User login
Receptor occupancy and drug response: Understanding the relationship
Most clinicians do not think about receptor occupancy when they prescribe a medication. Most simply assume that if they use a low dose of a medication, they will get a small effect, and if they use a higher dose, they will get a larger effect. However, this is frequently not accurate. Clinicians need to understand the relationship between receptor occupancy and drug response.
In general, when an antagonist of a neurotransmitter receptor is used, it must occupy a minimum of 65% to 70% of the target receptor to be effective. This is clearly the case when the target is a postsynaptic receptor, such as the dopamine D2 receptor.1-3 Similarly, despite significant variability in antidepressant response,4 blockade of 65% to 80% of presynaptic transport proteins—such as the serotonin reuptake pumps when considering serotoninergic antidepressants,5,6 or the norepinephrine reuptake pumps when considering noradrenergic agents such as nortriptyline7—is necessary for these medications to be effective.
It is reasonable to think of the drug response of such agents as following a “threshold” model (Figure 1). This model makes 2 predictions. The first prediction is that a low dose of the drug might result in <50% receptor occupancy, but is not associated with a smaller response; it is simply ineffective. The second prediction is that a very high dose of the drug (eg, one that may exceed 90% receptor occupancy) does not result in any additional benefit, but may cause additional adverse consequences.8

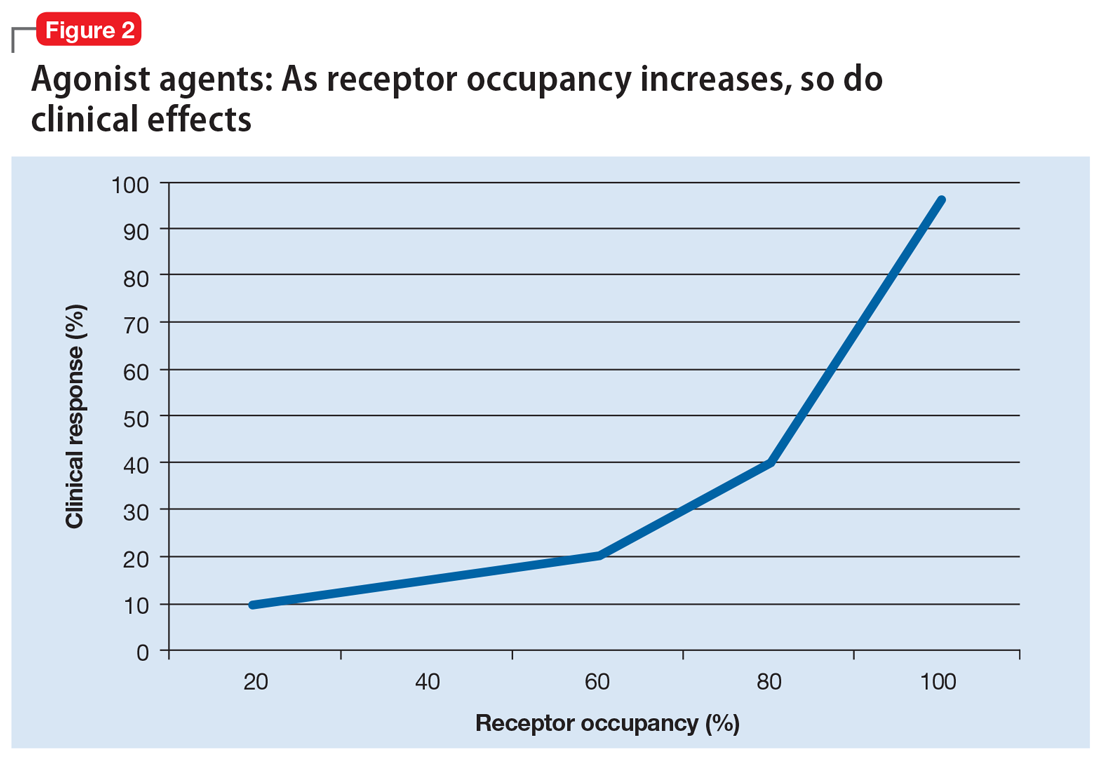
Alternatively, agonist medications, such as benzodiazepines or opiates, have their efficacy in a continuous dose-dependent fashion (Figure 2). Titrating these medications for clinical response is necessary, and minimal effective doses are highly individual. Agonist medications will not be addressed further in this article.
In this article, the term “response” is used to denote the average (population) symptom change in a study population. This term is not used as clinicians often use it to mean that their specific patient’s illness has improved, or that the patient has gone into remission. Furthermore, the information described in this article does not optimize clinical outcome, but instead is intended to help clinicians optimize the use of their pharmacologic tools.
Minimal effective dose
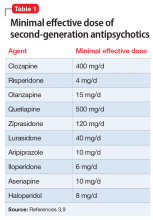
Medications that have a threshold for activity will display that clinically in a minimal effective dose (Table 13,9 and Table 25). The minimal effective dose of medications that act by blocking a neurotransmitter receptor is usually the dose that achieves 65% to 80% receptor occupancy in typical individuals (Table 25). The minimal effective doses for antipsychotics are listed in Table 1.3,9 These doses are known to occupy approximately 65% to 70% of postsynaptic D2 receptors in living humans as confirmed by positron emission tomography (PET) scans.10 Similar minimal effective doses can be determined for serotonin-reuptake inhibiting (SRI) antidepressants (Table 25). In placebo-controlled trials, doses that were smaller than the minimal effective dose did not provide any benefit.

There are important caveats to this. First is the use of partial agonists. Depending on the level of intrinsic activity of a partial agonist and clinical goal, the clinician may aim for a different level of receptor occupancy. For example, aripiprazole will act as a dopamine agonist at lower concentrations, but blocks the receptor at higher concentrations.11 Unlike antagonist antipsychotics, which require only 65% to 70% D2 receptor occupancy to be effective, aripiprazole receptor binding at effective antipsychotic doses is 90% to 95%.12-14 Since aripiprazole has an intrinsic activity of approximately 30% (ie, when it binds, it stimulates the D2 receptor to about 30% of the effect of dopamine binding to the receptor15), binding to 90% of the receptors, and displacing endogenous dopamine, allows aripiprazole to replace the background or tonic tone of dopamine, which has been measured at 19% in people with schizophrenia and 9% in controls.16 Clinically, this still appears as the minimal effective dose achieving maximal response17-19 without significant parkinsonism despite >90% receptor occupancy.12
Continue to: The second caveat is...
The second caveat is the action of low D2 receptor affinity antipsychotics, such as clozapine and quetiapine. These agents generally achieve adequate D2 receptor occupancy for only a brief period of time.20 It has been suggested that continuous receptor occupancy at ≥65% may not be necessary to obtain antipsychotic control.21,22 There may also be specific limbic and cortical (vs striatal) D2 receptor selectivity by clozapine23 compared with other second-generation antipsychotics such as risperidone and olanzapine,24,25 although this point remains debatable.26 Furthermore, the antipsychotic efficacy of low D2 receptor affinity drugs is unreliable, even in controlled, blinded studies (eg, a failed large quetiapine study27). Thus far, the actual antipsychotic mechanism of these agents is yet to be fully understood.
Minimal effective dose achieves maximal response
An interesting aspect of the threshold phenomenon of drug response is that once the minimal effective dose is reached, maximal response is achieved. In other words, there is no additional efficacy with additional dose increases. This is readily demonstrated in some studies in which patients were randomly assigned to different fixed doses or dose ranges. In these studies, there was generally no difference in response rates of different doses, so that once 65% to 80% receptor occupancy is achieved, minimal and maximal clinical response is simultaneously reached.18,28,29
For example, in the original risperidone studies, 6 mg/d was essentially equivalent to 16 mg/d.28 Similarly, lurasidone, 40 mg/d, achieves approximately 65% D2 occupancy.30 When the daily dose is increased to 120 mg, there is no additional benefit in controlling psychosis in schizophrenia.29 This pattern is also seen in partial agonists, where there are no differences between lower and higher doses in terms of response.18
Upon reading this, many clinicians may think “I don’t care what the studies say, I have seen additional benefits with additional doses.” There are several explanations for this. One is that individual patients have genetic variants that may prevent them from responding in a typical fashion. Hints of this are seen in an apparent disconnect between dosage and drug levels, so that it is not surprising that drug levels are a much better predictor of receptor occupancy than dosage.31 Nonetheless, as previously pointed out, for a population, dosage does predict receptor occupancy and outcome. However, for individuals, genetic variations make dosages less reliable. For example, ultrarapid metabolizers of cytochrome P450 (CYP) 2D6 may discontinue risperidone due to nonresponse, or require a higher dose or longer time period to respond.32,33 Similarly, patients who smoke may require an increase in doses of CYP1A2 substrates such as clozapine and olanzapine.34
Alternatively, the clinician may note improvement in mood, sleep, appetite, or other symptoms at lower doses, and then note additional improvements in psychosis or mania at higher doses.3 This occurs due to the varying affinity of different receptors. For example, in bipolar depression trials that used quetiapine in a fixed-dose design, patients who received 300 or 600 mg/d responded in the same fashion, with no additional benefit in improving depression with the higher dose.35 Similarly, in a flexible dose range study that evaluated lurasidone in bipolar depression, an average dose of 34 mg/d (range 20 to 60 mg/d) and an average dose of 83 mg/d (range 80 to 120 mg/d) both resulted in the same response (a 15.4-point reduction in depression ratings and an effect size of 0.51).36 For both quetiapine and lurasidone, higher doses are generally required to control psychosis.29,37 Note that for lurasidone, agitation, but not psychosis, improves with higher doses, which suggests that recruitment of additional receptors results in improvement in a different set of symptoms.9
Continue to: Clinical implications
Clinical implications
The implications for clinicians are relatively clear. Knowing the minimal effective doses for depression, psychosis, or mania informs the target dose. If improvement is seen at lower doses, the clinician needs to assess the profile of symptoms that improved, potential drug–drug interactions, or potential irregularities in the patient’s metabolic pathways. Clinicians need to increase doses above the minimally effective dose carefully, and expend additional effort in analyzing changes in their patient’s symptoms and adverse effects; this analysis should be performed with skepticism and willingness to reduce a dosage if no additional benefit is seen. Attention to these receptor-symptom interactions will improve response and reduce adverse consequences in the majority of patients.
Related Resource
- Lako IM, van den Heuvel ER, Knegtering H, et al. Estimating dopamine D2 receptor occupancy for doses of 8 antipsychotics: a meta-analysis. J Clin Psychopharmacol. 2013;33(5):675-681.
Drug Brand Names
Aripiprazole • Abilify
Asenapine • Saphris
Citalopram • Celexa
Clozapine • Clozaril
Fluoxetine • Prozac
Haloperidol • Haldol
Iloperidone • Fanapt
Lurasidone • Latuda
Nortriptyline • Pamelor
Olanzapine • Zyprexa
Paroxetine • Paxil
Quetiapine • Seroquel
Risperidone • Risperdal
Sertraline • Zoloft
Venlafaxine • Effexor
Ziprasidone • Geodon
1. Farde L, Nordström AL, Wiesel FA, et al. Positron emission tomographic analysis of central D1 and D2 dopamine receptor occupancy in patients treated with classical neuroleptics and clozapine. Relation to extrapyramidal side effects. Arch Gen Psychiatry. 1992;49(7):538-544.
2. Kapur S, Zipursky R, Jones C, et al. Relationship between dopamine D(2) occupancy, clinical response, and side effects: a double-blind PET study of first-episode schizophrenia. Am J Psychiatry. 2000;157(4):514-520.
3. Roberts RJ, Lohano KK, El-Mallakh RS. Antipsychotics as antidepressants. Asia Pacific Psychiatry. 2016;8(3):179-188.
4. Quitkin FM, Rabkin JG, Gerald J, et al. Validity of clinical trials of antidepressants. Am J Psychiatry. 2000;157(3):327-337.
5. Meyer JH, Wilson AA, Sagrati S, et al. Serotonin transporter occupancy of five selective serotonin reuptake inhibitors at different doses: an [11C]DASB positron emission tomography study. Am J Psychiatry. 2004;161(5):826-835.
6. Lundberg J, Tiger M, Landén M, et al. Serotonin transporter occupancy with TCAs and SSRIs: a PET study in patients with major depressive disorder. Int J Neuropsychopharmacol. 2012;15(8):1167-1172.
7. Takano H, Arakawa R, Nogami T, et al. Norepinephrine transporter occupancy by nortriptyline in patients with depression: a positron emission tomography study with (S,S)-[¹8F]FMeNER-D2. Int J Neuropsychopharmacol. 2014;17(4):553-560.
8. Johnson M, Kozielska M, Pilla Reddy V, et al. Dopamine D2 receptor occupancy as a predictor of catalepsy in rats: a pharmacokinetic-pharmacodynamic modeling approach. Pharm Res. 2014;31(10):2605-2617.
9. Allen MH, Citrome L, Pikalov A, et al. Efficacy of lurasidone in the treatment of agitation: a post hoc analysis of five short-term studies in acutely ill patients with schizophrenia. Gen Hosp Psychiatry. 2017;47:75-82.
10. Sekine M, Maeda J, Shimada H, et al. Central nervous system drug evaluation using positron emission tomography. Clin Psychopharmacol Neurosci. 2011;9(1):9-16.
11. Ma GF, Raivio N, Sabrià J, et al. Agonist and antagonist effects of aripiprazole on D2-like receptors controlling rat brain dopamine synthesis depend on the dopaminergic tone. Int J Neuropsychopharmacol. 2014;18(4):pii: pyu046. doi: 10.1093/ijnp/pyu046.
12. Yokoi F, Gründer G, Biziere K, et al. Dopamine D2 and D3 receptor occupancy in normal humans treated with the antipsychotic drug aripiprazole (OPC 14597): a study using positron emission tomography and [11C]raclopride. Neuropsychopharmacology. 2002;27(2):248-259.
13. Gründer G, Carlsson A, Wong DF. Mechanism of new antipsychotic medications: occupancy is not just antagonism. Arch Gen Psychiatry. 2003;60(10):974-977.
14. Mamo D, Graff A, Mizrahi R, et al. Differential effects of aripiprazole on D(2), 5-HT(2), and 5-HT(1A)receptor occupancy in patients with schizophrenia: a triple tracer PET study. Am J Psychiatry. 2007;164(9):1411-1417.
15. Weiden PJ, Preskorn SH, Fahnestock PA, et al. Translating the psychopharmacology of antipsychotics to individualized treatment for severe mental illness: a roadmap. J Clin Psychiatry. 2007;68(suppl 7):1-48.
16. Abi-Dargham A, Rodenhiser J, Printz D, et al. Increased baseline occupancy of D2 receptors by dopamine in schizophrenia. Proc Natl Acad Sci U S A. 2000;97(14):8104-8109.
17. Kane JM, Carson WH, Saha AR, et al. Efficacy and safety of aripiprazole and haloperidol versus placebo in patients with schizophrenia and schizoaffective disorder. J Clin Psychiatry. 2002;63(9):763-771.
18. Potkin SG, Saha AR, Kujawa MJ, et al. Aripiprazole, an antipsychotic with a novel mechanism of action, and risperidone vs placebo in patients with schizophrenia and schizoaffective disorder. Arch Gen Psychiatry. 2003;60(7):681-690.
19. Cutler AJ, Marcus RN, Hardy SA, et al. The efficacy and safety of lower doses of aripiprazole for the treatment of patients with acute exacerbation of schizophrenia. CNS Spectr. 2006;11(9):691-702; quiz 719.
20. Gründer G, Landvogt C, Vernaleken I, et al. The striatal and extrastriatal D2/D3 receptor-binding profile of clozapine in patients with schizophrenia. Neuropsychopharmacology. 2006;31(5):1027-1035.
21. Mizuno Y, Bies RR, Remington G, et al. Dopamine D2 receptor occupancy with risperidone or olanzapine during maintenance treatment of schizophrenia: a cross-sectional study. Prog Neuropsychopharmacol Biol Psychiatry. 2012;37(1):182-187.
22. Moriguchi S, Bies RR, Remington G, et al. Estimated dopamine D2 receptor occupancy and remission in schizophrenia: analysis of the CATIE data. J Clin Psychopharmacol. 2013;33(5):682-685.
23. Pilowsky LS, Mulligan RS, Acton PD, et al. Limbic selectivity of clozapine. Lancet. 1997;350(9076):490-491.
24. Ito H, Arakawa R, Takahashi H, et al. No regional difference in dopamine D2 receptor occupancy by the second-generation antipsychotic drug risperidone in humans: a positron emission tomography study. Int J Neuropsychopharmacol. 2009;12(5):667-675.
25. Arakawa R, Ito H, Okumura M, et al. Extrastriatal dopamine D(2) receptor occupancy in olanzapine-treated patients with schizophrenia. Eur Arch Psychiatry Clin Neurosci. 2010;260(4):345-350.
26. Xiberas X, Martinot JL, Mallet L, et al. Extrastriatal and striatal D(2) dopamine receptor blockade with haloperidol or new antipsychotic drugs in patients with schizophrenia. Br J Psychiatry. 2001;179:503-508.
27. Cutler AJ, Tran-Johnson T, Kalali A, et al. A failed 6-week, randomized, double-blind, placebo-controlled study of once-daily extended release quetiapine fumarate in patients with acute schizophrenia: lessons learned. Psychopharmacol Bull. 2010;43(4):37-69.
28. Marder SR, Meibach RC. Risperidone in the treatment of schizophrenia. Am J Psychiatry. 1994;151(6):825-835.
29. Meltzer HY, Cucchiaro J, Silva R, et al. Lurasidone in the treatment of schizophrenia: a randomized, double-blind, placebo- and olanzapine-controlled study. Am J Psychiatry. 2011;168(9):957-967.
30. Wong DF, Kuwabara H, Brašic JR, et al. Determination of dopamine D2 receptor occupancy by lurasidone using positron emission tomography in healthy male subjects. Psychopharmacology (Berl). 2013;229(2):245-252.
31. Potkin SG, Keator DB, Kesler-West ML, et al. D2 receptor occupancy following lurasidone treatment in patients with schizophrenia or schizoaffective disorder. CNS Spectr. 2014;19(2):176-181.
32. de Leon J, Susce MT, Pan RM, et al. The CYP2D6 poor metabolizer phenotype may be associated with risperidone adverse drug reactions and discontinuation. J Clin Psychiatry. 2005;66(1):15-27.
33. de Leon J, Susce MT, Pan RM, et al. A study of genetic (CYP2D6 and ABCB1) and environmental (drug inhibitors and inducers) variables that may influence plasma risperidone levels. Pharmacopsychiatry. 2007;40(3):93-102.
34. Narahari A, El-Mallakh RS, Kolikonda MK, et al. How coffee and cigarettes can affect the response to psychopharmacotherapy. Current Psychiatry. 2015;14(10):79-80.
35. Calabrese JR, Keck PE Jr, Macfadden W, et al. A randomized, double-blind, placebo-controlled trial of quetiapine in the treatment of bipolar I or II depression. Am J Psychiatry. 2005;162(7):1351-1360.
36. Loebel A, Cucchiaro J, Silva R, et al. Lurasidone monotherapy in the treatment of bipolar I depression: a randomized, double-blind, placebo-controlled study. Am J Psychiatry. 2014;171(2):160-168.
37. Lindenmayer JP, Brown D, Liu S, et al. The efficacy and tolerability of once-daily extended release quetiapine fumarate in hospitalized patients with acute schizophrenia: a 6-week randomized, double-blind, placebo-controlled study. Psychopharmacol Bull. 2008;41(3):11-35.
Most clinicians do not think about receptor occupancy when they prescribe a medication. Most simply assume that if they use a low dose of a medication, they will get a small effect, and if they use a higher dose, they will get a larger effect. However, this is frequently not accurate. Clinicians need to understand the relationship between receptor occupancy and drug response.
In general, when an antagonist of a neurotransmitter receptor is used, it must occupy a minimum of 65% to 70% of the target receptor to be effective. This is clearly the case when the target is a postsynaptic receptor, such as the dopamine D2 receptor.1-3 Similarly, despite significant variability in antidepressant response,4 blockade of 65% to 80% of presynaptic transport proteins—such as the serotonin reuptake pumps when considering serotoninergic antidepressants,5,6 or the norepinephrine reuptake pumps when considering noradrenergic agents such as nortriptyline7—is necessary for these medications to be effective.
It is reasonable to think of the drug response of such agents as following a “threshold” model (Figure 1). This model makes 2 predictions. The first prediction is that a low dose of the drug might result in <50% receptor occupancy, but is not associated with a smaller response; it is simply ineffective. The second prediction is that a very high dose of the drug (eg, one that may exceed 90% receptor occupancy) does not result in any additional benefit, but may cause additional adverse consequences.8

Alternatively, agonist medications, such as benzodiazepines or opiates, have their efficacy in a continuous dose-dependent fashion (Figure 2). Titrating these medications for clinical response is necessary, and minimal effective doses are highly individual. Agonist medications will not be addressed further in this article.

In this article, the term “response” is used to denote the average (population) symptom change in a study population. This term is not used as clinicians often use it to mean that their specific patient’s illness has improved, or that the patient has gone into remission. Furthermore, the information described in this article does not optimize clinical outcome, but instead is intended to help clinicians optimize the use of their pharmacologic tools.
Minimal effective dose
Medications that have a threshold for activity will display that clinically in a minimal effective dose (Table 13,9 and Table 25). The minimal effective dose of medications that act by blocking a neurotransmitter receptor is usually the dose that achieves 65% to 80% receptor occupancy in typical individuals (Table 25). The minimal effective doses for antipsychotics are listed in Table 1.3,9 These doses are known to occupy approximately 65% to 70% of postsynaptic D2 receptors in living humans as confirmed by positron emission tomography (PET) scans.10 Similar minimal effective doses can be determined for serotonin-reuptake inhibiting (SRI) antidepressants (Table 25). In placebo-controlled trials, doses that were smaller than the minimal effective dose did not provide any benefit.

There are important caveats to this. First is the use of partial agonists. Depending on the level of intrinsic activity of a partial agonist and clinical goal, the clinician may aim for a different level of receptor occupancy. For example, aripiprazole will act as a dopamine agonist at lower concentrations, but blocks the receptor at higher concentrations.11 Unlike antagonist antipsychotics, which require only 65% to 70% D2 receptor occupancy to be effective, aripiprazole receptor binding at effective antipsychotic doses is 90% to 95%.12-14 Since aripiprazole has an intrinsic activity of approximately 30% (ie, when it binds, it stimulates the D2 receptor to about 30% of the effect of dopamine binding to the receptor15), binding to 90% of the receptors, and displacing endogenous dopamine, allows aripiprazole to replace the background or tonic tone of dopamine, which has been measured at 19% in people with schizophrenia and 9% in controls.16 Clinically, this still appears as the minimal effective dose achieving maximal response17-19 without significant parkinsonism despite >90% receptor occupancy.12
Continue to: The second caveat is...
The second caveat is the action of low D2 receptor affinity antipsychotics, such as clozapine and quetiapine. These agents generally achieve adequate D2 receptor occupancy for only a brief period of time.20 It has been suggested that continuous receptor occupancy at ≥65% may not be necessary to obtain antipsychotic control.21,22 There may also be specific limbic and cortical (vs striatal) D2 receptor selectivity by clozapine23 compared with other second-generation antipsychotics such as risperidone and olanzapine,24,25 although this point remains debatable.26 Furthermore, the antipsychotic efficacy of low D2 receptor affinity drugs is unreliable, even in controlled, blinded studies (eg, a failed large quetiapine study27). Thus far, the actual antipsychotic mechanism of these agents is yet to be fully understood.
Minimal effective dose achieves maximal response
An interesting aspect of the threshold phenomenon of drug response is that once the minimal effective dose is reached, maximal response is achieved. In other words, there is no additional efficacy with additional dose increases. This is readily demonstrated in some studies in which patients were randomly assigned to different fixed doses or dose ranges. In these studies, there was generally no difference in response rates of different doses, so that once 65% to 80% receptor occupancy is achieved, minimal and maximal clinical response is simultaneously reached.18,28,29
For example, in the original risperidone studies, 6 mg/d was essentially equivalent to 16 mg/d.28 Similarly, lurasidone, 40 mg/d, achieves approximately 65% D2 occupancy.30 When the daily dose is increased to 120 mg, there is no additional benefit in controlling psychosis in schizophrenia.29 This pattern is also seen in partial agonists, where there are no differences between lower and higher doses in terms of response.18
Upon reading this, many clinicians may think “I don’t care what the studies say, I have seen additional benefits with additional doses.” There are several explanations for this. One is that individual patients have genetic variants that may prevent them from responding in a typical fashion. Hints of this are seen in an apparent disconnect between dosage and drug levels, so that it is not surprising that drug levels are a much better predictor of receptor occupancy than dosage.31 Nonetheless, as previously pointed out, for a population, dosage does predict receptor occupancy and outcome. However, for individuals, genetic variations make dosages less reliable. For example, ultrarapid metabolizers of cytochrome P450 (CYP) 2D6 may discontinue risperidone due to nonresponse, or require a higher dose or longer time period to respond.32,33 Similarly, patients who smoke may require an increase in doses of CYP1A2 substrates such as clozapine and olanzapine.34
Alternatively, the clinician may note improvement in mood, sleep, appetite, or other symptoms at lower doses, and then note additional improvements in psychosis or mania at higher doses.3 This occurs due to the varying affinity of different receptors. For example, in bipolar depression trials that used quetiapine in a fixed-dose design, patients who received 300 or 600 mg/d responded in the same fashion, with no additional benefit in improving depression with the higher dose.35 Similarly, in a flexible dose range study that evaluated lurasidone in bipolar depression, an average dose of 34 mg/d (range 20 to 60 mg/d) and an average dose of 83 mg/d (range 80 to 120 mg/d) both resulted in the same response (a 15.4-point reduction in depression ratings and an effect size of 0.51).36 For both quetiapine and lurasidone, higher doses are generally required to control psychosis.29,37 Note that for lurasidone, agitation, but not psychosis, improves with higher doses, which suggests that recruitment of additional receptors results in improvement in a different set of symptoms.9
Continue to: Clinical implications
Clinical implications
The implications for clinicians are relatively clear. Knowing the minimal effective doses for depression, psychosis, or mania informs the target dose. If improvement is seen at lower doses, the clinician needs to assess the profile of symptoms that improved, potential drug–drug interactions, or potential irregularities in the patient’s metabolic pathways. Clinicians need to increase doses above the minimally effective dose carefully, and expend additional effort in analyzing changes in their patient’s symptoms and adverse effects; this analysis should be performed with skepticism and willingness to reduce a dosage if no additional benefit is seen. Attention to these receptor-symptom interactions will improve response and reduce adverse consequences in the majority of patients.
Related Resource
- Lako IM, van den Heuvel ER, Knegtering H, et al. Estimating dopamine D2 receptor occupancy for doses of 8 antipsychotics: a meta-analysis. J Clin Psychopharmacol. 2013;33(5):675-681.
Drug Brand Names
Aripiprazole • Abilify
Asenapine • Saphris
Citalopram • Celexa
Clozapine • Clozaril
Fluoxetine • Prozac
Haloperidol • Haldol
Iloperidone • Fanapt
Lurasidone • Latuda
Nortriptyline • Pamelor
Olanzapine • Zyprexa
Paroxetine • Paxil
Quetiapine • Seroquel
Risperidone • Risperdal
Sertraline • Zoloft
Venlafaxine • Effexor
Ziprasidone • Geodon
Most clinicians do not think about receptor occupancy when they prescribe a medication. Most simply assume that if they use a low dose of a medication, they will get a small effect, and if they use a higher dose, they will get a larger effect. However, this is frequently not accurate. Clinicians need to understand the relationship between receptor occupancy and drug response.
In general, when an antagonist of a neurotransmitter receptor is used, it must occupy a minimum of 65% to 70% of the target receptor to be effective. This is clearly the case when the target is a postsynaptic receptor, such as the dopamine D2 receptor.1-3 Similarly, despite significant variability in antidepressant response,4 blockade of 65% to 80% of presynaptic transport proteins—such as the serotonin reuptake pumps when considering serotoninergic antidepressants,5,6 or the norepinephrine reuptake pumps when considering noradrenergic agents such as nortriptyline7—is necessary for these medications to be effective.
It is reasonable to think of the drug response of such agents as following a “threshold” model (Figure 1). This model makes 2 predictions. The first prediction is that a low dose of the drug might result in <50% receptor occupancy, but is not associated with a smaller response; it is simply ineffective. The second prediction is that a very high dose of the drug (eg, one that may exceed 90% receptor occupancy) does not result in any additional benefit, but may cause additional adverse consequences.8

Alternatively, agonist medications, such as benzodiazepines or opiates, have their efficacy in a continuous dose-dependent fashion (Figure 2). Titrating these medications for clinical response is necessary, and minimal effective doses are highly individual. Agonist medications will not be addressed further in this article.

In this article, the term “response” is used to denote the average (population) symptom change in a study population. This term is not used as clinicians often use it to mean that their specific patient’s illness has improved, or that the patient has gone into remission. Furthermore, the information described in this article does not optimize clinical outcome, but instead is intended to help clinicians optimize the use of their pharmacologic tools.
Minimal effective dose
Medications that have a threshold for activity will display that clinically in a minimal effective dose (Table 13,9 and Table 25). The minimal effective dose of medications that act by blocking a neurotransmitter receptor is usually the dose that achieves 65% to 80% receptor occupancy in typical individuals (Table 25). The minimal effective doses for antipsychotics are listed in Table 1.3,9 These doses are known to occupy approximately 65% to 70% of postsynaptic D2 receptors in living humans as confirmed by positron emission tomography (PET) scans.10 Similar minimal effective doses can be determined for serotonin-reuptake inhibiting (SRI) antidepressants (Table 25). In placebo-controlled trials, doses that were smaller than the minimal effective dose did not provide any benefit.

There are important caveats to this. First is the use of partial agonists. Depending on the level of intrinsic activity of a partial agonist and clinical goal, the clinician may aim for a different level of receptor occupancy. For example, aripiprazole will act as a dopamine agonist at lower concentrations, but blocks the receptor at higher concentrations.11 Unlike antagonist antipsychotics, which require only 65% to 70% D2 receptor occupancy to be effective, aripiprazole receptor binding at effective antipsychotic doses is 90% to 95%.12-14 Since aripiprazole has an intrinsic activity of approximately 30% (ie, when it binds, it stimulates the D2 receptor to about 30% of the effect of dopamine binding to the receptor15), binding to 90% of the receptors, and displacing endogenous dopamine, allows aripiprazole to replace the background or tonic tone of dopamine, which has been measured at 19% in people with schizophrenia and 9% in controls.16 Clinically, this still appears as the minimal effective dose achieving maximal response17-19 without significant parkinsonism despite >90% receptor occupancy.12
Continue to: The second caveat is...
The second caveat is the action of low D2 receptor affinity antipsychotics, such as clozapine and quetiapine. These agents generally achieve adequate D2 receptor occupancy for only a brief period of time.20 It has been suggested that continuous receptor occupancy at ≥65% may not be necessary to obtain antipsychotic control.21,22 There may also be specific limbic and cortical (vs striatal) D2 receptor selectivity by clozapine23 compared with other second-generation antipsychotics such as risperidone and olanzapine,24,25 although this point remains debatable.26 Furthermore, the antipsychotic efficacy of low D2 receptor affinity drugs is unreliable, even in controlled, blinded studies (eg, a failed large quetiapine study27). Thus far, the actual antipsychotic mechanism of these agents is yet to be fully understood.
Minimal effective dose achieves maximal response
An interesting aspect of the threshold phenomenon of drug response is that once the minimal effective dose is reached, maximal response is achieved. In other words, there is no additional efficacy with additional dose increases. This is readily demonstrated in some studies in which patients were randomly assigned to different fixed doses or dose ranges. In these studies, there was generally no difference in response rates of different doses, so that once 65% to 80% receptor occupancy is achieved, minimal and maximal clinical response is simultaneously reached.18,28,29
For example, in the original risperidone studies, 6 mg/d was essentially equivalent to 16 mg/d.28 Similarly, lurasidone, 40 mg/d, achieves approximately 65% D2 occupancy.30 When the daily dose is increased to 120 mg, there is no additional benefit in controlling psychosis in schizophrenia.29 This pattern is also seen in partial agonists, where there are no differences between lower and higher doses in terms of response.18
Upon reading this, many clinicians may think “I don’t care what the studies say, I have seen additional benefits with additional doses.” There are several explanations for this. One is that individual patients have genetic variants that may prevent them from responding in a typical fashion. Hints of this are seen in an apparent disconnect between dosage and drug levels, so that it is not surprising that drug levels are a much better predictor of receptor occupancy than dosage.31 Nonetheless, as previously pointed out, for a population, dosage does predict receptor occupancy and outcome. However, for individuals, genetic variations make dosages less reliable. For example, ultrarapid metabolizers of cytochrome P450 (CYP) 2D6 may discontinue risperidone due to nonresponse, or require a higher dose or longer time period to respond.32,33 Similarly, patients who smoke may require an increase in doses of CYP1A2 substrates such as clozapine and olanzapine.34
Alternatively, the clinician may note improvement in mood, sleep, appetite, or other symptoms at lower doses, and then note additional improvements in psychosis or mania at higher doses.3 This occurs due to the varying affinity of different receptors. For example, in bipolar depression trials that used quetiapine in a fixed-dose design, patients who received 300 or 600 mg/d responded in the same fashion, with no additional benefit in improving depression with the higher dose.35 Similarly, in a flexible dose range study that evaluated lurasidone in bipolar depression, an average dose of 34 mg/d (range 20 to 60 mg/d) and an average dose of 83 mg/d (range 80 to 120 mg/d) both resulted in the same response (a 15.4-point reduction in depression ratings and an effect size of 0.51).36 For both quetiapine and lurasidone, higher doses are generally required to control psychosis.29,37 Note that for lurasidone, agitation, but not psychosis, improves with higher doses, which suggests that recruitment of additional receptors results in improvement in a different set of symptoms.9
Continue to: Clinical implications
Clinical implications
The implications for clinicians are relatively clear. Knowing the minimal effective doses for depression, psychosis, or mania informs the target dose. If improvement is seen at lower doses, the clinician needs to assess the profile of symptoms that improved, potential drug–drug interactions, or potential irregularities in the patient’s metabolic pathways. Clinicians need to increase doses above the minimally effective dose carefully, and expend additional effort in analyzing changes in their patient’s symptoms and adverse effects; this analysis should be performed with skepticism and willingness to reduce a dosage if no additional benefit is seen. Attention to these receptor-symptom interactions will improve response and reduce adverse consequences in the majority of patients.
Related Resource
- Lako IM, van den Heuvel ER, Knegtering H, et al. Estimating dopamine D2 receptor occupancy for doses of 8 antipsychotics: a meta-analysis. J Clin Psychopharmacol. 2013;33(5):675-681.
Drug Brand Names
Aripiprazole • Abilify
Asenapine • Saphris
Citalopram • Celexa
Clozapine • Clozaril
Fluoxetine • Prozac
Haloperidol • Haldol
Iloperidone • Fanapt
Lurasidone • Latuda
Nortriptyline • Pamelor
Olanzapine • Zyprexa
Paroxetine • Paxil
Quetiapine • Seroquel
Risperidone • Risperdal
Sertraline • Zoloft
Venlafaxine • Effexor
Ziprasidone • Geodon
1. Farde L, Nordström AL, Wiesel FA, et al. Positron emission tomographic analysis of central D1 and D2 dopamine receptor occupancy in patients treated with classical neuroleptics and clozapine. Relation to extrapyramidal side effects. Arch Gen Psychiatry. 1992;49(7):538-544.
2. Kapur S, Zipursky R, Jones C, et al. Relationship between dopamine D(2) occupancy, clinical response, and side effects: a double-blind PET study of first-episode schizophrenia. Am J Psychiatry. 2000;157(4):514-520.
3. Roberts RJ, Lohano KK, El-Mallakh RS. Antipsychotics as antidepressants. Asia Pacific Psychiatry. 2016;8(3):179-188.
4. Quitkin FM, Rabkin JG, Gerald J, et al. Validity of clinical trials of antidepressants. Am J Psychiatry. 2000;157(3):327-337.
5. Meyer JH, Wilson AA, Sagrati S, et al. Serotonin transporter occupancy of five selective serotonin reuptake inhibitors at different doses: an [11C]DASB positron emission tomography study. Am J Psychiatry. 2004;161(5):826-835.
6. Lundberg J, Tiger M, Landén M, et al. Serotonin transporter occupancy with TCAs and SSRIs: a PET study in patients with major depressive disorder. Int J Neuropsychopharmacol. 2012;15(8):1167-1172.
7. Takano H, Arakawa R, Nogami T, et al. Norepinephrine transporter occupancy by nortriptyline in patients with depression: a positron emission tomography study with (S,S)-[¹8F]FMeNER-D2. Int J Neuropsychopharmacol. 2014;17(4):553-560.
8. Johnson M, Kozielska M, Pilla Reddy V, et al. Dopamine D2 receptor occupancy as a predictor of catalepsy in rats: a pharmacokinetic-pharmacodynamic modeling approach. Pharm Res. 2014;31(10):2605-2617.
9. Allen MH, Citrome L, Pikalov A, et al. Efficacy of lurasidone in the treatment of agitation: a post hoc analysis of five short-term studies in acutely ill patients with schizophrenia. Gen Hosp Psychiatry. 2017;47:75-82.
10. Sekine M, Maeda J, Shimada H, et al. Central nervous system drug evaluation using positron emission tomography. Clin Psychopharmacol Neurosci. 2011;9(1):9-16.
11. Ma GF, Raivio N, Sabrià J, et al. Agonist and antagonist effects of aripiprazole on D2-like receptors controlling rat brain dopamine synthesis depend on the dopaminergic tone. Int J Neuropsychopharmacol. 2014;18(4):pii: pyu046. doi: 10.1093/ijnp/pyu046.
12. Yokoi F, Gründer G, Biziere K, et al. Dopamine D2 and D3 receptor occupancy in normal humans treated with the antipsychotic drug aripiprazole (OPC 14597): a study using positron emission tomography and [11C]raclopride. Neuropsychopharmacology. 2002;27(2):248-259.
13. Gründer G, Carlsson A, Wong DF. Mechanism of new antipsychotic medications: occupancy is not just antagonism. Arch Gen Psychiatry. 2003;60(10):974-977.
14. Mamo D, Graff A, Mizrahi R, et al. Differential effects of aripiprazole on D(2), 5-HT(2), and 5-HT(1A)receptor occupancy in patients with schizophrenia: a triple tracer PET study. Am J Psychiatry. 2007;164(9):1411-1417.
15. Weiden PJ, Preskorn SH, Fahnestock PA, et al. Translating the psychopharmacology of antipsychotics to individualized treatment for severe mental illness: a roadmap. J Clin Psychiatry. 2007;68(suppl 7):1-48.
16. Abi-Dargham A, Rodenhiser J, Printz D, et al. Increased baseline occupancy of D2 receptors by dopamine in schizophrenia. Proc Natl Acad Sci U S A. 2000;97(14):8104-8109.
17. Kane JM, Carson WH, Saha AR, et al. Efficacy and safety of aripiprazole and haloperidol versus placebo in patients with schizophrenia and schizoaffective disorder. J Clin Psychiatry. 2002;63(9):763-771.
18. Potkin SG, Saha AR, Kujawa MJ, et al. Aripiprazole, an antipsychotic with a novel mechanism of action, and risperidone vs placebo in patients with schizophrenia and schizoaffective disorder. Arch Gen Psychiatry. 2003;60(7):681-690.
19. Cutler AJ, Marcus RN, Hardy SA, et al. The efficacy and safety of lower doses of aripiprazole for the treatment of patients with acute exacerbation of schizophrenia. CNS Spectr. 2006;11(9):691-702; quiz 719.
20. Gründer G, Landvogt C, Vernaleken I, et al. The striatal and extrastriatal D2/D3 receptor-binding profile of clozapine in patients with schizophrenia. Neuropsychopharmacology. 2006;31(5):1027-1035.
21. Mizuno Y, Bies RR, Remington G, et al. Dopamine D2 receptor occupancy with risperidone or olanzapine during maintenance treatment of schizophrenia: a cross-sectional study. Prog Neuropsychopharmacol Biol Psychiatry. 2012;37(1):182-187.
22. Moriguchi S, Bies RR, Remington G, et al. Estimated dopamine D2 receptor occupancy and remission in schizophrenia: analysis of the CATIE data. J Clin Psychopharmacol. 2013;33(5):682-685.
23. Pilowsky LS, Mulligan RS, Acton PD, et al. Limbic selectivity of clozapine. Lancet. 1997;350(9076):490-491.
24. Ito H, Arakawa R, Takahashi H, et al. No regional difference in dopamine D2 receptor occupancy by the second-generation antipsychotic drug risperidone in humans: a positron emission tomography study. Int J Neuropsychopharmacol. 2009;12(5):667-675.
25. Arakawa R, Ito H, Okumura M, et al. Extrastriatal dopamine D(2) receptor occupancy in olanzapine-treated patients with schizophrenia. Eur Arch Psychiatry Clin Neurosci. 2010;260(4):345-350.
26. Xiberas X, Martinot JL, Mallet L, et al. Extrastriatal and striatal D(2) dopamine receptor blockade with haloperidol or new antipsychotic drugs in patients with schizophrenia. Br J Psychiatry. 2001;179:503-508.
27. Cutler AJ, Tran-Johnson T, Kalali A, et al. A failed 6-week, randomized, double-blind, placebo-controlled study of once-daily extended release quetiapine fumarate in patients with acute schizophrenia: lessons learned. Psychopharmacol Bull. 2010;43(4):37-69.
28. Marder SR, Meibach RC. Risperidone in the treatment of schizophrenia. Am J Psychiatry. 1994;151(6):825-835.
29. Meltzer HY, Cucchiaro J, Silva R, et al. Lurasidone in the treatment of schizophrenia: a randomized, double-blind, placebo- and olanzapine-controlled study. Am J Psychiatry. 2011;168(9):957-967.
30. Wong DF, Kuwabara H, Brašic JR, et al. Determination of dopamine D2 receptor occupancy by lurasidone using positron emission tomography in healthy male subjects. Psychopharmacology (Berl). 2013;229(2):245-252.
31. Potkin SG, Keator DB, Kesler-West ML, et al. D2 receptor occupancy following lurasidone treatment in patients with schizophrenia or schizoaffective disorder. CNS Spectr. 2014;19(2):176-181.
32. de Leon J, Susce MT, Pan RM, et al. The CYP2D6 poor metabolizer phenotype may be associated with risperidone adverse drug reactions and discontinuation. J Clin Psychiatry. 2005;66(1):15-27.
33. de Leon J, Susce MT, Pan RM, et al. A study of genetic (CYP2D6 and ABCB1) and environmental (drug inhibitors and inducers) variables that may influence plasma risperidone levels. Pharmacopsychiatry. 2007;40(3):93-102.
34. Narahari A, El-Mallakh RS, Kolikonda MK, et al. How coffee and cigarettes can affect the response to psychopharmacotherapy. Current Psychiatry. 2015;14(10):79-80.
35. Calabrese JR, Keck PE Jr, Macfadden W, et al. A randomized, double-blind, placebo-controlled trial of quetiapine in the treatment of bipolar I or II depression. Am J Psychiatry. 2005;162(7):1351-1360.
36. Loebel A, Cucchiaro J, Silva R, et al. Lurasidone monotherapy in the treatment of bipolar I depression: a randomized, double-blind, placebo-controlled study. Am J Psychiatry. 2014;171(2):160-168.
37. Lindenmayer JP, Brown D, Liu S, et al. The efficacy and tolerability of once-daily extended release quetiapine fumarate in hospitalized patients with acute schizophrenia: a 6-week randomized, double-blind, placebo-controlled study. Psychopharmacol Bull. 2008;41(3):11-35.
1. Farde L, Nordström AL, Wiesel FA, et al. Positron emission tomographic analysis of central D1 and D2 dopamine receptor occupancy in patients treated with classical neuroleptics and clozapine. Relation to extrapyramidal side effects. Arch Gen Psychiatry. 1992;49(7):538-544.
2. Kapur S, Zipursky R, Jones C, et al. Relationship between dopamine D(2) occupancy, clinical response, and side effects: a double-blind PET study of first-episode schizophrenia. Am J Psychiatry. 2000;157(4):514-520.
3. Roberts RJ, Lohano KK, El-Mallakh RS. Antipsychotics as antidepressants. Asia Pacific Psychiatry. 2016;8(3):179-188.
4. Quitkin FM, Rabkin JG, Gerald J, et al. Validity of clinical trials of antidepressants. Am J Psychiatry. 2000;157(3):327-337.
5. Meyer JH, Wilson AA, Sagrati S, et al. Serotonin transporter occupancy of five selective serotonin reuptake inhibitors at different doses: an [11C]DASB positron emission tomography study. Am J Psychiatry. 2004;161(5):826-835.
6. Lundberg J, Tiger M, Landén M, et al. Serotonin transporter occupancy with TCAs and SSRIs: a PET study in patients with major depressive disorder. Int J Neuropsychopharmacol. 2012;15(8):1167-1172.
7. Takano H, Arakawa R, Nogami T, et al. Norepinephrine transporter occupancy by nortriptyline in patients with depression: a positron emission tomography study with (S,S)-[¹8F]FMeNER-D2. Int J Neuropsychopharmacol. 2014;17(4):553-560.
8. Johnson M, Kozielska M, Pilla Reddy V, et al. Dopamine D2 receptor occupancy as a predictor of catalepsy in rats: a pharmacokinetic-pharmacodynamic modeling approach. Pharm Res. 2014;31(10):2605-2617.
9. Allen MH, Citrome L, Pikalov A, et al. Efficacy of lurasidone in the treatment of agitation: a post hoc analysis of five short-term studies in acutely ill patients with schizophrenia. Gen Hosp Psychiatry. 2017;47:75-82.
10. Sekine M, Maeda J, Shimada H, et al. Central nervous system drug evaluation using positron emission tomography. Clin Psychopharmacol Neurosci. 2011;9(1):9-16.
11. Ma GF, Raivio N, Sabrià J, et al. Agonist and antagonist effects of aripiprazole on D2-like receptors controlling rat brain dopamine synthesis depend on the dopaminergic tone. Int J Neuropsychopharmacol. 2014;18(4):pii: pyu046. doi: 10.1093/ijnp/pyu046.
12. Yokoi F, Gründer G, Biziere K, et al. Dopamine D2 and D3 receptor occupancy in normal humans treated with the antipsychotic drug aripiprazole (OPC 14597): a study using positron emission tomography and [11C]raclopride. Neuropsychopharmacology. 2002;27(2):248-259.
13. Gründer G, Carlsson A, Wong DF. Mechanism of new antipsychotic medications: occupancy is not just antagonism. Arch Gen Psychiatry. 2003;60(10):974-977.
14. Mamo D, Graff A, Mizrahi R, et al. Differential effects of aripiprazole on D(2), 5-HT(2), and 5-HT(1A)receptor occupancy in patients with schizophrenia: a triple tracer PET study. Am J Psychiatry. 2007;164(9):1411-1417.
15. Weiden PJ, Preskorn SH, Fahnestock PA, et al. Translating the psychopharmacology of antipsychotics to individualized treatment for severe mental illness: a roadmap. J Clin Psychiatry. 2007;68(suppl 7):1-48.
16. Abi-Dargham A, Rodenhiser J, Printz D, et al. Increased baseline occupancy of D2 receptors by dopamine in schizophrenia. Proc Natl Acad Sci U S A. 2000;97(14):8104-8109.
17. Kane JM, Carson WH, Saha AR, et al. Efficacy and safety of aripiprazole and haloperidol versus placebo in patients with schizophrenia and schizoaffective disorder. J Clin Psychiatry. 2002;63(9):763-771.
18. Potkin SG, Saha AR, Kujawa MJ, et al. Aripiprazole, an antipsychotic with a novel mechanism of action, and risperidone vs placebo in patients with schizophrenia and schizoaffective disorder. Arch Gen Psychiatry. 2003;60(7):681-690.
19. Cutler AJ, Marcus RN, Hardy SA, et al. The efficacy and safety of lower doses of aripiprazole for the treatment of patients with acute exacerbation of schizophrenia. CNS Spectr. 2006;11(9):691-702; quiz 719.
20. Gründer G, Landvogt C, Vernaleken I, et al. The striatal and extrastriatal D2/D3 receptor-binding profile of clozapine in patients with schizophrenia. Neuropsychopharmacology. 2006;31(5):1027-1035.
21. Mizuno Y, Bies RR, Remington G, et al. Dopamine D2 receptor occupancy with risperidone or olanzapine during maintenance treatment of schizophrenia: a cross-sectional study. Prog Neuropsychopharmacol Biol Psychiatry. 2012;37(1):182-187.
22. Moriguchi S, Bies RR, Remington G, et al. Estimated dopamine D2 receptor occupancy and remission in schizophrenia: analysis of the CATIE data. J Clin Psychopharmacol. 2013;33(5):682-685.
23. Pilowsky LS, Mulligan RS, Acton PD, et al. Limbic selectivity of clozapine. Lancet. 1997;350(9076):490-491.
24. Ito H, Arakawa R, Takahashi H, et al. No regional difference in dopamine D2 receptor occupancy by the second-generation antipsychotic drug risperidone in humans: a positron emission tomography study. Int J Neuropsychopharmacol. 2009;12(5):667-675.
25. Arakawa R, Ito H, Okumura M, et al. Extrastriatal dopamine D(2) receptor occupancy in olanzapine-treated patients with schizophrenia. Eur Arch Psychiatry Clin Neurosci. 2010;260(4):345-350.
26. Xiberas X, Martinot JL, Mallet L, et al. Extrastriatal and striatal D(2) dopamine receptor blockade with haloperidol or new antipsychotic drugs in patients with schizophrenia. Br J Psychiatry. 2001;179:503-508.
27. Cutler AJ, Tran-Johnson T, Kalali A, et al. A failed 6-week, randomized, double-blind, placebo-controlled study of once-daily extended release quetiapine fumarate in patients with acute schizophrenia: lessons learned. Psychopharmacol Bull. 2010;43(4):37-69.
28. Marder SR, Meibach RC. Risperidone in the treatment of schizophrenia. Am J Psychiatry. 1994;151(6):825-835.
29. Meltzer HY, Cucchiaro J, Silva R, et al. Lurasidone in the treatment of schizophrenia: a randomized, double-blind, placebo- and olanzapine-controlled study. Am J Psychiatry. 2011;168(9):957-967.
30. Wong DF, Kuwabara H, Brašic JR, et al. Determination of dopamine D2 receptor occupancy by lurasidone using positron emission tomography in healthy male subjects. Psychopharmacology (Berl). 2013;229(2):245-252.
31. Potkin SG, Keator DB, Kesler-West ML, et al. D2 receptor occupancy following lurasidone treatment in patients with schizophrenia or schizoaffective disorder. CNS Spectr. 2014;19(2):176-181.
32. de Leon J, Susce MT, Pan RM, et al. The CYP2D6 poor metabolizer phenotype may be associated with risperidone adverse drug reactions and discontinuation. J Clin Psychiatry. 2005;66(1):15-27.
33. de Leon J, Susce MT, Pan RM, et al. A study of genetic (CYP2D6 and ABCB1) and environmental (drug inhibitors and inducers) variables that may influence plasma risperidone levels. Pharmacopsychiatry. 2007;40(3):93-102.
34. Narahari A, El-Mallakh RS, Kolikonda MK, et al. How coffee and cigarettes can affect the response to psychopharmacotherapy. Current Psychiatry. 2015;14(10):79-80.
35. Calabrese JR, Keck PE Jr, Macfadden W, et al. A randomized, double-blind, placebo-controlled trial of quetiapine in the treatment of bipolar I or II depression. Am J Psychiatry. 2005;162(7):1351-1360.
36. Loebel A, Cucchiaro J, Silva R, et al. Lurasidone monotherapy in the treatment of bipolar I depression: a randomized, double-blind, placebo-controlled study. Am J Psychiatry. 2014;171(2):160-168.
37. Lindenmayer JP, Brown D, Liu S, et al. The efficacy and tolerability of once-daily extended release quetiapine fumarate in hospitalized patients with acute schizophrenia: a 6-week randomized, double-blind, placebo-controlled study. Psychopharmacol Bull. 2008;41(3):11-35.
It takes guts to be mentally ill: Microbiota and psychopathology
What is the largest endocrine organ in the human body?
Here is a clue: It is also the largest immune organ in humans!
Still scratching your head? Here is another clue: This organ also contains a “second brain,” which is connected to big brain inside the head by the vagus nerve.
Okay, enough guessing: It’s the 30-foot long gastrointestinal (GI) tract, which is generally associated only with eating and digestion. But it is far more than a digestive tract. It is home to about 100 trillion diverse bacteria, including 1,000 known species, which together are known as “microbiota.” Its combined DNA is called the “microbiome” and is 10,000% larger than the human genome. Those trillions of bacteria in our guts are a symbiotic (commensal) organ that is vital for the normal functions of the human body.1
While this vast array of microorganisms is vital to sustaining a healthy human existence, it can also be involved in multiple psychiatric disorders, including depression, psychosis, anxiety, autism, and attention-deficit/hyperactivity disorder (ADHD). Humans acquire their unique sets of microbiota as they pass through the mother’s vagina at birth and while breastfeeding, as well as from exposure to various environmental sources in the first few months of life.2
The microbiota in the GI tract are an intimate neighbor of the “enteric brain,” comprised of 100 million neurons plus glia-like support cell structures. This “second brain” produces over 30 neurotransmitters, several of which (dopamine, serotonin, γ-aminobutyric acid [GABA], acetylcholine) have been implicated in major psychiatric disorders.3
The brain and gut have a dynamic bidirectional communication system, mediated by neural, hormonal, and immunological crosstalk and influences. The GI tract secretes dozens of peptides and other signaling molecules that influence the brain. The microbiota also interact with and are regulated by gut hormones such as oxytocin, ghrelin, neuropeptide Y, cholecystokinin, corticotrophin-releasing factor, and pancreatic polypeptide.4 The microbiota modulate brain development, functions, and behavior, and maintain the intestinal barrier, which, if disrupted, would result in the gut becoming “leaky” and triggering low-grade inflammation such as that associated with depression.5
Continue to: But don't overlook the importance of...
But don’t overlook the importance of diet. It is a major factor in shaping the composition of the microbiota. What we eat can have a preventative or reparative effect on neuroimmune or neuroinflammatory disease. An emerging body of evidence suggests that the diet and its effects on the gut microbiota can modify a person’s genes by epigenetic mechanisms (altering DNA methylation and histone effects). Probiotics can exert epigenetic effects by influencing cytokines, by producing short-chain fatty acids (SCFAs), by vitamin synthesis, and by producing several well-known neurotransmitters.6
The bidirectional trafficking across the microbiome-gut-brain axis includes reciprocal effects. The brain influences the microbiome composition by regulating satiety, the hypothalamic-pituitary axis, and with neuropeptides.7 In return, the microbiome conveys information to the brain about the intestinal status via infectious agents, intestinal neurotransmitters and modulators, cytokines, sensory vagal fibers, and various metabolites. Failure of these normal interactions can lead to a variety of pathological processes, including inflammatory, autoimmune, degenerative, metabolic, cognitive, mood, and behavioral adverse effects. Therapeutic interventions for these adverse consequences can be implemented through microbiome manipulations (such as fecal transplants), nutritional strategies, and reinforcement of the enteric and brain barriers.
Alterations in the microbiota, such as by the intake of antibiotics or by intestinal inflammation, can lead to psychiatric disorders.8 The following findings link gut microbiome disruptions with several psychiatric disorders:
Schizophrenia prodrome. Fecal bacteria show an increase in SCFAs, which can activate microglia (the initial step in triggering psychosis).9 These bacteria have been shown to lead to an increase in choline levels in the anterior cingulate, a known biomarker for membrane dysfunction, which is one of the biological models of schizophrenia.
Schizophrenia—first-episode. A recent study reported abnormalities in the gut microbiota of patients with first-episode psychosis, with a lower number of certain fecal bacteria (including bifidobacterium, E. coli, and lactobacillus) and high levels of Clostridium coccoides. After 6 months of risperidone treatment, the above changes were reversed.10
Continue to: Another study of fecal microbiota...
Another study of fecal microbiota in a first-episode psychosis cohort found significant differences in several bacterial strains compared with a healthy control group, and those with the strongest difference had more severe psychotic symptoms and poorer response after 12 months of antipsychotic treatment.11
Autism has been linked to increased microbiota diversity, and an excess of bacteroides has been associated with a higher diversity of autism. Fecal samples from autistic children were reported to have an increase in SCFAs. Interestingly, a certain strain of lactobacillus can modulate oxytocin or reverse some autistic symptoms.
Depression has been associated with increased diversity of microbiota alpha. Patients with depression have been reported to have low numbers of bifidobacterium
ADHD. Some studies suggest that ADHD may be linked to factors that can alter gut microbiota, including birthing mode, type of infant feeding, maternal health, and early stressors. In addition, dietary influences on gut microbiota can modify ADHD symptoms.14
Alzheimer’s disease. Metabolic dysregulation, such as obesity and diabetes, can inflame the gut microbiota, and are known risk factors for Alzheimer’s disease.15
Continue to: Irritable bowel sydrome...
Irritable bowel syndrome (IBS). Fecal microbiota transplantation has been shown to improve IBS by increasing the diversity of gut microbiota.16 It also improves patients’ mood, not just their IBS symptoms.
Alcohol use. Both alcohol consumption and alcohol withdrawal have been shown to cause immune dysregulation in the brain leading to neuroinflammation. This is attributed to the alteration in the composition of the microbiome (dysbiosis), which has a negative effect on the microbe-host homeostasis.17
The discovery of microbiome-gut-brain interactions and their bidirectional immune, endocrine, and neurotransmitter effects has been a momentous paradigm shift in health, neuroscience, and psychiatry.18 It has opened wide vistas of research for potential innovations in the prevention and treatment of various psychiatric disorders. Radical medical interventions that were previously inconceivable, such as fecal transplantation,19 are an example of the bold insights this new field of microbiome-gut-brain interaction is bringing to the landscape of medicine, including psychiatry. It has also highlighted the previously underappreciated importance of nutrition in health and disease.20
1. Nasrallah HA. Psychoneurogastroenterology: the abdominal brain, the microbiome, and psychiatry. Current Psychiatry. 2015;14(5):10-11.
2. Dinan TG, Borre YE, Cryan JF. Genomics of schizophrenia: time to consider the gut microbiome? Mol Psychiatry. 2014;19(12):1252-1257.
3. Alam R, Abdolmaleky HM, Zhou JR. Microbiome, inflammation, epigenetic alterations, and mental diseases. Am J Med Genet B Neuropsychiatr Genet. 2017;174(6):651-660.
4. Lach G, Schellekens H, Dinan TG, et al. Anxiety, depression, and the microbiome: a role for gut peptides. Neurotherapeutics. 2018;15(1):36-59.
5. Kelly JR, Kennedy PJ, Cryan JF, et al. Breaking down the barriers: the gut microbiome, intestinal permeability and stress-related psychiatric disorders. Front Cell Neurosci. 2015;9:392.
6. Rodrigues-Amorim D, Rivera-Baltanás T, Regueiro B, et al. The role of the gut microbiota in schizophrenia: current and future perspectives. World J Biol Psychiatry. 2018;21:1-15.
7. Petra AI, Panagiotidou S, Hatziagelaki E, et al. Gut-microbiota-brain axis and its effect on neuropsychiatric disorders with suspected immune dysregulation. Clin Ther. 2015;37(5):984-995.
8. Lurie I, Yang YX, Haynes K, et al. Antibiotic exposure and the risk for depression, anxiety, or psychosis: a nested case-control study. J Clin Psychiatry. 2015;76(11):1522-1528.
9. He Y, Kosciolek T, Tang J, et al. Gut microbiome and magnetic resonance spectroscopy study of subjects at ultra-high risk for psychosis may support the membrane hypothesis. Eur Psychiatry. 2018;53:37-45.
10. Yuan X, Zhang P, Wang Y, et al. Changes in metabolism and microbiota after 24-week risperidone treatment in drug naïve, normal weight patients with first episode schizophrenia. Schizophr Res. 2018;pii: S0920-9964(18)30274-3. [Epub ahead of print]. doi: 10.1016/j.schres.2018.05.017.
11. Dickerson F, Severance E, Yolken R. The microbiome, immunity, and schizophrenia and bipolar disorder. Brain Behav Immun. 2017;62:46-52.
12. Huang R, Wang K, Hu J. Effect of probiotics on depression: a systematic review and meta-analysis of randomized controlled trials. Nutrients. 2016;8(8):pii: E483. doi: 10.3390/nu8080483.
13. Carding S, Verbeke K, Vipond DT, et al. Dysbiosis of the gut microbiota in disease. Microb Ecol Health Dis. 2015;26:26191. doi: 10.3402/mehd.v26.26191.
14. Thapar A, Cooper M, Eyre O, et al. Practitioner review: what have we learnt about the causes of ADHD? J Child Psychol Psychiatry. 2013;54(1):3-16.
15. Jiang C, Li G, Huang P, et al. The gut microbiota and Alzheimer’s disease. J Alzheimers Dis. 2017;58(1):1-15.
16. Kurokawa S, Kishimoto T, Mizuno S, et al. The effect of fecal microbiota transplantation on psychiatric symptoms among patients with irritable bowel syndrome, functional diarrhea and functional constipation: an open-label observational study. J Affect Disord. 2018;235:506-512.
17. Hillemacher T, Bachmann O, Kahl KG, et al. Alcohol, microbiome, and their effect on psychiatric disorders. Prog Neuropsychopharmacol Biol Psychiatry. 2018;85:105-115.
18. Doré J, Multon MC, Béhier JM; participants of Giens XXXII, Round Table No. 2. The human gut microbiome as source of innovation for health: which physiological and therapeutic outcomes could we expect? Therapie. 2017;72(1):21-38.
19. Vemuri RC, Gundamaraju R, Shinde T, et al. Therapeutic interventions for gut dysbiosis and related disorders in the elderly: antibiotics, probiotics or faecal microbiota transplantation? Benef Microbes. 2017;8(2):179-192.
20. Lombardi VC, De Meirleir KL, Subramanian K, et al. Nutritional modulation of the intestinal microbiota; future opportunities for the prevention and treatment of neuroimmune and neuroinflammatory disease. J Nutr Biochem. 2018;61:1-16.
What is the largest endocrine organ in the human body?
Here is a clue: It is also the largest immune organ in humans!
Still scratching your head? Here is another clue: This organ also contains a “second brain,” which is connected to big brain inside the head by the vagus nerve.
Okay, enough guessing: It’s the 30-foot long gastrointestinal (GI) tract, which is generally associated only with eating and digestion. But it is far more than a digestive tract. It is home to about 100 trillion diverse bacteria, including 1,000 known species, which together are known as “microbiota.” Its combined DNA is called the “microbiome” and is 10,000% larger than the human genome. Those trillions of bacteria in our guts are a symbiotic (commensal) organ that is vital for the normal functions of the human body.1
While this vast array of microorganisms is vital to sustaining a healthy human existence, it can also be involved in multiple psychiatric disorders, including depression, psychosis, anxiety, autism, and attention-deficit/hyperactivity disorder (ADHD). Humans acquire their unique sets of microbiota as they pass through the mother’s vagina at birth and while breastfeeding, as well as from exposure to various environmental sources in the first few months of life.2
The microbiota in the GI tract are an intimate neighbor of the “enteric brain,” comprised of 100 million neurons plus glia-like support cell structures. This “second brain” produces over 30 neurotransmitters, several of which (dopamine, serotonin, γ-aminobutyric acid [GABA], acetylcholine) have been implicated in major psychiatric disorders.3
The brain and gut have a dynamic bidirectional communication system, mediated by neural, hormonal, and immunological crosstalk and influences. The GI tract secretes dozens of peptides and other signaling molecules that influence the brain. The microbiota also interact with and are regulated by gut hormones such as oxytocin, ghrelin, neuropeptide Y, cholecystokinin, corticotrophin-releasing factor, and pancreatic polypeptide.4 The microbiota modulate brain development, functions, and behavior, and maintain the intestinal barrier, which, if disrupted, would result in the gut becoming “leaky” and triggering low-grade inflammation such as that associated with depression.5
Continue to: But don't overlook the importance of...
But don’t overlook the importance of diet. It is a major factor in shaping the composition of the microbiota. What we eat can have a preventative or reparative effect on neuroimmune or neuroinflammatory disease. An emerging body of evidence suggests that the diet and its effects on the gut microbiota can modify a person’s genes by epigenetic mechanisms (altering DNA methylation and histone effects). Probiotics can exert epigenetic effects by influencing cytokines, by producing short-chain fatty acids (SCFAs), by vitamin synthesis, and by producing several well-known neurotransmitters.6
The bidirectional trafficking across the microbiome-gut-brain axis includes reciprocal effects. The brain influences the microbiome composition by regulating satiety, the hypothalamic-pituitary axis, and with neuropeptides.7 In return, the microbiome conveys information to the brain about the intestinal status via infectious agents, intestinal neurotransmitters and modulators, cytokines, sensory vagal fibers, and various metabolites. Failure of these normal interactions can lead to a variety of pathological processes, including inflammatory, autoimmune, degenerative, metabolic, cognitive, mood, and behavioral adverse effects. Therapeutic interventions for these adverse consequences can be implemented through microbiome manipulations (such as fecal transplants), nutritional strategies, and reinforcement of the enteric and brain barriers.
Alterations in the microbiota, such as by the intake of antibiotics or by intestinal inflammation, can lead to psychiatric disorders.8 The following findings link gut microbiome disruptions with several psychiatric disorders:
Schizophrenia prodrome. Fecal bacteria show an increase in SCFAs, which can activate microglia (the initial step in triggering psychosis).9 These bacteria have been shown to lead to an increase in choline levels in the anterior cingulate, a known biomarker for membrane dysfunction, which is one of the biological models of schizophrenia.
Schizophrenia—first-episode. A recent study reported abnormalities in the gut microbiota of patients with first-episode psychosis, with a lower number of certain fecal bacteria (including bifidobacterium, E. coli, and lactobacillus) and high levels of Clostridium coccoides. After 6 months of risperidone treatment, the above changes were reversed.10
Continue to: Another study of fecal microbiota...
Another study of fecal microbiota in a first-episode psychosis cohort found significant differences in several bacterial strains compared with a healthy control group, and those with the strongest difference had more severe psychotic symptoms and poorer response after 12 months of antipsychotic treatment.11
Autism has been linked to increased microbiota diversity, and an excess of bacteroides has been associated with a higher diversity of autism. Fecal samples from autistic children were reported to have an increase in SCFAs. Interestingly, a certain strain of lactobacillus can modulate oxytocin or reverse some autistic symptoms.
Depression has been associated with increased diversity of microbiota alpha. Patients with depression have been reported to have low numbers of bifidobacterium
ADHD. Some studies suggest that ADHD may be linked to factors that can alter gut microbiota, including birthing mode, type of infant feeding, maternal health, and early stressors. In addition, dietary influences on gut microbiota can modify ADHD symptoms.14
Alzheimer’s disease. Metabolic dysregulation, such as obesity and diabetes, can inflame the gut microbiota, and are known risk factors for Alzheimer’s disease.15
Continue to: Irritable bowel sydrome...
Irritable bowel syndrome (IBS). Fecal microbiota transplantation has been shown to improve IBS by increasing the diversity of gut microbiota.16 It also improves patients’ mood, not just their IBS symptoms.
Alcohol use. Both alcohol consumption and alcohol withdrawal have been shown to cause immune dysregulation in the brain leading to neuroinflammation. This is attributed to the alteration in the composition of the microbiome (dysbiosis), which has a negative effect on the microbe-host homeostasis.17
The discovery of microbiome-gut-brain interactions and their bidirectional immune, endocrine, and neurotransmitter effects has been a momentous paradigm shift in health, neuroscience, and psychiatry.18 It has opened wide vistas of research for potential innovations in the prevention and treatment of various psychiatric disorders. Radical medical interventions that were previously inconceivable, such as fecal transplantation,19 are an example of the bold insights this new field of microbiome-gut-brain interaction is bringing to the landscape of medicine, including psychiatry. It has also highlighted the previously underappreciated importance of nutrition in health and disease.20
What is the largest endocrine organ in the human body?
Here is a clue: It is also the largest immune organ in humans!
Still scratching your head? Here is another clue: This organ also contains a “second brain,” which is connected to big brain inside the head by the vagus nerve.
Okay, enough guessing: It’s the 30-foot long gastrointestinal (GI) tract, which is generally associated only with eating and digestion. But it is far more than a digestive tract. It is home to about 100 trillion diverse bacteria, including 1,000 known species, which together are known as “microbiota.” Its combined DNA is called the “microbiome” and is 10,000% larger than the human genome. Those trillions of bacteria in our guts are a symbiotic (commensal) organ that is vital for the normal functions of the human body.1
While this vast array of microorganisms is vital to sustaining a healthy human existence, it can also be involved in multiple psychiatric disorders, including depression, psychosis, anxiety, autism, and attention-deficit/hyperactivity disorder (ADHD). Humans acquire their unique sets of microbiota as they pass through the mother’s vagina at birth and while breastfeeding, as well as from exposure to various environmental sources in the first few months of life.2
The microbiota in the GI tract are an intimate neighbor of the “enteric brain,” comprised of 100 million neurons plus glia-like support cell structures. This “second brain” produces over 30 neurotransmitters, several of which (dopamine, serotonin, γ-aminobutyric acid [GABA], acetylcholine) have been implicated in major psychiatric disorders.3
The brain and gut have a dynamic bidirectional communication system, mediated by neural, hormonal, and immunological crosstalk and influences. The GI tract secretes dozens of peptides and other signaling molecules that influence the brain. The microbiota also interact with and are regulated by gut hormones such as oxytocin, ghrelin, neuropeptide Y, cholecystokinin, corticotrophin-releasing factor, and pancreatic polypeptide.4 The microbiota modulate brain development, functions, and behavior, and maintain the intestinal barrier, which, if disrupted, would result in the gut becoming “leaky” and triggering low-grade inflammation such as that associated with depression.5
Continue to: But don't overlook the importance of...
But don’t overlook the importance of diet. It is a major factor in shaping the composition of the microbiota. What we eat can have a preventative or reparative effect on neuroimmune or neuroinflammatory disease. An emerging body of evidence suggests that the diet and its effects on the gut microbiota can modify a person’s genes by epigenetic mechanisms (altering DNA methylation and histone effects). Probiotics can exert epigenetic effects by influencing cytokines, by producing short-chain fatty acids (SCFAs), by vitamin synthesis, and by producing several well-known neurotransmitters.6
The bidirectional trafficking across the microbiome-gut-brain axis includes reciprocal effects. The brain influences the microbiome composition by regulating satiety, the hypothalamic-pituitary axis, and with neuropeptides.7 In return, the microbiome conveys information to the brain about the intestinal status via infectious agents, intestinal neurotransmitters and modulators, cytokines, sensory vagal fibers, and various metabolites. Failure of these normal interactions can lead to a variety of pathological processes, including inflammatory, autoimmune, degenerative, metabolic, cognitive, mood, and behavioral adverse effects. Therapeutic interventions for these adverse consequences can be implemented through microbiome manipulations (such as fecal transplants), nutritional strategies, and reinforcement of the enteric and brain barriers.
Alterations in the microbiota, such as by the intake of antibiotics or by intestinal inflammation, can lead to psychiatric disorders.8 The following findings link gut microbiome disruptions with several psychiatric disorders:
Schizophrenia prodrome. Fecal bacteria show an increase in SCFAs, which can activate microglia (the initial step in triggering psychosis).9 These bacteria have been shown to lead to an increase in choline levels in the anterior cingulate, a known biomarker for membrane dysfunction, which is one of the biological models of schizophrenia.
Schizophrenia—first-episode. A recent study reported abnormalities in the gut microbiota of patients with first-episode psychosis, with a lower number of certain fecal bacteria (including bifidobacterium, E. coli, and lactobacillus) and high levels of Clostridium coccoides. After 6 months of risperidone treatment, the above changes were reversed.10
Continue to: Another study of fecal microbiota...
Another study of fecal microbiota in a first-episode psychosis cohort found significant differences in several bacterial strains compared with a healthy control group, and those with the strongest difference had more severe psychotic symptoms and poorer response after 12 months of antipsychotic treatment.11
Autism has been linked to increased microbiota diversity, and an excess of bacteroides has been associated with a higher diversity of autism. Fecal samples from autistic children were reported to have an increase in SCFAs. Interestingly, a certain strain of lactobacillus can modulate oxytocin or reverse some autistic symptoms.
Depression has been associated with increased diversity of microbiota alpha. Patients with depression have been reported to have low numbers of bifidobacterium
ADHD. Some studies suggest that ADHD may be linked to factors that can alter gut microbiota, including birthing mode, type of infant feeding, maternal health, and early stressors. In addition, dietary influences on gut microbiota can modify ADHD symptoms.14
Alzheimer’s disease. Metabolic dysregulation, such as obesity and diabetes, can inflame the gut microbiota, and are known risk factors for Alzheimer’s disease.15
Continue to: Irritable bowel sydrome...
Irritable bowel syndrome (IBS). Fecal microbiota transplantation has been shown to improve IBS by increasing the diversity of gut microbiota.16 It also improves patients’ mood, not just their IBS symptoms.
Alcohol use. Both alcohol consumption and alcohol withdrawal have been shown to cause immune dysregulation in the brain leading to neuroinflammation. This is attributed to the alteration in the composition of the microbiome (dysbiosis), which has a negative effect on the microbe-host homeostasis.17
The discovery of microbiome-gut-brain interactions and their bidirectional immune, endocrine, and neurotransmitter effects has been a momentous paradigm shift in health, neuroscience, and psychiatry.18 It has opened wide vistas of research for potential innovations in the prevention and treatment of various psychiatric disorders. Radical medical interventions that were previously inconceivable, such as fecal transplantation,19 are an example of the bold insights this new field of microbiome-gut-brain interaction is bringing to the landscape of medicine, including psychiatry. It has also highlighted the previously underappreciated importance of nutrition in health and disease.20
1. Nasrallah HA. Psychoneurogastroenterology: the abdominal brain, the microbiome, and psychiatry. Current Psychiatry. 2015;14(5):10-11.
2. Dinan TG, Borre YE, Cryan JF. Genomics of schizophrenia: time to consider the gut microbiome? Mol Psychiatry. 2014;19(12):1252-1257.
3. Alam R, Abdolmaleky HM, Zhou JR. Microbiome, inflammation, epigenetic alterations, and mental diseases. Am J Med Genet B Neuropsychiatr Genet. 2017;174(6):651-660.
4. Lach G, Schellekens H, Dinan TG, et al. Anxiety, depression, and the microbiome: a role for gut peptides. Neurotherapeutics. 2018;15(1):36-59.
5. Kelly JR, Kennedy PJ, Cryan JF, et al. Breaking down the barriers: the gut microbiome, intestinal permeability and stress-related psychiatric disorders. Front Cell Neurosci. 2015;9:392.
6. Rodrigues-Amorim D, Rivera-Baltanás T, Regueiro B, et al. The role of the gut microbiota in schizophrenia: current and future perspectives. World J Biol Psychiatry. 2018;21:1-15.
7. Petra AI, Panagiotidou S, Hatziagelaki E, et al. Gut-microbiota-brain axis and its effect on neuropsychiatric disorders with suspected immune dysregulation. Clin Ther. 2015;37(5):984-995.
8. Lurie I, Yang YX, Haynes K, et al. Antibiotic exposure and the risk for depression, anxiety, or psychosis: a nested case-control study. J Clin Psychiatry. 2015;76(11):1522-1528.
9. He Y, Kosciolek T, Tang J, et al. Gut microbiome and magnetic resonance spectroscopy study of subjects at ultra-high risk for psychosis may support the membrane hypothesis. Eur Psychiatry. 2018;53:37-45.
10. Yuan X, Zhang P, Wang Y, et al. Changes in metabolism and microbiota after 24-week risperidone treatment in drug naïve, normal weight patients with first episode schizophrenia. Schizophr Res. 2018;pii: S0920-9964(18)30274-3. [Epub ahead of print]. doi: 10.1016/j.schres.2018.05.017.
11. Dickerson F, Severance E, Yolken R. The microbiome, immunity, and schizophrenia and bipolar disorder. Brain Behav Immun. 2017;62:46-52.
12. Huang R, Wang K, Hu J. Effect of probiotics on depression: a systematic review and meta-analysis of randomized controlled trials. Nutrients. 2016;8(8):pii: E483. doi: 10.3390/nu8080483.
13. Carding S, Verbeke K, Vipond DT, et al. Dysbiosis of the gut microbiota in disease. Microb Ecol Health Dis. 2015;26:26191. doi: 10.3402/mehd.v26.26191.
14. Thapar A, Cooper M, Eyre O, et al. Practitioner review: what have we learnt about the causes of ADHD? J Child Psychol Psychiatry. 2013;54(1):3-16.
15. Jiang C, Li G, Huang P, et al. The gut microbiota and Alzheimer’s disease. J Alzheimers Dis. 2017;58(1):1-15.
16. Kurokawa S, Kishimoto T, Mizuno S, et al. The effect of fecal microbiota transplantation on psychiatric symptoms among patients with irritable bowel syndrome, functional diarrhea and functional constipation: an open-label observational study. J Affect Disord. 2018;235:506-512.
17. Hillemacher T, Bachmann O, Kahl KG, et al. Alcohol, microbiome, and their effect on psychiatric disorders. Prog Neuropsychopharmacol Biol Psychiatry. 2018;85:105-115.
18. Doré J, Multon MC, Béhier JM; participants of Giens XXXII, Round Table No. 2. The human gut microbiome as source of innovation for health: which physiological and therapeutic outcomes could we expect? Therapie. 2017;72(1):21-38.
19. Vemuri RC, Gundamaraju R, Shinde T, et al. Therapeutic interventions for gut dysbiosis and related disorders in the elderly: antibiotics, probiotics or faecal microbiota transplantation? Benef Microbes. 2017;8(2):179-192.
20. Lombardi VC, De Meirleir KL, Subramanian K, et al. Nutritional modulation of the intestinal microbiota; future opportunities for the prevention and treatment of neuroimmune and neuroinflammatory disease. J Nutr Biochem. 2018;61:1-16.
1. Nasrallah HA. Psychoneurogastroenterology: the abdominal brain, the microbiome, and psychiatry. Current Psychiatry. 2015;14(5):10-11.
2. Dinan TG, Borre YE, Cryan JF. Genomics of schizophrenia: time to consider the gut microbiome? Mol Psychiatry. 2014;19(12):1252-1257.
3. Alam R, Abdolmaleky HM, Zhou JR. Microbiome, inflammation, epigenetic alterations, and mental diseases. Am J Med Genet B Neuropsychiatr Genet. 2017;174(6):651-660.
4. Lach G, Schellekens H, Dinan TG, et al. Anxiety, depression, and the microbiome: a role for gut peptides. Neurotherapeutics. 2018;15(1):36-59.
5. Kelly JR, Kennedy PJ, Cryan JF, et al. Breaking down the barriers: the gut microbiome, intestinal permeability and stress-related psychiatric disorders. Front Cell Neurosci. 2015;9:392.
6. Rodrigues-Amorim D, Rivera-Baltanás T, Regueiro B, et al. The role of the gut microbiota in schizophrenia: current and future perspectives. World J Biol Psychiatry. 2018;21:1-15.
7. Petra AI, Panagiotidou S, Hatziagelaki E, et al. Gut-microbiota-brain axis and its effect on neuropsychiatric disorders with suspected immune dysregulation. Clin Ther. 2015;37(5):984-995.
8. Lurie I, Yang YX, Haynes K, et al. Antibiotic exposure and the risk for depression, anxiety, or psychosis: a nested case-control study. J Clin Psychiatry. 2015;76(11):1522-1528.
9. He Y, Kosciolek T, Tang J, et al. Gut microbiome and magnetic resonance spectroscopy study of subjects at ultra-high risk for psychosis may support the membrane hypothesis. Eur Psychiatry. 2018;53:37-45.
10. Yuan X, Zhang P, Wang Y, et al. Changes in metabolism and microbiota after 24-week risperidone treatment in drug naïve, normal weight patients with first episode schizophrenia. Schizophr Res. 2018;pii: S0920-9964(18)30274-3. [Epub ahead of print]. doi: 10.1016/j.schres.2018.05.017.
11. Dickerson F, Severance E, Yolken R. The microbiome, immunity, and schizophrenia and bipolar disorder. Brain Behav Immun. 2017;62:46-52.
12. Huang R, Wang K, Hu J. Effect of probiotics on depression: a systematic review and meta-analysis of randomized controlled trials. Nutrients. 2016;8(8):pii: E483. doi: 10.3390/nu8080483.
13. Carding S, Verbeke K, Vipond DT, et al. Dysbiosis of the gut microbiota in disease. Microb Ecol Health Dis. 2015;26:26191. doi: 10.3402/mehd.v26.26191.
14. Thapar A, Cooper M, Eyre O, et al. Practitioner review: what have we learnt about the causes of ADHD? J Child Psychol Psychiatry. 2013;54(1):3-16.
15. Jiang C, Li G, Huang P, et al. The gut microbiota and Alzheimer’s disease. J Alzheimers Dis. 2017;58(1):1-15.
16. Kurokawa S, Kishimoto T, Mizuno S, et al. The effect of fecal microbiota transplantation on psychiatric symptoms among patients with irritable bowel syndrome, functional diarrhea and functional constipation: an open-label observational study. J Affect Disord. 2018;235:506-512.
17. Hillemacher T, Bachmann O, Kahl KG, et al. Alcohol, microbiome, and their effect on psychiatric disorders. Prog Neuropsychopharmacol Biol Psychiatry. 2018;85:105-115.
18. Doré J, Multon MC, Béhier JM; participants of Giens XXXII, Round Table No. 2. The human gut microbiome as source of innovation for health: which physiological and therapeutic outcomes could we expect? Therapie. 2017;72(1):21-38.
19. Vemuri RC, Gundamaraju R, Shinde T, et al. Therapeutic interventions for gut dysbiosis and related disorders in the elderly: antibiotics, probiotics or faecal microbiota transplantation? Benef Microbes. 2017;8(2):179-192.
20. Lombardi VC, De Meirleir KL, Subramanian K, et al. Nutritional modulation of the intestinal microbiota; future opportunities for the prevention and treatment of neuroimmune and neuroinflammatory disease. J Nutr Biochem. 2018;61:1-16.
Mental illness and the criminal justice system: Reducing the risks
NEW YORK – The overrepresentation of people with serious mental illness (SMI) in the criminal justice system has led to creation of a resource from the Judges’ and Psychiatrists’ Leadership Initiative (JPLI) aimed at helping psychiatry and law enforcement address the problem.
The resource, “Supporting People with Serious Mental Illnesses and Reducing Their Risk of Contact with the Criminal Justice System: A Primer for Psychiatrists,” released last year, was designed to provide psychiatrists with specific knowledge and tools, according to Michael Champion, MD, forensic chief at the Hawaii State Department of Health, Adult Mental Health Division, Honolulu, and a member of the JPLI executive leadership team.
In developing the primer, the JPLI, which was created about 10 years ago by the American Psychiatric Association Foundation in partnership with the Council of State Governments Justice Center in response to the growing problem of such overrepresentation, sought to teach psychiatrists about what the criminal justice literature has dubbed “criminogenic risk” and to explore strategies to address those risks in community treatment settings, Dr. Champion said at the annual meeting of the American Psychiatric Association.
“The fact is that one in three Americans has a criminal record, and people with serious mental illness and criminal justice involvement are frequently part of our patient population – particularly in the public mental health sector,” Dr. Champion said. “Part of the challenge is that psychiatrists ... aren’t typically trained in these principles ... so the JPLI saw that this as an area that we could try to make some traction in and try to make a difference.”
The JPLI’s goals in publishing this resource are to reduce the risk of patient involvement in the criminal justice system, and to improve clinical and recovery outcomes by educating community psychiatrists about Risk-Need-Responsivity (RNR) principles. The JPLI also seeks to provide strategies for collaborating with criminal justice partners, incorporating criminal justice history into screening and assessment, and integrating criminogenic risk needs of patients into comprehensive treatment plans, Dr. Champion said.
Criminogenic risk and RNR
Many factors contribute to the involvement of people with serious mental illnesses in the criminal justice system, including higher rates of arrest, longer stays, recidivism, and limited access to health care, said Fred C. Osher, MD, former director of health systems and services policy for the Council of State Governments Justice Center.
“We used to think that ... if we could just get folks the health care that they need, they wouldn’t get involved with the criminal justice system. It turns out that that’s a gross oversimplification, in that their needs are terribly complex, and while treatment is a necessary component, it isn’t often sufficient for a large number of individuals,” said Dr. Osher, now a member of the JPLI executive leadership team.
Criminogenic risk – the likelihood that a person who has been arrested and jailed will commit a new crime after release or return to custody – helps explain why that is the case, he said, adding: “We have ways in which we can understand those risks.”
The risks are measured via static factors (unchanging conditions such as criminal history, age at first arrest, current age, and gender) and dynamic factors, he explained.
he said, noting that the research has shown there are eight specific criminogenic risk factors: substance abuse, history of antisocial behavior, antisocial personality pattern, antisocial cognition, antisocial associates, family and/or marital discord, poor school and/or work output, and having few leisure/recreation outlets.
Notably, mental illness is not a part of that list, he said.
“The reason for that is it’s not explanatory in and of itself,” he added.
However, research shows that people with mental illness have more of these dynamic risk factors, and research by Jennifer L. Skeem, PhD, and others shows that those with mental illness were coming back to jail not for new criminal activity, but for failing to comply with their conditions of release.
“These risks, then, have been brought into a paradigm that is central to our criminal justice operations, and it’s called the Risk-Need-Responsivity model,” Dr. Osher said. “This paradigm is what allows a criminal justice system to think about how to prioritize the resources – to think about who really needs to be wrapped tight, who needs to have close supervision, frequent reporting, lots of contact.”
The risk principle in the RNR model says that resources should be focused on high-risk cases, with limited supervision in lower-risk cases. This is based on experience demonstrating that recidivism is lower in high-risk individuals with close supervision but higher in low-risk individuals with close supervision.
The needs principle suggests that dynamic needs are “the issues that get folks in trouble,” he said.
“So, if we’re going to intervene, if we’re going to provide programming, if we’re going to try and help that individual stay out of jail or prison, we need to address these criminogenic needs,” he said, adding that the “big four” are related to their antisocial thinking and personality and friends.
Targeted interventions can help those individuals make better choices going forward, he noted.
The responsivity principle is an acknowledgment that individuals have different ways of learning, different cultural factors and backgrounds that influence them, and social determinants that are important to understand if they predict the ability to stay out of trouble.
“This is where mental illness fits in,” Dr. Osher said. “It’s absolutely important that we understand that.”
Examples would be patients with severe major depressive disorder who need their depression treated before they can participate in a group treatment setting designed to address criminogenic risks.
Dynamic risk factors are best treated with cognitive-behavioral interventions, Dr. Osher said, noting that the most effective interventions provide opportunities for participants to practice new behavior patterns and skills with feedback from program staff.
In many states, those interventions are being provided by criminal justice personnel, including probation officers, partly because of “an absence of [psychiatrists’] understanding, willingness, or ability to step forward.” The JPLI primer is designed to “really amp up our own excitement about, and willingness to learn how to develop interventions to help that individual stay out of trouble,” and it includes detailed descriptions of numerous well-researched, standardized, manualized interventions that people can access that make it less likely for them to have criminal justice access going forward, he said.
Those include programs such as “Thinking for a Change,” “Reasoning and Rehabilitation,” “Moral Reconation Therapy,” and “Interactive Journaling.”
A focus on the Sequential Intercept Model, which describes how individuals move through the criminal justice system, illustrates multiple points where psychiatrists can “do things better and differently to intervene,” he said, noting that the primer includes a framework for prioritizing the target population, and validated screening and assessment tools, including tools to help corrections officers identify mental health/substance abuse/criminogenic issues at the time individuals are booked into jail so they can be referred for appropriate interventions.
Achieving positive public health and safety outcomes requires changes to policy and practice, Dr. Osher said.
The JPLI primer is a step toward making such changes, and with it comes a set of four principles:
1. Conduct universal risk, substance use, and mental health screening at booking, and full assessments as appropriate, he said, noting that “13 million times this year (9 million unduplicated count), 2 million folks with serious mental illness are going to be arrested and brought to jail. Let’s make sure they get assessed, identified, and then a plan can be made.”
2. Get relevant information into the hands of decision makers in time to inform pretrial release decisions. For example, knowing if someone is eligible for a mental health court could lead to that person’s receiving necessary support and supervision, he said.
3. Use assessment information to connect people to appropriate jail-based services and post-release services and supervision, and ensure that there is communication between the two.
4. Ensure services and supervision are evidence based and hold systems accountable by measuring outcomes.
In addition, the goal is to partner with the criminal justice system through information-sharing agreements and integrating dynamic criminogenic risk factors into treatment plans, he said.
The intercepts
To demonstrate ways in which psychiatrists can intervene over the course of a patients’ journey toward involvement in the criminal justice system, Stephanie Le Melle, MD, provided a case example involving a 30-year-old African American man diagnosed with schizophrenia at age 18 years.
As a child, “Joe” was neglected and abused; both parents had a history of mental illness and substance use. He experienced homelessness, never finished high school, and was hospitalized or visited the emergency department more than 15 times after going off medications or because of intoxication.
His history with the legal system involved a first arrest at age 14 years for gang-related fighting and assault (after being bullied as a child and seeking safety in a gang), followed by 3 years in juvenile detention. He was released with supervision at age 17 years, was arrested several times after that for public intoxication and loitering, and was held for several days or weeks each time – then released with time served or summons paid. His first hospitalization occurred at age 18, when he was diagnosed with psychosis.
Subsequent experiences included treatment in a community mental health program at age 25 for heroin use and drinking. However, he was denied admission to a substance abuse program because of his history of psychosis and violence. After stopping his medications because of side effects, he tried to buy heroin, got into a fight, and was arrested for assault with a pocket knife. He resisted arrest and was tasered, handcuffed, and taken to prison, where he was held because he could not afford bail. Involvement in gang activity while in prison led to sanctions, including time in solitary confinement.
During all of his time in the criminal justice system, Joe refused treatment, because he was afraid he’d be considered “crazy” and would be preyed upon even more by other inmates. After about 3 years, he was released to a Forensic Assertive Community Treatment team for 2 years and completed that program, and is now receiving treatment in the community. He lives alone in supported housing and has Supplemental Security Income. He does not engage in clinic-related activities and has a lack of trust in the clinical team. He often is agitated and disruptive in the clinic. Staff members have concerns about his history of violence and drug use, and were reluctant to bring him into the program.
“Going back to ... the sequential intercept model, we can think about things, as psychiatrists, that we could have done for Joe all along the way to help him not get into the criminal justice system in the first place,” said Dr. Le Melle, director of public psychiatry education at Columbia University/New York State Psychiatric Institute, New York.
This is a framework for thinking through treatment for a patient like Joe:
Intercept 0 (community services). At this early stage, Joe would have been screened for adverse childhood experiences, and that could have led to trauma treatment, substance abuse treatment, and educational and vocational services. Awareness of his family illness, discord, and poverty would have led to parenting interventions, early school involvement, and promotion of meaningful activities, she said.
“These are things, again, that we can address as clinicians ... to intervene with families and with schools and communities to try to give young people an opportunity to not get into the criminal justice system,” she said, adding that providing early co-occurring treatment for mental health and substance use is particularly important.
Intercept 1 (law enforcement) also is a stage during which a psychiatrist can intervene by giving pertinent information when 911 is called by providing police or corrections with contact information for follow-up. For Joe, psychiatrist involvement at this intercept could have allowed for treatment recommendations or assessment for diversion programs, and in fact, at some point during his care, did allow for communication about his treatment needs, Dr. Le Melle said.
In general, psychiatrists also can participate at this stage through provision of crisis intervention team training for first responders or by being part of a co-response team, she said.
Intercept 2 (initial detention/initial court hearings). Attending court on behalf of a patient can make a real difference in outcomes, she noted.
“Judges want to know that someone is out there who can help, and they want to know that there’s a team of people who can intervene and try to get someone out of the criminal justice system,” she said.
At this stage, psychiatrists can help by recommending a treatment plan for a diversion program, and – within HIPAA guidelines – can share pertinent information about treatment needs and preferences.
Intercept 3 (jails/courts). At this in-the-system stage, information shared between corrections and community behavioral health would have led to Joe’s transfer to a mental health/observation unit; he would have been offered mental health treatment and been started on substance use treatment; and he would have participated in motivational treatment and cognitive-behavioral therapy targeting his criminogenic needs, she said.
Meeting with individuals while they are incarcerated can be helpful for “keeping them grounded.”
This also is a stage where psychiatrists could help individuals prepare for release by getting them into a GED program or other training.
Intercept 4 (reentry). With appropriate intervention at this stage, Joe would have his benefits, such as Medicaid and Supplemental Security Income, reinstated prior to reentry to the community. Also, his psychiatrist and treatment program would be contacted. He would be welcomed back into treatment, and he would have assistance finding a permanent place to live with services provided in the community.
Intercept 5 (community corrections). At this stage, community behavioral health clinicians would maintain awareness of their biases and fears about people involved in the criminal justice system and avoid making assumptions about Joe. His risks, needs, and priorities would be assessed and addressed, and he would be asked about his experiences with the system and about what could be done to help him avoid incarceration in the future.
He would receive help in incorporating alternative behaviors and thinking to address dynamic criminogenic risk, and evidence-based practices would be used in treatment.
The sequential intercept model reflects the fact that the criminal justice system and the people it serves are part of the community, Dr. Le Melle said.
“The community and the behavioral health system and the criminal justice system are partners in our shared mission of public safety and public health, so we are one and we can’t expect that our responsibility for providing people with the best care and services ends if someone is in the criminal justice system,” she said.
Dr. Champion, Dr. Osher, and Dr. Le Melle reported having no disclosures.
NEW YORK – The overrepresentation of people with serious mental illness (SMI) in the criminal justice system has led to creation of a resource from the Judges’ and Psychiatrists’ Leadership Initiative (JPLI) aimed at helping psychiatry and law enforcement address the problem.
The resource, “Supporting People with Serious Mental Illnesses and Reducing Their Risk of Contact with the Criminal Justice System: A Primer for Psychiatrists,” released last year, was designed to provide psychiatrists with specific knowledge and tools, according to Michael Champion, MD, forensic chief at the Hawaii State Department of Health, Adult Mental Health Division, Honolulu, and a member of the JPLI executive leadership team.
In developing the primer, the JPLI, which was created about 10 years ago by the American Psychiatric Association Foundation in partnership with the Council of State Governments Justice Center in response to the growing problem of such overrepresentation, sought to teach psychiatrists about what the criminal justice literature has dubbed “criminogenic risk” and to explore strategies to address those risks in community treatment settings, Dr. Champion said at the annual meeting of the American Psychiatric Association.
“The fact is that one in three Americans has a criminal record, and people with serious mental illness and criminal justice involvement are frequently part of our patient population – particularly in the public mental health sector,” Dr. Champion said. “Part of the challenge is that psychiatrists ... aren’t typically trained in these principles ... so the JPLI saw that this as an area that we could try to make some traction in and try to make a difference.”
The JPLI’s goals in publishing this resource are to reduce the risk of patient involvement in the criminal justice system, and to improve clinical and recovery outcomes by educating community psychiatrists about Risk-Need-Responsivity (RNR) principles. The JPLI also seeks to provide strategies for collaborating with criminal justice partners, incorporating criminal justice history into screening and assessment, and integrating criminogenic risk needs of patients into comprehensive treatment plans, Dr. Champion said.
Criminogenic risk and RNR
Many factors contribute to the involvement of people with serious mental illnesses in the criminal justice system, including higher rates of arrest, longer stays, recidivism, and limited access to health care, said Fred C. Osher, MD, former director of health systems and services policy for the Council of State Governments Justice Center.
“We used to think that ... if we could just get folks the health care that they need, they wouldn’t get involved with the criminal justice system. It turns out that that’s a gross oversimplification, in that their needs are terribly complex, and while treatment is a necessary component, it isn’t often sufficient for a large number of individuals,” said Dr. Osher, now a member of the JPLI executive leadership team.
Criminogenic risk – the likelihood that a person who has been arrested and jailed will commit a new crime after release or return to custody – helps explain why that is the case, he said, adding: “We have ways in which we can understand those risks.”
The risks are measured via static factors (unchanging conditions such as criminal history, age at first arrest, current age, and gender) and dynamic factors, he explained.
he said, noting that the research has shown there are eight specific criminogenic risk factors: substance abuse, history of antisocial behavior, antisocial personality pattern, antisocial cognition, antisocial associates, family and/or marital discord, poor school and/or work output, and having few leisure/recreation outlets.
Notably, mental illness is not a part of that list, he said.
“The reason for that is it’s not explanatory in and of itself,” he added.
However, research shows that people with mental illness have more of these dynamic risk factors, and research by Jennifer L. Skeem, PhD, and others shows that those with mental illness were coming back to jail not for new criminal activity, but for failing to comply with their conditions of release.
“These risks, then, have been brought into a paradigm that is central to our criminal justice operations, and it’s called the Risk-Need-Responsivity model,” Dr. Osher said. “This paradigm is what allows a criminal justice system to think about how to prioritize the resources – to think about who really needs to be wrapped tight, who needs to have close supervision, frequent reporting, lots of contact.”
The risk principle in the RNR model says that resources should be focused on high-risk cases, with limited supervision in lower-risk cases. This is based on experience demonstrating that recidivism is lower in high-risk individuals with close supervision but higher in low-risk individuals with close supervision.
The needs principle suggests that dynamic needs are “the issues that get folks in trouble,” he said.
“So, if we’re going to intervene, if we’re going to provide programming, if we’re going to try and help that individual stay out of jail or prison, we need to address these criminogenic needs,” he said, adding that the “big four” are related to their antisocial thinking and personality and friends.
Targeted interventions can help those individuals make better choices going forward, he noted.
The responsivity principle is an acknowledgment that individuals have different ways of learning, different cultural factors and backgrounds that influence them, and social determinants that are important to understand if they predict the ability to stay out of trouble.
“This is where mental illness fits in,” Dr. Osher said. “It’s absolutely important that we understand that.”
Examples would be patients with severe major depressive disorder who need their depression treated before they can participate in a group treatment setting designed to address criminogenic risks.
Dynamic risk factors are best treated with cognitive-behavioral interventions, Dr. Osher said, noting that the most effective interventions provide opportunities for participants to practice new behavior patterns and skills with feedback from program staff.
In many states, those interventions are being provided by criminal justice personnel, including probation officers, partly because of “an absence of [psychiatrists’] understanding, willingness, or ability to step forward.” The JPLI primer is designed to “really amp up our own excitement about, and willingness to learn how to develop interventions to help that individual stay out of trouble,” and it includes detailed descriptions of numerous well-researched, standardized, manualized interventions that people can access that make it less likely for them to have criminal justice access going forward, he said.
Those include programs such as “Thinking for a Change,” “Reasoning and Rehabilitation,” “Moral Reconation Therapy,” and “Interactive Journaling.”
A focus on the Sequential Intercept Model, which describes how individuals move through the criminal justice system, illustrates multiple points where psychiatrists can “do things better and differently to intervene,” he said, noting that the primer includes a framework for prioritizing the target population, and validated screening and assessment tools, including tools to help corrections officers identify mental health/substance abuse/criminogenic issues at the time individuals are booked into jail so they can be referred for appropriate interventions.
Achieving positive public health and safety outcomes requires changes to policy and practice, Dr. Osher said.
The JPLI primer is a step toward making such changes, and with it comes a set of four principles:
1. Conduct universal risk, substance use, and mental health screening at booking, and full assessments as appropriate, he said, noting that “13 million times this year (9 million unduplicated count), 2 million folks with serious mental illness are going to be arrested and brought to jail. Let’s make sure they get assessed, identified, and then a plan can be made.”
2. Get relevant information into the hands of decision makers in time to inform pretrial release decisions. For example, knowing if someone is eligible for a mental health court could lead to that person’s receiving necessary support and supervision, he said.
3. Use assessment information to connect people to appropriate jail-based services and post-release services and supervision, and ensure that there is communication between the two.
4. Ensure services and supervision are evidence based and hold systems accountable by measuring outcomes.
In addition, the goal is to partner with the criminal justice system through information-sharing agreements and integrating dynamic criminogenic risk factors into treatment plans, he said.
The intercepts
To demonstrate ways in which psychiatrists can intervene over the course of a patients’ journey toward involvement in the criminal justice system, Stephanie Le Melle, MD, provided a case example involving a 30-year-old African American man diagnosed with schizophrenia at age 18 years.
As a child, “Joe” was neglected and abused; both parents had a history of mental illness and substance use. He experienced homelessness, never finished high school, and was hospitalized or visited the emergency department more than 15 times after going off medications or because of intoxication.
His history with the legal system involved a first arrest at age 14 years for gang-related fighting and assault (after being bullied as a child and seeking safety in a gang), followed by 3 years in juvenile detention. He was released with supervision at age 17 years, was arrested several times after that for public intoxication and loitering, and was held for several days or weeks each time – then released with time served or summons paid. His first hospitalization occurred at age 18, when he was diagnosed with psychosis.
Subsequent experiences included treatment in a community mental health program at age 25 for heroin use and drinking. However, he was denied admission to a substance abuse program because of his history of psychosis and violence. After stopping his medications because of side effects, he tried to buy heroin, got into a fight, and was arrested for assault with a pocket knife. He resisted arrest and was tasered, handcuffed, and taken to prison, where he was held because he could not afford bail. Involvement in gang activity while in prison led to sanctions, including time in solitary confinement.
During all of his time in the criminal justice system, Joe refused treatment, because he was afraid he’d be considered “crazy” and would be preyed upon even more by other inmates. After about 3 years, he was released to a Forensic Assertive Community Treatment team for 2 years and completed that program, and is now receiving treatment in the community. He lives alone in supported housing and has Supplemental Security Income. He does not engage in clinic-related activities and has a lack of trust in the clinical team. He often is agitated and disruptive in the clinic. Staff members have concerns about his history of violence and drug use, and were reluctant to bring him into the program.
“Going back to ... the sequential intercept model, we can think about things, as psychiatrists, that we could have done for Joe all along the way to help him not get into the criminal justice system in the first place,” said Dr. Le Melle, director of public psychiatry education at Columbia University/New York State Psychiatric Institute, New York.
This is a framework for thinking through treatment for a patient like Joe:
Intercept 0 (community services). At this early stage, Joe would have been screened for adverse childhood experiences, and that could have led to trauma treatment, substance abuse treatment, and educational and vocational services. Awareness of his family illness, discord, and poverty would have led to parenting interventions, early school involvement, and promotion of meaningful activities, she said.
“These are things, again, that we can address as clinicians ... to intervene with families and with schools and communities to try to give young people an opportunity to not get into the criminal justice system,” she said, adding that providing early co-occurring treatment for mental health and substance use is particularly important.
Intercept 1 (law enforcement) also is a stage during which a psychiatrist can intervene by giving pertinent information when 911 is called by providing police or corrections with contact information for follow-up. For Joe, psychiatrist involvement at this intercept could have allowed for treatment recommendations or assessment for diversion programs, and in fact, at some point during his care, did allow for communication about his treatment needs, Dr. Le Melle said.
In general, psychiatrists also can participate at this stage through provision of crisis intervention team training for first responders or by being part of a co-response team, she said.
Intercept 2 (initial detention/initial court hearings). Attending court on behalf of a patient can make a real difference in outcomes, she noted.
“Judges want to know that someone is out there who can help, and they want to know that there’s a team of people who can intervene and try to get someone out of the criminal justice system,” she said.
At this stage, psychiatrists can help by recommending a treatment plan for a diversion program, and – within HIPAA guidelines – can share pertinent information about treatment needs and preferences.
Intercept 3 (jails/courts). At this in-the-system stage, information shared between corrections and community behavioral health would have led to Joe’s transfer to a mental health/observation unit; he would have been offered mental health treatment and been started on substance use treatment; and he would have participated in motivational treatment and cognitive-behavioral therapy targeting his criminogenic needs, she said.
Meeting with individuals while they are incarcerated can be helpful for “keeping them grounded.”
This also is a stage where psychiatrists could help individuals prepare for release by getting them into a GED program or other training.
Intercept 4 (reentry). With appropriate intervention at this stage, Joe would have his benefits, such as Medicaid and Supplemental Security Income, reinstated prior to reentry to the community. Also, his psychiatrist and treatment program would be contacted. He would be welcomed back into treatment, and he would have assistance finding a permanent place to live with services provided in the community.
Intercept 5 (community corrections). At this stage, community behavioral health clinicians would maintain awareness of their biases and fears about people involved in the criminal justice system and avoid making assumptions about Joe. His risks, needs, and priorities would be assessed and addressed, and he would be asked about his experiences with the system and about what could be done to help him avoid incarceration in the future.
He would receive help in incorporating alternative behaviors and thinking to address dynamic criminogenic risk, and evidence-based practices would be used in treatment.
The sequential intercept model reflects the fact that the criminal justice system and the people it serves are part of the community, Dr. Le Melle said.
“The community and the behavioral health system and the criminal justice system are partners in our shared mission of public safety and public health, so we are one and we can’t expect that our responsibility for providing people with the best care and services ends if someone is in the criminal justice system,” she said.
Dr. Champion, Dr. Osher, and Dr. Le Melle reported having no disclosures.
NEW YORK – The overrepresentation of people with serious mental illness (SMI) in the criminal justice system has led to creation of a resource from the Judges’ and Psychiatrists’ Leadership Initiative (JPLI) aimed at helping psychiatry and law enforcement address the problem.
The resource, “Supporting People with Serious Mental Illnesses and Reducing Their Risk of Contact with the Criminal Justice System: A Primer for Psychiatrists,” released last year, was designed to provide psychiatrists with specific knowledge and tools, according to Michael Champion, MD, forensic chief at the Hawaii State Department of Health, Adult Mental Health Division, Honolulu, and a member of the JPLI executive leadership team.
In developing the primer, the JPLI, which was created about 10 years ago by the American Psychiatric Association Foundation in partnership with the Council of State Governments Justice Center in response to the growing problem of such overrepresentation, sought to teach psychiatrists about what the criminal justice literature has dubbed “criminogenic risk” and to explore strategies to address those risks in community treatment settings, Dr. Champion said at the annual meeting of the American Psychiatric Association.
“The fact is that one in three Americans has a criminal record, and people with serious mental illness and criminal justice involvement are frequently part of our patient population – particularly in the public mental health sector,” Dr. Champion said. “Part of the challenge is that psychiatrists ... aren’t typically trained in these principles ... so the JPLI saw that this as an area that we could try to make some traction in and try to make a difference.”
The JPLI’s goals in publishing this resource are to reduce the risk of patient involvement in the criminal justice system, and to improve clinical and recovery outcomes by educating community psychiatrists about Risk-Need-Responsivity (RNR) principles. The JPLI also seeks to provide strategies for collaborating with criminal justice partners, incorporating criminal justice history into screening and assessment, and integrating criminogenic risk needs of patients into comprehensive treatment plans, Dr. Champion said.
Criminogenic risk and RNR
Many factors contribute to the involvement of people with serious mental illnesses in the criminal justice system, including higher rates of arrest, longer stays, recidivism, and limited access to health care, said Fred C. Osher, MD, former director of health systems and services policy for the Council of State Governments Justice Center.
“We used to think that ... if we could just get folks the health care that they need, they wouldn’t get involved with the criminal justice system. It turns out that that’s a gross oversimplification, in that their needs are terribly complex, and while treatment is a necessary component, it isn’t often sufficient for a large number of individuals,” said Dr. Osher, now a member of the JPLI executive leadership team.
Criminogenic risk – the likelihood that a person who has been arrested and jailed will commit a new crime after release or return to custody – helps explain why that is the case, he said, adding: “We have ways in which we can understand those risks.”
The risks are measured via static factors (unchanging conditions such as criminal history, age at first arrest, current age, and gender) and dynamic factors, he explained.
he said, noting that the research has shown there are eight specific criminogenic risk factors: substance abuse, history of antisocial behavior, antisocial personality pattern, antisocial cognition, antisocial associates, family and/or marital discord, poor school and/or work output, and having few leisure/recreation outlets.
Notably, mental illness is not a part of that list, he said.
“The reason for that is it’s not explanatory in and of itself,” he added.
However, research shows that people with mental illness have more of these dynamic risk factors, and research by Jennifer L. Skeem, PhD, and others shows that those with mental illness were coming back to jail not for new criminal activity, but for failing to comply with their conditions of release.
“These risks, then, have been brought into a paradigm that is central to our criminal justice operations, and it’s called the Risk-Need-Responsivity model,” Dr. Osher said. “This paradigm is what allows a criminal justice system to think about how to prioritize the resources – to think about who really needs to be wrapped tight, who needs to have close supervision, frequent reporting, lots of contact.”
The risk principle in the RNR model says that resources should be focused on high-risk cases, with limited supervision in lower-risk cases. This is based on experience demonstrating that recidivism is lower in high-risk individuals with close supervision but higher in low-risk individuals with close supervision.
The needs principle suggests that dynamic needs are “the issues that get folks in trouble,” he said.
“So, if we’re going to intervene, if we’re going to provide programming, if we’re going to try and help that individual stay out of jail or prison, we need to address these criminogenic needs,” he said, adding that the “big four” are related to their antisocial thinking and personality and friends.
Targeted interventions can help those individuals make better choices going forward, he noted.
The responsivity principle is an acknowledgment that individuals have different ways of learning, different cultural factors and backgrounds that influence them, and social determinants that are important to understand if they predict the ability to stay out of trouble.
“This is where mental illness fits in,” Dr. Osher said. “It’s absolutely important that we understand that.”
Examples would be patients with severe major depressive disorder who need their depression treated before they can participate in a group treatment setting designed to address criminogenic risks.
Dynamic risk factors are best treated with cognitive-behavioral interventions, Dr. Osher said, noting that the most effective interventions provide opportunities for participants to practice new behavior patterns and skills with feedback from program staff.
In many states, those interventions are being provided by criminal justice personnel, including probation officers, partly because of “an absence of [psychiatrists’] understanding, willingness, or ability to step forward.” The JPLI primer is designed to “really amp up our own excitement about, and willingness to learn how to develop interventions to help that individual stay out of trouble,” and it includes detailed descriptions of numerous well-researched, standardized, manualized interventions that people can access that make it less likely for them to have criminal justice access going forward, he said.
Those include programs such as “Thinking for a Change,” “Reasoning and Rehabilitation,” “Moral Reconation Therapy,” and “Interactive Journaling.”
A focus on the Sequential Intercept Model, which describes how individuals move through the criminal justice system, illustrates multiple points where psychiatrists can “do things better and differently to intervene,” he said, noting that the primer includes a framework for prioritizing the target population, and validated screening and assessment tools, including tools to help corrections officers identify mental health/substance abuse/criminogenic issues at the time individuals are booked into jail so they can be referred for appropriate interventions.
Achieving positive public health and safety outcomes requires changes to policy and practice, Dr. Osher said.
The JPLI primer is a step toward making such changes, and with it comes a set of four principles:
1. Conduct universal risk, substance use, and mental health screening at booking, and full assessments as appropriate, he said, noting that “13 million times this year (9 million unduplicated count), 2 million folks with serious mental illness are going to be arrested and brought to jail. Let’s make sure they get assessed, identified, and then a plan can be made.”
2. Get relevant information into the hands of decision makers in time to inform pretrial release decisions. For example, knowing if someone is eligible for a mental health court could lead to that person’s receiving necessary support and supervision, he said.
3. Use assessment information to connect people to appropriate jail-based services and post-release services and supervision, and ensure that there is communication between the two.
4. Ensure services and supervision are evidence based and hold systems accountable by measuring outcomes.
In addition, the goal is to partner with the criminal justice system through information-sharing agreements and integrating dynamic criminogenic risk factors into treatment plans, he said.
The intercepts
To demonstrate ways in which psychiatrists can intervene over the course of a patients’ journey toward involvement in the criminal justice system, Stephanie Le Melle, MD, provided a case example involving a 30-year-old African American man diagnosed with schizophrenia at age 18 years.
As a child, “Joe” was neglected and abused; both parents had a history of mental illness and substance use. He experienced homelessness, never finished high school, and was hospitalized or visited the emergency department more than 15 times after going off medications or because of intoxication.
His history with the legal system involved a first arrest at age 14 years for gang-related fighting and assault (after being bullied as a child and seeking safety in a gang), followed by 3 years in juvenile detention. He was released with supervision at age 17 years, was arrested several times after that for public intoxication and loitering, and was held for several days or weeks each time – then released with time served or summons paid. His first hospitalization occurred at age 18, when he was diagnosed with psychosis.
Subsequent experiences included treatment in a community mental health program at age 25 for heroin use and drinking. However, he was denied admission to a substance abuse program because of his history of psychosis and violence. After stopping his medications because of side effects, he tried to buy heroin, got into a fight, and was arrested for assault with a pocket knife. He resisted arrest and was tasered, handcuffed, and taken to prison, where he was held because he could not afford bail. Involvement in gang activity while in prison led to sanctions, including time in solitary confinement.
During all of his time in the criminal justice system, Joe refused treatment, because he was afraid he’d be considered “crazy” and would be preyed upon even more by other inmates. After about 3 years, he was released to a Forensic Assertive Community Treatment team for 2 years and completed that program, and is now receiving treatment in the community. He lives alone in supported housing and has Supplemental Security Income. He does not engage in clinic-related activities and has a lack of trust in the clinical team. He often is agitated and disruptive in the clinic. Staff members have concerns about his history of violence and drug use, and were reluctant to bring him into the program.
“Going back to ... the sequential intercept model, we can think about things, as psychiatrists, that we could have done for Joe all along the way to help him not get into the criminal justice system in the first place,” said Dr. Le Melle, director of public psychiatry education at Columbia University/New York State Psychiatric Institute, New York.
This is a framework for thinking through treatment for a patient like Joe:
Intercept 0 (community services). At this early stage, Joe would have been screened for adverse childhood experiences, and that could have led to trauma treatment, substance abuse treatment, and educational and vocational services. Awareness of his family illness, discord, and poverty would have led to parenting interventions, early school involvement, and promotion of meaningful activities, she said.
“These are things, again, that we can address as clinicians ... to intervene with families and with schools and communities to try to give young people an opportunity to not get into the criminal justice system,” she said, adding that providing early co-occurring treatment for mental health and substance use is particularly important.
Intercept 1 (law enforcement) also is a stage during which a psychiatrist can intervene by giving pertinent information when 911 is called by providing police or corrections with contact information for follow-up. For Joe, psychiatrist involvement at this intercept could have allowed for treatment recommendations or assessment for diversion programs, and in fact, at some point during his care, did allow for communication about his treatment needs, Dr. Le Melle said.
In general, psychiatrists also can participate at this stage through provision of crisis intervention team training for first responders or by being part of a co-response team, she said.
Intercept 2 (initial detention/initial court hearings). Attending court on behalf of a patient can make a real difference in outcomes, she noted.
“Judges want to know that someone is out there who can help, and they want to know that there’s a team of people who can intervene and try to get someone out of the criminal justice system,” she said.
At this stage, psychiatrists can help by recommending a treatment plan for a diversion program, and – within HIPAA guidelines – can share pertinent information about treatment needs and preferences.
Intercept 3 (jails/courts). At this in-the-system stage, information shared between corrections and community behavioral health would have led to Joe’s transfer to a mental health/observation unit; he would have been offered mental health treatment and been started on substance use treatment; and he would have participated in motivational treatment and cognitive-behavioral therapy targeting his criminogenic needs, she said.
Meeting with individuals while they are incarcerated can be helpful for “keeping them grounded.”
This also is a stage where psychiatrists could help individuals prepare for release by getting them into a GED program or other training.
Intercept 4 (reentry). With appropriate intervention at this stage, Joe would have his benefits, such as Medicaid and Supplemental Security Income, reinstated prior to reentry to the community. Also, his psychiatrist and treatment program would be contacted. He would be welcomed back into treatment, and he would have assistance finding a permanent place to live with services provided in the community.
Intercept 5 (community corrections). At this stage, community behavioral health clinicians would maintain awareness of their biases and fears about people involved in the criminal justice system and avoid making assumptions about Joe. His risks, needs, and priorities would be assessed and addressed, and he would be asked about his experiences with the system and about what could be done to help him avoid incarceration in the future.
He would receive help in incorporating alternative behaviors and thinking to address dynamic criminogenic risk, and evidence-based practices would be used in treatment.
The sequential intercept model reflects the fact that the criminal justice system and the people it serves are part of the community, Dr. Le Melle said.
“The community and the behavioral health system and the criminal justice system are partners in our shared mission of public safety and public health, so we are one and we can’t expect that our responsibility for providing people with the best care and services ends if someone is in the criminal justice system,” she said.
Dr. Champion, Dr. Osher, and Dr. Le Melle reported having no disclosures.
REPORTING FROM APA 2018
Childhood change of residence raises psychoses risk in young adults
Children and adolescents who moved longer distances or more frequently before 16 years of age were significantly more likely to develop psychosis in early adulthood than were those with less residential mobility, according to data from about 1.4 million children and adolescents in Sweden.
Data from previous studies have supported a link between childhood residential mobility and subsequent nonaffective psychoses, but no research has addressed the effects in later adolescence and young adulthood until now, wrote Ceri Price of Cardiff (Wales) University and colleagues.
In a study published in JAMA Psychiatry, the researchers reviewed data from a population-based cohort of individuals who were born in Sweden between Jan. 1, 1982, and Dec. 31, 1995, and lived in Sweden at age 16 years. The participants were followed from their 16th birthdays until a diagnosis of a nonaffective psychotic disorder, death, censorship because of emigration, or Dec. 31, 2011 – whichever came first.
Overall, the most sensitive range for an association between moving and psychosis was ages 16-19 years; the adjusted hazard ratio for a nonaffective psychotic disorder was 1.99 for participants who moved each year between ages 16 and 19 years, compared with those who never moved. In addition, moving greater distances before 16 years of age was independently associated with an increased risk of nonaffective psychosis (HR, 1.11) and the data suggested a nonlinear threshold effect when the distance moved exceeded 30 km.
A total of 4,537 individuals had a nonaffective psychotic disorder at a median 21 years of age, and a dose-response relationship emerged between more frequent moves and increased risk of nonaffective psychosis after controlling for confounding variables.
By contrast, a single move in young adulthood was not associated with increased psychosis risk, but moving at least four times during young adulthood was associated with an increased risk (adjusted HR, 1.82).
The study findings were strengthened by the longitudinal design and large population, but they were limited by several factors, including an absence of data on other adverse childhood experiences, such as family discord; peer relationships, such as friendships and bullying; and information on school changes and the disruption of peer relationships, the researchers wrote.
However, the results support the theory that psychosis risk can be affected by the disruption of social networks, peer support, and identity formation that occurs when children and adolescents move, and these results have potential implications for child health services and social policy, they noted.
“It is important that health, social, and educational practitioners ensure that children and adolescents who are newly resident to their neighborhoods receive adequate support to minimize the risks of adverse outcomes during adulthood, and every effort should be made to ensure the effective transfer of care for highly mobile children who are already in contact with health and social services,” they said.
The researchers had no financial conflicts to disclose. The study was supported in part by the Wellcome Trust and the Royal Society.
SOURCE: Price C et al. JAMA Psychiatry. 2018 Aug 22. doi: 10.1001/jamapsychiatry.2018.2233.
Children and adolescents who moved longer distances or more frequently before 16 years of age were significantly more likely to develop psychosis in early adulthood than were those with less residential mobility, according to data from about 1.4 million children and adolescents in Sweden.
Data from previous studies have supported a link between childhood residential mobility and subsequent nonaffective psychoses, but no research has addressed the effects in later adolescence and young adulthood until now, wrote Ceri Price of Cardiff (Wales) University and colleagues.
In a study published in JAMA Psychiatry, the researchers reviewed data from a population-based cohort of individuals who were born in Sweden between Jan. 1, 1982, and Dec. 31, 1995, and lived in Sweden at age 16 years. The participants were followed from their 16th birthdays until a diagnosis of a nonaffective psychotic disorder, death, censorship because of emigration, or Dec. 31, 2011 – whichever came first.
Overall, the most sensitive range for an association between moving and psychosis was ages 16-19 years; the adjusted hazard ratio for a nonaffective psychotic disorder was 1.99 for participants who moved each year between ages 16 and 19 years, compared with those who never moved. In addition, moving greater distances before 16 years of age was independently associated with an increased risk of nonaffective psychosis (HR, 1.11) and the data suggested a nonlinear threshold effect when the distance moved exceeded 30 km.
A total of 4,537 individuals had a nonaffective psychotic disorder at a median 21 years of age, and a dose-response relationship emerged between more frequent moves and increased risk of nonaffective psychosis after controlling for confounding variables.
By contrast, a single move in young adulthood was not associated with increased psychosis risk, but moving at least four times during young adulthood was associated with an increased risk (adjusted HR, 1.82).
The study findings were strengthened by the longitudinal design and large population, but they were limited by several factors, including an absence of data on other adverse childhood experiences, such as family discord; peer relationships, such as friendships and bullying; and information on school changes and the disruption of peer relationships, the researchers wrote.
However, the results support the theory that psychosis risk can be affected by the disruption of social networks, peer support, and identity formation that occurs when children and adolescents move, and these results have potential implications for child health services and social policy, they noted.
“It is important that health, social, and educational practitioners ensure that children and adolescents who are newly resident to their neighborhoods receive adequate support to minimize the risks of adverse outcomes during adulthood, and every effort should be made to ensure the effective transfer of care for highly mobile children who are already in contact with health and social services,” they said.
The researchers had no financial conflicts to disclose. The study was supported in part by the Wellcome Trust and the Royal Society.
SOURCE: Price C et al. JAMA Psychiatry. 2018 Aug 22. doi: 10.1001/jamapsychiatry.2018.2233.
Children and adolescents who moved longer distances or more frequently before 16 years of age were significantly more likely to develop psychosis in early adulthood than were those with less residential mobility, according to data from about 1.4 million children and adolescents in Sweden.
Data from previous studies have supported a link between childhood residential mobility and subsequent nonaffective psychoses, but no research has addressed the effects in later adolescence and young adulthood until now, wrote Ceri Price of Cardiff (Wales) University and colleagues.
In a study published in JAMA Psychiatry, the researchers reviewed data from a population-based cohort of individuals who were born in Sweden between Jan. 1, 1982, and Dec. 31, 1995, and lived in Sweden at age 16 years. The participants were followed from their 16th birthdays until a diagnosis of a nonaffective psychotic disorder, death, censorship because of emigration, or Dec. 31, 2011 – whichever came first.
Overall, the most sensitive range for an association between moving and psychosis was ages 16-19 years; the adjusted hazard ratio for a nonaffective psychotic disorder was 1.99 for participants who moved each year between ages 16 and 19 years, compared with those who never moved. In addition, moving greater distances before 16 years of age was independently associated with an increased risk of nonaffective psychosis (HR, 1.11) and the data suggested a nonlinear threshold effect when the distance moved exceeded 30 km.
A total of 4,537 individuals had a nonaffective psychotic disorder at a median 21 years of age, and a dose-response relationship emerged between more frequent moves and increased risk of nonaffective psychosis after controlling for confounding variables.
By contrast, a single move in young adulthood was not associated with increased psychosis risk, but moving at least four times during young adulthood was associated with an increased risk (adjusted HR, 1.82).
The study findings were strengthened by the longitudinal design and large population, but they were limited by several factors, including an absence of data on other adverse childhood experiences, such as family discord; peer relationships, such as friendships and bullying; and information on school changes and the disruption of peer relationships, the researchers wrote.
However, the results support the theory that psychosis risk can be affected by the disruption of social networks, peer support, and identity formation that occurs when children and adolescents move, and these results have potential implications for child health services and social policy, they noted.
“It is important that health, social, and educational practitioners ensure that children and adolescents who are newly resident to their neighborhoods receive adequate support to minimize the risks of adverse outcomes during adulthood, and every effort should be made to ensure the effective transfer of care for highly mobile children who are already in contact with health and social services,” they said.
The researchers had no financial conflicts to disclose. The study was supported in part by the Wellcome Trust and the Royal Society.
SOURCE: Price C et al. JAMA Psychiatry. 2018 Aug 22. doi: 10.1001/jamapsychiatry.2018.2233.
FROM JAMA PSYCHIATRY
Key clinical point: Clinicians and teachers should ensure that children and adolescents who are new to communities receive support “to minimize the risks of adverse outcomes during adulthood.”
Major finding: Those who moved residentially each year between 16 and 19 years of age were significantly more likely to develop nonaffective psychoses, compared with those who never moved (hazard ratio, 1.99).
Study details: The data come from a prospective cohort study of 1,440,383 youth living in Sweden.
Disclosures: The researchers had no financial conflicts to disclose. The study was supported in part by the Wellcome Trust and the Royal Society.
Source: Price C et al. JAMA Psychiatry. 2018 Aug 22. doi: 10.1001/jamapsychiatry.2018.2233.
Paliperidone palmitate: Adjusting dosing intervals and measuring serum concentrations
Mr. B, age 27, has a 10-year history of schizophrenia. Last year, he was doing well and working 4 hours/day 3 days/week while taking oral risperidone, 6 mg, at bedtime. However, during the past 2 weeks Mr. B began to have a return of auditory hallucinations and reports that he stopped taking his medication again 6 weeks ago.
As a result, he is started on paliperidone palmitate following the product label’s initiation dosing recommendation. On the first day he is given the first dose of the initiation regimen, 234 mg IM. One week later, the second dose of the initiation regimen, 156 mg IM, is given. One month later, the first maintenance dose of 117 mg IM every 28 days is given. All injections are in his deltoid muscle at his request.
After 3 weeks on the first maintenance dose of 117 mg, the voices begin to bother him again. Subsequently, Mr. B’s maintenance dose is increased first to 156 mg, and for the same problem with breakthrough hallucinations the following month to 234 mg, the maximum dose in the product label. After 6 months of receiving 234 mg IM every 28 days, the auditory hallucinations continue to bother him, but only for a few days prior to his next injection. He misses work 1 or 2 times before each injection.
Can the injection frequency for Mr. B’s paliperidone palmitate, 234 mg IM, in the deltoid muscle be increased to every 21 days to prevent the monthly exacerbations? Yes, the injection frequency can be increased, and doing so will increase the concentrations of paliperidone. The use of long-acting injectable antipsychotics (LAIs) is complicated by the lengthy time needed to reach steady state. In the case of paliperidone palmitate, an initiation regimen was developed that achieves therapeutic concentrations that are close to steady state before the oral antipsychotic’s effects are lost. This initiation strategy avoids the need for oral supplementation to maintain clinical efficacy. However, even using an initiation regimen or a loading dose does not decrease the time to final steady state after a dose adjustment due to the slow absorption of the medication from the injection site. The time to steady state is controlled by “flip-flop” pharmacokinetics. In this kind of pharmacokinetics, which is observed with all LAIs, the absorption rate from the injection site is lower than the elimination rate.1
Cleton et al2 reported the pharmacokinetics of paliperidone palmitate for deltoid and gluteal injection sites. By combining the median data for the deltoid injection route from the article by Cleton et al2 and the dosing from Mr. B’s case, I created a model using superposition of a sixth-degree polynomial fitted to the single dose data. Gluteal injections were not included because their increased complexity is beyond the scope of this article, but the time to maximum concentration (gluteal > deltoid) and peak concentration (deltoid > gluteal) are different for each route. The polynomial was a good fit with the adjusted r2 = 0.976, P < .0001. This model illustrates the paliperidone serum concentrations for Mr. B and is shown in the Figure. As you can see, by Day 9, the serum concentrations had reached the lower limit of the expected range of 20 to 60 ng/mL, shown in the shaded region of the Figure.3

Steady state at the routine maintenance dose of 117 mg every 28 days was never reached as the medication was not sufficient to suppress Mr. B’s hallucinations, and his doses needed to be increased each month. First, Mr. B’s dose was increased to 156 mg and then to the maximum recommended dose of 234 mg every 28 days. Steady state can be considered to have been achieved when 90% of the final steady state is reached after 3.3 half-lives. Because of the flip-flop pharmacokinetics, the important half-life is the absorption half-life of approximately 40 days or 132 days at the same dose. In Mr. B’s case, this was Day 221, where the trough concentration was 35 ng/mL. However, this regimen was still inadequate because he had breakthrough symptoms prior to the next injection.
By decreasing the injection interval from 28 days to 21 days, the concentrations will increase to a new steady state. This will take the same 132 days. With the reduced injection frequency of 21 days, 7 injections will have been given prior to reaching the new steady state. Steady state is not dependent on the number of injections, but only on the absorption half-life. This new steady state trough is substantially higher at 52 ng/mL, but still in the expected range for commonly used doses. Because Mr. B’s hallucinations only appeared at the end of the dosing interval, it is reasonable to expect that his new regimen would be successful in suppressing his hallucinations. However, monitoring for peak-related adverse effects is essential. Based upon controlled clinical trials, the potential dose-related adverse effects of paliperidone include akathisia, other extrapyramidal symptoms, weight gain, and QTc prolongation.
Continue to: Would monitoring a patient's paliperidone serum concentrations be useful?
Would monitoring a patient’s paliperidone serum concentrations be useful? Currently, measuring an individual’s paliperidone serum concentration is generally considered unwarranted.3,4 One of the major reasons is a lack of appropriately designed studies to determine a therapeutic range.5 Flexible dose designs, commonly used in registration studies, cloud the relationships between concentration, time, response, and adverse effects. There are additional problems that are the result of diagnostic heterogeneity and placebo responders. A well-designed study to determine the therapeutic range would have ≥1 fixed dose groups and be diagnostically homogeneous. There are currently only a limited number of clinical laboratories that have implemented suitable assays.
Given the lack of knowledge of a therapeutic range, assured knowledge of nonadherence to LAIs, and the absence of significant drug interactions for paliperidone, there remain a few reasonable justifications for obtaining a patient’s paliperidone serum concentration (Table). If the patient had a good response with mild adverse effects, there is no reason to obtain a paliperidone serum concentration or make any change in the medication or dose. However, if the patient had a good response accompanied by moderate or severe adverse effects, or the patient has a poor response, then obtaining the paliperidone serum concentration could help determine an appropriate course of action.

CASE CONTINUED
After the second dose at the increased frequency on Day 252, the paliperidone serum concentration was maintained above 40 ng/mL. Mr. B continued to tolerate the LAI well and no longer reported any breakthrough hallucinations.
Related Resources
- ARUP Laboratories. Paliperidone, serum or plasma. http://ltd.aruplab.com/tests/pub/2007949.
- LabCorp. Paliperidone Paliperidone (as 9-hydroxyrisperidone), serum or plasma. https://www.labcorp.com/test-menu/38351/paliperidone-as-9-hydroxyrisperidone-serum-or-plasma.
- Janssen Scientific Affairs. Educational dose illustrator. http://www.educationaldoseillustrator.com.
Drug Brand Names
Paliperidone palmitate • Invega Sustenna
Risperidone • Risperdal
1. Jann MW, Ereshefsky L, Saklad SR. Clinical pharmacokinetics of the depot antipsychotics. Clin Pharmacokinet. 1985;10(4):315-333.
2. Cleton A, Rossenu S, Crauwels H, et al. A single-dose, open-label, parallel, randomized, dose-proportionality study of paliperidone after intramuscular injections of paliperidone palmitate in the deltoid or gluteal muscle in patients with schizophrenia. J Clin Pharmacol. 2014;54(9):1048-1057.
3. Taylor D, Paton C, Kapur S. The Maudsley prescribing guidelines in psychiatry. 12th ed. Oxford, UK: John Wiley & Sons, Ltd.; 2015:1-10.
4. Hiemke C, Baumann P, Bergemann N, et al. AGNP consensus guidelines for therapeutic drug monitoring in psychiatry: update 2011. Pharmacopsychiatry. 2011;44(6):195-235.
5. Lopez LV, Kane JM. Plasma levels of second-generation antipsychotics and clinical response in acute psychosis: a review of the literature. Schizophr Res. 2013;147(2-3):368-374.
Mr. B, age 27, has a 10-year history of schizophrenia. Last year, he was doing well and working 4 hours/day 3 days/week while taking oral risperidone, 6 mg, at bedtime. However, during the past 2 weeks Mr. B began to have a return of auditory hallucinations and reports that he stopped taking his medication again 6 weeks ago.
As a result, he is started on paliperidone palmitate following the product label’s initiation dosing recommendation. On the first day he is given the first dose of the initiation regimen, 234 mg IM. One week later, the second dose of the initiation regimen, 156 mg IM, is given. One month later, the first maintenance dose of 117 mg IM every 28 days is given. All injections are in his deltoid muscle at his request.
After 3 weeks on the first maintenance dose of 117 mg, the voices begin to bother him again. Subsequently, Mr. B’s maintenance dose is increased first to 156 mg, and for the same problem with breakthrough hallucinations the following month to 234 mg, the maximum dose in the product label. After 6 months of receiving 234 mg IM every 28 days, the auditory hallucinations continue to bother him, but only for a few days prior to his next injection. He misses work 1 or 2 times before each injection.
Can the injection frequency for Mr. B’s paliperidone palmitate, 234 mg IM, in the deltoid muscle be increased to every 21 days to prevent the monthly exacerbations? Yes, the injection frequency can be increased, and doing so will increase the concentrations of paliperidone. The use of long-acting injectable antipsychotics (LAIs) is complicated by the lengthy time needed to reach steady state. In the case of paliperidone palmitate, an initiation regimen was developed that achieves therapeutic concentrations that are close to steady state before the oral antipsychotic’s effects are lost. This initiation strategy avoids the need for oral supplementation to maintain clinical efficacy. However, even using an initiation regimen or a loading dose does not decrease the time to final steady state after a dose adjustment due to the slow absorption of the medication from the injection site. The time to steady state is controlled by “flip-flop” pharmacokinetics. In this kind of pharmacokinetics, which is observed with all LAIs, the absorption rate from the injection site is lower than the elimination rate.1
Cleton et al2 reported the pharmacokinetics of paliperidone palmitate for deltoid and gluteal injection sites. By combining the median data for the deltoid injection route from the article by Cleton et al2 and the dosing from Mr. B’s case, I created a model using superposition of a sixth-degree polynomial fitted to the single dose data. Gluteal injections were not included because their increased complexity is beyond the scope of this article, but the time to maximum concentration (gluteal > deltoid) and peak concentration (deltoid > gluteal) are different for each route. The polynomial was a good fit with the adjusted r2 = 0.976, P < .0001. This model illustrates the paliperidone serum concentrations for Mr. B and is shown in the Figure. As you can see, by Day 9, the serum concentrations had reached the lower limit of the expected range of 20 to 60 ng/mL, shown in the shaded region of the Figure.3

Steady state at the routine maintenance dose of 117 mg every 28 days was never reached as the medication was not sufficient to suppress Mr. B’s hallucinations, and his doses needed to be increased each month. First, Mr. B’s dose was increased to 156 mg and then to the maximum recommended dose of 234 mg every 28 days. Steady state can be considered to have been achieved when 90% of the final steady state is reached after 3.3 half-lives. Because of the flip-flop pharmacokinetics, the important half-life is the absorption half-life of approximately 40 days or 132 days at the same dose. In Mr. B’s case, this was Day 221, where the trough concentration was 35 ng/mL. However, this regimen was still inadequate because he had breakthrough symptoms prior to the next injection.
By decreasing the injection interval from 28 days to 21 days, the concentrations will increase to a new steady state. This will take the same 132 days. With the reduced injection frequency of 21 days, 7 injections will have been given prior to reaching the new steady state. Steady state is not dependent on the number of injections, but only on the absorption half-life. This new steady state trough is substantially higher at 52 ng/mL, but still in the expected range for commonly used doses. Because Mr. B’s hallucinations only appeared at the end of the dosing interval, it is reasonable to expect that his new regimen would be successful in suppressing his hallucinations. However, monitoring for peak-related adverse effects is essential. Based upon controlled clinical trials, the potential dose-related adverse effects of paliperidone include akathisia, other extrapyramidal symptoms, weight gain, and QTc prolongation.
Continue to: Would monitoring a patient's paliperidone serum concentrations be useful?
Would monitoring a patient’s paliperidone serum concentrations be useful? Currently, measuring an individual’s paliperidone serum concentration is generally considered unwarranted.3,4 One of the major reasons is a lack of appropriately designed studies to determine a therapeutic range.5 Flexible dose designs, commonly used in registration studies, cloud the relationships between concentration, time, response, and adverse effects. There are additional problems that are the result of diagnostic heterogeneity and placebo responders. A well-designed study to determine the therapeutic range would have ≥1 fixed dose groups and be diagnostically homogeneous. There are currently only a limited number of clinical laboratories that have implemented suitable assays.
Given the lack of knowledge of a therapeutic range, assured knowledge of nonadherence to LAIs, and the absence of significant drug interactions for paliperidone, there remain a few reasonable justifications for obtaining a patient’s paliperidone serum concentration (Table). If the patient had a good response with mild adverse effects, there is no reason to obtain a paliperidone serum concentration or make any change in the medication or dose. However, if the patient had a good response accompanied by moderate or severe adverse effects, or the patient has a poor response, then obtaining the paliperidone serum concentration could help determine an appropriate course of action.

CASE CONTINUED
After the second dose at the increased frequency on Day 252, the paliperidone serum concentration was maintained above 40 ng/mL. Mr. B continued to tolerate the LAI well and no longer reported any breakthrough hallucinations.
Related Resources
- ARUP Laboratories. Paliperidone, serum or plasma. http://ltd.aruplab.com/tests/pub/2007949.
- LabCorp. Paliperidone Paliperidone (as 9-hydroxyrisperidone), serum or plasma. https://www.labcorp.com/test-menu/38351/paliperidone-as-9-hydroxyrisperidone-serum-or-plasma.
- Janssen Scientific Affairs. Educational dose illustrator. http://www.educationaldoseillustrator.com.
Drug Brand Names
Paliperidone palmitate • Invega Sustenna
Risperidone • Risperdal
Mr. B, age 27, has a 10-year history of schizophrenia. Last year, he was doing well and working 4 hours/day 3 days/week while taking oral risperidone, 6 mg, at bedtime. However, during the past 2 weeks Mr. B began to have a return of auditory hallucinations and reports that he stopped taking his medication again 6 weeks ago.
As a result, he is started on paliperidone palmitate following the product label’s initiation dosing recommendation. On the first day he is given the first dose of the initiation regimen, 234 mg IM. One week later, the second dose of the initiation regimen, 156 mg IM, is given. One month later, the first maintenance dose of 117 mg IM every 28 days is given. All injections are in his deltoid muscle at his request.
After 3 weeks on the first maintenance dose of 117 mg, the voices begin to bother him again. Subsequently, Mr. B’s maintenance dose is increased first to 156 mg, and for the same problem with breakthrough hallucinations the following month to 234 mg, the maximum dose in the product label. After 6 months of receiving 234 mg IM every 28 days, the auditory hallucinations continue to bother him, but only for a few days prior to his next injection. He misses work 1 or 2 times before each injection.
Can the injection frequency for Mr. B’s paliperidone palmitate, 234 mg IM, in the deltoid muscle be increased to every 21 days to prevent the monthly exacerbations? Yes, the injection frequency can be increased, and doing so will increase the concentrations of paliperidone. The use of long-acting injectable antipsychotics (LAIs) is complicated by the lengthy time needed to reach steady state. In the case of paliperidone palmitate, an initiation regimen was developed that achieves therapeutic concentrations that are close to steady state before the oral antipsychotic’s effects are lost. This initiation strategy avoids the need for oral supplementation to maintain clinical efficacy. However, even using an initiation regimen or a loading dose does not decrease the time to final steady state after a dose adjustment due to the slow absorption of the medication from the injection site. The time to steady state is controlled by “flip-flop” pharmacokinetics. In this kind of pharmacokinetics, which is observed with all LAIs, the absorption rate from the injection site is lower than the elimination rate.1
Cleton et al2 reported the pharmacokinetics of paliperidone palmitate for deltoid and gluteal injection sites. By combining the median data for the deltoid injection route from the article by Cleton et al2 and the dosing from Mr. B’s case, I created a model using superposition of a sixth-degree polynomial fitted to the single dose data. Gluteal injections were not included because their increased complexity is beyond the scope of this article, but the time to maximum concentration (gluteal > deltoid) and peak concentration (deltoid > gluteal) are different for each route. The polynomial was a good fit with the adjusted r2 = 0.976, P < .0001. This model illustrates the paliperidone serum concentrations for Mr. B and is shown in the Figure. As you can see, by Day 9, the serum concentrations had reached the lower limit of the expected range of 20 to 60 ng/mL, shown in the shaded region of the Figure.3

Steady state at the routine maintenance dose of 117 mg every 28 days was never reached as the medication was not sufficient to suppress Mr. B’s hallucinations, and his doses needed to be increased each month. First, Mr. B’s dose was increased to 156 mg and then to the maximum recommended dose of 234 mg every 28 days. Steady state can be considered to have been achieved when 90% of the final steady state is reached after 3.3 half-lives. Because of the flip-flop pharmacokinetics, the important half-life is the absorption half-life of approximately 40 days or 132 days at the same dose. In Mr. B’s case, this was Day 221, where the trough concentration was 35 ng/mL. However, this regimen was still inadequate because he had breakthrough symptoms prior to the next injection.
By decreasing the injection interval from 28 days to 21 days, the concentrations will increase to a new steady state. This will take the same 132 days. With the reduced injection frequency of 21 days, 7 injections will have been given prior to reaching the new steady state. Steady state is not dependent on the number of injections, but only on the absorption half-life. This new steady state trough is substantially higher at 52 ng/mL, but still in the expected range for commonly used doses. Because Mr. B’s hallucinations only appeared at the end of the dosing interval, it is reasonable to expect that his new regimen would be successful in suppressing his hallucinations. However, monitoring for peak-related adverse effects is essential. Based upon controlled clinical trials, the potential dose-related adverse effects of paliperidone include akathisia, other extrapyramidal symptoms, weight gain, and QTc prolongation.
Continue to: Would monitoring a patient's paliperidone serum concentrations be useful?
Would monitoring a patient’s paliperidone serum concentrations be useful? Currently, measuring an individual’s paliperidone serum concentration is generally considered unwarranted.3,4 One of the major reasons is a lack of appropriately designed studies to determine a therapeutic range.5 Flexible dose designs, commonly used in registration studies, cloud the relationships between concentration, time, response, and adverse effects. There are additional problems that are the result of diagnostic heterogeneity and placebo responders. A well-designed study to determine the therapeutic range would have ≥1 fixed dose groups and be diagnostically homogeneous. There are currently only a limited number of clinical laboratories that have implemented suitable assays.
Given the lack of knowledge of a therapeutic range, assured knowledge of nonadherence to LAIs, and the absence of significant drug interactions for paliperidone, there remain a few reasonable justifications for obtaining a patient’s paliperidone serum concentration (Table). If the patient had a good response with mild adverse effects, there is no reason to obtain a paliperidone serum concentration or make any change in the medication or dose. However, if the patient had a good response accompanied by moderate or severe adverse effects, or the patient has a poor response, then obtaining the paliperidone serum concentration could help determine an appropriate course of action.

CASE CONTINUED
After the second dose at the increased frequency on Day 252, the paliperidone serum concentration was maintained above 40 ng/mL. Mr. B continued to tolerate the LAI well and no longer reported any breakthrough hallucinations.
Related Resources
- ARUP Laboratories. Paliperidone, serum or plasma. http://ltd.aruplab.com/tests/pub/2007949.
- LabCorp. Paliperidone Paliperidone (as 9-hydroxyrisperidone), serum or plasma. https://www.labcorp.com/test-menu/38351/paliperidone-as-9-hydroxyrisperidone-serum-or-plasma.
- Janssen Scientific Affairs. Educational dose illustrator. http://www.educationaldoseillustrator.com.
Drug Brand Names
Paliperidone palmitate • Invega Sustenna
Risperidone • Risperdal
1. Jann MW, Ereshefsky L, Saklad SR. Clinical pharmacokinetics of the depot antipsychotics. Clin Pharmacokinet. 1985;10(4):315-333.
2. Cleton A, Rossenu S, Crauwels H, et al. A single-dose, open-label, parallel, randomized, dose-proportionality study of paliperidone after intramuscular injections of paliperidone palmitate in the deltoid or gluteal muscle in patients with schizophrenia. J Clin Pharmacol. 2014;54(9):1048-1057.
3. Taylor D, Paton C, Kapur S. The Maudsley prescribing guidelines in psychiatry. 12th ed. Oxford, UK: John Wiley & Sons, Ltd.; 2015:1-10.
4. Hiemke C, Baumann P, Bergemann N, et al. AGNP consensus guidelines for therapeutic drug monitoring in psychiatry: update 2011. Pharmacopsychiatry. 2011;44(6):195-235.
5. Lopez LV, Kane JM. Plasma levels of second-generation antipsychotics and clinical response in acute psychosis: a review of the literature. Schizophr Res. 2013;147(2-3):368-374.
1. Jann MW, Ereshefsky L, Saklad SR. Clinical pharmacokinetics of the depot antipsychotics. Clin Pharmacokinet. 1985;10(4):315-333.
2. Cleton A, Rossenu S, Crauwels H, et al. A single-dose, open-label, parallel, randomized, dose-proportionality study of paliperidone after intramuscular injections of paliperidone palmitate in the deltoid or gluteal muscle in patients with schizophrenia. J Clin Pharmacol. 2014;54(9):1048-1057.
3. Taylor D, Paton C, Kapur S. The Maudsley prescribing guidelines in psychiatry. 12th ed. Oxford, UK: John Wiley & Sons, Ltd.; 2015:1-10.
4. Hiemke C, Baumann P, Bergemann N, et al. AGNP consensus guidelines for therapeutic drug monitoring in psychiatry: update 2011. Pharmacopsychiatry. 2011;44(6):195-235.
5. Lopez LV, Kane JM. Plasma levels of second-generation antipsychotics and clinical response in acute psychosis: a review of the literature. Schizophr Res. 2013;147(2-3):368-374.
Address physical health risks of people with SMI
ROCKVILLE, MD. – The problem of medical comorbidities in people with serious mental illness (SMI) persists and must be addressed, researchers said at a National Institute on Mental Health conference on mental health services research. Part of that effort, they said, is a more careful consideration of risks tied to the off-label use of second-generation antipsychotics.
The researchers discussed strategies aimed at combating obesity and diabetes, as well as behaviors such as smoking and sedentary lifestyle.
“One of the things that jumps out is the tremendous need for evidence-based strategies to address these physical health problems that are common in general population but even more of a burden for people with serious mental illness,” said Susan T. Azrin, PhD, of the NIMH, in an interview.
A study published in 2015, estimated that people with schizophrenia, for example, might lose almost 30 years of life because of premature death. Individuals with serious mental illness also experience elevated morbidity from cardiovascular disease and cancer. The NIMH and other federal agencies have in recent years looked for ways to help people with SMI quit smoking, and better control their weight and cholesterol.
But approaches that sound promising for boosting physical fitness in this group of patients have not always proven successful. Joshua Breslau, PhD, ScD, of the Rand Corp. discussed findings from a 2014 paper where he and his colleagues reported somewhat disappointing results from a study of federal Primary and Behavioral Health Care Integration grants.
The researchers matched clinics receiving this funding with similar ones that did not. They found that people with mental illness treated at the clinics receiving the grants showed improvements in some indicators of physical health (diastolic blood pressure, total cholesterol, LDL cholesterol, and fasting plasma glucose) but not in others (systolic blood pressure, body mass index, HDL cholesterol, hemoglobin A1c, triglycerides, self-reported smoking). Dr. Breslau said he and his colleagues also found only limited benefits in quality of care for physical health conditions associated with the grant program. Still, he remains hopeful.
“There is some potential here,” Dr. Breslau said. “Sometimes, we are seeing positive effects, but it’s certainly not a slam-dunk.”
He noted that opening a new setting for primary care services could strain a workforce that’s already in short supply. In addition, he said, attempts to fold primary care services into mental health programs could, in some cases, result in replication of care of chronic conditions for certain patients with SMI.
We “may still not reach that portion of the target population that has the greatest need,” Dr. Breslau said in an interview. “The new services may turn out to be duplicative rather than filling a gap.”
In another session, Gail L. Daumit, MD, MHS, of Johns Hopkins University, Baltimore, discussed her plan to build on a past success in helping people with SMI lose weight.
In the ACHIEVE (Achieving Healthy Lifestyles In Psych Rehabilitation) trial, Dr. Daumit and her colleagues found that people enrolled in an intervention group lost an average of 3.2 kg more than did a control group after 18 months (N Engl J Med. 2013;368:1594-602). The intervention steps included alternating group and individual weight management sessions, on-site group physical activity three times weekly, and weigh-ins. The study had 291 patients who were randomized between the control and intervention groups.
that can be used more broadly. She’s seeking to scale up effective interventions to address cardiovascular risk factors in people with SMI.
“Our goal is not just to get process-of-care measures like ‘counseling was delivered,’ ‘a medicine was started,’ but to actually show impact on health outcomes,” Dr. Daumit said.
Risks tied to antipsychotics
People with SMI face cardiovascular risks not only from unhealthy behaviors but also from the medications used to treat their psychiatric conditions. The American Diabetes Association and American Psychiatric Association in 2004 released a consensus statement on the impact of antipsychotics such as clozapine, olanzapine, and risperidone on obesity and diabetes. It included guidelines for monitoring the metabolic status of patients both at baseline and after initiating treatment, including checking body mass index, waist circumference, blood pressure, fasting glucose, and fasting lipids.
Yet, substantial evidence suggests that the medical community still has not paid enough attention to the health risks of those medications, said Alisa Busch, MD, of Harvard Medical School, Boston.
“A slew of research has shown since then that we have done a very poor job in adhering to those monitoring guidelines,” Dr. Busch said.
A fellow panelist, Marcela Horvitz-Lennon, MD, MPH, of the Rand Corp., presented results from her study showing continued common use of second-generation antipsychotics for off-label use for treatment of anxiety, posttraumatic stress disorder, and dementia in people of all ages.
Consistent with previous research, Dr. Horvitz-Lennon and her colleagues found that off-label use of second-generation antipsychotics was common during 2008-2012 in the four states they studied. They looked at available data from fee-for-service Medicare, Medicaid, and dually (Medicaid-Medicare) covered adult beneficiaries in California, Georgia, Mississippi, and Oklahoma.
Throughout the study period, California had the highest rate of fee-for-service beneficiaries whose SGA use was consistently off label (44.6%). Georgia had the lowest rate of always off-label use (35.1%), while Mississippi (42%) and Oklahoma (36.3%) fell somewhere in the middle.
When second-generation antipsychotics have approved uses such as schizophrenia, the known profile of a medication gives some assurance that the benefit of the medications will exceed the risk for that patient, she said. “When the medication is used off label, the implication is that there is no good evidence that the benefits are there,” Dr. Horvitz-Lennon said. “Hence, the potential for harm is most likely exceeding that likely or unlikely benefit.”
Dr. Daumit, Dr. Breslau, and Dr. Horvitz-Lennon said they had no financial disclosures.
ROCKVILLE, MD. – The problem of medical comorbidities in people with serious mental illness (SMI) persists and must be addressed, researchers said at a National Institute on Mental Health conference on mental health services research. Part of that effort, they said, is a more careful consideration of risks tied to the off-label use of second-generation antipsychotics.
The researchers discussed strategies aimed at combating obesity and diabetes, as well as behaviors such as smoking and sedentary lifestyle.
“One of the things that jumps out is the tremendous need for evidence-based strategies to address these physical health problems that are common in general population but even more of a burden for people with serious mental illness,” said Susan T. Azrin, PhD, of the NIMH, in an interview.
A study published in 2015, estimated that people with schizophrenia, for example, might lose almost 30 years of life because of premature death. Individuals with serious mental illness also experience elevated morbidity from cardiovascular disease and cancer. The NIMH and other federal agencies have in recent years looked for ways to help people with SMI quit smoking, and better control their weight and cholesterol.
But approaches that sound promising for boosting physical fitness in this group of patients have not always proven successful. Joshua Breslau, PhD, ScD, of the Rand Corp. discussed findings from a 2014 paper where he and his colleagues reported somewhat disappointing results from a study of federal Primary and Behavioral Health Care Integration grants.
The researchers matched clinics receiving this funding with similar ones that did not. They found that people with mental illness treated at the clinics receiving the grants showed improvements in some indicators of physical health (diastolic blood pressure, total cholesterol, LDL cholesterol, and fasting plasma glucose) but not in others (systolic blood pressure, body mass index, HDL cholesterol, hemoglobin A1c, triglycerides, self-reported smoking). Dr. Breslau said he and his colleagues also found only limited benefits in quality of care for physical health conditions associated with the grant program. Still, he remains hopeful.
“There is some potential here,” Dr. Breslau said. “Sometimes, we are seeing positive effects, but it’s certainly not a slam-dunk.”
He noted that opening a new setting for primary care services could strain a workforce that’s already in short supply. In addition, he said, attempts to fold primary care services into mental health programs could, in some cases, result in replication of care of chronic conditions for certain patients with SMI.
We “may still not reach that portion of the target population that has the greatest need,” Dr. Breslau said in an interview. “The new services may turn out to be duplicative rather than filling a gap.”
In another session, Gail L. Daumit, MD, MHS, of Johns Hopkins University, Baltimore, discussed her plan to build on a past success in helping people with SMI lose weight.
In the ACHIEVE (Achieving Healthy Lifestyles In Psych Rehabilitation) trial, Dr. Daumit and her colleagues found that people enrolled in an intervention group lost an average of 3.2 kg more than did a control group after 18 months (N Engl J Med. 2013;368:1594-602). The intervention steps included alternating group and individual weight management sessions, on-site group physical activity three times weekly, and weigh-ins. The study had 291 patients who were randomized between the control and intervention groups.
that can be used more broadly. She’s seeking to scale up effective interventions to address cardiovascular risk factors in people with SMI.
“Our goal is not just to get process-of-care measures like ‘counseling was delivered,’ ‘a medicine was started,’ but to actually show impact on health outcomes,” Dr. Daumit said.
Risks tied to antipsychotics
People with SMI face cardiovascular risks not only from unhealthy behaviors but also from the medications used to treat their psychiatric conditions. The American Diabetes Association and American Psychiatric Association in 2004 released a consensus statement on the impact of antipsychotics such as clozapine, olanzapine, and risperidone on obesity and diabetes. It included guidelines for monitoring the metabolic status of patients both at baseline and after initiating treatment, including checking body mass index, waist circumference, blood pressure, fasting glucose, and fasting lipids.
Yet, substantial evidence suggests that the medical community still has not paid enough attention to the health risks of those medications, said Alisa Busch, MD, of Harvard Medical School, Boston.
“A slew of research has shown since then that we have done a very poor job in adhering to those monitoring guidelines,” Dr. Busch said.
A fellow panelist, Marcela Horvitz-Lennon, MD, MPH, of the Rand Corp., presented results from her study showing continued common use of second-generation antipsychotics for off-label use for treatment of anxiety, posttraumatic stress disorder, and dementia in people of all ages.
Consistent with previous research, Dr. Horvitz-Lennon and her colleagues found that off-label use of second-generation antipsychotics was common during 2008-2012 in the four states they studied. They looked at available data from fee-for-service Medicare, Medicaid, and dually (Medicaid-Medicare) covered adult beneficiaries in California, Georgia, Mississippi, and Oklahoma.
Throughout the study period, California had the highest rate of fee-for-service beneficiaries whose SGA use was consistently off label (44.6%). Georgia had the lowest rate of always off-label use (35.1%), while Mississippi (42%) and Oklahoma (36.3%) fell somewhere in the middle.
When second-generation antipsychotics have approved uses such as schizophrenia, the known profile of a medication gives some assurance that the benefit of the medications will exceed the risk for that patient, she said. “When the medication is used off label, the implication is that there is no good evidence that the benefits are there,” Dr. Horvitz-Lennon said. “Hence, the potential for harm is most likely exceeding that likely or unlikely benefit.”
Dr. Daumit, Dr. Breslau, and Dr. Horvitz-Lennon said they had no financial disclosures.
ROCKVILLE, MD. – The problem of medical comorbidities in people with serious mental illness (SMI) persists and must be addressed, researchers said at a National Institute on Mental Health conference on mental health services research. Part of that effort, they said, is a more careful consideration of risks tied to the off-label use of second-generation antipsychotics.
The researchers discussed strategies aimed at combating obesity and diabetes, as well as behaviors such as smoking and sedentary lifestyle.
“One of the things that jumps out is the tremendous need for evidence-based strategies to address these physical health problems that are common in general population but even more of a burden for people with serious mental illness,” said Susan T. Azrin, PhD, of the NIMH, in an interview.
A study published in 2015, estimated that people with schizophrenia, for example, might lose almost 30 years of life because of premature death. Individuals with serious mental illness also experience elevated morbidity from cardiovascular disease and cancer. The NIMH and other federal agencies have in recent years looked for ways to help people with SMI quit smoking, and better control their weight and cholesterol.
But approaches that sound promising for boosting physical fitness in this group of patients have not always proven successful. Joshua Breslau, PhD, ScD, of the Rand Corp. discussed findings from a 2014 paper where he and his colleagues reported somewhat disappointing results from a study of federal Primary and Behavioral Health Care Integration grants.
The researchers matched clinics receiving this funding with similar ones that did not. They found that people with mental illness treated at the clinics receiving the grants showed improvements in some indicators of physical health (diastolic blood pressure, total cholesterol, LDL cholesterol, and fasting plasma glucose) but not in others (systolic blood pressure, body mass index, HDL cholesterol, hemoglobin A1c, triglycerides, self-reported smoking). Dr. Breslau said he and his colleagues also found only limited benefits in quality of care for physical health conditions associated with the grant program. Still, he remains hopeful.
“There is some potential here,” Dr. Breslau said. “Sometimes, we are seeing positive effects, but it’s certainly not a slam-dunk.”
He noted that opening a new setting for primary care services could strain a workforce that’s already in short supply. In addition, he said, attempts to fold primary care services into mental health programs could, in some cases, result in replication of care of chronic conditions for certain patients with SMI.
We “may still not reach that portion of the target population that has the greatest need,” Dr. Breslau said in an interview. “The new services may turn out to be duplicative rather than filling a gap.”
In another session, Gail L. Daumit, MD, MHS, of Johns Hopkins University, Baltimore, discussed her plan to build on a past success in helping people with SMI lose weight.
In the ACHIEVE (Achieving Healthy Lifestyles In Psych Rehabilitation) trial, Dr. Daumit and her colleagues found that people enrolled in an intervention group lost an average of 3.2 kg more than did a control group after 18 months (N Engl J Med. 2013;368:1594-602). The intervention steps included alternating group and individual weight management sessions, on-site group physical activity three times weekly, and weigh-ins. The study had 291 patients who were randomized between the control and intervention groups.
that can be used more broadly. She’s seeking to scale up effective interventions to address cardiovascular risk factors in people with SMI.
“Our goal is not just to get process-of-care measures like ‘counseling was delivered,’ ‘a medicine was started,’ but to actually show impact on health outcomes,” Dr. Daumit said.
Risks tied to antipsychotics
People with SMI face cardiovascular risks not only from unhealthy behaviors but also from the medications used to treat their psychiatric conditions. The American Diabetes Association and American Psychiatric Association in 2004 released a consensus statement on the impact of antipsychotics such as clozapine, olanzapine, and risperidone on obesity and diabetes. It included guidelines for monitoring the metabolic status of patients both at baseline and after initiating treatment, including checking body mass index, waist circumference, blood pressure, fasting glucose, and fasting lipids.
Yet, substantial evidence suggests that the medical community still has not paid enough attention to the health risks of those medications, said Alisa Busch, MD, of Harvard Medical School, Boston.
“A slew of research has shown since then that we have done a very poor job in adhering to those monitoring guidelines,” Dr. Busch said.
A fellow panelist, Marcela Horvitz-Lennon, MD, MPH, of the Rand Corp., presented results from her study showing continued common use of second-generation antipsychotics for off-label use for treatment of anxiety, posttraumatic stress disorder, and dementia in people of all ages.
Consistent with previous research, Dr. Horvitz-Lennon and her colleagues found that off-label use of second-generation antipsychotics was common during 2008-2012 in the four states they studied. They looked at available data from fee-for-service Medicare, Medicaid, and dually (Medicaid-Medicare) covered adult beneficiaries in California, Georgia, Mississippi, and Oklahoma.
Throughout the study period, California had the highest rate of fee-for-service beneficiaries whose SGA use was consistently off label (44.6%). Georgia had the lowest rate of always off-label use (35.1%), while Mississippi (42%) and Oklahoma (36.3%) fell somewhere in the middle.
When second-generation antipsychotics have approved uses such as schizophrenia, the known profile of a medication gives some assurance that the benefit of the medications will exceed the risk for that patient, she said. “When the medication is used off label, the implication is that there is no good evidence that the benefits are there,” Dr. Horvitz-Lennon said. “Hence, the potential for harm is most likely exceeding that likely or unlikely benefit.”
Dr. Daumit, Dr. Breslau, and Dr. Horvitz-Lennon said they had no financial disclosures.
REPORTING FROM AN NIMH CONFERENCE
For people with SMI, disclosure still challenging
ROCKVILLE, MD. – People working in the mental health field are more likely to disclose their past or present treatment for psychosis than are professionals in other fields, a researcher said at a National Institute of Mental Health conference on mental health services research.
The researcher, Nev Jones, PhD, presented the findings of a small survey she conducted in 2014 and 2015 of adults with current or past experiences of psychotic disorder who described themselves as having had or having a successful career. The research was not conducted for publication purposes but as part of an effort to develop tools for students with psychosis as they continued in higher education, said Dr. Jones, of the department of mental health law and policy at the University of South Florida, Tampa.
One of those tools was a closed program that was akin to Facebook; people with early psychosis could use this to “look at successful adults across a wide range of careers and how they had navigated accommodations, disclosure, education as well as vocational choice,” she said.
Dr. Jones did not ask participants about their gender and race, but she did query them on highest degree earned. The poll was disseminated with the assistance of the National Alliance on Mental Illness and that of Stanford (Calif.) University, where Dr. Jones was a postdoctoral fellow. Of the sample presented, 33% had a masters degree (MSW, MBA), and 15% had a doctoral level degree (JD, MD, PhD).
People who worked in fields outside of mental health care were far less likely to have revealed their conditions to colleagues or employers, with 14 of 67 participants having made no disclosure. Of 14 who had made no disclosure, 12 were in fields such as banking, economics, secondary education, nursing, pediatrics, and computer programming.
Dr. Jones said she received several calls from students and staff at Stanford who were unwilling to fill out the survey.
“They were very concerned about the risks of inadvertent disclosure, even though it was anonymous, because they had unique, potentially identifiable career paths that they could not lay out in their responses without the fear that that would disclose [identify] them,” Dr. Jones said.
An additional 17 of the 67 participants made what Dr. Jones termed “selective disclosures,” such as telling a coworker who was considered a friend or a supportive boss. The majority of the respondents to Jones’s survey – 36 of the 67 participants – were open about their conditions. All but one of the respondents in this broad-disclosure group worked in mental health fields.
Dr. Jones described the broad-disclosure designation as “meaning that there is nobody in their life who doesn’t know.”
“They’re out professionally. They’ve published a book. They speak,” Dr. Jones said. “If you Google them on the Internet, you would quickly learn that they had a psychiatric disability or psychosis.”
Dr. Jones herself falls into that camp. She’s told media outlets, including the online newspaper MinnPost, about her own experience being diagnosed with schizophrenia while a PhD student. The online magazine Pacific Standard ran a full-length feature about her return to academia.
About 100,000 adolescents and young adults in the United States experience first-episode psychosis each year, and the peak onset hits between 15 and 25 years of age, according to the NIMH. About a decade ago, the NIMH launched its Recovery After an Initial Schizophrenia Episode (RAISE) initiative to examine use of coordinated specialty care treatments for people who were experiencing a first episode of psychosis. Congress in 2014 moved to provide a stream of federal funding for those kinds of efforts.
“We’re going to be soon starting to discharge, on an annual basis, potentially tens of thousands of young people from these specialized early intervention programs,” Dr. Jones said. “So it becomes really pressing to understand what’s happening to them in the context of reintegration.”
Another presenter at the panel, Marjorie L. Baldwin, PhD, of Arizona State University, Tempe, is an economist who has published a book based, in part, on her son’s struggles, “Beyond Schizophrenia: Living and Working With a Serious Mental Illness” (Lanham, Md.: Rowman & Littlefield Publishers, 2016).
She presented findings from a pilot study for a larger project looking at the issue of disclosures of serious mental illness in the workplace. She and her colleague in this work, Steven C. Marcus, PhD, of the University of Pennsylvania, Philadelphia, separately spoke about the difficulties in securing funding for the project, including five failed R01 grant applications.
“The 6th time was the charm with NIH,” Dr. Baldwin said.
An initial hurdle was finding a cost-effective way to identify workers with serious mental illness who hold or have held what she termed “competitive jobs,” which Dr. Baldwin described as those that paid at least minimum wage and are not subsidized for people with disabilities.
“You cannot do this kind of a study with random dialing because it would be way too expensive,” she said. “Schizophrenia and serious mental illnesses are not rare, but they are fairly uncommon.”
Several years ago, though, she learned of a long-running health survey into which she could “piggyback” questions on mental health status. She presented results of a pilot study with about 230 people with serious mental illness who had held or had competitive jobs. Of this group, 52% had left their most recent job for reasons other than mental illness, while those conditions had caused an additional 21% to leave. But Dr. Baldwin and her colleagues found 27% still working.
Like Dr. Jones, Dr. Baldwin said some of those workers were in professional fields, such as accounting, law, education; others worked in the service and construction industries.
“Contrary to the stereotypes, people with serious mental illness whose symptoms are reasonably well controlled can work, and many are capable of supporting themselves in mainstream competitive jobs,” Dr. Baldwin said.
Dr. Jones had no disclosures tied to her survey. Dr. Baldwin and Dr. Marcus had no disclosures other than the NIH R01 grant.
ROCKVILLE, MD. – People working in the mental health field are more likely to disclose their past or present treatment for psychosis than are professionals in other fields, a researcher said at a National Institute of Mental Health conference on mental health services research.
The researcher, Nev Jones, PhD, presented the findings of a small survey she conducted in 2014 and 2015 of adults with current or past experiences of psychotic disorder who described themselves as having had or having a successful career. The research was not conducted for publication purposes but as part of an effort to develop tools for students with psychosis as they continued in higher education, said Dr. Jones, of the department of mental health law and policy at the University of South Florida, Tampa.
One of those tools was a closed program that was akin to Facebook; people with early psychosis could use this to “look at successful adults across a wide range of careers and how they had navigated accommodations, disclosure, education as well as vocational choice,” she said.
Dr. Jones did not ask participants about their gender and race, but she did query them on highest degree earned. The poll was disseminated with the assistance of the National Alliance on Mental Illness and that of Stanford (Calif.) University, where Dr. Jones was a postdoctoral fellow. Of the sample presented, 33% had a masters degree (MSW, MBA), and 15% had a doctoral level degree (JD, MD, PhD).
People who worked in fields outside of mental health care were far less likely to have revealed their conditions to colleagues or employers, with 14 of 67 participants having made no disclosure. Of 14 who had made no disclosure, 12 were in fields such as banking, economics, secondary education, nursing, pediatrics, and computer programming.
Dr. Jones said she received several calls from students and staff at Stanford who were unwilling to fill out the survey.
“They were very concerned about the risks of inadvertent disclosure, even though it was anonymous, because they had unique, potentially identifiable career paths that they could not lay out in their responses without the fear that that would disclose [identify] them,” Dr. Jones said.
An additional 17 of the 67 participants made what Dr. Jones termed “selective disclosures,” such as telling a coworker who was considered a friend or a supportive boss. The majority of the respondents to Jones’s survey – 36 of the 67 participants – were open about their conditions. All but one of the respondents in this broad-disclosure group worked in mental health fields.
Dr. Jones described the broad-disclosure designation as “meaning that there is nobody in their life who doesn’t know.”
“They’re out professionally. They’ve published a book. They speak,” Dr. Jones said. “If you Google them on the Internet, you would quickly learn that they had a psychiatric disability or psychosis.”
Dr. Jones herself falls into that camp. She’s told media outlets, including the online newspaper MinnPost, about her own experience being diagnosed with schizophrenia while a PhD student. The online magazine Pacific Standard ran a full-length feature about her return to academia.
About 100,000 adolescents and young adults in the United States experience first-episode psychosis each year, and the peak onset hits between 15 and 25 years of age, according to the NIMH. About a decade ago, the NIMH launched its Recovery After an Initial Schizophrenia Episode (RAISE) initiative to examine use of coordinated specialty care treatments for people who were experiencing a first episode of psychosis. Congress in 2014 moved to provide a stream of federal funding for those kinds of efforts.
“We’re going to be soon starting to discharge, on an annual basis, potentially tens of thousands of young people from these specialized early intervention programs,” Dr. Jones said. “So it becomes really pressing to understand what’s happening to them in the context of reintegration.”
Another presenter at the panel, Marjorie L. Baldwin, PhD, of Arizona State University, Tempe, is an economist who has published a book based, in part, on her son’s struggles, “Beyond Schizophrenia: Living and Working With a Serious Mental Illness” (Lanham, Md.: Rowman & Littlefield Publishers, 2016).
She presented findings from a pilot study for a larger project looking at the issue of disclosures of serious mental illness in the workplace. She and her colleague in this work, Steven C. Marcus, PhD, of the University of Pennsylvania, Philadelphia, separately spoke about the difficulties in securing funding for the project, including five failed R01 grant applications.
“The 6th time was the charm with NIH,” Dr. Baldwin said.
An initial hurdle was finding a cost-effective way to identify workers with serious mental illness who hold or have held what she termed “competitive jobs,” which Dr. Baldwin described as those that paid at least minimum wage and are not subsidized for people with disabilities.
“You cannot do this kind of a study with random dialing because it would be way too expensive,” she said. “Schizophrenia and serious mental illnesses are not rare, but they are fairly uncommon.”
Several years ago, though, she learned of a long-running health survey into which she could “piggyback” questions on mental health status. She presented results of a pilot study with about 230 people with serious mental illness who had held or had competitive jobs. Of this group, 52% had left their most recent job for reasons other than mental illness, while those conditions had caused an additional 21% to leave. But Dr. Baldwin and her colleagues found 27% still working.
Like Dr. Jones, Dr. Baldwin said some of those workers were in professional fields, such as accounting, law, education; others worked in the service and construction industries.
“Contrary to the stereotypes, people with serious mental illness whose symptoms are reasonably well controlled can work, and many are capable of supporting themselves in mainstream competitive jobs,” Dr. Baldwin said.
Dr. Jones had no disclosures tied to her survey. Dr. Baldwin and Dr. Marcus had no disclosures other than the NIH R01 grant.
ROCKVILLE, MD. – People working in the mental health field are more likely to disclose their past or present treatment for psychosis than are professionals in other fields, a researcher said at a National Institute of Mental Health conference on mental health services research.
The researcher, Nev Jones, PhD, presented the findings of a small survey she conducted in 2014 and 2015 of adults with current or past experiences of psychotic disorder who described themselves as having had or having a successful career. The research was not conducted for publication purposes but as part of an effort to develop tools for students with psychosis as they continued in higher education, said Dr. Jones, of the department of mental health law and policy at the University of South Florida, Tampa.
One of those tools was a closed program that was akin to Facebook; people with early psychosis could use this to “look at successful adults across a wide range of careers and how they had navigated accommodations, disclosure, education as well as vocational choice,” she said.
Dr. Jones did not ask participants about their gender and race, but she did query them on highest degree earned. The poll was disseminated with the assistance of the National Alliance on Mental Illness and that of Stanford (Calif.) University, where Dr. Jones was a postdoctoral fellow. Of the sample presented, 33% had a masters degree (MSW, MBA), and 15% had a doctoral level degree (JD, MD, PhD).
People who worked in fields outside of mental health care were far less likely to have revealed their conditions to colleagues or employers, with 14 of 67 participants having made no disclosure. Of 14 who had made no disclosure, 12 were in fields such as banking, economics, secondary education, nursing, pediatrics, and computer programming.
Dr. Jones said she received several calls from students and staff at Stanford who were unwilling to fill out the survey.
“They were very concerned about the risks of inadvertent disclosure, even though it was anonymous, because they had unique, potentially identifiable career paths that they could not lay out in their responses without the fear that that would disclose [identify] them,” Dr. Jones said.
An additional 17 of the 67 participants made what Dr. Jones termed “selective disclosures,” such as telling a coworker who was considered a friend or a supportive boss. The majority of the respondents to Jones’s survey – 36 of the 67 participants – were open about their conditions. All but one of the respondents in this broad-disclosure group worked in mental health fields.
Dr. Jones described the broad-disclosure designation as “meaning that there is nobody in their life who doesn’t know.”
“They’re out professionally. They’ve published a book. They speak,” Dr. Jones said. “If you Google them on the Internet, you would quickly learn that they had a psychiatric disability or psychosis.”
Dr. Jones herself falls into that camp. She’s told media outlets, including the online newspaper MinnPost, about her own experience being diagnosed with schizophrenia while a PhD student. The online magazine Pacific Standard ran a full-length feature about her return to academia.
About 100,000 adolescents and young adults in the United States experience first-episode psychosis each year, and the peak onset hits between 15 and 25 years of age, according to the NIMH. About a decade ago, the NIMH launched its Recovery After an Initial Schizophrenia Episode (RAISE) initiative to examine use of coordinated specialty care treatments for people who were experiencing a first episode of psychosis. Congress in 2014 moved to provide a stream of federal funding for those kinds of efforts.
“We’re going to be soon starting to discharge, on an annual basis, potentially tens of thousands of young people from these specialized early intervention programs,” Dr. Jones said. “So it becomes really pressing to understand what’s happening to them in the context of reintegration.”
Another presenter at the panel, Marjorie L. Baldwin, PhD, of Arizona State University, Tempe, is an economist who has published a book based, in part, on her son’s struggles, “Beyond Schizophrenia: Living and Working With a Serious Mental Illness” (Lanham, Md.: Rowman & Littlefield Publishers, 2016).
She presented findings from a pilot study for a larger project looking at the issue of disclosures of serious mental illness in the workplace. She and her colleague in this work, Steven C. Marcus, PhD, of the University of Pennsylvania, Philadelphia, separately spoke about the difficulties in securing funding for the project, including five failed R01 grant applications.
“The 6th time was the charm with NIH,” Dr. Baldwin said.
An initial hurdle was finding a cost-effective way to identify workers with serious mental illness who hold or have held what she termed “competitive jobs,” which Dr. Baldwin described as those that paid at least minimum wage and are not subsidized for people with disabilities.
“You cannot do this kind of a study with random dialing because it would be way too expensive,” she said. “Schizophrenia and serious mental illnesses are not rare, but they are fairly uncommon.”
Several years ago, though, she learned of a long-running health survey into which she could “piggyback” questions on mental health status. She presented results of a pilot study with about 230 people with serious mental illness who had held or had competitive jobs. Of this group, 52% had left their most recent job for reasons other than mental illness, while those conditions had caused an additional 21% to leave. But Dr. Baldwin and her colleagues found 27% still working.
Like Dr. Jones, Dr. Baldwin said some of those workers were in professional fields, such as accounting, law, education; others worked in the service and construction industries.
“Contrary to the stereotypes, people with serious mental illness whose symptoms are reasonably well controlled can work, and many are capable of supporting themselves in mainstream competitive jobs,” Dr. Baldwin said.
Dr. Jones had no disclosures tied to her survey. Dr. Baldwin and Dr. Marcus had no disclosures other than the NIH R01 grant.
EXPERT ANALYSIS FROM AN NIMH CONFERENCE
Clozapine-induced GI hypomotility: From constipation to bowel obstruction
Patients who are treated with clozapine—a second-generation antipsychotic approved for treatment-resistant schizophrenia—require monitoring for serious adverse effects. Many of these adverse effects, such as agranulocytosis or seizures, are familiar to clinicians; however, gastrointestinal (GI) hypomotility is not always recognized as a potentially serious adverse effect, even though it is one of the most common causes for hospital admission.1 Its manifestations range from being relatively benign (nausea, vomiting, constipation) to potentially severe (fecal impaction) or even life-threatening (bowel obstruction, ileus, toxic megacolon).2
GI hypomotility is caused by clozapine’s strong anticholinergic properties, which lead to slowed smooth muscle contractions and delayed bowel transit time. It is further compounded by clozapine’s 5-HT3 antagonism, which is also known to slow bowel transit time. To avoid the potentially serious risks associated with GI hypomotility, we offer simple approaches for clinicians to follow when treating patients with clozapine.
Before starting a patient on clozapine, and at all subsequent visits, ask him or her about bowel habits and GI symptoms. Because the onset of GI hypomotility can be subtle, ask patients to pay close attention to their bowel habits and keep a diary to document GI symptoms and bowel movements. Signs of bowel obstruction can include an inability to pass stool, nausea, vomiting, abdominal pain, and a bloated abdomen. Staff who care for patients taking clozapine who live in a supervised setting should be educated about the relevance of a patient’s changing bowel habits or GI complaints. Also, teach patients about simple lifestyle modifications they can make to counteract constipation, including increased physical activity, adequate hydration, and consuming a fiber-rich diet.
If possible, avoid prescribing anticholinergic medications to a patient receiving clozapine because such agents may add to clozapine’s anticholinergic load. Some patients may require medical management of chronic constipation with stool softeners and stimulant laxatives. The Table outlines a typical bowel regimen we use for patients at our clinic. Some clinicians may prefer earlier and more regular use of senna glycoside. Also, patients might need a referral to their primary care physician if prevention has failed and fecal impaction requires enemas or mechanical disimpaction. An urgent referral to the emergency department is needed if a patient has a suspected bowel obstruction.
Acknowledgment
The authors thank Travis Baggett, MD, Department of Medicine, Massachusetts General Hospital, Boston, Massachusetts, for reviewing the medical management of constipation.
1. Stroup TS, Gerhard T, Crystal S, et al. Comparative effectiveness of clozapine and standard antipsychotic treatment in adults with schizophrenia. Am J Psychiatry. 2016;173(2):166-173.
2. Every-Palmer S, Ellis PM. Clozapine-induced gastrointestinal hypomotility: a 22-year bi-national pharmacovigilance study of serious or fatal ‘slow gut’ reactions, and comparison with international drug safety advice. CNS Drugs. 2017;31(8):699-709.
Patients who are treated with clozapine—a second-generation antipsychotic approved for treatment-resistant schizophrenia—require monitoring for serious adverse effects. Many of these adverse effects, such as agranulocytosis or seizures, are familiar to clinicians; however, gastrointestinal (GI) hypomotility is not always recognized as a potentially serious adverse effect, even though it is one of the most common causes for hospital admission.1 Its manifestations range from being relatively benign (nausea, vomiting, constipation) to potentially severe (fecal impaction) or even life-threatening (bowel obstruction, ileus, toxic megacolon).2
GI hypomotility is caused by clozapine’s strong anticholinergic properties, which lead to slowed smooth muscle contractions and delayed bowel transit time. It is further compounded by clozapine’s 5-HT3 antagonism, which is also known to slow bowel transit time. To avoid the potentially serious risks associated with GI hypomotility, we offer simple approaches for clinicians to follow when treating patients with clozapine.
Before starting a patient on clozapine, and at all subsequent visits, ask him or her about bowel habits and GI symptoms. Because the onset of GI hypomotility can be subtle, ask patients to pay close attention to their bowel habits and keep a diary to document GI symptoms and bowel movements. Signs of bowel obstruction can include an inability to pass stool, nausea, vomiting, abdominal pain, and a bloated abdomen. Staff who care for patients taking clozapine who live in a supervised setting should be educated about the relevance of a patient’s changing bowel habits or GI complaints. Also, teach patients about simple lifestyle modifications they can make to counteract constipation, including increased physical activity, adequate hydration, and consuming a fiber-rich diet.
If possible, avoid prescribing anticholinergic medications to a patient receiving clozapine because such agents may add to clozapine’s anticholinergic load. Some patients may require medical management of chronic constipation with stool softeners and stimulant laxatives. The Table outlines a typical bowel regimen we use for patients at our clinic. Some clinicians may prefer earlier and more regular use of senna glycoside. Also, patients might need a referral to their primary care physician if prevention has failed and fecal impaction requires enemas or mechanical disimpaction. An urgent referral to the emergency department is needed if a patient has a suspected bowel obstruction.
Acknowledgment
The authors thank Travis Baggett, MD, Department of Medicine, Massachusetts General Hospital, Boston, Massachusetts, for reviewing the medical management of constipation.
Patients who are treated with clozapine—a second-generation antipsychotic approved for treatment-resistant schizophrenia—require monitoring for serious adverse effects. Many of these adverse effects, such as agranulocytosis or seizures, are familiar to clinicians; however, gastrointestinal (GI) hypomotility is not always recognized as a potentially serious adverse effect, even though it is one of the most common causes for hospital admission.1 Its manifestations range from being relatively benign (nausea, vomiting, constipation) to potentially severe (fecal impaction) or even life-threatening (bowel obstruction, ileus, toxic megacolon).2
GI hypomotility is caused by clozapine’s strong anticholinergic properties, which lead to slowed smooth muscle contractions and delayed bowel transit time. It is further compounded by clozapine’s 5-HT3 antagonism, which is also known to slow bowel transit time. To avoid the potentially serious risks associated with GI hypomotility, we offer simple approaches for clinicians to follow when treating patients with clozapine.
Before starting a patient on clozapine, and at all subsequent visits, ask him or her about bowel habits and GI symptoms. Because the onset of GI hypomotility can be subtle, ask patients to pay close attention to their bowel habits and keep a diary to document GI symptoms and bowel movements. Signs of bowel obstruction can include an inability to pass stool, nausea, vomiting, abdominal pain, and a bloated abdomen. Staff who care for patients taking clozapine who live in a supervised setting should be educated about the relevance of a patient’s changing bowel habits or GI complaints. Also, teach patients about simple lifestyle modifications they can make to counteract constipation, including increased physical activity, adequate hydration, and consuming a fiber-rich diet.
If possible, avoid prescribing anticholinergic medications to a patient receiving clozapine because such agents may add to clozapine’s anticholinergic load. Some patients may require medical management of chronic constipation with stool softeners and stimulant laxatives. The Table outlines a typical bowel regimen we use for patients at our clinic. Some clinicians may prefer earlier and more regular use of senna glycoside. Also, patients might need a referral to their primary care physician if prevention has failed and fecal impaction requires enemas or mechanical disimpaction. An urgent referral to the emergency department is needed if a patient has a suspected bowel obstruction.
Acknowledgment
The authors thank Travis Baggett, MD, Department of Medicine, Massachusetts General Hospital, Boston, Massachusetts, for reviewing the medical management of constipation.
1. Stroup TS, Gerhard T, Crystal S, et al. Comparative effectiveness of clozapine and standard antipsychotic treatment in adults with schizophrenia. Am J Psychiatry. 2016;173(2):166-173.
2. Every-Palmer S, Ellis PM. Clozapine-induced gastrointestinal hypomotility: a 22-year bi-national pharmacovigilance study of serious or fatal ‘slow gut’ reactions, and comparison with international drug safety advice. CNS Drugs. 2017;31(8):699-709.
1. Stroup TS, Gerhard T, Crystal S, et al. Comparative effectiveness of clozapine and standard antipsychotic treatment in adults with schizophrenia. Am J Psychiatry. 2016;173(2):166-173.
2. Every-Palmer S, Ellis PM. Clozapine-induced gastrointestinal hypomotility: a 22-year bi-national pharmacovigilance study of serious or fatal ‘slow gut’ reactions, and comparison with international drug safety advice. CNS Drugs. 2017;31(8):699-709.
FAST and RAPID: Acronyms to prevent brain damage in stroke and psychosis
Psychosis and stroke are arguably the most serious acute threats to the integrity of the brain, and consequently the mind. Both are unquestionably associated with a grave outcome the longer their treatment is delayed.1,2
While the management of stroke has been elevated to the highest emergent priority because of the progressively deleterious impact of thrombotic ischemia in one of the cerebral arteries, rapid intervention for acute psychosis has never been regarded as an urgent neurologic condition with severe threats to the brain’s structure and function.3,4 There is extensive literature on the serious consequences of a long duration of untreated psychosis (DUP), including treatment resistance, frequent re-hospitalizations, more negative symptoms, and greater disability.5
Physical paralysis from a stroke receives much more attention than “mental paralysis” of psychosis. Both must be rapidly treated, whether for the regional ischemia to brain tissue following a stroke or for the neurotoxicity of neuroinflammation and oxidative stress that lead to widespread neurodegeneration during psychosis.6 While an acronym for the quick recognition of a stroke (FAST: Facial drooping, Arm weakness, Speech difficulties, and Time to call emergency services) is well established, no acronym for the urgency to treat psychosis has been developed. We propose the acronym RAPID (Readily Avoid Psychosis-Induced Damage). The acronym RAPID would hopefully expedite the urgently needed pharmacotherapeutic and psychosocial intervention in psychosis to halt ongoing brain tissue loss.
It is ironic that the legal obstacles for immediate treatment, which do not exist for stroke, often delay administering antipsychotic medication to patients with anosognosia (a neurologic delusional belief that one is not ill, leading to refusal of treatment) for their psychosis and end up harming patients by prolonging their DUP until a court order is obtained to force brain-saving treatment. Frequent psychotic relapses due to nonadherence with medications are also a very common cause for prolonged DUP due to the inexplicable reluctance of some psychiatric practitioners to employ a long-acting injectable antipsychotic (LAI) medication as soon as possible after the onset of psychosis to circumvent subsequent relapses due to the very high risk of poor adherence. Table 1 describes the optimal management of both stroke and acute psychosis once they are rapidly diagnosed, thanks to the FAST and RAPID reminders.
Tragically, the treatments of the mind have become falsely disengaged from the brain, the physical organ whose neurons, electrical impulses, synapses, and neurotransmitters generate the mind with its advanced human functions, such as self-awareness, will, thoughts, mood, speech, executive functions, memories, and social cognition. Recent editorials have challenged psychiatric practitioners to behave like cardiologists7 and oncologists8 by aggressively treating first-episode psychosis to prevent ongoing neurodegeneration due to recurrences. The brain loses 1% of its brain volume (~11 ml) after the first psychotic episode,8 which represents hundreds of millions of cells, billions of synapses, and substantial myelin. A second psychotic episode causes significant additional neuropil and white matter fiber damage and represents a different stage of schizophrenia9 with more severe tissue loss and disruption of neural pathways that trigger the process of treatment resistance and functional disability. Ensuring adherence with LAI antipsychotic formulations immediately after the first psychotic episode may allow many patients with schizophrenia to achieve a relapse-free remission and to return to their baseline functioning.10
In addition to significant brain tissue loss during psychotic episodes, mortality is also a very high risk following discharge from the first hospitalization for psychosis.11 LAI second-generation antipsychotic medications have been shown to be associated with lower mortality and neuroprotective effects,12 compared with oral or injectable first-generation antipsychotics. The highest mortality rate was reported to be associated with the lack of any antipsychotic medication,12 underscoring how untreated psychosis can be fatal.
Bottom line: Rapid treatment of stroke and psychosis is an absolute imperative for minimizing brain damage that respectively leads to physical or mental disability. The acronyms FAST and RAPID are essential reminders of the urgency needed to halt progressive neurodegeneration in those 2 devastating acute threats to the integrity of brain and mind. Intensive physical, psychological, and social rehabilitation must follow the acute treatment of stroke and psychosis, and the prevention of any recurrence is an absolute must. For psychosis, the use of a LAI second-generation antipsychotic before hospital discharge from a first episode of psychosis can be disease-modifying, with a more benign illness trajectory and outcome than the devastating deterioration that follows repetitive psychotic relapse, most often due to nonadherence with oral medications.
Continue to: Psychosis should be conceptualized as...
Psychosis should be conceptualized as a “stroke of the mind,” and it can be prevented in most patients with schizophrenia by adopting injectable antipsychotics as early after the onset of psychosis as possible. Yet, starting a LAI antipsychotic drug in first-episode psychosis before hospital discharge is rarely done, and the few patients who currently receive LAIs (10% of U.S. patients) generally receive them after multiple episodes and a protracted DUP. That’s like calling the fire department when much of the house has turned to ashes, instead of calling them when the first small flame is noticed. It makes so much sense, but the decades-old practice of postponing the use of LAIs continues to ruin the lives of young persons in the prime of life. By changing our practice habits to early use of LAIs, we have nothing to lose and our patients with psychosis may be spared a lifetime of suffering, poverty, stigma, incarceration, and functional disability. Wouldn’t we want to avoid that atrocious outcome for our own family members if they develop schizophrenia?
1. Benjamin EJ, Virani SS, Callaway CW, et al; American Heart Association Council on Epidemiology and Prevention Statistics Committee and Stroke Statistics Subcommittee. Heart Disease and Stroke Statistics-2018 Update: a report from the American Heart Association. Circulation. 2018;137(12):e67-e492.
2. Cechnicki A, Cichocki Ł, Kalisz A, et al. Duration of untreated psychosis (DUP) and the course of schizophrenia in a 20-year follow-up study. Psychiatry Res. 2014;219(3):420-425.
3. Davis J, Moylan S, Harvey BH, et al. Neuroprogression in schizophrenia: pathways underpinning clinical staging and therapeutic corollaries. Aust N Z J Psychiatry. 2014;48(6):512-529.
4. Olabi B, Ellison-Wright I, McIntosh AM, et al. Are there progressive brain changes in schizophrenia? A meta-analysis of structural magnetic resonance imaging studies. Biol Psychiatry. 2011;70(1):88-96.
5. Kane JM, Robinson DG, Schooler NR, et al. Comprehensive versus usual community care for first-episode psychosis: 2-year outcomes from the NIMH RAISE Early Treatment Program. Am J Psychiatry. 2016;173(4):362-372.
6. Najjar S, Pearlman DM. Neuroinflammation and white matter pathology in schizophrenia: systematic review. Schizophr Res. 2015;161(1):102-112.
7. Nasrallah HA. For first-episode psychosis, psychiatrists should behave like cardiologists. Current Psychiatry. 2017;16(8):4-7.
8. Cahn W, Hulshoff Pol HE, Lems EB, et al. Brain volume changes in first-episode schizophrenia: a 1-year follow-up study. Arch Gen Psychiatry. 2002;59(11):1002-1010.
9. McGorry P, Nelson B. Why we need a transdiagnostic staging approach to emerging psychopathology, early diagnosis, and treatment. JAMA Psychiatry. 2016;73(3):191-192.
10. Emsley R, Oosthuizen P, Koen L, et al. Remission in patients with first-episode schizophrenia receiving assured antipsychotic medication: a study with risperidone long-acting injection. Int Clin Psychopharmacol. 2008;23(6):325-331.
11. Nasrallah HA. The crisis of poor physical health and early mortality of psychiatric patients. Current Psychiatry. 2018;17(4):7-8,11.
12. Nasrallah HA. Triple advantages of injectable long acting second generation antipsychotics: relapse prevention, neuroprotection, and lower mortality. Schizophr Res. 2018;197:69-70.
Psychosis and stroke are arguably the most serious acute threats to the integrity of the brain, and consequently the mind. Both are unquestionably associated with a grave outcome the longer their treatment is delayed.1,2
While the management of stroke has been elevated to the highest emergent priority because of the progressively deleterious impact of thrombotic ischemia in one of the cerebral arteries, rapid intervention for acute psychosis has never been regarded as an urgent neurologic condition with severe threats to the brain’s structure and function.3,4 There is extensive literature on the serious consequences of a long duration of untreated psychosis (DUP), including treatment resistance, frequent re-hospitalizations, more negative symptoms, and greater disability.5
Physical paralysis from a stroke receives much more attention than “mental paralysis” of psychosis. Both must be rapidly treated, whether for the regional ischemia to brain tissue following a stroke or for the neurotoxicity of neuroinflammation and oxidative stress that lead to widespread neurodegeneration during psychosis.6 While an acronym for the quick recognition of a stroke (FAST: Facial drooping, Arm weakness, Speech difficulties, and Time to call emergency services) is well established, no acronym for the urgency to treat psychosis has been developed. We propose the acronym RAPID (Readily Avoid Psychosis-Induced Damage). The acronym RAPID would hopefully expedite the urgently needed pharmacotherapeutic and psychosocial intervention in psychosis to halt ongoing brain tissue loss.
It is ironic that the legal obstacles for immediate treatment, which do not exist for stroke, often delay administering antipsychotic medication to patients with anosognosia (a neurologic delusional belief that one is not ill, leading to refusal of treatment) for their psychosis and end up harming patients by prolonging their DUP until a court order is obtained to force brain-saving treatment. Frequent psychotic relapses due to nonadherence with medications are also a very common cause for prolonged DUP due to the inexplicable reluctance of some psychiatric practitioners to employ a long-acting injectable antipsychotic (LAI) medication as soon as possible after the onset of psychosis to circumvent subsequent relapses due to the very high risk of poor adherence. Table 1 describes the optimal management of both stroke and acute psychosis once they are rapidly diagnosed, thanks to the FAST and RAPID reminders.

Tragically, the treatments of the mind have become falsely disengaged from the brain, the physical organ whose neurons, electrical impulses, synapses, and neurotransmitters generate the mind with its advanced human functions, such as self-awareness, will, thoughts, mood, speech, executive functions, memories, and social cognition. Recent editorials have challenged psychiatric practitioners to behave like cardiologists7 and oncologists8 by aggressively treating first-episode psychosis to prevent ongoing neurodegeneration due to recurrences. The brain loses 1% of its brain volume (~11 ml) after the first psychotic episode,8 which represents hundreds of millions of cells, billions of synapses, and substantial myelin. A second psychotic episode causes significant additional neuropil and white matter fiber damage and represents a different stage of schizophrenia9 with more severe tissue loss and disruption of neural pathways that trigger the process of treatment resistance and functional disability. Ensuring adherence with LAI antipsychotic formulations immediately after the first psychotic episode may allow many patients with schizophrenia to achieve a relapse-free remission and to return to their baseline functioning.10
In addition to significant brain tissue loss during psychotic episodes, mortality is also a very high risk following discharge from the first hospitalization for psychosis.11 LAI second-generation antipsychotic medications have been shown to be associated with lower mortality and neuroprotective effects,12 compared with oral or injectable first-generation antipsychotics. The highest mortality rate was reported to be associated with the lack of any antipsychotic medication,12 underscoring how untreated psychosis can be fatal.
Bottom line: Rapid treatment of stroke and psychosis is an absolute imperative for minimizing brain damage that respectively leads to physical or mental disability. The acronyms FAST and RAPID are essential reminders of the urgency needed to halt progressive neurodegeneration in those 2 devastating acute threats to the integrity of brain and mind. Intensive physical, psychological, and social rehabilitation must follow the acute treatment of stroke and psychosis, and the prevention of any recurrence is an absolute must. For psychosis, the use of a LAI second-generation antipsychotic before hospital discharge from a first episode of psychosis can be disease-modifying, with a more benign illness trajectory and outcome than the devastating deterioration that follows repetitive psychotic relapse, most often due to nonadherence with oral medications.
Continue to: Psychosis should be conceptualized as...
Psychosis should be conceptualized as a “stroke of the mind,” and it can be prevented in most patients with schizophrenia by adopting injectable antipsychotics as early after the onset of psychosis as possible. Yet, starting a LAI antipsychotic drug in first-episode psychosis before hospital discharge is rarely done, and the few patients who currently receive LAIs (10% of U.S. patients) generally receive them after multiple episodes and a protracted DUP. That’s like calling the fire department when much of the house has turned to ashes, instead of calling them when the first small flame is noticed. It makes so much sense, but the decades-old practice of postponing the use of LAIs continues to ruin the lives of young persons in the prime of life. By changing our practice habits to early use of LAIs, we have nothing to lose and our patients with psychosis may be spared a lifetime of suffering, poverty, stigma, incarceration, and functional disability. Wouldn’t we want to avoid that atrocious outcome for our own family members if they develop schizophrenia?
Psychosis and stroke are arguably the most serious acute threats to the integrity of the brain, and consequently the mind. Both are unquestionably associated with a grave outcome the longer their treatment is delayed.1,2
While the management of stroke has been elevated to the highest emergent priority because of the progressively deleterious impact of thrombotic ischemia in one of the cerebral arteries, rapid intervention for acute psychosis has never been regarded as an urgent neurologic condition with severe threats to the brain’s structure and function.3,4 There is extensive literature on the serious consequences of a long duration of untreated psychosis (DUP), including treatment resistance, frequent re-hospitalizations, more negative symptoms, and greater disability.5
Physical paralysis from a stroke receives much more attention than “mental paralysis” of psychosis. Both must be rapidly treated, whether for the regional ischemia to brain tissue following a stroke or for the neurotoxicity of neuroinflammation and oxidative stress that lead to widespread neurodegeneration during psychosis.6 While an acronym for the quick recognition of a stroke (FAST: Facial drooping, Arm weakness, Speech difficulties, and Time to call emergency services) is well established, no acronym for the urgency to treat psychosis has been developed. We propose the acronym RAPID (Readily Avoid Psychosis-Induced Damage). The acronym RAPID would hopefully expedite the urgently needed pharmacotherapeutic and psychosocial intervention in psychosis to halt ongoing brain tissue loss.
It is ironic that the legal obstacles for immediate treatment, which do not exist for stroke, often delay administering antipsychotic medication to patients with anosognosia (a neurologic delusional belief that one is not ill, leading to refusal of treatment) for their psychosis and end up harming patients by prolonging their DUP until a court order is obtained to force brain-saving treatment. Frequent psychotic relapses due to nonadherence with medications are also a very common cause for prolonged DUP due to the inexplicable reluctance of some psychiatric practitioners to employ a long-acting injectable antipsychotic (LAI) medication as soon as possible after the onset of psychosis to circumvent subsequent relapses due to the very high risk of poor adherence. Table 1 describes the optimal management of both stroke and acute psychosis once they are rapidly diagnosed, thanks to the FAST and RAPID reminders.

Tragically, the treatments of the mind have become falsely disengaged from the brain, the physical organ whose neurons, electrical impulses, synapses, and neurotransmitters generate the mind with its advanced human functions, such as self-awareness, will, thoughts, mood, speech, executive functions, memories, and social cognition. Recent editorials have challenged psychiatric practitioners to behave like cardiologists7 and oncologists8 by aggressively treating first-episode psychosis to prevent ongoing neurodegeneration due to recurrences. The brain loses 1% of its brain volume (~11 ml) after the first psychotic episode,8 which represents hundreds of millions of cells, billions of synapses, and substantial myelin. A second psychotic episode causes significant additional neuropil and white matter fiber damage and represents a different stage of schizophrenia9 with more severe tissue loss and disruption of neural pathways that trigger the process of treatment resistance and functional disability. Ensuring adherence with LAI antipsychotic formulations immediately after the first psychotic episode may allow many patients with schizophrenia to achieve a relapse-free remission and to return to their baseline functioning.10
In addition to significant brain tissue loss during psychotic episodes, mortality is also a very high risk following discharge from the first hospitalization for psychosis.11 LAI second-generation antipsychotic medications have been shown to be associated with lower mortality and neuroprotective effects,12 compared with oral or injectable first-generation antipsychotics. The highest mortality rate was reported to be associated with the lack of any antipsychotic medication,12 underscoring how untreated psychosis can be fatal.
Bottom line: Rapid treatment of stroke and psychosis is an absolute imperative for minimizing brain damage that respectively leads to physical or mental disability. The acronyms FAST and RAPID are essential reminders of the urgency needed to halt progressive neurodegeneration in those 2 devastating acute threats to the integrity of brain and mind. Intensive physical, psychological, and social rehabilitation must follow the acute treatment of stroke and psychosis, and the prevention of any recurrence is an absolute must. For psychosis, the use of a LAI second-generation antipsychotic before hospital discharge from a first episode of psychosis can be disease-modifying, with a more benign illness trajectory and outcome than the devastating deterioration that follows repetitive psychotic relapse, most often due to nonadherence with oral medications.
Continue to: Psychosis should be conceptualized as...
Psychosis should be conceptualized as a “stroke of the mind,” and it can be prevented in most patients with schizophrenia by adopting injectable antipsychotics as early after the onset of psychosis as possible. Yet, starting a LAI antipsychotic drug in first-episode psychosis before hospital discharge is rarely done, and the few patients who currently receive LAIs (10% of U.S. patients) generally receive them after multiple episodes and a protracted DUP. That’s like calling the fire department when much of the house has turned to ashes, instead of calling them when the first small flame is noticed. It makes so much sense, but the decades-old practice of postponing the use of LAIs continues to ruin the lives of young persons in the prime of life. By changing our practice habits to early use of LAIs, we have nothing to lose and our patients with psychosis may be spared a lifetime of suffering, poverty, stigma, incarceration, and functional disability. Wouldn’t we want to avoid that atrocious outcome for our own family members if they develop schizophrenia?
1. Benjamin EJ, Virani SS, Callaway CW, et al; American Heart Association Council on Epidemiology and Prevention Statistics Committee and Stroke Statistics Subcommittee. Heart Disease and Stroke Statistics-2018 Update: a report from the American Heart Association. Circulation. 2018;137(12):e67-e492.
2. Cechnicki A, Cichocki Ł, Kalisz A, et al. Duration of untreated psychosis (DUP) and the course of schizophrenia in a 20-year follow-up study. Psychiatry Res. 2014;219(3):420-425.
3. Davis J, Moylan S, Harvey BH, et al. Neuroprogression in schizophrenia: pathways underpinning clinical staging and therapeutic corollaries. Aust N Z J Psychiatry. 2014;48(6):512-529.
4. Olabi B, Ellison-Wright I, McIntosh AM, et al. Are there progressive brain changes in schizophrenia? A meta-analysis of structural magnetic resonance imaging studies. Biol Psychiatry. 2011;70(1):88-96.
5. Kane JM, Robinson DG, Schooler NR, et al. Comprehensive versus usual community care for first-episode psychosis: 2-year outcomes from the NIMH RAISE Early Treatment Program. Am J Psychiatry. 2016;173(4):362-372.
6. Najjar S, Pearlman DM. Neuroinflammation and white matter pathology in schizophrenia: systematic review. Schizophr Res. 2015;161(1):102-112.
7. Nasrallah HA. For first-episode psychosis, psychiatrists should behave like cardiologists. Current Psychiatry. 2017;16(8):4-7.
8. Cahn W, Hulshoff Pol HE, Lems EB, et al. Brain volume changes in first-episode schizophrenia: a 1-year follow-up study. Arch Gen Psychiatry. 2002;59(11):1002-1010.
9. McGorry P, Nelson B. Why we need a transdiagnostic staging approach to emerging psychopathology, early diagnosis, and treatment. JAMA Psychiatry. 2016;73(3):191-192.
10. Emsley R, Oosthuizen P, Koen L, et al. Remission in patients with first-episode schizophrenia receiving assured antipsychotic medication: a study with risperidone long-acting injection. Int Clin Psychopharmacol. 2008;23(6):325-331.
11. Nasrallah HA. The crisis of poor physical health and early mortality of psychiatric patients. Current Psychiatry. 2018;17(4):7-8,11.
12. Nasrallah HA. Triple advantages of injectable long acting second generation antipsychotics: relapse prevention, neuroprotection, and lower mortality. Schizophr Res. 2018;197:69-70.
1. Benjamin EJ, Virani SS, Callaway CW, et al; American Heart Association Council on Epidemiology and Prevention Statistics Committee and Stroke Statistics Subcommittee. Heart Disease and Stroke Statistics-2018 Update: a report from the American Heart Association. Circulation. 2018;137(12):e67-e492.
2. Cechnicki A, Cichocki Ł, Kalisz A, et al. Duration of untreated psychosis (DUP) and the course of schizophrenia in a 20-year follow-up study. Psychiatry Res. 2014;219(3):420-425.
3. Davis J, Moylan S, Harvey BH, et al. Neuroprogression in schizophrenia: pathways underpinning clinical staging and therapeutic corollaries. Aust N Z J Psychiatry. 2014;48(6):512-529.
4. Olabi B, Ellison-Wright I, McIntosh AM, et al. Are there progressive brain changes in schizophrenia? A meta-analysis of structural magnetic resonance imaging studies. Biol Psychiatry. 2011;70(1):88-96.
5. Kane JM, Robinson DG, Schooler NR, et al. Comprehensive versus usual community care for first-episode psychosis: 2-year outcomes from the NIMH RAISE Early Treatment Program. Am J Psychiatry. 2016;173(4):362-372.
6. Najjar S, Pearlman DM. Neuroinflammation and white matter pathology in schizophrenia: systematic review. Schizophr Res. 2015;161(1):102-112.
7. Nasrallah HA. For first-episode psychosis, psychiatrists should behave like cardiologists. Current Psychiatry. 2017;16(8):4-7.
8. Cahn W, Hulshoff Pol HE, Lems EB, et al. Brain volume changes in first-episode schizophrenia: a 1-year follow-up study. Arch Gen Psychiatry. 2002;59(11):1002-1010.
9. McGorry P, Nelson B. Why we need a transdiagnostic staging approach to emerging psychopathology, early diagnosis, and treatment. JAMA Psychiatry. 2016;73(3):191-192.
10. Emsley R, Oosthuizen P, Koen L, et al. Remission in patients with first-episode schizophrenia receiving assured antipsychotic medication: a study with risperidone long-acting injection. Int Clin Psychopharmacol. 2008;23(6):325-331.
11. Nasrallah HA. The crisis of poor physical health and early mortality of psychiatric patients. Current Psychiatry. 2018;17(4):7-8,11.
12. Nasrallah HA. Triple advantages of injectable long acting second generation antipsychotics: relapse prevention, neuroprotection, and lower mortality. Schizophr Res. 2018;197:69-70.
Judge seeks replication of efforts to support people with SMI
Florida’s Miami-Dade County reportedly has the largest percentage of residents with serious mental illnesses (SMI) among large U.S. communities. And a Florida judge who helped develop approaches aimed at sparing his state’s residents with mental illness from harmful, avoidable, and expensive bouts of prison time wants to see his strategies replicated.
“There is something terribly wrong with a society that is willing to spend more money to incarcerate people who are ill than to treat them,” Judge Steve Leifman of the 11th judicial circuit court said at a National Institute of Mental Health conference on mental health services research.
Judge Leifman in 2000 created the Criminal Mental Health Project. It’s been recognized for its success in keeping people with SMI from becoming ensnared in the criminal justice system because of minor offenses. It also helps those who do spend time in jail from returning.
In Miami-Dade County, for example, 97 people were a significant driver of costs in the criminal justice system in a study that was completed in 2010, Judge Leifman said. The members of this group were largely men who suffered from schizophrenia spectrum disorders and were homeless with a co-occurring disorder. Combined, the number of arrests for this group was about 2,200 over a 5-year period, Judge Leifman said. They spent 27,000 days in the Miami-Dade County jail – costing taxpayers about $13.7 million.
Judge Leifman said. “They would have had a shot at an education. They would have had housing. They probably would have done a lot better.”
Through the Criminal Mental Health Project, Judge Leifman and his colleagues seek to both prevent people with mental illness from being arrested and jailed for minor offenses, and to provide a support network for those who have reached jail. The project’s “prebooking diversion” efforts are built on a model developed in Memphis, Tenn., in the late 1980s. Through it, police officers get special training in recognizing mental illness and resolving crises in which people who have these disorders are involved.
The project’s “postbooking diversion” techniques require participants to voluntarily consent to mental health treatment and services. The program is open only to those less serious felonies, which can include drug charges and theft. Through participation in the Criminal Mental Health Project, people can have charges dismissed or reduced. The program provides them with connections to community-based treatment, support, and housing services, according to its website.
Participants in the program who were charged with minor felonies had 75% fewer jail bookings and jail days after enrolling in the Criminal Mental Health Project (N Engl J Med. 2016;374:1701-3).
Judge Leifman said the postbooking jail diversion program has, since 2001, served more than 4,000 individuals. Recidivism rates among participants charged with misdemeanors dropped from roughly 75% to 20%, he said.
Still, Judge Leifman describes his role as a judge as making him a “gatekeeper to the largest psychiatric facility in Florida – the Miami-Dade County Jail.” The jail houses about 1,200 people with serious mental illness on any given day, according to the Criminal Mental Health Project’s website.
Judge Leifman said that, ultimately, he wants more communities to devote more resources to providing medical care for people with mental illness.
Florida’s Miami-Dade County reportedly has the largest percentage of residents with serious mental illnesses (SMI) among large U.S. communities. And a Florida judge who helped develop approaches aimed at sparing his state’s residents with mental illness from harmful, avoidable, and expensive bouts of prison time wants to see his strategies replicated.
“There is something terribly wrong with a society that is willing to spend more money to incarcerate people who are ill than to treat them,” Judge Steve Leifman of the 11th judicial circuit court said at a National Institute of Mental Health conference on mental health services research.
Judge Leifman in 2000 created the Criminal Mental Health Project. It’s been recognized for its success in keeping people with SMI from becoming ensnared in the criminal justice system because of minor offenses. It also helps those who do spend time in jail from returning.
In Miami-Dade County, for example, 97 people were a significant driver of costs in the criminal justice system in a study that was completed in 2010, Judge Leifman said. The members of this group were largely men who suffered from schizophrenia spectrum disorders and were homeless with a co-occurring disorder. Combined, the number of arrests for this group was about 2,200 over a 5-year period, Judge Leifman said. They spent 27,000 days in the Miami-Dade County jail – costing taxpayers about $13.7 million.
Judge Leifman said. “They would have had a shot at an education. They would have had housing. They probably would have done a lot better.”
Through the Criminal Mental Health Project, Judge Leifman and his colleagues seek to both prevent people with mental illness from being arrested and jailed for minor offenses, and to provide a support network for those who have reached jail. The project’s “prebooking diversion” efforts are built on a model developed in Memphis, Tenn., in the late 1980s. Through it, police officers get special training in recognizing mental illness and resolving crises in which people who have these disorders are involved.
The project’s “postbooking diversion” techniques require participants to voluntarily consent to mental health treatment and services. The program is open only to those less serious felonies, which can include drug charges and theft. Through participation in the Criminal Mental Health Project, people can have charges dismissed or reduced. The program provides them with connections to community-based treatment, support, and housing services, according to its website.
Participants in the program who were charged with minor felonies had 75% fewer jail bookings and jail days after enrolling in the Criminal Mental Health Project (N Engl J Med. 2016;374:1701-3).
Judge Leifman said the postbooking jail diversion program has, since 2001, served more than 4,000 individuals. Recidivism rates among participants charged with misdemeanors dropped from roughly 75% to 20%, he said.
Still, Judge Leifman describes his role as a judge as making him a “gatekeeper to the largest psychiatric facility in Florida – the Miami-Dade County Jail.” The jail houses about 1,200 people with serious mental illness on any given day, according to the Criminal Mental Health Project’s website.
Judge Leifman said that, ultimately, he wants more communities to devote more resources to providing medical care for people with mental illness.
Florida’s Miami-Dade County reportedly has the largest percentage of residents with serious mental illnesses (SMI) among large U.S. communities. And a Florida judge who helped develop approaches aimed at sparing his state’s residents with mental illness from harmful, avoidable, and expensive bouts of prison time wants to see his strategies replicated.
“There is something terribly wrong with a society that is willing to spend more money to incarcerate people who are ill than to treat them,” Judge Steve Leifman of the 11th judicial circuit court said at a National Institute of Mental Health conference on mental health services research.
Judge Leifman in 2000 created the Criminal Mental Health Project. It’s been recognized for its success in keeping people with SMI from becoming ensnared in the criminal justice system because of minor offenses. It also helps those who do spend time in jail from returning.
In Miami-Dade County, for example, 97 people were a significant driver of costs in the criminal justice system in a study that was completed in 2010, Judge Leifman said. The members of this group were largely men who suffered from schizophrenia spectrum disorders and were homeless with a co-occurring disorder. Combined, the number of arrests for this group was about 2,200 over a 5-year period, Judge Leifman said. They spent 27,000 days in the Miami-Dade County jail – costing taxpayers about $13.7 million.
Judge Leifman said. “They would have had a shot at an education. They would have had housing. They probably would have done a lot better.”
Through the Criminal Mental Health Project, Judge Leifman and his colleagues seek to both prevent people with mental illness from being arrested and jailed for minor offenses, and to provide a support network for those who have reached jail. The project’s “prebooking diversion” efforts are built on a model developed in Memphis, Tenn., in the late 1980s. Through it, police officers get special training in recognizing mental illness and resolving crises in which people who have these disorders are involved.
The project’s “postbooking diversion” techniques require participants to voluntarily consent to mental health treatment and services. The program is open only to those less serious felonies, which can include drug charges and theft. Through participation in the Criminal Mental Health Project, people can have charges dismissed or reduced. The program provides them with connections to community-based treatment, support, and housing services, according to its website.
Participants in the program who were charged with minor felonies had 75% fewer jail bookings and jail days after enrolling in the Criminal Mental Health Project (N Engl J Med. 2016;374:1701-3).
Judge Leifman said the postbooking jail diversion program has, since 2001, served more than 4,000 individuals. Recidivism rates among participants charged with misdemeanors dropped from roughly 75% to 20%, he said.
Still, Judge Leifman describes his role as a judge as making him a “gatekeeper to the largest psychiatric facility in Florida – the Miami-Dade County Jail.” The jail houses about 1,200 people with serious mental illness on any given day, according to the Criminal Mental Health Project’s website.
Judge Leifman said that, ultimately, he wants more communities to devote more resources to providing medical care for people with mental illness.
REPORTING FROM AN NIMH CONFERENCE