User login
Dr. Dinah Miller, Dr. Annette Hanson on involuntary psychiatric care
Dr. Miller is a private practice psychiatrist and professor at Johns Hopkins University. Dr. Hanson is the director of the forensic psychiatry fellowship at the University of Maryland. Dr. Miller and Dr. Hanson are board members for Clinical Psychiatry News.
Dr. Miller is a private practice psychiatrist and professor at Johns Hopkins University. Dr. Hanson is the director of the forensic psychiatry fellowship at the University of Maryland. Dr. Miller and Dr. Hanson are board members for Clinical Psychiatry News.
Dr. Miller is a private practice psychiatrist and professor at Johns Hopkins University. Dr. Hanson is the director of the forensic psychiatry fellowship at the University of Maryland. Dr. Miller and Dr. Hanson are board members for Clinical Psychiatry News.
A call for psychiatrists with tardive dyskinesia expertise
The CURESZ Foundation was founded in 2016 to bring hope to people suffering from schizophrenia and those who love and care for them. CURESZ was established by Bethany Yeiser and her psychiatrist, Current Psychiatry Editor-in-Chief Henry Nasrallah, MD, and was inspired by Bethany's complete recovery from schizophrenia after 4 years of delusions, hallucinations, homelessness, and disability. Bethany returned to her normal life and graduated from college with honors, thanks to clozapine, which cured her symptoms when several other medications did not work (for more of Bethany’s story, see From the Editor, Current Psychiatry. October 2014, p. 21,24-25).
We previously assembled a panel of clozapine experts to whom the CURESZ Foundation would refer patients who have never had a trial of clozapine despite ongoing delusions or hallucinations. We now have a panel of 80 clozapine experts around the country who are willing to receive referrals.
In an unexpected turn of events, after several years of receiving clozapine, Bethany developed tardive dyskinesia (TD) which, fortunately, was successfully treated. Bethany would not have been able to recover from her TD had it not been for the recent FDA approval of effective treatments. The embarrassing personal experience of oro-buccal TD movements that Bethany went through before she improved led her and me to establish a panel of experts in the recognition and treatment of TD around the country. It is estimated that hundreds of thousands of patients with schizophrenia, schizo-affective disorders, bipolar disorder and major depression, all of whom receive first or second generation antipsychotic agents, currently have TD that is not being diagnosed or treated.
We are therefore calling for psychiatric practitioners who have had experience in recognizing TD movements and have treated patients with FDA-approved treatments, to contact the CURESZ Foundation. Henry Nasrallah, MD, the Scientific Director of the CURESZ Foundation, who has had many years of federally funded research experience in TD, will serve as the Chair of this TD Panel.
This is a call for readers of Current Psychiatry who are treating TD and who practice in settings that can accommodate additional patients seeking treatment for their involuntary TD muscle movements in their face, trunk, and extremities. We hope to assemble between 50 to 100 experts to join this national TD panel.
If you would like to be a member of this national CURESZ TD Panel, please go to https://curesz.org/tardive-dyskinesia-panel/ and enter your name, email, work address, and office phone number. We will later organize the list by state and city so that patients and families around the country can contact the nearest expert to get an evaluation for assessment and treatment of their TD.
Thank you and we look forward to working with the experts who say “YESZ” to joining the TD Panel, sponsored by the CURESZ Foundation.
The CURESZ Foundation was founded in 2016 to bring hope to people suffering from schizophrenia and those who love and care for them. CURESZ was established by Bethany Yeiser and her psychiatrist, Current Psychiatry Editor-in-Chief Henry Nasrallah, MD, and was inspired by Bethany's complete recovery from schizophrenia after 4 years of delusions, hallucinations, homelessness, and disability. Bethany returned to her normal life and graduated from college with honors, thanks to clozapine, which cured her symptoms when several other medications did not work (for more of Bethany’s story, see From the Editor, Current Psychiatry. October 2014, p. 21,24-25).
We previously assembled a panel of clozapine experts to whom the CURESZ Foundation would refer patients who have never had a trial of clozapine despite ongoing delusions or hallucinations. We now have a panel of 80 clozapine experts around the country who are willing to receive referrals.
In an unexpected turn of events, after several years of receiving clozapine, Bethany developed tardive dyskinesia (TD) which, fortunately, was successfully treated. Bethany would not have been able to recover from her TD had it not been for the recent FDA approval of effective treatments. The embarrassing personal experience of oro-buccal TD movements that Bethany went through before she improved led her and me to establish a panel of experts in the recognition and treatment of TD around the country. It is estimated that hundreds of thousands of patients with schizophrenia, schizo-affective disorders, bipolar disorder and major depression, all of whom receive first or second generation antipsychotic agents, currently have TD that is not being diagnosed or treated.
We are therefore calling for psychiatric practitioners who have had experience in recognizing TD movements and have treated patients with FDA-approved treatments, to contact the CURESZ Foundation. Henry Nasrallah, MD, the Scientific Director of the CURESZ Foundation, who has had many years of federally funded research experience in TD, will serve as the Chair of this TD Panel.
This is a call for readers of Current Psychiatry who are treating TD and who practice in settings that can accommodate additional patients seeking treatment for their involuntary TD muscle movements in their face, trunk, and extremities. We hope to assemble between 50 to 100 experts to join this national TD panel.
If you would like to be a member of this national CURESZ TD Panel, please go to https://curesz.org/tardive-dyskinesia-panel/ and enter your name, email, work address, and office phone number. We will later organize the list by state and city so that patients and families around the country can contact the nearest expert to get an evaluation for assessment and treatment of their TD.
Thank you and we look forward to working with the experts who say “YESZ” to joining the TD Panel, sponsored by the CURESZ Foundation.
The CURESZ Foundation was founded in 2016 to bring hope to people suffering from schizophrenia and those who love and care for them. CURESZ was established by Bethany Yeiser and her psychiatrist, Current Psychiatry Editor-in-Chief Henry Nasrallah, MD, and was inspired by Bethany's complete recovery from schizophrenia after 4 years of delusions, hallucinations, homelessness, and disability. Bethany returned to her normal life and graduated from college with honors, thanks to clozapine, which cured her symptoms when several other medications did not work (for more of Bethany’s story, see From the Editor, Current Psychiatry. October 2014, p. 21,24-25).
We previously assembled a panel of clozapine experts to whom the CURESZ Foundation would refer patients who have never had a trial of clozapine despite ongoing delusions or hallucinations. We now have a panel of 80 clozapine experts around the country who are willing to receive referrals.
In an unexpected turn of events, after several years of receiving clozapine, Bethany developed tardive dyskinesia (TD) which, fortunately, was successfully treated. Bethany would not have been able to recover from her TD had it not been for the recent FDA approval of effective treatments. The embarrassing personal experience of oro-buccal TD movements that Bethany went through before she improved led her and me to establish a panel of experts in the recognition and treatment of TD around the country. It is estimated that hundreds of thousands of patients with schizophrenia, schizo-affective disorders, bipolar disorder and major depression, all of whom receive first or second generation antipsychotic agents, currently have TD that is not being diagnosed or treated.
We are therefore calling for psychiatric practitioners who have had experience in recognizing TD movements and have treated patients with FDA-approved treatments, to contact the CURESZ Foundation. Henry Nasrallah, MD, the Scientific Director of the CURESZ Foundation, who has had many years of federally funded research experience in TD, will serve as the Chair of this TD Panel.
This is a call for readers of Current Psychiatry who are treating TD and who practice in settings that can accommodate additional patients seeking treatment for their involuntary TD muscle movements in their face, trunk, and extremities. We hope to assemble between 50 to 100 experts to join this national TD panel.
If you would like to be a member of this national CURESZ TD Panel, please go to https://curesz.org/tardive-dyskinesia-panel/ and enter your name, email, work address, and office phone number. We will later organize the list by state and city so that patients and families around the country can contact the nearest expert to get an evaluation for assessment and treatment of their TD.
Thank you and we look forward to working with the experts who say “YESZ” to joining the TD Panel, sponsored by the CURESZ Foundation.
Suicidal ideation, maladaptive beliefs appear linked in some psychotic disorders
Maladaptive beliefs and face emotion processing appear associated with suicidal ideation and behavior in psychosis, results of a retrospective, cross-sectional study suggest.
“The present findings suggest that maladaptive beliefs are associated with a tendency to misperceive neutral stimuli as threatening, and are associated with suicidal ideation and behavior,” wrote Jennifer Villa of San Diego State University and her associates.
In the study, published in the Journal of Psychiatric Research, 101 outpatients aged 18 and older with psychotic disorders were assessed via the Interpersonal Needs Questionnaire–15 (INQ-15) and the Penn Emotion Recognition Task (ER-40). The participants also were assessed via several other measures, including the Modified Scale for Suicidal Ideation (MSSI), reported Ms. Villa. The INQ-15, a self-report measure, assesses interpersonal beliefs that underlie the desire for suicide. The MSSI, an 18-item instrument, measures the presence of ideation in the previous 48 hours.
Ms. Villa and her associates found that , compared with those without any past history of attempts or current ideation. In addition, MSSI total scores were correlated with INQ total scores (P = .002). When comparing disorders, patients with bipolar disorder with MSSI current ideation had higher INQ total scores than did patients without ideation (P = .010) vs. no MSSI current ideation.
They cited several limitations. One is that the findings might not be generalizable, because causal relationships between maladaptive beliefs, emotion recognition, or the risk of suicidal ideation or behavior cannot be inferred. Also, because most of the patients in the sample were middle-aged adults, the findings might not apply to patients who are experiencing first-episode psychosis. Nevertheless, they said, “the results are of clinical interest demonstrating the growing importance of social cognition to the cumulative evaluation of suicide risk in psychosis and identification of potential targets for suicide prevention.”
Ms. Villa reported no conflicts of interest. Two of the authors reported relationships with several pharmaceutical companies.
SOURCE: Villa J et al. J Psychiatr Res. 2018 Ma;100:107-12.
Maladaptive beliefs and face emotion processing appear associated with suicidal ideation and behavior in psychosis, results of a retrospective, cross-sectional study suggest.
“The present findings suggest that maladaptive beliefs are associated with a tendency to misperceive neutral stimuli as threatening, and are associated with suicidal ideation and behavior,” wrote Jennifer Villa of San Diego State University and her associates.
In the study, published in the Journal of Psychiatric Research, 101 outpatients aged 18 and older with psychotic disorders were assessed via the Interpersonal Needs Questionnaire–15 (INQ-15) and the Penn Emotion Recognition Task (ER-40). The participants also were assessed via several other measures, including the Modified Scale for Suicidal Ideation (MSSI), reported Ms. Villa. The INQ-15, a self-report measure, assesses interpersonal beliefs that underlie the desire for suicide. The MSSI, an 18-item instrument, measures the presence of ideation in the previous 48 hours.
Ms. Villa and her associates found that , compared with those without any past history of attempts or current ideation. In addition, MSSI total scores were correlated with INQ total scores (P = .002). When comparing disorders, patients with bipolar disorder with MSSI current ideation had higher INQ total scores than did patients without ideation (P = .010) vs. no MSSI current ideation.
They cited several limitations. One is that the findings might not be generalizable, because causal relationships between maladaptive beliefs, emotion recognition, or the risk of suicidal ideation or behavior cannot be inferred. Also, because most of the patients in the sample were middle-aged adults, the findings might not apply to patients who are experiencing first-episode psychosis. Nevertheless, they said, “the results are of clinical interest demonstrating the growing importance of social cognition to the cumulative evaluation of suicide risk in psychosis and identification of potential targets for suicide prevention.”
Ms. Villa reported no conflicts of interest. Two of the authors reported relationships with several pharmaceutical companies.
SOURCE: Villa J et al. J Psychiatr Res. 2018 Ma;100:107-12.
Maladaptive beliefs and face emotion processing appear associated with suicidal ideation and behavior in psychosis, results of a retrospective, cross-sectional study suggest.
“The present findings suggest that maladaptive beliefs are associated with a tendency to misperceive neutral stimuli as threatening, and are associated with suicidal ideation and behavior,” wrote Jennifer Villa of San Diego State University and her associates.
In the study, published in the Journal of Psychiatric Research, 101 outpatients aged 18 and older with psychotic disorders were assessed via the Interpersonal Needs Questionnaire–15 (INQ-15) and the Penn Emotion Recognition Task (ER-40). The participants also were assessed via several other measures, including the Modified Scale for Suicidal Ideation (MSSI), reported Ms. Villa. The INQ-15, a self-report measure, assesses interpersonal beliefs that underlie the desire for suicide. The MSSI, an 18-item instrument, measures the presence of ideation in the previous 48 hours.
Ms. Villa and her associates found that , compared with those without any past history of attempts or current ideation. In addition, MSSI total scores were correlated with INQ total scores (P = .002). When comparing disorders, patients with bipolar disorder with MSSI current ideation had higher INQ total scores than did patients without ideation (P = .010) vs. no MSSI current ideation.
They cited several limitations. One is that the findings might not be generalizable, because causal relationships between maladaptive beliefs, emotion recognition, or the risk of suicidal ideation or behavior cannot be inferred. Also, because most of the patients in the sample were middle-aged adults, the findings might not apply to patients who are experiencing first-episode psychosis. Nevertheless, they said, “the results are of clinical interest demonstrating the growing importance of social cognition to the cumulative evaluation of suicide risk in psychosis and identification of potential targets for suicide prevention.”
Ms. Villa reported no conflicts of interest. Two of the authors reported relationships with several pharmaceutical companies.
SOURCE: Villa J et al. J Psychiatr Res. 2018 Ma;100:107-12.
FROM THE JOURNAL OF PSYCHIATRIC RESEARCH
Comorbidities provide possible path to schizophrenia subtypes
NEW YORK – Psychiatric disorders comorbid with schizophrenia often pose a diagnostic challenge, but they can be the key for improving outcomes when they are addressed along with the core schizophrenia symptoms, according to a workshop on treatment-refractory disease at the American Psychiatric Association annual meeting.
Looking for comorbidities such as depression, panic disorder, and obsessive-compulsive disorder (OCD) is important, because treatment directed at those conditions “can have a significant effect on the psychosis as well,” reported Jeffrey P. Kahn, MD, clinical associate professor of psychiatry, Cornell University, New York.
“Schizophrenia is a syndrome. It is a collection of symptoms, not a disease,” explained Michael Y. Hwang, MD, an attending physician at Veterans Affairs Hudson Valley Healthcare System, Montrose, N.Y. He believes that comorbid OCD, like comorbid panic disorder, might identify a subtype of schizophrenia that requires distinct therapeutic strategies.
The principle that schizophrenia represents a group of distinct illnesses with similar presentations is several decades old. Both Dr. Kahn and Dr. Hwang, who have published on this topic, acknowledged that efforts so far to identify those distinct diseases have failed. However, they suggested that the presence of comorbidities probably establishes clinically relevant subtypes.
Among psychiatric comorbidities, major depression is the most common, identified in more than half of patients with schizophrenia in some studies, according to Dr. Hwang. He reported that diagnostic criteria for OCD is met by about 12%, while Dr. Kahn estimated the prevalence of panic disorder in individuals with schizophrenia at about 20%.
In an individual with active positive symptoms of schizophrenia, isolating comorbid OCD or panic disorder often is a challenge. In older textbooks and guidelines on schizophrenia, comorbidities were not even discussed, Dr. Hwang said. Although the DSM-5 now acknowledges elevated rates of OCD and panic disorder in individuals with schizophrenia, Dr. Hwang and Dr. Kahn each indicated that the comorbidities still are not receiving adequate attention from clinicians.
“In general, individuals with schizophrenia who have comorbid OCD tend to do worse,” said Dr. Hwang, explaining the rationale for both looking for and treating OCD. He presented data from several studies, including his own, suggesting that symptoms of OCD can be improved with individualized pharmacotherapy. In addition to such considerations as the potential for interactions between OCD therapies and concurrent atypical antipsychotics, heterogeneity in response underlies the recommendation for individualized therapy. He added that in those with preserved executive functioning, cognitive-behavioral therapy “is always more helpful that pharmacotherapy alone.”
Comorbid panic disorder also can be difficult to recognize, particularly in individuals with active symptoms of schizophrenia, according to Dr. Kahn. . He advised against trivializing the importance of panic disorder symptoms.
“The tendency is to think, so what if the patient has symptoms of panic. Wouldn’t you if you had schizophrenia?” Dr. Kahn noted. However, identifying and then treating the panic has implications for better control of schizophrenia, he said. For the panic symptoms, there is evidence to suggest that clonazepam is effective, he said, but he advised a slow upward titration to avoid sedation and other risks that include addiction. He, like Dr. Hwang, also advised that the value of psychotherapy should not be underestimated.
“It is very important, particularly with outpatients, to spend time with that patient in order to build a therapeutic alliance and from there, progress to supportive psychotherapy,” Dr. Kahn said.
Dr. Kahn and Dr. Hwang reported no potential conflicts of interest related to this topic.
NEW YORK – Psychiatric disorders comorbid with schizophrenia often pose a diagnostic challenge, but they can be the key for improving outcomes when they are addressed along with the core schizophrenia symptoms, according to a workshop on treatment-refractory disease at the American Psychiatric Association annual meeting.
Looking for comorbidities such as depression, panic disorder, and obsessive-compulsive disorder (OCD) is important, because treatment directed at those conditions “can have a significant effect on the psychosis as well,” reported Jeffrey P. Kahn, MD, clinical associate professor of psychiatry, Cornell University, New York.
“Schizophrenia is a syndrome. It is a collection of symptoms, not a disease,” explained Michael Y. Hwang, MD, an attending physician at Veterans Affairs Hudson Valley Healthcare System, Montrose, N.Y. He believes that comorbid OCD, like comorbid panic disorder, might identify a subtype of schizophrenia that requires distinct therapeutic strategies.
The principle that schizophrenia represents a group of distinct illnesses with similar presentations is several decades old. Both Dr. Kahn and Dr. Hwang, who have published on this topic, acknowledged that efforts so far to identify those distinct diseases have failed. However, they suggested that the presence of comorbidities probably establishes clinically relevant subtypes.
Among psychiatric comorbidities, major depression is the most common, identified in more than half of patients with schizophrenia in some studies, according to Dr. Hwang. He reported that diagnostic criteria for OCD is met by about 12%, while Dr. Kahn estimated the prevalence of panic disorder in individuals with schizophrenia at about 20%.
In an individual with active positive symptoms of schizophrenia, isolating comorbid OCD or panic disorder often is a challenge. In older textbooks and guidelines on schizophrenia, comorbidities were not even discussed, Dr. Hwang said. Although the DSM-5 now acknowledges elevated rates of OCD and panic disorder in individuals with schizophrenia, Dr. Hwang and Dr. Kahn each indicated that the comorbidities still are not receiving adequate attention from clinicians.
“In general, individuals with schizophrenia who have comorbid OCD tend to do worse,” said Dr. Hwang, explaining the rationale for both looking for and treating OCD. He presented data from several studies, including his own, suggesting that symptoms of OCD can be improved with individualized pharmacotherapy. In addition to such considerations as the potential for interactions between OCD therapies and concurrent atypical antipsychotics, heterogeneity in response underlies the recommendation for individualized therapy. He added that in those with preserved executive functioning, cognitive-behavioral therapy “is always more helpful that pharmacotherapy alone.”
Comorbid panic disorder also can be difficult to recognize, particularly in individuals with active symptoms of schizophrenia, according to Dr. Kahn. . He advised against trivializing the importance of panic disorder symptoms.
“The tendency is to think, so what if the patient has symptoms of panic. Wouldn’t you if you had schizophrenia?” Dr. Kahn noted. However, identifying and then treating the panic has implications for better control of schizophrenia, he said. For the panic symptoms, there is evidence to suggest that clonazepam is effective, he said, but he advised a slow upward titration to avoid sedation and other risks that include addiction. He, like Dr. Hwang, also advised that the value of psychotherapy should not be underestimated.
“It is very important, particularly with outpatients, to spend time with that patient in order to build a therapeutic alliance and from there, progress to supportive psychotherapy,” Dr. Kahn said.
Dr. Kahn and Dr. Hwang reported no potential conflicts of interest related to this topic.
NEW YORK – Psychiatric disorders comorbid with schizophrenia often pose a diagnostic challenge, but they can be the key for improving outcomes when they are addressed along with the core schizophrenia symptoms, according to a workshop on treatment-refractory disease at the American Psychiatric Association annual meeting.
Looking for comorbidities such as depression, panic disorder, and obsessive-compulsive disorder (OCD) is important, because treatment directed at those conditions “can have a significant effect on the psychosis as well,” reported Jeffrey P. Kahn, MD, clinical associate professor of psychiatry, Cornell University, New York.
“Schizophrenia is a syndrome. It is a collection of symptoms, not a disease,” explained Michael Y. Hwang, MD, an attending physician at Veterans Affairs Hudson Valley Healthcare System, Montrose, N.Y. He believes that comorbid OCD, like comorbid panic disorder, might identify a subtype of schizophrenia that requires distinct therapeutic strategies.
The principle that schizophrenia represents a group of distinct illnesses with similar presentations is several decades old. Both Dr. Kahn and Dr. Hwang, who have published on this topic, acknowledged that efforts so far to identify those distinct diseases have failed. However, they suggested that the presence of comorbidities probably establishes clinically relevant subtypes.
Among psychiatric comorbidities, major depression is the most common, identified in more than half of patients with schizophrenia in some studies, according to Dr. Hwang. He reported that diagnostic criteria for OCD is met by about 12%, while Dr. Kahn estimated the prevalence of panic disorder in individuals with schizophrenia at about 20%.
In an individual with active positive symptoms of schizophrenia, isolating comorbid OCD or panic disorder often is a challenge. In older textbooks and guidelines on schizophrenia, comorbidities were not even discussed, Dr. Hwang said. Although the DSM-5 now acknowledges elevated rates of OCD and panic disorder in individuals with schizophrenia, Dr. Hwang and Dr. Kahn each indicated that the comorbidities still are not receiving adequate attention from clinicians.
“In general, individuals with schizophrenia who have comorbid OCD tend to do worse,” said Dr. Hwang, explaining the rationale for both looking for and treating OCD. He presented data from several studies, including his own, suggesting that symptoms of OCD can be improved with individualized pharmacotherapy. In addition to such considerations as the potential for interactions between OCD therapies and concurrent atypical antipsychotics, heterogeneity in response underlies the recommendation for individualized therapy. He added that in those with preserved executive functioning, cognitive-behavioral therapy “is always more helpful that pharmacotherapy alone.”
Comorbid panic disorder also can be difficult to recognize, particularly in individuals with active symptoms of schizophrenia, according to Dr. Kahn. . He advised against trivializing the importance of panic disorder symptoms.
“The tendency is to think, so what if the patient has symptoms of panic. Wouldn’t you if you had schizophrenia?” Dr. Kahn noted. However, identifying and then treating the panic has implications for better control of schizophrenia, he said. For the panic symptoms, there is evidence to suggest that clonazepam is effective, he said, but he advised a slow upward titration to avoid sedation and other risks that include addiction. He, like Dr. Hwang, also advised that the value of psychotherapy should not be underestimated.
“It is very important, particularly with outpatients, to spend time with that patient in order to build a therapeutic alliance and from there, progress to supportive psychotherapy,” Dr. Kahn said.
Dr. Kahn and Dr. Hwang reported no potential conflicts of interest related to this topic.
REPORTING FROM APA
Experts explore issues, controversies around medical marijuana use
I live and work in Maryland, where medical marijuana dispensaries are just beginning to open. So far, my patients have been content to smoke illegal marijuana, even after my admonishments. Last week, however, a patient who suffers from chronic pain told me that one of her doctors suggested she try medical marijuana. What did I think? The patient is in her 70s, and she has not tolerated opiates. She lives an active life, and she drives. I didn’t know what to think and was left to tell her that I had no experience and would not object if she wanted to try it. The timing was right for “Issues and Controversies Around Marijuana Use: What’s the Buzz?” at the American Psychiatric Association’s annual meeting in New York this week.
William Iacono, PhD, a professor of psychiatry at the University of Minnesota, Minneapolis, started with a session called “Does Adolescent Marijuana Use Cause Cognitive Decline?” Dr. Iacono and all the speakers who followed him pointed out how difficult it is to research these issues. The research is largely retrospective, and the questions are complex. The degree of use is determined by self-report, and there are questions about acute versus chronic use, whether cognitive decline is temporary or permanent, whether the age of initiating drug use is important, and finally, which tests are used to measure cognitive abilities. Dr. Iacono noted that results are inconsistent and mentioned a large population study done in Dunedin, New Zealand, which measured a decrease in verbal IQ and vocabulary measures at age 38 years if the user began smoking cannabis as an adolescent. Dr. Iacono’s twin studies showed that marijuana users scored lower on these measures in childhood, well before they began smoking, and poor academic performance predisposes to marijuana use.
“Adolescents who use cannabis are not the same as those who don’t,” Dr. Iacono said, “and heavy or daily use does not cause cognitive decline in those who begin smoking as adults.”
Dr. Pearlson introduced the second speaker by saying, “It’s easier to get funding to show the ill effects of cannabis than to show medicinal effects.” Sue Sisley, MD, director of Midtown Roots, a medical marijuana dispensary in Phoenix, conducts cannabis trials for the treatment of PTSD in veterans and noted that she has had a long and difficult road with marijuana research, and hers is the only controlled trial on cannabis for PTSD. When her Schedule I license was approved by the Food and Drug Administration, she was able to receive marijuana from the National Institute on Drug Abuse that was grown by the University of Mississippi in Oxford – the only federal growing facility. The marijuana was delivered by FedEx, and the drug was the consistency of talcum powder. It was a challenge to find a lab that could verify the components of the test drug, and when she did, she found the tetrahydrocannabinol content was considerably lower than marijuana sold on the black market. Also, the product contained both mold and lead. “As a physician, how do you hand out mold weed to our veterans?”
Her trials are still in progress, and more veterans are needed. Anecdotally, she says, a decrease has been seen in the use of both opiates and Viagra by the research subjects.
Michael Stevens, PhD, adjunct professor of psychiatry at Yale University, New Haven, Conn., discussed the risk of motor vehicle accidents in marijuana smokers and the logistical issues enforcement poses for law enforcement officials. “There is evidence that marijuana increases the risk for accidents.” Dr. Stevens went on to say that the elevated risk is notably less than that associated with the use of alcohol or stimulants. Studying the effects of marijuana on driving is difficult, as driving simulators do not necessarily reflect on-road experiences, and cognitive testing does not always translate into impairment. “We can’t give marijuana to teens and test them, and you can’t tell people who smoke every day that you’ll check in with them in a few years and check their driving records.”
In terms of law enforcement issues, roadside sobriety tests have not been validated for marijuana use, and plasma levels of the drug drop within minutes of use. “The alcohol model works well with alcohol, but cannabis is not alcohol.”
Deborah Hasin, PhD, professor of epidemiology (psychiatry) at Columbia University, New York, talked about trends of cannabis use in the United States. “Looking at states before and after legalization, we see that there is an increase in both cannabis use and cannabis disorders in adults.” Adolescents, however, are not smoking more, and “Kids are just not socializing; they are in their bedrooms with their smartphones. Depression is increasing in teens, but substance abuse is not.”
The last speaker was Deepak Cyril D’Souza, MD, a professor of psychiatry at Yale University, who talked about cannabis and psychosis. He defined three distinct relationships: acute transient psychosis that resolves fairly quickly, acute persistent psychosis that takes days or weeks to resolve, and psychotic reactions that are associated with recurrent psychotic symptoms. Studies suggest that those who have a psychotic reaction to marijuana are at elevated risk of being diagnosed with schizophrenia later, and that timing of exposure to marijuana may be important.
With regard to the important question of whether marijuana causes schizophrenia, Dr. D’Souza noted that “it’s neither a necessary nor sufficient component, but it does appear it hastens psychosis in schizophrenia and earlier symptoms are associated with a worse prognosis.”
I’ll see what happens with my patient. A Canadian physician in the audience noted that he has treated thousands of patients, and most find medical marijuana to be helpful. In our country, marijuana continues to be a controversial topic with strong opinions about its usefulness and a conversation that is limited by our lack of research.
Dr. Miller is coauthor of “Committed: The Battle Over Involuntary Psychiatric Care,” (Baltimore: Johns Hopkins University Press, 2016). She practices in Baltimore.
I live and work in Maryland, where medical marijuana dispensaries are just beginning to open. So far, my patients have been content to smoke illegal marijuana, even after my admonishments. Last week, however, a patient who suffers from chronic pain told me that one of her doctors suggested she try medical marijuana. What did I think? The patient is in her 70s, and she has not tolerated opiates. She lives an active life, and she drives. I didn’t know what to think and was left to tell her that I had no experience and would not object if she wanted to try it. The timing was right for “Issues and Controversies Around Marijuana Use: What’s the Buzz?” at the American Psychiatric Association’s annual meeting in New York this week.
William Iacono, PhD, a professor of psychiatry at the University of Minnesota, Minneapolis, started with a session called “Does Adolescent Marijuana Use Cause Cognitive Decline?” Dr. Iacono and all the speakers who followed him pointed out how difficult it is to research these issues. The research is largely retrospective, and the questions are complex. The degree of use is determined by self-report, and there are questions about acute versus chronic use, whether cognitive decline is temporary or permanent, whether the age of initiating drug use is important, and finally, which tests are used to measure cognitive abilities. Dr. Iacono noted that results are inconsistent and mentioned a large population study done in Dunedin, New Zealand, which measured a decrease in verbal IQ and vocabulary measures at age 38 years if the user began smoking cannabis as an adolescent. Dr. Iacono’s twin studies showed that marijuana users scored lower on these measures in childhood, well before they began smoking, and poor academic performance predisposes to marijuana use.
“Adolescents who use cannabis are not the same as those who don’t,” Dr. Iacono said, “and heavy or daily use does not cause cognitive decline in those who begin smoking as adults.”
Dr. Pearlson introduced the second speaker by saying, “It’s easier to get funding to show the ill effects of cannabis than to show medicinal effects.” Sue Sisley, MD, director of Midtown Roots, a medical marijuana dispensary in Phoenix, conducts cannabis trials for the treatment of PTSD in veterans and noted that she has had a long and difficult road with marijuana research, and hers is the only controlled trial on cannabis for PTSD. When her Schedule I license was approved by the Food and Drug Administration, she was able to receive marijuana from the National Institute on Drug Abuse that was grown by the University of Mississippi in Oxford – the only federal growing facility. The marijuana was delivered by FedEx, and the drug was the consistency of talcum powder. It was a challenge to find a lab that could verify the components of the test drug, and when she did, she found the tetrahydrocannabinol content was considerably lower than marijuana sold on the black market. Also, the product contained both mold and lead. “As a physician, how do you hand out mold weed to our veterans?”
Her trials are still in progress, and more veterans are needed. Anecdotally, she says, a decrease has been seen in the use of both opiates and Viagra by the research subjects.
Michael Stevens, PhD, adjunct professor of psychiatry at Yale University, New Haven, Conn., discussed the risk of motor vehicle accidents in marijuana smokers and the logistical issues enforcement poses for law enforcement officials. “There is evidence that marijuana increases the risk for accidents.” Dr. Stevens went on to say that the elevated risk is notably less than that associated with the use of alcohol or stimulants. Studying the effects of marijuana on driving is difficult, as driving simulators do not necessarily reflect on-road experiences, and cognitive testing does not always translate into impairment. “We can’t give marijuana to teens and test them, and you can’t tell people who smoke every day that you’ll check in with them in a few years and check their driving records.”
In terms of law enforcement issues, roadside sobriety tests have not been validated for marijuana use, and plasma levels of the drug drop within minutes of use. “The alcohol model works well with alcohol, but cannabis is not alcohol.”
Deborah Hasin, PhD, professor of epidemiology (psychiatry) at Columbia University, New York, talked about trends of cannabis use in the United States. “Looking at states before and after legalization, we see that there is an increase in both cannabis use and cannabis disorders in adults.” Adolescents, however, are not smoking more, and “Kids are just not socializing; they are in their bedrooms with their smartphones. Depression is increasing in teens, but substance abuse is not.”
The last speaker was Deepak Cyril D’Souza, MD, a professor of psychiatry at Yale University, who talked about cannabis and psychosis. He defined three distinct relationships: acute transient psychosis that resolves fairly quickly, acute persistent psychosis that takes days or weeks to resolve, and psychotic reactions that are associated with recurrent psychotic symptoms. Studies suggest that those who have a psychotic reaction to marijuana are at elevated risk of being diagnosed with schizophrenia later, and that timing of exposure to marijuana may be important.
With regard to the important question of whether marijuana causes schizophrenia, Dr. D’Souza noted that “it’s neither a necessary nor sufficient component, but it does appear it hastens psychosis in schizophrenia and earlier symptoms are associated with a worse prognosis.”
I’ll see what happens with my patient. A Canadian physician in the audience noted that he has treated thousands of patients, and most find medical marijuana to be helpful. In our country, marijuana continues to be a controversial topic with strong opinions about its usefulness and a conversation that is limited by our lack of research.
Dr. Miller is coauthor of “Committed: The Battle Over Involuntary Psychiatric Care,” (Baltimore: Johns Hopkins University Press, 2016). She practices in Baltimore.
I live and work in Maryland, where medical marijuana dispensaries are just beginning to open. So far, my patients have been content to smoke illegal marijuana, even after my admonishments. Last week, however, a patient who suffers from chronic pain told me that one of her doctors suggested she try medical marijuana. What did I think? The patient is in her 70s, and she has not tolerated opiates. She lives an active life, and she drives. I didn’t know what to think and was left to tell her that I had no experience and would not object if she wanted to try it. The timing was right for “Issues and Controversies Around Marijuana Use: What’s the Buzz?” at the American Psychiatric Association’s annual meeting in New York this week.
William Iacono, PhD, a professor of psychiatry at the University of Minnesota, Minneapolis, started with a session called “Does Adolescent Marijuana Use Cause Cognitive Decline?” Dr. Iacono and all the speakers who followed him pointed out how difficult it is to research these issues. The research is largely retrospective, and the questions are complex. The degree of use is determined by self-report, and there are questions about acute versus chronic use, whether cognitive decline is temporary or permanent, whether the age of initiating drug use is important, and finally, which tests are used to measure cognitive abilities. Dr. Iacono noted that results are inconsistent and mentioned a large population study done in Dunedin, New Zealand, which measured a decrease in verbal IQ and vocabulary measures at age 38 years if the user began smoking cannabis as an adolescent. Dr. Iacono’s twin studies showed that marijuana users scored lower on these measures in childhood, well before they began smoking, and poor academic performance predisposes to marijuana use.
“Adolescents who use cannabis are not the same as those who don’t,” Dr. Iacono said, “and heavy or daily use does not cause cognitive decline in those who begin smoking as adults.”
Dr. Pearlson introduced the second speaker by saying, “It’s easier to get funding to show the ill effects of cannabis than to show medicinal effects.” Sue Sisley, MD, director of Midtown Roots, a medical marijuana dispensary in Phoenix, conducts cannabis trials for the treatment of PTSD in veterans and noted that she has had a long and difficult road with marijuana research, and hers is the only controlled trial on cannabis for PTSD. When her Schedule I license was approved by the Food and Drug Administration, she was able to receive marijuana from the National Institute on Drug Abuse that was grown by the University of Mississippi in Oxford – the only federal growing facility. The marijuana was delivered by FedEx, and the drug was the consistency of talcum powder. It was a challenge to find a lab that could verify the components of the test drug, and when she did, she found the tetrahydrocannabinol content was considerably lower than marijuana sold on the black market. Also, the product contained both mold and lead. “As a physician, how do you hand out mold weed to our veterans?”
Her trials are still in progress, and more veterans are needed. Anecdotally, she says, a decrease has been seen in the use of both opiates and Viagra by the research subjects.
Michael Stevens, PhD, adjunct professor of psychiatry at Yale University, New Haven, Conn., discussed the risk of motor vehicle accidents in marijuana smokers and the logistical issues enforcement poses for law enforcement officials. “There is evidence that marijuana increases the risk for accidents.” Dr. Stevens went on to say that the elevated risk is notably less than that associated with the use of alcohol or stimulants. Studying the effects of marijuana on driving is difficult, as driving simulators do not necessarily reflect on-road experiences, and cognitive testing does not always translate into impairment. “We can’t give marijuana to teens and test them, and you can’t tell people who smoke every day that you’ll check in with them in a few years and check their driving records.”
In terms of law enforcement issues, roadside sobriety tests have not been validated for marijuana use, and plasma levels of the drug drop within minutes of use. “The alcohol model works well with alcohol, but cannabis is not alcohol.”
Deborah Hasin, PhD, professor of epidemiology (psychiatry) at Columbia University, New York, talked about trends of cannabis use in the United States. “Looking at states before and after legalization, we see that there is an increase in both cannabis use and cannabis disorders in adults.” Adolescents, however, are not smoking more, and “Kids are just not socializing; they are in their bedrooms with their smartphones. Depression is increasing in teens, but substance abuse is not.”
The last speaker was Deepak Cyril D’Souza, MD, a professor of psychiatry at Yale University, who talked about cannabis and psychosis. He defined three distinct relationships: acute transient psychosis that resolves fairly quickly, acute persistent psychosis that takes days or weeks to resolve, and psychotic reactions that are associated with recurrent psychotic symptoms. Studies suggest that those who have a psychotic reaction to marijuana are at elevated risk of being diagnosed with schizophrenia later, and that timing of exposure to marijuana may be important.
With regard to the important question of whether marijuana causes schizophrenia, Dr. D’Souza noted that “it’s neither a necessary nor sufficient component, but it does appear it hastens psychosis in schizophrenia and earlier symptoms are associated with a worse prognosis.”
I’ll see what happens with my patient. A Canadian physician in the audience noted that he has treated thousands of patients, and most find medical marijuana to be helpful. In our country, marijuana continues to be a controversial topic with strong opinions about its usefulness and a conversation that is limited by our lack of research.
Dr. Miller is coauthor of “Committed: The Battle Over Involuntary Psychiatric Care,” (Baltimore: Johns Hopkins University Press, 2016). She practices in Baltimore.
Off-label prescribing of SGAs in children varies by diagnosis
TORONTO – On-label prescribing of second-generation antipsychotics in hospitalized pediatric patients with bipolar disorder and schizophrenia was greater than off-label prescribing, but not for patients with autism spectrum disorder, results from a single-center study showed.
“There is an increasing trend for using second-generation antipsychotics (SGAs) in children,” lead study author Debra V. McQuade, MD, PhD, said in an interview in advance of the Pediatric Academic Societies meeting. “This trend has already been widely acknowledged as concerning, due to the relatively high adverse event burden associated with them. Also, and perhaps more concerning, the great majority of these prescriptions are prescribed as ‘off-label’ (without Food and Drug Administration approval or indication). This is true in all settings, whether outpatient or inpatient. And, this off-label use is really high – usually reported as 60%-95% of all SGA prescriptions to children. We wanted to understand more about this practice of off-label prescribing of SGAs to children – to understand what drives it, to see if there were any organizing principles that might help to better explain it.”
Dr. McQuade and her associates found that for the three diagnostic groups overall, on-label prescribing of SGAs was greater than off-label prescribing (65% vs. 35%, respectively; P less than .001). Prescriptions for patients with bipolar disorder and schizophrenia followed this pattern, with on-label and off-label prescribing rates of 73% vs. 27%, for bipolar disorder, and 85% vs. 14% for schizophrenia. However, this prescription pattern was reversed for patients with autism spectrum disorder (35% vs. 64%; P less than .001), a finding that the researchers did not expect.
She said that while off-label prescribing of SGAs to pediatric patients remains widespread and is a concerning trend, the study findings suggested that prescribers “do select and prescribe SGAs in ways that are directly or indirectly influenced by FDA guidelines, mitigating risk to their pediatric patients.” Dr. McQuade acknowledged certain weaknesses of the study, including the fact that it was limited to one hospital setting and by the small number of prescribers who contributed to the overall pool of data.
She reported having no relevant financial disclosures.
TORONTO – On-label prescribing of second-generation antipsychotics in hospitalized pediatric patients with bipolar disorder and schizophrenia was greater than off-label prescribing, but not for patients with autism spectrum disorder, results from a single-center study showed.
“There is an increasing trend for using second-generation antipsychotics (SGAs) in children,” lead study author Debra V. McQuade, MD, PhD, said in an interview in advance of the Pediatric Academic Societies meeting. “This trend has already been widely acknowledged as concerning, due to the relatively high adverse event burden associated with them. Also, and perhaps more concerning, the great majority of these prescriptions are prescribed as ‘off-label’ (without Food and Drug Administration approval or indication). This is true in all settings, whether outpatient or inpatient. And, this off-label use is really high – usually reported as 60%-95% of all SGA prescriptions to children. We wanted to understand more about this practice of off-label prescribing of SGAs to children – to understand what drives it, to see if there were any organizing principles that might help to better explain it.”
Dr. McQuade and her associates found that for the three diagnostic groups overall, on-label prescribing of SGAs was greater than off-label prescribing (65% vs. 35%, respectively; P less than .001). Prescriptions for patients with bipolar disorder and schizophrenia followed this pattern, with on-label and off-label prescribing rates of 73% vs. 27%, for bipolar disorder, and 85% vs. 14% for schizophrenia. However, this prescription pattern was reversed for patients with autism spectrum disorder (35% vs. 64%; P less than .001), a finding that the researchers did not expect.
She said that while off-label prescribing of SGAs to pediatric patients remains widespread and is a concerning trend, the study findings suggested that prescribers “do select and prescribe SGAs in ways that are directly or indirectly influenced by FDA guidelines, mitigating risk to their pediatric patients.” Dr. McQuade acknowledged certain weaknesses of the study, including the fact that it was limited to one hospital setting and by the small number of prescribers who contributed to the overall pool of data.
She reported having no relevant financial disclosures.
TORONTO – On-label prescribing of second-generation antipsychotics in hospitalized pediatric patients with bipolar disorder and schizophrenia was greater than off-label prescribing, but not for patients with autism spectrum disorder, results from a single-center study showed.
“There is an increasing trend for using second-generation antipsychotics (SGAs) in children,” lead study author Debra V. McQuade, MD, PhD, said in an interview in advance of the Pediatric Academic Societies meeting. “This trend has already been widely acknowledged as concerning, due to the relatively high adverse event burden associated with them. Also, and perhaps more concerning, the great majority of these prescriptions are prescribed as ‘off-label’ (without Food and Drug Administration approval or indication). This is true in all settings, whether outpatient or inpatient. And, this off-label use is really high – usually reported as 60%-95% of all SGA prescriptions to children. We wanted to understand more about this practice of off-label prescribing of SGAs to children – to understand what drives it, to see if there were any organizing principles that might help to better explain it.”
Dr. McQuade and her associates found that for the three diagnostic groups overall, on-label prescribing of SGAs was greater than off-label prescribing (65% vs. 35%, respectively; P less than .001). Prescriptions for patients with bipolar disorder and schizophrenia followed this pattern, with on-label and off-label prescribing rates of 73% vs. 27%, for bipolar disorder, and 85% vs. 14% for schizophrenia. However, this prescription pattern was reversed for patients with autism spectrum disorder (35% vs. 64%; P less than .001), a finding that the researchers did not expect.
She said that while off-label prescribing of SGAs to pediatric patients remains widespread and is a concerning trend, the study findings suggested that prescribers “do select and prescribe SGAs in ways that are directly or indirectly influenced by FDA guidelines, mitigating risk to their pediatric patients.” Dr. McQuade acknowledged certain weaknesses of the study, including the fact that it was limited to one hospital setting and by the small number of prescribers who contributed to the overall pool of data.
She reported having no relevant financial disclosures.
Key clinical point:
Major finding: Overall, on-label prescribing of SGAs was greater than off-label prescribing (65% vs. 35%, respectively; P less than .001), but this prescription pattern did not hold true for patients with autism spectrum disorder.
Study details: A retrospective review of 86 hospitalized pediatric inpatients with a diagnosis of autism spectrum disorder, bipolar disorder, or schizophrenia.
Disclosures: Dr. McQuade reported having no financial disclosures.
VIDEO: Research underscores murky relationship between mental illness, gun violence
NEW YORK – Legislation enacted in some states in the wake of mass shootings seeks to limit access to firearms for people with mental illness, but research presented at the annual meeting of the American Psychiatric Association raises questions about the value of that approach.
During a workshop entitled “The ‘Crazed Gunman’ Myth: Examining Mental Illness and Firearm Violence,” researchers from the Yale University in New Haven, Conn., presented new findings that support existing data calling into question whether laws considered to be “common-sense approaches” to stopping gun violence really can reduce the likelihood of mass shootings.
It appears, based on the frequency and context of firearm use in more than 400 crimes that resulted in an insanity acquittal in Connecticut, for example, that individuals with mental illness are less likely than others to misuse firearms.
In this video, workshop chair Reena Kapoor, MD, also of Yale University, discusses the findings and notes that she and her colleagues seek to move past politics and ideology to focus on science that can guide policy and legislative efforts in a potentially more effective direction.
“We’ve also found that in spite of the media narrative, there has also been a slight decrease in how often [mentally ill offenders] use guns, over the years in the study,” she said. “Although the data are preliminary, it doesn’t support this idea that mentally ill people are more dangerous than ever, that they’re using guns more often in their violence; it actually says quite the opposite.”
Dr. Kapoor reported having no disclosures.
SOURCE: Kapoor R et al. APA 2018 Workshop.
NEW YORK – Legislation enacted in some states in the wake of mass shootings seeks to limit access to firearms for people with mental illness, but research presented at the annual meeting of the American Psychiatric Association raises questions about the value of that approach.
During a workshop entitled “The ‘Crazed Gunman’ Myth: Examining Mental Illness and Firearm Violence,” researchers from the Yale University in New Haven, Conn., presented new findings that support existing data calling into question whether laws considered to be “common-sense approaches” to stopping gun violence really can reduce the likelihood of mass shootings.
It appears, based on the frequency and context of firearm use in more than 400 crimes that resulted in an insanity acquittal in Connecticut, for example, that individuals with mental illness are less likely than others to misuse firearms.
In this video, workshop chair Reena Kapoor, MD, also of Yale University, discusses the findings and notes that she and her colleagues seek to move past politics and ideology to focus on science that can guide policy and legislative efforts in a potentially more effective direction.
“We’ve also found that in spite of the media narrative, there has also been a slight decrease in how often [mentally ill offenders] use guns, over the years in the study,” she said. “Although the data are preliminary, it doesn’t support this idea that mentally ill people are more dangerous than ever, that they’re using guns more often in their violence; it actually says quite the opposite.”
Dr. Kapoor reported having no disclosures.
SOURCE: Kapoor R et al. APA 2018 Workshop.
NEW YORK – Legislation enacted in some states in the wake of mass shootings seeks to limit access to firearms for people with mental illness, but research presented at the annual meeting of the American Psychiatric Association raises questions about the value of that approach.
During a workshop entitled “The ‘Crazed Gunman’ Myth: Examining Mental Illness and Firearm Violence,” researchers from the Yale University in New Haven, Conn., presented new findings that support existing data calling into question whether laws considered to be “common-sense approaches” to stopping gun violence really can reduce the likelihood of mass shootings.
It appears, based on the frequency and context of firearm use in more than 400 crimes that resulted in an insanity acquittal in Connecticut, for example, that individuals with mental illness are less likely than others to misuse firearms.
In this video, workshop chair Reena Kapoor, MD, also of Yale University, discusses the findings and notes that she and her colleagues seek to move past politics and ideology to focus on science that can guide policy and legislative efforts in a potentially more effective direction.
“We’ve also found that in spite of the media narrative, there has also been a slight decrease in how often [mentally ill offenders] use guns, over the years in the study,” she said. “Although the data are preliminary, it doesn’t support this idea that mentally ill people are more dangerous than ever, that they’re using guns more often in their violence; it actually says quite the opposite.”
Dr. Kapoor reported having no disclosures.
SOURCE: Kapoor R et al. APA 2018 Workshop.
REPORTING FROM APA
The case for long-acting injectables
Allowing patients with a first episode to experience a second is unacceptable, Henry A. Nasrallah, MD, editor in chief of Current Psychiatry, said in his interview with Lorenzo Norris, MD, editor in chief of MDedge Psychiatry.
“It’s not too late to use injectables after the first episode, but my God, why are we letting them lose so much tissue and have so much suffering, so much PTSD, so much stigma, so much poverty, so much incarceration that comes from having repetitive psychotic episodes and deteriorating?” Dr. Nasrallah asked. “We can prevent all of that by utilizing, exploiting those agents.”
Dr. Nasrallah serves as a consultant and on the advisory boards of several companies, including Acadia, Alkermes, Allergan, Boehringer-Ingelheim, Janssen, Lundbeck, Neurocrine, Otsuka, Sunovion, and Teva. He also serves on the speakers bureaus of several companies. Dr. Norris has no disclosures.
Allowing patients with a first episode to experience a second is unacceptable, Henry A. Nasrallah, MD, editor in chief of Current Psychiatry, said in his interview with Lorenzo Norris, MD, editor in chief of MDedge Psychiatry.
“It’s not too late to use injectables after the first episode, but my God, why are we letting them lose so much tissue and have so much suffering, so much PTSD, so much stigma, so much poverty, so much incarceration that comes from having repetitive psychotic episodes and deteriorating?” Dr. Nasrallah asked. “We can prevent all of that by utilizing, exploiting those agents.”
Dr. Nasrallah serves as a consultant and on the advisory boards of several companies, including Acadia, Alkermes, Allergan, Boehringer-Ingelheim, Janssen, Lundbeck, Neurocrine, Otsuka, Sunovion, and Teva. He also serves on the speakers bureaus of several companies. Dr. Norris has no disclosures.
Allowing patients with a first episode to experience a second is unacceptable, Henry A. Nasrallah, MD, editor in chief of Current Psychiatry, said in his interview with Lorenzo Norris, MD, editor in chief of MDedge Psychiatry.
“It’s not too late to use injectables after the first episode, but my God, why are we letting them lose so much tissue and have so much suffering, so much PTSD, so much stigma, so much poverty, so much incarceration that comes from having repetitive psychotic episodes and deteriorating?” Dr. Nasrallah asked. “We can prevent all of that by utilizing, exploiting those agents.”
Dr. Nasrallah serves as a consultant and on the advisory boards of several companies, including Acadia, Alkermes, Allergan, Boehringer-Ingelheim, Janssen, Lundbeck, Neurocrine, Otsuka, Sunovion, and Teva. He also serves on the speakers bureaus of several companies. Dr. Norris has no disclosures.
Treating psychosis in patients with HIV/AIDS
Mr. S, age 56, has human immunodeficiency virus (HIV) and schizoaffective disorder. He presents to your clinic with increased auditory hallucinations, disorganized behavior, and worsened tremors that have begun to seriously disrupt his daily life. Mr. S is prescribed risperidone; however, he reports that he has not been taking it lately due to the tremor despite being controlled on his medication regimen for nearly 1 year. His Abnormal Involuntary Movement Scale (AIMS) score reveals an increased wrist rigidity compared with previous clinic visits. Mr. S has a 40 pack-year history of smoking and history of IV drug use. Furthermore, he has a medical history of type 2 diabetes mellitus, hypertension, and hyperlipidemia.
His medication regimen includes atazanavir sulfate, 200 mg/d, ritonavir, 100 mg/d, efavirenz/emtricitabine/tenofovir disoproxil fumarate, 600/200/300 mg/d, risperidone, 6 mg/d, bupropion extended-release, 300 mg/d, gabapentin, 600 mg/d, amlodipine, 5 mg/d, pravastatin, 40 mg/d, metformin, 1000 mg twice daily, and glipizide, 10 mg twice daily. Today, his laboratory findings show that his CD4 count is 405 cell/mm3, and his viral load is <40 copies/mL, indicating his HIV is well managed. A hepatitis C virus antibody test result is negative and serum creatinine level is 1.0 mg/dL. Total cholesterol is 212 mg/dL, high-density lipoprotein cholesterol is 43 mg/dL, low-density lipoprotein cholesterol is 121 mg/dL, and triglycerides level is 238 mg/dL. Electrocardiography reveals a QTc interval of 426 ms. Mr. S’s blood pressure is 105/65 mm Hg. Based on this clinic visit, the treatment team decides to change Mr. S’s antipsychotic.
Psychiatric illness and HIV/AIDS
There is a strong link between mental illness and HIV/AIDS; 50% or more of patients with HIV/AIDS have a comorbid psychiatric disorder.1 The prevalence of mental illness in patients with HIV/AIDS is reported to be 8 times higher than in those without HIV/AIDS.2 Depression, bipolar disorder, anxiety disorders, delirium, substance abuse, and schizophrenia have all been identified in persons receiving highly active antiretroviral therapy (HAART). Patients with HIV/AIDS and psychiatric illness have a decreased quality of life, poor adherence to medications, faster disease progression, and increased mortality. Care of these individuals is complicated by the stigma of HIV/AIDS and the prevalence of the illness in underserved populations, as well as the need for complex medication regimens and the possibility of drug–drug interactions (DDIs).1,2 If left untreated, psychiatric illness in patients with HIV/AIDS may lead to further transmission of HIV as a result of patients engaging in high-risk behaviors, along with poor adherence to HAART.3
Individuals diagnosed with schizophrenia, schizoaffective disorder, and bipolar disorder are at greater risk for HIV infection.3 Patients with HIV/AIDS with primary psychosis may have poor medication adherence rates due to illness-related confusion or paranoia about medications. Furthermore, they may lack the resources to manage the complications and stress related to living with HIV/AIDS.
New-onset, or secondary psychosis, has been reported in individuals with late-stage HIV/AIDS with CD4 counts <200 who have not been diagnosed with a psychotic disorder previously.3 These patients may experience more persecutory and grandiose delusions rather than hallucinations. Neuropsychiatric symptoms in patients with HIV/AIDS may be due to the presence of HIV or other infections in the CNS, tumors, or other inflammatory illnesses. Medications that have been implicated in neuropsychiatric symptoms include efavirenz, rilpivirine, and other HAART regimens; interferon; metoclopramide; corticosteroids; muscle relaxants; and clonidine. It is possible that symptoms may continue even after the medications are discontinued.3
Antipsychotics remain the treatment of choice for psychosis in HIV/AIDS, regardless of the cause of the symptoms. Many factors must be taken into consideration when choosing an antipsychotic, such as DDIs, adverse effect profiles, patient history of antipsychotic use, cost, and patient preference. Here we focus primarily on DDIs and adverse effect profiles.
When treating psychosis in patients with HIV/AIDS, it is crucial to consider potential DDIs. Many antipsychotics and antiretroviral medications utilize cytochrome P450 (CYP) enzymes for their metabolism. The CYP enzyme system is responsible for the oxidative reactions that constitute the phase I reactions necessary for the metabolism of most drugs. Inhibition and induction of CYP enzymes are among the most common causes of pharmacokinetic DDIs. Antipsychotics are predominately metabolized by CYP3A4, CYP1A2, and CYP2D6.4
Continue to: The DDIs arise because...
The DDIs arise because many antiretroviral medications inhibit, or in some cases, induce, these CYP enzymes, thereby altering substrate-drug metabolism. Inhibiting a CYP enzyme pathway can decrease substrate-drug clearance and lead to increased levels of that drug. This, in turn, can cause an increased risk of adverse effects, such as extrapyramidal symptoms (EPS) or QTc prolongation, which are both types of pharmacodynamic DDIs.4-28 However, because antipsychotics often have more than one pathway of metabolism, it can be challenging to understand the full effect of CYP-related DDIs. Furthermore, CYP enzyme inducers can decrease drug levels, and in the case of antipsychotics, lead to subtherapeutic responses.
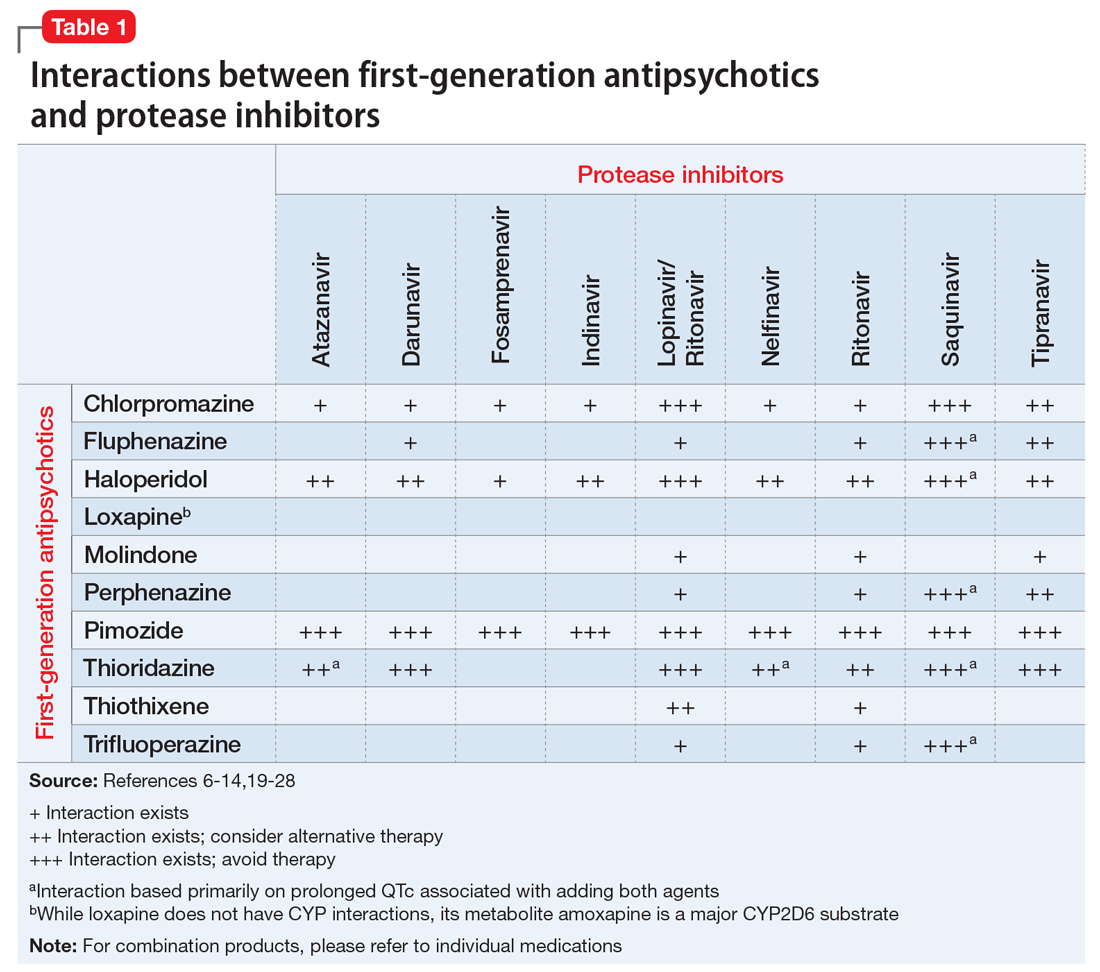
Table 1,6-14,19-28 Table 2,15-28 Table 3,6-14,19-28 and Table 415-28 list many of the known CYP enzyme-related DDIs that may occur with combination antipsychotic and antiretroviral medication therapy and aim to predict CYP induction or inhibition based on a particular combination. The following antiretroviral medications do not have any CYP-related interactions and therefore are not included in the Tables: abacavir, didanosine, emtricitabine, lamivudine, stavudine, tenofovir disoproxil, zidovudine, enfuvirtide, maraviroc, and raltegravir.
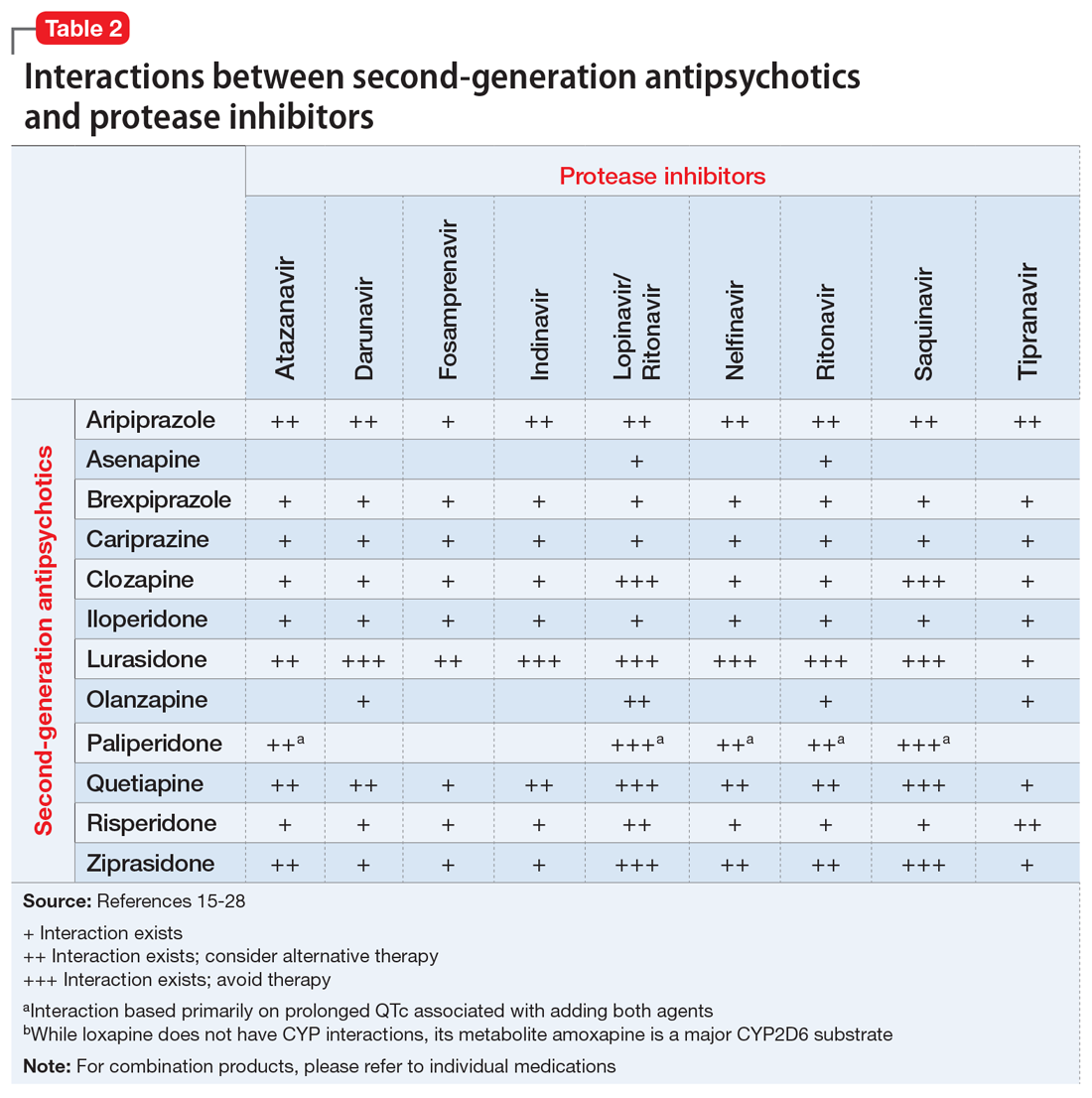
These Tables include the risk ratings for all D-rated (consider alternative therapy) and X-rated (avoid therapy) combinations. The majority of D-rated interactions are caused by CYP inhibition or induction that could potentially lead to altered antipsychotic levels. The majority of X-rated interactions are caused by increased QTc prolongation that may or may not be due to CYP-related DDIs. For example, paliperidone is not believed to be affected by the CYP enzyme system, but it does present a high risk of QTc prolongation on its own. When combined with an antiretroviral that also has a high risk of QTc prolongation, such as lopinavir, then the risk further increases.
Non-nucleoside reverse transcriptase inhibitors and protease inhibitors (PIs) are the antiretroviral medications most likely to cause DDIs with antipsychotics. Other antiretroviral classes, such as nucleoside/nucleotide reverse transcriptase inhibitors (NRTIs), fusion inhibitors, chemokine receptor 5 inhibitors, and integrase inhibitors, are not associated with CYP-related DDIs.19-28 For the most part, the severity of the CYP-related DDIs have not been well studied; therefore, most recommendations call for closer patient monitoring when combining antiretroviral medications and antipsychotics.6-18 The goal is to monitor for any changes in medication efficacy or adverse effects.
Continue to: Consider adverse effect profiles
Consider adverse effect profiles
When selecting an antipsychotic agent for a patient receiving HIV therapy, also consider adverse effect profiles. The emergence of adverse effects can greatly impact patients’ quality of life, leading to consequences of medication nonadherence and exacerbation of mental illness.
Extrapyramidal symptoms. Patients with HIV have a higher sensitivity to treatment-emergent EPS from antipsychotics.2 This sensitivity is generally thought to arise from the involvement of HIV on the basal ganglia. Historically, psychotic symptoms in HIV have been managed with second-generation antipsychotics (SGAs) at the lowest effective dose because these medications are less likely to cause EPS.1,29 The antipsychotic with the lowest rate of EPS is clozapine, followed by quetiapine, olanzapine, ziprasidone, and aripiprazole. Conversely, high-potency first-generation antipsychotics (FGAs) have the highest rates of EPS, followed by intermediate-potency FGAs and risperidone.30
Metabolic disturbances are another concern with concomitant antipsychotic/antiretroviral therapy. Patients with HIV who are receiving NRTIs or PIs can present with drug-induced lipodystrophy syndrome, which is associated with hyperglycemia, hyperinsulinemia, hyperlipidemia, and hypertension, and ultimately may cause metabolic syndrome.29 The prevalence of metabolic syndrome in patients receiving PI therapy has a vast range—2% to 84%—which can be attributed to inconsistent definitions, criteria, and assessment methodology.29 Use of a PI is considered to be the most prominent risk factor for developing lipodystrophy.29 Among the PIs, metabolic disturbances in regards to lipids are most often seen with lopinavir/ritonavir (LPV/r), saquinavir/ritonavir, tipranavir/ritonavir, and fosamprenavir/ritonavir.31 In comparison with LPV/r, darunavir showed improvement in lipids.32 Atazanavir (ATV) boosted with ritonavir has not shown clinically significant adverse effects on lipids.31 Additionally, amprenavir, LPV/r, and ritonavir demonstrated more glucose uptake inhibition via blockade of the glucose transporter type 4 than ATV.31 Of the NRTIs, lipodystrophy syndrome is most commonly seen with stavudine, which is used minimally in practice.2
The rates of metabolic disturbance with antipsychotic use range from 2% to 36%.2 The American Psychiatric Association recommends selecting one of the SGAs least likely to affect metabolic parameters.29 Aripiprazole and ziprasidone are associated with the lowest risk of weight gain, hyperglycemia, and hyperlipidemia. They are followed by risperidone and quetiapine, which are associated with moderate risk, and then clozapine and olanzapine, which are associated with high risk.2,30,33
Continue to: Management of metabolic adverse effects involves...
Management of metabolic adverse effects involves switching the antiretroviral agent and/or antipsychotic agent to an alternative associated with lower metabolic risk. Antipsychotics with low metabolic risk include aripiprazole, lurasidone, and ziprasidone. Lifestyle modifications are encouraged. Additionally, medication interventions, such as metformin, are also recommended in patients meeting criteria for pre-diabetes or type 2 diabetes mellitus.2 Lipid panels and metabolic parameters should be monitored periodically, according to guidelines.25,34
Bone marrow toxicity and blood dyscrasias. Lastly, consider the risk of bone marrow suppression. Patients receiving clozapine for treatment-resistant schizophrenia should be closely monitored for neutropenia and agranulocytosis. Although zidovudine is rarely used, its use is associated with adverse myelosuppressive effects, and the combination of clozapine and zidovudine could pose danger to the patient.2,35,36
CASE CONTINUED
Because Mr. S’s diagnosis of HIV puts him at a higher risk of developing EPS, and because he is already experiencing increased wrist rigidity, the treatment team decides to switch his antipsychotic therapy to an agent with a lower risk of EPS. His comorbidities, including type 2 diabetes mellitus, hypertension, and hyperlipidemia, are taken into account, and an SGA with a benign metabolic profile is considered. Aripiprazole and ziprasidone are favorable options. However, because efavirenz, ATZ, and ritonavir may cause QTc prolongation, ziprasidone, the SGA with the highest rate of QTc prolongation, is not the preferred option.
Mr. S’s SGA therapy is switched from risperidone to aripiprazole. Because potential CYP-related interactions between aripiprazole and Mr. S’s current antiretroviral therapy could lead to increased aripiprazole levels. Mr. S is started on a low dose (5 mg/d) with the goal to titrate based on response and tolerability. Increased levels of aripiprazole may increase the risk of akathisia, drowsiness, headaches, and fatigue. Mr. S is monitored closely for improvement of EPS, adverse effects of medication, and metabolic parameters. Furthermore, if the treatment team believes there is a more preferred antipsychotic for the patient that it did not prescribe because of the risk of DDIs, it may be worthwhile to consider discussing the HAART regimen with the patient’s infectious disease treatment team.
Continue to: Acknowledgements
Acknowledgements
This material is the result of work supported with resources and the use of facilities at the Chillicothe Veterans Affairs Medical Center in Chillicothe, Ohio. The contents of this paper do not represent the views of the U.S. Department of Veterans Affairs or the U.S. government.
Related Resources
- Cohen MA. HIV: How to provide compassionate care. Current Psychiatry. 2013;12(6):19-23,A,B.
- Khan AY, Zaidi SN. Reducing morbidity and mortality from common medical conditions in schizophrenia. Current Psychiatry. 2016;15(3):30-32,34-38,40.
Drug Brand Names
Abacavir • Ziagen
Amlodipine • Norvasc
Amprenavir • Agenerase
Aripiprazole • Abilify
Asenapine • Saphris
Atazanavir • Reyataz
Brexpiprazole • Rexulti
Bupropion ER • Wellbutrin SR
Cariprazine • Vraylar
Chlorpromazine • Thorazine
Clonidine • Catapres
Clozapine • Clozaril
Darunavir • Prezista
Delavirdine • Rescriptor
Didanosine • Videx EC
Efavirenz • Sustiva
Efavirenz/emtricitabine/tenofovir disoproxil fumarate • Atripla
Enfuvirtide • Fuzeon
Emtricitabine • Emtriva
Etravirine • Intelence
Fluphenazine • Prolixin
Fosamprenavir • Lexiva
Gabapentin • Neurontin
Glipizide • Glucotrol
Haloperidol • Haldol
Iloperidone • Fanapt
Indinavir • Crixivan
Lamivudine • Epivir
Lopinavir/ritonavir • Kaletra
Loxapine • Loxitane
Lurasidone • Latuda
Maraviroc • Selzentry
Metformin • Glucophage
Metoclopramide • Reglan
Molindone • Moban
Nelfinavir • Viracept
Nevirapine • Viramune
Olanzapine • Zyprexa
Paliperidone • Invega
Perphenazine • Trilafon
Pimozide • Orap
Pravastatin • Pravachol
Quetiapine • Seroquel
Raltegravir • Isentress
Rilpivirine • Edurant
Risperidone • Risperdal
Ritonavir • Norvir
Saquinavir • Invirase
Stavudine • Zerit
Tenofovir disoproxil • Viread
Thioridazine • Mellaril
Thiothixene • Navane
Tipranavir • Aptivus
Trifluoperazine • Stelazine
Zidovudine • Retrovir
Ziprasidone • Geodon
1. Freudenreich O, Goforth HW, Cozza KL, et al. Psychiatric treatment of persons with HIV/AIDS: An HIV-psychiatry consensus survey of current practices. Psychosomatics. 2010;51(6):480-488.
2. Hill L, Lee KC. Pharmacotherapy considerations in patients with HIV and psychiatric disorders: Focus on antidepressants and antipsychotics. Ann Pharmacother. 2013;47(1):75-89.
3. Watkins CC, Treisman GJ. Neuropsychiatric complications of aging with HIV. J Neurovirol. 2012;18(4):277-290.
4. Prior TI, Baker GB. Interactions between the cytochrome P450 system and the second-generation antipsychotics. J Psychiatry Neurosci. 2003;28(2):99-112.
5. Ponte ML, Keller GA, Di Girolamo G. Mechanisms of drug induced QT interval prolongation. Curr Drug Saf. 2010;5(1):44-53
6. Reyataz [package insert]. Princeton, NJ: Bristol-Myers Squibb Company; 2017.
7. Prezista [package insert]. Toronto, ON: Janssen Inc.; 2017.
8. Lexiva [package insert]. Research Triangle Park, NC: Viiv Healthcare; 2017
9. Crixivan [package insert]. Whitehouse Station, NJ; Merck; 2016.
10. Kaletra [package insert]. North Chicago, IL: AbbVie Inc; 2017
11. Viracept [package insert]. Kirkland, QC: Pfizer Canada Inc.; 201
12. Norvir tablets and oral solution [package insert]. North Chicago, IL: AbbVie Inc; 2017
13. Invirase [package insert]. South San Francisco, CA: Genentech USA, Inc.; 2016.
14. Aptivus [package insert]. Ridgefield, CT: Boehringer Ingelheim Pharmaceuticals Inc.; 2016.
15. Sustiva [package insert]. Princeton, NJ: Bristol-Myers Squibb Company; 2017
16. Intelence [package insert]. Titusville, NJ: Tibotec Pharmaceuticals; 2014.
17. Viramune [package insert]. Ridgefield, CT: Boehringer Ingelheim Pharmaceuticals Inc.; 2017.
18. Rescriptor [package insert]. Laval, QC: ViiV Healthcare ULC; 2013.
19. Ziagen [package insert]. Research Triangle Park, NC: GlaxoSmithKline; 2017.
20. Videx EC [package insert]. Princeton, NJ: Bristol-Myers Squibb; 2015.
21. Emtriva [package insert]. Foster City, CA: Gilead Sciences, Inc.; 2017.
22. Epivir [package insert]. Research Triangle Park, NC: ViiV Healthcare; 2017.
23. Zerit [package insert]. Princeton, NJ: Bristol-Myers Squibb; 2017.
24. Viread [package insert]. Foster City, CA: Gilead Sciences, Inc.; 2017.
25. Retrovir [package insert]. Research Triangle Park, NC: ViiV Healthcare; 2015.
26. Fuzeon [package insert]. South San Francisco, CA: Genentech USA, Inc; 2017.
27. Selzentry [package insert]. Research Triangle Park, NC: ViiV Healthcare; 2016.
28. Isentress [package insert]. Whitehouse Station, NJ: Merck Sharp & Dohme Corp.; 2017.
29. American Psychiatry Association. Practice guidelines for treatment of patients with HIV/AIDS. http://psychiatryonline.org/pb/assets/raw/sitewide/practice_guidelines/guidelines/hivaids.pdf. Published 2010. Accessed March 1, 2018.
30. Buchanan RW, Kreyenbuhl J, Kelly DL, et al; Schizophrenia Patient Outcomes Research Team (PORT). The 2009 Schizophrenia PORT psychopharmacological treatment recommendations and summary statements. Schizophr Bull. 2010;36(1):71-93.
31. Hughes PJ, Cretton-Scott E, Teague A, et al. Protease inhibitors for patients with HIV-1 infection. P T. 2011;36(6):332-336,341-345.
32. Ortiz R, Dejesus E, Khanlou H, et al. Efficacy and safety of once-daily darunavir/ritonavir versus lopinavir/ritonavir in treatment-naive HIV-1-infected patients at week 48. AIDS. 2008;22(12):1389-1397.
33. Leucht S, Cipriani A, Spineli L, et al. Comparative efficacy and tolerability of 15 antipsychotic drugs in schizophrenia: a multiple-treatments meta-analysis. Lancet. 2013;382(9896):951-962.
34. Zeier K, Connell R, Resch W, et al. Recommendations for lab monitoring of atypical antipsychotics. Current Psychiatry. 2013;12(9):51-54.
35. Singh D, Goodkin K. Choice of antipsychotic in HIV-infected patients. J Clin Psychiatry. 2007;68(3):479-480.
36. Max B, Sherer R. Management of the adverse effects of antiretroviral therapy and medication adherence. Clin Infect Dis. 2000;30(suppl 2):S96-S116.
Mr. S, age 56, has human immunodeficiency virus (HIV) and schizoaffective disorder. He presents to your clinic with increased auditory hallucinations, disorganized behavior, and worsened tremors that have begun to seriously disrupt his daily life. Mr. S is prescribed risperidone; however, he reports that he has not been taking it lately due to the tremor despite being controlled on his medication regimen for nearly 1 year. His Abnormal Involuntary Movement Scale (AIMS) score reveals an increased wrist rigidity compared with previous clinic visits. Mr. S has a 40 pack-year history of smoking and history of IV drug use. Furthermore, he has a medical history of type 2 diabetes mellitus, hypertension, and hyperlipidemia.
His medication regimen includes atazanavir sulfate, 200 mg/d, ritonavir, 100 mg/d, efavirenz/emtricitabine/tenofovir disoproxil fumarate, 600/200/300 mg/d, risperidone, 6 mg/d, bupropion extended-release, 300 mg/d, gabapentin, 600 mg/d, amlodipine, 5 mg/d, pravastatin, 40 mg/d, metformin, 1000 mg twice daily, and glipizide, 10 mg twice daily. Today, his laboratory findings show that his CD4 count is 405 cell/mm3, and his viral load is <40 copies/mL, indicating his HIV is well managed. A hepatitis C virus antibody test result is negative and serum creatinine level is 1.0 mg/dL. Total cholesterol is 212 mg/dL, high-density lipoprotein cholesterol is 43 mg/dL, low-density lipoprotein cholesterol is 121 mg/dL, and triglycerides level is 238 mg/dL. Electrocardiography reveals a QTc interval of 426 ms. Mr. S’s blood pressure is 105/65 mm Hg. Based on this clinic visit, the treatment team decides to change Mr. S’s antipsychotic.
Psychiatric illness and HIV/AIDS
There is a strong link between mental illness and HIV/AIDS; 50% or more of patients with HIV/AIDS have a comorbid psychiatric disorder.1 The prevalence of mental illness in patients with HIV/AIDS is reported to be 8 times higher than in those without HIV/AIDS.2 Depression, bipolar disorder, anxiety disorders, delirium, substance abuse, and schizophrenia have all been identified in persons receiving highly active antiretroviral therapy (HAART). Patients with HIV/AIDS and psychiatric illness have a decreased quality of life, poor adherence to medications, faster disease progression, and increased mortality. Care of these individuals is complicated by the stigma of HIV/AIDS and the prevalence of the illness in underserved populations, as well as the need for complex medication regimens and the possibility of drug–drug interactions (DDIs).1,2 If left untreated, psychiatric illness in patients with HIV/AIDS may lead to further transmission of HIV as a result of patients engaging in high-risk behaviors, along with poor adherence to HAART.3
Individuals diagnosed with schizophrenia, schizoaffective disorder, and bipolar disorder are at greater risk for HIV infection.3 Patients with HIV/AIDS with primary psychosis may have poor medication adherence rates due to illness-related confusion or paranoia about medications. Furthermore, they may lack the resources to manage the complications and stress related to living with HIV/AIDS.
New-onset, or secondary psychosis, has been reported in individuals with late-stage HIV/AIDS with CD4 counts <200 who have not been diagnosed with a psychotic disorder previously.3 These patients may experience more persecutory and grandiose delusions rather than hallucinations. Neuropsychiatric symptoms in patients with HIV/AIDS may be due to the presence of HIV or other infections in the CNS, tumors, or other inflammatory illnesses. Medications that have been implicated in neuropsychiatric symptoms include efavirenz, rilpivirine, and other HAART regimens; interferon; metoclopramide; corticosteroids; muscle relaxants; and clonidine. It is possible that symptoms may continue even after the medications are discontinued.3
Antipsychotics remain the treatment of choice for psychosis in HIV/AIDS, regardless of the cause of the symptoms. Many factors must be taken into consideration when choosing an antipsychotic, such as DDIs, adverse effect profiles, patient history of antipsychotic use, cost, and patient preference. Here we focus primarily on DDIs and adverse effect profiles.
When treating psychosis in patients with HIV/AIDS, it is crucial to consider potential DDIs. Many antipsychotics and antiretroviral medications utilize cytochrome P450 (CYP) enzymes for their metabolism. The CYP enzyme system is responsible for the oxidative reactions that constitute the phase I reactions necessary for the metabolism of most drugs. Inhibition and induction of CYP enzymes are among the most common causes of pharmacokinetic DDIs. Antipsychotics are predominately metabolized by CYP3A4, CYP1A2, and CYP2D6.4
Continue to: The DDIs arise because...
The DDIs arise because many antiretroviral medications inhibit, or in some cases, induce, these CYP enzymes, thereby altering substrate-drug metabolism. Inhibiting a CYP enzyme pathway can decrease substrate-drug clearance and lead to increased levels of that drug. This, in turn, can cause an increased risk of adverse effects, such as extrapyramidal symptoms (EPS) or QTc prolongation, which are both types of pharmacodynamic DDIs.4-28 However, because antipsychotics often have more than one pathway of metabolism, it can be challenging to understand the full effect of CYP-related DDIs. Furthermore, CYP enzyme inducers can decrease drug levels, and in the case of antipsychotics, lead to subtherapeutic responses.

Table 1,6-14,19-28 Table 2,15-28 Table 3,6-14,19-28 and Table 415-28 list many of the known CYP enzyme-related DDIs that may occur with combination antipsychotic and antiretroviral medication therapy and aim to predict CYP induction or inhibition based on a particular combination. The following antiretroviral medications do not have any CYP-related interactions and therefore are not included in the Tables: abacavir, didanosine, emtricitabine, lamivudine, stavudine, tenofovir disoproxil, zidovudine, enfuvirtide, maraviroc, and raltegravir.

These Tables include the risk ratings for all D-rated (consider alternative therapy) and X-rated (avoid therapy) combinations. The majority of D-rated interactions are caused by CYP inhibition or induction that could potentially lead to altered antipsychotic levels. The majority of X-rated interactions are caused by increased QTc prolongation that may or may not be due to CYP-related DDIs. For example, paliperidone is not believed to be affected by the CYP enzyme system, but it does present a high risk of QTc prolongation on its own. When combined with an antiretroviral that also has a high risk of QTc prolongation, such as lopinavir, then the risk further increases.
Non-nucleoside reverse transcriptase inhibitors and protease inhibitors (PIs) are the antiretroviral medications most likely to cause DDIs with antipsychotics. Other antiretroviral classes, such as nucleoside/nucleotide reverse transcriptase inhibitors (NRTIs), fusion inhibitors, chemokine receptor 5 inhibitors, and integrase inhibitors, are not associated with CYP-related DDIs.19-28 For the most part, the severity of the CYP-related DDIs have not been well studied; therefore, most recommendations call for closer patient monitoring when combining antiretroviral medications and antipsychotics.6-18 The goal is to monitor for any changes in medication efficacy or adverse effects.
Continue to: Consider adverse effect profiles
Consider adverse effect profiles
When selecting an antipsychotic agent for a patient receiving HIV therapy, also consider adverse effect profiles. The emergence of adverse effects can greatly impact patients’ quality of life, leading to consequences of medication nonadherence and exacerbation of mental illness.
Extrapyramidal symptoms. Patients with HIV have a higher sensitivity to treatment-emergent EPS from antipsychotics.2 This sensitivity is generally thought to arise from the involvement of HIV on the basal ganglia. Historically, psychotic symptoms in HIV have been managed with second-generation antipsychotics (SGAs) at the lowest effective dose because these medications are less likely to cause EPS.1,29 The antipsychotic with the lowest rate of EPS is clozapine, followed by quetiapine, olanzapine, ziprasidone, and aripiprazole. Conversely, high-potency first-generation antipsychotics (FGAs) have the highest rates of EPS, followed by intermediate-potency FGAs and risperidone.30
Metabolic disturbances are another concern with concomitant antipsychotic/antiretroviral therapy. Patients with HIV who are receiving NRTIs or PIs can present with drug-induced lipodystrophy syndrome, which is associated with hyperglycemia, hyperinsulinemia, hyperlipidemia, and hypertension, and ultimately may cause metabolic syndrome.29 The prevalence of metabolic syndrome in patients receiving PI therapy has a vast range—2% to 84%—which can be attributed to inconsistent definitions, criteria, and assessment methodology.29 Use of a PI is considered to be the most prominent risk factor for developing lipodystrophy.29 Among the PIs, metabolic disturbances in regards to lipids are most often seen with lopinavir/ritonavir (LPV/r), saquinavir/ritonavir, tipranavir/ritonavir, and fosamprenavir/ritonavir.31 In comparison with LPV/r, darunavir showed improvement in lipids.32 Atazanavir (ATV) boosted with ritonavir has not shown clinically significant adverse effects on lipids.31 Additionally, amprenavir, LPV/r, and ritonavir demonstrated more glucose uptake inhibition via blockade of the glucose transporter type 4 than ATV.31 Of the NRTIs, lipodystrophy syndrome is most commonly seen with stavudine, which is used minimally in practice.2
The rates of metabolic disturbance with antipsychotic use range from 2% to 36%.2 The American Psychiatric Association recommends selecting one of the SGAs least likely to affect metabolic parameters.29 Aripiprazole and ziprasidone are associated with the lowest risk of weight gain, hyperglycemia, and hyperlipidemia. They are followed by risperidone and quetiapine, which are associated with moderate risk, and then clozapine and olanzapine, which are associated with high risk.2,30,33
Continue to: Management of metabolic adverse effects involves...
Management of metabolic adverse effects involves switching the antiretroviral agent and/or antipsychotic agent to an alternative associated with lower metabolic risk. Antipsychotics with low metabolic risk include aripiprazole, lurasidone, and ziprasidone. Lifestyle modifications are encouraged. Additionally, medication interventions, such as metformin, are also recommended in patients meeting criteria for pre-diabetes or type 2 diabetes mellitus.2 Lipid panels and metabolic parameters should be monitored periodically, according to guidelines.25,34
Bone marrow toxicity and blood dyscrasias. Lastly, consider the risk of bone marrow suppression. Patients receiving clozapine for treatment-resistant schizophrenia should be closely monitored for neutropenia and agranulocytosis. Although zidovudine is rarely used, its use is associated with adverse myelosuppressive effects, and the combination of clozapine and zidovudine could pose danger to the patient.2,35,36
CASE CONTINUED
Because Mr. S’s diagnosis of HIV puts him at a higher risk of developing EPS, and because he is already experiencing increased wrist rigidity, the treatment team decides to switch his antipsychotic therapy to an agent with a lower risk of EPS. His comorbidities, including type 2 diabetes mellitus, hypertension, and hyperlipidemia, are taken into account, and an SGA with a benign metabolic profile is considered. Aripiprazole and ziprasidone are favorable options. However, because efavirenz, ATZ, and ritonavir may cause QTc prolongation, ziprasidone, the SGA with the highest rate of QTc prolongation, is not the preferred option.
Mr. S’s SGA therapy is switched from risperidone to aripiprazole. Because potential CYP-related interactions between aripiprazole and Mr. S’s current antiretroviral therapy could lead to increased aripiprazole levels. Mr. S is started on a low dose (5 mg/d) with the goal to titrate based on response and tolerability. Increased levels of aripiprazole may increase the risk of akathisia, drowsiness, headaches, and fatigue. Mr. S is monitored closely for improvement of EPS, adverse effects of medication, and metabolic parameters. Furthermore, if the treatment team believes there is a more preferred antipsychotic for the patient that it did not prescribe because of the risk of DDIs, it may be worthwhile to consider discussing the HAART regimen with the patient’s infectious disease treatment team.
Continue to: Acknowledgements
Acknowledgements
This material is the result of work supported with resources and the use of facilities at the Chillicothe Veterans Affairs Medical Center in Chillicothe, Ohio. The contents of this paper do not represent the views of the U.S. Department of Veterans Affairs or the U.S. government.
Related Resources
- Cohen MA. HIV: How to provide compassionate care. Current Psychiatry. 2013;12(6):19-23,A,B.
- Khan AY, Zaidi SN. Reducing morbidity and mortality from common medical conditions in schizophrenia. Current Psychiatry. 2016;15(3):30-32,34-38,40.
Drug Brand Names
Abacavir • Ziagen
Amlodipine • Norvasc
Amprenavir • Agenerase
Aripiprazole • Abilify
Asenapine • Saphris
Atazanavir • Reyataz
Brexpiprazole • Rexulti
Bupropion ER • Wellbutrin SR
Cariprazine • Vraylar
Chlorpromazine • Thorazine
Clonidine • Catapres
Clozapine • Clozaril
Darunavir • Prezista
Delavirdine • Rescriptor
Didanosine • Videx EC
Efavirenz • Sustiva
Efavirenz/emtricitabine/tenofovir disoproxil fumarate • Atripla
Enfuvirtide • Fuzeon
Emtricitabine • Emtriva
Etravirine • Intelence
Fluphenazine • Prolixin
Fosamprenavir • Lexiva
Gabapentin • Neurontin
Glipizide • Glucotrol
Haloperidol • Haldol
Iloperidone • Fanapt
Indinavir • Crixivan
Lamivudine • Epivir
Lopinavir/ritonavir • Kaletra
Loxapine • Loxitane
Lurasidone • Latuda
Maraviroc • Selzentry
Metformin • Glucophage
Metoclopramide • Reglan
Molindone • Moban
Nelfinavir • Viracept
Nevirapine • Viramune
Olanzapine • Zyprexa
Paliperidone • Invega
Perphenazine • Trilafon
Pimozide • Orap
Pravastatin • Pravachol
Quetiapine • Seroquel
Raltegravir • Isentress
Rilpivirine • Edurant
Risperidone • Risperdal
Ritonavir • Norvir
Saquinavir • Invirase
Stavudine • Zerit
Tenofovir disoproxil • Viread
Thioridazine • Mellaril
Thiothixene • Navane
Tipranavir • Aptivus
Trifluoperazine • Stelazine
Zidovudine • Retrovir
Ziprasidone • Geodon
Mr. S, age 56, has human immunodeficiency virus (HIV) and schizoaffective disorder. He presents to your clinic with increased auditory hallucinations, disorganized behavior, and worsened tremors that have begun to seriously disrupt his daily life. Mr. S is prescribed risperidone; however, he reports that he has not been taking it lately due to the tremor despite being controlled on his medication regimen for nearly 1 year. His Abnormal Involuntary Movement Scale (AIMS) score reveals an increased wrist rigidity compared with previous clinic visits. Mr. S has a 40 pack-year history of smoking and history of IV drug use. Furthermore, he has a medical history of type 2 diabetes mellitus, hypertension, and hyperlipidemia.
His medication regimen includes atazanavir sulfate, 200 mg/d, ritonavir, 100 mg/d, efavirenz/emtricitabine/tenofovir disoproxil fumarate, 600/200/300 mg/d, risperidone, 6 mg/d, bupropion extended-release, 300 mg/d, gabapentin, 600 mg/d, amlodipine, 5 mg/d, pravastatin, 40 mg/d, metformin, 1000 mg twice daily, and glipizide, 10 mg twice daily. Today, his laboratory findings show that his CD4 count is 405 cell/mm3, and his viral load is <40 copies/mL, indicating his HIV is well managed. A hepatitis C virus antibody test result is negative and serum creatinine level is 1.0 mg/dL. Total cholesterol is 212 mg/dL, high-density lipoprotein cholesterol is 43 mg/dL, low-density lipoprotein cholesterol is 121 mg/dL, and triglycerides level is 238 mg/dL. Electrocardiography reveals a QTc interval of 426 ms. Mr. S’s blood pressure is 105/65 mm Hg. Based on this clinic visit, the treatment team decides to change Mr. S’s antipsychotic.
Psychiatric illness and HIV/AIDS
There is a strong link between mental illness and HIV/AIDS; 50% or more of patients with HIV/AIDS have a comorbid psychiatric disorder.1 The prevalence of mental illness in patients with HIV/AIDS is reported to be 8 times higher than in those without HIV/AIDS.2 Depression, bipolar disorder, anxiety disorders, delirium, substance abuse, and schizophrenia have all been identified in persons receiving highly active antiretroviral therapy (HAART). Patients with HIV/AIDS and psychiatric illness have a decreased quality of life, poor adherence to medications, faster disease progression, and increased mortality. Care of these individuals is complicated by the stigma of HIV/AIDS and the prevalence of the illness in underserved populations, as well as the need for complex medication regimens and the possibility of drug–drug interactions (DDIs).1,2 If left untreated, psychiatric illness in patients with HIV/AIDS may lead to further transmission of HIV as a result of patients engaging in high-risk behaviors, along with poor adherence to HAART.3
Individuals diagnosed with schizophrenia, schizoaffective disorder, and bipolar disorder are at greater risk for HIV infection.3 Patients with HIV/AIDS with primary psychosis may have poor medication adherence rates due to illness-related confusion or paranoia about medications. Furthermore, they may lack the resources to manage the complications and stress related to living with HIV/AIDS.
New-onset, or secondary psychosis, has been reported in individuals with late-stage HIV/AIDS with CD4 counts <200 who have not been diagnosed with a psychotic disorder previously.3 These patients may experience more persecutory and grandiose delusions rather than hallucinations. Neuropsychiatric symptoms in patients with HIV/AIDS may be due to the presence of HIV or other infections in the CNS, tumors, or other inflammatory illnesses. Medications that have been implicated in neuropsychiatric symptoms include efavirenz, rilpivirine, and other HAART regimens; interferon; metoclopramide; corticosteroids; muscle relaxants; and clonidine. It is possible that symptoms may continue even after the medications are discontinued.3
Antipsychotics remain the treatment of choice for psychosis in HIV/AIDS, regardless of the cause of the symptoms. Many factors must be taken into consideration when choosing an antipsychotic, such as DDIs, adverse effect profiles, patient history of antipsychotic use, cost, and patient preference. Here we focus primarily on DDIs and adverse effect profiles.
When treating psychosis in patients with HIV/AIDS, it is crucial to consider potential DDIs. Many antipsychotics and antiretroviral medications utilize cytochrome P450 (CYP) enzymes for their metabolism. The CYP enzyme system is responsible for the oxidative reactions that constitute the phase I reactions necessary for the metabolism of most drugs. Inhibition and induction of CYP enzymes are among the most common causes of pharmacokinetic DDIs. Antipsychotics are predominately metabolized by CYP3A4, CYP1A2, and CYP2D6.4
Continue to: The DDIs arise because...
The DDIs arise because many antiretroviral medications inhibit, or in some cases, induce, these CYP enzymes, thereby altering substrate-drug metabolism. Inhibiting a CYP enzyme pathway can decrease substrate-drug clearance and lead to increased levels of that drug. This, in turn, can cause an increased risk of adverse effects, such as extrapyramidal symptoms (EPS) or QTc prolongation, which are both types of pharmacodynamic DDIs.4-28 However, because antipsychotics often have more than one pathway of metabolism, it can be challenging to understand the full effect of CYP-related DDIs. Furthermore, CYP enzyme inducers can decrease drug levels, and in the case of antipsychotics, lead to subtherapeutic responses.

Table 1,6-14,19-28 Table 2,15-28 Table 3,6-14,19-28 and Table 415-28 list many of the known CYP enzyme-related DDIs that may occur with combination antipsychotic and antiretroviral medication therapy and aim to predict CYP induction or inhibition based on a particular combination. The following antiretroviral medications do not have any CYP-related interactions and therefore are not included in the Tables: abacavir, didanosine, emtricitabine, lamivudine, stavudine, tenofovir disoproxil, zidovudine, enfuvirtide, maraviroc, and raltegravir.

These Tables include the risk ratings for all D-rated (consider alternative therapy) and X-rated (avoid therapy) combinations. The majority of D-rated interactions are caused by CYP inhibition or induction that could potentially lead to altered antipsychotic levels. The majority of X-rated interactions are caused by increased QTc prolongation that may or may not be due to CYP-related DDIs. For example, paliperidone is not believed to be affected by the CYP enzyme system, but it does present a high risk of QTc prolongation on its own. When combined with an antiretroviral that also has a high risk of QTc prolongation, such as lopinavir, then the risk further increases.
Non-nucleoside reverse transcriptase inhibitors and protease inhibitors (PIs) are the antiretroviral medications most likely to cause DDIs with antipsychotics. Other antiretroviral classes, such as nucleoside/nucleotide reverse transcriptase inhibitors (NRTIs), fusion inhibitors, chemokine receptor 5 inhibitors, and integrase inhibitors, are not associated with CYP-related DDIs.19-28 For the most part, the severity of the CYP-related DDIs have not been well studied; therefore, most recommendations call for closer patient monitoring when combining antiretroviral medications and antipsychotics.6-18 The goal is to monitor for any changes in medication efficacy or adverse effects.
Continue to: Consider adverse effect profiles
Consider adverse effect profiles
When selecting an antipsychotic agent for a patient receiving HIV therapy, also consider adverse effect profiles. The emergence of adverse effects can greatly impact patients’ quality of life, leading to consequences of medication nonadherence and exacerbation of mental illness.
Extrapyramidal symptoms. Patients with HIV have a higher sensitivity to treatment-emergent EPS from antipsychotics.2 This sensitivity is generally thought to arise from the involvement of HIV on the basal ganglia. Historically, psychotic symptoms in HIV have been managed with second-generation antipsychotics (SGAs) at the lowest effective dose because these medications are less likely to cause EPS.1,29 The antipsychotic with the lowest rate of EPS is clozapine, followed by quetiapine, olanzapine, ziprasidone, and aripiprazole. Conversely, high-potency first-generation antipsychotics (FGAs) have the highest rates of EPS, followed by intermediate-potency FGAs and risperidone.30
Metabolic disturbances are another concern with concomitant antipsychotic/antiretroviral therapy. Patients with HIV who are receiving NRTIs or PIs can present with drug-induced lipodystrophy syndrome, which is associated with hyperglycemia, hyperinsulinemia, hyperlipidemia, and hypertension, and ultimately may cause metabolic syndrome.29 The prevalence of metabolic syndrome in patients receiving PI therapy has a vast range—2% to 84%—which can be attributed to inconsistent definitions, criteria, and assessment methodology.29 Use of a PI is considered to be the most prominent risk factor for developing lipodystrophy.29 Among the PIs, metabolic disturbances in regards to lipids are most often seen with lopinavir/ritonavir (LPV/r), saquinavir/ritonavir, tipranavir/ritonavir, and fosamprenavir/ritonavir.31 In comparison with LPV/r, darunavir showed improvement in lipids.32 Atazanavir (ATV) boosted with ritonavir has not shown clinically significant adverse effects on lipids.31 Additionally, amprenavir, LPV/r, and ritonavir demonstrated more glucose uptake inhibition via blockade of the glucose transporter type 4 than ATV.31 Of the NRTIs, lipodystrophy syndrome is most commonly seen with stavudine, which is used minimally in practice.2
The rates of metabolic disturbance with antipsychotic use range from 2% to 36%.2 The American Psychiatric Association recommends selecting one of the SGAs least likely to affect metabolic parameters.29 Aripiprazole and ziprasidone are associated with the lowest risk of weight gain, hyperglycemia, and hyperlipidemia. They are followed by risperidone and quetiapine, which are associated with moderate risk, and then clozapine and olanzapine, which are associated with high risk.2,30,33
Continue to: Management of metabolic adverse effects involves...
Management of metabolic adverse effects involves switching the antiretroviral agent and/or antipsychotic agent to an alternative associated with lower metabolic risk. Antipsychotics with low metabolic risk include aripiprazole, lurasidone, and ziprasidone. Lifestyle modifications are encouraged. Additionally, medication interventions, such as metformin, are also recommended in patients meeting criteria for pre-diabetes or type 2 diabetes mellitus.2 Lipid panels and metabolic parameters should be monitored periodically, according to guidelines.25,34
Bone marrow toxicity and blood dyscrasias. Lastly, consider the risk of bone marrow suppression. Patients receiving clozapine for treatment-resistant schizophrenia should be closely monitored for neutropenia and agranulocytosis. Although zidovudine is rarely used, its use is associated with adverse myelosuppressive effects, and the combination of clozapine and zidovudine could pose danger to the patient.2,35,36
CASE CONTINUED
Because Mr. S’s diagnosis of HIV puts him at a higher risk of developing EPS, and because he is already experiencing increased wrist rigidity, the treatment team decides to switch his antipsychotic therapy to an agent with a lower risk of EPS. His comorbidities, including type 2 diabetes mellitus, hypertension, and hyperlipidemia, are taken into account, and an SGA with a benign metabolic profile is considered. Aripiprazole and ziprasidone are favorable options. However, because efavirenz, ATZ, and ritonavir may cause QTc prolongation, ziprasidone, the SGA with the highest rate of QTc prolongation, is not the preferred option.
Mr. S’s SGA therapy is switched from risperidone to aripiprazole. Because potential CYP-related interactions between aripiprazole and Mr. S’s current antiretroviral therapy could lead to increased aripiprazole levels. Mr. S is started on a low dose (5 mg/d) with the goal to titrate based on response and tolerability. Increased levels of aripiprazole may increase the risk of akathisia, drowsiness, headaches, and fatigue. Mr. S is monitored closely for improvement of EPS, adverse effects of medication, and metabolic parameters. Furthermore, if the treatment team believes there is a more preferred antipsychotic for the patient that it did not prescribe because of the risk of DDIs, it may be worthwhile to consider discussing the HAART regimen with the patient’s infectious disease treatment team.
Continue to: Acknowledgements
Acknowledgements
This material is the result of work supported with resources and the use of facilities at the Chillicothe Veterans Affairs Medical Center in Chillicothe, Ohio. The contents of this paper do not represent the views of the U.S. Department of Veterans Affairs or the U.S. government.
Related Resources
- Cohen MA. HIV: How to provide compassionate care. Current Psychiatry. 2013;12(6):19-23,A,B.
- Khan AY, Zaidi SN. Reducing morbidity and mortality from common medical conditions in schizophrenia. Current Psychiatry. 2016;15(3):30-32,34-38,40.
Drug Brand Names
Abacavir • Ziagen
Amlodipine • Norvasc
Amprenavir • Agenerase
Aripiprazole • Abilify
Asenapine • Saphris
Atazanavir • Reyataz
Brexpiprazole • Rexulti
Bupropion ER • Wellbutrin SR
Cariprazine • Vraylar
Chlorpromazine • Thorazine
Clonidine • Catapres
Clozapine • Clozaril
Darunavir • Prezista
Delavirdine • Rescriptor
Didanosine • Videx EC
Efavirenz • Sustiva
Efavirenz/emtricitabine/tenofovir disoproxil fumarate • Atripla
Enfuvirtide • Fuzeon
Emtricitabine • Emtriva
Etravirine • Intelence
Fluphenazine • Prolixin
Fosamprenavir • Lexiva
Gabapentin • Neurontin
Glipizide • Glucotrol
Haloperidol • Haldol
Iloperidone • Fanapt
Indinavir • Crixivan
Lamivudine • Epivir
Lopinavir/ritonavir • Kaletra
Loxapine • Loxitane
Lurasidone • Latuda
Maraviroc • Selzentry
Metformin • Glucophage
Metoclopramide • Reglan
Molindone • Moban
Nelfinavir • Viracept
Nevirapine • Viramune
Olanzapine • Zyprexa
Paliperidone • Invega
Perphenazine • Trilafon
Pimozide • Orap
Pravastatin • Pravachol
Quetiapine • Seroquel
Raltegravir • Isentress
Rilpivirine • Edurant
Risperidone • Risperdal
Ritonavir • Norvir
Saquinavir • Invirase
Stavudine • Zerit
Tenofovir disoproxil • Viread
Thioridazine • Mellaril
Thiothixene • Navane
Tipranavir • Aptivus
Trifluoperazine • Stelazine
Zidovudine • Retrovir
Ziprasidone • Geodon
1. Freudenreich O, Goforth HW, Cozza KL, et al. Psychiatric treatment of persons with HIV/AIDS: An HIV-psychiatry consensus survey of current practices. Psychosomatics. 2010;51(6):480-488.
2. Hill L, Lee KC. Pharmacotherapy considerations in patients with HIV and psychiatric disorders: Focus on antidepressants and antipsychotics. Ann Pharmacother. 2013;47(1):75-89.
3. Watkins CC, Treisman GJ. Neuropsychiatric complications of aging with HIV. J Neurovirol. 2012;18(4):277-290.
4. Prior TI, Baker GB. Interactions between the cytochrome P450 system and the second-generation antipsychotics. J Psychiatry Neurosci. 2003;28(2):99-112.
5. Ponte ML, Keller GA, Di Girolamo G. Mechanisms of drug induced QT interval prolongation. Curr Drug Saf. 2010;5(1):44-53
6. Reyataz [package insert]. Princeton, NJ: Bristol-Myers Squibb Company; 2017.
7. Prezista [package insert]. Toronto, ON: Janssen Inc.; 2017.
8. Lexiva [package insert]. Research Triangle Park, NC: Viiv Healthcare; 2017
9. Crixivan [package insert]. Whitehouse Station, NJ; Merck; 2016.
10. Kaletra [package insert]. North Chicago, IL: AbbVie Inc; 2017
11. Viracept [package insert]. Kirkland, QC: Pfizer Canada Inc.; 201
12. Norvir tablets and oral solution [package insert]. North Chicago, IL: AbbVie Inc; 2017
13. Invirase [package insert]. South San Francisco, CA: Genentech USA, Inc.; 2016.
14. Aptivus [package insert]. Ridgefield, CT: Boehringer Ingelheim Pharmaceuticals Inc.; 2016.
15. Sustiva [package insert]. Princeton, NJ: Bristol-Myers Squibb Company; 2017
16. Intelence [package insert]. Titusville, NJ: Tibotec Pharmaceuticals; 2014.
17. Viramune [package insert]. Ridgefield, CT: Boehringer Ingelheim Pharmaceuticals Inc.; 2017.
18. Rescriptor [package insert]. Laval, QC: ViiV Healthcare ULC; 2013.
19. Ziagen [package insert]. Research Triangle Park, NC: GlaxoSmithKline; 2017.
20. Videx EC [package insert]. Princeton, NJ: Bristol-Myers Squibb; 2015.
21. Emtriva [package insert]. Foster City, CA: Gilead Sciences, Inc.; 2017.
22. Epivir [package insert]. Research Triangle Park, NC: ViiV Healthcare; 2017.
23. Zerit [package insert]. Princeton, NJ: Bristol-Myers Squibb; 2017.
24. Viread [package insert]. Foster City, CA: Gilead Sciences, Inc.; 2017.
25. Retrovir [package insert]. Research Triangle Park, NC: ViiV Healthcare; 2015.
26. Fuzeon [package insert]. South San Francisco, CA: Genentech USA, Inc; 2017.
27. Selzentry [package insert]. Research Triangle Park, NC: ViiV Healthcare; 2016.
28. Isentress [package insert]. Whitehouse Station, NJ: Merck Sharp & Dohme Corp.; 2017.
29. American Psychiatry Association. Practice guidelines for treatment of patients with HIV/AIDS. http://psychiatryonline.org/pb/assets/raw/sitewide/practice_guidelines/guidelines/hivaids.pdf. Published 2010. Accessed March 1, 2018.
30. Buchanan RW, Kreyenbuhl J, Kelly DL, et al; Schizophrenia Patient Outcomes Research Team (PORT). The 2009 Schizophrenia PORT psychopharmacological treatment recommendations and summary statements. Schizophr Bull. 2010;36(1):71-93.
31. Hughes PJ, Cretton-Scott E, Teague A, et al. Protease inhibitors for patients with HIV-1 infection. P T. 2011;36(6):332-336,341-345.
32. Ortiz R, Dejesus E, Khanlou H, et al. Efficacy and safety of once-daily darunavir/ritonavir versus lopinavir/ritonavir in treatment-naive HIV-1-infected patients at week 48. AIDS. 2008;22(12):1389-1397.
33. Leucht S, Cipriani A, Spineli L, et al. Comparative efficacy and tolerability of 15 antipsychotic drugs in schizophrenia: a multiple-treatments meta-analysis. Lancet. 2013;382(9896):951-962.
34. Zeier K, Connell R, Resch W, et al. Recommendations for lab monitoring of atypical antipsychotics. Current Psychiatry. 2013;12(9):51-54.
35. Singh D, Goodkin K. Choice of antipsychotic in HIV-infected patients. J Clin Psychiatry. 2007;68(3):479-480.
36. Max B, Sherer R. Management of the adverse effects of antiretroviral therapy and medication adherence. Clin Infect Dis. 2000;30(suppl 2):S96-S116.
1. Freudenreich O, Goforth HW, Cozza KL, et al. Psychiatric treatment of persons with HIV/AIDS: An HIV-psychiatry consensus survey of current practices. Psychosomatics. 2010;51(6):480-488.
2. Hill L, Lee KC. Pharmacotherapy considerations in patients with HIV and psychiatric disorders: Focus on antidepressants and antipsychotics. Ann Pharmacother. 2013;47(1):75-89.
3. Watkins CC, Treisman GJ. Neuropsychiatric complications of aging with HIV. J Neurovirol. 2012;18(4):277-290.
4. Prior TI, Baker GB. Interactions between the cytochrome P450 system and the second-generation antipsychotics. J Psychiatry Neurosci. 2003;28(2):99-112.
5. Ponte ML, Keller GA, Di Girolamo G. Mechanisms of drug induced QT interval prolongation. Curr Drug Saf. 2010;5(1):44-53
6. Reyataz [package insert]. Princeton, NJ: Bristol-Myers Squibb Company; 2017.
7. Prezista [package insert]. Toronto, ON: Janssen Inc.; 2017.
8. Lexiva [package insert]. Research Triangle Park, NC: Viiv Healthcare; 2017
9. Crixivan [package insert]. Whitehouse Station, NJ; Merck; 2016.
10. Kaletra [package insert]. North Chicago, IL: AbbVie Inc; 2017
11. Viracept [package insert]. Kirkland, QC: Pfizer Canada Inc.; 201
12. Norvir tablets and oral solution [package insert]. North Chicago, IL: AbbVie Inc; 2017
13. Invirase [package insert]. South San Francisco, CA: Genentech USA, Inc.; 2016.
14. Aptivus [package insert]. Ridgefield, CT: Boehringer Ingelheim Pharmaceuticals Inc.; 2016.
15. Sustiva [package insert]. Princeton, NJ: Bristol-Myers Squibb Company; 2017
16. Intelence [package insert]. Titusville, NJ: Tibotec Pharmaceuticals; 2014.
17. Viramune [package insert]. Ridgefield, CT: Boehringer Ingelheim Pharmaceuticals Inc.; 2017.
18. Rescriptor [package insert]. Laval, QC: ViiV Healthcare ULC; 2013.
19. Ziagen [package insert]. Research Triangle Park, NC: GlaxoSmithKline; 2017.
20. Videx EC [package insert]. Princeton, NJ: Bristol-Myers Squibb; 2015.
21. Emtriva [package insert]. Foster City, CA: Gilead Sciences, Inc.; 2017.
22. Epivir [package insert]. Research Triangle Park, NC: ViiV Healthcare; 2017.
23. Zerit [package insert]. Princeton, NJ: Bristol-Myers Squibb; 2017.
24. Viread [package insert]. Foster City, CA: Gilead Sciences, Inc.; 2017.
25. Retrovir [package insert]. Research Triangle Park, NC: ViiV Healthcare; 2015.
26. Fuzeon [package insert]. South San Francisco, CA: Genentech USA, Inc; 2017.
27. Selzentry [package insert]. Research Triangle Park, NC: ViiV Healthcare; 2016.
28. Isentress [package insert]. Whitehouse Station, NJ: Merck Sharp & Dohme Corp.; 2017.
29. American Psychiatry Association. Practice guidelines for treatment of patients with HIV/AIDS. http://psychiatryonline.org/pb/assets/raw/sitewide/practice_guidelines/guidelines/hivaids.pdf. Published 2010. Accessed March 1, 2018.
30. Buchanan RW, Kreyenbuhl J, Kelly DL, et al; Schizophrenia Patient Outcomes Research Team (PORT). The 2009 Schizophrenia PORT psychopharmacological treatment recommendations and summary statements. Schizophr Bull. 2010;36(1):71-93.
31. Hughes PJ, Cretton-Scott E, Teague A, et al. Protease inhibitors for patients with HIV-1 infection. P T. 2011;36(6):332-336,341-345.
32. Ortiz R, Dejesus E, Khanlou H, et al. Efficacy and safety of once-daily darunavir/ritonavir versus lopinavir/ritonavir in treatment-naive HIV-1-infected patients at week 48. AIDS. 2008;22(12):1389-1397.
33. Leucht S, Cipriani A, Spineli L, et al. Comparative efficacy and tolerability of 15 antipsychotic drugs in schizophrenia: a multiple-treatments meta-analysis. Lancet. 2013;382(9896):951-962.
34. Zeier K, Connell R, Resch W, et al. Recommendations for lab monitoring of atypical antipsychotics. Current Psychiatry. 2013;12(9):51-54.
35. Singh D, Goodkin K. Choice of antipsychotic in HIV-infected patients. J Clin Psychiatry. 2007;68(3):479-480.
36. Max B, Sherer R. Management of the adverse effects of antiretroviral therapy and medication adherence. Clin Infect Dis. 2000;30(suppl 2):S96-S116.