User login
Algorithm trims time to treatment of acute hypertension in pregnancy
Use of a semiautonomous algorithm to initiate treatment for hypertension emergencies in pregnancy significantly increased the number of individuals treated promptly, based on data from 959 obstetric patients.
Data show poor compliance with the current American College of Obstetricians and Gynecologists recommendations for treatment of acute severe hypertension with no more than 30-60 minutes’ delay; low compliance may be caused by “multiple factors including lack of intravenous access, inadequate health care practitioner or nursing availability, and implicit racial biases,” wrote Courtney Martin, DO, of Loma Linda (Calif.) University School of Medicine and colleagues.
Semiautomated treatment algorithms have been used to improve timely treatment of conditions including myocardial infarction, heart failure, acute stroke, and asthma, but their use in obstetrics to date has been limited, the researchers noted.
In a retrospective cohort study published in Obstetrics & Gynecology, the researchers identified pregnant and postpartum women treated for severe hypertension at a single center between January 2017 and March 2020. A semiautonomous treatment algorithm was implemented between May 2018 and March 2019. The algorithm included vital sign monitoring, blood pressure thresholds for diagnosis of severe hypertension, and automated order sets for recommended first-line antihypertensive therapy. The primary outcomes were treatment with antihypertensive therapy within 15, 30, and 60 minutes of diagnosis. “Severe hypertension was defined as systolic blood pressure 160 mm Hg or higher or diastolic blood pressure 110 mm Hg or higher,” the researchers said.
The study population was divided into three groups; a preimplementation group (373 patients) managed between January 2017 and April 2018, a during-implementation group (334 patients) managed between May 2018 and March 2019, and a postimplementation group (252 patients) managed between April 2019 and March 2020. Patient demographics were similar among all three groups.
Timely treatment improves with algorithm
Overall, treatment of severe hypertension within 15 minutes of diagnosis was 36.5% preimplementation, 45.8% during implementation, and 55.6% postimplementation. Severe hypertension treatment within 30 minutes of diagnosis was 65.9% preimplementation, 77.8% during implementation, and 79.0% post implementation. Differences were significant between pre- and post implementation for 15 minutes and 30 minutes, but no significant differences occurred in the patients treated within 60 minutes before and after implementation of the algorithm.
The study findings were limited by several factors, including the inability to separate peer-to-peer education and other training from the impact of the algorithm, as well as a lack of data on the effect of the algorithm on maternal or neonatal outcomes, the researchers noted.
However, the results support the potential of a semiautonomous algorithm to significantly improve adherence to the recommended treatment guidelines for severe hypertension in pregnancy and post partum, they said. Given the expected increase in hypertensive disorders in pregnancy because of the trends in older age and higher obesity rates in pregnant women, “Integration of semiautonomous treatment algorithms similar to ours into routine obstetric practices could help reduce the health care burden and improve clinical outcomes, especially in areas with limited health care resources,” they concluded.
Algorithm may reduce disparities
The overall rise in maternal mortality in the United States remains a concern, but “Even more concerning are the disturbing racial disparities that persist across socioeconomic strata,” wrote Alisse Hauspurg, MD, of the University of Pittsburgh in an accompanying editorial. “There is clear evidence that expeditious treatment of obstetric hypertensive emergency reduces the risk of severe morbidities including stroke, eclampsia, and maternal death,” she emphasized, but compliance with the ACOG recommendations to treat severe hypertension within 30-60 minutes of confirmation remains low, she said.
In this study, not only did use of the algorithm reduce time to antihypertensive therapy, but more than 50% of patients were treated for severe hypertension within 15 minutes, and more than 90% within 60 minutes, “which was sustained after the implementation phase,” and aligns with the ACOG recommendations, Dr. Hauspurg said. “Although Martin et al.’s algorithm was limited to the initial management of obstetric hypertensive emergency, it could readily be expanded to follow the full ACOG algorithm for management of hypertension in pregnancy,” she noted.
In addition, Black women are more frequently diagnosed with hypertensive disorders of pregnancy, including severe hypertension, and the algorithm might improve disparities, she said.
“It is plausible that widespread implementation of such a semiautonomous algorithm at hospitals across the country could reduce delays in treatment and prevent hypertension-related morbidities,” said Dr. Hauspurg. “The use of innovative approaches to management of severe hypertension and other obstetric emergencies has the potential to allow provision of more equitable care by overcoming health care practitioner and system biases, which could meaningfully reduce disparities in care and change the trajectory of maternal morbidity and mortality in the United States,” she emphasized.
Need to create culture of safety
“Maternal mortality in the United States is the highest among developed nations, and shocking disparities exist in outcomes for non-Hispanic Black and American Indian/Alaskan Native women,” said Lisa Hollier, MD, of Texas Children’s Health Plan in Bellaire. “In a California review of maternal deaths, the greatest quality improvement opportunities were missed diagnosis and ineffective treatment of preeclampsia and related diseases, which occurred in 65% of the cases where women died of preeclampsia/eclampsia,” she said.
The current study “is very timely as more and more states across the nation are participating in the AIM (Alliance for Innovation on Maternal Health) programs to prevent pregnancy-related mortality,” Dr. Hollier noted.
“This study demonstrated a significant association between implementation of the algorithm and an increased percentage of treatment of severe hypertension within 30 minutes,” Dr. Hollier said. “With the implementation of a comprehensive program that included treatment algorithms, the Illinois Perinatal Quality Collaborative improved timely treatment for women with severe high blood pressure, increasing the percentage of patients treated within 60 minutes from 41% at baseline to 79% in the first year of the project.”
The take-home message is that “implementation of the semiautonomous treatment algorithm can address important clinical variation, including delays in appropriate treatment of severe hypertension,” said Dr. Hollier. However, “One of the potential barriers [to use of an algorithm] is the need for accurate, real-time clinical assessment. Resources must be available to ensure appropriate monitoring,” Dr. Hollier noted. “Collaboration and support of implementation of these treatment algorithms must extend through the nursing staff, the physicians, and advanced-practice providers. Medical staff and administrative leaders are essential in creating a culture of safety and continuous process improvement,” she said.
In addition, “long-term follow-up on the implementation of broader quality improvement programs is essential,” Dr. Hollier said. “While implementation of an algorithm can, and did, result in process improvements, assessment of broader implementation of evidence-based bundles, combined with a systematic approach to redesign of multiple related processes needs to occur and include outcomes of severe maternal morbidity and mortality,” she explained.
The study received no outside funding. The researchers had no financial conflicts to disclose.
Neither Dr. Hauspurg nor Dr. Hollier had financial conflicts to disclose.
Use of a semiautonomous algorithm to initiate treatment for hypertension emergencies in pregnancy significantly increased the number of individuals treated promptly, based on data from 959 obstetric patients.
Data show poor compliance with the current American College of Obstetricians and Gynecologists recommendations for treatment of acute severe hypertension with no more than 30-60 minutes’ delay; low compliance may be caused by “multiple factors including lack of intravenous access, inadequate health care practitioner or nursing availability, and implicit racial biases,” wrote Courtney Martin, DO, of Loma Linda (Calif.) University School of Medicine and colleagues.
Semiautomated treatment algorithms have been used to improve timely treatment of conditions including myocardial infarction, heart failure, acute stroke, and asthma, but their use in obstetrics to date has been limited, the researchers noted.
In a retrospective cohort study published in Obstetrics & Gynecology, the researchers identified pregnant and postpartum women treated for severe hypertension at a single center between January 2017 and March 2020. A semiautonomous treatment algorithm was implemented between May 2018 and March 2019. The algorithm included vital sign monitoring, blood pressure thresholds for diagnosis of severe hypertension, and automated order sets for recommended first-line antihypertensive therapy. The primary outcomes were treatment with antihypertensive therapy within 15, 30, and 60 minutes of diagnosis. “Severe hypertension was defined as systolic blood pressure 160 mm Hg or higher or diastolic blood pressure 110 mm Hg or higher,” the researchers said.
The study population was divided into three groups; a preimplementation group (373 patients) managed between January 2017 and April 2018, a during-implementation group (334 patients) managed between May 2018 and March 2019, and a postimplementation group (252 patients) managed between April 2019 and March 2020. Patient demographics were similar among all three groups.
Timely treatment improves with algorithm
Overall, treatment of severe hypertension within 15 minutes of diagnosis was 36.5% preimplementation, 45.8% during implementation, and 55.6% postimplementation. Severe hypertension treatment within 30 minutes of diagnosis was 65.9% preimplementation, 77.8% during implementation, and 79.0% post implementation. Differences were significant between pre- and post implementation for 15 minutes and 30 minutes, but no significant differences occurred in the patients treated within 60 minutes before and after implementation of the algorithm.
The study findings were limited by several factors, including the inability to separate peer-to-peer education and other training from the impact of the algorithm, as well as a lack of data on the effect of the algorithm on maternal or neonatal outcomes, the researchers noted.
However, the results support the potential of a semiautonomous algorithm to significantly improve adherence to the recommended treatment guidelines for severe hypertension in pregnancy and post partum, they said. Given the expected increase in hypertensive disorders in pregnancy because of the trends in older age and higher obesity rates in pregnant women, “Integration of semiautonomous treatment algorithms similar to ours into routine obstetric practices could help reduce the health care burden and improve clinical outcomes, especially in areas with limited health care resources,” they concluded.
Algorithm may reduce disparities
The overall rise in maternal mortality in the United States remains a concern, but “Even more concerning are the disturbing racial disparities that persist across socioeconomic strata,” wrote Alisse Hauspurg, MD, of the University of Pittsburgh in an accompanying editorial. “There is clear evidence that expeditious treatment of obstetric hypertensive emergency reduces the risk of severe morbidities including stroke, eclampsia, and maternal death,” she emphasized, but compliance with the ACOG recommendations to treat severe hypertension within 30-60 minutes of confirmation remains low, she said.
In this study, not only did use of the algorithm reduce time to antihypertensive therapy, but more than 50% of patients were treated for severe hypertension within 15 minutes, and more than 90% within 60 minutes, “which was sustained after the implementation phase,” and aligns with the ACOG recommendations, Dr. Hauspurg said. “Although Martin et al.’s algorithm was limited to the initial management of obstetric hypertensive emergency, it could readily be expanded to follow the full ACOG algorithm for management of hypertension in pregnancy,” she noted.
In addition, Black women are more frequently diagnosed with hypertensive disorders of pregnancy, including severe hypertension, and the algorithm might improve disparities, she said.
“It is plausible that widespread implementation of such a semiautonomous algorithm at hospitals across the country could reduce delays in treatment and prevent hypertension-related morbidities,” said Dr. Hauspurg. “The use of innovative approaches to management of severe hypertension and other obstetric emergencies has the potential to allow provision of more equitable care by overcoming health care practitioner and system biases, which could meaningfully reduce disparities in care and change the trajectory of maternal morbidity and mortality in the United States,” she emphasized.
Need to create culture of safety
“Maternal mortality in the United States is the highest among developed nations, and shocking disparities exist in outcomes for non-Hispanic Black and American Indian/Alaskan Native women,” said Lisa Hollier, MD, of Texas Children’s Health Plan in Bellaire. “In a California review of maternal deaths, the greatest quality improvement opportunities were missed diagnosis and ineffective treatment of preeclampsia and related diseases, which occurred in 65% of the cases where women died of preeclampsia/eclampsia,” she said.
The current study “is very timely as more and more states across the nation are participating in the AIM (Alliance for Innovation on Maternal Health) programs to prevent pregnancy-related mortality,” Dr. Hollier noted.
“This study demonstrated a significant association between implementation of the algorithm and an increased percentage of treatment of severe hypertension within 30 minutes,” Dr. Hollier said. “With the implementation of a comprehensive program that included treatment algorithms, the Illinois Perinatal Quality Collaborative improved timely treatment for women with severe high blood pressure, increasing the percentage of patients treated within 60 minutes from 41% at baseline to 79% in the first year of the project.”
The take-home message is that “implementation of the semiautonomous treatment algorithm can address important clinical variation, including delays in appropriate treatment of severe hypertension,” said Dr. Hollier. However, “One of the potential barriers [to use of an algorithm] is the need for accurate, real-time clinical assessment. Resources must be available to ensure appropriate monitoring,” Dr. Hollier noted. “Collaboration and support of implementation of these treatment algorithms must extend through the nursing staff, the physicians, and advanced-practice providers. Medical staff and administrative leaders are essential in creating a culture of safety and continuous process improvement,” she said.
In addition, “long-term follow-up on the implementation of broader quality improvement programs is essential,” Dr. Hollier said. “While implementation of an algorithm can, and did, result in process improvements, assessment of broader implementation of evidence-based bundles, combined with a systematic approach to redesign of multiple related processes needs to occur and include outcomes of severe maternal morbidity and mortality,” she explained.
The study received no outside funding. The researchers had no financial conflicts to disclose.
Neither Dr. Hauspurg nor Dr. Hollier had financial conflicts to disclose.
Use of a semiautonomous algorithm to initiate treatment for hypertension emergencies in pregnancy significantly increased the number of individuals treated promptly, based on data from 959 obstetric patients.
Data show poor compliance with the current American College of Obstetricians and Gynecologists recommendations for treatment of acute severe hypertension with no more than 30-60 minutes’ delay; low compliance may be caused by “multiple factors including lack of intravenous access, inadequate health care practitioner or nursing availability, and implicit racial biases,” wrote Courtney Martin, DO, of Loma Linda (Calif.) University School of Medicine and colleagues.
Semiautomated treatment algorithms have been used to improve timely treatment of conditions including myocardial infarction, heart failure, acute stroke, and asthma, but their use in obstetrics to date has been limited, the researchers noted.
In a retrospective cohort study published in Obstetrics & Gynecology, the researchers identified pregnant and postpartum women treated for severe hypertension at a single center between January 2017 and March 2020. A semiautonomous treatment algorithm was implemented between May 2018 and March 2019. The algorithm included vital sign monitoring, blood pressure thresholds for diagnosis of severe hypertension, and automated order sets for recommended first-line antihypertensive therapy. The primary outcomes were treatment with antihypertensive therapy within 15, 30, and 60 minutes of diagnosis. “Severe hypertension was defined as systolic blood pressure 160 mm Hg or higher or diastolic blood pressure 110 mm Hg or higher,” the researchers said.
The study population was divided into three groups; a preimplementation group (373 patients) managed between January 2017 and April 2018, a during-implementation group (334 patients) managed between May 2018 and March 2019, and a postimplementation group (252 patients) managed between April 2019 and March 2020. Patient demographics were similar among all three groups.
Timely treatment improves with algorithm
Overall, treatment of severe hypertension within 15 minutes of diagnosis was 36.5% preimplementation, 45.8% during implementation, and 55.6% postimplementation. Severe hypertension treatment within 30 minutes of diagnosis was 65.9% preimplementation, 77.8% during implementation, and 79.0% post implementation. Differences were significant between pre- and post implementation for 15 minutes and 30 minutes, but no significant differences occurred in the patients treated within 60 minutes before and after implementation of the algorithm.
The study findings were limited by several factors, including the inability to separate peer-to-peer education and other training from the impact of the algorithm, as well as a lack of data on the effect of the algorithm on maternal or neonatal outcomes, the researchers noted.
However, the results support the potential of a semiautonomous algorithm to significantly improve adherence to the recommended treatment guidelines for severe hypertension in pregnancy and post partum, they said. Given the expected increase in hypertensive disorders in pregnancy because of the trends in older age and higher obesity rates in pregnant women, “Integration of semiautonomous treatment algorithms similar to ours into routine obstetric practices could help reduce the health care burden and improve clinical outcomes, especially in areas with limited health care resources,” they concluded.
Algorithm may reduce disparities
The overall rise in maternal mortality in the United States remains a concern, but “Even more concerning are the disturbing racial disparities that persist across socioeconomic strata,” wrote Alisse Hauspurg, MD, of the University of Pittsburgh in an accompanying editorial. “There is clear evidence that expeditious treatment of obstetric hypertensive emergency reduces the risk of severe morbidities including stroke, eclampsia, and maternal death,” she emphasized, but compliance with the ACOG recommendations to treat severe hypertension within 30-60 minutes of confirmation remains low, she said.
In this study, not only did use of the algorithm reduce time to antihypertensive therapy, but more than 50% of patients were treated for severe hypertension within 15 minutes, and more than 90% within 60 minutes, “which was sustained after the implementation phase,” and aligns with the ACOG recommendations, Dr. Hauspurg said. “Although Martin et al.’s algorithm was limited to the initial management of obstetric hypertensive emergency, it could readily be expanded to follow the full ACOG algorithm for management of hypertension in pregnancy,” she noted.
In addition, Black women are more frequently diagnosed with hypertensive disorders of pregnancy, including severe hypertension, and the algorithm might improve disparities, she said.
“It is plausible that widespread implementation of such a semiautonomous algorithm at hospitals across the country could reduce delays in treatment and prevent hypertension-related morbidities,” said Dr. Hauspurg. “The use of innovative approaches to management of severe hypertension and other obstetric emergencies has the potential to allow provision of more equitable care by overcoming health care practitioner and system biases, which could meaningfully reduce disparities in care and change the trajectory of maternal morbidity and mortality in the United States,” she emphasized.
Need to create culture of safety
“Maternal mortality in the United States is the highest among developed nations, and shocking disparities exist in outcomes for non-Hispanic Black and American Indian/Alaskan Native women,” said Lisa Hollier, MD, of Texas Children’s Health Plan in Bellaire. “In a California review of maternal deaths, the greatest quality improvement opportunities were missed diagnosis and ineffective treatment of preeclampsia and related diseases, which occurred in 65% of the cases where women died of preeclampsia/eclampsia,” she said.
The current study “is very timely as more and more states across the nation are participating in the AIM (Alliance for Innovation on Maternal Health) programs to prevent pregnancy-related mortality,” Dr. Hollier noted.
“This study demonstrated a significant association between implementation of the algorithm and an increased percentage of treatment of severe hypertension within 30 minutes,” Dr. Hollier said. “With the implementation of a comprehensive program that included treatment algorithms, the Illinois Perinatal Quality Collaborative improved timely treatment for women with severe high blood pressure, increasing the percentage of patients treated within 60 minutes from 41% at baseline to 79% in the first year of the project.”
The take-home message is that “implementation of the semiautonomous treatment algorithm can address important clinical variation, including delays in appropriate treatment of severe hypertension,” said Dr. Hollier. However, “One of the potential barriers [to use of an algorithm] is the need for accurate, real-time clinical assessment. Resources must be available to ensure appropriate monitoring,” Dr. Hollier noted. “Collaboration and support of implementation of these treatment algorithms must extend through the nursing staff, the physicians, and advanced-practice providers. Medical staff and administrative leaders are essential in creating a culture of safety and continuous process improvement,” she said.
In addition, “long-term follow-up on the implementation of broader quality improvement programs is essential,” Dr. Hollier said. “While implementation of an algorithm can, and did, result in process improvements, assessment of broader implementation of evidence-based bundles, combined with a systematic approach to redesign of multiple related processes needs to occur and include outcomes of severe maternal morbidity and mortality,” she explained.
The study received no outside funding. The researchers had no financial conflicts to disclose.
Neither Dr. Hauspurg nor Dr. Hollier had financial conflicts to disclose.
FROM OBSTETRICS & GYNECOLOGY
Gestational diabetes carries CVD risk years later
Women who’ve had gestational diabetes are 40% more likely to develop coronary artery calcification later in life than are women haven’t, and attaining normal glycemic levels doesn’t diminish their midlife risk for atherosclerotic cardiovascular disease.
“The new finding from this study is that women with gestational diabetes had twice the risk of coronary artery calcium, compared to women who never had gestational diabetes, even though both groups attained normal blood sugar levels many years after pregnancy,” lead author Erica P. Gunderson, PhD, MS, MPH, said in an interview about a community-based prospective cohort study of young adults followed for up to 25 years, which was published in Circulation (2021 Feb 1. doi: 10.1161/CIRCULATIONAHA.120.047320).
Previous studies have reported a higher risk of heart disease in women who had gestational diabetes (GD) and later developed type 2 diabetes, but they didn’t elucidate whether that risk carried over in GD patients whose glycemic levels were normal after pregnancy. In 2018, the American College of Cardiology/American Heart Association Cholesterol Clinical Practice Guidelines specified that a history of GD increases women’s risk for coronary artery calcification (CAC).
This study analyzed data of 1,133 women ages 18-30 enrolled in the Coronary Artery Risk Development in Young Adults (CARDIA) study who had no diabetes in the baseline years of 1985-1986 and had given birth at least once in the ensuing 25 years. They had glucose tolerance testing at baseline and up to five times through the study period, along with evaluation for GD status and coronary artery calcification CAC measurements at least once at years 15, 20 and 25 (2001-2011).
CARDIA enrolled 5,155 young Black and White men and women ages 18-30 from four distinct geographic areas: Birmingham, Ala.; Chicago; Minneapolis; and Oakland, Calif. About 52% of the study population was Black.
Of the women who’d given birth, 139 (12%) had GD. Their average age at follow-up was 47.6 years, and 25% of the GD patients (34) had CAC, compared with 15% (149/994) in the non-GD group.
Dr. Gunderson noted that the same relative risk for CAC applied to women who had GD and went on to develop prediabetes or were diagnosed with type 2 diabetes during follow-up.
Risks persist even in normoglycemia
In the GD group, the adjusted hazard ratio for having CAC with normoglycemia was 2.3 (95% confidence interval, 1.34-4.09). The researchers also calculated HRs for prediabetes and incident diabetes: 1.5 (95% CI, 1.06-2.24) in no-GD and 2.1 (95% CI, 1.09-4.17) for GD for prediabetes; and 2.2 (95% CI, 1.3-3.62) and 2.02 (95% CI, 0.98-4.19), respectively, for incident diabetes (P = .003).
“This means the risk of heart disease may be increased substantially in women with a history of gestational diabetes and may not diminish even if their blood-sugar levels remain normal for years later,” said Dr. Gunderson, an epidemiologist and senior research scientist at the Kaiser Permanente Northern California Division of Research in Oakland.
“The clinical implications of our findings are that women with previous GD may benefit from enhanced traditional CVD [cardiovascular disease] risk factor testing – i.e., for hypertension, dyslipidemia, and hyperinsulinemia,” Dr. Gunderson said. “Our findings also suggest that it could be beneficial to incorporate history of GD into risk calculators to improve CVD risk stratification and prevention.”
Strong findings argue for more frequent CVD screening
These study results may be the strongest data to date on the long-term effects of GD, said Prakash Deedwania, MD, professor of cardiology at the University of California, San Francisco. “It’s the strongest in the sense in that it’s sponsored, involved four different communities in different parts of the United States, enrolled individuals when they were young and followed them, and saw very few patients drop out for such a long-term study.” The study reported follow-up data on 72% of patients at 25 years, a rate Dr. Deedwania noted was “excellent.”
“Patients who have had GD should be screened aggressively – for not only diabetes, but other cardiovascular risk factors – early on to minimize the subsequent risk of cardiovascular disease is a very important point of this study,” he added. In the absence of a clinical guideline, Dr. Deedwania suggested women with GD might have screening for CV risk factors every 5-7 years depending on their risk profile, but emphasized that parameter isn’t settled.
Future research should focus on the link between GD and CVD risk, Dr. Gunderson said. “Research is needed to better characterize the severity of GD in relation to CVD outcomes, and to identify critical pregnancy-related periods to modify cardiometabolic risk.” The latter would include life-course studies across the full pregnancy continuum from preconception to lactation. “Interventions for primary prevention of CVD and the importance of modifiable lifestyle behaviors with the highest relevance to reduce both diabetes and CVD risks during the first year post partum merit increased research investigation,” she added.
Future studies might also explore the role of inflammation in the GD-CVD relationship, Dr. Deedwania said. “My hypothesis is, and it’s purely a hypothesis, that perhaps the presence of coronary artery calcification scores score in these individuals who were described as having normal glucose but who could be at risk could very well be related to the beginning of inflammation.”
Dr. Gunderson and Dr. Deedwania have no financial relationships to disclose. The study was funded by the National Institute of Diabetes and Digestive and Kidney Diseases and the National Heart, Lung, and Blood Institute.
Women who’ve had gestational diabetes are 40% more likely to develop coronary artery calcification later in life than are women haven’t, and attaining normal glycemic levels doesn’t diminish their midlife risk for atherosclerotic cardiovascular disease.
“The new finding from this study is that women with gestational diabetes had twice the risk of coronary artery calcium, compared to women who never had gestational diabetes, even though both groups attained normal blood sugar levels many years after pregnancy,” lead author Erica P. Gunderson, PhD, MS, MPH, said in an interview about a community-based prospective cohort study of young adults followed for up to 25 years, which was published in Circulation (2021 Feb 1. doi: 10.1161/CIRCULATIONAHA.120.047320).
Previous studies have reported a higher risk of heart disease in women who had gestational diabetes (GD) and later developed type 2 diabetes, but they didn’t elucidate whether that risk carried over in GD patients whose glycemic levels were normal after pregnancy. In 2018, the American College of Cardiology/American Heart Association Cholesterol Clinical Practice Guidelines specified that a history of GD increases women’s risk for coronary artery calcification (CAC).
This study analyzed data of 1,133 women ages 18-30 enrolled in the Coronary Artery Risk Development in Young Adults (CARDIA) study who had no diabetes in the baseline years of 1985-1986 and had given birth at least once in the ensuing 25 years. They had glucose tolerance testing at baseline and up to five times through the study period, along with evaluation for GD status and coronary artery calcification CAC measurements at least once at years 15, 20 and 25 (2001-2011).
CARDIA enrolled 5,155 young Black and White men and women ages 18-30 from four distinct geographic areas: Birmingham, Ala.; Chicago; Minneapolis; and Oakland, Calif. About 52% of the study population was Black.
Of the women who’d given birth, 139 (12%) had GD. Their average age at follow-up was 47.6 years, and 25% of the GD patients (34) had CAC, compared with 15% (149/994) in the non-GD group.
Dr. Gunderson noted that the same relative risk for CAC applied to women who had GD and went on to develop prediabetes or were diagnosed with type 2 diabetes during follow-up.
Risks persist even in normoglycemia
In the GD group, the adjusted hazard ratio for having CAC with normoglycemia was 2.3 (95% confidence interval, 1.34-4.09). The researchers also calculated HRs for prediabetes and incident diabetes: 1.5 (95% CI, 1.06-2.24) in no-GD and 2.1 (95% CI, 1.09-4.17) for GD for prediabetes; and 2.2 (95% CI, 1.3-3.62) and 2.02 (95% CI, 0.98-4.19), respectively, for incident diabetes (P = .003).
“This means the risk of heart disease may be increased substantially in women with a history of gestational diabetes and may not diminish even if their blood-sugar levels remain normal for years later,” said Dr. Gunderson, an epidemiologist and senior research scientist at the Kaiser Permanente Northern California Division of Research in Oakland.
“The clinical implications of our findings are that women with previous GD may benefit from enhanced traditional CVD [cardiovascular disease] risk factor testing – i.e., for hypertension, dyslipidemia, and hyperinsulinemia,” Dr. Gunderson said. “Our findings also suggest that it could be beneficial to incorporate history of GD into risk calculators to improve CVD risk stratification and prevention.”
Strong findings argue for more frequent CVD screening
These study results may be the strongest data to date on the long-term effects of GD, said Prakash Deedwania, MD, professor of cardiology at the University of California, San Francisco. “It’s the strongest in the sense in that it’s sponsored, involved four different communities in different parts of the United States, enrolled individuals when they were young and followed them, and saw very few patients drop out for such a long-term study.” The study reported follow-up data on 72% of patients at 25 years, a rate Dr. Deedwania noted was “excellent.”
“Patients who have had GD should be screened aggressively – for not only diabetes, but other cardiovascular risk factors – early on to minimize the subsequent risk of cardiovascular disease is a very important point of this study,” he added. In the absence of a clinical guideline, Dr. Deedwania suggested women with GD might have screening for CV risk factors every 5-7 years depending on their risk profile, but emphasized that parameter isn’t settled.
Future research should focus on the link between GD and CVD risk, Dr. Gunderson said. “Research is needed to better characterize the severity of GD in relation to CVD outcomes, and to identify critical pregnancy-related periods to modify cardiometabolic risk.” The latter would include life-course studies across the full pregnancy continuum from preconception to lactation. “Interventions for primary prevention of CVD and the importance of modifiable lifestyle behaviors with the highest relevance to reduce both diabetes and CVD risks during the first year post partum merit increased research investigation,” she added.
Future studies might also explore the role of inflammation in the GD-CVD relationship, Dr. Deedwania said. “My hypothesis is, and it’s purely a hypothesis, that perhaps the presence of coronary artery calcification scores score in these individuals who were described as having normal glucose but who could be at risk could very well be related to the beginning of inflammation.”
Dr. Gunderson and Dr. Deedwania have no financial relationships to disclose. The study was funded by the National Institute of Diabetes and Digestive and Kidney Diseases and the National Heart, Lung, and Blood Institute.
Women who’ve had gestational diabetes are 40% more likely to develop coronary artery calcification later in life than are women haven’t, and attaining normal glycemic levels doesn’t diminish their midlife risk for atherosclerotic cardiovascular disease.
“The new finding from this study is that women with gestational diabetes had twice the risk of coronary artery calcium, compared to women who never had gestational diabetes, even though both groups attained normal blood sugar levels many years after pregnancy,” lead author Erica P. Gunderson, PhD, MS, MPH, said in an interview about a community-based prospective cohort study of young adults followed for up to 25 years, which was published in Circulation (2021 Feb 1. doi: 10.1161/CIRCULATIONAHA.120.047320).
Previous studies have reported a higher risk of heart disease in women who had gestational diabetes (GD) and later developed type 2 diabetes, but they didn’t elucidate whether that risk carried over in GD patients whose glycemic levels were normal after pregnancy. In 2018, the American College of Cardiology/American Heart Association Cholesterol Clinical Practice Guidelines specified that a history of GD increases women’s risk for coronary artery calcification (CAC).
This study analyzed data of 1,133 women ages 18-30 enrolled in the Coronary Artery Risk Development in Young Adults (CARDIA) study who had no diabetes in the baseline years of 1985-1986 and had given birth at least once in the ensuing 25 years. They had glucose tolerance testing at baseline and up to five times through the study period, along with evaluation for GD status and coronary artery calcification CAC measurements at least once at years 15, 20 and 25 (2001-2011).
CARDIA enrolled 5,155 young Black and White men and women ages 18-30 from four distinct geographic areas: Birmingham, Ala.; Chicago; Minneapolis; and Oakland, Calif. About 52% of the study population was Black.
Of the women who’d given birth, 139 (12%) had GD. Their average age at follow-up was 47.6 years, and 25% of the GD patients (34) had CAC, compared with 15% (149/994) in the non-GD group.
Dr. Gunderson noted that the same relative risk for CAC applied to women who had GD and went on to develop prediabetes or were diagnosed with type 2 diabetes during follow-up.
Risks persist even in normoglycemia
In the GD group, the adjusted hazard ratio for having CAC with normoglycemia was 2.3 (95% confidence interval, 1.34-4.09). The researchers also calculated HRs for prediabetes and incident diabetes: 1.5 (95% CI, 1.06-2.24) in no-GD and 2.1 (95% CI, 1.09-4.17) for GD for prediabetes; and 2.2 (95% CI, 1.3-3.62) and 2.02 (95% CI, 0.98-4.19), respectively, for incident diabetes (P = .003).
“This means the risk of heart disease may be increased substantially in women with a history of gestational diabetes and may not diminish even if their blood-sugar levels remain normal for years later,” said Dr. Gunderson, an epidemiologist and senior research scientist at the Kaiser Permanente Northern California Division of Research in Oakland.
“The clinical implications of our findings are that women with previous GD may benefit from enhanced traditional CVD [cardiovascular disease] risk factor testing – i.e., for hypertension, dyslipidemia, and hyperinsulinemia,” Dr. Gunderson said. “Our findings also suggest that it could be beneficial to incorporate history of GD into risk calculators to improve CVD risk stratification and prevention.”
Strong findings argue for more frequent CVD screening
These study results may be the strongest data to date on the long-term effects of GD, said Prakash Deedwania, MD, professor of cardiology at the University of California, San Francisco. “It’s the strongest in the sense in that it’s sponsored, involved four different communities in different parts of the United States, enrolled individuals when they were young and followed them, and saw very few patients drop out for such a long-term study.” The study reported follow-up data on 72% of patients at 25 years, a rate Dr. Deedwania noted was “excellent.”
“Patients who have had GD should be screened aggressively – for not only diabetes, but other cardiovascular risk factors – early on to minimize the subsequent risk of cardiovascular disease is a very important point of this study,” he added. In the absence of a clinical guideline, Dr. Deedwania suggested women with GD might have screening for CV risk factors every 5-7 years depending on their risk profile, but emphasized that parameter isn’t settled.
Future research should focus on the link between GD and CVD risk, Dr. Gunderson said. “Research is needed to better characterize the severity of GD in relation to CVD outcomes, and to identify critical pregnancy-related periods to modify cardiometabolic risk.” The latter would include life-course studies across the full pregnancy continuum from preconception to lactation. “Interventions for primary prevention of CVD and the importance of modifiable lifestyle behaviors with the highest relevance to reduce both diabetes and CVD risks during the first year post partum merit increased research investigation,” she added.
Future studies might also explore the role of inflammation in the GD-CVD relationship, Dr. Deedwania said. “My hypothesis is, and it’s purely a hypothesis, that perhaps the presence of coronary artery calcification scores score in these individuals who were described as having normal glucose but who could be at risk could very well be related to the beginning of inflammation.”
Dr. Gunderson and Dr. Deedwania have no financial relationships to disclose. The study was funded by the National Institute of Diabetes and Digestive and Kidney Diseases and the National Heart, Lung, and Blood Institute.
FROM CIRCULATION
Pelvic pain
A 34-year-old woman with no significant past medical history presented as a new patient to our family medicine clinic with 2 weeks of intermittent lower abdominal and pelvic pain. She was sexually active with 1 partner and denied abnormal vaginal discharge or bleeding. She mentioned she’d had an intrauterine contraceptive device (IUD) placed a few weeks ago. The patient was afebrile, and her pelvic examination was unremarkable.
Physical examination showed mild tenderness to palpation over the lower abdomen without rebound tenderness or guarding. A complete metabolic panel revealed no significant abnormalities, and her human chorionic gonadotropin levels were normal.
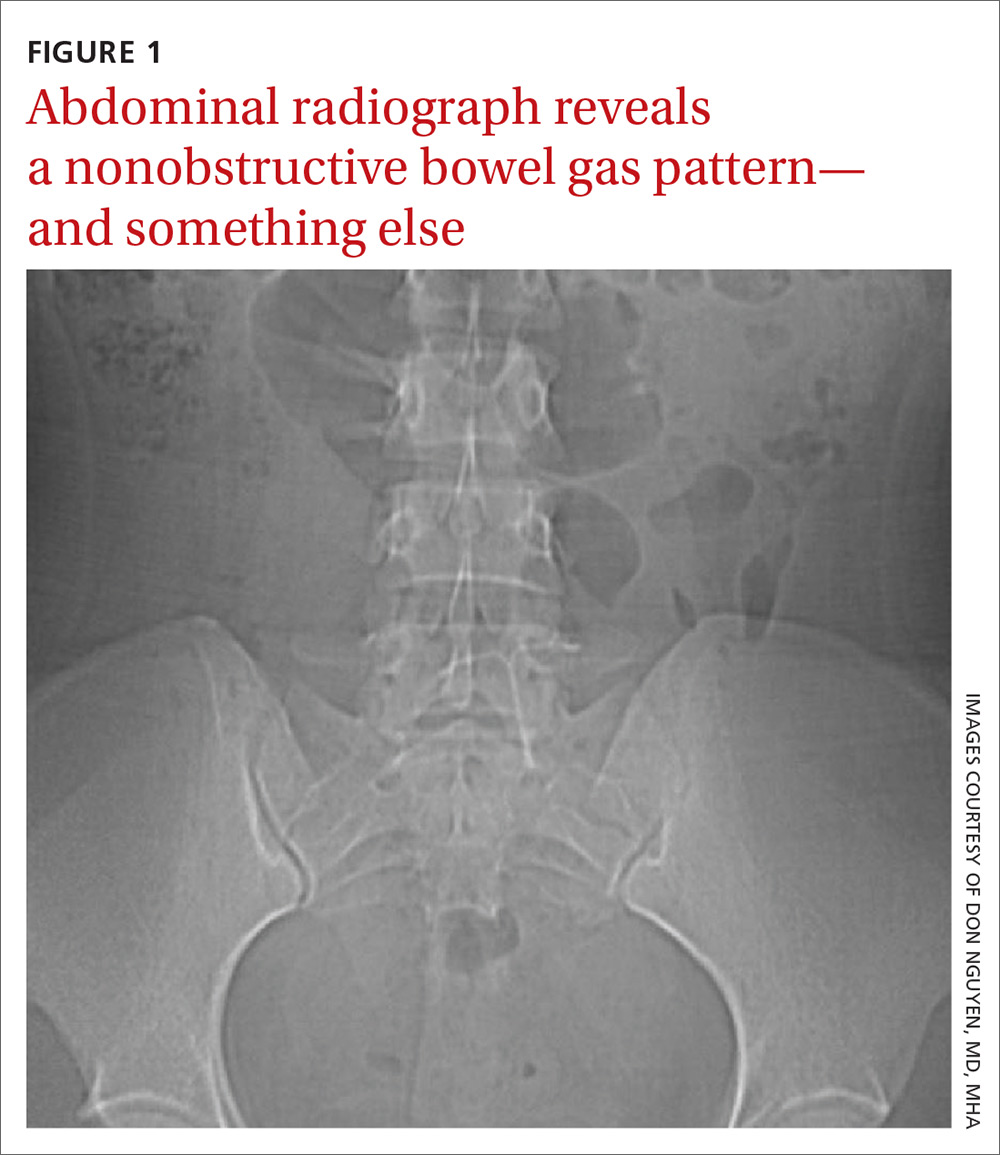
Findings from the physical exam and her clinical history prompted the need for imaging. An abdominal radiograph (FIGURE 1) and noncontrast computed tomography (FIGURES 2A and 2B) were subsequently ordered.
WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Dx: Intra-abdominal IUD migration
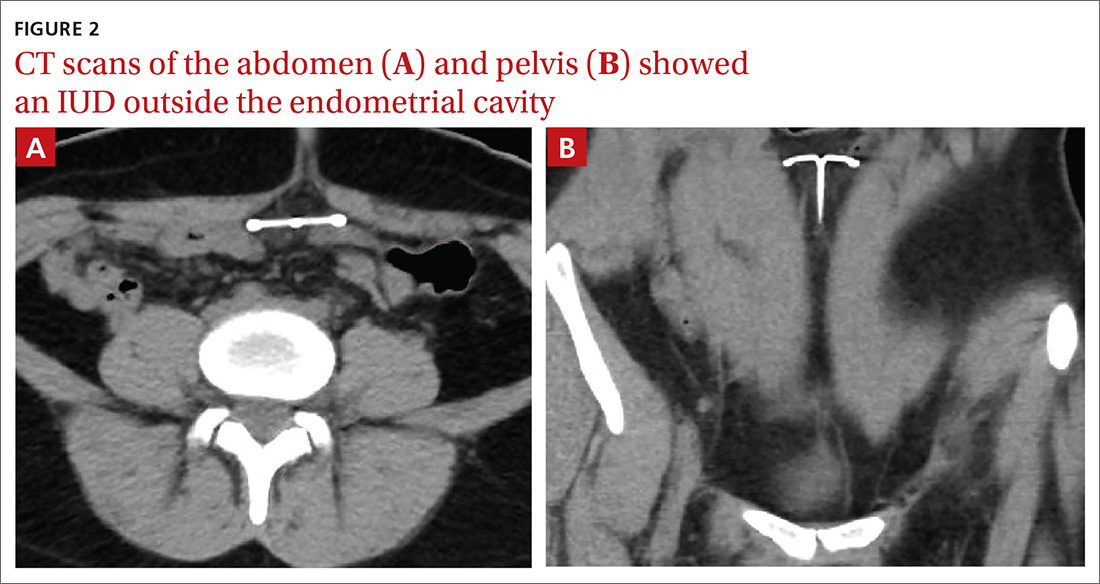
The abdominal radiograph revealed a nonobstructive bowel gas pattern with an IUD overlaying the central lower abdomen and pelvis at the L5-S1 level (FIGURE 1). Computed tomography (CT) of her abdomen and pelvis showed that the IUD was outside the endometrial cavity (FIGURES 2A and 2B). There was no evidence of pneumoperitoneum or bowel perforation. Based on the work-up and imaging, the patient’s pain was due to intra-abdominal IUD malpositioning.
Diagnostic criteria for IUD malpositioning include device migration into 1 of several locations, such as the lower uterine segment or cervix. IUD malpositioning can involve the rotation or protrusion of the device into or through the myometrium. On imaging, a well-positioned IUD should have a straight stem contained within the endometrial cavity, with the arms of the IUD extending laterally at the uterine fundus.
For our patient, an abdominal radiograph showed that her IUD was superiorly displaced outside the expected region of the endometrial cavity. CT helped to confirm this.
Complications with IUDs are few
Using an IUD is an increasingly popular method of contraception because it is effective and generally well tolerated, with minimal adverse effects or complications. In a multicenter retrospective chart review of 2138 patients who had IUDs, Aoun et al found that serious complications included pelvic inflammatory disease (2%), IUD expulsion (6%), and pregnancy (1%).1 In a retrospective cohort study examining complications among 90,489 women with IUDs, Berenson et al found ectopic pregnancy and uterine perforation affected < 1%.2
A less serious complication is IUD malpositioning. Although it does seem to occur more often than other, more serious complications, the exact incidence is unknown. In a retrospective case-control study, Braaten et al reported the rate for IUD malpositioning was 10.4% among 182 women.3 Malpositioned IUDs may be more likely to occur in those with suspected adenomyosis.3 In a study by de Kroon et al, the estimated prevalence rate for an abnormal IUD position ranged from 4% to 7.7% among 195 patients.4
Continue to: The clinical presentation of IUD migration
The clinical presentation of IUD migration
Identification of a malpositioned IUD is needed to avoid the possible increased risk for uterine perforation, IUD expulsion, or pregnancy.5
IUDs that have perforated the uterus float freely in the pelvis or abdomen and can result in injury to adjacent structures as well as peritonitis, fistulas, and hemorrhage.5-7 In addition, adhesion formation over the IUD can lead to intestinal obstruction, infertility, and chronic pain.6
Common symptoms of IUD malpositioning include abdominal or pelvic pain and abnormal bleeding, although many patients may be asymptomatic.8 In a retrospective study of 167 patients with IUDs who underwent pelvic ultrasound, 28 patients were found to have an IUD in an abnormal position.8 Rates of bleeding and pain were higher in patients with malpositioned IUDs (35.7% and 39.3%, respectively) than in those with a normally positioned IUD (15.1% and 19.4%, respectively).8
The differential Dx includes endometriosis and fibroids
IUD malpositioning can be distinguished from other diagnoses that cause pelvic pain and have similar presentations—including endometriosis, ectopic pregnancy, and fibroids—through imaging study findings, clinical history, and presentation.
Other conditions that may need to be ruled out include pelvic inflammatory disease, acute appendicitis, and ovarian cysts.9 A thorough history and physical examination can help rule out these conditions by organ system, and laboratory and imaging studies can help to confirm the diagnosis.
Continue to: Which imaging tool to use, and when
Which imaging tool to use, and when
Assessment of intrauterine contraception placement requires evaluation of the uterine cavity; gynecologic examination alone is not sufficient to fully evaluate for IUD position. Certain imaging studies are particularly helpful for revealing possible IUD migration.
Ultrasound—a widely available, radiation-free modality—is the first-line imaging tool for evaluation of an IUD’s position.10 In addition, ultrasound can provide effective evaluation of other pelvic structures, which is helpful in identifying or eliminating other causes of pain or abnormal bleeding.
Conventional radiography. If the IUD is not visualized on ultrasound, the American College of Obstetricians and Gynecologists (ACOG) recommends radiography to determine if the IUD has been expelled or has migrated to an extra-uterine position.6
CT may be best suited for the evaluation of more severe complications of IUD malpositioning, including visceral perforation, abscess formation, or bowel obstruction. CT should be considered if the patient’s clinical presentation is suspicious for a more serious intra-abdominal pathology.
Management depends on the IUD’s position
For patients whose IUD has an uncertain position or nonvisualized intravaginal strings, ACOG’s first-line recommendations include ruling out pregnancy, using an alternative method for contraception, and ordering pelvic ultrasonography.6 ACOG recommendations for the management of IUD malpositioning depend on the device’s location and the patient’s symptomatology.
Continue to: Management of low-lying IUDs
Management of low-lying IUDs is complex. An IUD that is malpositioned in the cervix is considered partially expelled and should be completely removed.6 For asymptomatic patients with an IUD located in the lower uterine segment and above the internal cervical os, there should be strong consideration given to leaving the IUD in place because removal is associated with higher rates of pregnancy given the low rates of initiation of effective contraception following removal.6
IUD malpositioning in the peritoneal cavity requires surgical intervention. Although ACOG’s first-line recommendation is laparoscopic intervention, laparotomy can be considered if laparoscopy does not result in the removal of the IUD or the patient has more severe complications (sepsis or bowel perforation).6 At the time of IUD removal, the clinician should also discuss and/or prescribe interim contraception.
Treatment for our patient included uncomplicated laparoscopic surgical removal of the intra-abdominal IUD. The patient’s symptoms went away following the procedure, and she was subsequently switched to an oral contraceptive.
1. Aoun J, Dines VA, Stovall DW, et al. Effects of age, parity, and device type on complications and discontinuation of intrauterine devices. Obstet Gynecol. 2014;123:585-592.
2. Berenson AB, Tan A, Hirth JM, et al. Complications and continuation of intrauterine device use among commercially insured teenagers. Obstet Gynecol. 2013;121:951-958.
3. Braaten KP, Benson CB, Maurer R, et al. Malpositioned intrauterine contraceptive devices: risk factors, outcomes, and future pregnancies. Obstet Gynecol. 2011;118:1014-1020.
4. de Kroon CD, van Houwelingen JC, Trimbos JB, et al. The value of transvaginal ultrasound to monitor the position of an intrauterine device after insertion. A technology assessment study. Hum Reprod. 2003;18:2323-2327.
5. Thonneau P, Almont T, de La Rochebrochard E, et al. Risk factors for IUD failure: results of a large multicentre case-control study. Hum Reprod. 2006;21:2612-2616.
6. ACOG Committee on Gynecologic Practice. Committee Opinion No 672: clinical challenges of long-acting reversible contraceptive methods. American College of Obstetricians and Gynecologists. Obstet Gynecol. 2016;128:e69-e77.
7. Heinemann K, Reed S, Moehner S, et al. Risk of uterine perforation with levonorgestrel-releasing and copper intrauterine devices in the European Active Surveillance Study on Intrauterine Devices. Contraception. 2015;91:274-279.
8. Benacerraf BR, Shipp TD, Bromley B. Three-dimensional ultrasound detection of abnormally located intrauterine contraceptive devices which are a source of pelvic pain and abnormal bleeding. Ultrasound Obstet Gynecol. 2009;34:110-115.
9. Bhavasr AK, Felner EJ, Shorma T. Common questions about the evaluation of acute pelvic pain. Am Fam Physician. 2016;93:41-48.
10. Peri N, Graham D, Levine D. Imaging of intrauterine contraceptive devices. J Ultrasound Med. 2007;26:1389-1401.
A 34-year-old woman with no significant past medical history presented as a new patient to our family medicine clinic with 2 weeks of intermittent lower abdominal and pelvic pain. She was sexually active with 1 partner and denied abnormal vaginal discharge or bleeding. She mentioned she’d had an intrauterine contraceptive device (IUD) placed a few weeks ago. The patient was afebrile, and her pelvic examination was unremarkable.
Physical examination showed mild tenderness to palpation over the lower abdomen without rebound tenderness or guarding. A complete metabolic panel revealed no significant abnormalities, and her human chorionic gonadotropin levels were normal.

Findings from the physical exam and her clinical history prompted the need for imaging. An abdominal radiograph (FIGURE 1) and noncontrast computed tomography (FIGURES 2A and 2B) were subsequently ordered.

WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Dx: Intra-abdominal IUD migration
The abdominal radiograph revealed a nonobstructive bowel gas pattern with an IUD overlaying the central lower abdomen and pelvis at the L5-S1 level (FIGURE 1). Computed tomography (CT) of her abdomen and pelvis showed that the IUD was outside the endometrial cavity (FIGURES 2A and 2B). There was no evidence of pneumoperitoneum or bowel perforation. Based on the work-up and imaging, the patient’s pain was due to intra-abdominal IUD malpositioning.
Diagnostic criteria for IUD malpositioning include device migration into 1 of several locations, such as the lower uterine segment or cervix. IUD malpositioning can involve the rotation or protrusion of the device into or through the myometrium. On imaging, a well-positioned IUD should have a straight stem contained within the endometrial cavity, with the arms of the IUD extending laterally at the uterine fundus.
For our patient, an abdominal radiograph showed that her IUD was superiorly displaced outside the expected region of the endometrial cavity. CT helped to confirm this.
Complications with IUDs are few
Using an IUD is an increasingly popular method of contraception because it is effective and generally well tolerated, with minimal adverse effects or complications. In a multicenter retrospective chart review of 2138 patients who had IUDs, Aoun et al found that serious complications included pelvic inflammatory disease (2%), IUD expulsion (6%), and pregnancy (1%).1 In a retrospective cohort study examining complications among 90,489 women with IUDs, Berenson et al found ectopic pregnancy and uterine perforation affected < 1%.2
A less serious complication is IUD malpositioning. Although it does seem to occur more often than other, more serious complications, the exact incidence is unknown. In a retrospective case-control study, Braaten et al reported the rate for IUD malpositioning was 10.4% among 182 women.3 Malpositioned IUDs may be more likely to occur in those with suspected adenomyosis.3 In a study by de Kroon et al, the estimated prevalence rate for an abnormal IUD position ranged from 4% to 7.7% among 195 patients.4
Continue to: The clinical presentation of IUD migration
The clinical presentation of IUD migration
Identification of a malpositioned IUD is needed to avoid the possible increased risk for uterine perforation, IUD expulsion, or pregnancy.5
IUDs that have perforated the uterus float freely in the pelvis or abdomen and can result in injury to adjacent structures as well as peritonitis, fistulas, and hemorrhage.5-7 In addition, adhesion formation over the IUD can lead to intestinal obstruction, infertility, and chronic pain.6
Common symptoms of IUD malpositioning include abdominal or pelvic pain and abnormal bleeding, although many patients may be asymptomatic.8 In a retrospective study of 167 patients with IUDs who underwent pelvic ultrasound, 28 patients were found to have an IUD in an abnormal position.8 Rates of bleeding and pain were higher in patients with malpositioned IUDs (35.7% and 39.3%, respectively) than in those with a normally positioned IUD (15.1% and 19.4%, respectively).8
The differential Dx includes endometriosis and fibroids
IUD malpositioning can be distinguished from other diagnoses that cause pelvic pain and have similar presentations—including endometriosis, ectopic pregnancy, and fibroids—through imaging study findings, clinical history, and presentation.
Other conditions that may need to be ruled out include pelvic inflammatory disease, acute appendicitis, and ovarian cysts.9 A thorough history and physical examination can help rule out these conditions by organ system, and laboratory and imaging studies can help to confirm the diagnosis.
Continue to: Which imaging tool to use, and when
Which imaging tool to use, and when
Assessment of intrauterine contraception placement requires evaluation of the uterine cavity; gynecologic examination alone is not sufficient to fully evaluate for IUD position. Certain imaging studies are particularly helpful for revealing possible IUD migration.
Ultrasound—a widely available, radiation-free modality—is the first-line imaging tool for evaluation of an IUD’s position.10 In addition, ultrasound can provide effective evaluation of other pelvic structures, which is helpful in identifying or eliminating other causes of pain or abnormal bleeding.
Conventional radiography. If the IUD is not visualized on ultrasound, the American College of Obstetricians and Gynecologists (ACOG) recommends radiography to determine if the IUD has been expelled or has migrated to an extra-uterine position.6
CT may be best suited for the evaluation of more severe complications of IUD malpositioning, including visceral perforation, abscess formation, or bowel obstruction. CT should be considered if the patient’s clinical presentation is suspicious for a more serious intra-abdominal pathology.
Management depends on the IUD’s position
For patients whose IUD has an uncertain position or nonvisualized intravaginal strings, ACOG’s first-line recommendations include ruling out pregnancy, using an alternative method for contraception, and ordering pelvic ultrasonography.6 ACOG recommendations for the management of IUD malpositioning depend on the device’s location and the patient’s symptomatology.
Continue to: Management of low-lying IUDs
Management of low-lying IUDs is complex. An IUD that is malpositioned in the cervix is considered partially expelled and should be completely removed.6 For asymptomatic patients with an IUD located in the lower uterine segment and above the internal cervical os, there should be strong consideration given to leaving the IUD in place because removal is associated with higher rates of pregnancy given the low rates of initiation of effective contraception following removal.6
IUD malpositioning in the peritoneal cavity requires surgical intervention. Although ACOG’s first-line recommendation is laparoscopic intervention, laparotomy can be considered if laparoscopy does not result in the removal of the IUD or the patient has more severe complications (sepsis or bowel perforation).6 At the time of IUD removal, the clinician should also discuss and/or prescribe interim contraception.
Treatment for our patient included uncomplicated laparoscopic surgical removal of the intra-abdominal IUD. The patient’s symptoms went away following the procedure, and she was subsequently switched to an oral contraceptive.
A 34-year-old woman with no significant past medical history presented as a new patient to our family medicine clinic with 2 weeks of intermittent lower abdominal and pelvic pain. She was sexually active with 1 partner and denied abnormal vaginal discharge or bleeding. She mentioned she’d had an intrauterine contraceptive device (IUD) placed a few weeks ago. The patient was afebrile, and her pelvic examination was unremarkable.
Physical examination showed mild tenderness to palpation over the lower abdomen without rebound tenderness or guarding. A complete metabolic panel revealed no significant abnormalities, and her human chorionic gonadotropin levels were normal.

Findings from the physical exam and her clinical history prompted the need for imaging. An abdominal radiograph (FIGURE 1) and noncontrast computed tomography (FIGURES 2A and 2B) were subsequently ordered.

WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Dx: Intra-abdominal IUD migration
The abdominal radiograph revealed a nonobstructive bowel gas pattern with an IUD overlaying the central lower abdomen and pelvis at the L5-S1 level (FIGURE 1). Computed tomography (CT) of her abdomen and pelvis showed that the IUD was outside the endometrial cavity (FIGURES 2A and 2B). There was no evidence of pneumoperitoneum or bowel perforation. Based on the work-up and imaging, the patient’s pain was due to intra-abdominal IUD malpositioning.
Diagnostic criteria for IUD malpositioning include device migration into 1 of several locations, such as the lower uterine segment or cervix. IUD malpositioning can involve the rotation or protrusion of the device into or through the myometrium. On imaging, a well-positioned IUD should have a straight stem contained within the endometrial cavity, with the arms of the IUD extending laterally at the uterine fundus.
For our patient, an abdominal radiograph showed that her IUD was superiorly displaced outside the expected region of the endometrial cavity. CT helped to confirm this.
Complications with IUDs are few
Using an IUD is an increasingly popular method of contraception because it is effective and generally well tolerated, with minimal adverse effects or complications. In a multicenter retrospective chart review of 2138 patients who had IUDs, Aoun et al found that serious complications included pelvic inflammatory disease (2%), IUD expulsion (6%), and pregnancy (1%).1 In a retrospective cohort study examining complications among 90,489 women with IUDs, Berenson et al found ectopic pregnancy and uterine perforation affected < 1%.2
A less serious complication is IUD malpositioning. Although it does seem to occur more often than other, more serious complications, the exact incidence is unknown. In a retrospective case-control study, Braaten et al reported the rate for IUD malpositioning was 10.4% among 182 women.3 Malpositioned IUDs may be more likely to occur in those with suspected adenomyosis.3 In a study by de Kroon et al, the estimated prevalence rate for an abnormal IUD position ranged from 4% to 7.7% among 195 patients.4
Continue to: The clinical presentation of IUD migration
The clinical presentation of IUD migration
Identification of a malpositioned IUD is needed to avoid the possible increased risk for uterine perforation, IUD expulsion, or pregnancy.5
IUDs that have perforated the uterus float freely in the pelvis or abdomen and can result in injury to adjacent structures as well as peritonitis, fistulas, and hemorrhage.5-7 In addition, adhesion formation over the IUD can lead to intestinal obstruction, infertility, and chronic pain.6
Common symptoms of IUD malpositioning include abdominal or pelvic pain and abnormal bleeding, although many patients may be asymptomatic.8 In a retrospective study of 167 patients with IUDs who underwent pelvic ultrasound, 28 patients were found to have an IUD in an abnormal position.8 Rates of bleeding and pain were higher in patients with malpositioned IUDs (35.7% and 39.3%, respectively) than in those with a normally positioned IUD (15.1% and 19.4%, respectively).8
The differential Dx includes endometriosis and fibroids
IUD malpositioning can be distinguished from other diagnoses that cause pelvic pain and have similar presentations—including endometriosis, ectopic pregnancy, and fibroids—through imaging study findings, clinical history, and presentation.
Other conditions that may need to be ruled out include pelvic inflammatory disease, acute appendicitis, and ovarian cysts.9 A thorough history and physical examination can help rule out these conditions by organ system, and laboratory and imaging studies can help to confirm the diagnosis.
Continue to: Which imaging tool to use, and when
Which imaging tool to use, and when
Assessment of intrauterine contraception placement requires evaluation of the uterine cavity; gynecologic examination alone is not sufficient to fully evaluate for IUD position. Certain imaging studies are particularly helpful for revealing possible IUD migration.
Ultrasound—a widely available, radiation-free modality—is the first-line imaging tool for evaluation of an IUD’s position.10 In addition, ultrasound can provide effective evaluation of other pelvic structures, which is helpful in identifying or eliminating other causes of pain or abnormal bleeding.
Conventional radiography. If the IUD is not visualized on ultrasound, the American College of Obstetricians and Gynecologists (ACOG) recommends radiography to determine if the IUD has been expelled or has migrated to an extra-uterine position.6
CT may be best suited for the evaluation of more severe complications of IUD malpositioning, including visceral perforation, abscess formation, or bowel obstruction. CT should be considered if the patient’s clinical presentation is suspicious for a more serious intra-abdominal pathology.
Management depends on the IUD’s position
For patients whose IUD has an uncertain position or nonvisualized intravaginal strings, ACOG’s first-line recommendations include ruling out pregnancy, using an alternative method for contraception, and ordering pelvic ultrasonography.6 ACOG recommendations for the management of IUD malpositioning depend on the device’s location and the patient’s symptomatology.
Continue to: Management of low-lying IUDs
Management of low-lying IUDs is complex. An IUD that is malpositioned in the cervix is considered partially expelled and should be completely removed.6 For asymptomatic patients with an IUD located in the lower uterine segment and above the internal cervical os, there should be strong consideration given to leaving the IUD in place because removal is associated with higher rates of pregnancy given the low rates of initiation of effective contraception following removal.6
IUD malpositioning in the peritoneal cavity requires surgical intervention. Although ACOG’s first-line recommendation is laparoscopic intervention, laparotomy can be considered if laparoscopy does not result in the removal of the IUD or the patient has more severe complications (sepsis or bowel perforation).6 At the time of IUD removal, the clinician should also discuss and/or prescribe interim contraception.
Treatment for our patient included uncomplicated laparoscopic surgical removal of the intra-abdominal IUD. The patient’s symptoms went away following the procedure, and she was subsequently switched to an oral contraceptive.
1. Aoun J, Dines VA, Stovall DW, et al. Effects of age, parity, and device type on complications and discontinuation of intrauterine devices. Obstet Gynecol. 2014;123:585-592.
2. Berenson AB, Tan A, Hirth JM, et al. Complications and continuation of intrauterine device use among commercially insured teenagers. Obstet Gynecol. 2013;121:951-958.
3. Braaten KP, Benson CB, Maurer R, et al. Malpositioned intrauterine contraceptive devices: risk factors, outcomes, and future pregnancies. Obstet Gynecol. 2011;118:1014-1020.
4. de Kroon CD, van Houwelingen JC, Trimbos JB, et al. The value of transvaginal ultrasound to monitor the position of an intrauterine device after insertion. A technology assessment study. Hum Reprod. 2003;18:2323-2327.
5. Thonneau P, Almont T, de La Rochebrochard E, et al. Risk factors for IUD failure: results of a large multicentre case-control study. Hum Reprod. 2006;21:2612-2616.
6. ACOG Committee on Gynecologic Practice. Committee Opinion No 672: clinical challenges of long-acting reversible contraceptive methods. American College of Obstetricians and Gynecologists. Obstet Gynecol. 2016;128:e69-e77.
7. Heinemann K, Reed S, Moehner S, et al. Risk of uterine perforation with levonorgestrel-releasing and copper intrauterine devices in the European Active Surveillance Study on Intrauterine Devices. Contraception. 2015;91:274-279.
8. Benacerraf BR, Shipp TD, Bromley B. Three-dimensional ultrasound detection of abnormally located intrauterine contraceptive devices which are a source of pelvic pain and abnormal bleeding. Ultrasound Obstet Gynecol. 2009;34:110-115.
9. Bhavasr AK, Felner EJ, Shorma T. Common questions about the evaluation of acute pelvic pain. Am Fam Physician. 2016;93:41-48.
10. Peri N, Graham D, Levine D. Imaging of intrauterine contraceptive devices. J Ultrasound Med. 2007;26:1389-1401.
1. Aoun J, Dines VA, Stovall DW, et al. Effects of age, parity, and device type on complications and discontinuation of intrauterine devices. Obstet Gynecol. 2014;123:585-592.
2. Berenson AB, Tan A, Hirth JM, et al. Complications and continuation of intrauterine device use among commercially insured teenagers. Obstet Gynecol. 2013;121:951-958.
3. Braaten KP, Benson CB, Maurer R, et al. Malpositioned intrauterine contraceptive devices: risk factors, outcomes, and future pregnancies. Obstet Gynecol. 2011;118:1014-1020.
4. de Kroon CD, van Houwelingen JC, Trimbos JB, et al. The value of transvaginal ultrasound to monitor the position of an intrauterine device after insertion. A technology assessment study. Hum Reprod. 2003;18:2323-2327.
5. Thonneau P, Almont T, de La Rochebrochard E, et al. Risk factors for IUD failure: results of a large multicentre case-control study. Hum Reprod. 2006;21:2612-2616.
6. ACOG Committee on Gynecologic Practice. Committee Opinion No 672: clinical challenges of long-acting reversible contraceptive methods. American College of Obstetricians and Gynecologists. Obstet Gynecol. 2016;128:e69-e77.
7. Heinemann K, Reed S, Moehner S, et al. Risk of uterine perforation with levonorgestrel-releasing and copper intrauterine devices in the European Active Surveillance Study on Intrauterine Devices. Contraception. 2015;91:274-279.
8. Benacerraf BR, Shipp TD, Bromley B. Three-dimensional ultrasound detection of abnormally located intrauterine contraceptive devices which are a source of pelvic pain and abnormal bleeding. Ultrasound Obstet Gynecol. 2009;34:110-115.
9. Bhavasr AK, Felner EJ, Shorma T. Common questions about the evaluation of acute pelvic pain. Am Fam Physician. 2016;93:41-48.
10. Peri N, Graham D, Levine D. Imaging of intrauterine contraceptive devices. J Ultrasound Med. 2007;26:1389-1401.
First mammography guidelines for older breast cancer survivors
For women who have a life expectancy of 5-10 years, the guidelines recommend that consideration be given to discontinuing mammography.
Overall, the guidelines encourage shared decision-making that is individualized for each woman after weighing the benefits and harms associated with surveillance mammography and patient preferences.
The panel also recommended that patients with clinical findings and symptoms receive ongoing clinical breast examinations and diagnostic mammography and that patients be reassured that these practices will continue.
Guidelines on breast cancer screening for healthy women already “acknowledge the limitations of mammograms and the need to consider one’s health status and preferences when making decisions on how and when to stop routine mammograms,” said the article’s first author, Rachel A. Freedman, MD, MPH, of the Dana-Farber Cancer Institute, Boston.
However, “we don’t have this kind of consensus for women with a history of breast cancer,” she continued. “Current follow-up care guidelines simply state that women with a history of breast cancer with intact breasts should have annual mammography without any guidance.
“In practice, the use of mammograms is highly variable, with less than 50% of breast cancer survivors who have limited life expectancy having annual mammograms, according to survey data we have from prior work,” Dr. Freedman said in an interview.
The guidelines were published online Jan. 28 in JAMA Oncology.
Clinicians discuss how to have these discussions
As part of the process of developing these expert consensus guidelines, the researchers held several clinical focus groups that involved primary care physicians from Brigham and Women’s Hospital and oncology clinicians (including breast surgeons and medical oncologists) from the Dana-Farber Cancer Institute.
All clinicians felt that having expert guidelines and talking points to guide discussions would be helpful, the researchers report.
“However, some oncology clinicians felt that 75 years is often ‘too young’ to stop surveillance mammography and that 80 years may be a more comfortable age to stop routine testing,” they write. “Most clinicians felt that estimations of life expectancy, more than age, should inform the timing of this discussion.”
In contrast to primary and geriatric care clinicians, oncology clinicians reported discomfort with such discussions. They appreciated having the information but “felt it was easier to communicate findings indirectly, without specifically revealing life expectancy to patients. One oncology clinician, however, felt it would be ‘sneaky’ to calculate life expectancy without communicating this to patients, supporting more open discussions,” the authors report.
“All clinicians acknowledged that framing the conversation around patients’ low risk for in-breast cancer events and how mammography will not benefit them was more appealing than discussing life expectancy,” the researchers continue. Their literature review found that the risk of these individuals developing second breast cancers was similar to that of a healthy woman developing a first breast cancer, leading one clinician to comment: “If their risk is really equivalent to the general population – that is very powerful.”
“Some clinicians reported that they ‘focus on the risks’ or frame such discussions by asking: ‘If you were to find something on [a] mammogram, would you do anything about it?’ If a patient answered no, clinicians felt this was a signal to stop mammography,” they noted.
Literature review finds very low risk
Dr. Freedman and colleagues conducted a literature review of the risk for ipsilateral and contralateral breast cancer events among survivors and of the harms and benefits associated with mammography. Following the literature review, a multidisciplinary expert panel, which included patients and patient advocates, was convened to develop consensus guidelines.
The literature review confirmed that there was a low risk for in-breast cancer events in this population and that the risk was particularly low among patients who undergo treatment with endocrine therapy. Among those who did not receive systemic therapy for ERBB2-positive or triple-negative cancers, the rates of ipsilateral recurrence were estimated to be higher.
On the basis of the literature review, the estimated 10-year risk for in-breast cancer events ranged from 1% to 15% for ipsilateral breast cancers and from 1% to 5% for contralateral cancers. Among women in the same age group who did not have a history of breast cancer, the 5-year risk of developing the disease (average risk) was 2.2%.
The authors note that these findings mirror their estimates for new breast cancers among survivors who had low-risk disease. The findings are also similar to those cited in a large-scale mammography study, in which breast cancer survivors aged 70-80 years had a 1.1% annual risk for in-breast cancers. The risk was 0.7%-0.9% for similarly aged patients who did not have a history of breast cancer.
The benefits associated with mammography for older women are not well defined, but the literature suggests that mammography offers little to modest clinical benefit for patients in this age group. The limited benefits are likely because of the more than 10-year time lag that is needed to detect the small improvements in breast cancer mortality; slow-growing tumors generally do not affect the life expectancy of older women, they point out.
“Through our expert consensus process and after iterative feedback from clinicians, we created guidelines to support patients and clinicians in making individualized decisions on how and when to stop mammography,” said Dr. Freedman. “These guidelines are based on the risk of a breast cancer returning in the breast, one’s underlying health, and one’s preferences.”
The guidelines are also intended to provide information to patients on the benefits and harms of mammography in this setting, in addition to “how much we anticipate a mammogram may or may not continue to help a woman over time,” she said.
A companion guide for patients on these guidelines will be published in the coming months.
Dr. Freedman has received institutional clinical trial funding from Eisai and Puma Biotechnology outside the submitted work.
A version of this article first appeared on Medscape.com.
For women who have a life expectancy of 5-10 years, the guidelines recommend that consideration be given to discontinuing mammography.
Overall, the guidelines encourage shared decision-making that is individualized for each woman after weighing the benefits and harms associated with surveillance mammography and patient preferences.
The panel also recommended that patients with clinical findings and symptoms receive ongoing clinical breast examinations and diagnostic mammography and that patients be reassured that these practices will continue.
Guidelines on breast cancer screening for healthy women already “acknowledge the limitations of mammograms and the need to consider one’s health status and preferences when making decisions on how and when to stop routine mammograms,” said the article’s first author, Rachel A. Freedman, MD, MPH, of the Dana-Farber Cancer Institute, Boston.
However, “we don’t have this kind of consensus for women with a history of breast cancer,” she continued. “Current follow-up care guidelines simply state that women with a history of breast cancer with intact breasts should have annual mammography without any guidance.
“In practice, the use of mammograms is highly variable, with less than 50% of breast cancer survivors who have limited life expectancy having annual mammograms, according to survey data we have from prior work,” Dr. Freedman said in an interview.
The guidelines were published online Jan. 28 in JAMA Oncology.
Clinicians discuss how to have these discussions
As part of the process of developing these expert consensus guidelines, the researchers held several clinical focus groups that involved primary care physicians from Brigham and Women’s Hospital and oncology clinicians (including breast surgeons and medical oncologists) from the Dana-Farber Cancer Institute.
All clinicians felt that having expert guidelines and talking points to guide discussions would be helpful, the researchers report.
“However, some oncology clinicians felt that 75 years is often ‘too young’ to stop surveillance mammography and that 80 years may be a more comfortable age to stop routine testing,” they write. “Most clinicians felt that estimations of life expectancy, more than age, should inform the timing of this discussion.”
In contrast to primary and geriatric care clinicians, oncology clinicians reported discomfort with such discussions. They appreciated having the information but “felt it was easier to communicate findings indirectly, without specifically revealing life expectancy to patients. One oncology clinician, however, felt it would be ‘sneaky’ to calculate life expectancy without communicating this to patients, supporting more open discussions,” the authors report.
“All clinicians acknowledged that framing the conversation around patients’ low risk for in-breast cancer events and how mammography will not benefit them was more appealing than discussing life expectancy,” the researchers continue. Their literature review found that the risk of these individuals developing second breast cancers was similar to that of a healthy woman developing a first breast cancer, leading one clinician to comment: “If their risk is really equivalent to the general population – that is very powerful.”
“Some clinicians reported that they ‘focus on the risks’ or frame such discussions by asking: ‘If you were to find something on [a] mammogram, would you do anything about it?’ If a patient answered no, clinicians felt this was a signal to stop mammography,” they noted.
Literature review finds very low risk
Dr. Freedman and colleagues conducted a literature review of the risk for ipsilateral and contralateral breast cancer events among survivors and of the harms and benefits associated with mammography. Following the literature review, a multidisciplinary expert panel, which included patients and patient advocates, was convened to develop consensus guidelines.
The literature review confirmed that there was a low risk for in-breast cancer events in this population and that the risk was particularly low among patients who undergo treatment with endocrine therapy. Among those who did not receive systemic therapy for ERBB2-positive or triple-negative cancers, the rates of ipsilateral recurrence were estimated to be higher.
On the basis of the literature review, the estimated 10-year risk for in-breast cancer events ranged from 1% to 15% for ipsilateral breast cancers and from 1% to 5% for contralateral cancers. Among women in the same age group who did not have a history of breast cancer, the 5-year risk of developing the disease (average risk) was 2.2%.
The authors note that these findings mirror their estimates for new breast cancers among survivors who had low-risk disease. The findings are also similar to those cited in a large-scale mammography study, in which breast cancer survivors aged 70-80 years had a 1.1% annual risk for in-breast cancers. The risk was 0.7%-0.9% for similarly aged patients who did not have a history of breast cancer.
The benefits associated with mammography for older women are not well defined, but the literature suggests that mammography offers little to modest clinical benefit for patients in this age group. The limited benefits are likely because of the more than 10-year time lag that is needed to detect the small improvements in breast cancer mortality; slow-growing tumors generally do not affect the life expectancy of older women, they point out.
“Through our expert consensus process and after iterative feedback from clinicians, we created guidelines to support patients and clinicians in making individualized decisions on how and when to stop mammography,” said Dr. Freedman. “These guidelines are based on the risk of a breast cancer returning in the breast, one’s underlying health, and one’s preferences.”
The guidelines are also intended to provide information to patients on the benefits and harms of mammography in this setting, in addition to “how much we anticipate a mammogram may or may not continue to help a woman over time,” she said.
A companion guide for patients on these guidelines will be published in the coming months.
Dr. Freedman has received institutional clinical trial funding from Eisai and Puma Biotechnology outside the submitted work.
A version of this article first appeared on Medscape.com.
For women who have a life expectancy of 5-10 years, the guidelines recommend that consideration be given to discontinuing mammography.
Overall, the guidelines encourage shared decision-making that is individualized for each woman after weighing the benefits and harms associated with surveillance mammography and patient preferences.
The panel also recommended that patients with clinical findings and symptoms receive ongoing clinical breast examinations and diagnostic mammography and that patients be reassured that these practices will continue.
Guidelines on breast cancer screening for healthy women already “acknowledge the limitations of mammograms and the need to consider one’s health status and preferences when making decisions on how and when to stop routine mammograms,” said the article’s first author, Rachel A. Freedman, MD, MPH, of the Dana-Farber Cancer Institute, Boston.
However, “we don’t have this kind of consensus for women with a history of breast cancer,” she continued. “Current follow-up care guidelines simply state that women with a history of breast cancer with intact breasts should have annual mammography without any guidance.
“In practice, the use of mammograms is highly variable, with less than 50% of breast cancer survivors who have limited life expectancy having annual mammograms, according to survey data we have from prior work,” Dr. Freedman said in an interview.
The guidelines were published online Jan. 28 in JAMA Oncology.
Clinicians discuss how to have these discussions
As part of the process of developing these expert consensus guidelines, the researchers held several clinical focus groups that involved primary care physicians from Brigham and Women’s Hospital and oncology clinicians (including breast surgeons and medical oncologists) from the Dana-Farber Cancer Institute.
All clinicians felt that having expert guidelines and talking points to guide discussions would be helpful, the researchers report.
“However, some oncology clinicians felt that 75 years is often ‘too young’ to stop surveillance mammography and that 80 years may be a more comfortable age to stop routine testing,” they write. “Most clinicians felt that estimations of life expectancy, more than age, should inform the timing of this discussion.”
In contrast to primary and geriatric care clinicians, oncology clinicians reported discomfort with such discussions. They appreciated having the information but “felt it was easier to communicate findings indirectly, without specifically revealing life expectancy to patients. One oncology clinician, however, felt it would be ‘sneaky’ to calculate life expectancy without communicating this to patients, supporting more open discussions,” the authors report.
“All clinicians acknowledged that framing the conversation around patients’ low risk for in-breast cancer events and how mammography will not benefit them was more appealing than discussing life expectancy,” the researchers continue. Their literature review found that the risk of these individuals developing second breast cancers was similar to that of a healthy woman developing a first breast cancer, leading one clinician to comment: “If their risk is really equivalent to the general population – that is very powerful.”
“Some clinicians reported that they ‘focus on the risks’ or frame such discussions by asking: ‘If you were to find something on [a] mammogram, would you do anything about it?’ If a patient answered no, clinicians felt this was a signal to stop mammography,” they noted.
Literature review finds very low risk
Dr. Freedman and colleagues conducted a literature review of the risk for ipsilateral and contralateral breast cancer events among survivors and of the harms and benefits associated with mammography. Following the literature review, a multidisciplinary expert panel, which included patients and patient advocates, was convened to develop consensus guidelines.
The literature review confirmed that there was a low risk for in-breast cancer events in this population and that the risk was particularly low among patients who undergo treatment with endocrine therapy. Among those who did not receive systemic therapy for ERBB2-positive or triple-negative cancers, the rates of ipsilateral recurrence were estimated to be higher.
On the basis of the literature review, the estimated 10-year risk for in-breast cancer events ranged from 1% to 15% for ipsilateral breast cancers and from 1% to 5% for contralateral cancers. Among women in the same age group who did not have a history of breast cancer, the 5-year risk of developing the disease (average risk) was 2.2%.
The authors note that these findings mirror their estimates for new breast cancers among survivors who had low-risk disease. The findings are also similar to those cited in a large-scale mammography study, in which breast cancer survivors aged 70-80 years had a 1.1% annual risk for in-breast cancers. The risk was 0.7%-0.9% for similarly aged patients who did not have a history of breast cancer.
The benefits associated with mammography for older women are not well defined, but the literature suggests that mammography offers little to modest clinical benefit for patients in this age group. The limited benefits are likely because of the more than 10-year time lag that is needed to detect the small improvements in breast cancer mortality; slow-growing tumors generally do not affect the life expectancy of older women, they point out.
“Through our expert consensus process and after iterative feedback from clinicians, we created guidelines to support patients and clinicians in making individualized decisions on how and when to stop mammography,” said Dr. Freedman. “These guidelines are based on the risk of a breast cancer returning in the breast, one’s underlying health, and one’s preferences.”
The guidelines are also intended to provide information to patients on the benefits and harms of mammography in this setting, in addition to “how much we anticipate a mammogram may or may not continue to help a woman over time,” she said.
A companion guide for patients on these guidelines will be published in the coming months.
Dr. Freedman has received institutional clinical trial funding from Eisai and Puma Biotechnology outside the submitted work.
A version of this article first appeared on Medscape.com.
Meta-analysis finds much less lupus than expected
The prevalence of systemic lupus erythematosus (SLE) appears to be much lower than previously believed and may pose “a potential risk to research funding for the disease,” according to results of a meta-analysis involving a network of population-based registries.
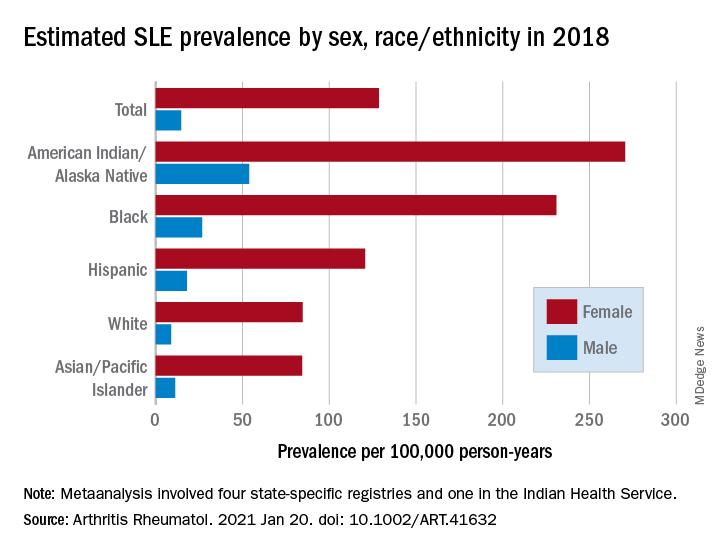
“When we started this study, a widely cited lupus statistic was that approximately 1.5 million Americans were affected. Our meta-analysis found the actual prevalence to be slightly more than 200,000: a number that approaches the [Food and Drug Administration’s] definition of a rare disease,” Emily Somers, PhD, ScM, senior author and associate professor of rheumatology and environmental health sciences at the University of Michigan, Ann Arbor, said in a written statement.
Their estimates, published online in Arthritis & Rheumatology, put the overall SLE prevalence in the United States at 72.8 per 100,000 person-years in 2018, with nearly nine times more females affected (128.7 cases per 100,000) than males (14.6 per 100,000). Race and ethnicity also play a role, as prevalence was highest among American Indian/Alaska Native and Black females, with Hispanic females lower but still higher than White and Asian/Pacific Islander females, Peter M. Izmirly, MD, MSc, of New York University, the lead author, and associates said.
SLE prevalence was distributed similarly in men, although there was a greater relative margin between American Indians/Alaska Natives (53.8 cases per 100,000 person-years) and Blacks (26.7 per 100,000), and Asians/Pacific Islanders were higher than Whites (11.2 vs. 8.9), the investigators reported.
The meta-analysis leveraged data from the Centers for Disease Control and Prevention’s national lupus registries, which include four state-specific SLE registries and a fifth in the Indian Health Service. All cases of SLE occurred in 2002-2009, and the data were age adjusted to the 2000 U.S. population and separately extrapolated to the 2018 U.S. Census population, they explained.
The analysis was funded by cooperative agreements between the New York City Department of Health and Mental Hygiene and New York University, and the CDC and National Institute of Health.
The prevalence of systemic lupus erythematosus (SLE) appears to be much lower than previously believed and may pose “a potential risk to research funding for the disease,” according to results of a meta-analysis involving a network of population-based registries.

“When we started this study, a widely cited lupus statistic was that approximately 1.5 million Americans were affected. Our meta-analysis found the actual prevalence to be slightly more than 200,000: a number that approaches the [Food and Drug Administration’s] definition of a rare disease,” Emily Somers, PhD, ScM, senior author and associate professor of rheumatology and environmental health sciences at the University of Michigan, Ann Arbor, said in a written statement.
Their estimates, published online in Arthritis & Rheumatology, put the overall SLE prevalence in the United States at 72.8 per 100,000 person-years in 2018, with nearly nine times more females affected (128.7 cases per 100,000) than males (14.6 per 100,000). Race and ethnicity also play a role, as prevalence was highest among American Indian/Alaska Native and Black females, with Hispanic females lower but still higher than White and Asian/Pacific Islander females, Peter M. Izmirly, MD, MSc, of New York University, the lead author, and associates said.
SLE prevalence was distributed similarly in men, although there was a greater relative margin between American Indians/Alaska Natives (53.8 cases per 100,000 person-years) and Blacks (26.7 per 100,000), and Asians/Pacific Islanders were higher than Whites (11.2 vs. 8.9), the investigators reported.
The meta-analysis leveraged data from the Centers for Disease Control and Prevention’s national lupus registries, which include four state-specific SLE registries and a fifth in the Indian Health Service. All cases of SLE occurred in 2002-2009, and the data were age adjusted to the 2000 U.S. population and separately extrapolated to the 2018 U.S. Census population, they explained.
The analysis was funded by cooperative agreements between the New York City Department of Health and Mental Hygiene and New York University, and the CDC and National Institute of Health.
The prevalence of systemic lupus erythematosus (SLE) appears to be much lower than previously believed and may pose “a potential risk to research funding for the disease,” according to results of a meta-analysis involving a network of population-based registries.

“When we started this study, a widely cited lupus statistic was that approximately 1.5 million Americans were affected. Our meta-analysis found the actual prevalence to be slightly more than 200,000: a number that approaches the [Food and Drug Administration’s] definition of a rare disease,” Emily Somers, PhD, ScM, senior author and associate professor of rheumatology and environmental health sciences at the University of Michigan, Ann Arbor, said in a written statement.
Their estimates, published online in Arthritis & Rheumatology, put the overall SLE prevalence in the United States at 72.8 per 100,000 person-years in 2018, with nearly nine times more females affected (128.7 cases per 100,000) than males (14.6 per 100,000). Race and ethnicity also play a role, as prevalence was highest among American Indian/Alaska Native and Black females, with Hispanic females lower but still higher than White and Asian/Pacific Islander females, Peter M. Izmirly, MD, MSc, of New York University, the lead author, and associates said.
SLE prevalence was distributed similarly in men, although there was a greater relative margin between American Indians/Alaska Natives (53.8 cases per 100,000 person-years) and Blacks (26.7 per 100,000), and Asians/Pacific Islanders were higher than Whites (11.2 vs. 8.9), the investigators reported.
The meta-analysis leveraged data from the Centers for Disease Control and Prevention’s national lupus registries, which include four state-specific SLE registries and a fifth in the Indian Health Service. All cases of SLE occurred in 2002-2009, and the data were age adjusted to the 2000 U.S. population and separately extrapolated to the 2018 U.S. Census population, they explained.
The analysis was funded by cooperative agreements between the New York City Department of Health and Mental Hygiene and New York University, and the CDC and National Institute of Health.
FROM ARTHRITIS & RHEUMATOLOGY
Severe maternal morbidity promotes long-term mortality
Women who experienced severe complications during pregnancy were more than twice as likely to die at any time after their last pregnancy, including post partum and beyond, based on data from more than 1 million women.
“Current data suggest that up to 88% of maternal deaths are preceded by severe maternal morbidity,” but the long-term risk of mortality and the effect of severe maternal morbidity has not been well studied, wrote U. Vivian Ukah, MPH, PhD, of McGill University, Montreal, and colleagues.
In a longitudinal cohort study published in Obstetrics & Gynecology, the researchers identified 1,229,306 pregnant women who delivered in Quebec between 1989 and 2016.
The primary outcome of in-hospital mortality after the last pregnancy, either post partum (within 42 days of delivery) or long term (43 days to 29 years after delivery).
Overall, 2.9% of the study population experienced severe maternal morbidity, with an associated mortality rate of 0.86 per 1,000 person-years versus 0.41 per 1,000 person-years in women without severe maternal morbidity. The median time to death for women with severe maternal mortality was 6.8 years, compared with 151 years for those without severe maternal morbidity.
The death rate at any time after delivery, post partum and beyond, was twice as high among women with severe maternal morbidity. The morbidities most often associated with long-term mortality after 42 days were severe cardiac complications (hazard ratio, 7.00), acute renal failure (HR, 4.35), and cerebrovascular accidents (HR, 4.03).
However, the mortality risk following severe maternal morbidity decreased over time, the researchers noted. Severe maternal morbidity was associated with 6.73 times the mortality risk, compared with no morbidity, during the period from 43 days to 11 months, but this difference dropped to 1.91, 1.77, and 1.18 times the risk, compared with no comorbidity, at 1-4 years, 5-9 years, and 10-29 years, respectively.
The study findings were limited by several factors, including the inability to prove causality, the use of only in-hospital mortality data, and the potential for missed cases that fell outside the Canadian Perinatal Surveillance System definition of severe maternal morbidity, the researchers noted.
However, the results suggest that identifying severe maternal morbidity may help identify women at risk for postpartum and long-term premature mortality. “Women with severe maternal morbidity may benefit from continued surveillance and preventative interventions to reduce the risk of premature mortality,” they concluded.
Increased morbidity rates drive need for research
“In this retrospective longitudinal cohort study of over 1.2 million women delivering in Quebec between 1989 and 2016, Dr. Ukah and her colleagues demonstrated the association between severe maternal morbidity [SMM] and an accelerated risk of mortality beyond the postpartum period, compared with women who do not experience SMM,” Rachel Humphrey, MD, a maternal-fetal medicine specialist at Advent Health in Orlando, said in an interview. “This study is important as there has been a steady increase in SMM in recent years. In the United States, the CDC reports that SMM affected more than 50,000 women in 2014 alone. Across multiple countries the decline in overall health of women giving birth is felt to contribute to SMM. As the rates of preexisting conditions such as maternal obesity, hypertension, diabetes, and advanced age increases, we can assume that SMM will increase as well. This study clearly depicts the association between SMM and maternal death at 43 days to years after the complicated delivery. We can assume that, as SMM increases, so will the risk of mortality beyond the postpartum period for these women who initially survive their serious pregnancy complication.”
Dr. Humphrey said that, in some respects, the study results are to be expected. “It is logical to assume that a patient with a life-threatening issue at delivery such as severe cardiac complications, acute renal failure and cerebrovascular accident would be at higher risk for long-term morbidity and mortality. This study also adds to the large body of evidence linking socioeconomic deprivation with SMM. But there were unexpected findings in this study. I did not expect certain types of SMM to be associated with an increased risk of death years after the event.For example, hysterectomy at delivery carried a hazard ratio of 3.54 for death at 5-9 years after the event. The association between severe hemorrhage and fully adjusted hazard ratio was similarly increased at 2.96 [2.37-3.71].”
More screening and prospective studies needed
“Recognizing the association between SMM and accelerated long-term risk of mortality is a first step in determining what interventions might improve health and longevity in women who experience SMM,” said Dr. Humphrey. “With the absence of prospective studies, it still is logical to assume that close medical follow-up and lifestyle interventions are appropriate in this population. Screening for and actively managing chronic conditions such as diabetes, dyslipidemia, and hypertension seems appropriate for these patients.”
As for further research, “I am interested to know through prospective clinical trials if specific health maintenance screens and interventions would have a positive impact on the life expectancy of survivors of SMM,” said Dr. Humphrey. “I applaud this team for providing data up to 27 years after an obstetric complication, and I am interested to see if Dr. Ukah and her team will continue their research to determine if there is a ‘second peak’ in mortality in the survivors of SMM when they are elderly. Finally, I would be interested to see more detailed data from this team on the associations between socioeconomic deprivation and short- and long-term mortality for women in their study. This information may help further fuel the movement toward social changes to maximize the health of the women and families we serve.”
The study was supported by the Heart & Stroke Foundation of Canada and awards to the lead author and others from the Fonds de recherche du Québec-Santé. The researchers and Dr. Humphrey had no financial conflicts to disclose.
Women who experienced severe complications during pregnancy were more than twice as likely to die at any time after their last pregnancy, including post partum and beyond, based on data from more than 1 million women.
“Current data suggest that up to 88% of maternal deaths are preceded by severe maternal morbidity,” but the long-term risk of mortality and the effect of severe maternal morbidity has not been well studied, wrote U. Vivian Ukah, MPH, PhD, of McGill University, Montreal, and colleagues.
In a longitudinal cohort study published in Obstetrics & Gynecology, the researchers identified 1,229,306 pregnant women who delivered in Quebec between 1989 and 2016.
The primary outcome of in-hospital mortality after the last pregnancy, either post partum (within 42 days of delivery) or long term (43 days to 29 years after delivery).
Overall, 2.9% of the study population experienced severe maternal morbidity, with an associated mortality rate of 0.86 per 1,000 person-years versus 0.41 per 1,000 person-years in women without severe maternal morbidity. The median time to death for women with severe maternal mortality was 6.8 years, compared with 151 years for those without severe maternal morbidity.
The death rate at any time after delivery, post partum and beyond, was twice as high among women with severe maternal morbidity. The morbidities most often associated with long-term mortality after 42 days were severe cardiac complications (hazard ratio, 7.00), acute renal failure (HR, 4.35), and cerebrovascular accidents (HR, 4.03).
However, the mortality risk following severe maternal morbidity decreased over time, the researchers noted. Severe maternal morbidity was associated with 6.73 times the mortality risk, compared with no morbidity, during the period from 43 days to 11 months, but this difference dropped to 1.91, 1.77, and 1.18 times the risk, compared with no comorbidity, at 1-4 years, 5-9 years, and 10-29 years, respectively.
The study findings were limited by several factors, including the inability to prove causality, the use of only in-hospital mortality data, and the potential for missed cases that fell outside the Canadian Perinatal Surveillance System definition of severe maternal morbidity, the researchers noted.
However, the results suggest that identifying severe maternal morbidity may help identify women at risk for postpartum and long-term premature mortality. “Women with severe maternal morbidity may benefit from continued surveillance and preventative interventions to reduce the risk of premature mortality,” they concluded.
Increased morbidity rates drive need for research
“In this retrospective longitudinal cohort study of over 1.2 million women delivering in Quebec between 1989 and 2016, Dr. Ukah and her colleagues demonstrated the association between severe maternal morbidity [SMM] and an accelerated risk of mortality beyond the postpartum period, compared with women who do not experience SMM,” Rachel Humphrey, MD, a maternal-fetal medicine specialist at Advent Health in Orlando, said in an interview. “This study is important as there has been a steady increase in SMM in recent years. In the United States, the CDC reports that SMM affected more than 50,000 women in 2014 alone. Across multiple countries the decline in overall health of women giving birth is felt to contribute to SMM. As the rates of preexisting conditions such as maternal obesity, hypertension, diabetes, and advanced age increases, we can assume that SMM will increase as well. This study clearly depicts the association between SMM and maternal death at 43 days to years after the complicated delivery. We can assume that, as SMM increases, so will the risk of mortality beyond the postpartum period for these women who initially survive their serious pregnancy complication.”
Dr. Humphrey said that, in some respects, the study results are to be expected. “It is logical to assume that a patient with a life-threatening issue at delivery such as severe cardiac complications, acute renal failure and cerebrovascular accident would be at higher risk for long-term morbidity and mortality. This study also adds to the large body of evidence linking socioeconomic deprivation with SMM. But there were unexpected findings in this study. I did not expect certain types of SMM to be associated with an increased risk of death years after the event.For example, hysterectomy at delivery carried a hazard ratio of 3.54 for death at 5-9 years after the event. The association between severe hemorrhage and fully adjusted hazard ratio was similarly increased at 2.96 [2.37-3.71].”
More screening and prospective studies needed
“Recognizing the association between SMM and accelerated long-term risk of mortality is a first step in determining what interventions might improve health and longevity in women who experience SMM,” said Dr. Humphrey. “With the absence of prospective studies, it still is logical to assume that close medical follow-up and lifestyle interventions are appropriate in this population. Screening for and actively managing chronic conditions such as diabetes, dyslipidemia, and hypertension seems appropriate for these patients.”
As for further research, “I am interested to know through prospective clinical trials if specific health maintenance screens and interventions would have a positive impact on the life expectancy of survivors of SMM,” said Dr. Humphrey. “I applaud this team for providing data up to 27 years after an obstetric complication, and I am interested to see if Dr. Ukah and her team will continue their research to determine if there is a ‘second peak’ in mortality in the survivors of SMM when they are elderly. Finally, I would be interested to see more detailed data from this team on the associations between socioeconomic deprivation and short- and long-term mortality for women in their study. This information may help further fuel the movement toward social changes to maximize the health of the women and families we serve.”
The study was supported by the Heart & Stroke Foundation of Canada and awards to the lead author and others from the Fonds de recherche du Québec-Santé. The researchers and Dr. Humphrey had no financial conflicts to disclose.
Women who experienced severe complications during pregnancy were more than twice as likely to die at any time after their last pregnancy, including post partum and beyond, based on data from more than 1 million women.
“Current data suggest that up to 88% of maternal deaths are preceded by severe maternal morbidity,” but the long-term risk of mortality and the effect of severe maternal morbidity has not been well studied, wrote U. Vivian Ukah, MPH, PhD, of McGill University, Montreal, and colleagues.
In a longitudinal cohort study published in Obstetrics & Gynecology, the researchers identified 1,229,306 pregnant women who delivered in Quebec between 1989 and 2016.
The primary outcome of in-hospital mortality after the last pregnancy, either post partum (within 42 days of delivery) or long term (43 days to 29 years after delivery).
Overall, 2.9% of the study population experienced severe maternal morbidity, with an associated mortality rate of 0.86 per 1,000 person-years versus 0.41 per 1,000 person-years in women without severe maternal morbidity. The median time to death for women with severe maternal mortality was 6.8 years, compared with 151 years for those without severe maternal morbidity.
The death rate at any time after delivery, post partum and beyond, was twice as high among women with severe maternal morbidity. The morbidities most often associated with long-term mortality after 42 days were severe cardiac complications (hazard ratio, 7.00), acute renal failure (HR, 4.35), and cerebrovascular accidents (HR, 4.03).
However, the mortality risk following severe maternal morbidity decreased over time, the researchers noted. Severe maternal morbidity was associated with 6.73 times the mortality risk, compared with no morbidity, during the period from 43 days to 11 months, but this difference dropped to 1.91, 1.77, and 1.18 times the risk, compared with no comorbidity, at 1-4 years, 5-9 years, and 10-29 years, respectively.
The study findings were limited by several factors, including the inability to prove causality, the use of only in-hospital mortality data, and the potential for missed cases that fell outside the Canadian Perinatal Surveillance System definition of severe maternal morbidity, the researchers noted.
However, the results suggest that identifying severe maternal morbidity may help identify women at risk for postpartum and long-term premature mortality. “Women with severe maternal morbidity may benefit from continued surveillance and preventative interventions to reduce the risk of premature mortality,” they concluded.
Increased morbidity rates drive need for research
“In this retrospective longitudinal cohort study of over 1.2 million women delivering in Quebec between 1989 and 2016, Dr. Ukah and her colleagues demonstrated the association between severe maternal morbidity [SMM] and an accelerated risk of mortality beyond the postpartum period, compared with women who do not experience SMM,” Rachel Humphrey, MD, a maternal-fetal medicine specialist at Advent Health in Orlando, said in an interview. “This study is important as there has been a steady increase in SMM in recent years. In the United States, the CDC reports that SMM affected more than 50,000 women in 2014 alone. Across multiple countries the decline in overall health of women giving birth is felt to contribute to SMM. As the rates of preexisting conditions such as maternal obesity, hypertension, diabetes, and advanced age increases, we can assume that SMM will increase as well. This study clearly depicts the association between SMM and maternal death at 43 days to years after the complicated delivery. We can assume that, as SMM increases, so will the risk of mortality beyond the postpartum period for these women who initially survive their serious pregnancy complication.”
Dr. Humphrey said that, in some respects, the study results are to be expected. “It is logical to assume that a patient with a life-threatening issue at delivery such as severe cardiac complications, acute renal failure and cerebrovascular accident would be at higher risk for long-term morbidity and mortality. This study also adds to the large body of evidence linking socioeconomic deprivation with SMM. But there were unexpected findings in this study. I did not expect certain types of SMM to be associated with an increased risk of death years after the event.For example, hysterectomy at delivery carried a hazard ratio of 3.54 for death at 5-9 years after the event. The association between severe hemorrhage and fully adjusted hazard ratio was similarly increased at 2.96 [2.37-3.71].”
More screening and prospective studies needed
“Recognizing the association between SMM and accelerated long-term risk of mortality is a first step in determining what interventions might improve health and longevity in women who experience SMM,” said Dr. Humphrey. “With the absence of prospective studies, it still is logical to assume that close medical follow-up and lifestyle interventions are appropriate in this population. Screening for and actively managing chronic conditions such as diabetes, dyslipidemia, and hypertension seems appropriate for these patients.”
As for further research, “I am interested to know through prospective clinical trials if specific health maintenance screens and interventions would have a positive impact on the life expectancy of survivors of SMM,” said Dr. Humphrey. “I applaud this team for providing data up to 27 years after an obstetric complication, and I am interested to see if Dr. Ukah and her team will continue their research to determine if there is a ‘second peak’ in mortality in the survivors of SMM when they are elderly. Finally, I would be interested to see more detailed data from this team on the associations between socioeconomic deprivation and short- and long-term mortality for women in their study. This information may help further fuel the movement toward social changes to maximize the health of the women and families we serve.”
The study was supported by the Heart & Stroke Foundation of Canada and awards to the lead author and others from the Fonds de recherche du Québec-Santé. The researchers and Dr. Humphrey had no financial conflicts to disclose.
FROM OBSTETRICS & GYNECOLOGY
Menopause, not aging, may influence brain volume
Postmenopausal women not only have larger brain volume than women who are premenopausal, but they also experience larger reductions in brain volume over time, reported Ananthan Ambikairajah of the Centre for Research on Ageing, Health and Wellbeing, Australian National University, Canberra, and associates. Their report was published in Menopause.
In this large population-based cohort of 5,072 women aged 37-73 years, the goal of the study was to look at links between brain volume and measures of menstruation history, such as menopausal status, age at menopause, age at menarche, and the duration of a woman’s reproductive stage, but to do so within the context of how it relates to dementia prevalence. Citing a study in The Lancet Neurology, the authors noted that the age-standardized prevalence for dementia is 17% higher in women than in men, and they speculated that it may be important to look beyond age for answers.
What about menstrual history and Alzheimer’s disease?
According to the Framingham Study in Neurology, the remaining lifetime risk of Alzheimer’s disease (AD) is nearly double for a 65-year-old woman (12%) compared with a 65-year-old man (6.3%), leading Mr. Ambikairajah and associates to conclude that “menstruation history may also be particularly relevant, given that it is unique to female aging.” They further speculated, citing several related studies, that because AD pathology is initiated decades prior to the onset of clinical signs, menstruation history and its effects on brain health may, in fact, be reflected in brain volume.
Postmenopausal women had 0.82% and 1.33% larger total brain and hippocampal volume, respectively, compared with premenopausal women. Postmenopausal women had a 23% greater decrease in total brain volume but not in hippocampal volume over time, compared with premenopausal women.
As Braak and Braak illustrated in Acta Neuropathologica, chronic inflammation has been linked to brain shrinkage “consistent with the pattern of results in the present study,” Mr. Ambikairajah and colleagues noted, adding that longitudinal neuroimaging/biomarker studies are needed to explore this further.
What made this study unique was its ability to match pre- and postmenopausal women for age, a critically important attribute “given that aging and menopause both progress concurrently, which can make it difficult to determine the individual contribution of each for measures of brain health,” the authors explained.
In an interview, Constance Bohon, MD, an ob.gyn. in private practice and assistant clinical professor, George Washington University, Washington, observed: “The conclusion [in this study] is that an early age of menarche, delayed age of menopause and increased duration of the reproductive stage is negatively associated with brain volume.”
What of the neuroprotective effects of endogenous estrogen?
“Their findings are not consistent with a neuroprotective effect of endogenous estrogen exposure on brain volume,” she noted, adding that the study “did not assess the effect of exogenous estrogen on brain volume. Neither was the effect of exogenous or endogenous estrogen on cerebral blood flow assessed. In a study published in Obstetrics & Gynecology, the conclusion was that oophorectomy before the age of natural menopause is associated with a decrease in cognitive impairment and dementia. There was no assessment of brain volume or cerebral blood flow. Likewise in a report published in Neurobiology of Aging, Maki P and Resnick S M. concluded that estrogen helps maintain hippocampal and prefrontal function as women age,” observed Dr. Bohon, noting that the study did not assess brain volume.
“It is unclear whether the most predictive assessment for worsening cognition and dementia is the finding of decreased total brain volume, decreased hippocampal volume, or decreased cerebral blood flow. The effect of both endogenous and exogenous estrogen on the risk for dementia needs further evaluation,” she cautioned.
Mr. Ambikairajah cited one financial disclosure; the remaining contributors had no relevant disclosures.
Postmenopausal women not only have larger brain volume than women who are premenopausal, but they also experience larger reductions in brain volume over time, reported Ananthan Ambikairajah of the Centre for Research on Ageing, Health and Wellbeing, Australian National University, Canberra, and associates. Their report was published in Menopause.
In this large population-based cohort of 5,072 women aged 37-73 years, the goal of the study was to look at links between brain volume and measures of menstruation history, such as menopausal status, age at menopause, age at menarche, and the duration of a woman’s reproductive stage, but to do so within the context of how it relates to dementia prevalence. Citing a study in The Lancet Neurology, the authors noted that the age-standardized prevalence for dementia is 17% higher in women than in men, and they speculated that it may be important to look beyond age for answers.
What about menstrual history and Alzheimer’s disease?
According to the Framingham Study in Neurology, the remaining lifetime risk of Alzheimer’s disease (AD) is nearly double for a 65-year-old woman (12%) compared with a 65-year-old man (6.3%), leading Mr. Ambikairajah and associates to conclude that “menstruation history may also be particularly relevant, given that it is unique to female aging.” They further speculated, citing several related studies, that because AD pathology is initiated decades prior to the onset of clinical signs, menstruation history and its effects on brain health may, in fact, be reflected in brain volume.
Postmenopausal women had 0.82% and 1.33% larger total brain and hippocampal volume, respectively, compared with premenopausal women. Postmenopausal women had a 23% greater decrease in total brain volume but not in hippocampal volume over time, compared with premenopausal women.
As Braak and Braak illustrated in Acta Neuropathologica, chronic inflammation has been linked to brain shrinkage “consistent with the pattern of results in the present study,” Mr. Ambikairajah and colleagues noted, adding that longitudinal neuroimaging/biomarker studies are needed to explore this further.
What made this study unique was its ability to match pre- and postmenopausal women for age, a critically important attribute “given that aging and menopause both progress concurrently, which can make it difficult to determine the individual contribution of each for measures of brain health,” the authors explained.
In an interview, Constance Bohon, MD, an ob.gyn. in private practice and assistant clinical professor, George Washington University, Washington, observed: “The conclusion [in this study] is that an early age of menarche, delayed age of menopause and increased duration of the reproductive stage is negatively associated with brain volume.”
What of the neuroprotective effects of endogenous estrogen?
“Their findings are not consistent with a neuroprotective effect of endogenous estrogen exposure on brain volume,” she noted, adding that the study “did not assess the effect of exogenous estrogen on brain volume. Neither was the effect of exogenous or endogenous estrogen on cerebral blood flow assessed. In a study published in Obstetrics & Gynecology, the conclusion was that oophorectomy before the age of natural menopause is associated with a decrease in cognitive impairment and dementia. There was no assessment of brain volume or cerebral blood flow. Likewise in a report published in Neurobiology of Aging, Maki P and Resnick S M. concluded that estrogen helps maintain hippocampal and prefrontal function as women age,” observed Dr. Bohon, noting that the study did not assess brain volume.
“It is unclear whether the most predictive assessment for worsening cognition and dementia is the finding of decreased total brain volume, decreased hippocampal volume, or decreased cerebral blood flow. The effect of both endogenous and exogenous estrogen on the risk for dementia needs further evaluation,” she cautioned.
Mr. Ambikairajah cited one financial disclosure; the remaining contributors had no relevant disclosures.
Postmenopausal women not only have larger brain volume than women who are premenopausal, but they also experience larger reductions in brain volume over time, reported Ananthan Ambikairajah of the Centre for Research on Ageing, Health and Wellbeing, Australian National University, Canberra, and associates. Their report was published in Menopause.
In this large population-based cohort of 5,072 women aged 37-73 years, the goal of the study was to look at links between brain volume and measures of menstruation history, such as menopausal status, age at menopause, age at menarche, and the duration of a woman’s reproductive stage, but to do so within the context of how it relates to dementia prevalence. Citing a study in The Lancet Neurology, the authors noted that the age-standardized prevalence for dementia is 17% higher in women than in men, and they speculated that it may be important to look beyond age for answers.
What about menstrual history and Alzheimer’s disease?
According to the Framingham Study in Neurology, the remaining lifetime risk of Alzheimer’s disease (AD) is nearly double for a 65-year-old woman (12%) compared with a 65-year-old man (6.3%), leading Mr. Ambikairajah and associates to conclude that “menstruation history may also be particularly relevant, given that it is unique to female aging.” They further speculated, citing several related studies, that because AD pathology is initiated decades prior to the onset of clinical signs, menstruation history and its effects on brain health may, in fact, be reflected in brain volume.
Postmenopausal women had 0.82% and 1.33% larger total brain and hippocampal volume, respectively, compared with premenopausal women. Postmenopausal women had a 23% greater decrease in total brain volume but not in hippocampal volume over time, compared with premenopausal women.
As Braak and Braak illustrated in Acta Neuropathologica, chronic inflammation has been linked to brain shrinkage “consistent with the pattern of results in the present study,” Mr. Ambikairajah and colleagues noted, adding that longitudinal neuroimaging/biomarker studies are needed to explore this further.
What made this study unique was its ability to match pre- and postmenopausal women for age, a critically important attribute “given that aging and menopause both progress concurrently, which can make it difficult to determine the individual contribution of each for measures of brain health,” the authors explained.
In an interview, Constance Bohon, MD, an ob.gyn. in private practice and assistant clinical professor, George Washington University, Washington, observed: “The conclusion [in this study] is that an early age of menarche, delayed age of menopause and increased duration of the reproductive stage is negatively associated with brain volume.”
What of the neuroprotective effects of endogenous estrogen?
“Their findings are not consistent with a neuroprotective effect of endogenous estrogen exposure on brain volume,” she noted, adding that the study “did not assess the effect of exogenous estrogen on brain volume. Neither was the effect of exogenous or endogenous estrogen on cerebral blood flow assessed. In a study published in Obstetrics & Gynecology, the conclusion was that oophorectomy before the age of natural menopause is associated with a decrease in cognitive impairment and dementia. There was no assessment of brain volume or cerebral blood flow. Likewise in a report published in Neurobiology of Aging, Maki P and Resnick S M. concluded that estrogen helps maintain hippocampal and prefrontal function as women age,” observed Dr. Bohon, noting that the study did not assess brain volume.
“It is unclear whether the most predictive assessment for worsening cognition and dementia is the finding of decreased total brain volume, decreased hippocampal volume, or decreased cerebral blood flow. The effect of both endogenous and exogenous estrogen on the risk for dementia needs further evaluation,” she cautioned.
Mr. Ambikairajah cited one financial disclosure; the remaining contributors had no relevant disclosures.
FROM MENOPAUSE
Theory of Planned Behavior Provides A Theoretical Explanation For Enhanced Behavior Change With Genetic-Based Lifestyle Interventions
Study Overview
Objective. To determine the impact of providing genetically tailored and population-based lifestyle advice for weight management on key constructs of the Theory of Planned Behavior (TPB), a widely accepted theory used to help predict human lifestyle-related behaviors.
Design. Pragmatic, cluster, randomized controlled trial.
Settings and participants. This study took place at the East Elgin Family Health Team, a primary care clinic in Aylmer, Ontario, Canada. Recruitment occurred between April 2017 and September 2018, with staggered intervention cohorts occurring from May 2017 to September 2019. Participants enrolled in a weight management program at the clinic were invited to participate in the study if they met the following inclusion criteria: body mass index (BMI) ≥25 kg/m2, >18 years of age, English-speaking, willing to undergo genetic testing, having access to a computer with internet at least 1 day per week, and not seeing another health care provider for weight loss advice outside of the study. Exclusion criteria included pregnancy and lactation. All participants provided written informed consent.
Interventions. At baseline, weight management program cohorts (average cohort size was 14 participants) were randomized (1:1) to receive either the standard population-based intervention (Group Lifestyle Balance, or GLB) or a modified GLB intervention, which included the provision of lifestyle genomics (LGx) information and advice (GLB+LGx). Both interventions aimed to assist participants with weight management and healthy lifestyle change, with particular focus on nutrition and physical activity (PA). Interventions were 12 months long, consisting of 23 group-based sessions and 3 one-on-one sessions with a registered dietitian after 3, 6, and 12 months (all sessions were face-to-face). To improve intervention adherence, participants were given reminder calls for their one-on-one appointments and for the start of their program. A sample size was calculated based on the primary outcome indicating that a total of 74 participants were needed (n = 37 per group) for this trial. By September 2019, this sample size was exceeded with 10 randomized groups (n = 140).
The 5 randomized standard GLB groups followed the established GLB program curriculum comprising population-based information and advice while focusing on following a calorie-controlled, moderate-fat (25% of calories) nutrition plan with at least 150 minutes of weekly moderate-intensity PA. Participants were also provided with a 1-page summary report of their nutrition and PA guidelines at the first group meeting outlining population-based targets, including acceptable macronutrient distribution ranges for protein, total fat, saturated fat, monounsaturated fat, polyunsaturated fat, sodium, calories, snacking, and PA.
The 5 randomized modified GLB+LGx groups followed a modified GLB program curriculum in which participants were given genetic-based information and advice, which differed from the advice given to the standard GLB group, while focusing on following a calorie-controlled nutrition plan. The nutrition and PA targets were personalized based on their individual genetic variation. For example, participants with the AA variant of FTO (rs9939609) were advised to engage in at least 30 to 60 minutes of PA daily 6 days per week, with muscle-strengthening activities at least 2 days per week, rather than receiving the standard population-based advice to aim for 150 minutes weekly of PA with at least 2 days per week of muscle-strengthening activity. Participants were also provided with a 1-page summary report of their nutrition and PA guidelines at the first group meeting, which outlined genetic-based information and advice related to protein, total fat, saturated fat, monounsaturated fat, polyunsaturated fat, sodium, calories, snacking, and PA.
Measures and analysis. Change in the TPB components (attitudes, subjective norms and perceived behavioral control) were measured via a TPB questionnaire at 5 time points: baseline (2-week run-in period), immediately after the first group session (where participants received a summary report of either population-based or genetic-based recommendations depending on group assignment), and after 3-, 6- and 12-month follow-ups. Attitudes, subjective norms, and perceived behavioral control were measured on a Likert scale from 1 through 7. Self-reported measures of actual behavioral control (including annual household income, perceptions about events arising in one’s day-to-day life that suddenly take up one’s free time, perceptions about the frequency of feeling ill or tired, and highest achieved level of education) were collected via survey questions and assessed on a Likert scale of 1 through 7. Stage of change was also measured, based on the Transtheoretical Model, using a Likert scale of 1 through 6.
Linear mixed models were used to conduct within- and between-group analyses using SPSS version 26.0, while controlling for measures of actual behavioral control. All analyses were intention-to-treat by originally assigned groups, with mean value imputation conducted for missing data. A Bonferroni correction for multiple testing was used. For all statistical analyses, the level of significance was set at P < 0.05 and trending towards significance at P = 0.05–0.06.
Main results. Participants consisted of primarily middle-age, middle-income, Caucasian females. Baseline attitudes towards the effectiveness of nutrition and PA for weight management were generally positive, and participants perceived that undergoing genetic testing would assist with weight management. Participants had overall neutral subjective norms related to friends and family consuming a healthy diet and engaging in PA, but perceived that their friends, family, and health care team (HCT) believed it was important for them to achieve their nutrition and PA recommendations. Participants overall also perceived that their HCT believed genetic testing could assist with weight management. Baseline measures of perceived behavioral control were overall neutral, with baseline stage of change between “motivation” and “action” (short-term; <3 months).
In within-group analyses, significant improvements (P < 0.05) in attitudes towards the effectiveness of nutrition and PA recommendations for weight management, subjective norms related to both friends and family consuming a healthy diet, and perceived behavioral control in changing PA/dietary intake and managing weight tended to be short-term in the GLB group and long-term for the GLB+LGx group. In all cases of between-group differences for changes in TPB components, the GLB group exhibited reductions in scores, whereas the GLB+LGx group exhibited increases or improvements. Between-group differences (short-term and long-term) in several measures of subjective norms were observed. For example, after 3 months, significant between-group differences were observed in changes in perception that friends believed LGx would help with weight management (P = 0.024). After 12 months, between-group differences trending towards significance were also observed in changes in perception that family members believed genetic testing would help with weight management (P = 0.05). Significant between-group differences and differences trending towards significance were also observed at 12 months for changes in perception that family believed it was important for the participant to achieve the PA recommendations (P = 0.049) and nutrition recommendations (P = 0.05). Between-group differences trending towards significance were also observed at 3 months in attitudes towards the effectiveness of LGx for weight management (P = 0.06). There were no significant between-group differences observed in changes in perceived behavioral control.
Conclusion. Results from this study support the hypothesis that the TPB can help provide a theoretical explanation for why genetically tailored lifestyle information and advice can lead to improvements in lifestyle behavior change.
Commentary
Because health behaviors are critical in areas such as prevention, treatment, and rehabilitation, it is important to describe and understand what drives these behaviors.1 Theories are important tools in this effort as they aim to explain and predict health behavior and are used in the design and evaluation of interventions.1 The TPB is one of the most widely accepted behavior change theories and posits that attitudes, subjective norms (or social pressures and behaviors), and perceived behavioral control are significant predictors of an individual’s intention to engage in behaviors.2 TPB has been highlighted in the literature as a validated theory for predicting nutrition and PA intentions and resulting behaviors.3,4
Motivating lifestyle behavior change in clinical practice can be challenging, but some studies have demonstrated how providing genetic information and advice (or lifestyle genomics) can help motivate changes in nutrition and PA among patients.5-7 Because this has yet to be explained using the TPB, this study is an important contribution to the literature as it aimed to determine the impact of providing genetically tailored and population-based lifestyle advice for weight management on key constructs of the TPB. Briefly, results from within-group analyses in this study demonstrated that the provision of genetically tailored lifestyle information and advice (via the GLB+LGx intervention) tended to impact antecedents of behavior change, more so over the long-term, while population-based advice (via the standard GLB intervention) tended to impact antecedents of behavior change over the short-term (eg, attitudes towards dietary fat intake, perceptions that friends and family consume a healthy diet, and perceptions about the impact of genetic-based advice for weight management). In addition, between-group differences in subjective norms observed at 12 months suggested that social pressures and norms may be influencing long-term changes in lifestyle habits.
While key strengths of this study include its pragmatic cluster randomized controlled trial design, 12-month intervention duration, and intent-to-treat analyses, there are some study limitations, which are acknowledged by the authors. Generalizability is limited to the demographic characteristics of the study population (ie, middle-aged, middle-income, Caucasian females enrolled in a lifestyle change weight management program). Thus, replication of the study is needed in more diverse study populations and with health-related outcomes beyond weight management. In addition, as the authors indicate, future research should ensure the inclusion of theory-based questionnaires in genetic-based intervention studies assessing lifestyle behavior change to elucidate theory-based mechanisms of change.
Applications for Clinical Practice
Population-based research has consistently indicated that nutrition interventions typically impact short-term dietary changes. Confronting the challenge of long-term adherence to nutrition and PA recommendations requires an understanding of factors impacting long-term motivation and behavior change. With increased attention on and research into genetically tailored lifestyle advice (or lifestyle genomics), it is important for clinical practitioners to be familiar with the evidence supporting these approaches. In addition, this research highlights the need to consider individual factors (attitudes, subjective norms, and perceived behavioral control) that may predict successful change in lifestyle habits when providing nutrition and PA recommendations, whether population-based or genetically tailored.
—Katrina F. Mateo, PhD, MPH
1. Lippke S, Ziegelmann JP. Theory-based health behavior change: Developing, testing, and applying theories for evidence-based interventions. Appl Psychol. 2008;57:698-716.
2. Ajzen I. The Theory of planned behaviour: reactions and reflections. Psychol Health. 2011;26:1113-1127.
3. McDermott MS, Oliver M, Simnadis T, et al. The Theory of Planned Behaviour and dietary patterns: A systematic review and meta-analysis. Prev Med (Baltim). 2015;81:150-156.
4. McEachan RRC, Conner M, Taylor NJ, Lawton RJ. Prospective prediction of health-related behaviours with the theory of planned behaviour: A meta-analysis. Health Psychol Rev. 2011;5:97-144.
5. Hietaranta-Luoma H-L, Tahvonen R, Iso-Touru T, et al A. An intervention study of individual, APOE genotype-based dietary and physical-activity advice: impact on health behavior. J Nutrigenet Nutrigenomics. 2014;7:161-174.
6. Nielsen DE, El-Sohemy A. Disclosure of genetic information and change in dietary intake: a randomized controlled trial. DeAngelis MM, ed. PLoS One. 2014;9(11):e112665.
7. Egglestone C, Morris A, O’Brien A. Effect of direct‐to‐consumer genetic tests on health behaviour and anxiety: a survey of consumers and potential consumers. J Genet Couns. 2013;22:565-575.
Study Overview
Objective. To determine the impact of providing genetically tailored and population-based lifestyle advice for weight management on key constructs of the Theory of Planned Behavior (TPB), a widely accepted theory used to help predict human lifestyle-related behaviors.
Design. Pragmatic, cluster, randomized controlled trial.
Settings and participants. This study took place at the East Elgin Family Health Team, a primary care clinic in Aylmer, Ontario, Canada. Recruitment occurred between April 2017 and September 2018, with staggered intervention cohorts occurring from May 2017 to September 2019. Participants enrolled in a weight management program at the clinic were invited to participate in the study if they met the following inclusion criteria: body mass index (BMI) ≥25 kg/m2, >18 years of age, English-speaking, willing to undergo genetic testing, having access to a computer with internet at least 1 day per week, and not seeing another health care provider for weight loss advice outside of the study. Exclusion criteria included pregnancy and lactation. All participants provided written informed consent.
Interventions. At baseline, weight management program cohorts (average cohort size was 14 participants) were randomized (1:1) to receive either the standard population-based intervention (Group Lifestyle Balance, or GLB) or a modified GLB intervention, which included the provision of lifestyle genomics (LGx) information and advice (GLB+LGx). Both interventions aimed to assist participants with weight management and healthy lifestyle change, with particular focus on nutrition and physical activity (PA). Interventions were 12 months long, consisting of 23 group-based sessions and 3 one-on-one sessions with a registered dietitian after 3, 6, and 12 months (all sessions were face-to-face). To improve intervention adherence, participants were given reminder calls for their one-on-one appointments and for the start of their program. A sample size was calculated based on the primary outcome indicating that a total of 74 participants were needed (n = 37 per group) for this trial. By September 2019, this sample size was exceeded with 10 randomized groups (n = 140).
The 5 randomized standard GLB groups followed the established GLB program curriculum comprising population-based information and advice while focusing on following a calorie-controlled, moderate-fat (25% of calories) nutrition plan with at least 150 minutes of weekly moderate-intensity PA. Participants were also provided with a 1-page summary report of their nutrition and PA guidelines at the first group meeting outlining population-based targets, including acceptable macronutrient distribution ranges for protein, total fat, saturated fat, monounsaturated fat, polyunsaturated fat, sodium, calories, snacking, and PA.
The 5 randomized modified GLB+LGx groups followed a modified GLB program curriculum in which participants were given genetic-based information and advice, which differed from the advice given to the standard GLB group, while focusing on following a calorie-controlled nutrition plan. The nutrition and PA targets were personalized based on their individual genetic variation. For example, participants with the AA variant of FTO (rs9939609) were advised to engage in at least 30 to 60 minutes of PA daily 6 days per week, with muscle-strengthening activities at least 2 days per week, rather than receiving the standard population-based advice to aim for 150 minutes weekly of PA with at least 2 days per week of muscle-strengthening activity. Participants were also provided with a 1-page summary report of their nutrition and PA guidelines at the first group meeting, which outlined genetic-based information and advice related to protein, total fat, saturated fat, monounsaturated fat, polyunsaturated fat, sodium, calories, snacking, and PA.
Measures and analysis. Change in the TPB components (attitudes, subjective norms and perceived behavioral control) were measured via a TPB questionnaire at 5 time points: baseline (2-week run-in period), immediately after the first group session (where participants received a summary report of either population-based or genetic-based recommendations depending on group assignment), and after 3-, 6- and 12-month follow-ups. Attitudes, subjective norms, and perceived behavioral control were measured on a Likert scale from 1 through 7. Self-reported measures of actual behavioral control (including annual household income, perceptions about events arising in one’s day-to-day life that suddenly take up one’s free time, perceptions about the frequency of feeling ill or tired, and highest achieved level of education) were collected via survey questions and assessed on a Likert scale of 1 through 7. Stage of change was also measured, based on the Transtheoretical Model, using a Likert scale of 1 through 6.
Linear mixed models were used to conduct within- and between-group analyses using SPSS version 26.0, while controlling for measures of actual behavioral control. All analyses were intention-to-treat by originally assigned groups, with mean value imputation conducted for missing data. A Bonferroni correction for multiple testing was used. For all statistical analyses, the level of significance was set at P < 0.05 and trending towards significance at P = 0.05–0.06.
Main results. Participants consisted of primarily middle-age, middle-income, Caucasian females. Baseline attitudes towards the effectiveness of nutrition and PA for weight management were generally positive, and participants perceived that undergoing genetic testing would assist with weight management. Participants had overall neutral subjective norms related to friends and family consuming a healthy diet and engaging in PA, but perceived that their friends, family, and health care team (HCT) believed it was important for them to achieve their nutrition and PA recommendations. Participants overall also perceived that their HCT believed genetic testing could assist with weight management. Baseline measures of perceived behavioral control were overall neutral, with baseline stage of change between “motivation” and “action” (short-term; <3 months).
In within-group analyses, significant improvements (P < 0.05) in attitudes towards the effectiveness of nutrition and PA recommendations for weight management, subjective norms related to both friends and family consuming a healthy diet, and perceived behavioral control in changing PA/dietary intake and managing weight tended to be short-term in the GLB group and long-term for the GLB+LGx group. In all cases of between-group differences for changes in TPB components, the GLB group exhibited reductions in scores, whereas the GLB+LGx group exhibited increases or improvements. Between-group differences (short-term and long-term) in several measures of subjective norms were observed. For example, after 3 months, significant between-group differences were observed in changes in perception that friends believed LGx would help with weight management (P = 0.024). After 12 months, between-group differences trending towards significance were also observed in changes in perception that family members believed genetic testing would help with weight management (P = 0.05). Significant between-group differences and differences trending towards significance were also observed at 12 months for changes in perception that family believed it was important for the participant to achieve the PA recommendations (P = 0.049) and nutrition recommendations (P = 0.05). Between-group differences trending towards significance were also observed at 3 months in attitudes towards the effectiveness of LGx for weight management (P = 0.06). There were no significant between-group differences observed in changes in perceived behavioral control.
Conclusion. Results from this study support the hypothesis that the TPB can help provide a theoretical explanation for why genetically tailored lifestyle information and advice can lead to improvements in lifestyle behavior change.
Commentary
Because health behaviors are critical in areas such as prevention, treatment, and rehabilitation, it is important to describe and understand what drives these behaviors.1 Theories are important tools in this effort as they aim to explain and predict health behavior and are used in the design and evaluation of interventions.1 The TPB is one of the most widely accepted behavior change theories and posits that attitudes, subjective norms (or social pressures and behaviors), and perceived behavioral control are significant predictors of an individual’s intention to engage in behaviors.2 TPB has been highlighted in the literature as a validated theory for predicting nutrition and PA intentions and resulting behaviors.3,4
Motivating lifestyle behavior change in clinical practice can be challenging, but some studies have demonstrated how providing genetic information and advice (or lifestyle genomics) can help motivate changes in nutrition and PA among patients.5-7 Because this has yet to be explained using the TPB, this study is an important contribution to the literature as it aimed to determine the impact of providing genetically tailored and population-based lifestyle advice for weight management on key constructs of the TPB. Briefly, results from within-group analyses in this study demonstrated that the provision of genetically tailored lifestyle information and advice (via the GLB+LGx intervention) tended to impact antecedents of behavior change, more so over the long-term, while population-based advice (via the standard GLB intervention) tended to impact antecedents of behavior change over the short-term (eg, attitudes towards dietary fat intake, perceptions that friends and family consume a healthy diet, and perceptions about the impact of genetic-based advice for weight management). In addition, between-group differences in subjective norms observed at 12 months suggested that social pressures and norms may be influencing long-term changes in lifestyle habits.
While key strengths of this study include its pragmatic cluster randomized controlled trial design, 12-month intervention duration, and intent-to-treat analyses, there are some study limitations, which are acknowledged by the authors. Generalizability is limited to the demographic characteristics of the study population (ie, middle-aged, middle-income, Caucasian females enrolled in a lifestyle change weight management program). Thus, replication of the study is needed in more diverse study populations and with health-related outcomes beyond weight management. In addition, as the authors indicate, future research should ensure the inclusion of theory-based questionnaires in genetic-based intervention studies assessing lifestyle behavior change to elucidate theory-based mechanisms of change.
Applications for Clinical Practice
Population-based research has consistently indicated that nutrition interventions typically impact short-term dietary changes. Confronting the challenge of long-term adherence to nutrition and PA recommendations requires an understanding of factors impacting long-term motivation and behavior change. With increased attention on and research into genetically tailored lifestyle advice (or lifestyle genomics), it is important for clinical practitioners to be familiar with the evidence supporting these approaches. In addition, this research highlights the need to consider individual factors (attitudes, subjective norms, and perceived behavioral control) that may predict successful change in lifestyle habits when providing nutrition and PA recommendations, whether population-based or genetically tailored.
—Katrina F. Mateo, PhD, MPH
Study Overview
Objective. To determine the impact of providing genetically tailored and population-based lifestyle advice for weight management on key constructs of the Theory of Planned Behavior (TPB), a widely accepted theory used to help predict human lifestyle-related behaviors.
Design. Pragmatic, cluster, randomized controlled trial.
Settings and participants. This study took place at the East Elgin Family Health Team, a primary care clinic in Aylmer, Ontario, Canada. Recruitment occurred between April 2017 and September 2018, with staggered intervention cohorts occurring from May 2017 to September 2019. Participants enrolled in a weight management program at the clinic were invited to participate in the study if they met the following inclusion criteria: body mass index (BMI) ≥25 kg/m2, >18 years of age, English-speaking, willing to undergo genetic testing, having access to a computer with internet at least 1 day per week, and not seeing another health care provider for weight loss advice outside of the study. Exclusion criteria included pregnancy and lactation. All participants provided written informed consent.
Interventions. At baseline, weight management program cohorts (average cohort size was 14 participants) were randomized (1:1) to receive either the standard population-based intervention (Group Lifestyle Balance, or GLB) or a modified GLB intervention, which included the provision of lifestyle genomics (LGx) information and advice (GLB+LGx). Both interventions aimed to assist participants with weight management and healthy lifestyle change, with particular focus on nutrition and physical activity (PA). Interventions were 12 months long, consisting of 23 group-based sessions and 3 one-on-one sessions with a registered dietitian after 3, 6, and 12 months (all sessions were face-to-face). To improve intervention adherence, participants were given reminder calls for their one-on-one appointments and for the start of their program. A sample size was calculated based on the primary outcome indicating that a total of 74 participants were needed (n = 37 per group) for this trial. By September 2019, this sample size was exceeded with 10 randomized groups (n = 140).
The 5 randomized standard GLB groups followed the established GLB program curriculum comprising population-based information and advice while focusing on following a calorie-controlled, moderate-fat (25% of calories) nutrition plan with at least 150 minutes of weekly moderate-intensity PA. Participants were also provided with a 1-page summary report of their nutrition and PA guidelines at the first group meeting outlining population-based targets, including acceptable macronutrient distribution ranges for protein, total fat, saturated fat, monounsaturated fat, polyunsaturated fat, sodium, calories, snacking, and PA.
The 5 randomized modified GLB+LGx groups followed a modified GLB program curriculum in which participants were given genetic-based information and advice, which differed from the advice given to the standard GLB group, while focusing on following a calorie-controlled nutrition plan. The nutrition and PA targets were personalized based on their individual genetic variation. For example, participants with the AA variant of FTO (rs9939609) were advised to engage in at least 30 to 60 minutes of PA daily 6 days per week, with muscle-strengthening activities at least 2 days per week, rather than receiving the standard population-based advice to aim for 150 minutes weekly of PA with at least 2 days per week of muscle-strengthening activity. Participants were also provided with a 1-page summary report of their nutrition and PA guidelines at the first group meeting, which outlined genetic-based information and advice related to protein, total fat, saturated fat, monounsaturated fat, polyunsaturated fat, sodium, calories, snacking, and PA.
Measures and analysis. Change in the TPB components (attitudes, subjective norms and perceived behavioral control) were measured via a TPB questionnaire at 5 time points: baseline (2-week run-in period), immediately after the first group session (where participants received a summary report of either population-based or genetic-based recommendations depending on group assignment), and after 3-, 6- and 12-month follow-ups. Attitudes, subjective norms, and perceived behavioral control were measured on a Likert scale from 1 through 7. Self-reported measures of actual behavioral control (including annual household income, perceptions about events arising in one’s day-to-day life that suddenly take up one’s free time, perceptions about the frequency of feeling ill or tired, and highest achieved level of education) were collected via survey questions and assessed on a Likert scale of 1 through 7. Stage of change was also measured, based on the Transtheoretical Model, using a Likert scale of 1 through 6.
Linear mixed models were used to conduct within- and between-group analyses using SPSS version 26.0, while controlling for measures of actual behavioral control. All analyses were intention-to-treat by originally assigned groups, with mean value imputation conducted for missing data. A Bonferroni correction for multiple testing was used. For all statistical analyses, the level of significance was set at P < 0.05 and trending towards significance at P = 0.05–0.06.
Main results. Participants consisted of primarily middle-age, middle-income, Caucasian females. Baseline attitudes towards the effectiveness of nutrition and PA for weight management were generally positive, and participants perceived that undergoing genetic testing would assist with weight management. Participants had overall neutral subjective norms related to friends and family consuming a healthy diet and engaging in PA, but perceived that their friends, family, and health care team (HCT) believed it was important for them to achieve their nutrition and PA recommendations. Participants overall also perceived that their HCT believed genetic testing could assist with weight management. Baseline measures of perceived behavioral control were overall neutral, with baseline stage of change between “motivation” and “action” (short-term; <3 months).
In within-group analyses, significant improvements (P < 0.05) in attitudes towards the effectiveness of nutrition and PA recommendations for weight management, subjective norms related to both friends and family consuming a healthy diet, and perceived behavioral control in changing PA/dietary intake and managing weight tended to be short-term in the GLB group and long-term for the GLB+LGx group. In all cases of between-group differences for changes in TPB components, the GLB group exhibited reductions in scores, whereas the GLB+LGx group exhibited increases or improvements. Between-group differences (short-term and long-term) in several measures of subjective norms were observed. For example, after 3 months, significant between-group differences were observed in changes in perception that friends believed LGx would help with weight management (P = 0.024). After 12 months, between-group differences trending towards significance were also observed in changes in perception that family members believed genetic testing would help with weight management (P = 0.05). Significant between-group differences and differences trending towards significance were also observed at 12 months for changes in perception that family believed it was important for the participant to achieve the PA recommendations (P = 0.049) and nutrition recommendations (P = 0.05). Between-group differences trending towards significance were also observed at 3 months in attitudes towards the effectiveness of LGx for weight management (P = 0.06). There were no significant between-group differences observed in changes in perceived behavioral control.
Conclusion. Results from this study support the hypothesis that the TPB can help provide a theoretical explanation for why genetically tailored lifestyle information and advice can lead to improvements in lifestyle behavior change.
Commentary
Because health behaviors are critical in areas such as prevention, treatment, and rehabilitation, it is important to describe and understand what drives these behaviors.1 Theories are important tools in this effort as they aim to explain and predict health behavior and are used in the design and evaluation of interventions.1 The TPB is one of the most widely accepted behavior change theories and posits that attitudes, subjective norms (or social pressures and behaviors), and perceived behavioral control are significant predictors of an individual’s intention to engage in behaviors.2 TPB has been highlighted in the literature as a validated theory for predicting nutrition and PA intentions and resulting behaviors.3,4
Motivating lifestyle behavior change in clinical practice can be challenging, but some studies have demonstrated how providing genetic information and advice (or lifestyle genomics) can help motivate changes in nutrition and PA among patients.5-7 Because this has yet to be explained using the TPB, this study is an important contribution to the literature as it aimed to determine the impact of providing genetically tailored and population-based lifestyle advice for weight management on key constructs of the TPB. Briefly, results from within-group analyses in this study demonstrated that the provision of genetically tailored lifestyle information and advice (via the GLB+LGx intervention) tended to impact antecedents of behavior change, more so over the long-term, while population-based advice (via the standard GLB intervention) tended to impact antecedents of behavior change over the short-term (eg, attitudes towards dietary fat intake, perceptions that friends and family consume a healthy diet, and perceptions about the impact of genetic-based advice for weight management). In addition, between-group differences in subjective norms observed at 12 months suggested that social pressures and norms may be influencing long-term changes in lifestyle habits.
While key strengths of this study include its pragmatic cluster randomized controlled trial design, 12-month intervention duration, and intent-to-treat analyses, there are some study limitations, which are acknowledged by the authors. Generalizability is limited to the demographic characteristics of the study population (ie, middle-aged, middle-income, Caucasian females enrolled in a lifestyle change weight management program). Thus, replication of the study is needed in more diverse study populations and with health-related outcomes beyond weight management. In addition, as the authors indicate, future research should ensure the inclusion of theory-based questionnaires in genetic-based intervention studies assessing lifestyle behavior change to elucidate theory-based mechanisms of change.
Applications for Clinical Practice
Population-based research has consistently indicated that nutrition interventions typically impact short-term dietary changes. Confronting the challenge of long-term adherence to nutrition and PA recommendations requires an understanding of factors impacting long-term motivation and behavior change. With increased attention on and research into genetically tailored lifestyle advice (or lifestyle genomics), it is important for clinical practitioners to be familiar with the evidence supporting these approaches. In addition, this research highlights the need to consider individual factors (attitudes, subjective norms, and perceived behavioral control) that may predict successful change in lifestyle habits when providing nutrition and PA recommendations, whether population-based or genetically tailored.
—Katrina F. Mateo, PhD, MPH
1. Lippke S, Ziegelmann JP. Theory-based health behavior change: Developing, testing, and applying theories for evidence-based interventions. Appl Psychol. 2008;57:698-716.
2. Ajzen I. The Theory of planned behaviour: reactions and reflections. Psychol Health. 2011;26:1113-1127.
3. McDermott MS, Oliver M, Simnadis T, et al. The Theory of Planned Behaviour and dietary patterns: A systematic review and meta-analysis. Prev Med (Baltim). 2015;81:150-156.
4. McEachan RRC, Conner M, Taylor NJ, Lawton RJ. Prospective prediction of health-related behaviours with the theory of planned behaviour: A meta-analysis. Health Psychol Rev. 2011;5:97-144.
5. Hietaranta-Luoma H-L, Tahvonen R, Iso-Touru T, et al A. An intervention study of individual, APOE genotype-based dietary and physical-activity advice: impact on health behavior. J Nutrigenet Nutrigenomics. 2014;7:161-174.
6. Nielsen DE, El-Sohemy A. Disclosure of genetic information and change in dietary intake: a randomized controlled trial. DeAngelis MM, ed. PLoS One. 2014;9(11):e112665.
7. Egglestone C, Morris A, O’Brien A. Effect of direct‐to‐consumer genetic tests on health behaviour and anxiety: a survey of consumers and potential consumers. J Genet Couns. 2013;22:565-575.
1. Lippke S, Ziegelmann JP. Theory-based health behavior change: Developing, testing, and applying theories for evidence-based interventions. Appl Psychol. 2008;57:698-716.
2. Ajzen I. The Theory of planned behaviour: reactions and reflections. Psychol Health. 2011;26:1113-1127.
3. McDermott MS, Oliver M, Simnadis T, et al. The Theory of Planned Behaviour and dietary patterns: A systematic review and meta-analysis. Prev Med (Baltim). 2015;81:150-156.
4. McEachan RRC, Conner M, Taylor NJ, Lawton RJ. Prospective prediction of health-related behaviours with the theory of planned behaviour: A meta-analysis. Health Psychol Rev. 2011;5:97-144.
5. Hietaranta-Luoma H-L, Tahvonen R, Iso-Touru T, et al A. An intervention study of individual, APOE genotype-based dietary and physical-activity advice: impact on health behavior. J Nutrigenet Nutrigenomics. 2014;7:161-174.
6. Nielsen DE, El-Sohemy A. Disclosure of genetic information and change in dietary intake: a randomized controlled trial. DeAngelis MM, ed. PLoS One. 2014;9(11):e112665.
7. Egglestone C, Morris A, O’Brien A. Effect of direct‐to‐consumer genetic tests on health behaviour and anxiety: a survey of consumers and potential consumers. J Genet Couns. 2013;22:565-575.
Dermatologist survey spotlights psoriasis care deficiencies in reproductive-age women
In a , Jenny Murase, MD, reported at the annual congress of the European Academy of Dermatology and Venereology.
“In Germany, the UK, and the United States, dermatologists face challenges in discussing pregnancy and child-bearing aspiration with women of reproductive age, in recommending compatible treatments during pregnancy, and engaging patients in the shared decision-making process. These challenges may exist due to suboptimal knowledge, skills, confidence, and attitude in respective areas of care,” said Dr. Murase, a dermatologist at the University of California, San Francisco, and coeditor-in-chief of the International Journal of Women’s Dermatology.
These shortcomings were documented in a survey, which began with Dr. Murase and her coinvestigators conducting detailed, 45-minute-long, semistructured telephone interviews with 24 dermatologists in the three countries. Those interviews provided the basis for subsequent development of a 20-minute online survey on psoriasis and pregnancy completed by 167 American, German, and UK dermatologists. The survey incorporated multiple choice questions and quantitative rating scales.
“Participants expressed challenges engaging in family planning counseling and reproductive health care as part of risk assessments for psoriasis,” Dr. Murase said.
Among the key findings:
- Forty-seven percent of respondents considered their knowledge of the impact of psoriasis on women’s reproductive health to be suboptimal. This knowledge gap was most common among American dermatologists, 59% of whom rated themselves as having suboptimal knowledge, and least common among German practitioners, only 27% of whom reported deficiencies in this area.
Fifty percent of dermatologists rated themselves as having suboptimal skills in discussing contraceptive methods with their psoriasis patients of childbearing potential.
- Forty-eight percent of respondents – and 59% of the American dermatologists – indicated they prefer to leave pregnancy-related discussions to ob.gyns.
- Fifty-five percent of dermatologists had only limited knowledge of the safety data and indications for prescribing biologic therapies before, during, and after pregnancy. Respondents gave themselves an average score of 58 out of 100 in terms of their confidence in prescribing biologics during pregnancy, compared to 74 out of 100 when prescribing before or after pregnancy.
- Forty-eight percent of participants indicated they had suboptimal skills in helping patients counter obstacles to treatment adherence.
Consideration of treatment of psoriasis in pregnancy requires balancing potential medication risks to the fetus versus the possible maternal and fetal harms of under- or nontreatment of their chronic inflammatory skin disease. It’s a matter that calls for shared decision-making between dermatologist and patient. But the survey showed that shared decision-making was often poorly integrated into clinical practice. Ninety-seven percent of the U.S. dermatologists were unaware of the existence of shared decision-making practice guidelines or models, as were 80% of UK respondents and 85% of the Germans. Of the relatively few dermatologists who were aware of such guidance, nearly half dismissed it as inapplicable to their clinical practice. More than one-third of respondents admitted having suboptimal skills in assessing their patients’ desired level of involvement in medical decisions. And one-third of the German dermatologists and roughly one-quarter of those from the United States and United Kingdom reported feeling pressure to make treatment decisions quickly and without patient input.
Dr. Murase added that the survey findings make a strong case for future interventions designed to help dermatologists appreciate the value of shared decision-making and develop the requisite patient-engagement skills. Dr. Murase reported serving as a paid consultant to UCB Pharma, which funded the survey via an educational grant.
In a , Jenny Murase, MD, reported at the annual congress of the European Academy of Dermatology and Venereology.
“In Germany, the UK, and the United States, dermatologists face challenges in discussing pregnancy and child-bearing aspiration with women of reproductive age, in recommending compatible treatments during pregnancy, and engaging patients in the shared decision-making process. These challenges may exist due to suboptimal knowledge, skills, confidence, and attitude in respective areas of care,” said Dr. Murase, a dermatologist at the University of California, San Francisco, and coeditor-in-chief of the International Journal of Women’s Dermatology.
These shortcomings were documented in a survey, which began with Dr. Murase and her coinvestigators conducting detailed, 45-minute-long, semistructured telephone interviews with 24 dermatologists in the three countries. Those interviews provided the basis for subsequent development of a 20-minute online survey on psoriasis and pregnancy completed by 167 American, German, and UK dermatologists. The survey incorporated multiple choice questions and quantitative rating scales.
“Participants expressed challenges engaging in family planning counseling and reproductive health care as part of risk assessments for psoriasis,” Dr. Murase said.
Among the key findings:
- Forty-seven percent of respondents considered their knowledge of the impact of psoriasis on women’s reproductive health to be suboptimal. This knowledge gap was most common among American dermatologists, 59% of whom rated themselves as having suboptimal knowledge, and least common among German practitioners, only 27% of whom reported deficiencies in this area.
Fifty percent of dermatologists rated themselves as having suboptimal skills in discussing contraceptive methods with their psoriasis patients of childbearing potential.
- Forty-eight percent of respondents – and 59% of the American dermatologists – indicated they prefer to leave pregnancy-related discussions to ob.gyns.
- Fifty-five percent of dermatologists had only limited knowledge of the safety data and indications for prescribing biologic therapies before, during, and after pregnancy. Respondents gave themselves an average score of 58 out of 100 in terms of their confidence in prescribing biologics during pregnancy, compared to 74 out of 100 when prescribing before or after pregnancy.
- Forty-eight percent of participants indicated they had suboptimal skills in helping patients counter obstacles to treatment adherence.
Consideration of treatment of psoriasis in pregnancy requires balancing potential medication risks to the fetus versus the possible maternal and fetal harms of under- or nontreatment of their chronic inflammatory skin disease. It’s a matter that calls for shared decision-making between dermatologist and patient. But the survey showed that shared decision-making was often poorly integrated into clinical practice. Ninety-seven percent of the U.S. dermatologists were unaware of the existence of shared decision-making practice guidelines or models, as were 80% of UK respondents and 85% of the Germans. Of the relatively few dermatologists who were aware of such guidance, nearly half dismissed it as inapplicable to their clinical practice. More than one-third of respondents admitted having suboptimal skills in assessing their patients’ desired level of involvement in medical decisions. And one-third of the German dermatologists and roughly one-quarter of those from the United States and United Kingdom reported feeling pressure to make treatment decisions quickly and without patient input.
Dr. Murase added that the survey findings make a strong case for future interventions designed to help dermatologists appreciate the value of shared decision-making and develop the requisite patient-engagement skills. Dr. Murase reported serving as a paid consultant to UCB Pharma, which funded the survey via an educational grant.
In a , Jenny Murase, MD, reported at the annual congress of the European Academy of Dermatology and Venereology.
“In Germany, the UK, and the United States, dermatologists face challenges in discussing pregnancy and child-bearing aspiration with women of reproductive age, in recommending compatible treatments during pregnancy, and engaging patients in the shared decision-making process. These challenges may exist due to suboptimal knowledge, skills, confidence, and attitude in respective areas of care,” said Dr. Murase, a dermatologist at the University of California, San Francisco, and coeditor-in-chief of the International Journal of Women’s Dermatology.
These shortcomings were documented in a survey, which began with Dr. Murase and her coinvestigators conducting detailed, 45-minute-long, semistructured telephone interviews with 24 dermatologists in the three countries. Those interviews provided the basis for subsequent development of a 20-minute online survey on psoriasis and pregnancy completed by 167 American, German, and UK dermatologists. The survey incorporated multiple choice questions and quantitative rating scales.
“Participants expressed challenges engaging in family planning counseling and reproductive health care as part of risk assessments for psoriasis,” Dr. Murase said.
Among the key findings:
- Forty-seven percent of respondents considered their knowledge of the impact of psoriasis on women’s reproductive health to be suboptimal. This knowledge gap was most common among American dermatologists, 59% of whom rated themselves as having suboptimal knowledge, and least common among German practitioners, only 27% of whom reported deficiencies in this area.
Fifty percent of dermatologists rated themselves as having suboptimal skills in discussing contraceptive methods with their psoriasis patients of childbearing potential.
- Forty-eight percent of respondents – and 59% of the American dermatologists – indicated they prefer to leave pregnancy-related discussions to ob.gyns.
- Fifty-five percent of dermatologists had only limited knowledge of the safety data and indications for prescribing biologic therapies before, during, and after pregnancy. Respondents gave themselves an average score of 58 out of 100 in terms of their confidence in prescribing biologics during pregnancy, compared to 74 out of 100 when prescribing before or after pregnancy.
- Forty-eight percent of participants indicated they had suboptimal skills in helping patients counter obstacles to treatment adherence.
Consideration of treatment of psoriasis in pregnancy requires balancing potential medication risks to the fetus versus the possible maternal and fetal harms of under- or nontreatment of their chronic inflammatory skin disease. It’s a matter that calls for shared decision-making between dermatologist and patient. But the survey showed that shared decision-making was often poorly integrated into clinical practice. Ninety-seven percent of the U.S. dermatologists were unaware of the existence of shared decision-making practice guidelines or models, as were 80% of UK respondents and 85% of the Germans. Of the relatively few dermatologists who were aware of such guidance, nearly half dismissed it as inapplicable to their clinical practice. More than one-third of respondents admitted having suboptimal skills in assessing their patients’ desired level of involvement in medical decisions. And one-third of the German dermatologists and roughly one-quarter of those from the United States and United Kingdom reported feeling pressure to make treatment decisions quickly and without patient input.
Dr. Murase added that the survey findings make a strong case for future interventions designed to help dermatologists appreciate the value of shared decision-making and develop the requisite patient-engagement skills. Dr. Murase reported serving as a paid consultant to UCB Pharma, which funded the survey via an educational grant.
FROM THE EADV CONGRESS
20-year-old man • sudden-onset chest pain • worsening pain with cough and exertion • Dx?
THE CASE
A 20-year-old man presented to our clinic with a 3-day history of nonradiating chest pain located at the center of his chest. Past medical history included idiopathic neonatal giant-cell hepatitis and subsequent liver transplant at 1 month of age; he had been followed by the transplant team without rejection or infection and was in otherwise good health prior to the chest pain.
On the day of symptom onset, he was walking inside his house and fell to his knees with a chest pain described as “a punch” to the center of the chest that lasted for a few seconds. He was able to continue his daily activities without limitation despite a constant, squeezing, centrally located chest pain. The pain worsened with cough and exertion.
A few hours later, he went to an urgent care center for evaluation. There, he reported, his chest radiograph and electrocardiogram (EKG) results were normal and he was given a diagnosis of musculoskeletal chest pain. Over the next 3 days, his chest pain persisted but did not worsen. He was taking 500 mg of naproxen every 8 hours with no improvement. No other acute or chronic medications were being taken. He had no significant family history. A review of systems was otherwise negative.
On physical exam, his vital statistics included a height of 6’4”; weight, 261 lb; body mass index, 31.8; temperature, 98.7 °F; blood pressure, 134/77 mm Hg; heart rate, 92 beats/min; respiratory rate, 18 breaths/min; and oxygen saturation, 96%. Throughout the exam, he demonstrated no acute distress, appeared well, and was talkative; however, he reported having a “constant, squeezing” chest pain that did not worsen with palpation of the chest. The rest of his physical exam was unremarkable.
Although he reported that his EKG and chest radiograph were normal 3 days prior, repeat chest radiograph and EKG were ordered due to his unexplained, active chest pain and the lack of immediate access to the prior results.
THE DIAGNOSIS
The chest radiograph (FIGURE 1A) showed a “mildly ectatic ascending thoracic aorta” that had increased since a chest radiograph from 6 years prior (FIGURE 1B) and “was concerning for an aneurysm.” Computed tomography (CT) angiography (FIGURE 2) then confirmed a 7-cm aneurysm of the ascending aorta, with findings suggestive of a retrograde ascending aortic dissection.
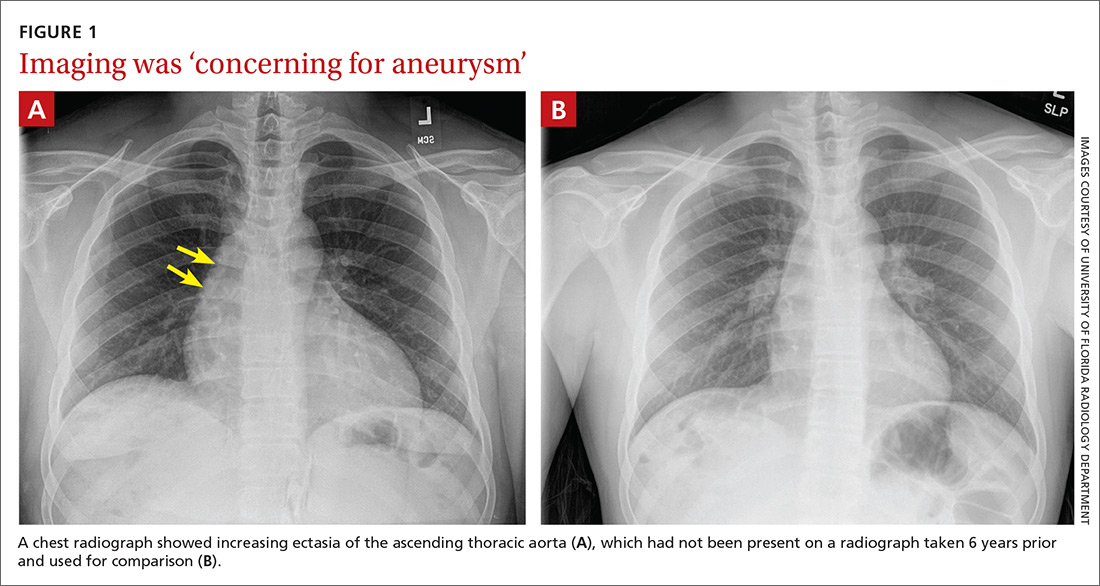
DISCUSSION
The average age of a patient with acute aortic dissection (AAD) is 63 years; only 7% occur in people younger than 40.1 AAD is often accompanied by a predisposing risk factor such as a connective tissue disease, bicuspid aortic valve, longstanding hypertension, trauma, or larger aortic dimensions.2,3 Younger patients are more likely to have predisposing risk factors of Marfan syndrome, prior aortic surgery, or a bicuspid aortic valve.3
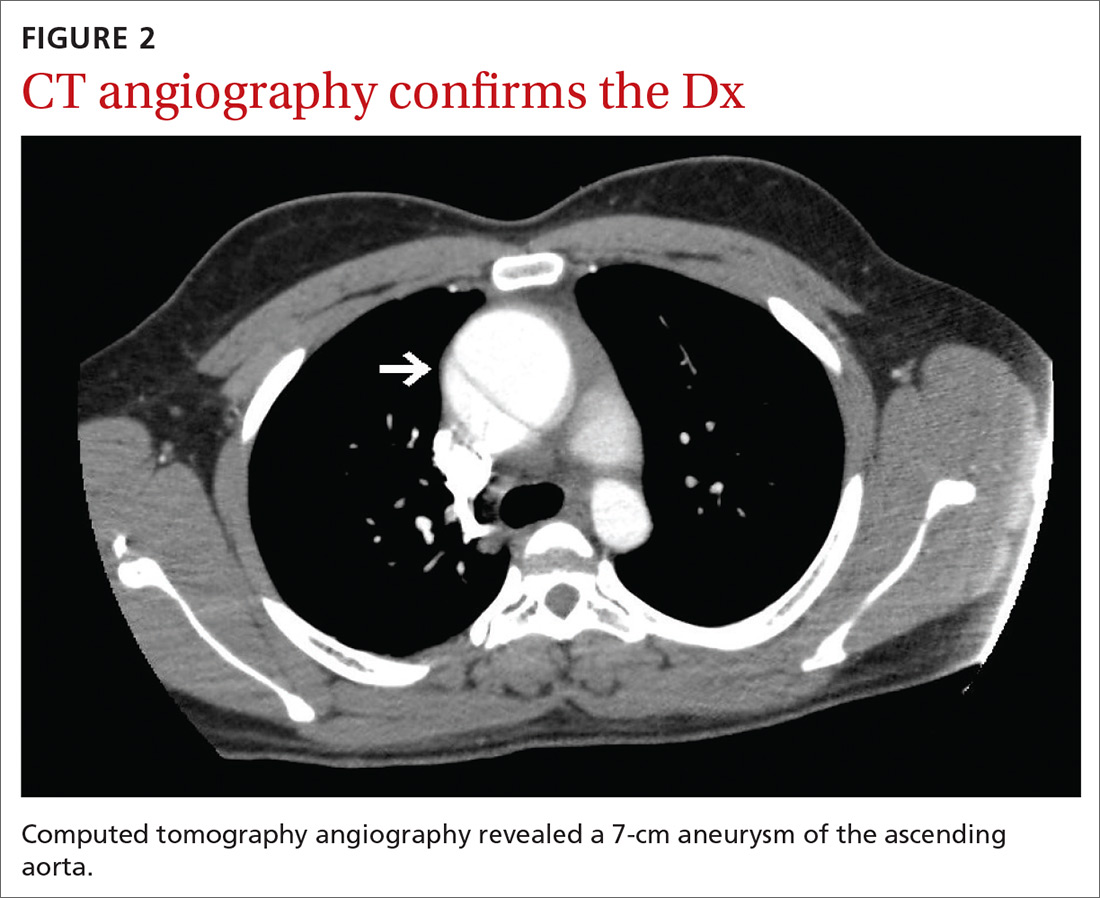
Continue to: A literature review did not reveal...
A literature review did not reveal any known correlation between the patient’s history of giant-cell hepatitis or antirejection therapy with thoracic aortic dissection. Furthermore, liver transplant is not known to be a specific risk factor for AAD in pediatric patients or outside the immediate postoperative period. Therefore, there were no known predisposing risk factors for AAD in our patient.
The most common clinical feature of AAD is chest pain, which occurs in 75% of patients.1 Other clinical symptoms include hypertension and diaphoresis.2,4 However, classic clinical findings are not always displayed, making the diagnosis difficult.2,4 The classical description of “tearing pain” is seen in only 51% of patients, and 5% to 15% of patients present without any pain.1
Commonly missed or misdiagnosed. The diagnosis of AAD has been missed during the initial exam in 38% of patients.4 As seen in our case, symptoms may be initially diagnosed as musculoskeletal chest pain. Based on symptoms, AAD can be incorrectly diagnosed as an acute myocardial infarction or vascular embolization.2,4
Every hour after symptom onset, the mortality rate of untreated AAD increases 1% to 2%,with no difference based on age.3,4 Different reports have shown mortality rates between 7% and 30%.4
Effective imaging is crucial to the diagnosis and treatment of AAD, given the occurrence of atypical presentation, missed diagnosis, and high mortality rate.4 A chest radiograph will show a widened mediastinum, but the preferred diagnostic tests are a CT or transthoracic echocardiogram.2,4 Once the diagnosis of AAD is confirmed, an aortic angiogram is the preferred test to determine the extent of the dissection prior to surgical treatment.2
Continue to: Classification dictates treatment
Classification dictates treatment. AAD is classified based on where the dissection of the aorta occurs. If the dissection involves the ascending aorta, it is classified as a type A AAD and should immediately be treated with emergent surgery in order to prevent complications including myocardial infarction, cardiac tamponade, and aortic rupture.2,4,5 If the dissection is limited to the descending aorta, it is classified as a type B AAD and can be medically managed by controlling pain and lowering blood pressure; if symptoms persist, surgical management may be required.2 After hospital discharge, AAD patients are followed closely with medical therapy, serial imaging, and reoperation if necessary.4
Our patient underwent emergent surgery for aortic root/ascending aortic replacement with a mechanical valve. He tolerated the procedure well. Surgical tissue pathology of the aortic segment showed a wall of elastic vessel with medial degeneration and dissection, and the tissue pathology of the aorta leaflets showed valvular tissue with myxoid degeneration.
THE TAKEAWAY
It is critical to keep AAD in the differential diagnosis of a patient presenting with acute onset of chest pain, as AAD often has an atypical presentation and can easily be misdiagnosed. Effective imaging is crucial to diagnosis, and immediate treatment is essential to patient survival.
CORRESPONDENCE
Rachel A. Reedy, PA, University of Florida, Department of General Pediatrics, 7046 SW Archer Road, Gainesville, FL 32608; rreedy@ufl.edu
1. Pineault J, Ouimet D, Pichette V, Vallée M. A case of aortic dissection in a young adult: a refresher of the literature of this “great masquerader.” Int J Gen Med. 2011;4:889-893.
2. Agabegi SS, Agabegi ElD, Ring AC. Diseases of the cardiovascular system. In: Jackson A, ed. Step-up to Medicine. 3rd ed. Lippincott Williams & Wilkins; 2012:54-55.
3. Januzzi JL, Isselbacher EM, Fattori R, et al. Characterizing the young patient with aortic dissection: results from the International Registry of Aortic Dissection (IRAD). J Am Coll Cardiol. 2004;43:665-669.
4. Tsai TT, Trimarchi S, Nienaber CA. Acute aortic dissection: perspectives from the International Registry of Acute Aortic Dissection (IRAD). Eur J Vasc Endovasc Surg. 2009;37:149-159.
5. Trimarchi S, Eagle KA, Nienaber CA, et al. Role of age in acute type A aortic dissection outcome: Report from the International Registry of Acute Aortic Dissection (IRAD). J Thorac Cardiovasc Surg. 2010;140:784-789.
THE CASE
A 20-year-old man presented to our clinic with a 3-day history of nonradiating chest pain located at the center of his chest. Past medical history included idiopathic neonatal giant-cell hepatitis and subsequent liver transplant at 1 month of age; he had been followed by the transplant team without rejection or infection and was in otherwise good health prior to the chest pain.
On the day of symptom onset, he was walking inside his house and fell to his knees with a chest pain described as “a punch” to the center of the chest that lasted for a few seconds. He was able to continue his daily activities without limitation despite a constant, squeezing, centrally located chest pain. The pain worsened with cough and exertion.
A few hours later, he went to an urgent care center for evaluation. There, he reported, his chest radiograph and electrocardiogram (EKG) results were normal and he was given a diagnosis of musculoskeletal chest pain. Over the next 3 days, his chest pain persisted but did not worsen. He was taking 500 mg of naproxen every 8 hours with no improvement. No other acute or chronic medications were being taken. He had no significant family history. A review of systems was otherwise negative.
On physical exam, his vital statistics included a height of 6’4”; weight, 261 lb; body mass index, 31.8; temperature, 98.7 °F; blood pressure, 134/77 mm Hg; heart rate, 92 beats/min; respiratory rate, 18 breaths/min; and oxygen saturation, 96%. Throughout the exam, he demonstrated no acute distress, appeared well, and was talkative; however, he reported having a “constant, squeezing” chest pain that did not worsen with palpation of the chest. The rest of his physical exam was unremarkable.
Although he reported that his EKG and chest radiograph were normal 3 days prior, repeat chest radiograph and EKG were ordered due to his unexplained, active chest pain and the lack of immediate access to the prior results.
THE DIAGNOSIS
The chest radiograph (FIGURE 1A) showed a “mildly ectatic ascending thoracic aorta” that had increased since a chest radiograph from 6 years prior (FIGURE 1B) and “was concerning for an aneurysm.” Computed tomography (CT) angiography (FIGURE 2) then confirmed a 7-cm aneurysm of the ascending aorta, with findings suggestive of a retrograde ascending aortic dissection.

DISCUSSION
The average age of a patient with acute aortic dissection (AAD) is 63 years; only 7% occur in people younger than 40.1 AAD is often accompanied by a predisposing risk factor such as a connective tissue disease, bicuspid aortic valve, longstanding hypertension, trauma, or larger aortic dimensions.2,3 Younger patients are more likely to have predisposing risk factors of Marfan syndrome, prior aortic surgery, or a bicuspid aortic valve.3

Continue to: A literature review did not reveal...
A literature review did not reveal any known correlation between the patient’s history of giant-cell hepatitis or antirejection therapy with thoracic aortic dissection. Furthermore, liver transplant is not known to be a specific risk factor for AAD in pediatric patients or outside the immediate postoperative period. Therefore, there were no known predisposing risk factors for AAD in our patient.
The most common clinical feature of AAD is chest pain, which occurs in 75% of patients.1 Other clinical symptoms include hypertension and diaphoresis.2,4 However, classic clinical findings are not always displayed, making the diagnosis difficult.2,4 The classical description of “tearing pain” is seen in only 51% of patients, and 5% to 15% of patients present without any pain.1
Commonly missed or misdiagnosed. The diagnosis of AAD has been missed during the initial exam in 38% of patients.4 As seen in our case, symptoms may be initially diagnosed as musculoskeletal chest pain. Based on symptoms, AAD can be incorrectly diagnosed as an acute myocardial infarction or vascular embolization.2,4
Every hour after symptom onset, the mortality rate of untreated AAD increases 1% to 2%,with no difference based on age.3,4 Different reports have shown mortality rates between 7% and 30%.4
Effective imaging is crucial to the diagnosis and treatment of AAD, given the occurrence of atypical presentation, missed diagnosis, and high mortality rate.4 A chest radiograph will show a widened mediastinum, but the preferred diagnostic tests are a CT or transthoracic echocardiogram.2,4 Once the diagnosis of AAD is confirmed, an aortic angiogram is the preferred test to determine the extent of the dissection prior to surgical treatment.2
Continue to: Classification dictates treatment
Classification dictates treatment. AAD is classified based on where the dissection of the aorta occurs. If the dissection involves the ascending aorta, it is classified as a type A AAD and should immediately be treated with emergent surgery in order to prevent complications including myocardial infarction, cardiac tamponade, and aortic rupture.2,4,5 If the dissection is limited to the descending aorta, it is classified as a type B AAD and can be medically managed by controlling pain and lowering blood pressure; if symptoms persist, surgical management may be required.2 After hospital discharge, AAD patients are followed closely with medical therapy, serial imaging, and reoperation if necessary.4
Our patient underwent emergent surgery for aortic root/ascending aortic replacement with a mechanical valve. He tolerated the procedure well. Surgical tissue pathology of the aortic segment showed a wall of elastic vessel with medial degeneration and dissection, and the tissue pathology of the aorta leaflets showed valvular tissue with myxoid degeneration.
THE TAKEAWAY
It is critical to keep AAD in the differential diagnosis of a patient presenting with acute onset of chest pain, as AAD often has an atypical presentation and can easily be misdiagnosed. Effective imaging is crucial to diagnosis, and immediate treatment is essential to patient survival.
CORRESPONDENCE
Rachel A. Reedy, PA, University of Florida, Department of General Pediatrics, 7046 SW Archer Road, Gainesville, FL 32608; rreedy@ufl.edu
THE CASE
A 20-year-old man presented to our clinic with a 3-day history of nonradiating chest pain located at the center of his chest. Past medical history included idiopathic neonatal giant-cell hepatitis and subsequent liver transplant at 1 month of age; he had been followed by the transplant team without rejection or infection and was in otherwise good health prior to the chest pain.
On the day of symptom onset, he was walking inside his house and fell to his knees with a chest pain described as “a punch” to the center of the chest that lasted for a few seconds. He was able to continue his daily activities without limitation despite a constant, squeezing, centrally located chest pain. The pain worsened with cough and exertion.
A few hours later, he went to an urgent care center for evaluation. There, he reported, his chest radiograph and electrocardiogram (EKG) results were normal and he was given a diagnosis of musculoskeletal chest pain. Over the next 3 days, his chest pain persisted but did not worsen. He was taking 500 mg of naproxen every 8 hours with no improvement. No other acute or chronic medications were being taken. He had no significant family history. A review of systems was otherwise negative.
On physical exam, his vital statistics included a height of 6’4”; weight, 261 lb; body mass index, 31.8; temperature, 98.7 °F; blood pressure, 134/77 mm Hg; heart rate, 92 beats/min; respiratory rate, 18 breaths/min; and oxygen saturation, 96%. Throughout the exam, he demonstrated no acute distress, appeared well, and was talkative; however, he reported having a “constant, squeezing” chest pain that did not worsen with palpation of the chest. The rest of his physical exam was unremarkable.
Although he reported that his EKG and chest radiograph were normal 3 days prior, repeat chest radiograph and EKG were ordered due to his unexplained, active chest pain and the lack of immediate access to the prior results.
THE DIAGNOSIS
The chest radiograph (FIGURE 1A) showed a “mildly ectatic ascending thoracic aorta” that had increased since a chest radiograph from 6 years prior (FIGURE 1B) and “was concerning for an aneurysm.” Computed tomography (CT) angiography (FIGURE 2) then confirmed a 7-cm aneurysm of the ascending aorta, with findings suggestive of a retrograde ascending aortic dissection.

DISCUSSION
The average age of a patient with acute aortic dissection (AAD) is 63 years; only 7% occur in people younger than 40.1 AAD is often accompanied by a predisposing risk factor such as a connective tissue disease, bicuspid aortic valve, longstanding hypertension, trauma, or larger aortic dimensions.2,3 Younger patients are more likely to have predisposing risk factors of Marfan syndrome, prior aortic surgery, or a bicuspid aortic valve.3

Continue to: A literature review did not reveal...
A literature review did not reveal any known correlation between the patient’s history of giant-cell hepatitis or antirejection therapy with thoracic aortic dissection. Furthermore, liver transplant is not known to be a specific risk factor for AAD in pediatric patients or outside the immediate postoperative period. Therefore, there were no known predisposing risk factors for AAD in our patient.
The most common clinical feature of AAD is chest pain, which occurs in 75% of patients.1 Other clinical symptoms include hypertension and diaphoresis.2,4 However, classic clinical findings are not always displayed, making the diagnosis difficult.2,4 The classical description of “tearing pain” is seen in only 51% of patients, and 5% to 15% of patients present without any pain.1
Commonly missed or misdiagnosed. The diagnosis of AAD has been missed during the initial exam in 38% of patients.4 As seen in our case, symptoms may be initially diagnosed as musculoskeletal chest pain. Based on symptoms, AAD can be incorrectly diagnosed as an acute myocardial infarction or vascular embolization.2,4
Every hour after symptom onset, the mortality rate of untreated AAD increases 1% to 2%,with no difference based on age.3,4 Different reports have shown mortality rates between 7% and 30%.4
Effective imaging is crucial to the diagnosis and treatment of AAD, given the occurrence of atypical presentation, missed diagnosis, and high mortality rate.4 A chest radiograph will show a widened mediastinum, but the preferred diagnostic tests are a CT or transthoracic echocardiogram.2,4 Once the diagnosis of AAD is confirmed, an aortic angiogram is the preferred test to determine the extent of the dissection prior to surgical treatment.2
Continue to: Classification dictates treatment
Classification dictates treatment. AAD is classified based on where the dissection of the aorta occurs. If the dissection involves the ascending aorta, it is classified as a type A AAD and should immediately be treated with emergent surgery in order to prevent complications including myocardial infarction, cardiac tamponade, and aortic rupture.2,4,5 If the dissection is limited to the descending aorta, it is classified as a type B AAD and can be medically managed by controlling pain and lowering blood pressure; if symptoms persist, surgical management may be required.2 After hospital discharge, AAD patients are followed closely with medical therapy, serial imaging, and reoperation if necessary.4
Our patient underwent emergent surgery for aortic root/ascending aortic replacement with a mechanical valve. He tolerated the procedure well. Surgical tissue pathology of the aortic segment showed a wall of elastic vessel with medial degeneration and dissection, and the tissue pathology of the aorta leaflets showed valvular tissue with myxoid degeneration.
THE TAKEAWAY
It is critical to keep AAD in the differential diagnosis of a patient presenting with acute onset of chest pain, as AAD often has an atypical presentation and can easily be misdiagnosed. Effective imaging is crucial to diagnosis, and immediate treatment is essential to patient survival.
CORRESPONDENCE
Rachel A. Reedy, PA, University of Florida, Department of General Pediatrics, 7046 SW Archer Road, Gainesville, FL 32608; rreedy@ufl.edu
1. Pineault J, Ouimet D, Pichette V, Vallée M. A case of aortic dissection in a young adult: a refresher of the literature of this “great masquerader.” Int J Gen Med. 2011;4:889-893.
2. Agabegi SS, Agabegi ElD, Ring AC. Diseases of the cardiovascular system. In: Jackson A, ed. Step-up to Medicine. 3rd ed. Lippincott Williams & Wilkins; 2012:54-55.
3. Januzzi JL, Isselbacher EM, Fattori R, et al. Characterizing the young patient with aortic dissection: results from the International Registry of Aortic Dissection (IRAD). J Am Coll Cardiol. 2004;43:665-669.
4. Tsai TT, Trimarchi S, Nienaber CA. Acute aortic dissection: perspectives from the International Registry of Acute Aortic Dissection (IRAD). Eur J Vasc Endovasc Surg. 2009;37:149-159.
5. Trimarchi S, Eagle KA, Nienaber CA, et al. Role of age in acute type A aortic dissection outcome: Report from the International Registry of Acute Aortic Dissection (IRAD). J Thorac Cardiovasc Surg. 2010;140:784-789.
1. Pineault J, Ouimet D, Pichette V, Vallée M. A case of aortic dissection in a young adult: a refresher of the literature of this “great masquerader.” Int J Gen Med. 2011;4:889-893.
2. Agabegi SS, Agabegi ElD, Ring AC. Diseases of the cardiovascular system. In: Jackson A, ed. Step-up to Medicine. 3rd ed. Lippincott Williams & Wilkins; 2012:54-55.
3. Januzzi JL, Isselbacher EM, Fattori R, et al. Characterizing the young patient with aortic dissection: results from the International Registry of Aortic Dissection (IRAD). J Am Coll Cardiol. 2004;43:665-669.
4. Tsai TT, Trimarchi S, Nienaber CA. Acute aortic dissection: perspectives from the International Registry of Acute Aortic Dissection (IRAD). Eur J Vasc Endovasc Surg. 2009;37:149-159.
5. Trimarchi S, Eagle KA, Nienaber CA, et al. Role of age in acute type A aortic dissection outcome: Report from the International Registry of Acute Aortic Dissection (IRAD). J Thorac Cardiovasc Surg. 2010;140:784-789.