User login
A Case of Birt-Hogg-Dubé Syndrome: A Rare but Essential Diagnosis to Consider
A Case of Birt-Hogg-Dubé Syndrome: A Rare but Essential Diagnosis to Consider
Birt-Hogg-Dubé syndrome (BHD) is an autosomal dominant disease that arises from loss-of-function mutations in the FLCN gene. FLCN encodes folliculin, which is presumed to function as a tumor suppressor, though its precise role is incompletely understood.1,2 BHD is characterized by multiple pulmonary cysts leading to recurrent spontaneous pneumothoraces, cutaneous lesions—specifically fibrofolliculomas—and an increased risk of renal malignancies. Diagnosing BHD is challenging due to the variable presentation of the disease. Some patients may only have cystic lung diseases, while others may not have characteristic skin lesions.3-5 It is important to maintain awareness of BHD, especially when the diagnosis dictates the need for genetic counseling.
Case Presentation
A male veteran in his 60s, who was a lifelong nonsmoker with a history of extensive bullous emphysema and recurrent pneumothoraces, presented to the Veterans Affairs Greater Los Angeles Healthcare System pulmonary clinic while transferring care from a separate institution.
According to the patient, the first pneumothorax episode occurred about 20 years before presentation, followed by a recurrence a few years later after he was diagnosed with emphysema. He underwent pleurodesis of the right lung during his service abroad. Another episode nearly a decade after the first pneumothorax necessitated pleurodesis of the left lung (Figure 1). The patient's family history revealed pulmonary cysts in 1 immediate family member but no history of renal tumors. Notably, his mother passed away at a young age due to tuberculosis.
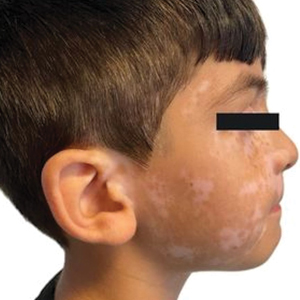
On physical examination, numerous skin tags and acrochordons on the face were observed, which had been stable for > 30 years. Despite a slow decline in exercise capacity following pleurodesis, the patient could still walk multiple miles daily and climb 3 flights of stairs before needing to rest. Pulmonary function testing (PFT) showed a forced expiratory volume in 1 second (FEV1)/forced vital capacity ratio of 0.84 with reduced FEV1, total lung capacity (TLC), and diffusion capacity for carbon monoxide (DLCO), indicating a mild restrictive ventilatory defect and reduced diffusing capacity.
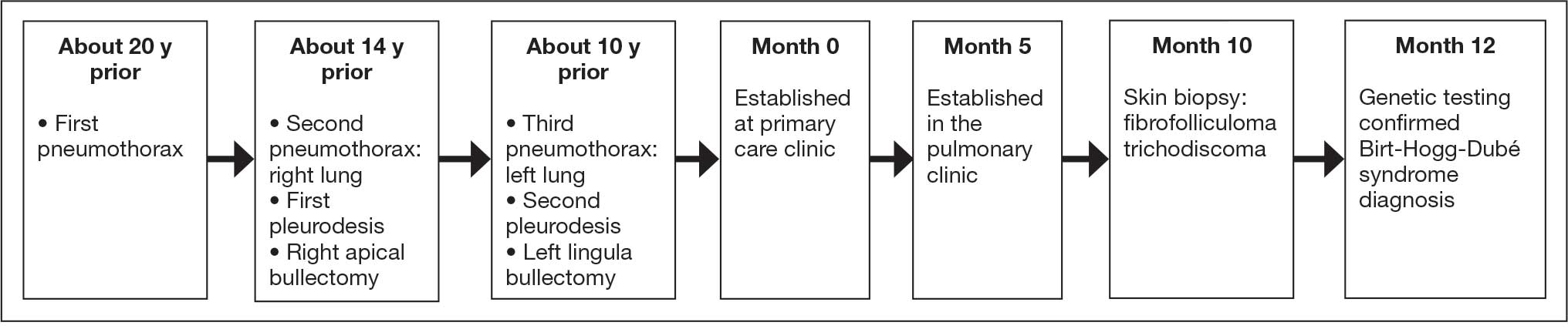
Laboratory results revealed a normal α-1 antitrypsin level: 133 mg/dL (reference, 83-199 mg/dL), with a Pi*MS phenotype and undetectable antinuclear antibodies. The most recent chest computed tomography (CT) in 2019, displayed paraseptal and centrilobular emphysema, scattered blebs, and scarring consistent with prior pleurodesis procedures (Figure 2).
Genetic testing for the FLCN gene revealed heterozygous pathogenic mutation: c.1285del and p.His429Thrfs*39, which confirmed the diagnosis of BHD. A shave biopsy of a postauricular papular lesion confirmed a histologic pattern of fibrofolliculoma/trichodiscoma.
Follow-up and Outcomes
After confirmation of the BHD diagnosis, the patient was referred to genetic counseling and scheduled for annual magnetic resonance imaging (MRI) of the abdomen and pelvis to screen for renal malignancies. As the patient was able to establish care with a new long-term primary care practitioner in the outpatient setting, he continues regular follow-up visits in the pulmonary clinic with stable respiratory symptoms and no recurrent pneumothoraces thus far.
Discussion
Differential Diagnoses of Cystic Pulmonary Lesions
BHD is an important differential diagnosis to consider in the presentation of diffuse cystic lung diseases. Still, 2 other crucial considerations are pulmonary Langerhans cell histiocytosis (PLCH) and lymphangioleiomyomatosis (LAM), which occur at slightly higher frequencies than BHD.6
One of the first steps in radiographically evaluating cystic lung diseases is to characterize the cysts. The Fleischner Society defines true cysts as a “round parenchymal lucency or low-attenuating area with a well-defined interface with normal lung.”7 Mimics of cystic lesions may include cavitary lung lesions, thick-walled spaces within another area of mass, nodule, or consolidation. Another mimic is a pneumatocele, a pseudocyst that lacks epithelial lining and may be secondary to bacterial pneumonia, pneumocystis infections, trauma, or prior mechanical ventilation.8After characterizing true cysts, different patterns of cystic lesions can also be associated with specific diseases. Cysts in PLCH are commonly more uniform and round, whereas the cysts in LAM may be more irregularly shaped. 9 Cysts in BHD may be larger and predominantly located in basal and paramediastinal areas.4LAM is associated with tuberous sclerosis, which can also present with skin lesions (angiofibromas) and renal tumors (angiomyolipomas), thus creating a very similar picture to BHD. Therefore, tissue biopsies of skin lesions are essential as histopathology can identify characteristic fibrofolliculomas specific to BHD. While genetic testing would also strongly support the diagnosis of BHD, it is essential to note that negative genetic testing does not rule out BHD.4Lastly, lymphoid interstitial pneumonia (LIP) is another important consideration in the differential diagnosis of cystic lung diseases. LIP presents with not only perivascular cysts and centrilobular nodules but also diffuse ground-glass attenuation.10 In contrast to BHD, LIP is associated with autoimmune diseases such as Sjögren syndrome and infectious diseases such as HIV; thus, it may be differentiated from BHD by the presence of underlying disease processes and may warrant serologic testing for potential rheumatologic disorders.
Characteristics and Diagnostic Criteria
Cystic lung disease is the most common presentation of BHD. It presents in > 80% of cases and confers a 50-fold increase in the risk of spontaneous pneumothorax compared with the general population.4,11 Recurrent pneumothoraces are observed in about 25% to 30% of patients with BHD, typically occurring between the third and fifth decades of life and at significantly decreased rates after 50 years of age.12 A spontaneous pneumothorax might serve as the initial and perhaps the sole clinical presentation for some patients with BHD, but others may present with other respiratory symptoms such as cough and exertional dyspnea. PFT results may be normal or reveal a mild restrictive ventilatory defect and reduced DLCO, as reported in a few cases.6 The management of pulmonary complications primarily revolves around reducing the risk of pneumothoraces, which includes precautions such as avoiding positive pressure ventilation and air travel. Early pleurodesis with the first occurrence of a spontaneous pneumothorax is considered in some cases.13
The distinctive dermatologic features associated with BHD include multiple white papules primarily found on the nose and face. Pathologically, these manifestations have a range of histologic distinctions, from fibrofolliculomas to benign hamartomas of the hair follicles and trichodiscomas.5 The diagnostic criteria outlined by Menko et al note that confirmation of BHD requires the presence of either ≥ 5 pathologically confirmed fibrofolliculomas or trichodiscomas, a documented pathogenic FLCN gene mutation, or the fulfillment of 2 minor criteria. These minor criteria include the presence of multiple lung cysts, early-onset renal cancer, or a first-degree relative with BHD.5
Recurrent Pneumothoraces Management
After the first episode of spontaneous pneumothorax, early pleurodesis is indicated as the risk of recurrence can be as high as 75%.4,14 Specific pleurodesis modalities have shown promising results, such as total pleural covering with cellulose mesh. In a small retrospective review, cellulose mesh demonstrated a significant reduction in the recurrence rate of pneumothorax at 7.5 years for patients with BHD compared with partial covering.15 Apart from preventing further pneumothorax episodes in the affected lung, it is also important to highlight patient education and monitoring after initial pleurodesis, as the contralateral lung is also at risk. As demonstrated in this case, the patient had received pleurodesis of his right lung but experienced another pneumothorax of his contralateral lung a few years later.
Lastly, the patient was advised to avoid air travel altogether; however, current data may suggest that air travel may not be an absolute contraindication for patients with BHD. Although the literature on this topic is limited, a retrospective study by Johannesma et al involving 158 patients with BHD surveyed on pneumothorax occurrence after air travel indicated a calculated risk of 0.63% per flight. Notably, only 3 of 13 patients with BHD and recurrent pneumothoraces after travel had undergone pleurodesis in the past.16 Therefore, counseling patients on the potential risks of air travel and allowing essential flights while diligently monitoring for symptoms during and after travel may be a reasonable, patient-centered approach in contrast to a complete restriction on air travel.
Timing to Diagnosis
Diagnosing BHD is challenging and often delayed. In a 2022 study by Steinlein et al, the average delay in BHD diagnoses in their cohort was 9.3 years, with 4 patients also diagnosed with renal malignancy during the study period.17 The difficulty in diagnosis can be attributed to the heterogeneous presentation among affected family members, some of whom may exclusively exhibit pulmonary cystic lesions without dermatologic findings.
A lack of longitudinal care for this patient may have contributed to the diagnostic delay. The patient had pneumothorax events across separate care settings and locations, and due to employment-related relocations, he often re-established care at various health care systems. This case highlights the importance of continuity of care, especially in BHD, where monitoring for renal tumors is also essential to long-term management.17,18
Renal Tumor Monitoring
Finally, once BHD is diagnosed, one of the most important considerations is to begin routine monitoring for renal malignancies. Current recommendations advise starting lifelong renal cancer screening, even as early as age 20 years, with annual MRIs, as renal ultrasound may not be sufficiently sensitive to detect smaller lesions.19 The screening interval can be extended to every 2 years for patients without a family history of renal tumors or suspicious renal lesions. If tumors are found, then nephron-sparing surgery is recommended, given the potential for the development of chronic renal insufficiency in patients with BHD.20
Conclusions
BHD is a rare and complex syndrome in which early recognition and diagnosis play a pivotal role in preventing potentially severe complications such as renal malignancies. Suspicion of a genetic disorder, such as BHD, LAM, or PLCH, should arise in patients who experience spontaneous pneumothorax, especially in the presence of multiple cystic lesions or a family history of pneumothoraces. Early consideration of pleurodesis after the first spontaneous pneumothorax is advisable. The complex presentation of BHD contributes to the difficulty of diagnosis and may delay recognition, which can be exacerbated by variable continuity of care.
- Schmidt LS, Linehan WM. Molecular genetics and clinical features of Birt-Hogg-Dubé-Syndrome. Nat Rev Urol. 2015;12:558-569. doi:10.1038/nrurol.2015.206
- Lim DHK, Rehal PK, Nahorski MS, et al. A new locus-specific database (LSDB) for mutations in the folliculin (FLCN) gene. Hum Mutat. 2010;31:E1043-1051. doi:10.1002/humu.21130
- Aivaz O, Berkman S, Middelton L, et al. Comedonal and cystic fibrofolliculomas in Birt-Hogg-Dube syndrome. JAMA Dermatology. 2015;151:770-774. doi:10.1001/jamadermatol.2015.0215
- Daccord C, Good JM, Morren MA, et al. Birt–Hogg–Dubé syndrome. Eur Respir Rev. 2020;29:200042. doi:10.1183/16000617.0042-2020
- Menko FH, van Steensel MA, Giraud S, et al. Birt-Hogg-Dubé syndrome: diagnosis and management. The Lancet Oncology. 2009;10:1199-1206. doi:10.1016/S1470-2045(09)70188-3
- Daccord C, Cottin V, Prévot G, et al. Lung function in Birt-Hogg-Dubé syndrome: a retrospective analysis of 96 patients. Orphanet J Rare Dis. 2020;15:120. doi:10.1186/s13023-020-01402-y
- Hansell DM, Bankier AA, MacMahon H, et al. Fleischner Society: glossary of terms for thoracic imaging. Radiology. 2008;246:697-722. doi:10.1148/radiol.2462070712
- Jamil A, Kasi A. Pneumatocele. In: StatPearls. StatPearls Publishing; 2024. Accessed March 2, 2026. http://www.ncbi.nlm.nih.gov/books/NBK556146/
- Bhardwaj H, Bhardwaj B. Differentiating pulmonary lymphangioleiomyomatosis from pulmonary langerhans cell histiocytosis and Birt-Hogg-Dube syndrome. Lung India. 2013;30:372-373. doi:10.4103/0970-2113.120611
- Swigris JJ, Berry GJ, Raffin TA, et al. Lymphoid interstitial pneumonia: a narrative review. Chest. 2002;122:2150-2164. doi:10.1378/chest.122.6.2150
- Zbar B, Alvord WG, Glenn G, et al. Risk of renal and colonic neoplasms and spontaneous pneumothorax in the Birt-Hogg-Dubé syndrome. Cancer Epidemiol Biomarkers Prev. 2002;11:393-400.
- Sattler EC, Steinlein OK. Delayed diagnosis of Birt-Hogg-Dubé syndrome due to marked intrafamilial clinical variability: a case report. BMC Med Genet. 2018;19:45. doi:10.1186/s12881-018-0558-0
- Gupta N, Seyama K, McCormack FX. Pulmonary manifestations of Birt-Hogg-Dubé syndrome. Fam Cancer. 2013;12:387-396. doi:10.1007/s10689-013-9660-9
- Gupta N, Kopras EJ, Henske EP, et al. Spontaneous pneumothoraces in patients with Birt–Hogg–Dubé syndrome. Ann Am Thorac Soc. 2017;14:706-713. doi:10.1513/AnnalsATS.201611-886OC
- Mizobuchi T, Kurihara M, Ebana H, et al. A total pleural covering of absorbable cellulose mesh prevents pneumothorax recurrence in patients with Birt-Hogg-Dubé syndrome. Orphanet J Rare Dis. 2018;13:78. doi:10.1186/s13023-018-0790-x
- Johannesma PC, van de Beek I, van der Wel JWT, et al. Risk of spontaneous pneumothorax due to air travel and diving in patients with Birt–Hogg–Dubé syndrome. Springerplus. 2016;5:1506. doi:10.1186/s40064-016-3009-4
- Steinlein OK, Reithmair M, Syunyaeva Z, et al. Delayed diagnosis of Birt-Hogg-Dubé syndrome might be aggravated by gender bias. eClinicalMedicine. 2022;51:101572. doi:10.1016/j.eclinm.2022.101572
- Pereira Gray DJ, Sidaway-Lee K, White E, et al. Continuity of care with doctors—a matter of life and death? A systematic review of continuity of care and mortality. BMJ Open. 2018;8:e021161. doi:10.1136/bmjopen-2017-021161
- Sattler EC, Steinlein OK. GeneReviews Birt-Hogg-Dubé syndrome. January 30, 2020. Accessed March 2, 2026. https://www.ncbi.nlm.nih.gov/books/NBK1522/table
- Stamatakis L, Metwalli AR, Middelton LA, et al. Diagnosis and management of BHD-associated kidney cancer. Fam Cancer. 2013;12:397-402. doi:10.1007/s10689-013-9657-4
Birt-Hogg-Dubé syndrome (BHD) is an autosomal dominant disease that arises from loss-of-function mutations in the FLCN gene. FLCN encodes folliculin, which is presumed to function as a tumor suppressor, though its precise role is incompletely understood.1,2 BHD is characterized by multiple pulmonary cysts leading to recurrent spontaneous pneumothoraces, cutaneous lesions—specifically fibrofolliculomas—and an increased risk of renal malignancies. Diagnosing BHD is challenging due to the variable presentation of the disease. Some patients may only have cystic lung diseases, while others may not have characteristic skin lesions.3-5 It is important to maintain awareness of BHD, especially when the diagnosis dictates the need for genetic counseling.
Case Presentation
A male veteran in his 60s, who was a lifelong nonsmoker with a history of extensive bullous emphysema and recurrent pneumothoraces, presented to the Veterans Affairs Greater Los Angeles Healthcare System pulmonary clinic while transferring care from a separate institution.
According to the patient, the first pneumothorax episode occurred about 20 years before presentation, followed by a recurrence a few years later after he was diagnosed with emphysema. He underwent pleurodesis of the right lung during his service abroad. Another episode nearly a decade after the first pneumothorax necessitated pleurodesis of the left lung (Figure 1). The patient's family history revealed pulmonary cysts in 1 immediate family member but no history of renal tumors. Notably, his mother passed away at a young age due to tuberculosis.

On physical examination, numerous skin tags and acrochordons on the face were observed, which had been stable for > 30 years. Despite a slow decline in exercise capacity following pleurodesis, the patient could still walk multiple miles daily and climb 3 flights of stairs before needing to rest. Pulmonary function testing (PFT) showed a forced expiratory volume in 1 second (FEV1)/forced vital capacity ratio of 0.84 with reduced FEV1, total lung capacity (TLC), and diffusion capacity for carbon monoxide (DLCO), indicating a mild restrictive ventilatory defect and reduced diffusing capacity.
Laboratory results revealed a normal α-1 antitrypsin level: 133 mg/dL (reference, 83-199 mg/dL), with a Pi*MS phenotype and undetectable antinuclear antibodies. The most recent chest computed tomography (CT) in 2019, displayed paraseptal and centrilobular emphysema, scattered blebs, and scarring consistent with prior pleurodesis procedures (Figure 2).

Genetic testing for the FLCN gene revealed heterozygous pathogenic mutation: c.1285del and p.His429Thrfs*39, which confirmed the diagnosis of BHD. A shave biopsy of a postauricular papular lesion confirmed a histologic pattern of fibrofolliculoma/trichodiscoma.
Follow-up and Outcomes
After confirmation of the BHD diagnosis, the patient was referred to genetic counseling and scheduled for annual magnetic resonance imaging (MRI) of the abdomen and pelvis to screen for renal malignancies. As the patient was able to establish care with a new long-term primary care practitioner in the outpatient setting, he continues regular follow-up visits in the pulmonary clinic with stable respiratory symptoms and no recurrent pneumothoraces thus far.
Discussion
Differential Diagnoses of Cystic Pulmonary Lesions
BHD is an important differential diagnosis to consider in the presentation of diffuse cystic lung diseases. Still, 2 other crucial considerations are pulmonary Langerhans cell histiocytosis (PLCH) and lymphangioleiomyomatosis (LAM), which occur at slightly higher frequencies than BHD.6
One of the first steps in radiographically evaluating cystic lung diseases is to characterize the cysts. The Fleischner Society defines true cysts as a “round parenchymal lucency or low-attenuating area with a well-defined interface with normal lung.”7 Mimics of cystic lesions may include cavitary lung lesions, thick-walled spaces within another area of mass, nodule, or consolidation. Another mimic is a pneumatocele, a pseudocyst that lacks epithelial lining and may be secondary to bacterial pneumonia, pneumocystis infections, trauma, or prior mechanical ventilation.8After characterizing true cysts, different patterns of cystic lesions can also be associated with specific diseases. Cysts in PLCH are commonly more uniform and round, whereas the cysts in LAM may be more irregularly shaped. 9 Cysts in BHD may be larger and predominantly located in basal and paramediastinal areas.4LAM is associated with tuberous sclerosis, which can also present with skin lesions (angiofibromas) and renal tumors (angiomyolipomas), thus creating a very similar picture to BHD. Therefore, tissue biopsies of skin lesions are essential as histopathology can identify characteristic fibrofolliculomas specific to BHD. While genetic testing would also strongly support the diagnosis of BHD, it is essential to note that negative genetic testing does not rule out BHD.4Lastly, lymphoid interstitial pneumonia (LIP) is another important consideration in the differential diagnosis of cystic lung diseases. LIP presents with not only perivascular cysts and centrilobular nodules but also diffuse ground-glass attenuation.10 In contrast to BHD, LIP is associated with autoimmune diseases such as Sjögren syndrome and infectious diseases such as HIV; thus, it may be differentiated from BHD by the presence of underlying disease processes and may warrant serologic testing for potential rheumatologic disorders.
Characteristics and Diagnostic Criteria
Cystic lung disease is the most common presentation of BHD. It presents in > 80% of cases and confers a 50-fold increase in the risk of spontaneous pneumothorax compared with the general population.4,11 Recurrent pneumothoraces are observed in about 25% to 30% of patients with BHD, typically occurring between the third and fifth decades of life and at significantly decreased rates after 50 years of age.12 A spontaneous pneumothorax might serve as the initial and perhaps the sole clinical presentation for some patients with BHD, but others may present with other respiratory symptoms such as cough and exertional dyspnea. PFT results may be normal or reveal a mild restrictive ventilatory defect and reduced DLCO, as reported in a few cases.6 The management of pulmonary complications primarily revolves around reducing the risk of pneumothoraces, which includes precautions such as avoiding positive pressure ventilation and air travel. Early pleurodesis with the first occurrence of a spontaneous pneumothorax is considered in some cases.13
The distinctive dermatologic features associated with BHD include multiple white papules primarily found on the nose and face. Pathologically, these manifestations have a range of histologic distinctions, from fibrofolliculomas to benign hamartomas of the hair follicles and trichodiscomas.5 The diagnostic criteria outlined by Menko et al note that confirmation of BHD requires the presence of either ≥ 5 pathologically confirmed fibrofolliculomas or trichodiscomas, a documented pathogenic FLCN gene mutation, or the fulfillment of 2 minor criteria. These minor criteria include the presence of multiple lung cysts, early-onset renal cancer, or a first-degree relative with BHD.5
Recurrent Pneumothoraces Management
After the first episode of spontaneous pneumothorax, early pleurodesis is indicated as the risk of recurrence can be as high as 75%.4,14 Specific pleurodesis modalities have shown promising results, such as total pleural covering with cellulose mesh. In a small retrospective review, cellulose mesh demonstrated a significant reduction in the recurrence rate of pneumothorax at 7.5 years for patients with BHD compared with partial covering.15 Apart from preventing further pneumothorax episodes in the affected lung, it is also important to highlight patient education and monitoring after initial pleurodesis, as the contralateral lung is also at risk. As demonstrated in this case, the patient had received pleurodesis of his right lung but experienced another pneumothorax of his contralateral lung a few years later.
Lastly, the patient was advised to avoid air travel altogether; however, current data may suggest that air travel may not be an absolute contraindication for patients with BHD. Although the literature on this topic is limited, a retrospective study by Johannesma et al involving 158 patients with BHD surveyed on pneumothorax occurrence after air travel indicated a calculated risk of 0.63% per flight. Notably, only 3 of 13 patients with BHD and recurrent pneumothoraces after travel had undergone pleurodesis in the past.16 Therefore, counseling patients on the potential risks of air travel and allowing essential flights while diligently monitoring for symptoms during and after travel may be a reasonable, patient-centered approach in contrast to a complete restriction on air travel.
Timing to Diagnosis
Diagnosing BHD is challenging and often delayed. In a 2022 study by Steinlein et al, the average delay in BHD diagnoses in their cohort was 9.3 years, with 4 patients also diagnosed with renal malignancy during the study period.17 The difficulty in diagnosis can be attributed to the heterogeneous presentation among affected family members, some of whom may exclusively exhibit pulmonary cystic lesions without dermatologic findings.
A lack of longitudinal care for this patient may have contributed to the diagnostic delay. The patient had pneumothorax events across separate care settings and locations, and due to employment-related relocations, he often re-established care at various health care systems. This case highlights the importance of continuity of care, especially in BHD, where monitoring for renal tumors is also essential to long-term management.17,18
Renal Tumor Monitoring
Finally, once BHD is diagnosed, one of the most important considerations is to begin routine monitoring for renal malignancies. Current recommendations advise starting lifelong renal cancer screening, even as early as age 20 years, with annual MRIs, as renal ultrasound may not be sufficiently sensitive to detect smaller lesions.19 The screening interval can be extended to every 2 years for patients without a family history of renal tumors or suspicious renal lesions. If tumors are found, then nephron-sparing surgery is recommended, given the potential for the development of chronic renal insufficiency in patients with BHD.20
Conclusions
BHD is a rare and complex syndrome in which early recognition and diagnosis play a pivotal role in preventing potentially severe complications such as renal malignancies. Suspicion of a genetic disorder, such as BHD, LAM, or PLCH, should arise in patients who experience spontaneous pneumothorax, especially in the presence of multiple cystic lesions or a family history of pneumothoraces. Early consideration of pleurodesis after the first spontaneous pneumothorax is advisable. The complex presentation of BHD contributes to the difficulty of diagnosis and may delay recognition, which can be exacerbated by variable continuity of care.
Birt-Hogg-Dubé syndrome (BHD) is an autosomal dominant disease that arises from loss-of-function mutations in the FLCN gene. FLCN encodes folliculin, which is presumed to function as a tumor suppressor, though its precise role is incompletely understood.1,2 BHD is characterized by multiple pulmonary cysts leading to recurrent spontaneous pneumothoraces, cutaneous lesions—specifically fibrofolliculomas—and an increased risk of renal malignancies. Diagnosing BHD is challenging due to the variable presentation of the disease. Some patients may only have cystic lung diseases, while others may not have characteristic skin lesions.3-5 It is important to maintain awareness of BHD, especially when the diagnosis dictates the need for genetic counseling.
Case Presentation
A male veteran in his 60s, who was a lifelong nonsmoker with a history of extensive bullous emphysema and recurrent pneumothoraces, presented to the Veterans Affairs Greater Los Angeles Healthcare System pulmonary clinic while transferring care from a separate institution.
According to the patient, the first pneumothorax episode occurred about 20 years before presentation, followed by a recurrence a few years later after he was diagnosed with emphysema. He underwent pleurodesis of the right lung during his service abroad. Another episode nearly a decade after the first pneumothorax necessitated pleurodesis of the left lung (Figure 1). The patient's family history revealed pulmonary cysts in 1 immediate family member but no history of renal tumors. Notably, his mother passed away at a young age due to tuberculosis.

On physical examination, numerous skin tags and acrochordons on the face were observed, which had been stable for > 30 years. Despite a slow decline in exercise capacity following pleurodesis, the patient could still walk multiple miles daily and climb 3 flights of stairs before needing to rest. Pulmonary function testing (PFT) showed a forced expiratory volume in 1 second (FEV1)/forced vital capacity ratio of 0.84 with reduced FEV1, total lung capacity (TLC), and diffusion capacity for carbon monoxide (DLCO), indicating a mild restrictive ventilatory defect and reduced diffusing capacity.
Laboratory results revealed a normal α-1 antitrypsin level: 133 mg/dL (reference, 83-199 mg/dL), with a Pi*MS phenotype and undetectable antinuclear antibodies. The most recent chest computed tomography (CT) in 2019, displayed paraseptal and centrilobular emphysema, scattered blebs, and scarring consistent with prior pleurodesis procedures (Figure 2).

Genetic testing for the FLCN gene revealed heterozygous pathogenic mutation: c.1285del and p.His429Thrfs*39, which confirmed the diagnosis of BHD. A shave biopsy of a postauricular papular lesion confirmed a histologic pattern of fibrofolliculoma/trichodiscoma.
Follow-up and Outcomes
After confirmation of the BHD diagnosis, the patient was referred to genetic counseling and scheduled for annual magnetic resonance imaging (MRI) of the abdomen and pelvis to screen for renal malignancies. As the patient was able to establish care with a new long-term primary care practitioner in the outpatient setting, he continues regular follow-up visits in the pulmonary clinic with stable respiratory symptoms and no recurrent pneumothoraces thus far.
Discussion
Differential Diagnoses of Cystic Pulmonary Lesions
BHD is an important differential diagnosis to consider in the presentation of diffuse cystic lung diseases. Still, 2 other crucial considerations are pulmonary Langerhans cell histiocytosis (PLCH) and lymphangioleiomyomatosis (LAM), which occur at slightly higher frequencies than BHD.6
One of the first steps in radiographically evaluating cystic lung diseases is to characterize the cysts. The Fleischner Society defines true cysts as a “round parenchymal lucency or low-attenuating area with a well-defined interface with normal lung.”7 Mimics of cystic lesions may include cavitary lung lesions, thick-walled spaces within another area of mass, nodule, or consolidation. Another mimic is a pneumatocele, a pseudocyst that lacks epithelial lining and may be secondary to bacterial pneumonia, pneumocystis infections, trauma, or prior mechanical ventilation.8After characterizing true cysts, different patterns of cystic lesions can also be associated with specific diseases. Cysts in PLCH are commonly more uniform and round, whereas the cysts in LAM may be more irregularly shaped. 9 Cysts in BHD may be larger and predominantly located in basal and paramediastinal areas.4LAM is associated with tuberous sclerosis, which can also present with skin lesions (angiofibromas) and renal tumors (angiomyolipomas), thus creating a very similar picture to BHD. Therefore, tissue biopsies of skin lesions are essential as histopathology can identify characteristic fibrofolliculomas specific to BHD. While genetic testing would also strongly support the diagnosis of BHD, it is essential to note that negative genetic testing does not rule out BHD.4Lastly, lymphoid interstitial pneumonia (LIP) is another important consideration in the differential diagnosis of cystic lung diseases. LIP presents with not only perivascular cysts and centrilobular nodules but also diffuse ground-glass attenuation.10 In contrast to BHD, LIP is associated with autoimmune diseases such as Sjögren syndrome and infectious diseases such as HIV; thus, it may be differentiated from BHD by the presence of underlying disease processes and may warrant serologic testing for potential rheumatologic disorders.
Characteristics and Diagnostic Criteria
Cystic lung disease is the most common presentation of BHD. It presents in > 80% of cases and confers a 50-fold increase in the risk of spontaneous pneumothorax compared with the general population.4,11 Recurrent pneumothoraces are observed in about 25% to 30% of patients with BHD, typically occurring between the third and fifth decades of life and at significantly decreased rates after 50 years of age.12 A spontaneous pneumothorax might serve as the initial and perhaps the sole clinical presentation for some patients with BHD, but others may present with other respiratory symptoms such as cough and exertional dyspnea. PFT results may be normal or reveal a mild restrictive ventilatory defect and reduced DLCO, as reported in a few cases.6 The management of pulmonary complications primarily revolves around reducing the risk of pneumothoraces, which includes precautions such as avoiding positive pressure ventilation and air travel. Early pleurodesis with the first occurrence of a spontaneous pneumothorax is considered in some cases.13
The distinctive dermatologic features associated with BHD include multiple white papules primarily found on the nose and face. Pathologically, these manifestations have a range of histologic distinctions, from fibrofolliculomas to benign hamartomas of the hair follicles and trichodiscomas.5 The diagnostic criteria outlined by Menko et al note that confirmation of BHD requires the presence of either ≥ 5 pathologically confirmed fibrofolliculomas or trichodiscomas, a documented pathogenic FLCN gene mutation, or the fulfillment of 2 minor criteria. These minor criteria include the presence of multiple lung cysts, early-onset renal cancer, or a first-degree relative with BHD.5
Recurrent Pneumothoraces Management
After the first episode of spontaneous pneumothorax, early pleurodesis is indicated as the risk of recurrence can be as high as 75%.4,14 Specific pleurodesis modalities have shown promising results, such as total pleural covering with cellulose mesh. In a small retrospective review, cellulose mesh demonstrated a significant reduction in the recurrence rate of pneumothorax at 7.5 years for patients with BHD compared with partial covering.15 Apart from preventing further pneumothorax episodes in the affected lung, it is also important to highlight patient education and monitoring after initial pleurodesis, as the contralateral lung is also at risk. As demonstrated in this case, the patient had received pleurodesis of his right lung but experienced another pneumothorax of his contralateral lung a few years later.
Lastly, the patient was advised to avoid air travel altogether; however, current data may suggest that air travel may not be an absolute contraindication for patients with BHD. Although the literature on this topic is limited, a retrospective study by Johannesma et al involving 158 patients with BHD surveyed on pneumothorax occurrence after air travel indicated a calculated risk of 0.63% per flight. Notably, only 3 of 13 patients with BHD and recurrent pneumothoraces after travel had undergone pleurodesis in the past.16 Therefore, counseling patients on the potential risks of air travel and allowing essential flights while diligently monitoring for symptoms during and after travel may be a reasonable, patient-centered approach in contrast to a complete restriction on air travel.
Timing to Diagnosis
Diagnosing BHD is challenging and often delayed. In a 2022 study by Steinlein et al, the average delay in BHD diagnoses in their cohort was 9.3 years, with 4 patients also diagnosed with renal malignancy during the study period.17 The difficulty in diagnosis can be attributed to the heterogeneous presentation among affected family members, some of whom may exclusively exhibit pulmonary cystic lesions without dermatologic findings.
A lack of longitudinal care for this patient may have contributed to the diagnostic delay. The patient had pneumothorax events across separate care settings and locations, and due to employment-related relocations, he often re-established care at various health care systems. This case highlights the importance of continuity of care, especially in BHD, where monitoring for renal tumors is also essential to long-term management.17,18
Renal Tumor Monitoring
Finally, once BHD is diagnosed, one of the most important considerations is to begin routine monitoring for renal malignancies. Current recommendations advise starting lifelong renal cancer screening, even as early as age 20 years, with annual MRIs, as renal ultrasound may not be sufficiently sensitive to detect smaller lesions.19 The screening interval can be extended to every 2 years for patients without a family history of renal tumors or suspicious renal lesions. If tumors are found, then nephron-sparing surgery is recommended, given the potential for the development of chronic renal insufficiency in patients with BHD.20
Conclusions
BHD is a rare and complex syndrome in which early recognition and diagnosis play a pivotal role in preventing potentially severe complications such as renal malignancies. Suspicion of a genetic disorder, such as BHD, LAM, or PLCH, should arise in patients who experience spontaneous pneumothorax, especially in the presence of multiple cystic lesions or a family history of pneumothoraces. Early consideration of pleurodesis after the first spontaneous pneumothorax is advisable. The complex presentation of BHD contributes to the difficulty of diagnosis and may delay recognition, which can be exacerbated by variable continuity of care.
- Schmidt LS, Linehan WM. Molecular genetics and clinical features of Birt-Hogg-Dubé-Syndrome. Nat Rev Urol. 2015;12:558-569. doi:10.1038/nrurol.2015.206
- Lim DHK, Rehal PK, Nahorski MS, et al. A new locus-specific database (LSDB) for mutations in the folliculin (FLCN) gene. Hum Mutat. 2010;31:E1043-1051. doi:10.1002/humu.21130
- Aivaz O, Berkman S, Middelton L, et al. Comedonal and cystic fibrofolliculomas in Birt-Hogg-Dube syndrome. JAMA Dermatology. 2015;151:770-774. doi:10.1001/jamadermatol.2015.0215
- Daccord C, Good JM, Morren MA, et al. Birt–Hogg–Dubé syndrome. Eur Respir Rev. 2020;29:200042. doi:10.1183/16000617.0042-2020
- Menko FH, van Steensel MA, Giraud S, et al. Birt-Hogg-Dubé syndrome: diagnosis and management. The Lancet Oncology. 2009;10:1199-1206. doi:10.1016/S1470-2045(09)70188-3
- Daccord C, Cottin V, Prévot G, et al. Lung function in Birt-Hogg-Dubé syndrome: a retrospective analysis of 96 patients. Orphanet J Rare Dis. 2020;15:120. doi:10.1186/s13023-020-01402-y
- Hansell DM, Bankier AA, MacMahon H, et al. Fleischner Society: glossary of terms for thoracic imaging. Radiology. 2008;246:697-722. doi:10.1148/radiol.2462070712
- Jamil A, Kasi A. Pneumatocele. In: StatPearls. StatPearls Publishing; 2024. Accessed March 2, 2026. http://www.ncbi.nlm.nih.gov/books/NBK556146/
- Bhardwaj H, Bhardwaj B. Differentiating pulmonary lymphangioleiomyomatosis from pulmonary langerhans cell histiocytosis and Birt-Hogg-Dube syndrome. Lung India. 2013;30:372-373. doi:10.4103/0970-2113.120611
- Swigris JJ, Berry GJ, Raffin TA, et al. Lymphoid interstitial pneumonia: a narrative review. Chest. 2002;122:2150-2164. doi:10.1378/chest.122.6.2150
- Zbar B, Alvord WG, Glenn G, et al. Risk of renal and colonic neoplasms and spontaneous pneumothorax in the Birt-Hogg-Dubé syndrome. Cancer Epidemiol Biomarkers Prev. 2002;11:393-400.
- Sattler EC, Steinlein OK. Delayed diagnosis of Birt-Hogg-Dubé syndrome due to marked intrafamilial clinical variability: a case report. BMC Med Genet. 2018;19:45. doi:10.1186/s12881-018-0558-0
- Gupta N, Seyama K, McCormack FX. Pulmonary manifestations of Birt-Hogg-Dubé syndrome. Fam Cancer. 2013;12:387-396. doi:10.1007/s10689-013-9660-9
- Gupta N, Kopras EJ, Henske EP, et al. Spontaneous pneumothoraces in patients with Birt–Hogg–Dubé syndrome. Ann Am Thorac Soc. 2017;14:706-713. doi:10.1513/AnnalsATS.201611-886OC
- Mizobuchi T, Kurihara M, Ebana H, et al. A total pleural covering of absorbable cellulose mesh prevents pneumothorax recurrence in patients with Birt-Hogg-Dubé syndrome. Orphanet J Rare Dis. 2018;13:78. doi:10.1186/s13023-018-0790-x
- Johannesma PC, van de Beek I, van der Wel JWT, et al. Risk of spontaneous pneumothorax due to air travel and diving in patients with Birt–Hogg–Dubé syndrome. Springerplus. 2016;5:1506. doi:10.1186/s40064-016-3009-4
- Steinlein OK, Reithmair M, Syunyaeva Z, et al. Delayed diagnosis of Birt-Hogg-Dubé syndrome might be aggravated by gender bias. eClinicalMedicine. 2022;51:101572. doi:10.1016/j.eclinm.2022.101572
- Pereira Gray DJ, Sidaway-Lee K, White E, et al. Continuity of care with doctors—a matter of life and death? A systematic review of continuity of care and mortality. BMJ Open. 2018;8:e021161. doi:10.1136/bmjopen-2017-021161
- Sattler EC, Steinlein OK. GeneReviews Birt-Hogg-Dubé syndrome. January 30, 2020. Accessed March 2, 2026. https://www.ncbi.nlm.nih.gov/books/NBK1522/table
- Stamatakis L, Metwalli AR, Middelton LA, et al. Diagnosis and management of BHD-associated kidney cancer. Fam Cancer. 2013;12:397-402. doi:10.1007/s10689-013-9657-4
- Schmidt LS, Linehan WM. Molecular genetics and clinical features of Birt-Hogg-Dubé-Syndrome. Nat Rev Urol. 2015;12:558-569. doi:10.1038/nrurol.2015.206
- Lim DHK, Rehal PK, Nahorski MS, et al. A new locus-specific database (LSDB) for mutations in the folliculin (FLCN) gene. Hum Mutat. 2010;31:E1043-1051. doi:10.1002/humu.21130
- Aivaz O, Berkman S, Middelton L, et al. Comedonal and cystic fibrofolliculomas in Birt-Hogg-Dube syndrome. JAMA Dermatology. 2015;151:770-774. doi:10.1001/jamadermatol.2015.0215
- Daccord C, Good JM, Morren MA, et al. Birt–Hogg–Dubé syndrome. Eur Respir Rev. 2020;29:200042. doi:10.1183/16000617.0042-2020
- Menko FH, van Steensel MA, Giraud S, et al. Birt-Hogg-Dubé syndrome: diagnosis and management. The Lancet Oncology. 2009;10:1199-1206. doi:10.1016/S1470-2045(09)70188-3
- Daccord C, Cottin V, Prévot G, et al. Lung function in Birt-Hogg-Dubé syndrome: a retrospective analysis of 96 patients. Orphanet J Rare Dis. 2020;15:120. doi:10.1186/s13023-020-01402-y
- Hansell DM, Bankier AA, MacMahon H, et al. Fleischner Society: glossary of terms for thoracic imaging. Radiology. 2008;246:697-722. doi:10.1148/radiol.2462070712
- Jamil A, Kasi A. Pneumatocele. In: StatPearls. StatPearls Publishing; 2024. Accessed March 2, 2026. http://www.ncbi.nlm.nih.gov/books/NBK556146/
- Bhardwaj H, Bhardwaj B. Differentiating pulmonary lymphangioleiomyomatosis from pulmonary langerhans cell histiocytosis and Birt-Hogg-Dube syndrome. Lung India. 2013;30:372-373. doi:10.4103/0970-2113.120611
- Swigris JJ, Berry GJ, Raffin TA, et al. Lymphoid interstitial pneumonia: a narrative review. Chest. 2002;122:2150-2164. doi:10.1378/chest.122.6.2150
- Zbar B, Alvord WG, Glenn G, et al. Risk of renal and colonic neoplasms and spontaneous pneumothorax in the Birt-Hogg-Dubé syndrome. Cancer Epidemiol Biomarkers Prev. 2002;11:393-400.
- Sattler EC, Steinlein OK. Delayed diagnosis of Birt-Hogg-Dubé syndrome due to marked intrafamilial clinical variability: a case report. BMC Med Genet. 2018;19:45. doi:10.1186/s12881-018-0558-0
- Gupta N, Seyama K, McCormack FX. Pulmonary manifestations of Birt-Hogg-Dubé syndrome. Fam Cancer. 2013;12:387-396. doi:10.1007/s10689-013-9660-9
- Gupta N, Kopras EJ, Henske EP, et al. Spontaneous pneumothoraces in patients with Birt–Hogg–Dubé syndrome. Ann Am Thorac Soc. 2017;14:706-713. doi:10.1513/AnnalsATS.201611-886OC
- Mizobuchi T, Kurihara M, Ebana H, et al. A total pleural covering of absorbable cellulose mesh prevents pneumothorax recurrence in patients with Birt-Hogg-Dubé syndrome. Orphanet J Rare Dis. 2018;13:78. doi:10.1186/s13023-018-0790-x
- Johannesma PC, van de Beek I, van der Wel JWT, et al. Risk of spontaneous pneumothorax due to air travel and diving in patients with Birt–Hogg–Dubé syndrome. Springerplus. 2016;5:1506. doi:10.1186/s40064-016-3009-4
- Steinlein OK, Reithmair M, Syunyaeva Z, et al. Delayed diagnosis of Birt-Hogg-Dubé syndrome might be aggravated by gender bias. eClinicalMedicine. 2022;51:101572. doi:10.1016/j.eclinm.2022.101572
- Pereira Gray DJ, Sidaway-Lee K, White E, et al. Continuity of care with doctors—a matter of life and death? A systematic review of continuity of care and mortality. BMJ Open. 2018;8:e021161. doi:10.1136/bmjopen-2017-021161
- Sattler EC, Steinlein OK. GeneReviews Birt-Hogg-Dubé syndrome. January 30, 2020. Accessed March 2, 2026. https://www.ncbi.nlm.nih.gov/books/NBK1522/table
- Stamatakis L, Metwalli AR, Middelton LA, et al. Diagnosis and management of BHD-associated kidney cancer. Fam Cancer. 2013;12:397-402. doi:10.1007/s10689-013-9657-4
A Case of Birt-Hogg-Dubé Syndrome: A Rare but Essential Diagnosis to Consider
A Case of Birt-Hogg-Dubé Syndrome: A Rare but Essential Diagnosis to Consider
Recliner Butt
Recliner Butt
DIAGNOSIS
Senile gluteal dermatosis (SGD). SGD is a friction- related skin injury, also known as recliner butt, chronic tissue injury, or grandfather’s disease.1-4 The hallmarks include blanchable erythematous plaques and/or purplish discoloration of the fleshy part of the buttocks or posterior thighs, with little to no change over months to years. Additional findings may include skin erosions, lichenification, and ridging. SGD is most commonly seen in older adults with impaired mobility who spend prolonged periods in a reclined position, particularly those who slide down in a chair, “scoot,” or drag the buttocks during transfers or repositioning.
The pathogenesis of SGD is thought to involve microischemia associated with prolonged sitting.4 Histopathologic findings are nonspecific and may include hyperkeratosis, psoriasiform epidermal hyperplasia, vascular dilatation or proliferation in the superficial dermis, and reactive lymphohistiocytic perivascular infiltrate.4 The condition is poorly recognized and is likely underreported. Treatment involves reducing frictional injury by avoiding the reclined position, minimizing sliding during transfers, and frequent repositioning. Petroleum-based ointments may be applied to reduce friction and protect the skin barrier. Heat-dissipating chair cushions can be used to offload pressure and improve the local microclimate. Friction-related skin injuries need to be differentiated from pressure injuries, in which pressure and shear are the driving forces, and lesions are located over bony prominences.
Unlike SGD, chronic lichen sclerosus typically occurs in the anogenital area, including the scrotum and vulva, and is typically intensely pruritic, with white, atrophic plaques.
A stage 2 pressure injury is characterized by an area of partial-thickness skin loss with exposed dermis, usually overlying a bony prominence. Although friction-related skin injuries may contain erosions, they are often maroon or purple and are not located over a bony prominence.
Deep tissue injury (DTI) is characterized by nonblanchable dark red or purple skin discoloration, with intact or nonintact skin. While friction injuries may mimic DTIs, they lack the characteristic anatomic location over a bony prominence and the predictable evolution pattern seen in DTIs.
Incontinence-associated dermatitis (IAD) results from prolonged exposure to urine and/or feces and presents with erythema, inflammation, and epidermal erosion. Although IAD can look similar or coincide with SGD, the affected area is typically red, not purple. Skin ridging and lichenification are also not seen in IAD cases.
Sedentary behavior is prevalent among older adults, with nearly 60% spending > 4 hours per day sitting.5 Prolonged sitting puts them at risk for friction-related skin injuries. Even though friction-related skin injuries are typically nonprogressive, these patients are also at risk for pressure injuries that are typically acquired in a sitting position (eg, ischial and sacrococcygeal). Therefore, it is imperative that clinicians not only address SGD but also implement a pressure injury prevention plan.
- Berke CT. Pathology and clinical presentation of friction injuries case series and literature review. J Wound Ostomy Continence Nurs. 2105;42:47-61. doi:10.1097/WON.0000000000000087
- Mahoney MF, Rozenboom BJ. Definition and characteristics of chronic tissue injury: a unique form of skin damage. J Wound Ostomy Continence Nurs. 2019;46:187-191. doi:10.1097/WON.0000000000000527
- Kelechi, TJ. Commentary: chronic tissue injury. Making the case for a new form of skin damage. J Wound Ostomy Continence Nurs. 2019;46:192-193. doi:10.1097/WON.0000000000000533
- Majid I, Jairam D, Baheti K, et al. Senile guletal dermatosis: update on etiopathogenesis, Diagnostic Criteria, and Management. Dermatol Ther. 2024;37:e5556190.
- Harvey JA, Chastin SF, Skelton DA. Prevalence of sedentary behavior in older adults: a systematic review. Int J Environ Res Public Health. 2013;10:6645-6661. doi:10.3390/ijerph10126645
DIAGNOSIS
Senile gluteal dermatosis (SGD). SGD is a friction- related skin injury, also known as recliner butt, chronic tissue injury, or grandfather’s disease.1-4 The hallmarks include blanchable erythematous plaques and/or purplish discoloration of the fleshy part of the buttocks or posterior thighs, with little to no change over months to years. Additional findings may include skin erosions, lichenification, and ridging. SGD is most commonly seen in older adults with impaired mobility who spend prolonged periods in a reclined position, particularly those who slide down in a chair, “scoot,” or drag the buttocks during transfers or repositioning.
The pathogenesis of SGD is thought to involve microischemia associated with prolonged sitting.4 Histopathologic findings are nonspecific and may include hyperkeratosis, psoriasiform epidermal hyperplasia, vascular dilatation or proliferation in the superficial dermis, and reactive lymphohistiocytic perivascular infiltrate.4 The condition is poorly recognized and is likely underreported. Treatment involves reducing frictional injury by avoiding the reclined position, minimizing sliding during transfers, and frequent repositioning. Petroleum-based ointments may be applied to reduce friction and protect the skin barrier. Heat-dissipating chair cushions can be used to offload pressure and improve the local microclimate. Friction-related skin injuries need to be differentiated from pressure injuries, in which pressure and shear are the driving forces, and lesions are located over bony prominences.
Unlike SGD, chronic lichen sclerosus typically occurs in the anogenital area, including the scrotum and vulva, and is typically intensely pruritic, with white, atrophic plaques.
A stage 2 pressure injury is characterized by an area of partial-thickness skin loss with exposed dermis, usually overlying a bony prominence. Although friction-related skin injuries may contain erosions, they are often maroon or purple and are not located over a bony prominence.
Deep tissue injury (DTI) is characterized by nonblanchable dark red or purple skin discoloration, with intact or nonintact skin. While friction injuries may mimic DTIs, they lack the characteristic anatomic location over a bony prominence and the predictable evolution pattern seen in DTIs.
Incontinence-associated dermatitis (IAD) results from prolonged exposure to urine and/or feces and presents with erythema, inflammation, and epidermal erosion. Although IAD can look similar or coincide with SGD, the affected area is typically red, not purple. Skin ridging and lichenification are also not seen in IAD cases.
Sedentary behavior is prevalent among older adults, with nearly 60% spending > 4 hours per day sitting.5 Prolonged sitting puts them at risk for friction-related skin injuries. Even though friction-related skin injuries are typically nonprogressive, these patients are also at risk for pressure injuries that are typically acquired in a sitting position (eg, ischial and sacrococcygeal). Therefore, it is imperative that clinicians not only address SGD but also implement a pressure injury prevention plan.
DIAGNOSIS
Senile gluteal dermatosis (SGD). SGD is a friction- related skin injury, also known as recliner butt, chronic tissue injury, or grandfather’s disease.1-4 The hallmarks include blanchable erythematous plaques and/or purplish discoloration of the fleshy part of the buttocks or posterior thighs, with little to no change over months to years. Additional findings may include skin erosions, lichenification, and ridging. SGD is most commonly seen in older adults with impaired mobility who spend prolonged periods in a reclined position, particularly those who slide down in a chair, “scoot,” or drag the buttocks during transfers or repositioning.
The pathogenesis of SGD is thought to involve microischemia associated with prolonged sitting.4 Histopathologic findings are nonspecific and may include hyperkeratosis, psoriasiform epidermal hyperplasia, vascular dilatation or proliferation in the superficial dermis, and reactive lymphohistiocytic perivascular infiltrate.4 The condition is poorly recognized and is likely underreported. Treatment involves reducing frictional injury by avoiding the reclined position, minimizing sliding during transfers, and frequent repositioning. Petroleum-based ointments may be applied to reduce friction and protect the skin barrier. Heat-dissipating chair cushions can be used to offload pressure and improve the local microclimate. Friction-related skin injuries need to be differentiated from pressure injuries, in which pressure and shear are the driving forces, and lesions are located over bony prominences.
Unlike SGD, chronic lichen sclerosus typically occurs in the anogenital area, including the scrotum and vulva, and is typically intensely pruritic, with white, atrophic plaques.
A stage 2 pressure injury is characterized by an area of partial-thickness skin loss with exposed dermis, usually overlying a bony prominence. Although friction-related skin injuries may contain erosions, they are often maroon or purple and are not located over a bony prominence.
Deep tissue injury (DTI) is characterized by nonblanchable dark red or purple skin discoloration, with intact or nonintact skin. While friction injuries may mimic DTIs, they lack the characteristic anatomic location over a bony prominence and the predictable evolution pattern seen in DTIs.
Incontinence-associated dermatitis (IAD) results from prolonged exposure to urine and/or feces and presents with erythema, inflammation, and epidermal erosion. Although IAD can look similar or coincide with SGD, the affected area is typically red, not purple. Skin ridging and lichenification are also not seen in IAD cases.
Sedentary behavior is prevalent among older adults, with nearly 60% spending > 4 hours per day sitting.5 Prolonged sitting puts them at risk for friction-related skin injuries. Even though friction-related skin injuries are typically nonprogressive, these patients are also at risk for pressure injuries that are typically acquired in a sitting position (eg, ischial and sacrococcygeal). Therefore, it is imperative that clinicians not only address SGD but also implement a pressure injury prevention plan.
- Berke CT. Pathology and clinical presentation of friction injuries case series and literature review. J Wound Ostomy Continence Nurs. 2105;42:47-61. doi:10.1097/WON.0000000000000087
- Mahoney MF, Rozenboom BJ. Definition and characteristics of chronic tissue injury: a unique form of skin damage. J Wound Ostomy Continence Nurs. 2019;46:187-191. doi:10.1097/WON.0000000000000527
- Kelechi, TJ. Commentary: chronic tissue injury. Making the case for a new form of skin damage. J Wound Ostomy Continence Nurs. 2019;46:192-193. doi:10.1097/WON.0000000000000533
- Majid I, Jairam D, Baheti K, et al. Senile guletal dermatosis: update on etiopathogenesis, Diagnostic Criteria, and Management. Dermatol Ther. 2024;37:e5556190.
- Harvey JA, Chastin SF, Skelton DA. Prevalence of sedentary behavior in older adults: a systematic review. Int J Environ Res Public Health. 2013;10:6645-6661. doi:10.3390/ijerph10126645
- Berke CT. Pathology and clinical presentation of friction injuries case series and literature review. J Wound Ostomy Continence Nurs. 2105;42:47-61. doi:10.1097/WON.0000000000000087
- Mahoney MF, Rozenboom BJ. Definition and characteristics of chronic tissue injury: a unique form of skin damage. J Wound Ostomy Continence Nurs. 2019;46:187-191. doi:10.1097/WON.0000000000000527
- Kelechi, TJ. Commentary: chronic tissue injury. Making the case for a new form of skin damage. J Wound Ostomy Continence Nurs. 2019;46:192-193. doi:10.1097/WON.0000000000000533
- Majid I, Jairam D, Baheti K, et al. Senile guletal dermatosis: update on etiopathogenesis, Diagnostic Criteria, and Management. Dermatol Ther. 2024;37:e5556190.
- Harvey JA, Chastin SF, Skelton DA. Prevalence of sedentary behavior in older adults: a systematic review. Int J Environ Res Public Health. 2013;10:6645-6661. doi:10.3390/ijerph10126645
Recliner Butt
Recliner Butt
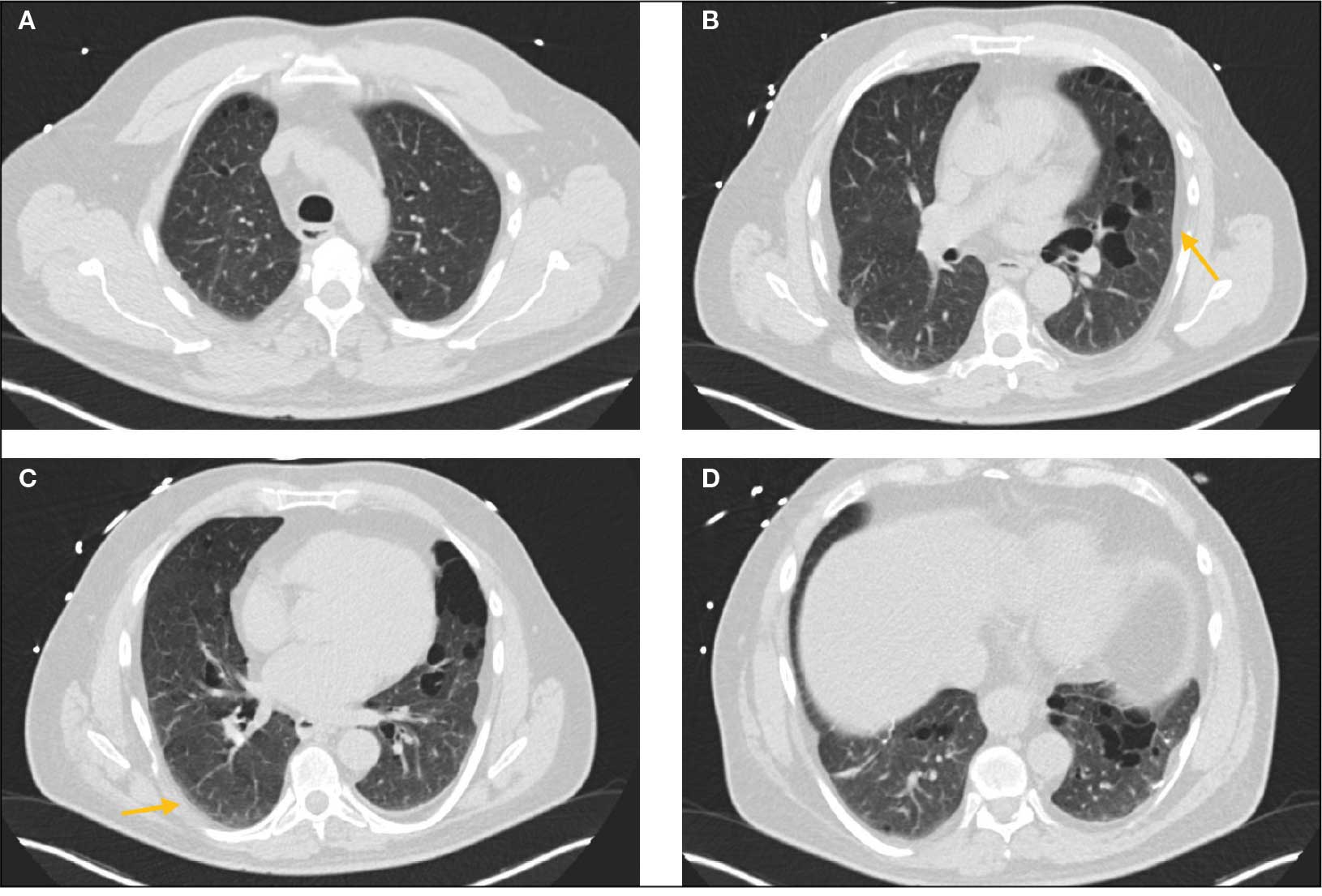
An 89-year-old male veteran with a history of obesity (body mass index, 33), osteoarthritis, anemia, pulmonary embolism, and urinary incontinence presented for evaluation of gluteal skin lesions (Figure). The patient had poor mobility and spent most of the day in a recliner chair. He also slept in the recliner due to chronic dyspnea and orthopnea.
The gluteal region demonstrated purplish discoloration with blanchable erythema and superficial ulcerations. The affected area was not pruritic and had remained unchanged for 3 months.
A punch biopsy of the discolored gluteal area was performed. Histopathologic examination revealed hyperkeratosis, orthokeratosis, irregular acanthosis, and mild spongiosis. Vascular proliferation and papillary dermal edema were also noted.
Predictors of Unplanned Postoperative Visits in a Veterans Affairs Hand Surgery Practice
Predictors of Unplanned Postoperative Visits in a Veterans Affairs Hand Surgery Practice
Patients make unplanned appointments after elective soft tissue hand surgery for real or perceived complications when they experience pain, anxiety, or fear. Unplanned appointments can create travel and financial burdens for patients and families. These appointments take time away from scheduled appointments and can contribute to late arrivals and delays in other clinics. Unscheduled appointments contribute to poor access when staff are diverted from scheduled appointments. If predictive factors can be identified, unplanned appointments may either be ameliorated or avoided with better perioperative risk management or education.
Methods
The US Department of Veterans Affairs (VA) North Florida/South Georgia Veterans Health System (NFSGVAHS) and University of Florida Institutional Review Board approved a retrospective chart review of all plastic surgery cases performed at the Malcom Randall VA Medical Center (MRVAMC) and Lake City VAMC operating rooms from July 1, 2018, through December 31, 2019, and January 1, 2021, through June 30, 2022 (nonurgent surgeries were discouraged during the COVID-19 pandemic). Elective soft tissue hand surgery cases were identified based on the operative description found in the Surgical Service Surgeon Staffing Report reviewed monthly by the Service Chief. Potential indicators of unplanned visits were recorded, including age; sex; diagnosis of diabetes, depression, anxiety, or posttraumatic stress disorder (PTSD); current smoking status; and residential zip code. We used the first 3 digits of the patients’ zip codes, which indicate region, as an estimate of proximity to the MRVAMC, which has a 50-county catchment area across North Florida and South Georgia. Diagnoses were found on the “problem list” from the electronic health record documented in the history and physical examinations before surgery. Clinic notes were examined for 3 months postsurgery to identify unplanned postoperative visits and the reason for the appointment. A χ2 analysis was conducted using Excel Version 2402. P < .05 was used to determine whether age (> 60 years), sex, proximity to MRVAMC, diabetes, smoking, depression, anxiety, or PTSD were statistically significant independent risk factors for these appointments.
Results
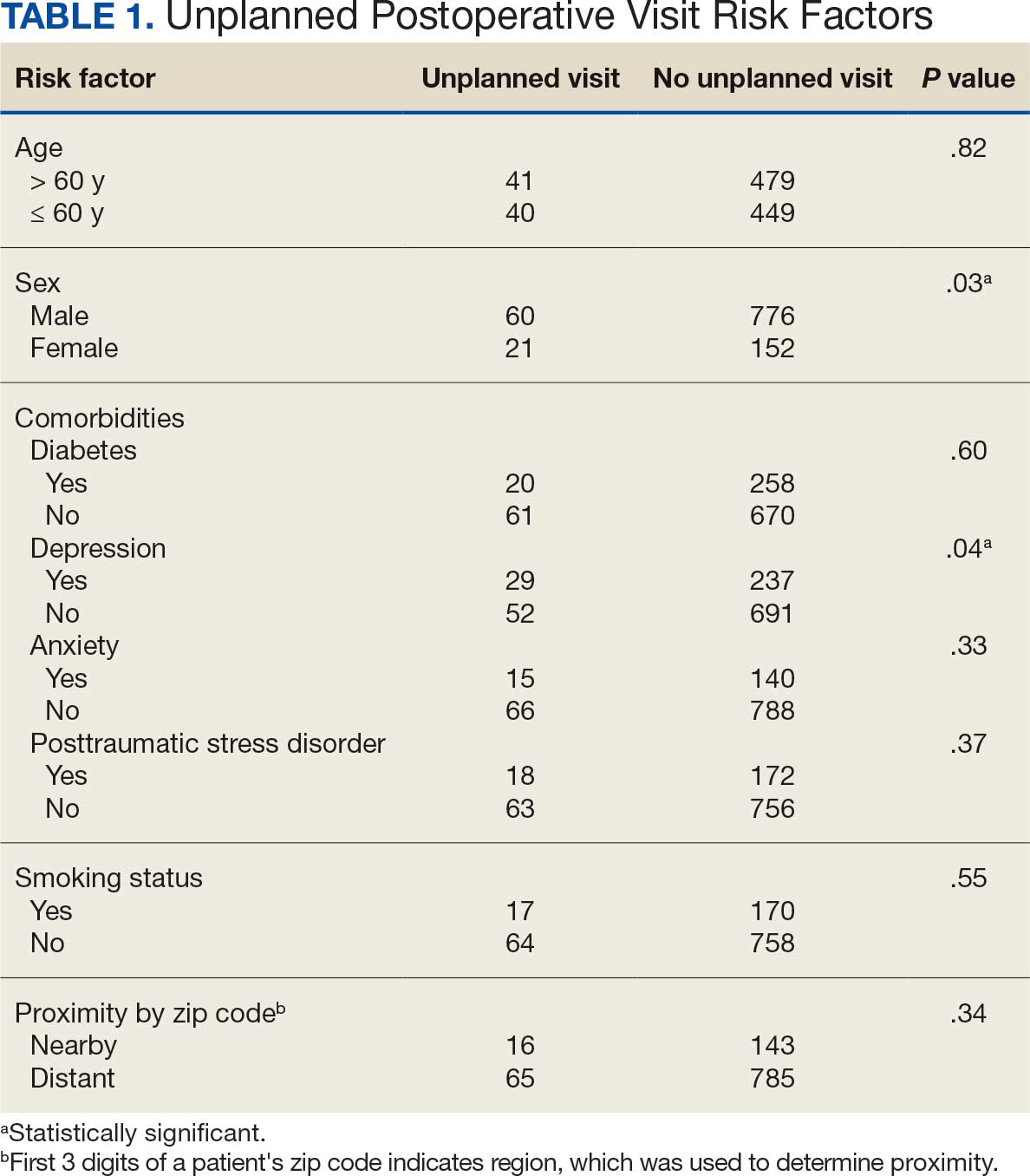
A total of 1009 elective soft tissue hand surgeries at MRVAMC were reviewed. The patients median age was 61 years. Patients included 173 women (17.1%) and 836 men (82.9%). Eighty-one patients (8.0%) returned for unplanned visits. Age (P = .82); proximity to MRVAMC (P = .34); and diabetes (P = .60), smoking (P = .55), anxiety (P = .33), or PTSD (P = .37) were not statistically significant predictors of unplanned appointments. Depression diagnosis (P = .04) and female sex (P = .03) were found to be independent risk factors for an unplanned appointment (Table 1). The most common indication for the requested appointment was pain-related, followed closely by noninfectious wound concerns and persistent symptoms (Table 2).

Discussion
Improved access, quality, and efficiency for patients are goals for the VA.1-3 The MRVAMC Plastic and Hand Surgery service provides care for the NFSGVAHS and receives an average of 15 to 20 consultation requests daily. The Veterans Health Administration is frequently challenged by staff shortages, and surgical services struggle to respond to consultation requests and treat patients within reasonable time frames.4,5
The objective of this study was to identify risk factors for unplanned postoperative appointments following elective hand surgery. Unplanned appointments prevent scheduled patients from being seen on time and contribute to backlogs and delays. When patients schedule multiple appointments on the same day, delays in the first clinic’s scheduled appointments create delays for the second and third clinics. Hand surgery clinics can provide a better experience for patients and staff by identifying and mitigating factors prompting unplanned visits.
We anticipated that wound complications would prompt unscheduled visits. Diabetes is a known risk factor for wound healing complications after plastic and hand surgery.6,7 A hemoglobin A1c (HbA1c) screening protocol used by the NFSGVAHS plastic surgery service since 2015 to identify poorly controlled patients with diabetes before surgery may partially explain this finding.8 We did not find a statistically significant difference between patients with diabetes and patients without diabetes for scheduling unplanned appointments. The plastic surgery service does not perform elective hand surgery unless the patient’s HbA1c level is < 9%, or violate the flexor sheath unless HbA1c level is < 8%. However, Zhuang et al found an increase in soft tissue infections after hand surgery with HbA1c levels ≥ 7%.9
Smoking is a potential factor in postoperative hand surgery complications.10,11 Lans et al found an increased incidence of 30-day emergency room visits in current and former smokers after outpatient upper extremity fracture surgery.12 The MRVAMC Plastic Surgery Service counsels patients about the risk of skin necrosis and delayed wound healing, but does not cancel cases or obtain laboratory values to verify abstinence in patients undergoing hand surgery. The VA has multiple resources available for patients interested in smoking cessation through mental health services.13
MRVAMC patients have been known to resist returning for scheduled appointments due to the costs or availability of transportation. We suspected that patients who lived further from MRVAMC would be less likely to return for unscheduled visits. We used the first 3 digits of the patients’ mailing zip code to estimate residential proximity to MRVAMC. An acknowledged limitation to this approach is that some veterans have primary addresses in other regions but still spend significant time in the MRVAMC catchment area and use the facility for their health care during the winter months. These “snowbirds” might reside near the facility despite having official addresses that are more distant. Additionally, there was no increased risk of unplanned visits after hand surgery in patients aged > 61 years (the median age of study participants) (P = .82). Dependence on a third party for transportation in older veterans could impact this finding.
Based on the observation that most patients needed reassurance rather than an intervention when they returned for unscheduled appointments, diagnoses of depression, anxiety, and PTSD were evaluated as separate predictive factors. In previous research, anxiety was found to be a risk factor for problematic recovery following carpal tunnel surgery.14 In the current study, depression was found to be a statistically significant predictor of unscheduled postoperative appointments (P = .04), while anxiety (P = .33) and PTSD (P = .37) were not statistically significant predictors. This is consistent with other studies that have found preexisting depression can predict complications after hand surgery.15,16 Vranceanu et al found that depression predicted pain intensity and disability after elective hand surgery.16 Similarly, Oflazoglu et al found a 12% incidence of depression based on the Patient Health Questionnaire-9 in new and returning hand patients who presented to an academic practice.17 They suggest patients should be assessed at all levels of care and that those with poor responses to surgical or nonsurgical management should be evaluated for depression. MRVAMC has a large mental health service consisting of psychiatrists, psychologists, addiction specialists, social workers, and homeless outreach, and patients tend to already have a diagnosis and mental health practitioner when they present to the clinic.
Recent studies found that wound problems, pain, and stiffness were the most common reasons for return visits.18,19 Shetty et al identified younger age, worse preoperative pain scores, and poor access to transportation as predictors of preventable emergency room visits, which generate higher health care expenditures than an office visit.19 Our study’s top reasons for appointments (pain, wound/scar concerns, persistent symptoms) can be addressed with additional presurgery patient and family education. Additionally, clinicians encourage nonnarcotic pain management strategies including anti-inflammatories, acetaminophen, elevation, splinting, and hand therapy, and the hospital employs experienced, fellowship-trained anesthesia block faculty who help limit perioperative narcotic use. Patients are advised that pain can be used to guide them through the postoperative recovery by preventing overuse and alerting them to a problem that would be masked with narcotics, and long-standing problems such as chronic nerve compressions may continue to cause pain after surgery.
Patients and families can be given consistent and repetitive verbal and written information, instructions, and expectations at the initial consultation, preoperative appointment, and on the day of surgery. Postoperatively, outside their scheduled appointments, patients are encouraged to use the My HealtheVet secure messaging system or call the clinic to access an experienced registered nurse before making a long drive. Access to virtual or phone visits can reduce emergent in-person visits in a VA population.20
Ozdag et al found that 42% of patients who had elective carpal tunnel surgery made unplanned electronic messages or phone contact within 2 weeks postsurgery. The authors point out the uncompensated administrative burden on the staff answering these messages and suggest pre-empting the contacts with more up-front education regarding postoperative pain expectations and management strategies.21
Fisher et al found that attending hand therapy reduced the number of emergency department visits in postoperative infection cases.22 At MRVAMC, a postoperative emergency department visit for a patient prompts an urgent unplanned appointment to the plastic surgery clinic, often on the same day. The MRVAMC occupational therapy clinic employed 3 on-site certified hand therapists during the study period. Because all hand surgery patients at the clinic receive hand therapy on the same day as their first postoperative appointment, attendance at hand therapy was not evaluated as a predictor of unplanned visits. Scheduled hand therapy is another point of contact where the clinic can provide reassurance and patient education.
While females made up 17.1% of the patients in this study, they constituted 12.5% of all veterans in Florida in fiscal year 2023.23 This study found that women were more likely to present for unplanned postoperative appointments (P = .03). This is consistent with existing literature which has found that women are higher users of health care and office-based appointments.24,25 This finding suggests the need for further study into whether our methods of communicating instructions to female patients undergoing plastic surgery may not be optimal.
Strengths and Limitations
As a retrospective review, the authors used information documented by multiple different health care practitioners, including trainees. The electronic medical record problem lists and templates provide consistency of information; however, less seasoned clinicians may interpret what they see and hear differently from more experienced clinicians in the postoperative setting. This study occurred in one part of the country with demographics that may not mirror other VA systems or the general population. The authors hope this study can be a starting point for other health care facilities to investigate ways to minimize the burden of unscheduled appointments. A strength of the study is that it was conducted within a closed system, as patients tend to stay within the VA system and documentation and communication among clinicians, even outside the immediate facility, are easily accessed through the electronic health record.
Conclusions
This study found that depression diagnosis and female sex are statistically significant predictors of unplanned postoperative visits after elective soft tissue hand surgery. More effective patient education during the preoperative period, particularly in patients with depression, may be warranted.
- Apaydin EA, Paige NM, Begashaw MM, et al. Veterans Health Administration (VA) vs. non-VA healthcare quality: a systematic review. J Gen Intern Med. 2023;38:2179-2188. doi:10.1007/s11606-023-08207-2
- Blegen M, Ko J, Salzman G, et al. Comparing quality of surgical care between the US Department of Veterans Affairs and non-Veterans Affairs settings: a systematic review. J Am Coll Surg. 2023;237:352-361. doi:10.1097/XCS.0000000000000720
- Valsangkar NP, Eppstein AC, Lawson RA, et al. Effect of lean processes on surgical wait times and efficiency in a tertiary care veterans affairs medical center. JAMA Surg. 2017;152:42-47. doi:10.1001/jamasurg.2016.2808
- National Association of Veterans Affairs Physicians and Dentists. Physicians remain at top of staffing shortage in VA. NAVAPD. December 20, 2023. Accessed March 16, 2026. https://www.navapd.org/news/physicians-remain-at-top-of-staffing-shortage-in-va
- OIG Determination of Veterans Health Administration’s severe occupational staffing shortages fiscal year 2024. Veterans Affairs Office of Inspector General. August 7, 2024. Accessed February 4, 2026. https://www.vaoig.gov/reports/national-healthcare-review/oig-determination-veterans-health-administrations-severe-0
- Goltsman D, Morrison KA, Ascherman JA. Defining the association between diabetes and plastic surgery outcomes: an analysis of nearly 40,000 patients. Plast Reconstr Surg Glob Open. 2017;5:e1461. doi:10.1097/GOX.0000000000001461 7.
- Cox CT, Sierra S, Egan A, et al. Elevated hemoglobin A1c and the risk of postoperative complications in elective hand and upper extremity surgery. Cureus. 2023;15:e48373. doi:10.7759/cureus.48373
- Coady-Fariborzian L, Anstead C. HbA1c and infection in diabetic elective hand surgery: a Veterans Affair Medical Center experience 2012-2018. Hand (NY). 2023;18:994-998. doi:10.1177/1558944720937363<
- Zhuang T, Shapiro LM, Fogel N, et al. Perioperative laboratory markers as risk factors for surgical site infection after elective hand surgery. J Hand Surg Am. 2021;46:675-684. doi:10.1016/j.jhsa.2021.04.001
- Cho BH, Aziz KT, Giladi AM. The impact of smoking on early postoperative complications in hand surgery. J Hand Surg Am. 2021;46:336.e1-336.e11. doi:10.1016/j.jhsa.2020.07.01411.
- Del Core MA, Ahn J, Golden AS, et al. Effect of smoking on short-term postoperative complications after elective upper extremity surgery. Hand (N Y). 2022;17:231-238. doi:10.1177/1558944720926638
- Lans J, Beagles CB, Watkins IT, et al. Unplanned postoperative emergency department visits after upper extremity fracture surgery. J Orthop Trauma. 2025;39:22-27. doi:10.1097/BOT.0000000000002925
- Tobacco and health - how to quit. US Dept of Veterans Affairs. Updated October 29, 2025. Accessed February 4, 2026. https://www.mentalhealth.va.gov/quit-tobacco/how-to-quit.asp
- Ryan C, Miner H, Ramachandran S, et al. General anxiety is associated with problematic initial recovery after carpal tunnel release. Clin Orthop Relat Res. 2022;480:1576-1581. doi:10.1097/CORR.0000000000002115
- Crijns TJ, Bernstein DN, Ring D, et al. Depression and pain interference correlate with physical function in patients recovering from hand surgery. Hand (N Y). 2019;14:830-835. doi:10.1177/1558944718777814
- Vranceanu AM, Jupiter JB, Mudgal CS, et al. Predictors of pain intensity and disability after minor hand surgery. J Hand Surg Am. 2010;35:956-960. doi:10.1016/j.jhsa.2010.02.00117.
- Oflazoglu K, Mellema JJ, Menendez ME, et al. Prevalence of and factors associated with major depression in patients with upper extremity conditions. J Hand Surg Am. 2016;41:263-269. doi:10.1016/j.jhsa.2015.11.019
- Townsend CB, Henry TW, Lutsky KF, et al. Unplanned office visits following outpatient hand surgery. Hand (N Y). 2022;17:1264-1268. doi:10.1177/15589447211028932
- Shetty PN, Guarino GM, Zhang G, et al. Risk factors for preventable emergency department use after outpatient hand surgery. J Hand Surg Am. 2022;47:855-864. doi:10.1016/j.jhsa.2022.05.012
- Sommers-Olson B, Christianson J, Neumann T, et al. Reducing nonemergent visits to the emergency department in a Veterans Affairs multistate system. J Emerg Nurs. 2023;49:539-545. doi:10.1016/j.jen.2023.02.010
- Ozdag Y, Manzar S, El Koussaify J, et al. Unplanned postoperative phone calls and electronic messages for patients with and without opioid prescriptions after carpal tunnel release. J Hand Surg Glob Online. 2024;6:363-368. doi:10.1016/j.jhsg.2024.02.006
- Fisher AH, Gandhi J, Nelson Z, et al. Immediate interventions after surgery to reduce readmission for upper extremity infections. Ann Plast Surg. 2022;88:S163-S169. doi:10.1097/SAP.0000000000003141
- Florida Department of Veterans Affairs Fast Facts. Florida Department of Veterans Affairs. Accessed February 4, 2026. https://floridavets.org/our-veterans/profilefast-facts/
- Bertakis KD, Azari R, Helms LJ, et al. Gender differences in the utilization of health care services. J Fam Pract. 2000;49:147-152.
- Ashman JJ, Santo L, Okeyode T. Characteristics of office-based physician visits, 2018. NCHS Data Brief. 2021;408:1-8.
Patients make unplanned appointments after elective soft tissue hand surgery for real or perceived complications when they experience pain, anxiety, or fear. Unplanned appointments can create travel and financial burdens for patients and families. These appointments take time away from scheduled appointments and can contribute to late arrivals and delays in other clinics. Unscheduled appointments contribute to poor access when staff are diverted from scheduled appointments. If predictive factors can be identified, unplanned appointments may either be ameliorated or avoided with better perioperative risk management or education.
Methods
The US Department of Veterans Affairs (VA) North Florida/South Georgia Veterans Health System (NFSGVAHS) and University of Florida Institutional Review Board approved a retrospective chart review of all plastic surgery cases performed at the Malcom Randall VA Medical Center (MRVAMC) and Lake City VAMC operating rooms from July 1, 2018, through December 31, 2019, and January 1, 2021, through June 30, 2022 (nonurgent surgeries were discouraged during the COVID-19 pandemic). Elective soft tissue hand surgery cases were identified based on the operative description found in the Surgical Service Surgeon Staffing Report reviewed monthly by the Service Chief. Potential indicators of unplanned visits were recorded, including age; sex; diagnosis of diabetes, depression, anxiety, or posttraumatic stress disorder (PTSD); current smoking status; and residential zip code. We used the first 3 digits of the patients’ zip codes, which indicate region, as an estimate of proximity to the MRVAMC, which has a 50-county catchment area across North Florida and South Georgia. Diagnoses were found on the “problem list” from the electronic health record documented in the history and physical examinations before surgery. Clinic notes were examined for 3 months postsurgery to identify unplanned postoperative visits and the reason for the appointment. A χ2 analysis was conducted using Excel Version 2402. P < .05 was used to determine whether age (> 60 years), sex, proximity to MRVAMC, diabetes, smoking, depression, anxiety, or PTSD were statistically significant independent risk factors for these appointments.
Results
A total of 1009 elective soft tissue hand surgeries at MRVAMC were reviewed. The patients median age was 61 years. Patients included 173 women (17.1%) and 836 men (82.9%). Eighty-one patients (8.0%) returned for unplanned visits. Age (P = .82); proximity to MRVAMC (P = .34); and diabetes (P = .60), smoking (P = .55), anxiety (P = .33), or PTSD (P = .37) were not statistically significant predictors of unplanned appointments. Depression diagnosis (P = .04) and female sex (P = .03) were found to be independent risk factors for an unplanned appointment (Table 1). The most common indication for the requested appointment was pain-related, followed closely by noninfectious wound concerns and persistent symptoms (Table 2).


Discussion
Improved access, quality, and efficiency for patients are goals for the VA.1-3 The MRVAMC Plastic and Hand Surgery service provides care for the NFSGVAHS and receives an average of 15 to 20 consultation requests daily. The Veterans Health Administration is frequently challenged by staff shortages, and surgical services struggle to respond to consultation requests and treat patients within reasonable time frames.4,5
The objective of this study was to identify risk factors for unplanned postoperative appointments following elective hand surgery. Unplanned appointments prevent scheduled patients from being seen on time and contribute to backlogs and delays. When patients schedule multiple appointments on the same day, delays in the first clinic’s scheduled appointments create delays for the second and third clinics. Hand surgery clinics can provide a better experience for patients and staff by identifying and mitigating factors prompting unplanned visits.
We anticipated that wound complications would prompt unscheduled visits. Diabetes is a known risk factor for wound healing complications after plastic and hand surgery.6,7 A hemoglobin A1c (HbA1c) screening protocol used by the NFSGVAHS plastic surgery service since 2015 to identify poorly controlled patients with diabetes before surgery may partially explain this finding.8 We did not find a statistically significant difference between patients with diabetes and patients without diabetes for scheduling unplanned appointments. The plastic surgery service does not perform elective hand surgery unless the patient’s HbA1c level is < 9%, or violate the flexor sheath unless HbA1c level is < 8%. However, Zhuang et al found an increase in soft tissue infections after hand surgery with HbA1c levels ≥ 7%.9
Smoking is a potential factor in postoperative hand surgery complications.10,11 Lans et al found an increased incidence of 30-day emergency room visits in current and former smokers after outpatient upper extremity fracture surgery.12 The MRVAMC Plastic Surgery Service counsels patients about the risk of skin necrosis and delayed wound healing, but does not cancel cases or obtain laboratory values to verify abstinence in patients undergoing hand surgery. The VA has multiple resources available for patients interested in smoking cessation through mental health services.13
MRVAMC patients have been known to resist returning for scheduled appointments due to the costs or availability of transportation. We suspected that patients who lived further from MRVAMC would be less likely to return for unscheduled visits. We used the first 3 digits of the patients’ mailing zip code to estimate residential proximity to MRVAMC. An acknowledged limitation to this approach is that some veterans have primary addresses in other regions but still spend significant time in the MRVAMC catchment area and use the facility for their health care during the winter months. These “snowbirds” might reside near the facility despite having official addresses that are more distant. Additionally, there was no increased risk of unplanned visits after hand surgery in patients aged > 61 years (the median age of study participants) (P = .82). Dependence on a third party for transportation in older veterans could impact this finding.
Based on the observation that most patients needed reassurance rather than an intervention when they returned for unscheduled appointments, diagnoses of depression, anxiety, and PTSD were evaluated as separate predictive factors. In previous research, anxiety was found to be a risk factor for problematic recovery following carpal tunnel surgery.14 In the current study, depression was found to be a statistically significant predictor of unscheduled postoperative appointments (P = .04), while anxiety (P = .33) and PTSD (P = .37) were not statistically significant predictors. This is consistent with other studies that have found preexisting depression can predict complications after hand surgery.15,16 Vranceanu et al found that depression predicted pain intensity and disability after elective hand surgery.16 Similarly, Oflazoglu et al found a 12% incidence of depression based on the Patient Health Questionnaire-9 in new and returning hand patients who presented to an academic practice.17 They suggest patients should be assessed at all levels of care and that those with poor responses to surgical or nonsurgical management should be evaluated for depression. MRVAMC has a large mental health service consisting of psychiatrists, psychologists, addiction specialists, social workers, and homeless outreach, and patients tend to already have a diagnosis and mental health practitioner when they present to the clinic.
Recent studies found that wound problems, pain, and stiffness were the most common reasons for return visits.18,19 Shetty et al identified younger age, worse preoperative pain scores, and poor access to transportation as predictors of preventable emergency room visits, which generate higher health care expenditures than an office visit.19 Our study’s top reasons for appointments (pain, wound/scar concerns, persistent symptoms) can be addressed with additional presurgery patient and family education. Additionally, clinicians encourage nonnarcotic pain management strategies including anti-inflammatories, acetaminophen, elevation, splinting, and hand therapy, and the hospital employs experienced, fellowship-trained anesthesia block faculty who help limit perioperative narcotic use. Patients are advised that pain can be used to guide them through the postoperative recovery by preventing overuse and alerting them to a problem that would be masked with narcotics, and long-standing problems such as chronic nerve compressions may continue to cause pain after surgery.
Patients and families can be given consistent and repetitive verbal and written information, instructions, and expectations at the initial consultation, preoperative appointment, and on the day of surgery. Postoperatively, outside their scheduled appointments, patients are encouraged to use the My HealtheVet secure messaging system or call the clinic to access an experienced registered nurse before making a long drive. Access to virtual or phone visits can reduce emergent in-person visits in a VA population.20
Ozdag et al found that 42% of patients who had elective carpal tunnel surgery made unplanned electronic messages or phone contact within 2 weeks postsurgery. The authors point out the uncompensated administrative burden on the staff answering these messages and suggest pre-empting the contacts with more up-front education regarding postoperative pain expectations and management strategies.21
Fisher et al found that attending hand therapy reduced the number of emergency department visits in postoperative infection cases.22 At MRVAMC, a postoperative emergency department visit for a patient prompts an urgent unplanned appointment to the plastic surgery clinic, often on the same day. The MRVAMC occupational therapy clinic employed 3 on-site certified hand therapists during the study period. Because all hand surgery patients at the clinic receive hand therapy on the same day as their first postoperative appointment, attendance at hand therapy was not evaluated as a predictor of unplanned visits. Scheduled hand therapy is another point of contact where the clinic can provide reassurance and patient education.
While females made up 17.1% of the patients in this study, they constituted 12.5% of all veterans in Florida in fiscal year 2023.23 This study found that women were more likely to present for unplanned postoperative appointments (P = .03). This is consistent with existing literature which has found that women are higher users of health care and office-based appointments.24,25 This finding suggests the need for further study into whether our methods of communicating instructions to female patients undergoing plastic surgery may not be optimal.
Strengths and Limitations
As a retrospective review, the authors used information documented by multiple different health care practitioners, including trainees. The electronic medical record problem lists and templates provide consistency of information; however, less seasoned clinicians may interpret what they see and hear differently from more experienced clinicians in the postoperative setting. This study occurred in one part of the country with demographics that may not mirror other VA systems or the general population. The authors hope this study can be a starting point for other health care facilities to investigate ways to minimize the burden of unscheduled appointments. A strength of the study is that it was conducted within a closed system, as patients tend to stay within the VA system and documentation and communication among clinicians, even outside the immediate facility, are easily accessed through the electronic health record.
Conclusions
This study found that depression diagnosis and female sex are statistically significant predictors of unplanned postoperative visits after elective soft tissue hand surgery. More effective patient education during the preoperative period, particularly in patients with depression, may be warranted.
Patients make unplanned appointments after elective soft tissue hand surgery for real or perceived complications when they experience pain, anxiety, or fear. Unplanned appointments can create travel and financial burdens for patients and families. These appointments take time away from scheduled appointments and can contribute to late arrivals and delays in other clinics. Unscheduled appointments contribute to poor access when staff are diverted from scheduled appointments. If predictive factors can be identified, unplanned appointments may either be ameliorated or avoided with better perioperative risk management or education.
Methods
The US Department of Veterans Affairs (VA) North Florida/South Georgia Veterans Health System (NFSGVAHS) and University of Florida Institutional Review Board approved a retrospective chart review of all plastic surgery cases performed at the Malcom Randall VA Medical Center (MRVAMC) and Lake City VAMC operating rooms from July 1, 2018, through December 31, 2019, and January 1, 2021, through June 30, 2022 (nonurgent surgeries were discouraged during the COVID-19 pandemic). Elective soft tissue hand surgery cases were identified based on the operative description found in the Surgical Service Surgeon Staffing Report reviewed monthly by the Service Chief. Potential indicators of unplanned visits were recorded, including age; sex; diagnosis of diabetes, depression, anxiety, or posttraumatic stress disorder (PTSD); current smoking status; and residential zip code. We used the first 3 digits of the patients’ zip codes, which indicate region, as an estimate of proximity to the MRVAMC, which has a 50-county catchment area across North Florida and South Georgia. Diagnoses were found on the “problem list” from the electronic health record documented in the history and physical examinations before surgery. Clinic notes were examined for 3 months postsurgery to identify unplanned postoperative visits and the reason for the appointment. A χ2 analysis was conducted using Excel Version 2402. P < .05 was used to determine whether age (> 60 years), sex, proximity to MRVAMC, diabetes, smoking, depression, anxiety, or PTSD were statistically significant independent risk factors for these appointments.
Results
A total of 1009 elective soft tissue hand surgeries at MRVAMC were reviewed. The patients median age was 61 years. Patients included 173 women (17.1%) and 836 men (82.9%). Eighty-one patients (8.0%) returned for unplanned visits. Age (P = .82); proximity to MRVAMC (P = .34); and diabetes (P = .60), smoking (P = .55), anxiety (P = .33), or PTSD (P = .37) were not statistically significant predictors of unplanned appointments. Depression diagnosis (P = .04) and female sex (P = .03) were found to be independent risk factors for an unplanned appointment (Table 1). The most common indication for the requested appointment was pain-related, followed closely by noninfectious wound concerns and persistent symptoms (Table 2).


Discussion
Improved access, quality, and efficiency for patients are goals for the VA.1-3 The MRVAMC Plastic and Hand Surgery service provides care for the NFSGVAHS and receives an average of 15 to 20 consultation requests daily. The Veterans Health Administration is frequently challenged by staff shortages, and surgical services struggle to respond to consultation requests and treat patients within reasonable time frames.4,5
The objective of this study was to identify risk factors for unplanned postoperative appointments following elective hand surgery. Unplanned appointments prevent scheduled patients from being seen on time and contribute to backlogs and delays. When patients schedule multiple appointments on the same day, delays in the first clinic’s scheduled appointments create delays for the second and third clinics. Hand surgery clinics can provide a better experience for patients and staff by identifying and mitigating factors prompting unplanned visits.
We anticipated that wound complications would prompt unscheduled visits. Diabetes is a known risk factor for wound healing complications after plastic and hand surgery.6,7 A hemoglobin A1c (HbA1c) screening protocol used by the NFSGVAHS plastic surgery service since 2015 to identify poorly controlled patients with diabetes before surgery may partially explain this finding.8 We did not find a statistically significant difference between patients with diabetes and patients without diabetes for scheduling unplanned appointments. The plastic surgery service does not perform elective hand surgery unless the patient’s HbA1c level is < 9%, or violate the flexor sheath unless HbA1c level is < 8%. However, Zhuang et al found an increase in soft tissue infections after hand surgery with HbA1c levels ≥ 7%.9
Smoking is a potential factor in postoperative hand surgery complications.10,11 Lans et al found an increased incidence of 30-day emergency room visits in current and former smokers after outpatient upper extremity fracture surgery.12 The MRVAMC Plastic Surgery Service counsels patients about the risk of skin necrosis and delayed wound healing, but does not cancel cases or obtain laboratory values to verify abstinence in patients undergoing hand surgery. The VA has multiple resources available for patients interested in smoking cessation through mental health services.13
MRVAMC patients have been known to resist returning for scheduled appointments due to the costs or availability of transportation. We suspected that patients who lived further from MRVAMC would be less likely to return for unscheduled visits. We used the first 3 digits of the patients’ mailing zip code to estimate residential proximity to MRVAMC. An acknowledged limitation to this approach is that some veterans have primary addresses in other regions but still spend significant time in the MRVAMC catchment area and use the facility for their health care during the winter months. These “snowbirds” might reside near the facility despite having official addresses that are more distant. Additionally, there was no increased risk of unplanned visits after hand surgery in patients aged > 61 years (the median age of study participants) (P = .82). Dependence on a third party for transportation in older veterans could impact this finding.
Based on the observation that most patients needed reassurance rather than an intervention when they returned for unscheduled appointments, diagnoses of depression, anxiety, and PTSD were evaluated as separate predictive factors. In previous research, anxiety was found to be a risk factor for problematic recovery following carpal tunnel surgery.14 In the current study, depression was found to be a statistically significant predictor of unscheduled postoperative appointments (P = .04), while anxiety (P = .33) and PTSD (P = .37) were not statistically significant predictors. This is consistent with other studies that have found preexisting depression can predict complications after hand surgery.15,16 Vranceanu et al found that depression predicted pain intensity and disability after elective hand surgery.16 Similarly, Oflazoglu et al found a 12% incidence of depression based on the Patient Health Questionnaire-9 in new and returning hand patients who presented to an academic practice.17 They suggest patients should be assessed at all levels of care and that those with poor responses to surgical or nonsurgical management should be evaluated for depression. MRVAMC has a large mental health service consisting of psychiatrists, psychologists, addiction specialists, social workers, and homeless outreach, and patients tend to already have a diagnosis and mental health practitioner when they present to the clinic.
Recent studies found that wound problems, pain, and stiffness were the most common reasons for return visits.18,19 Shetty et al identified younger age, worse preoperative pain scores, and poor access to transportation as predictors of preventable emergency room visits, which generate higher health care expenditures than an office visit.19 Our study’s top reasons for appointments (pain, wound/scar concerns, persistent symptoms) can be addressed with additional presurgery patient and family education. Additionally, clinicians encourage nonnarcotic pain management strategies including anti-inflammatories, acetaminophen, elevation, splinting, and hand therapy, and the hospital employs experienced, fellowship-trained anesthesia block faculty who help limit perioperative narcotic use. Patients are advised that pain can be used to guide them through the postoperative recovery by preventing overuse and alerting them to a problem that would be masked with narcotics, and long-standing problems such as chronic nerve compressions may continue to cause pain after surgery.
Patients and families can be given consistent and repetitive verbal and written information, instructions, and expectations at the initial consultation, preoperative appointment, and on the day of surgery. Postoperatively, outside their scheduled appointments, patients are encouraged to use the My HealtheVet secure messaging system or call the clinic to access an experienced registered nurse before making a long drive. Access to virtual or phone visits can reduce emergent in-person visits in a VA population.20
Ozdag et al found that 42% of patients who had elective carpal tunnel surgery made unplanned electronic messages or phone contact within 2 weeks postsurgery. The authors point out the uncompensated administrative burden on the staff answering these messages and suggest pre-empting the contacts with more up-front education regarding postoperative pain expectations and management strategies.21
Fisher et al found that attending hand therapy reduced the number of emergency department visits in postoperative infection cases.22 At MRVAMC, a postoperative emergency department visit for a patient prompts an urgent unplanned appointment to the plastic surgery clinic, often on the same day. The MRVAMC occupational therapy clinic employed 3 on-site certified hand therapists during the study period. Because all hand surgery patients at the clinic receive hand therapy on the same day as their first postoperative appointment, attendance at hand therapy was not evaluated as a predictor of unplanned visits. Scheduled hand therapy is another point of contact where the clinic can provide reassurance and patient education.
While females made up 17.1% of the patients in this study, they constituted 12.5% of all veterans in Florida in fiscal year 2023.23 This study found that women were more likely to present for unplanned postoperative appointments (P = .03). This is consistent with existing literature which has found that women are higher users of health care and office-based appointments.24,25 This finding suggests the need for further study into whether our methods of communicating instructions to female patients undergoing plastic surgery may not be optimal.
Strengths and Limitations
As a retrospective review, the authors used information documented by multiple different health care practitioners, including trainees. The electronic medical record problem lists and templates provide consistency of information; however, less seasoned clinicians may interpret what they see and hear differently from more experienced clinicians in the postoperative setting. This study occurred in one part of the country with demographics that may not mirror other VA systems or the general population. The authors hope this study can be a starting point for other health care facilities to investigate ways to minimize the burden of unscheduled appointments. A strength of the study is that it was conducted within a closed system, as patients tend to stay within the VA system and documentation and communication among clinicians, even outside the immediate facility, are easily accessed through the electronic health record.
Conclusions
This study found that depression diagnosis and female sex are statistically significant predictors of unplanned postoperative visits after elective soft tissue hand surgery. More effective patient education during the preoperative period, particularly in patients with depression, may be warranted.
- Apaydin EA, Paige NM, Begashaw MM, et al. Veterans Health Administration (VA) vs. non-VA healthcare quality: a systematic review. J Gen Intern Med. 2023;38:2179-2188. doi:10.1007/s11606-023-08207-2
- Blegen M, Ko J, Salzman G, et al. Comparing quality of surgical care between the US Department of Veterans Affairs and non-Veterans Affairs settings: a systematic review. J Am Coll Surg. 2023;237:352-361. doi:10.1097/XCS.0000000000000720
- Valsangkar NP, Eppstein AC, Lawson RA, et al. Effect of lean processes on surgical wait times and efficiency in a tertiary care veterans affairs medical center. JAMA Surg. 2017;152:42-47. doi:10.1001/jamasurg.2016.2808
- National Association of Veterans Affairs Physicians and Dentists. Physicians remain at top of staffing shortage in VA. NAVAPD. December 20, 2023. Accessed March 16, 2026. https://www.navapd.org/news/physicians-remain-at-top-of-staffing-shortage-in-va
- OIG Determination of Veterans Health Administration’s severe occupational staffing shortages fiscal year 2024. Veterans Affairs Office of Inspector General. August 7, 2024. Accessed February 4, 2026. https://www.vaoig.gov/reports/national-healthcare-review/oig-determination-veterans-health-administrations-severe-0
- Goltsman D, Morrison KA, Ascherman JA. Defining the association between diabetes and plastic surgery outcomes: an analysis of nearly 40,000 patients. Plast Reconstr Surg Glob Open. 2017;5:e1461. doi:10.1097/GOX.0000000000001461 7.
- Cox CT, Sierra S, Egan A, et al. Elevated hemoglobin A1c and the risk of postoperative complications in elective hand and upper extremity surgery. Cureus. 2023;15:e48373. doi:10.7759/cureus.48373
- Coady-Fariborzian L, Anstead C. HbA1c and infection in diabetic elective hand surgery: a Veterans Affair Medical Center experience 2012-2018. Hand (NY). 2023;18:994-998. doi:10.1177/1558944720937363<
- Zhuang T, Shapiro LM, Fogel N, et al. Perioperative laboratory markers as risk factors for surgical site infection after elective hand surgery. J Hand Surg Am. 2021;46:675-684. doi:10.1016/j.jhsa.2021.04.001
- Cho BH, Aziz KT, Giladi AM. The impact of smoking on early postoperative complications in hand surgery. J Hand Surg Am. 2021;46:336.e1-336.e11. doi:10.1016/j.jhsa.2020.07.01411.
- Del Core MA, Ahn J, Golden AS, et al. Effect of smoking on short-term postoperative complications after elective upper extremity surgery. Hand (N Y). 2022;17:231-238. doi:10.1177/1558944720926638
- Lans J, Beagles CB, Watkins IT, et al. Unplanned postoperative emergency department visits after upper extremity fracture surgery. J Orthop Trauma. 2025;39:22-27. doi:10.1097/BOT.0000000000002925
- Tobacco and health - how to quit. US Dept of Veterans Affairs. Updated October 29, 2025. Accessed February 4, 2026. https://www.mentalhealth.va.gov/quit-tobacco/how-to-quit.asp
- Ryan C, Miner H, Ramachandran S, et al. General anxiety is associated with problematic initial recovery after carpal tunnel release. Clin Orthop Relat Res. 2022;480:1576-1581. doi:10.1097/CORR.0000000000002115
- Crijns TJ, Bernstein DN, Ring D, et al. Depression and pain interference correlate with physical function in patients recovering from hand surgery. Hand (N Y). 2019;14:830-835. doi:10.1177/1558944718777814
- Vranceanu AM, Jupiter JB, Mudgal CS, et al. Predictors of pain intensity and disability after minor hand surgery. J Hand Surg Am. 2010;35:956-960. doi:10.1016/j.jhsa.2010.02.00117.
- Oflazoglu K, Mellema JJ, Menendez ME, et al. Prevalence of and factors associated with major depression in patients with upper extremity conditions. J Hand Surg Am. 2016;41:263-269. doi:10.1016/j.jhsa.2015.11.019
- Townsend CB, Henry TW, Lutsky KF, et al. Unplanned office visits following outpatient hand surgery. Hand (N Y). 2022;17:1264-1268. doi:10.1177/15589447211028932
- Shetty PN, Guarino GM, Zhang G, et al. Risk factors for preventable emergency department use after outpatient hand surgery. J Hand Surg Am. 2022;47:855-864. doi:10.1016/j.jhsa.2022.05.012
- Sommers-Olson B, Christianson J, Neumann T, et al. Reducing nonemergent visits to the emergency department in a Veterans Affairs multistate system. J Emerg Nurs. 2023;49:539-545. doi:10.1016/j.jen.2023.02.010
- Ozdag Y, Manzar S, El Koussaify J, et al. Unplanned postoperative phone calls and electronic messages for patients with and without opioid prescriptions after carpal tunnel release. J Hand Surg Glob Online. 2024;6:363-368. doi:10.1016/j.jhsg.2024.02.006
- Fisher AH, Gandhi J, Nelson Z, et al. Immediate interventions after surgery to reduce readmission for upper extremity infections. Ann Plast Surg. 2022;88:S163-S169. doi:10.1097/SAP.0000000000003141
- Florida Department of Veterans Affairs Fast Facts. Florida Department of Veterans Affairs. Accessed February 4, 2026. https://floridavets.org/our-veterans/profilefast-facts/
- Bertakis KD, Azari R, Helms LJ, et al. Gender differences in the utilization of health care services. J Fam Pract. 2000;49:147-152.
- Ashman JJ, Santo L, Okeyode T. Characteristics of office-based physician visits, 2018. NCHS Data Brief. 2021;408:1-8.
- Apaydin EA, Paige NM, Begashaw MM, et al. Veterans Health Administration (VA) vs. non-VA healthcare quality: a systematic review. J Gen Intern Med. 2023;38:2179-2188. doi:10.1007/s11606-023-08207-2
- Blegen M, Ko J, Salzman G, et al. Comparing quality of surgical care between the US Department of Veterans Affairs and non-Veterans Affairs settings: a systematic review. J Am Coll Surg. 2023;237:352-361. doi:10.1097/XCS.0000000000000720
- Valsangkar NP, Eppstein AC, Lawson RA, et al. Effect of lean processes on surgical wait times and efficiency in a tertiary care veterans affairs medical center. JAMA Surg. 2017;152:42-47. doi:10.1001/jamasurg.2016.2808
- National Association of Veterans Affairs Physicians and Dentists. Physicians remain at top of staffing shortage in VA. NAVAPD. December 20, 2023. Accessed March 16, 2026. https://www.navapd.org/news/physicians-remain-at-top-of-staffing-shortage-in-va
- OIG Determination of Veterans Health Administration’s severe occupational staffing shortages fiscal year 2024. Veterans Affairs Office of Inspector General. August 7, 2024. Accessed February 4, 2026. https://www.vaoig.gov/reports/national-healthcare-review/oig-determination-veterans-health-administrations-severe-0
- Goltsman D, Morrison KA, Ascherman JA. Defining the association between diabetes and plastic surgery outcomes: an analysis of nearly 40,000 patients. Plast Reconstr Surg Glob Open. 2017;5:e1461. doi:10.1097/GOX.0000000000001461 7.
- Cox CT, Sierra S, Egan A, et al. Elevated hemoglobin A1c and the risk of postoperative complications in elective hand and upper extremity surgery. Cureus. 2023;15:e48373. doi:10.7759/cureus.48373
- Coady-Fariborzian L, Anstead C. HbA1c and infection in diabetic elective hand surgery: a Veterans Affair Medical Center experience 2012-2018. Hand (NY). 2023;18:994-998. doi:10.1177/1558944720937363<
- Zhuang T, Shapiro LM, Fogel N, et al. Perioperative laboratory markers as risk factors for surgical site infection after elective hand surgery. J Hand Surg Am. 2021;46:675-684. doi:10.1016/j.jhsa.2021.04.001
- Cho BH, Aziz KT, Giladi AM. The impact of smoking on early postoperative complications in hand surgery. J Hand Surg Am. 2021;46:336.e1-336.e11. doi:10.1016/j.jhsa.2020.07.01411.
- Del Core MA, Ahn J, Golden AS, et al. Effect of smoking on short-term postoperative complications after elective upper extremity surgery. Hand (N Y). 2022;17:231-238. doi:10.1177/1558944720926638
- Lans J, Beagles CB, Watkins IT, et al. Unplanned postoperative emergency department visits after upper extremity fracture surgery. J Orthop Trauma. 2025;39:22-27. doi:10.1097/BOT.0000000000002925
- Tobacco and health - how to quit. US Dept of Veterans Affairs. Updated October 29, 2025. Accessed February 4, 2026. https://www.mentalhealth.va.gov/quit-tobacco/how-to-quit.asp
- Ryan C, Miner H, Ramachandran S, et al. General anxiety is associated with problematic initial recovery after carpal tunnel release. Clin Orthop Relat Res. 2022;480:1576-1581. doi:10.1097/CORR.0000000000002115
- Crijns TJ, Bernstein DN, Ring D, et al. Depression and pain interference correlate with physical function in patients recovering from hand surgery. Hand (N Y). 2019;14:830-835. doi:10.1177/1558944718777814
- Vranceanu AM, Jupiter JB, Mudgal CS, et al. Predictors of pain intensity and disability after minor hand surgery. J Hand Surg Am. 2010;35:956-960. doi:10.1016/j.jhsa.2010.02.00117.
- Oflazoglu K, Mellema JJ, Menendez ME, et al. Prevalence of and factors associated with major depression in patients with upper extremity conditions. J Hand Surg Am. 2016;41:263-269. doi:10.1016/j.jhsa.2015.11.019
- Townsend CB, Henry TW, Lutsky KF, et al. Unplanned office visits following outpatient hand surgery. Hand (N Y). 2022;17:1264-1268. doi:10.1177/15589447211028932
- Shetty PN, Guarino GM, Zhang G, et al. Risk factors for preventable emergency department use after outpatient hand surgery. J Hand Surg Am. 2022;47:855-864. doi:10.1016/j.jhsa.2022.05.012
- Sommers-Olson B, Christianson J, Neumann T, et al. Reducing nonemergent visits to the emergency department in a Veterans Affairs multistate system. J Emerg Nurs. 2023;49:539-545. doi:10.1016/j.jen.2023.02.010
- Ozdag Y, Manzar S, El Koussaify J, et al. Unplanned postoperative phone calls and electronic messages for patients with and without opioid prescriptions after carpal tunnel release. J Hand Surg Glob Online. 2024;6:363-368. doi:10.1016/j.jhsg.2024.02.006
- Fisher AH, Gandhi J, Nelson Z, et al. Immediate interventions after surgery to reduce readmission for upper extremity infections. Ann Plast Surg. 2022;88:S163-S169. doi:10.1097/SAP.0000000000003141
- Florida Department of Veterans Affairs Fast Facts. Florida Department of Veterans Affairs. Accessed February 4, 2026. https://floridavets.org/our-veterans/profilefast-facts/
- Bertakis KD, Azari R, Helms LJ, et al. Gender differences in the utilization of health care services. J Fam Pract. 2000;49:147-152.
- Ashman JJ, Santo L, Okeyode T. Characteristics of office-based physician visits, 2018. NCHS Data Brief. 2021;408:1-8.
Predictors of Unplanned Postoperative Visits in a Veterans Affairs Hand Surgery Practice
Predictors of Unplanned Postoperative Visits in a Veterans Affairs Hand Surgery Practice
Is Ken Kizer’s Legacy at Risk? The Future of the Veterans Health Administration
Is Ken Kizer’s Legacy at Risk? The Future of the Veterans Health Administration
Mostly it is loss that teaches us about the worth of things.
Arthur Schopenhauer1
One of the leaders I most respected in my US Department of Veterans Affairs (VA) career was a man who had worked his way up through the ranks to become a medical center director. Usually calm, cool, and collected, he would wax poetic when discussing the hero of the VA Health Care System revolution, Ken Kizer, MD, MPH.
In this issue of Federal Practitioner, journalist Randy Dotinga interviews Kizer about the current challenges facing the VA. Though many readers will have participated in or at least know about Kizer’s unprecedented overhaul of the agency, many others, especially those new to VA, may not. It seemed a fitting time to offer an outline of the immense and positive accomplishments that occurred in the agency during his tenure, especially as, under the current administration, many of his most forward-thinking initiatives seem to be moving backward.2
When President Clinton nominated Kizer to serve as the Under Secretary for Health for the Veterans Health Administration in 1994, the poor quality care the agency delivered was castigated in popular movies like Born on the Fourth of July. Veterans who were seen in that era, and who eventually returned to a far better, kinder VA thanks to Kizer, would often tell me, “Doc, the VA was really bad then, and I was afraid to come back.” The critique of VA health care in the mid-1990s sounds like a bureaucratic déjà vu of many of the concerns Kizer raised in his interview, including fragmentation of care, access barriers, and poor coordination of treatment.3
If anyone was prepared and qualified to take on this seeming mission impossible, it was Kizer. A US Navy veteran with 6 board certifications, he came to the VA following a brave and innovative stint as the top health official in California, where he successfully took on the tobacco lobby and dramatically reduced the state’s rates of smoking and related diseases.4
Long before it was the subject of reality TV shows, Kizer dubbed his major renovation of the VA’s antiquated structure an “extreme makeover.”3 Though this description is an oversimplification of Kizer’s monumental efforts, the makeover can be considered in 4 to 6 buckets, depending on how various health policy experts parse the re-engineering efforts.5-7
Decentralization. Kizer instituted the Veterans Integrated Service Network (VISN) system to coordinate the management and operations of all the hospitals, clinics, and other VA health care entities in what is roughly a region. The locus of decision-making shifted from the VA Central Office to the VISNs, intended to promote more efficient, economical, and streamlined health care delivery.
Capitation. Accompanying this restructuring was a shift to a capitated system focused on preventive care. The Veterans Equitable Resource Allocation system was designed to logically link workload and funding. This was a major shift away from VA’s previous emphasis on inpatient and specialty care and resulted in the closing of multiple hospitals.4
Information Systems. I can still remember the first time I sat down at a prehistoric computer to use the Computerized Patient Record System (CPRS). Though now much maligned, then it was like something out of Star Trek, at a time when almost every other health care institution was buried in paper charts. With CPRS, VA suddenly had a pioneering and much-envied electronic medical record that facilitated continuity of care, communication between professionals, and accuracy and completeness of documentation.
Data Driven Performance Improvement. The VISNs and information systems inaugurated a new era of data-driven quality improvement. The assembly and analysis of data enabled VISNs to have real-time input about comparative facility performance.
Performance Measures. The data enabled evidence-based performance measures to be developed and monitored. Though these have now become the bane of many Federal Practitioner readers’ existence, they were originally intended for VISN directors and members of the senior executive service at VA central office. These were tied to incentives that, though recently the subject of watchdog investigation, were intended to motivate and reward high-quality care.6
Even this cursory look at Kizer’s accomplishments is more than enough to demonstrate the magnitude of the makeover, and when the time frame of the achievements is factored in, the transformation is the equivalent of a planet changing its orbit at light speed. Rhetoric aside, there are now hundreds of research articles published in top medical and health policy journals, many of them authored by Kizer,7,8 that have amply demonstrated that when he departed the VA in 1999, it had become “the best care anywhere.” 9 For example, a 2000 New England Journal of Medicine article found that from 1994 to 2000, the percentage of veterans whose care met ≥ 90% of 9 of 17 quality standards was > 70% for 13 of the measures, outperforming fee-for-service Medicare.10
There had been uncertainty about whether Kizer would seek a second term as Under Secretary when he announced that he was leaving. With concise modesty, Kizer said he had met his charge to, “re-engineer the veterans’ health care system so that it could effectively function in the 21st century.”11
Despite openly and critically discussing the many difficulties the VA currently confronts, Kizer ends his interview on a note of hope. Since he likely knows more about VA than any person alive, we need to trust his judgment that his legacy, which currently seems more in jeopardy than ever before, will somehow prevail. Perhaps I am too melancholic, but I believe it will take a professional of the stature of Dr. Kizer to take us back to that future, and I fear we will not see his likes again.
- Schopenhaur A, translated by Saunders TB. Parerga and Paralipomena: A Collection of Philosophical Essays. Cosimo Classics: 2007.
- Spotswood, S. Massive VA restructuring would cut number of VISNS, reduce high-level leadership. U.S. Medicine. January 15, 2026. Accessed March 23, 2026. https://www .usmedicine.com/non-clinical-topics/policy/massive -va-restructuring-would-cut-number-of-visns-reduce-high -level-leadership/
- Kizer KW, Dudley RA. Extreme makeover: transformation of the veterans health ca re system. Annu Rev Public Health. 2009;30:313-339. doi:10.1146/annurev.publhealth.29.020907.090940
- Payne D. How Kizer healed the VA. BMJ. 2012;344:e3324. doi:10.1136/bmj.e3324
- Jha AK. What can the rest of the health care system learn from VA’s quality and safety transformation? Agency for Healthcare Research and Quality. Patient Safety Network. September 1, 2006. Accessed March 23, 2026. https://psnet .ahrq.gov/perspective/what-can-rest-health-care-system -learn-vas-quality-and-safety-transformation
- US Department of Veterans Affairs, Office of Inspector General. VA improperly awarded 10.8 million in incentives to central office senior executives. Report # 23-03773-169. May 9, 2024. Accessed March 23, 2026. https://www.vaoig.gov /reports/administrative-investigation/va-improperly-awarded -108-million-incentives-central-office
- Kizer KW. The “new VA”: a national laboratory for health care quality management. Am J Med Qual. 1999;14:3-20. doi:10.1177/106286069901400103
- Kizer KW, Pane GA. The “new VA”: delivering health care value through integrated service networks. Ann Emerg Med. 1997;30:804-807. doi:10.1016/s0196-0644(97)70053-2
- Longman P. Best Care Anywhere: Why VA Health Care is Better than Yours. 3rd ed. Berrett-Koehler Publishers; 2012.
- Jha AK, Perlin JB, Kizer KW, Dudley RA. Effect of the transformation of the Veterans Affairs Health Care System on the quality of care. N Engl J Med. 2003;348:2218-2227. doi:10.1056/NEJMsa021899
- US Department of Veterans Affairs. Kizer withdraws name from consideration for second term as VA Under Secretary for Health. Vanguard. June/July 1999. Accessed March 23, 2026. https://www.va.gov/opa/publications/archives /vanguard/99junjulvg.pdf
Mostly it is loss that teaches us about the worth of things.
Arthur Schopenhauer1
One of the leaders I most respected in my US Department of Veterans Affairs (VA) career was a man who had worked his way up through the ranks to become a medical center director. Usually calm, cool, and collected, he would wax poetic when discussing the hero of the VA Health Care System revolution, Ken Kizer, MD, MPH.
In this issue of Federal Practitioner, journalist Randy Dotinga interviews Kizer about the current challenges facing the VA. Though many readers will have participated in or at least know about Kizer’s unprecedented overhaul of the agency, many others, especially those new to VA, may not. It seemed a fitting time to offer an outline of the immense and positive accomplishments that occurred in the agency during his tenure, especially as, under the current administration, many of his most forward-thinking initiatives seem to be moving backward.2
When President Clinton nominated Kizer to serve as the Under Secretary for Health for the Veterans Health Administration in 1994, the poor quality care the agency delivered was castigated in popular movies like Born on the Fourth of July. Veterans who were seen in that era, and who eventually returned to a far better, kinder VA thanks to Kizer, would often tell me, “Doc, the VA was really bad then, and I was afraid to come back.” The critique of VA health care in the mid-1990s sounds like a bureaucratic déjà vu of many of the concerns Kizer raised in his interview, including fragmentation of care, access barriers, and poor coordination of treatment.3
If anyone was prepared and qualified to take on this seeming mission impossible, it was Kizer. A US Navy veteran with 6 board certifications, he came to the VA following a brave and innovative stint as the top health official in California, where he successfully took on the tobacco lobby and dramatically reduced the state’s rates of smoking and related diseases.4
Long before it was the subject of reality TV shows, Kizer dubbed his major renovation of the VA’s antiquated structure an “extreme makeover.”3 Though this description is an oversimplification of Kizer’s monumental efforts, the makeover can be considered in 4 to 6 buckets, depending on how various health policy experts parse the re-engineering efforts.5-7
Decentralization. Kizer instituted the Veterans Integrated Service Network (VISN) system to coordinate the management and operations of all the hospitals, clinics, and other VA health care entities in what is roughly a region. The locus of decision-making shifted from the VA Central Office to the VISNs, intended to promote more efficient, economical, and streamlined health care delivery.
Capitation. Accompanying this restructuring was a shift to a capitated system focused on preventive care. The Veterans Equitable Resource Allocation system was designed to logically link workload and funding. This was a major shift away from VA’s previous emphasis on inpatient and specialty care and resulted in the closing of multiple hospitals.4
Information Systems. I can still remember the first time I sat down at a prehistoric computer to use the Computerized Patient Record System (CPRS). Though now much maligned, then it was like something out of Star Trek, at a time when almost every other health care institution was buried in paper charts. With CPRS, VA suddenly had a pioneering and much-envied electronic medical record that facilitated continuity of care, communication between professionals, and accuracy and completeness of documentation.
Data Driven Performance Improvement. The VISNs and information systems inaugurated a new era of data-driven quality improvement. The assembly and analysis of data enabled VISNs to have real-time input about comparative facility performance.
Performance Measures. The data enabled evidence-based performance measures to be developed and monitored. Though these have now become the bane of many Federal Practitioner readers’ existence, they were originally intended for VISN directors and members of the senior executive service at VA central office. These were tied to incentives that, though recently the subject of watchdog investigation, were intended to motivate and reward high-quality care.6
Even this cursory look at Kizer’s accomplishments is more than enough to demonstrate the magnitude of the makeover, and when the time frame of the achievements is factored in, the transformation is the equivalent of a planet changing its orbit at light speed. Rhetoric aside, there are now hundreds of research articles published in top medical and health policy journals, many of them authored by Kizer,7,8 that have amply demonstrated that when he departed the VA in 1999, it had become “the best care anywhere.” 9 For example, a 2000 New England Journal of Medicine article found that from 1994 to 2000, the percentage of veterans whose care met ≥ 90% of 9 of 17 quality standards was > 70% for 13 of the measures, outperforming fee-for-service Medicare.10
There had been uncertainty about whether Kizer would seek a second term as Under Secretary when he announced that he was leaving. With concise modesty, Kizer said he had met his charge to, “re-engineer the veterans’ health care system so that it could effectively function in the 21st century.”11
Despite openly and critically discussing the many difficulties the VA currently confronts, Kizer ends his interview on a note of hope. Since he likely knows more about VA than any person alive, we need to trust his judgment that his legacy, which currently seems more in jeopardy than ever before, will somehow prevail. Perhaps I am too melancholic, but I believe it will take a professional of the stature of Dr. Kizer to take us back to that future, and I fear we will not see his likes again.
Mostly it is loss that teaches us about the worth of things.
Arthur Schopenhauer1
One of the leaders I most respected in my US Department of Veterans Affairs (VA) career was a man who had worked his way up through the ranks to become a medical center director. Usually calm, cool, and collected, he would wax poetic when discussing the hero of the VA Health Care System revolution, Ken Kizer, MD, MPH.
In this issue of Federal Practitioner, journalist Randy Dotinga interviews Kizer about the current challenges facing the VA. Though many readers will have participated in or at least know about Kizer’s unprecedented overhaul of the agency, many others, especially those new to VA, may not. It seemed a fitting time to offer an outline of the immense and positive accomplishments that occurred in the agency during his tenure, especially as, under the current administration, many of his most forward-thinking initiatives seem to be moving backward.2
When President Clinton nominated Kizer to serve as the Under Secretary for Health for the Veterans Health Administration in 1994, the poor quality care the agency delivered was castigated in popular movies like Born on the Fourth of July. Veterans who were seen in that era, and who eventually returned to a far better, kinder VA thanks to Kizer, would often tell me, “Doc, the VA was really bad then, and I was afraid to come back.” The critique of VA health care in the mid-1990s sounds like a bureaucratic déjà vu of many of the concerns Kizer raised in his interview, including fragmentation of care, access barriers, and poor coordination of treatment.3
If anyone was prepared and qualified to take on this seeming mission impossible, it was Kizer. A US Navy veteran with 6 board certifications, he came to the VA following a brave and innovative stint as the top health official in California, where he successfully took on the tobacco lobby and dramatically reduced the state’s rates of smoking and related diseases.4
Long before it was the subject of reality TV shows, Kizer dubbed his major renovation of the VA’s antiquated structure an “extreme makeover.”3 Though this description is an oversimplification of Kizer’s monumental efforts, the makeover can be considered in 4 to 6 buckets, depending on how various health policy experts parse the re-engineering efforts.5-7
Decentralization. Kizer instituted the Veterans Integrated Service Network (VISN) system to coordinate the management and operations of all the hospitals, clinics, and other VA health care entities in what is roughly a region. The locus of decision-making shifted from the VA Central Office to the VISNs, intended to promote more efficient, economical, and streamlined health care delivery.
Capitation. Accompanying this restructuring was a shift to a capitated system focused on preventive care. The Veterans Equitable Resource Allocation system was designed to logically link workload and funding. This was a major shift away from VA’s previous emphasis on inpatient and specialty care and resulted in the closing of multiple hospitals.4
Information Systems. I can still remember the first time I sat down at a prehistoric computer to use the Computerized Patient Record System (CPRS). Though now much maligned, then it was like something out of Star Trek, at a time when almost every other health care institution was buried in paper charts. With CPRS, VA suddenly had a pioneering and much-envied electronic medical record that facilitated continuity of care, communication between professionals, and accuracy and completeness of documentation.
Data Driven Performance Improvement. The VISNs and information systems inaugurated a new era of data-driven quality improvement. The assembly and analysis of data enabled VISNs to have real-time input about comparative facility performance.
Performance Measures. The data enabled evidence-based performance measures to be developed and monitored. Though these have now become the bane of many Federal Practitioner readers’ existence, they were originally intended for VISN directors and members of the senior executive service at VA central office. These were tied to incentives that, though recently the subject of watchdog investigation, were intended to motivate and reward high-quality care.6
Even this cursory look at Kizer’s accomplishments is more than enough to demonstrate the magnitude of the makeover, and when the time frame of the achievements is factored in, the transformation is the equivalent of a planet changing its orbit at light speed. Rhetoric aside, there are now hundreds of research articles published in top medical and health policy journals, many of them authored by Kizer,7,8 that have amply demonstrated that when he departed the VA in 1999, it had become “the best care anywhere.” 9 For example, a 2000 New England Journal of Medicine article found that from 1994 to 2000, the percentage of veterans whose care met ≥ 90% of 9 of 17 quality standards was > 70% for 13 of the measures, outperforming fee-for-service Medicare.10
There had been uncertainty about whether Kizer would seek a second term as Under Secretary when he announced that he was leaving. With concise modesty, Kizer said he had met his charge to, “re-engineer the veterans’ health care system so that it could effectively function in the 21st century.”11
Despite openly and critically discussing the many difficulties the VA currently confronts, Kizer ends his interview on a note of hope. Since he likely knows more about VA than any person alive, we need to trust his judgment that his legacy, which currently seems more in jeopardy than ever before, will somehow prevail. Perhaps I am too melancholic, but I believe it will take a professional of the stature of Dr. Kizer to take us back to that future, and I fear we will not see his likes again.
- Schopenhaur A, translated by Saunders TB. Parerga and Paralipomena: A Collection of Philosophical Essays. Cosimo Classics: 2007.
- Spotswood, S. Massive VA restructuring would cut number of VISNS, reduce high-level leadership. U.S. Medicine. January 15, 2026. Accessed March 23, 2026. https://www .usmedicine.com/non-clinical-topics/policy/massive -va-restructuring-would-cut-number-of-visns-reduce-high -level-leadership/
- Kizer KW, Dudley RA. Extreme makeover: transformation of the veterans health ca re system. Annu Rev Public Health. 2009;30:313-339. doi:10.1146/annurev.publhealth.29.020907.090940
- Payne D. How Kizer healed the VA. BMJ. 2012;344:e3324. doi:10.1136/bmj.e3324
- Jha AK. What can the rest of the health care system learn from VA’s quality and safety transformation? Agency for Healthcare Research and Quality. Patient Safety Network. September 1, 2006. Accessed March 23, 2026. https://psnet .ahrq.gov/perspective/what-can-rest-health-care-system -learn-vas-quality-and-safety-transformation
- US Department of Veterans Affairs, Office of Inspector General. VA improperly awarded 10.8 million in incentives to central office senior executives. Report # 23-03773-169. May 9, 2024. Accessed March 23, 2026. https://www.vaoig.gov /reports/administrative-investigation/va-improperly-awarded -108-million-incentives-central-office
- Kizer KW. The “new VA”: a national laboratory for health care quality management. Am J Med Qual. 1999;14:3-20. doi:10.1177/106286069901400103
- Kizer KW, Pane GA. The “new VA”: delivering health care value through integrated service networks. Ann Emerg Med. 1997;30:804-807. doi:10.1016/s0196-0644(97)70053-2
- Longman P. Best Care Anywhere: Why VA Health Care is Better than Yours. 3rd ed. Berrett-Koehler Publishers; 2012.
- Jha AK, Perlin JB, Kizer KW, Dudley RA. Effect of the transformation of the Veterans Affairs Health Care System on the quality of care. N Engl J Med. 2003;348:2218-2227. doi:10.1056/NEJMsa021899
- US Department of Veterans Affairs. Kizer withdraws name from consideration for second term as VA Under Secretary for Health. Vanguard. June/July 1999. Accessed March 23, 2026. https://www.va.gov/opa/publications/archives /vanguard/99junjulvg.pdf
- Schopenhaur A, translated by Saunders TB. Parerga and Paralipomena: A Collection of Philosophical Essays. Cosimo Classics: 2007.
- Spotswood, S. Massive VA restructuring would cut number of VISNS, reduce high-level leadership. U.S. Medicine. January 15, 2026. Accessed March 23, 2026. https://www .usmedicine.com/non-clinical-topics/policy/massive -va-restructuring-would-cut-number-of-visns-reduce-high -level-leadership/
- Kizer KW, Dudley RA. Extreme makeover: transformation of the veterans health ca re system. Annu Rev Public Health. 2009;30:313-339. doi:10.1146/annurev.publhealth.29.020907.090940
- Payne D. How Kizer healed the VA. BMJ. 2012;344:e3324. doi:10.1136/bmj.e3324
- Jha AK. What can the rest of the health care system learn from VA’s quality and safety transformation? Agency for Healthcare Research and Quality. Patient Safety Network. September 1, 2006. Accessed March 23, 2026. https://psnet .ahrq.gov/perspective/what-can-rest-health-care-system -learn-vas-quality-and-safety-transformation
- US Department of Veterans Affairs, Office of Inspector General. VA improperly awarded 10.8 million in incentives to central office senior executives. Report # 23-03773-169. May 9, 2024. Accessed March 23, 2026. https://www.vaoig.gov /reports/administrative-investigation/va-improperly-awarded -108-million-incentives-central-office
- Kizer KW. The “new VA”: a national laboratory for health care quality management. Am J Med Qual. 1999;14:3-20. doi:10.1177/106286069901400103
- Kizer KW, Pane GA. The “new VA”: delivering health care value through integrated service networks. Ann Emerg Med. 1997;30:804-807. doi:10.1016/s0196-0644(97)70053-2
- Longman P. Best Care Anywhere: Why VA Health Care is Better than Yours. 3rd ed. Berrett-Koehler Publishers; 2012.
- Jha AK, Perlin JB, Kizer KW, Dudley RA. Effect of the transformation of the Veterans Affairs Health Care System on the quality of care. N Engl J Med. 2003;348:2218-2227. doi:10.1056/NEJMsa021899
- US Department of Veterans Affairs. Kizer withdraws name from consideration for second term as VA Under Secretary for Health. Vanguard. June/July 1999. Accessed March 23, 2026. https://www.va.gov/opa/publications/archives /vanguard/99junjulvg.pdf
Is Ken Kizer’s Legacy at Risk? The Future of the Veterans Health Administration
Is Ken Kizer’s Legacy at Risk? The Future of the Veterans Health Administration
Asymptomatic Plaque and Nodule on the Nose
Asymptomatic Plaque and Nodule on the Nose
THE DIAGNOSIS: Coexisting Squamous Cell Carcinoma and Basal Cell Carcinoma
Dermoscopy of the plaque showed central ulceration with blood spots surrounded by branched linear vessels, which was suggestive of squamous cell carcinoma (SCC)(Figure 1A). The nodule showed shiny, white-red, structureless areas with small gray spots, bright white crystalline streaks, and short fine telangiectasias suggestive of basal cell carcinoma (BCC)(Figure 1B). Histopathology showed that the plaque had irregular nests, cords, and sheets of neoplastic keratinocytes invading the dermis (Figure 2A) and the nodule had discrete nests of basaloid cells with peripheral palisading in the dermis (Figure 2B), which confirmed the diagnosis of coexisting SCC and BCC. The patient underwent surgical excision of the lesions, which achieved clear margins. At the 2-year follow-up, there was no sign of recurrence.
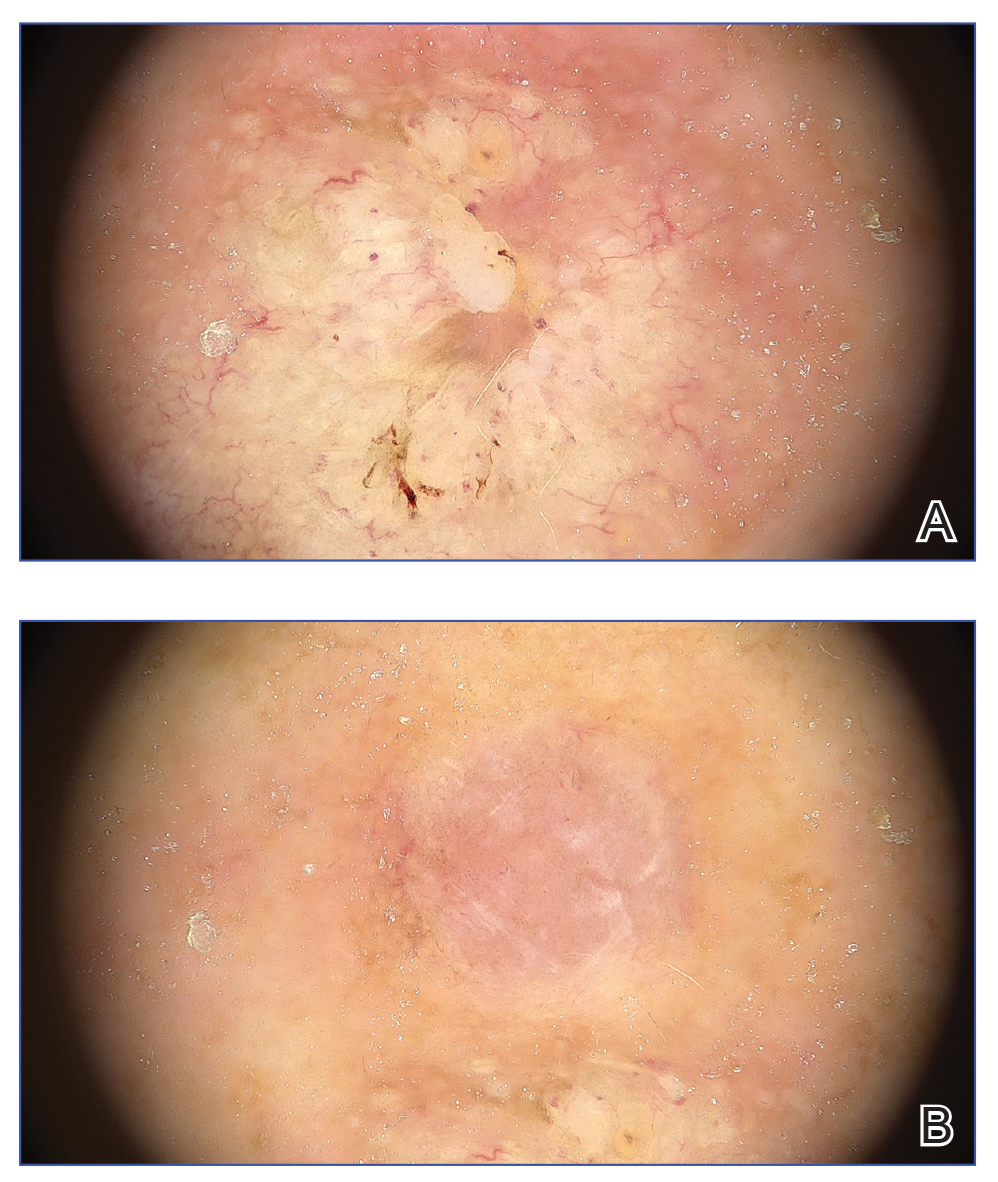
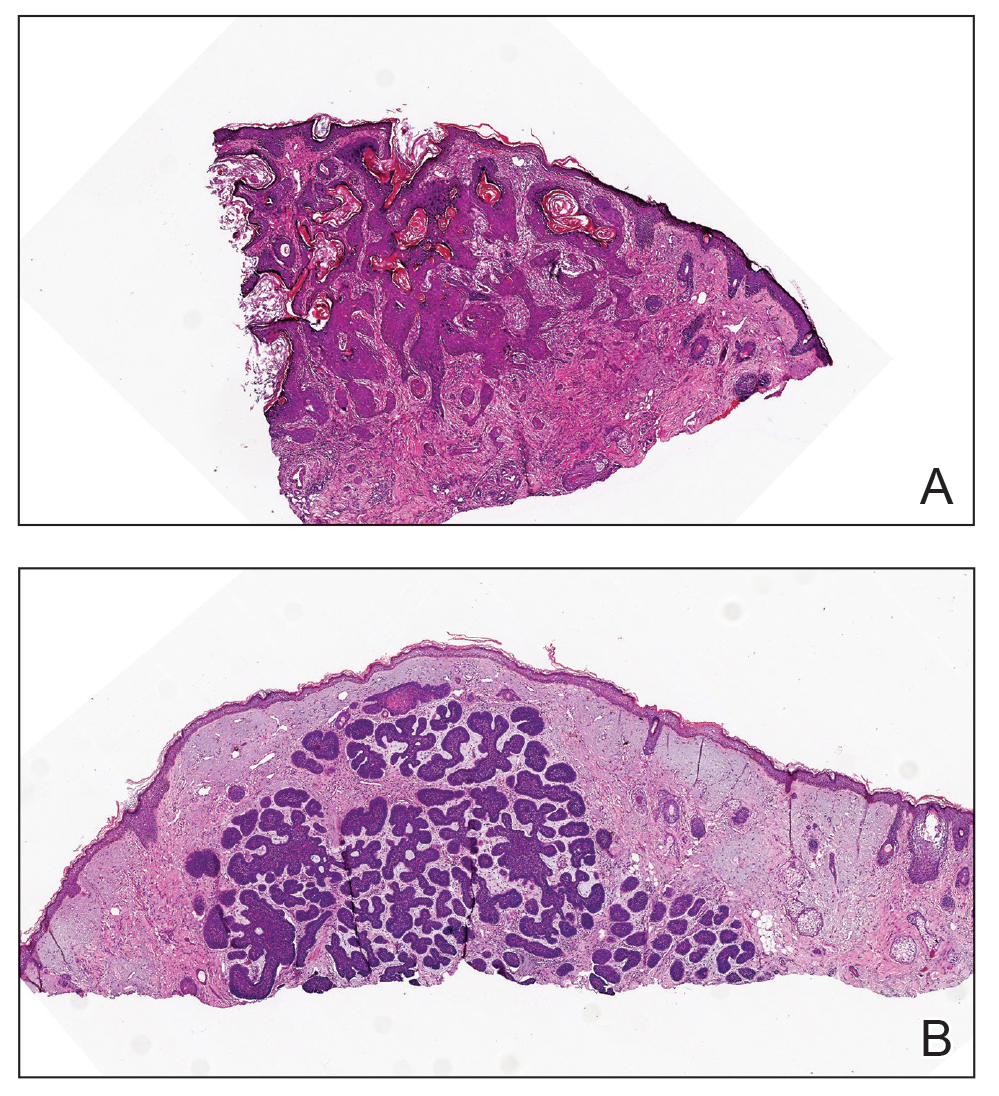
Squamous cell carcinoma is the second most frequent cancer in humans. Older patients are more susceptible due to chronic UV exposure.1 Basal cell carcinoma is the most common human cancer worldwide.2 These skin cancers have different clinical manifestations, pathologic features, treatment methods, and prognoses. The coexistence of 2 types of skin cancer presents a diagnostic challenge. Possible causes of this phenomenon are not clear. It may simply be a coincidence since the lesions typically occur in sun-exposed areas such as the nose, which may be affected by photodamage.3 According to the field cancerization theory, chronically sun-exposed areas are at higher risk for development of coexisting skin cancers.4 A more interesting explanation is the interaction theory, which suggests that one tumor produces epidermal or stromal changes that induce the formation of a second independent tumor via the paracrine effect (ie, growth mediators from nearby cells).4
Dermoscopy is an important noninvasive diagnostic tool for the evaluation of skin cancer, particularly early detection. Dermoscopic findings of blood vessels, ulcers, the fiber sign, blood spots, white structureless areas, keratin, and centered vessels indicate a diagnosis of SCC.5 In contrast, common dermoscopic findings for BCC include arborizing vessels, ulceration, shiny white structures, and blue-gray ovoid nests or globules.6
Irritated seborrheic keratosis is an inflammatory variant of seborrheic keratosis, which often is challenging to identify clinically due to its similar features with SCC; however, SCC is more likely to demonstrate dotted or branched vessels, white structureless areas, white circles around follicles, irregular or peripheral vessel patterns, and central scales on dermoscopy. In contrast, irritated seborrheic keratosis is more likely to have hairpin vessels, regular vessel patterns, and white halos around vessels, which may aid in the differentiation between the two entities.6
Due to the higher sensitivity of dermoscopy for detecting pigmented BCC compared to nonpigmented BCC, it holds substantial diagnostic value in Asian populations, in whom pigmented BCC is the most common subtype.6,8 However, the lack of pigmentation in the nodule in our case posed a diagnostic challenge, as the diagnosis of BCC had to rely on subtle vascular and shiny white structures rather than more obvious pigment clues. This absence of pigment, however, also helped rule out pigmented BCC as a diagnosis for the nodule. Short fine telangiectasias is the second most common vascular pattern in BCC, and bright white structures are highly suggestive of nonpigmented BCC.6 Therefore, dermoscopic findings of bright-white structures with fine telangiectasias should be alerted to the possibility of nonpigmented BCC.
Basosquamous carcinoma has clinical and dermoscopic features between SCC and BCC, and the presence of dermatoscopic features from both BCC and SCC should raise suspicion, but the diagnosis is particularly challenging because its presentation is nonspecific.9 We need to be vigilant about the possibility of coexistence of 2 types of skin cancer, and that regular physical examination and dermatoscopy are very important for early detection and diagnosis.
- Corchado-Cobos R, García-Sancha N, González-Sarmiento R, et al. Cutaneous squamous cell carcinoma: from biology to therapy. Int J Mol Sci. 2020;21:2956. doi:10.3390/ijms21082956
- Cameron MC, Lee E, Hibler BP, et al. Basal cell carcinoma: epidemiology; pathophysiology; clinical and histological subtypes; and disease associations. J Am Acad Dermatol. 2019;80:303-317. doi:10.1016/j.jaad.2018.03.060
- Kraemer KH, Lee MM, Scotto J. Xeroderma pigmentosum. Cutaneous, ocular, and neurologic abnormalities in 830 published cases. Arch Dermatol. 1987;123:241-250. doi:10.1001/archderm.123.2.241
- Cornejo KM, Deng AC. Malignant melanoma within squamous cell carcinoma and basal cell carcinoma: is it a combined or collision tumor? a case report and review of the literature. Am J Dermatopathol. 2013;35:226-34. doi:10.1097/DAD.0b013e3182545e27
- Ertop Dog˘an P, Akay BN, Okçu Heper A, et al. Dermatoscopic findings and dermatopathological correlates in clinical variants of actinic keratosis, Bowen’s disease, keratoacanthoma, and squamous cell carcinoma. Dermatol Ther. 2021;34:E14877. doi:10.1111/dth.14877.
- Álvarez-Salafranca M, Ara M, Zaballos P. Dermoscopy in basal cell carcinoma: an updated review. Actas Dermosifiliogr (Engl Ed). 2021;112:330-338. doi:10.1016/j.ad.2020.11.011
- Papageorgiou C, Spyridis I, Manoli SM, et al. Accuracy of dermoscopic criteria for the differential diagnosis between irritated seborrheic keratosis and squamous cell carcinoma. J Am Acad Dermatol. 2021;85:1143-1150. doi:10.1016/j.jaad.2020.02.019
- Cheng SY, Luk NM, Chong LY. Special features of non-melanoma skin cancer in Hong Kong Chinese patients: 10-year retrospective study. Hong Kong Med J. 2001;7:22-28.
- Murgia G, Denaro N, Boggio F, et al. Basosquamous carcinoma: comprehensive clinical and histopathological aspects, novel imaging tools, and therapeutic approaches. Cells. 2023;23:2737. doi:10.3390/cells12232737
THE DIAGNOSIS: Coexisting Squamous Cell Carcinoma and Basal Cell Carcinoma
Dermoscopy of the plaque showed central ulceration with blood spots surrounded by branched linear vessels, which was suggestive of squamous cell carcinoma (SCC)(Figure 1A). The nodule showed shiny, white-red, structureless areas with small gray spots, bright white crystalline streaks, and short fine telangiectasias suggestive of basal cell carcinoma (BCC)(Figure 1B). Histopathology showed that the plaque had irregular nests, cords, and sheets of neoplastic keratinocytes invading the dermis (Figure 2A) and the nodule had discrete nests of basaloid cells with peripheral palisading in the dermis (Figure 2B), which confirmed the diagnosis of coexisting SCC and BCC. The patient underwent surgical excision of the lesions, which achieved clear margins. At the 2-year follow-up, there was no sign of recurrence.


Squamous cell carcinoma is the second most frequent cancer in humans. Older patients are more susceptible due to chronic UV exposure.1 Basal cell carcinoma is the most common human cancer worldwide.2 These skin cancers have different clinical manifestations, pathologic features, treatment methods, and prognoses. The coexistence of 2 types of skin cancer presents a diagnostic challenge. Possible causes of this phenomenon are not clear. It may simply be a coincidence since the lesions typically occur in sun-exposed areas such as the nose, which may be affected by photodamage.3 According to the field cancerization theory, chronically sun-exposed areas are at higher risk for development of coexisting skin cancers.4 A more interesting explanation is the interaction theory, which suggests that one tumor produces epidermal or stromal changes that induce the formation of a second independent tumor via the paracrine effect (ie, growth mediators from nearby cells).4
Dermoscopy is an important noninvasive diagnostic tool for the evaluation of skin cancer, particularly early detection. Dermoscopic findings of blood vessels, ulcers, the fiber sign, blood spots, white structureless areas, keratin, and centered vessels indicate a diagnosis of SCC.5 In contrast, common dermoscopic findings for BCC include arborizing vessels, ulceration, shiny white structures, and blue-gray ovoid nests or globules.6
Irritated seborrheic keratosis is an inflammatory variant of seborrheic keratosis, which often is challenging to identify clinically due to its similar features with SCC; however, SCC is more likely to demonstrate dotted or branched vessels, white structureless areas, white circles around follicles, irregular or peripheral vessel patterns, and central scales on dermoscopy. In contrast, irritated seborrheic keratosis is more likely to have hairpin vessels, regular vessel patterns, and white halos around vessels, which may aid in the differentiation between the two entities.6
Due to the higher sensitivity of dermoscopy for detecting pigmented BCC compared to nonpigmented BCC, it holds substantial diagnostic value in Asian populations, in whom pigmented BCC is the most common subtype.6,8 However, the lack of pigmentation in the nodule in our case posed a diagnostic challenge, as the diagnosis of BCC had to rely on subtle vascular and shiny white structures rather than more obvious pigment clues. This absence of pigment, however, also helped rule out pigmented BCC as a diagnosis for the nodule. Short fine telangiectasias is the second most common vascular pattern in BCC, and bright white structures are highly suggestive of nonpigmented BCC.6 Therefore, dermoscopic findings of bright-white structures with fine telangiectasias should be alerted to the possibility of nonpigmented BCC.
Basosquamous carcinoma has clinical and dermoscopic features between SCC and BCC, and the presence of dermatoscopic features from both BCC and SCC should raise suspicion, but the diagnosis is particularly challenging because its presentation is nonspecific.9 We need to be vigilant about the possibility of coexistence of 2 types of skin cancer, and that regular physical examination and dermatoscopy are very important for early detection and diagnosis.
THE DIAGNOSIS: Coexisting Squamous Cell Carcinoma and Basal Cell Carcinoma
Dermoscopy of the plaque showed central ulceration with blood spots surrounded by branched linear vessels, which was suggestive of squamous cell carcinoma (SCC)(Figure 1A). The nodule showed shiny, white-red, structureless areas with small gray spots, bright white crystalline streaks, and short fine telangiectasias suggestive of basal cell carcinoma (BCC)(Figure 1B). Histopathology showed that the plaque had irregular nests, cords, and sheets of neoplastic keratinocytes invading the dermis (Figure 2A) and the nodule had discrete nests of basaloid cells with peripheral palisading in the dermis (Figure 2B), which confirmed the diagnosis of coexisting SCC and BCC. The patient underwent surgical excision of the lesions, which achieved clear margins. At the 2-year follow-up, there was no sign of recurrence.


Squamous cell carcinoma is the second most frequent cancer in humans. Older patients are more susceptible due to chronic UV exposure.1 Basal cell carcinoma is the most common human cancer worldwide.2 These skin cancers have different clinical manifestations, pathologic features, treatment methods, and prognoses. The coexistence of 2 types of skin cancer presents a diagnostic challenge. Possible causes of this phenomenon are not clear. It may simply be a coincidence since the lesions typically occur in sun-exposed areas such as the nose, which may be affected by photodamage.3 According to the field cancerization theory, chronically sun-exposed areas are at higher risk for development of coexisting skin cancers.4 A more interesting explanation is the interaction theory, which suggests that one tumor produces epidermal or stromal changes that induce the formation of a second independent tumor via the paracrine effect (ie, growth mediators from nearby cells).4
Dermoscopy is an important noninvasive diagnostic tool for the evaluation of skin cancer, particularly early detection. Dermoscopic findings of blood vessels, ulcers, the fiber sign, blood spots, white structureless areas, keratin, and centered vessels indicate a diagnosis of SCC.5 In contrast, common dermoscopic findings for BCC include arborizing vessels, ulceration, shiny white structures, and blue-gray ovoid nests or globules.6
Irritated seborrheic keratosis is an inflammatory variant of seborrheic keratosis, which often is challenging to identify clinically due to its similar features with SCC; however, SCC is more likely to demonstrate dotted or branched vessels, white structureless areas, white circles around follicles, irregular or peripheral vessel patterns, and central scales on dermoscopy. In contrast, irritated seborrheic keratosis is more likely to have hairpin vessels, regular vessel patterns, and white halos around vessels, which may aid in the differentiation between the two entities.6
Due to the higher sensitivity of dermoscopy for detecting pigmented BCC compared to nonpigmented BCC, it holds substantial diagnostic value in Asian populations, in whom pigmented BCC is the most common subtype.6,8 However, the lack of pigmentation in the nodule in our case posed a diagnostic challenge, as the diagnosis of BCC had to rely on subtle vascular and shiny white structures rather than more obvious pigment clues. This absence of pigment, however, also helped rule out pigmented BCC as a diagnosis for the nodule. Short fine telangiectasias is the second most common vascular pattern in BCC, and bright white structures are highly suggestive of nonpigmented BCC.6 Therefore, dermoscopic findings of bright-white structures with fine telangiectasias should be alerted to the possibility of nonpigmented BCC.
Basosquamous carcinoma has clinical and dermoscopic features between SCC and BCC, and the presence of dermatoscopic features from both BCC and SCC should raise suspicion, but the diagnosis is particularly challenging because its presentation is nonspecific.9 We need to be vigilant about the possibility of coexistence of 2 types of skin cancer, and that regular physical examination and dermatoscopy are very important for early detection and diagnosis.
- Corchado-Cobos R, García-Sancha N, González-Sarmiento R, et al. Cutaneous squamous cell carcinoma: from biology to therapy. Int J Mol Sci. 2020;21:2956. doi:10.3390/ijms21082956
- Cameron MC, Lee E, Hibler BP, et al. Basal cell carcinoma: epidemiology; pathophysiology; clinical and histological subtypes; and disease associations. J Am Acad Dermatol. 2019;80:303-317. doi:10.1016/j.jaad.2018.03.060
- Kraemer KH, Lee MM, Scotto J. Xeroderma pigmentosum. Cutaneous, ocular, and neurologic abnormalities in 830 published cases. Arch Dermatol. 1987;123:241-250. doi:10.1001/archderm.123.2.241
- Cornejo KM, Deng AC. Malignant melanoma within squamous cell carcinoma and basal cell carcinoma: is it a combined or collision tumor? a case report and review of the literature. Am J Dermatopathol. 2013;35:226-34. doi:10.1097/DAD.0b013e3182545e27
- Ertop Dog˘an P, Akay BN, Okçu Heper A, et al. Dermatoscopic findings and dermatopathological correlates in clinical variants of actinic keratosis, Bowen’s disease, keratoacanthoma, and squamous cell carcinoma. Dermatol Ther. 2021;34:E14877. doi:10.1111/dth.14877.
- Álvarez-Salafranca M, Ara M, Zaballos P. Dermoscopy in basal cell carcinoma: an updated review. Actas Dermosifiliogr (Engl Ed). 2021;112:330-338. doi:10.1016/j.ad.2020.11.011
- Papageorgiou C, Spyridis I, Manoli SM, et al. Accuracy of dermoscopic criteria for the differential diagnosis between irritated seborrheic keratosis and squamous cell carcinoma. J Am Acad Dermatol. 2021;85:1143-1150. doi:10.1016/j.jaad.2020.02.019
- Cheng SY, Luk NM, Chong LY. Special features of non-melanoma skin cancer in Hong Kong Chinese patients: 10-year retrospective study. Hong Kong Med J. 2001;7:22-28.
- Murgia G, Denaro N, Boggio F, et al. Basosquamous carcinoma: comprehensive clinical and histopathological aspects, novel imaging tools, and therapeutic approaches. Cells. 2023;23:2737. doi:10.3390/cells12232737
- Corchado-Cobos R, García-Sancha N, González-Sarmiento R, et al. Cutaneous squamous cell carcinoma: from biology to therapy. Int J Mol Sci. 2020;21:2956. doi:10.3390/ijms21082956
- Cameron MC, Lee E, Hibler BP, et al. Basal cell carcinoma: epidemiology; pathophysiology; clinical and histological subtypes; and disease associations. J Am Acad Dermatol. 2019;80:303-317. doi:10.1016/j.jaad.2018.03.060
- Kraemer KH, Lee MM, Scotto J. Xeroderma pigmentosum. Cutaneous, ocular, and neurologic abnormalities in 830 published cases. Arch Dermatol. 1987;123:241-250. doi:10.1001/archderm.123.2.241
- Cornejo KM, Deng AC. Malignant melanoma within squamous cell carcinoma and basal cell carcinoma: is it a combined or collision tumor? a case report and review of the literature. Am J Dermatopathol. 2013;35:226-34. doi:10.1097/DAD.0b013e3182545e27
- Ertop Dog˘an P, Akay BN, Okçu Heper A, et al. Dermatoscopic findings and dermatopathological correlates in clinical variants of actinic keratosis, Bowen’s disease, keratoacanthoma, and squamous cell carcinoma. Dermatol Ther. 2021;34:E14877. doi:10.1111/dth.14877.
- Álvarez-Salafranca M, Ara M, Zaballos P. Dermoscopy in basal cell carcinoma: an updated review. Actas Dermosifiliogr (Engl Ed). 2021;112:330-338. doi:10.1016/j.ad.2020.11.011
- Papageorgiou C, Spyridis I, Manoli SM, et al. Accuracy of dermoscopic criteria for the differential diagnosis between irritated seborrheic keratosis and squamous cell carcinoma. J Am Acad Dermatol. 2021;85:1143-1150. doi:10.1016/j.jaad.2020.02.019
- Cheng SY, Luk NM, Chong LY. Special features of non-melanoma skin cancer in Hong Kong Chinese patients: 10-year retrospective study. Hong Kong Med J. 2001;7:22-28.
- Murgia G, Denaro N, Boggio F, et al. Basosquamous carcinoma: comprehensive clinical and histopathological aspects, novel imaging tools, and therapeutic approaches. Cells. 2023;23:2737. doi:10.3390/cells12232737
Asymptomatic Plaque and Nodule on the Nose
Asymptomatic Plaque and Nodule on the Nose
An 80-year-old Asian woman presented to the hospital dermatology clinic for evaluation of 2 lesions on the nose of 2 years’ duration. The patient reported that the lesions had initially manifested as an asymptomatic red rash, but within the past month, the affected area had grown in diameter with a rough surface and occasional bleeding. Additionally, a smaller new rash appeared around the original plaque. She had no personal or family history of skin cancer. Physical examination revealed a 1.5-cm reddish plaque on the tip of the nose with a central ulcer filled with viscous exudate. Another 0.5-cm reddish nodule with a smooth surface also was noted adjacent to the plaque. Dermoscopy and a skin biopsy of both lesions were performed.
Pseudoleukonychia of the Distal Fingernails
Pseudoleukonychia of the Distal Fingernails
THE DIAGNOSIS: Pseudopsoriatic Nails With Pterygium Inversum Unguis
Based on the clinical findings and the patient’s history of gel manicures, a diagnosis of pseudopsoriatic nails with pterygium inversum unguis (PIU) was made. The patient was advised to avoid gel manicures and any other chemical or mechanical trauma to the nails. No other treatment was administered. Improvements including healthy nail growth and disappearing color and structure changes within the nail plates were noted at 2 months’ follow-up.
The durability and availability of gel manicures has been increasingly popular due to their ideal cosmetic results. A gel manicure involves applying a gel nail polish (GNP) containing acrylate or methacrylate monomers that harden after exposure to UV light through a photopolymerization reaction. Acrylate polymers including ethylene glycol dimethacrylate, 2-hydroxyethyl acrylate, 2-hydroxyethyl methacrylate, 2-hydroxypropyl methacrylate, methyl methacrylate, and tetrahydrofurfuryl methacrylate are known to cause allergic contact dermatitis in patients who wear acrylate-based GNP.1Hydroxyethyl methacrylate is the most common sensitizer among these acrylates. Fingertip dry dermatitis, fissured painful pulpitis of the fingers, and periungual erythema are the most common manifestations of methacrylate allergy; however, there also are reports of onycholysis and onychodystrophy in patients with severe allergic contact dermatitis caused by acrylates.2,3
In contrast to common public misconception that GNP may strengthen the nails, scientific evidence has shown otherwise. Besides allergic contact dermatitis, mechanical damage and UV-induced skin manifestations have been reported in association with GNP.1,3,4 Pseudopsoriatic nails are characterized by onycholysis accompanied by subungual hyperkeratosis, closely resembling the nail findings seen in psoriasis. This condition may occur due to mechanical damage and acrylate sensitization.2,4 Pterygium inversum unguis, also known as ventral pterygium, occurs as a result of hyponychium trauma due to either application or removal processes of GNP and/ or exposure to chemical ingredients and is one of the most striking clinical manifestations of GNP use.5 In our patient, all fingernails were affected by PIU.
Patients presenting with pseudopsoriatic nail changes and/or PIU should be questioned about potential exposure to GNP and/or sculpted nails, also known as custom artificial nails or nail prostheses. Diagnosis primarily is made clinically, but microbial cultures or skin biopsy may be required to exclude psoriasis and fungal infections in some patients. Patch testing with acrylate series in particular also is necessary in patients presenting with hand dermatitis. As it is the most common contact sensitizer in the acrylic material of the GNPs, screening for 2-hydroxyethyl methacrylate allergy is recommended in these patients.1 Almost all adverse effects related to use of GNP may be reversible upon discontinuation of exposure.
- Litaiem N, Baklouti M, Zeglaoui F. Side effects of gel nail polish: a systematic review. Clin Dermatol. 2022;40:706-715. doi:10.1016 /j.clindermatol.2022.07.008
- Engelina S, Shim TN. Atypical cases of pseudo-psoriatic nails associated with acrylate contact allergy. Contact Dermatitis. 2021; 84:342-344. doi:10.1111/cod.13741
- Draelos ZD. Nail cosmetics and adornment. Dermatol Clinics. 2021;39:351-359. doi:10.1016/j.det.2021.01.001
- Rieder EA, Tosti A. Cosmetically induced disorders of the nail with update on contemporary nail manicures. J Clin Aesthet Dermatol. 2016;9:39-44.
- Cervantes J, Sanchez M, Eber AE, et al. Pterygium inversum unguis secondary to gel polish. J Eur Acad Dermatol Venereol. 2018;32:160-163. doi:10.1111/jdv.14603
THE DIAGNOSIS: Pseudopsoriatic Nails With Pterygium Inversum Unguis
Based on the clinical findings and the patient’s history of gel manicures, a diagnosis of pseudopsoriatic nails with pterygium inversum unguis (PIU) was made. The patient was advised to avoid gel manicures and any other chemical or mechanical trauma to the nails. No other treatment was administered. Improvements including healthy nail growth and disappearing color and structure changes within the nail plates were noted at 2 months’ follow-up.
The durability and availability of gel manicures has been increasingly popular due to their ideal cosmetic results. A gel manicure involves applying a gel nail polish (GNP) containing acrylate or methacrylate monomers that harden after exposure to UV light through a photopolymerization reaction. Acrylate polymers including ethylene glycol dimethacrylate, 2-hydroxyethyl acrylate, 2-hydroxyethyl methacrylate, 2-hydroxypropyl methacrylate, methyl methacrylate, and tetrahydrofurfuryl methacrylate are known to cause allergic contact dermatitis in patients who wear acrylate-based GNP.1Hydroxyethyl methacrylate is the most common sensitizer among these acrylates. Fingertip dry dermatitis, fissured painful pulpitis of the fingers, and periungual erythema are the most common manifestations of methacrylate allergy; however, there also are reports of onycholysis and onychodystrophy in patients with severe allergic contact dermatitis caused by acrylates.2,3
In contrast to common public misconception that GNP may strengthen the nails, scientific evidence has shown otherwise. Besides allergic contact dermatitis, mechanical damage and UV-induced skin manifestations have been reported in association with GNP.1,3,4 Pseudopsoriatic nails are characterized by onycholysis accompanied by subungual hyperkeratosis, closely resembling the nail findings seen in psoriasis. This condition may occur due to mechanical damage and acrylate sensitization.2,4 Pterygium inversum unguis, also known as ventral pterygium, occurs as a result of hyponychium trauma due to either application or removal processes of GNP and/ or exposure to chemical ingredients and is one of the most striking clinical manifestations of GNP use.5 In our patient, all fingernails were affected by PIU.
Patients presenting with pseudopsoriatic nail changes and/or PIU should be questioned about potential exposure to GNP and/or sculpted nails, also known as custom artificial nails or nail prostheses. Diagnosis primarily is made clinically, but microbial cultures or skin biopsy may be required to exclude psoriasis and fungal infections in some patients. Patch testing with acrylate series in particular also is necessary in patients presenting with hand dermatitis. As it is the most common contact sensitizer in the acrylic material of the GNPs, screening for 2-hydroxyethyl methacrylate allergy is recommended in these patients.1 Almost all adverse effects related to use of GNP may be reversible upon discontinuation of exposure.
THE DIAGNOSIS: Pseudopsoriatic Nails With Pterygium Inversum Unguis
Based on the clinical findings and the patient’s history of gel manicures, a diagnosis of pseudopsoriatic nails with pterygium inversum unguis (PIU) was made. The patient was advised to avoid gel manicures and any other chemical or mechanical trauma to the nails. No other treatment was administered. Improvements including healthy nail growth and disappearing color and structure changes within the nail plates were noted at 2 months’ follow-up.
The durability and availability of gel manicures has been increasingly popular due to their ideal cosmetic results. A gel manicure involves applying a gel nail polish (GNP) containing acrylate or methacrylate monomers that harden after exposure to UV light through a photopolymerization reaction. Acrylate polymers including ethylene glycol dimethacrylate, 2-hydroxyethyl acrylate, 2-hydroxyethyl methacrylate, 2-hydroxypropyl methacrylate, methyl methacrylate, and tetrahydrofurfuryl methacrylate are known to cause allergic contact dermatitis in patients who wear acrylate-based GNP.1Hydroxyethyl methacrylate is the most common sensitizer among these acrylates. Fingertip dry dermatitis, fissured painful pulpitis of the fingers, and periungual erythema are the most common manifestations of methacrylate allergy; however, there also are reports of onycholysis and onychodystrophy in patients with severe allergic contact dermatitis caused by acrylates.2,3
In contrast to common public misconception that GNP may strengthen the nails, scientific evidence has shown otherwise. Besides allergic contact dermatitis, mechanical damage and UV-induced skin manifestations have been reported in association with GNP.1,3,4 Pseudopsoriatic nails are characterized by onycholysis accompanied by subungual hyperkeratosis, closely resembling the nail findings seen in psoriasis. This condition may occur due to mechanical damage and acrylate sensitization.2,4 Pterygium inversum unguis, also known as ventral pterygium, occurs as a result of hyponychium trauma due to either application or removal processes of GNP and/ or exposure to chemical ingredients and is one of the most striking clinical manifestations of GNP use.5 In our patient, all fingernails were affected by PIU.
Patients presenting with pseudopsoriatic nail changes and/or PIU should be questioned about potential exposure to GNP and/or sculpted nails, also known as custom artificial nails or nail prostheses. Diagnosis primarily is made clinically, but microbial cultures or skin biopsy may be required to exclude psoriasis and fungal infections in some patients. Patch testing with acrylate series in particular also is necessary in patients presenting with hand dermatitis. As it is the most common contact sensitizer in the acrylic material of the GNPs, screening for 2-hydroxyethyl methacrylate allergy is recommended in these patients.1 Almost all adverse effects related to use of GNP may be reversible upon discontinuation of exposure.
- Litaiem N, Baklouti M, Zeglaoui F. Side effects of gel nail polish: a systematic review. Clin Dermatol. 2022;40:706-715. doi:10.1016 /j.clindermatol.2022.07.008
- Engelina S, Shim TN. Atypical cases of pseudo-psoriatic nails associated with acrylate contact allergy. Contact Dermatitis. 2021; 84:342-344. doi:10.1111/cod.13741
- Draelos ZD. Nail cosmetics and adornment. Dermatol Clinics. 2021;39:351-359. doi:10.1016/j.det.2021.01.001
- Rieder EA, Tosti A. Cosmetically induced disorders of the nail with update on contemporary nail manicures. J Clin Aesthet Dermatol. 2016;9:39-44.
- Cervantes J, Sanchez M, Eber AE, et al. Pterygium inversum unguis secondary to gel polish. J Eur Acad Dermatol Venereol. 2018;32:160-163. doi:10.1111/jdv.14603
- Litaiem N, Baklouti M, Zeglaoui F. Side effects of gel nail polish: a systematic review. Clin Dermatol. 2022;40:706-715. doi:10.1016 /j.clindermatol.2022.07.008
- Engelina S, Shim TN. Atypical cases of pseudo-psoriatic nails associated with acrylate contact allergy. Contact Dermatitis. 2021; 84:342-344. doi:10.1111/cod.13741
- Draelos ZD. Nail cosmetics and adornment. Dermatol Clinics. 2021;39:351-359. doi:10.1016/j.det.2021.01.001
- Rieder EA, Tosti A. Cosmetically induced disorders of the nail with update on contemporary nail manicures. J Clin Aesthet Dermatol. 2016;9:39-44.
- Cervantes J, Sanchez M, Eber AE, et al. Pterygium inversum unguis secondary to gel polish. J Eur Acad Dermatol Venereol. 2018;32:160-163. doi:10.1111/jdv.14603
Pseudoleukonychia of the Distal Fingernails
Pseudoleukonychia of the Distal Fingernails
An otherwise healthy 36-year-old woman presented to the dermatology department for evaluation of disfiguring nail changes and subungual verrucous skin lesions of 3 weeks’ duration. A review of systems and the patient’s personal and family history were unremarkable. She denied any recent trauma or chemical exposure but noted that she had regularly been patronizing a beauty salon for gel manicures over the past year; her most recent visit was 6 weeks prior to the current presentation. She previously was treated at another dermatology clinic with local corticosteroid creams without any improvement. Dermatologic examination revealed pseudoleukonychia of the distal fingernails surrounded by an erythematous and/or haemorrhagic border. Overgrowth and adherence of the hyponychium to the nail plate also was noted in almost all the fingernails. A prior complete blood cell count and biochemistry panel were within reference range.
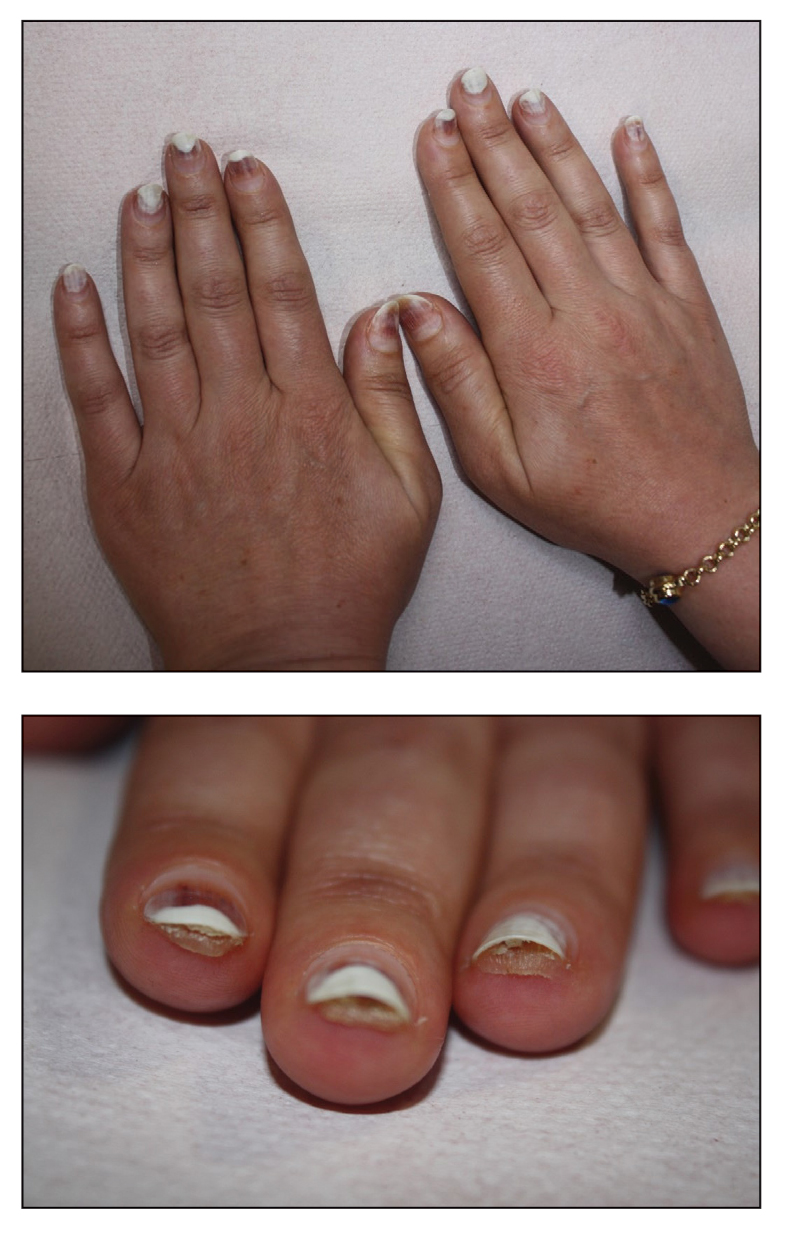
Leaders Helping Leaders: The American Dermatological Association as a Model of Lifelong Professional Community
Leaders Helping Leaders: The American Dermatological Association as a Model of Lifelong Professional Community
Have you ever heard of the American Dermatological Association (ADA)? While many residents may not yet be familiar with this group, its members are among the most respected leaders in dermatology. They serve as current and past presidents of influential organizations including the American Academy of Dermatology (Susan C. Taylor, MD [Philadelphia, Pennsylvania]), the American Society for Dermatologic Surgery (M. Laurin Council, MD, MBA [Creve Coeur, Missouri]), and the Association of Professors of Dermatology (Sewon Kang, MD [Baltimore, Maryland]). Others lead certification boards or serve as editors of key journals like the Journal of the American Academy of Dermatology (Dirk Elston, MD [Charleston, South Carolina]), JAMA Dermatology (Kanade Shinkai, MD [San Francisco, California], and Cutis (Vincent A. DeLeo, MD [Los Angeles, California]).
The ADA is celebrating its 150th anniversary in 2026. What makes the organization so enduring is not just its history, but its culture. The members of the ADA foster deep, long-lasting relationships, and its meetings are purposefully designed to balance structured scientific sessions with unscheduled time for reflection, conversation, and connection. That intentional design cultivates learning, innovation, and wellness.
Steven Covey’s The 7 Habits of Highly Effective People1 highlights the importance of renewal and relationship building, as does the Harvard Study of Adult Development, one of the longest-running research projects on well-being.2-4 The key conclusion? Relationships are the strongest predictors of long, healthy, and fulfilling lives, not wealth or achievement. Medical training is intense, and the emphasis often falls squarely on achievement. But the friendships you form in medical school, residency, and early career are just as formative. Membership with the ADA continues this spirit of connection throughout one’s professional life, with meetings that welcome spouses and partners and encourage engagement across generations.
A hallmark of ADA culture is its commitment to mentoring and mutual support. Need advice about transitioning from private practice to academia? Navigating department leadership? Applying for a grant? Considering industry, editorial, or global health roles? Within the ADA, there’s someone who has done it and is eager to help. Recent meetings have addressed future-facing topics such as artificial intelligence, bedside diagnostics, workforce advocacy, and global health while also carving out time for rejuvenating activities: book clubs with best-selling authors, sessions on the arts, storytelling, wellness, and travel. This holistic programming reflects the ADA’s belief in supporting the whole physician.3 Members understand the value of relationships and appreciate these opportunities to learn about the passions and interests of their colleagues (Table).
Candidates are nominated by current members and must be board certified and at least 10 years beyond completion of their training. Members vote upon candidates in a rank voting system each year. If someone is nominated and not selected, they did not fail—they may be nominated again. The idea behind this membership process is to keep the organization small enough that members can get to know one another—there are currently 552 active members. Importantly, the ADA has embraced diversity and inclusion. While historically male- and White-dominated, recent inductee classes now reflect gender parity and a broader range of backgrounds, enriching the organization with fresh perspectives.5-8
For residents and fellows, the lesson is clear: friendships, mentorship, and time for reflection are not luxuries—they are essential. Burnout stems from relentless output in isolation; however, in cultures that prioritize renewal, authenticity, and community, physicians can flourish.9 Membership in small professional organizations is an important step towards avoiding isolation. We encourage you to be active in your local, state, and national organizations.
The ADA stands as a powerful example of how professional societies can help you build the kind of life and career you want, not just a résumé. From informal beachside conversations to high-level scientific discussions, its enduring strength is this: leaders helping others lead.
- Covey SR. The 7 Habits of Highly Effective People: Powerful Lessons in Personal Change. Simon & Schuster; 1989.
- Waldinger R, Schulz M. The Good Life: Lessons From the World’s Longest Scientific Study of Happiness. Simon & Schuster; 2023.
- Malone JC, Liu SR, Vaillant GE, et al. Midlife Eriksonian psychosocial development: setting the stage for late-life cognitive and emotional health. Dev Psychol. 2016;52:496-508. doi:10.1037/a0039875
- Vaillant GE, Milofsky E. Natural history of male psychological health: IX. Empirical evidence for Erikson’s model of the life cycle. Am J Psychiatry. 1980;137:1348-59. doi:10.1176/ajp.137.11.1348
- American Dermatological Association. Accessed August 8, 2025. https://www.ada1.org
- Pariser DM. Illustrated History of the American Dermatological Association, 1876–2020. American Dermatological Association; 2020.
- Smith JG Jr, Johnson ML. 125th anniversary of the American Dermatological Association. Arch Dermatol. 2001;137:1520. doi:10.1001/archderm.137.11.1520
- Rodriguez R, Anderson L, Woolhiser E, et al. Diversity among American Dermatological Association members by sex and geographic region. JMIR Dermatol. 2024;7:E47802. doi:10.2196/47802
- Grant A. Think Again: The Power of Knowing What You Don’t Know. Viking; 2021.
Have you ever heard of the American Dermatological Association (ADA)? While many residents may not yet be familiar with this group, its members are among the most respected leaders in dermatology. They serve as current and past presidents of influential organizations including the American Academy of Dermatology (Susan C. Taylor, MD [Philadelphia, Pennsylvania]), the American Society for Dermatologic Surgery (M. Laurin Council, MD, MBA [Creve Coeur, Missouri]), and the Association of Professors of Dermatology (Sewon Kang, MD [Baltimore, Maryland]). Others lead certification boards or serve as editors of key journals like the Journal of the American Academy of Dermatology (Dirk Elston, MD [Charleston, South Carolina]), JAMA Dermatology (Kanade Shinkai, MD [San Francisco, California], and Cutis (Vincent A. DeLeo, MD [Los Angeles, California]).
The ADA is celebrating its 150th anniversary in 2026. What makes the organization so enduring is not just its history, but its culture. The members of the ADA foster deep, long-lasting relationships, and its meetings are purposefully designed to balance structured scientific sessions with unscheduled time for reflection, conversation, and connection. That intentional design cultivates learning, innovation, and wellness.
Steven Covey’s The 7 Habits of Highly Effective People1 highlights the importance of renewal and relationship building, as does the Harvard Study of Adult Development, one of the longest-running research projects on well-being.2-4 The key conclusion? Relationships are the strongest predictors of long, healthy, and fulfilling lives, not wealth or achievement. Medical training is intense, and the emphasis often falls squarely on achievement. But the friendships you form in medical school, residency, and early career are just as formative. Membership with the ADA continues this spirit of connection throughout one’s professional life, with meetings that welcome spouses and partners and encourage engagement across generations.
A hallmark of ADA culture is its commitment to mentoring and mutual support. Need advice about transitioning from private practice to academia? Navigating department leadership? Applying for a grant? Considering industry, editorial, or global health roles? Within the ADA, there’s someone who has done it and is eager to help. Recent meetings have addressed future-facing topics such as artificial intelligence, bedside diagnostics, workforce advocacy, and global health while also carving out time for rejuvenating activities: book clubs with best-selling authors, sessions on the arts, storytelling, wellness, and travel. This holistic programming reflects the ADA’s belief in supporting the whole physician.3 Members understand the value of relationships and appreciate these opportunities to learn about the passions and interests of their colleagues (Table).

Candidates are nominated by current members and must be board certified and at least 10 years beyond completion of their training. Members vote upon candidates in a rank voting system each year. If someone is nominated and not selected, they did not fail—they may be nominated again. The idea behind this membership process is to keep the organization small enough that members can get to know one another—there are currently 552 active members. Importantly, the ADA has embraced diversity and inclusion. While historically male- and White-dominated, recent inductee classes now reflect gender parity and a broader range of backgrounds, enriching the organization with fresh perspectives.5-8
For residents and fellows, the lesson is clear: friendships, mentorship, and time for reflection are not luxuries—they are essential. Burnout stems from relentless output in isolation; however, in cultures that prioritize renewal, authenticity, and community, physicians can flourish.9 Membership in small professional organizations is an important step towards avoiding isolation. We encourage you to be active in your local, state, and national organizations.
The ADA stands as a powerful example of how professional societies can help you build the kind of life and career you want, not just a résumé. From informal beachside conversations to high-level scientific discussions, its enduring strength is this: leaders helping others lead.
Have you ever heard of the American Dermatological Association (ADA)? While many residents may not yet be familiar with this group, its members are among the most respected leaders in dermatology. They serve as current and past presidents of influential organizations including the American Academy of Dermatology (Susan C. Taylor, MD [Philadelphia, Pennsylvania]), the American Society for Dermatologic Surgery (M. Laurin Council, MD, MBA [Creve Coeur, Missouri]), and the Association of Professors of Dermatology (Sewon Kang, MD [Baltimore, Maryland]). Others lead certification boards or serve as editors of key journals like the Journal of the American Academy of Dermatology (Dirk Elston, MD [Charleston, South Carolina]), JAMA Dermatology (Kanade Shinkai, MD [San Francisco, California], and Cutis (Vincent A. DeLeo, MD [Los Angeles, California]).
The ADA is celebrating its 150th anniversary in 2026. What makes the organization so enduring is not just its history, but its culture. The members of the ADA foster deep, long-lasting relationships, and its meetings are purposefully designed to balance structured scientific sessions with unscheduled time for reflection, conversation, and connection. That intentional design cultivates learning, innovation, and wellness.
Steven Covey’s The 7 Habits of Highly Effective People1 highlights the importance of renewal and relationship building, as does the Harvard Study of Adult Development, one of the longest-running research projects on well-being.2-4 The key conclusion? Relationships are the strongest predictors of long, healthy, and fulfilling lives, not wealth or achievement. Medical training is intense, and the emphasis often falls squarely on achievement. But the friendships you form in medical school, residency, and early career are just as formative. Membership with the ADA continues this spirit of connection throughout one’s professional life, with meetings that welcome spouses and partners and encourage engagement across generations.
A hallmark of ADA culture is its commitment to mentoring and mutual support. Need advice about transitioning from private practice to academia? Navigating department leadership? Applying for a grant? Considering industry, editorial, or global health roles? Within the ADA, there’s someone who has done it and is eager to help. Recent meetings have addressed future-facing topics such as artificial intelligence, bedside diagnostics, workforce advocacy, and global health while also carving out time for rejuvenating activities: book clubs with best-selling authors, sessions on the arts, storytelling, wellness, and travel. This holistic programming reflects the ADA’s belief in supporting the whole physician.3 Members understand the value of relationships and appreciate these opportunities to learn about the passions and interests of their colleagues (Table).

Candidates are nominated by current members and must be board certified and at least 10 years beyond completion of their training. Members vote upon candidates in a rank voting system each year. If someone is nominated and not selected, they did not fail—they may be nominated again. The idea behind this membership process is to keep the organization small enough that members can get to know one another—there are currently 552 active members. Importantly, the ADA has embraced diversity and inclusion. While historically male- and White-dominated, recent inductee classes now reflect gender parity and a broader range of backgrounds, enriching the organization with fresh perspectives.5-8
For residents and fellows, the lesson is clear: friendships, mentorship, and time for reflection are not luxuries—they are essential. Burnout stems from relentless output in isolation; however, in cultures that prioritize renewal, authenticity, and community, physicians can flourish.9 Membership in small professional organizations is an important step towards avoiding isolation. We encourage you to be active in your local, state, and national organizations.
The ADA stands as a powerful example of how professional societies can help you build the kind of life and career you want, not just a résumé. From informal beachside conversations to high-level scientific discussions, its enduring strength is this: leaders helping others lead.
- Covey SR. The 7 Habits of Highly Effective People: Powerful Lessons in Personal Change. Simon & Schuster; 1989.
- Waldinger R, Schulz M. The Good Life: Lessons From the World’s Longest Scientific Study of Happiness. Simon & Schuster; 2023.
- Malone JC, Liu SR, Vaillant GE, et al. Midlife Eriksonian psychosocial development: setting the stage for late-life cognitive and emotional health. Dev Psychol. 2016;52:496-508. doi:10.1037/a0039875
- Vaillant GE, Milofsky E. Natural history of male psychological health: IX. Empirical evidence for Erikson’s model of the life cycle. Am J Psychiatry. 1980;137:1348-59. doi:10.1176/ajp.137.11.1348
- American Dermatological Association. Accessed August 8, 2025. https://www.ada1.org
- Pariser DM. Illustrated History of the American Dermatological Association, 1876–2020. American Dermatological Association; 2020.
- Smith JG Jr, Johnson ML. 125th anniversary of the American Dermatological Association. Arch Dermatol. 2001;137:1520. doi:10.1001/archderm.137.11.1520
- Rodriguez R, Anderson L, Woolhiser E, et al. Diversity among American Dermatological Association members by sex and geographic region. JMIR Dermatol. 2024;7:E47802. doi:10.2196/47802
- Grant A. Think Again: The Power of Knowing What You Don’t Know. Viking; 2021.
- Covey SR. The 7 Habits of Highly Effective People: Powerful Lessons in Personal Change. Simon & Schuster; 1989.
- Waldinger R, Schulz M. The Good Life: Lessons From the World’s Longest Scientific Study of Happiness. Simon & Schuster; 2023.
- Malone JC, Liu SR, Vaillant GE, et al. Midlife Eriksonian psychosocial development: setting the stage for late-life cognitive and emotional health. Dev Psychol. 2016;52:496-508. doi:10.1037/a0039875
- Vaillant GE, Milofsky E. Natural history of male psychological health: IX. Empirical evidence for Erikson’s model of the life cycle. Am J Psychiatry. 1980;137:1348-59. doi:10.1176/ajp.137.11.1348
- American Dermatological Association. Accessed August 8, 2025. https://www.ada1.org
- Pariser DM. Illustrated History of the American Dermatological Association, 1876–2020. American Dermatological Association; 2020.
- Smith JG Jr, Johnson ML. 125th anniversary of the American Dermatological Association. Arch Dermatol. 2001;137:1520. doi:10.1001/archderm.137.11.1520
- Rodriguez R, Anderson L, Woolhiser E, et al. Diversity among American Dermatological Association members by sex and geographic region. JMIR Dermatol. 2024;7:E47802. doi:10.2196/47802
- Grant A. Think Again: The Power of Knowing What You Don’t Know. Viking; 2021.
Leaders Helping Leaders: The American Dermatological Association as a Model of Lifelong Professional Community
Leaders Helping Leaders: The American Dermatological Association as a Model of Lifelong Professional Community
Practice Points
- Professional relationships are a critical determinant of career longevity and personal well-being. Structured opportunities for mentorship, reflection, and intergenerational dialogue within professional societies can help mitigate burnout and foster sustained leadership development.
- Medical societies serve as leadership incubators. Through selective membership, cross-organizational representation, and sustained engagement, the American Dermatological Association models how professional community strengthens the specialty beyond scientific exchange alone.
- Physicians benefit from intentional engagement in organized dermatology. Participation in local, state, and national societies cultivates mentorship networks, broadens perspective, and lays the foundation for future leadership opportunities.
Hospital Dermatology: Review of Research in 2024-2025
Hospital Dermatology: Review of Research in 2024-2025
Dermatologists play a central role in the care of hospitalized patients with skin disease. This review summarizes research from January 2024 to December 2025 on severe cutaneous adverse drug reactions, emerging infectious diseases, hidradenitis suppurativa (HS), and inpatient dermatology workforce issues. Key developments include improved recognition and management of drug reactions; updated diagnostic and prognostic tools for Stevens-Johnson syndrome (SJS) and toxic epidermal necrolysis (TEN); and guidance for emerging infections such as measles, dengue, mpox, orthopoxviruses, and resistant dermatophytes. Evidence-based strategies for HS aim to reduce unnecessary admissions and optimize care. Workforce challenges, including limited access, high call burden, and potential for artificial intelligence (AI)–assisted diagnosis, are also highlighted. These findings emphasize the critical contributions of dermatologists to hospital-based care and provide emerging evidence to guide clinical practice.
Dermatologists play a critical role in the care of hospitalized patients. Herein, we review the research developments between January 2024 and December 2025 most relevant to the care of hospitalized patients with skin disease, including severe cutaneous adverse reactions (SCARs), emerging and re-emerging infectious diseases, hidradenitis suppurativa (HS), and access to inpatient dermatology services.
Severe Cutaneous Adverse Drug Reactions
Severe cutaneous adverse drug reactions are among the most frequent reasons for inpatient dermatology consultation. A National Inpatient Sample study identified more than 160,000 cases of drug rash with eosinophilia and systemic symptoms (DRESS syndrome) between January 2016 and December 2020.1 The overall mortality rate was 2.0%, substantially lower than the rates of up to 10% reported in earlier studies.2 Case burden and mortality peaked during the fall months, possibly due to either increased use of antibiotics or increased viral infection or reactivation during these months.1
A retrospective cohort study of patients with probable or definite DRESS syndrome showed that, among 93 patients with at least 1 viral marker tested, human herpesvirus (HHV) reactivation was found in 42% (39/93), including HHV-6 (28%)(24/85), Epstein-Barr virus (17%)(15/87), and cytomegalovirus (20%)(18/89); furthermore, viral reactivation was associated with higher 1-year mortality (odds ratio, 3.9), dialysis initiation, flares of disease, and longer hospital stay (all P<.05).1 Multiple reactivations were associated with higher inpatient mortality and 1-year mortality; however, despite apparent prognostic importance, the role of screening for viral reactivation in DRESS syndrome is undefined.
A systematic review of 610 studies including 2122 patients with DRESS syndrome demonstrated that, among 193 causal agents identified, 14 drugs accounted for more than 1% of cases each and therefore were considered high risk. Seventy-eight percent of cases were attributed to these 14 drugs (Table).5 A TriNetX Query study analyzed antibiotic exposures across SCARs and reported that sulfonamides (hazard ratio [HR], 7.5), aminoglycosides (HR, 3.7), and tetracyclines (HR, 1.7) were associated with an elevated risk for SCARs. Sulfonamides had the highest absolute incidence of SCARs, followed by cephalosporins and penicillins.6
A multicenter randomized clinical trial7 compared high-potency topical corticosteroids (clobetasol 30 g/d) to systemic corticosteroids (prednisone 0.5 mg/kg/d) for treatment of moderate DRESS syndrome. On day 30, 53.8% (14/26) of patients in the topical group had achieved remission of visceral involvement, compared to 72.0% (18/25) in the systemic group. Before day 30, 23.1% (6/26) of patients in the topical group worsened, necessitating transition to high-dose systemic steroids. When inpatient monitoring is available, low-dose systemic corticosteroids or high-potency topical steroids may be reasonable management strategies for moderate DRESS syndrome7; however, the frequent need for treatment intensification suggests limitations to this strategy.
Since prolonged courses of systemic steroids generally are necessary for management of DRESS syndrome, steroid-sparing options are needed. A retrospective case series examined interleukin 5 inhibition in patients with possible DRESS syndrome (Registry of Severe Cutaneous Adverse Reactions score ≥3). All patients demonstrated rapid eosinophil reduction within 1 to 3 days (mean [SD] time to resolution, 1.4 [0.9] days) after treatment with mepolizumab or benralizumab, with clinical improvement occurring at a mean (SD) of 16 (3.7) days (range, 13-21 days).8
A French cohort study of 1221 adult patients with Stevens-Johnson syndrome (SJS)/toxic epidermal necrolysis (TEN) reported in-hospital mortality of 19% and a total mortality of 34% at 1 year.9 Risk factors contributing to in-hospital mortality included age, history of/current diagnosis of cancer, dementia, and liver disease, while postdischarge mortality was associated with acute kidney injury and sepsis. Long-term complications included ophthalmologic and mood disorders.9
A new set of diagnostic criteria for SJS/TEN, known as the Niigata criteria,10 includes 3 main items: severe mucosal lesions in cutaneous-mucosal transition zones (eg, eyes, lips, vulva) or generalized erythema with necrotic lesions; fever of 38.5 °C or higher; and necrosis of the epidermis seen on histopathology. Because epidermal detachment involving 10% of the body surface area (BSA) is an important mortality risk predicter, SJS is defined as less than 10% BSA involvement, and TEN has been redefined as 10% or more BSA involvement (not ≥30%). A new prognostic score—clinical risk score for TEN (CRISTEN)—can be tabulated at the point of care without laboratory values. It was developed based on the 10 most important risk factors for death in a retrospective study of 382 patients, which included age 65 years or older; epidermal detachment involving 10% BSA or higher; an antibiotic as causative agent; systemic corticosteroid therapy before the onset of SJS/TEN; involvement of all 3 mucosal surfaces; and medical comorbidities such as renal impairment, diabetes, cardiac disease, active cancer, and bacterial infection.11
New potential therapeutic targets for SJS/TEN include PC111 (monoclonal antibody to Fas ligand), formyl peptide receptor 1 antagonists (which inhibit necroptosis induced by formyl peptide receptor 1–annexin A1 interaction), daratumumab (which depletes cytotoxic CD8-positive and CD38-positive T cells), and Janus kinase (JAK) inhibitors.10 Spatial proteomics showed marked enrichment of type I and type II interferon signatures as well as activation of signal transducer and activator of transcription 1. In vitro, tofacitinib reduced keratinocyte-directed cytotoxicity, and in vivo JAK inhibitors ameliorated disease severity in 2 TEN mouse models. Patients with TEN that was refractory to corticosteroid therapy received rescue treatment with JAK inhibitors and had re-epithelization within several days with marked reduction in levels of phosphorylated signal transducer and activator of transcription 1.12 Controlled studies are needed to assess the potential role of JAK inhibitors for SJS/TEN.
Emerging and Re-emerging Infectious Diseases
Dermatologists may encounter emerging or re-emerging infections, performing an essential public health role in the process. In 2025, a total of 2281 confirmed cases of measles had been reported across 45 of the United States.13 During the COVID-19 pandemic, measles vaccine coverage in the United States dropped to 93%—down from 95% to 97% prepandemic. Worldwide, 2022 saw an increase of 1.4 million measles cases (18% increase) and 41,200 excess deaths (43% increase) compared to the previous year. Complications of measles include pneumonia, blindness, otitis media, and encephalitis, with 1 in 5 (20%) unvaccinated people with measles in the United States requiring hospitalization.14 A vaccine coverage rate higher than 95% is needed to prevent community spread of disease. Since efforts to detect and rapidly isolate cases of measles are critical, dermatologists should consider measles in the differential of morbilliform eruptions with viral symptoms and ask about vaccination status.
Since 2023, dengue infection rates have tripled in the Americas, representing the highest levels recorded since tracking began in 1980. In 2024, there were more than 12 million cases, with approximately 8000 deaths reported. Ninety percent of cases occur in Argentina, Brazil, Colombia, and Mexico, but local transmission has been reported in Arizona, California, Florida, Hawaii, and Texas.15 The characteristic exanthem of dengue is diffuse erythema with islands of sparing.<
Unlike during the 2022 outbreak of mpox clade II, which predominantly impacted men who have sex with men, there now is an ongoing outbreak of mpox clades 1a and 1b in the Democratic Republic of the Congo and surrounding countries that more commonly affects children and heterosexual adults. It is also more transmissible and virulent. Cases of mpox clade I have been reported in several European countries and across the United States, mostly among travelers from areas of active transmission. Vaccination of at-risk individuals is considered effective; however, tecovirimat is not.16
Outbreaks of 2 emerging zoonotic orthopoxviruses recently have been reported. Buffalopox virus (BPXV) is transmitted via direct contact with the skin of infected cattle and buffalo as well as fomites and has been responsible for human cases in South Asia. Characteristics of BPXV include macules, umbilicated papules, vesicles, pustules, and eschars that evolve over several weeks, with a predilection for the hands and face. It can manifest with prodromal symptoms of fever, malaise, and lymphadenopathy.17 Borealpox virus (formerly known as Alaskapox) has similar manifestations. Its reservoir includes small mammals such as voles and shrews, but it also has been found in cats and dogs and has been responsible for at least one human fatality. Cidofovir may be an effective therapy for both BPXV and borealpox virus, and prior smallpox vaccination may provide protection.18 These outbreaks demonstrate the continued importance of research for more effective vaccines and therapies against smallpox and other orthopoxviruses.19 A recent review provided a detailed overview of the epidemiology, transmission, dermatologic findings, and management strategies associated with smallpox and other bioweapons.20
In 2023, a case was reported of a patient in a New York City hospital with tinea that was refractory to multiple rounds of topical antifungals, which called attention to the presence of Trichophyton indotineae in the United States.21 Since then, additional reports and case series have characterized the clinical presentation of T indotineae as widespread and atypical, refractory to traditional therapies, and most often encountered in travelers returning from Bangladesh or elsewhere in South Asia.22 The diagnosis should be confirmed via DNA testing of fungal culture. Itraconazole 100 to 200 mg/d is the antifungal therapy of choice.23
Other series have reported cases of tinea genitalis caused by Trichophyton mentagrophytes type VII seen predominately in sex workers and others engaging in high-risk sexual contact, highlighting the spread of dermatophytes through sexual activity.24-26 Lastly, it is important to culture pustules and consider atypical pathogens in patients with chronic folliculitis not responding to typical therapies such as tetracycline antibiotics. A case series reported the presence of pustules in the beard area of 7 men who have sex with men, with culture data showing Klebsiella aerogenes. Prolonged courses of fluoroquinolones were necessary for clearance.27
Reducing HS Admissions Through Evidence-Based Management
Hidradenitis suppurativa is a frequent cause of emergency department visits and hospital admissions. In an analysis of the Nationwide Readmissions Database, 17.8% (392/2204) of patients admitted to the hospital with HS were readmitted within 30 days, a number comparable to that of heart failure.28
Flaring HS can produce symptoms that mimic sepsis. A retrospective cohort study examining sepsislike features in HS showed that more than 50% (30/58) of those admitted to the hospital with an HS flare were misdiagnosed with sepsis, and more than 80% (53/64) of those patients received intravenous antibiotics.29 A National Inpatient Sample (January 2016-December 2018) study demonstrated minimal rates of true infection in patients admitted with HS flares,30 while patients with HS diagnosed as sepsis do not sustain the mortality expected from true sepsis. Improving recognition of HS and differentiation of the disease from true sepsis could decrease unnecessary antibiotic use, hospital admissions, and cost, underscoring the need for a framework to reliably and reproducibly distinguish sepsis from HS flare.31
While severe HS is difficult to manage, there may be a window of opportunity in which appropriate treatment of early disease may prevent progression and decrease inpatient utilization. A prospective cohort study of 335 biologic-naïve patients with mild to moderate HS (Hurley stages I and II) followed over a median of 2 years showed that active smoking, body mass index higher than 25, and the presence of disease in 2 or more anatomic areas were factors predictive of progression to severe disease.32
Despite high utilization of emergency and inpatient care, there has been no consensus on inpatient management of HS. A Delphi consensus exercise including 26 expert dermatologists reached consensus on 40 statements.33 Specific recommendations involve multidisciplinary care, including from a dermatologist; consideration of comorbid medical conditions; supportive care measures (wound care, pain control); evidence-based medical management, including initiation or adjustment of biologic therapies; targeted surgical intervention; nutritional support and maintenance of glycemic control; and attention to transitional care at discharge, including home health services, verification of insurance status, and timely outpatient dermatology follow-up.34 A retrospective review of 98 patients treated with intravenous ertapenem for a mean duration of 13 weeks demonstrated improvement in clinical and inflammatory markers.35 Patients with severe or treatment-refractory HS, including those admitted to the hospital, may benefit from initiation of this therapy in select circumstances.
Hospital Dermatology Workforce
Inpatient dermatology consultations are extremely valuable for improving diagnostic accuracy, reducing admissions for pseudocellulitis and inflammatory skin conditions, and keeping cancer patients on needed therapies.36-38 Despite this clear value added, a cross-sectional analysis of inpatient Medicare claims data from January 2013 to December 2019 found that the number of dermatologists performing more than 10 inpatient consults per year decreased from 356 to 281.39 Additionally, medical centers in which dermatology encounters occurred decreased from 239 to 157 during the same period. Ninety-eight percent of inpatient dermatologists were in metropolitan areas, with large regions lacking access to inpatient dermatology consultation altogether.39
A survey of Society for Pediatric Dermatology members similarly characterized the state of the pediatric dermatology workforce performing hospital consultation.40 Seventy-five percent reported a high call burden, defined as more than 11 days or nights per month, more than 1 weekend per month, and/or more than 5 hours per week seeing patients. Ninety-one percent of consultation services are based within academic institutions, reflecting disparities in access.40 A prospective cohort study of academic pediatric dermatologists reported that 310 curbside consultations were performed over 24 weeks; of these calls, 17% occurred during weeknights and 23% on weekends. None of these curbside interactions was reimbursed.41 These findings underscore the burden of uncompensated time a subset of pediatric dermatologists dedicates to inpatient consultations, highlighting the need for improved financial and administrative support and an increased number of physicians performing this role.
A survey study42 suggested that unfamiliarity with the inpatient setting, rather than medical knowledge, is the most important barrier to inpatient work among clinical dermatologists. Proposed interventions include resource guides (eg, hospital maps, pager numbers for key individuals, and protocols for urgent specimens). Reference guides and refresher courses may decrease gaps in knowledge or awareness among dermatologists in ambulatory practice.42 Another way to bolster the inpatient dermatology workforce may be to provide more guidance to qualified advanced practice providers to triage and address dermatologic emergencies.43
Artificial intelligence (AI) also has been explored as a tool for diagnosing complex dermatologic conditions. One study presented 15 published inpatient dermatology cases to 7 dermatologists. Participants were asked to formulate their top 3 differential diagnoses and were then shown AI-generated differentials and asked to submit a revised differential. Participants showed a diagnostic accuracy of 69% before seeing the AI-generated differential diagnosis and 79% after; however, in cases in which the AI differential was incorrect, diagnostic accuracy of the dermatologists decreased after being shown the AI model.44
Final Thoughts
This January 2024 to December 2025 review of research relevant to hospital dermatology highlights important developments and ongoing challenges in SCARs, emerging and re-emerging infectious diseases, HS, and the inpatient dermatology workforce. Dermatologists continue to play a critical role in the care of hospitalized patients with skin disease.
- Desai AD, Thomas C. Seasonal trends in drug reaction with eosinophilia and systemic symptoms. J Am Acad Dermatol. 2025;92:183-185.
- Wei BM, Fox LP, Kaffenberger BH, et al. Drug-induced hypersensitivity syndrome/drug reaction with eosinophilia and systemic symptoms. Part I. Epidemiology, pathogenesis, clinicopathological features, and prognosis. J Am Acad Dermatol. 2024;90:885-908. doi:10.1016/j.jaad.2023.02.072
- Chan LCE, Sultana R, Choo KJL, et al. Viral reactivation and clinical outcomes in drug reaction with eosinophilia and systemic symptoms (DRESS). Sci Rep. 2024;14:28492.
- Brüggen MC, Walsh S, Ameri MM, et al. Management of adult patients with drug reaction with eosinophilia and systemic symptoms: a Delphi-based international consensus. JAMA Dermatol. 2024;160:37-44
- Hansen E, Gallardo M, Yan A, et al. Risk assessment of drugs associated with DRESS syndrome based on publication frequency: a systematic review. J Am Acad Dermatol. 2024;91:962-966.
- Neubauer ZJK, Chan R, Singal A, et al. SCAR-ed by antibiotics: a retrospective cohort study of severe cutaneous adverse reactions (SCAR) relative risk. J Am Acad Dermatol. 2025;92:1143-1145.
- Ingen-Housz-Oro S, Guichard E, Milpied B, et al. Topical versus oral corticosteroids in moderate drug reaction with eosinophilia and systemic symptoms: a multicenter randomized clinical trial. J Am Acad Dermatol. 2024;91:544-547.
- Hijaz B, Nambudiri VE, Imadojemu S. IL-5 inhibitor treatment in drug reaction with eosinophilia and systemic symptoms. JAMA Dermatol. 2025;161:661-663.
- Bettuzzi T, Lebrun-Vignes B, Ingen-Housz-Oro S, et al. Incidence, in-hospital and long-term mortality, and sequelae of epidermal necrolysis in adults. JAMA Dermatol. 2024;160:1288-1296.
- Hama N, Aoki S, Chen CB, et al. Recent progress in Stevens-Johnson syndrome/toxic epidermal necrolysis: diagnostic criteria, pathogenesis and treatment. Br J Dermatol. 2024;192:9-18.
- Hama N, Sunaga Y, Ochiai H, et al. Development and validation of a novel score to predict mortality in Stevens-Johnson syndrome and toxic epidermal necrolysis: CRISTEN. J Allergy Clin Immunol Pract. 2023;11:3161-3168.e2.
- Nordmann TM, Anderton H, Hasegawa A, et al. Spatial proteomics identifies JAKi as treatment for a lethal skin disease. Nature. 2024;635:1001-1009.
- Centers for Disease Control and Prevention. Measles cases and outbreaks. Updated January 7, 2026. Accessed January 12, 2026. https://www.cdc.gov/measles/data-research/
- Rubin R. Despite safe and effective vaccine, measles cases and deaths increased worldwide from 2021 to 2022. JAMA. 2024;331:188-189.
- Orrall A. Dengue cases in the Americas highest recorded. JAMA. 2025;333:452.
- Harris E. As mpox cases surge in Africa, WHO declares a global emergency-here’s what to know. JAMA. 2024;332:862-864.
- Burningham KM, Hinojosa T, Cavazos A, et al. Buffalopox: an emerging cutaneous disease in humans. J Eur Acad Dermatol Venereol. 2025;39:404-406.
- Parker ER. Emergence of Alaskapox infection: what dermatologists need to know. J Am Acad Dermatol. 2024;91:397-399.
- Gostin LO, Singaravelu S, Hynes N. Smallpox readiness: modern strategies against an ancient disease. JAMA. 2024;332:873-874.
- Osborne S, Kam O, Thacker S, et al. Review of category A bioweapons with cutaneous features: epidemiology, clinical presentation, and contemporary management strategies. J Am Acad Dermatol. 2025;93:165-175.
- Caplan AS, Chaturvedi S, Zhu Y, et al. Notes from the field: first reported U.S. cases of tinea caused by Trichophyton indotineae - New York City, December 2021-March 2023. MMWR Morb Mortal Wkly Rep. 2023;72:536-537.
- McKenna M. Why the rise of this drug-resistant fungus is raising international concern. JAMA. 2024;332:859-861.
- Caplan AS, Todd GC, Zhu Y, et al. Clinical course, antifungal susceptibility, and genomic sequencing of Trichophyton indotineae. JAMA Dermatol. 2024;160:701-709.
- Jabet A, Bérot V, Chiarabini T, et al. Trichophyton mentagrophytes ITS genotype VII infections among men who have sex with men in France: an ongoing phenomenon. J Eur Acad Dermatol Venereol. 2025;39:407-415.
- Luchsinger I, Bosshard PP, Kasper RS, et al. Tinea genitalis: a new entity of sexually transmitted infection? Case series and review of the literature. Sex Transm Infect. 2015;91:493-496.
- Khurana A, Sharath S, Sardana K, et al. Therapeutic updates on the management of tinea corporis or cruris in the era of Trichophyton indotineae: separating evidence from hype-a narrative review. Indian J Dermatol. 2023;68:525-540.
- Bérot V, Monsel G, Dauendorffer JN, et al; Groupe Infectiologie Dermatologique et Infections Sexuellement Transmissibles (GrIDIST) de la Société Française de Dermatologie. Klebsiella aerogenes-related facial folliculitis in men having sex with men: a hypothetical new STI?J Eur Acad Dermatol Venereol. 2025;39:E10-E12.
- Edigin E, Kaul S, Eseaton PO, et al. At 180 days hidradenitis suppurativa readmission rate is comparable to heart failure: analysis of the Nationwide Readmissions Database. J Am Acad Dermatol. 2022;87:188-192.
- AbdelHameid D, Wang L, Mauskar MM, et al. Sepsis-like features in hidradenitis suppurativa flares requiring admission: a retrospective cohort study. J Am Acad Dermatol. 2024;90:1291-1294.
- Ehizogie E, Maghari I, Lo S, et al. Hidradenitis suppurativa, systemic inflammatory response syndrome and sepsis: a database study. Br J Dermatol. 2024;191:451-453.
- Maghari I, Abiad H, Griffin T, et al. Hidradenitis suppurativa (HS), systemic inflammatory response syndrome and sepsis, sepsis caused by HS: an empty systematic review. Br J Dermatol. 2024;191:449-450.
- Kjærsgaard Andersen R, Pedersen O, Eidsmo L, et al. Initial steps towards developing a predictive algorithm of disease progression for hidradenitis suppurativa (HS): results from a Cox proportional hazard regression analysis on disease progression among a cohort of 335 Danish patients with HS. Br J Dermatol. 2024;190:904-914.
- Needham M, Pichardo R, Alavi A, et al. Inpatient management of hidradenitis suppurativa: a Delphi consensus study. Cutis. 2024;113:251-254.
- Maskan Bermudez N, Elman SA, Kirsner RS, et al. Management of hidradenitis suppurativa in the inpatient setting: a clinical guide. Arch Dermatol Res. 2025;317:202.
- Nosrati A, Ch’en PY, Torpey ME, et al. Efficacy and durability of intravenous ertapenem therapy for recalcitrant hidradenitis suppurativa. JAMA Dermatol. 2024;160:312-318.
- Tracey EH, Forrestel A, Rosenbach M, et al. Inpatient dermatology consultation in patients with hematologic malignancies. J Am Acad Dermatol. 2016;75:835-836.
- Li DG, Xia FD, Khosravi H, et al. Outcomes of early dermatology consultation for inpatients diagnosed with cellulitis. JAMA Dermatol. 2018;154:537-543.
- Jacoby TV, Shah N, Asdourian MS, et al. Dermatology evaluation for cutaneous immune-related adverse events is associated with improved survival in cancer patients treated with checkpoint inhibition. J Am Acad Dermatol. 2023;88:711-714.
- Hydol-Smith JA, Gallardo MA, Korman A, et al. The United States dermatology inpatient workforce between 2013 and 2019: a Medicare analysis reveals contraction of the workforce and vast access deserts-a cross-sectional analysis. Arch Dermatol Res. 2024;316:103.
- Pineider JL, Rangu SA, Shaw KS, et al. Pediatric consultative dermatology: a survey of the Society for Pediatric Dermatology workforce reveals shortcomings in existing practice models of pediatric dermatology consult services in the United States. Pediatr Dermatol. 2024;41:270-274.
- Puar NK, Canty KM, Newell BD, et al. An evaluation of pediatric dermatology curbside consultations in an academic center: a prospective cohort study. J Am Acad Dermatol. 2024;90:1258-1260.
- Lau CB, Smith GP. Strategies for improving dermatologist comfort and quality of patient care in inpatient settings: a cross-sectional survey study. Arch Dermatol Res. 2024;316:575.
- Hazim AH. Empowering advanced clinical practitioners in managing acute dermatological emergencies. Br J Nurs. 2024;33:448-455.
- Macklis P, Kaffenberger B, Kirven R, et al. Dermatology diagnostic accuracy is improved by artificial intelligence-generated differential diagnoses. Int J Dermatol. 2025;64:960-962.
Dermatologists play a central role in the care of hospitalized patients with skin disease. This review summarizes research from January 2024 to December 2025 on severe cutaneous adverse drug reactions, emerging infectious diseases, hidradenitis suppurativa (HS), and inpatient dermatology workforce issues. Key developments include improved recognition and management of drug reactions; updated diagnostic and prognostic tools for Stevens-Johnson syndrome (SJS) and toxic epidermal necrolysis (TEN); and guidance for emerging infections such as measles, dengue, mpox, orthopoxviruses, and resistant dermatophytes. Evidence-based strategies for HS aim to reduce unnecessary admissions and optimize care. Workforce challenges, including limited access, high call burden, and potential for artificial intelligence (AI)–assisted diagnosis, are also highlighted. These findings emphasize the critical contributions of dermatologists to hospital-based care and provide emerging evidence to guide clinical practice.
Dermatologists play a critical role in the care of hospitalized patients. Herein, we review the research developments between January 2024 and December 2025 most relevant to the care of hospitalized patients with skin disease, including severe cutaneous adverse reactions (SCARs), emerging and re-emerging infectious diseases, hidradenitis suppurativa (HS), and access to inpatient dermatology services.
Severe Cutaneous Adverse Drug Reactions
Severe cutaneous adverse drug reactions are among the most frequent reasons for inpatient dermatology consultation. A National Inpatient Sample study identified more than 160,000 cases of drug rash with eosinophilia and systemic symptoms (DRESS syndrome) between January 2016 and December 2020.1 The overall mortality rate was 2.0%, substantially lower than the rates of up to 10% reported in earlier studies.2 Case burden and mortality peaked during the fall months, possibly due to either increased use of antibiotics or increased viral infection or reactivation during these months.1
A retrospective cohort study of patients with probable or definite DRESS syndrome showed that, among 93 patients with at least 1 viral marker tested, human herpesvirus (HHV) reactivation was found in 42% (39/93), including HHV-6 (28%)(24/85), Epstein-Barr virus (17%)(15/87), and cytomegalovirus (20%)(18/89); furthermore, viral reactivation was associated with higher 1-year mortality (odds ratio, 3.9), dialysis initiation, flares of disease, and longer hospital stay (all P<.05).1 Multiple reactivations were associated with higher inpatient mortality and 1-year mortality; however, despite apparent prognostic importance, the role of screening for viral reactivation in DRESS syndrome is undefined.
A systematic review of 610 studies including 2122 patients with DRESS syndrome demonstrated that, among 193 causal agents identified, 14 drugs accounted for more than 1% of cases each and therefore were considered high risk. Seventy-eight percent of cases were attributed to these 14 drugs (Table).5 A TriNetX Query study analyzed antibiotic exposures across SCARs and reported that sulfonamides (hazard ratio [HR], 7.5), aminoglycosides (HR, 3.7), and tetracyclines (HR, 1.7) were associated with an elevated risk for SCARs. Sulfonamides had the highest absolute incidence of SCARs, followed by cephalosporins and penicillins.6

A multicenter randomized clinical trial7 compared high-potency topical corticosteroids (clobetasol 30 g/d) to systemic corticosteroids (prednisone 0.5 mg/kg/d) for treatment of moderate DRESS syndrome. On day 30, 53.8% (14/26) of patients in the topical group had achieved remission of visceral involvement, compared to 72.0% (18/25) in the systemic group. Before day 30, 23.1% (6/26) of patients in the topical group worsened, necessitating transition to high-dose systemic steroids. When inpatient monitoring is available, low-dose systemic corticosteroids or high-potency topical steroids may be reasonable management strategies for moderate DRESS syndrome7; however, the frequent need for treatment intensification suggests limitations to this strategy.
Since prolonged courses of systemic steroids generally are necessary for management of DRESS syndrome, steroid-sparing options are needed. A retrospective case series examined interleukin 5 inhibition in patients with possible DRESS syndrome (Registry of Severe Cutaneous Adverse Reactions score ≥3). All patients demonstrated rapid eosinophil reduction within 1 to 3 days (mean [SD] time to resolution, 1.4 [0.9] days) after treatment with mepolizumab or benralizumab, with clinical improvement occurring at a mean (SD) of 16 (3.7) days (range, 13-21 days).8
A French cohort study of 1221 adult patients with Stevens-Johnson syndrome (SJS)/toxic epidermal necrolysis (TEN) reported in-hospital mortality of 19% and a total mortality of 34% at 1 year.9 Risk factors contributing to in-hospital mortality included age, history of/current diagnosis of cancer, dementia, and liver disease, while postdischarge mortality was associated with acute kidney injury and sepsis. Long-term complications included ophthalmologic and mood disorders.9
A new set of diagnostic criteria for SJS/TEN, known as the Niigata criteria,10 includes 3 main items: severe mucosal lesions in cutaneous-mucosal transition zones (eg, eyes, lips, vulva) or generalized erythema with necrotic lesions; fever of 38.5 °C or higher; and necrosis of the epidermis seen on histopathology. Because epidermal detachment involving 10% of the body surface area (BSA) is an important mortality risk predicter, SJS is defined as less than 10% BSA involvement, and TEN has been redefined as 10% or more BSA involvement (not ≥30%). A new prognostic score—clinical risk score for TEN (CRISTEN)—can be tabulated at the point of care without laboratory values. It was developed based on the 10 most important risk factors for death in a retrospective study of 382 patients, which included age 65 years or older; epidermal detachment involving 10% BSA or higher; an antibiotic as causative agent; systemic corticosteroid therapy before the onset of SJS/TEN; involvement of all 3 mucosal surfaces; and medical comorbidities such as renal impairment, diabetes, cardiac disease, active cancer, and bacterial infection.11
New potential therapeutic targets for SJS/TEN include PC111 (monoclonal antibody to Fas ligand), formyl peptide receptor 1 antagonists (which inhibit necroptosis induced by formyl peptide receptor 1–annexin A1 interaction), daratumumab (which depletes cytotoxic CD8-positive and CD38-positive T cells), and Janus kinase (JAK) inhibitors.10 Spatial proteomics showed marked enrichment of type I and type II interferon signatures as well as activation of signal transducer and activator of transcription 1. In vitro, tofacitinib reduced keratinocyte-directed cytotoxicity, and in vivo JAK inhibitors ameliorated disease severity in 2 TEN mouse models. Patients with TEN that was refractory to corticosteroid therapy received rescue treatment with JAK inhibitors and had re-epithelization within several days with marked reduction in levels of phosphorylated signal transducer and activator of transcription 1.12 Controlled studies are needed to assess the potential role of JAK inhibitors for SJS/TEN.
Emerging and Re-emerging Infectious Diseases
Dermatologists may encounter emerging or re-emerging infections, performing an essential public health role in the process. In 2025, a total of 2281 confirmed cases of measles had been reported across 45 of the United States.13 During the COVID-19 pandemic, measles vaccine coverage in the United States dropped to 93%—down from 95% to 97% prepandemic. Worldwide, 2022 saw an increase of 1.4 million measles cases (18% increase) and 41,200 excess deaths (43% increase) compared to the previous year. Complications of measles include pneumonia, blindness, otitis media, and encephalitis, with 1 in 5 (20%) unvaccinated people with measles in the United States requiring hospitalization.14 A vaccine coverage rate higher than 95% is needed to prevent community spread of disease. Since efforts to detect and rapidly isolate cases of measles are critical, dermatologists should consider measles in the differential of morbilliform eruptions with viral symptoms and ask about vaccination status.
Since 2023, dengue infection rates have tripled in the Americas, representing the highest levels recorded since tracking began in 1980. In 2024, there were more than 12 million cases, with approximately 8000 deaths reported. Ninety percent of cases occur in Argentina, Brazil, Colombia, and Mexico, but local transmission has been reported in Arizona, California, Florida, Hawaii, and Texas.15 The characteristic exanthem of dengue is diffuse erythema with islands of sparing.<
Unlike during the 2022 outbreak of mpox clade II, which predominantly impacted men who have sex with men, there now is an ongoing outbreak of mpox clades 1a and 1b in the Democratic Republic of the Congo and surrounding countries that more commonly affects children and heterosexual adults. It is also more transmissible and virulent. Cases of mpox clade I have been reported in several European countries and across the United States, mostly among travelers from areas of active transmission. Vaccination of at-risk individuals is considered effective; however, tecovirimat is not.16
Outbreaks of 2 emerging zoonotic orthopoxviruses recently have been reported. Buffalopox virus (BPXV) is transmitted via direct contact with the skin of infected cattle and buffalo as well as fomites and has been responsible for human cases in South Asia. Characteristics of BPXV include macules, umbilicated papules, vesicles, pustules, and eschars that evolve over several weeks, with a predilection for the hands and face. It can manifest with prodromal symptoms of fever, malaise, and lymphadenopathy.17 Borealpox virus (formerly known as Alaskapox) has similar manifestations. Its reservoir includes small mammals such as voles and shrews, but it also has been found in cats and dogs and has been responsible for at least one human fatality. Cidofovir may be an effective therapy for both BPXV and borealpox virus, and prior smallpox vaccination may provide protection.18 These outbreaks demonstrate the continued importance of research for more effective vaccines and therapies against smallpox and other orthopoxviruses.19 A recent review provided a detailed overview of the epidemiology, transmission, dermatologic findings, and management strategies associated with smallpox and other bioweapons.20
In 2023, a case was reported of a patient in a New York City hospital with tinea that was refractory to multiple rounds of topical antifungals, which called attention to the presence of Trichophyton indotineae in the United States.21 Since then, additional reports and case series have characterized the clinical presentation of T indotineae as widespread and atypical, refractory to traditional therapies, and most often encountered in travelers returning from Bangladesh or elsewhere in South Asia.22 The diagnosis should be confirmed via DNA testing of fungal culture. Itraconazole 100 to 200 mg/d is the antifungal therapy of choice.23
Other series have reported cases of tinea genitalis caused by Trichophyton mentagrophytes type VII seen predominately in sex workers and others engaging in high-risk sexual contact, highlighting the spread of dermatophytes through sexual activity.24-26 Lastly, it is important to culture pustules and consider atypical pathogens in patients with chronic folliculitis not responding to typical therapies such as tetracycline antibiotics. A case series reported the presence of pustules in the beard area of 7 men who have sex with men, with culture data showing Klebsiella aerogenes. Prolonged courses of fluoroquinolones were necessary for clearance.27
Reducing HS Admissions Through Evidence-Based Management
Hidradenitis suppurativa is a frequent cause of emergency department visits and hospital admissions. In an analysis of the Nationwide Readmissions Database, 17.8% (392/2204) of patients admitted to the hospital with HS were readmitted within 30 days, a number comparable to that of heart failure.28
Flaring HS can produce symptoms that mimic sepsis. A retrospective cohort study examining sepsislike features in HS showed that more than 50% (30/58) of those admitted to the hospital with an HS flare were misdiagnosed with sepsis, and more than 80% (53/64) of those patients received intravenous antibiotics.29 A National Inpatient Sample (January 2016-December 2018) study demonstrated minimal rates of true infection in patients admitted with HS flares,30 while patients with HS diagnosed as sepsis do not sustain the mortality expected from true sepsis. Improving recognition of HS and differentiation of the disease from true sepsis could decrease unnecessary antibiotic use, hospital admissions, and cost, underscoring the need for a framework to reliably and reproducibly distinguish sepsis from HS flare.31
While severe HS is difficult to manage, there may be a window of opportunity in which appropriate treatment of early disease may prevent progression and decrease inpatient utilization. A prospective cohort study of 335 biologic-naïve patients with mild to moderate HS (Hurley stages I and II) followed over a median of 2 years showed that active smoking, body mass index higher than 25, and the presence of disease in 2 or more anatomic areas were factors predictive of progression to severe disease.32
Despite high utilization of emergency and inpatient care, there has been no consensus on inpatient management of HS. A Delphi consensus exercise including 26 expert dermatologists reached consensus on 40 statements.33 Specific recommendations involve multidisciplinary care, including from a dermatologist; consideration of comorbid medical conditions; supportive care measures (wound care, pain control); evidence-based medical management, including initiation or adjustment of biologic therapies; targeted surgical intervention; nutritional support and maintenance of glycemic control; and attention to transitional care at discharge, including home health services, verification of insurance status, and timely outpatient dermatology follow-up.34 A retrospective review of 98 patients treated with intravenous ertapenem for a mean duration of 13 weeks demonstrated improvement in clinical and inflammatory markers.35 Patients with severe or treatment-refractory HS, including those admitted to the hospital, may benefit from initiation of this therapy in select circumstances.
Hospital Dermatology Workforce
Inpatient dermatology consultations are extremely valuable for improving diagnostic accuracy, reducing admissions for pseudocellulitis and inflammatory skin conditions, and keeping cancer patients on needed therapies.36-38 Despite this clear value added, a cross-sectional analysis of inpatient Medicare claims data from January 2013 to December 2019 found that the number of dermatologists performing more than 10 inpatient consults per year decreased from 356 to 281.39 Additionally, medical centers in which dermatology encounters occurred decreased from 239 to 157 during the same period. Ninety-eight percent of inpatient dermatologists were in metropolitan areas, with large regions lacking access to inpatient dermatology consultation altogether.39
A survey of Society for Pediatric Dermatology members similarly characterized the state of the pediatric dermatology workforce performing hospital consultation.40 Seventy-five percent reported a high call burden, defined as more than 11 days or nights per month, more than 1 weekend per month, and/or more than 5 hours per week seeing patients. Ninety-one percent of consultation services are based within academic institutions, reflecting disparities in access.40 A prospective cohort study of academic pediatric dermatologists reported that 310 curbside consultations were performed over 24 weeks; of these calls, 17% occurred during weeknights and 23% on weekends. None of these curbside interactions was reimbursed.41 These findings underscore the burden of uncompensated time a subset of pediatric dermatologists dedicates to inpatient consultations, highlighting the need for improved financial and administrative support and an increased number of physicians performing this role.
A survey study42 suggested that unfamiliarity with the inpatient setting, rather than medical knowledge, is the most important barrier to inpatient work among clinical dermatologists. Proposed interventions include resource guides (eg, hospital maps, pager numbers for key individuals, and protocols for urgent specimens). Reference guides and refresher courses may decrease gaps in knowledge or awareness among dermatologists in ambulatory practice.42 Another way to bolster the inpatient dermatology workforce may be to provide more guidance to qualified advanced practice providers to triage and address dermatologic emergencies.43
Artificial intelligence (AI) also has been explored as a tool for diagnosing complex dermatologic conditions. One study presented 15 published inpatient dermatology cases to 7 dermatologists. Participants were asked to formulate their top 3 differential diagnoses and were then shown AI-generated differentials and asked to submit a revised differential. Participants showed a diagnostic accuracy of 69% before seeing the AI-generated differential diagnosis and 79% after; however, in cases in which the AI differential was incorrect, diagnostic accuracy of the dermatologists decreased after being shown the AI model.44
Final Thoughts
This January 2024 to December 2025 review of research relevant to hospital dermatology highlights important developments and ongoing challenges in SCARs, emerging and re-emerging infectious diseases, HS, and the inpatient dermatology workforce. Dermatologists continue to play a critical role in the care of hospitalized patients with skin disease.
Dermatologists play a central role in the care of hospitalized patients with skin disease. This review summarizes research from January 2024 to December 2025 on severe cutaneous adverse drug reactions, emerging infectious diseases, hidradenitis suppurativa (HS), and inpatient dermatology workforce issues. Key developments include improved recognition and management of drug reactions; updated diagnostic and prognostic tools for Stevens-Johnson syndrome (SJS) and toxic epidermal necrolysis (TEN); and guidance for emerging infections such as measles, dengue, mpox, orthopoxviruses, and resistant dermatophytes. Evidence-based strategies for HS aim to reduce unnecessary admissions and optimize care. Workforce challenges, including limited access, high call burden, and potential for artificial intelligence (AI)–assisted diagnosis, are also highlighted. These findings emphasize the critical contributions of dermatologists to hospital-based care and provide emerging evidence to guide clinical practice.
Dermatologists play a critical role in the care of hospitalized patients. Herein, we review the research developments between January 2024 and December 2025 most relevant to the care of hospitalized patients with skin disease, including severe cutaneous adverse reactions (SCARs), emerging and re-emerging infectious diseases, hidradenitis suppurativa (HS), and access to inpatient dermatology services.
Severe Cutaneous Adverse Drug Reactions
Severe cutaneous adverse drug reactions are among the most frequent reasons for inpatient dermatology consultation. A National Inpatient Sample study identified more than 160,000 cases of drug rash with eosinophilia and systemic symptoms (DRESS syndrome) between January 2016 and December 2020.1 The overall mortality rate was 2.0%, substantially lower than the rates of up to 10% reported in earlier studies.2 Case burden and mortality peaked during the fall months, possibly due to either increased use of antibiotics or increased viral infection or reactivation during these months.1
A retrospective cohort study of patients with probable or definite DRESS syndrome showed that, among 93 patients with at least 1 viral marker tested, human herpesvirus (HHV) reactivation was found in 42% (39/93), including HHV-6 (28%)(24/85), Epstein-Barr virus (17%)(15/87), and cytomegalovirus (20%)(18/89); furthermore, viral reactivation was associated with higher 1-year mortality (odds ratio, 3.9), dialysis initiation, flares of disease, and longer hospital stay (all P<.05).1 Multiple reactivations were associated with higher inpatient mortality and 1-year mortality; however, despite apparent prognostic importance, the role of screening for viral reactivation in DRESS syndrome is undefined.
A systematic review of 610 studies including 2122 patients with DRESS syndrome demonstrated that, among 193 causal agents identified, 14 drugs accounted for more than 1% of cases each and therefore were considered high risk. Seventy-eight percent of cases were attributed to these 14 drugs (Table).5 A TriNetX Query study analyzed antibiotic exposures across SCARs and reported that sulfonamides (hazard ratio [HR], 7.5), aminoglycosides (HR, 3.7), and tetracyclines (HR, 1.7) were associated with an elevated risk for SCARs. Sulfonamides had the highest absolute incidence of SCARs, followed by cephalosporins and penicillins.6

A multicenter randomized clinical trial7 compared high-potency topical corticosteroids (clobetasol 30 g/d) to systemic corticosteroids (prednisone 0.5 mg/kg/d) for treatment of moderate DRESS syndrome. On day 30, 53.8% (14/26) of patients in the topical group had achieved remission of visceral involvement, compared to 72.0% (18/25) in the systemic group. Before day 30, 23.1% (6/26) of patients in the topical group worsened, necessitating transition to high-dose systemic steroids. When inpatient monitoring is available, low-dose systemic corticosteroids or high-potency topical steroids may be reasonable management strategies for moderate DRESS syndrome7; however, the frequent need for treatment intensification suggests limitations to this strategy.
Since prolonged courses of systemic steroids generally are necessary for management of DRESS syndrome, steroid-sparing options are needed. A retrospective case series examined interleukin 5 inhibition in patients with possible DRESS syndrome (Registry of Severe Cutaneous Adverse Reactions score ≥3). All patients demonstrated rapid eosinophil reduction within 1 to 3 days (mean [SD] time to resolution, 1.4 [0.9] days) after treatment with mepolizumab or benralizumab, with clinical improvement occurring at a mean (SD) of 16 (3.7) days (range, 13-21 days).8
A French cohort study of 1221 adult patients with Stevens-Johnson syndrome (SJS)/toxic epidermal necrolysis (TEN) reported in-hospital mortality of 19% and a total mortality of 34% at 1 year.9 Risk factors contributing to in-hospital mortality included age, history of/current diagnosis of cancer, dementia, and liver disease, while postdischarge mortality was associated with acute kidney injury and sepsis. Long-term complications included ophthalmologic and mood disorders.9
A new set of diagnostic criteria for SJS/TEN, known as the Niigata criteria,10 includes 3 main items: severe mucosal lesions in cutaneous-mucosal transition zones (eg, eyes, lips, vulva) or generalized erythema with necrotic lesions; fever of 38.5 °C or higher; and necrosis of the epidermis seen on histopathology. Because epidermal detachment involving 10% of the body surface area (BSA) is an important mortality risk predicter, SJS is defined as less than 10% BSA involvement, and TEN has been redefined as 10% or more BSA involvement (not ≥30%). A new prognostic score—clinical risk score for TEN (CRISTEN)—can be tabulated at the point of care without laboratory values. It was developed based on the 10 most important risk factors for death in a retrospective study of 382 patients, which included age 65 years or older; epidermal detachment involving 10% BSA or higher; an antibiotic as causative agent; systemic corticosteroid therapy before the onset of SJS/TEN; involvement of all 3 mucosal surfaces; and medical comorbidities such as renal impairment, diabetes, cardiac disease, active cancer, and bacterial infection.11
New potential therapeutic targets for SJS/TEN include PC111 (monoclonal antibody to Fas ligand), formyl peptide receptor 1 antagonists (which inhibit necroptosis induced by formyl peptide receptor 1–annexin A1 interaction), daratumumab (which depletes cytotoxic CD8-positive and CD38-positive T cells), and Janus kinase (JAK) inhibitors.10 Spatial proteomics showed marked enrichment of type I and type II interferon signatures as well as activation of signal transducer and activator of transcription 1. In vitro, tofacitinib reduced keratinocyte-directed cytotoxicity, and in vivo JAK inhibitors ameliorated disease severity in 2 TEN mouse models. Patients with TEN that was refractory to corticosteroid therapy received rescue treatment with JAK inhibitors and had re-epithelization within several days with marked reduction in levels of phosphorylated signal transducer and activator of transcription 1.12 Controlled studies are needed to assess the potential role of JAK inhibitors for SJS/TEN.
Emerging and Re-emerging Infectious Diseases
Dermatologists may encounter emerging or re-emerging infections, performing an essential public health role in the process. In 2025, a total of 2281 confirmed cases of measles had been reported across 45 of the United States.13 During the COVID-19 pandemic, measles vaccine coverage in the United States dropped to 93%—down from 95% to 97% prepandemic. Worldwide, 2022 saw an increase of 1.4 million measles cases (18% increase) and 41,200 excess deaths (43% increase) compared to the previous year. Complications of measles include pneumonia, blindness, otitis media, and encephalitis, with 1 in 5 (20%) unvaccinated people with measles in the United States requiring hospitalization.14 A vaccine coverage rate higher than 95% is needed to prevent community spread of disease. Since efforts to detect and rapidly isolate cases of measles are critical, dermatologists should consider measles in the differential of morbilliform eruptions with viral symptoms and ask about vaccination status.
Since 2023, dengue infection rates have tripled in the Americas, representing the highest levels recorded since tracking began in 1980. In 2024, there were more than 12 million cases, with approximately 8000 deaths reported. Ninety percent of cases occur in Argentina, Brazil, Colombia, and Mexico, but local transmission has been reported in Arizona, California, Florida, Hawaii, and Texas.15 The characteristic exanthem of dengue is diffuse erythema with islands of sparing.<
Unlike during the 2022 outbreak of mpox clade II, which predominantly impacted men who have sex with men, there now is an ongoing outbreak of mpox clades 1a and 1b in the Democratic Republic of the Congo and surrounding countries that more commonly affects children and heterosexual adults. It is also more transmissible and virulent. Cases of mpox clade I have been reported in several European countries and across the United States, mostly among travelers from areas of active transmission. Vaccination of at-risk individuals is considered effective; however, tecovirimat is not.16
Outbreaks of 2 emerging zoonotic orthopoxviruses recently have been reported. Buffalopox virus (BPXV) is transmitted via direct contact with the skin of infected cattle and buffalo as well as fomites and has been responsible for human cases in South Asia. Characteristics of BPXV include macules, umbilicated papules, vesicles, pustules, and eschars that evolve over several weeks, with a predilection for the hands and face. It can manifest with prodromal symptoms of fever, malaise, and lymphadenopathy.17 Borealpox virus (formerly known as Alaskapox) has similar manifestations. Its reservoir includes small mammals such as voles and shrews, but it also has been found in cats and dogs and has been responsible for at least one human fatality. Cidofovir may be an effective therapy for both BPXV and borealpox virus, and prior smallpox vaccination may provide protection.18 These outbreaks demonstrate the continued importance of research for more effective vaccines and therapies against smallpox and other orthopoxviruses.19 A recent review provided a detailed overview of the epidemiology, transmission, dermatologic findings, and management strategies associated with smallpox and other bioweapons.20
In 2023, a case was reported of a patient in a New York City hospital with tinea that was refractory to multiple rounds of topical antifungals, which called attention to the presence of Trichophyton indotineae in the United States.21 Since then, additional reports and case series have characterized the clinical presentation of T indotineae as widespread and atypical, refractory to traditional therapies, and most often encountered in travelers returning from Bangladesh or elsewhere in South Asia.22 The diagnosis should be confirmed via DNA testing of fungal culture. Itraconazole 100 to 200 mg/d is the antifungal therapy of choice.23
Other series have reported cases of tinea genitalis caused by Trichophyton mentagrophytes type VII seen predominately in sex workers and others engaging in high-risk sexual contact, highlighting the spread of dermatophytes through sexual activity.24-26 Lastly, it is important to culture pustules and consider atypical pathogens in patients with chronic folliculitis not responding to typical therapies such as tetracycline antibiotics. A case series reported the presence of pustules in the beard area of 7 men who have sex with men, with culture data showing Klebsiella aerogenes. Prolonged courses of fluoroquinolones were necessary for clearance.27
Reducing HS Admissions Through Evidence-Based Management
Hidradenitis suppurativa is a frequent cause of emergency department visits and hospital admissions. In an analysis of the Nationwide Readmissions Database, 17.8% (392/2204) of patients admitted to the hospital with HS were readmitted within 30 days, a number comparable to that of heart failure.28
Flaring HS can produce symptoms that mimic sepsis. A retrospective cohort study examining sepsislike features in HS showed that more than 50% (30/58) of those admitted to the hospital with an HS flare were misdiagnosed with sepsis, and more than 80% (53/64) of those patients received intravenous antibiotics.29 A National Inpatient Sample (January 2016-December 2018) study demonstrated minimal rates of true infection in patients admitted with HS flares,30 while patients with HS diagnosed as sepsis do not sustain the mortality expected from true sepsis. Improving recognition of HS and differentiation of the disease from true sepsis could decrease unnecessary antibiotic use, hospital admissions, and cost, underscoring the need for a framework to reliably and reproducibly distinguish sepsis from HS flare.31
While severe HS is difficult to manage, there may be a window of opportunity in which appropriate treatment of early disease may prevent progression and decrease inpatient utilization. A prospective cohort study of 335 biologic-naïve patients with mild to moderate HS (Hurley stages I and II) followed over a median of 2 years showed that active smoking, body mass index higher than 25, and the presence of disease in 2 or more anatomic areas were factors predictive of progression to severe disease.32
Despite high utilization of emergency and inpatient care, there has been no consensus on inpatient management of HS. A Delphi consensus exercise including 26 expert dermatologists reached consensus on 40 statements.33 Specific recommendations involve multidisciplinary care, including from a dermatologist; consideration of comorbid medical conditions; supportive care measures (wound care, pain control); evidence-based medical management, including initiation or adjustment of biologic therapies; targeted surgical intervention; nutritional support and maintenance of glycemic control; and attention to transitional care at discharge, including home health services, verification of insurance status, and timely outpatient dermatology follow-up.34 A retrospective review of 98 patients treated with intravenous ertapenem for a mean duration of 13 weeks demonstrated improvement in clinical and inflammatory markers.35 Patients with severe or treatment-refractory HS, including those admitted to the hospital, may benefit from initiation of this therapy in select circumstances.
Hospital Dermatology Workforce
Inpatient dermatology consultations are extremely valuable for improving diagnostic accuracy, reducing admissions for pseudocellulitis and inflammatory skin conditions, and keeping cancer patients on needed therapies.36-38 Despite this clear value added, a cross-sectional analysis of inpatient Medicare claims data from January 2013 to December 2019 found that the number of dermatologists performing more than 10 inpatient consults per year decreased from 356 to 281.39 Additionally, medical centers in which dermatology encounters occurred decreased from 239 to 157 during the same period. Ninety-eight percent of inpatient dermatologists were in metropolitan areas, with large regions lacking access to inpatient dermatology consultation altogether.39
A survey of Society for Pediatric Dermatology members similarly characterized the state of the pediatric dermatology workforce performing hospital consultation.40 Seventy-five percent reported a high call burden, defined as more than 11 days or nights per month, more than 1 weekend per month, and/or more than 5 hours per week seeing patients. Ninety-one percent of consultation services are based within academic institutions, reflecting disparities in access.40 A prospective cohort study of academic pediatric dermatologists reported that 310 curbside consultations were performed over 24 weeks; of these calls, 17% occurred during weeknights and 23% on weekends. None of these curbside interactions was reimbursed.41 These findings underscore the burden of uncompensated time a subset of pediatric dermatologists dedicates to inpatient consultations, highlighting the need for improved financial and administrative support and an increased number of physicians performing this role.
A survey study42 suggested that unfamiliarity with the inpatient setting, rather than medical knowledge, is the most important barrier to inpatient work among clinical dermatologists. Proposed interventions include resource guides (eg, hospital maps, pager numbers for key individuals, and protocols for urgent specimens). Reference guides and refresher courses may decrease gaps in knowledge or awareness among dermatologists in ambulatory practice.42 Another way to bolster the inpatient dermatology workforce may be to provide more guidance to qualified advanced practice providers to triage and address dermatologic emergencies.43
Artificial intelligence (AI) also has been explored as a tool for diagnosing complex dermatologic conditions. One study presented 15 published inpatient dermatology cases to 7 dermatologists. Participants were asked to formulate their top 3 differential diagnoses and were then shown AI-generated differentials and asked to submit a revised differential. Participants showed a diagnostic accuracy of 69% before seeing the AI-generated differential diagnosis and 79% after; however, in cases in which the AI differential was incorrect, diagnostic accuracy of the dermatologists decreased after being shown the AI model.44
Final Thoughts
This January 2024 to December 2025 review of research relevant to hospital dermatology highlights important developments and ongoing challenges in SCARs, emerging and re-emerging infectious diseases, HS, and the inpatient dermatology workforce. Dermatologists continue to play a critical role in the care of hospitalized patients with skin disease.
- Desai AD, Thomas C. Seasonal trends in drug reaction with eosinophilia and systemic symptoms. J Am Acad Dermatol. 2025;92:183-185.
- Wei BM, Fox LP, Kaffenberger BH, et al. Drug-induced hypersensitivity syndrome/drug reaction with eosinophilia and systemic symptoms. Part I. Epidemiology, pathogenesis, clinicopathological features, and prognosis. J Am Acad Dermatol. 2024;90:885-908. doi:10.1016/j.jaad.2023.02.072
- Chan LCE, Sultana R, Choo KJL, et al. Viral reactivation and clinical outcomes in drug reaction with eosinophilia and systemic symptoms (DRESS). Sci Rep. 2024;14:28492.
- Brüggen MC, Walsh S, Ameri MM, et al. Management of adult patients with drug reaction with eosinophilia and systemic symptoms: a Delphi-based international consensus. JAMA Dermatol. 2024;160:37-44
- Hansen E, Gallardo M, Yan A, et al. Risk assessment of drugs associated with DRESS syndrome based on publication frequency: a systematic review. J Am Acad Dermatol. 2024;91:962-966.
- Neubauer ZJK, Chan R, Singal A, et al. SCAR-ed by antibiotics: a retrospective cohort study of severe cutaneous adverse reactions (SCAR) relative risk. J Am Acad Dermatol. 2025;92:1143-1145.
- Ingen-Housz-Oro S, Guichard E, Milpied B, et al. Topical versus oral corticosteroids in moderate drug reaction with eosinophilia and systemic symptoms: a multicenter randomized clinical trial. J Am Acad Dermatol. 2024;91:544-547.
- Hijaz B, Nambudiri VE, Imadojemu S. IL-5 inhibitor treatment in drug reaction with eosinophilia and systemic symptoms. JAMA Dermatol. 2025;161:661-663.
- Bettuzzi T, Lebrun-Vignes B, Ingen-Housz-Oro S, et al. Incidence, in-hospital and long-term mortality, and sequelae of epidermal necrolysis in adults. JAMA Dermatol. 2024;160:1288-1296.
- Hama N, Aoki S, Chen CB, et al. Recent progress in Stevens-Johnson syndrome/toxic epidermal necrolysis: diagnostic criteria, pathogenesis and treatment. Br J Dermatol. 2024;192:9-18.
- Hama N, Sunaga Y, Ochiai H, et al. Development and validation of a novel score to predict mortality in Stevens-Johnson syndrome and toxic epidermal necrolysis: CRISTEN. J Allergy Clin Immunol Pract. 2023;11:3161-3168.e2.
- Nordmann TM, Anderton H, Hasegawa A, et al. Spatial proteomics identifies JAKi as treatment for a lethal skin disease. Nature. 2024;635:1001-1009.
- Centers for Disease Control and Prevention. Measles cases and outbreaks. Updated January 7, 2026. Accessed January 12, 2026. https://www.cdc.gov/measles/data-research/
- Rubin R. Despite safe and effective vaccine, measles cases and deaths increased worldwide from 2021 to 2022. JAMA. 2024;331:188-189.
- Orrall A. Dengue cases in the Americas highest recorded. JAMA. 2025;333:452.
- Harris E. As mpox cases surge in Africa, WHO declares a global emergency-here’s what to know. JAMA. 2024;332:862-864.
- Burningham KM, Hinojosa T, Cavazos A, et al. Buffalopox: an emerging cutaneous disease in humans. J Eur Acad Dermatol Venereol. 2025;39:404-406.
- Parker ER. Emergence of Alaskapox infection: what dermatologists need to know. J Am Acad Dermatol. 2024;91:397-399.
- Gostin LO, Singaravelu S, Hynes N. Smallpox readiness: modern strategies against an ancient disease. JAMA. 2024;332:873-874.
- Osborne S, Kam O, Thacker S, et al. Review of category A bioweapons with cutaneous features: epidemiology, clinical presentation, and contemporary management strategies. J Am Acad Dermatol. 2025;93:165-175.
- Caplan AS, Chaturvedi S, Zhu Y, et al. Notes from the field: first reported U.S. cases of tinea caused by Trichophyton indotineae - New York City, December 2021-March 2023. MMWR Morb Mortal Wkly Rep. 2023;72:536-537.
- McKenna M. Why the rise of this drug-resistant fungus is raising international concern. JAMA. 2024;332:859-861.
- Caplan AS, Todd GC, Zhu Y, et al. Clinical course, antifungal susceptibility, and genomic sequencing of Trichophyton indotineae. JAMA Dermatol. 2024;160:701-709.
- Jabet A, Bérot V, Chiarabini T, et al. Trichophyton mentagrophytes ITS genotype VII infections among men who have sex with men in France: an ongoing phenomenon. J Eur Acad Dermatol Venereol. 2025;39:407-415.
- Luchsinger I, Bosshard PP, Kasper RS, et al. Tinea genitalis: a new entity of sexually transmitted infection? Case series and review of the literature. Sex Transm Infect. 2015;91:493-496.
- Khurana A, Sharath S, Sardana K, et al. Therapeutic updates on the management of tinea corporis or cruris in the era of Trichophyton indotineae: separating evidence from hype-a narrative review. Indian J Dermatol. 2023;68:525-540.
- Bérot V, Monsel G, Dauendorffer JN, et al; Groupe Infectiologie Dermatologique et Infections Sexuellement Transmissibles (GrIDIST) de la Société Française de Dermatologie. Klebsiella aerogenes-related facial folliculitis in men having sex with men: a hypothetical new STI?J Eur Acad Dermatol Venereol. 2025;39:E10-E12.
- Edigin E, Kaul S, Eseaton PO, et al. At 180 days hidradenitis suppurativa readmission rate is comparable to heart failure: analysis of the Nationwide Readmissions Database. J Am Acad Dermatol. 2022;87:188-192.
- AbdelHameid D, Wang L, Mauskar MM, et al. Sepsis-like features in hidradenitis suppurativa flares requiring admission: a retrospective cohort study. J Am Acad Dermatol. 2024;90:1291-1294.
- Ehizogie E, Maghari I, Lo S, et al. Hidradenitis suppurativa, systemic inflammatory response syndrome and sepsis: a database study. Br J Dermatol. 2024;191:451-453.
- Maghari I, Abiad H, Griffin T, et al. Hidradenitis suppurativa (HS), systemic inflammatory response syndrome and sepsis, sepsis caused by HS: an empty systematic review. Br J Dermatol. 2024;191:449-450.
- Kjærsgaard Andersen R, Pedersen O, Eidsmo L, et al. Initial steps towards developing a predictive algorithm of disease progression for hidradenitis suppurativa (HS): results from a Cox proportional hazard regression analysis on disease progression among a cohort of 335 Danish patients with HS. Br J Dermatol. 2024;190:904-914.
- Needham M, Pichardo R, Alavi A, et al. Inpatient management of hidradenitis suppurativa: a Delphi consensus study. Cutis. 2024;113:251-254.
- Maskan Bermudez N, Elman SA, Kirsner RS, et al. Management of hidradenitis suppurativa in the inpatient setting: a clinical guide. Arch Dermatol Res. 2025;317:202.
- Nosrati A, Ch’en PY, Torpey ME, et al. Efficacy and durability of intravenous ertapenem therapy for recalcitrant hidradenitis suppurativa. JAMA Dermatol. 2024;160:312-318.
- Tracey EH, Forrestel A, Rosenbach M, et al. Inpatient dermatology consultation in patients with hematologic malignancies. J Am Acad Dermatol. 2016;75:835-836.
- Li DG, Xia FD, Khosravi H, et al. Outcomes of early dermatology consultation for inpatients diagnosed with cellulitis. JAMA Dermatol. 2018;154:537-543.
- Jacoby TV, Shah N, Asdourian MS, et al. Dermatology evaluation for cutaneous immune-related adverse events is associated with improved survival in cancer patients treated with checkpoint inhibition. J Am Acad Dermatol. 2023;88:711-714.
- Hydol-Smith JA, Gallardo MA, Korman A, et al. The United States dermatology inpatient workforce between 2013 and 2019: a Medicare analysis reveals contraction of the workforce and vast access deserts-a cross-sectional analysis. Arch Dermatol Res. 2024;316:103.
- Pineider JL, Rangu SA, Shaw KS, et al. Pediatric consultative dermatology: a survey of the Society for Pediatric Dermatology workforce reveals shortcomings in existing practice models of pediatric dermatology consult services in the United States. Pediatr Dermatol. 2024;41:270-274.
- Puar NK, Canty KM, Newell BD, et al. An evaluation of pediatric dermatology curbside consultations in an academic center: a prospective cohort study. J Am Acad Dermatol. 2024;90:1258-1260.
- Lau CB, Smith GP. Strategies for improving dermatologist comfort and quality of patient care in inpatient settings: a cross-sectional survey study. Arch Dermatol Res. 2024;316:575.
- Hazim AH. Empowering advanced clinical practitioners in managing acute dermatological emergencies. Br J Nurs. 2024;33:448-455.
- Macklis P, Kaffenberger B, Kirven R, et al. Dermatology diagnostic accuracy is improved by artificial intelligence-generated differential diagnoses. Int J Dermatol. 2025;64:960-962.
- Desai AD, Thomas C. Seasonal trends in drug reaction with eosinophilia and systemic symptoms. J Am Acad Dermatol. 2025;92:183-185.
- Wei BM, Fox LP, Kaffenberger BH, et al. Drug-induced hypersensitivity syndrome/drug reaction with eosinophilia and systemic symptoms. Part I. Epidemiology, pathogenesis, clinicopathological features, and prognosis. J Am Acad Dermatol. 2024;90:885-908. doi:10.1016/j.jaad.2023.02.072
- Chan LCE, Sultana R, Choo KJL, et al. Viral reactivation and clinical outcomes in drug reaction with eosinophilia and systemic symptoms (DRESS). Sci Rep. 2024;14:28492.
- Brüggen MC, Walsh S, Ameri MM, et al. Management of adult patients with drug reaction with eosinophilia and systemic symptoms: a Delphi-based international consensus. JAMA Dermatol. 2024;160:37-44
- Hansen E, Gallardo M, Yan A, et al. Risk assessment of drugs associated with DRESS syndrome based on publication frequency: a systematic review. J Am Acad Dermatol. 2024;91:962-966.
- Neubauer ZJK, Chan R, Singal A, et al. SCAR-ed by antibiotics: a retrospective cohort study of severe cutaneous adverse reactions (SCAR) relative risk. J Am Acad Dermatol. 2025;92:1143-1145.
- Ingen-Housz-Oro S, Guichard E, Milpied B, et al. Topical versus oral corticosteroids in moderate drug reaction with eosinophilia and systemic symptoms: a multicenter randomized clinical trial. J Am Acad Dermatol. 2024;91:544-547.
- Hijaz B, Nambudiri VE, Imadojemu S. IL-5 inhibitor treatment in drug reaction with eosinophilia and systemic symptoms. JAMA Dermatol. 2025;161:661-663.
- Bettuzzi T, Lebrun-Vignes B, Ingen-Housz-Oro S, et al. Incidence, in-hospital and long-term mortality, and sequelae of epidermal necrolysis in adults. JAMA Dermatol. 2024;160:1288-1296.
- Hama N, Aoki S, Chen CB, et al. Recent progress in Stevens-Johnson syndrome/toxic epidermal necrolysis: diagnostic criteria, pathogenesis and treatment. Br J Dermatol. 2024;192:9-18.
- Hama N, Sunaga Y, Ochiai H, et al. Development and validation of a novel score to predict mortality in Stevens-Johnson syndrome and toxic epidermal necrolysis: CRISTEN. J Allergy Clin Immunol Pract. 2023;11:3161-3168.e2.
- Nordmann TM, Anderton H, Hasegawa A, et al. Spatial proteomics identifies JAKi as treatment for a lethal skin disease. Nature. 2024;635:1001-1009.
- Centers for Disease Control and Prevention. Measles cases and outbreaks. Updated January 7, 2026. Accessed January 12, 2026. https://www.cdc.gov/measles/data-research/
- Rubin R. Despite safe and effective vaccine, measles cases and deaths increased worldwide from 2021 to 2022. JAMA. 2024;331:188-189.
- Orrall A. Dengue cases in the Americas highest recorded. JAMA. 2025;333:452.
- Harris E. As mpox cases surge in Africa, WHO declares a global emergency-here’s what to know. JAMA. 2024;332:862-864.
- Burningham KM, Hinojosa T, Cavazos A, et al. Buffalopox: an emerging cutaneous disease in humans. J Eur Acad Dermatol Venereol. 2025;39:404-406.
- Parker ER. Emergence of Alaskapox infection: what dermatologists need to know. J Am Acad Dermatol. 2024;91:397-399.
- Gostin LO, Singaravelu S, Hynes N. Smallpox readiness: modern strategies against an ancient disease. JAMA. 2024;332:873-874.
- Osborne S, Kam O, Thacker S, et al. Review of category A bioweapons with cutaneous features: epidemiology, clinical presentation, and contemporary management strategies. J Am Acad Dermatol. 2025;93:165-175.
- Caplan AS, Chaturvedi S, Zhu Y, et al. Notes from the field: first reported U.S. cases of tinea caused by Trichophyton indotineae - New York City, December 2021-March 2023. MMWR Morb Mortal Wkly Rep. 2023;72:536-537.
- McKenna M. Why the rise of this drug-resistant fungus is raising international concern. JAMA. 2024;332:859-861.
- Caplan AS, Todd GC, Zhu Y, et al. Clinical course, antifungal susceptibility, and genomic sequencing of Trichophyton indotineae. JAMA Dermatol. 2024;160:701-709.
- Jabet A, Bérot V, Chiarabini T, et al. Trichophyton mentagrophytes ITS genotype VII infections among men who have sex with men in France: an ongoing phenomenon. J Eur Acad Dermatol Venereol. 2025;39:407-415.
- Luchsinger I, Bosshard PP, Kasper RS, et al. Tinea genitalis: a new entity of sexually transmitted infection? Case series and review of the literature. Sex Transm Infect. 2015;91:493-496.
- Khurana A, Sharath S, Sardana K, et al. Therapeutic updates on the management of tinea corporis or cruris in the era of Trichophyton indotineae: separating evidence from hype-a narrative review. Indian J Dermatol. 2023;68:525-540.
- Bérot V, Monsel G, Dauendorffer JN, et al; Groupe Infectiologie Dermatologique et Infections Sexuellement Transmissibles (GrIDIST) de la Société Française de Dermatologie. Klebsiella aerogenes-related facial folliculitis in men having sex with men: a hypothetical new STI?J Eur Acad Dermatol Venereol. 2025;39:E10-E12.
- Edigin E, Kaul S, Eseaton PO, et al. At 180 days hidradenitis suppurativa readmission rate is comparable to heart failure: analysis of the Nationwide Readmissions Database. J Am Acad Dermatol. 2022;87:188-192.
- AbdelHameid D, Wang L, Mauskar MM, et al. Sepsis-like features in hidradenitis suppurativa flares requiring admission: a retrospective cohort study. J Am Acad Dermatol. 2024;90:1291-1294.
- Ehizogie E, Maghari I, Lo S, et al. Hidradenitis suppurativa, systemic inflammatory response syndrome and sepsis: a database study. Br J Dermatol. 2024;191:451-453.
- Maghari I, Abiad H, Griffin T, et al. Hidradenitis suppurativa (HS), systemic inflammatory response syndrome and sepsis, sepsis caused by HS: an empty systematic review. Br J Dermatol. 2024;191:449-450.
- Kjærsgaard Andersen R, Pedersen O, Eidsmo L, et al. Initial steps towards developing a predictive algorithm of disease progression for hidradenitis suppurativa (HS): results from a Cox proportional hazard regression analysis on disease progression among a cohort of 335 Danish patients with HS. Br J Dermatol. 2024;190:904-914.
- Needham M, Pichardo R, Alavi A, et al. Inpatient management of hidradenitis suppurativa: a Delphi consensus study. Cutis. 2024;113:251-254.
- Maskan Bermudez N, Elman SA, Kirsner RS, et al. Management of hidradenitis suppurativa in the inpatient setting: a clinical guide. Arch Dermatol Res. 2025;317:202.
- Nosrati A, Ch’en PY, Torpey ME, et al. Efficacy and durability of intravenous ertapenem therapy for recalcitrant hidradenitis suppurativa. JAMA Dermatol. 2024;160:312-318.
- Tracey EH, Forrestel A, Rosenbach M, et al. Inpatient dermatology consultation in patients with hematologic malignancies. J Am Acad Dermatol. 2016;75:835-836.
- Li DG, Xia FD, Khosravi H, et al. Outcomes of early dermatology consultation for inpatients diagnosed with cellulitis. JAMA Dermatol. 2018;154:537-543.
- Jacoby TV, Shah N, Asdourian MS, et al. Dermatology evaluation for cutaneous immune-related adverse events is associated with improved survival in cancer patients treated with checkpoint inhibition. J Am Acad Dermatol. 2023;88:711-714.
- Hydol-Smith JA, Gallardo MA, Korman A, et al. The United States dermatology inpatient workforce between 2013 and 2019: a Medicare analysis reveals contraction of the workforce and vast access deserts-a cross-sectional analysis. Arch Dermatol Res. 2024;316:103.
- Pineider JL, Rangu SA, Shaw KS, et al. Pediatric consultative dermatology: a survey of the Society for Pediatric Dermatology workforce reveals shortcomings in existing practice models of pediatric dermatology consult services in the United States. Pediatr Dermatol. 2024;41:270-274.
- Puar NK, Canty KM, Newell BD, et al. An evaluation of pediatric dermatology curbside consultations in an academic center: a prospective cohort study. J Am Acad Dermatol. 2024;90:1258-1260.
- Lau CB, Smith GP. Strategies for improving dermatologist comfort and quality of patient care in inpatient settings: a cross-sectional survey study. Arch Dermatol Res. 2024;316:575.
- Hazim AH. Empowering advanced clinical practitioners in managing acute dermatological emergencies. Br J Nurs. 2024;33:448-455.
- Macklis P, Kaffenberger B, Kirven R, et al. Dermatology diagnostic accuracy is improved by artificial intelligence-generated differential diagnoses. Int J Dermatol. 2025;64:960-962.
Hospital Dermatology: Review of Research in 2024-2025
Hospital Dermatology: Review of Research in 2024-2025
Practice Points
- In suspected drug reaction with eosinophilia and systemic symptoms, discontinue the offending drug; test for human herpesvirus 6, Epstein-Barr virus, and cytomegalovirus when available; and treat moderate cases with low-dose corticosteroids. Reserve interleukin 5 inhibitors for refractory disease.
- For Stevens-Johnson syndrome and toxic epidermal necrolysis (TEN), apply Niigata diagnostic criteria and clinical risk score for TEN, refer patients with 10% or more body surface area detachment to higher-level or burn care, and consider targeted therapies for refractory cases.
- When assessing infectious rashes, consider measles, dengue, mpox, orthopoxviruses, and resistant dermatophytes. Review the patient’s vaccination and travel history, isolate suspected measles cases, and confirm atypical tinea with culture or DNA testing.
- To reduce unnecessary admissions and optimize care for hidradenitis suppurativa, avoid misdiagnosing flares as sepsis, implement multidisciplinary protocols, consider selective intravenous antibiotics, and support expanded inpatient dermatology coverage.
Phototherapy for Pediatric Segmental Vitiligo
Phototherapy for Pediatric Segmental Vitiligo
To the Editor:
Segmental vitiligo (SV) accounts for a minority of vitiligo cases and most frequently occurs in children.1 It characteristically manifests unilaterally and affects a single body area with a sharp midline demarcation. In contrast to nonsegmental vitiligo (NSV), SV typically stabilizes early in the disease progression.1 The pathophysiology of this vitiligo subtype is not well established, but possible autoinflammatory mechanisms associated with somatic mosaicism, neuronal mechanisms, and/or microvascular skin-homing have been proposed.2 We present the case of a pediatric patient with segmental vitiligo of the right hemiface treated with a combination of a topical calcineurin inhibitor and narrow-band UVB (NB-UVB) phototherapy.
An otherwise healthy 7-year-old boy presented to the dermatology department for evaluation of depigmented macules and patches affecting the right hemiface (temporal, periorbital, malar, perioral, preauricular, and mandibular regions) and neck associated with homolateral leukotrichia of the scalp and facial hair as well as the eyelashes of 5 years’ duration. The findings were consistent with SV (Figure 1). The patient previously had been diagnosed based on the clinical findings and treated with continuous application of topical calcineurin inhibitors plus oral cyclosporine (3 mg/kg/d) for 1 year, but the response was poor. The condition had a severe impact on the patient’s quality of life and social relationships. Therapeutic options were discussed with the patient’s caregivers, and ultimately NB-UVB phototherapy was started twice weekly with 10% increases in the dose at each treatment. Topical tacrolimus ointment (1 mg/g) also was started, and the cyclosporine was stopped. Evaluation of treatment progress occurred every 3 months, with progressive repigmentation of the patches following a perifollicular pattern. After 6 months of phototherapy, there was notable repigmentation of the affected areas, particularly in the malar, perioral, and perinasal regions (Figure 2) and the therapeutic response improved after 1 year of treatment (Figure 3). No adverse events were noted during the treatment period.
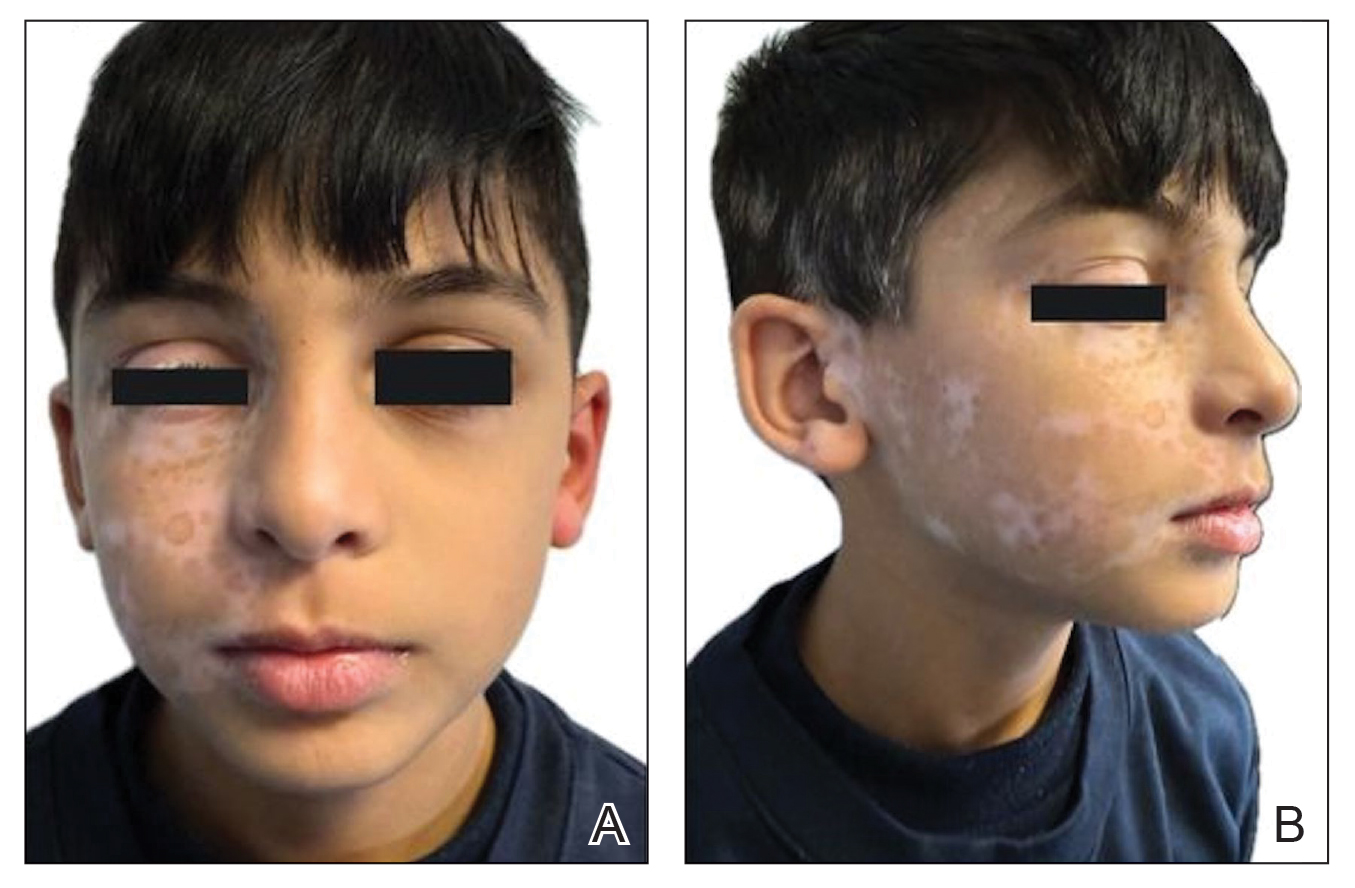
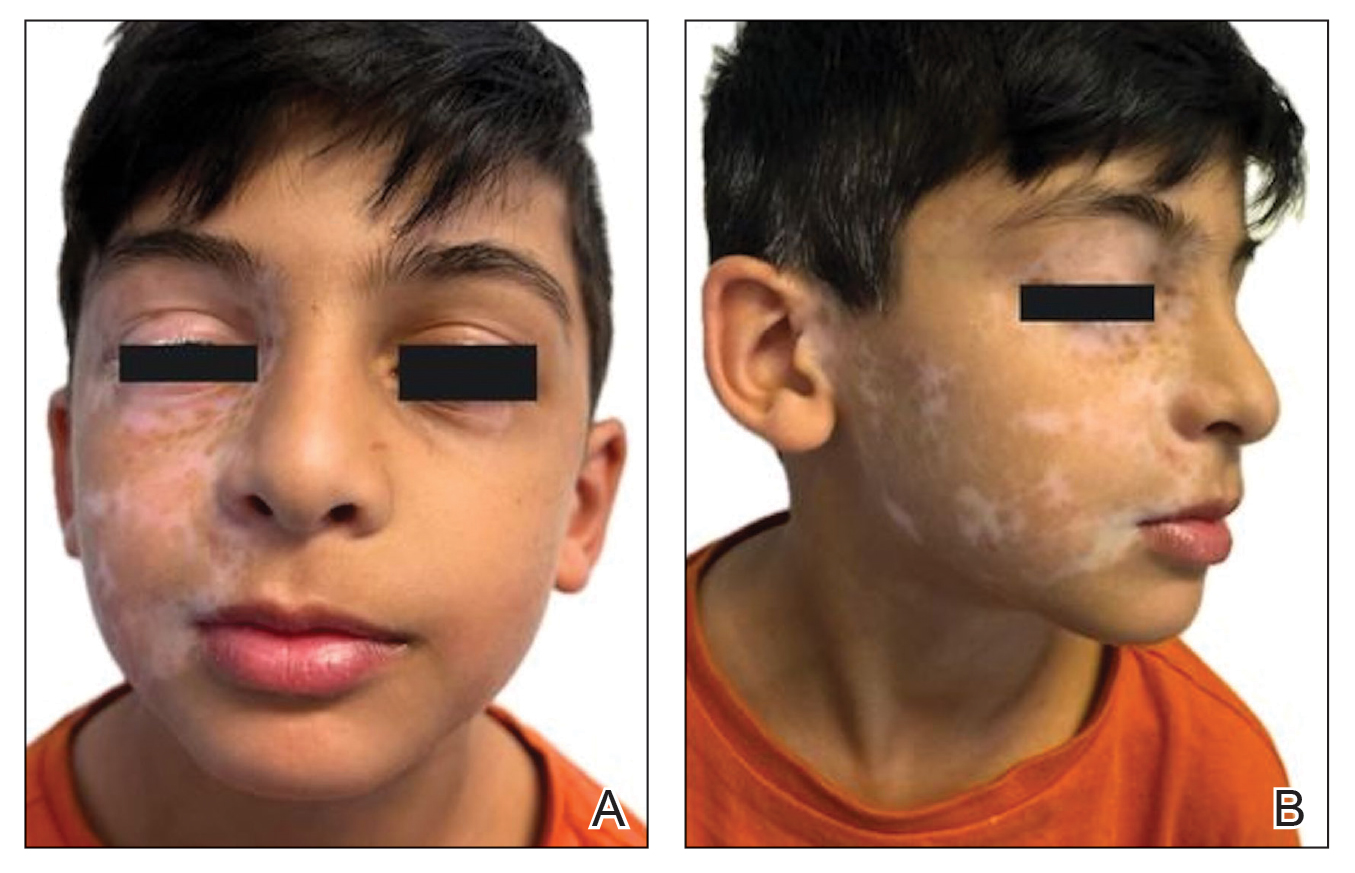
Segmental vitiligo lacks consistently effective treatment options. This subtype of vitiligo is classically resistant to conventional therapeutic options. Surgery may be a more effective and long-lasting treatment option but is not suitable for every patient.1,3 Janus kinase (JAK) inhibitors are the newest treatment options being explored for topical and systemic treatment of vitiligo, with promising results in active and stable NSV lesions4,5; however, SV rarely is represented in case reports and clinical trials. The topical JAK inhibitor ruxolitinib has been approved for use in NSV,5 and a phase 2 trial with oral ritlecitinib only included patients with NSV.4 Furthermore, JAK inhibitors have been studied and approved for children aged 12 years or older as well as for adults,4,5 but younger age groups (4-10 years)—in whom SV most frequently manifests, as in our patient—have been excluded from these studies.1 We present a novel case of SV of the right hemiface in a child that was successfully treated with NB-UVB phototherapy in association with topical calcineurin inhibitors.
The role of phototherapy for the treatment of vitiligo has been well documented, and it frequently is combined with other therapeutic modalities, such as topical anti-inflammatory drugs or, most recently, laser and micrografting techniques.6,7 The most frequently used modality is NB-UVB. In the active phase, it performs an immunomodulatory role, while in the stable phase, it stimulates migration and activity of perilesional and hair follicle melanocytes.8 Initiating therapy early is advisable, particularly during the first 6 months of progression, as there is a higher probability of response1,3,8; nevertheless, a good response was achieved despite the 5-year evolution of vitiligo in our patient. This is a safe option for a skin condition that may begin early in life and require long-term treatment.8 A main concern would be an increased risk for skin cancer associated with repeated NB-UVB exposure, which has not been verified in a recent analysis.9
Segmental vitiligo can considerably impact the patient’s quality of life, affecting social interactions and self-perception, particularly in younger patients with facial involvement; thus, effective and safe therapeutic strategies adapted to the individual and their vitiligo lesions should be discussed. Classical treatment options remain valid and provide good results for some patients; therefore, they should not be disregarded even with the rise of innovative therapies.
- Speeckaert R, Lambert J, Bulat V, et al. Autoimmunity in segmental vitiligo. Front Immunol. 2020;11:568447. doi:10.3389/fimmu.2020.568447
- Lin X, Meng X, Lin J. Segmental vitiligo: autoimmune pathogenesis, neuronal mechanisms, and somatic mosaicism. Int J Dermatol. 2025;64:490-498. doi:10.1111/ijd.17627
- Khalili M, Amiri R, Mohammadi S, et al. Efficacy and safety of traditional and surgical treatment modalities in segmental vitiligo: a review article. J Cosmet Dermatol. 2022;21:2360-2373. doi:10.1111/jocd.14899
- Yamaguchi Y, Peeva E, Del Duca E, et al. Ritlecitinib, a JAK3/TEC family kinase inhibitor, stabilizes active lesions and repigments stable lesions in vitiligo. Arch Dermatol Res. 2024;316:478. doi:10.1007/s00403-024-03182-y
- Rosmarin D, Passeron T, Pandya AG, et al. Two phase 3, randomized, controlled trials of ruxolitinib cream for vitiligo. N Engl J Med. 2022;387:1445-1455. doi:10.1056/NEJMoa2118828
- Chavez-Alvarez S, Herz-Ruelas M, Ocampo-Candiani J, et al. Stable segmental vitiligo treated with punch mini-grafts and narrow band UVB phototherapy. Australas J Dermatol. 2020;61:83-85. doi:10.1111/ajd.13105
- Kim WI, Kim S, Lee SH, et al. The efficacy of fractional carbon dioxide laser combined with narrow-band ultraviolet B phototherapy for non-segmental vitiligo: a systematic review and meta-analysis. Lasers Med Sci. 2021;36:165-173. doi:10.1007/s10103-020-03069-0
- Esmat S, Hegazy RA, Shalaby S, et al. Phototherapy and combination therapies for vitiligo. Dermatol Clin. 2017;35:171-192. doi:10.1016/j.det.2016.11.008
- Mimouni I, Shulman J, Unes AA, et al. Frequency of skin cancer among psoriasis, vitiligo, and mycosis fungoides patients treated with narrowband ultraviolet B phototherapy. Photodermatol Photoimmunol Photomed. 2024;40:E12936. doi:10.1111/phpp.12936
To the Editor:
Segmental vitiligo (SV) accounts for a minority of vitiligo cases and most frequently occurs in children.1 It characteristically manifests unilaterally and affects a single body area with a sharp midline demarcation. In contrast to nonsegmental vitiligo (NSV), SV typically stabilizes early in the disease progression.1 The pathophysiology of this vitiligo subtype is not well established, but possible autoinflammatory mechanisms associated with somatic mosaicism, neuronal mechanisms, and/or microvascular skin-homing have been proposed.2 We present the case of a pediatric patient with segmental vitiligo of the right hemiface treated with a combination of a topical calcineurin inhibitor and narrow-band UVB (NB-UVB) phototherapy.
An otherwise healthy 7-year-old boy presented to the dermatology department for evaluation of depigmented macules and patches affecting the right hemiface (temporal, periorbital, malar, perioral, preauricular, and mandibular regions) and neck associated with homolateral leukotrichia of the scalp and facial hair as well as the eyelashes of 5 years’ duration. The findings were consistent with SV (Figure 1). The patient previously had been diagnosed based on the clinical findings and treated with continuous application of topical calcineurin inhibitors plus oral cyclosporine (3 mg/kg/d) for 1 year, but the response was poor. The condition had a severe impact on the patient’s quality of life and social relationships. Therapeutic options were discussed with the patient’s caregivers, and ultimately NB-UVB phototherapy was started twice weekly with 10% increases in the dose at each treatment. Topical tacrolimus ointment (1 mg/g) also was started, and the cyclosporine was stopped. Evaluation of treatment progress occurred every 3 months, with progressive repigmentation of the patches following a perifollicular pattern. After 6 months of phototherapy, there was notable repigmentation of the affected areas, particularly in the malar, perioral, and perinasal regions (Figure 2) and the therapeutic response improved after 1 year of treatment (Figure 3). No adverse events were noted during the treatment period.



Segmental vitiligo lacks consistently effective treatment options. This subtype of vitiligo is classically resistant to conventional therapeutic options. Surgery may be a more effective and long-lasting treatment option but is not suitable for every patient.1,3 Janus kinase (JAK) inhibitors are the newest treatment options being explored for topical and systemic treatment of vitiligo, with promising results in active and stable NSV lesions4,5; however, SV rarely is represented in case reports and clinical trials. The topical JAK inhibitor ruxolitinib has been approved for use in NSV,5 and a phase 2 trial with oral ritlecitinib only included patients with NSV.4 Furthermore, JAK inhibitors have been studied and approved for children aged 12 years or older as well as for adults,4,5 but younger age groups (4-10 years)—in whom SV most frequently manifests, as in our patient—have been excluded from these studies.1 We present a novel case of SV of the right hemiface in a child that was successfully treated with NB-UVB phototherapy in association with topical calcineurin inhibitors.
The role of phototherapy for the treatment of vitiligo has been well documented, and it frequently is combined with other therapeutic modalities, such as topical anti-inflammatory drugs or, most recently, laser and micrografting techniques.6,7 The most frequently used modality is NB-UVB. In the active phase, it performs an immunomodulatory role, while in the stable phase, it stimulates migration and activity of perilesional and hair follicle melanocytes.8 Initiating therapy early is advisable, particularly during the first 6 months of progression, as there is a higher probability of response1,3,8; nevertheless, a good response was achieved despite the 5-year evolution of vitiligo in our patient. This is a safe option for a skin condition that may begin early in life and require long-term treatment.8 A main concern would be an increased risk for skin cancer associated with repeated NB-UVB exposure, which has not been verified in a recent analysis.9
Segmental vitiligo can considerably impact the patient’s quality of life, affecting social interactions and self-perception, particularly in younger patients with facial involvement; thus, effective and safe therapeutic strategies adapted to the individual and their vitiligo lesions should be discussed. Classical treatment options remain valid and provide good results for some patients; therefore, they should not be disregarded even with the rise of innovative therapies.
To the Editor:
Segmental vitiligo (SV) accounts for a minority of vitiligo cases and most frequently occurs in children.1 It characteristically manifests unilaterally and affects a single body area with a sharp midline demarcation. In contrast to nonsegmental vitiligo (NSV), SV typically stabilizes early in the disease progression.1 The pathophysiology of this vitiligo subtype is not well established, but possible autoinflammatory mechanisms associated with somatic mosaicism, neuronal mechanisms, and/or microvascular skin-homing have been proposed.2 We present the case of a pediatric patient with segmental vitiligo of the right hemiface treated with a combination of a topical calcineurin inhibitor and narrow-band UVB (NB-UVB) phototherapy.
An otherwise healthy 7-year-old boy presented to the dermatology department for evaluation of depigmented macules and patches affecting the right hemiface (temporal, periorbital, malar, perioral, preauricular, and mandibular regions) and neck associated with homolateral leukotrichia of the scalp and facial hair as well as the eyelashes of 5 years’ duration. The findings were consistent with SV (Figure 1). The patient previously had been diagnosed based on the clinical findings and treated with continuous application of topical calcineurin inhibitors plus oral cyclosporine (3 mg/kg/d) for 1 year, but the response was poor. The condition had a severe impact on the patient’s quality of life and social relationships. Therapeutic options were discussed with the patient’s caregivers, and ultimately NB-UVB phototherapy was started twice weekly with 10% increases in the dose at each treatment. Topical tacrolimus ointment (1 mg/g) also was started, and the cyclosporine was stopped. Evaluation of treatment progress occurred every 3 months, with progressive repigmentation of the patches following a perifollicular pattern. After 6 months of phototherapy, there was notable repigmentation of the affected areas, particularly in the malar, perioral, and perinasal regions (Figure 2) and the therapeutic response improved after 1 year of treatment (Figure 3). No adverse events were noted during the treatment period.



Segmental vitiligo lacks consistently effective treatment options. This subtype of vitiligo is classically resistant to conventional therapeutic options. Surgery may be a more effective and long-lasting treatment option but is not suitable for every patient.1,3 Janus kinase (JAK) inhibitors are the newest treatment options being explored for topical and systemic treatment of vitiligo, with promising results in active and stable NSV lesions4,5; however, SV rarely is represented in case reports and clinical trials. The topical JAK inhibitor ruxolitinib has been approved for use in NSV,5 and a phase 2 trial with oral ritlecitinib only included patients with NSV.4 Furthermore, JAK inhibitors have been studied and approved for children aged 12 years or older as well as for adults,4,5 but younger age groups (4-10 years)—in whom SV most frequently manifests, as in our patient—have been excluded from these studies.1 We present a novel case of SV of the right hemiface in a child that was successfully treated with NB-UVB phototherapy in association with topical calcineurin inhibitors.
The role of phototherapy for the treatment of vitiligo has been well documented, and it frequently is combined with other therapeutic modalities, such as topical anti-inflammatory drugs or, most recently, laser and micrografting techniques.6,7 The most frequently used modality is NB-UVB. In the active phase, it performs an immunomodulatory role, while in the stable phase, it stimulates migration and activity of perilesional and hair follicle melanocytes.8 Initiating therapy early is advisable, particularly during the first 6 months of progression, as there is a higher probability of response1,3,8; nevertheless, a good response was achieved despite the 5-year evolution of vitiligo in our patient. This is a safe option for a skin condition that may begin early in life and require long-term treatment.8 A main concern would be an increased risk for skin cancer associated with repeated NB-UVB exposure, which has not been verified in a recent analysis.9
Segmental vitiligo can considerably impact the patient’s quality of life, affecting social interactions and self-perception, particularly in younger patients with facial involvement; thus, effective and safe therapeutic strategies adapted to the individual and their vitiligo lesions should be discussed. Classical treatment options remain valid and provide good results for some patients; therefore, they should not be disregarded even with the rise of innovative therapies.
- Speeckaert R, Lambert J, Bulat V, et al. Autoimmunity in segmental vitiligo. Front Immunol. 2020;11:568447. doi:10.3389/fimmu.2020.568447
- Lin X, Meng X, Lin J. Segmental vitiligo: autoimmune pathogenesis, neuronal mechanisms, and somatic mosaicism. Int J Dermatol. 2025;64:490-498. doi:10.1111/ijd.17627
- Khalili M, Amiri R, Mohammadi S, et al. Efficacy and safety of traditional and surgical treatment modalities in segmental vitiligo: a review article. J Cosmet Dermatol. 2022;21:2360-2373. doi:10.1111/jocd.14899
- Yamaguchi Y, Peeva E, Del Duca E, et al. Ritlecitinib, a JAK3/TEC family kinase inhibitor, stabilizes active lesions and repigments stable lesions in vitiligo. Arch Dermatol Res. 2024;316:478. doi:10.1007/s00403-024-03182-y
- Rosmarin D, Passeron T, Pandya AG, et al. Two phase 3, randomized, controlled trials of ruxolitinib cream for vitiligo. N Engl J Med. 2022;387:1445-1455. doi:10.1056/NEJMoa2118828
- Chavez-Alvarez S, Herz-Ruelas M, Ocampo-Candiani J, et al. Stable segmental vitiligo treated with punch mini-grafts and narrow band UVB phototherapy. Australas J Dermatol. 2020;61:83-85. doi:10.1111/ajd.13105
- Kim WI, Kim S, Lee SH, et al. The efficacy of fractional carbon dioxide laser combined with narrow-band ultraviolet B phototherapy for non-segmental vitiligo: a systematic review and meta-analysis. Lasers Med Sci. 2021;36:165-173. doi:10.1007/s10103-020-03069-0
- Esmat S, Hegazy RA, Shalaby S, et al. Phototherapy and combination therapies for vitiligo. Dermatol Clin. 2017;35:171-192. doi:10.1016/j.det.2016.11.008
- Mimouni I, Shulman J, Unes AA, et al. Frequency of skin cancer among psoriasis, vitiligo, and mycosis fungoides patients treated with narrowband ultraviolet B phototherapy. Photodermatol Photoimmunol Photomed. 2024;40:E12936. doi:10.1111/phpp.12936
- Speeckaert R, Lambert J, Bulat V, et al. Autoimmunity in segmental vitiligo. Front Immunol. 2020;11:568447. doi:10.3389/fimmu.2020.568447
- Lin X, Meng X, Lin J. Segmental vitiligo: autoimmune pathogenesis, neuronal mechanisms, and somatic mosaicism. Int J Dermatol. 2025;64:490-498. doi:10.1111/ijd.17627
- Khalili M, Amiri R, Mohammadi S, et al. Efficacy and safety of traditional and surgical treatment modalities in segmental vitiligo: a review article. J Cosmet Dermatol. 2022;21:2360-2373. doi:10.1111/jocd.14899
- Yamaguchi Y, Peeva E, Del Duca E, et al. Ritlecitinib, a JAK3/TEC family kinase inhibitor, stabilizes active lesions and repigments stable lesions in vitiligo. Arch Dermatol Res. 2024;316:478. doi:10.1007/s00403-024-03182-y
- Rosmarin D, Passeron T, Pandya AG, et al. Two phase 3, randomized, controlled trials of ruxolitinib cream for vitiligo. N Engl J Med. 2022;387:1445-1455. doi:10.1056/NEJMoa2118828
- Chavez-Alvarez S, Herz-Ruelas M, Ocampo-Candiani J, et al. Stable segmental vitiligo treated with punch mini-grafts and narrow band UVB phototherapy. Australas J Dermatol. 2020;61:83-85. doi:10.1111/ajd.13105
- Kim WI, Kim S, Lee SH, et al. The efficacy of fractional carbon dioxide laser combined with narrow-band ultraviolet B phototherapy for non-segmental vitiligo: a systematic review and meta-analysis. Lasers Med Sci. 2021;36:165-173. doi:10.1007/s10103-020-03069-0
- Esmat S, Hegazy RA, Shalaby S, et al. Phototherapy and combination therapies for vitiligo. Dermatol Clin. 2017;35:171-192. doi:10.1016/j.det.2016.11.008
- Mimouni I, Shulman J, Unes AA, et al. Frequency of skin cancer among psoriasis, vitiligo, and mycosis fungoides patients treated with narrowband ultraviolet B phototherapy. Photodermatol Photoimmunol Photomed. 2024;40:E12936. doi:10.1111/phpp.12936
Phototherapy for Pediatric Segmental Vitiligo
Phototherapy for Pediatric Segmental Vitiligo
Practice Points
- Narrow-band UVB in combination with topical tacrolimus may be an effective treatment option for pediatric segmental vitiligo (SV), even in longstanding disease.
- Current evidence for Janus kinase (JAK) inhibitors largely is derived from studies in nonsegmental vitiligo and in patients aged 12 years or older.
- Segmental vitiligo is underrepresented in clinical trials, and topical and systemic JAK inhibitors are not approved for younger children, in whom SV most commonly occurs.
Cancer Data Trends 2026
The annual issue of Cancer Data Trends, produced in collaboration with the Association of VA Hematology/Oncology (AVAHO), highlights the latest research in some of the top cancers impacting US veterans.
In this issue:
- Prostate Cancer: Exposures, Racial Differences, and Treatment Trends
- Expanding Breast Cancer Care for Women Veterans: Genetic Testing and New Therapies
- Multiple Myeloma: Herbicide Exposure, BMI, and Novel Quadruplet Regimens
- Melanoma in Veterans: Higher Risk, Delayed Diagnosis, and Evolving Solutions
- Colorectal Cancer Trends and Digital Interventions in Veterans
- Advancing HCC Management: Updated Guidelines and VHA's Innovative Screening Trial
- Mental Health Care in Veterans With Cancer
The annual issue of Cancer Data Trends, produced in collaboration with the Association of VA Hematology/Oncology (AVAHO), highlights the latest research in some of the top cancers impacting US veterans.
In this issue:
- Prostate Cancer: Exposures, Racial Differences, and Treatment Trends
- Expanding Breast Cancer Care for Women Veterans: Genetic Testing and New Therapies
- Multiple Myeloma: Herbicide Exposure, BMI, and Novel Quadruplet Regimens
- Melanoma in Veterans: Higher Risk, Delayed Diagnosis, and Evolving Solutions
- Colorectal Cancer Trends and Digital Interventions in Veterans
- Advancing HCC Management: Updated Guidelines and VHA's Innovative Screening Trial
- Mental Health Care in Veterans With Cancer
The annual issue of Cancer Data Trends, produced in collaboration with the Association of VA Hematology/Oncology (AVAHO), highlights the latest research in some of the top cancers impacting US veterans.
In this issue:
- Prostate Cancer: Exposures, Racial Differences, and Treatment Trends
- Expanding Breast Cancer Care for Women Veterans: Genetic Testing and New Therapies
- Multiple Myeloma: Herbicide Exposure, BMI, and Novel Quadruplet Regimens
- Melanoma in Veterans: Higher Risk, Delayed Diagnosis, and Evolving Solutions
- Colorectal Cancer Trends and Digital Interventions in Veterans
- Advancing HCC Management: Updated Guidelines and VHA's Innovative Screening Trial
- Mental Health Care in Veterans With Cancer