User login
Monkeypox: Another emerging threat?
CASE Pregnant woman’s husband is ill after traveling
A 29-year-old primigravid woman at 18 weeks’ gestation just returned from a 10-day trip to Nigeria with her husband. While in Nigeria, the couple went on safari. On several occasions during the safari, they consumed bushmeat prepared by their guides. Her husband now has severe malaise, fever, chills, myalgias, cough, and prominent submandibular, cervical, and inguinal adenopathy. In addition, he has developed a diffuse papular-vesicular rash on his trunk and extremities.
- What is the most likely diagnosis?
- Does this condition pose a danger to his wife?
- What treatment is indicated for his wife?
What we know
In recent weeks, the specter of another poorly understood biological threat has emerged in the medical literature and lay press: monkeypox. This article will first review the epidemiology, clinical manifestations, and diagnosis of this infection, followed by a discussion of how to prevent and treat the condition, with special emphasis on the risks that this infection poses in pregnant women.
Virology
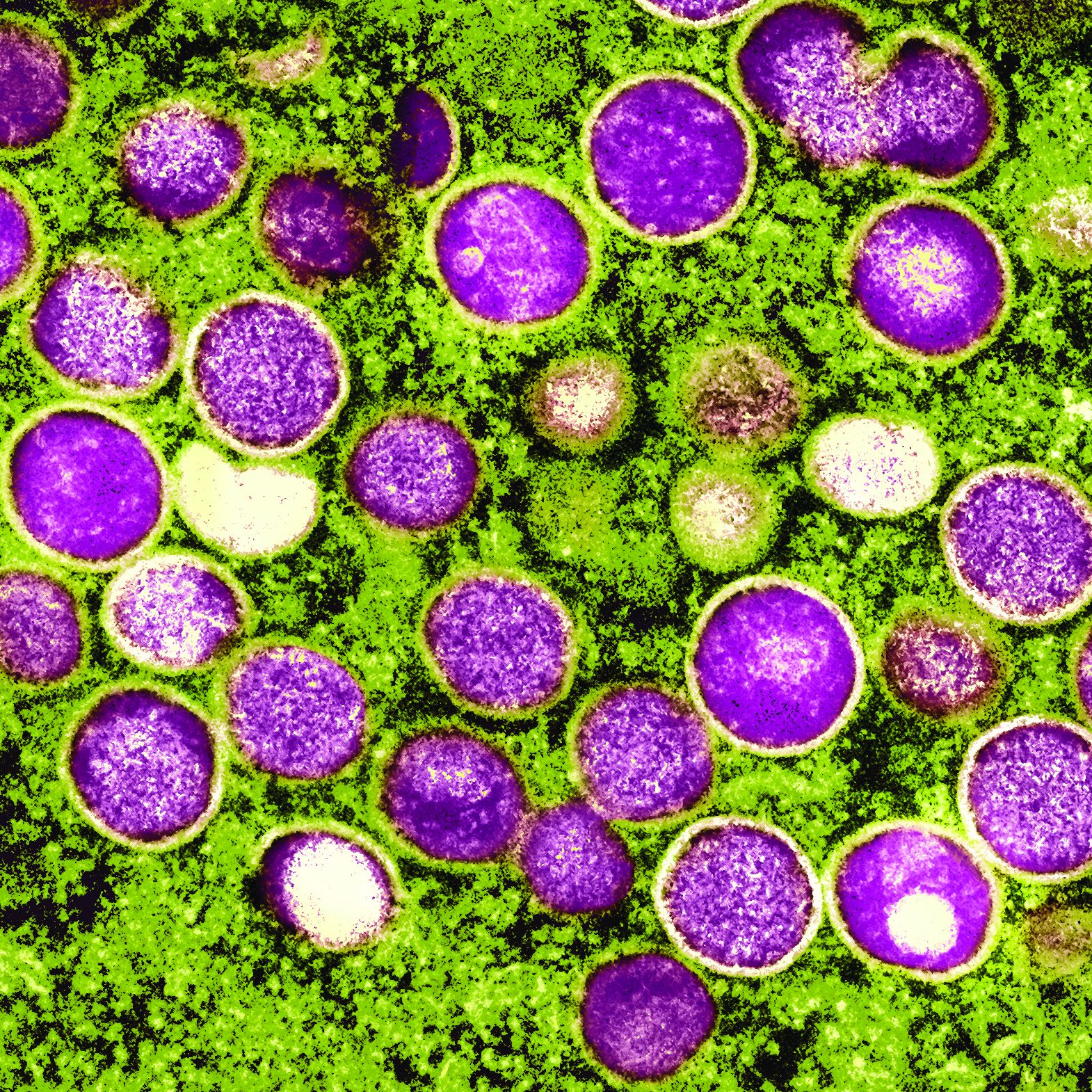
The monkeypox virus is a member of the orthopoxvirus genus. The variola (smallpox) virus and vaccinia virus are included in this genus. It is one of the largest of all viruses, measuring 200-250 nm. It is enveloped and contains double-stranded DNA. Its natural reservoir is probably African rodents. Two distinct strains of monkeypox exist in different geographical regions of Africa: the Central African clade and the West African clade. The Central African clade is significantly more virulent than the latter, with a mortality rate approaching 10%, versus 1% in the West African clade. The incubation period of the virus ranges from 4-20 days and averages 12 days.1,2
Epidemiology
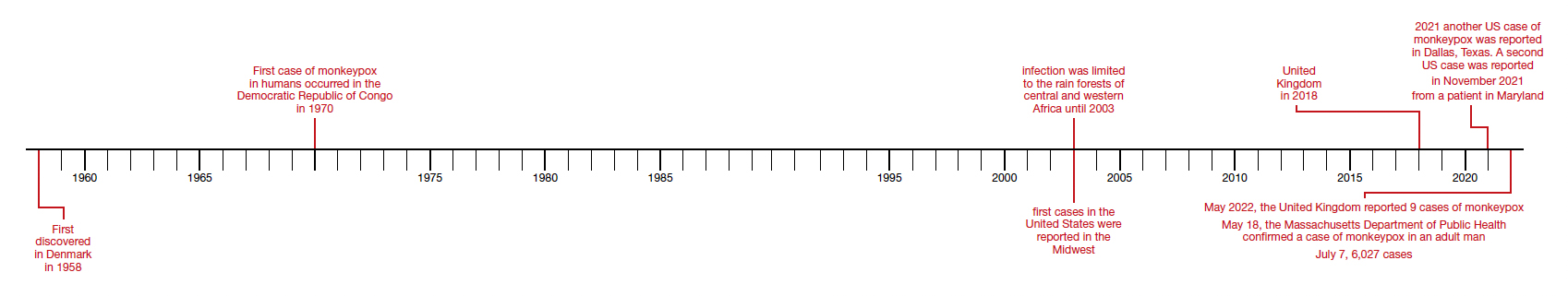
Monkeypox was first discovered in 1958 by Preben von Magnus in a colony of research monkeys in Copenhagen, Denmark. The first case of monkeypox in humans occurred in the Democratic Republic of Congo in 1970 in a 9-year-old boy. Subsequently, cases were reported in the Ivory Coast, Liberia, Nigeria, and Sierra Leone. The infection was limited to the rain forests of central and western Africa until 2003. At that time, the first cases in the United States were reported. The US cases occurred in the Midwest and were traced to exposure to pet prairie dogs. These animals all came from a single distributor, and they apparently were infected when they were housed in the same space with Gambian rats, which are well recognized reservoirs of monkeypox in their native habitat in Africa.1-3
A limited outbreak of monkeypox occurred in the United Kingdom in 2018. Seventy-one cases, with no fatalities, were reported. In 2021 another US case of monkeypox was reported in Dallas, Texas, in an individual who had recently traveled to the United States from Nigeria. A second US case was reported in November 2021 from a patient in Maryland who had returned from a visit to Nigeria. Those were the only 2 reported cases of monkeypox in the United States in 2021.1-3
Then in early May 2022, the United Kingdom reported 9 cases of monkeypox. The first infected patient had recently traveled to Nigeria and, subsequently, infected 2 members of his family.4 On May 18, the Massachusetts Department of Public Health confirmed a case of monkeypox in an adult man who had recently traveled to Canada. As of July 7, 6,027 cases have been reported from at least 39 countries.
The current outbreak is unusual in that, previously, almost all cases occurred in western and central Africa in remote tropical rain forests. Infection usually resulted from close exposure to rats, rabbits, squirrels, monkeys, porcupines, and gazelles. Exposure occurred when persons captured, slaughtered, prepared, and then ate these animals for food without properly cooking the flesh.
The leading theory is that the present outbreak originated among men who had sex with men at 2 raves held in Spain and Belgium. The virus appears to have been spread by skin-to-skin contact, by respiratory droplets, by contact with contaminated bedding, and probably by sperm.2,4,6
Continue to: Clinical manifestations...
Clinical manifestations
Monkeypox evolves through 2 stages: a pre-eruptive stage and an eruptive stage. Prodromal symptoms include malaise, severe headache, myalgias, fever, drenching sweats, backache, fatigue, sore throat, dyspnea, and cough. Within 2-3 days, the characteristic skin eruption develops. The lesions usually begin on the face and then spread in a centrifugal manner to the trunk and extremities, including the palms of the hands and soles of the feet. The lesions typically progress from macules to papules to vesicles to pustules. They then crust and scab over. An interesting additional finding is the presence of prominent lymphadenopathy behind the ear, beneath the mandible, in the neck, and in the groin.1
Several different illnesses must be considered in the differential diagnosis of monkeypox infection. They include measles, scabies, secondary syphilis, and medication-associated allergic reactions. However, the 2 conditions most likely to be confused with monkeypox are chickenpox (varicella) and smallpox. Lymphadenopathy is much more prominent in monkeypox compared with chickenpox. Moreover, with monkeypox, all lesions tend to be at the same stage of evolution as opposed to appearing in crops as they do in chickenpox. Smallpox would be extremely unlikely in the absence of a recognized laboratory accident or a bioterrorism incident.7
Diagnosis
The presumptive diagnosis of monkeypox infection is made primarily based on clinical examination. However, laboratory testing is indicated to definitively differentiate monkeypox from other orthopoxvirus infections such as varicella and smallpox.
In specialized laboratories that employ highly trained personnel and maintain strict safety precautions, the virus can be isolated in mammalian cell cultures. Electron microscopy is a valuable tool for identifying the characteristic brick-shaped poxvirus virions. Routine histologic examination of a lesion will show ballooning degeneration of keratinocytes, prominent spongiosis, dermal edema, and acute inflammation, although these findings are not unique to monkeypox.1
The Centers for Disease Control and Prevention (CDC) has developed serologic tests that detect immunoglobulin (Ig) M- and IgG-specific antibody. However, the most useful and practical diagnostic test is assessment of a skin scraping by polymerase chain reaction (PCR). This test is more sensitive than assessment of serum PCR.1
When the diagnosis of monkeypox is being considered, the clinician should coordinate testing through the local and state public health departments and through the CDC. Effective communication with all agencies will ensure that laboratory specimens are processed in a timely and efficient manner. The CDC website presents information on specimen collection.8
How do we manage monkeypox?
Prevention
The first step in prevention of infection is to isolate infected individuals until all lesions have dried and crusted over. Susceptible people should avoid close contact with skin lesions, respiratory and genital secretions, and bedding of patients who are infected.
The ultimate preventive measure, however, is vaccination of susceptible people either immediately before exposure (eg, military personnel, first responders, infection control investigators, health care workers) or immediately after exposure (general population). Older individuals who received the original smallpox vaccine likely have immunity to monkeypox infection. Unfortunately, very few women who currently are of reproductive age received this vaccine because its use was discontinued in the United States in the early 1970s. Therefore, the vast majority of our patients are uniquely susceptible to this infection and should be vaccinated if there is an outbreak of monkeypox in their locality.7,9
The current preferred vaccine for prevention of both smallpox and monkeypox is the Jynneos (Bavarian Nordic A/S) vaccine.10 This agent incorporates a replication-deficient live virus and does not pose the same risk for adverse events as the original versions of the smallpox vaccine. Jynneos is administered subcutaneously rather than by scarification. Two 0.5-mL doses, delivered 28 days apart, are required for optimal effect. The vaccine must be obtained from local and state health departments, in consultation with the CDC.7,9
There is very little published information on the safety of the Jynneos vaccine in pregnant or lactating women, although animal data are reassuring. Moreover, the dangers of monkeypox infection are significant, and in the event of an outbreak, vaccination of susceptible individuals, including pregnant women, is indicated.
- Monkeypox is a member of the orthopoxvirus genus and is closely related to the smallpox virus. It is a large, double-stranded, enveloped DNA virus.
- The virus is transmitted primarily by close contact with infected animals or other humans or by consumption of contaminated bushmeat.
- The infection evolves in 2 phases. The pre-eruptive phase is characterized by severe flu-like symptoms and signs. The eruptive phase is distinguished by a diffuse papular-vesicular rash.
- The most valuable test for confirming the diagnosis is a polymerase chain reaction test of a fresh skin lesion.
- In women who are pregnant, monkeypox has been associated with spontaneous abortion and fetal death.
- Three antiviral agents may be of value in treating infected patients: cidofovir, brincidofovir, and tecovirimat. Only the latter has an acceptable safety profile for women who are pregnant or lactating.
- The new nonreplicating smallpox vaccine Jynneos (Bavarian Nordic A/S) is of great value for pre- and post-exposure prophylaxis.
Continue to: Treatment...
Treatment
Infected pregnant women should receive acetaminophen 1,000 mg orally every 8 hours, to control fever and provide analgesia. An antihistamine such as diphenhydramine 25 mg orally every 6-8 hours, may be used to control pruritus and provide mild sedation. Adequate fluid intake and optimal nutrition should be encouraged. Skin lesions should be inspected regularly to detect signs of superimposed bacterial infections. Small, localized bacterial skin infections can be treated with topical application of mupirocin ointment 2%, 3 times daily for 7-14 days. For diffuse and more severe bacterial skin infections, a systemic antibiotic may be necessary. Reasonable choices include amoxicillin-clavulanate 875 mg/125 mg orally every 12 hours, or trimethoprim-sulfamethoxazole double strength 800 mg/160 mg orally every 12 hours.11 The latter agent should be avoided in the first trimester of pregnancy because of potential teratogenic effects.
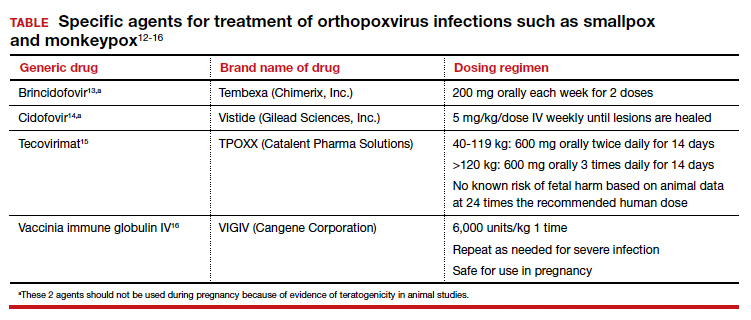
Several specific agents are available through the CDC for treatment of orthopoxvirus infections such as smallpox and monkeypox. Information about these agents is summarized in the TABLE.12-16
Unique considerations in pregnancy
Because monkeypox is so rare, there is very little information about the effects of this infection in pregnant women. The report most commonly cited in the literature is that by Mbala et al, which was published in 2017.17 These authors described 4 pregnant patients in the Democratic Republic of Congo who contracted monkeypox infection over a 4-year period. All 4 women were hospitalized and treated with systemic antibiotics, antiparasitic medications, and analgesics. One patient delivered a healthy infant. Two women had spontaneous abortions in the first trimester. The fourth patient experienced a stillbirth at 22 weeks’ gestation. At postmortem examination, the fetus had diffuse cutaneous lesions, prominent hepatomegaly, and hydrops. No structural malformations were noted. The placenta demonstrated numerous punctate hemorrhages, and high concentrations of virus were recovered from the placenta and from fetal tissue.
Although the information on pregnancy outcome is quite limited, it seems clear that the virus can cross the placenta and cause adverse effects such as spontaneous abortion and fetal death. Accordingly, I think the following guidelines are a reasonable approach to a pregnant patient who has been exposed to monkeypox or who has developed manifestations of infection.3,7,9
- In the event of a community outbreak, bioterrorism event, or exposure to a person with suspected or confirmed monkeypox infection, the pregnant patient should receive the Jynneos vaccine.
- The pregnant patient should be isolated from any individual with suspected or confirmed monkeypox.
- If infection develops despite these measures, the patient should be treated with either tecovirimat or vaccinia immune globulin IV. Hospitalization may be necessary for seriously ill individuals.
- Within 2 weeks of infection, a comprehensive ultrasound examination should be performed to assess for structural abnormalities in the fetus.
- Subsequently, serial ultrasound examinations should be performed at intervals of 4-6 weeks to assess fetal growth and re-evaluate fetal anatomy.
- Following delivery, a detailed neonatal examination should be performed to assess for evidence of viral injury. Neonatal skin lesions and neonatal serum can be assessed by PCR for monkeypox virus. The newborn should be isolated from the mother until all the mother’s lesions have dried and crusted over.
CASE Resolved
Given the husband’s recent travel to Nigeria and consumption of bushmeat, he most likely has monkeypox. The infection can be spread from person to person by close contact; thus, his wife is at risk. The couple should isolate until all of his lesions have dried and crusted over. The woman also should receive the Jynneos vaccine. If she becomes symptomatic, she should be treated with tecovirimat or vaccinia immune globulin IV. ●
- Isaacs SN, Shenoy ES. Monkeypox. UpToDate. Updated June 28,2022. Accessed July 1, 2022. https://www.uptodate.com /contents/monkeypox?topicRef=8349&source=see_link
- Graham MB. Monkeypox. Medscape. Updated June 29, 2022. Accessed July 1, 2022. https://emedicine.medscape.com /article/1134714-overview.
- Khalil A, Samara A, O’Brien P, et al. Monkeypox and pregnancy: what do obstetricians need to know? Ultrasound Obstet Gynecol. 2022;60:22-27. doi:10.1002/uog.24968.
- World Health Organization. Monkeypox-United Kingdom of Great Britain and Northern Ireland. May 18, 2022. Accessed July 1, 2022. https://www.who.int/emergencies/diseaseoutbreak-news/item/2022-DON383.
- WHO reports two new monkeypox deaths, cases in new areas. Reuters. July 7, 2022. https://www.reuters.com/world /who-reports-two-new-monkeypox-deaths-2022-07-07/. Accessed July 19, 2022.
- World Health Organization. Multi-country monkeypox outbreak in non-endemic countries: update. May 29, 2022. Accessed July 1, 2022. https://www.who.int /emergencies/disease-outbreak-news/item/2022 -DON388#:~:text=Multi%2Dcountry%20monkeypox%20 outbreak%20in%20non%2Dendemic%20countries%3A%20 Update,-29%20May%202022&text=Since%2013%20 May%202022%2C%20monkeypox,Epidemiological%20 investigations%20are%20ongoing.
- Cono J, Cragan JD, Jamieson DJ, Rasmussen SA. Prophylaxis and treatment of pregnant women for emerging infections andbioterrorism emergencies. Emerg Infect Dis. 2006;12:16311637. doi:10.3201/eid1211.060618.
- Centers for Disease Control and Prevention. Preparation and collection of specimens. Reviewed June 29, 2022. Accessed July 6, 2022. https://www.cdc.gov/poxvirus /monkeypox/clinicians/prep-collection-specimens.html.
- Rao AK, Petersen BW, Whitehill F, et al. Monkeypox vaccination. MMWR Morb Mortal Wkly Rep. 2022;71:734-742. doi:10.15585/mmwr.mm7122e1.
- Smallpox and monkeypox vaccine, live, nonreplicating. Package insert. Bavarian Nordic A/S; 2021. Accessed July 1, 2022. https://www.fda.gov/media/131078/download.
- Duff P. Commonly used antibiotics in ObGyn practice. OBG Manag. 2022;34:29, 36-40. doi:10.12788/obgm.0191.
- Centers for Disease Control and Prevention. Treatment information for healthcare professionals: interim clinical guidance for the treatment of monkeypox. Reviewed June 17, 2022. Accessed July 1, 2022. https://www.cdc.gov/poxvirus /monkeypox/clinicians/treatment.html.
- Brincidofovir. Prescribing information. Chimerix, Inc.; 2021. Accessed July 1, 2022. https://www.accessdata.fda.gov /drugsatfda_docs/label/2021/214460s000,214461s000lbl.pdf.
- Cidofovir. Package insert. Gilead Sciences, Inc.; 2010. Accessed July 1, 2022. https://www.gilead.com/~/media /Files/pdfs/medicines/other/vistide/vistide.pdf.
- Tecovirimat. Prescribing information. Catalent Pharma Solutions; 2022. Accessed July 1, 2022. https://www.accessdata.fda.gov/drugsatfda_docs /label/2022/214518s000lbl.pdf.
- Vaccinia immune globulin IV. Prescribing information. Cangene Corporation; 2010. Accessed July 1, 2022. https: //www.fda.gov/media/77004/download.
- Mbala PK, Huggins JW, Riu-Rovira T, et al. Maternal and fetal outcomes among pregnant women with human monkeypox infection in the Democratic Republic of Congo. J Infect Dis. 2017;216:824-828. doi:10.1093/infdis/jix260.
CASE Pregnant woman’s husband is ill after traveling
A 29-year-old primigravid woman at 18 weeks’ gestation just returned from a 10-day trip to Nigeria with her husband. While in Nigeria, the couple went on safari. On several occasions during the safari, they consumed bushmeat prepared by their guides. Her husband now has severe malaise, fever, chills, myalgias, cough, and prominent submandibular, cervical, and inguinal adenopathy. In addition, he has developed a diffuse papular-vesicular rash on his trunk and extremities.
- What is the most likely diagnosis?
- Does this condition pose a danger to his wife?
- What treatment is indicated for his wife?
What we know
In recent weeks, the specter of another poorly understood biological threat has emerged in the medical literature and lay press: monkeypox. This article will first review the epidemiology, clinical manifestations, and diagnosis of this infection, followed by a discussion of how to prevent and treat the condition, with special emphasis on the risks that this infection poses in pregnant women.
Virology
The monkeypox virus is a member of the orthopoxvirus genus. The variola (smallpox) virus and vaccinia virus are included in this genus. It is one of the largest of all viruses, measuring 200-250 nm. It is enveloped and contains double-stranded DNA. Its natural reservoir is probably African rodents. Two distinct strains of monkeypox exist in different geographical regions of Africa: the Central African clade and the West African clade. The Central African clade is significantly more virulent than the latter, with a mortality rate approaching 10%, versus 1% in the West African clade. The incubation period of the virus ranges from 4-20 days and averages 12 days.1,2
Epidemiology
Monkeypox was first discovered in 1958 by Preben von Magnus in a colony of research monkeys in Copenhagen, Denmark. The first case of monkeypox in humans occurred in the Democratic Republic of Congo in 1970 in a 9-year-old boy. Subsequently, cases were reported in the Ivory Coast, Liberia, Nigeria, and Sierra Leone. The infection was limited to the rain forests of central and western Africa until 2003. At that time, the first cases in the United States were reported. The US cases occurred in the Midwest and were traced to exposure to pet prairie dogs. These animals all came from a single distributor, and they apparently were infected when they were housed in the same space with Gambian rats, which are well recognized reservoirs of monkeypox in their native habitat in Africa.1-3
A limited outbreak of monkeypox occurred in the United Kingdom in 2018. Seventy-one cases, with no fatalities, were reported. In 2021 another US case of monkeypox was reported in Dallas, Texas, in an individual who had recently traveled to the United States from Nigeria. A second US case was reported in November 2021 from a patient in Maryland who had returned from a visit to Nigeria. Those were the only 2 reported cases of monkeypox in the United States in 2021.1-3
Then in early May 2022, the United Kingdom reported 9 cases of monkeypox. The first infected patient had recently traveled to Nigeria and, subsequently, infected 2 members of his family.4 On May 18, the Massachusetts Department of Public Health confirmed a case of monkeypox in an adult man who had recently traveled to Canada. As of July 7, 6,027 cases have been reported from at least 39 countries.
The current outbreak is unusual in that, previously, almost all cases occurred in western and central Africa in remote tropical rain forests. Infection usually resulted from close exposure to rats, rabbits, squirrels, monkeys, porcupines, and gazelles. Exposure occurred when persons captured, slaughtered, prepared, and then ate these animals for food without properly cooking the flesh.
The leading theory is that the present outbreak originated among men who had sex with men at 2 raves held in Spain and Belgium. The virus appears to have been spread by skin-to-skin contact, by respiratory droplets, by contact with contaminated bedding, and probably by sperm.2,4,6
Continue to: Clinical manifestations...
Clinical manifestations
Monkeypox evolves through 2 stages: a pre-eruptive stage and an eruptive stage. Prodromal symptoms include malaise, severe headache, myalgias, fever, drenching sweats, backache, fatigue, sore throat, dyspnea, and cough. Within 2-3 days, the characteristic skin eruption develops. The lesions usually begin on the face and then spread in a centrifugal manner to the trunk and extremities, including the palms of the hands and soles of the feet. The lesions typically progress from macules to papules to vesicles to pustules. They then crust and scab over. An interesting additional finding is the presence of prominent lymphadenopathy behind the ear, beneath the mandible, in the neck, and in the groin.1
Several different illnesses must be considered in the differential diagnosis of monkeypox infection. They include measles, scabies, secondary syphilis, and medication-associated allergic reactions. However, the 2 conditions most likely to be confused with monkeypox are chickenpox (varicella) and smallpox. Lymphadenopathy is much more prominent in monkeypox compared with chickenpox. Moreover, with monkeypox, all lesions tend to be at the same stage of evolution as opposed to appearing in crops as they do in chickenpox. Smallpox would be extremely unlikely in the absence of a recognized laboratory accident or a bioterrorism incident.7
Diagnosis
The presumptive diagnosis of monkeypox infection is made primarily based on clinical examination. However, laboratory testing is indicated to definitively differentiate monkeypox from other orthopoxvirus infections such as varicella and smallpox.
In specialized laboratories that employ highly trained personnel and maintain strict safety precautions, the virus can be isolated in mammalian cell cultures. Electron microscopy is a valuable tool for identifying the characteristic brick-shaped poxvirus virions. Routine histologic examination of a lesion will show ballooning degeneration of keratinocytes, prominent spongiosis, dermal edema, and acute inflammation, although these findings are not unique to monkeypox.1
The Centers for Disease Control and Prevention (CDC) has developed serologic tests that detect immunoglobulin (Ig) M- and IgG-specific antibody. However, the most useful and practical diagnostic test is assessment of a skin scraping by polymerase chain reaction (PCR). This test is more sensitive than assessment of serum PCR.1
When the diagnosis of monkeypox is being considered, the clinician should coordinate testing through the local and state public health departments and through the CDC. Effective communication with all agencies will ensure that laboratory specimens are processed in a timely and efficient manner. The CDC website presents information on specimen collection.8
How do we manage monkeypox?
Prevention
The first step in prevention of infection is to isolate infected individuals until all lesions have dried and crusted over. Susceptible people should avoid close contact with skin lesions, respiratory and genital secretions, and bedding of patients who are infected.
The ultimate preventive measure, however, is vaccination of susceptible people either immediately before exposure (eg, military personnel, first responders, infection control investigators, health care workers) or immediately after exposure (general population). Older individuals who received the original smallpox vaccine likely have immunity to monkeypox infection. Unfortunately, very few women who currently are of reproductive age received this vaccine because its use was discontinued in the United States in the early 1970s. Therefore, the vast majority of our patients are uniquely susceptible to this infection and should be vaccinated if there is an outbreak of monkeypox in their locality.7,9
The current preferred vaccine for prevention of both smallpox and monkeypox is the Jynneos (Bavarian Nordic A/S) vaccine.10 This agent incorporates a replication-deficient live virus and does not pose the same risk for adverse events as the original versions of the smallpox vaccine. Jynneos is administered subcutaneously rather than by scarification. Two 0.5-mL doses, delivered 28 days apart, are required for optimal effect. The vaccine must be obtained from local and state health departments, in consultation with the CDC.7,9
There is very little published information on the safety of the Jynneos vaccine in pregnant or lactating women, although animal data are reassuring. Moreover, the dangers of monkeypox infection are significant, and in the event of an outbreak, vaccination of susceptible individuals, including pregnant women, is indicated.
- Monkeypox is a member of the orthopoxvirus genus and is closely related to the smallpox virus. It is a large, double-stranded, enveloped DNA virus.
- The virus is transmitted primarily by close contact with infected animals or other humans or by consumption of contaminated bushmeat.
- The infection evolves in 2 phases. The pre-eruptive phase is characterized by severe flu-like symptoms and signs. The eruptive phase is distinguished by a diffuse papular-vesicular rash.
- The most valuable test for confirming the diagnosis is a polymerase chain reaction test of a fresh skin lesion.
- In women who are pregnant, monkeypox has been associated with spontaneous abortion and fetal death.
- Three antiviral agents may be of value in treating infected patients: cidofovir, brincidofovir, and tecovirimat. Only the latter has an acceptable safety profile for women who are pregnant or lactating.
- The new nonreplicating smallpox vaccine Jynneos (Bavarian Nordic A/S) is of great value for pre- and post-exposure prophylaxis.
Continue to: Treatment...
Treatment
Infected pregnant women should receive acetaminophen 1,000 mg orally every 8 hours, to control fever and provide analgesia. An antihistamine such as diphenhydramine 25 mg orally every 6-8 hours, may be used to control pruritus and provide mild sedation. Adequate fluid intake and optimal nutrition should be encouraged. Skin lesions should be inspected regularly to detect signs of superimposed bacterial infections. Small, localized bacterial skin infections can be treated with topical application of mupirocin ointment 2%, 3 times daily for 7-14 days. For diffuse and more severe bacterial skin infections, a systemic antibiotic may be necessary. Reasonable choices include amoxicillin-clavulanate 875 mg/125 mg orally every 12 hours, or trimethoprim-sulfamethoxazole double strength 800 mg/160 mg orally every 12 hours.11 The latter agent should be avoided in the first trimester of pregnancy because of potential teratogenic effects.
Several specific agents are available through the CDC for treatment of orthopoxvirus infections such as smallpox and monkeypox. Information about these agents is summarized in the TABLE.12-16

Unique considerations in pregnancy
Because monkeypox is so rare, there is very little information about the effects of this infection in pregnant women. The report most commonly cited in the literature is that by Mbala et al, which was published in 2017.17 These authors described 4 pregnant patients in the Democratic Republic of Congo who contracted monkeypox infection over a 4-year period. All 4 women were hospitalized and treated with systemic antibiotics, antiparasitic medications, and analgesics. One patient delivered a healthy infant. Two women had spontaneous abortions in the first trimester. The fourth patient experienced a stillbirth at 22 weeks’ gestation. At postmortem examination, the fetus had diffuse cutaneous lesions, prominent hepatomegaly, and hydrops. No structural malformations were noted. The placenta demonstrated numerous punctate hemorrhages, and high concentrations of virus were recovered from the placenta and from fetal tissue.
Although the information on pregnancy outcome is quite limited, it seems clear that the virus can cross the placenta and cause adverse effects such as spontaneous abortion and fetal death. Accordingly, I think the following guidelines are a reasonable approach to a pregnant patient who has been exposed to monkeypox or who has developed manifestations of infection.3,7,9
- In the event of a community outbreak, bioterrorism event, or exposure to a person with suspected or confirmed monkeypox infection, the pregnant patient should receive the Jynneos vaccine.
- The pregnant patient should be isolated from any individual with suspected or confirmed monkeypox.
- If infection develops despite these measures, the patient should be treated with either tecovirimat or vaccinia immune globulin IV. Hospitalization may be necessary for seriously ill individuals.
- Within 2 weeks of infection, a comprehensive ultrasound examination should be performed to assess for structural abnormalities in the fetus.
- Subsequently, serial ultrasound examinations should be performed at intervals of 4-6 weeks to assess fetal growth and re-evaluate fetal anatomy.
- Following delivery, a detailed neonatal examination should be performed to assess for evidence of viral injury. Neonatal skin lesions and neonatal serum can be assessed by PCR for monkeypox virus. The newborn should be isolated from the mother until all the mother’s lesions have dried and crusted over.
CASE Resolved
Given the husband’s recent travel to Nigeria and consumption of bushmeat, he most likely has monkeypox. The infection can be spread from person to person by close contact; thus, his wife is at risk. The couple should isolate until all of his lesions have dried and crusted over. The woman also should receive the Jynneos vaccine. If she becomes symptomatic, she should be treated with tecovirimat or vaccinia immune globulin IV. ●

CASE Pregnant woman’s husband is ill after traveling
A 29-year-old primigravid woman at 18 weeks’ gestation just returned from a 10-day trip to Nigeria with her husband. While in Nigeria, the couple went on safari. On several occasions during the safari, they consumed bushmeat prepared by their guides. Her husband now has severe malaise, fever, chills, myalgias, cough, and prominent submandibular, cervical, and inguinal adenopathy. In addition, he has developed a diffuse papular-vesicular rash on his trunk and extremities.
- What is the most likely diagnosis?
- Does this condition pose a danger to his wife?
- What treatment is indicated for his wife?
What we know
In recent weeks, the specter of another poorly understood biological threat has emerged in the medical literature and lay press: monkeypox. This article will first review the epidemiology, clinical manifestations, and diagnosis of this infection, followed by a discussion of how to prevent and treat the condition, with special emphasis on the risks that this infection poses in pregnant women.
Virology
The monkeypox virus is a member of the orthopoxvirus genus. The variola (smallpox) virus and vaccinia virus are included in this genus. It is one of the largest of all viruses, measuring 200-250 nm. It is enveloped and contains double-stranded DNA. Its natural reservoir is probably African rodents. Two distinct strains of monkeypox exist in different geographical regions of Africa: the Central African clade and the West African clade. The Central African clade is significantly more virulent than the latter, with a mortality rate approaching 10%, versus 1% in the West African clade. The incubation period of the virus ranges from 4-20 days and averages 12 days.1,2
Epidemiology
Monkeypox was first discovered in 1958 by Preben von Magnus in a colony of research monkeys in Copenhagen, Denmark. The first case of monkeypox in humans occurred in the Democratic Republic of Congo in 1970 in a 9-year-old boy. Subsequently, cases were reported in the Ivory Coast, Liberia, Nigeria, and Sierra Leone. The infection was limited to the rain forests of central and western Africa until 2003. At that time, the first cases in the United States were reported. The US cases occurred in the Midwest and were traced to exposure to pet prairie dogs. These animals all came from a single distributor, and they apparently were infected when they were housed in the same space with Gambian rats, which are well recognized reservoirs of monkeypox in their native habitat in Africa.1-3
A limited outbreak of monkeypox occurred in the United Kingdom in 2018. Seventy-one cases, with no fatalities, were reported. In 2021 another US case of monkeypox was reported in Dallas, Texas, in an individual who had recently traveled to the United States from Nigeria. A second US case was reported in November 2021 from a patient in Maryland who had returned from a visit to Nigeria. Those were the only 2 reported cases of monkeypox in the United States in 2021.1-3
Then in early May 2022, the United Kingdom reported 9 cases of monkeypox. The first infected patient had recently traveled to Nigeria and, subsequently, infected 2 members of his family.4 On May 18, the Massachusetts Department of Public Health confirmed a case of monkeypox in an adult man who had recently traveled to Canada. As of July 7, 6,027 cases have been reported from at least 39 countries.
The current outbreak is unusual in that, previously, almost all cases occurred in western and central Africa in remote tropical rain forests. Infection usually resulted from close exposure to rats, rabbits, squirrels, monkeys, porcupines, and gazelles. Exposure occurred when persons captured, slaughtered, prepared, and then ate these animals for food without properly cooking the flesh.
The leading theory is that the present outbreak originated among men who had sex with men at 2 raves held in Spain and Belgium. The virus appears to have been spread by skin-to-skin contact, by respiratory droplets, by contact with contaminated bedding, and probably by sperm.2,4,6
Continue to: Clinical manifestations...
Clinical manifestations
Monkeypox evolves through 2 stages: a pre-eruptive stage and an eruptive stage. Prodromal symptoms include malaise, severe headache, myalgias, fever, drenching sweats, backache, fatigue, sore throat, dyspnea, and cough. Within 2-3 days, the characteristic skin eruption develops. The lesions usually begin on the face and then spread in a centrifugal manner to the trunk and extremities, including the palms of the hands and soles of the feet. The lesions typically progress from macules to papules to vesicles to pustules. They then crust and scab over. An interesting additional finding is the presence of prominent lymphadenopathy behind the ear, beneath the mandible, in the neck, and in the groin.1
Several different illnesses must be considered in the differential diagnosis of monkeypox infection. They include measles, scabies, secondary syphilis, and medication-associated allergic reactions. However, the 2 conditions most likely to be confused with monkeypox are chickenpox (varicella) and smallpox. Lymphadenopathy is much more prominent in monkeypox compared with chickenpox. Moreover, with monkeypox, all lesions tend to be at the same stage of evolution as opposed to appearing in crops as they do in chickenpox. Smallpox would be extremely unlikely in the absence of a recognized laboratory accident or a bioterrorism incident.7
Diagnosis
The presumptive diagnosis of monkeypox infection is made primarily based on clinical examination. However, laboratory testing is indicated to definitively differentiate monkeypox from other orthopoxvirus infections such as varicella and smallpox.
In specialized laboratories that employ highly trained personnel and maintain strict safety precautions, the virus can be isolated in mammalian cell cultures. Electron microscopy is a valuable tool for identifying the characteristic brick-shaped poxvirus virions. Routine histologic examination of a lesion will show ballooning degeneration of keratinocytes, prominent spongiosis, dermal edema, and acute inflammation, although these findings are not unique to monkeypox.1
The Centers for Disease Control and Prevention (CDC) has developed serologic tests that detect immunoglobulin (Ig) M- and IgG-specific antibody. However, the most useful and practical diagnostic test is assessment of a skin scraping by polymerase chain reaction (PCR). This test is more sensitive than assessment of serum PCR.1
When the diagnosis of monkeypox is being considered, the clinician should coordinate testing through the local and state public health departments and through the CDC. Effective communication with all agencies will ensure that laboratory specimens are processed in a timely and efficient manner. The CDC website presents information on specimen collection.8
How do we manage monkeypox?
Prevention
The first step in prevention of infection is to isolate infected individuals until all lesions have dried and crusted over. Susceptible people should avoid close contact with skin lesions, respiratory and genital secretions, and bedding of patients who are infected.
The ultimate preventive measure, however, is vaccination of susceptible people either immediately before exposure (eg, military personnel, first responders, infection control investigators, health care workers) or immediately after exposure (general population). Older individuals who received the original smallpox vaccine likely have immunity to monkeypox infection. Unfortunately, very few women who currently are of reproductive age received this vaccine because its use was discontinued in the United States in the early 1970s. Therefore, the vast majority of our patients are uniquely susceptible to this infection and should be vaccinated if there is an outbreak of monkeypox in their locality.7,9
The current preferred vaccine for prevention of both smallpox and monkeypox is the Jynneos (Bavarian Nordic A/S) vaccine.10 This agent incorporates a replication-deficient live virus and does not pose the same risk for adverse events as the original versions of the smallpox vaccine. Jynneos is administered subcutaneously rather than by scarification. Two 0.5-mL doses, delivered 28 days apart, are required for optimal effect. The vaccine must be obtained from local and state health departments, in consultation with the CDC.7,9
There is very little published information on the safety of the Jynneos vaccine in pregnant or lactating women, although animal data are reassuring. Moreover, the dangers of monkeypox infection are significant, and in the event of an outbreak, vaccination of susceptible individuals, including pregnant women, is indicated.
- Monkeypox is a member of the orthopoxvirus genus and is closely related to the smallpox virus. It is a large, double-stranded, enveloped DNA virus.
- The virus is transmitted primarily by close contact with infected animals or other humans or by consumption of contaminated bushmeat.
- The infection evolves in 2 phases. The pre-eruptive phase is characterized by severe flu-like symptoms and signs. The eruptive phase is distinguished by a diffuse papular-vesicular rash.
- The most valuable test for confirming the diagnosis is a polymerase chain reaction test of a fresh skin lesion.
- In women who are pregnant, monkeypox has been associated with spontaneous abortion and fetal death.
- Three antiviral agents may be of value in treating infected patients: cidofovir, brincidofovir, and tecovirimat. Only the latter has an acceptable safety profile for women who are pregnant or lactating.
- The new nonreplicating smallpox vaccine Jynneos (Bavarian Nordic A/S) is of great value for pre- and post-exposure prophylaxis.
Continue to: Treatment...
Treatment
Infected pregnant women should receive acetaminophen 1,000 mg orally every 8 hours, to control fever and provide analgesia. An antihistamine such as diphenhydramine 25 mg orally every 6-8 hours, may be used to control pruritus and provide mild sedation. Adequate fluid intake and optimal nutrition should be encouraged. Skin lesions should be inspected regularly to detect signs of superimposed bacterial infections. Small, localized bacterial skin infections can be treated with topical application of mupirocin ointment 2%, 3 times daily for 7-14 days. For diffuse and more severe bacterial skin infections, a systemic antibiotic may be necessary. Reasonable choices include amoxicillin-clavulanate 875 mg/125 mg orally every 12 hours, or trimethoprim-sulfamethoxazole double strength 800 mg/160 mg orally every 12 hours.11 The latter agent should be avoided in the first trimester of pregnancy because of potential teratogenic effects.
Several specific agents are available through the CDC for treatment of orthopoxvirus infections such as smallpox and monkeypox. Information about these agents is summarized in the TABLE.12-16

Unique considerations in pregnancy
Because monkeypox is so rare, there is very little information about the effects of this infection in pregnant women. The report most commonly cited in the literature is that by Mbala et al, which was published in 2017.17 These authors described 4 pregnant patients in the Democratic Republic of Congo who contracted monkeypox infection over a 4-year period. All 4 women were hospitalized and treated with systemic antibiotics, antiparasitic medications, and analgesics. One patient delivered a healthy infant. Two women had spontaneous abortions in the first trimester. The fourth patient experienced a stillbirth at 22 weeks’ gestation. At postmortem examination, the fetus had diffuse cutaneous lesions, prominent hepatomegaly, and hydrops. No structural malformations were noted. The placenta demonstrated numerous punctate hemorrhages, and high concentrations of virus were recovered from the placenta and from fetal tissue.
Although the information on pregnancy outcome is quite limited, it seems clear that the virus can cross the placenta and cause adverse effects such as spontaneous abortion and fetal death. Accordingly, I think the following guidelines are a reasonable approach to a pregnant patient who has been exposed to monkeypox or who has developed manifestations of infection.3,7,9
- In the event of a community outbreak, bioterrorism event, or exposure to a person with suspected or confirmed monkeypox infection, the pregnant patient should receive the Jynneos vaccine.
- The pregnant patient should be isolated from any individual with suspected or confirmed monkeypox.
- If infection develops despite these measures, the patient should be treated with either tecovirimat or vaccinia immune globulin IV. Hospitalization may be necessary for seriously ill individuals.
- Within 2 weeks of infection, a comprehensive ultrasound examination should be performed to assess for structural abnormalities in the fetus.
- Subsequently, serial ultrasound examinations should be performed at intervals of 4-6 weeks to assess fetal growth and re-evaluate fetal anatomy.
- Following delivery, a detailed neonatal examination should be performed to assess for evidence of viral injury. Neonatal skin lesions and neonatal serum can be assessed by PCR for monkeypox virus. The newborn should be isolated from the mother until all the mother’s lesions have dried and crusted over.
CASE Resolved
Given the husband’s recent travel to Nigeria and consumption of bushmeat, he most likely has monkeypox. The infection can be spread from person to person by close contact; thus, his wife is at risk. The couple should isolate until all of his lesions have dried and crusted over. The woman also should receive the Jynneos vaccine. If she becomes symptomatic, she should be treated with tecovirimat or vaccinia immune globulin IV. ●

- Isaacs SN, Shenoy ES. Monkeypox. UpToDate. Updated June 28,2022. Accessed July 1, 2022. https://www.uptodate.com /contents/monkeypox?topicRef=8349&source=see_link
- Graham MB. Monkeypox. Medscape. Updated June 29, 2022. Accessed July 1, 2022. https://emedicine.medscape.com /article/1134714-overview.
- Khalil A, Samara A, O’Brien P, et al. Monkeypox and pregnancy: what do obstetricians need to know? Ultrasound Obstet Gynecol. 2022;60:22-27. doi:10.1002/uog.24968.
- World Health Organization. Monkeypox-United Kingdom of Great Britain and Northern Ireland. May 18, 2022. Accessed July 1, 2022. https://www.who.int/emergencies/diseaseoutbreak-news/item/2022-DON383.
- WHO reports two new monkeypox deaths, cases in new areas. Reuters. July 7, 2022. https://www.reuters.com/world /who-reports-two-new-monkeypox-deaths-2022-07-07/. Accessed July 19, 2022.
- World Health Organization. Multi-country monkeypox outbreak in non-endemic countries: update. May 29, 2022. Accessed July 1, 2022. https://www.who.int /emergencies/disease-outbreak-news/item/2022 -DON388#:~:text=Multi%2Dcountry%20monkeypox%20 outbreak%20in%20non%2Dendemic%20countries%3A%20 Update,-29%20May%202022&text=Since%2013%20 May%202022%2C%20monkeypox,Epidemiological%20 investigations%20are%20ongoing.
- Cono J, Cragan JD, Jamieson DJ, Rasmussen SA. Prophylaxis and treatment of pregnant women for emerging infections andbioterrorism emergencies. Emerg Infect Dis. 2006;12:16311637. doi:10.3201/eid1211.060618.
- Centers for Disease Control and Prevention. Preparation and collection of specimens. Reviewed June 29, 2022. Accessed July 6, 2022. https://www.cdc.gov/poxvirus /monkeypox/clinicians/prep-collection-specimens.html.
- Rao AK, Petersen BW, Whitehill F, et al. Monkeypox vaccination. MMWR Morb Mortal Wkly Rep. 2022;71:734-742. doi:10.15585/mmwr.mm7122e1.
- Smallpox and monkeypox vaccine, live, nonreplicating. Package insert. Bavarian Nordic A/S; 2021. Accessed July 1, 2022. https://www.fda.gov/media/131078/download.
- Duff P. Commonly used antibiotics in ObGyn practice. OBG Manag. 2022;34:29, 36-40. doi:10.12788/obgm.0191.
- Centers for Disease Control and Prevention. Treatment information for healthcare professionals: interim clinical guidance for the treatment of monkeypox. Reviewed June 17, 2022. Accessed July 1, 2022. https://www.cdc.gov/poxvirus /monkeypox/clinicians/treatment.html.
- Brincidofovir. Prescribing information. Chimerix, Inc.; 2021. Accessed July 1, 2022. https://www.accessdata.fda.gov /drugsatfda_docs/label/2021/214460s000,214461s000lbl.pdf.
- Cidofovir. Package insert. Gilead Sciences, Inc.; 2010. Accessed July 1, 2022. https://www.gilead.com/~/media /Files/pdfs/medicines/other/vistide/vistide.pdf.
- Tecovirimat. Prescribing information. Catalent Pharma Solutions; 2022. Accessed July 1, 2022. https://www.accessdata.fda.gov/drugsatfda_docs /label/2022/214518s000lbl.pdf.
- Vaccinia immune globulin IV. Prescribing information. Cangene Corporation; 2010. Accessed July 1, 2022. https: //www.fda.gov/media/77004/download.
- Mbala PK, Huggins JW, Riu-Rovira T, et al. Maternal and fetal outcomes among pregnant women with human monkeypox infection in the Democratic Republic of Congo. J Infect Dis. 2017;216:824-828. doi:10.1093/infdis/jix260.
- Isaacs SN, Shenoy ES. Monkeypox. UpToDate. Updated June 28,2022. Accessed July 1, 2022. https://www.uptodate.com /contents/monkeypox?topicRef=8349&source=see_link
- Graham MB. Monkeypox. Medscape. Updated June 29, 2022. Accessed July 1, 2022. https://emedicine.medscape.com /article/1134714-overview.
- Khalil A, Samara A, O’Brien P, et al. Monkeypox and pregnancy: what do obstetricians need to know? Ultrasound Obstet Gynecol. 2022;60:22-27. doi:10.1002/uog.24968.
- World Health Organization. Monkeypox-United Kingdom of Great Britain and Northern Ireland. May 18, 2022. Accessed July 1, 2022. https://www.who.int/emergencies/diseaseoutbreak-news/item/2022-DON383.
- WHO reports two new monkeypox deaths, cases in new areas. Reuters. July 7, 2022. https://www.reuters.com/world /who-reports-two-new-monkeypox-deaths-2022-07-07/. Accessed July 19, 2022.
- World Health Organization. Multi-country monkeypox outbreak in non-endemic countries: update. May 29, 2022. Accessed July 1, 2022. https://www.who.int /emergencies/disease-outbreak-news/item/2022 -DON388#:~:text=Multi%2Dcountry%20monkeypox%20 outbreak%20in%20non%2Dendemic%20countries%3A%20 Update,-29%20May%202022&text=Since%2013%20 May%202022%2C%20monkeypox,Epidemiological%20 investigations%20are%20ongoing.
- Cono J, Cragan JD, Jamieson DJ, Rasmussen SA. Prophylaxis and treatment of pregnant women for emerging infections andbioterrorism emergencies. Emerg Infect Dis. 2006;12:16311637. doi:10.3201/eid1211.060618.
- Centers for Disease Control and Prevention. Preparation and collection of specimens. Reviewed June 29, 2022. Accessed July 6, 2022. https://www.cdc.gov/poxvirus /monkeypox/clinicians/prep-collection-specimens.html.
- Rao AK, Petersen BW, Whitehill F, et al. Monkeypox vaccination. MMWR Morb Mortal Wkly Rep. 2022;71:734-742. doi:10.15585/mmwr.mm7122e1.
- Smallpox and monkeypox vaccine, live, nonreplicating. Package insert. Bavarian Nordic A/S; 2021. Accessed July 1, 2022. https://www.fda.gov/media/131078/download.
- Duff P. Commonly used antibiotics in ObGyn practice. OBG Manag. 2022;34:29, 36-40. doi:10.12788/obgm.0191.
- Centers for Disease Control and Prevention. Treatment information for healthcare professionals: interim clinical guidance for the treatment of monkeypox. Reviewed June 17, 2022. Accessed July 1, 2022. https://www.cdc.gov/poxvirus /monkeypox/clinicians/treatment.html.
- Brincidofovir. Prescribing information. Chimerix, Inc.; 2021. Accessed July 1, 2022. https://www.accessdata.fda.gov /drugsatfda_docs/label/2021/214460s000,214461s000lbl.pdf.
- Cidofovir. Package insert. Gilead Sciences, Inc.; 2010. Accessed July 1, 2022. https://www.gilead.com/~/media /Files/pdfs/medicines/other/vistide/vistide.pdf.
- Tecovirimat. Prescribing information. Catalent Pharma Solutions; 2022. Accessed July 1, 2022. https://www.accessdata.fda.gov/drugsatfda_docs /label/2022/214518s000lbl.pdf.
- Vaccinia immune globulin IV. Prescribing information. Cangene Corporation; 2010. Accessed July 1, 2022. https: //www.fda.gov/media/77004/download.
- Mbala PK, Huggins JW, Riu-Rovira T, et al. Maternal and fetal outcomes among pregnant women with human monkeypox infection in the Democratic Republic of Congo. J Infect Dis. 2017;216:824-828. doi:10.1093/infdis/jix260.
Single dose of HPV vaccine is ‘game changer,’ says WHO
The World Health Organization’s Strategic Advisory Group of Experts on Immunization (SAGE) has changed the recommendation for vaccines against human papillomavirus (HPV).
From the available evidence, SAGE has concluded that a single dose of vaccine offers solid protection against HPV, comparable to that achieved with two-dose schedules.
This could be a “game-changer for the prevention of the disease,” as it would allow “more doses of the life-saving jab reach more girls,” the WHO declared in a press release.
SAGE recommends updating HPV dose schedules as follows:
- One- or two-dose schedule for the primary target of girls aged 9-14 years.
- One- or two-dose schedule for young women aged 15-20.
- Two doses with a 6-month interval for women older than 21.
The HPV vaccine is highly effective for the prevention of HPV serotypes 16 and 18, which cause 70% of cases of cervical cancer, said Alejandro Cravioto, MD, PhD, SAGE chair, in a statement.
“SAGE urges all countries to introduce HPV vaccines and prioritize multi-age cohort catch up of missed and older cohorts of girls. These recommendations will enable more girls and women to be vaccinated and thus preventing them from having cervical cancer and all its consequences over the course of their lifetimes,” he added.
For individuals who are immunocompromised, including those with HIV, three doses of the vaccine should be given if feasible, and if not, then at least two doses. There is limited evidence regarding the efficacy of a single dose in this group, the advisory group noted.
Policy makers need to make changes
Now that the WHO has deemed that one dose of HPV vaccine is sufficient, policy makers should make changes, say experts in a recent editorial comment published in The Lancet Oncology.
“Policy makers should consider modifying their HPV immunization schedules for girls aged 9-14 years from a two-dose regimen to a one-dose regimen,” wrote Jeff D’Souza, PhD, Institute for Better Health, Trillium Health Partners, Mississauga, Ont., and David Nderitu, PhD, Egerton University, Nakuru County, Kenya.
Policy makers also need to consider reorienting their efforts on cervical cancer screening and treatment, and they should ensure that all girls globally have access to an effective HPV vaccination schedule, they add.
The editorialists also make a radical proposal.
Existing supply constraints of the HPV vaccine at the country level are expected to continue for the next 3 years, and the vast majority of new cervical cancer cases and related deaths occur in low- and middle-income countries (LMICs).
To overcome these problems, they suggest that “high-income countries that currently offer two-dose regimens to girls aged 9-14 years should consider opting for a one-dose vaccination schedule, and give any excess of vaccines to countries in greater need of them.”
Two doses in high-income countries
But it is unclear whether high-income countries are ready to move to a one-dose schedule.
Approached for comment, Maurie Markman, MD, president of medicine and science at Cancer Treatment Centers of America, Philadelphia, told this news organization that while he can’t say for certain, he suspects that the United States will be slower to accept this recommendation for a single dose of HPV vaccine “as a component of a ‘standard-of-care’ approach.”
However, it “might formally acknowledge that if an individual/parent will only accept a single vaccine dose (or ultimately refuses to return for a recommended second dose), this will be considered a favorable outcome, both for the individual and society.
“I do not know if regulatory bodies in the United States will accept the existing studies performed to address the one-dose vaccination strategy to rather dramatically change the approach in our country,” he said. “The issue would be that if a single dose was stated to be a clinically acceptable option in the United States, it would rapidly become the standard approach, and the regulators would want to be as certain as possible that this would not have a negative effect on what is now recognized to be a remarkably safe and effective cancer prevention effort.”
Another expert who was approached for comment, Stephanie V. Blank, MD, professor of gynecologic oncology at the Icahn School of Medicine at Mount Sinai, New York, said: “In higher-resourced countries, two doses are still preferred, as they are more effective than one.
“The modeling on which the SAGE recommendation is based is all from studies in LMICs and other modeling studies,” she added.
At present, the Centers for Disease Control and Prevention recommends a two-dose schedule of HPV vaccines for individuals who receive the first dose before their 15th birthday. The three-dose schedule is recommended for those who receive the first dose on or after their 15th birthday and for people with certain immunocompromising conditions.
Studies have shown that two doses of HPV vaccine given to children aged 9-14 years provide as good or better protection than three doses given to older adolescents or young adults.
But even with a two-dose schedule, the WHO reports that uptake of the vaccine has been slow, and coverage is much lower than their 90% target. In 2020, global coverage with two doses was only 13%.
Factors that have influenced the slow uptake and low coverage of HPV vaccines include supply challenges, programmatic challenges, and costs related to delivering a two-dose regimen to older girls who are not typically included in childhood vaccination programs. The relatively high cost of HPV vaccines has also been problematic, particularly for middle-income countries.
Trials of one-dose schedules
The one-dose vaccine schedule has garnered a lot of interest, with several studies showing efficacy.
The KEN SHE trial, based in Kenya, showed that a single dose of the HPV vaccine was highly effective at preventing oncogenic infection, rivaling the protection offered by multidose regimens. Vaccine efficacy was 97.5% (P < .001) against HPV 16/18 for both the bivalent and monovalent vaccines, which is “comparable to that seen in multidose vaccine trials,” the researchers noted.
A study in India found that efficacy against persistent HPV 16 and 18 infection among participants evaluable for the endpoint was 95.4% for the single dose, 93.1% for the two-dose schedule, and 93.3% for the three-dose series.
Commenting on this trial in India in a recent interview with this news organization, Geoffroy Canlorbe, MD, PhD, of the department of gynecologic and breast surgery and oncology, Pitié-Salpêtrière Hospital, Paris, said the findings from India would need “to be confirmed by other studies.” The results were nonetheless “excellent news for developing countries where there are challenges when it comes to access to vaccination.”
Speaking at the 45th Congress of the French Society for Colposcopy and Cervical and Vaginal Diseases, he emphasized that at this stage, the findings “cannot be extrapolated” to France. HPV vaccination coverage is low in France (it is estimated that the rate is 23.7%, placing the country 28th of 31 countries in Europe), and he recommended continuing with the two- or three-dose schedule for the time being.
“This poor coverage has nothing to do with health care–related logistical or organizational issues; instead, it has to do with people’s mistrust when it comes to vaccination. Here, people who get the first dose get the subsequent ones,” said Dr. Canlorbe. “The very fact of getting two to three doses allows the person’s body to increase the production of antibodies and get a longer-lasting response to the vaccine.”
Ethics of the vaccine
In their editorial, Dr. D’Souza and Dr. Nderitu note that there are ethical considerations with the HPV vaccine that can “help guide deliberations, covering nonmaleficence, beneficence, health equity, stewardship, and solidarity.”
It would be inequitable and unjustifiable, they write, to offer a two-dose regimen to girls aged 9-14 years without also introducing multi-age cohort catch-up campaigns or programs for women who do not have access. “When it comes to an effective HPV vaccination schedule, no woman or girl should be left behind,” they say.
To achieve the goal of eliminating cervical cancer, “countries must ensure that 90% of girls are vaccinated, 70% of women are screened, and 90% of women with precancerous lesions receive treatment and care,” they write. “Given resource constraints, particularly in low-middle income countries, policy makers have a responsibility to ensure that resources are used in an optimal manner that promotes the right to health of all individuals.”
Thus, countries that are lagging far behind in cervical cancer education, screening, and treatment should consider opting for a one-dose regimen for girls aged 9-14 years, as well as using additional resources to close the gap in these other areas.
Dr. Markman has relationships with Genentech, AstraZeneca, Celgene, Clovis, and Amgen; he is also a regular contributor to Medscape Oncology with the Markamn on Oncology video column. Dr. D’Souza and Dr. Nderitu have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The World Health Organization’s Strategic Advisory Group of Experts on Immunization (SAGE) has changed the recommendation for vaccines against human papillomavirus (HPV).
From the available evidence, SAGE has concluded that a single dose of vaccine offers solid protection against HPV, comparable to that achieved with two-dose schedules.
This could be a “game-changer for the prevention of the disease,” as it would allow “more doses of the life-saving jab reach more girls,” the WHO declared in a press release.
SAGE recommends updating HPV dose schedules as follows:
- One- or two-dose schedule for the primary target of girls aged 9-14 years.
- One- or two-dose schedule for young women aged 15-20.
- Two doses with a 6-month interval for women older than 21.
The HPV vaccine is highly effective for the prevention of HPV serotypes 16 and 18, which cause 70% of cases of cervical cancer, said Alejandro Cravioto, MD, PhD, SAGE chair, in a statement.
“SAGE urges all countries to introduce HPV vaccines and prioritize multi-age cohort catch up of missed and older cohorts of girls. These recommendations will enable more girls and women to be vaccinated and thus preventing them from having cervical cancer and all its consequences over the course of their lifetimes,” he added.
For individuals who are immunocompromised, including those with HIV, three doses of the vaccine should be given if feasible, and if not, then at least two doses. There is limited evidence regarding the efficacy of a single dose in this group, the advisory group noted.
Policy makers need to make changes
Now that the WHO has deemed that one dose of HPV vaccine is sufficient, policy makers should make changes, say experts in a recent editorial comment published in The Lancet Oncology.
“Policy makers should consider modifying their HPV immunization schedules for girls aged 9-14 years from a two-dose regimen to a one-dose regimen,” wrote Jeff D’Souza, PhD, Institute for Better Health, Trillium Health Partners, Mississauga, Ont., and David Nderitu, PhD, Egerton University, Nakuru County, Kenya.
Policy makers also need to consider reorienting their efforts on cervical cancer screening and treatment, and they should ensure that all girls globally have access to an effective HPV vaccination schedule, they add.
The editorialists also make a radical proposal.
Existing supply constraints of the HPV vaccine at the country level are expected to continue for the next 3 years, and the vast majority of new cervical cancer cases and related deaths occur in low- and middle-income countries (LMICs).
To overcome these problems, they suggest that “high-income countries that currently offer two-dose regimens to girls aged 9-14 years should consider opting for a one-dose vaccination schedule, and give any excess of vaccines to countries in greater need of them.”
Two doses in high-income countries
But it is unclear whether high-income countries are ready to move to a one-dose schedule.
Approached for comment, Maurie Markman, MD, president of medicine and science at Cancer Treatment Centers of America, Philadelphia, told this news organization that while he can’t say for certain, he suspects that the United States will be slower to accept this recommendation for a single dose of HPV vaccine “as a component of a ‘standard-of-care’ approach.”
However, it “might formally acknowledge that if an individual/parent will only accept a single vaccine dose (or ultimately refuses to return for a recommended second dose), this will be considered a favorable outcome, both for the individual and society.
“I do not know if regulatory bodies in the United States will accept the existing studies performed to address the one-dose vaccination strategy to rather dramatically change the approach in our country,” he said. “The issue would be that if a single dose was stated to be a clinically acceptable option in the United States, it would rapidly become the standard approach, and the regulators would want to be as certain as possible that this would not have a negative effect on what is now recognized to be a remarkably safe and effective cancer prevention effort.”
Another expert who was approached for comment, Stephanie V. Blank, MD, professor of gynecologic oncology at the Icahn School of Medicine at Mount Sinai, New York, said: “In higher-resourced countries, two doses are still preferred, as they are more effective than one.
“The modeling on which the SAGE recommendation is based is all from studies in LMICs and other modeling studies,” she added.
At present, the Centers for Disease Control and Prevention recommends a two-dose schedule of HPV vaccines for individuals who receive the first dose before their 15th birthday. The three-dose schedule is recommended for those who receive the first dose on or after their 15th birthday and for people with certain immunocompromising conditions.
Studies have shown that two doses of HPV vaccine given to children aged 9-14 years provide as good or better protection than three doses given to older adolescents or young adults.
But even with a two-dose schedule, the WHO reports that uptake of the vaccine has been slow, and coverage is much lower than their 90% target. In 2020, global coverage with two doses was only 13%.
Factors that have influenced the slow uptake and low coverage of HPV vaccines include supply challenges, programmatic challenges, and costs related to delivering a two-dose regimen to older girls who are not typically included in childhood vaccination programs. The relatively high cost of HPV vaccines has also been problematic, particularly for middle-income countries.
Trials of one-dose schedules
The one-dose vaccine schedule has garnered a lot of interest, with several studies showing efficacy.
The KEN SHE trial, based in Kenya, showed that a single dose of the HPV vaccine was highly effective at preventing oncogenic infection, rivaling the protection offered by multidose regimens. Vaccine efficacy was 97.5% (P < .001) against HPV 16/18 for both the bivalent and monovalent vaccines, which is “comparable to that seen in multidose vaccine trials,” the researchers noted.
A study in India found that efficacy against persistent HPV 16 and 18 infection among participants evaluable for the endpoint was 95.4% for the single dose, 93.1% for the two-dose schedule, and 93.3% for the three-dose series.
Commenting on this trial in India in a recent interview with this news organization, Geoffroy Canlorbe, MD, PhD, of the department of gynecologic and breast surgery and oncology, Pitié-Salpêtrière Hospital, Paris, said the findings from India would need “to be confirmed by other studies.” The results were nonetheless “excellent news for developing countries where there are challenges when it comes to access to vaccination.”
Speaking at the 45th Congress of the French Society for Colposcopy and Cervical and Vaginal Diseases, he emphasized that at this stage, the findings “cannot be extrapolated” to France. HPV vaccination coverage is low in France (it is estimated that the rate is 23.7%, placing the country 28th of 31 countries in Europe), and he recommended continuing with the two- or three-dose schedule for the time being.
“This poor coverage has nothing to do with health care–related logistical or organizational issues; instead, it has to do with people’s mistrust when it comes to vaccination. Here, people who get the first dose get the subsequent ones,” said Dr. Canlorbe. “The very fact of getting two to three doses allows the person’s body to increase the production of antibodies and get a longer-lasting response to the vaccine.”
Ethics of the vaccine
In their editorial, Dr. D’Souza and Dr. Nderitu note that there are ethical considerations with the HPV vaccine that can “help guide deliberations, covering nonmaleficence, beneficence, health equity, stewardship, and solidarity.”
It would be inequitable and unjustifiable, they write, to offer a two-dose regimen to girls aged 9-14 years without also introducing multi-age cohort catch-up campaigns or programs for women who do not have access. “When it comes to an effective HPV vaccination schedule, no woman or girl should be left behind,” they say.
To achieve the goal of eliminating cervical cancer, “countries must ensure that 90% of girls are vaccinated, 70% of women are screened, and 90% of women with precancerous lesions receive treatment and care,” they write. “Given resource constraints, particularly in low-middle income countries, policy makers have a responsibility to ensure that resources are used in an optimal manner that promotes the right to health of all individuals.”
Thus, countries that are lagging far behind in cervical cancer education, screening, and treatment should consider opting for a one-dose regimen for girls aged 9-14 years, as well as using additional resources to close the gap in these other areas.
Dr. Markman has relationships with Genentech, AstraZeneca, Celgene, Clovis, and Amgen; he is also a regular contributor to Medscape Oncology with the Markamn on Oncology video column. Dr. D’Souza and Dr. Nderitu have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The World Health Organization’s Strategic Advisory Group of Experts on Immunization (SAGE) has changed the recommendation for vaccines against human papillomavirus (HPV).
From the available evidence, SAGE has concluded that a single dose of vaccine offers solid protection against HPV, comparable to that achieved with two-dose schedules.
This could be a “game-changer for the prevention of the disease,” as it would allow “more doses of the life-saving jab reach more girls,” the WHO declared in a press release.
SAGE recommends updating HPV dose schedules as follows:
- One- or two-dose schedule for the primary target of girls aged 9-14 years.
- One- or two-dose schedule for young women aged 15-20.
- Two doses with a 6-month interval for women older than 21.
The HPV vaccine is highly effective for the prevention of HPV serotypes 16 and 18, which cause 70% of cases of cervical cancer, said Alejandro Cravioto, MD, PhD, SAGE chair, in a statement.
“SAGE urges all countries to introduce HPV vaccines and prioritize multi-age cohort catch up of missed and older cohorts of girls. These recommendations will enable more girls and women to be vaccinated and thus preventing them from having cervical cancer and all its consequences over the course of their lifetimes,” he added.
For individuals who are immunocompromised, including those with HIV, three doses of the vaccine should be given if feasible, and if not, then at least two doses. There is limited evidence regarding the efficacy of a single dose in this group, the advisory group noted.
Policy makers need to make changes
Now that the WHO has deemed that one dose of HPV vaccine is sufficient, policy makers should make changes, say experts in a recent editorial comment published in The Lancet Oncology.
“Policy makers should consider modifying their HPV immunization schedules for girls aged 9-14 years from a two-dose regimen to a one-dose regimen,” wrote Jeff D’Souza, PhD, Institute for Better Health, Trillium Health Partners, Mississauga, Ont., and David Nderitu, PhD, Egerton University, Nakuru County, Kenya.
Policy makers also need to consider reorienting their efforts on cervical cancer screening and treatment, and they should ensure that all girls globally have access to an effective HPV vaccination schedule, they add.
The editorialists also make a radical proposal.
Existing supply constraints of the HPV vaccine at the country level are expected to continue for the next 3 years, and the vast majority of new cervical cancer cases and related deaths occur in low- and middle-income countries (LMICs).
To overcome these problems, they suggest that “high-income countries that currently offer two-dose regimens to girls aged 9-14 years should consider opting for a one-dose vaccination schedule, and give any excess of vaccines to countries in greater need of them.”
Two doses in high-income countries
But it is unclear whether high-income countries are ready to move to a one-dose schedule.
Approached for comment, Maurie Markman, MD, president of medicine and science at Cancer Treatment Centers of America, Philadelphia, told this news organization that while he can’t say for certain, he suspects that the United States will be slower to accept this recommendation for a single dose of HPV vaccine “as a component of a ‘standard-of-care’ approach.”
However, it “might formally acknowledge that if an individual/parent will only accept a single vaccine dose (or ultimately refuses to return for a recommended second dose), this will be considered a favorable outcome, both for the individual and society.
“I do not know if regulatory bodies in the United States will accept the existing studies performed to address the one-dose vaccination strategy to rather dramatically change the approach in our country,” he said. “The issue would be that if a single dose was stated to be a clinically acceptable option in the United States, it would rapidly become the standard approach, and the regulators would want to be as certain as possible that this would not have a negative effect on what is now recognized to be a remarkably safe and effective cancer prevention effort.”
Another expert who was approached for comment, Stephanie V. Blank, MD, professor of gynecologic oncology at the Icahn School of Medicine at Mount Sinai, New York, said: “In higher-resourced countries, two doses are still preferred, as they are more effective than one.
“The modeling on which the SAGE recommendation is based is all from studies in LMICs and other modeling studies,” she added.
At present, the Centers for Disease Control and Prevention recommends a two-dose schedule of HPV vaccines for individuals who receive the first dose before their 15th birthday. The three-dose schedule is recommended for those who receive the first dose on or after their 15th birthday and for people with certain immunocompromising conditions.
Studies have shown that two doses of HPV vaccine given to children aged 9-14 years provide as good or better protection than three doses given to older adolescents or young adults.
But even with a two-dose schedule, the WHO reports that uptake of the vaccine has been slow, and coverage is much lower than their 90% target. In 2020, global coverage with two doses was only 13%.
Factors that have influenced the slow uptake and low coverage of HPV vaccines include supply challenges, programmatic challenges, and costs related to delivering a two-dose regimen to older girls who are not typically included in childhood vaccination programs. The relatively high cost of HPV vaccines has also been problematic, particularly for middle-income countries.
Trials of one-dose schedules
The one-dose vaccine schedule has garnered a lot of interest, with several studies showing efficacy.
The KEN SHE trial, based in Kenya, showed that a single dose of the HPV vaccine was highly effective at preventing oncogenic infection, rivaling the protection offered by multidose regimens. Vaccine efficacy was 97.5% (P < .001) against HPV 16/18 for both the bivalent and monovalent vaccines, which is “comparable to that seen in multidose vaccine trials,” the researchers noted.
A study in India found that efficacy against persistent HPV 16 and 18 infection among participants evaluable for the endpoint was 95.4% for the single dose, 93.1% for the two-dose schedule, and 93.3% for the three-dose series.
Commenting on this trial in India in a recent interview with this news organization, Geoffroy Canlorbe, MD, PhD, of the department of gynecologic and breast surgery and oncology, Pitié-Salpêtrière Hospital, Paris, said the findings from India would need “to be confirmed by other studies.” The results were nonetheless “excellent news for developing countries where there are challenges when it comes to access to vaccination.”
Speaking at the 45th Congress of the French Society for Colposcopy and Cervical and Vaginal Diseases, he emphasized that at this stage, the findings “cannot be extrapolated” to France. HPV vaccination coverage is low in France (it is estimated that the rate is 23.7%, placing the country 28th of 31 countries in Europe), and he recommended continuing with the two- or three-dose schedule for the time being.
“This poor coverage has nothing to do with health care–related logistical or organizational issues; instead, it has to do with people’s mistrust when it comes to vaccination. Here, people who get the first dose get the subsequent ones,” said Dr. Canlorbe. “The very fact of getting two to three doses allows the person’s body to increase the production of antibodies and get a longer-lasting response to the vaccine.”
Ethics of the vaccine
In their editorial, Dr. D’Souza and Dr. Nderitu note that there are ethical considerations with the HPV vaccine that can “help guide deliberations, covering nonmaleficence, beneficence, health equity, stewardship, and solidarity.”
It would be inequitable and unjustifiable, they write, to offer a two-dose regimen to girls aged 9-14 years without also introducing multi-age cohort catch-up campaigns or programs for women who do not have access. “When it comes to an effective HPV vaccination schedule, no woman or girl should be left behind,” they say.
To achieve the goal of eliminating cervical cancer, “countries must ensure that 90% of girls are vaccinated, 70% of women are screened, and 90% of women with precancerous lesions receive treatment and care,” they write. “Given resource constraints, particularly in low-middle income countries, policy makers have a responsibility to ensure that resources are used in an optimal manner that promotes the right to health of all individuals.”
Thus, countries that are lagging far behind in cervical cancer education, screening, and treatment should consider opting for a one-dose regimen for girls aged 9-14 years, as well as using additional resources to close the gap in these other areas.
Dr. Markman has relationships with Genentech, AstraZeneca, Celgene, Clovis, and Amgen; he is also a regular contributor to Medscape Oncology with the Markamn on Oncology video column. Dr. D’Souza and Dr. Nderitu have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
To gauge monkeypox spread, researchers eye cases in women
As cases of monkeypox continue to mount in the United States and abroad, infectious disease experts are closely monitoring one group of people in particular: women.
So far, the overwhelming majority of cases of the viral disease have been reported in men who have sex with men. But in recent days, officials have learned of a handful of cases in women – possibly indicating that the outbreak may be widening.
Researchers are keeping close tabs on the proportion of cases in women to “assess whether the outbreak is moving away” from networks of men who have sex with men, where most of the initial cases have been identified, according to a briefing from the UK Health Security Agency (UKHSA).
“There is insufficient evidence to support a change in the transmission dynamics,” the agency said. “However, over the last few weeks the proportion of female cases has been increasing, so this trend needs to be monitored closely.”
A global collaboration of researchers and clinicians recently described 528 cases of monkeypox in 16 countries – but none were in women.
Since data collection for that study ended in June, the research group has confirmed cases in women, said study coauthor John P. Thornhill, MD, PhD, consultant physician in sexual health and HIV and clinical senior lecturer at Barts Health NHS Trust and Queen Mary University of London.
“Cases in women have certainly been reported but are currently far less common,” Dr. Thornhill told this news organization.
Although infections in women have been outliers during the current outbreak, they can be severe when they do occur. Several women in England have been hospitalized with severe symptoms.
A similar pattern has been seen in New York City, where just one woman is among the 639 total cases, according to a July 21 report from the city’s health agency.
Researchers have recently published guidance on monkeypox for ob.gyns., maternal-fetal medicine subspecialists, and people who are pregnant or breastfeeding in anticipation of the possibility of more cases in women.
The Centers for Disease Control and Prevention advises that “pregnant, recently pregnant, and breastfeeding people should be prioritized for medical treatment” of monkeypox if needed.
One monkeypox vaccine, Jynneos, can be offered to people who are pregnant or breastfeeding and are otherwise eligible for vaccination on the basis of confirmed or likely contact with cases, ideally within 4 days of exposure. Some people at high risk for exposure, such as laboratory workers, may receive the vaccine preemptively.
Another vaccine, ACAM2000, is contraindicated in people who are pregnant or breastfeeding, according to the CDC.
Transmission dynamics
Investigators have not yet identified substantial spread of monkeypox beyond men who have sex with men, although transmission among household contacts, including women and children, has been reported.
Most initial infections during the current outbreak occurred during sexual activity. But monkeypox can spread through any close contact with skin lesions or body fluids and possibly through touching contaminated items like clothing or linens, according to the CDC. It also may spread from mother to child in utero.
Infected pets have been known to spread the disease as well. A multistate monkeypox outbreak in the United States in 2003 was linked to pet prairie dogs, including in childcare and school settings. That year, 55% of the 71 cases occurred in female patients.
More testing, higher positivity rates in men
Since May, more men than women in the United Kingdom have undergone testing for monkeypox, with 3,467 tests in men versus 447 tests in women. Among those tested, the positivity rate has been far higher in men than in women, 54% versus 2.2%, respectively.
As of July 20, about 0.65% of U.K. cases with known gender were in women. Two weeks prior, about 0.4% were in women.
In all, 13 monkeypox cases in England have been in women, and four had severe manifestations that required hospitalization, according to the UKHSA.
Globally, more than 16,000 monkeypox cases have been reported, according to the World Health Organization. The agency said that it plans to rename the disease to reduce stigma.
Monkeypox and pregnancy
Ob.gyns. are often on the “front line in terms of identifying people with infectious diseases,” said Denise J. Jamieson, MD, MPH, Emory University, Atlanta. Dr. Jamieson coauthored “A Primer on Monkeypox Virus for Obstetrician-Gynecologists,” published in Obstetrics & Gynecology.
“Obstetricians need to be aware of what infectious diseases are circulating and be aware of what is going on in the community,” she said.
With monkeypox, “it is anybody’s guess as to how widespread this is going to be,” Dr. Jamieson said.
“The initial monkeypox cases in the current outbreak have been predominately but not exclusively among men who have sex with men; enhanced transmission in this group may be facilitated by sexual activity and spread through complex sexual networks,” Dr. Thornhill said. “As the outbreak continues, we will likely see more monkeypox infections” outside that group.
“Those working in sexual health should have a high index of suspicion in all individuals presenting with genital and oral ulcers and those with proctitis,” he added.
During previous monkeypox outbreaks, the chain of household transmissions has been short, typically two or three people, said Chloe M. Orkin, MD, professor of HIV medicine at Queen Mary University of London. Dr. Orkin directs the Sexual Health and HIV All East Research (SHARE) Collaborative, which has worked to compile the international case series.
Though monkeypox has mainly been transmitted among men who have sex with men, not all identify as gay and some may also have female and nonbinary partners, Dr. Orkin said.
“Clinicians should bear this in mind when examining any person,” she said.
A version of this article first appeared on Medscape.com.
As cases of monkeypox continue to mount in the United States and abroad, infectious disease experts are closely monitoring one group of people in particular: women.
So far, the overwhelming majority of cases of the viral disease have been reported in men who have sex with men. But in recent days, officials have learned of a handful of cases in women – possibly indicating that the outbreak may be widening.
Researchers are keeping close tabs on the proportion of cases in women to “assess whether the outbreak is moving away” from networks of men who have sex with men, where most of the initial cases have been identified, according to a briefing from the UK Health Security Agency (UKHSA).
“There is insufficient evidence to support a change in the transmission dynamics,” the agency said. “However, over the last few weeks the proportion of female cases has been increasing, so this trend needs to be monitored closely.”
A global collaboration of researchers and clinicians recently described 528 cases of monkeypox in 16 countries – but none were in women.
Since data collection for that study ended in June, the research group has confirmed cases in women, said study coauthor John P. Thornhill, MD, PhD, consultant physician in sexual health and HIV and clinical senior lecturer at Barts Health NHS Trust and Queen Mary University of London.
“Cases in women have certainly been reported but are currently far less common,” Dr. Thornhill told this news organization.
Although infections in women have been outliers during the current outbreak, they can be severe when they do occur. Several women in England have been hospitalized with severe symptoms.
A similar pattern has been seen in New York City, where just one woman is among the 639 total cases, according to a July 21 report from the city’s health agency.
Researchers have recently published guidance on monkeypox for ob.gyns., maternal-fetal medicine subspecialists, and people who are pregnant or breastfeeding in anticipation of the possibility of more cases in women.
The Centers for Disease Control and Prevention advises that “pregnant, recently pregnant, and breastfeeding people should be prioritized for medical treatment” of monkeypox if needed.
One monkeypox vaccine, Jynneos, can be offered to people who are pregnant or breastfeeding and are otherwise eligible for vaccination on the basis of confirmed or likely contact with cases, ideally within 4 days of exposure. Some people at high risk for exposure, such as laboratory workers, may receive the vaccine preemptively.
Another vaccine, ACAM2000, is contraindicated in people who are pregnant or breastfeeding, according to the CDC.
Transmission dynamics
Investigators have not yet identified substantial spread of monkeypox beyond men who have sex with men, although transmission among household contacts, including women and children, has been reported.
Most initial infections during the current outbreak occurred during sexual activity. But monkeypox can spread through any close contact with skin lesions or body fluids and possibly through touching contaminated items like clothing or linens, according to the CDC. It also may spread from mother to child in utero.
Infected pets have been known to spread the disease as well. A multistate monkeypox outbreak in the United States in 2003 was linked to pet prairie dogs, including in childcare and school settings. That year, 55% of the 71 cases occurred in female patients.
More testing, higher positivity rates in men
Since May, more men than women in the United Kingdom have undergone testing for monkeypox, with 3,467 tests in men versus 447 tests in women. Among those tested, the positivity rate has been far higher in men than in women, 54% versus 2.2%, respectively.
As of July 20, about 0.65% of U.K. cases with known gender were in women. Two weeks prior, about 0.4% were in women.
In all, 13 monkeypox cases in England have been in women, and four had severe manifestations that required hospitalization, according to the UKHSA.
Globally, more than 16,000 monkeypox cases have been reported, according to the World Health Organization. The agency said that it plans to rename the disease to reduce stigma.
Monkeypox and pregnancy
Ob.gyns. are often on the “front line in terms of identifying people with infectious diseases,” said Denise J. Jamieson, MD, MPH, Emory University, Atlanta. Dr. Jamieson coauthored “A Primer on Monkeypox Virus for Obstetrician-Gynecologists,” published in Obstetrics & Gynecology.
“Obstetricians need to be aware of what infectious diseases are circulating and be aware of what is going on in the community,” she said.
With monkeypox, “it is anybody’s guess as to how widespread this is going to be,” Dr. Jamieson said.
“The initial monkeypox cases in the current outbreak have been predominately but not exclusively among men who have sex with men; enhanced transmission in this group may be facilitated by sexual activity and spread through complex sexual networks,” Dr. Thornhill said. “As the outbreak continues, we will likely see more monkeypox infections” outside that group.
“Those working in sexual health should have a high index of suspicion in all individuals presenting with genital and oral ulcers and those with proctitis,” he added.
During previous monkeypox outbreaks, the chain of household transmissions has been short, typically two or three people, said Chloe M. Orkin, MD, professor of HIV medicine at Queen Mary University of London. Dr. Orkin directs the Sexual Health and HIV All East Research (SHARE) Collaborative, which has worked to compile the international case series.
Though monkeypox has mainly been transmitted among men who have sex with men, not all identify as gay and some may also have female and nonbinary partners, Dr. Orkin said.
“Clinicians should bear this in mind when examining any person,” she said.
A version of this article first appeared on Medscape.com.
As cases of monkeypox continue to mount in the United States and abroad, infectious disease experts are closely monitoring one group of people in particular: women.
So far, the overwhelming majority of cases of the viral disease have been reported in men who have sex with men. But in recent days, officials have learned of a handful of cases in women – possibly indicating that the outbreak may be widening.
Researchers are keeping close tabs on the proportion of cases in women to “assess whether the outbreak is moving away” from networks of men who have sex with men, where most of the initial cases have been identified, according to a briefing from the UK Health Security Agency (UKHSA).
“There is insufficient evidence to support a change in the transmission dynamics,” the agency said. “However, over the last few weeks the proportion of female cases has been increasing, so this trend needs to be monitored closely.”
A global collaboration of researchers and clinicians recently described 528 cases of monkeypox in 16 countries – but none were in women.
Since data collection for that study ended in June, the research group has confirmed cases in women, said study coauthor John P. Thornhill, MD, PhD, consultant physician in sexual health and HIV and clinical senior lecturer at Barts Health NHS Trust and Queen Mary University of London.
“Cases in women have certainly been reported but are currently far less common,” Dr. Thornhill told this news organization.
Although infections in women have been outliers during the current outbreak, they can be severe when they do occur. Several women in England have been hospitalized with severe symptoms.
A similar pattern has been seen in New York City, where just one woman is among the 639 total cases, according to a July 21 report from the city’s health agency.
Researchers have recently published guidance on monkeypox for ob.gyns., maternal-fetal medicine subspecialists, and people who are pregnant or breastfeeding in anticipation of the possibility of more cases in women.
The Centers for Disease Control and Prevention advises that “pregnant, recently pregnant, and breastfeeding people should be prioritized for medical treatment” of monkeypox if needed.
One monkeypox vaccine, Jynneos, can be offered to people who are pregnant or breastfeeding and are otherwise eligible for vaccination on the basis of confirmed or likely contact with cases, ideally within 4 days of exposure. Some people at high risk for exposure, such as laboratory workers, may receive the vaccine preemptively.
Another vaccine, ACAM2000, is contraindicated in people who are pregnant or breastfeeding, according to the CDC.
Transmission dynamics
Investigators have not yet identified substantial spread of monkeypox beyond men who have sex with men, although transmission among household contacts, including women and children, has been reported.
Most initial infections during the current outbreak occurred during sexual activity. But monkeypox can spread through any close contact with skin lesions or body fluids and possibly through touching contaminated items like clothing or linens, according to the CDC. It also may spread from mother to child in utero.
Infected pets have been known to spread the disease as well. A multistate monkeypox outbreak in the United States in 2003 was linked to pet prairie dogs, including in childcare and school settings. That year, 55% of the 71 cases occurred in female patients.
More testing, higher positivity rates in men
Since May, more men than women in the United Kingdom have undergone testing for monkeypox, with 3,467 tests in men versus 447 tests in women. Among those tested, the positivity rate has been far higher in men than in women, 54% versus 2.2%, respectively.
As of July 20, about 0.65% of U.K. cases with known gender were in women. Two weeks prior, about 0.4% were in women.
In all, 13 monkeypox cases in England have been in women, and four had severe manifestations that required hospitalization, according to the UKHSA.
Globally, more than 16,000 monkeypox cases have been reported, according to the World Health Organization. The agency said that it plans to rename the disease to reduce stigma.
Monkeypox and pregnancy
Ob.gyns. are often on the “front line in terms of identifying people with infectious diseases,” said Denise J. Jamieson, MD, MPH, Emory University, Atlanta. Dr. Jamieson coauthored “A Primer on Monkeypox Virus for Obstetrician-Gynecologists,” published in Obstetrics & Gynecology.
“Obstetricians need to be aware of what infectious diseases are circulating and be aware of what is going on in the community,” she said.
With monkeypox, “it is anybody’s guess as to how widespread this is going to be,” Dr. Jamieson said.
“The initial monkeypox cases in the current outbreak have been predominately but not exclusively among men who have sex with men; enhanced transmission in this group may be facilitated by sexual activity and spread through complex sexual networks,” Dr. Thornhill said. “As the outbreak continues, we will likely see more monkeypox infections” outside that group.
“Those working in sexual health should have a high index of suspicion in all individuals presenting with genital and oral ulcers and those with proctitis,” he added.
During previous monkeypox outbreaks, the chain of household transmissions has been short, typically two or three people, said Chloe M. Orkin, MD, professor of HIV medicine at Queen Mary University of London. Dr. Orkin directs the Sexual Health and HIV All East Research (SHARE) Collaborative, which has worked to compile the international case series.
Though monkeypox has mainly been transmitted among men who have sex with men, not all identify as gay and some may also have female and nonbinary partners, Dr. Orkin said.
“Clinicians should bear this in mind when examining any person,” she said.
A version of this article first appeared on Medscape.com.
Hormone therapy didn’t increase recurrence or mortality in women treated for breast cancer
Hormone therapy did not increase mortality in postmenopausal women treated for early-stage estrogen receptor–positive breast cancer, but, in longitudinal data from Denmark, there was a recurrence risk with vaginal estrogen therapy among those treated with aromatase inhibitors.
Genitourinary syndrome of menopause (GSM) – including vaginal dryness, burning, and urinary incontinence – is common in women treated for breast cancer. Adjuvant endocrine therapy, particularly aromatase inhibitors, can aggravate these symptoms. Both local and systemic estrogen therapy are recommended for alleviating GSM symptoms in healthy women, but concerns have been raised about their use in women with breast cancer. Previous studies examining this have suggested possible risks for breast cancer recurrence, but those studies have had several limitations including small samples and short follow-up, particularly for vaginal estrogen therapy.
In the new study, from a national Danish cohort of 8,461 postmenopausal women diagnosed between 1997 and 2004 and treated for early-stage invasive estrogen receptor–positive nonmetastatic breast cancer, neither systemic menopausal hormone therapy (MHT) nor local vaginal estrogen therapy (VET) were associated with an overall increased risk for either breast cancer recurrence or mortality. However, in the subset who had received an aromatase inhibitor – with or without tamoxifen – there was a statistically significant increased risk for breast cancer recurrence, but not mortality.
The results were published in the Journal of the National Cancer Institute.
“The data are reassuring for the majority of women with no adjuvant therapy or tamoxifen. But for those using adjuvant aromatase inhibitors, there might be a small risk,” study lead author Søren Cold, MD, PhD, senior oncologist in the department of oncology at Odense (Denmark) University Hospital, Odense, said in an interview.
Moreover, Dr. Cold noted, while this study didn’t find an increased recurrence risk with MHT for women taking aromatase inhibitors, other studies have. One in particular was stopped because of harm. The reason for the difference here is likely that the previous sample was small – just 133 women.
“Our study is mainly focusing on the use of vaginal estrogen. We had so few patients using systemic menopausal hormone therapy, those data don’t mean much. ... The risk with systemic therapy has been established. The vaginal use hasn’t been thoroughly studied before,” he noted.
Breast cancer recurrence elevated with VET and aromatase inhibitors
The study pool was 9,710 women who underwent complete resection for estrogen-positive breast cancer and were all allocated to 5 years of adjuvant endocrine treatment or no adjuvant treatment, according to guidelines. Overall, 3,112 received no adjuvant endocrine treatment, 2,007 were treated with tamoxifen only, 403 with an aromatase inhibitor, and 2,939 with a sequence of tamoxifen and an aromatase inhibitor.
After exclusion of 1,249 who had received VET or MHT prior to breast cancer diagnosis, there were 6,391 not prescribed any estrogen hormonal treatment, 1,957 prescribed VET, and 133 prescribed MHT with or without VET.
During an estimated median 9.8 years’ follow-up, 1,333 women (16%) had a breast cancer recurrence. Of those, 111 had received VET, 16 MHT, and 1,206 neither. Compared with those receiving no hormonal treatment, the adjusted risk of recurrence was similar for the VET users (hazard ratio, 1.08; 95% confidence interval, 0.89-1.32).
However, there was an increased risk for recurrence associated with initiating VET during aromatase inhibitor treatment (HR, 1.39, 95% CI, 1.04-1.85). For women receiving MHT, the adjusted relative risk of recurrence with aromatase inhibitors wasn’t significant (HR, 1.05; 95% CI, 0.62-1.78).
Overall, compared with women who never used hormonal treatment, the absolute 10-year breast cancer recurrence risk was 19.2% for never-users of VET or MHT, 15.4% in VET users, and 17.1% in MHT users.
No differences found for mortality
Of the 8,461 women in the study, 40% (3,370) died during an estimated median follow-up of 15.2 years. Of those, 497 had received VET, 47 MHT, and 2,826 neither. Compared with the never-users of estrogen therapy, the adjusted HR for overall survival in VET users was 0.78 (95% CI, 0.71-0.87). The analysis stratified by adjuvant endocrine therapy didn’t show an increase in VET users by use of aromatase inhibitors (aHR, 0.94, 95% CI, 0.70-1.26). The same was found for women prescribed MHT, compared with never-users (aHR, 0.94; 95% CI, 0.70-1.26).
Never-users of VET or MHT had an absolute 10-year overall survival of 73.8% versus 79.5% and 80.5% among the women who used VET or MHT, respectively.
Asked to comment, Nanette Santoro, MD, professor and E. Stewart Taylor Chair of Obstetrics & Gynecology at the University of Colorado at Denver, Aurora, said in an interview: “It is important to look at this issue. These findings raise but don’t answer the question that vaginal estradiol may not be as safe as we hope it is for women with breast cancer using an aromatase inhibitor.”
However, she also pointed out that “the overall increase in risk is not enormous; mortality risk was not increased. Women need to consider that there may be some risk associated with this option in their decision making about taking it. Having a satisfying sex life is also important for many women! It is really compassionate use for quality of life, so there is always that unknown element of risk in the discussion. That unknown risk has to be balanced against the benefit that the estrogen provides.”
And, Dr. Santoro also noted that the use of prescription data poses limitations. “It cannot tell us what was going on in the minds of the patient and the prescriber. There may be differences in the prescriber’s impression of the patient’s risk of recurrence that influenced the decision to provide a prescription. ... Women using AIs [aromatase inhibitors] often get pretty severe vaginal dryness symptoms and may need more estrogen to be comfortable with intercourse, but we really cannot tell this from what is in this paper.”
Indeed, Dr. Cold said: “We admit it’s not a randomized study, but we’ve done what was possible to take [confounding] factors into account, including age, tumor size, nodal status, histology, and comorbidities.”
He suggested that a potential therapeutic approach to reducing the recurrence risk might be to switch VET-treated women to tamoxifen after 2-3 years of aromatase inhibitors.
This work was supported by Breast Friends, a part of the Danish Cancer Society. Dr. Cold received support from Breast Friends for the current study. Some of the other coauthors have pharmaceutical company disclosures. Dr. Santoro is a member of the scientific advisory boards for Astellas, Menogenix, Que Oncology, and Amazon Ember, and is a consultant for Ansh Labs.
Hormone therapy did not increase mortality in postmenopausal women treated for early-stage estrogen receptor–positive breast cancer, but, in longitudinal data from Denmark, there was a recurrence risk with vaginal estrogen therapy among those treated with aromatase inhibitors.
Genitourinary syndrome of menopause (GSM) – including vaginal dryness, burning, and urinary incontinence – is common in women treated for breast cancer. Adjuvant endocrine therapy, particularly aromatase inhibitors, can aggravate these symptoms. Both local and systemic estrogen therapy are recommended for alleviating GSM symptoms in healthy women, but concerns have been raised about their use in women with breast cancer. Previous studies examining this have suggested possible risks for breast cancer recurrence, but those studies have had several limitations including small samples and short follow-up, particularly for vaginal estrogen therapy.
In the new study, from a national Danish cohort of 8,461 postmenopausal women diagnosed between 1997 and 2004 and treated for early-stage invasive estrogen receptor–positive nonmetastatic breast cancer, neither systemic menopausal hormone therapy (MHT) nor local vaginal estrogen therapy (VET) were associated with an overall increased risk for either breast cancer recurrence or mortality. However, in the subset who had received an aromatase inhibitor – with or without tamoxifen – there was a statistically significant increased risk for breast cancer recurrence, but not mortality.
The results were published in the Journal of the National Cancer Institute.
“The data are reassuring for the majority of women with no adjuvant therapy or tamoxifen. But for those using adjuvant aromatase inhibitors, there might be a small risk,” study lead author Søren Cold, MD, PhD, senior oncologist in the department of oncology at Odense (Denmark) University Hospital, Odense, said in an interview.
Moreover, Dr. Cold noted, while this study didn’t find an increased recurrence risk with MHT for women taking aromatase inhibitors, other studies have. One in particular was stopped because of harm. The reason for the difference here is likely that the previous sample was small – just 133 women.
“Our study is mainly focusing on the use of vaginal estrogen. We had so few patients using systemic menopausal hormone therapy, those data don’t mean much. ... The risk with systemic therapy has been established. The vaginal use hasn’t been thoroughly studied before,” he noted.
Breast cancer recurrence elevated with VET and aromatase inhibitors
The study pool was 9,710 women who underwent complete resection for estrogen-positive breast cancer and were all allocated to 5 years of adjuvant endocrine treatment or no adjuvant treatment, according to guidelines. Overall, 3,112 received no adjuvant endocrine treatment, 2,007 were treated with tamoxifen only, 403 with an aromatase inhibitor, and 2,939 with a sequence of tamoxifen and an aromatase inhibitor.
After exclusion of 1,249 who had received VET or MHT prior to breast cancer diagnosis, there were 6,391 not prescribed any estrogen hormonal treatment, 1,957 prescribed VET, and 133 prescribed MHT with or without VET.
During an estimated median 9.8 years’ follow-up, 1,333 women (16%) had a breast cancer recurrence. Of those, 111 had received VET, 16 MHT, and 1,206 neither. Compared with those receiving no hormonal treatment, the adjusted risk of recurrence was similar for the VET users (hazard ratio, 1.08; 95% confidence interval, 0.89-1.32).
However, there was an increased risk for recurrence associated with initiating VET during aromatase inhibitor treatment (HR, 1.39, 95% CI, 1.04-1.85). For women receiving MHT, the adjusted relative risk of recurrence with aromatase inhibitors wasn’t significant (HR, 1.05; 95% CI, 0.62-1.78).
Overall, compared with women who never used hormonal treatment, the absolute 10-year breast cancer recurrence risk was 19.2% for never-users of VET or MHT, 15.4% in VET users, and 17.1% in MHT users.
No differences found for mortality
Of the 8,461 women in the study, 40% (3,370) died during an estimated median follow-up of 15.2 years. Of those, 497 had received VET, 47 MHT, and 2,826 neither. Compared with the never-users of estrogen therapy, the adjusted HR for overall survival in VET users was 0.78 (95% CI, 0.71-0.87). The analysis stratified by adjuvant endocrine therapy didn’t show an increase in VET users by use of aromatase inhibitors (aHR, 0.94, 95% CI, 0.70-1.26). The same was found for women prescribed MHT, compared with never-users (aHR, 0.94; 95% CI, 0.70-1.26).
Never-users of VET or MHT had an absolute 10-year overall survival of 73.8% versus 79.5% and 80.5% among the women who used VET or MHT, respectively.
Asked to comment, Nanette Santoro, MD, professor and E. Stewart Taylor Chair of Obstetrics & Gynecology at the University of Colorado at Denver, Aurora, said in an interview: “It is important to look at this issue. These findings raise but don’t answer the question that vaginal estradiol may not be as safe as we hope it is for women with breast cancer using an aromatase inhibitor.”
However, she also pointed out that “the overall increase in risk is not enormous; mortality risk was not increased. Women need to consider that there may be some risk associated with this option in their decision making about taking it. Having a satisfying sex life is also important for many women! It is really compassionate use for quality of life, so there is always that unknown element of risk in the discussion. That unknown risk has to be balanced against the benefit that the estrogen provides.”
And, Dr. Santoro also noted that the use of prescription data poses limitations. “It cannot tell us what was going on in the minds of the patient and the prescriber. There may be differences in the prescriber’s impression of the patient’s risk of recurrence that influenced the decision to provide a prescription. ... Women using AIs [aromatase inhibitors] often get pretty severe vaginal dryness symptoms and may need more estrogen to be comfortable with intercourse, but we really cannot tell this from what is in this paper.”
Indeed, Dr. Cold said: “We admit it’s not a randomized study, but we’ve done what was possible to take [confounding] factors into account, including age, tumor size, nodal status, histology, and comorbidities.”
He suggested that a potential therapeutic approach to reducing the recurrence risk might be to switch VET-treated women to tamoxifen after 2-3 years of aromatase inhibitors.
This work was supported by Breast Friends, a part of the Danish Cancer Society. Dr. Cold received support from Breast Friends for the current study. Some of the other coauthors have pharmaceutical company disclosures. Dr. Santoro is a member of the scientific advisory boards for Astellas, Menogenix, Que Oncology, and Amazon Ember, and is a consultant for Ansh Labs.
Hormone therapy did not increase mortality in postmenopausal women treated for early-stage estrogen receptor–positive breast cancer, but, in longitudinal data from Denmark, there was a recurrence risk with vaginal estrogen therapy among those treated with aromatase inhibitors.
Genitourinary syndrome of menopause (GSM) – including vaginal dryness, burning, and urinary incontinence – is common in women treated for breast cancer. Adjuvant endocrine therapy, particularly aromatase inhibitors, can aggravate these symptoms. Both local and systemic estrogen therapy are recommended for alleviating GSM symptoms in healthy women, but concerns have been raised about their use in women with breast cancer. Previous studies examining this have suggested possible risks for breast cancer recurrence, but those studies have had several limitations including small samples and short follow-up, particularly for vaginal estrogen therapy.
In the new study, from a national Danish cohort of 8,461 postmenopausal women diagnosed between 1997 and 2004 and treated for early-stage invasive estrogen receptor–positive nonmetastatic breast cancer, neither systemic menopausal hormone therapy (MHT) nor local vaginal estrogen therapy (VET) were associated with an overall increased risk for either breast cancer recurrence or mortality. However, in the subset who had received an aromatase inhibitor – with or without tamoxifen – there was a statistically significant increased risk for breast cancer recurrence, but not mortality.
The results were published in the Journal of the National Cancer Institute.
“The data are reassuring for the majority of women with no adjuvant therapy or tamoxifen. But for those using adjuvant aromatase inhibitors, there might be a small risk,” study lead author Søren Cold, MD, PhD, senior oncologist in the department of oncology at Odense (Denmark) University Hospital, Odense, said in an interview.
Moreover, Dr. Cold noted, while this study didn’t find an increased recurrence risk with MHT for women taking aromatase inhibitors, other studies have. One in particular was stopped because of harm. The reason for the difference here is likely that the previous sample was small – just 133 women.
“Our study is mainly focusing on the use of vaginal estrogen. We had so few patients using systemic menopausal hormone therapy, those data don’t mean much. ... The risk with systemic therapy has been established. The vaginal use hasn’t been thoroughly studied before,” he noted.
Breast cancer recurrence elevated with VET and aromatase inhibitors
The study pool was 9,710 women who underwent complete resection for estrogen-positive breast cancer and were all allocated to 5 years of adjuvant endocrine treatment or no adjuvant treatment, according to guidelines. Overall, 3,112 received no adjuvant endocrine treatment, 2,007 were treated with tamoxifen only, 403 with an aromatase inhibitor, and 2,939 with a sequence of tamoxifen and an aromatase inhibitor.
After exclusion of 1,249 who had received VET or MHT prior to breast cancer diagnosis, there were 6,391 not prescribed any estrogen hormonal treatment, 1,957 prescribed VET, and 133 prescribed MHT with or without VET.
During an estimated median 9.8 years’ follow-up, 1,333 women (16%) had a breast cancer recurrence. Of those, 111 had received VET, 16 MHT, and 1,206 neither. Compared with those receiving no hormonal treatment, the adjusted risk of recurrence was similar for the VET users (hazard ratio, 1.08; 95% confidence interval, 0.89-1.32).
However, there was an increased risk for recurrence associated with initiating VET during aromatase inhibitor treatment (HR, 1.39, 95% CI, 1.04-1.85). For women receiving MHT, the adjusted relative risk of recurrence with aromatase inhibitors wasn’t significant (HR, 1.05; 95% CI, 0.62-1.78).
Overall, compared with women who never used hormonal treatment, the absolute 10-year breast cancer recurrence risk was 19.2% for never-users of VET or MHT, 15.4% in VET users, and 17.1% in MHT users.
No differences found for mortality
Of the 8,461 women in the study, 40% (3,370) died during an estimated median follow-up of 15.2 years. Of those, 497 had received VET, 47 MHT, and 2,826 neither. Compared with the never-users of estrogen therapy, the adjusted HR for overall survival in VET users was 0.78 (95% CI, 0.71-0.87). The analysis stratified by adjuvant endocrine therapy didn’t show an increase in VET users by use of aromatase inhibitors (aHR, 0.94, 95% CI, 0.70-1.26). The same was found for women prescribed MHT, compared with never-users (aHR, 0.94; 95% CI, 0.70-1.26).
Never-users of VET or MHT had an absolute 10-year overall survival of 73.8% versus 79.5% and 80.5% among the women who used VET or MHT, respectively.
Asked to comment, Nanette Santoro, MD, professor and E. Stewart Taylor Chair of Obstetrics & Gynecology at the University of Colorado at Denver, Aurora, said in an interview: “It is important to look at this issue. These findings raise but don’t answer the question that vaginal estradiol may not be as safe as we hope it is for women with breast cancer using an aromatase inhibitor.”
However, she also pointed out that “the overall increase in risk is not enormous; mortality risk was not increased. Women need to consider that there may be some risk associated with this option in their decision making about taking it. Having a satisfying sex life is also important for many women! It is really compassionate use for quality of life, so there is always that unknown element of risk in the discussion. That unknown risk has to be balanced against the benefit that the estrogen provides.”
And, Dr. Santoro also noted that the use of prescription data poses limitations. “It cannot tell us what was going on in the minds of the patient and the prescriber. There may be differences in the prescriber’s impression of the patient’s risk of recurrence that influenced the decision to provide a prescription. ... Women using AIs [aromatase inhibitors] often get pretty severe vaginal dryness symptoms and may need more estrogen to be comfortable with intercourse, but we really cannot tell this from what is in this paper.”
Indeed, Dr. Cold said: “We admit it’s not a randomized study, but we’ve done what was possible to take [confounding] factors into account, including age, tumor size, nodal status, histology, and comorbidities.”
He suggested that a potential therapeutic approach to reducing the recurrence risk might be to switch VET-treated women to tamoxifen after 2-3 years of aromatase inhibitors.
This work was supported by Breast Friends, a part of the Danish Cancer Society. Dr. Cold received support from Breast Friends for the current study. Some of the other coauthors have pharmaceutical company disclosures. Dr. Santoro is a member of the scientific advisory boards for Astellas, Menogenix, Que Oncology, and Amazon Ember, and is a consultant for Ansh Labs.
FROM THE JOURNAL OF THE NATIONAL CANCER INSTITUTE
Some have heavier periods after COVID vaccine
Many women who got a COVID-19 vaccine have reported heavier bleeding during their periods since they had the shots.
A team of researchers investigated the trend and set out to find out who among the vaccinated were more likely to experience the menstruation changes.
The researchers were led by Katharine M.N. Lee, PhD, MS, of the division of public health sciences at Washington University in St. Louis. Their findings were published ahead of print in Science Advances.
The investigators analyzed more than 139,000 responses from an online survey from both currently and formerly menstruating women.
They found that, among people who have regular periods, about the same percentage had heavier bleeding after they got a COVID vaccine as had no change in bleeding after the vaccine (44% vs. 42%, respectively).
“A much smaller portion had lighter periods,” they write.
The phenomenon has been difficult to study because questions about changes in menstruation are not a standard part of vaccine trials.
Date of last period is often tracked in clinical trials to make sure a participant is not pregnant, but the questions about periods often stop there.
Additionally, periods are different for everyone and can be influenced by all sorts of environmental factors, so making associations regarding exposures is problematic.
No changes found to fertility
The authors emphasized that, generally, changes to menstrual bleeding are not uncommon nor dangerous. They also emphasized that the changes in bleeding don’t mean changes to fertility.
The uterine reproductive system is flexible when the body is under stress, they note.
“We know that running a marathon may influence hormone concentrations in the short term while not rendering that person infertile,” the authors write.
However, they acknowledge that investigating these reports is critical in building trust in medicine.
This report includes information that hasn’t been available through the clinical trial follow-up process.
For instance, the authors write, “To the best of our knowledge, our work is the first to examine breakthrough bleeding after vaccination in either pre- or postmenopausal people.”
Reports of changes to periods after vaccination started emerging in 2021. But without data, reports were largely dismissed, fueling criticism from those waging campaigns against COVID vaccines.
Dr. Lee and colleagues gathered data from those who responded to the online survey and detailed some trends.
People who were bleeding more heavily after vaccination were more likely to be older, Hispanic, had vaccine side effects of fever and fatigue, had been pregnant at some point, or had given birth.
People with regular periods who had endometriosis, prolonged bleeding during their periods, polycystic ovarian syndrome (PCOS) or fibroids were also more likely to have increased bleeding after a COVID vaccine.
Breakthrough bleeding
For people who don’t menstruate, but have not reached menopause, breakthrough bleeding happened more often in women who had been pregnant and/or had given birth.
Among respondents who were postmenopausal, breakthrough bleeding happened more often in younger people and/or those who are Hispanic.
More than a third of the respondents (39%) who use gender-affirming hormones that eliminate menstruation reported breakthrough bleeding after vaccination.
The majority of premenopausal people on long-acting, reversible contraception (71%) and the majority of postmenopausal respondents (66%) had breakthrough bleeding as well.
The authors note that you can’t compare the percentages who report these experiences in the survey with the incidence of those who would experience changes in menstrual bleeding in the general population.
The nature of the online survey means it may be naturally biased because the people who responded may be more often those who noted some change in their own menstrual experiences, particularly if that involved discomfort, pain, or fear.
Researchers also acknowledge that Black, Indigenous, Latinx, and other respondents of color are underrepresented in this research and that represents a limitation in the work.
Alison Edelman, MD, MPH, with the department of obstetrics and gynecology at Oregon Health & Science University in Portland, was not involved with Dr. Lee and associates’ study but has also studied the relationship between COVID vaccines and menstruation.
Her team’s study found that COVID vaccination is associated with a small change in time between periods but not length of periods.
She said about the work by Dr. Lee and colleagues, “This work really elevates the voices of the public and what they’re experiencing.”
The association makes sense, Dr. Edelman says, in that the reproductive system and the immune system talk to each other and inflammation in the immune system is going to be noticed by the system governing periods.
Lack of data on the relationship between exposures and menstruation didn’t start with COVID. “There has been a signal in the population before with other vaccines that’s been dismissed,” she said.
Tracking menstruation information in clinical trials can help physicians counsel women on what may be coming with any vaccine and alleviate fears and vaccine hesitancy, Dr. Edelman explained. It can also help vaccine developers know what to include in information about their product.
“When you are counseled about what to expect, it’s not as scary. That provides trust in the system,” she said. She likened it to original lack of data on whether COVID-19 vaccines would affect pregnancy.
“We have great science now that COVID vaccine does not affect fertility and [vaccine] does not impact pregnancy.”
Another important aspect of this paper is that it included subgroups not studied before regarding menstruation and breakthrough bleeding, such as those taking gender-affirming hormones, she added.
Menstruation has been often overlooked as important in clinical trial exposures but Dr. Edelman hopes this recent attention and question will escalate and prompt more research.
“I’m hoping with the immense outpouring from the public about how important this is, that future studies will look at this a little bit better,” she says.
She said when the National Institutes of Health opened up funding for trials on COVID-19 vaccines and menstruation, researchers got flooded with requests from women to share their stories.
“As a researcher – I’ve been doing research for over 20 years – that’s not something that usually happens. I would love to have that happen for every research project.”
The authors and Dr. Edelman declare that they have no competing interests. This research was supported in part by the University of Illinois Beckman Institute for Advanced Science and Technology, the University of Illinois Interdisciplinary Health Sciences Institute, the National Institutes of Health, the Foundation for Barnes-Jewish Hospital, and the Siteman Cancer Center.
Many women who got a COVID-19 vaccine have reported heavier bleeding during their periods since they had the shots.
A team of researchers investigated the trend and set out to find out who among the vaccinated were more likely to experience the menstruation changes.
The researchers were led by Katharine M.N. Lee, PhD, MS, of the division of public health sciences at Washington University in St. Louis. Their findings were published ahead of print in Science Advances.
The investigators analyzed more than 139,000 responses from an online survey from both currently and formerly menstruating women.
They found that, among people who have regular periods, about the same percentage had heavier bleeding after they got a COVID vaccine as had no change in bleeding after the vaccine (44% vs. 42%, respectively).
“A much smaller portion had lighter periods,” they write.
The phenomenon has been difficult to study because questions about changes in menstruation are not a standard part of vaccine trials.
Date of last period is often tracked in clinical trials to make sure a participant is not pregnant, but the questions about periods often stop there.
Additionally, periods are different for everyone and can be influenced by all sorts of environmental factors, so making associations regarding exposures is problematic.
No changes found to fertility
The authors emphasized that, generally, changes to menstrual bleeding are not uncommon nor dangerous. They also emphasized that the changes in bleeding don’t mean changes to fertility.
The uterine reproductive system is flexible when the body is under stress, they note.
“We know that running a marathon may influence hormone concentrations in the short term while not rendering that person infertile,” the authors write.
However, they acknowledge that investigating these reports is critical in building trust in medicine.
This report includes information that hasn’t been available through the clinical trial follow-up process.
For instance, the authors write, “To the best of our knowledge, our work is the first to examine breakthrough bleeding after vaccination in either pre- or postmenopausal people.”
Reports of changes to periods after vaccination started emerging in 2021. But without data, reports were largely dismissed, fueling criticism from those waging campaigns against COVID vaccines.
Dr. Lee and colleagues gathered data from those who responded to the online survey and detailed some trends.
People who were bleeding more heavily after vaccination were more likely to be older, Hispanic, had vaccine side effects of fever and fatigue, had been pregnant at some point, or had given birth.
People with regular periods who had endometriosis, prolonged bleeding during their periods, polycystic ovarian syndrome (PCOS) or fibroids were also more likely to have increased bleeding after a COVID vaccine.
Breakthrough bleeding
For people who don’t menstruate, but have not reached menopause, breakthrough bleeding happened more often in women who had been pregnant and/or had given birth.
Among respondents who were postmenopausal, breakthrough bleeding happened more often in younger people and/or those who are Hispanic.
More than a third of the respondents (39%) who use gender-affirming hormones that eliminate menstruation reported breakthrough bleeding after vaccination.
The majority of premenopausal people on long-acting, reversible contraception (71%) and the majority of postmenopausal respondents (66%) had breakthrough bleeding as well.
The authors note that you can’t compare the percentages who report these experiences in the survey with the incidence of those who would experience changes in menstrual bleeding in the general population.
The nature of the online survey means it may be naturally biased because the people who responded may be more often those who noted some change in their own menstrual experiences, particularly if that involved discomfort, pain, or fear.
Researchers also acknowledge that Black, Indigenous, Latinx, and other respondents of color are underrepresented in this research and that represents a limitation in the work.
Alison Edelman, MD, MPH, with the department of obstetrics and gynecology at Oregon Health & Science University in Portland, was not involved with Dr. Lee and associates’ study but has also studied the relationship between COVID vaccines and menstruation.
Her team’s study found that COVID vaccination is associated with a small change in time between periods but not length of periods.
She said about the work by Dr. Lee and colleagues, “This work really elevates the voices of the public and what they’re experiencing.”
The association makes sense, Dr. Edelman says, in that the reproductive system and the immune system talk to each other and inflammation in the immune system is going to be noticed by the system governing periods.
Lack of data on the relationship between exposures and menstruation didn’t start with COVID. “There has been a signal in the population before with other vaccines that’s been dismissed,” she said.
Tracking menstruation information in clinical trials can help physicians counsel women on what may be coming with any vaccine and alleviate fears and vaccine hesitancy, Dr. Edelman explained. It can also help vaccine developers know what to include in information about their product.
“When you are counseled about what to expect, it’s not as scary. That provides trust in the system,” she said. She likened it to original lack of data on whether COVID-19 vaccines would affect pregnancy.
“We have great science now that COVID vaccine does not affect fertility and [vaccine] does not impact pregnancy.”
Another important aspect of this paper is that it included subgroups not studied before regarding menstruation and breakthrough bleeding, such as those taking gender-affirming hormones, she added.
Menstruation has been often overlooked as important in clinical trial exposures but Dr. Edelman hopes this recent attention and question will escalate and prompt more research.
“I’m hoping with the immense outpouring from the public about how important this is, that future studies will look at this a little bit better,” she says.
She said when the National Institutes of Health opened up funding for trials on COVID-19 vaccines and menstruation, researchers got flooded with requests from women to share their stories.
“As a researcher – I’ve been doing research for over 20 years – that’s not something that usually happens. I would love to have that happen for every research project.”
The authors and Dr. Edelman declare that they have no competing interests. This research was supported in part by the University of Illinois Beckman Institute for Advanced Science and Technology, the University of Illinois Interdisciplinary Health Sciences Institute, the National Institutes of Health, the Foundation for Barnes-Jewish Hospital, and the Siteman Cancer Center.
Many women who got a COVID-19 vaccine have reported heavier bleeding during their periods since they had the shots.
A team of researchers investigated the trend and set out to find out who among the vaccinated were more likely to experience the menstruation changes.
The researchers were led by Katharine M.N. Lee, PhD, MS, of the division of public health sciences at Washington University in St. Louis. Their findings were published ahead of print in Science Advances.
The investigators analyzed more than 139,000 responses from an online survey from both currently and formerly menstruating women.
They found that, among people who have regular periods, about the same percentage had heavier bleeding after they got a COVID vaccine as had no change in bleeding after the vaccine (44% vs. 42%, respectively).
“A much smaller portion had lighter periods,” they write.
The phenomenon has been difficult to study because questions about changes in menstruation are not a standard part of vaccine trials.
Date of last period is often tracked in clinical trials to make sure a participant is not pregnant, but the questions about periods often stop there.
Additionally, periods are different for everyone and can be influenced by all sorts of environmental factors, so making associations regarding exposures is problematic.
No changes found to fertility
The authors emphasized that, generally, changes to menstrual bleeding are not uncommon nor dangerous. They also emphasized that the changes in bleeding don’t mean changes to fertility.
The uterine reproductive system is flexible when the body is under stress, they note.
“We know that running a marathon may influence hormone concentrations in the short term while not rendering that person infertile,” the authors write.
However, they acknowledge that investigating these reports is critical in building trust in medicine.
This report includes information that hasn’t been available through the clinical trial follow-up process.
For instance, the authors write, “To the best of our knowledge, our work is the first to examine breakthrough bleeding after vaccination in either pre- or postmenopausal people.”
Reports of changes to periods after vaccination started emerging in 2021. But without data, reports were largely dismissed, fueling criticism from those waging campaigns against COVID vaccines.
Dr. Lee and colleagues gathered data from those who responded to the online survey and detailed some trends.
People who were bleeding more heavily after vaccination were more likely to be older, Hispanic, had vaccine side effects of fever and fatigue, had been pregnant at some point, or had given birth.
People with regular periods who had endometriosis, prolonged bleeding during their periods, polycystic ovarian syndrome (PCOS) or fibroids were also more likely to have increased bleeding after a COVID vaccine.
Breakthrough bleeding
For people who don’t menstruate, but have not reached menopause, breakthrough bleeding happened more often in women who had been pregnant and/or had given birth.
Among respondents who were postmenopausal, breakthrough bleeding happened more often in younger people and/or those who are Hispanic.
More than a third of the respondents (39%) who use gender-affirming hormones that eliminate menstruation reported breakthrough bleeding after vaccination.
The majority of premenopausal people on long-acting, reversible contraception (71%) and the majority of postmenopausal respondents (66%) had breakthrough bleeding as well.
The authors note that you can’t compare the percentages who report these experiences in the survey with the incidence of those who would experience changes in menstrual bleeding in the general population.
The nature of the online survey means it may be naturally biased because the people who responded may be more often those who noted some change in their own menstrual experiences, particularly if that involved discomfort, pain, or fear.
Researchers also acknowledge that Black, Indigenous, Latinx, and other respondents of color are underrepresented in this research and that represents a limitation in the work.
Alison Edelman, MD, MPH, with the department of obstetrics and gynecology at Oregon Health & Science University in Portland, was not involved with Dr. Lee and associates’ study but has also studied the relationship between COVID vaccines and menstruation.
Her team’s study found that COVID vaccination is associated with a small change in time between periods but not length of periods.
She said about the work by Dr. Lee and colleagues, “This work really elevates the voices of the public and what they’re experiencing.”
The association makes sense, Dr. Edelman says, in that the reproductive system and the immune system talk to each other and inflammation in the immune system is going to be noticed by the system governing periods.
Lack of data on the relationship between exposures and menstruation didn’t start with COVID. “There has been a signal in the population before with other vaccines that’s been dismissed,” she said.
Tracking menstruation information in clinical trials can help physicians counsel women on what may be coming with any vaccine and alleviate fears and vaccine hesitancy, Dr. Edelman explained. It can also help vaccine developers know what to include in information about their product.
“When you are counseled about what to expect, it’s not as scary. That provides trust in the system,” she said. She likened it to original lack of data on whether COVID-19 vaccines would affect pregnancy.
“We have great science now that COVID vaccine does not affect fertility and [vaccine] does not impact pregnancy.”
Another important aspect of this paper is that it included subgroups not studied before regarding menstruation and breakthrough bleeding, such as those taking gender-affirming hormones, she added.
Menstruation has been often overlooked as important in clinical trial exposures but Dr. Edelman hopes this recent attention and question will escalate and prompt more research.
“I’m hoping with the immense outpouring from the public about how important this is, that future studies will look at this a little bit better,” she says.
She said when the National Institutes of Health opened up funding for trials on COVID-19 vaccines and menstruation, researchers got flooded with requests from women to share their stories.
“As a researcher – I’ve been doing research for over 20 years – that’s not something that usually happens. I would love to have that happen for every research project.”
The authors and Dr. Edelman declare that they have no competing interests. This research was supported in part by the University of Illinois Beckman Institute for Advanced Science and Technology, the University of Illinois Interdisciplinary Health Sciences Institute, the National Institutes of Health, the Foundation for Barnes-Jewish Hospital, and the Siteman Cancer Center.
FROM SCIENCE ADVANCES
Five-year cervical screening interval safe for HPV-negative women
A 5-year cervical screening interval is as safe and effective for women who test negative for human papillomavirus (HPV) as are 3-year intervals, according to a new ‘real life’ study led by King’s College London (KCL) with researchers from the University of Manchester, and the NHS, on behalf of the HPV pilot steering group.
The study, published in The BMJ, used data from the HPV screening pilot to assess rates of detection of high-grade cervical intraepithelial neoplasia (CIN3+) and of cervical cancer following a negative HPV test. It confirmed that 5-yearly screening prevents as many cancers as screening at 3-year intervals, even in women who are not vaccinated against HPV.
Change to primary HPV testing since 2019
Before 2019, the NHS cervical screening program conducted cytology testing first, testing for HPV only if abnormalities were found. In 2019, following reporting of early results of the HPV pilot by the same researchers, the program in England switched to testing for HPV first, on the grounds that since having HPV infection comes before having abnormal cells, HPV testing would detect more women at risk of cervical cancer.
Following the switch to primary HPV testing, the same screening intervals were retained, meaning 3-yearly screening for those aged 24-49 years and testing every 5 years for women aged 50-64 years, or 3 years if they tested positive. However, the National Screening Committee had recommended that invites should be changed from 3 to 5 years for those in the under-50 age group found not to have high-risk HPV at their routine screening test.
For the latest study, funded by Cancer Research UK, the steering group researchers analyzed details for more than 1.3 million women who had attended screening for two rounds of the HPV screening pilot, the first from 2013 to 2016, with a follow-up to the end of 2019. By this time, the data set had doubled in size from the pilot study, and results had been linked with the national cancer registry.
They confirmed that HPV testing was more accurate than a cytology test, irrespective of whether the HPV test assay was DNA- or mRNA-based. With HPV testing, the risk of subsequent cytological changes more than halved overall. Eligible women under 50 who had a negative HPV screen in the first round had a much lower risk of detection of CIN3+ in the second round, with a rate of 1.21 in 1,000, compared with 4.52 in 1,000 after a negative cytology test.
Data support extension of the testing interval
“The study confirms that women in this age group are much less likely to develop clinically relevant cervical lesions and cervical cancer, 3 years after a negative HPV screen, compared with a negative smear test,” the researchers said.
They suggested that most women do not need to be screened as frequently as the current program allows, and that the data support an extension of the screening intervals, regardless of the test assay used, to 5 years after a negative HPV test in women aged 25-49 years, and even longer for women aged 50 years and older.
However, the screening interval for HPV-positive women who have negative HPV tests at early recall should be kept at 3 years, they said.
“These results are very reassuring,” said lead author Matejka Rebolj, PhD, senior epidemiologist at KCL. “They build on previous research that shows that following the introduction of HPV testing for cervical screening, a 5-year interval is at least as safe as the previous 3-year interval. Changing to 5-yearly screening will mean we can prevent just as many cancers as before, while allowing for fewer screens.”
Michelle Mitchell, Cancer Research UK’s chief executive, said: “This large study shows that offering cervical screening using HPV testing effectively prevents cervical cancer, without having to be screened as often. This builds on findings from years of research showing HPV testing is more accurate at predicting who is at risk of developing cervical cancer compared to the previous way of testing. As changes to the screening [programs] are made, they will be monitored to help ensure that cervical screening is as effective as possible for all who take part.”
If HPV is present, testing interval should remain every 3 years
Responding to the study, Theresa Freeman-Wang, MBChB, consultant gynecologist, president of the British Society for Colposcopy and Cervical Pathology, and spokesperson for the Royal College of Obstetricians and Gynaecologists, told this news organization: “England, Scotland, and Wales and many other countries now use HPV primary screening, which is much better at assessing risk than previous methods. HPV testing is more sensitive and accurate, so changes are picked up earlier.
“Studies have confirmed that if someone is HPV negative (i.e., HPV is not present in the screen test), intervals between tests can very safely be increased from 3 to 5 years.
“If HPV is present, then the program will automatically look for any abnormal cells. If there are no abnormalities, the woman will be advised to have a repeat screen test in a year. If the HPV remains present over 3 successive years or if abnormal cells are detected at any stage, she will be referred for a more detailed screening examination called a colposcopy.
“It’s important that with any change like this, there is clear information available to explain what these changes mean.
“We have an effective cervical screening program in the UK that has significantly reduced the number of cases and deaths from this preventable cancer.
“HPV screening every 5 years is safe and to be fully effective it is vital that women take up the invitation for cervical screening when called.”
A version of this article first appeared on Medscape UK.
A 5-year cervical screening interval is as safe and effective for women who test negative for human papillomavirus (HPV) as are 3-year intervals, according to a new ‘real life’ study led by King’s College London (KCL) with researchers from the University of Manchester, and the NHS, on behalf of the HPV pilot steering group.
The study, published in The BMJ, used data from the HPV screening pilot to assess rates of detection of high-grade cervical intraepithelial neoplasia (CIN3+) and of cervical cancer following a negative HPV test. It confirmed that 5-yearly screening prevents as many cancers as screening at 3-year intervals, even in women who are not vaccinated against HPV.
Change to primary HPV testing since 2019
Before 2019, the NHS cervical screening program conducted cytology testing first, testing for HPV only if abnormalities were found. In 2019, following reporting of early results of the HPV pilot by the same researchers, the program in England switched to testing for HPV first, on the grounds that since having HPV infection comes before having abnormal cells, HPV testing would detect more women at risk of cervical cancer.
Following the switch to primary HPV testing, the same screening intervals were retained, meaning 3-yearly screening for those aged 24-49 years and testing every 5 years for women aged 50-64 years, or 3 years if they tested positive. However, the National Screening Committee had recommended that invites should be changed from 3 to 5 years for those in the under-50 age group found not to have high-risk HPV at their routine screening test.
For the latest study, funded by Cancer Research UK, the steering group researchers analyzed details for more than 1.3 million women who had attended screening for two rounds of the HPV screening pilot, the first from 2013 to 2016, with a follow-up to the end of 2019. By this time, the data set had doubled in size from the pilot study, and results had been linked with the national cancer registry.
They confirmed that HPV testing was more accurate than a cytology test, irrespective of whether the HPV test assay was DNA- or mRNA-based. With HPV testing, the risk of subsequent cytological changes more than halved overall. Eligible women under 50 who had a negative HPV screen in the first round had a much lower risk of detection of CIN3+ in the second round, with a rate of 1.21 in 1,000, compared with 4.52 in 1,000 after a negative cytology test.
Data support extension of the testing interval
“The study confirms that women in this age group are much less likely to develop clinically relevant cervical lesions and cervical cancer, 3 years after a negative HPV screen, compared with a negative smear test,” the researchers said.
They suggested that most women do not need to be screened as frequently as the current program allows, and that the data support an extension of the screening intervals, regardless of the test assay used, to 5 years after a negative HPV test in women aged 25-49 years, and even longer for women aged 50 years and older.
However, the screening interval for HPV-positive women who have negative HPV tests at early recall should be kept at 3 years, they said.
“These results are very reassuring,” said lead author Matejka Rebolj, PhD, senior epidemiologist at KCL. “They build on previous research that shows that following the introduction of HPV testing for cervical screening, a 5-year interval is at least as safe as the previous 3-year interval. Changing to 5-yearly screening will mean we can prevent just as many cancers as before, while allowing for fewer screens.”
Michelle Mitchell, Cancer Research UK’s chief executive, said: “This large study shows that offering cervical screening using HPV testing effectively prevents cervical cancer, without having to be screened as often. This builds on findings from years of research showing HPV testing is more accurate at predicting who is at risk of developing cervical cancer compared to the previous way of testing. As changes to the screening [programs] are made, they will be monitored to help ensure that cervical screening is as effective as possible for all who take part.”
If HPV is present, testing interval should remain every 3 years
Responding to the study, Theresa Freeman-Wang, MBChB, consultant gynecologist, president of the British Society for Colposcopy and Cervical Pathology, and spokesperson for the Royal College of Obstetricians and Gynaecologists, told this news organization: “England, Scotland, and Wales and many other countries now use HPV primary screening, which is much better at assessing risk than previous methods. HPV testing is more sensitive and accurate, so changes are picked up earlier.
“Studies have confirmed that if someone is HPV negative (i.e., HPV is not present in the screen test), intervals between tests can very safely be increased from 3 to 5 years.
“If HPV is present, then the program will automatically look for any abnormal cells. If there are no abnormalities, the woman will be advised to have a repeat screen test in a year. If the HPV remains present over 3 successive years or if abnormal cells are detected at any stage, she will be referred for a more detailed screening examination called a colposcopy.
“It’s important that with any change like this, there is clear information available to explain what these changes mean.
“We have an effective cervical screening program in the UK that has significantly reduced the number of cases and deaths from this preventable cancer.
“HPV screening every 5 years is safe and to be fully effective it is vital that women take up the invitation for cervical screening when called.”
A version of this article first appeared on Medscape UK.
A 5-year cervical screening interval is as safe and effective for women who test negative for human papillomavirus (HPV) as are 3-year intervals, according to a new ‘real life’ study led by King’s College London (KCL) with researchers from the University of Manchester, and the NHS, on behalf of the HPV pilot steering group.
The study, published in The BMJ, used data from the HPV screening pilot to assess rates of detection of high-grade cervical intraepithelial neoplasia (CIN3+) and of cervical cancer following a negative HPV test. It confirmed that 5-yearly screening prevents as many cancers as screening at 3-year intervals, even in women who are not vaccinated against HPV.
Change to primary HPV testing since 2019
Before 2019, the NHS cervical screening program conducted cytology testing first, testing for HPV only if abnormalities were found. In 2019, following reporting of early results of the HPV pilot by the same researchers, the program in England switched to testing for HPV first, on the grounds that since having HPV infection comes before having abnormal cells, HPV testing would detect more women at risk of cervical cancer.
Following the switch to primary HPV testing, the same screening intervals were retained, meaning 3-yearly screening for those aged 24-49 years and testing every 5 years for women aged 50-64 years, or 3 years if they tested positive. However, the National Screening Committee had recommended that invites should be changed from 3 to 5 years for those in the under-50 age group found not to have high-risk HPV at their routine screening test.
For the latest study, funded by Cancer Research UK, the steering group researchers analyzed details for more than 1.3 million women who had attended screening for two rounds of the HPV screening pilot, the first from 2013 to 2016, with a follow-up to the end of 2019. By this time, the data set had doubled in size from the pilot study, and results had been linked with the national cancer registry.
They confirmed that HPV testing was more accurate than a cytology test, irrespective of whether the HPV test assay was DNA- or mRNA-based. With HPV testing, the risk of subsequent cytological changes more than halved overall. Eligible women under 50 who had a negative HPV screen in the first round had a much lower risk of detection of CIN3+ in the second round, with a rate of 1.21 in 1,000, compared with 4.52 in 1,000 after a negative cytology test.
Data support extension of the testing interval
“The study confirms that women in this age group are much less likely to develop clinically relevant cervical lesions and cervical cancer, 3 years after a negative HPV screen, compared with a negative smear test,” the researchers said.
They suggested that most women do not need to be screened as frequently as the current program allows, and that the data support an extension of the screening intervals, regardless of the test assay used, to 5 years after a negative HPV test in women aged 25-49 years, and even longer for women aged 50 years and older.
However, the screening interval for HPV-positive women who have negative HPV tests at early recall should be kept at 3 years, they said.
“These results are very reassuring,” said lead author Matejka Rebolj, PhD, senior epidemiologist at KCL. “They build on previous research that shows that following the introduction of HPV testing for cervical screening, a 5-year interval is at least as safe as the previous 3-year interval. Changing to 5-yearly screening will mean we can prevent just as many cancers as before, while allowing for fewer screens.”
Michelle Mitchell, Cancer Research UK’s chief executive, said: “This large study shows that offering cervical screening using HPV testing effectively prevents cervical cancer, without having to be screened as often. This builds on findings from years of research showing HPV testing is more accurate at predicting who is at risk of developing cervical cancer compared to the previous way of testing. As changes to the screening [programs] are made, they will be monitored to help ensure that cervical screening is as effective as possible for all who take part.”
If HPV is present, testing interval should remain every 3 years
Responding to the study, Theresa Freeman-Wang, MBChB, consultant gynecologist, president of the British Society for Colposcopy and Cervical Pathology, and spokesperson for the Royal College of Obstetricians and Gynaecologists, told this news organization: “England, Scotland, and Wales and many other countries now use HPV primary screening, which is much better at assessing risk than previous methods. HPV testing is more sensitive and accurate, so changes are picked up earlier.
“Studies have confirmed that if someone is HPV negative (i.e., HPV is not present in the screen test), intervals between tests can very safely be increased from 3 to 5 years.
“If HPV is present, then the program will automatically look for any abnormal cells. If there are no abnormalities, the woman will be advised to have a repeat screen test in a year. If the HPV remains present over 3 successive years or if abnormal cells are detected at any stage, she will be referred for a more detailed screening examination called a colposcopy.
“It’s important that with any change like this, there is clear information available to explain what these changes mean.
“We have an effective cervical screening program in the UK that has significantly reduced the number of cases and deaths from this preventable cancer.
“HPV screening every 5 years is safe and to be fully effective it is vital that women take up the invitation for cervical screening when called.”
A version of this article first appeared on Medscape UK.
FROM THE BMJ
New test might transform male infertility
A new study suggests that, at least for certain male patients, the answer to infertility might lie with epigenetics.
According to the study, a commercially available test of epigenetic anomalies – factors that affect how genes express themselves – can grade the likelihood that sperm are viable for conception.
“The uniqueness of epigenetics is that some of the abnormalities detected have the potential to be modified with lifestyle,” said Larry I. Lipshultz, MD, head of the Division of Male Reproductive Medicine and Surgery at Baylor College of Medicine, Houston, who presented the new findings at the 2022 annual meeting of the American Urological Association.
For decades, semen analysis has been based on motility, morphology, and concentration. But these measures, while useful, are limited. Semen can still have low capacity for producing a pregnancy even when all three parameters are normal, Dr. Lipshultz told this news organization.
The test, called Path SpermQT (Inherent Biosciences) detects unstable gene promotors, which are the epigenetic markers for gene expression. In previous work with more than 1,300 gene samples, expression of the specific genes regulated by these promoters were linked to a wide variety of functions relevant to fertilization, such as spermatogenesis.
The test does not attempt to look for expression of specific unstable promoters but rather quantifies them to characterize sperm quality as excellent (≤ 10 unstable promoters), average (11-42), or poor (≥ 43).
In the studies that led to development of the SpermQT test, the number of unstable promoters correlated with pregnancy success. Pregnancy was achieved even among those in the group with poor sperm quality – but at very low rates.
Of the 172 semen samples collected so far in the ongoing analysis, sperm quality was characterized as excellent in 31%, average in 59%, and poor in 10%.
The stratifications for sperm quality were not significantly correlated with common measures of sperm viability, such as concentration, Dr. Lipshultz reported.
Certain patient characteristics were associated with greater sperm quality. These included use of antioxidant supplementation and low estrogen levels, as seen in men who had taken aromatase inhibitors.
So far, only one natural conception has occurred in the group with poor sperm versus eight in those with average or excellent quality.
The prognostic role of the test is only part of the picture.
“The exciting thing about this area of research is that epigenetics can be changed,” Dr. Lipshultz said in an interview. Based on the data so far, he said he is already starting to consider antioxidant supplementation and hormone modifications when sperm quality is poor.
The value of the Path SpermQT test for identifying treatment targets might eventually revolutionize the management of male infertility, Dr. Lipshultz said, but it has more immediate potential in helping couples decide whether to proceed with in vitro fertilization (IVF).
“We often see patients at an impasse when they are trying to decide to move to IVF,” he said. “A test like this could provide some direction. If sperm quality is good, the advice might be to keep trying. If poor, then a couple might want to move to IVF more quickly.”
A test of sperm quality on the basis of epigenetics “could change how we look at couples attempting to conceive,” agreed Peter N. Schlegel, MD, professor of urology and reproductive medicine, Weill Cornell Medicine, New York.
Dr. Schlegel praised several characteristics of the epigenetics test, including that 70%-80% of men with poor quality with SpermQT have normal results on standard assessments of semen. This finding suggests the tool is providing unique information about patients. He also noted that the studies so far indicate that sperm of poor quality for natural conception is still viable for IVF fertilization – which could be useful for couples weighing their options.
However, while the test is already available, Dr. Schlegel cautioned that much of the promise has yet to be documented.
“The results to date, despite being statistically significant, have only been gleaned from a small group of patients,” he said. “Much larger studies are needed before a change in practice is warranted.”
The value of SpermQT for identifying modifiable risks might be even further away.
“It is well recognized that environmental and lifestyle changes can affect methylation, but it is not known if the abnormalities seen so far could be influenced by lifestyle changes,” Dr. Schlegel said. Among the numerous steps needed to answer this question, he suggested that it might first be important “to evaluate why such changes in methylation occur.”
Dr. Lipshultz has financial relationships with several pharmaceutical companies, including Inherent Biosciences, which is marketing the SpermQT test. Dr. Schlegel has financial relationships with Theralogix, Posterity Health, and Roman Health.
A version of this article first appeared on Medscape.com.
A new study suggests that, at least for certain male patients, the answer to infertility might lie with epigenetics.
According to the study, a commercially available test of epigenetic anomalies – factors that affect how genes express themselves – can grade the likelihood that sperm are viable for conception.
“The uniqueness of epigenetics is that some of the abnormalities detected have the potential to be modified with lifestyle,” said Larry I. Lipshultz, MD, head of the Division of Male Reproductive Medicine and Surgery at Baylor College of Medicine, Houston, who presented the new findings at the 2022 annual meeting of the American Urological Association.
For decades, semen analysis has been based on motility, morphology, and concentration. But these measures, while useful, are limited. Semen can still have low capacity for producing a pregnancy even when all three parameters are normal, Dr. Lipshultz told this news organization.
The test, called Path SpermQT (Inherent Biosciences) detects unstable gene promotors, which are the epigenetic markers for gene expression. In previous work with more than 1,300 gene samples, expression of the specific genes regulated by these promoters were linked to a wide variety of functions relevant to fertilization, such as spermatogenesis.
The test does not attempt to look for expression of specific unstable promoters but rather quantifies them to characterize sperm quality as excellent (≤ 10 unstable promoters), average (11-42), or poor (≥ 43).
In the studies that led to development of the SpermQT test, the number of unstable promoters correlated with pregnancy success. Pregnancy was achieved even among those in the group with poor sperm quality – but at very low rates.
Of the 172 semen samples collected so far in the ongoing analysis, sperm quality was characterized as excellent in 31%, average in 59%, and poor in 10%.
The stratifications for sperm quality were not significantly correlated with common measures of sperm viability, such as concentration, Dr. Lipshultz reported.
Certain patient characteristics were associated with greater sperm quality. These included use of antioxidant supplementation and low estrogen levels, as seen in men who had taken aromatase inhibitors.
So far, only one natural conception has occurred in the group with poor sperm versus eight in those with average or excellent quality.
The prognostic role of the test is only part of the picture.
“The exciting thing about this area of research is that epigenetics can be changed,” Dr. Lipshultz said in an interview. Based on the data so far, he said he is already starting to consider antioxidant supplementation and hormone modifications when sperm quality is poor.
The value of the Path SpermQT test for identifying treatment targets might eventually revolutionize the management of male infertility, Dr. Lipshultz said, but it has more immediate potential in helping couples decide whether to proceed with in vitro fertilization (IVF).
“We often see patients at an impasse when they are trying to decide to move to IVF,” he said. “A test like this could provide some direction. If sperm quality is good, the advice might be to keep trying. If poor, then a couple might want to move to IVF more quickly.”
A test of sperm quality on the basis of epigenetics “could change how we look at couples attempting to conceive,” agreed Peter N. Schlegel, MD, professor of urology and reproductive medicine, Weill Cornell Medicine, New York.
Dr. Schlegel praised several characteristics of the epigenetics test, including that 70%-80% of men with poor quality with SpermQT have normal results on standard assessments of semen. This finding suggests the tool is providing unique information about patients. He also noted that the studies so far indicate that sperm of poor quality for natural conception is still viable for IVF fertilization – which could be useful for couples weighing their options.
However, while the test is already available, Dr. Schlegel cautioned that much of the promise has yet to be documented.
“The results to date, despite being statistically significant, have only been gleaned from a small group of patients,” he said. “Much larger studies are needed before a change in practice is warranted.”
The value of SpermQT for identifying modifiable risks might be even further away.
“It is well recognized that environmental and lifestyle changes can affect methylation, but it is not known if the abnormalities seen so far could be influenced by lifestyle changes,” Dr. Schlegel said. Among the numerous steps needed to answer this question, he suggested that it might first be important “to evaluate why such changes in methylation occur.”
Dr. Lipshultz has financial relationships with several pharmaceutical companies, including Inherent Biosciences, which is marketing the SpermQT test. Dr. Schlegel has financial relationships with Theralogix, Posterity Health, and Roman Health.
A version of this article first appeared on Medscape.com.
A new study suggests that, at least for certain male patients, the answer to infertility might lie with epigenetics.
According to the study, a commercially available test of epigenetic anomalies – factors that affect how genes express themselves – can grade the likelihood that sperm are viable for conception.
“The uniqueness of epigenetics is that some of the abnormalities detected have the potential to be modified with lifestyle,” said Larry I. Lipshultz, MD, head of the Division of Male Reproductive Medicine and Surgery at Baylor College of Medicine, Houston, who presented the new findings at the 2022 annual meeting of the American Urological Association.
For decades, semen analysis has been based on motility, morphology, and concentration. But these measures, while useful, are limited. Semen can still have low capacity for producing a pregnancy even when all three parameters are normal, Dr. Lipshultz told this news organization.
The test, called Path SpermQT (Inherent Biosciences) detects unstable gene promotors, which are the epigenetic markers for gene expression. In previous work with more than 1,300 gene samples, expression of the specific genes regulated by these promoters were linked to a wide variety of functions relevant to fertilization, such as spermatogenesis.
The test does not attempt to look for expression of specific unstable promoters but rather quantifies them to characterize sperm quality as excellent (≤ 10 unstable promoters), average (11-42), or poor (≥ 43).
In the studies that led to development of the SpermQT test, the number of unstable promoters correlated with pregnancy success. Pregnancy was achieved even among those in the group with poor sperm quality – but at very low rates.
Of the 172 semen samples collected so far in the ongoing analysis, sperm quality was characterized as excellent in 31%, average in 59%, and poor in 10%.
The stratifications for sperm quality were not significantly correlated with common measures of sperm viability, such as concentration, Dr. Lipshultz reported.
Certain patient characteristics were associated with greater sperm quality. These included use of antioxidant supplementation and low estrogen levels, as seen in men who had taken aromatase inhibitors.
So far, only one natural conception has occurred in the group with poor sperm versus eight in those with average or excellent quality.
The prognostic role of the test is only part of the picture.
“The exciting thing about this area of research is that epigenetics can be changed,” Dr. Lipshultz said in an interview. Based on the data so far, he said he is already starting to consider antioxidant supplementation and hormone modifications when sperm quality is poor.
The value of the Path SpermQT test for identifying treatment targets might eventually revolutionize the management of male infertility, Dr. Lipshultz said, but it has more immediate potential in helping couples decide whether to proceed with in vitro fertilization (IVF).
“We often see patients at an impasse when they are trying to decide to move to IVF,” he said. “A test like this could provide some direction. If sperm quality is good, the advice might be to keep trying. If poor, then a couple might want to move to IVF more quickly.”
A test of sperm quality on the basis of epigenetics “could change how we look at couples attempting to conceive,” agreed Peter N. Schlegel, MD, professor of urology and reproductive medicine, Weill Cornell Medicine, New York.
Dr. Schlegel praised several characteristics of the epigenetics test, including that 70%-80% of men with poor quality with SpermQT have normal results on standard assessments of semen. This finding suggests the tool is providing unique information about patients. He also noted that the studies so far indicate that sperm of poor quality for natural conception is still viable for IVF fertilization – which could be useful for couples weighing their options.
However, while the test is already available, Dr. Schlegel cautioned that much of the promise has yet to be documented.
“The results to date, despite being statistically significant, have only been gleaned from a small group of patients,” he said. “Much larger studies are needed before a change in practice is warranted.”
The value of SpermQT for identifying modifiable risks might be even further away.
“It is well recognized that environmental and lifestyle changes can affect methylation, but it is not known if the abnormalities seen so far could be influenced by lifestyle changes,” Dr. Schlegel said. Among the numerous steps needed to answer this question, he suggested that it might first be important “to evaluate why such changes in methylation occur.”
Dr. Lipshultz has financial relationships with several pharmaceutical companies, including Inherent Biosciences, which is marketing the SpermQT test. Dr. Schlegel has financial relationships with Theralogix, Posterity Health, and Roman Health.
A version of this article first appeared on Medscape.com.
FROM 2022 AMERICAN UROLOGICAL ASSOCIATION
Double the pleasure: Stim patch delays early ejaculation: Study
A wearable patch that delivers electrical stimulation to the perineum may postpone premature ejaculation, according to research presented at the annual meeting of the American Urological Association. The disposable device appears to work by helping men contract the muscles in the pelvic floor, allowing them to postpone climax.
Among 34 men with a lifelong history of premature ejaculation, average intravaginal ejaculatory latency time – the time from vaginal penetration to ejaculation – increased from about 67 seconds at baseline to 123 seconds when they used the device.
Another 17 participants received a sham treatment – stimulation they could feel but that did not activate muscles. In this group, time to ejaculation increased from 63 seconds to 81 seconds.
The longer duration with active treatment was statistically significant (P < .0001), whereas the increase in the control group was not (P = .1653), said Ege Can Serefoglu, MD, a researcher at Biruni University, Istanbul, and editor-in-chief of the International Journal of Impotence Research.
Dr. Serefoglu is a member of the scientific advisory board for Virility Medical, a company in Hod Hasharon, Israel, that is developing the stimulator. Marketed as vPatch, the device is expected to be available in 2023, Dr. Serefoglu said. It was cleared by the Food and Drug Administration in November and has CE-mark approval in Europe, according to the company.
Common problem, limited options
Research shows that 20%-30% of men are not happy with their time to ejaculation, Dr. Serefoglu said.
The International Society for Sexual Medicine defines premature ejaculation as ejaculation which always or almost always occurs within about 1 minute of penetration, the patient is unable to delay this occurrence, and the condition causes personal distress.
“Unfortunately, in spite of its high prevalence we do not really have any satisfying treatment options,” Dr. Serefoglu said.
Topical anesthetics may be used to decrease the sensitivity of the glans penis, and selective serotonin reuptake inhibitors may help delay ejaculation. But these options have limited efficacy and low adherence, he said.
Preclinical studies have shown that injection of botulinum toxin into the bulbospongiosus muscles is associated with a dose-dependent increase in ejaculation latency in rats.
Data on ClinicalTrials.gov show that this approach also may increase ejaculation latency in men, Dr. Serefoglu said. Although investigators found no safety concerns, drugmaker Allergan made a strategic business decision to stop developing this treatment approach, according to the registration entry for the study.
The idea for vPatch came from researchers wondering if instead of paralyzing the muscles with botulinum toxin, they used electrical stimulation to cause contraction of those muscles, Dr. Serefoglu said. A smaller proof-of-concept study demonstrated the feasibility and safety of this technique.
To further assess the safety and efficacy of a transcutaneous perineal electrical stimulator for the treatment of premature ejaculation, investigators conducted the randomized, double-blind, sham-controlled trial at Rambam Medical Centre, Haifa, Israel, and Villa Donatello Clinic, Florence, Italy.
The trial included males with premature ejaculation aged 18-60 years. Their female partners measured IELT using a stopwatch during four sexual intercourse sessions before treatment, and four times on treatment, at home.
In addition to the increased time to ejaculation, perceived control over ejaculation, satisfaction with sexual intercourse, personal distress related to ejaculation, and interpersonal difficulty related to ejaculation all significantly improved with vPatch, the researchers found.
Of participants who received active treatment, 73.5% reported a subjective sense of improvement versus 41.2% of the control group.
Potential reactions
No serious adverse events were observed, Dr. Serefoglu reported. Potential adverse reactions include redness, discomfort, and localized pain, according to the company’s website.
Men should not use vPatch if they have been diagnosed with pelvic cancer, or if they have an implanted electronic device, diabetes with peripheral neuropathy, or perineal dermatologic diseases, irritations, or lesions. Other precautions include avoiding use of the vPatch in water or humid environments. The device has not been tested on use with a pregnant partner.
The disposable patches are meant for one-time use. “The miniaturized perineal stimulation device may become an on-demand, drug-free therapeutic option,” Dr. Serefoglu said.
Combining electrical stimulation with other treatment approaches may provide additional benefit, said Bradley Schwartz, DO, professor and chairman of urology at Southern Illinois University, Springfield, who moderated the session at the AUA meeting at which the results of the study were presented.
“You go from 1 to 2 minutes just with this device,” Dr. Schwartz said. “If you went from 2 to 3 minutes, you would essentially be tripling their pleasure or their time, which might make a significant difference.”
Serefoglu agreed that combining the stimulator with other treatment approaches such as topical anesthetics could increase patient satisfaction.
Comoderator Kelly Healy, MD, assistant professor of urology at Columbia University Medical Center, New York, highlighted a direction for future research: examining outcomes according to different types of relationships, as well as partner satisfaction.
“That is a perfect question that should also be considered in the future trials,” Dr. Serefoglu said. “This was mainly focused on the man’s satisfaction. But men are trying to delay their ejaculation to satisfy their partner.”
Dr. Serefoglu is on the scientific advisory board for Virility Medical, which sponsored the study. Dr. Healy had no disclosures. Dr. Schwartz disclosed ties to Cook Medical.
A version of this article first appeared on Medscape.com.
A wearable patch that delivers electrical stimulation to the perineum may postpone premature ejaculation, according to research presented at the annual meeting of the American Urological Association. The disposable device appears to work by helping men contract the muscles in the pelvic floor, allowing them to postpone climax.
Among 34 men with a lifelong history of premature ejaculation, average intravaginal ejaculatory latency time – the time from vaginal penetration to ejaculation – increased from about 67 seconds at baseline to 123 seconds when they used the device.
Another 17 participants received a sham treatment – stimulation they could feel but that did not activate muscles. In this group, time to ejaculation increased from 63 seconds to 81 seconds.
The longer duration with active treatment was statistically significant (P < .0001), whereas the increase in the control group was not (P = .1653), said Ege Can Serefoglu, MD, a researcher at Biruni University, Istanbul, and editor-in-chief of the International Journal of Impotence Research.
Dr. Serefoglu is a member of the scientific advisory board for Virility Medical, a company in Hod Hasharon, Israel, that is developing the stimulator. Marketed as vPatch, the device is expected to be available in 2023, Dr. Serefoglu said. It was cleared by the Food and Drug Administration in November and has CE-mark approval in Europe, according to the company.
Common problem, limited options
Research shows that 20%-30% of men are not happy with their time to ejaculation, Dr. Serefoglu said.
The International Society for Sexual Medicine defines premature ejaculation as ejaculation which always or almost always occurs within about 1 minute of penetration, the patient is unable to delay this occurrence, and the condition causes personal distress.
“Unfortunately, in spite of its high prevalence we do not really have any satisfying treatment options,” Dr. Serefoglu said.
Topical anesthetics may be used to decrease the sensitivity of the glans penis, and selective serotonin reuptake inhibitors may help delay ejaculation. But these options have limited efficacy and low adherence, he said.
Preclinical studies have shown that injection of botulinum toxin into the bulbospongiosus muscles is associated with a dose-dependent increase in ejaculation latency in rats.
Data on ClinicalTrials.gov show that this approach also may increase ejaculation latency in men, Dr. Serefoglu said. Although investigators found no safety concerns, drugmaker Allergan made a strategic business decision to stop developing this treatment approach, according to the registration entry for the study.
The idea for vPatch came from researchers wondering if instead of paralyzing the muscles with botulinum toxin, they used electrical stimulation to cause contraction of those muscles, Dr. Serefoglu said. A smaller proof-of-concept study demonstrated the feasibility and safety of this technique.
To further assess the safety and efficacy of a transcutaneous perineal electrical stimulator for the treatment of premature ejaculation, investigators conducted the randomized, double-blind, sham-controlled trial at Rambam Medical Centre, Haifa, Israel, and Villa Donatello Clinic, Florence, Italy.
The trial included males with premature ejaculation aged 18-60 years. Their female partners measured IELT using a stopwatch during four sexual intercourse sessions before treatment, and four times on treatment, at home.
In addition to the increased time to ejaculation, perceived control over ejaculation, satisfaction with sexual intercourse, personal distress related to ejaculation, and interpersonal difficulty related to ejaculation all significantly improved with vPatch, the researchers found.
Of participants who received active treatment, 73.5% reported a subjective sense of improvement versus 41.2% of the control group.
Potential reactions
No serious adverse events were observed, Dr. Serefoglu reported. Potential adverse reactions include redness, discomfort, and localized pain, according to the company’s website.
Men should not use vPatch if they have been diagnosed with pelvic cancer, or if they have an implanted electronic device, diabetes with peripheral neuropathy, or perineal dermatologic diseases, irritations, or lesions. Other precautions include avoiding use of the vPatch in water or humid environments. The device has not been tested on use with a pregnant partner.
The disposable patches are meant for one-time use. “The miniaturized perineal stimulation device may become an on-demand, drug-free therapeutic option,” Dr. Serefoglu said.
Combining electrical stimulation with other treatment approaches may provide additional benefit, said Bradley Schwartz, DO, professor and chairman of urology at Southern Illinois University, Springfield, who moderated the session at the AUA meeting at which the results of the study were presented.
“You go from 1 to 2 minutes just with this device,” Dr. Schwartz said. “If you went from 2 to 3 minutes, you would essentially be tripling their pleasure or their time, which might make a significant difference.”
Serefoglu agreed that combining the stimulator with other treatment approaches such as topical anesthetics could increase patient satisfaction.
Comoderator Kelly Healy, MD, assistant professor of urology at Columbia University Medical Center, New York, highlighted a direction for future research: examining outcomes according to different types of relationships, as well as partner satisfaction.
“That is a perfect question that should also be considered in the future trials,” Dr. Serefoglu said. “This was mainly focused on the man’s satisfaction. But men are trying to delay their ejaculation to satisfy their partner.”
Dr. Serefoglu is on the scientific advisory board for Virility Medical, which sponsored the study. Dr. Healy had no disclosures. Dr. Schwartz disclosed ties to Cook Medical.
A version of this article first appeared on Medscape.com.
A wearable patch that delivers electrical stimulation to the perineum may postpone premature ejaculation, according to research presented at the annual meeting of the American Urological Association. The disposable device appears to work by helping men contract the muscles in the pelvic floor, allowing them to postpone climax.
Among 34 men with a lifelong history of premature ejaculation, average intravaginal ejaculatory latency time – the time from vaginal penetration to ejaculation – increased from about 67 seconds at baseline to 123 seconds when they used the device.
Another 17 participants received a sham treatment – stimulation they could feel but that did not activate muscles. In this group, time to ejaculation increased from 63 seconds to 81 seconds.
The longer duration with active treatment was statistically significant (P < .0001), whereas the increase in the control group was not (P = .1653), said Ege Can Serefoglu, MD, a researcher at Biruni University, Istanbul, and editor-in-chief of the International Journal of Impotence Research.
Dr. Serefoglu is a member of the scientific advisory board for Virility Medical, a company in Hod Hasharon, Israel, that is developing the stimulator. Marketed as vPatch, the device is expected to be available in 2023, Dr. Serefoglu said. It was cleared by the Food and Drug Administration in November and has CE-mark approval in Europe, according to the company.
Common problem, limited options
Research shows that 20%-30% of men are not happy with their time to ejaculation, Dr. Serefoglu said.
The International Society for Sexual Medicine defines premature ejaculation as ejaculation which always or almost always occurs within about 1 minute of penetration, the patient is unable to delay this occurrence, and the condition causes personal distress.
“Unfortunately, in spite of its high prevalence we do not really have any satisfying treatment options,” Dr. Serefoglu said.
Topical anesthetics may be used to decrease the sensitivity of the glans penis, and selective serotonin reuptake inhibitors may help delay ejaculation. But these options have limited efficacy and low adherence, he said.
Preclinical studies have shown that injection of botulinum toxin into the bulbospongiosus muscles is associated with a dose-dependent increase in ejaculation latency in rats.
Data on ClinicalTrials.gov show that this approach also may increase ejaculation latency in men, Dr. Serefoglu said. Although investigators found no safety concerns, drugmaker Allergan made a strategic business decision to stop developing this treatment approach, according to the registration entry for the study.
The idea for vPatch came from researchers wondering if instead of paralyzing the muscles with botulinum toxin, they used electrical stimulation to cause contraction of those muscles, Dr. Serefoglu said. A smaller proof-of-concept study demonstrated the feasibility and safety of this technique.
To further assess the safety and efficacy of a transcutaneous perineal electrical stimulator for the treatment of premature ejaculation, investigators conducted the randomized, double-blind, sham-controlled trial at Rambam Medical Centre, Haifa, Israel, and Villa Donatello Clinic, Florence, Italy.
The trial included males with premature ejaculation aged 18-60 years. Their female partners measured IELT using a stopwatch during four sexual intercourse sessions before treatment, and four times on treatment, at home.
In addition to the increased time to ejaculation, perceived control over ejaculation, satisfaction with sexual intercourse, personal distress related to ejaculation, and interpersonal difficulty related to ejaculation all significantly improved with vPatch, the researchers found.
Of participants who received active treatment, 73.5% reported a subjective sense of improvement versus 41.2% of the control group.
Potential reactions
No serious adverse events were observed, Dr. Serefoglu reported. Potential adverse reactions include redness, discomfort, and localized pain, according to the company’s website.
Men should not use vPatch if they have been diagnosed with pelvic cancer, or if they have an implanted electronic device, diabetes with peripheral neuropathy, or perineal dermatologic diseases, irritations, or lesions. Other precautions include avoiding use of the vPatch in water or humid environments. The device has not been tested on use with a pregnant partner.
The disposable patches are meant for one-time use. “The miniaturized perineal stimulation device may become an on-demand, drug-free therapeutic option,” Dr. Serefoglu said.
Combining electrical stimulation with other treatment approaches may provide additional benefit, said Bradley Schwartz, DO, professor and chairman of urology at Southern Illinois University, Springfield, who moderated the session at the AUA meeting at which the results of the study were presented.
“You go from 1 to 2 minutes just with this device,” Dr. Schwartz said. “If you went from 2 to 3 minutes, you would essentially be tripling their pleasure or their time, which might make a significant difference.”
Serefoglu agreed that combining the stimulator with other treatment approaches such as topical anesthetics could increase patient satisfaction.
Comoderator Kelly Healy, MD, assistant professor of urology at Columbia University Medical Center, New York, highlighted a direction for future research: examining outcomes according to different types of relationships, as well as partner satisfaction.
“That is a perfect question that should also be considered in the future trials,” Dr. Serefoglu said. “This was mainly focused on the man’s satisfaction. But men are trying to delay their ejaculation to satisfy their partner.”
Dr. Serefoglu is on the scientific advisory board for Virility Medical, which sponsored the study. Dr. Healy had no disclosures. Dr. Schwartz disclosed ties to Cook Medical.
A version of this article first appeared on Medscape.com.
Doxycycline bests azithromycin for anorectal chlamydia in women
NEW YORK (Reuters) – A one-week course of doxycycline was superior to a single dose of azithromycin in women with concurrent vaginal and anorectal chlamydia infection in an unblinded randomized controlled trial, mirroring previous results in men.
Researchers suggest that doxycycline should be the first-line therapy for chlamydia infection in women.
“It is clear we must consider that any woman with a urogenital infection must have an effective treatment for the anal infection, since nearly 80% of women have an anal infection concomitant with the vaginal infection,” Dr. Bertille de Barbeyrac of the University of Bordeaux, France, told Reuters Health by email.
However, she noted that “even [though] the study shows that doxycycline is more effective than azithromycin on anal infection, other studies are needed to prove that residual anal infection after treatment with azithromycin can be a source of vaginal contamination and therefore justify changing practices and eliminating azithromycin as a treatment for lower urogenital chlamydial infection in women.”
“There are other reasons [to make] this change,” she added, “such as the acquisition of macrolide resistance by M. genitalium following heavy use of azithromycin.”
As reported in The Lancet Infectious Diseases, Dr. Barbeyrac and colleagues randomly assigned 460 women (median age, 21) to either doxycycline or azithromycin in a multicenter, open-label superiority trial.
Participants received either azithromycin (a single 1-g dose, with or without food) or doxycycline (100 mg in the morning and evening at mealtimes for 7 days – that is, 100 mg of doxycycline twice daily).
The primary outcome was that the microbiological anorectal cure rate, defined as a C. trachomatis-negative nucleic acid amplification test (NAAT), resulted in anorectal specimens six weeks after treatment initiation among women who had a baseline positive result (about half the women in each treatment group).
Ninety-four percent of the doxycycline group versus 85% of the azithromycin group had an anorectal cure (adjusted odds ratio with imputation of missing values, 0.43).
Adverse events possibly related to treatment occurred in 11% of the doxycycline group versus 13% of the azithromycin group. Gastrointestinal disorders were most frequent, occurring in 8% of the doxycycline and 11% of the azithromycin groups.
Summing up, the authors write, “The microbiological anorectal cure rate was significantly lower among women who received a single dose of azithromycin than among those who received a 1-week course of doxycycline. This finding suggests that doxycycline should be the first-line therapy for C trachomatis infection in women.”
Dr. Meleen Chuang, medical director of women’s health at the Family Health Centers at NYU Langone, Brooklyn, commented in an email to Reuters Health that after reviewing this study “as well as CDC and WHO recommendations updated as of 2022, health care providers should be treating C. trachomatis infections with doxycycline 100 mg twice a day for seven days as first-line therapy rather than azithromycin, [given] concerns of increasing macrolide drug resistance against Mycoplasma genitalium and Neisseria gonorrhea.”
“Our clinicians also see the growing uptick of syphilis, gonorrhea, and chlamydia infections in our population, similarly to the rest of the United States since 2020,” she noted. “With the increase in STD infection ... treatment with doxycycline therapy with an important caveat to the patient to complete the one-week treatment regimen is extremely important.”
Dr. Latasha Murphy of the Gynecologic Care Institute at Mercy, Baltimore, also commented in an email to Reuters Health. She noted, “this study does not mirror my clinical experience. More patients have side effects from doxycycline than azithromycin in my experience. Also, anorectal screening is not routine in STD screening.”
“If any major changes to clinical care are made,” she said, “it may be for more consistent screening for anorectal disease. This may ultimately lead to doxycycline being the first line-treatment. More research is needed before making any definitive changes.”
Reuters Health Information © 2022
NEW YORK (Reuters) – A one-week course of doxycycline was superior to a single dose of azithromycin in women with concurrent vaginal and anorectal chlamydia infection in an unblinded randomized controlled trial, mirroring previous results in men.
Researchers suggest that doxycycline should be the first-line therapy for chlamydia infection in women.
“It is clear we must consider that any woman with a urogenital infection must have an effective treatment for the anal infection, since nearly 80% of women have an anal infection concomitant with the vaginal infection,” Dr. Bertille de Barbeyrac of the University of Bordeaux, France, told Reuters Health by email.
However, she noted that “even [though] the study shows that doxycycline is more effective than azithromycin on anal infection, other studies are needed to prove that residual anal infection after treatment with azithromycin can be a source of vaginal contamination and therefore justify changing practices and eliminating azithromycin as a treatment for lower urogenital chlamydial infection in women.”
“There are other reasons [to make] this change,” she added, “such as the acquisition of macrolide resistance by M. genitalium following heavy use of azithromycin.”
As reported in The Lancet Infectious Diseases, Dr. Barbeyrac and colleagues randomly assigned 460 women (median age, 21) to either doxycycline or azithromycin in a multicenter, open-label superiority trial.
Participants received either azithromycin (a single 1-g dose, with or without food) or doxycycline (100 mg in the morning and evening at mealtimes for 7 days – that is, 100 mg of doxycycline twice daily).
The primary outcome was that the microbiological anorectal cure rate, defined as a C. trachomatis-negative nucleic acid amplification test (NAAT), resulted in anorectal specimens six weeks after treatment initiation among women who had a baseline positive result (about half the women in each treatment group).
Ninety-four percent of the doxycycline group versus 85% of the azithromycin group had an anorectal cure (adjusted odds ratio with imputation of missing values, 0.43).
Adverse events possibly related to treatment occurred in 11% of the doxycycline group versus 13% of the azithromycin group. Gastrointestinal disorders were most frequent, occurring in 8% of the doxycycline and 11% of the azithromycin groups.
Summing up, the authors write, “The microbiological anorectal cure rate was significantly lower among women who received a single dose of azithromycin than among those who received a 1-week course of doxycycline. This finding suggests that doxycycline should be the first-line therapy for C trachomatis infection in women.”
Dr. Meleen Chuang, medical director of women’s health at the Family Health Centers at NYU Langone, Brooklyn, commented in an email to Reuters Health that after reviewing this study “as well as CDC and WHO recommendations updated as of 2022, health care providers should be treating C. trachomatis infections with doxycycline 100 mg twice a day for seven days as first-line therapy rather than azithromycin, [given] concerns of increasing macrolide drug resistance against Mycoplasma genitalium and Neisseria gonorrhea.”
“Our clinicians also see the growing uptick of syphilis, gonorrhea, and chlamydia infections in our population, similarly to the rest of the United States since 2020,” she noted. “With the increase in STD infection ... treatment with doxycycline therapy with an important caveat to the patient to complete the one-week treatment regimen is extremely important.”
Dr. Latasha Murphy of the Gynecologic Care Institute at Mercy, Baltimore, also commented in an email to Reuters Health. She noted, “this study does not mirror my clinical experience. More patients have side effects from doxycycline than azithromycin in my experience. Also, anorectal screening is not routine in STD screening.”
“If any major changes to clinical care are made,” she said, “it may be for more consistent screening for anorectal disease. This may ultimately lead to doxycycline being the first line-treatment. More research is needed before making any definitive changes.”
Reuters Health Information © 2022
NEW YORK (Reuters) – A one-week course of doxycycline was superior to a single dose of azithromycin in women with concurrent vaginal and anorectal chlamydia infection in an unblinded randomized controlled trial, mirroring previous results in men.
Researchers suggest that doxycycline should be the first-line therapy for chlamydia infection in women.
“It is clear we must consider that any woman with a urogenital infection must have an effective treatment for the anal infection, since nearly 80% of women have an anal infection concomitant with the vaginal infection,” Dr. Bertille de Barbeyrac of the University of Bordeaux, France, told Reuters Health by email.
However, she noted that “even [though] the study shows that doxycycline is more effective than azithromycin on anal infection, other studies are needed to prove that residual anal infection after treatment with azithromycin can be a source of vaginal contamination and therefore justify changing practices and eliminating azithromycin as a treatment for lower urogenital chlamydial infection in women.”
“There are other reasons [to make] this change,” she added, “such as the acquisition of macrolide resistance by M. genitalium following heavy use of azithromycin.”
As reported in The Lancet Infectious Diseases, Dr. Barbeyrac and colleagues randomly assigned 460 women (median age, 21) to either doxycycline or azithromycin in a multicenter, open-label superiority trial.
Participants received either azithromycin (a single 1-g dose, with or without food) or doxycycline (100 mg in the morning and evening at mealtimes for 7 days – that is, 100 mg of doxycycline twice daily).
The primary outcome was that the microbiological anorectal cure rate, defined as a C. trachomatis-negative nucleic acid amplification test (NAAT), resulted in anorectal specimens six weeks after treatment initiation among women who had a baseline positive result (about half the women in each treatment group).
Ninety-four percent of the doxycycline group versus 85% of the azithromycin group had an anorectal cure (adjusted odds ratio with imputation of missing values, 0.43).
Adverse events possibly related to treatment occurred in 11% of the doxycycline group versus 13% of the azithromycin group. Gastrointestinal disorders were most frequent, occurring in 8% of the doxycycline and 11% of the azithromycin groups.
Summing up, the authors write, “The microbiological anorectal cure rate was significantly lower among women who received a single dose of azithromycin than among those who received a 1-week course of doxycycline. This finding suggests that doxycycline should be the first-line therapy for C trachomatis infection in women.”
Dr. Meleen Chuang, medical director of women’s health at the Family Health Centers at NYU Langone, Brooklyn, commented in an email to Reuters Health that after reviewing this study “as well as CDC and WHO recommendations updated as of 2022, health care providers should be treating C. trachomatis infections with doxycycline 100 mg twice a day for seven days as first-line therapy rather than azithromycin, [given] concerns of increasing macrolide drug resistance against Mycoplasma genitalium and Neisseria gonorrhea.”
“Our clinicians also see the growing uptick of syphilis, gonorrhea, and chlamydia infections in our population, similarly to the rest of the United States since 2020,” she noted. “With the increase in STD infection ... treatment with doxycycline therapy with an important caveat to the patient to complete the one-week treatment regimen is extremely important.”
Dr. Latasha Murphy of the Gynecologic Care Institute at Mercy, Baltimore, also commented in an email to Reuters Health. She noted, “this study does not mirror my clinical experience. More patients have side effects from doxycycline than azithromycin in my experience. Also, anorectal screening is not routine in STD screening.”
“If any major changes to clinical care are made,” she said, “it may be for more consistent screening for anorectal disease. This may ultimately lead to doxycycline being the first line-treatment. More research is needed before making any definitive changes.”
Reuters Health Information © 2022
OTC meds, supplements, and other drugs may interact with HIV antiretrovirals
Over-the-counter medications, food supplements, and other drugs may interact with antiretroviral therapy (ART) in people living with HIV and be harmful, an industry-sponsored clinical survey from Denmark reports.
“Our study confirms that polypharmacy and being on a protease inhibitor–based regimen increase the risk of potential drug-drug interactions [PDDIs] considerably and highlights the importance of questioning people living with HIV [PLWH] about dietary supplement intake,” the authors, led by Michaela Tinggaard, MD, Copenhagen University Hospital, wrote in HIV Medicine.
“Potential drug-drug interactions were common among our study population. Although the clinical significance of the majority of the identified PDDIs may be low, most of them were avoidable through a change or discontinuation of the comedication, a change in ART or by spacing drugs,” they added.
Senior author Thomas Benfield, MD, DTMH, DMSc, a professor of infectious diseases at the University of Copenhagen, and colleagues collected information on prescription medication, over-the-counter medication, and dietary supplements from adults living with HIV who received ART from two outpatient clinics.
The researchers estimated the prevalence of non-HIV comedications, and they used the University of Liverpool HIV Drug Interactions database to identify potential drug-drug interactions. They evaluated PDDIs and used logistic regression models to investigate links between PDDIs and relevant variables.
The study included 337 people living with HIV receiving ART. The median age was 53 years, 77% of them were male, and 96% were virally suppressed, with HIV-RNA viral load less than 50 copies/mL.
Overall, 26% of participants received five or more comedications, and 56% took dietary supplements.
In the medication lists of 52% of patients, the authors identified coadministration of drugs that required dose adjustment or monitoring; 4.5% of patients were taking drugs that should not be coadministered.
The researchers detected several factors that independently predicted PDDIs:
- Male sex (odds ratio, 1.9; 95% confidence interval, 1.0-3.4)
- Being on a protease inhibitor (OR, 4.3; 95% CI, 1.9-9.7)
- Receiving five or more comedications (OR, 3.3; 95% CI, 1.5-7.2)
- Taking over-the-counter medications (OR, 1.9; 95% CI, 1.1-3.3)
- Taking dietary supplements (OR, 2.0; 95% CI, 1.2-3.3)
Comorbidities and OTC medications increase in aging people with HIV
Indira Brar, MD, an infectious diseases senior staff physician and the medical director of HIV services at Henry Ford Health in Detroit, called the study and important resource for educating providers and patients about over-the-counter drugs.
“The main strength of the study is that it includes a decent number of aging patients living with HIV, the age group in which we worry about drug interactions,” she said in an interview.
“As patients get older, they have increased comorbidities. As comorbidities increase, the number of medications increases. As the number of medications increases, the drug interactions increase,” said Dr. Brar, who was not involved in the study. “Also, as patients get older, they tend to take more over-the-counter drugs.”
Dr. Brar explained how drug-drug interactions can harm patients.
“Drugs added to a patient who is already on ART could decrease the level of the ART and cause the patient to develop a drug-resistant HIV infection,” she said. “Or the ART the patient is on can increase the levels of the new drugs that have been added, and that could have potential toxicity and side effects.
“Food supplements, including multivitamins, calcium, and magnesium, are often overlooked because we think they’re benign. But these drugs can bind our new antiretrovirals, the integrase inhibitors. They can decrease their levels in the patient and cause drug-resistant HIV infection.
“In our clinic, we always tell our patients to please call us before they take any medication, so we can make sure there is no drug interaction,” Dr. Brar said.
Nan Wang, PharmD, a clinical pharmacy specialist at University Hospitals Cleveland Medical Center, noted in an email that drug-drug interactions with ARTs are common.
“Understanding the prevalence of antiretroviral drug interactions in a patient population can help identify certain medications that require enhanced vigilance and can guide our clinical interventions,” said Dr. Wang, who was not associated with the research.
Joseph Alvarnas, MD, a hematologist and oncologist at City of Hope Comprehensive Cancer Center in Duarte, Calif., said that this is “a methodologically sound and well-designed study that’s a timely, important reminder that providers need to think carefully and comprehensively when caring for their patients living with HIV.”
Dr. Alvarnas, who was not involved in the study, said that, with the widespread availability of ART, HIV has become a chronic, manageable condition in an aging population.
“ART agents, particularly the ritonavir-boosted protease inhibitors, increase the likelihood of patients having a potentially significant drug-drug interaction with one of their chronic care medications,” he added. “Even seemingly low-risk supplements such as multivitamins may result in a negative impact upon effective ART treatment of PLWH.”
“The essential next step is that these findings are integrated carefully into decision-support systems, electronic health record prescribing systems, and pharmacy safety-check systems to ensure that we reduce the risk of patient harm,” Dr. Alvarnas advised.
Dr. Benfield and several study coauthors reported financial relationships with GlaxoSmithKline and other pharmaceutical companies. Other coauthors, as well as Dr. Alvarnas, Dr. Brar, and Dr. Wang, reported no relevant financial relationships. The study was supported by GlaxoSmithKline.
A version of this article first appeared on Medscape.com.
Over-the-counter medications, food supplements, and other drugs may interact with antiretroviral therapy (ART) in people living with HIV and be harmful, an industry-sponsored clinical survey from Denmark reports.
“Our study confirms that polypharmacy and being on a protease inhibitor–based regimen increase the risk of potential drug-drug interactions [PDDIs] considerably and highlights the importance of questioning people living with HIV [PLWH] about dietary supplement intake,” the authors, led by Michaela Tinggaard, MD, Copenhagen University Hospital, wrote in HIV Medicine.
“Potential drug-drug interactions were common among our study population. Although the clinical significance of the majority of the identified PDDIs may be low, most of them were avoidable through a change or discontinuation of the comedication, a change in ART or by spacing drugs,” they added.
Senior author Thomas Benfield, MD, DTMH, DMSc, a professor of infectious diseases at the University of Copenhagen, and colleagues collected information on prescription medication, over-the-counter medication, and dietary supplements from adults living with HIV who received ART from two outpatient clinics.
The researchers estimated the prevalence of non-HIV comedications, and they used the University of Liverpool HIV Drug Interactions database to identify potential drug-drug interactions. They evaluated PDDIs and used logistic regression models to investigate links between PDDIs and relevant variables.
The study included 337 people living with HIV receiving ART. The median age was 53 years, 77% of them were male, and 96% were virally suppressed, with HIV-RNA viral load less than 50 copies/mL.
Overall, 26% of participants received five or more comedications, and 56% took dietary supplements.
In the medication lists of 52% of patients, the authors identified coadministration of drugs that required dose adjustment or monitoring; 4.5% of patients were taking drugs that should not be coadministered.
The researchers detected several factors that independently predicted PDDIs:
- Male sex (odds ratio, 1.9; 95% confidence interval, 1.0-3.4)
- Being on a protease inhibitor (OR, 4.3; 95% CI, 1.9-9.7)
- Receiving five or more comedications (OR, 3.3; 95% CI, 1.5-7.2)
- Taking over-the-counter medications (OR, 1.9; 95% CI, 1.1-3.3)
- Taking dietary supplements (OR, 2.0; 95% CI, 1.2-3.3)
Comorbidities and OTC medications increase in aging people with HIV
Indira Brar, MD, an infectious diseases senior staff physician and the medical director of HIV services at Henry Ford Health in Detroit, called the study and important resource for educating providers and patients about over-the-counter drugs.
“The main strength of the study is that it includes a decent number of aging patients living with HIV, the age group in which we worry about drug interactions,” she said in an interview.
“As patients get older, they have increased comorbidities. As comorbidities increase, the number of medications increases. As the number of medications increases, the drug interactions increase,” said Dr. Brar, who was not involved in the study. “Also, as patients get older, they tend to take more over-the-counter drugs.”
Dr. Brar explained how drug-drug interactions can harm patients.
“Drugs added to a patient who is already on ART could decrease the level of the ART and cause the patient to develop a drug-resistant HIV infection,” she said. “Or the ART the patient is on can increase the levels of the new drugs that have been added, and that could have potential toxicity and side effects.
“Food supplements, including multivitamins, calcium, and magnesium, are often overlooked because we think they’re benign. But these drugs can bind our new antiretrovirals, the integrase inhibitors. They can decrease their levels in the patient and cause drug-resistant HIV infection.
“In our clinic, we always tell our patients to please call us before they take any medication, so we can make sure there is no drug interaction,” Dr. Brar said.
Nan Wang, PharmD, a clinical pharmacy specialist at University Hospitals Cleveland Medical Center, noted in an email that drug-drug interactions with ARTs are common.
“Understanding the prevalence of antiretroviral drug interactions in a patient population can help identify certain medications that require enhanced vigilance and can guide our clinical interventions,” said Dr. Wang, who was not associated with the research.
Joseph Alvarnas, MD, a hematologist and oncologist at City of Hope Comprehensive Cancer Center in Duarte, Calif., said that this is “a methodologically sound and well-designed study that’s a timely, important reminder that providers need to think carefully and comprehensively when caring for their patients living with HIV.”
Dr. Alvarnas, who was not involved in the study, said that, with the widespread availability of ART, HIV has become a chronic, manageable condition in an aging population.
“ART agents, particularly the ritonavir-boosted protease inhibitors, increase the likelihood of patients having a potentially significant drug-drug interaction with one of their chronic care medications,” he added. “Even seemingly low-risk supplements such as multivitamins may result in a negative impact upon effective ART treatment of PLWH.”
“The essential next step is that these findings are integrated carefully into decision-support systems, electronic health record prescribing systems, and pharmacy safety-check systems to ensure that we reduce the risk of patient harm,” Dr. Alvarnas advised.
Dr. Benfield and several study coauthors reported financial relationships with GlaxoSmithKline and other pharmaceutical companies. Other coauthors, as well as Dr. Alvarnas, Dr. Brar, and Dr. Wang, reported no relevant financial relationships. The study was supported by GlaxoSmithKline.
A version of this article first appeared on Medscape.com.
Over-the-counter medications, food supplements, and other drugs may interact with antiretroviral therapy (ART) in people living with HIV and be harmful, an industry-sponsored clinical survey from Denmark reports.
“Our study confirms that polypharmacy and being on a protease inhibitor–based regimen increase the risk of potential drug-drug interactions [PDDIs] considerably and highlights the importance of questioning people living with HIV [PLWH] about dietary supplement intake,” the authors, led by Michaela Tinggaard, MD, Copenhagen University Hospital, wrote in HIV Medicine.
“Potential drug-drug interactions were common among our study population. Although the clinical significance of the majority of the identified PDDIs may be low, most of them were avoidable through a change or discontinuation of the comedication, a change in ART or by spacing drugs,” they added.
Senior author Thomas Benfield, MD, DTMH, DMSc, a professor of infectious diseases at the University of Copenhagen, and colleagues collected information on prescription medication, over-the-counter medication, and dietary supplements from adults living with HIV who received ART from two outpatient clinics.
The researchers estimated the prevalence of non-HIV comedications, and they used the University of Liverpool HIV Drug Interactions database to identify potential drug-drug interactions. They evaluated PDDIs and used logistic regression models to investigate links between PDDIs and relevant variables.
The study included 337 people living with HIV receiving ART. The median age was 53 years, 77% of them were male, and 96% were virally suppressed, with HIV-RNA viral load less than 50 copies/mL.
Overall, 26% of participants received five or more comedications, and 56% took dietary supplements.
In the medication lists of 52% of patients, the authors identified coadministration of drugs that required dose adjustment or monitoring; 4.5% of patients were taking drugs that should not be coadministered.
The researchers detected several factors that independently predicted PDDIs:
- Male sex (odds ratio, 1.9; 95% confidence interval, 1.0-3.4)
- Being on a protease inhibitor (OR, 4.3; 95% CI, 1.9-9.7)
- Receiving five or more comedications (OR, 3.3; 95% CI, 1.5-7.2)
- Taking over-the-counter medications (OR, 1.9; 95% CI, 1.1-3.3)
- Taking dietary supplements (OR, 2.0; 95% CI, 1.2-3.3)
Comorbidities and OTC medications increase in aging people with HIV
Indira Brar, MD, an infectious diseases senior staff physician and the medical director of HIV services at Henry Ford Health in Detroit, called the study and important resource for educating providers and patients about over-the-counter drugs.
“The main strength of the study is that it includes a decent number of aging patients living with HIV, the age group in which we worry about drug interactions,” she said in an interview.
“As patients get older, they have increased comorbidities. As comorbidities increase, the number of medications increases. As the number of medications increases, the drug interactions increase,” said Dr. Brar, who was not involved in the study. “Also, as patients get older, they tend to take more over-the-counter drugs.”
Dr. Brar explained how drug-drug interactions can harm patients.
“Drugs added to a patient who is already on ART could decrease the level of the ART and cause the patient to develop a drug-resistant HIV infection,” she said. “Or the ART the patient is on can increase the levels of the new drugs that have been added, and that could have potential toxicity and side effects.
“Food supplements, including multivitamins, calcium, and magnesium, are often overlooked because we think they’re benign. But these drugs can bind our new antiretrovirals, the integrase inhibitors. They can decrease their levels in the patient and cause drug-resistant HIV infection.
“In our clinic, we always tell our patients to please call us before they take any medication, so we can make sure there is no drug interaction,” Dr. Brar said.
Nan Wang, PharmD, a clinical pharmacy specialist at University Hospitals Cleveland Medical Center, noted in an email that drug-drug interactions with ARTs are common.
“Understanding the prevalence of antiretroviral drug interactions in a patient population can help identify certain medications that require enhanced vigilance and can guide our clinical interventions,” said Dr. Wang, who was not associated with the research.
Joseph Alvarnas, MD, a hematologist and oncologist at City of Hope Comprehensive Cancer Center in Duarte, Calif., said that this is “a methodologically sound and well-designed study that’s a timely, important reminder that providers need to think carefully and comprehensively when caring for their patients living with HIV.”
Dr. Alvarnas, who was not involved in the study, said that, with the widespread availability of ART, HIV has become a chronic, manageable condition in an aging population.
“ART agents, particularly the ritonavir-boosted protease inhibitors, increase the likelihood of patients having a potentially significant drug-drug interaction with one of their chronic care medications,” he added. “Even seemingly low-risk supplements such as multivitamins may result in a negative impact upon effective ART treatment of PLWH.”
“The essential next step is that these findings are integrated carefully into decision-support systems, electronic health record prescribing systems, and pharmacy safety-check systems to ensure that we reduce the risk of patient harm,” Dr. Alvarnas advised.
Dr. Benfield and several study coauthors reported financial relationships with GlaxoSmithKline and other pharmaceutical companies. Other coauthors, as well as Dr. Alvarnas, Dr. Brar, and Dr. Wang, reported no relevant financial relationships. The study was supported by GlaxoSmithKline.
A version of this article first appeared on Medscape.com.
FROM HIV MEDICINE