the best of our knowledge," said Dr. Moore, an interventional radiologist in Charlotte, N.C.
The American Urological Association delegate, Dr. William Gee, did not mince words when it came to the PSA screening guidelines.
"The [USPSTF] did not use an open process and ignored the public in reaching their conclusions," he said.
But Dr. Sally J. Trippel, a preventive medicine specialist at the Mayo Clinic and delegate from Minnesota, defended the USPSTF and its process. The task force is "about as politically independent as any national organization can get," and "about as free of conflict of interest as is possible in any organization developing guidelines for American clinicians," she said.
She quoted from task force documents showing that urologists provided peer review of the PSA evidence review. "So there were experts from urology involved in the development of that guideline," and also for the one on screening mammography.
To further show its consternation with the task force, AMA delegates also approved a report that stated that starting at age 40, women should be "eligible for screening mammography," and encouraging physicians "to regularly discuss with their individual patients the benefits and risks of screening mammography, and whether screening is appropriate for each clinical situation given that the balance of benefits and risks will be viewed differently by each patient."
Primary care delegates from the American Academy of Family Physicians and the American College of Physicians opposed the resolutions of concern against the USPSTF and the mammography report.
The AAFP supported the task force when it issued its mammography recommendations "because it was the most comprehensive pattern and set of preventive guidelines using current methodology with what was available in science at that time," said Dr. Roland Goertz, AAFP delegate.
The task force basically recommends what the AMA report urged: that physicians have discussions with their patients about risks and benefits, he said. The problem is not the USPSTF recommendations, but that they are being used to deny payment, he added.
Dr. Richard Reiling, a delegate from the American College of Surgeons, said that the task force had confused patients with its mammography recommendation, and called for the AMA to convene all interested parties to craft a single guideline. The USPSTF was "wrong in presenting this report without listening to the stakeholders in the past," he said, adding, "let’s get one guideline out there."
Dr. Goertz agreed that there needed to be a common guideline.
ACP Delegate Dr. William Golden expressed the ACP’s view that the House of Delegates was not the appropriate venue for voting on particular guidelines. "The House should not be in the position of voting on what guideline is best," he said.
CHICAGO – Delegates to the American Medical Association’s legislative body are not happy with the U.S. Preventive Services Task Force.
They voted overwhelmingly (322 to 93) to express "concern" over the task force’s recommendations on screening mammography and prostate-specific antigen screening. Both recommendations attracted a firestorm of criticism when they were issued – the mammography recommendation in 2009 and those on PSA testing in May.
Many delegates also said they thought that the USPSTF, a quasi-governmental group operating under the auspices of the Agency for Healthcare Research and Quality, had reached its decisions without proper input from specialty societies or experts in each field.
In a second resolution, the delegates voted to encourage the USPSTF to "implement procedures that allow for meaningful input on recommendation development from specialists and stakeholders in the topic area under study."
Dr. Arl Van Moore Jr., a delegate from the American College of Radiology, said that neither the ACR nor any prominent breast imaging or surgical societies were contacted by the USPSTF in creating the screening mammography recommendations.
"None of the recognized experts in the field were contacted, to the best of our knowledge," said Dr. Moore, an interventional radiologist in Charlotte, N.C.
The American Urological Association delegate, Dr. William Gee, did not mince words when it came to the PSA screening guidelines.
"The [USPSTF] did not use an open process and ignored the public in reaching their conclusions," he said.
But Dr. Sally J. Trippel, a preventive medicine specialist at the Mayo Clinic and delegate from Minnesota, defended the USPSTF and its process. The task force is "about as politically independent as any national organization can get," and "about as free of conflict of interest as is possible in any organization developing guidelines for American clinicians," she said.
She quoted from task force documents showing that urologists provided peer review of the PSA evidence review. "So there were experts from urology involved in the development of that guideline," and also for the one on screening mammography.
To further show its consternation with the task force, AMA delegates also approved a report that stated that starting at age 40, women should be "eligible for screening mammography," and encouraging physicians "to regularly discuss with their individual patients the benefits and risks of screening mammography, and whether screening is appropriate for each clinical situation given that the balance of benefits and risks will be viewed differently by each patient."
Primary care delegates from the American Academy of Family Physicians and the American College of Physicians opposed the resolutions of concern against the USPSTF and the mammography report.
The AAFP supported the task force when it issued its mammography recommendations "because it was the most comprehensive pattern and set of preventive guidelines using current methodology with what was available in science at that time," said Dr. Roland Goertz, AAFP delegate.
The task force basically recommends what the AMA report urged: that physicians have discussions with their patients about risks and benefits, he said. The problem is not the USPSTF recommendations, but that they are being used to deny payment, he added.
Dr. Richard Reiling, a delegate from the American College of Surgeons, said that the task force had confused patients with its mammography recommendation, and called for the AMA to convene all interested parties to craft a single guideline. The USPSTF was "wrong in presenting this report without listening to the stakeholders in the past," he said, adding, "let’s get one guideline out there."
Dr. Goertz agreed that there needed to be a common guideline.
ACP Delegate Dr. William Golden expressed the ACP’s view that the House of Delegates was not the appropriate venue for voting on particular guidelines. "The House should not be in the position of voting on what guideline is best," he said.
AT THE ANNUAL MEETING OF THE AMERICAN MEDICAL ASSOCIATION'S HOUSE OF DELEGATES
Insurers Paying Faster, But Prior Authorization on Rise
CHICAGO – Health insurers are paying claims more quickly and with greater accuracy, but they’re also erecting barriers by requiring more prior authorization, a new report from the American Medical Association has found.
The AMA’s fifth annual National Health Insurer Report Card shows that error rates on paid claims dropped from 19% in 2011 to just over 9% in 2012. The organization took some credit for the improvement.
"The AMA has been working constructively with insurers, and we are encouraged by their response to our concerns regarding errors, inefficiency, and waste that take a heavy toll on patients and physicians," Dr. Robert M. Wah, chairman of the AMA board of trustees, said in a statement.
The AMA estimated that the reduction in errors saved the health system $8 billion. If insurers paid claims in a more consistent fashion, another $7 billion could be saved, according to the report card.
The report analyzed claims for seven private insurers –- Aetna, Anthem Blue Cross/Blue Shield, Cigna, Health Care Service Corporation (HCSC), Humana, Regence, and UnitedHealthcare – as well as Medicare. It looked at data based on 1.8 million services billed on 1.1 million claims in February and March of 2012. Some 12,000 physicians submitted data.
Over the last several years, both timeliness and accuracy have improved for this group. From 2008 to the current report, response times have risen by 17%. HCSC and Humana had the fastest response times, at a median of 6 days. Aetna was the slowest, with a median response of 14 days.
Aetna was the third most accurate payer, however, with a 95% accuracy rate. Medicare scored highest, with a 99% rate, followed by United at 98%. Humana and HCSC were the least accurate at 87%.
The good news was tempered by two troubling trends, according to the AMA. After declining from 2008 to 2011, claim denials grew in 2012. In 2011, the overall denial rate among private insurers was 2%; that rose to almost 3.5% in 2012. Anthem Blue Cross/Blue Shield had the highest denial rate (5%).
Private insurers also began making more use of prior authorization. Humana’s prior authorization rate rose from 5% of claims in 2011 to 14% in 2012. That was the highest rate among the seven insurers and Medicare. Cigna had the second highest frequency, at 7%, up from 6% in 2011. Regence and Medicare had the lowest rates of prior authorization, at about 0.7%.
"The costly administrative burdens of the prior authorization process can complicate medical decisions and delay or interrupt patient care," said Dr. Wah. He said that the AMA is seeking to replace "the largely manual process with an automated decision support system that will enhance patient care and reduce paperwork costs."
CHICAGO – Health insurers are paying claims more quickly and with greater accuracy, but they’re also erecting barriers by requiring more prior authorization, a new report from the American Medical Association has found.
The AMA’s fifth annual National Health Insurer Report Card shows that error rates on paid claims dropped from 19% in 2011 to just over 9% in 2012. The organization took some credit for the improvement.
"The AMA has been working constructively with insurers, and we are encouraged by their response to our concerns regarding errors, inefficiency, and waste that take a heavy toll on patients and physicians," Dr. Robert M. Wah, chairman of the AMA board of trustees, said in a statement.
The AMA estimated that the reduction in errors saved the health system $8 billion. If insurers paid claims in a more consistent fashion, another $7 billion could be saved, according to the report card.
The report analyzed claims for seven private insurers –- Aetna, Anthem Blue Cross/Blue Shield, Cigna, Health Care Service Corporation (HCSC), Humana, Regence, and UnitedHealthcare – as well as Medicare. It looked at data based on 1.8 million services billed on 1.1 million claims in February and March of 2012. Some 12,000 physicians submitted data.
Over the last several years, both timeliness and accuracy have improved for this group. From 2008 to the current report, response times have risen by 17%. HCSC and Humana had the fastest response times, at a median of 6 days. Aetna was the slowest, with a median response of 14 days.
Aetna was the third most accurate payer, however, with a 95% accuracy rate. Medicare scored highest, with a 99% rate, followed by United at 98%. Humana and HCSC were the least accurate at 87%.
The good news was tempered by two troubling trends, according to the AMA. After declining from 2008 to 2011, claim denials grew in 2012. In 2011, the overall denial rate among private insurers was 2%; that rose to almost 3.5% in 2012. Anthem Blue Cross/Blue Shield had the highest denial rate (5%).
Private insurers also began making more use of prior authorization. Humana’s prior authorization rate rose from 5% of claims in 2011 to 14% in 2012. That was the highest rate among the seven insurers and Medicare. Cigna had the second highest frequency, at 7%, up from 6% in 2011. Regence and Medicare had the lowest rates of prior authorization, at about 0.7%.
"The costly administrative burdens of the prior authorization process can complicate medical decisions and delay or interrupt patient care," said Dr. Wah. He said that the AMA is seeking to replace "the largely manual process with an automated decision support system that will enhance patient care and reduce paperwork costs."
CHICAGO – Health insurers are paying claims more quickly and with greater accuracy, but they’re also erecting barriers by requiring more prior authorization, a new report from the American Medical Association has found.
The AMA’s fifth annual National Health Insurer Report Card shows that error rates on paid claims dropped from 19% in 2011 to just over 9% in 2012. The organization took some credit for the improvement.
"The AMA has been working constructively with insurers, and we are encouraged by their response to our concerns regarding errors, inefficiency, and waste that take a heavy toll on patients and physicians," Dr. Robert M. Wah, chairman of the AMA board of trustees, said in a statement.
The AMA estimated that the reduction in errors saved the health system $8 billion. If insurers paid claims in a more consistent fashion, another $7 billion could be saved, according to the report card.
The report analyzed claims for seven private insurers –- Aetna, Anthem Blue Cross/Blue Shield, Cigna, Health Care Service Corporation (HCSC), Humana, Regence, and UnitedHealthcare – as well as Medicare. It looked at data based on 1.8 million services billed on 1.1 million claims in February and March of 2012. Some 12,000 physicians submitted data.
Over the last several years, both timeliness and accuracy have improved for this group. From 2008 to the current report, response times have risen by 17%. HCSC and Humana had the fastest response times, at a median of 6 days. Aetna was the slowest, with a median response of 14 days.
Aetna was the third most accurate payer, however, with a 95% accuracy rate. Medicare scored highest, with a 99% rate, followed by United at 98%. Humana and HCSC were the least accurate at 87%.
The good news was tempered by two troubling trends, according to the AMA. After declining from 2008 to 2011, claim denials grew in 2012. In 2011, the overall denial rate among private insurers was 2%; that rose to almost 3.5% in 2012. Anthem Blue Cross/Blue Shield had the highest denial rate (5%).
Private insurers also began making more use of prior authorization. Humana’s prior authorization rate rose from 5% of claims in 2011 to 14% in 2012. That was the highest rate among the seven insurers and Medicare. Cigna had the second highest frequency, at 7%, up from 6% in 2011. Regence and Medicare had the lowest rates of prior authorization, at about 0.7%.
"The costly administrative burdens of the prior authorization process can complicate medical decisions and delay or interrupt patient care," said Dr. Wah. He said that the AMA is seeking to replace "the largely manual process with an automated decision support system that will enhance patient care and reduce paperwork costs."
AT THE AMERICAN MEDICAL ASSOCIATION ANNUAL HOUSE OF DELEGATES MEETING
AMA Tries to Rally Members With New Vision
CHICAGO – As its annual House of Delegates meeting opened, officials from the American Medical Association sought to convince members that the organization remains as vital as ever to advancing physicians’ cause in Washington, in the state houses, and with insurers.
The pep talk came as the officials noted that, after a slide in membership over the last few years, the AMA added 1,636 members in 2011, for a gain of 0.8%. The group ended the year with 217,490 members.
Overall, there are about 954,000 physicians in America, according to AMA data.
"This past year, our membership increased modestly," said Dr. James Madara, executive vice president and chief executive officer of the AMA. "And while we are pleased, we are hardly complacent."
The AMA continues to feel the fallout related to its support of the Affordable Care Act (ACA). Its endorsement led to membership defections and fractious debate at the House of Delegates meetings over the last few years.
While the current meeting was shaping up to be less controversial, in committee meetings on Sunday – held in advance of Monday and Tuesday’s floor debates and votes – some delegates expressed their continued dismay with the health reform law, in particular the Independent Payment Advisory Board (IPAB).
In his address on Saturday, AMA President Peter Carmel made oblique reference to the organization’s role in health reform. He said that, by using the AMA’s position as "the indisputable voice of America’s physicians," the organization had become an advocate for health reform and for the uninsured in 2007.
Without citing the ACA by name, Dr. Carmel highlighted its achievements. He noted that 2.5 million Americans under age 26 years had gained insurance through their parents, and that 100 million people no longer had to worry about lifetime insurance caps. Another 54 million had gained prevention and wellness coverage, Dr. Carmel said.
"All of this has already happened – today – regardless of what the Supreme Court decides," he added.
His recitations drew lukewarm applause.
Dr. Carmel acknowledged that the ACA and the changing health care environment had put physicians on edge. "The only thing worse than that anxiety is the fear many physicians have: the fear that they’re being lied to," he said. For instance, said Dr. Carmel, "If Congress really wants to improve the health care system, why hasn’t it eliminated the [Sustainable Growth Rate formula]?"
He said that the AMA had notched several recent legislative and regulatory victories, including leaning on the Centers for Medicare and Medicaid Services to make accountable care organization criteria more physician friendly and to delay the ICD-10 implementation.
But Dr. Madara said it was not enough. "The AMA must bring a tighter focus to what we do so that our efforts are concentrated on the long term," he said.
The AMA’s senior management has spent months putting together what he called a "rolling 5-year strategic plan." The goal is to ensure that the AMA attracts a broader physician base and has the "credibility, competence, and resources to make a significant impact," he said.
The areas of most intense focus will be improving health outcomes, accelerating change in medical education, and "shaping delivery and payment models that demonstrate high-quality care and value while enhancing physician satisfaction and practice sustainability," Dr. Madara said.
In the outcomes area, the AMA will build on its Physician Consortium for Performance Improvement. It will select two to three outcomes this year to put on a national "AMA dashboard" that will be promoted to quality improvement organizations and physicians. The outcomes will be tracked and used to evaluate impact on productivity and costs.
The goal in the education area is to close the gap between how physicians are being trained and the future needs of the health system, Dr. Madara said. The AMA will push for a new emphasis on learning around patient-centered care and performance improvement and understanding of health care financing and delivery.
Finally, the AMA aims to study what makes physicians happy and then use that data to help develop new practice models, he said. "We will use this information to drive and implement change across practice settings by showcasing delivery and payment models that demonstrate high quality and value while preserving, restoring, and enhancing professional satisfaction for physicians," said Dr. Madara.
The process won’t be easy, but he said that the AMA had hired Dr. Francis J. Crosson to help. Dr. Crosson, a former executive on the physician side of Kaiser Permanente and a former Medicare Payment Advisory Commission member, will join the AMA on July 1 as the vice president of professional satisfaction, care delivery, and payment.
Dr. Madara said that the AMA had "changed the world during its 165-year history" and that he expected more in the future. "No other organization has done more to shape health and health care in this country than has our AMA," he said.
CHICAGO – As its annual House of Delegates meeting opened, officials from the American Medical Association sought to convince members that the organization remains as vital as ever to advancing physicians’ cause in Washington, in the state houses, and with insurers.
The pep talk came as the officials noted that, after a slide in membership over the last few years, the AMA added 1,636 members in 2011, for a gain of 0.8%. The group ended the year with 217,490 members.
Overall, there are about 954,000 physicians in America, according to AMA data.
"This past year, our membership increased modestly," said Dr. James Madara, executive vice president and chief executive officer of the AMA. "And while we are pleased, we are hardly complacent."
The AMA continues to feel the fallout related to its support of the Affordable Care Act (ACA). Its endorsement led to membership defections and fractious debate at the House of Delegates meetings over the last few years.
While the current meeting was shaping up to be less controversial, in committee meetings on Sunday – held in advance of Monday and Tuesday’s floor debates and votes – some delegates expressed their continued dismay with the health reform law, in particular the Independent Payment Advisory Board (IPAB).
In his address on Saturday, AMA President Peter Carmel made oblique reference to the organization’s role in health reform. He said that, by using the AMA’s position as "the indisputable voice of America’s physicians," the organization had become an advocate for health reform and for the uninsured in 2007.
Without citing the ACA by name, Dr. Carmel highlighted its achievements. He noted that 2.5 million Americans under age 26 years had gained insurance through their parents, and that 100 million people no longer had to worry about lifetime insurance caps. Another 54 million had gained prevention and wellness coverage, Dr. Carmel said.
"All of this has already happened – today – regardless of what the Supreme Court decides," he added.
His recitations drew lukewarm applause.
Dr. Carmel acknowledged that the ACA and the changing health care environment had put physicians on edge. "The only thing worse than that anxiety is the fear many physicians have: the fear that they’re being lied to," he said. For instance, said Dr. Carmel, "If Congress really wants to improve the health care system, why hasn’t it eliminated the [Sustainable Growth Rate formula]?"
He said that the AMA had notched several recent legislative and regulatory victories, including leaning on the Centers for Medicare and Medicaid Services to make accountable care organization criteria more physician friendly and to delay the ICD-10 implementation.
But Dr. Madara said it was not enough. "The AMA must bring a tighter focus to what we do so that our efforts are concentrated on the long term," he said.
The AMA’s senior management has spent months putting together what he called a "rolling 5-year strategic plan." The goal is to ensure that the AMA attracts a broader physician base and has the "credibility, competence, and resources to make a significant impact," he said.
The areas of most intense focus will be improving health outcomes, accelerating change in medical education, and "shaping delivery and payment models that demonstrate high-quality care and value while enhancing physician satisfaction and practice sustainability," Dr. Madara said.
In the outcomes area, the AMA will build on its Physician Consortium for Performance Improvement. It will select two to three outcomes this year to put on a national "AMA dashboard" that will be promoted to quality improvement organizations and physicians. The outcomes will be tracked and used to evaluate impact on productivity and costs.
The goal in the education area is to close the gap between how physicians are being trained and the future needs of the health system, Dr. Madara said. The AMA will push for a new emphasis on learning around patient-centered care and performance improvement and understanding of health care financing and delivery.
Finally, the AMA aims to study what makes physicians happy and then use that data to help develop new practice models, he said. "We will use this information to drive and implement change across practice settings by showcasing delivery and payment models that demonstrate high quality and value while preserving, restoring, and enhancing professional satisfaction for physicians," said Dr. Madara.
The process won’t be easy, but he said that the AMA had hired Dr. Francis J. Crosson to help. Dr. Crosson, a former executive on the physician side of Kaiser Permanente and a former Medicare Payment Advisory Commission member, will join the AMA on July 1 as the vice president of professional satisfaction, care delivery, and payment.
Dr. Madara said that the AMA had "changed the world during its 165-year history" and that he expected more in the future. "No other organization has done more to shape health and health care in this country than has our AMA," he said.
CHICAGO – As its annual House of Delegates meeting opened, officials from the American Medical Association sought to convince members that the organization remains as vital as ever to advancing physicians’ cause in Washington, in the state houses, and with insurers.
The pep talk came as the officials noted that, after a slide in membership over the last few years, the AMA added 1,636 members in 2011, for a gain of 0.8%. The group ended the year with 217,490 members.
Overall, there are about 954,000 physicians in America, according to AMA data.
"This past year, our membership increased modestly," said Dr. James Madara, executive vice president and chief executive officer of the AMA. "And while we are pleased, we are hardly complacent."
The AMA continues to feel the fallout related to its support of the Affordable Care Act (ACA). Its endorsement led to membership defections and fractious debate at the House of Delegates meetings over the last few years.
While the current meeting was shaping up to be less controversial, in committee meetings on Sunday – held in advance of Monday and Tuesday’s floor debates and votes – some delegates expressed their continued dismay with the health reform law, in particular the Independent Payment Advisory Board (IPAB).
In his address on Saturday, AMA President Peter Carmel made oblique reference to the organization’s role in health reform. He said that, by using the AMA’s position as "the indisputable voice of America’s physicians," the organization had become an advocate for health reform and for the uninsured in 2007.
Without citing the ACA by name, Dr. Carmel highlighted its achievements. He noted that 2.5 million Americans under age 26 years had gained insurance through their parents, and that 100 million people no longer had to worry about lifetime insurance caps. Another 54 million had gained prevention and wellness coverage, Dr. Carmel said.
"All of this has already happened – today – regardless of what the Supreme Court decides," he added.
His recitations drew lukewarm applause.
Dr. Carmel acknowledged that the ACA and the changing health care environment had put physicians on edge. "The only thing worse than that anxiety is the fear many physicians have: the fear that they’re being lied to," he said. For instance, said Dr. Carmel, "If Congress really wants to improve the health care system, why hasn’t it eliminated the [Sustainable Growth Rate formula]?"
He said that the AMA had notched several recent legislative and regulatory victories, including leaning on the Centers for Medicare and Medicaid Services to make accountable care organization criteria more physician friendly and to delay the ICD-10 implementation.
But Dr. Madara said it was not enough. "The AMA must bring a tighter focus to what we do so that our efforts are concentrated on the long term," he said.
The AMA’s senior management has spent months putting together what he called a "rolling 5-year strategic plan." The goal is to ensure that the AMA attracts a broader physician base and has the "credibility, competence, and resources to make a significant impact," he said.
The areas of most intense focus will be improving health outcomes, accelerating change in medical education, and "shaping delivery and payment models that demonstrate high-quality care and value while enhancing physician satisfaction and practice sustainability," Dr. Madara said.
In the outcomes area, the AMA will build on its Physician Consortium for Performance Improvement. It will select two to three outcomes this year to put on a national "AMA dashboard" that will be promoted to quality improvement organizations and physicians. The outcomes will be tracked and used to evaluate impact on productivity and costs.
The goal in the education area is to close the gap between how physicians are being trained and the future needs of the health system, Dr. Madara said. The AMA will push for a new emphasis on learning around patient-centered care and performance improvement and understanding of health care financing and delivery.
Finally, the AMA aims to study what makes physicians happy and then use that data to help develop new practice models, he said. "We will use this information to drive and implement change across practice settings by showcasing delivery and payment models that demonstrate high quality and value while preserving, restoring, and enhancing professional satisfaction for physicians," said Dr. Madara.
The process won’t be easy, but he said that the AMA had hired Dr. Francis J. Crosson to help. Dr. Crosson, a former executive on the physician side of Kaiser Permanente and a former Medicare Payment Advisory Commission member, will join the AMA on July 1 as the vice president of professional satisfaction, care delivery, and payment.
Dr. Madara said that the AMA had "changed the world during its 165-year history" and that he expected more in the future. "No other organization has done more to shape health and health care in this country than has our AMA," he said.
AT THE ANNUAL MEETING OF THE AMERICAN MEDICAL ASSOCIATION’S HOUSE OF DELEGATES
Pediatric Medical Home Model Cuts Neurology Costs
NEW ORLEANS – Use of a variation on the traditional medical home model for pediatric neurology cases in an underserved population led to a decrease in hospitalizations and less frequent use of the emergency department in a study that compared outcomes and health care resource utilization between the two models.
The new model also led to faster referrals and fewer broken appointments, said Dr. David Urion, director of the learning disabilities and behavioral neurology program at Children’s Hospital in Boston, who led the study.
It is already well known that access to primary care is not as great for Americans who have a lower socioeconomic status, he said. The rate of access is even lower among non-English speakers, hovering around 70%, Dr. Urion said at the annual meeting of the American Academy of Neurology.
Access to specialists is generally lower than access to primary care.
In Boston, the Community Health Center movement has tried to improve access, in part by using a medical home model. Dr. Urion and his colleagues studied two different medical home models utilized by two centers – the South End Community Health Center and the Martha Eliot Community Health Center. Both are located in predominantly Hispanic and poorer areas.
One medical home model was what would be considered "traditional," in which patients were referred by the health center to a specialist at a hospital outpatient clinic. The other was an "itinerant" model, where the specialist would hold office hours at the center on a set day of the week. Patients were referred by a primary care physician.
From 2007 to 2011, the traditional model saw about 105 patients a year, while the itinerant model had about 103 patients a year. This was the equivalent of one session per week per provider, Dr. Urion said.
For the traditional model, the median time to an appointment was 90 days. The itinerant model cut that by 76 days to a wait of only 14 days for an appointment. Only 67% of new patients kept their appointment with the traditional model, compared with 95% of those in the nontraditional model. For existing patients, only 50% kept an appointment in the traditional model, compared with 85% of those in the newer model.
The use of the emergency department was reduced 18-fold, with 1.8 visits per patient-year for the traditional model and 0.1 per patient-year for the nontraditional model. Hospitalization days per neurologic diagnosis were 0.4 days per patient-year for the traditional model, compared with 0.1 for the itinerant model.
There was a huge difference in costs. The traditional model’s charges were $14,650 per year, compared with only $1,850 for the itinerant model.
Physicians in the itinerant model also won out financially. With missed appointments and follow-ups, those in the traditional model lost $17,250 a year, compared with only $4,080 a year with the itinerant model.
Thus, the itinerant model delivered superior care, said Dr. Urion, who reported having no relevant disclosures.
NEW ORLEANS – Use of a variation on the traditional medical home model for pediatric neurology cases in an underserved population led to a decrease in hospitalizations and less frequent use of the emergency department in a study that compared outcomes and health care resource utilization between the two models.
The new model also led to faster referrals and fewer broken appointments, said Dr. David Urion, director of the learning disabilities and behavioral neurology program at Children’s Hospital in Boston, who led the study.
It is already well known that access to primary care is not as great for Americans who have a lower socioeconomic status, he said. The rate of access is even lower among non-English speakers, hovering around 70%, Dr. Urion said at the annual meeting of the American Academy of Neurology.
Access to specialists is generally lower than access to primary care.
In Boston, the Community Health Center movement has tried to improve access, in part by using a medical home model. Dr. Urion and his colleagues studied two different medical home models utilized by two centers – the South End Community Health Center and the Martha Eliot Community Health Center. Both are located in predominantly Hispanic and poorer areas.
One medical home model was what would be considered "traditional," in which patients were referred by the health center to a specialist at a hospital outpatient clinic. The other was an "itinerant" model, where the specialist would hold office hours at the center on a set day of the week. Patients were referred by a primary care physician.
From 2007 to 2011, the traditional model saw about 105 patients a year, while the itinerant model had about 103 patients a year. This was the equivalent of one session per week per provider, Dr. Urion said.
For the traditional model, the median time to an appointment was 90 days. The itinerant model cut that by 76 days to a wait of only 14 days for an appointment. Only 67% of new patients kept their appointment with the traditional model, compared with 95% of those in the nontraditional model. For existing patients, only 50% kept an appointment in the traditional model, compared with 85% of those in the newer model.
The use of the emergency department was reduced 18-fold, with 1.8 visits per patient-year for the traditional model and 0.1 per patient-year for the nontraditional model. Hospitalization days per neurologic diagnosis were 0.4 days per patient-year for the traditional model, compared with 0.1 for the itinerant model.
There was a huge difference in costs. The traditional model’s charges were $14,650 per year, compared with only $1,850 for the itinerant model.
Physicians in the itinerant model also won out financially. With missed appointments and follow-ups, those in the traditional model lost $17,250 a year, compared with only $4,080 a year with the itinerant model.
Thus, the itinerant model delivered superior care, said Dr. Urion, who reported having no relevant disclosures.
NEW ORLEANS – Use of a variation on the traditional medical home model for pediatric neurology cases in an underserved population led to a decrease in hospitalizations and less frequent use of the emergency department in a study that compared outcomes and health care resource utilization between the two models.
The new model also led to faster referrals and fewer broken appointments, said Dr. David Urion, director of the learning disabilities and behavioral neurology program at Children’s Hospital in Boston, who led the study.
It is already well known that access to primary care is not as great for Americans who have a lower socioeconomic status, he said. The rate of access is even lower among non-English speakers, hovering around 70%, Dr. Urion said at the annual meeting of the American Academy of Neurology.
Access to specialists is generally lower than access to primary care.
In Boston, the Community Health Center movement has tried to improve access, in part by using a medical home model. Dr. Urion and his colleagues studied two different medical home models utilized by two centers – the South End Community Health Center and the Martha Eliot Community Health Center. Both are located in predominantly Hispanic and poorer areas.
One medical home model was what would be considered "traditional," in which patients were referred by the health center to a specialist at a hospital outpatient clinic. The other was an "itinerant" model, where the specialist would hold office hours at the center on a set day of the week. Patients were referred by a primary care physician.
From 2007 to 2011, the traditional model saw about 105 patients a year, while the itinerant model had about 103 patients a year. This was the equivalent of one session per week per provider, Dr. Urion said.
For the traditional model, the median time to an appointment was 90 days. The itinerant model cut that by 76 days to a wait of only 14 days for an appointment. Only 67% of new patients kept their appointment with the traditional model, compared with 95% of those in the nontraditional model. For existing patients, only 50% kept an appointment in the traditional model, compared with 85% of those in the newer model.
The use of the emergency department was reduced 18-fold, with 1.8 visits per patient-year for the traditional model and 0.1 per patient-year for the nontraditional model. Hospitalization days per neurologic diagnosis were 0.4 days per patient-year for the traditional model, compared with 0.1 for the itinerant model.
There was a huge difference in costs. The traditional model’s charges were $14,650 per year, compared with only $1,850 for the itinerant model.
Physicians in the itinerant model also won out financially. With missed appointments and follow-ups, those in the traditional model lost $17,250 a year, compared with only $4,080 a year with the itinerant model.
Thus, the itinerant model delivered superior care, said Dr. Urion, who reported having no relevant disclosures.
FROM THE ANNUAL MEETING OF THE AMERICAN ACADEMY OF NEUROLOGY
Vismodegib Continues to Prove BCC Effectiveness
Two small, early-phase studies found that vismodegib is effective for the treatment of basal cell nevus syndrome and advanced basal cell carcinoma, according to results published June 6 in the New England Journal of Medicine.
Basal cell malignancies are among the most common nonmelanoma skin cancers in the United States, accounting for at least 80% of the 2.1 million cases. It is a highly disfiguring cancer but is only sometimes life threatening. Almost all basal cell carcinomas (BCCs) are caused by alterations in the hedgehog signaling pathway.
Vismodegib (Erivedge) was approved by the Food and Drug Administration in January for treating locally advanced and metastatic BCC. Vismodegib (manufactured by Genentech) is a small molecule inhibitor that steps in to repair the hedgehog pathway.
Study 1: Locally Advanced and Metastatic BCC
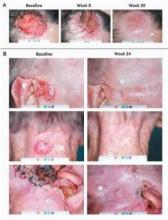
In the first study, conducted before vismodegib received FDA approval for locally advanced and metastatic BCC, Dr. Aleksandar Sekulic and colleagues conducted a phase II, nonrandomized, two-cohort trial at multiple sites around the United States and overseas. Vismodegib was given to 33 patients with metastatic BCC and 63 patients with locally advanced disease. Both cohorts had inoperable disease or were not eligible for surgery.
All the patients were treated with 150 mg of oral vismodegib daily. The median age of patients was 62 years and all were white, noted Dr. Sekulic of the Mayo Clinic in Scottsdale, Ariz.
The patients were treated with vismodegib until disease progression, until side effects became intolerable, or until end of study. Patients could stop the drug for up to a month if side effects were intolerable.
There was no standard end point for measuring response in locally advanced BCC when the study was designed. The definition used was a decrease of 30% or more of the externally visible or radiographic dimension or complete resolution of ulceration. Progressive disease was an increase of 20% or more in the externally visible or radiographic dimension, new ulceration, or a new lesion.
The response rate for the metastatic group treated with vismodegib was 30%. All of the responses were partial, defined as an absence of residual BCC in a biopsy specimen. In locally advanced disease, the response rate was 43%. Thirteen of the 63 patients had a complete response. The median duration of response was 7.6 months for metastatic and locally advanced BCC patients (N. Engl. J. Med. 2012;366:2171-9).
Half of the patients stopped the drug early, with 18% of metastatic patients discontinuing because of disease progression. Twenty-five percent of locally advanced patients decided on their own to stop therapy, for unknown reasons, according to the investigators. Adverse events – primarily muscle spasms, weight loss, fatigue, and loss of appetite – took a toll on patients. A quarter reported serious adverse events.
Seven patients died, but the relation to vismodegib is not clear at this point. "The deaths were considered by the site investigator to be unrelated to vismodegib," the investigators wrote.
The locally advanced and metastatic BCC trial was funded by Genentech and was jointly designed by Genetech and Dr. Sekulic. A majority of his colleagues reported being employees and/or receiving grants or financial support from the company.
In an interview, Dr. Ali Hendi, assistant clinical professor of dermatology at Georgetown University Medical Center, Washington, said that he felt the investigators in the study were overly subjective in defining locally advanced tumors. "These are tumors that are aggressive, but that does not make them inoperable in the right hands," said Dr. Hendi, who is also a Mohs surgeon.
He also noted the slim complete response rate, which, when coupled with the toxicity and the high cost of the therapy – which might run as much as $70,000 per treatment course – make it a less desirable therapy for many patients.
"For locally advanced [disease] I think the utility is limited if there is good surgical care available," he said. Vismodegib may be more useful for BCC nevus syndrome and for metastatic disease, where it may be the only therapy available, he added.
Dr. Hendi reported no conflicts.
Study 2: Basal Cell Nevus Syndrome
In a second randomized, double-blind, placebo-controlled trial of 41 patients with basal cell nevus syndrome, Dr. Jean Y. Tang and colleagues found that vismodegib reduced BCC tumors and blocked the development of new BCCs.
Basal cell nevus syndrome is rare, but it causes hundreds or thousands of carcinomas in each patient. There is no therapy, and often patients have to undergo multiple surgeries to remove the tumors.
Patients with the condition have one defective copy of the tumor suppressor gene (PTCH1), which inhibits the hedgehog signaling pathway, noted Dr. Tang of Stanford University (Calif.), and her colleagues.
The study enrolled patients from Sept. 2009 through Jan. 2011, but in Dec. 2010 the data safety and monitoring board decided to end the placebo treatment because the results so highly favored vismodegib. Patients were randomized to placebo or 150 mg daily of the drug for a planned 18 months.
The primary end point was the comparative rate of appearance of new BCCs that were eligible for surgery. Patients were followed for a mean of 8 months. Those who took vismodegib had a much lower rate of surgically eligible disease: 2 vs. 29 cases for placebo. Existing carcinomas shrunk by 65% in vismodegib-treated patients, compared with only 11% for placebo patients. Vismodegib patients also had fewer surgeries than patients receiving placebo: 0.31 per patient compared with 4.4 per patient, respectively.
At 1 month, there was a 90% reduction in hedgehog target-gene expression. There was no residual carcinoma in 83% of the biopsy samples taken from sites where the disease had been judged to be clinically regressed (N. Engl. J. Med. 2012;366:2180-8).
Fifty-four percent of study patients discontinued treatment because of adverse events. Patients treated with vismodegib were more likely to have grade 1 or 2 dysgeusia, muscle cramps, hair loss, and weight loss. They also had more grade 3 or 4 adverse events. The authors said that these side effects were similar to those reported in phase 1 and 2 trials of vismodegib, and also in other studies of therapies that inhibit the hedgehog pathway.
The authors concluded that vismodegib reduces tumor burden and blocks new tumor growth, but whether some of the clinically regressed tumors still harbor residual tumor cells cannot be ruled out – which could explain why there is tumor regrowth after therapy is stopped.
However, the "findings confirm the essential role of the hedgehog pathway in basal cell carcinomas," they wrote.
The trial was funded by Genentech and by grants from the National Institutes of Health, the National Institute of Arthritis and Musculoskeletal and Skin Diseases, the National Cancer Institute, the Swim Across America Foundation, and the Michael J. Rainen Family Foundation. Dr. Tang reported receiving consulting fees from Genentech. Several coauthors of the study reported receiving grants or fees from Genentech.
The two studies show that vismodegib is highly effective. "It is a landmark day for patients with basal cell carcinoma and all those involved in their care – the greatest advance in therapy yet seen for this disease," Dr. John T. Lear wrote in an accompanying editorial.
The response rates were impressive, especially in patients with basal cell nevus syndrome.
"However, the side effects are considerable and frequent, resulting in high rates of drug discontinuation, and these rates will probably be even higher in clinical practice," he said.
Another question raised is whether hedgehog pathway inhibition truly clears the BCC or whether it leaves residual resistant cells that could give rise to a recurrence. Follow-up studies are needed to address this question, Dr. Lear said (N. Engl. J. Med. 2012;366:2225-6).
Intermittent dosing might allow more patients to benefit by preventing new lesions. This would be especially important for BCC nevus syndrome. And for patients with large, symptomatic lesions, "the usefulness of local control in improving quality of life should not be underestimated," he noted.
Dr. Lear is affiliated with the Manchester Academic Health Science Centre at Manchester (England) University and the departments of dermatology at Salford Royal Hospital and Central Manchester National Health Service Foundation Trusts. He has received consulting fees from Novartis for an ongoing BCC study.
The two studies show that vismodegib is highly effective. "It is a landmark day for patients with basal cell carcinoma and all those involved in their care – the greatest advance in therapy yet seen for this disease," Dr. John T. Lear wrote in an accompanying editorial.
The response rates were impressive, especially in patients with basal cell nevus syndrome.
"However, the side effects are considerable and frequent, resulting in high rates of drug discontinuation, and these rates will probably be even higher in clinical practice," he said.
Another question raised is whether hedgehog pathway inhibition truly clears the BCC or whether it leaves residual resistant cells that could give rise to a recurrence. Follow-up studies are needed to address this question, Dr. Lear said (N. Engl. J. Med. 2012;366:2225-6).
Intermittent dosing might allow more patients to benefit by preventing new lesions. This would be especially important for BCC nevus syndrome. And for patients with large, symptomatic lesions, "the usefulness of local control in improving quality of life should not be underestimated," he noted.
Dr. Lear is affiliated with the Manchester Academic Health Science Centre at Manchester (England) University and the departments of dermatology at Salford Royal Hospital and Central Manchester National Health Service Foundation Trusts. He has received consulting fees from Novartis for an ongoing BCC study.
The two studies show that vismodegib is highly effective. "It is a landmark day for patients with basal cell carcinoma and all those involved in their care – the greatest advance in therapy yet seen for this disease," Dr. John T. Lear wrote in an accompanying editorial.
The response rates were impressive, especially in patients with basal cell nevus syndrome.
"However, the side effects are considerable and frequent, resulting in high rates of drug discontinuation, and these rates will probably be even higher in clinical practice," he said.
Another question raised is whether hedgehog pathway inhibition truly clears the BCC or whether it leaves residual resistant cells that could give rise to a recurrence. Follow-up studies are needed to address this question, Dr. Lear said (N. Engl. J. Med. 2012;366:2225-6).
Intermittent dosing might allow more patients to benefit by preventing new lesions. This would be especially important for BCC nevus syndrome. And for patients with large, symptomatic lesions, "the usefulness of local control in improving quality of life should not be underestimated," he noted.
Dr. Lear is affiliated with the Manchester Academic Health Science Centre at Manchester (England) University and the departments of dermatology at Salford Royal Hospital and Central Manchester National Health Service Foundation Trusts. He has received consulting fees from Novartis for an ongoing BCC study.
Two small, early-phase studies found that vismodegib is effective for the treatment of basal cell nevus syndrome and advanced basal cell carcinoma, according to results published June 6 in the New England Journal of Medicine.
Basal cell malignancies are among the most common nonmelanoma skin cancers in the United States, accounting for at least 80% of the 2.1 million cases. It is a highly disfiguring cancer but is only sometimes life threatening. Almost all basal cell carcinomas (BCCs) are caused by alterations in the hedgehog signaling pathway.
Vismodegib (Erivedge) was approved by the Food and Drug Administration in January for treating locally advanced and metastatic BCC. Vismodegib (manufactured by Genentech) is a small molecule inhibitor that steps in to repair the hedgehog pathway.
Study 1: Locally Advanced and Metastatic BCC
In the first study, conducted before vismodegib received FDA approval for locally advanced and metastatic BCC, Dr. Aleksandar Sekulic and colleagues conducted a phase II, nonrandomized, two-cohort trial at multiple sites around the United States and overseas. Vismodegib was given to 33 patients with metastatic BCC and 63 patients with locally advanced disease. Both cohorts had inoperable disease or were not eligible for surgery.
All the patients were treated with 150 mg of oral vismodegib daily. The median age of patients was 62 years and all were white, noted Dr. Sekulic of the Mayo Clinic in Scottsdale, Ariz.
The patients were treated with vismodegib until disease progression, until side effects became intolerable, or until end of study. Patients could stop the drug for up to a month if side effects were intolerable.
There was no standard end point for measuring response in locally advanced BCC when the study was designed. The definition used was a decrease of 30% or more of the externally visible or radiographic dimension or complete resolution of ulceration. Progressive disease was an increase of 20% or more in the externally visible or radiographic dimension, new ulceration, or a new lesion.
The response rate for the metastatic group treated with vismodegib was 30%. All of the responses were partial, defined as an absence of residual BCC in a biopsy specimen. In locally advanced disease, the response rate was 43%. Thirteen of the 63 patients had a complete response. The median duration of response was 7.6 months for metastatic and locally advanced BCC patients (N. Engl. J. Med. 2012;366:2171-9).
Half of the patients stopped the drug early, with 18% of metastatic patients discontinuing because of disease progression. Twenty-five percent of locally advanced patients decided on their own to stop therapy, for unknown reasons, according to the investigators. Adverse events – primarily muscle spasms, weight loss, fatigue, and loss of appetite – took a toll on patients. A quarter reported serious adverse events.
Seven patients died, but the relation to vismodegib is not clear at this point. "The deaths were considered by the site investigator to be unrelated to vismodegib," the investigators wrote.
The locally advanced and metastatic BCC trial was funded by Genentech and was jointly designed by Genetech and Dr. Sekulic. A majority of his colleagues reported being employees and/or receiving grants or financial support from the company.
In an interview, Dr. Ali Hendi, assistant clinical professor of dermatology at Georgetown University Medical Center, Washington, said that he felt the investigators in the study were overly subjective in defining locally advanced tumors. "These are tumors that are aggressive, but that does not make them inoperable in the right hands," said Dr. Hendi, who is also a Mohs surgeon.
He also noted the slim complete response rate, which, when coupled with the toxicity and the high cost of the therapy – which might run as much as $70,000 per treatment course – make it a less desirable therapy for many patients.
"For locally advanced [disease] I think the utility is limited if there is good surgical care available," he said. Vismodegib may be more useful for BCC nevus syndrome and for metastatic disease, where it may be the only therapy available, he added.
Dr. Hendi reported no conflicts.
Study 2: Basal Cell Nevus Syndrome
In a second randomized, double-blind, placebo-controlled trial of 41 patients with basal cell nevus syndrome, Dr. Jean Y. Tang and colleagues found that vismodegib reduced BCC tumors and blocked the development of new BCCs.
Basal cell nevus syndrome is rare, but it causes hundreds or thousands of carcinomas in each patient. There is no therapy, and often patients have to undergo multiple surgeries to remove the tumors.
Patients with the condition have one defective copy of the tumor suppressor gene (PTCH1), which inhibits the hedgehog signaling pathway, noted Dr. Tang of Stanford University (Calif.), and her colleagues.
The study enrolled patients from Sept. 2009 through Jan. 2011, but in Dec. 2010 the data safety and monitoring board decided to end the placebo treatment because the results so highly favored vismodegib. Patients were randomized to placebo or 150 mg daily of the drug for a planned 18 months.
The primary end point was the comparative rate of appearance of new BCCs that were eligible for surgery. Patients were followed for a mean of 8 months. Those who took vismodegib had a much lower rate of surgically eligible disease: 2 vs. 29 cases for placebo. Existing carcinomas shrunk by 65% in vismodegib-treated patients, compared with only 11% for placebo patients. Vismodegib patients also had fewer surgeries than patients receiving placebo: 0.31 per patient compared with 4.4 per patient, respectively.
At 1 month, there was a 90% reduction in hedgehog target-gene expression. There was no residual carcinoma in 83% of the biopsy samples taken from sites where the disease had been judged to be clinically regressed (N. Engl. J. Med. 2012;366:2180-8).
Fifty-four percent of study patients discontinued treatment because of adverse events. Patients treated with vismodegib were more likely to have grade 1 or 2 dysgeusia, muscle cramps, hair loss, and weight loss. They also had more grade 3 or 4 adverse events. The authors said that these side effects were similar to those reported in phase 1 and 2 trials of vismodegib, and also in other studies of therapies that inhibit the hedgehog pathway.
The authors concluded that vismodegib reduces tumor burden and blocks new tumor growth, but whether some of the clinically regressed tumors still harbor residual tumor cells cannot be ruled out – which could explain why there is tumor regrowth after therapy is stopped.
However, the "findings confirm the essential role of the hedgehog pathway in basal cell carcinomas," they wrote.
The trial was funded by Genentech and by grants from the National Institutes of Health, the National Institute of Arthritis and Musculoskeletal and Skin Diseases, the National Cancer Institute, the Swim Across America Foundation, and the Michael J. Rainen Family Foundation. Dr. Tang reported receiving consulting fees from Genentech. Several coauthors of the study reported receiving grants or fees from Genentech.
Two small, early-phase studies found that vismodegib is effective for the treatment of basal cell nevus syndrome and advanced basal cell carcinoma, according to results published June 6 in the New England Journal of Medicine.
Basal cell malignancies are among the most common nonmelanoma skin cancers in the United States, accounting for at least 80% of the 2.1 million cases. It is a highly disfiguring cancer but is only sometimes life threatening. Almost all basal cell carcinomas (BCCs) are caused by alterations in the hedgehog signaling pathway.
Vismodegib (Erivedge) was approved by the Food and Drug Administration in January for treating locally advanced and metastatic BCC. Vismodegib (manufactured by Genentech) is a small molecule inhibitor that steps in to repair the hedgehog pathway.
Study 1: Locally Advanced and Metastatic BCC
In the first study, conducted before vismodegib received FDA approval for locally advanced and metastatic BCC, Dr. Aleksandar Sekulic and colleagues conducted a phase II, nonrandomized, two-cohort trial at multiple sites around the United States and overseas. Vismodegib was given to 33 patients with metastatic BCC and 63 patients with locally advanced disease. Both cohorts had inoperable disease or were not eligible for surgery.
All the patients were treated with 150 mg of oral vismodegib daily. The median age of patients was 62 years and all were white, noted Dr. Sekulic of the Mayo Clinic in Scottsdale, Ariz.
The patients were treated with vismodegib until disease progression, until side effects became intolerable, or until end of study. Patients could stop the drug for up to a month if side effects were intolerable.
There was no standard end point for measuring response in locally advanced BCC when the study was designed. The definition used was a decrease of 30% or more of the externally visible or radiographic dimension or complete resolution of ulceration. Progressive disease was an increase of 20% or more in the externally visible or radiographic dimension, new ulceration, or a new lesion.
The response rate for the metastatic group treated with vismodegib was 30%. All of the responses were partial, defined as an absence of residual BCC in a biopsy specimen. In locally advanced disease, the response rate was 43%. Thirteen of the 63 patients had a complete response. The median duration of response was 7.6 months for metastatic and locally advanced BCC patients (N. Engl. J. Med. 2012;366:2171-9).
Half of the patients stopped the drug early, with 18% of metastatic patients discontinuing because of disease progression. Twenty-five percent of locally advanced patients decided on their own to stop therapy, for unknown reasons, according to the investigators. Adverse events – primarily muscle spasms, weight loss, fatigue, and loss of appetite – took a toll on patients. A quarter reported serious adverse events.
Seven patients died, but the relation to vismodegib is not clear at this point. "The deaths were considered by the site investigator to be unrelated to vismodegib," the investigators wrote.
The locally advanced and metastatic BCC trial was funded by Genentech and was jointly designed by Genetech and Dr. Sekulic. A majority of his colleagues reported being employees and/or receiving grants or financial support from the company.
In an interview, Dr. Ali Hendi, assistant clinical professor of dermatology at Georgetown University Medical Center, Washington, said that he felt the investigators in the study were overly subjective in defining locally advanced tumors. "These are tumors that are aggressive, but that does not make them inoperable in the right hands," said Dr. Hendi, who is also a Mohs surgeon.
He also noted the slim complete response rate, which, when coupled with the toxicity and the high cost of the therapy – which might run as much as $70,000 per treatment course – make it a less desirable therapy for many patients.
"For locally advanced [disease] I think the utility is limited if there is good surgical care available," he said. Vismodegib may be more useful for BCC nevus syndrome and for metastatic disease, where it may be the only therapy available, he added.
Dr. Hendi reported no conflicts.
Study 2: Basal Cell Nevus Syndrome
In a second randomized, double-blind, placebo-controlled trial of 41 patients with basal cell nevus syndrome, Dr. Jean Y. Tang and colleagues found that vismodegib reduced BCC tumors and blocked the development of new BCCs.
Basal cell nevus syndrome is rare, but it causes hundreds or thousands of carcinomas in each patient. There is no therapy, and often patients have to undergo multiple surgeries to remove the tumors.
Patients with the condition have one defective copy of the tumor suppressor gene (PTCH1), which inhibits the hedgehog signaling pathway, noted Dr. Tang of Stanford University (Calif.), and her colleagues.
The study enrolled patients from Sept. 2009 through Jan. 2011, but in Dec. 2010 the data safety and monitoring board decided to end the placebo treatment because the results so highly favored vismodegib. Patients were randomized to placebo or 150 mg daily of the drug for a planned 18 months.
The primary end point was the comparative rate of appearance of new BCCs that were eligible for surgery. Patients were followed for a mean of 8 months. Those who took vismodegib had a much lower rate of surgically eligible disease: 2 vs. 29 cases for placebo. Existing carcinomas shrunk by 65% in vismodegib-treated patients, compared with only 11% for placebo patients. Vismodegib patients also had fewer surgeries than patients receiving placebo: 0.31 per patient compared with 4.4 per patient, respectively.
At 1 month, there was a 90% reduction in hedgehog target-gene expression. There was no residual carcinoma in 83% of the biopsy samples taken from sites where the disease had been judged to be clinically regressed (N. Engl. J. Med. 2012;366:2180-8).
Fifty-four percent of study patients discontinued treatment because of adverse events. Patients treated with vismodegib were more likely to have grade 1 or 2 dysgeusia, muscle cramps, hair loss, and weight loss. They also had more grade 3 or 4 adverse events. The authors said that these side effects were similar to those reported in phase 1 and 2 trials of vismodegib, and also in other studies of therapies that inhibit the hedgehog pathway.
The authors concluded that vismodegib reduces tumor burden and blocks new tumor growth, but whether some of the clinically regressed tumors still harbor residual tumor cells cannot be ruled out – which could explain why there is tumor regrowth after therapy is stopped.
However, the "findings confirm the essential role of the hedgehog pathway in basal cell carcinomas," they wrote.
The trial was funded by Genentech and by grants from the National Institutes of Health, the National Institute of Arthritis and Musculoskeletal and Skin Diseases, the National Cancer Institute, the Swim Across America Foundation, and the Michael J. Rainen Family Foundation. Dr. Tang reported receiving consulting fees from Genentech. Several coauthors of the study reported receiving grants or fees from Genentech.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Major Finding: In the first trial, the response rate for vismodegib-treated patients was 30% for the metastatic group and 43% for the locally advanced BCC group. In the basal cell nevus syndrome trial, vismodegib-treated patients had a lower rate of surgically eligible disease: 2 cases vs. 29 cases for placebo.
Data Source: In the first trial, the data came from a nonrandomized, dual-cohort, multicenter, phase II trial of 96 patients. The data for the second trial came from a randomized, placebo-controlled study of 41 patients.
Disclosures: The locally advanced and metastatic BCC trial was funded by Genentech and was jointly designed by Genentech and Dr. Sekulic. A majority of his colleagues reported being employees and/or receiving grants or financial support from the company. The basal cell nevus syndrome trial was funded by Genentech and by grants from the National Institutes of Health, the National Institute of Arthritis and Musculoskeletal and Skin Diseases, the National Cancer Institute, the Swim Across America Foundation, and the Michael J. Rainen Family Foundation. Dr. Tang reported receiving consulting fees from Genentech. Several coauthors of the study reported receiving grants or fees from Genentech.
FDA: Drug Shortages Decline but Persist
The Food and Drug Administration has prevented at least 50 drug shortages so far this year, mainly as a result of manufacturers giving the agency early warning of impending problems, Dr. Sandra Kweder announced June 4.
Shortages persist, however, especially for key oncology therapies, according to Dr. Kweder, deputy director of the FDA Office of New Drugs, and oncologists who participated in a press briefing at the annual meeting of the American Society of Clinical Oncologists (ASCO) in Chicago.
Since the White House issued an executive order last Oct. 31 encouraging manufacturers to give the FDA "early notification" of potential disruptions, the agency, working with physicians, manufacturers, pharmacists, and others, has been able to avoid more than 150 drug shortages, Dr. Kweder said.
"Early notification by manufacturers of when they are having production difficulties makes a huge difference in our ability to prevent the public from bearing the burden of drug shortages," she said.
"The good news is that the frequency of drug shortages has begun to decline," agreed Dr. Richard Schilsky, chair of the ASCO Government Relations Committee and chief of the hematology/oncology section at the University of Chicago.
However, unpredictability still exists, which means that oncologists are "never sure when a generic is going to go out of supply," said Dr. Schilsky. "That creates a tremendous amount of uncertainty and anxiety for our patients."
Dr. Michael P. Link, ASCO president, said that several commonly used chemotherapy agents remain in short supply, including fluorouracil, nitrogen mustard, and paclitaxel. In addition, although a methotrexate shortage has been addressed, there is now a shortage of sodium bicarbonate, which is needed to give high-dose injections of the drug, said Dr. Link, chief of pediatric hematology/oncology at the Lucile Packard Children’s Hospital at Stanford (Calif.) University.
Shortages often lead to delays in treatment or less-than-desirable substitutions, said Dr. W. Charles Penley, the incoming ASCO Government Relations Committee chair. His practice – Tennessee Oncology in Nashville – is large enough that it generally has been able to locate products in short supply through its purchasing groups. But a leucovorin shortage last summer led him to switch some patients to oral capecitabine (Xeloda).
That drug is more expensive and is not tolerated well by some patients, he said.
And even that product almost went into short supply, Dr. Kweder said. There were some quality control issues that led the agency to temporarily import capecitabine while the manufacturer, Roche, fixed the problem, she said.
Manufacturing and quality control – especially for generic sterile injectables – continue to be behind many shortages, Dr. Kweder said. Sterility has been a consistent problem, with many products found to contain glass particles or metal shavings. She described a zero tolerance policy for such issues with injectables. "No patient should ever be exposed to risks of those sorts," she said.
When asked why generic injectables seem to be having more frequent quality control issues, Dr. Kweder said that the requirements and rules for good manufacturing have not changed over the years, but that the explosion of generic products in the last decade has perhaps put more pressure on manufacturers.
Dr. Schilsky and Dr. Penley said they were hopeful that legislation creating user fees for generic drugmakers would help address the shortage issue. Bills to reauthorize all FDA user fee programs have passed both the House and the Senate; differences are being hashed out in conference right now. The generic user fee – expected to add $1.5 billion to the FDA budget over the next few years – is expected to be retained. Experts expect the president to sign the final bill, said Dr. Schilsky.
The additional funds would give the FDA the ability to cut generic drug review times from the current 24-30 months to about 6-10 months, Dr. Kweder said.
It also would give the agency more resources to eradicate a current backlog of reviews and move more quickly on manufacturing issues, she said.
The legislation includes several additional items on ASCO’s wish list, including a requirement that manufacturers give the FDA early warning of impending shortages. But it does not include any penalties for noncompliance, said Dr. Schilsky.
The Food and Drug Administration has prevented at least 50 drug shortages so far this year, mainly as a result of manufacturers giving the agency early warning of impending problems, Dr. Sandra Kweder announced June 4.
Shortages persist, however, especially for key oncology therapies, according to Dr. Kweder, deputy director of the FDA Office of New Drugs, and oncologists who participated in a press briefing at the annual meeting of the American Society of Clinical Oncologists (ASCO) in Chicago.
Since the White House issued an executive order last Oct. 31 encouraging manufacturers to give the FDA "early notification" of potential disruptions, the agency, working with physicians, manufacturers, pharmacists, and others, has been able to avoid more than 150 drug shortages, Dr. Kweder said.
"Early notification by manufacturers of when they are having production difficulties makes a huge difference in our ability to prevent the public from bearing the burden of drug shortages," she said.
"The good news is that the frequency of drug shortages has begun to decline," agreed Dr. Richard Schilsky, chair of the ASCO Government Relations Committee and chief of the hematology/oncology section at the University of Chicago.
However, unpredictability still exists, which means that oncologists are "never sure when a generic is going to go out of supply," said Dr. Schilsky. "That creates a tremendous amount of uncertainty and anxiety for our patients."
Dr. Michael P. Link, ASCO president, said that several commonly used chemotherapy agents remain in short supply, including fluorouracil, nitrogen mustard, and paclitaxel. In addition, although a methotrexate shortage has been addressed, there is now a shortage of sodium bicarbonate, which is needed to give high-dose injections of the drug, said Dr. Link, chief of pediatric hematology/oncology at the Lucile Packard Children’s Hospital at Stanford (Calif.) University.
Shortages often lead to delays in treatment or less-than-desirable substitutions, said Dr. W. Charles Penley, the incoming ASCO Government Relations Committee chair. His practice – Tennessee Oncology in Nashville – is large enough that it generally has been able to locate products in short supply through its purchasing groups. But a leucovorin shortage last summer led him to switch some patients to oral capecitabine (Xeloda).
That drug is more expensive and is not tolerated well by some patients, he said.
And even that product almost went into short supply, Dr. Kweder said. There were some quality control issues that led the agency to temporarily import capecitabine while the manufacturer, Roche, fixed the problem, she said.
Manufacturing and quality control – especially for generic sterile injectables – continue to be behind many shortages, Dr. Kweder said. Sterility has been a consistent problem, with many products found to contain glass particles or metal shavings. She described a zero tolerance policy for such issues with injectables. "No patient should ever be exposed to risks of those sorts," she said.
When asked why generic injectables seem to be having more frequent quality control issues, Dr. Kweder said that the requirements and rules for good manufacturing have not changed over the years, but that the explosion of generic products in the last decade has perhaps put more pressure on manufacturers.
Dr. Schilsky and Dr. Penley said they were hopeful that legislation creating user fees for generic drugmakers would help address the shortage issue. Bills to reauthorize all FDA user fee programs have passed both the House and the Senate; differences are being hashed out in conference right now. The generic user fee – expected to add $1.5 billion to the FDA budget over the next few years – is expected to be retained. Experts expect the president to sign the final bill, said Dr. Schilsky.
The additional funds would give the FDA the ability to cut generic drug review times from the current 24-30 months to about 6-10 months, Dr. Kweder said.
It also would give the agency more resources to eradicate a current backlog of reviews and move more quickly on manufacturing issues, she said.
The legislation includes several additional items on ASCO’s wish list, including a requirement that manufacturers give the FDA early warning of impending shortages. But it does not include any penalties for noncompliance, said Dr. Schilsky.
The Food and Drug Administration has prevented at least 50 drug shortages so far this year, mainly as a result of manufacturers giving the agency early warning of impending problems, Dr. Sandra Kweder announced June 4.
Shortages persist, however, especially for key oncology therapies, according to Dr. Kweder, deputy director of the FDA Office of New Drugs, and oncologists who participated in a press briefing at the annual meeting of the American Society of Clinical Oncologists (ASCO) in Chicago.
Since the White House issued an executive order last Oct. 31 encouraging manufacturers to give the FDA "early notification" of potential disruptions, the agency, working with physicians, manufacturers, pharmacists, and others, has been able to avoid more than 150 drug shortages, Dr. Kweder said.
"Early notification by manufacturers of when they are having production difficulties makes a huge difference in our ability to prevent the public from bearing the burden of drug shortages," she said.
"The good news is that the frequency of drug shortages has begun to decline," agreed Dr. Richard Schilsky, chair of the ASCO Government Relations Committee and chief of the hematology/oncology section at the University of Chicago.
However, unpredictability still exists, which means that oncologists are "never sure when a generic is going to go out of supply," said Dr. Schilsky. "That creates a tremendous amount of uncertainty and anxiety for our patients."
Dr. Michael P. Link, ASCO president, said that several commonly used chemotherapy agents remain in short supply, including fluorouracil, nitrogen mustard, and paclitaxel. In addition, although a methotrexate shortage has been addressed, there is now a shortage of sodium bicarbonate, which is needed to give high-dose injections of the drug, said Dr. Link, chief of pediatric hematology/oncology at the Lucile Packard Children’s Hospital at Stanford (Calif.) University.
Shortages often lead to delays in treatment or less-than-desirable substitutions, said Dr. W. Charles Penley, the incoming ASCO Government Relations Committee chair. His practice – Tennessee Oncology in Nashville – is large enough that it generally has been able to locate products in short supply through its purchasing groups. But a leucovorin shortage last summer led him to switch some patients to oral capecitabine (Xeloda).
That drug is more expensive and is not tolerated well by some patients, he said.
And even that product almost went into short supply, Dr. Kweder said. There were some quality control issues that led the agency to temporarily import capecitabine while the manufacturer, Roche, fixed the problem, she said.
Manufacturing and quality control – especially for generic sterile injectables – continue to be behind many shortages, Dr. Kweder said. Sterility has been a consistent problem, with many products found to contain glass particles or metal shavings. She described a zero tolerance policy for such issues with injectables. "No patient should ever be exposed to risks of those sorts," she said.
When asked why generic injectables seem to be having more frequent quality control issues, Dr. Kweder said that the requirements and rules for good manufacturing have not changed over the years, but that the explosion of generic products in the last decade has perhaps put more pressure on manufacturers.
Dr. Schilsky and Dr. Penley said they were hopeful that legislation creating user fees for generic drugmakers would help address the shortage issue. Bills to reauthorize all FDA user fee programs have passed both the House and the Senate; differences are being hashed out in conference right now. The generic user fee – expected to add $1.5 billion to the FDA budget over the next few years – is expected to be retained. Experts expect the president to sign the final bill, said Dr. Schilsky.
The additional funds would give the FDA the ability to cut generic drug review times from the current 24-30 months to about 6-10 months, Dr. Kweder said.
It also would give the agency more resources to eradicate a current backlog of reviews and move more quickly on manufacturing issues, she said.
The legislation includes several additional items on ASCO’s wish list, including a requirement that manufacturers give the FDA early warning of impending shortages. But it does not include any penalties for noncompliance, said Dr. Schilsky.
FROM THE ANNUAL MEETING OF THE AMERICAN SOCIETY OF CLINICAL ONCOLOGY
Consumer Reports Issues Ratings on 487 Physician Practices
Consumers in Massachusetts are about to find out about what their fellow residents think about the quality of primary care and pediatric care in the state.
Consumer Reports, in collaboration with Massachusetts Health Quality Partners, released ratings on 329 adult primary care practices and 158 pediatric practices based on survey responses from 47,565 adults and 16,530 parents about their experiences in receiving care at those practices.
Massachusetts – which enacted a health reform law in 2006 requiring residents to buy insurance – is considered a model for the federal Affordable Care Act.
Interestingly, although demand for care expanded, survey responses showed that, for the most part, residents did not have trouble getting to see a physician. A total of 38% said that they did not get an appointment that they needed right away.
The ratings are included in a special 23-page insert in the newest Consumer Reports magazine and will also be available free of charge on the Web sites of both MHQP and Consumer Reports.
MHQP designed the survey, and its questions reflect the Institute of Medicine’s evidence-based recommendations on what makes good primary care, Barbra Rabson, executive director of MHQP, said in an interview. The survey has been used by the nonprofit group every 2 years since 2006 to measure patient experiences and feed them back to physicians in an effort to improve the quality of care in Massachusetts, Ms. Rabson said.
The MHQP board decided a year ago to partner with Consumer Reports as a way to make the data more widely available, she explained, adding that, although there were always plenty of hits on the MHQP Web site every time it released new survey data, interest would fade with time and many consumers weren’t even aware that MHQP had such data.
MHQP aims to reach as many consumers as possible and get them to be more interested in the quality of their care. "We’re really trying to get the public to be, certainly more engaged, but we also want them to increase their expectations about how good their health care could be," she said, adding that the survey data can help build those expectations.
For physicians, it is crucial for them to hear the patient, said Ms. Rabson. "If you don’t listen to the patient’s voice, how can you possibly have a patient-centered health care system?"
MHQP – established in 1995 – is a coalition that includes physicians, hospitals, health plans, purchasers, patient and public representatives, academics, and government agencies. When the group began discussing the Consumer Reports collaboration, some physicians were in favor. They felt that the MHQP survey was validated and evidence based, which meant that good data would be widely distributed. Others were hesitant because they did not want to be considered a product like a toaster or an automobile – the traditional purview of Consumer Reports’ reviews, she said.
The data should not be a surprise to physicians, since they already received the survey results last fall, said Ms. Rabson.
In the ratings, physicians are given a score from 1 to 4 (with 4 being the highest) in each of five performance measures: how well doctors communicate with patients; how well they coordinate care; how well they know their patients; whether patients can get timely appointments, care, and information; and whether patients get courteous and respectful help from office staff. Patients also were asked whether they would recommend the practice to a friend or family member.
A total of 13 of the 329 adult practices and 1 of the 158 pediatric practices had the highest score in each of the five measures. Only practices with three or more physicians were included. The data were not broken down further into how many practices were top, middle, or bottom tier. High-scoring practices were found across the state – in rural, suburban, and urban areas – and in a variety of settings, such as in academic and private practices.
All of the survey respondents had health insurance. Individual physicians were not rated.
Only a fraction of the survey questions that were asked were included in the Consumer Reports special insert. The rest of the data are available on the MHQP Web site.
In the excerpts published in the insert, a majority of patients – more than 80% – said that they felt their physician always explained things in a way that was easy to understand, listened carefully to them, and spent enough time with them. This was not always the case, said Ms. Rabson. Low marks in previous surveys have spurred physicians in the state to work hard at improving the quality of their communications with patients, she said.
Doctors received lower marks for coordinating care with specialists, for their knowledge of the patient as a person, for including the patient’s values and beliefs, and for asking about stress and depression.
Only 39% of patients surveyed said that their doctor always asked them whether they had periods when they felt sad, empty, or depressed.
MHQP would like to repeat the Consumer Reports collaboration when it surveys patients again in 2013, but it must line up funding. The partnership was funded by a grant from the Robert Wood Johnson Foundation.
RWJF also is supporting Consumer Reports’ collaborations with two other quality improvement organizations: Minnesota Community Measurement and the Wisconsin Collaborative for Healthcare Quality. Minnesota will release clinical quality and cost data to the public in September and Wisconsin will issue ratings on preventive care measures later this year.
Consumers in Massachusetts are about to find out about what their fellow residents think about the quality of primary care and pediatric care in the state.
Consumer Reports, in collaboration with Massachusetts Health Quality Partners, released ratings on 329 adult primary care practices and 158 pediatric practices based on survey responses from 47,565 adults and 16,530 parents about their experiences in receiving care at those practices.
Massachusetts – which enacted a health reform law in 2006 requiring residents to buy insurance – is considered a model for the federal Affordable Care Act.
Interestingly, although demand for care expanded, survey responses showed that, for the most part, residents did not have trouble getting to see a physician. A total of 38% said that they did not get an appointment that they needed right away.
The ratings are included in a special 23-page insert in the newest Consumer Reports magazine and will also be available free of charge on the Web sites of both MHQP and Consumer Reports.
MHQP designed the survey, and its questions reflect the Institute of Medicine’s evidence-based recommendations on what makes good primary care, Barbra Rabson, executive director of MHQP, said in an interview. The survey has been used by the nonprofit group every 2 years since 2006 to measure patient experiences and feed them back to physicians in an effort to improve the quality of care in Massachusetts, Ms. Rabson said.
The MHQP board decided a year ago to partner with Consumer Reports as a way to make the data more widely available, she explained, adding that, although there were always plenty of hits on the MHQP Web site every time it released new survey data, interest would fade with time and many consumers weren’t even aware that MHQP had such data.
MHQP aims to reach as many consumers as possible and get them to be more interested in the quality of their care. "We’re really trying to get the public to be, certainly more engaged, but we also want them to increase their expectations about how good their health care could be," she said, adding that the survey data can help build those expectations.
For physicians, it is crucial for them to hear the patient, said Ms. Rabson. "If you don’t listen to the patient’s voice, how can you possibly have a patient-centered health care system?"
MHQP – established in 1995 – is a coalition that includes physicians, hospitals, health plans, purchasers, patient and public representatives, academics, and government agencies. When the group began discussing the Consumer Reports collaboration, some physicians were in favor. They felt that the MHQP survey was validated and evidence based, which meant that good data would be widely distributed. Others were hesitant because they did not want to be considered a product like a toaster or an automobile – the traditional purview of Consumer Reports’ reviews, she said.
The data should not be a surprise to physicians, since they already received the survey results last fall, said Ms. Rabson.
In the ratings, physicians are given a score from 1 to 4 (with 4 being the highest) in each of five performance measures: how well doctors communicate with patients; how well they coordinate care; how well they know their patients; whether patients can get timely appointments, care, and information; and whether patients get courteous and respectful help from office staff. Patients also were asked whether they would recommend the practice to a friend or family member.
A total of 13 of the 329 adult practices and 1 of the 158 pediatric practices had the highest score in each of the five measures. Only practices with three or more physicians were included. The data were not broken down further into how many practices were top, middle, or bottom tier. High-scoring practices were found across the state – in rural, suburban, and urban areas – and in a variety of settings, such as in academic and private practices.
All of the survey respondents had health insurance. Individual physicians were not rated.
Only a fraction of the survey questions that were asked were included in the Consumer Reports special insert. The rest of the data are available on the MHQP Web site.
In the excerpts published in the insert, a majority of patients – more than 80% – said that they felt their physician always explained things in a way that was easy to understand, listened carefully to them, and spent enough time with them. This was not always the case, said Ms. Rabson. Low marks in previous surveys have spurred physicians in the state to work hard at improving the quality of their communications with patients, she said.
Doctors received lower marks for coordinating care with specialists, for their knowledge of the patient as a person, for including the patient’s values and beliefs, and for asking about stress and depression.
Only 39% of patients surveyed said that their doctor always asked them whether they had periods when they felt sad, empty, or depressed.
MHQP would like to repeat the Consumer Reports collaboration when it surveys patients again in 2013, but it must line up funding. The partnership was funded by a grant from the Robert Wood Johnson Foundation.
RWJF also is supporting Consumer Reports’ collaborations with two other quality improvement organizations: Minnesota Community Measurement and the Wisconsin Collaborative for Healthcare Quality. Minnesota will release clinical quality and cost data to the public in September and Wisconsin will issue ratings on preventive care measures later this year.
Consumers in Massachusetts are about to find out about what their fellow residents think about the quality of primary care and pediatric care in the state.
Consumer Reports, in collaboration with Massachusetts Health Quality Partners, released ratings on 329 adult primary care practices and 158 pediatric practices based on survey responses from 47,565 adults and 16,530 parents about their experiences in receiving care at those practices.
Massachusetts – which enacted a health reform law in 2006 requiring residents to buy insurance – is considered a model for the federal Affordable Care Act.
Interestingly, although demand for care expanded, survey responses showed that, for the most part, residents did not have trouble getting to see a physician. A total of 38% said that they did not get an appointment that they needed right away.
The ratings are included in a special 23-page insert in the newest Consumer Reports magazine and will also be available free of charge on the Web sites of both MHQP and Consumer Reports.
MHQP designed the survey, and its questions reflect the Institute of Medicine’s evidence-based recommendations on what makes good primary care, Barbra Rabson, executive director of MHQP, said in an interview. The survey has been used by the nonprofit group every 2 years since 2006 to measure patient experiences and feed them back to physicians in an effort to improve the quality of care in Massachusetts, Ms. Rabson said.
The MHQP board decided a year ago to partner with Consumer Reports as a way to make the data more widely available, she explained, adding that, although there were always plenty of hits on the MHQP Web site every time it released new survey data, interest would fade with time and many consumers weren’t even aware that MHQP had such data.
MHQP aims to reach as many consumers as possible and get them to be more interested in the quality of their care. "We’re really trying to get the public to be, certainly more engaged, but we also want them to increase their expectations about how good their health care could be," she said, adding that the survey data can help build those expectations.
For physicians, it is crucial for them to hear the patient, said Ms. Rabson. "If you don’t listen to the patient’s voice, how can you possibly have a patient-centered health care system?"
MHQP – established in 1995 – is a coalition that includes physicians, hospitals, health plans, purchasers, patient and public representatives, academics, and government agencies. When the group began discussing the Consumer Reports collaboration, some physicians were in favor. They felt that the MHQP survey was validated and evidence based, which meant that good data would be widely distributed. Others were hesitant because they did not want to be considered a product like a toaster or an automobile – the traditional purview of Consumer Reports’ reviews, she said.
The data should not be a surprise to physicians, since they already received the survey results last fall, said Ms. Rabson.
In the ratings, physicians are given a score from 1 to 4 (with 4 being the highest) in each of five performance measures: how well doctors communicate with patients; how well they coordinate care; how well they know their patients; whether patients can get timely appointments, care, and information; and whether patients get courteous and respectful help from office staff. Patients also were asked whether they would recommend the practice to a friend or family member.
A total of 13 of the 329 adult practices and 1 of the 158 pediatric practices had the highest score in each of the five measures. Only practices with three or more physicians were included. The data were not broken down further into how many practices were top, middle, or bottom tier. High-scoring practices were found across the state – in rural, suburban, and urban areas – and in a variety of settings, such as in academic and private practices.
All of the survey respondents had health insurance. Individual physicians were not rated.
Only a fraction of the survey questions that were asked were included in the Consumer Reports special insert. The rest of the data are available on the MHQP Web site.
In the excerpts published in the insert, a majority of patients – more than 80% – said that they felt their physician always explained things in a way that was easy to understand, listened carefully to them, and spent enough time with them. This was not always the case, said Ms. Rabson. Low marks in previous surveys have spurred physicians in the state to work hard at improving the quality of their communications with patients, she said.
Doctors received lower marks for coordinating care with specialists, for their knowledge of the patient as a person, for including the patient’s values and beliefs, and for asking about stress and depression.
Only 39% of patients surveyed said that their doctor always asked them whether they had periods when they felt sad, empty, or depressed.
MHQP would like to repeat the Consumer Reports collaboration when it surveys patients again in 2013, but it must line up funding. The partnership was funded by a grant from the Robert Wood Johnson Foundation.
RWJF also is supporting Consumer Reports’ collaborations with two other quality improvement organizations: Minnesota Community Measurement and the Wisconsin Collaborative for Healthcare Quality. Minnesota will release clinical quality and cost data to the public in September and Wisconsin will issue ratings on preventive care measures later this year.
Physician-Led PACs Rise in Number, Influence
They may have come relatively late to the party, but physician-led political action committees have become true players on the political stage and are poised to exert their influence in this year’s elections.
Their efforts are beginning to pay off, PAC leaders say. Members of Congress are "much more forthcoming when you’ve built relationships in support of their campaign," said Dr. James N. Martin Jr., chair of Ob-GynPAC, representing the American Congress of Obstetricians and Gynecologists (ACOG).
"Nothing has put the Academy at the table like BrainPAC has," Michael J. Amery, legislative counsel to the American Academy of Neurology, said in an interview. "We literally did not exist in the eyes of Congress before the PAC was created."
As of early March, PACs in the overall health sector had donated some $72 million to federal candidates – those running for the White House or for seats in the House or Senate – in the 2011-2012 election cycle. That level of spending ranked it as the fifth-leading source of money by sector, according to OpenSecrets.org, an election-information database compiled by the Center for Responsive Politics from Federal Elections Commission records.
OpenSecrets defines the health sector as pharmaceutical companies, medical device makers, hospitals, insurers, nursing homes, pharmacy benefit management companies, and professional societies, among others.
So far in the 2011-2012 cycle, physician-led organizations account for 5 of the top 11 donors in the health sector: the California-based Cooperative of American Physicians, the American Association of Orthopaedic Surgeons, the American College of Radiology, the American Society of Anesthesiologists, and the American College of Emergency Physicians.
PACs that represent health professionals tend to give evenly to Democrats and Republicans. A particular candidate’s position on an issue generally holds more weight than does his or her party affiliation, experts told this news organization.
Although the first PAC was formed in 1944, doctors’ PACs are a much more recent phenomenon. "Physicians historically have not been very active in the political process," Dr. Howard T. "Bo" Walpole, chair of the American College of Cardiology’s ACC PAC, said in an interview. "It’s been a learning experience for most of us."
Most physician PACs were formed in the last 5-10 years. Generally, PACs are formed by unions, corporations, or professional organizations to pool money and donate it to candidates who support their agendas. PACs can give up to $5,000 to a candidate or $15,000 to a national party committee per election cycle. Donations to PACs are limited to $5,000.
AMPAC – representing the American Medical Association – was seen as the voice of American medicine for decades. But as other groups have become more politically active, specialty-specific PACs have grown, and AMPAC, at least in the current cycle, seems to be lagging.
In 2008, AMPAC contributed $1.5 million to candidates. Two years later, that donation dropped to $1.3 million, and just edged out NEMPAC (the National Emergency Medicine PAC sponsored by the American College of Emergency Physicians) for the top spot among health professionals’ PACs. And this year, AMPAC fund-raising has slipped. At about $173,000 for the AMA and its affiliates, it is being dwarfed by a number of other physician PACs. The AMA did not respond to requests for interviews.
As recently as 2000, the American College of Physicians and the American Academy of Family Physicians were both debating whether to establish a PAC. Now, primary care is holding its own. The ACP says it represents 116,000 members; the AAFP, 100,300 members. In the 2012 cycle, FamMedPAC has donated $221,500 (making it the 14th biggest health professionals PAC), while ACP Services PAC has donated $95,000 (No. 31), according to OpenSecrets.
At the 9th position among health professionals PACs for this election cycle, ACC PAC (donating $310,000) has trumpeted its PAC success on its blog. ACC PAC began in 2002, and has been a leading contributor over the last few election cycles. With $1 million raised so far, it is among the 10 largest health sector PACs, according to Dr. Walpole.
The AAN, with 25,000 members, also has quickly grown BrainPAC, established in 2007. The PAC has raised more than $500,000 in that time, most of it coming over the last two election cycles. And that’s with only 5%-6% of the membership participating, said Mr. Amery.
Dr. Martin of Ob-GynPAC noted that the PAC contributions help physicians gain access. "Those legislators will hear you out and be open to hearing what you want," he said. The PAC is pushing for liability reform, protection of Medicaid funding, and any legislation that helps benefit women’s health, he said.
Ob-GynPAC is the 12th largest donor in the current cycle, having contributed $272,500. ACOG has 55,000 members.
In addition to working in the federal sector, Ob-GynPAC is getting involved in the states, Dr. Martin said. This year, the PAC has set aside some $50,000 of its funds to support state candidates.
Five years ago, the American College of Rheumatology started RheumPAC alongside efforts to increase its advocacy efforts, according to RheumPAC chair Dr. Gary Bryant.
A lot of the PAC’s work has been "getting the word out that we exist and the importance of participating in the political process," said Dr. Bryant, associate professor at the University of Minnesota. He added that he views as a recent success the introduction of the Patients Access to Treatments Act of 2012 (H.R. 4209), sponsored by Rep. David McKinley (R-W.Va.), which would limit patients’ cost-sharing for certain medications.
Even though it was founded in 2001, the American Association of Clinical Endocrinologists’ AACEPAC also is trying to raise its 6,500 members’ interest in participating, said Dr. Jonathan Leffert, chair of AACEPAC. Some of the diseases that endocrinologists treat, specifically diabetes and osteoporosis, have a huge impact on Medicare spending, "and we want to lend our voice to that," he said. The PAC has donated $20,000 to $30,000 to candidates in each of the last two cycles, and about half that so far in the 2012 cycle.
"Despite our small size and our small PAC dollar amount, because of our persistence and our focus, I think we’ve been extremely successful in making ourselves a force on Capitol Hill," Dr. Leffert said, noting at least a temporary victory in increasing payments for bone densitometry measurement in physician offices and securing introduction of the Diabetes Clinical Care Commission Act, which would create a board to enhance diabetes care.
Some physician groups, such as the American Academy of Pediatrics and the American Society of Clinical Oncology, have decided not to establish PACs. Dr. Marion Burton, immediate past president of the AAP, said that a PAC is not necessary for pediatricians. He says unlike many other specialties, pediatricians command attention just by nature of their patients. The AAP "has a huge respect in Congress because most of the time we come with our hands and hearts held out for children," said Dr. Burton.
They may have come relatively late to the party, but physician-led political action committees have become true players on the political stage and are poised to exert their influence in this year’s elections.
Their efforts are beginning to pay off, PAC leaders say. Members of Congress are "much more forthcoming when you’ve built relationships in support of their campaign," said Dr. James N. Martin Jr., chair of Ob-GynPAC, representing the American Congress of Obstetricians and Gynecologists (ACOG).
"Nothing has put the Academy at the table like BrainPAC has," Michael J. Amery, legislative counsel to the American Academy of Neurology, said in an interview. "We literally did not exist in the eyes of Congress before the PAC was created."
As of early March, PACs in the overall health sector had donated some $72 million to federal candidates – those running for the White House or for seats in the House or Senate – in the 2011-2012 election cycle. That level of spending ranked it as the fifth-leading source of money by sector, according to OpenSecrets.org, an election-information database compiled by the Center for Responsive Politics from Federal Elections Commission records.
OpenSecrets defines the health sector as pharmaceutical companies, medical device makers, hospitals, insurers, nursing homes, pharmacy benefit management companies, and professional societies, among others.
So far in the 2011-2012 cycle, physician-led organizations account for 5 of the top 11 donors in the health sector: the California-based Cooperative of American Physicians, the American Association of Orthopaedic Surgeons, the American College of Radiology, the American Society of Anesthesiologists, and the American College of Emergency Physicians.
PACs that represent health professionals tend to give evenly to Democrats and Republicans. A particular candidate’s position on an issue generally holds more weight than does his or her party affiliation, experts told this news organization.
Although the first PAC was formed in 1944, doctors’ PACs are a much more recent phenomenon. "Physicians historically have not been very active in the political process," Dr. Howard T. "Bo" Walpole, chair of the American College of Cardiology’s ACC PAC, said in an interview. "It’s been a learning experience for most of us."
Most physician PACs were formed in the last 5-10 years. Generally, PACs are formed by unions, corporations, or professional organizations to pool money and donate it to candidates who support their agendas. PACs can give up to $5,000 to a candidate or $15,000 to a national party committee per election cycle. Donations to PACs are limited to $5,000.
AMPAC – representing the American Medical Association – was seen as the voice of American medicine for decades. But as other groups have become more politically active, specialty-specific PACs have grown, and AMPAC, at least in the current cycle, seems to be lagging.
In 2008, AMPAC contributed $1.5 million to candidates. Two years later, that donation dropped to $1.3 million, and just edged out NEMPAC (the National Emergency Medicine PAC sponsored by the American College of Emergency Physicians) for the top spot among health professionals’ PACs. And this year, AMPAC fund-raising has slipped. At about $173,000 for the AMA and its affiliates, it is being dwarfed by a number of other physician PACs. The AMA did not respond to requests for interviews.
As recently as 2000, the American College of Physicians and the American Academy of Family Physicians were both debating whether to establish a PAC. Now, primary care is holding its own. The ACP says it represents 116,000 members; the AAFP, 100,300 members. In the 2012 cycle, FamMedPAC has donated $221,500 (making it the 14th biggest health professionals PAC), while ACP Services PAC has donated $95,000 (No. 31), according to OpenSecrets.
At the 9th position among health professionals PACs for this election cycle, ACC PAC (donating $310,000) has trumpeted its PAC success on its blog. ACC PAC began in 2002, and has been a leading contributor over the last few election cycles. With $1 million raised so far, it is among the 10 largest health sector PACs, according to Dr. Walpole.
The AAN, with 25,000 members, also has quickly grown BrainPAC, established in 2007. The PAC has raised more than $500,000 in that time, most of it coming over the last two election cycles. And that’s with only 5%-6% of the membership participating, said Mr. Amery.
Dr. Martin of Ob-GynPAC noted that the PAC contributions help physicians gain access. "Those legislators will hear you out and be open to hearing what you want," he said. The PAC is pushing for liability reform, protection of Medicaid funding, and any legislation that helps benefit women’s health, he said.
Ob-GynPAC is the 12th largest donor in the current cycle, having contributed $272,500. ACOG has 55,000 members.
In addition to working in the federal sector, Ob-GynPAC is getting involved in the states, Dr. Martin said. This year, the PAC has set aside some $50,000 of its funds to support state candidates.
Five years ago, the American College of Rheumatology started RheumPAC alongside efforts to increase its advocacy efforts, according to RheumPAC chair Dr. Gary Bryant.
A lot of the PAC’s work has been "getting the word out that we exist and the importance of participating in the political process," said Dr. Bryant, associate professor at the University of Minnesota. He added that he views as a recent success the introduction of the Patients Access to Treatments Act of 2012 (H.R. 4209), sponsored by Rep. David McKinley (R-W.Va.), which would limit patients’ cost-sharing for certain medications.
Even though it was founded in 2001, the American Association of Clinical Endocrinologists’ AACEPAC also is trying to raise its 6,500 members’ interest in participating, said Dr. Jonathan Leffert, chair of AACEPAC. Some of the diseases that endocrinologists treat, specifically diabetes and osteoporosis, have a huge impact on Medicare spending, "and we want to lend our voice to that," he said. The PAC has donated $20,000 to $30,000 to candidates in each of the last two cycles, and about half that so far in the 2012 cycle.
"Despite our small size and our small PAC dollar amount, because of our persistence and our focus, I think we’ve been extremely successful in making ourselves a force on Capitol Hill," Dr. Leffert said, noting at least a temporary victory in increasing payments for bone densitometry measurement in physician offices and securing introduction of the Diabetes Clinical Care Commission Act, which would create a board to enhance diabetes care.
Some physician groups, such as the American Academy of Pediatrics and the American Society of Clinical Oncology, have decided not to establish PACs. Dr. Marion Burton, immediate past president of the AAP, said that a PAC is not necessary for pediatricians. He says unlike many other specialties, pediatricians command attention just by nature of their patients. The AAP "has a huge respect in Congress because most of the time we come with our hands and hearts held out for children," said Dr. Burton.
They may have come relatively late to the party, but physician-led political action committees have become true players on the political stage and are poised to exert their influence in this year’s elections.
Their efforts are beginning to pay off, PAC leaders say. Members of Congress are "much more forthcoming when you’ve built relationships in support of their campaign," said Dr. James N. Martin Jr., chair of Ob-GynPAC, representing the American Congress of Obstetricians and Gynecologists (ACOG).
"Nothing has put the Academy at the table like BrainPAC has," Michael J. Amery, legislative counsel to the American Academy of Neurology, said in an interview. "We literally did not exist in the eyes of Congress before the PAC was created."
As of early March, PACs in the overall health sector had donated some $72 million to federal candidates – those running for the White House or for seats in the House or Senate – in the 2011-2012 election cycle. That level of spending ranked it as the fifth-leading source of money by sector, according to OpenSecrets.org, an election-information database compiled by the Center for Responsive Politics from Federal Elections Commission records.
OpenSecrets defines the health sector as pharmaceutical companies, medical device makers, hospitals, insurers, nursing homes, pharmacy benefit management companies, and professional societies, among others.
So far in the 2011-2012 cycle, physician-led organizations account for 5 of the top 11 donors in the health sector: the California-based Cooperative of American Physicians, the American Association of Orthopaedic Surgeons, the American College of Radiology, the American Society of Anesthesiologists, and the American College of Emergency Physicians.
PACs that represent health professionals tend to give evenly to Democrats and Republicans. A particular candidate’s position on an issue generally holds more weight than does his or her party affiliation, experts told this news organization.
Although the first PAC was formed in 1944, doctors’ PACs are a much more recent phenomenon. "Physicians historically have not been very active in the political process," Dr. Howard T. "Bo" Walpole, chair of the American College of Cardiology’s ACC PAC, said in an interview. "It’s been a learning experience for most of us."
Most physician PACs were formed in the last 5-10 years. Generally, PACs are formed by unions, corporations, or professional organizations to pool money and donate it to candidates who support their agendas. PACs can give up to $5,000 to a candidate or $15,000 to a national party committee per election cycle. Donations to PACs are limited to $5,000.
AMPAC – representing the American Medical Association – was seen as the voice of American medicine for decades. But as other groups have become more politically active, specialty-specific PACs have grown, and AMPAC, at least in the current cycle, seems to be lagging.
In 2008, AMPAC contributed $1.5 million to candidates. Two years later, that donation dropped to $1.3 million, and just edged out NEMPAC (the National Emergency Medicine PAC sponsored by the American College of Emergency Physicians) for the top spot among health professionals’ PACs. And this year, AMPAC fund-raising has slipped. At about $173,000 for the AMA and its affiliates, it is being dwarfed by a number of other physician PACs. The AMA did not respond to requests for interviews.
As recently as 2000, the American College of Physicians and the American Academy of Family Physicians were both debating whether to establish a PAC. Now, primary care is holding its own. The ACP says it represents 116,000 members; the AAFP, 100,300 members. In the 2012 cycle, FamMedPAC has donated $221,500 (making it the 14th biggest health professionals PAC), while ACP Services PAC has donated $95,000 (No. 31), according to OpenSecrets.
At the 9th position among health professionals PACs for this election cycle, ACC PAC (donating $310,000) has trumpeted its PAC success on its blog. ACC PAC began in 2002, and has been a leading contributor over the last few election cycles. With $1 million raised so far, it is among the 10 largest health sector PACs, according to Dr. Walpole.
The AAN, with 25,000 members, also has quickly grown BrainPAC, established in 2007. The PAC has raised more than $500,000 in that time, most of it coming over the last two election cycles. And that’s with only 5%-6% of the membership participating, said Mr. Amery.
Dr. Martin of Ob-GynPAC noted that the PAC contributions help physicians gain access. "Those legislators will hear you out and be open to hearing what you want," he said. The PAC is pushing for liability reform, protection of Medicaid funding, and any legislation that helps benefit women’s health, he said.
Ob-GynPAC is the 12th largest donor in the current cycle, having contributed $272,500. ACOG has 55,000 members.
In addition to working in the federal sector, Ob-GynPAC is getting involved in the states, Dr. Martin said. This year, the PAC has set aside some $50,000 of its funds to support state candidates.
Five years ago, the American College of Rheumatology started RheumPAC alongside efforts to increase its advocacy efforts, according to RheumPAC chair Dr. Gary Bryant.
A lot of the PAC’s work has been "getting the word out that we exist and the importance of participating in the political process," said Dr. Bryant, associate professor at the University of Minnesota. He added that he views as a recent success the introduction of the Patients Access to Treatments Act of 2012 (H.R. 4209), sponsored by Rep. David McKinley (R-W.Va.), which would limit patients’ cost-sharing for certain medications.
Even though it was founded in 2001, the American Association of Clinical Endocrinologists’ AACEPAC also is trying to raise its 6,500 members’ interest in participating, said Dr. Jonathan Leffert, chair of AACEPAC. Some of the diseases that endocrinologists treat, specifically diabetes and osteoporosis, have a huge impact on Medicare spending, "and we want to lend our voice to that," he said. The PAC has donated $20,000 to $30,000 to candidates in each of the last two cycles, and about half that so far in the 2012 cycle.
"Despite our small size and our small PAC dollar amount, because of our persistence and our focus, I think we’ve been extremely successful in making ourselves a force on Capitol Hill," Dr. Leffert said, noting at least a temporary victory in increasing payments for bone densitometry measurement in physician offices and securing introduction of the Diabetes Clinical Care Commission Act, which would create a board to enhance diabetes care.
Some physician groups, such as the American Academy of Pediatrics and the American Society of Clinical Oncology, have decided not to establish PACs. Dr. Marion Burton, immediate past president of the AAP, said that a PAC is not necessary for pediatricians. He says unlike many other specialties, pediatricians command attention just by nature of their patients. The AAP "has a huge respect in Congress because most of the time we come with our hands and hearts held out for children," said Dr. Burton.