User login
Antipsychotics show no link to increased risk of major congenital malformations
LAS VEGAS – Assessing the risk of major congenital malformations related to antipsychotic exposure requires detailed assessment of other sources of risk, including those related to the diagnosis and associated behaviors, according to Jonathan M. Meyer, MD.
However, the largest study to date showed no significant difference in rates of major congenital malformations for those with one or more prescriptions for atypical antipsychotics in the first trimester, compared with pregnancies with no first trimester antipsychotic exposure.
In the U.S. general population, the estimated risk of major birth defects is 2%-4%, Dr. Meyer, a clinical professor of psychiatry at the University of California, San Diego, said at an annual psychopharmacology update held by the Nevada Psychiatric Association.
Medications represent one source of risk for major congenital malformations in patients with psychiatric illness. Other factors include lifestyle factors such substance abuse and smoking, diet and physical activity, adherence with medical/prenatal care regimens, general medical disease burden, and unknown genetic risk because of the illness itself.
Until recently, published studies examining antipsychotic exposure and the risk for congenital malformations have been flawed because of numerous factors, Dr. Meyer said, including the small sample size of live births, absence of systematic collection of risk data prior to and during pregnancy, and failure to examine all possible covariates that might moderate the risk potentially attributable to the medication itself.
For example, researchers led by Frank Habermann, PhD, prospectively evaluated three cohorts who were followed in a psychiatry consultation in Freiburg, Germany: 453 women who received atypical antipsychotics in the first trimester of pregnancy (group A); 238 women who received typical antipsychotics in the first trimester of pregnancy (group B); and 1,104 women who had no records of treatment with medications associated with harmful fetal effects (group C).
Covariates included maternal age, alcohol consumption, smoking habits, number of previous spontaneous abortions, number of previous malformed children, and gestational week at delivery (J Clin Psychopharmacol. 2013;33[4]:453-62).
The researchers found that 5.2% of women in group A gave birth to a child with a major congenital malformation, compared with 5% in group B and 2.5% in group C. Nonsignificant associations were observed between group A vs. B (adjusted odds ratio, 1.26; 95% confidence interval, 0.57-2.82) and group B vs. C (adjusted OR, 1.71; 95% CI, 0.78-3.76). The only significant association noted was between group A and C (adjusted OR, 2.17; 95% CI, 1.20-3.91). However, Dr. Meyer emphasized limitations of the study, including its small sample size and certain missing covariates, including illegal substance use.
In addition, since subjects were enrolled in a consultation clinic, surveillance bias might have detected a higher number of CV malformations. “I don’t have a lot of confidence in this study because there were enormous sources of risk that were not controlled for,” said Dr. Meyer, who is also a psychopharmacology consultant for the California State Department of Hospitals.
In a separate study, researchers led by Lee S. Cohen, MD, assessed data from 487 women in the National Pregnancy Registry for Atypical Antipsychotics based at the Massachusetts General Hospital Center for Women’s Health, Boston. Of the 487 women, 353 were on atypical antipsychotics and 134 served as controls (Am J Psychiatry. 2016;73[3]:263-70). Medical records were obtained at baseline, month 7, and postpartum for 82% of subjects, which left 303 women in the final analysis. Covariates included demographic characteristics, medication use and dosage changes, social habits including smoking, use of alcohol and illicit drugs, medical and psychiatric history, and family history of birth defects.
Of 214 live births with first-trimester exposure to atypical antipsychotics, three major malformations were confirmed, while among the control group of 89 women, one major malformation was confirmed. The absolute risk of major malformations was 1.4% for exposed infants and 1.1% for unexposed infants. Meanwhile, the OR for major malformations comparing exposed infants was 1.25, which did not reach statistical significance (95% CI, 0.13-12.19). Limitations of the study include the small sample size of live births and the low overall rate of malformations, said Dr. Meyer. “This group of women living in the Boston area might not be representative of the general population based on the extremely low rates of congenital malformations for both cohorts in this study,” he commented.
In what Dr. Meyer said was the most robust study of its kind to date, researchers led by Krista F. Huybrechts, MS, PhD, drew from Medicaid data from 2000-2010 and included only women who were enrolled from 3 months before their last menstrual period through at least 1 month after delivery (JAMA Psychiatry. 2016;73[9]:938-46). The sample consisted of 1,341,715 pregnancies. Among those pregnancies, 9,258 women filled at least one prescription for an atypical antipsychotic, and 733 filled at least one prescription for a typical antipsychotic, for a total of 9,991 pregnancies. The researchers used propensity score matching to match for risk of antipsychotic exposure. They also balanced the antipsychotic-exposed and nonexposed groups for covariates that might be related to the outcome of interest (major congenital malformations), including (but not limited to) calendar year, age, race, smoking history, multiple gestation, indications for antipsychotic use, other maternal morbidity, concomitant medication use, and general markers of illness burden in the 3 months prior to pregnancy.
The atypical antipsychotics used included quetiapine (n = 4,221), followed by aripiprazole (n = 1,756), risperidone (n = 1,566), olanzapine (n = 1,394), and ziprasidone (n = 697). The absolute risks for congenital malformations per 1,000 live-born infants was 38.2 (95% CI, 26.6-54.7) for those treated with typicals and 44.5 (95% CI, 40.5-48.9) for those treated with atypicals versus 32.7 (95% CI, 32.4-33.0) for untreated women. In the fully adjusted analysis, the risk ratio was not statistically different for those exposed to atypical antipsychotics, compared with the control group, for malformations overall (relative risk, 1.05; 95% CI, 0.96-1.16) nor for cardiac malformations (RR, 1.06; 95% CI, 0.90-1.24). However, the risk remained elevated for risperidone for overall malformations (RR, 1.26; 1.02-1.56) and cardiac malformations (RR, 1.26; 95% CI, 0.88-1.81).
When Dr. Huybrechts and her colleagues redefined exposure as having filled two more prescriptions or having at least a 1-day supply in the first trimester, the results did not meaningfully change. However, the association appeared to strengthen somewhat for risperidone when filling two or more prescriptions (RR, 1.46 for any malformation; 95% CI, 1.01-2.10; RR, 1.87 for cardiac malformations; 95% CI, 1.09-3.19). The researchers observed no evidence of a dose-response relationship for any of the individual antipsychotics except risperidone. Risperidone dosages of 2 mg/day or more were associated with an increased risk for cardiac malformation (RR, 2.08; 95% CI, 1.32-3.28).
“The small increase in absolute risk and RR for malformations observed with risperidone should be interpreted with caution because no apparent biological mechanism can readily explain this outcome and the possibility of a chance finding cannot be ruled out,” the authors wrote in their study. “This finding should therefore be interpreted as a potential safety signal that will require follow-up in other studies.”
If the finding in this study is replicated, Dr. Meyer said, a number needed to harm analysis suggests that compared with no antipsychotic use. “Given the risks of bad outcomes that might occur related to medication switching or nonadherence, the risk-benefit ratio may tilt towards continuing risperidone, especially when a long-acting injectable [LAI] antipsychotic is needed in someone who responds to risperidone and either doesn’t respond to or tolerate the medications available in other LAI preparations such aripiprazole, haloperidol, or fluphenazine,” he said. “This need to balance the risks of exacerbating the mental disorder and the extremely small chance of an adverse pregnancy outcome is part of a clinical discussion one should have with the patient and her family.”
Dr. Meyer reported having received speaking or advising fees from Acadia, Alkermes, Allergan, Intracellular Therapies, Merck, Neurocrine, Otsuka America, Sunovion, and Teva.
LAS VEGAS – Assessing the risk of major congenital malformations related to antipsychotic exposure requires detailed assessment of other sources of risk, including those related to the diagnosis and associated behaviors, according to Jonathan M. Meyer, MD.
However, the largest study to date showed no significant difference in rates of major congenital malformations for those with one or more prescriptions for atypical antipsychotics in the first trimester, compared with pregnancies with no first trimester antipsychotic exposure.
In the U.S. general population, the estimated risk of major birth defects is 2%-4%, Dr. Meyer, a clinical professor of psychiatry at the University of California, San Diego, said at an annual psychopharmacology update held by the Nevada Psychiatric Association.
Medications represent one source of risk for major congenital malformations in patients with psychiatric illness. Other factors include lifestyle factors such substance abuse and smoking, diet and physical activity, adherence with medical/prenatal care regimens, general medical disease burden, and unknown genetic risk because of the illness itself.
Until recently, published studies examining antipsychotic exposure and the risk for congenital malformations have been flawed because of numerous factors, Dr. Meyer said, including the small sample size of live births, absence of systematic collection of risk data prior to and during pregnancy, and failure to examine all possible covariates that might moderate the risk potentially attributable to the medication itself.
For example, researchers led by Frank Habermann, PhD, prospectively evaluated three cohorts who were followed in a psychiatry consultation in Freiburg, Germany: 453 women who received atypical antipsychotics in the first trimester of pregnancy (group A); 238 women who received typical antipsychotics in the first trimester of pregnancy (group B); and 1,104 women who had no records of treatment with medications associated with harmful fetal effects (group C).
Covariates included maternal age, alcohol consumption, smoking habits, number of previous spontaneous abortions, number of previous malformed children, and gestational week at delivery (J Clin Psychopharmacol. 2013;33[4]:453-62).
The researchers found that 5.2% of women in group A gave birth to a child with a major congenital malformation, compared with 5% in group B and 2.5% in group C. Nonsignificant associations were observed between group A vs. B (adjusted odds ratio, 1.26; 95% confidence interval, 0.57-2.82) and group B vs. C (adjusted OR, 1.71; 95% CI, 0.78-3.76). The only significant association noted was between group A and C (adjusted OR, 2.17; 95% CI, 1.20-3.91). However, Dr. Meyer emphasized limitations of the study, including its small sample size and certain missing covariates, including illegal substance use.
In addition, since subjects were enrolled in a consultation clinic, surveillance bias might have detected a higher number of CV malformations. “I don’t have a lot of confidence in this study because there were enormous sources of risk that were not controlled for,” said Dr. Meyer, who is also a psychopharmacology consultant for the California State Department of Hospitals.
In a separate study, researchers led by Lee S. Cohen, MD, assessed data from 487 women in the National Pregnancy Registry for Atypical Antipsychotics based at the Massachusetts General Hospital Center for Women’s Health, Boston. Of the 487 women, 353 were on atypical antipsychotics and 134 served as controls (Am J Psychiatry. 2016;73[3]:263-70). Medical records were obtained at baseline, month 7, and postpartum for 82% of subjects, which left 303 women in the final analysis. Covariates included demographic characteristics, medication use and dosage changes, social habits including smoking, use of alcohol and illicit drugs, medical and psychiatric history, and family history of birth defects.
Of 214 live births with first-trimester exposure to atypical antipsychotics, three major malformations were confirmed, while among the control group of 89 women, one major malformation was confirmed. The absolute risk of major malformations was 1.4% for exposed infants and 1.1% for unexposed infants. Meanwhile, the OR for major malformations comparing exposed infants was 1.25, which did not reach statistical significance (95% CI, 0.13-12.19). Limitations of the study include the small sample size of live births and the low overall rate of malformations, said Dr. Meyer. “This group of women living in the Boston area might not be representative of the general population based on the extremely low rates of congenital malformations for both cohorts in this study,” he commented.
In what Dr. Meyer said was the most robust study of its kind to date, researchers led by Krista F. Huybrechts, MS, PhD, drew from Medicaid data from 2000-2010 and included only women who were enrolled from 3 months before their last menstrual period through at least 1 month after delivery (JAMA Psychiatry. 2016;73[9]:938-46). The sample consisted of 1,341,715 pregnancies. Among those pregnancies, 9,258 women filled at least one prescription for an atypical antipsychotic, and 733 filled at least one prescription for a typical antipsychotic, for a total of 9,991 pregnancies. The researchers used propensity score matching to match for risk of antipsychotic exposure. They also balanced the antipsychotic-exposed and nonexposed groups for covariates that might be related to the outcome of interest (major congenital malformations), including (but not limited to) calendar year, age, race, smoking history, multiple gestation, indications for antipsychotic use, other maternal morbidity, concomitant medication use, and general markers of illness burden in the 3 months prior to pregnancy.
The atypical antipsychotics used included quetiapine (n = 4,221), followed by aripiprazole (n = 1,756), risperidone (n = 1,566), olanzapine (n = 1,394), and ziprasidone (n = 697). The absolute risks for congenital malformations per 1,000 live-born infants was 38.2 (95% CI, 26.6-54.7) for those treated with typicals and 44.5 (95% CI, 40.5-48.9) for those treated with atypicals versus 32.7 (95% CI, 32.4-33.0) for untreated women. In the fully adjusted analysis, the risk ratio was not statistically different for those exposed to atypical antipsychotics, compared with the control group, for malformations overall (relative risk, 1.05; 95% CI, 0.96-1.16) nor for cardiac malformations (RR, 1.06; 95% CI, 0.90-1.24). However, the risk remained elevated for risperidone for overall malformations (RR, 1.26; 1.02-1.56) and cardiac malformations (RR, 1.26; 95% CI, 0.88-1.81).
When Dr. Huybrechts and her colleagues redefined exposure as having filled two more prescriptions or having at least a 1-day supply in the first trimester, the results did not meaningfully change. However, the association appeared to strengthen somewhat for risperidone when filling two or more prescriptions (RR, 1.46 for any malformation; 95% CI, 1.01-2.10; RR, 1.87 for cardiac malformations; 95% CI, 1.09-3.19). The researchers observed no evidence of a dose-response relationship for any of the individual antipsychotics except risperidone. Risperidone dosages of 2 mg/day or more were associated with an increased risk for cardiac malformation (RR, 2.08; 95% CI, 1.32-3.28).
“The small increase in absolute risk and RR for malformations observed with risperidone should be interpreted with caution because no apparent biological mechanism can readily explain this outcome and the possibility of a chance finding cannot be ruled out,” the authors wrote in their study. “This finding should therefore be interpreted as a potential safety signal that will require follow-up in other studies.”
If the finding in this study is replicated, Dr. Meyer said, a number needed to harm analysis suggests that compared with no antipsychotic use. “Given the risks of bad outcomes that might occur related to medication switching or nonadherence, the risk-benefit ratio may tilt towards continuing risperidone, especially when a long-acting injectable [LAI] antipsychotic is needed in someone who responds to risperidone and either doesn’t respond to or tolerate the medications available in other LAI preparations such aripiprazole, haloperidol, or fluphenazine,” he said. “This need to balance the risks of exacerbating the mental disorder and the extremely small chance of an adverse pregnancy outcome is part of a clinical discussion one should have with the patient and her family.”
Dr. Meyer reported having received speaking or advising fees from Acadia, Alkermes, Allergan, Intracellular Therapies, Merck, Neurocrine, Otsuka America, Sunovion, and Teva.
LAS VEGAS – Assessing the risk of major congenital malformations related to antipsychotic exposure requires detailed assessment of other sources of risk, including those related to the diagnosis and associated behaviors, according to Jonathan M. Meyer, MD.
However, the largest study to date showed no significant difference in rates of major congenital malformations for those with one or more prescriptions for atypical antipsychotics in the first trimester, compared with pregnancies with no first trimester antipsychotic exposure.
In the U.S. general population, the estimated risk of major birth defects is 2%-4%, Dr. Meyer, a clinical professor of psychiatry at the University of California, San Diego, said at an annual psychopharmacology update held by the Nevada Psychiatric Association.
Medications represent one source of risk for major congenital malformations in patients with psychiatric illness. Other factors include lifestyle factors such substance abuse and smoking, diet and physical activity, adherence with medical/prenatal care regimens, general medical disease burden, and unknown genetic risk because of the illness itself.
Until recently, published studies examining antipsychotic exposure and the risk for congenital malformations have been flawed because of numerous factors, Dr. Meyer said, including the small sample size of live births, absence of systematic collection of risk data prior to and during pregnancy, and failure to examine all possible covariates that might moderate the risk potentially attributable to the medication itself.
For example, researchers led by Frank Habermann, PhD, prospectively evaluated three cohorts who were followed in a psychiatry consultation in Freiburg, Germany: 453 women who received atypical antipsychotics in the first trimester of pregnancy (group A); 238 women who received typical antipsychotics in the first trimester of pregnancy (group B); and 1,104 women who had no records of treatment with medications associated with harmful fetal effects (group C).
Covariates included maternal age, alcohol consumption, smoking habits, number of previous spontaneous abortions, number of previous malformed children, and gestational week at delivery (J Clin Psychopharmacol. 2013;33[4]:453-62).
The researchers found that 5.2% of women in group A gave birth to a child with a major congenital malformation, compared with 5% in group B and 2.5% in group C. Nonsignificant associations were observed between group A vs. B (adjusted odds ratio, 1.26; 95% confidence interval, 0.57-2.82) and group B vs. C (adjusted OR, 1.71; 95% CI, 0.78-3.76). The only significant association noted was between group A and C (adjusted OR, 2.17; 95% CI, 1.20-3.91). However, Dr. Meyer emphasized limitations of the study, including its small sample size and certain missing covariates, including illegal substance use.
In addition, since subjects were enrolled in a consultation clinic, surveillance bias might have detected a higher number of CV malformations. “I don’t have a lot of confidence in this study because there were enormous sources of risk that were not controlled for,” said Dr. Meyer, who is also a psychopharmacology consultant for the California State Department of Hospitals.
In a separate study, researchers led by Lee S. Cohen, MD, assessed data from 487 women in the National Pregnancy Registry for Atypical Antipsychotics based at the Massachusetts General Hospital Center for Women’s Health, Boston. Of the 487 women, 353 were on atypical antipsychotics and 134 served as controls (Am J Psychiatry. 2016;73[3]:263-70). Medical records were obtained at baseline, month 7, and postpartum for 82% of subjects, which left 303 women in the final analysis. Covariates included demographic characteristics, medication use and dosage changes, social habits including smoking, use of alcohol and illicit drugs, medical and psychiatric history, and family history of birth defects.
Of 214 live births with first-trimester exposure to atypical antipsychotics, three major malformations were confirmed, while among the control group of 89 women, one major malformation was confirmed. The absolute risk of major malformations was 1.4% for exposed infants and 1.1% for unexposed infants. Meanwhile, the OR for major malformations comparing exposed infants was 1.25, which did not reach statistical significance (95% CI, 0.13-12.19). Limitations of the study include the small sample size of live births and the low overall rate of malformations, said Dr. Meyer. “This group of women living in the Boston area might not be representative of the general population based on the extremely low rates of congenital malformations for both cohorts in this study,” he commented.
In what Dr. Meyer said was the most robust study of its kind to date, researchers led by Krista F. Huybrechts, MS, PhD, drew from Medicaid data from 2000-2010 and included only women who were enrolled from 3 months before their last menstrual period through at least 1 month after delivery (JAMA Psychiatry. 2016;73[9]:938-46). The sample consisted of 1,341,715 pregnancies. Among those pregnancies, 9,258 women filled at least one prescription for an atypical antipsychotic, and 733 filled at least one prescription for a typical antipsychotic, for a total of 9,991 pregnancies. The researchers used propensity score matching to match for risk of antipsychotic exposure. They also balanced the antipsychotic-exposed and nonexposed groups for covariates that might be related to the outcome of interest (major congenital malformations), including (but not limited to) calendar year, age, race, smoking history, multiple gestation, indications for antipsychotic use, other maternal morbidity, concomitant medication use, and general markers of illness burden in the 3 months prior to pregnancy.
The atypical antipsychotics used included quetiapine (n = 4,221), followed by aripiprazole (n = 1,756), risperidone (n = 1,566), olanzapine (n = 1,394), and ziprasidone (n = 697). The absolute risks for congenital malformations per 1,000 live-born infants was 38.2 (95% CI, 26.6-54.7) for those treated with typicals and 44.5 (95% CI, 40.5-48.9) for those treated with atypicals versus 32.7 (95% CI, 32.4-33.0) for untreated women. In the fully adjusted analysis, the risk ratio was not statistically different for those exposed to atypical antipsychotics, compared with the control group, for malformations overall (relative risk, 1.05; 95% CI, 0.96-1.16) nor for cardiac malformations (RR, 1.06; 95% CI, 0.90-1.24). However, the risk remained elevated for risperidone for overall malformations (RR, 1.26; 1.02-1.56) and cardiac malformations (RR, 1.26; 95% CI, 0.88-1.81).
When Dr. Huybrechts and her colleagues redefined exposure as having filled two more prescriptions or having at least a 1-day supply in the first trimester, the results did not meaningfully change. However, the association appeared to strengthen somewhat for risperidone when filling two or more prescriptions (RR, 1.46 for any malformation; 95% CI, 1.01-2.10; RR, 1.87 for cardiac malformations; 95% CI, 1.09-3.19). The researchers observed no evidence of a dose-response relationship for any of the individual antipsychotics except risperidone. Risperidone dosages of 2 mg/day or more were associated with an increased risk for cardiac malformation (RR, 2.08; 95% CI, 1.32-3.28).
“The small increase in absolute risk and RR for malformations observed with risperidone should be interpreted with caution because no apparent biological mechanism can readily explain this outcome and the possibility of a chance finding cannot be ruled out,” the authors wrote in their study. “This finding should therefore be interpreted as a potential safety signal that will require follow-up in other studies.”
If the finding in this study is replicated, Dr. Meyer said, a number needed to harm analysis suggests that compared with no antipsychotic use. “Given the risks of bad outcomes that might occur related to medication switching or nonadherence, the risk-benefit ratio may tilt towards continuing risperidone, especially when a long-acting injectable [LAI] antipsychotic is needed in someone who responds to risperidone and either doesn’t respond to or tolerate the medications available in other LAI preparations such aripiprazole, haloperidol, or fluphenazine,” he said. “This need to balance the risks of exacerbating the mental disorder and the extremely small chance of an adverse pregnancy outcome is part of a clinical discussion one should have with the patient and her family.”
Dr. Meyer reported having received speaking or advising fees from Acadia, Alkermes, Allergan, Intracellular Therapies, Merck, Neurocrine, Otsuka America, Sunovion, and Teva.
EXPERT ANALYSIS FROM NPA 2019
Switching long-acting injectables deemed safe in schizophrenia
Patients with schizophrenia who have efficacy or tolerability concerns with paliperidone palmitate or risperidone long-acting injection can be switched safely to aripiprazole lauroxil, a small prospective, open-label study suggests.
“To our knowledge, this is the first prospective study of the safety of switching from other long-acting injectable antipsychotics to [aripiprazole lauroxil],” wrote Brian J. Miller, MD, and his associates.
The 6-month study included 51 patients (mean age, 40.6 years; 72.5% male) who were switched to aripiprazole lauroxil from either of the other long-acting injectables, reported Dr. Miller of Augusta University, Georgia, and his associates. They observed rates of discontinuation for any reason and discontinuation related to the new medication regimen. The study found that, at 6 months, all-cause discontinuation was 30.4% and medication-related discontinuation was 9.2% (Schizophr Res. 2019 Feb 7. doi: 10.1016/jschres.2019.01.38).
Statistically significant improvements were seen with aripiprazole lauroxil based on scores on the Clinical Global Impressions–Severity and the Brief Psychiatric Rating Scale. Safety was assessed by tracking adverse events; the observed adverse events were consistent with aripiprazole lauroxil’s known safety profile. Those improvements and tolerability seen with aripiprazole lauroxil are important, because the reasons for the switch had included experiencing residual symptoms or issues of tolerability with the previous treatment.
“The clinical benefit observed in the study occurred irrespective of the investigator-determined [aripiprazole lauroxil] dosing regimen, suggesting that clinicians have the flexibility to select the regimen that is most compatible with the individual needs of their patients,” the authors added.
The full report can be found in Schizophrenia Research.
Patients with schizophrenia who have efficacy or tolerability concerns with paliperidone palmitate or risperidone long-acting injection can be switched safely to aripiprazole lauroxil, a small prospective, open-label study suggests.
“To our knowledge, this is the first prospective study of the safety of switching from other long-acting injectable antipsychotics to [aripiprazole lauroxil],” wrote Brian J. Miller, MD, and his associates.
The 6-month study included 51 patients (mean age, 40.6 years; 72.5% male) who were switched to aripiprazole lauroxil from either of the other long-acting injectables, reported Dr. Miller of Augusta University, Georgia, and his associates. They observed rates of discontinuation for any reason and discontinuation related to the new medication regimen. The study found that, at 6 months, all-cause discontinuation was 30.4% and medication-related discontinuation was 9.2% (Schizophr Res. 2019 Feb 7. doi: 10.1016/jschres.2019.01.38).
Statistically significant improvements were seen with aripiprazole lauroxil based on scores on the Clinical Global Impressions–Severity and the Brief Psychiatric Rating Scale. Safety was assessed by tracking adverse events; the observed adverse events were consistent with aripiprazole lauroxil’s known safety profile. Those improvements and tolerability seen with aripiprazole lauroxil are important, because the reasons for the switch had included experiencing residual symptoms or issues of tolerability with the previous treatment.
“The clinical benefit observed in the study occurred irrespective of the investigator-determined [aripiprazole lauroxil] dosing regimen, suggesting that clinicians have the flexibility to select the regimen that is most compatible with the individual needs of their patients,” the authors added.
The full report can be found in Schizophrenia Research.
Patients with schizophrenia who have efficacy or tolerability concerns with paliperidone palmitate or risperidone long-acting injection can be switched safely to aripiprazole lauroxil, a small prospective, open-label study suggests.
“To our knowledge, this is the first prospective study of the safety of switching from other long-acting injectable antipsychotics to [aripiprazole lauroxil],” wrote Brian J. Miller, MD, and his associates.
The 6-month study included 51 patients (mean age, 40.6 years; 72.5% male) who were switched to aripiprazole lauroxil from either of the other long-acting injectables, reported Dr. Miller of Augusta University, Georgia, and his associates. They observed rates of discontinuation for any reason and discontinuation related to the new medication regimen. The study found that, at 6 months, all-cause discontinuation was 30.4% and medication-related discontinuation was 9.2% (Schizophr Res. 2019 Feb 7. doi: 10.1016/jschres.2019.01.38).
Statistically significant improvements were seen with aripiprazole lauroxil based on scores on the Clinical Global Impressions–Severity and the Brief Psychiatric Rating Scale. Safety was assessed by tracking adverse events; the observed adverse events were consistent with aripiprazole lauroxil’s known safety profile. Those improvements and tolerability seen with aripiprazole lauroxil are important, because the reasons for the switch had included experiencing residual symptoms or issues of tolerability with the previous treatment.
“The clinical benefit observed in the study occurred irrespective of the investigator-determined [aripiprazole lauroxil] dosing regimen, suggesting that clinicians have the flexibility to select the regimen that is most compatible with the individual needs of their patients,” the authors added.
The full report can be found in Schizophrenia Research.
FROM SCHIZOPHRENIA RESEARCH
Medical students and psychiatry
I have the unfortunate task of trying to teach medical students about psychiatry. I say “unfortunate,” as most of them find psychiatry a difficult art to understand, and they seem reluctant to classify psychiatry as a branch of medicine.
In my efforts to keep things simple, I tell that them psychiatry is one of the most difficult branches of medicine as there are very few objective measures we can rely on to make sense of people’s behavior. Regrettably, the American Psychiatric Association’s Diagnostic and Statistical Manual only seems to confuse them more. So, I remind them that, in medicine, 90%-95% of diagnoses can be obtained from doing a good history, and, if we are lucky a drug level will show drugs in the system, a CT scan without contrast will show cerebral atrophy, or there will be a lab result that will be abnormal and point to a diagnosis. But mostly what they will be seeing is unusual behavior they are unable to classify.
So I identifiable brain damage, psychosis, affective disorders, anxiety disorders, and personality disorders. Under the brain damage category, I include the short- and long-term effects of drugs, major neurocognitive disorders (called dementia before DSM-5), cerebrovascular infarcts, traumatic brain injury, and neurodevelopmental disorders. For their exams and, if they are interested in psychiatry, I tell them to study the DSM. I explain to them that when I was in medical school my dermatology professor told us that if we could recognize the 10 most common dermatologic disorders, we would be able to recognize 90% of the skin disorders we would see. It is similar in psychiatry – thus, my five categories.
However, because I do not want them thinking that only schizophrenia causes psychosis, I let them know that at least 40 different factors cause people to be psychotic indicated by auditory hallucinations. Those 40 factors are: 1) acute alcohol intoxication, 2) alcohol withdrawal, 3) alcoholism, 4) Alzheimer’s disease, 5) benzodiazepine withdrawal, 6) cocaine abuse and addiction, 7) chemical poisoning, 8) dehydration, 9) delirium, 10) dissociative disorders, 11) electrolyte imbalances, 12) encephalopathy of various forms, 13) ecstasy, 14) extreme fatigue, 15) falling asleep, 16) fetal alcohol exposure, 17) grief, 18) hallucinogen use, 19) heroin abuse and dependence, 20) high fever, 21) hyperglycemia, 22) hypoglycemia, 23) intellectual disability, 24) lupus, 25) major depression, 26) mania, 27) methamphetamine use, 28) Parkinson’s disease, 29) phencyclidine, 30) postictal states, 31) posttraumatic stress disorder, 32) schizoid or schizotypal personality disorder, 33) schizophrenia, 34) sleep deprivation, 35) sleep paralysis, 36) solvent abuse, 37) traumatic brain injury, 38) temporal lobe epilepsy, 39) uremia. Lastly, I ask them about No. 40 – “normal” (For example, have you ever been walking down the street and thought you heard someone calling your name, but when you turned around no one was there?). Of course, there are many more causes of psychosis, but keeping it simple makes the principle easier to remember.
Regarding affective disorders, I point out to them, as I did in a previous column, that there is a huge difference between major depressive disorders, unhappiness, or sadness, grief, and demoralization. Regarding anxiety disorders, I let the medical students know that, like personality disorders, there is a lot of comorbidity. Yet, if they can distinguish brain damage, psychosis, and affective disorders from anxiety and personality disorders, that will be good enough.
In keeping with trying to help medical students not make assumptions, I always ask them what’s wrong with people who wash their hands 30 times a day. Invariably, the answer is obsessive-compulsive disorder. So, next I ask: Isn’t it possible that the person who washes his hands 30 times a day is a surgeon – or perhaps a patient with schizophrenia who thinks that Martians are beaming germs to his hands?
I guess I raise this issue because I am concerned with the future of psychiatry, and I think that my approach to medical school education provides a framework that can help students learn how to think about and provide care for psychiatric patients.
Dr. Bell is a staff psychiatrist at Jackson Park Hospital’s Medical/Surgical-Psychiatry Inpatient Unit in Chicago, clinical psychiatrist emeritus in the department of psychiatry at the University of Illinois at Chicago, former president/CEO of the Community Mental Health Council, and former director of the Institute for Juvenile Research (birthplace of child psychiatry), also in Chicago. If you have tricks of the medical school teaching trade that you would like to share, email Dr. Bell at cpnews@mededge.com.
I have the unfortunate task of trying to teach medical students about psychiatry. I say “unfortunate,” as most of them find psychiatry a difficult art to understand, and they seem reluctant to classify psychiatry as a branch of medicine.
In my efforts to keep things simple, I tell that them psychiatry is one of the most difficult branches of medicine as there are very few objective measures we can rely on to make sense of people’s behavior. Regrettably, the American Psychiatric Association’s Diagnostic and Statistical Manual only seems to confuse them more. So, I remind them that, in medicine, 90%-95% of diagnoses can be obtained from doing a good history, and, if we are lucky a drug level will show drugs in the system, a CT scan without contrast will show cerebral atrophy, or there will be a lab result that will be abnormal and point to a diagnosis. But mostly what they will be seeing is unusual behavior they are unable to classify.
So I identifiable brain damage, psychosis, affective disorders, anxiety disorders, and personality disorders. Under the brain damage category, I include the short- and long-term effects of drugs, major neurocognitive disorders (called dementia before DSM-5), cerebrovascular infarcts, traumatic brain injury, and neurodevelopmental disorders. For their exams and, if they are interested in psychiatry, I tell them to study the DSM. I explain to them that when I was in medical school my dermatology professor told us that if we could recognize the 10 most common dermatologic disorders, we would be able to recognize 90% of the skin disorders we would see. It is similar in psychiatry – thus, my five categories.
However, because I do not want them thinking that only schizophrenia causes psychosis, I let them know that at least 40 different factors cause people to be psychotic indicated by auditory hallucinations. Those 40 factors are: 1) acute alcohol intoxication, 2) alcohol withdrawal, 3) alcoholism, 4) Alzheimer’s disease, 5) benzodiazepine withdrawal, 6) cocaine abuse and addiction, 7) chemical poisoning, 8) dehydration, 9) delirium, 10) dissociative disorders, 11) electrolyte imbalances, 12) encephalopathy of various forms, 13) ecstasy, 14) extreme fatigue, 15) falling asleep, 16) fetal alcohol exposure, 17) grief, 18) hallucinogen use, 19) heroin abuse and dependence, 20) high fever, 21) hyperglycemia, 22) hypoglycemia, 23) intellectual disability, 24) lupus, 25) major depression, 26) mania, 27) methamphetamine use, 28) Parkinson’s disease, 29) phencyclidine, 30) postictal states, 31) posttraumatic stress disorder, 32) schizoid or schizotypal personality disorder, 33) schizophrenia, 34) sleep deprivation, 35) sleep paralysis, 36) solvent abuse, 37) traumatic brain injury, 38) temporal lobe epilepsy, 39) uremia. Lastly, I ask them about No. 40 – “normal” (For example, have you ever been walking down the street and thought you heard someone calling your name, but when you turned around no one was there?). Of course, there are many more causes of psychosis, but keeping it simple makes the principle easier to remember.
Regarding affective disorders, I point out to them, as I did in a previous column, that there is a huge difference between major depressive disorders, unhappiness, or sadness, grief, and demoralization. Regarding anxiety disorders, I let the medical students know that, like personality disorders, there is a lot of comorbidity. Yet, if they can distinguish brain damage, psychosis, and affective disorders from anxiety and personality disorders, that will be good enough.
In keeping with trying to help medical students not make assumptions, I always ask them what’s wrong with people who wash their hands 30 times a day. Invariably, the answer is obsessive-compulsive disorder. So, next I ask: Isn’t it possible that the person who washes his hands 30 times a day is a surgeon – or perhaps a patient with schizophrenia who thinks that Martians are beaming germs to his hands?
I guess I raise this issue because I am concerned with the future of psychiatry, and I think that my approach to medical school education provides a framework that can help students learn how to think about and provide care for psychiatric patients.
Dr. Bell is a staff psychiatrist at Jackson Park Hospital’s Medical/Surgical-Psychiatry Inpatient Unit in Chicago, clinical psychiatrist emeritus in the department of psychiatry at the University of Illinois at Chicago, former president/CEO of the Community Mental Health Council, and former director of the Institute for Juvenile Research (birthplace of child psychiatry), also in Chicago. If you have tricks of the medical school teaching trade that you would like to share, email Dr. Bell at cpnews@mededge.com.
I have the unfortunate task of trying to teach medical students about psychiatry. I say “unfortunate,” as most of them find psychiatry a difficult art to understand, and they seem reluctant to classify psychiatry as a branch of medicine.
In my efforts to keep things simple, I tell that them psychiatry is one of the most difficult branches of medicine as there are very few objective measures we can rely on to make sense of people’s behavior. Regrettably, the American Psychiatric Association’s Diagnostic and Statistical Manual only seems to confuse them more. So, I remind them that, in medicine, 90%-95% of diagnoses can be obtained from doing a good history, and, if we are lucky a drug level will show drugs in the system, a CT scan without contrast will show cerebral atrophy, or there will be a lab result that will be abnormal and point to a diagnosis. But mostly what they will be seeing is unusual behavior they are unable to classify.
So I identifiable brain damage, psychosis, affective disorders, anxiety disorders, and personality disorders. Under the brain damage category, I include the short- and long-term effects of drugs, major neurocognitive disorders (called dementia before DSM-5), cerebrovascular infarcts, traumatic brain injury, and neurodevelopmental disorders. For their exams and, if they are interested in psychiatry, I tell them to study the DSM. I explain to them that when I was in medical school my dermatology professor told us that if we could recognize the 10 most common dermatologic disorders, we would be able to recognize 90% of the skin disorders we would see. It is similar in psychiatry – thus, my five categories.
However, because I do not want them thinking that only schizophrenia causes psychosis, I let them know that at least 40 different factors cause people to be psychotic indicated by auditory hallucinations. Those 40 factors are: 1) acute alcohol intoxication, 2) alcohol withdrawal, 3) alcoholism, 4) Alzheimer’s disease, 5) benzodiazepine withdrawal, 6) cocaine abuse and addiction, 7) chemical poisoning, 8) dehydration, 9) delirium, 10) dissociative disorders, 11) electrolyte imbalances, 12) encephalopathy of various forms, 13) ecstasy, 14) extreme fatigue, 15) falling asleep, 16) fetal alcohol exposure, 17) grief, 18) hallucinogen use, 19) heroin abuse and dependence, 20) high fever, 21) hyperglycemia, 22) hypoglycemia, 23) intellectual disability, 24) lupus, 25) major depression, 26) mania, 27) methamphetamine use, 28) Parkinson’s disease, 29) phencyclidine, 30) postictal states, 31) posttraumatic stress disorder, 32) schizoid or schizotypal personality disorder, 33) schizophrenia, 34) sleep deprivation, 35) sleep paralysis, 36) solvent abuse, 37) traumatic brain injury, 38) temporal lobe epilepsy, 39) uremia. Lastly, I ask them about No. 40 – “normal” (For example, have you ever been walking down the street and thought you heard someone calling your name, but when you turned around no one was there?). Of course, there are many more causes of psychosis, but keeping it simple makes the principle easier to remember.
Regarding affective disorders, I point out to them, as I did in a previous column, that there is a huge difference between major depressive disorders, unhappiness, or sadness, grief, and demoralization. Regarding anxiety disorders, I let the medical students know that, like personality disorders, there is a lot of comorbidity. Yet, if they can distinguish brain damage, psychosis, and affective disorders from anxiety and personality disorders, that will be good enough.
In keeping with trying to help medical students not make assumptions, I always ask them what’s wrong with people who wash their hands 30 times a day. Invariably, the answer is obsessive-compulsive disorder. So, next I ask: Isn’t it possible that the person who washes his hands 30 times a day is a surgeon – or perhaps a patient with schizophrenia who thinks that Martians are beaming germs to his hands?
I guess I raise this issue because I am concerned with the future of psychiatry, and I think that my approach to medical school education provides a framework that can help students learn how to think about and provide care for psychiatric patients.
Dr. Bell is a staff psychiatrist at Jackson Park Hospital’s Medical/Surgical-Psychiatry Inpatient Unit in Chicago, clinical psychiatrist emeritus in the department of psychiatry at the University of Illinois at Chicago, former president/CEO of the Community Mental Health Council, and former director of the Institute for Juvenile Research (birthplace of child psychiatry), also in Chicago. If you have tricks of the medical school teaching trade that you would like to share, email Dr. Bell at cpnews@mededge.com.
Can taming inflammation help reduce aggression?
Several psychiatric disorders, including depression, schizophrenia, bipolar disorder, Alzheimer’s disease, traumatic brain injury, autism, and posttraumatic stress disorder, are associated with a dysregulated immune response and elevated levels of inflammatory biomarkers. Inflammation has long been associated with an increased risk of aggressive behavior.1,2 By taming immune system dysregulation, we might be able to more effectively reduce inflammation, and thus reduce aggression, in patients with psychiatric illness.
Inflammation and psychiatric symptoms
An overactivated immune response has been empirically correlated to the development of psychiatric symptoms. Inducing systemic inflammation has adverse effects on cognition and behavior, whereas suppressing inflammation can dramatically improve sensorium and mood. Brain regions involved in arousal and alarm are particularly susceptible to inflammation. Subcortical areas, such as the basal ganglia, and cortical circuits, such as the amygdala and anterior insula, are affected by neuroinflammation. Several modifiable factors, including a diet rich in high glycemic food, improper sleep hygiene, tobacco use, a sedentary lifestyle, obesity, and excess psychosocial stressors, can contribute to systemic inflammation and the development of psychiatric symptoms. Oral diseases, such as tooth decay, periodontitis, and gingivitis, also contribute significantly to overall inflammation.
Anti-inflammatory agents
Using nonsteroidal anti-inflammatory drugs as augmentation to standard treatments has shown promise in several psychiatric illnesses. For example, low-dose aspirin, 81 mg/d, has demonstrated reliable results as an adjunctive treatment for depression.3 Research also has shown that the use of ibuprofen may reduce the chances of individuals seeking psychiatric care.3
Individuals who are at high risk for psychosis and schizophrenia have measurable increases in inflammatory microglial activity.4 The severity of psychotic symptoms corresponds to the magnitude of the immune response; this suggests that neuroinflammation is a risk factor for psychosis, and that anti-inflammatory treatments might help prevent or ameliorate psychosis.
In a double-blind, placebo-controlled study, 70 patients diagnosed with schizophrenia who were taking an antipsychotic were randomized to adjunctive aspirin, 1,000 mg/d, or placebo.5 Participants who received aspirin had significant improvement as measured by changes in Positive and Negative Syndrome Scale total score.5
Targeting C-reactive protein
Inflammation has long been associated with impulsive aggression. C-reactive protein (CRP) is a biomarker produced in the liver in response to inflammatory triggers. In a study of 213 inpatients with schizophrenia, researchers compared 57 patients with higher levels of CRP (>1 mg/dL) with 156 patients with normal levels (<1 mg/dL).2 Compared with patients with normal CRP levels, those with higher levels displayed increased aggressive behavior. Researchers found that the chance of being physically restrained during hospitalization was almost 2.5 times greater for patients with elevated CRP levels on admission compared with those with normal CRP levels.
Statins have long been used to reduce C-reactive peptides in patients with cardiovascular conditions. The use of simvastatin has been shown to significantly reduce negative symptoms in patients with schizophrenia.6
Continue to: Vitamin C also can effectively...
Vitamin C also can effectively lower CRP levels. In a 2-month study, 396 participants with elevated CRP levels received vitamin C, 1,000 mg/d, vitamin E, 800 IU/d, or placebo.7 Although vitamin E didn’t reduce CRP levels, vitamin C reduced CRP by 25.3% compared with placebo. Vitamin C is as effective as statins in controlling this biomarker.
Several nonpharmacologic measures also can help reduce the immune system’s activation of CRP, including increased physical activity, increased intake of low glycemic food and supplemental omega-3 fatty acids, improved dental hygiene, and enhanced sleep.
Using a relatively simple and inexpensive laboratory test for measuring CRP might help predict or stratify the risk of aggressive behavior among psychiatric inpatients. For psychiatric patients with elevated inflammatory markers, the interventions described here may be useful as adjunctive treatments to help reduce aggression and injury in an inpatient setting.
1. Coccaro EF, Lee R, Coussons-Read M. Elevated plasma inflammatory markers in individuals with intermittent explosive disorder and correlation with aggression in humans. JAMA Psychiatry. 2014;71(2):158-165.
2. Barzilay R, Lobel T, Krivoy A, et al. Elevated C-reactive protein levels in schizophrenia inpatients is associated with aggressive behavior. Eur Psychiatry. 2016;31:8-12.
3. Köhler O, Peterson L, Mors O, et al. Inflammation and depression: combined use of selective serotonin reuptake inhibitors and NSAIDs or paracetamol and psychiatric outcomes. Brain and Behavior. 2015;5(8):e00338. doi: 10.1002/brb3.338.
4. Bloomfield PS, Selvaraj S, Veronese M, et al. M icroglial activity in people at ultra high risk of psychosis and in schizophrenia; an [11C]PBR28 PET brain imaging study. Am J Psychiatry. 2016;173(1):44-52.
5. Laan W, Grobbee DE, Selten JP, et al. Adjuvant aspirin therapy reduces symptoms of schizophrenia spectrum disorders: results from a randomized, double-blind, placebo-controlled trial. J Clin Psychiatry. 2010;71(5):520-527.
6. Tajik-Esmaeeli S, Moazen-Zadeh E, Abbasi N, et al. Simvastatin adjunct therapy for negative symptoms of schizophrenia: a randomized double-blind placebo-controlled trial. Int Clin Psychopharmacol. 2017;32(2):87-94.
7. Block G, Jensen CD, Dalvi TB, et al. Vitamin C treatment reduces elevated C-reactive protein. Free Radic Biol Med. 2009;46(1):70-77.
Several psychiatric disorders, including depression, schizophrenia, bipolar disorder, Alzheimer’s disease, traumatic brain injury, autism, and posttraumatic stress disorder, are associated with a dysregulated immune response and elevated levels of inflammatory biomarkers. Inflammation has long been associated with an increased risk of aggressive behavior.1,2 By taming immune system dysregulation, we might be able to more effectively reduce inflammation, and thus reduce aggression, in patients with psychiatric illness.
Inflammation and psychiatric symptoms
An overactivated immune response has been empirically correlated to the development of psychiatric symptoms. Inducing systemic inflammation has adverse effects on cognition and behavior, whereas suppressing inflammation can dramatically improve sensorium and mood. Brain regions involved in arousal and alarm are particularly susceptible to inflammation. Subcortical areas, such as the basal ganglia, and cortical circuits, such as the amygdala and anterior insula, are affected by neuroinflammation. Several modifiable factors, including a diet rich in high glycemic food, improper sleep hygiene, tobacco use, a sedentary lifestyle, obesity, and excess psychosocial stressors, can contribute to systemic inflammation and the development of psychiatric symptoms. Oral diseases, such as tooth decay, periodontitis, and gingivitis, also contribute significantly to overall inflammation.
Anti-inflammatory agents
Using nonsteroidal anti-inflammatory drugs as augmentation to standard treatments has shown promise in several psychiatric illnesses. For example, low-dose aspirin, 81 mg/d, has demonstrated reliable results as an adjunctive treatment for depression.3 Research also has shown that the use of ibuprofen may reduce the chances of individuals seeking psychiatric care.3
Individuals who are at high risk for psychosis and schizophrenia have measurable increases in inflammatory microglial activity.4 The severity of psychotic symptoms corresponds to the magnitude of the immune response; this suggests that neuroinflammation is a risk factor for psychosis, and that anti-inflammatory treatments might help prevent or ameliorate psychosis.
In a double-blind, placebo-controlled study, 70 patients diagnosed with schizophrenia who were taking an antipsychotic were randomized to adjunctive aspirin, 1,000 mg/d, or placebo.5 Participants who received aspirin had significant improvement as measured by changes in Positive and Negative Syndrome Scale total score.5
Targeting C-reactive protein
Inflammation has long been associated with impulsive aggression. C-reactive protein (CRP) is a biomarker produced in the liver in response to inflammatory triggers. In a study of 213 inpatients with schizophrenia, researchers compared 57 patients with higher levels of CRP (>1 mg/dL) with 156 patients with normal levels (<1 mg/dL).2 Compared with patients with normal CRP levels, those with higher levels displayed increased aggressive behavior. Researchers found that the chance of being physically restrained during hospitalization was almost 2.5 times greater for patients with elevated CRP levels on admission compared with those with normal CRP levels.
Statins have long been used to reduce C-reactive peptides in patients with cardiovascular conditions. The use of simvastatin has been shown to significantly reduce negative symptoms in patients with schizophrenia.6
Continue to: Vitamin C also can effectively...
Vitamin C also can effectively lower CRP levels. In a 2-month study, 396 participants with elevated CRP levels received vitamin C, 1,000 mg/d, vitamin E, 800 IU/d, or placebo.7 Although vitamin E didn’t reduce CRP levels, vitamin C reduced CRP by 25.3% compared with placebo. Vitamin C is as effective as statins in controlling this biomarker.
Several nonpharmacologic measures also can help reduce the immune system’s activation of CRP, including increased physical activity, increased intake of low glycemic food and supplemental omega-3 fatty acids, improved dental hygiene, and enhanced sleep.
Using a relatively simple and inexpensive laboratory test for measuring CRP might help predict or stratify the risk of aggressive behavior among psychiatric inpatients. For psychiatric patients with elevated inflammatory markers, the interventions described here may be useful as adjunctive treatments to help reduce aggression and injury in an inpatient setting.
Several psychiatric disorders, including depression, schizophrenia, bipolar disorder, Alzheimer’s disease, traumatic brain injury, autism, and posttraumatic stress disorder, are associated with a dysregulated immune response and elevated levels of inflammatory biomarkers. Inflammation has long been associated with an increased risk of aggressive behavior.1,2 By taming immune system dysregulation, we might be able to more effectively reduce inflammation, and thus reduce aggression, in patients with psychiatric illness.
Inflammation and psychiatric symptoms
An overactivated immune response has been empirically correlated to the development of psychiatric symptoms. Inducing systemic inflammation has adverse effects on cognition and behavior, whereas suppressing inflammation can dramatically improve sensorium and mood. Brain regions involved in arousal and alarm are particularly susceptible to inflammation. Subcortical areas, such as the basal ganglia, and cortical circuits, such as the amygdala and anterior insula, are affected by neuroinflammation. Several modifiable factors, including a diet rich in high glycemic food, improper sleep hygiene, tobacco use, a sedentary lifestyle, obesity, and excess psychosocial stressors, can contribute to systemic inflammation and the development of psychiatric symptoms. Oral diseases, such as tooth decay, periodontitis, and gingivitis, also contribute significantly to overall inflammation.
Anti-inflammatory agents
Using nonsteroidal anti-inflammatory drugs as augmentation to standard treatments has shown promise in several psychiatric illnesses. For example, low-dose aspirin, 81 mg/d, has demonstrated reliable results as an adjunctive treatment for depression.3 Research also has shown that the use of ibuprofen may reduce the chances of individuals seeking psychiatric care.3
Individuals who are at high risk for psychosis and schizophrenia have measurable increases in inflammatory microglial activity.4 The severity of psychotic symptoms corresponds to the magnitude of the immune response; this suggests that neuroinflammation is a risk factor for psychosis, and that anti-inflammatory treatments might help prevent or ameliorate psychosis.
In a double-blind, placebo-controlled study, 70 patients diagnosed with schizophrenia who were taking an antipsychotic were randomized to adjunctive aspirin, 1,000 mg/d, or placebo.5 Participants who received aspirin had significant improvement as measured by changes in Positive and Negative Syndrome Scale total score.5
Targeting C-reactive protein
Inflammation has long been associated with impulsive aggression. C-reactive protein (CRP) is a biomarker produced in the liver in response to inflammatory triggers. In a study of 213 inpatients with schizophrenia, researchers compared 57 patients with higher levels of CRP (>1 mg/dL) with 156 patients with normal levels (<1 mg/dL).2 Compared with patients with normal CRP levels, those with higher levels displayed increased aggressive behavior. Researchers found that the chance of being physically restrained during hospitalization was almost 2.5 times greater for patients with elevated CRP levels on admission compared with those with normal CRP levels.
Statins have long been used to reduce C-reactive peptides in patients with cardiovascular conditions. The use of simvastatin has been shown to significantly reduce negative symptoms in patients with schizophrenia.6
Continue to: Vitamin C also can effectively...
Vitamin C also can effectively lower CRP levels. In a 2-month study, 396 participants with elevated CRP levels received vitamin C, 1,000 mg/d, vitamin E, 800 IU/d, or placebo.7 Although vitamin E didn’t reduce CRP levels, vitamin C reduced CRP by 25.3% compared with placebo. Vitamin C is as effective as statins in controlling this biomarker.
Several nonpharmacologic measures also can help reduce the immune system’s activation of CRP, including increased physical activity, increased intake of low glycemic food and supplemental omega-3 fatty acids, improved dental hygiene, and enhanced sleep.
Using a relatively simple and inexpensive laboratory test for measuring CRP might help predict or stratify the risk of aggressive behavior among psychiatric inpatients. For psychiatric patients with elevated inflammatory markers, the interventions described here may be useful as adjunctive treatments to help reduce aggression and injury in an inpatient setting.
1. Coccaro EF, Lee R, Coussons-Read M. Elevated plasma inflammatory markers in individuals with intermittent explosive disorder and correlation with aggression in humans. JAMA Psychiatry. 2014;71(2):158-165.
2. Barzilay R, Lobel T, Krivoy A, et al. Elevated C-reactive protein levels in schizophrenia inpatients is associated with aggressive behavior. Eur Psychiatry. 2016;31:8-12.
3. Köhler O, Peterson L, Mors O, et al. Inflammation and depression: combined use of selective serotonin reuptake inhibitors and NSAIDs or paracetamol and psychiatric outcomes. Brain and Behavior. 2015;5(8):e00338. doi: 10.1002/brb3.338.
4. Bloomfield PS, Selvaraj S, Veronese M, et al. M icroglial activity in people at ultra high risk of psychosis and in schizophrenia; an [11C]PBR28 PET brain imaging study. Am J Psychiatry. 2016;173(1):44-52.
5. Laan W, Grobbee DE, Selten JP, et al. Adjuvant aspirin therapy reduces symptoms of schizophrenia spectrum disorders: results from a randomized, double-blind, placebo-controlled trial. J Clin Psychiatry. 2010;71(5):520-527.
6. Tajik-Esmaeeli S, Moazen-Zadeh E, Abbasi N, et al. Simvastatin adjunct therapy for negative symptoms of schizophrenia: a randomized double-blind placebo-controlled trial. Int Clin Psychopharmacol. 2017;32(2):87-94.
7. Block G, Jensen CD, Dalvi TB, et al. Vitamin C treatment reduces elevated C-reactive protein. Free Radic Biol Med. 2009;46(1):70-77.
1. Coccaro EF, Lee R, Coussons-Read M. Elevated plasma inflammatory markers in individuals with intermittent explosive disorder and correlation with aggression in humans. JAMA Psychiatry. 2014;71(2):158-165.
2. Barzilay R, Lobel T, Krivoy A, et al. Elevated C-reactive protein levels in schizophrenia inpatients is associated with aggressive behavior. Eur Psychiatry. 2016;31:8-12.
3. Köhler O, Peterson L, Mors O, et al. Inflammation and depression: combined use of selective serotonin reuptake inhibitors and NSAIDs or paracetamol and psychiatric outcomes. Brain and Behavior. 2015;5(8):e00338. doi: 10.1002/brb3.338.
4. Bloomfield PS, Selvaraj S, Veronese M, et al. M icroglial activity in people at ultra high risk of psychosis and in schizophrenia; an [11C]PBR28 PET brain imaging study. Am J Psychiatry. 2016;173(1):44-52.
5. Laan W, Grobbee DE, Selten JP, et al. Adjuvant aspirin therapy reduces symptoms of schizophrenia spectrum disorders: results from a randomized, double-blind, placebo-controlled trial. J Clin Psychiatry. 2010;71(5):520-527.
6. Tajik-Esmaeeli S, Moazen-Zadeh E, Abbasi N, et al. Simvastatin adjunct therapy for negative symptoms of schizophrenia: a randomized double-blind placebo-controlled trial. Int Clin Psychopharmacol. 2017;32(2):87-94.
7. Block G, Jensen CD, Dalvi TB, et al. Vitamin C treatment reduces elevated C-reactive protein. Free Radic Biol Med. 2009;46(1):70-77.
Fever, tachycardia, and tachypnea during a psychotic exacerbation
CASE Posing a threat to his family
Mr. C, age 23, who was diagnosed with schizophrenia with daily auditory hallucinations 4 years earlier, is transferred from an outside psychiatric hospital to our emergency department (ED) after developing fever, tachycardia, headache, and nasal congestion for the past day. He had been admitted to the psychiatric hospital 3 weeks ago due to concerns he was experiencing increased hallucinations and delusions and posed a threat to his sister and her children, with whom he had been living.
Mr. C tells us that while at the psychiatric hospital, he had been started on clozapine, 250 mg/d. He said that prior to clozapine, he had been taking risperidone. We are unable to confirm past treatment information with the psychiatric hospital, including exactly when the clozapine had been started or how fast it had been titrated. We also were not able to obtain information on his prior medication regimen.
In the ED, Mr. C is febrile (39.4°C; 102.9°F), tachycardic (160 beats per minute; reference range 60 to 100), and tachypneic (24 breaths per minute; reference range 12 to 20). His blood pressure is 130/68 mm Hg, and his lactate level is 2.3 mmol/L (reference range <1.9 mmol/L). After he receives 3 liters of fluid, Mr. C’s heart rate decreases to 117 and his lactate level to 1.1 mmol/L. His white blood cell count is 10.6 × 103/mm3 (reference range 4.0 to 10.0 × 103/mm3); a differential can be found in the Table. His electrocardiogram (ECG) demonstrates sinus tachycardia and a QTc of 510 ms (reference range <430 ms), but is otherwise unremarkable. His creatinine kinase (CK) level is within normal limits at 76 U/L (reference range 52 to 336 U/L). A C-reactive protein (CRP) level was not drawn at this time. Other than marijuana and cocaine use, Mr. C’s medical history is unremarkable.
Mr. C is admitted to the hospital and is started on treatment for sepsis. On the evening of Day 1, Mr. C experiences worsening tachycardia (140 beats per minute) and tachypnea (≥40 breaths per minute). His temperature increases to 103.3°F, and his blood pressure drops to 97/55 mm Hg. His troponin level is 19.0 ng/mL (reference range <0.01 ng/mL) and CK level is 491 U/L.
As Mr. C continues to deteriorate, a rapid response is called and he is placed on non-rebreather oxygen and transferred to the medical intensive care unit (MICU).
[polldaddy:10226034]
The authors’ observations
With Mr. C’s presenting symptoms, multiple conditions were included in the differential diagnosis. The initial concern was for sepsis. Sepsis is defined as life-threatening organ dysfunction caused by a dysregulated host response to infection.1 Organ dysfunction is defined by a quick Sepsis-Related Organ Failure Assessment (qSOFA) score ≥2 and is associated with an increased probability of mortality (>10%). Although no infection had been identified in Mr. C, the combination of fever, altered vital signs, and elevated lactate level in the setting of a qSOFA score of 2 (for respiratory rate and blood pressure) raised suspicion enough to start empiric treatment.
With Mr. C’s subsequent deterioration on the evening of Day 1, we considered cardiopulmonary etiologies. His symptoms of dyspnea, hypotension, tachycardia, tachypnea, and fever were nonspecific and thus required consideration of multiple life-threatening etiologies. Thygesen et al2 published an expert consensus of the definition of myocardial infarction, which was of concern given our patient’s elevated troponin level. Because there was already concern for sepsis, the addition of cardiac symptoms required us to consider infectious endocarditis.3 Sudden onset of dyspnea and a drop in blood pressure were concerning for pulmonary embolism, although our patient did not have the usual risk factors (cancer, immobilization, recent surgery, etc.).4 Additionally, in light of Mr. C’s psychiatric history and recent stressors of being moved from his sister’s house and admitted to a psychiatric hospital, coupled with dyspnea and hypotension, we included Takotsubo cardiomyopathy in the differential.5,6 This disease often occurs in response to an emotional or physical stressor and is characterized by transient systolic dysfunction in the setting of ventricular wall-motion abnormalities reaching beyond the distribution of a single coronary artery. Acute ECG and biomarker findings mimic those of myocardial infarction.6
Continue to: Finally, we needed to consider...
Finally, we needed to consider the potential adverse effects of clozapine. Clozapine is a second-generation antipsychotic (SGA) used to treat patients with schizophrenia for whom other antipsychotic medications are ineffective. Clozapine has been shown to be more effective than first-generation antipsychotics (FGA) in reducing symptoms of schizophrenia.7 It has also been shown to be more effective than several SGAs, including quetiapine, risperidone, and olanzapine.7 In fact, in patients with an insufficient therapeutic response to an SGA, clozapine proves to be more effective than switching to a different SGA. As a result of more than 20 years of research, clozapine is the gold-standard for treatment-resistant schizophrenia.7 Yet despite this strong evidence supporting its use in patients with treatment-resistant schizophrenia, the medication continues to be underutilized, especially in patients at risk for suicide.7
It appears that clozapine remains a third-choice medication in the treatment of schizophrenia largely due to its serious adverse effect profile.7 The medication includes several black-box warnings, including severe neutropenia, orthostatic hypotension, bradycardia, syncope, seizures, myocarditis, cardiomyopathy, and mitral valve incompetence.8 Tachycardia, bradycardia, and orthostatic hypotension are all clozapine-related adverse effects associated with autonomic dysfunction, which can result in serious long-term cardiac complications.9 With regards to the drug’s neutropenia risk, the establishment of the Clozapine Risk Evaluation and Mitigation Strategy (REMS) program has allowed for safer use of clozapine and reduced deaths due to clozapine-induced agranulocytosis. Clinicians and pharmacists must be certified in order to prescribe clozapine, and patients must be registered and undergo frequent absolute neutrophil count (ANC) monitoring.
Clozapine-induced myocarditis, a condition observed in up to 3% of patients started on the medication,9 is more likely to develop early on during treatment, with a median time of detection of 16 days following drug initiation.10 Myocarditis often presents with nonspecific signs and symptoms that include chest pain, tachycardia, palpitations, dyspnea, fever, flu-like symptoms, and/or hypotension.
[polldaddy:10226036]
The authors’ observations
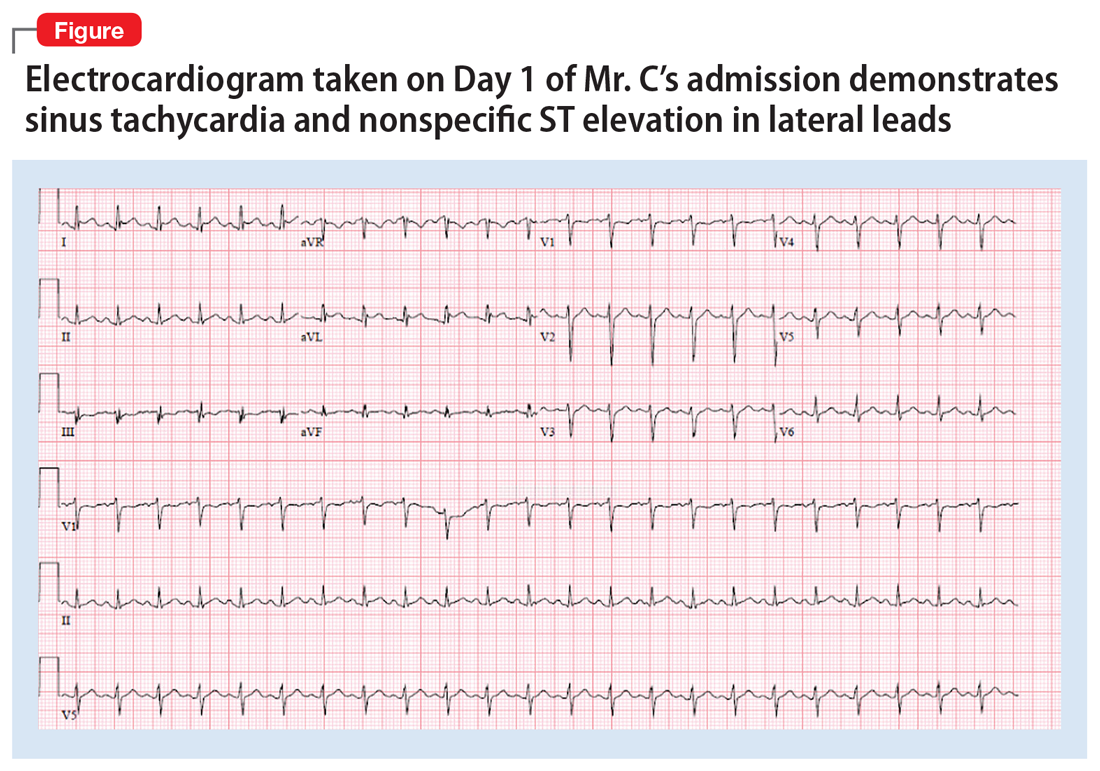
Initial workup in the MICU for Mr. C included an ABG analysis, ECG, and cardiology consult. The ABG analysis demonstrated metabolic alkalosis; his ECG demonstrated sinus tachycardia and nonspecific ST elevation in the lateral leads (Figure). The cardiology consult team started Mr. C on treatment for a non-ST-elevation myocardial infarction (NSTEMI), which it believed to be most likely due to myocarditis with secondary demand ischemia, and less likely acute coronary syndrome. The cardiology consult team also recommended performing a workup for pulmonary emboli and infectious endocarditis if Mr. C’s symptoms persist or the infectious source could not be identified.
EVALUATION Gradual improvement
Mr. C demonstrates gradual improvement as his workup continues, and clozapine is held on the recommendation of the cardiac consult team. By Day 2, he stops complaining of auditory hallucinations, and does not report their return during the rest of his stay. His troponin level decreases to 8.6 ng/mL and lactate level to 1.4 mmol/L; trending is stopped for both. The erythrocyte sedimentation rate (ESR) is elevated at 59 mm/hr (reference range 0 to 22 mm/hr), along with a CRP level of 21 mg/L (reference range <8.0 mg/L). An echocardiogram demonstrates a 40% ejection fraction (reference range 55% to 75%) and moderate global hypokinesis. The cardiology consult team is concerned for Takotsubo cardiomyopathy with sepsis as a source of adrenergic surge vs myopericarditis of viral etiology. The cardiology team also suggests continued stoppage of clozapine, because the medication can cause hypotension and tachycardia.
Continue to: On Day 3...
On Day 3, Mr. C’s ST elevation resolves on ECG, and his CK level decreases to 70 U/L, at which point trending is stopped. On Day 5, Mr. C undergoes MRI, which demonstrates an ejection fraction of 55% and confirms myocarditis. No infectious source is identified.
By Day 6, with all other sources ruled out, clozapine is confirmed as the source of myocarditis for Mr. C.
The authors’ observations
Close cardiovascular monitoring should occur during the first 4 weeks after starting clozapine because 80% of cases of clozapine-induced myocarditis occur within 4 weeks of clozapine initiation.10 Baseline CRP, troponin I/T, and vital signs should be obtained before starting clozapine.11 Vital signs must be monitored to assess for fever, tachycardia, and deviations from baseline blood pressures.11 Although eosinophil counts and percentages can also be considered in addition to a baseline CRP value, they have not proven to be sensitive or specific for clozapine-induced myocarditis.12 A baseline echocardiogram can also be obtained, but is not necessary, especially given that it may not be readily available in all clinics, and could therefore delay initiation of clozapine and limit its use. C-reactive protein and troponin levels should be assessed weekly during the first 6 weeks of clozapine therapy.11 For symptomatic patients presenting with concern for clozapine-induced myocarditis, a CRP level >100 mg/L has 100% sensitivity in detecting clozapine-induced myocarditis.13 Clozapine should also be stopped if troponins levels reach twice the upper limit of normal. More mild elevations of CRP and troponins in the setting of persistent tachycardia or signs of an infectious process should be followed by daily CRP and troponins levels until these features resolve.11
Mr. C’s case highlights clinical features that clinicians should consider when screening for myocarditis. The development of myocarditis is associated with quick titrations of clozapine during Days 1 to 9. In this case, Mr. C had recently been titrated at an outside hospital, and the time frame during which this titration occurred was unknown. Given this lack of information, the potential for a rapid titration should alert the clinician to the risk of developing myocarditis. Increased age is also associated with an increased risk of myocarditis, with a 31% increase for each decade. Further, the concomitant use of valproate sodium during the titration period also increases the risk of myocarditis 2.5-fold.14
When evaluating a patient such as Mr. C, an important clinical sign that must not be overlooked is that an elevation of body temperature of 1°C is expected to give rise to a 10-beats-per-minute increase in heart rate when the fever is the result of an infection.15 During Day 1 of his hospitalization, Mr. C was tachycardic to 160 beats per minute, with a fever of 39.4°C. Thus, his heart rate was elevated well beyond what would be expected from a fever secondary to an infectious process. This further illustrates the need to consider adverse effects caused by medication, such as clozapine-induced tachycardia.
Continue to: While clozapine had already been stopped...
While clozapine had already been stopped in Mr. C, it is conceivable that other patients would potentially continue receiving it because of the medication’s demonstrated efficacy in reducing hallucinations; however, this would result in worsening and potentially serious cardiac symptoms.
[polldaddy:10226037]
The authors’ observations
A diagnosis of clozapine-induced myocarditis should be followed by a prompt discontinuation of clozapine. Discontinuation of the drug should lead to spontaneous resolution of the myocarditis, with significantly improved left ventricular function observed within 5 days.13 Historically, rechallenging a patient with clozapine was not recommended, due to fear of recurrence of myocarditis. However, recent case studies indicate that myocarditis need not be an absolute contraindication to restarting clozapine.16 Rather, the risks must be balanced against demonstrated efficacy in patients who had a limited response to other antipsychotics, as was the case with Mr. C. For these patients, the decision to rechallenge should be made with the patient’s informed consent and involve slow dose titration and increased monitoring.17 Should this rechallenge fail, another antipsychotic plus augmentation with a mood stabilizer or ECT may be more efficacious than an antipsychotic alone.18,19
OUTCOME Return to the psychiatric hospital
On Day 8, Mr. C is medically cleared; he had not reported auditory hallucinations since Day 2. He is discharged back to the psychiatric hospital for additional medication management of his schizophrenia.
Bottom Line
Clozapine-induced myocarditis should be included in the differential diagnosis for patients who present with nonspecific complaints and have an incomplete history pertaining to clozapine use. After discontinuing clozapine, and after myocarditis symptoms resolve, consider restarting clozapine in patients who have limited response to other treatments. If rechallenging fails, another antipsychotic plus augmentation with a mood stabilizer or electroconvulsive therapy may be more efficacious than an antipsychotic alone.
Related Resources
- Clozapine Risk Evaluation and Mitigation Strategy [REMS] Program. What is the Clozapine REMS Program? https://www.clozapinerems.com.
- Keating D, McWilliams S, Schneider I, et al. Pharmacological guidelines for schizophrenia: a systematic review and comparison of recommendations for the first episode. BMJ Open. 2017;7(1):e013881.
- Curto M, Girardi N, Lionetto L, et al. Systematic review of clozapine cardiotoxicity. Curr Psychiatry Rep. 2016;18(7):68.
Drug Brand Names
Clozapine • Clozaril
Olanzapine • Zyprexa
Quetiapine • Seroquel
Risperidone • Risperdal
Valproate • Depacon
1. Singer M, Deutschman CS, Seymour CW, et al. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA. 2016;315(8):801-810.
2. Thygesen K, Alpert JS, Jaffe AS, et al. Third universal definition of myocardial infarction. Eur Heart J. 2012;33(20):2551-2567.
3. Cahill TJ, Prendergast BD. Infective endocarditis. Lancet. 2016;387(10021):882-893.
4. Stein PD, Terrin ML, Hales CA, et al. Clinical, laboratory, roentgenographic, and electrocardiographic findings in patients with acute pulmonary embolism and no pre-existing cardiac or pulmonary disease. Chest. 1991;100(3):598-603.
5. Summers MR, Lennon RJ, Prasad A. Pre-morbid psychiatric and cardiovascular diseases in apical ballooning syndrome (tako-tsubo/stress-induced cardiomyopathy): potential pre-disposing factors? J Am Coll Cardiol. 2010;55(7):700-701.
6. Templin C, Ghadri JR, Diekmann J, et al. Clinical features and outcomes of Takotsubo (stress) cardiomyopathy. N Engl J Med. 2015;373(10):929-938.
7. Warnez S, Alessi-Severini S. Clozapine: a review of clinical practice guidelines and prescribing trends. BMC Psychiatry. 2014;14:102.
8. Clozaril [package insert]. Rosemont, PA: HLS Therapeutics (USA), Inc.; 2016.
9. Ronaldson KJ. Cardiovascular disease in clozapine-treated Patients: evidence, mechanisms and management. CNS Drugs. 2017;31(9):777-795.
10. Haas SJ, Hill R, Krum H, et al. Clozapine-associated myocarditis: a review of 116 cases of suspected myocarditis associated with the use of clozapine in Australia during 1993-2003. Drug Saf. 2007;30(1):47-57.
11. Goldsmith DR, Cotes RO. An unmet need: a clozapine-induced myocarditis screening protocol. Prim Care Companion CNS Disord. 2017;19(4): doi: 10.4088/PCC.16l02083.
12. Ronaldson KJ, Fitzgerald PB, McNeil JJ. Evolution of troponin, C-reactive protein and eosinophil count with the onset of clozapine-induced myocarditis. Aust N Z J Psychiatry. 2015;49(5):486-487.
13. Ronaldson KJ, Fitzgerald PB, Taylor AJ, et al. A new monitoring protocol for clozapine-induced myocarditis based on an analysis of 75 cases and 94 controls. Aust N Z J Psychiatry. 2011;45(6):458-465.
14. Ronaldson KJ, Fitzgerald PB, Taylor AJ, et al. Rapid clozapine dose titration and concomitant sodium valproate increase the risk of myocarditis with clozapine: a case-control study. Schizophr Res. 2012;141(2-3):173-178.
15. Davies P, Maconochie I. The relationship between body temperature, heart rate and respiratory rate in children. Emerg Med J. 2009;26(9):641-643.
16. Cook SC, Ferguson BA, Cotes RO, et al. Clozapine-induced myocarditis: prevention and considerations in rechallenge. Psychosomatics. 2015;56(6):685-690.
17. Ronaldson KJ, Fitzgerald PB, Taylor AJ, et al. Observations from 8 cases of clozapine rechallenge after development of myocarditis. J Clin Psychiatry. 2012;73(2):252-254.
18. Singh SP, Singh V, Kar N, et al. Efficacy of antidepressants in treating the negative symptoms of chronic schizophrenia: meta-analysis. Br J Psychiatry. 2010;197(3):174-179.
19. Wenzheng W, Chengcheng PU, Jiangling Jiang, et al. Efficacy and safety of treating patients with refractory schizophrenia with antipsychotic medication and adjunctive electroconvulsive therapy: a systematic review and meta-analysis. Shanghai Arch Psychiatry. 2015;27(4):206-219.
CASE Posing a threat to his family
Mr. C, age 23, who was diagnosed with schizophrenia with daily auditory hallucinations 4 years earlier, is transferred from an outside psychiatric hospital to our emergency department (ED) after developing fever, tachycardia, headache, and nasal congestion for the past day. He had been admitted to the psychiatric hospital 3 weeks ago due to concerns he was experiencing increased hallucinations and delusions and posed a threat to his sister and her children, with whom he had been living.
Mr. C tells us that while at the psychiatric hospital, he had been started on clozapine, 250 mg/d. He said that prior to clozapine, he had been taking risperidone. We are unable to confirm past treatment information with the psychiatric hospital, including exactly when the clozapine had been started or how fast it had been titrated. We also were not able to obtain information on his prior medication regimen.
In the ED, Mr. C is febrile (39.4°C; 102.9°F), tachycardic (160 beats per minute; reference range 60 to 100), and tachypneic (24 breaths per minute; reference range 12 to 20). His blood pressure is 130/68 mm Hg, and his lactate level is 2.3 mmol/L (reference range <1.9 mmol/L). After he receives 3 liters of fluid, Mr. C’s heart rate decreases to 117 and his lactate level to 1.1 mmol/L. His white blood cell count is 10.6 × 103/mm3 (reference range 4.0 to 10.0 × 103/mm3); a differential can be found in the Table. His electrocardiogram (ECG) demonstrates sinus tachycardia and a QTc of 510 ms (reference range <430 ms), but is otherwise unremarkable. His creatinine kinase (CK) level is within normal limits at 76 U/L (reference range 52 to 336 U/L). A C-reactive protein (CRP) level was not drawn at this time. Other than marijuana and cocaine use, Mr. C’s medical history is unremarkable.
Mr. C is admitted to the hospital and is started on treatment for sepsis. On the evening of Day 1, Mr. C experiences worsening tachycardia (140 beats per minute) and tachypnea (≥40 breaths per minute). His temperature increases to 103.3°F, and his blood pressure drops to 97/55 mm Hg. His troponin level is 19.0 ng/mL (reference range <0.01 ng/mL) and CK level is 491 U/L.
As Mr. C continues to deteriorate, a rapid response is called and he is placed on non-rebreather oxygen and transferred to the medical intensive care unit (MICU).
[polldaddy:10226034]
The authors’ observations
With Mr. C’s presenting symptoms, multiple conditions were included in the differential diagnosis. The initial concern was for sepsis. Sepsis is defined as life-threatening organ dysfunction caused by a dysregulated host response to infection.1 Organ dysfunction is defined by a quick Sepsis-Related Organ Failure Assessment (qSOFA) score ≥2 and is associated with an increased probability of mortality (>10%). Although no infection had been identified in Mr. C, the combination of fever, altered vital signs, and elevated lactate level in the setting of a qSOFA score of 2 (for respiratory rate and blood pressure) raised suspicion enough to start empiric treatment.
With Mr. C’s subsequent deterioration on the evening of Day 1, we considered cardiopulmonary etiologies. His symptoms of dyspnea, hypotension, tachycardia, tachypnea, and fever were nonspecific and thus required consideration of multiple life-threatening etiologies. Thygesen et al2 published an expert consensus of the definition of myocardial infarction, which was of concern given our patient’s elevated troponin level. Because there was already concern for sepsis, the addition of cardiac symptoms required us to consider infectious endocarditis.3 Sudden onset of dyspnea and a drop in blood pressure were concerning for pulmonary embolism, although our patient did not have the usual risk factors (cancer, immobilization, recent surgery, etc.).4 Additionally, in light of Mr. C’s psychiatric history and recent stressors of being moved from his sister’s house and admitted to a psychiatric hospital, coupled with dyspnea and hypotension, we included Takotsubo cardiomyopathy in the differential.5,6 This disease often occurs in response to an emotional or physical stressor and is characterized by transient systolic dysfunction in the setting of ventricular wall-motion abnormalities reaching beyond the distribution of a single coronary artery. Acute ECG and biomarker findings mimic those of myocardial infarction.6
Continue to: Finally, we needed to consider...
Finally, we needed to consider the potential adverse effects of clozapine. Clozapine is a second-generation antipsychotic (SGA) used to treat patients with schizophrenia for whom other antipsychotic medications are ineffective. Clozapine has been shown to be more effective than first-generation antipsychotics (FGA) in reducing symptoms of schizophrenia.7 It has also been shown to be more effective than several SGAs, including quetiapine, risperidone, and olanzapine.7 In fact, in patients with an insufficient therapeutic response to an SGA, clozapine proves to be more effective than switching to a different SGA. As a result of more than 20 years of research, clozapine is the gold-standard for treatment-resistant schizophrenia.7 Yet despite this strong evidence supporting its use in patients with treatment-resistant schizophrenia, the medication continues to be underutilized, especially in patients at risk for suicide.7
It appears that clozapine remains a third-choice medication in the treatment of schizophrenia largely due to its serious adverse effect profile.7 The medication includes several black-box warnings, including severe neutropenia, orthostatic hypotension, bradycardia, syncope, seizures, myocarditis, cardiomyopathy, and mitral valve incompetence.8 Tachycardia, bradycardia, and orthostatic hypotension are all clozapine-related adverse effects associated with autonomic dysfunction, which can result in serious long-term cardiac complications.9 With regards to the drug’s neutropenia risk, the establishment of the Clozapine Risk Evaluation and Mitigation Strategy (REMS) program has allowed for safer use of clozapine and reduced deaths due to clozapine-induced agranulocytosis. Clinicians and pharmacists must be certified in order to prescribe clozapine, and patients must be registered and undergo frequent absolute neutrophil count (ANC) monitoring.
Clozapine-induced myocarditis, a condition observed in up to 3% of patients started on the medication,9 is more likely to develop early on during treatment, with a median time of detection of 16 days following drug initiation.10 Myocarditis often presents with nonspecific signs and symptoms that include chest pain, tachycardia, palpitations, dyspnea, fever, flu-like symptoms, and/or hypotension.
[polldaddy:10226036]
The authors’ observations
Initial workup in the MICU for Mr. C included an ABG analysis, ECG, and cardiology consult. The ABG analysis demonstrated metabolic alkalosis; his ECG demonstrated sinus tachycardia and nonspecific ST elevation in the lateral leads (Figure). The cardiology consult team started Mr. C on treatment for a non-ST-elevation myocardial infarction (NSTEMI), which it believed to be most likely due to myocarditis with secondary demand ischemia, and less likely acute coronary syndrome. The cardiology consult team also recommended performing a workup for pulmonary emboli and infectious endocarditis if Mr. C’s symptoms persist or the infectious source could not be identified.

EVALUATION Gradual improvement
Mr. C demonstrates gradual improvement as his workup continues, and clozapine is held on the recommendation of the cardiac consult team. By Day 2, he stops complaining of auditory hallucinations, and does not report their return during the rest of his stay. His troponin level decreases to 8.6 ng/mL and lactate level to 1.4 mmol/L; trending is stopped for both. The erythrocyte sedimentation rate (ESR) is elevated at 59 mm/hr (reference range 0 to 22 mm/hr), along with a CRP level of 21 mg/L (reference range <8.0 mg/L). An echocardiogram demonstrates a 40% ejection fraction (reference range 55% to 75%) and moderate global hypokinesis. The cardiology consult team is concerned for Takotsubo cardiomyopathy with sepsis as a source of adrenergic surge vs myopericarditis of viral etiology. The cardiology team also suggests continued stoppage of clozapine, because the medication can cause hypotension and tachycardia.
Continue to: On Day 3...
On Day 3, Mr. C’s ST elevation resolves on ECG, and his CK level decreases to 70 U/L, at which point trending is stopped. On Day 5, Mr. C undergoes MRI, which demonstrates an ejection fraction of 55% and confirms myocarditis. No infectious source is identified.
By Day 6, with all other sources ruled out, clozapine is confirmed as the source of myocarditis for Mr. C.
The authors’ observations
Close cardiovascular monitoring should occur during the first 4 weeks after starting clozapine because 80% of cases of clozapine-induced myocarditis occur within 4 weeks of clozapine initiation.10 Baseline CRP, troponin I/T, and vital signs should be obtained before starting clozapine.11 Vital signs must be monitored to assess for fever, tachycardia, and deviations from baseline blood pressures.11 Although eosinophil counts and percentages can also be considered in addition to a baseline CRP value, they have not proven to be sensitive or specific for clozapine-induced myocarditis.12 A baseline echocardiogram can also be obtained, but is not necessary, especially given that it may not be readily available in all clinics, and could therefore delay initiation of clozapine and limit its use. C-reactive protein and troponin levels should be assessed weekly during the first 6 weeks of clozapine therapy.11 For symptomatic patients presenting with concern for clozapine-induced myocarditis, a CRP level >100 mg/L has 100% sensitivity in detecting clozapine-induced myocarditis.13 Clozapine should also be stopped if troponins levels reach twice the upper limit of normal. More mild elevations of CRP and troponins in the setting of persistent tachycardia or signs of an infectious process should be followed by daily CRP and troponins levels until these features resolve.11
Mr. C’s case highlights clinical features that clinicians should consider when screening for myocarditis. The development of myocarditis is associated with quick titrations of clozapine during Days 1 to 9. In this case, Mr. C had recently been titrated at an outside hospital, and the time frame during which this titration occurred was unknown. Given this lack of information, the potential for a rapid titration should alert the clinician to the risk of developing myocarditis. Increased age is also associated with an increased risk of myocarditis, with a 31% increase for each decade. Further, the concomitant use of valproate sodium during the titration period also increases the risk of myocarditis 2.5-fold.14
When evaluating a patient such as Mr. C, an important clinical sign that must not be overlooked is that an elevation of body temperature of 1°C is expected to give rise to a 10-beats-per-minute increase in heart rate when the fever is the result of an infection.15 During Day 1 of his hospitalization, Mr. C was tachycardic to 160 beats per minute, with a fever of 39.4°C. Thus, his heart rate was elevated well beyond what would be expected from a fever secondary to an infectious process. This further illustrates the need to consider adverse effects caused by medication, such as clozapine-induced tachycardia.
Continue to: While clozapine had already been stopped...
While clozapine had already been stopped in Mr. C, it is conceivable that other patients would potentially continue receiving it because of the medication’s demonstrated efficacy in reducing hallucinations; however, this would result in worsening and potentially serious cardiac symptoms.
[polldaddy:10226037]
The authors’ observations
A diagnosis of clozapine-induced myocarditis should be followed by a prompt discontinuation of clozapine. Discontinuation of the drug should lead to spontaneous resolution of the myocarditis, with significantly improved left ventricular function observed within 5 days.13 Historically, rechallenging a patient with clozapine was not recommended, due to fear of recurrence of myocarditis. However, recent case studies indicate that myocarditis need not be an absolute contraindication to restarting clozapine.16 Rather, the risks must be balanced against demonstrated efficacy in patients who had a limited response to other antipsychotics, as was the case with Mr. C. For these patients, the decision to rechallenge should be made with the patient’s informed consent and involve slow dose titration and increased monitoring.17 Should this rechallenge fail, another antipsychotic plus augmentation with a mood stabilizer or ECT may be more efficacious than an antipsychotic alone.18,19
OUTCOME Return to the psychiatric hospital
On Day 8, Mr. C is medically cleared; he had not reported auditory hallucinations since Day 2. He is discharged back to the psychiatric hospital for additional medication management of his schizophrenia.
Bottom Line
Clozapine-induced myocarditis should be included in the differential diagnosis for patients who present with nonspecific complaints and have an incomplete history pertaining to clozapine use. After discontinuing clozapine, and after myocarditis symptoms resolve, consider restarting clozapine in patients who have limited response to other treatments. If rechallenging fails, another antipsychotic plus augmentation with a mood stabilizer or electroconvulsive therapy may be more efficacious than an antipsychotic alone.
Related Resources
- Clozapine Risk Evaluation and Mitigation Strategy [REMS] Program. What is the Clozapine REMS Program? https://www.clozapinerems.com.
- Keating D, McWilliams S, Schneider I, et al. Pharmacological guidelines for schizophrenia: a systematic review and comparison of recommendations for the first episode. BMJ Open. 2017;7(1):e013881.
- Curto M, Girardi N, Lionetto L, et al. Systematic review of clozapine cardiotoxicity. Curr Psychiatry Rep. 2016;18(7):68.
Drug Brand Names
Clozapine • Clozaril
Olanzapine • Zyprexa
Quetiapine • Seroquel
Risperidone • Risperdal
Valproate • Depacon
CASE Posing a threat to his family
Mr. C, age 23, who was diagnosed with schizophrenia with daily auditory hallucinations 4 years earlier, is transferred from an outside psychiatric hospital to our emergency department (ED) after developing fever, tachycardia, headache, and nasal congestion for the past day. He had been admitted to the psychiatric hospital 3 weeks ago due to concerns he was experiencing increased hallucinations and delusions and posed a threat to his sister and her children, with whom he had been living.
Mr. C tells us that while at the psychiatric hospital, he had been started on clozapine, 250 mg/d. He said that prior to clozapine, he had been taking risperidone. We are unable to confirm past treatment information with the psychiatric hospital, including exactly when the clozapine had been started or how fast it had been titrated. We also were not able to obtain information on his prior medication regimen.
In the ED, Mr. C is febrile (39.4°C; 102.9°F), tachycardic (160 beats per minute; reference range 60 to 100), and tachypneic (24 breaths per minute; reference range 12 to 20). His blood pressure is 130/68 mm Hg, and his lactate level is 2.3 mmol/L (reference range <1.9 mmol/L). After he receives 3 liters of fluid, Mr. C’s heart rate decreases to 117 and his lactate level to 1.1 mmol/L. His white blood cell count is 10.6 × 103/mm3 (reference range 4.0 to 10.0 × 103/mm3); a differential can be found in the Table. His electrocardiogram (ECG) demonstrates sinus tachycardia and a QTc of 510 ms (reference range <430 ms), but is otherwise unremarkable. His creatinine kinase (CK) level is within normal limits at 76 U/L (reference range 52 to 336 U/L). A C-reactive protein (CRP) level was not drawn at this time. Other than marijuana and cocaine use, Mr. C’s medical history is unremarkable.
Mr. C is admitted to the hospital and is started on treatment for sepsis. On the evening of Day 1, Mr. C experiences worsening tachycardia (140 beats per minute) and tachypnea (≥40 breaths per minute). His temperature increases to 103.3°F, and his blood pressure drops to 97/55 mm Hg. His troponin level is 19.0 ng/mL (reference range <0.01 ng/mL) and CK level is 491 U/L.
As Mr. C continues to deteriorate, a rapid response is called and he is placed on non-rebreather oxygen and transferred to the medical intensive care unit (MICU).
[polldaddy:10226034]
The authors’ observations
With Mr. C’s presenting symptoms, multiple conditions were included in the differential diagnosis. The initial concern was for sepsis. Sepsis is defined as life-threatening organ dysfunction caused by a dysregulated host response to infection.1 Organ dysfunction is defined by a quick Sepsis-Related Organ Failure Assessment (qSOFA) score ≥2 and is associated with an increased probability of mortality (>10%). Although no infection had been identified in Mr. C, the combination of fever, altered vital signs, and elevated lactate level in the setting of a qSOFA score of 2 (for respiratory rate and blood pressure) raised suspicion enough to start empiric treatment.
With Mr. C’s subsequent deterioration on the evening of Day 1, we considered cardiopulmonary etiologies. His symptoms of dyspnea, hypotension, tachycardia, tachypnea, and fever were nonspecific and thus required consideration of multiple life-threatening etiologies. Thygesen et al2 published an expert consensus of the definition of myocardial infarction, which was of concern given our patient’s elevated troponin level. Because there was already concern for sepsis, the addition of cardiac symptoms required us to consider infectious endocarditis.3 Sudden onset of dyspnea and a drop in blood pressure were concerning for pulmonary embolism, although our patient did not have the usual risk factors (cancer, immobilization, recent surgery, etc.).4 Additionally, in light of Mr. C’s psychiatric history and recent stressors of being moved from his sister’s house and admitted to a psychiatric hospital, coupled with dyspnea and hypotension, we included Takotsubo cardiomyopathy in the differential.5,6 This disease often occurs in response to an emotional or physical stressor and is characterized by transient systolic dysfunction in the setting of ventricular wall-motion abnormalities reaching beyond the distribution of a single coronary artery. Acute ECG and biomarker findings mimic those of myocardial infarction.6
Continue to: Finally, we needed to consider...
Finally, we needed to consider the potential adverse effects of clozapine. Clozapine is a second-generation antipsychotic (SGA) used to treat patients with schizophrenia for whom other antipsychotic medications are ineffective. Clozapine has been shown to be more effective than first-generation antipsychotics (FGA) in reducing symptoms of schizophrenia.7 It has also been shown to be more effective than several SGAs, including quetiapine, risperidone, and olanzapine.7 In fact, in patients with an insufficient therapeutic response to an SGA, clozapine proves to be more effective than switching to a different SGA. As a result of more than 20 years of research, clozapine is the gold-standard for treatment-resistant schizophrenia.7 Yet despite this strong evidence supporting its use in patients with treatment-resistant schizophrenia, the medication continues to be underutilized, especially in patients at risk for suicide.7
It appears that clozapine remains a third-choice medication in the treatment of schizophrenia largely due to its serious adverse effect profile.7 The medication includes several black-box warnings, including severe neutropenia, orthostatic hypotension, bradycardia, syncope, seizures, myocarditis, cardiomyopathy, and mitral valve incompetence.8 Tachycardia, bradycardia, and orthostatic hypotension are all clozapine-related adverse effects associated with autonomic dysfunction, which can result in serious long-term cardiac complications.9 With regards to the drug’s neutropenia risk, the establishment of the Clozapine Risk Evaluation and Mitigation Strategy (REMS) program has allowed for safer use of clozapine and reduced deaths due to clozapine-induced agranulocytosis. Clinicians and pharmacists must be certified in order to prescribe clozapine, and patients must be registered and undergo frequent absolute neutrophil count (ANC) monitoring.
Clozapine-induced myocarditis, a condition observed in up to 3% of patients started on the medication,9 is more likely to develop early on during treatment, with a median time of detection of 16 days following drug initiation.10 Myocarditis often presents with nonspecific signs and symptoms that include chest pain, tachycardia, palpitations, dyspnea, fever, flu-like symptoms, and/or hypotension.
[polldaddy:10226036]
The authors’ observations
Initial workup in the MICU for Mr. C included an ABG analysis, ECG, and cardiology consult. The ABG analysis demonstrated metabolic alkalosis; his ECG demonstrated sinus tachycardia and nonspecific ST elevation in the lateral leads (Figure). The cardiology consult team started Mr. C on treatment for a non-ST-elevation myocardial infarction (NSTEMI), which it believed to be most likely due to myocarditis with secondary demand ischemia, and less likely acute coronary syndrome. The cardiology consult team also recommended performing a workup for pulmonary emboli and infectious endocarditis if Mr. C’s symptoms persist or the infectious source could not be identified.

EVALUATION Gradual improvement
Mr. C demonstrates gradual improvement as his workup continues, and clozapine is held on the recommendation of the cardiac consult team. By Day 2, he stops complaining of auditory hallucinations, and does not report their return during the rest of his stay. His troponin level decreases to 8.6 ng/mL and lactate level to 1.4 mmol/L; trending is stopped for both. The erythrocyte sedimentation rate (ESR) is elevated at 59 mm/hr (reference range 0 to 22 mm/hr), along with a CRP level of 21 mg/L (reference range <8.0 mg/L). An echocardiogram demonstrates a 40% ejection fraction (reference range 55% to 75%) and moderate global hypokinesis. The cardiology consult team is concerned for Takotsubo cardiomyopathy with sepsis as a source of adrenergic surge vs myopericarditis of viral etiology. The cardiology team also suggests continued stoppage of clozapine, because the medication can cause hypotension and tachycardia.
Continue to: On Day 3...
On Day 3, Mr. C’s ST elevation resolves on ECG, and his CK level decreases to 70 U/L, at which point trending is stopped. On Day 5, Mr. C undergoes MRI, which demonstrates an ejection fraction of 55% and confirms myocarditis. No infectious source is identified.
By Day 6, with all other sources ruled out, clozapine is confirmed as the source of myocarditis for Mr. C.
The authors’ observations
Close cardiovascular monitoring should occur during the first 4 weeks after starting clozapine because 80% of cases of clozapine-induced myocarditis occur within 4 weeks of clozapine initiation.10 Baseline CRP, troponin I/T, and vital signs should be obtained before starting clozapine.11 Vital signs must be monitored to assess for fever, tachycardia, and deviations from baseline blood pressures.11 Although eosinophil counts and percentages can also be considered in addition to a baseline CRP value, they have not proven to be sensitive or specific for clozapine-induced myocarditis.12 A baseline echocardiogram can also be obtained, but is not necessary, especially given that it may not be readily available in all clinics, and could therefore delay initiation of clozapine and limit its use. C-reactive protein and troponin levels should be assessed weekly during the first 6 weeks of clozapine therapy.11 For symptomatic patients presenting with concern for clozapine-induced myocarditis, a CRP level >100 mg/L has 100% sensitivity in detecting clozapine-induced myocarditis.13 Clozapine should also be stopped if troponins levels reach twice the upper limit of normal. More mild elevations of CRP and troponins in the setting of persistent tachycardia or signs of an infectious process should be followed by daily CRP and troponins levels until these features resolve.11
Mr. C’s case highlights clinical features that clinicians should consider when screening for myocarditis. The development of myocarditis is associated with quick titrations of clozapine during Days 1 to 9. In this case, Mr. C had recently been titrated at an outside hospital, and the time frame during which this titration occurred was unknown. Given this lack of information, the potential for a rapid titration should alert the clinician to the risk of developing myocarditis. Increased age is also associated with an increased risk of myocarditis, with a 31% increase for each decade. Further, the concomitant use of valproate sodium during the titration period also increases the risk of myocarditis 2.5-fold.14
When evaluating a patient such as Mr. C, an important clinical sign that must not be overlooked is that an elevation of body temperature of 1°C is expected to give rise to a 10-beats-per-minute increase in heart rate when the fever is the result of an infection.15 During Day 1 of his hospitalization, Mr. C was tachycardic to 160 beats per minute, with a fever of 39.4°C. Thus, his heart rate was elevated well beyond what would be expected from a fever secondary to an infectious process. This further illustrates the need to consider adverse effects caused by medication, such as clozapine-induced tachycardia.
Continue to: While clozapine had already been stopped...
While clozapine had already been stopped in Mr. C, it is conceivable that other patients would potentially continue receiving it because of the medication’s demonstrated efficacy in reducing hallucinations; however, this would result in worsening and potentially serious cardiac symptoms.
[polldaddy:10226037]
The authors’ observations
A diagnosis of clozapine-induced myocarditis should be followed by a prompt discontinuation of clozapine. Discontinuation of the drug should lead to spontaneous resolution of the myocarditis, with significantly improved left ventricular function observed within 5 days.13 Historically, rechallenging a patient with clozapine was not recommended, due to fear of recurrence of myocarditis. However, recent case studies indicate that myocarditis need not be an absolute contraindication to restarting clozapine.16 Rather, the risks must be balanced against demonstrated efficacy in patients who had a limited response to other antipsychotics, as was the case with Mr. C. For these patients, the decision to rechallenge should be made with the patient’s informed consent and involve slow dose titration and increased monitoring.17 Should this rechallenge fail, another antipsychotic plus augmentation with a mood stabilizer or ECT may be more efficacious than an antipsychotic alone.18,19
OUTCOME Return to the psychiatric hospital
On Day 8, Mr. C is medically cleared; he had not reported auditory hallucinations since Day 2. He is discharged back to the psychiatric hospital for additional medication management of his schizophrenia.
Bottom Line
Clozapine-induced myocarditis should be included in the differential diagnosis for patients who present with nonspecific complaints and have an incomplete history pertaining to clozapine use. After discontinuing clozapine, and after myocarditis symptoms resolve, consider restarting clozapine in patients who have limited response to other treatments. If rechallenging fails, another antipsychotic plus augmentation with a mood stabilizer or electroconvulsive therapy may be more efficacious than an antipsychotic alone.
Related Resources
- Clozapine Risk Evaluation and Mitigation Strategy [REMS] Program. What is the Clozapine REMS Program? https://www.clozapinerems.com.
- Keating D, McWilliams S, Schneider I, et al. Pharmacological guidelines for schizophrenia: a systematic review and comparison of recommendations for the first episode. BMJ Open. 2017;7(1):e013881.
- Curto M, Girardi N, Lionetto L, et al. Systematic review of clozapine cardiotoxicity. Curr Psychiatry Rep. 2016;18(7):68.
Drug Brand Names
Clozapine • Clozaril
Olanzapine • Zyprexa
Quetiapine • Seroquel
Risperidone • Risperdal
Valproate • Depacon
1. Singer M, Deutschman CS, Seymour CW, et al. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA. 2016;315(8):801-810.
2. Thygesen K, Alpert JS, Jaffe AS, et al. Third universal definition of myocardial infarction. Eur Heart J. 2012;33(20):2551-2567.
3. Cahill TJ, Prendergast BD. Infective endocarditis. Lancet. 2016;387(10021):882-893.
4. Stein PD, Terrin ML, Hales CA, et al. Clinical, laboratory, roentgenographic, and electrocardiographic findings in patients with acute pulmonary embolism and no pre-existing cardiac or pulmonary disease. Chest. 1991;100(3):598-603.
5. Summers MR, Lennon RJ, Prasad A. Pre-morbid psychiatric and cardiovascular diseases in apical ballooning syndrome (tako-tsubo/stress-induced cardiomyopathy): potential pre-disposing factors? J Am Coll Cardiol. 2010;55(7):700-701.
6. Templin C, Ghadri JR, Diekmann J, et al. Clinical features and outcomes of Takotsubo (stress) cardiomyopathy. N Engl J Med. 2015;373(10):929-938.
7. Warnez S, Alessi-Severini S. Clozapine: a review of clinical practice guidelines and prescribing trends. BMC Psychiatry. 2014;14:102.
8. Clozaril [package insert]. Rosemont, PA: HLS Therapeutics (USA), Inc.; 2016.
9. Ronaldson KJ. Cardiovascular disease in clozapine-treated Patients: evidence, mechanisms and management. CNS Drugs. 2017;31(9):777-795.
10. Haas SJ, Hill R, Krum H, et al. Clozapine-associated myocarditis: a review of 116 cases of suspected myocarditis associated with the use of clozapine in Australia during 1993-2003. Drug Saf. 2007;30(1):47-57.
11. Goldsmith DR, Cotes RO. An unmet need: a clozapine-induced myocarditis screening protocol. Prim Care Companion CNS Disord. 2017;19(4): doi: 10.4088/PCC.16l02083.
12. Ronaldson KJ, Fitzgerald PB, McNeil JJ. Evolution of troponin, C-reactive protein and eosinophil count with the onset of clozapine-induced myocarditis. Aust N Z J Psychiatry. 2015;49(5):486-487.
13. Ronaldson KJ, Fitzgerald PB, Taylor AJ, et al. A new monitoring protocol for clozapine-induced myocarditis based on an analysis of 75 cases and 94 controls. Aust N Z J Psychiatry. 2011;45(6):458-465.
14. Ronaldson KJ, Fitzgerald PB, Taylor AJ, et al. Rapid clozapine dose titration and concomitant sodium valproate increase the risk of myocarditis with clozapine: a case-control study. Schizophr Res. 2012;141(2-3):173-178.
15. Davies P, Maconochie I. The relationship between body temperature, heart rate and respiratory rate in children. Emerg Med J. 2009;26(9):641-643.
16. Cook SC, Ferguson BA, Cotes RO, et al. Clozapine-induced myocarditis: prevention and considerations in rechallenge. Psychosomatics. 2015;56(6):685-690.
17. Ronaldson KJ, Fitzgerald PB, Taylor AJ, et al. Observations from 8 cases of clozapine rechallenge after development of myocarditis. J Clin Psychiatry. 2012;73(2):252-254.
18. Singh SP, Singh V, Kar N, et al. Efficacy of antidepressants in treating the negative symptoms of chronic schizophrenia: meta-analysis. Br J Psychiatry. 2010;197(3):174-179.
19. Wenzheng W, Chengcheng PU, Jiangling Jiang, et al. Efficacy and safety of treating patients with refractory schizophrenia with antipsychotic medication and adjunctive electroconvulsive therapy: a systematic review and meta-analysis. Shanghai Arch Psychiatry. 2015;27(4):206-219.
1. Singer M, Deutschman CS, Seymour CW, et al. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA. 2016;315(8):801-810.
2. Thygesen K, Alpert JS, Jaffe AS, et al. Third universal definition of myocardial infarction. Eur Heart J. 2012;33(20):2551-2567.
3. Cahill TJ, Prendergast BD. Infective endocarditis. Lancet. 2016;387(10021):882-893.
4. Stein PD, Terrin ML, Hales CA, et al. Clinical, laboratory, roentgenographic, and electrocardiographic findings in patients with acute pulmonary embolism and no pre-existing cardiac or pulmonary disease. Chest. 1991;100(3):598-603.
5. Summers MR, Lennon RJ, Prasad A. Pre-morbid psychiatric and cardiovascular diseases in apical ballooning syndrome (tako-tsubo/stress-induced cardiomyopathy): potential pre-disposing factors? J Am Coll Cardiol. 2010;55(7):700-701.
6. Templin C, Ghadri JR, Diekmann J, et al. Clinical features and outcomes of Takotsubo (stress) cardiomyopathy. N Engl J Med. 2015;373(10):929-938.
7. Warnez S, Alessi-Severini S. Clozapine: a review of clinical practice guidelines and prescribing trends. BMC Psychiatry. 2014;14:102.
8. Clozaril [package insert]. Rosemont, PA: HLS Therapeutics (USA), Inc.; 2016.
9. Ronaldson KJ. Cardiovascular disease in clozapine-treated Patients: evidence, mechanisms and management. CNS Drugs. 2017;31(9):777-795.
10. Haas SJ, Hill R, Krum H, et al. Clozapine-associated myocarditis: a review of 116 cases of suspected myocarditis associated with the use of clozapine in Australia during 1993-2003. Drug Saf. 2007;30(1):47-57.
11. Goldsmith DR, Cotes RO. An unmet need: a clozapine-induced myocarditis screening protocol. Prim Care Companion CNS Disord. 2017;19(4): doi: 10.4088/PCC.16l02083.
12. Ronaldson KJ, Fitzgerald PB, McNeil JJ. Evolution of troponin, C-reactive protein and eosinophil count with the onset of clozapine-induced myocarditis. Aust N Z J Psychiatry. 2015;49(5):486-487.
13. Ronaldson KJ, Fitzgerald PB, Taylor AJ, et al. A new monitoring protocol for clozapine-induced myocarditis based on an analysis of 75 cases and 94 controls. Aust N Z J Psychiatry. 2011;45(6):458-465.
14. Ronaldson KJ, Fitzgerald PB, Taylor AJ, et al. Rapid clozapine dose titration and concomitant sodium valproate increase the risk of myocarditis with clozapine: a case-control study. Schizophr Res. 2012;141(2-3):173-178.
15. Davies P, Maconochie I. The relationship between body temperature, heart rate and respiratory rate in children. Emerg Med J. 2009;26(9):641-643.
16. Cook SC, Ferguson BA, Cotes RO, et al. Clozapine-induced myocarditis: prevention and considerations in rechallenge. Psychosomatics. 2015;56(6):685-690.
17. Ronaldson KJ, Fitzgerald PB, Taylor AJ, et al. Observations from 8 cases of clozapine rechallenge after development of myocarditis. J Clin Psychiatry. 2012;73(2):252-254.
18. Singh SP, Singh V, Kar N, et al. Efficacy of antidepressants in treating the negative symptoms of chronic schizophrenia: meta-analysis. Br J Psychiatry. 2010;197(3):174-179.
19. Wenzheng W, Chengcheng PU, Jiangling Jiang, et al. Efficacy and safety of treating patients with refractory schizophrenia with antipsychotic medication and adjunctive electroconvulsive therapy: a systematic review and meta-analysis. Shanghai Arch Psychiatry. 2015;27(4):206-219.
Differentiating serotonin syndrome and neuroleptic malignant syndrome
Serotonin syndrome (SS) and neuroleptic malignant syndrome (NMS) are each rare psychiatric emergencies that can lead to fatal outcomes. Their clinical presentations can overlap, which can make it difficult to differentiate between the 2 syndromes; however, their treatments are distinct, and it is imperative to know how to identify symptoms and accurately diagnose each of them to provide appropriate intervention. This article summarizes the 2 syndromes and their treatments, with a focus on how clinicians can distinguish them, provide prompt intervention, and prevent occurrence.
Serotonin syndrome
Mechanism. The decarboxylation and hydroxylation of tryptophan forms serotonin, also known as 5-hydroxytryptamine (5-HT), which can then be metabolized by monoamine oxidase-A (MAO-A) into 5-hydroxyindoleacetic acid (5-HIAA).1Medications can disrupt this pathway of serotonin production or its metabolism, and result in excessive levels of serotonin, which subsequently leads to an overactivation of central and peripheral serotonin receptors.1 Increased receptor activation leads to further upregulation, and ultimately more serotonin transmission. This can be caused by monotherapy or use of multiple serotonergic agents, polypharmacy with a combination of medication classes, drug interactions, or overdose. The wide variety of medications often prescribed by different clinicians can make identification of excessive serotonergic activity difficult, especially because mood stabilizers such as lithium,2 and non-psychiatric medications such as
- inhibition of serotonin uptake (seen with selective serotonin reuptake inhibitors [SSRIs], serotonin-norepinephrine reuptake inhibitors [SNRIs], and tricyclic antidepressants [TCAs])
- inhibition of serotonin metabolism (seen with monoamine oxidase inhibitors [MAOIs])
- increased serotonin synthesis (seen with stimulants)
- increased serotonin release (seen with stimulants and opiates)
- activation of serotonin receptors (seen with lithium)
- inhibition of certain cytochrome P450 (CYP450) enzymes (seen with ciprofloxacin, fluconazole, etc.).

It is important to recognize that various serotonergic agents are involved in the CYP450 system. Inhibition of the CYP450 pathway by common antibiotics such as ciprofloxacin, or antifungals such as fluconazole, may result in an accumulation of serotonergic agents and place patients at increased risk for developing SS.
Clinical presentation. The clinical presentation of SS can range from mild to fatal. There is no specific laboratory test for diagnosis, although an elevation of the total creatine kinase (CK) and leukocyte count, as well as increased transaminase levels or lower bicarbonate levels, have been reported in the literature.4
Symptoms of SS generally present within 24 hours of starting/changing therapy and include a triad of mental status changes (altered mental status [AMS]), autonomic instability, and abnormalities of neuromuscular tone. Examples of AMS include agitation, anxiety, disorientation, and restlessness. Symptoms of autonomic instability include hypertension, tachycardia, tachypnea, hyperthermia, diaphoresis, flushed skin, vomiting, diarrhea, and arrhythmias. Symptoms stemming from changes in neuromuscular tone include tremors, clonus, hyperreflexia, and muscle rigidity.1 The multiple possible clinical presentations, as well as symptoms that overlap with those of other syndromes, can make SS difficult to recognize quickly in a clinical setting.
Diagnostic criteria. Sternbach’s diagnostic criteria for SS are defined as the presence of 3 or more of the 10 most common clinical features (Table 25). Due to concerns that Sternbach’s diagnostic criteria overemphasized an abnormal mental state (leading to possible confusion of SS with other AMS syndromes), the Hunter serotonin toxicity criteria6 (Figure6) were developed in 2003, and were found to be more sensitive and specific than Sternbach’s criteria. Both tools are often used in clinical practice.

Treatment. Treatment of SS begins with prompt discontinuation of all serotonergic agents. The intensity of treatment depends on the severity of the symptoms. Mild symptoms can be managed with supportive care,3 and in such cases, the syndrome generally resolves within 24 hours.7 Clinicians may use supportive care to normalize vital signs (oxygenation to maintain SpO2 >94%, IV fluids for volume depletion, cooling agents, antihypertensives, benzodiazepines for sedation or control of agitation, etc.). Patients who are more ill may require more aggressive treatment, such as the use of a serotonergic antagonist (ie, cyproheptadine) and those who are severely hyperthermic (temperature >41.1ºC) may require neuromuscular sedation, paralysis, and possibly endotracheal intubation.3
Continue to: Management pitfalls include...
Management pitfalls include misdiagnosis of SS, failure to recognize its rapid rate of progression, and adverse effects of pharmacologic therapy.3 The most effective treatment for SS is prevention. SS can be prevented by astute pharmacologic understanding, avoidance of polypharmacy, and physician education.3
Neuroleptic malignant syndrome
Possible mechanisms. Neuromuscular malignant syndrome is thought to result from dopamine receptor antagonism leading to a hypodopaminergic state in the striatum and hypothalamus.8 The pathophysiology behind NMS has not fully been elucidated; however, several hypotheses attempt to explain this life-threatening reaction. The first focuses on dopamine D2 receptor antagonism, because many of the neuroleptic (antipsychotic) medications that can precipitate NMS are involved in dopamine blockade. In this theory, blocking dopamine D2 receptors in the anterior hypothalamus explains the hyperthermia seen in NMS, while blockade in the corpus striatum is believed to lead to muscle rigidity.9
The second hypothesis suggests that neuroleptics may have a direct toxic effect to muscle cells. Neuroleptics influence calcium transport across the sarcoplasmic reticulum and can lead to increased calcium release, which may contribute to the muscle rigidity and hyperthermia seen in NMS.9
The third hypothesis involves hyperactivity of the sympathetic nervous system; it is thought that psychologic stressors alter frontal lobe function, with neuroleptics disrupting the inhibitory pathways of the sympathetic nervous system. The autonomic nervous system innervates multiple organ systems, so this excessively dysregulated sympathetic nervous system may be responsible for multiple NMS symptoms (hyperthermia, muscle rigidity, hypertension, diaphoresis, tachycardia, elevated CK.10
NMS can be caused by neuroleptic agents (both first- and second-generation antipsychotics) as well as antiemetics (Table 31). The time between use of these medications and onset of symptoms is highly variable. NMS can occur after a single dose, after a dose adjustment, or possibly after years of treatment with the same medication. It is not dose-dependent.11 In certain individuals, NMS may occur at therapeutic doses.

Continue to: Clinical presentation
Clinical presentation. Patients with NMS typically present with a tetrad of symptoms: mental status changes, muscular rigidity, hyperthermia, and autonomic instability.12 Mental status changes can include confusion and agitation, as well as catatonic signs and mutism. The muscular rigidity of NMS is characterized by “lead pipe rigidity” and may be accompanied by tremor, dystonia, or dyskinesias. Laboratory findings include elevated serum CK (from severe rigidity), often >1,000 U/L, although normal levels can be observed if rigidity has not yet developed.13
Treatment. The first step for treatment is to discontinue the causative medication.14 Initiate supportive therapy immediately to restrict the progression of symptoms. Interventions include cooling blankets, fluid resuscitation, and antihypertensives to maintain autonomic stability15 or benzodiazepines to control agitation. In severe cases, muscular rigidity may extend to the airways and intubation may be required. The severity of these symptoms may warrant admission to the ICU for close monitoring. Pharmacologic treatment with
Differentiating between SS and NMS
Differentiating between these 2 syndromes (Table 417) is critical to direct appropriate intervention. Table 517 outlines the treatment overview for SS and NMS.

Detailed history. A detailed history is imperative in making accurate diagnoses. Useful components of the history include a patient’s duration of symptoms and medication history (prescription medications as well as over-the-counter medications, supplements, and illicit drugs). Also assess for medical comorbidities, because certain medical diagnoses may alert the clinician that it is likely the patient had been prescribed serotonergic agents or neuroleptics, and renal or liver impairment may alert the clinician of decreased metabolism rates. Medication history is arguably the most useful piece of the interview, because serotonergic agents can cause SS, whereas dopamine blockers cause NMS. It should be noted that excess serotonin acts as a true toxidrome and is concentration-dependent in causing SS, whereas NMS is an idiosyncratic reaction to a drug.

Physical exam. Although there are many overlapping clinical manifestations, SS produces neuromuscular hyperactivity (ie, clonus, hyperreflexia), whereas NMS is characterized by more sluggish responses (ie, rigidity, bradyreflexia).18
Continue to: Laboratory findings
Laboratory findings. Overlap between NMS and SS also occurs with lab findings; both syndromes can result in leukocytosis, elevated CK from muscle damage, and low serum iron levels. However, these findings are more commonly associated with NMS and are seen in 75% of cases.17,19
Course of illness. Duration of symptoms can also help differentiate the 2 syndromes. SS typically develops within 24 hours of starting/changing therapy, whereas NMS symptoms can be present for days to weeks. Resolution of symptoms may also be helpful in differentiation because SS typically resolves within a few days of initiating treatment, whereas NMS resolves within 9 to 14 days of starting treatment.19
Bottom Line
The clinical presentations of serotonin syndrome (SS) and neuroleptic malignant syndrome (NMS) overlap, which can make them difficult to differentiate; however, they each have distinct approaches to treatment. Features in SS that are distinct from NMS include a history of serotonergic agents, rapid onset of symptoms, hyperreflexia, and clonus. NMS is slower in onset and can be found in patients who are prescribed dopamine antagonists, with distinct symptoms of rigidity and hyporeflexia.
Related Resources
- Kimmel R. Serotonin syndrome or NMS? Clues to diagnosis. Current Psychiatry. 2010;9(2):92.
- Strawn JR, Keck Jr PE, Caroff SN. Neuroleptic malignant syndrome: Answers to 6 tough questions. Current Psychiatry. 2008;7(1):95-101.
Drug Brand Names
Amantadine • Symmetrel
Amitriptyline • Elavil, Endep
Aripiprazole • Abilify
Bromocriptine • Cycloset, Parlodel
Bupropion • Wellbutrin, Zyban
Buspirone • BuSpar
Carbamazepine • Carbatrol, Tegretol
Chlorpromazine • Thorazine
Ciprofloxacin • Cipro
Citalopram • Celexa
Clomipramine • Anafranil
Clozapine • Clozaril
Cyclobenzaprine • Amrix, Flexeril
Cyproheptadine • Periactin
Dantrolene • Dantrium
Desipramine • Norpramin
Desvenlafaxine • Pristiq
Dextromethorphan • Benylin, Dexalone
Dolasetron • Anzemet
Doxepin • Silenor
Droperidol • Inapsine
Duloxetine • Cymbalt
Escitalopram • Lexapro
Fentanyl • Actiq, Duragesic
Fluconazole • Diflucan
Fluoxetine • Prozac
Fluphenazine • Prolixin
Fluvoxamine • Luvox
Granisetron • Kytril
Haloperidol • Haldol
Isocarboxazid • Marplan
Levomilnacipran • Fetzima
Linezolid • Zyvox
Lithium • Eskalith, Lithobid
Meperidone • Demerol
Metoclopramide • Reglan
Milnacipran • Savella
Nefazodone • Serzone
Olanzapine • Zyprexa
Ondansetron • Zofran
Paliperidone • Invega
Palonosetron • Aloxi
Paroxetine • Paxil
Pentazocine • Talwin, Talacen
Perphenazine • Trilafon
Phenelzine • Nardil
Procarbazine • Matulane
Prochlorperazine • Compazine
Promethazine • Phenergan
Quetiapine • Seroquel
Rasagiline • Azilect
Risperidone • Risperdal
Safinamide • Xadago
Selegiline • Eldepryl, Zelapar
Sertraline • Zoloft
Sibutramine • Meridia
Tedizolid • Sivextro
Thioridazine • Mellaril
Tranylcypromine • Parnate
Tramadol • Ultram
Trazodone • Desyrel, Oleptro
Venlafaxine • Effexor
Vilazodone • Viibryd
Vortioxetine • Trintellix
Valproate • Depacon
Ziprasidone • Geodon
1. Volpi-Abadie J, Kaye AM, Kaye AD. Serotonin syndrome. Ochsner J. 2013;13(4):533-540.
2. Werneke U, Jamshidi F, Taylor D, et al. Conundrums in neurology: diagnosing serotonin syndrome – a meta-analysis of cases. BMC Neurol. 2016;16:97.
3. Boyer EW, Shannon M. The serotonin syndrome. N Engl J Med. 2005;352(11):1112-1120.
4. Birmes P, Coppin D, Schmitt L, et al. Serotonin syndrome: a brief review. CMAJ. 2003;168(11):1439-1442.
5. Sternbach H. The serotonin syndrome. Am J Psychiatry. 1991;148:705-713.
6. Dunkley EJ, Isbister GK, Sibbritt D, et al. The Hunter serotonin toxicity criteria: simple and accurate diagnostic decision rules for serotonin toxicity. QJM. 2003; 96(9):635-642.
7. Lappin RI, Auchincloss EL. Treatment of the serotonin syndrome with cyproheptadine. N Engl J Med. 1994;331(15):1021-1022.
8. Nisijima K. Serotonin syndrome overlapping with neuroleptic malignant syndrome: A case report and approaches for differentially diagnosing the two syndromes. Asian J Psychiatr. 2015;18:100-101.
9. Adnet P, Lestavel P, Krivosic-Horber R. Neuroleptic malignant syndrome. Br J Anaesth. 2000;85(1):129-135.
10. Gurrera R. Sympathoadrenal hyperactivity and the etiology of neuroleptic malignant syndrome. Am J Psychiatry. 1999;156:169-180.
11. Pope HG Jr, Aizley HG, Keck PE Jr, et al. Neuroleptic malignant syndrome: long-term follow-up of 20 cases. J Clin Psychiatry. 1991;52(5):208-212.
12. Velamoor VR, Norman RM, Caroff SN, et al. Progression of symptoms in neuroleptic malignant syndrome. J Nerv Ment Dis. 1994;182(3):168-173.
13. Caroff SN, Mann SC. Neuroleptic malignant syndrome. Med Clin North Am. 1993;77(1):185-202.
14. Pileggi DJ, Cook AM. Neuroleptic malignant syndrome. Ann Pharmacother. 2016;50(11):973-981.
15. San Gabriel MC, Eddula-Changala B, Tan Y, et al. Electroconvulsive in a schizophrenic patient with neuroleptic malignant syndrome and rhabdomyolysis. J ECT. 2015;31(3):197-200.
16. Buggenhout S, Vandenberghe J, Sienaert P. Electroconvulsion therapy for neuroleptic malignant syndrome. Tijdschr Psychiatr. 2014;56(9):612-615.
17. Perry PJ, Wilborn CA. Serotonin syndrome vs neuroleptic malignant syndrome: a contrast of causes, diagnoses, and management. Ann Clin Psychiatry. 2012;24(2):155-162.
18. Mills KC. Serotonin syndrome. A clinical update. Crit Care Clin. 1997;13(4):763-783.
19. Dosi R, Ambaliya A, Joshi H, et al. Serotonin syndrome versus neuroleptic malignant syndrome: a challenge clinical quandary. BMJ Case Rep. 2014;2014:bcr201404154. doi:10.1136/bcr-2014-204154.
Serotonin syndrome (SS) and neuroleptic malignant syndrome (NMS) are each rare psychiatric emergencies that can lead to fatal outcomes. Their clinical presentations can overlap, which can make it difficult to differentiate between the 2 syndromes; however, their treatments are distinct, and it is imperative to know how to identify symptoms and accurately diagnose each of them to provide appropriate intervention. This article summarizes the 2 syndromes and their treatments, with a focus on how clinicians can distinguish them, provide prompt intervention, and prevent occurrence.
Serotonin syndrome
Mechanism. The decarboxylation and hydroxylation of tryptophan forms serotonin, also known as 5-hydroxytryptamine (5-HT), which can then be metabolized by monoamine oxidase-A (MAO-A) into 5-hydroxyindoleacetic acid (5-HIAA).1Medications can disrupt this pathway of serotonin production or its metabolism, and result in excessive levels of serotonin, which subsequently leads to an overactivation of central and peripheral serotonin receptors.1 Increased receptor activation leads to further upregulation, and ultimately more serotonin transmission. This can be caused by monotherapy or use of multiple serotonergic agents, polypharmacy with a combination of medication classes, drug interactions, or overdose. The wide variety of medications often prescribed by different clinicians can make identification of excessive serotonergic activity difficult, especially because mood stabilizers such as lithium,2 and non-psychiatric medications such as
- inhibition of serotonin uptake (seen with selective serotonin reuptake inhibitors [SSRIs], serotonin-norepinephrine reuptake inhibitors [SNRIs], and tricyclic antidepressants [TCAs])
- inhibition of serotonin metabolism (seen with monoamine oxidase inhibitors [MAOIs])
- increased serotonin synthesis (seen with stimulants)
- increased serotonin release (seen with stimulants and opiates)
- activation of serotonin receptors (seen with lithium)
- inhibition of certain cytochrome P450 (CYP450) enzymes (seen with ciprofloxacin, fluconazole, etc.).

It is important to recognize that various serotonergic agents are involved in the CYP450 system. Inhibition of the CYP450 pathway by common antibiotics such as ciprofloxacin, or antifungals such as fluconazole, may result in an accumulation of serotonergic agents and place patients at increased risk for developing SS.
Clinical presentation. The clinical presentation of SS can range from mild to fatal. There is no specific laboratory test for diagnosis, although an elevation of the total creatine kinase (CK) and leukocyte count, as well as increased transaminase levels or lower bicarbonate levels, have been reported in the literature.4
Symptoms of SS generally present within 24 hours of starting/changing therapy and include a triad of mental status changes (altered mental status [AMS]), autonomic instability, and abnormalities of neuromuscular tone. Examples of AMS include agitation, anxiety, disorientation, and restlessness. Symptoms of autonomic instability include hypertension, tachycardia, tachypnea, hyperthermia, diaphoresis, flushed skin, vomiting, diarrhea, and arrhythmias. Symptoms stemming from changes in neuromuscular tone include tremors, clonus, hyperreflexia, and muscle rigidity.1 The multiple possible clinical presentations, as well as symptoms that overlap with those of other syndromes, can make SS difficult to recognize quickly in a clinical setting.
Diagnostic criteria. Sternbach’s diagnostic criteria for SS are defined as the presence of 3 or more of the 10 most common clinical features (Table 25). Due to concerns that Sternbach’s diagnostic criteria overemphasized an abnormal mental state (leading to possible confusion of SS with other AMS syndromes), the Hunter serotonin toxicity criteria6 (Figure6) were developed in 2003, and were found to be more sensitive and specific than Sternbach’s criteria. Both tools are often used in clinical practice.

Treatment. Treatment of SS begins with prompt discontinuation of all serotonergic agents. The intensity of treatment depends on the severity of the symptoms. Mild symptoms can be managed with supportive care,3 and in such cases, the syndrome generally resolves within 24 hours.7 Clinicians may use supportive care to normalize vital signs (oxygenation to maintain SpO2 >94%, IV fluids for volume depletion, cooling agents, antihypertensives, benzodiazepines for sedation or control of agitation, etc.). Patients who are more ill may require more aggressive treatment, such as the use of a serotonergic antagonist (ie, cyproheptadine) and those who are severely hyperthermic (temperature >41.1ºC) may require neuromuscular sedation, paralysis, and possibly endotracheal intubation.3
Continue to: Management pitfalls include...
Management pitfalls include misdiagnosis of SS, failure to recognize its rapid rate of progression, and adverse effects of pharmacologic therapy.3 The most effective treatment for SS is prevention. SS can be prevented by astute pharmacologic understanding, avoidance of polypharmacy, and physician education.3
Neuroleptic malignant syndrome
Possible mechanisms. Neuromuscular malignant syndrome is thought to result from dopamine receptor antagonism leading to a hypodopaminergic state in the striatum and hypothalamus.8 The pathophysiology behind NMS has not fully been elucidated; however, several hypotheses attempt to explain this life-threatening reaction. The first focuses on dopamine D2 receptor antagonism, because many of the neuroleptic (antipsychotic) medications that can precipitate NMS are involved in dopamine blockade. In this theory, blocking dopamine D2 receptors in the anterior hypothalamus explains the hyperthermia seen in NMS, while blockade in the corpus striatum is believed to lead to muscle rigidity.9
The second hypothesis suggests that neuroleptics may have a direct toxic effect to muscle cells. Neuroleptics influence calcium transport across the sarcoplasmic reticulum and can lead to increased calcium release, which may contribute to the muscle rigidity and hyperthermia seen in NMS.9
The third hypothesis involves hyperactivity of the sympathetic nervous system; it is thought that psychologic stressors alter frontal lobe function, with neuroleptics disrupting the inhibitory pathways of the sympathetic nervous system. The autonomic nervous system innervates multiple organ systems, so this excessively dysregulated sympathetic nervous system may be responsible for multiple NMS symptoms (hyperthermia, muscle rigidity, hypertension, diaphoresis, tachycardia, elevated CK.10
NMS can be caused by neuroleptic agents (both first- and second-generation antipsychotics) as well as antiemetics (Table 31). The time between use of these medications and onset of symptoms is highly variable. NMS can occur after a single dose, after a dose adjustment, or possibly after years of treatment with the same medication. It is not dose-dependent.11 In certain individuals, NMS may occur at therapeutic doses.

Continue to: Clinical presentation
Clinical presentation. Patients with NMS typically present with a tetrad of symptoms: mental status changes, muscular rigidity, hyperthermia, and autonomic instability.12 Mental status changes can include confusion and agitation, as well as catatonic signs and mutism. The muscular rigidity of NMS is characterized by “lead pipe rigidity” and may be accompanied by tremor, dystonia, or dyskinesias. Laboratory findings include elevated serum CK (from severe rigidity), often >1,000 U/L, although normal levels can be observed if rigidity has not yet developed.13
Treatment. The first step for treatment is to discontinue the causative medication.14 Initiate supportive therapy immediately to restrict the progression of symptoms. Interventions include cooling blankets, fluid resuscitation, and antihypertensives to maintain autonomic stability15 or benzodiazepines to control agitation. In severe cases, muscular rigidity may extend to the airways and intubation may be required. The severity of these symptoms may warrant admission to the ICU for close monitoring. Pharmacologic treatment with
Differentiating between SS and NMS
Differentiating between these 2 syndromes (Table 417) is critical to direct appropriate intervention. Table 517 outlines the treatment overview for SS and NMS.

Detailed history. A detailed history is imperative in making accurate diagnoses. Useful components of the history include a patient’s duration of symptoms and medication history (prescription medications as well as over-the-counter medications, supplements, and illicit drugs). Also assess for medical comorbidities, because certain medical diagnoses may alert the clinician that it is likely the patient had been prescribed serotonergic agents or neuroleptics, and renal or liver impairment may alert the clinician of decreased metabolism rates. Medication history is arguably the most useful piece of the interview, because serotonergic agents can cause SS, whereas dopamine blockers cause NMS. It should be noted that excess serotonin acts as a true toxidrome and is concentration-dependent in causing SS, whereas NMS is an idiosyncratic reaction to a drug.

Physical exam. Although there are many overlapping clinical manifestations, SS produces neuromuscular hyperactivity (ie, clonus, hyperreflexia), whereas NMS is characterized by more sluggish responses (ie, rigidity, bradyreflexia).18
Continue to: Laboratory findings
Laboratory findings. Overlap between NMS and SS also occurs with lab findings; both syndromes can result in leukocytosis, elevated CK from muscle damage, and low serum iron levels. However, these findings are more commonly associated with NMS and are seen in 75% of cases.17,19
Course of illness. Duration of symptoms can also help differentiate the 2 syndromes. SS typically develops within 24 hours of starting/changing therapy, whereas NMS symptoms can be present for days to weeks. Resolution of symptoms may also be helpful in differentiation because SS typically resolves within a few days of initiating treatment, whereas NMS resolves within 9 to 14 days of starting treatment.19
Bottom Line
The clinical presentations of serotonin syndrome (SS) and neuroleptic malignant syndrome (NMS) overlap, which can make them difficult to differentiate; however, they each have distinct approaches to treatment. Features in SS that are distinct from NMS include a history of serotonergic agents, rapid onset of symptoms, hyperreflexia, and clonus. NMS is slower in onset and can be found in patients who are prescribed dopamine antagonists, with distinct symptoms of rigidity and hyporeflexia.
Related Resources
- Kimmel R. Serotonin syndrome or NMS? Clues to diagnosis. Current Psychiatry. 2010;9(2):92.
- Strawn JR, Keck Jr PE, Caroff SN. Neuroleptic malignant syndrome: Answers to 6 tough questions. Current Psychiatry. 2008;7(1):95-101.
Drug Brand Names
Amantadine • Symmetrel
Amitriptyline • Elavil, Endep
Aripiprazole • Abilify
Bromocriptine • Cycloset, Parlodel
Bupropion • Wellbutrin, Zyban
Buspirone • BuSpar
Carbamazepine • Carbatrol, Tegretol
Chlorpromazine • Thorazine
Ciprofloxacin • Cipro
Citalopram • Celexa
Clomipramine • Anafranil
Clozapine • Clozaril
Cyclobenzaprine • Amrix, Flexeril
Cyproheptadine • Periactin
Dantrolene • Dantrium
Desipramine • Norpramin
Desvenlafaxine • Pristiq
Dextromethorphan • Benylin, Dexalone
Dolasetron • Anzemet
Doxepin • Silenor
Droperidol • Inapsine
Duloxetine • Cymbalt
Escitalopram • Lexapro
Fentanyl • Actiq, Duragesic
Fluconazole • Diflucan
Fluoxetine • Prozac
Fluphenazine • Prolixin
Fluvoxamine • Luvox
Granisetron • Kytril
Haloperidol • Haldol
Isocarboxazid • Marplan
Levomilnacipran • Fetzima
Linezolid • Zyvox
Lithium • Eskalith, Lithobid
Meperidone • Demerol
Metoclopramide • Reglan
Milnacipran • Savella
Nefazodone • Serzone
Olanzapine • Zyprexa
Ondansetron • Zofran
Paliperidone • Invega
Palonosetron • Aloxi
Paroxetine • Paxil
Pentazocine • Talwin, Talacen
Perphenazine • Trilafon
Phenelzine • Nardil
Procarbazine • Matulane
Prochlorperazine • Compazine
Promethazine • Phenergan
Quetiapine • Seroquel
Rasagiline • Azilect
Risperidone • Risperdal
Safinamide • Xadago
Selegiline • Eldepryl, Zelapar
Sertraline • Zoloft
Sibutramine • Meridia
Tedizolid • Sivextro
Thioridazine • Mellaril
Tranylcypromine • Parnate
Tramadol • Ultram
Trazodone • Desyrel, Oleptro
Venlafaxine • Effexor
Vilazodone • Viibryd
Vortioxetine • Trintellix
Valproate • Depacon
Ziprasidone • Geodon
Serotonin syndrome (SS) and neuroleptic malignant syndrome (NMS) are each rare psychiatric emergencies that can lead to fatal outcomes. Their clinical presentations can overlap, which can make it difficult to differentiate between the 2 syndromes; however, their treatments are distinct, and it is imperative to know how to identify symptoms and accurately diagnose each of them to provide appropriate intervention. This article summarizes the 2 syndromes and their treatments, with a focus on how clinicians can distinguish them, provide prompt intervention, and prevent occurrence.
Serotonin syndrome
Mechanism. The decarboxylation and hydroxylation of tryptophan forms serotonin, also known as 5-hydroxytryptamine (5-HT), which can then be metabolized by monoamine oxidase-A (MAO-A) into 5-hydroxyindoleacetic acid (5-HIAA).1Medications can disrupt this pathway of serotonin production or its metabolism, and result in excessive levels of serotonin, which subsequently leads to an overactivation of central and peripheral serotonin receptors.1 Increased receptor activation leads to further upregulation, and ultimately more serotonin transmission. This can be caused by monotherapy or use of multiple serotonergic agents, polypharmacy with a combination of medication classes, drug interactions, or overdose. The wide variety of medications often prescribed by different clinicians can make identification of excessive serotonergic activity difficult, especially because mood stabilizers such as lithium,2 and non-psychiatric medications such as
- inhibition of serotonin uptake (seen with selective serotonin reuptake inhibitors [SSRIs], serotonin-norepinephrine reuptake inhibitors [SNRIs], and tricyclic antidepressants [TCAs])
- inhibition of serotonin metabolism (seen with monoamine oxidase inhibitors [MAOIs])
- increased serotonin synthesis (seen with stimulants)
- increased serotonin release (seen with stimulants and opiates)
- activation of serotonin receptors (seen with lithium)
- inhibition of certain cytochrome P450 (CYP450) enzymes (seen with ciprofloxacin, fluconazole, etc.).

It is important to recognize that various serotonergic agents are involved in the CYP450 system. Inhibition of the CYP450 pathway by common antibiotics such as ciprofloxacin, or antifungals such as fluconazole, may result in an accumulation of serotonergic agents and place patients at increased risk for developing SS.
Clinical presentation. The clinical presentation of SS can range from mild to fatal. There is no specific laboratory test for diagnosis, although an elevation of the total creatine kinase (CK) and leukocyte count, as well as increased transaminase levels or lower bicarbonate levels, have been reported in the literature.4
Symptoms of SS generally present within 24 hours of starting/changing therapy and include a triad of mental status changes (altered mental status [AMS]), autonomic instability, and abnormalities of neuromuscular tone. Examples of AMS include agitation, anxiety, disorientation, and restlessness. Symptoms of autonomic instability include hypertension, tachycardia, tachypnea, hyperthermia, diaphoresis, flushed skin, vomiting, diarrhea, and arrhythmias. Symptoms stemming from changes in neuromuscular tone include tremors, clonus, hyperreflexia, and muscle rigidity.1 The multiple possible clinical presentations, as well as symptoms that overlap with those of other syndromes, can make SS difficult to recognize quickly in a clinical setting.
Diagnostic criteria. Sternbach’s diagnostic criteria for SS are defined as the presence of 3 or more of the 10 most common clinical features (Table 25). Due to concerns that Sternbach’s diagnostic criteria overemphasized an abnormal mental state (leading to possible confusion of SS with other AMS syndromes), the Hunter serotonin toxicity criteria6 (Figure6) were developed in 2003, and were found to be more sensitive and specific than Sternbach’s criteria. Both tools are often used in clinical practice.

Treatment. Treatment of SS begins with prompt discontinuation of all serotonergic agents. The intensity of treatment depends on the severity of the symptoms. Mild symptoms can be managed with supportive care,3 and in such cases, the syndrome generally resolves within 24 hours.7 Clinicians may use supportive care to normalize vital signs (oxygenation to maintain SpO2 >94%, IV fluids for volume depletion, cooling agents, antihypertensives, benzodiazepines for sedation or control of agitation, etc.). Patients who are more ill may require more aggressive treatment, such as the use of a serotonergic antagonist (ie, cyproheptadine) and those who are severely hyperthermic (temperature >41.1ºC) may require neuromuscular sedation, paralysis, and possibly endotracheal intubation.3
Continue to: Management pitfalls include...
Management pitfalls include misdiagnosis of SS, failure to recognize its rapid rate of progression, and adverse effects of pharmacologic therapy.3 The most effective treatment for SS is prevention. SS can be prevented by astute pharmacologic understanding, avoidance of polypharmacy, and physician education.3
Neuroleptic malignant syndrome
Possible mechanisms. Neuromuscular malignant syndrome is thought to result from dopamine receptor antagonism leading to a hypodopaminergic state in the striatum and hypothalamus.8 The pathophysiology behind NMS has not fully been elucidated; however, several hypotheses attempt to explain this life-threatening reaction. The first focuses on dopamine D2 receptor antagonism, because many of the neuroleptic (antipsychotic) medications that can precipitate NMS are involved in dopamine blockade. In this theory, blocking dopamine D2 receptors in the anterior hypothalamus explains the hyperthermia seen in NMS, while blockade in the corpus striatum is believed to lead to muscle rigidity.9
The second hypothesis suggests that neuroleptics may have a direct toxic effect to muscle cells. Neuroleptics influence calcium transport across the sarcoplasmic reticulum and can lead to increased calcium release, which may contribute to the muscle rigidity and hyperthermia seen in NMS.9
The third hypothesis involves hyperactivity of the sympathetic nervous system; it is thought that psychologic stressors alter frontal lobe function, with neuroleptics disrupting the inhibitory pathways of the sympathetic nervous system. The autonomic nervous system innervates multiple organ systems, so this excessively dysregulated sympathetic nervous system may be responsible for multiple NMS symptoms (hyperthermia, muscle rigidity, hypertension, diaphoresis, tachycardia, elevated CK.10
NMS can be caused by neuroleptic agents (both first- and second-generation antipsychotics) as well as antiemetics (Table 31). The time between use of these medications and onset of symptoms is highly variable. NMS can occur after a single dose, after a dose adjustment, or possibly after years of treatment with the same medication. It is not dose-dependent.11 In certain individuals, NMS may occur at therapeutic doses.

Continue to: Clinical presentation
Clinical presentation. Patients with NMS typically present with a tetrad of symptoms: mental status changes, muscular rigidity, hyperthermia, and autonomic instability.12 Mental status changes can include confusion and agitation, as well as catatonic signs and mutism. The muscular rigidity of NMS is characterized by “lead pipe rigidity” and may be accompanied by tremor, dystonia, or dyskinesias. Laboratory findings include elevated serum CK (from severe rigidity), often >1,000 U/L, although normal levels can be observed if rigidity has not yet developed.13
Treatment. The first step for treatment is to discontinue the causative medication.14 Initiate supportive therapy immediately to restrict the progression of symptoms. Interventions include cooling blankets, fluid resuscitation, and antihypertensives to maintain autonomic stability15 or benzodiazepines to control agitation. In severe cases, muscular rigidity may extend to the airways and intubation may be required. The severity of these symptoms may warrant admission to the ICU for close monitoring. Pharmacologic treatment with
Differentiating between SS and NMS
Differentiating between these 2 syndromes (Table 417) is critical to direct appropriate intervention. Table 517 outlines the treatment overview for SS and NMS.

Detailed history. A detailed history is imperative in making accurate diagnoses. Useful components of the history include a patient’s duration of symptoms and medication history (prescription medications as well as over-the-counter medications, supplements, and illicit drugs). Also assess for medical comorbidities, because certain medical diagnoses may alert the clinician that it is likely the patient had been prescribed serotonergic agents or neuroleptics, and renal or liver impairment may alert the clinician of decreased metabolism rates. Medication history is arguably the most useful piece of the interview, because serotonergic agents can cause SS, whereas dopamine blockers cause NMS. It should be noted that excess serotonin acts as a true toxidrome and is concentration-dependent in causing SS, whereas NMS is an idiosyncratic reaction to a drug.

Physical exam. Although there are many overlapping clinical manifestations, SS produces neuromuscular hyperactivity (ie, clonus, hyperreflexia), whereas NMS is characterized by more sluggish responses (ie, rigidity, bradyreflexia).18
Continue to: Laboratory findings
Laboratory findings. Overlap between NMS and SS also occurs with lab findings; both syndromes can result in leukocytosis, elevated CK from muscle damage, and low serum iron levels. However, these findings are more commonly associated with NMS and are seen in 75% of cases.17,19
Course of illness. Duration of symptoms can also help differentiate the 2 syndromes. SS typically develops within 24 hours of starting/changing therapy, whereas NMS symptoms can be present for days to weeks. Resolution of symptoms may also be helpful in differentiation because SS typically resolves within a few days of initiating treatment, whereas NMS resolves within 9 to 14 days of starting treatment.19
Bottom Line
The clinical presentations of serotonin syndrome (SS) and neuroleptic malignant syndrome (NMS) overlap, which can make them difficult to differentiate; however, they each have distinct approaches to treatment. Features in SS that are distinct from NMS include a history of serotonergic agents, rapid onset of symptoms, hyperreflexia, and clonus. NMS is slower in onset and can be found in patients who are prescribed dopamine antagonists, with distinct symptoms of rigidity and hyporeflexia.
Related Resources
- Kimmel R. Serotonin syndrome or NMS? Clues to diagnosis. Current Psychiatry. 2010;9(2):92.
- Strawn JR, Keck Jr PE, Caroff SN. Neuroleptic malignant syndrome: Answers to 6 tough questions. Current Psychiatry. 2008;7(1):95-101.
Drug Brand Names
Amantadine • Symmetrel
Amitriptyline • Elavil, Endep
Aripiprazole • Abilify
Bromocriptine • Cycloset, Parlodel
Bupropion • Wellbutrin, Zyban
Buspirone • BuSpar
Carbamazepine • Carbatrol, Tegretol
Chlorpromazine • Thorazine
Ciprofloxacin • Cipro
Citalopram • Celexa
Clomipramine • Anafranil
Clozapine • Clozaril
Cyclobenzaprine • Amrix, Flexeril
Cyproheptadine • Periactin
Dantrolene • Dantrium
Desipramine • Norpramin
Desvenlafaxine • Pristiq
Dextromethorphan • Benylin, Dexalone
Dolasetron • Anzemet
Doxepin • Silenor
Droperidol • Inapsine
Duloxetine • Cymbalt
Escitalopram • Lexapro
Fentanyl • Actiq, Duragesic
Fluconazole • Diflucan
Fluoxetine • Prozac
Fluphenazine • Prolixin
Fluvoxamine • Luvox
Granisetron • Kytril
Haloperidol • Haldol
Isocarboxazid • Marplan
Levomilnacipran • Fetzima
Linezolid • Zyvox
Lithium • Eskalith, Lithobid
Meperidone • Demerol
Metoclopramide • Reglan
Milnacipran • Savella
Nefazodone • Serzone
Olanzapine • Zyprexa
Ondansetron • Zofran
Paliperidone • Invega
Palonosetron • Aloxi
Paroxetine • Paxil
Pentazocine • Talwin, Talacen
Perphenazine • Trilafon
Phenelzine • Nardil
Procarbazine • Matulane
Prochlorperazine • Compazine
Promethazine • Phenergan
Quetiapine • Seroquel
Rasagiline • Azilect
Risperidone • Risperdal
Safinamide • Xadago
Selegiline • Eldepryl, Zelapar
Sertraline • Zoloft
Sibutramine • Meridia
Tedizolid • Sivextro
Thioridazine • Mellaril
Tranylcypromine • Parnate
Tramadol • Ultram
Trazodone • Desyrel, Oleptro
Venlafaxine • Effexor
Vilazodone • Viibryd
Vortioxetine • Trintellix
Valproate • Depacon
Ziprasidone • Geodon
1. Volpi-Abadie J, Kaye AM, Kaye AD. Serotonin syndrome. Ochsner J. 2013;13(4):533-540.
2. Werneke U, Jamshidi F, Taylor D, et al. Conundrums in neurology: diagnosing serotonin syndrome – a meta-analysis of cases. BMC Neurol. 2016;16:97.
3. Boyer EW, Shannon M. The serotonin syndrome. N Engl J Med. 2005;352(11):1112-1120.
4. Birmes P, Coppin D, Schmitt L, et al. Serotonin syndrome: a brief review. CMAJ. 2003;168(11):1439-1442.
5. Sternbach H. The serotonin syndrome. Am J Psychiatry. 1991;148:705-713.
6. Dunkley EJ, Isbister GK, Sibbritt D, et al. The Hunter serotonin toxicity criteria: simple and accurate diagnostic decision rules for serotonin toxicity. QJM. 2003; 96(9):635-642.
7. Lappin RI, Auchincloss EL. Treatment of the serotonin syndrome with cyproheptadine. N Engl J Med. 1994;331(15):1021-1022.
8. Nisijima K. Serotonin syndrome overlapping with neuroleptic malignant syndrome: A case report and approaches for differentially diagnosing the two syndromes. Asian J Psychiatr. 2015;18:100-101.
9. Adnet P, Lestavel P, Krivosic-Horber R. Neuroleptic malignant syndrome. Br J Anaesth. 2000;85(1):129-135.
10. Gurrera R. Sympathoadrenal hyperactivity and the etiology of neuroleptic malignant syndrome. Am J Psychiatry. 1999;156:169-180.
11. Pope HG Jr, Aizley HG, Keck PE Jr, et al. Neuroleptic malignant syndrome: long-term follow-up of 20 cases. J Clin Psychiatry. 1991;52(5):208-212.
12. Velamoor VR, Norman RM, Caroff SN, et al. Progression of symptoms in neuroleptic malignant syndrome. J Nerv Ment Dis. 1994;182(3):168-173.
13. Caroff SN, Mann SC. Neuroleptic malignant syndrome. Med Clin North Am. 1993;77(1):185-202.
14. Pileggi DJ, Cook AM. Neuroleptic malignant syndrome. Ann Pharmacother. 2016;50(11):973-981.
15. San Gabriel MC, Eddula-Changala B, Tan Y, et al. Electroconvulsive in a schizophrenic patient with neuroleptic malignant syndrome and rhabdomyolysis. J ECT. 2015;31(3):197-200.
16. Buggenhout S, Vandenberghe J, Sienaert P. Electroconvulsion therapy for neuroleptic malignant syndrome. Tijdschr Psychiatr. 2014;56(9):612-615.
17. Perry PJ, Wilborn CA. Serotonin syndrome vs neuroleptic malignant syndrome: a contrast of causes, diagnoses, and management. Ann Clin Psychiatry. 2012;24(2):155-162.
18. Mills KC. Serotonin syndrome. A clinical update. Crit Care Clin. 1997;13(4):763-783.
19. Dosi R, Ambaliya A, Joshi H, et al. Serotonin syndrome versus neuroleptic malignant syndrome: a challenge clinical quandary. BMJ Case Rep. 2014;2014:bcr201404154. doi:10.1136/bcr-2014-204154.
1. Volpi-Abadie J, Kaye AM, Kaye AD. Serotonin syndrome. Ochsner J. 2013;13(4):533-540.
2. Werneke U, Jamshidi F, Taylor D, et al. Conundrums in neurology: diagnosing serotonin syndrome – a meta-analysis of cases. BMC Neurol. 2016;16:97.
3. Boyer EW, Shannon M. The serotonin syndrome. N Engl J Med. 2005;352(11):1112-1120.
4. Birmes P, Coppin D, Schmitt L, et al. Serotonin syndrome: a brief review. CMAJ. 2003;168(11):1439-1442.
5. Sternbach H. The serotonin syndrome. Am J Psychiatry. 1991;148:705-713.
6. Dunkley EJ, Isbister GK, Sibbritt D, et al. The Hunter serotonin toxicity criteria: simple and accurate diagnostic decision rules for serotonin toxicity. QJM. 2003; 96(9):635-642.
7. Lappin RI, Auchincloss EL. Treatment of the serotonin syndrome with cyproheptadine. N Engl J Med. 1994;331(15):1021-1022.
8. Nisijima K. Serotonin syndrome overlapping with neuroleptic malignant syndrome: A case report and approaches for differentially diagnosing the two syndromes. Asian J Psychiatr. 2015;18:100-101.
9. Adnet P, Lestavel P, Krivosic-Horber R. Neuroleptic malignant syndrome. Br J Anaesth. 2000;85(1):129-135.
10. Gurrera R. Sympathoadrenal hyperactivity and the etiology of neuroleptic malignant syndrome. Am J Psychiatry. 1999;156:169-180.
11. Pope HG Jr, Aizley HG, Keck PE Jr, et al. Neuroleptic malignant syndrome: long-term follow-up of 20 cases. J Clin Psychiatry. 1991;52(5):208-212.
12. Velamoor VR, Norman RM, Caroff SN, et al. Progression of symptoms in neuroleptic malignant syndrome. J Nerv Ment Dis. 1994;182(3):168-173.
13. Caroff SN, Mann SC. Neuroleptic malignant syndrome. Med Clin North Am. 1993;77(1):185-202.
14. Pileggi DJ, Cook AM. Neuroleptic malignant syndrome. Ann Pharmacother. 2016;50(11):973-981.
15. San Gabriel MC, Eddula-Changala B, Tan Y, et al. Electroconvulsive in a schizophrenic patient with neuroleptic malignant syndrome and rhabdomyolysis. J ECT. 2015;31(3):197-200.
16. Buggenhout S, Vandenberghe J, Sienaert P. Electroconvulsion therapy for neuroleptic malignant syndrome. Tijdschr Psychiatr. 2014;56(9):612-615.
17. Perry PJ, Wilborn CA. Serotonin syndrome vs neuroleptic malignant syndrome: a contrast of causes, diagnoses, and management. Ann Clin Psychiatry. 2012;24(2):155-162.
18. Mills KC. Serotonin syndrome. A clinical update. Crit Care Clin. 1997;13(4):763-783.
19. Dosi R, Ambaliya A, Joshi H, et al. Serotonin syndrome versus neuroleptic malignant syndrome: a challenge clinical quandary. BMJ Case Rep. 2014;2014:bcr201404154. doi:10.1136/bcr-2014-204154.
Early lead exposure tied to greater psychopathology in adulthood
Lead exposure during childhood appears tied to a significant increase in the risk of psychopathology in adulthood, results of a multidecade, prospective cohort study show.
“These results suggest that early life lead exposure in the era of leaded gasoline experienced by individuals who are currently adults may have contributed to subtle, lifelong differences in emotion and behavior that are detectable at least up to 38 years of age,” Aaron Reuben and his coauthors wrote in JAMA Psychiatry.
The ongoing Dunedin longitudinal cohort study in New Zealand has followed 1,037 individuals born during 1972-1973. Of these individuals, 579 were tested for lead exposure at 11 years of age. The study assessed their mental health at 18, 21, 26, 32, and 38 years of age.
“Although follow-up studies of lead-tested children have reported the persistence of lead-related cognitive deficits well into adulthood, apart from antisocial outcomes, the long-term mental and behavioral health consequences of early life lead exposure have not been fully characterized,” wrote Mr. Reuben, a PhD student in the department of psychology and neuroscience at Duke University in Durham, N.C., and his coauthors.
Researchers saw that, for each 5-mcg/dL increase in childhood blood lead level, there was a significant 1.34-point increase in general psychopathology (P = 0.03), which was largely driven by a 1.41-point increase in internalizing (P = 0.02) and 1.30-point increase in thought-disorder symptoms (P = 0.04). Those associations were seen after adjustment for covariates, such as family socioeconomic status, maternal IQ, and family history of mental illness.
Adults who had higher lead exposure during childhood also were described by their informants – close friends or family members – as being significantly more neurotic, less agreeable, and less conscientious. However, they showed no significant differences in extroversion or in openness to experience, compared with those with less lead exposure.
“These results suggest that early-life lead exposure in the era of leaded gasoline experienced by individuals who are currently adults may have contributed to subtle, lifelong differences in emotion and behavior that are detectable at least up to 38 years of age,” the authors wrote.
They noted that the size of the effect was around one-third the size of the associations seen between psychopathology and other risk factors, such as family history of mental illness and childhood maltreatment. However, the effects of lead exposure on adult psychopathology were similar to its effects on IQ and stronger than the associations seen between lead exposure and criminal offending.
The researchers also examined how early these psychopathology symptoms could be detected with use of parent- and teacher-reported measures of antisocial behavior, hyperactivity, and internalizing from 11 years of age. This showed that individuals with higher lead exposure scored higher on these measures even at 11 years of age, “suggesting that
Mr. Reuben and his associates cited several limitations. One is that the study used a cohort that was predominantly white and born in the 1970s. Also, as an observational study, it does not establish causality between lead exposure and psychopathology.
Nevertheless, they wrote, the study results suggest that adult patients who were exposed to high levels of lead as children might benefit from increased screening and access to mental health services.
The Dunedin study is supported by the New Zealand Health Research Council and the New Zealand Ministry of Business, Innovation, and Employment. This study was supported by several entities, including the National Institute on Aging, the U.K. Medical Research Council, the National Institute of Child Health and Human Development, and the National Institute of Environmental Health Sciences. The authors reported no conflicts of interest.
SOURCE: Reuben A et al. JAMA Psychiatry. 2019 Jan 23. doi: 10.1001/jamapsychiatry.2018.4192.
Lead exposure during childhood appears tied to a significant increase in the risk of psychopathology in adulthood, results of a multidecade, prospective cohort study show.
“These results suggest that early life lead exposure in the era of leaded gasoline experienced by individuals who are currently adults may have contributed to subtle, lifelong differences in emotion and behavior that are detectable at least up to 38 years of age,” Aaron Reuben and his coauthors wrote in JAMA Psychiatry.
The ongoing Dunedin longitudinal cohort study in New Zealand has followed 1,037 individuals born during 1972-1973. Of these individuals, 579 were tested for lead exposure at 11 years of age. The study assessed their mental health at 18, 21, 26, 32, and 38 years of age.
“Although follow-up studies of lead-tested children have reported the persistence of lead-related cognitive deficits well into adulthood, apart from antisocial outcomes, the long-term mental and behavioral health consequences of early life lead exposure have not been fully characterized,” wrote Mr. Reuben, a PhD student in the department of psychology and neuroscience at Duke University in Durham, N.C., and his coauthors.
Researchers saw that, for each 5-mcg/dL increase in childhood blood lead level, there was a significant 1.34-point increase in general psychopathology (P = 0.03), which was largely driven by a 1.41-point increase in internalizing (P = 0.02) and 1.30-point increase in thought-disorder symptoms (P = 0.04). Those associations were seen after adjustment for covariates, such as family socioeconomic status, maternal IQ, and family history of mental illness.
Adults who had higher lead exposure during childhood also were described by their informants – close friends or family members – as being significantly more neurotic, less agreeable, and less conscientious. However, they showed no significant differences in extroversion or in openness to experience, compared with those with less lead exposure.
“These results suggest that early-life lead exposure in the era of leaded gasoline experienced by individuals who are currently adults may have contributed to subtle, lifelong differences in emotion and behavior that are detectable at least up to 38 years of age,” the authors wrote.
They noted that the size of the effect was around one-third the size of the associations seen between psychopathology and other risk factors, such as family history of mental illness and childhood maltreatment. However, the effects of lead exposure on adult psychopathology were similar to its effects on IQ and stronger than the associations seen between lead exposure and criminal offending.
The researchers also examined how early these psychopathology symptoms could be detected with use of parent- and teacher-reported measures of antisocial behavior, hyperactivity, and internalizing from 11 years of age. This showed that individuals with higher lead exposure scored higher on these measures even at 11 years of age, “suggesting that
Mr. Reuben and his associates cited several limitations. One is that the study used a cohort that was predominantly white and born in the 1970s. Also, as an observational study, it does not establish causality between lead exposure and psychopathology.
Nevertheless, they wrote, the study results suggest that adult patients who were exposed to high levels of lead as children might benefit from increased screening and access to mental health services.
The Dunedin study is supported by the New Zealand Health Research Council and the New Zealand Ministry of Business, Innovation, and Employment. This study was supported by several entities, including the National Institute on Aging, the U.K. Medical Research Council, the National Institute of Child Health and Human Development, and the National Institute of Environmental Health Sciences. The authors reported no conflicts of interest.
SOURCE: Reuben A et al. JAMA Psychiatry. 2019 Jan 23. doi: 10.1001/jamapsychiatry.2018.4192.
Lead exposure during childhood appears tied to a significant increase in the risk of psychopathology in adulthood, results of a multidecade, prospective cohort study show.
“These results suggest that early life lead exposure in the era of leaded gasoline experienced by individuals who are currently adults may have contributed to subtle, lifelong differences in emotion and behavior that are detectable at least up to 38 years of age,” Aaron Reuben and his coauthors wrote in JAMA Psychiatry.
The ongoing Dunedin longitudinal cohort study in New Zealand has followed 1,037 individuals born during 1972-1973. Of these individuals, 579 were tested for lead exposure at 11 years of age. The study assessed their mental health at 18, 21, 26, 32, and 38 years of age.
“Although follow-up studies of lead-tested children have reported the persistence of lead-related cognitive deficits well into adulthood, apart from antisocial outcomes, the long-term mental and behavioral health consequences of early life lead exposure have not been fully characterized,” wrote Mr. Reuben, a PhD student in the department of psychology and neuroscience at Duke University in Durham, N.C., and his coauthors.
Researchers saw that, for each 5-mcg/dL increase in childhood blood lead level, there was a significant 1.34-point increase in general psychopathology (P = 0.03), which was largely driven by a 1.41-point increase in internalizing (P = 0.02) and 1.30-point increase in thought-disorder symptoms (P = 0.04). Those associations were seen after adjustment for covariates, such as family socioeconomic status, maternal IQ, and family history of mental illness.
Adults who had higher lead exposure during childhood also were described by their informants – close friends or family members – as being significantly more neurotic, less agreeable, and less conscientious. However, they showed no significant differences in extroversion or in openness to experience, compared with those with less lead exposure.
“These results suggest that early-life lead exposure in the era of leaded gasoline experienced by individuals who are currently adults may have contributed to subtle, lifelong differences in emotion and behavior that are detectable at least up to 38 years of age,” the authors wrote.
They noted that the size of the effect was around one-third the size of the associations seen between psychopathology and other risk factors, such as family history of mental illness and childhood maltreatment. However, the effects of lead exposure on adult psychopathology were similar to its effects on IQ and stronger than the associations seen between lead exposure and criminal offending.
The researchers also examined how early these psychopathology symptoms could be detected with use of parent- and teacher-reported measures of antisocial behavior, hyperactivity, and internalizing from 11 years of age. This showed that individuals with higher lead exposure scored higher on these measures even at 11 years of age, “suggesting that
Mr. Reuben and his associates cited several limitations. One is that the study used a cohort that was predominantly white and born in the 1970s. Also, as an observational study, it does not establish causality between lead exposure and psychopathology.
Nevertheless, they wrote, the study results suggest that adult patients who were exposed to high levels of lead as children might benefit from increased screening and access to mental health services.
The Dunedin study is supported by the New Zealand Health Research Council and the New Zealand Ministry of Business, Innovation, and Employment. This study was supported by several entities, including the National Institute on Aging, the U.K. Medical Research Council, the National Institute of Child Health and Human Development, and the National Institute of Environmental Health Sciences. The authors reported no conflicts of interest.
SOURCE: Reuben A et al. JAMA Psychiatry. 2019 Jan 23. doi: 10.1001/jamapsychiatry.2018.4192.
FROM JAMA PSYCHIATRY
Key clinical point: Higher lead exposure in childhood is linked to psychopathology in adulthood.
Major finding: Children who experienced more lead exposure in childhood show more internalizing and thought-disorder symptoms in adulthood.
Study details: Longitudinal cohort study of 579 individuals.
Disclosures: The Dunedin study is supported by the New Zealand Health Research Council and the New Zealand Ministry of Business, Innovation, and Employment. This study was supported by several entities, including the National Institute on Aging, the U.K. Medical Research Council, the National Institute of Child Health and Human Development, and the National Institute of Environmental Health Sciences. The authors reported no conflicts of interest.
Source: Reuben A et al. JAMA Psychiatry. 2019 Jan 23. doi: 10.1001/jamapsychiatry.2018.4192.
FDA: Clozapine REMS modified
The Food and Drug Administration has modified the Risk Evaluation and Mitigation Strategy (REMS) Program for clozapine, a second-generation antipsychotic used for patients who do not respond adequately to standard antipsychotic treatment. Use of clozapine comes with the risk of neutropenia, which can make patients vulnerable to serious infections, so routine monitoring of absolute neutrophil counts is a must.
The new requirements, including one that requires both prescribers and pharmacies to be certified in the clozapine REMS program, take effect Feb. 28.
More information about clozapine and this change can be found on the FDA web page for the drug.
The Food and Drug Administration has modified the Risk Evaluation and Mitigation Strategy (REMS) Program for clozapine, a second-generation antipsychotic used for patients who do not respond adequately to standard antipsychotic treatment. Use of clozapine comes with the risk of neutropenia, which can make patients vulnerable to serious infections, so routine monitoring of absolute neutrophil counts is a must.
The new requirements, including one that requires both prescribers and pharmacies to be certified in the clozapine REMS program, take effect Feb. 28.
More information about clozapine and this change can be found on the FDA web page for the drug.
The Food and Drug Administration has modified the Risk Evaluation and Mitigation Strategy (REMS) Program for clozapine, a second-generation antipsychotic used for patients who do not respond adequately to standard antipsychotic treatment. Use of clozapine comes with the risk of neutropenia, which can make patients vulnerable to serious infections, so routine monitoring of absolute neutrophil counts is a must.
The new requirements, including one that requires both prescribers and pharmacies to be certified in the clozapine REMS program, take effect Feb. 28.
More information about clozapine and this change can be found on the FDA web page for the drug.
Know the red flags for synaptic autoimmune psychosis
BARCELONA – Consider the possibility of an autoantibody-related etiology in all cases of first-onset psychosis, Josep Dalmau, MD, PhD, urged at the annual congress of the European College of Neuropsychopharmacology.
“There are patients in our clinics all of us – neurologists and psychiatrists – are missing. These patients are believed to have psychiatric presentations, but they do not. They are autoimmune,” said Dr. Dalmau, professor of neurology at the University of Barcelona.
Dr. Dalmau urged psychiatrists to become familiar with the red flags suggestive of synaptic autoimmunity as the underlying cause of first-episode, out-of-the-blue psychosis.
“If you have a patient with a classical presentation of schizophrenia or bipolar disorder, you probably won’t find antibodies,” according to the neurologist.
It’s important to have a high index of suspicion, because anti–NMDA receptor encephalitis is treatable with immunotherapy. And firm evidence shows that earlier recognition and treatment lead to improved outcomes. Also, the disorder is refractory to antipsychotics; indeed, 
Manifestations of anti–NMDA receptor encephalitis follow a characteristic pattern, beginning with a prodromal flulike phase lasting several days to a week. This is followed by acute-onset bizarre behavioral changes, irritability, and psychosis with delusions and/or hallucinations, often progressing to catatonia. After 1-4 weeks of this, florid neurologic symptoms usually appear, including seizures, abnormal movements, autonomic dysregulation, and hypoventilation requiring prolonged ICU support for weeks to months. This is followed by a prolonged recovery phase lasting 5-24 months, and a period marked by deficits in executive function and working memory, impulsivity, and disinhibition. Impressively, the patient has no memory of the illness.
In one large series of patients with confirmed anti–NMDA receptor encephalitis reported by Dr. Dalmau and coinvestigators, psychiatric symptoms occurred in isolation without subsequent neurologic involvement in just 4% of cases (JAMA Neurol. 2013 Sep 1;70[9]:1133-9).
Dr. Dalmau was senior author of an international cohort study including 577 patients with anti-NMDA receptor encephalitis with serial follow-up for 24 months. The study provided an unprecedented picture of the epidemiology and clinical features of the disorder.
“It’s a disease predominantly of women and young people,” he observed.
Indeed, the median age of the study population was 21 years, and 37% of subjects were less than 18 years of age. Roughly 80% of patients were female and most of them had a benign ovarian teratoma, which played a key role in their neuropsychiatric disease (Lancet Neurol. 2013 Feb;12[2]:157-65). These benign tumors express the NMDA receptor in ectopic nerve tissue, triggering a systemic immune response.
One or more relapses – again treatable via immunotherapy – occurred in 12% of patients during 24 months of follow-up.
When a red flag suggestive of synaptic autoimmunity is present, it’s important to obtain a cerebrospinal fluid (CSF) sample for analysis, along with an EEG and/or brain MRI.
“I don’t know if you as psychiatrists are set up to do spinal taps in all persons with first presentation of psychosis, but this would be my suggestion. It’s extremely useful in this situation,” Dr. Dalmau said.
The vast majority of patients with anti–NMDA receptor encephalitis have CSF pleocytosis with a mild lymphocytic predominance. The MRI is abnormal in about 35% of cases. EEG abnormalities are common but nonspecific. The diagnosis is confirmed by identification of anti–NMDA receptor antibodies in the CSF.
First-line therapy is corticosteroids, intravenous immunoglobulin, and/or plasma exchange to remove the pathogenic antibodies, along with resection of the tumor if present. These treatments are effective in almost half of affected patients. When they’re not, the second-line options are rituximab (Rituxan) and cyclophosphamide, alone or combined.
Antibodies to the NMDA receptor are far and away the most common cause of synaptic autoimmunity-induced psychosis, but other targets of autoimmunity have been documented as well, including the alpha-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA) receptor, contactin-associated protein-like 2 (CASPR2), and neurexin-3-alpha.
Dr. Dalmau and various collaborators continue to advance the understanding of this novel category of neuropsychiatric disease. They have developed a simple 5-point score, known as the NEOS score, that predicts 1-year functional status in patients with anti–NMDA receptor encephalitis (Neurology. 2018 Dec 21. doi: 10.1212/WNL.0000000000006783). He and his colleagues have also recently shown in a prospective study that herpes simplex encephalitis can result in an autoimmune encephalitis, with NMDA receptor antibodies present in most cases (Lancet Neurol. 2018 Sep;17[9]:760-72).
Dr. Dalmau’s research is supported by the U.S. National Institute of Neurological Disorders and Stroke, the Spanish Ministry of Health, and Spanish research foundations. He reported receiving royalties from the use of several neuronal antibody tests.
BARCELONA – Consider the possibility of an autoantibody-related etiology in all cases of first-onset psychosis, Josep Dalmau, MD, PhD, urged at the annual congress of the European College of Neuropsychopharmacology.
“There are patients in our clinics all of us – neurologists and psychiatrists – are missing. These patients are believed to have psychiatric presentations, but they do not. They are autoimmune,” said Dr. Dalmau, professor of neurology at the University of Barcelona.
Dr. Dalmau urged psychiatrists to become familiar with the red flags suggestive of synaptic autoimmunity as the underlying cause of first-episode, out-of-the-blue psychosis.
“If you have a patient with a classical presentation of schizophrenia or bipolar disorder, you probably won’t find antibodies,” according to the neurologist.
It’s important to have a high index of suspicion, because anti–NMDA receptor encephalitis is treatable with immunotherapy. And firm evidence shows that earlier recognition and treatment lead to improved outcomes. Also, the disorder is refractory to antipsychotics; indeed, 
Manifestations of anti–NMDA receptor encephalitis follow a characteristic pattern, beginning with a prodromal flulike phase lasting several days to a week. This is followed by acute-onset bizarre behavioral changes, irritability, and psychosis with delusions and/or hallucinations, often progressing to catatonia. After 1-4 weeks of this, florid neurologic symptoms usually appear, including seizures, abnormal movements, autonomic dysregulation, and hypoventilation requiring prolonged ICU support for weeks to months. This is followed by a prolonged recovery phase lasting 5-24 months, and a period marked by deficits in executive function and working memory, impulsivity, and disinhibition. Impressively, the patient has no memory of the illness.
In one large series of patients with confirmed anti–NMDA receptor encephalitis reported by Dr. Dalmau and coinvestigators, psychiatric symptoms occurred in isolation without subsequent neurologic involvement in just 4% of cases (JAMA Neurol. 2013 Sep 1;70[9]:1133-9).
Dr. Dalmau was senior author of an international cohort study including 577 patients with anti-NMDA receptor encephalitis with serial follow-up for 24 months. The study provided an unprecedented picture of the epidemiology and clinical features of the disorder.
“It’s a disease predominantly of women and young people,” he observed.
Indeed, the median age of the study population was 21 years, and 37% of subjects were less than 18 years of age. Roughly 80% of patients were female and most of them had a benign ovarian teratoma, which played a key role in their neuropsychiatric disease (Lancet Neurol. 2013 Feb;12[2]:157-65). These benign tumors express the NMDA receptor in ectopic nerve tissue, triggering a systemic immune response.
One or more relapses – again treatable via immunotherapy – occurred in 12% of patients during 24 months of follow-up.
When a red flag suggestive of synaptic autoimmunity is present, it’s important to obtain a cerebrospinal fluid (CSF) sample for analysis, along with an EEG and/or brain MRI.
“I don’t know if you as psychiatrists are set up to do spinal taps in all persons with first presentation of psychosis, but this would be my suggestion. It’s extremely useful in this situation,” Dr. Dalmau said.
The vast majority of patients with anti–NMDA receptor encephalitis have CSF pleocytosis with a mild lymphocytic predominance. The MRI is abnormal in about 35% of cases. EEG abnormalities are common but nonspecific. The diagnosis is confirmed by identification of anti–NMDA receptor antibodies in the CSF.
First-line therapy is corticosteroids, intravenous immunoglobulin, and/or plasma exchange to remove the pathogenic antibodies, along with resection of the tumor if present. These treatments are effective in almost half of affected patients. When they’re not, the second-line options are rituximab (Rituxan) and cyclophosphamide, alone or combined.
Antibodies to the NMDA receptor are far and away the most common cause of synaptic autoimmunity-induced psychosis, but other targets of autoimmunity have been documented as well, including the alpha-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA) receptor, contactin-associated protein-like 2 (CASPR2), and neurexin-3-alpha.
Dr. Dalmau and various collaborators continue to advance the understanding of this novel category of neuropsychiatric disease. They have developed a simple 5-point score, known as the NEOS score, that predicts 1-year functional status in patients with anti–NMDA receptor encephalitis (Neurology. 2018 Dec 21. doi: 10.1212/WNL.0000000000006783). He and his colleagues have also recently shown in a prospective study that herpes simplex encephalitis can result in an autoimmune encephalitis, with NMDA receptor antibodies present in most cases (Lancet Neurol. 2018 Sep;17[9]:760-72).
Dr. Dalmau’s research is supported by the U.S. National Institute of Neurological Disorders and Stroke, the Spanish Ministry of Health, and Spanish research foundations. He reported receiving royalties from the use of several neuronal antibody tests.
BARCELONA – Consider the possibility of an autoantibody-related etiology in all cases of first-onset psychosis, Josep Dalmau, MD, PhD, urged at the annual congress of the European College of Neuropsychopharmacology.
“There are patients in our clinics all of us – neurologists and psychiatrists – are missing. These patients are believed to have psychiatric presentations, but they do not. They are autoimmune,” said Dr. Dalmau, professor of neurology at the University of Barcelona.
Dr. Dalmau urged psychiatrists to become familiar with the red flags suggestive of synaptic autoimmunity as the underlying cause of first-episode, out-of-the-blue psychosis.
“If you have a patient with a classical presentation of schizophrenia or bipolar disorder, you probably won’t find antibodies,” according to the neurologist.
It’s important to have a high index of suspicion, because anti–NMDA receptor encephalitis is treatable with immunotherapy. And firm evidence shows that earlier recognition and treatment lead to improved outcomes. Also, the disorder is refractory to antipsychotics; indeed, 
Manifestations of anti–NMDA receptor encephalitis follow a characteristic pattern, beginning with a prodromal flulike phase lasting several days to a week. This is followed by acute-onset bizarre behavioral changes, irritability, and psychosis with delusions and/or hallucinations, often progressing to catatonia. After 1-4 weeks of this, florid neurologic symptoms usually appear, including seizures, abnormal movements, autonomic dysregulation, and hypoventilation requiring prolonged ICU support for weeks to months. This is followed by a prolonged recovery phase lasting 5-24 months, and a period marked by deficits in executive function and working memory, impulsivity, and disinhibition. Impressively, the patient has no memory of the illness.
In one large series of patients with confirmed anti–NMDA receptor encephalitis reported by Dr. Dalmau and coinvestigators, psychiatric symptoms occurred in isolation without subsequent neurologic involvement in just 4% of cases (JAMA Neurol. 2013 Sep 1;70[9]:1133-9).
Dr. Dalmau was senior author of an international cohort study including 577 patients with anti-NMDA receptor encephalitis with serial follow-up for 24 months. The study provided an unprecedented picture of the epidemiology and clinical features of the disorder.
“It’s a disease predominantly of women and young people,” he observed.
Indeed, the median age of the study population was 21 years, and 37% of subjects were less than 18 years of age. Roughly 80% of patients were female and most of them had a benign ovarian teratoma, which played a key role in their neuropsychiatric disease (Lancet Neurol. 2013 Feb;12[2]:157-65). These benign tumors express the NMDA receptor in ectopic nerve tissue, triggering a systemic immune response.
One or more relapses – again treatable via immunotherapy – occurred in 12% of patients during 24 months of follow-up.
When a red flag suggestive of synaptic autoimmunity is present, it’s important to obtain a cerebrospinal fluid (CSF) sample for analysis, along with an EEG and/or brain MRI.
“I don’t know if you as psychiatrists are set up to do spinal taps in all persons with first presentation of psychosis, but this would be my suggestion. It’s extremely useful in this situation,” Dr. Dalmau said.
The vast majority of patients with anti–NMDA receptor encephalitis have CSF pleocytosis with a mild lymphocytic predominance. The MRI is abnormal in about 35% of cases. EEG abnormalities are common but nonspecific. The diagnosis is confirmed by identification of anti–NMDA receptor antibodies in the CSF.
First-line therapy is corticosteroids, intravenous immunoglobulin, and/or plasma exchange to remove the pathogenic antibodies, along with resection of the tumor if present. These treatments are effective in almost half of affected patients. When they’re not, the second-line options are rituximab (Rituxan) and cyclophosphamide, alone or combined.
Antibodies to the NMDA receptor are far and away the most common cause of synaptic autoimmunity-induced psychosis, but other targets of autoimmunity have been documented as well, including the alpha-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA) receptor, contactin-associated protein-like 2 (CASPR2), and neurexin-3-alpha.
Dr. Dalmau and various collaborators continue to advance the understanding of this novel category of neuropsychiatric disease. They have developed a simple 5-point score, known as the NEOS score, that predicts 1-year functional status in patients with anti–NMDA receptor encephalitis (Neurology. 2018 Dec 21. doi: 10.1212/WNL.0000000000006783). He and his colleagues have also recently shown in a prospective study that herpes simplex encephalitis can result in an autoimmune encephalitis, with NMDA receptor antibodies present in most cases (Lancet Neurol. 2018 Sep;17[9]:760-72).
Dr. Dalmau’s research is supported by the U.S. National Institute of Neurological Disorders and Stroke, the Spanish Ministry of Health, and Spanish research foundations. He reported receiving royalties from the use of several neuronal antibody tests.
REPORTING FROM THE ECNP CONGRESS
A prescription for ‘deprescribing’: A case report
In 2016, Swapnil Gupta, MD, and John Daniel Cahill, MD, PhD, challenged the field of psychiatry to reexamine our prescribing patterns. They warned against our use of polypharmacy when not attached to improvement in functioning for our patients.1 They were concerned about the lack of evidence for those treatment regimens and for our diagnostic criteria. In their inspiring article, they described how psychiatrists might proceed in the process of “deprescribing” – which they define as a process of pharmacologic regimen optimization through reducing or ending medications for which “benefits no longer outweigh risks.”1
In my practice, I routinely confront medication regimens that I have never encountered in the literature. The evidence for two psychotropics is limited but certainly available, in particular adjunct treatment of antidepressants2 and mood stabilizers.3 The evidence supporting the use of more than two psychotropics, however, is quite sparse. Yet, patients often enter my office on more than five psychotropics. I am also confronted with poorly defined diagnostic labels – which present more as means to justify polypharmacy than a thorough review of the patient’s current state.
Dr. Gupta and Dr. Cahill recommend a series of steps aimed at attempting the deprescription of psychotropics. Those steps include timeliness, knowledge of the patient’s current regimen, discussion about the risk of prescriptions, discussion about deprescribing, choosing the right medications to stop, a plan for describing, and monitoring. In the case presented below, I used some of those steps in an effort to provide the best care for the patient. Key details of the case have been changed, including the name, to protect the patient’s confidentiality.
Overview of the case
Rosalie Bertin is a 54-year-old female who has been treated for depression by a variety of primary care physicians for the better part of the last 30 years. She had tried an array of antidepressants, including sertraline, citalopram, duloxetine, and mirtazapine, over that time. Each seemed to provide some benefit when reviewing the notes, but there is no mention of why she was continued on those medications despite the absence of continuing symptoms. Occasionally, Rosalie would present to her clinician tearful and endorsing sadness, though the record did not comment on reports of energy, concentration, sleep, appetite, and interest.
In 2014, Rosalie’s husband passed away from lung cancer. His death was fairly quick, and initially, Rosalie did not mention any significant emotional complaints. However, when visiting her primary care physician 4 months later, she was noted to experience auditory hallucinations. “Sometimes I hear my husband when I am alone in my home,” she said. Rosalie was referred to a psychiatrist with a diagnosis of “psychosis not otherwise specified.”
When discussing her condition with the psychiatrist, Rosalie mentioned experiencing low mood, and having diminished interest in engaging in activities. “I miss Marc when I go places; I used to do everything with him.” She reported hearing him often but only when at her home. He would say things like, “I miss you,” or ask her about her day. She was diagnosed with “major depressive disorder with psychotic features.” Risperidone was added to the escitalopram, buspirone, and gabapentin that had been started by her primary care physician.
After several months of psychotropic management, the dose of risperidone was titrated to 8 mg per day. Her mood symptoms were unchanged, but she now was complaining of poor concentration and memory. The psychiatrist performed a Mini-Mental State Examination (MMSE). It was noted that taking the MMSE engendered significant anxiety for the patient. Rosalie received a score suggesting mild cognitive impairment. She was started on donepezil for the memory complaint, quetiapine for the continued voices, and recommended for disability.
Once on short-term disability, the patient relocated to live closer to her mother in San Diego and subsequently contacted me about continuing psychiatric care.
Initial visit
Rosalie is a petite white woman, raised in the Midwest, who married her high school sweetheart, and subsequently became an administrative assistant. Rosalie and Marc were unable to have children. Marc was an engineer, and a longtime smoker. She describes their lives as simple – “few friends, few vacations, few problems, few regrets.” She states she misses her husband and often cries when thinking about him.
When asked about psychiatric diagnoses, she answered: “I have psychosis. … My doctor said maybe schizophrenia, but he is not sure yet.” She described schizophrenia as hearing voices. Rosalie also mentioned having memory problems: “They cannot tell if it is Alzheimer’s disease until I die and they look at my brain, but the medication should delay the progression.”
She reported no significant effect from her prior antidepressant trials: “I am not sure if or how they helped.” Rosalie could not explain the role of the medication. “I take medications as prescribed by my doctor,” she said.
When discussing her antipsychotics, she mentioned: “Those are strong medications; it is hard for me to stay awake with them.” She declared having had no changes in the voices while on the risperidone but said they went away since also being on the quetiapine: “I wonder if the combination of the two really fixed my brain imbalance.”
Assessment
I admit that I have a critical bias against the overuse of psychotropics, and this might have painted how I interpreted Rosalie’s story. Nonetheless, I was honest with her and told her of my concerns. I informed her that her diagnosis was not consistent with my understanding of mood and thought disorders. Her initial reports of depression neither met the DSM criteria for depression nor felt consistent with my conceptualization of the illness. She had retained appropriate functioning and seemed to be responding with the sadness expected when facing difficult challenges like grief.
Her subsequent reports of auditory hallucinations were not associated with delusions or forms of disorganization that I would expect in someone with a thought disorder. Furthermore, the context of the onset gave me the impression that this was part of her process of grief. Her poor result in the dementia screen was most surprising and inconsistent with my evaluation. I told her that I suspected that she was not suffering from Alzheimer’s but from being overmedicated and from anxiety at the time of the testing.
She was excited and hesitant about my report. She was surprised by the length of our visit and interested in hearing more from me. Strangely, I wished she had challenged my different approach. I think that I was hoping she would question my conceptualization, the way I hoped she would have done with her prior clinicians. Nonetheless, she agreed to make a plan with me.
Treatment plan
We decided to review each of her medications and discuss their benefits and risks over a couple of visits. She was most eager to discontinue the donepezil, which had caused diarrhea. She was concerned when I informed her of the potential side effects of antipsychotics. “My doctor asked me if I had any side effects at each visit; I answered that I felt nothing wrong; I had not realized that side effects could appear later.”
She was adamant about staying on buspirone, as she felt it helped her the most with her anxiety at social events. She voiced concern about discontinuing the antipsychotics despite being unsettled by my review of their risks. She asked that we taper them slowly.
In regard to receiving psychosocial support throughout this period of deprescribing, Rosalie declined weekly psychotherapy. She reported having a good social network in San Diego that she wanted to rely on.
Outcome
I often worry about consequences of stopping a medication, especially when I was not present at the time of its initiation. I agonize that the patient might relapse from my need to carry out my agenda on deprescribing. I try to remind myself that the evidence supports my decision making. The risks of psychotropics often are slow to show up, making the benefit of deprescribing less tangible. However, this case was straightforward.
Rosalie quickly improved. Tapering the antipsychotics was astonishing to her: “I can think clearly again.” Within 6 months, she was on buspirone only – though willing to discuss its discontinuation. She had a lead for a job and was hoping to return to work soon. Rosalie continued to miss her husband but had not heard him in some time. She has not had symptoms of psychosis or depression. Her cognition and mood were intact on my clinical assessment.
Discussion
Sadly and shockingly, cases like that of Rosalie are common. In my practice, I routinely see patients on multiple psychotropics – often on more than one antipsychotic. Their diagnoses are vague and dubious, and include diagnoses such as “unspecified psychosis” and “cognitive impairments.” Clinicians occasionally worry about relapse and promote a narrative that treatment must be not only long term but lifelong.4 There is some evidence for this perspective in a research context, but the clinical world also is filled with patients like Rosalie.
Her reports of auditory hallucinations were better explained by her grief than by a psychotic process.5 Her memory complaints were better explained by anxiety at the time of her testing while suffering from the side effects from her numerous psychotropics.6 Her depressive complaints were better explained by appropriate sadness in response to stressors. Several months later with fewer diagnoses and far fewer psychotropics, she is functioning better.
Take-home points
- Polypharmacy can lead to psychiatric symptoms and functional impairment.
- Patients often are unaware of the complete risks of psychotropics.
- Psychiatric symptoms are not always associated with a psychiatric disorder.
- Deprescribing can be performed safely and effectively.
- Deprescribing can be performed with the patient’s informed consent and agreement.
References
1. Psychiatr Serv. 2016 Aug 1;67(8):904-7.
2. Focus. 2016 Apr 13; doi: 10.1176/appi.focus.20150041.
3. Bipolar Disord. 2016 Dec;18(8):684-91.
4. Am J Psychiatry. 2017 Sep 1;174(9):840-9.
5. World Psychiatry. 2009 Jun;8(2):67-74.
6. Hosp Community Psychiatry. 1983 Sep;34(9):830-5.
Dr. Badre is a forensic psychiatrist in San Diego and an expert in correctional mental health. He holds teaching positions at the University of California, San Diego, and the University of San Diego. He teaches medical education, psychopharmacology, ethics in psychiatry, and correctional care. Among his writings is chapter 7 in the new book “Critical Psychiatry: Controversies and Clinical Implications” (Springer, 2019).
*This column was updated 1/11/2019.
In 2016, Swapnil Gupta, MD, and John Daniel Cahill, MD, PhD, challenged the field of psychiatry to reexamine our prescribing patterns. They warned against our use of polypharmacy when not attached to improvement in functioning for our patients.1 They were concerned about the lack of evidence for those treatment regimens and for our diagnostic criteria. In their inspiring article, they described how psychiatrists might proceed in the process of “deprescribing” – which they define as a process of pharmacologic regimen optimization through reducing or ending medications for which “benefits no longer outweigh risks.”1
In my practice, I routinely confront medication regimens that I have never encountered in the literature. The evidence for two psychotropics is limited but certainly available, in particular adjunct treatment of antidepressants2 and mood stabilizers.3 The evidence supporting the use of more than two psychotropics, however, is quite sparse. Yet, patients often enter my office on more than five psychotropics. I am also confronted with poorly defined diagnostic labels – which present more as means to justify polypharmacy than a thorough review of the patient’s current state.
Dr. Gupta and Dr. Cahill recommend a series of steps aimed at attempting the deprescription of psychotropics. Those steps include timeliness, knowledge of the patient’s current regimen, discussion about the risk of prescriptions, discussion about deprescribing, choosing the right medications to stop, a plan for describing, and monitoring. In the case presented below, I used some of those steps in an effort to provide the best care for the patient. Key details of the case have been changed, including the name, to protect the patient’s confidentiality.
Overview of the case
Rosalie Bertin is a 54-year-old female who has been treated for depression by a variety of primary care physicians for the better part of the last 30 years. She had tried an array of antidepressants, including sertraline, citalopram, duloxetine, and mirtazapine, over that time. Each seemed to provide some benefit when reviewing the notes, but there is no mention of why she was continued on those medications despite the absence of continuing symptoms. Occasionally, Rosalie would present to her clinician tearful and endorsing sadness, though the record did not comment on reports of energy, concentration, sleep, appetite, and interest.
In 2014, Rosalie’s husband passed away from lung cancer. His death was fairly quick, and initially, Rosalie did not mention any significant emotional complaints. However, when visiting her primary care physician 4 months later, she was noted to experience auditory hallucinations. “Sometimes I hear my husband when I am alone in my home,” she said. Rosalie was referred to a psychiatrist with a diagnosis of “psychosis not otherwise specified.”
When discussing her condition with the psychiatrist, Rosalie mentioned experiencing low mood, and having diminished interest in engaging in activities. “I miss Marc when I go places; I used to do everything with him.” She reported hearing him often but only when at her home. He would say things like, “I miss you,” or ask her about her day. She was diagnosed with “major depressive disorder with psychotic features.” Risperidone was added to the escitalopram, buspirone, and gabapentin that had been started by her primary care physician.
After several months of psychotropic management, the dose of risperidone was titrated to 8 mg per day. Her mood symptoms were unchanged, but she now was complaining of poor concentration and memory. The psychiatrist performed a Mini-Mental State Examination (MMSE). It was noted that taking the MMSE engendered significant anxiety for the patient. Rosalie received a score suggesting mild cognitive impairment. She was started on donepezil for the memory complaint, quetiapine for the continued voices, and recommended for disability.
Once on short-term disability, the patient relocated to live closer to her mother in San Diego and subsequently contacted me about continuing psychiatric care.
Initial visit
Rosalie is a petite white woman, raised in the Midwest, who married her high school sweetheart, and subsequently became an administrative assistant. Rosalie and Marc were unable to have children. Marc was an engineer, and a longtime smoker. She describes their lives as simple – “few friends, few vacations, few problems, few regrets.” She states she misses her husband and often cries when thinking about him.
When asked about psychiatric diagnoses, she answered: “I have psychosis. … My doctor said maybe schizophrenia, but he is not sure yet.” She described schizophrenia as hearing voices. Rosalie also mentioned having memory problems: “They cannot tell if it is Alzheimer’s disease until I die and they look at my brain, but the medication should delay the progression.”
She reported no significant effect from her prior antidepressant trials: “I am not sure if or how they helped.” Rosalie could not explain the role of the medication. “I take medications as prescribed by my doctor,” she said.
When discussing her antipsychotics, she mentioned: “Those are strong medications; it is hard for me to stay awake with them.” She declared having had no changes in the voices while on the risperidone but said they went away since also being on the quetiapine: “I wonder if the combination of the two really fixed my brain imbalance.”
Assessment
I admit that I have a critical bias against the overuse of psychotropics, and this might have painted how I interpreted Rosalie’s story. Nonetheless, I was honest with her and told her of my concerns. I informed her that her diagnosis was not consistent with my understanding of mood and thought disorders. Her initial reports of depression neither met the DSM criteria for depression nor felt consistent with my conceptualization of the illness. She had retained appropriate functioning and seemed to be responding with the sadness expected when facing difficult challenges like grief.
Her subsequent reports of auditory hallucinations were not associated with delusions or forms of disorganization that I would expect in someone with a thought disorder. Furthermore, the context of the onset gave me the impression that this was part of her process of grief. Her poor result in the dementia screen was most surprising and inconsistent with my evaluation. I told her that I suspected that she was not suffering from Alzheimer’s but from being overmedicated and from anxiety at the time of the testing.
She was excited and hesitant about my report. She was surprised by the length of our visit and interested in hearing more from me. Strangely, I wished she had challenged my different approach. I think that I was hoping she would question my conceptualization, the way I hoped she would have done with her prior clinicians. Nonetheless, she agreed to make a plan with me.
Treatment plan
We decided to review each of her medications and discuss their benefits and risks over a couple of visits. She was most eager to discontinue the donepezil, which had caused diarrhea. She was concerned when I informed her of the potential side effects of antipsychotics. “My doctor asked me if I had any side effects at each visit; I answered that I felt nothing wrong; I had not realized that side effects could appear later.”
She was adamant about staying on buspirone, as she felt it helped her the most with her anxiety at social events. She voiced concern about discontinuing the antipsychotics despite being unsettled by my review of their risks. She asked that we taper them slowly.
In regard to receiving psychosocial support throughout this period of deprescribing, Rosalie declined weekly psychotherapy. She reported having a good social network in San Diego that she wanted to rely on.
Outcome
I often worry about consequences of stopping a medication, especially when I was not present at the time of its initiation. I agonize that the patient might relapse from my need to carry out my agenda on deprescribing. I try to remind myself that the evidence supports my decision making. The risks of psychotropics often are slow to show up, making the benefit of deprescribing less tangible. However, this case was straightforward.
Rosalie quickly improved. Tapering the antipsychotics was astonishing to her: “I can think clearly again.” Within 6 months, she was on buspirone only – though willing to discuss its discontinuation. She had a lead for a job and was hoping to return to work soon. Rosalie continued to miss her husband but had not heard him in some time. She has not had symptoms of psychosis or depression. Her cognition and mood were intact on my clinical assessment.
Discussion
Sadly and shockingly, cases like that of Rosalie are common. In my practice, I routinely see patients on multiple psychotropics – often on more than one antipsychotic. Their diagnoses are vague and dubious, and include diagnoses such as “unspecified psychosis” and “cognitive impairments.” Clinicians occasionally worry about relapse and promote a narrative that treatment must be not only long term but lifelong.4 There is some evidence for this perspective in a research context, but the clinical world also is filled with patients like Rosalie.
Her reports of auditory hallucinations were better explained by her grief than by a psychotic process.5 Her memory complaints were better explained by anxiety at the time of her testing while suffering from the side effects from her numerous psychotropics.6 Her depressive complaints were better explained by appropriate sadness in response to stressors. Several months later with fewer diagnoses and far fewer psychotropics, she is functioning better.
Take-home points
- Polypharmacy can lead to psychiatric symptoms and functional impairment.
- Patients often are unaware of the complete risks of psychotropics.
- Psychiatric symptoms are not always associated with a psychiatric disorder.
- Deprescribing can be performed safely and effectively.
- Deprescribing can be performed with the patient’s informed consent and agreement.
References
1. Psychiatr Serv. 2016 Aug 1;67(8):904-7.
2. Focus. 2016 Apr 13; doi: 10.1176/appi.focus.20150041.
3. Bipolar Disord. 2016 Dec;18(8):684-91.
4. Am J Psychiatry. 2017 Sep 1;174(9):840-9.
5. World Psychiatry. 2009 Jun;8(2):67-74.
6. Hosp Community Psychiatry. 1983 Sep;34(9):830-5.
Dr. Badre is a forensic psychiatrist in San Diego and an expert in correctional mental health. He holds teaching positions at the University of California, San Diego, and the University of San Diego. He teaches medical education, psychopharmacology, ethics in psychiatry, and correctional care. Among his writings is chapter 7 in the new book “Critical Psychiatry: Controversies and Clinical Implications” (Springer, 2019).
*This column was updated 1/11/2019.
In 2016, Swapnil Gupta, MD, and John Daniel Cahill, MD, PhD, challenged the field of psychiatry to reexamine our prescribing patterns. They warned against our use of polypharmacy when not attached to improvement in functioning for our patients.1 They were concerned about the lack of evidence for those treatment regimens and for our diagnostic criteria. In their inspiring article, they described how psychiatrists might proceed in the process of “deprescribing” – which they define as a process of pharmacologic regimen optimization through reducing or ending medications for which “benefits no longer outweigh risks.”1
In my practice, I routinely confront medication regimens that I have never encountered in the literature. The evidence for two psychotropics is limited but certainly available, in particular adjunct treatment of antidepressants2 and mood stabilizers.3 The evidence supporting the use of more than two psychotropics, however, is quite sparse. Yet, patients often enter my office on more than five psychotropics. I am also confronted with poorly defined diagnostic labels – which present more as means to justify polypharmacy than a thorough review of the patient’s current state.
Dr. Gupta and Dr. Cahill recommend a series of steps aimed at attempting the deprescription of psychotropics. Those steps include timeliness, knowledge of the patient’s current regimen, discussion about the risk of prescriptions, discussion about deprescribing, choosing the right medications to stop, a plan for describing, and monitoring. In the case presented below, I used some of those steps in an effort to provide the best care for the patient. Key details of the case have been changed, including the name, to protect the patient’s confidentiality.
Overview of the case
Rosalie Bertin is a 54-year-old female who has been treated for depression by a variety of primary care physicians for the better part of the last 30 years. She had tried an array of antidepressants, including sertraline, citalopram, duloxetine, and mirtazapine, over that time. Each seemed to provide some benefit when reviewing the notes, but there is no mention of why she was continued on those medications despite the absence of continuing symptoms. Occasionally, Rosalie would present to her clinician tearful and endorsing sadness, though the record did not comment on reports of energy, concentration, sleep, appetite, and interest.
In 2014, Rosalie’s husband passed away from lung cancer. His death was fairly quick, and initially, Rosalie did not mention any significant emotional complaints. However, when visiting her primary care physician 4 months later, she was noted to experience auditory hallucinations. “Sometimes I hear my husband when I am alone in my home,” she said. Rosalie was referred to a psychiatrist with a diagnosis of “psychosis not otherwise specified.”
When discussing her condition with the psychiatrist, Rosalie mentioned experiencing low mood, and having diminished interest in engaging in activities. “I miss Marc when I go places; I used to do everything with him.” She reported hearing him often but only when at her home. He would say things like, “I miss you,” or ask her about her day. She was diagnosed with “major depressive disorder with psychotic features.” Risperidone was added to the escitalopram, buspirone, and gabapentin that had been started by her primary care physician.
After several months of psychotropic management, the dose of risperidone was titrated to 8 mg per day. Her mood symptoms were unchanged, but she now was complaining of poor concentration and memory. The psychiatrist performed a Mini-Mental State Examination (MMSE). It was noted that taking the MMSE engendered significant anxiety for the patient. Rosalie received a score suggesting mild cognitive impairment. She was started on donepezil for the memory complaint, quetiapine for the continued voices, and recommended for disability.
Once on short-term disability, the patient relocated to live closer to her mother in San Diego and subsequently contacted me about continuing psychiatric care.
Initial visit
Rosalie is a petite white woman, raised in the Midwest, who married her high school sweetheart, and subsequently became an administrative assistant. Rosalie and Marc were unable to have children. Marc was an engineer, and a longtime smoker. She describes their lives as simple – “few friends, few vacations, few problems, few regrets.” She states she misses her husband and often cries when thinking about him.
When asked about psychiatric diagnoses, she answered: “I have psychosis. … My doctor said maybe schizophrenia, but he is not sure yet.” She described schizophrenia as hearing voices. Rosalie also mentioned having memory problems: “They cannot tell if it is Alzheimer’s disease until I die and they look at my brain, but the medication should delay the progression.”
She reported no significant effect from her prior antidepressant trials: “I am not sure if or how they helped.” Rosalie could not explain the role of the medication. “I take medications as prescribed by my doctor,” she said.
When discussing her antipsychotics, she mentioned: “Those are strong medications; it is hard for me to stay awake with them.” She declared having had no changes in the voices while on the risperidone but said they went away since also being on the quetiapine: “I wonder if the combination of the two really fixed my brain imbalance.”
Assessment
I admit that I have a critical bias against the overuse of psychotropics, and this might have painted how I interpreted Rosalie’s story. Nonetheless, I was honest with her and told her of my concerns. I informed her that her diagnosis was not consistent with my understanding of mood and thought disorders. Her initial reports of depression neither met the DSM criteria for depression nor felt consistent with my conceptualization of the illness. She had retained appropriate functioning and seemed to be responding with the sadness expected when facing difficult challenges like grief.
Her subsequent reports of auditory hallucinations were not associated with delusions or forms of disorganization that I would expect in someone with a thought disorder. Furthermore, the context of the onset gave me the impression that this was part of her process of grief. Her poor result in the dementia screen was most surprising and inconsistent with my evaluation. I told her that I suspected that she was not suffering from Alzheimer’s but from being overmedicated and from anxiety at the time of the testing.
She was excited and hesitant about my report. She was surprised by the length of our visit and interested in hearing more from me. Strangely, I wished she had challenged my different approach. I think that I was hoping she would question my conceptualization, the way I hoped she would have done with her prior clinicians. Nonetheless, she agreed to make a plan with me.
Treatment plan
We decided to review each of her medications and discuss their benefits and risks over a couple of visits. She was most eager to discontinue the donepezil, which had caused diarrhea. She was concerned when I informed her of the potential side effects of antipsychotics. “My doctor asked me if I had any side effects at each visit; I answered that I felt nothing wrong; I had not realized that side effects could appear later.”
She was adamant about staying on buspirone, as she felt it helped her the most with her anxiety at social events. She voiced concern about discontinuing the antipsychotics despite being unsettled by my review of their risks. She asked that we taper them slowly.
In regard to receiving psychosocial support throughout this period of deprescribing, Rosalie declined weekly psychotherapy. She reported having a good social network in San Diego that she wanted to rely on.
Outcome
I often worry about consequences of stopping a medication, especially when I was not present at the time of its initiation. I agonize that the patient might relapse from my need to carry out my agenda on deprescribing. I try to remind myself that the evidence supports my decision making. The risks of psychotropics often are slow to show up, making the benefit of deprescribing less tangible. However, this case was straightforward.
Rosalie quickly improved. Tapering the antipsychotics was astonishing to her: “I can think clearly again.” Within 6 months, she was on buspirone only – though willing to discuss its discontinuation. She had a lead for a job and was hoping to return to work soon. Rosalie continued to miss her husband but had not heard him in some time. She has not had symptoms of psychosis or depression. Her cognition and mood were intact on my clinical assessment.
Discussion
Sadly and shockingly, cases like that of Rosalie are common. In my practice, I routinely see patients on multiple psychotropics – often on more than one antipsychotic. Their diagnoses are vague and dubious, and include diagnoses such as “unspecified psychosis” and “cognitive impairments.” Clinicians occasionally worry about relapse and promote a narrative that treatment must be not only long term but lifelong.4 There is some evidence for this perspective in a research context, but the clinical world also is filled with patients like Rosalie.
Her reports of auditory hallucinations were better explained by her grief than by a psychotic process.5 Her memory complaints were better explained by anxiety at the time of her testing while suffering from the side effects from her numerous psychotropics.6 Her depressive complaints were better explained by appropriate sadness in response to stressors. Several months later with fewer diagnoses and far fewer psychotropics, she is functioning better.
Take-home points
- Polypharmacy can lead to psychiatric symptoms and functional impairment.
- Patients often are unaware of the complete risks of psychotropics.
- Psychiatric symptoms are not always associated with a psychiatric disorder.
- Deprescribing can be performed safely and effectively.
- Deprescribing can be performed with the patient’s informed consent and agreement.
References
1. Psychiatr Serv. 2016 Aug 1;67(8):904-7.
2. Focus. 2016 Apr 13; doi: 10.1176/appi.focus.20150041.
3. Bipolar Disord. 2016 Dec;18(8):684-91.
4. Am J Psychiatry. 2017 Sep 1;174(9):840-9.
5. World Psychiatry. 2009 Jun;8(2):67-74.
6. Hosp Community Psychiatry. 1983 Sep;34(9):830-5.
Dr. Badre is a forensic psychiatrist in San Diego and an expert in correctional mental health. He holds teaching positions at the University of California, San Diego, and the University of San Diego. He teaches medical education, psychopharmacology, ethics in psychiatry, and correctional care. Among his writings is chapter 7 in the new book “Critical Psychiatry: Controversies and Clinical Implications” (Springer, 2019).
*This column was updated 1/11/2019.











