User login
Schizophrenia patients not getting secondary cardiovascular prevention
Secondary prevention of cardiovascular disease could significantly reduce cardiac mortality among patients with schizophrenia, yet is underused, a study published Oct. 24 has found.
The retrospective study, which included a Danish nationwide cohort of 105,018 patients with myocardial infarction, including 684 patients with schizophrenia, showed that individuals with schizophrenia who did not receive secondary cardioprotective treatment had a more than eight times higher mortality, compared with people in the general population who did receive treatment (hazard ratio, 8.78; 95% confidence interval, 4.37-17.64), Pirathiv Kugathasan and his associates reported in JAMA Psychiatry.
In contrast, the investigators found, patients with schizophrenia who received cardioprotective treatment had a 97% higher mortality (HR, 1.97; 95% CI, 1.25-310), compared with the treated general population, which was not statistically different from individuals in the general population who did not receive treatment (HR, 2.95; 95% CI, 2.62-3.32) after adjustment for baseline characteristics.
“Given the increased cardiovascular risk among patients with schizophrenia, we believe that the current findings support the use of intensive cardioprotective treatments in patients with schizophrenia,” reported Mr. Kugathasan, a PhD candidate in the department of psychiatry at Aalborg University Hospital in Denmark.
However, 7.8% of patients diagnosed with schizophrenia received no prescriptions for cardioprotective medications after a myocardial infarction, compared with 3.3% of the general population. They were significantly less likely than were individuals from the general population to receive a prescription for antiplatelets (84.9% vs. 91.8%), vitamin K antagonists (15.9% vs. 24.2%), beta-blockers (74.1% vs. 84.9%), ACE inhibitors (70.9% vs. 86.6%), and statins (72.2% vs. 87.3%).
The mortality rates were not significantly different between untreated patients with schizophrenia and untreated participants from the general population.
When the researchers examined the effects of different treatment types, they found that mortality rates were still higher in treated patients with schizophrenia, compared with treated patients from the general population – with the exception of those treated with antiplatelets and statins. For ACE inhibitors, treated patients with schizophrenia had a twofold higher mortality than that of treated patients from the general population, while the mortality was more than twofold higher in the case of vitamin K antagonists.
“Previous studies have found that patients with schizophrenia have increased cardiovascular mortality and a lower prescription rate for cardioprotective treatment compared with the general population,” the authors wrote.
They noted that patients with schizophrenia might have more problems with medication adherence, and pointed to another study showing this in patients with schizophrenia and diabetes. Other studies also showed that patients with schizophrenia were less likely to talk to a cardiologist after a cardiac event.
“Together, ,” they wrote. “This hypothesis could explain the results of a generally increased mortality rate among patients with schizophrenia, especially when the results suggest that these patients die of cardiovascular causes that might be treatable.”
They called for patients with schizophrenia to be followed up during treatment, and to increase treatment intensity after cardiac events, “because a diagnosis of schizophrenia may be associated with an increased cardiac risk, which potentially can be countered by secondary preventive cardiac treatment.”
In patients who received triple therapy, the mortality rates were similar for those with schizophrenia and the general population. However, there were much higher mortality rate differences between patients with schizophrenia and the general population for those receiving dual or monotherapy.
Significantly more patients from the general population were readmitted with myocardial infarction and more underwent percutaneous coronary intervention than from the group of patients with schizophrenia.
The prevalence of hypertension was similar between the two populations, but more patients with schizophrenia had diabetes and substance abuse, compared with the general population.
The authors noted that they did not have information on the severity of myocardial infarction or the effect of other lifestyle factors.
“Future research should attempt to assess the degree to which these factors contribute to the increased mortality in patients with schizophrenia,” they wrote.
One author declared grants, speaking fees and advisory roles with the pharmaceutical industry. No other conflicts of interest were declared.
SOURCE: Kugathasan P et al. JAMA Psychiatry 2018. Oct 24. doi: 10.1001/jamapsychiatry.2018.2742.
Individuals with schizophrenia are known to have higher mortality compared with the general population, yet the mechanisms underlying this are poorly understood. This study suggests the poor quality of cardiovascular care might play a role.
The outcomes of this study show the dire consequences of failing to provide care that addresses both disability and mortality among individuals with comorbid psychiatric and medical conditions. We need better coordination between mental health clinicians and medical specialists, and between inpatient and outpatient services.
Despite the high quality of health care in Denmark, these individuals are being left behind, and work is needed to develop policy and programs to enable patients with schizophrenia to enjoy an equal share in medical advances aimed at improving health and longevity.
Benjamin G. Druss, MD, MPH, is affiliated with the Rollins School of Public Health at Emory University in Atlanta. These comments are taken from an accompanying editorial (JAMA Psychiatry. 2018 Oct 24. doi: 10.1001/jamapsychiatry.2018.2726). He reported no conflicts of interest.
Individuals with schizophrenia are known to have higher mortality compared with the general population, yet the mechanisms underlying this are poorly understood. This study suggests the poor quality of cardiovascular care might play a role.
The outcomes of this study show the dire consequences of failing to provide care that addresses both disability and mortality among individuals with comorbid psychiatric and medical conditions. We need better coordination between mental health clinicians and medical specialists, and between inpatient and outpatient services.
Despite the high quality of health care in Denmark, these individuals are being left behind, and work is needed to develop policy and programs to enable patients with schizophrenia to enjoy an equal share in medical advances aimed at improving health and longevity.
Benjamin G. Druss, MD, MPH, is affiliated with the Rollins School of Public Health at Emory University in Atlanta. These comments are taken from an accompanying editorial (JAMA Psychiatry. 2018 Oct 24. doi: 10.1001/jamapsychiatry.2018.2726). He reported no conflicts of interest.
Individuals with schizophrenia are known to have higher mortality compared with the general population, yet the mechanisms underlying this are poorly understood. This study suggests the poor quality of cardiovascular care might play a role.
The outcomes of this study show the dire consequences of failing to provide care that addresses both disability and mortality among individuals with comorbid psychiatric and medical conditions. We need better coordination between mental health clinicians and medical specialists, and between inpatient and outpatient services.
Despite the high quality of health care in Denmark, these individuals are being left behind, and work is needed to develop policy and programs to enable patients with schizophrenia to enjoy an equal share in medical advances aimed at improving health and longevity.
Benjamin G. Druss, MD, MPH, is affiliated with the Rollins School of Public Health at Emory University in Atlanta. These comments are taken from an accompanying editorial (JAMA Psychiatry. 2018 Oct 24. doi: 10.1001/jamapsychiatry.2018.2726). He reported no conflicts of interest.
Secondary prevention of cardiovascular disease could significantly reduce cardiac mortality among patients with schizophrenia, yet is underused, a study published Oct. 24 has found.
The retrospective study, which included a Danish nationwide cohort of 105,018 patients with myocardial infarction, including 684 patients with schizophrenia, showed that individuals with schizophrenia who did not receive secondary cardioprotective treatment had a more than eight times higher mortality, compared with people in the general population who did receive treatment (hazard ratio, 8.78; 95% confidence interval, 4.37-17.64), Pirathiv Kugathasan and his associates reported in JAMA Psychiatry.
In contrast, the investigators found, patients with schizophrenia who received cardioprotective treatment had a 97% higher mortality (HR, 1.97; 95% CI, 1.25-310), compared with the treated general population, which was not statistically different from individuals in the general population who did not receive treatment (HR, 2.95; 95% CI, 2.62-3.32) after adjustment for baseline characteristics.
“Given the increased cardiovascular risk among patients with schizophrenia, we believe that the current findings support the use of intensive cardioprotective treatments in patients with schizophrenia,” reported Mr. Kugathasan, a PhD candidate in the department of psychiatry at Aalborg University Hospital in Denmark.
However, 7.8% of patients diagnosed with schizophrenia received no prescriptions for cardioprotective medications after a myocardial infarction, compared with 3.3% of the general population. They were significantly less likely than were individuals from the general population to receive a prescription for antiplatelets (84.9% vs. 91.8%), vitamin K antagonists (15.9% vs. 24.2%), beta-blockers (74.1% vs. 84.9%), ACE inhibitors (70.9% vs. 86.6%), and statins (72.2% vs. 87.3%).
The mortality rates were not significantly different between untreated patients with schizophrenia and untreated participants from the general population.
When the researchers examined the effects of different treatment types, they found that mortality rates were still higher in treated patients with schizophrenia, compared with treated patients from the general population – with the exception of those treated with antiplatelets and statins. For ACE inhibitors, treated patients with schizophrenia had a twofold higher mortality than that of treated patients from the general population, while the mortality was more than twofold higher in the case of vitamin K antagonists.
“Previous studies have found that patients with schizophrenia have increased cardiovascular mortality and a lower prescription rate for cardioprotective treatment compared with the general population,” the authors wrote.
They noted that patients with schizophrenia might have more problems with medication adherence, and pointed to another study showing this in patients with schizophrenia and diabetes. Other studies also showed that patients with schizophrenia were less likely to talk to a cardiologist after a cardiac event.
“Together, ,” they wrote. “This hypothesis could explain the results of a generally increased mortality rate among patients with schizophrenia, especially when the results suggest that these patients die of cardiovascular causes that might be treatable.”
They called for patients with schizophrenia to be followed up during treatment, and to increase treatment intensity after cardiac events, “because a diagnosis of schizophrenia may be associated with an increased cardiac risk, which potentially can be countered by secondary preventive cardiac treatment.”
In patients who received triple therapy, the mortality rates were similar for those with schizophrenia and the general population. However, there were much higher mortality rate differences between patients with schizophrenia and the general population for those receiving dual or monotherapy.
Significantly more patients from the general population were readmitted with myocardial infarction and more underwent percutaneous coronary intervention than from the group of patients with schizophrenia.
The prevalence of hypertension was similar between the two populations, but more patients with schizophrenia had diabetes and substance abuse, compared with the general population.
The authors noted that they did not have information on the severity of myocardial infarction or the effect of other lifestyle factors.
“Future research should attempt to assess the degree to which these factors contribute to the increased mortality in patients with schizophrenia,” they wrote.
One author declared grants, speaking fees and advisory roles with the pharmaceutical industry. No other conflicts of interest were declared.
SOURCE: Kugathasan P et al. JAMA Psychiatry 2018. Oct 24. doi: 10.1001/jamapsychiatry.2018.2742.
Secondary prevention of cardiovascular disease could significantly reduce cardiac mortality among patients with schizophrenia, yet is underused, a study published Oct. 24 has found.
The retrospective study, which included a Danish nationwide cohort of 105,018 patients with myocardial infarction, including 684 patients with schizophrenia, showed that individuals with schizophrenia who did not receive secondary cardioprotective treatment had a more than eight times higher mortality, compared with people in the general population who did receive treatment (hazard ratio, 8.78; 95% confidence interval, 4.37-17.64), Pirathiv Kugathasan and his associates reported in JAMA Psychiatry.
In contrast, the investigators found, patients with schizophrenia who received cardioprotective treatment had a 97% higher mortality (HR, 1.97; 95% CI, 1.25-310), compared with the treated general population, which was not statistically different from individuals in the general population who did not receive treatment (HR, 2.95; 95% CI, 2.62-3.32) after adjustment for baseline characteristics.
“Given the increased cardiovascular risk among patients with schizophrenia, we believe that the current findings support the use of intensive cardioprotective treatments in patients with schizophrenia,” reported Mr. Kugathasan, a PhD candidate in the department of psychiatry at Aalborg University Hospital in Denmark.
However, 7.8% of patients diagnosed with schizophrenia received no prescriptions for cardioprotective medications after a myocardial infarction, compared with 3.3% of the general population. They were significantly less likely than were individuals from the general population to receive a prescription for antiplatelets (84.9% vs. 91.8%), vitamin K antagonists (15.9% vs. 24.2%), beta-blockers (74.1% vs. 84.9%), ACE inhibitors (70.9% vs. 86.6%), and statins (72.2% vs. 87.3%).
The mortality rates were not significantly different between untreated patients with schizophrenia and untreated participants from the general population.
When the researchers examined the effects of different treatment types, they found that mortality rates were still higher in treated patients with schizophrenia, compared with treated patients from the general population – with the exception of those treated with antiplatelets and statins. For ACE inhibitors, treated patients with schizophrenia had a twofold higher mortality than that of treated patients from the general population, while the mortality was more than twofold higher in the case of vitamin K antagonists.
“Previous studies have found that patients with schizophrenia have increased cardiovascular mortality and a lower prescription rate for cardioprotective treatment compared with the general population,” the authors wrote.
They noted that patients with schizophrenia might have more problems with medication adherence, and pointed to another study showing this in patients with schizophrenia and diabetes. Other studies also showed that patients with schizophrenia were less likely to talk to a cardiologist after a cardiac event.
“Together, ,” they wrote. “This hypothesis could explain the results of a generally increased mortality rate among patients with schizophrenia, especially when the results suggest that these patients die of cardiovascular causes that might be treatable.”
They called for patients with schizophrenia to be followed up during treatment, and to increase treatment intensity after cardiac events, “because a diagnosis of schizophrenia may be associated with an increased cardiac risk, which potentially can be countered by secondary preventive cardiac treatment.”
In patients who received triple therapy, the mortality rates were similar for those with schizophrenia and the general population. However, there were much higher mortality rate differences between patients with schizophrenia and the general population for those receiving dual or monotherapy.
Significantly more patients from the general population were readmitted with myocardial infarction and more underwent percutaneous coronary intervention than from the group of patients with schizophrenia.
The prevalence of hypertension was similar between the two populations, but more patients with schizophrenia had diabetes and substance abuse, compared with the general population.
The authors noted that they did not have information on the severity of myocardial infarction or the effect of other lifestyle factors.
“Future research should attempt to assess the degree to which these factors contribute to the increased mortality in patients with schizophrenia,” they wrote.
One author declared grants, speaking fees and advisory roles with the pharmaceutical industry. No other conflicts of interest were declared.
SOURCE: Kugathasan P et al. JAMA Psychiatry 2018. Oct 24. doi: 10.1001/jamapsychiatry.2018.2742.
FROM JAMA PSYCHIATRY
Key clinical point: Secondary prevention of cardiovascular disease shows significant benefits in patients with schizophrenia but is underused.
Major finding: Patients with schizophrenia are significantly less likely to receive all forms of secondary cardiovascular protective medications.
Study details: Nationwide cohort study of 105,018 patients with myocardial infarction.
Disclosures: One author declared grants, speaking fees and advisory roles with the pharmaceutical industry. No other conflicts of interest were declared.
Source: Kugathasan P et al. JAMA Psychiatry. 2018 Oct 24. doi: 10.1001/jamapsychiatry.2018.2742.
Chinese American man with high risk of psychosis
Editors’ Note: Curbside Consult is an occasional column aimed at helping psychiatrists think through family and cultural considerations when treating patients. It examines case vignettes and is written by two Group for the Advancement of Psychiatry (GAP) committees – the Committee on Family Psychiatry and the Committee on Cultural Psychiatry. The contributors have revised selected patient details to shield the identities of the patients/cases and to comply with HIPAA requirements.
Case vignette
Bill is a 25-year-old man of Chinese descent who sought psychiatric evaluation of his psychosis risk. His parents emigrated from China to Canada more than 30 years ago; Bill was born in Canada, and moved to the United States with his parents and two siblings at age 7.
His family has a strong history of mental illness. His older sister was diagnosed with schizophrenia; she frequently got into verbal altercations with her parents. When Bill was 15, she walked out of the house after a fight with the family and never returned. Bill’s family thinks his father has had paranoid delusions. In the past, he attempted to call the police multiple times because he suspected the neighbors had planted a listening device in his front yard. The family stopped him from making the actual calls. However, the family never brought him to psychiatric evaluation because of perceived stigma and social discrimination in their community. He also was emotionally and physically abusive to Bill during his childhood by calling him names and hitting him with a belt. As an adult, Bill still has posttraumatic stress disorder symptoms including flashbacks, nightmares, and avoidance when thinking about his father.
Bill identifies himself as Chinese American and speaks English only. He often perceives himself as a newcomer to U.S. society, making comments such as: “I guess I should live my life like this to fulfill the American dream.” Bill’s parents placed a strong emphasis on academic success, often at the expense of their children’s social interaction and playtime activities. Bill describes himself as a “loner” with few friends. He maintained high academic achievement during high school and was accepted by a prestigious college. Although he was interested in music composition, Bill was “forced” by his parents to major in courses deemed good preparation for law school.
During college, he suffered severe depression with insomnia, low energy, hopelessness, anhedonia, social withdrawal, poor appetite with weight loss, and ruminative thoughts but without delusional thoughts or perceptual disturbances. He had one near-lethal suicide attempt, during which he impulsively took the contents of an entire bottle of Tylenol in the context of family conflicts, resulting in psychiatric hospitalization. Bill recalled with animosity the inpatient psychiatrists who put him on psychotropic medications during a 3-day hospitalization. He was not adherent to the medication and did not follow up with postdischarge outpatient care. He did not remember the medication trial he had during the hospitalization, nor did he give consent to obtain medical records from there. Bill withdrew from college in sophomore year, because of his declining academic performance secondary to his mental illness. He currently works at a gas station.
Over the last year, Bill’s interpersonal communication has become disorganized in both work and social settings, and he has developed thought blocking, causing him substantial distress. He intermittently hears voices of strangers in the background; these have gradually become more frequent, now occurring 3-4 times a week. Bill also is more depressed, with frequent crying episodes and worsening social isolation. He often thinks that life is not worth living, but he has no active suicidal plans or intent.
Bill’s supervisor and coworkers strongly suggested that he seek medical evaluation. As an outpatient, Bill started weekly cognitive-behavioral therapy (CBT) and biweekly medication sessions for early psychosis symptoms, receiving low-dose risperidone (1 mg b.i.d.) and fluoxetine (20 mg daily). Despite initial improvement, he was very skeptical about continuing the medications because of concern that they will cause a “change in his identity” by altering his body chemistry. His parents have been reluctant to join family meetings, because they were ambivalent about Bill’s ongoing psychiatric treatment.
Treatment team’s impressions
Clinical high risk (CHR) syndrome refers to the prodromal phase before a full psychotic disorder. As one of the three subcategories of CHR, genetic risk and deterioration (GRD) prodromal syndrome is defined by having a genetic risk for psychosis (first-degree relative with a psychotic disorder or meeting criteria for schizotypal personality disorder) and a recent decline in daily functioning equivalent to a 30% drop in Global Assessment of Functioning rating.1 Due to Bill’s family history of psychosis, new difficulties in self-care, psychotic-spectrum symptoms, and declining social/executive function, he meets criteria for GRD prodromal syndrome. In addition, major depressive disorder should be considered on his differential diagnosis.
Bill has not received a diagnosis of acute psychosis, and instead is judged to be in the CHR spectrum for psychosis, because of his level of insight that the occasional perceptual disturbance and abnormal thought content are in his own mind. Since 26% of individuals with CHR in mainland China2 and 35% in the general U.S. population develop fully psychotic symptoms within 2-3 years, Bill’s current presentation warrants secondary preventive care (early intervention) to promote improved clinical outcomes. Given the high rates of comorbid depressive and anxiety disorders among individuals with CHR, Bill’s mood symptoms and passive suicidality also require psychiatric intervention. The treatment team raised three questions, which are answered below.
1. How can we understand Bill’s and his family’s resistance to mental health treatment?
Chinese Americans tend to access mental health services at a lower rate than that of the general U.S. population.3 They also tend to exhibit elevated discontinuation from mental health care, compared with non-Latino whites.4 Since first- and second-generation Chinese Americans have similar use rates, it is likely the barriers to care are not immigration specific but also related to factors that endure across generations, including culture-related aspects. These include cultural concepts of illness and how to interpret prodromal symptoms such as Bill’s, stigma and interpersonal shame regarding mental illness and psychiatric treatment, and value orientations such as self-reliance, avoidance of direct expressions of interpersonal conflict, and family privacy.
For example, Chinese Americans tend to emphasize the physical-symptom components of psychiatric problems, partly because of mind-body holism – which combines physical and psychological symptoms into cultural syndromes and idioms of distress – and partly because of concerns about stigma and shame regarding mental health symptoms. Hence, they are more likely to seek help from primary care clinicians for psychological distress before any mental health provider. Many Chinese Americans interpret mild to moderate psychological distress as “mental weakness” or “excessive thinking” (xiang tai duo), which does not require clinical evaluation. In this view, only alarmingly bizarre or disruptive behaviors warrant formal psychiatric treatment.
Some Chinese parents perceive psychosocial stressors, including hardships, interpersonal conflicts, and academic burdens, as understandable triggers for symptoms that clinicians would classify as schizophrenia or attenuated psychosis syndrome.5 Those views of illness can be associated in the Chinese American community with delayed diagnosis and intervention for emerging psychiatric disorders.
Social stigma regarding mental illness is pervasive in many Chinese American communities. Stigma hinders service use, to avoid shame and save the family’s “face.” Although many Chinese Americans acknowledge the efficacy of biomedical treatments for mental illness, they also remain concerned about enduring shame in their communities if they access those services.6 In line with collectivistic values traditionally held by many Chinese Americans, individuals tend to turn to relatives for help first and keep mental illness confidential within the family group to avoid disgracing the family name.7 Hence, social stigma in the Chinese community can be a key barrier to early detection and early intervention for youth at high risk of psychosis.
Culturally influenced cognitions, emotions, and values also might contribute to underuse of formal mental health services in this population. Many Chinese Americans perceive the roots of mental illness in a lack of willpower and the unwise indulgence of morbid thoughts. In these communities, direct communication of strong emotions can be discouraged – in the name of maintaining harmony, collective interests, and tolerance.8 Hence, many Chinese Americans find Western models of psychotherapy that focus on intrapsychic conflicts and/or intense interpersonal emotions incongruous with their treatment expectations. Psychiatric interview processes that explore gloomy or disturbing thoughts can be perceived as disquieting and antithetical to the goals of treatment. In addition, some individuals rooted in collectivist communities would rather keep personal psychological problems private within the family than seek counseling from an expert who is an out-group stranger.
2. How does Bill’s cultural and social context affect his prognosis?
Individuals with psychosis and their close relatives are generally vulnerable to stigma and discrimination. Mental health stigma has a substantial effect on the lives of patients with psychosis and their family members. The magnitude of the perceived stigma tends to be greater if the patient has more severe positive symptoms, is more educated, or resides in a highly urbanized area.
Acutely ill patients usually face more negative community responses than do milder cases, since their close relatives are blamed for failing to uphold the moral and legal responsibility of ensuring that the patients control their behavior. The effect of stigma in Chinese society also is greater among male patients with early-onset illness, because of the expectation that men marry and become the family breadwinner to attain higher social status. Hence, young males who are unable to achieve these socially determined adult milestones can be considered socially inferior, and suffer more community discrimination and exclusion, which are risk factors for clinical deterioration and functional impairment.
Social stigma can intensify relationship conflicts within the family and magnify expressed emotion (EE), which is defined as caregivers’ attitude toward a person with mental illness as reflected by their comments and interaction patterns. “High EE” comprises three behavioral patterns: criticism, hostility, and emotional overinvolvement. High EE is associated with psychiatric symptom relapse among individuals with schizophrenia and other disorders.9
Currently, most of the literature on EE is limited to white samples in Western countries. Some researchers have studied the relationship between the EE index of emotional overinvolvement and schizophrenia relapse among Hispanic populations.10 However, there are limited data on cultural congruence of EE research in Asian populations. Therefore, clinicians should carefully evaluate the contribution of high EE to Bill’s family’s situation during his course of treatment.
Higher education often is associated with greater levels of EE and can result in anxious and fearful responses to the person’s illness.11 This may be attributable to more negative reactions to actual or feared stigma and discrimination, possibly because relatives feel they have more to lose regarding the family’s social status, especially in densely populated urban areas where it might be harder to keep the patient’s mental illness as a “family secret.”
On the other hand, certain explanatory models of psychosis can modulate Chinese community members’ perceptions and allow ill individuals to remain socially integrated. Cultural idioms such as “excessive thinking” (xiang tai duo), “taking things too hard” (xiang bu kai), and “narrow-mindedness” (xiao xin yan) promote socially accommodating behaviors that facilitate acceptance of mildly to moderately ill individuals as full-status community members.12
Another important contributor to psychosis risk is Bill’s acculturative stress about his cultural identity. Linguistic challenges, limited social support, perceived discrimination, and an acculturation gap between parents and children are major sources of acculturative stress among Chinese American college students.13 Greater acculturative stress elevates the risk of mental illness and symptom deterioration. However, highly acculturated Chinese Americans with above-average bicultural self-efficacy tend to express more positive attitudes toward mental health services.
3. Are there culturally appropriate interventions that can help Bill and his family?
A major predictor among Chinese Americans of the intent to use services is the perceived credibility of the treatment and the provider. Ethnic-specific services staffed by bicultural/bilingual mental health clinicians delivering culturally responsive interventions are increasingly available in many metropolitan areas with major Asian communities. These programs have shown clinical efficacy in encouraging service use and promoting treatment persistence. Bill and his family may benefit from referral to ethnic-specific services, where they can obtain culturally sensitive psychoeducation about his mental illness and treatment plan.
Other services that might be useful for Bill and his family include family psychoeducation programs and supportive groups specifically designed for Chinese American families; these can improve the entire family’s psychosocial health, promote medication adherence, and reduce the risk of symptom relapse through family-centered intervention models.14 Connecting with local National Alliance on Mental Illness (NAMI) programs might help Bill’s parents obtain social support from Chinese American families with similar caregiver experiences.
However, services that are not designed specifically for Chinese-origin patients also can provide excellent care for these patients, and be perceived as credible and effective. A thorough cultural assessment is necessary, as well as inclusion of the information obtained in the treatment plan. As described in the DSM-5 Outline for Cultural Formulation and operationalized in the Cultural Formulation Interview, clinicians should assess possible cultural differences among Bill, his family, his community, and his clinicians regarding their cultural concepts of distress and illness and expectations of care in order to formulate a treatment plan acceptable to patient and family. Practical cultural barriers should be addressed, such as Bill’s parents’ limited English proficiency, in which case a bilingual clinician or trained interpreter should be included in the treatment team.
With Bill’s consent, the treatment team also should consider reaching out to his parents, especially his mother, to understand and empathize with their cultural concepts of distress and illness as well as expectations for care. In addition to providing psychoeducation, the clinicians should validate the parents’ experience of shame, fear, and worry about their son. Bill’s brother, for example, might be a useful bridge in communicating with the parents given his higher acculturation and potentially greater acceptance of psychiatric care. He might help alleviate the tension between Bill and his parents and encourage them to seek family-based help.
Take-home points
- Clinical training programs should offer cultural competency training about underserved populations, including communities of color.
- Certain key concepts, such as traditional idioms of distress and explanatory models, social stigma, and acculturative stress, should be included in these trainings and evaluated in a comprehensive psychosocial assessment.
- High expressed emotion among family caregivers is associated with higher rates of psychiatric symptom relapse, whereas families with above-average bicultural self-efficacy have more positive attitudes toward mental health services.
- Clinicians should incorporate culturally appropriate educational materials (for example, CHR warning signs) and interventions to engage underserved patients and their families in mental health treatment.
Contributors
Emily Wu, MD – Harvard Medical School, Boston
Francis Lu, MD – University of California, Davis
John Sargent, MD – Tufts Medical Center, Boston
Roberto Lewis-Fernández, MD – Columbia College of Physicians & Surgeons, New York
If you would like to a submit case in which your understanding and treatment are affected by challenging cultural and family values, send it to cpnews@mdedge.com. We will then write back with our best answers about how one might proceed in such a case. Your case and our response will then be published at mdedge.com/psychiatry. This column is meant to be educational and does not constitute medical advice. The opinions expressed are those of the contributors and do not represent those of the organizations they are employed by or affiliated with or the Group for the Advancement of Psychiatry.
References
1. J Nerv Ment Dis. 2013 Jun;20(6);484-9.
2. Schizophr Res. 2014 Feb;152(2-3):391-9.
3. Perspect Psychiatr Care. 2013;49(4):288-92.
4. Ment Health Serv Res. 2001 Dec;3(4):201-14.
5. Br J Psychiatry. 2000 Jul;177;20-5.
6. Cultr Divers Ethnic Minor Psychol. 2008 Jan;14(1):10-8.
7. Couns Psychol. 2003 May1;31:343-61.
8. Emotion. 2002 Dec;2(4):341-60.
9. Am J Psychiatry. 1986 Nov;143(11):1361-73.
10. J Nerv Ment Dis. 2013 Oct;201(10):833-40.
11. Schizophr Bull. 1981;7(1):43-4.
12. Schizophr Bull. 2010 Jul;36(4):836-45.
13. Am J Orthopsychiatry. 2011 Oct;81(4):489-97.
14. Patient Educ Couns. 2009 Apr;75(1):67-76.
Editors’ Note: Curbside Consult is an occasional column aimed at helping psychiatrists think through family and cultural considerations when treating patients. It examines case vignettes and is written by two Group for the Advancement of Psychiatry (GAP) committees – the Committee on Family Psychiatry and the Committee on Cultural Psychiatry. The contributors have revised selected patient details to shield the identities of the patients/cases and to comply with HIPAA requirements.
Case vignette
Bill is a 25-year-old man of Chinese descent who sought psychiatric evaluation of his psychosis risk. His parents emigrated from China to Canada more than 30 years ago; Bill was born in Canada, and moved to the United States with his parents and two siblings at age 7.
His family has a strong history of mental illness. His older sister was diagnosed with schizophrenia; she frequently got into verbal altercations with her parents. When Bill was 15, she walked out of the house after a fight with the family and never returned. Bill’s family thinks his father has had paranoid delusions. In the past, he attempted to call the police multiple times because he suspected the neighbors had planted a listening device in his front yard. The family stopped him from making the actual calls. However, the family never brought him to psychiatric evaluation because of perceived stigma and social discrimination in their community. He also was emotionally and physically abusive to Bill during his childhood by calling him names and hitting him with a belt. As an adult, Bill still has posttraumatic stress disorder symptoms including flashbacks, nightmares, and avoidance when thinking about his father.
Bill identifies himself as Chinese American and speaks English only. He often perceives himself as a newcomer to U.S. society, making comments such as: “I guess I should live my life like this to fulfill the American dream.” Bill’s parents placed a strong emphasis on academic success, often at the expense of their children’s social interaction and playtime activities. Bill describes himself as a “loner” with few friends. He maintained high academic achievement during high school and was accepted by a prestigious college. Although he was interested in music composition, Bill was “forced” by his parents to major in courses deemed good preparation for law school.
During college, he suffered severe depression with insomnia, low energy, hopelessness, anhedonia, social withdrawal, poor appetite with weight loss, and ruminative thoughts but without delusional thoughts or perceptual disturbances. He had one near-lethal suicide attempt, during which he impulsively took the contents of an entire bottle of Tylenol in the context of family conflicts, resulting in psychiatric hospitalization. Bill recalled with animosity the inpatient psychiatrists who put him on psychotropic medications during a 3-day hospitalization. He was not adherent to the medication and did not follow up with postdischarge outpatient care. He did not remember the medication trial he had during the hospitalization, nor did he give consent to obtain medical records from there. Bill withdrew from college in sophomore year, because of his declining academic performance secondary to his mental illness. He currently works at a gas station.
Over the last year, Bill’s interpersonal communication has become disorganized in both work and social settings, and he has developed thought blocking, causing him substantial distress. He intermittently hears voices of strangers in the background; these have gradually become more frequent, now occurring 3-4 times a week. Bill also is more depressed, with frequent crying episodes and worsening social isolation. He often thinks that life is not worth living, but he has no active suicidal plans or intent.
Bill’s supervisor and coworkers strongly suggested that he seek medical evaluation. As an outpatient, Bill started weekly cognitive-behavioral therapy (CBT) and biweekly medication sessions for early psychosis symptoms, receiving low-dose risperidone (1 mg b.i.d.) and fluoxetine (20 mg daily). Despite initial improvement, he was very skeptical about continuing the medications because of concern that they will cause a “change in his identity” by altering his body chemistry. His parents have been reluctant to join family meetings, because they were ambivalent about Bill’s ongoing psychiatric treatment.
Treatment team’s impressions
Clinical high risk (CHR) syndrome refers to the prodromal phase before a full psychotic disorder. As one of the three subcategories of CHR, genetic risk and deterioration (GRD) prodromal syndrome is defined by having a genetic risk for psychosis (first-degree relative with a psychotic disorder or meeting criteria for schizotypal personality disorder) and a recent decline in daily functioning equivalent to a 30% drop in Global Assessment of Functioning rating.1 Due to Bill’s family history of psychosis, new difficulties in self-care, psychotic-spectrum symptoms, and declining social/executive function, he meets criteria for GRD prodromal syndrome. In addition, major depressive disorder should be considered on his differential diagnosis.
Bill has not received a diagnosis of acute psychosis, and instead is judged to be in the CHR spectrum for psychosis, because of his level of insight that the occasional perceptual disturbance and abnormal thought content are in his own mind. Since 26% of individuals with CHR in mainland China2 and 35% in the general U.S. population develop fully psychotic symptoms within 2-3 years, Bill’s current presentation warrants secondary preventive care (early intervention) to promote improved clinical outcomes. Given the high rates of comorbid depressive and anxiety disorders among individuals with CHR, Bill’s mood symptoms and passive suicidality also require psychiatric intervention. The treatment team raised three questions, which are answered below.
1. How can we understand Bill’s and his family’s resistance to mental health treatment?
Chinese Americans tend to access mental health services at a lower rate than that of the general U.S. population.3 They also tend to exhibit elevated discontinuation from mental health care, compared with non-Latino whites.4 Since first- and second-generation Chinese Americans have similar use rates, it is likely the barriers to care are not immigration specific but also related to factors that endure across generations, including culture-related aspects. These include cultural concepts of illness and how to interpret prodromal symptoms such as Bill’s, stigma and interpersonal shame regarding mental illness and psychiatric treatment, and value orientations such as self-reliance, avoidance of direct expressions of interpersonal conflict, and family privacy.
For example, Chinese Americans tend to emphasize the physical-symptom components of psychiatric problems, partly because of mind-body holism – which combines physical and psychological symptoms into cultural syndromes and idioms of distress – and partly because of concerns about stigma and shame regarding mental health symptoms. Hence, they are more likely to seek help from primary care clinicians for psychological distress before any mental health provider. Many Chinese Americans interpret mild to moderate psychological distress as “mental weakness” or “excessive thinking” (xiang tai duo), which does not require clinical evaluation. In this view, only alarmingly bizarre or disruptive behaviors warrant formal psychiatric treatment.
Some Chinese parents perceive psychosocial stressors, including hardships, interpersonal conflicts, and academic burdens, as understandable triggers for symptoms that clinicians would classify as schizophrenia or attenuated psychosis syndrome.5 Those views of illness can be associated in the Chinese American community with delayed diagnosis and intervention for emerging psychiatric disorders.
Social stigma regarding mental illness is pervasive in many Chinese American communities. Stigma hinders service use, to avoid shame and save the family’s “face.” Although many Chinese Americans acknowledge the efficacy of biomedical treatments for mental illness, they also remain concerned about enduring shame in their communities if they access those services.6 In line with collectivistic values traditionally held by many Chinese Americans, individuals tend to turn to relatives for help first and keep mental illness confidential within the family group to avoid disgracing the family name.7 Hence, social stigma in the Chinese community can be a key barrier to early detection and early intervention for youth at high risk of psychosis.
Culturally influenced cognitions, emotions, and values also might contribute to underuse of formal mental health services in this population. Many Chinese Americans perceive the roots of mental illness in a lack of willpower and the unwise indulgence of morbid thoughts. In these communities, direct communication of strong emotions can be discouraged – in the name of maintaining harmony, collective interests, and tolerance.8 Hence, many Chinese Americans find Western models of psychotherapy that focus on intrapsychic conflicts and/or intense interpersonal emotions incongruous with their treatment expectations. Psychiatric interview processes that explore gloomy or disturbing thoughts can be perceived as disquieting and antithetical to the goals of treatment. In addition, some individuals rooted in collectivist communities would rather keep personal psychological problems private within the family than seek counseling from an expert who is an out-group stranger.
2. How does Bill’s cultural and social context affect his prognosis?
Individuals with psychosis and their close relatives are generally vulnerable to stigma and discrimination. Mental health stigma has a substantial effect on the lives of patients with psychosis and their family members. The magnitude of the perceived stigma tends to be greater if the patient has more severe positive symptoms, is more educated, or resides in a highly urbanized area.
Acutely ill patients usually face more negative community responses than do milder cases, since their close relatives are blamed for failing to uphold the moral and legal responsibility of ensuring that the patients control their behavior. The effect of stigma in Chinese society also is greater among male patients with early-onset illness, because of the expectation that men marry and become the family breadwinner to attain higher social status. Hence, young males who are unable to achieve these socially determined adult milestones can be considered socially inferior, and suffer more community discrimination and exclusion, which are risk factors for clinical deterioration and functional impairment.
Social stigma can intensify relationship conflicts within the family and magnify expressed emotion (EE), which is defined as caregivers’ attitude toward a person with mental illness as reflected by their comments and interaction patterns. “High EE” comprises three behavioral patterns: criticism, hostility, and emotional overinvolvement. High EE is associated with psychiatric symptom relapse among individuals with schizophrenia and other disorders.9
Currently, most of the literature on EE is limited to white samples in Western countries. Some researchers have studied the relationship between the EE index of emotional overinvolvement and schizophrenia relapse among Hispanic populations.10 However, there are limited data on cultural congruence of EE research in Asian populations. Therefore, clinicians should carefully evaluate the contribution of high EE to Bill’s family’s situation during his course of treatment.
Higher education often is associated with greater levels of EE and can result in anxious and fearful responses to the person’s illness.11 This may be attributable to more negative reactions to actual or feared stigma and discrimination, possibly because relatives feel they have more to lose regarding the family’s social status, especially in densely populated urban areas where it might be harder to keep the patient’s mental illness as a “family secret.”
On the other hand, certain explanatory models of psychosis can modulate Chinese community members’ perceptions and allow ill individuals to remain socially integrated. Cultural idioms such as “excessive thinking” (xiang tai duo), “taking things too hard” (xiang bu kai), and “narrow-mindedness” (xiao xin yan) promote socially accommodating behaviors that facilitate acceptance of mildly to moderately ill individuals as full-status community members.12
Another important contributor to psychosis risk is Bill’s acculturative stress about his cultural identity. Linguistic challenges, limited social support, perceived discrimination, and an acculturation gap between parents and children are major sources of acculturative stress among Chinese American college students.13 Greater acculturative stress elevates the risk of mental illness and symptom deterioration. However, highly acculturated Chinese Americans with above-average bicultural self-efficacy tend to express more positive attitudes toward mental health services.
3. Are there culturally appropriate interventions that can help Bill and his family?
A major predictor among Chinese Americans of the intent to use services is the perceived credibility of the treatment and the provider. Ethnic-specific services staffed by bicultural/bilingual mental health clinicians delivering culturally responsive interventions are increasingly available in many metropolitan areas with major Asian communities. These programs have shown clinical efficacy in encouraging service use and promoting treatment persistence. Bill and his family may benefit from referral to ethnic-specific services, where they can obtain culturally sensitive psychoeducation about his mental illness and treatment plan.
Other services that might be useful for Bill and his family include family psychoeducation programs and supportive groups specifically designed for Chinese American families; these can improve the entire family’s psychosocial health, promote medication adherence, and reduce the risk of symptom relapse through family-centered intervention models.14 Connecting with local National Alliance on Mental Illness (NAMI) programs might help Bill’s parents obtain social support from Chinese American families with similar caregiver experiences.
However, services that are not designed specifically for Chinese-origin patients also can provide excellent care for these patients, and be perceived as credible and effective. A thorough cultural assessment is necessary, as well as inclusion of the information obtained in the treatment plan. As described in the DSM-5 Outline for Cultural Formulation and operationalized in the Cultural Formulation Interview, clinicians should assess possible cultural differences among Bill, his family, his community, and his clinicians regarding their cultural concepts of distress and illness and expectations of care in order to formulate a treatment plan acceptable to patient and family. Practical cultural barriers should be addressed, such as Bill’s parents’ limited English proficiency, in which case a bilingual clinician or trained interpreter should be included in the treatment team.
With Bill’s consent, the treatment team also should consider reaching out to his parents, especially his mother, to understand and empathize with their cultural concepts of distress and illness as well as expectations for care. In addition to providing psychoeducation, the clinicians should validate the parents’ experience of shame, fear, and worry about their son. Bill’s brother, for example, might be a useful bridge in communicating with the parents given his higher acculturation and potentially greater acceptance of psychiatric care. He might help alleviate the tension between Bill and his parents and encourage them to seek family-based help.
Take-home points
- Clinical training programs should offer cultural competency training about underserved populations, including communities of color.
- Certain key concepts, such as traditional idioms of distress and explanatory models, social stigma, and acculturative stress, should be included in these trainings and evaluated in a comprehensive psychosocial assessment.
- High expressed emotion among family caregivers is associated with higher rates of psychiatric symptom relapse, whereas families with above-average bicultural self-efficacy have more positive attitudes toward mental health services.
- Clinicians should incorporate culturally appropriate educational materials (for example, CHR warning signs) and interventions to engage underserved patients and their families in mental health treatment.
Contributors
Emily Wu, MD – Harvard Medical School, Boston
Francis Lu, MD – University of California, Davis
John Sargent, MD – Tufts Medical Center, Boston
Roberto Lewis-Fernández, MD – Columbia College of Physicians & Surgeons, New York
If you would like to a submit case in which your understanding and treatment are affected by challenging cultural and family values, send it to cpnews@mdedge.com. We will then write back with our best answers about how one might proceed in such a case. Your case and our response will then be published at mdedge.com/psychiatry. This column is meant to be educational and does not constitute medical advice. The opinions expressed are those of the contributors and do not represent those of the organizations they are employed by or affiliated with or the Group for the Advancement of Psychiatry.
References
1. J Nerv Ment Dis. 2013 Jun;20(6);484-9.
2. Schizophr Res. 2014 Feb;152(2-3):391-9.
3. Perspect Psychiatr Care. 2013;49(4):288-92.
4. Ment Health Serv Res. 2001 Dec;3(4):201-14.
5. Br J Psychiatry. 2000 Jul;177;20-5.
6. Cultr Divers Ethnic Minor Psychol. 2008 Jan;14(1):10-8.
7. Couns Psychol. 2003 May1;31:343-61.
8. Emotion. 2002 Dec;2(4):341-60.
9. Am J Psychiatry. 1986 Nov;143(11):1361-73.
10. J Nerv Ment Dis. 2013 Oct;201(10):833-40.
11. Schizophr Bull. 1981;7(1):43-4.
12. Schizophr Bull. 2010 Jul;36(4):836-45.
13. Am J Orthopsychiatry. 2011 Oct;81(4):489-97.
14. Patient Educ Couns. 2009 Apr;75(1):67-76.
Editors’ Note: Curbside Consult is an occasional column aimed at helping psychiatrists think through family and cultural considerations when treating patients. It examines case vignettes and is written by two Group for the Advancement of Psychiatry (GAP) committees – the Committee on Family Psychiatry and the Committee on Cultural Psychiatry. The contributors have revised selected patient details to shield the identities of the patients/cases and to comply with HIPAA requirements.
Case vignette
Bill is a 25-year-old man of Chinese descent who sought psychiatric evaluation of his psychosis risk. His parents emigrated from China to Canada more than 30 years ago; Bill was born in Canada, and moved to the United States with his parents and two siblings at age 7.
His family has a strong history of mental illness. His older sister was diagnosed with schizophrenia; she frequently got into verbal altercations with her parents. When Bill was 15, she walked out of the house after a fight with the family and never returned. Bill’s family thinks his father has had paranoid delusions. In the past, he attempted to call the police multiple times because he suspected the neighbors had planted a listening device in his front yard. The family stopped him from making the actual calls. However, the family never brought him to psychiatric evaluation because of perceived stigma and social discrimination in their community. He also was emotionally and physically abusive to Bill during his childhood by calling him names and hitting him with a belt. As an adult, Bill still has posttraumatic stress disorder symptoms including flashbacks, nightmares, and avoidance when thinking about his father.
Bill identifies himself as Chinese American and speaks English only. He often perceives himself as a newcomer to U.S. society, making comments such as: “I guess I should live my life like this to fulfill the American dream.” Bill’s parents placed a strong emphasis on academic success, often at the expense of their children’s social interaction and playtime activities. Bill describes himself as a “loner” with few friends. He maintained high academic achievement during high school and was accepted by a prestigious college. Although he was interested in music composition, Bill was “forced” by his parents to major in courses deemed good preparation for law school.
During college, he suffered severe depression with insomnia, low energy, hopelessness, anhedonia, social withdrawal, poor appetite with weight loss, and ruminative thoughts but without delusional thoughts or perceptual disturbances. He had one near-lethal suicide attempt, during which he impulsively took the contents of an entire bottle of Tylenol in the context of family conflicts, resulting in psychiatric hospitalization. Bill recalled with animosity the inpatient psychiatrists who put him on psychotropic medications during a 3-day hospitalization. He was not adherent to the medication and did not follow up with postdischarge outpatient care. He did not remember the medication trial he had during the hospitalization, nor did he give consent to obtain medical records from there. Bill withdrew from college in sophomore year, because of his declining academic performance secondary to his mental illness. He currently works at a gas station.
Over the last year, Bill’s interpersonal communication has become disorganized in both work and social settings, and he has developed thought blocking, causing him substantial distress. He intermittently hears voices of strangers in the background; these have gradually become more frequent, now occurring 3-4 times a week. Bill also is more depressed, with frequent crying episodes and worsening social isolation. He often thinks that life is not worth living, but he has no active suicidal plans or intent.
Bill’s supervisor and coworkers strongly suggested that he seek medical evaluation. As an outpatient, Bill started weekly cognitive-behavioral therapy (CBT) and biweekly medication sessions for early psychosis symptoms, receiving low-dose risperidone (1 mg b.i.d.) and fluoxetine (20 mg daily). Despite initial improvement, he was very skeptical about continuing the medications because of concern that they will cause a “change in his identity” by altering his body chemistry. His parents have been reluctant to join family meetings, because they were ambivalent about Bill’s ongoing psychiatric treatment.
Treatment team’s impressions
Clinical high risk (CHR) syndrome refers to the prodromal phase before a full psychotic disorder. As one of the three subcategories of CHR, genetic risk and deterioration (GRD) prodromal syndrome is defined by having a genetic risk for psychosis (first-degree relative with a psychotic disorder or meeting criteria for schizotypal personality disorder) and a recent decline in daily functioning equivalent to a 30% drop in Global Assessment of Functioning rating.1 Due to Bill’s family history of psychosis, new difficulties in self-care, psychotic-spectrum symptoms, and declining social/executive function, he meets criteria for GRD prodromal syndrome. In addition, major depressive disorder should be considered on his differential diagnosis.
Bill has not received a diagnosis of acute psychosis, and instead is judged to be in the CHR spectrum for psychosis, because of his level of insight that the occasional perceptual disturbance and abnormal thought content are in his own mind. Since 26% of individuals with CHR in mainland China2 and 35% in the general U.S. population develop fully psychotic symptoms within 2-3 years, Bill’s current presentation warrants secondary preventive care (early intervention) to promote improved clinical outcomes. Given the high rates of comorbid depressive and anxiety disorders among individuals with CHR, Bill’s mood symptoms and passive suicidality also require psychiatric intervention. The treatment team raised three questions, which are answered below.
1. How can we understand Bill’s and his family’s resistance to mental health treatment?
Chinese Americans tend to access mental health services at a lower rate than that of the general U.S. population.3 They also tend to exhibit elevated discontinuation from mental health care, compared with non-Latino whites.4 Since first- and second-generation Chinese Americans have similar use rates, it is likely the barriers to care are not immigration specific but also related to factors that endure across generations, including culture-related aspects. These include cultural concepts of illness and how to interpret prodromal symptoms such as Bill’s, stigma and interpersonal shame regarding mental illness and psychiatric treatment, and value orientations such as self-reliance, avoidance of direct expressions of interpersonal conflict, and family privacy.
For example, Chinese Americans tend to emphasize the physical-symptom components of psychiatric problems, partly because of mind-body holism – which combines physical and psychological symptoms into cultural syndromes and idioms of distress – and partly because of concerns about stigma and shame regarding mental health symptoms. Hence, they are more likely to seek help from primary care clinicians for psychological distress before any mental health provider. Many Chinese Americans interpret mild to moderate psychological distress as “mental weakness” or “excessive thinking” (xiang tai duo), which does not require clinical evaluation. In this view, only alarmingly bizarre or disruptive behaviors warrant formal psychiatric treatment.
Some Chinese parents perceive psychosocial stressors, including hardships, interpersonal conflicts, and academic burdens, as understandable triggers for symptoms that clinicians would classify as schizophrenia or attenuated psychosis syndrome.5 Those views of illness can be associated in the Chinese American community with delayed diagnosis and intervention for emerging psychiatric disorders.
Social stigma regarding mental illness is pervasive in many Chinese American communities. Stigma hinders service use, to avoid shame and save the family’s “face.” Although many Chinese Americans acknowledge the efficacy of biomedical treatments for mental illness, they also remain concerned about enduring shame in their communities if they access those services.6 In line with collectivistic values traditionally held by many Chinese Americans, individuals tend to turn to relatives for help first and keep mental illness confidential within the family group to avoid disgracing the family name.7 Hence, social stigma in the Chinese community can be a key barrier to early detection and early intervention for youth at high risk of psychosis.
Culturally influenced cognitions, emotions, and values also might contribute to underuse of formal mental health services in this population. Many Chinese Americans perceive the roots of mental illness in a lack of willpower and the unwise indulgence of morbid thoughts. In these communities, direct communication of strong emotions can be discouraged – in the name of maintaining harmony, collective interests, and tolerance.8 Hence, many Chinese Americans find Western models of psychotherapy that focus on intrapsychic conflicts and/or intense interpersonal emotions incongruous with their treatment expectations. Psychiatric interview processes that explore gloomy or disturbing thoughts can be perceived as disquieting and antithetical to the goals of treatment. In addition, some individuals rooted in collectivist communities would rather keep personal psychological problems private within the family than seek counseling from an expert who is an out-group stranger.
2. How does Bill’s cultural and social context affect his prognosis?
Individuals with psychosis and their close relatives are generally vulnerable to stigma and discrimination. Mental health stigma has a substantial effect on the lives of patients with psychosis and their family members. The magnitude of the perceived stigma tends to be greater if the patient has more severe positive symptoms, is more educated, or resides in a highly urbanized area.
Acutely ill patients usually face more negative community responses than do milder cases, since their close relatives are blamed for failing to uphold the moral and legal responsibility of ensuring that the patients control their behavior. The effect of stigma in Chinese society also is greater among male patients with early-onset illness, because of the expectation that men marry and become the family breadwinner to attain higher social status. Hence, young males who are unable to achieve these socially determined adult milestones can be considered socially inferior, and suffer more community discrimination and exclusion, which are risk factors for clinical deterioration and functional impairment.
Social stigma can intensify relationship conflicts within the family and magnify expressed emotion (EE), which is defined as caregivers’ attitude toward a person with mental illness as reflected by their comments and interaction patterns. “High EE” comprises three behavioral patterns: criticism, hostility, and emotional overinvolvement. High EE is associated with psychiatric symptom relapse among individuals with schizophrenia and other disorders.9
Currently, most of the literature on EE is limited to white samples in Western countries. Some researchers have studied the relationship between the EE index of emotional overinvolvement and schizophrenia relapse among Hispanic populations.10 However, there are limited data on cultural congruence of EE research in Asian populations. Therefore, clinicians should carefully evaluate the contribution of high EE to Bill’s family’s situation during his course of treatment.
Higher education often is associated with greater levels of EE and can result in anxious and fearful responses to the person’s illness.11 This may be attributable to more negative reactions to actual or feared stigma and discrimination, possibly because relatives feel they have more to lose regarding the family’s social status, especially in densely populated urban areas where it might be harder to keep the patient’s mental illness as a “family secret.”
On the other hand, certain explanatory models of psychosis can modulate Chinese community members’ perceptions and allow ill individuals to remain socially integrated. Cultural idioms such as “excessive thinking” (xiang tai duo), “taking things too hard” (xiang bu kai), and “narrow-mindedness” (xiao xin yan) promote socially accommodating behaviors that facilitate acceptance of mildly to moderately ill individuals as full-status community members.12
Another important contributor to psychosis risk is Bill’s acculturative stress about his cultural identity. Linguistic challenges, limited social support, perceived discrimination, and an acculturation gap between parents and children are major sources of acculturative stress among Chinese American college students.13 Greater acculturative stress elevates the risk of mental illness and symptom deterioration. However, highly acculturated Chinese Americans with above-average bicultural self-efficacy tend to express more positive attitudes toward mental health services.
3. Are there culturally appropriate interventions that can help Bill and his family?
A major predictor among Chinese Americans of the intent to use services is the perceived credibility of the treatment and the provider. Ethnic-specific services staffed by bicultural/bilingual mental health clinicians delivering culturally responsive interventions are increasingly available in many metropolitan areas with major Asian communities. These programs have shown clinical efficacy in encouraging service use and promoting treatment persistence. Bill and his family may benefit from referral to ethnic-specific services, where they can obtain culturally sensitive psychoeducation about his mental illness and treatment plan.
Other services that might be useful for Bill and his family include family psychoeducation programs and supportive groups specifically designed for Chinese American families; these can improve the entire family’s psychosocial health, promote medication adherence, and reduce the risk of symptom relapse through family-centered intervention models.14 Connecting with local National Alliance on Mental Illness (NAMI) programs might help Bill’s parents obtain social support from Chinese American families with similar caregiver experiences.
However, services that are not designed specifically for Chinese-origin patients also can provide excellent care for these patients, and be perceived as credible and effective. A thorough cultural assessment is necessary, as well as inclusion of the information obtained in the treatment plan. As described in the DSM-5 Outline for Cultural Formulation and operationalized in the Cultural Formulation Interview, clinicians should assess possible cultural differences among Bill, his family, his community, and his clinicians regarding their cultural concepts of distress and illness and expectations of care in order to formulate a treatment plan acceptable to patient and family. Practical cultural barriers should be addressed, such as Bill’s parents’ limited English proficiency, in which case a bilingual clinician or trained interpreter should be included in the treatment team.
With Bill’s consent, the treatment team also should consider reaching out to his parents, especially his mother, to understand and empathize with their cultural concepts of distress and illness as well as expectations for care. In addition to providing psychoeducation, the clinicians should validate the parents’ experience of shame, fear, and worry about their son. Bill’s brother, for example, might be a useful bridge in communicating with the parents given his higher acculturation and potentially greater acceptance of psychiatric care. He might help alleviate the tension between Bill and his parents and encourage them to seek family-based help.
Take-home points
- Clinical training programs should offer cultural competency training about underserved populations, including communities of color.
- Certain key concepts, such as traditional idioms of distress and explanatory models, social stigma, and acculturative stress, should be included in these trainings and evaluated in a comprehensive psychosocial assessment.
- High expressed emotion among family caregivers is associated with higher rates of psychiatric symptom relapse, whereas families with above-average bicultural self-efficacy have more positive attitudes toward mental health services.
- Clinicians should incorporate culturally appropriate educational materials (for example, CHR warning signs) and interventions to engage underserved patients and their families in mental health treatment.
Contributors
Emily Wu, MD – Harvard Medical School, Boston
Francis Lu, MD – University of California, Davis
John Sargent, MD – Tufts Medical Center, Boston
Roberto Lewis-Fernández, MD – Columbia College of Physicians & Surgeons, New York
If you would like to a submit case in which your understanding and treatment are affected by challenging cultural and family values, send it to cpnews@mdedge.com. We will then write back with our best answers about how one might proceed in such a case. Your case and our response will then be published at mdedge.com/psychiatry. This column is meant to be educational and does not constitute medical advice. The opinions expressed are those of the contributors and do not represent those of the organizations they are employed by or affiliated with or the Group for the Advancement of Psychiatry.
References
1. J Nerv Ment Dis. 2013 Jun;20(6);484-9.
2. Schizophr Res. 2014 Feb;152(2-3):391-9.
3. Perspect Psychiatr Care. 2013;49(4):288-92.
4. Ment Health Serv Res. 2001 Dec;3(4):201-14.
5. Br J Psychiatry. 2000 Jul;177;20-5.
6. Cultr Divers Ethnic Minor Psychol. 2008 Jan;14(1):10-8.
7. Couns Psychol. 2003 May1;31:343-61.
8. Emotion. 2002 Dec;2(4):341-60.
9. Am J Psychiatry. 1986 Nov;143(11):1361-73.
10. J Nerv Ment Dis. 2013 Oct;201(10):833-40.
11. Schizophr Bull. 1981;7(1):43-4.
12. Schizophr Bull. 2010 Jul;36(4):836-45.
13. Am J Orthopsychiatry. 2011 Oct;81(4):489-97.
14. Patient Educ Couns. 2009 Apr;75(1):67-76.
Brain mapping takes next step toward precision psychiatry
Brain mapping patterns varied with clinical mental health diagnoses in a study of 218 patients with schizophrenia spectrum disorders.
“Psychiatry is now the last area of medicine in which diseases are diagnosed solely on the basis of symptoms, and biomarkers to assist treatment remain to be developed,” wrote Thomas Wolfers of Radboud University, Nijmegen, the Netherlands, and his colleagues. They also said schizophrenia and bipolar disorder “are excellent examples of highly heterogeneous mental disorders.”
To explore brain structure homogeneity, the researchers used brain scans and mapping models to compare results in 218 adults aged 18-65 years with schizophrenia disorders (163 with schizophrenia and 190 with bipolar disorder) and 256 healthy controls. Demographics were similar between the groups.
The MRI data showed that the same abnormalities in more than 2% of patients with the same disorder occurred in very few loci. Schizophrenia patients showed significantly reduced gray matter in the frontal, cerebellar, and temporal regions; most bipolar patients showed changes in the cerebellar region.
The researchers identified extreme deviations across patients and controls in gray and white matter.
In gray matter, schizophrenia patients had a significantly higher percentage of extreme negative deviations across voxels (0.9% of voxels), compared with both bipolar patients and healthy controls (0.24% and 0.23%, respectively).
In white matter, a similar pattern emerged; schizophrenia patients had a significantly higher percentage of extreme negative deviations (0.62%), compared with healthy controls and bipolar patients (0.25% and 0.41%, respectively). In addition, extreme positive deviations were significantly higher among the controls (1.14% of voxels), compared with schizophrenia and bipolar patients (0.83% for both).
The findings support data from previous studies suggesting reduced cortical volume in schizophrenia patients, compared with healthy controls and bipolar disorder patients, the researchers noted.
“In this study, patients with schizophrenia and bipolar disorder differed extremely on an individual level; the lack of substantial overlap among patients in terms of extreme deviations from the normative model is evidence of the high degree of biological heterogeneity in both disorders,” wrote Mr. Wolfers and his colleagues.
The main limitations of the study were the inability to control for confounding variables and to make conclusions about causality, the researchers said. However, the absence of overlap in patients with the same disorder supports the use of brain mapping to study individual pathophysiologic signatures in schizophrenia and bipolar disorder patients, they concluded.
Mr. Wolfers had no financial conflicts to disclose. The study was supported by grants from multiple organizations, including the Netherlands Organisation for Scientific Research and the Wellcome Trust U.K. Strategic Award.
SOURCE: Wolfers T et al. JAMA Psychiatry. 2018. doi: 10.1001/jamapsychiatry.2018.2467.
A growing body of evidence suggests that “conventional psychiatric diagnoses of serious mental illness (SMI), when tested, do not show a common biology,” wrote Carol A. Tamminga, MD, and Elena I. Ivleva, MD, PhD, in an accompanying editorial. The editorialists noted the work of the Bipolar-Schizophrenia Network on Intermediate Phenotypes consortium to identify subtype disease clusters.
However, they wrote: “We question whether these biotype structures represent disease groups as opposed to mere brain biomarker clusters. Dr. Tamminga and Dr. Ivleva also expressed concern about whether biologically based subgroups would be clinically useful and questioned what evidence would be needed to accept such subtypes as disease subgroups.
Their ideas for further exploration included identifying a characteristic genetic fingerprint to help determine a common pathophysiology. In addition, “a disorder clustered by biological features might also show a distinctive pharmacological profile,” they wrote.
Next steps include making severe mental illness into a condition diagnosable based on biomarkers that also can serve as a foundation for treatment, which would be “a revolution in our ability to understand and treat complex brain disorders,” they noted (JAMA Psychiatry. 2018 Oct. doi: 10.1001/jamapsychiatry.2018.2451).
Dr. Tamminga and Dr. Ivleva are affiliated with the University of Texas, Dallas. Both of them serve as researchers with the Bipolar-Schizophrenia Network on Intermediate Phenotypes consortium and reported funding from the National Institute for Mental Health.
A growing body of evidence suggests that “conventional psychiatric diagnoses of serious mental illness (SMI), when tested, do not show a common biology,” wrote Carol A. Tamminga, MD, and Elena I. Ivleva, MD, PhD, in an accompanying editorial. The editorialists noted the work of the Bipolar-Schizophrenia Network on Intermediate Phenotypes consortium to identify subtype disease clusters.
However, they wrote: “We question whether these biotype structures represent disease groups as opposed to mere brain biomarker clusters. Dr. Tamminga and Dr. Ivleva also expressed concern about whether biologically based subgroups would be clinically useful and questioned what evidence would be needed to accept such subtypes as disease subgroups.
Their ideas for further exploration included identifying a characteristic genetic fingerprint to help determine a common pathophysiology. In addition, “a disorder clustered by biological features might also show a distinctive pharmacological profile,” they wrote.
Next steps include making severe mental illness into a condition diagnosable based on biomarkers that also can serve as a foundation for treatment, which would be “a revolution in our ability to understand and treat complex brain disorders,” they noted (JAMA Psychiatry. 2018 Oct. doi: 10.1001/jamapsychiatry.2018.2451).
Dr. Tamminga and Dr. Ivleva are affiliated with the University of Texas, Dallas. Both of them serve as researchers with the Bipolar-Schizophrenia Network on Intermediate Phenotypes consortium and reported funding from the National Institute for Mental Health.
A growing body of evidence suggests that “conventional psychiatric diagnoses of serious mental illness (SMI), when tested, do not show a common biology,” wrote Carol A. Tamminga, MD, and Elena I. Ivleva, MD, PhD, in an accompanying editorial. The editorialists noted the work of the Bipolar-Schizophrenia Network on Intermediate Phenotypes consortium to identify subtype disease clusters.
However, they wrote: “We question whether these biotype structures represent disease groups as opposed to mere brain biomarker clusters. Dr. Tamminga and Dr. Ivleva also expressed concern about whether biologically based subgroups would be clinically useful and questioned what evidence would be needed to accept such subtypes as disease subgroups.
Their ideas for further exploration included identifying a characteristic genetic fingerprint to help determine a common pathophysiology. In addition, “a disorder clustered by biological features might also show a distinctive pharmacological profile,” they wrote.
Next steps include making severe mental illness into a condition diagnosable based on biomarkers that also can serve as a foundation for treatment, which would be “a revolution in our ability to understand and treat complex brain disorders,” they noted (JAMA Psychiatry. 2018 Oct. doi: 10.1001/jamapsychiatry.2018.2451).
Dr. Tamminga and Dr. Ivleva are affiliated with the University of Texas, Dallas. Both of them serve as researchers with the Bipolar-Schizophrenia Network on Intermediate Phenotypes consortium and reported funding from the National Institute for Mental Health.
Brain mapping patterns varied with clinical mental health diagnoses in a study of 218 patients with schizophrenia spectrum disorders.
“Psychiatry is now the last area of medicine in which diseases are diagnosed solely on the basis of symptoms, and biomarkers to assist treatment remain to be developed,” wrote Thomas Wolfers of Radboud University, Nijmegen, the Netherlands, and his colleagues. They also said schizophrenia and bipolar disorder “are excellent examples of highly heterogeneous mental disorders.”
To explore brain structure homogeneity, the researchers used brain scans and mapping models to compare results in 218 adults aged 18-65 years with schizophrenia disorders (163 with schizophrenia and 190 with bipolar disorder) and 256 healthy controls. Demographics were similar between the groups.
The MRI data showed that the same abnormalities in more than 2% of patients with the same disorder occurred in very few loci. Schizophrenia patients showed significantly reduced gray matter in the frontal, cerebellar, and temporal regions; most bipolar patients showed changes in the cerebellar region.
The researchers identified extreme deviations across patients and controls in gray and white matter.
In gray matter, schizophrenia patients had a significantly higher percentage of extreme negative deviations across voxels (0.9% of voxels), compared with both bipolar patients and healthy controls (0.24% and 0.23%, respectively).
In white matter, a similar pattern emerged; schizophrenia patients had a significantly higher percentage of extreme negative deviations (0.62%), compared with healthy controls and bipolar patients (0.25% and 0.41%, respectively). In addition, extreme positive deviations were significantly higher among the controls (1.14% of voxels), compared with schizophrenia and bipolar patients (0.83% for both).
The findings support data from previous studies suggesting reduced cortical volume in schizophrenia patients, compared with healthy controls and bipolar disorder patients, the researchers noted.
“In this study, patients with schizophrenia and bipolar disorder differed extremely on an individual level; the lack of substantial overlap among patients in terms of extreme deviations from the normative model is evidence of the high degree of biological heterogeneity in both disorders,” wrote Mr. Wolfers and his colleagues.
The main limitations of the study were the inability to control for confounding variables and to make conclusions about causality, the researchers said. However, the absence of overlap in patients with the same disorder supports the use of brain mapping to study individual pathophysiologic signatures in schizophrenia and bipolar disorder patients, they concluded.
Mr. Wolfers had no financial conflicts to disclose. The study was supported by grants from multiple organizations, including the Netherlands Organisation for Scientific Research and the Wellcome Trust U.K. Strategic Award.
SOURCE: Wolfers T et al. JAMA Psychiatry. 2018. doi: 10.1001/jamapsychiatry.2018.2467.
Brain mapping patterns varied with clinical mental health diagnoses in a study of 218 patients with schizophrenia spectrum disorders.
“Psychiatry is now the last area of medicine in which diseases are diagnosed solely on the basis of symptoms, and biomarkers to assist treatment remain to be developed,” wrote Thomas Wolfers of Radboud University, Nijmegen, the Netherlands, and his colleagues. They also said schizophrenia and bipolar disorder “are excellent examples of highly heterogeneous mental disorders.”
To explore brain structure homogeneity, the researchers used brain scans and mapping models to compare results in 218 adults aged 18-65 years with schizophrenia disorders (163 with schizophrenia and 190 with bipolar disorder) and 256 healthy controls. Demographics were similar between the groups.
The MRI data showed that the same abnormalities in more than 2% of patients with the same disorder occurred in very few loci. Schizophrenia patients showed significantly reduced gray matter in the frontal, cerebellar, and temporal regions; most bipolar patients showed changes in the cerebellar region.
The researchers identified extreme deviations across patients and controls in gray and white matter.
In gray matter, schizophrenia patients had a significantly higher percentage of extreme negative deviations across voxels (0.9% of voxels), compared with both bipolar patients and healthy controls (0.24% and 0.23%, respectively).
In white matter, a similar pattern emerged; schizophrenia patients had a significantly higher percentage of extreme negative deviations (0.62%), compared with healthy controls and bipolar patients (0.25% and 0.41%, respectively). In addition, extreme positive deviations were significantly higher among the controls (1.14% of voxels), compared with schizophrenia and bipolar patients (0.83% for both).
The findings support data from previous studies suggesting reduced cortical volume in schizophrenia patients, compared with healthy controls and bipolar disorder patients, the researchers noted.
“In this study, patients with schizophrenia and bipolar disorder differed extremely on an individual level; the lack of substantial overlap among patients in terms of extreme deviations from the normative model is evidence of the high degree of biological heterogeneity in both disorders,” wrote Mr. Wolfers and his colleagues.
The main limitations of the study were the inability to control for confounding variables and to make conclusions about causality, the researchers said. However, the absence of overlap in patients with the same disorder supports the use of brain mapping to study individual pathophysiologic signatures in schizophrenia and bipolar disorder patients, they concluded.
Mr. Wolfers had no financial conflicts to disclose. The study was supported by grants from multiple organizations, including the Netherlands Organisation for Scientific Research and the Wellcome Trust U.K. Strategic Award.
SOURCE: Wolfers T et al. JAMA Psychiatry. 2018. doi: 10.1001/jamapsychiatry.2018.2467.
FROM JAMA PSYCHIATRY
Key clinical point: Technology that allows for brain mapping offers a framework for greater use of precision medicine in psychiatry.
Major finding: Few brain loci showed extreme deviations in more than 2% of patients with schizophrenia disorders in a brain mapping model.
Study details: The data come from MRI scans of 218 patients with schizophrenia or bipolar disorder and 256 healthy controls.
Disclosures: Mr. Wolfers had no financial conflicts to disclose. The study was supported by grants from multiple organizations, including the Netherlands Organisation for Scientific Research and the Wellcome Trust U.K. Strategic Award.
Source: Wolfers T et al. JAMA Psychiatry. 2018. doi: 10.1001/jamapsychiatry.2018.2467.
Inflammation in schizophrenia tied to poor functioning
Targeting inflammation in people with schizophrenia could lead to improvements in daily functioning, a study by Sophia Kogan, MD, PhD, and her associates at the Icahn School of Medicine at Mount Sinai, New York, has suggested.
“Our findings invite speculation about the underlying link between inflammation and neurocognitive dysfunction in schizophrenia,” Dr. Kogan and her associates wrote in Brain, Behavior, and Immunity. “Our results suggest that these associations also extend to poor daily functioning in this population.”
to analyze neurocognition, functions of daily living, and inflammatory markers in 41 adults aged 18-55 years with schizophrenia, schizoaffective disorder, or psychosis. Among the individuals in the trial, 36% were female, the average age was 37 years, and the average body mass index was 31 kg/m2. In addition, all of the individuals were either taking antipsychotics or were on doses of injectable depot antipsychotics. People with active substance use and mild depressive symptoms were excluded.
Dr. Kogan and her associates found that poorer neurocognition was tied to increased levels of peripheral tumor necrosis factor–alpha (TNF-a) and interleukin-12 (IL-12p70).
“For TNF-a, these findings were driven primarily by significant associations with poorer neurocognitive performance in the domains of speed of processing (P = .02), visual learning (P = .02), and reasoning and problem solving (P = .03),” wrote Dr. Kogan and her associates. “Similarly, the association between neurocognition and IL-12p70 was driven primarily by decreased speed of processing (P less than .01) and visual learning (P = .05).”
The small sample size was cited as a limitation, as was the exclusion of people with substance use and mild depressive symptoms.
The National Institute of Mental Health funded the study. Dr. Kogan reported no conflicts of interest. Coauthor David Kimhy, PhD, is a consultant to NeuroCog Trials relating to another project.
SOURCE: Kogan S et al. Brain Behav Immun. 2018 Sep 12. doi: 10.1016/j.bbi.2018.09.016.
Targeting inflammation in people with schizophrenia could lead to improvements in daily functioning, a study by Sophia Kogan, MD, PhD, and her associates at the Icahn School of Medicine at Mount Sinai, New York, has suggested.
“Our findings invite speculation about the underlying link between inflammation and neurocognitive dysfunction in schizophrenia,” Dr. Kogan and her associates wrote in Brain, Behavior, and Immunity. “Our results suggest that these associations also extend to poor daily functioning in this population.”
to analyze neurocognition, functions of daily living, and inflammatory markers in 41 adults aged 18-55 years with schizophrenia, schizoaffective disorder, or psychosis. Among the individuals in the trial, 36% were female, the average age was 37 years, and the average body mass index was 31 kg/m2. In addition, all of the individuals were either taking antipsychotics or were on doses of injectable depot antipsychotics. People with active substance use and mild depressive symptoms were excluded.
Dr. Kogan and her associates found that poorer neurocognition was tied to increased levels of peripheral tumor necrosis factor–alpha (TNF-a) and interleukin-12 (IL-12p70).
“For TNF-a, these findings were driven primarily by significant associations with poorer neurocognitive performance in the domains of speed of processing (P = .02), visual learning (P = .02), and reasoning and problem solving (P = .03),” wrote Dr. Kogan and her associates. “Similarly, the association between neurocognition and IL-12p70 was driven primarily by decreased speed of processing (P less than .01) and visual learning (P = .05).”
The small sample size was cited as a limitation, as was the exclusion of people with substance use and mild depressive symptoms.
The National Institute of Mental Health funded the study. Dr. Kogan reported no conflicts of interest. Coauthor David Kimhy, PhD, is a consultant to NeuroCog Trials relating to another project.
SOURCE: Kogan S et al. Brain Behav Immun. 2018 Sep 12. doi: 10.1016/j.bbi.2018.09.016.
Targeting inflammation in people with schizophrenia could lead to improvements in daily functioning, a study by Sophia Kogan, MD, PhD, and her associates at the Icahn School of Medicine at Mount Sinai, New York, has suggested.
“Our findings invite speculation about the underlying link between inflammation and neurocognitive dysfunction in schizophrenia,” Dr. Kogan and her associates wrote in Brain, Behavior, and Immunity. “Our results suggest that these associations also extend to poor daily functioning in this population.”
to analyze neurocognition, functions of daily living, and inflammatory markers in 41 adults aged 18-55 years with schizophrenia, schizoaffective disorder, or psychosis. Among the individuals in the trial, 36% were female, the average age was 37 years, and the average body mass index was 31 kg/m2. In addition, all of the individuals were either taking antipsychotics or were on doses of injectable depot antipsychotics. People with active substance use and mild depressive symptoms were excluded.
Dr. Kogan and her associates found that poorer neurocognition was tied to increased levels of peripheral tumor necrosis factor–alpha (TNF-a) and interleukin-12 (IL-12p70).
“For TNF-a, these findings were driven primarily by significant associations with poorer neurocognitive performance in the domains of speed of processing (P = .02), visual learning (P = .02), and reasoning and problem solving (P = .03),” wrote Dr. Kogan and her associates. “Similarly, the association between neurocognition and IL-12p70 was driven primarily by decreased speed of processing (P less than .01) and visual learning (P = .05).”
The small sample size was cited as a limitation, as was the exclusion of people with substance use and mild depressive symptoms.
The National Institute of Mental Health funded the study. Dr. Kogan reported no conflicts of interest. Coauthor David Kimhy, PhD, is a consultant to NeuroCog Trials relating to another project.
SOURCE: Kogan S et al. Brain Behav Immun. 2018 Sep 12. doi: 10.1016/j.bbi.2018.09.016.
FROM BRAIN, BEHAVIOR, AND IMMUNITY
Antipsychotics for patients with dementia: The road less traveled
As psychiatrists treating an aging population, we frequently face the daunting challenges of managing medically complex and behaviorally unstable patients whose fragile condition tests the brightest among us. As our population enters late life, not only are physicians confronted with aging patients whose bodies have decreased renal and hepatic function, but we also face the challenges of the aging brain, severed neuronal networks, and neurotransmitter diminution. These physiological changes can alter treatment response, increase the frequency of adverse effects, and increase the likelihood of emergence of behavioral and psychological symptoms.
During the past decade, the number of people reaching age 65 has dramatically increased. As life expectancy improves, the “oldest old”—those age 85 and older—are the fastest-growing segment of the population. The prevalence of cognitive impairment, including mild cognitive impairment and dementia, in this cohort is >40%.1 Roughly 90% of patients with dementia will develop clinically significant behavioral problems at some point in the course of their illness.2
Behavioral and psychological symptoms of dementia (BPSD) have a tremendous impact on the quality of life for both patients and their caregivers. We are experts in understanding these behaviors and crafting nonpharmacologic treatment plans to manage them. Understanding the context in which behaviors emerge allows us to modify the environment, communication strategies, and other potential triggers, in turn reducing the need for pharmacologic intervention.
However, when nonpharmacologic interventions have been exhausted, what are the options? Antipsychotics have been one of the approaches used to address the challenges of behavioral disturbances and psychosis occurring in dementia. Unfortunately, there is conflicting evidence regarding the risks and benefits associated with the use of antipsychotics in this population. In this article, we provide a roadmap for the judicious use of antipsychotics for patients with dementia.
Weighing the risks and benefits of antipsychotics
Until better treatment options become available, second-generation antipsychotics (SGAs) continue to have an important but limited role in the treatment of behavioral disturbances in dementia. Although safety risks exist, they can be minimized through the careful selection of appropriate patients for treatment, close monitoring, and effective communication with patients and caregivers before and during treatment.
Several studies examining the efficacy of antipsychotics in the treatment of BPSD have demonstrated an increased risk of cerebrovascular events, including stroke and death due to any cause.3 This evidence prompted the FDA to issue a “black-box” warning in 2005 to highlight the increased risk of mortality for patients with dementia who are treated with SGAs.4 Both first-generation antipsychotics (FGAs) and SGAs have been associated with higher rates of mortality than most other psychotropic classes, except anticonvulsants. This increased mortality risk has been shown to persist for at least 6 to 12 months.5,6 FGAs appear to be associated with a greater mortality risk compared with SGAs. As a result, if antipsychotic treatment is necessary, the use of FGAs in this population is not recommended.
The potential mechanisms leading to stroke and death remain unclear. They could include orthostatic hypotension, anticholinergic adverse effects, QT prolongation, platelet aggregation effects, and venous thromboembolism. The presence of cardiovascular and vascular risk factors, electrolyte imbalances, cardiac arrhythmias, and concomitant use of medications that prolong the QTc interval may confer additional risks.
Continued to: Although the use of antipsychotics for patients with dementia...
Although the use of antipsychotics for patients with dementia may increase the risk of mortality, the absolute increased risk to a given individual, at least with short-term treatment, is likely small. The risk may also vary depending on the choice of SGA. Patients who were treated with quetiapine had a slightly lower risk of death than those who were treated risperidone.5 Death rates among patients prescribed aripiprazole, olanzapine, and ziprasidone were similar to the death rates of patients who were treated with risperidone. Compared with patients who were treated with risperidone, patients who were treated with the FGA haloperidol were twice as likely to die during a subsequent 6-month observation period. The largest number of deaths occurred during the first 40 days of treatment.5
While this increased risk of mortality is an important factor to discuss with patients and caregivers when deciding whether to initiate antipsychotic treatment, it is also important to put it into perspective. For example, the risk of suddenly dying from a stroke or heart attack for a person with dementia who is not taking an antipsychotic is approximately 2%. When an individual is started on one of these agents, that risk increases to approximately 4%. While the mortality risk is doubled, it remains relatively small.4 When faced with verbal or physical assaults, hostility, paranoid ideations, or other psychotic symptoms, many families feel that this relatively low risk does not outweigh the potential benefits of reducing caregiver and patient distress. If nonpharmacologic and/or other pharmacologic interventions have failed, the treatment has reached a point of no good alternatives and therapy should then focus on minimizing risk.
Informed consent is essential. A discussion of risks and benefits with the patient, family, or other decision-makers should focus on the risk of stroke, potential metabolic effects, and mortality, as well as potential worsening of cognitive decline associated with antipsychotic treatment. This should be weighed together with the evidence that suggests psychosis and agitation are associated with earlier nursing home admission and death.7,8 Families should be given ample time and opportunity to ask questions. Alternatives to immediate initiation of antipsychotics should be thoroughly reviewed.
Despite the above-noted risks, expert consensus suggests that the use of antipsychotics in the treatment of individuals with dementia can be appropriate, particularly in individuals with dangerous agitation or psychosis.9 These agents can minimize the risk of violence, reduce patient distress, improve the patient’s quality of life, and reduce caregiver burden. In clinical trials, the benefits of antipsychotics have been modest. Nevertheless, evidence has shown that these agents can reduce psychosis, agitation, aggression, hostility, and suspiciousness, which makes them a valid option when other interventions have proven insufficient.
Target specific symptoms
Despite this article’s focus on the appropriate use of antipsychotics for patients with BPSD, it is important to emphasize that the first-line approach to the management of BPSD in this population should always be a person-centered, psychosocial, multidisciplinary, nonpharmacologic approach that focuses on identifying triggers and treating potentially modifiable contributors to behavioral symptoms. Table 110 outlines common underlying causes of BPSD in dementia that should be assessed before prescribing an antipsychotic.
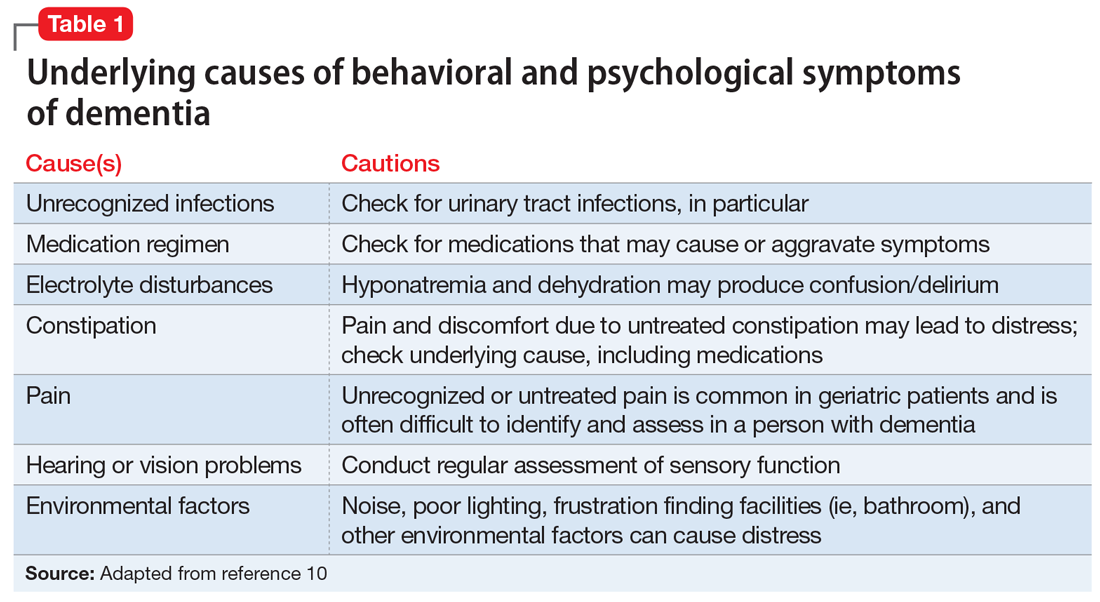
Continued to: Alternative psychopharmacologic treatments...
Alternative psychopharmacologic treatments based on a psychobehavioral metaphor should also be considered (Table 211). This approach matches the dominant target symptoms to the most relevant medication class.11 For example, in the case of a verbally and physically agitated patient who is also irritable, negative, socially withdrawn, and appears dysphoric, we might first undertake a trial of an antidepressant. Conversely, if the patient shows agitation in the context of increased motor activity, loud and rapid speech, and affective lability, we might consider the use of a mood stabilizer. Pharmacologic treatment should be aimed at the modification of clearly identified and documented target behaviors.

Indications to use antipsychotics for patients with dementia include:
- severe agitation and aggression associated with risk of harm
- delusions and hallucinations
- comorbid preexisting mental health conditions (eg, bipolar disorder, schizophrenia, treatment-resistant depression, etc.).
Symptoms that do not usually respond to an antipsychotic include wandering, social withdrawal, shouting, pacing, touching, cognitive defects, and incontinence.12 These symptoms may respond to interventions such as changes to the environment.
Continued to: Choosing an antipsychotic
Choosing an antipsychotic
Once you have identified that an antipsychotic is truly indicated, the choice of an agent will focus on patient-related factors. Considerations such as frailty, comorbid medical conditions including diabetes, history of falls, hepatic insufficiency, cardiac arrhythmias, and cerebrovascular risk factors, should all be analyzed prior to initiating an antipsychotic. The presence of these conditions will increase the likelihood that adverse effects may occur. It will also guide the dose trajectory and the target dose for discontinuation. Antipsychotics differ with respect to their efficacy and adverse effect profile. For practical purposes, adverse effects typically guide the selection of these agents when used for patients with dementia.
Continued to: Gradual structural changes occur...
Gradual structural changes occur in the dopaminergic system with age and increase the propensity for antipsychotic adverse effects. The number of dopaminergic neurons and D2 receptors decreases approximately 10% per decade. In order to avoid the development of adverse effects related to extrapyramidal symptoms, approximately 20% of receptors need to be free. FGAs tend to block approximately 90% of D2 receptors, whereas SGAs block less than 70% to 80% and dissociate more rapidly from D2 receptors.13 FGAs should therefore be avoided, as they have been associated with numerous adverse effects, including parkinsonism, tardive dyskinesia, akathisia, sedation, peripheral and central anticholinergic effects, postural hypotension, cardiac conduction defects, and falls. As noted above, they have been linked to a greater risk of mortality (Figure14 ).
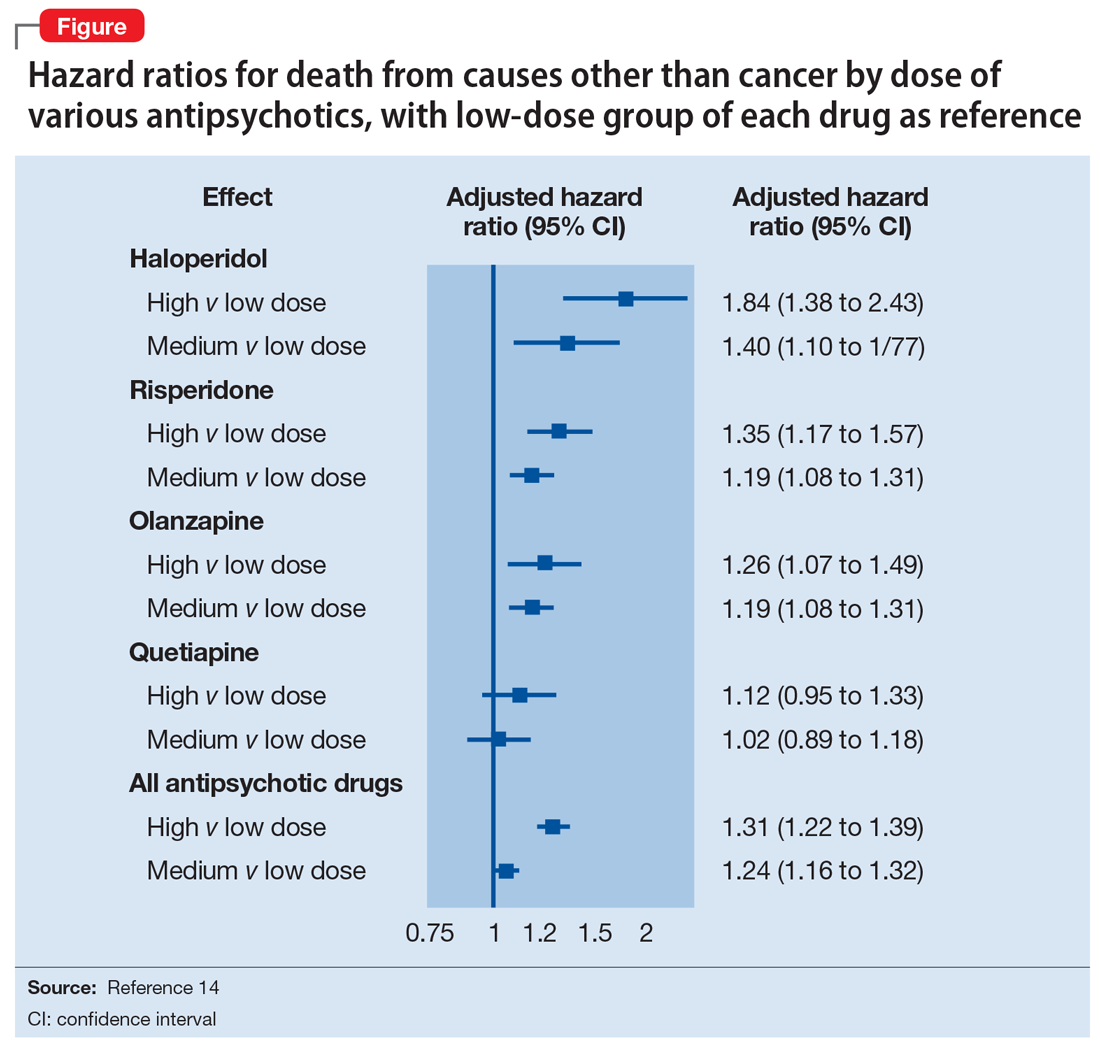
When the decision to use an antipsychotic agent is made for a person with dementia, SGAs appear to be a better choice. There appear to be modest differences within the class of SGAs in terms of effectiveness, tolerability, and adverse effect profile. Although the association between the dose of an antipsychotic and the risk of mortality or stroke remains undefined, other common adverse effects, such as sedation, extrapyramidal symptoms, and risk of falls, can be reduced by starting at the lowest dose possible and titrating slowly.
Dosing considerations
Dose increments should be modest and, in a nonemergent setting, may be adjusted at weekly intervals depending on response. Prior to starting a treatment trial, it is advisable to estimate what will constitute a worthwhile clinical response, the duration of treatment, and the maximum dose. Avoid high doses or prolonged use of antipsychotics that have not significantly improved the target behavior.
When the decision to use a SGA is made, choosing the initial starting dose is challenging given that none of these medications has an indication for use in this population. We propose doses that have been used in completed randomized trials that reflect the best information available about the dose likely to maximize benefit and minimize risk. On the basis of those trials, reasonable starting doses would be15-22:
- quetiapine 25 to 50 mg/d
- risperidone 0.5 to 1 mg/d
- aripiprazole 2 to 10 mg/d
- olanzapine 2.5 to 5 mg/d
- ziprasidone 20 mg/d
Continued to: The highest doses tested...
The highest doses tested for each of these compounds in randomized clinical trials for this population were: risperidone 2 mg/d, olanzapine 10 mg/d, and aripiprazole 15 mg/d. A wide variety of maximum doses of quetiapine were studied in clinical trials, with a top dose of 200 mg being most common. It is worth noting that doses higher than these have been used for other indications.15-22
Quetiapine. One of the most commonly prescribed antipsychotics for the treatment of BPSD in individuals with memory disorders is quetiapine. The reasons for this preference include a low risk of extrapyramidal adverse effects, flexibility of dosing, ability to use lower dosages, and evidence of the lower risk of mortality when compared with other second-generation agents.5,15 If an antipsychotic is indicated, quetiapine should be considered as a first-line antipsychotic therapy. Quetiapine has well-established effects on mood, anxiety, and sleep, all of which can be disrupted in dementia and can act as drivers for agitation.5,15 Starting quetiapine may mitigate the need for separate agents to treat insomnia, loss of appetite, or anxiety, although it is not FDA-indicated for these comorbid conditions. Quetiapine is also less likely to exacerbate motor symptoms compared with other SGAs but has the potential to increase the risk of falls, and orthostasis, and carries a considerable anticholinergic burden.5,15
Risperidone has been shown to provide modest improvements in some people exhibiting symptoms of aggression, agitation, and psychosis.5,15 There is no evidence that risperidone is any more effective than other SGAs, but it has been tested on more geriatric patients than other SGAs. The fact that it is also available in an orally disintegrating tablet makes it a practical treatment in certain populations of patients, such as those who have difficulty swallowing. Risperidone carries the highest extrapyramidal symptom burden among the SGAs due to its potent D2 receptor binding. 5,15
Aripiprazole. There have been several studies of aripiprazole for the treatment of psychosis and agitation in Alzheimer’s dementia.15 This medication showed modest effect and was generally well tolerated. Aripiprazole appears to have less associated weight gain, which may be pertinent for some patients. It also appears to be less sedating than many of the other SGAs. However, some patients may experience activation or insomnia with this agent, particularly with doses <15 mg/d. This activating effect may be beneficial for treating comorbid depressive symptoms, although lower doses could theoretically worsen psychosis due to the activating effects.
Aripiprazole has also been studied in Parkinson’s disease. While some patients had favorable responses with improvement in psychosis and behavioral disturbances, this medication was also associated with worsening of motor symptoms. Certain individuals also experienced a worsening of their psychosis.23 For this reason, it is unlikely to be a useful agent for patients displaying evidence of parkinsonism, Parkinson’s dementia, or dementia with Lewy bodies.
Olanzapine. Several studies have shown that low-dose olanzapine has been modestly effective in decreasing agitation and aggression in patients suffering from Alzheimer’s and vascular dementias.24 The medication is also available in an orally disintegrating form, which may be beneficial when treating individuals whose swallowing abilities are compromised. Olanzapine also has been associated with significant weight gain and metabolic syndrome.24
Continued to: Ziprasidone
Ziprasidone. There are no specific studies of ziprasidone for geriatric patients and none for patients with dementia. However, case reports have suggested both oral and injectable forms of the medication may be well tolerated and have some benefit in treating agitation in this population.25 Based on evidence from younger populations, ziprasidone is less likely to be associated with weight gain or orthostatic hypotension. Medication has been associated with QTc prolongation and should be used with caution and monitored with an ECG.
The initial dosing and potential adverse effects of quetiapine, risperidone, aripiprazole, olanzapine, and ziprasidone are highlighted in Table 3.10

Other SGAs. Newer antipsychotics have recently become available and may serve as additional tools for managing BPSD in the future. Unfortunately, there are currently no available studies regarding their efficacy in the treatment of agitation and psychosis in dementia. One notable exception is pimavaserin, a serotonin 2A receptor inverse agonist. This medication has recently been FDA-approved for the treatment of Parkinson’s disease psychosis. The medication was extensively studied in older patients. It appeared to be effective in reducing delusions and hallucinations while not impairing motor function or causing sedation or hypotension.23 Additional studies are currently ongoing for the treatment of Alzheimer’s dementia psychosis.
Monitor treatment, consider discontinuation
American Psychiatric Association guidelines on the use of antipsychotics to treat agitation or psychosis in patients with dementia currently recommend that clinicians use a quantitative measure to track symptoms and response to treatment.26 These measures may be formal, such as an overall assessment of symptom severity on a Likert scale, or as simple as monitoring the changes in the frequency of periods of agitation.
After starting an antipsychotic, a follow-up appointment should typically take place within 1 month. If the patient is at high risk for developing adverse effects, or if the symptoms are severe, a follow-up appointment for monitoring the response to treatment and potential adverse effects should occur within 1 week. At a minimum, expert consensus suggests follow-up visits should occur every 3 months.
If there is no clinical response after 4 weeks of adequate dosing of an antipsychotic, the medication should be tapered and withdrawn. Switching to an alternative agent may be appropriate.
Many patients will have only partial remission of target symptoms. Therefore, increasing the dose or switching to an alternative agent may be necessary. Concurrent use of multiple antipsychotic agents should be avoided.
Continued to: Maintenance treatment may be appropriate
Maintenance treatment may be appropriate for patients who have demonstrated a clear benefit from antipsychotic treatment without undue adverse effects, and in whom a trial dose reduction has resulted in reappearance of the target symptoms. A formal monitoring plan to assess changes in response and the significance of adverse effects should be in place. Review the target behavior, changes in function, and significance of adverse effects at least every 3 months.
How to approach discontinuation
Behavioral and psychological symptoms of dementia are frequently temporary. If the patient has been stable, gradual dose reduction and eventual discontinuation of antipsychotics should be attempted every 3 months. Studies have reported that most patients who were taken off antipsychotics for treating BPSD showed no worsening of behavioral symptoms.27
Discontinuation of antipsychotics should be done gradually by reducing the dose by 50% every 2 weeks, and then stopping after 2 weeks on the minimum dose, with monitoring for recurrence of target symptoms or emergence of new ones. The longer a medication has been prescribed, the slower the withdrawal occurs. Thus, the possibility of emerging symptoms related to drug withdrawal will lessen.
A roadmap for judicious prescribing

When underlying treatable or reversible causes of BPSD in dementia have been ruled out or nonpharmacologic treatments have failed, a trial of an antipsychotic may be indicated. The choice of agent should focus on patient-related factors and on clearly identified target behaviors. Treatment should be started at a low dose and titrated cautiously to the lowest effective dose.
Behavioral and psychological symptoms of dementia are frequently temporary. Therefore, a gradual reduction and eventual withdrawal of antipsychotic medications should be attempted every 3 months. Studies indicate that most patients are able to tolerate elimination of antipsychotic medications with no worsening of behavioral symptoms.
Despite the limitations of treatment, SGAs remain a valid consideration when other interventions have proven insufficient. However, judicious use of these agents remains the cornerstone of therapy.
Bottom Line
Until better treatment options become available, second-generation antipsychotics (SGAs) continue to have an important, albeit limited, role in the treatment of behavioral disturbances in dementia. Despite the limitations of treatment, SGAs remain a valid consideration when other interventions have proven insufficient. However, judicious use of these agents remains the cornerstone of therapy.
Related Resources
- Kales HC, Mulsant BH, Sajatovic MS. Prescribing antipsychotics in geriatric patients: Focus on dementia. Third of 3 parts. Current Psychiatry. 2017;16(12):24-30.
- Meeks TW, Jeste DV. Antipsychotics in dementia: Beyond ‘black-box’ warnings. Current Psychiatry. 2008;7(6):51-52, 55-58, 64-65.
Drug Brand Names
Aripiprazole • Abilify
Haloperidol • Haldol
Olanzapine • Zyprexa
Pimavanserin • Nuplazid
Risperidone • Risperdal
Quetiapine • Seroquel
Ziprasidone • Geodon
1. Gardner RC, Valcour V, Yaffe K. Dementia in the oldest old: a multi-factorial and growing public health issue. Alzheimers Res Ther. 2013;5(4):27.
2. Tariot PN, Blazina L. The psychopathology of dementia. In: Morris JC, ed. Handbook of dementing illnesses. New York, NY: Marcel Dekker Inc.; 1993:461-475.
3. Schneider LS, Dagerman KS, Insel P. Risk of death with atypical antipsychotic drug treatment for dementia: meta-analysis of randomized placebo-controlled trials. JAMA. 2005;294:1934-1943.
4. Lenzer J. FDA warns about using antipsychotic drugs for dementia. BMJ. 2005;330(7497):922.
5. Kales HC, Valenstein M, Kim HM, et al. Mortality risk in patients with dementia treated with antipsychotics versus other psychiatric medications. Am J Psychiatry. 2007;164(10):1568-1576; quiz 1623.
6. Gill SS, Bronskill SE, Normand SL, et al. Antipsychotic drug use and mortality in older adults with dementia. Ann Intern Med. 2007;146(11):775-786.
7. Okura T, Plassman BL, Steffens DC, et al. Neuropsychiatric symptoms and the risk of institutionalization and death: the aging, demographics, and memory study. J Am Geriatr Soc. 2011;59:473-481.
8. Banerjee S, Murray J, Foley B, et al. Predictors of institutionalisation in people with dementia. J Neurol Neurosurg Psychiatry. 2003;74:1315-1316.
9. Alexopoulos GS, Jeste DV, Chung H, et al. The expert consensus guideline series. Treatment of dementia and its behavioral disturbances. Introduction: methods, commentary, and summary. Postgrad Med. 2005;Spec No:6-22.
10. Burke AD, Hall G, Yaari R, et al. Pocket reference to Alzheimer’s disease management. Philadelphia, PA: Springer Healthcare Communications; 2015:39-46
11. Burke AD, Burke WJ, Tariot PN. Drug treatments for the behavioural and psychiatric symptoms of dementia. In: Ames D, O’Brien JT, Burns A, eds. Dementia, 5th ed. Boca Raton, FL: CRC Press; 2016:231-252.
12. Royal Australian and New Zealand College of Psychiatrists. Antipsychotics in dementia: best practice guide. https://bpac.org.nz/a4d/resources/docs/bpac_A4D_best_practice_guide.pdf. Accessed September 4, 2018.
13. Nyberg L, Backman L. Cognitive aging: a view from brain imaging. In: Dixon RA, Backman L, Nilsson LG, eds. New frontiers in cognitive aging. Oxford: Oxford Univ Press; 2004:135-60.
14. Huybrechts KF, Gerhard T, Crystal S, et al. Differential risk of death in older residents in nursing homes prescribed specific antipsychotic drugs: population based cohort study. BMJ. 2012;344:e977. doi: 10.1136/bmj.e977.
15. Burke AD, Tariot PN. Atypical antipsychotics in the elderly: a review of therapeutic trends and clinical outcomes. Expert Opin Pharmacother. 2009;10(15):2407-2414.
16. De Deyn PP, Rabheru K, Rasmussen A, et al. A randomized trial of risperidone, placebo, and haloperidol for behavioral symptoms of dementia. Neurology.1999;53(5):946-955.
17. De Deyn PP, Jeste DV, Auby P, et al. Aripiprazole in dementia of the Alzheimer’s type. Poster presented at: 16th Annual Meeting of American Association for Geriatric Psychiatry; March 1-4, 2003; Honolulu, HI.
18. Lopez OL, Becker JT, Chang YF, et al. The long-term effects of conventional and atypical antipsychotics in patients with probable Alzheimer’s disease. Am J Psychiatry. 2013;170(9):1051-1058.
19. Mintzer J, Weiner M, Greenspan A, et al. Efficacy and safety of a flexible dose of risperidone versus placebo in the treatment of psychosis of Alzheimer’s disease. In: International College of Geriatric Psychopharmacology. Basel, Switzerland; 2004.
20. Mintzer JE, Tune LE, Breder CD, et al. Aripiprazole for the treatment of psychoses in institutionalized patients with Alzheimer dementia: a multicenter, randomized, double-blind, placebo-controlled assessment of three fixed doses. Am J Geriatr Psychiatry. 2007;15(11):918-931.
21. Sultzer DL, Davis SM, Tariot PN, et al; CATIE-AD Study Group. Clinical symptom responses to atypical antipsychotic medications in Alzheimer’s disease: phase 1 outcomes from the CATIE-AD effectiveness trial. Am J Psychiatry. 2008;165(7):844-854.
22. Zhong KX, Tariot PN, Mintzer J, et al. Quetiapine to treat agitation in dementia: a randomized, double-blind, placebo-controlled study. Curr Alzheimer Res. 2007;4(1):81-93.
23. Bozymski KM, Lowe DK, Pasternak KM, et al. Pimavanserin: a novel antipsychotic for Parkinson’s disease psychosis. Ann Pharmacother. 2017;51(6):479-487.
24. Moretti R, Torre R, Antonello T, et al. Olanzapine as a possible treatment of behavioral symptoms in vascular dementia: risks of cerebrovascular events. J Neurol. 2005;252:1186.
25. Cole SA, Saleem R, Shea WP, et al. Ziprasidone for agitation or psychosis in dementia: four cases. Int J Psychiatry Med. 2005;35(1):91-98.
26. Reus VI, Fochtmann LJ, Eyler AE, et al. The American Psychiatric Association practice guideline on the use of antipsychotics to treat agitation or psychosis in patients with dementia. Am J Psychiatry. 2016;173(5):543-546.
27. Horwitz GJ, Tariot PN, Mead K, et al. Discontinuation of antipsychotics in nursing home patients with dementia. Am J Geriatr Psychiatry. 1995;3(4):290-299.
As psychiatrists treating an aging population, we frequently face the daunting challenges of managing medically complex and behaviorally unstable patients whose fragile condition tests the brightest among us. As our population enters late life, not only are physicians confronted with aging patients whose bodies have decreased renal and hepatic function, but we also face the challenges of the aging brain, severed neuronal networks, and neurotransmitter diminution. These physiological changes can alter treatment response, increase the frequency of adverse effects, and increase the likelihood of emergence of behavioral and psychological symptoms.
During the past decade, the number of people reaching age 65 has dramatically increased. As life expectancy improves, the “oldest old”—those age 85 and older—are the fastest-growing segment of the population. The prevalence of cognitive impairment, including mild cognitive impairment and dementia, in this cohort is >40%.1 Roughly 90% of patients with dementia will develop clinically significant behavioral problems at some point in the course of their illness.2
Behavioral and psychological symptoms of dementia (BPSD) have a tremendous impact on the quality of life for both patients and their caregivers. We are experts in understanding these behaviors and crafting nonpharmacologic treatment plans to manage them. Understanding the context in which behaviors emerge allows us to modify the environment, communication strategies, and other potential triggers, in turn reducing the need for pharmacologic intervention.
However, when nonpharmacologic interventions have been exhausted, what are the options? Antipsychotics have been one of the approaches used to address the challenges of behavioral disturbances and psychosis occurring in dementia. Unfortunately, there is conflicting evidence regarding the risks and benefits associated with the use of antipsychotics in this population. In this article, we provide a roadmap for the judicious use of antipsychotics for patients with dementia.
Weighing the risks and benefits of antipsychotics
Until better treatment options become available, second-generation antipsychotics (SGAs) continue to have an important but limited role in the treatment of behavioral disturbances in dementia. Although safety risks exist, they can be minimized through the careful selection of appropriate patients for treatment, close monitoring, and effective communication with patients and caregivers before and during treatment.
Several studies examining the efficacy of antipsychotics in the treatment of BPSD have demonstrated an increased risk of cerebrovascular events, including stroke and death due to any cause.3 This evidence prompted the FDA to issue a “black-box” warning in 2005 to highlight the increased risk of mortality for patients with dementia who are treated with SGAs.4 Both first-generation antipsychotics (FGAs) and SGAs have been associated with higher rates of mortality than most other psychotropic classes, except anticonvulsants. This increased mortality risk has been shown to persist for at least 6 to 12 months.5,6 FGAs appear to be associated with a greater mortality risk compared with SGAs. As a result, if antipsychotic treatment is necessary, the use of FGAs in this population is not recommended.
The potential mechanisms leading to stroke and death remain unclear. They could include orthostatic hypotension, anticholinergic adverse effects, QT prolongation, platelet aggregation effects, and venous thromboembolism. The presence of cardiovascular and vascular risk factors, electrolyte imbalances, cardiac arrhythmias, and concomitant use of medications that prolong the QTc interval may confer additional risks.
Continued to: Although the use of antipsychotics for patients with dementia...
Although the use of antipsychotics for patients with dementia may increase the risk of mortality, the absolute increased risk to a given individual, at least with short-term treatment, is likely small. The risk may also vary depending on the choice of SGA. Patients who were treated with quetiapine had a slightly lower risk of death than those who were treated risperidone.5 Death rates among patients prescribed aripiprazole, olanzapine, and ziprasidone were similar to the death rates of patients who were treated with risperidone. Compared with patients who were treated with risperidone, patients who were treated with the FGA haloperidol were twice as likely to die during a subsequent 6-month observation period. The largest number of deaths occurred during the first 40 days of treatment.5
While this increased risk of mortality is an important factor to discuss with patients and caregivers when deciding whether to initiate antipsychotic treatment, it is also important to put it into perspective. For example, the risk of suddenly dying from a stroke or heart attack for a person with dementia who is not taking an antipsychotic is approximately 2%. When an individual is started on one of these agents, that risk increases to approximately 4%. While the mortality risk is doubled, it remains relatively small.4 When faced with verbal or physical assaults, hostility, paranoid ideations, or other psychotic symptoms, many families feel that this relatively low risk does not outweigh the potential benefits of reducing caregiver and patient distress. If nonpharmacologic and/or other pharmacologic interventions have failed, the treatment has reached a point of no good alternatives and therapy should then focus on minimizing risk.
Informed consent is essential. A discussion of risks and benefits with the patient, family, or other decision-makers should focus on the risk of stroke, potential metabolic effects, and mortality, as well as potential worsening of cognitive decline associated with antipsychotic treatment. This should be weighed together with the evidence that suggests psychosis and agitation are associated with earlier nursing home admission and death.7,8 Families should be given ample time and opportunity to ask questions. Alternatives to immediate initiation of antipsychotics should be thoroughly reviewed.
Despite the above-noted risks, expert consensus suggests that the use of antipsychotics in the treatment of individuals with dementia can be appropriate, particularly in individuals with dangerous agitation or psychosis.9 These agents can minimize the risk of violence, reduce patient distress, improve the patient’s quality of life, and reduce caregiver burden. In clinical trials, the benefits of antipsychotics have been modest. Nevertheless, evidence has shown that these agents can reduce psychosis, agitation, aggression, hostility, and suspiciousness, which makes them a valid option when other interventions have proven insufficient.
Target specific symptoms
Despite this article’s focus on the appropriate use of antipsychotics for patients with BPSD, it is important to emphasize that the first-line approach to the management of BPSD in this population should always be a person-centered, psychosocial, multidisciplinary, nonpharmacologic approach that focuses on identifying triggers and treating potentially modifiable contributors to behavioral symptoms. Table 110 outlines common underlying causes of BPSD in dementia that should be assessed before prescribing an antipsychotic.

Continued to: Alternative psychopharmacologic treatments...
Alternative psychopharmacologic treatments based on a psychobehavioral metaphor should also be considered (Table 211). This approach matches the dominant target symptoms to the most relevant medication class.11 For example, in the case of a verbally and physically agitated patient who is also irritable, negative, socially withdrawn, and appears dysphoric, we might first undertake a trial of an antidepressant. Conversely, if the patient shows agitation in the context of increased motor activity, loud and rapid speech, and affective lability, we might consider the use of a mood stabilizer. Pharmacologic treatment should be aimed at the modification of clearly identified and documented target behaviors.

Indications to use antipsychotics for patients with dementia include:
- severe agitation and aggression associated with risk of harm
- delusions and hallucinations
- comorbid preexisting mental health conditions (eg, bipolar disorder, schizophrenia, treatment-resistant depression, etc.).
Symptoms that do not usually respond to an antipsychotic include wandering, social withdrawal, shouting, pacing, touching, cognitive defects, and incontinence.12 These symptoms may respond to interventions such as changes to the environment.
Continued to: Choosing an antipsychotic
Choosing an antipsychotic
Once you have identified that an antipsychotic is truly indicated, the choice of an agent will focus on patient-related factors. Considerations such as frailty, comorbid medical conditions including diabetes, history of falls, hepatic insufficiency, cardiac arrhythmias, and cerebrovascular risk factors, should all be analyzed prior to initiating an antipsychotic. The presence of these conditions will increase the likelihood that adverse effects may occur. It will also guide the dose trajectory and the target dose for discontinuation. Antipsychotics differ with respect to their efficacy and adverse effect profile. For practical purposes, adverse effects typically guide the selection of these agents when used for patients with dementia.
Continued to: Gradual structural changes occur...
Gradual structural changes occur in the dopaminergic system with age and increase the propensity for antipsychotic adverse effects. The number of dopaminergic neurons and D2 receptors decreases approximately 10% per decade. In order to avoid the development of adverse effects related to extrapyramidal symptoms, approximately 20% of receptors need to be free. FGAs tend to block approximately 90% of D2 receptors, whereas SGAs block less than 70% to 80% and dissociate more rapidly from D2 receptors.13 FGAs should therefore be avoided, as they have been associated with numerous adverse effects, including parkinsonism, tardive dyskinesia, akathisia, sedation, peripheral and central anticholinergic effects, postural hypotension, cardiac conduction defects, and falls. As noted above, they have been linked to a greater risk of mortality (Figure14 ).

When the decision to use an antipsychotic agent is made for a person with dementia, SGAs appear to be a better choice. There appear to be modest differences within the class of SGAs in terms of effectiveness, tolerability, and adverse effect profile. Although the association between the dose of an antipsychotic and the risk of mortality or stroke remains undefined, other common adverse effects, such as sedation, extrapyramidal symptoms, and risk of falls, can be reduced by starting at the lowest dose possible and titrating slowly.
Dosing considerations
Dose increments should be modest and, in a nonemergent setting, may be adjusted at weekly intervals depending on response. Prior to starting a treatment trial, it is advisable to estimate what will constitute a worthwhile clinical response, the duration of treatment, and the maximum dose. Avoid high doses or prolonged use of antipsychotics that have not significantly improved the target behavior.
When the decision to use a SGA is made, choosing the initial starting dose is challenging given that none of these medications has an indication for use in this population. We propose doses that have been used in completed randomized trials that reflect the best information available about the dose likely to maximize benefit and minimize risk. On the basis of those trials, reasonable starting doses would be15-22:
- quetiapine 25 to 50 mg/d
- risperidone 0.5 to 1 mg/d
- aripiprazole 2 to 10 mg/d
- olanzapine 2.5 to 5 mg/d
- ziprasidone 20 mg/d
Continued to: The highest doses tested...
The highest doses tested for each of these compounds in randomized clinical trials for this population were: risperidone 2 mg/d, olanzapine 10 mg/d, and aripiprazole 15 mg/d. A wide variety of maximum doses of quetiapine were studied in clinical trials, with a top dose of 200 mg being most common. It is worth noting that doses higher than these have been used for other indications.15-22
Quetiapine. One of the most commonly prescribed antipsychotics for the treatment of BPSD in individuals with memory disorders is quetiapine. The reasons for this preference include a low risk of extrapyramidal adverse effects, flexibility of dosing, ability to use lower dosages, and evidence of the lower risk of mortality when compared with other second-generation agents.5,15 If an antipsychotic is indicated, quetiapine should be considered as a first-line antipsychotic therapy. Quetiapine has well-established effects on mood, anxiety, and sleep, all of which can be disrupted in dementia and can act as drivers for agitation.5,15 Starting quetiapine may mitigate the need for separate agents to treat insomnia, loss of appetite, or anxiety, although it is not FDA-indicated for these comorbid conditions. Quetiapine is also less likely to exacerbate motor symptoms compared with other SGAs but has the potential to increase the risk of falls, and orthostasis, and carries a considerable anticholinergic burden.5,15
Risperidone has been shown to provide modest improvements in some people exhibiting symptoms of aggression, agitation, and psychosis.5,15 There is no evidence that risperidone is any more effective than other SGAs, but it has been tested on more geriatric patients than other SGAs. The fact that it is also available in an orally disintegrating tablet makes it a practical treatment in certain populations of patients, such as those who have difficulty swallowing. Risperidone carries the highest extrapyramidal symptom burden among the SGAs due to its potent D2 receptor binding. 5,15
Aripiprazole. There have been several studies of aripiprazole for the treatment of psychosis and agitation in Alzheimer’s dementia.15 This medication showed modest effect and was generally well tolerated. Aripiprazole appears to have less associated weight gain, which may be pertinent for some patients. It also appears to be less sedating than many of the other SGAs. However, some patients may experience activation or insomnia with this agent, particularly with doses <15 mg/d. This activating effect may be beneficial for treating comorbid depressive symptoms, although lower doses could theoretically worsen psychosis due to the activating effects.
Aripiprazole has also been studied in Parkinson’s disease. While some patients had favorable responses with improvement in psychosis and behavioral disturbances, this medication was also associated with worsening of motor symptoms. Certain individuals also experienced a worsening of their psychosis.23 For this reason, it is unlikely to be a useful agent for patients displaying evidence of parkinsonism, Parkinson’s dementia, or dementia with Lewy bodies.
Olanzapine. Several studies have shown that low-dose olanzapine has been modestly effective in decreasing agitation and aggression in patients suffering from Alzheimer’s and vascular dementias.24 The medication is also available in an orally disintegrating form, which may be beneficial when treating individuals whose swallowing abilities are compromised. Olanzapine also has been associated with significant weight gain and metabolic syndrome.24
Continued to: Ziprasidone
Ziprasidone. There are no specific studies of ziprasidone for geriatric patients and none for patients with dementia. However, case reports have suggested both oral and injectable forms of the medication may be well tolerated and have some benefit in treating agitation in this population.25 Based on evidence from younger populations, ziprasidone is less likely to be associated with weight gain or orthostatic hypotension. Medication has been associated with QTc prolongation and should be used with caution and monitored with an ECG.
The initial dosing and potential adverse effects of quetiapine, risperidone, aripiprazole, olanzapine, and ziprasidone are highlighted in Table 3.10

Other SGAs. Newer antipsychotics have recently become available and may serve as additional tools for managing BPSD in the future. Unfortunately, there are currently no available studies regarding their efficacy in the treatment of agitation and psychosis in dementia. One notable exception is pimavaserin, a serotonin 2A receptor inverse agonist. This medication has recently been FDA-approved for the treatment of Parkinson’s disease psychosis. The medication was extensively studied in older patients. It appeared to be effective in reducing delusions and hallucinations while not impairing motor function or causing sedation or hypotension.23 Additional studies are currently ongoing for the treatment of Alzheimer’s dementia psychosis.
Monitor treatment, consider discontinuation
American Psychiatric Association guidelines on the use of antipsychotics to treat agitation or psychosis in patients with dementia currently recommend that clinicians use a quantitative measure to track symptoms and response to treatment.26 These measures may be formal, such as an overall assessment of symptom severity on a Likert scale, or as simple as monitoring the changes in the frequency of periods of agitation.
After starting an antipsychotic, a follow-up appointment should typically take place within 1 month. If the patient is at high risk for developing adverse effects, or if the symptoms are severe, a follow-up appointment for monitoring the response to treatment and potential adverse effects should occur within 1 week. At a minimum, expert consensus suggests follow-up visits should occur every 3 months.
If there is no clinical response after 4 weeks of adequate dosing of an antipsychotic, the medication should be tapered and withdrawn. Switching to an alternative agent may be appropriate.
Many patients will have only partial remission of target symptoms. Therefore, increasing the dose or switching to an alternative agent may be necessary. Concurrent use of multiple antipsychotic agents should be avoided.
Continued to: Maintenance treatment may be appropriate
Maintenance treatment may be appropriate for patients who have demonstrated a clear benefit from antipsychotic treatment without undue adverse effects, and in whom a trial dose reduction has resulted in reappearance of the target symptoms. A formal monitoring plan to assess changes in response and the significance of adverse effects should be in place. Review the target behavior, changes in function, and significance of adverse effects at least every 3 months.
How to approach discontinuation
Behavioral and psychological symptoms of dementia are frequently temporary. If the patient has been stable, gradual dose reduction and eventual discontinuation of antipsychotics should be attempted every 3 months. Studies have reported that most patients who were taken off antipsychotics for treating BPSD showed no worsening of behavioral symptoms.27
Discontinuation of antipsychotics should be done gradually by reducing the dose by 50% every 2 weeks, and then stopping after 2 weeks on the minimum dose, with monitoring for recurrence of target symptoms or emergence of new ones. The longer a medication has been prescribed, the slower the withdrawal occurs. Thus, the possibility of emerging symptoms related to drug withdrawal will lessen.
A roadmap for judicious prescribing

When underlying treatable or reversible causes of BPSD in dementia have been ruled out or nonpharmacologic treatments have failed, a trial of an antipsychotic may be indicated. The choice of agent should focus on patient-related factors and on clearly identified target behaviors. Treatment should be started at a low dose and titrated cautiously to the lowest effective dose.
Behavioral and psychological symptoms of dementia are frequently temporary. Therefore, a gradual reduction and eventual withdrawal of antipsychotic medications should be attempted every 3 months. Studies indicate that most patients are able to tolerate elimination of antipsychotic medications with no worsening of behavioral symptoms.
Despite the limitations of treatment, SGAs remain a valid consideration when other interventions have proven insufficient. However, judicious use of these agents remains the cornerstone of therapy.
Bottom Line
Until better treatment options become available, second-generation antipsychotics (SGAs) continue to have an important, albeit limited, role in the treatment of behavioral disturbances in dementia. Despite the limitations of treatment, SGAs remain a valid consideration when other interventions have proven insufficient. However, judicious use of these agents remains the cornerstone of therapy.
Related Resources
- Kales HC, Mulsant BH, Sajatovic MS. Prescribing antipsychotics in geriatric patients: Focus on dementia. Third of 3 parts. Current Psychiatry. 2017;16(12):24-30.
- Meeks TW, Jeste DV. Antipsychotics in dementia: Beyond ‘black-box’ warnings. Current Psychiatry. 2008;7(6):51-52, 55-58, 64-65.
Drug Brand Names
Aripiprazole • Abilify
Haloperidol • Haldol
Olanzapine • Zyprexa
Pimavanserin • Nuplazid
Risperidone • Risperdal
Quetiapine • Seroquel
Ziprasidone • Geodon
As psychiatrists treating an aging population, we frequently face the daunting challenges of managing medically complex and behaviorally unstable patients whose fragile condition tests the brightest among us. As our population enters late life, not only are physicians confronted with aging patients whose bodies have decreased renal and hepatic function, but we also face the challenges of the aging brain, severed neuronal networks, and neurotransmitter diminution. These physiological changes can alter treatment response, increase the frequency of adverse effects, and increase the likelihood of emergence of behavioral and psychological symptoms.
During the past decade, the number of people reaching age 65 has dramatically increased. As life expectancy improves, the “oldest old”—those age 85 and older—are the fastest-growing segment of the population. The prevalence of cognitive impairment, including mild cognitive impairment and dementia, in this cohort is >40%.1 Roughly 90% of patients with dementia will develop clinically significant behavioral problems at some point in the course of their illness.2
Behavioral and psychological symptoms of dementia (BPSD) have a tremendous impact on the quality of life for both patients and their caregivers. We are experts in understanding these behaviors and crafting nonpharmacologic treatment plans to manage them. Understanding the context in which behaviors emerge allows us to modify the environment, communication strategies, and other potential triggers, in turn reducing the need for pharmacologic intervention.
However, when nonpharmacologic interventions have been exhausted, what are the options? Antipsychotics have been one of the approaches used to address the challenges of behavioral disturbances and psychosis occurring in dementia. Unfortunately, there is conflicting evidence regarding the risks and benefits associated with the use of antipsychotics in this population. In this article, we provide a roadmap for the judicious use of antipsychotics for patients with dementia.
Weighing the risks and benefits of antipsychotics
Until better treatment options become available, second-generation antipsychotics (SGAs) continue to have an important but limited role in the treatment of behavioral disturbances in dementia. Although safety risks exist, they can be minimized through the careful selection of appropriate patients for treatment, close monitoring, and effective communication with patients and caregivers before and during treatment.
Several studies examining the efficacy of antipsychotics in the treatment of BPSD have demonstrated an increased risk of cerebrovascular events, including stroke and death due to any cause.3 This evidence prompted the FDA to issue a “black-box” warning in 2005 to highlight the increased risk of mortality for patients with dementia who are treated with SGAs.4 Both first-generation antipsychotics (FGAs) and SGAs have been associated with higher rates of mortality than most other psychotropic classes, except anticonvulsants. This increased mortality risk has been shown to persist for at least 6 to 12 months.5,6 FGAs appear to be associated with a greater mortality risk compared with SGAs. As a result, if antipsychotic treatment is necessary, the use of FGAs in this population is not recommended.
The potential mechanisms leading to stroke and death remain unclear. They could include orthostatic hypotension, anticholinergic adverse effects, QT prolongation, platelet aggregation effects, and venous thromboembolism. The presence of cardiovascular and vascular risk factors, electrolyte imbalances, cardiac arrhythmias, and concomitant use of medications that prolong the QTc interval may confer additional risks.
Continued to: Although the use of antipsychotics for patients with dementia...
Although the use of antipsychotics for patients with dementia may increase the risk of mortality, the absolute increased risk to a given individual, at least with short-term treatment, is likely small. The risk may also vary depending on the choice of SGA. Patients who were treated with quetiapine had a slightly lower risk of death than those who were treated risperidone.5 Death rates among patients prescribed aripiprazole, olanzapine, and ziprasidone were similar to the death rates of patients who were treated with risperidone. Compared with patients who were treated with risperidone, patients who were treated with the FGA haloperidol were twice as likely to die during a subsequent 6-month observation period. The largest number of deaths occurred during the first 40 days of treatment.5
While this increased risk of mortality is an important factor to discuss with patients and caregivers when deciding whether to initiate antipsychotic treatment, it is also important to put it into perspective. For example, the risk of suddenly dying from a stroke or heart attack for a person with dementia who is not taking an antipsychotic is approximately 2%. When an individual is started on one of these agents, that risk increases to approximately 4%. While the mortality risk is doubled, it remains relatively small.4 When faced with verbal or physical assaults, hostility, paranoid ideations, or other psychotic symptoms, many families feel that this relatively low risk does not outweigh the potential benefits of reducing caregiver and patient distress. If nonpharmacologic and/or other pharmacologic interventions have failed, the treatment has reached a point of no good alternatives and therapy should then focus on minimizing risk.
Informed consent is essential. A discussion of risks and benefits with the patient, family, or other decision-makers should focus on the risk of stroke, potential metabolic effects, and mortality, as well as potential worsening of cognitive decline associated with antipsychotic treatment. This should be weighed together with the evidence that suggests psychosis and agitation are associated with earlier nursing home admission and death.7,8 Families should be given ample time and opportunity to ask questions. Alternatives to immediate initiation of antipsychotics should be thoroughly reviewed.
Despite the above-noted risks, expert consensus suggests that the use of antipsychotics in the treatment of individuals with dementia can be appropriate, particularly in individuals with dangerous agitation or psychosis.9 These agents can minimize the risk of violence, reduce patient distress, improve the patient’s quality of life, and reduce caregiver burden. In clinical trials, the benefits of antipsychotics have been modest. Nevertheless, evidence has shown that these agents can reduce psychosis, agitation, aggression, hostility, and suspiciousness, which makes them a valid option when other interventions have proven insufficient.
Target specific symptoms
Despite this article’s focus on the appropriate use of antipsychotics for patients with BPSD, it is important to emphasize that the first-line approach to the management of BPSD in this population should always be a person-centered, psychosocial, multidisciplinary, nonpharmacologic approach that focuses on identifying triggers and treating potentially modifiable contributors to behavioral symptoms. Table 110 outlines common underlying causes of BPSD in dementia that should be assessed before prescribing an antipsychotic.

Continued to: Alternative psychopharmacologic treatments...
Alternative psychopharmacologic treatments based on a psychobehavioral metaphor should also be considered (Table 211). This approach matches the dominant target symptoms to the most relevant medication class.11 For example, in the case of a verbally and physically agitated patient who is also irritable, negative, socially withdrawn, and appears dysphoric, we might first undertake a trial of an antidepressant. Conversely, if the patient shows agitation in the context of increased motor activity, loud and rapid speech, and affective lability, we might consider the use of a mood stabilizer. Pharmacologic treatment should be aimed at the modification of clearly identified and documented target behaviors.

Indications to use antipsychotics for patients with dementia include:
- severe agitation and aggression associated with risk of harm
- delusions and hallucinations
- comorbid preexisting mental health conditions (eg, bipolar disorder, schizophrenia, treatment-resistant depression, etc.).
Symptoms that do not usually respond to an antipsychotic include wandering, social withdrawal, shouting, pacing, touching, cognitive defects, and incontinence.12 These symptoms may respond to interventions such as changes to the environment.
Continued to: Choosing an antipsychotic
Choosing an antipsychotic
Once you have identified that an antipsychotic is truly indicated, the choice of an agent will focus on patient-related factors. Considerations such as frailty, comorbid medical conditions including diabetes, history of falls, hepatic insufficiency, cardiac arrhythmias, and cerebrovascular risk factors, should all be analyzed prior to initiating an antipsychotic. The presence of these conditions will increase the likelihood that adverse effects may occur. It will also guide the dose trajectory and the target dose for discontinuation. Antipsychotics differ with respect to their efficacy and adverse effect profile. For practical purposes, adverse effects typically guide the selection of these agents when used for patients with dementia.
Continued to: Gradual structural changes occur...
Gradual structural changes occur in the dopaminergic system with age and increase the propensity for antipsychotic adverse effects. The number of dopaminergic neurons and D2 receptors decreases approximately 10% per decade. In order to avoid the development of adverse effects related to extrapyramidal symptoms, approximately 20% of receptors need to be free. FGAs tend to block approximately 90% of D2 receptors, whereas SGAs block less than 70% to 80% and dissociate more rapidly from D2 receptors.13 FGAs should therefore be avoided, as they have been associated with numerous adverse effects, including parkinsonism, tardive dyskinesia, akathisia, sedation, peripheral and central anticholinergic effects, postural hypotension, cardiac conduction defects, and falls. As noted above, they have been linked to a greater risk of mortality (Figure14 ).

When the decision to use an antipsychotic agent is made for a person with dementia, SGAs appear to be a better choice. There appear to be modest differences within the class of SGAs in terms of effectiveness, tolerability, and adverse effect profile. Although the association between the dose of an antipsychotic and the risk of mortality or stroke remains undefined, other common adverse effects, such as sedation, extrapyramidal symptoms, and risk of falls, can be reduced by starting at the lowest dose possible and titrating slowly.
Dosing considerations
Dose increments should be modest and, in a nonemergent setting, may be adjusted at weekly intervals depending on response. Prior to starting a treatment trial, it is advisable to estimate what will constitute a worthwhile clinical response, the duration of treatment, and the maximum dose. Avoid high doses or prolonged use of antipsychotics that have not significantly improved the target behavior.
When the decision to use a SGA is made, choosing the initial starting dose is challenging given that none of these medications has an indication for use in this population. We propose doses that have been used in completed randomized trials that reflect the best information available about the dose likely to maximize benefit and minimize risk. On the basis of those trials, reasonable starting doses would be15-22:
- quetiapine 25 to 50 mg/d
- risperidone 0.5 to 1 mg/d
- aripiprazole 2 to 10 mg/d
- olanzapine 2.5 to 5 mg/d
- ziprasidone 20 mg/d
Continued to: The highest doses tested...
The highest doses tested for each of these compounds in randomized clinical trials for this population were: risperidone 2 mg/d, olanzapine 10 mg/d, and aripiprazole 15 mg/d. A wide variety of maximum doses of quetiapine were studied in clinical trials, with a top dose of 200 mg being most common. It is worth noting that doses higher than these have been used for other indications.15-22
Quetiapine. One of the most commonly prescribed antipsychotics for the treatment of BPSD in individuals with memory disorders is quetiapine. The reasons for this preference include a low risk of extrapyramidal adverse effects, flexibility of dosing, ability to use lower dosages, and evidence of the lower risk of mortality when compared with other second-generation agents.5,15 If an antipsychotic is indicated, quetiapine should be considered as a first-line antipsychotic therapy. Quetiapine has well-established effects on mood, anxiety, and sleep, all of which can be disrupted in dementia and can act as drivers for agitation.5,15 Starting quetiapine may mitigate the need for separate agents to treat insomnia, loss of appetite, or anxiety, although it is not FDA-indicated for these comorbid conditions. Quetiapine is also less likely to exacerbate motor symptoms compared with other SGAs but has the potential to increase the risk of falls, and orthostasis, and carries a considerable anticholinergic burden.5,15
Risperidone has been shown to provide modest improvements in some people exhibiting symptoms of aggression, agitation, and psychosis.5,15 There is no evidence that risperidone is any more effective than other SGAs, but it has been tested on more geriatric patients than other SGAs. The fact that it is also available in an orally disintegrating tablet makes it a practical treatment in certain populations of patients, such as those who have difficulty swallowing. Risperidone carries the highest extrapyramidal symptom burden among the SGAs due to its potent D2 receptor binding. 5,15
Aripiprazole. There have been several studies of aripiprazole for the treatment of psychosis and agitation in Alzheimer’s dementia.15 This medication showed modest effect and was generally well tolerated. Aripiprazole appears to have less associated weight gain, which may be pertinent for some patients. It also appears to be less sedating than many of the other SGAs. However, some patients may experience activation or insomnia with this agent, particularly with doses <15 mg/d. This activating effect may be beneficial for treating comorbid depressive symptoms, although lower doses could theoretically worsen psychosis due to the activating effects.
Aripiprazole has also been studied in Parkinson’s disease. While some patients had favorable responses with improvement in psychosis and behavioral disturbances, this medication was also associated with worsening of motor symptoms. Certain individuals also experienced a worsening of their psychosis.23 For this reason, it is unlikely to be a useful agent for patients displaying evidence of parkinsonism, Parkinson’s dementia, or dementia with Lewy bodies.
Olanzapine. Several studies have shown that low-dose olanzapine has been modestly effective in decreasing agitation and aggression in patients suffering from Alzheimer’s and vascular dementias.24 The medication is also available in an orally disintegrating form, which may be beneficial when treating individuals whose swallowing abilities are compromised. Olanzapine also has been associated with significant weight gain and metabolic syndrome.24
Continued to: Ziprasidone
Ziprasidone. There are no specific studies of ziprasidone for geriatric patients and none for patients with dementia. However, case reports have suggested both oral and injectable forms of the medication may be well tolerated and have some benefit in treating agitation in this population.25 Based on evidence from younger populations, ziprasidone is less likely to be associated with weight gain or orthostatic hypotension. Medication has been associated with QTc prolongation and should be used with caution and monitored with an ECG.
The initial dosing and potential adverse effects of quetiapine, risperidone, aripiprazole, olanzapine, and ziprasidone are highlighted in Table 3.10

Other SGAs. Newer antipsychotics have recently become available and may serve as additional tools for managing BPSD in the future. Unfortunately, there are currently no available studies regarding their efficacy in the treatment of agitation and psychosis in dementia. One notable exception is pimavaserin, a serotonin 2A receptor inverse agonist. This medication has recently been FDA-approved for the treatment of Parkinson’s disease psychosis. The medication was extensively studied in older patients. It appeared to be effective in reducing delusions and hallucinations while not impairing motor function or causing sedation or hypotension.23 Additional studies are currently ongoing for the treatment of Alzheimer’s dementia psychosis.
Monitor treatment, consider discontinuation
American Psychiatric Association guidelines on the use of antipsychotics to treat agitation or psychosis in patients with dementia currently recommend that clinicians use a quantitative measure to track symptoms and response to treatment.26 These measures may be formal, such as an overall assessment of symptom severity on a Likert scale, or as simple as monitoring the changes in the frequency of periods of agitation.
After starting an antipsychotic, a follow-up appointment should typically take place within 1 month. If the patient is at high risk for developing adverse effects, or if the symptoms are severe, a follow-up appointment for monitoring the response to treatment and potential adverse effects should occur within 1 week. At a minimum, expert consensus suggests follow-up visits should occur every 3 months.
If there is no clinical response after 4 weeks of adequate dosing of an antipsychotic, the medication should be tapered and withdrawn. Switching to an alternative agent may be appropriate.
Many patients will have only partial remission of target symptoms. Therefore, increasing the dose or switching to an alternative agent may be necessary. Concurrent use of multiple antipsychotic agents should be avoided.
Continued to: Maintenance treatment may be appropriate
Maintenance treatment may be appropriate for patients who have demonstrated a clear benefit from antipsychotic treatment without undue adverse effects, and in whom a trial dose reduction has resulted in reappearance of the target symptoms. A formal monitoring plan to assess changes in response and the significance of adverse effects should be in place. Review the target behavior, changes in function, and significance of adverse effects at least every 3 months.
How to approach discontinuation
Behavioral and psychological symptoms of dementia are frequently temporary. If the patient has been stable, gradual dose reduction and eventual discontinuation of antipsychotics should be attempted every 3 months. Studies have reported that most patients who were taken off antipsychotics for treating BPSD showed no worsening of behavioral symptoms.27
Discontinuation of antipsychotics should be done gradually by reducing the dose by 50% every 2 weeks, and then stopping after 2 weeks on the minimum dose, with monitoring for recurrence of target symptoms or emergence of new ones. The longer a medication has been prescribed, the slower the withdrawal occurs. Thus, the possibility of emerging symptoms related to drug withdrawal will lessen.
A roadmap for judicious prescribing

When underlying treatable or reversible causes of BPSD in dementia have been ruled out or nonpharmacologic treatments have failed, a trial of an antipsychotic may be indicated. The choice of agent should focus on patient-related factors and on clearly identified target behaviors. Treatment should be started at a low dose and titrated cautiously to the lowest effective dose.
Behavioral and psychological symptoms of dementia are frequently temporary. Therefore, a gradual reduction and eventual withdrawal of antipsychotic medications should be attempted every 3 months. Studies indicate that most patients are able to tolerate elimination of antipsychotic medications with no worsening of behavioral symptoms.
Despite the limitations of treatment, SGAs remain a valid consideration when other interventions have proven insufficient. However, judicious use of these agents remains the cornerstone of therapy.
Bottom Line
Until better treatment options become available, second-generation antipsychotics (SGAs) continue to have an important, albeit limited, role in the treatment of behavioral disturbances in dementia. Despite the limitations of treatment, SGAs remain a valid consideration when other interventions have proven insufficient. However, judicious use of these agents remains the cornerstone of therapy.
Related Resources
- Kales HC, Mulsant BH, Sajatovic MS. Prescribing antipsychotics in geriatric patients: Focus on dementia. Third of 3 parts. Current Psychiatry. 2017;16(12):24-30.
- Meeks TW, Jeste DV. Antipsychotics in dementia: Beyond ‘black-box’ warnings. Current Psychiatry. 2008;7(6):51-52, 55-58, 64-65.
Drug Brand Names
Aripiprazole • Abilify
Haloperidol • Haldol
Olanzapine • Zyprexa
Pimavanserin • Nuplazid
Risperidone • Risperdal
Quetiapine • Seroquel
Ziprasidone • Geodon
1. Gardner RC, Valcour V, Yaffe K. Dementia in the oldest old: a multi-factorial and growing public health issue. Alzheimers Res Ther. 2013;5(4):27.
2. Tariot PN, Blazina L. The psychopathology of dementia. In: Morris JC, ed. Handbook of dementing illnesses. New York, NY: Marcel Dekker Inc.; 1993:461-475.
3. Schneider LS, Dagerman KS, Insel P. Risk of death with atypical antipsychotic drug treatment for dementia: meta-analysis of randomized placebo-controlled trials. JAMA. 2005;294:1934-1943.
4. Lenzer J. FDA warns about using antipsychotic drugs for dementia. BMJ. 2005;330(7497):922.
5. Kales HC, Valenstein M, Kim HM, et al. Mortality risk in patients with dementia treated with antipsychotics versus other psychiatric medications. Am J Psychiatry. 2007;164(10):1568-1576; quiz 1623.
6. Gill SS, Bronskill SE, Normand SL, et al. Antipsychotic drug use and mortality in older adults with dementia. Ann Intern Med. 2007;146(11):775-786.
7. Okura T, Plassman BL, Steffens DC, et al. Neuropsychiatric symptoms and the risk of institutionalization and death: the aging, demographics, and memory study. J Am Geriatr Soc. 2011;59:473-481.
8. Banerjee S, Murray J, Foley B, et al. Predictors of institutionalisation in people with dementia. J Neurol Neurosurg Psychiatry. 2003;74:1315-1316.
9. Alexopoulos GS, Jeste DV, Chung H, et al. The expert consensus guideline series. Treatment of dementia and its behavioral disturbances. Introduction: methods, commentary, and summary. Postgrad Med. 2005;Spec No:6-22.
10. Burke AD, Hall G, Yaari R, et al. Pocket reference to Alzheimer’s disease management. Philadelphia, PA: Springer Healthcare Communications; 2015:39-46
11. Burke AD, Burke WJ, Tariot PN. Drug treatments for the behavioural and psychiatric symptoms of dementia. In: Ames D, O’Brien JT, Burns A, eds. Dementia, 5th ed. Boca Raton, FL: CRC Press; 2016:231-252.
12. Royal Australian and New Zealand College of Psychiatrists. Antipsychotics in dementia: best practice guide. https://bpac.org.nz/a4d/resources/docs/bpac_A4D_best_practice_guide.pdf. Accessed September 4, 2018.
13. Nyberg L, Backman L. Cognitive aging: a view from brain imaging. In: Dixon RA, Backman L, Nilsson LG, eds. New frontiers in cognitive aging. Oxford: Oxford Univ Press; 2004:135-60.
14. Huybrechts KF, Gerhard T, Crystal S, et al. Differential risk of death in older residents in nursing homes prescribed specific antipsychotic drugs: population based cohort study. BMJ. 2012;344:e977. doi: 10.1136/bmj.e977.
15. Burke AD, Tariot PN. Atypical antipsychotics in the elderly: a review of therapeutic trends and clinical outcomes. Expert Opin Pharmacother. 2009;10(15):2407-2414.
16. De Deyn PP, Rabheru K, Rasmussen A, et al. A randomized trial of risperidone, placebo, and haloperidol for behavioral symptoms of dementia. Neurology.1999;53(5):946-955.
17. De Deyn PP, Jeste DV, Auby P, et al. Aripiprazole in dementia of the Alzheimer’s type. Poster presented at: 16th Annual Meeting of American Association for Geriatric Psychiatry; March 1-4, 2003; Honolulu, HI.
18. Lopez OL, Becker JT, Chang YF, et al. The long-term effects of conventional and atypical antipsychotics in patients with probable Alzheimer’s disease. Am J Psychiatry. 2013;170(9):1051-1058.
19. Mintzer J, Weiner M, Greenspan A, et al. Efficacy and safety of a flexible dose of risperidone versus placebo in the treatment of psychosis of Alzheimer’s disease. In: International College of Geriatric Psychopharmacology. Basel, Switzerland; 2004.
20. Mintzer JE, Tune LE, Breder CD, et al. Aripiprazole for the treatment of psychoses in institutionalized patients with Alzheimer dementia: a multicenter, randomized, double-blind, placebo-controlled assessment of three fixed doses. Am J Geriatr Psychiatry. 2007;15(11):918-931.
21. Sultzer DL, Davis SM, Tariot PN, et al; CATIE-AD Study Group. Clinical symptom responses to atypical antipsychotic medications in Alzheimer’s disease: phase 1 outcomes from the CATIE-AD effectiveness trial. Am J Psychiatry. 2008;165(7):844-854.
22. Zhong KX, Tariot PN, Mintzer J, et al. Quetiapine to treat agitation in dementia: a randomized, double-blind, placebo-controlled study. Curr Alzheimer Res. 2007;4(1):81-93.
23. Bozymski KM, Lowe DK, Pasternak KM, et al. Pimavanserin: a novel antipsychotic for Parkinson’s disease psychosis. Ann Pharmacother. 2017;51(6):479-487.
24. Moretti R, Torre R, Antonello T, et al. Olanzapine as a possible treatment of behavioral symptoms in vascular dementia: risks of cerebrovascular events. J Neurol. 2005;252:1186.
25. Cole SA, Saleem R, Shea WP, et al. Ziprasidone for agitation or psychosis in dementia: four cases. Int J Psychiatry Med. 2005;35(1):91-98.
26. Reus VI, Fochtmann LJ, Eyler AE, et al. The American Psychiatric Association practice guideline on the use of antipsychotics to treat agitation or psychosis in patients with dementia. Am J Psychiatry. 2016;173(5):543-546.
27. Horwitz GJ, Tariot PN, Mead K, et al. Discontinuation of antipsychotics in nursing home patients with dementia. Am J Geriatr Psychiatry. 1995;3(4):290-299.
1. Gardner RC, Valcour V, Yaffe K. Dementia in the oldest old: a multi-factorial and growing public health issue. Alzheimers Res Ther. 2013;5(4):27.
2. Tariot PN, Blazina L. The psychopathology of dementia. In: Morris JC, ed. Handbook of dementing illnesses. New York, NY: Marcel Dekker Inc.; 1993:461-475.
3. Schneider LS, Dagerman KS, Insel P. Risk of death with atypical antipsychotic drug treatment for dementia: meta-analysis of randomized placebo-controlled trials. JAMA. 2005;294:1934-1943.
4. Lenzer J. FDA warns about using antipsychotic drugs for dementia. BMJ. 2005;330(7497):922.
5. Kales HC, Valenstein M, Kim HM, et al. Mortality risk in patients with dementia treated with antipsychotics versus other psychiatric medications. Am J Psychiatry. 2007;164(10):1568-1576; quiz 1623.
6. Gill SS, Bronskill SE, Normand SL, et al. Antipsychotic drug use and mortality in older adults with dementia. Ann Intern Med. 2007;146(11):775-786.
7. Okura T, Plassman BL, Steffens DC, et al. Neuropsychiatric symptoms and the risk of institutionalization and death: the aging, demographics, and memory study. J Am Geriatr Soc. 2011;59:473-481.
8. Banerjee S, Murray J, Foley B, et al. Predictors of institutionalisation in people with dementia. J Neurol Neurosurg Psychiatry. 2003;74:1315-1316.
9. Alexopoulos GS, Jeste DV, Chung H, et al. The expert consensus guideline series. Treatment of dementia and its behavioral disturbances. Introduction: methods, commentary, and summary. Postgrad Med. 2005;Spec No:6-22.
10. Burke AD, Hall G, Yaari R, et al. Pocket reference to Alzheimer’s disease management. Philadelphia, PA: Springer Healthcare Communications; 2015:39-46
11. Burke AD, Burke WJ, Tariot PN. Drug treatments for the behavioural and psychiatric symptoms of dementia. In: Ames D, O’Brien JT, Burns A, eds. Dementia, 5th ed. Boca Raton, FL: CRC Press; 2016:231-252.
12. Royal Australian and New Zealand College of Psychiatrists. Antipsychotics in dementia: best practice guide. https://bpac.org.nz/a4d/resources/docs/bpac_A4D_best_practice_guide.pdf. Accessed September 4, 2018.
13. Nyberg L, Backman L. Cognitive aging: a view from brain imaging. In: Dixon RA, Backman L, Nilsson LG, eds. New frontiers in cognitive aging. Oxford: Oxford Univ Press; 2004:135-60.
14. Huybrechts KF, Gerhard T, Crystal S, et al. Differential risk of death in older residents in nursing homes prescribed specific antipsychotic drugs: population based cohort study. BMJ. 2012;344:e977. doi: 10.1136/bmj.e977.
15. Burke AD, Tariot PN. Atypical antipsychotics in the elderly: a review of therapeutic trends and clinical outcomes. Expert Opin Pharmacother. 2009;10(15):2407-2414.
16. De Deyn PP, Rabheru K, Rasmussen A, et al. A randomized trial of risperidone, placebo, and haloperidol for behavioral symptoms of dementia. Neurology.1999;53(5):946-955.
17. De Deyn PP, Jeste DV, Auby P, et al. Aripiprazole in dementia of the Alzheimer’s type. Poster presented at: 16th Annual Meeting of American Association for Geriatric Psychiatry; March 1-4, 2003; Honolulu, HI.
18. Lopez OL, Becker JT, Chang YF, et al. The long-term effects of conventional and atypical antipsychotics in patients with probable Alzheimer’s disease. Am J Psychiatry. 2013;170(9):1051-1058.
19. Mintzer J, Weiner M, Greenspan A, et al. Efficacy and safety of a flexible dose of risperidone versus placebo in the treatment of psychosis of Alzheimer’s disease. In: International College of Geriatric Psychopharmacology. Basel, Switzerland; 2004.
20. Mintzer JE, Tune LE, Breder CD, et al. Aripiprazole for the treatment of psychoses in institutionalized patients with Alzheimer dementia: a multicenter, randomized, double-blind, placebo-controlled assessment of three fixed doses. Am J Geriatr Psychiatry. 2007;15(11):918-931.
21. Sultzer DL, Davis SM, Tariot PN, et al; CATIE-AD Study Group. Clinical symptom responses to atypical antipsychotic medications in Alzheimer’s disease: phase 1 outcomes from the CATIE-AD effectiveness trial. Am J Psychiatry. 2008;165(7):844-854.
22. Zhong KX, Tariot PN, Mintzer J, et al. Quetiapine to treat agitation in dementia: a randomized, double-blind, placebo-controlled study. Curr Alzheimer Res. 2007;4(1):81-93.
23. Bozymski KM, Lowe DK, Pasternak KM, et al. Pimavanserin: a novel antipsychotic for Parkinson’s disease psychosis. Ann Pharmacother. 2017;51(6):479-487.
24. Moretti R, Torre R, Antonello T, et al. Olanzapine as a possible treatment of behavioral symptoms in vascular dementia: risks of cerebrovascular events. J Neurol. 2005;252:1186.
25. Cole SA, Saleem R, Shea WP, et al. Ziprasidone for agitation or psychosis in dementia: four cases. Int J Psychiatry Med. 2005;35(1):91-98.
26. Reus VI, Fochtmann LJ, Eyler AE, et al. The American Psychiatric Association practice guideline on the use of antipsychotics to treat agitation or psychosis in patients with dementia. Am J Psychiatry. 2016;173(5):543-546.
27. Horwitz GJ, Tariot PN, Mead K, et al. Discontinuation of antipsychotics in nursing home patients with dementia. Am J Geriatr Psychiatry. 1995;3(4):290-299.
5 Strategies for managing antipsychotic-induced hyperprolactinemia
There is a well-established relationship between antipsychotic treatment and hyperprolactinemia. Most antipsychotics have been linked to increased prolactin levels, and the risk appears to be dose-related.1 Antipsychotic-induced hyperprolactinemia can be asymptomatic, but it also has been associated with several adverse effects, including menstrual irregularity, osteoporosis, gynecomastia, and sexual dysfunction. Here I discuss what to do before starting a patient on an antipsychotic, and 5 treatment strategies for addressing antipsychotic-induced hyperprolactinemia.
Get a baseline prolactin level
Before starting a patient on an antipsychotic, obtain a baseline prolactin level measurement. If the patient later develops hyperprolactinemia, having a baseline measurement will make it easier to determine if the antipsychotic is a potential cause. Also, it is helpful to gather additional information regarding baseline psychosexual function and menstruation before starting an antipsychotic.

It is critical to determine if a temporal relationship exists between exposure to an antipsychotic and increase in prolactin levels.3 If the time course is unclear, laboratory tests need to be performed, including assessing liver, renal, and thyroid function or imaging of the pituitary gland. Also, hyperprolactinemia should not be diagnosed based on a single blood test result, because emotional and physical stress can elevate prolactin levels.
Continued to: 5 strategies for addressing hyperprolactinemia
5 strategies for addressing hyperprolactinemia
1. Reduce the antipsychotic dose. Because the risk of hyperprolactinemia is dose-dependent, reducing the antipsychotic dose could be helpful for some patients.
2. Switch to a prolactin-sparing antipsychotic, such as clozapine, quetiapine, olanzapine, or ziprasidone. However, it is often difficult to predict positive outcomes because switching antipsychotics may cause new adverse effects or trigger a psychotic relapse.
3. Consider sex hormone replacement therapy. A combined oral contraceptive could prevent osteoporosis and help estrogen deficiency symptoms in women who require antipsychotic medication. However, this treatment approach may worsen galactorrhea.
4. Use a dopamine receptor agonist. Dopamine receptor agonists, such as cabergoline or bromocriptine, have been shown to suppress prolactin secretion. Clinicians should always proceed cautiously because these medications can potentially increase the risk of psychosis.
5. Examine the potential benefits of adding aripiprazole because it can be used for augmentation to reduce prolactin levels in patients receiving other antipsychotics. In some cases, dopamine receptors can be exposed to competition between a partial agonist (aripiprazole) and an antagonist (the current antipsychotic). This competition may decrease the effectiveness of the current antipsychotic.1 Also, adding another antipsychotic could increase overall adverse effects.
1. Montejo ÁL, Arango C, Bernardo M, et al. Multidisciplinary consensus on the therapeutic recommendations for iatrogenic hyperprolactinemia secondary to antipsychotics. Front Neuroendocrinol. 2017;45:25-34.
2. Taylor D, Paton C, Kapur S. Schizophrenia. In: Taylor D, Paton C, Kapur S. The Maudsley Prescribing Guidelines in psychiatry. 12th ed. Chichester, UK: Wiley Blackwell; 2015:133-134.
3. Miyamoto BE, Galecki M, Francois D. Guidelines for antipsychotic-induced hyperprolactinemia. Psychiatr Ann. 2015;45(5):266,268,270-272.
There is a well-established relationship between antipsychotic treatment and hyperprolactinemia. Most antipsychotics have been linked to increased prolactin levels, and the risk appears to be dose-related.1 Antipsychotic-induced hyperprolactinemia can be asymptomatic, but it also has been associated with several adverse effects, including menstrual irregularity, osteoporosis, gynecomastia, and sexual dysfunction. Here I discuss what to do before starting a patient on an antipsychotic, and 5 treatment strategies for addressing antipsychotic-induced hyperprolactinemia.
Get a baseline prolactin level
Before starting a patient on an antipsychotic, obtain a baseline prolactin level measurement. If the patient later develops hyperprolactinemia, having a baseline measurement will make it easier to determine if the antipsychotic is a potential cause. Also, it is helpful to gather additional information regarding baseline psychosexual function and menstruation before starting an antipsychotic.

It is critical to determine if a temporal relationship exists between exposure to an antipsychotic and increase in prolactin levels.3 If the time course is unclear, laboratory tests need to be performed, including assessing liver, renal, and thyroid function or imaging of the pituitary gland. Also, hyperprolactinemia should not be diagnosed based on a single blood test result, because emotional and physical stress can elevate prolactin levels.
Continued to: 5 strategies for addressing hyperprolactinemia
5 strategies for addressing hyperprolactinemia
1. Reduce the antipsychotic dose. Because the risk of hyperprolactinemia is dose-dependent, reducing the antipsychotic dose could be helpful for some patients.
2. Switch to a prolactin-sparing antipsychotic, such as clozapine, quetiapine, olanzapine, or ziprasidone. However, it is often difficult to predict positive outcomes because switching antipsychotics may cause new adverse effects or trigger a psychotic relapse.
3. Consider sex hormone replacement therapy. A combined oral contraceptive could prevent osteoporosis and help estrogen deficiency symptoms in women who require antipsychotic medication. However, this treatment approach may worsen galactorrhea.
4. Use a dopamine receptor agonist. Dopamine receptor agonists, such as cabergoline or bromocriptine, have been shown to suppress prolactin secretion. Clinicians should always proceed cautiously because these medications can potentially increase the risk of psychosis.
5. Examine the potential benefits of adding aripiprazole because it can be used for augmentation to reduce prolactin levels in patients receiving other antipsychotics. In some cases, dopamine receptors can be exposed to competition between a partial agonist (aripiprazole) and an antagonist (the current antipsychotic). This competition may decrease the effectiveness of the current antipsychotic.1 Also, adding another antipsychotic could increase overall adverse effects.
There is a well-established relationship between antipsychotic treatment and hyperprolactinemia. Most antipsychotics have been linked to increased prolactin levels, and the risk appears to be dose-related.1 Antipsychotic-induced hyperprolactinemia can be asymptomatic, but it also has been associated with several adverse effects, including menstrual irregularity, osteoporosis, gynecomastia, and sexual dysfunction. Here I discuss what to do before starting a patient on an antipsychotic, and 5 treatment strategies for addressing antipsychotic-induced hyperprolactinemia.
Get a baseline prolactin level
Before starting a patient on an antipsychotic, obtain a baseline prolactin level measurement. If the patient later develops hyperprolactinemia, having a baseline measurement will make it easier to determine if the antipsychotic is a potential cause. Also, it is helpful to gather additional information regarding baseline psychosexual function and menstruation before starting an antipsychotic.

It is critical to determine if a temporal relationship exists between exposure to an antipsychotic and increase in prolactin levels.3 If the time course is unclear, laboratory tests need to be performed, including assessing liver, renal, and thyroid function or imaging of the pituitary gland. Also, hyperprolactinemia should not be diagnosed based on a single blood test result, because emotional and physical stress can elevate prolactin levels.
Continued to: 5 strategies for addressing hyperprolactinemia
5 strategies for addressing hyperprolactinemia
1. Reduce the antipsychotic dose. Because the risk of hyperprolactinemia is dose-dependent, reducing the antipsychotic dose could be helpful for some patients.
2. Switch to a prolactin-sparing antipsychotic, such as clozapine, quetiapine, olanzapine, or ziprasidone. However, it is often difficult to predict positive outcomes because switching antipsychotics may cause new adverse effects or trigger a psychotic relapse.
3. Consider sex hormone replacement therapy. A combined oral contraceptive could prevent osteoporosis and help estrogen deficiency symptoms in women who require antipsychotic medication. However, this treatment approach may worsen galactorrhea.
4. Use a dopamine receptor agonist. Dopamine receptor agonists, such as cabergoline or bromocriptine, have been shown to suppress prolactin secretion. Clinicians should always proceed cautiously because these medications can potentially increase the risk of psychosis.
5. Examine the potential benefits of adding aripiprazole because it can be used for augmentation to reduce prolactin levels in patients receiving other antipsychotics. In some cases, dopamine receptors can be exposed to competition between a partial agonist (aripiprazole) and an antagonist (the current antipsychotic). This competition may decrease the effectiveness of the current antipsychotic.1 Also, adding another antipsychotic could increase overall adverse effects.
1. Montejo ÁL, Arango C, Bernardo M, et al. Multidisciplinary consensus on the therapeutic recommendations for iatrogenic hyperprolactinemia secondary to antipsychotics. Front Neuroendocrinol. 2017;45:25-34.
2. Taylor D, Paton C, Kapur S. Schizophrenia. In: Taylor D, Paton C, Kapur S. The Maudsley Prescribing Guidelines in psychiatry. 12th ed. Chichester, UK: Wiley Blackwell; 2015:133-134.
3. Miyamoto BE, Galecki M, Francois D. Guidelines for antipsychotic-induced hyperprolactinemia. Psychiatr Ann. 2015;45(5):266,268,270-272.
1. Montejo ÁL, Arango C, Bernardo M, et al. Multidisciplinary consensus on the therapeutic recommendations for iatrogenic hyperprolactinemia secondary to antipsychotics. Front Neuroendocrinol. 2017;45:25-34.
2. Taylor D, Paton C, Kapur S. Schizophrenia. In: Taylor D, Paton C, Kapur S. The Maudsley Prescribing Guidelines in psychiatry. 12th ed. Chichester, UK: Wiley Blackwell; 2015:133-134.
3. Miyamoto BE, Galecki M, Francois D. Guidelines for antipsychotic-induced hyperprolactinemia. Psychiatr Ann. 2015;45(5):266,268,270-272.
Preventing brain damage in psychosis
I read with great interest Dr. Nasrallah’s editorial, “FAST and RAPID: Acronyms to prevent brain damage in stroke and psychosis” (From the Editor,
Mitchell L. Glaser, MD
Board-Certified Child/Adolescent and General Psychiatrist
Assistant Professor of Psychiatry
Rush University Medical Center
Chairman
Department of Psychiatry
Medical Director of Child/Adolescent Psychiatry
St. Mary/Elizabeth Medical Center
Clinical Assistant Professor of Psychiatry
Rosalind Franklin University
Chicago, Illinois
Thank you, Dr. Nasrallah, for your incisive thinking and for bringing our attention as psychiatrists to the crucial issues of our clinical practice. I’d like to offer some nuance on the RAPID acronym. First, I’d like to counterpropose DASH: Delusions, Auditory hallucinations, Strange behavior, Hospital now. This is more in line with getting physicians to tune in to the symptoms that should alarm them and bring them to action. I agree that neurodegeneration and illness recurrence are the problems to address. One unsettled issue remains: With early intervention, can we eventually taper patients off antipsychotics to spare them the metabolic and immune morbidity associated with these medications? There is some evidence that this is possible, but it is difficult to collect data. One of the factors delaying treatment, other than lack of recognition, is the general public’s belief that the treatment is sometimes worse than the disease. If we can address this issue in a nuanced fashion, we may get more “early adopters” of these neuron-sparing treatments.
Michael S. Diamond, MD
Private psychiatric practice
Chevy Chase, Maryland
Dr. Nasrallah is right to focus on brain injury patterns, including inflammation and de-myelination, during psychotic episodes. He and Dr. Roque note that starting a patient on a long-acting injectable antipsychotic as soon as possible may prevent subsequent relapse and further brain damage. However, their editorial omits 2 treatments—minocycline and clemastine—that can help stop CNS inflammation, reduce brain damage, and promote remyelination.
Minocycline has been shown to reduce stroke infarct penumbra size and improve outcomes in functional recovery from stroke.1,2 Minocycline’s effects as a potent CNS anti-inflammatory and antiapoptotic agent are well established.
Clemastine has been shown to improve function in multiple sclerosis by activating oligodendrocyte precursor cells into active agents of myelination and fiber bundle stabilization.3 Clemastine reverses acute leukoencephalopathy.4
If we are to treat acute psychosis as a neurologic emergency, we cannot rely on long-acting injectable antipsychotics as the sole treatment. Psychiatric medication alone is not sufficient across every neuropsychiatric condition in which inflammation and white matter damage are part of the etiology, destruction, and pattern of relapse.
The adverse effects risk of adjunctive minocycline and clemastine is low compared with the potential benefits of stopping inflammation, reducing apoptosis, and jump-starting white matter repair. Doses of oral minocycline in the 50- to 100-mg/d range and oral clemastine in the 1.34- to 2.68-mg/d range together can lead to reduced cranial heat, improved cranial suture mobility, and improved elasticity of white matter bundle tracts palpable on physical examination. Both medications show clinical results in improved emotional self-regulation, according to family reports and clinical observations in the outpatient setting. There is no reason to delay neurologic-based adjunctive treatment when our goal is to prevent and reverse brain damage.
Daniel Kerlinsky, MD
Child Psychiatrist
Clinical Assistant Professor
Burrell College of Osteopathic Medicine
Albuquerque, New Mexico
References
1. Hess DH, Fagan SC. Repurposing an old drug to improve the use and safety of tissue plasminogen activator for acute ischemic stroke: minocycline. Rev Neurol Dis. 2010;30(7 pt 2):55S-61S.
2. Vedantam S, Moller AR. Minocycline: a novel stroke therapy. J Neurol Stroke.
3. Green AJ, Gelfand JM, Cree BA, et al. Clemastine fumarate as a remyelinating therapy for multiple sclerosis (ReBUILD): a randomised, controlled, double-blind, crossover trial. Lancet. 2017;390(10111):2481-2489.
4. Cree BAC, Niu J, Hoi KK, et al. Clemastine rescues myelination defects and promotes functional recovery in hypoxic brain injury. Brain. 2018;141(1):85-98.
Continue to: Dr. Nasrallah responds
Dr. Nasrallah responds
Thanks to my colleagues, Drs. Diamond, Glaser, and Kerlinsky, for their cogent letters about my editorial.
To Dr. Glaser: The “ethics” of conducting placebo-controlled studies when developing a new antipsychotic has been raging for some time. For decades, the FDA has insisted on using a placebo group because around 25% to 30% of research participants respond to placebo, and because participants receiving placebo also complain of many adverse effects. So a new drug has to demonstrate a statistically higher efficacy than a placebo, and the adverse effect profile of the placebo group will put the safety and tolerability profile of a new drug in proper perspective. However, in Europe, they do not conduct placebo-controlled studies; instead, they conduct what is called a “non-inferiority” trial of a new antipsychotic compared with a well-established antipsychotic.
Interestingly, even though the discovery of the neurodegenerative effects of untreated psychosis was only 20 years ago (in 1997 after serial MRI scans revealed progressive atrophy), in the 1960s, the first antipsychotic, chlorpromazine, was compared with placebo in a large national study for 6 months. This study showed without a doubt that chlorpromazine has a higher efficacy than placebo. After the study was done, Dr. Philip May at University of California, Los Angeles looked at what happened to the psychotic patients who received placebo for 6 months and found that they became less responsive to treatment, were re-hospitalized more often, and had more negative symptoms and a poorer overall outcome. That was a clue that untreated psychosis can be harmful, and it supports your point about the ethics of using placebo. In contemporary studies, a trial of oral antipsychotics is 6 weeks, not 6 months. In the year-long, placebo-controlled studies of injectable antipsychotics in stable patients, those who show the slightest increase in delusions, hallucinations, or suicidal/homicidal behavior were promptly taken out of the study and treated. This reduced the “harm,” although not completely. Perhaps the FDA will change its policies and adopt the non-inferiority model. That’s what is done with nonpsychiatric disorders such as pneumonia, stroke, or diabetes. But one last fact has to be stated: The placebo response in anxiety, depression, or psychosis is much higher (25% to 35%) than the 1% placebo response in pneumonia.
To Dr. Diamond: I really like DASH, and it is an acronym for quick symptomatic diagnosis. Speedy treatment then follows with the acronym RAPID to prevent brain damage that gets worse with delay.
As for the second issue of tapering off the antipsychotic medication, the evidence is overwhelming in favor of continuous pharmacotherapy. Just as hypertension and diabetes will return if medications are tapered or stopped, so will psychosis, and vengefully so because treatment resistance increases with each relapse.1 This is also true for bipolar disorder recurrences.2 A recent 20-year follow-up study showed that stopping antipsychotic treatment is associated with a much higher mortality rate than continuation therapy.3 Another 7-year study showed the same thing.4 It is literally deadly, and not just neurodegenerative, for persons with schizophrenia to stop their medications.
To Dr. Kerlinsky: I agree with you about using certain adjunctive pharmacotherapies for acute psychosis, which is associated with neuroinflammation, oxidative stress, and neuropil and myelin damage. I support using agents with anti-inflammatory effects (such as minocycline and omega-3 fatty acid), antioxidant effects (such as N-acetylcysteine), and neuroprotective effects (such as minocycline, clemastine, lithium, vitamin D, erythropoietin, etc.). I refer you to my past editorial, “Are you neuroprotecting your patients? 10 Adjunctive therapies to consider,”5 in which I mentioned all the above. I also pointed out the many neuroprotective effects of atypical antipsychotics in another editorial.6 Although off-label, those supplements can be useful interventions that can ameliorate the gray and white matter damage associated with acute psychotic relapses in patients with schizophrenia.
Henry A. Nasrallah, MD
Editor-in-Chief
The Sydney W. Souers Endowed Chair
Professor and Chairman
Department of Psychiatry and Behavioral Neuroscience
Saint Louis University School of Medicine
St. Louis, Missouri
References
1. Emsley R, Oosthuizen P, Koen L, et al. Comparison of treatment response in second-episode versus first-episode schizophrenia. J Clin Psychopharmacol. 2013;33(1):80-83.
2. Post RM. Preventing the maligna
3. Tiihonen J, Tanskanen A, Taipale H. 20-year nationwide follow-up study on discontinuation of antipsychotic treatment in first-episode schizophrenia. Am J Psychiatry. 2018;175(8):765-773.
4. Taipale H, Mittendorfer-Rutz E, Alexanderson K, et al. Antipsychotics and mortality in a nationwide cohort of 29,823 patients with schizophrenia. Schizophr Res. 2018;197:274-280.
5. Nasrallah HA. Are you neuroprotecting your patients? 10 Adjunctive therapies to consider. Current Psychiatry. 2016;15(12):12-14.
6. Nasrallah HA. A decade after the CATIE study, the focus has shifted from effectiveness to neuroprotection. Current Psychiatry. 2015;14(2):19-21.
I read with great interest Dr. Nasrallah’s editorial, “FAST and RAPID: Acronyms to prevent brain damage in stroke and psychosis” (From the Editor,
Mitchell L. Glaser, MD
Board-Certified Child/Adolescent and General Psychiatrist
Assistant Professor of Psychiatry
Rush University Medical Center
Chairman
Department of Psychiatry
Medical Director of Child/Adolescent Psychiatry
St. Mary/Elizabeth Medical Center
Clinical Assistant Professor of Psychiatry
Rosalind Franklin University
Chicago, Illinois
Thank you, Dr. Nasrallah, for your incisive thinking and for bringing our attention as psychiatrists to the crucial issues of our clinical practice. I’d like to offer some nuance on the RAPID acronym. First, I’d like to counterpropose DASH: Delusions, Auditory hallucinations, Strange behavior, Hospital now. This is more in line with getting physicians to tune in to the symptoms that should alarm them and bring them to action. I agree that neurodegeneration and illness recurrence are the problems to address. One unsettled issue remains: With early intervention, can we eventually taper patients off antipsychotics to spare them the metabolic and immune morbidity associated with these medications? There is some evidence that this is possible, but it is difficult to collect data. One of the factors delaying treatment, other than lack of recognition, is the general public’s belief that the treatment is sometimes worse than the disease. If we can address this issue in a nuanced fashion, we may get more “early adopters” of these neuron-sparing treatments.
Michael S. Diamond, MD
Private psychiatric practice
Chevy Chase, Maryland
Dr. Nasrallah is right to focus on brain injury patterns, including inflammation and de-myelination, during psychotic episodes. He and Dr. Roque note that starting a patient on a long-acting injectable antipsychotic as soon as possible may prevent subsequent relapse and further brain damage. However, their editorial omits 2 treatments—minocycline and clemastine—that can help stop CNS inflammation, reduce brain damage, and promote remyelination.
Minocycline has been shown to reduce stroke infarct penumbra size and improve outcomes in functional recovery from stroke.1,2 Minocycline’s effects as a potent CNS anti-inflammatory and antiapoptotic agent are well established.
Clemastine has been shown to improve function in multiple sclerosis by activating oligodendrocyte precursor cells into active agents of myelination and fiber bundle stabilization.3 Clemastine reverses acute leukoencephalopathy.4
If we are to treat acute psychosis as a neurologic emergency, we cannot rely on long-acting injectable antipsychotics as the sole treatment. Psychiatric medication alone is not sufficient across every neuropsychiatric condition in which inflammation and white matter damage are part of the etiology, destruction, and pattern of relapse.
The adverse effects risk of adjunctive minocycline and clemastine is low compared with the potential benefits of stopping inflammation, reducing apoptosis, and jump-starting white matter repair. Doses of oral minocycline in the 50- to 100-mg/d range and oral clemastine in the 1.34- to 2.68-mg/d range together can lead to reduced cranial heat, improved cranial suture mobility, and improved elasticity of white matter bundle tracts palpable on physical examination. Both medications show clinical results in improved emotional self-regulation, according to family reports and clinical observations in the outpatient setting. There is no reason to delay neurologic-based adjunctive treatment when our goal is to prevent and reverse brain damage.
Daniel Kerlinsky, MD
Child Psychiatrist
Clinical Assistant Professor
Burrell College of Osteopathic Medicine
Albuquerque, New Mexico
References
1. Hess DH, Fagan SC. Repurposing an old drug to improve the use and safety of tissue plasminogen activator for acute ischemic stroke: minocycline. Rev Neurol Dis. 2010;30(7 pt 2):55S-61S.
2. Vedantam S, Moller AR. Minocycline: a novel stroke therapy. J Neurol Stroke.
3. Green AJ, Gelfand JM, Cree BA, et al. Clemastine fumarate as a remyelinating therapy for multiple sclerosis (ReBUILD): a randomised, controlled, double-blind, crossover trial. Lancet. 2017;390(10111):2481-2489.
4. Cree BAC, Niu J, Hoi KK, et al. Clemastine rescues myelination defects and promotes functional recovery in hypoxic brain injury. Brain. 2018;141(1):85-98.
Continue to: Dr. Nasrallah responds
Dr. Nasrallah responds
Thanks to my colleagues, Drs. Diamond, Glaser, and Kerlinsky, for their cogent letters about my editorial.
To Dr. Glaser: The “ethics” of conducting placebo-controlled studies when developing a new antipsychotic has been raging for some time. For decades, the FDA has insisted on using a placebo group because around 25% to 30% of research participants respond to placebo, and because participants receiving placebo also complain of many adverse effects. So a new drug has to demonstrate a statistically higher efficacy than a placebo, and the adverse effect profile of the placebo group will put the safety and tolerability profile of a new drug in proper perspective. However, in Europe, they do not conduct placebo-controlled studies; instead, they conduct what is called a “non-inferiority” trial of a new antipsychotic compared with a well-established antipsychotic.
Interestingly, even though the discovery of the neurodegenerative effects of untreated psychosis was only 20 years ago (in 1997 after serial MRI scans revealed progressive atrophy), in the 1960s, the first antipsychotic, chlorpromazine, was compared with placebo in a large national study for 6 months. This study showed without a doubt that chlorpromazine has a higher efficacy than placebo. After the study was done, Dr. Philip May at University of California, Los Angeles looked at what happened to the psychotic patients who received placebo for 6 months and found that they became less responsive to treatment, were re-hospitalized more often, and had more negative symptoms and a poorer overall outcome. That was a clue that untreated psychosis can be harmful, and it supports your point about the ethics of using placebo. In contemporary studies, a trial of oral antipsychotics is 6 weeks, not 6 months. In the year-long, placebo-controlled studies of injectable antipsychotics in stable patients, those who show the slightest increase in delusions, hallucinations, or suicidal/homicidal behavior were promptly taken out of the study and treated. This reduced the “harm,” although not completely. Perhaps the FDA will change its policies and adopt the non-inferiority model. That’s what is done with nonpsychiatric disorders such as pneumonia, stroke, or diabetes. But one last fact has to be stated: The placebo response in anxiety, depression, or psychosis is much higher (25% to 35%) than the 1% placebo response in pneumonia.
To Dr. Diamond: I really like DASH, and it is an acronym for quick symptomatic diagnosis. Speedy treatment then follows with the acronym RAPID to prevent brain damage that gets worse with delay.
As for the second issue of tapering off the antipsychotic medication, the evidence is overwhelming in favor of continuous pharmacotherapy. Just as hypertension and diabetes will return if medications are tapered or stopped, so will psychosis, and vengefully so because treatment resistance increases with each relapse.1 This is also true for bipolar disorder recurrences.2 A recent 20-year follow-up study showed that stopping antipsychotic treatment is associated with a much higher mortality rate than continuation therapy.3 Another 7-year study showed the same thing.4 It is literally deadly, and not just neurodegenerative, for persons with schizophrenia to stop their medications.
To Dr. Kerlinsky: I agree with you about using certain adjunctive pharmacotherapies for acute psychosis, which is associated with neuroinflammation, oxidative stress, and neuropil and myelin damage. I support using agents with anti-inflammatory effects (such as minocycline and omega-3 fatty acid), antioxidant effects (such as N-acetylcysteine), and neuroprotective effects (such as minocycline, clemastine, lithium, vitamin D, erythropoietin, etc.). I refer you to my past editorial, “Are you neuroprotecting your patients? 10 Adjunctive therapies to consider,”5 in which I mentioned all the above. I also pointed out the many neuroprotective effects of atypical antipsychotics in another editorial.6 Although off-label, those supplements can be useful interventions that can ameliorate the gray and white matter damage associated with acute psychotic relapses in patients with schizophrenia.
Henry A. Nasrallah, MD
Editor-in-Chief
The Sydney W. Souers Endowed Chair
Professor and Chairman
Department of Psychiatry and Behavioral Neuroscience
Saint Louis University School of Medicine
St. Louis, Missouri
References
1. Emsley R, Oosthuizen P, Koen L, et al. Comparison of treatment response in second-episode versus first-episode schizophrenia. J Clin Psychopharmacol. 2013;33(1):80-83.
2. Post RM. Preventing the maligna
3. Tiihonen J, Tanskanen A, Taipale H. 20-year nationwide follow-up study on discontinuation of antipsychotic treatment in first-episode schizophrenia. Am J Psychiatry. 2018;175(8):765-773.
4. Taipale H, Mittendorfer-Rutz E, Alexanderson K, et al. Antipsychotics and mortality in a nationwide cohort of 29,823 patients with schizophrenia. Schizophr Res. 2018;197:274-280.
5. Nasrallah HA. Are you neuroprotecting your patients? 10 Adjunctive therapies to consider. Current Psychiatry. 2016;15(12):12-14.
6. Nasrallah HA. A decade after the CATIE study, the focus has shifted from effectiveness to neuroprotection. Current Psychiatry. 2015;14(2):19-21.
I read with great interest Dr. Nasrallah’s editorial, “FAST and RAPID: Acronyms to prevent brain damage in stroke and psychosis” (From the Editor,
Mitchell L. Glaser, MD
Board-Certified Child/Adolescent and General Psychiatrist
Assistant Professor of Psychiatry
Rush University Medical Center
Chairman
Department of Psychiatry
Medical Director of Child/Adolescent Psychiatry
St. Mary/Elizabeth Medical Center
Clinical Assistant Professor of Psychiatry
Rosalind Franklin University
Chicago, Illinois
Thank you, Dr. Nasrallah, for your incisive thinking and for bringing our attention as psychiatrists to the crucial issues of our clinical practice. I’d like to offer some nuance on the RAPID acronym. First, I’d like to counterpropose DASH: Delusions, Auditory hallucinations, Strange behavior, Hospital now. This is more in line with getting physicians to tune in to the symptoms that should alarm them and bring them to action. I agree that neurodegeneration and illness recurrence are the problems to address. One unsettled issue remains: With early intervention, can we eventually taper patients off antipsychotics to spare them the metabolic and immune morbidity associated with these medications? There is some evidence that this is possible, but it is difficult to collect data. One of the factors delaying treatment, other than lack of recognition, is the general public’s belief that the treatment is sometimes worse than the disease. If we can address this issue in a nuanced fashion, we may get more “early adopters” of these neuron-sparing treatments.
Michael S. Diamond, MD
Private psychiatric practice
Chevy Chase, Maryland
Dr. Nasrallah is right to focus on brain injury patterns, including inflammation and de-myelination, during psychotic episodes. He and Dr. Roque note that starting a patient on a long-acting injectable antipsychotic as soon as possible may prevent subsequent relapse and further brain damage. However, their editorial omits 2 treatments—minocycline and clemastine—that can help stop CNS inflammation, reduce brain damage, and promote remyelination.
Minocycline has been shown to reduce stroke infarct penumbra size and improve outcomes in functional recovery from stroke.1,2 Minocycline’s effects as a potent CNS anti-inflammatory and antiapoptotic agent are well established.
Clemastine has been shown to improve function in multiple sclerosis by activating oligodendrocyte precursor cells into active agents of myelination and fiber bundle stabilization.3 Clemastine reverses acute leukoencephalopathy.4
If we are to treat acute psychosis as a neurologic emergency, we cannot rely on long-acting injectable antipsychotics as the sole treatment. Psychiatric medication alone is not sufficient across every neuropsychiatric condition in which inflammation and white matter damage are part of the etiology, destruction, and pattern of relapse.
The adverse effects risk of adjunctive minocycline and clemastine is low compared with the potential benefits of stopping inflammation, reducing apoptosis, and jump-starting white matter repair. Doses of oral minocycline in the 50- to 100-mg/d range and oral clemastine in the 1.34- to 2.68-mg/d range together can lead to reduced cranial heat, improved cranial suture mobility, and improved elasticity of white matter bundle tracts palpable on physical examination. Both medications show clinical results in improved emotional self-regulation, according to family reports and clinical observations in the outpatient setting. There is no reason to delay neurologic-based adjunctive treatment when our goal is to prevent and reverse brain damage.
Daniel Kerlinsky, MD
Child Psychiatrist
Clinical Assistant Professor
Burrell College of Osteopathic Medicine
Albuquerque, New Mexico
References
1. Hess DH, Fagan SC. Repurposing an old drug to improve the use and safety of tissue plasminogen activator for acute ischemic stroke: minocycline. Rev Neurol Dis. 2010;30(7 pt 2):55S-61S.
2. Vedantam S, Moller AR. Minocycline: a novel stroke therapy. J Neurol Stroke.
3. Green AJ, Gelfand JM, Cree BA, et al. Clemastine fumarate as a remyelinating therapy for multiple sclerosis (ReBUILD): a randomised, controlled, double-blind, crossover trial. Lancet. 2017;390(10111):2481-2489.
4. Cree BAC, Niu J, Hoi KK, et al. Clemastine rescues myelination defects and promotes functional recovery in hypoxic brain injury. Brain. 2018;141(1):85-98.
Continue to: Dr. Nasrallah responds
Dr. Nasrallah responds
Thanks to my colleagues, Drs. Diamond, Glaser, and Kerlinsky, for their cogent letters about my editorial.
To Dr. Glaser: The “ethics” of conducting placebo-controlled studies when developing a new antipsychotic has been raging for some time. For decades, the FDA has insisted on using a placebo group because around 25% to 30% of research participants respond to placebo, and because participants receiving placebo also complain of many adverse effects. So a new drug has to demonstrate a statistically higher efficacy than a placebo, and the adverse effect profile of the placebo group will put the safety and tolerability profile of a new drug in proper perspective. However, in Europe, they do not conduct placebo-controlled studies; instead, they conduct what is called a “non-inferiority” trial of a new antipsychotic compared with a well-established antipsychotic.
Interestingly, even though the discovery of the neurodegenerative effects of untreated psychosis was only 20 years ago (in 1997 after serial MRI scans revealed progressive atrophy), in the 1960s, the first antipsychotic, chlorpromazine, was compared with placebo in a large national study for 6 months. This study showed without a doubt that chlorpromazine has a higher efficacy than placebo. After the study was done, Dr. Philip May at University of California, Los Angeles looked at what happened to the psychotic patients who received placebo for 6 months and found that they became less responsive to treatment, were re-hospitalized more often, and had more negative symptoms and a poorer overall outcome. That was a clue that untreated psychosis can be harmful, and it supports your point about the ethics of using placebo. In contemporary studies, a trial of oral antipsychotics is 6 weeks, not 6 months. In the year-long, placebo-controlled studies of injectable antipsychotics in stable patients, those who show the slightest increase in delusions, hallucinations, or suicidal/homicidal behavior were promptly taken out of the study and treated. This reduced the “harm,” although not completely. Perhaps the FDA will change its policies and adopt the non-inferiority model. That’s what is done with nonpsychiatric disorders such as pneumonia, stroke, or diabetes. But one last fact has to be stated: The placebo response in anxiety, depression, or psychosis is much higher (25% to 35%) than the 1% placebo response in pneumonia.
To Dr. Diamond: I really like DASH, and it is an acronym for quick symptomatic diagnosis. Speedy treatment then follows with the acronym RAPID to prevent brain damage that gets worse with delay.
As for the second issue of tapering off the antipsychotic medication, the evidence is overwhelming in favor of continuous pharmacotherapy. Just as hypertension and diabetes will return if medications are tapered or stopped, so will psychosis, and vengefully so because treatment resistance increases with each relapse.1 This is also true for bipolar disorder recurrences.2 A recent 20-year follow-up study showed that stopping antipsychotic treatment is associated with a much higher mortality rate than continuation therapy.3 Another 7-year study showed the same thing.4 It is literally deadly, and not just neurodegenerative, for persons with schizophrenia to stop their medications.
To Dr. Kerlinsky: I agree with you about using certain adjunctive pharmacotherapies for acute psychosis, which is associated with neuroinflammation, oxidative stress, and neuropil and myelin damage. I support using agents with anti-inflammatory effects (such as minocycline and omega-3 fatty acid), antioxidant effects (such as N-acetylcysteine), and neuroprotective effects (such as minocycline, clemastine, lithium, vitamin D, erythropoietin, etc.). I refer you to my past editorial, “Are you neuroprotecting your patients? 10 Adjunctive therapies to consider,”5 in which I mentioned all the above. I also pointed out the many neuroprotective effects of atypical antipsychotics in another editorial.6 Although off-label, those supplements can be useful interventions that can ameliorate the gray and white matter damage associated with acute psychotic relapses in patients with schizophrenia.
Henry A. Nasrallah, MD
Editor-in-Chief
The Sydney W. Souers Endowed Chair
Professor and Chairman
Department of Psychiatry and Behavioral Neuroscience
Saint Louis University School of Medicine
St. Louis, Missouri
References
1. Emsley R, Oosthuizen P, Koen L, et al. Comparison of treatment response in second-episode versus first-episode schizophrenia. J Clin Psychopharmacol. 2013;33(1):80-83.
2. Post RM. Preventing the maligna
3. Tiihonen J, Tanskanen A, Taipale H. 20-year nationwide follow-up study on discontinuation of antipsychotic treatment in first-episode schizophrenia. Am J Psychiatry. 2018;175(8):765-773.
4. Taipale H, Mittendorfer-Rutz E, Alexanderson K, et al. Antipsychotics and mortality in a nationwide cohort of 29,823 patients with schizophrenia. Schizophr Res. 2018;197:274-280.
5. Nasrallah HA. Are you neuroprotecting your patients? 10 Adjunctive therapies to consider. Current Psychiatry. 2016;15(12):12-14.
6. Nasrallah HA. A decade after the CATIE study, the focus has shifted from effectiveness to neuroprotection. Current Psychiatry. 2015;14(2):19-21.
Vitamin B6 for tardive dyskinesia?
Although antipsychotics have revolutionized the treatment of severe mental illnesses, adverse effects often present a substantial obstacle to adherence. One of the most tenacious and difficult-to-treat adverse effects is tardive dyskinesia (TD), a neuromotor syndrome with characteristic involuntary repetitive movements, typically of the muscles of the jaw, lips, and tongue. In addition to spasms and grimacing, patients can have choreoathetoid movements of the neck. In more extreme presentations, some patients can have difficulty breathing. TD is a largely irreversible condition. It is often a disfiguring lifelong disability that can further stigmatize patients who already suffer scorn and derision. TD usually has a delayed onset after a patient is started on an antipsychotic.1 The syndrome is more commonly associated with first-generation antipsychotics, but affects up to 20% of patients who are treated with second-generation antipsychotics.1 In the United States, TD affects as many as 500,000 patients.1
There are several palliative interventions for TD, but the evidence for a consistently reliable treatment is weak. Branched-chain amino acids, ginkgo biloba, melatonin, and vitamin E have been investigated as interventions. Other approaches include switching to an alternate antipsychotic such as clozapine, adjusting the antipsychotic dose, using anticholinergic medications, adjunctive amantadine, gamma aminobutyric acid agonists, or adding tetrabenazine.
The FDA recently approved two vesicular monoamine transporter 2 (VMAT2) inhibitors, deutetrabenazine and valbenazine, for addressing symptoms of TD. However, these medications can cost tens of thousands of dollars per year, and also carry the risk of adverse effects such as sedation, akathisia, urinary retention, constipation, and muscle pain.2 When treating a patient who develops TD, one might consider other potentially effective therapies with low adverse effect profiles that may be more cost-effective than existing treatments. The bioactive form of vitamin B6 (pyridoxine), pyridoxal-5-phosphate, has been used to treat various antipsychotic-induced movement disorders. Preliminary evidence suggests that vitamin B6 may help reduce the symptoms of TD.
A recent Cochrane Database Review (2015)3 of pyridoxal-5-phosphate treatment for TD found a significant improvement in symptoms compared with placebo. Although the studies included in this review were limited by modest sample sizes and short follow-up periods, 2 of the investigations revealed improvements of >40% in extrapyramidal symptoms with vitamin B6 compared with placebo. Lerner et al (2001)4 conducted a randomized, double-blind, placebo-controlled crossover trial in which 15 inpatients with schizophrenia who met the criteria for TD were assigned to vitamin B6, 400 mg/d, or placebo for 4 weeks. After a 2-week washout period, the placebo group was given vitamin B6 and vice versa. Compared with placebo, mean scores on the parkinsonism and dyskinetic movement subscales of the Extrapyramidal Symptom Rating Scale were significantly better in the third week of treatment with vitamin B6.
Lerner et al (2007)5 later conducted a separate crossover study using the same design with a washout period. This trial included a larger sample size (50 inpatients with DSM-IV diagnoses of schizophrenia or schizoaffective disorder and TD) and the dosage of vitamin B6 was increased to 1,200 mg/d over 26 weeks. Patients who received vitamin B6 experienced a significantly greater decrease in Extrapyramidal Symptom Rating Scale scores compared with those in the placebo group.
Continued to: A 29-year-old woman with treatment-resistant schizophrenia...
Umar et al (2016)6 published a case review of a 29-year-old woman with treatment-resistant schizophrenia with TD who was treated with clozapine, 400 mg/d. She was started on vitamin B6, 450 mg/d, for 4 weeks, and then her dose was increased to 600 mg/d. At 6 months, she experienced a 78% reduction in the severity of her TD symptoms, as measured by the Abnormal Involuntary Movement Scale. The authors reported that this improvement was maintained for 1 year after vitamin B6 was stopped.
Miodownik et al (2008)7 reported in a study of 89 patients with schizophrenia that those with TD (n = 40) had diminished amounts of vitamin B6 in their plasma compared with patients without symptoms of motor disturbances (n = 49).
Vitamin B6 has been known to improve other psychotropic-induced movement disorders. In a study of lithium-induced tremors, treatment with pyridoxine, 900 to 1,200 mg/d, resulted in “impressive improvement until total disappearance of tremor.”8 Lerner et al (2004)9 also reported significant improvement for patients with neuroleptic-induced akathisia who were treated with vitamin B6.
Some proposed mechanisms of action
Pyridoxal-5-phosphate is a coenzyme in the synthesis of dopamine and other neurotransmitters. This might explain in part the biochemical mechanism of vitamin B6 in attenuating motor symptoms following long-term dopamine blockade. Chronic neurotransmitter antagonism may result in an upregulation of dopamine receptors in response. This compensatory reaction might create a dopamine receptor super-sensitivity in the nigrostriatal pathways.10
Another potential mechanism of action might be vitamin B6’s potent antioxidant properties and its scavenging of free radicals. The neurotoxicity of oxidative stress has been implicated in various movement disorders and psychiatric conditions.
In all of the studies described here, patients continued to receive daily antipsychotic treatment. In these trials, the adverse effects of vitamin B6 were minimal or negligible. In one study, vitamin B6 was reported to have had a better adverse effect profile than placebo.4
1. Carbon M, Hsieh CH, Kane JM, et al. Tardive dyskinesia prevalence in the period of second-generation antipsychotic use: a meta-analysis. J Clin Psychiatry. 2017;78(3):e264-e278.
2. Smith Mosley LL, Mosely II JF, Fleischfresser JR, et al. Vesicular monoamine transporter type 2 (VMAT2) inhibitors in the management of tardive dyskinesia. Clin Med Rev Case Rep. 2017;4(12):1-5.
3. Adelufosi AO, Abayomi O, Ojo M. Pyridoxal 5 phosphate for neuroleptic-induced tardive dyskinesia. Cochrane Database Syst Rev. 2015;(4):CD010501.
4. Lerner V, Miodownik C, Kapstan A, et al. Vitamin B(6) in the treatment of tardive dyskinesia: a double-blind, placebo-controlled, crossover study. Am J Psychiatry. 2001;158(9):1511-1514.
5. Lerner V, Miodownik C, Kapstan A, et al. Vitamin B6 treatment for tardive dyskinesia: a randomized, double-blind, placebo-controlled, crossover study. J Clin Psychiatry. 2007;68(11):1648-1654.
6. Umar MU, Isa AA, Abba AH. High dose pyridoxine for the treatment of tardive dyskinesia: clinical case and review of literature. Ther Adv Psychopharmacol. 2016;6(2):152-156.
7. Miodownik C, Meoded A, Libov I, et al. Pyridoxal plasma level in schizophrenic and schizoaffective patients with and without tardive dyskinesia. Clin Neuropharmacol. 2008;31(4):197-203.
8. Miodownik C, Witztum E, Lerner V. Lithium-induced tremor treated with vitamin B6: a preliminary case series. Int J Psychiatry Med. 2002;32(1):103-108.
9. Lerner V, Bergman J, Statsenko N, et al. Vitamin B6 treatment in acute neuroleptic-induced akathisia: a randomized, double-blind, placebo-controlled study. J Clin Psychiatry. 2004;65(11):1550-1554.
10. Miller, BJ. Tardive dyskinesia: a review of the literature. Psychiatric Times. http://www.psychiatrictimes.com/articles/tardive-dyskinesia-review-literature. Published June 27, 2017. Accessed July 31, 2018.
Although antipsychotics have revolutionized the treatment of severe mental illnesses, adverse effects often present a substantial obstacle to adherence. One of the most tenacious and difficult-to-treat adverse effects is tardive dyskinesia (TD), a neuromotor syndrome with characteristic involuntary repetitive movements, typically of the muscles of the jaw, lips, and tongue. In addition to spasms and grimacing, patients can have choreoathetoid movements of the neck. In more extreme presentations, some patients can have difficulty breathing. TD is a largely irreversible condition. It is often a disfiguring lifelong disability that can further stigmatize patients who already suffer scorn and derision. TD usually has a delayed onset after a patient is started on an antipsychotic.1 The syndrome is more commonly associated with first-generation antipsychotics, but affects up to 20% of patients who are treated with second-generation antipsychotics.1 In the United States, TD affects as many as 500,000 patients.1
There are several palliative interventions for TD, but the evidence for a consistently reliable treatment is weak. Branched-chain amino acids, ginkgo biloba, melatonin, and vitamin E have been investigated as interventions. Other approaches include switching to an alternate antipsychotic such as clozapine, adjusting the antipsychotic dose, using anticholinergic medications, adjunctive amantadine, gamma aminobutyric acid agonists, or adding tetrabenazine.
The FDA recently approved two vesicular monoamine transporter 2 (VMAT2) inhibitors, deutetrabenazine and valbenazine, for addressing symptoms of TD. However, these medications can cost tens of thousands of dollars per year, and also carry the risk of adverse effects such as sedation, akathisia, urinary retention, constipation, and muscle pain.2 When treating a patient who develops TD, one might consider other potentially effective therapies with low adverse effect profiles that may be more cost-effective than existing treatments. The bioactive form of vitamin B6 (pyridoxine), pyridoxal-5-phosphate, has been used to treat various antipsychotic-induced movement disorders. Preliminary evidence suggests that vitamin B6 may help reduce the symptoms of TD.
A recent Cochrane Database Review (2015)3 of pyridoxal-5-phosphate treatment for TD found a significant improvement in symptoms compared with placebo. Although the studies included in this review were limited by modest sample sizes and short follow-up periods, 2 of the investigations revealed improvements of >40% in extrapyramidal symptoms with vitamin B6 compared with placebo. Lerner et al (2001)4 conducted a randomized, double-blind, placebo-controlled crossover trial in which 15 inpatients with schizophrenia who met the criteria for TD were assigned to vitamin B6, 400 mg/d, or placebo for 4 weeks. After a 2-week washout period, the placebo group was given vitamin B6 and vice versa. Compared with placebo, mean scores on the parkinsonism and dyskinetic movement subscales of the Extrapyramidal Symptom Rating Scale were significantly better in the third week of treatment with vitamin B6.
Lerner et al (2007)5 later conducted a separate crossover study using the same design with a washout period. This trial included a larger sample size (50 inpatients with DSM-IV diagnoses of schizophrenia or schizoaffective disorder and TD) and the dosage of vitamin B6 was increased to 1,200 mg/d over 26 weeks. Patients who received vitamin B6 experienced a significantly greater decrease in Extrapyramidal Symptom Rating Scale scores compared with those in the placebo group.
Continued to: A 29-year-old woman with treatment-resistant schizophrenia...
Umar et al (2016)6 published a case review of a 29-year-old woman with treatment-resistant schizophrenia with TD who was treated with clozapine, 400 mg/d. She was started on vitamin B6, 450 mg/d, for 4 weeks, and then her dose was increased to 600 mg/d. At 6 months, she experienced a 78% reduction in the severity of her TD symptoms, as measured by the Abnormal Involuntary Movement Scale. The authors reported that this improvement was maintained for 1 year after vitamin B6 was stopped.
Miodownik et al (2008)7 reported in a study of 89 patients with schizophrenia that those with TD (n = 40) had diminished amounts of vitamin B6 in their plasma compared with patients without symptoms of motor disturbances (n = 49).
Vitamin B6 has been known to improve other psychotropic-induced movement disorders. In a study of lithium-induced tremors, treatment with pyridoxine, 900 to 1,200 mg/d, resulted in “impressive improvement until total disappearance of tremor.”8 Lerner et al (2004)9 also reported significant improvement for patients with neuroleptic-induced akathisia who were treated with vitamin B6.
Some proposed mechanisms of action
Pyridoxal-5-phosphate is a coenzyme in the synthesis of dopamine and other neurotransmitters. This might explain in part the biochemical mechanism of vitamin B6 in attenuating motor symptoms following long-term dopamine blockade. Chronic neurotransmitter antagonism may result in an upregulation of dopamine receptors in response. This compensatory reaction might create a dopamine receptor super-sensitivity in the nigrostriatal pathways.10
Another potential mechanism of action might be vitamin B6’s potent antioxidant properties and its scavenging of free radicals. The neurotoxicity of oxidative stress has been implicated in various movement disorders and psychiatric conditions.
In all of the studies described here, patients continued to receive daily antipsychotic treatment. In these trials, the adverse effects of vitamin B6 were minimal or negligible. In one study, vitamin B6 was reported to have had a better adverse effect profile than placebo.4
Although antipsychotics have revolutionized the treatment of severe mental illnesses, adverse effects often present a substantial obstacle to adherence. One of the most tenacious and difficult-to-treat adverse effects is tardive dyskinesia (TD), a neuromotor syndrome with characteristic involuntary repetitive movements, typically of the muscles of the jaw, lips, and tongue. In addition to spasms and grimacing, patients can have choreoathetoid movements of the neck. In more extreme presentations, some patients can have difficulty breathing. TD is a largely irreversible condition. It is often a disfiguring lifelong disability that can further stigmatize patients who already suffer scorn and derision. TD usually has a delayed onset after a patient is started on an antipsychotic.1 The syndrome is more commonly associated with first-generation antipsychotics, but affects up to 20% of patients who are treated with second-generation antipsychotics.1 In the United States, TD affects as many as 500,000 patients.1
There are several palliative interventions for TD, but the evidence for a consistently reliable treatment is weak. Branched-chain amino acids, ginkgo biloba, melatonin, and vitamin E have been investigated as interventions. Other approaches include switching to an alternate antipsychotic such as clozapine, adjusting the antipsychotic dose, using anticholinergic medications, adjunctive amantadine, gamma aminobutyric acid agonists, or adding tetrabenazine.
The FDA recently approved two vesicular monoamine transporter 2 (VMAT2) inhibitors, deutetrabenazine and valbenazine, for addressing symptoms of TD. However, these medications can cost tens of thousands of dollars per year, and also carry the risk of adverse effects such as sedation, akathisia, urinary retention, constipation, and muscle pain.2 When treating a patient who develops TD, one might consider other potentially effective therapies with low adverse effect profiles that may be more cost-effective than existing treatments. The bioactive form of vitamin B6 (pyridoxine), pyridoxal-5-phosphate, has been used to treat various antipsychotic-induced movement disorders. Preliminary evidence suggests that vitamin B6 may help reduce the symptoms of TD.
A recent Cochrane Database Review (2015)3 of pyridoxal-5-phosphate treatment for TD found a significant improvement in symptoms compared with placebo. Although the studies included in this review were limited by modest sample sizes and short follow-up periods, 2 of the investigations revealed improvements of >40% in extrapyramidal symptoms with vitamin B6 compared with placebo. Lerner et al (2001)4 conducted a randomized, double-blind, placebo-controlled crossover trial in which 15 inpatients with schizophrenia who met the criteria for TD were assigned to vitamin B6, 400 mg/d, or placebo for 4 weeks. After a 2-week washout period, the placebo group was given vitamin B6 and vice versa. Compared with placebo, mean scores on the parkinsonism and dyskinetic movement subscales of the Extrapyramidal Symptom Rating Scale were significantly better in the third week of treatment with vitamin B6.
Lerner et al (2007)5 later conducted a separate crossover study using the same design with a washout period. This trial included a larger sample size (50 inpatients with DSM-IV diagnoses of schizophrenia or schizoaffective disorder and TD) and the dosage of vitamin B6 was increased to 1,200 mg/d over 26 weeks. Patients who received vitamin B6 experienced a significantly greater decrease in Extrapyramidal Symptom Rating Scale scores compared with those in the placebo group.
Continued to: A 29-year-old woman with treatment-resistant schizophrenia...
Umar et al (2016)6 published a case review of a 29-year-old woman with treatment-resistant schizophrenia with TD who was treated with clozapine, 400 mg/d. She was started on vitamin B6, 450 mg/d, for 4 weeks, and then her dose was increased to 600 mg/d. At 6 months, she experienced a 78% reduction in the severity of her TD symptoms, as measured by the Abnormal Involuntary Movement Scale. The authors reported that this improvement was maintained for 1 year after vitamin B6 was stopped.
Miodownik et al (2008)7 reported in a study of 89 patients with schizophrenia that those with TD (n = 40) had diminished amounts of vitamin B6 in their plasma compared with patients without symptoms of motor disturbances (n = 49).
Vitamin B6 has been known to improve other psychotropic-induced movement disorders. In a study of lithium-induced tremors, treatment with pyridoxine, 900 to 1,200 mg/d, resulted in “impressive improvement until total disappearance of tremor.”8 Lerner et al (2004)9 also reported significant improvement for patients with neuroleptic-induced akathisia who were treated with vitamin B6.
Some proposed mechanisms of action
Pyridoxal-5-phosphate is a coenzyme in the synthesis of dopamine and other neurotransmitters. This might explain in part the biochemical mechanism of vitamin B6 in attenuating motor symptoms following long-term dopamine blockade. Chronic neurotransmitter antagonism may result in an upregulation of dopamine receptors in response. This compensatory reaction might create a dopamine receptor super-sensitivity in the nigrostriatal pathways.10
Another potential mechanism of action might be vitamin B6’s potent antioxidant properties and its scavenging of free radicals. The neurotoxicity of oxidative stress has been implicated in various movement disorders and psychiatric conditions.
In all of the studies described here, patients continued to receive daily antipsychotic treatment. In these trials, the adverse effects of vitamin B6 were minimal or negligible. In one study, vitamin B6 was reported to have had a better adverse effect profile than placebo.4
1. Carbon M, Hsieh CH, Kane JM, et al. Tardive dyskinesia prevalence in the period of second-generation antipsychotic use: a meta-analysis. J Clin Psychiatry. 2017;78(3):e264-e278.
2. Smith Mosley LL, Mosely II JF, Fleischfresser JR, et al. Vesicular monoamine transporter type 2 (VMAT2) inhibitors in the management of tardive dyskinesia. Clin Med Rev Case Rep. 2017;4(12):1-5.
3. Adelufosi AO, Abayomi O, Ojo M. Pyridoxal 5 phosphate for neuroleptic-induced tardive dyskinesia. Cochrane Database Syst Rev. 2015;(4):CD010501.
4. Lerner V, Miodownik C, Kapstan A, et al. Vitamin B(6) in the treatment of tardive dyskinesia: a double-blind, placebo-controlled, crossover study. Am J Psychiatry. 2001;158(9):1511-1514.
5. Lerner V, Miodownik C, Kapstan A, et al. Vitamin B6 treatment for tardive dyskinesia: a randomized, double-blind, placebo-controlled, crossover study. J Clin Psychiatry. 2007;68(11):1648-1654.
6. Umar MU, Isa AA, Abba AH. High dose pyridoxine for the treatment of tardive dyskinesia: clinical case and review of literature. Ther Adv Psychopharmacol. 2016;6(2):152-156.
7. Miodownik C, Meoded A, Libov I, et al. Pyridoxal plasma level in schizophrenic and schizoaffective patients with and without tardive dyskinesia. Clin Neuropharmacol. 2008;31(4):197-203.
8. Miodownik C, Witztum E, Lerner V. Lithium-induced tremor treated with vitamin B6: a preliminary case series. Int J Psychiatry Med. 2002;32(1):103-108.
9. Lerner V, Bergman J, Statsenko N, et al. Vitamin B6 treatment in acute neuroleptic-induced akathisia: a randomized, double-blind, placebo-controlled study. J Clin Psychiatry. 2004;65(11):1550-1554.
10. Miller, BJ. Tardive dyskinesia: a review of the literature. Psychiatric Times. http://www.psychiatrictimes.com/articles/tardive-dyskinesia-review-literature. Published June 27, 2017. Accessed July 31, 2018.
1. Carbon M, Hsieh CH, Kane JM, et al. Tardive dyskinesia prevalence in the period of second-generation antipsychotic use: a meta-analysis. J Clin Psychiatry. 2017;78(3):e264-e278.
2. Smith Mosley LL, Mosely II JF, Fleischfresser JR, et al. Vesicular monoamine transporter type 2 (VMAT2) inhibitors in the management of tardive dyskinesia. Clin Med Rev Case Rep. 2017;4(12):1-5.
3. Adelufosi AO, Abayomi O, Ojo M. Pyridoxal 5 phosphate for neuroleptic-induced tardive dyskinesia. Cochrane Database Syst Rev. 2015;(4):CD010501.
4. Lerner V, Miodownik C, Kapstan A, et al. Vitamin B(6) in the treatment of tardive dyskinesia: a double-blind, placebo-controlled, crossover study. Am J Psychiatry. 2001;158(9):1511-1514.
5. Lerner V, Miodownik C, Kapstan A, et al. Vitamin B6 treatment for tardive dyskinesia: a randomized, double-blind, placebo-controlled, crossover study. J Clin Psychiatry. 2007;68(11):1648-1654.
6. Umar MU, Isa AA, Abba AH. High dose pyridoxine for the treatment of tardive dyskinesia: clinical case and review of literature. Ther Adv Psychopharmacol. 2016;6(2):152-156.
7. Miodownik C, Meoded A, Libov I, et al. Pyridoxal plasma level in schizophrenic and schizoaffective patients with and without tardive dyskinesia. Clin Neuropharmacol. 2008;31(4):197-203.
8. Miodownik C, Witztum E, Lerner V. Lithium-induced tremor treated with vitamin B6: a preliminary case series. Int J Psychiatry Med. 2002;32(1):103-108.
9. Lerner V, Bergman J, Statsenko N, et al. Vitamin B6 treatment in acute neuroleptic-induced akathisia: a randomized, double-blind, placebo-controlled study. J Clin Psychiatry. 2004;65(11):1550-1554.
10. Miller, BJ. Tardive dyskinesia: a review of the literature. Psychiatric Times. http://www.psychiatrictimes.com/articles/tardive-dyskinesia-review-literature. Published June 27, 2017. Accessed July 31, 2018.
FDA review supports Nuplazid’s safety
Pimavanserin (Nuplazid) remains an acceptable treatment for the hallucinations and delusions associated with Parkinson’s disease, according to a statement issued by the Food and Drug Administration after the agency conducted a postmarketing review of deaths and serious adverse events associated with the drug.
“Based on an analysis of all available data, FDA did not identify any new or unexpected safety findings” associated with the drug, according to the Sept. 20 statement.
However, the FDA researchers identified prescribing patterns that might increase the risk of serious adverse events, such as the concomitant use of pimavanserin and other antipsychotic drugs or drugs that can cause QT prolongation. The QT prolongation risk is listed on the drug label, which also includes a Boxed Warning about increased mortality risk in elderly patients.
The FDA statement reminds clinicians to know the risks described in the label and to be aware that no other antipsychotics are currently approved for psychosis in Parkinson’s patients.
The review was prompted by the number of reports of serious adverse events and deaths associated with pimavanserin, based on data obtained from multiple sources including the FDA Adverse Event Reporting System (FAERS), drug utilization data, safety data from the new drug application, the sponsor’s Periodic Adverse Drug Experience Reports, the sponsor’s analysis of fatal adverse event reports, and data from published medical literature.
When conducting the review, the FDA considered several factors, including the fact that Parkinson’s disease patients have higher mortality in general because of older age, advanced disease, and other medical comorbidities. In addition, pimavanserin adverse events and deaths are more likely to be reported because the drug is distributed mainly through a patient-support program and specialty pharmacy. The FDA also found no pattern suggestive of a drug effect on causes of death in patients whose deaths were reported through FAERS.
“Overall, the postmarketing data were consistent with the safety data obtained from the premarketing controlled clinical trials of Nuplazid for Parkinson’s disease psychosis,” according to the FDA statement.
Pimavanserin (Nuplazid) remains an acceptable treatment for the hallucinations and delusions associated with Parkinson’s disease, according to a statement issued by the Food and Drug Administration after the agency conducted a postmarketing review of deaths and serious adverse events associated with the drug.
“Based on an analysis of all available data, FDA did not identify any new or unexpected safety findings” associated with the drug, according to the Sept. 20 statement.
However, the FDA researchers identified prescribing patterns that might increase the risk of serious adverse events, such as the concomitant use of pimavanserin and other antipsychotic drugs or drugs that can cause QT prolongation. The QT prolongation risk is listed on the drug label, which also includes a Boxed Warning about increased mortality risk in elderly patients.
The FDA statement reminds clinicians to know the risks described in the label and to be aware that no other antipsychotics are currently approved for psychosis in Parkinson’s patients.
The review was prompted by the number of reports of serious adverse events and deaths associated with pimavanserin, based on data obtained from multiple sources including the FDA Adverse Event Reporting System (FAERS), drug utilization data, safety data from the new drug application, the sponsor’s Periodic Adverse Drug Experience Reports, the sponsor’s analysis of fatal adverse event reports, and data from published medical literature.
When conducting the review, the FDA considered several factors, including the fact that Parkinson’s disease patients have higher mortality in general because of older age, advanced disease, and other medical comorbidities. In addition, pimavanserin adverse events and deaths are more likely to be reported because the drug is distributed mainly through a patient-support program and specialty pharmacy. The FDA also found no pattern suggestive of a drug effect on causes of death in patients whose deaths were reported through FAERS.
“Overall, the postmarketing data were consistent with the safety data obtained from the premarketing controlled clinical trials of Nuplazid for Parkinson’s disease psychosis,” according to the FDA statement.
Pimavanserin (Nuplazid) remains an acceptable treatment for the hallucinations and delusions associated with Parkinson’s disease, according to a statement issued by the Food and Drug Administration after the agency conducted a postmarketing review of deaths and serious adverse events associated with the drug.
“Based on an analysis of all available data, FDA did not identify any new or unexpected safety findings” associated with the drug, according to the Sept. 20 statement.
However, the FDA researchers identified prescribing patterns that might increase the risk of serious adverse events, such as the concomitant use of pimavanserin and other antipsychotic drugs or drugs that can cause QT prolongation. The QT prolongation risk is listed on the drug label, which also includes a Boxed Warning about increased mortality risk in elderly patients.
The FDA statement reminds clinicians to know the risks described in the label and to be aware that no other antipsychotics are currently approved for psychosis in Parkinson’s patients.
The review was prompted by the number of reports of serious adverse events and deaths associated with pimavanserin, based on data obtained from multiple sources including the FDA Adverse Event Reporting System (FAERS), drug utilization data, safety data from the new drug application, the sponsor’s Periodic Adverse Drug Experience Reports, the sponsor’s analysis of fatal adverse event reports, and data from published medical literature.
When conducting the review, the FDA considered several factors, including the fact that Parkinson’s disease patients have higher mortality in general because of older age, advanced disease, and other medical comorbidities. In addition, pimavanserin adverse events and deaths are more likely to be reported because the drug is distributed mainly through a patient-support program and specialty pharmacy. The FDA also found no pattern suggestive of a drug effect on causes of death in patients whose deaths were reported through FAERS.
“Overall, the postmarketing data were consistent with the safety data obtained from the premarketing controlled clinical trials of Nuplazid for Parkinson’s disease psychosis,” according to the FDA statement.
Maternal infanticide and postpartum psychosis
Postpartum psychosis is extremely rare, but when it does occur, the consequences can prove catastrophic.
An article in The Atlantic chronicles the nightmare suffered by mothers driven to kill their children.
Cara Angelotta, MD, a forensic psychiatrist affiliated with Northwestern University in Chicago, says the presence of postpartum psychosis also puts mothers’ lives at risk. “Suicide is a major contributor to maternal death in the first year postpartum. [And] postpartum psychosis increases the risk of suicide,” she says.
“People don’t get it,” says Susan Benjamin Feingold, PsyD, a psychologist at the Illinois School of Professional Psychology in Chicago and an expert in postpartum disorders. “They see baby killers. These women are really sick and need treatment, not punishment.”
On June 1, Illinois became the first state to implement a law that allows judges to consider postpartum psychosis during sentencing. When left untreated, postpartum psychosis might raise the risk of infanticide by about 4%, the article in The Atlantic noted.
Thank you for being a friend
Some senior adults are benefiting from the friendship, security, and invigoration provided by an initiative of several universities in which they share living spaces with students. The arrangement, according to the Canadian Broadcasting Corporation, saves the student on housing costs and provides the senior with companionship and assistance with household chores.
But these arrangements have deeper and more enriching benefits.
About 25% of older adults lack family assistance with everyday tasks and live without the companionship that is a hallmark of multiperson households. “It actually gives some peace of mind to that older person,” says Samir Sinha, MD, director of geriatrics at Toronto’s Mount Sinai Hospital.
“It gives them a greater sense of security and the ability to feel like they can maintain their independence for that much longer.”
Jobs and the deaf community
Hearing loss can cause a retreat from the world. But in one San Francisco neighborhood, it is not a barrier to work. As reported by National Public Radio (NPR), every staff member at Mozzeria is partly or fully deaf. Using pen-and-paper ordering or the tried and true pointing at the menu that eases the interchange between hearing-enabled patrons not versed in American Sign Language and hearing-challenged staff, Mozzeria owners Melody and Russ Stein provide solid employment for people for whom a job could otherwise be elusive. Only about 48% of the deaf community is employed in the United States (compared with 72% of hearing individuals), according to the National Deaf Center.
“We envision that each Mozzeria location will be looked at as a source of both local and national pride,” Melody Stein wrote in a recent article.
An alternative to opioids?
Jelena M. Janjic, PhD, a pain researcher at Duquesne University in Pittsburgh, is drawing on her expertise in nanotechnology and her personal experience to develop a means of delivering nonopioid drugs directly to cells in the body. If successful in humans, as it has been in rats, the method would free people from using addictive opioids for pain relief.
As reported by NPR, a motivation for the tact is Dr. Janjic’s own decades-long battle with chronic pain. Personal introspection and literature searches led her to the view that chronic pain can be rooted in inflammation. Quelling the inflammation without disabling the good that the immune system does in fighting infections and other harms might help ease the pain.
“As a patient, I want an answer,” Dr. Janjic says in the interview. “I want to figure out this.”
Marshmallows and self-control
The world recently bid goodbye to Walter Mischel, PhD, who died on Sept. 12. Dr. Mischel is chiefly remembered for his ionic marshmallow test.
In the test of delayed gratification, children were presented with a treat (sometimes the famous marshmallow) then asked to hold off gobbling down the treat in exchange for an even more treats later.
As described in The New Yorker, an inspiration for Dr. Mischel was his repeated attempts to quit his prodigious smoking addiction. His research was one of the first glimpses into the role of self-control as a beneficial strategy in life. Learning to mentally temper that got-to-have-it moment is, according to Dr. Mischel, key to self-control.
“If we have the skills to allow us to make discriminations about when we do or don’t do something, when we do or don’t drink something, and when we do and when we don’t wait for something, we are no longer victims of our desires,” he said in the New Yorker interview.
Postpartum psychosis is extremely rare, but when it does occur, the consequences can prove catastrophic.
An article in The Atlantic chronicles the nightmare suffered by mothers driven to kill their children.
Cara Angelotta, MD, a forensic psychiatrist affiliated with Northwestern University in Chicago, says the presence of postpartum psychosis also puts mothers’ lives at risk. “Suicide is a major contributor to maternal death in the first year postpartum. [And] postpartum psychosis increases the risk of suicide,” she says.
“People don’t get it,” says Susan Benjamin Feingold, PsyD, a psychologist at the Illinois School of Professional Psychology in Chicago and an expert in postpartum disorders. “They see baby killers. These women are really sick and need treatment, not punishment.”
On June 1, Illinois became the first state to implement a law that allows judges to consider postpartum psychosis during sentencing. When left untreated, postpartum psychosis might raise the risk of infanticide by about 4%, the article in The Atlantic noted.
Thank you for being a friend
Some senior adults are benefiting from the friendship, security, and invigoration provided by an initiative of several universities in which they share living spaces with students. The arrangement, according to the Canadian Broadcasting Corporation, saves the student on housing costs and provides the senior with companionship and assistance with household chores.
But these arrangements have deeper and more enriching benefits.
About 25% of older adults lack family assistance with everyday tasks and live without the companionship that is a hallmark of multiperson households. “It actually gives some peace of mind to that older person,” says Samir Sinha, MD, director of geriatrics at Toronto’s Mount Sinai Hospital.
“It gives them a greater sense of security and the ability to feel like they can maintain their independence for that much longer.”
Jobs and the deaf community
Hearing loss can cause a retreat from the world. But in one San Francisco neighborhood, it is not a barrier to work. As reported by National Public Radio (NPR), every staff member at Mozzeria is partly or fully deaf. Using pen-and-paper ordering or the tried and true pointing at the menu that eases the interchange between hearing-enabled patrons not versed in American Sign Language and hearing-challenged staff, Mozzeria owners Melody and Russ Stein provide solid employment for people for whom a job could otherwise be elusive. Only about 48% of the deaf community is employed in the United States (compared with 72% of hearing individuals), according to the National Deaf Center.
“We envision that each Mozzeria location will be looked at as a source of both local and national pride,” Melody Stein wrote in a recent article.
An alternative to opioids?
Jelena M. Janjic, PhD, a pain researcher at Duquesne University in Pittsburgh, is drawing on her expertise in nanotechnology and her personal experience to develop a means of delivering nonopioid drugs directly to cells in the body. If successful in humans, as it has been in rats, the method would free people from using addictive opioids for pain relief.
As reported by NPR, a motivation for the tact is Dr. Janjic’s own decades-long battle with chronic pain. Personal introspection and literature searches led her to the view that chronic pain can be rooted in inflammation. Quelling the inflammation without disabling the good that the immune system does in fighting infections and other harms might help ease the pain.
“As a patient, I want an answer,” Dr. Janjic says in the interview. “I want to figure out this.”
Marshmallows and self-control
The world recently bid goodbye to Walter Mischel, PhD, who died on Sept. 12. Dr. Mischel is chiefly remembered for his ionic marshmallow test.
In the test of delayed gratification, children were presented with a treat (sometimes the famous marshmallow) then asked to hold off gobbling down the treat in exchange for an even more treats later.
As described in The New Yorker, an inspiration for Dr. Mischel was his repeated attempts to quit his prodigious smoking addiction. His research was one of the first glimpses into the role of self-control as a beneficial strategy in life. Learning to mentally temper that got-to-have-it moment is, according to Dr. Mischel, key to self-control.
“If we have the skills to allow us to make discriminations about when we do or don’t do something, when we do or don’t drink something, and when we do and when we don’t wait for something, we are no longer victims of our desires,” he said in the New Yorker interview.
Postpartum psychosis is extremely rare, but when it does occur, the consequences can prove catastrophic.
An article in The Atlantic chronicles the nightmare suffered by mothers driven to kill their children.
Cara Angelotta, MD, a forensic psychiatrist affiliated with Northwestern University in Chicago, says the presence of postpartum psychosis also puts mothers’ lives at risk. “Suicide is a major contributor to maternal death in the first year postpartum. [And] postpartum psychosis increases the risk of suicide,” she says.
“People don’t get it,” says Susan Benjamin Feingold, PsyD, a psychologist at the Illinois School of Professional Psychology in Chicago and an expert in postpartum disorders. “They see baby killers. These women are really sick and need treatment, not punishment.”
On June 1, Illinois became the first state to implement a law that allows judges to consider postpartum psychosis during sentencing. When left untreated, postpartum psychosis might raise the risk of infanticide by about 4%, the article in The Atlantic noted.
Thank you for being a friend
Some senior adults are benefiting from the friendship, security, and invigoration provided by an initiative of several universities in which they share living spaces with students. The arrangement, according to the Canadian Broadcasting Corporation, saves the student on housing costs and provides the senior with companionship and assistance with household chores.
But these arrangements have deeper and more enriching benefits.
About 25% of older adults lack family assistance with everyday tasks and live without the companionship that is a hallmark of multiperson households. “It actually gives some peace of mind to that older person,” says Samir Sinha, MD, director of geriatrics at Toronto’s Mount Sinai Hospital.
“It gives them a greater sense of security and the ability to feel like they can maintain their independence for that much longer.”
Jobs and the deaf community
Hearing loss can cause a retreat from the world. But in one San Francisco neighborhood, it is not a barrier to work. As reported by National Public Radio (NPR), every staff member at Mozzeria is partly or fully deaf. Using pen-and-paper ordering or the tried and true pointing at the menu that eases the interchange between hearing-enabled patrons not versed in American Sign Language and hearing-challenged staff, Mozzeria owners Melody and Russ Stein provide solid employment for people for whom a job could otherwise be elusive. Only about 48% of the deaf community is employed in the United States (compared with 72% of hearing individuals), according to the National Deaf Center.
“We envision that each Mozzeria location will be looked at as a source of both local and national pride,” Melody Stein wrote in a recent article.
An alternative to opioids?
Jelena M. Janjic, PhD, a pain researcher at Duquesne University in Pittsburgh, is drawing on her expertise in nanotechnology and her personal experience to develop a means of delivering nonopioid drugs directly to cells in the body. If successful in humans, as it has been in rats, the method would free people from using addictive opioids for pain relief.
As reported by NPR, a motivation for the tact is Dr. Janjic’s own decades-long battle with chronic pain. Personal introspection and literature searches led her to the view that chronic pain can be rooted in inflammation. Quelling the inflammation without disabling the good that the immune system does in fighting infections and other harms might help ease the pain.
“As a patient, I want an answer,” Dr. Janjic says in the interview. “I want to figure out this.”
Marshmallows and self-control
The world recently bid goodbye to Walter Mischel, PhD, who died on Sept. 12. Dr. Mischel is chiefly remembered for his ionic marshmallow test.
In the test of delayed gratification, children were presented with a treat (sometimes the famous marshmallow) then asked to hold off gobbling down the treat in exchange for an even more treats later.
As described in The New Yorker, an inspiration for Dr. Mischel was his repeated attempts to quit his prodigious smoking addiction. His research was one of the first glimpses into the role of self-control as a beneficial strategy in life. Learning to mentally temper that got-to-have-it moment is, according to Dr. Mischel, key to self-control.
“If we have the skills to allow us to make discriminations about when we do or don’t do something, when we do or don’t drink something, and when we do and when we don’t wait for something, we are no longer victims of our desires,” he said in the New Yorker interview.









