User login
Postpartum psychosis: Protecting mother and infant
A new mother drowned her 6-month-old daughter in the bathtub. The married woman, who had a history of schizoaffective disorder, had been high functioning and worked in a managerial role prior to giving birth. However, within a day of delivery, her mental state deteriorated. She quickly became convinced that her daughter had a genetic disorder such as achondroplasia. Physical examinations, genetic testing, and x-rays all failed to alleviate her concerns. Examination of her computer revealed thousands of searches for various medical conditions and surgical treatments. After the baby’s death, the mother was admitted to a psychiatric hospital. She eventually pled guilty to manslaughter.1
Mothers with postpartum psychosis (PPP) typically present fulminantly within days to weeks of giving birth. Symptoms of PPP may include not only psychosis, but also confusion and dysphoric mania. These symptoms often wax and wane, which can make it challenging to establish the diagnosis. In addition, many mothers hide their symptoms due to poor insight, delusions, or fear of loss of custody of their infant. In the vast majority of cases, psychiatric hospitalization is required to protect both mother and baby; untreated, there is an elevated risk of both maternal suicide and infanticide. This article discusses the presentation of PPP, its differential diagnosis, risk factors for developing PPP, suicide and infanticide risk assessment, treatment (including during breastfeeding), and prevention.
The bipolar connection
While multiple factors may increase the risk of PPP (Table 12), women with bipolar disorder have a particularly elevated risk. After experiencing incipient postpartum affective psychosis, a woman has a 50% to 80% chance of having another psychiatric episode, usually within the bipolar spectrum.2 Of all women with PPP, 70% to 90% have bipolar illness or schizoaffective disorder, while approximately 12% have schizophrenia.3,4Women with bipolar disorder are more likely to experience a postpartum psychiatric admission than mothers with any other psychiatric diagnosis5 and have an increased risk of PPP by a factor of 100 over the general population.2
For women with bipolar disorder, PPP should be understood as a recurrence of the chronic disease. Recent evidence does suggest, however, that a significant minority of women progress to experience mood and psychotic symptoms only in the postpartum period.6,7 It is hypothesized that this subgroup of women has a biologic vulnerability to affective psychosis that is limited to the postpartum period. Clinically, understanding a woman’s disease course is important because it may guide decision-making about prophylactic medications during or after pregnancy.
A rapid, delirium-like presentation
Postpartum psychosis is a rare disorder, with a prevalence of 1 to 2 cases per 1,000 childbirths.3 While symptoms may begin days to weeks postpartum, the typical time of onset is between 3 to 10 days after birth, occurring after a woman has been discharged from the hospital and during a time of change and uncertainty. This can make the presentation of PPP a confusing and distressing experience for both the new mother and the family, resulting in delays in seeking care.
Subtle prodromal symptoms may include insomnia, mood fluctuation, and irritability. As symptoms progress, PPP is notable for a rapid onset and a delirium-like appearance that may include waxing and waning cognitive symptoms such as disorientation and confusion.8 Grossly disorganized behaviors and rapid mood fluctuations are typical. Distinct from mood episodes outside the peripartum period, women with PPP often experience mood-incongruent delusions and obsessive thoughts, often focused on their child.9 Women with PPP appear less likely to experience thought insertion or withdrawal or auditory hallucinations that give a running commentary.2
Differential diagnosis includes depression, OCD
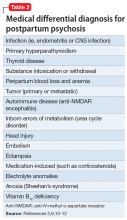
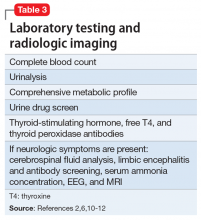
When evaluating a woman with possible postpartum psychotic symptoms or delirium, it is important to include a thorough history, physical examination, and relevant laboratory and/or imaging investigations to assess for organic causes or contributors (Table 22,6,10-12 and Table 32,6,10-12). A detailed psychiatric history should establish whether the patient is presenting with new-onset psychosis or has had previous mood or psychotic episodes that may have gone undetected. Important perinatal psychiatric differential diagnoses should include “baby blues,” postpartum depression (PPD), and obsessive-compulsive disorder (OCD).
Continue to: PPP vs "baby blues."
PPP vs “baby blues.” “Baby blues” is not an official DSM-5 diagnosis but rather a normative postpartum experience that affects 50% to 80% of postpartum women. A woman with the “baby blues” may feel weepy or have mild mood lability, irritability, or anxiety; however, these symptoms do not significantly impair function. Peak symptoms typically occur between 2 to 5 days postpartum and generally resolve within 2 weeks. Women who have the “baby blues” are at an increased risk for PPD and should be monitored over time.13,14
PPP vs PPD. Postpartum depression affects approximately 10% to 15% of new mothers.15 Women with PPD may experience feelings of persistent and severe sadness, feelings of detachment, insomnia, and fatigue. Symptoms of PPD can interfere with a mother’s interest in caring for her baby and present a barrier to maternal bonding.16,17
As the awareness of PPD has increased in recent years, screening for depressive symptoms during and after pregnancy has increasingly become the standard of care.18 When evaluating a postpartum woman for PPD, it is important to consider PPP in the differential. Women with severe or persistent depressive symptoms may also develop psychotic symptoms. Furthermore, suicidal thoughts or thoughts of harming the infant may be present in either PPD or PPP. One study found that 41% of mothers with depression endorsed thoughts of harming their infants.19
PPP vs postpartum OCD. Postpartum obsessive-compulsive symptoms commonly occur comorbidly with PPD,9 and OCD often presents for the first time in the postpartum period.20 Obsessive-compulsive disorder affects between 2% to 9% of new mothers.21,22 It is critical to properly differentiate PPP from postpartum OCD. Clinical questions should be posed with a non-judgmental stance. Just as delusions in PPP are often focused on the infant, for women with OCD, obsessive thoughts may center on worries about the infant’s safety. Distressing obsessions about violence are common in OCD.23 Mothers with OCD may experience intrusive thinking about accidentally or purposefully harming their infant. For example, they may intrusively worry that they will accidentally put the baby in the microwave or oven, leave the baby in a hot car, or throw the baby down the stairs. However, a postpartum woman with OCD may be reluctant to share her ego-dystonic thoughts of infant harm. Mothers with OCD are not out of touch with reality; instead, their intrusive thoughts are ego-dystonic and distressing. These are thoughts and fears that they focus on and try to avoid, rather than plan. The psychiatrist must carefully differentiate between ego-syntonic and ego-dystonic thoughts. These patients often avoid seeking treatment because of their shame and guilt.23 Clinicians often under-recognize OCD and risk inappropriate hospitalization, treatment, and inappropriate referral to Child Protective Services (CPS).23
Perinatal psychiatric risk assessment
When a mother develops PPP, consider the risks of suicide, child harm, and infanticide. Although suicide risk is generally lower in the postpartum period, suicide is the cause of 20% of postpartum deaths.24,25 When PPP is untreated, suicide risk is elevated. A careful suicide risk assessment should be completed.
Continue to: Particularly in PPP...
Particularly in PPP, a mother may be at risk of child neglect or abuse due to her confused or delusional thinking and mood state.26 For example, one mother heated empty bottles and gave them to her baby, and then became frustrated when the baby continued to cry.
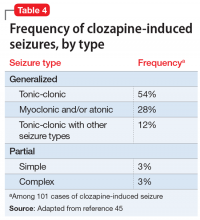
The risk of infanticide is also elevated in untreated PPP, with approximately 4% of these women committing infanticide.9 There are 5 motives for infanticide (Table 427). Altruistic and acutely psychotic motives are more likely to be related to PPP, while fatal maltreatment, unwanted child, and partner revenge motives are less likely to be related to PPP. Among mothers who kill both their child and themselves (filicide-suicide), altruistic motives were the most common.28 Mothers in psychiatric samples who kill their children have often experienced psychosis, suicidality, depression, and significant life stresses.27 Both infanticidal ideas and behaviors have been associated with psychotic thinking about the infant,29 so it is critical to ascertain whether the mother’s delusions or hallucinations involve the infant.30 In contrast, neonaticide (murder in the first day of life) is rarely related to PPP because PPP typically has a later onset.31

Treating acute PPP
The fulminant nature of PPP can make its treatment difficult. Thinking through the case in an organized fashion is critical (Table 5).

Hospitalization. Postpartum psychosis is a psychiatric emergency with a rapid onset of symptoms. Hospitalization is required in almost all cases for diagnostic evaluation, assessment and management of safety, and initiation of treatment. While maternal-infant bonding in the perinatal period is important, infant safety is critical and usually requires maternal psychiatric hospitalization.
The specialized mother-baby psychiatric unit (MBU) is a model of care first developed in the United Kingdom and is now available in many European countries as well as in New Zealand and Australia. Mother-baby psychiatric units admit the mother and the baby together and provide dyadic treatment to allow for enhanced bonding and parenting support, and often to encourage breastfeeding.30 In the United States, there has been growing interest in specialized inpatient settings that acknowledge the importance of maternal-infant attachment in the treatment of perinatal disorders and provide care with a dyadic focus; however, differences in the health care payer system have been a barrier to full-scale MBUs. The Perinatal Psychiatry Inpatient Unit at University of North Carolina-Chapel Hill is among the first of such a model in the United States.32
Continue to: Although this specialized treatment setting...
Although this specialized treatment setting is unlikely to be available in most American cities, treatment should still consider the maternal role. When possible, the infant should stay with the father or family members during the mother’s hospitalization, and supervised visits should be arranged when appropriate. If the mother is breastfeeding, or plans to breastfeed after the hospitalization, the treatment team may consider providing supervised use of a breast pump and making arrangements for breast milk storage. During the mother’s hospitalization, staff should provide psychoeducation and convey hopefulness and support.
Medication management. Mood stabilizers and second-generation antipsychotics (SGAs) are often used for acute management of PPP. The choice of medication is determined by individual symptoms, severity of presentation, previous response to medication, and maternal adverse effects.30 In a naturalistic study of 64 women admitted for new-onset PPP, sequential administration of benzodiazepines, antipsychotics, and lithium was found to be effective in achieving remission for 99% of patients, with 80% sustaining remission at 9 months postpartum.6 Second-generation antipsychotics such as
Breastfeeding. It is important to discuss breastfeeding with the mother and her partner or family. The patient’s preference, the maternal and infant benefits of breastfeeding, the potential for sleep disruption, and the safety profile of needed medications should all be considered. Because sleep loss is a modifiable risk factor in PPP, the benefits of breastfeeding may be outweighed by the risks for some patients.9 For others, breastfeeding during the day and bottle-feeding at night may be preferred.
What to consider during discharge planning
Discharge arrangements require careful consideration (Table 6). Meet with the family prior to discharge to provide psychoeducation and to underscore the importance of family involvement with both mother and infant. It is important to ensure adequate support at home, including at night, since sleep is critical to improved stability. Encourage the patient and her family to monitor for early warning signs of relapse, which might include refractory insomnia, mood instability, poor judgment, or hypomanic symptoms.35 She should be followed closely as an outpatient. Having her partner (or another close family member) and infant present during appointments can help in obtaining collateral information and assessing mother-infant bonding. The clinician should also consider whether it is necessary to contact CPS. Many mothers with mental illness appropriately parent their child, but CPS should be alerted when there is a reasonable concern about safe parenting—abuse, neglect, or significant risk.36

Take steps for prevention
An important part of managing PPP is prevention. This involves providing preconception counseling to the woman and her partner.30 Preconception advice should be individualized and include discussion of:
- risks of relapse in pregnancy and the postpartum period
- optimal physical and mental health
- potential risks and benefits of medication options in pregnancy
- potential effects of untreated illness for the fetus, infant, and family
- a strategy outlining whether medication is continued in pregnancy or started in the postpartum period.
Continue to: For women at risk of PPP...
For women at risk of PPP, the risks of medications need to be balanced with the risks of untreated illness. To reduce the risk of PPP relapse, guidelines recommend a robust antenatal care plan that should include37,38:
- close monitoring of a woman’s mental state for early warning signs of PPP, with active participation from the woman’s partner and family
- ongoing discussion of the risks and benefits of pharmacotherapy (and, for women who prefer to not take medication in the first trimester, a plan for when medications will be restarted)
- collaboration with other professionals involved in care during pregnancy and postpartum (eg, obstetricians, midwives, family practitioners, pediatricians)
- planning to minimize risk factors associated with relapse (eg, sleep deprivation, lack of social supports, domestic violence, and substance abuse).
Evidence clearly suggests that women with bipolar disorder are at increased risk for illness recurrence without continued maintenance medication.39 A subgroup of women with PPP go on to have psychosis limited to the postpartum period, and reinstating prophylactic medication in late pregnancy (preferably) or immediately after birth should be discussed.2 The choice of prophylactic medication should be determined by the woman’s previous response.
Regarding prophylaxis, the most evidence exists for lithium.6 Lithium use during the first trimester carries a risk of Ebstein’s anomaly. However, a recent systematic review and meta-analysis have concluded that the teratogenic risks of lithium have been overestimated.40,41
Lamotrigine is an alternative mood stabilizer with a favorable safety profile in pregnancy. In a small naturalistic study in which lamotrigine was continued in pregnancy in women with bipolar disorder, the medication was effective in preventing relapse in pregnancy and postpartum.42 A small population-based cohort study found lamotrigine was as effective as lithium in preventing severe postpartum relapse in women with bipolar disorder,43 although this study was limited by its observational design. Recently published studies have found no significant association between
Box
It is essential to consider the patient’s individual symptoms and treatment history when making pharmacologic recommendations during pregnancy. Discussion with the patient about the risks and benefits of lithium is recommended. For women who continue to use lithium during pregnancy, ongoing pharmacokinetic changes warrant more frequent monitoring (some experts advise monthly monitoring throughout pregnancy, moving to more frequent monitoring at 36 weeks).47 During labor, the team might consider temporary cessation of lithium and particular attention to hydration status.30 In the postpartum period, there is a quick return to baseline glomerular filtration rate and a rapid decrease in vascular volume, so it is advisable to restart the patient at her pre-pregnancy lithium dosage. It is recommended to check lithium levels within 24 hours of delivery.47 While lithium is not an absolute contraindication to breastfeeding, there is particular concern in situations of prematurity or neonatal dehydration. Collaboration with and close monitoring by the pediatrician is essential to determine an infant monitoring plan.48
If lamotrigine is used during pregnancy, be aware that pregnancy-related pharmacokinetic changes result in increased lamotrigine clearance, which will vary in magnitude among individuals. Faster clearance may necessitate dose increases during pregnancy and a taper back to pre-pregnancy dose in the postpartum period. Dosing should always take clinical symptoms into account.
Pharmacotherapy can reduce relapse risk
To prevent relapse in the postpartum period, consider initiating treatment with mood stabilizers and/or SGAs, particularly for women with bipolar disorder who do not take medication during pregnancy. A recent meta-analysis found a high postpartum relapse rate (66%) in women with bipolar disorder who did not take prophylactic medication, compared with a relapse rate of 23% for women who did take such medication. In women with psychosis limited to the postpartum period, prophylaxis with lithium or antipsychotics in the immediate postpartum can prevent relapse.39 The SGAs olanzapine and quetiapine are often used to manage acute symptoms because they are considered acceptable during breastfeeding.33 The use of lithium when breastfeeding is complex to manage48 and may require advice to not breastfeed, which can be an important consideration for patients and their families.
Bottom Line
Postpartum psychosis (PPP) typically presents with a rapid onset of hallucinations, delusions, confusion, and mood swings within days to weeks of giving birth. Mothers with PPP almost always require hospitalization for the safety of their infants and themselves. Mood stabilizers and second-generation antipsychotics are used for acute management.
Related Resources
- Clark CT, Wisner KL. Treatment of peripartum bipolar disorder. Obstet Gynecol Clin N Am. 2018;45:403-417.
- Massachusetts General Hospital Center for Women’s Mental Health. https://womensmentalhealth.org/. 2018.
- Postpartum Support International. Postpartum psychosis. http://www.postpartum.net/learn-more/postpartumpsychosis/. 2019.
Drug Brand Names
Bromocriptine • Cycloset, Parlodel
Cabergoline • Dostinex
Lamotrigine • Lamictal
Lithium • Eskalith, Lithobid
Olanzapine • Zyprexa
Quetiapine • Seroquel
1. Hall L. Mother who killed baby believing she was a dwarf should not be jailed, court told. The Sydney Morning Herald. https://www.smh.com.au/national/nsw/mother-who-killed-baby-believing-she-was-a-dwarf-should-not-be-jailed-court-told-20170428-gvud4d.html. Published April 28, 2017. Accessed March 12, 2019.
2. Bergink V, Rasgon N, Wisner KL. Postpartum psychosis: madness, mania, and melancholia in motherhood. Am J Psychiatry. 2016;173(12):1179-1188.
3. Sit D, Rothschild AJ, Wisner KL. A review of postpartum psychosis. J Womens Health (Larchmt). 2006;15(4):352-368.
4. Kendell RE, Chalmers JC, Platz C. Epidemiology of puerperal psychoses. Br J Psychiatry. 1987;150(5):662-673.
5. Munk-Olsen T, Laursen TM, Mendelson T, et al. Risks and predictors of readmission for a mental disorder during the postpartum period. Arch Gen Psychiatry. 2009;66(2):189-195.
6. Bergink V, Burgerhout KM, Koorengevel KM, et al. Treatment of psychosis and mania in the postpartum period. Am J Psychiatry. 2015;172(2):115-123.
7. Wesseloo R, Kamperman AM, Munk-Olsen T, et al. Risk of postpartum relapse in bipolar disorder and postpartum psychosis: a systematic review and meta-analysis. Am J Psychiatry. 2015;173(2):117-127.
8. Wisner KL, Peindl K, Hanusa BH. Symptomatology of affective and psychotic illnesses related to childbearing. J Affect Disord. 1994;30(2):77-87.
9. Spinelli MG. Postpartum psychosis: detection of risk and management. Am J Psychiatry. 2009;166(4):405-408.
10. Fassier T, Guffon N, Acquaviva C, et al. Misdiagnosed postpartum psychosis revealing a late-onset urea cycle disorder. Am J Psychiatry. 2011;168(6):576-580.
11. Yu AYX, Moore FG. Paraneoplastic encephalitis presenting as postpartum psychosis. Psychosomatics. 2011;52(6):568-570.
12. Patil NJ, Yadav SS, Gokhale YA, et al. Primary hypoparathyroidism: psychosis in postpartum period. J Assoc Physicians India. 2010;58:506-508.
13. O’Hara MW, Schlechte JA, Lewis DA, et al. Prospective study of postpartum blues: biologic and psychosocial factors. Arch Gen Psychiatry. 1991;48(9):801-806.
14. Burt VK, Hendrick VC. Clinical manual of women’s mental health. Washington, DC. American Psychiatric Association Publishing; 2007:79-80.
15. Melzer-Brody S. Postpartum depression: what to tell patients who breast-feed. Current Psychiatry. 2008;7(5):87-95.
16. Alhusen JL, Gross D, Hayat MJ, et al. The role of mental health on maternal‐fetal attachment in low‐income women. J Obstet Gynecol Neonatal Nurs. 2012;41(6):E71-E81.
17. McLearn KT, Minkovitz CS, Strobino DM, et al. Maternal depressive symptoms at 2 to 4 months postpartum and early parenting practices. Arch Pediatr Adolesc Med. 2006;160(3):279-284.
18. Committee on Obstetric Practice. The American College of Obstetricians and Gynecologists Committee Opinion no. 630. Screening for perinatal depression. Obstet Gynecol. 2015;125(5):1268-1271.
19. Jennings KD, Ross S, Popper S. Thoughts of harming infants in depressed and nondepressed mothers. J Affect Disord. 1999;54(1-2):21-28.
20. Miller ES, Hoxha D, Wisner KL, et al. Obsessions and compulsions in postpartum women without obsessive compulsive disorder. J Womens Health. 2015;24(10):825-830.
21. Russell EJ, Fawcett JM, Mazmanian D. Risk of obsessive-compulsive disorder in pregnant and postpartum women: a meta-analysis. J Clin Psychiatry. 2013;74(4):377-385.
22. Zambaldi CF, Cantilino A, Montenegro AC, et al. Postpartum obsessive-compulsive disorder: prevalence and clinical characteristics. Compr Psychiatry. 2009;50(6):503-509.
23. Booth BD, Friedman SH, Curry S, et al. Obsessions of child murder: underrecognized manifestations of obsessive-compulsive disorder. J Am Acad Psychiatry Law. 2014;42(1):66-74.
24. Lindahl V, Pearson JL, Colpe L. Prevalence of suicidality during pregnancy and the postpartum. Arch Womens Ment Health. 2005;8(2):77-87.
25. Samandari G, Martin SL, Kupper LL, et al. Are pregnant and postpartum women: at increased risk for violent death? Suicide and homicide findings from North Carolina. Matern Child Health J. 2011;15(5):660-669.
26. Friedman SH, Sorrentino R. Commentary: postpartum psychosis, infanticide, and insanity—implications for forensic psychiatry. J Am Acad Psychiatry Law. 2012;40(3):326-332.
27. Friedman SH, Resnick PJ. Child murder by mothers: patterns and prevention. World Psychiatry. 2007;6(3):137-141.
28. Friedman SH, Hrouda DR, Holden CE, et al. Filicide-suicide: common factors in parents who kill their children and themselves. J Am Acad Psychiatry Law. 2005;33(4):496-504.
29. Chandra PS, Venkatasubramanian G, Thomas T. Infanticidal ideas and infanticidal behavior in Indian women with severe postpartum psychiatric disorders. J Nerv Ment Dis. 2002;190(7):457-461.
30. Jones I, Chandra PS, Dazzan P, et al. Bipolar disorder, affective psychosis, and schizophrenia in pregnancy and the post-partum period. Lancet. 2014;384(9956):1789-1799.
31. Friedman SH. Neonaticide. In: Friedman SH. Family murder: pathologies of love and hate. Washington, DC: American Psychiatric Association Publishing; 2018:53-67.
32. Meltzer-Brody S, Brandon AR, Pearson B, et al. Evaluating the clinical effectiveness of a specialized perinatal psychiatry inpatient unit. Arch Womens Ment Health. 2014;17(2):107-113.
33. Klinger G, Stahl B, Fusar-Poli P, et al. Antipsychotic drugs and breastfeeding. Pediatri Endocrinol Rev. 2013;10(3):308-317.
34. Focht A, Kellner CH. Electroconvulsive therapy (ECT) in the treatment of postpartum psychosis. J ECT. 2012;28(1):31-33.
35. Heron J, McGuinness M, Blackmore ER, et al. Early postpartum symptoms in puerperal psychosis. BJOG. 2008;115(3):348-353.
36. McEwan M, Friedman SH. Violence by parents against their children: reporting of maltreatment suspicions, child protection, and risk in mental illness. Psychiatr Clin North Am. 2016;39(4):691-700.
37. Centre of Perinatal Excellence. National Perinatal Mental Health Guideline. http://cope.org.au/about/review-of-new-perinatal-mental-health-guidelines/. Published October 27, 2017. Accessed November 22, 2018.
38. National Institute for Health and Care Excellence. Antenatal and postnatal mental health overview. https://pathways.nice.org.uk/pathways/antenatal-and-postnatal-mental-health. 2017. Accessed November 22, 2018.
39. Wesseloo R, Kamperman AM, Olsen TM, et al. Risk of postpartum relapse in bipolar disorder and postpartum psychosis: a systematic review and meta-analysis. Am J Psychiatry. 2016;173(2):117-127.
40. McKnight RF, Adida M, Budge K, et al. Lithium toxicity profile: a systematic review and meta-analysis. Lancet. 2012;379(9817):721-728.
41. Munk-Olsen T, Liu X, Viktorin A, et al. Maternal and infant outcomes associated with lithium use in pregnancy: an international collaborative meta-analysis of six cohort studies. Lancet Psychiatry. 2018;5(8):644-652.
42. Prakash C, Friedman SH, Moller-Olsen C, et al. Maternal and fetal outcomes after lamotrigine use in pregnancy: a retrospective analysis from an urban maternal mental health centre in New Zealand. Psychopharmacology Bull. 2016;46(2):63-69.
43. Wesseloo R, Liu X, Clark CT, et al. Risk of postpartum episodes in women with bipolar disorder after lamotrigine or lithium use in pregnancy: a population-based cohort study. J Affect Disord. 2017;218:394-397.
44. Dolk H, Wang H, Loane M, et al. Lamotrigine use in pregnancy and risk of orofacial cleft and other congenital anomalies. Neurology. 2016;86(18):1716-1725.
45. Diav-Citrin O, Shechtman S, Zvi N, et al. Is it safe to use lamotrigine during pregnancy? A prospective comparative observational study. Birth Defects Res. 2017;109(15):1196-1203.
46. Kong L, Zhou T, Wang B, et al. The risks associated with the use of lamotrigine during pregnancy. Int J Psychiatry Clin Pract. 2018;22(1):2-5.
47. Deligiannidis KM, Byatt N, Freeman MP. Pharmacotherapy for mood disorders in pregnancy: a review of pharmacokinetic changes and clinical recommendations for therapeutic drug monitoring. J Clin Psychopharmacol. 2014;34(2):244.
48. Bogen DL, Sit D, Genovese A, et al. Three cases of lithium exposure and exclusive breastfeeding. Arch Womens Ment Health. 2012;15(1):69-72.
A new mother drowned her 6-month-old daughter in the bathtub. The married woman, who had a history of schizoaffective disorder, had been high functioning and worked in a managerial role prior to giving birth. However, within a day of delivery, her mental state deteriorated. She quickly became convinced that her daughter had a genetic disorder such as achondroplasia. Physical examinations, genetic testing, and x-rays all failed to alleviate her concerns. Examination of her computer revealed thousands of searches for various medical conditions and surgical treatments. After the baby’s death, the mother was admitted to a psychiatric hospital. She eventually pled guilty to manslaughter.1
Mothers with postpartum psychosis (PPP) typically present fulminantly within days to weeks of giving birth. Symptoms of PPP may include not only psychosis, but also confusion and dysphoric mania. These symptoms often wax and wane, which can make it challenging to establish the diagnosis. In addition, many mothers hide their symptoms due to poor insight, delusions, or fear of loss of custody of their infant. In the vast majority of cases, psychiatric hospitalization is required to protect both mother and baby; untreated, there is an elevated risk of both maternal suicide and infanticide. This article discusses the presentation of PPP, its differential diagnosis, risk factors for developing PPP, suicide and infanticide risk assessment, treatment (including during breastfeeding), and prevention.
The bipolar connection
While multiple factors may increase the risk of PPP (Table 12), women with bipolar disorder have a particularly elevated risk. After experiencing incipient postpartum affective psychosis, a woman has a 50% to 80% chance of having another psychiatric episode, usually within the bipolar spectrum.2 Of all women with PPP, 70% to 90% have bipolar illness or schizoaffective disorder, while approximately 12% have schizophrenia.3,4Women with bipolar disorder are more likely to experience a postpartum psychiatric admission than mothers with any other psychiatric diagnosis5 and have an increased risk of PPP by a factor of 100 over the general population.2
For women with bipolar disorder, PPP should be understood as a recurrence of the chronic disease. Recent evidence does suggest, however, that a significant minority of women progress to experience mood and psychotic symptoms only in the postpartum period.6,7 It is hypothesized that this subgroup of women has a biologic vulnerability to affective psychosis that is limited to the postpartum period. Clinically, understanding a woman’s disease course is important because it may guide decision-making about prophylactic medications during or after pregnancy.
A rapid, delirium-like presentation
Postpartum psychosis is a rare disorder, with a prevalence of 1 to 2 cases per 1,000 childbirths.3 While symptoms may begin days to weeks postpartum, the typical time of onset is between 3 to 10 days after birth, occurring after a woman has been discharged from the hospital and during a time of change and uncertainty. This can make the presentation of PPP a confusing and distressing experience for both the new mother and the family, resulting in delays in seeking care.
Subtle prodromal symptoms may include insomnia, mood fluctuation, and irritability. As symptoms progress, PPP is notable for a rapid onset and a delirium-like appearance that may include waxing and waning cognitive symptoms such as disorientation and confusion.8 Grossly disorganized behaviors and rapid mood fluctuations are typical. Distinct from mood episodes outside the peripartum period, women with PPP often experience mood-incongruent delusions and obsessive thoughts, often focused on their child.9 Women with PPP appear less likely to experience thought insertion or withdrawal or auditory hallucinations that give a running commentary.2
Differential diagnosis includes depression, OCD
When evaluating a woman with possible postpartum psychotic symptoms or delirium, it is important to include a thorough history, physical examination, and relevant laboratory and/or imaging investigations to assess for organic causes or contributors (Table 22,6,10-12 and Table 32,6,10-12). A detailed psychiatric history should establish whether the patient is presenting with new-onset psychosis or has had previous mood or psychotic episodes that may have gone undetected. Important perinatal psychiatric differential diagnoses should include “baby blues,” postpartum depression (PPD), and obsessive-compulsive disorder (OCD).
Continue to: PPP vs "baby blues."
PPP vs “baby blues.” “Baby blues” is not an official DSM-5 diagnosis but rather a normative postpartum experience that affects 50% to 80% of postpartum women. A woman with the “baby blues” may feel weepy or have mild mood lability, irritability, or anxiety; however, these symptoms do not significantly impair function. Peak symptoms typically occur between 2 to 5 days postpartum and generally resolve within 2 weeks. Women who have the “baby blues” are at an increased risk for PPD and should be monitored over time.13,14
PPP vs PPD. Postpartum depression affects approximately 10% to 15% of new mothers.15 Women with PPD may experience feelings of persistent and severe sadness, feelings of detachment, insomnia, and fatigue. Symptoms of PPD can interfere with a mother’s interest in caring for her baby and present a barrier to maternal bonding.16,17
As the awareness of PPD has increased in recent years, screening for depressive symptoms during and after pregnancy has increasingly become the standard of care.18 When evaluating a postpartum woman for PPD, it is important to consider PPP in the differential. Women with severe or persistent depressive symptoms may also develop psychotic symptoms. Furthermore, suicidal thoughts or thoughts of harming the infant may be present in either PPD or PPP. One study found that 41% of mothers with depression endorsed thoughts of harming their infants.19
PPP vs postpartum OCD. Postpartum obsessive-compulsive symptoms commonly occur comorbidly with PPD,9 and OCD often presents for the first time in the postpartum period.20 Obsessive-compulsive disorder affects between 2% to 9% of new mothers.21,22 It is critical to properly differentiate PPP from postpartum OCD. Clinical questions should be posed with a non-judgmental stance. Just as delusions in PPP are often focused on the infant, for women with OCD, obsessive thoughts may center on worries about the infant’s safety. Distressing obsessions about violence are common in OCD.23 Mothers with OCD may experience intrusive thinking about accidentally or purposefully harming their infant. For example, they may intrusively worry that they will accidentally put the baby in the microwave or oven, leave the baby in a hot car, or throw the baby down the stairs. However, a postpartum woman with OCD may be reluctant to share her ego-dystonic thoughts of infant harm. Mothers with OCD are not out of touch with reality; instead, their intrusive thoughts are ego-dystonic and distressing. These are thoughts and fears that they focus on and try to avoid, rather than plan. The psychiatrist must carefully differentiate between ego-syntonic and ego-dystonic thoughts. These patients often avoid seeking treatment because of their shame and guilt.23 Clinicians often under-recognize OCD and risk inappropriate hospitalization, treatment, and inappropriate referral to Child Protective Services (CPS).23
Perinatal psychiatric risk assessment
When a mother develops PPP, consider the risks of suicide, child harm, and infanticide. Although suicide risk is generally lower in the postpartum period, suicide is the cause of 20% of postpartum deaths.24,25 When PPP is untreated, suicide risk is elevated. A careful suicide risk assessment should be completed.
Continue to: Particularly in PPP...
Particularly in PPP, a mother may be at risk of child neglect or abuse due to her confused or delusional thinking and mood state.26 For example, one mother heated empty bottles and gave them to her baby, and then became frustrated when the baby continued to cry.
The risk of infanticide is also elevated in untreated PPP, with approximately 4% of these women committing infanticide.9 There are 5 motives for infanticide (Table 427). Altruistic and acutely psychotic motives are more likely to be related to PPP, while fatal maltreatment, unwanted child, and partner revenge motives are less likely to be related to PPP. Among mothers who kill both their child and themselves (filicide-suicide), altruistic motives were the most common.28 Mothers in psychiatric samples who kill their children have often experienced psychosis, suicidality, depression, and significant life stresses.27 Both infanticidal ideas and behaviors have been associated with psychotic thinking about the infant,29 so it is critical to ascertain whether the mother’s delusions or hallucinations involve the infant.30 In contrast, neonaticide (murder in the first day of life) is rarely related to PPP because PPP typically has a later onset.31

Treating acute PPP
The fulminant nature of PPP can make its treatment difficult. Thinking through the case in an organized fashion is critical (Table 5).

Hospitalization. Postpartum psychosis is a psychiatric emergency with a rapid onset of symptoms. Hospitalization is required in almost all cases for diagnostic evaluation, assessment and management of safety, and initiation of treatment. While maternal-infant bonding in the perinatal period is important, infant safety is critical and usually requires maternal psychiatric hospitalization.
The specialized mother-baby psychiatric unit (MBU) is a model of care first developed in the United Kingdom and is now available in many European countries as well as in New Zealand and Australia. Mother-baby psychiatric units admit the mother and the baby together and provide dyadic treatment to allow for enhanced bonding and parenting support, and often to encourage breastfeeding.30 In the United States, there has been growing interest in specialized inpatient settings that acknowledge the importance of maternal-infant attachment in the treatment of perinatal disorders and provide care with a dyadic focus; however, differences in the health care payer system have been a barrier to full-scale MBUs. The Perinatal Psychiatry Inpatient Unit at University of North Carolina-Chapel Hill is among the first of such a model in the United States.32
Continue to: Although this specialized treatment setting...
Although this specialized treatment setting is unlikely to be available in most American cities, treatment should still consider the maternal role. When possible, the infant should stay with the father or family members during the mother’s hospitalization, and supervised visits should be arranged when appropriate. If the mother is breastfeeding, or plans to breastfeed after the hospitalization, the treatment team may consider providing supervised use of a breast pump and making arrangements for breast milk storage. During the mother’s hospitalization, staff should provide psychoeducation and convey hopefulness and support.
Medication management. Mood stabilizers and second-generation antipsychotics (SGAs) are often used for acute management of PPP. The choice of medication is determined by individual symptoms, severity of presentation, previous response to medication, and maternal adverse effects.30 In a naturalistic study of 64 women admitted for new-onset PPP, sequential administration of benzodiazepines, antipsychotics, and lithium was found to be effective in achieving remission for 99% of patients, with 80% sustaining remission at 9 months postpartum.6 Second-generation antipsychotics such as
Breastfeeding. It is important to discuss breastfeeding with the mother and her partner or family. The patient’s preference, the maternal and infant benefits of breastfeeding, the potential for sleep disruption, and the safety profile of needed medications should all be considered. Because sleep loss is a modifiable risk factor in PPP, the benefits of breastfeeding may be outweighed by the risks for some patients.9 For others, breastfeeding during the day and bottle-feeding at night may be preferred.
What to consider during discharge planning
Discharge arrangements require careful consideration (Table 6). Meet with the family prior to discharge to provide psychoeducation and to underscore the importance of family involvement with both mother and infant. It is important to ensure adequate support at home, including at night, since sleep is critical to improved stability. Encourage the patient and her family to monitor for early warning signs of relapse, which might include refractory insomnia, mood instability, poor judgment, or hypomanic symptoms.35 She should be followed closely as an outpatient. Having her partner (or another close family member) and infant present during appointments can help in obtaining collateral information and assessing mother-infant bonding. The clinician should also consider whether it is necessary to contact CPS. Many mothers with mental illness appropriately parent their child, but CPS should be alerted when there is a reasonable concern about safe parenting—abuse, neglect, or significant risk.36

Take steps for prevention
An important part of managing PPP is prevention. This involves providing preconception counseling to the woman and her partner.30 Preconception advice should be individualized and include discussion of:
- risks of relapse in pregnancy and the postpartum period
- optimal physical and mental health
- potential risks and benefits of medication options in pregnancy
- potential effects of untreated illness for the fetus, infant, and family
- a strategy outlining whether medication is continued in pregnancy or started in the postpartum period.
Continue to: For women at risk of PPP...
For women at risk of PPP, the risks of medications need to be balanced with the risks of untreated illness. To reduce the risk of PPP relapse, guidelines recommend a robust antenatal care plan that should include37,38:
- close monitoring of a woman’s mental state for early warning signs of PPP, with active participation from the woman’s partner and family
- ongoing discussion of the risks and benefits of pharmacotherapy (and, for women who prefer to not take medication in the first trimester, a plan for when medications will be restarted)
- collaboration with other professionals involved in care during pregnancy and postpartum (eg, obstetricians, midwives, family practitioners, pediatricians)
- planning to minimize risk factors associated with relapse (eg, sleep deprivation, lack of social supports, domestic violence, and substance abuse).
Evidence clearly suggests that women with bipolar disorder are at increased risk for illness recurrence without continued maintenance medication.39 A subgroup of women with PPP go on to have psychosis limited to the postpartum period, and reinstating prophylactic medication in late pregnancy (preferably) or immediately after birth should be discussed.2 The choice of prophylactic medication should be determined by the woman’s previous response.
Regarding prophylaxis, the most evidence exists for lithium.6 Lithium use during the first trimester carries a risk of Ebstein’s anomaly. However, a recent systematic review and meta-analysis have concluded that the teratogenic risks of lithium have been overestimated.40,41
Lamotrigine is an alternative mood stabilizer with a favorable safety profile in pregnancy. In a small naturalistic study in which lamotrigine was continued in pregnancy in women with bipolar disorder, the medication was effective in preventing relapse in pregnancy and postpartum.42 A small population-based cohort study found lamotrigine was as effective as lithium in preventing severe postpartum relapse in women with bipolar disorder,43 although this study was limited by its observational design. Recently published studies have found no significant association between
Box
It is essential to consider the patient’s individual symptoms and treatment history when making pharmacologic recommendations during pregnancy. Discussion with the patient about the risks and benefits of lithium is recommended. For women who continue to use lithium during pregnancy, ongoing pharmacokinetic changes warrant more frequent monitoring (some experts advise monthly monitoring throughout pregnancy, moving to more frequent monitoring at 36 weeks).47 During labor, the team might consider temporary cessation of lithium and particular attention to hydration status.30 In the postpartum period, there is a quick return to baseline glomerular filtration rate and a rapid decrease in vascular volume, so it is advisable to restart the patient at her pre-pregnancy lithium dosage. It is recommended to check lithium levels within 24 hours of delivery.47 While lithium is not an absolute contraindication to breastfeeding, there is particular concern in situations of prematurity or neonatal dehydration. Collaboration with and close monitoring by the pediatrician is essential to determine an infant monitoring plan.48
If lamotrigine is used during pregnancy, be aware that pregnancy-related pharmacokinetic changes result in increased lamotrigine clearance, which will vary in magnitude among individuals. Faster clearance may necessitate dose increases during pregnancy and a taper back to pre-pregnancy dose in the postpartum period. Dosing should always take clinical symptoms into account.
Pharmacotherapy can reduce relapse risk
To prevent relapse in the postpartum period, consider initiating treatment with mood stabilizers and/or SGAs, particularly for women with bipolar disorder who do not take medication during pregnancy. A recent meta-analysis found a high postpartum relapse rate (66%) in women with bipolar disorder who did not take prophylactic medication, compared with a relapse rate of 23% for women who did take such medication. In women with psychosis limited to the postpartum period, prophylaxis with lithium or antipsychotics in the immediate postpartum can prevent relapse.39 The SGAs olanzapine and quetiapine are often used to manage acute symptoms because they are considered acceptable during breastfeeding.33 The use of lithium when breastfeeding is complex to manage48 and may require advice to not breastfeed, which can be an important consideration for patients and their families.
Bottom Line
Postpartum psychosis (PPP) typically presents with a rapid onset of hallucinations, delusions, confusion, and mood swings within days to weeks of giving birth. Mothers with PPP almost always require hospitalization for the safety of their infants and themselves. Mood stabilizers and second-generation antipsychotics are used for acute management.
Related Resources
- Clark CT, Wisner KL. Treatment of peripartum bipolar disorder. Obstet Gynecol Clin N Am. 2018;45:403-417.
- Massachusetts General Hospital Center for Women’s Mental Health. https://womensmentalhealth.org/. 2018.
- Postpartum Support International. Postpartum psychosis. http://www.postpartum.net/learn-more/postpartumpsychosis/. 2019.
Drug Brand Names
Bromocriptine • Cycloset, Parlodel
Cabergoline • Dostinex
Lamotrigine • Lamictal
Lithium • Eskalith, Lithobid
Olanzapine • Zyprexa
Quetiapine • Seroquel
A new mother drowned her 6-month-old daughter in the bathtub. The married woman, who had a history of schizoaffective disorder, had been high functioning and worked in a managerial role prior to giving birth. However, within a day of delivery, her mental state deteriorated. She quickly became convinced that her daughter had a genetic disorder such as achondroplasia. Physical examinations, genetic testing, and x-rays all failed to alleviate her concerns. Examination of her computer revealed thousands of searches for various medical conditions and surgical treatments. After the baby’s death, the mother was admitted to a psychiatric hospital. She eventually pled guilty to manslaughter.1
Mothers with postpartum psychosis (PPP) typically present fulminantly within days to weeks of giving birth. Symptoms of PPP may include not only psychosis, but also confusion and dysphoric mania. These symptoms often wax and wane, which can make it challenging to establish the diagnosis. In addition, many mothers hide their symptoms due to poor insight, delusions, or fear of loss of custody of their infant. In the vast majority of cases, psychiatric hospitalization is required to protect both mother and baby; untreated, there is an elevated risk of both maternal suicide and infanticide. This article discusses the presentation of PPP, its differential diagnosis, risk factors for developing PPP, suicide and infanticide risk assessment, treatment (including during breastfeeding), and prevention.
The bipolar connection
While multiple factors may increase the risk of PPP (Table 12), women with bipolar disorder have a particularly elevated risk. After experiencing incipient postpartum affective psychosis, a woman has a 50% to 80% chance of having another psychiatric episode, usually within the bipolar spectrum.2 Of all women with PPP, 70% to 90% have bipolar illness or schizoaffective disorder, while approximately 12% have schizophrenia.3,4Women with bipolar disorder are more likely to experience a postpartum psychiatric admission than mothers with any other psychiatric diagnosis5 and have an increased risk of PPP by a factor of 100 over the general population.2
For women with bipolar disorder, PPP should be understood as a recurrence of the chronic disease. Recent evidence does suggest, however, that a significant minority of women progress to experience mood and psychotic symptoms only in the postpartum period.6,7 It is hypothesized that this subgroup of women has a biologic vulnerability to affective psychosis that is limited to the postpartum period. Clinically, understanding a woman’s disease course is important because it may guide decision-making about prophylactic medications during or after pregnancy.
A rapid, delirium-like presentation
Postpartum psychosis is a rare disorder, with a prevalence of 1 to 2 cases per 1,000 childbirths.3 While symptoms may begin days to weeks postpartum, the typical time of onset is between 3 to 10 days after birth, occurring after a woman has been discharged from the hospital and during a time of change and uncertainty. This can make the presentation of PPP a confusing and distressing experience for both the new mother and the family, resulting in delays in seeking care.
Subtle prodromal symptoms may include insomnia, mood fluctuation, and irritability. As symptoms progress, PPP is notable for a rapid onset and a delirium-like appearance that may include waxing and waning cognitive symptoms such as disorientation and confusion.8 Grossly disorganized behaviors and rapid mood fluctuations are typical. Distinct from mood episodes outside the peripartum period, women with PPP often experience mood-incongruent delusions and obsessive thoughts, often focused on their child.9 Women with PPP appear less likely to experience thought insertion or withdrawal or auditory hallucinations that give a running commentary.2
Differential diagnosis includes depression, OCD
When evaluating a woman with possible postpartum psychotic symptoms or delirium, it is important to include a thorough history, physical examination, and relevant laboratory and/or imaging investigations to assess for organic causes or contributors (Table 22,6,10-12 and Table 32,6,10-12). A detailed psychiatric history should establish whether the patient is presenting with new-onset psychosis or has had previous mood or psychotic episodes that may have gone undetected. Important perinatal psychiatric differential diagnoses should include “baby blues,” postpartum depression (PPD), and obsessive-compulsive disorder (OCD).
Continue to: PPP vs "baby blues."
PPP vs “baby blues.” “Baby blues” is not an official DSM-5 diagnosis but rather a normative postpartum experience that affects 50% to 80% of postpartum women. A woman with the “baby blues” may feel weepy or have mild mood lability, irritability, or anxiety; however, these symptoms do not significantly impair function. Peak symptoms typically occur between 2 to 5 days postpartum and generally resolve within 2 weeks. Women who have the “baby blues” are at an increased risk for PPD and should be monitored over time.13,14
PPP vs PPD. Postpartum depression affects approximately 10% to 15% of new mothers.15 Women with PPD may experience feelings of persistent and severe sadness, feelings of detachment, insomnia, and fatigue. Symptoms of PPD can interfere with a mother’s interest in caring for her baby and present a barrier to maternal bonding.16,17
As the awareness of PPD has increased in recent years, screening for depressive symptoms during and after pregnancy has increasingly become the standard of care.18 When evaluating a postpartum woman for PPD, it is important to consider PPP in the differential. Women with severe or persistent depressive symptoms may also develop psychotic symptoms. Furthermore, suicidal thoughts or thoughts of harming the infant may be present in either PPD or PPP. One study found that 41% of mothers with depression endorsed thoughts of harming their infants.19
PPP vs postpartum OCD. Postpartum obsessive-compulsive symptoms commonly occur comorbidly with PPD,9 and OCD often presents for the first time in the postpartum period.20 Obsessive-compulsive disorder affects between 2% to 9% of new mothers.21,22 It is critical to properly differentiate PPP from postpartum OCD. Clinical questions should be posed with a non-judgmental stance. Just as delusions in PPP are often focused on the infant, for women with OCD, obsessive thoughts may center on worries about the infant’s safety. Distressing obsessions about violence are common in OCD.23 Mothers with OCD may experience intrusive thinking about accidentally or purposefully harming their infant. For example, they may intrusively worry that they will accidentally put the baby in the microwave or oven, leave the baby in a hot car, or throw the baby down the stairs. However, a postpartum woman with OCD may be reluctant to share her ego-dystonic thoughts of infant harm. Mothers with OCD are not out of touch with reality; instead, their intrusive thoughts are ego-dystonic and distressing. These are thoughts and fears that they focus on and try to avoid, rather than plan. The psychiatrist must carefully differentiate between ego-syntonic and ego-dystonic thoughts. These patients often avoid seeking treatment because of their shame and guilt.23 Clinicians often under-recognize OCD and risk inappropriate hospitalization, treatment, and inappropriate referral to Child Protective Services (CPS).23
Perinatal psychiatric risk assessment
When a mother develops PPP, consider the risks of suicide, child harm, and infanticide. Although suicide risk is generally lower in the postpartum period, suicide is the cause of 20% of postpartum deaths.24,25 When PPP is untreated, suicide risk is elevated. A careful suicide risk assessment should be completed.
Continue to: Particularly in PPP...
Particularly in PPP, a mother may be at risk of child neglect or abuse due to her confused or delusional thinking and mood state.26 For example, one mother heated empty bottles and gave them to her baby, and then became frustrated when the baby continued to cry.
The risk of infanticide is also elevated in untreated PPP, with approximately 4% of these women committing infanticide.9 There are 5 motives for infanticide (Table 427). Altruistic and acutely psychotic motives are more likely to be related to PPP, while fatal maltreatment, unwanted child, and partner revenge motives are less likely to be related to PPP. Among mothers who kill both their child and themselves (filicide-suicide), altruistic motives were the most common.28 Mothers in psychiatric samples who kill their children have often experienced psychosis, suicidality, depression, and significant life stresses.27 Both infanticidal ideas and behaviors have been associated with psychotic thinking about the infant,29 so it is critical to ascertain whether the mother’s delusions or hallucinations involve the infant.30 In contrast, neonaticide (murder in the first day of life) is rarely related to PPP because PPP typically has a later onset.31

Treating acute PPP
The fulminant nature of PPP can make its treatment difficult. Thinking through the case in an organized fashion is critical (Table 5).

Hospitalization. Postpartum psychosis is a psychiatric emergency with a rapid onset of symptoms. Hospitalization is required in almost all cases for diagnostic evaluation, assessment and management of safety, and initiation of treatment. While maternal-infant bonding in the perinatal period is important, infant safety is critical and usually requires maternal psychiatric hospitalization.
The specialized mother-baby psychiatric unit (MBU) is a model of care first developed in the United Kingdom and is now available in many European countries as well as in New Zealand and Australia. Mother-baby psychiatric units admit the mother and the baby together and provide dyadic treatment to allow for enhanced bonding and parenting support, and often to encourage breastfeeding.30 In the United States, there has been growing interest in specialized inpatient settings that acknowledge the importance of maternal-infant attachment in the treatment of perinatal disorders and provide care with a dyadic focus; however, differences in the health care payer system have been a barrier to full-scale MBUs. The Perinatal Psychiatry Inpatient Unit at University of North Carolina-Chapel Hill is among the first of such a model in the United States.32
Continue to: Although this specialized treatment setting...
Although this specialized treatment setting is unlikely to be available in most American cities, treatment should still consider the maternal role. When possible, the infant should stay with the father or family members during the mother’s hospitalization, and supervised visits should be arranged when appropriate. If the mother is breastfeeding, or plans to breastfeed after the hospitalization, the treatment team may consider providing supervised use of a breast pump and making arrangements for breast milk storage. During the mother’s hospitalization, staff should provide psychoeducation and convey hopefulness and support.
Medication management. Mood stabilizers and second-generation antipsychotics (SGAs) are often used for acute management of PPP. The choice of medication is determined by individual symptoms, severity of presentation, previous response to medication, and maternal adverse effects.30 In a naturalistic study of 64 women admitted for new-onset PPP, sequential administration of benzodiazepines, antipsychotics, and lithium was found to be effective in achieving remission for 99% of patients, with 80% sustaining remission at 9 months postpartum.6 Second-generation antipsychotics such as
Breastfeeding. It is important to discuss breastfeeding with the mother and her partner or family. The patient’s preference, the maternal and infant benefits of breastfeeding, the potential for sleep disruption, and the safety profile of needed medications should all be considered. Because sleep loss is a modifiable risk factor in PPP, the benefits of breastfeeding may be outweighed by the risks for some patients.9 For others, breastfeeding during the day and bottle-feeding at night may be preferred.
What to consider during discharge planning
Discharge arrangements require careful consideration (Table 6). Meet with the family prior to discharge to provide psychoeducation and to underscore the importance of family involvement with both mother and infant. It is important to ensure adequate support at home, including at night, since sleep is critical to improved stability. Encourage the patient and her family to monitor for early warning signs of relapse, which might include refractory insomnia, mood instability, poor judgment, or hypomanic symptoms.35 She should be followed closely as an outpatient. Having her partner (or another close family member) and infant present during appointments can help in obtaining collateral information and assessing mother-infant bonding. The clinician should also consider whether it is necessary to contact CPS. Many mothers with mental illness appropriately parent their child, but CPS should be alerted when there is a reasonable concern about safe parenting—abuse, neglect, or significant risk.36

Take steps for prevention
An important part of managing PPP is prevention. This involves providing preconception counseling to the woman and her partner.30 Preconception advice should be individualized and include discussion of:
- risks of relapse in pregnancy and the postpartum period
- optimal physical and mental health
- potential risks and benefits of medication options in pregnancy
- potential effects of untreated illness for the fetus, infant, and family
- a strategy outlining whether medication is continued in pregnancy or started in the postpartum period.
Continue to: For women at risk of PPP...
For women at risk of PPP, the risks of medications need to be balanced with the risks of untreated illness. To reduce the risk of PPP relapse, guidelines recommend a robust antenatal care plan that should include37,38:
- close monitoring of a woman’s mental state for early warning signs of PPP, with active participation from the woman’s partner and family
- ongoing discussion of the risks and benefits of pharmacotherapy (and, for women who prefer to not take medication in the first trimester, a plan for when medications will be restarted)
- collaboration with other professionals involved in care during pregnancy and postpartum (eg, obstetricians, midwives, family practitioners, pediatricians)
- planning to minimize risk factors associated with relapse (eg, sleep deprivation, lack of social supports, domestic violence, and substance abuse).
Evidence clearly suggests that women with bipolar disorder are at increased risk for illness recurrence without continued maintenance medication.39 A subgroup of women with PPP go on to have psychosis limited to the postpartum period, and reinstating prophylactic medication in late pregnancy (preferably) or immediately after birth should be discussed.2 The choice of prophylactic medication should be determined by the woman’s previous response.
Regarding prophylaxis, the most evidence exists for lithium.6 Lithium use during the first trimester carries a risk of Ebstein’s anomaly. However, a recent systematic review and meta-analysis have concluded that the teratogenic risks of lithium have been overestimated.40,41
Lamotrigine is an alternative mood stabilizer with a favorable safety profile in pregnancy. In a small naturalistic study in which lamotrigine was continued in pregnancy in women with bipolar disorder, the medication was effective in preventing relapse in pregnancy and postpartum.42 A small population-based cohort study found lamotrigine was as effective as lithium in preventing severe postpartum relapse in women with bipolar disorder,43 although this study was limited by its observational design. Recently published studies have found no significant association between
Box
It is essential to consider the patient’s individual symptoms and treatment history when making pharmacologic recommendations during pregnancy. Discussion with the patient about the risks and benefits of lithium is recommended. For women who continue to use lithium during pregnancy, ongoing pharmacokinetic changes warrant more frequent monitoring (some experts advise monthly monitoring throughout pregnancy, moving to more frequent monitoring at 36 weeks).47 During labor, the team might consider temporary cessation of lithium and particular attention to hydration status.30 In the postpartum period, there is a quick return to baseline glomerular filtration rate and a rapid decrease in vascular volume, so it is advisable to restart the patient at her pre-pregnancy lithium dosage. It is recommended to check lithium levels within 24 hours of delivery.47 While lithium is not an absolute contraindication to breastfeeding, there is particular concern in situations of prematurity or neonatal dehydration. Collaboration with and close monitoring by the pediatrician is essential to determine an infant monitoring plan.48
If lamotrigine is used during pregnancy, be aware that pregnancy-related pharmacokinetic changes result in increased lamotrigine clearance, which will vary in magnitude among individuals. Faster clearance may necessitate dose increases during pregnancy and a taper back to pre-pregnancy dose in the postpartum period. Dosing should always take clinical symptoms into account.
Pharmacotherapy can reduce relapse risk
To prevent relapse in the postpartum period, consider initiating treatment with mood stabilizers and/or SGAs, particularly for women with bipolar disorder who do not take medication during pregnancy. A recent meta-analysis found a high postpartum relapse rate (66%) in women with bipolar disorder who did not take prophylactic medication, compared with a relapse rate of 23% for women who did take such medication. In women with psychosis limited to the postpartum period, prophylaxis with lithium or antipsychotics in the immediate postpartum can prevent relapse.39 The SGAs olanzapine and quetiapine are often used to manage acute symptoms because they are considered acceptable during breastfeeding.33 The use of lithium when breastfeeding is complex to manage48 and may require advice to not breastfeed, which can be an important consideration for patients and their families.
Bottom Line
Postpartum psychosis (PPP) typically presents with a rapid onset of hallucinations, delusions, confusion, and mood swings within days to weeks of giving birth. Mothers with PPP almost always require hospitalization for the safety of their infants and themselves. Mood stabilizers and second-generation antipsychotics are used for acute management.
Related Resources
- Clark CT, Wisner KL. Treatment of peripartum bipolar disorder. Obstet Gynecol Clin N Am. 2018;45:403-417.
- Massachusetts General Hospital Center for Women’s Mental Health. https://womensmentalhealth.org/. 2018.
- Postpartum Support International. Postpartum psychosis. http://www.postpartum.net/learn-more/postpartumpsychosis/. 2019.
Drug Brand Names
Bromocriptine • Cycloset, Parlodel
Cabergoline • Dostinex
Lamotrigine • Lamictal
Lithium • Eskalith, Lithobid
Olanzapine • Zyprexa
Quetiapine • Seroquel
1. Hall L. Mother who killed baby believing she was a dwarf should not be jailed, court told. The Sydney Morning Herald. https://www.smh.com.au/national/nsw/mother-who-killed-baby-believing-she-was-a-dwarf-should-not-be-jailed-court-told-20170428-gvud4d.html. Published April 28, 2017. Accessed March 12, 2019.
2. Bergink V, Rasgon N, Wisner KL. Postpartum psychosis: madness, mania, and melancholia in motherhood. Am J Psychiatry. 2016;173(12):1179-1188.
3. Sit D, Rothschild AJ, Wisner KL. A review of postpartum psychosis. J Womens Health (Larchmt). 2006;15(4):352-368.
4. Kendell RE, Chalmers JC, Platz C. Epidemiology of puerperal psychoses. Br J Psychiatry. 1987;150(5):662-673.
5. Munk-Olsen T, Laursen TM, Mendelson T, et al. Risks and predictors of readmission for a mental disorder during the postpartum period. Arch Gen Psychiatry. 2009;66(2):189-195.
6. Bergink V, Burgerhout KM, Koorengevel KM, et al. Treatment of psychosis and mania in the postpartum period. Am J Psychiatry. 2015;172(2):115-123.
7. Wesseloo R, Kamperman AM, Munk-Olsen T, et al. Risk of postpartum relapse in bipolar disorder and postpartum psychosis: a systematic review and meta-analysis. Am J Psychiatry. 2015;173(2):117-127.
8. Wisner KL, Peindl K, Hanusa BH. Symptomatology of affective and psychotic illnesses related to childbearing. J Affect Disord. 1994;30(2):77-87.
9. Spinelli MG. Postpartum psychosis: detection of risk and management. Am J Psychiatry. 2009;166(4):405-408.
10. Fassier T, Guffon N, Acquaviva C, et al. Misdiagnosed postpartum psychosis revealing a late-onset urea cycle disorder. Am J Psychiatry. 2011;168(6):576-580.
11. Yu AYX, Moore FG. Paraneoplastic encephalitis presenting as postpartum psychosis. Psychosomatics. 2011;52(6):568-570.
12. Patil NJ, Yadav SS, Gokhale YA, et al. Primary hypoparathyroidism: psychosis in postpartum period. J Assoc Physicians India. 2010;58:506-508.
13. O’Hara MW, Schlechte JA, Lewis DA, et al. Prospective study of postpartum blues: biologic and psychosocial factors. Arch Gen Psychiatry. 1991;48(9):801-806.
14. Burt VK, Hendrick VC. Clinical manual of women’s mental health. Washington, DC. American Psychiatric Association Publishing; 2007:79-80.
15. Melzer-Brody S. Postpartum depression: what to tell patients who breast-feed. Current Psychiatry. 2008;7(5):87-95.
16. Alhusen JL, Gross D, Hayat MJ, et al. The role of mental health on maternal‐fetal attachment in low‐income women. J Obstet Gynecol Neonatal Nurs. 2012;41(6):E71-E81.
17. McLearn KT, Minkovitz CS, Strobino DM, et al. Maternal depressive symptoms at 2 to 4 months postpartum and early parenting practices. Arch Pediatr Adolesc Med. 2006;160(3):279-284.
18. Committee on Obstetric Practice. The American College of Obstetricians and Gynecologists Committee Opinion no. 630. Screening for perinatal depression. Obstet Gynecol. 2015;125(5):1268-1271.
19. Jennings KD, Ross S, Popper S. Thoughts of harming infants in depressed and nondepressed mothers. J Affect Disord. 1999;54(1-2):21-28.
20. Miller ES, Hoxha D, Wisner KL, et al. Obsessions and compulsions in postpartum women without obsessive compulsive disorder. J Womens Health. 2015;24(10):825-830.
21. Russell EJ, Fawcett JM, Mazmanian D. Risk of obsessive-compulsive disorder in pregnant and postpartum women: a meta-analysis. J Clin Psychiatry. 2013;74(4):377-385.
22. Zambaldi CF, Cantilino A, Montenegro AC, et al. Postpartum obsessive-compulsive disorder: prevalence and clinical characteristics. Compr Psychiatry. 2009;50(6):503-509.
23. Booth BD, Friedman SH, Curry S, et al. Obsessions of child murder: underrecognized manifestations of obsessive-compulsive disorder. J Am Acad Psychiatry Law. 2014;42(1):66-74.
24. Lindahl V, Pearson JL, Colpe L. Prevalence of suicidality during pregnancy and the postpartum. Arch Womens Ment Health. 2005;8(2):77-87.
25. Samandari G, Martin SL, Kupper LL, et al. Are pregnant and postpartum women: at increased risk for violent death? Suicide and homicide findings from North Carolina. Matern Child Health J. 2011;15(5):660-669.
26. Friedman SH, Sorrentino R. Commentary: postpartum psychosis, infanticide, and insanity—implications for forensic psychiatry. J Am Acad Psychiatry Law. 2012;40(3):326-332.
27. Friedman SH, Resnick PJ. Child murder by mothers: patterns and prevention. World Psychiatry. 2007;6(3):137-141.
28. Friedman SH, Hrouda DR, Holden CE, et al. Filicide-suicide: common factors in parents who kill their children and themselves. J Am Acad Psychiatry Law. 2005;33(4):496-504.
29. Chandra PS, Venkatasubramanian G, Thomas T. Infanticidal ideas and infanticidal behavior in Indian women with severe postpartum psychiatric disorders. J Nerv Ment Dis. 2002;190(7):457-461.
30. Jones I, Chandra PS, Dazzan P, et al. Bipolar disorder, affective psychosis, and schizophrenia in pregnancy and the post-partum period. Lancet. 2014;384(9956):1789-1799.
31. Friedman SH. Neonaticide. In: Friedman SH. Family murder: pathologies of love and hate. Washington, DC: American Psychiatric Association Publishing; 2018:53-67.
32. Meltzer-Brody S, Brandon AR, Pearson B, et al. Evaluating the clinical effectiveness of a specialized perinatal psychiatry inpatient unit. Arch Womens Ment Health. 2014;17(2):107-113.
33. Klinger G, Stahl B, Fusar-Poli P, et al. Antipsychotic drugs and breastfeeding. Pediatri Endocrinol Rev. 2013;10(3):308-317.
34. Focht A, Kellner CH. Electroconvulsive therapy (ECT) in the treatment of postpartum psychosis. J ECT. 2012;28(1):31-33.
35. Heron J, McGuinness M, Blackmore ER, et al. Early postpartum symptoms in puerperal psychosis. BJOG. 2008;115(3):348-353.
36. McEwan M, Friedman SH. Violence by parents against their children: reporting of maltreatment suspicions, child protection, and risk in mental illness. Psychiatr Clin North Am. 2016;39(4):691-700.
37. Centre of Perinatal Excellence. National Perinatal Mental Health Guideline. http://cope.org.au/about/review-of-new-perinatal-mental-health-guidelines/. Published October 27, 2017. Accessed November 22, 2018.
38. National Institute for Health and Care Excellence. Antenatal and postnatal mental health overview. https://pathways.nice.org.uk/pathways/antenatal-and-postnatal-mental-health. 2017. Accessed November 22, 2018.
39. Wesseloo R, Kamperman AM, Olsen TM, et al. Risk of postpartum relapse in bipolar disorder and postpartum psychosis: a systematic review and meta-analysis. Am J Psychiatry. 2016;173(2):117-127.
40. McKnight RF, Adida M, Budge K, et al. Lithium toxicity profile: a systematic review and meta-analysis. Lancet. 2012;379(9817):721-728.
41. Munk-Olsen T, Liu X, Viktorin A, et al. Maternal and infant outcomes associated with lithium use in pregnancy: an international collaborative meta-analysis of six cohort studies. Lancet Psychiatry. 2018;5(8):644-652.
42. Prakash C, Friedman SH, Moller-Olsen C, et al. Maternal and fetal outcomes after lamotrigine use in pregnancy: a retrospective analysis from an urban maternal mental health centre in New Zealand. Psychopharmacology Bull. 2016;46(2):63-69.
43. Wesseloo R, Liu X, Clark CT, et al. Risk of postpartum episodes in women with bipolar disorder after lamotrigine or lithium use in pregnancy: a population-based cohort study. J Affect Disord. 2017;218:394-397.
44. Dolk H, Wang H, Loane M, et al. Lamotrigine use in pregnancy and risk of orofacial cleft and other congenital anomalies. Neurology. 2016;86(18):1716-1725.
45. Diav-Citrin O, Shechtman S, Zvi N, et al. Is it safe to use lamotrigine during pregnancy? A prospective comparative observational study. Birth Defects Res. 2017;109(15):1196-1203.
46. Kong L, Zhou T, Wang B, et al. The risks associated with the use of lamotrigine during pregnancy. Int J Psychiatry Clin Pract. 2018;22(1):2-5.
47. Deligiannidis KM, Byatt N, Freeman MP. Pharmacotherapy for mood disorders in pregnancy: a review of pharmacokinetic changes and clinical recommendations for therapeutic drug monitoring. J Clin Psychopharmacol. 2014;34(2):244.
48. Bogen DL, Sit D, Genovese A, et al. Three cases of lithium exposure and exclusive breastfeeding. Arch Womens Ment Health. 2012;15(1):69-72.
1. Hall L. Mother who killed baby believing she was a dwarf should not be jailed, court told. The Sydney Morning Herald. https://www.smh.com.au/national/nsw/mother-who-killed-baby-believing-she-was-a-dwarf-should-not-be-jailed-court-told-20170428-gvud4d.html. Published April 28, 2017. Accessed March 12, 2019.
2. Bergink V, Rasgon N, Wisner KL. Postpartum psychosis: madness, mania, and melancholia in motherhood. Am J Psychiatry. 2016;173(12):1179-1188.
3. Sit D, Rothschild AJ, Wisner KL. A review of postpartum psychosis. J Womens Health (Larchmt). 2006;15(4):352-368.
4. Kendell RE, Chalmers JC, Platz C. Epidemiology of puerperal psychoses. Br J Psychiatry. 1987;150(5):662-673.
5. Munk-Olsen T, Laursen TM, Mendelson T, et al. Risks and predictors of readmission for a mental disorder during the postpartum period. Arch Gen Psychiatry. 2009;66(2):189-195.
6. Bergink V, Burgerhout KM, Koorengevel KM, et al. Treatment of psychosis and mania in the postpartum period. Am J Psychiatry. 2015;172(2):115-123.
7. Wesseloo R, Kamperman AM, Munk-Olsen T, et al. Risk of postpartum relapse in bipolar disorder and postpartum psychosis: a systematic review and meta-analysis. Am J Psychiatry. 2015;173(2):117-127.
8. Wisner KL, Peindl K, Hanusa BH. Symptomatology of affective and psychotic illnesses related to childbearing. J Affect Disord. 1994;30(2):77-87.
9. Spinelli MG. Postpartum psychosis: detection of risk and management. Am J Psychiatry. 2009;166(4):405-408.
10. Fassier T, Guffon N, Acquaviva C, et al. Misdiagnosed postpartum psychosis revealing a late-onset urea cycle disorder. Am J Psychiatry. 2011;168(6):576-580.
11. Yu AYX, Moore FG. Paraneoplastic encephalitis presenting as postpartum psychosis. Psychosomatics. 2011;52(6):568-570.
12. Patil NJ, Yadav SS, Gokhale YA, et al. Primary hypoparathyroidism: psychosis in postpartum period. J Assoc Physicians India. 2010;58:506-508.
13. O’Hara MW, Schlechte JA, Lewis DA, et al. Prospective study of postpartum blues: biologic and psychosocial factors. Arch Gen Psychiatry. 1991;48(9):801-806.
14. Burt VK, Hendrick VC. Clinical manual of women’s mental health. Washington, DC. American Psychiatric Association Publishing; 2007:79-80.
15. Melzer-Brody S. Postpartum depression: what to tell patients who breast-feed. Current Psychiatry. 2008;7(5):87-95.
16. Alhusen JL, Gross D, Hayat MJ, et al. The role of mental health on maternal‐fetal attachment in low‐income women. J Obstet Gynecol Neonatal Nurs. 2012;41(6):E71-E81.
17. McLearn KT, Minkovitz CS, Strobino DM, et al. Maternal depressive symptoms at 2 to 4 months postpartum and early parenting practices. Arch Pediatr Adolesc Med. 2006;160(3):279-284.
18. Committee on Obstetric Practice. The American College of Obstetricians and Gynecologists Committee Opinion no. 630. Screening for perinatal depression. Obstet Gynecol. 2015;125(5):1268-1271.
19. Jennings KD, Ross S, Popper S. Thoughts of harming infants in depressed and nondepressed mothers. J Affect Disord. 1999;54(1-2):21-28.
20. Miller ES, Hoxha D, Wisner KL, et al. Obsessions and compulsions in postpartum women without obsessive compulsive disorder. J Womens Health. 2015;24(10):825-830.
21. Russell EJ, Fawcett JM, Mazmanian D. Risk of obsessive-compulsive disorder in pregnant and postpartum women: a meta-analysis. J Clin Psychiatry. 2013;74(4):377-385.
22. Zambaldi CF, Cantilino A, Montenegro AC, et al. Postpartum obsessive-compulsive disorder: prevalence and clinical characteristics. Compr Psychiatry. 2009;50(6):503-509.
23. Booth BD, Friedman SH, Curry S, et al. Obsessions of child murder: underrecognized manifestations of obsessive-compulsive disorder. J Am Acad Psychiatry Law. 2014;42(1):66-74.
24. Lindahl V, Pearson JL, Colpe L. Prevalence of suicidality during pregnancy and the postpartum. Arch Womens Ment Health. 2005;8(2):77-87.
25. Samandari G, Martin SL, Kupper LL, et al. Are pregnant and postpartum women: at increased risk for violent death? Suicide and homicide findings from North Carolina. Matern Child Health J. 2011;15(5):660-669.
26. Friedman SH, Sorrentino R. Commentary: postpartum psychosis, infanticide, and insanity—implications for forensic psychiatry. J Am Acad Psychiatry Law. 2012;40(3):326-332.
27. Friedman SH, Resnick PJ. Child murder by mothers: patterns and prevention. World Psychiatry. 2007;6(3):137-141.
28. Friedman SH, Hrouda DR, Holden CE, et al. Filicide-suicide: common factors in parents who kill their children and themselves. J Am Acad Psychiatry Law. 2005;33(4):496-504.
29. Chandra PS, Venkatasubramanian G, Thomas T. Infanticidal ideas and infanticidal behavior in Indian women with severe postpartum psychiatric disorders. J Nerv Ment Dis. 2002;190(7):457-461.
30. Jones I, Chandra PS, Dazzan P, et al. Bipolar disorder, affective psychosis, and schizophrenia in pregnancy and the post-partum period. Lancet. 2014;384(9956):1789-1799.
31. Friedman SH. Neonaticide. In: Friedman SH. Family murder: pathologies of love and hate. Washington, DC: American Psychiatric Association Publishing; 2018:53-67.
32. Meltzer-Brody S, Brandon AR, Pearson B, et al. Evaluating the clinical effectiveness of a specialized perinatal psychiatry inpatient unit. Arch Womens Ment Health. 2014;17(2):107-113.
33. Klinger G, Stahl B, Fusar-Poli P, et al. Antipsychotic drugs and breastfeeding. Pediatri Endocrinol Rev. 2013;10(3):308-317.
34. Focht A, Kellner CH. Electroconvulsive therapy (ECT) in the treatment of postpartum psychosis. J ECT. 2012;28(1):31-33.
35. Heron J, McGuinness M, Blackmore ER, et al. Early postpartum symptoms in puerperal psychosis. BJOG. 2008;115(3):348-353.
36. McEwan M, Friedman SH. Violence by parents against their children: reporting of maltreatment suspicions, child protection, and risk in mental illness. Psychiatr Clin North Am. 2016;39(4):691-700.
37. Centre of Perinatal Excellence. National Perinatal Mental Health Guideline. http://cope.org.au/about/review-of-new-perinatal-mental-health-guidelines/. Published October 27, 2017. Accessed November 22, 2018.
38. National Institute for Health and Care Excellence. Antenatal and postnatal mental health overview. https://pathways.nice.org.uk/pathways/antenatal-and-postnatal-mental-health. 2017. Accessed November 22, 2018.
39. Wesseloo R, Kamperman AM, Olsen TM, et al. Risk of postpartum relapse in bipolar disorder and postpartum psychosis: a systematic review and meta-analysis. Am J Psychiatry. 2016;173(2):117-127.
40. McKnight RF, Adida M, Budge K, et al. Lithium toxicity profile: a systematic review and meta-analysis. Lancet. 2012;379(9817):721-728.
41. Munk-Olsen T, Liu X, Viktorin A, et al. Maternal and infant outcomes associated with lithium use in pregnancy: an international collaborative meta-analysis of six cohort studies. Lancet Psychiatry. 2018;5(8):644-652.
42. Prakash C, Friedman SH, Moller-Olsen C, et al. Maternal and fetal outcomes after lamotrigine use in pregnancy: a retrospective analysis from an urban maternal mental health centre in New Zealand. Psychopharmacology Bull. 2016;46(2):63-69.
43. Wesseloo R, Liu X, Clark CT, et al. Risk of postpartum episodes in women with bipolar disorder after lamotrigine or lithium use in pregnancy: a population-based cohort study. J Affect Disord. 2017;218:394-397.
44. Dolk H, Wang H, Loane M, et al. Lamotrigine use in pregnancy and risk of orofacial cleft and other congenital anomalies. Neurology. 2016;86(18):1716-1725.
45. Diav-Citrin O, Shechtman S, Zvi N, et al. Is it safe to use lamotrigine during pregnancy? A prospective comparative observational study. Birth Defects Res. 2017;109(15):1196-1203.
46. Kong L, Zhou T, Wang B, et al. The risks associated with the use of lamotrigine during pregnancy. Int J Psychiatry Clin Pract. 2018;22(1):2-5.
47. Deligiannidis KM, Byatt N, Freeman MP. Pharmacotherapy for mood disorders in pregnancy: a review of pharmacokinetic changes and clinical recommendations for therapeutic drug monitoring. J Clin Psychopharmacol. 2014;34(2):244.
48. Bogen DL, Sit D, Genovese A, et al. Three cases of lithium exposure and exclusive breastfeeding. Arch Womens Ment Health. 2012;15(1):69-72.
COMBS: Feeling positive about negative symptoms of schizophrenia
Negative symptoms of schizophrenia—such as social withdrawal, avolition, avoidance, lack of spontaneity, anhedonia, poverty of speech, and blunted affect—often persist after successful treatment of positive symptoms, such as hallucinations and delusions.1 Negative symptoms can be debilitating and are associated with poor social and occupational outcomes, as well as cognitive dysfunction. Currently, treatments for negative symptoms are not nearly as effective as treatments for positive symptoms. The mnemonic COMBS can be used to easily recall 3 treatment modalities often used to address negative symptoms.
COgnitive-behavioral therapy
Cognitive-behavioral therapy (CBT) and other psychosocial therapies derived from it, such as social skills training, recovery-oriented cognitive therapy, motivation and enhancement therapy, and cognitive-behavioral social skills training (CBSST), have shown to be effective for treating negative symptoms.2 In a study of 149 patients with schizophrenia, CBSST reduced symptoms of avolition and apathy and improved functioning outcomes.2
Medications
Antipsychotics. Although second-generation antipsychotics (SGAs) were initially promising, accumulating clinical experience and research have shown that these agents have limited efficacy for treating negative symptoms.1 Unlike first-generation antipsychotics, SGAs do not cause affective blunting, and are effective at treating depressive symptoms; however, depressive symptoms can sometimes be difficult to distinguish from negative symptoms. Improvement of depressive symptoms observed with SGA treatment could be mistakenly interpreted as alleviation of negative symptoms; however, clinical trials that focused specifically on treating negative symptoms have found no specific efficacy of SGAs.1
Antidepressants. Although clinical trials and meta-analyses have had mixed results,1 antidepressants appear to be safe add-on treatments with small efficacy for negative symptoms.
Anticonvulsants have long been used as augmentation to antipsychotics for patients with treatment-resistant schizophrenia; however, there is no evidence that these medications can improve negative symptoms.1
Stimulants. There is no strong evidence that stimulants could be an efficacious treatment for negative symptoms.1
Other pharmacologic agents,1 such as acetylcholine-related medications, oxytocin, and medications with a mechanism of action that is related to an inflammatory response and immunologic pathways (ie, minocycline), are being evaluated for treating negative symptoms. Research into the efficacy of glutamate-related agents also appears to be continuing.1
Continue to: Brain Stimulation therapies
Brain Stimulation therapies
Transcranial magnetic stimulation (TMS), transdirect current stimulation (tDCS), vagus nerve stimulation, and deep brain stimulation have been evaluated for treating negative symptoms. A recent meta-analysis of randomized controlled trials comparing the effects of brain stimulation with sham interventions in patients with schizophrenia found that TMS and tDCS that targeted the left dorsolateral prefrontal cortex effectively reduced the severity of negative symptoms.3
The Table1-3 summarizes available treatments for negative symptoms of schizophrenia and their efficacies. Although research investigating the improvement of negative symptoms is currently insufficient, CBT-related therapies and antidepressants appear to be helpful. For more information, see “Treating negative symptoms of schizophrenia” (C

1. Remington G, Foussias G, Fervaha G, et al. Treating negative symptoms in schizophrenia: an update. Curr Treat Options Psychiatry. 2016;3:133-150.
2. Granholm E, Holden J, Worley M. Improvement in negative symptoms and functioning in cognitive-behavioral social skills training for schizophrenia: mediation by defeatist performance attitudes and asocial beliefs. Schizophr Bull. 2018;44(3):653-661.
3. Kennedy NI, Lee WH. Efficacy of non-invasive brain stimulation on the symptom dimensions of schizophrenia: a meta-analysis of randomized controlled trials. Eur Psychiatry. 2018;49:69-77.
Negative symptoms of schizophrenia—such as social withdrawal, avolition, avoidance, lack of spontaneity, anhedonia, poverty of speech, and blunted affect—often persist after successful treatment of positive symptoms, such as hallucinations and delusions.1 Negative symptoms can be debilitating and are associated with poor social and occupational outcomes, as well as cognitive dysfunction. Currently, treatments for negative symptoms are not nearly as effective as treatments for positive symptoms. The mnemonic COMBS can be used to easily recall 3 treatment modalities often used to address negative symptoms.
COgnitive-behavioral therapy
Cognitive-behavioral therapy (CBT) and other psychosocial therapies derived from it, such as social skills training, recovery-oriented cognitive therapy, motivation and enhancement therapy, and cognitive-behavioral social skills training (CBSST), have shown to be effective for treating negative symptoms.2 In a study of 149 patients with schizophrenia, CBSST reduced symptoms of avolition and apathy and improved functioning outcomes.2
Medications
Antipsychotics. Although second-generation antipsychotics (SGAs) were initially promising, accumulating clinical experience and research have shown that these agents have limited efficacy for treating negative symptoms.1 Unlike first-generation antipsychotics, SGAs do not cause affective blunting, and are effective at treating depressive symptoms; however, depressive symptoms can sometimes be difficult to distinguish from negative symptoms. Improvement of depressive symptoms observed with SGA treatment could be mistakenly interpreted as alleviation of negative symptoms; however, clinical trials that focused specifically on treating negative symptoms have found no specific efficacy of SGAs.1
Antidepressants. Although clinical trials and meta-analyses have had mixed results,1 antidepressants appear to be safe add-on treatments with small efficacy for negative symptoms.
Anticonvulsants have long been used as augmentation to antipsychotics for patients with treatment-resistant schizophrenia; however, there is no evidence that these medications can improve negative symptoms.1
Stimulants. There is no strong evidence that stimulants could be an efficacious treatment for negative symptoms.1
Other pharmacologic agents,1 such as acetylcholine-related medications, oxytocin, and medications with a mechanism of action that is related to an inflammatory response and immunologic pathways (ie, minocycline), are being evaluated for treating negative symptoms. Research into the efficacy of glutamate-related agents also appears to be continuing.1
Continue to: Brain Stimulation therapies
Brain Stimulation therapies
Transcranial magnetic stimulation (TMS), transdirect current stimulation (tDCS), vagus nerve stimulation, and deep brain stimulation have been evaluated for treating negative symptoms. A recent meta-analysis of randomized controlled trials comparing the effects of brain stimulation with sham interventions in patients with schizophrenia found that TMS and tDCS that targeted the left dorsolateral prefrontal cortex effectively reduced the severity of negative symptoms.3
The Table1-3 summarizes available treatments for negative symptoms of schizophrenia and their efficacies. Although research investigating the improvement of negative symptoms is currently insufficient, CBT-related therapies and antidepressants appear to be helpful. For more information, see “Treating negative symptoms of schizophrenia” (C

Negative symptoms of schizophrenia—such as social withdrawal, avolition, avoidance, lack of spontaneity, anhedonia, poverty of speech, and blunted affect—often persist after successful treatment of positive symptoms, such as hallucinations and delusions.1 Negative symptoms can be debilitating and are associated with poor social and occupational outcomes, as well as cognitive dysfunction. Currently, treatments for negative symptoms are not nearly as effective as treatments for positive symptoms. The mnemonic COMBS can be used to easily recall 3 treatment modalities often used to address negative symptoms.
COgnitive-behavioral therapy
Cognitive-behavioral therapy (CBT) and other psychosocial therapies derived from it, such as social skills training, recovery-oriented cognitive therapy, motivation and enhancement therapy, and cognitive-behavioral social skills training (CBSST), have shown to be effective for treating negative symptoms.2 In a study of 149 patients with schizophrenia, CBSST reduced symptoms of avolition and apathy and improved functioning outcomes.2
Medications
Antipsychotics. Although second-generation antipsychotics (SGAs) were initially promising, accumulating clinical experience and research have shown that these agents have limited efficacy for treating negative symptoms.1 Unlike first-generation antipsychotics, SGAs do not cause affective blunting, and are effective at treating depressive symptoms; however, depressive symptoms can sometimes be difficult to distinguish from negative symptoms. Improvement of depressive symptoms observed with SGA treatment could be mistakenly interpreted as alleviation of negative symptoms; however, clinical trials that focused specifically on treating negative symptoms have found no specific efficacy of SGAs.1
Antidepressants. Although clinical trials and meta-analyses have had mixed results,1 antidepressants appear to be safe add-on treatments with small efficacy for negative symptoms.
Anticonvulsants have long been used as augmentation to antipsychotics for patients with treatment-resistant schizophrenia; however, there is no evidence that these medications can improve negative symptoms.1
Stimulants. There is no strong evidence that stimulants could be an efficacious treatment for negative symptoms.1
Other pharmacologic agents,1 such as acetylcholine-related medications, oxytocin, and medications with a mechanism of action that is related to an inflammatory response and immunologic pathways (ie, minocycline), are being evaluated for treating negative symptoms. Research into the efficacy of glutamate-related agents also appears to be continuing.1
Continue to: Brain Stimulation therapies
Brain Stimulation therapies
Transcranial magnetic stimulation (TMS), transdirect current stimulation (tDCS), vagus nerve stimulation, and deep brain stimulation have been evaluated for treating negative symptoms. A recent meta-analysis of randomized controlled trials comparing the effects of brain stimulation with sham interventions in patients with schizophrenia found that TMS and tDCS that targeted the left dorsolateral prefrontal cortex effectively reduced the severity of negative symptoms.3
The Table1-3 summarizes available treatments for negative symptoms of schizophrenia and their efficacies. Although research investigating the improvement of negative symptoms is currently insufficient, CBT-related therapies and antidepressants appear to be helpful. For more information, see “Treating negative symptoms of schizophrenia” (C

1. Remington G, Foussias G, Fervaha G, et al. Treating negative symptoms in schizophrenia: an update. Curr Treat Options Psychiatry. 2016;3:133-150.
2. Granholm E, Holden J, Worley M. Improvement in negative symptoms and functioning in cognitive-behavioral social skills training for schizophrenia: mediation by defeatist performance attitudes and asocial beliefs. Schizophr Bull. 2018;44(3):653-661.
3. Kennedy NI, Lee WH. Efficacy of non-invasive brain stimulation on the symptom dimensions of schizophrenia: a meta-analysis of randomized controlled trials. Eur Psychiatry. 2018;49:69-77.
1. Remington G, Foussias G, Fervaha G, et al. Treating negative symptoms in schizophrenia: an update. Curr Treat Options Psychiatry. 2016;3:133-150.
2. Granholm E, Holden J, Worley M. Improvement in negative symptoms and functioning in cognitive-behavioral social skills training for schizophrenia: mediation by defeatist performance attitudes and asocial beliefs. Schizophr Bull. 2018;44(3):653-661.
3. Kennedy NI, Lee WH. Efficacy of non-invasive brain stimulation on the symptom dimensions of schizophrenia: a meta-analysis of randomized controlled trials. Eur Psychiatry. 2018;49:69-77.
Pimavanserin: A potentially safer alternative to clozapine for refractory hallucinations and delusions
Up to 30% of patients with schizophrenia do not respond to dopamine antagonists, which include all first- and second-generation antipsychotics. They are labeled as “treatment-resistant” if they have a partial response, or “treatment-refractory” if their hallucinations and/or delusions do not improve at all despite multiple trials of antipsychotics.
That’s why clozapine is considered a “lifesaver” for such patients, a last-resort medication that unshackles patients with refractory psychotic symptoms from the tyranny of auditory and/or visual hallucinations and the reality distortion of fixed false beliefs such as paranoid delusions.
Many long-suffering patients with refractory psychosis recover and return to their baseline, thanks to clozapine. In a past editorial, I discussed how one of my patients, Bethany, who had dropped out of college and became homeless for 4 years with refractory delusions and hallucinations, recovered completely when she received clozapine.1 She then returned to college, graduated with honors, and authored a book about her journey of recovery.2 She and I later established a nonprofit foundation we called CURESZ (Comprehensive Understanding via Research and Education in Schizophrenia), and assembled a panel of 80 clozapine experts across the country to provide access to clozapine for the hundreds of thousands of individuals with refractory psychosis who never received a trial of clozapine from their psychiatrists or psychiatric nurse practitioners. (Visit CURESZ.org for details.)
Bethany was very lucky to respond and recover completely, because only 40% of patients with refractory psychosis respond to clozapine. She does not mind having her blood drawn every week to measure her white blood cell count for early detection of potentially fatal agranulocytosis. Many refractory, often homeless patients with chronic schizophrenia refuse to have weekly phlebotomy and therefore are not treated with clozapine. Bethany was also fortunate to experience only 1 adverse effect of clozapine: extreme sedation that forced her to sleep up to 15 hours a day (this was reduced to 9 to 10 hours a day with adjunctive modafinil). Fortunately, she was spared the multiple other serious adverse effects of clozapine, which include excessive salivation, extreme weight gain, diabetes, hyperlipidemia, cardiomyopathy, pancreatitis, seizures, and ileus.3 Clozapine is also associated with sudden death more than any other antipsychotic agent.4
So, what can be done for patients with refractory hallucinations and delusions who are among the 60% who fail to respond to clozapine, or who experience intolerable adverse effects or safety problems, or who refuse to take clozapine and have their blood drawn every week? This is a desperately ill and seriously disabled group of patients who are deemed to be beyond the reach of medical intervention by psychiatry. They are often treated with various off-label medications as adjunctive therapy to clozapine, to which they failed to respond. This includes adding lamotrigine5 or benzoate,6 but none have been approved as an efficacious and safe monotherapy alternative to clozapine. So, what can be done for patients with refractory illness?
Enter pimavanserin. This new medication is an inverse agonist of serotonin 5-HT2A receptors and (to a lesser extent) serotonin 5-HT2C receptors. It was recently FDA-approved for treating the hallucinations and delusions of Parkinson’s disease psychosis,7 which is estimated to develop in up to 50% of individuals with Parkinson’s disease. It does not have any affinity to any dopamine receptors, which makes it an ideal antipsychotic for Parkinson’s disease, where any dopamine antagonism can worsen the motor symptoms (rigidity, hypokinesia, and tremors) associated with that movement disorder. Thus, pimavanserin became the first ever non-dopaminergic antipsychotic in the world and is indicated only for Parkinson’s disease psychosis.
Our clinical team made a serendipitous discovery about the efficacy of pimavanserin in patients with schizophrenia who failed to respond to clozapine therapy after several months at clinically adequate doses. Our findings were published online last month in the highly respected journal Schizophrenia Research.8 We reported the successful treatment with pimavanserin in 2 groups:
- patients who had not responded to clozapine received pimavanserin as an add-on to clozapine in doses of 34 mg/d, the same dose recommended for patients with Parkinson’s disease hallucinations and/or delusions.
- patients who had hallucinations and delusions that failed to respond to several non-clozapine antipsychotics received pimavanserin monotherapy instead of clozapine to avoid blood draws and serious adverse effects.
Continue to: Pimavanserin successfully treated...
Pimavanserin successfully treated the hallucinations and delusions of all 10 patients in both groups. Remission occurred within 1 month in most cases, and after 2 months in 1 patient. Those patients no longer required hospitalization as they did prior to taking pimavanserin, and they maintained their response for several months of follow-up. We were also pleased to note that most patients became more sociable and affable, with improved mood and affect, after their hallucinations and delusions disappeared with pimavanserin. We did have a few patients who did not respond to 34 mg/d of pimavanserin, and some who responded for several months but then showed signs of recurrence. We are considering increasing the dose to 68 mg/d in such patients because it is possible that a higher dose may be needed in some patients with refractory illness, who may vary in symptom severity or biology.
We are now planning to apply for a research grant to conduct a controlled trial to confirm our very encouraging clinical findings, and we hope other investigators will also conduct clinical trials in patients with refractory psychosis comparing pimavanserin with placebo or pimavanserin with clozapine in double-blind studies.
As a disclosure, our clinical findings were obtained without any knowledge of, or funding from, the company that makes pimavanserin (Acadia Pharmaceuticals Inc.). The company was informed of our findings only after our article was accepted for publication.
I hope this important finding of a potentially safer alternative to clozapine may address a major unmet need in psychiatry, involving the treatment of hundreds of thousands of patients with treatment-resistant or treatment-refractory psychosis, which includes patients with schizophrenia, schizoaffective disorder, or psychotic bipolar disorder.
To comment on this editorial or other topics of interest: henry.nasrallah@currentpsychiatry.com.
1. Nasrallah HA. Clozapine is a vastly underutilized, unique agent with multiple applications. Current Psychiatry. 2014;13(10):21, 24-25.
2. Yeiser B. Mind estranged: my journey from schizophrenia and homelessness to recovery. Seattle, WA: Amazon; 2014.
3. Raja M, Raja S. Clozapine safety, 40 years later. Curr Drug Saf. 2014;9(3):163-195.
4. Manu P, Kane JM, Corell CU. Sudden deaths in psychiatric patients. J Clin Psychiatry. 2011;72(7):936-941.
5. Tiihonen J, Wahlbeck K, Kiviniemi V. The efficacy of lamotrigine in clozapine-resistant schizophrenia: a systematic review and meta-analysis. Schizophrenia Research. 2009;109(1-3):10-14.
6. Lin CH, Lin CH, Chang YC, et al. Sodium benzoate, a D-amino acid oxidase inhibitor, added to clozapine for the treatment of schizophrenia: a randomized, double-blind, placebo-controlled trial. Biol Psychiatry. 2017;84(6):422-432.
7. Ballard C, Banister C, Khan Z, et al; ADP Investigators. Evaluation of the safety, tolerability, and efficacy of pimavanserin versus placebo in p atients with Alzheimer’s disease psychosis: a phase 2, randomised, placebo-controlled, double-blind study. Lancet Neurol. 2018;17(3):213-222.
8. Nasrallah HA, Fedora R, Morton R. Successful treatment of clozapine-nonresponsive refractory hallucinations and delusions with pimavanserin, a serotonin 5HT-2A receptor inverse agonist [Epub ahead of print March 2, 2019]. Schizophrenia Res. 2019. doi: 10.1016/j.schres.2019.02.018.
Up to 30% of patients with schizophrenia do not respond to dopamine antagonists, which include all first- and second-generation antipsychotics. They are labeled as “treatment-resistant” if they have a partial response, or “treatment-refractory” if their hallucinations and/or delusions do not improve at all despite multiple trials of antipsychotics.
That’s why clozapine is considered a “lifesaver” for such patients, a last-resort medication that unshackles patients with refractory psychotic symptoms from the tyranny of auditory and/or visual hallucinations and the reality distortion of fixed false beliefs such as paranoid delusions.
Many long-suffering patients with refractory psychosis recover and return to their baseline, thanks to clozapine. In a past editorial, I discussed how one of my patients, Bethany, who had dropped out of college and became homeless for 4 years with refractory delusions and hallucinations, recovered completely when she received clozapine.1 She then returned to college, graduated with honors, and authored a book about her journey of recovery.2 She and I later established a nonprofit foundation we called CURESZ (Comprehensive Understanding via Research and Education in Schizophrenia), and assembled a panel of 80 clozapine experts across the country to provide access to clozapine for the hundreds of thousands of individuals with refractory psychosis who never received a trial of clozapine from their psychiatrists or psychiatric nurse practitioners. (Visit CURESZ.org for details.)
Bethany was very lucky to respond and recover completely, because only 40% of patients with refractory psychosis respond to clozapine. She does not mind having her blood drawn every week to measure her white blood cell count for early detection of potentially fatal agranulocytosis. Many refractory, often homeless patients with chronic schizophrenia refuse to have weekly phlebotomy and therefore are not treated with clozapine. Bethany was also fortunate to experience only 1 adverse effect of clozapine: extreme sedation that forced her to sleep up to 15 hours a day (this was reduced to 9 to 10 hours a day with adjunctive modafinil). Fortunately, she was spared the multiple other serious adverse effects of clozapine, which include excessive salivation, extreme weight gain, diabetes, hyperlipidemia, cardiomyopathy, pancreatitis, seizures, and ileus.3 Clozapine is also associated with sudden death more than any other antipsychotic agent.4
So, what can be done for patients with refractory hallucinations and delusions who are among the 60% who fail to respond to clozapine, or who experience intolerable adverse effects or safety problems, or who refuse to take clozapine and have their blood drawn every week? This is a desperately ill and seriously disabled group of patients who are deemed to be beyond the reach of medical intervention by psychiatry. They are often treated with various off-label medications as adjunctive therapy to clozapine, to which they failed to respond. This includes adding lamotrigine5 or benzoate,6 but none have been approved as an efficacious and safe monotherapy alternative to clozapine. So, what can be done for patients with refractory illness?
Enter pimavanserin. This new medication is an inverse agonist of serotonin 5-HT2A receptors and (to a lesser extent) serotonin 5-HT2C receptors. It was recently FDA-approved for treating the hallucinations and delusions of Parkinson’s disease psychosis,7 which is estimated to develop in up to 50% of individuals with Parkinson’s disease. It does not have any affinity to any dopamine receptors, which makes it an ideal antipsychotic for Parkinson’s disease, where any dopamine antagonism can worsen the motor symptoms (rigidity, hypokinesia, and tremors) associated with that movement disorder. Thus, pimavanserin became the first ever non-dopaminergic antipsychotic in the world and is indicated only for Parkinson’s disease psychosis.
Our clinical team made a serendipitous discovery about the efficacy of pimavanserin in patients with schizophrenia who failed to respond to clozapine therapy after several months at clinically adequate doses. Our findings were published online last month in the highly respected journal Schizophrenia Research.8 We reported the successful treatment with pimavanserin in 2 groups:
- patients who had not responded to clozapine received pimavanserin as an add-on to clozapine in doses of 34 mg/d, the same dose recommended for patients with Parkinson’s disease hallucinations and/or delusions.
- patients who had hallucinations and delusions that failed to respond to several non-clozapine antipsychotics received pimavanserin monotherapy instead of clozapine to avoid blood draws and serious adverse effects.
Continue to: Pimavanserin successfully treated...
Pimavanserin successfully treated the hallucinations and delusions of all 10 patients in both groups. Remission occurred within 1 month in most cases, and after 2 months in 1 patient. Those patients no longer required hospitalization as they did prior to taking pimavanserin, and they maintained their response for several months of follow-up. We were also pleased to note that most patients became more sociable and affable, with improved mood and affect, after their hallucinations and delusions disappeared with pimavanserin. We did have a few patients who did not respond to 34 mg/d of pimavanserin, and some who responded for several months but then showed signs of recurrence. We are considering increasing the dose to 68 mg/d in such patients because it is possible that a higher dose may be needed in some patients with refractory illness, who may vary in symptom severity or biology.
We are now planning to apply for a research grant to conduct a controlled trial to confirm our very encouraging clinical findings, and we hope other investigators will also conduct clinical trials in patients with refractory psychosis comparing pimavanserin with placebo or pimavanserin with clozapine in double-blind studies.
As a disclosure, our clinical findings were obtained without any knowledge of, or funding from, the company that makes pimavanserin (Acadia Pharmaceuticals Inc.). The company was informed of our findings only after our article was accepted for publication.
I hope this important finding of a potentially safer alternative to clozapine may address a major unmet need in psychiatry, involving the treatment of hundreds of thousands of patients with treatment-resistant or treatment-refractory psychosis, which includes patients with schizophrenia, schizoaffective disorder, or psychotic bipolar disorder.
To comment on this editorial or other topics of interest: henry.nasrallah@currentpsychiatry.com.
Up to 30% of patients with schizophrenia do not respond to dopamine antagonists, which include all first- and second-generation antipsychotics. They are labeled as “treatment-resistant” if they have a partial response, or “treatment-refractory” if their hallucinations and/or delusions do not improve at all despite multiple trials of antipsychotics.
That’s why clozapine is considered a “lifesaver” for such patients, a last-resort medication that unshackles patients with refractory psychotic symptoms from the tyranny of auditory and/or visual hallucinations and the reality distortion of fixed false beliefs such as paranoid delusions.
Many long-suffering patients with refractory psychosis recover and return to their baseline, thanks to clozapine. In a past editorial, I discussed how one of my patients, Bethany, who had dropped out of college and became homeless for 4 years with refractory delusions and hallucinations, recovered completely when she received clozapine.1 She then returned to college, graduated with honors, and authored a book about her journey of recovery.2 She and I later established a nonprofit foundation we called CURESZ (Comprehensive Understanding via Research and Education in Schizophrenia), and assembled a panel of 80 clozapine experts across the country to provide access to clozapine for the hundreds of thousands of individuals with refractory psychosis who never received a trial of clozapine from their psychiatrists or psychiatric nurse practitioners. (Visit CURESZ.org for details.)
Bethany was very lucky to respond and recover completely, because only 40% of patients with refractory psychosis respond to clozapine. She does not mind having her blood drawn every week to measure her white blood cell count for early detection of potentially fatal agranulocytosis. Many refractory, often homeless patients with chronic schizophrenia refuse to have weekly phlebotomy and therefore are not treated with clozapine. Bethany was also fortunate to experience only 1 adverse effect of clozapine: extreme sedation that forced her to sleep up to 15 hours a day (this was reduced to 9 to 10 hours a day with adjunctive modafinil). Fortunately, she was spared the multiple other serious adverse effects of clozapine, which include excessive salivation, extreme weight gain, diabetes, hyperlipidemia, cardiomyopathy, pancreatitis, seizures, and ileus.3 Clozapine is also associated with sudden death more than any other antipsychotic agent.4
So, what can be done for patients with refractory hallucinations and delusions who are among the 60% who fail to respond to clozapine, or who experience intolerable adverse effects or safety problems, or who refuse to take clozapine and have their blood drawn every week? This is a desperately ill and seriously disabled group of patients who are deemed to be beyond the reach of medical intervention by psychiatry. They are often treated with various off-label medications as adjunctive therapy to clozapine, to which they failed to respond. This includes adding lamotrigine5 or benzoate,6 but none have been approved as an efficacious and safe monotherapy alternative to clozapine. So, what can be done for patients with refractory illness?
Enter pimavanserin. This new medication is an inverse agonist of serotonin 5-HT2A receptors and (to a lesser extent) serotonin 5-HT2C receptors. It was recently FDA-approved for treating the hallucinations and delusions of Parkinson’s disease psychosis,7 which is estimated to develop in up to 50% of individuals with Parkinson’s disease. It does not have any affinity to any dopamine receptors, which makes it an ideal antipsychotic for Parkinson’s disease, where any dopamine antagonism can worsen the motor symptoms (rigidity, hypokinesia, and tremors) associated with that movement disorder. Thus, pimavanserin became the first ever non-dopaminergic antipsychotic in the world and is indicated only for Parkinson’s disease psychosis.
Our clinical team made a serendipitous discovery about the efficacy of pimavanserin in patients with schizophrenia who failed to respond to clozapine therapy after several months at clinically adequate doses. Our findings were published online last month in the highly respected journal Schizophrenia Research.8 We reported the successful treatment with pimavanserin in 2 groups:
- patients who had not responded to clozapine received pimavanserin as an add-on to clozapine in doses of 34 mg/d, the same dose recommended for patients with Parkinson’s disease hallucinations and/or delusions.
- patients who had hallucinations and delusions that failed to respond to several non-clozapine antipsychotics received pimavanserin monotherapy instead of clozapine to avoid blood draws and serious adverse effects.
Continue to: Pimavanserin successfully treated...
Pimavanserin successfully treated the hallucinations and delusions of all 10 patients in both groups. Remission occurred within 1 month in most cases, and after 2 months in 1 patient. Those patients no longer required hospitalization as they did prior to taking pimavanserin, and they maintained their response for several months of follow-up. We were also pleased to note that most patients became more sociable and affable, with improved mood and affect, after their hallucinations and delusions disappeared with pimavanserin. We did have a few patients who did not respond to 34 mg/d of pimavanserin, and some who responded for several months but then showed signs of recurrence. We are considering increasing the dose to 68 mg/d in such patients because it is possible that a higher dose may be needed in some patients with refractory illness, who may vary in symptom severity or biology.
We are now planning to apply for a research grant to conduct a controlled trial to confirm our very encouraging clinical findings, and we hope other investigators will also conduct clinical trials in patients with refractory psychosis comparing pimavanserin with placebo or pimavanserin with clozapine in double-blind studies.
As a disclosure, our clinical findings were obtained without any knowledge of, or funding from, the company that makes pimavanserin (Acadia Pharmaceuticals Inc.). The company was informed of our findings only after our article was accepted for publication.
I hope this important finding of a potentially safer alternative to clozapine may address a major unmet need in psychiatry, involving the treatment of hundreds of thousands of patients with treatment-resistant or treatment-refractory psychosis, which includes patients with schizophrenia, schizoaffective disorder, or psychotic bipolar disorder.
To comment on this editorial or other topics of interest: henry.nasrallah@currentpsychiatry.com.
1. Nasrallah HA. Clozapine is a vastly underutilized, unique agent with multiple applications. Current Psychiatry. 2014;13(10):21, 24-25.
2. Yeiser B. Mind estranged: my journey from schizophrenia and homelessness to recovery. Seattle, WA: Amazon; 2014.
3. Raja M, Raja S. Clozapine safety, 40 years later. Curr Drug Saf. 2014;9(3):163-195.
4. Manu P, Kane JM, Corell CU. Sudden deaths in psychiatric patients. J Clin Psychiatry. 2011;72(7):936-941.
5. Tiihonen J, Wahlbeck K, Kiviniemi V. The efficacy of lamotrigine in clozapine-resistant schizophrenia: a systematic review and meta-analysis. Schizophrenia Research. 2009;109(1-3):10-14.
6. Lin CH, Lin CH, Chang YC, et al. Sodium benzoate, a D-amino acid oxidase inhibitor, added to clozapine for the treatment of schizophrenia: a randomized, double-blind, placebo-controlled trial. Biol Psychiatry. 2017;84(6):422-432.
7. Ballard C, Banister C, Khan Z, et al; ADP Investigators. Evaluation of the safety, tolerability, and efficacy of pimavanserin versus placebo in p atients with Alzheimer’s disease psychosis: a phase 2, randomised, placebo-controlled, double-blind study. Lancet Neurol. 2018;17(3):213-222.
8. Nasrallah HA, Fedora R, Morton R. Successful treatment of clozapine-nonresponsive refractory hallucinations and delusions with pimavanserin, a serotonin 5HT-2A receptor inverse agonist [Epub ahead of print March 2, 2019]. Schizophrenia Res. 2019. doi: 10.1016/j.schres.2019.02.018.
1. Nasrallah HA. Clozapine is a vastly underutilized, unique agent with multiple applications. Current Psychiatry. 2014;13(10):21, 24-25.
2. Yeiser B. Mind estranged: my journey from schizophrenia and homelessness to recovery. Seattle, WA: Amazon; 2014.
3. Raja M, Raja S. Clozapine safety, 40 years later. Curr Drug Saf. 2014;9(3):163-195.
4. Manu P, Kane JM, Corell CU. Sudden deaths in psychiatric patients. J Clin Psychiatry. 2011;72(7):936-941.
5. Tiihonen J, Wahlbeck K, Kiviniemi V. The efficacy of lamotrigine in clozapine-resistant schizophrenia: a systematic review and meta-analysis. Schizophrenia Research. 2009;109(1-3):10-14.
6. Lin CH, Lin CH, Chang YC, et al. Sodium benzoate, a D-amino acid oxidase inhibitor, added to clozapine for the treatment of schizophrenia: a randomized, double-blind, placebo-controlled trial. Biol Psychiatry. 2017;84(6):422-432.
7. Ballard C, Banister C, Khan Z, et al; ADP Investigators. Evaluation of the safety, tolerability, and efficacy of pimavanserin versus placebo in p atients with Alzheimer’s disease psychosis: a phase 2, randomised, placebo-controlled, double-blind study. Lancet Neurol. 2018;17(3):213-222.
8. Nasrallah HA, Fedora R, Morton R. Successful treatment of clozapine-nonresponsive refractory hallucinations and delusions with pimavanserin, a serotonin 5HT-2A receptor inverse agonist [Epub ahead of print March 2, 2019]. Schizophrenia Res. 2019. doi: 10.1016/j.schres.2019.02.018.
Atypical used in Parkinson’s lifts hallucinations, delusions in refractory schizophrenia
Pimavanserin (Nuplazid), an atypical antipsychotic approved to treat hallucinations and delusions in Parkinson’s disease, shows promise as a treatment for patients with refractory schizophrenia who fail to respond to clozapine, a retrospective study suggests.
“Within a month, sometimes 2 months, hallucinations and delusions that have persisted for years were completely gone,” said lead author Henry A. Nasrallah, MD, in an interview. The study was published in Schizophrenia Research.
Dr. Nasrallah and his colleagues launched the study in a bid to help “the most desperate group of patients” with schizophrenia – the 60% of those with refractory psychosis who do not respond to clozapine.
“This group of patients is so desperate that psychiatrists have used everything in our pharmacopeia,” said Dr. Nasrallah, the Sydney W. Souers Endowed Chair and professor and chairman of the department of psychiatry and behavioral neuroscience at Saint Louis University. “Nothing has been shown to work. We decided to give them this medication [pimavanserin], which was approved by the FDA [Food and Drug Administration] 2 years ago for hallucinations and delusions for Parkinson’s disease.”
For the new study, Dr. Nasrallah and his coauthors gave 34 mg/day of pimavanserin to 10 patients, aged 21-77 years, with schizophrenia or schizoaffective disorder and refractory hallucinations and delusions. The subjects, all of whom live in a residential group home, had either failed clozapine (n = 6) or failed several antipsychotics but had not yet received clozapine (n = 4).
The results, Dr. Nasrallah said, were remarkable. “Not only did they get relief from their delusions and hallucinations, but nursing staff reported they were much more sociable and affable, getting out of their rooms, and mixing and mingling. It seems to help them beyond suppressing delusions and hallucinations. It made them more sociable and pleasant.”
Patients were able to avoid blood tests and the “sometimes life-threatening side effects of clozapine,” he said. According to the study, no patients needed to discontinue treatment because of safety or tolerability.
However, pimavanserin is expensive. According to GoodRx.com, monthly prices for 60 tablets of 17 mg pimavanserin – equal to the daily dose in this study – run from $2,759 to $2,907 with a free coupon.
Should psychiatrists prescribe the drug now for treatment-resistant schizophrenia? “We use drugs off label all the time for patients who do not have any FDA-approved medication,” Dr. Nasrallah said. “Sometimes, off-label use in psychiatry is a necessity, because around 80% of DSM-5 disorders do not have any approved drugs at this time.”
“It would also be interesting to test pimavanserin in first-episode psychosis to identify a ‘serotonergic subtype’ of the schizophrenia syndrome but also to completely avoid the extrapyramidal side effects of dopamine antagonists, to which first-episode psychosis patients are especially susceptible.”
No outside funding was reported. Dr. Nasrallah reported advisory board and consultant and speaker’s bureau relationships with Acadia, Alkermes, Allergan, Janssen, Lundbeck, Neurocrine Biosciences, Otsuka Pharmaceutical, Sunovion, and Teva. Another author reported no disclosures, and a third author reported numerous disclosures.
SOURCE: Nasrallah HA et al. Schizophr Res. 2019 Mar 2. doi: 10/1016/j.schres.2019.02.018.
Pimavanserin (Nuplazid), an atypical antipsychotic approved to treat hallucinations and delusions in Parkinson’s disease, shows promise as a treatment for patients with refractory schizophrenia who fail to respond to clozapine, a retrospective study suggests.
“Within a month, sometimes 2 months, hallucinations and delusions that have persisted for years were completely gone,” said lead author Henry A. Nasrallah, MD, in an interview. The study was published in Schizophrenia Research.
Dr. Nasrallah and his colleagues launched the study in a bid to help “the most desperate group of patients” with schizophrenia – the 60% of those with refractory psychosis who do not respond to clozapine.
“This group of patients is so desperate that psychiatrists have used everything in our pharmacopeia,” said Dr. Nasrallah, the Sydney W. Souers Endowed Chair and professor and chairman of the department of psychiatry and behavioral neuroscience at Saint Louis University. “Nothing has been shown to work. We decided to give them this medication [pimavanserin], which was approved by the FDA [Food and Drug Administration] 2 years ago for hallucinations and delusions for Parkinson’s disease.”
For the new study, Dr. Nasrallah and his coauthors gave 34 mg/day of pimavanserin to 10 patients, aged 21-77 years, with schizophrenia or schizoaffective disorder and refractory hallucinations and delusions. The subjects, all of whom live in a residential group home, had either failed clozapine (n = 6) or failed several antipsychotics but had not yet received clozapine (n = 4).
The results, Dr. Nasrallah said, were remarkable. “Not only did they get relief from their delusions and hallucinations, but nursing staff reported they were much more sociable and affable, getting out of their rooms, and mixing and mingling. It seems to help them beyond suppressing delusions and hallucinations. It made them more sociable and pleasant.”
Patients were able to avoid blood tests and the “sometimes life-threatening side effects of clozapine,” he said. According to the study, no patients needed to discontinue treatment because of safety or tolerability.
However, pimavanserin is expensive. According to GoodRx.com, monthly prices for 60 tablets of 17 mg pimavanserin – equal to the daily dose in this study – run from $2,759 to $2,907 with a free coupon.
Should psychiatrists prescribe the drug now for treatment-resistant schizophrenia? “We use drugs off label all the time for patients who do not have any FDA-approved medication,” Dr. Nasrallah said. “Sometimes, off-label use in psychiatry is a necessity, because around 80% of DSM-5 disorders do not have any approved drugs at this time.”
“It would also be interesting to test pimavanserin in first-episode psychosis to identify a ‘serotonergic subtype’ of the schizophrenia syndrome but also to completely avoid the extrapyramidal side effects of dopamine antagonists, to which first-episode psychosis patients are especially susceptible.”
No outside funding was reported. Dr. Nasrallah reported advisory board and consultant and speaker’s bureau relationships with Acadia, Alkermes, Allergan, Janssen, Lundbeck, Neurocrine Biosciences, Otsuka Pharmaceutical, Sunovion, and Teva. Another author reported no disclosures, and a third author reported numerous disclosures.
SOURCE: Nasrallah HA et al. Schizophr Res. 2019 Mar 2. doi: 10/1016/j.schres.2019.02.018.
Pimavanserin (Nuplazid), an atypical antipsychotic approved to treat hallucinations and delusions in Parkinson’s disease, shows promise as a treatment for patients with refractory schizophrenia who fail to respond to clozapine, a retrospective study suggests.
“Within a month, sometimes 2 months, hallucinations and delusions that have persisted for years were completely gone,” said lead author Henry A. Nasrallah, MD, in an interview. The study was published in Schizophrenia Research.
Dr. Nasrallah and his colleagues launched the study in a bid to help “the most desperate group of patients” with schizophrenia – the 60% of those with refractory psychosis who do not respond to clozapine.
“This group of patients is so desperate that psychiatrists have used everything in our pharmacopeia,” said Dr. Nasrallah, the Sydney W. Souers Endowed Chair and professor and chairman of the department of psychiatry and behavioral neuroscience at Saint Louis University. “Nothing has been shown to work. We decided to give them this medication [pimavanserin], which was approved by the FDA [Food and Drug Administration] 2 years ago for hallucinations and delusions for Parkinson’s disease.”
For the new study, Dr. Nasrallah and his coauthors gave 34 mg/day of pimavanserin to 10 patients, aged 21-77 years, with schizophrenia or schizoaffective disorder and refractory hallucinations and delusions. The subjects, all of whom live in a residential group home, had either failed clozapine (n = 6) or failed several antipsychotics but had not yet received clozapine (n = 4).
The results, Dr. Nasrallah said, were remarkable. “Not only did they get relief from their delusions and hallucinations, but nursing staff reported they were much more sociable and affable, getting out of their rooms, and mixing and mingling. It seems to help them beyond suppressing delusions and hallucinations. It made them more sociable and pleasant.”
Patients were able to avoid blood tests and the “sometimes life-threatening side effects of clozapine,” he said. According to the study, no patients needed to discontinue treatment because of safety or tolerability.
However, pimavanserin is expensive. According to GoodRx.com, monthly prices for 60 tablets of 17 mg pimavanserin – equal to the daily dose in this study – run from $2,759 to $2,907 with a free coupon.
Should psychiatrists prescribe the drug now for treatment-resistant schizophrenia? “We use drugs off label all the time for patients who do not have any FDA-approved medication,” Dr. Nasrallah said. “Sometimes, off-label use in psychiatry is a necessity, because around 80% of DSM-5 disorders do not have any approved drugs at this time.”
“It would also be interesting to test pimavanserin in first-episode psychosis to identify a ‘serotonergic subtype’ of the schizophrenia syndrome but also to completely avoid the extrapyramidal side effects of dopamine antagonists, to which first-episode psychosis patients are especially susceptible.”
No outside funding was reported. Dr. Nasrallah reported advisory board and consultant and speaker’s bureau relationships with Acadia, Alkermes, Allergan, Janssen, Lundbeck, Neurocrine Biosciences, Otsuka Pharmaceutical, Sunovion, and Teva. Another author reported no disclosures, and a third author reported numerous disclosures.
SOURCE: Nasrallah HA et al. Schizophr Res. 2019 Mar 2. doi: 10/1016/j.schres.2019.02.018.
FROM SCHIZOPHRENIA RESEARCH
Amphetamine tied to higher risk of new-onset psychosis than methylphenidate
Greater risk applies only to adolescents, young adults with ADHD treated in primary care
Adolescents and young adults with ADHD who start on amphetamine might have twice the risk of developing new-onset psychosis as do those who start on methylphenidate, a cohort study of more than 220,000 patients suggests.
“The percentage of patients who had a psychotic episode was 0.10% among patients who received methylphenidate and 0.21% among patients who received amphetamine, reported Lauren V. Moran, MD, of the division of pharmacoepidemiology and pharmacoeconomics at Brigham and Women’s Hospital in Boston and her colleagues. The study was published by the New England Journal of Medicine.
(aged 13-25 years) with ADHD between January 2004 and September 2015 who were prescribed methylphenidate or amphetamine (both 110,923 patients; 143,286 total person-years of follow-up). They looked for an ICD-9 or ICD-10 code for new-onset psychosis followed by a prescription for an antipsychotic medication the same day or within 60 days of the psychosis diagnosis. Hazard ratios were calculated by matching patients taking methylphenidate with patients taking amphetamine across both databases and calculating the incidence rate of psychosis in each group.
The researchers found 343 new cases of psychosis overall, with an incidence of 2.4 cases per 1,000 person-years. There were 106 episodes of psychosis among patients receiving methylphenidate (0.10%) and 237 new cases among patients receiving amphetamine (0.21%). There was an incidence rate of 1.78 cases per 1,000 person-years for methylphenidate patients and 2.83 cases per 1,000 person-years for amphetamine patients. Across both databases, the pooled hazard ratio for amphetamine use and new-onset psychosis, compared with matched patients, was 1.65 (95% confidence interval, 1.31-2.09).
“The attribution of the higher risk of psychosis to amphetamine use was supported by negative control outcome analyses, which showed that there was no difference in the risk of other psychiatric events between the two stimulant groups,” Dr. Moran and her colleagues reported. “The different biologic mechanisms of methylphenidate and amphetamine activity on neurotransmitters could explain our findings.”
Patients who were prescribed amphetamine by family medicine physicians, internists, and pediatricians were at a higher risk of developing psychosis. That risk, however, did not extend to patients prescribed amphetamine by psychiatrists, the researchers said.
“Psychosis may develop in these patients regardless of stimulant treatment. Alternatively, psychiatrists may prescribe amphetamine more cautiously than other providers and may screen for risk factors for psychosis,” Dr. Moran and her colleagues wrote.
The researchers said the study was limited by unmeasured confounders, such as substance or stimulant misuse; the rate of diversion for amphetamine; and lack of information on race, gender, or socioeconomic status. In addition, they noted, the results could not be generalized to patients with public insurance or no insurance, “which disproportionately applies to patients who are black or Hispanic.”
Dr. Moran reported receiving grants from National Institute of Mental Health (NIMH). The other authors reported grants, personal fees, and other relationships with several entities, including Boehringer Ingelheim, the Food and Drug Administration, the NIMH, and Takeda.
SOURCE: Moran LV et al. N Engl J Med. 2019. doi: 10.1056/NEJMoa1813751.
The findings by Moran et al. are consistent with other randomized controlled trials that suggest a better safety profile for methylphenidate over amphetamine. But the data cannot determine causality in this patient population, Samuele Cortese, MD, PhD, wrote in a related editorial.
“The findings of the current study should not be considered definitive. Observational studies such as this one can provide information on uncommon adverse events in real-world clinical practice that are challenging to assess in randomized trials performed over brief periods,” he said. “However, even sophisticated approaches, such as the ones used in this study to address possible biases, do not have the advantages of randomized trials in excluding confounding factors.”
It is still unclear why some patients developed psychosis, such as in cases of patients with stimulant use and had a “low” or “high” vulnerability to developing psychosis after exposure. The lack of association between psychosis and prescribing amphetamines among psychiatrists also might indicate that those clinicians identified risk factors in patients that predicted the development of psychosis and thus avoided prescribing amphetamines to these patients, he said.
“Currently, it is not possible to predict which patients will have psychotic episodes after stimulant treatment,” Dr. Cortese concluded. “Perhaps techniques such as machine learning applied to large data sets from randomized trials, combined with observational data, will provide predictors at the individual patient level.”
Dr. Cortese is affiliated with the Center for Innovation in Mental Health at the University of Southampton (England). These comments summarize his accompanying editorial (N Engl J Med. 2019. doi: 10.1056/NEJMe1900887 ). He reported nonfinancial relationships with the Association for Child and Adolescent Central Health and the Healthcare Convention & Exhibitors Association.
Greater risk applies only to adolescents, young adults with ADHD treated in primary care
Greater risk applies only to adolescents, young adults with ADHD treated in primary care
The findings by Moran et al. are consistent with other randomized controlled trials that suggest a better safety profile for methylphenidate over amphetamine. But the data cannot determine causality in this patient population, Samuele Cortese, MD, PhD, wrote in a related editorial.
“The findings of the current study should not be considered definitive. Observational studies such as this one can provide information on uncommon adverse events in real-world clinical practice that are challenging to assess in randomized trials performed over brief periods,” he said. “However, even sophisticated approaches, such as the ones used in this study to address possible biases, do not have the advantages of randomized trials in excluding confounding factors.”
It is still unclear why some patients developed psychosis, such as in cases of patients with stimulant use and had a “low” or “high” vulnerability to developing psychosis after exposure. The lack of association between psychosis and prescribing amphetamines among psychiatrists also might indicate that those clinicians identified risk factors in patients that predicted the development of psychosis and thus avoided prescribing amphetamines to these patients, he said.
“Currently, it is not possible to predict which patients will have psychotic episodes after stimulant treatment,” Dr. Cortese concluded. “Perhaps techniques such as machine learning applied to large data sets from randomized trials, combined with observational data, will provide predictors at the individual patient level.”
Dr. Cortese is affiliated with the Center for Innovation in Mental Health at the University of Southampton (England). These comments summarize his accompanying editorial (N Engl J Med. 2019. doi: 10.1056/NEJMe1900887 ). He reported nonfinancial relationships with the Association for Child and Adolescent Central Health and the Healthcare Convention & Exhibitors Association.
The findings by Moran et al. are consistent with other randomized controlled trials that suggest a better safety profile for methylphenidate over amphetamine. But the data cannot determine causality in this patient population, Samuele Cortese, MD, PhD, wrote in a related editorial.
“The findings of the current study should not be considered definitive. Observational studies such as this one can provide information on uncommon adverse events in real-world clinical practice that are challenging to assess in randomized trials performed over brief periods,” he said. “However, even sophisticated approaches, such as the ones used in this study to address possible biases, do not have the advantages of randomized trials in excluding confounding factors.”
It is still unclear why some patients developed psychosis, such as in cases of patients with stimulant use and had a “low” or “high” vulnerability to developing psychosis after exposure. The lack of association between psychosis and prescribing amphetamines among psychiatrists also might indicate that those clinicians identified risk factors in patients that predicted the development of psychosis and thus avoided prescribing amphetamines to these patients, he said.
“Currently, it is not possible to predict which patients will have psychotic episodes after stimulant treatment,” Dr. Cortese concluded. “Perhaps techniques such as machine learning applied to large data sets from randomized trials, combined with observational data, will provide predictors at the individual patient level.”
Dr. Cortese is affiliated with the Center for Innovation in Mental Health at the University of Southampton (England). These comments summarize his accompanying editorial (N Engl J Med. 2019. doi: 10.1056/NEJMe1900887 ). He reported nonfinancial relationships with the Association for Child and Adolescent Central Health and the Healthcare Convention & Exhibitors Association.
Adolescents and young adults with ADHD who start on amphetamine might have twice the risk of developing new-onset psychosis as do those who start on methylphenidate, a cohort study of more than 220,000 patients suggests.
“The percentage of patients who had a psychotic episode was 0.10% among patients who received methylphenidate and 0.21% among patients who received amphetamine, reported Lauren V. Moran, MD, of the division of pharmacoepidemiology and pharmacoeconomics at Brigham and Women’s Hospital in Boston and her colleagues. The study was published by the New England Journal of Medicine.
(aged 13-25 years) with ADHD between January 2004 and September 2015 who were prescribed methylphenidate or amphetamine (both 110,923 patients; 143,286 total person-years of follow-up). They looked for an ICD-9 or ICD-10 code for new-onset psychosis followed by a prescription for an antipsychotic medication the same day or within 60 days of the psychosis diagnosis. Hazard ratios were calculated by matching patients taking methylphenidate with patients taking amphetamine across both databases and calculating the incidence rate of psychosis in each group.
The researchers found 343 new cases of psychosis overall, with an incidence of 2.4 cases per 1,000 person-years. There were 106 episodes of psychosis among patients receiving methylphenidate (0.10%) and 237 new cases among patients receiving amphetamine (0.21%). There was an incidence rate of 1.78 cases per 1,000 person-years for methylphenidate patients and 2.83 cases per 1,000 person-years for amphetamine patients. Across both databases, the pooled hazard ratio for amphetamine use and new-onset psychosis, compared with matched patients, was 1.65 (95% confidence interval, 1.31-2.09).
“The attribution of the higher risk of psychosis to amphetamine use was supported by negative control outcome analyses, which showed that there was no difference in the risk of other psychiatric events between the two stimulant groups,” Dr. Moran and her colleagues reported. “The different biologic mechanisms of methylphenidate and amphetamine activity on neurotransmitters could explain our findings.”
Patients who were prescribed amphetamine by family medicine physicians, internists, and pediatricians were at a higher risk of developing psychosis. That risk, however, did not extend to patients prescribed amphetamine by psychiatrists, the researchers said.
“Psychosis may develop in these patients regardless of stimulant treatment. Alternatively, psychiatrists may prescribe amphetamine more cautiously than other providers and may screen for risk factors for psychosis,” Dr. Moran and her colleagues wrote.
The researchers said the study was limited by unmeasured confounders, such as substance or stimulant misuse; the rate of diversion for amphetamine; and lack of information on race, gender, or socioeconomic status. In addition, they noted, the results could not be generalized to patients with public insurance or no insurance, “which disproportionately applies to patients who are black or Hispanic.”
Dr. Moran reported receiving grants from National Institute of Mental Health (NIMH). The other authors reported grants, personal fees, and other relationships with several entities, including Boehringer Ingelheim, the Food and Drug Administration, the NIMH, and Takeda.
SOURCE: Moran LV et al. N Engl J Med. 2019. doi: 10.1056/NEJMoa1813751.
Adolescents and young adults with ADHD who start on amphetamine might have twice the risk of developing new-onset psychosis as do those who start on methylphenidate, a cohort study of more than 220,000 patients suggests.
“The percentage of patients who had a psychotic episode was 0.10% among patients who received methylphenidate and 0.21% among patients who received amphetamine, reported Lauren V. Moran, MD, of the division of pharmacoepidemiology and pharmacoeconomics at Brigham and Women’s Hospital in Boston and her colleagues. The study was published by the New England Journal of Medicine.
(aged 13-25 years) with ADHD between January 2004 and September 2015 who were prescribed methylphenidate or amphetamine (both 110,923 patients; 143,286 total person-years of follow-up). They looked for an ICD-9 or ICD-10 code for new-onset psychosis followed by a prescription for an antipsychotic medication the same day or within 60 days of the psychosis diagnosis. Hazard ratios were calculated by matching patients taking methylphenidate with patients taking amphetamine across both databases and calculating the incidence rate of psychosis in each group.
The researchers found 343 new cases of psychosis overall, with an incidence of 2.4 cases per 1,000 person-years. There were 106 episodes of psychosis among patients receiving methylphenidate (0.10%) and 237 new cases among patients receiving amphetamine (0.21%). There was an incidence rate of 1.78 cases per 1,000 person-years for methylphenidate patients and 2.83 cases per 1,000 person-years for amphetamine patients. Across both databases, the pooled hazard ratio for amphetamine use and new-onset psychosis, compared with matched patients, was 1.65 (95% confidence interval, 1.31-2.09).
“The attribution of the higher risk of psychosis to amphetamine use was supported by negative control outcome analyses, which showed that there was no difference in the risk of other psychiatric events between the two stimulant groups,” Dr. Moran and her colleagues reported. “The different biologic mechanisms of methylphenidate and amphetamine activity on neurotransmitters could explain our findings.”
Patients who were prescribed amphetamine by family medicine physicians, internists, and pediatricians were at a higher risk of developing psychosis. That risk, however, did not extend to patients prescribed amphetamine by psychiatrists, the researchers said.
“Psychosis may develop in these patients regardless of stimulant treatment. Alternatively, psychiatrists may prescribe amphetamine more cautiously than other providers and may screen for risk factors for psychosis,” Dr. Moran and her colleagues wrote.
The researchers said the study was limited by unmeasured confounders, such as substance or stimulant misuse; the rate of diversion for amphetamine; and lack of information on race, gender, or socioeconomic status. In addition, they noted, the results could not be generalized to patients with public insurance or no insurance, “which disproportionately applies to patients who are black or Hispanic.”
Dr. Moran reported receiving grants from National Institute of Mental Health (NIMH). The other authors reported grants, personal fees, and other relationships with several entities, including Boehringer Ingelheim, the Food and Drug Administration, the NIMH, and Takeda.
SOURCE: Moran LV et al. N Engl J Med. 2019. doi: 10.1056/NEJMoa1813751.
Cannabis use, potency linked to psychotic disorder risk
Daily cannabis use, particularly high-potency cannabis, might be a significant contributor to the incidence of psychotic disorder, results of a multicenter, case-control study suggest.
“We provide the first direct evidence that cannabis use has an effect on variation in the incidence of psychotic disorders,” Marta Di Forti, PhD, and her coauthors wrote in the Lancet.
In the study, Dr. Di Forti and her coauthors looked at cannabis use in 901 patients presenting with a first psychotic episode to one of 11 sites across Europe and Brazil, compared with 1,237 population controls from the same locations. They found that daily cannabis users had more than threefold higher odds of psychotic disorder, compared with individuals who had never used cannabis (odds ratio, 3.2; P less than .0001), even after adjusting for sociodemographic factors and use of tobacco, stimulants, ketamine, and hallucinogenics.
Those who reported using high-potency cannabis (delta 9-tetrahydrocannabinol greater than or equal to 10%) – also showed a significant 60% increase in the odds of psychotic disorder, compared with never-users, which decreased slightly to 50% after controlling for daily use.
“The large sample size and the different types of cannabis available across Europe have allowed us to report that the dose-response relationship characterizing the association between cannabis use and psychosis reflects not only the use of high-potency cannabis but also the daily use of types with an amount of THC consistent with more traditional varieties,” wrote Dr. Di Forti, of the Social, Genetic, and Developmental Psychiatry Centre at King’s College London, and her coauthors.
When the authors looked at the population-attributable fractions, they calculated that 12.2% of cases of first-episode psychosis would be avoided if high-potency cannabis were not available.
(P = .0122), while those who started using high-potency cannabis at that age had more than a doubling of risk (OR, 2.3).
Similarly, those who used high-potency cannabis on a daily basis had nearly fivefold higher odds of psychotic disorder, compared with never-users, while daily users of low-potency had a 2.2-fold increase in risk.
Researchers also examined patterns of cannabis use and psychotic disorder across the 11 sites, which included Amsterdam, London; Cambridge, England; Madrid; Palermo, Italy; Paris; and Ribeirão Preto, Brazil.
They noted that there were significant variations in the incidence of psychotic disorder across the study sites, and that those variations correlated with the prevalence of daily cannabis use.
London and Amsterdam, where daily use was the most common, had the highest adjusted incidence rates of psychotic disorder (45.7 cases per 100,000 person-years in London and 37.9 per 100,000 person-years in Amsterdam). In contrast, the incidence in Bologna, Italy – where daily use was less frequent – was half that of London.
They estimated that 43% of new cases of psychotic disorder in Amsterdam were attributable to daily use of cannabis, and 50.3% were attributable to high-potency cannabis, compared with 1.2% and 2.3% of cases in Puy de Dôme in France.
“Use of high-potency cannabis was a strong predictor of psychotic disorder in Amsterdam, London, and Paris, where high-potency cannabis was widely available, by contrast with sites such as Palermo where this type was not yet available,” the authors wrote. “Our results show that, in areas where daily use and use of high-potency cannabis are more prevalent in the general population, there is an excess of cases of psychotic disorder.”
The authors did point out that the study relied on self-reported cannabis use, rather than biological sampling measures. But previous studies have shown self-reported use to be a reliable measure, they said.
“Education is needed to inform the public about the mental health hazards of regular use of high-potency cannabis, which is becoming increasingly available worldwide,” they wrote.
The study was supported by the Medical Research Council, the European Community’s Seventh Framework Program, the São Paulo Research Foundation, the National Institute for Health Research Biomedical Research Centre, and the Wellcome Trust. Five authors declared personal fees and grants from the pharmaceutical industry. No other conflicts of interest were declared.
SOURCE: Di Forti M et al. Lancet. 2019 Mar 19. doi: 10.1016/S2215-0366(19)30048-3.
Epidemiologic and experimental studies generally have established a link between heavy cannabis use and psychosis. However, a long-running issue has been that, while cannabis use has increased in some populations, the rates of psychosis have not necessarily done the same. The results of this study go against that, suggesting that differing rates and intensity of cannabis use across Europe appear to correlate with differing rates of psychosis.
This does not necessarily imply causality. For example, genetic studies suggest that individuals predisposed to psychosis also may have a predisposition to use cannabis. Another possibility is that subclinical mental health issues existed in those participants before the start of cannabis use. The challenge, therefore, remains to identify which individuals are most at risk from psychosis related to cannabis use, and to develop strategies aimed at mitigating this risk.
Suzanne H. Gage, PhD, is affiliated with the department of psychological sciences at the University of Liverpool in England. These comments are adapted from an accompanying editorial (Lancet. 2019 Mar 19. doi: 10.1016/ S2215-0366[19]30086-0). No conflicts of interest were declared.
Epidemiologic and experimental studies generally have established a link between heavy cannabis use and psychosis. However, a long-running issue has been that, while cannabis use has increased in some populations, the rates of psychosis have not necessarily done the same. The results of this study go against that, suggesting that differing rates and intensity of cannabis use across Europe appear to correlate with differing rates of psychosis.
This does not necessarily imply causality. For example, genetic studies suggest that individuals predisposed to psychosis also may have a predisposition to use cannabis. Another possibility is that subclinical mental health issues existed in those participants before the start of cannabis use. The challenge, therefore, remains to identify which individuals are most at risk from psychosis related to cannabis use, and to develop strategies aimed at mitigating this risk.
Suzanne H. Gage, PhD, is affiliated with the department of psychological sciences at the University of Liverpool in England. These comments are adapted from an accompanying editorial (Lancet. 2019 Mar 19. doi: 10.1016/ S2215-0366[19]30086-0). No conflicts of interest were declared.
Epidemiologic and experimental studies generally have established a link between heavy cannabis use and psychosis. However, a long-running issue has been that, while cannabis use has increased in some populations, the rates of psychosis have not necessarily done the same. The results of this study go against that, suggesting that differing rates and intensity of cannabis use across Europe appear to correlate with differing rates of psychosis.
This does not necessarily imply causality. For example, genetic studies suggest that individuals predisposed to psychosis also may have a predisposition to use cannabis. Another possibility is that subclinical mental health issues existed in those participants before the start of cannabis use. The challenge, therefore, remains to identify which individuals are most at risk from psychosis related to cannabis use, and to develop strategies aimed at mitigating this risk.
Suzanne H. Gage, PhD, is affiliated with the department of psychological sciences at the University of Liverpool in England. These comments are adapted from an accompanying editorial (Lancet. 2019 Mar 19. doi: 10.1016/ S2215-0366[19]30086-0). No conflicts of interest were declared.
Daily cannabis use, particularly high-potency cannabis, might be a significant contributor to the incidence of psychotic disorder, results of a multicenter, case-control study suggest.
“We provide the first direct evidence that cannabis use has an effect on variation in the incidence of psychotic disorders,” Marta Di Forti, PhD, and her coauthors wrote in the Lancet.
In the study, Dr. Di Forti and her coauthors looked at cannabis use in 901 patients presenting with a first psychotic episode to one of 11 sites across Europe and Brazil, compared with 1,237 population controls from the same locations. They found that daily cannabis users had more than threefold higher odds of psychotic disorder, compared with individuals who had never used cannabis (odds ratio, 3.2; P less than .0001), even after adjusting for sociodemographic factors and use of tobacco, stimulants, ketamine, and hallucinogenics.
Those who reported using high-potency cannabis (delta 9-tetrahydrocannabinol greater than or equal to 10%) – also showed a significant 60% increase in the odds of psychotic disorder, compared with never-users, which decreased slightly to 50% after controlling for daily use.
“The large sample size and the different types of cannabis available across Europe have allowed us to report that the dose-response relationship characterizing the association between cannabis use and psychosis reflects not only the use of high-potency cannabis but also the daily use of types with an amount of THC consistent with more traditional varieties,” wrote Dr. Di Forti, of the Social, Genetic, and Developmental Psychiatry Centre at King’s College London, and her coauthors.
When the authors looked at the population-attributable fractions, they calculated that 12.2% of cases of first-episode psychosis would be avoided if high-potency cannabis were not available.
(P = .0122), while those who started using high-potency cannabis at that age had more than a doubling of risk (OR, 2.3).
Similarly, those who used high-potency cannabis on a daily basis had nearly fivefold higher odds of psychotic disorder, compared with never-users, while daily users of low-potency had a 2.2-fold increase in risk.
Researchers also examined patterns of cannabis use and psychotic disorder across the 11 sites, which included Amsterdam, London; Cambridge, England; Madrid; Palermo, Italy; Paris; and Ribeirão Preto, Brazil.
They noted that there were significant variations in the incidence of psychotic disorder across the study sites, and that those variations correlated with the prevalence of daily cannabis use.
London and Amsterdam, where daily use was the most common, had the highest adjusted incidence rates of psychotic disorder (45.7 cases per 100,000 person-years in London and 37.9 per 100,000 person-years in Amsterdam). In contrast, the incidence in Bologna, Italy – where daily use was less frequent – was half that of London.
They estimated that 43% of new cases of psychotic disorder in Amsterdam were attributable to daily use of cannabis, and 50.3% were attributable to high-potency cannabis, compared with 1.2% and 2.3% of cases in Puy de Dôme in France.
“Use of high-potency cannabis was a strong predictor of psychotic disorder in Amsterdam, London, and Paris, where high-potency cannabis was widely available, by contrast with sites such as Palermo where this type was not yet available,” the authors wrote. “Our results show that, in areas where daily use and use of high-potency cannabis are more prevalent in the general population, there is an excess of cases of psychotic disorder.”
The authors did point out that the study relied on self-reported cannabis use, rather than biological sampling measures. But previous studies have shown self-reported use to be a reliable measure, they said.
“Education is needed to inform the public about the mental health hazards of regular use of high-potency cannabis, which is becoming increasingly available worldwide,” they wrote.
The study was supported by the Medical Research Council, the European Community’s Seventh Framework Program, the São Paulo Research Foundation, the National Institute for Health Research Biomedical Research Centre, and the Wellcome Trust. Five authors declared personal fees and grants from the pharmaceutical industry. No other conflicts of interest were declared.
SOURCE: Di Forti M et al. Lancet. 2019 Mar 19. doi: 10.1016/S2215-0366(19)30048-3.
Daily cannabis use, particularly high-potency cannabis, might be a significant contributor to the incidence of psychotic disorder, results of a multicenter, case-control study suggest.
“We provide the first direct evidence that cannabis use has an effect on variation in the incidence of psychotic disorders,” Marta Di Forti, PhD, and her coauthors wrote in the Lancet.
In the study, Dr. Di Forti and her coauthors looked at cannabis use in 901 patients presenting with a first psychotic episode to one of 11 sites across Europe and Brazil, compared with 1,237 population controls from the same locations. They found that daily cannabis users had more than threefold higher odds of psychotic disorder, compared with individuals who had never used cannabis (odds ratio, 3.2; P less than .0001), even after adjusting for sociodemographic factors and use of tobacco, stimulants, ketamine, and hallucinogenics.
Those who reported using high-potency cannabis (delta 9-tetrahydrocannabinol greater than or equal to 10%) – also showed a significant 60% increase in the odds of psychotic disorder, compared with never-users, which decreased slightly to 50% after controlling for daily use.
“The large sample size and the different types of cannabis available across Europe have allowed us to report that the dose-response relationship characterizing the association between cannabis use and psychosis reflects not only the use of high-potency cannabis but also the daily use of types with an amount of THC consistent with more traditional varieties,” wrote Dr. Di Forti, of the Social, Genetic, and Developmental Psychiatry Centre at King’s College London, and her coauthors.
When the authors looked at the population-attributable fractions, they calculated that 12.2% of cases of first-episode psychosis would be avoided if high-potency cannabis were not available.
(P = .0122), while those who started using high-potency cannabis at that age had more than a doubling of risk (OR, 2.3).
Similarly, those who used high-potency cannabis on a daily basis had nearly fivefold higher odds of psychotic disorder, compared with never-users, while daily users of low-potency had a 2.2-fold increase in risk.
Researchers also examined patterns of cannabis use and psychotic disorder across the 11 sites, which included Amsterdam, London; Cambridge, England; Madrid; Palermo, Italy; Paris; and Ribeirão Preto, Brazil.
They noted that there were significant variations in the incidence of psychotic disorder across the study sites, and that those variations correlated with the prevalence of daily cannabis use.
London and Amsterdam, where daily use was the most common, had the highest adjusted incidence rates of psychotic disorder (45.7 cases per 100,000 person-years in London and 37.9 per 100,000 person-years in Amsterdam). In contrast, the incidence in Bologna, Italy – where daily use was less frequent – was half that of London.
They estimated that 43% of new cases of psychotic disorder in Amsterdam were attributable to daily use of cannabis, and 50.3% were attributable to high-potency cannabis, compared with 1.2% and 2.3% of cases in Puy de Dôme in France.
“Use of high-potency cannabis was a strong predictor of psychotic disorder in Amsterdam, London, and Paris, where high-potency cannabis was widely available, by contrast with sites such as Palermo where this type was not yet available,” the authors wrote. “Our results show that, in areas where daily use and use of high-potency cannabis are more prevalent in the general population, there is an excess of cases of psychotic disorder.”
The authors did point out that the study relied on self-reported cannabis use, rather than biological sampling measures. But previous studies have shown self-reported use to be a reliable measure, they said.
“Education is needed to inform the public about the mental health hazards of regular use of high-potency cannabis, which is becoming increasingly available worldwide,” they wrote.
The study was supported by the Medical Research Council, the European Community’s Seventh Framework Program, the São Paulo Research Foundation, the National Institute for Health Research Biomedical Research Centre, and the Wellcome Trust. Five authors declared personal fees and grants from the pharmaceutical industry. No other conflicts of interest were declared.
SOURCE: Di Forti M et al. Lancet. 2019 Mar 19. doi: 10.1016/S2215-0366(19)30048-3.
FROM THE LANCET
‘The birth of a mother is a complex process’
Softening the blow to women and families of severe perinatal, postpartum psychiatric disorders
Editor’s Note: Alison M. Heru, MD, the Families in Psychiatry columnist, invited Dr. Reinstein to address this topic.
“But this was not what I expected!” That’s a statement I have heard from countless new mothers.
Women often envision pregnancy and the postpartum period as a time of pure joy. The glow of an expectant woman and the excitement of the arrival of a new baby masks the reality that many women struggle emotionally when transitioning to motherhood. Like the birth of a child, the birth of a mother is a complex process. Upholding the myth that all women seamlessly transform into mothers can have devastating effects and hinder access to mental health care.
As a psychiatrist working on a women’s inpatient unit with a perinatal program, I treat women at times of crisis. What may have begun as mild anxiety or depression sometimes quickly spirals into severe psychiatric illness. The sheer force of these severe perinatal and postpartum psychiatric disorders often leaves women and families shocked and confused, wondering what happened to their crumbled dreams of early motherhood.
What must general psychiatrists know about perinatal and postpartum psychiatric disorders? Why is maternal mental health so important? What are the barriers to treatment for these women? How can general psychiatrists best support and treat these new mothers and their families?
What data show
Maternal depression is now known to be one of the most common complications of pregnancy. Studies have suggested that about 11% of women experience depression during pregnancy1 and approximately 17% of women are depressed in the postpartum period.2 Perinatal generalized anxiety disorder has been shown to have a prevalence of 8.5%-10.5% during pregnancy with a wider variance post partum.3 Approximately 3% of women in the general community develop PTSD symptoms following childbirth.4 Research suggests that about 2% of women develop obsessive-compulsive disorder symptoms in the postpartum period.5 Postpartum psychosis, a rare but potentially devastating illness, occurs after 0.1%-0.2% of births.6
Importance of maternal mental health
There is a growing body of literature supporting both obstetric and pediatric adverse outcomes related to untreated psychiatric illness. Untreated maternal depression has been associated with obstetric complications, such as preterm delivery, preeclampsia, low birth weight, as well as the child’s developing cognitive function.7 Anxiety during pregnancy has been associated with both a shorter gestational period and adverse implications for fetal neurodevelopment.
These adverse effects were found to be even more potent in “pregnancy anxiety,” or anxiety specifically focused on the pregnancy, the birth experience, and the transition to motherhood.8 The psychotic symptoms occurring during postpartum psychosis can jeopardize the lives of both a woman and her child and carries a 4% risk of infanticide.9 Although there are limited data about the long-term effects of postpartum obsessive-compulsive disorders and PTSD, it is reasonable to assume that they might carry negative long-term implications for the mother and possibly her child.
Barriers to treatment
Despite the significant rates of mental illness, pregnant and new mothers often face barriers to receiving treatment. Many psychiatrists are hesitant to prescribe psychiatric medication to pregnant women because of concerns about teratogenic potential of psychiatric medications; similar concerns exist for newborn babies when prescribing medications to lactating mothers. In addition, the field of reproductive psychiatry is evolving at a rapid pace, making it difficult for busy psychiatrists to keep up with the ever-growing literature.
Also, it is hard to imagine a population that has more barriers to attending outpatient appointments. For many new mothers, the exhaustion and all-consuming work involved with taking care of a newborn are insurmountable barriers to obtaining mental health care. In addition, despite the awareness that new mothers often are more emotional, families can be slow to recognize the developing severity of a psychiatric illness during the peripartum and postpartum periods.
Supporting and treating new mothers
As general psychiatrists, there are several ways to directly help these women.
1. Expect the expected. Even in women with no prior psychiatric history, a significant percentage of expectant and postpartum women will develop acute psychiatric symptoms. Learn about the different presentations and treatments of perinatal and postpartum psychiatric disorders. For example, a woman might have thoughts of harming her baby in both postpartum psychosis and obsessive-compulsive disorder. However, the acuity and treatment of these two conditions drastically differ.
2. Learn more about psychiatric medications. Several apps and websites are available to psychiatrists to learn about the safety profile of psychiatric medications, such as Reprotox.org, mothertobaby.org, lactmed, and womensmentalhealth.org. Many medications are considered to be relatively safe during pregnancy and breastfeeding. It is important for psychiatrists to appreciate the risks when choosing not to prescribe to pregnant and postpartum women. Sometimes a known risk of a specific medication may be preferable to the unknown risk of leaving a woman susceptible to a severe psychiatric decompensation.
3. Involve all members of the family. A mother’s mental health has significant implications for the entire family. Psychoeducation for the family as well as frequent family sessions are key tools when treating this population. In addition, prescribing to pregnant women provides the opportunity for a psychiatrist to refine skills in joint decision making; it is crucial to involve both a patient and her spouse when discussing psychiatric medications.
4. Provide ready access and collaborate care. It is important to understand the potential rapid onset of psychiatric symptoms during the pregnancy and postpartum period. Psychiatrists should be prepared to collaborate care with other specialties. It is important to establish relationships with community psychotherapists specializing in maternal mental health, pediatricians, as well as obstetricians.
5. Learn when to seek a higher level of care. Although many women with perinatal and postpartum psychiatric symptoms can be managed as outpatients, women at times need a higher level of care. Similar to general psychiatry, women who are acutely suicidal or homicidal or have a sudden onset of psychotic and manic symptoms all should be evaluated immediately for inpatient hospitalization. Women with less severe symptoms but who require a higher level of care than typically offered in standard outpatient treatment should be candidates for partial hospitalization programs.
General intensive programs usually can accommodate these women, but it is ideal to refer this population to perinatal intensive programs. Postpartum Support International (postpartum.net) lists the nationwide inpatient and partial perinatal programs as well as regional and local services. An example of inpatient perinatal care is the women’s unit at Zucker Hillside Hospital (Northwell Health System, Glen Oaks, N.Y.), which houses an inpatient perinatal program. As a psychiatrist on the unit, I treat acute symptoms such as depression, anxiety, psychosis, mania, and catatonia that occur during the perinatal and postpartum periods. Given the severity of symptoms, I use a wide range of psychiatric medications with the possibility of electroconvulsive therapy when indicated. Psychotherapy staff on the unit offer specialized perinatal, mothers, and dialectical behavioral therapy groups. Breast pumps are available for women who wish to breastfeed. Accommodations are made for babies and children to visit their mother when clinically appropriate. Once discharged, women often are referred to Zucker Hillside’s own perinatal outpatient clinic for continued treatment. Similar models exist in select inpatient units as well as an increasing number of partial programs across the United States.
Conclusion
Psychiatric care for pregnant and new mothers can be challenging, but it is also immensely rewarding. Restoring a mother’s mental health usually leads to increased emotional stability for her entire family. Given the prevalence of maternal mental health disorders, psychiatrists in nearly every setting will encounter this population of women. With dedicated time devoted to reviewing the literature and learning about local resources, psychiatrists can feel comfortable treating women throughout the childbearing experience.
References
1. J Affect Disord. 2017 Sep;219:86-92.
2. J Psychiatr Res. 2018 Sep;104:235-48.
3. J Womens Health. (Larchmt). 2015 Sep;24(9):762-70.
4. Clin Psychol Rev. 2014 Jul;34(5):389-401.
5. Compr Psychiatry. 2009 Nov-Dec;50(6):503-9.
6. Int Rev Psychiatry. 2003 Aug;15(3):231-42.
7. Clin Obstet Gynecol. 2018 Sep;61(3):533-43.
8. Curr Opinion Psychiatry. 2012 Mar;25(2):141-8.
9. Am J Psychiatry. 2009 Apr;166(4):405-8.
Dr. Reinstein is a psychiatry attending at Zucker Hillside Hospital. Her clinical interests include reproductive psychiatry and family therapy, with a specific focus on maternal mental health. Dr. Reinstein completed her adult psychiatry residency training at Montefiore Hospital/Albert Einstein College of Medicine, New York, after graduating from the Albert Einstein College of Medicine and Yeshiva University, New York, with a BA in biology. She is one of the recipients of the 4th Annual Resident Recognition Award for Excellence in Family Oriented Care.
Softening the blow to women and families of severe perinatal, postpartum psychiatric disorders
Softening the blow to women and families of severe perinatal, postpartum psychiatric disorders
Editor’s Note: Alison M. Heru, MD, the Families in Psychiatry columnist, invited Dr. Reinstein to address this topic.
“But this was not what I expected!” That’s a statement I have heard from countless new mothers.
Women often envision pregnancy and the postpartum period as a time of pure joy. The glow of an expectant woman and the excitement of the arrival of a new baby masks the reality that many women struggle emotionally when transitioning to motherhood. Like the birth of a child, the birth of a mother is a complex process. Upholding the myth that all women seamlessly transform into mothers can have devastating effects and hinder access to mental health care.
As a psychiatrist working on a women’s inpatient unit with a perinatal program, I treat women at times of crisis. What may have begun as mild anxiety or depression sometimes quickly spirals into severe psychiatric illness. The sheer force of these severe perinatal and postpartum psychiatric disorders often leaves women and families shocked and confused, wondering what happened to their crumbled dreams of early motherhood.
What must general psychiatrists know about perinatal and postpartum psychiatric disorders? Why is maternal mental health so important? What are the barriers to treatment for these women? How can general psychiatrists best support and treat these new mothers and their families?
What data show
Maternal depression is now known to be one of the most common complications of pregnancy. Studies have suggested that about 11% of women experience depression during pregnancy1 and approximately 17% of women are depressed in the postpartum period.2 Perinatal generalized anxiety disorder has been shown to have a prevalence of 8.5%-10.5% during pregnancy with a wider variance post partum.3 Approximately 3% of women in the general community develop PTSD symptoms following childbirth.4 Research suggests that about 2% of women develop obsessive-compulsive disorder symptoms in the postpartum period.5 Postpartum psychosis, a rare but potentially devastating illness, occurs after 0.1%-0.2% of births.6
Importance of maternal mental health
There is a growing body of literature supporting both obstetric and pediatric adverse outcomes related to untreated psychiatric illness. Untreated maternal depression has been associated with obstetric complications, such as preterm delivery, preeclampsia, low birth weight, as well as the child’s developing cognitive function.7 Anxiety during pregnancy has been associated with both a shorter gestational period and adverse implications for fetal neurodevelopment.
These adverse effects were found to be even more potent in “pregnancy anxiety,” or anxiety specifically focused on the pregnancy, the birth experience, and the transition to motherhood.8 The psychotic symptoms occurring during postpartum psychosis can jeopardize the lives of both a woman and her child and carries a 4% risk of infanticide.9 Although there are limited data about the long-term effects of postpartum obsessive-compulsive disorders and PTSD, it is reasonable to assume that they might carry negative long-term implications for the mother and possibly her child.
Barriers to treatment
Despite the significant rates of mental illness, pregnant and new mothers often face barriers to receiving treatment. Many psychiatrists are hesitant to prescribe psychiatric medication to pregnant women because of concerns about teratogenic potential of psychiatric medications; similar concerns exist for newborn babies when prescribing medications to lactating mothers. In addition, the field of reproductive psychiatry is evolving at a rapid pace, making it difficult for busy psychiatrists to keep up with the ever-growing literature.
Also, it is hard to imagine a population that has more barriers to attending outpatient appointments. For many new mothers, the exhaustion and all-consuming work involved with taking care of a newborn are insurmountable barriers to obtaining mental health care. In addition, despite the awareness that new mothers often are more emotional, families can be slow to recognize the developing severity of a psychiatric illness during the peripartum and postpartum periods.
Supporting and treating new mothers
As general psychiatrists, there are several ways to directly help these women.
1. Expect the expected. Even in women with no prior psychiatric history, a significant percentage of expectant and postpartum women will develop acute psychiatric symptoms. Learn about the different presentations and treatments of perinatal and postpartum psychiatric disorders. For example, a woman might have thoughts of harming her baby in both postpartum psychosis and obsessive-compulsive disorder. However, the acuity and treatment of these two conditions drastically differ.
2. Learn more about psychiatric medications. Several apps and websites are available to psychiatrists to learn about the safety profile of psychiatric medications, such as Reprotox.org, mothertobaby.org, lactmed, and womensmentalhealth.org. Many medications are considered to be relatively safe during pregnancy and breastfeeding. It is important for psychiatrists to appreciate the risks when choosing not to prescribe to pregnant and postpartum women. Sometimes a known risk of a specific medication may be preferable to the unknown risk of leaving a woman susceptible to a severe psychiatric decompensation.
3. Involve all members of the family. A mother’s mental health has significant implications for the entire family. Psychoeducation for the family as well as frequent family sessions are key tools when treating this population. In addition, prescribing to pregnant women provides the opportunity for a psychiatrist to refine skills in joint decision making; it is crucial to involve both a patient and her spouse when discussing psychiatric medications.
4. Provide ready access and collaborate care. It is important to understand the potential rapid onset of psychiatric symptoms during the pregnancy and postpartum period. Psychiatrists should be prepared to collaborate care with other specialties. It is important to establish relationships with community psychotherapists specializing in maternal mental health, pediatricians, as well as obstetricians.
5. Learn when to seek a higher level of care. Although many women with perinatal and postpartum psychiatric symptoms can be managed as outpatients, women at times need a higher level of care. Similar to general psychiatry, women who are acutely suicidal or homicidal or have a sudden onset of psychotic and manic symptoms all should be evaluated immediately for inpatient hospitalization. Women with less severe symptoms but who require a higher level of care than typically offered in standard outpatient treatment should be candidates for partial hospitalization programs.
General intensive programs usually can accommodate these women, but it is ideal to refer this population to perinatal intensive programs. Postpartum Support International (postpartum.net) lists the nationwide inpatient and partial perinatal programs as well as regional and local services. An example of inpatient perinatal care is the women’s unit at Zucker Hillside Hospital (Northwell Health System, Glen Oaks, N.Y.), which houses an inpatient perinatal program. As a psychiatrist on the unit, I treat acute symptoms such as depression, anxiety, psychosis, mania, and catatonia that occur during the perinatal and postpartum periods. Given the severity of symptoms, I use a wide range of psychiatric medications with the possibility of electroconvulsive therapy when indicated. Psychotherapy staff on the unit offer specialized perinatal, mothers, and dialectical behavioral therapy groups. Breast pumps are available for women who wish to breastfeed. Accommodations are made for babies and children to visit their mother when clinically appropriate. Once discharged, women often are referred to Zucker Hillside’s own perinatal outpatient clinic for continued treatment. Similar models exist in select inpatient units as well as an increasing number of partial programs across the United States.
Conclusion
Psychiatric care for pregnant and new mothers can be challenging, but it is also immensely rewarding. Restoring a mother’s mental health usually leads to increased emotional stability for her entire family. Given the prevalence of maternal mental health disorders, psychiatrists in nearly every setting will encounter this population of women. With dedicated time devoted to reviewing the literature and learning about local resources, psychiatrists can feel comfortable treating women throughout the childbearing experience.
References
1. J Affect Disord. 2017 Sep;219:86-92.
2. J Psychiatr Res. 2018 Sep;104:235-48.
3. J Womens Health. (Larchmt). 2015 Sep;24(9):762-70.
4. Clin Psychol Rev. 2014 Jul;34(5):389-401.
5. Compr Psychiatry. 2009 Nov-Dec;50(6):503-9.
6. Int Rev Psychiatry. 2003 Aug;15(3):231-42.
7. Clin Obstet Gynecol. 2018 Sep;61(3):533-43.
8. Curr Opinion Psychiatry. 2012 Mar;25(2):141-8.
9. Am J Psychiatry. 2009 Apr;166(4):405-8.
Dr. Reinstein is a psychiatry attending at Zucker Hillside Hospital. Her clinical interests include reproductive psychiatry and family therapy, with a specific focus on maternal mental health. Dr. Reinstein completed her adult psychiatry residency training at Montefiore Hospital/Albert Einstein College of Medicine, New York, after graduating from the Albert Einstein College of Medicine and Yeshiva University, New York, with a BA in biology. She is one of the recipients of the 4th Annual Resident Recognition Award for Excellence in Family Oriented Care.
Editor’s Note: Alison M. Heru, MD, the Families in Psychiatry columnist, invited Dr. Reinstein to address this topic.
“But this was not what I expected!” That’s a statement I have heard from countless new mothers.
Women often envision pregnancy and the postpartum period as a time of pure joy. The glow of an expectant woman and the excitement of the arrival of a new baby masks the reality that many women struggle emotionally when transitioning to motherhood. Like the birth of a child, the birth of a mother is a complex process. Upholding the myth that all women seamlessly transform into mothers can have devastating effects and hinder access to mental health care.
As a psychiatrist working on a women’s inpatient unit with a perinatal program, I treat women at times of crisis. What may have begun as mild anxiety or depression sometimes quickly spirals into severe psychiatric illness. The sheer force of these severe perinatal and postpartum psychiatric disorders often leaves women and families shocked and confused, wondering what happened to their crumbled dreams of early motherhood.
What must general psychiatrists know about perinatal and postpartum psychiatric disorders? Why is maternal mental health so important? What are the barriers to treatment for these women? How can general psychiatrists best support and treat these new mothers and their families?
What data show
Maternal depression is now known to be one of the most common complications of pregnancy. Studies have suggested that about 11% of women experience depression during pregnancy1 and approximately 17% of women are depressed in the postpartum period.2 Perinatal generalized anxiety disorder has been shown to have a prevalence of 8.5%-10.5% during pregnancy with a wider variance post partum.3 Approximately 3% of women in the general community develop PTSD symptoms following childbirth.4 Research suggests that about 2% of women develop obsessive-compulsive disorder symptoms in the postpartum period.5 Postpartum psychosis, a rare but potentially devastating illness, occurs after 0.1%-0.2% of births.6
Importance of maternal mental health
There is a growing body of literature supporting both obstetric and pediatric adverse outcomes related to untreated psychiatric illness. Untreated maternal depression has been associated with obstetric complications, such as preterm delivery, preeclampsia, low birth weight, as well as the child’s developing cognitive function.7 Anxiety during pregnancy has been associated with both a shorter gestational period and adverse implications for fetal neurodevelopment.
These adverse effects were found to be even more potent in “pregnancy anxiety,” or anxiety specifically focused on the pregnancy, the birth experience, and the transition to motherhood.8 The psychotic symptoms occurring during postpartum psychosis can jeopardize the lives of both a woman and her child and carries a 4% risk of infanticide.9 Although there are limited data about the long-term effects of postpartum obsessive-compulsive disorders and PTSD, it is reasonable to assume that they might carry negative long-term implications for the mother and possibly her child.
Barriers to treatment
Despite the significant rates of mental illness, pregnant and new mothers often face barriers to receiving treatment. Many psychiatrists are hesitant to prescribe psychiatric medication to pregnant women because of concerns about teratogenic potential of psychiatric medications; similar concerns exist for newborn babies when prescribing medications to lactating mothers. In addition, the field of reproductive psychiatry is evolving at a rapid pace, making it difficult for busy psychiatrists to keep up with the ever-growing literature.
Also, it is hard to imagine a population that has more barriers to attending outpatient appointments. For many new mothers, the exhaustion and all-consuming work involved with taking care of a newborn are insurmountable barriers to obtaining mental health care. In addition, despite the awareness that new mothers often are more emotional, families can be slow to recognize the developing severity of a psychiatric illness during the peripartum and postpartum periods.
Supporting and treating new mothers
As general psychiatrists, there are several ways to directly help these women.
1. Expect the expected. Even in women with no prior psychiatric history, a significant percentage of expectant and postpartum women will develop acute psychiatric symptoms. Learn about the different presentations and treatments of perinatal and postpartum psychiatric disorders. For example, a woman might have thoughts of harming her baby in both postpartum psychosis and obsessive-compulsive disorder. However, the acuity and treatment of these two conditions drastically differ.
2. Learn more about psychiatric medications. Several apps and websites are available to psychiatrists to learn about the safety profile of psychiatric medications, such as Reprotox.org, mothertobaby.org, lactmed, and womensmentalhealth.org. Many medications are considered to be relatively safe during pregnancy and breastfeeding. It is important for psychiatrists to appreciate the risks when choosing not to prescribe to pregnant and postpartum women. Sometimes a known risk of a specific medication may be preferable to the unknown risk of leaving a woman susceptible to a severe psychiatric decompensation.
3. Involve all members of the family. A mother’s mental health has significant implications for the entire family. Psychoeducation for the family as well as frequent family sessions are key tools when treating this population. In addition, prescribing to pregnant women provides the opportunity for a psychiatrist to refine skills in joint decision making; it is crucial to involve both a patient and her spouse when discussing psychiatric medications.
4. Provide ready access and collaborate care. It is important to understand the potential rapid onset of psychiatric symptoms during the pregnancy and postpartum period. Psychiatrists should be prepared to collaborate care with other specialties. It is important to establish relationships with community psychotherapists specializing in maternal mental health, pediatricians, as well as obstetricians.
5. Learn when to seek a higher level of care. Although many women with perinatal and postpartum psychiatric symptoms can be managed as outpatients, women at times need a higher level of care. Similar to general psychiatry, women who are acutely suicidal or homicidal or have a sudden onset of psychotic and manic symptoms all should be evaluated immediately for inpatient hospitalization. Women with less severe symptoms but who require a higher level of care than typically offered in standard outpatient treatment should be candidates for partial hospitalization programs.
General intensive programs usually can accommodate these women, but it is ideal to refer this population to perinatal intensive programs. Postpartum Support International (postpartum.net) lists the nationwide inpatient and partial perinatal programs as well as regional and local services. An example of inpatient perinatal care is the women’s unit at Zucker Hillside Hospital (Northwell Health System, Glen Oaks, N.Y.), which houses an inpatient perinatal program. As a psychiatrist on the unit, I treat acute symptoms such as depression, anxiety, psychosis, mania, and catatonia that occur during the perinatal and postpartum periods. Given the severity of symptoms, I use a wide range of psychiatric medications with the possibility of electroconvulsive therapy when indicated. Psychotherapy staff on the unit offer specialized perinatal, mothers, and dialectical behavioral therapy groups. Breast pumps are available for women who wish to breastfeed. Accommodations are made for babies and children to visit their mother when clinically appropriate. Once discharged, women often are referred to Zucker Hillside’s own perinatal outpatient clinic for continued treatment. Similar models exist in select inpatient units as well as an increasing number of partial programs across the United States.
Conclusion
Psychiatric care for pregnant and new mothers can be challenging, but it is also immensely rewarding. Restoring a mother’s mental health usually leads to increased emotional stability for her entire family. Given the prevalence of maternal mental health disorders, psychiatrists in nearly every setting will encounter this population of women. With dedicated time devoted to reviewing the literature and learning about local resources, psychiatrists can feel comfortable treating women throughout the childbearing experience.
References
1. J Affect Disord. 2017 Sep;219:86-92.
2. J Psychiatr Res. 2018 Sep;104:235-48.
3. J Womens Health. (Larchmt). 2015 Sep;24(9):762-70.
4. Clin Psychol Rev. 2014 Jul;34(5):389-401.
5. Compr Psychiatry. 2009 Nov-Dec;50(6):503-9.
6. Int Rev Psychiatry. 2003 Aug;15(3):231-42.
7. Clin Obstet Gynecol. 2018 Sep;61(3):533-43.
8. Curr Opinion Psychiatry. 2012 Mar;25(2):141-8.
9. Am J Psychiatry. 2009 Apr;166(4):405-8.
Dr. Reinstein is a psychiatry attending at Zucker Hillside Hospital. Her clinical interests include reproductive psychiatry and family therapy, with a specific focus on maternal mental health. Dr. Reinstein completed her adult psychiatry residency training at Montefiore Hospital/Albert Einstein College of Medicine, New York, after graduating from the Albert Einstein College of Medicine and Yeshiva University, New York, with a BA in biology. She is one of the recipients of the 4th Annual Resident Recognition Award for Excellence in Family Oriented Care.
Antipsychotics and seizures: What are the risks?
Antipsychotics, especially second-generation antipsychotics (SGAs), have been proven effective for treating psychosis as well as mood disorders.1,2 Because antipsychotics can lower the epileptogenic threshold, seizures are a serious potential adverse effect. Antipsychotics can cause isolated EEG abnormalities in 7% of patients with no history of epilepsy, and clinical seizures in .5% to 1.2% of such patients.3 Additionally, the neuropathophysiology underlying epilepsy can predispose patients to psychiatric disorders4; the estimated prevalence of psychosis in patients with epilepsy is approximately 7%.5 This review will shed light on the risk of clinical seizures related to antipsychotics.
Comparing seizure risk among antipsychotics
In a review of the World Health Organization’s adverse drug reactions database, Kumlien and Lundberg6 calculated the ratio of the number of reports of seizures to the total number of reports for each drug. They found that approximately 9% of all adverse drug reaction reports involving clozapine were due to seizures. Equivalent ratios were 5.90% for quetiapine, 4.91% for olanzapine, 3.68% for risperidone, 3.27% for haloperidol, and 2.59% for aripiprazole. Using the database of the Pharmacovigilance Unit of the Basque Country, Lertxundi et al7 reported a 3.2-fold increased risk of seizure with SGAs in comparison with first-generation antipsychotics (FGAs) (95% confidence interval [CI], 2.21 to 4.63), which went down to 2.08 (CI, 1.39 to 3.12) once clozapine was excluded. However, as the authors of both studies noted, the quality and relevance of this data are limited because it relies on spontaneous reporting.
Overall, the evidence regarding the seizure risk associated with antipsychotics is scarce. To the best of our knowledge, only 2 large observational studies have compared the seizure risks associated with different antipsychotics.
Using data from the UK-based Clinical Practice Research Datalink between 1998 and 2013, Bloechlinger et al8 examined the incidence rates of seizures among patients newly diagnosed with schizophrenia, affective disorders, or dementia who were prescribed antipsychotics. They excluded patients with a history of seizures or antiepileptic use. In the cohort of 60,121 patients, the incidence rates of seizures per 10,000 person-years were 11.7 (CI, 10.0 to 13.4) for those who did not use antipsychotics, 12.4 (CI, 10.9 to 13.8) for past users, 115.4 (CI, 50.1 to 180.7) for current users of haloperidol, 48.8 (CI, 30.7 to 66.9) for current users of quetiapine, 25.9 (CI, 11.8 to 40.0) for current users of risperidone, and 19.0 (CI, 8.7 to 29.3) for current users of olanzapine. No data were available about clozapine use.
In subsequent analyses, the authors found that among patients with affective disorders, only current use of medium- to high-potency FGAs (haloperidol, prochlorperazine, and trifluoperazine) was associated with a significantly increased risk of seizures (adjusted odds ratio: 2.51, CI, 1.51 to 4.18) compared with non-users.8 Among patients with dementia, current use of olanzapine or quetiapine and current use of any FGAs were associated with significantly increased odds of seizures. This study suggests that the underlying mental illness might modulate the seizure risk associated with antipsychotics.8
Wu et al9 conducted a study based on the National Health Insurance Research Database in Taiwan. They examined the 1-year incidence of new-onset seizures among patients diagnosed with schizophrenia or mood disorders who were new to antipsychotic treatment, and calculated the risk of seizure associated with each antipsychotic in reference to risperidone. They found that those receiving clozapine, thioridazine, and haloperidol were 2 to 3 times more likely to develop seizures than those treated with risperidone; risks associated with the rest of the FGAs were similar to that of risperidone.
The results of these 2 large cohort studies are somewhat concurrent in indicating that, other than clozapine, SGAs incur similar risks of seizures; furthermore, they specify that, contrary to earlier studies,10 haloperidol is associated with significantly higher odds of seizures. While both of these cohort studies controlled for several sociodemographic and clinical confounders, they have several limitations. First, diagnoses of seizures were based on information available in databases, which might be subject to inaccuracies. Second, neither study evaluated the effect of drug dosage and duration of exposure on new-onset seizures.
Continue to: Most evidence is from case reports
Most evidence is from case reports
Other than these 2 large studies, most of the evidence addressing the relationship between the use of antipsychotics and incidence of seizures is low quality and relies on case reports or expert opinions. Older studies found that, among FGAs, seizure risk is highest with chlorpromazine and promazine, and lowest with thioridazine and haloperidol.10 As for SGAs, case reports have described seizuresassociated with the use of quetiapine, aripiprazole, risperidone, paliperidone, and olanzapine.
Quetiapine. Three case reports published between 2002 and 2010 describe generalized
Aripiprazole. Five case reports described staring spells and tonic-clonic seizures in patients receiving 10 to 15 mg of aripiprazole.15-19 In the New Drug Application (NDA) for aripiprazole, the incidence of seizures was estimated to be .11% (1 of 926 patients) in placebo-controlled trials and .46% (3 of 859 patients) in haloperidol-controlled trials.20
Risperidone’s product labeling suggests the drug should be used with caution in patients with a history of seizures or conditions that could result in a lower seizure threshold. In Phase III placebo-controlled trials, seizures occurred in .3% of patients treated with risperidone, although in some cases, the seizures were induced by electrolyte disturbances such as hyponatremia.21 Gonzalez-Heydrich et al22 and Holzhausen et al23 found no increase in seizure activity among patients with epilepsy who were receiving risperidone. Lane et al24 published a case report of a geriatric woman who presented with a generalized tonic-clonic seizure related to rapid titration of risperidone; however, with slower titration and lower doses, she stopped having seizures without adding any antiepileptic drugs. Komossa et al25 found that risperidone is less epileptogenic than clozapine, with a relative risk of .22.
Paliperidone is the active metabolite of risperidone and does not have pharmacokinetic interactions with drugs metabolized by the cytochrome P450 (CYP) enzymes. Its labeling indicates that the drug should be used with caution in patients with a history of seizures.26 In Phase III placebo-controlled trials of paliperidone, the rate of seizures was .22%.27 Two case reports suggest close monitoring of seizure risk in patients receiving paliperidone.28,29 Liang et al29 reported that co-administration of valproic acid could mask an underlying decrease of the seizure threshold caused by antipsychotics such as paliperidone.
Continue to: Olanzapine
Olanzapine is a thienobenzodiazepine derivative and is chemically related to clozapine.30 The olanzapine NDA31 shows that 23 of 3,139 patients developed seizures, mainly tonic-clonic, with evidence suggesting that the seizures may have been due to confounding factors such as a history of seizures or metabolic abnormalities. There were no statistically significant differences in the rate of seizures associated with olanzapine compared with placebo or haloperidol (P = .252 and .168, respectively).
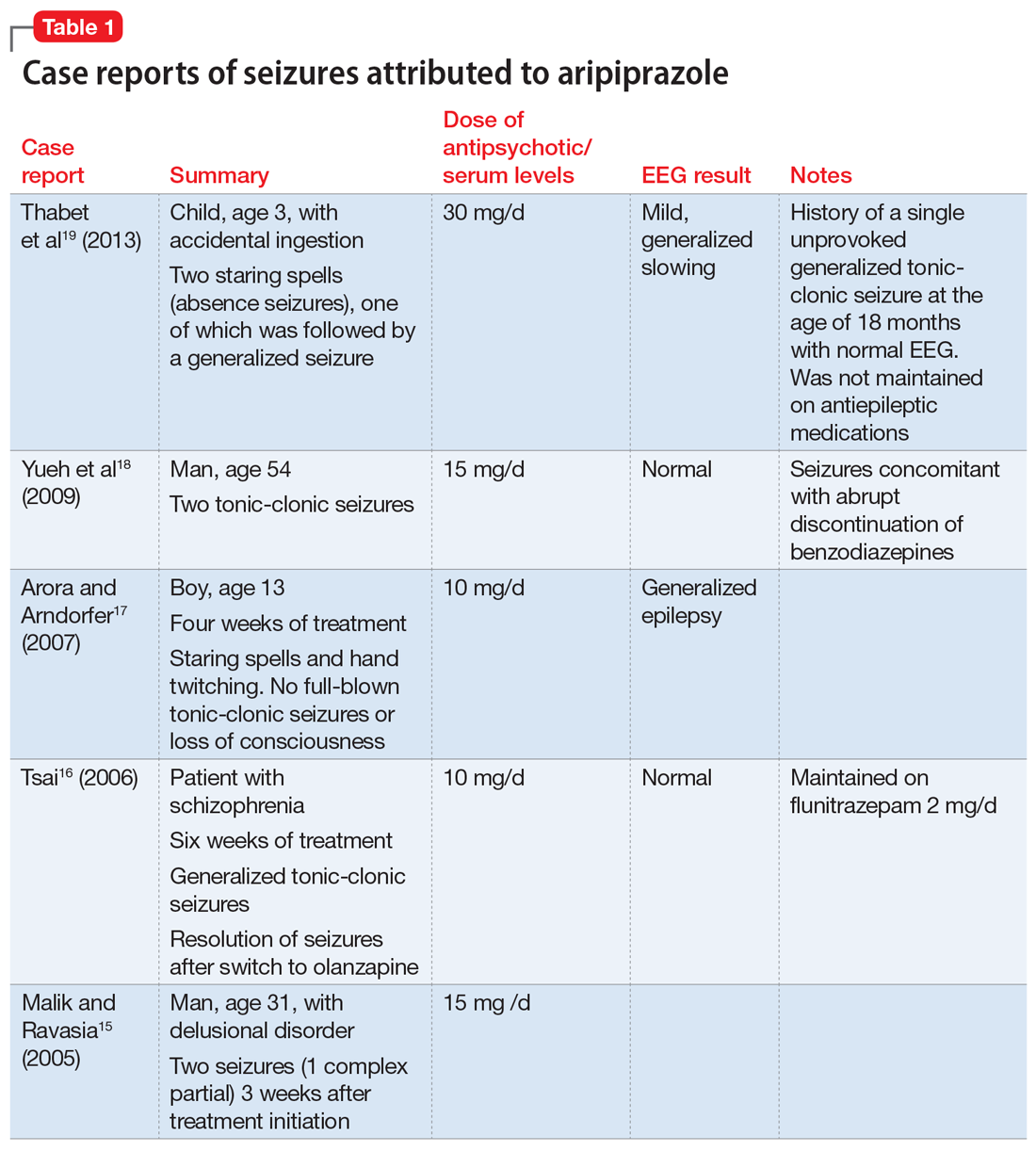
A literature review for olanzapine yielded 1 case report of repetitive focal seizures and lingual dystonia,32 5 case reports of generalized tonic-clonic seizures and myoclonus,33-37 and 2 case reports of status epilepticus.38,39 Olanzapine’s clearance is 25% to 30% lower in women, and most of these case reports occurred women.40
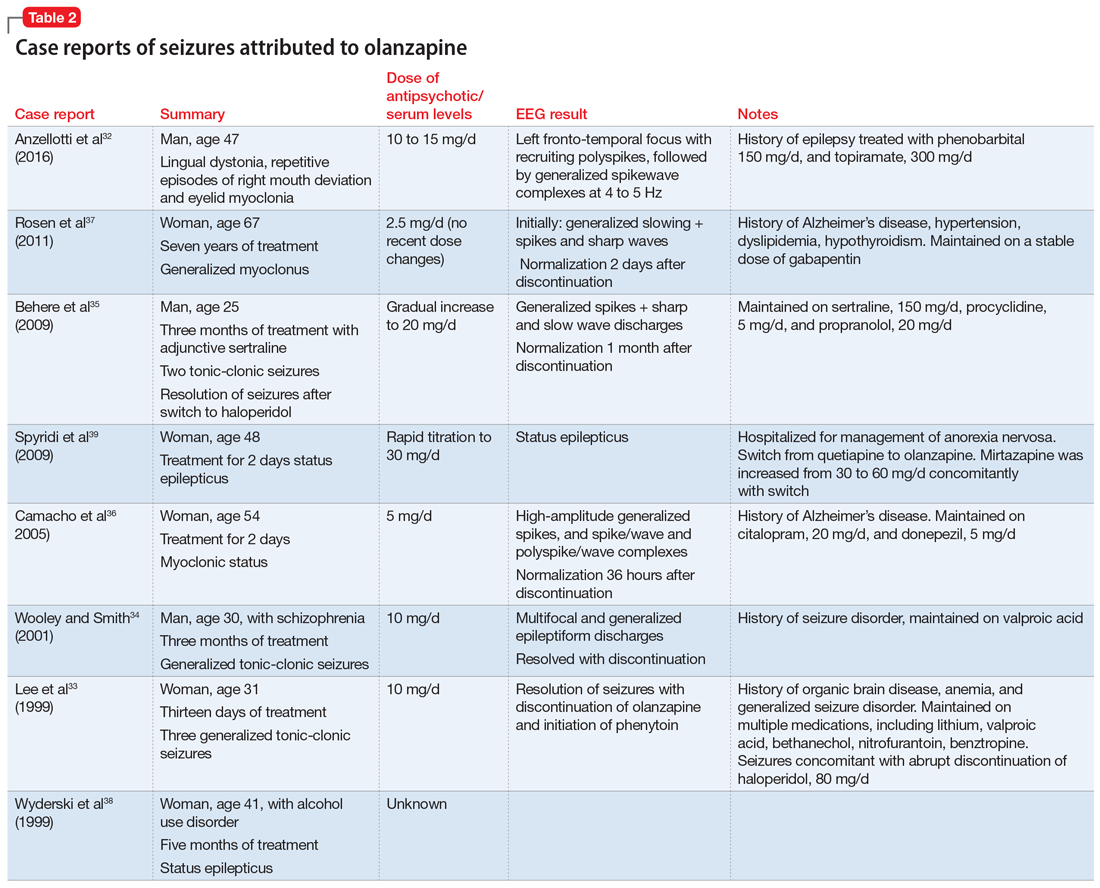
Details of the above case reports are summarized in Table 1 (aripiprazole15-19), Table 2 (olanzapine32-39), and Table 3 (paliperidone,28,29 quetiapine,11-13 and risperidone22-24).
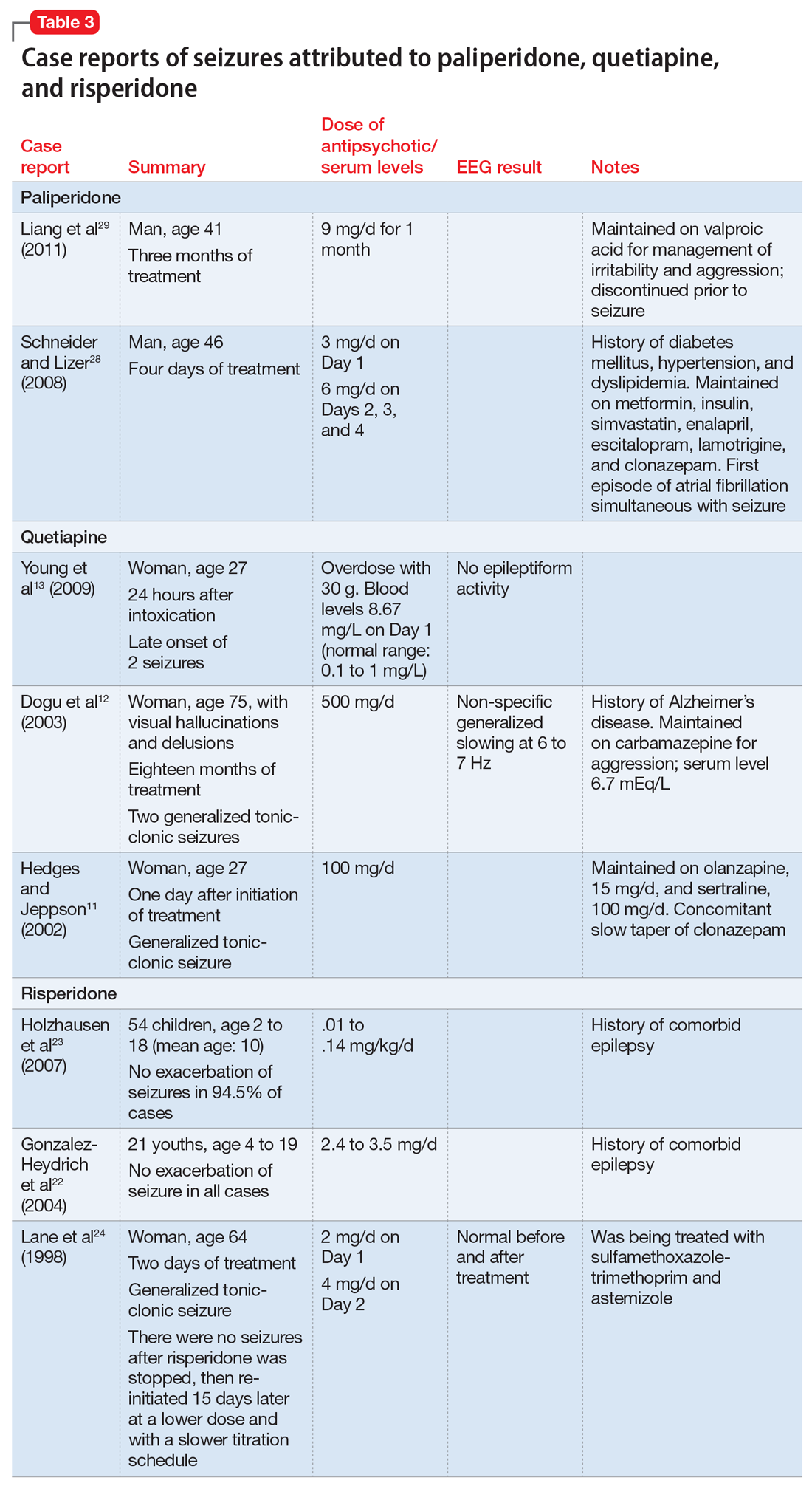
Ziprasidone. According to the NDA safety database, the seizure rate attributed to ziprasidone was 1.8 per 100 subject-years or 0.54% of participants (12 of 2,588).41 No additional studies have been published regarding its seizure risk.
Clozapine has a black-box warning
To the best of our knowledge, clozapine is the only antipsychotic that carries an FDA “black-box” warning regarding its risk of inducing seizures.42 Devinsky and Pacia43 reported a cumulative risk of 10% after 3.8 years of treatment. The literature has described clozapine-induced generalized tonic-clonic, myoclonic, simple and complex partial, and absence seizures.44 Table 445 lists the estimated frequency of each seizure type based on 101 cases of clozapine-induced seizures. Myoclonic seizures and drop attacks could be precursors/warning signs of grand mal tonic-clonic seizures.46,47 Seizures have been observed at all stages of treatment, but were more common during initiation of clozapine, which emphasizes the importance of a progressive and slow titration.43,48 The incidence of seizures was estimated to be 6% in a sample of 216 patients with schizophrenia with no history of epilepsy who were prescribed clozapine.49
Continue to: Regarding a possible association between...
Regarding a possible association between clozapine dose or clozapine plasma levels and seizure risk, there is a positive linear relationship between the dose of clozapine and its serum concentration over a dosing range of 25 to 800 mg/d.50 However, the plasma concentration is also significantly affected by factors such as smoking, gender, age, drug interactions, and CYP genotypes. Therefore, the same clozapine dose will yield a lower serum concentration in an older male who smokes compared with a younger, non-smoking female.51 Perry et al52 suggested a dosing nomogram to calculate the influence of gender and smoking. Seizure risk, especially for tonic-clonic seizures, has been reported to increase with clozapine doses >600 mg/d,53 and with plasma concentrations exceeding 1,000 to 1,300 mg/L.54 However, in a 2011 regression analysis, Varma et al55 found no statistically significant relationship between seizure risk and clozapine oral dose; there was not enough data to test a correlation between clozapine plasma levels and the incidence of seizures.
How antipsychotics might lower the seizure threshold
Researchers have suggested several possible mechanisms to explain how antipsychotics might lower the seizure threshold. Antagonism of dopamine D4, histamine H1, and acetylcholine-muscarinic receptors seems to induce EEG alterations and increase the risk of seizures.56 Additionally, modulation of the N-methyl-
Watch for pharmacokinetic interactions
The CYP enzymes involved in drug metabolism include CYP1A2, CYP2C9, CYP2C19, CYP2D6, and CYP3A4. Most commonly used antiepileptics and antipsychotics are metabolized by CYP enzymes, and may also act as inhibitors or inducers of these enzymes.61 Drug interactions may impair seizure control, which is why monotherapy is preferable to combination treatment in patients with epilepsy.62 Carbamazepine and phenytoin are inducers of both CYP1A2 (which metabolizes olanzapine and clozapine), and CYP3A4 (which metabolizes haloperidol, risperidone, quetiapine, ziprasidone and clozapine). Paliperidone is not metabolized by CYP enzymes.62 Discontinuing an enzyme-inducing agent may result in increased antipsychotic plasma concentrations, which might lead to an increased risk of seizures.
Valproic acid, which is often used to prevent or treat clozapine-induced seizures, has an unclear effect on clozapine plasma concentrations.63 Although valproic acid is known to inhibit clozapine metabolism, 2 reports have suggested that the plasma concentrations of clozapine and its metabolites may decrease after adding valproic acid.64,65 Other studies have found that valproic acid increases plasma concentrations of clozapine while it decreases plasma concentrations of norclozapine; norclozapine is the main clozapine metabolite responsible for inducing seizures.66,67
Steps for minimizing seizure risk
Determining the seizure risk for a patient taking an antipsychotic is challenging because doing so depends not only on the seizurogenic potential of each drug but also on individualized predisposing factors.11,57,68 Choosing the “best” antipsychotic therefore largely depends on each patient’s profile. The predisposing factors consist mainly of the individually inherited seizure threshold (personal history of febrile convulsions or a family history of seizures) and other comorbid seizurogenic conditions, such as a history of head trauma, brain injury, intellectual disability, cerebral arteriosclerosis, neurodegenerative diseases, encephalopathy, chronic renal insufficiency, and hyponatremia. Furthermore, seizure risk depends on the antipsychotic dose administered and the rate of titration.11
Continue to: There is not enough evidence...
There is not enough evidence to recommend performing an EEG in all patients taking antipsychotics. Such testing is recommended only for patients who have predisposing factors for seizures. If an EEG shows any abnormality in a patient taking clozapine, consider decreasing the clozapine dose69,70 or adding an antiepileptic drug such as valproic acid or lamotrigine.44,70
Although clozapine carries a black-box warning of increased risk of causing seizures, there is no consensus regarding the efficacy of co-prescribing an antiepileptic. Some studies have suggested prescribing valproic acid prophylactically,71 after the occurrence of 1 seizure,59 or after 2 seizures.54,72 Others have recommended prescribing prophylactic valproic acid for patients taking ≥600 mg/d of clozapine or whose clozapine plasma levels are >500 mg/L.73 Varma et al55 recommended starting an antiepileptic medication if there are clear epileptiform discharges on EEG, if the patient develops stuttering or speech difficulties, or if seizures occur. Liukkonen et al72 advised initiating an antiepileptic at the start of clozapine treatment in patients who are taking other epileptogenic medications, patients with pre-existing seizure disorder, and patients with neurologic abnormalities. On the other hand, Caetano51 argued against primary prevention of seizures for patients receiving >600 mg/d of clozapine, suggesting that “the risk of seizures would be better managed by close clinical monitoring and measures of clozapine serum concentration rather than adding an anticonvulsant drug.”
Current recommendations for primary and secondary prevention of clozapine-induced seizures are detailed in Table 5.42,44,45,51,55,57,69,74,75

Studies addressing the seizurogenic potential of SGAs other than clozapine have a low level of evidence and include patients who had comorbid conditions and were taking other medications that could cause seizures. Additionally, clinical trials of SGAs rarely include patients with seizure disorders; this might underestimate the risk of seizures.4
The effect of the mental illness itself on the seizure threshold needs to be considered.43 Bloechlinger et al8 found that dementia might be inherently associated with a higher risk of antipsychotic-related seizures. Moreover, numerous qualitative EEG studies have found abnormalities in 20% to 60% of patients with schizophrenia.56 Other quantitative studies have reported mild and nonspecific EEG abnormalities, such as increased delta and/or theta activity, in many non-medicated patients with schizophrenia.10,76 Additionally, brain tissue analysis of deceased patients who had schizophrenia has shown a significant increase in dopamine concentrations in the left amygdala compared with controls, and this might be responsible for enhanced electrical activity in this region.10 Some studies have described EEG slowing in the frontal brain regions of patients with schizophrenia,77 and was selectively normalized in these areas with antipsychotics.78
As always, start low, go slow
Mounting evidence suggests that antipsychotic medications decrease the seizure threshold. Practitioners should thus be cautious in prescribing antipsychotics and should target reaching the minimal effective dose with slow titration, especially in patients with predisposing factors for epilepsy.
Continue to: Although evidence suggests...
Although evidence suggests antipsychotics can induce different types of epileptic seizures, the quality of this evidence is low. Randomized controlled trials are needed to determine which antipsychotics increase seizure risk and whether there is a dose-effect relationship.
Bottom Line
Among second-generation antipsychotics, clozapine appears to increase the risk of clinical seizure the most. Correlations with dosage and/or plasma levels have not been proven. Psychiatrists should be vigilant for pharmacokinetic interactions between antipsychotics and antiepileptics, notably via CYP1A2 and CYP3A4.
Related Resources
- Druschky K, Bleich S, Grohmann R, et al. Seizure rates under treatment with antipsychotic drugs: Data from the AMSP project. World J Biol Psychiatry. 2018;15:1-10.
- Epilepsy Foundation. For professionals: Antipsychotics. https://www.epilepsy.com/learn/professionals/diagnosistreatment/psychotropic-drugs-developmental-disabilities/comorbid-5.
Drug Brand Names
Aripiprazole • Abilify
Benztropine • Cogentin
Bethanechol • Duvoid
Carbamazepine • Carbatrol, Tegretol
Chlorpromazine • Thorazine
Cimetidine • Tagamet
Ciprofloxacin • Cipro
Citalopram • Celexa
Clonazepam • Klonopin
Clozapine • Clozaril
Donepezil • Aricept
Enalapril • Vasotec
Erythromycin • Erythrocin
Escitalopram • Lexapro
Flunitrazepam • Rohypnol
Fluvoxamine • Luvox
Gabapentin • Neurontin
Haloperidol • Haldol
Lamotrigine • Lamictal
Lithium • Eskalith, Lithobid
Metformin • Fortamet, Glucophage
Mirtazapine • Remeron
Nitrofurantoin • Furadantin
Olanzapine • Zyprexa
Paliperidone • Invega
Phenobarbital • Luminal
Phenytoin • Dilantin
Prochlorperazine • Compazine
Procyclidine • Kemadrin
Propranolol • Inderal
Quetiapine • Seroquel
Risperidone • Risperdal
Sertraline • Zoloft
Simvastatin • Zocor
Sulfamethoxazole/trimethoprim • Bactrim, Sulfatrim
Topiramate • Topamax
Trifluoperazine • Stelazine
Valproic acid • Depakene, Depakote
Ziprasidone • Geodon
1. Bruijnzeel D, Suryadevara U, Tandon R. Antipsychotic treatment of schizophrenia: an update. Asian J Psychiatr. 2014;11:3-7.
2. Hrdlicka M, Dudova I. Atypical antipsychotics in the treatment of early-onset schizophrenia. Neuropsychiatr Dis Treat. 2015;11:907-913.
3. Koch-Stoecker S. Antipsychotic drugs and epilepsy: indications and treatment guidelines. Epilepsia. 2002;43(suppl 2):19-24.
4. Alper K, Schwartz KA, Kolts RL, et al. Seizure incidence in psychopharmacological clinical trials: an analysis of Food and Drug Administration (FDA) summary basis of approval reports. Biol Psychiatry. 2007;62(4):345-354.
5. Torta R, Keller R. Behavioral, psychotic, and anxiety disorders in epilepsy: etiology, clinical features, and therapeutic implications. Epilepsia. 1999;40(suppl 10):S2-S20.
6. Kumlien E, Lundberg PO. Seizure risk associated with neuroactive drugs: data from the WHO adverse drug reactions database. Seizure. 2010;19(2):69-73.
7. Lertxundi U, Hernandez R, Medrano J, et al. Antipsychotics and seizures: higher risk with atypicals? Seizure. 2013;22(2):141-143.
8. Bloechliger M, Rüegg S, Jick SS, et al. Antipsychotic drug use and the risk of seizures: follow-up study with a nested case-control analysis. CNS Drugs. 2015;29(7):591-603.
9. Wu CS, Wang SC, Yeh IJ, et al. Comparative risk of seizure with use of first- and second-generation antipsychotics in patients with schizophrenia and mood disorders. J Clin Psychiatry. 2016;77(5):e573-e579.
10. Cold JA, Wells BG, Froemming JH. Seizure activity associated with antipsychotic therapy. [Erratum in DICP. 1990;24(10):1012.] DICP. 1990;24(6):601-606.
11. Hedges DW, Jeppson KG. New-onset seizure associated with quetiapine and olanzapine. Ann Pharmacother. 2002;36(3):437-439.
12. Dogu O, Sevim S, Kaleagasi HS. Seizures associated with quetiapine treatment. Ann Pharmacother. 2003;37(9):1224-1227.
13. Young AC, Kleinschmidt KC, Wax PM. Late-onset seizures associated with quetiapine poisoning. J Med Toxicol. 2009;5(1):24-26.
14. US Food and Drug Administration. Recommendation of approvable action for quetiapine fumarate extended release (Seroquel® XR) for the treatment of schizophrenia. http://www.accessdata.fda.gov/drugsatfda_docs/nda/2007/022047Orig1s000MedR.pdf. April 24, 2007. Accessed January 28, 2019.
15. Malik AR, Ravasia S. Aripiprazole-induced seizure. Can J Psychiatry. 2005;50(3):186.
16. Tsai JF. Aripiprazole-associated seizure. J Clin Psychiatry. 2006;67(6):995-996.
17. Arora M, Arndorfer L. EEG abnormalities in a patient taking aripiprazole. Psychiatry (Edgmont). 2007;4(7):18-19.
18. Yueh CL, Yu SL, Chen HM, et al. Aripiprazole-induced seizure: a second case report. BMJ case reports. 2009;2009:bcr03.2009.1693. doi: 10.1136/bcr.03.2009.1693.
19. Thabet FI, Sweis RT, Joseph SA. Aripiprazole-induced seizure in a 3-year-old child: a case report and literature review. Clin Neuropharmacol. 2013;36(1):29-30.
20. US Food and Drug Administration. Abilify (Aripiprazole) tablets. Medical Review Part 2. http://www.accessdata.fda.gov/drugsatfda_docs/nda/2002/21-436_Abilify_medr_P2.pdf. Published March 07, 2003. Accessed January 28, 2019.
21. US Food and Drug Administration. Drugs@FDA: FDA approved drug products. Risperdal tablets, Risperdal oral solution & Risperdal M-tab orally disintegrating tablets. http://www.accessdata.fda.gov/drugsatfda_docs/nda/2003/021444_S004_RISPERDAL_TABLETS.pdf. Published September 10, 2003. Accessed January 28, 2019.
22. Gonzalez-Heydrich J, Pandina GJ, Fleisher CA, et al. No seizure exacerbation from risperidone in youth with comorbid epilepsy and psychiatric disorders: a case series. J Child Adolesc Psychopharmacol. 2004;14(2):295-310.
23. Holzhausen SPF, Guerreiro MM, Baccin CE, et al. Use of risperidone in children with epilepsy. Epilepsy Behav. 2007;10(3):412-416.
24. Lane HY, Chang WH, Chou JC. Seizure during risperidone treatment in an elderly woman treated with concomitant medications. J Clinl Psychiatry. 1998;59(2):81-82.
25. Komossa K, Rummel-Kluge C, Schwarz S, et al. Risperidone versus other atypical antipsychotics for schizophrenia. Cochrane Database Syst Rev. 2011;(1):19:CD006626.
26. Paliperidone [package insert]. Mountainville, CA: Janssen Pharmaceuticals, Inc.; 2007.
27. Brugge, MD; US Food and Drug Administration. Paliperidone OROS oral formulation. http://www.accessdata.fda.gov/drugsatfda_docs/nda/2006/021999s000_MedR_Part4.pdf. Accessed January 28, 2019.
28. Schneider RA, Lizer MH. Apparent seizure and atrial fibrillation associated with paliperidone. Am J Health System Pharm. 2008;65(22):2122-2125.
29. Liang CS, Yang FW, Chiang KT. Paliperidone-associated seizure after discontinuation of sodium valproate: a case report. J Clin Psychopharmacol. 2011;31(2):246-247.
30. Fulton B, Goa KL. Olanzapine. A review of its pharmacological properties and therapeutic efficacy in the management of schizophrenia and related psychoses. Drugs. 1997;53(2):281-298.
31. US Food and Drug Administration. Drugs@FDA: FDA approved drug products: Zyprexa (olanzapine). ORIG-1. http://www.accessdata.fda.gov/drugsatfda_docs/nda/96/020592_Original_Approval_Pkg%20.pdf. Published September 30, 1996. Accessed January 28, 2019.
32. Anzellotti F, Capasso M, Frazzini V, et al. Olanzapine-related repetitive focal seizures with lingual dystonia. Epileptic Disord. 2016;18(1):83-86.
33. Lee JW, Crismon ML, Dorson PG. Seizure associated with olanzapine. Ann Pharmac. 1999;33(5):554-556.
34. Woolley J, Smith S. Lowered seizure threshold on olanzapine. Br J Psychiatry. 2001;178(1):85-86.
35. Behere RV, Anjith D, Rao NP, et al. Olanzapine-induced clinical seizure: a case report. Clin Neuropharmacol. 2009;32(5):297-298.
36. Camacho A, García-Navarro M, Martínez B, et al. Olanzapine-induced myoclonic status. Clin Neuropharmacol. 2005;28(3):145-147.
37. Rosen JB, Milstein MJ, Haut SR. Olanzapine-associated myoclonus. Epilepsy Res. 2012;98(2-3):247-250.
38. Wyderski RJ, Starrett WG, Abou-Saif A. Fatal status epilepticus associated with olanzapine therapy. Ann Pharmacother. 1999;33(7-8):787-789.
39. Spyridi S, Sokolaki S, Nimatoudis J, et al. Status epilepticus in a patient treated with olanzapine and mirtazapine. Int J Clin Pharmacol Ther. 2009;47(2):120-123.
40. Schatzberg AF, Nemeroff CB. Essentials of clinical psychopharmacology. 2nd ed. Arlington, Virginia: American Psychiatric Publishing; 2006.
41. US Food and Drug Administration. Drug approval package: Geodon (Ziprasidone HCI) Capsules. Medical Review Part 2. http://www.accessdata.fda.gov/drugsatfda_docs/nda/2001/20-825_Geodan_medr_P2.pdf. Published February 5, 2001. Accessed January 29, 2019.
42. Clozaril [package insert]. East Hanover, NJ: Novartis; 2008.
43. Devinsky O, Pacia SV. Seizures during clozapine therapy. J Clin Psychiatry. 1994;55(suppl B):153-156.
44. Williams AM, Park SH. Seizure associated with clozapine: incidence, etiology, and management. CNS Drugs. 2015;29(2):101-111.
45. Wong J, Delva N. Clozapine-induced seizures: recognition and treatment. Can J Psychiatry. 2007;52(7):457-463.
46. Berman I, Zalma A, DuRand CJ, et al. Clozapine-induced myoclonic jerks and drop attacks. J Clin Psychiatry. 1992;53(9):329-330.
47. Gouzoulis E, Ozdaglar A, Kasper J. Myoclonic seizures followed by grand mal seizures during clozapine treatment. Am J Psychiatry. 1993;150(7):1128.
48. Sajatovic M, Meltzer HY. Clozapine-induced myoclonus and generalized seizures. Biol Psychiatry. 1996;39(5):367-370.
49. Grover S, Hazari N, Chakrabarti S, et al. Association of clozapine with seizures: a brief report involving 222 patients prescribed clozapine. East Asian Arch Psychiatry. 2015;25(2):73-78.
50. Byerly MJ, DeVane CL. Pharmacokinetics of clozapine and risperidone: a review of recent literature. J Clin Psychopharmacol. 1996;16(2):177-187.
51. Caetano D. Use of anticonvulsants as prophylaxis for seizures in patients on clozapine. Australas Psychiatry. 2014;22(1):78-83.
52. Perry PJ, Bever KA, Arndt S, et al. Relationship between patient variables and plasma clozapine concentrations: a dosing nomogram. Biol Psychiatry.1998;44(8):733-738.
53. Dumortier G, Mahé V, Pons D, et al. Clonic seizure associated with high clozapine plasma level. J Neuropsychiatry Clin Neurosci. 2001;13(2):302-303.
54. Funderburg LG, Vertrees JE, True JE, et al. Seizure following addition of erythromycin to clozapine treatment. Am J Psychiatry. 1994;151(12):1840-1841.
55. Varma S, Bishara D, Besag FMC, et al. Clozapine-related EEG changes and seizures: dose and plasma-level relationships. Ther Adv Psychopharmacol. 2011;1(2):47-66.
56. Amann BL, Pogarell O, Mergl R, et al. EEG abnormalities associated with antipsychotics: a comparison of quetiapine, olanzapine, haloperidol and healthy subjects. Hum Psychopharmacol. 2003;18(8):641-646.
57. Pisani F, Oteri G, Costa C, et al. Effects of psychotropic drugs on seizure threshold. Drug Saf. 2002;25(2):91-110.
58. Maurice T, Phan VL, Urani A, et al. Neuroactive neurosteroids as endogenous effectors for the sigma1 (sigma1) receptor: pharmacological evidence and therapeutic opportunities. Jpn J Pharmacol. 1999;81(2):125-155.
59. Haller E, Binder RL. Clozapine and seizures. Am J Psychiatry. 1990;147(8):1069-1071.
60. Torta R, Monaco F. Atypical antipsychotics and serotoninergic antidepressants in patients with epilepsy: pharmacodynamic considerations. Epilepsia. 2002;43(suppl 2):8-13.
61. Spina E. Drug interactions. In: Shorvon S, Perucca E, Engel J Jr, eds. The treatment of epilepsy. 3rd ed. Oxford, UK: Blackwell Publishing; 2009:361-377.
62. Spina E, Perucca E. Clinical significance of pharmacokinetic interactions between antiepileptic and psychotropic drugs. Epilepsia. 2002;43(suppl 2):37-44.
63. de Leon J, Santoro V, D’Arrigo C, et al. Interactions between antiepileptics and second-generation antipsychotics. Expert Opin Drug Metab Toxicol. 2012;8(3):311-334.
64. Finley P, Warner D. Potential impact of valproic acid therapy on clozapine disposition. Biol Psychiatry. 1994;36(7):487-488.
65. Longo LP, Salzman C. Valproic acid effects on serum concentrations of clozapine and norclozapine. Am J Psychiatry. 1995;152(4):650.
66. Centorrino F, Baldessarini RJ, Kando J, et al. Serum concentrations of clozapine and its major metabolites: effects of cotreatment with fluoxetine or valproate. Am J Psychiatry. 1994;151(1):123-125.
67. Facciolà G, Avenoso A, Scordo MG, et al. Small effects of valproic acid on the plasma concentrations of clozapine and its major metabolites in patients with schizophrenic or affective disorders. Ther Drug Monit. 1999;21(3):341-345.
68. Hyde TM, Weinberger DR. Seizures and schizophrenia. Schizophr Bull. 1997;23(4):611-622.
69. Muzyk A, Gala G, Kahn DA. Use of lamotrigine in a patient with a clozapine-related seizure. J Psychiatr Pract. 2010;16(2):125-128.
70. Kikuchi YS, Sato W, Ataka K, et al. Clozapine-induced seizures, electroencephalography abnormalities, and clinical responses in Japanese patients with schizophrenia. Neuropsychiatr Dis Treat. 2014;10:1973-1978.
71. Taner E, Coşar B, Işik E. Clozapine-induced myoclonic seizures and valproic acid. Int J Psychiatry Clin Pract. 1998;2(1):53-55.
72. Liukkonen J, Koponen HJ, Nousiainen U. Clinical picture and long-term course of epileptic seizures that occur during clozapine treatment. Psychiatry Res. 1992;44(2):107-112.
73. Devinsky O, Honigfeld G, Patin J. Clozapine-related seizures. Neurology. 1991;41(3):369-371.
74. Foster R, Olajide D. A case of clozapine-induced tonic-clonic seizures managed with valproate: implications for clinical care. J Psychopharmacol. 2005;19(1):93-96.
75. Gandelman-Marton R, Theitler J, Klein C, et al. Phenytoin intoxication in a clozapine-related prolonged seizure. J Emerg Med. 2008;35(4):407-409.
76. Primavera A, Giberti L, Scotto P, et al. Nonconvulsive status epilepticus as a cause of confusion in later life: a report of 5 cases. Neuropsychobiology. 1994;30(2-3):148-152.
77. Boutros NN, Arfken C, Galderisi S, et al. The status of spectral EEG abnormality as a diagnostic test for schizophrenia. Schizophrenia Res. 2008;99(1-3):225-237.
78. Takahashi T, Cho RY, Mizuno T, et al. Antipsychotics reverse abnormal EEG complexity in drug-naïve schizophrenia: a multiscale entropy analysis. Neuroimage. 2010;51(1):173-182.
Antipsychotics, especially second-generation antipsychotics (SGAs), have been proven effective for treating psychosis as well as mood disorders.1,2 Because antipsychotics can lower the epileptogenic threshold, seizures are a serious potential adverse effect. Antipsychotics can cause isolated EEG abnormalities in 7% of patients with no history of epilepsy, and clinical seizures in .5% to 1.2% of such patients.3 Additionally, the neuropathophysiology underlying epilepsy can predispose patients to psychiatric disorders4; the estimated prevalence of psychosis in patients with epilepsy is approximately 7%.5 This review will shed light on the risk of clinical seizures related to antipsychotics.
Comparing seizure risk among antipsychotics
In a review of the World Health Organization’s adverse drug reactions database, Kumlien and Lundberg6 calculated the ratio of the number of reports of seizures to the total number of reports for each drug. They found that approximately 9% of all adverse drug reaction reports involving clozapine were due to seizures. Equivalent ratios were 5.90% for quetiapine, 4.91% for olanzapine, 3.68% for risperidone, 3.27% for haloperidol, and 2.59% for aripiprazole. Using the database of the Pharmacovigilance Unit of the Basque Country, Lertxundi et al7 reported a 3.2-fold increased risk of seizure with SGAs in comparison with first-generation antipsychotics (FGAs) (95% confidence interval [CI], 2.21 to 4.63), which went down to 2.08 (CI, 1.39 to 3.12) once clozapine was excluded. However, as the authors of both studies noted, the quality and relevance of this data are limited because it relies on spontaneous reporting.
Overall, the evidence regarding the seizure risk associated with antipsychotics is scarce. To the best of our knowledge, only 2 large observational studies have compared the seizure risks associated with different antipsychotics.
Using data from the UK-based Clinical Practice Research Datalink between 1998 and 2013, Bloechlinger et al8 examined the incidence rates of seizures among patients newly diagnosed with schizophrenia, affective disorders, or dementia who were prescribed antipsychotics. They excluded patients with a history of seizures or antiepileptic use. In the cohort of 60,121 patients, the incidence rates of seizures per 10,000 person-years were 11.7 (CI, 10.0 to 13.4) for those who did not use antipsychotics, 12.4 (CI, 10.9 to 13.8) for past users, 115.4 (CI, 50.1 to 180.7) for current users of haloperidol, 48.8 (CI, 30.7 to 66.9) for current users of quetiapine, 25.9 (CI, 11.8 to 40.0) for current users of risperidone, and 19.0 (CI, 8.7 to 29.3) for current users of olanzapine. No data were available about clozapine use.
In subsequent analyses, the authors found that among patients with affective disorders, only current use of medium- to high-potency FGAs (haloperidol, prochlorperazine, and trifluoperazine) was associated with a significantly increased risk of seizures (adjusted odds ratio: 2.51, CI, 1.51 to 4.18) compared with non-users.8 Among patients with dementia, current use of olanzapine or quetiapine and current use of any FGAs were associated with significantly increased odds of seizures. This study suggests that the underlying mental illness might modulate the seizure risk associated with antipsychotics.8
Wu et al9 conducted a study based on the National Health Insurance Research Database in Taiwan. They examined the 1-year incidence of new-onset seizures among patients diagnosed with schizophrenia or mood disorders who were new to antipsychotic treatment, and calculated the risk of seizure associated with each antipsychotic in reference to risperidone. They found that those receiving clozapine, thioridazine, and haloperidol were 2 to 3 times more likely to develop seizures than those treated with risperidone; risks associated with the rest of the FGAs were similar to that of risperidone.
The results of these 2 large cohort studies are somewhat concurrent in indicating that, other than clozapine, SGAs incur similar risks of seizures; furthermore, they specify that, contrary to earlier studies,10 haloperidol is associated with significantly higher odds of seizures. While both of these cohort studies controlled for several sociodemographic and clinical confounders, they have several limitations. First, diagnoses of seizures were based on information available in databases, which might be subject to inaccuracies. Second, neither study evaluated the effect of drug dosage and duration of exposure on new-onset seizures.
Continue to: Most evidence is from case reports
Most evidence is from case reports
Other than these 2 large studies, most of the evidence addressing the relationship between the use of antipsychotics and incidence of seizures is low quality and relies on case reports or expert opinions. Older studies found that, among FGAs, seizure risk is highest with chlorpromazine and promazine, and lowest with thioridazine and haloperidol.10 As for SGAs, case reports have described seizuresassociated with the use of quetiapine, aripiprazole, risperidone, paliperidone, and olanzapine.
Quetiapine. Three case reports published between 2002 and 2010 describe generalized
Aripiprazole. Five case reports described staring spells and tonic-clonic seizures in patients receiving 10 to 15 mg of aripiprazole.15-19 In the New Drug Application (NDA) for aripiprazole, the incidence of seizures was estimated to be .11% (1 of 926 patients) in placebo-controlled trials and .46% (3 of 859 patients) in haloperidol-controlled trials.20
Risperidone’s product labeling suggests the drug should be used with caution in patients with a history of seizures or conditions that could result in a lower seizure threshold. In Phase III placebo-controlled trials, seizures occurred in .3% of patients treated with risperidone, although in some cases, the seizures were induced by electrolyte disturbances such as hyponatremia.21 Gonzalez-Heydrich et al22 and Holzhausen et al23 found no increase in seizure activity among patients with epilepsy who were receiving risperidone. Lane et al24 published a case report of a geriatric woman who presented with a generalized tonic-clonic seizure related to rapid titration of risperidone; however, with slower titration and lower doses, she stopped having seizures without adding any antiepileptic drugs. Komossa et al25 found that risperidone is less epileptogenic than clozapine, with a relative risk of .22.
Paliperidone is the active metabolite of risperidone and does not have pharmacokinetic interactions with drugs metabolized by the cytochrome P450 (CYP) enzymes. Its labeling indicates that the drug should be used with caution in patients with a history of seizures.26 In Phase III placebo-controlled trials of paliperidone, the rate of seizures was .22%.27 Two case reports suggest close monitoring of seizure risk in patients receiving paliperidone.28,29 Liang et al29 reported that co-administration of valproic acid could mask an underlying decrease of the seizure threshold caused by antipsychotics such as paliperidone.
Continue to: Olanzapine
Olanzapine is a thienobenzodiazepine derivative and is chemically related to clozapine.30 The olanzapine NDA31 shows that 23 of 3,139 patients developed seizures, mainly tonic-clonic, with evidence suggesting that the seizures may have been due to confounding factors such as a history of seizures or metabolic abnormalities. There were no statistically significant differences in the rate of seizures associated with olanzapine compared with placebo or haloperidol (P = .252 and .168, respectively).

A literature review for olanzapine yielded 1 case report of repetitive focal seizures and lingual dystonia,32 5 case reports of generalized tonic-clonic seizures and myoclonus,33-37 and 2 case reports of status epilepticus.38,39 Olanzapine’s clearance is 25% to 30% lower in women, and most of these case reports occurred women.40

Details of the above case reports are summarized in Table 1 (aripiprazole15-19), Table 2 (olanzapine32-39), and Table 3 (paliperidone,28,29 quetiapine,11-13 and risperidone22-24).

Ziprasidone. According to the NDA safety database, the seizure rate attributed to ziprasidone was 1.8 per 100 subject-years or 0.54% of participants (12 of 2,588).41 No additional studies have been published regarding its seizure risk.
Clozapine has a black-box warning
To the best of our knowledge, clozapine is the only antipsychotic that carries an FDA “black-box” warning regarding its risk of inducing seizures.42 Devinsky and Pacia43 reported a cumulative risk of 10% after 3.8 years of treatment. The literature has described clozapine-induced generalized tonic-clonic, myoclonic, simple and complex partial, and absence seizures.44 Table 445 lists the estimated frequency of each seizure type based on 101 cases of clozapine-induced seizures. Myoclonic seizures and drop attacks could be precursors/warning signs of grand mal tonic-clonic seizures.46,47 Seizures have been observed at all stages of treatment, but were more common during initiation of clozapine, which emphasizes the importance of a progressive and slow titration.43,48 The incidence of seizures was estimated to be 6% in a sample of 216 patients with schizophrenia with no history of epilepsy who were prescribed clozapine.49
Continue to: Regarding a possible association between...
Regarding a possible association between clozapine dose or clozapine plasma levels and seizure risk, there is a positive linear relationship between the dose of clozapine and its serum concentration over a dosing range of 25 to 800 mg/d.50 However, the plasma concentration is also significantly affected by factors such as smoking, gender, age, drug interactions, and CYP genotypes. Therefore, the same clozapine dose will yield a lower serum concentration in an older male who smokes compared with a younger, non-smoking female.51 Perry et al52 suggested a dosing nomogram to calculate the influence of gender and smoking. Seizure risk, especially for tonic-clonic seizures, has been reported to increase with clozapine doses >600 mg/d,53 and with plasma concentrations exceeding 1,000 to 1,300 mg/L.54 However, in a 2011 regression analysis, Varma et al55 found no statistically significant relationship between seizure risk and clozapine oral dose; there was not enough data to test a correlation between clozapine plasma levels and the incidence of seizures.
How antipsychotics might lower the seizure threshold
Researchers have suggested several possible mechanisms to explain how antipsychotics might lower the seizure threshold. Antagonism of dopamine D4, histamine H1, and acetylcholine-muscarinic receptors seems to induce EEG alterations and increase the risk of seizures.56 Additionally, modulation of the N-methyl-
Watch for pharmacokinetic interactions
The CYP enzymes involved in drug metabolism include CYP1A2, CYP2C9, CYP2C19, CYP2D6, and CYP3A4. Most commonly used antiepileptics and antipsychotics are metabolized by CYP enzymes, and may also act as inhibitors or inducers of these enzymes.61 Drug interactions may impair seizure control, which is why monotherapy is preferable to combination treatment in patients with epilepsy.62 Carbamazepine and phenytoin are inducers of both CYP1A2 (which metabolizes olanzapine and clozapine), and CYP3A4 (which metabolizes haloperidol, risperidone, quetiapine, ziprasidone and clozapine). Paliperidone is not metabolized by CYP enzymes.62 Discontinuing an enzyme-inducing agent may result in increased antipsychotic plasma concentrations, which might lead to an increased risk of seizures.
Valproic acid, which is often used to prevent or treat clozapine-induced seizures, has an unclear effect on clozapine plasma concentrations.63 Although valproic acid is known to inhibit clozapine metabolism, 2 reports have suggested that the plasma concentrations of clozapine and its metabolites may decrease after adding valproic acid.64,65 Other studies have found that valproic acid increases plasma concentrations of clozapine while it decreases plasma concentrations of norclozapine; norclozapine is the main clozapine metabolite responsible for inducing seizures.66,67
Steps for minimizing seizure risk
Determining the seizure risk for a patient taking an antipsychotic is challenging because doing so depends not only on the seizurogenic potential of each drug but also on individualized predisposing factors.11,57,68 Choosing the “best” antipsychotic therefore largely depends on each patient’s profile. The predisposing factors consist mainly of the individually inherited seizure threshold (personal history of febrile convulsions or a family history of seizures) and other comorbid seizurogenic conditions, such as a history of head trauma, brain injury, intellectual disability, cerebral arteriosclerosis, neurodegenerative diseases, encephalopathy, chronic renal insufficiency, and hyponatremia. Furthermore, seizure risk depends on the antipsychotic dose administered and the rate of titration.11
Continue to: There is not enough evidence...
There is not enough evidence to recommend performing an EEG in all patients taking antipsychotics. Such testing is recommended only for patients who have predisposing factors for seizures. If an EEG shows any abnormality in a patient taking clozapine, consider decreasing the clozapine dose69,70 or adding an antiepileptic drug such as valproic acid or lamotrigine.44,70
Although clozapine carries a black-box warning of increased risk of causing seizures, there is no consensus regarding the efficacy of co-prescribing an antiepileptic. Some studies have suggested prescribing valproic acid prophylactically,71 after the occurrence of 1 seizure,59 or after 2 seizures.54,72 Others have recommended prescribing prophylactic valproic acid for patients taking ≥600 mg/d of clozapine or whose clozapine plasma levels are >500 mg/L.73 Varma et al55 recommended starting an antiepileptic medication if there are clear epileptiform discharges on EEG, if the patient develops stuttering or speech difficulties, or if seizures occur. Liukkonen et al72 advised initiating an antiepileptic at the start of clozapine treatment in patients who are taking other epileptogenic medications, patients with pre-existing seizure disorder, and patients with neurologic abnormalities. On the other hand, Caetano51 argued against primary prevention of seizures for patients receiving >600 mg/d of clozapine, suggesting that “the risk of seizures would be better managed by close clinical monitoring and measures of clozapine serum concentration rather than adding an anticonvulsant drug.”
Current recommendations for primary and secondary prevention of clozapine-induced seizures are detailed in Table 5.42,44,45,51,55,57,69,74,75

Studies addressing the seizurogenic potential of SGAs other than clozapine have a low level of evidence and include patients who had comorbid conditions and were taking other medications that could cause seizures. Additionally, clinical trials of SGAs rarely include patients with seizure disorders; this might underestimate the risk of seizures.4
The effect of the mental illness itself on the seizure threshold needs to be considered.43 Bloechlinger et al8 found that dementia might be inherently associated with a higher risk of antipsychotic-related seizures. Moreover, numerous qualitative EEG studies have found abnormalities in 20% to 60% of patients with schizophrenia.56 Other quantitative studies have reported mild and nonspecific EEG abnormalities, such as increased delta and/or theta activity, in many non-medicated patients with schizophrenia.10,76 Additionally, brain tissue analysis of deceased patients who had schizophrenia has shown a significant increase in dopamine concentrations in the left amygdala compared with controls, and this might be responsible for enhanced electrical activity in this region.10 Some studies have described EEG slowing in the frontal brain regions of patients with schizophrenia,77 and was selectively normalized in these areas with antipsychotics.78
As always, start low, go slow
Mounting evidence suggests that antipsychotic medications decrease the seizure threshold. Practitioners should thus be cautious in prescribing antipsychotics and should target reaching the minimal effective dose with slow titration, especially in patients with predisposing factors for epilepsy.
Continue to: Although evidence suggests...
Although evidence suggests antipsychotics can induce different types of epileptic seizures, the quality of this evidence is low. Randomized controlled trials are needed to determine which antipsychotics increase seizure risk and whether there is a dose-effect relationship.
Bottom Line
Among second-generation antipsychotics, clozapine appears to increase the risk of clinical seizure the most. Correlations with dosage and/or plasma levels have not been proven. Psychiatrists should be vigilant for pharmacokinetic interactions between antipsychotics and antiepileptics, notably via CYP1A2 and CYP3A4.
Related Resources
- Druschky K, Bleich S, Grohmann R, et al. Seizure rates under treatment with antipsychotic drugs: Data from the AMSP project. World J Biol Psychiatry. 2018;15:1-10.
- Epilepsy Foundation. For professionals: Antipsychotics. https://www.epilepsy.com/learn/professionals/diagnosistreatment/psychotropic-drugs-developmental-disabilities/comorbid-5.
Drug Brand Names
Aripiprazole • Abilify
Benztropine • Cogentin
Bethanechol • Duvoid
Carbamazepine • Carbatrol, Tegretol
Chlorpromazine • Thorazine
Cimetidine • Tagamet
Ciprofloxacin • Cipro
Citalopram • Celexa
Clonazepam • Klonopin
Clozapine • Clozaril
Donepezil • Aricept
Enalapril • Vasotec
Erythromycin • Erythrocin
Escitalopram • Lexapro
Flunitrazepam • Rohypnol
Fluvoxamine • Luvox
Gabapentin • Neurontin
Haloperidol • Haldol
Lamotrigine • Lamictal
Lithium • Eskalith, Lithobid
Metformin • Fortamet, Glucophage
Mirtazapine • Remeron
Nitrofurantoin • Furadantin
Olanzapine • Zyprexa
Paliperidone • Invega
Phenobarbital • Luminal
Phenytoin • Dilantin
Prochlorperazine • Compazine
Procyclidine • Kemadrin
Propranolol • Inderal
Quetiapine • Seroquel
Risperidone • Risperdal
Sertraline • Zoloft
Simvastatin • Zocor
Sulfamethoxazole/trimethoprim • Bactrim, Sulfatrim
Topiramate • Topamax
Trifluoperazine • Stelazine
Valproic acid • Depakene, Depakote
Ziprasidone • Geodon
Antipsychotics, especially second-generation antipsychotics (SGAs), have been proven effective for treating psychosis as well as mood disorders.1,2 Because antipsychotics can lower the epileptogenic threshold, seizures are a serious potential adverse effect. Antipsychotics can cause isolated EEG abnormalities in 7% of patients with no history of epilepsy, and clinical seizures in .5% to 1.2% of such patients.3 Additionally, the neuropathophysiology underlying epilepsy can predispose patients to psychiatric disorders4; the estimated prevalence of psychosis in patients with epilepsy is approximately 7%.5 This review will shed light on the risk of clinical seizures related to antipsychotics.
Comparing seizure risk among antipsychotics
In a review of the World Health Organization’s adverse drug reactions database, Kumlien and Lundberg6 calculated the ratio of the number of reports of seizures to the total number of reports for each drug. They found that approximately 9% of all adverse drug reaction reports involving clozapine were due to seizures. Equivalent ratios were 5.90% for quetiapine, 4.91% for olanzapine, 3.68% for risperidone, 3.27% for haloperidol, and 2.59% for aripiprazole. Using the database of the Pharmacovigilance Unit of the Basque Country, Lertxundi et al7 reported a 3.2-fold increased risk of seizure with SGAs in comparison with first-generation antipsychotics (FGAs) (95% confidence interval [CI], 2.21 to 4.63), which went down to 2.08 (CI, 1.39 to 3.12) once clozapine was excluded. However, as the authors of both studies noted, the quality and relevance of this data are limited because it relies on spontaneous reporting.
Overall, the evidence regarding the seizure risk associated with antipsychotics is scarce. To the best of our knowledge, only 2 large observational studies have compared the seizure risks associated with different antipsychotics.
Using data from the UK-based Clinical Practice Research Datalink between 1998 and 2013, Bloechlinger et al8 examined the incidence rates of seizures among patients newly diagnosed with schizophrenia, affective disorders, or dementia who were prescribed antipsychotics. They excluded patients with a history of seizures or antiepileptic use. In the cohort of 60,121 patients, the incidence rates of seizures per 10,000 person-years were 11.7 (CI, 10.0 to 13.4) for those who did not use antipsychotics, 12.4 (CI, 10.9 to 13.8) for past users, 115.4 (CI, 50.1 to 180.7) for current users of haloperidol, 48.8 (CI, 30.7 to 66.9) for current users of quetiapine, 25.9 (CI, 11.8 to 40.0) for current users of risperidone, and 19.0 (CI, 8.7 to 29.3) for current users of olanzapine. No data were available about clozapine use.
In subsequent analyses, the authors found that among patients with affective disorders, only current use of medium- to high-potency FGAs (haloperidol, prochlorperazine, and trifluoperazine) was associated with a significantly increased risk of seizures (adjusted odds ratio: 2.51, CI, 1.51 to 4.18) compared with non-users.8 Among patients with dementia, current use of olanzapine or quetiapine and current use of any FGAs were associated with significantly increased odds of seizures. This study suggests that the underlying mental illness might modulate the seizure risk associated with antipsychotics.8
Wu et al9 conducted a study based on the National Health Insurance Research Database in Taiwan. They examined the 1-year incidence of new-onset seizures among patients diagnosed with schizophrenia or mood disorders who were new to antipsychotic treatment, and calculated the risk of seizure associated with each antipsychotic in reference to risperidone. They found that those receiving clozapine, thioridazine, and haloperidol were 2 to 3 times more likely to develop seizures than those treated with risperidone; risks associated with the rest of the FGAs were similar to that of risperidone.
The results of these 2 large cohort studies are somewhat concurrent in indicating that, other than clozapine, SGAs incur similar risks of seizures; furthermore, they specify that, contrary to earlier studies,10 haloperidol is associated with significantly higher odds of seizures. While both of these cohort studies controlled for several sociodemographic and clinical confounders, they have several limitations. First, diagnoses of seizures were based on information available in databases, which might be subject to inaccuracies. Second, neither study evaluated the effect of drug dosage and duration of exposure on new-onset seizures.
Continue to: Most evidence is from case reports
Most evidence is from case reports
Other than these 2 large studies, most of the evidence addressing the relationship between the use of antipsychotics and incidence of seizures is low quality and relies on case reports or expert opinions. Older studies found that, among FGAs, seizure risk is highest with chlorpromazine and promazine, and lowest with thioridazine and haloperidol.10 As for SGAs, case reports have described seizuresassociated with the use of quetiapine, aripiprazole, risperidone, paliperidone, and olanzapine.
Quetiapine. Three case reports published between 2002 and 2010 describe generalized
Aripiprazole. Five case reports described staring spells and tonic-clonic seizures in patients receiving 10 to 15 mg of aripiprazole.15-19 In the New Drug Application (NDA) for aripiprazole, the incidence of seizures was estimated to be .11% (1 of 926 patients) in placebo-controlled trials and .46% (3 of 859 patients) in haloperidol-controlled trials.20
Risperidone’s product labeling suggests the drug should be used with caution in patients with a history of seizures or conditions that could result in a lower seizure threshold. In Phase III placebo-controlled trials, seizures occurred in .3% of patients treated with risperidone, although in some cases, the seizures were induced by electrolyte disturbances such as hyponatremia.21 Gonzalez-Heydrich et al22 and Holzhausen et al23 found no increase in seizure activity among patients with epilepsy who were receiving risperidone. Lane et al24 published a case report of a geriatric woman who presented with a generalized tonic-clonic seizure related to rapid titration of risperidone; however, with slower titration and lower doses, she stopped having seizures without adding any antiepileptic drugs. Komossa et al25 found that risperidone is less epileptogenic than clozapine, with a relative risk of .22.
Paliperidone is the active metabolite of risperidone and does not have pharmacokinetic interactions with drugs metabolized by the cytochrome P450 (CYP) enzymes. Its labeling indicates that the drug should be used with caution in patients with a history of seizures.26 In Phase III placebo-controlled trials of paliperidone, the rate of seizures was .22%.27 Two case reports suggest close monitoring of seizure risk in patients receiving paliperidone.28,29 Liang et al29 reported that co-administration of valproic acid could mask an underlying decrease of the seizure threshold caused by antipsychotics such as paliperidone.
Continue to: Olanzapine
Olanzapine is a thienobenzodiazepine derivative and is chemically related to clozapine.30 The olanzapine NDA31 shows that 23 of 3,139 patients developed seizures, mainly tonic-clonic, with evidence suggesting that the seizures may have been due to confounding factors such as a history of seizures or metabolic abnormalities. There were no statistically significant differences in the rate of seizures associated with olanzapine compared with placebo or haloperidol (P = .252 and .168, respectively).

A literature review for olanzapine yielded 1 case report of repetitive focal seizures and lingual dystonia,32 5 case reports of generalized tonic-clonic seizures and myoclonus,33-37 and 2 case reports of status epilepticus.38,39 Olanzapine’s clearance is 25% to 30% lower in women, and most of these case reports occurred women.40

Details of the above case reports are summarized in Table 1 (aripiprazole15-19), Table 2 (olanzapine32-39), and Table 3 (paliperidone,28,29 quetiapine,11-13 and risperidone22-24).

Ziprasidone. According to the NDA safety database, the seizure rate attributed to ziprasidone was 1.8 per 100 subject-years or 0.54% of participants (12 of 2,588).41 No additional studies have been published regarding its seizure risk.
Clozapine has a black-box warning
To the best of our knowledge, clozapine is the only antipsychotic that carries an FDA “black-box” warning regarding its risk of inducing seizures.42 Devinsky and Pacia43 reported a cumulative risk of 10% after 3.8 years of treatment. The literature has described clozapine-induced generalized tonic-clonic, myoclonic, simple and complex partial, and absence seizures.44 Table 445 lists the estimated frequency of each seizure type based on 101 cases of clozapine-induced seizures. Myoclonic seizures and drop attacks could be precursors/warning signs of grand mal tonic-clonic seizures.46,47 Seizures have been observed at all stages of treatment, but were more common during initiation of clozapine, which emphasizes the importance of a progressive and slow titration.43,48 The incidence of seizures was estimated to be 6% in a sample of 216 patients with schizophrenia with no history of epilepsy who were prescribed clozapine.49
Continue to: Regarding a possible association between...
Regarding a possible association between clozapine dose or clozapine plasma levels and seizure risk, there is a positive linear relationship between the dose of clozapine and its serum concentration over a dosing range of 25 to 800 mg/d.50 However, the plasma concentration is also significantly affected by factors such as smoking, gender, age, drug interactions, and CYP genotypes. Therefore, the same clozapine dose will yield a lower serum concentration in an older male who smokes compared with a younger, non-smoking female.51 Perry et al52 suggested a dosing nomogram to calculate the influence of gender and smoking. Seizure risk, especially for tonic-clonic seizures, has been reported to increase with clozapine doses >600 mg/d,53 and with plasma concentrations exceeding 1,000 to 1,300 mg/L.54 However, in a 2011 regression analysis, Varma et al55 found no statistically significant relationship between seizure risk and clozapine oral dose; there was not enough data to test a correlation between clozapine plasma levels and the incidence of seizures.
How antipsychotics might lower the seizure threshold
Researchers have suggested several possible mechanisms to explain how antipsychotics might lower the seizure threshold. Antagonism of dopamine D4, histamine H1, and acetylcholine-muscarinic receptors seems to induce EEG alterations and increase the risk of seizures.56 Additionally, modulation of the N-methyl-
Watch for pharmacokinetic interactions
The CYP enzymes involved in drug metabolism include CYP1A2, CYP2C9, CYP2C19, CYP2D6, and CYP3A4. Most commonly used antiepileptics and antipsychotics are metabolized by CYP enzymes, and may also act as inhibitors or inducers of these enzymes.61 Drug interactions may impair seizure control, which is why monotherapy is preferable to combination treatment in patients with epilepsy.62 Carbamazepine and phenytoin are inducers of both CYP1A2 (which metabolizes olanzapine and clozapine), and CYP3A4 (which metabolizes haloperidol, risperidone, quetiapine, ziprasidone and clozapine). Paliperidone is not metabolized by CYP enzymes.62 Discontinuing an enzyme-inducing agent may result in increased antipsychotic plasma concentrations, which might lead to an increased risk of seizures.
Valproic acid, which is often used to prevent or treat clozapine-induced seizures, has an unclear effect on clozapine plasma concentrations.63 Although valproic acid is known to inhibit clozapine metabolism, 2 reports have suggested that the plasma concentrations of clozapine and its metabolites may decrease after adding valproic acid.64,65 Other studies have found that valproic acid increases plasma concentrations of clozapine while it decreases plasma concentrations of norclozapine; norclozapine is the main clozapine metabolite responsible for inducing seizures.66,67
Steps for minimizing seizure risk
Determining the seizure risk for a patient taking an antipsychotic is challenging because doing so depends not only on the seizurogenic potential of each drug but also on individualized predisposing factors.11,57,68 Choosing the “best” antipsychotic therefore largely depends on each patient’s profile. The predisposing factors consist mainly of the individually inherited seizure threshold (personal history of febrile convulsions or a family history of seizures) and other comorbid seizurogenic conditions, such as a history of head trauma, brain injury, intellectual disability, cerebral arteriosclerosis, neurodegenerative diseases, encephalopathy, chronic renal insufficiency, and hyponatremia. Furthermore, seizure risk depends on the antipsychotic dose administered and the rate of titration.11
Continue to: There is not enough evidence...
There is not enough evidence to recommend performing an EEG in all patients taking antipsychotics. Such testing is recommended only for patients who have predisposing factors for seizures. If an EEG shows any abnormality in a patient taking clozapine, consider decreasing the clozapine dose69,70 or adding an antiepileptic drug such as valproic acid or lamotrigine.44,70
Although clozapine carries a black-box warning of increased risk of causing seizures, there is no consensus regarding the efficacy of co-prescribing an antiepileptic. Some studies have suggested prescribing valproic acid prophylactically,71 after the occurrence of 1 seizure,59 or after 2 seizures.54,72 Others have recommended prescribing prophylactic valproic acid for patients taking ≥600 mg/d of clozapine or whose clozapine plasma levels are >500 mg/L.73 Varma et al55 recommended starting an antiepileptic medication if there are clear epileptiform discharges on EEG, if the patient develops stuttering or speech difficulties, or if seizures occur. Liukkonen et al72 advised initiating an antiepileptic at the start of clozapine treatment in patients who are taking other epileptogenic medications, patients with pre-existing seizure disorder, and patients with neurologic abnormalities. On the other hand, Caetano51 argued against primary prevention of seizures for patients receiving >600 mg/d of clozapine, suggesting that “the risk of seizures would be better managed by close clinical monitoring and measures of clozapine serum concentration rather than adding an anticonvulsant drug.”
Current recommendations for primary and secondary prevention of clozapine-induced seizures are detailed in Table 5.42,44,45,51,55,57,69,74,75

Studies addressing the seizurogenic potential of SGAs other than clozapine have a low level of evidence and include patients who had comorbid conditions and were taking other medications that could cause seizures. Additionally, clinical trials of SGAs rarely include patients with seizure disorders; this might underestimate the risk of seizures.4
The effect of the mental illness itself on the seizure threshold needs to be considered.43 Bloechlinger et al8 found that dementia might be inherently associated with a higher risk of antipsychotic-related seizures. Moreover, numerous qualitative EEG studies have found abnormalities in 20% to 60% of patients with schizophrenia.56 Other quantitative studies have reported mild and nonspecific EEG abnormalities, such as increased delta and/or theta activity, in many non-medicated patients with schizophrenia.10,76 Additionally, brain tissue analysis of deceased patients who had schizophrenia has shown a significant increase in dopamine concentrations in the left amygdala compared with controls, and this might be responsible for enhanced electrical activity in this region.10 Some studies have described EEG slowing in the frontal brain regions of patients with schizophrenia,77 and was selectively normalized in these areas with antipsychotics.78
As always, start low, go slow
Mounting evidence suggests that antipsychotic medications decrease the seizure threshold. Practitioners should thus be cautious in prescribing antipsychotics and should target reaching the minimal effective dose with slow titration, especially in patients with predisposing factors for epilepsy.
Continue to: Although evidence suggests...
Although evidence suggests antipsychotics can induce different types of epileptic seizures, the quality of this evidence is low. Randomized controlled trials are needed to determine which antipsychotics increase seizure risk and whether there is a dose-effect relationship.
Bottom Line
Among second-generation antipsychotics, clozapine appears to increase the risk of clinical seizure the most. Correlations with dosage and/or plasma levels have not been proven. Psychiatrists should be vigilant for pharmacokinetic interactions between antipsychotics and antiepileptics, notably via CYP1A2 and CYP3A4.
Related Resources
- Druschky K, Bleich S, Grohmann R, et al. Seizure rates under treatment with antipsychotic drugs: Data from the AMSP project. World J Biol Psychiatry. 2018;15:1-10.
- Epilepsy Foundation. For professionals: Antipsychotics. https://www.epilepsy.com/learn/professionals/diagnosistreatment/psychotropic-drugs-developmental-disabilities/comorbid-5.
Drug Brand Names
Aripiprazole • Abilify
Benztropine • Cogentin
Bethanechol • Duvoid
Carbamazepine • Carbatrol, Tegretol
Chlorpromazine • Thorazine
Cimetidine • Tagamet
Ciprofloxacin • Cipro
Citalopram • Celexa
Clonazepam • Klonopin
Clozapine • Clozaril
Donepezil • Aricept
Enalapril • Vasotec
Erythromycin • Erythrocin
Escitalopram • Lexapro
Flunitrazepam • Rohypnol
Fluvoxamine • Luvox
Gabapentin • Neurontin
Haloperidol • Haldol
Lamotrigine • Lamictal
Lithium • Eskalith, Lithobid
Metformin • Fortamet, Glucophage
Mirtazapine • Remeron
Nitrofurantoin • Furadantin
Olanzapine • Zyprexa
Paliperidone • Invega
Phenobarbital • Luminal
Phenytoin • Dilantin
Prochlorperazine • Compazine
Procyclidine • Kemadrin
Propranolol • Inderal
Quetiapine • Seroquel
Risperidone • Risperdal
Sertraline • Zoloft
Simvastatin • Zocor
Sulfamethoxazole/trimethoprim • Bactrim, Sulfatrim
Topiramate • Topamax
Trifluoperazine • Stelazine
Valproic acid • Depakene, Depakote
Ziprasidone • Geodon
1. Bruijnzeel D, Suryadevara U, Tandon R. Antipsychotic treatment of schizophrenia: an update. Asian J Psychiatr. 2014;11:3-7.
2. Hrdlicka M, Dudova I. Atypical antipsychotics in the treatment of early-onset schizophrenia. Neuropsychiatr Dis Treat. 2015;11:907-913.
3. Koch-Stoecker S. Antipsychotic drugs and epilepsy: indications and treatment guidelines. Epilepsia. 2002;43(suppl 2):19-24.
4. Alper K, Schwartz KA, Kolts RL, et al. Seizure incidence in psychopharmacological clinical trials: an analysis of Food and Drug Administration (FDA) summary basis of approval reports. Biol Psychiatry. 2007;62(4):345-354.
5. Torta R, Keller R. Behavioral, psychotic, and anxiety disorders in epilepsy: etiology, clinical features, and therapeutic implications. Epilepsia. 1999;40(suppl 10):S2-S20.
6. Kumlien E, Lundberg PO. Seizure risk associated with neuroactive drugs: data from the WHO adverse drug reactions database. Seizure. 2010;19(2):69-73.
7. Lertxundi U, Hernandez R, Medrano J, et al. Antipsychotics and seizures: higher risk with atypicals? Seizure. 2013;22(2):141-143.
8. Bloechliger M, Rüegg S, Jick SS, et al. Antipsychotic drug use and the risk of seizures: follow-up study with a nested case-control analysis. CNS Drugs. 2015;29(7):591-603.
9. Wu CS, Wang SC, Yeh IJ, et al. Comparative risk of seizure with use of first- and second-generation antipsychotics in patients with schizophrenia and mood disorders. J Clin Psychiatry. 2016;77(5):e573-e579.
10. Cold JA, Wells BG, Froemming JH. Seizure activity associated with antipsychotic therapy. [Erratum in DICP. 1990;24(10):1012.] DICP. 1990;24(6):601-606.
11. Hedges DW, Jeppson KG. New-onset seizure associated with quetiapine and olanzapine. Ann Pharmacother. 2002;36(3):437-439.
12. Dogu O, Sevim S, Kaleagasi HS. Seizures associated with quetiapine treatment. Ann Pharmacother. 2003;37(9):1224-1227.
13. Young AC, Kleinschmidt KC, Wax PM. Late-onset seizures associated with quetiapine poisoning. J Med Toxicol. 2009;5(1):24-26.
14. US Food and Drug Administration. Recommendation of approvable action for quetiapine fumarate extended release (Seroquel® XR) for the treatment of schizophrenia. http://www.accessdata.fda.gov/drugsatfda_docs/nda/2007/022047Orig1s000MedR.pdf. April 24, 2007. Accessed January 28, 2019.
15. Malik AR, Ravasia S. Aripiprazole-induced seizure. Can J Psychiatry. 2005;50(3):186.
16. Tsai JF. Aripiprazole-associated seizure. J Clin Psychiatry. 2006;67(6):995-996.
17. Arora M, Arndorfer L. EEG abnormalities in a patient taking aripiprazole. Psychiatry (Edgmont). 2007;4(7):18-19.
18. Yueh CL, Yu SL, Chen HM, et al. Aripiprazole-induced seizure: a second case report. BMJ case reports. 2009;2009:bcr03.2009.1693. doi: 10.1136/bcr.03.2009.1693.
19. Thabet FI, Sweis RT, Joseph SA. Aripiprazole-induced seizure in a 3-year-old child: a case report and literature review. Clin Neuropharmacol. 2013;36(1):29-30.
20. US Food and Drug Administration. Abilify (Aripiprazole) tablets. Medical Review Part 2. http://www.accessdata.fda.gov/drugsatfda_docs/nda/2002/21-436_Abilify_medr_P2.pdf. Published March 07, 2003. Accessed January 28, 2019.
21. US Food and Drug Administration. Drugs@FDA: FDA approved drug products. Risperdal tablets, Risperdal oral solution & Risperdal M-tab orally disintegrating tablets. http://www.accessdata.fda.gov/drugsatfda_docs/nda/2003/021444_S004_RISPERDAL_TABLETS.pdf. Published September 10, 2003. Accessed January 28, 2019.
22. Gonzalez-Heydrich J, Pandina GJ, Fleisher CA, et al. No seizure exacerbation from risperidone in youth with comorbid epilepsy and psychiatric disorders: a case series. J Child Adolesc Psychopharmacol. 2004;14(2):295-310.
23. Holzhausen SPF, Guerreiro MM, Baccin CE, et al. Use of risperidone in children with epilepsy. Epilepsy Behav. 2007;10(3):412-416.
24. Lane HY, Chang WH, Chou JC. Seizure during risperidone treatment in an elderly woman treated with concomitant medications. J Clinl Psychiatry. 1998;59(2):81-82.
25. Komossa K, Rummel-Kluge C, Schwarz S, et al. Risperidone versus other atypical antipsychotics for schizophrenia. Cochrane Database Syst Rev. 2011;(1):19:CD006626.
26. Paliperidone [package insert]. Mountainville, CA: Janssen Pharmaceuticals, Inc.; 2007.
27. Brugge, MD; US Food and Drug Administration. Paliperidone OROS oral formulation. http://www.accessdata.fda.gov/drugsatfda_docs/nda/2006/021999s000_MedR_Part4.pdf. Accessed January 28, 2019.
28. Schneider RA, Lizer MH. Apparent seizure and atrial fibrillation associated with paliperidone. Am J Health System Pharm. 2008;65(22):2122-2125.
29. Liang CS, Yang FW, Chiang KT. Paliperidone-associated seizure after discontinuation of sodium valproate: a case report. J Clin Psychopharmacol. 2011;31(2):246-247.
30. Fulton B, Goa KL. Olanzapine. A review of its pharmacological properties and therapeutic efficacy in the management of schizophrenia and related psychoses. Drugs. 1997;53(2):281-298.
31. US Food and Drug Administration. Drugs@FDA: FDA approved drug products: Zyprexa (olanzapine). ORIG-1. http://www.accessdata.fda.gov/drugsatfda_docs/nda/96/020592_Original_Approval_Pkg%20.pdf. Published September 30, 1996. Accessed January 28, 2019.
32. Anzellotti F, Capasso M, Frazzini V, et al. Olanzapine-related repetitive focal seizures with lingual dystonia. Epileptic Disord. 2016;18(1):83-86.
33. Lee JW, Crismon ML, Dorson PG. Seizure associated with olanzapine. Ann Pharmac. 1999;33(5):554-556.
34. Woolley J, Smith S. Lowered seizure threshold on olanzapine. Br J Psychiatry. 2001;178(1):85-86.
35. Behere RV, Anjith D, Rao NP, et al. Olanzapine-induced clinical seizure: a case report. Clin Neuropharmacol. 2009;32(5):297-298.
36. Camacho A, García-Navarro M, Martínez B, et al. Olanzapine-induced myoclonic status. Clin Neuropharmacol. 2005;28(3):145-147.
37. Rosen JB, Milstein MJ, Haut SR. Olanzapine-associated myoclonus. Epilepsy Res. 2012;98(2-3):247-250.
38. Wyderski RJ, Starrett WG, Abou-Saif A. Fatal status epilepticus associated with olanzapine therapy. Ann Pharmacother. 1999;33(7-8):787-789.
39. Spyridi S, Sokolaki S, Nimatoudis J, et al. Status epilepticus in a patient treated with olanzapine and mirtazapine. Int J Clin Pharmacol Ther. 2009;47(2):120-123.
40. Schatzberg AF, Nemeroff CB. Essentials of clinical psychopharmacology. 2nd ed. Arlington, Virginia: American Psychiatric Publishing; 2006.
41. US Food and Drug Administration. Drug approval package: Geodon (Ziprasidone HCI) Capsules. Medical Review Part 2. http://www.accessdata.fda.gov/drugsatfda_docs/nda/2001/20-825_Geodan_medr_P2.pdf. Published February 5, 2001. Accessed January 29, 2019.
42. Clozaril [package insert]. East Hanover, NJ: Novartis; 2008.
43. Devinsky O, Pacia SV. Seizures during clozapine therapy. J Clin Psychiatry. 1994;55(suppl B):153-156.
44. Williams AM, Park SH. Seizure associated with clozapine: incidence, etiology, and management. CNS Drugs. 2015;29(2):101-111.
45. Wong J, Delva N. Clozapine-induced seizures: recognition and treatment. Can J Psychiatry. 2007;52(7):457-463.
46. Berman I, Zalma A, DuRand CJ, et al. Clozapine-induced myoclonic jerks and drop attacks. J Clin Psychiatry. 1992;53(9):329-330.
47. Gouzoulis E, Ozdaglar A, Kasper J. Myoclonic seizures followed by grand mal seizures during clozapine treatment. Am J Psychiatry. 1993;150(7):1128.
48. Sajatovic M, Meltzer HY. Clozapine-induced myoclonus and generalized seizures. Biol Psychiatry. 1996;39(5):367-370.
49. Grover S, Hazari N, Chakrabarti S, et al. Association of clozapine with seizures: a brief report involving 222 patients prescribed clozapine. East Asian Arch Psychiatry. 2015;25(2):73-78.
50. Byerly MJ, DeVane CL. Pharmacokinetics of clozapine and risperidone: a review of recent literature. J Clin Psychopharmacol. 1996;16(2):177-187.
51. Caetano D. Use of anticonvulsants as prophylaxis for seizures in patients on clozapine. Australas Psychiatry. 2014;22(1):78-83.
52. Perry PJ, Bever KA, Arndt S, et al. Relationship between patient variables and plasma clozapine concentrations: a dosing nomogram. Biol Psychiatry.1998;44(8):733-738.
53. Dumortier G, Mahé V, Pons D, et al. Clonic seizure associated with high clozapine plasma level. J Neuropsychiatry Clin Neurosci. 2001;13(2):302-303.
54. Funderburg LG, Vertrees JE, True JE, et al. Seizure following addition of erythromycin to clozapine treatment. Am J Psychiatry. 1994;151(12):1840-1841.
55. Varma S, Bishara D, Besag FMC, et al. Clozapine-related EEG changes and seizures: dose and plasma-level relationships. Ther Adv Psychopharmacol. 2011;1(2):47-66.
56. Amann BL, Pogarell O, Mergl R, et al. EEG abnormalities associated with antipsychotics: a comparison of quetiapine, olanzapine, haloperidol and healthy subjects. Hum Psychopharmacol. 2003;18(8):641-646.
57. Pisani F, Oteri G, Costa C, et al. Effects of psychotropic drugs on seizure threshold. Drug Saf. 2002;25(2):91-110.
58. Maurice T, Phan VL, Urani A, et al. Neuroactive neurosteroids as endogenous effectors for the sigma1 (sigma1) receptor: pharmacological evidence and therapeutic opportunities. Jpn J Pharmacol. 1999;81(2):125-155.
59. Haller E, Binder RL. Clozapine and seizures. Am J Psychiatry. 1990;147(8):1069-1071.
60. Torta R, Monaco F. Atypical antipsychotics and serotoninergic antidepressants in patients with epilepsy: pharmacodynamic considerations. Epilepsia. 2002;43(suppl 2):8-13.
61. Spina E. Drug interactions. In: Shorvon S, Perucca E, Engel J Jr, eds. The treatment of epilepsy. 3rd ed. Oxford, UK: Blackwell Publishing; 2009:361-377.
62. Spina E, Perucca E. Clinical significance of pharmacokinetic interactions between antiepileptic and psychotropic drugs. Epilepsia. 2002;43(suppl 2):37-44.
63. de Leon J, Santoro V, D’Arrigo C, et al. Interactions between antiepileptics and second-generation antipsychotics. Expert Opin Drug Metab Toxicol. 2012;8(3):311-334.
64. Finley P, Warner D. Potential impact of valproic acid therapy on clozapine disposition. Biol Psychiatry. 1994;36(7):487-488.
65. Longo LP, Salzman C. Valproic acid effects on serum concentrations of clozapine and norclozapine. Am J Psychiatry. 1995;152(4):650.
66. Centorrino F, Baldessarini RJ, Kando J, et al. Serum concentrations of clozapine and its major metabolites: effects of cotreatment with fluoxetine or valproate. Am J Psychiatry. 1994;151(1):123-125.
67. Facciolà G, Avenoso A, Scordo MG, et al. Small effects of valproic acid on the plasma concentrations of clozapine and its major metabolites in patients with schizophrenic or affective disorders. Ther Drug Monit. 1999;21(3):341-345.
68. Hyde TM, Weinberger DR. Seizures and schizophrenia. Schizophr Bull. 1997;23(4):611-622.
69. Muzyk A, Gala G, Kahn DA. Use of lamotrigine in a patient with a clozapine-related seizure. J Psychiatr Pract. 2010;16(2):125-128.
70. Kikuchi YS, Sato W, Ataka K, et al. Clozapine-induced seizures, electroencephalography abnormalities, and clinical responses in Japanese patients with schizophrenia. Neuropsychiatr Dis Treat. 2014;10:1973-1978.
71. Taner E, Coşar B, Işik E. Clozapine-induced myoclonic seizures and valproic acid. Int J Psychiatry Clin Pract. 1998;2(1):53-55.
72. Liukkonen J, Koponen HJ, Nousiainen U. Clinical picture and long-term course of epileptic seizures that occur during clozapine treatment. Psychiatry Res. 1992;44(2):107-112.
73. Devinsky O, Honigfeld G, Patin J. Clozapine-related seizures. Neurology. 1991;41(3):369-371.
74. Foster R, Olajide D. A case of clozapine-induced tonic-clonic seizures managed with valproate: implications for clinical care. J Psychopharmacol. 2005;19(1):93-96.
75. Gandelman-Marton R, Theitler J, Klein C, et al. Phenytoin intoxication in a clozapine-related prolonged seizure. J Emerg Med. 2008;35(4):407-409.
76. Primavera A, Giberti L, Scotto P, et al. Nonconvulsive status epilepticus as a cause of confusion in later life: a report of 5 cases. Neuropsychobiology. 1994;30(2-3):148-152.
77. Boutros NN, Arfken C, Galderisi S, et al. The status of spectral EEG abnormality as a diagnostic test for schizophrenia. Schizophrenia Res. 2008;99(1-3):225-237.
78. Takahashi T, Cho RY, Mizuno T, et al. Antipsychotics reverse abnormal EEG complexity in drug-naïve schizophrenia: a multiscale entropy analysis. Neuroimage. 2010;51(1):173-182.
1. Bruijnzeel D, Suryadevara U, Tandon R. Antipsychotic treatment of schizophrenia: an update. Asian J Psychiatr. 2014;11:3-7.
2. Hrdlicka M, Dudova I. Atypical antipsychotics in the treatment of early-onset schizophrenia. Neuropsychiatr Dis Treat. 2015;11:907-913.
3. Koch-Stoecker S. Antipsychotic drugs and epilepsy: indications and treatment guidelines. Epilepsia. 2002;43(suppl 2):19-24.
4. Alper K, Schwartz KA, Kolts RL, et al. Seizure incidence in psychopharmacological clinical trials: an analysis of Food and Drug Administration (FDA) summary basis of approval reports. Biol Psychiatry. 2007;62(4):345-354.
5. Torta R, Keller R. Behavioral, psychotic, and anxiety disorders in epilepsy: etiology, clinical features, and therapeutic implications. Epilepsia. 1999;40(suppl 10):S2-S20.
6. Kumlien E, Lundberg PO. Seizure risk associated with neuroactive drugs: data from the WHO adverse drug reactions database. Seizure. 2010;19(2):69-73.
7. Lertxundi U, Hernandez R, Medrano J, et al. Antipsychotics and seizures: higher risk with atypicals? Seizure. 2013;22(2):141-143.
8. Bloechliger M, Rüegg S, Jick SS, et al. Antipsychotic drug use and the risk of seizures: follow-up study with a nested case-control analysis. CNS Drugs. 2015;29(7):591-603.
9. Wu CS, Wang SC, Yeh IJ, et al. Comparative risk of seizure with use of first- and second-generation antipsychotics in patients with schizophrenia and mood disorders. J Clin Psychiatry. 2016;77(5):e573-e579.
10. Cold JA, Wells BG, Froemming JH. Seizure activity associated with antipsychotic therapy. [Erratum in DICP. 1990;24(10):1012.] DICP. 1990;24(6):601-606.
11. Hedges DW, Jeppson KG. New-onset seizure associated with quetiapine and olanzapine. Ann Pharmacother. 2002;36(3):437-439.
12. Dogu O, Sevim S, Kaleagasi HS. Seizures associated with quetiapine treatment. Ann Pharmacother. 2003;37(9):1224-1227.
13. Young AC, Kleinschmidt KC, Wax PM. Late-onset seizures associated with quetiapine poisoning. J Med Toxicol. 2009;5(1):24-26.
14. US Food and Drug Administration. Recommendation of approvable action for quetiapine fumarate extended release (Seroquel® XR) for the treatment of schizophrenia. http://www.accessdata.fda.gov/drugsatfda_docs/nda/2007/022047Orig1s000MedR.pdf. April 24, 2007. Accessed January 28, 2019.
15. Malik AR, Ravasia S. Aripiprazole-induced seizure. Can J Psychiatry. 2005;50(3):186.
16. Tsai JF. Aripiprazole-associated seizure. J Clin Psychiatry. 2006;67(6):995-996.
17. Arora M, Arndorfer L. EEG abnormalities in a patient taking aripiprazole. Psychiatry (Edgmont). 2007;4(7):18-19.
18. Yueh CL, Yu SL, Chen HM, et al. Aripiprazole-induced seizure: a second case report. BMJ case reports. 2009;2009:bcr03.2009.1693. doi: 10.1136/bcr.03.2009.1693.
19. Thabet FI, Sweis RT, Joseph SA. Aripiprazole-induced seizure in a 3-year-old child: a case report and literature review. Clin Neuropharmacol. 2013;36(1):29-30.
20. US Food and Drug Administration. Abilify (Aripiprazole) tablets. Medical Review Part 2. http://www.accessdata.fda.gov/drugsatfda_docs/nda/2002/21-436_Abilify_medr_P2.pdf. Published March 07, 2003. Accessed January 28, 2019.
21. US Food and Drug Administration. Drugs@FDA: FDA approved drug products. Risperdal tablets, Risperdal oral solution & Risperdal M-tab orally disintegrating tablets. http://www.accessdata.fda.gov/drugsatfda_docs/nda/2003/021444_S004_RISPERDAL_TABLETS.pdf. Published September 10, 2003. Accessed January 28, 2019.
22. Gonzalez-Heydrich J, Pandina GJ, Fleisher CA, et al. No seizure exacerbation from risperidone in youth with comorbid epilepsy and psychiatric disorders: a case series. J Child Adolesc Psychopharmacol. 2004;14(2):295-310.
23. Holzhausen SPF, Guerreiro MM, Baccin CE, et al. Use of risperidone in children with epilepsy. Epilepsy Behav. 2007;10(3):412-416.
24. Lane HY, Chang WH, Chou JC. Seizure during risperidone treatment in an elderly woman treated with concomitant medications. J Clinl Psychiatry. 1998;59(2):81-82.
25. Komossa K, Rummel-Kluge C, Schwarz S, et al. Risperidone versus other atypical antipsychotics for schizophrenia. Cochrane Database Syst Rev. 2011;(1):19:CD006626.
26. Paliperidone [package insert]. Mountainville, CA: Janssen Pharmaceuticals, Inc.; 2007.
27. Brugge, MD; US Food and Drug Administration. Paliperidone OROS oral formulation. http://www.accessdata.fda.gov/drugsatfda_docs/nda/2006/021999s000_MedR_Part4.pdf. Accessed January 28, 2019.
28. Schneider RA, Lizer MH. Apparent seizure and atrial fibrillation associated with paliperidone. Am J Health System Pharm. 2008;65(22):2122-2125.
29. Liang CS, Yang FW, Chiang KT. Paliperidone-associated seizure after discontinuation of sodium valproate: a case report. J Clin Psychopharmacol. 2011;31(2):246-247.
30. Fulton B, Goa KL. Olanzapine. A review of its pharmacological properties and therapeutic efficacy in the management of schizophrenia and related psychoses. Drugs. 1997;53(2):281-298.
31. US Food and Drug Administration. Drugs@FDA: FDA approved drug products: Zyprexa (olanzapine). ORIG-1. http://www.accessdata.fda.gov/drugsatfda_docs/nda/96/020592_Original_Approval_Pkg%20.pdf. Published September 30, 1996. Accessed January 28, 2019.
32. Anzellotti F, Capasso M, Frazzini V, et al. Olanzapine-related repetitive focal seizures with lingual dystonia. Epileptic Disord. 2016;18(1):83-86.
33. Lee JW, Crismon ML, Dorson PG. Seizure associated with olanzapine. Ann Pharmac. 1999;33(5):554-556.
34. Woolley J, Smith S. Lowered seizure threshold on olanzapine. Br J Psychiatry. 2001;178(1):85-86.
35. Behere RV, Anjith D, Rao NP, et al. Olanzapine-induced clinical seizure: a case report. Clin Neuropharmacol. 2009;32(5):297-298.
36. Camacho A, García-Navarro M, Martínez B, et al. Olanzapine-induced myoclonic status. Clin Neuropharmacol. 2005;28(3):145-147.
37. Rosen JB, Milstein MJ, Haut SR. Olanzapine-associated myoclonus. Epilepsy Res. 2012;98(2-3):247-250.
38. Wyderski RJ, Starrett WG, Abou-Saif A. Fatal status epilepticus associated with olanzapine therapy. Ann Pharmacother. 1999;33(7-8):787-789.
39. Spyridi S, Sokolaki S, Nimatoudis J, et al. Status epilepticus in a patient treated with olanzapine and mirtazapine. Int J Clin Pharmacol Ther. 2009;47(2):120-123.
40. Schatzberg AF, Nemeroff CB. Essentials of clinical psychopharmacology. 2nd ed. Arlington, Virginia: American Psychiatric Publishing; 2006.
41. US Food and Drug Administration. Drug approval package: Geodon (Ziprasidone HCI) Capsules. Medical Review Part 2. http://www.accessdata.fda.gov/drugsatfda_docs/nda/2001/20-825_Geodan_medr_P2.pdf. Published February 5, 2001. Accessed January 29, 2019.
42. Clozaril [package insert]. East Hanover, NJ: Novartis; 2008.
43. Devinsky O, Pacia SV. Seizures during clozapine therapy. J Clin Psychiatry. 1994;55(suppl B):153-156.
44. Williams AM, Park SH. Seizure associated with clozapine: incidence, etiology, and management. CNS Drugs. 2015;29(2):101-111.
45. Wong J, Delva N. Clozapine-induced seizures: recognition and treatment. Can J Psychiatry. 2007;52(7):457-463.
46. Berman I, Zalma A, DuRand CJ, et al. Clozapine-induced myoclonic jerks and drop attacks. J Clin Psychiatry. 1992;53(9):329-330.
47. Gouzoulis E, Ozdaglar A, Kasper J. Myoclonic seizures followed by grand mal seizures during clozapine treatment. Am J Psychiatry. 1993;150(7):1128.
48. Sajatovic M, Meltzer HY. Clozapine-induced myoclonus and generalized seizures. Biol Psychiatry. 1996;39(5):367-370.
49. Grover S, Hazari N, Chakrabarti S, et al. Association of clozapine with seizures: a brief report involving 222 patients prescribed clozapine. East Asian Arch Psychiatry. 2015;25(2):73-78.
50. Byerly MJ, DeVane CL. Pharmacokinetics of clozapine and risperidone: a review of recent literature. J Clin Psychopharmacol. 1996;16(2):177-187.
51. Caetano D. Use of anticonvulsants as prophylaxis for seizures in patients on clozapine. Australas Psychiatry. 2014;22(1):78-83.
52. Perry PJ, Bever KA, Arndt S, et al. Relationship between patient variables and plasma clozapine concentrations: a dosing nomogram. Biol Psychiatry.1998;44(8):733-738.
53. Dumortier G, Mahé V, Pons D, et al. Clonic seizure associated with high clozapine plasma level. J Neuropsychiatry Clin Neurosci. 2001;13(2):302-303.
54. Funderburg LG, Vertrees JE, True JE, et al. Seizure following addition of erythromycin to clozapine treatment. Am J Psychiatry. 1994;151(12):1840-1841.
55. Varma S, Bishara D, Besag FMC, et al. Clozapine-related EEG changes and seizures: dose and plasma-level relationships. Ther Adv Psychopharmacol. 2011;1(2):47-66.
56. Amann BL, Pogarell O, Mergl R, et al. EEG abnormalities associated with antipsychotics: a comparison of quetiapine, olanzapine, haloperidol and healthy subjects. Hum Psychopharmacol. 2003;18(8):641-646.
57. Pisani F, Oteri G, Costa C, et al. Effects of psychotropic drugs on seizure threshold. Drug Saf. 2002;25(2):91-110.
58. Maurice T, Phan VL, Urani A, et al. Neuroactive neurosteroids as endogenous effectors for the sigma1 (sigma1) receptor: pharmacological evidence and therapeutic opportunities. Jpn J Pharmacol. 1999;81(2):125-155.
59. Haller E, Binder RL. Clozapine and seizures. Am J Psychiatry. 1990;147(8):1069-1071.
60. Torta R, Monaco F. Atypical antipsychotics and serotoninergic antidepressants in patients with epilepsy: pharmacodynamic considerations. Epilepsia. 2002;43(suppl 2):8-13.
61. Spina E. Drug interactions. In: Shorvon S, Perucca E, Engel J Jr, eds. The treatment of epilepsy. 3rd ed. Oxford, UK: Blackwell Publishing; 2009:361-377.
62. Spina E, Perucca E. Clinical significance of pharmacokinetic interactions between antiepileptic and psychotropic drugs. Epilepsia. 2002;43(suppl 2):37-44.
63. de Leon J, Santoro V, D’Arrigo C, et al. Interactions between antiepileptics and second-generation antipsychotics. Expert Opin Drug Metab Toxicol. 2012;8(3):311-334.
64. Finley P, Warner D. Potential impact of valproic acid therapy on clozapine disposition. Biol Psychiatry. 1994;36(7):487-488.
65. Longo LP, Salzman C. Valproic acid effects on serum concentrations of clozapine and norclozapine. Am J Psychiatry. 1995;152(4):650.
66. Centorrino F, Baldessarini RJ, Kando J, et al. Serum concentrations of clozapine and its major metabolites: effects of cotreatment with fluoxetine or valproate. Am J Psychiatry. 1994;151(1):123-125.
67. Facciolà G, Avenoso A, Scordo MG, et al. Small effects of valproic acid on the plasma concentrations of clozapine and its major metabolites in patients with schizophrenic or affective disorders. Ther Drug Monit. 1999;21(3):341-345.
68. Hyde TM, Weinberger DR. Seizures and schizophrenia. Schizophr Bull. 1997;23(4):611-622.
69. Muzyk A, Gala G, Kahn DA. Use of lamotrigine in a patient with a clozapine-related seizure. J Psychiatr Pract. 2010;16(2):125-128.
70. Kikuchi YS, Sato W, Ataka K, et al. Clozapine-induced seizures, electroencephalography abnormalities, and clinical responses in Japanese patients with schizophrenia. Neuropsychiatr Dis Treat. 2014;10:1973-1978.
71. Taner E, Coşar B, Işik E. Clozapine-induced myoclonic seizures and valproic acid. Int J Psychiatry Clin Pract. 1998;2(1):53-55.
72. Liukkonen J, Koponen HJ, Nousiainen U. Clinical picture and long-term course of epileptic seizures that occur during clozapine treatment. Psychiatry Res. 1992;44(2):107-112.
73. Devinsky O, Honigfeld G, Patin J. Clozapine-related seizures. Neurology. 1991;41(3):369-371.
74. Foster R, Olajide D. A case of clozapine-induced tonic-clonic seizures managed with valproate: implications for clinical care. J Psychopharmacol. 2005;19(1):93-96.
75. Gandelman-Marton R, Theitler J, Klein C, et al. Phenytoin intoxication in a clozapine-related prolonged seizure. J Emerg Med. 2008;35(4):407-409.
76. Primavera A, Giberti L, Scotto P, et al. Nonconvulsive status epilepticus as a cause of confusion in later life: a report of 5 cases. Neuropsychobiology. 1994;30(2-3):148-152.
77. Boutros NN, Arfken C, Galderisi S, et al. The status of spectral EEG abnormality as a diagnostic test for schizophrenia. Schizophrenia Res. 2008;99(1-3):225-237.
78. Takahashi T, Cho RY, Mizuno T, et al. Antipsychotics reverse abnormal EEG complexity in drug-naïve schizophrenia: a multiscale entropy analysis. Neuroimage. 2010;51(1):173-182.
Antidepressants may be best add-on to antipsychotics in schizophrenia
Antidepressants could be the best adjunctive treatment for adult outpatients with schizophrenia who are taking a second-generation antipsychotic and need a change in medication, results of an observational study suggest. Patients who added antidepressants to their treatment had a lower risk of psychiatric hospitalization and emergency room visits than did those who tried an alternative antipsychotic, and those who took mood stabilizers and benzodiazepines were significantly more likely to die over 365 days.
Specifically, “the possibility that adjunctive use of gabapentin is associated with increased risk of death raises a serious concern,” wrote T. Scott Stroup, MD, MPH, of the department of psychiatry at Columbia University, New York, and his associates in JAMA Psychiatry.
Often, Dr. Stroup and his associates noted, second-generation antipsychotics often are insufficient to alleviate symptoms and leave patients with functional limitations. said Dr. Stroup, who is also affiliated with the New York State Psychiatric Institute, and his associates.
Using a Medicaid database, the researchers retrospectively tracked 81,921 outpatients with schizophrenia (aged 18-64 years; mean age, 41 years; 46% women) who were treated with a single antipsychotic from 2001-2010. Each patient added an antidepressant (31,117), a benzodiazepine (11,941), a mood stabilizer (12,849), or another second-generation antipsychotic (26,014).
The researchers examined treatment outcomes over a yearlong period after patients began their new treatment and compared the various groups to the reference group (those who began taking an additional antipsychotic medication).
Compared with the reference group, patients who took an antidepressant had a lower risk of psychiatric hospitalization (hazard ratio, 0.84; 95% confidence interval, 0.80-0.88), while the benzodiazepine group had a higher risk (HR, 1.08; 95% CI, 1.02-1.15), and the mood stabilizer group saw no major difference (HR, 0.98; 95% CI, 0.94-1.03). Similar results were found for the risk of psychiatric emergency department visits, compared with the reference group: The HR with the addition of an antidepressant was 0.92 (95% CI, 0.88-0.96), 1.12 with a benzodiazepine (95% CI, 1.07-1.19), and 0.99 with a mood stabilizer (95% CI, 0.94-1.04).
In regard to mortality, the researchers found that mood stabilizers and benzodiazepines stood apart on the risk front with HRs of 1.31 (95% CI, 1.04-1.66) and 1.22 (95% CI, 0.98-1.52), respectively. Among mood stabilizer use, Gabapentin accounted for 1,755 initiations (13.7%) and was associated with 45 deaths (28.0%), the researchers reported. “No other mood stabilizer appeared to be associated with a higher rate of death than the others.”
Dr. Stroup and his associates cited several limitations. One is that the results might not be generalizable because the investigators looked only at patients who were enrolled in the Medicaid program. Nevertheless, “improved pharmacologic treatment of schizophrenia and consequent reduced need for hospitalization and ED visits associated with more antidepressant and less benzodiazepine use would represent a significant benefit for individuals and for public health,” they wrote.
The study authors reported various relationships with drugmakers, including Auspex, Intra-Cellular Therapies, Eli Lilly, Bristol-Myers Squibb, and Merck. The study was funded by a Patient-Centered Outcomes Research Institute award.
SOURCE: Stroup TS et al. JAMA Psychiatry. 2019 Feb 20. doi: 10.1001/jamapsychiatry.2018.4489.
Much of the research into adjunctive therapy in schizophrenia is of poor quality, and other hurdles make it difficult to understand the best treatment approach. The new study links the addition of an antidepressant to a substantial lowering of psychiatric hospitalization risk, compared with initiating another antipsychotic, wrote Donald C. Goff, MD. Previous randomized controlled trials (RCTs) have suggested that adding on antidepressants can moderately reduce symptoms – mainly negative ones – in schizophrenia. The study findings are preliminary and suggest that an RCT is in order.
Dr. Goff disclosed grants from Avanir.
These statements are based on an accompanying editorial by Dr. Goff of New York University (JAMA Psychiatry. 2019 Feb 20. doi: 10.1001/jamapsychiatry.2018.4318).
Much of the research into adjunctive therapy in schizophrenia is of poor quality, and other hurdles make it difficult to understand the best treatment approach. The new study links the addition of an antidepressant to a substantial lowering of psychiatric hospitalization risk, compared with initiating another antipsychotic, wrote Donald C. Goff, MD. Previous randomized controlled trials (RCTs) have suggested that adding on antidepressants can moderately reduce symptoms – mainly negative ones – in schizophrenia. The study findings are preliminary and suggest that an RCT is in order.
Dr. Goff disclosed grants from Avanir.
These statements are based on an accompanying editorial by Dr. Goff of New York University (JAMA Psychiatry. 2019 Feb 20. doi: 10.1001/jamapsychiatry.2018.4318).
Much of the research into adjunctive therapy in schizophrenia is of poor quality, and other hurdles make it difficult to understand the best treatment approach. The new study links the addition of an antidepressant to a substantial lowering of psychiatric hospitalization risk, compared with initiating another antipsychotic, wrote Donald C. Goff, MD. Previous randomized controlled trials (RCTs) have suggested that adding on antidepressants can moderately reduce symptoms – mainly negative ones – in schizophrenia. The study findings are preliminary and suggest that an RCT is in order.
Dr. Goff disclosed grants from Avanir.
These statements are based on an accompanying editorial by Dr. Goff of New York University (JAMA Psychiatry. 2019 Feb 20. doi: 10.1001/jamapsychiatry.2018.4318).
Antidepressants could be the best adjunctive treatment for adult outpatients with schizophrenia who are taking a second-generation antipsychotic and need a change in medication, results of an observational study suggest. Patients who added antidepressants to their treatment had a lower risk of psychiatric hospitalization and emergency room visits than did those who tried an alternative antipsychotic, and those who took mood stabilizers and benzodiazepines were significantly more likely to die over 365 days.
Specifically, “the possibility that adjunctive use of gabapentin is associated with increased risk of death raises a serious concern,” wrote T. Scott Stroup, MD, MPH, of the department of psychiatry at Columbia University, New York, and his associates in JAMA Psychiatry.
Often, Dr. Stroup and his associates noted, second-generation antipsychotics often are insufficient to alleviate symptoms and leave patients with functional limitations. said Dr. Stroup, who is also affiliated with the New York State Psychiatric Institute, and his associates.
Using a Medicaid database, the researchers retrospectively tracked 81,921 outpatients with schizophrenia (aged 18-64 years; mean age, 41 years; 46% women) who were treated with a single antipsychotic from 2001-2010. Each patient added an antidepressant (31,117), a benzodiazepine (11,941), a mood stabilizer (12,849), or another second-generation antipsychotic (26,014).
The researchers examined treatment outcomes over a yearlong period after patients began their new treatment and compared the various groups to the reference group (those who began taking an additional antipsychotic medication).
Compared with the reference group, patients who took an antidepressant had a lower risk of psychiatric hospitalization (hazard ratio, 0.84; 95% confidence interval, 0.80-0.88), while the benzodiazepine group had a higher risk (HR, 1.08; 95% CI, 1.02-1.15), and the mood stabilizer group saw no major difference (HR, 0.98; 95% CI, 0.94-1.03). Similar results were found for the risk of psychiatric emergency department visits, compared with the reference group: The HR with the addition of an antidepressant was 0.92 (95% CI, 0.88-0.96), 1.12 with a benzodiazepine (95% CI, 1.07-1.19), and 0.99 with a mood stabilizer (95% CI, 0.94-1.04).
In regard to mortality, the researchers found that mood stabilizers and benzodiazepines stood apart on the risk front with HRs of 1.31 (95% CI, 1.04-1.66) and 1.22 (95% CI, 0.98-1.52), respectively. Among mood stabilizer use, Gabapentin accounted for 1,755 initiations (13.7%) and was associated with 45 deaths (28.0%), the researchers reported. “No other mood stabilizer appeared to be associated with a higher rate of death than the others.”
Dr. Stroup and his associates cited several limitations. One is that the results might not be generalizable because the investigators looked only at patients who were enrolled in the Medicaid program. Nevertheless, “improved pharmacologic treatment of schizophrenia and consequent reduced need for hospitalization and ED visits associated with more antidepressant and less benzodiazepine use would represent a significant benefit for individuals and for public health,” they wrote.
The study authors reported various relationships with drugmakers, including Auspex, Intra-Cellular Therapies, Eli Lilly, Bristol-Myers Squibb, and Merck. The study was funded by a Patient-Centered Outcomes Research Institute award.
SOURCE: Stroup TS et al. JAMA Psychiatry. 2019 Feb 20. doi: 10.1001/jamapsychiatry.2018.4489.
Antidepressants could be the best adjunctive treatment for adult outpatients with schizophrenia who are taking a second-generation antipsychotic and need a change in medication, results of an observational study suggest. Patients who added antidepressants to their treatment had a lower risk of psychiatric hospitalization and emergency room visits than did those who tried an alternative antipsychotic, and those who took mood stabilizers and benzodiazepines were significantly more likely to die over 365 days.
Specifically, “the possibility that adjunctive use of gabapentin is associated with increased risk of death raises a serious concern,” wrote T. Scott Stroup, MD, MPH, of the department of psychiatry at Columbia University, New York, and his associates in JAMA Psychiatry.
Often, Dr. Stroup and his associates noted, second-generation antipsychotics often are insufficient to alleviate symptoms and leave patients with functional limitations. said Dr. Stroup, who is also affiliated with the New York State Psychiatric Institute, and his associates.
Using a Medicaid database, the researchers retrospectively tracked 81,921 outpatients with schizophrenia (aged 18-64 years; mean age, 41 years; 46% women) who were treated with a single antipsychotic from 2001-2010. Each patient added an antidepressant (31,117), a benzodiazepine (11,941), a mood stabilizer (12,849), or another second-generation antipsychotic (26,014).
The researchers examined treatment outcomes over a yearlong period after patients began their new treatment and compared the various groups to the reference group (those who began taking an additional antipsychotic medication).
Compared with the reference group, patients who took an antidepressant had a lower risk of psychiatric hospitalization (hazard ratio, 0.84; 95% confidence interval, 0.80-0.88), while the benzodiazepine group had a higher risk (HR, 1.08; 95% CI, 1.02-1.15), and the mood stabilizer group saw no major difference (HR, 0.98; 95% CI, 0.94-1.03). Similar results were found for the risk of psychiatric emergency department visits, compared with the reference group: The HR with the addition of an antidepressant was 0.92 (95% CI, 0.88-0.96), 1.12 with a benzodiazepine (95% CI, 1.07-1.19), and 0.99 with a mood stabilizer (95% CI, 0.94-1.04).
In regard to mortality, the researchers found that mood stabilizers and benzodiazepines stood apart on the risk front with HRs of 1.31 (95% CI, 1.04-1.66) and 1.22 (95% CI, 0.98-1.52), respectively. Among mood stabilizer use, Gabapentin accounted for 1,755 initiations (13.7%) and was associated with 45 deaths (28.0%), the researchers reported. “No other mood stabilizer appeared to be associated with a higher rate of death than the others.”
Dr. Stroup and his associates cited several limitations. One is that the results might not be generalizable because the investigators looked only at patients who were enrolled in the Medicaid program. Nevertheless, “improved pharmacologic treatment of schizophrenia and consequent reduced need for hospitalization and ED visits associated with more antidepressant and less benzodiazepine use would represent a significant benefit for individuals and for public health,” they wrote.
The study authors reported various relationships with drugmakers, including Auspex, Intra-Cellular Therapies, Eli Lilly, Bristol-Myers Squibb, and Merck. The study was funded by a Patient-Centered Outcomes Research Institute award.
SOURCE: Stroup TS et al. JAMA Psychiatry. 2019 Feb 20. doi: 10.1001/jamapsychiatry.2018.4489.
FROM JAMA PSYCHIATRY
Aripiprazole/clozapine combo more effective than monotherapies
Certain antipsychotic polytherapies, including aripiprazole and clozapine, are associated with a lower risk of psychiatric rehospitalization in patients with schizophrenia, according to Jari Tiihonen, MD, PhD, of the Karolinska Institute in Stockholm, and his associates.
The study population included a total of 62,250 patients from the Finnish Hospital Discharge register who were treated for schizophrenia in the inpatient setting from 1972 to 2014. The median patient age was 45.6 years and the median length of follow-up was 14.1 years. Over the study period, 58.8% of this cohort were readmitted for psychiatric inpatient care, 67.2% used antipsychotic polypharmacy during the follow-up, and 57.5% were exposed to antipsychotic polypharmacy for at least 90 days, Dr. Tiihonen and his associates wrote in JAMA Psychiatry.
The combination of aripiprazole and clozapine was associated with the lowest risk of psychiatric rehospitalization, compared with those who received no therapy (hazard ratio, 0.42, 95% confidence interval, 0.39-0.46). Clozapine alone was the most effective antipsychotic monotherapy (HR, 0.49; 95% CI, 0.47-0.51), and when aripiprazole/clozapine was compared with clozapine alone, the polytherapy was significantly more effective (HR, 0.86; 95% CI, 0.79-0.94).
The difference between aripiprazole/clozapine and clozapine alone was even greater in patients who initially were hospitalized for their first episode of schizophrenia (HR, 0.78; 95% CI, 0.63-0.96). Overall, any antipsychotic polypharmacy was associated with a 7%-13% lower risk of hospitalization, compared with any monotherapy; , the authors noted.
“It should be acknowledged that statements about a preferential use of antipsychotic monotherapy for maintenance treatment of schizophrenia lack evidence, and that currently available evidence – although gathered with few nonrandomized cohort studies that have their own limitations – indicates the opposite,” they concluded.
Dr. Tiihonen and several of his associates reported numerous conflicts of interest.
SOURCE: Tiihonen J et al. JAMA Psychiatry. 2019 Feb 20. doi: 10.1001/jamapsychiatry.2018.4320.
Certain antipsychotic polytherapies, including aripiprazole and clozapine, are associated with a lower risk of psychiatric rehospitalization in patients with schizophrenia, according to Jari Tiihonen, MD, PhD, of the Karolinska Institute in Stockholm, and his associates.
The study population included a total of 62,250 patients from the Finnish Hospital Discharge register who were treated for schizophrenia in the inpatient setting from 1972 to 2014. The median patient age was 45.6 years and the median length of follow-up was 14.1 years. Over the study period, 58.8% of this cohort were readmitted for psychiatric inpatient care, 67.2% used antipsychotic polypharmacy during the follow-up, and 57.5% were exposed to antipsychotic polypharmacy for at least 90 days, Dr. Tiihonen and his associates wrote in JAMA Psychiatry.
The combination of aripiprazole and clozapine was associated with the lowest risk of psychiatric rehospitalization, compared with those who received no therapy (hazard ratio, 0.42, 95% confidence interval, 0.39-0.46). Clozapine alone was the most effective antipsychotic monotherapy (HR, 0.49; 95% CI, 0.47-0.51), and when aripiprazole/clozapine was compared with clozapine alone, the polytherapy was significantly more effective (HR, 0.86; 95% CI, 0.79-0.94).
The difference between aripiprazole/clozapine and clozapine alone was even greater in patients who initially were hospitalized for their first episode of schizophrenia (HR, 0.78; 95% CI, 0.63-0.96). Overall, any antipsychotic polypharmacy was associated with a 7%-13% lower risk of hospitalization, compared with any monotherapy; , the authors noted.
“It should be acknowledged that statements about a preferential use of antipsychotic monotherapy for maintenance treatment of schizophrenia lack evidence, and that currently available evidence – although gathered with few nonrandomized cohort studies that have their own limitations – indicates the opposite,” they concluded.
Dr. Tiihonen and several of his associates reported numerous conflicts of interest.
SOURCE: Tiihonen J et al. JAMA Psychiatry. 2019 Feb 20. doi: 10.1001/jamapsychiatry.2018.4320.
Certain antipsychotic polytherapies, including aripiprazole and clozapine, are associated with a lower risk of psychiatric rehospitalization in patients with schizophrenia, according to Jari Tiihonen, MD, PhD, of the Karolinska Institute in Stockholm, and his associates.
The study population included a total of 62,250 patients from the Finnish Hospital Discharge register who were treated for schizophrenia in the inpatient setting from 1972 to 2014. The median patient age was 45.6 years and the median length of follow-up was 14.1 years. Over the study period, 58.8% of this cohort were readmitted for psychiatric inpatient care, 67.2% used antipsychotic polypharmacy during the follow-up, and 57.5% were exposed to antipsychotic polypharmacy for at least 90 days, Dr. Tiihonen and his associates wrote in JAMA Psychiatry.
The combination of aripiprazole and clozapine was associated with the lowest risk of psychiatric rehospitalization, compared with those who received no therapy (hazard ratio, 0.42, 95% confidence interval, 0.39-0.46). Clozapine alone was the most effective antipsychotic monotherapy (HR, 0.49; 95% CI, 0.47-0.51), and when aripiprazole/clozapine was compared with clozapine alone, the polytherapy was significantly more effective (HR, 0.86; 95% CI, 0.79-0.94).
The difference between aripiprazole/clozapine and clozapine alone was even greater in patients who initially were hospitalized for their first episode of schizophrenia (HR, 0.78; 95% CI, 0.63-0.96). Overall, any antipsychotic polypharmacy was associated with a 7%-13% lower risk of hospitalization, compared with any monotherapy; , the authors noted.
“It should be acknowledged that statements about a preferential use of antipsychotic monotherapy for maintenance treatment of schizophrenia lack evidence, and that currently available evidence – although gathered with few nonrandomized cohort studies that have their own limitations – indicates the opposite,” they concluded.
Dr. Tiihonen and several of his associates reported numerous conflicts of interest.
SOURCE: Tiihonen J et al. JAMA Psychiatry. 2019 Feb 20. doi: 10.1001/jamapsychiatry.2018.4320.
FROM JAMA PSYCHIATRY