User login
Adverse Events Associated With At-Home Microcurrent Facial Devices
Adverse Events Associated With At-Home Microcurrent Facial Devices
To the Editor:
At-home microcurrent facial devices have gained rapid popularity for cosmetic rejuvenation, promising improvements in skin tone, contour, and collagen production.¹ In particular, the post–COVID-19 era has seen a surge in at-home beauty practices driven by social media influence, with the global microcurrent facial market estimated at $372.9 million in 2022 and projected to grow at a compound annual growth rate of 7.3% through 2030.1 Microcurrent devices deliver low-level electrical currents to the skin and underlying muscles. Given the limited exploration of the long-term safety, we aimed to collate existing data and identify trends in reports of adverse events (AEs) associated with these microcurrent devices.
On April 15, 2025, the US Food and Drug Administration’s Manufacturer and User Facility Device Experience (MAUDE) database was queried for medical device reports from January 1, 2013, through March 31, 2025, using product names and keywords including NuFACE, TheraFace, FOREO, and microcurrent device. Search terms were limited to brands for which complaint data existed in the MAUDE database at the time of query. To ensure accuracy, reports were manually reviewed to eliminate duplicates and irrelevant entries.
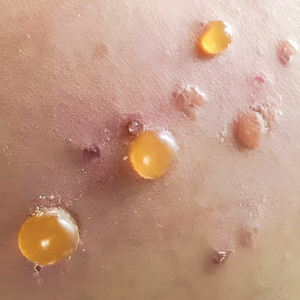
A total of 28 unique AE reports associated with at-home microcurrent devices were identified (eTable). The majority involved NuFACE devices (ie, NuFACE Trinity, NuFACE Mini, and NuFACE Trinity+)(NuFACE)(n=25), followed by the TheraFace PRO (Therabody, Inc)(n=2) and the FOREO BEAR (FOREO)(n=1). The most frequently documented AEs associated with the NuFACE devices included arrhythmia (7/25 [28%]), pain (6/25 [24%]), dizziness (4/25 [16%]), headache (4/25 [16%]), and inflammation (4/25 [16%]). There was 1 (4%) case of retinal detachment. The TheraFace PRO was associated with device overheating (2/2 [100%]), and the FOREO BEAR was associated with facial deformity/disfigurement (1/1 [100%]).

While microcurrent therapy is widely marketed to consumers through social media influencers and at-home beauty platforms,1 randomized controlled trials (RCTs) evaluating AEs related to use of this technology are lacking, possibly due to nonstringent regulation of nonprescription cosmetic devices.² Contrary to our findings, RCTs of microcurrent devices have reported minimal or no AEs; for instance, an RCT evaluating 56 participants treated 5 times weekly for 12 weeks with a microcurrent device that was not included in our analysis reported only mild erythema in all experimental group participants.2 In another RCT of 30 participants, 15 of whom were treated with a microcurrent device and 15 with placebo for 30 minutes once daily over a period of 10 days, no AEs were reported.3 A cohort analysis of 34 patients also provided preliminary evidence supporting the use of microcurrent therapy for chronic back and neck pain, beyond its cosmetic applications.4 Despite the lack of reported AEs in the literature, there is a notable absence of large-scale, rigorous studies on this topic.
Our analysis was subject to the limitations of the MAUDE database, in which reports of severe AEs are more likely to be reported than transient ones. Additionally, the small sample size and lack of a known denominator make it difficult to compare frequencies of AEs among different microcurrent tools. The products chosen for this study were the select few that reported complaint data, but there is a large existing market of devices that may be associated with AEs that have yet to be reported, potentially because of their novelty.
Our findings suggest that, despite their over-the-counter availability, microcurrent facial devices may carry major risks—particularly in at-home settings. While short-term studies have highlighted potential benefits, the small sample sizes and limited follow-up make it difficult to comprehensively characterize long-term safety risks. Among available studies on microcurrent beauty treatments, the longest follow-up was only 12 weeks.2 Our findings support the need for further large-scale and longitudinal studies to evaluate both the efficacy and safety of at-home microcurrent therapy, especially with increasing consumer interest. The diversity of the products available adds to the challenge of broad safety guidelines, in addition to the lack of long-term clinical studies.
- Microcurrent Facial Market Size, Growth & Trends Report 2030. Grand View Research. Published 2023. Accessed March 3, 2026. https://www.grandviewresearch.com/industry-analysis/microcurrent-facial-market-report
- Bu P, Duan R, Luo J, et al. Development of home beauty devices for facial rejuvenation: establishment of efficacy evaluation system. Clin Cosmet Investig Dermatol. 2024;17:553-563.
- Jain S, Arora M. Effect of microcurrent facial muscle toning on fine wrinkles & firmness of face. IAMR J Physiother. 2012;1:13-19.
- Armstrong K, Gokal R, Chevalier A, et al. Microcurrent point stimulation applied to lower back acupuncture points for the treatment of nonspecific neck pain. J Altern Complement Med. 2017;23:295-299.
To the Editor:
At-home microcurrent facial devices have gained rapid popularity for cosmetic rejuvenation, promising improvements in skin tone, contour, and collagen production.¹ In particular, the post–COVID-19 era has seen a surge in at-home beauty practices driven by social media influence, with the global microcurrent facial market estimated at $372.9 million in 2022 and projected to grow at a compound annual growth rate of 7.3% through 2030.1 Microcurrent devices deliver low-level electrical currents to the skin and underlying muscles. Given the limited exploration of the long-term safety, we aimed to collate existing data and identify trends in reports of adverse events (AEs) associated with these microcurrent devices.
On April 15, 2025, the US Food and Drug Administration’s Manufacturer and User Facility Device Experience (MAUDE) database was queried for medical device reports from January 1, 2013, through March 31, 2025, using product names and keywords including NuFACE, TheraFace, FOREO, and microcurrent device. Search terms were limited to brands for which complaint data existed in the MAUDE database at the time of query. To ensure accuracy, reports were manually reviewed to eliminate duplicates and irrelevant entries.
A total of 28 unique AE reports associated with at-home microcurrent devices were identified (eTable). The majority involved NuFACE devices (ie, NuFACE Trinity, NuFACE Mini, and NuFACE Trinity+)(NuFACE)(n=25), followed by the TheraFace PRO (Therabody, Inc)(n=2) and the FOREO BEAR (FOREO)(n=1). The most frequently documented AEs associated with the NuFACE devices included arrhythmia (7/25 [28%]), pain (6/25 [24%]), dizziness (4/25 [16%]), headache (4/25 [16%]), and inflammation (4/25 [16%]). There was 1 (4%) case of retinal detachment. The TheraFace PRO was associated with device overheating (2/2 [100%]), and the FOREO BEAR was associated with facial deformity/disfigurement (1/1 [100%]).

While microcurrent therapy is widely marketed to consumers through social media influencers and at-home beauty platforms,1 randomized controlled trials (RCTs) evaluating AEs related to use of this technology are lacking, possibly due to nonstringent regulation of nonprescription cosmetic devices.² Contrary to our findings, RCTs of microcurrent devices have reported minimal or no AEs; for instance, an RCT evaluating 56 participants treated 5 times weekly for 12 weeks with a microcurrent device that was not included in our analysis reported only mild erythema in all experimental group participants.2 In another RCT of 30 participants, 15 of whom were treated with a microcurrent device and 15 with placebo for 30 minutes once daily over a period of 10 days, no AEs were reported.3 A cohort analysis of 34 patients also provided preliminary evidence supporting the use of microcurrent therapy for chronic back and neck pain, beyond its cosmetic applications.4 Despite the lack of reported AEs in the literature, there is a notable absence of large-scale, rigorous studies on this topic.
Our analysis was subject to the limitations of the MAUDE database, in which reports of severe AEs are more likely to be reported than transient ones. Additionally, the small sample size and lack of a known denominator make it difficult to compare frequencies of AEs among different microcurrent tools. The products chosen for this study were the select few that reported complaint data, but there is a large existing market of devices that may be associated with AEs that have yet to be reported, potentially because of their novelty.
Our findings suggest that, despite their over-the-counter availability, microcurrent facial devices may carry major risks—particularly in at-home settings. While short-term studies have highlighted potential benefits, the small sample sizes and limited follow-up make it difficult to comprehensively characterize long-term safety risks. Among available studies on microcurrent beauty treatments, the longest follow-up was only 12 weeks.2 Our findings support the need for further large-scale and longitudinal studies to evaluate both the efficacy and safety of at-home microcurrent therapy, especially with increasing consumer interest. The diversity of the products available adds to the challenge of broad safety guidelines, in addition to the lack of long-term clinical studies.
To the Editor:
At-home microcurrent facial devices have gained rapid popularity for cosmetic rejuvenation, promising improvements in skin tone, contour, and collagen production.¹ In particular, the post–COVID-19 era has seen a surge in at-home beauty practices driven by social media influence, with the global microcurrent facial market estimated at $372.9 million in 2022 and projected to grow at a compound annual growth rate of 7.3% through 2030.1 Microcurrent devices deliver low-level electrical currents to the skin and underlying muscles. Given the limited exploration of the long-term safety, we aimed to collate existing data and identify trends in reports of adverse events (AEs) associated with these microcurrent devices.
On April 15, 2025, the US Food and Drug Administration’s Manufacturer and User Facility Device Experience (MAUDE) database was queried for medical device reports from January 1, 2013, through March 31, 2025, using product names and keywords including NuFACE, TheraFace, FOREO, and microcurrent device. Search terms were limited to brands for which complaint data existed in the MAUDE database at the time of query. To ensure accuracy, reports were manually reviewed to eliminate duplicates and irrelevant entries.
A total of 28 unique AE reports associated with at-home microcurrent devices were identified (eTable). The majority involved NuFACE devices (ie, NuFACE Trinity, NuFACE Mini, and NuFACE Trinity+)(NuFACE)(n=25), followed by the TheraFace PRO (Therabody, Inc)(n=2) and the FOREO BEAR (FOREO)(n=1). The most frequently documented AEs associated with the NuFACE devices included arrhythmia (7/25 [28%]), pain (6/25 [24%]), dizziness (4/25 [16%]), headache (4/25 [16%]), and inflammation (4/25 [16%]). There was 1 (4%) case of retinal detachment. The TheraFace PRO was associated with device overheating (2/2 [100%]), and the FOREO BEAR was associated with facial deformity/disfigurement (1/1 [100%]).

While microcurrent therapy is widely marketed to consumers through social media influencers and at-home beauty platforms,1 randomized controlled trials (RCTs) evaluating AEs related to use of this technology are lacking, possibly due to nonstringent regulation of nonprescription cosmetic devices.² Contrary to our findings, RCTs of microcurrent devices have reported minimal or no AEs; for instance, an RCT evaluating 56 participants treated 5 times weekly for 12 weeks with a microcurrent device that was not included in our analysis reported only mild erythema in all experimental group participants.2 In another RCT of 30 participants, 15 of whom were treated with a microcurrent device and 15 with placebo for 30 minutes once daily over a period of 10 days, no AEs were reported.3 A cohort analysis of 34 patients also provided preliminary evidence supporting the use of microcurrent therapy for chronic back and neck pain, beyond its cosmetic applications.4 Despite the lack of reported AEs in the literature, there is a notable absence of large-scale, rigorous studies on this topic.
Our analysis was subject to the limitations of the MAUDE database, in which reports of severe AEs are more likely to be reported than transient ones. Additionally, the small sample size and lack of a known denominator make it difficult to compare frequencies of AEs among different microcurrent tools. The products chosen for this study were the select few that reported complaint data, but there is a large existing market of devices that may be associated with AEs that have yet to be reported, potentially because of their novelty.
Our findings suggest that, despite their over-the-counter availability, microcurrent facial devices may carry major risks—particularly in at-home settings. While short-term studies have highlighted potential benefits, the small sample sizes and limited follow-up make it difficult to comprehensively characterize long-term safety risks. Among available studies on microcurrent beauty treatments, the longest follow-up was only 12 weeks.2 Our findings support the need for further large-scale and longitudinal studies to evaluate both the efficacy and safety of at-home microcurrent therapy, especially with increasing consumer interest. The diversity of the products available adds to the challenge of broad safety guidelines, in addition to the lack of long-term clinical studies.
- Microcurrent Facial Market Size, Growth & Trends Report 2030. Grand View Research. Published 2023. Accessed March 3, 2026. https://www.grandviewresearch.com/industry-analysis/microcurrent-facial-market-report
- Bu P, Duan R, Luo J, et al. Development of home beauty devices for facial rejuvenation: establishment of efficacy evaluation system. Clin Cosmet Investig Dermatol. 2024;17:553-563.
- Jain S, Arora M. Effect of microcurrent facial muscle toning on fine wrinkles & firmness of face. IAMR J Physiother. 2012;1:13-19.
- Armstrong K, Gokal R, Chevalier A, et al. Microcurrent point stimulation applied to lower back acupuncture points for the treatment of nonspecific neck pain. J Altern Complement Med. 2017;23:295-299.
- Microcurrent Facial Market Size, Growth & Trends Report 2030. Grand View Research. Published 2023. Accessed March 3, 2026. https://www.grandviewresearch.com/industry-analysis/microcurrent-facial-market-report
- Bu P, Duan R, Luo J, et al. Development of home beauty devices for facial rejuvenation: establishment of efficacy evaluation system. Clin Cosmet Investig Dermatol. 2024;17:553-563.
- Jain S, Arora M. Effect of microcurrent facial muscle toning on fine wrinkles & firmness of face. IAMR J Physiother. 2012;1:13-19.
- Armstrong K, Gokal R, Chevalier A, et al. Microcurrent point stimulation applied to lower back acupuncture points for the treatment of nonspecific neck pain. J Altern Complement Med. 2017;23:295-299.
Adverse Events Associated With At-Home Microcurrent Facial Devices
Adverse Events Associated With At-Home Microcurrent Facial Devices
PRACTICE POINTS
- At-home microcurrent facial devices have been associated with serious adverse events, including arrhythmia, pain, dizziness, and retinal detachment, based on US Food and Drug Administration Manufacturer and User Facility Device Experience database reports, underscoring the importance of counseling patients about potential risks prior to use.
- Existing randomized controlled trials of microcurrent devices are limited by small sample sizes and short follow-up periods (maximum 12 weeks), making it difficult to characterize the long-term safety profile of these increasingly popular devices.
- Dermatologists should be aware that the largely unregulated at-home microcurrent device market lacks robust, large-scale safety data. Patients, particularly those with cardiac conditions or implanted electrical devices, should be advised to consult a physician before use.
Optimizing Patch Testing in Clinical Practice: Insights From Amber Reck Atwater, MD
Optimizing Patch Testing in Clinical Practice: Insights From Amber Reck Atwater, MD
What evidence exists on the impact of systemic immunosuppressants, biologics, and small-molecule inhibitors on patch test sensitivity and specificity?
DR. ATWATER: Guidance on this topic recently was published by the North American Contact Dermatitis Group (NACDG) in the Journal of the American Academy of Dermatology in June 2025. The article outlined expert recommendations on whether systemic immunosuppressants, biologics, and small-molecule inhibitors should be held before patch testing, how long they should be withheld, and the maximum recommended doses that can be used during testing.
How can dermatologists perform patch testing or use alternative diagnostic strategies when systemic therapy cannot safely be withheld?
DR. ATWATER: When systemic therapy cannot safely be withheld and patch testing is needed for diagnostic purposes, I typically proceed with the understanding that there is risk of false-negative reactions. If the patient has dermatitis on systemic therapy, it suggests that an allergic response on patch testing is also possible. I generally follow the NACDG guidelines mentioned above, and I hold systemic medications during the week of testing, when possible. If the patient has diffuse dermatitis on systemic therapy and their skin is not clear enough to proceed, I prescribe a prednisone taper and patch test on 10 mg per day for the entire week of testing. In patients taking systemic medications, I typically consider doubtful (+/−) patch test reactions to be the equivalent of a positive (1+) reaction.
One alternative diagnostic strategy is to create a safe list that avoids common allergens and have the patient use only products on this list. If their skin clears with avoidance, it suggests that they may have a contact allergy, and you can proceed with patch testing.
In patients with a convincing history of contact dermatitis but a negative patch test, what are the most common causes of false negatives, and how do you distinguish those from true negatives?
DR. ATWATER: In this setting, the most common cause of a false-negative patch test is not testing the correct allergens. This may occur when too few allergens are tested or when relevant allergens are not tested. Other potential causes of false negatives are incorrect timing of allergen exposure and readings, inadequate allergen adherence, expired allergens, and testing with the incorrect vehicle or concentration of allergen. Some immunosuppressant medications also can cause a false-negative patch test. The only way to distinguish false negatives from true negatives is to be aware of these potential pitfalls and continuously work to avoid errors whenever possible.
What technical and practical factors most influence false negatives/positives, and what steps do you recommend to standardize and improve test yield?
DR. ATWATER: Not testing the correct allergens is a potential pitfall in patch testing. For example, when comparing the 35 allergens in the T.R.U.E. Test (thin layer rapid-use epicutaneous test) to the 80 allergens tested by the NACDG in 2021 to 2022, up to 48% of NACDG allergens are missed when testing with only the T.R.U.E. Test. This argues for comprehensive patch testing and testing of at least 80 to 90 allergens whenever possible. Another example is failure to test allergens relevant to occupational exposures, such as in the case of a hairdresser or nail technician. When patches are not applied for the correct period (48 hours) and the final reading isn’t completed in the recommended timeframe (96 to 168 hours), there is increased risk for false negatives and positives. Both false negatives and positives can occur if you complete your final reading too early, whereas false negatives may be more likely if you complete your final reading late. Poor allergen adherence, which can be caused by hair, sweat, poor tape application, water, and exercise, also can result in false negatives. Allergen concentration that is too low to elicit a reaction, as well as too little allergen placed into the chamber, also could increase the risk of false negatives. Too much allergen in the chamber or too high a concentration of the allergen can result in false positives.
Topical medications applied to the patch test site prior to or during patch testing, as well as phototherapy, sun exposure, intramuscular triamcinolone, immunosuppressants, biologics, and small-molecule inhibitors can suppress the immune response to allergen exposure.
To identify true positive patch test reactions, use side lighting and palpate the skin. Be aware of the appearance of irritant reactions, patch test reaction variants such as follicular reactions, and the poral reaction, which can be seen with cobalt. Strong knowledge of how to read patch test reactions will decrease your risk of false-positive and -negative reactions. Training and protocols are vital for standardization and accurate patch testing. We train our staff on the technicalities of patch testing and utilize patch test orders and checklists in our office. We take photos to confirm application sites and visually track reactions between visits. We also provide verbal and written patch test care instructions for our patients and reinforce instructions at each clinic visit.
What are your top practical tips for dermatologists to maximize diagnostic accuracy and patient safety?
DR. ATWATER: My first tip is to develop patch test protocols that are followed by staff and physicians—every time—for every patient. My second tip is to make sure you understand and are comfortable with the patch test process. There are several great patch test resources that can help, including Introduction to Patch Testing, a recently developed CME module in the AAD Learning Center (https://learning.aad.org/Listing/Introduction-to-Patch- Testing-20366). There also are patch test training courses and other resources offered by the American Contact Dermatitis Society (https://www.contactderm.org/).
What single guideline change would most improve patch testing?
DR. ATWATER: The single guideline change that would most improve the practice of patch testing is removal of payer limitations on the number of patches that can be applied per day. For many payers in the United States, this limit is 80 to 90 allergens, and it is sometimes lower. Limits on the number of allergens that can be applied per day may result in false-negative patch testing (when limits are applied), patient inconvenience (if testing is completed over 2 different application days), and insufficient reimbursement (if more allergens are tested than the limit allows).
What evidence exists on the impact of systemic immunosuppressants, biologics, and small-molecule inhibitors on patch test sensitivity and specificity?
DR. ATWATER: Guidance on this topic recently was published by the North American Contact Dermatitis Group (NACDG) in the Journal of the American Academy of Dermatology in June 2025. The article outlined expert recommendations on whether systemic immunosuppressants, biologics, and small-molecule inhibitors should be held before patch testing, how long they should be withheld, and the maximum recommended doses that can be used during testing.
How can dermatologists perform patch testing or use alternative diagnostic strategies when systemic therapy cannot safely be withheld?
DR. ATWATER: When systemic therapy cannot safely be withheld and patch testing is needed for diagnostic purposes, I typically proceed with the understanding that there is risk of false-negative reactions. If the patient has dermatitis on systemic therapy, it suggests that an allergic response on patch testing is also possible. I generally follow the NACDG guidelines mentioned above, and I hold systemic medications during the week of testing, when possible. If the patient has diffuse dermatitis on systemic therapy and their skin is not clear enough to proceed, I prescribe a prednisone taper and patch test on 10 mg per day for the entire week of testing. In patients taking systemic medications, I typically consider doubtful (+/−) patch test reactions to be the equivalent of a positive (1+) reaction.
One alternative diagnostic strategy is to create a safe list that avoids common allergens and have the patient use only products on this list. If their skin clears with avoidance, it suggests that they may have a contact allergy, and you can proceed with patch testing.
In patients with a convincing history of contact dermatitis but a negative patch test, what are the most common causes of false negatives, and how do you distinguish those from true negatives?
DR. ATWATER: In this setting, the most common cause of a false-negative patch test is not testing the correct allergens. This may occur when too few allergens are tested or when relevant allergens are not tested. Other potential causes of false negatives are incorrect timing of allergen exposure and readings, inadequate allergen adherence, expired allergens, and testing with the incorrect vehicle or concentration of allergen. Some immunosuppressant medications also can cause a false-negative patch test. The only way to distinguish false negatives from true negatives is to be aware of these potential pitfalls and continuously work to avoid errors whenever possible.
What technical and practical factors most influence false negatives/positives, and what steps do you recommend to standardize and improve test yield?
DR. ATWATER: Not testing the correct allergens is a potential pitfall in patch testing. For example, when comparing the 35 allergens in the T.R.U.E. Test (thin layer rapid-use epicutaneous test) to the 80 allergens tested by the NACDG in 2021 to 2022, up to 48% of NACDG allergens are missed when testing with only the T.R.U.E. Test. This argues for comprehensive patch testing and testing of at least 80 to 90 allergens whenever possible. Another example is failure to test allergens relevant to occupational exposures, such as in the case of a hairdresser or nail technician. When patches are not applied for the correct period (48 hours) and the final reading isn’t completed in the recommended timeframe (96 to 168 hours), there is increased risk for false negatives and positives. Both false negatives and positives can occur if you complete your final reading too early, whereas false negatives may be more likely if you complete your final reading late. Poor allergen adherence, which can be caused by hair, sweat, poor tape application, water, and exercise, also can result in false negatives. Allergen concentration that is too low to elicit a reaction, as well as too little allergen placed into the chamber, also could increase the risk of false negatives. Too much allergen in the chamber or too high a concentration of the allergen can result in false positives.
Topical medications applied to the patch test site prior to or during patch testing, as well as phototherapy, sun exposure, intramuscular triamcinolone, immunosuppressants, biologics, and small-molecule inhibitors can suppress the immune response to allergen exposure.
To identify true positive patch test reactions, use side lighting and palpate the skin. Be aware of the appearance of irritant reactions, patch test reaction variants such as follicular reactions, and the poral reaction, which can be seen with cobalt. Strong knowledge of how to read patch test reactions will decrease your risk of false-positive and -negative reactions. Training and protocols are vital for standardization and accurate patch testing. We train our staff on the technicalities of patch testing and utilize patch test orders and checklists in our office. We take photos to confirm application sites and visually track reactions between visits. We also provide verbal and written patch test care instructions for our patients and reinforce instructions at each clinic visit.
What are your top practical tips for dermatologists to maximize diagnostic accuracy and patient safety?
DR. ATWATER: My first tip is to develop patch test protocols that are followed by staff and physicians—every time—for every patient. My second tip is to make sure you understand and are comfortable with the patch test process. There are several great patch test resources that can help, including Introduction to Patch Testing, a recently developed CME module in the AAD Learning Center (https://learning.aad.org/Listing/Introduction-to-Patch- Testing-20366). There also are patch test training courses and other resources offered by the American Contact Dermatitis Society (https://www.contactderm.org/).
What single guideline change would most improve patch testing?
DR. ATWATER: The single guideline change that would most improve the practice of patch testing is removal of payer limitations on the number of patches that can be applied per day. For many payers in the United States, this limit is 80 to 90 allergens, and it is sometimes lower. Limits on the number of allergens that can be applied per day may result in false-negative patch testing (when limits are applied), patient inconvenience (if testing is completed over 2 different application days), and insufficient reimbursement (if more allergens are tested than the limit allows).
What evidence exists on the impact of systemic immunosuppressants, biologics, and small-molecule inhibitors on patch test sensitivity and specificity?
DR. ATWATER: Guidance on this topic recently was published by the North American Contact Dermatitis Group (NACDG) in the Journal of the American Academy of Dermatology in June 2025. The article outlined expert recommendations on whether systemic immunosuppressants, biologics, and small-molecule inhibitors should be held before patch testing, how long they should be withheld, and the maximum recommended doses that can be used during testing.
How can dermatologists perform patch testing or use alternative diagnostic strategies when systemic therapy cannot safely be withheld?
DR. ATWATER: When systemic therapy cannot safely be withheld and patch testing is needed for diagnostic purposes, I typically proceed with the understanding that there is risk of false-negative reactions. If the patient has dermatitis on systemic therapy, it suggests that an allergic response on patch testing is also possible. I generally follow the NACDG guidelines mentioned above, and I hold systemic medications during the week of testing, when possible. If the patient has diffuse dermatitis on systemic therapy and their skin is not clear enough to proceed, I prescribe a prednisone taper and patch test on 10 mg per day for the entire week of testing. In patients taking systemic medications, I typically consider doubtful (+/−) patch test reactions to be the equivalent of a positive (1+) reaction.
One alternative diagnostic strategy is to create a safe list that avoids common allergens and have the patient use only products on this list. If their skin clears with avoidance, it suggests that they may have a contact allergy, and you can proceed with patch testing.
In patients with a convincing history of contact dermatitis but a negative patch test, what are the most common causes of false negatives, and how do you distinguish those from true negatives?
DR. ATWATER: In this setting, the most common cause of a false-negative patch test is not testing the correct allergens. This may occur when too few allergens are tested or when relevant allergens are not tested. Other potential causes of false negatives are incorrect timing of allergen exposure and readings, inadequate allergen adherence, expired allergens, and testing with the incorrect vehicle or concentration of allergen. Some immunosuppressant medications also can cause a false-negative patch test. The only way to distinguish false negatives from true negatives is to be aware of these potential pitfalls and continuously work to avoid errors whenever possible.
What technical and practical factors most influence false negatives/positives, and what steps do you recommend to standardize and improve test yield?
DR. ATWATER: Not testing the correct allergens is a potential pitfall in patch testing. For example, when comparing the 35 allergens in the T.R.U.E. Test (thin layer rapid-use epicutaneous test) to the 80 allergens tested by the NACDG in 2021 to 2022, up to 48% of NACDG allergens are missed when testing with only the T.R.U.E. Test. This argues for comprehensive patch testing and testing of at least 80 to 90 allergens whenever possible. Another example is failure to test allergens relevant to occupational exposures, such as in the case of a hairdresser or nail technician. When patches are not applied for the correct period (48 hours) and the final reading isn’t completed in the recommended timeframe (96 to 168 hours), there is increased risk for false negatives and positives. Both false negatives and positives can occur if you complete your final reading too early, whereas false negatives may be more likely if you complete your final reading late. Poor allergen adherence, which can be caused by hair, sweat, poor tape application, water, and exercise, also can result in false negatives. Allergen concentration that is too low to elicit a reaction, as well as too little allergen placed into the chamber, also could increase the risk of false negatives. Too much allergen in the chamber or too high a concentration of the allergen can result in false positives.
Topical medications applied to the patch test site prior to or during patch testing, as well as phototherapy, sun exposure, intramuscular triamcinolone, immunosuppressants, biologics, and small-molecule inhibitors can suppress the immune response to allergen exposure.
To identify true positive patch test reactions, use side lighting and palpate the skin. Be aware of the appearance of irritant reactions, patch test reaction variants such as follicular reactions, and the poral reaction, which can be seen with cobalt. Strong knowledge of how to read patch test reactions will decrease your risk of false-positive and -negative reactions. Training and protocols are vital for standardization and accurate patch testing. We train our staff on the technicalities of patch testing and utilize patch test orders and checklists in our office. We take photos to confirm application sites and visually track reactions between visits. We also provide verbal and written patch test care instructions for our patients and reinforce instructions at each clinic visit.
What are your top practical tips for dermatologists to maximize diagnostic accuracy and patient safety?
DR. ATWATER: My first tip is to develop patch test protocols that are followed by staff and physicians—every time—for every patient. My second tip is to make sure you understand and are comfortable with the patch test process. There are several great patch test resources that can help, including Introduction to Patch Testing, a recently developed CME module in the AAD Learning Center (https://learning.aad.org/Listing/Introduction-to-Patch- Testing-20366). There also are patch test training courses and other resources offered by the American Contact Dermatitis Society (https://www.contactderm.org/).
What single guideline change would most improve patch testing?
DR. ATWATER: The single guideline change that would most improve the practice of patch testing is removal of payer limitations on the number of patches that can be applied per day. For many payers in the United States, this limit is 80 to 90 allergens, and it is sometimes lower. Limits on the number of allergens that can be applied per day may result in false-negative patch testing (when limits are applied), patient inconvenience (if testing is completed over 2 different application days), and insufficient reimbursement (if more allergens are tested than the limit allows).
Optimizing Patch Testing in Clinical Practice: Insights From Amber Reck Atwater, MD
Optimizing Patch Testing in Clinical Practice: Insights From Amber Reck Atwater, MD
A Hybrid Suture Technique: Suture Modification With Dental Roll Insertion
A Hybrid Suture Technique: Suture Modification With Dental Roll Insertion
Practice Gap
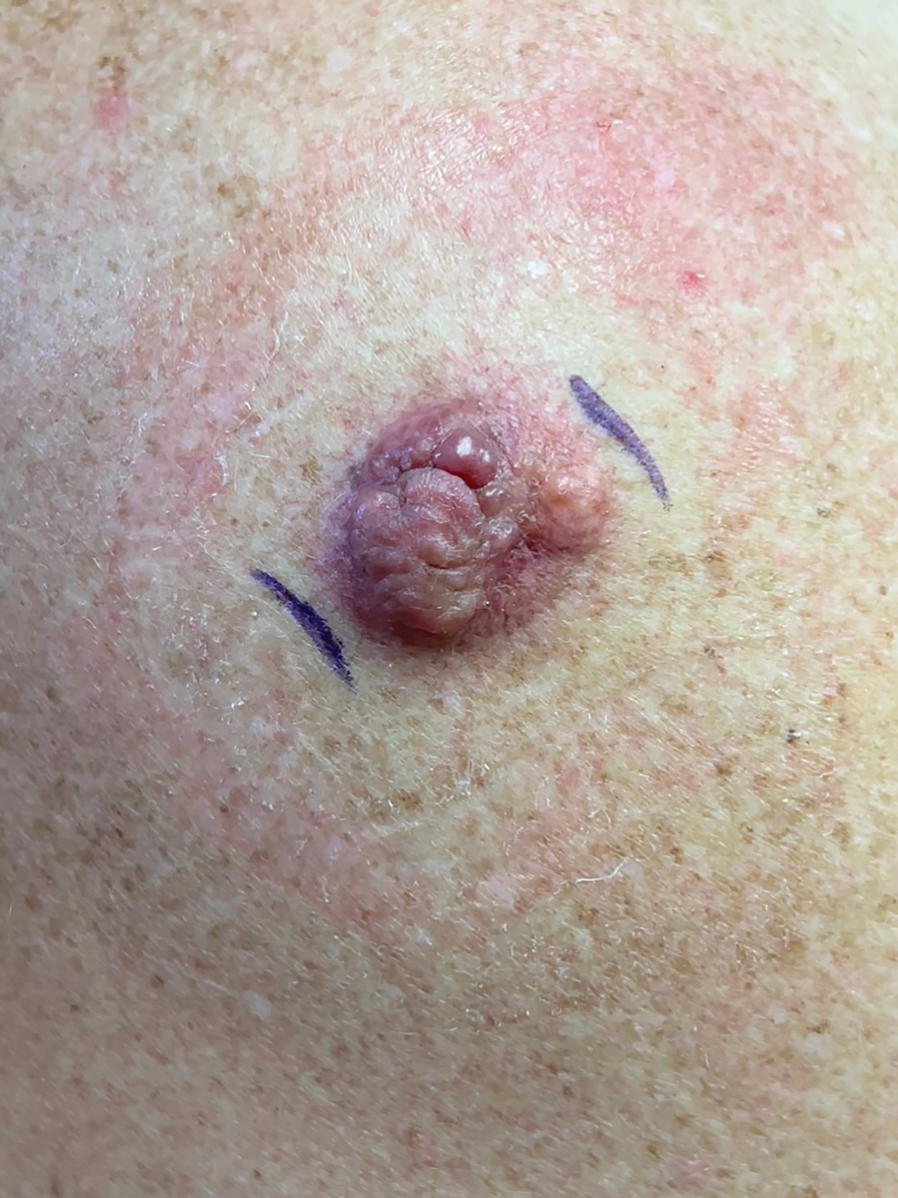
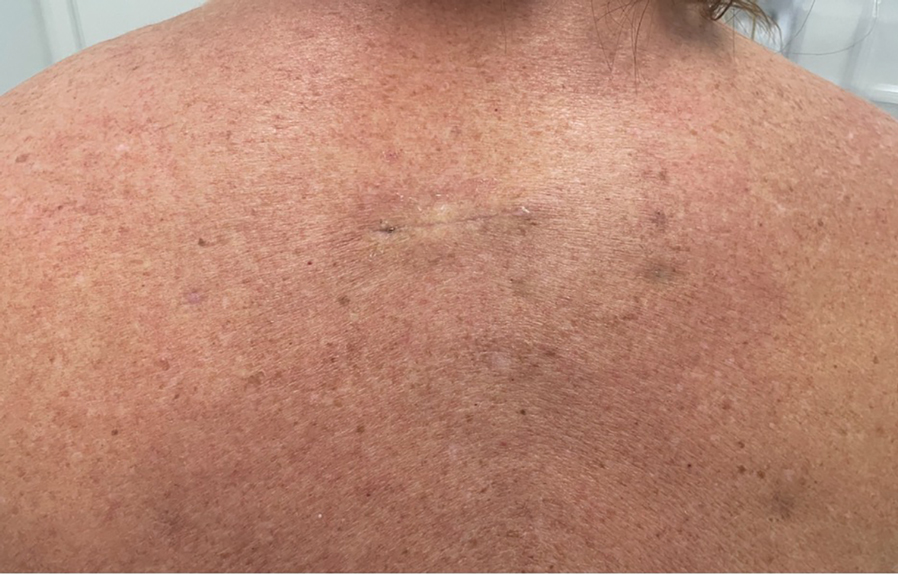
If not cared for properly, epidermal suture knots can cause discomfort, skin irritation, and an increased risk for infection. There is limited guidance on a simple adaptable method to reduce tram-track marks from epidermal sutures exerting pressure on the epidermis while still facilitating healing in dermatologic procedures such as excision of cysts or lipomas. We present a hybrid suture method that combines elements of traditional simple interrupted and retention sutures with a layer of sterile, absorbent rolled gauze or a dental roll placed beneath the suture knots.
The Technique
Traditional epidermal sutures concentrate pressure at the knot, increasing the risk for tram-track marks and patient discomfort. To address this, we developed a hybrid technique combining simple interrupted sutures with a sterile dental roll beneath the knots to reduce pressure, protect the wound, and promote comfortable wound healing.
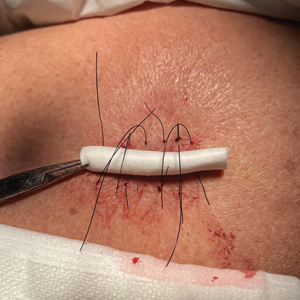
After excision of a cyst, we approximated the wound edges with buried vertical mattress sutures for eversion (a set-back buried dermal suture also may be used). The sutures initially were placed loosely but were left untied (eFigure 1A). A sterile dental roll with sterile petrolatum on the underside was positioned over the wound before the knots were secured, similar to a bolster dressing (eFigures 1B and 1C). The dressing then was covered and left in place for 24 to 48 hours. After removal of the dressing, no bandage was needed because the wound was clean and hemostatic and the dental roll had absorbed minimal drainage and protected the incision edges during the initial healing period. The patient applied petrolatum daily to prevent the dental roll from drying out. Sutures and the bolster were removed at 14 days without complications or complaints.
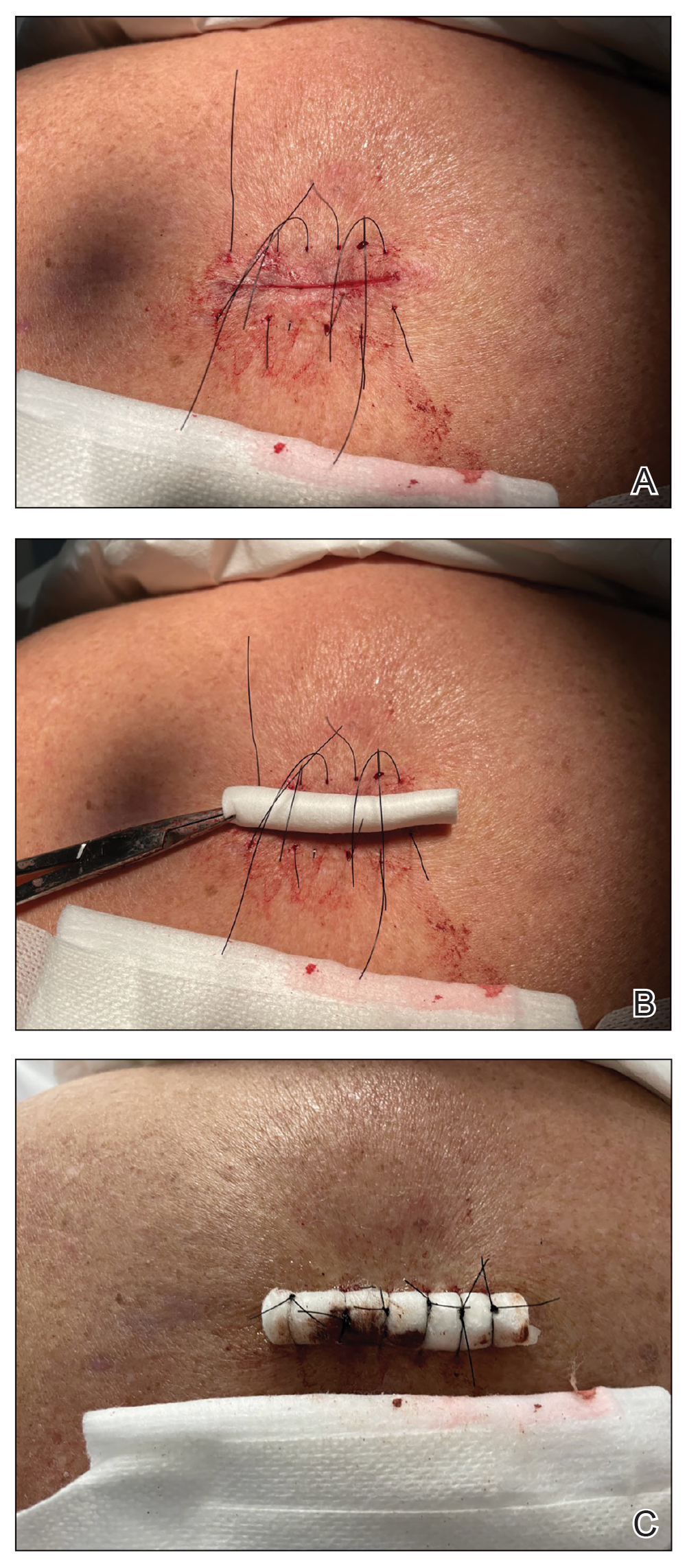
Rolled gauze may be used as an alternative to the dental roll. To maintain a clean surgical field, nonsterile gauze may be soaked in a disinfectant (eg, alcohol) and wrung out to remove excess moisture before placement on the skin. The side of the gauze in contact with the skin also should be lubricated with petroleum jelly to prevent sticking. If the sutures slip during knot tying, one end can be secured with a needle driver or hemostat. Patients should be advised to keep the dental roll dry to prevent maceration and promote optimal wound healing, but minor dampness is permissible if followed by air-drying.
This suturing method is most suitable for low- to moderate-tension closures such as cyst or lipoma excisions. The serosanguinous drainage can be absorbed by the gauze or dental roll while pressure is simultaneously applied to the wound. We do not recommend this technique for high-tension wounds in which large surface areas are removed (eg, skin cancer excisions on the posterior shoulder that require wide margins). Close monitoring of the wound for dehiscence is needed. As the sutures stretch and swelling decreases, the pressure is distributed accordingly without excessive compression to the wound line. Depending on the location, the sutures and dental roll can be removed in 7 to 14 days.
Practice Implications
Placing a dental roll or rolled gauze beneath suture knots can prevent tram-track scarring by eliminating direct knot-to-skin contact (eFigure 2).1,2 This technique distributes tension evenly, reduces the risk for wound edge necrosis, and absorbs serosanguinous drainage while providing hemostasis. The modification is quick, inexpensive, and especially beneficial for patients who may struggle with complex wound care, maintaining a clean environment until sutures are removed.
- Li E, Zhang T, Ma Q, et al. Effect of modified Allgöwer-Donati suture technique on wound cosmetics in spinal surgery. Orthop Surg. 2022;14:678-685. doi:10.1111/os.13188
- Park L, Lum ZC, Joo JS. Allgöwer-Donati suture: a technique to preserve skin microcirculation in dermatologic surgery. J Am Acad Dermatol. 2024;90:E129-E130. doi:10.1016/j.jaad.2023.05.052
Practice Gap
If not cared for properly, epidermal suture knots can cause discomfort, skin irritation, and an increased risk for infection. There is limited guidance on a simple adaptable method to reduce tram-track marks from epidermal sutures exerting pressure on the epidermis while still facilitating healing in dermatologic procedures such as excision of cysts or lipomas. We present a hybrid suture method that combines elements of traditional simple interrupted and retention sutures with a layer of sterile, absorbent rolled gauze or a dental roll placed beneath the suture knots.
The Technique
Traditional epidermal sutures concentrate pressure at the knot, increasing the risk for tram-track marks and patient discomfort. To address this, we developed a hybrid technique combining simple interrupted sutures with a sterile dental roll beneath the knots to reduce pressure, protect the wound, and promote comfortable wound healing.
After excision of a cyst, we approximated the wound edges with buried vertical mattress sutures for eversion (a set-back buried dermal suture also may be used). The sutures initially were placed loosely but were left untied (eFigure 1A). A sterile dental roll with sterile petrolatum on the underside was positioned over the wound before the knots were secured, similar to a bolster dressing (eFigures 1B and 1C). The dressing then was covered and left in place for 24 to 48 hours. After removal of the dressing, no bandage was needed because the wound was clean and hemostatic and the dental roll had absorbed minimal drainage and protected the incision edges during the initial healing period. The patient applied petrolatum daily to prevent the dental roll from drying out. Sutures and the bolster were removed at 14 days without complications or complaints.

Rolled gauze may be used as an alternative to the dental roll. To maintain a clean surgical field, nonsterile gauze may be soaked in a disinfectant (eg, alcohol) and wrung out to remove excess moisture before placement on the skin. The side of the gauze in contact with the skin also should be lubricated with petroleum jelly to prevent sticking. If the sutures slip during knot tying, one end can be secured with a needle driver or hemostat. Patients should be advised to keep the dental roll dry to prevent maceration and promote optimal wound healing, but minor dampness is permissible if followed by air-drying.
This suturing method is most suitable for low- to moderate-tension closures such as cyst or lipoma excisions. The serosanguinous drainage can be absorbed by the gauze or dental roll while pressure is simultaneously applied to the wound. We do not recommend this technique for high-tension wounds in which large surface areas are removed (eg, skin cancer excisions on the posterior shoulder that require wide margins). Close monitoring of the wound for dehiscence is needed. As the sutures stretch and swelling decreases, the pressure is distributed accordingly without excessive compression to the wound line. Depending on the location, the sutures and dental roll can be removed in 7 to 14 days.
Practice Implications
Placing a dental roll or rolled gauze beneath suture knots can prevent tram-track scarring by eliminating direct knot-to-skin contact (eFigure 2).1,2 This technique distributes tension evenly, reduces the risk for wound edge necrosis, and absorbs serosanguinous drainage while providing hemostasis. The modification is quick, inexpensive, and especially beneficial for patients who may struggle with complex wound care, maintaining a clean environment until sutures are removed.

Practice Gap
If not cared for properly, epidermal suture knots can cause discomfort, skin irritation, and an increased risk for infection. There is limited guidance on a simple adaptable method to reduce tram-track marks from epidermal sutures exerting pressure on the epidermis while still facilitating healing in dermatologic procedures such as excision of cysts or lipomas. We present a hybrid suture method that combines elements of traditional simple interrupted and retention sutures with a layer of sterile, absorbent rolled gauze or a dental roll placed beneath the suture knots.
The Technique
Traditional epidermal sutures concentrate pressure at the knot, increasing the risk for tram-track marks and patient discomfort. To address this, we developed a hybrid technique combining simple interrupted sutures with a sterile dental roll beneath the knots to reduce pressure, protect the wound, and promote comfortable wound healing.
After excision of a cyst, we approximated the wound edges with buried vertical mattress sutures for eversion (a set-back buried dermal suture also may be used). The sutures initially were placed loosely but were left untied (eFigure 1A). A sterile dental roll with sterile petrolatum on the underside was positioned over the wound before the knots were secured, similar to a bolster dressing (eFigures 1B and 1C). The dressing then was covered and left in place for 24 to 48 hours. After removal of the dressing, no bandage was needed because the wound was clean and hemostatic and the dental roll had absorbed minimal drainage and protected the incision edges during the initial healing period. The patient applied petrolatum daily to prevent the dental roll from drying out. Sutures and the bolster were removed at 14 days without complications or complaints.

Rolled gauze may be used as an alternative to the dental roll. To maintain a clean surgical field, nonsterile gauze may be soaked in a disinfectant (eg, alcohol) and wrung out to remove excess moisture before placement on the skin. The side of the gauze in contact with the skin also should be lubricated with petroleum jelly to prevent sticking. If the sutures slip during knot tying, one end can be secured with a needle driver or hemostat. Patients should be advised to keep the dental roll dry to prevent maceration and promote optimal wound healing, but minor dampness is permissible if followed by air-drying.
This suturing method is most suitable for low- to moderate-tension closures such as cyst or lipoma excisions. The serosanguinous drainage can be absorbed by the gauze or dental roll while pressure is simultaneously applied to the wound. We do not recommend this technique for high-tension wounds in which large surface areas are removed (eg, skin cancer excisions on the posterior shoulder that require wide margins). Close monitoring of the wound for dehiscence is needed. As the sutures stretch and swelling decreases, the pressure is distributed accordingly without excessive compression to the wound line. Depending on the location, the sutures and dental roll can be removed in 7 to 14 days.
Practice Implications
Placing a dental roll or rolled gauze beneath suture knots can prevent tram-track scarring by eliminating direct knot-to-skin contact (eFigure 2).1,2 This technique distributes tension evenly, reduces the risk for wound edge necrosis, and absorbs serosanguinous drainage while providing hemostasis. The modification is quick, inexpensive, and especially beneficial for patients who may struggle with complex wound care, maintaining a clean environment until sutures are removed.

- Li E, Zhang T, Ma Q, et al. Effect of modified Allgöwer-Donati suture technique on wound cosmetics in spinal surgery. Orthop Surg. 2022;14:678-685. doi:10.1111/os.13188
- Park L, Lum ZC, Joo JS. Allgöwer-Donati suture: a technique to preserve skin microcirculation in dermatologic surgery. J Am Acad Dermatol. 2024;90:E129-E130. doi:10.1016/j.jaad.2023.05.052
- Li E, Zhang T, Ma Q, et al. Effect of modified Allgöwer-Donati suture technique on wound cosmetics in spinal surgery. Orthop Surg. 2022;14:678-685. doi:10.1111/os.13188
- Park L, Lum ZC, Joo JS. Allgöwer-Donati suture: a technique to preserve skin microcirculation in dermatologic surgery. J Am Acad Dermatol. 2024;90:E129-E130. doi:10.1016/j.jaad.2023.05.052
A Hybrid Suture Technique: Suture Modification With Dental Roll Insertion
A Hybrid Suture Technique: Suture Modification With Dental Roll Insertion
Cutaneous Manifestations of Neglected Infectious Diseases in US Military Personnel
Cutaneous Manifestations of Neglected Infectious Diseases in US Military Personnel
Infectious diseases historically have posed major challenges to the operations and health of military forces. In recent conflicts, nonbattle injuries including infections have caused more evacuations than combat trauma.1 Deployment to endemic regions, poor sanitation, and trauma increase susceptibility to both common and rare infections, many of which have cutaneous manifestations.
Surveillance programs such as the Armed Forces Health Surveillance Division serve a critical role in monitoring outbreaks among deployed personnel.2 Cutaneous manifestations of systemic disease often serve as early clinical indicators, especially in settings with limited diagnostic resources. This review describes rarely encountered infectious agents for which US military personnel are at increased risk and outlines management strategies for clinicians practicing in austere environments.
EPIDEMIOLOGIC RISK FACTORS IN MILITARY POPULATIONS
United States military personnel face an elevated risk for infectious diseases when deployed in tropical and subtropical regions where endemic pathogens are prevalent. Exposure to soil, water, and insect vectors facilitates transmission. Direct exposure during combat or training combined with high-density housing, combat-related trauma, and constraints on hygiene access during operations increases infection risk.3
REGION-SPECIFIC PATHOGENS
Middle East
Leishmania species—Leishmania, a protozoa transmitted via sand fly bites, has caused multiple documented outbreaks among US troops in Iraq and Afghanistan, with a reported incidence of 14%.4 Leishmaniasis can present as 3 main clinical variants: cutaneous, visceral, and mucocutaneous. Cutaneous leishmaniasis typically manifests as painless ulcers covered with hemorrhagic crusts on exposed regions of the body. While typically self-limited, lesions frequently result in irreversible scarring. Many Leishmania species respond well to antimonials such as sodium stibogluconate. Preventive measures include wearing protective clothing and sleeping inside insecticide-treated bed nets.5
Coxiella burnetii—Coxiella burnetii transmits through inhalation of aerosolized particles originating from the urine, feces, birth products, or milk of infected bovine. In 2003, a small number of cases were identified in US service members exposed to livestock while serving in Iraq.6 Outbreaks also occurred during World War II, but it is unclear whether they were caused by naturally occurring C burnetii or biowarfare.7 Though primarily a systemic illness with severe pneumonia, Q fever may manifest with an associated purpuric or morbilliform rash.8 Doxycycline is recommended both for treatment and empiric coverage.6
Acinetobacter baumannii—This multidrug-resistant organism is known to infect combat wounds and is associated with nosocomial outbreaks in military hospitals. Studies suggest environmental contamination and health care transmission contribute substantially to outbreaks in military hospitals.9 Cutaneous manifestations can include cellulitis with a peau d’orange appearance or necrotizing fasciitis; however, pneumonia and bacteremia have been reported. Prompt culture and antibiotic initiation with debridement are essential for treatment.10 Antibiotic stewardship and strict infection control are critical to prevent outbreaks and limit resistance.9
Africa
Plasmodium species—Malaria remains a life-threatening disease found in tropical and subtropical areas around the world. Despite preventive measures, 30 cases among US service members were reported in 2024.11 Cutaneous findings include purpura fulminans, petechiae, acral necrosis, or reticulated erythema.12 Service members stationed in endemic areas should take prophylactic antimalarials. Symptoms include fevers, headaches, and malaise, with possible rapid deterioration.13
Mycobacterium ulcerans—Mycobacterium ulcerans causes extensive necrotic ulcers—commonly known as Buruli ulcers—which generally begin as a nodule, plaque, papule, or edematous lesion, eventually progressing to extensive ulceration. Despite no documented cases of US personnel contracting Buruli ulcers, those stationed in endemic regions remain at risk. Environmental reservoirs of M ulcerans are unknown, but its DNA has been isolated from water sources.14,15 These ulcers take months to heal, making wound management and antimycobacterial therapy essential. Primary preventive measures include avoidance of swimming in rivers or agricultural work in endemic areas.14
Mpox Virus—During the 2022 mpox outbreak, male service members who engaged in sexual activity with other men were at the highest risk, with 88.8% of infected service members reporting this practice.16 While the virus is endemic to Africa, 89.0% of cases were reported from service members stationed in the United States.17 Typical infection results in fever, headache, lymphadenopathy, and myalgias, followed by a facial rash that spreads over the body, palms, and soles. Safe-sex practices help prevent transmission, and there is a vaccine available for high-risk patients.16
Southeast Asia
Leptospira species—Leptospira is an aerobic spirochete found in tropical regions worldwide. Transmission occurs when water contaminated with urine from infected animals exposes humans to the organism. Infection manifests as a mild febrile illness, though approximately 10% of patients develop Weil syndrome, consisting of conjunctival suffusion, jaundice, and acute kidney injury. Treatment and prophylaxis include doxycycline, but severe disease warrants intravenous antibiotics.17,18 A 2014 outbreak among Marines in Japan highlighted poor prophylactic compliance as a key risk factor.19 Proper education for those at risk is essential to prevent future outbreaks.
Mycobacterium leprae—Leprosy is an acid-fast mycobacterium that remains endemic in the Pacific Islands and Southeast Asia. Case reports of US service members diagnosed with leprosy exist, though only in patients who emigrated from endemic areas.20 This disease has a spectrum of manifestations depending on the immune response, with tuberculoid leprosy showing a cell-mediated (T helper 1) response and lepromatous leprosy having more of a humoral (T helper 2) response.21 It manifests with hypopigmented anesthetic macules and peripheral neuropathy. Diagnosis is made by skin biopsy, which shows perineural lymphohistiocytic inflammation and non-necrotizing granulomas.20 The infection typically is curable with a multidrug regimen.21
Strongyloides stercoralis—This nematode causes infection by transdermal penetration of bare feet. They then migrate to the lungs where the patient coughs and swallows the nematode into the gastrointestinal tract. Strongyloides stercoralis autoinfect by penetrating the intestinal wall, resulting in chronic digestive, respiratory, and cutaneous symptoms. Worldwide prevalence of S stercoralis infection is estimated to be 10% to 40%, with foreign-born US military members at increased risk compared to the general military population.22,23 Larva currens may manifest with a pruritic erythematous plaque at the site of penetration that progresses to an intensely pruritic, creeping dermatitis as the nematode migrates under the skin. Avoidance of barefoot soil exposure and treatment with ivermectin are effective preventive and therapeutic measures.23
South America
Ancylostoma braziliense—Found throughout the subtropical world, this hookworm primarily infects dogs and cats and is found in their stool. Larva currens has a similar manifestation and life cycle to cutaneous larva migrans, but autoinfection does not occur. Transmission occurs similarly to S stercoralis and responds well to oral albendazole or ivermectin; however, the infection is self-limited.24 Military cases have been reported,25 though overall morbidity remains poorly characterized.
Dengue Virus—An arbovirus transmitted by Aedes mosquitoes, dengue remains a major military threat. Service members in the Vietnam War experienced an attack rate as high as 80%.26,27 Infection often manifests with retro-orbital pain and a morbilliform rash that occurs 2 to 5 days after fever, though severe cases may progress to hemorrhagic dengue with skin petechiae or ecchymosis.28 Immediate intervention is essential in symptomatic patients to prevent life-threatening progression. There are no dengue vaccines approved by the US Food and Drug Administration for adults, thus military personnel in endemic areas remain at risk.27
Trypanosoma cruzi—Chagas disease is transmitted when a reduviid infected with T cruzi bites and defecates on the patient’s skin. A skin nodule (chagoma) or painless eyelid edema (Romaña sign) may appear at the site of parasite entry. Chronic disease may result in dilated cardiomyopathy.29 Several cases of Chagas disease have been reported in South American military operations, including an outbreak in 9 Columbian military personnel.30 Cases in the southwestern United States have recently emerged, emphasizing the need for increased awareness.31 Proper insect repellent helps to ward off reduviid bugs. Nifurtimox and benznidazole are the only drugs with proven efficacy against T cruzi.29
Continental United States of America
Coccidioides immitis—The first reported case of coccidiomycosis was described in 1892 in a service member with debilitating masses and ulcers.32 Endemic to arid regions of the western United States, coccidioidomycosis affects military trainees at rates up to 32% annually in high-risk settings.33 Primary infection occurs in the lungs and may spread hematologically. The fungi prefer dry desert soils, which may aerosolize during military maneuvers. Coccidioidomycosis occasionally causes erythema nodosum, and diffuse infection shows verrucous plaques, ulcers, or abscesses. Dust avoidance and mask wearing are advised for those in endemic regions. Ketoconazole and amphotericin B are the only treatments approved by the US Food and Drug Administration.32 When starting immunosuppressive drugs, clinicians should inquire if patients have previously been stationed in Coccidioides-endemic areas, such as Fort Irwin, California, to avoid reactivation of the fungi.33
Francisella tularensis—Acquired via ticks or contact with wild animals, tularemia causes an ulceroglandular disease with regional lymphadenopathy. Inoculation requires as few as 10 to 25 organisms; thus it is considered a Category A agent for bioterror.34 Natural outbreaks have occurred during war times, most recently during the civil wars in Bosnia and Kosovo.35 Patients may present with a painful ulcer that enlarges to form a plaque with raised borders. Personnel in wooded areas should use tick precautions and handle wild animals cautiously. Treatment includes gentamicin for severe disease, with tetracyclines effective in mild cases.34
PREVENTION AND MANAGEMENT STRATEGIES IN AUSTERE SETTINGS
For health care professionals practicing in military settings, austere environments can provide a challenge for diagnosis of neglected diseases. Despite a lack of advanced diagnostic tools, practical options can be applied to the diagnostic process; for example, teledermatology is utilized for treatment of service members deployed to remote environments.36
Management of uncommon infectious diseases in military personnel often requires treatments outside those practiced in domestic clinics. Field management may indicate prompt empiric therapy while balancing the risks of overtreatment against those of missed diagnoses37; however, medical evacuation to a higher level of care may be indicated in patients with severe presentations to expedite diagnosis and treatment.38
Prevention remains the primary goal to avoid local outbreaks. Long-sleeved uniforms, DEET (N, N-diethyl-meta-toluamide)–based repellents, permethrin-impregnated clothing, and bed nets are effective for vector protection. Prophylactic medications and vaccinations often are provided when personnel are deployed to endemic locations.39
Onsite entomology teams also can provide surveillance of the local insect populations. They may contribute to vector control through insecticide application and environmental modification. The Armed Forces Health Surveillance Division and the Global Emerging Infections Surveillance Program monitor infectious threats in real time to locate any potential outbreaks, guiding operational responses.40
FINAL THOUGHTS
Dermatologic signs often provide early evidence of infection in military personnel. With increasing antimicrobial resistance and the emergence of new pathogens, it is imperative for clinicians treating members of the military to recognize cutaneous signs, employ efficient diagnostic strategies, and encourage proactive prevention. A collaborative approach spanning dermatology, infectious disease, and public health is essential to protect the modern service member.
- Murray CK. Infectious disease complications of combat-related injuries. Crit Care Med. 2008;36(7 suppl):S358-S364. doi:10.1097/CCM.0b013e31817e2ffc
- Armed Forces Health Surveillance Division. AFHSD Annual Report. Defense Health Agency; 2023. Accessed March 5, 2026. https://www.health.mil/Reference-Center/Reports/2024/09/19/AFHSD-Annual-Report-2023
- Murray CK, Yun HC, Markelz AE, et al. Operation United Assistance: infectious disease threats to deployed military personnel. Military Medicine. 2015;180:626-651. doi:10.7205/MILMED-D-14-00691
- Niba Rawlings N, Bailey M, Courtenay O. Leishmaniasis in deployed military populations: a systematic review and meta-analysis. PLoS Negl Trop Dis. 2025;19:E0012680. doi:10.1371/journal.pntd.0012680
- de Vries HJC, Schallig HD. Cutaneous leishmaniasis: a 2022 updated narrative review into diagnosis and management developments. Am J Clin Dermatol. 2022;23:823-840. doi:10.1007/s40257-022-00726-8
- Anderson AD, Smoak B, Shuping E, et al. Q fever and the US military. Emerg Infect Dis. 2005;11:1320-1322. doi:10.3201/eid1108.050314
- Madariaga MG, Rezai K, Trenholme GM, et al. Q fever: a biological weapon in your backyard. Lancet Infect Dis. 2003;3:709-721. doi:10.1016/S1473-3099(03)00804-1
- Uribe Pulido N, Escorcia García C, Cabrera Orrego R, et al. Acute Q fever with dermatologic manifestations, molecular diagnosis, and no seroconversion. Open Forum Infect Dis. 2021;8:ofab458. doi:10.1093/ofid/ofab458
- Scott P, Deye G, Srinivasan A, et al. An outbreak of multidrug-resistant acinetobacter baumannii-calcoaceticus complex infection in the US Military Health Care System associated with military operations in Iraq. Clin Infect Dis. 2007;44:1577-1584. doi:10.1086/518170
- Guerrero DM, Perez F, Conger NG, et al. Acinetobacter baumannii-associated skin and soft tissue infections: recognizing a broadening spectrum of disease. Surg Infect (Larchmt). 2010;11:49-57. doi:10.1089/sur.2009.022
- Armed Forces Health Surveillance Division. Malaria among members of the US Armed Forces, 2024. MSMR. 2025;32:22-28.
- Farkouh CS, Abdi P, Amatul-Hadi F, et al. Cutaneous manifestations of malaria and their prognostic windows: a narrative review. Cureus. 2023;15:E41706. doi:10.7759/cureus.41706
- Shahbodaghi SD, Rathjen NA. Malaria: prevention, diagnosis, and treatment. Am Fam Physician. 2022;106:270-278.
- Yotsu RR, Suzuki K, Simmonds RE, et al. Buruli ulcer: a review of the current knowledge. Curr Trop Med Rep. 2018;5:247-256. doi:10.1007/s40475-018-0166-2
- Portaels F, Meyers WM, Ablordey A, et al. First cultivation and characterization of Mycobacterium ulcerans from the environment. PLoS Negl Trop Dis. 2008;2:E178. doi:10.1371/journal.pntd.0000178
- Metcalf-Kelly M, Garrison M, Stidham R. Characteristics of mpox cases diagnosed in Military Health System beneficiaries, May 2022-April 2024. MSMR. 2024;31:7-11.
- Rajapakse S. Leptospirosis: clinical aspects. Clin Med (Lond). 2022;22:14-17. doi:10.7861/clinmed.2021-0784
- Heath CW, Alexander AD, Galton MM. Leptospirosis in the United States: a of 483 cases in man, 1949–1961. N Engl J Med. 1965;273:857-864. doi:10.1056/NEJM196510142731606
- Mason V. Mystery outbreak investigation 2014—Leptospirosis licerasiae. November 17, 2017. Accessed March 5, 2026. https://usupulse.blogspot.com/2017/11/mystery-outbreak-investigation-2014.html
- Berjohn CM, DuPlessis CA, Tieu K, et al. Multibacillary leprosy in an active duty military member. Emerg Infect Dis. 2015;21:1077-1078. doi:10.3201/eid2106.141666
- Scollard DM, Adams LB, Gillis TP, et al. The continuing challenges of leprosy. Clin Microbiol Rev. 2006;19:338-381. doi:10.1128/CMR.19.2.338-381.2006
- Schär F, Trostdorf U, Giardina F, et al. Strongyloides stercoralis: global distribution and risk factors. PLoS Negl Trop Dis. 2013;7:E2288. doi:10.1371/journal.pntd.0002288
- Taheri B, Kuo HC, Hockenbury N, et al. Strongyloides stercoralis in the US Military Health System. Open Forum Infect Dis. 2023;10:ofad127. doi:10.1093/ofid/ofad127
- Bowman DD, Montgomery SP, Zajac AM, et al. Hookworms of dogs and cats as agents of cutaneous larva migrans. Trends Parasitol. 2010;26:162-167. doi:10.1016/j.pt.2010.01.005
- Inglis DM, Bailey MS. Unusual presentations of cutaneous larva migrans in British military personnel. BMJ Mil Health. 2023;169:E78-E81. doi:10.1136/bmjmilitary-2020-001677
- Halstead SB, Udomsakdi S, Singharaj P, et al. Dengue and chikungunya virus infection in man in Thailand, 1962–1964. 3. clinical, epidemiologic, and virologic observations on disease in non-indigenous white persons. Am J Trop Med Hyg. 1969;18:984-996. doi:10.4269/ajtmh.1969.18.984
- Gibbons RV, Streitz M, Babina T, et al. Dengue and US military operations from the Spanish-American War through today. Emerg Infect Dis. 2012;18:623-630. doi:10.3201/eid1804.110134
- Guzman MG, Harris E. Dengue. Lancet. 2015;385:453-465. doi:10.1016/S0140-6736(14)60572-9
- Bern C. Chagas’ disease. N Engl J Med. 2015;373:456-466. doi:10.1056/NEJMra1410150
- Vergara HD, Gómez CH, Faccini-Martínez ÁA, et al. Acute Chagas disease outbreak among military personnel, Colombia, 2021. Emerg Infect Dis. 2023;29:1882-1885. doi:10.3201/eid2909.230886
- Harris N, Woc-Colburn L, Gunter SM, et al. Autochthonous Chagas disease in the southern United States: a case report of suspected residential and military exposures. Zoonoses Public Health. 2017;64:491-493. doi:10.1111/zph.12360
- Crum NF. Coccidioidomycosis: a contemporary review. Infect Dis Ther. 2022;11:713-742. doi:10.1007/s40121-022-00606-y
- Crum NF, Potter M, Pappagianis D. Seroincidence of coccidioidomycosis during military desert training exercises. J Clin Microbiol. 2004;42:4552-4555. doi:10.1128/JCM.42.10.4552-4555.2004
- Antonello RM, Giacomelli A, Riccardi N. Tularemia for clinicians: an up-to-date review on epidemiology, diagnosis, prevention and treatment. Eur J Intern Med. 2025;135:25-32. doi:10.1016/j.ejim.2025.03.013
- Nelson CA, Sjöstedt A. Tularemia: a storied history, an ongoing threat. Clin Infect Dis. 2024;78(supplement_1):S1-S3. doi:10.1093/cid/ciad681
- Pendlebury GA, Roman J, Shrivastava V, et al. A call to action: evidence for the military integration of teledermoscopy in a pandemic era. Dermatopathology (Basel). 2022;9:327-342. doi:10.3390/dermatopathology9040039
- Bhagchandani R, Singhi S, Peter JV, et al. Tropical fevers: management guidelines. Indian J Crit Care Med. 2014;18:62-69. doi:10.4103/0972-5229.126074
- Joint Chiefs of Staff. Joint Publication 4-02: Joint Health Services. December 11, 2017. Accessed March 5, 2026. https://cdmrp.health.mil/pubs/pdf/Joint%20Health%20Services%20Publication%20JP%204-02.pdf
- Armed Services Pest Management Board. Technical Guide No. 36: Personal Protective Measures Against Insects and Other Arthropods of Military Significance. Updated November 2015. Accessed March 5, 2026. https://www.acq.osd.mil/eie/afpmb/docs/techguides/tg36.pdf
- Global Emerging Infections Surveillance. Armed Forces Health Surveillance Division Annual Report 2024. Defense Health Agency; 2024:15-28. Accessed March 17, 2026. https://www.health.mil/Reference-Center/Reports/2026/01/05/AFHSD-Annual-Report-2024
Infectious diseases historically have posed major challenges to the operations and health of military forces. In recent conflicts, nonbattle injuries including infections have caused more evacuations than combat trauma.1 Deployment to endemic regions, poor sanitation, and trauma increase susceptibility to both common and rare infections, many of which have cutaneous manifestations.
Surveillance programs such as the Armed Forces Health Surveillance Division serve a critical role in monitoring outbreaks among deployed personnel.2 Cutaneous manifestations of systemic disease often serve as early clinical indicators, especially in settings with limited diagnostic resources. This review describes rarely encountered infectious agents for which US military personnel are at increased risk and outlines management strategies for clinicians practicing in austere environments.
EPIDEMIOLOGIC RISK FACTORS IN MILITARY POPULATIONS
United States military personnel face an elevated risk for infectious diseases when deployed in tropical and subtropical regions where endemic pathogens are prevalent. Exposure to soil, water, and insect vectors facilitates transmission. Direct exposure during combat or training combined with high-density housing, combat-related trauma, and constraints on hygiene access during operations increases infection risk.3
REGION-SPECIFIC PATHOGENS
Middle East
Leishmania species—Leishmania, a protozoa transmitted via sand fly bites, has caused multiple documented outbreaks among US troops in Iraq and Afghanistan, with a reported incidence of 14%.4 Leishmaniasis can present as 3 main clinical variants: cutaneous, visceral, and mucocutaneous. Cutaneous leishmaniasis typically manifests as painless ulcers covered with hemorrhagic crusts on exposed regions of the body. While typically self-limited, lesions frequently result in irreversible scarring. Many Leishmania species respond well to antimonials such as sodium stibogluconate. Preventive measures include wearing protective clothing and sleeping inside insecticide-treated bed nets.5
Coxiella burnetii—Coxiella burnetii transmits through inhalation of aerosolized particles originating from the urine, feces, birth products, or milk of infected bovine. In 2003, a small number of cases were identified in US service members exposed to livestock while serving in Iraq.6 Outbreaks also occurred during World War II, but it is unclear whether they were caused by naturally occurring C burnetii or biowarfare.7 Though primarily a systemic illness with severe pneumonia, Q fever may manifest with an associated purpuric or morbilliform rash.8 Doxycycline is recommended both for treatment and empiric coverage.6
Acinetobacter baumannii—This multidrug-resistant organism is known to infect combat wounds and is associated with nosocomial outbreaks in military hospitals. Studies suggest environmental contamination and health care transmission contribute substantially to outbreaks in military hospitals.9 Cutaneous manifestations can include cellulitis with a peau d’orange appearance or necrotizing fasciitis; however, pneumonia and bacteremia have been reported. Prompt culture and antibiotic initiation with debridement are essential for treatment.10 Antibiotic stewardship and strict infection control are critical to prevent outbreaks and limit resistance.9
Africa
Plasmodium species—Malaria remains a life-threatening disease found in tropical and subtropical areas around the world. Despite preventive measures, 30 cases among US service members were reported in 2024.11 Cutaneous findings include purpura fulminans, petechiae, acral necrosis, or reticulated erythema.12 Service members stationed in endemic areas should take prophylactic antimalarials. Symptoms include fevers, headaches, and malaise, with possible rapid deterioration.13
Mycobacterium ulcerans—Mycobacterium ulcerans causes extensive necrotic ulcers—commonly known as Buruli ulcers—which generally begin as a nodule, plaque, papule, or edematous lesion, eventually progressing to extensive ulceration. Despite no documented cases of US personnel contracting Buruli ulcers, those stationed in endemic regions remain at risk. Environmental reservoirs of M ulcerans are unknown, but its DNA has been isolated from water sources.14,15 These ulcers take months to heal, making wound management and antimycobacterial therapy essential. Primary preventive measures include avoidance of swimming in rivers or agricultural work in endemic areas.14
Mpox Virus—During the 2022 mpox outbreak, male service members who engaged in sexual activity with other men were at the highest risk, with 88.8% of infected service members reporting this practice.16 While the virus is endemic to Africa, 89.0% of cases were reported from service members stationed in the United States.17 Typical infection results in fever, headache, lymphadenopathy, and myalgias, followed by a facial rash that spreads over the body, palms, and soles. Safe-sex practices help prevent transmission, and there is a vaccine available for high-risk patients.16
Southeast Asia
Leptospira species—Leptospira is an aerobic spirochete found in tropical regions worldwide. Transmission occurs when water contaminated with urine from infected animals exposes humans to the organism. Infection manifests as a mild febrile illness, though approximately 10% of patients develop Weil syndrome, consisting of conjunctival suffusion, jaundice, and acute kidney injury. Treatment and prophylaxis include doxycycline, but severe disease warrants intravenous antibiotics.17,18 A 2014 outbreak among Marines in Japan highlighted poor prophylactic compliance as a key risk factor.19 Proper education for those at risk is essential to prevent future outbreaks.
Mycobacterium leprae—Leprosy is an acid-fast mycobacterium that remains endemic in the Pacific Islands and Southeast Asia. Case reports of US service members diagnosed with leprosy exist, though only in patients who emigrated from endemic areas.20 This disease has a spectrum of manifestations depending on the immune response, with tuberculoid leprosy showing a cell-mediated (T helper 1) response and lepromatous leprosy having more of a humoral (T helper 2) response.21 It manifests with hypopigmented anesthetic macules and peripheral neuropathy. Diagnosis is made by skin biopsy, which shows perineural lymphohistiocytic inflammation and non-necrotizing granulomas.20 The infection typically is curable with a multidrug regimen.21
Strongyloides stercoralis—This nematode causes infection by transdermal penetration of bare feet. They then migrate to the lungs where the patient coughs and swallows the nematode into the gastrointestinal tract. Strongyloides stercoralis autoinfect by penetrating the intestinal wall, resulting in chronic digestive, respiratory, and cutaneous symptoms. Worldwide prevalence of S stercoralis infection is estimated to be 10% to 40%, with foreign-born US military members at increased risk compared to the general military population.22,23 Larva currens may manifest with a pruritic erythematous plaque at the site of penetration that progresses to an intensely pruritic, creeping dermatitis as the nematode migrates under the skin. Avoidance of barefoot soil exposure and treatment with ivermectin are effective preventive and therapeutic measures.23
South America
Ancylostoma braziliense—Found throughout the subtropical world, this hookworm primarily infects dogs and cats and is found in their stool. Larva currens has a similar manifestation and life cycle to cutaneous larva migrans, but autoinfection does not occur. Transmission occurs similarly to S stercoralis and responds well to oral albendazole or ivermectin; however, the infection is self-limited.24 Military cases have been reported,25 though overall morbidity remains poorly characterized.
Dengue Virus—An arbovirus transmitted by Aedes mosquitoes, dengue remains a major military threat. Service members in the Vietnam War experienced an attack rate as high as 80%.26,27 Infection often manifests with retro-orbital pain and a morbilliform rash that occurs 2 to 5 days after fever, though severe cases may progress to hemorrhagic dengue with skin petechiae or ecchymosis.28 Immediate intervention is essential in symptomatic patients to prevent life-threatening progression. There are no dengue vaccines approved by the US Food and Drug Administration for adults, thus military personnel in endemic areas remain at risk.27
Trypanosoma cruzi—Chagas disease is transmitted when a reduviid infected with T cruzi bites and defecates on the patient’s skin. A skin nodule (chagoma) or painless eyelid edema (Romaña sign) may appear at the site of parasite entry. Chronic disease may result in dilated cardiomyopathy.29 Several cases of Chagas disease have been reported in South American military operations, including an outbreak in 9 Columbian military personnel.30 Cases in the southwestern United States have recently emerged, emphasizing the need for increased awareness.31 Proper insect repellent helps to ward off reduviid bugs. Nifurtimox and benznidazole are the only drugs with proven efficacy against T cruzi.29
Continental United States of America
Coccidioides immitis—The first reported case of coccidiomycosis was described in 1892 in a service member with debilitating masses and ulcers.32 Endemic to arid regions of the western United States, coccidioidomycosis affects military trainees at rates up to 32% annually in high-risk settings.33 Primary infection occurs in the lungs and may spread hematologically. The fungi prefer dry desert soils, which may aerosolize during military maneuvers. Coccidioidomycosis occasionally causes erythema nodosum, and diffuse infection shows verrucous plaques, ulcers, or abscesses. Dust avoidance and mask wearing are advised for those in endemic regions. Ketoconazole and amphotericin B are the only treatments approved by the US Food and Drug Administration.32 When starting immunosuppressive drugs, clinicians should inquire if patients have previously been stationed in Coccidioides-endemic areas, such as Fort Irwin, California, to avoid reactivation of the fungi.33
Francisella tularensis—Acquired via ticks or contact with wild animals, tularemia causes an ulceroglandular disease with regional lymphadenopathy. Inoculation requires as few as 10 to 25 organisms; thus it is considered a Category A agent for bioterror.34 Natural outbreaks have occurred during war times, most recently during the civil wars in Bosnia and Kosovo.35 Patients may present with a painful ulcer that enlarges to form a plaque with raised borders. Personnel in wooded areas should use tick precautions and handle wild animals cautiously. Treatment includes gentamicin for severe disease, with tetracyclines effective in mild cases.34
PREVENTION AND MANAGEMENT STRATEGIES IN AUSTERE SETTINGS
For health care professionals practicing in military settings, austere environments can provide a challenge for diagnosis of neglected diseases. Despite a lack of advanced diagnostic tools, practical options can be applied to the diagnostic process; for example, teledermatology is utilized for treatment of service members deployed to remote environments.36
Management of uncommon infectious diseases in military personnel often requires treatments outside those practiced in domestic clinics. Field management may indicate prompt empiric therapy while balancing the risks of overtreatment against those of missed diagnoses37; however, medical evacuation to a higher level of care may be indicated in patients with severe presentations to expedite diagnosis and treatment.38
Prevention remains the primary goal to avoid local outbreaks. Long-sleeved uniforms, DEET (N, N-diethyl-meta-toluamide)–based repellents, permethrin-impregnated clothing, and bed nets are effective for vector protection. Prophylactic medications and vaccinations often are provided when personnel are deployed to endemic locations.39
Onsite entomology teams also can provide surveillance of the local insect populations. They may contribute to vector control through insecticide application and environmental modification. The Armed Forces Health Surveillance Division and the Global Emerging Infections Surveillance Program monitor infectious threats in real time to locate any potential outbreaks, guiding operational responses.40
FINAL THOUGHTS
Dermatologic signs often provide early evidence of infection in military personnel. With increasing antimicrobial resistance and the emergence of new pathogens, it is imperative for clinicians treating members of the military to recognize cutaneous signs, employ efficient diagnostic strategies, and encourage proactive prevention. A collaborative approach spanning dermatology, infectious disease, and public health is essential to protect the modern service member.
Infectious diseases historically have posed major challenges to the operations and health of military forces. In recent conflicts, nonbattle injuries including infections have caused more evacuations than combat trauma.1 Deployment to endemic regions, poor sanitation, and trauma increase susceptibility to both common and rare infections, many of which have cutaneous manifestations.
Surveillance programs such as the Armed Forces Health Surveillance Division serve a critical role in monitoring outbreaks among deployed personnel.2 Cutaneous manifestations of systemic disease often serve as early clinical indicators, especially in settings with limited diagnostic resources. This review describes rarely encountered infectious agents for which US military personnel are at increased risk and outlines management strategies for clinicians practicing in austere environments.
EPIDEMIOLOGIC RISK FACTORS IN MILITARY POPULATIONS
United States military personnel face an elevated risk for infectious diseases when deployed in tropical and subtropical regions where endemic pathogens are prevalent. Exposure to soil, water, and insect vectors facilitates transmission. Direct exposure during combat or training combined with high-density housing, combat-related trauma, and constraints on hygiene access during operations increases infection risk.3
REGION-SPECIFIC PATHOGENS
Middle East
Leishmania species—Leishmania, a protozoa transmitted via sand fly bites, has caused multiple documented outbreaks among US troops in Iraq and Afghanistan, with a reported incidence of 14%.4 Leishmaniasis can present as 3 main clinical variants: cutaneous, visceral, and mucocutaneous. Cutaneous leishmaniasis typically manifests as painless ulcers covered with hemorrhagic crusts on exposed regions of the body. While typically self-limited, lesions frequently result in irreversible scarring. Many Leishmania species respond well to antimonials such as sodium stibogluconate. Preventive measures include wearing protective clothing and sleeping inside insecticide-treated bed nets.5
Coxiella burnetii—Coxiella burnetii transmits through inhalation of aerosolized particles originating from the urine, feces, birth products, or milk of infected bovine. In 2003, a small number of cases were identified in US service members exposed to livestock while serving in Iraq.6 Outbreaks also occurred during World War II, but it is unclear whether they were caused by naturally occurring C burnetii or biowarfare.7 Though primarily a systemic illness with severe pneumonia, Q fever may manifest with an associated purpuric or morbilliform rash.8 Doxycycline is recommended both for treatment and empiric coverage.6
Acinetobacter baumannii—This multidrug-resistant organism is known to infect combat wounds and is associated with nosocomial outbreaks in military hospitals. Studies suggest environmental contamination and health care transmission contribute substantially to outbreaks in military hospitals.9 Cutaneous manifestations can include cellulitis with a peau d’orange appearance or necrotizing fasciitis; however, pneumonia and bacteremia have been reported. Prompt culture and antibiotic initiation with debridement are essential for treatment.10 Antibiotic stewardship and strict infection control are critical to prevent outbreaks and limit resistance.9
Africa
Plasmodium species—Malaria remains a life-threatening disease found in tropical and subtropical areas around the world. Despite preventive measures, 30 cases among US service members were reported in 2024.11 Cutaneous findings include purpura fulminans, petechiae, acral necrosis, or reticulated erythema.12 Service members stationed in endemic areas should take prophylactic antimalarials. Symptoms include fevers, headaches, and malaise, with possible rapid deterioration.13
Mycobacterium ulcerans—Mycobacterium ulcerans causes extensive necrotic ulcers—commonly known as Buruli ulcers—which generally begin as a nodule, plaque, papule, or edematous lesion, eventually progressing to extensive ulceration. Despite no documented cases of US personnel contracting Buruli ulcers, those stationed in endemic regions remain at risk. Environmental reservoirs of M ulcerans are unknown, but its DNA has been isolated from water sources.14,15 These ulcers take months to heal, making wound management and antimycobacterial therapy essential. Primary preventive measures include avoidance of swimming in rivers or agricultural work in endemic areas.14
Mpox Virus—During the 2022 mpox outbreak, male service members who engaged in sexual activity with other men were at the highest risk, with 88.8% of infected service members reporting this practice.16 While the virus is endemic to Africa, 89.0% of cases were reported from service members stationed in the United States.17 Typical infection results in fever, headache, lymphadenopathy, and myalgias, followed by a facial rash that spreads over the body, palms, and soles. Safe-sex practices help prevent transmission, and there is a vaccine available for high-risk patients.16
Southeast Asia
Leptospira species—Leptospira is an aerobic spirochete found in tropical regions worldwide. Transmission occurs when water contaminated with urine from infected animals exposes humans to the organism. Infection manifests as a mild febrile illness, though approximately 10% of patients develop Weil syndrome, consisting of conjunctival suffusion, jaundice, and acute kidney injury. Treatment and prophylaxis include doxycycline, but severe disease warrants intravenous antibiotics.17,18 A 2014 outbreak among Marines in Japan highlighted poor prophylactic compliance as a key risk factor.19 Proper education for those at risk is essential to prevent future outbreaks.
Mycobacterium leprae—Leprosy is an acid-fast mycobacterium that remains endemic in the Pacific Islands and Southeast Asia. Case reports of US service members diagnosed with leprosy exist, though only in patients who emigrated from endemic areas.20 This disease has a spectrum of manifestations depending on the immune response, with tuberculoid leprosy showing a cell-mediated (T helper 1) response and lepromatous leprosy having more of a humoral (T helper 2) response.21 It manifests with hypopigmented anesthetic macules and peripheral neuropathy. Diagnosis is made by skin biopsy, which shows perineural lymphohistiocytic inflammation and non-necrotizing granulomas.20 The infection typically is curable with a multidrug regimen.21
Strongyloides stercoralis—This nematode causes infection by transdermal penetration of bare feet. They then migrate to the lungs where the patient coughs and swallows the nematode into the gastrointestinal tract. Strongyloides stercoralis autoinfect by penetrating the intestinal wall, resulting in chronic digestive, respiratory, and cutaneous symptoms. Worldwide prevalence of S stercoralis infection is estimated to be 10% to 40%, with foreign-born US military members at increased risk compared to the general military population.22,23 Larva currens may manifest with a pruritic erythematous plaque at the site of penetration that progresses to an intensely pruritic, creeping dermatitis as the nematode migrates under the skin. Avoidance of barefoot soil exposure and treatment with ivermectin are effective preventive and therapeutic measures.23
South America
Ancylostoma braziliense—Found throughout the subtropical world, this hookworm primarily infects dogs and cats and is found in their stool. Larva currens has a similar manifestation and life cycle to cutaneous larva migrans, but autoinfection does not occur. Transmission occurs similarly to S stercoralis and responds well to oral albendazole or ivermectin; however, the infection is self-limited.24 Military cases have been reported,25 though overall morbidity remains poorly characterized.
Dengue Virus—An arbovirus transmitted by Aedes mosquitoes, dengue remains a major military threat. Service members in the Vietnam War experienced an attack rate as high as 80%.26,27 Infection often manifests with retro-orbital pain and a morbilliform rash that occurs 2 to 5 days after fever, though severe cases may progress to hemorrhagic dengue with skin petechiae or ecchymosis.28 Immediate intervention is essential in symptomatic patients to prevent life-threatening progression. There are no dengue vaccines approved by the US Food and Drug Administration for adults, thus military personnel in endemic areas remain at risk.27
Trypanosoma cruzi—Chagas disease is transmitted when a reduviid infected with T cruzi bites and defecates on the patient’s skin. A skin nodule (chagoma) or painless eyelid edema (Romaña sign) may appear at the site of parasite entry. Chronic disease may result in dilated cardiomyopathy.29 Several cases of Chagas disease have been reported in South American military operations, including an outbreak in 9 Columbian military personnel.30 Cases in the southwestern United States have recently emerged, emphasizing the need for increased awareness.31 Proper insect repellent helps to ward off reduviid bugs. Nifurtimox and benznidazole are the only drugs with proven efficacy against T cruzi.29
Continental United States of America
Coccidioides immitis—The first reported case of coccidiomycosis was described in 1892 in a service member with debilitating masses and ulcers.32 Endemic to arid regions of the western United States, coccidioidomycosis affects military trainees at rates up to 32% annually in high-risk settings.33 Primary infection occurs in the lungs and may spread hematologically. The fungi prefer dry desert soils, which may aerosolize during military maneuvers. Coccidioidomycosis occasionally causes erythema nodosum, and diffuse infection shows verrucous plaques, ulcers, or abscesses. Dust avoidance and mask wearing are advised for those in endemic regions. Ketoconazole and amphotericin B are the only treatments approved by the US Food and Drug Administration.32 When starting immunosuppressive drugs, clinicians should inquire if patients have previously been stationed in Coccidioides-endemic areas, such as Fort Irwin, California, to avoid reactivation of the fungi.33
Francisella tularensis—Acquired via ticks or contact with wild animals, tularemia causes an ulceroglandular disease with regional lymphadenopathy. Inoculation requires as few as 10 to 25 organisms; thus it is considered a Category A agent for bioterror.34 Natural outbreaks have occurred during war times, most recently during the civil wars in Bosnia and Kosovo.35 Patients may present with a painful ulcer that enlarges to form a plaque with raised borders. Personnel in wooded areas should use tick precautions and handle wild animals cautiously. Treatment includes gentamicin for severe disease, with tetracyclines effective in mild cases.34
PREVENTION AND MANAGEMENT STRATEGIES IN AUSTERE SETTINGS
For health care professionals practicing in military settings, austere environments can provide a challenge for diagnosis of neglected diseases. Despite a lack of advanced diagnostic tools, practical options can be applied to the diagnostic process; for example, teledermatology is utilized for treatment of service members deployed to remote environments.36
Management of uncommon infectious diseases in military personnel often requires treatments outside those practiced in domestic clinics. Field management may indicate prompt empiric therapy while balancing the risks of overtreatment against those of missed diagnoses37; however, medical evacuation to a higher level of care may be indicated in patients with severe presentations to expedite diagnosis and treatment.38
Prevention remains the primary goal to avoid local outbreaks. Long-sleeved uniforms, DEET (N, N-diethyl-meta-toluamide)–based repellents, permethrin-impregnated clothing, and bed nets are effective for vector protection. Prophylactic medications and vaccinations often are provided when personnel are deployed to endemic locations.39
Onsite entomology teams also can provide surveillance of the local insect populations. They may contribute to vector control through insecticide application and environmental modification. The Armed Forces Health Surveillance Division and the Global Emerging Infections Surveillance Program monitor infectious threats in real time to locate any potential outbreaks, guiding operational responses.40
FINAL THOUGHTS
Dermatologic signs often provide early evidence of infection in military personnel. With increasing antimicrobial resistance and the emergence of new pathogens, it is imperative for clinicians treating members of the military to recognize cutaneous signs, employ efficient diagnostic strategies, and encourage proactive prevention. A collaborative approach spanning dermatology, infectious disease, and public health is essential to protect the modern service member.
- Murray CK. Infectious disease complications of combat-related injuries. Crit Care Med. 2008;36(7 suppl):S358-S364. doi:10.1097/CCM.0b013e31817e2ffc
- Armed Forces Health Surveillance Division. AFHSD Annual Report. Defense Health Agency; 2023. Accessed March 5, 2026. https://www.health.mil/Reference-Center/Reports/2024/09/19/AFHSD-Annual-Report-2023
- Murray CK, Yun HC, Markelz AE, et al. Operation United Assistance: infectious disease threats to deployed military personnel. Military Medicine. 2015;180:626-651. doi:10.7205/MILMED-D-14-00691
- Niba Rawlings N, Bailey M, Courtenay O. Leishmaniasis in deployed military populations: a systematic review and meta-analysis. PLoS Negl Trop Dis. 2025;19:E0012680. doi:10.1371/journal.pntd.0012680
- de Vries HJC, Schallig HD. Cutaneous leishmaniasis: a 2022 updated narrative review into diagnosis and management developments. Am J Clin Dermatol. 2022;23:823-840. doi:10.1007/s40257-022-00726-8
- Anderson AD, Smoak B, Shuping E, et al. Q fever and the US military. Emerg Infect Dis. 2005;11:1320-1322. doi:10.3201/eid1108.050314
- Madariaga MG, Rezai K, Trenholme GM, et al. Q fever: a biological weapon in your backyard. Lancet Infect Dis. 2003;3:709-721. doi:10.1016/S1473-3099(03)00804-1
- Uribe Pulido N, Escorcia García C, Cabrera Orrego R, et al. Acute Q fever with dermatologic manifestations, molecular diagnosis, and no seroconversion. Open Forum Infect Dis. 2021;8:ofab458. doi:10.1093/ofid/ofab458
- Scott P, Deye G, Srinivasan A, et al. An outbreak of multidrug-resistant acinetobacter baumannii-calcoaceticus complex infection in the US Military Health Care System associated with military operations in Iraq. Clin Infect Dis. 2007;44:1577-1584. doi:10.1086/518170
- Guerrero DM, Perez F, Conger NG, et al. Acinetobacter baumannii-associated skin and soft tissue infections: recognizing a broadening spectrum of disease. Surg Infect (Larchmt). 2010;11:49-57. doi:10.1089/sur.2009.022
- Armed Forces Health Surveillance Division. Malaria among members of the US Armed Forces, 2024. MSMR. 2025;32:22-28.
- Farkouh CS, Abdi P, Amatul-Hadi F, et al. Cutaneous manifestations of malaria and their prognostic windows: a narrative review. Cureus. 2023;15:E41706. doi:10.7759/cureus.41706
- Shahbodaghi SD, Rathjen NA. Malaria: prevention, diagnosis, and treatment. Am Fam Physician. 2022;106:270-278.
- Yotsu RR, Suzuki K, Simmonds RE, et al. Buruli ulcer: a review of the current knowledge. Curr Trop Med Rep. 2018;5:247-256. doi:10.1007/s40475-018-0166-2
- Portaels F, Meyers WM, Ablordey A, et al. First cultivation and characterization of Mycobacterium ulcerans from the environment. PLoS Negl Trop Dis. 2008;2:E178. doi:10.1371/journal.pntd.0000178
- Metcalf-Kelly M, Garrison M, Stidham R. Characteristics of mpox cases diagnosed in Military Health System beneficiaries, May 2022-April 2024. MSMR. 2024;31:7-11.
- Rajapakse S. Leptospirosis: clinical aspects. Clin Med (Lond). 2022;22:14-17. doi:10.7861/clinmed.2021-0784
- Heath CW, Alexander AD, Galton MM. Leptospirosis in the United States: a of 483 cases in man, 1949–1961. N Engl J Med. 1965;273:857-864. doi:10.1056/NEJM196510142731606
- Mason V. Mystery outbreak investigation 2014—Leptospirosis licerasiae. November 17, 2017. Accessed March 5, 2026. https://usupulse.blogspot.com/2017/11/mystery-outbreak-investigation-2014.html
- Berjohn CM, DuPlessis CA, Tieu K, et al. Multibacillary leprosy in an active duty military member. Emerg Infect Dis. 2015;21:1077-1078. doi:10.3201/eid2106.141666
- Scollard DM, Adams LB, Gillis TP, et al. The continuing challenges of leprosy. Clin Microbiol Rev. 2006;19:338-381. doi:10.1128/CMR.19.2.338-381.2006
- Schär F, Trostdorf U, Giardina F, et al. Strongyloides stercoralis: global distribution and risk factors. PLoS Negl Trop Dis. 2013;7:E2288. doi:10.1371/journal.pntd.0002288
- Taheri B, Kuo HC, Hockenbury N, et al. Strongyloides stercoralis in the US Military Health System. Open Forum Infect Dis. 2023;10:ofad127. doi:10.1093/ofid/ofad127
- Bowman DD, Montgomery SP, Zajac AM, et al. Hookworms of dogs and cats as agents of cutaneous larva migrans. Trends Parasitol. 2010;26:162-167. doi:10.1016/j.pt.2010.01.005
- Inglis DM, Bailey MS. Unusual presentations of cutaneous larva migrans in British military personnel. BMJ Mil Health. 2023;169:E78-E81. doi:10.1136/bmjmilitary-2020-001677
- Halstead SB, Udomsakdi S, Singharaj P, et al. Dengue and chikungunya virus infection in man in Thailand, 1962–1964. 3. clinical, epidemiologic, and virologic observations on disease in non-indigenous white persons. Am J Trop Med Hyg. 1969;18:984-996. doi:10.4269/ajtmh.1969.18.984
- Gibbons RV, Streitz M, Babina T, et al. Dengue and US military operations from the Spanish-American War through today. Emerg Infect Dis. 2012;18:623-630. doi:10.3201/eid1804.110134
- Guzman MG, Harris E. Dengue. Lancet. 2015;385:453-465. doi:10.1016/S0140-6736(14)60572-9
- Bern C. Chagas’ disease. N Engl J Med. 2015;373:456-466. doi:10.1056/NEJMra1410150
- Vergara HD, Gómez CH, Faccini-Martínez ÁA, et al. Acute Chagas disease outbreak among military personnel, Colombia, 2021. Emerg Infect Dis. 2023;29:1882-1885. doi:10.3201/eid2909.230886
- Harris N, Woc-Colburn L, Gunter SM, et al. Autochthonous Chagas disease in the southern United States: a case report of suspected residential and military exposures. Zoonoses Public Health. 2017;64:491-493. doi:10.1111/zph.12360
- Crum NF. Coccidioidomycosis: a contemporary review. Infect Dis Ther. 2022;11:713-742. doi:10.1007/s40121-022-00606-y
- Crum NF, Potter M, Pappagianis D. Seroincidence of coccidioidomycosis during military desert training exercises. J Clin Microbiol. 2004;42:4552-4555. doi:10.1128/JCM.42.10.4552-4555.2004
- Antonello RM, Giacomelli A, Riccardi N. Tularemia for clinicians: an up-to-date review on epidemiology, diagnosis, prevention and treatment. Eur J Intern Med. 2025;135:25-32. doi:10.1016/j.ejim.2025.03.013
- Nelson CA, Sjöstedt A. Tularemia: a storied history, an ongoing threat. Clin Infect Dis. 2024;78(supplement_1):S1-S3. doi:10.1093/cid/ciad681
- Pendlebury GA, Roman J, Shrivastava V, et al. A call to action: evidence for the military integration of teledermoscopy in a pandemic era. Dermatopathology (Basel). 2022;9:327-342. doi:10.3390/dermatopathology9040039
- Bhagchandani R, Singhi S, Peter JV, et al. Tropical fevers: management guidelines. Indian J Crit Care Med. 2014;18:62-69. doi:10.4103/0972-5229.126074
- Joint Chiefs of Staff. Joint Publication 4-02: Joint Health Services. December 11, 2017. Accessed March 5, 2026. https://cdmrp.health.mil/pubs/pdf/Joint%20Health%20Services%20Publication%20JP%204-02.pdf
- Armed Services Pest Management Board. Technical Guide No. 36: Personal Protective Measures Against Insects and Other Arthropods of Military Significance. Updated November 2015. Accessed March 5, 2026. https://www.acq.osd.mil/eie/afpmb/docs/techguides/tg36.pdf
- Global Emerging Infections Surveillance. Armed Forces Health Surveillance Division Annual Report 2024. Defense Health Agency; 2024:15-28. Accessed March 17, 2026. https://www.health.mil/Reference-Center/Reports/2026/01/05/AFHSD-Annual-Report-2024
- Murray CK. Infectious disease complications of combat-related injuries. Crit Care Med. 2008;36(7 suppl):S358-S364. doi:10.1097/CCM.0b013e31817e2ffc
- Armed Forces Health Surveillance Division. AFHSD Annual Report. Defense Health Agency; 2023. Accessed March 5, 2026. https://www.health.mil/Reference-Center/Reports/2024/09/19/AFHSD-Annual-Report-2023
- Murray CK, Yun HC, Markelz AE, et al. Operation United Assistance: infectious disease threats to deployed military personnel. Military Medicine. 2015;180:626-651. doi:10.7205/MILMED-D-14-00691
- Niba Rawlings N, Bailey M, Courtenay O. Leishmaniasis in deployed military populations: a systematic review and meta-analysis. PLoS Negl Trop Dis. 2025;19:E0012680. doi:10.1371/journal.pntd.0012680
- de Vries HJC, Schallig HD. Cutaneous leishmaniasis: a 2022 updated narrative review into diagnosis and management developments. Am J Clin Dermatol. 2022;23:823-840. doi:10.1007/s40257-022-00726-8
- Anderson AD, Smoak B, Shuping E, et al. Q fever and the US military. Emerg Infect Dis. 2005;11:1320-1322. doi:10.3201/eid1108.050314
- Madariaga MG, Rezai K, Trenholme GM, et al. Q fever: a biological weapon in your backyard. Lancet Infect Dis. 2003;3:709-721. doi:10.1016/S1473-3099(03)00804-1
- Uribe Pulido N, Escorcia García C, Cabrera Orrego R, et al. Acute Q fever with dermatologic manifestations, molecular diagnosis, and no seroconversion. Open Forum Infect Dis. 2021;8:ofab458. doi:10.1093/ofid/ofab458
- Scott P, Deye G, Srinivasan A, et al. An outbreak of multidrug-resistant acinetobacter baumannii-calcoaceticus complex infection in the US Military Health Care System associated with military operations in Iraq. Clin Infect Dis. 2007;44:1577-1584. doi:10.1086/518170
- Guerrero DM, Perez F, Conger NG, et al. Acinetobacter baumannii-associated skin and soft tissue infections: recognizing a broadening spectrum of disease. Surg Infect (Larchmt). 2010;11:49-57. doi:10.1089/sur.2009.022
- Armed Forces Health Surveillance Division. Malaria among members of the US Armed Forces, 2024. MSMR. 2025;32:22-28.
- Farkouh CS, Abdi P, Amatul-Hadi F, et al. Cutaneous manifestations of malaria and their prognostic windows: a narrative review. Cureus. 2023;15:E41706. doi:10.7759/cureus.41706
- Shahbodaghi SD, Rathjen NA. Malaria: prevention, diagnosis, and treatment. Am Fam Physician. 2022;106:270-278.
- Yotsu RR, Suzuki K, Simmonds RE, et al. Buruli ulcer: a review of the current knowledge. Curr Trop Med Rep. 2018;5:247-256. doi:10.1007/s40475-018-0166-2
- Portaels F, Meyers WM, Ablordey A, et al. First cultivation and characterization of Mycobacterium ulcerans from the environment. PLoS Negl Trop Dis. 2008;2:E178. doi:10.1371/journal.pntd.0000178
- Metcalf-Kelly M, Garrison M, Stidham R. Characteristics of mpox cases diagnosed in Military Health System beneficiaries, May 2022-April 2024. MSMR. 2024;31:7-11.
- Rajapakse S. Leptospirosis: clinical aspects. Clin Med (Lond). 2022;22:14-17. doi:10.7861/clinmed.2021-0784
- Heath CW, Alexander AD, Galton MM. Leptospirosis in the United States: a of 483 cases in man, 1949–1961. N Engl J Med. 1965;273:857-864. doi:10.1056/NEJM196510142731606
- Mason V. Mystery outbreak investigation 2014—Leptospirosis licerasiae. November 17, 2017. Accessed March 5, 2026. https://usupulse.blogspot.com/2017/11/mystery-outbreak-investigation-2014.html
- Berjohn CM, DuPlessis CA, Tieu K, et al. Multibacillary leprosy in an active duty military member. Emerg Infect Dis. 2015;21:1077-1078. doi:10.3201/eid2106.141666
- Scollard DM, Adams LB, Gillis TP, et al. The continuing challenges of leprosy. Clin Microbiol Rev. 2006;19:338-381. doi:10.1128/CMR.19.2.338-381.2006
- Schär F, Trostdorf U, Giardina F, et al. Strongyloides stercoralis: global distribution and risk factors. PLoS Negl Trop Dis. 2013;7:E2288. doi:10.1371/journal.pntd.0002288
- Taheri B, Kuo HC, Hockenbury N, et al. Strongyloides stercoralis in the US Military Health System. Open Forum Infect Dis. 2023;10:ofad127. doi:10.1093/ofid/ofad127
- Bowman DD, Montgomery SP, Zajac AM, et al. Hookworms of dogs and cats as agents of cutaneous larva migrans. Trends Parasitol. 2010;26:162-167. doi:10.1016/j.pt.2010.01.005
- Inglis DM, Bailey MS. Unusual presentations of cutaneous larva migrans in British military personnel. BMJ Mil Health. 2023;169:E78-E81. doi:10.1136/bmjmilitary-2020-001677
- Halstead SB, Udomsakdi S, Singharaj P, et al. Dengue and chikungunya virus infection in man in Thailand, 1962–1964. 3. clinical, epidemiologic, and virologic observations on disease in non-indigenous white persons. Am J Trop Med Hyg. 1969;18:984-996. doi:10.4269/ajtmh.1969.18.984
- Gibbons RV, Streitz M, Babina T, et al. Dengue and US military operations from the Spanish-American War through today. Emerg Infect Dis. 2012;18:623-630. doi:10.3201/eid1804.110134
- Guzman MG, Harris E. Dengue. Lancet. 2015;385:453-465. doi:10.1016/S0140-6736(14)60572-9
- Bern C. Chagas’ disease. N Engl J Med. 2015;373:456-466. doi:10.1056/NEJMra1410150
- Vergara HD, Gómez CH, Faccini-Martínez ÁA, et al. Acute Chagas disease outbreak among military personnel, Colombia, 2021. Emerg Infect Dis. 2023;29:1882-1885. doi:10.3201/eid2909.230886
- Harris N, Woc-Colburn L, Gunter SM, et al. Autochthonous Chagas disease in the southern United States: a case report of suspected residential and military exposures. Zoonoses Public Health. 2017;64:491-493. doi:10.1111/zph.12360
- Crum NF. Coccidioidomycosis: a contemporary review. Infect Dis Ther. 2022;11:713-742. doi:10.1007/s40121-022-00606-y
- Crum NF, Potter M, Pappagianis D. Seroincidence of coccidioidomycosis during military desert training exercises. J Clin Microbiol. 2004;42:4552-4555. doi:10.1128/JCM.42.10.4552-4555.2004
- Antonello RM, Giacomelli A, Riccardi N. Tularemia for clinicians: an up-to-date review on epidemiology, diagnosis, prevention and treatment. Eur J Intern Med. 2025;135:25-32. doi:10.1016/j.ejim.2025.03.013
- Nelson CA, Sjöstedt A. Tularemia: a storied history, an ongoing threat. Clin Infect Dis. 2024;78(supplement_1):S1-S3. doi:10.1093/cid/ciad681
- Pendlebury GA, Roman J, Shrivastava V, et al. A call to action: evidence for the military integration of teledermoscopy in a pandemic era. Dermatopathology (Basel). 2022;9:327-342. doi:10.3390/dermatopathology9040039
- Bhagchandani R, Singhi S, Peter JV, et al. Tropical fevers: management guidelines. Indian J Crit Care Med. 2014;18:62-69. doi:10.4103/0972-5229.126074
- Joint Chiefs of Staff. Joint Publication 4-02: Joint Health Services. December 11, 2017. Accessed March 5, 2026. https://cdmrp.health.mil/pubs/pdf/Joint%20Health%20Services%20Publication%20JP%204-02.pdf
- Armed Services Pest Management Board. Technical Guide No. 36: Personal Protective Measures Against Insects and Other Arthropods of Military Significance. Updated November 2015. Accessed March 5, 2026. https://www.acq.osd.mil/eie/afpmb/docs/techguides/tg36.pdf
- Global Emerging Infections Surveillance. Armed Forces Health Surveillance Division Annual Report 2024. Defense Health Agency; 2024:15-28. Accessed March 17, 2026. https://www.health.mil/Reference-Center/Reports/2026/01/05/AFHSD-Annual-Report-2024
Cutaneous Manifestations of Neglected Infectious Diseases in US Military Personnel
Cutaneous Manifestations of Neglected Infectious Diseases in US Military Personnel
Practice Points
- Military personnel stationed overseas are at risk for encountering infectious organisms that are not regularly observed domestically—many of which have cutaneous manifestations.
- Health care professionals treating military personnel should consider uncommonly encountered infections in the differential diagnosis for certain dermatologic presentations.
- Clinicians should inquire if patients have been stationed in Coccidioides immitis–endemic areas prior to the initiation of immunosuppression.
Solitary Papule on the Upper Back
Solitary Papule on the Upper Back
THE DIAGNOSIS: Plexiform Palisaded Encapsulated Neuroma
Microscopically, there was a superficial to deep dermal proliferation of tapered spindle cells in fascicles that were well circumscribed in nodules throughout the dermis with pale background stroma, mild mucin, and a thin capsule. The tapered spindle cells stained positive for SOX-10 and negative for Melan-A (Figure 1A). Staining for epithelial membrane antigen highlighted delicate cells around the periphery of the nodules, consistent with perineurium (Figure 1B). A diagnosis of plexiform palisaded encapsulated neuroma was made. No additional treatment was pursued due to the benign nature of the condition.
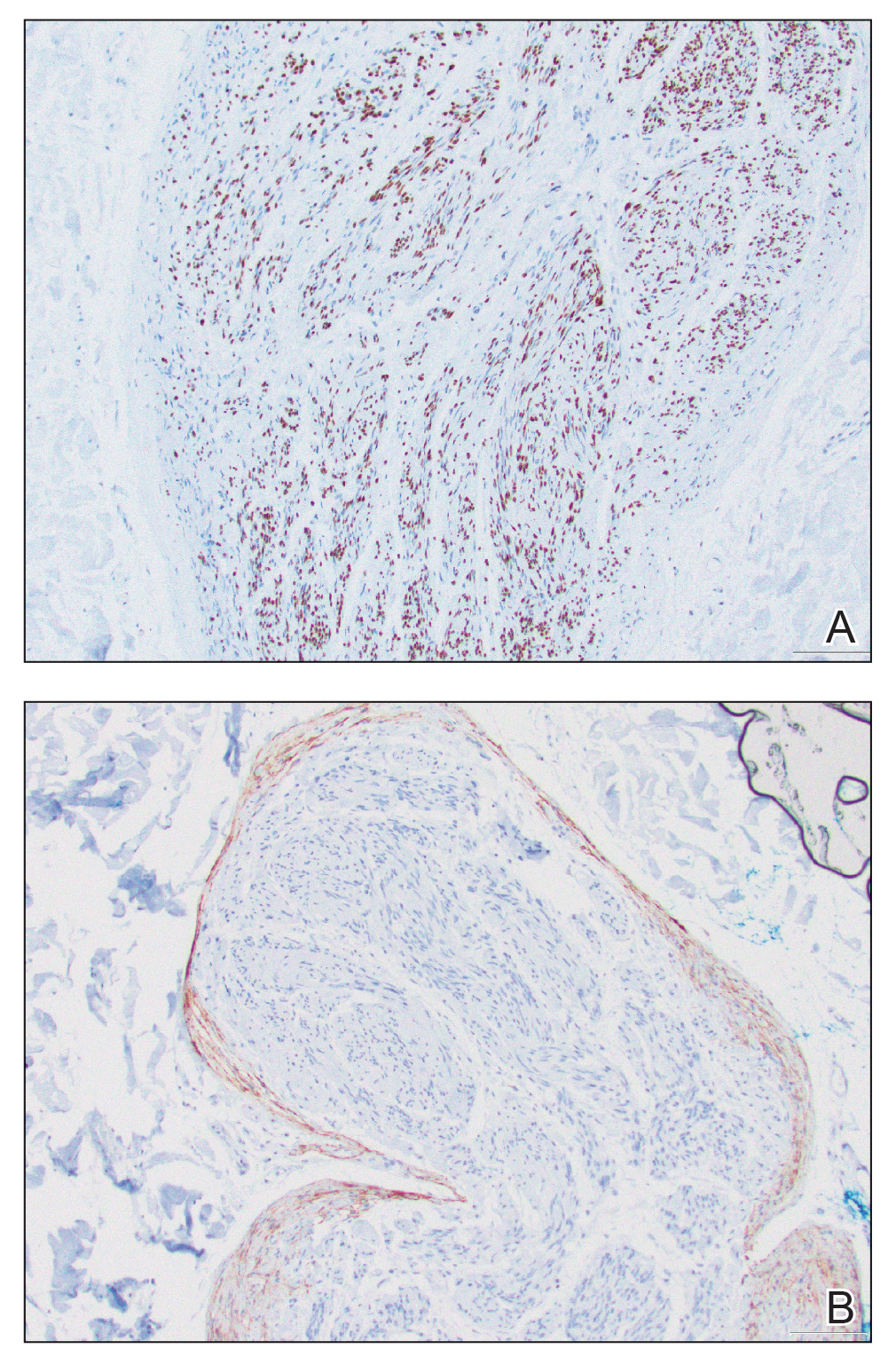
Palisaded encapsulated neuroma (PEN), also referred to as solitary circumscribed neuroma,1 is a benign, generally solitary neurogenic tumor that manifests predominantly on the skin, particularly in areas of frequent outside trauma such as the face. Lesions also may occur on mucosal and acral sites.2 First described by Reed et al3 in 1972, PEN characteristically manifests as a well-circumscribed, dermal nodule with a distinctive palisading pattern of Schwann cells and axons within a delicate perineurial capsule, the latter of which may be incomplete.3 Palisaded encapsulated neuroma frequently exhibits clefting between the tumor and the surrounding dermis. While PEN generally is sporadic, rare cases have been reported in association with Cowden syndrome and neurofibromatosis type 2.4,5
While the nodular growth pattern is most common, PEN also may present in epithelioid, plexiform, multinodular, or fungating subtypes.6 The plexiform subtype of PEN is rare. It has a complex growth pattern and a tendency to involve multiple adjacent nerve bundles in a plexiform arrangement.6,7 In two independent reviews characterizing the predominant growth patterns of PEN, nonnodular growth patterns were observed in a minority of the 85 cases: 18.8% (16/85) were plexiform, 7.1% (6/85) were multinodular or multilobular, and 7.1% (6/85) were fungating.6,7
The clinical presentation of plexiform PEN often includes a painless, slow-growing mass, and it predominantly occurs in middle-aged adults.2 Immunohistochemical staining reveals diffuse positivity for SOX-10 and S-100, which highlights the neural origin of the tumor.6 This variant, like conventional PEN, lacks notable atypia or mitotic activity.
Palisaded encapsulated neuroma, regardless of subtype, has an excellent prognosis, with no known cases of malignant transformation, and surgical excision with clear margins is curative.8
The differential diagnosis for plexiform PEN includes plexiform variants of neurofibroma and schwannoma, traumatic neuroma, and malignant peripheral nerve sheath tumor.
Neurofibromas are nonencapsulated lesions composed of spindle cells with wavy nuclei dispersed in a myxoid background.8 Neurofibromas can manifest in various locations throughout the body, including the skin, subcutaneous tissues, and internal organs. They are slow-growing tumors but may accelerate during periods of hormonal changes, such as pregnancy and puberty, or in cases of malignant transformation.8 Although plexiform neurofibromas are benign, malignant transformation can occur, particularly in patients with neurofibromatosis type 1 (NF1).8,9 Neurofibromas may assume one of 3 growth patterns: localized, diffuse, or plexiform.8 Plexiform neurofibromas exhibit a multinodular, ropelike growth pattern with a mix of Schwann cells and fibroblasts (Figure 2).8,9 These lesions are pathognomonic for NF1 and can infiltrate the surrounding tissue. They may involve large nerve trunks, leading to a more complex growth pattern compared to solitary neurofibromas.8,9 The plexiform variants of both neurofibromas and PEN demonstrate a multinodular growth pattern; however, plexiform neurofibromas are nonencapsulated and show a more diffuse infiltrative nature, whereas plexiform PEN remains well circumscribed. Additionally, plexiform neurofibromas are associated with NF1, while plexiform PEN lacks this genetic association.
Schwannomas are encapsulated tumors that originate from the outer sheath of peripheral nerves, usually positioned eccentrically to the nerve fibers. Schwannomas are characterized by Antoni A and Antoni B areas, which usually are absent in PEN. Antoni A areas are composed of compact spindle cells arranged in palisades with Verocay bodies, while Antoni B areas are more loosely arranged and have a myxoid background (Figure 3).8,9 Schwannomas stain positive for S-100 and often show degenerative changes such as cystic degeneration or calcification, particularly in larger lesions.8,9 Plexiform schwannoma is a rare variant of schwannoma, and while it carries a substantial risk for local recurrence with rates as high as 50%, it has not been shown to possess malignant or metastatic potential.10 Unlike PEN, schwannomas have a consistent capsule but share S-100 positivity with PEN. Verocay bodies occasionally can be observed in PENs, with studies reporting their presence in 20% to 36% of cases.7,11,12 Additionally, some schwannomas may exhibit few Verocay bodies or poorly developed forms, which can make histopathologic distinction more challenging.7,11,12
Traumatic neuromas result from nerve regeneration following any type of outside trauma that is deep enough to cause nerve injury. The lesion often is painful and associated with prior trauma or surgery. Under optimal conditions, the severed ends of a nerve reconnect through the orderly growth of axons from the proximal stump to the distal stump, guided by tubes formed by proliferating Schwann cells. If the nerve ends are not properly aligned or if the distal stump is absent, the axons may proliferate in a disorganized manner at the proximal stump, resulting in the formation of a traumatic neuroma.8 Histologically, these lesions exhibit disorganized, proliferating nerve fibers intermixed with fibrous stroma.8,13 The nerve fibers are not encapsulated, and there is an irregular arrangement of axons and Schwann cells (Figure 4).8,13 Unlike PEN, which usually is encapsulated and well organized with fascicular architecture, traumatic neuromas exhibit a disorganized, haphazard arrangement of neural elements and lack a capsule.8 Clinically, traumatic neuromas also are more likely to be painful.
Malignant peripheral nerve sheath tumors are aggressive malignant spindle-cell tumors that may be associated with NF1 or occur sporadically.9,14 The spindle cells are arranged in fascicles, and these tumors can have areas of necrosis, hemorrhage, and high mitotic activity.9,15 The spindle cells may be arranged in a herringbone pattern, and alternating areas of hypocellularity and hypercellularity impart a marbled appearance (Figure 5).16 Malignant peripheral nerve sheath tumors frequently exhibit inactivation of the SWI/SNF-related, matrix- associated, actin-dependent regulator of chromatin subfamily B member 1 gene and loss of integrase interactor 1 protein. Transformation from plexiform neurofibroma to malignant peripheral nerve sheath tumor frequently is accompanied by progressive genomic changes.17 Malignant peripheral nerve sheath tumors differ substantially from PEN in their aggressive histologic features, including nuclear atypia and mitotic figures, which are absent in PEN.
- Fletcher CD. Solitary circumscribed neuroma of the skin (so-called palisaded, encapsulated neuroma). a clinicopathologic and immunohistochemical study. Am J Surg Pathol. 1989;13:574-580. doi:10.1097/00000478-198907000-00005
- Jokinen CH, Ragsdale BD, Argenyi ZB. Expanding the clinicopathologic spectrum of palisaded encapsulated neuroma. J Cutan Pathol. 2010;37:43-48. doi:10.1111/j.1600-0560.2009.01380.x
- Reed RJ, Fine RM, Meltzer HD. Palisaded encapsulated neuromas of the skin. Arch Dermatol. 1972;106:865-870.
- Harris E, Mir A. Acral plexiform palisaded encapsulated neuromas as the initial cutaneous manifestation of Cowden syndrome. Pediatr Dermatol. 2017;34:E219-E220. doi:10.1111/pde.13161
- Arole V, Shaker N, Kim LR, et al. Multiple cutaneous solitary circumscribed neuroma in a patient with neurofibromatosis type 2: an “incidentaloma” or new association?. Int J Surg Pathol. 2023;31:734-737. doi:10.1177/10668969221120782
- Argenyi ZB, Cooper PH, Cruz DS. Plexiform and other unusual variants of palisaded encapsulated neuroma. J Cutan Pathol. 1993;20:34-39. doi:10.1111/j.1600-0560.1993.tb01246.x
- Leblebici C, Savli TC, Yeni B, et al. Palisaded encapsulated (solitary circumscribed) neuroma: a review of 30 cases. Int J Surg Pathol. 2019;27:506-514. doi:10.1177/1066896919833172
- Weiss SW, Goldblum JR. Enzinger and Weiss’s Soft Tissue Tumors. 6th ed. Elsevier Saunders; 2014.
- Rodriguez FJ, Folpe AL, Giannini C, et al. Pathology of peripheral nerve sheath tumors: diagnostic overview and update on selected diagnostic problems. Acta Neuropathol. 2012;123:295-319. doi:10.1007 /s00401-012-0954-z
- Berg JC, Scheithauer BW, Spinner RJ, et al. Plexiform schwannoma: a clinicopathologic overview with emphasis on the head and neck region. Hum Pathol. 2008;39:633-640. doi:10.1016 /j.humpath.2007.10.029
- Koutlas IG, Scheithauer BW. Palisaded encapsulated (“solitary circumscribed”) neuroma of the oral cavity: a review of 55 cases. Head Neck Pathol. 2010;4:15-26. doi:10.1007/s12105-010-0162-x
- Kossard S, Kumar A, Wilkinson B. Neural spectrum: palisaded encapsulated neuroma and verocay body poor dermal schwannoma. J Cutan Pathol. 1999;26:31-36. doi:10.1111/j.1600-0560.1999 .tb01787.x
- Yang H, Dong Y, Wang Z, et al. Traumatic neuromas of peripheral nerves: diagnosis, management and future perspectives. Front Neurol. 2023;13:1039529. doi:10.3389/fneur.2022.1039529
- Knight SWE, Knight TE, Santiago T, et al. Malignant peripheral nerve sheath tumors-a comprehensive review of pathophysiology, diagnosis, and multidisciplinary management. Children (Basel). 2022;9:38. doi:10.3390/children9010038
- Perry A, Gutmann DH. Malignant peripheral nerve sheath tumors: clinical and genetic aspects of pathogenesis. Clin Neuropathol. 2000;19:105-114.
- Lindberg G, Lucas D, Cassarino D, et al, eds. Diagnostic Pathology: Soft Tissue Tumors. 3rd ed. Elsevier; 2023.
- Miettinen MM, Antonescu CR, Fletcher CDM, et al. Histopathologic evaluation of atypical neurofibromatous tumors and their transformation into malignant peripheral nerve sheath tumor in patients with neurofibromatosis 1-a consensus overview. Hum Pathol. 2017;67:1-10. doi:10.1016/j.humpath.2017.05.010
THE DIAGNOSIS: Plexiform Palisaded Encapsulated Neuroma
Microscopically, there was a superficial to deep dermal proliferation of tapered spindle cells in fascicles that were well circumscribed in nodules throughout the dermis with pale background stroma, mild mucin, and a thin capsule. The tapered spindle cells stained positive for SOX-10 and negative for Melan-A (Figure 1A). Staining for epithelial membrane antigen highlighted delicate cells around the periphery of the nodules, consistent with perineurium (Figure 1B). A diagnosis of plexiform palisaded encapsulated neuroma was made. No additional treatment was pursued due to the benign nature of the condition.

Palisaded encapsulated neuroma (PEN), also referred to as solitary circumscribed neuroma,1 is a benign, generally solitary neurogenic tumor that manifests predominantly on the skin, particularly in areas of frequent outside trauma such as the face. Lesions also may occur on mucosal and acral sites.2 First described by Reed et al3 in 1972, PEN characteristically manifests as a well-circumscribed, dermal nodule with a distinctive palisading pattern of Schwann cells and axons within a delicate perineurial capsule, the latter of which may be incomplete.3 Palisaded encapsulated neuroma frequently exhibits clefting between the tumor and the surrounding dermis. While PEN generally is sporadic, rare cases have been reported in association with Cowden syndrome and neurofibromatosis type 2.4,5
While the nodular growth pattern is most common, PEN also may present in epithelioid, plexiform, multinodular, or fungating subtypes.6 The plexiform subtype of PEN is rare. It has a complex growth pattern and a tendency to involve multiple adjacent nerve bundles in a plexiform arrangement.6,7 In two independent reviews characterizing the predominant growth patterns of PEN, nonnodular growth patterns were observed in a minority of the 85 cases: 18.8% (16/85) were plexiform, 7.1% (6/85) were multinodular or multilobular, and 7.1% (6/85) were fungating.6,7
The clinical presentation of plexiform PEN often includes a painless, slow-growing mass, and it predominantly occurs in middle-aged adults.2 Immunohistochemical staining reveals diffuse positivity for SOX-10 and S-100, which highlights the neural origin of the tumor.6 This variant, like conventional PEN, lacks notable atypia or mitotic activity.
Palisaded encapsulated neuroma, regardless of subtype, has an excellent prognosis, with no known cases of malignant transformation, and surgical excision with clear margins is curative.8
The differential diagnosis for plexiform PEN includes plexiform variants of neurofibroma and schwannoma, traumatic neuroma, and malignant peripheral nerve sheath tumor.
Neurofibromas are nonencapsulated lesions composed of spindle cells with wavy nuclei dispersed in a myxoid background.8 Neurofibromas can manifest in various locations throughout the body, including the skin, subcutaneous tissues, and internal organs. They are slow-growing tumors but may accelerate during periods of hormonal changes, such as pregnancy and puberty, or in cases of malignant transformation.8 Although plexiform neurofibromas are benign, malignant transformation can occur, particularly in patients with neurofibromatosis type 1 (NF1).8,9 Neurofibromas may assume one of 3 growth patterns: localized, diffuse, or plexiform.8 Plexiform neurofibromas exhibit a multinodular, ropelike growth pattern with a mix of Schwann cells and fibroblasts (Figure 2).8,9 These lesions are pathognomonic for NF1 and can infiltrate the surrounding tissue. They may involve large nerve trunks, leading to a more complex growth pattern compared to solitary neurofibromas.8,9 The plexiform variants of both neurofibromas and PEN demonstrate a multinodular growth pattern; however, plexiform neurofibromas are nonencapsulated and show a more diffuse infiltrative nature, whereas plexiform PEN remains well circumscribed. Additionally, plexiform neurofibromas are associated with NF1, while plexiform PEN lacks this genetic association.

Schwannomas are encapsulated tumors that originate from the outer sheath of peripheral nerves, usually positioned eccentrically to the nerve fibers. Schwannomas are characterized by Antoni A and Antoni B areas, which usually are absent in PEN. Antoni A areas are composed of compact spindle cells arranged in palisades with Verocay bodies, while Antoni B areas are more loosely arranged and have a myxoid background (Figure 3).8,9 Schwannomas stain positive for S-100 and often show degenerative changes such as cystic degeneration or calcification, particularly in larger lesions.8,9 Plexiform schwannoma is a rare variant of schwannoma, and while it carries a substantial risk for local recurrence with rates as high as 50%, it has not been shown to possess malignant or metastatic potential.10 Unlike PEN, schwannomas have a consistent capsule but share S-100 positivity with PEN. Verocay bodies occasionally can be observed in PENs, with studies reporting their presence in 20% to 36% of cases.7,11,12 Additionally, some schwannomas may exhibit few Verocay bodies or poorly developed forms, which can make histopathologic distinction more challenging.7,11,12

Traumatic neuromas result from nerve regeneration following any type of outside trauma that is deep enough to cause nerve injury. The lesion often is painful and associated with prior trauma or surgery. Under optimal conditions, the severed ends of a nerve reconnect through the orderly growth of axons from the proximal stump to the distal stump, guided by tubes formed by proliferating Schwann cells. If the nerve ends are not properly aligned or if the distal stump is absent, the axons may proliferate in a disorganized manner at the proximal stump, resulting in the formation of a traumatic neuroma.8 Histologically, these lesions exhibit disorganized, proliferating nerve fibers intermixed with fibrous stroma.8,13 The nerve fibers are not encapsulated, and there is an irregular arrangement of axons and Schwann cells (Figure 4).8,13 Unlike PEN, which usually is encapsulated and well organized with fascicular architecture, traumatic neuromas exhibit a disorganized, haphazard arrangement of neural elements and lack a capsule.8 Clinically, traumatic neuromas also are more likely to be painful.

Malignant peripheral nerve sheath tumors are aggressive malignant spindle-cell tumors that may be associated with NF1 or occur sporadically.9,14 The spindle cells are arranged in fascicles, and these tumors can have areas of necrosis, hemorrhage, and high mitotic activity.9,15 The spindle cells may be arranged in a herringbone pattern, and alternating areas of hypocellularity and hypercellularity impart a marbled appearance (Figure 5).16 Malignant peripheral nerve sheath tumors frequently exhibit inactivation of the SWI/SNF-related, matrix- associated, actin-dependent regulator of chromatin subfamily B member 1 gene and loss of integrase interactor 1 protein. Transformation from plexiform neurofibroma to malignant peripheral nerve sheath tumor frequently is accompanied by progressive genomic changes.17 Malignant peripheral nerve sheath tumors differ substantially from PEN in their aggressive histologic features, including nuclear atypia and mitotic figures, which are absent in PEN.

THE DIAGNOSIS: Plexiform Palisaded Encapsulated Neuroma
Microscopically, there was a superficial to deep dermal proliferation of tapered spindle cells in fascicles that were well circumscribed in nodules throughout the dermis with pale background stroma, mild mucin, and a thin capsule. The tapered spindle cells stained positive for SOX-10 and negative for Melan-A (Figure 1A). Staining for epithelial membrane antigen highlighted delicate cells around the periphery of the nodules, consistent with perineurium (Figure 1B). A diagnosis of plexiform palisaded encapsulated neuroma was made. No additional treatment was pursued due to the benign nature of the condition.

Palisaded encapsulated neuroma (PEN), also referred to as solitary circumscribed neuroma,1 is a benign, generally solitary neurogenic tumor that manifests predominantly on the skin, particularly in areas of frequent outside trauma such as the face. Lesions also may occur on mucosal and acral sites.2 First described by Reed et al3 in 1972, PEN characteristically manifests as a well-circumscribed, dermal nodule with a distinctive palisading pattern of Schwann cells and axons within a delicate perineurial capsule, the latter of which may be incomplete.3 Palisaded encapsulated neuroma frequently exhibits clefting between the tumor and the surrounding dermis. While PEN generally is sporadic, rare cases have been reported in association with Cowden syndrome and neurofibromatosis type 2.4,5
While the nodular growth pattern is most common, PEN also may present in epithelioid, plexiform, multinodular, or fungating subtypes.6 The plexiform subtype of PEN is rare. It has a complex growth pattern and a tendency to involve multiple adjacent nerve bundles in a plexiform arrangement.6,7 In two independent reviews characterizing the predominant growth patterns of PEN, nonnodular growth patterns were observed in a minority of the 85 cases: 18.8% (16/85) were plexiform, 7.1% (6/85) were multinodular or multilobular, and 7.1% (6/85) were fungating.6,7
The clinical presentation of plexiform PEN often includes a painless, slow-growing mass, and it predominantly occurs in middle-aged adults.2 Immunohistochemical staining reveals diffuse positivity for SOX-10 and S-100, which highlights the neural origin of the tumor.6 This variant, like conventional PEN, lacks notable atypia or mitotic activity.
Palisaded encapsulated neuroma, regardless of subtype, has an excellent prognosis, with no known cases of malignant transformation, and surgical excision with clear margins is curative.8
The differential diagnosis for plexiform PEN includes plexiform variants of neurofibroma and schwannoma, traumatic neuroma, and malignant peripheral nerve sheath tumor.
Neurofibromas are nonencapsulated lesions composed of spindle cells with wavy nuclei dispersed in a myxoid background.8 Neurofibromas can manifest in various locations throughout the body, including the skin, subcutaneous tissues, and internal organs. They are slow-growing tumors but may accelerate during periods of hormonal changes, such as pregnancy and puberty, or in cases of malignant transformation.8 Although plexiform neurofibromas are benign, malignant transformation can occur, particularly in patients with neurofibromatosis type 1 (NF1).8,9 Neurofibromas may assume one of 3 growth patterns: localized, diffuse, or plexiform.8 Plexiform neurofibromas exhibit a multinodular, ropelike growth pattern with a mix of Schwann cells and fibroblasts (Figure 2).8,9 These lesions are pathognomonic for NF1 and can infiltrate the surrounding tissue. They may involve large nerve trunks, leading to a more complex growth pattern compared to solitary neurofibromas.8,9 The plexiform variants of both neurofibromas and PEN demonstrate a multinodular growth pattern; however, plexiform neurofibromas are nonencapsulated and show a more diffuse infiltrative nature, whereas plexiform PEN remains well circumscribed. Additionally, plexiform neurofibromas are associated with NF1, while plexiform PEN lacks this genetic association.

Schwannomas are encapsulated tumors that originate from the outer sheath of peripheral nerves, usually positioned eccentrically to the nerve fibers. Schwannomas are characterized by Antoni A and Antoni B areas, which usually are absent in PEN. Antoni A areas are composed of compact spindle cells arranged in palisades with Verocay bodies, while Antoni B areas are more loosely arranged and have a myxoid background (Figure 3).8,9 Schwannomas stain positive for S-100 and often show degenerative changes such as cystic degeneration or calcification, particularly in larger lesions.8,9 Plexiform schwannoma is a rare variant of schwannoma, and while it carries a substantial risk for local recurrence with rates as high as 50%, it has not been shown to possess malignant or metastatic potential.10 Unlike PEN, schwannomas have a consistent capsule but share S-100 positivity with PEN. Verocay bodies occasionally can be observed in PENs, with studies reporting their presence in 20% to 36% of cases.7,11,12 Additionally, some schwannomas may exhibit few Verocay bodies or poorly developed forms, which can make histopathologic distinction more challenging.7,11,12

Traumatic neuromas result from nerve regeneration following any type of outside trauma that is deep enough to cause nerve injury. The lesion often is painful and associated with prior trauma or surgery. Under optimal conditions, the severed ends of a nerve reconnect through the orderly growth of axons from the proximal stump to the distal stump, guided by tubes formed by proliferating Schwann cells. If the nerve ends are not properly aligned or if the distal stump is absent, the axons may proliferate in a disorganized manner at the proximal stump, resulting in the formation of a traumatic neuroma.8 Histologically, these lesions exhibit disorganized, proliferating nerve fibers intermixed with fibrous stroma.8,13 The nerve fibers are not encapsulated, and there is an irregular arrangement of axons and Schwann cells (Figure 4).8,13 Unlike PEN, which usually is encapsulated and well organized with fascicular architecture, traumatic neuromas exhibit a disorganized, haphazard arrangement of neural elements and lack a capsule.8 Clinically, traumatic neuromas also are more likely to be painful.

Malignant peripheral nerve sheath tumors are aggressive malignant spindle-cell tumors that may be associated with NF1 or occur sporadically.9,14 The spindle cells are arranged in fascicles, and these tumors can have areas of necrosis, hemorrhage, and high mitotic activity.9,15 The spindle cells may be arranged in a herringbone pattern, and alternating areas of hypocellularity and hypercellularity impart a marbled appearance (Figure 5).16 Malignant peripheral nerve sheath tumors frequently exhibit inactivation of the SWI/SNF-related, matrix- associated, actin-dependent regulator of chromatin subfamily B member 1 gene and loss of integrase interactor 1 protein. Transformation from plexiform neurofibroma to malignant peripheral nerve sheath tumor frequently is accompanied by progressive genomic changes.17 Malignant peripheral nerve sheath tumors differ substantially from PEN in their aggressive histologic features, including nuclear atypia and mitotic figures, which are absent in PEN.

- Fletcher CD. Solitary circumscribed neuroma of the skin (so-called palisaded, encapsulated neuroma). a clinicopathologic and immunohistochemical study. Am J Surg Pathol. 1989;13:574-580. doi:10.1097/00000478-198907000-00005
- Jokinen CH, Ragsdale BD, Argenyi ZB. Expanding the clinicopathologic spectrum of palisaded encapsulated neuroma. J Cutan Pathol. 2010;37:43-48. doi:10.1111/j.1600-0560.2009.01380.x
- Reed RJ, Fine RM, Meltzer HD. Palisaded encapsulated neuromas of the skin. Arch Dermatol. 1972;106:865-870.
- Harris E, Mir A. Acral plexiform palisaded encapsulated neuromas as the initial cutaneous manifestation of Cowden syndrome. Pediatr Dermatol. 2017;34:E219-E220. doi:10.1111/pde.13161
- Arole V, Shaker N, Kim LR, et al. Multiple cutaneous solitary circumscribed neuroma in a patient with neurofibromatosis type 2: an “incidentaloma” or new association?. Int J Surg Pathol. 2023;31:734-737. doi:10.1177/10668969221120782
- Argenyi ZB, Cooper PH, Cruz DS. Plexiform and other unusual variants of palisaded encapsulated neuroma. J Cutan Pathol. 1993;20:34-39. doi:10.1111/j.1600-0560.1993.tb01246.x
- Leblebici C, Savli TC, Yeni B, et al. Palisaded encapsulated (solitary circumscribed) neuroma: a review of 30 cases. Int J Surg Pathol. 2019;27:506-514. doi:10.1177/1066896919833172
- Weiss SW, Goldblum JR. Enzinger and Weiss’s Soft Tissue Tumors. 6th ed. Elsevier Saunders; 2014.
- Rodriguez FJ, Folpe AL, Giannini C, et al. Pathology of peripheral nerve sheath tumors: diagnostic overview and update on selected diagnostic problems. Acta Neuropathol. 2012;123:295-319. doi:10.1007 /s00401-012-0954-z
- Berg JC, Scheithauer BW, Spinner RJ, et al. Plexiform schwannoma: a clinicopathologic overview with emphasis on the head and neck region. Hum Pathol. 2008;39:633-640. doi:10.1016 /j.humpath.2007.10.029
- Koutlas IG, Scheithauer BW. Palisaded encapsulated (“solitary circumscribed”) neuroma of the oral cavity: a review of 55 cases. Head Neck Pathol. 2010;4:15-26. doi:10.1007/s12105-010-0162-x
- Kossard S, Kumar A, Wilkinson B. Neural spectrum: palisaded encapsulated neuroma and verocay body poor dermal schwannoma. J Cutan Pathol. 1999;26:31-36. doi:10.1111/j.1600-0560.1999 .tb01787.x
- Yang H, Dong Y, Wang Z, et al. Traumatic neuromas of peripheral nerves: diagnosis, management and future perspectives. Front Neurol. 2023;13:1039529. doi:10.3389/fneur.2022.1039529
- Knight SWE, Knight TE, Santiago T, et al. Malignant peripheral nerve sheath tumors-a comprehensive review of pathophysiology, diagnosis, and multidisciplinary management. Children (Basel). 2022;9:38. doi:10.3390/children9010038
- Perry A, Gutmann DH. Malignant peripheral nerve sheath tumors: clinical and genetic aspects of pathogenesis. Clin Neuropathol. 2000;19:105-114.
- Lindberg G, Lucas D, Cassarino D, et al, eds. Diagnostic Pathology: Soft Tissue Tumors. 3rd ed. Elsevier; 2023.
- Miettinen MM, Antonescu CR, Fletcher CDM, et al. Histopathologic evaluation of atypical neurofibromatous tumors and their transformation into malignant peripheral nerve sheath tumor in patients with neurofibromatosis 1-a consensus overview. Hum Pathol. 2017;67:1-10. doi:10.1016/j.humpath.2017.05.010
- Fletcher CD. Solitary circumscribed neuroma of the skin (so-called palisaded, encapsulated neuroma). a clinicopathologic and immunohistochemical study. Am J Surg Pathol. 1989;13:574-580. doi:10.1097/00000478-198907000-00005
- Jokinen CH, Ragsdale BD, Argenyi ZB. Expanding the clinicopathologic spectrum of palisaded encapsulated neuroma. J Cutan Pathol. 2010;37:43-48. doi:10.1111/j.1600-0560.2009.01380.x
- Reed RJ, Fine RM, Meltzer HD. Palisaded encapsulated neuromas of the skin. Arch Dermatol. 1972;106:865-870.
- Harris E, Mir A. Acral plexiform palisaded encapsulated neuromas as the initial cutaneous manifestation of Cowden syndrome. Pediatr Dermatol. 2017;34:E219-E220. doi:10.1111/pde.13161
- Arole V, Shaker N, Kim LR, et al. Multiple cutaneous solitary circumscribed neuroma in a patient with neurofibromatosis type 2: an “incidentaloma” or new association?. Int J Surg Pathol. 2023;31:734-737. doi:10.1177/10668969221120782
- Argenyi ZB, Cooper PH, Cruz DS. Plexiform and other unusual variants of palisaded encapsulated neuroma. J Cutan Pathol. 1993;20:34-39. doi:10.1111/j.1600-0560.1993.tb01246.x
- Leblebici C, Savli TC, Yeni B, et al. Palisaded encapsulated (solitary circumscribed) neuroma: a review of 30 cases. Int J Surg Pathol. 2019;27:506-514. doi:10.1177/1066896919833172
- Weiss SW, Goldblum JR. Enzinger and Weiss’s Soft Tissue Tumors. 6th ed. Elsevier Saunders; 2014.
- Rodriguez FJ, Folpe AL, Giannini C, et al. Pathology of peripheral nerve sheath tumors: diagnostic overview and update on selected diagnostic problems. Acta Neuropathol. 2012;123:295-319. doi:10.1007 /s00401-012-0954-z
- Berg JC, Scheithauer BW, Spinner RJ, et al. Plexiform schwannoma: a clinicopathologic overview with emphasis on the head and neck region. Hum Pathol. 2008;39:633-640. doi:10.1016 /j.humpath.2007.10.029
- Koutlas IG, Scheithauer BW. Palisaded encapsulated (“solitary circumscribed”) neuroma of the oral cavity: a review of 55 cases. Head Neck Pathol. 2010;4:15-26. doi:10.1007/s12105-010-0162-x
- Kossard S, Kumar A, Wilkinson B. Neural spectrum: palisaded encapsulated neuroma and verocay body poor dermal schwannoma. J Cutan Pathol. 1999;26:31-36. doi:10.1111/j.1600-0560.1999 .tb01787.x
- Yang H, Dong Y, Wang Z, et al. Traumatic neuromas of peripheral nerves: diagnosis, management and future perspectives. Front Neurol. 2023;13:1039529. doi:10.3389/fneur.2022.1039529
- Knight SWE, Knight TE, Santiago T, et al. Malignant peripheral nerve sheath tumors-a comprehensive review of pathophysiology, diagnosis, and multidisciplinary management. Children (Basel). 2022;9:38. doi:10.3390/children9010038
- Perry A, Gutmann DH. Malignant peripheral nerve sheath tumors: clinical and genetic aspects of pathogenesis. Clin Neuropathol. 2000;19:105-114.
- Lindberg G, Lucas D, Cassarino D, et al, eds. Diagnostic Pathology: Soft Tissue Tumors. 3rd ed. Elsevier; 2023.
- Miettinen MM, Antonescu CR, Fletcher CDM, et al. Histopathologic evaluation of atypical neurofibromatous tumors and their transformation into malignant peripheral nerve sheath tumor in patients with neurofibromatosis 1-a consensus overview. Hum Pathol. 2017;67:1-10. doi:10.1016/j.humpath.2017.05.010
Solitary Papule on the Upper Back
Solitary Papule on the Upper Back
An 88-year-old woman presented to the dermatology clinic with an asymptomatic papule on the left upper back of unknown duration. The patient reported that her medical history was negative for eczematous dermatitis, hypertension, and osteoarthritis. Physical examination revealed a firm, well-circumscribed, flesh-colored, 6-mm papule with no overlying scale or ulceration. No other concerning lesions were noted on full skin examination. A punch biopsy of the papule was performed.
Dupilumab for Bullous Pemphigoid: To Treat or Not to Treat?
Dupilumab for Bullous Pemphigoid: To Treat or Not to Treat?
Bullous pemphigoid (BP) is a chronic autoimmune skin condition characterized by large, fluid-filled blisters (bullae) on the skin. There usually is severe itching, which may antedate the bullae by several weeks. Bullous pemphigoid most commonly affects adults aged 60 years and older with comorbidities such as metabolic syndrome or other autoimmune diseases. The typical blistering is subepidermal, and its occurrence seems to be driven by autoantibodies targeting the epidermal basement membrane zone.
Conventional treatment for BP has long included systemic corticosteroids and immunosuppressants; this combination of drugs effectively controls inflammation but also is associated with considerable risks such as infection, hyperglycemia with worsening diabetes, hypertension, osteoporosis and fracture risk, and delirium, particularly in older patients. Recent developments in biologic therapy have supported the utilization of potentially safer alternatives. Specifically, dupilumab has demonstrated efficacy in the treatment of atopic dermatitis. Case reports and small research studies included in a recent review have suggested that dupilumab could be effective in managing BP, potentially offering a safer treatment option.1 Importantly, dupilumab recently was approved for BP by the US Food and Drug Administration, making it the first biologic approved for treating this condition.2 In this article, we discuss dupilumab as an option for clinicians to prescribe as an initial therapy for BP.
Pathophysiology of Bullous Pemphigoid and Dupilumab
Bullous pemphigoid pathophysiology is driven by 2 main components: immunologic and inflammatory processes. The immune-mediated attack consists of autoantibodies against the hemidesmosomal proteins BP180 and BP230 in the basement membrane zone, which anchor the epidermis to the dermis.1 When these proteins are targeted, a cascade of responses is triggered, including the release of inflammatory cells, particularly eosinophils and neutrophils, into the skin. These cells release inflammatory mediators that break down the basement membrane, resulting in the separation of the epidermis from the dermis and leading to subepidermal blister formation.
The inflammation associated with BP largely is driven by a T helper 2–dominant immune response, with increased levels of cytokines such as interleukin (IL) 4 and IL-13.1 The chronic inflammation contributes to the characteristic blisters and intense pruritus seen in patients with BP. Traditional treatments such as corticosteroids work broadly to suppress this inflammation by reducing the production of all proinflammatory cytokines; however, they lack specificity, which dampens the targeted immune response against BP and broadly suppresses the immune system, increasing the risk for infection and other adverse effects. For older patients with multiple health conditions, these adverse effects can impact quality of life and pose health risks.
Interleukin 4 and IL-13 contribute to eosinophilia by upregulating the expression of eosinophil chemoattractant cytokines that promote eosinophil migration and adhesion.1 This is where dupilumab presents a promising alternative. Dupilumab is a monoclonal antibody that selectively targets the key cytokines implicated in BP. It binds to the IL-4 receptor alpha subunit, which IL-4 and IL-13 share, thereby inhibiting the signaling of both cytokines.1
Dupilumab Reports and Case Series
Current evidence supporting the use of dupilumab for BP consists primarily of case series and uncontrolled clinical observations, both of which are useful to formulate but not to test hypotheses.3 One case series of 13 patients diagnosed with BP reported that 92.3% (12/13) of patients achieved disease clearance or satisfactory control, with 53.8% (7/13) achieving total lesion clearance and no adverse events observed.4 In a larger case series, 87.0% of 146 patients achieved disease control with an initial dose of dupilumab 600 mg followed by 300 mg every 2 weeks for 4 weeks, though the study also looked at secondary outcomes for up to 64 weeks.5 Both these findings are compatible with the idea that dupilumab has efficacy in BP, but they cannot be used to test hypotheses.3,4
In a single case report of refractory BP, the combination of dupilumab and omalizumab—a monoclonal antibody that blocks the binding of IgE to mast cells, basophils, and dendritic cells—has shown efficacy, particularly when BP is unresponsive to standard therapies.6 The case involved a 72-year-old woman with unresponsive BP who was treated with a combination of dupilumab and omalizumab. She experienced marked improvement in pruritus and bullae accompanied by normalization of anti-BP180 autoantibody levels. This result points to the possibility that IL-4 and IL-13 blockade could influence autoantibody production. The authors attributed the reduction in pruritus to dupilumab, although the drug was administered in combination with omalizumab.6
Small Randomized Trial of Dupilumab
In addition to these uncontrolled clinical observations, dupilumab recently demonstrated positive results in a small randomized trial.7 Patients who were assigned to receive dupilumab were 5 times more likely to achieve sustained remission compared to those assigned to a placebo. All randomized patients also were treated with a standard-of-care oral corticosteroid regimen, which complicated the ability to isolate the specific effects of dupilumab.7 Finally, a completed totality of evidence on the prescription of dupilumab for refractory BP should include more reliable results from larger randomized trials.8
Final Thoughts
Dupilumab is now approved by the US Food and Drug Administration for the treatment of BP, marking an important milestone as the first targeted biologic therapy for this condition. While the totality of evidence is still evolving, the available data indicate both efficacy and a favorable safety profile when compared to traditional corticosteroids and immunosuppressants. Based on the available data, we suggest that health care providers consider dupilumab as a first-line monotherapy in appropriate patients with BP without specific contraindications, particularly as a practical and safe alternative to corticosteroids and immunosuppressive therapies in older adults with comorbidities. If the patient demonstrates clinical improvement in symptom relief, the therapy can be continued. This strategy minimizes initial exposure to more potent biologics or therapies with higher toxicity profiles. For patients without adequate disease control or with severe initial presentations requiring rapid improvement, clinicians may consider escalation to adjunctive or alternative therapies, such as rituximab, omalizumab, or a short course of systemic corticosteroids for acute management.
Emerging data suggest that dupilumab is a promising therapy for BP. We suggest that it may be a safer targeted alternative to traditional corticosteroids and immunosuppressive therapies. The ability of dupilumab to mitigate inflammation without broadly suppressing the immune system suggests that it may turn out to be an especially valuable option for older patients and those with comorbid conditions.
Acknowledgement—The authors thank Jodi Fiedler, MD (Boca Raton, FL), for her expert advice and assistance.
- Zhang L, Chen Z, Wang L, et al. Bullous pemphigoid: the role of type 2 inflammation in its pathogenesis and the prospect of targeted therapy. Front Immunol. 2023;14:1115083. doi:10.3389/fimmu.2023.1115083
- Sanofi. Dupixent approved in the US as the only targeted medicine to treat patients with bullous pemphigoid. News release. June 20, 2025. Accessed September 8, 2025. https://www.sanofi.com/en/media-room/press-releases/2025/2025-06-20-05-00-00-3102518
- Hennekens CH, DeMets D. Statistical association and causation: contributions of different types of evidence. JAMA. 2011;306:1134-1136. doi:10.1001/jama.2011.322
- Abdat R, Waldman RA, de Bedout V, et al. Dupilumab as a novel therapy for bullous pemphigoid: a multicenter case series. J Am Acad Dermatol. 2020;83:46-52. doi:10.1016/j.jaad.2020.01.089
- Zhao L, Wang Q, Liang G, et al. Evaluation of dupilumab in patients with bullous pemphigoid. JAMA Dermatol. 2023;159:953-960. doi:10.1001/jamadermatol.2023.2428
- Seyed Jafari SM, Feldmeyer L, Bossart S, et al. Case report: combination of omalizumab and dupilumab for recalcitrant bullous pemphigoid. Front Immunol. 2021;11:611549. doi:10.3389/fimmu.2020.611549
- US National Library of Medicine. A multicenter, randomized, double-blind, placebo-controlled, parallel-group study to evaluate the efficacy and safety of dupilumab in adult patients with bullous pemphigoid. ClinicalTrials.gov. Published 2019. Accessed March 10, 2026. https://clinicaltrials.gov/ct2/show/NCT04206553
- Hennekens CH, DeMets D. The need for large scale randomized evidence without undue emphasis on small trials, meta-analyses or subgroup analyses. JAMA. 2009;302:2361-2362. doi:10.1001/jama.2009.1829
Bullous pemphigoid (BP) is a chronic autoimmune skin condition characterized by large, fluid-filled blisters (bullae) on the skin. There usually is severe itching, which may antedate the bullae by several weeks. Bullous pemphigoid most commonly affects adults aged 60 years and older with comorbidities such as metabolic syndrome or other autoimmune diseases. The typical blistering is subepidermal, and its occurrence seems to be driven by autoantibodies targeting the epidermal basement membrane zone.
Conventional treatment for BP has long included systemic corticosteroids and immunosuppressants; this combination of drugs effectively controls inflammation but also is associated with considerable risks such as infection, hyperglycemia with worsening diabetes, hypertension, osteoporosis and fracture risk, and delirium, particularly in older patients. Recent developments in biologic therapy have supported the utilization of potentially safer alternatives. Specifically, dupilumab has demonstrated efficacy in the treatment of atopic dermatitis. Case reports and small research studies included in a recent review have suggested that dupilumab could be effective in managing BP, potentially offering a safer treatment option.1 Importantly, dupilumab recently was approved for BP by the US Food and Drug Administration, making it the first biologic approved for treating this condition.2 In this article, we discuss dupilumab as an option for clinicians to prescribe as an initial therapy for BP.
Pathophysiology of Bullous Pemphigoid and Dupilumab
Bullous pemphigoid pathophysiology is driven by 2 main components: immunologic and inflammatory processes. The immune-mediated attack consists of autoantibodies against the hemidesmosomal proteins BP180 and BP230 in the basement membrane zone, which anchor the epidermis to the dermis.1 When these proteins are targeted, a cascade of responses is triggered, including the release of inflammatory cells, particularly eosinophils and neutrophils, into the skin. These cells release inflammatory mediators that break down the basement membrane, resulting in the separation of the epidermis from the dermis and leading to subepidermal blister formation.
The inflammation associated with BP largely is driven by a T helper 2–dominant immune response, with increased levels of cytokines such as interleukin (IL) 4 and IL-13.1 The chronic inflammation contributes to the characteristic blisters and intense pruritus seen in patients with BP. Traditional treatments such as corticosteroids work broadly to suppress this inflammation by reducing the production of all proinflammatory cytokines; however, they lack specificity, which dampens the targeted immune response against BP and broadly suppresses the immune system, increasing the risk for infection and other adverse effects. For older patients with multiple health conditions, these adverse effects can impact quality of life and pose health risks.
Interleukin 4 and IL-13 contribute to eosinophilia by upregulating the expression of eosinophil chemoattractant cytokines that promote eosinophil migration and adhesion.1 This is where dupilumab presents a promising alternative. Dupilumab is a monoclonal antibody that selectively targets the key cytokines implicated in BP. It binds to the IL-4 receptor alpha subunit, which IL-4 and IL-13 share, thereby inhibiting the signaling of both cytokines.1
Dupilumab Reports and Case Series
Current evidence supporting the use of dupilumab for BP consists primarily of case series and uncontrolled clinical observations, both of which are useful to formulate but not to test hypotheses.3 One case series of 13 patients diagnosed with BP reported that 92.3% (12/13) of patients achieved disease clearance or satisfactory control, with 53.8% (7/13) achieving total lesion clearance and no adverse events observed.4 In a larger case series, 87.0% of 146 patients achieved disease control with an initial dose of dupilumab 600 mg followed by 300 mg every 2 weeks for 4 weeks, though the study also looked at secondary outcomes for up to 64 weeks.5 Both these findings are compatible with the idea that dupilumab has efficacy in BP, but they cannot be used to test hypotheses.3,4
In a single case report of refractory BP, the combination of dupilumab and omalizumab—a monoclonal antibody that blocks the binding of IgE to mast cells, basophils, and dendritic cells—has shown efficacy, particularly when BP is unresponsive to standard therapies.6 The case involved a 72-year-old woman with unresponsive BP who was treated with a combination of dupilumab and omalizumab. She experienced marked improvement in pruritus and bullae accompanied by normalization of anti-BP180 autoantibody levels. This result points to the possibility that IL-4 and IL-13 blockade could influence autoantibody production. The authors attributed the reduction in pruritus to dupilumab, although the drug was administered in combination with omalizumab.6
Small Randomized Trial of Dupilumab
In addition to these uncontrolled clinical observations, dupilumab recently demonstrated positive results in a small randomized trial.7 Patients who were assigned to receive dupilumab were 5 times more likely to achieve sustained remission compared to those assigned to a placebo. All randomized patients also were treated with a standard-of-care oral corticosteroid regimen, which complicated the ability to isolate the specific effects of dupilumab.7 Finally, a completed totality of evidence on the prescription of dupilumab for refractory BP should include more reliable results from larger randomized trials.8
Final Thoughts
Dupilumab is now approved by the US Food and Drug Administration for the treatment of BP, marking an important milestone as the first targeted biologic therapy for this condition. While the totality of evidence is still evolving, the available data indicate both efficacy and a favorable safety profile when compared to traditional corticosteroids and immunosuppressants. Based on the available data, we suggest that health care providers consider dupilumab as a first-line monotherapy in appropriate patients with BP without specific contraindications, particularly as a practical and safe alternative to corticosteroids and immunosuppressive therapies in older adults with comorbidities. If the patient demonstrates clinical improvement in symptom relief, the therapy can be continued. This strategy minimizes initial exposure to more potent biologics or therapies with higher toxicity profiles. For patients without adequate disease control or with severe initial presentations requiring rapid improvement, clinicians may consider escalation to adjunctive or alternative therapies, such as rituximab, omalizumab, or a short course of systemic corticosteroids for acute management.
Emerging data suggest that dupilumab is a promising therapy for BP. We suggest that it may be a safer targeted alternative to traditional corticosteroids and immunosuppressive therapies. The ability of dupilumab to mitigate inflammation without broadly suppressing the immune system suggests that it may turn out to be an especially valuable option for older patients and those with comorbid conditions.
Acknowledgement—The authors thank Jodi Fiedler, MD (Boca Raton, FL), for her expert advice and assistance.
Bullous pemphigoid (BP) is a chronic autoimmune skin condition characterized by large, fluid-filled blisters (bullae) on the skin. There usually is severe itching, which may antedate the bullae by several weeks. Bullous pemphigoid most commonly affects adults aged 60 years and older with comorbidities such as metabolic syndrome or other autoimmune diseases. The typical blistering is subepidermal, and its occurrence seems to be driven by autoantibodies targeting the epidermal basement membrane zone.
Conventional treatment for BP has long included systemic corticosteroids and immunosuppressants; this combination of drugs effectively controls inflammation but also is associated with considerable risks such as infection, hyperglycemia with worsening diabetes, hypertension, osteoporosis and fracture risk, and delirium, particularly in older patients. Recent developments in biologic therapy have supported the utilization of potentially safer alternatives. Specifically, dupilumab has demonstrated efficacy in the treatment of atopic dermatitis. Case reports and small research studies included in a recent review have suggested that dupilumab could be effective in managing BP, potentially offering a safer treatment option.1 Importantly, dupilumab recently was approved for BP by the US Food and Drug Administration, making it the first biologic approved for treating this condition.2 In this article, we discuss dupilumab as an option for clinicians to prescribe as an initial therapy for BP.
Pathophysiology of Bullous Pemphigoid and Dupilumab
Bullous pemphigoid pathophysiology is driven by 2 main components: immunologic and inflammatory processes. The immune-mediated attack consists of autoantibodies against the hemidesmosomal proteins BP180 and BP230 in the basement membrane zone, which anchor the epidermis to the dermis.1 When these proteins are targeted, a cascade of responses is triggered, including the release of inflammatory cells, particularly eosinophils and neutrophils, into the skin. These cells release inflammatory mediators that break down the basement membrane, resulting in the separation of the epidermis from the dermis and leading to subepidermal blister formation.
The inflammation associated with BP largely is driven by a T helper 2–dominant immune response, with increased levels of cytokines such as interleukin (IL) 4 and IL-13.1 The chronic inflammation contributes to the characteristic blisters and intense pruritus seen in patients with BP. Traditional treatments such as corticosteroids work broadly to suppress this inflammation by reducing the production of all proinflammatory cytokines; however, they lack specificity, which dampens the targeted immune response against BP and broadly suppresses the immune system, increasing the risk for infection and other adverse effects. For older patients with multiple health conditions, these adverse effects can impact quality of life and pose health risks.
Interleukin 4 and IL-13 contribute to eosinophilia by upregulating the expression of eosinophil chemoattractant cytokines that promote eosinophil migration and adhesion.1 This is where dupilumab presents a promising alternative. Dupilumab is a monoclonal antibody that selectively targets the key cytokines implicated in BP. It binds to the IL-4 receptor alpha subunit, which IL-4 and IL-13 share, thereby inhibiting the signaling of both cytokines.1
Dupilumab Reports and Case Series
Current evidence supporting the use of dupilumab for BP consists primarily of case series and uncontrolled clinical observations, both of which are useful to formulate but not to test hypotheses.3 One case series of 13 patients diagnosed with BP reported that 92.3% (12/13) of patients achieved disease clearance or satisfactory control, with 53.8% (7/13) achieving total lesion clearance and no adverse events observed.4 In a larger case series, 87.0% of 146 patients achieved disease control with an initial dose of dupilumab 600 mg followed by 300 mg every 2 weeks for 4 weeks, though the study also looked at secondary outcomes for up to 64 weeks.5 Both these findings are compatible with the idea that dupilumab has efficacy in BP, but they cannot be used to test hypotheses.3,4
In a single case report of refractory BP, the combination of dupilumab and omalizumab—a monoclonal antibody that blocks the binding of IgE to mast cells, basophils, and dendritic cells—has shown efficacy, particularly when BP is unresponsive to standard therapies.6 The case involved a 72-year-old woman with unresponsive BP who was treated with a combination of dupilumab and omalizumab. She experienced marked improvement in pruritus and bullae accompanied by normalization of anti-BP180 autoantibody levels. This result points to the possibility that IL-4 and IL-13 blockade could influence autoantibody production. The authors attributed the reduction in pruritus to dupilumab, although the drug was administered in combination with omalizumab.6
Small Randomized Trial of Dupilumab
In addition to these uncontrolled clinical observations, dupilumab recently demonstrated positive results in a small randomized trial.7 Patients who were assigned to receive dupilumab were 5 times more likely to achieve sustained remission compared to those assigned to a placebo. All randomized patients also were treated with a standard-of-care oral corticosteroid regimen, which complicated the ability to isolate the specific effects of dupilumab.7 Finally, a completed totality of evidence on the prescription of dupilumab for refractory BP should include more reliable results from larger randomized trials.8
Final Thoughts
Dupilumab is now approved by the US Food and Drug Administration for the treatment of BP, marking an important milestone as the first targeted biologic therapy for this condition. While the totality of evidence is still evolving, the available data indicate both efficacy and a favorable safety profile when compared to traditional corticosteroids and immunosuppressants. Based on the available data, we suggest that health care providers consider dupilumab as a first-line monotherapy in appropriate patients with BP without specific contraindications, particularly as a practical and safe alternative to corticosteroids and immunosuppressive therapies in older adults with comorbidities. If the patient demonstrates clinical improvement in symptom relief, the therapy can be continued. This strategy minimizes initial exposure to more potent biologics or therapies with higher toxicity profiles. For patients without adequate disease control or with severe initial presentations requiring rapid improvement, clinicians may consider escalation to adjunctive or alternative therapies, such as rituximab, omalizumab, or a short course of systemic corticosteroids for acute management.
Emerging data suggest that dupilumab is a promising therapy for BP. We suggest that it may be a safer targeted alternative to traditional corticosteroids and immunosuppressive therapies. The ability of dupilumab to mitigate inflammation without broadly suppressing the immune system suggests that it may turn out to be an especially valuable option for older patients and those with comorbid conditions.
Acknowledgement—The authors thank Jodi Fiedler, MD (Boca Raton, FL), for her expert advice and assistance.
- Zhang L, Chen Z, Wang L, et al. Bullous pemphigoid: the role of type 2 inflammation in its pathogenesis and the prospect of targeted therapy. Front Immunol. 2023;14:1115083. doi:10.3389/fimmu.2023.1115083
- Sanofi. Dupixent approved in the US as the only targeted medicine to treat patients with bullous pemphigoid. News release. June 20, 2025. Accessed September 8, 2025. https://www.sanofi.com/en/media-room/press-releases/2025/2025-06-20-05-00-00-3102518
- Hennekens CH, DeMets D. Statistical association and causation: contributions of different types of evidence. JAMA. 2011;306:1134-1136. doi:10.1001/jama.2011.322
- Abdat R, Waldman RA, de Bedout V, et al. Dupilumab as a novel therapy for bullous pemphigoid: a multicenter case series. J Am Acad Dermatol. 2020;83:46-52. doi:10.1016/j.jaad.2020.01.089
- Zhao L, Wang Q, Liang G, et al. Evaluation of dupilumab in patients with bullous pemphigoid. JAMA Dermatol. 2023;159:953-960. doi:10.1001/jamadermatol.2023.2428
- Seyed Jafari SM, Feldmeyer L, Bossart S, et al. Case report: combination of omalizumab and dupilumab for recalcitrant bullous pemphigoid. Front Immunol. 2021;11:611549. doi:10.3389/fimmu.2020.611549
- US National Library of Medicine. A multicenter, randomized, double-blind, placebo-controlled, parallel-group study to evaluate the efficacy and safety of dupilumab in adult patients with bullous pemphigoid. ClinicalTrials.gov. Published 2019. Accessed March 10, 2026. https://clinicaltrials.gov/ct2/show/NCT04206553
- Hennekens CH, DeMets D. The need for large scale randomized evidence without undue emphasis on small trials, meta-analyses or subgroup analyses. JAMA. 2009;302:2361-2362. doi:10.1001/jama.2009.1829
- Zhang L, Chen Z, Wang L, et al. Bullous pemphigoid: the role of type 2 inflammation in its pathogenesis and the prospect of targeted therapy. Front Immunol. 2023;14:1115083. doi:10.3389/fimmu.2023.1115083
- Sanofi. Dupixent approved in the US as the only targeted medicine to treat patients with bullous pemphigoid. News release. June 20, 2025. Accessed September 8, 2025. https://www.sanofi.com/en/media-room/press-releases/2025/2025-06-20-05-00-00-3102518
- Hennekens CH, DeMets D. Statistical association and causation: contributions of different types of evidence. JAMA. 2011;306:1134-1136. doi:10.1001/jama.2011.322
- Abdat R, Waldman RA, de Bedout V, et al. Dupilumab as a novel therapy for bullous pemphigoid: a multicenter case series. J Am Acad Dermatol. 2020;83:46-52. doi:10.1016/j.jaad.2020.01.089
- Zhao L, Wang Q, Liang G, et al. Evaluation of dupilumab in patients with bullous pemphigoid. JAMA Dermatol. 2023;159:953-960. doi:10.1001/jamadermatol.2023.2428
- Seyed Jafari SM, Feldmeyer L, Bossart S, et al. Case report: combination of omalizumab and dupilumab for recalcitrant bullous pemphigoid. Front Immunol. 2021;11:611549. doi:10.3389/fimmu.2020.611549
- US National Library of Medicine. A multicenter, randomized, double-blind, placebo-controlled, parallel-group study to evaluate the efficacy and safety of dupilumab in adult patients with bullous pemphigoid. ClinicalTrials.gov. Published 2019. Accessed March 10, 2026. https://clinicaltrials.gov/ct2/show/NCT04206553
- Hennekens CH, DeMets D. The need for large scale randomized evidence without undue emphasis on small trials, meta-analyses or subgroup analyses. JAMA. 2009;302:2361-2362. doi:10.1001/jama.2009.1829
Dupilumab for Bullous Pemphigoid: To Treat or Not to Treat?
Dupilumab for Bullous Pemphigoid: To Treat or Not to Treat?
PRACTICE POINTS
- Bullous pemphigoid (BP) is a pruritic autoimmune blistering disease in older adults that often requires systemic therapy with associated risks.
- Dupilumab targets interleukin (IL) 4/IL-13–driven inflammation and shows promising efficacy in BP.
- As the first approved biologic for BP, dupilumab may offer a safer alternative to corticosteroids in select patients.
Social Drivers of Health Curriculum for Dermatology Residents: the UCSF Experience
Social Drivers of Health Curriculum for Dermatology Residents: the UCSF Experience
Social drivers of health (SDH) describe the conditions in which an individual is born, grows, lives, works, and ages—all of which collectively influence their health. Examples of SDH include employment status, literacy level, education level, housing status, food access, income level, and social cohesion. Social drivers of health are critical catalysts to attaining health equity. Effectively applying an understanding of how SDH affect the care of all patients is an essential competency for physicians practicing in the modern era of rising income inequality and housing instability and increasing racial, ethnic, language, religious, and cultural diversity in the United States; however, in dermatology residency, this skill set often is developed by the hidden curriculum (ie, the informal curriculum that is based on what patient scenarios a resident happens to face) rather than one represented by formal educational objectives.2
Adding to this challenge of limited formal curricula is that caring for minoritized, marginalized, and other populations facing specific barriers can evoke feelings of frustration, helplessness, and even anger. These feelings can test the limits of a physician’s identity as a healer, leading to burnout and self-protective attitudes such as distancing (emotionally, physically, or both) from these patients.3 This is particularly relevant given that the majority (76%-79% each year from 2007-2019) of medical student matriculants come from families with incomes in the top 2 quintiles nationwide, and fewer than 6% come from the lowest quintile earners.4,5 These data indicate that most trainees have not experienced (and may even have a hard time imagining) the degree of economic and housing instability faced by many of their low-income patients, the care of whom disproportionately falls to large academic medical centers, which sponsor dermatology training programs.6 Many trainees may feel uncomfortable communicating across the broad range of racial, socioeconomic, linguistic, and cultural differences they encounter during training and in practice. Structured opportunities to provide care in a supervised supportive environment combined with didactics that emphasize practical, evidence-based strategies can build empathy, improve attitudes toward patients from diverse backgrounds, and strengthen self-efficacy in challenging scenarios.3
In the past decade, there has been a push toward integrating our understanding of SDH into formal medical training.7 Other specialty training programs—including psychiatry,8 internal medicine,9 pediatrics,10 and family medicine11—have incorporated these elements into their curricula and competency evaluations. In dermatology, as in other specialties, making and implementing effective, patient-centered care plans requires attention to the various social and structural drivers that may influence outcomes. Dermatologists therefore should be educated about SDH during their training programs and empowered to address the ways they affect patient care.
At the University of California San Francisco (UCSF)(San Francisco, California), our dermatology trainees care for patients in several hospital systems citywide, including a tertiary academic medical center with multiple locations, a county hospital, and a Veterans Affairs medical center. Given the diversity of patient populations across our training sites—including many racially and ethnically minoritized individuals, immigrants, patients with limited English proficiency, people experiencing homelessness, and sexual and gender diverse individuals—we identified a critical opportunity to enhance our training through formal didactics and hands-on experiences that integrate SDH into existing curricula and strengthen trainees’ ability to provide high-quality care to all patients.
Implementing an SDH Curriculum
In May 2020, UCSF dermatology faculty with an interest in SDH collaborated with departmental educational leadership to develop a formal SDH curriculum centered around 8 core learning objectives for residents (eTable 1). To achieve these objectives, we organized a 3-year didactic and experiential curriculum consisting of lectures (eTable 2), grand rounds sessions, journal clubs, and community engagement opportunities. Residents also spend 7 months during their training rotating at San Francisco’s city and county hospital (Zuckerberg San Francisco General Hospital [San Francisco, California]) where all faculty are members of the core SDH curriculum development team and where residents can put into practice many of the skills learned in formal didactics to develop patient-centered care plans for low-income patients, approximately 40% of whom have limited English proficiency.

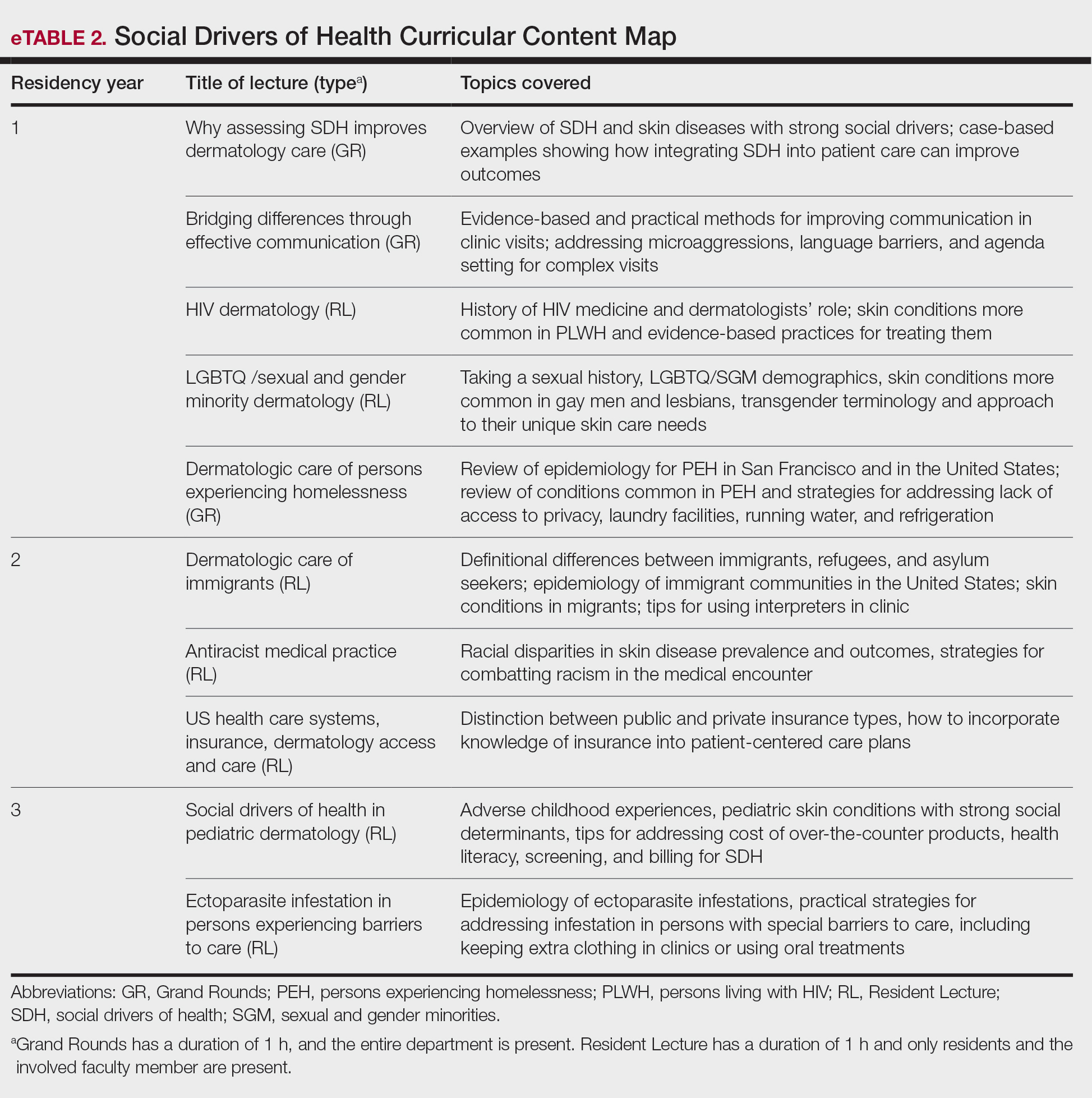
To further center the importance of SDH and health equity in our training program, we developed a Health Equity Chief leadership role for senior dermatology residents. Each year, 2 to 4 residents volunteer for and serve in this role, wherein they work with core faculty to review and improve SDH curriculum elements. They also work to enhance community engagement opportunities for residents (eg, pathway programs aimed at diversifying the dermatology workforce by introducing historically excluded local high school and college students to dermatology as a career path) and improve dermatology trainees’ awareness of the history and health needs of the specific communities we serve in San Francisco. They also are prepared to become leaders in the field of health equity and to improve the care of diverse patient populations after residency. Our faculty curriculum leaders meet quarterly with our Health Equity Chiefs to review their individual and collective goals and strategize ways to improve learner and community engagement. Departmental funds are made available to support these efforts.
Leadership at our safety-net county hospital also developed a patient navigator position to improve our ability to care for patients with the most complex medical conditions and social needs. This role is held by a medical student taking a funded gap year and incorporates aspects of social work (eg, identifying barriers to care and connecting patients with resources such as transportation), quality improvement, and clinical research.12
Assessing Residents’ Experience of a New SDH Curriculum
Prior to curriculum implementation, we surveyed graduating UCSF dermatology residents in June 2020 to assess their familiarity with SDH and the social and medical needs of various populations facing barriers to care, their comfort level with specific challenging clinical situations, and their desire for additional training. Responses were measured using a 5-part Likert scale, with additional options for free-text response. After initiating the SDH curriculum in July 2020, we sent the same survey each year to all senior residents immediately prior to their graduation, offering a small financial incentive ($15 cash gift card) to those who completed the survey. We obtained UCSF Institutional Review Board approval to utilize these survey data to better understand and to enhance residents’ experience of the SDH curriculum.
All 8 residents invited in 2020 completed the survey assessing curriculum efficacy (100% response rate). For the 2023 and 2024 classes, data were analyzed in aggregate (n=14), with a 50% response rate. After implementation of the SDH curriculum, there was improvement in learners’ awareness of challenges faced by every patient population, from a mean (SD) of 3.12 (0.66) to 4.52 (SD, 0.69)(P<.05). Learners were more comfortable handling hypothetical clinical scenarios requiring them to identify and address specific SDH after vs before implementation of the curriculum (mean [SD], 3.5 [1.06] before vs 4.0 [1.16] after)(P>.05), though this difference was not statistically learners’ awareness of challenges faced by every patient population, from a mean (SD) of 3.12 (0.66) to 4.52 (0.69)(P<.05). Learners were more comfortable handling hypothetical clinical scenarios requiring them to identify and address specific SDH after vs before implementation of the curriculum (mean [SD], 3.5 [1.06] before vs 4.0 [1.16] after)(P>.05), though this difference was not statistically significant. Finally, many respondents expressed appreciation that our curriculum improved their ability to care for patients in complex social circumstances. Residents suggested in the free-text responses that learning more about the historical underpinnings of health disparities, opportunities for grassroots activism, and how to provide more culturally competent care of Native American populations could improve our curriculum.
Implications for Dermatology Training
Our survey results indicate that a formal SDH curriculum can improve dermatology residents’ ability to care for populations with complex social needs. We advocate for implementing SDH curricula into dermatology training programs nationwide, as has been recommended by others.13,14 We also propose that structural competency should eventually be a key dermatologic competency as determined by the Accreditation Council for Graduate Medical Education, in line with the American Medical Association’s recommendation that structural competency is a learned skill required to end health inequity.15 The Accreditation Council for Graduate Medical Education specialty program requirements currently are being revised; interested individuals can engage in this process by submitting this suggestion for public comment (https://www.acgme.org/programs-and-institutions/programs/review-and-comment/).
Limitations of a survey include the relatively small sample size (7-8 per year) and variable response rates. In addition, we did not survey each class of residents at the beginning and end of their training; our comparisons therefore were limited by comparing different individuals with distinct backgrounds and experiences. Furthermore, we acknowledge that the experience of developing this curriculum in San Francisco may be distinct from other communities, where access to dermatologic care may vary according to both the availability of public health insurance and the treatments covered by public insurers. In San Francisco, insurance coverage is near universal, such that residents in our training program regularly care for undocumented immigrants, persons experiencing homelessness, and other populations that might find it challenging to present to dermatology clinics in other settings nationwide.
Final Thoughts
Future directions of our curriculum include exploration of novel curriculum delivery methods (including a problem-based curriculum approach and other more experiential didactics), increased opportunities for community engagement, greater focus on advocacy with an emphasis on broader social and structural policies and their downstream effects, and focusing more specifically on the history and needs of specific low-income San Francisco neighborhoods and diverse patient populations.
- US Department of Health and Human Services. Health Equity in Healthy People 2030. Accessed January 7, 2025. https://odphp.healtwh.gov/healthypeople/priority-areas/health-equity-healthy-people-2030
- Axelson DJ, Stull MJ, Coates WC. Social determinants of health: a missing link in emergency medicine training. AEM Educ Train. 2018;2:66-68. doi:10.1002/aet2.10056
- Brenner AM, Guerrero APS, Beresin EV, et al. Teaching medical students and residents about homelessness: complex, evidence-based, and imperative. Acad Psychiatry. 2016;40:572-575. doi:10.1007/s40596-016-0571-6
- Youngclaus J, Roskovensky L. An Updated Look at the Economic Diversity of U.S. Medical Students. American Association of Medical Colleges Analysis in Brief. 2018;18. https://www.aamc.org/media/9596/download?attachment
- Shahriar AA, Puram VV, Miller JM, et al. Socioeconomic diversity of the matriculating US medical student body by race, ethnicity, and sex, 2017-2019. JAMA Netw Open. 2022;5:E222621. doi:10.1001/jamanetworkopen.2022.2621
- Williams JC, Maxey AE, Wei ML, et al. A cross-sectional analysis of Medicaid acceptance among US dermatology residency training programs. J Am Acad Dermatol. 2022;86:453-455. doi:10.1016/j.jaad.2021.09.046
- Daniel H, Bornstein S, Kane G; Health and Public Policy Committee of the American College of Physicians. Addressing social determinants to improve patient care and promote health equity: an American College of Physicians position paper. Ann Intern Med. 2018;168:577-578. doi:10.2105/AJPH
- Hansen H, Kline N, Braslow J, et al. From cultural to structural competency—training psychiatry residents to act on social determinants of health and institutional racism. JAMA Psychiatry. 2018;75:117-118. doi:10.1001/jamapsychiatry.2017.3894
- Schmidt S, Higgins S, George M, et al. An experiential resident module for understanding social determinants of health at an academic safety-net hospital. MedEdPORTAL. 2017;26:10647. doi:10.15766/mep_2374-8265.10647
- Hoffman BD, Rose J, Best D, et al. The community pediatrics training initiative project planning tool: a practical approach to community-based advocacy. MedEdPORTAL. 2017;13:10630.
- Chrisman-Khawam L, Abdullah N, Dhoopar A. Teaching health-care trainees empathy and homelessness IQ through service learning, reflective practice, and altruistic attribution. Int J Psychiatry Med. 2017;52:245-254. doi:10.1177/0091217417730288
- Sanchez-Anguiano ME, Klufas D, Amerson E. Screening for cardiometabolic risk factors in patients with psoriasis and hidradenitis suppurativa: a pilot study in a safety net population. J Am Acad Dermatol. 2024;91:1269-1272. doi:10.1016/j.jaad.2024.07.1518
- Riley C, Vasquez R, Pritchett EN. Equipping dermatologists to address structural and social drivers of inequities—structural competency. JAMA Dermatol. 2024;160:1037-1038. doi:10.1001/jamadermatol.2024.2351
- Crawl-Bey A, Pritchett EN, Riley C. 54338 Structural competency in dermatology: a pilot curriculum for equipping residents to address structural factors that contribute to health inequity. J Am Acad Dermatol. 2024;91(3 suppl):AB318. doi:10.1016/j.jaad.2024.07.1264
- Smith TM. New competency focus involves structural factors in health. American Medical Association. December 18, 202. Accessed March 23, 2026. https://www.ama-assn.org/education/changemeded-initiative/new-competency-focus-involves-structural-factors-health
Social drivers of health (SDH) describe the conditions in which an individual is born, grows, lives, works, and ages—all of which collectively influence their health. Examples of SDH include employment status, literacy level, education level, housing status, food access, income level, and social cohesion. Social drivers of health are critical catalysts to attaining health equity. Effectively applying an understanding of how SDH affect the care of all patients is an essential competency for physicians practicing in the modern era of rising income inequality and housing instability and increasing racial, ethnic, language, religious, and cultural diversity in the United States; however, in dermatology residency, this skill set often is developed by the hidden curriculum (ie, the informal curriculum that is based on what patient scenarios a resident happens to face) rather than one represented by formal educational objectives.2
Adding to this challenge of limited formal curricula is that caring for minoritized, marginalized, and other populations facing specific barriers can evoke feelings of frustration, helplessness, and even anger. These feelings can test the limits of a physician’s identity as a healer, leading to burnout and self-protective attitudes such as distancing (emotionally, physically, or both) from these patients.3 This is particularly relevant given that the majority (76%-79% each year from 2007-2019) of medical student matriculants come from families with incomes in the top 2 quintiles nationwide, and fewer than 6% come from the lowest quintile earners.4,5 These data indicate that most trainees have not experienced (and may even have a hard time imagining) the degree of economic and housing instability faced by many of their low-income patients, the care of whom disproportionately falls to large academic medical centers, which sponsor dermatology training programs.6 Many trainees may feel uncomfortable communicating across the broad range of racial, socioeconomic, linguistic, and cultural differences they encounter during training and in practice. Structured opportunities to provide care in a supervised supportive environment combined with didactics that emphasize practical, evidence-based strategies can build empathy, improve attitudes toward patients from diverse backgrounds, and strengthen self-efficacy in challenging scenarios.3
In the past decade, there has been a push toward integrating our understanding of SDH into formal medical training.7 Other specialty training programs—including psychiatry,8 internal medicine,9 pediatrics,10 and family medicine11—have incorporated these elements into their curricula and competency evaluations. In dermatology, as in other specialties, making and implementing effective, patient-centered care plans requires attention to the various social and structural drivers that may influence outcomes. Dermatologists therefore should be educated about SDH during their training programs and empowered to address the ways they affect patient care.
At the University of California San Francisco (UCSF)(San Francisco, California), our dermatology trainees care for patients in several hospital systems citywide, including a tertiary academic medical center with multiple locations, a county hospital, and a Veterans Affairs medical center. Given the diversity of patient populations across our training sites—including many racially and ethnically minoritized individuals, immigrants, patients with limited English proficiency, people experiencing homelessness, and sexual and gender diverse individuals—we identified a critical opportunity to enhance our training through formal didactics and hands-on experiences that integrate SDH into existing curricula and strengthen trainees’ ability to provide high-quality care to all patients.
Implementing an SDH Curriculum
In May 2020, UCSF dermatology faculty with an interest in SDH collaborated with departmental educational leadership to develop a formal SDH curriculum centered around 8 core learning objectives for residents (eTable 1). To achieve these objectives, we organized a 3-year didactic and experiential curriculum consisting of lectures (eTable 2), grand rounds sessions, journal clubs, and community engagement opportunities. Residents also spend 7 months during their training rotating at San Francisco’s city and county hospital (Zuckerberg San Francisco General Hospital [San Francisco, California]) where all faculty are members of the core SDH curriculum development team and where residents can put into practice many of the skills learned in formal didactics to develop patient-centered care plans for low-income patients, approximately 40% of whom have limited English proficiency.


To further center the importance of SDH and health equity in our training program, we developed a Health Equity Chief leadership role for senior dermatology residents. Each year, 2 to 4 residents volunteer for and serve in this role, wherein they work with core faculty to review and improve SDH curriculum elements. They also work to enhance community engagement opportunities for residents (eg, pathway programs aimed at diversifying the dermatology workforce by introducing historically excluded local high school and college students to dermatology as a career path) and improve dermatology trainees’ awareness of the history and health needs of the specific communities we serve in San Francisco. They also are prepared to become leaders in the field of health equity and to improve the care of diverse patient populations after residency. Our faculty curriculum leaders meet quarterly with our Health Equity Chiefs to review their individual and collective goals and strategize ways to improve learner and community engagement. Departmental funds are made available to support these efforts.
Leadership at our safety-net county hospital also developed a patient navigator position to improve our ability to care for patients with the most complex medical conditions and social needs. This role is held by a medical student taking a funded gap year and incorporates aspects of social work (eg, identifying barriers to care and connecting patients with resources such as transportation), quality improvement, and clinical research.12
Assessing Residents’ Experience of a New SDH Curriculum
Prior to curriculum implementation, we surveyed graduating UCSF dermatology residents in June 2020 to assess their familiarity with SDH and the social and medical needs of various populations facing barriers to care, their comfort level with specific challenging clinical situations, and their desire for additional training. Responses were measured using a 5-part Likert scale, with additional options for free-text response. After initiating the SDH curriculum in July 2020, we sent the same survey each year to all senior residents immediately prior to their graduation, offering a small financial incentive ($15 cash gift card) to those who completed the survey. We obtained UCSF Institutional Review Board approval to utilize these survey data to better understand and to enhance residents’ experience of the SDH curriculum.
All 8 residents invited in 2020 completed the survey assessing curriculum efficacy (100% response rate). For the 2023 and 2024 classes, data were analyzed in aggregate (n=14), with a 50% response rate. After implementation of the SDH curriculum, there was improvement in learners’ awareness of challenges faced by every patient population, from a mean (SD) of 3.12 (0.66) to 4.52 (SD, 0.69)(P<.05). Learners were more comfortable handling hypothetical clinical scenarios requiring them to identify and address specific SDH after vs before implementation of the curriculum (mean [SD], 3.5 [1.06] before vs 4.0 [1.16] after)(P>.05), though this difference was not statistically learners’ awareness of challenges faced by every patient population, from a mean (SD) of 3.12 (0.66) to 4.52 (0.69)(P<.05). Learners were more comfortable handling hypothetical clinical scenarios requiring them to identify and address specific SDH after vs before implementation of the curriculum (mean [SD], 3.5 [1.06] before vs 4.0 [1.16] after)(P>.05), though this difference was not statistically significant. Finally, many respondents expressed appreciation that our curriculum improved their ability to care for patients in complex social circumstances. Residents suggested in the free-text responses that learning more about the historical underpinnings of health disparities, opportunities for grassroots activism, and how to provide more culturally competent care of Native American populations could improve our curriculum.
Implications for Dermatology Training
Our survey results indicate that a formal SDH curriculum can improve dermatology residents’ ability to care for populations with complex social needs. We advocate for implementing SDH curricula into dermatology training programs nationwide, as has been recommended by others.13,14 We also propose that structural competency should eventually be a key dermatologic competency as determined by the Accreditation Council for Graduate Medical Education, in line with the American Medical Association’s recommendation that structural competency is a learned skill required to end health inequity.15 The Accreditation Council for Graduate Medical Education specialty program requirements currently are being revised; interested individuals can engage in this process by submitting this suggestion for public comment (https://www.acgme.org/programs-and-institutions/programs/review-and-comment/).
Limitations of a survey include the relatively small sample size (7-8 per year) and variable response rates. In addition, we did not survey each class of residents at the beginning and end of their training; our comparisons therefore were limited by comparing different individuals with distinct backgrounds and experiences. Furthermore, we acknowledge that the experience of developing this curriculum in San Francisco may be distinct from other communities, where access to dermatologic care may vary according to both the availability of public health insurance and the treatments covered by public insurers. In San Francisco, insurance coverage is near universal, such that residents in our training program regularly care for undocumented immigrants, persons experiencing homelessness, and other populations that might find it challenging to present to dermatology clinics in other settings nationwide.
Final Thoughts
Future directions of our curriculum include exploration of novel curriculum delivery methods (including a problem-based curriculum approach and other more experiential didactics), increased opportunities for community engagement, greater focus on advocacy with an emphasis on broader social and structural policies and their downstream effects, and focusing more specifically on the history and needs of specific low-income San Francisco neighborhoods and diverse patient populations.
Social drivers of health (SDH) describe the conditions in which an individual is born, grows, lives, works, and ages—all of which collectively influence their health. Examples of SDH include employment status, literacy level, education level, housing status, food access, income level, and social cohesion. Social drivers of health are critical catalysts to attaining health equity. Effectively applying an understanding of how SDH affect the care of all patients is an essential competency for physicians practicing in the modern era of rising income inequality and housing instability and increasing racial, ethnic, language, religious, and cultural diversity in the United States; however, in dermatology residency, this skill set often is developed by the hidden curriculum (ie, the informal curriculum that is based on what patient scenarios a resident happens to face) rather than one represented by formal educational objectives.2
Adding to this challenge of limited formal curricula is that caring for minoritized, marginalized, and other populations facing specific barriers can evoke feelings of frustration, helplessness, and even anger. These feelings can test the limits of a physician’s identity as a healer, leading to burnout and self-protective attitudes such as distancing (emotionally, physically, or both) from these patients.3 This is particularly relevant given that the majority (76%-79% each year from 2007-2019) of medical student matriculants come from families with incomes in the top 2 quintiles nationwide, and fewer than 6% come from the lowest quintile earners.4,5 These data indicate that most trainees have not experienced (and may even have a hard time imagining) the degree of economic and housing instability faced by many of their low-income patients, the care of whom disproportionately falls to large academic medical centers, which sponsor dermatology training programs.6 Many trainees may feel uncomfortable communicating across the broad range of racial, socioeconomic, linguistic, and cultural differences they encounter during training and in practice. Structured opportunities to provide care in a supervised supportive environment combined with didactics that emphasize practical, evidence-based strategies can build empathy, improve attitudes toward patients from diverse backgrounds, and strengthen self-efficacy in challenging scenarios.3
In the past decade, there has been a push toward integrating our understanding of SDH into formal medical training.7 Other specialty training programs—including psychiatry,8 internal medicine,9 pediatrics,10 and family medicine11—have incorporated these elements into their curricula and competency evaluations. In dermatology, as in other specialties, making and implementing effective, patient-centered care plans requires attention to the various social and structural drivers that may influence outcomes. Dermatologists therefore should be educated about SDH during their training programs and empowered to address the ways they affect patient care.
At the University of California San Francisco (UCSF)(San Francisco, California), our dermatology trainees care for patients in several hospital systems citywide, including a tertiary academic medical center with multiple locations, a county hospital, and a Veterans Affairs medical center. Given the diversity of patient populations across our training sites—including many racially and ethnically minoritized individuals, immigrants, patients with limited English proficiency, people experiencing homelessness, and sexual and gender diverse individuals—we identified a critical opportunity to enhance our training through formal didactics and hands-on experiences that integrate SDH into existing curricula and strengthen trainees’ ability to provide high-quality care to all patients.
Implementing an SDH Curriculum
In May 2020, UCSF dermatology faculty with an interest in SDH collaborated with departmental educational leadership to develop a formal SDH curriculum centered around 8 core learning objectives for residents (eTable 1). To achieve these objectives, we organized a 3-year didactic and experiential curriculum consisting of lectures (eTable 2), grand rounds sessions, journal clubs, and community engagement opportunities. Residents also spend 7 months during their training rotating at San Francisco’s city and county hospital (Zuckerberg San Francisco General Hospital [San Francisco, California]) where all faculty are members of the core SDH curriculum development team and where residents can put into practice many of the skills learned in formal didactics to develop patient-centered care plans for low-income patients, approximately 40% of whom have limited English proficiency.


To further center the importance of SDH and health equity in our training program, we developed a Health Equity Chief leadership role for senior dermatology residents. Each year, 2 to 4 residents volunteer for and serve in this role, wherein they work with core faculty to review and improve SDH curriculum elements. They also work to enhance community engagement opportunities for residents (eg, pathway programs aimed at diversifying the dermatology workforce by introducing historically excluded local high school and college students to dermatology as a career path) and improve dermatology trainees’ awareness of the history and health needs of the specific communities we serve in San Francisco. They also are prepared to become leaders in the field of health equity and to improve the care of diverse patient populations after residency. Our faculty curriculum leaders meet quarterly with our Health Equity Chiefs to review their individual and collective goals and strategize ways to improve learner and community engagement. Departmental funds are made available to support these efforts.
Leadership at our safety-net county hospital also developed a patient navigator position to improve our ability to care for patients with the most complex medical conditions and social needs. This role is held by a medical student taking a funded gap year and incorporates aspects of social work (eg, identifying barriers to care and connecting patients with resources such as transportation), quality improvement, and clinical research.12
Assessing Residents’ Experience of a New SDH Curriculum
Prior to curriculum implementation, we surveyed graduating UCSF dermatology residents in June 2020 to assess their familiarity with SDH and the social and medical needs of various populations facing barriers to care, their comfort level with specific challenging clinical situations, and their desire for additional training. Responses were measured using a 5-part Likert scale, with additional options for free-text response. After initiating the SDH curriculum in July 2020, we sent the same survey each year to all senior residents immediately prior to their graduation, offering a small financial incentive ($15 cash gift card) to those who completed the survey. We obtained UCSF Institutional Review Board approval to utilize these survey data to better understand and to enhance residents’ experience of the SDH curriculum.
All 8 residents invited in 2020 completed the survey assessing curriculum efficacy (100% response rate). For the 2023 and 2024 classes, data were analyzed in aggregate (n=14), with a 50% response rate. After implementation of the SDH curriculum, there was improvement in learners’ awareness of challenges faced by every patient population, from a mean (SD) of 3.12 (0.66) to 4.52 (SD, 0.69)(P<.05). Learners were more comfortable handling hypothetical clinical scenarios requiring them to identify and address specific SDH after vs before implementation of the curriculum (mean [SD], 3.5 [1.06] before vs 4.0 [1.16] after)(P>.05), though this difference was not statistically learners’ awareness of challenges faced by every patient population, from a mean (SD) of 3.12 (0.66) to 4.52 (0.69)(P<.05). Learners were more comfortable handling hypothetical clinical scenarios requiring them to identify and address specific SDH after vs before implementation of the curriculum (mean [SD], 3.5 [1.06] before vs 4.0 [1.16] after)(P>.05), though this difference was not statistically significant. Finally, many respondents expressed appreciation that our curriculum improved their ability to care for patients in complex social circumstances. Residents suggested in the free-text responses that learning more about the historical underpinnings of health disparities, opportunities for grassroots activism, and how to provide more culturally competent care of Native American populations could improve our curriculum.
Implications for Dermatology Training
Our survey results indicate that a formal SDH curriculum can improve dermatology residents’ ability to care for populations with complex social needs. We advocate for implementing SDH curricula into dermatology training programs nationwide, as has been recommended by others.13,14 We also propose that structural competency should eventually be a key dermatologic competency as determined by the Accreditation Council for Graduate Medical Education, in line with the American Medical Association’s recommendation that structural competency is a learned skill required to end health inequity.15 The Accreditation Council for Graduate Medical Education specialty program requirements currently are being revised; interested individuals can engage in this process by submitting this suggestion for public comment (https://www.acgme.org/programs-and-institutions/programs/review-and-comment/).
Limitations of a survey include the relatively small sample size (7-8 per year) and variable response rates. In addition, we did not survey each class of residents at the beginning and end of their training; our comparisons therefore were limited by comparing different individuals with distinct backgrounds and experiences. Furthermore, we acknowledge that the experience of developing this curriculum in San Francisco may be distinct from other communities, where access to dermatologic care may vary according to both the availability of public health insurance and the treatments covered by public insurers. In San Francisco, insurance coverage is near universal, such that residents in our training program regularly care for undocumented immigrants, persons experiencing homelessness, and other populations that might find it challenging to present to dermatology clinics in other settings nationwide.
Final Thoughts
Future directions of our curriculum include exploration of novel curriculum delivery methods (including a problem-based curriculum approach and other more experiential didactics), increased opportunities for community engagement, greater focus on advocacy with an emphasis on broader social and structural policies and their downstream effects, and focusing more specifically on the history and needs of specific low-income San Francisco neighborhoods and diverse patient populations.
- US Department of Health and Human Services. Health Equity in Healthy People 2030. Accessed January 7, 2025. https://odphp.healtwh.gov/healthypeople/priority-areas/health-equity-healthy-people-2030
- Axelson DJ, Stull MJ, Coates WC. Social determinants of health: a missing link in emergency medicine training. AEM Educ Train. 2018;2:66-68. doi:10.1002/aet2.10056
- Brenner AM, Guerrero APS, Beresin EV, et al. Teaching medical students and residents about homelessness: complex, evidence-based, and imperative. Acad Psychiatry. 2016;40:572-575. doi:10.1007/s40596-016-0571-6
- Youngclaus J, Roskovensky L. An Updated Look at the Economic Diversity of U.S. Medical Students. American Association of Medical Colleges Analysis in Brief. 2018;18. https://www.aamc.org/media/9596/download?attachment
- Shahriar AA, Puram VV, Miller JM, et al. Socioeconomic diversity of the matriculating US medical student body by race, ethnicity, and sex, 2017-2019. JAMA Netw Open. 2022;5:E222621. doi:10.1001/jamanetworkopen.2022.2621
- Williams JC, Maxey AE, Wei ML, et al. A cross-sectional analysis of Medicaid acceptance among US dermatology residency training programs. J Am Acad Dermatol. 2022;86:453-455. doi:10.1016/j.jaad.2021.09.046
- Daniel H, Bornstein S, Kane G; Health and Public Policy Committee of the American College of Physicians. Addressing social determinants to improve patient care and promote health equity: an American College of Physicians position paper. Ann Intern Med. 2018;168:577-578. doi:10.2105/AJPH
- Hansen H, Kline N, Braslow J, et al. From cultural to structural competency—training psychiatry residents to act on social determinants of health and institutional racism. JAMA Psychiatry. 2018;75:117-118. doi:10.1001/jamapsychiatry.2017.3894
- Schmidt S, Higgins S, George M, et al. An experiential resident module for understanding social determinants of health at an academic safety-net hospital. MedEdPORTAL. 2017;26:10647. doi:10.15766/mep_2374-8265.10647
- Hoffman BD, Rose J, Best D, et al. The community pediatrics training initiative project planning tool: a practical approach to community-based advocacy. MedEdPORTAL. 2017;13:10630.
- Chrisman-Khawam L, Abdullah N, Dhoopar A. Teaching health-care trainees empathy and homelessness IQ through service learning, reflective practice, and altruistic attribution. Int J Psychiatry Med. 2017;52:245-254. doi:10.1177/0091217417730288
- Sanchez-Anguiano ME, Klufas D, Amerson E. Screening for cardiometabolic risk factors in patients with psoriasis and hidradenitis suppurativa: a pilot study in a safety net population. J Am Acad Dermatol. 2024;91:1269-1272. doi:10.1016/j.jaad.2024.07.1518
- Riley C, Vasquez R, Pritchett EN. Equipping dermatologists to address structural and social drivers of inequities—structural competency. JAMA Dermatol. 2024;160:1037-1038. doi:10.1001/jamadermatol.2024.2351
- Crawl-Bey A, Pritchett EN, Riley C. 54338 Structural competency in dermatology: a pilot curriculum for equipping residents to address structural factors that contribute to health inequity. J Am Acad Dermatol. 2024;91(3 suppl):AB318. doi:10.1016/j.jaad.2024.07.1264
- Smith TM. New competency focus involves structural factors in health. American Medical Association. December 18, 202. Accessed March 23, 2026. https://www.ama-assn.org/education/changemeded-initiative/new-competency-focus-involves-structural-factors-health
- US Department of Health and Human Services. Health Equity in Healthy People 2030. Accessed January 7, 2025. https://odphp.healtwh.gov/healthypeople/priority-areas/health-equity-healthy-people-2030
- Axelson DJ, Stull MJ, Coates WC. Social determinants of health: a missing link in emergency medicine training. AEM Educ Train. 2018;2:66-68. doi:10.1002/aet2.10056
- Brenner AM, Guerrero APS, Beresin EV, et al. Teaching medical students and residents about homelessness: complex, evidence-based, and imperative. Acad Psychiatry. 2016;40:572-575. doi:10.1007/s40596-016-0571-6
- Youngclaus J, Roskovensky L. An Updated Look at the Economic Diversity of U.S. Medical Students. American Association of Medical Colleges Analysis in Brief. 2018;18. https://www.aamc.org/media/9596/download?attachment
- Shahriar AA, Puram VV, Miller JM, et al. Socioeconomic diversity of the matriculating US medical student body by race, ethnicity, and sex, 2017-2019. JAMA Netw Open. 2022;5:E222621. doi:10.1001/jamanetworkopen.2022.2621
- Williams JC, Maxey AE, Wei ML, et al. A cross-sectional analysis of Medicaid acceptance among US dermatology residency training programs. J Am Acad Dermatol. 2022;86:453-455. doi:10.1016/j.jaad.2021.09.046
- Daniel H, Bornstein S, Kane G; Health and Public Policy Committee of the American College of Physicians. Addressing social determinants to improve patient care and promote health equity: an American College of Physicians position paper. Ann Intern Med. 2018;168:577-578. doi:10.2105/AJPH
- Hansen H, Kline N, Braslow J, et al. From cultural to structural competency—training psychiatry residents to act on social determinants of health and institutional racism. JAMA Psychiatry. 2018;75:117-118. doi:10.1001/jamapsychiatry.2017.3894
- Schmidt S, Higgins S, George M, et al. An experiential resident module for understanding social determinants of health at an academic safety-net hospital. MedEdPORTAL. 2017;26:10647. doi:10.15766/mep_2374-8265.10647
- Hoffman BD, Rose J, Best D, et al. The community pediatrics training initiative project planning tool: a practical approach to community-based advocacy. MedEdPORTAL. 2017;13:10630.
- Chrisman-Khawam L, Abdullah N, Dhoopar A. Teaching health-care trainees empathy and homelessness IQ through service learning, reflective practice, and altruistic attribution. Int J Psychiatry Med. 2017;52:245-254. doi:10.1177/0091217417730288
- Sanchez-Anguiano ME, Klufas D, Amerson E. Screening for cardiometabolic risk factors in patients with psoriasis and hidradenitis suppurativa: a pilot study in a safety net population. J Am Acad Dermatol. 2024;91:1269-1272. doi:10.1016/j.jaad.2024.07.1518
- Riley C, Vasquez R, Pritchett EN. Equipping dermatologists to address structural and social drivers of inequities—structural competency. JAMA Dermatol. 2024;160:1037-1038. doi:10.1001/jamadermatol.2024.2351
- Crawl-Bey A, Pritchett EN, Riley C. 54338 Structural competency in dermatology: a pilot curriculum for equipping residents to address structural factors that contribute to health inequity. J Am Acad Dermatol. 2024;91(3 suppl):AB318. doi:10.1016/j.jaad.2024.07.1264
- Smith TM. New competency focus involves structural factors in health. American Medical Association. December 18, 202. Accessed March 23, 2026. https://www.ama-assn.org/education/changemeded-initiative/new-competency-focus-involves-structural-factors-health
Social Drivers of Health Curriculum for Dermatology Residents: the UCSF Experience
Social Drivers of Health Curriculum for Dermatology Residents: the UCSF Experience
Practice Points
- Integrating a formal curriculum on social drivers of health, including didactics on structural racism, cultural humility, communication, and practical strategies, can help residents learn to routinely assess social needs and develop feasible patient-centered care plans.
- Classroom learning paired with experiential rotations and community engagement in safety-net hospitals can help to build empathy, practical skills, and comfort managing real-world social barriers.
- Creation of trainee leadership roles and dedicated program support (eg, departmental funding) can sustain curriculum improvements, foster advocacy skills, and diversify the workforce pipeline.
Choosing the Right Electronic Medical Record: Key Features and Considerations for Dermatology Practices
Choosing the Right Electronic Medical Record: Key Features and Considerations for Dermatology Practices
Choosing an electronic medical record (EMR) is one of the most important clinical and financial decisions a dermatology practice will make. An effective system can help streamline workflows, support high-quality patient care, and protect revenue, while the wrong choice can slow clinicians down and add to the administrative burden.
Dermatology workflows involve unique documentation, imaging, and billing needs that are not always well served by generic EMR platforms. To help guide the selection of an EMR, the following framework outlines key features and practice considerations specific to dermatology practices.
While many general EMRs offer customization, dermatology practices benefit greatly from ready-built, specialty-specific documentation tools. Key elements to evaluate include the following:
- Preconfigured dermatology templates for common conditions and procedures (eg, acne, psoriasis, melanoma, biopsies, cosmetic treatments)
- Smart-phrase libraries tailored to dermatologic language and examinations
- Ability to create, modify, and share custom templates across providers
Why It Matters—Efficient charting reduces documentation time, improves consistency, and supports accurate coding.
Dermatology is a highly visual specialty, making clinical photography and image management essential. Important capabilities of an EMR include the following:
- Easy capture, annotation, and longitudinal tracking of clinical images
- Seamless embedding of photographs directly into the patient chart
- Side-by-side comparison of current and prior images
- Secure image storage and camera integration
- Body-mapping tools to mark and track lesion locations visually
Why It Matters—A high-quality image workflow supports diagnosis, treatment planning, patient education, and medicolegal documentation.
For insurance-based practices, robust billing and revenue cycle management (RCM) tools are critical. For direct-care models, some of these items may be prioritized lower. Key features to compare include the following:
- Support for International Classification of Diseases, 10th Revision, Clinical Modification; Current Procedural Terminology; and dermatology-specific code sets
- Automated coding suggestions tied to clinical documentation
- Reviewing claims for errors and inconsistencies prior to submitting to payers’ insurance eligibility verification
- Electronic Remittance Advice/Explanation of Benefits posting and denial management workflows
- Support for cosmetic and self-pay billing
- Ability to generate superbills (itemized receipts for medical services that include International Classification of Diseases Tenth Revision and Current Procedural Terminology codes; patients can submit these directly to their insurance company for reimbursement) for direct-pay practices
Why It Matters—Strong RCM functionality protects revenue, reduces denials, and minimizes staff workload.
The most effective EMRs tightly integrate clinical charting with daily practice operations. Features to evaluate include the following:
- Integrated scheduling with color-coded calendars
- Appointment-type templates and block scheduling
- Automated patient reminders via text or email
- Support for multiprovider and multilocation practices
- Integration with outside pathology or lab services
Why It Matters—Clear and templated scheduling and practice integration help practices run more smoothly by reducing administrative workload and errors and coordinating communication between providers and even ancillary services.
Patient communication and virtual care are increasingly important in dermatology. When evaluating EMRs, compare the following:
- Built-in telehealth functionality vs third-party integrations
- Automated appointment reminders
- Patient portal features (forms, messaging, results)
- Online booking capabilities
Why It Matters—Integrated telehealth and patient communication tools improve access to care, enhance patient engagement, and streamline scheduling, messaging, and virtual visits within dermatology workflows.
Reporting capabilities support clinical quality, compliance, and business decision-making. Key reporting areas include the following:
- Clinical reports (outcomes, lesion tracking, disease management)
- Financial reports (revenue per provider, payer mix, visit types)
- Customizable or exportable reporting tools
Why It Matters—Robust reporting and analytics help dermatology practices track clinical outcomes, monitor financial performance, and make data-driven decisions to improve both patient care and practice management.
The user experience after implementation of the EMR is just as important as the software itself. Evaluate the following after the EMR is implemented:
- Initial training and onboarding resources
- Availability of dermatology-specific support teams
- Ongoing education, help centers, or user communities
- Access to dedicated implementation or success managers
Why It Matters—Strong training and support resources help ensure a smoother EMR implementation, faster staff adoption, and ongoing optimization of the system for dermatology workflows.
Finally, look beyond the sticker price. The total cost of ownership includes far more than monthly fees. Compare the following:
- Upfront costs (implementation, data migration, training)
- Subscription pricing (per provider or per user)
- Billing or RCM fees (including percentages of collections if applicable) and payment processing fees
- Costs for add-on modules (telehealth, imaging, analytics)
- Contract length and termination terms
Why It Matters—Understanding the full cost of ownership helps dermatology practices choose an EMR that fits their budget long-term while avoiding unexpected fees and contractual limitations.
There is no single “best” EMR for every dermatology practice. The right choice depends on your practice model, payer mix, clinical focus, and growth plans. By evaluating EMRs through a dermatology-specific lens and asking the right questions, you can choose a system that supports both excellent patient care and long-term practice success.
Choosing an electronic medical record (EMR) is one of the most important clinical and financial decisions a dermatology practice will make. An effective system can help streamline workflows, support high-quality patient care, and protect revenue, while the wrong choice can slow clinicians down and add to the administrative burden.
Dermatology workflows involve unique documentation, imaging, and billing needs that are not always well served by generic EMR platforms. To help guide the selection of an EMR, the following framework outlines key features and practice considerations specific to dermatology practices.
While many general EMRs offer customization, dermatology practices benefit greatly from ready-built, specialty-specific documentation tools. Key elements to evaluate include the following:
- Preconfigured dermatology templates for common conditions and procedures (eg, acne, psoriasis, melanoma, biopsies, cosmetic treatments)
- Smart-phrase libraries tailored to dermatologic language and examinations
- Ability to create, modify, and share custom templates across providers
Why It Matters—Efficient charting reduces documentation time, improves consistency, and supports accurate coding.
Dermatology is a highly visual specialty, making clinical photography and image management essential. Important capabilities of an EMR include the following:
- Easy capture, annotation, and longitudinal tracking of clinical images
- Seamless embedding of photographs directly into the patient chart
- Side-by-side comparison of current and prior images
- Secure image storage and camera integration
- Body-mapping tools to mark and track lesion locations visually
Why It Matters—A high-quality image workflow supports diagnosis, treatment planning, patient education, and medicolegal documentation.
For insurance-based practices, robust billing and revenue cycle management (RCM) tools are critical. For direct-care models, some of these items may be prioritized lower. Key features to compare include the following:
- Support for International Classification of Diseases, 10th Revision, Clinical Modification; Current Procedural Terminology; and dermatology-specific code sets
- Automated coding suggestions tied to clinical documentation
- Reviewing claims for errors and inconsistencies prior to submitting to payers’ insurance eligibility verification
- Electronic Remittance Advice/Explanation of Benefits posting and denial management workflows
- Support for cosmetic and self-pay billing
- Ability to generate superbills (itemized receipts for medical services that include International Classification of Diseases Tenth Revision and Current Procedural Terminology codes; patients can submit these directly to their insurance company for reimbursement) for direct-pay practices
Why It Matters—Strong RCM functionality protects revenue, reduces denials, and minimizes staff workload.
The most effective EMRs tightly integrate clinical charting with daily practice operations. Features to evaluate include the following:
- Integrated scheduling with color-coded calendars
- Appointment-type templates and block scheduling
- Automated patient reminders via text or email
- Support for multiprovider and multilocation practices
- Integration with outside pathology or lab services
Why It Matters—Clear and templated scheduling and practice integration help practices run more smoothly by reducing administrative workload and errors and coordinating communication between providers and even ancillary services.
Patient communication and virtual care are increasingly important in dermatology. When evaluating EMRs, compare the following:
- Built-in telehealth functionality vs third-party integrations
- Automated appointment reminders
- Patient portal features (forms, messaging, results)
- Online booking capabilities
Why It Matters—Integrated telehealth and patient communication tools improve access to care, enhance patient engagement, and streamline scheduling, messaging, and virtual visits within dermatology workflows.
Reporting capabilities support clinical quality, compliance, and business decision-making. Key reporting areas include the following:
- Clinical reports (outcomes, lesion tracking, disease management)
- Financial reports (revenue per provider, payer mix, visit types)
- Customizable or exportable reporting tools
Why It Matters—Robust reporting and analytics help dermatology practices track clinical outcomes, monitor financial performance, and make data-driven decisions to improve both patient care and practice management.
The user experience after implementation of the EMR is just as important as the software itself. Evaluate the following after the EMR is implemented:
- Initial training and onboarding resources
- Availability of dermatology-specific support teams
- Ongoing education, help centers, or user communities
- Access to dedicated implementation or success managers
Why It Matters—Strong training and support resources help ensure a smoother EMR implementation, faster staff adoption, and ongoing optimization of the system for dermatology workflows.
Finally, look beyond the sticker price. The total cost of ownership includes far more than monthly fees. Compare the following:
- Upfront costs (implementation, data migration, training)
- Subscription pricing (per provider or per user)
- Billing or RCM fees (including percentages of collections if applicable) and payment processing fees
- Costs for add-on modules (telehealth, imaging, analytics)
- Contract length and termination terms
Why It Matters—Understanding the full cost of ownership helps dermatology practices choose an EMR that fits their budget long-term while avoiding unexpected fees and contractual limitations.
There is no single “best” EMR for every dermatology practice. The right choice depends on your practice model, payer mix, clinical focus, and growth plans. By evaluating EMRs through a dermatology-specific lens and asking the right questions, you can choose a system that supports both excellent patient care and long-term practice success.
Choosing an electronic medical record (EMR) is one of the most important clinical and financial decisions a dermatology practice will make. An effective system can help streamline workflows, support high-quality patient care, and protect revenue, while the wrong choice can slow clinicians down and add to the administrative burden.
Dermatology workflows involve unique documentation, imaging, and billing needs that are not always well served by generic EMR platforms. To help guide the selection of an EMR, the following framework outlines key features and practice considerations specific to dermatology practices.
While many general EMRs offer customization, dermatology practices benefit greatly from ready-built, specialty-specific documentation tools. Key elements to evaluate include the following:
- Preconfigured dermatology templates for common conditions and procedures (eg, acne, psoriasis, melanoma, biopsies, cosmetic treatments)
- Smart-phrase libraries tailored to dermatologic language and examinations
- Ability to create, modify, and share custom templates across providers
Why It Matters—Efficient charting reduces documentation time, improves consistency, and supports accurate coding.
Dermatology is a highly visual specialty, making clinical photography and image management essential. Important capabilities of an EMR include the following:
- Easy capture, annotation, and longitudinal tracking of clinical images
- Seamless embedding of photographs directly into the patient chart
- Side-by-side comparison of current and prior images
- Secure image storage and camera integration
- Body-mapping tools to mark and track lesion locations visually
Why It Matters—A high-quality image workflow supports diagnosis, treatment planning, patient education, and medicolegal documentation.
For insurance-based practices, robust billing and revenue cycle management (RCM) tools are critical. For direct-care models, some of these items may be prioritized lower. Key features to compare include the following:
- Support for International Classification of Diseases, 10th Revision, Clinical Modification; Current Procedural Terminology; and dermatology-specific code sets
- Automated coding suggestions tied to clinical documentation
- Reviewing claims for errors and inconsistencies prior to submitting to payers’ insurance eligibility verification
- Electronic Remittance Advice/Explanation of Benefits posting and denial management workflows
- Support for cosmetic and self-pay billing
- Ability to generate superbills (itemized receipts for medical services that include International Classification of Diseases Tenth Revision and Current Procedural Terminology codes; patients can submit these directly to their insurance company for reimbursement) for direct-pay practices
Why It Matters—Strong RCM functionality protects revenue, reduces denials, and minimizes staff workload.
The most effective EMRs tightly integrate clinical charting with daily practice operations. Features to evaluate include the following:
- Integrated scheduling with color-coded calendars
- Appointment-type templates and block scheduling
- Automated patient reminders via text or email
- Support for multiprovider and multilocation practices
- Integration with outside pathology or lab services
Why It Matters—Clear and templated scheduling and practice integration help practices run more smoothly by reducing administrative workload and errors and coordinating communication between providers and even ancillary services.
Patient communication and virtual care are increasingly important in dermatology. When evaluating EMRs, compare the following:
- Built-in telehealth functionality vs third-party integrations
- Automated appointment reminders
- Patient portal features (forms, messaging, results)
- Online booking capabilities
Why It Matters—Integrated telehealth and patient communication tools improve access to care, enhance patient engagement, and streamline scheduling, messaging, and virtual visits within dermatology workflows.
Reporting capabilities support clinical quality, compliance, and business decision-making. Key reporting areas include the following:
- Clinical reports (outcomes, lesion tracking, disease management)
- Financial reports (revenue per provider, payer mix, visit types)
- Customizable or exportable reporting tools
Why It Matters—Robust reporting and analytics help dermatology practices track clinical outcomes, monitor financial performance, and make data-driven decisions to improve both patient care and practice management.
The user experience after implementation of the EMR is just as important as the software itself. Evaluate the following after the EMR is implemented:
- Initial training and onboarding resources
- Availability of dermatology-specific support teams
- Ongoing education, help centers, or user communities
- Access to dedicated implementation or success managers
Why It Matters—Strong training and support resources help ensure a smoother EMR implementation, faster staff adoption, and ongoing optimization of the system for dermatology workflows.
Finally, look beyond the sticker price. The total cost of ownership includes far more than monthly fees. Compare the following:
- Upfront costs (implementation, data migration, training)
- Subscription pricing (per provider or per user)
- Billing or RCM fees (including percentages of collections if applicable) and payment processing fees
- Costs for add-on modules (telehealth, imaging, analytics)
- Contract length and termination terms
Why It Matters—Understanding the full cost of ownership helps dermatology practices choose an EMR that fits their budget long-term while avoiding unexpected fees and contractual limitations.
There is no single “best” EMR for every dermatology practice. The right choice depends on your practice model, payer mix, clinical focus, and growth plans. By evaluating EMRs through a dermatology-specific lens and asking the right questions, you can choose a system that supports both excellent patient care and long-term practice success.
Choosing the Right Electronic Medical Record: Key Features and Considerations for Dermatology Practices
Choosing the Right Electronic Medical Record: Key Features and Considerations for Dermatology Practices
PRACTICE POINTS
- Choosing an electronic medical record (EMR) built for dermatology workflow is a critical part of practice management.
- Features of an EMR that should be evaluated include support for clinical documentation, scheduling and billing, and customer support.
- The proper EMR can reduce administrative tasks and protect practice revenue, but there is no one-size-fits-all option.
Beyond Conventional Paradigms: Rethinking the Evaluation and Management of Melasma
Beyond Conventional Paradigms: Rethinking the Evaluation and Management of Melasma
Melasma is a chronic hyperpigmentation disorder with a high recurrence rate. Advances in pathogenesis, diagnosis, and treatment have shifted dermatologic approaches for both initial management and long-term care. These advances highlight the need to move beyond conventional paradigms for melasma to adopt a more comprehensive and holistic approach to evaluation and management.
Diagnosis: Beyond the Wood Lamp
Diagnosis of melasma is primarily clinical, based on visual examination, with tools such as a Wood lamp (320-400 nm) aiding in classification as epidermal, dermal, or mixed types.1 Dermoscopy is another useful tool that commonly demonstrates a brown reticular pseudonetwork, vascular patterns, and brown clods.2 Reflectance confocal microscopy allows detailed assessment of pigment distribution across skin layers and may serve as a useful diagnostic adjunct.3 Two-photon excitation microscopy also has shown concordance with reflectance confocal microscopy in identifying key pathologic features of melasma and allows quantitative assessment of pigment burden.4,5 Biopsy remains warranted in cases when the diagnosis is unclear.6,7 These advanced tools provide additional options for noninvasive imaging of melasma, which may be useful during both diagnosis and treatment.
Pathogenesis: Beyond the Melanocyte
Recent insights into pathogenesis have shifted the view of melasma from a purely melanocyte-driven disorder to one involving complex epidermal-dermal interactions influenced by chronic UV and visible light exposure and sustained cutaneous inflammation. Evidence suggests involvement of additional cell types and structural components, including epidermal barrier dysfunction, basement membrane alterations, senescent fibroblasts, mast cell activity, vascular remodeling, and dermal solar elastosis. Senescent fibroblasts secrete melanogenic and angiogenic mediators (including stem cell factor, vascular endothelial growth factor, endothelin 1, and matrix metalloproteinases) while mast cell degranulation releases histamine, tryptase, and other proteases that drive melanocyte activation, extracellular matrix degradation, and persistent hyperpigmentation.8 New research highlights the importance of epidermal-dermal communication in melasma disease activity.8,9 Exposure to UV and visible light leads to keratinocyte injury and fibroblast dysfunction, promoting the release of inflammatory and melanogenic mediators that stimulate melanocytes and weaken the basement membrane.8 Mast cell activation and vascular signaling also have been implicated, reinforcing the contribution of inflammation and dermal remodeling to ongoing pigmentation.9 This broader understanding helps explain the difficulty of sustaining long-term remission.8,9 The multifaceted and evolving understanding of melasma’s complex pathogenesis highlights areas for future therapeutic targets beyond melanin, which may help lead to greater clearance and remission periods.
Treatment: Beyond Hydroquinone
Melasma treatment has shifted toward a longitudinal approach reflecting its chronic relapsing nature. Effective management should include combination therapy of broad-spectrum photoprotection, topical therapies, systemic therapies (when appropriate), and adjunctive procedural modalities, while emphasizing the importance of maintenance therapy and relapse prevention.10,11 Growing evidence demonstrates that UV and visible light contribute to melanogenesis and prolonged pigmentation, particularly in individuals with skin of color.12,13 Visible light, particularly within the high-energy blue wavelength spectrum (400-500 nm), induces reactive oxygen species and stimulates melanogenesis, resulting in persistent hyperpigmentation and exacerbation of melasma in darker skin types.13 Iron oxide, a mineral-based physical filter commonly used in tinted mineral sunscreens, enhances protection from visible light, thus improving melasma outcomes.12,13 In a randomized double-blind placebo-controlled trial, oral polypodium leucotomos extract, an antioxidant with photoprotective effects, improved Melasma Area and Severity Index (MASI)/modified MASI (mMASI) scores and melasma-rated quality of life measures when used as an adjunct to sunscreen with or without topical hydroquinone.14
Topical therapy serves as the mainstay of melasma treatment. Hydroquinone- and retinoid-based regimens including triple-combination therapy remain an effective gold standard for active disease; however, there still are concerns about irritant dermatitis with both as well as ochronosis with long-term use of hydroquinone.15 Recent studies highlight the efficacy of nonhydroquinone topical therapies such as azelaic acid, tranexamic acid (TXA), thiamidol, cysteamine, metformin, malassezin, 2-mercaptonicotinoyl glycine, niacinamide, kojic acid, ascorbic acid, and botanical-derived compounds, many of which demonstrate reductions in disease severity and more favorable tolerability profiles.16-19 Combinations of these ingredients can allow for addressing the various pathogenic factors of melasma beyond pigmentation, including vascularity, inflammation, and solar damage.
Systemic therapy, particularly oral TXA, has emerged as an option for patients with moderate to severe or refractory melasma. Clinical trials and observational studies demonstrate improvement with oral TXA; however, relapse after discontinuation is common, emphasizing the importance of careful patient selection, established treatment duration, and concurrent maintenance therapy.16,20 Although it is well tolerated, adverse effects include gastrointestinal discomfort, menstrual irregularities, and headache. Additionally, prescreening for contraindications and clotting risk factors is needed to avoid thromboembolic events.21
Procedural interventions are best used as adjunctive therapies rather than primary treatment. Superficial and medium peels can be used depending on baseline skin type.22 Platelet-rich plasma is a promising adjunctive therapy for melasma, demonstrating reductions in MASI scores following intradermal or microneedling-assisted delivery, likely mediated through growth factor–driven inhibition of melanogenesis and modulation of tyrosinase activity. Platelet-rich plasma promotes dermal remodeling through angiogenesis, collagen synthesis, and extracellular matrix production improving skin texture and pigmentation.23 Botulinum toxin A may improve pigmentation by modulating melanogenesis as well as inflammatory and vascular pathways.24
Radiofrequency microneedling provides a dermal-targeting therapeutic approach, with some evidence showing that monthly treatments can help maintain improvements achieved with conventional therapy by modulating photoaged dermal structures impacted by melanogenic signaling.25 Histopathologic correlation studies suggest that microneedling used alone or as a delivery platform alongside topical agents may be beneficial in dermal-predominant disease, while laser-based therapies should be reserved for recalcitrant cases due to the risk for postinflammatory hyperpigmentation and melasma recurrence, particularly in those with darker skin types.26 Lasers such as Q-switched 1064-nm Nd:YAG as well as nonablative lasers used in conservative settings are options for melasma treatment, including in patients with skin of color.26,27 Laser toning with serial low-fluence (<3 J/cm2), large-spot 1064-nm Nd:YAG has been used to reduce inflammation and risk for postinflammatory hyperpigmentation.28 Additionally, randomized split-face data demonstrate that picosecond 1064-nm devices achieve comparable reductions in mMASI scores to traditional nanosecond devices, and combination strategies incorporating intense pulsed light (IPL) have shown greater MASI improvement compared with IPL alone.28,29 However, IPL generally is limited to lighter skin phototypes due to the risk for postinflammatory hyperpigmentation in darker skin.30
Lastly, long-term management requires patient education on the importance of year-round maintenance therapy, consistency, and compliance. Reinforcing adherence to photoprotection, supporting barrier repair, and setting expectations regarding prognosis are essential components to optimizing melasma treatment outcomes.12
Future Insights: Beyond the Current State of Melasma Research and Treatment
Future advances in melasma management likely will focus on improving disease assessment and long-term treatment durability. Tools such as the MASI and mMASI are widely used but are limited by interobserver variability and reduced sensitivity in darker skin types, highlighting the need to incorporate objective measures such as colorimetry.10,26 There also is a need for studies that relate clinical improvement to underlying pathology. Histopathologic correlation remains limited across most therapeutic trials, and more studies should assess long-term outcomes and quality-of-life measures given the relapsing nature of melasma.26
Future interventions will continue to move past the treatment of pigment alone. Increasing recognition of vascular, inflammatory, and barrier-related contributions to melasma, as well as targeting of other cell types affected by melasma, will be important considerations in future treatment innovations.8,11 In summary, improved inclusion of patients with skin of color in clinical trials, expansion of clinical outcomes measures and objective parameters of disease evaluation, novel approaches to treatment and relapse prevention, and greater attention to patient adherence and education will be essential to achieving advancements in melasma care.12
- Sanchez NP, Pathak MA, Sato S, et al. Melasma: a clinical, light microscopic, ultrastructural, and immunofluorescence study. J Am Acad Dermatol. 1981;4:698-710.
- Bostan E, Cakir A. The dermoscopic characteristics of melasma in relation to different skin phototypes, distribution patterns and wood lamp findings: a cross-sectional study of 236 melasma lesions. Arch Dermatol Res. 2023;315:1927-1938.
- Guida S, Longo C, Ronga R, et al. Melasma and reflectance confocal microscopy: from baseline to treatment monitoring. Int J Dermatol. 2024;63:1007-1012.
- Ning X, Yang J, Ouyang H, et al. In vivo evaluation of melasma pathologic features and treatment response by 2-photon microscopy. JAMA Dermatol. 2025;161:1039-1047.
- Chen K-J, Han Y, Wang Z-Y, et al. Submicron resolution techniques: multiphoton microscopy in skin disease. Exp Dermatol. 2023;32:1613-1623.
- Philipp-Dormston WG. Melasma: a step-by-step approach towards a multimodal combination therapy. Clin Cosmet Investig Dermatol. 2024;17:1203-1216.
- Doolan B, Gupta M. Melasma. Aust J Gen Pract. 2021;50:880-885.
- Ali L, Al Niaimi F. Pathogenesis of melasma explained. Int J Dermatol. 2025;64:1201-1212.
- Artzi O, Horovitz T, Bar-Ilan E, et al. The pathogenesis of melasma and implications for treatment. J Cosmet Dermatol. 2021;20:3432-3445.
- Gan C, Rodrigues M. An update on new and existing treatments for the management of melasma. Am J Clin Dermatol. 2024;25:717-733.
- Sarkar R, Desai SR, Sinha S, et al. Delphi consensus on melasma management by international experts and pigmentary disorders society. J Eur Acad Dermatol Venereol. Published online September 25, 2025.
- Desai SR, Alexis AF, Elbuluk N, et al. Best practices in the treatment of melasma with a focus on patients with skin of color. J Am Acad Dermatol. 2024;90:269-279.
- Lim HW, Kohli I, Ruvolo E, et al. Impact of visible light on skin health: the role of antioxidants and free radical quenchers in skin protection. J Am Acad Deramtol. 2022;86(3 suppl):S27-S37.
- Goh CL, Chuah SY, Tien S, et al. Double-blind, placebo-controlled trial to evaluate the effectiveness of polypodium leucotomos extract in the treatment of melasma in Asian skin: a pilot study. J Clin Aesthet Dermatol. 2018;11:14-19.
- Mahajan VK, Patil A, Blicharz L, et al. Medical therapies for melasma. J Cosmet Dermatol. 2022;21:3707-3728.
- Suliman RS, Alhuwayshil J, Almuflehi AA, et al. Emerging topical therapies for melasma: a comparative analysis of efficacy and safety. J Dermatolog Treat. 2025;36:2591502.
- Austin E, Nguyen JK, Jagdeo J. Topical treatments for melasma: a systematic review of randomized controlled trials. J Drugs Dermatol. 2019;18:S1545961619P1156X.
- Grimes PE, Dias S, Oparaugo NC, et al. A randomized, controlled, split-face, double-blind study comparing topical malassezin to hydroquinone 4% for melasma. J Drugs Dermatol. 2026;25:25-29.
- Passeron T, Kerob D, Le Dantec G, et al. Efficacy and tolerability of a new facial 2-mercaptonicotinoyl glycine-containing depigmenting serum versus hydroquinone 4% over 3-month treatment of facial melasma. Dermatol Ther (Heidelb). 2025;15:2379-2390.
- McKesey J, Tovar-Garza A, Pandya AG. Melasma treatment: an evidence-based review. Am J Clin Dermatol. 2020;21:173-225.
- Bala HR, Lee S, Wong C, et al. Oral tranexamic acid for the treatment of melasma: a review. Dermatol Surg. 2018;44:814-825.
- Sarkar R, Katoch S. Chemical peels in treatment of melasma. Dermatol Clin. 2024;42:21-32.
- Sarkar R, Gupta M. Platelet-rich plasma in melasma—a systematic review. Dermatol Surg. 2022;48:131-134.
- Pongklaokam J, Manuskiatti W, Wanitphakdeedecha R, et al. Efficacy and safety of letibotulinum toxin A for the treatment of melasma in two different dilutions: a randomized double-blind split-face study. Toxins (Basel). 2025;17:349.
- Han HJ, Kim JC, Park YJ, et al. Targeting the dermis for melasma maintenance treatment. Sci Rep. 2024;14:949.
- Zhang AD, Lazar M, Akhundova E, et al. A scoping review on melasma treatments and their histopathologic correlates. Dermatopathology (Basel). 2025;12:13.
- Goldman MP, Wu DC, Khetarpal S, et al. Dual 1550-nm erbium glass fiber and 1927-nm thulium fiber non-ablative fractional laser system treatment in patients with skin of color: a review of clinical studies and unmet needs. Lasers Surg Med. Published online February 10, 2026.
- Chen J, Yu N, Peng L, et al. Efficacy of low-fluence 1064 nm Q-switched Nd: YAG laser for the treatment of melasma: a meta-analysis and systematic review. J Cosmet Dermatol. 2022;21.
- Feng J, Huang L. Comparison of picosecond and nanosecond Nd:YAG 1064-nm lasers in the treatment of melasma: a split-face randomized clinical trial. Plast Reconstr Surg. 2023;151:772-777.
- Passeron T, Genedy R, Salah L, et al., Laser treatment of hyperpigmented lesions: position statement of the European Society of Laser in Dermatology. J Eur Acad Dermatol Venereol. 2019;33:987-1005.
Melasma is a chronic hyperpigmentation disorder with a high recurrence rate. Advances in pathogenesis, diagnosis, and treatment have shifted dermatologic approaches for both initial management and long-term care. These advances highlight the need to move beyond conventional paradigms for melasma to adopt a more comprehensive and holistic approach to evaluation and management.
Diagnosis: Beyond the Wood Lamp
Diagnosis of melasma is primarily clinical, based on visual examination, with tools such as a Wood lamp (320-400 nm) aiding in classification as epidermal, dermal, or mixed types.1 Dermoscopy is another useful tool that commonly demonstrates a brown reticular pseudonetwork, vascular patterns, and brown clods.2 Reflectance confocal microscopy allows detailed assessment of pigment distribution across skin layers and may serve as a useful diagnostic adjunct.3 Two-photon excitation microscopy also has shown concordance with reflectance confocal microscopy in identifying key pathologic features of melasma and allows quantitative assessment of pigment burden.4,5 Biopsy remains warranted in cases when the diagnosis is unclear.6,7 These advanced tools provide additional options for noninvasive imaging of melasma, which may be useful during both diagnosis and treatment.
Pathogenesis: Beyond the Melanocyte
Recent insights into pathogenesis have shifted the view of melasma from a purely melanocyte-driven disorder to one involving complex epidermal-dermal interactions influenced by chronic UV and visible light exposure and sustained cutaneous inflammation. Evidence suggests involvement of additional cell types and structural components, including epidermal barrier dysfunction, basement membrane alterations, senescent fibroblasts, mast cell activity, vascular remodeling, and dermal solar elastosis. Senescent fibroblasts secrete melanogenic and angiogenic mediators (including stem cell factor, vascular endothelial growth factor, endothelin 1, and matrix metalloproteinases) while mast cell degranulation releases histamine, tryptase, and other proteases that drive melanocyte activation, extracellular matrix degradation, and persistent hyperpigmentation.8 New research highlights the importance of epidermal-dermal communication in melasma disease activity.8,9 Exposure to UV and visible light leads to keratinocyte injury and fibroblast dysfunction, promoting the release of inflammatory and melanogenic mediators that stimulate melanocytes and weaken the basement membrane.8 Mast cell activation and vascular signaling also have been implicated, reinforcing the contribution of inflammation and dermal remodeling to ongoing pigmentation.9 This broader understanding helps explain the difficulty of sustaining long-term remission.8,9 The multifaceted and evolving understanding of melasma’s complex pathogenesis highlights areas for future therapeutic targets beyond melanin, which may help lead to greater clearance and remission periods.
Treatment: Beyond Hydroquinone
Melasma treatment has shifted toward a longitudinal approach reflecting its chronic relapsing nature. Effective management should include combination therapy of broad-spectrum photoprotection, topical therapies, systemic therapies (when appropriate), and adjunctive procedural modalities, while emphasizing the importance of maintenance therapy and relapse prevention.10,11 Growing evidence demonstrates that UV and visible light contribute to melanogenesis and prolonged pigmentation, particularly in individuals with skin of color.12,13 Visible light, particularly within the high-energy blue wavelength spectrum (400-500 nm), induces reactive oxygen species and stimulates melanogenesis, resulting in persistent hyperpigmentation and exacerbation of melasma in darker skin types.13 Iron oxide, a mineral-based physical filter commonly used in tinted mineral sunscreens, enhances protection from visible light, thus improving melasma outcomes.12,13 In a randomized double-blind placebo-controlled trial, oral polypodium leucotomos extract, an antioxidant with photoprotective effects, improved Melasma Area and Severity Index (MASI)/modified MASI (mMASI) scores and melasma-rated quality of life measures when used as an adjunct to sunscreen with or without topical hydroquinone.14
Topical therapy serves as the mainstay of melasma treatment. Hydroquinone- and retinoid-based regimens including triple-combination therapy remain an effective gold standard for active disease; however, there still are concerns about irritant dermatitis with both as well as ochronosis with long-term use of hydroquinone.15 Recent studies highlight the efficacy of nonhydroquinone topical therapies such as azelaic acid, tranexamic acid (TXA), thiamidol, cysteamine, metformin, malassezin, 2-mercaptonicotinoyl glycine, niacinamide, kojic acid, ascorbic acid, and botanical-derived compounds, many of which demonstrate reductions in disease severity and more favorable tolerability profiles.16-19 Combinations of these ingredients can allow for addressing the various pathogenic factors of melasma beyond pigmentation, including vascularity, inflammation, and solar damage.
Systemic therapy, particularly oral TXA, has emerged as an option for patients with moderate to severe or refractory melasma. Clinical trials and observational studies demonstrate improvement with oral TXA; however, relapse after discontinuation is common, emphasizing the importance of careful patient selection, established treatment duration, and concurrent maintenance therapy.16,20 Although it is well tolerated, adverse effects include gastrointestinal discomfort, menstrual irregularities, and headache. Additionally, prescreening for contraindications and clotting risk factors is needed to avoid thromboembolic events.21
Procedural interventions are best used as adjunctive therapies rather than primary treatment. Superficial and medium peels can be used depending on baseline skin type.22 Platelet-rich plasma is a promising adjunctive therapy for melasma, demonstrating reductions in MASI scores following intradermal or microneedling-assisted delivery, likely mediated through growth factor–driven inhibition of melanogenesis and modulation of tyrosinase activity. Platelet-rich plasma promotes dermal remodeling through angiogenesis, collagen synthesis, and extracellular matrix production improving skin texture and pigmentation.23 Botulinum toxin A may improve pigmentation by modulating melanogenesis as well as inflammatory and vascular pathways.24
Radiofrequency microneedling provides a dermal-targeting therapeutic approach, with some evidence showing that monthly treatments can help maintain improvements achieved with conventional therapy by modulating photoaged dermal structures impacted by melanogenic signaling.25 Histopathologic correlation studies suggest that microneedling used alone or as a delivery platform alongside topical agents may be beneficial in dermal-predominant disease, while laser-based therapies should be reserved for recalcitrant cases due to the risk for postinflammatory hyperpigmentation and melasma recurrence, particularly in those with darker skin types.26 Lasers such as Q-switched 1064-nm Nd:YAG as well as nonablative lasers used in conservative settings are options for melasma treatment, including in patients with skin of color.26,27 Laser toning with serial low-fluence (<3 J/cm2), large-spot 1064-nm Nd:YAG has been used to reduce inflammation and risk for postinflammatory hyperpigmentation.28 Additionally, randomized split-face data demonstrate that picosecond 1064-nm devices achieve comparable reductions in mMASI scores to traditional nanosecond devices, and combination strategies incorporating intense pulsed light (IPL) have shown greater MASI improvement compared with IPL alone.28,29 However, IPL generally is limited to lighter skin phototypes due to the risk for postinflammatory hyperpigmentation in darker skin.30
Lastly, long-term management requires patient education on the importance of year-round maintenance therapy, consistency, and compliance. Reinforcing adherence to photoprotection, supporting barrier repair, and setting expectations regarding prognosis are essential components to optimizing melasma treatment outcomes.12
Future Insights: Beyond the Current State of Melasma Research and Treatment
Future advances in melasma management likely will focus on improving disease assessment and long-term treatment durability. Tools such as the MASI and mMASI are widely used but are limited by interobserver variability and reduced sensitivity in darker skin types, highlighting the need to incorporate objective measures such as colorimetry.10,26 There also is a need for studies that relate clinical improvement to underlying pathology. Histopathologic correlation remains limited across most therapeutic trials, and more studies should assess long-term outcomes and quality-of-life measures given the relapsing nature of melasma.26
Future interventions will continue to move past the treatment of pigment alone. Increasing recognition of vascular, inflammatory, and barrier-related contributions to melasma, as well as targeting of other cell types affected by melasma, will be important considerations in future treatment innovations.8,11 In summary, improved inclusion of patients with skin of color in clinical trials, expansion of clinical outcomes measures and objective parameters of disease evaluation, novel approaches to treatment and relapse prevention, and greater attention to patient adherence and education will be essential to achieving advancements in melasma care.12
Melasma is a chronic hyperpigmentation disorder with a high recurrence rate. Advances in pathogenesis, diagnosis, and treatment have shifted dermatologic approaches for both initial management and long-term care. These advances highlight the need to move beyond conventional paradigms for melasma to adopt a more comprehensive and holistic approach to evaluation and management.
Diagnosis: Beyond the Wood Lamp
Diagnosis of melasma is primarily clinical, based on visual examination, with tools such as a Wood lamp (320-400 nm) aiding in classification as epidermal, dermal, or mixed types.1 Dermoscopy is another useful tool that commonly demonstrates a brown reticular pseudonetwork, vascular patterns, and brown clods.2 Reflectance confocal microscopy allows detailed assessment of pigment distribution across skin layers and may serve as a useful diagnostic adjunct.3 Two-photon excitation microscopy also has shown concordance with reflectance confocal microscopy in identifying key pathologic features of melasma and allows quantitative assessment of pigment burden.4,5 Biopsy remains warranted in cases when the diagnosis is unclear.6,7 These advanced tools provide additional options for noninvasive imaging of melasma, which may be useful during both diagnosis and treatment.
Pathogenesis: Beyond the Melanocyte
Recent insights into pathogenesis have shifted the view of melasma from a purely melanocyte-driven disorder to one involving complex epidermal-dermal interactions influenced by chronic UV and visible light exposure and sustained cutaneous inflammation. Evidence suggests involvement of additional cell types and structural components, including epidermal barrier dysfunction, basement membrane alterations, senescent fibroblasts, mast cell activity, vascular remodeling, and dermal solar elastosis. Senescent fibroblasts secrete melanogenic and angiogenic mediators (including stem cell factor, vascular endothelial growth factor, endothelin 1, and matrix metalloproteinases) while mast cell degranulation releases histamine, tryptase, and other proteases that drive melanocyte activation, extracellular matrix degradation, and persistent hyperpigmentation.8 New research highlights the importance of epidermal-dermal communication in melasma disease activity.8,9 Exposure to UV and visible light leads to keratinocyte injury and fibroblast dysfunction, promoting the release of inflammatory and melanogenic mediators that stimulate melanocytes and weaken the basement membrane.8 Mast cell activation and vascular signaling also have been implicated, reinforcing the contribution of inflammation and dermal remodeling to ongoing pigmentation.9 This broader understanding helps explain the difficulty of sustaining long-term remission.8,9 The multifaceted and evolving understanding of melasma’s complex pathogenesis highlights areas for future therapeutic targets beyond melanin, which may help lead to greater clearance and remission periods.
Treatment: Beyond Hydroquinone
Melasma treatment has shifted toward a longitudinal approach reflecting its chronic relapsing nature. Effective management should include combination therapy of broad-spectrum photoprotection, topical therapies, systemic therapies (when appropriate), and adjunctive procedural modalities, while emphasizing the importance of maintenance therapy and relapse prevention.10,11 Growing evidence demonstrates that UV and visible light contribute to melanogenesis and prolonged pigmentation, particularly in individuals with skin of color.12,13 Visible light, particularly within the high-energy blue wavelength spectrum (400-500 nm), induces reactive oxygen species and stimulates melanogenesis, resulting in persistent hyperpigmentation and exacerbation of melasma in darker skin types.13 Iron oxide, a mineral-based physical filter commonly used in tinted mineral sunscreens, enhances protection from visible light, thus improving melasma outcomes.12,13 In a randomized double-blind placebo-controlled trial, oral polypodium leucotomos extract, an antioxidant with photoprotective effects, improved Melasma Area and Severity Index (MASI)/modified MASI (mMASI) scores and melasma-rated quality of life measures when used as an adjunct to sunscreen with or without topical hydroquinone.14
Topical therapy serves as the mainstay of melasma treatment. Hydroquinone- and retinoid-based regimens including triple-combination therapy remain an effective gold standard for active disease; however, there still are concerns about irritant dermatitis with both as well as ochronosis with long-term use of hydroquinone.15 Recent studies highlight the efficacy of nonhydroquinone topical therapies such as azelaic acid, tranexamic acid (TXA), thiamidol, cysteamine, metformin, malassezin, 2-mercaptonicotinoyl glycine, niacinamide, kojic acid, ascorbic acid, and botanical-derived compounds, many of which demonstrate reductions in disease severity and more favorable tolerability profiles.16-19 Combinations of these ingredients can allow for addressing the various pathogenic factors of melasma beyond pigmentation, including vascularity, inflammation, and solar damage.
Systemic therapy, particularly oral TXA, has emerged as an option for patients with moderate to severe or refractory melasma. Clinical trials and observational studies demonstrate improvement with oral TXA; however, relapse after discontinuation is common, emphasizing the importance of careful patient selection, established treatment duration, and concurrent maintenance therapy.16,20 Although it is well tolerated, adverse effects include gastrointestinal discomfort, menstrual irregularities, and headache. Additionally, prescreening for contraindications and clotting risk factors is needed to avoid thromboembolic events.21
Procedural interventions are best used as adjunctive therapies rather than primary treatment. Superficial and medium peels can be used depending on baseline skin type.22 Platelet-rich plasma is a promising adjunctive therapy for melasma, demonstrating reductions in MASI scores following intradermal or microneedling-assisted delivery, likely mediated through growth factor–driven inhibition of melanogenesis and modulation of tyrosinase activity. Platelet-rich plasma promotes dermal remodeling through angiogenesis, collagen synthesis, and extracellular matrix production improving skin texture and pigmentation.23 Botulinum toxin A may improve pigmentation by modulating melanogenesis as well as inflammatory and vascular pathways.24
Radiofrequency microneedling provides a dermal-targeting therapeutic approach, with some evidence showing that monthly treatments can help maintain improvements achieved with conventional therapy by modulating photoaged dermal structures impacted by melanogenic signaling.25 Histopathologic correlation studies suggest that microneedling used alone or as a delivery platform alongside topical agents may be beneficial in dermal-predominant disease, while laser-based therapies should be reserved for recalcitrant cases due to the risk for postinflammatory hyperpigmentation and melasma recurrence, particularly in those with darker skin types.26 Lasers such as Q-switched 1064-nm Nd:YAG as well as nonablative lasers used in conservative settings are options for melasma treatment, including in patients with skin of color.26,27 Laser toning with serial low-fluence (<3 J/cm2), large-spot 1064-nm Nd:YAG has been used to reduce inflammation and risk for postinflammatory hyperpigmentation.28 Additionally, randomized split-face data demonstrate that picosecond 1064-nm devices achieve comparable reductions in mMASI scores to traditional nanosecond devices, and combination strategies incorporating intense pulsed light (IPL) have shown greater MASI improvement compared with IPL alone.28,29 However, IPL generally is limited to lighter skin phototypes due to the risk for postinflammatory hyperpigmentation in darker skin.30
Lastly, long-term management requires patient education on the importance of year-round maintenance therapy, consistency, and compliance. Reinforcing adherence to photoprotection, supporting barrier repair, and setting expectations regarding prognosis are essential components to optimizing melasma treatment outcomes.12
Future Insights: Beyond the Current State of Melasma Research and Treatment
Future advances in melasma management likely will focus on improving disease assessment and long-term treatment durability. Tools such as the MASI and mMASI are widely used but are limited by interobserver variability and reduced sensitivity in darker skin types, highlighting the need to incorporate objective measures such as colorimetry.10,26 There also is a need for studies that relate clinical improvement to underlying pathology. Histopathologic correlation remains limited across most therapeutic trials, and more studies should assess long-term outcomes and quality-of-life measures given the relapsing nature of melasma.26
Future interventions will continue to move past the treatment of pigment alone. Increasing recognition of vascular, inflammatory, and barrier-related contributions to melasma, as well as targeting of other cell types affected by melasma, will be important considerations in future treatment innovations.8,11 In summary, improved inclusion of patients with skin of color in clinical trials, expansion of clinical outcomes measures and objective parameters of disease evaluation, novel approaches to treatment and relapse prevention, and greater attention to patient adherence and education will be essential to achieving advancements in melasma care.12
- Sanchez NP, Pathak MA, Sato S, et al. Melasma: a clinical, light microscopic, ultrastructural, and immunofluorescence study. J Am Acad Dermatol. 1981;4:698-710.
- Bostan E, Cakir A. The dermoscopic characteristics of melasma in relation to different skin phototypes, distribution patterns and wood lamp findings: a cross-sectional study of 236 melasma lesions. Arch Dermatol Res. 2023;315:1927-1938.
- Guida S, Longo C, Ronga R, et al. Melasma and reflectance confocal microscopy: from baseline to treatment monitoring. Int J Dermatol. 2024;63:1007-1012.
- Ning X, Yang J, Ouyang H, et al. In vivo evaluation of melasma pathologic features and treatment response by 2-photon microscopy. JAMA Dermatol. 2025;161:1039-1047.
- Chen K-J, Han Y, Wang Z-Y, et al. Submicron resolution techniques: multiphoton microscopy in skin disease. Exp Dermatol. 2023;32:1613-1623.
- Philipp-Dormston WG. Melasma: a step-by-step approach towards a multimodal combination therapy. Clin Cosmet Investig Dermatol. 2024;17:1203-1216.
- Doolan B, Gupta M. Melasma. Aust J Gen Pract. 2021;50:880-885.
- Ali L, Al Niaimi F. Pathogenesis of melasma explained. Int J Dermatol. 2025;64:1201-1212.
- Artzi O, Horovitz T, Bar-Ilan E, et al. The pathogenesis of melasma and implications for treatment. J Cosmet Dermatol. 2021;20:3432-3445.
- Gan C, Rodrigues M. An update on new and existing treatments for the management of melasma. Am J Clin Dermatol. 2024;25:717-733.
- Sarkar R, Desai SR, Sinha S, et al. Delphi consensus on melasma management by international experts and pigmentary disorders society. J Eur Acad Dermatol Venereol. Published online September 25, 2025.
- Desai SR, Alexis AF, Elbuluk N, et al. Best practices in the treatment of melasma with a focus on patients with skin of color. J Am Acad Dermatol. 2024;90:269-279.
- Lim HW, Kohli I, Ruvolo E, et al. Impact of visible light on skin health: the role of antioxidants and free radical quenchers in skin protection. J Am Acad Deramtol. 2022;86(3 suppl):S27-S37.
- Goh CL, Chuah SY, Tien S, et al. Double-blind, placebo-controlled trial to evaluate the effectiveness of polypodium leucotomos extract in the treatment of melasma in Asian skin: a pilot study. J Clin Aesthet Dermatol. 2018;11:14-19.
- Mahajan VK, Patil A, Blicharz L, et al. Medical therapies for melasma. J Cosmet Dermatol. 2022;21:3707-3728.
- Suliman RS, Alhuwayshil J, Almuflehi AA, et al. Emerging topical therapies for melasma: a comparative analysis of efficacy and safety. J Dermatolog Treat. 2025;36:2591502.
- Austin E, Nguyen JK, Jagdeo J. Topical treatments for melasma: a systematic review of randomized controlled trials. J Drugs Dermatol. 2019;18:S1545961619P1156X.
- Grimes PE, Dias S, Oparaugo NC, et al. A randomized, controlled, split-face, double-blind study comparing topical malassezin to hydroquinone 4% for melasma. J Drugs Dermatol. 2026;25:25-29.
- Passeron T, Kerob D, Le Dantec G, et al. Efficacy and tolerability of a new facial 2-mercaptonicotinoyl glycine-containing depigmenting serum versus hydroquinone 4% over 3-month treatment of facial melasma. Dermatol Ther (Heidelb). 2025;15:2379-2390.
- McKesey J, Tovar-Garza A, Pandya AG. Melasma treatment: an evidence-based review. Am J Clin Dermatol. 2020;21:173-225.
- Bala HR, Lee S, Wong C, et al. Oral tranexamic acid for the treatment of melasma: a review. Dermatol Surg. 2018;44:814-825.
- Sarkar R, Katoch S. Chemical peels in treatment of melasma. Dermatol Clin. 2024;42:21-32.
- Sarkar R, Gupta M. Platelet-rich plasma in melasma—a systematic review. Dermatol Surg. 2022;48:131-134.
- Pongklaokam J, Manuskiatti W, Wanitphakdeedecha R, et al. Efficacy and safety of letibotulinum toxin A for the treatment of melasma in two different dilutions: a randomized double-blind split-face study. Toxins (Basel). 2025;17:349.
- Han HJ, Kim JC, Park YJ, et al. Targeting the dermis for melasma maintenance treatment. Sci Rep. 2024;14:949.
- Zhang AD, Lazar M, Akhundova E, et al. A scoping review on melasma treatments and their histopathologic correlates. Dermatopathology (Basel). 2025;12:13.
- Goldman MP, Wu DC, Khetarpal S, et al. Dual 1550-nm erbium glass fiber and 1927-nm thulium fiber non-ablative fractional laser system treatment in patients with skin of color: a review of clinical studies and unmet needs. Lasers Surg Med. Published online February 10, 2026.
- Chen J, Yu N, Peng L, et al. Efficacy of low-fluence 1064 nm Q-switched Nd: YAG laser for the treatment of melasma: a meta-analysis and systematic review. J Cosmet Dermatol. 2022;21.
- Feng J, Huang L. Comparison of picosecond and nanosecond Nd:YAG 1064-nm lasers in the treatment of melasma: a split-face randomized clinical trial. Plast Reconstr Surg. 2023;151:772-777.
- Passeron T, Genedy R, Salah L, et al., Laser treatment of hyperpigmented lesions: position statement of the European Society of Laser in Dermatology. J Eur Acad Dermatol Venereol. 2019;33:987-1005.
- Sanchez NP, Pathak MA, Sato S, et al. Melasma: a clinical, light microscopic, ultrastructural, and immunofluorescence study. J Am Acad Dermatol. 1981;4:698-710.
- Bostan E, Cakir A. The dermoscopic characteristics of melasma in relation to different skin phototypes, distribution patterns and wood lamp findings: a cross-sectional study of 236 melasma lesions. Arch Dermatol Res. 2023;315:1927-1938.
- Guida S, Longo C, Ronga R, et al. Melasma and reflectance confocal microscopy: from baseline to treatment monitoring. Int J Dermatol. 2024;63:1007-1012.
- Ning X, Yang J, Ouyang H, et al. In vivo evaluation of melasma pathologic features and treatment response by 2-photon microscopy. JAMA Dermatol. 2025;161:1039-1047.
- Chen K-J, Han Y, Wang Z-Y, et al. Submicron resolution techniques: multiphoton microscopy in skin disease. Exp Dermatol. 2023;32:1613-1623.
- Philipp-Dormston WG. Melasma: a step-by-step approach towards a multimodal combination therapy. Clin Cosmet Investig Dermatol. 2024;17:1203-1216.
- Doolan B, Gupta M. Melasma. Aust J Gen Pract. 2021;50:880-885.
- Ali L, Al Niaimi F. Pathogenesis of melasma explained. Int J Dermatol. 2025;64:1201-1212.
- Artzi O, Horovitz T, Bar-Ilan E, et al. The pathogenesis of melasma and implications for treatment. J Cosmet Dermatol. 2021;20:3432-3445.
- Gan C, Rodrigues M. An update on new and existing treatments for the management of melasma. Am J Clin Dermatol. 2024;25:717-733.
- Sarkar R, Desai SR, Sinha S, et al. Delphi consensus on melasma management by international experts and pigmentary disorders society. J Eur Acad Dermatol Venereol. Published online September 25, 2025.
- Desai SR, Alexis AF, Elbuluk N, et al. Best practices in the treatment of melasma with a focus on patients with skin of color. J Am Acad Dermatol. 2024;90:269-279.
- Lim HW, Kohli I, Ruvolo E, et al. Impact of visible light on skin health: the role of antioxidants and free radical quenchers in skin protection. J Am Acad Deramtol. 2022;86(3 suppl):S27-S37.
- Goh CL, Chuah SY, Tien S, et al. Double-blind, placebo-controlled trial to evaluate the effectiveness of polypodium leucotomos extract in the treatment of melasma in Asian skin: a pilot study. J Clin Aesthet Dermatol. 2018;11:14-19.
- Mahajan VK, Patil A, Blicharz L, et al. Medical therapies for melasma. J Cosmet Dermatol. 2022;21:3707-3728.
- Suliman RS, Alhuwayshil J, Almuflehi AA, et al. Emerging topical therapies for melasma: a comparative analysis of efficacy and safety. J Dermatolog Treat. 2025;36:2591502.
- Austin E, Nguyen JK, Jagdeo J. Topical treatments for melasma: a systematic review of randomized controlled trials. J Drugs Dermatol. 2019;18:S1545961619P1156X.
- Grimes PE, Dias S, Oparaugo NC, et al. A randomized, controlled, split-face, double-blind study comparing topical malassezin to hydroquinone 4% for melasma. J Drugs Dermatol. 2026;25:25-29.
- Passeron T, Kerob D, Le Dantec G, et al. Efficacy and tolerability of a new facial 2-mercaptonicotinoyl glycine-containing depigmenting serum versus hydroquinone 4% over 3-month treatment of facial melasma. Dermatol Ther (Heidelb). 2025;15:2379-2390.
- McKesey J, Tovar-Garza A, Pandya AG. Melasma treatment: an evidence-based review. Am J Clin Dermatol. 2020;21:173-225.
- Bala HR, Lee S, Wong C, et al. Oral tranexamic acid for the treatment of melasma: a review. Dermatol Surg. 2018;44:814-825.
- Sarkar R, Katoch S. Chemical peels in treatment of melasma. Dermatol Clin. 2024;42:21-32.
- Sarkar R, Gupta M. Platelet-rich plasma in melasma—a systematic review. Dermatol Surg. 2022;48:131-134.
- Pongklaokam J, Manuskiatti W, Wanitphakdeedecha R, et al. Efficacy and safety of letibotulinum toxin A for the treatment of melasma in two different dilutions: a randomized double-blind split-face study. Toxins (Basel). 2025;17:349.
- Han HJ, Kim JC, Park YJ, et al. Targeting the dermis for melasma maintenance treatment. Sci Rep. 2024;14:949.
- Zhang AD, Lazar M, Akhundova E, et al. A scoping review on melasma treatments and their histopathologic correlates. Dermatopathology (Basel). 2025;12:13.
- Goldman MP, Wu DC, Khetarpal S, et al. Dual 1550-nm erbium glass fiber and 1927-nm thulium fiber non-ablative fractional laser system treatment in patients with skin of color: a review of clinical studies and unmet needs. Lasers Surg Med. Published online February 10, 2026.
- Chen J, Yu N, Peng L, et al. Efficacy of low-fluence 1064 nm Q-switched Nd: YAG laser for the treatment of melasma: a meta-analysis and systematic review. J Cosmet Dermatol. 2022;21.
- Feng J, Huang L. Comparison of picosecond and nanosecond Nd:YAG 1064-nm lasers in the treatment of melasma: a split-face randomized clinical trial. Plast Reconstr Surg. 2023;151:772-777.
- Passeron T, Genedy R, Salah L, et al., Laser treatment of hyperpigmented lesions: position statement of the European Society of Laser in Dermatology. J Eur Acad Dermatol Venereol. 2019;33:987-1005.
Beyond Conventional Paradigms: Rethinking the Evaluation and Management of Melasma
Beyond Conventional Paradigms: Rethinking the Evaluation and Management of Melasma
Painful, Purpuric, Nodular Lesion With an Irregular Surface on the Shoulder
Painful, Purpuric, Nodular Lesion With an Irregular Surface on the Shoulder
THE DIAGNOSIS: Cutaneous Leiomyosarcoma
Based on the clinical and histopathologic findings, our patient was diagnosed with primary cutaneous leiomyosarcoma (LMS), a rare soft-tissue neoplasm that arises from smooth muscle and typically manifests as a firm pink nodule.1 The neoplasm may occur in the area of a prior traumatic injury or develop spontaneously without an identifiable cause.1-3 Cutaneous LMS represents 2% to 3% of all soft-tissue sarcomas worldwide, with an estimated incidence of 1 in 500,000 annually.1,4 Men who are in their fifth to seventh decades of life are at the highest risk for LMS.1
Histologically, cutaneous LMS can be subclassified as dermal, which has a low metastatic risk and excellent prognosis, or subcutaneous, which is associated with poorer outcomes and vascular muscle origin.1 In our case, hematoxylin and eosin staining revealed fascicles of smooth muscle fibers with hypercellularity, atypia, and mitotic figures (Figure). The neoplasm stained positive for desmin, vimentin, and smooth muscle actin and negative for SOX10, Melan-A, PRAME (preferentially expressed antigen in melanoma), CD34, and Factor XIIIa.1
Standard treatment for LMS is surgical excision.5 Poor prognostic factors include lesions with a diameter of 5 cm or larger, deep subcutaneous tumor invasion, and distant metastases.2,5
The differential diagnosis may include dermatofibrosarcoma protuberans, which can have a similar pink nodular appearance and also may manifest after injury6; however, this lesion would stain positive for CD34 on histopathology.1 Nodular melanoma also can manifest as a solitary red, raised lesion, but it would stain positive for SOX10, PRAME, and Melan-A on histopathology.7 Basal cell carcinoma, which also may have a similar clinical appearance, is associated with nests of basaloid cells and palisading nuclei histologically.8 Lastly, atypical fibroxanthoma also manifests as a red nodule or plaque and is associated with atypical mitotic figures on histology; however, it notably stains negative for desmin.9
In summary, cutaneous LMS should be included in the differential diagnosis for raised, pink nodules. Given its nonspecific clinical presentation, this rare and malignant neoplasm requires biopsy and immunohistochemical staining for accurate diagnosis.
- Soares Queirós C, Filipe P, Soares de Almeida L. Cutaneous leiomyosarcoma: a 20-year retrospective study and review of the literature. Anais Brasileiros de Dermatologia. 2021;96:278-283. doi:10.1016/j.abd.2020.10.003
- Kim NG, Kim JO, Park YJ, et al. Cutaneous leiomyosarcoma of the face. Arch Craniofac Surg. 2017;18:145-148. doi:10.7181/acfs.2017.18.2.145
- Litaiem N, Tabka M, Nefiss M, et al. Cutaneous leiomyosarcoma mimicking arteriovenous malformation. Clin Case Rep. 2020;8:2538-2540. doi:10.1002/ccr3.3222
- Hmida L, Letaief F, Doghri R, et al. Cutaneous leiomyosarcoma on the trunk: an unusual presentation with an aggressive course - case report and review of literature. Pan Afr Med J. 2018;31:190. doi:10.11604/pamj.2018.31.190.16682
- Cazzato G, Sergi MC, Sablone S, et al. Advanced cutaneous leiomyosarcoma of the forearm. Dermatopathology (Basel). 2021;8:40-44. doi:10.3390/dermatopathology8010008
- Li Y, Wang C, Xiang B, et al. Clinical features, pathological findings and treatment of recurrent dermatofibrosarcoma protuberans. J Cancer. 2017;8:1319-1323. doi:10.7150/jca.17988
- Hernandez LE, Frech FS, Mohsin N, et al. Nodular melanoma: a review of pathogenesis, presentation, diagnosis and treatment. J Dermatol & Skin Sci. 2021;3:25-30. doi:10.29245/2767-5092/2021/3.1144
- Steele RB, Puckette Y. Basal cell carcinoma. StatPearls [Internet]. StatPearls Publishing; 2026. Updated November 7, 2025. Accessed March 3, 2026. https://www.ncbi.nlm.nih.gov/books/NBK482439/
- Kolb L, Schmieder GJ. Atypical fibroxanthoma. StatPearls [Internet]. StatPearls Publishing; 2026. Updated September 18, 2022. Accessed March 3, 2026. http://www.ncbi.nlm.nih.gov/books/NBK459342/
THE DIAGNOSIS: Cutaneous Leiomyosarcoma
Based on the clinical and histopathologic findings, our patient was diagnosed with primary cutaneous leiomyosarcoma (LMS), a rare soft-tissue neoplasm that arises from smooth muscle and typically manifests as a firm pink nodule.1 The neoplasm may occur in the area of a prior traumatic injury or develop spontaneously without an identifiable cause.1-3 Cutaneous LMS represents 2% to 3% of all soft-tissue sarcomas worldwide, with an estimated incidence of 1 in 500,000 annually.1,4 Men who are in their fifth to seventh decades of life are at the highest risk for LMS.1
Histologically, cutaneous LMS can be subclassified as dermal, which has a low metastatic risk and excellent prognosis, or subcutaneous, which is associated with poorer outcomes and vascular muscle origin.1 In our case, hematoxylin and eosin staining revealed fascicles of smooth muscle fibers with hypercellularity, atypia, and mitotic figures (Figure). The neoplasm stained positive for desmin, vimentin, and smooth muscle actin and negative for SOX10, Melan-A, PRAME (preferentially expressed antigen in melanoma), CD34, and Factor XIIIa.1

Standard treatment for LMS is surgical excision.5 Poor prognostic factors include lesions with a diameter of 5 cm or larger, deep subcutaneous tumor invasion, and distant metastases.2,5
The differential diagnosis may include dermatofibrosarcoma protuberans, which can have a similar pink nodular appearance and also may manifest after injury6; however, this lesion would stain positive for CD34 on histopathology.1 Nodular melanoma also can manifest as a solitary red, raised lesion, but it would stain positive for SOX10, PRAME, and Melan-A on histopathology.7 Basal cell carcinoma, which also may have a similar clinical appearance, is associated with nests of basaloid cells and palisading nuclei histologically.8 Lastly, atypical fibroxanthoma also manifests as a red nodule or plaque and is associated with atypical mitotic figures on histology; however, it notably stains negative for desmin.9
In summary, cutaneous LMS should be included in the differential diagnosis for raised, pink nodules. Given its nonspecific clinical presentation, this rare and malignant neoplasm requires biopsy and immunohistochemical staining for accurate diagnosis.
THE DIAGNOSIS: Cutaneous Leiomyosarcoma
Based on the clinical and histopathologic findings, our patient was diagnosed with primary cutaneous leiomyosarcoma (LMS), a rare soft-tissue neoplasm that arises from smooth muscle and typically manifests as a firm pink nodule.1 The neoplasm may occur in the area of a prior traumatic injury or develop spontaneously without an identifiable cause.1-3 Cutaneous LMS represents 2% to 3% of all soft-tissue sarcomas worldwide, with an estimated incidence of 1 in 500,000 annually.1,4 Men who are in their fifth to seventh decades of life are at the highest risk for LMS.1
Histologically, cutaneous LMS can be subclassified as dermal, which has a low metastatic risk and excellent prognosis, or subcutaneous, which is associated with poorer outcomes and vascular muscle origin.1 In our case, hematoxylin and eosin staining revealed fascicles of smooth muscle fibers with hypercellularity, atypia, and mitotic figures (Figure). The neoplasm stained positive for desmin, vimentin, and smooth muscle actin and negative for SOX10, Melan-A, PRAME (preferentially expressed antigen in melanoma), CD34, and Factor XIIIa.1

Standard treatment for LMS is surgical excision.5 Poor prognostic factors include lesions with a diameter of 5 cm or larger, deep subcutaneous tumor invasion, and distant metastases.2,5
The differential diagnosis may include dermatofibrosarcoma protuberans, which can have a similar pink nodular appearance and also may manifest after injury6; however, this lesion would stain positive for CD34 on histopathology.1 Nodular melanoma also can manifest as a solitary red, raised lesion, but it would stain positive for SOX10, PRAME, and Melan-A on histopathology.7 Basal cell carcinoma, which also may have a similar clinical appearance, is associated with nests of basaloid cells and palisading nuclei histologically.8 Lastly, atypical fibroxanthoma also manifests as a red nodule or plaque and is associated with atypical mitotic figures on histology; however, it notably stains negative for desmin.9
In summary, cutaneous LMS should be included in the differential diagnosis for raised, pink nodules. Given its nonspecific clinical presentation, this rare and malignant neoplasm requires biopsy and immunohistochemical staining for accurate diagnosis.
- Soares Queirós C, Filipe P, Soares de Almeida L. Cutaneous leiomyosarcoma: a 20-year retrospective study and review of the literature. Anais Brasileiros de Dermatologia. 2021;96:278-283. doi:10.1016/j.abd.2020.10.003
- Kim NG, Kim JO, Park YJ, et al. Cutaneous leiomyosarcoma of the face. Arch Craniofac Surg. 2017;18:145-148. doi:10.7181/acfs.2017.18.2.145
- Litaiem N, Tabka M, Nefiss M, et al. Cutaneous leiomyosarcoma mimicking arteriovenous malformation. Clin Case Rep. 2020;8:2538-2540. doi:10.1002/ccr3.3222
- Hmida L, Letaief F, Doghri R, et al. Cutaneous leiomyosarcoma on the trunk: an unusual presentation with an aggressive course - case report and review of literature. Pan Afr Med J. 2018;31:190. doi:10.11604/pamj.2018.31.190.16682
- Cazzato G, Sergi MC, Sablone S, et al. Advanced cutaneous leiomyosarcoma of the forearm. Dermatopathology (Basel). 2021;8:40-44. doi:10.3390/dermatopathology8010008
- Li Y, Wang C, Xiang B, et al. Clinical features, pathological findings and treatment of recurrent dermatofibrosarcoma protuberans. J Cancer. 2017;8:1319-1323. doi:10.7150/jca.17988
- Hernandez LE, Frech FS, Mohsin N, et al. Nodular melanoma: a review of pathogenesis, presentation, diagnosis and treatment. J Dermatol & Skin Sci. 2021;3:25-30. doi:10.29245/2767-5092/2021/3.1144
- Steele RB, Puckette Y. Basal cell carcinoma. StatPearls [Internet]. StatPearls Publishing; 2026. Updated November 7, 2025. Accessed March 3, 2026. https://www.ncbi.nlm.nih.gov/books/NBK482439/
- Kolb L, Schmieder GJ. Atypical fibroxanthoma. StatPearls [Internet]. StatPearls Publishing; 2026. Updated September 18, 2022. Accessed March 3, 2026. http://www.ncbi.nlm.nih.gov/books/NBK459342/
- Soares Queirós C, Filipe P, Soares de Almeida L. Cutaneous leiomyosarcoma: a 20-year retrospective study and review of the literature. Anais Brasileiros de Dermatologia. 2021;96:278-283. doi:10.1016/j.abd.2020.10.003
- Kim NG, Kim JO, Park YJ, et al. Cutaneous leiomyosarcoma of the face. Arch Craniofac Surg. 2017;18:145-148. doi:10.7181/acfs.2017.18.2.145
- Litaiem N, Tabka M, Nefiss M, et al. Cutaneous leiomyosarcoma mimicking arteriovenous malformation. Clin Case Rep. 2020;8:2538-2540. doi:10.1002/ccr3.3222
- Hmida L, Letaief F, Doghri R, et al. Cutaneous leiomyosarcoma on the trunk: an unusual presentation with an aggressive course - case report and review of literature. Pan Afr Med J. 2018;31:190. doi:10.11604/pamj.2018.31.190.16682
- Cazzato G, Sergi MC, Sablone S, et al. Advanced cutaneous leiomyosarcoma of the forearm. Dermatopathology (Basel). 2021;8:40-44. doi:10.3390/dermatopathology8010008
- Li Y, Wang C, Xiang B, et al. Clinical features, pathological findings and treatment of recurrent dermatofibrosarcoma protuberans. J Cancer. 2017;8:1319-1323. doi:10.7150/jca.17988
- Hernandez LE, Frech FS, Mohsin N, et al. Nodular melanoma: a review of pathogenesis, presentation, diagnosis and treatment. J Dermatol & Skin Sci. 2021;3:25-30. doi:10.29245/2767-5092/2021/3.1144
- Steele RB, Puckette Y. Basal cell carcinoma. StatPearls [Internet]. StatPearls Publishing; 2026. Updated November 7, 2025. Accessed March 3, 2026. https://www.ncbi.nlm.nih.gov/books/NBK482439/
- Kolb L, Schmieder GJ. Atypical fibroxanthoma. StatPearls [Internet]. StatPearls Publishing; 2026. Updated September 18, 2022. Accessed March 3, 2026. http://www.ncbi.nlm.nih.gov/books/NBK459342/
Painful, Purpuric, Nodular Lesion With an Irregular Surface on the Shoulder
Painful, Purpuric, Nodular Lesion With an Irregular Surface on the Shoulder
A 53-year-old man presented to the dermatology clinic for evaluation of a painful, purpuric, nodular lesion on the left shoulder of 3 months’ duration. The lesion had an irregular surface that was surrounded by an erythematous ring. Biopsy revealed fascicles of eosinophilic cells within the dermis. The nuclei were heterogeneous in size and shape and had blunted ends. Frequent atypia and mitotic figures were observed, and the lesion extended into the subcutis. Immunostaining was positive for desmin and smooth muscle actin and negative for SOX10, Melan-A, PRAME (preferentially expressed antigen in melanoma), CD34, and Factor XIIIa.