User login
Bugs on her skin—but nobody else sees them
CASE Scratching, anxious, and hopeless
Ms. L, age 74, who is paraplegic and uses a wheelchair, presents to our hospital’s emergency department (ED) accompanied by staff from the nursing home where she resides. She reports that she can feel and see bugs crawling all over her skin, biting
Ms. L experiences generalized pruritus with excoriations scattered over her upper and lower extremities and her trunk. She copes with the pruritus by scratching. She reports that the bugs are present throughout the day and are worse at night when she tries to go to bed. Nothing she does provides relief from the infestation. Earlier, at the nursing home, Ms. L had obtained a detergent powder and used it in an attempt to purge the bugs. She now has large swaths of irritated skin, mostly on her lower back and perineal region.
She says the bug infestation became unbearable 3 weeks ago, but she can’t identify any precipitants for her symptoms. Ms. L reports that the impact of the bugs on her daily activity, sleep, and quality of life is enormous. Despite her complaints, neither the nursing home staff nor the ED staff can find any evidence of bugs on Ms. L’s clothes or skin.
Because Ms. L resorted to such drastic measures in her attempt to rid her body of the bugs, she is considered a safety risk and is admitted to the psychiatric unit, although she vehemently denies any intention to harm herself.
On the psychiatric unit, Ms. L states that the infestation began approximately 2 years ago. She began to experience severe worsening of her symptoms a few weeks before presenting to the ED.
During evaluation, Ms. L is alert and oriented to person, place, and situation. She is also quite cooperative but guarded in describing her infestation. There is some degree of suspiciousness and paranoia with regards to her infestation; she is very sensitive to how the clinical staff respond to her condition. She appears worried, and exhibits anxiety, sadness, hopelessness, and tearfulness. Her thought process is goal-directed, but preoccupied by the bugs.
[polldaddy:10064801]
Continue to: The authors' observations
The authors’ observations
Delusional parasitosis is a rare disorder that is defined by an individual having a fixed, false belief that he or she is being infected or grossly invaded by a living organism. Karl A. Ekbom, a Swedish neurologist, was the first practitioner to definitively describe this affliction in 1938.1
Primary delusional parasitosis is a disease defined by this single psychotic symptom without other classic symptoms of schizophrenia; this single symptom cannot be attributed to the effects of substance abuse or a medical condition. Many affected patients remain functional in their daily lives; only a minority of patients experience delusions that interfere with usual activity.2 Secondary delusional parasitosis is a symptom of another psychiatric or medical disease.
Morgellons disease is characterized by symptoms similar to primary delusional parasitosis, but symptoms of this condition also include the delusional belief that inanimate objects, usually fibers, are in the skin as well as the parasites.3
A population-based study among individuals living in Olmsted County, Minnesota from 1976 to 2010 found that the incidence of delusional infestation was 1.9 cases (95% confidence interval, 1.5 to 2.4) per 100,000 person-years.4 In a retrospective study of 147 patients with delusional parasitosis, 33% of these patients described themselves as disabled, 28% were retired, and 26% were employed.5 In this study, the mean age of diagnosis was 57, with a female-to-male ratio of 2.89:1.5
Continue to: HISTORY Prior psychiatric hospitalization
HISTORY Prior psychiatric hospitalization
Ms. L, who is divorced and retired, lives in a nursing home and has no pets, no exposure to scabies, no recent travel, no allergies, and no difficulty with her hygiene except at the peak of her illness. She denies any alcohol or illicit drug use but reports a 6 pack year history of smoking. She has a son, 2 grandchildren, and 2 great grandchildren who all live in town and see her regularly. She reports no history of arrests or legal problems.
Ms. L has a history of depression and anxiety that culminated in a “nervous breakdown” in 1985 with a brief stay in a psychiatric hospital. She reports that she had seen a therapist for 6 years as part of her treatment following that event. During her hospitalization, she was treated with a tricyclic antidepressant and received electroconvulsive therapy. She denies being suicidal during the incident in 1985 or at any point in time before or since then. She now takes venlafaxine, 75 mg/d, for depression and anxiety.
Ms. L’s paraplegia resulted from her sixth corrective surgery for scoliosis, which occurred 6 years ago. She has had chronic pain since this surgery. Her medical history also includes hypertension, atrial fibrillation, mild neurocognitive changes, and gastroesophageal reflux disease.
EVALUATION Skin examination, blood analysis normal
On admission, Ms. L undergoes a skin examination, which yields no evidence consistent with infestation with Pediculus humanus corporis (body louse) or Sarcoptes scabiei (scabies).6 Blood analysis shows no iron deficiency, renal failure, hyperbilirubinemia, or eosinophilia. In the ED, the medical team examines Ms. L and explores other medical and dermatological causes of her condition. Because dermatological causes had been ruled out before Ms. L was admitted to the inpatient psychiatric unit, no dermatology consult is requested.
Continue to: TREATMENT A first-generation antipsychotic
TREATMENT A first-generation antipsychotic
When Ms. L is admitted to the psychiatric unit, she is started
During the week, Ms. L’s perphenazine is titrated up to 24 mg twice daily and venlafaxine is titrated to 150 mg/d. A Montreal Cognitive Assessment (MoCA) is performed within the first 2 days of admission and she scores 16/30, indicating moderate cognitive impairment. On Friday, the attending physician explains that her medications should start to have therapeutic effect. During this time, this clinician engages in cognitive restructuring by providing validation of Ms. L’s suffering, verbal support, and medication compliance counseling. At this time, the treating team also suggests to Ms. L that she should expect the activity and effects of the bugs to dissipate. She is receptive to this suggestion. She also participates in the milieu, including unit activities, but is limited in her ability to engage in group therapy due to the intensity of her illness.
Throughout the weekend, the on-call physician also engages Ms. L and reports minor improvement.
OUTCOME Significant relief
On re-evaluation Monday morning—almost a week after Ms. L had been admitted to the inpatient psychiatric unit—she has achieved significant relief from her delusions. She says that she has no idea where the bugs have gone. Ms. L appears to be a completely different person. She no longer appears guarded. The suspiciousness, paranoia, hopelessness, and negative outlook she previously experienced have significantly diminished. Her MoCA score improves to 25/30, indicating no cognitive impairment (Table). She is discharged after a 7-night stay on the inpatient psychiatric unit.
Continue to: The authors' observations
The authors’ observations
During one of the clinical multidisciplinary treatment team meetings held for Ms. L, it was initially estimated that it would take at least 2 weeks for the delusional parasitosis to significantly respond to antipsychotic therapy. However, it is our professional opinion that the applied cognitive restructuring, with validation of her suffering, verbal support, and medication adherence counseling, expedited her recovery. This coincided with the aggressive titration of her antipsychotic and antidepressant, although the treatment team’s acknowledgment of Ms. L’s misery appeared to lower her guard and make her more susceptible to the power of cognitive restructuring. The efforts to validate the patient’s feelings and decrease hopelessness by telling her that the medication would make the bugs go away appeared to be the tipping point for her recovery. Patients with primary delusional parasitosis often are guarded and may feel alone in their predicament when they are met with perplexed responses from individuals with whom they discuss their symptoms. Compared with patients with schizophrenia, patients with delusional parasitosis maintain normal cognitive functioning, which may give them the insight to understand how their experience may be perceived as incompatible with reality.7 This understanding, coupled with some perceived helplessness, can lead a patient to fear having a severe mental decompensation, which can contribute to a delayed or complicated recovery.
The cognitive process described above might have been responsible for the difference in Ms. L’s MoCA scores because her performance in the initial test was hindered by her constant obsession with the bugs, which made her distracted during the test. By the time she responded to treatment, she gained significant clarity of thought, which enabled her to perform optimally in the test.
The difficulty in treating patients with delusional parasitosis may be further affected by lack of insight, and the fact that they often do not present to a psychiatrist for treatment in a timely manner because their delusion is impregnable and presents them with an alternate reality. These patients are more likely to seek out primary care physicians, dermatologists, infectious disease doctors, and entomologists because of the fervor of their delusion and the intensity of their discomfort. Because of this, a collaboration between these providers would likely lead to improved care and treatment acceptance for patients with delusional parasitosis.
Antipsychotics are the preferred medication for treating delusional parasitosis, and the literature supports their use for this purpose.6,8 The overall response rate is 60% to 100%.6 Previously, in small placebo-controlled trials, the first-generation antipsychotic (FGA) pimozide was considered first-line treatment for this disease.6 However, this antipsychotic is no longer favored because evidence is mounting that other FGAs result in comparable response rates with fewer tolerability issues.8,9
The bulk of data on the use of antipsychotics for treating delusional parasitosis comes from retrospective case reports and case series.6 Multiple antipsychotics have been shown to be effective in treating delusional parasitosis, including both FGAs and second-generation antipsychotics (SGAs).6,10 Published case reports and series have shown the effectiveness of the FGAs
Continue to: The SGAs risperidone, olanzapine, aripiprazole...
The SGAs
When selecting antipsychotic therapy for a patient diagnosed with delusional parasitosis, consider patient-specific factors, such as age, medication history, comorbidities, and the adverse-effect profile of the medication(s). These medications should be started at a low dose and titrated based on efficacy and safety. The optimal duration of therapy varies by patient. Patients should continually be assessed for possible treatment discontinuation, although if therapy is tapered off, patients need to be closely monitored for possible relapse or recurrence of symptoms.
Ms. L received perphenazine titrated up to 24 mg/d for the treatment of delusional parasitosis. The maximum dose used for Ms. L was higher than those used in previous reports, although she appeared to tolerate the medication well and respond rapidly. Her symptoms showed improvement within 1 week. Importantly, in published case reports, patients have been resistant to the use of psychotropic medications without other treatment modalities (eg, psychotherapy, various behavioral approaches). We conclude that Ms. L’s response was attributable to the use of the combination of psychotherapeutic techniques and the effectiveness of perphenazine and venlafaxine.
Bottom Line
Managing patients with primary delusional parasitosis can be challenging due to the fixed nature of the delusion. A combination of antipsychotics and psychotherapeutic techniques can benefit some patients. The optimal duration of treatment varies by patient.
Related Resource
- Trenton A, Pansare N, Tobia A, et al. Delusional parasitosis on the psychiatric consultation service-a longitudinal perspective: case study. BJPsych Open. 2017;3(3):154-158.
Drug Brand Names
Aripiprazole • Abilify
Haloperidol • Haldol
Olanzapine • Zyprexa
Paliperidone • Invega
Paliperidone palmitate • Invega Sustenna
Perphenazine • Trilafon
Pimozide • Orap
Quetiapine • Seroquel
Risperidone • Risperdal
Venlafaxine • Effexor
Ziprasidone • Geodon
1. Ekbom KA. Der präsenile dermatozoenwahn [in Swedish]. Acta Psychiatr Neurol Scand. 1938;13(3):227-259.
2. Lynch PJ. Delusions of parasitosis. Semin Dermatol. 1993;12(1):39-45.
3. Middelveen MJ, Fesler MC, Stricker RB. History of Morgellons disease: from delusion to definition. Clin Cosmet Investig Dermatol. 2018;11:71-90.
4. Bailey CH, Andersen LK, Lowe GC, et al. A population-based study of the incidence of delusional infestation in Olmsted County, Minnesota, 1976–2010. Br J Dermatol. 2014;170(5):1130-1135.
5. Foster AA, Hylwa SA, Bury JE, et al. Delusional infestation: clinical presentation in 147 patients seen at Mayo Clinic. J Am Acad Dermatol. 2012;67(4):673.e1-e10.
6. Lepping P, Russell I, Freudenmann RW. Antipsychotic treatment of primary delusional parasitosis: systematic review. Br J Psychiatry. 2007;191(3):198-205.
7. Freudenmann RW, Lepping P. Delusional infestation. Clin Microbiol Rev. 2009;22(4):690-732.
8. Mercan S, Altunay IK, Taskintuna N, et al. Atypical antipsychotic drugs in the treatment of delusional parasitosis. Intl J Psychiatry Med. 2007:37(1):29-37.
9. Trabert W. 100 years of delusional parasitosis. Meta-analysis of 1,223 case reports. Psychopathology. 1995;28(5):238-246.
10. Freudenmann RW, Lepping P. Second-generation antipsychotics in primary and secondary delusional parasitosis. J Clin Psychopharmacol. 2008;28(5):500-508.
11. Boggild AK, Nicks BA, Yen L, et al. Delusional parasitosis: six-year experience with 23 consecutive cases at an academic medical center. Int J Infect Dis. 2010;14(4):e317-e321.
CASE Scratching, anxious, and hopeless
Ms. L, age 74, who is paraplegic and uses a wheelchair, presents to our hospital’s emergency department (ED) accompanied by staff from the nursing home where she resides. She reports that she can feel and see bugs crawling all over her skin, biting
Ms. L experiences generalized pruritus with excoriations scattered over her upper and lower extremities and her trunk. She copes with the pruritus by scratching. She reports that the bugs are present throughout the day and are worse at night when she tries to go to bed. Nothing she does provides relief from the infestation. Earlier, at the nursing home, Ms. L had obtained a detergent powder and used it in an attempt to purge the bugs. She now has large swaths of irritated skin, mostly on her lower back and perineal region.
She says the bug infestation became unbearable 3 weeks ago, but she can’t identify any precipitants for her symptoms. Ms. L reports that the impact of the bugs on her daily activity, sleep, and quality of life is enormous. Despite her complaints, neither the nursing home staff nor the ED staff can find any evidence of bugs on Ms. L’s clothes or skin.
Because Ms. L resorted to such drastic measures in her attempt to rid her body of the bugs, she is considered a safety risk and is admitted to the psychiatric unit, although she vehemently denies any intention to harm herself.
On the psychiatric unit, Ms. L states that the infestation began approximately 2 years ago. She began to experience severe worsening of her symptoms a few weeks before presenting to the ED.
During evaluation, Ms. L is alert and oriented to person, place, and situation. She is also quite cooperative but guarded in describing her infestation. There is some degree of suspiciousness and paranoia with regards to her infestation; she is very sensitive to how the clinical staff respond to her condition. She appears worried, and exhibits anxiety, sadness, hopelessness, and tearfulness. Her thought process is goal-directed, but preoccupied by the bugs.
[polldaddy:10064801]
Continue to: The authors' observations
The authors’ observations
Delusional parasitosis is a rare disorder that is defined by an individual having a fixed, false belief that he or she is being infected or grossly invaded by a living organism. Karl A. Ekbom, a Swedish neurologist, was the first practitioner to definitively describe this affliction in 1938.1
Primary delusional parasitosis is a disease defined by this single psychotic symptom without other classic symptoms of schizophrenia; this single symptom cannot be attributed to the effects of substance abuse or a medical condition. Many affected patients remain functional in their daily lives; only a minority of patients experience delusions that interfere with usual activity.2 Secondary delusional parasitosis is a symptom of another psychiatric or medical disease.
Morgellons disease is characterized by symptoms similar to primary delusional parasitosis, but symptoms of this condition also include the delusional belief that inanimate objects, usually fibers, are in the skin as well as the parasites.3
A population-based study among individuals living in Olmsted County, Minnesota from 1976 to 2010 found that the incidence of delusional infestation was 1.9 cases (95% confidence interval, 1.5 to 2.4) per 100,000 person-years.4 In a retrospective study of 147 patients with delusional parasitosis, 33% of these patients described themselves as disabled, 28% were retired, and 26% were employed.5 In this study, the mean age of diagnosis was 57, with a female-to-male ratio of 2.89:1.5
Continue to: HISTORY Prior psychiatric hospitalization
HISTORY Prior psychiatric hospitalization
Ms. L, who is divorced and retired, lives in a nursing home and has no pets, no exposure to scabies, no recent travel, no allergies, and no difficulty with her hygiene except at the peak of her illness. She denies any alcohol or illicit drug use but reports a 6 pack year history of smoking. She has a son, 2 grandchildren, and 2 great grandchildren who all live in town and see her regularly. She reports no history of arrests or legal problems.
Ms. L has a history of depression and anxiety that culminated in a “nervous breakdown” in 1985 with a brief stay in a psychiatric hospital. She reports that she had seen a therapist for 6 years as part of her treatment following that event. During her hospitalization, she was treated with a tricyclic antidepressant and received electroconvulsive therapy. She denies being suicidal during the incident in 1985 or at any point in time before or since then. She now takes venlafaxine, 75 mg/d, for depression and anxiety.
Ms. L’s paraplegia resulted from her sixth corrective surgery for scoliosis, which occurred 6 years ago. She has had chronic pain since this surgery. Her medical history also includes hypertension, atrial fibrillation, mild neurocognitive changes, and gastroesophageal reflux disease.
EVALUATION Skin examination, blood analysis normal
On admission, Ms. L undergoes a skin examination, which yields no evidence consistent with infestation with Pediculus humanus corporis (body louse) or Sarcoptes scabiei (scabies).6 Blood analysis shows no iron deficiency, renal failure, hyperbilirubinemia, or eosinophilia. In the ED, the medical team examines Ms. L and explores other medical and dermatological causes of her condition. Because dermatological causes had been ruled out before Ms. L was admitted to the inpatient psychiatric unit, no dermatology consult is requested.
Continue to: TREATMENT A first-generation antipsychotic
TREATMENT A first-generation antipsychotic
When Ms. L is admitted to the psychiatric unit, she is started
During the week, Ms. L’s perphenazine is titrated up to 24 mg twice daily and venlafaxine is titrated to 150 mg/d. A Montreal Cognitive Assessment (MoCA) is performed within the first 2 days of admission and she scores 16/30, indicating moderate cognitive impairment. On Friday, the attending physician explains that her medications should start to have therapeutic effect. During this time, this clinician engages in cognitive restructuring by providing validation of Ms. L’s suffering, verbal support, and medication compliance counseling. At this time, the treating team also suggests to Ms. L that she should expect the activity and effects of the bugs to dissipate. She is receptive to this suggestion. She also participates in the milieu, including unit activities, but is limited in her ability to engage in group therapy due to the intensity of her illness.
Throughout the weekend, the on-call physician also engages Ms. L and reports minor improvement.
OUTCOME Significant relief
On re-evaluation Monday morning—almost a week after Ms. L had been admitted to the inpatient psychiatric unit—she has achieved significant relief from her delusions. She says that she has no idea where the bugs have gone. Ms. L appears to be a completely different person. She no longer appears guarded. The suspiciousness, paranoia, hopelessness, and negative outlook she previously experienced have significantly diminished. Her MoCA score improves to 25/30, indicating no cognitive impairment (Table). She is discharged after a 7-night stay on the inpatient psychiatric unit.
Continue to: The authors' observations
The authors’ observations
During one of the clinical multidisciplinary treatment team meetings held for Ms. L, it was initially estimated that it would take at least 2 weeks for the delusional parasitosis to significantly respond to antipsychotic therapy. However, it is our professional opinion that the applied cognitive restructuring, with validation of her suffering, verbal support, and medication adherence counseling, expedited her recovery. This coincided with the aggressive titration of her antipsychotic and antidepressant, although the treatment team’s acknowledgment of Ms. L’s misery appeared to lower her guard and make her more susceptible to the power of cognitive restructuring. The efforts to validate the patient’s feelings and decrease hopelessness by telling her that the medication would make the bugs go away appeared to be the tipping point for her recovery. Patients with primary delusional parasitosis often are guarded and may feel alone in their predicament when they are met with perplexed responses from individuals with whom they discuss their symptoms. Compared with patients with schizophrenia, patients with delusional parasitosis maintain normal cognitive functioning, which may give them the insight to understand how their experience may be perceived as incompatible with reality.7 This understanding, coupled with some perceived helplessness, can lead a patient to fear having a severe mental decompensation, which can contribute to a delayed or complicated recovery.
The cognitive process described above might have been responsible for the difference in Ms. L’s MoCA scores because her performance in the initial test was hindered by her constant obsession with the bugs, which made her distracted during the test. By the time she responded to treatment, she gained significant clarity of thought, which enabled her to perform optimally in the test.
The difficulty in treating patients with delusional parasitosis may be further affected by lack of insight, and the fact that they often do not present to a psychiatrist for treatment in a timely manner because their delusion is impregnable and presents them with an alternate reality. These patients are more likely to seek out primary care physicians, dermatologists, infectious disease doctors, and entomologists because of the fervor of their delusion and the intensity of their discomfort. Because of this, a collaboration between these providers would likely lead to improved care and treatment acceptance for patients with delusional parasitosis.
Antipsychotics are the preferred medication for treating delusional parasitosis, and the literature supports their use for this purpose.6,8 The overall response rate is 60% to 100%.6 Previously, in small placebo-controlled trials, the first-generation antipsychotic (FGA) pimozide was considered first-line treatment for this disease.6 However, this antipsychotic is no longer favored because evidence is mounting that other FGAs result in comparable response rates with fewer tolerability issues.8,9
The bulk of data on the use of antipsychotics for treating delusional parasitosis comes from retrospective case reports and case series.6 Multiple antipsychotics have been shown to be effective in treating delusional parasitosis, including both FGAs and second-generation antipsychotics (SGAs).6,10 Published case reports and series have shown the effectiveness of the FGAs
Continue to: The SGAs risperidone, olanzapine, aripiprazole...
The SGAs
When selecting antipsychotic therapy for a patient diagnosed with delusional parasitosis, consider patient-specific factors, such as age, medication history, comorbidities, and the adverse-effect profile of the medication(s). These medications should be started at a low dose and titrated based on efficacy and safety. The optimal duration of therapy varies by patient. Patients should continually be assessed for possible treatment discontinuation, although if therapy is tapered off, patients need to be closely monitored for possible relapse or recurrence of symptoms.
Ms. L received perphenazine titrated up to 24 mg/d for the treatment of delusional parasitosis. The maximum dose used for Ms. L was higher than those used in previous reports, although she appeared to tolerate the medication well and respond rapidly. Her symptoms showed improvement within 1 week. Importantly, in published case reports, patients have been resistant to the use of psychotropic medications without other treatment modalities (eg, psychotherapy, various behavioral approaches). We conclude that Ms. L’s response was attributable to the use of the combination of psychotherapeutic techniques and the effectiveness of perphenazine and venlafaxine.
Bottom Line
Managing patients with primary delusional parasitosis can be challenging due to the fixed nature of the delusion. A combination of antipsychotics and psychotherapeutic techniques can benefit some patients. The optimal duration of treatment varies by patient.
Related Resource
- Trenton A, Pansare N, Tobia A, et al. Delusional parasitosis on the psychiatric consultation service-a longitudinal perspective: case study. BJPsych Open. 2017;3(3):154-158.
Drug Brand Names
Aripiprazole • Abilify
Haloperidol • Haldol
Olanzapine • Zyprexa
Paliperidone • Invega
Paliperidone palmitate • Invega Sustenna
Perphenazine • Trilafon
Pimozide • Orap
Quetiapine • Seroquel
Risperidone • Risperdal
Venlafaxine • Effexor
Ziprasidone • Geodon
CASE Scratching, anxious, and hopeless
Ms. L, age 74, who is paraplegic and uses a wheelchair, presents to our hospital’s emergency department (ED) accompanied by staff from the nursing home where she resides. She reports that she can feel and see bugs crawling all over her skin, biting
Ms. L experiences generalized pruritus with excoriations scattered over her upper and lower extremities and her trunk. She copes with the pruritus by scratching. She reports that the bugs are present throughout the day and are worse at night when she tries to go to bed. Nothing she does provides relief from the infestation. Earlier, at the nursing home, Ms. L had obtained a detergent powder and used it in an attempt to purge the bugs. She now has large swaths of irritated skin, mostly on her lower back and perineal region.
She says the bug infestation became unbearable 3 weeks ago, but she can’t identify any precipitants for her symptoms. Ms. L reports that the impact of the bugs on her daily activity, sleep, and quality of life is enormous. Despite her complaints, neither the nursing home staff nor the ED staff can find any evidence of bugs on Ms. L’s clothes or skin.
Because Ms. L resorted to such drastic measures in her attempt to rid her body of the bugs, she is considered a safety risk and is admitted to the psychiatric unit, although she vehemently denies any intention to harm herself.
On the psychiatric unit, Ms. L states that the infestation began approximately 2 years ago. She began to experience severe worsening of her symptoms a few weeks before presenting to the ED.
During evaluation, Ms. L is alert and oriented to person, place, and situation. She is also quite cooperative but guarded in describing her infestation. There is some degree of suspiciousness and paranoia with regards to her infestation; she is very sensitive to how the clinical staff respond to her condition. She appears worried, and exhibits anxiety, sadness, hopelessness, and tearfulness. Her thought process is goal-directed, but preoccupied by the bugs.
[polldaddy:10064801]
Continue to: The authors' observations
The authors’ observations
Delusional parasitosis is a rare disorder that is defined by an individual having a fixed, false belief that he or she is being infected or grossly invaded by a living organism. Karl A. Ekbom, a Swedish neurologist, was the first practitioner to definitively describe this affliction in 1938.1
Primary delusional parasitosis is a disease defined by this single psychotic symptom without other classic symptoms of schizophrenia; this single symptom cannot be attributed to the effects of substance abuse or a medical condition. Many affected patients remain functional in their daily lives; only a minority of patients experience delusions that interfere with usual activity.2 Secondary delusional parasitosis is a symptom of another psychiatric or medical disease.
Morgellons disease is characterized by symptoms similar to primary delusional parasitosis, but symptoms of this condition also include the delusional belief that inanimate objects, usually fibers, are in the skin as well as the parasites.3
A population-based study among individuals living in Olmsted County, Minnesota from 1976 to 2010 found that the incidence of delusional infestation was 1.9 cases (95% confidence interval, 1.5 to 2.4) per 100,000 person-years.4 In a retrospective study of 147 patients with delusional parasitosis, 33% of these patients described themselves as disabled, 28% were retired, and 26% were employed.5 In this study, the mean age of diagnosis was 57, with a female-to-male ratio of 2.89:1.5
Continue to: HISTORY Prior psychiatric hospitalization
HISTORY Prior psychiatric hospitalization
Ms. L, who is divorced and retired, lives in a nursing home and has no pets, no exposure to scabies, no recent travel, no allergies, and no difficulty with her hygiene except at the peak of her illness. She denies any alcohol or illicit drug use but reports a 6 pack year history of smoking. She has a son, 2 grandchildren, and 2 great grandchildren who all live in town and see her regularly. She reports no history of arrests or legal problems.
Ms. L has a history of depression and anxiety that culminated in a “nervous breakdown” in 1985 with a brief stay in a psychiatric hospital. She reports that she had seen a therapist for 6 years as part of her treatment following that event. During her hospitalization, she was treated with a tricyclic antidepressant and received electroconvulsive therapy. She denies being suicidal during the incident in 1985 or at any point in time before or since then. She now takes venlafaxine, 75 mg/d, for depression and anxiety.
Ms. L’s paraplegia resulted from her sixth corrective surgery for scoliosis, which occurred 6 years ago. She has had chronic pain since this surgery. Her medical history also includes hypertension, atrial fibrillation, mild neurocognitive changes, and gastroesophageal reflux disease.
EVALUATION Skin examination, blood analysis normal
On admission, Ms. L undergoes a skin examination, which yields no evidence consistent with infestation with Pediculus humanus corporis (body louse) or Sarcoptes scabiei (scabies).6 Blood analysis shows no iron deficiency, renal failure, hyperbilirubinemia, or eosinophilia. In the ED, the medical team examines Ms. L and explores other medical and dermatological causes of her condition. Because dermatological causes had been ruled out before Ms. L was admitted to the inpatient psychiatric unit, no dermatology consult is requested.
Continue to: TREATMENT A first-generation antipsychotic
TREATMENT A first-generation antipsychotic
When Ms. L is admitted to the psychiatric unit, she is started
During the week, Ms. L’s perphenazine is titrated up to 24 mg twice daily and venlafaxine is titrated to 150 mg/d. A Montreal Cognitive Assessment (MoCA) is performed within the first 2 days of admission and she scores 16/30, indicating moderate cognitive impairment. On Friday, the attending physician explains that her medications should start to have therapeutic effect. During this time, this clinician engages in cognitive restructuring by providing validation of Ms. L’s suffering, verbal support, and medication compliance counseling. At this time, the treating team also suggests to Ms. L that she should expect the activity and effects of the bugs to dissipate. She is receptive to this suggestion. She also participates in the milieu, including unit activities, but is limited in her ability to engage in group therapy due to the intensity of her illness.
Throughout the weekend, the on-call physician also engages Ms. L and reports minor improvement.
OUTCOME Significant relief
On re-evaluation Monday morning—almost a week after Ms. L had been admitted to the inpatient psychiatric unit—she has achieved significant relief from her delusions. She says that she has no idea where the bugs have gone. Ms. L appears to be a completely different person. She no longer appears guarded. The suspiciousness, paranoia, hopelessness, and negative outlook she previously experienced have significantly diminished. Her MoCA score improves to 25/30, indicating no cognitive impairment (Table). She is discharged after a 7-night stay on the inpatient psychiatric unit.
Continue to: The authors' observations
The authors’ observations
During one of the clinical multidisciplinary treatment team meetings held for Ms. L, it was initially estimated that it would take at least 2 weeks for the delusional parasitosis to significantly respond to antipsychotic therapy. However, it is our professional opinion that the applied cognitive restructuring, with validation of her suffering, verbal support, and medication adherence counseling, expedited her recovery. This coincided with the aggressive titration of her antipsychotic and antidepressant, although the treatment team’s acknowledgment of Ms. L’s misery appeared to lower her guard and make her more susceptible to the power of cognitive restructuring. The efforts to validate the patient’s feelings and decrease hopelessness by telling her that the medication would make the bugs go away appeared to be the tipping point for her recovery. Patients with primary delusional parasitosis often are guarded and may feel alone in their predicament when they are met with perplexed responses from individuals with whom they discuss their symptoms. Compared with patients with schizophrenia, patients with delusional parasitosis maintain normal cognitive functioning, which may give them the insight to understand how their experience may be perceived as incompatible with reality.7 This understanding, coupled with some perceived helplessness, can lead a patient to fear having a severe mental decompensation, which can contribute to a delayed or complicated recovery.
The cognitive process described above might have been responsible for the difference in Ms. L’s MoCA scores because her performance in the initial test was hindered by her constant obsession with the bugs, which made her distracted during the test. By the time she responded to treatment, she gained significant clarity of thought, which enabled her to perform optimally in the test.
The difficulty in treating patients with delusional parasitosis may be further affected by lack of insight, and the fact that they often do not present to a psychiatrist for treatment in a timely manner because their delusion is impregnable and presents them with an alternate reality. These patients are more likely to seek out primary care physicians, dermatologists, infectious disease doctors, and entomologists because of the fervor of their delusion and the intensity of their discomfort. Because of this, a collaboration between these providers would likely lead to improved care and treatment acceptance for patients with delusional parasitosis.
Antipsychotics are the preferred medication for treating delusional parasitosis, and the literature supports their use for this purpose.6,8 The overall response rate is 60% to 100%.6 Previously, in small placebo-controlled trials, the first-generation antipsychotic (FGA) pimozide was considered first-line treatment for this disease.6 However, this antipsychotic is no longer favored because evidence is mounting that other FGAs result in comparable response rates with fewer tolerability issues.8,9
The bulk of data on the use of antipsychotics for treating delusional parasitosis comes from retrospective case reports and case series.6 Multiple antipsychotics have been shown to be effective in treating delusional parasitosis, including both FGAs and second-generation antipsychotics (SGAs).6,10 Published case reports and series have shown the effectiveness of the FGAs
Continue to: The SGAs risperidone, olanzapine, aripiprazole...
The SGAs
When selecting antipsychotic therapy for a patient diagnosed with delusional parasitosis, consider patient-specific factors, such as age, medication history, comorbidities, and the adverse-effect profile of the medication(s). These medications should be started at a low dose and titrated based on efficacy and safety. The optimal duration of therapy varies by patient. Patients should continually be assessed for possible treatment discontinuation, although if therapy is tapered off, patients need to be closely monitored for possible relapse or recurrence of symptoms.
Ms. L received perphenazine titrated up to 24 mg/d for the treatment of delusional parasitosis. The maximum dose used for Ms. L was higher than those used in previous reports, although she appeared to tolerate the medication well and respond rapidly. Her symptoms showed improvement within 1 week. Importantly, in published case reports, patients have been resistant to the use of psychotropic medications without other treatment modalities (eg, psychotherapy, various behavioral approaches). We conclude that Ms. L’s response was attributable to the use of the combination of psychotherapeutic techniques and the effectiveness of perphenazine and venlafaxine.
Bottom Line
Managing patients with primary delusional parasitosis can be challenging due to the fixed nature of the delusion. A combination of antipsychotics and psychotherapeutic techniques can benefit some patients. The optimal duration of treatment varies by patient.
Related Resource
- Trenton A, Pansare N, Tobia A, et al. Delusional parasitosis on the psychiatric consultation service-a longitudinal perspective: case study. BJPsych Open. 2017;3(3):154-158.
Drug Brand Names
Aripiprazole • Abilify
Haloperidol • Haldol
Olanzapine • Zyprexa
Paliperidone • Invega
Paliperidone palmitate • Invega Sustenna
Perphenazine • Trilafon
Pimozide • Orap
Quetiapine • Seroquel
Risperidone • Risperdal
Venlafaxine • Effexor
Ziprasidone • Geodon
1. Ekbom KA. Der präsenile dermatozoenwahn [in Swedish]. Acta Psychiatr Neurol Scand. 1938;13(3):227-259.
2. Lynch PJ. Delusions of parasitosis. Semin Dermatol. 1993;12(1):39-45.
3. Middelveen MJ, Fesler MC, Stricker RB. History of Morgellons disease: from delusion to definition. Clin Cosmet Investig Dermatol. 2018;11:71-90.
4. Bailey CH, Andersen LK, Lowe GC, et al. A population-based study of the incidence of delusional infestation in Olmsted County, Minnesota, 1976–2010. Br J Dermatol. 2014;170(5):1130-1135.
5. Foster AA, Hylwa SA, Bury JE, et al. Delusional infestation: clinical presentation in 147 patients seen at Mayo Clinic. J Am Acad Dermatol. 2012;67(4):673.e1-e10.
6. Lepping P, Russell I, Freudenmann RW. Antipsychotic treatment of primary delusional parasitosis: systematic review. Br J Psychiatry. 2007;191(3):198-205.
7. Freudenmann RW, Lepping P. Delusional infestation. Clin Microbiol Rev. 2009;22(4):690-732.
8. Mercan S, Altunay IK, Taskintuna N, et al. Atypical antipsychotic drugs in the treatment of delusional parasitosis. Intl J Psychiatry Med. 2007:37(1):29-37.
9. Trabert W. 100 years of delusional parasitosis. Meta-analysis of 1,223 case reports. Psychopathology. 1995;28(5):238-246.
10. Freudenmann RW, Lepping P. Second-generation antipsychotics in primary and secondary delusional parasitosis. J Clin Psychopharmacol. 2008;28(5):500-508.
11. Boggild AK, Nicks BA, Yen L, et al. Delusional parasitosis: six-year experience with 23 consecutive cases at an academic medical center. Int J Infect Dis. 2010;14(4):e317-e321.
1. Ekbom KA. Der präsenile dermatozoenwahn [in Swedish]. Acta Psychiatr Neurol Scand. 1938;13(3):227-259.
2. Lynch PJ. Delusions of parasitosis. Semin Dermatol. 1993;12(1):39-45.
3. Middelveen MJ, Fesler MC, Stricker RB. History of Morgellons disease: from delusion to definition. Clin Cosmet Investig Dermatol. 2018;11:71-90.
4. Bailey CH, Andersen LK, Lowe GC, et al. A population-based study of the incidence of delusional infestation in Olmsted County, Minnesota, 1976–2010. Br J Dermatol. 2014;170(5):1130-1135.
5. Foster AA, Hylwa SA, Bury JE, et al. Delusional infestation: clinical presentation in 147 patients seen at Mayo Clinic. J Am Acad Dermatol. 2012;67(4):673.e1-e10.
6. Lepping P, Russell I, Freudenmann RW. Antipsychotic treatment of primary delusional parasitosis: systematic review. Br J Psychiatry. 2007;191(3):198-205.
7. Freudenmann RW, Lepping P. Delusional infestation. Clin Microbiol Rev. 2009;22(4):690-732.
8. Mercan S, Altunay IK, Taskintuna N, et al. Atypical antipsychotic drugs in the treatment of delusional parasitosis. Intl J Psychiatry Med. 2007:37(1):29-37.
9. Trabert W. 100 years of delusional parasitosis. Meta-analysis of 1,223 case reports. Psychopathology. 1995;28(5):238-246.
10. Freudenmann RW, Lepping P. Second-generation antipsychotics in primary and secondary delusional parasitosis. J Clin Psychopharmacol. 2008;28(5):500-508.
11. Boggild AK, Nicks BA, Yen L, et al. Delusional parasitosis: six-year experience with 23 consecutive cases at an academic medical center. Int J Infect Dis. 2010;14(4):e317-e321.
Prescribing psychotropics to pediatric patients
Writing about prescribing psychotropics to children for depression, anxiety, or attention-deficit/hyperactivity disorder (ADHD) sometimes brings conspiratorial accusations from readers, pediatrician Perri Klass, MD, writes in her column, “The Checkup” in the New York Times.
Some readers react to these discussions by suggesting that Dr. Klass is beholden to pharmaceutical companies. Others suggest that she wants to medicate young patients for behaviors that are a normal part of childhood. Of course, prescribing those medications to young patients should never be taken lightly, she says.
“It is a big deal, and there are side effects to worry about and doctors should listen to families’ concerns,” writes Dr. Klass, professor of journalism and pediatrics at New York University. “But when a child is suffering and struggling, families need help and medications are often part of the discussion.”
Dr. Klass goes on to interview Doris M. Greenberg, MD, and psychiatrist Timothy Wilens, MD, about the way they approach the treatment of children with psychiatric illness.
Click here to read Dr. Klass’s article in the Times.
Writing about prescribing psychotropics to children for depression, anxiety, or attention-deficit/hyperactivity disorder (ADHD) sometimes brings conspiratorial accusations from readers, pediatrician Perri Klass, MD, writes in her column, “The Checkup” in the New York Times.
Some readers react to these discussions by suggesting that Dr. Klass is beholden to pharmaceutical companies. Others suggest that she wants to medicate young patients for behaviors that are a normal part of childhood. Of course, prescribing those medications to young patients should never be taken lightly, she says.
“It is a big deal, and there are side effects to worry about and doctors should listen to families’ concerns,” writes Dr. Klass, professor of journalism and pediatrics at New York University. “But when a child is suffering and struggling, families need help and medications are often part of the discussion.”
Dr. Klass goes on to interview Doris M. Greenberg, MD, and psychiatrist Timothy Wilens, MD, about the way they approach the treatment of children with psychiatric illness.
Click here to read Dr. Klass’s article in the Times.
Writing about prescribing psychotropics to children for depression, anxiety, or attention-deficit/hyperactivity disorder (ADHD) sometimes brings conspiratorial accusations from readers, pediatrician Perri Klass, MD, writes in her column, “The Checkup” in the New York Times.
Some readers react to these discussions by suggesting that Dr. Klass is beholden to pharmaceutical companies. Others suggest that she wants to medicate young patients for behaviors that are a normal part of childhood. Of course, prescribing those medications to young patients should never be taken lightly, she says.
“It is a big deal, and there are side effects to worry about and doctors should listen to families’ concerns,” writes Dr. Klass, professor of journalism and pediatrics at New York University. “But when a child is suffering and struggling, families need help and medications are often part of the discussion.”
Dr. Klass goes on to interview Doris M. Greenberg, MD, and psychiatrist Timothy Wilens, MD, about the way they approach the treatment of children with psychiatric illness.
Click here to read Dr. Klass’s article in the Times.
Peer-comparison letters reduce physician quetiapine prescribing
A behavioral “nudge” intervention, targeting primary care prescribers who have particularly high off-label prescription rates of the antipsychotic quetiapine fumarate to older and disabled adults, has shown significant and long-lasting reductions in prescriptions.
A study, published Aug. 1 online by JAMA Psychiatry, looked at the effect of a “peer-comparison” letter, compared with a placebo letter, sent to 5,055 high quetiapine-prescribing primary care physicians in the Medicare program.
The letters said that the physicians’ quetiapine prescribing was extremely high, compared with their peers’ prescribing in the same state. Furthermore, the letters said the high-volume prescribers’ practices were under review because of concerns over medically unjustified use. They also encouraged the doctors to review their prescribing habits, while the placebo letter simply discussed an unrelated Medicare enrollment regulation.
Over the 9-month study, researchers saw a significant 11.1% reduction in the total number days of quetiapine prescribing among physicians who received the intervention letter, compared with those who received the control letter (95% confidence interval, –13.1 to –9.2 days; P less than .001; adjusted difference, –319 days; 95% CI, –374 to –263 days; P less than .001). At 2 years, the cumulative reduction was 15.6% fewer days in the intervention group (95% CI, –18.1 to –13.0; P less than .001), compared with the control group.
The study also used Medicare data to look at the impact on patients and found that individuals whose physicians were in the intervention arm had 3.9% fewer days of quetiapine usage over the 9 months (95% CI, –5.0 to –2.9; P less than 0.11), compared with those in the control arm. The reduction was even greater among patients whose indications for quetiapine were deemed to be of “low value,” as opposed to those who were prescribed for guideline-concordant indications, reported Adam Sacarny, PhD, of Columbia University, New York, and his coauthors.
When researchers looked in more detail at the reductions in prescriptions for guideline-concordant patients, they found that much of this was offset by prescriptions from other prescribers; in particular, physicians with psychiatric specialization or other study prescribers who the patient had not previously received a quetiapine prescription from.
The authors noted that the reductions for guideline-concordant patients could have negative effects if prescribers were reducing their quetiapine prescriptions indiscriminately.
“If this represented a harmful change for patients, we may have expected to see higher rates of adverse outcomes in the guideline-concordant patient group as prescribing rates decreased,” wrote Dr. Sacarny, and his coauthors. “However, if anything, guideline-concordant patients experienced lower rates of hospital encounters after the intervention.”
The study did not see any evidence of substitution to other antipsychotics, nor was any significant difference found in hospital use or mortality between the two arms of the study.
Dr. Sacarny and his coauthors cited several limitations. One is that the analysis looked at prescribing covered by Medicare Part D only. Nevertheless, they said, the results show the impact that peer comparison letters can have on prescribing patterns.
“These results provide encouraging evidence that high prescribers of antipsychotics can decrease quetiapine prescribing, without adverse clinical consequences, in response to a letter highlighting their overall high rates of prescribing,” the authors wrote.
The study was supported by the Robert Wood Johnson Foundation, Abdul Latif Jameel Poverty Action Lab North America, and the Laura and John Arnold Foundation. No conflicts of interest were reported.
SOURCE: Sacarny A et al. JAMA Psychiatry. 2018 Aug 1. doi: 10.1001/jamapsychiatry.2018.1867.
A behavioral “nudge” intervention, targeting primary care prescribers who have particularly high off-label prescription rates of the antipsychotic quetiapine fumarate to older and disabled adults, has shown significant and long-lasting reductions in prescriptions.
A study, published Aug. 1 online by JAMA Psychiatry, looked at the effect of a “peer-comparison” letter, compared with a placebo letter, sent to 5,055 high quetiapine-prescribing primary care physicians in the Medicare program.
The letters said that the physicians’ quetiapine prescribing was extremely high, compared with their peers’ prescribing in the same state. Furthermore, the letters said the high-volume prescribers’ practices were under review because of concerns over medically unjustified use. They also encouraged the doctors to review their prescribing habits, while the placebo letter simply discussed an unrelated Medicare enrollment regulation.
Over the 9-month study, researchers saw a significant 11.1% reduction in the total number days of quetiapine prescribing among physicians who received the intervention letter, compared with those who received the control letter (95% confidence interval, –13.1 to –9.2 days; P less than .001; adjusted difference, –319 days; 95% CI, –374 to –263 days; P less than .001). At 2 years, the cumulative reduction was 15.6% fewer days in the intervention group (95% CI, –18.1 to –13.0; P less than .001), compared with the control group.
The study also used Medicare data to look at the impact on patients and found that individuals whose physicians were in the intervention arm had 3.9% fewer days of quetiapine usage over the 9 months (95% CI, –5.0 to –2.9; P less than 0.11), compared with those in the control arm. The reduction was even greater among patients whose indications for quetiapine were deemed to be of “low value,” as opposed to those who were prescribed for guideline-concordant indications, reported Adam Sacarny, PhD, of Columbia University, New York, and his coauthors.
When researchers looked in more detail at the reductions in prescriptions for guideline-concordant patients, they found that much of this was offset by prescriptions from other prescribers; in particular, physicians with psychiatric specialization or other study prescribers who the patient had not previously received a quetiapine prescription from.
The authors noted that the reductions for guideline-concordant patients could have negative effects if prescribers were reducing their quetiapine prescriptions indiscriminately.
“If this represented a harmful change for patients, we may have expected to see higher rates of adverse outcomes in the guideline-concordant patient group as prescribing rates decreased,” wrote Dr. Sacarny, and his coauthors. “However, if anything, guideline-concordant patients experienced lower rates of hospital encounters after the intervention.”
The study did not see any evidence of substitution to other antipsychotics, nor was any significant difference found in hospital use or mortality between the two arms of the study.
Dr. Sacarny and his coauthors cited several limitations. One is that the analysis looked at prescribing covered by Medicare Part D only. Nevertheless, they said, the results show the impact that peer comparison letters can have on prescribing patterns.
“These results provide encouraging evidence that high prescribers of antipsychotics can decrease quetiapine prescribing, without adverse clinical consequences, in response to a letter highlighting their overall high rates of prescribing,” the authors wrote.
The study was supported by the Robert Wood Johnson Foundation, Abdul Latif Jameel Poverty Action Lab North America, and the Laura and John Arnold Foundation. No conflicts of interest were reported.
SOURCE: Sacarny A et al. JAMA Psychiatry. 2018 Aug 1. doi: 10.1001/jamapsychiatry.2018.1867.
A behavioral “nudge” intervention, targeting primary care prescribers who have particularly high off-label prescription rates of the antipsychotic quetiapine fumarate to older and disabled adults, has shown significant and long-lasting reductions in prescriptions.
A study, published Aug. 1 online by JAMA Psychiatry, looked at the effect of a “peer-comparison” letter, compared with a placebo letter, sent to 5,055 high quetiapine-prescribing primary care physicians in the Medicare program.
The letters said that the physicians’ quetiapine prescribing was extremely high, compared with their peers’ prescribing in the same state. Furthermore, the letters said the high-volume prescribers’ practices were under review because of concerns over medically unjustified use. They also encouraged the doctors to review their prescribing habits, while the placebo letter simply discussed an unrelated Medicare enrollment regulation.
Over the 9-month study, researchers saw a significant 11.1% reduction in the total number days of quetiapine prescribing among physicians who received the intervention letter, compared with those who received the control letter (95% confidence interval, –13.1 to –9.2 days; P less than .001; adjusted difference, –319 days; 95% CI, –374 to –263 days; P less than .001). At 2 years, the cumulative reduction was 15.6% fewer days in the intervention group (95% CI, –18.1 to –13.0; P less than .001), compared with the control group.
The study also used Medicare data to look at the impact on patients and found that individuals whose physicians were in the intervention arm had 3.9% fewer days of quetiapine usage over the 9 months (95% CI, –5.0 to –2.9; P less than 0.11), compared with those in the control arm. The reduction was even greater among patients whose indications for quetiapine were deemed to be of “low value,” as opposed to those who were prescribed for guideline-concordant indications, reported Adam Sacarny, PhD, of Columbia University, New York, and his coauthors.
When researchers looked in more detail at the reductions in prescriptions for guideline-concordant patients, they found that much of this was offset by prescriptions from other prescribers; in particular, physicians with psychiatric specialization or other study prescribers who the patient had not previously received a quetiapine prescription from.
The authors noted that the reductions for guideline-concordant patients could have negative effects if prescribers were reducing their quetiapine prescriptions indiscriminately.
“If this represented a harmful change for patients, we may have expected to see higher rates of adverse outcomes in the guideline-concordant patient group as prescribing rates decreased,” wrote Dr. Sacarny, and his coauthors. “However, if anything, guideline-concordant patients experienced lower rates of hospital encounters after the intervention.”
The study did not see any evidence of substitution to other antipsychotics, nor was any significant difference found in hospital use or mortality between the two arms of the study.
Dr. Sacarny and his coauthors cited several limitations. One is that the analysis looked at prescribing covered by Medicare Part D only. Nevertheless, they said, the results show the impact that peer comparison letters can have on prescribing patterns.
“These results provide encouraging evidence that high prescribers of antipsychotics can decrease quetiapine prescribing, without adverse clinical consequences, in response to a letter highlighting their overall high rates of prescribing,” the authors wrote.
The study was supported by the Robert Wood Johnson Foundation, Abdul Latif Jameel Poverty Action Lab North America, and the Laura and John Arnold Foundation. No conflicts of interest were reported.
SOURCE: Sacarny A et al. JAMA Psychiatry. 2018 Aug 1. doi: 10.1001/jamapsychiatry.2018.1867.
FROM JAMA PSYCHIATRY
Key clinical point: Letter intervention significantly reduces quetiapine prescription rates by physicians.
Major finding: Peer-comparison letters achieved an 11.1% reduction in days of quetiapine prescribed (95% confidence interval, –13.1 to –9.2 days; P less than .001; adjusted difference, –319 days; 95% CI, –374 to –263 days; P less than .001).
Study details: Randomized controlled trial in 5,055 high quetiapine-prescribing rates by primary care physicians.
Disclosures: The study was supported by the Robert Wood Johnson Foundation, Abdul Latif Jameel Poverty Action Lab North America, and the Laura and John Arnold Foundation. No conflicts of interest were declared.
Source: Sacarny A et al. JAMA Psychiatry. 2018 Aug 1. doi: 10.1001/jamapsychiatry.2018.1867.
Catatonia: How to identify and treat it
Is catatonia a rare condition that belongs in the history books, or is it more prevalent than we think? If we think we don’t see it often, how will we recognize it? And how do we treat it? This article reviews the evolution of our understanding of the phenomenology and therapy of this interesting and complex condition.
History of the concept
In 1874, Kahlbaum1,2 was the first to propose a syndrome of motor dysfunction characterized by mutism, immobility, staring gaze, negativism, stereotyped behavior, waxy flexibility, and verbal stereotypies that he called catatonia. Kahlbaum conceptualized catatonia as a distinct disorder,3 but Kraepelin reformulated it as a feature of dementia praecox.4 Although Bleuler felt that catatonia could occur in other psychiatric disorders and in normal people,4 he also included catatonia as a marker of schizophrenia, where it remained from DSM-I through DSM-IV.3 As was believed to be true of schizophrenia, Kraepelin considered catatonia to be characterized by poor prognosis, whereas Bleuler eliminated poor prognosis as a criterion for catatonia.3
In DSM-IV, catatonia was still a subtype of schizophrenia, but for the first time it was expanded diagnostically to become both a specifier in mood disorders, and a syndrome resulting from a general medical condition.5,6 In DSM-5, catatonic schizophrenia was deleted, and catatonia became a specifier for 10 disorders, including schizophrenia, mood disorders, and general medical conditions.3,5-9 In ICD-10, however, catatonia is still associated primarily with schizophrenia.10
A wide range of presentations
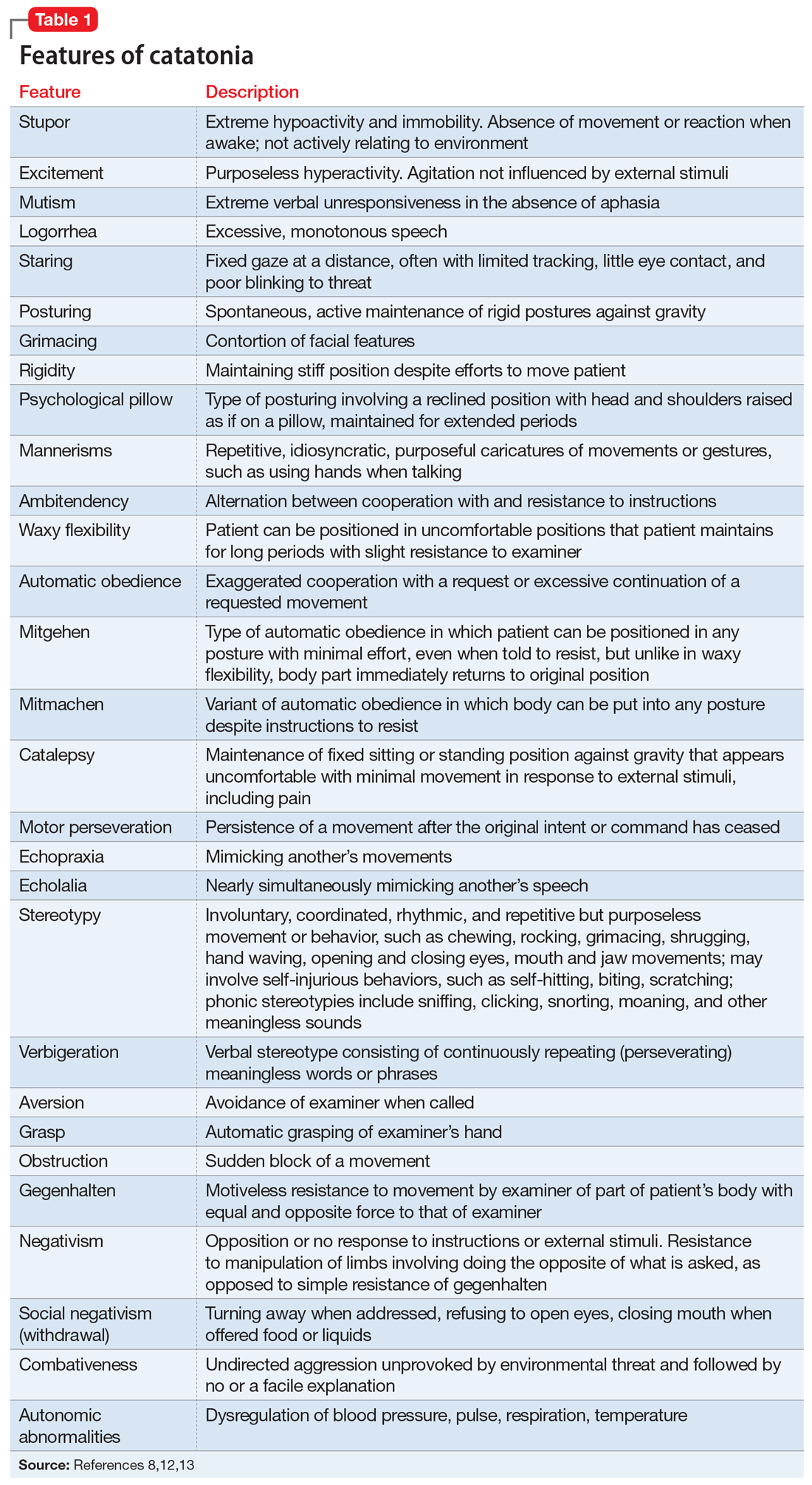
Catatonia is a cyclical syndrome characterized by alterations in motor, behavioral, and vocal signs occurring in the context of medical, neurologic, and psychiatric disorders.8 The most common features are immobility, waxy flexibility, stupor, mutism, negativism, echolalia, echopraxia, peculiarities of voluntary movement, and rigidity.7,11 Features of catatonia that have been repeatedly described through the years are summarized in Table 1.8,12,13 In general, presentations of catatonia are not specific to any psychiatric or medical etiology.13,14
Catatonia often is described along a continuum from retarded/stuporous to excited,14,15 and from benign to malignant.13 Examples of these ranges of presentation include5,12,13,15-19:
Stuporous/retarded catatonia (Kahlbaum syndrome) is a primarily negative syndrome in which stupor, mutism, negativism, obsessional slowness, and posturing predominate. Akinetic mutism and coma vigil are sometimes considered to be types of stuporous catatonia, as occasionally are locked-in syndrome and abulia caused by anterior cingulate lesions.
Excited catatonia (hyperkinetic variant, Bell’s mania, oneirophrenia, oneroid state/syndrome, catatonia raptus) is characterized by agitation, combativeness, verbigeration, stereotypies, grimacing, and echo phenomena (echopraxia and echolalia).
Continue to: Malignant (lethal) catatonia
Malignant (lethal) catatonia consists of catatonia accompanied by excitement, stupor, altered level of consciousness, catalepsy, hyperthermia, and autonomic instability with tachycardia, tachypnea, hypertension, and labile blood pressure. Autonomic dysregulation, fever, rhabdomyolysis, and acute renal failure can be causes of morbidity and mortality. Neuroleptic malignant syndrome (NMS)—which is associated with dopamine antagonists, especially antipsychotics—is considered a form of malignant catatonia and has a mortality rate of 10% to 20%. Signs of NMS include muscle rigidity, fever, diaphoresis, rigor, altered consciousness, mutism, tachycardia, hypertension, leukocytosis, and laboratory evidence of muscle damage. Serotonin syndrome can be difficult to distinguish from malignant catatonia, but it is usually not associated with waxy flexibility and rigidity.
Several specific subtypes of catatonia that may exist anywhere along dimensions of activity and severity also have been described:
Periodic catatonia. In 1908, Kraepelin described a form of periodic catatonia, with rapid shifts from excitement to stupor.4 Later, Gjessing described periodic catatonia in schizophrenia and reported success treating it with high doses of thyroid hormone.4 Today, periodic catatonia refers to the rapid onset of recurrent, brief hypokinetic or hyperkinetic episodes lasting 4 to 10 days and recurring during the course of weeks to years. Patients often are asymptomatic between episodes except for grimacing, stereotypies, and negativism later in the course.13,15 At least some forms of periodic catatonia are familial,4 with autosomal dominant transmission possibly linked to chromosome 15q15.13
A familial form of catatonia has been described that has a poor response to standard therapies (benzodiazepines and electroconvulsive therapy [ECT]), but in view of the high comorbidity of catatonia and bipolar disorder, it is difficult to determine whether this is a separate condition, or a group of patients with bipolar disorder.5
Late (ie, late-onset) catatonia is well described in the Japanese literature.10 Reported primarily in women without a known medical illness or brain disorder, late catatonia begins with prodromal hypochondriacal or depressive symptoms during a stressful situation, followed by unprovoked anxiety and agitation. Some patients develop hallucinations, delusions, and recurrent excitement, along with anxiety and agitation. The next stage involves typical catatonic features (mainly excitement, retardation, negativism, and autonomic disturbance), progressing to stupor, mutism, verbal stereotypies, and negativism, including refusal of food. Most patients have residual symptoms following improvement. A few cases have been noted to remit with ECT, with relapse when treatment was discontinued. Late catatonia has been thought to be associated with late-onset schizophrenia or bipolar disorder, or to be an independent entity.
Continue to: Untreated catatonia can have...
Untreated catatonia can have serious medical complications, including deep vein thrombosis, pulmonary embolism, aspiration pneumonia, infection, metabolic disorders, decubitus ulcers, malnutrition, dehydration, contractures, thrombosis, urinary retention, rhabdomyolysis, acute renal failure, sepsis, disseminated intravascular coagulation, and cardiac arrest.11,12,16,20,21 Mortality approaches 10%.12 In children and adolescents, catatonia increases the risk of premature death (including by suicide) 60-fold.22
Not as rare as you might think
With the shift from inpatient to outpatient care driven by deinstitutionalization, longitudinal close observation became less common, and clinicians got the impression that the dramatic catatonia that was common in the hospital had become rare.3 The impression that catatonia was unimportant was strengthened by expanding industry promotion of antipsychotic medications while ignoring catatonia, for which the industry had no specific treatment.3 With recent research, however, catatonia has been reported in 7% to 38% of adult psychiatric patients, including 9% to 25% of inpatients, 20% to 25% of patients with mania,3,5 and 20% of patients with major depressive episodes.7 Catatonia has been noted in .6% to 18% of adolescent psychiatric inpatients (especially in communication and social disorders programs),5,8,22 some children,5 and 6% to 18% of adult and juvenile patients with autism spectrum disorder (ASD).23 In the medical setting, catatonia occurs in 12% to 37% of patients with delirium,8,14,17,18,20,24 7% to 45% of medically ill patients, including those with no psychiatric history,12,13 and 4% of ICU patients.12 Several substances have been linked to catatonia; these are discussed later.11 Contrary to earlier impressions, catatonia is more common in mood disorders, particularly mixed bipolar disorder, especially mania,5 than in schizophrenia.7,8,17,25
Pathophysiology/etiology
Conditions associated with catatonia have different features that act through a final common pathway,7 possibly related to the neurobiology of an extreme fear response called tonic immobility that has been conserved through evolution.8 This mechanism may be mediated by decreased dopamine signaling in basal ganglia, orbitofrontal, and limbic systems, including the hypothalamus and basal forebrain.3,17,20 Subcortical reduction of dopaminergic neurotransmission appears to be related to reduced GABAA receptor signaling and dysfunction of N-methyl-
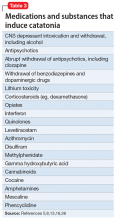
Up to one-quarter of cases of catatonia are secondary to medical (mostly neurologic) factors or substances.15 Table 25,13,15 lists common medical and neurological causes. Medications and substances known to cause catatonia are noted in Table 3.5,8,13,16,26
Catatonia can be a specifier, or a separate condition
DSM-5 criteria for catatonia are summarized in Table 4.28 With these features, catatonia can be a specifier for depressive, bipolar, or psychotic disorders; a complication of a medical disorder; or another separate diagnosis.8 The diagnosis of catatonia in DSM-5 is made when the clinical picture is dominated by ≥3 of the following core features8,15:
- motoric immobility as evidenced by catalepsy (including waxy flexibility) or stupor
- excessive purposeless motor activity that is not influenced by external stimuli
- extreme negativism or mutism
- peculiarities of voluntary movement such as posturing, stereotyped movements, prominent mannerisms, or prominent grimacing
- echolalia or echopraxia.
Continue to: DSM-5 criteria for the diagnosis of catatonia are more...
DSM-5 criteria for the diagnosis of catatonia are more restrictive than DSM-IV criteria. As a result, they exclude a significant number of patients who would be considered catatonic in other systems.29 For example, DSM-5 criteria do not include common features noted in Table 1,8,12,13 such as rigidity and staring.14,29 If the diagnosis is not obvious, it might be suspected in the presence of >1 of posturing, automatic obedience, or waxy flexibility, or >2 of echopraxia/echolalia, gegenhalten, negativism, mitgehen, or stereotypy/vergiberation.12 Clues to catatonia that are not included in formal diagnostic systems and are easily confused with features of psychosis include whispered or robotic speech, uncharacteristic foreign accent, tiptoe walking, hopping, rituals, and odd mannerisms.5
There are several catatonia rating scales containing between 14 and 40 items that are useful in diagnosing and following treatment response in catatonia (Table 58,13,15,29). Of these, the Kanner Scale is primarily applied in neuropsychiatric settings, while the Bush-Francis Catatonia Rating Scale (BFCRS) has had the most widespread use. The BFCRS consists of 23 items, the first 14 of which are used as a screening instrument. It requires 2 of its first 14 items to diagnose catatonia, while DSM-5 requires 3 of 12 signs.29 If the diagnosis remains in doubt, a benzodiazepine agonist test can be instructive.9,12 The presence of catatonia is suggested by significant improvement, ideally assessed prospectively by improvement of BFCRS scores, shortly after administration of a single dose of 1 to 2 mg lorazepam or 5 mg diazepam IV, or 10 mg zolpidem orally. Further evaluation generally consists of a careful medical and psychiatric histories of patient and family, review of all medications, history of substance use with toxicology as indicated, physical examination focusing on autonomic dysregulation, examination for delirium, and laboratory tests as suggested by the history and examination that may include complete blood count, creatine kinase, serum iron, blood urea nitrogen, electrolytes, creatinine, prolactin, anti-NMDA antibodies, thyroid function tests, serology, metabolic panel, human immunodeficiency virus testing, EEG, and neuroimaging.8,15,16
A complex differential diagnosis
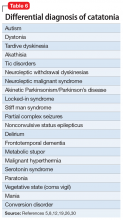
Manifestations of numerous psychiatric and neurologic disorders can mimic or be identical to those of catatonia. The differential diagnosis is complicated by the fact that some of these disorders can cause catatonia, which is then masked by the primary disorder; some disorders (eg, NMS) are forms of catatonia. Table 65,8,12,19,26,30 lists conditions to consider.
Some of these conditions warrant discussion. ASD may have catatonia-like features such as echolalia, echopraxia, excitement, combativeness, grimacing, mutism, logorrhea, verbigeration, catalepsy, mannerisms, rigidity, staring and withdrawal.8 Catatonia may also be a stage of deterioration of autism, in which case it is characterized by increases in slowness of movement and speech, reliance on physical or verbal prompting from others, passivity, and lack of motivation.23 At the same time, catatonic features such as mutism, stereotypic speech, repetitive behavior, echolalia, posturing, mannerisms, purposeless agitation, and rigidity in catatonia can be misinterpreted as signs of ASD.8 Catatonia should be suspected as a complication of longstanding ASD in the presence of a consistent, marked change in motor behavior, such as immobility, decreased speech, stupor, excitement, or mixtures or alternations of stupor and excitement.8 Freezing while doing something, difficulty crossing lines, or uncharacteristic persistence of a particular behavior may also herald the presence of catatonia with ASD.8
Catatonia caused by a neurologic or metabolic factor or a substance can be difficult to distinguish from delirium complicated by catatonia. Delirium may be identified in patients with catatonia by the presence of a waxing and waning level of consciousness (vs fluctuating behavior in catatonia) and slowing of the EEG.12,15 Antipsychotic medications can improve delirium but worsen catatonia, while benzodiazepines can improve catatonia but worsen delirium.
Continue to: Among other neurologic syndromes...
Among other neurologic syndromes that can be confused with catatonia, locked-in syndrome consists of total immobility except for vertical extraocular movements and blinking. In this state, patients attempt to communicate with their eyes, while catatonic patients do not try to communicate. There is no response to a lorazepam challenge test. Stiff man syndrome is associated with painful spasms precipitated by touch, noise, or emotional stimuli. Baclofen can resolve stiff man syndrome, but it can induce catatonia. Paratonia refers to generalized increased motor tone that is idiopathic, or associated with neurodegeneration, encephalopathy, or medications. The only motor sign is increased tone, and other signs of catatonia are absent. Catatonia is usually associated with some motor behaviors and interaction with the environment, even if it is negative, while the coma vigil patient is completely unresponsive. Frontotemporal dementia is progressive, while catatonia usually improves without residual dementia.30
Benzodiazepines, ECT are the usual treatments
Experience dictates that the general principles of treatment noted in Table 712,15,23,31 apply to all patients with catatonia. Since the first reported improvement of catatonia with amobarbital in 1930,6 there have been no controlled studies of specific treatments of catatonia.13 Meaningful treatment trials are either naturalistic, or have been performed only for NMS and malignant catatonia.5 However, multiple case reports and case series suggest that treatments with agents that have anticonvulsant properties (benzodiazepines, barbiturates) and ECT are effective.5
Benzodiazepines and related compounds. Case series have suggested a 60% to 80% remission rate of catatonia with benzodiazepines, the most commonly utilized of which has been lorazepam.7,13,32 Treatment begins with a lorazepam challenge test of 1 to 2 mg in adults and 0.5 to 1 mg in children and geriatric patients,9,15 administered orally (including via nasogastric tube), IM, or IV. Following a response (≥50% improvement), the dose is increased to 2 mg 3 times per day. The dose is further increased to 6 to 16 mg/d, and sometimes up to 30 mg/d.9,11 Oral is less effective than sublingual or IM administration.11 Diazepam can be helpful at doses 5 times the lorazepam dose.9,17 A zo
One alternative benzodiazepine protocol utilizes an initial IV dose of 2 mg lorazepam, repeated 3 to 5 times per day; the dose is increased to 10 to 12 mg/d if the first doses are partially effective.16 A lorazepam/diazepam approach involves a combination of IM lorazepam and IV diazepam.11 The protocol begins with 2 mg of IM lorazepam. If there is no effect within 2 hours, a second 2 mg dose is administered, followed by an IV infusion of 10 mg diazepam in 500 ml of normal saline at 1.25 mg/hour until catatonia remits.
An Indian study of 107 patients (mean age 26) receiving relatively low doses of lorazepam (3 to 6 mg/d for at least 3 days) found that factors suggesting a robust response include a shorter duration of catatonia and waxy flexibility, while passivity, mutism, and auditory hallucinations describing the patient in the third person were associated with a poorer acute response.31 Catatonia with marked retardation and mutism complicating schizophrenia, especially with chronic negative symptoms, may be associated with a lower response rate to benzodiazepines.20,33 Maintenance lorazepam has been effective in reducing relapse and recurrence.11 There are no controlled studies of maintenance treatment with benzodiazepines, but clinical reports suggest that doses in the range of 4 to 10 mg/d are effective.32
Continue to: ECT was used for catatonia in 1934...
ECT was first used for catatonia in 1934, when Laszlo Meduna used chemically induced seizures in catatonic patients who had been on tube feeding for months and no longer needed it after treatment.6,7 As was true for other disorders, this approach was replaced by ECT.7 In various case series, the effectiveness of ECT in catatonia has been 53% to 100%.7,13,15 Right unilateral ECT has been reported to be effective with 1 treatment.21 However, the best-established approach is with bitemporal ECT with a suprathreshold stimulus,9 usually with an acute course of 6 to 20 treatments.20 ECT has been reported to be equally safe and effective in adolescents and adults.34 Continued ECT is usually necessary until the patient has returned to baseline.9
ECT usually is recommended within 24 hours for treatment-resistant malignant catatonia or refusal to eat or drink, and within 2 to 3 days if medications are not sufficiently effective in other forms of catatonia.12,15,20 If ECT is initiated after a benzodiazepine trial, the benzodiazepine antagonist flumazenil is administered first to reverse the anticonvulsant effect.9 Some experts recommend using a muscle relaxant other than succinylcholine in the presence of evidence of muscle damage.7
Alternatives to benzodiazepines and ECT. Based on case reports, the treatments described in Table 813,15,17,20,25 have been used for patients with catatonia who do not tolerate or respond to standard treatments. The largest number of case reports have been with NMDA antagonists, while the presumed involvement of reduced dopamine signaling suggests that dopaminergic medications should be helpful. Dantrolene, which blocks release of calcium from intracellular stores and has been used to treat malignant hyperthermia, is sometimes used for NMS, often with disappointing results.
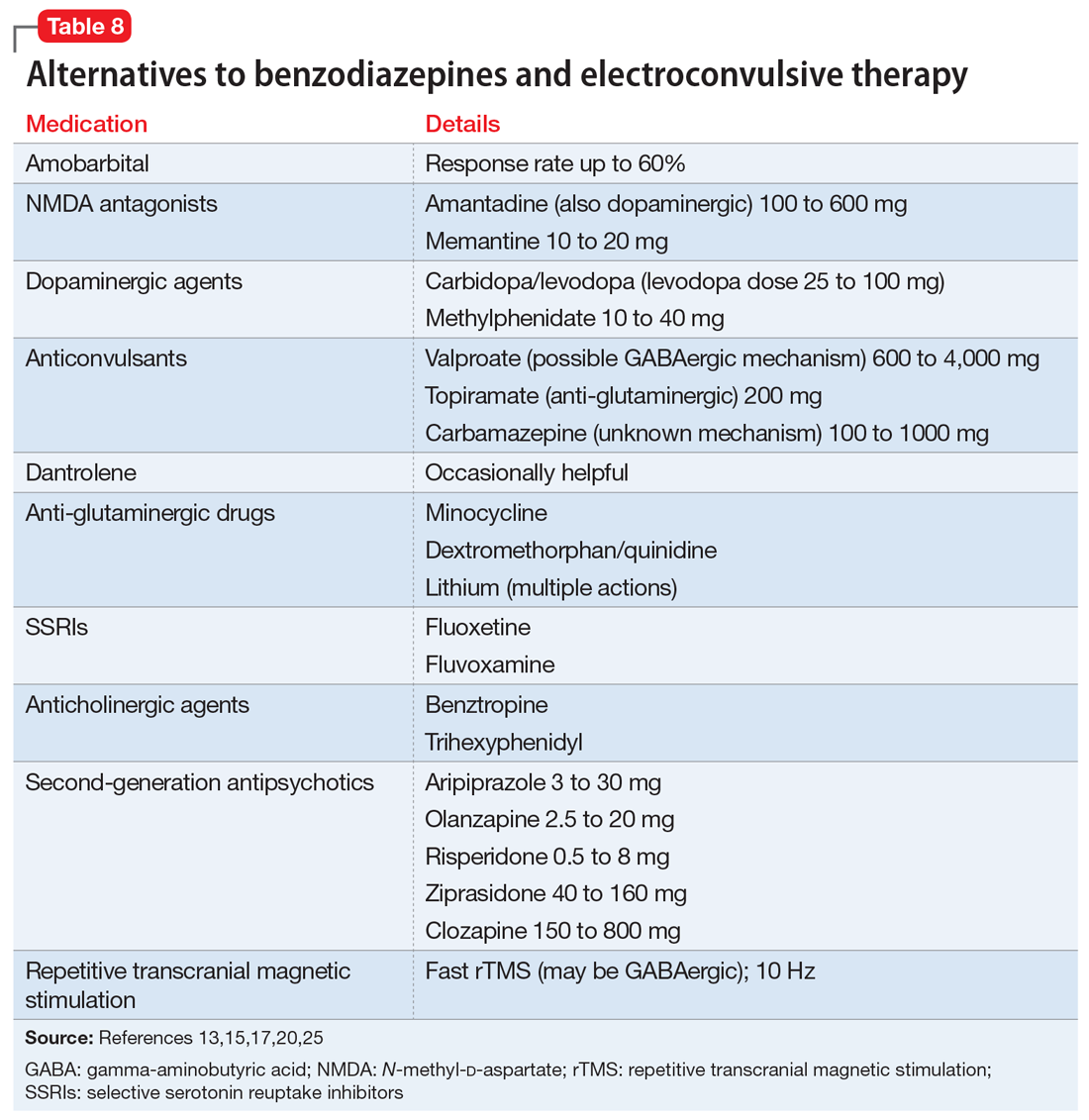
Whereas first-generation antipsychotics definitely increase the risk of catatonia and second-generation antipsychotics (SGAs) probably do so, SGAs are sometimes necessary to treat persistent psychosis in patients with schizophrenia who develop catatonia. Of these medications, clozapine may be most desirable because of low potency for dopamine receptor blockade and modulation of glutamatergic signaling. Partial dopamine agonism by aripiprazole, and the potential for increased subcortical prefrontal dopamine release resulting from serotonin 5HT2A antagonism and 5HT1A agonism by other SGAs, could also be helpful or at least not harmful in catatonia. Lorazepam is usually administered along with these medications to ameliorate treatment-emergent exacerbation of catatonia.
There are no controlled studies of any of these treatments. Based on case reports, most experts would recommend initiating treatment of catatonia with lorazepam, followed by ECT if necessary or in the presence of life-threatening catatonia. If ECT is not available, ineffective, or not tolerated, the first alternatives to be considered would be an NMDA antagonist or an anticonvulsant.20
Continue to: Course varies by patient, underlying cause
Course varies by patient, underlying cause
The response to benzodiazepines or ECT can vary from episode to episode11 and is similar in adults and younger patients.22 Many patients recover completely after a single episode, while relapse after remission occurs repeatedly in periodic catatonia, which involves chronic alternating stupor and excitement waxing and waning over years.11 Relapses may occur frequently, or every few years.11 Some cases of catatonia initially have an episodic course and become chronic and deteriorating, possibly paralleling the original descriptions of the natural history of untreated catatonia, while malignant catatonia can be complicated by medical morbidity or death.4 The long-term prognosis generally depends on the underlying cause of catatonia.5
Bottom Line
Much more common than many clinicians realize, catatonia can be overlooked because symptoms can mimic or overlap with features of an underlying medical or neurologic disorder. Suspect catatonia when one of these illnesses has an unexpected course or an inadequate treatment response. Be alert to characteristic changes in behavior and speech. A benzodiazepine challenge can be used to diagnose and begin treatment of catatonia. Consider electroconvulsive therapy sooner rather than later, especially for severely ill patients.
Related Resources
- Gibson RC, Walcott G. Benzodiazepines for catatonia in people with schizophrenia and other serious mental illnesses. Cochrane Database Syst Rev. 2008;(4):CD006570.
- Newcastle University. Catatonia. https://youtu.be/_s1lzxHRO4U.
Drug Brand Names
Amantadine • Symmetrel
Amobarbital • Amytal
Aripiprazole • Abilify
Azithromycin • Zithromax
Baclofen • Lioresal
Benztropine • Cogentin
Carbamazepine • Carbatrol, Tegretol
Carbidopa/levodopa • Sinemet
Ciprofloxacin • Cipro
Clozapine • Clozaril
Dantrolene • Dantrium
Dexamethasone • Decadron
Dextromethorphan/quinidine • Neudexta
Diazepam • Valium
Disulfiram • Antabuse
Flumazenil • Romazicon
Fluoxetine • Prozac
Fluvoxamine • Luvox
Levetiracetam • Keppra
Lithium • Eskalith, Lithobid
Lorazepam • Ativan
Memantine • Namenda
Methylphenidate • Ritalin
Minocycline • Minocin
Olanzapine • Zyprexa
Risperidone • Risperdal
Succinylcholine • Anectine
Topiramate • Topamax
Trihexyphenidyl • Artane
Valproate • Depakote
Ziprasidone • Geodon
Zolpidem • Ambien
1. Kahlbaum KL. Catatonia. Baltimore, MD: John Hopkins University Press; 1973.
2. Kahlbaum KL. Die Katatonie oder das Spannungsirresein. Berlin: Hirschwald; 1874.
3. Tang VM, Duffin J. Catatonia in the history of psychiatry: construction and deconstruction of a disease concept. Perspect Biol Med. 2014;57(4):524-537.
4. Carroll BT. Kahlbaum’s catatonia revisited. Psychiatry Clin Neurosci. 2001;55(5):431-436.
5. Taylor MA, Fink M. Catatonia in psychiatric classification: a home of its own. Am J Psychiatry. 2003;160(7):1233-1241.
6. Fink M, Fricchione GL, Rummans T, et al. Catatonia is a systemic medical syndrome. Acta Psychiatr Scand. 2016;133(3):250-251.
7. Medda P, Toni C, Luchini F, et al. Catatonia in 26 patients with bipolar disorder: clinical features and response to electroconvulsive therapy. Bipolar Disord. 2015;17(8):892-901.
8. Mazzone L, Postorino V, Valeri G, et al. Catatonia in patients with autism: prevalence and management. CNS Drugs. 2014;28(3):205-215.
9. Fink M, Kellner CH, McCall WV. Optimizing ECT technique in treating catatonia. J ECT. 2016;32(3):149-150.
10. Kocha H, Moriguchi S, Mimura M. Revisiting the concept of late catatonia. Compr Psychiatry. 2014;55(7):1485-1490.
11. Lin CC, Hung YL, Tsai MC, et al. Relapses and recurrences of catatonia: 30-case analysis and literature review. Compr Psychiatry. 2016;66:157-165.
12. Saddawi-Konefka D, Berg SM, Nejad SH, et al. Catatonia in the ICU: An important and underdiagnosed cause of altered mental status. A case series and review of the literature. Crit Care Med. 2013;42(3):e234-e241.
13. Wijemanne S, Jankovic J. Movement disorders in catatonia. J Neurol Neurosurg Psychiatry. 2015;86(8):825-832.
14. Grover S, Chakrabarti S, Ghormode D, et al. Catatonia in inpatients with psychiatric disorders: a comparison of schizophrenia and mood disorders. Psychiatry Res. 2015;229(3):919-925.
15. Oldham MA, Lee HB. Catatonia vis-à-vis delirium: the significance of recognizing catatonia in altered mental status. Gen Hosp Psychiatry. 2015;37(6):554-559.
16. Tuerlings JH, van Waarde JA, Verwey B. A retrospective study of 34 catatonic patients: analysis of clinical ‘care and treatment. Gen Hosp Psychiatry. 2010;32(6):631-635.
17. Ohi K, Kuwata A, Shimada T, et al. Response to benzodiazepines and the clinical course in malignant catatonia associated with schizophrenia: a case report. Medicine (Baltimore). 2017;96(16):e6566. doi: 10.1097/MD.0000000000006566.
18. Komatsu T, Nomura T, Takami H, et al. Catatonic symptoms appearing before autonomic symptoms help distinguish neuroleptic malignant syndrome from malignant catatonia. Intern Med. 2016;55(19):2893-2897.
19. Lang FU, Lang S, Becker T, et al. Neuroleptic malignant syndrome or catatonia? Trying to solve the catatonic dilemma. Psychopharmacology (Berl). 2015;232(1):1-5.
20. Beach SR, Gomez-Bernal F, Huffman JC, et al. Alternative treatment strategies for catatonia: a systematic review. Gen Hosp Psychiatry. 2017;48:1-19.
21. Kugler JL, Hauptman AJ, Collier SJ, et al. Treatment of catatonia with ultrabrief right unilateral electroconvulsive therapy: a case series. J ECT. 2015;31(3):192-196.
22. Raffin M, Zugaj-Bensaou L, Bodeau N, et al. Treatment use in a prospective naturalistic cohort of children and adolescents with catatonia. Eur Child Adolesc Psychiatry. 2015;24(4):441-449.
23. DeJong H, Bunton P, Hare DJ. A systematic review of interventions used to treat catatonic symptoms in people with autistic spectrum disorders. J Autism Dev Disord. 2014;44(9):2127-2136.
24. Wachtel L, Commins E, Park MH, et al. Neuroleptic malignant syndrome and delirious mania as malignant catatonia in autism: prompt relief with electroconvulsive therapy. Acta Psychiatr Scand. 2015;132(4):319-320.
25. Fink M, Taylor MA. Catatonia: subtype or syndrome in DSM? Am J Psychiatry. 2006;163(11):1875-1876.
26. Khan M, Pace L, Truong A, et al. Catatonia secondary to synthetic cannabinoid use in two patients with no previous psychosis. Am J Addictions. 2016;25(1):25-27.
27. Komatsu T, Nomura T, Takami H, et al. Catatonic symptoms appearing before autonomic symptoms help distinguish neuroleptic malignant syndrome from malignant catatonia. Intern Med. 2016;55(19):2893-2897.
28. Diagnostic and statistical manual of mental disorders, 5th ed. Washington, DC: American Psychiatric Association; 2013.
29. Wilson JE, Niu K, Nicolson SE, et al. The diagnostic criteria and structure of catatonia. Schizophr Res. 2015;164(1-3):256-262.
30. Ducharme S, Dickerson BC, Larvie M, et al. Differentiating frontotemporal dementia from catatonia: a complex neuropsychiatric challenge. J Neuropsychiatry Clin Neurosci. 2015;27(2):e174-e176.
31. Narayanaswamy JC, Tibrewal P, Zutshi A, et al. Clinical predictors of response to treatment in catatonia. Gen Hosp Psychiatry. 2012;34(3):312-316.
32. Thamizh JS, Harshini M, Selvakumar N, et al. Maintenance lorazepam for treatment of recurrent catatonic states: a case series and implications. Asian J Psychiatr. 2016;22:147-149
33. Ungvari GS, Chiu HF, Chow LY, et al. Lorazepam for chronic catatonia: a randomized, double-blind, placebo-controlled cross-over study. Psychopharmacology (Berl). 1999;142(4):393-398.
34. Flamarique I, Baeza I, de la Serna E, et al. Long-term effectiveness of electroconvulsive therapy in adolescents with schizophrenia spectrum disorders. Eur Child Adolesc Psychiatry. 2015;24(5):517-524.
Is catatonia a rare condition that belongs in the history books, or is it more prevalent than we think? If we think we don’t see it often, how will we recognize it? And how do we treat it? This article reviews the evolution of our understanding of the phenomenology and therapy of this interesting and complex condition.
History of the concept
In 1874, Kahlbaum1,2 was the first to propose a syndrome of motor dysfunction characterized by mutism, immobility, staring gaze, negativism, stereotyped behavior, waxy flexibility, and verbal stereotypies that he called catatonia. Kahlbaum conceptualized catatonia as a distinct disorder,3 but Kraepelin reformulated it as a feature of dementia praecox.4 Although Bleuler felt that catatonia could occur in other psychiatric disorders and in normal people,4 he also included catatonia as a marker of schizophrenia, where it remained from DSM-I through DSM-IV.3 As was believed to be true of schizophrenia, Kraepelin considered catatonia to be characterized by poor prognosis, whereas Bleuler eliminated poor prognosis as a criterion for catatonia.3
In DSM-IV, catatonia was still a subtype of schizophrenia, but for the first time it was expanded diagnostically to become both a specifier in mood disorders, and a syndrome resulting from a general medical condition.5,6 In DSM-5, catatonic schizophrenia was deleted, and catatonia became a specifier for 10 disorders, including schizophrenia, mood disorders, and general medical conditions.3,5-9 In ICD-10, however, catatonia is still associated primarily with schizophrenia.10
A wide range of presentations
Catatonia is a cyclical syndrome characterized by alterations in motor, behavioral, and vocal signs occurring in the context of medical, neurologic, and psychiatric disorders.8 The most common features are immobility, waxy flexibility, stupor, mutism, negativism, echolalia, echopraxia, peculiarities of voluntary movement, and rigidity.7,11 Features of catatonia that have been repeatedly described through the years are summarized in Table 1.8,12,13 In general, presentations of catatonia are not specific to any psychiatric or medical etiology.13,14

Catatonia often is described along a continuum from retarded/stuporous to excited,14,15 and from benign to malignant.13 Examples of these ranges of presentation include5,12,13,15-19:
Stuporous/retarded catatonia (Kahlbaum syndrome) is a primarily negative syndrome in which stupor, mutism, negativism, obsessional slowness, and posturing predominate. Akinetic mutism and coma vigil are sometimes considered to be types of stuporous catatonia, as occasionally are locked-in syndrome and abulia caused by anterior cingulate lesions.
Excited catatonia (hyperkinetic variant, Bell’s mania, oneirophrenia, oneroid state/syndrome, catatonia raptus) is characterized by agitation, combativeness, verbigeration, stereotypies, grimacing, and echo phenomena (echopraxia and echolalia).
Continue to: Malignant (lethal) catatonia
Malignant (lethal) catatonia consists of catatonia accompanied by excitement, stupor, altered level of consciousness, catalepsy, hyperthermia, and autonomic instability with tachycardia, tachypnea, hypertension, and labile blood pressure. Autonomic dysregulation, fever, rhabdomyolysis, and acute renal failure can be causes of morbidity and mortality. Neuroleptic malignant syndrome (NMS)—which is associated with dopamine antagonists, especially antipsychotics—is considered a form of malignant catatonia and has a mortality rate of 10% to 20%. Signs of NMS include muscle rigidity, fever, diaphoresis, rigor, altered consciousness, mutism, tachycardia, hypertension, leukocytosis, and laboratory evidence of muscle damage. Serotonin syndrome can be difficult to distinguish from malignant catatonia, but it is usually not associated with waxy flexibility and rigidity.
Several specific subtypes of catatonia that may exist anywhere along dimensions of activity and severity also have been described:
Periodic catatonia. In 1908, Kraepelin described a form of periodic catatonia, with rapid shifts from excitement to stupor.4 Later, Gjessing described periodic catatonia in schizophrenia and reported success treating it with high doses of thyroid hormone.4 Today, periodic catatonia refers to the rapid onset of recurrent, brief hypokinetic or hyperkinetic episodes lasting 4 to 10 days and recurring during the course of weeks to years. Patients often are asymptomatic between episodes except for grimacing, stereotypies, and negativism later in the course.13,15 At least some forms of periodic catatonia are familial,4 with autosomal dominant transmission possibly linked to chromosome 15q15.13
A familial form of catatonia has been described that has a poor response to standard therapies (benzodiazepines and electroconvulsive therapy [ECT]), but in view of the high comorbidity of catatonia and bipolar disorder, it is difficult to determine whether this is a separate condition, or a group of patients with bipolar disorder.5
Late (ie, late-onset) catatonia is well described in the Japanese literature.10 Reported primarily in women without a known medical illness or brain disorder, late catatonia begins with prodromal hypochondriacal or depressive symptoms during a stressful situation, followed by unprovoked anxiety and agitation. Some patients develop hallucinations, delusions, and recurrent excitement, along with anxiety and agitation. The next stage involves typical catatonic features (mainly excitement, retardation, negativism, and autonomic disturbance), progressing to stupor, mutism, verbal stereotypies, and negativism, including refusal of food. Most patients have residual symptoms following improvement. A few cases have been noted to remit with ECT, with relapse when treatment was discontinued. Late catatonia has been thought to be associated with late-onset schizophrenia or bipolar disorder, or to be an independent entity.
Continue to: Untreated catatonia can have...
Untreated catatonia can have serious medical complications, including deep vein thrombosis, pulmonary embolism, aspiration pneumonia, infection, metabolic disorders, decubitus ulcers, malnutrition, dehydration, contractures, thrombosis, urinary retention, rhabdomyolysis, acute renal failure, sepsis, disseminated intravascular coagulation, and cardiac arrest.11,12,16,20,21 Mortality approaches 10%.12 In children and adolescents, catatonia increases the risk of premature death (including by suicide) 60-fold.22
Not as rare as you might think
With the shift from inpatient to outpatient care driven by deinstitutionalization, longitudinal close observation became less common, and clinicians got the impression that the dramatic catatonia that was common in the hospital had become rare.3 The impression that catatonia was unimportant was strengthened by expanding industry promotion of antipsychotic medications while ignoring catatonia, for which the industry had no specific treatment.3 With recent research, however, catatonia has been reported in 7% to 38% of adult psychiatric patients, including 9% to 25% of inpatients, 20% to 25% of patients with mania,3,5 and 20% of patients with major depressive episodes.7 Catatonia has been noted in .6% to 18% of adolescent psychiatric inpatients (especially in communication and social disorders programs),5,8,22 some children,5 and 6% to 18% of adult and juvenile patients with autism spectrum disorder (ASD).23 In the medical setting, catatonia occurs in 12% to 37% of patients with delirium,8,14,17,18,20,24 7% to 45% of medically ill patients, including those with no psychiatric history,12,13 and 4% of ICU patients.12 Several substances have been linked to catatonia; these are discussed later.11 Contrary to earlier impressions, catatonia is more common in mood disorders, particularly mixed bipolar disorder, especially mania,5 than in schizophrenia.7,8,17,25
Pathophysiology/etiology
Conditions associated with catatonia have different features that act through a final common pathway,7 possibly related to the neurobiology of an extreme fear response called tonic immobility that has been conserved through evolution.8 This mechanism may be mediated by decreased dopamine signaling in basal ganglia, orbitofrontal, and limbic systems, including the hypothalamus and basal forebrain.3,17,20 Subcortical reduction of dopaminergic neurotransmission appears to be related to reduced GABAA receptor signaling and dysfunction of N-methyl-
Up to one-quarter of cases of catatonia are secondary to medical (mostly neurologic) factors or substances.15 Table 25,13,15 lists common medical and neurological causes. Medications and substances known to cause catatonia are noted in Table 3.5,8,13,16,26
Catatonia can be a specifier, or a separate condition
DSM-5 criteria for catatonia are summarized in Table 4.28 With these features, catatonia can be a specifier for depressive, bipolar, or psychotic disorders; a complication of a medical disorder; or another separate diagnosis.8 The diagnosis of catatonia in DSM-5 is made when the clinical picture is dominated by ≥3 of the following core features8,15:
- motoric immobility as evidenced by catalepsy (including waxy flexibility) or stupor
- excessive purposeless motor activity that is not influenced by external stimuli
- extreme negativism or mutism
- peculiarities of voluntary movement such as posturing, stereotyped movements, prominent mannerisms, or prominent grimacing
- echolalia or echopraxia.
Continue to: DSM-5 criteria for the diagnosis of catatonia are more...
DSM-5 criteria for the diagnosis of catatonia are more restrictive than DSM-IV criteria. As a result, they exclude a significant number of patients who would be considered catatonic in other systems.29 For example, DSM-5 criteria do not include common features noted in Table 1,8,12,13 such as rigidity and staring.14,29 If the diagnosis is not obvious, it might be suspected in the presence of >1 of posturing, automatic obedience, or waxy flexibility, or >2 of echopraxia/echolalia, gegenhalten, negativism, mitgehen, or stereotypy/vergiberation.12 Clues to catatonia that are not included in formal diagnostic systems and are easily confused with features of psychosis include whispered or robotic speech, uncharacteristic foreign accent, tiptoe walking, hopping, rituals, and odd mannerisms.5
There are several catatonia rating scales containing between 14 and 40 items that are useful in diagnosing and following treatment response in catatonia (Table 58,13,15,29). Of these, the Kanner Scale is primarily applied in neuropsychiatric settings, while the Bush-Francis Catatonia Rating Scale (BFCRS) has had the most widespread use. The BFCRS consists of 23 items, the first 14 of which are used as a screening instrument. It requires 2 of its first 14 items to diagnose catatonia, while DSM-5 requires 3 of 12 signs.29 If the diagnosis remains in doubt, a benzodiazepine agonist test can be instructive.9,12 The presence of catatonia is suggested by significant improvement, ideally assessed prospectively by improvement of BFCRS scores, shortly after administration of a single dose of 1 to 2 mg lorazepam or 5 mg diazepam IV, or 10 mg zolpidem orally. Further evaluation generally consists of a careful medical and psychiatric histories of patient and family, review of all medications, history of substance use with toxicology as indicated, physical examination focusing on autonomic dysregulation, examination for delirium, and laboratory tests as suggested by the history and examination that may include complete blood count, creatine kinase, serum iron, blood urea nitrogen, electrolytes, creatinine, prolactin, anti-NMDA antibodies, thyroid function tests, serology, metabolic panel, human immunodeficiency virus testing, EEG, and neuroimaging.8,15,16
A complex differential diagnosis
Manifestations of numerous psychiatric and neurologic disorders can mimic or be identical to those of catatonia. The differential diagnosis is complicated by the fact that some of these disorders can cause catatonia, which is then masked by the primary disorder; some disorders (eg, NMS) are forms of catatonia. Table 65,8,12,19,26,30 lists conditions to consider.
Some of these conditions warrant discussion. ASD may have catatonia-like features such as echolalia, echopraxia, excitement, combativeness, grimacing, mutism, logorrhea, verbigeration, catalepsy, mannerisms, rigidity, staring and withdrawal.8 Catatonia may also be a stage of deterioration of autism, in which case it is characterized by increases in slowness of movement and speech, reliance on physical or verbal prompting from others, passivity, and lack of motivation.23 At the same time, catatonic features such as mutism, stereotypic speech, repetitive behavior, echolalia, posturing, mannerisms, purposeless agitation, and rigidity in catatonia can be misinterpreted as signs of ASD.8 Catatonia should be suspected as a complication of longstanding ASD in the presence of a consistent, marked change in motor behavior, such as immobility, decreased speech, stupor, excitement, or mixtures or alternations of stupor and excitement.8 Freezing while doing something, difficulty crossing lines, or uncharacteristic persistence of a particular behavior may also herald the presence of catatonia with ASD.8
Catatonia caused by a neurologic or metabolic factor or a substance can be difficult to distinguish from delirium complicated by catatonia. Delirium may be identified in patients with catatonia by the presence of a waxing and waning level of consciousness (vs fluctuating behavior in catatonia) and slowing of the EEG.12,15 Antipsychotic medications can improve delirium but worsen catatonia, while benzodiazepines can improve catatonia but worsen delirium.
Continue to: Among other neurologic syndromes...
Among other neurologic syndromes that can be confused with catatonia, locked-in syndrome consists of total immobility except for vertical extraocular movements and blinking. In this state, patients attempt to communicate with their eyes, while catatonic patients do not try to communicate. There is no response to a lorazepam challenge test. Stiff man syndrome is associated with painful spasms precipitated by touch, noise, or emotional stimuli. Baclofen can resolve stiff man syndrome, but it can induce catatonia. Paratonia refers to generalized increased motor tone that is idiopathic, or associated with neurodegeneration, encephalopathy, or medications. The only motor sign is increased tone, and other signs of catatonia are absent. Catatonia is usually associated with some motor behaviors and interaction with the environment, even if it is negative, while the coma vigil patient is completely unresponsive. Frontotemporal dementia is progressive, while catatonia usually improves without residual dementia.30
Benzodiazepines, ECT are the usual treatments
Experience dictates that the general principles of treatment noted in Table 712,15,23,31 apply to all patients with catatonia. Since the first reported improvement of catatonia with amobarbital in 1930,6 there have been no controlled studies of specific treatments of catatonia.13 Meaningful treatment trials are either naturalistic, or have been performed only for NMS and malignant catatonia.5 However, multiple case reports and case series suggest that treatments with agents that have anticonvulsant properties (benzodiazepines, barbiturates) and ECT are effective.5
Benzodiazepines and related compounds. Case series have suggested a 60% to 80% remission rate of catatonia with benzodiazepines, the most commonly utilized of which has been lorazepam.7,13,32 Treatment begins with a lorazepam challenge test of 1 to 2 mg in adults and 0.5 to 1 mg in children and geriatric patients,9,15 administered orally (including via nasogastric tube), IM, or IV. Following a response (≥50% improvement), the dose is increased to 2 mg 3 times per day. The dose is further increased to 6 to 16 mg/d, and sometimes up to 30 mg/d.9,11 Oral is less effective than sublingual or IM administration.11 Diazepam can be helpful at doses 5 times the lorazepam dose.9,17 A zo
One alternative benzodiazepine protocol utilizes an initial IV dose of 2 mg lorazepam, repeated 3 to 5 times per day; the dose is increased to 10 to 12 mg/d if the first doses are partially effective.16 A lorazepam/diazepam approach involves a combination of IM lorazepam and IV diazepam.11 The protocol begins with 2 mg of IM lorazepam. If there is no effect within 2 hours, a second 2 mg dose is administered, followed by an IV infusion of 10 mg diazepam in 500 ml of normal saline at 1.25 mg/hour until catatonia remits.
An Indian study of 107 patients (mean age 26) receiving relatively low doses of lorazepam (3 to 6 mg/d for at least 3 days) found that factors suggesting a robust response include a shorter duration of catatonia and waxy flexibility, while passivity, mutism, and auditory hallucinations describing the patient in the third person were associated with a poorer acute response.31 Catatonia with marked retardation and mutism complicating schizophrenia, especially with chronic negative symptoms, may be associated with a lower response rate to benzodiazepines.20,33 Maintenance lorazepam has been effective in reducing relapse and recurrence.11 There are no controlled studies of maintenance treatment with benzodiazepines, but clinical reports suggest that doses in the range of 4 to 10 mg/d are effective.32
Continue to: ECT was used for catatonia in 1934...
ECT was first used for catatonia in 1934, when Laszlo Meduna used chemically induced seizures in catatonic patients who had been on tube feeding for months and no longer needed it after treatment.6,7 As was true for other disorders, this approach was replaced by ECT.7 In various case series, the effectiveness of ECT in catatonia has been 53% to 100%.7,13,15 Right unilateral ECT has been reported to be effective with 1 treatment.21 However, the best-established approach is with bitemporal ECT with a suprathreshold stimulus,9 usually with an acute course of 6 to 20 treatments.20 ECT has been reported to be equally safe and effective in adolescents and adults.34 Continued ECT is usually necessary until the patient has returned to baseline.9
ECT usually is recommended within 24 hours for treatment-resistant malignant catatonia or refusal to eat or drink, and within 2 to 3 days if medications are not sufficiently effective in other forms of catatonia.12,15,20 If ECT is initiated after a benzodiazepine trial, the benzodiazepine antagonist flumazenil is administered first to reverse the anticonvulsant effect.9 Some experts recommend using a muscle relaxant other than succinylcholine in the presence of evidence of muscle damage.7
Alternatives to benzodiazepines and ECT. Based on case reports, the treatments described in Table 813,15,17,20,25 have been used for patients with catatonia who do not tolerate or respond to standard treatments. The largest number of case reports have been with NMDA antagonists, while the presumed involvement of reduced dopamine signaling suggests that dopaminergic medications should be helpful. Dantrolene, which blocks release of calcium from intracellular stores and has been used to treat malignant hyperthermia, is sometimes used for NMS, often with disappointing results.

Whereas first-generation antipsychotics definitely increase the risk of catatonia and second-generation antipsychotics (SGAs) probably do so, SGAs are sometimes necessary to treat persistent psychosis in patients with schizophrenia who develop catatonia. Of these medications, clozapine may be most desirable because of low potency for dopamine receptor blockade and modulation of glutamatergic signaling. Partial dopamine agonism by aripiprazole, and the potential for increased subcortical prefrontal dopamine release resulting from serotonin 5HT2A antagonism and 5HT1A agonism by other SGAs, could also be helpful or at least not harmful in catatonia. Lorazepam is usually administered along with these medications to ameliorate treatment-emergent exacerbation of catatonia.
There are no controlled studies of any of these treatments. Based on case reports, most experts would recommend initiating treatment of catatonia with lorazepam, followed by ECT if necessary or in the presence of life-threatening catatonia. If ECT is not available, ineffective, or not tolerated, the first alternatives to be considered would be an NMDA antagonist or an anticonvulsant.20
Continue to: Course varies by patient, underlying cause
Course varies by patient, underlying cause
The response to benzodiazepines or ECT can vary from episode to episode11 and is similar in adults and younger patients.22 Many patients recover completely after a single episode, while relapse after remission occurs repeatedly in periodic catatonia, which involves chronic alternating stupor and excitement waxing and waning over years.11 Relapses may occur frequently, or every few years.11 Some cases of catatonia initially have an episodic course and become chronic and deteriorating, possibly paralleling the original descriptions of the natural history of untreated catatonia, while malignant catatonia can be complicated by medical morbidity or death.4 The long-term prognosis generally depends on the underlying cause of catatonia.5
Bottom Line
Much more common than many clinicians realize, catatonia can be overlooked because symptoms can mimic or overlap with features of an underlying medical or neurologic disorder. Suspect catatonia when one of these illnesses has an unexpected course or an inadequate treatment response. Be alert to characteristic changes in behavior and speech. A benzodiazepine challenge can be used to diagnose and begin treatment of catatonia. Consider electroconvulsive therapy sooner rather than later, especially for severely ill patients.
Related Resources
- Gibson RC, Walcott G. Benzodiazepines for catatonia in people with schizophrenia and other serious mental illnesses. Cochrane Database Syst Rev. 2008;(4):CD006570.
- Newcastle University. Catatonia. https://youtu.be/_s1lzxHRO4U.
Drug Brand Names
Amantadine • Symmetrel
Amobarbital • Amytal
Aripiprazole • Abilify
Azithromycin • Zithromax
Baclofen • Lioresal
Benztropine • Cogentin
Carbamazepine • Carbatrol, Tegretol
Carbidopa/levodopa • Sinemet
Ciprofloxacin • Cipro
Clozapine • Clozaril
Dantrolene • Dantrium
Dexamethasone • Decadron
Dextromethorphan/quinidine • Neudexta
Diazepam • Valium
Disulfiram • Antabuse
Flumazenil • Romazicon
Fluoxetine • Prozac
Fluvoxamine • Luvox
Levetiracetam • Keppra
Lithium • Eskalith, Lithobid
Lorazepam • Ativan
Memantine • Namenda
Methylphenidate • Ritalin
Minocycline • Minocin
Olanzapine • Zyprexa
Risperidone • Risperdal
Succinylcholine • Anectine
Topiramate • Topamax
Trihexyphenidyl • Artane
Valproate • Depakote
Ziprasidone • Geodon
Zolpidem • Ambien
Is catatonia a rare condition that belongs in the history books, or is it more prevalent than we think? If we think we don’t see it often, how will we recognize it? And how do we treat it? This article reviews the evolution of our understanding of the phenomenology and therapy of this interesting and complex condition.
History of the concept
In 1874, Kahlbaum1,2 was the first to propose a syndrome of motor dysfunction characterized by mutism, immobility, staring gaze, negativism, stereotyped behavior, waxy flexibility, and verbal stereotypies that he called catatonia. Kahlbaum conceptualized catatonia as a distinct disorder,3 but Kraepelin reformulated it as a feature of dementia praecox.4 Although Bleuler felt that catatonia could occur in other psychiatric disorders and in normal people,4 he also included catatonia as a marker of schizophrenia, where it remained from DSM-I through DSM-IV.3 As was believed to be true of schizophrenia, Kraepelin considered catatonia to be characterized by poor prognosis, whereas Bleuler eliminated poor prognosis as a criterion for catatonia.3
In DSM-IV, catatonia was still a subtype of schizophrenia, but for the first time it was expanded diagnostically to become both a specifier in mood disorders, and a syndrome resulting from a general medical condition.5,6 In DSM-5, catatonic schizophrenia was deleted, and catatonia became a specifier for 10 disorders, including schizophrenia, mood disorders, and general medical conditions.3,5-9 In ICD-10, however, catatonia is still associated primarily with schizophrenia.10
A wide range of presentations
Catatonia is a cyclical syndrome characterized by alterations in motor, behavioral, and vocal signs occurring in the context of medical, neurologic, and psychiatric disorders.8 The most common features are immobility, waxy flexibility, stupor, mutism, negativism, echolalia, echopraxia, peculiarities of voluntary movement, and rigidity.7,11 Features of catatonia that have been repeatedly described through the years are summarized in Table 1.8,12,13 In general, presentations of catatonia are not specific to any psychiatric or medical etiology.13,14

Catatonia often is described along a continuum from retarded/stuporous to excited,14,15 and from benign to malignant.13 Examples of these ranges of presentation include5,12,13,15-19:
Stuporous/retarded catatonia (Kahlbaum syndrome) is a primarily negative syndrome in which stupor, mutism, negativism, obsessional slowness, and posturing predominate. Akinetic mutism and coma vigil are sometimes considered to be types of stuporous catatonia, as occasionally are locked-in syndrome and abulia caused by anterior cingulate lesions.
Excited catatonia (hyperkinetic variant, Bell’s mania, oneirophrenia, oneroid state/syndrome, catatonia raptus) is characterized by agitation, combativeness, verbigeration, stereotypies, grimacing, and echo phenomena (echopraxia and echolalia).
Continue to: Malignant (lethal) catatonia
Malignant (lethal) catatonia consists of catatonia accompanied by excitement, stupor, altered level of consciousness, catalepsy, hyperthermia, and autonomic instability with tachycardia, tachypnea, hypertension, and labile blood pressure. Autonomic dysregulation, fever, rhabdomyolysis, and acute renal failure can be causes of morbidity and mortality. Neuroleptic malignant syndrome (NMS)—which is associated with dopamine antagonists, especially antipsychotics—is considered a form of malignant catatonia and has a mortality rate of 10% to 20%. Signs of NMS include muscle rigidity, fever, diaphoresis, rigor, altered consciousness, mutism, tachycardia, hypertension, leukocytosis, and laboratory evidence of muscle damage. Serotonin syndrome can be difficult to distinguish from malignant catatonia, but it is usually not associated with waxy flexibility and rigidity.
Several specific subtypes of catatonia that may exist anywhere along dimensions of activity and severity also have been described:
Periodic catatonia. In 1908, Kraepelin described a form of periodic catatonia, with rapid shifts from excitement to stupor.4 Later, Gjessing described periodic catatonia in schizophrenia and reported success treating it with high doses of thyroid hormone.4 Today, periodic catatonia refers to the rapid onset of recurrent, brief hypokinetic or hyperkinetic episodes lasting 4 to 10 days and recurring during the course of weeks to years. Patients often are asymptomatic between episodes except for grimacing, stereotypies, and negativism later in the course.13,15 At least some forms of periodic catatonia are familial,4 with autosomal dominant transmission possibly linked to chromosome 15q15.13
A familial form of catatonia has been described that has a poor response to standard therapies (benzodiazepines and electroconvulsive therapy [ECT]), but in view of the high comorbidity of catatonia and bipolar disorder, it is difficult to determine whether this is a separate condition, or a group of patients with bipolar disorder.5
Late (ie, late-onset) catatonia is well described in the Japanese literature.10 Reported primarily in women without a known medical illness or brain disorder, late catatonia begins with prodromal hypochondriacal or depressive symptoms during a stressful situation, followed by unprovoked anxiety and agitation. Some patients develop hallucinations, delusions, and recurrent excitement, along with anxiety and agitation. The next stage involves typical catatonic features (mainly excitement, retardation, negativism, and autonomic disturbance), progressing to stupor, mutism, verbal stereotypies, and negativism, including refusal of food. Most patients have residual symptoms following improvement. A few cases have been noted to remit with ECT, with relapse when treatment was discontinued. Late catatonia has been thought to be associated with late-onset schizophrenia or bipolar disorder, or to be an independent entity.
Continue to: Untreated catatonia can have...
Untreated catatonia can have serious medical complications, including deep vein thrombosis, pulmonary embolism, aspiration pneumonia, infection, metabolic disorders, decubitus ulcers, malnutrition, dehydration, contractures, thrombosis, urinary retention, rhabdomyolysis, acute renal failure, sepsis, disseminated intravascular coagulation, and cardiac arrest.11,12,16,20,21 Mortality approaches 10%.12 In children and adolescents, catatonia increases the risk of premature death (including by suicide) 60-fold.22
Not as rare as you might think
With the shift from inpatient to outpatient care driven by deinstitutionalization, longitudinal close observation became less common, and clinicians got the impression that the dramatic catatonia that was common in the hospital had become rare.3 The impression that catatonia was unimportant was strengthened by expanding industry promotion of antipsychotic medications while ignoring catatonia, for which the industry had no specific treatment.3 With recent research, however, catatonia has been reported in 7% to 38% of adult psychiatric patients, including 9% to 25% of inpatients, 20% to 25% of patients with mania,3,5 and 20% of patients with major depressive episodes.7 Catatonia has been noted in .6% to 18% of adolescent psychiatric inpatients (especially in communication and social disorders programs),5,8,22 some children,5 and 6% to 18% of adult and juvenile patients with autism spectrum disorder (ASD).23 In the medical setting, catatonia occurs in 12% to 37% of patients with delirium,8,14,17,18,20,24 7% to 45% of medically ill patients, including those with no psychiatric history,12,13 and 4% of ICU patients.12 Several substances have been linked to catatonia; these are discussed later.11 Contrary to earlier impressions, catatonia is more common in mood disorders, particularly mixed bipolar disorder, especially mania,5 than in schizophrenia.7,8,17,25
Pathophysiology/etiology
Conditions associated with catatonia have different features that act through a final common pathway,7 possibly related to the neurobiology of an extreme fear response called tonic immobility that has been conserved through evolution.8 This mechanism may be mediated by decreased dopamine signaling in basal ganglia, orbitofrontal, and limbic systems, including the hypothalamus and basal forebrain.3,17,20 Subcortical reduction of dopaminergic neurotransmission appears to be related to reduced GABAA receptor signaling and dysfunction of N-methyl-
Up to one-quarter of cases of catatonia are secondary to medical (mostly neurologic) factors or substances.15 Table 25,13,15 lists common medical and neurological causes. Medications and substances known to cause catatonia are noted in Table 3.5,8,13,16,26
Catatonia can be a specifier, or a separate condition
DSM-5 criteria for catatonia are summarized in Table 4.28 With these features, catatonia can be a specifier for depressive, bipolar, or psychotic disorders; a complication of a medical disorder; or another separate diagnosis.8 The diagnosis of catatonia in DSM-5 is made when the clinical picture is dominated by ≥3 of the following core features8,15:
- motoric immobility as evidenced by catalepsy (including waxy flexibility) or stupor
- excessive purposeless motor activity that is not influenced by external stimuli
- extreme negativism or mutism
- peculiarities of voluntary movement such as posturing, stereotyped movements, prominent mannerisms, or prominent grimacing
- echolalia or echopraxia.
Continue to: DSM-5 criteria for the diagnosis of catatonia are more...
DSM-5 criteria for the diagnosis of catatonia are more restrictive than DSM-IV criteria. As a result, they exclude a significant number of patients who would be considered catatonic in other systems.29 For example, DSM-5 criteria do not include common features noted in Table 1,8,12,13 such as rigidity and staring.14,29 If the diagnosis is not obvious, it might be suspected in the presence of >1 of posturing, automatic obedience, or waxy flexibility, or >2 of echopraxia/echolalia, gegenhalten, negativism, mitgehen, or stereotypy/vergiberation.12 Clues to catatonia that are not included in formal diagnostic systems and are easily confused with features of psychosis include whispered or robotic speech, uncharacteristic foreign accent, tiptoe walking, hopping, rituals, and odd mannerisms.5
There are several catatonia rating scales containing between 14 and 40 items that are useful in diagnosing and following treatment response in catatonia (Table 58,13,15,29). Of these, the Kanner Scale is primarily applied in neuropsychiatric settings, while the Bush-Francis Catatonia Rating Scale (BFCRS) has had the most widespread use. The BFCRS consists of 23 items, the first 14 of which are used as a screening instrument. It requires 2 of its first 14 items to diagnose catatonia, while DSM-5 requires 3 of 12 signs.29 If the diagnosis remains in doubt, a benzodiazepine agonist test can be instructive.9,12 The presence of catatonia is suggested by significant improvement, ideally assessed prospectively by improvement of BFCRS scores, shortly after administration of a single dose of 1 to 2 mg lorazepam or 5 mg diazepam IV, or 10 mg zolpidem orally. Further evaluation generally consists of a careful medical and psychiatric histories of patient and family, review of all medications, history of substance use with toxicology as indicated, physical examination focusing on autonomic dysregulation, examination for delirium, and laboratory tests as suggested by the history and examination that may include complete blood count, creatine kinase, serum iron, blood urea nitrogen, electrolytes, creatinine, prolactin, anti-NMDA antibodies, thyroid function tests, serology, metabolic panel, human immunodeficiency virus testing, EEG, and neuroimaging.8,15,16
A complex differential diagnosis
Manifestations of numerous psychiatric and neurologic disorders can mimic or be identical to those of catatonia. The differential diagnosis is complicated by the fact that some of these disorders can cause catatonia, which is then masked by the primary disorder; some disorders (eg, NMS) are forms of catatonia. Table 65,8,12,19,26,30 lists conditions to consider.
Some of these conditions warrant discussion. ASD may have catatonia-like features such as echolalia, echopraxia, excitement, combativeness, grimacing, mutism, logorrhea, verbigeration, catalepsy, mannerisms, rigidity, staring and withdrawal.8 Catatonia may also be a stage of deterioration of autism, in which case it is characterized by increases in slowness of movement and speech, reliance on physical or verbal prompting from others, passivity, and lack of motivation.23 At the same time, catatonic features such as mutism, stereotypic speech, repetitive behavior, echolalia, posturing, mannerisms, purposeless agitation, and rigidity in catatonia can be misinterpreted as signs of ASD.8 Catatonia should be suspected as a complication of longstanding ASD in the presence of a consistent, marked change in motor behavior, such as immobility, decreased speech, stupor, excitement, or mixtures or alternations of stupor and excitement.8 Freezing while doing something, difficulty crossing lines, or uncharacteristic persistence of a particular behavior may also herald the presence of catatonia with ASD.8
Catatonia caused by a neurologic or metabolic factor or a substance can be difficult to distinguish from delirium complicated by catatonia. Delirium may be identified in patients with catatonia by the presence of a waxing and waning level of consciousness (vs fluctuating behavior in catatonia) and slowing of the EEG.12,15 Antipsychotic medications can improve delirium but worsen catatonia, while benzodiazepines can improve catatonia but worsen delirium.
Continue to: Among other neurologic syndromes...
Among other neurologic syndromes that can be confused with catatonia, locked-in syndrome consists of total immobility except for vertical extraocular movements and blinking. In this state, patients attempt to communicate with their eyes, while catatonic patients do not try to communicate. There is no response to a lorazepam challenge test. Stiff man syndrome is associated with painful spasms precipitated by touch, noise, or emotional stimuli. Baclofen can resolve stiff man syndrome, but it can induce catatonia. Paratonia refers to generalized increased motor tone that is idiopathic, or associated with neurodegeneration, encephalopathy, or medications. The only motor sign is increased tone, and other signs of catatonia are absent. Catatonia is usually associated with some motor behaviors and interaction with the environment, even if it is negative, while the coma vigil patient is completely unresponsive. Frontotemporal dementia is progressive, while catatonia usually improves without residual dementia.30
Benzodiazepines, ECT are the usual treatments
Experience dictates that the general principles of treatment noted in Table 712,15,23,31 apply to all patients with catatonia. Since the first reported improvement of catatonia with amobarbital in 1930,6 there have been no controlled studies of specific treatments of catatonia.13 Meaningful treatment trials are either naturalistic, or have been performed only for NMS and malignant catatonia.5 However, multiple case reports and case series suggest that treatments with agents that have anticonvulsant properties (benzodiazepines, barbiturates) and ECT are effective.5
Benzodiazepines and related compounds. Case series have suggested a 60% to 80% remission rate of catatonia with benzodiazepines, the most commonly utilized of which has been lorazepam.7,13,32 Treatment begins with a lorazepam challenge test of 1 to 2 mg in adults and 0.5 to 1 mg in children and geriatric patients,9,15 administered orally (including via nasogastric tube), IM, or IV. Following a response (≥50% improvement), the dose is increased to 2 mg 3 times per day. The dose is further increased to 6 to 16 mg/d, and sometimes up to 30 mg/d.9,11 Oral is less effective than sublingual or IM administration.11 Diazepam can be helpful at doses 5 times the lorazepam dose.9,17 A zo
One alternative benzodiazepine protocol utilizes an initial IV dose of 2 mg lorazepam, repeated 3 to 5 times per day; the dose is increased to 10 to 12 mg/d if the first doses are partially effective.16 A lorazepam/diazepam approach involves a combination of IM lorazepam and IV diazepam.11 The protocol begins with 2 mg of IM lorazepam. If there is no effect within 2 hours, a second 2 mg dose is administered, followed by an IV infusion of 10 mg diazepam in 500 ml of normal saline at 1.25 mg/hour until catatonia remits.
An Indian study of 107 patients (mean age 26) receiving relatively low doses of lorazepam (3 to 6 mg/d for at least 3 days) found that factors suggesting a robust response include a shorter duration of catatonia and waxy flexibility, while passivity, mutism, and auditory hallucinations describing the patient in the third person were associated with a poorer acute response.31 Catatonia with marked retardation and mutism complicating schizophrenia, especially with chronic negative symptoms, may be associated with a lower response rate to benzodiazepines.20,33 Maintenance lorazepam has been effective in reducing relapse and recurrence.11 There are no controlled studies of maintenance treatment with benzodiazepines, but clinical reports suggest that doses in the range of 4 to 10 mg/d are effective.32
Continue to: ECT was used for catatonia in 1934...
ECT was first used for catatonia in 1934, when Laszlo Meduna used chemically induced seizures in catatonic patients who had been on tube feeding for months and no longer needed it after treatment.6,7 As was true for other disorders, this approach was replaced by ECT.7 In various case series, the effectiveness of ECT in catatonia has been 53% to 100%.7,13,15 Right unilateral ECT has been reported to be effective with 1 treatment.21 However, the best-established approach is with bitemporal ECT with a suprathreshold stimulus,9 usually with an acute course of 6 to 20 treatments.20 ECT has been reported to be equally safe and effective in adolescents and adults.34 Continued ECT is usually necessary until the patient has returned to baseline.9
ECT usually is recommended within 24 hours for treatment-resistant malignant catatonia or refusal to eat or drink, and within 2 to 3 days if medications are not sufficiently effective in other forms of catatonia.12,15,20 If ECT is initiated after a benzodiazepine trial, the benzodiazepine antagonist flumazenil is administered first to reverse the anticonvulsant effect.9 Some experts recommend using a muscle relaxant other than succinylcholine in the presence of evidence of muscle damage.7
Alternatives to benzodiazepines and ECT. Based on case reports, the treatments described in Table 813,15,17,20,25 have been used for patients with catatonia who do not tolerate or respond to standard treatments. The largest number of case reports have been with NMDA antagonists, while the presumed involvement of reduced dopamine signaling suggests that dopaminergic medications should be helpful. Dantrolene, which blocks release of calcium from intracellular stores and has been used to treat malignant hyperthermia, is sometimes used for NMS, often with disappointing results.

Whereas first-generation antipsychotics definitely increase the risk of catatonia and second-generation antipsychotics (SGAs) probably do so, SGAs are sometimes necessary to treat persistent psychosis in patients with schizophrenia who develop catatonia. Of these medications, clozapine may be most desirable because of low potency for dopamine receptor blockade and modulation of glutamatergic signaling. Partial dopamine agonism by aripiprazole, and the potential for increased subcortical prefrontal dopamine release resulting from serotonin 5HT2A antagonism and 5HT1A agonism by other SGAs, could also be helpful or at least not harmful in catatonia. Lorazepam is usually administered along with these medications to ameliorate treatment-emergent exacerbation of catatonia.
There are no controlled studies of any of these treatments. Based on case reports, most experts would recommend initiating treatment of catatonia with lorazepam, followed by ECT if necessary or in the presence of life-threatening catatonia. If ECT is not available, ineffective, or not tolerated, the first alternatives to be considered would be an NMDA antagonist or an anticonvulsant.20
Continue to: Course varies by patient, underlying cause
Course varies by patient, underlying cause
The response to benzodiazepines or ECT can vary from episode to episode11 and is similar in adults and younger patients.22 Many patients recover completely after a single episode, while relapse after remission occurs repeatedly in periodic catatonia, which involves chronic alternating stupor and excitement waxing and waning over years.11 Relapses may occur frequently, or every few years.11 Some cases of catatonia initially have an episodic course and become chronic and deteriorating, possibly paralleling the original descriptions of the natural history of untreated catatonia, while malignant catatonia can be complicated by medical morbidity or death.4 The long-term prognosis generally depends on the underlying cause of catatonia.5
Bottom Line
Much more common than many clinicians realize, catatonia can be overlooked because symptoms can mimic or overlap with features of an underlying medical or neurologic disorder. Suspect catatonia when one of these illnesses has an unexpected course or an inadequate treatment response. Be alert to characteristic changes in behavior and speech. A benzodiazepine challenge can be used to diagnose and begin treatment of catatonia. Consider electroconvulsive therapy sooner rather than later, especially for severely ill patients.
Related Resources
- Gibson RC, Walcott G. Benzodiazepines for catatonia in people with schizophrenia and other serious mental illnesses. Cochrane Database Syst Rev. 2008;(4):CD006570.
- Newcastle University. Catatonia. https://youtu.be/_s1lzxHRO4U.
Drug Brand Names
Amantadine • Symmetrel
Amobarbital • Amytal
Aripiprazole • Abilify
Azithromycin • Zithromax
Baclofen • Lioresal
Benztropine • Cogentin
Carbamazepine • Carbatrol, Tegretol
Carbidopa/levodopa • Sinemet
Ciprofloxacin • Cipro
Clozapine • Clozaril
Dantrolene • Dantrium
Dexamethasone • Decadron
Dextromethorphan/quinidine • Neudexta
Diazepam • Valium
Disulfiram • Antabuse
Flumazenil • Romazicon
Fluoxetine • Prozac
Fluvoxamine • Luvox
Levetiracetam • Keppra
Lithium • Eskalith, Lithobid
Lorazepam • Ativan
Memantine • Namenda
Methylphenidate • Ritalin
Minocycline • Minocin
Olanzapine • Zyprexa
Risperidone • Risperdal
Succinylcholine • Anectine
Topiramate • Topamax
Trihexyphenidyl • Artane
Valproate • Depakote
Ziprasidone • Geodon
Zolpidem • Ambien
1. Kahlbaum KL. Catatonia. Baltimore, MD: John Hopkins University Press; 1973.
2. Kahlbaum KL. Die Katatonie oder das Spannungsirresein. Berlin: Hirschwald; 1874.
3. Tang VM, Duffin J. Catatonia in the history of psychiatry: construction and deconstruction of a disease concept. Perspect Biol Med. 2014;57(4):524-537.
4. Carroll BT. Kahlbaum’s catatonia revisited. Psychiatry Clin Neurosci. 2001;55(5):431-436.
5. Taylor MA, Fink M. Catatonia in psychiatric classification: a home of its own. Am J Psychiatry. 2003;160(7):1233-1241.
6. Fink M, Fricchione GL, Rummans T, et al. Catatonia is a systemic medical syndrome. Acta Psychiatr Scand. 2016;133(3):250-251.
7. Medda P, Toni C, Luchini F, et al. Catatonia in 26 patients with bipolar disorder: clinical features and response to electroconvulsive therapy. Bipolar Disord. 2015;17(8):892-901.
8. Mazzone L, Postorino V, Valeri G, et al. Catatonia in patients with autism: prevalence and management. CNS Drugs. 2014;28(3):205-215.
9. Fink M, Kellner CH, McCall WV. Optimizing ECT technique in treating catatonia. J ECT. 2016;32(3):149-150.
10. Kocha H, Moriguchi S, Mimura M. Revisiting the concept of late catatonia. Compr Psychiatry. 2014;55(7):1485-1490.
11. Lin CC, Hung YL, Tsai MC, et al. Relapses and recurrences of catatonia: 30-case analysis and literature review. Compr Psychiatry. 2016;66:157-165.
12. Saddawi-Konefka D, Berg SM, Nejad SH, et al. Catatonia in the ICU: An important and underdiagnosed cause of altered mental status. A case series and review of the literature. Crit Care Med. 2013;42(3):e234-e241.
13. Wijemanne S, Jankovic J. Movement disorders in catatonia. J Neurol Neurosurg Psychiatry. 2015;86(8):825-832.
14. Grover S, Chakrabarti S, Ghormode D, et al. Catatonia in inpatients with psychiatric disorders: a comparison of schizophrenia and mood disorders. Psychiatry Res. 2015;229(3):919-925.
15. Oldham MA, Lee HB. Catatonia vis-à-vis delirium: the significance of recognizing catatonia in altered mental status. Gen Hosp Psychiatry. 2015;37(6):554-559.
16. Tuerlings JH, van Waarde JA, Verwey B. A retrospective study of 34 catatonic patients: analysis of clinical ‘care and treatment. Gen Hosp Psychiatry. 2010;32(6):631-635.
17. Ohi K, Kuwata A, Shimada T, et al. Response to benzodiazepines and the clinical course in malignant catatonia associated with schizophrenia: a case report. Medicine (Baltimore). 2017;96(16):e6566. doi: 10.1097/MD.0000000000006566.
18. Komatsu T, Nomura T, Takami H, et al. Catatonic symptoms appearing before autonomic symptoms help distinguish neuroleptic malignant syndrome from malignant catatonia. Intern Med. 2016;55(19):2893-2897.
19. Lang FU, Lang S, Becker T, et al. Neuroleptic malignant syndrome or catatonia? Trying to solve the catatonic dilemma. Psychopharmacology (Berl). 2015;232(1):1-5.
20. Beach SR, Gomez-Bernal F, Huffman JC, et al. Alternative treatment strategies for catatonia: a systematic review. Gen Hosp Psychiatry. 2017;48:1-19.
21. Kugler JL, Hauptman AJ, Collier SJ, et al. Treatment of catatonia with ultrabrief right unilateral electroconvulsive therapy: a case series. J ECT. 2015;31(3):192-196.
22. Raffin M, Zugaj-Bensaou L, Bodeau N, et al. Treatment use in a prospective naturalistic cohort of children and adolescents with catatonia. Eur Child Adolesc Psychiatry. 2015;24(4):441-449.
23. DeJong H, Bunton P, Hare DJ. A systematic review of interventions used to treat catatonic symptoms in people with autistic spectrum disorders. J Autism Dev Disord. 2014;44(9):2127-2136.
24. Wachtel L, Commins E, Park MH, et al. Neuroleptic malignant syndrome and delirious mania as malignant catatonia in autism: prompt relief with electroconvulsive therapy. Acta Psychiatr Scand. 2015;132(4):319-320.
25. Fink M, Taylor MA. Catatonia: subtype or syndrome in DSM? Am J Psychiatry. 2006;163(11):1875-1876.
26. Khan M, Pace L, Truong A, et al. Catatonia secondary to synthetic cannabinoid use in two patients with no previous psychosis. Am J Addictions. 2016;25(1):25-27.
27. Komatsu T, Nomura T, Takami H, et al. Catatonic symptoms appearing before autonomic symptoms help distinguish neuroleptic malignant syndrome from malignant catatonia. Intern Med. 2016;55(19):2893-2897.
28. Diagnostic and statistical manual of mental disorders, 5th ed. Washington, DC: American Psychiatric Association; 2013.
29. Wilson JE, Niu K, Nicolson SE, et al. The diagnostic criteria and structure of catatonia. Schizophr Res. 2015;164(1-3):256-262.
30. Ducharme S, Dickerson BC, Larvie M, et al. Differentiating frontotemporal dementia from catatonia: a complex neuropsychiatric challenge. J Neuropsychiatry Clin Neurosci. 2015;27(2):e174-e176.
31. Narayanaswamy JC, Tibrewal P, Zutshi A, et al. Clinical predictors of response to treatment in catatonia. Gen Hosp Psychiatry. 2012;34(3):312-316.
32. Thamizh JS, Harshini M, Selvakumar N, et al. Maintenance lorazepam for treatment of recurrent catatonic states: a case series and implications. Asian J Psychiatr. 2016;22:147-149
33. Ungvari GS, Chiu HF, Chow LY, et al. Lorazepam for chronic catatonia: a randomized, double-blind, placebo-controlled cross-over study. Psychopharmacology (Berl). 1999;142(4):393-398.
34. Flamarique I, Baeza I, de la Serna E, et al. Long-term effectiveness of electroconvulsive therapy in adolescents with schizophrenia spectrum disorders. Eur Child Adolesc Psychiatry. 2015;24(5):517-524.
1. Kahlbaum KL. Catatonia. Baltimore, MD: John Hopkins University Press; 1973.
2. Kahlbaum KL. Die Katatonie oder das Spannungsirresein. Berlin: Hirschwald; 1874.
3. Tang VM, Duffin J. Catatonia in the history of psychiatry: construction and deconstruction of a disease concept. Perspect Biol Med. 2014;57(4):524-537.
4. Carroll BT. Kahlbaum’s catatonia revisited. Psychiatry Clin Neurosci. 2001;55(5):431-436.
5. Taylor MA, Fink M. Catatonia in psychiatric classification: a home of its own. Am J Psychiatry. 2003;160(7):1233-1241.
6. Fink M, Fricchione GL, Rummans T, et al. Catatonia is a systemic medical syndrome. Acta Psychiatr Scand. 2016;133(3):250-251.
7. Medda P, Toni C, Luchini F, et al. Catatonia in 26 patients with bipolar disorder: clinical features and response to electroconvulsive therapy. Bipolar Disord. 2015;17(8):892-901.
8. Mazzone L, Postorino V, Valeri G, et al. Catatonia in patients with autism: prevalence and management. CNS Drugs. 2014;28(3):205-215.
9. Fink M, Kellner CH, McCall WV. Optimizing ECT technique in treating catatonia. J ECT. 2016;32(3):149-150.
10. Kocha H, Moriguchi S, Mimura M. Revisiting the concept of late catatonia. Compr Psychiatry. 2014;55(7):1485-1490.
11. Lin CC, Hung YL, Tsai MC, et al. Relapses and recurrences of catatonia: 30-case analysis and literature review. Compr Psychiatry. 2016;66:157-165.
12. Saddawi-Konefka D, Berg SM, Nejad SH, et al. Catatonia in the ICU: An important and underdiagnosed cause of altered mental status. A case series and review of the literature. Crit Care Med. 2013;42(3):e234-e241.
13. Wijemanne S, Jankovic J. Movement disorders in catatonia. J Neurol Neurosurg Psychiatry. 2015;86(8):825-832.
14. Grover S, Chakrabarti S, Ghormode D, et al. Catatonia in inpatients with psychiatric disorders: a comparison of schizophrenia and mood disorders. Psychiatry Res. 2015;229(3):919-925.
15. Oldham MA, Lee HB. Catatonia vis-à-vis delirium: the significance of recognizing catatonia in altered mental status. Gen Hosp Psychiatry. 2015;37(6):554-559.
16. Tuerlings JH, van Waarde JA, Verwey B. A retrospective study of 34 catatonic patients: analysis of clinical ‘care and treatment. Gen Hosp Psychiatry. 2010;32(6):631-635.
17. Ohi K, Kuwata A, Shimada T, et al. Response to benzodiazepines and the clinical course in malignant catatonia associated with schizophrenia: a case report. Medicine (Baltimore). 2017;96(16):e6566. doi: 10.1097/MD.0000000000006566.
18. Komatsu T, Nomura T, Takami H, et al. Catatonic symptoms appearing before autonomic symptoms help distinguish neuroleptic malignant syndrome from malignant catatonia. Intern Med. 2016;55(19):2893-2897.
19. Lang FU, Lang S, Becker T, et al. Neuroleptic malignant syndrome or catatonia? Trying to solve the catatonic dilemma. Psychopharmacology (Berl). 2015;232(1):1-5.
20. Beach SR, Gomez-Bernal F, Huffman JC, et al. Alternative treatment strategies for catatonia: a systematic review. Gen Hosp Psychiatry. 2017;48:1-19.
21. Kugler JL, Hauptman AJ, Collier SJ, et al. Treatment of catatonia with ultrabrief right unilateral electroconvulsive therapy: a case series. J ECT. 2015;31(3):192-196.
22. Raffin M, Zugaj-Bensaou L, Bodeau N, et al. Treatment use in a prospective naturalistic cohort of children and adolescents with catatonia. Eur Child Adolesc Psychiatry. 2015;24(4):441-449.
23. DeJong H, Bunton P, Hare DJ. A systematic review of interventions used to treat catatonic symptoms in people with autistic spectrum disorders. J Autism Dev Disord. 2014;44(9):2127-2136.
24. Wachtel L, Commins E, Park MH, et al. Neuroleptic malignant syndrome and delirious mania as malignant catatonia in autism: prompt relief with electroconvulsive therapy. Acta Psychiatr Scand. 2015;132(4):319-320.
25. Fink M, Taylor MA. Catatonia: subtype or syndrome in DSM? Am J Psychiatry. 2006;163(11):1875-1876.
26. Khan M, Pace L, Truong A, et al. Catatonia secondary to synthetic cannabinoid use in two patients with no previous psychosis. Am J Addictions. 2016;25(1):25-27.
27. Komatsu T, Nomura T, Takami H, et al. Catatonic symptoms appearing before autonomic symptoms help distinguish neuroleptic malignant syndrome from malignant catatonia. Intern Med. 2016;55(19):2893-2897.
28. Diagnostic and statistical manual of mental disorders, 5th ed. Washington, DC: American Psychiatric Association; 2013.
29. Wilson JE, Niu K, Nicolson SE, et al. The diagnostic criteria and structure of catatonia. Schizophr Res. 2015;164(1-3):256-262.
30. Ducharme S, Dickerson BC, Larvie M, et al. Differentiating frontotemporal dementia from catatonia: a complex neuropsychiatric challenge. J Neuropsychiatry Clin Neurosci. 2015;27(2):e174-e176.
31. Narayanaswamy JC, Tibrewal P, Zutshi A, et al. Clinical predictors of response to treatment in catatonia. Gen Hosp Psychiatry. 2012;34(3):312-316.
32. Thamizh JS, Harshini M, Selvakumar N, et al. Maintenance lorazepam for treatment of recurrent catatonic states: a case series and implications. Asian J Psychiatr. 2016;22:147-149
33. Ungvari GS, Chiu HF, Chow LY, et al. Lorazepam for chronic catatonia: a randomized, double-blind, placebo-controlled cross-over study. Psychopharmacology (Berl). 1999;142(4):393-398.
34. Flamarique I, Baeza I, de la Serna E, et al. Long-term effectiveness of electroconvulsive therapy in adolescents with schizophrenia spectrum disorders. Eur Child Adolesc Psychiatry. 2015;24(5):517-524.
FDA approves Perseris for schizophrenia
The Food and Drug Administration has approved Perseris, a once-monthly, long-acting injectable formulation of risperidone, for the treatment of schizophrenia in adults, according to a press release from the drug’s developer, Indivior.
The depot formulation of the atypical antipsychotic, administered subcutaneously, provides sustained levels of risperidone for 1 month; the process of injecting it also releases some of the drug, which helps achieve peak plasma levels within the first 4-6 hours. There is no need for either loading doses or supplemental oral doses with Perseris.
Efficacy was based on a phase 3, randomized, double-blind, placebo-controlled, 8-week study of 354 patients. The primary endpoint was improvement in Positive and Negative Syndrome Scale by day 57. Safety was evaluated in 814 patients who had participated in clinical trials of Perseris and was in line with the known safety profile of risperidone.
The most common adverse reactions, occurring in more than 5% of patients, were increased weight, sedation/somnolence, and musculoskeletal pain. Other risks included neuroleptic malignant syndrome, tardive dyskinesia, and hyperprolactinemia. Full prescribing information can be found on the manufacturer’s website.
The Food and Drug Administration has approved Perseris, a once-monthly, long-acting injectable formulation of risperidone, for the treatment of schizophrenia in adults, according to a press release from the drug’s developer, Indivior.
The depot formulation of the atypical antipsychotic, administered subcutaneously, provides sustained levels of risperidone for 1 month; the process of injecting it also releases some of the drug, which helps achieve peak plasma levels within the first 4-6 hours. There is no need for either loading doses or supplemental oral doses with Perseris.
Efficacy was based on a phase 3, randomized, double-blind, placebo-controlled, 8-week study of 354 patients. The primary endpoint was improvement in Positive and Negative Syndrome Scale by day 57. Safety was evaluated in 814 patients who had participated in clinical trials of Perseris and was in line with the known safety profile of risperidone.
The most common adverse reactions, occurring in more than 5% of patients, were increased weight, sedation/somnolence, and musculoskeletal pain. Other risks included neuroleptic malignant syndrome, tardive dyskinesia, and hyperprolactinemia. Full prescribing information can be found on the manufacturer’s website.
The Food and Drug Administration has approved Perseris, a once-monthly, long-acting injectable formulation of risperidone, for the treatment of schizophrenia in adults, according to a press release from the drug’s developer, Indivior.
The depot formulation of the atypical antipsychotic, administered subcutaneously, provides sustained levels of risperidone for 1 month; the process of injecting it also releases some of the drug, which helps achieve peak plasma levels within the first 4-6 hours. There is no need for either loading doses or supplemental oral doses with Perseris.
Efficacy was based on a phase 3, randomized, double-blind, placebo-controlled, 8-week study of 354 patients. The primary endpoint was improvement in Positive and Negative Syndrome Scale by day 57. Safety was evaluated in 814 patients who had participated in clinical trials of Perseris and was in line with the known safety profile of risperidone.
The most common adverse reactions, occurring in more than 5% of patients, were increased weight, sedation/somnolence, and musculoskeletal pain. Other risks included neuroleptic malignant syndrome, tardive dyskinesia, and hyperprolactinemia. Full prescribing information can be found on the manufacturer’s website.
Fetal exposure to folic acid may reduce youth psychosis risk
Gestational exposure to grain products fortified with folic acid resulted in delayed thinning of the cerebral cortex, and thus may protect against later psychosis, according to results published in JAMA Psychiatry.
In a retrospective, observational cohort study of 292 patients aged 8-18 years, increases in cortical thickness were associated with folic acid exposure in the bilateral frontal and temporal regions of the brain (9.9%-11.6%; corrected P less than .001 to P = .03). Delayed, age-associated thinning in temporal and parietal regions was also observed (beta = –11.1 to –13.9; corrected P = .002), reported Hamdi Eryilmaz, PhD, of the department of psychiatry at Massachusetts General Hospital, Boston, and his coauthors.
The study authors first observed MRI scans in the Massachusetts General Hospital (MGH) cohort, which included the 292 patients aged 8-18 years, and then conducted subsequent analyses on two additional cohorts – the Philadelphia Neurodevelopmental Cohort (PNC) and the National Institutes of Health MRI Study of Normal Brain Development (NIH) cohort – to test the reliability and specificity of cortical development associations and the relevance of MRI changes to psychopathological characteristics, the authors wrote.
Using the U.S. implementation of folic acid fortification in grain foods in 1996 and 1997 to define exposure status, the investigators identified MRI scans of 97 prerollout (not exposed), 96 rollout (partly exposed), and 99 postrollout (fully exposed) unique individuals in the MGH group between January 2005 and March 2015, for patients born between January 1993 and December 2001. They also collected information on demographics; reason for MRI scan; and prior use of psychotropic medications, folic acid, or multivitamins.
The PNC cohort consisted of 861 patients, also aged 8-18 years, from community health settings in Philadelphia who had an MRI assessment and a clinical assessment of psychiatric symptoms. The NIH comparison cohort included 217 patients recruited from six health sites across the United States and born before the fortification rollout.
The MGH analysis contrasted mean cortical thickness and linear and quadratic models of age-associated change in cortical thickness within the fully exposed and nonexposed groups. PNC and NIH analyses evaluated quadratic associations of age with cortical thickness.
In the MGH cohort, increases in cortical thickness were associated with folic acid exposure in the bilateral frontal and temporal regions of the brain (9.9%-11.6%; corrected P less than .001 to P = .03), and delayed, age-associated thinning in the temporal and parietal regions was observed (corrected P = .002). Thickness was higher in the fully exposed group, compared with those in the nonexposed group. The effect was intermediate in the partly exposed group, Dr. Eryilmaz and his coauthors wrote.
In the PNC cohort, delayed, age-related thinning was observed in four clusters overlapping with the MGH analysis: left frontal, right inferior temporal, left inferior parietal, and right inferior parietal. In addition, onset of cortical thinning was found to be between 13.0 and 14.3 years of age in least-square regression analysis. In the comparison NIH cohort, though, only the left frontal cortex demonstrated significant quadratic thinning; the break point occurred at a significantly younger age, compared with those in the PNC group (P less than .001).
In the PNC group, 248 of the 861 patients included in the MRI analysis were typically developing, 199 had a psychosis diagnosis, 105 had attenuated psychotic symptoms, and 309 had various other psychiatric conditions. Best-fit local thinning slopes were calculated for each participant for each region with postfortification thinning, and in three of four regions, less-negative local slopes were associated with significantly reduced adjusted odds of psychosis spectrum diagnosis (odds ratio, 0.37-0.59; P less than .001 to P = .02), the authors reported.
The findings confirm that “fetal exposure to population-wide folic acid fortification was associated with subsequent alterations in cortical development among school-aged youths,” Dr. Eryilmaz and his coauthors wrote. “These cortical changes were associated with reduced risk of psychosis.”
The results also suggest that the protective effects of folic acid in gestation “may extend beyond prevention of neural tube defects and span neurodevelopment during childhood and adolescence,” they concluded.
The study was funded by MQ: Transforming Mental Health, with support from grants from several additional sources, including the National Institutes of Health.
SOURCE: Eryilmaz H et al. JAMA Psychiatry. 2018 Jul 3. doi: 10.1001/jamapsychiatry.2018.1381.
The results of this study complement previous studies in the Netherlands “addressing similar questions using plasma levels of folate in maternal blood during pregnancy,” Tomáš Paus, MD, PhD, wrote in an editorial (JAMA Psychiatry. 2018 Jul 3. doi: 10.1001/jamapsychiatry.2018.1255) published with the study. However, he added, “it is unfortunate that the authors did not report values of cortical surface area in the different groups of individuals studied.”
Previous studies have shown that the children of mothers with high folic acid levels during pregnancy showed greater head growth and larger brain volumes at 6-8 years of age, Dr. Paus wrote, adding that future research should explore the possibility that exposure to folic acid induces effects such as DNA methylation, that may persist over time.
“This folate-methylation hypothesis needs to be tested empirically in large data sets, ideally in conjunction with relevant brain phenotypes, to replicate and expand the initial findings reported” in this study, he concluded.
Dr. Paus is affiliated with the department of psychology at the Rotman Research Institute, Toronto. He reported no conflicts of interest.
The results of this study complement previous studies in the Netherlands “addressing similar questions using plasma levels of folate in maternal blood during pregnancy,” Tomáš Paus, MD, PhD, wrote in an editorial (JAMA Psychiatry. 2018 Jul 3. doi: 10.1001/jamapsychiatry.2018.1255) published with the study. However, he added, “it is unfortunate that the authors did not report values of cortical surface area in the different groups of individuals studied.”
Previous studies have shown that the children of mothers with high folic acid levels during pregnancy showed greater head growth and larger brain volumes at 6-8 years of age, Dr. Paus wrote, adding that future research should explore the possibility that exposure to folic acid induces effects such as DNA methylation, that may persist over time.
“This folate-methylation hypothesis needs to be tested empirically in large data sets, ideally in conjunction with relevant brain phenotypes, to replicate and expand the initial findings reported” in this study, he concluded.
Dr. Paus is affiliated with the department of psychology at the Rotman Research Institute, Toronto. He reported no conflicts of interest.
The results of this study complement previous studies in the Netherlands “addressing similar questions using plasma levels of folate in maternal blood during pregnancy,” Tomáš Paus, MD, PhD, wrote in an editorial (JAMA Psychiatry. 2018 Jul 3. doi: 10.1001/jamapsychiatry.2018.1255) published with the study. However, he added, “it is unfortunate that the authors did not report values of cortical surface area in the different groups of individuals studied.”
Previous studies have shown that the children of mothers with high folic acid levels during pregnancy showed greater head growth and larger brain volumes at 6-8 years of age, Dr. Paus wrote, adding that future research should explore the possibility that exposure to folic acid induces effects such as DNA methylation, that may persist over time.
“This folate-methylation hypothesis needs to be tested empirically in large data sets, ideally in conjunction with relevant brain phenotypes, to replicate and expand the initial findings reported” in this study, he concluded.
Dr. Paus is affiliated with the department of psychology at the Rotman Research Institute, Toronto. He reported no conflicts of interest.
Gestational exposure to grain products fortified with folic acid resulted in delayed thinning of the cerebral cortex, and thus may protect against later psychosis, according to results published in JAMA Psychiatry.
In a retrospective, observational cohort study of 292 patients aged 8-18 years, increases in cortical thickness were associated with folic acid exposure in the bilateral frontal and temporal regions of the brain (9.9%-11.6%; corrected P less than .001 to P = .03). Delayed, age-associated thinning in temporal and parietal regions was also observed (beta = –11.1 to –13.9; corrected P = .002), reported Hamdi Eryilmaz, PhD, of the department of psychiatry at Massachusetts General Hospital, Boston, and his coauthors.
The study authors first observed MRI scans in the Massachusetts General Hospital (MGH) cohort, which included the 292 patients aged 8-18 years, and then conducted subsequent analyses on two additional cohorts – the Philadelphia Neurodevelopmental Cohort (PNC) and the National Institutes of Health MRI Study of Normal Brain Development (NIH) cohort – to test the reliability and specificity of cortical development associations and the relevance of MRI changes to psychopathological characteristics, the authors wrote.
Using the U.S. implementation of folic acid fortification in grain foods in 1996 and 1997 to define exposure status, the investigators identified MRI scans of 97 prerollout (not exposed), 96 rollout (partly exposed), and 99 postrollout (fully exposed) unique individuals in the MGH group between January 2005 and March 2015, for patients born between January 1993 and December 2001. They also collected information on demographics; reason for MRI scan; and prior use of psychotropic medications, folic acid, or multivitamins.
The PNC cohort consisted of 861 patients, also aged 8-18 years, from community health settings in Philadelphia who had an MRI assessment and a clinical assessment of psychiatric symptoms. The NIH comparison cohort included 217 patients recruited from six health sites across the United States and born before the fortification rollout.
The MGH analysis contrasted mean cortical thickness and linear and quadratic models of age-associated change in cortical thickness within the fully exposed and nonexposed groups. PNC and NIH analyses evaluated quadratic associations of age with cortical thickness.
In the MGH cohort, increases in cortical thickness were associated with folic acid exposure in the bilateral frontal and temporal regions of the brain (9.9%-11.6%; corrected P less than .001 to P = .03), and delayed, age-associated thinning in the temporal and parietal regions was observed (corrected P = .002). Thickness was higher in the fully exposed group, compared with those in the nonexposed group. The effect was intermediate in the partly exposed group, Dr. Eryilmaz and his coauthors wrote.
In the PNC cohort, delayed, age-related thinning was observed in four clusters overlapping with the MGH analysis: left frontal, right inferior temporal, left inferior parietal, and right inferior parietal. In addition, onset of cortical thinning was found to be between 13.0 and 14.3 years of age in least-square regression analysis. In the comparison NIH cohort, though, only the left frontal cortex demonstrated significant quadratic thinning; the break point occurred at a significantly younger age, compared with those in the PNC group (P less than .001).
In the PNC group, 248 of the 861 patients included in the MRI analysis were typically developing, 199 had a psychosis diagnosis, 105 had attenuated psychotic symptoms, and 309 had various other psychiatric conditions. Best-fit local thinning slopes were calculated for each participant for each region with postfortification thinning, and in three of four regions, less-negative local slopes were associated with significantly reduced adjusted odds of psychosis spectrum diagnosis (odds ratio, 0.37-0.59; P less than .001 to P = .02), the authors reported.
The findings confirm that “fetal exposure to population-wide folic acid fortification was associated with subsequent alterations in cortical development among school-aged youths,” Dr. Eryilmaz and his coauthors wrote. “These cortical changes were associated with reduced risk of psychosis.”
The results also suggest that the protective effects of folic acid in gestation “may extend beyond prevention of neural tube defects and span neurodevelopment during childhood and adolescence,” they concluded.
The study was funded by MQ: Transforming Mental Health, with support from grants from several additional sources, including the National Institutes of Health.
SOURCE: Eryilmaz H et al. JAMA Psychiatry. 2018 Jul 3. doi: 10.1001/jamapsychiatry.2018.1381.
Gestational exposure to grain products fortified with folic acid resulted in delayed thinning of the cerebral cortex, and thus may protect against later psychosis, according to results published in JAMA Psychiatry.
In a retrospective, observational cohort study of 292 patients aged 8-18 years, increases in cortical thickness were associated with folic acid exposure in the bilateral frontal and temporal regions of the brain (9.9%-11.6%; corrected P less than .001 to P = .03). Delayed, age-associated thinning in temporal and parietal regions was also observed (beta = –11.1 to –13.9; corrected P = .002), reported Hamdi Eryilmaz, PhD, of the department of psychiatry at Massachusetts General Hospital, Boston, and his coauthors.
The study authors first observed MRI scans in the Massachusetts General Hospital (MGH) cohort, which included the 292 patients aged 8-18 years, and then conducted subsequent analyses on two additional cohorts – the Philadelphia Neurodevelopmental Cohort (PNC) and the National Institutes of Health MRI Study of Normal Brain Development (NIH) cohort – to test the reliability and specificity of cortical development associations and the relevance of MRI changes to psychopathological characteristics, the authors wrote.
Using the U.S. implementation of folic acid fortification in grain foods in 1996 and 1997 to define exposure status, the investigators identified MRI scans of 97 prerollout (not exposed), 96 rollout (partly exposed), and 99 postrollout (fully exposed) unique individuals in the MGH group between January 2005 and March 2015, for patients born between January 1993 and December 2001. They also collected information on demographics; reason for MRI scan; and prior use of psychotropic medications, folic acid, or multivitamins.
The PNC cohort consisted of 861 patients, also aged 8-18 years, from community health settings in Philadelphia who had an MRI assessment and a clinical assessment of psychiatric symptoms. The NIH comparison cohort included 217 patients recruited from six health sites across the United States and born before the fortification rollout.
The MGH analysis contrasted mean cortical thickness and linear and quadratic models of age-associated change in cortical thickness within the fully exposed and nonexposed groups. PNC and NIH analyses evaluated quadratic associations of age with cortical thickness.
In the MGH cohort, increases in cortical thickness were associated with folic acid exposure in the bilateral frontal and temporal regions of the brain (9.9%-11.6%; corrected P less than .001 to P = .03), and delayed, age-associated thinning in the temporal and parietal regions was observed (corrected P = .002). Thickness was higher in the fully exposed group, compared with those in the nonexposed group. The effect was intermediate in the partly exposed group, Dr. Eryilmaz and his coauthors wrote.
In the PNC cohort, delayed, age-related thinning was observed in four clusters overlapping with the MGH analysis: left frontal, right inferior temporal, left inferior parietal, and right inferior parietal. In addition, onset of cortical thinning was found to be between 13.0 and 14.3 years of age in least-square regression analysis. In the comparison NIH cohort, though, only the left frontal cortex demonstrated significant quadratic thinning; the break point occurred at a significantly younger age, compared with those in the PNC group (P less than .001).
In the PNC group, 248 of the 861 patients included in the MRI analysis were typically developing, 199 had a psychosis diagnosis, 105 had attenuated psychotic symptoms, and 309 had various other psychiatric conditions. Best-fit local thinning slopes were calculated for each participant for each region with postfortification thinning, and in three of four regions, less-negative local slopes were associated with significantly reduced adjusted odds of psychosis spectrum diagnosis (odds ratio, 0.37-0.59; P less than .001 to P = .02), the authors reported.
The findings confirm that “fetal exposure to population-wide folic acid fortification was associated with subsequent alterations in cortical development among school-aged youths,” Dr. Eryilmaz and his coauthors wrote. “These cortical changes were associated with reduced risk of psychosis.”
The results also suggest that the protective effects of folic acid in gestation “may extend beyond prevention of neural tube defects and span neurodevelopment during childhood and adolescence,” they concluded.
The study was funded by MQ: Transforming Mental Health, with support from grants from several additional sources, including the National Institutes of Health.
SOURCE: Eryilmaz H et al. JAMA Psychiatry. 2018 Jul 3. doi: 10.1001/jamapsychiatry.2018.1381.
FROM JAMA PSYCHIATRY
Key clinical point: Gestational exposure to grain products fortified with folic acid resulted in delayed thinning of the cerebral cortex, and thus may protect against later psychosis.
Major finding: Increases in cortical thickness were associated with folic acid exposure in the bilateral frontal and temporal regions of the brain (9.9%-11.6%; corrected P less than .001 to P = .03).
Study details: A retrospective, observational cohort study of 292 patients aged 8-18 years.
Disclosures: The study was funded by MQ: Transforming Mental Health, with support from grants from several additional sources, including the National Institutes of Health.
Source: Eryilmaz H et al. JAMA Psychiatry. 2018 Jul 3. doi: 10.1001/jamapsychiatry.2018.1381.
FDA approves Aristada Initio for schizophrenia
The Food and Drug Administration has approved aripiprazole lauroxil (Aristada Initio) for the initiation of aripiprazole lauroxil (Aristada) for treating schizophrenia in adults, the drug’s developer, Alkermes, announced July 2 in a press release.
“,” Craig Hopkinson, MD, chief medical officer at Alkermes, said in the press release.
The standard initiation regimen for aripiprazole lauroxil previously was 21 consecutive days of oral aripiprazole starting with the first dose of Aristada. The alternative initiation regimen allows patients to achieve “relevant levels of aripiprazole within 4 days of initiation,” according to Alkermes. The result gives health care providers “an additional tool to support patients.”
One important advantage of Aristada Initio is that, in addition to monthly and 6-week dosing options, it offers a 2-month option.
Aristada and Aristada Initio both contain aripiprazole lauroxil, but the drugs are not interchangeable because they have different pharmacokinetic profiles, the company said. In addition, Aristada Initio is to be administered a single time only.
Aristada Initio has exhibited some of the same adverse events as other atypical antipsychotics, including neuroleptic malignant syndrome, tardive dyskinesia, and metabolic changes. Injection-site reactions also have been observed.
Aristada Initio is expected to become available by mid-July.
Full prescribing information and boxed warnings can be found on the Alkermes website.
The Food and Drug Administration has approved aripiprazole lauroxil (Aristada Initio) for the initiation of aripiprazole lauroxil (Aristada) for treating schizophrenia in adults, the drug’s developer, Alkermes, announced July 2 in a press release.
“,” Craig Hopkinson, MD, chief medical officer at Alkermes, said in the press release.
The standard initiation regimen for aripiprazole lauroxil previously was 21 consecutive days of oral aripiprazole starting with the first dose of Aristada. The alternative initiation regimen allows patients to achieve “relevant levels of aripiprazole within 4 days of initiation,” according to Alkermes. The result gives health care providers “an additional tool to support patients.”
One important advantage of Aristada Initio is that, in addition to monthly and 6-week dosing options, it offers a 2-month option.
Aristada and Aristada Initio both contain aripiprazole lauroxil, but the drugs are not interchangeable because they have different pharmacokinetic profiles, the company said. In addition, Aristada Initio is to be administered a single time only.
Aristada Initio has exhibited some of the same adverse events as other atypical antipsychotics, including neuroleptic malignant syndrome, tardive dyskinesia, and metabolic changes. Injection-site reactions also have been observed.
Aristada Initio is expected to become available by mid-July.
Full prescribing information and boxed warnings can be found on the Alkermes website.
The Food and Drug Administration has approved aripiprazole lauroxil (Aristada Initio) for the initiation of aripiprazole lauroxil (Aristada) for treating schizophrenia in adults, the drug’s developer, Alkermes, announced July 2 in a press release.
“,” Craig Hopkinson, MD, chief medical officer at Alkermes, said in the press release.
The standard initiation regimen for aripiprazole lauroxil previously was 21 consecutive days of oral aripiprazole starting with the first dose of Aristada. The alternative initiation regimen allows patients to achieve “relevant levels of aripiprazole within 4 days of initiation,” according to Alkermes. The result gives health care providers “an additional tool to support patients.”
One important advantage of Aristada Initio is that, in addition to monthly and 6-week dosing options, it offers a 2-month option.
Aristada and Aristada Initio both contain aripiprazole lauroxil, but the drugs are not interchangeable because they have different pharmacokinetic profiles, the company said. In addition, Aristada Initio is to be administered a single time only.
Aristada Initio has exhibited some of the same adverse events as other atypical antipsychotics, including neuroleptic malignant syndrome, tardive dyskinesia, and metabolic changes. Injection-site reactions also have been observed.
Aristada Initio is expected to become available by mid-July.
Full prescribing information and boxed warnings can be found on the Alkermes website.
Long-acting injectable antipsychotics: What to do about missed doses
Antipsychotic agents are the mainstay of treatment for patients with schizophrenia,1-3 and when taken regularly, they can greatly improve patient outcomes. Unfortunately, many studies have documented poor adherence to antipsychotic regimens in patients with schizophrenia, which often leads to an exacerbation of symptoms and preventable hospitalizations.4-8 In order to improve adherence, many clinicians prescribe long-acting injectable antipsychotics (LAIAs).
LAIAs help improve adherence, but these benefits are seen only in patients who receive their injections within a specific time frame.9-11 LAIAs administered outside of this time frame (missed doses) can lead to reoccurrence or exacerbation of symptoms. This article explains how to adequately manage missed LAIA doses.
First-generation long-acting injectable antipsychotics
Two first-generation antipsychotics are available as a long-acting injectable formulation: haloperidol decanoate and fluphenazine decanoate. Due to the increased risk of extrapyramidal symptoms, use of these agents have decreased, and they are often less preferred than second-generation LAIAs. Furthermore, unlike many of the newer second-generation LAIAs, first-generation LAIAs lack literature on how to manage missed doses. Therefore, clinicians should analyze the pharmacokinetic properties of these agents (Table 112-28), as well as the patient’s medical history and clinical presentation, in order to determine how best to address missed doses.

Haloperidol decanoate plasma concentrations peak approximately 6 days after the injection.12 The medication has a half-life of 3 weeks. One study found that haloperidol plasma concentrations were detectable 13 weeks after the discontinuation of haloperidol decanoate.17 This same study also found that the change in plasma levels from 3 to 6 weeks after the last dose was minimal.17 Based on these findings, Figure 1 summarizes our recommendations for addressing missed haloperidol decanoate doses.

Fluphenazine decanoate levels peak 24 hours after the injection.18 An estimated therapeutic range for fluphenazine is 0.2 to 2 ng/mL.21-25 One study that evaluated fluphenazine decanoate levels following discontinuation after reaching steady state found there was no significant difference in plasma levels 6 weeks after the last dose of fluphenazine, but a significant decrease in levels 8 to 12 weeks after the last dose.26 Other studies found that fluphenazine levels were detectable 21 to 24 weeks following fluphenazine decanoate discontinuation.27,28 Based on these findings, Figure 2 summarizes our recommendations for addressing missed fluphenazine decanoate doses.

Continue to: Second-generation LAIAs
Second-generation LAIAs
Six second-generation LAIAs are available in the United States. Compared with the first-generation LAIAs, second-generation LAIAs have more extensive guidance on how to address missed doses.
Risperidone long-acting injection. When addressing missed doses of risperidone long-acting injection, first determine whether the medication has reached steady state. Steady state occurs approximately after the fourth consecutive injection (approximately 2 months).29
If a patient missed a dose but has not reached steady state, he or she should receive the next dose as well as oral antipsychotic supplementation for 3 weeks.30 If the patient has reached steady state and if it has been ≤6 weeks since the last injection, give the next injection as soon as possible. However, if steady state has been reached and it has been >6 weeks since the last injection, give the next injection, along with 3 weeks of oral antipsychotic supplementation (Figure 3).

Paliperidone palmitate monthly long-acting injection. Once the initiation dosing phase of paliperidone palmitate monthly long-acting injection (PP1M) is completed, the maintenance dose is administered every 4 weeks. When addressing missed doses of PP1M, first determine whether the patient is in the initiation or maintenance dosing phase.31
Initiation phase. Patients are in the initiation dosing phase during the first 2 injections of PP1M. During the initiation phase, the patient first receives 234 mg and then 156 mg 1 week later, both in the deltoid muscle. One month later, the patient receives a maintenance dose of PP1M (in the deltoid or gluteal muscle). The second initiation injection may be given 4 days before or after the scheduled administration date. The initiation doses should be adjusted in patients with mild renal function (creatinine clearance 50 to 80 mL/min).31 Figure 4 summarizes the guidance for addressing a missed or delayed second injection during the initiation phase.
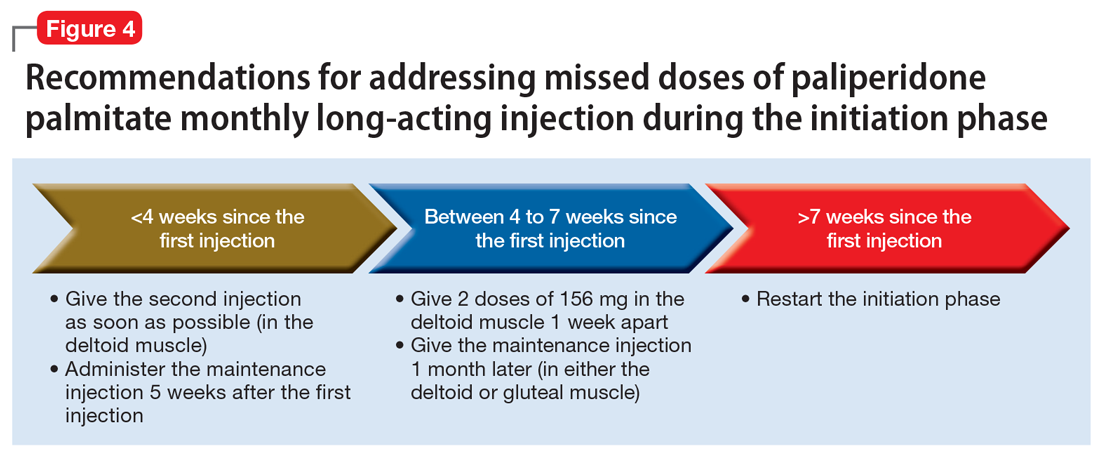
Continue to: Maintenance phase
Maintenance phase. During the maintenance phase, PP1M can be administered 7 days before or after the monthly due date. If the patient has missed a maintenance injection and it has been <6 weeks since the last dose, the maintenance injection can be given as soon as possible (Figure 5).31 If it has been 6 weeks to 6 months since the last injection, the patient should receive their prescribed maintenance dose as soon as possible and the same dose 1 week later, with both injections in the deltoid muscle. Following the second dose, the patient can resume their regular monthly maintenance schedule, in either the deltoid or gluteal muscle. For example, if the patient was maintained on 117 mg of PP1M and it had been 8 weeks since the last injection, the patient should receive 117 mg immediately, then 117 mg 1 week later, then 117 mg 1 month later. An exception to this is if a patient’s maintenance dose is 234 mg monthly. In this case, the patient should receive 156 mg of PP1M immediately, then 156 mg 1 week later, and then 234 mg 1 month later.31 If it has been >6 months since the last dose, the patient should start the initiation schedule as if he or she were receiving a new medication.31
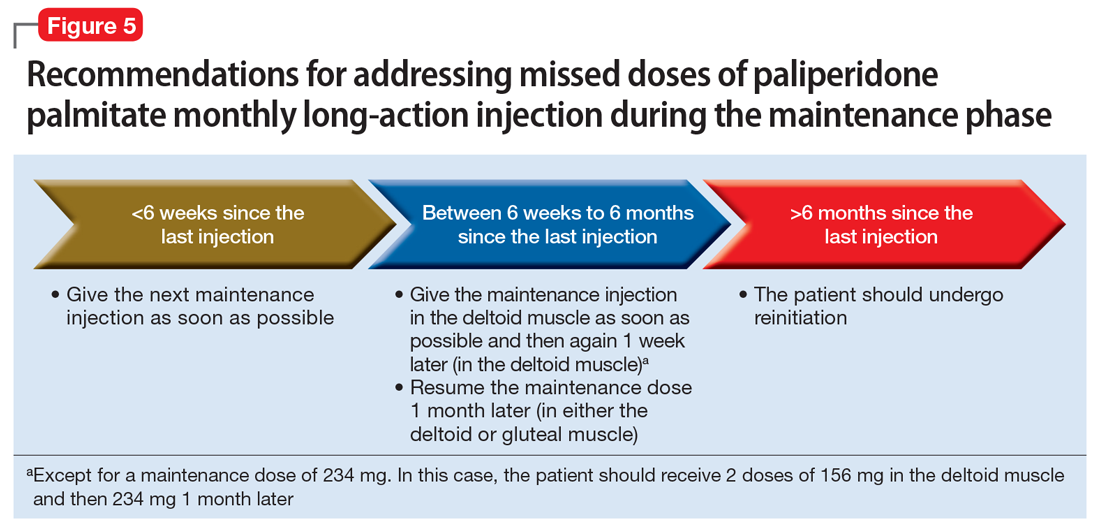
Paliperidone palmitate 3-month long-acting injection (PP3M) should be administered every 3 months. This injection can be given 2 weeks before or after the date of the scheduled dose.32
If the patient missed an injection and it has been <4 months since the last dose, the next scheduled dose should be given as soon as possible.32 If it has been 4 to 9 months since the last dose, the patient must return to PP1M for 2 booster injections 1 week apart. The dose of these PP1M booster injections depends on the dose of PP3M that the patient had been stabilized on:
- 78 mg if stabilized on 273 mg
- 117 mg if stabilized on 410 mg
- 156 mg if stabilized on 546 mg or 819 mg.32
After the second booster dose, PP3M can be restarted 1 month later.32 If it has been >9 months since the last PP3M dose, the patient should be restarted on PP1M. PP3M can be reconsidered once the patient has been stabilized on PP1M for ≥4 months (Figure 6).32
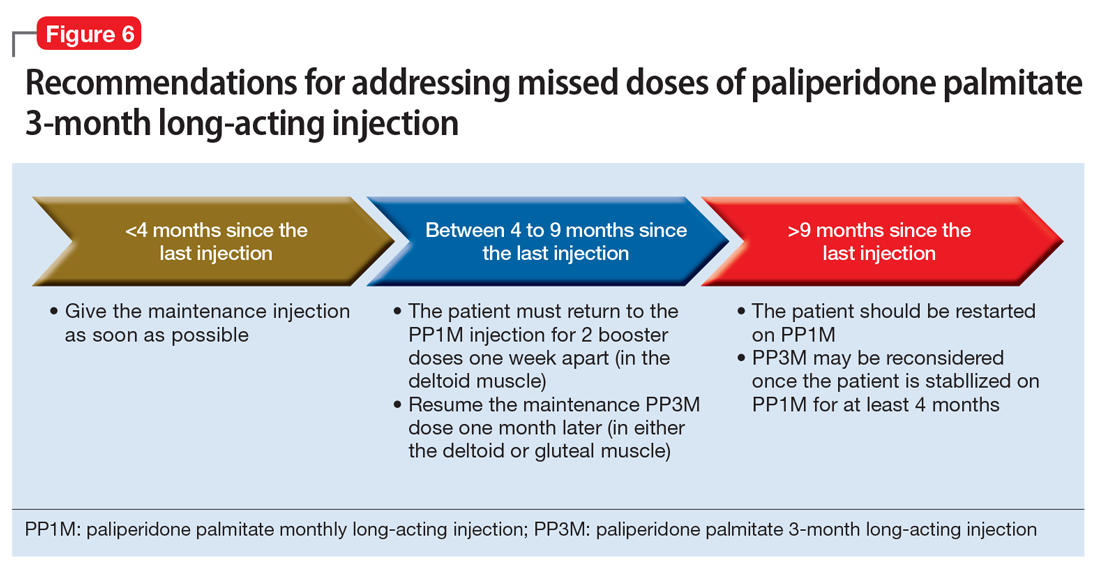
Continue to: Aripiprazole long-acting injection
Aripiprazole long-acting injection is administered every 4 weeks. If a patient misses an injection, first determine how many consecutive doses he or she has received.33 If the patient has missed the second or third injection, and it has been <5 weeks since the last dose, give the next injection as soon as possible. If it has been >5 weeks, give the next injection as soon as possible, plus oral aripiprazole supplementation for 2 weeks (Figure 7).

If the patient has received ≥4 consecutive doses and misses a dose and it has been <6 weeks since the last dose, administer an injection as soon as possible. If it has been >6 weeks since the last dose, give the next injection as soon as possible, plus with oral aripiprazole supplementation for 2 weeks.
Aripiprazole lauroxil long-acting injection. Depending on the dose, aripiprazole lauroxil can be administered monthly, every 6 weeks, or every 2 months. Aripiprazole lauroxil can be administered 14 days before or after the scheduled dose.34
The guidance for addressing missed or delayed doses of aripiprazole lauroxil differs depending on the dose the patient is stabilized on, and how long it has been since the last injection. Figure 8 summarizes how missed injections should be managed. When oral aripiprazole supplementation is needed, the following doses should be used:
- 10 mg/d if stabilized on 441 mg every month
- 15 mg/d if stabilized on 662 mg every month, 882 mg every 6 weeks, or 1,064 mg every 2 months
- 20 mg/d if stabilized on 882 mg every month.34

Olanzapine pamoate long-acting injection is a unique LAIA because it requires prescribers and patients to participate in a risk evaluation and mitigation strategies (REMS) program due the risk of post-injection delirium/sedation syndrome. It is administered every 2 to 4 weeks, with loading doses given for the first 2 months of treatment (Table 235). After 2 months, the patient can proceed to the maintenance dosing regimen.
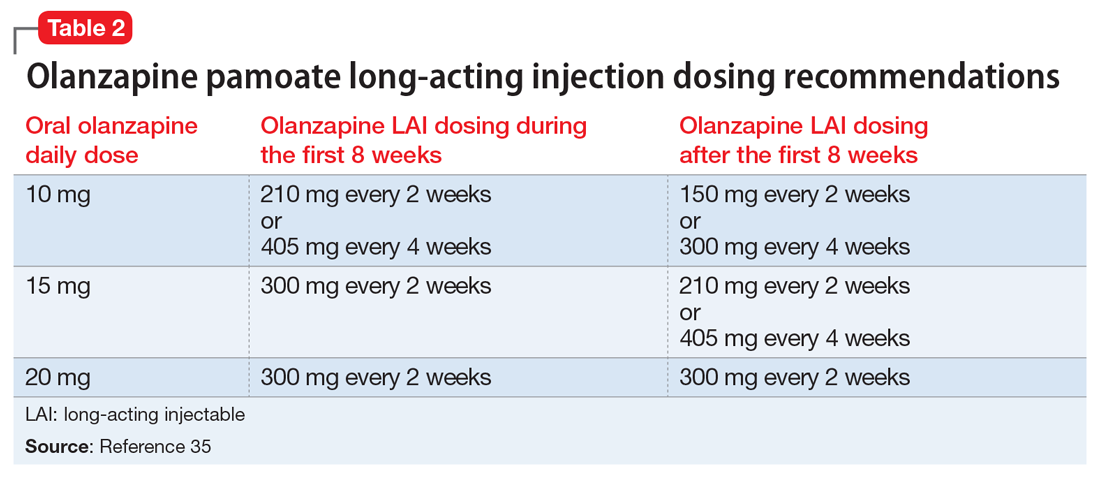
Continue to: Currently, there is no concrete guidance...
Currently, there is no concrete guidance on how to address missed doses of olanzapine long-acting injection; however, the pharmacokinetics of this formulation allow flexibility in dosing intervals. Therapeutic levels are present after the first injection, and the medication reaches steady-state levels in 3 months.35-37 As a result of its specific formulation, olanzapine pamoate long-acting injection provides sustained olanzapine pamoate plasma concentrations between injections, and has a half-life of 30 days.35 Consequently, therapeutic levels of the medication are still present 2 to 4 weeks after an injection.37 Additionally, clinically relevant plasma concentrations may be present 2 to 3 months after the last injection.36
In light of this information, if a patient has not reached steady state and has missed an injection, he or she should receive the recommended loading dose schedule. If the patient has reached steady state and it has been ≤2 months since the last dose, he or she should receive the next dose as soon as possible. If steady state has been reached and it has been >2 months since the last injection, the patient should receive the recommended loading dosing for 2 months (Figure 9). Because of the risk of post-injection delirium/sedation syndrome, and because therapeutic levels are achieved after the first injection, oral olanzapine supplementation is not recommended.

Use a stepwise approach
In general, clinicians can use a stepwise approach to managing missed doses of LAIAs (Figure 10). First, establish the number of LAIA doses the patient had received prior to the last dose, and whether these injections were administered on schedule. This will help you determine if the patient is in the initiation or maintenance phase and/or has reached steady state. The second step is to establish the date of the last injection. Use objective tools, such as pharmacy records or the medical chart, to determine the date of the last injection, rather than relying on patient reporting. For the third step, calculate the time that has passed since the last LAIA dose. Once you have completed these steps, use the specific medication recommendations described in this article to address the missed dose.

Continue to: Address barriers to adherence
Address barriers to adherence
When addressing missed LAIA doses, be sure to identify any barriers that may have led to a missed injection. These might include:
- bothersome adverse effects
- transportation difficulties
- issues with insurance/medication coverage
- comorbidities (ie, alcohol/substance use disorders)
- cognitive and functional impairment caused by the patient’s illness
- difficulty with keeping track of appointments.
Clinicians can work closely with patients and/or caregivers to address any barriers to ensure that patients receive their injections in a timely fashion.
The goal: Reducing relapse
LAIAs improve medication adherence. Although nonadherence is less frequent with LAIAs than with oral antipsychotics, when a LAIA dose is missed, it is important to properly follow a stepwise approach based on the unique properties of the specific LAIA prescribed. Proper management of LAIA missed doses can prevent relapse and reoccurrence of schizophrenia symptoms, thus possibly avoiding future hospitalizations.
Acknowledgments
The authors thank Brian Tschosik, JD, Mary Collen O’Rourke, MD, and Amanda Holloway, MD, for their assistance with this article.
Bottom Line
Although long-acting injectable antipsychotics (LAIAs) greatly assist with adherence, these agents are effective only when missed doses are avoided. When addressing missed LAIA doses, use a stepwise approach that takes into consideration the unique properties of the specific LAIA prescribed.
Related Resources
- Haddad P, Lambert T, Lauriello J, eds. Antipsychotic long-acting injections. 2nd ed. Oxford, UK: Oxford University Press; 2016.
- Diefenderfer LA. When should you consider combining 2 long-acting injectable antipsychotics? Current Psychiatry. 2017;16(10):42-46.
Drug Brand Names
Aripiprazole long-acting injection • Abilify Maintena
Aripiprazole lauroxil long-acting injection • Aristada
Fluphenazine decanoate • Prolixin decanoate
Haloperidol decanoate • Haldol decanoate
Olanzapine pamoate long-acting injection • Zyprexa Relprevv
Paliperidone palmitate monthly long-acting injection • Invega Sustenna
Paliperidone palmitate 3-month long-acting injection • Invega Trinza
Risperidone long-acting injection • Risperdal Consta
1. Olfson M, Mechanic D, Hansell S, et al. Predicting medication noncompliance after hospital discharge among patients with schizophrenia. Psychiatr Serv. 2000;51(2):216-222.
2. Lehman AF, Lieberman JA, Dixon LB, et al; American Psychiatric Association; Steering Committee on Practice Guidelines. Practice guideline for the treatment of patients with schizophrenia, second edition. Am J Psychiatry. 2004;161(suppl 2):1-56.
3. Kane JM, Garcia-Ribera C. Clinical guideline recommendations for antipsychotic long-acting injections. Br J Psychiatry Suppl. 2009;195(52):S63-S67.
4. Velligan DI, Weiden PJ, Sajatovic M, et al. Strategies for addressing adherence problems in patients with serious and persistent mental illness: recommendations from the expert consensus guidelines. J Psychiatr Pract. 2010;16(5):306-324.
5. Kishimoto T, Robenzadeh A, Leucht C, et al. Long-acting injectable vs oral antipsychotics for relapse prevention in schizophrenia: a meta-analysis of randomized trials. Schizophr Bull. 2014;40(1):192-213.
6. Andreasen NC. Symptoms, signs, and diagnosis of schizophrenia. Lancet. 1995;346(8973):477-481.
7. de Sena EP, Santos-Jesus R, Miranda-Scippa Â, et al. Relapse in patients with schizophrenia: a comparison between risperidone and haloperidol. Rev Bras Psiquiatr. 2003;25(4):220-223.
8. Chue P. Long-acting risperidone injection: efficacy, safety, and cost-effectiveness of the first long-acting atypical antipsychotic. Neuropsychiatr Dis Treat. 2007;3(1):13-39.
9. Lafeuille MH, Frois C, Cloutier M, et al. Factors associated with adherence to the HEDIS Quality Measure in medicaid patients with schizophrenia. Am Health Drug Benefits. 2016;9(7):399-410.
10. Kishimoto T, Nitta M, Borenstein M, et al. Long-acting injectable versus oral antipsychotics in schizophrenia: a systematic review and meta-analysis of mirror-image studies. J Clin Psychiatry. 2013;74(10):957-965.
11. Marcus SC, Zummo J, Pettit AR, et al. Antipsychotic adherence and rehospitalization in schizophrenia patients receiving oral versus long-acting injectable antipsychotics following hospital discharge. J Manag Care Spec Pharm. 2015;21(9):754-768.
12. Haldol Decanoate injection [package insert]. Titusville, NJ: Janssen Pharmaceuticals, Inc.; February 2017.
13. Magliozzi JR, Hollister LE, Arnold KV, et al. Relationship of serum haloperidol levels to clinical response in schizophrenic patients. Am J Psychiatry. 1981;138(3):365-367.
14. Mavroidis ML, Kanter DR, Hirschowitz J, et al. Clinical response and plasma haloperidol levels in schizophrenia. Psychopharmacology (Berl). 1983;81(4):354-356.
15. Reyntigens AJ, Heykants JJ, Woestenborghs RJ, et al. Pharmacokinetics of haloperidol decanoate. A 2-year follow-up. Int Pharmacopsychiatry. 1982;17(4):238-246.
16. Jann MW, Ereshefsky L, Saklad SR. Clinical pharmacokinetics of the depot antipsychotics. Clin Pharmacokinet. 1985;10(4):315-333.
17. Chang WH, Lin SK, Juang DJ, et al. Prolonged haloperidol and reduced haloperidol plasma concentrations after decanoate withdrawal. Schizophr Res. 1993;9(1):35-40.
18. Ereshefsky L, Saklad SR, Jann MW. Future of depot neuroleptic therapy: pharmacokinetic and pharmacodynamic approaches. J Clin Psychiatry.1984;45(5 pt 2):50-58.
19. Marder SR, Hawes EM, Van Putten T, et al. Fluphenazine plasma levels in patients receiving low and conventional doses of fluphenazine decanoate. Psychopharmacology (Berl). 1986;88(4):480-483.
20. Marder SR, Hubbard JW, Van Putten T, et al. Pharmacokinetics of long-acting injectable neuroleptic drugs: clinical implications. Psychopharmacology (Berl). 1989;98(4):433-439.
21. Mavroidis ML, Kanter DR, Hirschowitz J, et al. Fluphenazine plasma levels and clinical response. J Clin Psychiatry. 1984;45(9):370-373.
22. Balant-Gorgia AE, Balant LP, Andreoli A. Pharmacokinetic optimisation of the treatment of psychosis. Clin Pharmacokinet. 1993;25(3):217-236.
23. Van Putten T, Marder SR, Wirshing WC, et al. Neuroleptic plasma levels. Schizophr Bull. 1991;17(2):197-216.
24. Dahl SG. Plasma level monitoring of antipsychotic drugs. Clinical utility. Clin Pharmacokinet. 1986;11(1):36-61.
25. Miller RS, Peterson GM, McLean S, et al. Monitoring plasma levels of fluphenazine during chronic therapy with fluphenazine decanoate. J Clin Pharm Ther. 1995;20(2):55-62.
26. Gitlin MJ, Midha KK, Fogelson D, et al. Persistence of fluphenazine in plasma after decanoate withdrawal. J Clin Psychopharmacol. 1988;8(1):53-56.
27. Wistedt B, Wiles D, Kolakowska T. Slow decline of plasma drug and prolactin levels after discontinuation of chronic treatment with depot neuroleptics. Lancet. 1981;1(8230):1163.
28. Wistedt B, Jørgensen A, Wiles D. A depot neuroleptic withdrawal study. Plasma concentration of fluphenazine and flupenthixol and relapse frequency. Psychopharmacology (Berl). 1982;78(4):301-304.
29. Risperdal Consta [package insert]. Titusville, NJ: Janssen Pharmaceuticals, Inc.; February 2017.
30. Marder SR, Conley R, Ereshefsky L, et al. Clinical guidelines: dosing and switching strategies for long-acting risperidone. J Clin Psychiatry. 2003;64(suppl 16):41-46.
31. Invega Sustenna [package insert]. Titusville, NJ: Janssen Pharmaceuticals, Inc.; February 2017.
32. Invega Trinza [package insert]. Titusville, NJ: Janssen Pharmaceuticals, Inc.; February 2017.
33. Abilify Maintena [package insert]. Rockville, MD: Otsuka America Pharmaceutical, Inc.; December 2016.
34. Artistada [package insert]. Waltham, MA: Alkermes, Inc.; June 2017.
35. Zyprexa Relprevv [package insert]. Indianapolis; IN: Eli Lilly and Co.; February 2017.
36. Heres S, Kraemer S, Bergstrom RF, et al. Pharmacokinetics of olanzapine long-acting injection: the clinical perspective. Int Clin Psychopharmacol. 2014;29(6):299-312.
37. Detke HC, Zhao F, Garhyan P, et al. Dose correspondence between olanzapine long-acting injection and oral olanzapine: recommendations for switching. Int Clin Psychopharmacol. 2011;26(1):35-42.
Antipsychotic agents are the mainstay of treatment for patients with schizophrenia,1-3 and when taken regularly, they can greatly improve patient outcomes. Unfortunately, many studies have documented poor adherence to antipsychotic regimens in patients with schizophrenia, which often leads to an exacerbation of symptoms and preventable hospitalizations.4-8 In order to improve adherence, many clinicians prescribe long-acting injectable antipsychotics (LAIAs).
LAIAs help improve adherence, but these benefits are seen only in patients who receive their injections within a specific time frame.9-11 LAIAs administered outside of this time frame (missed doses) can lead to reoccurrence or exacerbation of symptoms. This article explains how to adequately manage missed LAIA doses.
First-generation long-acting injectable antipsychotics
Two first-generation antipsychotics are available as a long-acting injectable formulation: haloperidol decanoate and fluphenazine decanoate. Due to the increased risk of extrapyramidal symptoms, use of these agents have decreased, and they are often less preferred than second-generation LAIAs. Furthermore, unlike many of the newer second-generation LAIAs, first-generation LAIAs lack literature on how to manage missed doses. Therefore, clinicians should analyze the pharmacokinetic properties of these agents (Table 112-28), as well as the patient’s medical history and clinical presentation, in order to determine how best to address missed doses.

Haloperidol decanoate plasma concentrations peak approximately 6 days after the injection.12 The medication has a half-life of 3 weeks. One study found that haloperidol plasma concentrations were detectable 13 weeks after the discontinuation of haloperidol decanoate.17 This same study also found that the change in plasma levels from 3 to 6 weeks after the last dose was minimal.17 Based on these findings, Figure 1 summarizes our recommendations for addressing missed haloperidol decanoate doses.

Fluphenazine decanoate levels peak 24 hours after the injection.18 An estimated therapeutic range for fluphenazine is 0.2 to 2 ng/mL.21-25 One study that evaluated fluphenazine decanoate levels following discontinuation after reaching steady state found there was no significant difference in plasma levels 6 weeks after the last dose of fluphenazine, but a significant decrease in levels 8 to 12 weeks after the last dose.26 Other studies found that fluphenazine levels were detectable 21 to 24 weeks following fluphenazine decanoate discontinuation.27,28 Based on these findings, Figure 2 summarizes our recommendations for addressing missed fluphenazine decanoate doses.

Continue to: Second-generation LAIAs
Second-generation LAIAs
Six second-generation LAIAs are available in the United States. Compared with the first-generation LAIAs, second-generation LAIAs have more extensive guidance on how to address missed doses.
Risperidone long-acting injection. When addressing missed doses of risperidone long-acting injection, first determine whether the medication has reached steady state. Steady state occurs approximately after the fourth consecutive injection (approximately 2 months).29
If a patient missed a dose but has not reached steady state, he or she should receive the next dose as well as oral antipsychotic supplementation for 3 weeks.30 If the patient has reached steady state and if it has been ≤6 weeks since the last injection, give the next injection as soon as possible. However, if steady state has been reached and it has been >6 weeks since the last injection, give the next injection, along with 3 weeks of oral antipsychotic supplementation (Figure 3).

Paliperidone palmitate monthly long-acting injection. Once the initiation dosing phase of paliperidone palmitate monthly long-acting injection (PP1M) is completed, the maintenance dose is administered every 4 weeks. When addressing missed doses of PP1M, first determine whether the patient is in the initiation or maintenance dosing phase.31
Initiation phase. Patients are in the initiation dosing phase during the first 2 injections of PP1M. During the initiation phase, the patient first receives 234 mg and then 156 mg 1 week later, both in the deltoid muscle. One month later, the patient receives a maintenance dose of PP1M (in the deltoid or gluteal muscle). The second initiation injection may be given 4 days before or after the scheduled administration date. The initiation doses should be adjusted in patients with mild renal function (creatinine clearance 50 to 80 mL/min).31 Figure 4 summarizes the guidance for addressing a missed or delayed second injection during the initiation phase.

Continue to: Maintenance phase
Maintenance phase. During the maintenance phase, PP1M can be administered 7 days before or after the monthly due date. If the patient has missed a maintenance injection and it has been <6 weeks since the last dose, the maintenance injection can be given as soon as possible (Figure 5).31 If it has been 6 weeks to 6 months since the last injection, the patient should receive their prescribed maintenance dose as soon as possible and the same dose 1 week later, with both injections in the deltoid muscle. Following the second dose, the patient can resume their regular monthly maintenance schedule, in either the deltoid or gluteal muscle. For example, if the patient was maintained on 117 mg of PP1M and it had been 8 weeks since the last injection, the patient should receive 117 mg immediately, then 117 mg 1 week later, then 117 mg 1 month later. An exception to this is if a patient’s maintenance dose is 234 mg monthly. In this case, the patient should receive 156 mg of PP1M immediately, then 156 mg 1 week later, and then 234 mg 1 month later.31 If it has been >6 months since the last dose, the patient should start the initiation schedule as if he or she were receiving a new medication.31

Paliperidone palmitate 3-month long-acting injection (PP3M) should be administered every 3 months. This injection can be given 2 weeks before or after the date of the scheduled dose.32
If the patient missed an injection and it has been <4 months since the last dose, the next scheduled dose should be given as soon as possible.32 If it has been 4 to 9 months since the last dose, the patient must return to PP1M for 2 booster injections 1 week apart. The dose of these PP1M booster injections depends on the dose of PP3M that the patient had been stabilized on:
- 78 mg if stabilized on 273 mg
- 117 mg if stabilized on 410 mg
- 156 mg if stabilized on 546 mg or 819 mg.32
After the second booster dose, PP3M can be restarted 1 month later.32 If it has been >9 months since the last PP3M dose, the patient should be restarted on PP1M. PP3M can be reconsidered once the patient has been stabilized on PP1M for ≥4 months (Figure 6).32

Continue to: Aripiprazole long-acting injection
Aripiprazole long-acting injection is administered every 4 weeks. If a patient misses an injection, first determine how many consecutive doses he or she has received.33 If the patient has missed the second or third injection, and it has been <5 weeks since the last dose, give the next injection as soon as possible. If it has been >5 weeks, give the next injection as soon as possible, plus oral aripiprazole supplementation for 2 weeks (Figure 7).

If the patient has received ≥4 consecutive doses and misses a dose and it has been <6 weeks since the last dose, administer an injection as soon as possible. If it has been >6 weeks since the last dose, give the next injection as soon as possible, plus with oral aripiprazole supplementation for 2 weeks.
Aripiprazole lauroxil long-acting injection. Depending on the dose, aripiprazole lauroxil can be administered monthly, every 6 weeks, or every 2 months. Aripiprazole lauroxil can be administered 14 days before or after the scheduled dose.34
The guidance for addressing missed or delayed doses of aripiprazole lauroxil differs depending on the dose the patient is stabilized on, and how long it has been since the last injection. Figure 8 summarizes how missed injections should be managed. When oral aripiprazole supplementation is needed, the following doses should be used:
- 10 mg/d if stabilized on 441 mg every month
- 15 mg/d if stabilized on 662 mg every month, 882 mg every 6 weeks, or 1,064 mg every 2 months
- 20 mg/d if stabilized on 882 mg every month.34

Olanzapine pamoate long-acting injection is a unique LAIA because it requires prescribers and patients to participate in a risk evaluation and mitigation strategies (REMS) program due the risk of post-injection delirium/sedation syndrome. It is administered every 2 to 4 weeks, with loading doses given for the first 2 months of treatment (Table 235). After 2 months, the patient can proceed to the maintenance dosing regimen.

Continue to: Currently, there is no concrete guidance...
Currently, there is no concrete guidance on how to address missed doses of olanzapine long-acting injection; however, the pharmacokinetics of this formulation allow flexibility in dosing intervals. Therapeutic levels are present after the first injection, and the medication reaches steady-state levels in 3 months.35-37 As a result of its specific formulation, olanzapine pamoate long-acting injection provides sustained olanzapine pamoate plasma concentrations between injections, and has a half-life of 30 days.35 Consequently, therapeutic levels of the medication are still present 2 to 4 weeks after an injection.37 Additionally, clinically relevant plasma concentrations may be present 2 to 3 months after the last injection.36
In light of this information, if a patient has not reached steady state and has missed an injection, he or she should receive the recommended loading dose schedule. If the patient has reached steady state and it has been ≤2 months since the last dose, he or she should receive the next dose as soon as possible. If steady state has been reached and it has been >2 months since the last injection, the patient should receive the recommended loading dosing for 2 months (Figure 9). Because of the risk of post-injection delirium/sedation syndrome, and because therapeutic levels are achieved after the first injection, oral olanzapine supplementation is not recommended.

Use a stepwise approach
In general, clinicians can use a stepwise approach to managing missed doses of LAIAs (Figure 10). First, establish the number of LAIA doses the patient had received prior to the last dose, and whether these injections were administered on schedule. This will help you determine if the patient is in the initiation or maintenance phase and/or has reached steady state. The second step is to establish the date of the last injection. Use objective tools, such as pharmacy records or the medical chart, to determine the date of the last injection, rather than relying on patient reporting. For the third step, calculate the time that has passed since the last LAIA dose. Once you have completed these steps, use the specific medication recommendations described in this article to address the missed dose.

Continue to: Address barriers to adherence
Address barriers to adherence
When addressing missed LAIA doses, be sure to identify any barriers that may have led to a missed injection. These might include:
- bothersome adverse effects
- transportation difficulties
- issues with insurance/medication coverage
- comorbidities (ie, alcohol/substance use disorders)
- cognitive and functional impairment caused by the patient’s illness
- difficulty with keeping track of appointments.
Clinicians can work closely with patients and/or caregivers to address any barriers to ensure that patients receive their injections in a timely fashion.
The goal: Reducing relapse
LAIAs improve medication adherence. Although nonadherence is less frequent with LAIAs than with oral antipsychotics, when a LAIA dose is missed, it is important to properly follow a stepwise approach based on the unique properties of the specific LAIA prescribed. Proper management of LAIA missed doses can prevent relapse and reoccurrence of schizophrenia symptoms, thus possibly avoiding future hospitalizations.
Acknowledgments
The authors thank Brian Tschosik, JD, Mary Collen O’Rourke, MD, and Amanda Holloway, MD, for their assistance with this article.
Bottom Line
Although long-acting injectable antipsychotics (LAIAs) greatly assist with adherence, these agents are effective only when missed doses are avoided. When addressing missed LAIA doses, use a stepwise approach that takes into consideration the unique properties of the specific LAIA prescribed.
Related Resources
- Haddad P, Lambert T, Lauriello J, eds. Antipsychotic long-acting injections. 2nd ed. Oxford, UK: Oxford University Press; 2016.
- Diefenderfer LA. When should you consider combining 2 long-acting injectable antipsychotics? Current Psychiatry. 2017;16(10):42-46.
Drug Brand Names
Aripiprazole long-acting injection • Abilify Maintena
Aripiprazole lauroxil long-acting injection • Aristada
Fluphenazine decanoate • Prolixin decanoate
Haloperidol decanoate • Haldol decanoate
Olanzapine pamoate long-acting injection • Zyprexa Relprevv
Paliperidone palmitate monthly long-acting injection • Invega Sustenna
Paliperidone palmitate 3-month long-acting injection • Invega Trinza
Risperidone long-acting injection • Risperdal Consta
Antipsychotic agents are the mainstay of treatment for patients with schizophrenia,1-3 and when taken regularly, they can greatly improve patient outcomes. Unfortunately, many studies have documented poor adherence to antipsychotic regimens in patients with schizophrenia, which often leads to an exacerbation of symptoms and preventable hospitalizations.4-8 In order to improve adherence, many clinicians prescribe long-acting injectable antipsychotics (LAIAs).
LAIAs help improve adherence, but these benefits are seen only in patients who receive their injections within a specific time frame.9-11 LAIAs administered outside of this time frame (missed doses) can lead to reoccurrence or exacerbation of symptoms. This article explains how to adequately manage missed LAIA doses.
First-generation long-acting injectable antipsychotics
Two first-generation antipsychotics are available as a long-acting injectable formulation: haloperidol decanoate and fluphenazine decanoate. Due to the increased risk of extrapyramidal symptoms, use of these agents have decreased, and they are often less preferred than second-generation LAIAs. Furthermore, unlike many of the newer second-generation LAIAs, first-generation LAIAs lack literature on how to manage missed doses. Therefore, clinicians should analyze the pharmacokinetic properties of these agents (Table 112-28), as well as the patient’s medical history and clinical presentation, in order to determine how best to address missed doses.

Haloperidol decanoate plasma concentrations peak approximately 6 days after the injection.12 The medication has a half-life of 3 weeks. One study found that haloperidol plasma concentrations were detectable 13 weeks after the discontinuation of haloperidol decanoate.17 This same study also found that the change in plasma levels from 3 to 6 weeks after the last dose was minimal.17 Based on these findings, Figure 1 summarizes our recommendations for addressing missed haloperidol decanoate doses.

Fluphenazine decanoate levels peak 24 hours after the injection.18 An estimated therapeutic range for fluphenazine is 0.2 to 2 ng/mL.21-25 One study that evaluated fluphenazine decanoate levels following discontinuation after reaching steady state found there was no significant difference in plasma levels 6 weeks after the last dose of fluphenazine, but a significant decrease in levels 8 to 12 weeks after the last dose.26 Other studies found that fluphenazine levels were detectable 21 to 24 weeks following fluphenazine decanoate discontinuation.27,28 Based on these findings, Figure 2 summarizes our recommendations for addressing missed fluphenazine decanoate doses.

Continue to: Second-generation LAIAs
Second-generation LAIAs
Six second-generation LAIAs are available in the United States. Compared with the first-generation LAIAs, second-generation LAIAs have more extensive guidance on how to address missed doses.
Risperidone long-acting injection. When addressing missed doses of risperidone long-acting injection, first determine whether the medication has reached steady state. Steady state occurs approximately after the fourth consecutive injection (approximately 2 months).29
If a patient missed a dose but has not reached steady state, he or she should receive the next dose as well as oral antipsychotic supplementation for 3 weeks.30 If the patient has reached steady state and if it has been ≤6 weeks since the last injection, give the next injection as soon as possible. However, if steady state has been reached and it has been >6 weeks since the last injection, give the next injection, along with 3 weeks of oral antipsychotic supplementation (Figure 3).

Paliperidone palmitate monthly long-acting injection. Once the initiation dosing phase of paliperidone palmitate monthly long-acting injection (PP1M) is completed, the maintenance dose is administered every 4 weeks. When addressing missed doses of PP1M, first determine whether the patient is in the initiation or maintenance dosing phase.31
Initiation phase. Patients are in the initiation dosing phase during the first 2 injections of PP1M. During the initiation phase, the patient first receives 234 mg and then 156 mg 1 week later, both in the deltoid muscle. One month later, the patient receives a maintenance dose of PP1M (in the deltoid or gluteal muscle). The second initiation injection may be given 4 days before or after the scheduled administration date. The initiation doses should be adjusted in patients with mild renal function (creatinine clearance 50 to 80 mL/min).31 Figure 4 summarizes the guidance for addressing a missed or delayed second injection during the initiation phase.

Continue to: Maintenance phase
Maintenance phase. During the maintenance phase, PP1M can be administered 7 days before or after the monthly due date. If the patient has missed a maintenance injection and it has been <6 weeks since the last dose, the maintenance injection can be given as soon as possible (Figure 5).31 If it has been 6 weeks to 6 months since the last injection, the patient should receive their prescribed maintenance dose as soon as possible and the same dose 1 week later, with both injections in the deltoid muscle. Following the second dose, the patient can resume their regular monthly maintenance schedule, in either the deltoid or gluteal muscle. For example, if the patient was maintained on 117 mg of PP1M and it had been 8 weeks since the last injection, the patient should receive 117 mg immediately, then 117 mg 1 week later, then 117 mg 1 month later. An exception to this is if a patient’s maintenance dose is 234 mg monthly. In this case, the patient should receive 156 mg of PP1M immediately, then 156 mg 1 week later, and then 234 mg 1 month later.31 If it has been >6 months since the last dose, the patient should start the initiation schedule as if he or she were receiving a new medication.31

Paliperidone palmitate 3-month long-acting injection (PP3M) should be administered every 3 months. This injection can be given 2 weeks before or after the date of the scheduled dose.32
If the patient missed an injection and it has been <4 months since the last dose, the next scheduled dose should be given as soon as possible.32 If it has been 4 to 9 months since the last dose, the patient must return to PP1M for 2 booster injections 1 week apart. The dose of these PP1M booster injections depends on the dose of PP3M that the patient had been stabilized on:
- 78 mg if stabilized on 273 mg
- 117 mg if stabilized on 410 mg
- 156 mg if stabilized on 546 mg or 819 mg.32
After the second booster dose, PP3M can be restarted 1 month later.32 If it has been >9 months since the last PP3M dose, the patient should be restarted on PP1M. PP3M can be reconsidered once the patient has been stabilized on PP1M for ≥4 months (Figure 6).32

Continue to: Aripiprazole long-acting injection
Aripiprazole long-acting injection is administered every 4 weeks. If a patient misses an injection, first determine how many consecutive doses he or she has received.33 If the patient has missed the second or third injection, and it has been <5 weeks since the last dose, give the next injection as soon as possible. If it has been >5 weeks, give the next injection as soon as possible, plus oral aripiprazole supplementation for 2 weeks (Figure 7).

If the patient has received ≥4 consecutive doses and misses a dose and it has been <6 weeks since the last dose, administer an injection as soon as possible. If it has been >6 weeks since the last dose, give the next injection as soon as possible, plus with oral aripiprazole supplementation for 2 weeks.
Aripiprazole lauroxil long-acting injection. Depending on the dose, aripiprazole lauroxil can be administered monthly, every 6 weeks, or every 2 months. Aripiprazole lauroxil can be administered 14 days before or after the scheduled dose.34
The guidance for addressing missed or delayed doses of aripiprazole lauroxil differs depending on the dose the patient is stabilized on, and how long it has been since the last injection. Figure 8 summarizes how missed injections should be managed. When oral aripiprazole supplementation is needed, the following doses should be used:
- 10 mg/d if stabilized on 441 mg every month
- 15 mg/d if stabilized on 662 mg every month, 882 mg every 6 weeks, or 1,064 mg every 2 months
- 20 mg/d if stabilized on 882 mg every month.34

Olanzapine pamoate long-acting injection is a unique LAIA because it requires prescribers and patients to participate in a risk evaluation and mitigation strategies (REMS) program due the risk of post-injection delirium/sedation syndrome. It is administered every 2 to 4 weeks, with loading doses given for the first 2 months of treatment (Table 235). After 2 months, the patient can proceed to the maintenance dosing regimen.

Continue to: Currently, there is no concrete guidance...
Currently, there is no concrete guidance on how to address missed doses of olanzapine long-acting injection; however, the pharmacokinetics of this formulation allow flexibility in dosing intervals. Therapeutic levels are present after the first injection, and the medication reaches steady-state levels in 3 months.35-37 As a result of its specific formulation, olanzapine pamoate long-acting injection provides sustained olanzapine pamoate plasma concentrations between injections, and has a half-life of 30 days.35 Consequently, therapeutic levels of the medication are still present 2 to 4 weeks after an injection.37 Additionally, clinically relevant plasma concentrations may be present 2 to 3 months after the last injection.36
In light of this information, if a patient has not reached steady state and has missed an injection, he or she should receive the recommended loading dose schedule. If the patient has reached steady state and it has been ≤2 months since the last dose, he or she should receive the next dose as soon as possible. If steady state has been reached and it has been >2 months since the last injection, the patient should receive the recommended loading dosing for 2 months (Figure 9). Because of the risk of post-injection delirium/sedation syndrome, and because therapeutic levels are achieved after the first injection, oral olanzapine supplementation is not recommended.

Use a stepwise approach
In general, clinicians can use a stepwise approach to managing missed doses of LAIAs (Figure 10). First, establish the number of LAIA doses the patient had received prior to the last dose, and whether these injections were administered on schedule. This will help you determine if the patient is in the initiation or maintenance phase and/or has reached steady state. The second step is to establish the date of the last injection. Use objective tools, such as pharmacy records or the medical chart, to determine the date of the last injection, rather than relying on patient reporting. For the third step, calculate the time that has passed since the last LAIA dose. Once you have completed these steps, use the specific medication recommendations described in this article to address the missed dose.

Continue to: Address barriers to adherence
Address barriers to adherence
When addressing missed LAIA doses, be sure to identify any barriers that may have led to a missed injection. These might include:
- bothersome adverse effects
- transportation difficulties
- issues with insurance/medication coverage
- comorbidities (ie, alcohol/substance use disorders)
- cognitive and functional impairment caused by the patient’s illness
- difficulty with keeping track of appointments.
Clinicians can work closely with patients and/or caregivers to address any barriers to ensure that patients receive their injections in a timely fashion.
The goal: Reducing relapse
LAIAs improve medication adherence. Although nonadherence is less frequent with LAIAs than with oral antipsychotics, when a LAIA dose is missed, it is important to properly follow a stepwise approach based on the unique properties of the specific LAIA prescribed. Proper management of LAIA missed doses can prevent relapse and reoccurrence of schizophrenia symptoms, thus possibly avoiding future hospitalizations.
Acknowledgments
The authors thank Brian Tschosik, JD, Mary Collen O’Rourke, MD, and Amanda Holloway, MD, for their assistance with this article.
Bottom Line
Although long-acting injectable antipsychotics (LAIAs) greatly assist with adherence, these agents are effective only when missed doses are avoided. When addressing missed LAIA doses, use a stepwise approach that takes into consideration the unique properties of the specific LAIA prescribed.
Related Resources
- Haddad P, Lambert T, Lauriello J, eds. Antipsychotic long-acting injections. 2nd ed. Oxford, UK: Oxford University Press; 2016.
- Diefenderfer LA. When should you consider combining 2 long-acting injectable antipsychotics? Current Psychiatry. 2017;16(10):42-46.
Drug Brand Names
Aripiprazole long-acting injection • Abilify Maintena
Aripiprazole lauroxil long-acting injection • Aristada
Fluphenazine decanoate • Prolixin decanoate
Haloperidol decanoate • Haldol decanoate
Olanzapine pamoate long-acting injection • Zyprexa Relprevv
Paliperidone palmitate monthly long-acting injection • Invega Sustenna
Paliperidone palmitate 3-month long-acting injection • Invega Trinza
Risperidone long-acting injection • Risperdal Consta
1. Olfson M, Mechanic D, Hansell S, et al. Predicting medication noncompliance after hospital discharge among patients with schizophrenia. Psychiatr Serv. 2000;51(2):216-222.
2. Lehman AF, Lieberman JA, Dixon LB, et al; American Psychiatric Association; Steering Committee on Practice Guidelines. Practice guideline for the treatment of patients with schizophrenia, second edition. Am J Psychiatry. 2004;161(suppl 2):1-56.
3. Kane JM, Garcia-Ribera C. Clinical guideline recommendations for antipsychotic long-acting injections. Br J Psychiatry Suppl. 2009;195(52):S63-S67.
4. Velligan DI, Weiden PJ, Sajatovic M, et al. Strategies for addressing adherence problems in patients with serious and persistent mental illness: recommendations from the expert consensus guidelines. J Psychiatr Pract. 2010;16(5):306-324.
5. Kishimoto T, Robenzadeh A, Leucht C, et al. Long-acting injectable vs oral antipsychotics for relapse prevention in schizophrenia: a meta-analysis of randomized trials. Schizophr Bull. 2014;40(1):192-213.
6. Andreasen NC. Symptoms, signs, and diagnosis of schizophrenia. Lancet. 1995;346(8973):477-481.
7. de Sena EP, Santos-Jesus R, Miranda-Scippa Â, et al. Relapse in patients with schizophrenia: a comparison between risperidone and haloperidol. Rev Bras Psiquiatr. 2003;25(4):220-223.
8. Chue P. Long-acting risperidone injection: efficacy, safety, and cost-effectiveness of the first long-acting atypical antipsychotic. Neuropsychiatr Dis Treat. 2007;3(1):13-39.
9. Lafeuille MH, Frois C, Cloutier M, et al. Factors associated with adherence to the HEDIS Quality Measure in medicaid patients with schizophrenia. Am Health Drug Benefits. 2016;9(7):399-410.
10. Kishimoto T, Nitta M, Borenstein M, et al. Long-acting injectable versus oral antipsychotics in schizophrenia: a systematic review and meta-analysis of mirror-image studies. J Clin Psychiatry. 2013;74(10):957-965.
11. Marcus SC, Zummo J, Pettit AR, et al. Antipsychotic adherence and rehospitalization in schizophrenia patients receiving oral versus long-acting injectable antipsychotics following hospital discharge. J Manag Care Spec Pharm. 2015;21(9):754-768.
12. Haldol Decanoate injection [package insert]. Titusville, NJ: Janssen Pharmaceuticals, Inc.; February 2017.
13. Magliozzi JR, Hollister LE, Arnold KV, et al. Relationship of serum haloperidol levels to clinical response in schizophrenic patients. Am J Psychiatry. 1981;138(3):365-367.
14. Mavroidis ML, Kanter DR, Hirschowitz J, et al. Clinical response and plasma haloperidol levels in schizophrenia. Psychopharmacology (Berl). 1983;81(4):354-356.
15. Reyntigens AJ, Heykants JJ, Woestenborghs RJ, et al. Pharmacokinetics of haloperidol decanoate. A 2-year follow-up. Int Pharmacopsychiatry. 1982;17(4):238-246.
16. Jann MW, Ereshefsky L, Saklad SR. Clinical pharmacokinetics of the depot antipsychotics. Clin Pharmacokinet. 1985;10(4):315-333.
17. Chang WH, Lin SK, Juang DJ, et al. Prolonged haloperidol and reduced haloperidol plasma concentrations after decanoate withdrawal. Schizophr Res. 1993;9(1):35-40.
18. Ereshefsky L, Saklad SR, Jann MW. Future of depot neuroleptic therapy: pharmacokinetic and pharmacodynamic approaches. J Clin Psychiatry.1984;45(5 pt 2):50-58.
19. Marder SR, Hawes EM, Van Putten T, et al. Fluphenazine plasma levels in patients receiving low and conventional doses of fluphenazine decanoate. Psychopharmacology (Berl). 1986;88(4):480-483.
20. Marder SR, Hubbard JW, Van Putten T, et al. Pharmacokinetics of long-acting injectable neuroleptic drugs: clinical implications. Psychopharmacology (Berl). 1989;98(4):433-439.
21. Mavroidis ML, Kanter DR, Hirschowitz J, et al. Fluphenazine plasma levels and clinical response. J Clin Psychiatry. 1984;45(9):370-373.
22. Balant-Gorgia AE, Balant LP, Andreoli A. Pharmacokinetic optimisation of the treatment of psychosis. Clin Pharmacokinet. 1993;25(3):217-236.
23. Van Putten T, Marder SR, Wirshing WC, et al. Neuroleptic plasma levels. Schizophr Bull. 1991;17(2):197-216.
24. Dahl SG. Plasma level monitoring of antipsychotic drugs. Clinical utility. Clin Pharmacokinet. 1986;11(1):36-61.
25. Miller RS, Peterson GM, McLean S, et al. Monitoring plasma levels of fluphenazine during chronic therapy with fluphenazine decanoate. J Clin Pharm Ther. 1995;20(2):55-62.
26. Gitlin MJ, Midha KK, Fogelson D, et al. Persistence of fluphenazine in plasma after decanoate withdrawal. J Clin Psychopharmacol. 1988;8(1):53-56.
27. Wistedt B, Wiles D, Kolakowska T. Slow decline of plasma drug and prolactin levels after discontinuation of chronic treatment with depot neuroleptics. Lancet. 1981;1(8230):1163.
28. Wistedt B, Jørgensen A, Wiles D. A depot neuroleptic withdrawal study. Plasma concentration of fluphenazine and flupenthixol and relapse frequency. Psychopharmacology (Berl). 1982;78(4):301-304.
29. Risperdal Consta [package insert]. Titusville, NJ: Janssen Pharmaceuticals, Inc.; February 2017.
30. Marder SR, Conley R, Ereshefsky L, et al. Clinical guidelines: dosing and switching strategies for long-acting risperidone. J Clin Psychiatry. 2003;64(suppl 16):41-46.
31. Invega Sustenna [package insert]. Titusville, NJ: Janssen Pharmaceuticals, Inc.; February 2017.
32. Invega Trinza [package insert]. Titusville, NJ: Janssen Pharmaceuticals, Inc.; February 2017.
33. Abilify Maintena [package insert]. Rockville, MD: Otsuka America Pharmaceutical, Inc.; December 2016.
34. Artistada [package insert]. Waltham, MA: Alkermes, Inc.; June 2017.
35. Zyprexa Relprevv [package insert]. Indianapolis; IN: Eli Lilly and Co.; February 2017.
36. Heres S, Kraemer S, Bergstrom RF, et al. Pharmacokinetics of olanzapine long-acting injection: the clinical perspective. Int Clin Psychopharmacol. 2014;29(6):299-312.
37. Detke HC, Zhao F, Garhyan P, et al. Dose correspondence between olanzapine long-acting injection and oral olanzapine: recommendations for switching. Int Clin Psychopharmacol. 2011;26(1):35-42.
1. Olfson M, Mechanic D, Hansell S, et al. Predicting medication noncompliance after hospital discharge among patients with schizophrenia. Psychiatr Serv. 2000;51(2):216-222.
2. Lehman AF, Lieberman JA, Dixon LB, et al; American Psychiatric Association; Steering Committee on Practice Guidelines. Practice guideline for the treatment of patients with schizophrenia, second edition. Am J Psychiatry. 2004;161(suppl 2):1-56.
3. Kane JM, Garcia-Ribera C. Clinical guideline recommendations for antipsychotic long-acting injections. Br J Psychiatry Suppl. 2009;195(52):S63-S67.
4. Velligan DI, Weiden PJ, Sajatovic M, et al. Strategies for addressing adherence problems in patients with serious and persistent mental illness: recommendations from the expert consensus guidelines. J Psychiatr Pract. 2010;16(5):306-324.
5. Kishimoto T, Robenzadeh A, Leucht C, et al. Long-acting injectable vs oral antipsychotics for relapse prevention in schizophrenia: a meta-analysis of randomized trials. Schizophr Bull. 2014;40(1):192-213.
6. Andreasen NC. Symptoms, signs, and diagnosis of schizophrenia. Lancet. 1995;346(8973):477-481.
7. de Sena EP, Santos-Jesus R, Miranda-Scippa Â, et al. Relapse in patients with schizophrenia: a comparison between risperidone and haloperidol. Rev Bras Psiquiatr. 2003;25(4):220-223.
8. Chue P. Long-acting risperidone injection: efficacy, safety, and cost-effectiveness of the first long-acting atypical antipsychotic. Neuropsychiatr Dis Treat. 2007;3(1):13-39.
9. Lafeuille MH, Frois C, Cloutier M, et al. Factors associated with adherence to the HEDIS Quality Measure in medicaid patients with schizophrenia. Am Health Drug Benefits. 2016;9(7):399-410.
10. Kishimoto T, Nitta M, Borenstein M, et al. Long-acting injectable versus oral antipsychotics in schizophrenia: a systematic review and meta-analysis of mirror-image studies. J Clin Psychiatry. 2013;74(10):957-965.
11. Marcus SC, Zummo J, Pettit AR, et al. Antipsychotic adherence and rehospitalization in schizophrenia patients receiving oral versus long-acting injectable antipsychotics following hospital discharge. J Manag Care Spec Pharm. 2015;21(9):754-768.
12. Haldol Decanoate injection [package insert]. Titusville, NJ: Janssen Pharmaceuticals, Inc.; February 2017.
13. Magliozzi JR, Hollister LE, Arnold KV, et al. Relationship of serum haloperidol levels to clinical response in schizophrenic patients. Am J Psychiatry. 1981;138(3):365-367.
14. Mavroidis ML, Kanter DR, Hirschowitz J, et al. Clinical response and plasma haloperidol levels in schizophrenia. Psychopharmacology (Berl). 1983;81(4):354-356.
15. Reyntigens AJ, Heykants JJ, Woestenborghs RJ, et al. Pharmacokinetics of haloperidol decanoate. A 2-year follow-up. Int Pharmacopsychiatry. 1982;17(4):238-246.
16. Jann MW, Ereshefsky L, Saklad SR. Clinical pharmacokinetics of the depot antipsychotics. Clin Pharmacokinet. 1985;10(4):315-333.
17. Chang WH, Lin SK, Juang DJ, et al. Prolonged haloperidol and reduced haloperidol plasma concentrations after decanoate withdrawal. Schizophr Res. 1993;9(1):35-40.
18. Ereshefsky L, Saklad SR, Jann MW. Future of depot neuroleptic therapy: pharmacokinetic and pharmacodynamic approaches. J Clin Psychiatry.1984;45(5 pt 2):50-58.
19. Marder SR, Hawes EM, Van Putten T, et al. Fluphenazine plasma levels in patients receiving low and conventional doses of fluphenazine decanoate. Psychopharmacology (Berl). 1986;88(4):480-483.
20. Marder SR, Hubbard JW, Van Putten T, et al. Pharmacokinetics of long-acting injectable neuroleptic drugs: clinical implications. Psychopharmacology (Berl). 1989;98(4):433-439.
21. Mavroidis ML, Kanter DR, Hirschowitz J, et al. Fluphenazine plasma levels and clinical response. J Clin Psychiatry. 1984;45(9):370-373.
22. Balant-Gorgia AE, Balant LP, Andreoli A. Pharmacokinetic optimisation of the treatment of psychosis. Clin Pharmacokinet. 1993;25(3):217-236.
23. Van Putten T, Marder SR, Wirshing WC, et al. Neuroleptic plasma levels. Schizophr Bull. 1991;17(2):197-216.
24. Dahl SG. Plasma level monitoring of antipsychotic drugs. Clinical utility. Clin Pharmacokinet. 1986;11(1):36-61.
25. Miller RS, Peterson GM, McLean S, et al. Monitoring plasma levels of fluphenazine during chronic therapy with fluphenazine decanoate. J Clin Pharm Ther. 1995;20(2):55-62.
26. Gitlin MJ, Midha KK, Fogelson D, et al. Persistence of fluphenazine in plasma after decanoate withdrawal. J Clin Psychopharmacol. 1988;8(1):53-56.
27. Wistedt B, Wiles D, Kolakowska T. Slow decline of plasma drug and prolactin levels after discontinuation of chronic treatment with depot neuroleptics. Lancet. 1981;1(8230):1163.
28. Wistedt B, Jørgensen A, Wiles D. A depot neuroleptic withdrawal study. Plasma concentration of fluphenazine and flupenthixol and relapse frequency. Psychopharmacology (Berl). 1982;78(4):301-304.
29. Risperdal Consta [package insert]. Titusville, NJ: Janssen Pharmaceuticals, Inc.; February 2017.
30. Marder SR, Conley R, Ereshefsky L, et al. Clinical guidelines: dosing and switching strategies for long-acting risperidone. J Clin Psychiatry. 2003;64(suppl 16):41-46.
31. Invega Sustenna [package insert]. Titusville, NJ: Janssen Pharmaceuticals, Inc.; February 2017.
32. Invega Trinza [package insert]. Titusville, NJ: Janssen Pharmaceuticals, Inc.; February 2017.
33. Abilify Maintena [package insert]. Rockville, MD: Otsuka America Pharmaceutical, Inc.; December 2016.
34. Artistada [package insert]. Waltham, MA: Alkermes, Inc.; June 2017.
35. Zyprexa Relprevv [package insert]. Indianapolis; IN: Eli Lilly and Co.; February 2017.
36. Heres S, Kraemer S, Bergstrom RF, et al. Pharmacokinetics of olanzapine long-acting injection: the clinical perspective. Int Clin Psychopharmacol. 2014;29(6):299-312.
37. Detke HC, Zhao F, Garhyan P, et al. Dose correspondence between olanzapine long-acting injection and oral olanzapine: recommendations for switching. Int Clin Psychopharmacol. 2011;26(1):35-42.
Antipsychotics linked to increased body fat, insulin resistance in children
Twelve weeks of treatment with three different antipsychotics commonly used in children and adolescents with disruptive behavioral disorders was associated with rapid-onset adverse changes in adiposity and insulin sensitivity, results of a randomized clinical trial show.
The adverse effect on adiposity was greatest for children aged 6-18 years who received olanzapine, but was also seen in study participants randomized to receive risperidone or aripiprazole, according to results of the study published in JAMA Psychiatry.
These findings inform the risk-benefit analysis for off-label pediatric use of antipsychotics for disruptive behavior disorders, Ginger E. Nicol, MD, of Washington University, St. Louis, and colleagues wrote.
“The potential psychiatric benefits of antipsychotic use in this population, evident in this trial and others, should be carefully weighed against the potential for childhood onset of abdominal obesity and insulin resistance that – compared with adult onset – further increases long-term risk for [type 2 diabetes], cardiovascular disease, and related conditions,” the researchers wrote.
This randomized, prospective clinical trial included 144 antipsychotic-naive children and adolescents aged 6-18 years who had been diagnosed with one or more psychiatric disorders and clinically significant aggression. They were randomly and evenly assigned to 12 weeks of treatment with oral olanzapine, risperidone, or aripiprazole.
Those who received olanzapine had a 4.12% increase in percentage total body fat from baseline to week 12, as measured by dual-energy x-ray absorptiometry (DXA). That increase was significantly greater than the total body fat increases of 1.18% for risperidone and 1.66% for aripiprazole seen over that same time period (P less than .001 for time-by-treatment interaction), the researchers reported.
Insulin sensitivity decreased over time in the pooled study sample, with significant changes reported in insulin-stimulated glucose rate of disappearance, glucose rate of appearance, and glycerol rate of appearance. The changes did not differ significantly across the three treatment groups.
The lack of a placebo group in this study makes it unclear how much of the body fat and insulin sensitivity changes were attributable to antipsychotic medication. Psychiatric symptoms did improve significantly with treatment, the researchers noted.
Dr. Nicol reported research funding from the National Institute of Mental Health, Otsuka America Pharmaceutical, Alkermes PLC, The Sidney R. Baer Jr. Foundation, and the Center for Brain Research in Mood Disorders at Washington University in St Louis. Co-authors reported financial ties to Reviva Pharmaceuticals, Sunovion Pharmaceuticals, Indivior, Amgen, and other companies.
SOURCE: Nicol GE et al. JAMA Psychiatry. 2018 Jun 13. doi: 10.1001/jamapsychiatry.2018.1088.
The findings of this study by Nicol and colleagues corroborate previous reports that used conventional anthropometry to document the metabolic adverse effects of second-generation antipsychotic medications in children and adolescents, according to authors of an accompanying editorial.
The current study used “gold standard” methods of measurement to demonstrate that 12 weeks of treatment with low-dose olanzapine, risperidone, or aripiprazole led to rapid-onset changes in adiposity and insulin sensitivity, with larger increases in those who received olanzapine.
“As such, the study even more emphatically underlines the risks to which these vulnerable populations are exposed,” Marc De Hert, MD, PhD, and Johan Detraux wrote.
It is now is clear that the potential psychiatric benefits of off-label antipsychotics need to be weighed against the potential for childhood-onset obesity and insulin resistance, they added. However, longer-term studies that use hard end points, such as new-onset diabetes or cardiovascular disease are needed.
Marc De Hert, MD, PhD, and Johan Detraux, are with Katholieke Universiteit Leuven, Kortenberg, Belgium. These comments are from an accompanying editorial (JAMA Psychiatry. 2018 Jun 13. doi: 10.1001/jamapsychiatry.2018.1080 ). Dr. De Hert reported no disclosures. Mr. Detraux reported partial support by the Janssen Academy for work on the the Belgian Discussion Board on Antipsychotic Treatment.
The findings of this study by Nicol and colleagues corroborate previous reports that used conventional anthropometry to document the metabolic adverse effects of second-generation antipsychotic medications in children and adolescents, according to authors of an accompanying editorial.
The current study used “gold standard” methods of measurement to demonstrate that 12 weeks of treatment with low-dose olanzapine, risperidone, or aripiprazole led to rapid-onset changes in adiposity and insulin sensitivity, with larger increases in those who received olanzapine.
“As such, the study even more emphatically underlines the risks to which these vulnerable populations are exposed,” Marc De Hert, MD, PhD, and Johan Detraux wrote.
It is now is clear that the potential psychiatric benefits of off-label antipsychotics need to be weighed against the potential for childhood-onset obesity and insulin resistance, they added. However, longer-term studies that use hard end points, such as new-onset diabetes or cardiovascular disease are needed.
Marc De Hert, MD, PhD, and Johan Detraux, are with Katholieke Universiteit Leuven, Kortenberg, Belgium. These comments are from an accompanying editorial (JAMA Psychiatry. 2018 Jun 13. doi: 10.1001/jamapsychiatry.2018.1080 ). Dr. De Hert reported no disclosures. Mr. Detraux reported partial support by the Janssen Academy for work on the the Belgian Discussion Board on Antipsychotic Treatment.
The findings of this study by Nicol and colleagues corroborate previous reports that used conventional anthropometry to document the metabolic adverse effects of second-generation antipsychotic medications in children and adolescents, according to authors of an accompanying editorial.
The current study used “gold standard” methods of measurement to demonstrate that 12 weeks of treatment with low-dose olanzapine, risperidone, or aripiprazole led to rapid-onset changes in adiposity and insulin sensitivity, with larger increases in those who received olanzapine.
“As such, the study even more emphatically underlines the risks to which these vulnerable populations are exposed,” Marc De Hert, MD, PhD, and Johan Detraux wrote.
It is now is clear that the potential psychiatric benefits of off-label antipsychotics need to be weighed against the potential for childhood-onset obesity and insulin resistance, they added. However, longer-term studies that use hard end points, such as new-onset diabetes or cardiovascular disease are needed.
Marc De Hert, MD, PhD, and Johan Detraux, are with Katholieke Universiteit Leuven, Kortenberg, Belgium. These comments are from an accompanying editorial (JAMA Psychiatry. 2018 Jun 13. doi: 10.1001/jamapsychiatry.2018.1080 ). Dr. De Hert reported no disclosures. Mr. Detraux reported partial support by the Janssen Academy for work on the the Belgian Discussion Board on Antipsychotic Treatment.
Twelve weeks of treatment with three different antipsychotics commonly used in children and adolescents with disruptive behavioral disorders was associated with rapid-onset adverse changes in adiposity and insulin sensitivity, results of a randomized clinical trial show.
The adverse effect on adiposity was greatest for children aged 6-18 years who received olanzapine, but was also seen in study participants randomized to receive risperidone or aripiprazole, according to results of the study published in JAMA Psychiatry.
These findings inform the risk-benefit analysis for off-label pediatric use of antipsychotics for disruptive behavior disorders, Ginger E. Nicol, MD, of Washington University, St. Louis, and colleagues wrote.
“The potential psychiatric benefits of antipsychotic use in this population, evident in this trial and others, should be carefully weighed against the potential for childhood onset of abdominal obesity and insulin resistance that – compared with adult onset – further increases long-term risk for [type 2 diabetes], cardiovascular disease, and related conditions,” the researchers wrote.
This randomized, prospective clinical trial included 144 antipsychotic-naive children and adolescents aged 6-18 years who had been diagnosed with one or more psychiatric disorders and clinically significant aggression. They were randomly and evenly assigned to 12 weeks of treatment with oral olanzapine, risperidone, or aripiprazole.
Those who received olanzapine had a 4.12% increase in percentage total body fat from baseline to week 12, as measured by dual-energy x-ray absorptiometry (DXA). That increase was significantly greater than the total body fat increases of 1.18% for risperidone and 1.66% for aripiprazole seen over that same time period (P less than .001 for time-by-treatment interaction), the researchers reported.
Insulin sensitivity decreased over time in the pooled study sample, with significant changes reported in insulin-stimulated glucose rate of disappearance, glucose rate of appearance, and glycerol rate of appearance. The changes did not differ significantly across the three treatment groups.
The lack of a placebo group in this study makes it unclear how much of the body fat and insulin sensitivity changes were attributable to antipsychotic medication. Psychiatric symptoms did improve significantly with treatment, the researchers noted.
Dr. Nicol reported research funding from the National Institute of Mental Health, Otsuka America Pharmaceutical, Alkermes PLC, The Sidney R. Baer Jr. Foundation, and the Center for Brain Research in Mood Disorders at Washington University in St Louis. Co-authors reported financial ties to Reviva Pharmaceuticals, Sunovion Pharmaceuticals, Indivior, Amgen, and other companies.
SOURCE: Nicol GE et al. JAMA Psychiatry. 2018 Jun 13. doi: 10.1001/jamapsychiatry.2018.1088.
Twelve weeks of treatment with three different antipsychotics commonly used in children and adolescents with disruptive behavioral disorders was associated with rapid-onset adverse changes in adiposity and insulin sensitivity, results of a randomized clinical trial show.
The adverse effect on adiposity was greatest for children aged 6-18 years who received olanzapine, but was also seen in study participants randomized to receive risperidone or aripiprazole, according to results of the study published in JAMA Psychiatry.
These findings inform the risk-benefit analysis for off-label pediatric use of antipsychotics for disruptive behavior disorders, Ginger E. Nicol, MD, of Washington University, St. Louis, and colleagues wrote.
“The potential psychiatric benefits of antipsychotic use in this population, evident in this trial and others, should be carefully weighed against the potential for childhood onset of abdominal obesity and insulin resistance that – compared with adult onset – further increases long-term risk for [type 2 diabetes], cardiovascular disease, and related conditions,” the researchers wrote.
This randomized, prospective clinical trial included 144 antipsychotic-naive children and adolescents aged 6-18 years who had been diagnosed with one or more psychiatric disorders and clinically significant aggression. They were randomly and evenly assigned to 12 weeks of treatment with oral olanzapine, risperidone, or aripiprazole.
Those who received olanzapine had a 4.12% increase in percentage total body fat from baseline to week 12, as measured by dual-energy x-ray absorptiometry (DXA). That increase was significantly greater than the total body fat increases of 1.18% for risperidone and 1.66% for aripiprazole seen over that same time period (P less than .001 for time-by-treatment interaction), the researchers reported.
Insulin sensitivity decreased over time in the pooled study sample, with significant changes reported in insulin-stimulated glucose rate of disappearance, glucose rate of appearance, and glycerol rate of appearance. The changes did not differ significantly across the three treatment groups.
The lack of a placebo group in this study makes it unclear how much of the body fat and insulin sensitivity changes were attributable to antipsychotic medication. Psychiatric symptoms did improve significantly with treatment, the researchers noted.
Dr. Nicol reported research funding from the National Institute of Mental Health, Otsuka America Pharmaceutical, Alkermes PLC, The Sidney R. Baer Jr. Foundation, and the Center for Brain Research in Mood Disorders at Washington University in St Louis. Co-authors reported financial ties to Reviva Pharmaceuticals, Sunovion Pharmaceuticals, Indivior, Amgen, and other companies.
SOURCE: Nicol GE et al. JAMA Psychiatry. 2018 Jun 13. doi: 10.1001/jamapsychiatry.2018.1088.
FROM JAMA PSYCHIATRY
Key clinical point:
Major finding: Olanzapine-treated youths had the highest percentage total body fat increase (4.12%), but substantial increases were also noted for risperidone (1.18%) and aripiprazole (1.66%).
Study details: A randomized clinical trial including 144 antipsychotic-naive youths aged 6-18 years with one or more psychiatric disorders and clinically significant aggression.
Disclosures: Study authors reported disclosures related to Otsuka America Pharmaceutical, Alkermes PLC, Reviva Pharmaceuticals, Sunovion Pharmaceuticals, Indivior, Amgen, and other companies.
Source: Nicol GE et al. JAMA Psychiatry. 2018 Jun 13. doi:10.1001/jamapsychiatry.2018.1088.
Sudden-onset memory problems, visual hallucinations, and odd behaviors
CASE A rapid decline
Ms. D, age 62, presents to a psychiatric emergency room (ER) after experiencing visual hallucinations, exhibiting odd behaviors, and having memory problems. On interview, she is disoriented, distractible, tearful, and tangential. She plays with her shirt and glasses, and occasionally shouts. She perseverates on “the aerialists,” acrobatic children she has been seeing in her apartment. She becomes distressed and shouts, “I would love to just get them!”
Ms. D is unable to provide an account of her history. Collateral information is obtained from her daughter, who has brought Ms. D to the ER for evaluation. She reports that her mother has no relevant medical or psychiatric history, and does not take any medications, except a mixture of Chinese herbs that she brews into a tea.
Ms. D’s daughter says that her mother began to deteriorate 5 months ago, after she traveled to California to care for her sister, who was seriously ill and passed away. After Ms. D returned, she would cry frequently. She also appeared “spaced out,” complained of feeling dizzy, and frequently misplaced belongings. Three months before presenting to the ER, she began to experience weakness, fatigue, and difficulty walking. Her daughter became more worried 2 months ago, when Ms. D began sleeping with her purse and hiding her belongings around their house. When asked about these odd behaviors, Ms. D claimed that “the aerialists” were climbing through her windows at night and stealing her things.
A week before seeking treatment at the ER, Ms. D’s daughter had taken her to a neurologist at another facility for clinical evaluation. An MRI of the brain showed minimal dilation in the subarachnoid space and a focal 1 cm lipoma in the anterior falx cerebri, but was otherwise unremarkable. However, Ms. D’s symptoms continued to worsen, and began to interfere with her ability to care for herself.
The team in the psychiatric ER attributes Ms. D’s symptoms to a severe, psychotic depressive episode. They admit her to the psychiatric inpatient unit for further evaluation.
[polldaddy:10012742]
Continue to: The authors' observations
The authors’ observations
Ms. D was plagued by several mood and psychotic symptoms. Such symptoms can arise from many different psychiatric or organic etiologies. In Ms. D’s case, several aspects of her presentation suggest that her illness was psychiatric. The severe illness of a beloved family member is a significant stressor that could cause a great deal of grief and devastation, possibly leading to depression. Indeed, Ms. D’s daughter noticed that her mother was crying frequently, which is consistent with grief or depression.
Memory problems, which might manifest as misplacing belongings, can also indicate a depressive illness, especially in older patients. Moreover, impaired concentration, which can cause one to appear “spaced out” or distractible, is a core symptom of major depressive disorder. Sadness and grief also can be appropriate during bereavement and in response to significant losses. Therefore, in Ms. D’s case, it is possible her frequent crying, “spaced out” appearance, and other mood symptoms she experienced immediately after caring for her sister were an appropriate response to her sister’s illness and death.
However, other aspects of Ms. D’s presentation suggested an organic etiology. Her rapid deterioration and symptom onset relatively late in life were consistent with dementia and malignancy. Her complaint of feeling dizzy suggested a neurologic process was affecting her vestibular system. Finally, while psychiatric disorders can certainly cause visual hallucinations, they occur in only a small percentage of cases.1 Visual hallucinations are commonly associated with delirium, intoxication, and neurologic illness.
Continue to: EVALUATION Severe impairment
EVALUATION Severe impairment
On the psychiatric inpatient unit, Ms. D remains unable to give a coherent account of her illness or recent events. During interviews, she abruptly shifts from laughing to crying for no apparent reason. While answering questions, her responses trail off and she appears to forget what she had been saying. However, she continues to speak at length about “the aerialists,” stating that she sees them living in her wardrobe and jumping from rooftop to rooftop in her neighborhood.
A mental status examination finds evidence of severe cognitive impairment. Ms. D is unable to identify the correct date, time, or place, and appears surprised when told she is in a hospital. She can repeat the names of 3 objects but cannot recall them a few minutes later. Finally, she scores a 14 on the Mini-Mental State Examination (MMSE) and a 5 on the Montreal Cognitive Assessment (MoCA), indicating severe impairment.
On the unit, Ms. D cannot remember the location of her room or bathroom, and even when given directions, she needs to be escorted to her destination. Her gait is unsteady and wide-spaced, and she walks on her toes at times. When food is placed before her, she needs to be shown how to take the lids off containers, pick up utensils, and start eating.
All laboratory results are unremarkable, including a complete blood count, basic metabolic panel, liver function tests, gamma-glutamyl transpeptidase, magnesium, phosphate, thyroid-stimulating hormone, vitamin B12, methylmalonic acid, homocysteine, folate, erythrocyte sedimentation rate, C-reactive protein, antinuclear antibodies, rapid plasma reagin, human immunodeficiency virus, and Lyme titers. The team also considers Ms. D’s history of herbal medicine use, because herbal mixtures can contain heavy metals and other contaminants. However, all toxicology results are normal, including arsenic, mercury, lead, copper, and zinc.
To address her symptoms, Ms. D is given risperidone, 0.5 mg twice a day, and donepezil, 5 mg/d.
[polldaddy:10012743]
Continue to: The authors' observations
The authors’ observations
Despite her persistent psychiatric symptoms, Ms. D had several neurologic symptoms that warranted further investigation. Her abrupt shifts from laughter to tears for no apparent reason were consistent with pseudobulbar affect. Her inability to remember how to use utensils during meals was consistent with apraxia. Finally, her abnormal gait raised concern for a process affecting her motor system.
OUTCOME A rare disorder
Given the psychiatry team’s suspicions for a neurologic etiology of Ms. D’s symptoms, an MRI of her brain is repeated. The results are notable for abnormal restricted diffusion in the caudate and putamen bilaterally, which is consistent with Creutzfeldt-Jakob disease (CJD). EEG shows moderate diffuse cerebral dysfunction, frontal intermittent delta activity, and diffuse cortical hyperexcitability, consistent with early- to mid-onset prion disease. Upon evaluation by the neurology team, Ms. D appears fearful, suspicious, and disorganized, but her examination does not reveal additional significant sensorimotor findings.
Ms. D is transferred to the neurology service for further workup and management. A lumbar puncture is positive for real-time quaking-induced conversion (RT-QuIC) and 14-3-3 protein with elevated tau proteins; these findings also are consistent with CJD. She develops transaminitis, with an alanine transaminase (ALT) of 127 and aspartate transaminase (AST) of 355, and a malignancy is suspected. However, CT scans of the chest, abdomen, and pelvis show no evidence of malignancy, and an extensive gastrointestinal workup is unremarkable, including anti-smooth muscle antibodies, anti-liver-kidney microsomal antibody, antimitochondrial antibodies, gliadin antibody, alpha-1 antitrypsin, liver/kidney microsomal antibody, and hepatitis serologies. While on the neurology service, risperidone and donepezil are discontinued because
After discontinuing these medications, she is evaluated by the psychiatry consult team for mood lability. The psychiatry consult team recommends quetiapine, which is later started at 25 mg nightly at bedtime.
Clinically, Ms. D’s mental status continues to deteriorate. She becomes nonverbal and minimally able to follow commands. She is ultimately discharged to an inpatient hospice for end-of-life care and the team recommends that she continue with quetiapine once there.
Continue to: The authors' observations
The authors’ observations
CJD is a rare, rapidly progressive, fatal form of dementia. In the United States, the incidence is approximately 1 to 1.5 cases per 1 million people each year.2 There are various forms of the disease. Sporadic CJD is the most common, representing 85% of cases.3 Sporadic CJD typically occurs in patients in their 60s and quickly leads to death—50% of patients die within 5 months, and 90% of patients die within 1 year.2,3 The illness is hypothesized to arise from the production of misfolded prion proteins, ultimately leading to vacuolation, neuronal loss, and the spongiform appearance characteristic of CJD.3,4
Psychiatric symptoms have long been acknowledged as a feature of CJD. Recent data indicates that psychiatric symptoms occur in 90% to 92% of cases.5,6 Sleep disturbances and depressive symptoms, including vegetative symptoms, anhedonia, and tearfulness, appear to be most common.5 Psychotic symptoms occur in approximately 42% of cases and may include persecutory and paranoid delusions, as well as an array of vivid auditory, visual, and tactile hallucinations.5,7
There is also evidence that psychiatric symptoms may be an early marker of CJD.5,8 A Mayo Clinic study found that psychiatric symptoms occurred within the prodromal phase of CJD in 26% of cases, and psychiatric symptoms occurred within the first 100 days of illness in 86% of cases.5
Case reports have described patients with CJD who initially presented with depression, psychosis, and other psychiatric symptoms.9-11 Interestingly, there have been cases with only psychiatric symptoms, and no neurologic symptoms until relatively late in the illness.10,11 Several patients with CJD have been evaluated in psychiatric ERs, admitted to psychiatric hospitals, and treated with psychiatric medications and ECT.5,9 In one study, 44% of CJD cases were misdiagnosed as “psychiatric patients” due to the prominence of their psychiatric symptomatology.8
Continue to: Making the diagnosis in psychiatric settings
Making the diagnosis in psychiatric settings. Often, the most difficult aspect of CJD is making the diagnosis.3,12 Sporadic CJD in particular can vary widely in its clinical presentation.3 The core clinical feature of CJD is rapidly progressive dementia, so suspect CJD in these patients. However, CJD can be difficult to distinguish from other rapidly progressive dementias, such as autoimmune and paraneoplastic encephalopathies.2,3 The presence of neurologic features, specifically myoclonus, akinetic mutism, and visual, cerebellar, and extrapyramidal symptoms, should also be considered a red flag for the disorder3 (Table).
Finally, positive findings on MRI, EEG, or CSF assay can indicate a probable diagnosis of CJD.13 MRI, particularly diffusion weighted imaging (DWI) and fluid-attenuated inversion recovery (FLAIR), is recognized as the most studied, sensitive, and overall useful neuroimaging modality for detecting CJD.2,3,12 Although the appearance of CJD on MRI can vary widely, asymmetric hyperintensities in ≥3 cortical gyri, particularly in the frontal and parietal lobes, provide strong evidence of CJD and are observed in 80% to 81% of cases.4,12 Asymmetric hyperintensities in the basal ganglia, particularly the caudate and rostral putamen, are observed in 69% to 70% of cases.4,12,13
EEG and CSF assay also can be useful for making the diagnosis. While diffuse slowing and frontal rhythmic delta activity appear early in the course of CJD, periodic sharp wave complexes emerge later in the illness.4 However, EEG findings are not diagnostic, because periodic sharp wave complexes are seen in only two-thirds of CJD cases and also occur in other neurologic illnesses.3,4 In recent years, lumbar puncture with subsequent CSF testing has become increasingly useful in detecting the illness. The presence of the 14-3-3 protein and tau protein is highly sensitive, although not specific, for CJD.3 A definite diagnosis of CJD requires discovery of the misfolded prion proteins, such as by RT-QuIC or brain biopsy.2,3,13
Management of CJD in psychiatric patients. CJD is an invariably fatal disease for which there is no effective cure or disease modifying treatment.2 Therefore, supportive therapies are the mainstay of care. Psychotropic medications can be used to provide symptom relief. While the sleep disturbances, anxiety, and agitation/hallucinations associated with CJD appear to respond well to hypnotic, anxiolytic, and antipsychotic medications, respectively, antidepressants and mood-stabilizing medications appear to have little benefit for patients with CJD.5 During the final stages of the disease, patients may suffer from akinetic mutism and inability to swallow, which often leads to aspiration pneumonia.14 Patients should also be offered end-of-life counseling, planning, and care, and provided with other comfort measures wherever possible (Figure).
Continue to: Bottom Line
Bottom Line
Patients with Creutzfeldt-Jakob disease (CJD) may present to psychiatric settings, particularly to a psychiatric emergency room. Consider CJD as a possible etiology in patients with rapidly progressive dementia, depression, and psychosis. CJD is invariably fatal and there is no effective disease-modifying treatment. Supportive therapies are the mainstay of care.
Related Resources
- National Institute of Neurological Disorders and Stroke. Creutzfeldt-Jakob disease fact sheet. http://www.ninds.nih.gov/disorders/cjd/detail_cjd.htm.
- Centers for Disease Control and Prevention. Creutzfeldt-Jakob disease, classic (CJD). http://www.cdc.gov/prions/cjd.
Drug Brand Names
Donepezil • Aricept
Risperidone • Risperdal
Quetiapine • Seroquel
1. Resnick PJ. The detection of malingered psychosis. Psychiatr Clin North Am. 1999;22(1):159-172.
2. Bucelli RC, Ances BM. Diagnosis and evaluation of a patient with rapidly progressive dementia. Mo Med. 2013;110(5):422-428.
3. Manix M, Kalakoti P, Henry M, et al. Creutzfeldt-Jakob disease: updated diagnostic criteria, treatment algorithm, and the utility of brain biopsy. Neurosurg Focus. 2015;39(5):E2.
4. Puoti G, Bizzi A, Forloni G, et al. Sporadic human prion diseases: molecular insights and diagnosis. Lancet Neurol. 2012;11(7):618-628.
5. Wall CA, Rummans TA, Aksamit AJ, et al. Psychiatric manifestations of Creutzfeldt-Jakob disease: a 25-year analysis. J Neuropsychiatry Clin Neurosci. 2005;17(4):489-495.
6. Krasnianski A, Bohling GT, Harden M, et al. Psychiatric symptoms in patients with sporadic Creutzfeldt-Jakob disease in Germany. J Clin Psychiatry. 2015;76(9):1209-1215.
7. Javed Q, Alam F, Krishna S, et al. An unusual case of sporadic Creutzfeldt-Jakob disease (CJD). BMJ Case Rep. 2010;pii: bcr1220092576. doi:10.1136/bcr.12.2009.2576.
8. Abudy A, Juven-Wetzler A, Zohar J. The different faces of Creutzfeldt-Jacob disease CJD in psychiatry. Gen Hosp Psychiatry. 2014;36(3):245-248.
9. Jiang TT, Moses H, Gordon H, et al. Sporadic Creutzfeldt-Jakob disease presenting as major depression. South Med J. 1999;92(8):807-808.
10. Ali R, Baborie A, Larner AJ et al. Psychiatric presentation of sporadic Creutzfeldt-Jakob disease: a challenge to current diagnostic criteria. J Neuropsychiatry Clin Neurosci. 2013;25(4):335-338.
11. Gençer AG, Pelin Z, Küçükali CI., et al. Creutzfeldt-Jakob disease. Psychogeriatrics. 2011;11(2):119-124.
12. Caobelli F, Cobelli M, Pizzocaro C, et al. The role of neuroimaging in evaluating patients affected by Creutzfeldt-Jakob disease: a systematic review of the literature. J Neuroimaging. 2015;25(1):2-13.
13. Centers for Disease Control and Prevention. CDC's diagnostic criteria for Creutzfeldt-Jakob disease, 2010. http://www.cdc.gov/prions/cjd/diagnostic-criteria.html. Updated February 11, 2015. Accessed August 2, 2016.
14. Martindale JL, Geschwind MD, Miller BL. Psychiatric and neuroimaging findings in Creutzfeldt-Jakob disease. Curr Psychiatry Rep. 2003;5(1):43-46.
CASE A rapid decline
Ms. D, age 62, presents to a psychiatric emergency room (ER) after experiencing visual hallucinations, exhibiting odd behaviors, and having memory problems. On interview, she is disoriented, distractible, tearful, and tangential. She plays with her shirt and glasses, and occasionally shouts. She perseverates on “the aerialists,” acrobatic children she has been seeing in her apartment. She becomes distressed and shouts, “I would love to just get them!”
Ms. D is unable to provide an account of her history. Collateral information is obtained from her daughter, who has brought Ms. D to the ER for evaluation. She reports that her mother has no relevant medical or psychiatric history, and does not take any medications, except a mixture of Chinese herbs that she brews into a tea.
Ms. D’s daughter says that her mother began to deteriorate 5 months ago, after she traveled to California to care for her sister, who was seriously ill and passed away. After Ms. D returned, she would cry frequently. She also appeared “spaced out,” complained of feeling dizzy, and frequently misplaced belongings. Three months before presenting to the ER, she began to experience weakness, fatigue, and difficulty walking. Her daughter became more worried 2 months ago, when Ms. D began sleeping with her purse and hiding her belongings around their house. When asked about these odd behaviors, Ms. D claimed that “the aerialists” were climbing through her windows at night and stealing her things.
A week before seeking treatment at the ER, Ms. D’s daughter had taken her to a neurologist at another facility for clinical evaluation. An MRI of the brain showed minimal dilation in the subarachnoid space and a focal 1 cm lipoma in the anterior falx cerebri, but was otherwise unremarkable. However, Ms. D’s symptoms continued to worsen, and began to interfere with her ability to care for herself.
The team in the psychiatric ER attributes Ms. D’s symptoms to a severe, psychotic depressive episode. They admit her to the psychiatric inpatient unit for further evaluation.
[polldaddy:10012742]
Continue to: The authors' observations
The authors’ observations
Ms. D was plagued by several mood and psychotic symptoms. Such symptoms can arise from many different psychiatric or organic etiologies. In Ms. D’s case, several aspects of her presentation suggest that her illness was psychiatric. The severe illness of a beloved family member is a significant stressor that could cause a great deal of grief and devastation, possibly leading to depression. Indeed, Ms. D’s daughter noticed that her mother was crying frequently, which is consistent with grief or depression.
Memory problems, which might manifest as misplacing belongings, can also indicate a depressive illness, especially in older patients. Moreover, impaired concentration, which can cause one to appear “spaced out” or distractible, is a core symptom of major depressive disorder. Sadness and grief also can be appropriate during bereavement and in response to significant losses. Therefore, in Ms. D’s case, it is possible her frequent crying, “spaced out” appearance, and other mood symptoms she experienced immediately after caring for her sister were an appropriate response to her sister’s illness and death.
However, other aspects of Ms. D’s presentation suggested an organic etiology. Her rapid deterioration and symptom onset relatively late in life were consistent with dementia and malignancy. Her complaint of feeling dizzy suggested a neurologic process was affecting her vestibular system. Finally, while psychiatric disorders can certainly cause visual hallucinations, they occur in only a small percentage of cases.1 Visual hallucinations are commonly associated with delirium, intoxication, and neurologic illness.
Continue to: EVALUATION Severe impairment
EVALUATION Severe impairment
On the psychiatric inpatient unit, Ms. D remains unable to give a coherent account of her illness or recent events. During interviews, she abruptly shifts from laughing to crying for no apparent reason. While answering questions, her responses trail off and she appears to forget what she had been saying. However, she continues to speak at length about “the aerialists,” stating that she sees them living in her wardrobe and jumping from rooftop to rooftop in her neighborhood.
A mental status examination finds evidence of severe cognitive impairment. Ms. D is unable to identify the correct date, time, or place, and appears surprised when told she is in a hospital. She can repeat the names of 3 objects but cannot recall them a few minutes later. Finally, she scores a 14 on the Mini-Mental State Examination (MMSE) and a 5 on the Montreal Cognitive Assessment (MoCA), indicating severe impairment.
On the unit, Ms. D cannot remember the location of her room or bathroom, and even when given directions, she needs to be escorted to her destination. Her gait is unsteady and wide-spaced, and she walks on her toes at times. When food is placed before her, she needs to be shown how to take the lids off containers, pick up utensils, and start eating.
All laboratory results are unremarkable, including a complete blood count, basic metabolic panel, liver function tests, gamma-glutamyl transpeptidase, magnesium, phosphate, thyroid-stimulating hormone, vitamin B12, methylmalonic acid, homocysteine, folate, erythrocyte sedimentation rate, C-reactive protein, antinuclear antibodies, rapid plasma reagin, human immunodeficiency virus, and Lyme titers. The team also considers Ms. D’s history of herbal medicine use, because herbal mixtures can contain heavy metals and other contaminants. However, all toxicology results are normal, including arsenic, mercury, lead, copper, and zinc.
To address her symptoms, Ms. D is given risperidone, 0.5 mg twice a day, and donepezil, 5 mg/d.
[polldaddy:10012743]
Continue to: The authors' observations
The authors’ observations
Despite her persistent psychiatric symptoms, Ms. D had several neurologic symptoms that warranted further investigation. Her abrupt shifts from laughter to tears for no apparent reason were consistent with pseudobulbar affect. Her inability to remember how to use utensils during meals was consistent with apraxia. Finally, her abnormal gait raised concern for a process affecting her motor system.
OUTCOME A rare disorder
Given the psychiatry team’s suspicions for a neurologic etiology of Ms. D’s symptoms, an MRI of her brain is repeated. The results are notable for abnormal restricted diffusion in the caudate and putamen bilaterally, which is consistent with Creutzfeldt-Jakob disease (CJD). EEG shows moderate diffuse cerebral dysfunction, frontal intermittent delta activity, and diffuse cortical hyperexcitability, consistent with early- to mid-onset prion disease. Upon evaluation by the neurology team, Ms. D appears fearful, suspicious, and disorganized, but her examination does not reveal additional significant sensorimotor findings.
Ms. D is transferred to the neurology service for further workup and management. A lumbar puncture is positive for real-time quaking-induced conversion (RT-QuIC) and 14-3-3 protein with elevated tau proteins; these findings also are consistent with CJD. She develops transaminitis, with an alanine transaminase (ALT) of 127 and aspartate transaminase (AST) of 355, and a malignancy is suspected. However, CT scans of the chest, abdomen, and pelvis show no evidence of malignancy, and an extensive gastrointestinal workup is unremarkable, including anti-smooth muscle antibodies, anti-liver-kidney microsomal antibody, antimitochondrial antibodies, gliadin antibody, alpha-1 antitrypsin, liver/kidney microsomal antibody, and hepatitis serologies. While on the neurology service, risperidone and donepezil are discontinued because
After discontinuing these medications, she is evaluated by the psychiatry consult team for mood lability. The psychiatry consult team recommends quetiapine, which is later started at 25 mg nightly at bedtime.
Clinically, Ms. D’s mental status continues to deteriorate. She becomes nonverbal and minimally able to follow commands. She is ultimately discharged to an inpatient hospice for end-of-life care and the team recommends that she continue with quetiapine once there.
Continue to: The authors' observations
The authors’ observations
CJD is a rare, rapidly progressive, fatal form of dementia. In the United States, the incidence is approximately 1 to 1.5 cases per 1 million people each year.2 There are various forms of the disease. Sporadic CJD is the most common, representing 85% of cases.3 Sporadic CJD typically occurs in patients in their 60s and quickly leads to death—50% of patients die within 5 months, and 90% of patients die within 1 year.2,3 The illness is hypothesized to arise from the production of misfolded prion proteins, ultimately leading to vacuolation, neuronal loss, and the spongiform appearance characteristic of CJD.3,4
Psychiatric symptoms have long been acknowledged as a feature of CJD. Recent data indicates that psychiatric symptoms occur in 90% to 92% of cases.5,6 Sleep disturbances and depressive symptoms, including vegetative symptoms, anhedonia, and tearfulness, appear to be most common.5 Psychotic symptoms occur in approximately 42% of cases and may include persecutory and paranoid delusions, as well as an array of vivid auditory, visual, and tactile hallucinations.5,7
There is also evidence that psychiatric symptoms may be an early marker of CJD.5,8 A Mayo Clinic study found that psychiatric symptoms occurred within the prodromal phase of CJD in 26% of cases, and psychiatric symptoms occurred within the first 100 days of illness in 86% of cases.5
Case reports have described patients with CJD who initially presented with depression, psychosis, and other psychiatric symptoms.9-11 Interestingly, there have been cases with only psychiatric symptoms, and no neurologic symptoms until relatively late in the illness.10,11 Several patients with CJD have been evaluated in psychiatric ERs, admitted to psychiatric hospitals, and treated with psychiatric medications and ECT.5,9 In one study, 44% of CJD cases were misdiagnosed as “psychiatric patients” due to the prominence of their psychiatric symptomatology.8
Continue to: Making the diagnosis in psychiatric settings
Making the diagnosis in psychiatric settings. Often, the most difficult aspect of CJD is making the diagnosis.3,12 Sporadic CJD in particular can vary widely in its clinical presentation.3 The core clinical feature of CJD is rapidly progressive dementia, so suspect CJD in these patients. However, CJD can be difficult to distinguish from other rapidly progressive dementias, such as autoimmune and paraneoplastic encephalopathies.2,3 The presence of neurologic features, specifically myoclonus, akinetic mutism, and visual, cerebellar, and extrapyramidal symptoms, should also be considered a red flag for the disorder3 (Table).
Finally, positive findings on MRI, EEG, or CSF assay can indicate a probable diagnosis of CJD.13 MRI, particularly diffusion weighted imaging (DWI) and fluid-attenuated inversion recovery (FLAIR), is recognized as the most studied, sensitive, and overall useful neuroimaging modality for detecting CJD.2,3,12 Although the appearance of CJD on MRI can vary widely, asymmetric hyperintensities in ≥3 cortical gyri, particularly in the frontal and parietal lobes, provide strong evidence of CJD and are observed in 80% to 81% of cases.4,12 Asymmetric hyperintensities in the basal ganglia, particularly the caudate and rostral putamen, are observed in 69% to 70% of cases.4,12,13
EEG and CSF assay also can be useful for making the diagnosis. While diffuse slowing and frontal rhythmic delta activity appear early in the course of CJD, periodic sharp wave complexes emerge later in the illness.4 However, EEG findings are not diagnostic, because periodic sharp wave complexes are seen in only two-thirds of CJD cases and also occur in other neurologic illnesses.3,4 In recent years, lumbar puncture with subsequent CSF testing has become increasingly useful in detecting the illness. The presence of the 14-3-3 protein and tau protein is highly sensitive, although not specific, for CJD.3 A definite diagnosis of CJD requires discovery of the misfolded prion proteins, such as by RT-QuIC or brain biopsy.2,3,13
Management of CJD in psychiatric patients. CJD is an invariably fatal disease for which there is no effective cure or disease modifying treatment.2 Therefore, supportive therapies are the mainstay of care. Psychotropic medications can be used to provide symptom relief. While the sleep disturbances, anxiety, and agitation/hallucinations associated with CJD appear to respond well to hypnotic, anxiolytic, and antipsychotic medications, respectively, antidepressants and mood-stabilizing medications appear to have little benefit for patients with CJD.5 During the final stages of the disease, patients may suffer from akinetic mutism and inability to swallow, which often leads to aspiration pneumonia.14 Patients should also be offered end-of-life counseling, planning, and care, and provided with other comfort measures wherever possible (Figure).

Continue to: Bottom Line
Bottom Line
Patients with Creutzfeldt-Jakob disease (CJD) may present to psychiatric settings, particularly to a psychiatric emergency room. Consider CJD as a possible etiology in patients with rapidly progressive dementia, depression, and psychosis. CJD is invariably fatal and there is no effective disease-modifying treatment. Supportive therapies are the mainstay of care.
Related Resources
- National Institute of Neurological Disorders and Stroke. Creutzfeldt-Jakob disease fact sheet. http://www.ninds.nih.gov/disorders/cjd/detail_cjd.htm.
- Centers for Disease Control and Prevention. Creutzfeldt-Jakob disease, classic (CJD). http://www.cdc.gov/prions/cjd.
Drug Brand Names
Donepezil • Aricept
Risperidone • Risperdal
Quetiapine • Seroquel
CASE A rapid decline
Ms. D, age 62, presents to a psychiatric emergency room (ER) after experiencing visual hallucinations, exhibiting odd behaviors, and having memory problems. On interview, she is disoriented, distractible, tearful, and tangential. She plays with her shirt and glasses, and occasionally shouts. She perseverates on “the aerialists,” acrobatic children she has been seeing in her apartment. She becomes distressed and shouts, “I would love to just get them!”
Ms. D is unable to provide an account of her history. Collateral information is obtained from her daughter, who has brought Ms. D to the ER for evaluation. She reports that her mother has no relevant medical or psychiatric history, and does not take any medications, except a mixture of Chinese herbs that she brews into a tea.
Ms. D’s daughter says that her mother began to deteriorate 5 months ago, after she traveled to California to care for her sister, who was seriously ill and passed away. After Ms. D returned, she would cry frequently. She also appeared “spaced out,” complained of feeling dizzy, and frequently misplaced belongings. Three months before presenting to the ER, she began to experience weakness, fatigue, and difficulty walking. Her daughter became more worried 2 months ago, when Ms. D began sleeping with her purse and hiding her belongings around their house. When asked about these odd behaviors, Ms. D claimed that “the aerialists” were climbing through her windows at night and stealing her things.
A week before seeking treatment at the ER, Ms. D’s daughter had taken her to a neurologist at another facility for clinical evaluation. An MRI of the brain showed minimal dilation in the subarachnoid space and a focal 1 cm lipoma in the anterior falx cerebri, but was otherwise unremarkable. However, Ms. D’s symptoms continued to worsen, and began to interfere with her ability to care for herself.
The team in the psychiatric ER attributes Ms. D’s symptoms to a severe, psychotic depressive episode. They admit her to the psychiatric inpatient unit for further evaluation.
[polldaddy:10012742]
Continue to: The authors' observations
The authors’ observations
Ms. D was plagued by several mood and psychotic symptoms. Such symptoms can arise from many different psychiatric or organic etiologies. In Ms. D’s case, several aspects of her presentation suggest that her illness was psychiatric. The severe illness of a beloved family member is a significant stressor that could cause a great deal of grief and devastation, possibly leading to depression. Indeed, Ms. D’s daughter noticed that her mother was crying frequently, which is consistent with grief or depression.
Memory problems, which might manifest as misplacing belongings, can also indicate a depressive illness, especially in older patients. Moreover, impaired concentration, which can cause one to appear “spaced out” or distractible, is a core symptom of major depressive disorder. Sadness and grief also can be appropriate during bereavement and in response to significant losses. Therefore, in Ms. D’s case, it is possible her frequent crying, “spaced out” appearance, and other mood symptoms she experienced immediately after caring for her sister were an appropriate response to her sister’s illness and death.
However, other aspects of Ms. D’s presentation suggested an organic etiology. Her rapid deterioration and symptom onset relatively late in life were consistent with dementia and malignancy. Her complaint of feeling dizzy suggested a neurologic process was affecting her vestibular system. Finally, while psychiatric disorders can certainly cause visual hallucinations, they occur in only a small percentage of cases.1 Visual hallucinations are commonly associated with delirium, intoxication, and neurologic illness.
Continue to: EVALUATION Severe impairment
EVALUATION Severe impairment
On the psychiatric inpatient unit, Ms. D remains unable to give a coherent account of her illness or recent events. During interviews, she abruptly shifts from laughing to crying for no apparent reason. While answering questions, her responses trail off and she appears to forget what she had been saying. However, she continues to speak at length about “the aerialists,” stating that she sees them living in her wardrobe and jumping from rooftop to rooftop in her neighborhood.
A mental status examination finds evidence of severe cognitive impairment. Ms. D is unable to identify the correct date, time, or place, and appears surprised when told she is in a hospital. She can repeat the names of 3 objects but cannot recall them a few minutes later. Finally, she scores a 14 on the Mini-Mental State Examination (MMSE) and a 5 on the Montreal Cognitive Assessment (MoCA), indicating severe impairment.
On the unit, Ms. D cannot remember the location of her room or bathroom, and even when given directions, she needs to be escorted to her destination. Her gait is unsteady and wide-spaced, and she walks on her toes at times. When food is placed before her, she needs to be shown how to take the lids off containers, pick up utensils, and start eating.
All laboratory results are unremarkable, including a complete blood count, basic metabolic panel, liver function tests, gamma-glutamyl transpeptidase, magnesium, phosphate, thyroid-stimulating hormone, vitamin B12, methylmalonic acid, homocysteine, folate, erythrocyte sedimentation rate, C-reactive protein, antinuclear antibodies, rapid plasma reagin, human immunodeficiency virus, and Lyme titers. The team also considers Ms. D’s history of herbal medicine use, because herbal mixtures can contain heavy metals and other contaminants. However, all toxicology results are normal, including arsenic, mercury, lead, copper, and zinc.
To address her symptoms, Ms. D is given risperidone, 0.5 mg twice a day, and donepezil, 5 mg/d.
[polldaddy:10012743]
Continue to: The authors' observations
The authors’ observations
Despite her persistent psychiatric symptoms, Ms. D had several neurologic symptoms that warranted further investigation. Her abrupt shifts from laughter to tears for no apparent reason were consistent with pseudobulbar affect. Her inability to remember how to use utensils during meals was consistent with apraxia. Finally, her abnormal gait raised concern for a process affecting her motor system.
OUTCOME A rare disorder
Given the psychiatry team’s suspicions for a neurologic etiology of Ms. D’s symptoms, an MRI of her brain is repeated. The results are notable for abnormal restricted diffusion in the caudate and putamen bilaterally, which is consistent with Creutzfeldt-Jakob disease (CJD). EEG shows moderate diffuse cerebral dysfunction, frontal intermittent delta activity, and diffuse cortical hyperexcitability, consistent with early- to mid-onset prion disease. Upon evaluation by the neurology team, Ms. D appears fearful, suspicious, and disorganized, but her examination does not reveal additional significant sensorimotor findings.
Ms. D is transferred to the neurology service for further workup and management. A lumbar puncture is positive for real-time quaking-induced conversion (RT-QuIC) and 14-3-3 protein with elevated tau proteins; these findings also are consistent with CJD. She develops transaminitis, with an alanine transaminase (ALT) of 127 and aspartate transaminase (AST) of 355, and a malignancy is suspected. However, CT scans of the chest, abdomen, and pelvis show no evidence of malignancy, and an extensive gastrointestinal workup is unremarkable, including anti-smooth muscle antibodies, anti-liver-kidney microsomal antibody, antimitochondrial antibodies, gliadin antibody, alpha-1 antitrypsin, liver/kidney microsomal antibody, and hepatitis serologies. While on the neurology service, risperidone and donepezil are discontinued because
After discontinuing these medications, she is evaluated by the psychiatry consult team for mood lability. The psychiatry consult team recommends quetiapine, which is later started at 25 mg nightly at bedtime.
Clinically, Ms. D’s mental status continues to deteriorate. She becomes nonverbal and minimally able to follow commands. She is ultimately discharged to an inpatient hospice for end-of-life care and the team recommends that she continue with quetiapine once there.
Continue to: The authors' observations
The authors’ observations
CJD is a rare, rapidly progressive, fatal form of dementia. In the United States, the incidence is approximately 1 to 1.5 cases per 1 million people each year.2 There are various forms of the disease. Sporadic CJD is the most common, representing 85% of cases.3 Sporadic CJD typically occurs in patients in their 60s and quickly leads to death—50% of patients die within 5 months, and 90% of patients die within 1 year.2,3 The illness is hypothesized to arise from the production of misfolded prion proteins, ultimately leading to vacuolation, neuronal loss, and the spongiform appearance characteristic of CJD.3,4
Psychiatric symptoms have long been acknowledged as a feature of CJD. Recent data indicates that psychiatric symptoms occur in 90% to 92% of cases.5,6 Sleep disturbances and depressive symptoms, including vegetative symptoms, anhedonia, and tearfulness, appear to be most common.5 Psychotic symptoms occur in approximately 42% of cases and may include persecutory and paranoid delusions, as well as an array of vivid auditory, visual, and tactile hallucinations.5,7
There is also evidence that psychiatric symptoms may be an early marker of CJD.5,8 A Mayo Clinic study found that psychiatric symptoms occurred within the prodromal phase of CJD in 26% of cases, and psychiatric symptoms occurred within the first 100 days of illness in 86% of cases.5
Case reports have described patients with CJD who initially presented with depression, psychosis, and other psychiatric symptoms.9-11 Interestingly, there have been cases with only psychiatric symptoms, and no neurologic symptoms until relatively late in the illness.10,11 Several patients with CJD have been evaluated in psychiatric ERs, admitted to psychiatric hospitals, and treated with psychiatric medications and ECT.5,9 In one study, 44% of CJD cases were misdiagnosed as “psychiatric patients” due to the prominence of their psychiatric symptomatology.8
Continue to: Making the diagnosis in psychiatric settings
Making the diagnosis in psychiatric settings. Often, the most difficult aspect of CJD is making the diagnosis.3,12 Sporadic CJD in particular can vary widely in its clinical presentation.3 The core clinical feature of CJD is rapidly progressive dementia, so suspect CJD in these patients. However, CJD can be difficult to distinguish from other rapidly progressive dementias, such as autoimmune and paraneoplastic encephalopathies.2,3 The presence of neurologic features, specifically myoclonus, akinetic mutism, and visual, cerebellar, and extrapyramidal symptoms, should also be considered a red flag for the disorder3 (Table).
Finally, positive findings on MRI, EEG, or CSF assay can indicate a probable diagnosis of CJD.13 MRI, particularly diffusion weighted imaging (DWI) and fluid-attenuated inversion recovery (FLAIR), is recognized as the most studied, sensitive, and overall useful neuroimaging modality for detecting CJD.2,3,12 Although the appearance of CJD on MRI can vary widely, asymmetric hyperintensities in ≥3 cortical gyri, particularly in the frontal and parietal lobes, provide strong evidence of CJD and are observed in 80% to 81% of cases.4,12 Asymmetric hyperintensities in the basal ganglia, particularly the caudate and rostral putamen, are observed in 69% to 70% of cases.4,12,13
EEG and CSF assay also can be useful for making the diagnosis. While diffuse slowing and frontal rhythmic delta activity appear early in the course of CJD, periodic sharp wave complexes emerge later in the illness.4 However, EEG findings are not diagnostic, because periodic sharp wave complexes are seen in only two-thirds of CJD cases and also occur in other neurologic illnesses.3,4 In recent years, lumbar puncture with subsequent CSF testing has become increasingly useful in detecting the illness. The presence of the 14-3-3 protein and tau protein is highly sensitive, although not specific, for CJD.3 A definite diagnosis of CJD requires discovery of the misfolded prion proteins, such as by RT-QuIC or brain biopsy.2,3,13
Management of CJD in psychiatric patients. CJD is an invariably fatal disease for which there is no effective cure or disease modifying treatment.2 Therefore, supportive therapies are the mainstay of care. Psychotropic medications can be used to provide symptom relief. While the sleep disturbances, anxiety, and agitation/hallucinations associated with CJD appear to respond well to hypnotic, anxiolytic, and antipsychotic medications, respectively, antidepressants and mood-stabilizing medications appear to have little benefit for patients with CJD.5 During the final stages of the disease, patients may suffer from akinetic mutism and inability to swallow, which often leads to aspiration pneumonia.14 Patients should also be offered end-of-life counseling, planning, and care, and provided with other comfort measures wherever possible (Figure).

Continue to: Bottom Line
Bottom Line
Patients with Creutzfeldt-Jakob disease (CJD) may present to psychiatric settings, particularly to a psychiatric emergency room. Consider CJD as a possible etiology in patients with rapidly progressive dementia, depression, and psychosis. CJD is invariably fatal and there is no effective disease-modifying treatment. Supportive therapies are the mainstay of care.
Related Resources
- National Institute of Neurological Disorders and Stroke. Creutzfeldt-Jakob disease fact sheet. http://www.ninds.nih.gov/disorders/cjd/detail_cjd.htm.
- Centers for Disease Control and Prevention. Creutzfeldt-Jakob disease, classic (CJD). http://www.cdc.gov/prions/cjd.
Drug Brand Names
Donepezil • Aricept
Risperidone • Risperdal
Quetiapine • Seroquel
1. Resnick PJ. The detection of malingered psychosis. Psychiatr Clin North Am. 1999;22(1):159-172.
2. Bucelli RC, Ances BM. Diagnosis and evaluation of a patient with rapidly progressive dementia. Mo Med. 2013;110(5):422-428.
3. Manix M, Kalakoti P, Henry M, et al. Creutzfeldt-Jakob disease: updated diagnostic criteria, treatment algorithm, and the utility of brain biopsy. Neurosurg Focus. 2015;39(5):E2.
4. Puoti G, Bizzi A, Forloni G, et al. Sporadic human prion diseases: molecular insights and diagnosis. Lancet Neurol. 2012;11(7):618-628.
5. Wall CA, Rummans TA, Aksamit AJ, et al. Psychiatric manifestations of Creutzfeldt-Jakob disease: a 25-year analysis. J Neuropsychiatry Clin Neurosci. 2005;17(4):489-495.
6. Krasnianski A, Bohling GT, Harden M, et al. Psychiatric symptoms in patients with sporadic Creutzfeldt-Jakob disease in Germany. J Clin Psychiatry. 2015;76(9):1209-1215.
7. Javed Q, Alam F, Krishna S, et al. An unusual case of sporadic Creutzfeldt-Jakob disease (CJD). BMJ Case Rep. 2010;pii: bcr1220092576. doi:10.1136/bcr.12.2009.2576.
8. Abudy A, Juven-Wetzler A, Zohar J. The different faces of Creutzfeldt-Jacob disease CJD in psychiatry. Gen Hosp Psychiatry. 2014;36(3):245-248.
9. Jiang TT, Moses H, Gordon H, et al. Sporadic Creutzfeldt-Jakob disease presenting as major depression. South Med J. 1999;92(8):807-808.
10. Ali R, Baborie A, Larner AJ et al. Psychiatric presentation of sporadic Creutzfeldt-Jakob disease: a challenge to current diagnostic criteria. J Neuropsychiatry Clin Neurosci. 2013;25(4):335-338.
11. Gençer AG, Pelin Z, Küçükali CI., et al. Creutzfeldt-Jakob disease. Psychogeriatrics. 2011;11(2):119-124.
12. Caobelli F, Cobelli M, Pizzocaro C, et al. The role of neuroimaging in evaluating patients affected by Creutzfeldt-Jakob disease: a systematic review of the literature. J Neuroimaging. 2015;25(1):2-13.
13. Centers for Disease Control and Prevention. CDC's diagnostic criteria for Creutzfeldt-Jakob disease, 2010. http://www.cdc.gov/prions/cjd/diagnostic-criteria.html. Updated February 11, 2015. Accessed August 2, 2016.
14. Martindale JL, Geschwind MD, Miller BL. Psychiatric and neuroimaging findings in Creutzfeldt-Jakob disease. Curr Psychiatry Rep. 2003;5(1):43-46.
1. Resnick PJ. The detection of malingered psychosis. Psychiatr Clin North Am. 1999;22(1):159-172.
2. Bucelli RC, Ances BM. Diagnosis and evaluation of a patient with rapidly progressive dementia. Mo Med. 2013;110(5):422-428.
3. Manix M, Kalakoti P, Henry M, et al. Creutzfeldt-Jakob disease: updated diagnostic criteria, treatment algorithm, and the utility of brain biopsy. Neurosurg Focus. 2015;39(5):E2.
4. Puoti G, Bizzi A, Forloni G, et al. Sporadic human prion diseases: molecular insights and diagnosis. Lancet Neurol. 2012;11(7):618-628.
5. Wall CA, Rummans TA, Aksamit AJ, et al. Psychiatric manifestations of Creutzfeldt-Jakob disease: a 25-year analysis. J Neuropsychiatry Clin Neurosci. 2005;17(4):489-495.
6. Krasnianski A, Bohling GT, Harden M, et al. Psychiatric symptoms in patients with sporadic Creutzfeldt-Jakob disease in Germany. J Clin Psychiatry. 2015;76(9):1209-1215.
7. Javed Q, Alam F, Krishna S, et al. An unusual case of sporadic Creutzfeldt-Jakob disease (CJD). BMJ Case Rep. 2010;pii: bcr1220092576. doi:10.1136/bcr.12.2009.2576.
8. Abudy A, Juven-Wetzler A, Zohar J. The different faces of Creutzfeldt-Jacob disease CJD in psychiatry. Gen Hosp Psychiatry. 2014;36(3):245-248.
9. Jiang TT, Moses H, Gordon H, et al. Sporadic Creutzfeldt-Jakob disease presenting as major depression. South Med J. 1999;92(8):807-808.
10. Ali R, Baborie A, Larner AJ et al. Psychiatric presentation of sporadic Creutzfeldt-Jakob disease: a challenge to current diagnostic criteria. J Neuropsychiatry Clin Neurosci. 2013;25(4):335-338.
11. Gençer AG, Pelin Z, Küçükali CI., et al. Creutzfeldt-Jakob disease. Psychogeriatrics. 2011;11(2):119-124.
12. Caobelli F, Cobelli M, Pizzocaro C, et al. The role of neuroimaging in evaluating patients affected by Creutzfeldt-Jakob disease: a systematic review of the literature. J Neuroimaging. 2015;25(1):2-13.
13. Centers for Disease Control and Prevention. CDC's diagnostic criteria for Creutzfeldt-Jakob disease, 2010. http://www.cdc.gov/prions/cjd/diagnostic-criteria.html. Updated February 11, 2015. Accessed August 2, 2016.
14. Martindale JL, Geschwind MD, Miller BL. Psychiatric and neuroimaging findings in Creutzfeldt-Jakob disease. Curr Psychiatry Rep. 2003;5(1):43-46.