User login
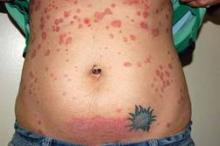
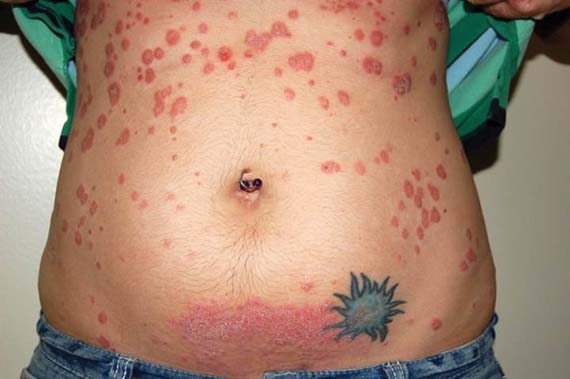
Psoriasis Flares Rapidly Postpartum
Managing psoriasis during pregnancy includes consideration for quality of life and medication safety issues, according to Dr. Alan Menter.
"Treatment of pregnant women with psoriasis should take into consideration the benefit of the therapy to her and the safety of her fetus. The full spectrum of safe and effective therapies during pregnancy needs to be reviewed," Dr. Menter said at the Women’s & Pediatric Dermatology Seminar, sponsored by the Skin Disease Education Foundation (SDEF).
"Most people present with psoriasis before age 40 years. The disease affects women and men equally, so many women with psoriasis are of reproductive age and are considering pregnancy," said Dr. Menter, chairman of the dermatology division at Baylor University Medical Center, Dallas.
"Maintaining quality of life is particularly important for females with psoriasis who are considering becoming pregnant, as well as during pregnancy and while breastfeeding."
Treatment Options
Patients with mild psoriasis can be treated safely with topical agents during pregnancy, most commonly with steroid and vitamin D preparations. For women with psoriasis unresponsive to topicals, a course of phototherapy with narrow-band UVB is safe and effective before conception, during pregnancy, and during the lactation period, Dr. Menter said.
Methotrexate, which should be stopped 3 months before conceiving, and the retinoid acitretin (Soriatane), which is contraindicated in females of childbearing potential, are pregnancy category X drugs and are not options during pregnancy.
As for cyclosporine, more than 20 years of pregnancy registry data on organ transplant recipients treated with the drug – who are on higher doses than used in psoriasis – indicate that cyclosporine is a "viable drug to be used during pregnancy if absolutely necessary," he said.
For moderate to severe psoriasis, "cyclosporine works very well at relatively low dosages, but has to be discontinued before the baby is born because it is excreted in breast milk," he added. A potential adverse effect of cyclosporine is that it may aggravate hypertension of pregnancy, and, in a small percentage of patients, it produces a pregnancy that is 3-4 weeks shorter than normal.
The biologic treatments etanercept (Enbrel), adalimumab (Humira), infliximab (Remicade), and ustekinumab (Stelara), which are all category B drugs, could interfere with normal embryonic development and affect fetal and newborn immunity. However, there are numerous pregnancy registries for both psoriasis and other inflammatory diseases, such as rheumatoid arthritis, Crohn’s disease, and ulcerative colitis, which show that biologics have had no long-term implications for the normal development of the infant immune system.
Psoriasis improves spontaneously in about 60% of women during pregnancy, although they are at an increased risk for postpartum flares. After delivery, "psoriasis can flare up very quickly and can spread from head to toe within days or weeks ... [and is] sometimes a lot worse than what they had previously," he said.
Patients should be advised to alert their physicians at the first sign of a flare after delivery. Phototherapy and topical treatments are options, but cyclosporine should not be used in breastfeeding patients. There is almost no excretion of the four injectable biologics into breast milk, which can thus be considered safe in lactating women.
Case Studies
Among the cases he presented at the conference was a 26-year-old female with a 5-year history of severe psoriasis who started treatment with cyclosporine. At 12 months, she was 90% clear and the dose was tapered. She became pregnant at 16 weeks. Cyclosporine was discontinued at 27 weeks gestation, and she remained relatively clear for the rest of her pregnancy and delivered a healthy baby at term.
She had a severe postpartum flare, with only a moderate response obtained with the reintroduction of cyclosporine after she stopped breastfeeding. She was then started on a 12-week course of the biologic agent alefacept and UVB treatment, which produced a moderate response. During the second month of alefacept treatment, she became pregnant again. The moderate response achieved with the alefacept was maintained throughout the pregnancy, even after the 12-week course of alefacept was stopped. She delivered a normal, healthy baby.
The same patient was started on infliximab for a significant flare of psoriasis a month after her second delivery. She was 95% clear after three induction infusions and was maintained on infliximab for 5 years. She became pregnant again during treatment with infliximab and had a healthy baby, with a normal delivery. She remains 90% clear to date on infliximab at a dose of 7.5 mg/kg every 8 weeks.
Another patient Dr. Menter described became pregnant while on methotrexate, and had a healthy baby despite the risk of miscarriage and fetal abnormalities with methotrexate. She stopped methotrexate treatment at week 13 of pregnancy, but developed a significant flare and started treatment with cyclosporine, which was tapered and discontinued at 8 months of pregnancy when the patient’s ustekinumab therapy was initiated. Three weeks later, she delivered a healthy baby who is "thriving" at age 3 years; her psoriasis remains 75% clear on a dose of 45 mg of ustekinumab every 12 weeks, Dr. Menter said.
Clinicians should be aware that women with psoriasis are at a slightly increased risk for spontaneous abortion and premature rupture of the membranes, he said, referring to the results of a case-control study that found an increased risk for these pregnancy complications, as well as macrosomia and induced abortions in women with psoriasis (J. Eur. Acad. Dermatol. Venereol. 2011:25;1041-7).
Dr. Menter has received research support from, been a consultant to, or served as a lecturer for Abbott, Amgen, and other companies.
SDEF and this news organization are owned by Frontline Medical Communications.
Managing psoriasis during pregnancy includes consideration for quality of life and medication safety issues, according to Dr. Alan Menter.
"Treatment of pregnant women with psoriasis should take into consideration the benefit of the therapy to her and the safety of her fetus. The full spectrum of safe and effective therapies during pregnancy needs to be reviewed," Dr. Menter said at the Women’s & Pediatric Dermatology Seminar, sponsored by the Skin Disease Education Foundation (SDEF).
"Most people present with psoriasis before age 40 years. The disease affects women and men equally, so many women with psoriasis are of reproductive age and are considering pregnancy," said Dr. Menter, chairman of the dermatology division at Baylor University Medical Center, Dallas.
"Maintaining quality of life is particularly important for females with psoriasis who are considering becoming pregnant, as well as during pregnancy and while breastfeeding."
Treatment Options
Patients with mild psoriasis can be treated safely with topical agents during pregnancy, most commonly with steroid and vitamin D preparations. For women with psoriasis unresponsive to topicals, a course of phototherapy with narrow-band UVB is safe and effective before conception, during pregnancy, and during the lactation period, Dr. Menter said.
Methotrexate, which should be stopped 3 months before conceiving, and the retinoid acitretin (Soriatane), which is contraindicated in females of childbearing potential, are pregnancy category X drugs and are not options during pregnancy.
As for cyclosporine, more than 20 years of pregnancy registry data on organ transplant recipients treated with the drug – who are on higher doses than used in psoriasis – indicate that cyclosporine is a "viable drug to be used during pregnancy if absolutely necessary," he said.
For moderate to severe psoriasis, "cyclosporine works very well at relatively low dosages, but has to be discontinued before the baby is born because it is excreted in breast milk," he added. A potential adverse effect of cyclosporine is that it may aggravate hypertension of pregnancy, and, in a small percentage of patients, it produces a pregnancy that is 3-4 weeks shorter than normal.
The biologic treatments etanercept (Enbrel), adalimumab (Humira), infliximab (Remicade), and ustekinumab (Stelara), which are all category B drugs, could interfere with normal embryonic development and affect fetal and newborn immunity. However, there are numerous pregnancy registries for both psoriasis and other inflammatory diseases, such as rheumatoid arthritis, Crohn’s disease, and ulcerative colitis, which show that biologics have had no long-term implications for the normal development of the infant immune system.
Psoriasis improves spontaneously in about 60% of women during pregnancy, although they are at an increased risk for postpartum flares. After delivery, "psoriasis can flare up very quickly and can spread from head to toe within days or weeks ... [and is] sometimes a lot worse than what they had previously," he said.
Patients should be advised to alert their physicians at the first sign of a flare after delivery. Phototherapy and topical treatments are options, but cyclosporine should not be used in breastfeeding patients. There is almost no excretion of the four injectable biologics into breast milk, which can thus be considered safe in lactating women.
Case Studies
Among the cases he presented at the conference was a 26-year-old female with a 5-year history of severe psoriasis who started treatment with cyclosporine. At 12 months, she was 90% clear and the dose was tapered. She became pregnant at 16 weeks. Cyclosporine was discontinued at 27 weeks gestation, and she remained relatively clear for the rest of her pregnancy and delivered a healthy baby at term.
She had a severe postpartum flare, with only a moderate response obtained with the reintroduction of cyclosporine after she stopped breastfeeding. She was then started on a 12-week course of the biologic agent alefacept and UVB treatment, which produced a moderate response. During the second month of alefacept treatment, she became pregnant again. The moderate response achieved with the alefacept was maintained throughout the pregnancy, even after the 12-week course of alefacept was stopped. She delivered a normal, healthy baby.
The same patient was started on infliximab for a significant flare of psoriasis a month after her second delivery. She was 95% clear after three induction infusions and was maintained on infliximab for 5 years. She became pregnant again during treatment with infliximab and had a healthy baby, with a normal delivery. She remains 90% clear to date on infliximab at a dose of 7.5 mg/kg every 8 weeks.
Another patient Dr. Menter described became pregnant while on methotrexate, and had a healthy baby despite the risk of miscarriage and fetal abnormalities with methotrexate. She stopped methotrexate treatment at week 13 of pregnancy, but developed a significant flare and started treatment with cyclosporine, which was tapered and discontinued at 8 months of pregnancy when the patient’s ustekinumab therapy was initiated. Three weeks later, she delivered a healthy baby who is "thriving" at age 3 years; her psoriasis remains 75% clear on a dose of 45 mg of ustekinumab every 12 weeks, Dr. Menter said.
Clinicians should be aware that women with psoriasis are at a slightly increased risk for spontaneous abortion and premature rupture of the membranes, he said, referring to the results of a case-control study that found an increased risk for these pregnancy complications, as well as macrosomia and induced abortions in women with psoriasis (J. Eur. Acad. Dermatol. Venereol. 2011:25;1041-7).
Dr. Menter has received research support from, been a consultant to, or served as a lecturer for Abbott, Amgen, and other companies.
SDEF and this news organization are owned by Frontline Medical Communications.
Managing psoriasis during pregnancy includes consideration for quality of life and medication safety issues, according to Dr. Alan Menter.
"Treatment of pregnant women with psoriasis should take into consideration the benefit of the therapy to her and the safety of her fetus. The full spectrum of safe and effective therapies during pregnancy needs to be reviewed," Dr. Menter said at the Women’s & Pediatric Dermatology Seminar, sponsored by the Skin Disease Education Foundation (SDEF).
"Most people present with psoriasis before age 40 years. The disease affects women and men equally, so many women with psoriasis are of reproductive age and are considering pregnancy," said Dr. Menter, chairman of the dermatology division at Baylor University Medical Center, Dallas.
"Maintaining quality of life is particularly important for females with psoriasis who are considering becoming pregnant, as well as during pregnancy and while breastfeeding."
Treatment Options
Patients with mild psoriasis can be treated safely with topical agents during pregnancy, most commonly with steroid and vitamin D preparations. For women with psoriasis unresponsive to topicals, a course of phototherapy with narrow-band UVB is safe and effective before conception, during pregnancy, and during the lactation period, Dr. Menter said.
Methotrexate, which should be stopped 3 months before conceiving, and the retinoid acitretin (Soriatane), which is contraindicated in females of childbearing potential, are pregnancy category X drugs and are not options during pregnancy.
As for cyclosporine, more than 20 years of pregnancy registry data on organ transplant recipients treated with the drug – who are on higher doses than used in psoriasis – indicate that cyclosporine is a "viable drug to be used during pregnancy if absolutely necessary," he said.
For moderate to severe psoriasis, "cyclosporine works very well at relatively low dosages, but has to be discontinued before the baby is born because it is excreted in breast milk," he added. A potential adverse effect of cyclosporine is that it may aggravate hypertension of pregnancy, and, in a small percentage of patients, it produces a pregnancy that is 3-4 weeks shorter than normal.
The biologic treatments etanercept (Enbrel), adalimumab (Humira), infliximab (Remicade), and ustekinumab (Stelara), which are all category B drugs, could interfere with normal embryonic development and affect fetal and newborn immunity. However, there are numerous pregnancy registries for both psoriasis and other inflammatory diseases, such as rheumatoid arthritis, Crohn’s disease, and ulcerative colitis, which show that biologics have had no long-term implications for the normal development of the infant immune system.
Psoriasis improves spontaneously in about 60% of women during pregnancy, although they are at an increased risk for postpartum flares. After delivery, "psoriasis can flare up very quickly and can spread from head to toe within days or weeks ... [and is] sometimes a lot worse than what they had previously," he said.
Patients should be advised to alert their physicians at the first sign of a flare after delivery. Phototherapy and topical treatments are options, but cyclosporine should not be used in breastfeeding patients. There is almost no excretion of the four injectable biologics into breast milk, which can thus be considered safe in lactating women.
Case Studies
Among the cases he presented at the conference was a 26-year-old female with a 5-year history of severe psoriasis who started treatment with cyclosporine. At 12 months, she was 90% clear and the dose was tapered. She became pregnant at 16 weeks. Cyclosporine was discontinued at 27 weeks gestation, and she remained relatively clear for the rest of her pregnancy and delivered a healthy baby at term.
She had a severe postpartum flare, with only a moderate response obtained with the reintroduction of cyclosporine after she stopped breastfeeding. She was then started on a 12-week course of the biologic agent alefacept and UVB treatment, which produced a moderate response. During the second month of alefacept treatment, she became pregnant again. The moderate response achieved with the alefacept was maintained throughout the pregnancy, even after the 12-week course of alefacept was stopped. She delivered a normal, healthy baby.
The same patient was started on infliximab for a significant flare of psoriasis a month after her second delivery. She was 95% clear after three induction infusions and was maintained on infliximab for 5 years. She became pregnant again during treatment with infliximab and had a healthy baby, with a normal delivery. She remains 90% clear to date on infliximab at a dose of 7.5 mg/kg every 8 weeks.
Another patient Dr. Menter described became pregnant while on methotrexate, and had a healthy baby despite the risk of miscarriage and fetal abnormalities with methotrexate. She stopped methotrexate treatment at week 13 of pregnancy, but developed a significant flare and started treatment with cyclosporine, which was tapered and discontinued at 8 months of pregnancy when the patient’s ustekinumab therapy was initiated. Three weeks later, she delivered a healthy baby who is "thriving" at age 3 years; her psoriasis remains 75% clear on a dose of 45 mg of ustekinumab every 12 weeks, Dr. Menter said.
Clinicians should be aware that women with psoriasis are at a slightly increased risk for spontaneous abortion and premature rupture of the membranes, he said, referring to the results of a case-control study that found an increased risk for these pregnancy complications, as well as macrosomia and induced abortions in women with psoriasis (J. Eur. Acad. Dermatol. Venereol. 2011:25;1041-7).
Dr. Menter has received research support from, been a consultant to, or served as a lecturer for Abbott, Amgen, and other companies.
SDEF and this news organization are owned by Frontline Medical Communications.
EXPERT ANALYSIS FROM THE SDEF WOMEN'S AND PEDIATRIC DERMATOLOGY SEMINAR
EULAR's Lupus Nephritis Guidelines Emphasize Early Biopsy
The European League Against Rheumatism has issued its first guidelines on management of lupus nephritis, and they advise renal biopsy at the first sign of kidney involvement, unlike the guidance issued by the American College of Rheumatology, which leaves timing of that testing up to the clinician’s judgment.
Other bright spots in EULAR’s guidelines, which it issued jointly with the European Renal Association-European Dialysis and Transplant Association (ERA-EDTA) are their position that mycophenolic acid should be the first choice for immunosuppressive therapy, the precise recommendations for steroid dosage, stratification of treatment plans according to disease severity, and explicit advice on switching therapies after one drug has failed.
The European guidelines were published in the November issue of Annals of the Rheumatic Diseases (2012;71:1771-82) and are intended for rheumatologists, nephrologists, and internists managing adult and pediatric patients with lupus nephritis. The EULAR guidelines differ in several key ways from those issued by the American College of Rheumatology (ACR) in June (Arthritis Care Res. 2012;64:797-808).
The EULAR guidelines are unequivocal in their support for renal biopsy at any sign of renal involvement, including when unexplained renal insufficiency is accompanied by normal urinary findings. The ACR guidelines, by contrast, allow for more physician latitude in determining whether to biopsy.
"This can be an emotional issue because rheumatologists do not do biopsies and may try to avoid them," said the EULAR guidelines’ lead author, rheumatologist Dr. Dimitrios T. Boumpas, who is professor of medicine and director of internal medicine/rheumatology at the University of Crete in Heraklion, Greece, in an interview. "We felt that someone should make a position statement. Biopsy is simply best care. If you’re dealing with something severe like LN [lupus nephritis] and you avoid biopsy, that’s not good medicine."
Dr. Boumpas said the EULAR task force had also moved to put mycophenolic acid in the first position as an initial immunosuppressant treatment for most cases of class III-IV LN. Low-dose intravenous cyclophosphamide in combination with steroids is also recommended for this patient group.
There are no 5-year data for mycophenolic acid as there are for cyclophosphamide, Dr. Boumpas said. But the task force that developed the guidelines weighed data on safety and efficacy and found mycophenolic acid "as the clear first choice, while at the same time recognizing the limitations of the studies," he noted.
The ACR guidelines do not recommend use of azathioprine (AZA) as induction treatment. The EULAR recommendations acknowledge that AZA has been associated with a higher risk of renal flares, and call for its use in certain patients who have no adverse clinical or histological risk factors. Patients treated with AZA need close follow-up. "This is particularly important for countries without access to MPA [mycophenolic acid]," Dr. Boumpas said.
The EULAR guidelines also recommend switching to an alternative agent when patients fail to improve in 3-4 months or do not achieve partial response after 6-12 months, or a complete response after 2 years.
"This is based on evidence from both controlled trials and observational cohort studies, which highlight the fact that immunosuppressive agents, particularly cyclophosphamide, may take up to 2 years to achieve complete renal response," Dr. George K. Bertsias, also of the University of Crete in Heraklion and the first author of the guidelines, said in an interview. "On the other hand, lack of improvement at early time points (3-6 months) is associated with adverse prognosis and should evoke discussions for treatment intensification or switch."
This is a different timetable from that described in the ACR guidelines, which advocate switching after patients fail to respond after 6 months of treatment based on the treating physician’s clinical impression.
For patients not responding to mycophenolic acid or cyclophosphamide, treatment may be switched from mycophenolic acid to cyclophosphamide or from cyclophosphamide to mycophenolic acid, according to the guidelines.
If switching fails, rituximab, a biological agent, may be given either as an add-on treatment or as monotherapy. Although randomized controlled trials have failed to demonstrate the superiority of rituximab over standard treatment in lupus nephritis, "there is culminating evidence from several uncontrolled studies and several groups worldwide that rituximab works in about half of patients with nephritis refractory to conventional immunosuppressive therapy," Dr. Boumpas said. "Since rituximab does not have adverse effects on the gonads – a significant issue in the care of young women with lupus – the committee decided to recommend it as an additional treatment resource."
The EULAR guidelines, in contrast to the ACR guidelines, contain specific dosing advice on steroids, advocating pulse steroids (500-1,000 mg of methylprednisolone daily for three doses) in combination with initial immunosuppressive therapy, followed by daily oral glucocorticoids (0.5-1.0 mg/kg per day), afterward tapering to the minimal amount necessary to control disease.
The EULAR guidelines also contain specific recommendations for patients planning pregnancy. In addition, they cover diagnosis and management of pediatric lupus nephritis, which largely follow adult recommendations. The pediatric recommendations are based on evidence in adults, and on nonrandomized evidence in children.
Work on the recommendations was funded by EULAR and the ERA-EDTA. The authors declared no conflicts of interest.
European Renal Association-European Dialysis and Transplant Association, ERA-EDTA, mycophenolic acid, immunosuppressive therapy, Annals of the Rheumatic Diseases, Dr. Dimitrios T. Boumpas,
The European League Against Rheumatism has issued its first guidelines on management of lupus nephritis, and they advise renal biopsy at the first sign of kidney involvement, unlike the guidance issued by the American College of Rheumatology, which leaves timing of that testing up to the clinician’s judgment.
Other bright spots in EULAR’s guidelines, which it issued jointly with the European Renal Association-European Dialysis and Transplant Association (ERA-EDTA) are their position that mycophenolic acid should be the first choice for immunosuppressive therapy, the precise recommendations for steroid dosage, stratification of treatment plans according to disease severity, and explicit advice on switching therapies after one drug has failed.
The European guidelines were published in the November issue of Annals of the Rheumatic Diseases (2012;71:1771-82) and are intended for rheumatologists, nephrologists, and internists managing adult and pediatric patients with lupus nephritis. The EULAR guidelines differ in several key ways from those issued by the American College of Rheumatology (ACR) in June (Arthritis Care Res. 2012;64:797-808).
The EULAR guidelines are unequivocal in their support for renal biopsy at any sign of renal involvement, including when unexplained renal insufficiency is accompanied by normal urinary findings. The ACR guidelines, by contrast, allow for more physician latitude in determining whether to biopsy.
"This can be an emotional issue because rheumatologists do not do biopsies and may try to avoid them," said the EULAR guidelines’ lead author, rheumatologist Dr. Dimitrios T. Boumpas, who is professor of medicine and director of internal medicine/rheumatology at the University of Crete in Heraklion, Greece, in an interview. "We felt that someone should make a position statement. Biopsy is simply best care. If you’re dealing with something severe like LN [lupus nephritis] and you avoid biopsy, that’s not good medicine."
Dr. Boumpas said the EULAR task force had also moved to put mycophenolic acid in the first position as an initial immunosuppressant treatment for most cases of class III-IV LN. Low-dose intravenous cyclophosphamide in combination with steroids is also recommended for this patient group.
There are no 5-year data for mycophenolic acid as there are for cyclophosphamide, Dr. Boumpas said. But the task force that developed the guidelines weighed data on safety and efficacy and found mycophenolic acid "as the clear first choice, while at the same time recognizing the limitations of the studies," he noted.
The ACR guidelines do not recommend use of azathioprine (AZA) as induction treatment. The EULAR recommendations acknowledge that AZA has been associated with a higher risk of renal flares, and call for its use in certain patients who have no adverse clinical or histological risk factors. Patients treated with AZA need close follow-up. "This is particularly important for countries without access to MPA [mycophenolic acid]," Dr. Boumpas said.
The EULAR guidelines also recommend switching to an alternative agent when patients fail to improve in 3-4 months or do not achieve partial response after 6-12 months, or a complete response after 2 years.
"This is based on evidence from both controlled trials and observational cohort studies, which highlight the fact that immunosuppressive agents, particularly cyclophosphamide, may take up to 2 years to achieve complete renal response," Dr. George K. Bertsias, also of the University of Crete in Heraklion and the first author of the guidelines, said in an interview. "On the other hand, lack of improvement at early time points (3-6 months) is associated with adverse prognosis and should evoke discussions for treatment intensification or switch."
This is a different timetable from that described in the ACR guidelines, which advocate switching after patients fail to respond after 6 months of treatment based on the treating physician’s clinical impression.
For patients not responding to mycophenolic acid or cyclophosphamide, treatment may be switched from mycophenolic acid to cyclophosphamide or from cyclophosphamide to mycophenolic acid, according to the guidelines.
If switching fails, rituximab, a biological agent, may be given either as an add-on treatment or as monotherapy. Although randomized controlled trials have failed to demonstrate the superiority of rituximab over standard treatment in lupus nephritis, "there is culminating evidence from several uncontrolled studies and several groups worldwide that rituximab works in about half of patients with nephritis refractory to conventional immunosuppressive therapy," Dr. Boumpas said. "Since rituximab does not have adverse effects on the gonads – a significant issue in the care of young women with lupus – the committee decided to recommend it as an additional treatment resource."
The EULAR guidelines, in contrast to the ACR guidelines, contain specific dosing advice on steroids, advocating pulse steroids (500-1,000 mg of methylprednisolone daily for three doses) in combination with initial immunosuppressive therapy, followed by daily oral glucocorticoids (0.5-1.0 mg/kg per day), afterward tapering to the minimal amount necessary to control disease.
The EULAR guidelines also contain specific recommendations for patients planning pregnancy. In addition, they cover diagnosis and management of pediatric lupus nephritis, which largely follow adult recommendations. The pediatric recommendations are based on evidence in adults, and on nonrandomized evidence in children.
Work on the recommendations was funded by EULAR and the ERA-EDTA. The authors declared no conflicts of interest.
The European League Against Rheumatism has issued its first guidelines on management of lupus nephritis, and they advise renal biopsy at the first sign of kidney involvement, unlike the guidance issued by the American College of Rheumatology, which leaves timing of that testing up to the clinician’s judgment.
Other bright spots in EULAR’s guidelines, which it issued jointly with the European Renal Association-European Dialysis and Transplant Association (ERA-EDTA) are their position that mycophenolic acid should be the first choice for immunosuppressive therapy, the precise recommendations for steroid dosage, stratification of treatment plans according to disease severity, and explicit advice on switching therapies after one drug has failed.
The European guidelines were published in the November issue of Annals of the Rheumatic Diseases (2012;71:1771-82) and are intended for rheumatologists, nephrologists, and internists managing adult and pediatric patients with lupus nephritis. The EULAR guidelines differ in several key ways from those issued by the American College of Rheumatology (ACR) in June (Arthritis Care Res. 2012;64:797-808).
The EULAR guidelines are unequivocal in their support for renal biopsy at any sign of renal involvement, including when unexplained renal insufficiency is accompanied by normal urinary findings. The ACR guidelines, by contrast, allow for more physician latitude in determining whether to biopsy.
"This can be an emotional issue because rheumatologists do not do biopsies and may try to avoid them," said the EULAR guidelines’ lead author, rheumatologist Dr. Dimitrios T. Boumpas, who is professor of medicine and director of internal medicine/rheumatology at the University of Crete in Heraklion, Greece, in an interview. "We felt that someone should make a position statement. Biopsy is simply best care. If you’re dealing with something severe like LN [lupus nephritis] and you avoid biopsy, that’s not good medicine."
Dr. Boumpas said the EULAR task force had also moved to put mycophenolic acid in the first position as an initial immunosuppressant treatment for most cases of class III-IV LN. Low-dose intravenous cyclophosphamide in combination with steroids is also recommended for this patient group.
There are no 5-year data for mycophenolic acid as there are for cyclophosphamide, Dr. Boumpas said. But the task force that developed the guidelines weighed data on safety and efficacy and found mycophenolic acid "as the clear first choice, while at the same time recognizing the limitations of the studies," he noted.
The ACR guidelines do not recommend use of azathioprine (AZA) as induction treatment. The EULAR recommendations acknowledge that AZA has been associated with a higher risk of renal flares, and call for its use in certain patients who have no adverse clinical or histological risk factors. Patients treated with AZA need close follow-up. "This is particularly important for countries without access to MPA [mycophenolic acid]," Dr. Boumpas said.
The EULAR guidelines also recommend switching to an alternative agent when patients fail to improve in 3-4 months or do not achieve partial response after 6-12 months, or a complete response after 2 years.
"This is based on evidence from both controlled trials and observational cohort studies, which highlight the fact that immunosuppressive agents, particularly cyclophosphamide, may take up to 2 years to achieve complete renal response," Dr. George K. Bertsias, also of the University of Crete in Heraklion and the first author of the guidelines, said in an interview. "On the other hand, lack of improvement at early time points (3-6 months) is associated with adverse prognosis and should evoke discussions for treatment intensification or switch."
This is a different timetable from that described in the ACR guidelines, which advocate switching after patients fail to respond after 6 months of treatment based on the treating physician’s clinical impression.
For patients not responding to mycophenolic acid or cyclophosphamide, treatment may be switched from mycophenolic acid to cyclophosphamide or from cyclophosphamide to mycophenolic acid, according to the guidelines.
If switching fails, rituximab, a biological agent, may be given either as an add-on treatment or as monotherapy. Although randomized controlled trials have failed to demonstrate the superiority of rituximab over standard treatment in lupus nephritis, "there is culminating evidence from several uncontrolled studies and several groups worldwide that rituximab works in about half of patients with nephritis refractory to conventional immunosuppressive therapy," Dr. Boumpas said. "Since rituximab does not have adverse effects on the gonads – a significant issue in the care of young women with lupus – the committee decided to recommend it as an additional treatment resource."
The EULAR guidelines, in contrast to the ACR guidelines, contain specific dosing advice on steroids, advocating pulse steroids (500-1,000 mg of methylprednisolone daily for three doses) in combination with initial immunosuppressive therapy, followed by daily oral glucocorticoids (0.5-1.0 mg/kg per day), afterward tapering to the minimal amount necessary to control disease.
The EULAR guidelines also contain specific recommendations for patients planning pregnancy. In addition, they cover diagnosis and management of pediatric lupus nephritis, which largely follow adult recommendations. The pediatric recommendations are based on evidence in adults, and on nonrandomized evidence in children.
Work on the recommendations was funded by EULAR and the ERA-EDTA. The authors declared no conflicts of interest.
European Renal Association-European Dialysis and Transplant Association, ERA-EDTA, mycophenolic acid, immunosuppressive therapy, Annals of the Rheumatic Diseases, Dr. Dimitrios T. Boumpas,
European Renal Association-European Dialysis and Transplant Association, ERA-EDTA, mycophenolic acid, immunosuppressive therapy, Annals of the Rheumatic Diseases, Dr. Dimitrios T. Boumpas,
FROM ANNALS OF THE RHEUMATIC DISEASES
Psoriasis Plus Diabetes Equals Heightened Vascular Risk
PRAGUE – Patients with diabetes and psoriasis are significantly more likely to develop new-onset, diabetes-related microvascular and macrovascular complications than are psoriasis-free patients with diabetes, according to the results of a large study.
"In view of this greater likelihood of developing microvascular and macrovascular complications, clinicians may wish to consider closer disease management of diabetic patients with psoriasis," said Dr. April W. Armstrong, director of clinical research and teledermatology for the department of dermatology at the University of California Davis Health System. The study is the first to examine the impact of comorbid psoriasis and diabetes – two diseases characterized by systemic inflammation – on the risk of diabetic vascular complications.
She and her coinvestigators identified 6,164 adult patients with psoriasis and diabetes in the Thomson Reuters MarketScan medical records database covering 2000-2006. A limitation of the database is its lack of information regarding how well the participants’ diabetes was controlled, Dr. Armstrong noted.
The patients were matched to an equal number of control patients (psoriasis-free patients with diabetes) based on sex, diabetes type, prior diabetic complications, diabetic complications, and a vascular complications propensity score. Then, the researchers analyzed the risk of incident diabetes-related microvascular and macrovascular complications over the subsequent 1, 3, and 5 years.
At 12 months’ follow-up, 18.3% of the patients with diabetes and psoriasis had developed new-onset diabetic microvascular complications – that is, retinopathy, neuropathy, or nephropathy – compared with 16.5% of controls. Similarly, 18.6% of patients with diabetes and psoriasis developed microvascular complications within this time frame, compared with 15.9% of controls.
Over the full 5 years of follow-up, 29.2% of the patients with diabetes and psoriasis experienced incident microvascular and 28.6% developed macrovascular complications, compared with 26.0% and 25.7% of control patients. This translated to a highly significant adjusted 14% increased relative risk of incident microvascular complications in the cohort with comorbid diabetes and psoriasis, as well as a 13% increased incidence of macrovascular complications such as MI, heart failure, or stroke, Dr. Armstrong reported at the annual congress of the European Academy of Dermatology and Venereology.
At baseline, investigators classified 73% of the 6,164 psoriasis patients who also had diabetes as having mild psoriasis based on their use of topical therapies only, while the remaining 27% had moderate to severe psoriasis defined by their use of systemic medications or phototherapy.
Next, the researchers sought to find out whether the risk of diabetic vascular complications in patients with comorbid psoriasis climbed with increasing severity of their dermatologic disease. The answer turned out to be "sort of," noted Dr. Armstrong.
That is, the adjusted 5-year risk of incident diabetic microvascular complications was 13% greater in patients with mild psoriasis than in psoriasis-free patients with diabetes, and 16% greater in those with moderate to severe psoriasis. The results were consistent with the notion that more severe psoriasis, with its greater attendant systemic inflammation, spells greater vascular risk.
However, the risk for macrovascular complications wasn’t as clear cut. While the risk of diabetic macrovascular complications was increased 16% in patients with diabetes and mild psoriasis, the relative risk in those with moderate to severe psoriasis was only 10% more than in psoriasis-free patients with diabetes – and that degree of elevation didn’t achieve statistical significance.
One plausible explanation for this discordant finding, according to Dr. Armstrong, is that the use of systemic therapies in the cohort with moderate to severe psoriasis quelled their systemic inflammation, which dampened their risk for macrovascular disease to a level similar to that seen in patients with diabetes without psoriasis, which, it must be emphasized, is still significantly greater than in the general population without diabetes.
Dr. Armstrong reported being the recipient of psoriasis research grants from Abbott Laboratories, which sponsored this study, as well as from Amgen and Janssen Pharmaceuticals.
PRAGUE – Patients with diabetes and psoriasis are significantly more likely to develop new-onset, diabetes-related microvascular and macrovascular complications than are psoriasis-free patients with diabetes, according to the results of a large study.
"In view of this greater likelihood of developing microvascular and macrovascular complications, clinicians may wish to consider closer disease management of diabetic patients with psoriasis," said Dr. April W. Armstrong, director of clinical research and teledermatology for the department of dermatology at the University of California Davis Health System. The study is the first to examine the impact of comorbid psoriasis and diabetes – two diseases characterized by systemic inflammation – on the risk of diabetic vascular complications.
She and her coinvestigators identified 6,164 adult patients with psoriasis and diabetes in the Thomson Reuters MarketScan medical records database covering 2000-2006. A limitation of the database is its lack of information regarding how well the participants’ diabetes was controlled, Dr. Armstrong noted.
The patients were matched to an equal number of control patients (psoriasis-free patients with diabetes) based on sex, diabetes type, prior diabetic complications, diabetic complications, and a vascular complications propensity score. Then, the researchers analyzed the risk of incident diabetes-related microvascular and macrovascular complications over the subsequent 1, 3, and 5 years.
At 12 months’ follow-up, 18.3% of the patients with diabetes and psoriasis had developed new-onset diabetic microvascular complications – that is, retinopathy, neuropathy, or nephropathy – compared with 16.5% of controls. Similarly, 18.6% of patients with diabetes and psoriasis developed microvascular complications within this time frame, compared with 15.9% of controls.
Over the full 5 years of follow-up, 29.2% of the patients with diabetes and psoriasis experienced incident microvascular and 28.6% developed macrovascular complications, compared with 26.0% and 25.7% of control patients. This translated to a highly significant adjusted 14% increased relative risk of incident microvascular complications in the cohort with comorbid diabetes and psoriasis, as well as a 13% increased incidence of macrovascular complications such as MI, heart failure, or stroke, Dr. Armstrong reported at the annual congress of the European Academy of Dermatology and Venereology.
At baseline, investigators classified 73% of the 6,164 psoriasis patients who also had diabetes as having mild psoriasis based on their use of topical therapies only, while the remaining 27% had moderate to severe psoriasis defined by their use of systemic medications or phototherapy.
Next, the researchers sought to find out whether the risk of diabetic vascular complications in patients with comorbid psoriasis climbed with increasing severity of their dermatologic disease. The answer turned out to be "sort of," noted Dr. Armstrong.
That is, the adjusted 5-year risk of incident diabetic microvascular complications was 13% greater in patients with mild psoriasis than in psoriasis-free patients with diabetes, and 16% greater in those with moderate to severe psoriasis. The results were consistent with the notion that more severe psoriasis, with its greater attendant systemic inflammation, spells greater vascular risk.
However, the risk for macrovascular complications wasn’t as clear cut. While the risk of diabetic macrovascular complications was increased 16% in patients with diabetes and mild psoriasis, the relative risk in those with moderate to severe psoriasis was only 10% more than in psoriasis-free patients with diabetes – and that degree of elevation didn’t achieve statistical significance.
One plausible explanation for this discordant finding, according to Dr. Armstrong, is that the use of systemic therapies in the cohort with moderate to severe psoriasis quelled their systemic inflammation, which dampened their risk for macrovascular disease to a level similar to that seen in patients with diabetes without psoriasis, which, it must be emphasized, is still significantly greater than in the general population without diabetes.
Dr. Armstrong reported being the recipient of psoriasis research grants from Abbott Laboratories, which sponsored this study, as well as from Amgen and Janssen Pharmaceuticals.
PRAGUE – Patients with diabetes and psoriasis are significantly more likely to develop new-onset, diabetes-related microvascular and macrovascular complications than are psoriasis-free patients with diabetes, according to the results of a large study.
"In view of this greater likelihood of developing microvascular and macrovascular complications, clinicians may wish to consider closer disease management of diabetic patients with psoriasis," said Dr. April W. Armstrong, director of clinical research and teledermatology for the department of dermatology at the University of California Davis Health System. The study is the first to examine the impact of comorbid psoriasis and diabetes – two diseases characterized by systemic inflammation – on the risk of diabetic vascular complications.
She and her coinvestigators identified 6,164 adult patients with psoriasis and diabetes in the Thomson Reuters MarketScan medical records database covering 2000-2006. A limitation of the database is its lack of information regarding how well the participants’ diabetes was controlled, Dr. Armstrong noted.
The patients were matched to an equal number of control patients (psoriasis-free patients with diabetes) based on sex, diabetes type, prior diabetic complications, diabetic complications, and a vascular complications propensity score. Then, the researchers analyzed the risk of incident diabetes-related microvascular and macrovascular complications over the subsequent 1, 3, and 5 years.
At 12 months’ follow-up, 18.3% of the patients with diabetes and psoriasis had developed new-onset diabetic microvascular complications – that is, retinopathy, neuropathy, or nephropathy – compared with 16.5% of controls. Similarly, 18.6% of patients with diabetes and psoriasis developed microvascular complications within this time frame, compared with 15.9% of controls.
Over the full 5 years of follow-up, 29.2% of the patients with diabetes and psoriasis experienced incident microvascular and 28.6% developed macrovascular complications, compared with 26.0% and 25.7% of control patients. This translated to a highly significant adjusted 14% increased relative risk of incident microvascular complications in the cohort with comorbid diabetes and psoriasis, as well as a 13% increased incidence of macrovascular complications such as MI, heart failure, or stroke, Dr. Armstrong reported at the annual congress of the European Academy of Dermatology and Venereology.
At baseline, investigators classified 73% of the 6,164 psoriasis patients who also had diabetes as having mild psoriasis based on their use of topical therapies only, while the remaining 27% had moderate to severe psoriasis defined by their use of systemic medications or phototherapy.
Next, the researchers sought to find out whether the risk of diabetic vascular complications in patients with comorbid psoriasis climbed with increasing severity of their dermatologic disease. The answer turned out to be "sort of," noted Dr. Armstrong.
That is, the adjusted 5-year risk of incident diabetic microvascular complications was 13% greater in patients with mild psoriasis than in psoriasis-free patients with diabetes, and 16% greater in those with moderate to severe psoriasis. The results were consistent with the notion that more severe psoriasis, with its greater attendant systemic inflammation, spells greater vascular risk.
However, the risk for macrovascular complications wasn’t as clear cut. While the risk of diabetic macrovascular complications was increased 16% in patients with diabetes and mild psoriasis, the relative risk in those with moderate to severe psoriasis was only 10% more than in psoriasis-free patients with diabetes – and that degree of elevation didn’t achieve statistical significance.
One plausible explanation for this discordant finding, according to Dr. Armstrong, is that the use of systemic therapies in the cohort with moderate to severe psoriasis quelled their systemic inflammation, which dampened their risk for macrovascular disease to a level similar to that seen in patients with diabetes without psoriasis, which, it must be emphasized, is still significantly greater than in the general population without diabetes.
Dr. Armstrong reported being the recipient of psoriasis research grants from Abbott Laboratories, which sponsored this study, as well as from Amgen and Janssen Pharmaceuticals.
AT THE ANNUAL CONGRESS OF THE EUROPEAN ACADEMY OF DERMATOLOGY AND VENEREOLOGY
Major Finding: Patients with diabetes and psoriasis had 5-year risks of new-onset microvascular and macrovascular diabetic complications that were 14% and 13% greater, respectively, than in matched psoriasis-free patients with diabetes.
Data Source: This was a retrospective study of 6,164 psoriasis patients with diabetes and an equal number of matched psoriasis-free patients with diabetes.
Disclosures: Dr. Armstrong reported being the recipient of psoriasis research grants from Abbott Laboratories, which sponsored this study, as well as from Amgen and Janssen Pharmaceuticals.
Experience Proved Rituximab Best for Vasculitis
NEWPORT BEACH, CALIF. – Rituximab is a good remission induction agent in patients with small vessel vasculitis, such as Wegener’s granulomatosis, microscopic polyangiitis, and Churg-Strauss syndrome, according to Dr. Leonard Calabrese, chair of clinical immunology and professor of medicine at the Cleveland Clinic.
It appears to be a better choice in some patients than cyclophosphamide, the traditional option, although cyclophosphamide remains the standard of care for remission induction in patients with antineutrophil cytoplasmic antibody (ANCA)–associated vasculitis, he said at Perspectives in Rheumatic Diseases 2012.
The Food and Drug Administration approved rituximab for small vessel vasculitis in 2011 based on a trial that pitted cyclophosphamide induction and azathioprine maintenance against a 6-month regimen of rituximab, with patients in both arms of the study receiving prednisone. They all had severe but not fulminant Wegener’s granulomatosis or microscopic polyangiitis; over half had renal involvement.
Of the 99 rituximab patients, 63 (64%) reached the primary end point of steroid-free remission at 6 months, compared with 52 (53%) of the 98 cyclophosphamide patients. (N. Engl. J. Med. 2010;363:221-32).
At 18 months follow-up, 36% of rituximab patients remained in remission without treatment.
"All the trends in the trial favored rituximab. Patients who came in with [new] disease had equal outcomes. If they had relapsed on [cyclophosphamide], rituximab was significantly better," Dr. Calabrese said.
Given the results, and the fact that rituximab might have fewer long-term side effects, it "is favored as the initial therapy for patients who would otherwise be offered cyclophosphamide. Clearly, rituximab is the drug of choice for [cyclophosphamide] relapsers," he said.
Also based on the results, Dr. Calabrese said he is comfortable stopping therapy and observing patients "with new-onset moderate disease that melts away" with rituximab.
But for patients with a relapsing course or who respond only partially to rituximab, "I don’t favor observation. I think repeated rituximab or an antimetabolite is in order. Most the time, I’ve been doing repeated rituximab," he said.
The optimal rituximab patient remains unclear. "There’s more work to be done on the [study] results. We are looking for predictors," he said.
Meanwhile, "cyclophosphamide and apheresis are still my go-to therapies for patients who are ventilator dependent or have a pulmonary or renal presentation," Dr. Calabrese said.
Azathioprine, methotrexate, leflunomide, and mycophenolate are among the options for step-down therapy when patients are treated traditionally with cyclophosphamide for remission, but findings from a recent study showed "there’s no doubt that azathioprine is more effective than mycophenolate for remission maintenance. Azathioprine, in those who can tolerate it, is quite good," he said (JAMA 2010;304:2381-8).
When patients don’t have life-threatening target organ involvement – no red blood–cell casts in the urine, no hypoxic lung involvement, and normal creatinines and liver functions – methotrexate alone might be the best option.
Such patients "tend to be young and have upper-airway-limited disease. These people are very good methotrexate candidates, 20-25 mg/day with high dose glucocorticoids," he said.
For mild, limited disease, "methotrexate works great," he said.
Dr. Calabrese is a consultant for Aventis, Bristol-Myers Squibb, Genentech, Janssen, and Pfizer. He is a speaker for Amgen. The meeting was held by Global Academy for Medical Education. GAME and this news organization are owned by Frontline Medical Communications.
NEWPORT BEACH, CALIF. – Rituximab is a good remission induction agent in patients with small vessel vasculitis, such as Wegener’s granulomatosis, microscopic polyangiitis, and Churg-Strauss syndrome, according to Dr. Leonard Calabrese, chair of clinical immunology and professor of medicine at the Cleveland Clinic.
It appears to be a better choice in some patients than cyclophosphamide, the traditional option, although cyclophosphamide remains the standard of care for remission induction in patients with antineutrophil cytoplasmic antibody (ANCA)–associated vasculitis, he said at Perspectives in Rheumatic Diseases 2012.
The Food and Drug Administration approved rituximab for small vessel vasculitis in 2011 based on a trial that pitted cyclophosphamide induction and azathioprine maintenance against a 6-month regimen of rituximab, with patients in both arms of the study receiving prednisone. They all had severe but not fulminant Wegener’s granulomatosis or microscopic polyangiitis; over half had renal involvement.
Of the 99 rituximab patients, 63 (64%) reached the primary end point of steroid-free remission at 6 months, compared with 52 (53%) of the 98 cyclophosphamide patients. (N. Engl. J. Med. 2010;363:221-32).
At 18 months follow-up, 36% of rituximab patients remained in remission without treatment.
"All the trends in the trial favored rituximab. Patients who came in with [new] disease had equal outcomes. If they had relapsed on [cyclophosphamide], rituximab was significantly better," Dr. Calabrese said.
Given the results, and the fact that rituximab might have fewer long-term side effects, it "is favored as the initial therapy for patients who would otherwise be offered cyclophosphamide. Clearly, rituximab is the drug of choice for [cyclophosphamide] relapsers," he said.
Also based on the results, Dr. Calabrese said he is comfortable stopping therapy and observing patients "with new-onset moderate disease that melts away" with rituximab.
But for patients with a relapsing course or who respond only partially to rituximab, "I don’t favor observation. I think repeated rituximab or an antimetabolite is in order. Most the time, I’ve been doing repeated rituximab," he said.
The optimal rituximab patient remains unclear. "There’s more work to be done on the [study] results. We are looking for predictors," he said.
Meanwhile, "cyclophosphamide and apheresis are still my go-to therapies for patients who are ventilator dependent or have a pulmonary or renal presentation," Dr. Calabrese said.
Azathioprine, methotrexate, leflunomide, and mycophenolate are among the options for step-down therapy when patients are treated traditionally with cyclophosphamide for remission, but findings from a recent study showed "there’s no doubt that azathioprine is more effective than mycophenolate for remission maintenance. Azathioprine, in those who can tolerate it, is quite good," he said (JAMA 2010;304:2381-8).
When patients don’t have life-threatening target organ involvement – no red blood–cell casts in the urine, no hypoxic lung involvement, and normal creatinines and liver functions – methotrexate alone might be the best option.
Such patients "tend to be young and have upper-airway-limited disease. These people are very good methotrexate candidates, 20-25 mg/day with high dose glucocorticoids," he said.
For mild, limited disease, "methotrexate works great," he said.
Dr. Calabrese is a consultant for Aventis, Bristol-Myers Squibb, Genentech, Janssen, and Pfizer. He is a speaker for Amgen. The meeting was held by Global Academy for Medical Education. GAME and this news organization are owned by Frontline Medical Communications.
NEWPORT BEACH, CALIF. – Rituximab is a good remission induction agent in patients with small vessel vasculitis, such as Wegener’s granulomatosis, microscopic polyangiitis, and Churg-Strauss syndrome, according to Dr. Leonard Calabrese, chair of clinical immunology and professor of medicine at the Cleveland Clinic.
It appears to be a better choice in some patients than cyclophosphamide, the traditional option, although cyclophosphamide remains the standard of care for remission induction in patients with antineutrophil cytoplasmic antibody (ANCA)–associated vasculitis, he said at Perspectives in Rheumatic Diseases 2012.
The Food and Drug Administration approved rituximab for small vessel vasculitis in 2011 based on a trial that pitted cyclophosphamide induction and azathioprine maintenance against a 6-month regimen of rituximab, with patients in both arms of the study receiving prednisone. They all had severe but not fulminant Wegener’s granulomatosis or microscopic polyangiitis; over half had renal involvement.
Of the 99 rituximab patients, 63 (64%) reached the primary end point of steroid-free remission at 6 months, compared with 52 (53%) of the 98 cyclophosphamide patients. (N. Engl. J. Med. 2010;363:221-32).
At 18 months follow-up, 36% of rituximab patients remained in remission without treatment.
"All the trends in the trial favored rituximab. Patients who came in with [new] disease had equal outcomes. If they had relapsed on [cyclophosphamide], rituximab was significantly better," Dr. Calabrese said.
Given the results, and the fact that rituximab might have fewer long-term side effects, it "is favored as the initial therapy for patients who would otherwise be offered cyclophosphamide. Clearly, rituximab is the drug of choice for [cyclophosphamide] relapsers," he said.
Also based on the results, Dr. Calabrese said he is comfortable stopping therapy and observing patients "with new-onset moderate disease that melts away" with rituximab.
But for patients with a relapsing course or who respond only partially to rituximab, "I don’t favor observation. I think repeated rituximab or an antimetabolite is in order. Most the time, I’ve been doing repeated rituximab," he said.
The optimal rituximab patient remains unclear. "There’s more work to be done on the [study] results. We are looking for predictors," he said.
Meanwhile, "cyclophosphamide and apheresis are still my go-to therapies for patients who are ventilator dependent or have a pulmonary or renal presentation," Dr. Calabrese said.
Azathioprine, methotrexate, leflunomide, and mycophenolate are among the options for step-down therapy when patients are treated traditionally with cyclophosphamide for remission, but findings from a recent study showed "there’s no doubt that azathioprine is more effective than mycophenolate for remission maintenance. Azathioprine, in those who can tolerate it, is quite good," he said (JAMA 2010;304:2381-8).
When patients don’t have life-threatening target organ involvement – no red blood–cell casts in the urine, no hypoxic lung involvement, and normal creatinines and liver functions – methotrexate alone might be the best option.
Such patients "tend to be young and have upper-airway-limited disease. These people are very good methotrexate candidates, 20-25 mg/day with high dose glucocorticoids," he said.
For mild, limited disease, "methotrexate works great," he said.
Dr. Calabrese is a consultant for Aventis, Bristol-Myers Squibb, Genentech, Janssen, and Pfizer. He is a speaker for Amgen. The meeting was held by Global Academy for Medical Education. GAME and this news organization are owned by Frontline Medical Communications.
EXPERT ANALYSIS FROM PERSPECTIVES IN RHEUMATIC DISEASES 2012
Adalimumab May Be Best Anti-TNF for Psoriasis in Obesity
NEWPORT BEACH, CALIF. – Monoclonal tumor necrosis factor inhibitors such as adalimumab are the go-to treatments for management of psoriasis in obese patients.
Some anti–tumor necrosis factor inhibitors (anti-TNFs) are good treatments for patients with a healthy weight whose psoriasis requires systemic treatment, according to Dr. Jennifer Cather, medical director of the Modern Dermatology-Aesthetics Center in Dallas.
"It would be great to be able to do a cream and light therapy, but for a lot of people that’s just not going to work. The TNF antagonists are my treatment of choice" in those cases, said Dr. Cather, also codirector of the Cutaneous Lymphoma Clinic at Baylor University Medical Center in Dallas.
But obese psoriasis patients "can have success with adalimumab even after failing or partially responding to etanercept," she said at Perspectives in Rheumatic Diseases 2012, held by Global Academy for Medical Education. GAME and this news organization are owned by Frontline Medical Communications.
Etanercept is her go-to, however, for people who aren’t overweight and "for my peripregnancy people. It seems like they can come on and off it multiple times [as needed and still] do okay." TNF inhibitors are pregnancy category B agents, but "people are reticent to use them" during pregnancy, Dr. Cather noted.
Infliximab is an option as well, but "I have all my infliximab administered by infectious disease [specialists so that patients can be tested for infection with hepatitis B or hepatitis C virus and tuberculosis], and rheumatologists assume care," she said.
Concomitant use of methotrexate can boost the efficacy of TNF inhibitors in the management of psoriasis. In addition, to find the drug that best suits the patient, various anti-TNF agents can be given a trial and stopped if there is no response, and another drug can be initiated. Dr. Cather said that she draws the line at two anti-TNF agents. If a patient has not responded to two of these agents, "usually I’m not going to do a third," she said.
"You want to make sure people are healthy when you put them on [these biologics]. I want to make sure they don’t have [infections or] cancer," both possible risks with treatment. "You have to ask women about cervical dysplasia," because immunosuppression may add fuel to a simmering neoplastic fire, she said.
New-onset, treatment-induced psoriasis is also a possibility. "Nobody really knows what to do with this. Everybody tries to treat through, but if they are getting localized palmoplantar pustular psoriasis, usually you are not going to be able to." Rotating between agents might help, as might adding methotrexate. The retinoid "acitretin works well for pustular psoriasis," Dr. Cather said.
"Remember," she added, "you’ve got to stop the picking in psoriasis." Shiny lesions are a hint that patients have picked off the scales. When they do that, "their plaques actually get thicker. A little bit of [the pain drug gabapentin], 100-300 mg at night, really helps people with this," she said.
Primary care providers may not know that psoriasis is a risk factor for cardiovascular disease and may not routinely screen psoriasis patients for other risk factors. So "when I have a new psoriasis patient, I will write their primary care doctor" and suggest they do so, she said.
Dr. Cather is a consultant, speaker, or researcher for Abbott, Amgen, Calgene, Centocor, Leo Pharma, Novartis, and Pfizer. SDEF and this news organization are owned by Frontline Medical Communications.
NEWPORT BEACH, CALIF. – Monoclonal tumor necrosis factor inhibitors such as adalimumab are the go-to treatments for management of psoriasis in obese patients.
Some anti–tumor necrosis factor inhibitors (anti-TNFs) are good treatments for patients with a healthy weight whose psoriasis requires systemic treatment, according to Dr. Jennifer Cather, medical director of the Modern Dermatology-Aesthetics Center in Dallas.
"It would be great to be able to do a cream and light therapy, but for a lot of people that’s just not going to work. The TNF antagonists are my treatment of choice" in those cases, said Dr. Cather, also codirector of the Cutaneous Lymphoma Clinic at Baylor University Medical Center in Dallas.
But obese psoriasis patients "can have success with adalimumab even after failing or partially responding to etanercept," she said at Perspectives in Rheumatic Diseases 2012, held by Global Academy for Medical Education. GAME and this news organization are owned by Frontline Medical Communications.
Etanercept is her go-to, however, for people who aren’t overweight and "for my peripregnancy people. It seems like they can come on and off it multiple times [as needed and still] do okay." TNF inhibitors are pregnancy category B agents, but "people are reticent to use them" during pregnancy, Dr. Cather noted.
Infliximab is an option as well, but "I have all my infliximab administered by infectious disease [specialists so that patients can be tested for infection with hepatitis B or hepatitis C virus and tuberculosis], and rheumatologists assume care," she said.
Concomitant use of methotrexate can boost the efficacy of TNF inhibitors in the management of psoriasis. In addition, to find the drug that best suits the patient, various anti-TNF agents can be given a trial and stopped if there is no response, and another drug can be initiated. Dr. Cather said that she draws the line at two anti-TNF agents. If a patient has not responded to two of these agents, "usually I’m not going to do a third," she said.
"You want to make sure people are healthy when you put them on [these biologics]. I want to make sure they don’t have [infections or] cancer," both possible risks with treatment. "You have to ask women about cervical dysplasia," because immunosuppression may add fuel to a simmering neoplastic fire, she said.
New-onset, treatment-induced psoriasis is also a possibility. "Nobody really knows what to do with this. Everybody tries to treat through, but if they are getting localized palmoplantar pustular psoriasis, usually you are not going to be able to." Rotating between agents might help, as might adding methotrexate. The retinoid "acitretin works well for pustular psoriasis," Dr. Cather said.
"Remember," she added, "you’ve got to stop the picking in psoriasis." Shiny lesions are a hint that patients have picked off the scales. When they do that, "their plaques actually get thicker. A little bit of [the pain drug gabapentin], 100-300 mg at night, really helps people with this," she said.
Primary care providers may not know that psoriasis is a risk factor for cardiovascular disease and may not routinely screen psoriasis patients for other risk factors. So "when I have a new psoriasis patient, I will write their primary care doctor" and suggest they do so, she said.
Dr. Cather is a consultant, speaker, or researcher for Abbott, Amgen, Calgene, Centocor, Leo Pharma, Novartis, and Pfizer. SDEF and this news organization are owned by Frontline Medical Communications.
NEWPORT BEACH, CALIF. – Monoclonal tumor necrosis factor inhibitors such as adalimumab are the go-to treatments for management of psoriasis in obese patients.
Some anti–tumor necrosis factor inhibitors (anti-TNFs) are good treatments for patients with a healthy weight whose psoriasis requires systemic treatment, according to Dr. Jennifer Cather, medical director of the Modern Dermatology-Aesthetics Center in Dallas.
"It would be great to be able to do a cream and light therapy, but for a lot of people that’s just not going to work. The TNF antagonists are my treatment of choice" in those cases, said Dr. Cather, also codirector of the Cutaneous Lymphoma Clinic at Baylor University Medical Center in Dallas.
But obese psoriasis patients "can have success with adalimumab even after failing or partially responding to etanercept," she said at Perspectives in Rheumatic Diseases 2012, held by Global Academy for Medical Education. GAME and this news organization are owned by Frontline Medical Communications.
Etanercept is her go-to, however, for people who aren’t overweight and "for my peripregnancy people. It seems like they can come on and off it multiple times [as needed and still] do okay." TNF inhibitors are pregnancy category B agents, but "people are reticent to use them" during pregnancy, Dr. Cather noted.
Infliximab is an option as well, but "I have all my infliximab administered by infectious disease [specialists so that patients can be tested for infection with hepatitis B or hepatitis C virus and tuberculosis], and rheumatologists assume care," she said.
Concomitant use of methotrexate can boost the efficacy of TNF inhibitors in the management of psoriasis. In addition, to find the drug that best suits the patient, various anti-TNF agents can be given a trial and stopped if there is no response, and another drug can be initiated. Dr. Cather said that she draws the line at two anti-TNF agents. If a patient has not responded to two of these agents, "usually I’m not going to do a third," she said.
"You want to make sure people are healthy when you put them on [these biologics]. I want to make sure they don’t have [infections or] cancer," both possible risks with treatment. "You have to ask women about cervical dysplasia," because immunosuppression may add fuel to a simmering neoplastic fire, she said.
New-onset, treatment-induced psoriasis is also a possibility. "Nobody really knows what to do with this. Everybody tries to treat through, but if they are getting localized palmoplantar pustular psoriasis, usually you are not going to be able to." Rotating between agents might help, as might adding methotrexate. The retinoid "acitretin works well for pustular psoriasis," Dr. Cather said.
"Remember," she added, "you’ve got to stop the picking in psoriasis." Shiny lesions are a hint that patients have picked off the scales. When they do that, "their plaques actually get thicker. A little bit of [the pain drug gabapentin], 100-300 mg at night, really helps people with this," she said.
Primary care providers may not know that psoriasis is a risk factor for cardiovascular disease and may not routinely screen psoriasis patients for other risk factors. So "when I have a new psoriasis patient, I will write their primary care doctor" and suggest they do so, she said.
Dr. Cather is a consultant, speaker, or researcher for Abbott, Amgen, Calgene, Centocor, Leo Pharma, Novartis, and Pfizer. SDEF and this news organization are owned by Frontline Medical Communications.
EXPERT ANALYSIS FROM PERSPECTIVES IN RHEUMATIC DISEASES 2012
Brodalumab Knocks Psoriasis Out of Park
PRAGUE – In a 48-week, phase-II study of brodalumab – a novel selective interleukin-17 inhibitor – 60% of psoriasis patients achieved a Psoriasis Area and Severity Index score of 100, reported Dr. Kim A. Papp.
There were 181 patients with moderate to severe plaque psoriasis in the multicenter, open-label study, and nearly 100% achieved at least a PASI 75 response, 80% achieved at least a PASI 90 response, and 60% achieved a PASI 100, Dr. Papp said at the annual congress of the European Academy of Dermatology and Venereology. Equally important, responses were maintained through week 48, essentially undiminished, he said.
Dr. Papp, director of research at Probity Medical Research, Waterloo, Ont., presented the results of the open-label extension of a previously published phase II, 12-week study (N. Engl. J. Med. 2012;366:1181-9). At the end of the 12-week study, participants were observed off therapy until their psoriasis relapsed, defined as a 50% loss of therapeutic benefit. At that point, they were treated with 210 mg of brodalumab administered subcutaneously every 2 weeks for 48 weeks. The peak response was achieved between weeks 8 and 12 or 16; few study dropouts occurred.
"The side effect profile was varied and included a mix of serious infections and development of atrial fibrillation and other cardiac disorders. Certainly, there were no obvious signals to suggest that we should have undue concerns going into the planned phase III program," said Dr. Papp.
Session cochair Dr. Peter van de Kerkhof was enthusiastic about the brodalumab data. "These are absolutely fabulous results. It’s really very impressive. This study gives us a lead as to where we’re going: to a new phase in biologic therapy where we get an efficacy which we couldn’t have dreamed of 10 years ago. A new bar is set," said Dr. van de Kerkhof, professor and chairman of the department of dermatology at Radboud University in Nijmegen, the Netherlands.
Brodalumab (Amgen) is a human monoclonal antibody directed against the interleukin-17A receptor as a means of quelling inflammatory cytokines. Another humanized monoclonal antibody that neutralizes interleukin-17, ixekizumab (Eli Lilly), was also the subject of a recently published, encouraging phase II study (N. Engl. J. Med. 2012;366:1190-9). Secukinumab (Novartis) is another fully-human monoclonal antibody directed against IL-17A, which has completed several successful phase II studies. These anti-IL-17 biologics are being considered as possible new therapies for other inflammatory diseases, including psoriatic arthritis and rheumatoid arthritis.
Dr. van de Kerkhof cautioned that from a safety standpoint, it will be important to scrutinize the upcoming large, definitive phase III clinical trials of these agents for evidence of an increase in infections, particularly Staphylococcus aureus and Candida albicans.
Interleukin-17A and IL-17F play roles in the normal host-immune defenses against microorganisms. Patients with genetic defects in IL-17 tend to have problems with recurrent chronic mucocutaneous candidiasis. Moreover, individuals with chronic mucocutaneous candidiasis have been shown to have antibodies to IL-17A, IL-17F, and IL-22, he noted.
The study was sponsored by Amgen. Dr. Papp reported receiving research funds from and serving as a consultant to Amgen and numerous other pharmaceutical companies. Dr. van de Kerkhof has received research funding and consultancy fees from more than a dozen pharmaceutical companies.
PRAGUE – In a 48-week, phase-II study of brodalumab – a novel selective interleukin-17 inhibitor – 60% of psoriasis patients achieved a Psoriasis Area and Severity Index score of 100, reported Dr. Kim A. Papp.
There were 181 patients with moderate to severe plaque psoriasis in the multicenter, open-label study, and nearly 100% achieved at least a PASI 75 response, 80% achieved at least a PASI 90 response, and 60% achieved a PASI 100, Dr. Papp said at the annual congress of the European Academy of Dermatology and Venereology. Equally important, responses were maintained through week 48, essentially undiminished, he said.
Dr. Papp, director of research at Probity Medical Research, Waterloo, Ont., presented the results of the open-label extension of a previously published phase II, 12-week study (N. Engl. J. Med. 2012;366:1181-9). At the end of the 12-week study, participants were observed off therapy until their psoriasis relapsed, defined as a 50% loss of therapeutic benefit. At that point, they were treated with 210 mg of brodalumab administered subcutaneously every 2 weeks for 48 weeks. The peak response was achieved between weeks 8 and 12 or 16; few study dropouts occurred.
"The side effect profile was varied and included a mix of serious infections and development of atrial fibrillation and other cardiac disorders. Certainly, there were no obvious signals to suggest that we should have undue concerns going into the planned phase III program," said Dr. Papp.
Session cochair Dr. Peter van de Kerkhof was enthusiastic about the brodalumab data. "These are absolutely fabulous results. It’s really very impressive. This study gives us a lead as to where we’re going: to a new phase in biologic therapy where we get an efficacy which we couldn’t have dreamed of 10 years ago. A new bar is set," said Dr. van de Kerkhof, professor and chairman of the department of dermatology at Radboud University in Nijmegen, the Netherlands.
Brodalumab (Amgen) is a human monoclonal antibody directed against the interleukin-17A receptor as a means of quelling inflammatory cytokines. Another humanized monoclonal antibody that neutralizes interleukin-17, ixekizumab (Eli Lilly), was also the subject of a recently published, encouraging phase II study (N. Engl. J. Med. 2012;366:1190-9). Secukinumab (Novartis) is another fully-human monoclonal antibody directed against IL-17A, which has completed several successful phase II studies. These anti-IL-17 biologics are being considered as possible new therapies for other inflammatory diseases, including psoriatic arthritis and rheumatoid arthritis.
Dr. van de Kerkhof cautioned that from a safety standpoint, it will be important to scrutinize the upcoming large, definitive phase III clinical trials of these agents for evidence of an increase in infections, particularly Staphylococcus aureus and Candida albicans.
Interleukin-17A and IL-17F play roles in the normal host-immune defenses against microorganisms. Patients with genetic defects in IL-17 tend to have problems with recurrent chronic mucocutaneous candidiasis. Moreover, individuals with chronic mucocutaneous candidiasis have been shown to have antibodies to IL-17A, IL-17F, and IL-22, he noted.
The study was sponsored by Amgen. Dr. Papp reported receiving research funds from and serving as a consultant to Amgen and numerous other pharmaceutical companies. Dr. van de Kerkhof has received research funding and consultancy fees from more than a dozen pharmaceutical companies.
PRAGUE – In a 48-week, phase-II study of brodalumab – a novel selective interleukin-17 inhibitor – 60% of psoriasis patients achieved a Psoriasis Area and Severity Index score of 100, reported Dr. Kim A. Papp.
There were 181 patients with moderate to severe plaque psoriasis in the multicenter, open-label study, and nearly 100% achieved at least a PASI 75 response, 80% achieved at least a PASI 90 response, and 60% achieved a PASI 100, Dr. Papp said at the annual congress of the European Academy of Dermatology and Venereology. Equally important, responses were maintained through week 48, essentially undiminished, he said.
Dr. Papp, director of research at Probity Medical Research, Waterloo, Ont., presented the results of the open-label extension of a previously published phase II, 12-week study (N. Engl. J. Med. 2012;366:1181-9). At the end of the 12-week study, participants were observed off therapy until their psoriasis relapsed, defined as a 50% loss of therapeutic benefit. At that point, they were treated with 210 mg of brodalumab administered subcutaneously every 2 weeks for 48 weeks. The peak response was achieved between weeks 8 and 12 or 16; few study dropouts occurred.
"The side effect profile was varied and included a mix of serious infections and development of atrial fibrillation and other cardiac disorders. Certainly, there were no obvious signals to suggest that we should have undue concerns going into the planned phase III program," said Dr. Papp.
Session cochair Dr. Peter van de Kerkhof was enthusiastic about the brodalumab data. "These are absolutely fabulous results. It’s really very impressive. This study gives us a lead as to where we’re going: to a new phase in biologic therapy where we get an efficacy which we couldn’t have dreamed of 10 years ago. A new bar is set," said Dr. van de Kerkhof, professor and chairman of the department of dermatology at Radboud University in Nijmegen, the Netherlands.
Brodalumab (Amgen) is a human monoclonal antibody directed against the interleukin-17A receptor as a means of quelling inflammatory cytokines. Another humanized monoclonal antibody that neutralizes interleukin-17, ixekizumab (Eli Lilly), was also the subject of a recently published, encouraging phase II study (N. Engl. J. Med. 2012;366:1190-9). Secukinumab (Novartis) is another fully-human monoclonal antibody directed against IL-17A, which has completed several successful phase II studies. These anti-IL-17 biologics are being considered as possible new therapies for other inflammatory diseases, including psoriatic arthritis and rheumatoid arthritis.
Dr. van de Kerkhof cautioned that from a safety standpoint, it will be important to scrutinize the upcoming large, definitive phase III clinical trials of these agents for evidence of an increase in infections, particularly Staphylococcus aureus and Candida albicans.
Interleukin-17A and IL-17F play roles in the normal host-immune defenses against microorganisms. Patients with genetic defects in IL-17 tend to have problems with recurrent chronic mucocutaneous candidiasis. Moreover, individuals with chronic mucocutaneous candidiasis have been shown to have antibodies to IL-17A, IL-17F, and IL-22, he noted.
The study was sponsored by Amgen. Dr. Papp reported receiving research funds from and serving as a consultant to Amgen and numerous other pharmaceutical companies. Dr. van de Kerkhof has received research funding and consultancy fees from more than a dozen pharmaceutical companies.
AT THE ANNUAL CONGRESS OF THE EUROPEAN ACADEMY OF DERMATOLOGY AND VENEREOLOGY
Major Finding: After 48 weeks of treatment with brodalumab, 60% of patients with moderate to severe psoriasis displayed a PASI 100 response.
Data Source: This was a multicenter, open-label study involving 181 psoriasis patients who received 210 mg of brodalumab administered subcutaneously every 2 weeks.
Disclosures: This study was sponsored by Amgen. Dr. Papp reported receiving research funds from and serving as a consultant to Amgen and numerous other pharmaceutical companies. Dr. van de Kerkhof has received research funding and consultancy fees from more than a dozen pharmaceutical companies.
Thalidomide Remains a Good Choice for Refractory Cutaneous Lupus
NEWPORT BEACH, CALIF. – Thalidomide remains a good therapeutic choice to control cutaneous lupus when steroid creams and antimalarials fail to do the job, according to Dr. Ruth Ann Vleugels.
Dr. Vleugels said that she sometimes turns to systemic therapy for cutaneous lupus to prevent the severe and disfiguring scarring the condition can cause. The presence of erythematous, scaling lesions suggest active disease amenable to treatment.
Hydroxychloroquine or chloroquine are her first choices for systemic therapy, with the option of adding quinacrine in cases when response is inadequate.
But what do you do "when patients fail to respond to antimalarials alone?" asked Dr. Vleugels, who is director of the connective tissue disease clinic at the Brigham and Women’s Hospital in Boston.
In those cases, she said she sometimes adds methotrexate, but "I keep them on their [hydroxychloroquine] because it has some photoprotective benefits and may lower the risk of developing more significant [disease] down the road."
"Our other favorite is thalidomide. We have good luck with thalidomide. It’s a really good drug to consider for patients who are quite refractory; 7 out of 10 will clear their rash," while on thalidomide, Dr. Vleugels said at the SDEF Perspectives in Rheumatic Diseases meeting, held by Global Academy for Medical Education. GAME and this news organization are owned by Frontline Medical Communications.
As with isotretinoin, another teratogen, physicians have to enroll in a registry to prescribe thalidomide, and patients have to come back to the office every month for their next prescription because there are no refills.
In addition to being a teratogen, thalidomide is a sedative and so has to be dosed at night. The drug cannot be given to people at increased risk for clotting. About 30% of people on thalidomide develop peripheral neuropathies as well. Dr. Vleugels said that she tells her patients that "if they get bilateral pins and needles sensations, they need to let me know right away. Most the time, you just stop the medicine at that point," she said.
In general, the work-up for cutaneous lupus is like that for systemic lupus erythematosus, she said. "We do a full review of systems to hit all the [American College of Rheumatology] criteria but also ask about other things such as Reynaud’s, hair loss, miscarriages, history of clotting, etc. For labs, we get at least blood work, serology, and a urinalysis."
A medication and herbal review is also critical. "Probably at least 20% of SCLE [subacute cutaneous lupus erythematosus] patients have drug-induced disease. Hydrochlorothiazide is the classic cause, but also calcium channel blockers, and multiple other agents. We are seeing a lot of terbinafine-induced SCLE," she said.
St. John’s wort, ginkgo biloba, and Echinacea are photosensitizers. "If you have a patient with skin lupus, or SLE, who you are trying to photoprotect, you’re going to have a really hard time controlling their skin disease" if they are on any of these agents or supplements, Dr. Vleugels said.
Corticosteroid creams are an option for nonsystemic treatment. For body rashes, Dr. Vleugels said that she prefers a potent agent like clobetasol or a midpotency agent such as triamcinolone.
She said that she uses a low-potency agent such as desonide for the face "except when I really want to treat someone’s face aggressively. If patients have discoid lesions that I don’t want to scar or an impressive malar rash, I’ll give them a potent topical steroid for their face, but I make sure they use it one week on, one week off," she said.
"If you write for ‘one tube,’ a patient will get 15 g," which is insufficient for body rashes. "I’ll give most of my patients with body rashes a minimum of 120 g, but more likely 240 g. You really have to get away from writing for ‘one tube,’ " she said.
Up to 20% of patients with discoid lupus develop systemic disease, a higher proportion than once thought, and systemic disease can take almost a decade to manifest (Br. J. Dermatol. 2011;164:1335-41).
"[We used to think] that if we screened patients with skin-limited disease for the first few years, they’re over the hump. Now we realize we at least have to do careful reviews of systems and baseline labs for patients for longer than the first few years," Dr. Vleugels said.
SDEF and this news organization are owned by Frontline Medical Communications. Dr. Vleugels said she has no disclosures.
NEWPORT BEACH, CALIF. – Thalidomide remains a good therapeutic choice to control cutaneous lupus when steroid creams and antimalarials fail to do the job, according to Dr. Ruth Ann Vleugels.
Dr. Vleugels said that she sometimes turns to systemic therapy for cutaneous lupus to prevent the severe and disfiguring scarring the condition can cause. The presence of erythematous, scaling lesions suggest active disease amenable to treatment.
Hydroxychloroquine or chloroquine are her first choices for systemic therapy, with the option of adding quinacrine in cases when response is inadequate.
But what do you do "when patients fail to respond to antimalarials alone?" asked Dr. Vleugels, who is director of the connective tissue disease clinic at the Brigham and Women’s Hospital in Boston.
In those cases, she said she sometimes adds methotrexate, but "I keep them on their [hydroxychloroquine] because it has some photoprotective benefits and may lower the risk of developing more significant [disease] down the road."
"Our other favorite is thalidomide. We have good luck with thalidomide. It’s a really good drug to consider for patients who are quite refractory; 7 out of 10 will clear their rash," while on thalidomide, Dr. Vleugels said at the SDEF Perspectives in Rheumatic Diseases meeting, held by Global Academy for Medical Education. GAME and this news organization are owned by Frontline Medical Communications.
As with isotretinoin, another teratogen, physicians have to enroll in a registry to prescribe thalidomide, and patients have to come back to the office every month for their next prescription because there are no refills.
In addition to being a teratogen, thalidomide is a sedative and so has to be dosed at night. The drug cannot be given to people at increased risk for clotting. About 30% of people on thalidomide develop peripheral neuropathies as well. Dr. Vleugels said that she tells her patients that "if they get bilateral pins and needles sensations, they need to let me know right away. Most the time, you just stop the medicine at that point," she said.
In general, the work-up for cutaneous lupus is like that for systemic lupus erythematosus, she said. "We do a full review of systems to hit all the [American College of Rheumatology] criteria but also ask about other things such as Reynaud’s, hair loss, miscarriages, history of clotting, etc. For labs, we get at least blood work, serology, and a urinalysis."
A medication and herbal review is also critical. "Probably at least 20% of SCLE [subacute cutaneous lupus erythematosus] patients have drug-induced disease. Hydrochlorothiazide is the classic cause, but also calcium channel blockers, and multiple other agents. We are seeing a lot of terbinafine-induced SCLE," she said.
St. John’s wort, ginkgo biloba, and Echinacea are photosensitizers. "If you have a patient with skin lupus, or SLE, who you are trying to photoprotect, you’re going to have a really hard time controlling their skin disease" if they are on any of these agents or supplements, Dr. Vleugels said.
Corticosteroid creams are an option for nonsystemic treatment. For body rashes, Dr. Vleugels said that she prefers a potent agent like clobetasol or a midpotency agent such as triamcinolone.
She said that she uses a low-potency agent such as desonide for the face "except when I really want to treat someone’s face aggressively. If patients have discoid lesions that I don’t want to scar or an impressive malar rash, I’ll give them a potent topical steroid for their face, but I make sure they use it one week on, one week off," she said.
"If you write for ‘one tube,’ a patient will get 15 g," which is insufficient for body rashes. "I’ll give most of my patients with body rashes a minimum of 120 g, but more likely 240 g. You really have to get away from writing for ‘one tube,’ " she said.
Up to 20% of patients with discoid lupus develop systemic disease, a higher proportion than once thought, and systemic disease can take almost a decade to manifest (Br. J. Dermatol. 2011;164:1335-41).
"[We used to think] that if we screened patients with skin-limited disease for the first few years, they’re over the hump. Now we realize we at least have to do careful reviews of systems and baseline labs for patients for longer than the first few years," Dr. Vleugels said.
SDEF and this news organization are owned by Frontline Medical Communications. Dr. Vleugels said she has no disclosures.
NEWPORT BEACH, CALIF. – Thalidomide remains a good therapeutic choice to control cutaneous lupus when steroid creams and antimalarials fail to do the job, according to Dr. Ruth Ann Vleugels.
Dr. Vleugels said that she sometimes turns to systemic therapy for cutaneous lupus to prevent the severe and disfiguring scarring the condition can cause. The presence of erythematous, scaling lesions suggest active disease amenable to treatment.
Hydroxychloroquine or chloroquine are her first choices for systemic therapy, with the option of adding quinacrine in cases when response is inadequate.
But what do you do "when patients fail to respond to antimalarials alone?" asked Dr. Vleugels, who is director of the connective tissue disease clinic at the Brigham and Women’s Hospital in Boston.
In those cases, she said she sometimes adds methotrexate, but "I keep them on their [hydroxychloroquine] because it has some photoprotective benefits and may lower the risk of developing more significant [disease] down the road."
"Our other favorite is thalidomide. We have good luck with thalidomide. It’s a really good drug to consider for patients who are quite refractory; 7 out of 10 will clear their rash," while on thalidomide, Dr. Vleugels said at the SDEF Perspectives in Rheumatic Diseases meeting, held by Global Academy for Medical Education. GAME and this news organization are owned by Frontline Medical Communications.
As with isotretinoin, another teratogen, physicians have to enroll in a registry to prescribe thalidomide, and patients have to come back to the office every month for their next prescription because there are no refills.
In addition to being a teratogen, thalidomide is a sedative and so has to be dosed at night. The drug cannot be given to people at increased risk for clotting. About 30% of people on thalidomide develop peripheral neuropathies as well. Dr. Vleugels said that she tells her patients that "if they get bilateral pins and needles sensations, they need to let me know right away. Most the time, you just stop the medicine at that point," she said.
In general, the work-up for cutaneous lupus is like that for systemic lupus erythematosus, she said. "We do a full review of systems to hit all the [American College of Rheumatology] criteria but also ask about other things such as Reynaud’s, hair loss, miscarriages, history of clotting, etc. For labs, we get at least blood work, serology, and a urinalysis."
A medication and herbal review is also critical. "Probably at least 20% of SCLE [subacute cutaneous lupus erythematosus] patients have drug-induced disease. Hydrochlorothiazide is the classic cause, but also calcium channel blockers, and multiple other agents. We are seeing a lot of terbinafine-induced SCLE," she said.
St. John’s wort, ginkgo biloba, and Echinacea are photosensitizers. "If you have a patient with skin lupus, or SLE, who you are trying to photoprotect, you’re going to have a really hard time controlling their skin disease" if they are on any of these agents or supplements, Dr. Vleugels said.
Corticosteroid creams are an option for nonsystemic treatment. For body rashes, Dr. Vleugels said that she prefers a potent agent like clobetasol or a midpotency agent such as triamcinolone.
She said that she uses a low-potency agent such as desonide for the face "except when I really want to treat someone’s face aggressively. If patients have discoid lesions that I don’t want to scar or an impressive malar rash, I’ll give them a potent topical steroid for their face, but I make sure they use it one week on, one week off," she said.
"If you write for ‘one tube,’ a patient will get 15 g," which is insufficient for body rashes. "I’ll give most of my patients with body rashes a minimum of 120 g, but more likely 240 g. You really have to get away from writing for ‘one tube,’ " she said.
Up to 20% of patients with discoid lupus develop systemic disease, a higher proportion than once thought, and systemic disease can take almost a decade to manifest (Br. J. Dermatol. 2011;164:1335-41).
"[We used to think] that if we screened patients with skin-limited disease for the first few years, they’re over the hump. Now we realize we at least have to do careful reviews of systems and baseline labs for patients for longer than the first few years," Dr. Vleugels said.
SDEF and this news organization are owned by Frontline Medical Communications. Dr. Vleugels said she has no disclosures.
EXPERT ANALYSIS FROM PERSPECTIVES IN RHEUMATIC DISEASES 2012
Antibiotics Helpful in Chlamydia-Induced Reactive Arthritis
Reactive arthritis that is due to Chlamydia trachomatis infection responds to antibiotic therapy. Other infectious causes of the condition do not.
So it is worth checking the synovial fluid of affected joints for evidence of chlamydia polymerase chain reaction (PCR), according to Dr. Atul Deodhar, professor of medicine at Oregon Health and Science University in Portland.
In a recent randomized trial, 6 months of rifampin plus either azithromycin or doxycycline significantly improved outcomes versus placebo in patients with chlamydia-induced reactive arthritis. Synovial fluid PCRs were positive for chlamydia in all 42 patients (Arthritis Rheum. 2010;62:1298-307).
The study "has changed my practice. I now send synovial fluid for PCR. I have found several patients" positive for chlamydia, "and we are treating them with antibiotics," Dr. Deodhar said; he also sends urine samples for chlamydia testing.
The primary end point in the study – an improvement of 20% or more in at least four of six variables such as swollen joint count – was achieved by 17 of 27 antibiotic patients (63%) but only 3 of 15 placebo patients (20%). Six patients treated with antibiotics but none of the patients in the placebo group went into complete remission during the trial. Patients on antibiotic were also more likely to clear chlamydia from their joints.
It’s a different story when reactive arthritis is triggered by gastrointestinal pathogens such as salmonella, shigella, campylobacter, and yersinia. In those cases, "avoid antibiotics," Dr. Deodhar said.
He and his colleagues found antibiotic therapy just didn’t help in a population study of 575 likely reactive arthritis cases among 6,379 people with culture-confirmed GI infections. His team confirmed reactive arthritis in 54 of the 82 (66%) subjects they were able to exam. Enthesitis was the most frequent finding; arthritis was less common (Ann. Rheum. Dis. 2008;67:1689-96).
Some patients had been given antibiotics for their GI infections, others not. It "didn’t really make any difference to patients developing or not developing reactive arthritis or the severity of it. [Antibiotics] are not going to prevent people [with dysentery] from developing reactive arthritis," Dr. Deodhar said.
They also found that the presence or absence of human leukocyte antigen B27 did not predict risk. In sporadic reactive arthritis cases, the presence of the antigen is "not actually that important in deciding if someone has or does not have reactive arthritis," he said.
Onset of reactive arthritis comes a few days to a maximum of several weeks following the inducing infection. Asymmetrical mono- or oligoarthritis of the lower extremity is the most common joint finding. Uveitis, dactylitis, and enthesitis are also possible.
Besides antibiotics for chlamydia-induced disease, sulfasalazine and tumor necrosis factor inhibitors may help with difficult cases.
Dr. Deodhar disclosed research and grant support from Abbott, Janssen, and UCB.
Reactive arthritis that is due to Chlamydia trachomatis infection responds to antibiotic therapy. Other infectious causes of the condition do not.
So it is worth checking the synovial fluid of affected joints for evidence of chlamydia polymerase chain reaction (PCR), according to Dr. Atul Deodhar, professor of medicine at Oregon Health and Science University in Portland.
In a recent randomized trial, 6 months of rifampin plus either azithromycin or doxycycline significantly improved outcomes versus placebo in patients with chlamydia-induced reactive arthritis. Synovial fluid PCRs were positive for chlamydia in all 42 patients (Arthritis Rheum. 2010;62:1298-307).
The study "has changed my practice. I now send synovial fluid for PCR. I have found several patients" positive for chlamydia, "and we are treating them with antibiotics," Dr. Deodhar said; he also sends urine samples for chlamydia testing.
The primary end point in the study – an improvement of 20% or more in at least four of six variables such as swollen joint count – was achieved by 17 of 27 antibiotic patients (63%) but only 3 of 15 placebo patients (20%). Six patients treated with antibiotics but none of the patients in the placebo group went into complete remission during the trial. Patients on antibiotic were also more likely to clear chlamydia from their joints.
It’s a different story when reactive arthritis is triggered by gastrointestinal pathogens such as salmonella, shigella, campylobacter, and yersinia. In those cases, "avoid antibiotics," Dr. Deodhar said.
He and his colleagues found antibiotic therapy just didn’t help in a population study of 575 likely reactive arthritis cases among 6,379 people with culture-confirmed GI infections. His team confirmed reactive arthritis in 54 of the 82 (66%) subjects they were able to exam. Enthesitis was the most frequent finding; arthritis was less common (Ann. Rheum. Dis. 2008;67:1689-96).
Some patients had been given antibiotics for their GI infections, others not. It "didn’t really make any difference to patients developing or not developing reactive arthritis or the severity of it. [Antibiotics] are not going to prevent people [with dysentery] from developing reactive arthritis," Dr. Deodhar said.
They also found that the presence or absence of human leukocyte antigen B27 did not predict risk. In sporadic reactive arthritis cases, the presence of the antigen is "not actually that important in deciding if someone has or does not have reactive arthritis," he said.
Onset of reactive arthritis comes a few days to a maximum of several weeks following the inducing infection. Asymmetrical mono- or oligoarthritis of the lower extremity is the most common joint finding. Uveitis, dactylitis, and enthesitis are also possible.
Besides antibiotics for chlamydia-induced disease, sulfasalazine and tumor necrosis factor inhibitors may help with difficult cases.
Dr. Deodhar disclosed research and grant support from Abbott, Janssen, and UCB.
Reactive arthritis that is due to Chlamydia trachomatis infection responds to antibiotic therapy. Other infectious causes of the condition do not.
So it is worth checking the synovial fluid of affected joints for evidence of chlamydia polymerase chain reaction (PCR), according to Dr. Atul Deodhar, professor of medicine at Oregon Health and Science University in Portland.
In a recent randomized trial, 6 months of rifampin plus either azithromycin or doxycycline significantly improved outcomes versus placebo in patients with chlamydia-induced reactive arthritis. Synovial fluid PCRs were positive for chlamydia in all 42 patients (Arthritis Rheum. 2010;62:1298-307).
The study "has changed my practice. I now send synovial fluid for PCR. I have found several patients" positive for chlamydia, "and we are treating them with antibiotics," Dr. Deodhar said; he also sends urine samples for chlamydia testing.
The primary end point in the study – an improvement of 20% or more in at least four of six variables such as swollen joint count – was achieved by 17 of 27 antibiotic patients (63%) but only 3 of 15 placebo patients (20%). Six patients treated with antibiotics but none of the patients in the placebo group went into complete remission during the trial. Patients on antibiotic were also more likely to clear chlamydia from their joints.
It’s a different story when reactive arthritis is triggered by gastrointestinal pathogens such as salmonella, shigella, campylobacter, and yersinia. In those cases, "avoid antibiotics," Dr. Deodhar said.
He and his colleagues found antibiotic therapy just didn’t help in a population study of 575 likely reactive arthritis cases among 6,379 people with culture-confirmed GI infections. His team confirmed reactive arthritis in 54 of the 82 (66%) subjects they were able to exam. Enthesitis was the most frequent finding; arthritis was less common (Ann. Rheum. Dis. 2008;67:1689-96).
Some patients had been given antibiotics for their GI infections, others not. It "didn’t really make any difference to patients developing or not developing reactive arthritis or the severity of it. [Antibiotics] are not going to prevent people [with dysentery] from developing reactive arthritis," Dr. Deodhar said.
They also found that the presence or absence of human leukocyte antigen B27 did not predict risk. In sporadic reactive arthritis cases, the presence of the antigen is "not actually that important in deciding if someone has or does not have reactive arthritis," he said.
Onset of reactive arthritis comes a few days to a maximum of several weeks following the inducing infection. Asymmetrical mono- or oligoarthritis of the lower extremity is the most common joint finding. Uveitis, dactylitis, and enthesitis are also possible.
Besides antibiotics for chlamydia-induced disease, sulfasalazine and tumor necrosis factor inhibitors may help with difficult cases.
Dr. Deodhar disclosed research and grant support from Abbott, Janssen, and UCB.
EXPERT ANALYSIS FROM PERSPECTIVES IN RHEUMATOLOGY
Using CORRONA Registry Data to Treat Rheumatic Diseases
The Consortium of Rheumatology Researchers of North America (or CORRONA) primarily manages a database, and data analysis, of rheumatic diseases. Dr. Joel Kremer, CORRONA president and CEO, offered an update on the group's work at the 2012 Perspectives of Rheumatic Diseases in Newport Beach, Calif., held by Global Academy for Medical Education. GAME and this news organization are owned by Frontline Medical Communications.
The Consortium of Rheumatology Researchers of North America (or CORRONA) primarily manages a database, and data analysis, of rheumatic diseases. Dr. Joel Kremer, CORRONA president and CEO, offered an update on the group's work at the 2012 Perspectives of Rheumatic Diseases in Newport Beach, Calif., held by Global Academy for Medical Education. GAME and this news organization are owned by Frontline Medical Communications.
The Consortium of Rheumatology Researchers of North America (or CORRONA) primarily manages a database, and data analysis, of rheumatic diseases. Dr. Joel Kremer, CORRONA president and CEO, offered an update on the group's work at the 2012 Perspectives of Rheumatic Diseases in Newport Beach, Calif., held by Global Academy for Medical Education. GAME and this news organization are owned by Frontline Medical Communications.
Severe Psoriasis Linked to Doubled Diabetes Risk
MUNICH – Patients with severe psoriasis have a twofold increased risk of developing new-onset diabetes based on a review of more than 4 million Danish children and adults, the first nationwide cohort to be evaluated for a link between the two diseases.
"Our results underline the importance of considering psoriatic patients as a high-risk population in terms of diabetes and cardiovascular risk," Mr. Usman Khalid said at the Annual Congress of the European Society of Cardiology. "Screening for diabetes and cardiovascular risk factors in patients with psoriasis is warranted," and follows existing guidelines for managing patients with psoriasis, said Mr. Khalid, a researcher in the cardiovascular research unit of Gentofte Hospital in Copenhagen.
The likely mechanism underlying the association is the inflammatory state of patients with psoriasis, he added.
"These observations are new, interesting, and important," commented Dr. Lars Rydén, professor of cardiology at the Karolinska Institute in Stockholm. Physicians should add "carefully looking for diabetes" to their existing routine screening in psoriasis patients, he said.
The study used Danish national population and patient records for more than 4.6 million Danish citizens aged 10 years or older from 1997 through the end of 2009. The researchers excluded the 97,000 people who had diabetes, psoriasis, or both at entry into the database, which left just more than 4.5 million people, of whom 52,613 developed new-onset psoriasis during follow-up, and 4,465,643 people without psoriasis who served as the reference population. The researchers defined severe psoriasis as a case that required hospitalization at least three times, or patients diagnosed with psoriatic arthritis. The cohort of children and adults with incidence psoriasis included 45,829 mild cases, and 6,784 severe cases.
The researchers tallied the number of people who developed new-onset diabetes, both among those who never had psoriasis during the study period, and among those who developed psoriasis. They identified new diabetes cases based on initiation of treatment with one or more glucose-lowering drugs. During follow-up, the number of new cases of diabetes was 3.67/1,000 person-years among those with no psoriasis, 6.93/1,000 patient-years among patients with mild psoriasis, and 9.65/1,000 patient-years among patients with severe psoriasis. The vast majority of the diabetes that developed was type 2.
Using adjustments that controlled for potential confounders at baseline, including age, sex, comorbidities, medications, and socioeconomic status, the researchers found that, compared with the people without psoriasis, those with mild psoriasis had a statistically significant 47% increased incidence of diabetes, and severe psoriasis linked with a statistically significant twofold increased risk for diabetes, Mr. Khalid reported. The median time from onset of psoriasis to the first treatment for diabetes was about 6 years.
The analysis notably focused on patients who developed diabetes following initial development of psoriasis, which provided insight into the sequence of the two diseases that had not been available in previously-reported studies, noted Dr. Ole Ahlehoff, a cardiology researcher at Gentofte Hospital and collaborator on the study.
"I suggest screening patients with psoriasis once a year for cardiovascular risk factors, including hypertension, dyslipidemia, lifestyle factors, and diabetes based on their glucose level," said Dr. Ahlehoff, who spoke about the research at a press conference. Medical groups have released guidelines that recommend annual risk assessment for patients with severe psoriasis, such as psoriatic arthritis, including the European League Against Arthritis (Ann. Rheum. Dis. 2010;69:325-31), and the Scottish Intercollegiate Guidelines Network.
Mr. Khalid and Dr. Ahlehoff said that they had no disclosures. Dr. Rydén had no relevant disclosures.
MUNICH – Patients with severe psoriasis have a twofold increased risk of developing new-onset diabetes based on a review of more than 4 million Danish children and adults, the first nationwide cohort to be evaluated for a link between the two diseases.
"Our results underline the importance of considering psoriatic patients as a high-risk population in terms of diabetes and cardiovascular risk," Mr. Usman Khalid said at the Annual Congress of the European Society of Cardiology. "Screening for diabetes and cardiovascular risk factors in patients with psoriasis is warranted," and follows existing guidelines for managing patients with psoriasis, said Mr. Khalid, a researcher in the cardiovascular research unit of Gentofte Hospital in Copenhagen.
The likely mechanism underlying the association is the inflammatory state of patients with psoriasis, he added.
"These observations are new, interesting, and important," commented Dr. Lars Rydén, professor of cardiology at the Karolinska Institute in Stockholm. Physicians should add "carefully looking for diabetes" to their existing routine screening in psoriasis patients, he said.
The study used Danish national population and patient records for more than 4.6 million Danish citizens aged 10 years or older from 1997 through the end of 2009. The researchers excluded the 97,000 people who had diabetes, psoriasis, or both at entry into the database, which left just more than 4.5 million people, of whom 52,613 developed new-onset psoriasis during follow-up, and 4,465,643 people without psoriasis who served as the reference population. The researchers defined severe psoriasis as a case that required hospitalization at least three times, or patients diagnosed with psoriatic arthritis. The cohort of children and adults with incidence psoriasis included 45,829 mild cases, and 6,784 severe cases.
The researchers tallied the number of people who developed new-onset diabetes, both among those who never had psoriasis during the study period, and among those who developed psoriasis. They identified new diabetes cases based on initiation of treatment with one or more glucose-lowering drugs. During follow-up, the number of new cases of diabetes was 3.67/1,000 person-years among those with no psoriasis, 6.93/1,000 patient-years among patients with mild psoriasis, and 9.65/1,000 patient-years among patients with severe psoriasis. The vast majority of the diabetes that developed was type 2.
Using adjustments that controlled for potential confounders at baseline, including age, sex, comorbidities, medications, and socioeconomic status, the researchers found that, compared with the people without psoriasis, those with mild psoriasis had a statistically significant 47% increased incidence of diabetes, and severe psoriasis linked with a statistically significant twofold increased risk for diabetes, Mr. Khalid reported. The median time from onset of psoriasis to the first treatment for diabetes was about 6 years.
The analysis notably focused on patients who developed diabetes following initial development of psoriasis, which provided insight into the sequence of the two diseases that had not been available in previously-reported studies, noted Dr. Ole Ahlehoff, a cardiology researcher at Gentofte Hospital and collaborator on the study.
"I suggest screening patients with psoriasis once a year for cardiovascular risk factors, including hypertension, dyslipidemia, lifestyle factors, and diabetes based on their glucose level," said Dr. Ahlehoff, who spoke about the research at a press conference. Medical groups have released guidelines that recommend annual risk assessment for patients with severe psoriasis, such as psoriatic arthritis, including the European League Against Arthritis (Ann. Rheum. Dis. 2010;69:325-31), and the Scottish Intercollegiate Guidelines Network.
Mr. Khalid and Dr. Ahlehoff said that they had no disclosures. Dr. Rydén had no relevant disclosures.
MUNICH – Patients with severe psoriasis have a twofold increased risk of developing new-onset diabetes based on a review of more than 4 million Danish children and adults, the first nationwide cohort to be evaluated for a link between the two diseases.
"Our results underline the importance of considering psoriatic patients as a high-risk population in terms of diabetes and cardiovascular risk," Mr. Usman Khalid said at the Annual Congress of the European Society of Cardiology. "Screening for diabetes and cardiovascular risk factors in patients with psoriasis is warranted," and follows existing guidelines for managing patients with psoriasis, said Mr. Khalid, a researcher in the cardiovascular research unit of Gentofte Hospital in Copenhagen.
The likely mechanism underlying the association is the inflammatory state of patients with psoriasis, he added.
"These observations are new, interesting, and important," commented Dr. Lars Rydén, professor of cardiology at the Karolinska Institute in Stockholm. Physicians should add "carefully looking for diabetes" to their existing routine screening in psoriasis patients, he said.
The study used Danish national population and patient records for more than 4.6 million Danish citizens aged 10 years or older from 1997 through the end of 2009. The researchers excluded the 97,000 people who had diabetes, psoriasis, or both at entry into the database, which left just more than 4.5 million people, of whom 52,613 developed new-onset psoriasis during follow-up, and 4,465,643 people without psoriasis who served as the reference population. The researchers defined severe psoriasis as a case that required hospitalization at least three times, or patients diagnosed with psoriatic arthritis. The cohort of children and adults with incidence psoriasis included 45,829 mild cases, and 6,784 severe cases.
The researchers tallied the number of people who developed new-onset diabetes, both among those who never had psoriasis during the study period, and among those who developed psoriasis. They identified new diabetes cases based on initiation of treatment with one or more glucose-lowering drugs. During follow-up, the number of new cases of diabetes was 3.67/1,000 person-years among those with no psoriasis, 6.93/1,000 patient-years among patients with mild psoriasis, and 9.65/1,000 patient-years among patients with severe psoriasis. The vast majority of the diabetes that developed was type 2.
Using adjustments that controlled for potential confounders at baseline, including age, sex, comorbidities, medications, and socioeconomic status, the researchers found that, compared with the people without psoriasis, those with mild psoriasis had a statistically significant 47% increased incidence of diabetes, and severe psoriasis linked with a statistically significant twofold increased risk for diabetes, Mr. Khalid reported. The median time from onset of psoriasis to the first treatment for diabetes was about 6 years.
The analysis notably focused on patients who developed diabetes following initial development of psoriasis, which provided insight into the sequence of the two diseases that had not been available in previously-reported studies, noted Dr. Ole Ahlehoff, a cardiology researcher at Gentofte Hospital and collaborator on the study.
"I suggest screening patients with psoriasis once a year for cardiovascular risk factors, including hypertension, dyslipidemia, lifestyle factors, and diabetes based on their glucose level," said Dr. Ahlehoff, who spoke about the research at a press conference. Medical groups have released guidelines that recommend annual risk assessment for patients with severe psoriasis, such as psoriatic arthritis, including the European League Against Arthritis (Ann. Rheum. Dis. 2010;69:325-31), and the Scottish Intercollegiate Guidelines Network.
Mr. Khalid and Dr. Ahlehoff said that they had no disclosures. Dr. Rydén had no relevant disclosures.
AT THE ANNUAL CONGRESS OF THE EUROPEAN SOCIETY OF CARDIOLOGY
Major Finding: New-onset, severe psoriasis doubled the risk for incident diabetes during a median of 6 years compared with people without psoriasis.
Data Source: Data came from an analysis of 4.5 million Danish children and adults followed during 1997-2009.
Disclosures: Mr. Khalid and Dr. Ahlehoff said that they had no disclosures. Dr. Rydén had no relevant disclosures.