User login
Transplantation palliative care: The time is ripe
Over 10 years ago, a challenge was made in a surgical publication for increased collaboration between the fields of transplantation and palliative care.1
Since that time not much progress has been made bringing these fields together in a consistent way that would mutually benefit patients and the specialties. However, other progress has been made, particularly in the field of palliative care, which could brighten the prospects and broaden the opportunities to accomplish collaboration between palliative care and transplantation.
Growth of palliative services
During the past decade there has been a robust proliferation of hospital-based palliative care programs in the United States. In all, 67% of U.S. hospitals with 50 or more beds report palliative care teams, up from 63% in 2011 and 53% in 2008.
Only a decade ago, critical care and palliative care were generally considered mutually exclusive. Evidence is trickling in to suggest that this is no longer the case. Although palliative care was not an integral part of critical care at that time, patients, families, and even practitioners began to demand these services. Cook and Rocker have eloquently advocated the rightful place of palliative care in the ICU.2
Studies in recent years have shown that the integration of palliative care into critical care decreases in length of ICU and hospital stay, decreases costs, enhances patient/family satisfaction, and promotes a more rapid consensus about goals of care, without increasing mortality. The ICU experience to date could be considered a reassuring precedent for transplantation palliative care.
Integration of palliative care with transplantation
Early palliative care intervention has been shown to improve symptom burden and depression scores in end-stage liver disease patients awaiting transplant. In addition, early palliative care consultation in conjunction with cancer treatment has been associated with increased survival in non–small-cell lung cancer patients. It has been demonstrated that early integration of palliative care in the surgical ICU alongside disease-directed curative care can be accomplished without change in mortality, while improving end-of-life practice in liver transplant patients.3
What palliative care can do for transplant patients
What does palliative care mean for the person (and family) awaiting transplantation? For the cirrhotic patient with cachexia, ascites, and encephalopathy, it means access to the services of a team trained in the management of these symptoms. Palliative care teams can also provide psychosocial and spiritual support for patients and families who are intimidated by the complex navigation of the health care system and the existential threat that end-stage organ failure presents to them. Skilled palliative care and services can be the difference between failing and extended life with a higher quality of life for these very sick patients
Resuscitation of a patient, whether through restoration of organ function or interdicting the progression of disease, begins with resuscitation of hope. Nothing achieves this more quickly than amelioration of burdensome symptoms for the patient and family.
The barriers for transplant surgeons and teams referring and incorporating palliative care services in their practices are multiple and profound. The unique dilemma facing the transplant team is to balance the treatment of the failing organ, the treatment of the patient (and family and friends), and the best use of the graft, a precious gift of society.
Palliative surgery has been defined as any invasive procedure in which the main intention is to mitigate physical symptoms in patients with noncurable disease without causing premature death. The very success of transplantation over the past 3 decades has obscured our memory of transplantation as a type of palliative surgery. It is a well-known axiom of reconstructive surgery that the reconstructed site should be compared to what was there, not to “normal.” Even in the current era of improved immunosuppression and posttransplant support services, one could hardly describe even a successful transplant patient’s experience as “normal.” These patients’ lives may be extended and/or enhanced but they need palliative care before, during, and after transplantation. The growing availability of trained palliative care clinicians and teams, the increased familiarity of palliative and end-of-life care to surgical residents and fellows, and quality metrics measuring palliative care outcomes will provide reassurance and guidance to address reservations about the convergence of the two seemingly opposite realities.
A modest proposal
We propose that palliative care be presented to the entire spectrum of transplantation care: on the ward, in the ICU, and after transplantation. More specific “triggers” for palliative care for referral of transplant patients should be identified. Wentlandt et al.4 have described a promising model for an ambulatory clinic, which provides early, integrated palliative care to patients awaiting and receiving organ transplantation. In addition, we propose an application for grant funding for a conference and eventual formation of a work group of transplant surgeons and team members, palliative care clinicians, and patient/families who have experienced one of the aspects of the transplant spectrum. We await the subspecialty certification in hospice and palliative medicine of a transplant surgeon. Outside of transplantation, every other surgical specialty in the United States has diplomates certified in hospice and palliative medicine. We await the benefits that will accrue from research about the merging of these fields.
1. Molmenti EP, Dunn GP: Transplantation and palliative care: The convergence of two seemingly opposite realities. Surg Clin North Am. 2005;85:373-82.
2. Cook D, Rocker G. Dying with dignity in the intensive care unit. N Engl J Med. 2014;370:2506-14.
3. Lamba S, Murphy P, McVicker S, Smith JH, and Mosenthal AC. Changing end-of-life care practice for liver transplant patients: structured palliative care intervention in the surgical intensive care unit. J Pain Symptom Manage. 2012; 44(4):508-19.
4. Wentlandt, K., Dall’Osto, A., Freeman, N., Le, L. W., Kaya, E., Ross, H., Singer, L. G., Abbey, S., Clarke, H. and Zimmermann, C. (2016), The Transplant Palliative Care Clinic: An early palliative care model for patients in a transplant program. Clin Transplant. 2016 Nov 4; doi: 10.1111/ctr.12838.
Dr. Azoulay is a transplantation specialist of Assistance Publique – Hôpitaux de Paris, and the University of Paris. Dr. Dunn is medical director of the Palliative Care Consultation Service at the University of Pittsburgh Medical Center Hamot, and vice-chair of the ACS Committee on Surgical Palliative Care.
Over 10 years ago, a challenge was made in a surgical publication for increased collaboration between the fields of transplantation and palliative care.1
Since that time not much progress has been made bringing these fields together in a consistent way that would mutually benefit patients and the specialties. However, other progress has been made, particularly in the field of palliative care, which could brighten the prospects and broaden the opportunities to accomplish collaboration between palliative care and transplantation.
Growth of palliative services
During the past decade there has been a robust proliferation of hospital-based palliative care programs in the United States. In all, 67% of U.S. hospitals with 50 or more beds report palliative care teams, up from 63% in 2011 and 53% in 2008.
Only a decade ago, critical care and palliative care were generally considered mutually exclusive. Evidence is trickling in to suggest that this is no longer the case. Although palliative care was not an integral part of critical care at that time, patients, families, and even practitioners began to demand these services. Cook and Rocker have eloquently advocated the rightful place of palliative care in the ICU.2
Studies in recent years have shown that the integration of palliative care into critical care decreases in length of ICU and hospital stay, decreases costs, enhances patient/family satisfaction, and promotes a more rapid consensus about goals of care, without increasing mortality. The ICU experience to date could be considered a reassuring precedent for transplantation palliative care.
Integration of palliative care with transplantation
Early palliative care intervention has been shown to improve symptom burden and depression scores in end-stage liver disease patients awaiting transplant. In addition, early palliative care consultation in conjunction with cancer treatment has been associated with increased survival in non–small-cell lung cancer patients. It has been demonstrated that early integration of palliative care in the surgical ICU alongside disease-directed curative care can be accomplished without change in mortality, while improving end-of-life practice in liver transplant patients.3
What palliative care can do for transplant patients
What does palliative care mean for the person (and family) awaiting transplantation? For the cirrhotic patient with cachexia, ascites, and encephalopathy, it means access to the services of a team trained in the management of these symptoms. Palliative care teams can also provide psychosocial and spiritual support for patients and families who are intimidated by the complex navigation of the health care system and the existential threat that end-stage organ failure presents to them. Skilled palliative care and services can be the difference between failing and extended life with a higher quality of life for these very sick patients
Resuscitation of a patient, whether through restoration of organ function or interdicting the progression of disease, begins with resuscitation of hope. Nothing achieves this more quickly than amelioration of burdensome symptoms for the patient and family.
The barriers for transplant surgeons and teams referring and incorporating palliative care services in their practices are multiple and profound. The unique dilemma facing the transplant team is to balance the treatment of the failing organ, the treatment of the patient (and family and friends), and the best use of the graft, a precious gift of society.
Palliative surgery has been defined as any invasive procedure in which the main intention is to mitigate physical symptoms in patients with noncurable disease without causing premature death. The very success of transplantation over the past 3 decades has obscured our memory of transplantation as a type of palliative surgery. It is a well-known axiom of reconstructive surgery that the reconstructed site should be compared to what was there, not to “normal.” Even in the current era of improved immunosuppression and posttransplant support services, one could hardly describe even a successful transplant patient’s experience as “normal.” These patients’ lives may be extended and/or enhanced but they need palliative care before, during, and after transplantation. The growing availability of trained palliative care clinicians and teams, the increased familiarity of palliative and end-of-life care to surgical residents and fellows, and quality metrics measuring palliative care outcomes will provide reassurance and guidance to address reservations about the convergence of the two seemingly opposite realities.
A modest proposal
We propose that palliative care be presented to the entire spectrum of transplantation care: on the ward, in the ICU, and after transplantation. More specific “triggers” for palliative care for referral of transplant patients should be identified. Wentlandt et al.4 have described a promising model for an ambulatory clinic, which provides early, integrated palliative care to patients awaiting and receiving organ transplantation. In addition, we propose an application for grant funding for a conference and eventual formation of a work group of transplant surgeons and team members, palliative care clinicians, and patient/families who have experienced one of the aspects of the transplant spectrum. We await the subspecialty certification in hospice and palliative medicine of a transplant surgeon. Outside of transplantation, every other surgical specialty in the United States has diplomates certified in hospice and palliative medicine. We await the benefits that will accrue from research about the merging of these fields.
1. Molmenti EP, Dunn GP: Transplantation and palliative care: The convergence of two seemingly opposite realities. Surg Clin North Am. 2005;85:373-82.
2. Cook D, Rocker G. Dying with dignity in the intensive care unit. N Engl J Med. 2014;370:2506-14.
3. Lamba S, Murphy P, McVicker S, Smith JH, and Mosenthal AC. Changing end-of-life care practice for liver transplant patients: structured palliative care intervention in the surgical intensive care unit. J Pain Symptom Manage. 2012; 44(4):508-19.
4. Wentlandt, K., Dall’Osto, A., Freeman, N., Le, L. W., Kaya, E., Ross, H., Singer, L. G., Abbey, S., Clarke, H. and Zimmermann, C. (2016), The Transplant Palliative Care Clinic: An early palliative care model for patients in a transplant program. Clin Transplant. 2016 Nov 4; doi: 10.1111/ctr.12838.
Dr. Azoulay is a transplantation specialist of Assistance Publique – Hôpitaux de Paris, and the University of Paris. Dr. Dunn is medical director of the Palliative Care Consultation Service at the University of Pittsburgh Medical Center Hamot, and vice-chair of the ACS Committee on Surgical Palliative Care.
Over 10 years ago, a challenge was made in a surgical publication for increased collaboration between the fields of transplantation and palliative care.1
Since that time not much progress has been made bringing these fields together in a consistent way that would mutually benefit patients and the specialties. However, other progress has been made, particularly in the field of palliative care, which could brighten the prospects and broaden the opportunities to accomplish collaboration between palliative care and transplantation.
Growth of palliative services
During the past decade there has been a robust proliferation of hospital-based palliative care programs in the United States. In all, 67% of U.S. hospitals with 50 or more beds report palliative care teams, up from 63% in 2011 and 53% in 2008.
Only a decade ago, critical care and palliative care were generally considered mutually exclusive. Evidence is trickling in to suggest that this is no longer the case. Although palliative care was not an integral part of critical care at that time, patients, families, and even practitioners began to demand these services. Cook and Rocker have eloquently advocated the rightful place of palliative care in the ICU.2
Studies in recent years have shown that the integration of palliative care into critical care decreases in length of ICU and hospital stay, decreases costs, enhances patient/family satisfaction, and promotes a more rapid consensus about goals of care, without increasing mortality. The ICU experience to date could be considered a reassuring precedent for transplantation palliative care.
Integration of palliative care with transplantation
Early palliative care intervention has been shown to improve symptom burden and depression scores in end-stage liver disease patients awaiting transplant. In addition, early palliative care consultation in conjunction with cancer treatment has been associated with increased survival in non–small-cell lung cancer patients. It has been demonstrated that early integration of palliative care in the surgical ICU alongside disease-directed curative care can be accomplished without change in mortality, while improving end-of-life practice in liver transplant patients.3
What palliative care can do for transplant patients
What does palliative care mean for the person (and family) awaiting transplantation? For the cirrhotic patient with cachexia, ascites, and encephalopathy, it means access to the services of a team trained in the management of these symptoms. Palliative care teams can also provide psychosocial and spiritual support for patients and families who are intimidated by the complex navigation of the health care system and the existential threat that end-stage organ failure presents to them. Skilled palliative care and services can be the difference between failing and extended life with a higher quality of life for these very sick patients
Resuscitation of a patient, whether through restoration of organ function or interdicting the progression of disease, begins with resuscitation of hope. Nothing achieves this more quickly than amelioration of burdensome symptoms for the patient and family.
The barriers for transplant surgeons and teams referring and incorporating palliative care services in their practices are multiple and profound. The unique dilemma facing the transplant team is to balance the treatment of the failing organ, the treatment of the patient (and family and friends), and the best use of the graft, a precious gift of society.
Palliative surgery has been defined as any invasive procedure in which the main intention is to mitigate physical symptoms in patients with noncurable disease without causing premature death. The very success of transplantation over the past 3 decades has obscured our memory of transplantation as a type of palliative surgery. It is a well-known axiom of reconstructive surgery that the reconstructed site should be compared to what was there, not to “normal.” Even in the current era of improved immunosuppression and posttransplant support services, one could hardly describe even a successful transplant patient’s experience as “normal.” These patients’ lives may be extended and/or enhanced but they need palliative care before, during, and after transplantation. The growing availability of trained palliative care clinicians and teams, the increased familiarity of palliative and end-of-life care to surgical residents and fellows, and quality metrics measuring palliative care outcomes will provide reassurance and guidance to address reservations about the convergence of the two seemingly opposite realities.
A modest proposal
We propose that palliative care be presented to the entire spectrum of transplantation care: on the ward, in the ICU, and after transplantation. More specific “triggers” for palliative care for referral of transplant patients should be identified. Wentlandt et al.4 have described a promising model for an ambulatory clinic, which provides early, integrated palliative care to patients awaiting and receiving organ transplantation. In addition, we propose an application for grant funding for a conference and eventual formation of a work group of transplant surgeons and team members, palliative care clinicians, and patient/families who have experienced one of the aspects of the transplant spectrum. We await the subspecialty certification in hospice and palliative medicine of a transplant surgeon. Outside of transplantation, every other surgical specialty in the United States has diplomates certified in hospice and palliative medicine. We await the benefits that will accrue from research about the merging of these fields.
1. Molmenti EP, Dunn GP: Transplantation and palliative care: The convergence of two seemingly opposite realities. Surg Clin North Am. 2005;85:373-82.
2. Cook D, Rocker G. Dying with dignity in the intensive care unit. N Engl J Med. 2014;370:2506-14.
3. Lamba S, Murphy P, McVicker S, Smith JH, and Mosenthal AC. Changing end-of-life care practice for liver transplant patients: structured palliative care intervention in the surgical intensive care unit. J Pain Symptom Manage. 2012; 44(4):508-19.
4. Wentlandt, K., Dall’Osto, A., Freeman, N., Le, L. W., Kaya, E., Ross, H., Singer, L. G., Abbey, S., Clarke, H. and Zimmermann, C. (2016), The Transplant Palliative Care Clinic: An early palliative care model for patients in a transplant program. Clin Transplant. 2016 Nov 4; doi: 10.1111/ctr.12838.
Dr. Azoulay is a transplantation specialist of Assistance Publique – Hôpitaux de Paris, and the University of Paris. Dr. Dunn is medical director of the Palliative Care Consultation Service at the University of Pittsburgh Medical Center Hamot, and vice-chair of the ACS Committee on Surgical Palliative Care.
Best Practices: Protecting Dry Vulnerable Skin with CeraVe® Healing Ointment
A supplement to Dermatology News. This advertising supplement is sponsored by Valeant Pharmaceuticals.
- Reinforcing the Skin Barrier
- NEA Seal of Acceptance
- A Preventative Approach to Dry, Cracked Skin
- CeraVe Ointment in the Clinical Setting
Faculty/Faculty Disclosure
Sheila Fallon Friedlander, MD
Professor of Clinical Dermatology & Pediatrics
Director, Pediatric Dermatology Fellowship Training Program
University of California at San Diego School of Medicine
Rady Children’s Hospital,
San Diego, California
Dr. Friedlander was compensated for her participation in the development of this article.
CeraVe is a registered trademark of Valeant Pharmaceuticals International, Inc. or its affiliates.
A supplement to Dermatology News. This advertising supplement is sponsored by Valeant Pharmaceuticals.
- Reinforcing the Skin Barrier
- NEA Seal of Acceptance
- A Preventative Approach to Dry, Cracked Skin
- CeraVe Ointment in the Clinical Setting
Faculty/Faculty Disclosure
Sheila Fallon Friedlander, MD
Professor of Clinical Dermatology & Pediatrics
Director, Pediatric Dermatology Fellowship Training Program
University of California at San Diego School of Medicine
Rady Children’s Hospital,
San Diego, California
Dr. Friedlander was compensated for her participation in the development of this article.
CeraVe is a registered trademark of Valeant Pharmaceuticals International, Inc. or its affiliates.
A supplement to Dermatology News. This advertising supplement is sponsored by Valeant Pharmaceuticals.
- Reinforcing the Skin Barrier
- NEA Seal of Acceptance
- A Preventative Approach to Dry, Cracked Skin
- CeraVe Ointment in the Clinical Setting
Faculty/Faculty Disclosure
Sheila Fallon Friedlander, MD
Professor of Clinical Dermatology & Pediatrics
Director, Pediatric Dermatology Fellowship Training Program
University of California at San Diego School of Medicine
Rady Children’s Hospital,
San Diego, California
Dr. Friedlander was compensated for her participation in the development of this article.
CeraVe is a registered trademark of Valeant Pharmaceuticals International, Inc. or its affiliates.
Involving Concerned Significant Others in Firearm Suicide Prevention: Development of the Family FireArms Secure Storage Training Intervention
Involving Concerned Significant Others in Firearm Suicide Prevention: Development of the Family FireArms Secure Storage Training Intervention
Veterans are at higher risk for suicide compared with civilian populations.1 Firearms are the most frequent cause of death in veteran deaths by suicide, likely because about 51% of veterans own ≥ 1 firearms and firearms are the most lethal and readily available mechanism.1-3 Unsecure firearm storage practices (eg, storing firearms unlocked, in an unsecure location, or loaded with ammunition) are associated with increased suicide risk.4 Conversely, secure firearm storage (ie, storing firearms locked and unloaded) is associated with lower suicide risk.5
A 2019 study of veterans who own firearms found that only 22.2% store all their firearms unloaded and locked, while 32.7% store ≥ 1 firearm unlocked and loaded, and 45.2% store firearms both unlocked and loaded or locked and unloaded. Only 6.3% of veterans strongly agreed that having a firearm at home increased suicide risk among household members; however, 77.2% indicated they would ensure a household member could not access firearms if they were concerned about their suicidal ideation.6
Another study found that 9.2% of veterans receive lethal means safety counseling from their US Department of Veterans Affairs (VA)-affiliated or non-VA health care professional.7 These data highlight a need to educate veterans about the increased risk for suicide associated with storing an unsecured firearm in the household and to connect this understanding to their values of service and protection of others, while simultaneously preparing them and their family members for a potential mental health crisis.
Consistent with the government’s public health approach to suicide prevention, prevention efforts should also enlist the participation of individuals outside health care.8 For example, prior research has found that family members are considered highly credible, and engaging them could expand the reach of lethal means safety conversations. A qualitative analysis of 29 veterans found that 17 (57%) said they preferred having a concerned significant other (CSO) (eg, spouse, adult friend, or relative) involved in their suicide prevention care, while 21 (72%) said they would prefer having a CSO assisting in the secure storage of firearms.9,10 Some veterans may be more amenable to a conversation about firearm access and suicide risk concerns initiated by a CSO rather than by a clinician, indicating the potential benefits of educating and involving CSOs in suicide prevention.11 Involving CSOs in secure firearm storage planning may also strengthen the veteran’s sense of social support, a key protective factor against suicidal ideation.12
CSO involvement in secure firearm storage can provide the following benefits: (1) helping the veteran create a secure storage plan, including developing approaches to secure storage; (2) understanding warning signs of suicide; (3) helping the veteran limit access to firearms during a suicidal crisis; (4) helping the veteran remember the secure storage plan; (5) helping the veteran connect with mental health services; and (6) enhancing social support. In most instances, CSOs are physically close to the veteran (eg, live in the same household) and have a greater practical ability to support and affect change with respect to changes in firearm storage practices.
This article describes the development of an intervention that incorporates CSO involvement in firearms safety efforts for veterans with guidance from VA mental health care practitioners (HCPs). The goal is to provide HCPs and other key stakeholders with a detailed description of the intervention and to suggest potential strategies for how to involve CSOs in suicide prevention.
This article follows the Guideline for Reporting Evidence-based Practice Educational interventions and Teaching checklist, which was developed to facilitate standardized reporting and replication for education interventions.13 Applicable portions of the checklist are outlined, with others (ie, incentives, planned/unplanned changes, attendance, and other outcomes) to be addressed in future research.
FFAST INTERVENTION
Training (FFAST) intervention promotes voluntary secure firearm storage, engages CSOs in veteran mental health care, and provides psychoeducation and skills to support crisis management. The intervention was developed for all veterans who do not securely store firearms.
Theory
The intervention incorporates motivational interviewing techniques, as ambivalence about changing firearm storage behaviors is common, particularly when veterans own firearms for safety or protection.6,14 Motivational interviewing is a collaborative approach that addresses a client’s ambivalence to change by eliciting and exploring the client’s own arguments related to change.14 An important aspect of developing this intervention was to ensure it would be culturally relevant to veteran firearm owners and their CSOs.15 Further, involvement of the CSO is intentional and meant to boost social support, a known buffering factor against suicide risk.12
Objectives
This intervention’s primary objective was for veteran participants to identify secure firearm storage practices and develop a plan for implementing them, including when a veteran or other household member experiences a mental health crisis. For CSOs, the primary objective is to learn how to help the veteran connect with mental health resources if needed and support secure firearm storage as necessary. The overall goal is to learn how to identify warning signs for suicide and how to respond to a mental health crisis through a collaborative process, including securing firearms in a crisis situation.
Materials, Educational Strategies, and Instructors
Training for delivering the intervention was provided via direct consultation with the developer of the intervention and manual. The manual contains pertinent background information to provide context for the intervention’s significance and rationale, including the role of firearms in suicides and current lethal means safety initiatives. It also describes the purpose and objective of each intervention component in detail in addition to providing a script for interventionists to follow to complete each objective.
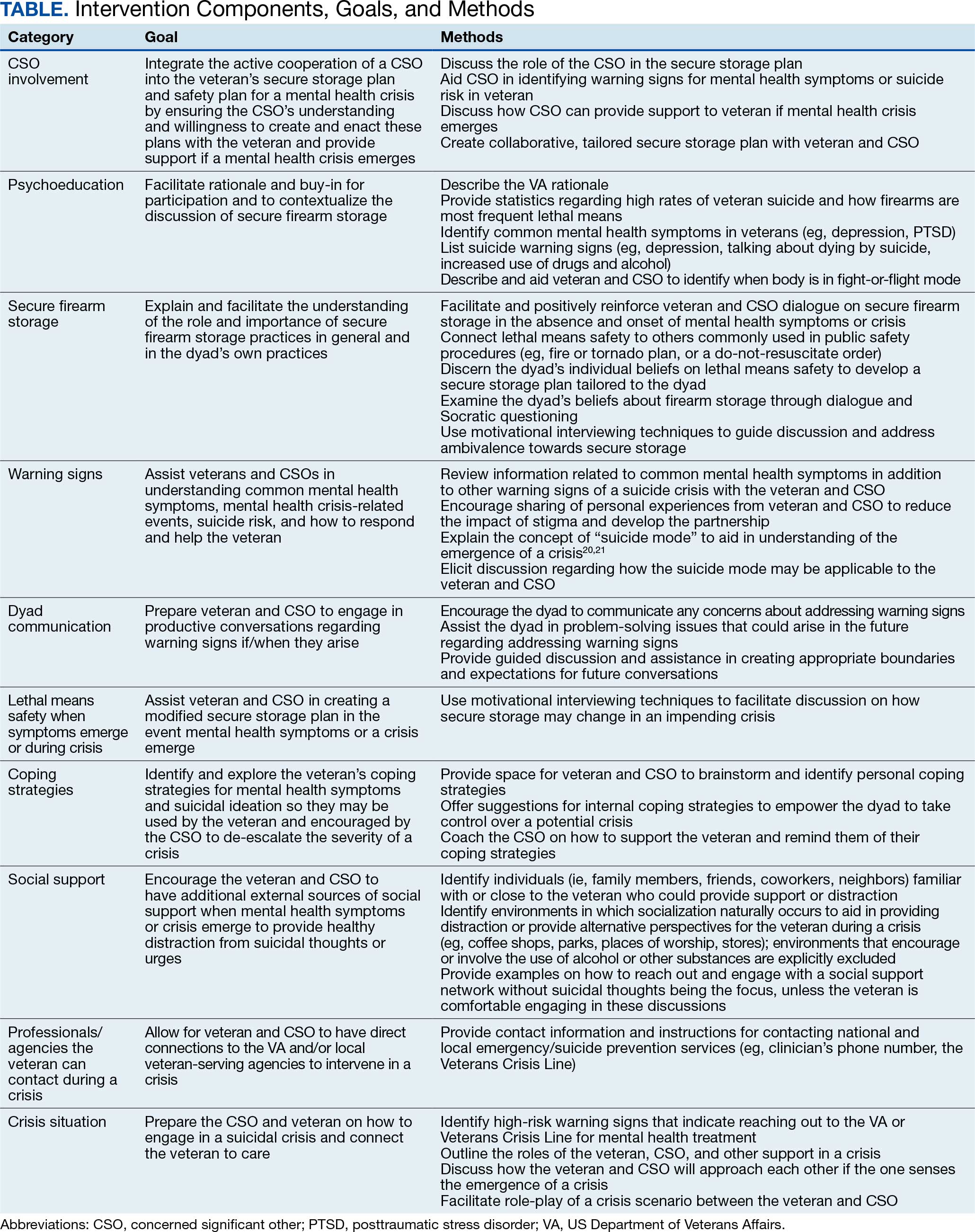
Training materials for veterans and CSOs include a single Firearms Secure Storage Planning worksheet completed during the intervention, with which the interventionist guides participants through the creation of a secure firearm storage plan (Table). Educational strategies include psychoeducation and Socratic questioning (eg, questioning focused on guiding participants toward the intervention goals) delivered verbally by the interventionist.
The intervention is delivered in person or virtually during a single 90-minute session with a veteran and CSO. Veterans and CSOs work with the interventionist to complete collaborative activities during the session and have self-directive learning activities or homework.
The intervention has 4 primary components: (1) CSO involvement; (2) psychoeducation; (3) secure firearm storage; and (4) how to respond to a mental health crisis. Each CSO should have an established relationship with the veteran, be willing and able to be present during the intervention, and remain an encouraging support person for the veteran. The interventionist emphasizes that it is part of the VA mission for staff to care about the veteran, and that initiating such contact with a CSO is meant to prioritize veteran safety and the safety of their family. Psychoeducation on mental health symptoms, suicide warning signs, veteran suicide rates and lethal means, and the benefits of secure firearm storage, is incorporated in the intervention.
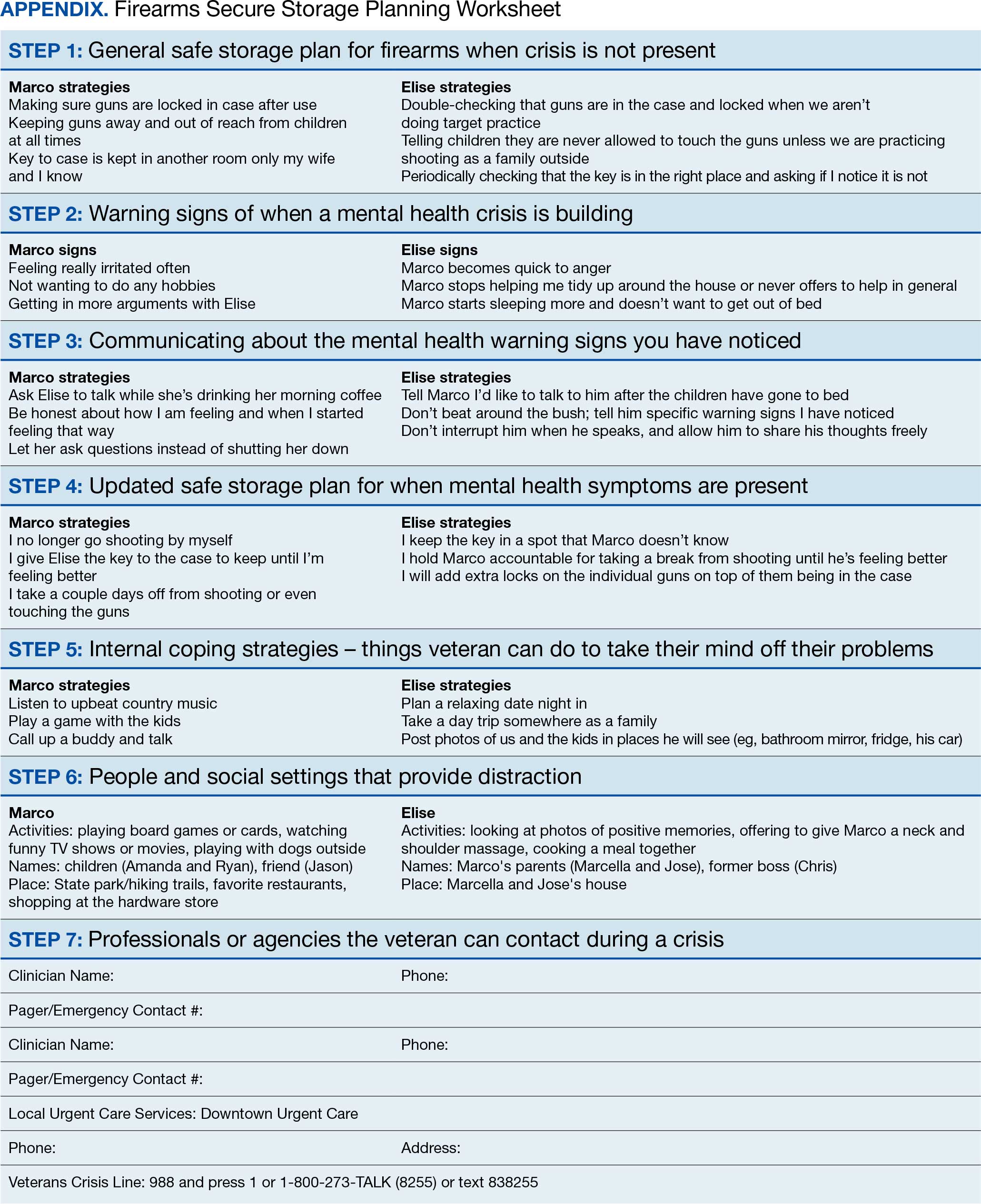
The secure firearm storage component consists of 7 subcomponents: (1) general lethal means secure storage; (2) warning signs; (3) dyad communication; (4) lethal means safety when symptoms emerge; (5) coping strategies; (6) social support; and (7) emergency contacts. A lethal means safety worksheet rooted in the Stanley and Brown suicide safety plan model and implemented in VA health care settings is used to facilitate discussions of secure storage (Appendix).16
CSOs typically have little or no suicidal crisis response training, yet they likely have more interaction with the veteran on a daily basis than HCPs, putting them in a vital position to identify a crisis early and connect the veteran with the proper care. The crisis component prepares the CSO and veteran to navigate a crisis scenario so they can practice their newly developed safety plan and increase their comfort in discussing mental health and suicidal crisis.
FICTIONAL CASE STUDY
Cole, aged 59 years, is a Persian Gulf War veteran and retired police officer. His medical history includes hypothyroidism, hypertension, type 2 diabetes mellitus, chronic posttraumatic stress disorder, major depressive disorder, and insomnia.
Cole's wife of > 30 years, Sheila, joined him for the FFAST intervention. They report having 4 firearms in the home, 3 of which are loaded but stored in a lockbox and 1 that Cole reports is kept on his person for protection. Cole reports passive suicidal ideation, but no plans or intent. When discussing warning signs that a mental health crisis is building, Cole describes feeling anxious, having a change in his speech patterns, and isolating himself. Sheila agrees, but also mentions that Cole is easily angered and becomes nonverbal. Cole and Sheila express difficulty communicating and appear to have a breakthrough moment when Cole says he does not like when Sheila repeats herself, as he feels like she is “poking” at him. Sheila shares concerns for his safety and that she only repeats herself because he refuses to talk.
Cole agrees to verbalize that he is safe but needs time to process his thoughts. Sheila agrees to give him space with a plan to revisit the conversation within an agreed upon timeline. When discussing an updated secure storage plan for their firearms when a mental health crisis is building, Cole commits to allowing Sheila to store the firearm currently on his person in their gun safe, with the ammunition stored separately, and to giving her the gun safe key. They agree to implement this practice until the mental health crisis has passed.
To mitigate a potential crisis, the interventionist discusses possible internal coping strategies for Cole, including writing, reading, walking the dog, listening to music, and baking. People and social settings that could provide distraction involve going to the gym, talking to his friend Carl or his daughter Kelly, and attending the men’s ministry at church. The intervention concludes by discussing professionals or agencies that Cole and Sheila could contact during a crisis. After the intervention, Cole and Sheila are asked to rate their likelihood of using the plan they established during the conversation on a scale of 0 to 10, with 0 being highly unlikely and 10 being extremely likely. Cole responds with 9 and Sheila responds with 10.
DISCUSSION
Lethal means safety remains a critical component of veteran suicide prevention. However, lethal means safety discussions are often implemented after suicide risk has been identified, which may be too late. Thus, having these conversations early and before a crisis may be imperative. Veterans have expressed a desire to have CSOs involved in their suicide prevention treatment, and CSOs can play a key role in recognizing risk factors during everyday life. The FFAST intervention addresses many of these gaps.
Having discussions in advance of a crisis allows veterans to consider an effective secure firearm storage plan outside of the context of a crisis. Including a CSO galvanizes another person to understand a veteran’s needs and assist with secure firearm storage, identify warning signs, and support them during a crisis. These discussions occur in a context where there is less pressure than during a crisis. Features that were more appealing to veterans and their CSOs were also incorporated, such as having the dyad build a plan that is conceptually similar to other public safety initiatives (eg, a fire safety plan, tornado plan, or hurricane plan). Previous research demonstrates that veterans appreciated the nonjudgmental approach and some preferred that clinicians approach the discussion of secure firearm storage within the context of general home and family safety.17 Additionally, this intervention can build on veterans’ prior military training in preparedness.
Other potential benefits associated with the FFAST intervention include creating an opportunity to strengthen communication between the veteran and CSO. While FFAST is intended to be used with all types of CSOs, this work is consistent with preliminary data from a couples-based suicide prevention study that indicated veterans and their partners reported increases in relationship functioning and marginal decreases in suicidal ideation.18 It is possible that communication strategies gained from the current intervention could improve veterans’ relationships with their CSOs, which are associated with a greater sense of social support and reduced suicide risk.12
The intervention is a brief, single session that may be appealing to veterans and CSOs with full schedules. Evidence suggests that even brief, single-session interventions have a significant impact on beliefs about secure firearm storage, knowledge of lethal means safety, and confidence in having secure firearm storage conversations.19 However, clinicians should be cautious when extrapolating from the findings of the current case example, which was a one-time intervention with no follow-up.
Future Directions
Pilot testing of the proposed intervention is underway, and future research will include feedback from veterans and CSOs, as well as feasibility and acceptability data collected during the pilot process. The pilot study uses a successive cohort design with an initial 2 sets of 5 veteran and CSO dyads, and subsequent funding has expanded the pilot study to include an additional 30 dyads. Qualitative interviews will be conducted separately with each veteran and CSO, and additional constructs such as feasibility, acceptability, barriers and facilitators to implementation, and changes in secure storage will be examined. This future research may provide a deeper understanding of the broader acceptability, feasibility, and satisfaction associated with a suicide prevention intervention focused on securing firearms and involving veterans and their CSOs. These data could be used to inform future implementation trials and inform the development of an implementation strategy. In the interim, the nature of the manual is summarized in the context of the urgency of suicide prevention in this at-risk population.
Conclusions
FFAST is a novel approach to veteran firearm suicide prevention. By involving CSOs and emphasizing mental health crisis preparedness between them and veterans, the dyad can work in association with HCPs to establish and exercise secure firearm storage practices as part of an at-home safety plan. Implementation of FFAST may be beneficial for all veterans, not only those who have been identified as being at high suicide risk.
- US Dept of Veterans Affairs Office of Suicide Prevention. 2024 national veteran suicide prevention annual report. December 2024. Accessed February 5, 2026. https://www.mentalhealth.va.gov/docs/data-sheets/2024/2024-Annual-Report-Part-2-of-2_508.pdf
- Fischer IC, Aunon FM, Nichter B, et al. Firearm ownership among a nationally representative sample of U.S. veterans. Am J Prev Med. 2023;65:1129-1133. doi:10.1016/j.amepre.2023.06.013
- Conner A, Azrael D, Miller M. Suicide case-fatality rates in the United States, 2007-2014: a nationwide population-based study. Ann Intern Med. 2019;171(12):885-895. doi:10.7326/M19-1324
- Dempsey CL, Benedek DM, Zuromski KL, et al. Association of firearm ownership, use, accessibility, and storage practices with suicide risk among US army soldiers. JAMA Netw Open. 2019;2:e195383. doi:10.1001/jamanetworkopen.2019.5383
- Butterworth SE, Daruwala SE, Anestis MD. Firearm storage and shooting experience: factors relevant to the practical capability for suicide. J Psychiatr Res. 2018;102:52-56. doi:10.1016/j.jpsychires.2018.03.010
- Simonetti JA, Azrael D, Miller M. Firearm storage practices and risk perceptions among a nationally representative sample of U.S. veterans with and without self-harm risk factors. Suicide Life Threat Behav. 2019;49:653-664. doi:10.1111/sltb.12463
- Simonetti JA, Azrael D, Zhang W, Miller M. Receipt of clinician-delivered firearm safety counseling among U.S. veterans: results from a 2019 national survey. Suicide Life Threat Behav. 2022;52:1121-1125. doi:10.1111/sltb.12906
- US Office of the Surgeon General. The surgeon general’s call to action to implement the national strategy for suicide prevention. January 2021. Accessed February 5, 2026. https://www.hhs.gov/sites/default/files/sprc-call-to-action.pdf
- DeBeer BB, Matthieu MM, Kittel JA, et al. Quality Improvement Evaluation of the Feasibility and Acceptability of Adding a Concerned Significant Other to Safety Planning for Suicide Prevention With Veterans. J Ment Health Couns. 2019;41:4-20. doi:10.17744/mehc.41.1.02
- DeBeer BB, Matthieu MM, Degutis LC, et al. Firearms lethal means safety among veterans: attitudes toward involving a concerned significant other. J Mil Veteran Fam Health. 2025;11:23-31.
- Monteith LL, Holliday R, Dorsey Holliman BA, et al. Understanding female veterans’ experiences and perspectives of firearms. J Clin Psychol. 2020;76:1736-1753. doi:10.1002/jclp.22952
- DeBeer BB, Kimbrel NA, Meyer EC, et al. Combined PTSD and depressive symptoms interact with post-deployment social support to predict suicidal ideation in Operation Enduring Freedom and Operation Iraqi Freedom veterans. Psychiatry Res. 2014;216:357-362. doi:10.1016/j.psychres.2014.02.010
- Phillips AC, Lewis LK, McEvoy MP, et al. Development and validation of the guideline for reporting evidence-based practice educational interventions and teaching (GREET). BMC Med Educ. 2016;16:237. doi:10.1186/s12909-016-0759-1
- Miller WR, Rollnick S. Motivational Interviewing: Helping People Change. 3rd ed. Guilford Press; 2013.
- Khazanov GK, Keddem S, Hoskins K, et al. Stakeholder perceptions of lethal means safety counseling: a qualitative systematic review. Front Psychiatry. 2022;13:993415. doi:10.3389/fpsyt.2022.993415
- Stanley B, Brown GK, Karlin B, et al. US Dept of Veterans Affairs. Safety plan treatment manual to reduce suicide risk: veteran version. August 20, 2008. Accessed February 5, 2026. https://www.mentalhealth.va.gov/mentalhealth/docs/va_safety_planning_manual.doc
- Dobscha SK, Clark KD, Newell S, et al. Strategies for discussing firearms storage safety in primary care: veteran perspectives. J Gen Intern Med. 2021;36:1492-1502. doi:10.1007/s11606-020-06412-x
- Khalifian CE, Leifker FR, Knopp K, et al. Utilizing the couple relationship to prevent suicide: a preliminary examination of treatment for relationships and safety together. J Clin Psych. 2022;78:747-757. doi:10.1002/jclp.23251
- Walsh A, Friedman K, Morrissey BH, et al. Project Safe Guard: evaluating a lethal means safety intervention to reduce firearm suicide in the National Guard. Mil Med. 2024;189:510-516. doi:10.1093/milmed/usae172
- Beck AT. Beyond belief: a theory of modes, personality, and psychopathology. In: Salkovkis PM, ed. Frontiers of Cognitive Therapy. Guilford Press;1996:1-25.
- Rudd MD. The suicidal mode: a cognitive-behavioral model of suicidality. Suicide Life Threat Behav. 2000;30(1):18-33.
Veterans are at higher risk for suicide compared with civilian populations.1 Firearms are the most frequent cause of death in veteran deaths by suicide, likely because about 51% of veterans own ≥ 1 firearms and firearms are the most lethal and readily available mechanism.1-3 Unsecure firearm storage practices (eg, storing firearms unlocked, in an unsecure location, or loaded with ammunition) are associated with increased suicide risk.4 Conversely, secure firearm storage (ie, storing firearms locked and unloaded) is associated with lower suicide risk.5
A 2019 study of veterans who own firearms found that only 22.2% store all their firearms unloaded and locked, while 32.7% store ≥ 1 firearm unlocked and loaded, and 45.2% store firearms both unlocked and loaded or locked and unloaded. Only 6.3% of veterans strongly agreed that having a firearm at home increased suicide risk among household members; however, 77.2% indicated they would ensure a household member could not access firearms if they were concerned about their suicidal ideation.6
Another study found that 9.2% of veterans receive lethal means safety counseling from their US Department of Veterans Affairs (VA)-affiliated or non-VA health care professional.7 These data highlight a need to educate veterans about the increased risk for suicide associated with storing an unsecured firearm in the household and to connect this understanding to their values of service and protection of others, while simultaneously preparing them and their family members for a potential mental health crisis.
Consistent with the government’s public health approach to suicide prevention, prevention efforts should also enlist the participation of individuals outside health care.8 For example, prior research has found that family members are considered highly credible, and engaging them could expand the reach of lethal means safety conversations. A qualitative analysis of 29 veterans found that 17 (57%) said they preferred having a concerned significant other (CSO) (eg, spouse, adult friend, or relative) involved in their suicide prevention care, while 21 (72%) said they would prefer having a CSO assisting in the secure storage of firearms.9,10 Some veterans may be more amenable to a conversation about firearm access and suicide risk concerns initiated by a CSO rather than by a clinician, indicating the potential benefits of educating and involving CSOs in suicide prevention.11 Involving CSOs in secure firearm storage planning may also strengthen the veteran’s sense of social support, a key protective factor against suicidal ideation.12
CSO involvement in secure firearm storage can provide the following benefits: (1) helping the veteran create a secure storage plan, including developing approaches to secure storage; (2) understanding warning signs of suicide; (3) helping the veteran limit access to firearms during a suicidal crisis; (4) helping the veteran remember the secure storage plan; (5) helping the veteran connect with mental health services; and (6) enhancing social support. In most instances, CSOs are physically close to the veteran (eg, live in the same household) and have a greater practical ability to support and affect change with respect to changes in firearm storage practices.
This article describes the development of an intervention that incorporates CSO involvement in firearms safety efforts for veterans with guidance from VA mental health care practitioners (HCPs). The goal is to provide HCPs and other key stakeholders with a detailed description of the intervention and to suggest potential strategies for how to involve CSOs in suicide prevention.
This article follows the Guideline for Reporting Evidence-based Practice Educational interventions and Teaching checklist, which was developed to facilitate standardized reporting and replication for education interventions.13 Applicable portions of the checklist are outlined, with others (ie, incentives, planned/unplanned changes, attendance, and other outcomes) to be addressed in future research.
FFAST INTERVENTION
Training (FFAST) intervention promotes voluntary secure firearm storage, engages CSOs in veteran mental health care, and provides psychoeducation and skills to support crisis management. The intervention was developed for all veterans who do not securely store firearms.
Theory
The intervention incorporates motivational interviewing techniques, as ambivalence about changing firearm storage behaviors is common, particularly when veterans own firearms for safety or protection.6,14 Motivational interviewing is a collaborative approach that addresses a client’s ambivalence to change by eliciting and exploring the client’s own arguments related to change.14 An important aspect of developing this intervention was to ensure it would be culturally relevant to veteran firearm owners and their CSOs.15 Further, involvement of the CSO is intentional and meant to boost social support, a known buffering factor against suicide risk.12
Objectives
This intervention’s primary objective was for veteran participants to identify secure firearm storage practices and develop a plan for implementing them, including when a veteran or other household member experiences a mental health crisis. For CSOs, the primary objective is to learn how to help the veteran connect with mental health resources if needed and support secure firearm storage as necessary. The overall goal is to learn how to identify warning signs for suicide and how to respond to a mental health crisis through a collaborative process, including securing firearms in a crisis situation.
Materials, Educational Strategies, and Instructors
Training for delivering the intervention was provided via direct consultation with the developer of the intervention and manual. The manual contains pertinent background information to provide context for the intervention’s significance and rationale, including the role of firearms in suicides and current lethal means safety initiatives. It also describes the purpose and objective of each intervention component in detail in addition to providing a script for interventionists to follow to complete each objective.
Training materials for veterans and CSOs include a single Firearms Secure Storage Planning worksheet completed during the intervention, with which the interventionist guides participants through the creation of a secure firearm storage plan (Table). Educational strategies include psychoeducation and Socratic questioning (eg, questioning focused on guiding participants toward the intervention goals) delivered verbally by the interventionist.

The intervention is delivered in person or virtually during a single 90-minute session with a veteran and CSO. Veterans and CSOs work with the interventionist to complete collaborative activities during the session and have self-directive learning activities or homework.
The intervention has 4 primary components: (1) CSO involvement; (2) psychoeducation; (3) secure firearm storage; and (4) how to respond to a mental health crisis. Each CSO should have an established relationship with the veteran, be willing and able to be present during the intervention, and remain an encouraging support person for the veteran. The interventionist emphasizes that it is part of the VA mission for staff to care about the veteran, and that initiating such contact with a CSO is meant to prioritize veteran safety and the safety of their family. Psychoeducation on mental health symptoms, suicide warning signs, veteran suicide rates and lethal means, and the benefits of secure firearm storage, is incorporated in the intervention.
The secure firearm storage component consists of 7 subcomponents: (1) general lethal means secure storage; (2) warning signs; (3) dyad communication; (4) lethal means safety when symptoms emerge; (5) coping strategies; (6) social support; and (7) emergency contacts. A lethal means safety worksheet rooted in the Stanley and Brown suicide safety plan model and implemented in VA health care settings is used to facilitate discussions of secure storage (Appendix).16

CSOs typically have little or no suicidal crisis response training, yet they likely have more interaction with the veteran on a daily basis than HCPs, putting them in a vital position to identify a crisis early and connect the veteran with the proper care. The crisis component prepares the CSO and veteran to navigate a crisis scenario so they can practice their newly developed safety plan and increase their comfort in discussing mental health and suicidal crisis.
FICTIONAL CASE STUDY
Cole, aged 59 years, is a Persian Gulf War veteran and retired police officer. His medical history includes hypothyroidism, hypertension, type 2 diabetes mellitus, chronic posttraumatic stress disorder, major depressive disorder, and insomnia.
Cole's wife of > 30 years, Sheila, joined him for the FFAST intervention. They report having 4 firearms in the home, 3 of which are loaded but stored in a lockbox and 1 that Cole reports is kept on his person for protection. Cole reports passive suicidal ideation, but no plans or intent. When discussing warning signs that a mental health crisis is building, Cole describes feeling anxious, having a change in his speech patterns, and isolating himself. Sheila agrees, but also mentions that Cole is easily angered and becomes nonverbal. Cole and Sheila express difficulty communicating and appear to have a breakthrough moment when Cole says he does not like when Sheila repeats herself, as he feels like she is “poking” at him. Sheila shares concerns for his safety and that she only repeats herself because he refuses to talk.
Cole agrees to verbalize that he is safe but needs time to process his thoughts. Sheila agrees to give him space with a plan to revisit the conversation within an agreed upon timeline. When discussing an updated secure storage plan for their firearms when a mental health crisis is building, Cole commits to allowing Sheila to store the firearm currently on his person in their gun safe, with the ammunition stored separately, and to giving her the gun safe key. They agree to implement this practice until the mental health crisis has passed.
To mitigate a potential crisis, the interventionist discusses possible internal coping strategies for Cole, including writing, reading, walking the dog, listening to music, and baking. People and social settings that could provide distraction involve going to the gym, talking to his friend Carl or his daughter Kelly, and attending the men’s ministry at church. The intervention concludes by discussing professionals or agencies that Cole and Sheila could contact during a crisis. After the intervention, Cole and Sheila are asked to rate their likelihood of using the plan they established during the conversation on a scale of 0 to 10, with 0 being highly unlikely and 10 being extremely likely. Cole responds with 9 and Sheila responds with 10.
DISCUSSION
Lethal means safety remains a critical component of veteran suicide prevention. However, lethal means safety discussions are often implemented after suicide risk has been identified, which may be too late. Thus, having these conversations early and before a crisis may be imperative. Veterans have expressed a desire to have CSOs involved in their suicide prevention treatment, and CSOs can play a key role in recognizing risk factors during everyday life. The FFAST intervention addresses many of these gaps.
Having discussions in advance of a crisis allows veterans to consider an effective secure firearm storage plan outside of the context of a crisis. Including a CSO galvanizes another person to understand a veteran’s needs and assist with secure firearm storage, identify warning signs, and support them during a crisis. These discussions occur in a context where there is less pressure than during a crisis. Features that were more appealing to veterans and their CSOs were also incorporated, such as having the dyad build a plan that is conceptually similar to other public safety initiatives (eg, a fire safety plan, tornado plan, or hurricane plan). Previous research demonstrates that veterans appreciated the nonjudgmental approach and some preferred that clinicians approach the discussion of secure firearm storage within the context of general home and family safety.17 Additionally, this intervention can build on veterans’ prior military training in preparedness.
Other potential benefits associated with the FFAST intervention include creating an opportunity to strengthen communication between the veteran and CSO. While FFAST is intended to be used with all types of CSOs, this work is consistent with preliminary data from a couples-based suicide prevention study that indicated veterans and their partners reported increases in relationship functioning and marginal decreases in suicidal ideation.18 It is possible that communication strategies gained from the current intervention could improve veterans’ relationships with their CSOs, which are associated with a greater sense of social support and reduced suicide risk.12
The intervention is a brief, single session that may be appealing to veterans and CSOs with full schedules. Evidence suggests that even brief, single-session interventions have a significant impact on beliefs about secure firearm storage, knowledge of lethal means safety, and confidence in having secure firearm storage conversations.19 However, clinicians should be cautious when extrapolating from the findings of the current case example, which was a one-time intervention with no follow-up.
Future Directions
Pilot testing of the proposed intervention is underway, and future research will include feedback from veterans and CSOs, as well as feasibility and acceptability data collected during the pilot process. The pilot study uses a successive cohort design with an initial 2 sets of 5 veteran and CSO dyads, and subsequent funding has expanded the pilot study to include an additional 30 dyads. Qualitative interviews will be conducted separately with each veteran and CSO, and additional constructs such as feasibility, acceptability, barriers and facilitators to implementation, and changes in secure storage will be examined. This future research may provide a deeper understanding of the broader acceptability, feasibility, and satisfaction associated with a suicide prevention intervention focused on securing firearms and involving veterans and their CSOs. These data could be used to inform future implementation trials and inform the development of an implementation strategy. In the interim, the nature of the manual is summarized in the context of the urgency of suicide prevention in this at-risk population.
Conclusions
FFAST is a novel approach to veteran firearm suicide prevention. By involving CSOs and emphasizing mental health crisis preparedness between them and veterans, the dyad can work in association with HCPs to establish and exercise secure firearm storage practices as part of an at-home safety plan. Implementation of FFAST may be beneficial for all veterans, not only those who have been identified as being at high suicide risk.
Veterans are at higher risk for suicide compared with civilian populations.1 Firearms are the most frequent cause of death in veteran deaths by suicide, likely because about 51% of veterans own ≥ 1 firearms and firearms are the most lethal and readily available mechanism.1-3 Unsecure firearm storage practices (eg, storing firearms unlocked, in an unsecure location, or loaded with ammunition) are associated with increased suicide risk.4 Conversely, secure firearm storage (ie, storing firearms locked and unloaded) is associated with lower suicide risk.5
A 2019 study of veterans who own firearms found that only 22.2% store all their firearms unloaded and locked, while 32.7% store ≥ 1 firearm unlocked and loaded, and 45.2% store firearms both unlocked and loaded or locked and unloaded. Only 6.3% of veterans strongly agreed that having a firearm at home increased suicide risk among household members; however, 77.2% indicated they would ensure a household member could not access firearms if they were concerned about their suicidal ideation.6
Another study found that 9.2% of veterans receive lethal means safety counseling from their US Department of Veterans Affairs (VA)-affiliated or non-VA health care professional.7 These data highlight a need to educate veterans about the increased risk for suicide associated with storing an unsecured firearm in the household and to connect this understanding to their values of service and protection of others, while simultaneously preparing them and their family members for a potential mental health crisis.
Consistent with the government’s public health approach to suicide prevention, prevention efforts should also enlist the participation of individuals outside health care.8 For example, prior research has found that family members are considered highly credible, and engaging them could expand the reach of lethal means safety conversations. A qualitative analysis of 29 veterans found that 17 (57%) said they preferred having a concerned significant other (CSO) (eg, spouse, adult friend, or relative) involved in their suicide prevention care, while 21 (72%) said they would prefer having a CSO assisting in the secure storage of firearms.9,10 Some veterans may be more amenable to a conversation about firearm access and suicide risk concerns initiated by a CSO rather than by a clinician, indicating the potential benefits of educating and involving CSOs in suicide prevention.11 Involving CSOs in secure firearm storage planning may also strengthen the veteran’s sense of social support, a key protective factor against suicidal ideation.12
CSO involvement in secure firearm storage can provide the following benefits: (1) helping the veteran create a secure storage plan, including developing approaches to secure storage; (2) understanding warning signs of suicide; (3) helping the veteran limit access to firearms during a suicidal crisis; (4) helping the veteran remember the secure storage plan; (5) helping the veteran connect with mental health services; and (6) enhancing social support. In most instances, CSOs are physically close to the veteran (eg, live in the same household) and have a greater practical ability to support and affect change with respect to changes in firearm storage practices.
This article describes the development of an intervention that incorporates CSO involvement in firearms safety efforts for veterans with guidance from VA mental health care practitioners (HCPs). The goal is to provide HCPs and other key stakeholders with a detailed description of the intervention and to suggest potential strategies for how to involve CSOs in suicide prevention.
This article follows the Guideline for Reporting Evidence-based Practice Educational interventions and Teaching checklist, which was developed to facilitate standardized reporting and replication for education interventions.13 Applicable portions of the checklist are outlined, with others (ie, incentives, planned/unplanned changes, attendance, and other outcomes) to be addressed in future research.
FFAST INTERVENTION
Training (FFAST) intervention promotes voluntary secure firearm storage, engages CSOs in veteran mental health care, and provides psychoeducation and skills to support crisis management. The intervention was developed for all veterans who do not securely store firearms.
Theory
The intervention incorporates motivational interviewing techniques, as ambivalence about changing firearm storage behaviors is common, particularly when veterans own firearms for safety or protection.6,14 Motivational interviewing is a collaborative approach that addresses a client’s ambivalence to change by eliciting and exploring the client’s own arguments related to change.14 An important aspect of developing this intervention was to ensure it would be culturally relevant to veteran firearm owners and their CSOs.15 Further, involvement of the CSO is intentional and meant to boost social support, a known buffering factor against suicide risk.12
Objectives
This intervention’s primary objective was for veteran participants to identify secure firearm storage practices and develop a plan for implementing them, including when a veteran or other household member experiences a mental health crisis. For CSOs, the primary objective is to learn how to help the veteran connect with mental health resources if needed and support secure firearm storage as necessary. The overall goal is to learn how to identify warning signs for suicide and how to respond to a mental health crisis through a collaborative process, including securing firearms in a crisis situation.
Materials, Educational Strategies, and Instructors
Training for delivering the intervention was provided via direct consultation with the developer of the intervention and manual. The manual contains pertinent background information to provide context for the intervention’s significance and rationale, including the role of firearms in suicides and current lethal means safety initiatives. It also describes the purpose and objective of each intervention component in detail in addition to providing a script for interventionists to follow to complete each objective.
Training materials for veterans and CSOs include a single Firearms Secure Storage Planning worksheet completed during the intervention, with which the interventionist guides participants through the creation of a secure firearm storage plan (Table). Educational strategies include psychoeducation and Socratic questioning (eg, questioning focused on guiding participants toward the intervention goals) delivered verbally by the interventionist.

The intervention is delivered in person or virtually during a single 90-minute session with a veteran and CSO. Veterans and CSOs work with the interventionist to complete collaborative activities during the session and have self-directive learning activities or homework.
The intervention has 4 primary components: (1) CSO involvement; (2) psychoeducation; (3) secure firearm storage; and (4) how to respond to a mental health crisis. Each CSO should have an established relationship with the veteran, be willing and able to be present during the intervention, and remain an encouraging support person for the veteran. The interventionist emphasizes that it is part of the VA mission for staff to care about the veteran, and that initiating such contact with a CSO is meant to prioritize veteran safety and the safety of their family. Psychoeducation on mental health symptoms, suicide warning signs, veteran suicide rates and lethal means, and the benefits of secure firearm storage, is incorporated in the intervention.
The secure firearm storage component consists of 7 subcomponents: (1) general lethal means secure storage; (2) warning signs; (3) dyad communication; (4) lethal means safety when symptoms emerge; (5) coping strategies; (6) social support; and (7) emergency contacts. A lethal means safety worksheet rooted in the Stanley and Brown suicide safety plan model and implemented in VA health care settings is used to facilitate discussions of secure storage (Appendix).16

CSOs typically have little or no suicidal crisis response training, yet they likely have more interaction with the veteran on a daily basis than HCPs, putting them in a vital position to identify a crisis early and connect the veteran with the proper care. The crisis component prepares the CSO and veteran to navigate a crisis scenario so they can practice their newly developed safety plan and increase their comfort in discussing mental health and suicidal crisis.
FICTIONAL CASE STUDY
Cole, aged 59 years, is a Persian Gulf War veteran and retired police officer. His medical history includes hypothyroidism, hypertension, type 2 diabetes mellitus, chronic posttraumatic stress disorder, major depressive disorder, and insomnia.
Cole's wife of > 30 years, Sheila, joined him for the FFAST intervention. They report having 4 firearms in the home, 3 of which are loaded but stored in a lockbox and 1 that Cole reports is kept on his person for protection. Cole reports passive suicidal ideation, but no plans or intent. When discussing warning signs that a mental health crisis is building, Cole describes feeling anxious, having a change in his speech patterns, and isolating himself. Sheila agrees, but also mentions that Cole is easily angered and becomes nonverbal. Cole and Sheila express difficulty communicating and appear to have a breakthrough moment when Cole says he does not like when Sheila repeats herself, as he feels like she is “poking” at him. Sheila shares concerns for his safety and that she only repeats herself because he refuses to talk.
Cole agrees to verbalize that he is safe but needs time to process his thoughts. Sheila agrees to give him space with a plan to revisit the conversation within an agreed upon timeline. When discussing an updated secure storage plan for their firearms when a mental health crisis is building, Cole commits to allowing Sheila to store the firearm currently on his person in their gun safe, with the ammunition stored separately, and to giving her the gun safe key. They agree to implement this practice until the mental health crisis has passed.
To mitigate a potential crisis, the interventionist discusses possible internal coping strategies for Cole, including writing, reading, walking the dog, listening to music, and baking. People and social settings that could provide distraction involve going to the gym, talking to his friend Carl or his daughter Kelly, and attending the men’s ministry at church. The intervention concludes by discussing professionals or agencies that Cole and Sheila could contact during a crisis. After the intervention, Cole and Sheila are asked to rate their likelihood of using the plan they established during the conversation on a scale of 0 to 10, with 0 being highly unlikely and 10 being extremely likely. Cole responds with 9 and Sheila responds with 10.
DISCUSSION
Lethal means safety remains a critical component of veteran suicide prevention. However, lethal means safety discussions are often implemented after suicide risk has been identified, which may be too late. Thus, having these conversations early and before a crisis may be imperative. Veterans have expressed a desire to have CSOs involved in their suicide prevention treatment, and CSOs can play a key role in recognizing risk factors during everyday life. The FFAST intervention addresses many of these gaps.
Having discussions in advance of a crisis allows veterans to consider an effective secure firearm storage plan outside of the context of a crisis. Including a CSO galvanizes another person to understand a veteran’s needs and assist with secure firearm storage, identify warning signs, and support them during a crisis. These discussions occur in a context where there is less pressure than during a crisis. Features that were more appealing to veterans and their CSOs were also incorporated, such as having the dyad build a plan that is conceptually similar to other public safety initiatives (eg, a fire safety plan, tornado plan, or hurricane plan). Previous research demonstrates that veterans appreciated the nonjudgmental approach and some preferred that clinicians approach the discussion of secure firearm storage within the context of general home and family safety.17 Additionally, this intervention can build on veterans’ prior military training in preparedness.
Other potential benefits associated with the FFAST intervention include creating an opportunity to strengthen communication between the veteran and CSO. While FFAST is intended to be used with all types of CSOs, this work is consistent with preliminary data from a couples-based suicide prevention study that indicated veterans and their partners reported increases in relationship functioning and marginal decreases in suicidal ideation.18 It is possible that communication strategies gained from the current intervention could improve veterans’ relationships with their CSOs, which are associated with a greater sense of social support and reduced suicide risk.12
The intervention is a brief, single session that may be appealing to veterans and CSOs with full schedules. Evidence suggests that even brief, single-session interventions have a significant impact on beliefs about secure firearm storage, knowledge of lethal means safety, and confidence in having secure firearm storage conversations.19 However, clinicians should be cautious when extrapolating from the findings of the current case example, which was a one-time intervention with no follow-up.
Future Directions
Pilot testing of the proposed intervention is underway, and future research will include feedback from veterans and CSOs, as well as feasibility and acceptability data collected during the pilot process. The pilot study uses a successive cohort design with an initial 2 sets of 5 veteran and CSO dyads, and subsequent funding has expanded the pilot study to include an additional 30 dyads. Qualitative interviews will be conducted separately with each veteran and CSO, and additional constructs such as feasibility, acceptability, barriers and facilitators to implementation, and changes in secure storage will be examined. This future research may provide a deeper understanding of the broader acceptability, feasibility, and satisfaction associated with a suicide prevention intervention focused on securing firearms and involving veterans and their CSOs. These data could be used to inform future implementation trials and inform the development of an implementation strategy. In the interim, the nature of the manual is summarized in the context of the urgency of suicide prevention in this at-risk population.
Conclusions
FFAST is a novel approach to veteran firearm suicide prevention. By involving CSOs and emphasizing mental health crisis preparedness between them and veterans, the dyad can work in association with HCPs to establish and exercise secure firearm storage practices as part of an at-home safety plan. Implementation of FFAST may be beneficial for all veterans, not only those who have been identified as being at high suicide risk.
- US Dept of Veterans Affairs Office of Suicide Prevention. 2024 national veteran suicide prevention annual report. December 2024. Accessed February 5, 2026. https://www.mentalhealth.va.gov/docs/data-sheets/2024/2024-Annual-Report-Part-2-of-2_508.pdf
- Fischer IC, Aunon FM, Nichter B, et al. Firearm ownership among a nationally representative sample of U.S. veterans. Am J Prev Med. 2023;65:1129-1133. doi:10.1016/j.amepre.2023.06.013
- Conner A, Azrael D, Miller M. Suicide case-fatality rates in the United States, 2007-2014: a nationwide population-based study. Ann Intern Med. 2019;171(12):885-895. doi:10.7326/M19-1324
- Dempsey CL, Benedek DM, Zuromski KL, et al. Association of firearm ownership, use, accessibility, and storage practices with suicide risk among US army soldiers. JAMA Netw Open. 2019;2:e195383. doi:10.1001/jamanetworkopen.2019.5383
- Butterworth SE, Daruwala SE, Anestis MD. Firearm storage and shooting experience: factors relevant to the practical capability for suicide. J Psychiatr Res. 2018;102:52-56. doi:10.1016/j.jpsychires.2018.03.010
- Simonetti JA, Azrael D, Miller M. Firearm storage practices and risk perceptions among a nationally representative sample of U.S. veterans with and without self-harm risk factors. Suicide Life Threat Behav. 2019;49:653-664. doi:10.1111/sltb.12463
- Simonetti JA, Azrael D, Zhang W, Miller M. Receipt of clinician-delivered firearm safety counseling among U.S. veterans: results from a 2019 national survey. Suicide Life Threat Behav. 2022;52:1121-1125. doi:10.1111/sltb.12906
- US Office of the Surgeon General. The surgeon general’s call to action to implement the national strategy for suicide prevention. January 2021. Accessed February 5, 2026. https://www.hhs.gov/sites/default/files/sprc-call-to-action.pdf
- DeBeer BB, Matthieu MM, Kittel JA, et al. Quality Improvement Evaluation of the Feasibility and Acceptability of Adding a Concerned Significant Other to Safety Planning for Suicide Prevention With Veterans. J Ment Health Couns. 2019;41:4-20. doi:10.17744/mehc.41.1.02
- DeBeer BB, Matthieu MM, Degutis LC, et al. Firearms lethal means safety among veterans: attitudes toward involving a concerned significant other. J Mil Veteran Fam Health. 2025;11:23-31.
- Monteith LL, Holliday R, Dorsey Holliman BA, et al. Understanding female veterans’ experiences and perspectives of firearms. J Clin Psychol. 2020;76:1736-1753. doi:10.1002/jclp.22952
- DeBeer BB, Kimbrel NA, Meyer EC, et al. Combined PTSD and depressive symptoms interact with post-deployment social support to predict suicidal ideation in Operation Enduring Freedom and Operation Iraqi Freedom veterans. Psychiatry Res. 2014;216:357-362. doi:10.1016/j.psychres.2014.02.010
- Phillips AC, Lewis LK, McEvoy MP, et al. Development and validation of the guideline for reporting evidence-based practice educational interventions and teaching (GREET). BMC Med Educ. 2016;16:237. doi:10.1186/s12909-016-0759-1
- Miller WR, Rollnick S. Motivational Interviewing: Helping People Change. 3rd ed. Guilford Press; 2013.
- Khazanov GK, Keddem S, Hoskins K, et al. Stakeholder perceptions of lethal means safety counseling: a qualitative systematic review. Front Psychiatry. 2022;13:993415. doi:10.3389/fpsyt.2022.993415
- Stanley B, Brown GK, Karlin B, et al. US Dept of Veterans Affairs. Safety plan treatment manual to reduce suicide risk: veteran version. August 20, 2008. Accessed February 5, 2026. https://www.mentalhealth.va.gov/mentalhealth/docs/va_safety_planning_manual.doc
- Dobscha SK, Clark KD, Newell S, et al. Strategies for discussing firearms storage safety in primary care: veteran perspectives. J Gen Intern Med. 2021;36:1492-1502. doi:10.1007/s11606-020-06412-x
- Khalifian CE, Leifker FR, Knopp K, et al. Utilizing the couple relationship to prevent suicide: a preliminary examination of treatment for relationships and safety together. J Clin Psych. 2022;78:747-757. doi:10.1002/jclp.23251
- Walsh A, Friedman K, Morrissey BH, et al. Project Safe Guard: evaluating a lethal means safety intervention to reduce firearm suicide in the National Guard. Mil Med. 2024;189:510-516. doi:10.1093/milmed/usae172
- Beck AT. Beyond belief: a theory of modes, personality, and psychopathology. In: Salkovkis PM, ed. Frontiers of Cognitive Therapy. Guilford Press;1996:1-25.
- Rudd MD. The suicidal mode: a cognitive-behavioral model of suicidality. Suicide Life Threat Behav. 2000;30(1):18-33.
- US Dept of Veterans Affairs Office of Suicide Prevention. 2024 national veteran suicide prevention annual report. December 2024. Accessed February 5, 2026. https://www.mentalhealth.va.gov/docs/data-sheets/2024/2024-Annual-Report-Part-2-of-2_508.pdf
- Fischer IC, Aunon FM, Nichter B, et al. Firearm ownership among a nationally representative sample of U.S. veterans. Am J Prev Med. 2023;65:1129-1133. doi:10.1016/j.amepre.2023.06.013
- Conner A, Azrael D, Miller M. Suicide case-fatality rates in the United States, 2007-2014: a nationwide population-based study. Ann Intern Med. 2019;171(12):885-895. doi:10.7326/M19-1324
- Dempsey CL, Benedek DM, Zuromski KL, et al. Association of firearm ownership, use, accessibility, and storage practices with suicide risk among US army soldiers. JAMA Netw Open. 2019;2:e195383. doi:10.1001/jamanetworkopen.2019.5383
- Butterworth SE, Daruwala SE, Anestis MD. Firearm storage and shooting experience: factors relevant to the practical capability for suicide. J Psychiatr Res. 2018;102:52-56. doi:10.1016/j.jpsychires.2018.03.010
- Simonetti JA, Azrael D, Miller M. Firearm storage practices and risk perceptions among a nationally representative sample of U.S. veterans with and without self-harm risk factors. Suicide Life Threat Behav. 2019;49:653-664. doi:10.1111/sltb.12463
- Simonetti JA, Azrael D, Zhang W, Miller M. Receipt of clinician-delivered firearm safety counseling among U.S. veterans: results from a 2019 national survey. Suicide Life Threat Behav. 2022;52:1121-1125. doi:10.1111/sltb.12906
- US Office of the Surgeon General. The surgeon general’s call to action to implement the national strategy for suicide prevention. January 2021. Accessed February 5, 2026. https://www.hhs.gov/sites/default/files/sprc-call-to-action.pdf
- DeBeer BB, Matthieu MM, Kittel JA, et al. Quality Improvement Evaluation of the Feasibility and Acceptability of Adding a Concerned Significant Other to Safety Planning for Suicide Prevention With Veterans. J Ment Health Couns. 2019;41:4-20. doi:10.17744/mehc.41.1.02
- DeBeer BB, Matthieu MM, Degutis LC, et al. Firearms lethal means safety among veterans: attitudes toward involving a concerned significant other. J Mil Veteran Fam Health. 2025;11:23-31.
- Monteith LL, Holliday R, Dorsey Holliman BA, et al. Understanding female veterans’ experiences and perspectives of firearms. J Clin Psychol. 2020;76:1736-1753. doi:10.1002/jclp.22952
- DeBeer BB, Kimbrel NA, Meyer EC, et al. Combined PTSD and depressive symptoms interact with post-deployment social support to predict suicidal ideation in Operation Enduring Freedom and Operation Iraqi Freedom veterans. Psychiatry Res. 2014;216:357-362. doi:10.1016/j.psychres.2014.02.010
- Phillips AC, Lewis LK, McEvoy MP, et al. Development and validation of the guideline for reporting evidence-based practice educational interventions and teaching (GREET). BMC Med Educ. 2016;16:237. doi:10.1186/s12909-016-0759-1
- Miller WR, Rollnick S. Motivational Interviewing: Helping People Change. 3rd ed. Guilford Press; 2013.
- Khazanov GK, Keddem S, Hoskins K, et al. Stakeholder perceptions of lethal means safety counseling: a qualitative systematic review. Front Psychiatry. 2022;13:993415. doi:10.3389/fpsyt.2022.993415
- Stanley B, Brown GK, Karlin B, et al. US Dept of Veterans Affairs. Safety plan treatment manual to reduce suicide risk: veteran version. August 20, 2008. Accessed February 5, 2026. https://www.mentalhealth.va.gov/mentalhealth/docs/va_safety_planning_manual.doc
- Dobscha SK, Clark KD, Newell S, et al. Strategies for discussing firearms storage safety in primary care: veteran perspectives. J Gen Intern Med. 2021;36:1492-1502. doi:10.1007/s11606-020-06412-x
- Khalifian CE, Leifker FR, Knopp K, et al. Utilizing the couple relationship to prevent suicide: a preliminary examination of treatment for relationships and safety together. J Clin Psych. 2022;78:747-757. doi:10.1002/jclp.23251
- Walsh A, Friedman K, Morrissey BH, et al. Project Safe Guard: evaluating a lethal means safety intervention to reduce firearm suicide in the National Guard. Mil Med. 2024;189:510-516. doi:10.1093/milmed/usae172
- Beck AT. Beyond belief: a theory of modes, personality, and psychopathology. In: Salkovkis PM, ed. Frontiers of Cognitive Therapy. Guilford Press;1996:1-25.
- Rudd MD. The suicidal mode: a cognitive-behavioral model of suicidality. Suicide Life Threat Behav. 2000;30(1):18-33.
Involving Concerned Significant Others in Firearm Suicide Prevention: Development of the Family FireArms Secure Storage Training Intervention
Involving Concerned Significant Others in Firearm Suicide Prevention: Development of the Family FireArms Secure Storage Training Intervention
Ask the Expert Geriatric Psychiatry: A VHA Email Consultation Program to Support Clinicians
Ask the Expert Geriatric Psychiatry: A VHA Email Consultation Program to Support Clinicians
T he US Census Bureau projects that the number of older adults (aged ≥ 65 years) will exceed 49 million by 2030, and an estimated 20% (nearly 10 million) of this population will experience cognitive or mental health disorders.1,2 The mental health workforce is not equipped to address the specialized mental health care needs of many older adults.2,3 For example, geriatric psychiatrists specialize in the diagnosis and treatment of mental illness and cognitive disorders in the later stages of life, but their numbers are few and declining. Only 33.5% of geriatric psychiatry fellowship training slots were filled from 2017 to 2021, and only 62 fellows trained during the 2021-2022 academic year.4 Board-certified geriatric psychiatrists also tend to be concentrated in larger, urban, academically-affiliated medical centers, often leaving rural areas and smaller facilities without access, including facilities in the Veterans Health Administration (VHA).5
The VHA has been optimizing access to specialty geriatric mental health services via regional and national virtual consultation services. Seven of 19 Veterans Integrated Service Network (VISN) Clinical Resource Hubs (CRHs) have geriatric mental health teams.6 These provide interdisciplinary geriatric mental telehealth services, including geriatric psychiatry, for older veterans with complex care needs.7,8 Likewise, the VHA National Expert Consultation & Specialized Services-Mental Health (NEXCSS- MH, formerly known as the National Telemental Health Center) sponsors video teleconsultations with board-certified geriatric psychiatrists and an Ask the Expert email consultation program.
This article describes the Ask the Expert Geriatric Psychiatry email program (one of several similar programs at NEXCSS-MH), building upon a symposium presented at the American Association for Geriatric Psychiatry (AAGP) annual meeting in March 2022.9 The program was initiated in June 2021 as a result of discussions between the National Mental Health Director, Geriatric Mental Health in the VHA Office of Mental Health and Suicide Prevention (now known as the Office of Mental Health [OMH]), and National Telemental Health Center leadership. VHA board certified geriatric psychiatrists were recruited to serve as expert consultants and respond to email questions submitted by VHA clinicians regarding the psychiatric care of older adult veterans. The results of this program identify educational needs among clinical staff and may inform the development of program materials for a range of clinicians.
Program Description
The national geriatric mental health director recruited prospective experts and met with each to assess interest and qualifications, consulting with OMH psychiatrist leaders before making selections. Five experts were initially selected; 1 later stepped down and was replaced by another, who also stepped down. The experts were board certified in psychiatry and geriatric psychiatry and held a variety of local and national leadership positions, including geriatric psychiatry fellowship director, US Department of Veterans Affairs (VA) research and clinical leader, and various roles in the AAGP; some had received teaching awards.
Operations
The national geriatric mental health director announced the program in June 2021 to VHA mental health and geriatric program email groups with reminders sent every few months. The announcement included information about the types of questions appropriate to submit, including examples of general clinical management questions that did not share patient-specific protected health information, and clarified that experts would not be conducting chart reviews because the time required for detailed chart reviews was not feasible for volunteer experts to integrate into their otherwise full-time jobs at their respective VA medical centers. The announcement also included brief biographies of the experts.
The Figure describes the daily operations of the Ask the Expert Geriatric Psychiatry email consultation program. The NEXCSS- MH developed a Microsoft Outlook mailbox and group email address where clinicians from across the VHA could submit questions. The experts, as well as the national geriatric mental health director and NEXCSS-MH staff, had access to this mailbox to track and/or respond to questions. One expert volunteered to be the program’s primary mailbox coordinator. The coordinator checked the inbox daily and assigned each question to one of the experts on a rotating basis using the color-coding feature in Outlook. The other experts were advised to check the email account at least once weekly and reply to any assigned questions.
Responding to a question entailed first determining whether the question was appropriate for the service. For example, if a question requested a chart review, the expert replied that experts could not provide chart reviews and requested that the question be reframed. Next, the expert often needed to define a specific clinical question from the information provided, as email questions often touched upon several topics. The expert provided personalized advice on diagnostic testing, nonpharmacologic treatment strategies, and/or pharmacologic treatment options. Experts also often attached relevant guidelines or review articles. The goal was to provide a response within 7 business days.
All email responses included a disclaimer indicating that the program was not intended for urgent or immediate medical advice and that the information provided was for VHA clinician education purposes only. The disclaimer explained that email communication did not establish a doctor-patient relationship between the expert and a specific veteran and that, if desired, a request for a clinical consultation could be submitted on a specific case (ie, a video teleconsultation).
Methods for Reviewing Questions
Descriptive statistics, including frequencies, means, and minimum and maximum ranges, were used to capture the number of questions the program received, type of requester, and length of time prior to response for emailed questions.9 Conventional content analysis procedures were used between January and October 2024 to analyze clinicians’ questions.10 Four subject matter experts (3 geriatric psychiatrists and 1 geropsychologist) served as coders, assigned in groups of 2 to review questions. Each coder independently reviewed assigned questions and identified preliminary themes. Themes were reviewed and revised using an iterative process during regular team meetings with coders to clarify and confirm interpretations. Discrepancies were discussed within team meetings to achieve consensus.
Questions received. Between February 2022 and December 2023, the program received 101 email questions. Requesters included 39 physicians, 17 nurse practitioners or physician assistants, 15 social workers, 14 psychologists, 9 nurses, 5 pharmacists, 1 dietitian, and 1 who was undetermined. Experts responded to the questions an average of 6 days after receipt (range, < 1-19); 73 responses (72%) met the 7-day goal.
Iterative changes to coded themes were made during group discussions. Multiple clinical questions were often posed within the same email. Initially, some coders identified themes solely based on reported symptoms; others identified themes based on reported and/or potential diagnostic conditions attributed to the symptom(s) described within the email. For example, some coders selected a primary theme of behavioral and psychological symptoms of dementia (BPSD) only if a behavior contributing to distress in the veteran or others was described, while others selected this theme when any psychiatric symptom (eg, psychosis) was present in the context of dementia. The group identified 1 primary theme per question based on the main clinical symptom or main concern presented. Co-occurring diagnostic conditions highlighted in the email requests were included as secondary themes, and each question could have > 1 secondary theme.
The most frequent requests related to clinical symptoms included questions about agitated behaviors, sleep and/or nightmares, and depression symptoms (Table 1). Twenty-seven of 33 email requests on agitated behaviors were related to a dementia diagnosis, as were several questions about sleep/nightmares, depression, psychosis/mania, and anxiety. Many diagnostic conditions were described in the email requests (Table 2). The most frequent condition was dementia, followed by a medical condition, depressive disorder, posttraumatic stress disorder, and/or serious mental illness.
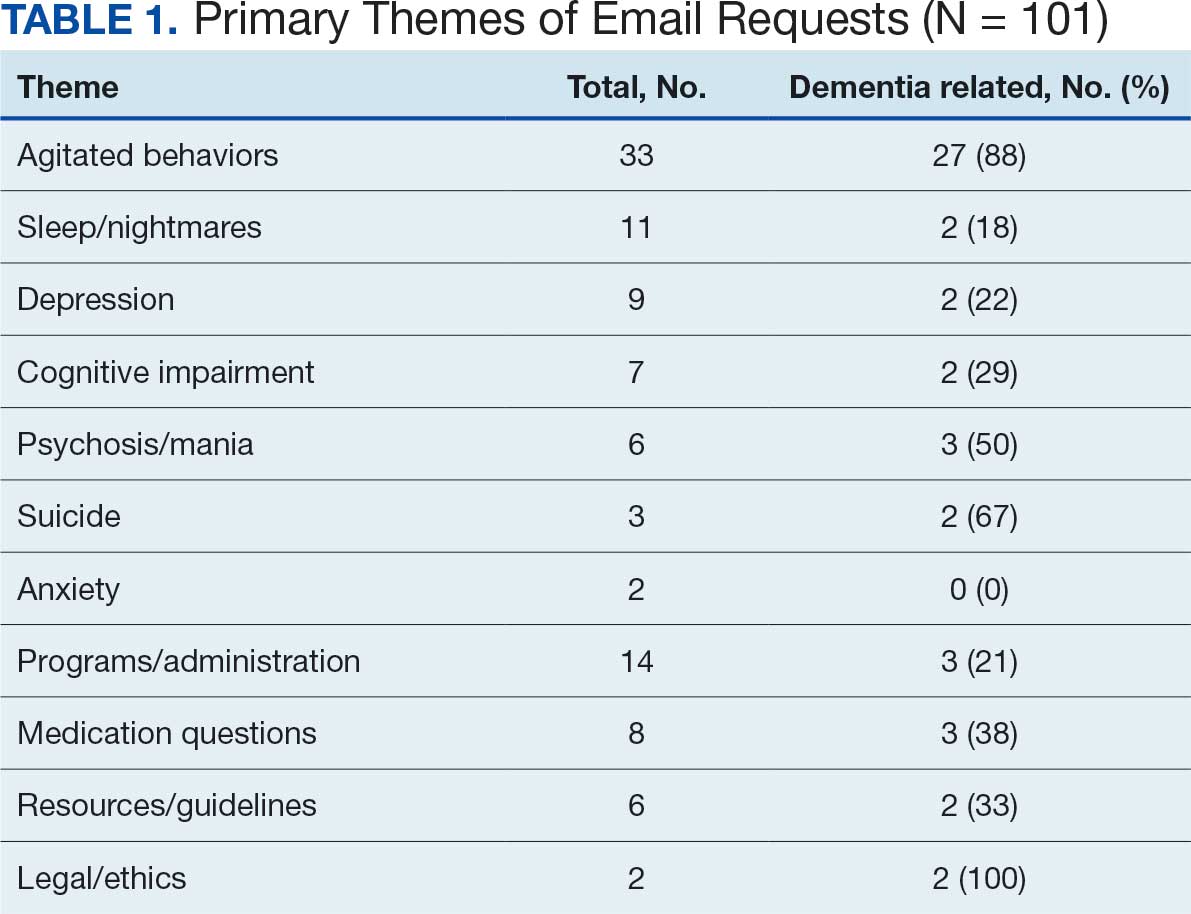

Request for Feedback. In February 2022, an email request was sent to the 64 clinicians who asked email questions from the start of the program in June 2021 through December 2021. A second request included 11 clinicians who asked questions from January through February 2022. These requests were sent as part of preparations for the symposium on the program presented at the AAGP annual meeting in March 2022.9 In May 2024, feedback was requested from 37 clinicians who submitted questions from May 1, 2023, through May 15, 2024.
Requests for feedback included 6 closed-ended and 1 open-ended question: (1) Did the answer you received help inform clinical practice? (2) Did you receive a timely response? (3) What type of information was useful to you in addressing your question (ie, direct/specific answer to a clinical scenario, guidelines, articles, VA resources)? (4) Do you have access to a geriatric psychiatrist at your facility? (5) Are you likely to use Ask the Expert Geriatric Psychiatry in the future? (6) Would you use a geriatric psychiatry teleconsultation service? (7) Share suggestions for improvement. Frequencies of response selection were obtained for each question. Text responses to the open-ended question asking for suggestions for improvement were reviewed and summarized.
Responses
Thirty users responded to the feedback request (27% response rate). Respondents considered the answers received extremely (n = 14; 47%) or very much (n = 12; 40%) helpful for their clinical practice. Twenty-three respondents (77%) felt an answer was provided promptly, 7 respondents (23%) felt the answer was not timely but still useful, and none felt that the answer was too late. Respondents reported that the most useful type of information in addressing their questions was a direct/specific answer to a clinical scenario (n = 27; 90%), followed by guidelines (n = 12; 40%), articles (n = 7; 23%), and VA resources (n = 4; 13%).
Sixteen respondents (53%) reported that they rarely had ready access to a geriatric psychiatrist at their facility, 3 (10%) had access sometimes, 4 (14%) had access usually, 3 (10%) had access regularly, and 3 (10%) never had access. Twenty-seven respondents (90%) indicated they would be very likely to use the service again. If geriatric psychiatry teleconsultation and/or e-consultation were offered, many respondents indicated they would be extremely (n = 10; 33%) or very (n = 12; 40%) likely to use teleconsultation and/or e-consultation.
Suggestions for improvement included supporting experts to perform chart reviews for email questions, developing a template or consult form, holding a biweekly drop-in meeting to present questions to and discuss cases with a panel of experts, and providing further help addressing complex decisional capacity issues, delirium, and care or placement for veterans with severe behavioral issues in a rural setting.
Discussion
Although many older adults experience cognitive and mental health disorders that may benefit from management by a geriatric psychiatrist, the number of trained geriatric psychiatrists available is insufficient to allow for direct care for each patient. The Ask the Expert Geriatric Psychiatry email consultation program is one aspect of a multicomponent strategy within the VHA to increase access to specialty geriatric mental health services for veterans. A key advantage of the program is that it is not resource intensive. Experts can participate voluntarily, providing timely feedback to clinicians around the country while continuing other duties at their respective VA medical centers. Email replies to the experts’ answers elicited positive feedback on the program, include: “I found this service to be extremely helpful and I have shared the information they sent me with several other coworkers!”, “It was great!”, and “I endorsed the service to our VISN Rehabilitation and Extended Care group.”
The coding of primary and secondary themes from 101 email questions that were retained revealed the range and relative frequencies of clinical and administrative topics with which clinicians needed help. The most common (33%) theme was agitated behaviors. Nearly half of the questions (48%) were related to underlying dementia, and 29% were related to a patient’s medical comorbidities. These findings suggest that the expertise of a geriatric psychiatrist is particularly relevant when caring for older patients experiencing BPSD or patients with complex, overlapping psychiatric and medical conditions.
Despite a 27% response rate, participant feedback has been helpful. The program reached its intended audience of clinicians in rural areas and at smaller facilities with 53% of requesters reporting they rarely had access to a geriatric psychiatrist. Suggestions for improvement indicated that some clinicians desired additional support, including chart reviews, meetings with experts, and a video teleconsultation service (available through NEXCSS-MH).
Many clinicians without training in specialty geriatric mental health may require help with complex clinical presentations. For example, 39 clinicians who submitted questions to the program were physicians. Accreditation Council for Graduate Medical Education program requirements for general psychiatry residency include 4 weeks of geriatric psychiatry.11 The findings of this study suggest that this level of training may not be adequate to independently care for every patient who experiences dementia or multimorbidity. Several training and mentoring initiatives have been developed to address the professional development need for psychiatrists.12-14
The need for geriatric workforce development is significant across health care, including other mental health professions.15,16 The VHA Geriatric Scholars program trains rural primary care practitioners, psychologists, and psychiatrists.17,18 Likewise, consultative geriatric specialty support for primary care practitioners in rural areas is provided via the Geriatric Research Education and Clinical Center Connect program.19 The Ask the Expert Geriatric Psychiatry email program is an additional economical model to support clinician educational development and provide rapid educational responses to inform patient care.
Ask the Expert received fewer email questions than anticipated. Enhanced optimization may require more frequent and widespread announcements about the program. Clinical staff may not be aware of the program due to an overload of email communications. Likewise, it may be challenging for busy clinicians to take the time to seek consultation or recognize a potential gap in their knowledge or skills. Had more questions been submitted, the 5 volunteer experts may have had more difficulty addressing the demand. Feedback from this project may inform development of a frequently asked questions document to share with VHA teams and a drop-in office hour to pose clinical questions of geriatric psychiatry experts, as recommended by a clinician who participated in the program.
Limitations
Not all requesters were sent a request for feedback, and the response rate for the request for feedback was only 27%. As the program has evolved, it began sending a request for feedback immediately after answering each question, which may increase the odds of response. The goal of experts answering questions within 7 business days was met 72% of the time, likely an artifact of experts integrating question answering with many other duties. The mailbox coordinator has since provided email prompts to experts immediately upon being assigned a question with the goal of improving timeliness. The program did not include chart reviews or patient consultations, as neither was feasible for volunteer experts. The email consultation service is a single component of virtual consultative specialty geriatric mental health services within the VHA, including video consultations via NEXCSS-MH and regional geriatric mental health teams.
Conclusions
The need for specialty geriatric mental health services is increasing in the VHA and across the US. However, there are too few board-certified geriatric psychiatrists to provide direct patient care to all older adults with cognitive and mental health disorders. The VHA has leveraged telehealth to improve access to geriatric mental health care. The VHA Ask the Expert Geriatric Psychiatry email consultation program is a low-resource service which provides rapid feedback to clinicians nationwide on challenging clinical scenarios, many of which are dementia-related. Most users of the service who responded to requests for feedback reported that answers to their questions were helpful and timely. The email consultation program should continue to be supplemented by more comprehensive geriatric telemental health services for particularly complex cases to meet the needs of older veterans.
- 2023 population projections for the nation by age, sex, race, Hispanic origin and nativity. United States Census Bureau. November 9, 2023. Accessed December 11, 2025. https://www.census.gov/newsroom/press-kits/2023/population-projections.html
- National Academies of Sciences Engineering and Medicine. Addressing the rising mental health needs of an aging population: proceedings of a workshop. 2024. Accessed December 11, 2025. doi.org:10.17226/27340
- Institute of Medicine. The mental health and substance use workforce for older adults: in whose hands? The National Academies Press; 2012. Accessed December 11, 2025. doi:10.17226/13400
- American Psychiatric Association. 2022 resident/fellow census. November 2023. Accessed December 11, 2025. https://www.psychiatry.org/getmedia/d80438af-f760-40f3-9d33-f91309b09564/APA-Resident-Census-2022.pdf
- Juul D, Colenda CC, Lyness JM, et al. Subspecialty training and certification in geriatric psychiatry: a 25-year overview. Am J Geriatr Psychiatry. 2017;25:445-453. doi:10.1016/j.jagp.2016.12.018
- Jaske E, Wheat CL, Rubenstein LV, et al. Understanding how contingency staffing programs can support mental health services in the Veterans Health Administration. Telemed J E Health. 2024;30:1857-1865. doi:10.1089/tmj.2023.0573
- Gould CE, Carlson C, Alfaro AJ, et al. Supporting veterans, caregivers, and providers in rural regions with tele-geriatric psychiatry consultation: a mixed methods pilot study. Am J Geriatr Psychiatry. 2023;31:279-290. doi:10.1016/j.jagp.2023.01.005
- Gould CE, Paiko L, Carlson C, et al. Implementation of tele-geriatricmental healthcare for rural veterans: factors influencing care models. Front Health Serv. 2024;4:1221899. doi:10.3389/frhs.2024.1221899
- Padala P, Schultz S, Khatkhate G, et al. Ask the expert geriatric psychiatry: VA program to support clinicians. Am J Geriatr Psychiatry. 2022;30:S18. doi:10.1016/j.jagp.2022.01.279
- Hsieh HF, Shannon SE. Three approaches to qualitative content analysis. Qual Health Res. 2005;15:1277-1288. doi:10.1177/1049732305276687
- Accreditation Council for Graduate Medical Education. Program requirements for graduate medical education in psychiatry. Revised September 3, 2025. Accessed December 11, 2025. https://www.acgme.org/globalassets/pfassets/programrequirements/2025-reformatted-requirements/400_psychiatry_2025_reformatted.pdf
- Fernandez J, Agarwal KS, Amspoker AB, et al. Outcomes from an interprofessional, dementia-focused, telementoring program: a brief report. Gerontol Geriatr Educ. 2024;45:601-606. doi:10.1080/02701960.2023.2253175
- Conroy ML, Garcia-Pittman EC, van Dyck LI, et al. The COVID-19 American Association for Geriatric Psychiatry (AAGP) online trainee curriculum: program evaluation and future directions. Am J Geriatr Psychiatry. 2025;33:308-314. doi:10.1016/j.jagp.2024.10.010
- Conroy ML, Garcia-Pittman EC, Ali H, et al. The COVID-19 AAGP online trainee curriculum: development and method of initial evaluation. Am J Geriatr Psychiatry. 2020;28:1004-1008. doi:10.1016/j.jagp.2020.06.003
- Flaherty E, Busby-Whitehead J, Potter J, et al. The geriatric workforce enhancement program: review of the coordinating center and examples of the GWEP in practice. Am J Geriatr Psychiatry. 2019;27:675-686. doi:10.1016/j.jagp.2019.04.010
- Hoge MA, Karel MJ, Zeiss AM, et al. Strengthening psychology’s workforce for older adults: implications of the Institute of Medicine’s report to Congress. Am Psychol. 2015;70:265-278. doi:10.1037/a0038927
- Kramer BJ, Creekmur B, Howe JL, et al. Veterans Affairs geriatric scholars program: enhancing existing primary care clinician skills in caring for older veterans. J Am Geriatr Soc. 2016;64:2343-2348. doi:10.1111/jgs.14382
- Gould CE, Rodriguez RL, Gregg JJ, et al. Preparing Veterans Health Administration psychologists to meet the complex needs of aging veterans. Fed Pract. 2024;41:S10-S15. doi:10.12788/fp.0466
- Pimentel CB, Gately M, Barczi SR, et al. GRECC Connect: Geriatrics telehealth to empower health care providers and improve management of older veterans in rural communities. Fed Pract. 2019;36:464-470.
T he US Census Bureau projects that the number of older adults (aged ≥ 65 years) will exceed 49 million by 2030, and an estimated 20% (nearly 10 million) of this population will experience cognitive or mental health disorders.1,2 The mental health workforce is not equipped to address the specialized mental health care needs of many older adults.2,3 For example, geriatric psychiatrists specialize in the diagnosis and treatment of mental illness and cognitive disorders in the later stages of life, but their numbers are few and declining. Only 33.5% of geriatric psychiatry fellowship training slots were filled from 2017 to 2021, and only 62 fellows trained during the 2021-2022 academic year.4 Board-certified geriatric psychiatrists also tend to be concentrated in larger, urban, academically-affiliated medical centers, often leaving rural areas and smaller facilities without access, including facilities in the Veterans Health Administration (VHA).5
The VHA has been optimizing access to specialty geriatric mental health services via regional and national virtual consultation services. Seven of 19 Veterans Integrated Service Network (VISN) Clinical Resource Hubs (CRHs) have geriatric mental health teams.6 These provide interdisciplinary geriatric mental telehealth services, including geriatric psychiatry, for older veterans with complex care needs.7,8 Likewise, the VHA National Expert Consultation & Specialized Services-Mental Health (NEXCSS- MH, formerly known as the National Telemental Health Center) sponsors video teleconsultations with board-certified geriatric psychiatrists and an Ask the Expert email consultation program.
This article describes the Ask the Expert Geriatric Psychiatry email program (one of several similar programs at NEXCSS-MH), building upon a symposium presented at the American Association for Geriatric Psychiatry (AAGP) annual meeting in March 2022.9 The program was initiated in June 2021 as a result of discussions between the National Mental Health Director, Geriatric Mental Health in the VHA Office of Mental Health and Suicide Prevention (now known as the Office of Mental Health [OMH]), and National Telemental Health Center leadership. VHA board certified geriatric psychiatrists were recruited to serve as expert consultants and respond to email questions submitted by VHA clinicians regarding the psychiatric care of older adult veterans. The results of this program identify educational needs among clinical staff and may inform the development of program materials for a range of clinicians.
Program Description
The national geriatric mental health director recruited prospective experts and met with each to assess interest and qualifications, consulting with OMH psychiatrist leaders before making selections. Five experts were initially selected; 1 later stepped down and was replaced by another, who also stepped down. The experts were board certified in psychiatry and geriatric psychiatry and held a variety of local and national leadership positions, including geriatric psychiatry fellowship director, US Department of Veterans Affairs (VA) research and clinical leader, and various roles in the AAGP; some had received teaching awards.
Operations
The national geriatric mental health director announced the program in June 2021 to VHA mental health and geriatric program email groups with reminders sent every few months. The announcement included information about the types of questions appropriate to submit, including examples of general clinical management questions that did not share patient-specific protected health information, and clarified that experts would not be conducting chart reviews because the time required for detailed chart reviews was not feasible for volunteer experts to integrate into their otherwise full-time jobs at their respective VA medical centers. The announcement also included brief biographies of the experts.
The Figure describes the daily operations of the Ask the Expert Geriatric Psychiatry email consultation program. The NEXCSS- MH developed a Microsoft Outlook mailbox and group email address where clinicians from across the VHA could submit questions. The experts, as well as the national geriatric mental health director and NEXCSS-MH staff, had access to this mailbox to track and/or respond to questions. One expert volunteered to be the program’s primary mailbox coordinator. The coordinator checked the inbox daily and assigned each question to one of the experts on a rotating basis using the color-coding feature in Outlook. The other experts were advised to check the email account at least once weekly and reply to any assigned questions.

Responding to a question entailed first determining whether the question was appropriate for the service. For example, if a question requested a chart review, the expert replied that experts could not provide chart reviews and requested that the question be reframed. Next, the expert often needed to define a specific clinical question from the information provided, as email questions often touched upon several topics. The expert provided personalized advice on diagnostic testing, nonpharmacologic treatment strategies, and/or pharmacologic treatment options. Experts also often attached relevant guidelines or review articles. The goal was to provide a response within 7 business days.
All email responses included a disclaimer indicating that the program was not intended for urgent or immediate medical advice and that the information provided was for VHA clinician education purposes only. The disclaimer explained that email communication did not establish a doctor-patient relationship between the expert and a specific veteran and that, if desired, a request for a clinical consultation could be submitted on a specific case (ie, a video teleconsultation).
Methods for Reviewing Questions
Descriptive statistics, including frequencies, means, and minimum and maximum ranges, were used to capture the number of questions the program received, type of requester, and length of time prior to response for emailed questions.9 Conventional content analysis procedures were used between January and October 2024 to analyze clinicians’ questions.10 Four subject matter experts (3 geriatric psychiatrists and 1 geropsychologist) served as coders, assigned in groups of 2 to review questions. Each coder independently reviewed assigned questions and identified preliminary themes. Themes were reviewed and revised using an iterative process during regular team meetings with coders to clarify and confirm interpretations. Discrepancies were discussed within team meetings to achieve consensus.
Questions received. Between February 2022 and December 2023, the program received 101 email questions. Requesters included 39 physicians, 17 nurse practitioners or physician assistants, 15 social workers, 14 psychologists, 9 nurses, 5 pharmacists, 1 dietitian, and 1 who was undetermined. Experts responded to the questions an average of 6 days after receipt (range, < 1-19); 73 responses (72%) met the 7-day goal.
Iterative changes to coded themes were made during group discussions. Multiple clinical questions were often posed within the same email. Initially, some coders identified themes solely based on reported symptoms; others identified themes based on reported and/or potential diagnostic conditions attributed to the symptom(s) described within the email. For example, some coders selected a primary theme of behavioral and psychological symptoms of dementia (BPSD) only if a behavior contributing to distress in the veteran or others was described, while others selected this theme when any psychiatric symptom (eg, psychosis) was present in the context of dementia. The group identified 1 primary theme per question based on the main clinical symptom or main concern presented. Co-occurring diagnostic conditions highlighted in the email requests were included as secondary themes, and each question could have > 1 secondary theme.
The most frequent requests related to clinical symptoms included questions about agitated behaviors, sleep and/or nightmares, and depression symptoms (Table 1). Twenty-seven of 33 email requests on agitated behaviors were related to a dementia diagnosis, as were several questions about sleep/nightmares, depression, psychosis/mania, and anxiety. Many diagnostic conditions were described in the email requests (Table 2). The most frequent condition was dementia, followed by a medical condition, depressive disorder, posttraumatic stress disorder, and/or serious mental illness.


Request for Feedback. In February 2022, an email request was sent to the 64 clinicians who asked email questions from the start of the program in June 2021 through December 2021. A second request included 11 clinicians who asked questions from January through February 2022. These requests were sent as part of preparations for the symposium on the program presented at the AAGP annual meeting in March 2022.9 In May 2024, feedback was requested from 37 clinicians who submitted questions from May 1, 2023, through May 15, 2024.
Requests for feedback included 6 closed-ended and 1 open-ended question: (1) Did the answer you received help inform clinical practice? (2) Did you receive a timely response? (3) What type of information was useful to you in addressing your question (ie, direct/specific answer to a clinical scenario, guidelines, articles, VA resources)? (4) Do you have access to a geriatric psychiatrist at your facility? (5) Are you likely to use Ask the Expert Geriatric Psychiatry in the future? (6) Would you use a geriatric psychiatry teleconsultation service? (7) Share suggestions for improvement. Frequencies of response selection were obtained for each question. Text responses to the open-ended question asking for suggestions for improvement were reviewed and summarized.
Responses
Thirty users responded to the feedback request (27% response rate). Respondents considered the answers received extremely (n = 14; 47%) or very much (n = 12; 40%) helpful for their clinical practice. Twenty-three respondents (77%) felt an answer was provided promptly, 7 respondents (23%) felt the answer was not timely but still useful, and none felt that the answer was too late. Respondents reported that the most useful type of information in addressing their questions was a direct/specific answer to a clinical scenario (n = 27; 90%), followed by guidelines (n = 12; 40%), articles (n = 7; 23%), and VA resources (n = 4; 13%).
Sixteen respondents (53%) reported that they rarely had ready access to a geriatric psychiatrist at their facility, 3 (10%) had access sometimes, 4 (14%) had access usually, 3 (10%) had access regularly, and 3 (10%) never had access. Twenty-seven respondents (90%) indicated they would be very likely to use the service again. If geriatric psychiatry teleconsultation and/or e-consultation were offered, many respondents indicated they would be extremely (n = 10; 33%) or very (n = 12; 40%) likely to use teleconsultation and/or e-consultation.
Suggestions for improvement included supporting experts to perform chart reviews for email questions, developing a template or consult form, holding a biweekly drop-in meeting to present questions to and discuss cases with a panel of experts, and providing further help addressing complex decisional capacity issues, delirium, and care or placement for veterans with severe behavioral issues in a rural setting.
Discussion
Although many older adults experience cognitive and mental health disorders that may benefit from management by a geriatric psychiatrist, the number of trained geriatric psychiatrists available is insufficient to allow for direct care for each patient. The Ask the Expert Geriatric Psychiatry email consultation program is one aspect of a multicomponent strategy within the VHA to increase access to specialty geriatric mental health services for veterans. A key advantage of the program is that it is not resource intensive. Experts can participate voluntarily, providing timely feedback to clinicians around the country while continuing other duties at their respective VA medical centers. Email replies to the experts’ answers elicited positive feedback on the program, include: “I found this service to be extremely helpful and I have shared the information they sent me with several other coworkers!”, “It was great!”, and “I endorsed the service to our VISN Rehabilitation and Extended Care group.”
The coding of primary and secondary themes from 101 email questions that were retained revealed the range and relative frequencies of clinical and administrative topics with which clinicians needed help. The most common (33%) theme was agitated behaviors. Nearly half of the questions (48%) were related to underlying dementia, and 29% were related to a patient’s medical comorbidities. These findings suggest that the expertise of a geriatric psychiatrist is particularly relevant when caring for older patients experiencing BPSD or patients with complex, overlapping psychiatric and medical conditions.
Despite a 27% response rate, participant feedback has been helpful. The program reached its intended audience of clinicians in rural areas and at smaller facilities with 53% of requesters reporting they rarely had access to a geriatric psychiatrist. Suggestions for improvement indicated that some clinicians desired additional support, including chart reviews, meetings with experts, and a video teleconsultation service (available through NEXCSS-MH).
Many clinicians without training in specialty geriatric mental health may require help with complex clinical presentations. For example, 39 clinicians who submitted questions to the program were physicians. Accreditation Council for Graduate Medical Education program requirements for general psychiatry residency include 4 weeks of geriatric psychiatry.11 The findings of this study suggest that this level of training may not be adequate to independently care for every patient who experiences dementia or multimorbidity. Several training and mentoring initiatives have been developed to address the professional development need for psychiatrists.12-14
The need for geriatric workforce development is significant across health care, including other mental health professions.15,16 The VHA Geriatric Scholars program trains rural primary care practitioners, psychologists, and psychiatrists.17,18 Likewise, consultative geriatric specialty support for primary care practitioners in rural areas is provided via the Geriatric Research Education and Clinical Center Connect program.19 The Ask the Expert Geriatric Psychiatry email program is an additional economical model to support clinician educational development and provide rapid educational responses to inform patient care.
Ask the Expert received fewer email questions than anticipated. Enhanced optimization may require more frequent and widespread announcements about the program. Clinical staff may not be aware of the program due to an overload of email communications. Likewise, it may be challenging for busy clinicians to take the time to seek consultation or recognize a potential gap in their knowledge or skills. Had more questions been submitted, the 5 volunteer experts may have had more difficulty addressing the demand. Feedback from this project may inform development of a frequently asked questions document to share with VHA teams and a drop-in office hour to pose clinical questions of geriatric psychiatry experts, as recommended by a clinician who participated in the program.
Limitations
Not all requesters were sent a request for feedback, and the response rate for the request for feedback was only 27%. As the program has evolved, it began sending a request for feedback immediately after answering each question, which may increase the odds of response. The goal of experts answering questions within 7 business days was met 72% of the time, likely an artifact of experts integrating question answering with many other duties. The mailbox coordinator has since provided email prompts to experts immediately upon being assigned a question with the goal of improving timeliness. The program did not include chart reviews or patient consultations, as neither was feasible for volunteer experts. The email consultation service is a single component of virtual consultative specialty geriatric mental health services within the VHA, including video consultations via NEXCSS-MH and regional geriatric mental health teams.
Conclusions
The need for specialty geriatric mental health services is increasing in the VHA and across the US. However, there are too few board-certified geriatric psychiatrists to provide direct patient care to all older adults with cognitive and mental health disorders. The VHA has leveraged telehealth to improve access to geriatric mental health care. The VHA Ask the Expert Geriatric Psychiatry email consultation program is a low-resource service which provides rapid feedback to clinicians nationwide on challenging clinical scenarios, many of which are dementia-related. Most users of the service who responded to requests for feedback reported that answers to their questions were helpful and timely. The email consultation program should continue to be supplemented by more comprehensive geriatric telemental health services for particularly complex cases to meet the needs of older veterans.
T he US Census Bureau projects that the number of older adults (aged ≥ 65 years) will exceed 49 million by 2030, and an estimated 20% (nearly 10 million) of this population will experience cognitive or mental health disorders.1,2 The mental health workforce is not equipped to address the specialized mental health care needs of many older adults.2,3 For example, geriatric psychiatrists specialize in the diagnosis and treatment of mental illness and cognitive disorders in the later stages of life, but their numbers are few and declining. Only 33.5% of geriatric psychiatry fellowship training slots were filled from 2017 to 2021, and only 62 fellows trained during the 2021-2022 academic year.4 Board-certified geriatric psychiatrists also tend to be concentrated in larger, urban, academically-affiliated medical centers, often leaving rural areas and smaller facilities without access, including facilities in the Veterans Health Administration (VHA).5
The VHA has been optimizing access to specialty geriatric mental health services via regional and national virtual consultation services. Seven of 19 Veterans Integrated Service Network (VISN) Clinical Resource Hubs (CRHs) have geriatric mental health teams.6 These provide interdisciplinary geriatric mental telehealth services, including geriatric psychiatry, for older veterans with complex care needs.7,8 Likewise, the VHA National Expert Consultation & Specialized Services-Mental Health (NEXCSS- MH, formerly known as the National Telemental Health Center) sponsors video teleconsultations with board-certified geriatric psychiatrists and an Ask the Expert email consultation program.
This article describes the Ask the Expert Geriatric Psychiatry email program (one of several similar programs at NEXCSS-MH), building upon a symposium presented at the American Association for Geriatric Psychiatry (AAGP) annual meeting in March 2022.9 The program was initiated in June 2021 as a result of discussions between the National Mental Health Director, Geriatric Mental Health in the VHA Office of Mental Health and Suicide Prevention (now known as the Office of Mental Health [OMH]), and National Telemental Health Center leadership. VHA board certified geriatric psychiatrists were recruited to serve as expert consultants and respond to email questions submitted by VHA clinicians regarding the psychiatric care of older adult veterans. The results of this program identify educational needs among clinical staff and may inform the development of program materials for a range of clinicians.
Program Description
The national geriatric mental health director recruited prospective experts and met with each to assess interest and qualifications, consulting with OMH psychiatrist leaders before making selections. Five experts were initially selected; 1 later stepped down and was replaced by another, who also stepped down. The experts were board certified in psychiatry and geriatric psychiatry and held a variety of local and national leadership positions, including geriatric psychiatry fellowship director, US Department of Veterans Affairs (VA) research and clinical leader, and various roles in the AAGP; some had received teaching awards.
Operations
The national geriatric mental health director announced the program in June 2021 to VHA mental health and geriatric program email groups with reminders sent every few months. The announcement included information about the types of questions appropriate to submit, including examples of general clinical management questions that did not share patient-specific protected health information, and clarified that experts would not be conducting chart reviews because the time required for detailed chart reviews was not feasible for volunteer experts to integrate into their otherwise full-time jobs at their respective VA medical centers. The announcement also included brief biographies of the experts.
The Figure describes the daily operations of the Ask the Expert Geriatric Psychiatry email consultation program. The NEXCSS- MH developed a Microsoft Outlook mailbox and group email address where clinicians from across the VHA could submit questions. The experts, as well as the national geriatric mental health director and NEXCSS-MH staff, had access to this mailbox to track and/or respond to questions. One expert volunteered to be the program’s primary mailbox coordinator. The coordinator checked the inbox daily and assigned each question to one of the experts on a rotating basis using the color-coding feature in Outlook. The other experts were advised to check the email account at least once weekly and reply to any assigned questions.

Responding to a question entailed first determining whether the question was appropriate for the service. For example, if a question requested a chart review, the expert replied that experts could not provide chart reviews and requested that the question be reframed. Next, the expert often needed to define a specific clinical question from the information provided, as email questions often touched upon several topics. The expert provided personalized advice on diagnostic testing, nonpharmacologic treatment strategies, and/or pharmacologic treatment options. Experts also often attached relevant guidelines or review articles. The goal was to provide a response within 7 business days.
All email responses included a disclaimer indicating that the program was not intended for urgent or immediate medical advice and that the information provided was for VHA clinician education purposes only. The disclaimer explained that email communication did not establish a doctor-patient relationship between the expert and a specific veteran and that, if desired, a request for a clinical consultation could be submitted on a specific case (ie, a video teleconsultation).
Methods for Reviewing Questions
Descriptive statistics, including frequencies, means, and minimum and maximum ranges, were used to capture the number of questions the program received, type of requester, and length of time prior to response for emailed questions.9 Conventional content analysis procedures were used between January and October 2024 to analyze clinicians’ questions.10 Four subject matter experts (3 geriatric psychiatrists and 1 geropsychologist) served as coders, assigned in groups of 2 to review questions. Each coder independently reviewed assigned questions and identified preliminary themes. Themes were reviewed and revised using an iterative process during regular team meetings with coders to clarify and confirm interpretations. Discrepancies were discussed within team meetings to achieve consensus.
Questions received. Between February 2022 and December 2023, the program received 101 email questions. Requesters included 39 physicians, 17 nurse practitioners or physician assistants, 15 social workers, 14 psychologists, 9 nurses, 5 pharmacists, 1 dietitian, and 1 who was undetermined. Experts responded to the questions an average of 6 days after receipt (range, < 1-19); 73 responses (72%) met the 7-day goal.
Iterative changes to coded themes were made during group discussions. Multiple clinical questions were often posed within the same email. Initially, some coders identified themes solely based on reported symptoms; others identified themes based on reported and/or potential diagnostic conditions attributed to the symptom(s) described within the email. For example, some coders selected a primary theme of behavioral and psychological symptoms of dementia (BPSD) only if a behavior contributing to distress in the veteran or others was described, while others selected this theme when any psychiatric symptom (eg, psychosis) was present in the context of dementia. The group identified 1 primary theme per question based on the main clinical symptom or main concern presented. Co-occurring diagnostic conditions highlighted in the email requests were included as secondary themes, and each question could have > 1 secondary theme.
The most frequent requests related to clinical symptoms included questions about agitated behaviors, sleep and/or nightmares, and depression symptoms (Table 1). Twenty-seven of 33 email requests on agitated behaviors were related to a dementia diagnosis, as were several questions about sleep/nightmares, depression, psychosis/mania, and anxiety. Many diagnostic conditions were described in the email requests (Table 2). The most frequent condition was dementia, followed by a medical condition, depressive disorder, posttraumatic stress disorder, and/or serious mental illness.


Request for Feedback. In February 2022, an email request was sent to the 64 clinicians who asked email questions from the start of the program in June 2021 through December 2021. A second request included 11 clinicians who asked questions from January through February 2022. These requests were sent as part of preparations for the symposium on the program presented at the AAGP annual meeting in March 2022.9 In May 2024, feedback was requested from 37 clinicians who submitted questions from May 1, 2023, through May 15, 2024.
Requests for feedback included 6 closed-ended and 1 open-ended question: (1) Did the answer you received help inform clinical practice? (2) Did you receive a timely response? (3) What type of information was useful to you in addressing your question (ie, direct/specific answer to a clinical scenario, guidelines, articles, VA resources)? (4) Do you have access to a geriatric psychiatrist at your facility? (5) Are you likely to use Ask the Expert Geriatric Psychiatry in the future? (6) Would you use a geriatric psychiatry teleconsultation service? (7) Share suggestions for improvement. Frequencies of response selection were obtained for each question. Text responses to the open-ended question asking for suggestions for improvement were reviewed and summarized.
Responses
Thirty users responded to the feedback request (27% response rate). Respondents considered the answers received extremely (n = 14; 47%) or very much (n = 12; 40%) helpful for their clinical practice. Twenty-three respondents (77%) felt an answer was provided promptly, 7 respondents (23%) felt the answer was not timely but still useful, and none felt that the answer was too late. Respondents reported that the most useful type of information in addressing their questions was a direct/specific answer to a clinical scenario (n = 27; 90%), followed by guidelines (n = 12; 40%), articles (n = 7; 23%), and VA resources (n = 4; 13%).
Sixteen respondents (53%) reported that they rarely had ready access to a geriatric psychiatrist at their facility, 3 (10%) had access sometimes, 4 (14%) had access usually, 3 (10%) had access regularly, and 3 (10%) never had access. Twenty-seven respondents (90%) indicated they would be very likely to use the service again. If geriatric psychiatry teleconsultation and/or e-consultation were offered, many respondents indicated they would be extremely (n = 10; 33%) or very (n = 12; 40%) likely to use teleconsultation and/or e-consultation.
Suggestions for improvement included supporting experts to perform chart reviews for email questions, developing a template or consult form, holding a biweekly drop-in meeting to present questions to and discuss cases with a panel of experts, and providing further help addressing complex decisional capacity issues, delirium, and care or placement for veterans with severe behavioral issues in a rural setting.
Discussion
Although many older adults experience cognitive and mental health disorders that may benefit from management by a geriatric psychiatrist, the number of trained geriatric psychiatrists available is insufficient to allow for direct care for each patient. The Ask the Expert Geriatric Psychiatry email consultation program is one aspect of a multicomponent strategy within the VHA to increase access to specialty geriatric mental health services for veterans. A key advantage of the program is that it is not resource intensive. Experts can participate voluntarily, providing timely feedback to clinicians around the country while continuing other duties at their respective VA medical centers. Email replies to the experts’ answers elicited positive feedback on the program, include: “I found this service to be extremely helpful and I have shared the information they sent me with several other coworkers!”, “It was great!”, and “I endorsed the service to our VISN Rehabilitation and Extended Care group.”
The coding of primary and secondary themes from 101 email questions that were retained revealed the range and relative frequencies of clinical and administrative topics with which clinicians needed help. The most common (33%) theme was agitated behaviors. Nearly half of the questions (48%) were related to underlying dementia, and 29% were related to a patient’s medical comorbidities. These findings suggest that the expertise of a geriatric psychiatrist is particularly relevant when caring for older patients experiencing BPSD or patients with complex, overlapping psychiatric and medical conditions.
Despite a 27% response rate, participant feedback has been helpful. The program reached its intended audience of clinicians in rural areas and at smaller facilities with 53% of requesters reporting they rarely had access to a geriatric psychiatrist. Suggestions for improvement indicated that some clinicians desired additional support, including chart reviews, meetings with experts, and a video teleconsultation service (available through NEXCSS-MH).
Many clinicians without training in specialty geriatric mental health may require help with complex clinical presentations. For example, 39 clinicians who submitted questions to the program were physicians. Accreditation Council for Graduate Medical Education program requirements for general psychiatry residency include 4 weeks of geriatric psychiatry.11 The findings of this study suggest that this level of training may not be adequate to independently care for every patient who experiences dementia or multimorbidity. Several training and mentoring initiatives have been developed to address the professional development need for psychiatrists.12-14
The need for geriatric workforce development is significant across health care, including other mental health professions.15,16 The VHA Geriatric Scholars program trains rural primary care practitioners, psychologists, and psychiatrists.17,18 Likewise, consultative geriatric specialty support for primary care practitioners in rural areas is provided via the Geriatric Research Education and Clinical Center Connect program.19 The Ask the Expert Geriatric Psychiatry email program is an additional economical model to support clinician educational development and provide rapid educational responses to inform patient care.
Ask the Expert received fewer email questions than anticipated. Enhanced optimization may require more frequent and widespread announcements about the program. Clinical staff may not be aware of the program due to an overload of email communications. Likewise, it may be challenging for busy clinicians to take the time to seek consultation or recognize a potential gap in their knowledge or skills. Had more questions been submitted, the 5 volunteer experts may have had more difficulty addressing the demand. Feedback from this project may inform development of a frequently asked questions document to share with VHA teams and a drop-in office hour to pose clinical questions of geriatric psychiatry experts, as recommended by a clinician who participated in the program.
Limitations
Not all requesters were sent a request for feedback, and the response rate for the request for feedback was only 27%. As the program has evolved, it began sending a request for feedback immediately after answering each question, which may increase the odds of response. The goal of experts answering questions within 7 business days was met 72% of the time, likely an artifact of experts integrating question answering with many other duties. The mailbox coordinator has since provided email prompts to experts immediately upon being assigned a question with the goal of improving timeliness. The program did not include chart reviews or patient consultations, as neither was feasible for volunteer experts. The email consultation service is a single component of virtual consultative specialty geriatric mental health services within the VHA, including video consultations via NEXCSS-MH and regional geriatric mental health teams.
Conclusions
The need for specialty geriatric mental health services is increasing in the VHA and across the US. However, there are too few board-certified geriatric psychiatrists to provide direct patient care to all older adults with cognitive and mental health disorders. The VHA has leveraged telehealth to improve access to geriatric mental health care. The VHA Ask the Expert Geriatric Psychiatry email consultation program is a low-resource service which provides rapid feedback to clinicians nationwide on challenging clinical scenarios, many of which are dementia-related. Most users of the service who responded to requests for feedback reported that answers to their questions were helpful and timely. The email consultation program should continue to be supplemented by more comprehensive geriatric telemental health services for particularly complex cases to meet the needs of older veterans.
- 2023 population projections for the nation by age, sex, race, Hispanic origin and nativity. United States Census Bureau. November 9, 2023. Accessed December 11, 2025. https://www.census.gov/newsroom/press-kits/2023/population-projections.html
- National Academies of Sciences Engineering and Medicine. Addressing the rising mental health needs of an aging population: proceedings of a workshop. 2024. Accessed December 11, 2025. doi.org:10.17226/27340
- Institute of Medicine. The mental health and substance use workforce for older adults: in whose hands? The National Academies Press; 2012. Accessed December 11, 2025. doi:10.17226/13400
- American Psychiatric Association. 2022 resident/fellow census. November 2023. Accessed December 11, 2025. https://www.psychiatry.org/getmedia/d80438af-f760-40f3-9d33-f91309b09564/APA-Resident-Census-2022.pdf
- Juul D, Colenda CC, Lyness JM, et al. Subspecialty training and certification in geriatric psychiatry: a 25-year overview. Am J Geriatr Psychiatry. 2017;25:445-453. doi:10.1016/j.jagp.2016.12.018
- Jaske E, Wheat CL, Rubenstein LV, et al. Understanding how contingency staffing programs can support mental health services in the Veterans Health Administration. Telemed J E Health. 2024;30:1857-1865. doi:10.1089/tmj.2023.0573
- Gould CE, Carlson C, Alfaro AJ, et al. Supporting veterans, caregivers, and providers in rural regions with tele-geriatric psychiatry consultation: a mixed methods pilot study. Am J Geriatr Psychiatry. 2023;31:279-290. doi:10.1016/j.jagp.2023.01.005
- Gould CE, Paiko L, Carlson C, et al. Implementation of tele-geriatricmental healthcare for rural veterans: factors influencing care models. Front Health Serv. 2024;4:1221899. doi:10.3389/frhs.2024.1221899
- Padala P, Schultz S, Khatkhate G, et al. Ask the expert geriatric psychiatry: VA program to support clinicians. Am J Geriatr Psychiatry. 2022;30:S18. doi:10.1016/j.jagp.2022.01.279
- Hsieh HF, Shannon SE. Three approaches to qualitative content analysis. Qual Health Res. 2005;15:1277-1288. doi:10.1177/1049732305276687
- Accreditation Council for Graduate Medical Education. Program requirements for graduate medical education in psychiatry. Revised September 3, 2025. Accessed December 11, 2025. https://www.acgme.org/globalassets/pfassets/programrequirements/2025-reformatted-requirements/400_psychiatry_2025_reformatted.pdf
- Fernandez J, Agarwal KS, Amspoker AB, et al. Outcomes from an interprofessional, dementia-focused, telementoring program: a brief report. Gerontol Geriatr Educ. 2024;45:601-606. doi:10.1080/02701960.2023.2253175
- Conroy ML, Garcia-Pittman EC, van Dyck LI, et al. The COVID-19 American Association for Geriatric Psychiatry (AAGP) online trainee curriculum: program evaluation and future directions. Am J Geriatr Psychiatry. 2025;33:308-314. doi:10.1016/j.jagp.2024.10.010
- Conroy ML, Garcia-Pittman EC, Ali H, et al. The COVID-19 AAGP online trainee curriculum: development and method of initial evaluation. Am J Geriatr Psychiatry. 2020;28:1004-1008. doi:10.1016/j.jagp.2020.06.003
- Flaherty E, Busby-Whitehead J, Potter J, et al. The geriatric workforce enhancement program: review of the coordinating center and examples of the GWEP in practice. Am J Geriatr Psychiatry. 2019;27:675-686. doi:10.1016/j.jagp.2019.04.010
- Hoge MA, Karel MJ, Zeiss AM, et al. Strengthening psychology’s workforce for older adults: implications of the Institute of Medicine’s report to Congress. Am Psychol. 2015;70:265-278. doi:10.1037/a0038927
- Kramer BJ, Creekmur B, Howe JL, et al. Veterans Affairs geriatric scholars program: enhancing existing primary care clinician skills in caring for older veterans. J Am Geriatr Soc. 2016;64:2343-2348. doi:10.1111/jgs.14382
- Gould CE, Rodriguez RL, Gregg JJ, et al. Preparing Veterans Health Administration psychologists to meet the complex needs of aging veterans. Fed Pract. 2024;41:S10-S15. doi:10.12788/fp.0466
- Pimentel CB, Gately M, Barczi SR, et al. GRECC Connect: Geriatrics telehealth to empower health care providers and improve management of older veterans in rural communities. Fed Pract. 2019;36:464-470.
- 2023 population projections for the nation by age, sex, race, Hispanic origin and nativity. United States Census Bureau. November 9, 2023. Accessed December 11, 2025. https://www.census.gov/newsroom/press-kits/2023/population-projections.html
- National Academies of Sciences Engineering and Medicine. Addressing the rising mental health needs of an aging population: proceedings of a workshop. 2024. Accessed December 11, 2025. doi.org:10.17226/27340
- Institute of Medicine. The mental health and substance use workforce for older adults: in whose hands? The National Academies Press; 2012. Accessed December 11, 2025. doi:10.17226/13400
- American Psychiatric Association. 2022 resident/fellow census. November 2023. Accessed December 11, 2025. https://www.psychiatry.org/getmedia/d80438af-f760-40f3-9d33-f91309b09564/APA-Resident-Census-2022.pdf
- Juul D, Colenda CC, Lyness JM, et al. Subspecialty training and certification in geriatric psychiatry: a 25-year overview. Am J Geriatr Psychiatry. 2017;25:445-453. doi:10.1016/j.jagp.2016.12.018
- Jaske E, Wheat CL, Rubenstein LV, et al. Understanding how contingency staffing programs can support mental health services in the Veterans Health Administration. Telemed J E Health. 2024;30:1857-1865. doi:10.1089/tmj.2023.0573
- Gould CE, Carlson C, Alfaro AJ, et al. Supporting veterans, caregivers, and providers in rural regions with tele-geriatric psychiatry consultation: a mixed methods pilot study. Am J Geriatr Psychiatry. 2023;31:279-290. doi:10.1016/j.jagp.2023.01.005
- Gould CE, Paiko L, Carlson C, et al. Implementation of tele-geriatricmental healthcare for rural veterans: factors influencing care models. Front Health Serv. 2024;4:1221899. doi:10.3389/frhs.2024.1221899
- Padala P, Schultz S, Khatkhate G, et al. Ask the expert geriatric psychiatry: VA program to support clinicians. Am J Geriatr Psychiatry. 2022;30:S18. doi:10.1016/j.jagp.2022.01.279
- Hsieh HF, Shannon SE. Three approaches to qualitative content analysis. Qual Health Res. 2005;15:1277-1288. doi:10.1177/1049732305276687
- Accreditation Council for Graduate Medical Education. Program requirements for graduate medical education in psychiatry. Revised September 3, 2025. Accessed December 11, 2025. https://www.acgme.org/globalassets/pfassets/programrequirements/2025-reformatted-requirements/400_psychiatry_2025_reformatted.pdf
- Fernandez J, Agarwal KS, Amspoker AB, et al. Outcomes from an interprofessional, dementia-focused, telementoring program: a brief report. Gerontol Geriatr Educ. 2024;45:601-606. doi:10.1080/02701960.2023.2253175
- Conroy ML, Garcia-Pittman EC, van Dyck LI, et al. The COVID-19 American Association for Geriatric Psychiatry (AAGP) online trainee curriculum: program evaluation and future directions. Am J Geriatr Psychiatry. 2025;33:308-314. doi:10.1016/j.jagp.2024.10.010
- Conroy ML, Garcia-Pittman EC, Ali H, et al. The COVID-19 AAGP online trainee curriculum: development and method of initial evaluation. Am J Geriatr Psychiatry. 2020;28:1004-1008. doi:10.1016/j.jagp.2020.06.003
- Flaherty E, Busby-Whitehead J, Potter J, et al. The geriatric workforce enhancement program: review of the coordinating center and examples of the GWEP in practice. Am J Geriatr Psychiatry. 2019;27:675-686. doi:10.1016/j.jagp.2019.04.010
- Hoge MA, Karel MJ, Zeiss AM, et al. Strengthening psychology’s workforce for older adults: implications of the Institute of Medicine’s report to Congress. Am Psychol. 2015;70:265-278. doi:10.1037/a0038927
- Kramer BJ, Creekmur B, Howe JL, et al. Veterans Affairs geriatric scholars program: enhancing existing primary care clinician skills in caring for older veterans. J Am Geriatr Soc. 2016;64:2343-2348. doi:10.1111/jgs.14382
- Gould CE, Rodriguez RL, Gregg JJ, et al. Preparing Veterans Health Administration psychologists to meet the complex needs of aging veterans. Fed Pract. 2024;41:S10-S15. doi:10.12788/fp.0466
- Pimentel CB, Gately M, Barczi SR, et al. GRECC Connect: Geriatrics telehealth to empower health care providers and improve management of older veterans in rural communities. Fed Pract. 2019;36:464-470.
Ask the Expert Geriatric Psychiatry: A VHA Email Consultation Program to Support Clinicians
Ask the Expert Geriatric Psychiatry: A VHA Email Consultation Program to Support Clinicians
Meeting the Needs of Those Who Have Served: The Role of VHA Specialized Mental Health Centers of Excellence
Meeting the Needs of Those Who Have Served: The Role of VHA Specialized Mental Health Centers of Excellence
Accessible and effective mental health services are a vital component of the Veterans Health Administration (VHA) mission to provide exceptional care that improves veterans’ health and well-being. Veterans are seeking mental health care at the VHA at significantly higher rates than in previous years. From 2009 through 2024, the number of veterans who received direct mental health care from the VHA increased 78%.1 The proportion of veterans enrolled in the VHA who also received direct mental health care expanded from 23% of total enrollees in 2009 to 33% in 2024. The increase in VHA mental health care delivery is also reflected in the number of outpatient mental health care and treatment visits at the VHA, which increased from 12.7 million to 21.5 million over the same period.
The Sergeant First Class (SFC) Heath Robinson Honoring Our Promise to Address Comprehensive Toxics (PACT) Act of 2022 expanded eligibility for VHA services, including mental health care and counseling, to new cohorts of toxic-exposed veterans. From 2022 to 2024, > 680,000 veterans enrolled in VHA care, and 45% of those were PACT Act-eligible cohorts.2 Research highlighted the high prevalence of physical and psychiatric comorbidities in toxic-exposed veterans.3 As such, the VHA may face greater demand for direct mental health care with these newly eligible cohorts of veterans.
Veterans often have mental health conditions (eg, depression, posttraumatic stress disorder [PTSD]), substance use disorders (SUDs), unique military experiences (eg, deployments), and injuries and illnesses (eg, traumatic brain injury [TBI]) that increase the complexity of their clinical presentation. The varied nature of these mental health conditions, as well as veterans’ unique military-related experiences, necessitates specialized centers focused on distinct high-priority areas of mental health in the VHA.
A series of public laws charged the US Department of Veterans Affairs (VA) with developing specialized mental health Centers of Excellence (CoEs) focused on high-priority areas of veteran mental health. The first of these laws, Public Law 98- 528, established the National Center for PTSD (NCPTSD), which opened in 1989.4 In 1996, Congress established specialized mental health CoEs known as Mental Illness Research, Education, and Clinical Centers (MIRECCs) across the VHA.5 To address the unique needs of post-9/11 veterans, 3 additional specialized centers were established in 2005.6 Finally, under the authority of the Secretary of the VA, specialized mental health CoEs were established to focus on SUD and integrated health care.
There are 17 geographically diverse mental health CoEs and MIRECCs across the VA (Table). CoEs are embedded in VA medical centers (VAMCs) with strong medical school academic affiliations. Organizational oversight of the CoEs is provided by the VA Office of Mental Health and Office of Suicide Prevention, respectively. As part of the oversight process, CoEs submit annual reports detailing their advancements in research, education and training, and clinical activities, as well as participate in a peer-reviewed renewal process.
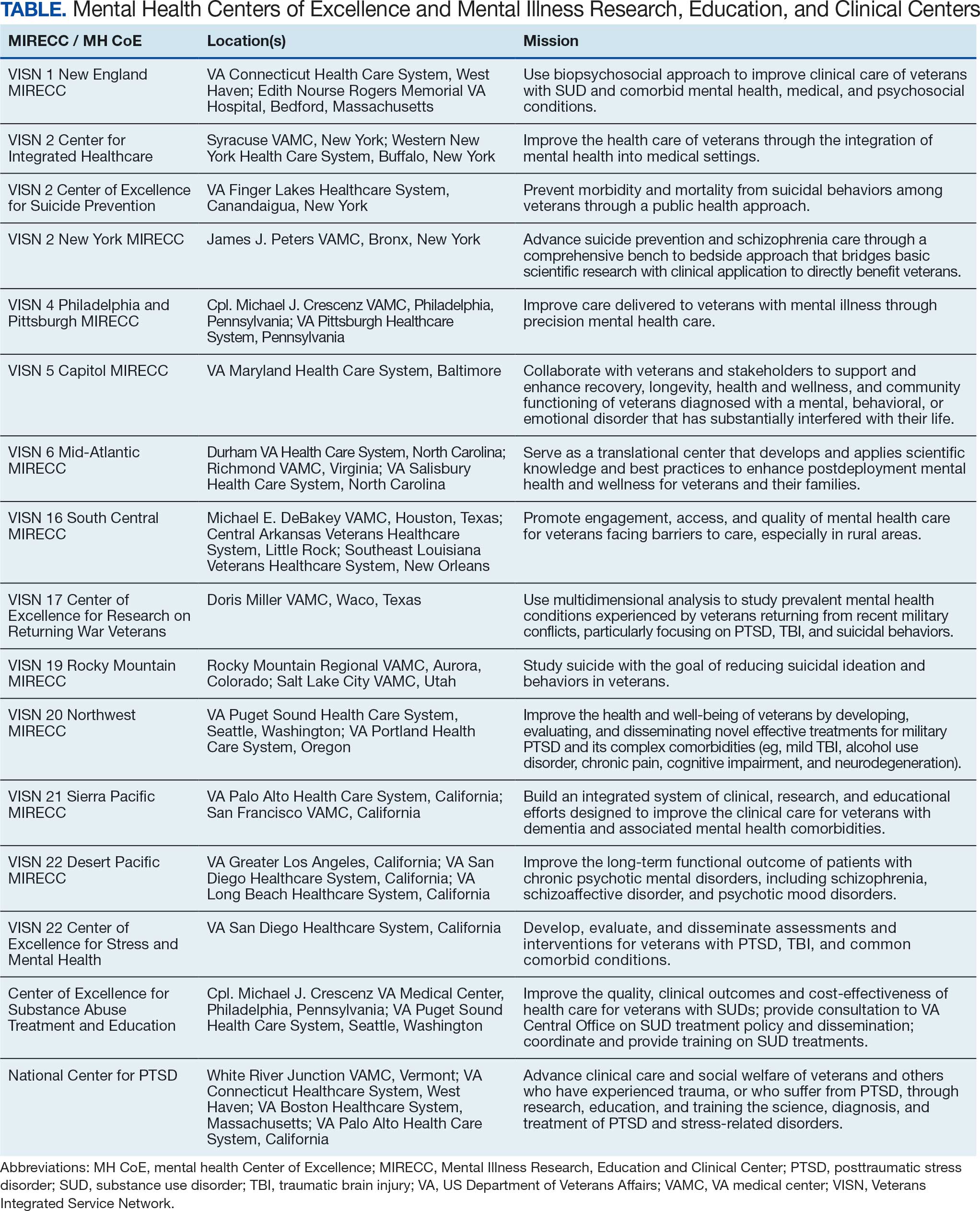
These specialized centers are united in a shared tripartite mission to generate new knowledge about the causes and treatments of mental health conditions, to educate and train VHA clinicians and personnel, and to develop and implement innovative clinical programs within the VHA. This combined focus on research, education, and improved clinical care reduces the time from discovery to implementation and improves the health and well-being of veterans.
Examples of this acceleration translation from scientific discovery to clinical practice are evident throughout mental health CoEs. The NCPTSD is a leader in research on PTSD and traumatic stress. Its work led to the development of national training programs for VHA clinicians, facilitating the broad dissemination of efficacious PTSD treatments across the enterprise.7 Researchers at the Veterans Integrated Service Network (VISN) 21 Sierra Pacific MIRECC identified depression as a significant risk factor for dementia and subsequently launched the first multisite trial of repetitive transcranial magnetic stimulation (rTMS) in the VHA in 2012 (CSP #556: the effectiveness of rTMS in depressed VA patients).8 This project laid the groundwork for the national clinical rTMS program launched in 2017, which is now clinically available at 60 VAMCs. In the largest pragmatic randomized clinical trial of its kind, the VISN 4 Philadelphia and Pittsburgh MIRECC found that pharmacogenomic testing significantly reduced the number of prescription medications with predicted drug-gene interactions and improved clinical outcomes among veterans with depression.9
Mental health CoEs are also leaders in suicide prevention, a top clinical priority for the VHA. The VISN 2 New York MIRECC developed Project Life Force, a safety planning skills group for veterans with suicidal ideation, now implemented across 10 VAMCs, including telehealth hubs, outpatient settings, and veteran peer programs.10 The VISN 2 CoE for Suicide Prevention and VISN 19 Rocky Mountain MIRECC coordinate key suicide prevention services for VA, including the analysis of suicide surveillance data; evaluation of national VA suicide prevention initiatives; the support of veterans, families, and clinicians; and enhanced access to evidence-based treatments for at-risk veterans.
Mental health CoEs are a key operational partner in VHA treatment of SUDs. The CoEs in Substance Addiction Treatment and Education (CESATEs) are national resources dedicated to improving the quality, clinical outcomes, and cost-effectiveness of VHA SUD treatment. CESATEs developed and implemented a national rollout of an effective treatment for stimulant use disorders, training staff at > 120 VA programs. The VISN 1 Mid-Atlantic MIRECC’s focus on SUD and comorbid/co-occurring mental health conditions has highlighted the significant prevalence of these conditions and the impact they have on treatment response.11
Serious mental illness (SMI) (eg, schizophrenia, schizoaffective disorder) impacts up to 5% of veterans.12 VISN 22 Desert Pacific MIRECC has developed interventions to improve the lives of veterans with SMI. Its research established supported employment as an effective intervention to improve outcomes in veterans with psychotic disorders and supported its implementation in the VHA.13 Peer specialists are a cornerstone in the VHA commitment to recovery-oriented services for veterans with SMI. VISN 5 Capitol MIRECC has long championed research, clinical training, and educational activities that contributed to the effective deployment of peer specialists across the VHA enterprise.
Veterans have unique military-related experiences (eg, deployment, traumatic stress, transition to civilian status) and injuries and illnesses (eg, TBI, posttraumatic headaches) that significantly impact their mental health and quality of life.
The period between active duty and transition to civilian status is a critical time in a veteran’s life. The VISN 17 CoE Veteran Sponsorship Initiative connects veterans with VA care within 30 days postdischarge, with the option of additional support in the community. The VISN 22 CoE for Stress and Mental Health (CESAMH) develops, evaluates, and disseminates diagnostics and treatments for veterans affected by traumatic events and brain injuries, with a unique focus on supporting their whole health needs. The VISN 6 Mid-Atlantic MIRECC leads the ongoing VISN-6 Post-Deployment Mental Health (PDMH) study, the largest biorepository of post-9/11 veterans. PDMH has greatly expanded the understanding of the unique needs of post-9/11 veterans, with > 100 peer-reviewed publications to date. Veterans with mild TBI frequently experience chronic posttraumatic headaches that can be disabling and nonresponsive to treatment. The VISN 20 Northwest MIRECC demonstrated that prazosin, a repurposed, low-cost, widely available, nonaddictive medication, can safely and effectively reduce the frequency of these headaches and improve functional impairment in veterans and active-duty service members.14
Increased and enhanced access to effective mental health treatment is a priority for VA. In 2007, the VA launched the National Primary Care Mental Health Integration program, which integrated mental health services into primary care settings. The Center for Integrated Healthcare (CIH) has supported the VA in these efforts. In 2024, CIH trained > 5000 health care staff on high-fidelity integration of behavioral health and medical care. VA has also focused on increasing access to mental health services via expanded telehealth offerings. The VISN 16 MIRECC, with its unique focus on increasing access to care for rural veterans via distance-based and digital health technology, supported the VA Offices of Mental Health and Connected Care to virtualize mental health care and promote adoption and sustained use of VA Video Connect across the enterprise.
Specialized MH CoEs are uniquely equipped to support the VHA in providing training and education to VA clinicians, veterans, care partners and family members, and the community on high-priority mental health topics. Education is a core component of the MH CoEs tripartite mission. As such, MH CoEs offer national trainings, conferences, consultation services, clinical demonstration projects, development of clinical dashboards and toolkits, and public awareness campaigns. Researchers, educators, and clinicians at the CoEs frequently serve as subject matter experts on topics aligned with their respective missions. Several national rollout programs that disseminated evidence-based treatments for mental health conditions to the field (eg, cognitive behavioral therapy for depression, cognitive behavioral therapy for insomnia, and prolonged exposure) were developed at specialized CoEs.
The VHA provides advanced training, residencies, and fellowships to > 120,000 trainees annually. Many of these trainees choose to remain at the VA. Seven of 10 VHA psychologists and 6 of 10 VHA physicians trained within the VHA prior to their employment.15 The MH CoEs and MIRECCs play an important role in preparing these trainees for VHA mental health careers. These centers are funded to provide advanced postdoctoral training to physicians as well as allied health professionals in clinical and counseling psychology, social work, pharmacy, and nursing. Training is not limited to postdoctoral fellows: graduate students, residents, and interns from affiliated accredited training programs may rotate through mental health CoEs each academic year.
Conclusions
For > 30 years, mental health CoEs have brought thousands of veterans advanced treatments for their mental health needs and helped reduce death by suicide. The centers have a bright future ahead, harnessing advances in artificial intelligence and genomics to permit the matching of the individual veterans to the treatment most likely to benefit them. Precision medicine, as espoused by the Hannon Act, will not only encourage the efficient use of health care resources but also rapidly reduce pain in veterans with mental health and SUDs.
- Congressionally Mandated Report: Report on Transparency in Mental Health Care Services. US Dept of Veterans Affairs; December 2022. Accessed December 5, 2025. https://www.govinfo.gov/content/pkg/CMR-VA1-00181657/pdf/CMR-VA1-00181657.pdf
- Beckman AL, Jacobs J, Elnahal SM. The PACT Act—expanding coverage and access for veterans. JAMA. 2024;332:1423-1424. doi:10.1001/jama.2024.16013
- Morse JL, Acheson DT, Almklov E, et al. Associations among environmental exposures and physical and psychiatric symptoms in a care-seeking sample of U.S. military veterans. Mil Med. 2024;189:e1397-e1402. doi:10.1093/milmed/usae035
- Veterans’ Health Care Act of 1984, 38 USC §98-528 (1984). Accessed March 27, 2026. https://www.congress.gov/bill/98th-congress/house-bill/5618/text
- Veterans’ Health Care Eligibility Reform Act of 1996, 38 USC §104-262 (1996). Accessed March 27, 2026. https://www.congress.gov/bill/104th-congress/house-bill/3118/text
- Military Quality of Life and Veterans Affairs Appropriations Act, 2006. Pub L No. 109-114, 119 Stat. 2372. Accessed March 27, 2026. https://www.congress.gov/bill/109th-congress/house-bill/2528/text
- Karlin BE, Ruzek JI, Chard KM, et al. Dissemination of evidence‐based psychological treatments for posttraumatic stress disorder in the Veterans Health Administration. J Trauma Stress. 2010;23:663-673. doi:10.1002/jts.20588
- Byers AL, Covinsky KE, Barnes DE, et al. Dysthymia and depression increase risk of dementia and mortality among older veterans. Am J Geriatr Psychiatry. 2012;20:664-672. doi:10.1097/JGP.0b013e31822001c1
- Oslin DW, Lynch KG, Shih MC, et al. Effect of pharmacogenomic testing for drug-gene interactions on medication selection and remission of symptoms in major depressive disorder: the PRIME Care randomized clinical trial. JAMA. 2022;328:151-161. doi:10.1001/jama.2022.9805
- Goodman M, Brown GK, Galfalvy HC, et al. Group (“Project Life Force”) versus individual suicide safety planning: a randomized clinical trial. Contemp Clin Trials Commun. 2020;17:100520. doi:10.1016/j.conctc.2020.100520
- Na PJ, Ralevski E, Jegede O, et al. Depression and/or PTSD comorbidity affects response to antidepressants in those with alcohol use disorder. Front Psychiatry. 2022;12:768318. doi:10.3389/fpsyt.2021.768318
- McCarthy JF, Blow FC, Valenstein M, et al. Veterans Affairs Health System and mental health treatment retention among patients with serious mental illness: evaluating accessibility and availability barriers. Health Serv Res. 2007;42:1042-1060. doi:10.1111/j.1475-6773.2006.00642.x
- Glynn SM, Marder SR, Noordsy DL, et al. An RCT evaluating the effects of skills training and medication type on work outcomes among patients with schizophrenia. Psychiatr Serv. 2016;67:500-506. doi:10.1176/appips201500171
- Mayer CL, Savage PJ, Engle CK, et al. Randomized controlled pilot trial of prazosin for prophylaxis of posttraumatic headaches in active-duty service members and veterans. Headache. 2023;63:751-762. doi:10.1111/head.14529
- Hill C. Medical education at VA: it’s all about the veterans. VA News. August 18, 2021. Accessed December 5, 2025. https://news.va.gov/93370/medical-education-at-va-its-all-about-the-veterans/
Accessible and effective mental health services are a vital component of the Veterans Health Administration (VHA) mission to provide exceptional care that improves veterans’ health and well-being. Veterans are seeking mental health care at the VHA at significantly higher rates than in previous years. From 2009 through 2024, the number of veterans who received direct mental health care from the VHA increased 78%.1 The proportion of veterans enrolled in the VHA who also received direct mental health care expanded from 23% of total enrollees in 2009 to 33% in 2024. The increase in VHA mental health care delivery is also reflected in the number of outpatient mental health care and treatment visits at the VHA, which increased from 12.7 million to 21.5 million over the same period.
The Sergeant First Class (SFC) Heath Robinson Honoring Our Promise to Address Comprehensive Toxics (PACT) Act of 2022 expanded eligibility for VHA services, including mental health care and counseling, to new cohorts of toxic-exposed veterans. From 2022 to 2024, > 680,000 veterans enrolled in VHA care, and 45% of those were PACT Act-eligible cohorts.2 Research highlighted the high prevalence of physical and psychiatric comorbidities in toxic-exposed veterans.3 As such, the VHA may face greater demand for direct mental health care with these newly eligible cohorts of veterans.
Veterans often have mental health conditions (eg, depression, posttraumatic stress disorder [PTSD]), substance use disorders (SUDs), unique military experiences (eg, deployments), and injuries and illnesses (eg, traumatic brain injury [TBI]) that increase the complexity of their clinical presentation. The varied nature of these mental health conditions, as well as veterans’ unique military-related experiences, necessitates specialized centers focused on distinct high-priority areas of mental health in the VHA.
A series of public laws charged the US Department of Veterans Affairs (VA) with developing specialized mental health Centers of Excellence (CoEs) focused on high-priority areas of veteran mental health. The first of these laws, Public Law 98- 528, established the National Center for PTSD (NCPTSD), which opened in 1989.4 In 1996, Congress established specialized mental health CoEs known as Mental Illness Research, Education, and Clinical Centers (MIRECCs) across the VHA.5 To address the unique needs of post-9/11 veterans, 3 additional specialized centers were established in 2005.6 Finally, under the authority of the Secretary of the VA, specialized mental health CoEs were established to focus on SUD and integrated health care.
There are 17 geographically diverse mental health CoEs and MIRECCs across the VA (Table). CoEs are embedded in VA medical centers (VAMCs) with strong medical school academic affiliations. Organizational oversight of the CoEs is provided by the VA Office of Mental Health and Office of Suicide Prevention, respectively. As part of the oversight process, CoEs submit annual reports detailing their advancements in research, education and training, and clinical activities, as well as participate in a peer-reviewed renewal process.

These specialized centers are united in a shared tripartite mission to generate new knowledge about the causes and treatments of mental health conditions, to educate and train VHA clinicians and personnel, and to develop and implement innovative clinical programs within the VHA. This combined focus on research, education, and improved clinical care reduces the time from discovery to implementation and improves the health and well-being of veterans.
Examples of this acceleration translation from scientific discovery to clinical practice are evident throughout mental health CoEs. The NCPTSD is a leader in research on PTSD and traumatic stress. Its work led to the development of national training programs for VHA clinicians, facilitating the broad dissemination of efficacious PTSD treatments across the enterprise.7 Researchers at the Veterans Integrated Service Network (VISN) 21 Sierra Pacific MIRECC identified depression as a significant risk factor for dementia and subsequently launched the first multisite trial of repetitive transcranial magnetic stimulation (rTMS) in the VHA in 2012 (CSP #556: the effectiveness of rTMS in depressed VA patients).8 This project laid the groundwork for the national clinical rTMS program launched in 2017, which is now clinically available at 60 VAMCs. In the largest pragmatic randomized clinical trial of its kind, the VISN 4 Philadelphia and Pittsburgh MIRECC found that pharmacogenomic testing significantly reduced the number of prescription medications with predicted drug-gene interactions and improved clinical outcomes among veterans with depression.9
Mental health CoEs are also leaders in suicide prevention, a top clinical priority for the VHA. The VISN 2 New York MIRECC developed Project Life Force, a safety planning skills group for veterans with suicidal ideation, now implemented across 10 VAMCs, including telehealth hubs, outpatient settings, and veteran peer programs.10 The VISN 2 CoE for Suicide Prevention and VISN 19 Rocky Mountain MIRECC coordinate key suicide prevention services for VA, including the analysis of suicide surveillance data; evaluation of national VA suicide prevention initiatives; the support of veterans, families, and clinicians; and enhanced access to evidence-based treatments for at-risk veterans.
Mental health CoEs are a key operational partner in VHA treatment of SUDs. The CoEs in Substance Addiction Treatment and Education (CESATEs) are national resources dedicated to improving the quality, clinical outcomes, and cost-effectiveness of VHA SUD treatment. CESATEs developed and implemented a national rollout of an effective treatment for stimulant use disorders, training staff at > 120 VA programs. The VISN 1 Mid-Atlantic MIRECC’s focus on SUD and comorbid/co-occurring mental health conditions has highlighted the significant prevalence of these conditions and the impact they have on treatment response.11
Serious mental illness (SMI) (eg, schizophrenia, schizoaffective disorder) impacts up to 5% of veterans.12 VISN 22 Desert Pacific MIRECC has developed interventions to improve the lives of veterans with SMI. Its research established supported employment as an effective intervention to improve outcomes in veterans with psychotic disorders and supported its implementation in the VHA.13 Peer specialists are a cornerstone in the VHA commitment to recovery-oriented services for veterans with SMI. VISN 5 Capitol MIRECC has long championed research, clinical training, and educational activities that contributed to the effective deployment of peer specialists across the VHA enterprise.
Veterans have unique military-related experiences (eg, deployment, traumatic stress, transition to civilian status) and injuries and illnesses (eg, TBI, posttraumatic headaches) that significantly impact their mental health and quality of life.
The period between active duty and transition to civilian status is a critical time in a veteran’s life. The VISN 17 CoE Veteran Sponsorship Initiative connects veterans with VA care within 30 days postdischarge, with the option of additional support in the community. The VISN 22 CoE for Stress and Mental Health (CESAMH) develops, evaluates, and disseminates diagnostics and treatments for veterans affected by traumatic events and brain injuries, with a unique focus on supporting their whole health needs. The VISN 6 Mid-Atlantic MIRECC leads the ongoing VISN-6 Post-Deployment Mental Health (PDMH) study, the largest biorepository of post-9/11 veterans. PDMH has greatly expanded the understanding of the unique needs of post-9/11 veterans, with > 100 peer-reviewed publications to date. Veterans with mild TBI frequently experience chronic posttraumatic headaches that can be disabling and nonresponsive to treatment. The VISN 20 Northwest MIRECC demonstrated that prazosin, a repurposed, low-cost, widely available, nonaddictive medication, can safely and effectively reduce the frequency of these headaches and improve functional impairment in veterans and active-duty service members.14
Increased and enhanced access to effective mental health treatment is a priority for VA. In 2007, the VA launched the National Primary Care Mental Health Integration program, which integrated mental health services into primary care settings. The Center for Integrated Healthcare (CIH) has supported the VA in these efforts. In 2024, CIH trained > 5000 health care staff on high-fidelity integration of behavioral health and medical care. VA has also focused on increasing access to mental health services via expanded telehealth offerings. The VISN 16 MIRECC, with its unique focus on increasing access to care for rural veterans via distance-based and digital health technology, supported the VA Offices of Mental Health and Connected Care to virtualize mental health care and promote adoption and sustained use of VA Video Connect across the enterprise.
Specialized MH CoEs are uniquely equipped to support the VHA in providing training and education to VA clinicians, veterans, care partners and family members, and the community on high-priority mental health topics. Education is a core component of the MH CoEs tripartite mission. As such, MH CoEs offer national trainings, conferences, consultation services, clinical demonstration projects, development of clinical dashboards and toolkits, and public awareness campaigns. Researchers, educators, and clinicians at the CoEs frequently serve as subject matter experts on topics aligned with their respective missions. Several national rollout programs that disseminated evidence-based treatments for mental health conditions to the field (eg, cognitive behavioral therapy for depression, cognitive behavioral therapy for insomnia, and prolonged exposure) were developed at specialized CoEs.
The VHA provides advanced training, residencies, and fellowships to > 120,000 trainees annually. Many of these trainees choose to remain at the VA. Seven of 10 VHA psychologists and 6 of 10 VHA physicians trained within the VHA prior to their employment.15 The MH CoEs and MIRECCs play an important role in preparing these trainees for VHA mental health careers. These centers are funded to provide advanced postdoctoral training to physicians as well as allied health professionals in clinical and counseling psychology, social work, pharmacy, and nursing. Training is not limited to postdoctoral fellows: graduate students, residents, and interns from affiliated accredited training programs may rotate through mental health CoEs each academic year.
Conclusions
For > 30 years, mental health CoEs have brought thousands of veterans advanced treatments for their mental health needs and helped reduce death by suicide. The centers have a bright future ahead, harnessing advances in artificial intelligence and genomics to permit the matching of the individual veterans to the treatment most likely to benefit them. Precision medicine, as espoused by the Hannon Act, will not only encourage the efficient use of health care resources but also rapidly reduce pain in veterans with mental health and SUDs.
Accessible and effective mental health services are a vital component of the Veterans Health Administration (VHA) mission to provide exceptional care that improves veterans’ health and well-being. Veterans are seeking mental health care at the VHA at significantly higher rates than in previous years. From 2009 through 2024, the number of veterans who received direct mental health care from the VHA increased 78%.1 The proportion of veterans enrolled in the VHA who also received direct mental health care expanded from 23% of total enrollees in 2009 to 33% in 2024. The increase in VHA mental health care delivery is also reflected in the number of outpatient mental health care and treatment visits at the VHA, which increased from 12.7 million to 21.5 million over the same period.
The Sergeant First Class (SFC) Heath Robinson Honoring Our Promise to Address Comprehensive Toxics (PACT) Act of 2022 expanded eligibility for VHA services, including mental health care and counseling, to new cohorts of toxic-exposed veterans. From 2022 to 2024, > 680,000 veterans enrolled in VHA care, and 45% of those were PACT Act-eligible cohorts.2 Research highlighted the high prevalence of physical and psychiatric comorbidities in toxic-exposed veterans.3 As such, the VHA may face greater demand for direct mental health care with these newly eligible cohorts of veterans.
Veterans often have mental health conditions (eg, depression, posttraumatic stress disorder [PTSD]), substance use disorders (SUDs), unique military experiences (eg, deployments), and injuries and illnesses (eg, traumatic brain injury [TBI]) that increase the complexity of their clinical presentation. The varied nature of these mental health conditions, as well as veterans’ unique military-related experiences, necessitates specialized centers focused on distinct high-priority areas of mental health in the VHA.
A series of public laws charged the US Department of Veterans Affairs (VA) with developing specialized mental health Centers of Excellence (CoEs) focused on high-priority areas of veteran mental health. The first of these laws, Public Law 98- 528, established the National Center for PTSD (NCPTSD), which opened in 1989.4 In 1996, Congress established specialized mental health CoEs known as Mental Illness Research, Education, and Clinical Centers (MIRECCs) across the VHA.5 To address the unique needs of post-9/11 veterans, 3 additional specialized centers were established in 2005.6 Finally, under the authority of the Secretary of the VA, specialized mental health CoEs were established to focus on SUD and integrated health care.
There are 17 geographically diverse mental health CoEs and MIRECCs across the VA (Table). CoEs are embedded in VA medical centers (VAMCs) with strong medical school academic affiliations. Organizational oversight of the CoEs is provided by the VA Office of Mental Health and Office of Suicide Prevention, respectively. As part of the oversight process, CoEs submit annual reports detailing their advancements in research, education and training, and clinical activities, as well as participate in a peer-reviewed renewal process.

These specialized centers are united in a shared tripartite mission to generate new knowledge about the causes and treatments of mental health conditions, to educate and train VHA clinicians and personnel, and to develop and implement innovative clinical programs within the VHA. This combined focus on research, education, and improved clinical care reduces the time from discovery to implementation and improves the health and well-being of veterans.
Examples of this acceleration translation from scientific discovery to clinical practice are evident throughout mental health CoEs. The NCPTSD is a leader in research on PTSD and traumatic stress. Its work led to the development of national training programs for VHA clinicians, facilitating the broad dissemination of efficacious PTSD treatments across the enterprise.7 Researchers at the Veterans Integrated Service Network (VISN) 21 Sierra Pacific MIRECC identified depression as a significant risk factor for dementia and subsequently launched the first multisite trial of repetitive transcranial magnetic stimulation (rTMS) in the VHA in 2012 (CSP #556: the effectiveness of rTMS in depressed VA patients).8 This project laid the groundwork for the national clinical rTMS program launched in 2017, which is now clinically available at 60 VAMCs. In the largest pragmatic randomized clinical trial of its kind, the VISN 4 Philadelphia and Pittsburgh MIRECC found that pharmacogenomic testing significantly reduced the number of prescription medications with predicted drug-gene interactions and improved clinical outcomes among veterans with depression.9
Mental health CoEs are also leaders in suicide prevention, a top clinical priority for the VHA. The VISN 2 New York MIRECC developed Project Life Force, a safety planning skills group for veterans with suicidal ideation, now implemented across 10 VAMCs, including telehealth hubs, outpatient settings, and veteran peer programs.10 The VISN 2 CoE for Suicide Prevention and VISN 19 Rocky Mountain MIRECC coordinate key suicide prevention services for VA, including the analysis of suicide surveillance data; evaluation of national VA suicide prevention initiatives; the support of veterans, families, and clinicians; and enhanced access to evidence-based treatments for at-risk veterans.
Mental health CoEs are a key operational partner in VHA treatment of SUDs. The CoEs in Substance Addiction Treatment and Education (CESATEs) are national resources dedicated to improving the quality, clinical outcomes, and cost-effectiveness of VHA SUD treatment. CESATEs developed and implemented a national rollout of an effective treatment for stimulant use disorders, training staff at > 120 VA programs. The VISN 1 Mid-Atlantic MIRECC’s focus on SUD and comorbid/co-occurring mental health conditions has highlighted the significant prevalence of these conditions and the impact they have on treatment response.11
Serious mental illness (SMI) (eg, schizophrenia, schizoaffective disorder) impacts up to 5% of veterans.12 VISN 22 Desert Pacific MIRECC has developed interventions to improve the lives of veterans with SMI. Its research established supported employment as an effective intervention to improve outcomes in veterans with psychotic disorders and supported its implementation in the VHA.13 Peer specialists are a cornerstone in the VHA commitment to recovery-oriented services for veterans with SMI. VISN 5 Capitol MIRECC has long championed research, clinical training, and educational activities that contributed to the effective deployment of peer specialists across the VHA enterprise.
Veterans have unique military-related experiences (eg, deployment, traumatic stress, transition to civilian status) and injuries and illnesses (eg, TBI, posttraumatic headaches) that significantly impact their mental health and quality of life.
The period between active duty and transition to civilian status is a critical time in a veteran’s life. The VISN 17 CoE Veteran Sponsorship Initiative connects veterans with VA care within 30 days postdischarge, with the option of additional support in the community. The VISN 22 CoE for Stress and Mental Health (CESAMH) develops, evaluates, and disseminates diagnostics and treatments for veterans affected by traumatic events and brain injuries, with a unique focus on supporting their whole health needs. The VISN 6 Mid-Atlantic MIRECC leads the ongoing VISN-6 Post-Deployment Mental Health (PDMH) study, the largest biorepository of post-9/11 veterans. PDMH has greatly expanded the understanding of the unique needs of post-9/11 veterans, with > 100 peer-reviewed publications to date. Veterans with mild TBI frequently experience chronic posttraumatic headaches that can be disabling and nonresponsive to treatment. The VISN 20 Northwest MIRECC demonstrated that prazosin, a repurposed, low-cost, widely available, nonaddictive medication, can safely and effectively reduce the frequency of these headaches and improve functional impairment in veterans and active-duty service members.14
Increased and enhanced access to effective mental health treatment is a priority for VA. In 2007, the VA launched the National Primary Care Mental Health Integration program, which integrated mental health services into primary care settings. The Center for Integrated Healthcare (CIH) has supported the VA in these efforts. In 2024, CIH trained > 5000 health care staff on high-fidelity integration of behavioral health and medical care. VA has also focused on increasing access to mental health services via expanded telehealth offerings. The VISN 16 MIRECC, with its unique focus on increasing access to care for rural veterans via distance-based and digital health technology, supported the VA Offices of Mental Health and Connected Care to virtualize mental health care and promote adoption and sustained use of VA Video Connect across the enterprise.
Specialized MH CoEs are uniquely equipped to support the VHA in providing training and education to VA clinicians, veterans, care partners and family members, and the community on high-priority mental health topics. Education is a core component of the MH CoEs tripartite mission. As such, MH CoEs offer national trainings, conferences, consultation services, clinical demonstration projects, development of clinical dashboards and toolkits, and public awareness campaigns. Researchers, educators, and clinicians at the CoEs frequently serve as subject matter experts on topics aligned with their respective missions. Several national rollout programs that disseminated evidence-based treatments for mental health conditions to the field (eg, cognitive behavioral therapy for depression, cognitive behavioral therapy for insomnia, and prolonged exposure) were developed at specialized CoEs.
The VHA provides advanced training, residencies, and fellowships to > 120,000 trainees annually. Many of these trainees choose to remain at the VA. Seven of 10 VHA psychologists and 6 of 10 VHA physicians trained within the VHA prior to their employment.15 The MH CoEs and MIRECCs play an important role in preparing these trainees for VHA mental health careers. These centers are funded to provide advanced postdoctoral training to physicians as well as allied health professionals in clinical and counseling psychology, social work, pharmacy, and nursing. Training is not limited to postdoctoral fellows: graduate students, residents, and interns from affiliated accredited training programs may rotate through mental health CoEs each academic year.
Conclusions
For > 30 years, mental health CoEs have brought thousands of veterans advanced treatments for their mental health needs and helped reduce death by suicide. The centers have a bright future ahead, harnessing advances in artificial intelligence and genomics to permit the matching of the individual veterans to the treatment most likely to benefit them. Precision medicine, as espoused by the Hannon Act, will not only encourage the efficient use of health care resources but also rapidly reduce pain in veterans with mental health and SUDs.
- Congressionally Mandated Report: Report on Transparency in Mental Health Care Services. US Dept of Veterans Affairs; December 2022. Accessed December 5, 2025. https://www.govinfo.gov/content/pkg/CMR-VA1-00181657/pdf/CMR-VA1-00181657.pdf
- Beckman AL, Jacobs J, Elnahal SM. The PACT Act—expanding coverage and access for veterans. JAMA. 2024;332:1423-1424. doi:10.1001/jama.2024.16013
- Morse JL, Acheson DT, Almklov E, et al. Associations among environmental exposures and physical and psychiatric symptoms in a care-seeking sample of U.S. military veterans. Mil Med. 2024;189:e1397-e1402. doi:10.1093/milmed/usae035
- Veterans’ Health Care Act of 1984, 38 USC §98-528 (1984). Accessed March 27, 2026. https://www.congress.gov/bill/98th-congress/house-bill/5618/text
- Veterans’ Health Care Eligibility Reform Act of 1996, 38 USC §104-262 (1996). Accessed March 27, 2026. https://www.congress.gov/bill/104th-congress/house-bill/3118/text
- Military Quality of Life and Veterans Affairs Appropriations Act, 2006. Pub L No. 109-114, 119 Stat. 2372. Accessed March 27, 2026. https://www.congress.gov/bill/109th-congress/house-bill/2528/text
- Karlin BE, Ruzek JI, Chard KM, et al. Dissemination of evidence‐based psychological treatments for posttraumatic stress disorder in the Veterans Health Administration. J Trauma Stress. 2010;23:663-673. doi:10.1002/jts.20588
- Byers AL, Covinsky KE, Barnes DE, et al. Dysthymia and depression increase risk of dementia and mortality among older veterans. Am J Geriatr Psychiatry. 2012;20:664-672. doi:10.1097/JGP.0b013e31822001c1
- Oslin DW, Lynch KG, Shih MC, et al. Effect of pharmacogenomic testing for drug-gene interactions on medication selection and remission of symptoms in major depressive disorder: the PRIME Care randomized clinical trial. JAMA. 2022;328:151-161. doi:10.1001/jama.2022.9805
- Goodman M, Brown GK, Galfalvy HC, et al. Group (“Project Life Force”) versus individual suicide safety planning: a randomized clinical trial. Contemp Clin Trials Commun. 2020;17:100520. doi:10.1016/j.conctc.2020.100520
- Na PJ, Ralevski E, Jegede O, et al. Depression and/or PTSD comorbidity affects response to antidepressants in those with alcohol use disorder. Front Psychiatry. 2022;12:768318. doi:10.3389/fpsyt.2021.768318
- McCarthy JF, Blow FC, Valenstein M, et al. Veterans Affairs Health System and mental health treatment retention among patients with serious mental illness: evaluating accessibility and availability barriers. Health Serv Res. 2007;42:1042-1060. doi:10.1111/j.1475-6773.2006.00642.x
- Glynn SM, Marder SR, Noordsy DL, et al. An RCT evaluating the effects of skills training and medication type on work outcomes among patients with schizophrenia. Psychiatr Serv. 2016;67:500-506. doi:10.1176/appips201500171
- Mayer CL, Savage PJ, Engle CK, et al. Randomized controlled pilot trial of prazosin for prophylaxis of posttraumatic headaches in active-duty service members and veterans. Headache. 2023;63:751-762. doi:10.1111/head.14529
- Hill C. Medical education at VA: it’s all about the veterans. VA News. August 18, 2021. Accessed December 5, 2025. https://news.va.gov/93370/medical-education-at-va-its-all-about-the-veterans/
- Congressionally Mandated Report: Report on Transparency in Mental Health Care Services. US Dept of Veterans Affairs; December 2022. Accessed December 5, 2025. https://www.govinfo.gov/content/pkg/CMR-VA1-00181657/pdf/CMR-VA1-00181657.pdf
- Beckman AL, Jacobs J, Elnahal SM. The PACT Act—expanding coverage and access for veterans. JAMA. 2024;332:1423-1424. doi:10.1001/jama.2024.16013
- Morse JL, Acheson DT, Almklov E, et al. Associations among environmental exposures and physical and psychiatric symptoms in a care-seeking sample of U.S. military veterans. Mil Med. 2024;189:e1397-e1402. doi:10.1093/milmed/usae035
- Veterans’ Health Care Act of 1984, 38 USC §98-528 (1984). Accessed March 27, 2026. https://www.congress.gov/bill/98th-congress/house-bill/5618/text
- Veterans’ Health Care Eligibility Reform Act of 1996, 38 USC §104-262 (1996). Accessed March 27, 2026. https://www.congress.gov/bill/104th-congress/house-bill/3118/text
- Military Quality of Life and Veterans Affairs Appropriations Act, 2006. Pub L No. 109-114, 119 Stat. 2372. Accessed March 27, 2026. https://www.congress.gov/bill/109th-congress/house-bill/2528/text
- Karlin BE, Ruzek JI, Chard KM, et al. Dissemination of evidence‐based psychological treatments for posttraumatic stress disorder in the Veterans Health Administration. J Trauma Stress. 2010;23:663-673. doi:10.1002/jts.20588
- Byers AL, Covinsky KE, Barnes DE, et al. Dysthymia and depression increase risk of dementia and mortality among older veterans. Am J Geriatr Psychiatry. 2012;20:664-672. doi:10.1097/JGP.0b013e31822001c1
- Oslin DW, Lynch KG, Shih MC, et al. Effect of pharmacogenomic testing for drug-gene interactions on medication selection and remission of symptoms in major depressive disorder: the PRIME Care randomized clinical trial. JAMA. 2022;328:151-161. doi:10.1001/jama.2022.9805
- Goodman M, Brown GK, Galfalvy HC, et al. Group (“Project Life Force”) versus individual suicide safety planning: a randomized clinical trial. Contemp Clin Trials Commun. 2020;17:100520. doi:10.1016/j.conctc.2020.100520
- Na PJ, Ralevski E, Jegede O, et al. Depression and/or PTSD comorbidity affects response to antidepressants in those with alcohol use disorder. Front Psychiatry. 2022;12:768318. doi:10.3389/fpsyt.2021.768318
- McCarthy JF, Blow FC, Valenstein M, et al. Veterans Affairs Health System and mental health treatment retention among patients with serious mental illness: evaluating accessibility and availability barriers. Health Serv Res. 2007;42:1042-1060. doi:10.1111/j.1475-6773.2006.00642.x
- Glynn SM, Marder SR, Noordsy DL, et al. An RCT evaluating the effects of skills training and medication type on work outcomes among patients with schizophrenia. Psychiatr Serv. 2016;67:500-506. doi:10.1176/appips201500171
- Mayer CL, Savage PJ, Engle CK, et al. Randomized controlled pilot trial of prazosin for prophylaxis of posttraumatic headaches in active-duty service members and veterans. Headache. 2023;63:751-762. doi:10.1111/head.14529
- Hill C. Medical education at VA: it’s all about the veterans. VA News. August 18, 2021. Accessed December 5, 2025. https://news.va.gov/93370/medical-education-at-va-its-all-about-the-veterans/
Meeting the Needs of Those Who Have Served: The Role of VHA Specialized Mental Health Centers of Excellence
Meeting the Needs of Those Who Have Served: The Role of VHA Specialized Mental Health Centers of Excellence
Accelerated Unified Protocol for Transdiagnostic Treatment of Anxiety Disorders in a VHA System
Accelerated Unified Protocol for Transdiagnostic Treatment of Anxiety Disorders in a VHA System
Cognitive behavioral therapy (CBT) is supported as an effective treatment for depression by clinical practice guidelines from the US Department of Veterans Affairs (VA) and US Department of Defense.1-3 Despite research supporting the use of evidence-based CBT for anxiety, mood, and emotional disorders, patient access to these interventions is limited.4 One barrier to CBT for anxiety, mood, and emotional disorders is the traditional use of single-disorder protocols (SDPs) to separately treat each disorder (eg, major depression, generalized anxiety disorder, panic disorder).
Use of SDPs places a high burden on clinicians, requiring them to learn and competently implement multiple different manualized interventions for each anxiety, mood, or emotional disorder encountered in practice.4 It is common for individuals who present with an anxiety, mood, or emotional disorder to experience co-occurring disorders.5 Traditional SDP-based CBT may require multiple SDPs to address co-occurring disorders, extending time in treatment and increasing training burden. There is evidence that even when an SDP is used, co-occurring difficulties may decrease in intensity.6 Thus, evidence-based CBT for 1 presenting concern may positively affect co-occurring difficulties that are not the primary treatment target.6
Unified Protocol (UP) is a transdiagnostic CBT intervention for anxiety and mood disorders. UP targets emotional experiences (eg, negative affect and emotional distress) that are present in multiple anxiety, depression, and emotional disorders. UP is organized into 8 modules and uses interventions present in other CBT SDPs, such as teaching objective assessment methods to clients, motivational enhancement and goal setting, emotion psychoeducation, mindful awareness, cognitive reframing, and exposure principles as mechanisms of change (Table 1).4 UP is an ideal intervention for addressing a number of anxiety, mood, and emotional disorders as well as addressing cooccurring disorders within the same course of treatment.
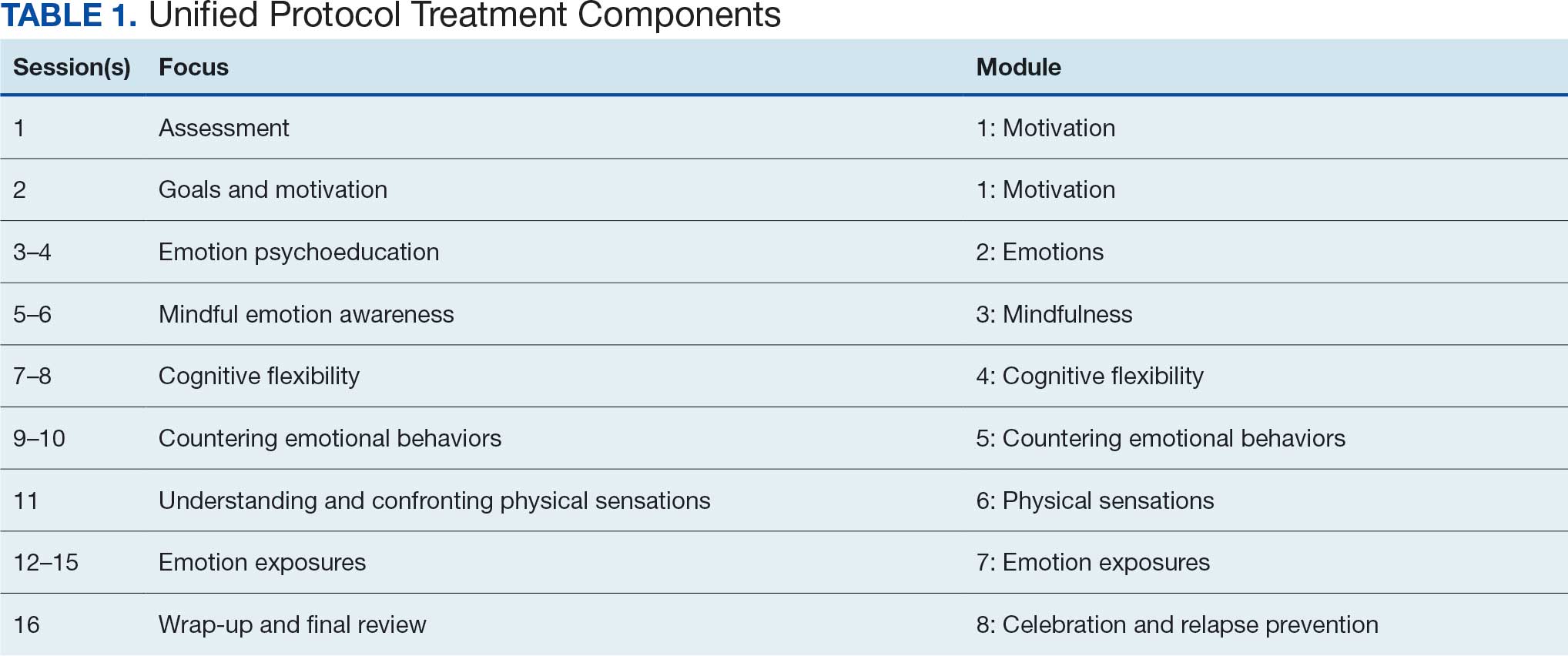
UP has been compared to SDPs; studies illustrate that UP is equivalent to SDPs at the end of treatment and at 6 months posttreatment.4,7 Additionally, patients who received UP experienced symptom reduction for multiple co-occurring disorders.7 Furthermore, patients were less likely to drop out of UP when compared with SDPs.4
Studies have reported positive impacts on affective disorders with UP. A 2019 meta-analysis found significant reductions in depression and anxiety-related disorders.8 Additionally, UP has been shown to be effective when delivered in person and via telehealth.9,10 UP has also been successfully used in veterans.11 While traditional models of UP (1-2 sessions weekly) have a lower dropout rate than SDPs, UP and CBT dropout rates still leave room for improvement.12-14 Specifically, rates of attrition from SDP CBT and transdiagnostic CBT protocols range from 9% to 35%, and dropout reduces the likelihood of attaining a full therapeutic dose of any course of CBT. Notably, accelerated delivery of CBT (ie, ≥ 3 sessions/wk) has been shown to reduce risk of dropout.14,15
Veterans are at increased risk for suicide, and anxiety and mood disorders are associated with increased risk of suicide attempt and death.16,17 Very few veterans who could benefit from high-quality CBT interventions, whether SDP or UP, are able to access them. Only 6.3% of veterans received ≥1 session of an evidence-based CBT SDP for posttraumatic stress disorder (PTSD) at 6 clinics evaluated in 2010.18
We identified 2 strategies to increase access to and completion of an evidence-based CBT course. First, the use of transdiagnostic UP instead of SDPs reduced burden on therapists and allowed them to address presenting and co-occurring disorders within the same course of care. Second, the use of an accelerated model of service delivery reduced dropout risk. Training clinicians to deliver UP is efficient and cost-effective, considering clinicians use core strategies that can be tailored and flexibly applied to a range of emotional difficulties. Thus, implementing UP may decrease barriers to receiving an optimal dose of an evidence-based CBT delivered with fidelity in a time- and cost-efficient manner.4 Two studies have found no evidence of differences in outcomes between UP and SDPs, suggesting that training and supervising clinicians in a single transdiagnostic UP intervention may prepare them to treat heterogeneous and co-occurring anxiety, mood, and emotional disorders with less burden than learning multiple SDPs.7,19
Delivering UP in an accelerated or massed format (≥4 sessions/wk) instead of the traditional spaced model (1 session/wk) has empirical support but has not been widely implemented. This approach, sometimes referred to as a UP-intensive outpatient program (UP-IOP) or UP-intensive outpatient track (UP-IOT), has been shown to be feasible, acceptable, and effective, with increased completion rates compared with traditional UP delivery (1-2 sessions/wk).20-22
Ragsdale et al describe a 2-week IOP with multiple treatment tracks, including a general track.20 The general track includes massed UP and additional standard services, including case management, wellness services, family services, and a single session effective behaviors group. Additional augmentation services are available when clinically indicated (eg, repetitive transcranial magnetic stimulation, transcranial direct current stimulation, psychoeducation, motivational interviewing, relapse prevention). In other words, this was an UP-IOP.20
Thompson-Brenner et al described a successful implementation of massed UP applied in intensive treatment settings, such as residential and day-hospital programs, for eating disorders. Patients reported improvements in 3 UP targets: experiential avoidance, mindfulness, and anxiety.21 Watkins et al evaluated a 2-week IOP using CBT for comorbid substance use and mental health disorders, including prolonged exposure, UP, and relapse prevention for substance use disorders. Participants were post-9/11 veterans and activeduty personnel. Results indicated that UP reduced PTSD and depressive symptoms following treatment. Furthermore, the retention rate (91%) was higher than retention in outpatient treatment (39%-65%), supporting the IOP model as a strategy to reduce dropout.22
Massed psychotherapy has been extended to IOP programs for PTSD treatment within the Veterans Health Administration (VHA). Yamokoski et al found that patients who completed an IOP that included massed CBT for PTSD had high retention, high satisfaction, and significant reduction in self-reported co-occurring depression symptoms. The authors also found that this model of care could be implemented and sustained within a VHA facility using minimal staffing resources.23
The UP-IOP models described by Ragsdale et al and Watkins et al included massed UP as the primary evidence-based practice (EBP) with adjunctive groups (eg, wellness and educational groups and access to complementary interventions such as mindfulness and yoga); they found that adding these groups increased retention and patient- reported satisfaction (ie, UP-IOP).20,22 The addition of wellness education alongside a primary EBP aligns with the VHA focus on whole health well-being and wellness. This includes understanding factors that motivate a patient toward health and well-being, providing health education, and offering access to complementary interventions such as mindfulness.24
Dryden et al described the whole health transformation within VHA as a proactive approach to addressing both employee and patient wellness. Their research found that the whole health model promoted wellbeing in patients and staff and these improvements were sustained during the COVID-19 pandemic. Dryden et al also noted that virtual technologies facilitated continued whole health implementation.25
The literature illustrates that (1) massed UP can be delivered with complementary education and wellness offerings that may increase retention and satisfaction by enriching treatment (eg, delivering UP-IOP); (2) whole health, including wellness education and complementary interventions (eg, mindfulness, motivational enhancement), promotes well-being in patients and clinicians; and (3) whole health education and complementary interventions can be delivered virtually.
IVET
Health Care Need
Veterans Affairs Pacific Islands Health Care System (VAPIHCS) provides medical services to veterans in the Hawaiian Islands, American Samoa, Guam, and Saipan spanning nearly 4000 miles across the Pacific Ocean. Prior to implementation of this program, veterans who received care at VAPIHCS had little to no access to UP in outpatient settings and no access to UP in residential settings. Access to UP depended on the presence of a therapist trained in UP within a given clinic and was geographically limited to the location of the UP-trained therapist. The limited outpatient access to UP was restricted to the traditional UP delivery model (eg, about 1 session/wk); thus, there was no access to accelerated UP for veterans served at VAPIHCS. In the fiscal year prior to implementation of the massed UP program, > 1000 VAPIHCS veterans had been diagnosed with obsessive-compulsive related disorder or anxiety. A massed-UP program with weekly rolling admissions would support access to UP for more veterans over 12 months, and the virtual treatment modality would reduce barriers for diverse and underserved veterans, making care more equitable and inclusive.
Successful implementation and sustainment of an EBP prompted the establishment of this UP program. In 2022, VAPIHCS launched the Intensive Virtual Evidence- Based Psychotherapy Team (iVET) for treatment of PTSD. This clinic and associated EBP project demonstrated that massed (≥3 individual sessions/wk) of prolonged exposure (PE) therapy, delivered virtually to a geographically diverse veteran population with PTSD, resulted in significant reductions in PTSD, depression, and anxiety symptoms and substance use risk factors, with very high retention rates. The iVET for PTSD program was feasible, acceptable, and effective, with veterans reporting significant improvement in quality of life and high satisfaction with their mental health services.15 Given the known benefits of transdiagnostic UP treatment (vs SDPs), the need for accelerated UP, and success with accelerated PE, our goal was to spread the EBP of massed (≥3 sessions/ wk) virtual psychotherapy to other presenting problems (eg, anxiety disorders with or without co-occurring unipolar depression) using transdiagnostic UP.
Program Description
The program implemented within outpatient mental health services at VAPIHCS was iVET for the treatment of anxiety with or without co-occurring depression. The program model consists of an accelerated course of UP and whole health education provided via VA Video Connect (VVC), the VA video telehealth platform. iVET is a 2- to 4-week program and consists of 3 parts: (1) massed individual UP for transdiagnostic treatment of anxiety and co-occurring depression, (2) group whole health and wellness classes, and (3) individual health coaching to address personal wellness goals. Programming is offered over 10-hour days to increase access across multiple time zones, especially to allow participation from Guam and Saipan.
When a patient is referred to iVET, the first contact is a video (or telephone) appointment with a registered nurse (RN) for a screening session. The screening session is designed to provide education about the program (including interventions, time commitment, and resources required for participation). Following education, the RN completes a safety screening, including screening for suicidal ideation and risk, as well as intimate partner violence risk. If urgent safety concerns are present, a licensed social worker or psychologist joins the screening to complete further risk assessment and address any safety concerns.
Following screening, patients are scheduled for a VVC telehealth intake with a licensed therapist (social worker or psychologist) to complete a diagnostic interview. Patients are sent a secure link to complete a measurement-based care (MBC) battery of self-report measures, including assessments of demographics, anxiety symptoms, depression symptoms, substance use, psychological flexibility, quality of life, and satisfaction with mental health care. The results of the diagnostic interview and self-report measures are discussed with the patient during the intake session to plan next steps and support shared decision-making. This initial VVC intake not only allows for fit assessment but also serves to troubleshoot technical difficulties with the virtual platforms.
Notably, there are minimal exclusion criteria for participation in iVET. These include active unmanaged psychosis or manic symptoms, recent suicidal crises (attempt within 8 weeks), active nonsuicidal self-injury (within 8 weeks), and moderate to severe cognitive impairment. Following intake, patients are scheduled to begin their course of care with iVET. Upon completion of intake, patients receive program materials for individual and group classes and are told they will receive email links for all VVC telehealth appointments. Patients are admitted to the iVET on a rolling basis, thereby increasing access compared with closed group and/or cohort models of care.
Patients receiving iVET attend 2 to 4 telehealth appointments daily and complete exercises between sessions. The primary iVET for Anxiety program EBP intervention is a massed or accelerated individual course of UP, which includes 8 primary components: assessment, goals and motivation, understanding emotions, mindful emotion awareness, cognitive flexibility, countering emotional behaviors, understanding and confronting physical sensations, and emotional exposures. UP is delivered in 4 to 8 individual sessions weekly (60-90 minutes each), allowing completion of the full UP protocol with fidelity in 2 to 4 weeks. In addition to primary EBP intervention, patients participate in 4 group sessions weekly (50 minutes each) of a whole health and wellness education class and have access to 1 individual health coaching session weekly (30- 60 minutes) to set wellness goals and receive coaching. During iVET, patients are invited to complete MBC batteries of self-report measures assessing anxiety symptoms, depression symptoms, substance use, psychological flexibility, quality of life, and satisfaction with mental health care at sessions 1, 5, 9, 13, and the final UP session. Following discharge from the iVET, patients are offered 1-, 3-, and 6-month individual postdischarge check-up sessions with a therapist, during which they are invited to complete MBC measures and review relapse prevention and maintenance of treatment gains. Likewise, patients are offered 1-, 3-, and 6-month individual postdischarge check-up sessions with an RN focused on maintaining wellness gains.
The iVET for Anxiety staff has 3 therapists (psychologists or social workers) and 1 RN. Additionally, the iVET for Anxiety is supported by a program manager and a program support assistant who support 2 programs total (the iVET for Anxiety plus another mental health program). The primary cost of the program is staff salary. Additional resources included computer equipment for staff and supplies (eg, printed materials for patients and office supplies). Due to the virtual environment of care, iVET staff telework and do not require physical space within VAPIHCS.
Outcomes
Veterans receiving iVET for Anxiety are invited to complete MBC multiple times, including pretreatment, during UP treatment (sessions 1, 5, 9, 13, and the final session), and posttreatment (1, 3, and 6 months). MBC measures include self-reported demographics; a 2-item measure of satisfaction with mental health services; the Acceptance and Action Questionnaire II,26 the Brief Addiction Monitor-Intensive Outpatient Program, 27 the Generalized Anxiety Disorder-7,28 the Patient Health Questionnaire (PHQ-9),29 and the Quality of Life Enjoyment and Satisfaction Questionnaire-Short Form.30
Forty-two veterans completed the iVET for Anxiety program, with a retention rate of 87.5% completing . 16 sessions of massed UP (Table 2). Veterans reported reduced anxiety (P < .001), depression (P < .001), and substance use risk (P < .001). Veterans experienced improved acceptance of emotional experiences (P < .001) and quality of life (P < .001), based on paired sample t tests comparing session 1 vs final session scores on the self-reported measures. Veterans also reported high satisfaction with mental health care in iVET for Anxiety.
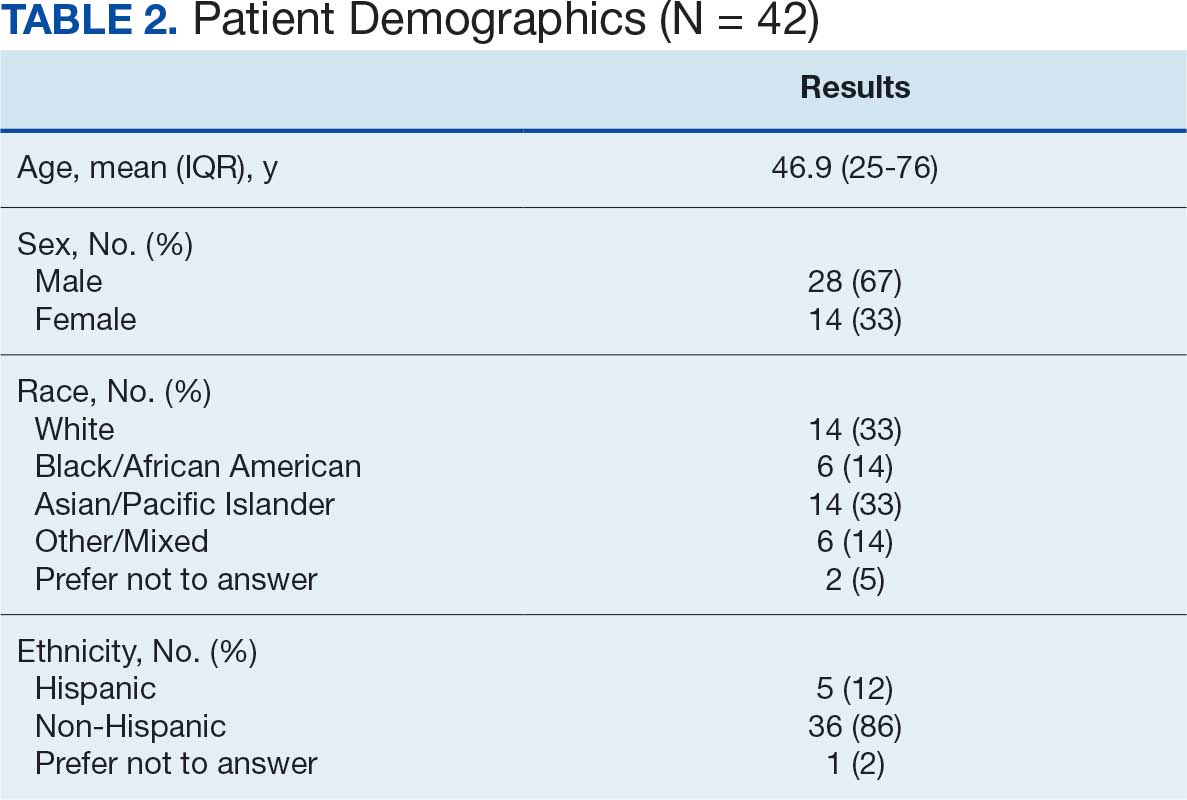
Veterans experienced reduced thoughts of death or suicidal ideation (SI) based on responses to item 9 of PHQ-9. When SI was categorically analyzed (presence vs absence) using PHQ-9 item 9, there was an association observed between absence of SI and completion of a course of massed UP that does not appear to be due to chance, (X2 [1, N = 42) = 3.94; P = .047). In addition, veterans who completed the program showed a significant decrease in SI severity measured continuously (range, 0-3) on PHQ-9 item 9 (P < .05) (Table 3).
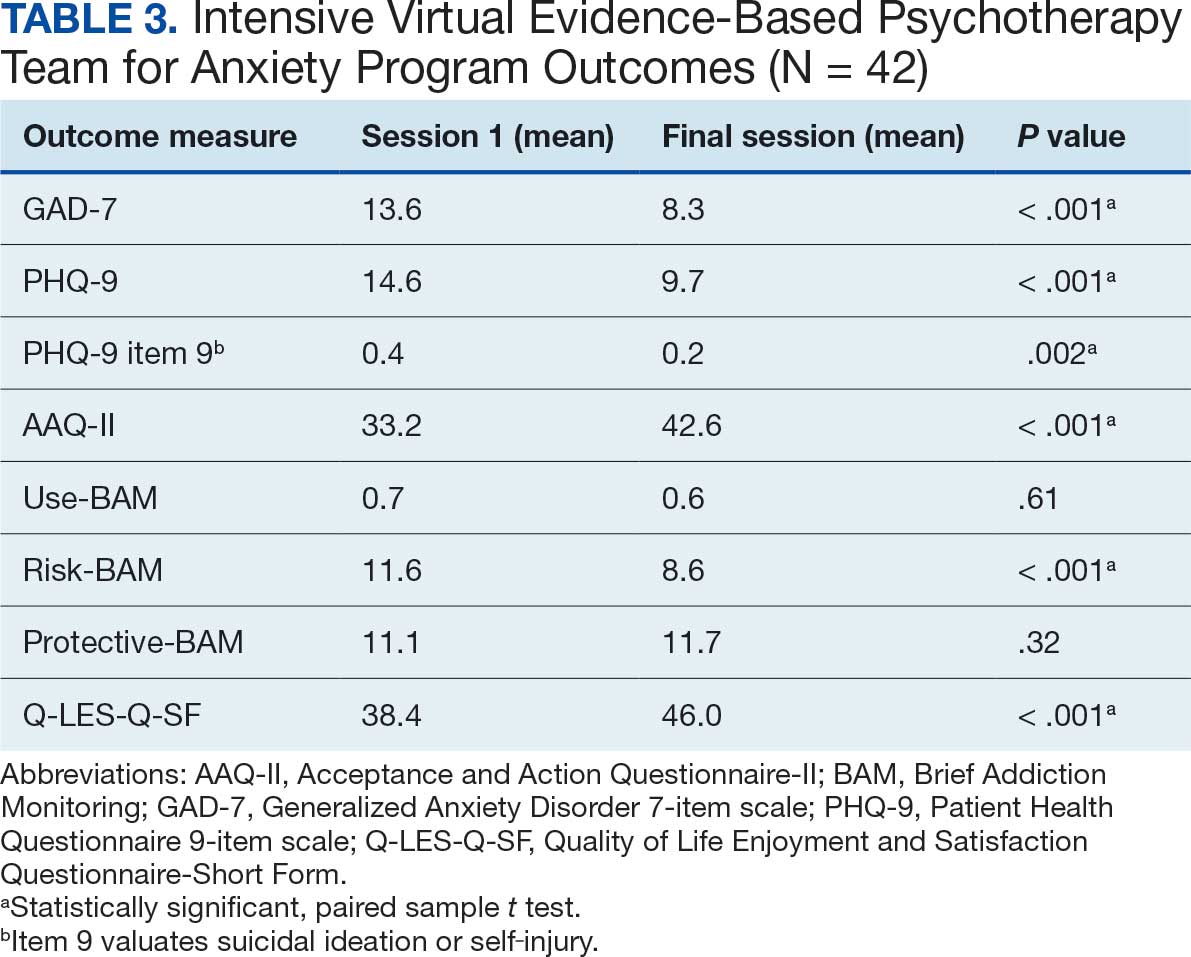
Future Directions
The aim of this program is to see sustained patient outcomes as iVET continues to serve more veterans. Another line of inquiry is longer- term follow-up, given that long-term follow- up was not addressed in this project. We also hope that the accelerated model of care can be applied to treatment of other presenting concerns (eg, relationship difficulties, insomnia). Expansion of accelerated mental health treatment into other federal and nonfederal health care settings is another area worthy of future inquiry. Exploration of staff satisfaction and burnout related to providing accelerated UP is another important future direction. Relatedly, assessment of the staff burden to learn 1 transdiagnostic EBP vs learning multiple SDPs is another future direction. Likewise, exploration of institutional benefits of investment in transdiagnostic training, supervision, and consultation for UP vs multiple SDPs may be important. These areas could also result in insightful, beneficial evidence of the effectiveness of massed UP to add to the existing literature.
Conclusions
UP for transdiagnostic treatment of anxiety, depressive, and emotional disorders has demonstrated reduced suffering and improved functioning and is supported by multiple clinical practice guidelines.1-4 Federal practitioners are positioned to improve access to this intervention, thereby reducing pain and improving lives. Indeed, it is crucial to envision a future state in which access to UP for a range of anxiety and depressive disorders is improved and broad, retention rates are dramatically improved, and clinicians providing UP do not experience the high burden and burnout associated with needing to learn and implement a variety of SDPs. Development of these programs, or similar tracks within existing programs, that provide massed or accelerated UP for transdiagnostic treatment of a range of anxiety and depressive disorders with virtual delivery options, is imperative to advance improved care for patients and clinicians.
Federal health care settings treating patients with anxiety and depression, such as those within the US Department of Defense, Indian Health Services, Bureau of Prisons, and VHA, are positioned to implement programs like iVET. Moreover, at the institutional level, investment in training and supervision in the transdiagnostic UP as opposed to multiple SDPs warrants consideration. We believe this model of care has great merit and foresee a future where all patients seeking treatment for anxiety and depression have the option to complete an accelerated or massed course of transdiagnostic care with UP if they so desire. Our experiences with iVET illustrate the feasibility, acceptability, and sustainability of such programs without requiring substantial staffing and financial resources.
- US Department of Veterans Affairs, US Department of Defense. VA/DoD clinical practice guideline for the management of major depressive disorder. Version 4. 2022. Accessed February 1, 2026. https://www.healthquality.va.gov/guidelines/MH/mdd/VADoDMDDCPGFinal508.pdf
- American Psychological Association. Clinical practice guideline for the treatment of depression across three age cohorts. February 2019. Accessed February 4, 2026. https://www.apa.org/depression-guideline/guideline.pdf
- Katzman MA, Bleau P, Blier P, et al. Canadian clinical practice guidelines for the management of anxiety, posttraumatic stress and obsessive compulsive disorders. BMC Psychiatry. 2014;14:S1. doi:10.1186/1471-244X-14-S1-S1
- Barlow DH, Farchione TJ, Bullis JR, et al. The unified protocol for transdiagnostic treatment of emotional disorders compared with diagnosis-specific protocols for anxiety disorders: a randomized clinical trial. JAMA Psychiatry. 2017;74:875-884. doi:10.1001/jamapsychiatry.2017.2164
- Calkins AW, et al. Comorbidity of anxiety and depression. In: Ressler KJ, Pine DS, Rothbaum BO, eds. Anxiety Disorders, Primer On. Oxford Academic; 2015. https://doi.org/10.1093/med/9780199395125.003.0021
- Manber R, Edinger JD, Gress JL, et al. Cognitive behavioral therapy for insomnia enhances depression outcome in patients with comorbid major depressive disorder and insomnia. Sleep. 2008;31:489-495. doi:10.1093/sleep/31.4.489
- Steele SJ, Farchione TJ, Cassiello-Robbins C, et al. Efficacy of the Unified Protocol for transdiagnostic treatment of comorbid psychopathology accompanying emotional disorders compared to treatments targeting single disorders. J Psychiatr Res. 2018;104:211-216. doi:10.1016/j.jpsychires.2018.08.005
- Sakiris N, Berle D. A systematic review and meta-analysis of the Unified Protocol as a transdiagnostic emotion regulation based intervention. Clin Psychol Rev. 2019;72:101751. doi:10.1016/j.cpr.2019.101751
- Cassiello-Robbins C, Rosenthal MZ, Ammirati RJ. Delivering transdiagnostic treatment over telehealth during the COVID-19 pandemic: application of the unified protocol. Cogn Behav Pract. 2021;28:555-572. doi:10.1016/j.cbpra.2021.04.007
- Meyer EC, Coe E, Pennington ML, et al. The unified protocol for transdiagnostic treatment of emotional disorders delivered to firefighters via videoconferencing: pilot outcomes highlighting improvements in alcohol use disorder and posttraumatic stress disorder symptoms. Cogn Behav Pract. 2024;31:215-229. doi:10.1016/j.cbpra.2022.08.004
- Varkovitzky RL, Sherrill AM, Reger GM. Effectiveness of the unified protocol for transdiagnostic treatment of emotional disorders among veterans with posttraumatic stress disorder: a pilot study. Behav Modif. 2018;42:210-230. doi:10.1177/0145445517724539
- Oliveira JT, Sousa I, Ribeiro AP, et al. Premature termination of the unified protocol for the transdiagnostic treatment of emotional disorders: The role of ambivalence towards change. Clin Psychol Psychother. 2022;29:1089-1100. doi:10.1002/cpp.2694
- Schaeuffele C, Homeyer S, Perea L, et al. The unified protocol as an internet-based intervention for emotional disorders: Randomized controlled trial. PLoS One. 2022;17:e0270178. doi:10.1371/journal.pone.0270178
- Bentley KH, Cohen ZD, Kim T, et al. The nature, timing, and symptom trajectories of dropout from transdiagnostic and single-diagnosis cognitive-behavioral therapy for anxiety disorders. Behav Ther. 2021;52:1364-1376. doi:10.1016/j.beth.2021.03.007
- Aosved AC, Brown TB, Bell JC, et al. Accelerated prolonged exposure therapy for posttraumatic stress disorder in a Veterans Health Administration system. Fed Pract. 2025;42:S6-S11. doi:10.12788/fp.0568
- Nepon J, Belik SL, Bolton J, et al. The relationship between anxiety disorders and suicide attempts: findings from the National Epidemiologic Survey on Alcohol and Related Conditions. Depress Anxiety. 2010;27:791-798. doi:10.1002/da.20674
- Shiner B, D’Avolio LW, Nguyen TM, et al. Measuring use of evidence based psychotherapy for posttraumatic stress disorder. Adm Policy Ment Health. 2013;40:311-318. doi:10.1007/s10488-012-0421-0
- Nichter B, Stein MB, Monteith LL, et al. Risk factors for suicide attempts among U.S. military veterans: A 7-year population-based, longitudinal cohort study. Suicide Life Threat Behav. 2022;52:303-316. doi:10.1111/sltb.12822
- McHugh RK, Barlow DH. The dissemination and implementation of evidence-based psychological treatments. A review of current efforts. Am Psychol. 2010;65:73-84. doi:10.1037/a0018121
- Ragsdale KA, Nichols AA, Mehta M, et al. Comorbid treatment of traumatic brain injury and mental health disorders. NeuroRehabilitation. 2024;55:375-384. doi:10.3233/NRE-230235
- Thompson-Brenner H, Brooks GE, Boswell JF, et al. Evidence-based implementation practices applied to the intensive treatment of eating disorders: summary of research and illustration of principles using a case example. Clin Psychol Sci Pract. 2018;25:e12221. doi:10.1111/cpsp.12221
- Watkins LE, Patton SC, Drexler K, et al. Clinical effectiveness of an intensive outpatient program for integrated treatment of comorbid substance abuse and mental health disorders. Cog Behav Pract. 2023;30:354-366.
- Yamokoski C, Flores H, Facemire V, et al. Feasibility of an intensive outpatient treatment program for post-traumatic stress disorder within the veterans health care administration. Psychol Serv. 2023;20:506-515. doi:10.1037/ser0000628
- Gaudet T, Kligler B. Whole health in the whole system of the Veterans Administration: how will we know we have reached this future state?. J Altern Complement Med. 2019;25:S7-S11. doi:10.1089/acm.2018.29061.gau
- Dryden EM, Bolton RE, Bokhour BG, et al. Leaning into whole health: sustaining system transformation while supporting patients and employees during COVID-19. Glob Adv Health Med. 2021;10:21649561211021047. doi:10.1177/21649561211021047
- Bond FW, Hayes SC, Baer RA, et al. Preliminary psychometric properties of the Acceptance and Action Questionnaire-II: a revised measure of psychological inflexibility and experiential avoidance. Behav Ther. 2011;42:676-688. doi:10.1016/j.beth.2011.03.007
- Cacciola JS, Alterman AI, Dephilippis D, et al. Development and initial evaluation of the Brief Addiction Monitor (BAM). J Subst Abuse Treat. 2013;44:256-263. doi:10.1016/j.jsat.2012.07.013
- Spitzer RL, Kroenke K, Williams JB, et al. A brief measure for assessing generalized anxiety disorder: the GAD-7. Arch Intern Med. 2006;166:1092-1097. doi:10.1001/archinte.166.10.1092
- Kroenke K, Spitzer RL, Williams JB. The PHQ-9: validity of a brief depression severity measure. J Gen Intern Med. 2001;16:606-613. doi:10.1046/j.1525-1497.2001.016009606.x
- Stevanovic D. Quality of life enjoyment and satisfaction questionnaire-short form for quality of life assessments in clinical practice: a psychometric study. J Psychiatr Ment Health Nurs. 2011;18:744-750. doi:10.1111/j.1365-2850.2011.01735.x
Cognitive behavioral therapy (CBT) is supported as an effective treatment for depression by clinical practice guidelines from the US Department of Veterans Affairs (VA) and US Department of Defense.1-3 Despite research supporting the use of evidence-based CBT for anxiety, mood, and emotional disorders, patient access to these interventions is limited.4 One barrier to CBT for anxiety, mood, and emotional disorders is the traditional use of single-disorder protocols (SDPs) to separately treat each disorder (eg, major depression, generalized anxiety disorder, panic disorder).
Use of SDPs places a high burden on clinicians, requiring them to learn and competently implement multiple different manualized interventions for each anxiety, mood, or emotional disorder encountered in practice.4 It is common for individuals who present with an anxiety, mood, or emotional disorder to experience co-occurring disorders.5 Traditional SDP-based CBT may require multiple SDPs to address co-occurring disorders, extending time in treatment and increasing training burden. There is evidence that even when an SDP is used, co-occurring difficulties may decrease in intensity.6 Thus, evidence-based CBT for 1 presenting concern may positively affect co-occurring difficulties that are not the primary treatment target.6
Unified Protocol (UP) is a transdiagnostic CBT intervention for anxiety and mood disorders. UP targets emotional experiences (eg, negative affect and emotional distress) that are present in multiple anxiety, depression, and emotional disorders. UP is organized into 8 modules and uses interventions present in other CBT SDPs, such as teaching objective assessment methods to clients, motivational enhancement and goal setting, emotion psychoeducation, mindful awareness, cognitive reframing, and exposure principles as mechanisms of change (Table 1).4 UP is an ideal intervention for addressing a number of anxiety, mood, and emotional disorders as well as addressing cooccurring disorders within the same course of treatment.

UP has been compared to SDPs; studies illustrate that UP is equivalent to SDPs at the end of treatment and at 6 months posttreatment.4,7 Additionally, patients who received UP experienced symptom reduction for multiple co-occurring disorders.7 Furthermore, patients were less likely to drop out of UP when compared with SDPs.4
Studies have reported positive impacts on affective disorders with UP. A 2019 meta-analysis found significant reductions in depression and anxiety-related disorders.8 Additionally, UP has been shown to be effective when delivered in person and via telehealth.9,10 UP has also been successfully used in veterans.11 While traditional models of UP (1-2 sessions weekly) have a lower dropout rate than SDPs, UP and CBT dropout rates still leave room for improvement.12-14 Specifically, rates of attrition from SDP CBT and transdiagnostic CBT protocols range from 9% to 35%, and dropout reduces the likelihood of attaining a full therapeutic dose of any course of CBT. Notably, accelerated delivery of CBT (ie, ≥ 3 sessions/wk) has been shown to reduce risk of dropout.14,15
Veterans are at increased risk for suicide, and anxiety and mood disorders are associated with increased risk of suicide attempt and death.16,17 Very few veterans who could benefit from high-quality CBT interventions, whether SDP or UP, are able to access them. Only 6.3% of veterans received ≥1 session of an evidence-based CBT SDP for posttraumatic stress disorder (PTSD) at 6 clinics evaluated in 2010.18
We identified 2 strategies to increase access to and completion of an evidence-based CBT course. First, the use of transdiagnostic UP instead of SDPs reduced burden on therapists and allowed them to address presenting and co-occurring disorders within the same course of care. Second, the use of an accelerated model of service delivery reduced dropout risk. Training clinicians to deliver UP is efficient and cost-effective, considering clinicians use core strategies that can be tailored and flexibly applied to a range of emotional difficulties. Thus, implementing UP may decrease barriers to receiving an optimal dose of an evidence-based CBT delivered with fidelity in a time- and cost-efficient manner.4 Two studies have found no evidence of differences in outcomes between UP and SDPs, suggesting that training and supervising clinicians in a single transdiagnostic UP intervention may prepare them to treat heterogeneous and co-occurring anxiety, mood, and emotional disorders with less burden than learning multiple SDPs.7,19
Delivering UP in an accelerated or massed format (≥4 sessions/wk) instead of the traditional spaced model (1 session/wk) has empirical support but has not been widely implemented. This approach, sometimes referred to as a UP-intensive outpatient program (UP-IOP) or UP-intensive outpatient track (UP-IOT), has been shown to be feasible, acceptable, and effective, with increased completion rates compared with traditional UP delivery (1-2 sessions/wk).20-22
Ragsdale et al describe a 2-week IOP with multiple treatment tracks, including a general track.20 The general track includes massed UP and additional standard services, including case management, wellness services, family services, and a single session effective behaviors group. Additional augmentation services are available when clinically indicated (eg, repetitive transcranial magnetic stimulation, transcranial direct current stimulation, psychoeducation, motivational interviewing, relapse prevention). In other words, this was an UP-IOP.20
Thompson-Brenner et al described a successful implementation of massed UP applied in intensive treatment settings, such as residential and day-hospital programs, for eating disorders. Patients reported improvements in 3 UP targets: experiential avoidance, mindfulness, and anxiety.21 Watkins et al evaluated a 2-week IOP using CBT for comorbid substance use and mental health disorders, including prolonged exposure, UP, and relapse prevention for substance use disorders. Participants were post-9/11 veterans and activeduty personnel. Results indicated that UP reduced PTSD and depressive symptoms following treatment. Furthermore, the retention rate (91%) was higher than retention in outpatient treatment (39%-65%), supporting the IOP model as a strategy to reduce dropout.22
Massed psychotherapy has been extended to IOP programs for PTSD treatment within the Veterans Health Administration (VHA). Yamokoski et al found that patients who completed an IOP that included massed CBT for PTSD had high retention, high satisfaction, and significant reduction in self-reported co-occurring depression symptoms. The authors also found that this model of care could be implemented and sustained within a VHA facility using minimal staffing resources.23
The UP-IOP models described by Ragsdale et al and Watkins et al included massed UP as the primary evidence-based practice (EBP) with adjunctive groups (eg, wellness and educational groups and access to complementary interventions such as mindfulness and yoga); they found that adding these groups increased retention and patient- reported satisfaction (ie, UP-IOP).20,22 The addition of wellness education alongside a primary EBP aligns with the VHA focus on whole health well-being and wellness. This includes understanding factors that motivate a patient toward health and well-being, providing health education, and offering access to complementary interventions such as mindfulness.24
Dryden et al described the whole health transformation within VHA as a proactive approach to addressing both employee and patient wellness. Their research found that the whole health model promoted wellbeing in patients and staff and these improvements were sustained during the COVID-19 pandemic. Dryden et al also noted that virtual technologies facilitated continued whole health implementation.25
The literature illustrates that (1) massed UP can be delivered with complementary education and wellness offerings that may increase retention and satisfaction by enriching treatment (eg, delivering UP-IOP); (2) whole health, including wellness education and complementary interventions (eg, mindfulness, motivational enhancement), promotes well-being in patients and clinicians; and (3) whole health education and complementary interventions can be delivered virtually.
IVET
Health Care Need
Veterans Affairs Pacific Islands Health Care System (VAPIHCS) provides medical services to veterans in the Hawaiian Islands, American Samoa, Guam, and Saipan spanning nearly 4000 miles across the Pacific Ocean. Prior to implementation of this program, veterans who received care at VAPIHCS had little to no access to UP in outpatient settings and no access to UP in residential settings. Access to UP depended on the presence of a therapist trained in UP within a given clinic and was geographically limited to the location of the UP-trained therapist. The limited outpatient access to UP was restricted to the traditional UP delivery model (eg, about 1 session/wk); thus, there was no access to accelerated UP for veterans served at VAPIHCS. In the fiscal year prior to implementation of the massed UP program, > 1000 VAPIHCS veterans had been diagnosed with obsessive-compulsive related disorder or anxiety. A massed-UP program with weekly rolling admissions would support access to UP for more veterans over 12 months, and the virtual treatment modality would reduce barriers for diverse and underserved veterans, making care more equitable and inclusive.
Successful implementation and sustainment of an EBP prompted the establishment of this UP program. In 2022, VAPIHCS launched the Intensive Virtual Evidence- Based Psychotherapy Team (iVET) for treatment of PTSD. This clinic and associated EBP project demonstrated that massed (≥3 individual sessions/wk) of prolonged exposure (PE) therapy, delivered virtually to a geographically diverse veteran population with PTSD, resulted in significant reductions in PTSD, depression, and anxiety symptoms and substance use risk factors, with very high retention rates. The iVET for PTSD program was feasible, acceptable, and effective, with veterans reporting significant improvement in quality of life and high satisfaction with their mental health services.15 Given the known benefits of transdiagnostic UP treatment (vs SDPs), the need for accelerated UP, and success with accelerated PE, our goal was to spread the EBP of massed (≥3 sessions/ wk) virtual psychotherapy to other presenting problems (eg, anxiety disorders with or without co-occurring unipolar depression) using transdiagnostic UP.
Program Description
The program implemented within outpatient mental health services at VAPIHCS was iVET for the treatment of anxiety with or without co-occurring depression. The program model consists of an accelerated course of UP and whole health education provided via VA Video Connect (VVC), the VA video telehealth platform. iVET is a 2- to 4-week program and consists of 3 parts: (1) massed individual UP for transdiagnostic treatment of anxiety and co-occurring depression, (2) group whole health and wellness classes, and (3) individual health coaching to address personal wellness goals. Programming is offered over 10-hour days to increase access across multiple time zones, especially to allow participation from Guam and Saipan.
When a patient is referred to iVET, the first contact is a video (or telephone) appointment with a registered nurse (RN) for a screening session. The screening session is designed to provide education about the program (including interventions, time commitment, and resources required for participation). Following education, the RN completes a safety screening, including screening for suicidal ideation and risk, as well as intimate partner violence risk. If urgent safety concerns are present, a licensed social worker or psychologist joins the screening to complete further risk assessment and address any safety concerns.
Following screening, patients are scheduled for a VVC telehealth intake with a licensed therapist (social worker or psychologist) to complete a diagnostic interview. Patients are sent a secure link to complete a measurement-based care (MBC) battery of self-report measures, including assessments of demographics, anxiety symptoms, depression symptoms, substance use, psychological flexibility, quality of life, and satisfaction with mental health care. The results of the diagnostic interview and self-report measures are discussed with the patient during the intake session to plan next steps and support shared decision-making. This initial VVC intake not only allows for fit assessment but also serves to troubleshoot technical difficulties with the virtual platforms.
Notably, there are minimal exclusion criteria for participation in iVET. These include active unmanaged psychosis or manic symptoms, recent suicidal crises (attempt within 8 weeks), active nonsuicidal self-injury (within 8 weeks), and moderate to severe cognitive impairment. Following intake, patients are scheduled to begin their course of care with iVET. Upon completion of intake, patients receive program materials for individual and group classes and are told they will receive email links for all VVC telehealth appointments. Patients are admitted to the iVET on a rolling basis, thereby increasing access compared with closed group and/or cohort models of care.
Patients receiving iVET attend 2 to 4 telehealth appointments daily and complete exercises between sessions. The primary iVET for Anxiety program EBP intervention is a massed or accelerated individual course of UP, which includes 8 primary components: assessment, goals and motivation, understanding emotions, mindful emotion awareness, cognitive flexibility, countering emotional behaviors, understanding and confronting physical sensations, and emotional exposures. UP is delivered in 4 to 8 individual sessions weekly (60-90 minutes each), allowing completion of the full UP protocol with fidelity in 2 to 4 weeks. In addition to primary EBP intervention, patients participate in 4 group sessions weekly (50 minutes each) of a whole health and wellness education class and have access to 1 individual health coaching session weekly (30- 60 minutes) to set wellness goals and receive coaching. During iVET, patients are invited to complete MBC batteries of self-report measures assessing anxiety symptoms, depression symptoms, substance use, psychological flexibility, quality of life, and satisfaction with mental health care at sessions 1, 5, 9, 13, and the final UP session. Following discharge from the iVET, patients are offered 1-, 3-, and 6-month individual postdischarge check-up sessions with a therapist, during which they are invited to complete MBC measures and review relapse prevention and maintenance of treatment gains. Likewise, patients are offered 1-, 3-, and 6-month individual postdischarge check-up sessions with an RN focused on maintaining wellness gains.
The iVET for Anxiety staff has 3 therapists (psychologists or social workers) and 1 RN. Additionally, the iVET for Anxiety is supported by a program manager and a program support assistant who support 2 programs total (the iVET for Anxiety plus another mental health program). The primary cost of the program is staff salary. Additional resources included computer equipment for staff and supplies (eg, printed materials for patients and office supplies). Due to the virtual environment of care, iVET staff telework and do not require physical space within VAPIHCS.
Outcomes
Veterans receiving iVET for Anxiety are invited to complete MBC multiple times, including pretreatment, during UP treatment (sessions 1, 5, 9, 13, and the final session), and posttreatment (1, 3, and 6 months). MBC measures include self-reported demographics; a 2-item measure of satisfaction with mental health services; the Acceptance and Action Questionnaire II,26 the Brief Addiction Monitor-Intensive Outpatient Program, 27 the Generalized Anxiety Disorder-7,28 the Patient Health Questionnaire (PHQ-9),29 and the Quality of Life Enjoyment and Satisfaction Questionnaire-Short Form.30
Forty-two veterans completed the iVET for Anxiety program, with a retention rate of 87.5% completing . 16 sessions of massed UP (Table 2). Veterans reported reduced anxiety (P < .001), depression (P < .001), and substance use risk (P < .001). Veterans experienced improved acceptance of emotional experiences (P < .001) and quality of life (P < .001), based on paired sample t tests comparing session 1 vs final session scores on the self-reported measures. Veterans also reported high satisfaction with mental health care in iVET for Anxiety.

Veterans experienced reduced thoughts of death or suicidal ideation (SI) based on responses to item 9 of PHQ-9. When SI was categorically analyzed (presence vs absence) using PHQ-9 item 9, there was an association observed between absence of SI and completion of a course of massed UP that does not appear to be due to chance, (X2 [1, N = 42) = 3.94; P = .047). In addition, veterans who completed the program showed a significant decrease in SI severity measured continuously (range, 0-3) on PHQ-9 item 9 (P < .05) (Table 3).

Future Directions
The aim of this program is to see sustained patient outcomes as iVET continues to serve more veterans. Another line of inquiry is longer- term follow-up, given that long-term follow- up was not addressed in this project. We also hope that the accelerated model of care can be applied to treatment of other presenting concerns (eg, relationship difficulties, insomnia). Expansion of accelerated mental health treatment into other federal and nonfederal health care settings is another area worthy of future inquiry. Exploration of staff satisfaction and burnout related to providing accelerated UP is another important future direction. Relatedly, assessment of the staff burden to learn 1 transdiagnostic EBP vs learning multiple SDPs is another future direction. Likewise, exploration of institutional benefits of investment in transdiagnostic training, supervision, and consultation for UP vs multiple SDPs may be important. These areas could also result in insightful, beneficial evidence of the effectiveness of massed UP to add to the existing literature.
Conclusions
UP for transdiagnostic treatment of anxiety, depressive, and emotional disorders has demonstrated reduced suffering and improved functioning and is supported by multiple clinical practice guidelines.1-4 Federal practitioners are positioned to improve access to this intervention, thereby reducing pain and improving lives. Indeed, it is crucial to envision a future state in which access to UP for a range of anxiety and depressive disorders is improved and broad, retention rates are dramatically improved, and clinicians providing UP do not experience the high burden and burnout associated with needing to learn and implement a variety of SDPs. Development of these programs, or similar tracks within existing programs, that provide massed or accelerated UP for transdiagnostic treatment of a range of anxiety and depressive disorders with virtual delivery options, is imperative to advance improved care for patients and clinicians.
Federal health care settings treating patients with anxiety and depression, such as those within the US Department of Defense, Indian Health Services, Bureau of Prisons, and VHA, are positioned to implement programs like iVET. Moreover, at the institutional level, investment in training and supervision in the transdiagnostic UP as opposed to multiple SDPs warrants consideration. We believe this model of care has great merit and foresee a future where all patients seeking treatment for anxiety and depression have the option to complete an accelerated or massed course of transdiagnostic care with UP if they so desire. Our experiences with iVET illustrate the feasibility, acceptability, and sustainability of such programs without requiring substantial staffing and financial resources.
Cognitive behavioral therapy (CBT) is supported as an effective treatment for depression by clinical practice guidelines from the US Department of Veterans Affairs (VA) and US Department of Defense.1-3 Despite research supporting the use of evidence-based CBT for anxiety, mood, and emotional disorders, patient access to these interventions is limited.4 One barrier to CBT for anxiety, mood, and emotional disorders is the traditional use of single-disorder protocols (SDPs) to separately treat each disorder (eg, major depression, generalized anxiety disorder, panic disorder).
Use of SDPs places a high burden on clinicians, requiring them to learn and competently implement multiple different manualized interventions for each anxiety, mood, or emotional disorder encountered in practice.4 It is common for individuals who present with an anxiety, mood, or emotional disorder to experience co-occurring disorders.5 Traditional SDP-based CBT may require multiple SDPs to address co-occurring disorders, extending time in treatment and increasing training burden. There is evidence that even when an SDP is used, co-occurring difficulties may decrease in intensity.6 Thus, evidence-based CBT for 1 presenting concern may positively affect co-occurring difficulties that are not the primary treatment target.6
Unified Protocol (UP) is a transdiagnostic CBT intervention for anxiety and mood disorders. UP targets emotional experiences (eg, negative affect and emotional distress) that are present in multiple anxiety, depression, and emotional disorders. UP is organized into 8 modules and uses interventions present in other CBT SDPs, such as teaching objective assessment methods to clients, motivational enhancement and goal setting, emotion psychoeducation, mindful awareness, cognitive reframing, and exposure principles as mechanisms of change (Table 1).4 UP is an ideal intervention for addressing a number of anxiety, mood, and emotional disorders as well as addressing cooccurring disorders within the same course of treatment.

UP has been compared to SDPs; studies illustrate that UP is equivalent to SDPs at the end of treatment and at 6 months posttreatment.4,7 Additionally, patients who received UP experienced symptom reduction for multiple co-occurring disorders.7 Furthermore, patients were less likely to drop out of UP when compared with SDPs.4
Studies have reported positive impacts on affective disorders with UP. A 2019 meta-analysis found significant reductions in depression and anxiety-related disorders.8 Additionally, UP has been shown to be effective when delivered in person and via telehealth.9,10 UP has also been successfully used in veterans.11 While traditional models of UP (1-2 sessions weekly) have a lower dropout rate than SDPs, UP and CBT dropout rates still leave room for improvement.12-14 Specifically, rates of attrition from SDP CBT and transdiagnostic CBT protocols range from 9% to 35%, and dropout reduces the likelihood of attaining a full therapeutic dose of any course of CBT. Notably, accelerated delivery of CBT (ie, ≥ 3 sessions/wk) has been shown to reduce risk of dropout.14,15
Veterans are at increased risk for suicide, and anxiety and mood disorders are associated with increased risk of suicide attempt and death.16,17 Very few veterans who could benefit from high-quality CBT interventions, whether SDP or UP, are able to access them. Only 6.3% of veterans received ≥1 session of an evidence-based CBT SDP for posttraumatic stress disorder (PTSD) at 6 clinics evaluated in 2010.18
We identified 2 strategies to increase access to and completion of an evidence-based CBT course. First, the use of transdiagnostic UP instead of SDPs reduced burden on therapists and allowed them to address presenting and co-occurring disorders within the same course of care. Second, the use of an accelerated model of service delivery reduced dropout risk. Training clinicians to deliver UP is efficient and cost-effective, considering clinicians use core strategies that can be tailored and flexibly applied to a range of emotional difficulties. Thus, implementing UP may decrease barriers to receiving an optimal dose of an evidence-based CBT delivered with fidelity in a time- and cost-efficient manner.4 Two studies have found no evidence of differences in outcomes between UP and SDPs, suggesting that training and supervising clinicians in a single transdiagnostic UP intervention may prepare them to treat heterogeneous and co-occurring anxiety, mood, and emotional disorders with less burden than learning multiple SDPs.7,19
Delivering UP in an accelerated or massed format (≥4 sessions/wk) instead of the traditional spaced model (1 session/wk) has empirical support but has not been widely implemented. This approach, sometimes referred to as a UP-intensive outpatient program (UP-IOP) or UP-intensive outpatient track (UP-IOT), has been shown to be feasible, acceptable, and effective, with increased completion rates compared with traditional UP delivery (1-2 sessions/wk).20-22
Ragsdale et al describe a 2-week IOP with multiple treatment tracks, including a general track.20 The general track includes massed UP and additional standard services, including case management, wellness services, family services, and a single session effective behaviors group. Additional augmentation services are available when clinically indicated (eg, repetitive transcranial magnetic stimulation, transcranial direct current stimulation, psychoeducation, motivational interviewing, relapse prevention). In other words, this was an UP-IOP.20
Thompson-Brenner et al described a successful implementation of massed UP applied in intensive treatment settings, such as residential and day-hospital programs, for eating disorders. Patients reported improvements in 3 UP targets: experiential avoidance, mindfulness, and anxiety.21 Watkins et al evaluated a 2-week IOP using CBT for comorbid substance use and mental health disorders, including prolonged exposure, UP, and relapse prevention for substance use disorders. Participants were post-9/11 veterans and activeduty personnel. Results indicated that UP reduced PTSD and depressive symptoms following treatment. Furthermore, the retention rate (91%) was higher than retention in outpatient treatment (39%-65%), supporting the IOP model as a strategy to reduce dropout.22
Massed psychotherapy has been extended to IOP programs for PTSD treatment within the Veterans Health Administration (VHA). Yamokoski et al found that patients who completed an IOP that included massed CBT for PTSD had high retention, high satisfaction, and significant reduction in self-reported co-occurring depression symptoms. The authors also found that this model of care could be implemented and sustained within a VHA facility using minimal staffing resources.23
The UP-IOP models described by Ragsdale et al and Watkins et al included massed UP as the primary evidence-based practice (EBP) with adjunctive groups (eg, wellness and educational groups and access to complementary interventions such as mindfulness and yoga); they found that adding these groups increased retention and patient- reported satisfaction (ie, UP-IOP).20,22 The addition of wellness education alongside a primary EBP aligns with the VHA focus on whole health well-being and wellness. This includes understanding factors that motivate a patient toward health and well-being, providing health education, and offering access to complementary interventions such as mindfulness.24
Dryden et al described the whole health transformation within VHA as a proactive approach to addressing both employee and patient wellness. Their research found that the whole health model promoted wellbeing in patients and staff and these improvements were sustained during the COVID-19 pandemic. Dryden et al also noted that virtual technologies facilitated continued whole health implementation.25
The literature illustrates that (1) massed UP can be delivered with complementary education and wellness offerings that may increase retention and satisfaction by enriching treatment (eg, delivering UP-IOP); (2) whole health, including wellness education and complementary interventions (eg, mindfulness, motivational enhancement), promotes well-being in patients and clinicians; and (3) whole health education and complementary interventions can be delivered virtually.
IVET
Health Care Need
Veterans Affairs Pacific Islands Health Care System (VAPIHCS) provides medical services to veterans in the Hawaiian Islands, American Samoa, Guam, and Saipan spanning nearly 4000 miles across the Pacific Ocean. Prior to implementation of this program, veterans who received care at VAPIHCS had little to no access to UP in outpatient settings and no access to UP in residential settings. Access to UP depended on the presence of a therapist trained in UP within a given clinic and was geographically limited to the location of the UP-trained therapist. The limited outpatient access to UP was restricted to the traditional UP delivery model (eg, about 1 session/wk); thus, there was no access to accelerated UP for veterans served at VAPIHCS. In the fiscal year prior to implementation of the massed UP program, > 1000 VAPIHCS veterans had been diagnosed with obsessive-compulsive related disorder or anxiety. A massed-UP program with weekly rolling admissions would support access to UP for more veterans over 12 months, and the virtual treatment modality would reduce barriers for diverse and underserved veterans, making care more equitable and inclusive.
Successful implementation and sustainment of an EBP prompted the establishment of this UP program. In 2022, VAPIHCS launched the Intensive Virtual Evidence- Based Psychotherapy Team (iVET) for treatment of PTSD. This clinic and associated EBP project demonstrated that massed (≥3 individual sessions/wk) of prolonged exposure (PE) therapy, delivered virtually to a geographically diverse veteran population with PTSD, resulted in significant reductions in PTSD, depression, and anxiety symptoms and substance use risk factors, with very high retention rates. The iVET for PTSD program was feasible, acceptable, and effective, with veterans reporting significant improvement in quality of life and high satisfaction with their mental health services.15 Given the known benefits of transdiagnostic UP treatment (vs SDPs), the need for accelerated UP, and success with accelerated PE, our goal was to spread the EBP of massed (≥3 sessions/ wk) virtual psychotherapy to other presenting problems (eg, anxiety disorders with or without co-occurring unipolar depression) using transdiagnostic UP.
Program Description
The program implemented within outpatient mental health services at VAPIHCS was iVET for the treatment of anxiety with or without co-occurring depression. The program model consists of an accelerated course of UP and whole health education provided via VA Video Connect (VVC), the VA video telehealth platform. iVET is a 2- to 4-week program and consists of 3 parts: (1) massed individual UP for transdiagnostic treatment of anxiety and co-occurring depression, (2) group whole health and wellness classes, and (3) individual health coaching to address personal wellness goals. Programming is offered over 10-hour days to increase access across multiple time zones, especially to allow participation from Guam and Saipan.
When a patient is referred to iVET, the first contact is a video (or telephone) appointment with a registered nurse (RN) for a screening session. The screening session is designed to provide education about the program (including interventions, time commitment, and resources required for participation). Following education, the RN completes a safety screening, including screening for suicidal ideation and risk, as well as intimate partner violence risk. If urgent safety concerns are present, a licensed social worker or psychologist joins the screening to complete further risk assessment and address any safety concerns.
Following screening, patients are scheduled for a VVC telehealth intake with a licensed therapist (social worker or psychologist) to complete a diagnostic interview. Patients are sent a secure link to complete a measurement-based care (MBC) battery of self-report measures, including assessments of demographics, anxiety symptoms, depression symptoms, substance use, psychological flexibility, quality of life, and satisfaction with mental health care. The results of the diagnostic interview and self-report measures are discussed with the patient during the intake session to plan next steps and support shared decision-making. This initial VVC intake not only allows for fit assessment but also serves to troubleshoot technical difficulties with the virtual platforms.
Notably, there are minimal exclusion criteria for participation in iVET. These include active unmanaged psychosis or manic symptoms, recent suicidal crises (attempt within 8 weeks), active nonsuicidal self-injury (within 8 weeks), and moderate to severe cognitive impairment. Following intake, patients are scheduled to begin their course of care with iVET. Upon completion of intake, patients receive program materials for individual and group classes and are told they will receive email links for all VVC telehealth appointments. Patients are admitted to the iVET on a rolling basis, thereby increasing access compared with closed group and/or cohort models of care.
Patients receiving iVET attend 2 to 4 telehealth appointments daily and complete exercises between sessions. The primary iVET for Anxiety program EBP intervention is a massed or accelerated individual course of UP, which includes 8 primary components: assessment, goals and motivation, understanding emotions, mindful emotion awareness, cognitive flexibility, countering emotional behaviors, understanding and confronting physical sensations, and emotional exposures. UP is delivered in 4 to 8 individual sessions weekly (60-90 minutes each), allowing completion of the full UP protocol with fidelity in 2 to 4 weeks. In addition to primary EBP intervention, patients participate in 4 group sessions weekly (50 minutes each) of a whole health and wellness education class and have access to 1 individual health coaching session weekly (30- 60 minutes) to set wellness goals and receive coaching. During iVET, patients are invited to complete MBC batteries of self-report measures assessing anxiety symptoms, depression symptoms, substance use, psychological flexibility, quality of life, and satisfaction with mental health care at sessions 1, 5, 9, 13, and the final UP session. Following discharge from the iVET, patients are offered 1-, 3-, and 6-month individual postdischarge check-up sessions with a therapist, during which they are invited to complete MBC measures and review relapse prevention and maintenance of treatment gains. Likewise, patients are offered 1-, 3-, and 6-month individual postdischarge check-up sessions with an RN focused on maintaining wellness gains.
The iVET for Anxiety staff has 3 therapists (psychologists or social workers) and 1 RN. Additionally, the iVET for Anxiety is supported by a program manager and a program support assistant who support 2 programs total (the iVET for Anxiety plus another mental health program). The primary cost of the program is staff salary. Additional resources included computer equipment for staff and supplies (eg, printed materials for patients and office supplies). Due to the virtual environment of care, iVET staff telework and do not require physical space within VAPIHCS.
Outcomes
Veterans receiving iVET for Anxiety are invited to complete MBC multiple times, including pretreatment, during UP treatment (sessions 1, 5, 9, 13, and the final session), and posttreatment (1, 3, and 6 months). MBC measures include self-reported demographics; a 2-item measure of satisfaction with mental health services; the Acceptance and Action Questionnaire II,26 the Brief Addiction Monitor-Intensive Outpatient Program, 27 the Generalized Anxiety Disorder-7,28 the Patient Health Questionnaire (PHQ-9),29 and the Quality of Life Enjoyment and Satisfaction Questionnaire-Short Form.30
Forty-two veterans completed the iVET for Anxiety program, with a retention rate of 87.5% completing . 16 sessions of massed UP (Table 2). Veterans reported reduced anxiety (P < .001), depression (P < .001), and substance use risk (P < .001). Veterans experienced improved acceptance of emotional experiences (P < .001) and quality of life (P < .001), based on paired sample t tests comparing session 1 vs final session scores on the self-reported measures. Veterans also reported high satisfaction with mental health care in iVET for Anxiety.

Veterans experienced reduced thoughts of death or suicidal ideation (SI) based on responses to item 9 of PHQ-9. When SI was categorically analyzed (presence vs absence) using PHQ-9 item 9, there was an association observed between absence of SI and completion of a course of massed UP that does not appear to be due to chance, (X2 [1, N = 42) = 3.94; P = .047). In addition, veterans who completed the program showed a significant decrease in SI severity measured continuously (range, 0-3) on PHQ-9 item 9 (P < .05) (Table 3).

Future Directions
The aim of this program is to see sustained patient outcomes as iVET continues to serve more veterans. Another line of inquiry is longer- term follow-up, given that long-term follow- up was not addressed in this project. We also hope that the accelerated model of care can be applied to treatment of other presenting concerns (eg, relationship difficulties, insomnia). Expansion of accelerated mental health treatment into other federal and nonfederal health care settings is another area worthy of future inquiry. Exploration of staff satisfaction and burnout related to providing accelerated UP is another important future direction. Relatedly, assessment of the staff burden to learn 1 transdiagnostic EBP vs learning multiple SDPs is another future direction. Likewise, exploration of institutional benefits of investment in transdiagnostic training, supervision, and consultation for UP vs multiple SDPs may be important. These areas could also result in insightful, beneficial evidence of the effectiveness of massed UP to add to the existing literature.
Conclusions
UP for transdiagnostic treatment of anxiety, depressive, and emotional disorders has demonstrated reduced suffering and improved functioning and is supported by multiple clinical practice guidelines.1-4 Federal practitioners are positioned to improve access to this intervention, thereby reducing pain and improving lives. Indeed, it is crucial to envision a future state in which access to UP for a range of anxiety and depressive disorders is improved and broad, retention rates are dramatically improved, and clinicians providing UP do not experience the high burden and burnout associated with needing to learn and implement a variety of SDPs. Development of these programs, or similar tracks within existing programs, that provide massed or accelerated UP for transdiagnostic treatment of a range of anxiety and depressive disorders with virtual delivery options, is imperative to advance improved care for patients and clinicians.
Federal health care settings treating patients with anxiety and depression, such as those within the US Department of Defense, Indian Health Services, Bureau of Prisons, and VHA, are positioned to implement programs like iVET. Moreover, at the institutional level, investment in training and supervision in the transdiagnostic UP as opposed to multiple SDPs warrants consideration. We believe this model of care has great merit and foresee a future where all patients seeking treatment for anxiety and depression have the option to complete an accelerated or massed course of transdiagnostic care with UP if they so desire. Our experiences with iVET illustrate the feasibility, acceptability, and sustainability of such programs without requiring substantial staffing and financial resources.
- US Department of Veterans Affairs, US Department of Defense. VA/DoD clinical practice guideline for the management of major depressive disorder. Version 4. 2022. Accessed February 1, 2026. https://www.healthquality.va.gov/guidelines/MH/mdd/VADoDMDDCPGFinal508.pdf
- American Psychological Association. Clinical practice guideline for the treatment of depression across three age cohorts. February 2019. Accessed February 4, 2026. https://www.apa.org/depression-guideline/guideline.pdf
- Katzman MA, Bleau P, Blier P, et al. Canadian clinical practice guidelines for the management of anxiety, posttraumatic stress and obsessive compulsive disorders. BMC Psychiatry. 2014;14:S1. doi:10.1186/1471-244X-14-S1-S1
- Barlow DH, Farchione TJ, Bullis JR, et al. The unified protocol for transdiagnostic treatment of emotional disorders compared with diagnosis-specific protocols for anxiety disorders: a randomized clinical trial. JAMA Psychiatry. 2017;74:875-884. doi:10.1001/jamapsychiatry.2017.2164
- Calkins AW, et al. Comorbidity of anxiety and depression. In: Ressler KJ, Pine DS, Rothbaum BO, eds. Anxiety Disorders, Primer On. Oxford Academic; 2015. https://doi.org/10.1093/med/9780199395125.003.0021
- Manber R, Edinger JD, Gress JL, et al. Cognitive behavioral therapy for insomnia enhances depression outcome in patients with comorbid major depressive disorder and insomnia. Sleep. 2008;31:489-495. doi:10.1093/sleep/31.4.489
- Steele SJ, Farchione TJ, Cassiello-Robbins C, et al. Efficacy of the Unified Protocol for transdiagnostic treatment of comorbid psychopathology accompanying emotional disorders compared to treatments targeting single disorders. J Psychiatr Res. 2018;104:211-216. doi:10.1016/j.jpsychires.2018.08.005
- Sakiris N, Berle D. A systematic review and meta-analysis of the Unified Protocol as a transdiagnostic emotion regulation based intervention. Clin Psychol Rev. 2019;72:101751. doi:10.1016/j.cpr.2019.101751
- Cassiello-Robbins C, Rosenthal MZ, Ammirati RJ. Delivering transdiagnostic treatment over telehealth during the COVID-19 pandemic: application of the unified protocol. Cogn Behav Pract. 2021;28:555-572. doi:10.1016/j.cbpra.2021.04.007
- Meyer EC, Coe E, Pennington ML, et al. The unified protocol for transdiagnostic treatment of emotional disorders delivered to firefighters via videoconferencing: pilot outcomes highlighting improvements in alcohol use disorder and posttraumatic stress disorder symptoms. Cogn Behav Pract. 2024;31:215-229. doi:10.1016/j.cbpra.2022.08.004
- Varkovitzky RL, Sherrill AM, Reger GM. Effectiveness of the unified protocol for transdiagnostic treatment of emotional disorders among veterans with posttraumatic stress disorder: a pilot study. Behav Modif. 2018;42:210-230. doi:10.1177/0145445517724539
- Oliveira JT, Sousa I, Ribeiro AP, et al. Premature termination of the unified protocol for the transdiagnostic treatment of emotional disorders: The role of ambivalence towards change. Clin Psychol Psychother. 2022;29:1089-1100. doi:10.1002/cpp.2694
- Schaeuffele C, Homeyer S, Perea L, et al. The unified protocol as an internet-based intervention for emotional disorders: Randomized controlled trial. PLoS One. 2022;17:e0270178. doi:10.1371/journal.pone.0270178
- Bentley KH, Cohen ZD, Kim T, et al. The nature, timing, and symptom trajectories of dropout from transdiagnostic and single-diagnosis cognitive-behavioral therapy for anxiety disorders. Behav Ther. 2021;52:1364-1376. doi:10.1016/j.beth.2021.03.007
- Aosved AC, Brown TB, Bell JC, et al. Accelerated prolonged exposure therapy for posttraumatic stress disorder in a Veterans Health Administration system. Fed Pract. 2025;42:S6-S11. doi:10.12788/fp.0568
- Nepon J, Belik SL, Bolton J, et al. The relationship between anxiety disorders and suicide attempts: findings from the National Epidemiologic Survey on Alcohol and Related Conditions. Depress Anxiety. 2010;27:791-798. doi:10.1002/da.20674
- Shiner B, D’Avolio LW, Nguyen TM, et al. Measuring use of evidence based psychotherapy for posttraumatic stress disorder. Adm Policy Ment Health. 2013;40:311-318. doi:10.1007/s10488-012-0421-0
- Nichter B, Stein MB, Monteith LL, et al. Risk factors for suicide attempts among U.S. military veterans: A 7-year population-based, longitudinal cohort study. Suicide Life Threat Behav. 2022;52:303-316. doi:10.1111/sltb.12822
- McHugh RK, Barlow DH. The dissemination and implementation of evidence-based psychological treatments. A review of current efforts. Am Psychol. 2010;65:73-84. doi:10.1037/a0018121
- Ragsdale KA, Nichols AA, Mehta M, et al. Comorbid treatment of traumatic brain injury and mental health disorders. NeuroRehabilitation. 2024;55:375-384. doi:10.3233/NRE-230235
- Thompson-Brenner H, Brooks GE, Boswell JF, et al. Evidence-based implementation practices applied to the intensive treatment of eating disorders: summary of research and illustration of principles using a case example. Clin Psychol Sci Pract. 2018;25:e12221. doi:10.1111/cpsp.12221
- Watkins LE, Patton SC, Drexler K, et al. Clinical effectiveness of an intensive outpatient program for integrated treatment of comorbid substance abuse and mental health disorders. Cog Behav Pract. 2023;30:354-366.
- Yamokoski C, Flores H, Facemire V, et al. Feasibility of an intensive outpatient treatment program for post-traumatic stress disorder within the veterans health care administration. Psychol Serv. 2023;20:506-515. doi:10.1037/ser0000628
- Gaudet T, Kligler B. Whole health in the whole system of the Veterans Administration: how will we know we have reached this future state?. J Altern Complement Med. 2019;25:S7-S11. doi:10.1089/acm.2018.29061.gau
- Dryden EM, Bolton RE, Bokhour BG, et al. Leaning into whole health: sustaining system transformation while supporting patients and employees during COVID-19. Glob Adv Health Med. 2021;10:21649561211021047. doi:10.1177/21649561211021047
- Bond FW, Hayes SC, Baer RA, et al. Preliminary psychometric properties of the Acceptance and Action Questionnaire-II: a revised measure of psychological inflexibility and experiential avoidance. Behav Ther. 2011;42:676-688. doi:10.1016/j.beth.2011.03.007
- Cacciola JS, Alterman AI, Dephilippis D, et al. Development and initial evaluation of the Brief Addiction Monitor (BAM). J Subst Abuse Treat. 2013;44:256-263. doi:10.1016/j.jsat.2012.07.013
- Spitzer RL, Kroenke K, Williams JB, et al. A brief measure for assessing generalized anxiety disorder: the GAD-7. Arch Intern Med. 2006;166:1092-1097. doi:10.1001/archinte.166.10.1092
- Kroenke K, Spitzer RL, Williams JB. The PHQ-9: validity of a brief depression severity measure. J Gen Intern Med. 2001;16:606-613. doi:10.1046/j.1525-1497.2001.016009606.x
- Stevanovic D. Quality of life enjoyment and satisfaction questionnaire-short form for quality of life assessments in clinical practice: a psychometric study. J Psychiatr Ment Health Nurs. 2011;18:744-750. doi:10.1111/j.1365-2850.2011.01735.x
- US Department of Veterans Affairs, US Department of Defense. VA/DoD clinical practice guideline for the management of major depressive disorder. Version 4. 2022. Accessed February 1, 2026. https://www.healthquality.va.gov/guidelines/MH/mdd/VADoDMDDCPGFinal508.pdf
- American Psychological Association. Clinical practice guideline for the treatment of depression across three age cohorts. February 2019. Accessed February 4, 2026. https://www.apa.org/depression-guideline/guideline.pdf
- Katzman MA, Bleau P, Blier P, et al. Canadian clinical practice guidelines for the management of anxiety, posttraumatic stress and obsessive compulsive disorders. BMC Psychiatry. 2014;14:S1. doi:10.1186/1471-244X-14-S1-S1
- Barlow DH, Farchione TJ, Bullis JR, et al. The unified protocol for transdiagnostic treatment of emotional disorders compared with diagnosis-specific protocols for anxiety disorders: a randomized clinical trial. JAMA Psychiatry. 2017;74:875-884. doi:10.1001/jamapsychiatry.2017.2164
- Calkins AW, et al. Comorbidity of anxiety and depression. In: Ressler KJ, Pine DS, Rothbaum BO, eds. Anxiety Disorders, Primer On. Oxford Academic; 2015. https://doi.org/10.1093/med/9780199395125.003.0021
- Manber R, Edinger JD, Gress JL, et al. Cognitive behavioral therapy for insomnia enhances depression outcome in patients with comorbid major depressive disorder and insomnia. Sleep. 2008;31:489-495. doi:10.1093/sleep/31.4.489
- Steele SJ, Farchione TJ, Cassiello-Robbins C, et al. Efficacy of the Unified Protocol for transdiagnostic treatment of comorbid psychopathology accompanying emotional disorders compared to treatments targeting single disorders. J Psychiatr Res. 2018;104:211-216. doi:10.1016/j.jpsychires.2018.08.005
- Sakiris N, Berle D. A systematic review and meta-analysis of the Unified Protocol as a transdiagnostic emotion regulation based intervention. Clin Psychol Rev. 2019;72:101751. doi:10.1016/j.cpr.2019.101751
- Cassiello-Robbins C, Rosenthal MZ, Ammirati RJ. Delivering transdiagnostic treatment over telehealth during the COVID-19 pandemic: application of the unified protocol. Cogn Behav Pract. 2021;28:555-572. doi:10.1016/j.cbpra.2021.04.007
- Meyer EC, Coe E, Pennington ML, et al. The unified protocol for transdiagnostic treatment of emotional disorders delivered to firefighters via videoconferencing: pilot outcomes highlighting improvements in alcohol use disorder and posttraumatic stress disorder symptoms. Cogn Behav Pract. 2024;31:215-229. doi:10.1016/j.cbpra.2022.08.004
- Varkovitzky RL, Sherrill AM, Reger GM. Effectiveness of the unified protocol for transdiagnostic treatment of emotional disorders among veterans with posttraumatic stress disorder: a pilot study. Behav Modif. 2018;42:210-230. doi:10.1177/0145445517724539
- Oliveira JT, Sousa I, Ribeiro AP, et al. Premature termination of the unified protocol for the transdiagnostic treatment of emotional disorders: The role of ambivalence towards change. Clin Psychol Psychother. 2022;29:1089-1100. doi:10.1002/cpp.2694
- Schaeuffele C, Homeyer S, Perea L, et al. The unified protocol as an internet-based intervention for emotional disorders: Randomized controlled trial. PLoS One. 2022;17:e0270178. doi:10.1371/journal.pone.0270178
- Bentley KH, Cohen ZD, Kim T, et al. The nature, timing, and symptom trajectories of dropout from transdiagnostic and single-diagnosis cognitive-behavioral therapy for anxiety disorders. Behav Ther. 2021;52:1364-1376. doi:10.1016/j.beth.2021.03.007
- Aosved AC, Brown TB, Bell JC, et al. Accelerated prolonged exposure therapy for posttraumatic stress disorder in a Veterans Health Administration system. Fed Pract. 2025;42:S6-S11. doi:10.12788/fp.0568
- Nepon J, Belik SL, Bolton J, et al. The relationship between anxiety disorders and suicide attempts: findings from the National Epidemiologic Survey on Alcohol and Related Conditions. Depress Anxiety. 2010;27:791-798. doi:10.1002/da.20674
- Shiner B, D’Avolio LW, Nguyen TM, et al. Measuring use of evidence based psychotherapy for posttraumatic stress disorder. Adm Policy Ment Health. 2013;40:311-318. doi:10.1007/s10488-012-0421-0
- Nichter B, Stein MB, Monteith LL, et al. Risk factors for suicide attempts among U.S. military veterans: A 7-year population-based, longitudinal cohort study. Suicide Life Threat Behav. 2022;52:303-316. doi:10.1111/sltb.12822
- McHugh RK, Barlow DH. The dissemination and implementation of evidence-based psychological treatments. A review of current efforts. Am Psychol. 2010;65:73-84. doi:10.1037/a0018121
- Ragsdale KA, Nichols AA, Mehta M, et al. Comorbid treatment of traumatic brain injury and mental health disorders. NeuroRehabilitation. 2024;55:375-384. doi:10.3233/NRE-230235
- Thompson-Brenner H, Brooks GE, Boswell JF, et al. Evidence-based implementation practices applied to the intensive treatment of eating disorders: summary of research and illustration of principles using a case example. Clin Psychol Sci Pract. 2018;25:e12221. doi:10.1111/cpsp.12221
- Watkins LE, Patton SC, Drexler K, et al. Clinical effectiveness of an intensive outpatient program for integrated treatment of comorbid substance abuse and mental health disorders. Cog Behav Pract. 2023;30:354-366.
- Yamokoski C, Flores H, Facemire V, et al. Feasibility of an intensive outpatient treatment program for post-traumatic stress disorder within the veterans health care administration. Psychol Serv. 2023;20:506-515. doi:10.1037/ser0000628
- Gaudet T, Kligler B. Whole health in the whole system of the Veterans Administration: how will we know we have reached this future state?. J Altern Complement Med. 2019;25:S7-S11. doi:10.1089/acm.2018.29061.gau
- Dryden EM, Bolton RE, Bokhour BG, et al. Leaning into whole health: sustaining system transformation while supporting patients and employees during COVID-19. Glob Adv Health Med. 2021;10:21649561211021047. doi:10.1177/21649561211021047
- Bond FW, Hayes SC, Baer RA, et al. Preliminary psychometric properties of the Acceptance and Action Questionnaire-II: a revised measure of psychological inflexibility and experiential avoidance. Behav Ther. 2011;42:676-688. doi:10.1016/j.beth.2011.03.007
- Cacciola JS, Alterman AI, Dephilippis D, et al. Development and initial evaluation of the Brief Addiction Monitor (BAM). J Subst Abuse Treat. 2013;44:256-263. doi:10.1016/j.jsat.2012.07.013
- Spitzer RL, Kroenke K, Williams JB, et al. A brief measure for assessing generalized anxiety disorder: the GAD-7. Arch Intern Med. 2006;166:1092-1097. doi:10.1001/archinte.166.10.1092
- Kroenke K, Spitzer RL, Williams JB. The PHQ-9: validity of a brief depression severity measure. J Gen Intern Med. 2001;16:606-613. doi:10.1046/j.1525-1497.2001.016009606.x
- Stevanovic D. Quality of life enjoyment and satisfaction questionnaire-short form for quality of life assessments in clinical practice: a psychometric study. J Psychiatr Ment Health Nurs. 2011;18:744-750. doi:10.1111/j.1365-2850.2011.01735.x
Accelerated Unified Protocol for Transdiagnostic Treatment of Anxiety Disorders in a VHA System
Accelerated Unified Protocol for Transdiagnostic Treatment of Anxiety Disorders in a VHA System
Adalimumab in Lichen Planus: A Narrative Review of Treatment and Paradoxical Reactions
Adalimumab in Lichen Planus: A Narrative Review of Treatment and Paradoxical Reactions
Lichen planus (LP) is a chronic inflammatory condition affecting the skin (cutaneous LP), mucous membranes (oral, ocular, or vulvar LP), hair (lichen planopilaris [LPP]), and nails that predominantly occurs in middle-aged adults. Although the true etiology remains unknown, the pathogenesis of LP is thought to involve multiple factors. Several human leukocyte antigen (HLA) alleles have been associated with LP and its variants, including HLA-B27, HLA-B51, HLA-DR1 (cutaneous and oral LP), HLA-DRB1*11, and HLA-DQB1*03 (LPP). Additionally, HLA-Bw57 has been reported to be associated with oral LP in a cohort of British patients.1 In addition to HLA alleles, genetic polymorphisms in cytokines including IL-4, IL-6, IL-18, interferon (IFN) γ, and tumor necrosis factor (TNF) α and its receptor have been found to be associated with LP.2 Beyond genetics, chronic viral infection has been implicated in the development of LP. Systemic infection with the hepatitis C virus has been linked to the development of oral LP by promoting the recruitment of hepatitis C virus–specific CD8+ T cells from peripheral blood to the oral lesions, where they exhibit a terminally differentiated effector status.3 Another report found an association between human herpesvirus 7 (HHV-7) and cutaneous LP; in this study, HHV-7 RNA was detected in plasmacytoid dendritic cells but not T cells and diminished after treatment, providing evidence for dendritic cells being involved in the HHV-7–mediated pathogenesis of cutaneous LP.4 These findings were further corroborated by another study of oral LP patients that found enhanced infiltration of plasmacytoid and myeloid dendritic cells and upregulation in toll-like receptor and IFN-γ signaling.4
In addition to immune cell dysregulation, LP and its variants have been linked to neurogenic inflammation. In oral LP lesions, neurokinin 1 receptor and substance P were highly expressed and demonstrated a positive correlation with the expression of apoptotic marker caspase-3 and proliferation marker Ki-67.5 These results suggest that neuropeptides may be involved in cell proliferation and turnover in oral LP. Similarly, in patients with LPP, substance P was more abundant in affected areas, whereas another neuropeptide, calcitonin gene-related peptide, was more highly expressed in unaffected areas,6 further supporting the pathogenic role of neurogenic inflammation in LP.
A mucosal variant that often goes undiagnosed is vulvar LP. Although no distinct pathologic mechanism for vulvar LP has been established, prior reports found an association with autoantibodies.7,8 In patients with erosive vulvar LP, epidermal-binding basement membrane zone antibodies were detected in epidermal skin biopsies and in circulation with reactivity to bullous pemphigoid antigens 180 (9/11 [81.8%] patients) and 230 (2/11 [18.2%] patients).7 A similar study in patients with vulvar lichen sclerosus found similar proportions of circulating antibodies reactive to bullous pemphigoid antigens 180 (6/7 [85.7%] patients) and 230 (1/7 [14.3%] patients).8 Erosive vulvar LP has been shown to be associated with autoimmune disease (eg, alopecia areata, celiac disease and pernicious anemia),9 which suggests that the previously reported autoreactive antibodies7,8 are secondary to autoimmunity rather than primary drivers of vulvar LP pathogenesis.
Certain medications also have been reported to cause cutaneous lichenoid drug eruptions. Although they can clinically and histologically mimic classic LP, lichenoid drug eruptions are a distinct entity. Common inciting medications include thiazide diuretics, angiotensin-converting enzyme inhibitors, anti-inflammatory drugs, antimalarials, checkpoint inhibitors, antimicrobials, antihypertensives, antidiabetics, and psychiatric drugs. The exact pathologic mechanism of lichenoid drug eruptions currently is unclear but is thought to involve the binding of drug molecules to the cell-surface proteins of the epidermis, creating an antigenic hapten stimulus for CD8+T cells and triggering apoptosis of keratinocytes.1
The clinical severity of LP can range from mild localized disease to widespread and debilitating involvement. Multiple treatment modalities have been developed for management of LP, including topical and intralesional corticosteroids, phototherapy, Janus kinase inhibitors, phosphodiesterase-4 inhibitors, and anti–TNF-α inhibitors. Herein, we provide a narrative review and summary of the use of the TNF-α inhibitor adalimumab as a potential effective treatment for patients with LP.
Methods
We conducted a PubMed search of articles indexed for MEDLINE from 2005 to 2025 using the terms adalimumab AND lichen planus or adalimumab AND lichen. Articles that reported cases of oral LP, cutaneous LP, LPP, or lichenoid eruptions and adalimumab therapy were included in our review. Articles that used non-adalimumab TNF-α inhibitors were excluded. Using the search terms, 2 independent reviewers (M.G. and N.E.) conducted the literature review then screened the articles based on the inclusion and exclusion criteria. Our literature search yielded 40 articles, of which 20 met the criteria for inclusion in our narrative review.
Results
Our literature search yielded 11 patients with LP who were treated with adalimumab across studies (Table 1).10-16 Prior LP treatments included topical corticosteroids (11/11 [100%]), disease-modifying antirheumatic drugs (6/11 [54.5%]), retinoids (4/11 [36.4%]), and psoralen plus UVA (1/11 [36.4%]). Adalimumab was administered subcutaneously following 4 treatment regimens: (1)
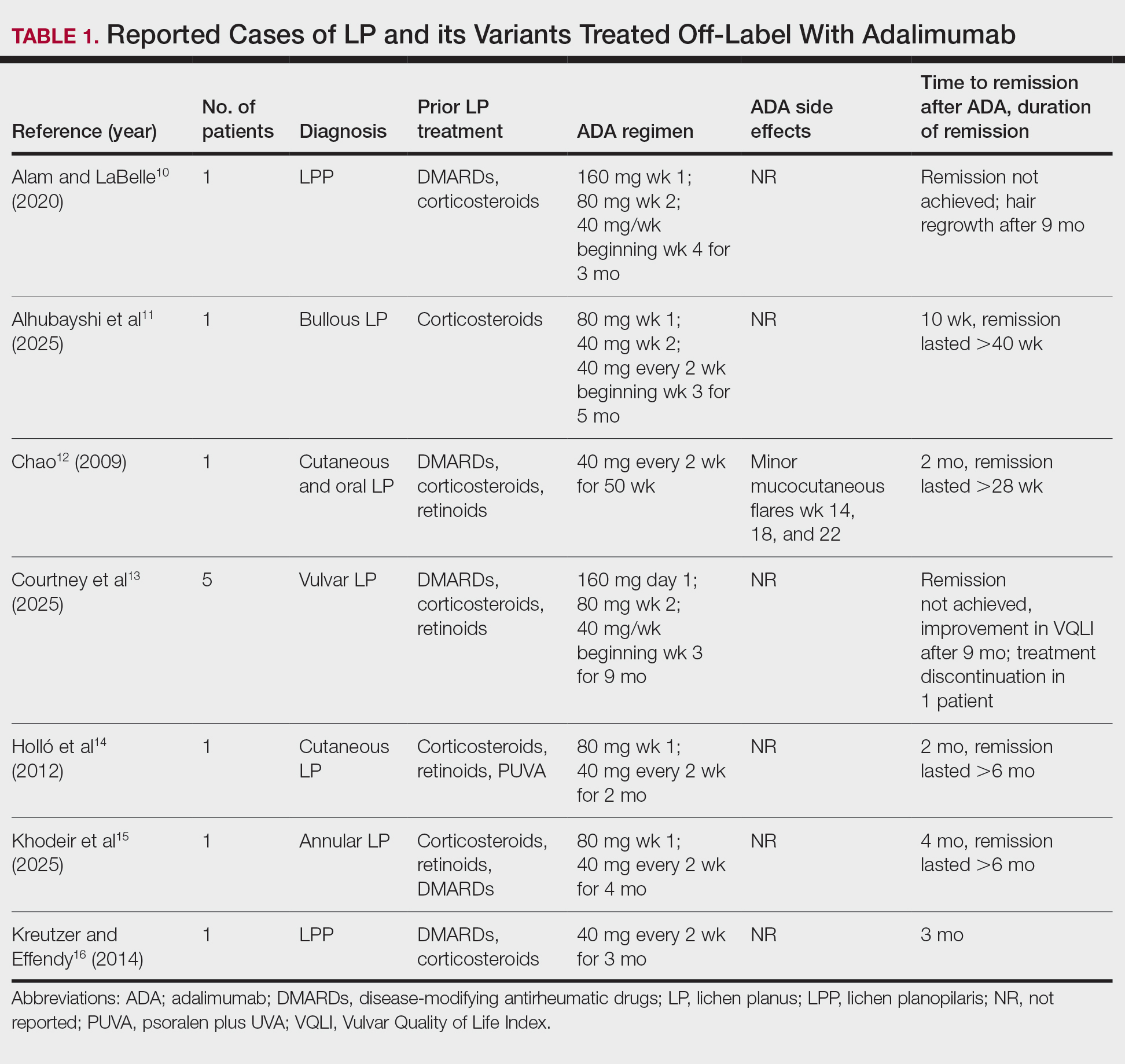
Paradoxically, our review of the literature yielded 12 patients in whom adalimumab was associated with lichenoid-type eruptions across 9 studies (Table 2).17-29 The conditions for which these patients were undergoing treatment with adalimumab included ulcerative colitis,17 psoriasis,18,19 Crohn disease,20,26 rheumatoid arthritis,21-23,26 oligoarthritis,24 and ankylosing spondylitis.25 Lichenoid drug eruptions occurred on the legs (5/12 [41.7%]), arms (3/12 [25%]), oral mucosa (2/12 [16.7%]), and forehead or scalp (2/12 [16.7%]). Onset of time to these lichenoid eruptions ranged from 2 weeks to 17 months, with a median of 4 months. Adalimumab was discontinued in 9 (75.0%) patients and was continued in 3 (25.0%). One patient who had an onset of their lichenoid eruption after 17 months of treatment with adalimumab continued to receive adalimumab therapy with the addition of topical corticosteroids, which led to resolution of their oral lesions and partial remission of their cutaneous lesions. In 1 (8.3%) patient with localized buccal lichenoid eruptions, discontinuation of adalimumab on its own was sufficient to completely clear the lesions. Seven patients (7/12 [58.3%]) received topical corticosteroids with minimal (2/12 [16.7%]) or moderate (4/12 [33.3%]) improvement, and 1 (8.3%) patient did not have reported outcomes data. Eosinophils were detected within the adalimumab-associated lichenoid eruptions in 3 (25.0%) patients.17,20,22
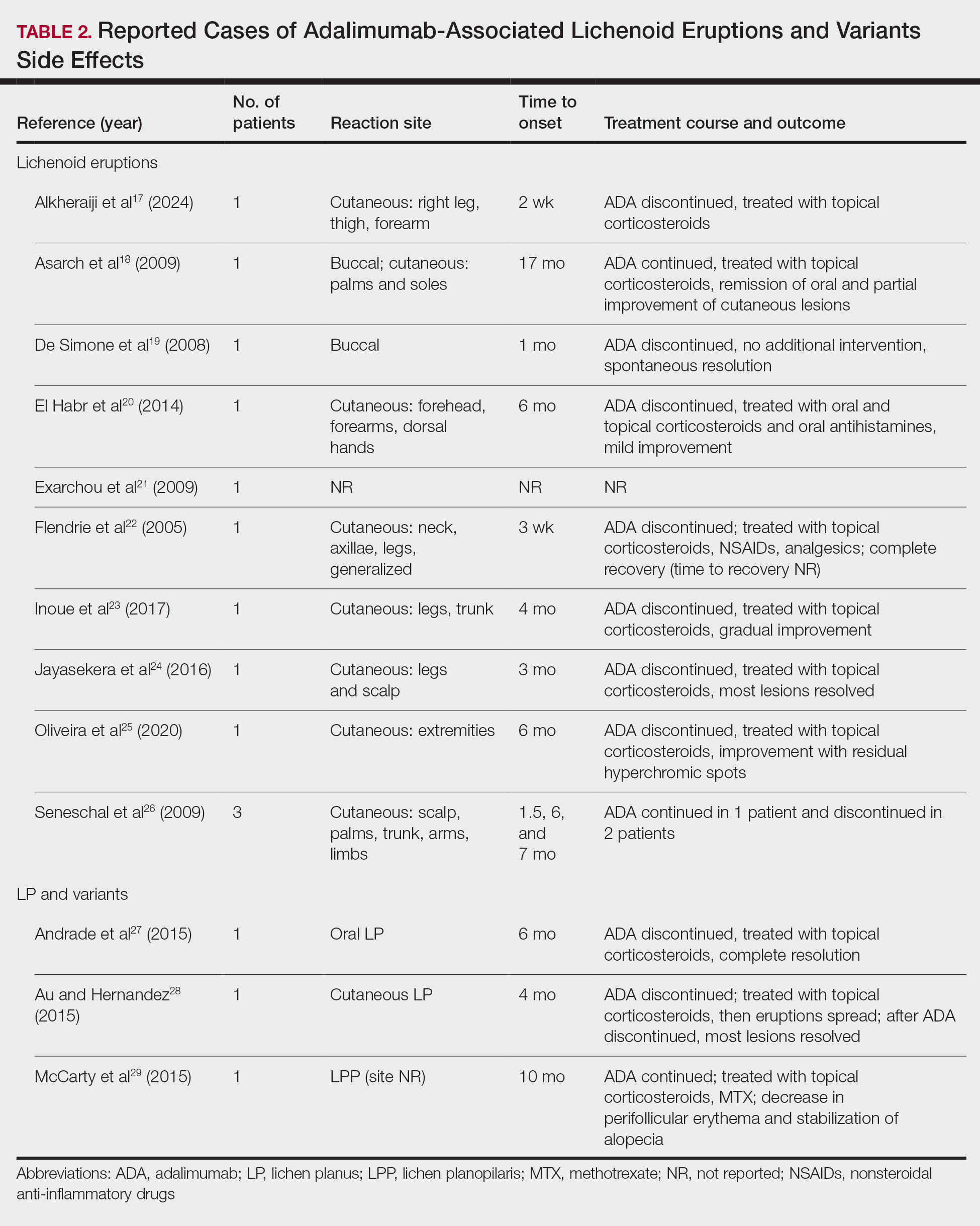
In addition to its association with lichenoid drug eruptions, adalimumab also was reported to induce LPP in a patient who was being treated for Behçet disease,29 oral LP in a patient being treated for Crohn disease,27 and cutaneous LP in a patient being treated for Crohn disease (Table 2).28 Time to onset ranged from 4 to 10 months, with a median of 6 months. Adalimumab was discontinued in 2 of 3 (66.7%) patients and was continued in the other patient (33.3%). After cessation of adalimumab therapy, administration of topical steroids led to complete resolution in the case of associated oral LP. In contrast, in adalimumab-induced cutaneous LP, initial topical corticosteroid treatment led to progression of lesions, which mostly resolved after adalimumab cessation. In 1 patient with LPP in whom adalimumab therapy could not be discontinued, topical corticosteroid and methotrexate therapy reduced the perifollicular erythema and stabilized the alopecia without full remission.
Comment
Conventional treatment modalities for LP often include topical corticosteroids as first-line therapy, with systemic corticosteroids, phototherapy, retinoids, or immunosuppressants (eg, cyclosporine or methotrexate) reserved for more severe or widespread disease. Historically, these approaches primarily have aimed to control symptoms rather than achieve long-term resolution; however, novel therapies including biologics and targeted immunomodulators show potential to induce sustained remission and improve quality of life for patients with refractory or mucosal LP.
In all reports where adalimumab was used to treat LP, patients initially received topical corticosteroids. While corticosteroids and other immunosuppressive agents are standard therapies, they often provide only temporary relief and may have an unfavorable side effect profile. Our review highlights the emerging role of adalimumab, a TNF-α inhibitor, in off-label management of LP subtypes, including cutaneous, mucosal, and vulvar LP and LPP. In several small case series and reports, patients treated with adalimumab experienced clinical improvement, including symptom resolution and quality-of-life enhancement, as well as complete remission, indicating a durable response.
The potential benefit of adalimumab in treating LP must be balanced with its paradoxical risk for inducing lichenoid eruptions as well as LP and its variants, as identified in our narrative review that included reports of patients receiving this biologic for other indications.17-29 Since adalimumab is a fully humanized antibody, the development of neutralizing antibodies may not account for drug-induced LP and lichenoid eruptions. Given that it blocks TNF-α, adalimumab may induce these lesions through a cytokine imbalance. This is supported by data demonstrating enhanced type I IFN-related proteins in plaques of patients with psoriasiform lesions treated with TNF-α inhibitors.26 These drug-induced eruptions often resolved or improved with topical corticosteroids after discontinuation, but their occurrence underscores the complexity of therapeutically targeting TNF-α in the management of LP. Our literature review suggests that adalimumab may offer therapeutic benefit in select cases of LP refractory to conventional therapy, especially when systemic control is required. Nonetheless, the risk for LP and lichenoid reactions necessitates cautious use and further investigation.
Conclusion
While the current evidence is limited to case reports and series, adalimumab shows promise as an effective and tolerable off-label treatment for LP, particularly in patients who are unresponsive to conventional immunosuppressive therapies. Remission or clinically significant improvement was achieved in several cases; however, the potential for adalimumab to induce LP and lichenoid eruptions underscores the need for careful patient selection and monitoring. Further prospective studies and larger cohorts are warranted to better define the safety and efficacy of adalimumab in treating LP lesions.
- Boch K, Langan EA, Kridin K, et al. Lichen planus. Front Med (Lausanne). 2021;8:737813.
- Gorouhi F, Davari P, Fazel N. Cutaneous and mucosal lichen planus: a comprehensive review of clinical subtypes, risk factors, diagnosis, and prognosis. ScientificWorldJournal. 2014;2014:742826.
- Pilli M, Penna A, Zerbini A, et al. Oral lichen planus pathogenesis: a role for the HCV-specific cellular immune response. Hepatology. 2002;36:1446-1452.
- Wang Y, Shang S, Sun Q, et al. Increased infiltration of CD11 c+/CD123+ dendritic cell subsets and upregulation of TLR/IFN-α signaling participate in pathogenesis of oral lichen planus. Oral Surg Oral Med Oral Pathol Oral Radiol. 2018;125:459-467.E2.
- González Moles M, Esteban F, Ruiz-Ávila I, et al. A role for the substance P/NK-1 receptor complex in cell proliferation and apoptosis in oral lichen planus. Oral Dis. 2009;15:162-169.
- Doche I, Wilcox GL, Ericson M, et al. Evidence for neurogenic inflammation in lichen planopilaris and frontal fibrosing alopecia pathogenic mechanism. Exp Dermatol. 2020;29:282-285.
- Cooper SM, Dean D, Allen J, et al. Erosive lichen planus of the vulva: weak circulating basement membrane zone antibodies are present. Clin Exp Dermatol. 2005;30:551-556.
- Howard A, Dean D, Cooper S, et al. Circulating basement membrane zone antibodies are found in lichen sclerosus of the vulva. Australas J Dermatol. 2004;45:12-15.
- Cooper SM, Ali I, Baldo M, et al. The association of lichen sclerosus and erosive lichen planus of the vulva with autoimmune disease: a case-control study. Arch Dermatol. 2008;144:1432-1435.
- Alam MS, LaBelle B. Treatment of lichen planopilaris with adalimumab in a patient with hidradenitis suppurativa and rheumatoid arthritis. JAAD Case Rep. 2020;6:219-221.
- Alhubayshi BS, Alnoshan AA, Alhumidi AA, et al. Bullous lichen planus treated with adalimumab: a case report. Case Rep Dermatol. 2025;17:42-47.
- Chao TJ. Adalimumab in the management of cutaneous and oral lichen planus. Cutis. 2009;84:325-328.
- Courtney A, Adamson SR, Veysey E. Adalimumab use in severe recalcitrant vulval lichen sclerosus and vulval lichen planus. J Low Genit Tract Dis. 2025;29:190-194.
- Holló P, Szakonyi J, Kiss D, et al. Successful treatment of lichen planus with adalimumab. Acta Derm Venereol. 2012;92:385-386.
- Khodeir J, Ohanian P, Ohanian M. Successful treatment of annular atrophic lichen planus with adalimumab. Clin Case Rep. 2025;13:E70036.
- Kreutzer K, Effendy I. Therapy-resistant folliculitis decalvans and lichen planopilaris successfully treated with adalimumab. J Dtsch Dermatol Ges. 2014;12:74-76.
- Alkheraiji A, Alotaibi H, Irfan Thalib H. Lichenoid drug eruption secondary to adalimumab: a case report. Cureus. 2024;16:E64013.
- Asarch A, Gottlieb AB, Lee J, et al. Lichen planus-like eruptions: an emerging side effect of tumor necrosis factor-alpha antagonists. J Am Acad Dermatol. 2009;61:104-111.
- De Simone C, Caldarola G, D’Agostino M, et al. Lichenoid reaction induced by adalimumab. J Eur Acad Dermatol Venereol. 2008;22:626-627.
- El Habr C, Meguerian Z, Sammour R. Adalimumab-induced lichenoid drug eruption. J Med Liban. 2014;62:238-240.
- Exarchou SA, Voulgari PV, Markatseli TE, et al. Immune-mediated skin lesions in patients treated with anti-tumour necrosis factor alpha inhibitors. Scand J Rheumatol. 2009;38:328-331.
- Flendrie M, Vissers WH, Creemers MC, et al. Dermatological conditions during TNF-α-blocking therapy in patients with rheumatoid arthritis: a prospective study. Arthritis Res Ther. 2005;7:R666-R676.
- Inoue A, Sawada Y, Yamaguchi T, et al. Lichenoid drug eruption caused by adalimumab: a case report and literature review. Eur J Dermatol. 2017;27:69-70.
- Jayasekera PSA, Walsh ML, Hurrell D, et al. Case report of lichen planopilaris occurring in a pediatric patient receiving a tumor necrosis factor α inhibitor and a review of the literature. Pediatr Dermatol. 2016;33:E143-E146.
- Oliveira SCD, Vasconcelos AHC, Magalhães EPB, et al. Clinical, histopathological and outcome analysis of five patients with lichenoid eruption following anti-tumor necrosis factor-alpha therapy for ankylosing spondylitis: report of one case and review of the literature. Cureus. 2020;12:E10598.
- Seneschal J, Milpied B, Vergier B, et al. Cytokine imbalance with increased production of interferon-alpha in psoriasiform eruptions associated with antitumour necrosis factor-alpha treatments. Br J Dermatol. 2009;161:1081-1088.
- Andrade P, Lopes S, Albuquerque A, et al. Oral lichen planus in IBD patients: a paradoxical adverse effect of anti-TNF-α therapy. Dig Dis Sci. 2015;60:2746-2749.
- Au S, Hernandez C. Paradoxical induction of psoriasis and lichen planus by tumor necrosis factor-α inhibitors. Skinmed. 2015;13:403-405.
- McCarty M, Basile A, Bair B, et al. Lichenoid reactions in association with tumor necrosis factor alpha inhibitors. J Clin Aesthet Dermatol. 2015;8:45-49.
Lichen planus (LP) is a chronic inflammatory condition affecting the skin (cutaneous LP), mucous membranes (oral, ocular, or vulvar LP), hair (lichen planopilaris [LPP]), and nails that predominantly occurs in middle-aged adults. Although the true etiology remains unknown, the pathogenesis of LP is thought to involve multiple factors. Several human leukocyte antigen (HLA) alleles have been associated with LP and its variants, including HLA-B27, HLA-B51, HLA-DR1 (cutaneous and oral LP), HLA-DRB1*11, and HLA-DQB1*03 (LPP). Additionally, HLA-Bw57 has been reported to be associated with oral LP in a cohort of British patients.1 In addition to HLA alleles, genetic polymorphisms in cytokines including IL-4, IL-6, IL-18, interferon (IFN) γ, and tumor necrosis factor (TNF) α and its receptor have been found to be associated with LP.2 Beyond genetics, chronic viral infection has been implicated in the development of LP. Systemic infection with the hepatitis C virus has been linked to the development of oral LP by promoting the recruitment of hepatitis C virus–specific CD8+ T cells from peripheral blood to the oral lesions, where they exhibit a terminally differentiated effector status.3 Another report found an association between human herpesvirus 7 (HHV-7) and cutaneous LP; in this study, HHV-7 RNA was detected in plasmacytoid dendritic cells but not T cells and diminished after treatment, providing evidence for dendritic cells being involved in the HHV-7–mediated pathogenesis of cutaneous LP.4 These findings were further corroborated by another study of oral LP patients that found enhanced infiltration of plasmacytoid and myeloid dendritic cells and upregulation in toll-like receptor and IFN-γ signaling.4
In addition to immune cell dysregulation, LP and its variants have been linked to neurogenic inflammation. In oral LP lesions, neurokinin 1 receptor and substance P were highly expressed and demonstrated a positive correlation with the expression of apoptotic marker caspase-3 and proliferation marker Ki-67.5 These results suggest that neuropeptides may be involved in cell proliferation and turnover in oral LP. Similarly, in patients with LPP, substance P was more abundant in affected areas, whereas another neuropeptide, calcitonin gene-related peptide, was more highly expressed in unaffected areas,6 further supporting the pathogenic role of neurogenic inflammation in LP.
A mucosal variant that often goes undiagnosed is vulvar LP. Although no distinct pathologic mechanism for vulvar LP has been established, prior reports found an association with autoantibodies.7,8 In patients with erosive vulvar LP, epidermal-binding basement membrane zone antibodies were detected in epidermal skin biopsies and in circulation with reactivity to bullous pemphigoid antigens 180 (9/11 [81.8%] patients) and 230 (2/11 [18.2%] patients).7 A similar study in patients with vulvar lichen sclerosus found similar proportions of circulating antibodies reactive to bullous pemphigoid antigens 180 (6/7 [85.7%] patients) and 230 (1/7 [14.3%] patients).8 Erosive vulvar LP has been shown to be associated with autoimmune disease (eg, alopecia areata, celiac disease and pernicious anemia),9 which suggests that the previously reported autoreactive antibodies7,8 are secondary to autoimmunity rather than primary drivers of vulvar LP pathogenesis.
Certain medications also have been reported to cause cutaneous lichenoid drug eruptions. Although they can clinically and histologically mimic classic LP, lichenoid drug eruptions are a distinct entity. Common inciting medications include thiazide diuretics, angiotensin-converting enzyme inhibitors, anti-inflammatory drugs, antimalarials, checkpoint inhibitors, antimicrobials, antihypertensives, antidiabetics, and psychiatric drugs. The exact pathologic mechanism of lichenoid drug eruptions currently is unclear but is thought to involve the binding of drug molecules to the cell-surface proteins of the epidermis, creating an antigenic hapten stimulus for CD8+T cells and triggering apoptosis of keratinocytes.1
The clinical severity of LP can range from mild localized disease to widespread and debilitating involvement. Multiple treatment modalities have been developed for management of LP, including topical and intralesional corticosteroids, phototherapy, Janus kinase inhibitors, phosphodiesterase-4 inhibitors, and anti–TNF-α inhibitors. Herein, we provide a narrative review and summary of the use of the TNF-α inhibitor adalimumab as a potential effective treatment for patients with LP.
Methods
We conducted a PubMed search of articles indexed for MEDLINE from 2005 to 2025 using the terms adalimumab AND lichen planus or adalimumab AND lichen. Articles that reported cases of oral LP, cutaneous LP, LPP, or lichenoid eruptions and adalimumab therapy were included in our review. Articles that used non-adalimumab TNF-α inhibitors were excluded. Using the search terms, 2 independent reviewers (M.G. and N.E.) conducted the literature review then screened the articles based on the inclusion and exclusion criteria. Our literature search yielded 40 articles, of which 20 met the criteria for inclusion in our narrative review.
Results
Our literature search yielded 11 patients with LP who were treated with adalimumab across studies (Table 1).10-16 Prior LP treatments included topical corticosteroids (11/11 [100%]), disease-modifying antirheumatic drugs (6/11 [54.5%]), retinoids (4/11 [36.4%]), and psoralen plus UVA (1/11 [36.4%]). Adalimumab was administered subcutaneously following 4 treatment regimens: (1)

Paradoxically, our review of the literature yielded 12 patients in whom adalimumab was associated with lichenoid-type eruptions across 9 studies (Table 2).17-29 The conditions for which these patients were undergoing treatment with adalimumab included ulcerative colitis,17 psoriasis,18,19 Crohn disease,20,26 rheumatoid arthritis,21-23,26 oligoarthritis,24 and ankylosing spondylitis.25 Lichenoid drug eruptions occurred on the legs (5/12 [41.7%]), arms (3/12 [25%]), oral mucosa (2/12 [16.7%]), and forehead or scalp (2/12 [16.7%]). Onset of time to these lichenoid eruptions ranged from 2 weeks to 17 months, with a median of 4 months. Adalimumab was discontinued in 9 (75.0%) patients and was continued in 3 (25.0%). One patient who had an onset of their lichenoid eruption after 17 months of treatment with adalimumab continued to receive adalimumab therapy with the addition of topical corticosteroids, which led to resolution of their oral lesions and partial remission of their cutaneous lesions. In 1 (8.3%) patient with localized buccal lichenoid eruptions, discontinuation of adalimumab on its own was sufficient to completely clear the lesions. Seven patients (7/12 [58.3%]) received topical corticosteroids with minimal (2/12 [16.7%]) or moderate (4/12 [33.3%]) improvement, and 1 (8.3%) patient did not have reported outcomes data. Eosinophils were detected within the adalimumab-associated lichenoid eruptions in 3 (25.0%) patients.17,20,22

In addition to its association with lichenoid drug eruptions, adalimumab also was reported to induce LPP in a patient who was being treated for Behçet disease,29 oral LP in a patient being treated for Crohn disease,27 and cutaneous LP in a patient being treated for Crohn disease (Table 2).28 Time to onset ranged from 4 to 10 months, with a median of 6 months. Adalimumab was discontinued in 2 of 3 (66.7%) patients and was continued in the other patient (33.3%). After cessation of adalimumab therapy, administration of topical steroids led to complete resolution in the case of associated oral LP. In contrast, in adalimumab-induced cutaneous LP, initial topical corticosteroid treatment led to progression of lesions, which mostly resolved after adalimumab cessation. In 1 patient with LPP in whom adalimumab therapy could not be discontinued, topical corticosteroid and methotrexate therapy reduced the perifollicular erythema and stabilized the alopecia without full remission.
Comment
Conventional treatment modalities for LP often include topical corticosteroids as first-line therapy, with systemic corticosteroids, phototherapy, retinoids, or immunosuppressants (eg, cyclosporine or methotrexate) reserved for more severe or widespread disease. Historically, these approaches primarily have aimed to control symptoms rather than achieve long-term resolution; however, novel therapies including biologics and targeted immunomodulators show potential to induce sustained remission and improve quality of life for patients with refractory or mucosal LP.
In all reports where adalimumab was used to treat LP, patients initially received topical corticosteroids. While corticosteroids and other immunosuppressive agents are standard therapies, they often provide only temporary relief and may have an unfavorable side effect profile. Our review highlights the emerging role of adalimumab, a TNF-α inhibitor, in off-label management of LP subtypes, including cutaneous, mucosal, and vulvar LP and LPP. In several small case series and reports, patients treated with adalimumab experienced clinical improvement, including symptom resolution and quality-of-life enhancement, as well as complete remission, indicating a durable response.
The potential benefit of adalimumab in treating LP must be balanced with its paradoxical risk for inducing lichenoid eruptions as well as LP and its variants, as identified in our narrative review that included reports of patients receiving this biologic for other indications.17-29 Since adalimumab is a fully humanized antibody, the development of neutralizing antibodies may not account for drug-induced LP and lichenoid eruptions. Given that it blocks TNF-α, adalimumab may induce these lesions through a cytokine imbalance. This is supported by data demonstrating enhanced type I IFN-related proteins in plaques of patients with psoriasiform lesions treated with TNF-α inhibitors.26 These drug-induced eruptions often resolved or improved with topical corticosteroids after discontinuation, but their occurrence underscores the complexity of therapeutically targeting TNF-α in the management of LP. Our literature review suggests that adalimumab may offer therapeutic benefit in select cases of LP refractory to conventional therapy, especially when systemic control is required. Nonetheless, the risk for LP and lichenoid reactions necessitates cautious use and further investigation.
Conclusion
While the current evidence is limited to case reports and series, adalimumab shows promise as an effective and tolerable off-label treatment for LP, particularly in patients who are unresponsive to conventional immunosuppressive therapies. Remission or clinically significant improvement was achieved in several cases; however, the potential for adalimumab to induce LP and lichenoid eruptions underscores the need for careful patient selection and monitoring. Further prospective studies and larger cohorts are warranted to better define the safety and efficacy of adalimumab in treating LP lesions.
Lichen planus (LP) is a chronic inflammatory condition affecting the skin (cutaneous LP), mucous membranes (oral, ocular, or vulvar LP), hair (lichen planopilaris [LPP]), and nails that predominantly occurs in middle-aged adults. Although the true etiology remains unknown, the pathogenesis of LP is thought to involve multiple factors. Several human leukocyte antigen (HLA) alleles have been associated with LP and its variants, including HLA-B27, HLA-B51, HLA-DR1 (cutaneous and oral LP), HLA-DRB1*11, and HLA-DQB1*03 (LPP). Additionally, HLA-Bw57 has been reported to be associated with oral LP in a cohort of British patients.1 In addition to HLA alleles, genetic polymorphisms in cytokines including IL-4, IL-6, IL-18, interferon (IFN) γ, and tumor necrosis factor (TNF) α and its receptor have been found to be associated with LP.2 Beyond genetics, chronic viral infection has been implicated in the development of LP. Systemic infection with the hepatitis C virus has been linked to the development of oral LP by promoting the recruitment of hepatitis C virus–specific CD8+ T cells from peripheral blood to the oral lesions, where they exhibit a terminally differentiated effector status.3 Another report found an association between human herpesvirus 7 (HHV-7) and cutaneous LP; in this study, HHV-7 RNA was detected in plasmacytoid dendritic cells but not T cells and diminished after treatment, providing evidence for dendritic cells being involved in the HHV-7–mediated pathogenesis of cutaneous LP.4 These findings were further corroborated by another study of oral LP patients that found enhanced infiltration of plasmacytoid and myeloid dendritic cells and upregulation in toll-like receptor and IFN-γ signaling.4
In addition to immune cell dysregulation, LP and its variants have been linked to neurogenic inflammation. In oral LP lesions, neurokinin 1 receptor and substance P were highly expressed and demonstrated a positive correlation with the expression of apoptotic marker caspase-3 and proliferation marker Ki-67.5 These results suggest that neuropeptides may be involved in cell proliferation and turnover in oral LP. Similarly, in patients with LPP, substance P was more abundant in affected areas, whereas another neuropeptide, calcitonin gene-related peptide, was more highly expressed in unaffected areas,6 further supporting the pathogenic role of neurogenic inflammation in LP.
A mucosal variant that often goes undiagnosed is vulvar LP. Although no distinct pathologic mechanism for vulvar LP has been established, prior reports found an association with autoantibodies.7,8 In patients with erosive vulvar LP, epidermal-binding basement membrane zone antibodies were detected in epidermal skin biopsies and in circulation with reactivity to bullous pemphigoid antigens 180 (9/11 [81.8%] patients) and 230 (2/11 [18.2%] patients).7 A similar study in patients with vulvar lichen sclerosus found similar proportions of circulating antibodies reactive to bullous pemphigoid antigens 180 (6/7 [85.7%] patients) and 230 (1/7 [14.3%] patients).8 Erosive vulvar LP has been shown to be associated with autoimmune disease (eg, alopecia areata, celiac disease and pernicious anemia),9 which suggests that the previously reported autoreactive antibodies7,8 are secondary to autoimmunity rather than primary drivers of vulvar LP pathogenesis.
Certain medications also have been reported to cause cutaneous lichenoid drug eruptions. Although they can clinically and histologically mimic classic LP, lichenoid drug eruptions are a distinct entity. Common inciting medications include thiazide diuretics, angiotensin-converting enzyme inhibitors, anti-inflammatory drugs, antimalarials, checkpoint inhibitors, antimicrobials, antihypertensives, antidiabetics, and psychiatric drugs. The exact pathologic mechanism of lichenoid drug eruptions currently is unclear but is thought to involve the binding of drug molecules to the cell-surface proteins of the epidermis, creating an antigenic hapten stimulus for CD8+T cells and triggering apoptosis of keratinocytes.1
The clinical severity of LP can range from mild localized disease to widespread and debilitating involvement. Multiple treatment modalities have been developed for management of LP, including topical and intralesional corticosteroids, phototherapy, Janus kinase inhibitors, phosphodiesterase-4 inhibitors, and anti–TNF-α inhibitors. Herein, we provide a narrative review and summary of the use of the TNF-α inhibitor adalimumab as a potential effective treatment for patients with LP.
Methods
We conducted a PubMed search of articles indexed for MEDLINE from 2005 to 2025 using the terms adalimumab AND lichen planus or adalimumab AND lichen. Articles that reported cases of oral LP, cutaneous LP, LPP, or lichenoid eruptions and adalimumab therapy were included in our review. Articles that used non-adalimumab TNF-α inhibitors were excluded. Using the search terms, 2 independent reviewers (M.G. and N.E.) conducted the literature review then screened the articles based on the inclusion and exclusion criteria. Our literature search yielded 40 articles, of which 20 met the criteria for inclusion in our narrative review.
Results
Our literature search yielded 11 patients with LP who were treated with adalimumab across studies (Table 1).10-16 Prior LP treatments included topical corticosteroids (11/11 [100%]), disease-modifying antirheumatic drugs (6/11 [54.5%]), retinoids (4/11 [36.4%]), and psoralen plus UVA (1/11 [36.4%]). Adalimumab was administered subcutaneously following 4 treatment regimens: (1)

Paradoxically, our review of the literature yielded 12 patients in whom adalimumab was associated with lichenoid-type eruptions across 9 studies (Table 2).17-29 The conditions for which these patients were undergoing treatment with adalimumab included ulcerative colitis,17 psoriasis,18,19 Crohn disease,20,26 rheumatoid arthritis,21-23,26 oligoarthritis,24 and ankylosing spondylitis.25 Lichenoid drug eruptions occurred on the legs (5/12 [41.7%]), arms (3/12 [25%]), oral mucosa (2/12 [16.7%]), and forehead or scalp (2/12 [16.7%]). Onset of time to these lichenoid eruptions ranged from 2 weeks to 17 months, with a median of 4 months. Adalimumab was discontinued in 9 (75.0%) patients and was continued in 3 (25.0%). One patient who had an onset of their lichenoid eruption after 17 months of treatment with adalimumab continued to receive adalimumab therapy with the addition of topical corticosteroids, which led to resolution of their oral lesions and partial remission of their cutaneous lesions. In 1 (8.3%) patient with localized buccal lichenoid eruptions, discontinuation of adalimumab on its own was sufficient to completely clear the lesions. Seven patients (7/12 [58.3%]) received topical corticosteroids with minimal (2/12 [16.7%]) or moderate (4/12 [33.3%]) improvement, and 1 (8.3%) patient did not have reported outcomes data. Eosinophils were detected within the adalimumab-associated lichenoid eruptions in 3 (25.0%) patients.17,20,22

In addition to its association with lichenoid drug eruptions, adalimumab also was reported to induce LPP in a patient who was being treated for Behçet disease,29 oral LP in a patient being treated for Crohn disease,27 and cutaneous LP in a patient being treated for Crohn disease (Table 2).28 Time to onset ranged from 4 to 10 months, with a median of 6 months. Adalimumab was discontinued in 2 of 3 (66.7%) patients and was continued in the other patient (33.3%). After cessation of adalimumab therapy, administration of topical steroids led to complete resolution in the case of associated oral LP. In contrast, in adalimumab-induced cutaneous LP, initial topical corticosteroid treatment led to progression of lesions, which mostly resolved after adalimumab cessation. In 1 patient with LPP in whom adalimumab therapy could not be discontinued, topical corticosteroid and methotrexate therapy reduced the perifollicular erythema and stabilized the alopecia without full remission.
Comment
Conventional treatment modalities for LP often include topical corticosteroids as first-line therapy, with systemic corticosteroids, phototherapy, retinoids, or immunosuppressants (eg, cyclosporine or methotrexate) reserved for more severe or widespread disease. Historically, these approaches primarily have aimed to control symptoms rather than achieve long-term resolution; however, novel therapies including biologics and targeted immunomodulators show potential to induce sustained remission and improve quality of life for patients with refractory or mucosal LP.
In all reports where adalimumab was used to treat LP, patients initially received topical corticosteroids. While corticosteroids and other immunosuppressive agents are standard therapies, they often provide only temporary relief and may have an unfavorable side effect profile. Our review highlights the emerging role of adalimumab, a TNF-α inhibitor, in off-label management of LP subtypes, including cutaneous, mucosal, and vulvar LP and LPP. In several small case series and reports, patients treated with adalimumab experienced clinical improvement, including symptom resolution and quality-of-life enhancement, as well as complete remission, indicating a durable response.
The potential benefit of adalimumab in treating LP must be balanced with its paradoxical risk for inducing lichenoid eruptions as well as LP and its variants, as identified in our narrative review that included reports of patients receiving this biologic for other indications.17-29 Since adalimumab is a fully humanized antibody, the development of neutralizing antibodies may not account for drug-induced LP and lichenoid eruptions. Given that it blocks TNF-α, adalimumab may induce these lesions through a cytokine imbalance. This is supported by data demonstrating enhanced type I IFN-related proteins in plaques of patients with psoriasiform lesions treated with TNF-α inhibitors.26 These drug-induced eruptions often resolved or improved with topical corticosteroids after discontinuation, but their occurrence underscores the complexity of therapeutically targeting TNF-α in the management of LP. Our literature review suggests that adalimumab may offer therapeutic benefit in select cases of LP refractory to conventional therapy, especially when systemic control is required. Nonetheless, the risk for LP and lichenoid reactions necessitates cautious use and further investigation.
Conclusion
While the current evidence is limited to case reports and series, adalimumab shows promise as an effective and tolerable off-label treatment for LP, particularly in patients who are unresponsive to conventional immunosuppressive therapies. Remission or clinically significant improvement was achieved in several cases; however, the potential for adalimumab to induce LP and lichenoid eruptions underscores the need for careful patient selection and monitoring. Further prospective studies and larger cohorts are warranted to better define the safety and efficacy of adalimumab in treating LP lesions.
- Boch K, Langan EA, Kridin K, et al. Lichen planus. Front Med (Lausanne). 2021;8:737813.
- Gorouhi F, Davari P, Fazel N. Cutaneous and mucosal lichen planus: a comprehensive review of clinical subtypes, risk factors, diagnosis, and prognosis. ScientificWorldJournal. 2014;2014:742826.
- Pilli M, Penna A, Zerbini A, et al. Oral lichen planus pathogenesis: a role for the HCV-specific cellular immune response. Hepatology. 2002;36:1446-1452.
- Wang Y, Shang S, Sun Q, et al. Increased infiltration of CD11 c+/CD123+ dendritic cell subsets and upregulation of TLR/IFN-α signaling participate in pathogenesis of oral lichen planus. Oral Surg Oral Med Oral Pathol Oral Radiol. 2018;125:459-467.E2.
- González Moles M, Esteban F, Ruiz-Ávila I, et al. A role for the substance P/NK-1 receptor complex in cell proliferation and apoptosis in oral lichen planus. Oral Dis. 2009;15:162-169.
- Doche I, Wilcox GL, Ericson M, et al. Evidence for neurogenic inflammation in lichen planopilaris and frontal fibrosing alopecia pathogenic mechanism. Exp Dermatol. 2020;29:282-285.
- Cooper SM, Dean D, Allen J, et al. Erosive lichen planus of the vulva: weak circulating basement membrane zone antibodies are present. Clin Exp Dermatol. 2005;30:551-556.
- Howard A, Dean D, Cooper S, et al. Circulating basement membrane zone antibodies are found in lichen sclerosus of the vulva. Australas J Dermatol. 2004;45:12-15.
- Cooper SM, Ali I, Baldo M, et al. The association of lichen sclerosus and erosive lichen planus of the vulva with autoimmune disease: a case-control study. Arch Dermatol. 2008;144:1432-1435.
- Alam MS, LaBelle B. Treatment of lichen planopilaris with adalimumab in a patient with hidradenitis suppurativa and rheumatoid arthritis. JAAD Case Rep. 2020;6:219-221.
- Alhubayshi BS, Alnoshan AA, Alhumidi AA, et al. Bullous lichen planus treated with adalimumab: a case report. Case Rep Dermatol. 2025;17:42-47.
- Chao TJ. Adalimumab in the management of cutaneous and oral lichen planus. Cutis. 2009;84:325-328.
- Courtney A, Adamson SR, Veysey E. Adalimumab use in severe recalcitrant vulval lichen sclerosus and vulval lichen planus. J Low Genit Tract Dis. 2025;29:190-194.
- Holló P, Szakonyi J, Kiss D, et al. Successful treatment of lichen planus with adalimumab. Acta Derm Venereol. 2012;92:385-386.
- Khodeir J, Ohanian P, Ohanian M. Successful treatment of annular atrophic lichen planus with adalimumab. Clin Case Rep. 2025;13:E70036.
- Kreutzer K, Effendy I. Therapy-resistant folliculitis decalvans and lichen planopilaris successfully treated with adalimumab. J Dtsch Dermatol Ges. 2014;12:74-76.
- Alkheraiji A, Alotaibi H, Irfan Thalib H. Lichenoid drug eruption secondary to adalimumab: a case report. Cureus. 2024;16:E64013.
- Asarch A, Gottlieb AB, Lee J, et al. Lichen planus-like eruptions: an emerging side effect of tumor necrosis factor-alpha antagonists. J Am Acad Dermatol. 2009;61:104-111.
- De Simone C, Caldarola G, D’Agostino M, et al. Lichenoid reaction induced by adalimumab. J Eur Acad Dermatol Venereol. 2008;22:626-627.
- El Habr C, Meguerian Z, Sammour R. Adalimumab-induced lichenoid drug eruption. J Med Liban. 2014;62:238-240.
- Exarchou SA, Voulgari PV, Markatseli TE, et al. Immune-mediated skin lesions in patients treated with anti-tumour necrosis factor alpha inhibitors. Scand J Rheumatol. 2009;38:328-331.
- Flendrie M, Vissers WH, Creemers MC, et al. Dermatological conditions during TNF-α-blocking therapy in patients with rheumatoid arthritis: a prospective study. Arthritis Res Ther. 2005;7:R666-R676.
- Inoue A, Sawada Y, Yamaguchi T, et al. Lichenoid drug eruption caused by adalimumab: a case report and literature review. Eur J Dermatol. 2017;27:69-70.
- Jayasekera PSA, Walsh ML, Hurrell D, et al. Case report of lichen planopilaris occurring in a pediatric patient receiving a tumor necrosis factor α inhibitor and a review of the literature. Pediatr Dermatol. 2016;33:E143-E146.
- Oliveira SCD, Vasconcelos AHC, Magalhães EPB, et al. Clinical, histopathological and outcome analysis of five patients with lichenoid eruption following anti-tumor necrosis factor-alpha therapy for ankylosing spondylitis: report of one case and review of the literature. Cureus. 2020;12:E10598.
- Seneschal J, Milpied B, Vergier B, et al. Cytokine imbalance with increased production of interferon-alpha in psoriasiform eruptions associated with antitumour necrosis factor-alpha treatments. Br J Dermatol. 2009;161:1081-1088.
- Andrade P, Lopes S, Albuquerque A, et al. Oral lichen planus in IBD patients: a paradoxical adverse effect of anti-TNF-α therapy. Dig Dis Sci. 2015;60:2746-2749.
- Au S, Hernandez C. Paradoxical induction of psoriasis and lichen planus by tumor necrosis factor-α inhibitors. Skinmed. 2015;13:403-405.
- McCarty M, Basile A, Bair B, et al. Lichenoid reactions in association with tumor necrosis factor alpha inhibitors. J Clin Aesthet Dermatol. 2015;8:45-49.
- Boch K, Langan EA, Kridin K, et al. Lichen planus. Front Med (Lausanne). 2021;8:737813.
- Gorouhi F, Davari P, Fazel N. Cutaneous and mucosal lichen planus: a comprehensive review of clinical subtypes, risk factors, diagnosis, and prognosis. ScientificWorldJournal. 2014;2014:742826.
- Pilli M, Penna A, Zerbini A, et al. Oral lichen planus pathogenesis: a role for the HCV-specific cellular immune response. Hepatology. 2002;36:1446-1452.
- Wang Y, Shang S, Sun Q, et al. Increased infiltration of CD11 c+/CD123+ dendritic cell subsets and upregulation of TLR/IFN-α signaling participate in pathogenesis of oral lichen planus. Oral Surg Oral Med Oral Pathol Oral Radiol. 2018;125:459-467.E2.
- González Moles M, Esteban F, Ruiz-Ávila I, et al. A role for the substance P/NK-1 receptor complex in cell proliferation and apoptosis in oral lichen planus. Oral Dis. 2009;15:162-169.
- Doche I, Wilcox GL, Ericson M, et al. Evidence for neurogenic inflammation in lichen planopilaris and frontal fibrosing alopecia pathogenic mechanism. Exp Dermatol. 2020;29:282-285.
- Cooper SM, Dean D, Allen J, et al. Erosive lichen planus of the vulva: weak circulating basement membrane zone antibodies are present. Clin Exp Dermatol. 2005;30:551-556.
- Howard A, Dean D, Cooper S, et al. Circulating basement membrane zone antibodies are found in lichen sclerosus of the vulva. Australas J Dermatol. 2004;45:12-15.
- Cooper SM, Ali I, Baldo M, et al. The association of lichen sclerosus and erosive lichen planus of the vulva with autoimmune disease: a case-control study. Arch Dermatol. 2008;144:1432-1435.
- Alam MS, LaBelle B. Treatment of lichen planopilaris with adalimumab in a patient with hidradenitis suppurativa and rheumatoid arthritis. JAAD Case Rep. 2020;6:219-221.
- Alhubayshi BS, Alnoshan AA, Alhumidi AA, et al. Bullous lichen planus treated with adalimumab: a case report. Case Rep Dermatol. 2025;17:42-47.
- Chao TJ. Adalimumab in the management of cutaneous and oral lichen planus. Cutis. 2009;84:325-328.
- Courtney A, Adamson SR, Veysey E. Adalimumab use in severe recalcitrant vulval lichen sclerosus and vulval lichen planus. J Low Genit Tract Dis. 2025;29:190-194.
- Holló P, Szakonyi J, Kiss D, et al. Successful treatment of lichen planus with adalimumab. Acta Derm Venereol. 2012;92:385-386.
- Khodeir J, Ohanian P, Ohanian M. Successful treatment of annular atrophic lichen planus with adalimumab. Clin Case Rep. 2025;13:E70036.
- Kreutzer K, Effendy I. Therapy-resistant folliculitis decalvans and lichen planopilaris successfully treated with adalimumab. J Dtsch Dermatol Ges. 2014;12:74-76.
- Alkheraiji A, Alotaibi H, Irfan Thalib H. Lichenoid drug eruption secondary to adalimumab: a case report. Cureus. 2024;16:E64013.
- Asarch A, Gottlieb AB, Lee J, et al. Lichen planus-like eruptions: an emerging side effect of tumor necrosis factor-alpha antagonists. J Am Acad Dermatol. 2009;61:104-111.
- De Simone C, Caldarola G, D’Agostino M, et al. Lichenoid reaction induced by adalimumab. J Eur Acad Dermatol Venereol. 2008;22:626-627.
- El Habr C, Meguerian Z, Sammour R. Adalimumab-induced lichenoid drug eruption. J Med Liban. 2014;62:238-240.
- Exarchou SA, Voulgari PV, Markatseli TE, et al. Immune-mediated skin lesions in patients treated with anti-tumour necrosis factor alpha inhibitors. Scand J Rheumatol. 2009;38:328-331.
- Flendrie M, Vissers WH, Creemers MC, et al. Dermatological conditions during TNF-α-blocking therapy in patients with rheumatoid arthritis: a prospective study. Arthritis Res Ther. 2005;7:R666-R676.
- Inoue A, Sawada Y, Yamaguchi T, et al. Lichenoid drug eruption caused by adalimumab: a case report and literature review. Eur J Dermatol. 2017;27:69-70.
- Jayasekera PSA, Walsh ML, Hurrell D, et al. Case report of lichen planopilaris occurring in a pediatric patient receiving a tumor necrosis factor α inhibitor and a review of the literature. Pediatr Dermatol. 2016;33:E143-E146.
- Oliveira SCD, Vasconcelos AHC, Magalhães EPB, et al. Clinical, histopathological and outcome analysis of five patients with lichenoid eruption following anti-tumor necrosis factor-alpha therapy for ankylosing spondylitis: report of one case and review of the literature. Cureus. 2020;12:E10598.
- Seneschal J, Milpied B, Vergier B, et al. Cytokine imbalance with increased production of interferon-alpha in psoriasiform eruptions associated with antitumour necrosis factor-alpha treatments. Br J Dermatol. 2009;161:1081-1088.
- Andrade P, Lopes S, Albuquerque A, et al. Oral lichen planus in IBD patients: a paradoxical adverse effect of anti-TNF-α therapy. Dig Dis Sci. 2015;60:2746-2749.
- Au S, Hernandez C. Paradoxical induction of psoriasis and lichen planus by tumor necrosis factor-α inhibitors. Skinmed. 2015;13:403-405.
- McCarty M, Basile A, Bair B, et al. Lichenoid reactions in association with tumor necrosis factor alpha inhibitors. J Clin Aesthet Dermatol. 2015;8:45-49.
Adalimumab in Lichen Planus: A Narrative Review of Treatment and Paradoxical Reactions
Adalimumab in Lichen Planus: A Narrative Review of Treatment and Paradoxical Reactions
Practice Points
- Adalimumab can be beneficial when used off label for treatment of lichen planus in patients who do not respond to conventional therapies, including corticosteroids and immunosuppressants.
- Clinicians should be aware that adalimumab could potentially lead to paradoxical lichenoid eruptions and should monitor patients closely during treatment.
Metastatic Primary Extramammary Paget Disease: A Case Series
Metastatic Primary Extramammary Paget Disease: A Case Series
Extramammary Paget disease (EMPD) is a rare cutaneous malignancy typically seen in apocrine-rich areas, including the axillae and anogenital region. It presents as a slow-growing, erythematous patch or plaque that commonly is misdiagnosed as an infectious or inflammatory condition.1,2 Primary EMPD occurs as a intraepithelial neoplasm, whereas secondary EMPD occurs due to epidermotropic metastases or direct extension of an underlying adenocarcinoma into the skin.1 Most commonly, primary EMPD occurs in situ; however, when present, dermal invasion and metastases from the skin are associated with poorer outcomes.3 Given the rarity of metastatic disease, existing literature is limited to case reports and case series.
We present 2 patients with metastatic primary EMPD who had evidence of invasion on initial biopsy and died secondary to metastatic EMPD. We conducted a comprehensive review of the literature for invasive and metastatic EMPD to highlight key clinicopathologic features, treatment considerations, and the potential for rapid disease progression in cases of invasive EMPD.
Case Series
Patient 1—A 68-year-old White man with a history of breast cancer (in remission) presented to our clinic for further management of biopsy-proven scrotal EMPD. Prior to biopsy, he described a 6-month history of worsening scrotal rash treated with topical antifungals, oral antibiotics, and topical steroids due to presumed diagnosis of intertrigo, cellulitis, and dermatitis, respectively. Clinical examination showed indurated, erythematous, ulcerated plaques involving the bilateral groin, genitalia, and perineum (Figure 1). Skin biopsy confirmed a diagnosis of EMPD with both dermal and lymphovascular invasion. An immunohistochemical profile was positive for CK7 and carcinoembryonic antigen (CEA) and negative for CK20 (Figure 2).
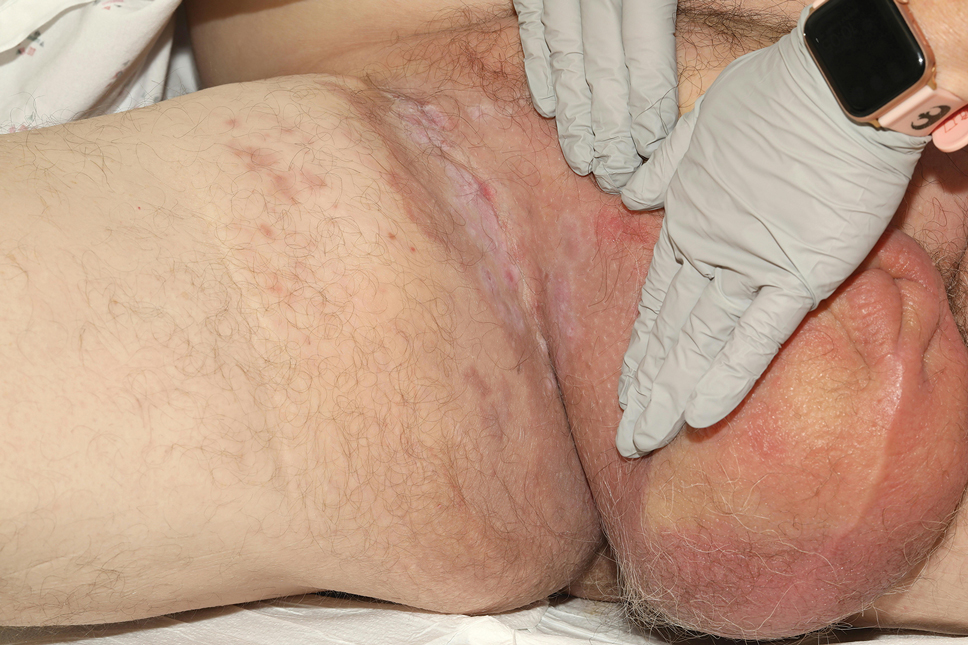
At presentation, the patient had palpable lymphadenopathy and scrotal edema concerning for inguinal and iliac lymph node metastases. Workup for an underlying adenocarcinoma included computed tomography (CT) of the chest, abdomen, and pelvis; urologic consultation with cystoscopy; and a screening colonoscopy. The CT scan revealed multiple enlarged inguinal and external iliac lymph nodes. Fine-needle aspiration revealed CK7- and CEA-positive neoplastic cells consistent with metastatic EMPD. The patient was treated with 6 cycles of carboplatin-paclitaxel, palliative radiation therapy, and pembrolizumab with minimal response to treatment and development of osteolytic vertebral lesions concerning for disease progression. He died 1 year after the initial diagnosis secondary to the disease.
Patient 2—A 79-year-old White man presented for further management of an outside diagnosis of superficially invasive primary EMPD of the bilateral inguinal folds and scrotum that had been present for 5 months prior to biopsy and diagnosis. Clinical examination at initial presentation revealed erythematous patches of the bilateral inguinal folds and scrotum, as well as an erythematous scaling plaque in the right axilla. There was no palpable clinical lymphadenopathy. Biopsy of the axilla and groin were both consistent with invasive EMPD with positive staining for CK7 and negative staining for CK20 and CDX2. Workup for underlying adenocarcinoma with whole-body positron emission tomography/CT, mammography, esophagogastroduodenoscopy, serum CEA, colonoscopy, and cystoscopy were all negative for a metastatic adenocarcinoma. There was no imaging or clinical evidence of lymphadenopathy. Complete circumferential peripheral and deep-margin assessment was performed in a staged manner on both sites, and negative margins were obtained.
Surveillance imaging 6 months after surgery revealed suspicious hepatic lesions. Fine-needle aspiration of the hepatic lesions demonstrated positive staining for CK7 and negative staining for CK20, CDX2, prostate-specific antigen, and thyroid transcription factor 1, consistent with metastatic EMPD. Oncology recommended carboplatin and docetaxel or docetaxel monotherapy chemotherapy. The patient was further managed by an outside oncologist due to ease of travel but died secondary to the disease 15 months following the initial diagnosis.
Comment
Extramammary Paget disease is an uncommon cutaneous malignancy that manifests as pruritic erythematous plaques within apocrine-rich areas such as the genitalia, axillae, or anal region. It most commonly occurs in patients older than 65 years, with White women and Asian men being affected at disproportionately higher rates.1,4 Delay in diagnosis is common, as EMPD can mimic other benign inflammatory or infectious conditions, including contact dermatitis, seborrheic dermatitis, tinea, candidiasis, and eczema.1
Metastatic and multifocal cases of primary EMPD are especially rare. According to a search of PubMed articles indexed for MEDLINE published through December 2023 using the terms extramammary Paget disease, EMPD, neoplasm metastasis, invasive extramammary, and neoplasm invasiveness, we identified 5040 cases of invasive EMPD and 477 cases of metastatic EMPD.5-37 Of the reports that disclosed patient demographic information, 3627 patients were female 1410 were male, and the mean age was 67 years. Sites of metastases included regional lymph nodes, liver, lungs, cervix, bladder, bone, brain, skin, kidney, and adrenal glands
Workup for EMPD—The initial steps for workup of EMPD include a thorough physical examination and lymph node assessment. A skin biopsy also should be performed for patients presenting with refractory, pruritic, and eczematous rashes in apocrine-rich areas to evaluate for EMPD.1 Characterization of large and complex tumors is better achieved through multiple biopsies with particular focus on nodular or thickened areas, as these may indicate invasive disease.2 Primary EMPD is characterized by pagetoid cells with abundant pale cytoplasm proliferating in a single-cell or nested pattern within the epidermis or dermis in invasive disease and often is accompanied by dermal lymphocytic inflammation.1 Immunohistochemistry demonstrates positive staining for CEA, CK7, and CK8, with negative staining for indicators of secondary EMPD including CK20 and CDX2.1,2
As part of the workup, it is critical to distinguish between primary disease and secondary EMPD.1 Beyond skin and clinical lymph node examination, additional workup should be based on age-appropriate and location-directed malignant neoplasm screenings, including colonoscopy, cystoscopy, prostate examination, mammography, and Papanicolaou test. Advanced imaging such as CT, positron emission tomography, or magnetic resonance imaging can be used to assess for metastatic disease if internal malignant neoplasms are present on initial screening or clinical lymphadenopathy is identified.2 Additionally, it can be helpful in the evaluation for nodal disease in cases of invasive EMPD.
The likelihood of associated underlying carcinomas varies depending on the site of involvement.38,39 For example, vulvar involvement constitutes approximately 65% of EMPD cases, with 11% to 20% of cases being associated with underlying gastrointestinal or genitourinary carcinomas. Involvement of the male genitalia, as in our 2 patients, is rare, accounting for approximately 14% of cases, 11% of which are associated with prostate, testicular, and bladder carcinoma. Perianal involvement comprises 20% of EMPD cases and has the greatest risk for underlying malignancy with an incidence of 33% to 86%, the majority of which are rectal or tubo-ovarian cancers.38,39 Consideration of the frequency and types of underlying carcinoma of respective sites of involvement can be helpful when ruling out secondary EMPD.
In both of our patients, palpable lymphadenopathy at the time of original diagnosis and histologic invasive disease on initial biopsy warranted thorough imaging and laboratory workup; there was no evidence of primary malignancy. Given the absence of an underlying carcinoma, both patients were classified as having metastatic primary EMPD.
Assessment of lymphadenopathy is an essential aspect of disease workup, as it is associated with a statistically higher rate of lymph node metastases. A study by Fujisawa et al20 demonstrated that 80% of patients with lymphadenopathy had regional metastases compared to only 15% of patients without clinical lymphadenopathy. The presence of invasive disease also has been shown to correspond with lymph node metastases.40 Ogata et al40 showed that 0% of cases with in situ EMPD had a positive sentinel lymph node biopsy (SLNB) compared to 4% and 43% in cases that showed evidence of microinvasion and dermal invasion, respectively. Lymph node metastases are associated with poor prognosis, with increasingly worse prognosis when there are multiple lymph nodes affected.41 In our case series, patient 1 had lymphadenopathy and both patients had invasive EMPD; they both later developed metastases and died.
Lymphadenopathy should be further investigated with imaging and biopsy or fine-needle aspiration.42 Recent expert consensus guidelines recommended this method of investigation over routine use of SLNB, as there is no evidence that a positive SLNB affects treatment that changes disease-specific survival.2
Treatment of EMPD—Surgical excision of the primary lesion is the first-line treatment of EMPD,1,2 which can be performed by wide local excision; however, studies have demonstrated higher recurrence-free survival with margin-controlled surgery (complete circumferential peripheral and deep margin assessment) or Mohs micrographic surgery (MMS), especially with CK7 immunostaining.2,37,43 The literature on MMS of invasive EMPD is sparse, accounting for 57 patients.25,37,44 Other reports describe management with surgical excision, wide local excision, regional resection, or vulvectomy, in addition to lymph node dissection, radiation therapy (RT), and/or chemotherapy.1-36,39,43-46 Despite the improved outcomes with MMS, the predominance of other surgical approaches in our search suggests that MMS may be currently underutilized for the treatment of invasive or locally advanced EMPD.
Among patients with unresectable disease or distant metastases, management includes RT with curative intent, chemotherapy, or a combination of both.1,2 In our review, 267 cases were treated using RT and 77 with chemotherapy. Radiation therapy is an effective therapeutic option with a reported response rate of 62% to 100% and can be employed as either primary or adjuvant treatment.3 For patients with lymph node metastasis the combination of RT and lymph node dissection has been shown to have improved outcomes compared to lymph node dissection alone, with 1 study showing a 5-year survival of 75% for patients who received adjuvant RT compared to 0% for lymph node dissection alone.45
There are currently no consensus guidelines on the best chemotherapeutic regimen for metastatic EMPD. Several regimens have been reported, including docetaxel monotherapy; low-dose 5-fluorouracil and cisplatin; combination chemotherapy FECOM (5-fluorouracil, epirubicin, carboplatin, vincristine, mitomycin); or combination therapy with cisplatin, epirubicin, and paclitaxel.1
Prognosis of Metastatic EMPD—Because invasive and metastatic EMPD is rare, its natural history is hard to predict. Poor prognosis is associated with nodule formation, tumor thickness, perianal or vaginal involvement, lymphovascular invasion, nodal metastasis, and distant metastasis. The 5-year survival for metastatic EMPD has been reported to be less than 10%.46 Our cases underscore the poor prognostic risk associated with metastatic EMPD.
For all cases of EMPD, close follow-up is warranted. Guidelines recommend physical examination with lymph node assessment every 3 to 6 months for 3 years and every 6 to 12 months for the subsequent 5 years.2 Specific recommendations for follow-up in invasive disease have not yet been described, though the 20% probability of developing an internal malignancy within 5 years after diagnosis and poor prognostic outcomes associated with invasive and metastatic disease support the need for close monitoring.2
Conclusion
Although in situ EMPD often is a slow-growing tumor with good prognosis, invasive disease has high potential to behave aggressively with high morbidity and mortality. Increased awareness and prompt identification of invasive EMPD, expedited comprehensive workup, and early multidisciplinary management might impact patient outcomes.
Acknowledgment—The authors would like to thank Ellen Aaronson, MLIS, AHIP (Mayo Clinic Libraries [Jacksonville, FL]), for creating and conducting the narrative literature search in the MEDLINE database.
- Hashimoto H, Ito T. Current management and treatment of extramammary Paget’s disease. Curr Treat Options Oncol. 2022;23:818-830. doi:10.1007/s11864-021-00923-3
- Kibbi N, Owen JL, Worley B, et al. Evidence-based clinical practice guidelines for extramammary Paget disease. JAMA Oncol. 2022;8:618-628. doi:10.1001/jamaoncol.2021.7148
- Morris CR, Hurst EA. Extramammary Paget’s disease: a review of the literature part II: treatment and prognosis. Dermatol Surg. 2020;46:305-311. doi:10.1097/DSS.0000000000002240
- Merritt BG, Degesys CA, Brodland DG. Extramammary Paget disease. Dermatol Clin. 2019;37:261-267. doi:10.1016/j.det.2019.02.002
- Aroche Gutierrez LL, Holloway SB, Donthi D, et al. Docetaxel treatment for widely metastatic invasive vulvar extramammary Paget’s disease with multifocal bone metastasis. Gynecol Oncol Rep. 2022;45:101114. doi:10.1016/j.gore.2022.101114
- Ueda M, Omori M, Sakai A. Invasive extramammary Paget’s disease with lymph node metastases and high-grade B-cell lymphoma. An Bras Dermatol. 2023;98:414-418. doi:10.1016/j.abd.2022.04.012
- Rathore R, Yadav D, Agarwal S, et al. Primary extra mammary Paget’s disease of vulva, with apocrine adenocarcinoma, signet ring cell differentiation and distant metastasis. J Family Reprod Health. 2020;14:276-280. doi:10.18502/jfrh.v14i4.5213
- Kawahara Y, Umeda Y, Yamaguchi B, et al. Long-term resolution of invasive extramammary Paget’s disease with multiple regional lymph node metastases solely with regional lymph node dissection. J Dermatol. 2021;48:E452-E453. doi:10.1111/1346-8138.16007
- Hanyu T, Fujitani S, Ito A, et al. Brain metastasis from extramammary Paget’s disease. Nagoya J Med Sci. 2020;82:791-798. doi:10.18999/nagjms.82.4.791
- Waki Y, Nobeyama Y, Ogawa T, et al. Case of extramammary Paget’s disease causing pulmonary tumor embolism. J Dermatol. 2020;47:E133-E134. doi:10.1111/1346-8138.15267
- Li ZG, Qin XJ. Extensive invasive extramammary Paget disease evaluated by F-18 FDG PET/CT: a case report. Medicine (Baltimore). 2015;94:E371. doi:10.1097/MD.0000000000000371
- Kato N, Matsue K, Sotodate A, et al. Extramammary Paget’s disease with distant skin metastasis. J Dermatol. 1996;23:408-414. doi:10.1111/j.1346-8138.1996.tb04043.x
- Hosomi M, Miyake O, Matsumiya K, et al. Extramammary Paget’s disease with a large mass in male genitalia: a case report. Article in Japanese. Hinyokika Kiyo. 1989;35:1981-1984.
- Hardy LE, Baxter L, Wan K, et al. Invasive cervical adenocarcinoma arising from extension of recurrent vulval Paget’s disease. BMJ Case Rep. 2020;13e232424. doi:10.1136/bcr-2019-232424
- Onaiwu CO, Ramirez PT, Kamat A, et al. Invasive extramammary Paget’s disease of the bladder diagnosed 18 years after noninvasive extramammary Paget’s disease of the vulva. Gynecol Oncol Case Rep. 2014;8:27-29. doi:10.1016/j.gynor.2014.03.004
- Yao H, Xie M, Fu S, et al. Survival analysis of patients with invasive extramammary Paget disease: implications of anatomic sites. BMC Cancer. 2018;18:403. doi:10.1186/s12885-018-4257-1
- Kato H, Watanabe S, Kariya K, et al. Efficacy of low-dose 5-fluorouracil/cisplatin therapy for invasive extramammary Paget’s disease. J Dermatol. 2018;45:560-563. doi:10.1111/1346-8138.14247
- Yoshino K, Fujisawa Y, Kiyohara Y, et al. Usefulness of docetaxel as first-line chemotherapy for metastatic extramammary Paget’s disease. J Dermatol. 2016;43:633-637. doi:10.1111/1346-8138.13200
- Shu B, Shen XX, Chen P, et al. Primary invasive extramammary Paget disease on penoscrotum: a clinicopathological analysis of 41 cases. Hum Pathol. 2016;47:70-77. doi:10.1016/j.humpath.2015.09.005References
- Fujisawa Y, Yoshino K, Kiyohara Y, et al. The role of sentinel lymph node biopsy in the management of invasive extramammary Paget’s disease: multi-center, retrospective study of 151 patients. J Dermatol Sci. 2015;79:38-42. doi:10.1016/j.jdermsci.2015.03.014
- Dai B, Kong YY, Chang K, et al. Primary invasive carcinoma associated with penoscrotal extramammary Paget’s disease: a clinicopathological analysis of 56 cases. BJU Int. 2015;115:153-160. doi:10.1111/bju.12776
- Shiomi T, Noguchi T, Nakayama H, et al. Clinicopathological study of invasive extramammary Paget’s disease: subgroup comparison according to invasion depth. J Eur Acad Dermatol Venereol. 2013;27:589-592. doi:10.1111/j.1468-3083.2012.04489.x
- Hatta N, Morita R, Yamada M, et al. Sentinel lymph node biopsy in patients with extramammary Paget’s disease. Dermatol Surg. 2004;30:1329-1334. doi:10.1111/j.1524-4725.2004.30377.x
- Karam A, Dorigo O. Treatment outcomes in a large cohort of patients with invasive extramammary Paget’s disease. Gynecol Oncol. 2012;125:346-351. doi:10.1016/j.ygyno.2012.01.032
- Guo L, Liu X, Li H, et al. Clinicopathological features of extramammary Paget’s disease: a report of 75 cases. Article in Chinese. Zhonghua Yi Xue Za Zhi. 2015;95:1751-1754.
- Kilts TP, Long B, Glasgow AE, et al. Invasive vulvar extramammary Paget’s disease in the United States. Gynecol Oncol. 2020;157:649-655. doi:10.1016/j.ygyno.2020.03.018
- Kusatake K, Harada Y, Mizumoto K, et al. Usefulness of sentinel lymph node biopsy for the detection of metastasis in the early stage of extramammary Paget’s disease. Eur J Dermatol. 2015;25:156-161. doi:10.1684/ejd.2015.2534
- Jeong BK, Kim KR. Invasive extramammary Paget disease of the vulva with signet ring cell morphology in a patient with signet ring cell carcinoma of the stomach: report of a case. Int J Gynecol Pathol. 2018;37:147-151. doi:10.1097/PGP.0000000000000405
- Pagnanelli M, De Nardi P, Martella S, et al. Local excision of a mucinous adenocarcinoma of the anal margin (extramammary Paget’s disease) and reconstruction with a bilateral V-Y flap. Case Rep Surg. 2019;2019:9073982. doi:10.1155/2019/9073982
- Sopracordevole F, Di Giuseppe J, De Piero G, et al. Surgical treatment of Paget disease of the vulva: prognostic significance of stromal invasion and surgical margin status. J Low Genit Tract Dis. 2016;20:184-188. doi:10.1097/LGT.0000000000000191
- Evans AT, Neven P. Invasive adenocarcinoma arising in extramammary Paget’s disease of the vulva. Histopathology. 1991;18:355-360. doi:10.1111/j.1365-2559.1991.tb00857.x
- Kitano A, Izumi M, Tamura K, et al. Brain metastasis from cutaneous squamous cell carcinoma coexistent with extramammary Paget’s disease: a case report. Pathol Int. 2019;69:619-625. doi:10.1111/pin.12846
- Miracco C, Francini E, Torre P, et al. Extramammary invasive Paget’s disease and apocrine angiomatous hamartoma: an unusual association. Eur J Dermatol. 2018;28:853-855. doi:10.1684/ejd.2018.3438
- Kambayashi Y, Fujimura T, Ohuchi K, et al. Advanced invasive extramammary Paget’s disease concomitant with cecal cancer possessing rare variant of TP53 single nucleotide polymorphism. Case Rep Oncol. 2019;12:855-860. doi:10.1159/000504339
- Fujimura T, Furudate S, Kambayashi Y, et al. Potential use of bisphosphonates in invasive extramammary Paget’s disease: an immunohistochemical investigation. Clin Dev Immunol. 2013;2013:164982. doi:10.1155/2013/164982
- Kawamura H, Ogata K, Miura H, et al. Patellar metastases. A report of two cases. Int Orthop. 1993;17:57-59. doi:10.1007/BF00195227
- Damavandy AA, Terushkin V, Zitelli JA, et al. Intraoperative immunostaining for cytokeratin-7 during Mohs micrographic surgery demonstrates low local recurrence rates in extramammary Paget’s disease. Dermatol Surg. 2018;44:354-364. doi:10.1097/DSS.0000000000001355
- Morris CR, Hurst EA. Extramammary Paget disease: a review of the literature-part I: history, epidemiology, pathogenesis, presentation, histopathology, and diagnostic work-up. Dermatol Surg. 2020;46:151-158. doi:10.1097/DSS.0000000000002064
- Simonds RM, Segal RJ, Sharma A. Extramammary Paget’s disease: a review of the literature. Int J Dermatol. 2019;58:871-879. doi:10.1111/ijd.14328
- Ogata D, Kiyohara Y, Yoshikawa S, et al. Usefulness of sentinel lymph node biopsy for prognostic prediction in extramammary Paget’s disease. Eur J Dermatol. 2016;26:254-259. doi:10.1684/ejd.2016.2744
- Ohara K, Fujisawa Y, Yoshino K, et al. A proposal for a TNM staging system for extramammary Paget disease: retrospective analysis of 301 patients with invasive primary tumors. J Dermatol Sci. 2016;83:234-239. doi:10.1016/j.jdermsci.2016.06.004
- Fujisawa Y, Yoshino K, Kiyohara Y, et al. The role of sentinel lymph node biopsy in the management of invasive extramammary Paget’s disease: multi-center, retrospective study of 151 patients. J Dermatol Sci. 2015;79:38-42. doi:10.1016/j.jdermsci.2015.03.014
- Kim SJ, Thompson AK, Zubair AS, et al. Surgical treatment and outcomes of patients with extramammary Paget disease: a cohort study. Dermatol Surg. 2017;43:708-714. doi:10.1097/DSS.0000000000001051
- Wollina U. Extensive invasive extramammary Paget’s disease: surgical treatment. J Cutan Aesthet Surg. 2013;6:41-44. doi:10.4103/0974-2077.110098
- Tsutsui K, Takahashi A, Muto Y, et al. Outcomes of lymph node dissection in the treatment of extramammary Paget’s disease: a single-institution study. J Dermatol. 2020;47:512-517. doi:10.1111/1346-8138.15285
- Guercio BJ, Iyer G, Kidwai WZ, et al. Treatment of metastatic extramammary Paget disease with combination ipilimumab and nivolumab: a case report. Case Rep Oncol. 2021;14:430-438. doi:10.1159/000514345
Extramammary Paget disease (EMPD) is a rare cutaneous malignancy typically seen in apocrine-rich areas, including the axillae and anogenital region. It presents as a slow-growing, erythematous patch or plaque that commonly is misdiagnosed as an infectious or inflammatory condition.1,2 Primary EMPD occurs as a intraepithelial neoplasm, whereas secondary EMPD occurs due to epidermotropic metastases or direct extension of an underlying adenocarcinoma into the skin.1 Most commonly, primary EMPD occurs in situ; however, when present, dermal invasion and metastases from the skin are associated with poorer outcomes.3 Given the rarity of metastatic disease, existing literature is limited to case reports and case series.
We present 2 patients with metastatic primary EMPD who had evidence of invasion on initial biopsy and died secondary to metastatic EMPD. We conducted a comprehensive review of the literature for invasive and metastatic EMPD to highlight key clinicopathologic features, treatment considerations, and the potential for rapid disease progression in cases of invasive EMPD.
Case Series
Patient 1—A 68-year-old White man with a history of breast cancer (in remission) presented to our clinic for further management of biopsy-proven scrotal EMPD. Prior to biopsy, he described a 6-month history of worsening scrotal rash treated with topical antifungals, oral antibiotics, and topical steroids due to presumed diagnosis of intertrigo, cellulitis, and dermatitis, respectively. Clinical examination showed indurated, erythematous, ulcerated plaques involving the bilateral groin, genitalia, and perineum (Figure 1). Skin biopsy confirmed a diagnosis of EMPD with both dermal and lymphovascular invasion. An immunohistochemical profile was positive for CK7 and carcinoembryonic antigen (CEA) and negative for CK20 (Figure 2).


At presentation, the patient had palpable lymphadenopathy and scrotal edema concerning for inguinal and iliac lymph node metastases. Workup for an underlying adenocarcinoma included computed tomography (CT) of the chest, abdomen, and pelvis; urologic consultation with cystoscopy; and a screening colonoscopy. The CT scan revealed multiple enlarged inguinal and external iliac lymph nodes. Fine-needle aspiration revealed CK7- and CEA-positive neoplastic cells consistent with metastatic EMPD. The patient was treated with 6 cycles of carboplatin-paclitaxel, palliative radiation therapy, and pembrolizumab with minimal response to treatment and development of osteolytic vertebral lesions concerning for disease progression. He died 1 year after the initial diagnosis secondary to the disease.
Patient 2—A 79-year-old White man presented for further management of an outside diagnosis of superficially invasive primary EMPD of the bilateral inguinal folds and scrotum that had been present for 5 months prior to biopsy and diagnosis. Clinical examination at initial presentation revealed erythematous patches of the bilateral inguinal folds and scrotum, as well as an erythematous scaling plaque in the right axilla. There was no palpable clinical lymphadenopathy. Biopsy of the axilla and groin were both consistent with invasive EMPD with positive staining for CK7 and negative staining for CK20 and CDX2. Workup for underlying adenocarcinoma with whole-body positron emission tomography/CT, mammography, esophagogastroduodenoscopy, serum CEA, colonoscopy, and cystoscopy were all negative for a metastatic adenocarcinoma. There was no imaging or clinical evidence of lymphadenopathy. Complete circumferential peripheral and deep-margin assessment was performed in a staged manner on both sites, and negative margins were obtained.
Surveillance imaging 6 months after surgery revealed suspicious hepatic lesions. Fine-needle aspiration of the hepatic lesions demonstrated positive staining for CK7 and negative staining for CK20, CDX2, prostate-specific antigen, and thyroid transcription factor 1, consistent with metastatic EMPD. Oncology recommended carboplatin and docetaxel or docetaxel monotherapy chemotherapy. The patient was further managed by an outside oncologist due to ease of travel but died secondary to the disease 15 months following the initial diagnosis.
Comment
Extramammary Paget disease is an uncommon cutaneous malignancy that manifests as pruritic erythematous plaques within apocrine-rich areas such as the genitalia, axillae, or anal region. It most commonly occurs in patients older than 65 years, with White women and Asian men being affected at disproportionately higher rates.1,4 Delay in diagnosis is common, as EMPD can mimic other benign inflammatory or infectious conditions, including contact dermatitis, seborrheic dermatitis, tinea, candidiasis, and eczema.1
Metastatic and multifocal cases of primary EMPD are especially rare. According to a search of PubMed articles indexed for MEDLINE published through December 2023 using the terms extramammary Paget disease, EMPD, neoplasm metastasis, invasive extramammary, and neoplasm invasiveness, we identified 5040 cases of invasive EMPD and 477 cases of metastatic EMPD.5-37 Of the reports that disclosed patient demographic information, 3627 patients were female 1410 were male, and the mean age was 67 years. Sites of metastases included regional lymph nodes, liver, lungs, cervix, bladder, bone, brain, skin, kidney, and adrenal glands
Workup for EMPD—The initial steps for workup of EMPD include a thorough physical examination and lymph node assessment. A skin biopsy also should be performed for patients presenting with refractory, pruritic, and eczematous rashes in apocrine-rich areas to evaluate for EMPD.1 Characterization of large and complex tumors is better achieved through multiple biopsies with particular focus on nodular or thickened areas, as these may indicate invasive disease.2 Primary EMPD is characterized by pagetoid cells with abundant pale cytoplasm proliferating in a single-cell or nested pattern within the epidermis or dermis in invasive disease and often is accompanied by dermal lymphocytic inflammation.1 Immunohistochemistry demonstrates positive staining for CEA, CK7, and CK8, with negative staining for indicators of secondary EMPD including CK20 and CDX2.1,2
As part of the workup, it is critical to distinguish between primary disease and secondary EMPD.1 Beyond skin and clinical lymph node examination, additional workup should be based on age-appropriate and location-directed malignant neoplasm screenings, including colonoscopy, cystoscopy, prostate examination, mammography, and Papanicolaou test. Advanced imaging such as CT, positron emission tomography, or magnetic resonance imaging can be used to assess for metastatic disease if internal malignant neoplasms are present on initial screening or clinical lymphadenopathy is identified.2 Additionally, it can be helpful in the evaluation for nodal disease in cases of invasive EMPD.
The likelihood of associated underlying carcinomas varies depending on the site of involvement.38,39 For example, vulvar involvement constitutes approximately 65% of EMPD cases, with 11% to 20% of cases being associated with underlying gastrointestinal or genitourinary carcinomas. Involvement of the male genitalia, as in our 2 patients, is rare, accounting for approximately 14% of cases, 11% of which are associated with prostate, testicular, and bladder carcinoma. Perianal involvement comprises 20% of EMPD cases and has the greatest risk for underlying malignancy with an incidence of 33% to 86%, the majority of which are rectal or tubo-ovarian cancers.38,39 Consideration of the frequency and types of underlying carcinoma of respective sites of involvement can be helpful when ruling out secondary EMPD.
In both of our patients, palpable lymphadenopathy at the time of original diagnosis and histologic invasive disease on initial biopsy warranted thorough imaging and laboratory workup; there was no evidence of primary malignancy. Given the absence of an underlying carcinoma, both patients were classified as having metastatic primary EMPD.
Assessment of lymphadenopathy is an essential aspect of disease workup, as it is associated with a statistically higher rate of lymph node metastases. A study by Fujisawa et al20 demonstrated that 80% of patients with lymphadenopathy had regional metastases compared to only 15% of patients without clinical lymphadenopathy. The presence of invasive disease also has been shown to correspond with lymph node metastases.40 Ogata et al40 showed that 0% of cases with in situ EMPD had a positive sentinel lymph node biopsy (SLNB) compared to 4% and 43% in cases that showed evidence of microinvasion and dermal invasion, respectively. Lymph node metastases are associated with poor prognosis, with increasingly worse prognosis when there are multiple lymph nodes affected.41 In our case series, patient 1 had lymphadenopathy and both patients had invasive EMPD; they both later developed metastases and died.
Lymphadenopathy should be further investigated with imaging and biopsy or fine-needle aspiration.42 Recent expert consensus guidelines recommended this method of investigation over routine use of SLNB, as there is no evidence that a positive SLNB affects treatment that changes disease-specific survival.2
Treatment of EMPD—Surgical excision of the primary lesion is the first-line treatment of EMPD,1,2 which can be performed by wide local excision; however, studies have demonstrated higher recurrence-free survival with margin-controlled surgery (complete circumferential peripheral and deep margin assessment) or Mohs micrographic surgery (MMS), especially with CK7 immunostaining.2,37,43 The literature on MMS of invasive EMPD is sparse, accounting for 57 patients.25,37,44 Other reports describe management with surgical excision, wide local excision, regional resection, or vulvectomy, in addition to lymph node dissection, radiation therapy (RT), and/or chemotherapy.1-36,39,43-46 Despite the improved outcomes with MMS, the predominance of other surgical approaches in our search suggests that MMS may be currently underutilized for the treatment of invasive or locally advanced EMPD.
Among patients with unresectable disease or distant metastases, management includes RT with curative intent, chemotherapy, or a combination of both.1,2 In our review, 267 cases were treated using RT and 77 with chemotherapy. Radiation therapy is an effective therapeutic option with a reported response rate of 62% to 100% and can be employed as either primary or adjuvant treatment.3 For patients with lymph node metastasis the combination of RT and lymph node dissection has been shown to have improved outcomes compared to lymph node dissection alone, with 1 study showing a 5-year survival of 75% for patients who received adjuvant RT compared to 0% for lymph node dissection alone.45
There are currently no consensus guidelines on the best chemotherapeutic regimen for metastatic EMPD. Several regimens have been reported, including docetaxel monotherapy; low-dose 5-fluorouracil and cisplatin; combination chemotherapy FECOM (5-fluorouracil, epirubicin, carboplatin, vincristine, mitomycin); or combination therapy with cisplatin, epirubicin, and paclitaxel.1
Prognosis of Metastatic EMPD—Because invasive and metastatic EMPD is rare, its natural history is hard to predict. Poor prognosis is associated with nodule formation, tumor thickness, perianal or vaginal involvement, lymphovascular invasion, nodal metastasis, and distant metastasis. The 5-year survival for metastatic EMPD has been reported to be less than 10%.46 Our cases underscore the poor prognostic risk associated with metastatic EMPD.
For all cases of EMPD, close follow-up is warranted. Guidelines recommend physical examination with lymph node assessment every 3 to 6 months for 3 years and every 6 to 12 months for the subsequent 5 years.2 Specific recommendations for follow-up in invasive disease have not yet been described, though the 20% probability of developing an internal malignancy within 5 years after diagnosis and poor prognostic outcomes associated with invasive and metastatic disease support the need for close monitoring.2
Conclusion
Although in situ EMPD often is a slow-growing tumor with good prognosis, invasive disease has high potential to behave aggressively with high morbidity and mortality. Increased awareness and prompt identification of invasive EMPD, expedited comprehensive workup, and early multidisciplinary management might impact patient outcomes.
Acknowledgment—The authors would like to thank Ellen Aaronson, MLIS, AHIP (Mayo Clinic Libraries [Jacksonville, FL]), for creating and conducting the narrative literature search in the MEDLINE database.
Extramammary Paget disease (EMPD) is a rare cutaneous malignancy typically seen in apocrine-rich areas, including the axillae and anogenital region. It presents as a slow-growing, erythematous patch or plaque that commonly is misdiagnosed as an infectious or inflammatory condition.1,2 Primary EMPD occurs as a intraepithelial neoplasm, whereas secondary EMPD occurs due to epidermotropic metastases or direct extension of an underlying adenocarcinoma into the skin.1 Most commonly, primary EMPD occurs in situ; however, when present, dermal invasion and metastases from the skin are associated with poorer outcomes.3 Given the rarity of metastatic disease, existing literature is limited to case reports and case series.
We present 2 patients with metastatic primary EMPD who had evidence of invasion on initial biopsy and died secondary to metastatic EMPD. We conducted a comprehensive review of the literature for invasive and metastatic EMPD to highlight key clinicopathologic features, treatment considerations, and the potential for rapid disease progression in cases of invasive EMPD.
Case Series
Patient 1—A 68-year-old White man with a history of breast cancer (in remission) presented to our clinic for further management of biopsy-proven scrotal EMPD. Prior to biopsy, he described a 6-month history of worsening scrotal rash treated with topical antifungals, oral antibiotics, and topical steroids due to presumed diagnosis of intertrigo, cellulitis, and dermatitis, respectively. Clinical examination showed indurated, erythematous, ulcerated plaques involving the bilateral groin, genitalia, and perineum (Figure 1). Skin biopsy confirmed a diagnosis of EMPD with both dermal and lymphovascular invasion. An immunohistochemical profile was positive for CK7 and carcinoembryonic antigen (CEA) and negative for CK20 (Figure 2).


At presentation, the patient had palpable lymphadenopathy and scrotal edema concerning for inguinal and iliac lymph node metastases. Workup for an underlying adenocarcinoma included computed tomography (CT) of the chest, abdomen, and pelvis; urologic consultation with cystoscopy; and a screening colonoscopy. The CT scan revealed multiple enlarged inguinal and external iliac lymph nodes. Fine-needle aspiration revealed CK7- and CEA-positive neoplastic cells consistent with metastatic EMPD. The patient was treated with 6 cycles of carboplatin-paclitaxel, palliative radiation therapy, and pembrolizumab with minimal response to treatment and development of osteolytic vertebral lesions concerning for disease progression. He died 1 year after the initial diagnosis secondary to the disease.
Patient 2—A 79-year-old White man presented for further management of an outside diagnosis of superficially invasive primary EMPD of the bilateral inguinal folds and scrotum that had been present for 5 months prior to biopsy and diagnosis. Clinical examination at initial presentation revealed erythematous patches of the bilateral inguinal folds and scrotum, as well as an erythematous scaling plaque in the right axilla. There was no palpable clinical lymphadenopathy. Biopsy of the axilla and groin were both consistent with invasive EMPD with positive staining for CK7 and negative staining for CK20 and CDX2. Workup for underlying adenocarcinoma with whole-body positron emission tomography/CT, mammography, esophagogastroduodenoscopy, serum CEA, colonoscopy, and cystoscopy were all negative for a metastatic adenocarcinoma. There was no imaging or clinical evidence of lymphadenopathy. Complete circumferential peripheral and deep-margin assessment was performed in a staged manner on both sites, and negative margins were obtained.
Surveillance imaging 6 months after surgery revealed suspicious hepatic lesions. Fine-needle aspiration of the hepatic lesions demonstrated positive staining for CK7 and negative staining for CK20, CDX2, prostate-specific antigen, and thyroid transcription factor 1, consistent with metastatic EMPD. Oncology recommended carboplatin and docetaxel or docetaxel monotherapy chemotherapy. The patient was further managed by an outside oncologist due to ease of travel but died secondary to the disease 15 months following the initial diagnosis.
Comment
Extramammary Paget disease is an uncommon cutaneous malignancy that manifests as pruritic erythematous plaques within apocrine-rich areas such as the genitalia, axillae, or anal region. It most commonly occurs in patients older than 65 years, with White women and Asian men being affected at disproportionately higher rates.1,4 Delay in diagnosis is common, as EMPD can mimic other benign inflammatory or infectious conditions, including contact dermatitis, seborrheic dermatitis, tinea, candidiasis, and eczema.1
Metastatic and multifocal cases of primary EMPD are especially rare. According to a search of PubMed articles indexed for MEDLINE published through December 2023 using the terms extramammary Paget disease, EMPD, neoplasm metastasis, invasive extramammary, and neoplasm invasiveness, we identified 5040 cases of invasive EMPD and 477 cases of metastatic EMPD.5-37 Of the reports that disclosed patient demographic information, 3627 patients were female 1410 were male, and the mean age was 67 years. Sites of metastases included regional lymph nodes, liver, lungs, cervix, bladder, bone, brain, skin, kidney, and adrenal glands
Workup for EMPD—The initial steps for workup of EMPD include a thorough physical examination and lymph node assessment. A skin biopsy also should be performed for patients presenting with refractory, pruritic, and eczematous rashes in apocrine-rich areas to evaluate for EMPD.1 Characterization of large and complex tumors is better achieved through multiple biopsies with particular focus on nodular or thickened areas, as these may indicate invasive disease.2 Primary EMPD is characterized by pagetoid cells with abundant pale cytoplasm proliferating in a single-cell or nested pattern within the epidermis or dermis in invasive disease and often is accompanied by dermal lymphocytic inflammation.1 Immunohistochemistry demonstrates positive staining for CEA, CK7, and CK8, with negative staining for indicators of secondary EMPD including CK20 and CDX2.1,2
As part of the workup, it is critical to distinguish between primary disease and secondary EMPD.1 Beyond skin and clinical lymph node examination, additional workup should be based on age-appropriate and location-directed malignant neoplasm screenings, including colonoscopy, cystoscopy, prostate examination, mammography, and Papanicolaou test. Advanced imaging such as CT, positron emission tomography, or magnetic resonance imaging can be used to assess for metastatic disease if internal malignant neoplasms are present on initial screening or clinical lymphadenopathy is identified.2 Additionally, it can be helpful in the evaluation for nodal disease in cases of invasive EMPD.
The likelihood of associated underlying carcinomas varies depending on the site of involvement.38,39 For example, vulvar involvement constitutes approximately 65% of EMPD cases, with 11% to 20% of cases being associated with underlying gastrointestinal or genitourinary carcinomas. Involvement of the male genitalia, as in our 2 patients, is rare, accounting for approximately 14% of cases, 11% of which are associated with prostate, testicular, and bladder carcinoma. Perianal involvement comprises 20% of EMPD cases and has the greatest risk for underlying malignancy with an incidence of 33% to 86%, the majority of which are rectal or tubo-ovarian cancers.38,39 Consideration of the frequency and types of underlying carcinoma of respective sites of involvement can be helpful when ruling out secondary EMPD.
In both of our patients, palpable lymphadenopathy at the time of original diagnosis and histologic invasive disease on initial biopsy warranted thorough imaging and laboratory workup; there was no evidence of primary malignancy. Given the absence of an underlying carcinoma, both patients were classified as having metastatic primary EMPD.
Assessment of lymphadenopathy is an essential aspect of disease workup, as it is associated with a statistically higher rate of lymph node metastases. A study by Fujisawa et al20 demonstrated that 80% of patients with lymphadenopathy had regional metastases compared to only 15% of patients without clinical lymphadenopathy. The presence of invasive disease also has been shown to correspond with lymph node metastases.40 Ogata et al40 showed that 0% of cases with in situ EMPD had a positive sentinel lymph node biopsy (SLNB) compared to 4% and 43% in cases that showed evidence of microinvasion and dermal invasion, respectively. Lymph node metastases are associated with poor prognosis, with increasingly worse prognosis when there are multiple lymph nodes affected.41 In our case series, patient 1 had lymphadenopathy and both patients had invasive EMPD; they both later developed metastases and died.
Lymphadenopathy should be further investigated with imaging and biopsy or fine-needle aspiration.42 Recent expert consensus guidelines recommended this method of investigation over routine use of SLNB, as there is no evidence that a positive SLNB affects treatment that changes disease-specific survival.2
Treatment of EMPD—Surgical excision of the primary lesion is the first-line treatment of EMPD,1,2 which can be performed by wide local excision; however, studies have demonstrated higher recurrence-free survival with margin-controlled surgery (complete circumferential peripheral and deep margin assessment) or Mohs micrographic surgery (MMS), especially with CK7 immunostaining.2,37,43 The literature on MMS of invasive EMPD is sparse, accounting for 57 patients.25,37,44 Other reports describe management with surgical excision, wide local excision, regional resection, or vulvectomy, in addition to lymph node dissection, radiation therapy (RT), and/or chemotherapy.1-36,39,43-46 Despite the improved outcomes with MMS, the predominance of other surgical approaches in our search suggests that MMS may be currently underutilized for the treatment of invasive or locally advanced EMPD.
Among patients with unresectable disease or distant metastases, management includes RT with curative intent, chemotherapy, or a combination of both.1,2 In our review, 267 cases were treated using RT and 77 with chemotherapy. Radiation therapy is an effective therapeutic option with a reported response rate of 62% to 100% and can be employed as either primary or adjuvant treatment.3 For patients with lymph node metastasis the combination of RT and lymph node dissection has been shown to have improved outcomes compared to lymph node dissection alone, with 1 study showing a 5-year survival of 75% for patients who received adjuvant RT compared to 0% for lymph node dissection alone.45
There are currently no consensus guidelines on the best chemotherapeutic regimen for metastatic EMPD. Several regimens have been reported, including docetaxel monotherapy; low-dose 5-fluorouracil and cisplatin; combination chemotherapy FECOM (5-fluorouracil, epirubicin, carboplatin, vincristine, mitomycin); or combination therapy with cisplatin, epirubicin, and paclitaxel.1
Prognosis of Metastatic EMPD—Because invasive and metastatic EMPD is rare, its natural history is hard to predict. Poor prognosis is associated with nodule formation, tumor thickness, perianal or vaginal involvement, lymphovascular invasion, nodal metastasis, and distant metastasis. The 5-year survival for metastatic EMPD has been reported to be less than 10%.46 Our cases underscore the poor prognostic risk associated with metastatic EMPD.
For all cases of EMPD, close follow-up is warranted. Guidelines recommend physical examination with lymph node assessment every 3 to 6 months for 3 years and every 6 to 12 months for the subsequent 5 years.2 Specific recommendations for follow-up in invasive disease have not yet been described, though the 20% probability of developing an internal malignancy within 5 years after diagnosis and poor prognostic outcomes associated with invasive and metastatic disease support the need for close monitoring.2
Conclusion
Although in situ EMPD often is a slow-growing tumor with good prognosis, invasive disease has high potential to behave aggressively with high morbidity and mortality. Increased awareness and prompt identification of invasive EMPD, expedited comprehensive workup, and early multidisciplinary management might impact patient outcomes.
Acknowledgment—The authors would like to thank Ellen Aaronson, MLIS, AHIP (Mayo Clinic Libraries [Jacksonville, FL]), for creating and conducting the narrative literature search in the MEDLINE database.
- Hashimoto H, Ito T. Current management and treatment of extramammary Paget’s disease. Curr Treat Options Oncol. 2022;23:818-830. doi:10.1007/s11864-021-00923-3
- Kibbi N, Owen JL, Worley B, et al. Evidence-based clinical practice guidelines for extramammary Paget disease. JAMA Oncol. 2022;8:618-628. doi:10.1001/jamaoncol.2021.7148
- Morris CR, Hurst EA. Extramammary Paget’s disease: a review of the literature part II: treatment and prognosis. Dermatol Surg. 2020;46:305-311. doi:10.1097/DSS.0000000000002240
- Merritt BG, Degesys CA, Brodland DG. Extramammary Paget disease. Dermatol Clin. 2019;37:261-267. doi:10.1016/j.det.2019.02.002
- Aroche Gutierrez LL, Holloway SB, Donthi D, et al. Docetaxel treatment for widely metastatic invasive vulvar extramammary Paget’s disease with multifocal bone metastasis. Gynecol Oncol Rep. 2022;45:101114. doi:10.1016/j.gore.2022.101114
- Ueda M, Omori M, Sakai A. Invasive extramammary Paget’s disease with lymph node metastases and high-grade B-cell lymphoma. An Bras Dermatol. 2023;98:414-418. doi:10.1016/j.abd.2022.04.012
- Rathore R, Yadav D, Agarwal S, et al. Primary extra mammary Paget’s disease of vulva, with apocrine adenocarcinoma, signet ring cell differentiation and distant metastasis. J Family Reprod Health. 2020;14:276-280. doi:10.18502/jfrh.v14i4.5213
- Kawahara Y, Umeda Y, Yamaguchi B, et al. Long-term resolution of invasive extramammary Paget’s disease with multiple regional lymph node metastases solely with regional lymph node dissection. J Dermatol. 2021;48:E452-E453. doi:10.1111/1346-8138.16007
- Hanyu T, Fujitani S, Ito A, et al. Brain metastasis from extramammary Paget’s disease. Nagoya J Med Sci. 2020;82:791-798. doi:10.18999/nagjms.82.4.791
- Waki Y, Nobeyama Y, Ogawa T, et al. Case of extramammary Paget’s disease causing pulmonary tumor embolism. J Dermatol. 2020;47:E133-E134. doi:10.1111/1346-8138.15267
- Li ZG, Qin XJ. Extensive invasive extramammary Paget disease evaluated by F-18 FDG PET/CT: a case report. Medicine (Baltimore). 2015;94:E371. doi:10.1097/MD.0000000000000371
- Kato N, Matsue K, Sotodate A, et al. Extramammary Paget’s disease with distant skin metastasis. J Dermatol. 1996;23:408-414. doi:10.1111/j.1346-8138.1996.tb04043.x
- Hosomi M, Miyake O, Matsumiya K, et al. Extramammary Paget’s disease with a large mass in male genitalia: a case report. Article in Japanese. Hinyokika Kiyo. 1989;35:1981-1984.
- Hardy LE, Baxter L, Wan K, et al. Invasive cervical adenocarcinoma arising from extension of recurrent vulval Paget’s disease. BMJ Case Rep. 2020;13e232424. doi:10.1136/bcr-2019-232424
- Onaiwu CO, Ramirez PT, Kamat A, et al. Invasive extramammary Paget’s disease of the bladder diagnosed 18 years after noninvasive extramammary Paget’s disease of the vulva. Gynecol Oncol Case Rep. 2014;8:27-29. doi:10.1016/j.gynor.2014.03.004
- Yao H, Xie M, Fu S, et al. Survival analysis of patients with invasive extramammary Paget disease: implications of anatomic sites. BMC Cancer. 2018;18:403. doi:10.1186/s12885-018-4257-1
- Kato H, Watanabe S, Kariya K, et al. Efficacy of low-dose 5-fluorouracil/cisplatin therapy for invasive extramammary Paget’s disease. J Dermatol. 2018;45:560-563. doi:10.1111/1346-8138.14247
- Yoshino K, Fujisawa Y, Kiyohara Y, et al. Usefulness of docetaxel as first-line chemotherapy for metastatic extramammary Paget’s disease. J Dermatol. 2016;43:633-637. doi:10.1111/1346-8138.13200
- Shu B, Shen XX, Chen P, et al. Primary invasive extramammary Paget disease on penoscrotum: a clinicopathological analysis of 41 cases. Hum Pathol. 2016;47:70-77. doi:10.1016/j.humpath.2015.09.005References
- Fujisawa Y, Yoshino K, Kiyohara Y, et al. The role of sentinel lymph node biopsy in the management of invasive extramammary Paget’s disease: multi-center, retrospective study of 151 patients. J Dermatol Sci. 2015;79:38-42. doi:10.1016/j.jdermsci.2015.03.014
- Dai B, Kong YY, Chang K, et al. Primary invasive carcinoma associated with penoscrotal extramammary Paget’s disease: a clinicopathological analysis of 56 cases. BJU Int. 2015;115:153-160. doi:10.1111/bju.12776
- Shiomi T, Noguchi T, Nakayama H, et al. Clinicopathological study of invasive extramammary Paget’s disease: subgroup comparison according to invasion depth. J Eur Acad Dermatol Venereol. 2013;27:589-592. doi:10.1111/j.1468-3083.2012.04489.x
- Hatta N, Morita R, Yamada M, et al. Sentinel lymph node biopsy in patients with extramammary Paget’s disease. Dermatol Surg. 2004;30:1329-1334. doi:10.1111/j.1524-4725.2004.30377.x
- Karam A, Dorigo O. Treatment outcomes in a large cohort of patients with invasive extramammary Paget’s disease. Gynecol Oncol. 2012;125:346-351. doi:10.1016/j.ygyno.2012.01.032
- Guo L, Liu X, Li H, et al. Clinicopathological features of extramammary Paget’s disease: a report of 75 cases. Article in Chinese. Zhonghua Yi Xue Za Zhi. 2015;95:1751-1754.
- Kilts TP, Long B, Glasgow AE, et al. Invasive vulvar extramammary Paget’s disease in the United States. Gynecol Oncol. 2020;157:649-655. doi:10.1016/j.ygyno.2020.03.018
- Kusatake K, Harada Y, Mizumoto K, et al. Usefulness of sentinel lymph node biopsy for the detection of metastasis in the early stage of extramammary Paget’s disease. Eur J Dermatol. 2015;25:156-161. doi:10.1684/ejd.2015.2534
- Jeong BK, Kim KR. Invasive extramammary Paget disease of the vulva with signet ring cell morphology in a patient with signet ring cell carcinoma of the stomach: report of a case. Int J Gynecol Pathol. 2018;37:147-151. doi:10.1097/PGP.0000000000000405
- Pagnanelli M, De Nardi P, Martella S, et al. Local excision of a mucinous adenocarcinoma of the anal margin (extramammary Paget’s disease) and reconstruction with a bilateral V-Y flap. Case Rep Surg. 2019;2019:9073982. doi:10.1155/2019/9073982
- Sopracordevole F, Di Giuseppe J, De Piero G, et al. Surgical treatment of Paget disease of the vulva: prognostic significance of stromal invasion and surgical margin status. J Low Genit Tract Dis. 2016;20:184-188. doi:10.1097/LGT.0000000000000191
- Evans AT, Neven P. Invasive adenocarcinoma arising in extramammary Paget’s disease of the vulva. Histopathology. 1991;18:355-360. doi:10.1111/j.1365-2559.1991.tb00857.x
- Kitano A, Izumi M, Tamura K, et al. Brain metastasis from cutaneous squamous cell carcinoma coexistent with extramammary Paget’s disease: a case report. Pathol Int. 2019;69:619-625. doi:10.1111/pin.12846
- Miracco C, Francini E, Torre P, et al. Extramammary invasive Paget’s disease and apocrine angiomatous hamartoma: an unusual association. Eur J Dermatol. 2018;28:853-855. doi:10.1684/ejd.2018.3438
- Kambayashi Y, Fujimura T, Ohuchi K, et al. Advanced invasive extramammary Paget’s disease concomitant with cecal cancer possessing rare variant of TP53 single nucleotide polymorphism. Case Rep Oncol. 2019;12:855-860. doi:10.1159/000504339
- Fujimura T, Furudate S, Kambayashi Y, et al. Potential use of bisphosphonates in invasive extramammary Paget’s disease: an immunohistochemical investigation. Clin Dev Immunol. 2013;2013:164982. doi:10.1155/2013/164982
- Kawamura H, Ogata K, Miura H, et al. Patellar metastases. A report of two cases. Int Orthop. 1993;17:57-59. doi:10.1007/BF00195227
- Damavandy AA, Terushkin V, Zitelli JA, et al. Intraoperative immunostaining for cytokeratin-7 during Mohs micrographic surgery demonstrates low local recurrence rates in extramammary Paget’s disease. Dermatol Surg. 2018;44:354-364. doi:10.1097/DSS.0000000000001355
- Morris CR, Hurst EA. Extramammary Paget disease: a review of the literature-part I: history, epidemiology, pathogenesis, presentation, histopathology, and diagnostic work-up. Dermatol Surg. 2020;46:151-158. doi:10.1097/DSS.0000000000002064
- Simonds RM, Segal RJ, Sharma A. Extramammary Paget’s disease: a review of the literature. Int J Dermatol. 2019;58:871-879. doi:10.1111/ijd.14328
- Ogata D, Kiyohara Y, Yoshikawa S, et al. Usefulness of sentinel lymph node biopsy for prognostic prediction in extramammary Paget’s disease. Eur J Dermatol. 2016;26:254-259. doi:10.1684/ejd.2016.2744
- Ohara K, Fujisawa Y, Yoshino K, et al. A proposal for a TNM staging system for extramammary Paget disease: retrospective analysis of 301 patients with invasive primary tumors. J Dermatol Sci. 2016;83:234-239. doi:10.1016/j.jdermsci.2016.06.004
- Fujisawa Y, Yoshino K, Kiyohara Y, et al. The role of sentinel lymph node biopsy in the management of invasive extramammary Paget’s disease: multi-center, retrospective study of 151 patients. J Dermatol Sci. 2015;79:38-42. doi:10.1016/j.jdermsci.2015.03.014
- Kim SJ, Thompson AK, Zubair AS, et al. Surgical treatment and outcomes of patients with extramammary Paget disease: a cohort study. Dermatol Surg. 2017;43:708-714. doi:10.1097/DSS.0000000000001051
- Wollina U. Extensive invasive extramammary Paget’s disease: surgical treatment. J Cutan Aesthet Surg. 2013;6:41-44. doi:10.4103/0974-2077.110098
- Tsutsui K, Takahashi A, Muto Y, et al. Outcomes of lymph node dissection in the treatment of extramammary Paget’s disease: a single-institution study. J Dermatol. 2020;47:512-517. doi:10.1111/1346-8138.15285
- Guercio BJ, Iyer G, Kidwai WZ, et al. Treatment of metastatic extramammary Paget disease with combination ipilimumab and nivolumab: a case report. Case Rep Oncol. 2021;14:430-438. doi:10.1159/000514345
- Hashimoto H, Ito T. Current management and treatment of extramammary Paget’s disease. Curr Treat Options Oncol. 2022;23:818-830. doi:10.1007/s11864-021-00923-3
- Kibbi N, Owen JL, Worley B, et al. Evidence-based clinical practice guidelines for extramammary Paget disease. JAMA Oncol. 2022;8:618-628. doi:10.1001/jamaoncol.2021.7148
- Morris CR, Hurst EA. Extramammary Paget’s disease: a review of the literature part II: treatment and prognosis. Dermatol Surg. 2020;46:305-311. doi:10.1097/DSS.0000000000002240
- Merritt BG, Degesys CA, Brodland DG. Extramammary Paget disease. Dermatol Clin. 2019;37:261-267. doi:10.1016/j.det.2019.02.002
- Aroche Gutierrez LL, Holloway SB, Donthi D, et al. Docetaxel treatment for widely metastatic invasive vulvar extramammary Paget’s disease with multifocal bone metastasis. Gynecol Oncol Rep. 2022;45:101114. doi:10.1016/j.gore.2022.101114
- Ueda M, Omori M, Sakai A. Invasive extramammary Paget’s disease with lymph node metastases and high-grade B-cell lymphoma. An Bras Dermatol. 2023;98:414-418. doi:10.1016/j.abd.2022.04.012
- Rathore R, Yadav D, Agarwal S, et al. Primary extra mammary Paget’s disease of vulva, with apocrine adenocarcinoma, signet ring cell differentiation and distant metastasis. J Family Reprod Health. 2020;14:276-280. doi:10.18502/jfrh.v14i4.5213
- Kawahara Y, Umeda Y, Yamaguchi B, et al. Long-term resolution of invasive extramammary Paget’s disease with multiple regional lymph node metastases solely with regional lymph node dissection. J Dermatol. 2021;48:E452-E453. doi:10.1111/1346-8138.16007
- Hanyu T, Fujitani S, Ito A, et al. Brain metastasis from extramammary Paget’s disease. Nagoya J Med Sci. 2020;82:791-798. doi:10.18999/nagjms.82.4.791
- Waki Y, Nobeyama Y, Ogawa T, et al. Case of extramammary Paget’s disease causing pulmonary tumor embolism. J Dermatol. 2020;47:E133-E134. doi:10.1111/1346-8138.15267
- Li ZG, Qin XJ. Extensive invasive extramammary Paget disease evaluated by F-18 FDG PET/CT: a case report. Medicine (Baltimore). 2015;94:E371. doi:10.1097/MD.0000000000000371
- Kato N, Matsue K, Sotodate A, et al. Extramammary Paget’s disease with distant skin metastasis. J Dermatol. 1996;23:408-414. doi:10.1111/j.1346-8138.1996.tb04043.x
- Hosomi M, Miyake O, Matsumiya K, et al. Extramammary Paget’s disease with a large mass in male genitalia: a case report. Article in Japanese. Hinyokika Kiyo. 1989;35:1981-1984.
- Hardy LE, Baxter L, Wan K, et al. Invasive cervical adenocarcinoma arising from extension of recurrent vulval Paget’s disease. BMJ Case Rep. 2020;13e232424. doi:10.1136/bcr-2019-232424
- Onaiwu CO, Ramirez PT, Kamat A, et al. Invasive extramammary Paget’s disease of the bladder diagnosed 18 years after noninvasive extramammary Paget’s disease of the vulva. Gynecol Oncol Case Rep. 2014;8:27-29. doi:10.1016/j.gynor.2014.03.004
- Yao H, Xie M, Fu S, et al. Survival analysis of patients with invasive extramammary Paget disease: implications of anatomic sites. BMC Cancer. 2018;18:403. doi:10.1186/s12885-018-4257-1
- Kato H, Watanabe S, Kariya K, et al. Efficacy of low-dose 5-fluorouracil/cisplatin therapy for invasive extramammary Paget’s disease. J Dermatol. 2018;45:560-563. doi:10.1111/1346-8138.14247
- Yoshino K, Fujisawa Y, Kiyohara Y, et al. Usefulness of docetaxel as first-line chemotherapy for metastatic extramammary Paget’s disease. J Dermatol. 2016;43:633-637. doi:10.1111/1346-8138.13200
- Shu B, Shen XX, Chen P, et al. Primary invasive extramammary Paget disease on penoscrotum: a clinicopathological analysis of 41 cases. Hum Pathol. 2016;47:70-77. doi:10.1016/j.humpath.2015.09.005References
- Fujisawa Y, Yoshino K, Kiyohara Y, et al. The role of sentinel lymph node biopsy in the management of invasive extramammary Paget’s disease: multi-center, retrospective study of 151 patients. J Dermatol Sci. 2015;79:38-42. doi:10.1016/j.jdermsci.2015.03.014
- Dai B, Kong YY, Chang K, et al. Primary invasive carcinoma associated with penoscrotal extramammary Paget’s disease: a clinicopathological analysis of 56 cases. BJU Int. 2015;115:153-160. doi:10.1111/bju.12776
- Shiomi T, Noguchi T, Nakayama H, et al. Clinicopathological study of invasive extramammary Paget’s disease: subgroup comparison according to invasion depth. J Eur Acad Dermatol Venereol. 2013;27:589-592. doi:10.1111/j.1468-3083.2012.04489.x
- Hatta N, Morita R, Yamada M, et al. Sentinel lymph node biopsy in patients with extramammary Paget’s disease. Dermatol Surg. 2004;30:1329-1334. doi:10.1111/j.1524-4725.2004.30377.x
- Karam A, Dorigo O. Treatment outcomes in a large cohort of patients with invasive extramammary Paget’s disease. Gynecol Oncol. 2012;125:346-351. doi:10.1016/j.ygyno.2012.01.032
- Guo L, Liu X, Li H, et al. Clinicopathological features of extramammary Paget’s disease: a report of 75 cases. Article in Chinese. Zhonghua Yi Xue Za Zhi. 2015;95:1751-1754.
- Kilts TP, Long B, Glasgow AE, et al. Invasive vulvar extramammary Paget’s disease in the United States. Gynecol Oncol. 2020;157:649-655. doi:10.1016/j.ygyno.2020.03.018
- Kusatake K, Harada Y, Mizumoto K, et al. Usefulness of sentinel lymph node biopsy for the detection of metastasis in the early stage of extramammary Paget’s disease. Eur J Dermatol. 2015;25:156-161. doi:10.1684/ejd.2015.2534
- Jeong BK, Kim KR. Invasive extramammary Paget disease of the vulva with signet ring cell morphology in a patient with signet ring cell carcinoma of the stomach: report of a case. Int J Gynecol Pathol. 2018;37:147-151. doi:10.1097/PGP.0000000000000405
- Pagnanelli M, De Nardi P, Martella S, et al. Local excision of a mucinous adenocarcinoma of the anal margin (extramammary Paget’s disease) and reconstruction with a bilateral V-Y flap. Case Rep Surg. 2019;2019:9073982. doi:10.1155/2019/9073982
- Sopracordevole F, Di Giuseppe J, De Piero G, et al. Surgical treatment of Paget disease of the vulva: prognostic significance of stromal invasion and surgical margin status. J Low Genit Tract Dis. 2016;20:184-188. doi:10.1097/LGT.0000000000000191
- Evans AT, Neven P. Invasive adenocarcinoma arising in extramammary Paget’s disease of the vulva. Histopathology. 1991;18:355-360. doi:10.1111/j.1365-2559.1991.tb00857.x
- Kitano A, Izumi M, Tamura K, et al. Brain metastasis from cutaneous squamous cell carcinoma coexistent with extramammary Paget’s disease: a case report. Pathol Int. 2019;69:619-625. doi:10.1111/pin.12846
- Miracco C, Francini E, Torre P, et al. Extramammary invasive Paget’s disease and apocrine angiomatous hamartoma: an unusual association. Eur J Dermatol. 2018;28:853-855. doi:10.1684/ejd.2018.3438
- Kambayashi Y, Fujimura T, Ohuchi K, et al. Advanced invasive extramammary Paget’s disease concomitant with cecal cancer possessing rare variant of TP53 single nucleotide polymorphism. Case Rep Oncol. 2019;12:855-860. doi:10.1159/000504339
- Fujimura T, Furudate S, Kambayashi Y, et al. Potential use of bisphosphonates in invasive extramammary Paget’s disease: an immunohistochemical investigation. Clin Dev Immunol. 2013;2013:164982. doi:10.1155/2013/164982
- Kawamura H, Ogata K, Miura H, et al. Patellar metastases. A report of two cases. Int Orthop. 1993;17:57-59. doi:10.1007/BF00195227
- Damavandy AA, Terushkin V, Zitelli JA, et al. Intraoperative immunostaining for cytokeratin-7 during Mohs micrographic surgery demonstrates low local recurrence rates in extramammary Paget’s disease. Dermatol Surg. 2018;44:354-364. doi:10.1097/DSS.0000000000001355
- Morris CR, Hurst EA. Extramammary Paget disease: a review of the literature-part I: history, epidemiology, pathogenesis, presentation, histopathology, and diagnostic work-up. Dermatol Surg. 2020;46:151-158. doi:10.1097/DSS.0000000000002064
- Simonds RM, Segal RJ, Sharma A. Extramammary Paget’s disease: a review of the literature. Int J Dermatol. 2019;58:871-879. doi:10.1111/ijd.14328
- Ogata D, Kiyohara Y, Yoshikawa S, et al. Usefulness of sentinel lymph node biopsy for prognostic prediction in extramammary Paget’s disease. Eur J Dermatol. 2016;26:254-259. doi:10.1684/ejd.2016.2744
- Ohara K, Fujisawa Y, Yoshino K, et al. A proposal for a TNM staging system for extramammary Paget disease: retrospective analysis of 301 patients with invasive primary tumors. J Dermatol Sci. 2016;83:234-239. doi:10.1016/j.jdermsci.2016.06.004
- Fujisawa Y, Yoshino K, Kiyohara Y, et al. The role of sentinel lymph node biopsy in the management of invasive extramammary Paget’s disease: multi-center, retrospective study of 151 patients. J Dermatol Sci. 2015;79:38-42. doi:10.1016/j.jdermsci.2015.03.014
- Kim SJ, Thompson AK, Zubair AS, et al. Surgical treatment and outcomes of patients with extramammary Paget disease: a cohort study. Dermatol Surg. 2017;43:708-714. doi:10.1097/DSS.0000000000001051
- Wollina U. Extensive invasive extramammary Paget’s disease: surgical treatment. J Cutan Aesthet Surg. 2013;6:41-44. doi:10.4103/0974-2077.110098
- Tsutsui K, Takahashi A, Muto Y, et al. Outcomes of lymph node dissection in the treatment of extramammary Paget’s disease: a single-institution study. J Dermatol. 2020;47:512-517. doi:10.1111/1346-8138.15285
- Guercio BJ, Iyer G, Kidwai WZ, et al. Treatment of metastatic extramammary Paget disease with combination ipilimumab and nivolumab: a case report. Case Rep Oncol. 2021;14:430-438. doi:10.1159/000514345
Metastatic Primary Extramammary Paget Disease: A Case Series
Metastatic Primary Extramammary Paget Disease: A Case Series
Practice Points
- Invasive primary extramammary Paget disease has a higher risk for lymph node metastasis.
- Consider extramammary Paget disease in patients presenting with erythematous pruritic plaques in apocrine-rich areas that fail to respond to topical steroids or antifungals.
- Prompt diagnosis can expedite comprehensive malignancy work-up and multidisciplinary management, potentially impacting patient outcomes.
Severe Cutaneous Adverse Reactions in the Setting of Antineoplastic Therapy: A Single-Institution Retrospective Study
Severe Cutaneous Adverse Reactions in the Setting of Antineoplastic Therapy: A Single-Institution Retrospective Study
To the Editor:
Severe cutaneous adverse reactions (SCARs) are rare, life-threatening reactions that include acute generalized exanthematous pustulosis (AGEP), drug reaction with eosinophilia and systemic symptoms (DRESS), and Stevens-Johnson syndrome (SJS)/toxic epidermal necrolysis (TEN).1 In addition to being associated with commonly implicated medications, SCARs also may occur in the setting of antineoplastic therapy.2,3 Although antineoplastic-associated SCARs have been described, diagnosis can be difficult due to varying latency periods and atypical clinical features, such as those observed with BRAF inhibitor–related DRESS during immunotherapy.4 Severe cutaneous adverse reactions can increase morbidity and mortality in the oncologic patient population due to both the clinical sequelae from the cutaneous reaction and the potential to interrupt cancer treatment.
The aim of this study was to evaluate the clinical characteristics, outcomes, and impact on cancer treatment among patients diagnosed with a SCAR while receiving active therapy for malignancy. We conducted a retrospective chart review of electronic medical records at Yale New Haven Hospital (New Haven, Connecticut) from 2013 to 2023, identifying patients receiving antineoplastic therapy who were diagnosed with a SCAR. Cases were identified through a search of the electronic medical record performed by the joint data analytics team using the keywords DRESS, SJS, TEN, AGEP, and generalized bullous fixed drug eruption, along with spelling variations (both abbreviations and full terms), in addition to manual review by one of the authors (K.V.) of the inpatient dermatology consultation log and dermatopathology database. Only patients for whom an antineoplastic agent was identified as a high-probability culprit by the dermatology and/or oncology teams were included.
In total, 20 patients (11 female, 9 male) were identified as having an antineoplastic-associated SCAR. All patients had metastatic or advanced disease. We identified 2 (10%) cases of AGEP, 16 (80%) cases of DRESS, and 3 (15%) cases of SJS/TEN. One patient on immunotherapy had 2 distinct SCARs (AGEP, DRESS) at different time points. Table 1 describes patient and SCAR characteristics as well as impact on cancer treatment. The median (interquartile range [IQR]) latency period for AGEP was 7.5 (4-11) days. The median (IQR) latency period for 13 of the 16 (81%) DRESS cases was 14 (10-32) days. For 3 DRESS cases with a potential second-hit phenomenon in the setting of current or antecedent immunotherapy,5 the median (IQR) latency period was 122 (96-426) days for the immunotherapy drug and 28 (21-52) days for the drug culprit. The median (IQR) latency period for SJS/TEN was 23 (20-27) days.

Patients received treatment with combination systemic corticosteroids and topical corticosteroids in 13 (65%) cases, systemic corticosteroid monotherapy in 6 (30%) cases, or combination systemic corticosteroids and etanercept in 1 (5%) case. All patients experienced resolution of the SCAR and survived to hospital discharge. Most (17/20 [85%]) patients experienced interruption or discontinuation of cancer treatment. Table 2 describes the implicated antineoplastic therapies, which included chemotherapy (3 DRESS, 1 SJS/TEN), hormonal therapy (1 DRESS), immunotherapy (1 AGEP, 4 DRESS), and targeted therapy (1 AGEP, 8 DRESS, 2 SJS/TEN).

Limitations of this study include the retrospective study design, the small sample size, and the challenge of drug culprit identification in oncologic patients on multiple high-probability medications.
Though rare, SCARs can be encountered in patients on antineoplastic therapy with a wide range of drug culprits. In our cohort, SCARs occurred with various antineoplastic agents, including chemotherapy, hormonal therapy, immunotherapy, and targeted therapy. The most common antineoplastic-associated SCAR was DRESS, which had the widest latency period in the setting of a potential second-hit phenomenon with another drug culprit. Although we did not observe any cases of SJS/TEN in the immunotherapy category, it is important to consider progressive immunotherapy-related mucocutaneous eruption in the differential diagnosis. Fortunately, all patients survived to hospital discharge and experienced SCAR resolution with systemic treatment; however, most patients experienced interruption of cancer therapy, which has the potential to affect oncologic outcomes. This interruption is not uncommon, as rechallenge of an antineoplastic agent in patients with a therapy-related SCAR generally is not recommended. The awareness and prompt management of SCARs in a patient on treatment for malignancy are critical in order to minimize negative outcomes in this vulnerable patient population.
- Duong TA, Valeyrie-Allanore L, Wolkenstein P, et al. Severe cutaneous adverse reactions to drugs. Lancet. 2017;390: 1996-2011.
- Chen CB, Wu MY, Ng CY, et al. Severe cutaneous adverse reactions induced by targeted anticancer therapies and immunotherapies. Cancer Manag Res. 2018;10:1259-1273.
- Ng CY, Chen CB, Wu MY, et al. Anticancer drugs induced severe adverse cutaneous drug reactions: an updated review on the risks associated with anticancer targeted therapy or immunotherapies. J Immunol Res. 2018;2018:5376476.
- Maloney NJ, Rana J, Yang JJ, et al. Clinical features of druginduced hypersensitivity syndrome to BRAF inhibitors with and without previous immune checkpoint inhibition: a review. Support Care Cancer. 2022;30:2839-2851.
- Hammond S, Olsson-Brown A, Grice S, et al. Does immune checkpoint inhibitor therapy increase the frequency of adverse reactions to concomitant medications? Clin Exp Allergy. 2022;52:600-603.
To the Editor:
Severe cutaneous adverse reactions (SCARs) are rare, life-threatening reactions that include acute generalized exanthematous pustulosis (AGEP), drug reaction with eosinophilia and systemic symptoms (DRESS), and Stevens-Johnson syndrome (SJS)/toxic epidermal necrolysis (TEN).1 In addition to being associated with commonly implicated medications, SCARs also may occur in the setting of antineoplastic therapy.2,3 Although antineoplastic-associated SCARs have been described, diagnosis can be difficult due to varying latency periods and atypical clinical features, such as those observed with BRAF inhibitor–related DRESS during immunotherapy.4 Severe cutaneous adverse reactions can increase morbidity and mortality in the oncologic patient population due to both the clinical sequelae from the cutaneous reaction and the potential to interrupt cancer treatment.
The aim of this study was to evaluate the clinical characteristics, outcomes, and impact on cancer treatment among patients diagnosed with a SCAR while receiving active therapy for malignancy. We conducted a retrospective chart review of electronic medical records at Yale New Haven Hospital (New Haven, Connecticut) from 2013 to 2023, identifying patients receiving antineoplastic therapy who were diagnosed with a SCAR. Cases were identified through a search of the electronic medical record performed by the joint data analytics team using the keywords DRESS, SJS, TEN, AGEP, and generalized bullous fixed drug eruption, along with spelling variations (both abbreviations and full terms), in addition to manual review by one of the authors (K.V.) of the inpatient dermatology consultation log and dermatopathology database. Only patients for whom an antineoplastic agent was identified as a high-probability culprit by the dermatology and/or oncology teams were included.
In total, 20 patients (11 female, 9 male) were identified as having an antineoplastic-associated SCAR. All patients had metastatic or advanced disease. We identified 2 (10%) cases of AGEP, 16 (80%) cases of DRESS, and 3 (15%) cases of SJS/TEN. One patient on immunotherapy had 2 distinct SCARs (AGEP, DRESS) at different time points. Table 1 describes patient and SCAR characteristics as well as impact on cancer treatment. The median (interquartile range [IQR]) latency period for AGEP was 7.5 (4-11) days. The median (IQR) latency period for 13 of the 16 (81%) DRESS cases was 14 (10-32) days. For 3 DRESS cases with a potential second-hit phenomenon in the setting of current or antecedent immunotherapy,5 the median (IQR) latency period was 122 (96-426) days for the immunotherapy drug and 28 (21-52) days for the drug culprit. The median (IQR) latency period for SJS/TEN was 23 (20-27) days.

Patients received treatment with combination systemic corticosteroids and topical corticosteroids in 13 (65%) cases, systemic corticosteroid monotherapy in 6 (30%) cases, or combination systemic corticosteroids and etanercept in 1 (5%) case. All patients experienced resolution of the SCAR and survived to hospital discharge. Most (17/20 [85%]) patients experienced interruption or discontinuation of cancer treatment. Table 2 describes the implicated antineoplastic therapies, which included chemotherapy (3 DRESS, 1 SJS/TEN), hormonal therapy (1 DRESS), immunotherapy (1 AGEP, 4 DRESS), and targeted therapy (1 AGEP, 8 DRESS, 2 SJS/TEN).

Limitations of this study include the retrospective study design, the small sample size, and the challenge of drug culprit identification in oncologic patients on multiple high-probability medications.
Though rare, SCARs can be encountered in patients on antineoplastic therapy with a wide range of drug culprits. In our cohort, SCARs occurred with various antineoplastic agents, including chemotherapy, hormonal therapy, immunotherapy, and targeted therapy. The most common antineoplastic-associated SCAR was DRESS, which had the widest latency period in the setting of a potential second-hit phenomenon with another drug culprit. Although we did not observe any cases of SJS/TEN in the immunotherapy category, it is important to consider progressive immunotherapy-related mucocutaneous eruption in the differential diagnosis. Fortunately, all patients survived to hospital discharge and experienced SCAR resolution with systemic treatment; however, most patients experienced interruption of cancer therapy, which has the potential to affect oncologic outcomes. This interruption is not uncommon, as rechallenge of an antineoplastic agent in patients with a therapy-related SCAR generally is not recommended. The awareness and prompt management of SCARs in a patient on treatment for malignancy are critical in order to minimize negative outcomes in this vulnerable patient population.
To the Editor:
Severe cutaneous adverse reactions (SCARs) are rare, life-threatening reactions that include acute generalized exanthematous pustulosis (AGEP), drug reaction with eosinophilia and systemic symptoms (DRESS), and Stevens-Johnson syndrome (SJS)/toxic epidermal necrolysis (TEN).1 In addition to being associated with commonly implicated medications, SCARs also may occur in the setting of antineoplastic therapy.2,3 Although antineoplastic-associated SCARs have been described, diagnosis can be difficult due to varying latency periods and atypical clinical features, such as those observed with BRAF inhibitor–related DRESS during immunotherapy.4 Severe cutaneous adverse reactions can increase morbidity and mortality in the oncologic patient population due to both the clinical sequelae from the cutaneous reaction and the potential to interrupt cancer treatment.
The aim of this study was to evaluate the clinical characteristics, outcomes, and impact on cancer treatment among patients diagnosed with a SCAR while receiving active therapy for malignancy. We conducted a retrospective chart review of electronic medical records at Yale New Haven Hospital (New Haven, Connecticut) from 2013 to 2023, identifying patients receiving antineoplastic therapy who were diagnosed with a SCAR. Cases were identified through a search of the electronic medical record performed by the joint data analytics team using the keywords DRESS, SJS, TEN, AGEP, and generalized bullous fixed drug eruption, along with spelling variations (both abbreviations and full terms), in addition to manual review by one of the authors (K.V.) of the inpatient dermatology consultation log and dermatopathology database. Only patients for whom an antineoplastic agent was identified as a high-probability culprit by the dermatology and/or oncology teams were included.
In total, 20 patients (11 female, 9 male) were identified as having an antineoplastic-associated SCAR. All patients had metastatic or advanced disease. We identified 2 (10%) cases of AGEP, 16 (80%) cases of DRESS, and 3 (15%) cases of SJS/TEN. One patient on immunotherapy had 2 distinct SCARs (AGEP, DRESS) at different time points. Table 1 describes patient and SCAR characteristics as well as impact on cancer treatment. The median (interquartile range [IQR]) latency period for AGEP was 7.5 (4-11) days. The median (IQR) latency period for 13 of the 16 (81%) DRESS cases was 14 (10-32) days. For 3 DRESS cases with a potential second-hit phenomenon in the setting of current or antecedent immunotherapy,5 the median (IQR) latency period was 122 (96-426) days for the immunotherapy drug and 28 (21-52) days for the drug culprit. The median (IQR) latency period for SJS/TEN was 23 (20-27) days.

Patients received treatment with combination systemic corticosteroids and topical corticosteroids in 13 (65%) cases, systemic corticosteroid monotherapy in 6 (30%) cases, or combination systemic corticosteroids and etanercept in 1 (5%) case. All patients experienced resolution of the SCAR and survived to hospital discharge. Most (17/20 [85%]) patients experienced interruption or discontinuation of cancer treatment. Table 2 describes the implicated antineoplastic therapies, which included chemotherapy (3 DRESS, 1 SJS/TEN), hormonal therapy (1 DRESS), immunotherapy (1 AGEP, 4 DRESS), and targeted therapy (1 AGEP, 8 DRESS, 2 SJS/TEN).

Limitations of this study include the retrospective study design, the small sample size, and the challenge of drug culprit identification in oncologic patients on multiple high-probability medications.
Though rare, SCARs can be encountered in patients on antineoplastic therapy with a wide range of drug culprits. In our cohort, SCARs occurred with various antineoplastic agents, including chemotherapy, hormonal therapy, immunotherapy, and targeted therapy. The most common antineoplastic-associated SCAR was DRESS, which had the widest latency period in the setting of a potential second-hit phenomenon with another drug culprit. Although we did not observe any cases of SJS/TEN in the immunotherapy category, it is important to consider progressive immunotherapy-related mucocutaneous eruption in the differential diagnosis. Fortunately, all patients survived to hospital discharge and experienced SCAR resolution with systemic treatment; however, most patients experienced interruption of cancer therapy, which has the potential to affect oncologic outcomes. This interruption is not uncommon, as rechallenge of an antineoplastic agent in patients with a therapy-related SCAR generally is not recommended. The awareness and prompt management of SCARs in a patient on treatment for malignancy are critical in order to minimize negative outcomes in this vulnerable patient population.
- Duong TA, Valeyrie-Allanore L, Wolkenstein P, et al. Severe cutaneous adverse reactions to drugs. Lancet. 2017;390: 1996-2011.
- Chen CB, Wu MY, Ng CY, et al. Severe cutaneous adverse reactions induced by targeted anticancer therapies and immunotherapies. Cancer Manag Res. 2018;10:1259-1273.
- Ng CY, Chen CB, Wu MY, et al. Anticancer drugs induced severe adverse cutaneous drug reactions: an updated review on the risks associated with anticancer targeted therapy or immunotherapies. J Immunol Res. 2018;2018:5376476.
- Maloney NJ, Rana J, Yang JJ, et al. Clinical features of druginduced hypersensitivity syndrome to BRAF inhibitors with and without previous immune checkpoint inhibition: a review. Support Care Cancer. 2022;30:2839-2851.
- Hammond S, Olsson-Brown A, Grice S, et al. Does immune checkpoint inhibitor therapy increase the frequency of adverse reactions to concomitant medications? Clin Exp Allergy. 2022;52:600-603.
- Duong TA, Valeyrie-Allanore L, Wolkenstein P, et al. Severe cutaneous adverse reactions to drugs. Lancet. 2017;390: 1996-2011.
- Chen CB, Wu MY, Ng CY, et al. Severe cutaneous adverse reactions induced by targeted anticancer therapies and immunotherapies. Cancer Manag Res. 2018;10:1259-1273.
- Ng CY, Chen CB, Wu MY, et al. Anticancer drugs induced severe adverse cutaneous drug reactions: an updated review on the risks associated with anticancer targeted therapy or immunotherapies. J Immunol Res. 2018;2018:5376476.
- Maloney NJ, Rana J, Yang JJ, et al. Clinical features of druginduced hypersensitivity syndrome to BRAF inhibitors with and without previous immune checkpoint inhibition: a review. Support Care Cancer. 2022;30:2839-2851.
- Hammond S, Olsson-Brown A, Grice S, et al. Does immune checkpoint inhibitor therapy increase the frequency of adverse reactions to concomitant medications? Clin Exp Allergy. 2022;52:600-603.
Severe Cutaneous Adverse Reactions in the Setting of Antineoplastic Therapy: A Single-Institution Retrospective Study
Severe Cutaneous Adverse Reactions in the Setting of Antineoplastic Therapy: A Single-Institution Retrospective Study
Practice Points
- Clinicians should be aware of the occurrence of severe cutaneous adverse reactions (SCARs) in patients on antineoplastic therapy to prevent delays in treatment and improve patient outcomes.
- Rapid initiation of treatment can be effective in resolving SCARs and ensuring full recovery.
- Close coordination between dermatology and oncology teams is crucial to manage SCARs while minimizing cancer treatment interruptions.