User login
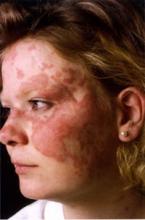
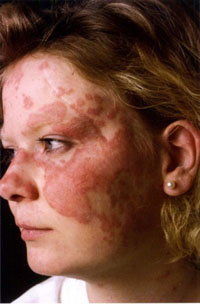
Antimalarial Response in CLE Takes Time
BOSTON – The use of adjunctive therapy and a lot of patience may go a long way to improving the response of cutaneous lupus erythematosus to antimalarial agents, according to Dr. Jennie T. Clarke.
Antimalarials are the go-to treatment for cutaneous lupus erythematosus (CLE), yet roughly a third of patients do not respond, said Dr. Clarke, who spoke at the American Academy of Dermatology’s Summer Academy Meeting.
Findings from some recent studies shed light on how to improve that dismal response rate. In one of these trials involving 128 patients with CLE, researchers found that slightly more than half of patients who initiated treatment with hydroxychloroquine monotherapy were responders (55%) (Arch. Dermatol. 2011;147:1261-7). When quinacrine was added to the treatment regimen of the nonresponders, two-thirds experienced a lessening of their disease. Improvement continued beyond 2 months in 43%.
"With antimalarials remember that patience is important. [These agents] have a slow onset. You have to give the drugs for 2-3 months before assessing for efficacy. Patients need to know this because otherwise they’re going to become frustrated and noncompliant," said Dr. Clarke of the department of dermatology at Pennsylvania State University, Hershey.
The other important lesson from the study is about combination antimalarial therapy. Quinacrine can be added to either hydroxychloroquine or chloroquine to achieve improvement in patients who don’t respond to a single antimalarial agent.
However, roughly a third of patients don’t respond to a combination of antimalarials. Some research in the last 18 months has tried to identify which patients fail antimalarial treatment and why, in order to improve subsequent treatment. Three possible reasons have been identified: dosing and compliance, disease severity, and smoking status.
Bioavailability and clearance of hydroxychloroquine seems to vary by individual, she said. In addition, noncompliance is estimated to be about 10%. In a study of 300 CLE patients, French researchers found that the median blood concentration of hydroxychloroquine correlated with response (Arch. Dermatol. 2012;148:479-84). Specifically, the median blood hydroxychloroquine concentration was significantly higher in patients with complete remission compared with those with either partial remission or treatment failure. Findings from the multivariate analysis showed that complete remission was associated with higher blood hydroxychloroquine concentrations and the absence of discoid lesions.
Concentration and response were correlated with actual rather than ideal body weight dosing. Smoking was not found to be related to concentration, and the subset of 170 patients with discoid lupus erythematosus (DLE) was less responsive to hydroxychloroquine.
Thirty patients (10%) had very low blood hydroxychloroquine concentrations (less than 200 ng/mL) and may be considered nonadherent to the treatment regimen, Dr. Clarke noted. However, additional study is needed to determine the optimal blood concentration of hydroxychloroquine and the impact of toxicity with actual weight-based dosing rather than ideal body weight dosing.
In another study, researchers assessed the clinical and pharmacogenic influences of disease severity on response to blood concentration of hydroxychloroquine (J. Invest. Dermatol. 2011;131:1981-6). They assessed 200 patients with DLE. Slightly more than a third (35%) of patients had not responded to hydroxychloroquine at 6 months. Poor response was associated with disease severity and concomitant systemic lupus erythematosus (SLE). However, response was not associated with the presence of cytochrome P450 genotype or smoking.
It has been long held medical dogma that smoking lessens patients’ response to antimalarial drugs. The impact of smoking in patients with CLE was assessed directly in another paper (Arch. Dermatol. 2012;148:317-22). The researchers included 218 patients with CLE or SLE with skin disease. They found that current smokers had more severe disease and poorer disease-related quality of life. Smokers were also more likely to receive combination antimalarial therapy. Current smokers responded better to antimalarials than past or never smokers. However, smokers responded worse than nonsmokers if antimalarials and immunomodulator/suppressives were required.
"This tells us that antimalarials can be effective for smokers, particularly in those with more mild disease. But we need to remember that smokers who don’t respond are likely going to have poorer outcomes than nonsmokers who don’t respond to antimalarials."
Dr. Clarke reported having no relevant financial conflicts.
BOSTON – The use of adjunctive therapy and a lot of patience may go a long way to improving the response of cutaneous lupus erythematosus to antimalarial agents, according to Dr. Jennie T. Clarke.
Antimalarials are the go-to treatment for cutaneous lupus erythematosus (CLE), yet roughly a third of patients do not respond, said Dr. Clarke, who spoke at the American Academy of Dermatology’s Summer Academy Meeting.
Findings from some recent studies shed light on how to improve that dismal response rate. In one of these trials involving 128 patients with CLE, researchers found that slightly more than half of patients who initiated treatment with hydroxychloroquine monotherapy were responders (55%) (Arch. Dermatol. 2011;147:1261-7). When quinacrine was added to the treatment regimen of the nonresponders, two-thirds experienced a lessening of their disease. Improvement continued beyond 2 months in 43%.
"With antimalarials remember that patience is important. [These agents] have a slow onset. You have to give the drugs for 2-3 months before assessing for efficacy. Patients need to know this because otherwise they’re going to become frustrated and noncompliant," said Dr. Clarke of the department of dermatology at Pennsylvania State University, Hershey.
The other important lesson from the study is about combination antimalarial therapy. Quinacrine can be added to either hydroxychloroquine or chloroquine to achieve improvement in patients who don’t respond to a single antimalarial agent.
However, roughly a third of patients don’t respond to a combination of antimalarials. Some research in the last 18 months has tried to identify which patients fail antimalarial treatment and why, in order to improve subsequent treatment. Three possible reasons have been identified: dosing and compliance, disease severity, and smoking status.
Bioavailability and clearance of hydroxychloroquine seems to vary by individual, she said. In addition, noncompliance is estimated to be about 10%. In a study of 300 CLE patients, French researchers found that the median blood concentration of hydroxychloroquine correlated with response (Arch. Dermatol. 2012;148:479-84). Specifically, the median blood hydroxychloroquine concentration was significantly higher in patients with complete remission compared with those with either partial remission or treatment failure. Findings from the multivariate analysis showed that complete remission was associated with higher blood hydroxychloroquine concentrations and the absence of discoid lesions.
Concentration and response were correlated with actual rather than ideal body weight dosing. Smoking was not found to be related to concentration, and the subset of 170 patients with discoid lupus erythematosus (DLE) was less responsive to hydroxychloroquine.
Thirty patients (10%) had very low blood hydroxychloroquine concentrations (less than 200 ng/mL) and may be considered nonadherent to the treatment regimen, Dr. Clarke noted. However, additional study is needed to determine the optimal blood concentration of hydroxychloroquine and the impact of toxicity with actual weight-based dosing rather than ideal body weight dosing.
In another study, researchers assessed the clinical and pharmacogenic influences of disease severity on response to blood concentration of hydroxychloroquine (J. Invest. Dermatol. 2011;131:1981-6). They assessed 200 patients with DLE. Slightly more than a third (35%) of patients had not responded to hydroxychloroquine at 6 months. Poor response was associated with disease severity and concomitant systemic lupus erythematosus (SLE). However, response was not associated with the presence of cytochrome P450 genotype or smoking.
It has been long held medical dogma that smoking lessens patients’ response to antimalarial drugs. The impact of smoking in patients with CLE was assessed directly in another paper (Arch. Dermatol. 2012;148:317-22). The researchers included 218 patients with CLE or SLE with skin disease. They found that current smokers had more severe disease and poorer disease-related quality of life. Smokers were also more likely to receive combination antimalarial therapy. Current smokers responded better to antimalarials than past or never smokers. However, smokers responded worse than nonsmokers if antimalarials and immunomodulator/suppressives were required.
"This tells us that antimalarials can be effective for smokers, particularly in those with more mild disease. But we need to remember that smokers who don’t respond are likely going to have poorer outcomes than nonsmokers who don’t respond to antimalarials."
Dr. Clarke reported having no relevant financial conflicts.
BOSTON – The use of adjunctive therapy and a lot of patience may go a long way to improving the response of cutaneous lupus erythematosus to antimalarial agents, according to Dr. Jennie T. Clarke.
Antimalarials are the go-to treatment for cutaneous lupus erythematosus (CLE), yet roughly a third of patients do not respond, said Dr. Clarke, who spoke at the American Academy of Dermatology’s Summer Academy Meeting.
Findings from some recent studies shed light on how to improve that dismal response rate. In one of these trials involving 128 patients with CLE, researchers found that slightly more than half of patients who initiated treatment with hydroxychloroquine monotherapy were responders (55%) (Arch. Dermatol. 2011;147:1261-7). When quinacrine was added to the treatment regimen of the nonresponders, two-thirds experienced a lessening of their disease. Improvement continued beyond 2 months in 43%.
"With antimalarials remember that patience is important. [These agents] have a slow onset. You have to give the drugs for 2-3 months before assessing for efficacy. Patients need to know this because otherwise they’re going to become frustrated and noncompliant," said Dr. Clarke of the department of dermatology at Pennsylvania State University, Hershey.
The other important lesson from the study is about combination antimalarial therapy. Quinacrine can be added to either hydroxychloroquine or chloroquine to achieve improvement in patients who don’t respond to a single antimalarial agent.
However, roughly a third of patients don’t respond to a combination of antimalarials. Some research in the last 18 months has tried to identify which patients fail antimalarial treatment and why, in order to improve subsequent treatment. Three possible reasons have been identified: dosing and compliance, disease severity, and smoking status.
Bioavailability and clearance of hydroxychloroquine seems to vary by individual, she said. In addition, noncompliance is estimated to be about 10%. In a study of 300 CLE patients, French researchers found that the median blood concentration of hydroxychloroquine correlated with response (Arch. Dermatol. 2012;148:479-84). Specifically, the median blood hydroxychloroquine concentration was significantly higher in patients with complete remission compared with those with either partial remission or treatment failure. Findings from the multivariate analysis showed that complete remission was associated with higher blood hydroxychloroquine concentrations and the absence of discoid lesions.
Concentration and response were correlated with actual rather than ideal body weight dosing. Smoking was not found to be related to concentration, and the subset of 170 patients with discoid lupus erythematosus (DLE) was less responsive to hydroxychloroquine.
Thirty patients (10%) had very low blood hydroxychloroquine concentrations (less than 200 ng/mL) and may be considered nonadherent to the treatment regimen, Dr. Clarke noted. However, additional study is needed to determine the optimal blood concentration of hydroxychloroquine and the impact of toxicity with actual weight-based dosing rather than ideal body weight dosing.
In another study, researchers assessed the clinical and pharmacogenic influences of disease severity on response to blood concentration of hydroxychloroquine (J. Invest. Dermatol. 2011;131:1981-6). They assessed 200 patients with DLE. Slightly more than a third (35%) of patients had not responded to hydroxychloroquine at 6 months. Poor response was associated with disease severity and concomitant systemic lupus erythematosus (SLE). However, response was not associated with the presence of cytochrome P450 genotype or smoking.
It has been long held medical dogma that smoking lessens patients’ response to antimalarial drugs. The impact of smoking in patients with CLE was assessed directly in another paper (Arch. Dermatol. 2012;148:317-22). The researchers included 218 patients with CLE or SLE with skin disease. They found that current smokers had more severe disease and poorer disease-related quality of life. Smokers were also more likely to receive combination antimalarial therapy. Current smokers responded better to antimalarials than past or never smokers. However, smokers responded worse than nonsmokers if antimalarials and immunomodulator/suppressives were required.
"This tells us that antimalarials can be effective for smokers, particularly in those with more mild disease. But we need to remember that smokers who don’t respond are likely going to have poorer outcomes than nonsmokers who don’t respond to antimalarials."
Dr. Clarke reported having no relevant financial conflicts.
EXPERT ANALYSIS FROM THE AMERICAN ACADEMY OF DERMATOLOGY'S SUMMER ACADEMY MEETING
Psoriasis Severity Linked to Tonsil Size
BOSTON – Patients with psoriasis are almost nine times more likely to have enlarged tonsils, compared with patients without psoriasis, according to the results of a small study.
"Our findings suggest that hypertrophic tonsils may be associated with a pathogenic role in psoriasis," Dr. Marianna Shvartsbeyn and her coinvestigators reported in a poster presented at the American Academy of Dermatology’s Summer Academy Meeting. But it is still too soon to know the clinical implications.
In all, 32 patients with psoriasis and 14 patients with noninflammatory skin conditions (common warts, melanoma, and nonmelanoma skin diseases) were recruited. Patients who previously underwent tonsillectomy were excluded.
Tonsils were examined by one investigator, using a 5-point standardized tonsillar hypertrophy grading scale (adopted from Am. Fam. Physician 2004;69:1147-55).
Tonsils that were entirely within the tonsillar fossa received a grade of 0. Tonsils occupying less than 25% of the lateral dimension of the oropharynx, as measured between the anterior tonsillar pillars, received a grade of 1; tonsils occupying less than 50% of the lateral dimension of the oropharynx were a 2; tonsils occupying less than 75% of the lateral dimension of the oropharynx were a 3; and tonsils occupying 75 % or more of the lateral dimension of the oropharynx received a grade of 4.
Chart reviews were conducted to collect information on patient age, sex, race, social history (tobacco, alcohol, and drug use), diagnosis of skin condition, and the duration/severity of disease, noted Dr. Shvartsbeyn and her colleague of the departments of pathology and dermatology at the New York University.
Patients with psoriasis were found to have had an odds ratio of 8.77 for having enlarged tonsils (grade 2 or greater), compared with healthy controls. Tonsillar size also was significantly larger in patients with psoriasis (mean tonsil grade, 1.78), than in control patients (mean tonsil grade, 0.86); the severity of psoriasis was positively associated with tonsil size, Dr. Shvartsbeyn and her colleagues reported.
Limited clinical data have suggested that there is an association between hypertrophic tonsils and inflammatory skin disease. Small studies have shown that among patients with psoriasis, the cutaneous lesions disappeared or improved after tonsillectomy. It is suspected that there may be a genetic predisposition that makes certain patient populations more susceptible, the researchers noted.
Histopathologic studies also point to the possible link between the robust immune response that takes place in the tonsils and the changes in the skin of patients with pustulosis palmaris et plantaris (PPP).
Histologic evaluation of tonsils obtained from patients with PPP has revealed enlargement of the secondary T nodules and atrophy of the lymph follicles, with a decrease in the number of the germinal center cells and fibrosis – changes typically seen in older tonsils. This finding provides indirect evidence of the intensely advanced stage of the immune response within the tonsils.
"Our hypothesis is that in chronic tonsillar hypertrophy, bacterial species that reside in the tonsils are released into the circulation and cause stimulation of T cells. As a result of this constant chronic stimulation, an autoreactive clone may be formed. The auto-clone may produce an antibody attacking the skin and drive inflammatory response. In some individuals, this exaggerated immune response may manifest as psoriasis," the investigators wrote.
And although there is empirical evidence "that tonsillectomy improved skin lesions in patients with psoriasis and pustulosis palmaris et plantaris in small retrospective studies, further studies are needed. ... The observed association needs validation and interventional study is needed to prove causation/contribution," Dr. Shvartsbeyn noted in an interview.
The study was supported by grants from the National Cancer Institute, the National Institute for Allergy and Infectious Diseases, and the National Institute of Dental and Craniofacial Research. The investigators did not report having any conflicts of interest.
BOSTON – Patients with psoriasis are almost nine times more likely to have enlarged tonsils, compared with patients without psoriasis, according to the results of a small study.
"Our findings suggest that hypertrophic tonsils may be associated with a pathogenic role in psoriasis," Dr. Marianna Shvartsbeyn and her coinvestigators reported in a poster presented at the American Academy of Dermatology’s Summer Academy Meeting. But it is still too soon to know the clinical implications.
In all, 32 patients with psoriasis and 14 patients with noninflammatory skin conditions (common warts, melanoma, and nonmelanoma skin diseases) were recruited. Patients who previously underwent tonsillectomy were excluded.
Tonsils were examined by one investigator, using a 5-point standardized tonsillar hypertrophy grading scale (adopted from Am. Fam. Physician 2004;69:1147-55).
Tonsils that were entirely within the tonsillar fossa received a grade of 0. Tonsils occupying less than 25% of the lateral dimension of the oropharynx, as measured between the anterior tonsillar pillars, received a grade of 1; tonsils occupying less than 50% of the lateral dimension of the oropharynx were a 2; tonsils occupying less than 75% of the lateral dimension of the oropharynx were a 3; and tonsils occupying 75 % or more of the lateral dimension of the oropharynx received a grade of 4.
Chart reviews were conducted to collect information on patient age, sex, race, social history (tobacco, alcohol, and drug use), diagnosis of skin condition, and the duration/severity of disease, noted Dr. Shvartsbeyn and her colleague of the departments of pathology and dermatology at the New York University.
Patients with psoriasis were found to have had an odds ratio of 8.77 for having enlarged tonsils (grade 2 or greater), compared with healthy controls. Tonsillar size also was significantly larger in patients with psoriasis (mean tonsil grade, 1.78), than in control patients (mean tonsil grade, 0.86); the severity of psoriasis was positively associated with tonsil size, Dr. Shvartsbeyn and her colleagues reported.
Limited clinical data have suggested that there is an association between hypertrophic tonsils and inflammatory skin disease. Small studies have shown that among patients with psoriasis, the cutaneous lesions disappeared or improved after tonsillectomy. It is suspected that there may be a genetic predisposition that makes certain patient populations more susceptible, the researchers noted.
Histopathologic studies also point to the possible link between the robust immune response that takes place in the tonsils and the changes in the skin of patients with pustulosis palmaris et plantaris (PPP).
Histologic evaluation of tonsils obtained from patients with PPP has revealed enlargement of the secondary T nodules and atrophy of the lymph follicles, with a decrease in the number of the germinal center cells and fibrosis – changes typically seen in older tonsils. This finding provides indirect evidence of the intensely advanced stage of the immune response within the tonsils.
"Our hypothesis is that in chronic tonsillar hypertrophy, bacterial species that reside in the tonsils are released into the circulation and cause stimulation of T cells. As a result of this constant chronic stimulation, an autoreactive clone may be formed. The auto-clone may produce an antibody attacking the skin and drive inflammatory response. In some individuals, this exaggerated immune response may manifest as psoriasis," the investigators wrote.
And although there is empirical evidence "that tonsillectomy improved skin lesions in patients with psoriasis and pustulosis palmaris et plantaris in small retrospective studies, further studies are needed. ... The observed association needs validation and interventional study is needed to prove causation/contribution," Dr. Shvartsbeyn noted in an interview.
The study was supported by grants from the National Cancer Institute, the National Institute for Allergy and Infectious Diseases, and the National Institute of Dental and Craniofacial Research. The investigators did not report having any conflicts of interest.
BOSTON – Patients with psoriasis are almost nine times more likely to have enlarged tonsils, compared with patients without psoriasis, according to the results of a small study.
"Our findings suggest that hypertrophic tonsils may be associated with a pathogenic role in psoriasis," Dr. Marianna Shvartsbeyn and her coinvestigators reported in a poster presented at the American Academy of Dermatology’s Summer Academy Meeting. But it is still too soon to know the clinical implications.
In all, 32 patients with psoriasis and 14 patients with noninflammatory skin conditions (common warts, melanoma, and nonmelanoma skin diseases) were recruited. Patients who previously underwent tonsillectomy were excluded.
Tonsils were examined by one investigator, using a 5-point standardized tonsillar hypertrophy grading scale (adopted from Am. Fam. Physician 2004;69:1147-55).
Tonsils that were entirely within the tonsillar fossa received a grade of 0. Tonsils occupying less than 25% of the lateral dimension of the oropharynx, as measured between the anterior tonsillar pillars, received a grade of 1; tonsils occupying less than 50% of the lateral dimension of the oropharynx were a 2; tonsils occupying less than 75% of the lateral dimension of the oropharynx were a 3; and tonsils occupying 75 % or more of the lateral dimension of the oropharynx received a grade of 4.
Chart reviews were conducted to collect information on patient age, sex, race, social history (tobacco, alcohol, and drug use), diagnosis of skin condition, and the duration/severity of disease, noted Dr. Shvartsbeyn and her colleague of the departments of pathology and dermatology at the New York University.
Patients with psoriasis were found to have had an odds ratio of 8.77 for having enlarged tonsils (grade 2 or greater), compared with healthy controls. Tonsillar size also was significantly larger in patients with psoriasis (mean tonsil grade, 1.78), than in control patients (mean tonsil grade, 0.86); the severity of psoriasis was positively associated with tonsil size, Dr. Shvartsbeyn and her colleagues reported.
Limited clinical data have suggested that there is an association between hypertrophic tonsils and inflammatory skin disease. Small studies have shown that among patients with psoriasis, the cutaneous lesions disappeared or improved after tonsillectomy. It is suspected that there may be a genetic predisposition that makes certain patient populations more susceptible, the researchers noted.
Histopathologic studies also point to the possible link between the robust immune response that takes place in the tonsils and the changes in the skin of patients with pustulosis palmaris et plantaris (PPP).
Histologic evaluation of tonsils obtained from patients with PPP has revealed enlargement of the secondary T nodules and atrophy of the lymph follicles, with a decrease in the number of the germinal center cells and fibrosis – changes typically seen in older tonsils. This finding provides indirect evidence of the intensely advanced stage of the immune response within the tonsils.
"Our hypothesis is that in chronic tonsillar hypertrophy, bacterial species that reside in the tonsils are released into the circulation and cause stimulation of T cells. As a result of this constant chronic stimulation, an autoreactive clone may be formed. The auto-clone may produce an antibody attacking the skin and drive inflammatory response. In some individuals, this exaggerated immune response may manifest as psoriasis," the investigators wrote.
And although there is empirical evidence "that tonsillectomy improved skin lesions in patients with psoriasis and pustulosis palmaris et plantaris in small retrospective studies, further studies are needed. ... The observed association needs validation and interventional study is needed to prove causation/contribution," Dr. Shvartsbeyn noted in an interview.
The study was supported by grants from the National Cancer Institute, the National Institute for Allergy and Infectious Diseases, and the National Institute of Dental and Craniofacial Research. The investigators did not report having any conflicts of interest.
AT THE AMERICAN ACADEMY OF DERMATOLOGY'S SUMMER ACADEMY MEETING
Major Finding: Patients with psoriasis had an odds ratio of 8.77 for having enlarged tonsils (grade 2 or greater), compared with healthy controls.
Data Source: The findings come from a prospective study of 32 patients with psoriasis and 14 patients with noninflammatory skin conditions, who served as controls.
Disclosures: The study was supported by grants from the National Cancer Institute, the National Institute for Allergy and Infectious Diseases, and the National Institute of Dental and Craniofacial Research. The investigators did not report having any conflicts of interest.
Anti-TNF Use Linked to Cardiovascular-Disease Drop in RA
Evidence continues to accumulate that the potent anti-inflammatory effect of drugs that block tumor necrosis factor can significantly dampen cardiovascular-disease risk in patients with rheumatoid arthritis, even though definitive proof from a prospective trial is still lacking.
Two recent pieces of suggestive evidence came from a meta-analysis of four placebo-controlled trials of adalimumab (Humira) that together included nearly 2,500 patients with rheumatoid arthritis, and from two prospective cohort studies of 828 RA patients that compared the outcomes of those treated with either adalimumab or etanercept (Enbrel) to the outcomes of similar patients who did not receive an anti–tumor necrosis factor drug.
In both studies, treatment with an anti-TNF agent was linked to a statistically significant cut in cardiovascular (CV) events of about 50%.
These results support another recent, similar finding reported in June at the Annual European Congress of Rheumatology in London. In that study, analysis of medical records from more than 109,000 U.S. patients with RA showed that every 6 months of treatment with an anti-TNF drug reduced the rate of CV events by 13%, compared with RA patients who did not receive a TNF blocker.
The meta-analysis of four trials included data collected in the ARMADA (Arthritis Rheum. 2003;48:35-45), DEO19 (Arthritis Rheum. 2004;50:1400-11), PREMIER (Arthritis Rheum. 2006;54:26-37), and OPTIMA (Ann. Rheum. Dis. 2012 May [doi: 10.1136/annrheumdis-2011-201247]) trials. Collectively, these four studies included 1,411 RA patients treated with both adalimumab and methotrexate, and 1,036 patients who received methotrexate but no anti-TNF drug. At baseline, patients in these two treatment groups had similar demographic and CV disease risk profiles.
During study periods that ranged from 16 to 104 weeks, the incidence of major adverse CV events was 1.3% in patients who received adalimumab, and 2% in those who didn’t. This difference represents a statistically significant, 2/3 drop in the risk for a major CV event in a proportional-hazard model, said Dr. Gerd Burmester, lead investigator for the analysis and a rheumatologist and professor of medicine at Charité Hospital in Berlin. His analysis also showed a statistically significant risk reduction with adalimumab in the rate of nonfatal myocardial infarction, but no significant effect of adalimumab on the rates of nonfatal myocardial infarction or nonfatal stroke compared with patients not receiving adalimumab.
A limitation of the analysis was that none of the four trials was powered to assess CV outcomes, Dr. Burmester said.
The second study used data collected from two cohorts: the CARRÉ (Cardiovascular Research and Rheumatoid Arthritis) study, a prospective Dutch cohort study of 309 randomly selected RA patients who were not treated with a TNF blocker; and the Biologics cohort, which involves 519 Dutch RA patients who have been followed since they began treatment with an anti-TNF drug for the first time, either adalimumab or etanercept.
During follow-up, there were 8 CV events per 1,000 patient-years in the cohort receiving an anti-TNF drug, compared with 23 events per 1,000 patient-years in patients not on an anti-TNF agent, a statistically significant reduction. In a proportional hazards model that adjusted for baseline differences in age and gender, treatment with an anti-TNF agent reduced the rate of CV events by about half, a statistically significant effect, said Dr. Alper M. van Sijl, a researcher at the Reade Centre for Rehabilitation and Rheumatology at the VU Medical Center in Amsterdam.
"Our observations confirm the association between strong suppression of inflammation [with an anti-TNF drug] and curbing the cardiovascular risk in RA," said Dr. van Sijl and his colleagues. But they cautioned that because of the design of the study it remains unclear whether this was a real effect of the anti-TNF drugs, or a bias to treat patients with lower CV disease risk with a TNF blocker.
Dr. Burmester said that he has been a consultant to, served as a speaker for, and received research support from Abbott, the company that markets adalimumab. Dr. van Silj said that he had no disclosures.
Evidence continues to accumulate that the potent anti-inflammatory effect of drugs that block tumor necrosis factor can significantly dampen cardiovascular-disease risk in patients with rheumatoid arthritis, even though definitive proof from a prospective trial is still lacking.
Two recent pieces of suggestive evidence came from a meta-analysis of four placebo-controlled trials of adalimumab (Humira) that together included nearly 2,500 patients with rheumatoid arthritis, and from two prospective cohort studies of 828 RA patients that compared the outcomes of those treated with either adalimumab or etanercept (Enbrel) to the outcomes of similar patients who did not receive an anti–tumor necrosis factor drug.
In both studies, treatment with an anti-TNF agent was linked to a statistically significant cut in cardiovascular (CV) events of about 50%.
These results support another recent, similar finding reported in June at the Annual European Congress of Rheumatology in London. In that study, analysis of medical records from more than 109,000 U.S. patients with RA showed that every 6 months of treatment with an anti-TNF drug reduced the rate of CV events by 13%, compared with RA patients who did not receive a TNF blocker.
The meta-analysis of four trials included data collected in the ARMADA (Arthritis Rheum. 2003;48:35-45), DEO19 (Arthritis Rheum. 2004;50:1400-11), PREMIER (Arthritis Rheum. 2006;54:26-37), and OPTIMA (Ann. Rheum. Dis. 2012 May [doi: 10.1136/annrheumdis-2011-201247]) trials. Collectively, these four studies included 1,411 RA patients treated with both adalimumab and methotrexate, and 1,036 patients who received methotrexate but no anti-TNF drug. At baseline, patients in these two treatment groups had similar demographic and CV disease risk profiles.
During study periods that ranged from 16 to 104 weeks, the incidence of major adverse CV events was 1.3% in patients who received adalimumab, and 2% in those who didn’t. This difference represents a statistically significant, 2/3 drop in the risk for a major CV event in a proportional-hazard model, said Dr. Gerd Burmester, lead investigator for the analysis and a rheumatologist and professor of medicine at Charité Hospital in Berlin. His analysis also showed a statistically significant risk reduction with adalimumab in the rate of nonfatal myocardial infarction, but no significant effect of adalimumab on the rates of nonfatal myocardial infarction or nonfatal stroke compared with patients not receiving adalimumab.
A limitation of the analysis was that none of the four trials was powered to assess CV outcomes, Dr. Burmester said.
The second study used data collected from two cohorts: the CARRÉ (Cardiovascular Research and Rheumatoid Arthritis) study, a prospective Dutch cohort study of 309 randomly selected RA patients who were not treated with a TNF blocker; and the Biologics cohort, which involves 519 Dutch RA patients who have been followed since they began treatment with an anti-TNF drug for the first time, either adalimumab or etanercept.
During follow-up, there were 8 CV events per 1,000 patient-years in the cohort receiving an anti-TNF drug, compared with 23 events per 1,000 patient-years in patients not on an anti-TNF agent, a statistically significant reduction. In a proportional hazards model that adjusted for baseline differences in age and gender, treatment with an anti-TNF agent reduced the rate of CV events by about half, a statistically significant effect, said Dr. Alper M. van Sijl, a researcher at the Reade Centre for Rehabilitation and Rheumatology at the VU Medical Center in Amsterdam.
"Our observations confirm the association between strong suppression of inflammation [with an anti-TNF drug] and curbing the cardiovascular risk in RA," said Dr. van Sijl and his colleagues. But they cautioned that because of the design of the study it remains unclear whether this was a real effect of the anti-TNF drugs, or a bias to treat patients with lower CV disease risk with a TNF blocker.
Dr. Burmester said that he has been a consultant to, served as a speaker for, and received research support from Abbott, the company that markets adalimumab. Dr. van Silj said that he had no disclosures.
Evidence continues to accumulate that the potent anti-inflammatory effect of drugs that block tumor necrosis factor can significantly dampen cardiovascular-disease risk in patients with rheumatoid arthritis, even though definitive proof from a prospective trial is still lacking.
Two recent pieces of suggestive evidence came from a meta-analysis of four placebo-controlled trials of adalimumab (Humira) that together included nearly 2,500 patients with rheumatoid arthritis, and from two prospective cohort studies of 828 RA patients that compared the outcomes of those treated with either adalimumab or etanercept (Enbrel) to the outcomes of similar patients who did not receive an anti–tumor necrosis factor drug.
In both studies, treatment with an anti-TNF agent was linked to a statistically significant cut in cardiovascular (CV) events of about 50%.
These results support another recent, similar finding reported in June at the Annual European Congress of Rheumatology in London. In that study, analysis of medical records from more than 109,000 U.S. patients with RA showed that every 6 months of treatment with an anti-TNF drug reduced the rate of CV events by 13%, compared with RA patients who did not receive a TNF blocker.
The meta-analysis of four trials included data collected in the ARMADA (Arthritis Rheum. 2003;48:35-45), DEO19 (Arthritis Rheum. 2004;50:1400-11), PREMIER (Arthritis Rheum. 2006;54:26-37), and OPTIMA (Ann. Rheum. Dis. 2012 May [doi: 10.1136/annrheumdis-2011-201247]) trials. Collectively, these four studies included 1,411 RA patients treated with both adalimumab and methotrexate, and 1,036 patients who received methotrexate but no anti-TNF drug. At baseline, patients in these two treatment groups had similar demographic and CV disease risk profiles.
During study periods that ranged from 16 to 104 weeks, the incidence of major adverse CV events was 1.3% in patients who received adalimumab, and 2% in those who didn’t. This difference represents a statistically significant, 2/3 drop in the risk for a major CV event in a proportional-hazard model, said Dr. Gerd Burmester, lead investigator for the analysis and a rheumatologist and professor of medicine at Charité Hospital in Berlin. His analysis also showed a statistically significant risk reduction with adalimumab in the rate of nonfatal myocardial infarction, but no significant effect of adalimumab on the rates of nonfatal myocardial infarction or nonfatal stroke compared with patients not receiving adalimumab.
A limitation of the analysis was that none of the four trials was powered to assess CV outcomes, Dr. Burmester said.
The second study used data collected from two cohorts: the CARRÉ (Cardiovascular Research and Rheumatoid Arthritis) study, a prospective Dutch cohort study of 309 randomly selected RA patients who were not treated with a TNF blocker; and the Biologics cohort, which involves 519 Dutch RA patients who have been followed since they began treatment with an anti-TNF drug for the first time, either adalimumab or etanercept.
During follow-up, there were 8 CV events per 1,000 patient-years in the cohort receiving an anti-TNF drug, compared with 23 events per 1,000 patient-years in patients not on an anti-TNF agent, a statistically significant reduction. In a proportional hazards model that adjusted for baseline differences in age and gender, treatment with an anti-TNF agent reduced the rate of CV events by about half, a statistically significant effect, said Dr. Alper M. van Sijl, a researcher at the Reade Centre for Rehabilitation and Rheumatology at the VU Medical Center in Amsterdam.
"Our observations confirm the association between strong suppression of inflammation [with an anti-TNF drug] and curbing the cardiovascular risk in RA," said Dr. van Sijl and his colleagues. But they cautioned that because of the design of the study it remains unclear whether this was a real effect of the anti-TNF drugs, or a bias to treat patients with lower CV disease risk with a TNF blocker.
Dr. Burmester said that he has been a consultant to, served as a speaker for, and received research support from Abbott, the company that markets adalimumab. Dr. van Silj said that he had no disclosures.
Psoriasis Patients Still Going Unscreened for Heart Disease
BOSTON – Despite the growing body of literature linking psoriasis to poor cardiovascular outcomes, most psoriasis patients are not being regularly screened for the major CV risk factors, according to the results of a new analysis.
"Screening for high blood pressure, diabetes, hypercholesterolemia, and obesity are not performed at most outpatient visits for psoriasis," wrote clinical research fellow Amir Al-Dabagh and colleagues at the center for dermatology research at Wake Forest University, Winston-Salem, N.C.
To determine whether and to what degree CV screening is taking place during outpatient psoriasis visits in the ambulatory care setting, the investigators reviewed data from the NAMCS (National Ambulatory Medical Care Survey) from 2005 to 2009. They calculated the probability of a patient’s being screened for at least one of four CV risk factors (blood pressure, glucose, cholesterol, and body mass index). They also compared screening rates by physician specialty, patient demographics, and clinical practice characteristics.
Approximately 11.4 million psoriasis patient visits were recorded during the study period. Age was found to be the only demographic factor that was significantly associated with all CV risk screens, Mr. Al-Dabagh reported in a poster presented at the American Academy of Dermatology’s Summer Academy Meeting. "Psoriasis had a statistically significant negative association on overall screening rates for blood pressure and BMI screening, but not for glucose or cholesterol."
A comparison of screening rates among patients with and without psoriasis showed that only 41.2% of the psoriasis patients were screened for at least one of the four risk factors, compared with 66.3% of patients without psoriasis. When looking at psoriasis patients only, the researchers found that screening for each of the risk factors occurred more frequently during nondermatology vs. dermatology visits, regardless of disease severity.
Specifically, among patients with severe and nonsevere psoriasis, respectively, 100% and 89.9% of psoriasis visits to nondermatology offices included screening for at least one risk factor, compared with 28.9% and 12.3% of psoriasis visits to dermatology offices, according to the analysis. In both settings, screening rates were higher among male patients, as well as among black and non-Hispanic patients, Mr. Al-Dabagh noted.
The majority of nondermatologist visits for patients with severe and nonsevere psoriasis included screening for blood pressure (100% for severe; 87.9% for not severe) and BMI (88.3% for severe; 54.4% for not severe). By comparison, during dermatology visits, 3.8% of patients with severe disease and 2.4% of those with mild to moderate disease had blood pressure screens, and 14.8% and 8.5%, respectively, had BMI recorded. Relatively few visits in either setting included glucose or cholesterol measurement, regardless of disease severity.
In 2008, the National Psoriasis Foundation issued a clinical consensus report recommending that screening psoriasis patients for cardiovascular risk factors begin as early as 20 years of age. The recommendation was based on mounting evidence from population-based studies that found psoriasis to be a risk factor for developing atherosclerosis and myocardial infarction (J. Amer. Acad. Derm. 2008;58:1031-42).
In addition to early, routine screening, psoriasis patients should be counseled to modify cholesterol levels when necessary, to take measures to control depression, to quit smoking, to moderate their alcohol intake, to eat a healthy diet, and to exercise at least three times a week, according to the consensus report.
The center for dermatology research at Wake Forest University is supported by an educational grant from Galderma. The study investigators reported financial relationships with multiple pharmaceutical companies, including Galderma.
BOSTON – Despite the growing body of literature linking psoriasis to poor cardiovascular outcomes, most psoriasis patients are not being regularly screened for the major CV risk factors, according to the results of a new analysis.
"Screening for high blood pressure, diabetes, hypercholesterolemia, and obesity are not performed at most outpatient visits for psoriasis," wrote clinical research fellow Amir Al-Dabagh and colleagues at the center for dermatology research at Wake Forest University, Winston-Salem, N.C.
To determine whether and to what degree CV screening is taking place during outpatient psoriasis visits in the ambulatory care setting, the investigators reviewed data from the NAMCS (National Ambulatory Medical Care Survey) from 2005 to 2009. They calculated the probability of a patient’s being screened for at least one of four CV risk factors (blood pressure, glucose, cholesterol, and body mass index). They also compared screening rates by physician specialty, patient demographics, and clinical practice characteristics.
Approximately 11.4 million psoriasis patient visits were recorded during the study period. Age was found to be the only demographic factor that was significantly associated with all CV risk screens, Mr. Al-Dabagh reported in a poster presented at the American Academy of Dermatology’s Summer Academy Meeting. "Psoriasis had a statistically significant negative association on overall screening rates for blood pressure and BMI screening, but not for glucose or cholesterol."
A comparison of screening rates among patients with and without psoriasis showed that only 41.2% of the psoriasis patients were screened for at least one of the four risk factors, compared with 66.3% of patients without psoriasis. When looking at psoriasis patients only, the researchers found that screening for each of the risk factors occurred more frequently during nondermatology vs. dermatology visits, regardless of disease severity.
Specifically, among patients with severe and nonsevere psoriasis, respectively, 100% and 89.9% of psoriasis visits to nondermatology offices included screening for at least one risk factor, compared with 28.9% and 12.3% of psoriasis visits to dermatology offices, according to the analysis. In both settings, screening rates were higher among male patients, as well as among black and non-Hispanic patients, Mr. Al-Dabagh noted.
The majority of nondermatologist visits for patients with severe and nonsevere psoriasis included screening for blood pressure (100% for severe; 87.9% for not severe) and BMI (88.3% for severe; 54.4% for not severe). By comparison, during dermatology visits, 3.8% of patients with severe disease and 2.4% of those with mild to moderate disease had blood pressure screens, and 14.8% and 8.5%, respectively, had BMI recorded. Relatively few visits in either setting included glucose or cholesterol measurement, regardless of disease severity.
In 2008, the National Psoriasis Foundation issued a clinical consensus report recommending that screening psoriasis patients for cardiovascular risk factors begin as early as 20 years of age. The recommendation was based on mounting evidence from population-based studies that found psoriasis to be a risk factor for developing atherosclerosis and myocardial infarction (J. Amer. Acad. Derm. 2008;58:1031-42).
In addition to early, routine screening, psoriasis patients should be counseled to modify cholesterol levels when necessary, to take measures to control depression, to quit smoking, to moderate their alcohol intake, to eat a healthy diet, and to exercise at least three times a week, according to the consensus report.
The center for dermatology research at Wake Forest University is supported by an educational grant from Galderma. The study investigators reported financial relationships with multiple pharmaceutical companies, including Galderma.
BOSTON – Despite the growing body of literature linking psoriasis to poor cardiovascular outcomes, most psoriasis patients are not being regularly screened for the major CV risk factors, according to the results of a new analysis.
"Screening for high blood pressure, diabetes, hypercholesterolemia, and obesity are not performed at most outpatient visits for psoriasis," wrote clinical research fellow Amir Al-Dabagh and colleagues at the center for dermatology research at Wake Forest University, Winston-Salem, N.C.
To determine whether and to what degree CV screening is taking place during outpatient psoriasis visits in the ambulatory care setting, the investigators reviewed data from the NAMCS (National Ambulatory Medical Care Survey) from 2005 to 2009. They calculated the probability of a patient’s being screened for at least one of four CV risk factors (blood pressure, glucose, cholesterol, and body mass index). They also compared screening rates by physician specialty, patient demographics, and clinical practice characteristics.
Approximately 11.4 million psoriasis patient visits were recorded during the study period. Age was found to be the only demographic factor that was significantly associated with all CV risk screens, Mr. Al-Dabagh reported in a poster presented at the American Academy of Dermatology’s Summer Academy Meeting. "Psoriasis had a statistically significant negative association on overall screening rates for blood pressure and BMI screening, but not for glucose or cholesterol."
A comparison of screening rates among patients with and without psoriasis showed that only 41.2% of the psoriasis patients were screened for at least one of the four risk factors, compared with 66.3% of patients without psoriasis. When looking at psoriasis patients only, the researchers found that screening for each of the risk factors occurred more frequently during nondermatology vs. dermatology visits, regardless of disease severity.
Specifically, among patients with severe and nonsevere psoriasis, respectively, 100% and 89.9% of psoriasis visits to nondermatology offices included screening for at least one risk factor, compared with 28.9% and 12.3% of psoriasis visits to dermatology offices, according to the analysis. In both settings, screening rates were higher among male patients, as well as among black and non-Hispanic patients, Mr. Al-Dabagh noted.
The majority of nondermatologist visits for patients with severe and nonsevere psoriasis included screening for blood pressure (100% for severe; 87.9% for not severe) and BMI (88.3% for severe; 54.4% for not severe). By comparison, during dermatology visits, 3.8% of patients with severe disease and 2.4% of those with mild to moderate disease had blood pressure screens, and 14.8% and 8.5%, respectively, had BMI recorded. Relatively few visits in either setting included glucose or cholesterol measurement, regardless of disease severity.
In 2008, the National Psoriasis Foundation issued a clinical consensus report recommending that screening psoriasis patients for cardiovascular risk factors begin as early as 20 years of age. The recommendation was based on mounting evidence from population-based studies that found psoriasis to be a risk factor for developing atherosclerosis and myocardial infarction (J. Amer. Acad. Derm. 2008;58:1031-42).
In addition to early, routine screening, psoriasis patients should be counseled to modify cholesterol levels when necessary, to take measures to control depression, to quit smoking, to moderate their alcohol intake, to eat a healthy diet, and to exercise at least three times a week, according to the consensus report.
The center for dermatology research at Wake Forest University is supported by an educational grant from Galderma. The study investigators reported financial relationships with multiple pharmaceutical companies, including Galderma.
AT THE AMERICAN ACADEMY OF DERMATOLOGY'S SUMMER ACADEMY MEETING
Major Finding: Approximately 41% of psoriasis patients were screened for at least one of four risk factors for cardiovascular disease during outpatient visits, compared with 66.3% of patients without the chronic inflammatory disease.
Data Source: Analysis of data from the National Ambulatory Medical Care Survey on outpatient psoriasis visits from 2005-2009.
Disclosures: The Center for Dermatology Research at Wake Forest University is supported by an educational grant from Galderma. The study investigators reported financial relationships with multiple pharmaceutical companies, including Galderma.
Biologic Agents for RA Don't Appear to Raise Malignancy Risk
The use of biologic agents to treat rheumatoid arthritis doesn’t appear to be associated with an increased risk of malignancy, compared with either placebo or with other disease-modifying drugs, according to a report in the Sept. 5 issue of JAMA.
In a meta-analysis of 63 randomized clinical trials of at least 24 weeks’ duration involving 29,423 adults with RA, there was no increase in the risk of cancer in general or in the risk of specific cancers. "Additional systematic reviews of observational studies are needed to establish risk in the longer term," said Dr. Maria A. Lopez-Olivo of the University of Texas M.D. Anderson Cancer Center, Houston, and her associates.
They described their study as the first systematic review and meta-analysis to assess the risk of any type of malignancy solely in RA patients who were taking any of the nine biologic agents approved for such use: abatacept, adalimumab, anakinra, certolizumab, etanercept, golimumab, infliximab, rituximab, or tocilizumab.
There has been concern that these agents could raise cancer risk because they interfere with the immune system. Some data have implicated tumor necrosis factor (TNF) inhibitors in particular, prompting the Food and Drug Administration to recommend adding a warning label citing an excess of cases of spontaneous lymphoma among children and adolescents taking TNF inhibitors.
"While trials in RA are relatively short and cannot evaluate the risk over longer-term exposure as observational studies do, we thought there was a need to conduct an updated meta-analysis of RCTs [randomized controlled trials] because of the older reports of the possible increase in malignancies and the more recent FDA advisory for TNF inhibitors, mostly based on studies in children," the researchers noted.
In the RCTs included in this meta-analysis, sample sizes ranged from 20 to 1,399 subjects. Most study subjects (79%) were white, and 76% were women. The mean duration of RA ranged from less than 1 year to 13 years.
Pharmaceutical companies sponsored 56 of the 63 trials, and another 3 did not disclose their funding sources. The remaining four trials were funded by national organizations, but the agents they used were provided free of charge by manufacturers. "There is evidence that industry-sponsored trials may overestimate the treatment effect and could possibly also overestimate safety," acacording to Dr. Lopez-Olivo and her associates.
A total of 15,989 study subjects were assigned to take biologic agents plus methotrexate and/or other disease-modifying antirheumatic drugs, while 3,615 were assigned to take the biologic agents alone and 9,819 served as control subjects who were given placebos.
A total of 211 malignancies developed during the trials, in 0.77% of the combination-therapy group, 0.64% of the monotherapy group, and 0.66% of the control group. These differences were not significant, the investigators said (JAMA 2012;308:898-908).
Skin cancers accounted for 48 of the malignancies (which included 4 melanomas), 14 were lymphomas, 26 were not specified, and 5 were hematologic nonlymphomas. The remaining 118 malignancies were solid tumors affecting a wide variety of organs including the adrenal glands, bladder, breast, GI system, liver, lung, ovary, pancreas, prostate, kidney, testes, thyroid, tongue, and uterus.
Thus there was no pattern involving a specific type of cancer.
Similarly, there was no pattern involving any of the individual biologic agents, Dr. Lopez-Olivo and her associates said.
"The only increased risk of malignancy we observed was in patients with RA treated with TNF inhibitors plus methotrexate at 52 weeks, for all cancers combined." However, this effect was not consistent across the three separate analytical methods the researchers used, did not occur in patients taking TNF inhibitors as monotherapy, and did not occur at any of the other three time points assessed.
The study findings "suggest that biologic, disease-modifying agents may be generally safe with respect to risk of malignancy in the short term," but their safety in RA patients who have concomitant cancer or risk factors for cancer remains unknown, the researchers said.
No sponsors were involved with this study, and no financial conflicts of interest were reported.
The use of biologic agents to treat rheumatoid arthritis doesn’t appear to be associated with an increased risk of malignancy, compared with either placebo or with other disease-modifying drugs, according to a report in the Sept. 5 issue of JAMA.
In a meta-analysis of 63 randomized clinical trials of at least 24 weeks’ duration involving 29,423 adults with RA, there was no increase in the risk of cancer in general or in the risk of specific cancers. "Additional systematic reviews of observational studies are needed to establish risk in the longer term," said Dr. Maria A. Lopez-Olivo of the University of Texas M.D. Anderson Cancer Center, Houston, and her associates.
They described their study as the first systematic review and meta-analysis to assess the risk of any type of malignancy solely in RA patients who were taking any of the nine biologic agents approved for such use: abatacept, adalimumab, anakinra, certolizumab, etanercept, golimumab, infliximab, rituximab, or tocilizumab.
There has been concern that these agents could raise cancer risk because they interfere with the immune system. Some data have implicated tumor necrosis factor (TNF) inhibitors in particular, prompting the Food and Drug Administration to recommend adding a warning label citing an excess of cases of spontaneous lymphoma among children and adolescents taking TNF inhibitors.
"While trials in RA are relatively short and cannot evaluate the risk over longer-term exposure as observational studies do, we thought there was a need to conduct an updated meta-analysis of RCTs [randomized controlled trials] because of the older reports of the possible increase in malignancies and the more recent FDA advisory for TNF inhibitors, mostly based on studies in children," the researchers noted.
In the RCTs included in this meta-analysis, sample sizes ranged from 20 to 1,399 subjects. Most study subjects (79%) were white, and 76% were women. The mean duration of RA ranged from less than 1 year to 13 years.
Pharmaceutical companies sponsored 56 of the 63 trials, and another 3 did not disclose their funding sources. The remaining four trials were funded by national organizations, but the agents they used were provided free of charge by manufacturers. "There is evidence that industry-sponsored trials may overestimate the treatment effect and could possibly also overestimate safety," acacording to Dr. Lopez-Olivo and her associates.
A total of 15,989 study subjects were assigned to take biologic agents plus methotrexate and/or other disease-modifying antirheumatic drugs, while 3,615 were assigned to take the biologic agents alone and 9,819 served as control subjects who were given placebos.
A total of 211 malignancies developed during the trials, in 0.77% of the combination-therapy group, 0.64% of the monotherapy group, and 0.66% of the control group. These differences were not significant, the investigators said (JAMA 2012;308:898-908).
Skin cancers accounted for 48 of the malignancies (which included 4 melanomas), 14 were lymphomas, 26 were not specified, and 5 were hematologic nonlymphomas. The remaining 118 malignancies were solid tumors affecting a wide variety of organs including the adrenal glands, bladder, breast, GI system, liver, lung, ovary, pancreas, prostate, kidney, testes, thyroid, tongue, and uterus.
Thus there was no pattern involving a specific type of cancer.
Similarly, there was no pattern involving any of the individual biologic agents, Dr. Lopez-Olivo and her associates said.
"The only increased risk of malignancy we observed was in patients with RA treated with TNF inhibitors plus methotrexate at 52 weeks, for all cancers combined." However, this effect was not consistent across the three separate analytical methods the researchers used, did not occur in patients taking TNF inhibitors as monotherapy, and did not occur at any of the other three time points assessed.
The study findings "suggest that biologic, disease-modifying agents may be generally safe with respect to risk of malignancy in the short term," but their safety in RA patients who have concomitant cancer or risk factors for cancer remains unknown, the researchers said.
No sponsors were involved with this study, and no financial conflicts of interest were reported.
The use of biologic agents to treat rheumatoid arthritis doesn’t appear to be associated with an increased risk of malignancy, compared with either placebo or with other disease-modifying drugs, according to a report in the Sept. 5 issue of JAMA.
In a meta-analysis of 63 randomized clinical trials of at least 24 weeks’ duration involving 29,423 adults with RA, there was no increase in the risk of cancer in general or in the risk of specific cancers. "Additional systematic reviews of observational studies are needed to establish risk in the longer term," said Dr. Maria A. Lopez-Olivo of the University of Texas M.D. Anderson Cancer Center, Houston, and her associates.
They described their study as the first systematic review and meta-analysis to assess the risk of any type of malignancy solely in RA patients who were taking any of the nine biologic agents approved for such use: abatacept, adalimumab, anakinra, certolizumab, etanercept, golimumab, infliximab, rituximab, or tocilizumab.
There has been concern that these agents could raise cancer risk because they interfere with the immune system. Some data have implicated tumor necrosis factor (TNF) inhibitors in particular, prompting the Food and Drug Administration to recommend adding a warning label citing an excess of cases of spontaneous lymphoma among children and adolescents taking TNF inhibitors.
"While trials in RA are relatively short and cannot evaluate the risk over longer-term exposure as observational studies do, we thought there was a need to conduct an updated meta-analysis of RCTs [randomized controlled trials] because of the older reports of the possible increase in malignancies and the more recent FDA advisory for TNF inhibitors, mostly based on studies in children," the researchers noted.
In the RCTs included in this meta-analysis, sample sizes ranged from 20 to 1,399 subjects. Most study subjects (79%) were white, and 76% were women. The mean duration of RA ranged from less than 1 year to 13 years.
Pharmaceutical companies sponsored 56 of the 63 trials, and another 3 did not disclose their funding sources. The remaining four trials were funded by national organizations, but the agents they used were provided free of charge by manufacturers. "There is evidence that industry-sponsored trials may overestimate the treatment effect and could possibly also overestimate safety," acacording to Dr. Lopez-Olivo and her associates.
A total of 15,989 study subjects were assigned to take biologic agents plus methotrexate and/or other disease-modifying antirheumatic drugs, while 3,615 were assigned to take the biologic agents alone and 9,819 served as control subjects who were given placebos.
A total of 211 malignancies developed during the trials, in 0.77% of the combination-therapy group, 0.64% of the monotherapy group, and 0.66% of the control group. These differences were not significant, the investigators said (JAMA 2012;308:898-908).
Skin cancers accounted for 48 of the malignancies (which included 4 melanomas), 14 were lymphomas, 26 were not specified, and 5 were hematologic nonlymphomas. The remaining 118 malignancies were solid tumors affecting a wide variety of organs including the adrenal glands, bladder, breast, GI system, liver, lung, ovary, pancreas, prostate, kidney, testes, thyroid, tongue, and uterus.
Thus there was no pattern involving a specific type of cancer.
Similarly, there was no pattern involving any of the individual biologic agents, Dr. Lopez-Olivo and her associates said.
"The only increased risk of malignancy we observed was in patients with RA treated with TNF inhibitors plus methotrexate at 52 weeks, for all cancers combined." However, this effect was not consistent across the three separate analytical methods the researchers used, did not occur in patients taking TNF inhibitors as monotherapy, and did not occur at any of the other three time points assessed.
The study findings "suggest that biologic, disease-modifying agents may be generally safe with respect to risk of malignancy in the short term," but their safety in RA patients who have concomitant cancer or risk factors for cancer remains unknown, the researchers said.
No sponsors were involved with this study, and no financial conflicts of interest were reported.
FROM JAMA
Major Finding: The incidence of malignancy was very low and comparable across the 3,615 patients who took biologic agents as monotherapy (0.64%), the 15,989 who took biologic agents with methotrexate (0.77%), and the 9,819 control subjects who took no biologic agents (0.66%).
Data Source: The data came from a meta-analysis of 63 randomized clinical trials of at least 24 weeks’ duration involving 29,423 adults with RA who were treated with abatacept, adalimumab, anakinra, certolizumab, etanercept, golimumab, infliximab, rituximab, or tocilizumab and then were followed for the development of cancer.
Disclosures: No sponsors were involved with this study, and no financial conflicts of interest were reported.