User login
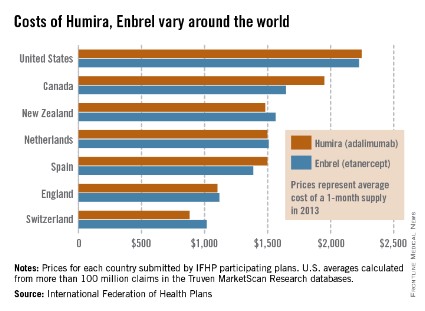
Humira, Enbrel costs highest in the United States
The costs of Humira and Enbrel are higher in the United States, compared with other industrialized countries included in the International Federation of Health Plans’ 2013 Comparative Price Report.
The average cost of a 1-month supply was $2,246 for Humira (adalimumab) in 2013 and $2,225 for Enbrel (etanercept). In Canada, which had second-highest cost for both drugs, Humira cost $1,950 per month and Enbrel cost $1,646 per month, the IFHP reported.
Switzerland had the lowest cost for both drugs among the countries included in the comparison: $881 for Humira and $1,017 for Enbrel.
In the United States, Humira is approved for the treatment of rheumatoid arthritis, juvenile idiopathic arthritis, psoriatic arthritis, ankylosing spondylitis, Crohn’s disease, ulcerative colitis, and plaque psoriasis. Enbrel is approved for RA, polyarticular juvenile idiopathic arthritis in patients aged 2 years or older, psoriatic arthritis, ankylosing spondylitis, and plaque psoriasis.
The IFHP comprises more than 100 member companies in 25 countries. For the survey, the price for each country was submitted by participating member plans. Some prices are drawn from the public sector, some from the private, and some from both. U.S. averages were calculated from more than 100 million claims in the Truven MarketScan Research databases.
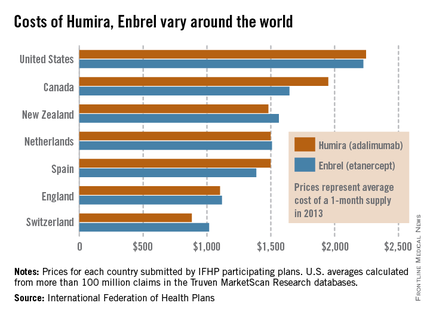
The costs of Humira and Enbrel are higher in the United States, compared with other industrialized countries included in the International Federation of Health Plans’ 2013 Comparative Price Report.
The average cost of a 1-month supply was $2,246 for Humira (adalimumab) in 2013 and $2,225 for Enbrel (etanercept). In Canada, which had second-highest cost for both drugs, Humira cost $1,950 per month and Enbrel cost $1,646 per month, the IFHP reported.
Switzerland had the lowest cost for both drugs among the countries included in the comparison: $881 for Humira and $1,017 for Enbrel.
In the United States, Humira is approved for the treatment of rheumatoid arthritis, juvenile idiopathic arthritis, psoriatic arthritis, ankylosing spondylitis, Crohn’s disease, ulcerative colitis, and plaque psoriasis. Enbrel is approved for RA, polyarticular juvenile idiopathic arthritis in patients aged 2 years or older, psoriatic arthritis, ankylosing spondylitis, and plaque psoriasis.
The IFHP comprises more than 100 member companies in 25 countries. For the survey, the price for each country was submitted by participating member plans. Some prices are drawn from the public sector, some from the private, and some from both. U.S. averages were calculated from more than 100 million claims in the Truven MarketScan Research databases.

The costs of Humira and Enbrel are higher in the United States, compared with other industrialized countries included in the International Federation of Health Plans’ 2013 Comparative Price Report.
The average cost of a 1-month supply was $2,246 for Humira (adalimumab) in 2013 and $2,225 for Enbrel (etanercept). In Canada, which had second-highest cost for both drugs, Humira cost $1,950 per month and Enbrel cost $1,646 per month, the IFHP reported.
Switzerland had the lowest cost for both drugs among the countries included in the comparison: $881 for Humira and $1,017 for Enbrel.
In the United States, Humira is approved for the treatment of rheumatoid arthritis, juvenile idiopathic arthritis, psoriatic arthritis, ankylosing spondylitis, Crohn’s disease, ulcerative colitis, and plaque psoriasis. Enbrel is approved for RA, polyarticular juvenile idiopathic arthritis in patients aged 2 years or older, psoriatic arthritis, ankylosing spondylitis, and plaque psoriasis.
The IFHP comprises more than 100 member companies in 25 countries. For the survey, the price for each country was submitted by participating member plans. Some prices are drawn from the public sector, some from the private, and some from both. U.S. averages were calculated from more than 100 million claims in the Truven MarketScan Research databases.

Survival gains seen with rituximab and mycophenolate in RA lung disease
LIVERPOOL, ENGLAND – Rituximab and mycophenolate mofetil could offer patients with rheumatoid arthritis and concomitant interstitial lung disease a survival advantage over other therapies, according to findings from a large, retrospective, multicenter study.
Data collated by the British Rheumatoid Interstitial Lung (BRILL) network found all-cause mortality in patients with rheumatoid arthritis (RA) and interstitial lung disease (ILD) treated with rituximab was 8% versus 31% for those treated with anti-tumor necrosis factor (TNF) alpha therapies (P = .03). Death from respiratory causes was also significantly lower in the rituximab-treated patients (4% vs. 15%, P = .04).
All-cause mortality was also significantly lower in patients with RA and ILD treated with mycophenolate mofetil (relative risk, 0.65) when compared with azathioprine (RR, 1.42) or other immunosuppressants, which included cyclophosphamide (RR, 1.65) and tacrolimus (RR, 1.74).
However, these data were retrospective, so prospective therapeutic trials are now needed, said Dr. Clive Kelly of Queen Elizabeth Hospital, Gateshead, England. Dr. Kelly, who was the driving force behind the set up of the BRILL network 2 years ago, stated that plans were already afoot to set up these trials with the intention of recruiting 200 patients with RA and ILD across the United Kingdom.
The aim of future studies would be to compare the use of rituximab versus anti-TNF therapies, with or without methotrexate, and mycophenolate mofetil versus azathioprine, with or without oral steroids, to see if the anticipated survival benefit associated with the newer agents is confirmed. Funding for the trials is yet to be secured, but it is hoped that recruitment can begin next year.
When people think of comorbidity in RA, they tend to think about cardiovascular disease first, Dr. Kelly noted at the British Society for Rheumatology annual conference. He observed that ILD has been associated with RA for well over 50 years and that respiratory disease, largely due to ILD and chronic lung damage, is now a "close second" behind heart disease as a cause of death in patients with RA.
Up to 40% of all cases of ILD are discovered in RA patients postmortem, he said, but with the increased use of high-resolution computed tomography (HRCT) in the 1990s, around a quarter of patients with RA were found to have changes on lung scanning.
Today, the prevalence of clinically significant lung disease is estimated to be around 5% in RA, with a lifetime risk thought to be around 7.7%. Patients with RA who develop ILD are around three times more likely to die than are those who do not, with a reported mean survival around 2.6 years from the diagnosis of lung disease until the advent of these recent studies.
The BRILL network consists of 16 centers located throughout the United Kingdom and was set up to establish a national database of all cases of ILD in RA patients that had been reported by these centers over a 25-year period, beginning in 1988. Patients were included in the database if clinical symptoms, such as shortness of breath or chest crackles, or pulmonary function tests had indicated ILD, and abnormal findings had been confirmed via HRCT.
For the current study, Dr. Kelly and coinvestigators from the BRILL Network looked at a subset of 188 patients with RA and ILD who had been diagnosed between 2000 and 2012. A control group of 188 patients with RA without ILD were included for comparison, matched for age, sex, and duration of RA.
Patients with ILD were more likely than those without to use biologics or immunosuppressants. Of the 188 patients with ILD, 57 were treated with either rituximab or anti-TNF drugs and 83 were treated with immunosuppressants.
The median age at which ILD was identified was 64 years, ranging from 42 to 87 years. The median duration of RA at ILD diagnosis was 9 years and the median duration of ILD was 4 years.
Dr. Kelly reported that RA preceded lung disease in the majority (83%) of cases, while 10% of patients developed ILD before the articular features of their disease became apparent. The onset of RA and ILD was "synchronous" in the remaining 7%.
HRCT results showed that the most common subtype of lung disease was interstitial pneumonia in 65% of cases, which carries the worst overall prognosis, he said. Nonspecific interstitial pneumonia, which is more responsive to early therapy, accounted for 24% of cases. The remaining patients had cryptogenic organizing pneumonia (6%) and or mixed subtypes (5%).
Survival over the 25 years improved, with 67% of RA-ILD patients dying from ILD in 1987-1993, compared with 30% in 2006-2012 (P less than .01). Nevertheless, these data are retrospective, Dr. Kelly observed, and trials are warranted to confirm the effects of specific treatments. He encouraged delegates at the meeting to participate in forthcoming BRILL network studies.
Dr. Kelly had no disclosures.
LIVERPOOL, ENGLAND – Rituximab and mycophenolate mofetil could offer patients with rheumatoid arthritis and concomitant interstitial lung disease a survival advantage over other therapies, according to findings from a large, retrospective, multicenter study.
Data collated by the British Rheumatoid Interstitial Lung (BRILL) network found all-cause mortality in patients with rheumatoid arthritis (RA) and interstitial lung disease (ILD) treated with rituximab was 8% versus 31% for those treated with anti-tumor necrosis factor (TNF) alpha therapies (P = .03). Death from respiratory causes was also significantly lower in the rituximab-treated patients (4% vs. 15%, P = .04).
All-cause mortality was also significantly lower in patients with RA and ILD treated with mycophenolate mofetil (relative risk, 0.65) when compared with azathioprine (RR, 1.42) or other immunosuppressants, which included cyclophosphamide (RR, 1.65) and tacrolimus (RR, 1.74).
However, these data were retrospective, so prospective therapeutic trials are now needed, said Dr. Clive Kelly of Queen Elizabeth Hospital, Gateshead, England. Dr. Kelly, who was the driving force behind the set up of the BRILL network 2 years ago, stated that plans were already afoot to set up these trials with the intention of recruiting 200 patients with RA and ILD across the United Kingdom.
The aim of future studies would be to compare the use of rituximab versus anti-TNF therapies, with or without methotrexate, and mycophenolate mofetil versus azathioprine, with or without oral steroids, to see if the anticipated survival benefit associated with the newer agents is confirmed. Funding for the trials is yet to be secured, but it is hoped that recruitment can begin next year.
When people think of comorbidity in RA, they tend to think about cardiovascular disease first, Dr. Kelly noted at the British Society for Rheumatology annual conference. He observed that ILD has been associated with RA for well over 50 years and that respiratory disease, largely due to ILD and chronic lung damage, is now a "close second" behind heart disease as a cause of death in patients with RA.
Up to 40% of all cases of ILD are discovered in RA patients postmortem, he said, but with the increased use of high-resolution computed tomography (HRCT) in the 1990s, around a quarter of patients with RA were found to have changes on lung scanning.
Today, the prevalence of clinically significant lung disease is estimated to be around 5% in RA, with a lifetime risk thought to be around 7.7%. Patients with RA who develop ILD are around three times more likely to die than are those who do not, with a reported mean survival around 2.6 years from the diagnosis of lung disease until the advent of these recent studies.
The BRILL network consists of 16 centers located throughout the United Kingdom and was set up to establish a national database of all cases of ILD in RA patients that had been reported by these centers over a 25-year period, beginning in 1988. Patients were included in the database if clinical symptoms, such as shortness of breath or chest crackles, or pulmonary function tests had indicated ILD, and abnormal findings had been confirmed via HRCT.
For the current study, Dr. Kelly and coinvestigators from the BRILL Network looked at a subset of 188 patients with RA and ILD who had been diagnosed between 2000 and 2012. A control group of 188 patients with RA without ILD were included for comparison, matched for age, sex, and duration of RA.
Patients with ILD were more likely than those without to use biologics or immunosuppressants. Of the 188 patients with ILD, 57 were treated with either rituximab or anti-TNF drugs and 83 were treated with immunosuppressants.
The median age at which ILD was identified was 64 years, ranging from 42 to 87 years. The median duration of RA at ILD diagnosis was 9 years and the median duration of ILD was 4 years.
Dr. Kelly reported that RA preceded lung disease in the majority (83%) of cases, while 10% of patients developed ILD before the articular features of their disease became apparent. The onset of RA and ILD was "synchronous" in the remaining 7%.
HRCT results showed that the most common subtype of lung disease was interstitial pneumonia in 65% of cases, which carries the worst overall prognosis, he said. Nonspecific interstitial pneumonia, which is more responsive to early therapy, accounted for 24% of cases. The remaining patients had cryptogenic organizing pneumonia (6%) and or mixed subtypes (5%).
Survival over the 25 years improved, with 67% of RA-ILD patients dying from ILD in 1987-1993, compared with 30% in 2006-2012 (P less than .01). Nevertheless, these data are retrospective, Dr. Kelly observed, and trials are warranted to confirm the effects of specific treatments. He encouraged delegates at the meeting to participate in forthcoming BRILL network studies.
Dr. Kelly had no disclosures.
LIVERPOOL, ENGLAND – Rituximab and mycophenolate mofetil could offer patients with rheumatoid arthritis and concomitant interstitial lung disease a survival advantage over other therapies, according to findings from a large, retrospective, multicenter study.
Data collated by the British Rheumatoid Interstitial Lung (BRILL) network found all-cause mortality in patients with rheumatoid arthritis (RA) and interstitial lung disease (ILD) treated with rituximab was 8% versus 31% for those treated with anti-tumor necrosis factor (TNF) alpha therapies (P = .03). Death from respiratory causes was also significantly lower in the rituximab-treated patients (4% vs. 15%, P = .04).
All-cause mortality was also significantly lower in patients with RA and ILD treated with mycophenolate mofetil (relative risk, 0.65) when compared with azathioprine (RR, 1.42) or other immunosuppressants, which included cyclophosphamide (RR, 1.65) and tacrolimus (RR, 1.74).
However, these data were retrospective, so prospective therapeutic trials are now needed, said Dr. Clive Kelly of Queen Elizabeth Hospital, Gateshead, England. Dr. Kelly, who was the driving force behind the set up of the BRILL network 2 years ago, stated that plans were already afoot to set up these trials with the intention of recruiting 200 patients with RA and ILD across the United Kingdom.
The aim of future studies would be to compare the use of rituximab versus anti-TNF therapies, with or without methotrexate, and mycophenolate mofetil versus azathioprine, with or without oral steroids, to see if the anticipated survival benefit associated with the newer agents is confirmed. Funding for the trials is yet to be secured, but it is hoped that recruitment can begin next year.
When people think of comorbidity in RA, they tend to think about cardiovascular disease first, Dr. Kelly noted at the British Society for Rheumatology annual conference. He observed that ILD has been associated with RA for well over 50 years and that respiratory disease, largely due to ILD and chronic lung damage, is now a "close second" behind heart disease as a cause of death in patients with RA.
Up to 40% of all cases of ILD are discovered in RA patients postmortem, he said, but with the increased use of high-resolution computed tomography (HRCT) in the 1990s, around a quarter of patients with RA were found to have changes on lung scanning.
Today, the prevalence of clinically significant lung disease is estimated to be around 5% in RA, with a lifetime risk thought to be around 7.7%. Patients with RA who develop ILD are around three times more likely to die than are those who do not, with a reported mean survival around 2.6 years from the diagnosis of lung disease until the advent of these recent studies.
The BRILL network consists of 16 centers located throughout the United Kingdom and was set up to establish a national database of all cases of ILD in RA patients that had been reported by these centers over a 25-year period, beginning in 1988. Patients were included in the database if clinical symptoms, such as shortness of breath or chest crackles, or pulmonary function tests had indicated ILD, and abnormal findings had been confirmed via HRCT.
For the current study, Dr. Kelly and coinvestigators from the BRILL Network looked at a subset of 188 patients with RA and ILD who had been diagnosed between 2000 and 2012. A control group of 188 patients with RA without ILD were included for comparison, matched for age, sex, and duration of RA.
Patients with ILD were more likely than those without to use biologics or immunosuppressants. Of the 188 patients with ILD, 57 were treated with either rituximab or anti-TNF drugs and 83 were treated with immunosuppressants.
The median age at which ILD was identified was 64 years, ranging from 42 to 87 years. The median duration of RA at ILD diagnosis was 9 years and the median duration of ILD was 4 years.
Dr. Kelly reported that RA preceded lung disease in the majority (83%) of cases, while 10% of patients developed ILD before the articular features of their disease became apparent. The onset of RA and ILD was "synchronous" in the remaining 7%.
HRCT results showed that the most common subtype of lung disease was interstitial pneumonia in 65% of cases, which carries the worst overall prognosis, he said. Nonspecific interstitial pneumonia, which is more responsive to early therapy, accounted for 24% of cases. The remaining patients had cryptogenic organizing pneumonia (6%) and or mixed subtypes (5%).
Survival over the 25 years improved, with 67% of RA-ILD patients dying from ILD in 1987-1993, compared with 30% in 2006-2012 (P less than .01). Nevertheless, these data are retrospective, Dr. Kelly observed, and trials are warranted to confirm the effects of specific treatments. He encouraged delegates at the meeting to participate in forthcoming BRILL network studies.
Dr. Kelly had no disclosures.
AT RHEUMATOLOGY 2014
Key clinical point: Early, retrospective data indicate that in RA-ILD patients, rituximab may improve survival over anti-TNF treatments, while mycophenolate mofetil may reduce mortality when compared with other immunosuppressants.
Major finding: Treatment of RA-ILD with rituximab vs. anti-TNF agents led to all-cause mortality of 8% vs. 31% (P = .03), respectively, and respiratory mortality of 4% vs. 15% (P = .04). The relative risk for death from any cause was 0.65 for mycophenolate treatment, compared with other immunosuppressants.
Data source: Multicenter, retrospective study of 188 patients with rheumatoid arthritis (RA) and interstitial lung disease and 188 with RA and no lung disease as controls.
Disclosures: Dr. Kelly had no conflicts of interest.
Biologic therapy improves fatigue of rheumatoid arthritis
LIVERPOOL, ENGLAND – Anti–tumor necrosis factor treatment significantly decreased fatigue in patients with rheumatoid arthritis in an analysis of data from the British Society for Rheumatology Biologics Registers Rheumatoid Arthritis Register.
Of 6,835 patients included in the analysis, 38.8% reported high levels of fatigue at baseline, which was defined as a score of 12.5 or less on the vitality domain of the Short Form (SF)-36. Of these, 66% reported clinically relevant improvements of 10 U or more on the SF-36 vitality domain 6 months after starting anti–tumor necrosis factor (TNF) treatment.
"Randomized controlled trials have shown modest improvements in fatigue with anti-TNF therapy, but fatigue has not been the primary research question in these trials, and so that led us to ask what the effect would be in patients who had clinically relevant symptoms," said Katie Druce, who performed the research as part of her postgraduate studies at the University of Aberdeen (Scotland).
Data on 13,122 patients with RA who started treatment with etanercept, adalimumab, or infliximab between 2000 and 2008 were initially considered, but only those who had data on fatigue available at baseline and at 6-month assessments were included in the analysis.
At baseline, the mean age of participants was 57 years, and most (77%) were female and white (97%). A total of 31% were not working because of illness. The patients had mean scores of 6.57 on the 28-joint Disease Activity Score, 21.1 on the bodily pain and 24.8 on the vitality domains of the SF-36, and 2.03 on the mean Health Assessment Questionnaire Disability Index.
Fatigue improved at 6 months
Ms. Druce reported at the British Society for Rheumatology (BSR) annual conference that there was an absolute improvement in fatigue at 6 months of 22.5 U in the 2,652 patients who had reported a high level of fatigue at baseline. This exceeded the value of 10 U or more that was conservatively set as the minimum clinically important difference (MCID). Furthermore, in the patients who improved – that is going from a high level to a low level of fatigue – the difference was even greater, with improvement three times the MCID, at about 32 U.
Baseline predictors of improvement included being in good mental health, no history of hypertension or depression, and not being unemployed because of ill health. Other predictors included being female, rheumatoid factor positive, having a low level of disability, and not using steroids. Patients with multiple baseline predictors of improvement appeared to have the largest absolute improvement in fatigue at 6 months.
"Importantly, we saw no association with disease activity or measures of inflammation," Ms. Druce said, and the specific anti-TNF drug used was also not influential on the findings. She noted that baseline predictors of fatigue might be able to help stratify patients who need specific fatigue management.
Pain drives fatigue in RA
The improvements in RA-related fatigue seen with biologic therapy were driven by decreases in pain and not disease activity, Ms. Druce said, reporting the finding of a separate analysis elsewhere at the conference.
"Anecdotally, from both clinicians and patients, we know that they consider fatigue to be caused by disease activity, but when we look at the evidence from the literature, we see that there is an inconsistent link between fatigue and disease activity," she said. "Yet we know that fatigue improves after anti-TNF therapy commences. So this led us to look at which variables are driving change in fatigue that we see after anti-TNF therapies are commenced," she said.
Ms. Druce and her colleagues used structural equation modeling, which evaluates the effect of one variable on an outcome and the pathway by which it has that effect, to find that for every 1–standard deviation increase in change in disease activity, there was a 0.05 increase in change in fatigue. Although this does indicate some improvement, change in pain had a greater effect, with every 1–standard deviation increase in change in pain linked to a 0.31 increase in change in fatigue.
"What we found was that 82% of the effect of change in disease activity is mediated through other variables in [our] model," Ms. Druce explained. Indeed, it was found that almost two-thirds of the change in fatigue was mediated by pain (or pain and mental health), 27% through disability (or disability and mental health), and 9% directly through mental health.
"This led us to conclude that improvements in fatigue after commencing anti-TNF therapies are driven by improvements in pain rather than directly from disease activity, but that mental health and disability are also important, and, along with pain, represent potentially modifiable factors for us to target in routine treatment," Ms. Druce said.
How can fatigue be assessed?
Fatigue is an important facet of RA that should be included in patient assessment, said Sara Hewlett, Ph.D., professor of nursing at the University of the West of England in Bristol.
Speaking during a session organized by the BSR’s Specialist Interest Group in RA, she noted that there are three reasons why fatigue is important to assess: It is important to patients, it provides information beyond disease activity, and it responds to several interventions, including physical activity, psychological strategies, and biologic medications.
Dr. Hewlett noted that there have been many instruments used to try to assess fatigue in clinical trials, but none was specifically developed to assess fatigue in patients with RA, and many were not developed using recommended methodology. That was until the development and validation of the Bristol Rheumatoid Arthritis Fatigue Numerical Rating Scales (BRAF NRS).
On the BRAF NRS, patients circle a number from 0 to 10 to indicate the average level of fatigue that they have experienced, the impact that fatigue has had on them, and how well they have subsequently coped, in the past 7 days.
Ms. Druce said she had no financial disclosures. One of her coauthors had received honoraria and research funding from Pfizer and had been a consultant for Merck Sharp & Dohme. Ms. Hewlett said she had no disclosures other than helping to develop the BRAF NRS.
LIVERPOOL, ENGLAND – Anti–tumor necrosis factor treatment significantly decreased fatigue in patients with rheumatoid arthritis in an analysis of data from the British Society for Rheumatology Biologics Registers Rheumatoid Arthritis Register.
Of 6,835 patients included in the analysis, 38.8% reported high levels of fatigue at baseline, which was defined as a score of 12.5 or less on the vitality domain of the Short Form (SF)-36. Of these, 66% reported clinically relevant improvements of 10 U or more on the SF-36 vitality domain 6 months after starting anti–tumor necrosis factor (TNF) treatment.
"Randomized controlled trials have shown modest improvements in fatigue with anti-TNF therapy, but fatigue has not been the primary research question in these trials, and so that led us to ask what the effect would be in patients who had clinically relevant symptoms," said Katie Druce, who performed the research as part of her postgraduate studies at the University of Aberdeen (Scotland).
Data on 13,122 patients with RA who started treatment with etanercept, adalimumab, or infliximab between 2000 and 2008 were initially considered, but only those who had data on fatigue available at baseline and at 6-month assessments were included in the analysis.
At baseline, the mean age of participants was 57 years, and most (77%) were female and white (97%). A total of 31% were not working because of illness. The patients had mean scores of 6.57 on the 28-joint Disease Activity Score, 21.1 on the bodily pain and 24.8 on the vitality domains of the SF-36, and 2.03 on the mean Health Assessment Questionnaire Disability Index.
Fatigue improved at 6 months
Ms. Druce reported at the British Society for Rheumatology (BSR) annual conference that there was an absolute improvement in fatigue at 6 months of 22.5 U in the 2,652 patients who had reported a high level of fatigue at baseline. This exceeded the value of 10 U or more that was conservatively set as the minimum clinically important difference (MCID). Furthermore, in the patients who improved – that is going from a high level to a low level of fatigue – the difference was even greater, with improvement three times the MCID, at about 32 U.
Baseline predictors of improvement included being in good mental health, no history of hypertension or depression, and not being unemployed because of ill health. Other predictors included being female, rheumatoid factor positive, having a low level of disability, and not using steroids. Patients with multiple baseline predictors of improvement appeared to have the largest absolute improvement in fatigue at 6 months.
"Importantly, we saw no association with disease activity or measures of inflammation," Ms. Druce said, and the specific anti-TNF drug used was also not influential on the findings. She noted that baseline predictors of fatigue might be able to help stratify patients who need specific fatigue management.
Pain drives fatigue in RA
The improvements in RA-related fatigue seen with biologic therapy were driven by decreases in pain and not disease activity, Ms. Druce said, reporting the finding of a separate analysis elsewhere at the conference.
"Anecdotally, from both clinicians and patients, we know that they consider fatigue to be caused by disease activity, but when we look at the evidence from the literature, we see that there is an inconsistent link between fatigue and disease activity," she said. "Yet we know that fatigue improves after anti-TNF therapy commences. So this led us to look at which variables are driving change in fatigue that we see after anti-TNF therapies are commenced," she said.
Ms. Druce and her colleagues used structural equation modeling, which evaluates the effect of one variable on an outcome and the pathway by which it has that effect, to find that for every 1–standard deviation increase in change in disease activity, there was a 0.05 increase in change in fatigue. Although this does indicate some improvement, change in pain had a greater effect, with every 1–standard deviation increase in change in pain linked to a 0.31 increase in change in fatigue.
"What we found was that 82% of the effect of change in disease activity is mediated through other variables in [our] model," Ms. Druce explained. Indeed, it was found that almost two-thirds of the change in fatigue was mediated by pain (or pain and mental health), 27% through disability (or disability and mental health), and 9% directly through mental health.
"This led us to conclude that improvements in fatigue after commencing anti-TNF therapies are driven by improvements in pain rather than directly from disease activity, but that mental health and disability are also important, and, along with pain, represent potentially modifiable factors for us to target in routine treatment," Ms. Druce said.
How can fatigue be assessed?
Fatigue is an important facet of RA that should be included in patient assessment, said Sara Hewlett, Ph.D., professor of nursing at the University of the West of England in Bristol.
Speaking during a session organized by the BSR’s Specialist Interest Group in RA, she noted that there are three reasons why fatigue is important to assess: It is important to patients, it provides information beyond disease activity, and it responds to several interventions, including physical activity, psychological strategies, and biologic medications.
Dr. Hewlett noted that there have been many instruments used to try to assess fatigue in clinical trials, but none was specifically developed to assess fatigue in patients with RA, and many were not developed using recommended methodology. That was until the development and validation of the Bristol Rheumatoid Arthritis Fatigue Numerical Rating Scales (BRAF NRS).
On the BRAF NRS, patients circle a number from 0 to 10 to indicate the average level of fatigue that they have experienced, the impact that fatigue has had on them, and how well they have subsequently coped, in the past 7 days.
Ms. Druce said she had no financial disclosures. One of her coauthors had received honoraria and research funding from Pfizer and had been a consultant for Merck Sharp & Dohme. Ms. Hewlett said she had no disclosures other than helping to develop the BRAF NRS.
LIVERPOOL, ENGLAND – Anti–tumor necrosis factor treatment significantly decreased fatigue in patients with rheumatoid arthritis in an analysis of data from the British Society for Rheumatology Biologics Registers Rheumatoid Arthritis Register.
Of 6,835 patients included in the analysis, 38.8% reported high levels of fatigue at baseline, which was defined as a score of 12.5 or less on the vitality domain of the Short Form (SF)-36. Of these, 66% reported clinically relevant improvements of 10 U or more on the SF-36 vitality domain 6 months after starting anti–tumor necrosis factor (TNF) treatment.
"Randomized controlled trials have shown modest improvements in fatigue with anti-TNF therapy, but fatigue has not been the primary research question in these trials, and so that led us to ask what the effect would be in patients who had clinically relevant symptoms," said Katie Druce, who performed the research as part of her postgraduate studies at the University of Aberdeen (Scotland).
Data on 13,122 patients with RA who started treatment with etanercept, adalimumab, or infliximab between 2000 and 2008 were initially considered, but only those who had data on fatigue available at baseline and at 6-month assessments were included in the analysis.
At baseline, the mean age of participants was 57 years, and most (77%) were female and white (97%). A total of 31% were not working because of illness. The patients had mean scores of 6.57 on the 28-joint Disease Activity Score, 21.1 on the bodily pain and 24.8 on the vitality domains of the SF-36, and 2.03 on the mean Health Assessment Questionnaire Disability Index.
Fatigue improved at 6 months
Ms. Druce reported at the British Society for Rheumatology (BSR) annual conference that there was an absolute improvement in fatigue at 6 months of 22.5 U in the 2,652 patients who had reported a high level of fatigue at baseline. This exceeded the value of 10 U or more that was conservatively set as the minimum clinically important difference (MCID). Furthermore, in the patients who improved – that is going from a high level to a low level of fatigue – the difference was even greater, with improvement three times the MCID, at about 32 U.
Baseline predictors of improvement included being in good mental health, no history of hypertension or depression, and not being unemployed because of ill health. Other predictors included being female, rheumatoid factor positive, having a low level of disability, and not using steroids. Patients with multiple baseline predictors of improvement appeared to have the largest absolute improvement in fatigue at 6 months.
"Importantly, we saw no association with disease activity or measures of inflammation," Ms. Druce said, and the specific anti-TNF drug used was also not influential on the findings. She noted that baseline predictors of fatigue might be able to help stratify patients who need specific fatigue management.
Pain drives fatigue in RA
The improvements in RA-related fatigue seen with biologic therapy were driven by decreases in pain and not disease activity, Ms. Druce said, reporting the finding of a separate analysis elsewhere at the conference.
"Anecdotally, from both clinicians and patients, we know that they consider fatigue to be caused by disease activity, but when we look at the evidence from the literature, we see that there is an inconsistent link between fatigue and disease activity," she said. "Yet we know that fatigue improves after anti-TNF therapy commences. So this led us to look at which variables are driving change in fatigue that we see after anti-TNF therapies are commenced," she said.
Ms. Druce and her colleagues used structural equation modeling, which evaluates the effect of one variable on an outcome and the pathway by which it has that effect, to find that for every 1–standard deviation increase in change in disease activity, there was a 0.05 increase in change in fatigue. Although this does indicate some improvement, change in pain had a greater effect, with every 1–standard deviation increase in change in pain linked to a 0.31 increase in change in fatigue.
"What we found was that 82% of the effect of change in disease activity is mediated through other variables in [our] model," Ms. Druce explained. Indeed, it was found that almost two-thirds of the change in fatigue was mediated by pain (or pain and mental health), 27% through disability (or disability and mental health), and 9% directly through mental health.
"This led us to conclude that improvements in fatigue after commencing anti-TNF therapies are driven by improvements in pain rather than directly from disease activity, but that mental health and disability are also important, and, along with pain, represent potentially modifiable factors for us to target in routine treatment," Ms. Druce said.
How can fatigue be assessed?
Fatigue is an important facet of RA that should be included in patient assessment, said Sara Hewlett, Ph.D., professor of nursing at the University of the West of England in Bristol.
Speaking during a session organized by the BSR’s Specialist Interest Group in RA, she noted that there are three reasons why fatigue is important to assess: It is important to patients, it provides information beyond disease activity, and it responds to several interventions, including physical activity, psychological strategies, and biologic medications.
Dr. Hewlett noted that there have been many instruments used to try to assess fatigue in clinical trials, but none was specifically developed to assess fatigue in patients with RA, and many were not developed using recommended methodology. That was until the development and validation of the Bristol Rheumatoid Arthritis Fatigue Numerical Rating Scales (BRAF NRS).
On the BRAF NRS, patients circle a number from 0 to 10 to indicate the average level of fatigue that they have experienced, the impact that fatigue has had on them, and how well they have subsequently coped, in the past 7 days.
Ms. Druce said she had no financial disclosures. One of her coauthors had received honoraria and research funding from Pfizer and had been a consultant for Merck Sharp & Dohme. Ms. Hewlett said she had no disclosures other than helping to develop the BRAF NRS.
AT RHEUMATOLOGY 2014
Key clinical point: Changes in fatigue levels after 6 months of anti-TNF treatment were associated most strongly with changes in pain, not disease activity.
Major finding: Of patients reporting high fatigue at baseline, 66% had improvement with 6 months of anti-TNF therapy.
Data source: British Society for Rheumatology Biologics Registers RA Register data on more than 6,000 patients treated with anti-TNF drugs and who reported fatigue at baseline and at 6 months.
Disclosures: Ms. Druce said she had no disclosures. One of her coauthors had received honoraria and research funding from Pfizer and been a consultant for Merck Sharp & Dohme. Ms Hewlett said she had no disclosures other than helping to develop the BRAF NRS.
Biologic agents do not up recurrent cancer risk in RA
LIVERPOOL, ENGLAND – The risk of recurrent cancer in patients with rheumatoid arthritis did not increase with the use of biologic therapies, according to data just released from the British Society for Rheumatology Biologics Registers’ Rheumatoid Arthritis Register.
In fact, the risk of repeated cancer in patients with prior malignancies treated with biologic therapies was apparently decreased by around 50%, when compared with treatment with nonbiologic disease-modifying antirheumatic drugs (nbDMARDs).
The hazard ratios, adjusted for age and sex, for recurrent cancer were 0.55 for patients treated with drugs directed at tumor necrosis factor (TNF)-alpha and 0.47 for patients treated, off-label, with rituximab vs. nbDMARDs.
However, Dr. Luca Silva-Fernandez, who presented the findings at the British Society for Rheumatology annual conference, noted that patients treated with nbDMARDs were perhaps at higher risk of recurrent disease than were those who were treated with biologic agents at the start of their treatment, so the data do not imply that biologic agents are less likely to cause recurrent disease than do the older RA therapies.
"Our data suggest that patients with RA and prior malignancy selected to receive either an anti-TNF or rituximab therapy in the U.K. do not seem to have an increased risk of future incident malignancy," said Dr. Silva-Fernandez.
Patients treated with anti-TNFs or rituximab were more likely to have incident cancers, added Dr. Silva-Fernandez of the Arthritis Research UK Epidemiology Unit at the University of Manchester, England, where the BSRBR-RA Register is run.
Previous data on the use of biologic therapies in patients with prior cancer were published from the British Society for Rheumatology Biologics Registers (BSRBR) 4 years ago (Arthritis Care Res. 2010;62:755-63). These considered 293 patients with a prior malignancy identified from over 14,000 patients with RA enrolled in the register at the time. The present analysis included 425 patients who had cancer before being enrolled in the BSRBR-RA Register, which at the time of the updated analysis included around 19,000 patients.
In total, 425 new malignancies were detected in 159 of 3,787 (1.7%) patients who had been treated with an nbDMARD, 243 of 14,168 (8.9%) patients treated with an anti-TNF drug, and 23 of 257 (4.2%) patients who had received rituximab for the treatment of their RA.
There were several differences in baseline characteristics among the three groups. The mean age of patients in each treatment arm was 66.1, 62.7, and 67.3 years, respectively. A higher percentage (81%) of anti-TNF-treated patients were female when compared with the nbDMARD (74%) and rituximab (65%) arms. Biologic-treated patients tended to have a longer mean RA disease duration (anti-TNF 12 years and rituximab 14 years) than did the nbDMARD-treated patients (8 years), and higher disease severity scores at enrollment into the BSRBR-RA Register. The median time between the previous and current malignancy was 7.9 years for nbDMARDs, 11.5 years for anti-TNFs, and 5.4 years for rituximab.
The crude incident malignancy rates were 47/1,000 person-years in the nbDMARD cohort, 24/1,000 person-years in the anti-TNF-treated patients, and 25/1,000 person-years in the group treated with rituximab. Dr. Silva-Fernandez reported that the median follow-up was much shorter in rituximab-treated patients, at 3.9 years, than both the nbDMARD- (6.6 years) and anti-TNF–treated (6.9 years) groups.
After her presentation, Dr. Silva-Fernandez was asked to comment on the apparent "protective" effect of anti-TNFs on the recurrence of cancer. She replied that the nbDMARD and biologic groups were not really comparable, referring back to the differences in baseline characteristics, so such an association cannot be claimed.
With regards to a question on the use of rituximab in RA patients with a history of prior cancer, which was "off label" in this instance as it wasn’t used after anti-TNFs but "up-front," this might reflect a "channeling bias" on the part of the physicians, commented Dr. Kimmie Hyrich.
"These patients were those who had received rituximab as their first biologic for a number of reasons," said Dr. Hyrich, one of the principal investigators for the BSRBR-RA Register. "For many of them, it may have been a past cancer that made that decision," she added.
"I think, as physicians, we have a comfort in using rituximab in patients with past cancer, because it is not an absolute contraindication, so I think this is physician choice, and that’s probably why an off-license decision to treat with rituximab was made in these patients," said Dr. Hyrich, also from the University of Manchester, England.
Although patients with nonmelanoma skin cancer (NSMC) were excluded from the present analysis, Dr. Hyrich also noted in response to a question that the BSRBR RA Register team had previously reported on the rates of recurrence in patients with and without a prior history of this type of skin cancer.
Findings had shown that anti-TNFs did not appear to increase the risk of NSMC in patients without a prior history of skin cancer. In patients with a history of the disease, there was elevated risk of recurrence, regardless of whether patients received nbDMARDs or anti-TNFs, and it did not seem to occur more in the biologic-treated patients.
"I think the most striking finding, however, was regardless of treatment, all patients in the register, overall, have a marked increased risk of skin cancer, compared with general population, so I think RA itself and its treatment is probably the strongest risk factor," Dr. Hyrich said.
The BSRBR is funded by a grant from the British Society for Rheumatology (BSR), which receives funding from multiple drug companies. This income finances a separate contract between the BSR and the University of Manchester that provides and runs the BSRBR RA Register. Dr. Silva-Fernandez and Dr. Hyrich had no personal conflicts of interest.
LIVERPOOL, ENGLAND – The risk of recurrent cancer in patients with rheumatoid arthritis did not increase with the use of biologic therapies, according to data just released from the British Society for Rheumatology Biologics Registers’ Rheumatoid Arthritis Register.
In fact, the risk of repeated cancer in patients with prior malignancies treated with biologic therapies was apparently decreased by around 50%, when compared with treatment with nonbiologic disease-modifying antirheumatic drugs (nbDMARDs).
The hazard ratios, adjusted for age and sex, for recurrent cancer were 0.55 for patients treated with drugs directed at tumor necrosis factor (TNF)-alpha and 0.47 for patients treated, off-label, with rituximab vs. nbDMARDs.
However, Dr. Luca Silva-Fernandez, who presented the findings at the British Society for Rheumatology annual conference, noted that patients treated with nbDMARDs were perhaps at higher risk of recurrent disease than were those who were treated with biologic agents at the start of their treatment, so the data do not imply that biologic agents are less likely to cause recurrent disease than do the older RA therapies.
"Our data suggest that patients with RA and prior malignancy selected to receive either an anti-TNF or rituximab therapy in the U.K. do not seem to have an increased risk of future incident malignancy," said Dr. Silva-Fernandez.
Patients treated with anti-TNFs or rituximab were more likely to have incident cancers, added Dr. Silva-Fernandez of the Arthritis Research UK Epidemiology Unit at the University of Manchester, England, where the BSRBR-RA Register is run.
Previous data on the use of biologic therapies in patients with prior cancer were published from the British Society for Rheumatology Biologics Registers (BSRBR) 4 years ago (Arthritis Care Res. 2010;62:755-63). These considered 293 patients with a prior malignancy identified from over 14,000 patients with RA enrolled in the register at the time. The present analysis included 425 patients who had cancer before being enrolled in the BSRBR-RA Register, which at the time of the updated analysis included around 19,000 patients.
In total, 425 new malignancies were detected in 159 of 3,787 (1.7%) patients who had been treated with an nbDMARD, 243 of 14,168 (8.9%) patients treated with an anti-TNF drug, and 23 of 257 (4.2%) patients who had received rituximab for the treatment of their RA.
There were several differences in baseline characteristics among the three groups. The mean age of patients in each treatment arm was 66.1, 62.7, and 67.3 years, respectively. A higher percentage (81%) of anti-TNF-treated patients were female when compared with the nbDMARD (74%) and rituximab (65%) arms. Biologic-treated patients tended to have a longer mean RA disease duration (anti-TNF 12 years and rituximab 14 years) than did the nbDMARD-treated patients (8 years), and higher disease severity scores at enrollment into the BSRBR-RA Register. The median time between the previous and current malignancy was 7.9 years for nbDMARDs, 11.5 years for anti-TNFs, and 5.4 years for rituximab.
The crude incident malignancy rates were 47/1,000 person-years in the nbDMARD cohort, 24/1,000 person-years in the anti-TNF-treated patients, and 25/1,000 person-years in the group treated with rituximab. Dr. Silva-Fernandez reported that the median follow-up was much shorter in rituximab-treated patients, at 3.9 years, than both the nbDMARD- (6.6 years) and anti-TNF–treated (6.9 years) groups.
After her presentation, Dr. Silva-Fernandez was asked to comment on the apparent "protective" effect of anti-TNFs on the recurrence of cancer. She replied that the nbDMARD and biologic groups were not really comparable, referring back to the differences in baseline characteristics, so such an association cannot be claimed.
With regards to a question on the use of rituximab in RA patients with a history of prior cancer, which was "off label" in this instance as it wasn’t used after anti-TNFs but "up-front," this might reflect a "channeling bias" on the part of the physicians, commented Dr. Kimmie Hyrich.
"These patients were those who had received rituximab as their first biologic for a number of reasons," said Dr. Hyrich, one of the principal investigators for the BSRBR-RA Register. "For many of them, it may have been a past cancer that made that decision," she added.
"I think, as physicians, we have a comfort in using rituximab in patients with past cancer, because it is not an absolute contraindication, so I think this is physician choice, and that’s probably why an off-license decision to treat with rituximab was made in these patients," said Dr. Hyrich, also from the University of Manchester, England.
Although patients with nonmelanoma skin cancer (NSMC) were excluded from the present analysis, Dr. Hyrich also noted in response to a question that the BSRBR RA Register team had previously reported on the rates of recurrence in patients with and without a prior history of this type of skin cancer.
Findings had shown that anti-TNFs did not appear to increase the risk of NSMC in patients without a prior history of skin cancer. In patients with a history of the disease, there was elevated risk of recurrence, regardless of whether patients received nbDMARDs or anti-TNFs, and it did not seem to occur more in the biologic-treated patients.
"I think the most striking finding, however, was regardless of treatment, all patients in the register, overall, have a marked increased risk of skin cancer, compared with general population, so I think RA itself and its treatment is probably the strongest risk factor," Dr. Hyrich said.
The BSRBR is funded by a grant from the British Society for Rheumatology (BSR), which receives funding from multiple drug companies. This income finances a separate contract between the BSR and the University of Manchester that provides and runs the BSRBR RA Register. Dr. Silva-Fernandez and Dr. Hyrich had no personal conflicts of interest.
LIVERPOOL, ENGLAND – The risk of recurrent cancer in patients with rheumatoid arthritis did not increase with the use of biologic therapies, according to data just released from the British Society for Rheumatology Biologics Registers’ Rheumatoid Arthritis Register.
In fact, the risk of repeated cancer in patients with prior malignancies treated with biologic therapies was apparently decreased by around 50%, when compared with treatment with nonbiologic disease-modifying antirheumatic drugs (nbDMARDs).
The hazard ratios, adjusted for age and sex, for recurrent cancer were 0.55 for patients treated with drugs directed at tumor necrosis factor (TNF)-alpha and 0.47 for patients treated, off-label, with rituximab vs. nbDMARDs.
However, Dr. Luca Silva-Fernandez, who presented the findings at the British Society for Rheumatology annual conference, noted that patients treated with nbDMARDs were perhaps at higher risk of recurrent disease than were those who were treated with biologic agents at the start of their treatment, so the data do not imply that biologic agents are less likely to cause recurrent disease than do the older RA therapies.
"Our data suggest that patients with RA and prior malignancy selected to receive either an anti-TNF or rituximab therapy in the U.K. do not seem to have an increased risk of future incident malignancy," said Dr. Silva-Fernandez.
Patients treated with anti-TNFs or rituximab were more likely to have incident cancers, added Dr. Silva-Fernandez of the Arthritis Research UK Epidemiology Unit at the University of Manchester, England, where the BSRBR-RA Register is run.
Previous data on the use of biologic therapies in patients with prior cancer were published from the British Society for Rheumatology Biologics Registers (BSRBR) 4 years ago (Arthritis Care Res. 2010;62:755-63). These considered 293 patients with a prior malignancy identified from over 14,000 patients with RA enrolled in the register at the time. The present analysis included 425 patients who had cancer before being enrolled in the BSRBR-RA Register, which at the time of the updated analysis included around 19,000 patients.
In total, 425 new malignancies were detected in 159 of 3,787 (1.7%) patients who had been treated with an nbDMARD, 243 of 14,168 (8.9%) patients treated with an anti-TNF drug, and 23 of 257 (4.2%) patients who had received rituximab for the treatment of their RA.
There were several differences in baseline characteristics among the three groups. The mean age of patients in each treatment arm was 66.1, 62.7, and 67.3 years, respectively. A higher percentage (81%) of anti-TNF-treated patients were female when compared with the nbDMARD (74%) and rituximab (65%) arms. Biologic-treated patients tended to have a longer mean RA disease duration (anti-TNF 12 years and rituximab 14 years) than did the nbDMARD-treated patients (8 years), and higher disease severity scores at enrollment into the BSRBR-RA Register. The median time between the previous and current malignancy was 7.9 years for nbDMARDs, 11.5 years for anti-TNFs, and 5.4 years for rituximab.
The crude incident malignancy rates were 47/1,000 person-years in the nbDMARD cohort, 24/1,000 person-years in the anti-TNF-treated patients, and 25/1,000 person-years in the group treated with rituximab. Dr. Silva-Fernandez reported that the median follow-up was much shorter in rituximab-treated patients, at 3.9 years, than both the nbDMARD- (6.6 years) and anti-TNF–treated (6.9 years) groups.
After her presentation, Dr. Silva-Fernandez was asked to comment on the apparent "protective" effect of anti-TNFs on the recurrence of cancer. She replied that the nbDMARD and biologic groups were not really comparable, referring back to the differences in baseline characteristics, so such an association cannot be claimed.
With regards to a question on the use of rituximab in RA patients with a history of prior cancer, which was "off label" in this instance as it wasn’t used after anti-TNFs but "up-front," this might reflect a "channeling bias" on the part of the physicians, commented Dr. Kimmie Hyrich.
"These patients were those who had received rituximab as their first biologic for a number of reasons," said Dr. Hyrich, one of the principal investigators for the BSRBR-RA Register. "For many of them, it may have been a past cancer that made that decision," she added.
"I think, as physicians, we have a comfort in using rituximab in patients with past cancer, because it is not an absolute contraindication, so I think this is physician choice, and that’s probably why an off-license decision to treat with rituximab was made in these patients," said Dr. Hyrich, also from the University of Manchester, England.
Although patients with nonmelanoma skin cancer (NSMC) were excluded from the present analysis, Dr. Hyrich also noted in response to a question that the BSRBR RA Register team had previously reported on the rates of recurrence in patients with and without a prior history of this type of skin cancer.
Findings had shown that anti-TNFs did not appear to increase the risk of NSMC in patients without a prior history of skin cancer. In patients with a history of the disease, there was elevated risk of recurrence, regardless of whether patients received nbDMARDs or anti-TNFs, and it did not seem to occur more in the biologic-treated patients.
"I think the most striking finding, however, was regardless of treatment, all patients in the register, overall, have a marked increased risk of skin cancer, compared with general population, so I think RA itself and its treatment is probably the strongest risk factor," Dr. Hyrich said.
The BSRBR is funded by a grant from the British Society for Rheumatology (BSR), which receives funding from multiple drug companies. This income finances a separate contract between the BSR and the University of Manchester that provides and runs the BSRBR RA Register. Dr. Silva-Fernandez and Dr. Hyrich had no personal conflicts of interest.
AT RHEUMATOLOGY 2014
Major finding: Age- and sex-adjusted hazard ratios for recurrent cancer vs. nbDMARDs were 0.55 for anti-TNFs and 0.47 for rituximab.
Data source: 425 patients with prior cancer who were enrolled in the BSRBR RA Register and treated with biologic or nonbiologic DMARDs.
Disclosures: The BSRBR is funded by a grant from the British Society for Rheumatology, which receives funding from multiple drug companies. This income finances a separate contract between the BSR and the University of Manchester that provides and runs the BSRBR RA Register. Dr. Silva-Fernandez and Dr. Hyrich had no personal conflicts of interest.
Anti-TNFs linked to lower heart attack risk in RA patients
LIVERPOOL, ENGLAND – A new analysis of data from the British Society for Rheumatology Biologics Registers’ Rheumatoid Arthritis Register confirms that anti–tumor necrosis factor treatment decreases patients’ risk for heart attack when compared with treatment with traditional, nonbiologic disease-modifying antirheumatic drugs, but a lower level of inflammation may not be the mechanism of action.
In the latest analysis, the risk of myocardial infarction was reduced by 40% if patients had been treated with an anti–tumor necrosis factor (anti-TNF) drug rather than a nonbiologic disease-modifying antirheumatic drug (nbDMARD) (adjusted hazard ratio, 0.6).
But this effect was not the result of a reduction in the severity of inflammation, said researcher Dr. Audrey Low of the Arthritis Research UK Epidemiology Unit at the University of Manchester, England, where the British Society for Rheumatology Biologics Registers’ Rheumatoid Arthritis (BSRBR-RA) Register is run. Nevertheless, the effect could still relate to attributes of these drugs themselves or better overall disease control, she and her coworkers reported at the British Society for Rheumatology annual conference.
"It is now well known that our patients with rheumatoid arthritis are at increased risk of cardiovascular disease," Dr. Low observed, adding that the risk of both cardiovascular morbidity and mortality is around 50% higher than in the general population. "In particular, the risk of MI [myocardial infarction] is increased," she noted.
Inflammation is thought to play an important role in the development of both atherosclerosis and rheumatoid arthritis, and thus dampening down the inflammatory response with anti-TNF drugs could potentially modify the elevated cardiovascular risk seen in RA patients.
Previously, Dr. Low reported that the risk of MI was lowered by 30% in patients treated with anti-TNFs versus nbDMARDs. The current study looked more deeply into the possible relationship, linking BSRBR-RA Register data to those in the Myocardial Ischaemia National Audit Project (MINAP), a large MI data set derived from all hospitalizations for heart attack in England and Wales. MINAP was established in 1999, 2 years before the BSRBR-RA Register, and it collects around 90,000 records of possible MI per year. As of 2012, MINAP consisted of more than 1 million records.
The aims of the current analysis, which included 14,258 patients with active RA, were to first look at the incidence of MI in patients treated with anti-TNFs (n = 11,200) versus nbDMARDs (n = 3,058), and then to see if the severity of MI was influenced by biologic treatment. The analysis included all patients who started treatment with one of three established anti-TNFs (etanercept, infliximab, and adalimumab) and were recruited into the BSRBR-RA Register between 2001 and 2008. Patients with prior cardiovascular disease were excluded.
To verify the occurrence of MI, data from the BSRBR-RA Register were linked to data from MINAP using patients’ forenames, surnames, date of birth, National Health Service number, postal code, and gender. Events occurring in the same 30-day time window in both data sets were considered the same event. MIs were then verified using criteria set by the American Heart Association and the European Society for Cardiology, with additional criteria of thrombolysis, angioplasty, and MI listed as the underlying cause of death on death certificates, based on the World Health Organization’s International Classification of Diseases, version 10.
A total of 252 first MIs were analyzed, of which 194 occurred in anti-TNF–treated patients at a median follow-up of 5.3 years and 58 occurred in those treated with nbDMARDs at a median follow-up of 3.5 years. The corresponding crude incidence rates were 3.5 and 5.6 per 1,000 patient-years.
"Looking at the MI severity, we only analyzed those MIs that had MINAP-associated data," which was just over half of the MIs (n = 143), Dr. Low said. While the absolute levels of three cardiac enzymes measured – peak troponin I, peak troponin T, and peak creatinine kinase – were higher in the anti-TNF cohort, compared with nbDMARDs, there were not enough events in each group to achieve statistical significance.
There were also no differences between the treatment groups in terms of other cardiac measures of severity, including the phenotype of MI (ST- vs. non-ST elevated), cardiac arrest, and length of hospitalization.
"So the second conclusion is that severity and post-MI mortality do not appear to be influenced by anti-TNF therapy," Dr. Low said.
BSR president Dr. Chris Deighton, who chaired the plenary session in which the findings were presented, noted that there seemed to be a high proportion of patients in the anti-TNF arm who were also treated with steroidal or nonsteroidal anti-inflammatory drugs (NSAIDs) at enrollment. Perhaps if the anti-TNFs were working well, then any reduction in the use of these other drugs over time might have had a confounding effect on the MI risk.
Dr. Low noted that the data on the use of steroids was quite crude and often the dose was not recorded, and even less information about NSAIDs was available. "This is something that we will have to work on, and it does remain to be a potential confounding factor in the analysis," she concluded.
The BSRBR is funded by a grant from the BSR, which receives funding from multiple drug companies. This income finances a separate contract between the BSR and the University of Manchester that provides and runs the BSRBR. Dr. Low had no personal conflicts of interest.
LIVERPOOL, ENGLAND – A new analysis of data from the British Society for Rheumatology Biologics Registers’ Rheumatoid Arthritis Register confirms that anti–tumor necrosis factor treatment decreases patients’ risk for heart attack when compared with treatment with traditional, nonbiologic disease-modifying antirheumatic drugs, but a lower level of inflammation may not be the mechanism of action.
In the latest analysis, the risk of myocardial infarction was reduced by 40% if patients had been treated with an anti–tumor necrosis factor (anti-TNF) drug rather than a nonbiologic disease-modifying antirheumatic drug (nbDMARD) (adjusted hazard ratio, 0.6).
But this effect was not the result of a reduction in the severity of inflammation, said researcher Dr. Audrey Low of the Arthritis Research UK Epidemiology Unit at the University of Manchester, England, where the British Society for Rheumatology Biologics Registers’ Rheumatoid Arthritis (BSRBR-RA) Register is run. Nevertheless, the effect could still relate to attributes of these drugs themselves or better overall disease control, she and her coworkers reported at the British Society for Rheumatology annual conference.
"It is now well known that our patients with rheumatoid arthritis are at increased risk of cardiovascular disease," Dr. Low observed, adding that the risk of both cardiovascular morbidity and mortality is around 50% higher than in the general population. "In particular, the risk of MI [myocardial infarction] is increased," she noted.
Inflammation is thought to play an important role in the development of both atherosclerosis and rheumatoid arthritis, and thus dampening down the inflammatory response with anti-TNF drugs could potentially modify the elevated cardiovascular risk seen in RA patients.
Previously, Dr. Low reported that the risk of MI was lowered by 30% in patients treated with anti-TNFs versus nbDMARDs. The current study looked more deeply into the possible relationship, linking BSRBR-RA Register data to those in the Myocardial Ischaemia National Audit Project (MINAP), a large MI data set derived from all hospitalizations for heart attack in England and Wales. MINAP was established in 1999, 2 years before the BSRBR-RA Register, and it collects around 90,000 records of possible MI per year. As of 2012, MINAP consisted of more than 1 million records.
The aims of the current analysis, which included 14,258 patients with active RA, were to first look at the incidence of MI in patients treated with anti-TNFs (n = 11,200) versus nbDMARDs (n = 3,058), and then to see if the severity of MI was influenced by biologic treatment. The analysis included all patients who started treatment with one of three established anti-TNFs (etanercept, infliximab, and adalimumab) and were recruited into the BSRBR-RA Register between 2001 and 2008. Patients with prior cardiovascular disease were excluded.
To verify the occurrence of MI, data from the BSRBR-RA Register were linked to data from MINAP using patients’ forenames, surnames, date of birth, National Health Service number, postal code, and gender. Events occurring in the same 30-day time window in both data sets were considered the same event. MIs were then verified using criteria set by the American Heart Association and the European Society for Cardiology, with additional criteria of thrombolysis, angioplasty, and MI listed as the underlying cause of death on death certificates, based on the World Health Organization’s International Classification of Diseases, version 10.
A total of 252 first MIs were analyzed, of which 194 occurred in anti-TNF–treated patients at a median follow-up of 5.3 years and 58 occurred in those treated with nbDMARDs at a median follow-up of 3.5 years. The corresponding crude incidence rates were 3.5 and 5.6 per 1,000 patient-years.
"Looking at the MI severity, we only analyzed those MIs that had MINAP-associated data," which was just over half of the MIs (n = 143), Dr. Low said. While the absolute levels of three cardiac enzymes measured – peak troponin I, peak troponin T, and peak creatinine kinase – were higher in the anti-TNF cohort, compared with nbDMARDs, there were not enough events in each group to achieve statistical significance.
There were also no differences between the treatment groups in terms of other cardiac measures of severity, including the phenotype of MI (ST- vs. non-ST elevated), cardiac arrest, and length of hospitalization.
"So the second conclusion is that severity and post-MI mortality do not appear to be influenced by anti-TNF therapy," Dr. Low said.
BSR president Dr. Chris Deighton, who chaired the plenary session in which the findings were presented, noted that there seemed to be a high proportion of patients in the anti-TNF arm who were also treated with steroidal or nonsteroidal anti-inflammatory drugs (NSAIDs) at enrollment. Perhaps if the anti-TNFs were working well, then any reduction in the use of these other drugs over time might have had a confounding effect on the MI risk.
Dr. Low noted that the data on the use of steroids was quite crude and often the dose was not recorded, and even less information about NSAIDs was available. "This is something that we will have to work on, and it does remain to be a potential confounding factor in the analysis," she concluded.
The BSRBR is funded by a grant from the BSR, which receives funding from multiple drug companies. This income finances a separate contract between the BSR and the University of Manchester that provides and runs the BSRBR. Dr. Low had no personal conflicts of interest.
LIVERPOOL, ENGLAND – A new analysis of data from the British Society for Rheumatology Biologics Registers’ Rheumatoid Arthritis Register confirms that anti–tumor necrosis factor treatment decreases patients’ risk for heart attack when compared with treatment with traditional, nonbiologic disease-modifying antirheumatic drugs, but a lower level of inflammation may not be the mechanism of action.
In the latest analysis, the risk of myocardial infarction was reduced by 40% if patients had been treated with an anti–tumor necrosis factor (anti-TNF) drug rather than a nonbiologic disease-modifying antirheumatic drug (nbDMARD) (adjusted hazard ratio, 0.6).
But this effect was not the result of a reduction in the severity of inflammation, said researcher Dr. Audrey Low of the Arthritis Research UK Epidemiology Unit at the University of Manchester, England, where the British Society for Rheumatology Biologics Registers’ Rheumatoid Arthritis (BSRBR-RA) Register is run. Nevertheless, the effect could still relate to attributes of these drugs themselves or better overall disease control, she and her coworkers reported at the British Society for Rheumatology annual conference.
"It is now well known that our patients with rheumatoid arthritis are at increased risk of cardiovascular disease," Dr. Low observed, adding that the risk of both cardiovascular morbidity and mortality is around 50% higher than in the general population. "In particular, the risk of MI [myocardial infarction] is increased," she noted.
Inflammation is thought to play an important role in the development of both atherosclerosis and rheumatoid arthritis, and thus dampening down the inflammatory response with anti-TNF drugs could potentially modify the elevated cardiovascular risk seen in RA patients.
Previously, Dr. Low reported that the risk of MI was lowered by 30% in patients treated with anti-TNFs versus nbDMARDs. The current study looked more deeply into the possible relationship, linking BSRBR-RA Register data to those in the Myocardial Ischaemia National Audit Project (MINAP), a large MI data set derived from all hospitalizations for heart attack in England and Wales. MINAP was established in 1999, 2 years before the BSRBR-RA Register, and it collects around 90,000 records of possible MI per year. As of 2012, MINAP consisted of more than 1 million records.
The aims of the current analysis, which included 14,258 patients with active RA, were to first look at the incidence of MI in patients treated with anti-TNFs (n = 11,200) versus nbDMARDs (n = 3,058), and then to see if the severity of MI was influenced by biologic treatment. The analysis included all patients who started treatment with one of three established anti-TNFs (etanercept, infliximab, and adalimumab) and were recruited into the BSRBR-RA Register between 2001 and 2008. Patients with prior cardiovascular disease were excluded.
To verify the occurrence of MI, data from the BSRBR-RA Register were linked to data from MINAP using patients’ forenames, surnames, date of birth, National Health Service number, postal code, and gender. Events occurring in the same 30-day time window in both data sets were considered the same event. MIs were then verified using criteria set by the American Heart Association and the European Society for Cardiology, with additional criteria of thrombolysis, angioplasty, and MI listed as the underlying cause of death on death certificates, based on the World Health Organization’s International Classification of Diseases, version 10.
A total of 252 first MIs were analyzed, of which 194 occurred in anti-TNF–treated patients at a median follow-up of 5.3 years and 58 occurred in those treated with nbDMARDs at a median follow-up of 3.5 years. The corresponding crude incidence rates were 3.5 and 5.6 per 1,000 patient-years.
"Looking at the MI severity, we only analyzed those MIs that had MINAP-associated data," which was just over half of the MIs (n = 143), Dr. Low said. While the absolute levels of three cardiac enzymes measured – peak troponin I, peak troponin T, and peak creatinine kinase – were higher in the anti-TNF cohort, compared with nbDMARDs, there were not enough events in each group to achieve statistical significance.
There were also no differences between the treatment groups in terms of other cardiac measures of severity, including the phenotype of MI (ST- vs. non-ST elevated), cardiac arrest, and length of hospitalization.
"So the second conclusion is that severity and post-MI mortality do not appear to be influenced by anti-TNF therapy," Dr. Low said.
BSR president Dr. Chris Deighton, who chaired the plenary session in which the findings were presented, noted that there seemed to be a high proportion of patients in the anti-TNF arm who were also treated with steroidal or nonsteroidal anti-inflammatory drugs (NSAIDs) at enrollment. Perhaps if the anti-TNFs were working well, then any reduction in the use of these other drugs over time might have had a confounding effect on the MI risk.
Dr. Low noted that the data on the use of steroids was quite crude and often the dose was not recorded, and even less information about NSAIDs was available. "This is something that we will have to work on, and it does remain to be a potential confounding factor in the analysis," she concluded.
The BSRBR is funded by a grant from the BSR, which receives funding from multiple drug companies. This income finances a separate contract between the BSR and the University of Manchester that provides and runs the BSRBR. Dr. Low had no personal conflicts of interest.
AT RHEUMATOLOGY 2014
Key clinical point: Linkage of BSRBR-RA Register data with a database of MIs in England and Wales further solidifies the evidence for lower risk of MI in RA patients who take anti-TNF drugs.
Major finding: Anti-TNFs were associated with a lower MI risk than traditional DMARDs (adjusted hazard ratio, 0.6).
Data source: More than 14,000 patients enrolled in the BSRBR-RA Register who were treated with anti-TNF therapy or nonbiologic DMARDs.
Disclosures: The BSRBR is funded by a grant from the BSR, which receives funding from multiple drug companies. This income finances a separate contract between the BSR and the University of Manchester that provides and runs the BSRBR-RA Register. Dr. Low had no personal conflicts of interest.
Reduced risk of RA in schizophrenia may be bias, not biology
The observed reduced risk of rheumatoid arthritis and osteoarthritis in patients with schizophrenia might be tied to reporting bias rather than disease-specific biology, Dr. Carl Sellgren and colleagues reported.
Data on more than 6 million people contained in the Swedish Population Register showed significantly reduced risks of both disorders, and of other musculoskeletal diseases, among schizophrenia patients, wrote Dr. Sellgren of the Karolinksa Institute, Stockholm, and his associates. Conversely, no increased risks were found among patients with bipolar disorder, despite a shared polygenic component of 68% between the two mental illnesses (Schizophr. Bull. 2014 Apr. 8 [doi:10.1093/schbul/sbu054]).
"In light of the more severe loss of function in schizophrenia and schizoaffective disorder patients in comparison to bipolar disease patients, this is compatible with differential misclassification bias in the observed inverse association between schizophrenia and rheumatoid arthritis," the researchers wrote.
Dr. Sellgren and his team were not ready to entirely scrap the possibility of some biologic link, however.
"Interestingly, we observed a significantly decreased risk for seronegative RA in children and siblings of schizophrenia probands," he said in an interview. "Correspondingly, the lowest point estimate for schizophrenia was observed in relatives of seronegative RA patients, and the lowest point estimate among schizophrenia patients was also observed for seronegative RA. Genetic causation may be present regarding the specific association between schizophrenia and seronegative RA."
Whatever the link, the study points up the importance of looking at schizophrenia patients holistically, rather than only focusing on their mental illnesses. "It appears that the more severely affected the patient is of the psychiatric disorder, the less likely is the clinician to identify a comorbid musculoskeletal disease," he said. "These data emphasize the need of assessing and being aware of nonpsychiatric symptoms in schizophrenia."
The Swedish Medical Research Council funded the study. Dr. Sellgren made no financial disclosures.
On Twitter @Alz_Gal
The observed reduced risk of rheumatoid arthritis and osteoarthritis in patients with schizophrenia might be tied to reporting bias rather than disease-specific biology, Dr. Carl Sellgren and colleagues reported.
Data on more than 6 million people contained in the Swedish Population Register showed significantly reduced risks of both disorders, and of other musculoskeletal diseases, among schizophrenia patients, wrote Dr. Sellgren of the Karolinksa Institute, Stockholm, and his associates. Conversely, no increased risks were found among patients with bipolar disorder, despite a shared polygenic component of 68% between the two mental illnesses (Schizophr. Bull. 2014 Apr. 8 [doi:10.1093/schbul/sbu054]).
"In light of the more severe loss of function in schizophrenia and schizoaffective disorder patients in comparison to bipolar disease patients, this is compatible with differential misclassification bias in the observed inverse association between schizophrenia and rheumatoid arthritis," the researchers wrote.
Dr. Sellgren and his team were not ready to entirely scrap the possibility of some biologic link, however.
"Interestingly, we observed a significantly decreased risk for seronegative RA in children and siblings of schizophrenia probands," he said in an interview. "Correspondingly, the lowest point estimate for schizophrenia was observed in relatives of seronegative RA patients, and the lowest point estimate among schizophrenia patients was also observed for seronegative RA. Genetic causation may be present regarding the specific association between schizophrenia and seronegative RA."
Whatever the link, the study points up the importance of looking at schizophrenia patients holistically, rather than only focusing on their mental illnesses. "It appears that the more severely affected the patient is of the psychiatric disorder, the less likely is the clinician to identify a comorbid musculoskeletal disease," he said. "These data emphasize the need of assessing and being aware of nonpsychiatric symptoms in schizophrenia."
The Swedish Medical Research Council funded the study. Dr. Sellgren made no financial disclosures.
On Twitter @Alz_Gal
The observed reduced risk of rheumatoid arthritis and osteoarthritis in patients with schizophrenia might be tied to reporting bias rather than disease-specific biology, Dr. Carl Sellgren and colleagues reported.
Data on more than 6 million people contained in the Swedish Population Register showed significantly reduced risks of both disorders, and of other musculoskeletal diseases, among schizophrenia patients, wrote Dr. Sellgren of the Karolinksa Institute, Stockholm, and his associates. Conversely, no increased risks were found among patients with bipolar disorder, despite a shared polygenic component of 68% between the two mental illnesses (Schizophr. Bull. 2014 Apr. 8 [doi:10.1093/schbul/sbu054]).
"In light of the more severe loss of function in schizophrenia and schizoaffective disorder patients in comparison to bipolar disease patients, this is compatible with differential misclassification bias in the observed inverse association between schizophrenia and rheumatoid arthritis," the researchers wrote.
Dr. Sellgren and his team were not ready to entirely scrap the possibility of some biologic link, however.
"Interestingly, we observed a significantly decreased risk for seronegative RA in children and siblings of schizophrenia probands," he said in an interview. "Correspondingly, the lowest point estimate for schizophrenia was observed in relatives of seronegative RA patients, and the lowest point estimate among schizophrenia patients was also observed for seronegative RA. Genetic causation may be present regarding the specific association between schizophrenia and seronegative RA."
Whatever the link, the study points up the importance of looking at schizophrenia patients holistically, rather than only focusing on their mental illnesses. "It appears that the more severely affected the patient is of the psychiatric disorder, the less likely is the clinician to identify a comorbid musculoskeletal disease," he said. "These data emphasize the need of assessing and being aware of nonpsychiatric symptoms in schizophrenia."
The Swedish Medical Research Council funded the study. Dr. Sellgren made no financial disclosures.
On Twitter @Alz_Gal
FROM SCHIZOPHRENIA BULLETIN
Major finding: The risk for RA was decreased (hazard ratio, 0.69; 95% confidence interval, 0.59-0.80) in schizophrenia, but not in bipolar disorder. These findings suggest that the risk reduction could be attributable to reporting and misclassification bias.
Data source: The review included more than 6 million people in the Swedish Population Register.
Disclosures: The Swedish Medical Research Council funded the study. Dr. Sellgren had no financial disclosures.
Stopping biologics in RA remission remains uncertain
Once a rheumatoid arthritis patient maintains relatively long-term remission on a regimen that combines a synthetic and a biologic disease-modifying drug, temptation mounts to try withdrawing one of the two drugs. Cost considerations alone can make it irresistible to patients to taper down or stop an expensive biologic drug to see whether the patient’s remission can sustain dose reduction or elimination.
But several American experts cautioned that despite growing evidence that at least some rheumatoid arthritis (RA) patients will stay in remission after their biologic disease-modifying antirheumatic drug (DMARD) is gone, the overall efficacy and safety of this approach remain uncertain and it’s not fully clear which patients are the best candidates. Biologic-drug withdrawal has not repeatedly been shown to be fully safe and worth attempting with results from rigorously controlled and adequately powered studies, an evidence base that may still be several years off.
"We can try withdrawing a drug," and many American RA patients do just that, a step more often than not initiated by the patient. "But we don’t know if it’s benign for radiographic progression or for cardiovascular disease events," said Dr. Arthur Weinstein, director of rheumatology at Washington (D.C.) Hospital Center. Even though RA patients in remission who taper down or fully stop their biologic DMARD are usually closely monitored for flare, "there is some evidence for a disconnect between radiographic progression and clinical status," raising the danger that a patient may appear to remain in remission when off the biologic drug while joint deterioration occurs. That, coupled with concern that an RA patient’s cardiovascular risk may worsen off their biologic, can make patients and physicians hesitate when faced with the prospect of messing with a stable remission on a combined biologic and synthetic DMARD regimen, Dr. Weinstein said in an interview.
Biologic withdrawal can succeed
Despite that, it’s undeniable that a substantial minority or even a majority in some series of RA patients in remission or low disease activity on both a biologic and synthetic DMARD can taper down or stop the biologic without a flare occurring. The flip side of that is, of course, that in many series a good number of patients who stop their biologic have a flare and need to have the drug restarted. The good news for these scenarios is that prompt restart of the biologic once a flare occurs generally seems capable of restoring remission with no long-term deleterious effects.
In its 2013 RA treatment guidelines, the European League Against Rheumatism (EULAR) said that "if a patient is in persistent remission after having tapered glucocorticoids, one can consider tapering biologic DMARDs, especially if this treatment is combined with a synthetic DMARD" (Ann. Rheum. Dis. 2014;73:492-509). The most recent RA management guidelines from the American College of Rheumatology, published in 2012, don’t address tapering down or withdrawing biologics. Dr. Weinstein noted that the EULAR recommendation is largely based on consensus expert opinion without a definitive evidence base to justify this approach.
Recent evidence documenting the success of biologic withdrawal includes a report earlier this year from a prespecified analysis in the OPTIMA (Optimal Protocol for Treatment Initiation with Methotrexate and Adalimumab) trial that detailed the outcomes of 207 patients who had stable, low disease activity on combined treatment with adalimumab (Humira) and methotrexate and were randomized to either adalimumab withdrawal or continuation. A year later, 70% of patients who maintained the two-drug regimen and 54% of those randomized to stop adalimumab remained at low disease activity and had no sign of radiographic progression, a between-group difference that was not statistically significant (Lancet 2014;383:321-32).
But in other studies, patients withdrawn from a biologic did not fare nearly as well. The PRESERVE (Maintenance, induction, or withdrawal of etanercept after treatment with etanercept and methotrexate in patients with moderate rheumatoid arthritis) trial randomized 604 RA patients with low disease activity on a combination of etanercept (Enbrel) and methotrexate to continue to receive 50 mg of etanercept plus methotrexate weekly, step down to 25 mg of etanercept plus methotrexate weekly, or completely stop etanercept and continue on methotrexate alone. A year later, the percentage of patients who remained in low disease activity tallied 83% in those who received at least one dose of 50 mg etanercept, 79% of patients in the 25-mg etanercept group, and 43% in the group maintained on methotrexate monotherapy, statistically significant differences between each of the two etanercept groups and the monotherapy arm (Lancet 2013;381:918-29).
A more real-world assessment included 717 RA patients at low disease activity who discontinued their first biologic drug while enrolled in the CORRONA (Consortium of Rheumatology Researchers of North America) registry. The results showed that after 1 year on monotherapy without a biologic, 73% of patients remained free from a flare, and that state persisted in 56% to 18 months, 42% after 2 years, and 28% after 3 years on monotherapy, Dr. Arthur F. Kavanaugh and his associates reported last year at the annual meeting of the American College of Rheumatology (Arthritis Rheum. 2013;65:S603).
Finding the right patients
Despite evidence like this, the right time and the right patient for withdrawing a biologic remain murky. "At this point it is investigational," said Dr. Kavanaugh in an interview. "In the United States, it is mostly initiated by patients. There are many factors to consider, including the duration of RA, how well the patients are doing, is the patient on another drug that should be stopped first such as prednisone, and can the synthetic DMARD like methotrexate sustain the response," said Dr. Kavanaugh, a rheumatologist who is professor of clinical medicine and director of the Center for Innovative Therapy at the University of California, San Diego.
Routinely attempting biologic taper-down or withdrawal from RA patients in remission or at low disease activity "is not yet standard practice, but many patients do it," said Dr. Joel M. Kremer, a rheumatologist and professor of medicine at Albany (N.Y.) Medical College. When he attempts withdrawing a biologic, he prefers to use a gradual taper-down of the dosage, and he said he is more reluctant to attempt this for patients who began treatment with severe RA, and that he is more likely to try it for patients who began treatment with early RA, although he admitted that this remains uncertain. Like all experts interviewed, he said the type of biologic a patient is on doesn’t seem to matter.
"I think this entire area needs more study," Dr. Kremer said in an interview. For example, "are there biomarkers associated with the ability to withdraw? I don’t believe we know much about who can or should withdraw." Once withdrawal occurs, he recommended careful follow-up to detect a flare quickly, and restarting the biologic if even a mild flare occurs.
Biologic withdrawal "is really more investigational" right now, said Dr. Eric L. Matteson, professor and chairman of rheumatology at the Mayo Clinic in Rochester, Minn. "No guideline establishes it as standard of practice. I like to see patients in full remission for a year before considering such a move," he said in an interview. Withdrawal depends on whether the RA is well controlled and not on whether treatment started on early or established RA, he added; nor did he think that a flare after withdrawal precludes future withdrawal attempts as long as the patient returns to a durable, full remission. But Dr. Matteson did caution about also considering extra-articular disease like vasculitis or iritis that may be active even when a patient’s joints are not. "Any evidence of recurrent disease should cause the patient and physician to consider ramping the therapy back up."
Like others, Dr. Matteson highlighted the lingering unknowns about biologic withdrawal that keep it from being standard practice. "We don’t know whether the biologic or synthetic DMARD should get withdrawn first. We don’t have a great handle on what biomarkers to use to help decide if there is subclinical disease activity that necessitates reimplementation of the biologic. We don’t know which patients may violently flare following withdrawal and which might not. We also don’t know if retreatment of the disease is as good when a biologic is withdrawn and then restarted. Finally, I think we overestimate the percent of our patients who are actually in remission or a low-disease-activity state and underestimate who really should continue their medications."
The STARA trial
Dr. Weinstein and his associates recently planned a U.S.-based trial that has come close to launching to address several of these issues, the STARA (Stopping TNF-Alpha Inhibitors in Rheumatoid Arthritis) trial.
"One aspect of STARA is to determine whether there is a profile of clinical, imaging, and laboratory factors that will reliably distinguish patients who will flare from those who will remain in remission" following withdrawal of a biologic DMARD, said Dr. Weinstein, STARA’s lead investigator and also a professor of medicine at Georgetown University in Washington. "Another aim is to find predictors of good candidates for withdrawal. The advantage of STARA is patients are drawn from real-world practice and not from a clinical trial." Dr. Weinstein faulted some of the published studies of biologic withdrawal, such as OPTIMA and PRESERVE, because they included trial patients exclusively and not patients managed in routine practice.
STARA’s design uses a sudden, full withdrawal of the biologic to allow a standardized approach to patients who could enter the study on etanercept, adalimumab, or infliximab (Remicade). For routine practice, gradual withdrawal is more common, though there is no evidence this produces better long-term outcomes. An important withdrawal question that STARA won’t address is: Which is the better drug to remove, the biologic or the synthetic DMARD? In most cases, the more expensive biologic is the patient’s choice, but ideally this issue should be addressed in a randomized trial, Dr. Weinstein said.
But as of late March, STARA’s status was in doubt, as anticipated funding was withdrawn. If STARA isn’t run, Dr. Weinstein predicts that attempts to cut biologics from dual regimens will continue and likely expand, but with unknown implications for long-term safety that he believes can only be addressed in a prospective, randomized trial.
Dr. Weinstein said that he had no relevant disclosures. Dr. Matteson had no disclosures. Dr. Kremer has received research support and been a consultant to more than 10 drug companies. Dr. Kavanaugh has received research support from more than 10 drug companies.
On Twitter @mitchelzoler
Once a rheumatoid arthritis patient maintains relatively long-term remission on a regimen that combines a synthetic and a biologic disease-modifying drug, temptation mounts to try withdrawing one of the two drugs. Cost considerations alone can make it irresistible to patients to taper down or stop an expensive biologic drug to see whether the patient’s remission can sustain dose reduction or elimination.
But several American experts cautioned that despite growing evidence that at least some rheumatoid arthritis (RA) patients will stay in remission after their biologic disease-modifying antirheumatic drug (DMARD) is gone, the overall efficacy and safety of this approach remain uncertain and it’s not fully clear which patients are the best candidates. Biologic-drug withdrawal has not repeatedly been shown to be fully safe and worth attempting with results from rigorously controlled and adequately powered studies, an evidence base that may still be several years off.
"We can try withdrawing a drug," and many American RA patients do just that, a step more often than not initiated by the patient. "But we don’t know if it’s benign for radiographic progression or for cardiovascular disease events," said Dr. Arthur Weinstein, director of rheumatology at Washington (D.C.) Hospital Center. Even though RA patients in remission who taper down or fully stop their biologic DMARD are usually closely monitored for flare, "there is some evidence for a disconnect between radiographic progression and clinical status," raising the danger that a patient may appear to remain in remission when off the biologic drug while joint deterioration occurs. That, coupled with concern that an RA patient’s cardiovascular risk may worsen off their biologic, can make patients and physicians hesitate when faced with the prospect of messing with a stable remission on a combined biologic and synthetic DMARD regimen, Dr. Weinstein said in an interview.
Biologic withdrawal can succeed
Despite that, it’s undeniable that a substantial minority or even a majority in some series of RA patients in remission or low disease activity on both a biologic and synthetic DMARD can taper down or stop the biologic without a flare occurring. The flip side of that is, of course, that in many series a good number of patients who stop their biologic have a flare and need to have the drug restarted. The good news for these scenarios is that prompt restart of the biologic once a flare occurs generally seems capable of restoring remission with no long-term deleterious effects.
In its 2013 RA treatment guidelines, the European League Against Rheumatism (EULAR) said that "if a patient is in persistent remission after having tapered glucocorticoids, one can consider tapering biologic DMARDs, especially if this treatment is combined with a synthetic DMARD" (Ann. Rheum. Dis. 2014;73:492-509). The most recent RA management guidelines from the American College of Rheumatology, published in 2012, don’t address tapering down or withdrawing biologics. Dr. Weinstein noted that the EULAR recommendation is largely based on consensus expert opinion without a definitive evidence base to justify this approach.
Recent evidence documenting the success of biologic withdrawal includes a report earlier this year from a prespecified analysis in the OPTIMA (Optimal Protocol for Treatment Initiation with Methotrexate and Adalimumab) trial that detailed the outcomes of 207 patients who had stable, low disease activity on combined treatment with adalimumab (Humira) and methotrexate and were randomized to either adalimumab withdrawal or continuation. A year later, 70% of patients who maintained the two-drug regimen and 54% of those randomized to stop adalimumab remained at low disease activity and had no sign of radiographic progression, a between-group difference that was not statistically significant (Lancet 2014;383:321-32).
But in other studies, patients withdrawn from a biologic did not fare nearly as well. The PRESERVE (Maintenance, induction, or withdrawal of etanercept after treatment with etanercept and methotrexate in patients with moderate rheumatoid arthritis) trial randomized 604 RA patients with low disease activity on a combination of etanercept (Enbrel) and methotrexate to continue to receive 50 mg of etanercept plus methotrexate weekly, step down to 25 mg of etanercept plus methotrexate weekly, or completely stop etanercept and continue on methotrexate alone. A year later, the percentage of patients who remained in low disease activity tallied 83% in those who received at least one dose of 50 mg etanercept, 79% of patients in the 25-mg etanercept group, and 43% in the group maintained on methotrexate monotherapy, statistically significant differences between each of the two etanercept groups and the monotherapy arm (Lancet 2013;381:918-29).
A more real-world assessment included 717 RA patients at low disease activity who discontinued their first biologic drug while enrolled in the CORRONA (Consortium of Rheumatology Researchers of North America) registry. The results showed that after 1 year on monotherapy without a biologic, 73% of patients remained free from a flare, and that state persisted in 56% to 18 months, 42% after 2 years, and 28% after 3 years on monotherapy, Dr. Arthur F. Kavanaugh and his associates reported last year at the annual meeting of the American College of Rheumatology (Arthritis Rheum. 2013;65:S603).
Finding the right patients
Despite evidence like this, the right time and the right patient for withdrawing a biologic remain murky. "At this point it is investigational," said Dr. Kavanaugh in an interview. "In the United States, it is mostly initiated by patients. There are many factors to consider, including the duration of RA, how well the patients are doing, is the patient on another drug that should be stopped first such as prednisone, and can the synthetic DMARD like methotrexate sustain the response," said Dr. Kavanaugh, a rheumatologist who is professor of clinical medicine and director of the Center for Innovative Therapy at the University of California, San Diego.
Routinely attempting biologic taper-down or withdrawal from RA patients in remission or at low disease activity "is not yet standard practice, but many patients do it," said Dr. Joel M. Kremer, a rheumatologist and professor of medicine at Albany (N.Y.) Medical College. When he attempts withdrawing a biologic, he prefers to use a gradual taper-down of the dosage, and he said he is more reluctant to attempt this for patients who began treatment with severe RA, and that he is more likely to try it for patients who began treatment with early RA, although he admitted that this remains uncertain. Like all experts interviewed, he said the type of biologic a patient is on doesn’t seem to matter.
"I think this entire area needs more study," Dr. Kremer said in an interview. For example, "are there biomarkers associated with the ability to withdraw? I don’t believe we know much about who can or should withdraw." Once withdrawal occurs, he recommended careful follow-up to detect a flare quickly, and restarting the biologic if even a mild flare occurs.
Biologic withdrawal "is really more investigational" right now, said Dr. Eric L. Matteson, professor and chairman of rheumatology at the Mayo Clinic in Rochester, Minn. "No guideline establishes it as standard of practice. I like to see patients in full remission for a year before considering such a move," he said in an interview. Withdrawal depends on whether the RA is well controlled and not on whether treatment started on early or established RA, he added; nor did he think that a flare after withdrawal precludes future withdrawal attempts as long as the patient returns to a durable, full remission. But Dr. Matteson did caution about also considering extra-articular disease like vasculitis or iritis that may be active even when a patient’s joints are not. "Any evidence of recurrent disease should cause the patient and physician to consider ramping the therapy back up."
Like others, Dr. Matteson highlighted the lingering unknowns about biologic withdrawal that keep it from being standard practice. "We don’t know whether the biologic or synthetic DMARD should get withdrawn first. We don’t have a great handle on what biomarkers to use to help decide if there is subclinical disease activity that necessitates reimplementation of the biologic. We don’t know which patients may violently flare following withdrawal and which might not. We also don’t know if retreatment of the disease is as good when a biologic is withdrawn and then restarted. Finally, I think we overestimate the percent of our patients who are actually in remission or a low-disease-activity state and underestimate who really should continue their medications."
The STARA trial
Dr. Weinstein and his associates recently planned a U.S.-based trial that has come close to launching to address several of these issues, the STARA (Stopping TNF-Alpha Inhibitors in Rheumatoid Arthritis) trial.
"One aspect of STARA is to determine whether there is a profile of clinical, imaging, and laboratory factors that will reliably distinguish patients who will flare from those who will remain in remission" following withdrawal of a biologic DMARD, said Dr. Weinstein, STARA’s lead investigator and also a professor of medicine at Georgetown University in Washington. "Another aim is to find predictors of good candidates for withdrawal. The advantage of STARA is patients are drawn from real-world practice and not from a clinical trial." Dr. Weinstein faulted some of the published studies of biologic withdrawal, such as OPTIMA and PRESERVE, because they included trial patients exclusively and not patients managed in routine practice.
STARA’s design uses a sudden, full withdrawal of the biologic to allow a standardized approach to patients who could enter the study on etanercept, adalimumab, or infliximab (Remicade). For routine practice, gradual withdrawal is more common, though there is no evidence this produces better long-term outcomes. An important withdrawal question that STARA won’t address is: Which is the better drug to remove, the biologic or the synthetic DMARD? In most cases, the more expensive biologic is the patient’s choice, but ideally this issue should be addressed in a randomized trial, Dr. Weinstein said.
But as of late March, STARA’s status was in doubt, as anticipated funding was withdrawn. If STARA isn’t run, Dr. Weinstein predicts that attempts to cut biologics from dual regimens will continue and likely expand, but with unknown implications for long-term safety that he believes can only be addressed in a prospective, randomized trial.
Dr. Weinstein said that he had no relevant disclosures. Dr. Matteson had no disclosures. Dr. Kremer has received research support and been a consultant to more than 10 drug companies. Dr. Kavanaugh has received research support from more than 10 drug companies.
On Twitter @mitchelzoler
Once a rheumatoid arthritis patient maintains relatively long-term remission on a regimen that combines a synthetic and a biologic disease-modifying drug, temptation mounts to try withdrawing one of the two drugs. Cost considerations alone can make it irresistible to patients to taper down or stop an expensive biologic drug to see whether the patient’s remission can sustain dose reduction or elimination.
But several American experts cautioned that despite growing evidence that at least some rheumatoid arthritis (RA) patients will stay in remission after their biologic disease-modifying antirheumatic drug (DMARD) is gone, the overall efficacy and safety of this approach remain uncertain and it’s not fully clear which patients are the best candidates. Biologic-drug withdrawal has not repeatedly been shown to be fully safe and worth attempting with results from rigorously controlled and adequately powered studies, an evidence base that may still be several years off.
"We can try withdrawing a drug," and many American RA patients do just that, a step more often than not initiated by the patient. "But we don’t know if it’s benign for radiographic progression or for cardiovascular disease events," said Dr. Arthur Weinstein, director of rheumatology at Washington (D.C.) Hospital Center. Even though RA patients in remission who taper down or fully stop their biologic DMARD are usually closely monitored for flare, "there is some evidence for a disconnect between radiographic progression and clinical status," raising the danger that a patient may appear to remain in remission when off the biologic drug while joint deterioration occurs. That, coupled with concern that an RA patient’s cardiovascular risk may worsen off their biologic, can make patients and physicians hesitate when faced with the prospect of messing with a stable remission on a combined biologic and synthetic DMARD regimen, Dr. Weinstein said in an interview.
Biologic withdrawal can succeed
Despite that, it’s undeniable that a substantial minority or even a majority in some series of RA patients in remission or low disease activity on both a biologic and synthetic DMARD can taper down or stop the biologic without a flare occurring. The flip side of that is, of course, that in many series a good number of patients who stop their biologic have a flare and need to have the drug restarted. The good news for these scenarios is that prompt restart of the biologic once a flare occurs generally seems capable of restoring remission with no long-term deleterious effects.
In its 2013 RA treatment guidelines, the European League Against Rheumatism (EULAR) said that "if a patient is in persistent remission after having tapered glucocorticoids, one can consider tapering biologic DMARDs, especially if this treatment is combined with a synthetic DMARD" (Ann. Rheum. Dis. 2014;73:492-509). The most recent RA management guidelines from the American College of Rheumatology, published in 2012, don’t address tapering down or withdrawing biologics. Dr. Weinstein noted that the EULAR recommendation is largely based on consensus expert opinion without a definitive evidence base to justify this approach.
Recent evidence documenting the success of biologic withdrawal includes a report earlier this year from a prespecified analysis in the OPTIMA (Optimal Protocol for Treatment Initiation with Methotrexate and Adalimumab) trial that detailed the outcomes of 207 patients who had stable, low disease activity on combined treatment with adalimumab (Humira) and methotrexate and were randomized to either adalimumab withdrawal or continuation. A year later, 70% of patients who maintained the two-drug regimen and 54% of those randomized to stop adalimumab remained at low disease activity and had no sign of radiographic progression, a between-group difference that was not statistically significant (Lancet 2014;383:321-32).
But in other studies, patients withdrawn from a biologic did not fare nearly as well. The PRESERVE (Maintenance, induction, or withdrawal of etanercept after treatment with etanercept and methotrexate in patients with moderate rheumatoid arthritis) trial randomized 604 RA patients with low disease activity on a combination of etanercept (Enbrel) and methotrexate to continue to receive 50 mg of etanercept plus methotrexate weekly, step down to 25 mg of etanercept plus methotrexate weekly, or completely stop etanercept and continue on methotrexate alone. A year later, the percentage of patients who remained in low disease activity tallied 83% in those who received at least one dose of 50 mg etanercept, 79% of patients in the 25-mg etanercept group, and 43% in the group maintained on methotrexate monotherapy, statistically significant differences between each of the two etanercept groups and the monotherapy arm (Lancet 2013;381:918-29).
A more real-world assessment included 717 RA patients at low disease activity who discontinued their first biologic drug while enrolled in the CORRONA (Consortium of Rheumatology Researchers of North America) registry. The results showed that after 1 year on monotherapy without a biologic, 73% of patients remained free from a flare, and that state persisted in 56% to 18 months, 42% after 2 years, and 28% after 3 years on monotherapy, Dr. Arthur F. Kavanaugh and his associates reported last year at the annual meeting of the American College of Rheumatology (Arthritis Rheum. 2013;65:S603).
Finding the right patients
Despite evidence like this, the right time and the right patient for withdrawing a biologic remain murky. "At this point it is investigational," said Dr. Kavanaugh in an interview. "In the United States, it is mostly initiated by patients. There are many factors to consider, including the duration of RA, how well the patients are doing, is the patient on another drug that should be stopped first such as prednisone, and can the synthetic DMARD like methotrexate sustain the response," said Dr. Kavanaugh, a rheumatologist who is professor of clinical medicine and director of the Center for Innovative Therapy at the University of California, San Diego.
Routinely attempting biologic taper-down or withdrawal from RA patients in remission or at low disease activity "is not yet standard practice, but many patients do it," said Dr. Joel M. Kremer, a rheumatologist and professor of medicine at Albany (N.Y.) Medical College. When he attempts withdrawing a biologic, he prefers to use a gradual taper-down of the dosage, and he said he is more reluctant to attempt this for patients who began treatment with severe RA, and that he is more likely to try it for patients who began treatment with early RA, although he admitted that this remains uncertain. Like all experts interviewed, he said the type of biologic a patient is on doesn’t seem to matter.
"I think this entire area needs more study," Dr. Kremer said in an interview. For example, "are there biomarkers associated with the ability to withdraw? I don’t believe we know much about who can or should withdraw." Once withdrawal occurs, he recommended careful follow-up to detect a flare quickly, and restarting the biologic if even a mild flare occurs.
Biologic withdrawal "is really more investigational" right now, said Dr. Eric L. Matteson, professor and chairman of rheumatology at the Mayo Clinic in Rochester, Minn. "No guideline establishes it as standard of practice. I like to see patients in full remission for a year before considering such a move," he said in an interview. Withdrawal depends on whether the RA is well controlled and not on whether treatment started on early or established RA, he added; nor did he think that a flare after withdrawal precludes future withdrawal attempts as long as the patient returns to a durable, full remission. But Dr. Matteson did caution about also considering extra-articular disease like vasculitis or iritis that may be active even when a patient’s joints are not. "Any evidence of recurrent disease should cause the patient and physician to consider ramping the therapy back up."
Like others, Dr. Matteson highlighted the lingering unknowns about biologic withdrawal that keep it from being standard practice. "We don’t know whether the biologic or synthetic DMARD should get withdrawn first. We don’t have a great handle on what biomarkers to use to help decide if there is subclinical disease activity that necessitates reimplementation of the biologic. We don’t know which patients may violently flare following withdrawal and which might not. We also don’t know if retreatment of the disease is as good when a biologic is withdrawn and then restarted. Finally, I think we overestimate the percent of our patients who are actually in remission or a low-disease-activity state and underestimate who really should continue their medications."
The STARA trial
Dr. Weinstein and his associates recently planned a U.S.-based trial that has come close to launching to address several of these issues, the STARA (Stopping TNF-Alpha Inhibitors in Rheumatoid Arthritis) trial.
"One aspect of STARA is to determine whether there is a profile of clinical, imaging, and laboratory factors that will reliably distinguish patients who will flare from those who will remain in remission" following withdrawal of a biologic DMARD, said Dr. Weinstein, STARA’s lead investigator and also a professor of medicine at Georgetown University in Washington. "Another aim is to find predictors of good candidates for withdrawal. The advantage of STARA is patients are drawn from real-world practice and not from a clinical trial." Dr. Weinstein faulted some of the published studies of biologic withdrawal, such as OPTIMA and PRESERVE, because they included trial patients exclusively and not patients managed in routine practice.
STARA’s design uses a sudden, full withdrawal of the biologic to allow a standardized approach to patients who could enter the study on etanercept, adalimumab, or infliximab (Remicade). For routine practice, gradual withdrawal is more common, though there is no evidence this produces better long-term outcomes. An important withdrawal question that STARA won’t address is: Which is the better drug to remove, the biologic or the synthetic DMARD? In most cases, the more expensive biologic is the patient’s choice, but ideally this issue should be addressed in a randomized trial, Dr. Weinstein said.
But as of late March, STARA’s status was in doubt, as anticipated funding was withdrawn. If STARA isn’t run, Dr. Weinstein predicts that attempts to cut biologics from dual regimens will continue and likely expand, but with unknown implications for long-term safety that he believes can only be addressed in a prospective, randomized trial.
Dr. Weinstein said that he had no relevant disclosures. Dr. Matteson had no disclosures. Dr. Kremer has received research support and been a consultant to more than 10 drug companies. Dr. Kavanaugh has received research support from more than 10 drug companies.
On Twitter @mitchelzoler
Chinese herbal remedy found noninferior to methotrexate in RA patients
A traditional Chinese herbal remedy used to treat joint pain, fever, edema, and local inflammation was found noninferior to methotrexate for controlling active rheumatoid arthritis in an open-label, randomized clinical trial involving 207 patients.
Tripterygium wilfordii Hook F (TwHF) is a relatively inexpensive remedy (about $10 per month) often used alone or in combination with methotrexate for rheumatoid arthritis (RA) in China, but it has not been assessed in a rigorous controlled study until now.
In this trial, patients at nine general hospitals in China were randomly assigned in a nonblinded fashion to receive 6 months of oral TwHF pills three times per day (69 patients), oral methotrexate once per week (69 patients), or TwHF plus methotrexate (69 patients), in addition to their stable doses of NSAIDs and oral corticosteroids, said Qian-wen Lv of the department of rheumatology and clinical immunology, Peking Union Medical College and the Chinese Academy of Medical Sciences, Beijing, and associates.
The primary efficacy measure was the proportion of patients who achieved an ACR50, or an American College of Rheumatology response of at least 50% improvement in tender and swollen joints and an improvement of at least 50% in three or more of the following: evaluator’s assessment of global health status, patient’s assessment of global health status, patient’s assessment of pain on a visual analog scale, patient’s assessment of function using the Health Assessment Questionnaire, and ESR or CRP level. This rate was 55.1% in the TwHF group, which was noninferior to the 46.4% rate in the methotrexate group. The combination of TwHF plus methotrexate was the most effective, with an ACR50 response rate of 76.8%.
A similar pattern was seen in secondary efficacy measures such as the remission rate, the ACR20 response, the ACR70 response, "good" responses on the Clinical Disease Activity Index, "good" responses on EULAR criteria, and the rate of Low Disease Activity. This pattern also persisted in a per-protocol analysis that excluded the 33 patients who withdrew from the study: The ACR50 response was 65.3% for TwHF alone, 59.2% for methotrexate alone, and 80.4% for combined TwHF plus methotrexate among patients who completed the study, the investigators said (Ann. Rheum. Dis. 2014 April 14 [doi:10.1136/annrheumdis.2013.204807]).
The main difference among the treatment groups in laboratory analyses was that TwHF monotherapy induced a much more rapid reduction in ESR than did methotrexate monotherapy. The numbers of adverse effects and severe adverse effects were comparable between TwHF and methotrexate, except that TwHF was associated with a slightly higher rate of irregular menses. This antifertility effect of the herbal remedy is well known in China, and study participants were warned about it before enrollment. It is usually reversible after treatment is discontinued, the investigators said.
This study was supported by the National Natural Science Foundation of China, the Beijing Municipal Natural Science Foundation, the Research Special Fund for Public Welfare Industry of Health, the Capital Health Research and Development of Special Fund, and the National Laboratory Special Fund. The investigators declared having no financial conflicts of interest.
A traditional Chinese herbal remedy used to treat joint pain, fever, edema, and local inflammation was found noninferior to methotrexate for controlling active rheumatoid arthritis in an open-label, randomized clinical trial involving 207 patients.
Tripterygium wilfordii Hook F (TwHF) is a relatively inexpensive remedy (about $10 per month) often used alone or in combination with methotrexate for rheumatoid arthritis (RA) in China, but it has not been assessed in a rigorous controlled study until now.
In this trial, patients at nine general hospitals in China were randomly assigned in a nonblinded fashion to receive 6 months of oral TwHF pills three times per day (69 patients), oral methotrexate once per week (69 patients), or TwHF plus methotrexate (69 patients), in addition to their stable doses of NSAIDs and oral corticosteroids, said Qian-wen Lv of the department of rheumatology and clinical immunology, Peking Union Medical College and the Chinese Academy of Medical Sciences, Beijing, and associates.
The primary efficacy measure was the proportion of patients who achieved an ACR50, or an American College of Rheumatology response of at least 50% improvement in tender and swollen joints and an improvement of at least 50% in three or more of the following: evaluator’s assessment of global health status, patient’s assessment of global health status, patient’s assessment of pain on a visual analog scale, patient’s assessment of function using the Health Assessment Questionnaire, and ESR or CRP level. This rate was 55.1% in the TwHF group, which was noninferior to the 46.4% rate in the methotrexate group. The combination of TwHF plus methotrexate was the most effective, with an ACR50 response rate of 76.8%.
A similar pattern was seen in secondary efficacy measures such as the remission rate, the ACR20 response, the ACR70 response, "good" responses on the Clinical Disease Activity Index, "good" responses on EULAR criteria, and the rate of Low Disease Activity. This pattern also persisted in a per-protocol analysis that excluded the 33 patients who withdrew from the study: The ACR50 response was 65.3% for TwHF alone, 59.2% for methotrexate alone, and 80.4% for combined TwHF plus methotrexate among patients who completed the study, the investigators said (Ann. Rheum. Dis. 2014 April 14 [doi:10.1136/annrheumdis.2013.204807]).
The main difference among the treatment groups in laboratory analyses was that TwHF monotherapy induced a much more rapid reduction in ESR than did methotrexate monotherapy. The numbers of adverse effects and severe adverse effects were comparable between TwHF and methotrexate, except that TwHF was associated with a slightly higher rate of irregular menses. This antifertility effect of the herbal remedy is well known in China, and study participants were warned about it before enrollment. It is usually reversible after treatment is discontinued, the investigators said.
This study was supported by the National Natural Science Foundation of China, the Beijing Municipal Natural Science Foundation, the Research Special Fund for Public Welfare Industry of Health, the Capital Health Research and Development of Special Fund, and the National Laboratory Special Fund. The investigators declared having no financial conflicts of interest.
A traditional Chinese herbal remedy used to treat joint pain, fever, edema, and local inflammation was found noninferior to methotrexate for controlling active rheumatoid arthritis in an open-label, randomized clinical trial involving 207 patients.
Tripterygium wilfordii Hook F (TwHF) is a relatively inexpensive remedy (about $10 per month) often used alone or in combination with methotrexate for rheumatoid arthritis (RA) in China, but it has not been assessed in a rigorous controlled study until now.
In this trial, patients at nine general hospitals in China were randomly assigned in a nonblinded fashion to receive 6 months of oral TwHF pills three times per day (69 patients), oral methotrexate once per week (69 patients), or TwHF plus methotrexate (69 patients), in addition to their stable doses of NSAIDs and oral corticosteroids, said Qian-wen Lv of the department of rheumatology and clinical immunology, Peking Union Medical College and the Chinese Academy of Medical Sciences, Beijing, and associates.
The primary efficacy measure was the proportion of patients who achieved an ACR50, or an American College of Rheumatology response of at least 50% improvement in tender and swollen joints and an improvement of at least 50% in three or more of the following: evaluator’s assessment of global health status, patient’s assessment of global health status, patient’s assessment of pain on a visual analog scale, patient’s assessment of function using the Health Assessment Questionnaire, and ESR or CRP level. This rate was 55.1% in the TwHF group, which was noninferior to the 46.4% rate in the methotrexate group. The combination of TwHF plus methotrexate was the most effective, with an ACR50 response rate of 76.8%.
A similar pattern was seen in secondary efficacy measures such as the remission rate, the ACR20 response, the ACR70 response, "good" responses on the Clinical Disease Activity Index, "good" responses on EULAR criteria, and the rate of Low Disease Activity. This pattern also persisted in a per-protocol analysis that excluded the 33 patients who withdrew from the study: The ACR50 response was 65.3% for TwHF alone, 59.2% for methotrexate alone, and 80.4% for combined TwHF plus methotrexate among patients who completed the study, the investigators said (Ann. Rheum. Dis. 2014 April 14 [doi:10.1136/annrheumdis.2013.204807]).
The main difference among the treatment groups in laboratory analyses was that TwHF monotherapy induced a much more rapid reduction in ESR than did methotrexate monotherapy. The numbers of adverse effects and severe adverse effects were comparable between TwHF and methotrexate, except that TwHF was associated with a slightly higher rate of irregular menses. This antifertility effect of the herbal remedy is well known in China, and study participants were warned about it before enrollment. It is usually reversible after treatment is discontinued, the investigators said.
This study was supported by the National Natural Science Foundation of China, the Beijing Municipal Natural Science Foundation, the Research Special Fund for Public Welfare Industry of Health, the Capital Health Research and Development of Special Fund, and the National Laboratory Special Fund. The investigators declared having no financial conflicts of interest.
FROM ANNALS OF THE RHEUMATIC DISEASES
Major Finding: The primary efficacy measure, the proportion of patients who achieved an ACR50 response, was 55.1% with TwHF alone, 46.4% with methotrexate alone, and 76.8% with combined TwHF plus methotrexate.
Data Source: A multicenter, open-label, randomized clinical trial involving 207 patients with active RA who were treated for 6 months with TwHF alone, methotrexate alone, or TwHF plus methotrexate.
Disclosures: This study was supported by the National Natural Science Foundation of China, the Beijing Municipal Natural Science Foundation, the Research Special Fund for Public Welfare Industry of Health, the Capital Health Research and Development of Special Fund, and the National Laboratory Special Fund. The investigators reported no financial conflicts of interest.
Current smoking strongly predicts rheumatoid arthritis radiographic progression
Current smoking at the time of early rheumatoid arthritis diagnosis was a strong risk factor for rapid radiographic progression in early rheumatoid arthritis, according to an analysis of patients in the multicenter, randomized SWEFOT trial.
Smoking emerged as an independent risk factor despite initial treatment with methotrexate, with or without subsequent adjunctive treatment with additional synthetic disease-modifying antirheumatic drugs or a biologic in nonresponders, wrote Dr. Saedis Saevarsdottir of the Karolinska Institute, Stockholm, and her associates in the trial’s study group.
The finding may be "perhaps not so surprising, since several older studies have previously reported an association between cigarette smoking and a more severe RA. ... [But] smoking habits have not been included in any of the recent studies on risk matrix of radiographic progression," she said.
The investigators identified 79 patients with radiographic progression among 311 patients in the trial who had radiographic data available at baseline and 1 year. Those who achieved a 28-joint disease activity score of less than 3.2 by 3-4 months remained on methotrexate (147 patients), whereas others were randomized again to add either infliximab (Remicade, 128 patients) or both sulfasalazine and hydroxychloroquine (130 patients). A total of 269 patients were included in a multivariable model (72 with radiographic progression) that included 80 who remained on methotrexate monotherapy, 94 who added infliximab, and 95 on triple therapy. The researchers defined radiographic progression as an increase in Sharp-van der Heijde score of 5 or greater after 1 year.
In the multivariable model, current smoking was the strongest baseline predictor of rapid radiographic progression (odds ratio, 2.78), and other independent predictors included erosions (OR, 2.21), C-reactive protein (CRP) level (OR, 1.49), and erythrocyte sedimentation rate (OR, 1.62) – all of which have been previously reported as baseline predictors of radiographic progression (Ann. Rheum. Dis. 2014 April 4 [doi:10.1136/annrheumdis-2013-204601]).
The investigators constructed a three-dimensional risk matrix for rapid radiographic progression based on their results with current smoking status, baseline erosions, and CRP level. At 1 year, 63% of patients who had all the predictors developed radiographic progression, compared with 12% of those without these three baseline predictors. The results were similar for men and women and within the different treatment subgroups. "Thus, the matrix may help to identify at baseline those patients at risk of radiographic progression, irrespective of which treatment is chosen on clinical grounds," the researchers wrote.
Two variables that had been significant predictors of radiographic progression in other studies and used in other risk matrices – swollen joint count and autoantibody positivity (rheumatoid factor and anti-cyclic citrullinated peptide antibodies [anti-CCP]) – were not independent predictors in the current study. In an exploratory analysis, the use of a lower Sharp-van der Heijde score cutoff of 1 or greater found both rheumatoid factor and anti-CCP positivity to be significant predictors of radiographic progression in unadjusted, but not adjusted, analyses. Tests of the current study’s risk matrix in anti-CCP negative and positive patients showed that current smokers with erosive disease had high risk for radiographic progression in both subsets of patients.
Data from the current study also performed "reasonably well" in a previous clinical trial–based risk matrix that had included autoantibody status instead of smoking status (Ann. Rheum. Dis. 2010;69:1333-7).
The study’s strength was its inclusion within a clinical trial of unselected early RA patients that reflects the common standard of care: giving methotrexate first, then adding a biologic or two additional synthetic disease-modifying antirheumatic drugs if low disease activity had not been achieved after 3-4 months of methotrexate monotherapy. Although patients’ pack-years of smoking were not available, the investigators noted that a previous study did not affect outcome when the smoking intensity was evaluated based on actual smoking status of current, past, or never smoker, "indicating that the actual smoking habits have most impact."
The study received no specific external funding. There were no relevant financial disclosures.
Current smoking at the time of early rheumatoid arthritis diagnosis was a strong risk factor for rapid radiographic progression in early rheumatoid arthritis, according to an analysis of patients in the multicenter, randomized SWEFOT trial.
Smoking emerged as an independent risk factor despite initial treatment with methotrexate, with or without subsequent adjunctive treatment with additional synthetic disease-modifying antirheumatic drugs or a biologic in nonresponders, wrote Dr. Saedis Saevarsdottir of the Karolinska Institute, Stockholm, and her associates in the trial’s study group.
The finding may be "perhaps not so surprising, since several older studies have previously reported an association between cigarette smoking and a more severe RA. ... [But] smoking habits have not been included in any of the recent studies on risk matrix of radiographic progression," she said.
The investigators identified 79 patients with radiographic progression among 311 patients in the trial who had radiographic data available at baseline and 1 year. Those who achieved a 28-joint disease activity score of less than 3.2 by 3-4 months remained on methotrexate (147 patients), whereas others were randomized again to add either infliximab (Remicade, 128 patients) or both sulfasalazine and hydroxychloroquine (130 patients). A total of 269 patients were included in a multivariable model (72 with radiographic progression) that included 80 who remained on methotrexate monotherapy, 94 who added infliximab, and 95 on triple therapy. The researchers defined radiographic progression as an increase in Sharp-van der Heijde score of 5 or greater after 1 year.
In the multivariable model, current smoking was the strongest baseline predictor of rapid radiographic progression (odds ratio, 2.78), and other independent predictors included erosions (OR, 2.21), C-reactive protein (CRP) level (OR, 1.49), and erythrocyte sedimentation rate (OR, 1.62) – all of which have been previously reported as baseline predictors of radiographic progression (Ann. Rheum. Dis. 2014 April 4 [doi:10.1136/annrheumdis-2013-204601]).
The investigators constructed a three-dimensional risk matrix for rapid radiographic progression based on their results with current smoking status, baseline erosions, and CRP level. At 1 year, 63% of patients who had all the predictors developed radiographic progression, compared with 12% of those without these three baseline predictors. The results were similar for men and women and within the different treatment subgroups. "Thus, the matrix may help to identify at baseline those patients at risk of radiographic progression, irrespective of which treatment is chosen on clinical grounds," the researchers wrote.
Two variables that had been significant predictors of radiographic progression in other studies and used in other risk matrices – swollen joint count and autoantibody positivity (rheumatoid factor and anti-cyclic citrullinated peptide antibodies [anti-CCP]) – were not independent predictors in the current study. In an exploratory analysis, the use of a lower Sharp-van der Heijde score cutoff of 1 or greater found both rheumatoid factor and anti-CCP positivity to be significant predictors of radiographic progression in unadjusted, but not adjusted, analyses. Tests of the current study’s risk matrix in anti-CCP negative and positive patients showed that current smokers with erosive disease had high risk for radiographic progression in both subsets of patients.
Data from the current study also performed "reasonably well" in a previous clinical trial–based risk matrix that had included autoantibody status instead of smoking status (Ann. Rheum. Dis. 2010;69:1333-7).
The study’s strength was its inclusion within a clinical trial of unselected early RA patients that reflects the common standard of care: giving methotrexate first, then adding a biologic or two additional synthetic disease-modifying antirheumatic drugs if low disease activity had not been achieved after 3-4 months of methotrexate monotherapy. Although patients’ pack-years of smoking were not available, the investigators noted that a previous study did not affect outcome when the smoking intensity was evaluated based on actual smoking status of current, past, or never smoker, "indicating that the actual smoking habits have most impact."
The study received no specific external funding. There were no relevant financial disclosures.
Current smoking at the time of early rheumatoid arthritis diagnosis was a strong risk factor for rapid radiographic progression in early rheumatoid arthritis, according to an analysis of patients in the multicenter, randomized SWEFOT trial.
Smoking emerged as an independent risk factor despite initial treatment with methotrexate, with or without subsequent adjunctive treatment with additional synthetic disease-modifying antirheumatic drugs or a biologic in nonresponders, wrote Dr. Saedis Saevarsdottir of the Karolinska Institute, Stockholm, and her associates in the trial’s study group.
The finding may be "perhaps not so surprising, since several older studies have previously reported an association between cigarette smoking and a more severe RA. ... [But] smoking habits have not been included in any of the recent studies on risk matrix of radiographic progression," she said.
The investigators identified 79 patients with radiographic progression among 311 patients in the trial who had radiographic data available at baseline and 1 year. Those who achieved a 28-joint disease activity score of less than 3.2 by 3-4 months remained on methotrexate (147 patients), whereas others were randomized again to add either infliximab (Remicade, 128 patients) or both sulfasalazine and hydroxychloroquine (130 patients). A total of 269 patients were included in a multivariable model (72 with radiographic progression) that included 80 who remained on methotrexate monotherapy, 94 who added infliximab, and 95 on triple therapy. The researchers defined radiographic progression as an increase in Sharp-van der Heijde score of 5 or greater after 1 year.
In the multivariable model, current smoking was the strongest baseline predictor of rapid radiographic progression (odds ratio, 2.78), and other independent predictors included erosions (OR, 2.21), C-reactive protein (CRP) level (OR, 1.49), and erythrocyte sedimentation rate (OR, 1.62) – all of which have been previously reported as baseline predictors of radiographic progression (Ann. Rheum. Dis. 2014 April 4 [doi:10.1136/annrheumdis-2013-204601]).
The investigators constructed a three-dimensional risk matrix for rapid radiographic progression based on their results with current smoking status, baseline erosions, and CRP level. At 1 year, 63% of patients who had all the predictors developed radiographic progression, compared with 12% of those without these three baseline predictors. The results were similar for men and women and within the different treatment subgroups. "Thus, the matrix may help to identify at baseline those patients at risk of radiographic progression, irrespective of which treatment is chosen on clinical grounds," the researchers wrote.
Two variables that had been significant predictors of radiographic progression in other studies and used in other risk matrices – swollen joint count and autoantibody positivity (rheumatoid factor and anti-cyclic citrullinated peptide antibodies [anti-CCP]) – were not independent predictors in the current study. In an exploratory analysis, the use of a lower Sharp-van der Heijde score cutoff of 1 or greater found both rheumatoid factor and anti-CCP positivity to be significant predictors of radiographic progression in unadjusted, but not adjusted, analyses. Tests of the current study’s risk matrix in anti-CCP negative and positive patients showed that current smokers with erosive disease had high risk for radiographic progression in both subsets of patients.
Data from the current study also performed "reasonably well" in a previous clinical trial–based risk matrix that had included autoantibody status instead of smoking status (Ann. Rheum. Dis. 2010;69:1333-7).
The study’s strength was its inclusion within a clinical trial of unselected early RA patients that reflects the common standard of care: giving methotrexate first, then adding a biologic or two additional synthetic disease-modifying antirheumatic drugs if low disease activity had not been achieved after 3-4 months of methotrexate monotherapy. Although patients’ pack-years of smoking were not available, the investigators noted that a previous study did not affect outcome when the smoking intensity was evaluated based on actual smoking status of current, past, or never smoker, "indicating that the actual smoking habits have most impact."
The study received no specific external funding. There were no relevant financial disclosures.
FROM ANNALS OF THE RHEUMATIC DISEASES
Major finding: At 1 year, 63% of patients who were current smokers and had erosive disease and high CRP levels at baseline developed radiographic progression, compared with 12% of those without the three baseline predictors.
Data source: An analysis of data from the multicenter, randomized SWEFOT study of patients with early RA.
Disclosures: The study received no specific external funding. There were no relevant financial disclosures.
Sirukumab improves rheumatoid arthritis symptoms in phase II trial
The investigational drug sirukumab significantly improved signs and symptoms of rheumatoid arthritis in a phase II trial of patients who had active disease despite receiving methotrexate.
The multicenter, randomized, placebo-controlled, double-blind study found that the drug, a human anti–interleukin (IL)-6 monoclonal antibody, led to significant improvements in American College of Rheumatology (ACR) criteria 50 and 20 responses, 28-joint count Disease Activity Score using C-reactive protein (DAS28-CRP), and scores on the Health Assessment Questionnaire-disability index (HAQ-DI) and Clinical Disease Activity Index (CDAI), compared with placebo. Of five sirukumab treatment arms in the trial, the most effective dose was 100 mg every 2 weeks.
Differences in ACR20 and ACR50 response rates between the sirukumab groups versus placebo at week 12 in the study "were generally broadly consistent with those achieved with the anti-IL-6 receptor antibodies tocilizumab and sarilumab, and the anti-IL-6 antibody clazakizumab in patients with an inadequate response to prior [methotrexate] therapy," wrote lead investigator Dr. Josef S. Smolen of the Medical University of Vienna and Hietzing Hospital, Vienna, and his colleagues.
In the two-part study, the investigators recruited adults from sites across Europe, North America, and Asia who had active RA despite methotrexate therapy. For the first part, 36 patients were randomized to receive either an injection of 100 mg of sirukumab or placebo every 2 weeks for 10 weeks and then switch to the other regimen for the next 12 weeks. For the second part, 151 patients were randomly assigned to one of five groups: injections of sirukumab at a dose of 100 mg every 2 weeks, 100 mg every 4 weeks, 50 mg every 4 weeks, 25 mg every 4 weeks for 24 weeks, and placebo every 2 weeks for 12 weeks (followed by 100 mg sirukumab every 2 weeks for 12 weeks).
In part 1, patients who received sirukumab at 100 mg every 2 weeks had a "significantly greater mean improvement from baseline to week 12 in DAS28-CRP (2.1 vs. 0.6)" than did those receiving placebo, the investigators said. Patients receiving sirukumab also had significantly greater ACR20 response rates (71% vs. 18%) and greater improvements in HAQ-DI (0.74 vs. 0.17) and CDAI (16.7 vs. 7.2) scores. At 12 weeks, 29% of patients treated with sirukumab achieved ACR50 responses, compared with 6% of patients who received placebo. Clinical responses to the 100-mg dose of sirukumab occurred as early as the second week of the study.
Patients who originally received placebo and were switched to sirukumab quickly achieved a clinical response similar to the patients who originally received the drug. Among patients switched from active drug to placebo, the clinical response lasted through the study’s end.
In part 2 of the study, only the group receiving 100 mg of sirukumab every 2 weeks achieved a statistically significant difference from placebo in ACR50 response rates at 12 weeks (the primary endpoint), occurring in 27% of the drug group versus 3% of the placebo group. ACR20 responses also were significantly higher in the 100-mg sirukumab groups versus placebo.
The authors noted that all four sirukumab groups had "a significantly greater mean improvement in DAS28-CRP at week 12 versus placebo." Six patients receiving 100 mg sirukumab every 2 weeks achieved DAS28-CRP remission versus none on placebo; patients receiving that sirukumab dose also had a greater improvement in CDAI scores (20.7 vs. 13.3).
"This study demonstrates proof-of-concept that IL-6 blockade by sirukumab provides a biologic therapy for RA," the authors wrote (Ann. Rheum. Dis. 2014 April 3 [doi:10.1136/annrheumdis-2013-205137).
Researchers observed decreases in white blood cells, neutrophils, and platelets in all sirukumab groups, generally within 2 weeks of starting treatment.
In part 1, 24 patients before crossover reported at least one adverse effect and 25 reported at least one after crossover. In part 2, 119 patients reported at least one adverse effect. Adverse effects were most often infections such as nasopharyngitis and upper respiratory tract infections. Serious adverse effects, including pneumonia, were reported by 1 patient in part 1 and 17 patients in part 2.
Phase III trials of the drug are ongoing.
This study was supported by Janssen Research & Development (a division of Johnson & Johnson). Three of the five study authors are employees of the study sponsor and own stock in Johnson & Johnson. Dr. Smolen and a coauthor have received grants and/or fees from multiple pharmaceutical companies, including Janssen.
The investigational drug sirukumab significantly improved signs and symptoms of rheumatoid arthritis in a phase II trial of patients who had active disease despite receiving methotrexate.
The multicenter, randomized, placebo-controlled, double-blind study found that the drug, a human anti–interleukin (IL)-6 monoclonal antibody, led to significant improvements in American College of Rheumatology (ACR) criteria 50 and 20 responses, 28-joint count Disease Activity Score using C-reactive protein (DAS28-CRP), and scores on the Health Assessment Questionnaire-disability index (HAQ-DI) and Clinical Disease Activity Index (CDAI), compared with placebo. Of five sirukumab treatment arms in the trial, the most effective dose was 100 mg every 2 weeks.
Differences in ACR20 and ACR50 response rates between the sirukumab groups versus placebo at week 12 in the study "were generally broadly consistent with those achieved with the anti-IL-6 receptor antibodies tocilizumab and sarilumab, and the anti-IL-6 antibody clazakizumab in patients with an inadequate response to prior [methotrexate] therapy," wrote lead investigator Dr. Josef S. Smolen of the Medical University of Vienna and Hietzing Hospital, Vienna, and his colleagues.
In the two-part study, the investigators recruited adults from sites across Europe, North America, and Asia who had active RA despite methotrexate therapy. For the first part, 36 patients were randomized to receive either an injection of 100 mg of sirukumab or placebo every 2 weeks for 10 weeks and then switch to the other regimen for the next 12 weeks. For the second part, 151 patients were randomly assigned to one of five groups: injections of sirukumab at a dose of 100 mg every 2 weeks, 100 mg every 4 weeks, 50 mg every 4 weeks, 25 mg every 4 weeks for 24 weeks, and placebo every 2 weeks for 12 weeks (followed by 100 mg sirukumab every 2 weeks for 12 weeks).
In part 1, patients who received sirukumab at 100 mg every 2 weeks had a "significantly greater mean improvement from baseline to week 12 in DAS28-CRP (2.1 vs. 0.6)" than did those receiving placebo, the investigators said. Patients receiving sirukumab also had significantly greater ACR20 response rates (71% vs. 18%) and greater improvements in HAQ-DI (0.74 vs. 0.17) and CDAI (16.7 vs. 7.2) scores. At 12 weeks, 29% of patients treated with sirukumab achieved ACR50 responses, compared with 6% of patients who received placebo. Clinical responses to the 100-mg dose of sirukumab occurred as early as the second week of the study.
Patients who originally received placebo and were switched to sirukumab quickly achieved a clinical response similar to the patients who originally received the drug. Among patients switched from active drug to placebo, the clinical response lasted through the study’s end.
In part 2 of the study, only the group receiving 100 mg of sirukumab every 2 weeks achieved a statistically significant difference from placebo in ACR50 response rates at 12 weeks (the primary endpoint), occurring in 27% of the drug group versus 3% of the placebo group. ACR20 responses also were significantly higher in the 100-mg sirukumab groups versus placebo.
The authors noted that all four sirukumab groups had "a significantly greater mean improvement in DAS28-CRP at week 12 versus placebo." Six patients receiving 100 mg sirukumab every 2 weeks achieved DAS28-CRP remission versus none on placebo; patients receiving that sirukumab dose also had a greater improvement in CDAI scores (20.7 vs. 13.3).
"This study demonstrates proof-of-concept that IL-6 blockade by sirukumab provides a biologic therapy for RA," the authors wrote (Ann. Rheum. Dis. 2014 April 3 [doi:10.1136/annrheumdis-2013-205137).
Researchers observed decreases in white blood cells, neutrophils, and platelets in all sirukumab groups, generally within 2 weeks of starting treatment.
In part 1, 24 patients before crossover reported at least one adverse effect and 25 reported at least one after crossover. In part 2, 119 patients reported at least one adverse effect. Adverse effects were most often infections such as nasopharyngitis and upper respiratory tract infections. Serious adverse effects, including pneumonia, were reported by 1 patient in part 1 and 17 patients in part 2.
Phase III trials of the drug are ongoing.
This study was supported by Janssen Research & Development (a division of Johnson & Johnson). Three of the five study authors are employees of the study sponsor and own stock in Johnson & Johnson. Dr. Smolen and a coauthor have received grants and/or fees from multiple pharmaceutical companies, including Janssen.
The investigational drug sirukumab significantly improved signs and symptoms of rheumatoid arthritis in a phase II trial of patients who had active disease despite receiving methotrexate.
The multicenter, randomized, placebo-controlled, double-blind study found that the drug, a human anti–interleukin (IL)-6 monoclonal antibody, led to significant improvements in American College of Rheumatology (ACR) criteria 50 and 20 responses, 28-joint count Disease Activity Score using C-reactive protein (DAS28-CRP), and scores on the Health Assessment Questionnaire-disability index (HAQ-DI) and Clinical Disease Activity Index (CDAI), compared with placebo. Of five sirukumab treatment arms in the trial, the most effective dose was 100 mg every 2 weeks.
Differences in ACR20 and ACR50 response rates between the sirukumab groups versus placebo at week 12 in the study "were generally broadly consistent with those achieved with the anti-IL-6 receptor antibodies tocilizumab and sarilumab, and the anti-IL-6 antibody clazakizumab in patients with an inadequate response to prior [methotrexate] therapy," wrote lead investigator Dr. Josef S. Smolen of the Medical University of Vienna and Hietzing Hospital, Vienna, and his colleagues.
In the two-part study, the investigators recruited adults from sites across Europe, North America, and Asia who had active RA despite methotrexate therapy. For the first part, 36 patients were randomized to receive either an injection of 100 mg of sirukumab or placebo every 2 weeks for 10 weeks and then switch to the other regimen for the next 12 weeks. For the second part, 151 patients were randomly assigned to one of five groups: injections of sirukumab at a dose of 100 mg every 2 weeks, 100 mg every 4 weeks, 50 mg every 4 weeks, 25 mg every 4 weeks for 24 weeks, and placebo every 2 weeks for 12 weeks (followed by 100 mg sirukumab every 2 weeks for 12 weeks).
In part 1, patients who received sirukumab at 100 mg every 2 weeks had a "significantly greater mean improvement from baseline to week 12 in DAS28-CRP (2.1 vs. 0.6)" than did those receiving placebo, the investigators said. Patients receiving sirukumab also had significantly greater ACR20 response rates (71% vs. 18%) and greater improvements in HAQ-DI (0.74 vs. 0.17) and CDAI (16.7 vs. 7.2) scores. At 12 weeks, 29% of patients treated with sirukumab achieved ACR50 responses, compared with 6% of patients who received placebo. Clinical responses to the 100-mg dose of sirukumab occurred as early as the second week of the study.
Patients who originally received placebo and were switched to sirukumab quickly achieved a clinical response similar to the patients who originally received the drug. Among patients switched from active drug to placebo, the clinical response lasted through the study’s end.
In part 2 of the study, only the group receiving 100 mg of sirukumab every 2 weeks achieved a statistically significant difference from placebo in ACR50 response rates at 12 weeks (the primary endpoint), occurring in 27% of the drug group versus 3% of the placebo group. ACR20 responses also were significantly higher in the 100-mg sirukumab groups versus placebo.
The authors noted that all four sirukumab groups had "a significantly greater mean improvement in DAS28-CRP at week 12 versus placebo." Six patients receiving 100 mg sirukumab every 2 weeks achieved DAS28-CRP remission versus none on placebo; patients receiving that sirukumab dose also had a greater improvement in CDAI scores (20.7 vs. 13.3).
"This study demonstrates proof-of-concept that IL-6 blockade by sirukumab provides a biologic therapy for RA," the authors wrote (Ann. Rheum. Dis. 2014 April 3 [doi:10.1136/annrheumdis-2013-205137).
Researchers observed decreases in white blood cells, neutrophils, and platelets in all sirukumab groups, generally within 2 weeks of starting treatment.
In part 1, 24 patients before crossover reported at least one adverse effect and 25 reported at least one after crossover. In part 2, 119 patients reported at least one adverse effect. Adverse effects were most often infections such as nasopharyngitis and upper respiratory tract infections. Serious adverse effects, including pneumonia, were reported by 1 patient in part 1 and 17 patients in part 2.
Phase III trials of the drug are ongoing.
This study was supported by Janssen Research & Development (a division of Johnson & Johnson). Three of the five study authors are employees of the study sponsor and own stock in Johnson & Johnson. Dr. Smolen and a coauthor have received grants and/or fees from multiple pharmaceutical companies, including Janssen.
FROM ANNALS OF THE RHEUMATIC DISEASES
Major finding: Sirukumab, an anti–interleukin-6 monoclonal antibody, improves the signs and symptoms of patients with active RA who are receiving methotrexate therapy.
Data source: A multicenter, randomized, placebo-controlled, double-blind phase II trial involving 187 patients.
Disclosures: This study was supported by Janssen Research & Development (a division of Johnson & Johnson). Three of the five study authors are employees of the study sponsor and own stock in Johnson & Johnson. The two lead authors have received grants and/or fees from several pharmaceutical companies, including Janssen.