User login
Improving Colorectal Cancer Screening via Mailed Fecal Immunochemical Testing in a Veterans Affairs Health System
Colorectal cancer (CRC) is among the most common cancers and causes of cancer-related deaths in the United States.1 Reflective of a nationwide trend, CRC screening rates at the Veterans Affairs Connecticut Healthcare System (VACHS) decreased during the COVID-19 pandemic.2-5 Contributing factors to this decrease included cancellations of elective colonoscopies during the initial phase of the pandemic and concurrent turnover of endoscopists. In 2021, the US Preventive Services Task Force lowered the recommended initial CRC screening age from 50 years to 45 years, further increasing the backlog of unscreened patients.6
Fecal immunochemical testing (FIT) is a noninvasive screening method in which antibodies are used to detect hemoglobin in the stool. The sensitivity and specificity of 1-time FIT are 79% to 80% and 94%, respectively, for the detection of CRC, with sensitivity improving with successive testing.7,8 Annual FIT is recognized as a tier 1 preferred screening method by the US Multi-Society Task Force on Colorectal Cancer.7,9 Programs that mail FIT kits to eligible patients outside of physician visits have been successfully implemented in health care systems.10,11
The VACHS designed and implemented a mailed FIT program using existing infrastructure and staffing.
Program Description
A team of local stakeholders comprised of VACHS leadership, primary care, nursing, and gastroenterology staff, as well as representatives from laboratory, informatics, mail services, and group practice management, was established to execute the project. The team met monthly to plan the project.
The team developed a dataset consisting of patients aged 45 to 75 years who were at average risk for CRC and due for CRC screening. Patients were defined as due for CRC screening if they had not had a colonoscopy in the previous 9 years or a FIT or fecal occult blood test in the previous 11 months. Average risk for CRC was defined by excluding patients with associated diagnosis codes for CRC, colectomy, inflammatory bowel disease, and anemia. The program also excluded patients with diagnosis codes associated with dementia, deferring discussions about cancer screening to their primary care practitioners (PCPs). Patients with invalid mailing addresses were also excluded, as well as those whose PCPs had indicated in the electronic health record that the patient received CRC screening outside the US Department of Veterans Affairs (VA) system.
Letter Templates
Two patient letter electronic health record templates were developed. The first was a primer letter, which was mailed to patients 2 to 3 weeks before the mailed FIT kit as an introduction to the program.12 The purpose of the primer letter was to give advance notice to patients that they could expect a FIT kit to arrive in the mail. The goal was to prepare patients to complete FIT when the kit arrived and prompt them to call the VA to opt out of the mailed FIT program if they were up to date with CRC screening or if they had a condition which made them at high risk for CRC.
The second FIT letter arrived with the FIT kit, introduced FIT and described the importance of CRC screening. The letter detailed instructions for completing FIT and automatically created a FIT order. It also included a list of common conditions that may exclude patients, with a recommendation for patients to contact their medical team if they felt they were not candidates for FIT.
Staff Education
A previous VACHS pilot project demonstrated the success of a mailed FIT program to increase FIT use. Implemented as part of the pilot program, staff education consisted of a session for clinicians about the role of FIT in CRC screening and an all-staff education session. An additional education session about CRC and FIT for all staff was repeated with the program launch.
Program Launch
The mailed FIT program was introduced during a VACHS primary care all-staff meeting. After the meeting, each patient aligned care team (PACT) received an encrypted email that included a list of the patients on their team who were candidates for the program, a patient-facing FIT instruction sheet, detailed instructions on how to send the FIT primer letter, and a FIT package consisting of the labeled FIT kit, FIT letter, and patient instruction sheet. A reminder letter was sent to each patient 3 weeks after the FIT package was mailed. The patient lists were populated into a shared, encrypted Microsoft Teams folder that was edited in real time by PACT teams and viewed by VACHS leadership to track progress.
Program Metrics
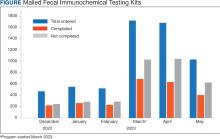
At program launch, the VACHS had 4642 patients due for CRC screening who were eligible for the mailed FIT program. On March 7, 2023, the data consisting of FIT tests ordered between December 2022 and May 2023—3 months before and after the launch of the program—were reviewed and categorized. In the 3 months before program launch, 1528 FIT were ordered and 714 were returned (46.7%). In the 3 months after the launch of the program, 4383 FIT were ordered and 1712 were returned (39.1%) (Figure). Test orders increased 287% from the preintervention to the postintervention period. The mean (SD) number of monthly FIT tests prelaunch was 509 (32.7), which increased to 1461 (331.6) postlaunch.
At the VACHS, 61.4% of patients aged 45 to 75 years were up to date with CRC screening before the program launch. In the 3 months after program launch, the rate increased to 63.8% among patients aged 45 to 75 years, the highest rate in our Veterans Integrated Services Network and exceeding the VA national average CRC screening rate, according to unpublished VA Monthly Management Report data.
In the 3 months following the program launch, 139 FIT kits tested positive for potential CRC. Of these, 79 (56.8%) patients had completed a diagnostic colonoscopy. PACT PCPs and nurses received reports on patients with positive FIT tests and those with no colonoscopy scheduled or completed and were asked to follow up.
Discussion
Through a proactive, population-based CRC screening program centered on mailed FIT kits outside of the traditional patient visit, the VACHS increased the use of FIT and rates of CRC screening. The numbers of FIT kits ordered and completed substantially increased in the 3 months after program launch.
Compared to mailed FIT programs described in the literature that rely on centralized processes in that a separate team operates the mailed FIT program for the entire organization, this program used existing PACT infrastructure and staff.10,11 This strategy allowed VACHS to design and implement the program in several months. Not needing to hire new staff or create a central team for the sole purpose of implementing the program allowed us to save on any organizational funding and efforts that would have accompanied the additional staff. The program described in this article may be more attainable for primary care practices or smaller health systems that do not have the capacity for the creation of a centralized process.
Limitations
Although the total number of FIT completions substantially increased during the program, the rate of FIT completion during the mailed FIT program was lower than the rate of completion prior to program launch. This decreased rate of FIT kit completion may be related to separation from a patient visit and potential loss of real-time education with a clinician. The program’s decentralized design increased the existing workload for primary care staff, and as a result, consideration must be given to local staffing levels. Additionally, the report of eligible patients depended on diagnosis codes and may have captured patients with higher-than-average risk of CRC, such as patients with prior history of adenomatous polyps, family history of CRC, or other medical or genetic conditions. We attempted to mitigate this by including a list of conditions that would exclude patients from FIT eligibility in the FIT letter and giving them the option to opt out.
Conclusions
CRC screening rates improved following implementation of a primary care team-centered quality improvement process to proactively identify patients appropriate for FIT and mail them FIT kits. This project highlights that population-health interventions around CRC screening via use of FIT can be successful within a primary care patient-centered medical home model, considering the increases in both CRC screening rates and increase in FIT tests ordered.
1. American Cancer Society. Key statistics for colorectal cancer. Revised January 29, 2024. Accessed June 11, 2024. https://www.cancer.org/cancer/types/colon-rectal-cancer/about/key-statistics.html
2. Chen RC, Haynes K, Du S, Barron J, Katz AJ. Association of cancer screening deficit in the United States with the COVID-19 pandemic. JAMA Oncol. 2021;7(6):878-884. doi:10.1001/jamaoncol.2021.0884
3. Mazidimoradi A, Tiznobaik A, Salehiniya H. Impact of the COVID-19 pandemic on colorectal cancer screening: a systematic review. J Gastrointest Cancer. 2022;53(3):730-744. doi:10.1007/s12029-021-00679-x
4. Adams MA, Kurlander JE, Gao Y, Yankey N, Saini SD. Impact of coronavirus disease 2019 on screening colonoscopy utilization in a large integrated health system. Gastroenterology. 2022;162(7):2098-2100.e2. doi:10.1053/j.gastro.2022.02.034
5. Sundaram S, Olson S, Sharma P, Rajendra S. A review of the impact of the COVID-19 pandemic on colorectal cancer screening: implications and solutions. Pathogens. 2021;10(11):558. doi:10.3390/pathogens10111508
6. US Preventive Services Task Force. Screening for colorectal cancer: US Preventive Services Task Force recommendation statement. JAMA. 2021;325(19):1965-1977. doi:10.1001/jama.2021.6238
7. Robertson DJ, Lee JK, Boland CR, et al. Recommendations on fecal immunochemical testing to screen for colorectal neoplasia: a consensus statement by the US Multi-Society Task Force on Colorectal Cancer. Gastrointest Endosc. 2017;85(1):2-21.e3. doi:10.1016/j.gie.2016.09.025
8. Lee JK, Liles EG, Bent S, Levin TR, Corley DA. Accuracy of fecal immunochemical tests for colorectal cancer: systematic review and meta-analysis. Ann Intern Med. 2014;160(3):171. doi:10.7326/M13-1484
9. Rex DK, Boland CR, Dominitz JA, et al. Colorectal cancer screening: recommendations for physicians and patients from the U.S. Multi-Society Task Force on Colorectal Cancer. Gastroenterology. 2017;153(1):307-323. doi:10.1053/j.gastro.2017.05.013
10. Deeds SA, Moore CB, Gunnink EJ, et al. Implementation of a mailed faecal immunochemical test programme for colorectal cancer screening among veterans. BMJ Open Qual. 2022;11(4):e001927. doi:10.1136/bmjoq-2022-001927
11. Selby K, Jensen CD, Levin TR, et al. Program components and results from an organized colorectal cancer screening program using annual fecal immunochemical testing. Clin Gastroenterol Hepatol. 2022;20(1):145-152. doi:10.1016/j.cgh.2020.09.042
12. Deeds S, Liu T, Schuttner L, et al. A postcard primer prior to mailed fecal immunochemical test among veterans: a randomized controlled trial. J Gen Intern Med. 2023:38(14):3235-3241. doi:10.1007/s11606-023-08248-7
Colorectal cancer (CRC) is among the most common cancers and causes of cancer-related deaths in the United States.1 Reflective of a nationwide trend, CRC screening rates at the Veterans Affairs Connecticut Healthcare System (VACHS) decreased during the COVID-19 pandemic.2-5 Contributing factors to this decrease included cancellations of elective colonoscopies during the initial phase of the pandemic and concurrent turnover of endoscopists. In 2021, the US Preventive Services Task Force lowered the recommended initial CRC screening age from 50 years to 45 years, further increasing the backlog of unscreened patients.6
Fecal immunochemical testing (FIT) is a noninvasive screening method in which antibodies are used to detect hemoglobin in the stool. The sensitivity and specificity of 1-time FIT are 79% to 80% and 94%, respectively, for the detection of CRC, with sensitivity improving with successive testing.7,8 Annual FIT is recognized as a tier 1 preferred screening method by the US Multi-Society Task Force on Colorectal Cancer.7,9 Programs that mail FIT kits to eligible patients outside of physician visits have been successfully implemented in health care systems.10,11
The VACHS designed and implemented a mailed FIT program using existing infrastructure and staffing.
Program Description
A team of local stakeholders comprised of VACHS leadership, primary care, nursing, and gastroenterology staff, as well as representatives from laboratory, informatics, mail services, and group practice management, was established to execute the project. The team met monthly to plan the project.
The team developed a dataset consisting of patients aged 45 to 75 years who were at average risk for CRC and due for CRC screening. Patients were defined as due for CRC screening if they had not had a colonoscopy in the previous 9 years or a FIT or fecal occult blood test in the previous 11 months. Average risk for CRC was defined by excluding patients with associated diagnosis codes for CRC, colectomy, inflammatory bowel disease, and anemia. The program also excluded patients with diagnosis codes associated with dementia, deferring discussions about cancer screening to their primary care practitioners (PCPs). Patients with invalid mailing addresses were also excluded, as well as those whose PCPs had indicated in the electronic health record that the patient received CRC screening outside the US Department of Veterans Affairs (VA) system.
Letter Templates
Two patient letter electronic health record templates were developed. The first was a primer letter, which was mailed to patients 2 to 3 weeks before the mailed FIT kit as an introduction to the program.12 The purpose of the primer letter was to give advance notice to patients that they could expect a FIT kit to arrive in the mail. The goal was to prepare patients to complete FIT when the kit arrived and prompt them to call the VA to opt out of the mailed FIT program if they were up to date with CRC screening or if they had a condition which made them at high risk for CRC.
The second FIT letter arrived with the FIT kit, introduced FIT and described the importance of CRC screening. The letter detailed instructions for completing FIT and automatically created a FIT order. It also included a list of common conditions that may exclude patients, with a recommendation for patients to contact their medical team if they felt they were not candidates for FIT.
Staff Education
A previous VACHS pilot project demonstrated the success of a mailed FIT program to increase FIT use. Implemented as part of the pilot program, staff education consisted of a session for clinicians about the role of FIT in CRC screening and an all-staff education session. An additional education session about CRC and FIT for all staff was repeated with the program launch.
Program Launch
The mailed FIT program was introduced during a VACHS primary care all-staff meeting. After the meeting, each patient aligned care team (PACT) received an encrypted email that included a list of the patients on their team who were candidates for the program, a patient-facing FIT instruction sheet, detailed instructions on how to send the FIT primer letter, and a FIT package consisting of the labeled FIT kit, FIT letter, and patient instruction sheet. A reminder letter was sent to each patient 3 weeks after the FIT package was mailed. The patient lists were populated into a shared, encrypted Microsoft Teams folder that was edited in real time by PACT teams and viewed by VACHS leadership to track progress.
Program Metrics
At program launch, the VACHS had 4642 patients due for CRC screening who were eligible for the mailed FIT program. On March 7, 2023, the data consisting of FIT tests ordered between December 2022 and May 2023—3 months before and after the launch of the program—were reviewed and categorized. In the 3 months before program launch, 1528 FIT were ordered and 714 were returned (46.7%). In the 3 months after the launch of the program, 4383 FIT were ordered and 1712 were returned (39.1%) (Figure). Test orders increased 287% from the preintervention to the postintervention period. The mean (SD) number of monthly FIT tests prelaunch was 509 (32.7), which increased to 1461 (331.6) postlaunch.
At the VACHS, 61.4% of patients aged 45 to 75 years were up to date with CRC screening before the program launch. In the 3 months after program launch, the rate increased to 63.8% among patients aged 45 to 75 years, the highest rate in our Veterans Integrated Services Network and exceeding the VA national average CRC screening rate, according to unpublished VA Monthly Management Report data.
In the 3 months following the program launch, 139 FIT kits tested positive for potential CRC. Of these, 79 (56.8%) patients had completed a diagnostic colonoscopy. PACT PCPs and nurses received reports on patients with positive FIT tests and those with no colonoscopy scheduled or completed and were asked to follow up.
Discussion
Through a proactive, population-based CRC screening program centered on mailed FIT kits outside of the traditional patient visit, the VACHS increased the use of FIT and rates of CRC screening. The numbers of FIT kits ordered and completed substantially increased in the 3 months after program launch.
Compared to mailed FIT programs described in the literature that rely on centralized processes in that a separate team operates the mailed FIT program for the entire organization, this program used existing PACT infrastructure and staff.10,11 This strategy allowed VACHS to design and implement the program in several months. Not needing to hire new staff or create a central team for the sole purpose of implementing the program allowed us to save on any organizational funding and efforts that would have accompanied the additional staff. The program described in this article may be more attainable for primary care practices or smaller health systems that do not have the capacity for the creation of a centralized process.
Limitations
Although the total number of FIT completions substantially increased during the program, the rate of FIT completion during the mailed FIT program was lower than the rate of completion prior to program launch. This decreased rate of FIT kit completion may be related to separation from a patient visit and potential loss of real-time education with a clinician. The program’s decentralized design increased the existing workload for primary care staff, and as a result, consideration must be given to local staffing levels. Additionally, the report of eligible patients depended on diagnosis codes and may have captured patients with higher-than-average risk of CRC, such as patients with prior history of adenomatous polyps, family history of CRC, or other medical or genetic conditions. We attempted to mitigate this by including a list of conditions that would exclude patients from FIT eligibility in the FIT letter and giving them the option to opt out.
Conclusions
CRC screening rates improved following implementation of a primary care team-centered quality improvement process to proactively identify patients appropriate for FIT and mail them FIT kits. This project highlights that population-health interventions around CRC screening via use of FIT can be successful within a primary care patient-centered medical home model, considering the increases in both CRC screening rates and increase in FIT tests ordered.
Colorectal cancer (CRC) is among the most common cancers and causes of cancer-related deaths in the United States.1 Reflective of a nationwide trend, CRC screening rates at the Veterans Affairs Connecticut Healthcare System (VACHS) decreased during the COVID-19 pandemic.2-5 Contributing factors to this decrease included cancellations of elective colonoscopies during the initial phase of the pandemic and concurrent turnover of endoscopists. In 2021, the US Preventive Services Task Force lowered the recommended initial CRC screening age from 50 years to 45 years, further increasing the backlog of unscreened patients.6
Fecal immunochemical testing (FIT) is a noninvasive screening method in which antibodies are used to detect hemoglobin in the stool. The sensitivity and specificity of 1-time FIT are 79% to 80% and 94%, respectively, for the detection of CRC, with sensitivity improving with successive testing.7,8 Annual FIT is recognized as a tier 1 preferred screening method by the US Multi-Society Task Force on Colorectal Cancer.7,9 Programs that mail FIT kits to eligible patients outside of physician visits have been successfully implemented in health care systems.10,11
The VACHS designed and implemented a mailed FIT program using existing infrastructure and staffing.
Program Description
A team of local stakeholders comprised of VACHS leadership, primary care, nursing, and gastroenterology staff, as well as representatives from laboratory, informatics, mail services, and group practice management, was established to execute the project. The team met monthly to plan the project.
The team developed a dataset consisting of patients aged 45 to 75 years who were at average risk for CRC and due for CRC screening. Patients were defined as due for CRC screening if they had not had a colonoscopy in the previous 9 years or a FIT or fecal occult blood test in the previous 11 months. Average risk for CRC was defined by excluding patients with associated diagnosis codes for CRC, colectomy, inflammatory bowel disease, and anemia. The program also excluded patients with diagnosis codes associated with dementia, deferring discussions about cancer screening to their primary care practitioners (PCPs). Patients with invalid mailing addresses were also excluded, as well as those whose PCPs had indicated in the electronic health record that the patient received CRC screening outside the US Department of Veterans Affairs (VA) system.
Letter Templates
Two patient letter electronic health record templates were developed. The first was a primer letter, which was mailed to patients 2 to 3 weeks before the mailed FIT kit as an introduction to the program.12 The purpose of the primer letter was to give advance notice to patients that they could expect a FIT kit to arrive in the mail. The goal was to prepare patients to complete FIT when the kit arrived and prompt them to call the VA to opt out of the mailed FIT program if they were up to date with CRC screening or if they had a condition which made them at high risk for CRC.
The second FIT letter arrived with the FIT kit, introduced FIT and described the importance of CRC screening. The letter detailed instructions for completing FIT and automatically created a FIT order. It also included a list of common conditions that may exclude patients, with a recommendation for patients to contact their medical team if they felt they were not candidates for FIT.
Staff Education
A previous VACHS pilot project demonstrated the success of a mailed FIT program to increase FIT use. Implemented as part of the pilot program, staff education consisted of a session for clinicians about the role of FIT in CRC screening and an all-staff education session. An additional education session about CRC and FIT for all staff was repeated with the program launch.
Program Launch
The mailed FIT program was introduced during a VACHS primary care all-staff meeting. After the meeting, each patient aligned care team (PACT) received an encrypted email that included a list of the patients on their team who were candidates for the program, a patient-facing FIT instruction sheet, detailed instructions on how to send the FIT primer letter, and a FIT package consisting of the labeled FIT kit, FIT letter, and patient instruction sheet. A reminder letter was sent to each patient 3 weeks after the FIT package was mailed. The patient lists were populated into a shared, encrypted Microsoft Teams folder that was edited in real time by PACT teams and viewed by VACHS leadership to track progress.
Program Metrics
At program launch, the VACHS had 4642 patients due for CRC screening who were eligible for the mailed FIT program. On March 7, 2023, the data consisting of FIT tests ordered between December 2022 and May 2023—3 months before and after the launch of the program—were reviewed and categorized. In the 3 months before program launch, 1528 FIT were ordered and 714 were returned (46.7%). In the 3 months after the launch of the program, 4383 FIT were ordered and 1712 were returned (39.1%) (Figure). Test orders increased 287% from the preintervention to the postintervention period. The mean (SD) number of monthly FIT tests prelaunch was 509 (32.7), which increased to 1461 (331.6) postlaunch.
At the VACHS, 61.4% of patients aged 45 to 75 years were up to date with CRC screening before the program launch. In the 3 months after program launch, the rate increased to 63.8% among patients aged 45 to 75 years, the highest rate in our Veterans Integrated Services Network and exceeding the VA national average CRC screening rate, according to unpublished VA Monthly Management Report data.
In the 3 months following the program launch, 139 FIT kits tested positive for potential CRC. Of these, 79 (56.8%) patients had completed a diagnostic colonoscopy. PACT PCPs and nurses received reports on patients with positive FIT tests and those with no colonoscopy scheduled or completed and were asked to follow up.
Discussion
Through a proactive, population-based CRC screening program centered on mailed FIT kits outside of the traditional patient visit, the VACHS increased the use of FIT and rates of CRC screening. The numbers of FIT kits ordered and completed substantially increased in the 3 months after program launch.
Compared to mailed FIT programs described in the literature that rely on centralized processes in that a separate team operates the mailed FIT program for the entire organization, this program used existing PACT infrastructure and staff.10,11 This strategy allowed VACHS to design and implement the program in several months. Not needing to hire new staff or create a central team for the sole purpose of implementing the program allowed us to save on any organizational funding and efforts that would have accompanied the additional staff. The program described in this article may be more attainable for primary care practices or smaller health systems that do not have the capacity for the creation of a centralized process.
Limitations
Although the total number of FIT completions substantially increased during the program, the rate of FIT completion during the mailed FIT program was lower than the rate of completion prior to program launch. This decreased rate of FIT kit completion may be related to separation from a patient visit and potential loss of real-time education with a clinician. The program’s decentralized design increased the existing workload for primary care staff, and as a result, consideration must be given to local staffing levels. Additionally, the report of eligible patients depended on diagnosis codes and may have captured patients with higher-than-average risk of CRC, such as patients with prior history of adenomatous polyps, family history of CRC, or other medical or genetic conditions. We attempted to mitigate this by including a list of conditions that would exclude patients from FIT eligibility in the FIT letter and giving them the option to opt out.
Conclusions
CRC screening rates improved following implementation of a primary care team-centered quality improvement process to proactively identify patients appropriate for FIT and mail them FIT kits. This project highlights that population-health interventions around CRC screening via use of FIT can be successful within a primary care patient-centered medical home model, considering the increases in both CRC screening rates and increase in FIT tests ordered.
1. American Cancer Society. Key statistics for colorectal cancer. Revised January 29, 2024. Accessed June 11, 2024. https://www.cancer.org/cancer/types/colon-rectal-cancer/about/key-statistics.html
2. Chen RC, Haynes K, Du S, Barron J, Katz AJ. Association of cancer screening deficit in the United States with the COVID-19 pandemic. JAMA Oncol. 2021;7(6):878-884. doi:10.1001/jamaoncol.2021.0884
3. Mazidimoradi A, Tiznobaik A, Salehiniya H. Impact of the COVID-19 pandemic on colorectal cancer screening: a systematic review. J Gastrointest Cancer. 2022;53(3):730-744. doi:10.1007/s12029-021-00679-x
4. Adams MA, Kurlander JE, Gao Y, Yankey N, Saini SD. Impact of coronavirus disease 2019 on screening colonoscopy utilization in a large integrated health system. Gastroenterology. 2022;162(7):2098-2100.e2. doi:10.1053/j.gastro.2022.02.034
5. Sundaram S, Olson S, Sharma P, Rajendra S. A review of the impact of the COVID-19 pandemic on colorectal cancer screening: implications and solutions. Pathogens. 2021;10(11):558. doi:10.3390/pathogens10111508
6. US Preventive Services Task Force. Screening for colorectal cancer: US Preventive Services Task Force recommendation statement. JAMA. 2021;325(19):1965-1977. doi:10.1001/jama.2021.6238
7. Robertson DJ, Lee JK, Boland CR, et al. Recommendations on fecal immunochemical testing to screen for colorectal neoplasia: a consensus statement by the US Multi-Society Task Force on Colorectal Cancer. Gastrointest Endosc. 2017;85(1):2-21.e3. doi:10.1016/j.gie.2016.09.025
8. Lee JK, Liles EG, Bent S, Levin TR, Corley DA. Accuracy of fecal immunochemical tests for colorectal cancer: systematic review and meta-analysis. Ann Intern Med. 2014;160(3):171. doi:10.7326/M13-1484
9. Rex DK, Boland CR, Dominitz JA, et al. Colorectal cancer screening: recommendations for physicians and patients from the U.S. Multi-Society Task Force on Colorectal Cancer. Gastroenterology. 2017;153(1):307-323. doi:10.1053/j.gastro.2017.05.013
10. Deeds SA, Moore CB, Gunnink EJ, et al. Implementation of a mailed faecal immunochemical test programme for colorectal cancer screening among veterans. BMJ Open Qual. 2022;11(4):e001927. doi:10.1136/bmjoq-2022-001927
11. Selby K, Jensen CD, Levin TR, et al. Program components and results from an organized colorectal cancer screening program using annual fecal immunochemical testing. Clin Gastroenterol Hepatol. 2022;20(1):145-152. doi:10.1016/j.cgh.2020.09.042
12. Deeds S, Liu T, Schuttner L, et al. A postcard primer prior to mailed fecal immunochemical test among veterans: a randomized controlled trial. J Gen Intern Med. 2023:38(14):3235-3241. doi:10.1007/s11606-023-08248-7
1. American Cancer Society. Key statistics for colorectal cancer. Revised January 29, 2024. Accessed June 11, 2024. https://www.cancer.org/cancer/types/colon-rectal-cancer/about/key-statistics.html
2. Chen RC, Haynes K, Du S, Barron J, Katz AJ. Association of cancer screening deficit in the United States with the COVID-19 pandemic. JAMA Oncol. 2021;7(6):878-884. doi:10.1001/jamaoncol.2021.0884
3. Mazidimoradi A, Tiznobaik A, Salehiniya H. Impact of the COVID-19 pandemic on colorectal cancer screening: a systematic review. J Gastrointest Cancer. 2022;53(3):730-744. doi:10.1007/s12029-021-00679-x
4. Adams MA, Kurlander JE, Gao Y, Yankey N, Saini SD. Impact of coronavirus disease 2019 on screening colonoscopy utilization in a large integrated health system. Gastroenterology. 2022;162(7):2098-2100.e2. doi:10.1053/j.gastro.2022.02.034
5. Sundaram S, Olson S, Sharma P, Rajendra S. A review of the impact of the COVID-19 pandemic on colorectal cancer screening: implications and solutions. Pathogens. 2021;10(11):558. doi:10.3390/pathogens10111508
6. US Preventive Services Task Force. Screening for colorectal cancer: US Preventive Services Task Force recommendation statement. JAMA. 2021;325(19):1965-1977. doi:10.1001/jama.2021.6238
7. Robertson DJ, Lee JK, Boland CR, et al. Recommendations on fecal immunochemical testing to screen for colorectal neoplasia: a consensus statement by the US Multi-Society Task Force on Colorectal Cancer. Gastrointest Endosc. 2017;85(1):2-21.e3. doi:10.1016/j.gie.2016.09.025
8. Lee JK, Liles EG, Bent S, Levin TR, Corley DA. Accuracy of fecal immunochemical tests for colorectal cancer: systematic review and meta-analysis. Ann Intern Med. 2014;160(3):171. doi:10.7326/M13-1484
9. Rex DK, Boland CR, Dominitz JA, et al. Colorectal cancer screening: recommendations for physicians and patients from the U.S. Multi-Society Task Force on Colorectal Cancer. Gastroenterology. 2017;153(1):307-323. doi:10.1053/j.gastro.2017.05.013
10. Deeds SA, Moore CB, Gunnink EJ, et al. Implementation of a mailed faecal immunochemical test programme for colorectal cancer screening among veterans. BMJ Open Qual. 2022;11(4):e001927. doi:10.1136/bmjoq-2022-001927
11. Selby K, Jensen CD, Levin TR, et al. Program components and results from an organized colorectal cancer screening program using annual fecal immunochemical testing. Clin Gastroenterol Hepatol. 2022;20(1):145-152. doi:10.1016/j.cgh.2020.09.042
12. Deeds S, Liu T, Schuttner L, et al. A postcard primer prior to mailed fecal immunochemical test among veterans: a randomized controlled trial. J Gen Intern Med. 2023:38(14):3235-3241. doi:10.1007/s11606-023-08248-7
Military Women Survive Ovarian Cancer at Higher Rates
Military Women Survive Ovarian Cancer at Higher Rates
Women with epithelial ovarian cancer treated in the US Department of Defense (DoD) universal health care system demonstrate better 5-year survival compared with similar patients from the national population. The survival advantage persists across multiple age groups and disease stages, with particularly notable improvements in patients aged 35-49 years and those with stage III disease.
METHODOLOGY:
- Researchers compared 1504 patients with invasive stage I-IV epithelial ovarian carcinoma from the Automated Center Tumor Registry (ACTUR) for the DoD with 6016 matched patients from the 18-region Surveillance, Epidemiology, and End Results (SEER) program between 1987 and 2013.
- Patients from ACTUR were matched in a 1:4 ratio with SEER patients stratified for age, race, year of diagnosis, and histology, including serous carcinoma, clear cell carcinoma, mucinous carcinoma, and endometrioid carcinoma with adenocarcinoma subtypes.
- Five-year overall survival was evaluated using the Kaplan-Meier method and compared using log-rank test, with median follow-up time of 46 months in ACTUR and 44 months in SEER.
- Adjusted hazard ratio (AHR) and 95% CI for all-cause mortality were estimated from multivariable Cox proportional regression modeling controlling for age, race, year of diagnosis, region of diagnosis, stage, histology, and grade.
TAKEAWAY:
- Overall survival differs between registries: 5-year survival of 53.2% in ACTUR vs 47.7% in matched SEER cohort (log-rank P = .001).
- In the primary adjusted model, ACTUR is associated with a lower risk for all-cause mortality vs SEER (AHR, 0.83; 95% CI, 0.76-0.91; P < .0001).
- Subset results retain lower adjusted risk for death for ACTUR vs SEER among ages 35-49 years (AHR, 0.66; 95% CI, 0.52-0.83; P = .0005), ages ≥ 65 years (AHR, 0.82; 95% CI, 0.70-0.96; P = .016), and stage III cancer (AHR, 0.79; 95% CI, 0.69-0.91; P = .0015).
- Histology-stratified findings show lower adjusted risk for death in ACTUR vs SEER for clear cell carcinoma (AHR, 0.63; 95% CI, 0.43-0.93; P =.02) and for endometrioid and other adenocarcinomas (AHR, 0.68; 95% CI, 0.56-0.81; P < .0001).
IN PRACTICE:
"This study is envisioned to be a stepping stone to further investigations of survival and other cancer health outcomes starting with patients diagnosed between 2014 and 2024 with epithelial carcinoma of the ovary, fallopian tube, or primary peritoneum in the DoD Healthcare System versus the national population or other Healthcare Systems,” wrote the authors of the study. “Dedicated funding and support in the [Military Health System] are needed to invest in infrastructure, technology, security, education, and research.”
SOURCE:
The study was led by Kathleen M. Darcy, PhD, and Christopher M. Tarney, MD, from the Gynecologic Cancer Center of Excellence, Department of Gynecologic Surgery & Obstetrics, Uniformed Services University, Walter Reed National Military Medical Center in Bethesda, Maryland. It was published online in Military Medicine.
LIMITATIONS:
The retrospective cohort study design limits causal inference. Although groups were balanced by age, race, year, and region of diagnosis, other demographic factors and socioeconomic variables such as patient comorbidities, educational attainment, household income, and health insurance status were not available and may have affected results. The databases fundamentally differ in how data are acquired, with ACTUR following hospital-based Facility Oncology Registry Data Standards and SEER being a national population-based registry, potentially affecting data quality, consistency, and reliability of survival outcome comparisons. The inclusion of patients diagnosed only through 2013 represents a limitation as it does not allow for contemporary evaluation of survival outcomes, particularly given advances over the past decade including maximal cytoreductive effort to no residual disease, increased adoption of neoadjuvant chemotherapy, and introduction of targeted maintenance agents. The study could not incorporate details regarding residual disease status or control for specifics regarding surgical and medical management, including primary vs interval debulking surgery or the type and timing of agents utilized in first-line, maintenance, and recurrent disease settings. Data regarding circulating biomarkers including CA125, molecular subtypes or alterations, and stratification by homologous recombination deficiency vs proficiency status were not available. Epithelial carcinomas of the fallopian tube and primary peritoneum were excluded from this study, which now are commonly incorporated with ovarian carcinomas. Results may not be generalizable to other populations given the unique characteristics of the Military Health System beneficiary population.
DISCLOSURES:
This research received funding from the Uniformed Services University from the Defense Health Program to the Henry M. Jackson Foundation for the Advancement of Military Medicine Inc., including award HU0001-18-2-0032 to the Murtha Cancer Center Research Program and awards HU0001-19-2-0031 and HU0001-24-2-0047 to the Gynecologic Cancer Center of Excellence Program. All coauthors disclosed no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
Women with epithelial ovarian cancer treated in the US Department of Defense (DoD) universal health care system demonstrate better 5-year survival compared with similar patients from the national population. The survival advantage persists across multiple age groups and disease stages, with particularly notable improvements in patients aged 35-49 years and those with stage III disease.
METHODOLOGY:
- Researchers compared 1504 patients with invasive stage I-IV epithelial ovarian carcinoma from the Automated Center Tumor Registry (ACTUR) for the DoD with 6016 matched patients from the 18-region Surveillance, Epidemiology, and End Results (SEER) program between 1987 and 2013.
- Patients from ACTUR were matched in a 1:4 ratio with SEER patients stratified for age, race, year of diagnosis, and histology, including serous carcinoma, clear cell carcinoma, mucinous carcinoma, and endometrioid carcinoma with adenocarcinoma subtypes.
- Five-year overall survival was evaluated using the Kaplan-Meier method and compared using log-rank test, with median follow-up time of 46 months in ACTUR and 44 months in SEER.
- Adjusted hazard ratio (AHR) and 95% CI for all-cause mortality were estimated from multivariable Cox proportional regression modeling controlling for age, race, year of diagnosis, region of diagnosis, stage, histology, and grade.
TAKEAWAY:
- Overall survival differs between registries: 5-year survival of 53.2% in ACTUR vs 47.7% in matched SEER cohort (log-rank P = .001).
- In the primary adjusted model, ACTUR is associated with a lower risk for all-cause mortality vs SEER (AHR, 0.83; 95% CI, 0.76-0.91; P < .0001).
- Subset results retain lower adjusted risk for death for ACTUR vs SEER among ages 35-49 years (AHR, 0.66; 95% CI, 0.52-0.83; P = .0005), ages ≥ 65 years (AHR, 0.82; 95% CI, 0.70-0.96; P = .016), and stage III cancer (AHR, 0.79; 95% CI, 0.69-0.91; P = .0015).
- Histology-stratified findings show lower adjusted risk for death in ACTUR vs SEER for clear cell carcinoma (AHR, 0.63; 95% CI, 0.43-0.93; P =.02) and for endometrioid and other adenocarcinomas (AHR, 0.68; 95% CI, 0.56-0.81; P < .0001).
IN PRACTICE:
"This study is envisioned to be a stepping stone to further investigations of survival and other cancer health outcomes starting with patients diagnosed between 2014 and 2024 with epithelial carcinoma of the ovary, fallopian tube, or primary peritoneum in the DoD Healthcare System versus the national population or other Healthcare Systems,” wrote the authors of the study. “Dedicated funding and support in the [Military Health System] are needed to invest in infrastructure, technology, security, education, and research.”
SOURCE:
The study was led by Kathleen M. Darcy, PhD, and Christopher M. Tarney, MD, from the Gynecologic Cancer Center of Excellence, Department of Gynecologic Surgery & Obstetrics, Uniformed Services University, Walter Reed National Military Medical Center in Bethesda, Maryland. It was published online in Military Medicine.
LIMITATIONS:
The retrospective cohort study design limits causal inference. Although groups were balanced by age, race, year, and region of diagnosis, other demographic factors and socioeconomic variables such as patient comorbidities, educational attainment, household income, and health insurance status were not available and may have affected results. The databases fundamentally differ in how data are acquired, with ACTUR following hospital-based Facility Oncology Registry Data Standards and SEER being a national population-based registry, potentially affecting data quality, consistency, and reliability of survival outcome comparisons. The inclusion of patients diagnosed only through 2013 represents a limitation as it does not allow for contemporary evaluation of survival outcomes, particularly given advances over the past decade including maximal cytoreductive effort to no residual disease, increased adoption of neoadjuvant chemotherapy, and introduction of targeted maintenance agents. The study could not incorporate details regarding residual disease status or control for specifics regarding surgical and medical management, including primary vs interval debulking surgery or the type and timing of agents utilized in first-line, maintenance, and recurrent disease settings. Data regarding circulating biomarkers including CA125, molecular subtypes or alterations, and stratification by homologous recombination deficiency vs proficiency status were not available. Epithelial carcinomas of the fallopian tube and primary peritoneum were excluded from this study, which now are commonly incorporated with ovarian carcinomas. Results may not be generalizable to other populations given the unique characteristics of the Military Health System beneficiary population.
DISCLOSURES:
This research received funding from the Uniformed Services University from the Defense Health Program to the Henry M. Jackson Foundation for the Advancement of Military Medicine Inc., including award HU0001-18-2-0032 to the Murtha Cancer Center Research Program and awards HU0001-19-2-0031 and HU0001-24-2-0047 to the Gynecologic Cancer Center of Excellence Program. All coauthors disclosed no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
Women with epithelial ovarian cancer treated in the US Department of Defense (DoD) universal health care system demonstrate better 5-year survival compared with similar patients from the national population. The survival advantage persists across multiple age groups and disease stages, with particularly notable improvements in patients aged 35-49 years and those with stage III disease.
METHODOLOGY:
- Researchers compared 1504 patients with invasive stage I-IV epithelial ovarian carcinoma from the Automated Center Tumor Registry (ACTUR) for the DoD with 6016 matched patients from the 18-region Surveillance, Epidemiology, and End Results (SEER) program between 1987 and 2013.
- Patients from ACTUR were matched in a 1:4 ratio with SEER patients stratified for age, race, year of diagnosis, and histology, including serous carcinoma, clear cell carcinoma, mucinous carcinoma, and endometrioid carcinoma with adenocarcinoma subtypes.
- Five-year overall survival was evaluated using the Kaplan-Meier method and compared using log-rank test, with median follow-up time of 46 months in ACTUR and 44 months in SEER.
- Adjusted hazard ratio (AHR) and 95% CI for all-cause mortality were estimated from multivariable Cox proportional regression modeling controlling for age, race, year of diagnosis, region of diagnosis, stage, histology, and grade.
TAKEAWAY:
- Overall survival differs between registries: 5-year survival of 53.2% in ACTUR vs 47.7% in matched SEER cohort (log-rank P = .001).
- In the primary adjusted model, ACTUR is associated with a lower risk for all-cause mortality vs SEER (AHR, 0.83; 95% CI, 0.76-0.91; P < .0001).
- Subset results retain lower adjusted risk for death for ACTUR vs SEER among ages 35-49 years (AHR, 0.66; 95% CI, 0.52-0.83; P = .0005), ages ≥ 65 years (AHR, 0.82; 95% CI, 0.70-0.96; P = .016), and stage III cancer (AHR, 0.79; 95% CI, 0.69-0.91; P = .0015).
- Histology-stratified findings show lower adjusted risk for death in ACTUR vs SEER for clear cell carcinoma (AHR, 0.63; 95% CI, 0.43-0.93; P =.02) and for endometrioid and other adenocarcinomas (AHR, 0.68; 95% CI, 0.56-0.81; P < .0001).
IN PRACTICE:
"This study is envisioned to be a stepping stone to further investigations of survival and other cancer health outcomes starting with patients diagnosed between 2014 and 2024 with epithelial carcinoma of the ovary, fallopian tube, or primary peritoneum in the DoD Healthcare System versus the national population or other Healthcare Systems,” wrote the authors of the study. “Dedicated funding and support in the [Military Health System] are needed to invest in infrastructure, technology, security, education, and research.”
SOURCE:
The study was led by Kathleen M. Darcy, PhD, and Christopher M. Tarney, MD, from the Gynecologic Cancer Center of Excellence, Department of Gynecologic Surgery & Obstetrics, Uniformed Services University, Walter Reed National Military Medical Center in Bethesda, Maryland. It was published online in Military Medicine.
LIMITATIONS:
The retrospective cohort study design limits causal inference. Although groups were balanced by age, race, year, and region of diagnosis, other demographic factors and socioeconomic variables such as patient comorbidities, educational attainment, household income, and health insurance status were not available and may have affected results. The databases fundamentally differ in how data are acquired, with ACTUR following hospital-based Facility Oncology Registry Data Standards and SEER being a national population-based registry, potentially affecting data quality, consistency, and reliability of survival outcome comparisons. The inclusion of patients diagnosed only through 2013 represents a limitation as it does not allow for contemporary evaluation of survival outcomes, particularly given advances over the past decade including maximal cytoreductive effort to no residual disease, increased adoption of neoadjuvant chemotherapy, and introduction of targeted maintenance agents. The study could not incorporate details regarding residual disease status or control for specifics regarding surgical and medical management, including primary vs interval debulking surgery or the type and timing of agents utilized in first-line, maintenance, and recurrent disease settings. Data regarding circulating biomarkers including CA125, molecular subtypes or alterations, and stratification by homologous recombination deficiency vs proficiency status were not available. Epithelial carcinomas of the fallopian tube and primary peritoneum were excluded from this study, which now are commonly incorporated with ovarian carcinomas. Results may not be generalizable to other populations given the unique characteristics of the Military Health System beneficiary population.
DISCLOSURES:
This research received funding from the Uniformed Services University from the Defense Health Program to the Henry M. Jackson Foundation for the Advancement of Military Medicine Inc., including award HU0001-18-2-0032 to the Murtha Cancer Center Research Program and awards HU0001-19-2-0031 and HU0001-24-2-0047 to the Gynecologic Cancer Center of Excellence Program. All coauthors disclosed no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
Military Women Survive Ovarian Cancer at Higher Rates
Military Women Survive Ovarian Cancer at Higher Rates
No Survival Gain With Adjuvant Therapy in Stage III Melanoma
Offering adjuvant therapy to patients with stage III melanoma offers no melanoma-specific or overall survival benefit, reveals extended follow-up from the first population-based national study to estimate the impact of the treatment.
Hildur Helgadottir, MD, PhD, presented the new findings at the 22nd European Association of Dermato-Oncology (EADO) Congress 2026 on April 24 and described the lead-up to the latest update on the study.
To investigate the impact of adjuvant treatment in patients with stage III melanoma, researchers initially conducted a study in which they used the Swedish Melanoma Registry (SweMR) to identify a precohort of those treated before the introduction of adjuvant therapy in 2018 and a postcohort of those treated subsequently, following both groups out to 2023, she explained.
The analysis revealed no significant difference in melanoma-specific survival between the two groups, at a hazard ratio of 0.92, nor in overall survival, at a hazard ratio of 0.93 (P = .60 for both). However, median follow-up differed between the groups, at 69 months vs 39 months for the precohort vs the postcohort.
Helgadottir, who is a senior research specialist at the Karolinska Comprehensive Cancer Center in Stockholm, Sweden, said that when the earlier results were presented at the European Society for Medical Oncology 2024, there was some criticism that the follow-up was not long enough and that there was no information on the actual adjuvant treatment received in the postcohort patients.
The researchers therefore extended their study out to 2024 to increase the median follow-up to 60 months vs 92 months in the postcohort group vs the precohort group.
They also focused patient selection on patients aged less than 75 years because exposure to adjuvant therapy in older patients was low and restricted the analysis to sentinel lymph node-positive stage IIIB-D cutaneous melanoma diagnosed between 2016 and 2020. This was because adjuvant exposure in stage IIIA disease was low, and patients with clinically detected stage III melanoma started to receive neoadjuvant therapy from 2022 onward.
The current analysis, which was recently published in the European Journal of Cancer, involved 287 patients in the precohort and 349 in the postcohort, who had a median age of 60.0 years and 61.0 years, respectively, and of whom 62.0% and 60.5%, respectively, were male. The groups were well balanced in terms of baseline disease characteristics.
Helgadottir explained that 73% of patients in the postcohort received some form of adjuvant treatment, with the majority treated with PD-1 inhibitors, and a smaller proportion given B-Raf serine-threonine kinase inhibitors. The main reasons for not giving adjuvant therapy were favorable tumor characteristics and the presence of comorbidities.
Five-year melanoma-specific survival rates in the precohorts and postcohorts were 71.4% vs 73.2%, at a hazard ratio adjusted for age, sex, and American Joint Committee on Cancer stage of 1.01 (P = .931). Five-year overall survival rates were 67.3% vs 70.1%, at an adjusted hazard ratio of 0.96 (P = .791).
Helgadottir showed that there were also no significant survival differences in any of the prespecified subgroups for neither melanoma-specific nor overall survival.
There were, again, no significant differences in survival outcomes between the two patient groups, she reported.
The latest results are similar to those from another study conducted in Netherlands and a Danish analysis, Helgadottir said.
Taken together, and “considering the side effects and the costs, it is possible that we will go back to closely following up our patients and treating only at relapse,” she said, “and optimally, of course, that will be already in the neoadjuvant setting.”
“And of course we will need biomarkers because there could be some patients that really need adjuvant treatment, but we need to identify these patients,” continued Helgadottir. Overall survival results from KEYNOTE-054, which compares pembrolizumab with placebo after resection of high-risk stage III melanoma, are awaited, she continued.
Helgadottir explained that adjuvant treatment for stage III melanoma was approved in Sweden in 2018, with treatments freely available to all Swedish residents.
The SweMR is a population-based national register that has near-complete and detailed data on primary cutaneous melanomas, including nodal status and satellite and in-transit disease, and is linked to the national Cause of Death Registry. Helgadottir noted, however, that the SweMR does not contain any information on relapses or the nature of the oncologic treatment received by patients with melanoma.
Following her presentation, she was challenged by an audience member as to whether, on the basis of her findings, she would go back to following up with patients and treating at relapses.
“Maybe we should do that and believe in our own data, and we do. But still, the gold standard must always be the randomized clinical trial,” Helgadottir responded. “So I think, although that we believe in this data, we also want to see the results of the randomized studies.”
The audience member commented that she can see in the data from her own institution that they treat fewer and fewer patients with melanoma with adjuvant therapy by discussing it more thoroughly and being stricter on who should receive it.
Helgadottir agreed, adding that “based on this experience, we did not introduce it to stage II patients because it’s always harder to go back” once a group of patients has started to receive a treatment.
The research was supported by Regional Cancer Centres in Sweden and with grants from the Swedish Cancer Society, Region Stockholm, and the Cancer Research Funds of Radiumhemmet. Helgadottir declared having relationships with Bristol Myers Squibb, Merck Sharp & Dohme, Pierre Fabre, and Novartis.
The trial was supported by SkinVision. The researchers declared having no relevant financial relationships.
This article was previously published by Medscape.
Offering adjuvant therapy to patients with stage III melanoma offers no melanoma-specific or overall survival benefit, reveals extended follow-up from the first population-based national study to estimate the impact of the treatment.
Hildur Helgadottir, MD, PhD, presented the new findings at the 22nd European Association of Dermato-Oncology (EADO) Congress 2026 on April 24 and described the lead-up to the latest update on the study.
To investigate the impact of adjuvant treatment in patients with stage III melanoma, researchers initially conducted a study in which they used the Swedish Melanoma Registry (SweMR) to identify a precohort of those treated before the introduction of adjuvant therapy in 2018 and a postcohort of those treated subsequently, following both groups out to 2023, she explained.
The analysis revealed no significant difference in melanoma-specific survival between the two groups, at a hazard ratio of 0.92, nor in overall survival, at a hazard ratio of 0.93 (P = .60 for both). However, median follow-up differed between the groups, at 69 months vs 39 months for the precohort vs the postcohort.
Helgadottir, who is a senior research specialist at the Karolinska Comprehensive Cancer Center in Stockholm, Sweden, said that when the earlier results were presented at the European Society for Medical Oncology 2024, there was some criticism that the follow-up was not long enough and that there was no information on the actual adjuvant treatment received in the postcohort patients.
The researchers therefore extended their study out to 2024 to increase the median follow-up to 60 months vs 92 months in the postcohort group vs the precohort group.
They also focused patient selection on patients aged less than 75 years because exposure to adjuvant therapy in older patients was low and restricted the analysis to sentinel lymph node-positive stage IIIB-D cutaneous melanoma diagnosed between 2016 and 2020. This was because adjuvant exposure in stage IIIA disease was low, and patients with clinically detected stage III melanoma started to receive neoadjuvant therapy from 2022 onward.
The current analysis, which was recently published in the European Journal of Cancer, involved 287 patients in the precohort and 349 in the postcohort, who had a median age of 60.0 years and 61.0 years, respectively, and of whom 62.0% and 60.5%, respectively, were male. The groups were well balanced in terms of baseline disease characteristics.
Helgadottir explained that 73% of patients in the postcohort received some form of adjuvant treatment, with the majority treated with PD-1 inhibitors, and a smaller proportion given B-Raf serine-threonine kinase inhibitors. The main reasons for not giving adjuvant therapy were favorable tumor characteristics and the presence of comorbidities.
Five-year melanoma-specific survival rates in the precohorts and postcohorts were 71.4% vs 73.2%, at a hazard ratio adjusted for age, sex, and American Joint Committee on Cancer stage of 1.01 (P = .931). Five-year overall survival rates were 67.3% vs 70.1%, at an adjusted hazard ratio of 0.96 (P = .791).
Helgadottir showed that there were also no significant survival differences in any of the prespecified subgroups for neither melanoma-specific nor overall survival.
There were, again, no significant differences in survival outcomes between the two patient groups, she reported.
The latest results are similar to those from another study conducted in Netherlands and a Danish analysis, Helgadottir said.
Taken together, and “considering the side effects and the costs, it is possible that we will go back to closely following up our patients and treating only at relapse,” she said, “and optimally, of course, that will be already in the neoadjuvant setting.”
“And of course we will need biomarkers because there could be some patients that really need adjuvant treatment, but we need to identify these patients,” continued Helgadottir. Overall survival results from KEYNOTE-054, which compares pembrolizumab with placebo after resection of high-risk stage III melanoma, are awaited, she continued.
Helgadottir explained that adjuvant treatment for stage III melanoma was approved in Sweden in 2018, with treatments freely available to all Swedish residents.
The SweMR is a population-based national register that has near-complete and detailed data on primary cutaneous melanomas, including nodal status and satellite and in-transit disease, and is linked to the national Cause of Death Registry. Helgadottir noted, however, that the SweMR does not contain any information on relapses or the nature of the oncologic treatment received by patients with melanoma.
Following her presentation, she was challenged by an audience member as to whether, on the basis of her findings, she would go back to following up with patients and treating at relapses.
“Maybe we should do that and believe in our own data, and we do. But still, the gold standard must always be the randomized clinical trial,” Helgadottir responded. “So I think, although that we believe in this data, we also want to see the results of the randomized studies.”
The audience member commented that she can see in the data from her own institution that they treat fewer and fewer patients with melanoma with adjuvant therapy by discussing it more thoroughly and being stricter on who should receive it.
Helgadottir agreed, adding that “based on this experience, we did not introduce it to stage II patients because it’s always harder to go back” once a group of patients has started to receive a treatment.
The research was supported by Regional Cancer Centres in Sweden and with grants from the Swedish Cancer Society, Region Stockholm, and the Cancer Research Funds of Radiumhemmet. Helgadottir declared having relationships with Bristol Myers Squibb, Merck Sharp & Dohme, Pierre Fabre, and Novartis.
The trial was supported by SkinVision. The researchers declared having no relevant financial relationships.
This article was previously published by Medscape.
Offering adjuvant therapy to patients with stage III melanoma offers no melanoma-specific or overall survival benefit, reveals extended follow-up from the first population-based national study to estimate the impact of the treatment.
Hildur Helgadottir, MD, PhD, presented the new findings at the 22nd European Association of Dermato-Oncology (EADO) Congress 2026 on April 24 and described the lead-up to the latest update on the study.
To investigate the impact of adjuvant treatment in patients with stage III melanoma, researchers initially conducted a study in which they used the Swedish Melanoma Registry (SweMR) to identify a precohort of those treated before the introduction of adjuvant therapy in 2018 and a postcohort of those treated subsequently, following both groups out to 2023, she explained.
The analysis revealed no significant difference in melanoma-specific survival between the two groups, at a hazard ratio of 0.92, nor in overall survival, at a hazard ratio of 0.93 (P = .60 for both). However, median follow-up differed between the groups, at 69 months vs 39 months for the precohort vs the postcohort.
Helgadottir, who is a senior research specialist at the Karolinska Comprehensive Cancer Center in Stockholm, Sweden, said that when the earlier results were presented at the European Society for Medical Oncology 2024, there was some criticism that the follow-up was not long enough and that there was no information on the actual adjuvant treatment received in the postcohort patients.
The researchers therefore extended their study out to 2024 to increase the median follow-up to 60 months vs 92 months in the postcohort group vs the precohort group.
They also focused patient selection on patients aged less than 75 years because exposure to adjuvant therapy in older patients was low and restricted the analysis to sentinel lymph node-positive stage IIIB-D cutaneous melanoma diagnosed between 2016 and 2020. This was because adjuvant exposure in stage IIIA disease was low, and patients with clinically detected stage III melanoma started to receive neoadjuvant therapy from 2022 onward.
The current analysis, which was recently published in the European Journal of Cancer, involved 287 patients in the precohort and 349 in the postcohort, who had a median age of 60.0 years and 61.0 years, respectively, and of whom 62.0% and 60.5%, respectively, were male. The groups were well balanced in terms of baseline disease characteristics.
Helgadottir explained that 73% of patients in the postcohort received some form of adjuvant treatment, with the majority treated with PD-1 inhibitors, and a smaller proportion given B-Raf serine-threonine kinase inhibitors. The main reasons for not giving adjuvant therapy were favorable tumor characteristics and the presence of comorbidities.
Five-year melanoma-specific survival rates in the precohorts and postcohorts were 71.4% vs 73.2%, at a hazard ratio adjusted for age, sex, and American Joint Committee on Cancer stage of 1.01 (P = .931). Five-year overall survival rates were 67.3% vs 70.1%, at an adjusted hazard ratio of 0.96 (P = .791).
Helgadottir showed that there were also no significant survival differences in any of the prespecified subgroups for neither melanoma-specific nor overall survival.
There were, again, no significant differences in survival outcomes between the two patient groups, she reported.
The latest results are similar to those from another study conducted in Netherlands and a Danish analysis, Helgadottir said.
Taken together, and “considering the side effects and the costs, it is possible that we will go back to closely following up our patients and treating only at relapse,” she said, “and optimally, of course, that will be already in the neoadjuvant setting.”
“And of course we will need biomarkers because there could be some patients that really need adjuvant treatment, but we need to identify these patients,” continued Helgadottir. Overall survival results from KEYNOTE-054, which compares pembrolizumab with placebo after resection of high-risk stage III melanoma, are awaited, she continued.
Helgadottir explained that adjuvant treatment for stage III melanoma was approved in Sweden in 2018, with treatments freely available to all Swedish residents.
The SweMR is a population-based national register that has near-complete and detailed data on primary cutaneous melanomas, including nodal status and satellite and in-transit disease, and is linked to the national Cause of Death Registry. Helgadottir noted, however, that the SweMR does not contain any information on relapses or the nature of the oncologic treatment received by patients with melanoma.
Following her presentation, she was challenged by an audience member as to whether, on the basis of her findings, she would go back to following up with patients and treating at relapses.
“Maybe we should do that and believe in our own data, and we do. But still, the gold standard must always be the randomized clinical trial,” Helgadottir responded. “So I think, although that we believe in this data, we also want to see the results of the randomized studies.”
The audience member commented that she can see in the data from her own institution that they treat fewer and fewer patients with melanoma with adjuvant therapy by discussing it more thoroughly and being stricter on who should receive it.
Helgadottir agreed, adding that “based on this experience, we did not introduce it to stage II patients because it’s always harder to go back” once a group of patients has started to receive a treatment.
The research was supported by Regional Cancer Centres in Sweden and with grants from the Swedish Cancer Society, Region Stockholm, and the Cancer Research Funds of Radiumhemmet. Helgadottir declared having relationships with Bristol Myers Squibb, Merck Sharp & Dohme, Pierre Fabre, and Novartis.
The trial was supported by SkinVision. The researchers declared having no relevant financial relationships.
This article was previously published by Medscape.
Wildfire Smoke Linked to Potential Risks for Some Cancers
Wildfire smoke exposure may be associated with increased risks for multiple types of cancer, suggests an analysis of prospective cohort data from over 90,000 individuals.
To determine how this widespread pollution might be affecting cancer risk, senior author Shuguang Leng, MBBS, PhD, and colleagues analyzed data from the Prostate, Lung, Colorectal, and Ovarian (PLCO) Cancer Screening Trial. That prospective national study enrolled approximately 154,000 participants between 1993 and 2001 and tracked cancer incidence through 2018. Of these, 91,460 participants had wildfire smoke exposure data and were included in the analysis.
During the 2006-2018 exposure period, the investigators identified incident cases of 242 ovarian, 800 colorectal, 896 bladder, 1696 hematopoietic, 1739 breast, and 1758 lung cancers, as well as 1127 melanoma cases. The median 36-month moving average for wildfire smoke PM2.5 (fine particulate matter) across the cohort was 0.37 µg/m3.
Wildfire smoke exposure was significantly associated with increased risks for lung, colorectal, breast, bladder, and hematopoietic cancer, according to the results of the study presented by Leng at American Association for Cancer Research (AACR) Annual Meeting 2026.
Each 1 µg/m3 increase in the 36-month moving average of wildfire smoke PM2.5 was associated with a 63% higher risk for hematopoietic cancer (HR, 1.63; 95% CI, 1.02-2.60), a nearly twofold higher risk for lung cancer (hazard ratio [HR], 1.92; 95% CI, 1.18-3.15), more than twofold higher risks for breast cancer (HR, 2.09; 95% CI, 1.34-3.26) and colorectal cancer (HR, 2.31; 95% CI, 1.11-4.81), and a more than threefold higher risk for bladder cancer (HR, 3.49; 95% CI, 1.66-7.34). No significant associations were observed for ovarian cancer or melanoma.
The investigators quantified wildfire smoke exposure at each participant’s residence on a monthly basis using three measures: near-ground wildfire smoke PM2.5, wildfire smoke black carbon, and satellite-derived wildfire smoke plume-day counts, with measurements available from 2006 until first cancer diagnosis or last contact.
Given evidence that 3 years of air pollution exposure can influence the development of epidermal growth factor receptor-positive lung adenocarcinoma, the team modeled exposure as a time-varying variable using 36-month moving averages preceding each month. HRs were estimated using Cox proportional hazards models stratified by study center, with restricted cubic splines applied to evaluate dose-response relationships. Models were adjusted for age, sex, race and ethnicity, education, smoking history, BMI, and trial arm.
All five cancer types linked with wildfire smoke exposure showed linear dose-response relationships, Leng noted, “which means the higher the exposure, the higher the cancer risk.”
Results based on wildfire smoke plume-day counts were generally consistent with those for PM2.5, while associations for black carbon exposure were observed only for breast and bladder cancers.
With wildfires on the rise, these findings suggest that the resulting smoke may become a “major driver for cancer burden in the US in the coming decades,” said Leng, of the University of New Mexico Comprehensive Cancer Center, Albuquerque, New Mexico.
“Wildfire smoke has become a major source of air pollution in the United States,” he continued. Large fires in the US are three times more common than they were 50 years ago, and the “tons of toxicants and particles” released by these fires “can travel hundreds of miles to affect communities far away.”
The investigators also conducted histology-specific analyses, finding that adenocarcinoma showed the strongest association with wildfire smoke among lung cancer subtypes. Among colorectal cancers, proximal tumors appeared more sensitive to wildfire smoke exposure, while among bladder cancers, the association was strongest for muscle-invasive disease.
Wildfire Smoke Exposure Expected to Rise
Under even the most conservative climate projections, wildfire smoke exposure in the US is expected to rise over the next 20-30 years, Leng said.
Annual average wildfire smoke PM2.5 levels, currently estimated at around 0.5 µg/m3, could rise to 1 µg/m3. Based on the study’s dose-response data, this would correspond to substantially greater cancer risk.
There will be “a much larger area” of the US exposed “at a much higher dose,” Leng predicted.
Mitigating the Risks of Wildfire Smoke
This is a “strong hypothesis-generating study,” Jun Wu, PhD, professor of environmental and occupational health at the UC Irvine Program in Public Health, Irvine, California, told Medscape Medical News.
“This is one of the first large, prospective US cohort studies to examine wildfire smoke specifically in relation to cancer risk, especially cancer sites beyond the lung,” Wu said. “A major strength is that the PLCO platform has around 91,000 participants with longitudinal follow-up and detailed covariate data, including smoking history, which is often a weak point in previous air pollution-cancer studies.”
According to Wu, who was not involved in the analysis but recently published data linking wildfire smoke exposure to preterm birth, the reported risks for colorectal, breast, bladder, and hematopoietic cancers represent novel contributions to the literature. However, she cautioned against viewing the specific HRs as a precise estimates of risk due to wide confidence intervals.
The findings should encourage individuals, public health officials, and clinicians to mitigate the risks of wildfire smoke, Wu said.
Specifically, she suggested that public health assessments expand beyond acute outcomes like emergency department visits to include long-term endpoints such as cancer, while community clean-air shelters need to be made more widely available.
She advised clinicians to incorporate wildfire exposure into routine patient histories and to provide vulnerable patients — such as those with asthma, chronic obstructive pulmonary disease, heart failure, or pregnancy — with smoke-season action plans.
Risk mitigation begins with awareness, according to Wu, who advised individuals check their local air quality index on AirNow.gov or PurpleAir.
On smoky days, she suggested prioritizing indoor air quality by keeping windows closed and running air purifiers. If going outside on such days is necessary, she suggested an N95 or KN95 mask, as these offer “meaningful protection,” while cloth and surgical masks do not.
These preventive steps may have once been out of the ordinary, Wu said, but the risk for wildfire smoke exposure is becoming a part of everyday life.
“The common thread is a shift in framing,” Wu said. “Wildfire smoke has traditionally been treated as an acute event, but the emerging evidence points to a chronic environmental exposure. Both our clinical and public health systems have room to grow into that reality.”
The analysis was funded by the National Institutes of Health. The investigators and Wu reported having no conflicts of interest.
This article was previously published on Medscape.
Wildfire smoke exposure may be associated with increased risks for multiple types of cancer, suggests an analysis of prospective cohort data from over 90,000 individuals.
To determine how this widespread pollution might be affecting cancer risk, senior author Shuguang Leng, MBBS, PhD, and colleagues analyzed data from the Prostate, Lung, Colorectal, and Ovarian (PLCO) Cancer Screening Trial. That prospective national study enrolled approximately 154,000 participants between 1993 and 2001 and tracked cancer incidence through 2018. Of these, 91,460 participants had wildfire smoke exposure data and were included in the analysis.
During the 2006-2018 exposure period, the investigators identified incident cases of 242 ovarian, 800 colorectal, 896 bladder, 1696 hematopoietic, 1739 breast, and 1758 lung cancers, as well as 1127 melanoma cases. The median 36-month moving average for wildfire smoke PM2.5 (fine particulate matter) across the cohort was 0.37 µg/m3.
Wildfire smoke exposure was significantly associated with increased risks for lung, colorectal, breast, bladder, and hematopoietic cancer, according to the results of the study presented by Leng at American Association for Cancer Research (AACR) Annual Meeting 2026.
Each 1 µg/m3 increase in the 36-month moving average of wildfire smoke PM2.5 was associated with a 63% higher risk for hematopoietic cancer (HR, 1.63; 95% CI, 1.02-2.60), a nearly twofold higher risk for lung cancer (hazard ratio [HR], 1.92; 95% CI, 1.18-3.15), more than twofold higher risks for breast cancer (HR, 2.09; 95% CI, 1.34-3.26) and colorectal cancer (HR, 2.31; 95% CI, 1.11-4.81), and a more than threefold higher risk for bladder cancer (HR, 3.49; 95% CI, 1.66-7.34). No significant associations were observed for ovarian cancer or melanoma.
The investigators quantified wildfire smoke exposure at each participant’s residence on a monthly basis using three measures: near-ground wildfire smoke PM2.5, wildfire smoke black carbon, and satellite-derived wildfire smoke plume-day counts, with measurements available from 2006 until first cancer diagnosis or last contact.
Given evidence that 3 years of air pollution exposure can influence the development of epidermal growth factor receptor-positive lung adenocarcinoma, the team modeled exposure as a time-varying variable using 36-month moving averages preceding each month. HRs were estimated using Cox proportional hazards models stratified by study center, with restricted cubic splines applied to evaluate dose-response relationships. Models were adjusted for age, sex, race and ethnicity, education, smoking history, BMI, and trial arm.
All five cancer types linked with wildfire smoke exposure showed linear dose-response relationships, Leng noted, “which means the higher the exposure, the higher the cancer risk.”
Results based on wildfire smoke plume-day counts were generally consistent with those for PM2.5, while associations for black carbon exposure were observed only for breast and bladder cancers.
With wildfires on the rise, these findings suggest that the resulting smoke may become a “major driver for cancer burden in the US in the coming decades,” said Leng, of the University of New Mexico Comprehensive Cancer Center, Albuquerque, New Mexico.
“Wildfire smoke has become a major source of air pollution in the United States,” he continued. Large fires in the US are three times more common than they were 50 years ago, and the “tons of toxicants and particles” released by these fires “can travel hundreds of miles to affect communities far away.”
The investigators also conducted histology-specific analyses, finding that adenocarcinoma showed the strongest association with wildfire smoke among lung cancer subtypes. Among colorectal cancers, proximal tumors appeared more sensitive to wildfire smoke exposure, while among bladder cancers, the association was strongest for muscle-invasive disease.
Wildfire Smoke Exposure Expected to Rise
Under even the most conservative climate projections, wildfire smoke exposure in the US is expected to rise over the next 20-30 years, Leng said.
Annual average wildfire smoke PM2.5 levels, currently estimated at around 0.5 µg/m3, could rise to 1 µg/m3. Based on the study’s dose-response data, this would correspond to substantially greater cancer risk.
There will be “a much larger area” of the US exposed “at a much higher dose,” Leng predicted.
Mitigating the Risks of Wildfire Smoke
This is a “strong hypothesis-generating study,” Jun Wu, PhD, professor of environmental and occupational health at the UC Irvine Program in Public Health, Irvine, California, told Medscape Medical News.
“This is one of the first large, prospective US cohort studies to examine wildfire smoke specifically in relation to cancer risk, especially cancer sites beyond the lung,” Wu said. “A major strength is that the PLCO platform has around 91,000 participants with longitudinal follow-up and detailed covariate data, including smoking history, which is often a weak point in previous air pollution-cancer studies.”
According to Wu, who was not involved in the analysis but recently published data linking wildfire smoke exposure to preterm birth, the reported risks for colorectal, breast, bladder, and hematopoietic cancers represent novel contributions to the literature. However, she cautioned against viewing the specific HRs as a precise estimates of risk due to wide confidence intervals.
The findings should encourage individuals, public health officials, and clinicians to mitigate the risks of wildfire smoke, Wu said.
Specifically, she suggested that public health assessments expand beyond acute outcomes like emergency department visits to include long-term endpoints such as cancer, while community clean-air shelters need to be made more widely available.
She advised clinicians to incorporate wildfire exposure into routine patient histories and to provide vulnerable patients — such as those with asthma, chronic obstructive pulmonary disease, heart failure, or pregnancy — with smoke-season action plans.
Risk mitigation begins with awareness, according to Wu, who advised individuals check their local air quality index on AirNow.gov or PurpleAir.
On smoky days, she suggested prioritizing indoor air quality by keeping windows closed and running air purifiers. If going outside on such days is necessary, she suggested an N95 or KN95 mask, as these offer “meaningful protection,” while cloth and surgical masks do not.
These preventive steps may have once been out of the ordinary, Wu said, but the risk for wildfire smoke exposure is becoming a part of everyday life.
“The common thread is a shift in framing,” Wu said. “Wildfire smoke has traditionally been treated as an acute event, but the emerging evidence points to a chronic environmental exposure. Both our clinical and public health systems have room to grow into that reality.”
The analysis was funded by the National Institutes of Health. The investigators and Wu reported having no conflicts of interest.
This article was previously published on Medscape.
Wildfire smoke exposure may be associated with increased risks for multiple types of cancer, suggests an analysis of prospective cohort data from over 90,000 individuals.
To determine how this widespread pollution might be affecting cancer risk, senior author Shuguang Leng, MBBS, PhD, and colleagues analyzed data from the Prostate, Lung, Colorectal, and Ovarian (PLCO) Cancer Screening Trial. That prospective national study enrolled approximately 154,000 participants between 1993 and 2001 and tracked cancer incidence through 2018. Of these, 91,460 participants had wildfire smoke exposure data and were included in the analysis.
During the 2006-2018 exposure period, the investigators identified incident cases of 242 ovarian, 800 colorectal, 896 bladder, 1696 hematopoietic, 1739 breast, and 1758 lung cancers, as well as 1127 melanoma cases. The median 36-month moving average for wildfire smoke PM2.5 (fine particulate matter) across the cohort was 0.37 µg/m3.
Wildfire smoke exposure was significantly associated with increased risks for lung, colorectal, breast, bladder, and hematopoietic cancer, according to the results of the study presented by Leng at American Association for Cancer Research (AACR) Annual Meeting 2026.
Each 1 µg/m3 increase in the 36-month moving average of wildfire smoke PM2.5 was associated with a 63% higher risk for hematopoietic cancer (HR, 1.63; 95% CI, 1.02-2.60), a nearly twofold higher risk for lung cancer (hazard ratio [HR], 1.92; 95% CI, 1.18-3.15), more than twofold higher risks for breast cancer (HR, 2.09; 95% CI, 1.34-3.26) and colorectal cancer (HR, 2.31; 95% CI, 1.11-4.81), and a more than threefold higher risk for bladder cancer (HR, 3.49; 95% CI, 1.66-7.34). No significant associations were observed for ovarian cancer or melanoma.
The investigators quantified wildfire smoke exposure at each participant’s residence on a monthly basis using three measures: near-ground wildfire smoke PM2.5, wildfire smoke black carbon, and satellite-derived wildfire smoke plume-day counts, with measurements available from 2006 until first cancer diagnosis or last contact.
Given evidence that 3 years of air pollution exposure can influence the development of epidermal growth factor receptor-positive lung adenocarcinoma, the team modeled exposure as a time-varying variable using 36-month moving averages preceding each month. HRs were estimated using Cox proportional hazards models stratified by study center, with restricted cubic splines applied to evaluate dose-response relationships. Models were adjusted for age, sex, race and ethnicity, education, smoking history, BMI, and trial arm.
All five cancer types linked with wildfire smoke exposure showed linear dose-response relationships, Leng noted, “which means the higher the exposure, the higher the cancer risk.”
Results based on wildfire smoke plume-day counts were generally consistent with those for PM2.5, while associations for black carbon exposure were observed only for breast and bladder cancers.
With wildfires on the rise, these findings suggest that the resulting smoke may become a “major driver for cancer burden in the US in the coming decades,” said Leng, of the University of New Mexico Comprehensive Cancer Center, Albuquerque, New Mexico.
“Wildfire smoke has become a major source of air pollution in the United States,” he continued. Large fires in the US are three times more common than they were 50 years ago, and the “tons of toxicants and particles” released by these fires “can travel hundreds of miles to affect communities far away.”
The investigators also conducted histology-specific analyses, finding that adenocarcinoma showed the strongest association with wildfire smoke among lung cancer subtypes. Among colorectal cancers, proximal tumors appeared more sensitive to wildfire smoke exposure, while among bladder cancers, the association was strongest for muscle-invasive disease.
Wildfire Smoke Exposure Expected to Rise
Under even the most conservative climate projections, wildfire smoke exposure in the US is expected to rise over the next 20-30 years, Leng said.
Annual average wildfire smoke PM2.5 levels, currently estimated at around 0.5 µg/m3, could rise to 1 µg/m3. Based on the study’s dose-response data, this would correspond to substantially greater cancer risk.
There will be “a much larger area” of the US exposed “at a much higher dose,” Leng predicted.
Mitigating the Risks of Wildfire Smoke
This is a “strong hypothesis-generating study,” Jun Wu, PhD, professor of environmental and occupational health at the UC Irvine Program in Public Health, Irvine, California, told Medscape Medical News.
“This is one of the first large, prospective US cohort studies to examine wildfire smoke specifically in relation to cancer risk, especially cancer sites beyond the lung,” Wu said. “A major strength is that the PLCO platform has around 91,000 participants with longitudinal follow-up and detailed covariate data, including smoking history, which is often a weak point in previous air pollution-cancer studies.”
According to Wu, who was not involved in the analysis but recently published data linking wildfire smoke exposure to preterm birth, the reported risks for colorectal, breast, bladder, and hematopoietic cancers represent novel contributions to the literature. However, she cautioned against viewing the specific HRs as a precise estimates of risk due to wide confidence intervals.
The findings should encourage individuals, public health officials, and clinicians to mitigate the risks of wildfire smoke, Wu said.
Specifically, she suggested that public health assessments expand beyond acute outcomes like emergency department visits to include long-term endpoints such as cancer, while community clean-air shelters need to be made more widely available.
She advised clinicians to incorporate wildfire exposure into routine patient histories and to provide vulnerable patients — such as those with asthma, chronic obstructive pulmonary disease, heart failure, or pregnancy — with smoke-season action plans.
Risk mitigation begins with awareness, according to Wu, who advised individuals check their local air quality index on AirNow.gov or PurpleAir.
On smoky days, she suggested prioritizing indoor air quality by keeping windows closed and running air purifiers. If going outside on such days is necessary, she suggested an N95 or KN95 mask, as these offer “meaningful protection,” while cloth and surgical masks do not.
These preventive steps may have once been out of the ordinary, Wu said, but the risk for wildfire smoke exposure is becoming a part of everyday life.
“The common thread is a shift in framing,” Wu said. “Wildfire smoke has traditionally been treated as an acute event, but the emerging evidence points to a chronic environmental exposure. Both our clinical and public health systems have room to grow into that reality.”
The analysis was funded by the National Institutes of Health. The investigators and Wu reported having no conflicts of interest.
This article was previously published on Medscape.
Many Veterans With H&N Cancer Face Access, Equity Barriers
TOPLINE: In 75,453 veterans with head and neck squamous cell carcinoma (HNSCC), 36.4% live in rural or highly rural areas and 32.0% have a national area deprivation index (ADI) ≥ 75. The average drive time to the nearest tertiary or complex US Department of Veterans Affairs (VA) facility is 94 minutes, highlighting potential access and equity barriers related to rurality, deprivation, and distance.
METHODOLOGY:
A retrospective descriptive study using nationwide VA data from fiscal years 2012 to 2022 identified 75,453 veterans with head and neck squamous cell carcinoma (HNSCC) by International Classification of Diseases, Ninth Revision and International Classification of Diseases, Tenth Revision codes.
Patients were grouped into 5 primary tumor subsites: oral cavity, oropharynx, hypopharynx, larynx, and nasopharynx.
Rurality was classified using Rural-Urban Commuting Area-derived urban, rural, and highly rural scores; socioeconomic disadvantage was measured with national ADI scores.
Travel burden was assessed using time and distance to the nearest primary, secondary, and tertiary VA facilities.
TAKEAWAY:
Oropharyngeal cancer (OPC) cases among veterans increased from 26.3% in 2012 to 46.0% in 2022, while laryngeal cancers decreased from 41.2% to 29.3%.
HNSCCs locations included 35.6% in the larynx, 34.4% in the oropharynx, 22.6% in the oral cavity 3.7% in the hypopharynx, and 3.7% in the nasopharynx.
Veterans with OPC were younger than non-OPC patients and more likely to be White; > 70% were current or former smokers.
IN PRACTICE: “Understanding the geographic and socioeconomic landscape of veterans with HNSCC will allow us to tease out the factors associated with poor outcomes and ultimately design interventions that target high-risk veteran populations to improve overall health outcomes,” the authors argued.
SOURCE: The study was led by researchers at the Veterans Affairs Pittsburgh Healthcare System. It was published online in Head & Neck.
LIMITATIONS: The inability to extract accurate data from a large dataset, challenges in obtaining tumor stage information due to varying documentation practices across physicians and treatment courses, and the inability to assess HPV or p16 data for the cohort represents significant limitations that may have impacted interpretation of results. Clinical outcome measures and cause of death assessment were limited in this national database, affecting the ability to draw conclusions regarding the impact of rurality, area deprivation, and travel time on outcomes.
DISCLOSURES: Chad Brenner reported holding several patents related to the development and use of circulating tumor DNA tests in patients with HNSCC. Jose P. Zevallos disclosed being the founder, equity shareholder, and board member of Droplet Biosciences and Echogenesis Therapeutics, serving as chief scientific advisor and shareholder of Vine Medical, and acting as a consultant for Merck and Johnson & Johnson. Matthew E. Spector reported serving as a consultant for Hologic. Kristen L. Zayan, Jennifer L. McCoy, Monique Y. Boudreaux-Kelly, Zachary Hahn, John Hotchkiss, and Jessica H. Maxwell declared no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
TOPLINE: In 75,453 veterans with head and neck squamous cell carcinoma (HNSCC), 36.4% live in rural or highly rural areas and 32.0% have a national area deprivation index (ADI) ≥ 75. The average drive time to the nearest tertiary or complex US Department of Veterans Affairs (VA) facility is 94 minutes, highlighting potential access and equity barriers related to rurality, deprivation, and distance.
METHODOLOGY:
A retrospective descriptive study using nationwide VA data from fiscal years 2012 to 2022 identified 75,453 veterans with head and neck squamous cell carcinoma (HNSCC) by International Classification of Diseases, Ninth Revision and International Classification of Diseases, Tenth Revision codes.
Patients were grouped into 5 primary tumor subsites: oral cavity, oropharynx, hypopharynx, larynx, and nasopharynx.
Rurality was classified using Rural-Urban Commuting Area-derived urban, rural, and highly rural scores; socioeconomic disadvantage was measured with national ADI scores.
Travel burden was assessed using time and distance to the nearest primary, secondary, and tertiary VA facilities.
TAKEAWAY:
Oropharyngeal cancer (OPC) cases among veterans increased from 26.3% in 2012 to 46.0% in 2022, while laryngeal cancers decreased from 41.2% to 29.3%.
HNSCCs locations included 35.6% in the larynx, 34.4% in the oropharynx, 22.6% in the oral cavity 3.7% in the hypopharynx, and 3.7% in the nasopharynx.
Veterans with OPC were younger than non-OPC patients and more likely to be White; > 70% were current or former smokers.
IN PRACTICE: “Understanding the geographic and socioeconomic landscape of veterans with HNSCC will allow us to tease out the factors associated with poor outcomes and ultimately design interventions that target high-risk veteran populations to improve overall health outcomes,” the authors argued.
SOURCE: The study was led by researchers at the Veterans Affairs Pittsburgh Healthcare System. It was published online in Head & Neck.
LIMITATIONS: The inability to extract accurate data from a large dataset, challenges in obtaining tumor stage information due to varying documentation practices across physicians and treatment courses, and the inability to assess HPV or p16 data for the cohort represents significant limitations that may have impacted interpretation of results. Clinical outcome measures and cause of death assessment were limited in this national database, affecting the ability to draw conclusions regarding the impact of rurality, area deprivation, and travel time on outcomes.
DISCLOSURES: Chad Brenner reported holding several patents related to the development and use of circulating tumor DNA tests in patients with HNSCC. Jose P. Zevallos disclosed being the founder, equity shareholder, and board member of Droplet Biosciences and Echogenesis Therapeutics, serving as chief scientific advisor and shareholder of Vine Medical, and acting as a consultant for Merck and Johnson & Johnson. Matthew E. Spector reported serving as a consultant for Hologic. Kristen L. Zayan, Jennifer L. McCoy, Monique Y. Boudreaux-Kelly, Zachary Hahn, John Hotchkiss, and Jessica H. Maxwell declared no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
TOPLINE: In 75,453 veterans with head and neck squamous cell carcinoma (HNSCC), 36.4% live in rural or highly rural areas and 32.0% have a national area deprivation index (ADI) ≥ 75. The average drive time to the nearest tertiary or complex US Department of Veterans Affairs (VA) facility is 94 minutes, highlighting potential access and equity barriers related to rurality, deprivation, and distance.
METHODOLOGY:
A retrospective descriptive study using nationwide VA data from fiscal years 2012 to 2022 identified 75,453 veterans with head and neck squamous cell carcinoma (HNSCC) by International Classification of Diseases, Ninth Revision and International Classification of Diseases, Tenth Revision codes.
Patients were grouped into 5 primary tumor subsites: oral cavity, oropharynx, hypopharynx, larynx, and nasopharynx.
Rurality was classified using Rural-Urban Commuting Area-derived urban, rural, and highly rural scores; socioeconomic disadvantage was measured with national ADI scores.
Travel burden was assessed using time and distance to the nearest primary, secondary, and tertiary VA facilities.
TAKEAWAY:
Oropharyngeal cancer (OPC) cases among veterans increased from 26.3% in 2012 to 46.0% in 2022, while laryngeal cancers decreased from 41.2% to 29.3%.
HNSCCs locations included 35.6% in the larynx, 34.4% in the oropharynx, 22.6% in the oral cavity 3.7% in the hypopharynx, and 3.7% in the nasopharynx.
Veterans with OPC were younger than non-OPC patients and more likely to be White; > 70% were current or former smokers.
IN PRACTICE: “Understanding the geographic and socioeconomic landscape of veterans with HNSCC will allow us to tease out the factors associated with poor outcomes and ultimately design interventions that target high-risk veteran populations to improve overall health outcomes,” the authors argued.
SOURCE: The study was led by researchers at the Veterans Affairs Pittsburgh Healthcare System. It was published online in Head & Neck.
LIMITATIONS: The inability to extract accurate data from a large dataset, challenges in obtaining tumor stage information due to varying documentation practices across physicians and treatment courses, and the inability to assess HPV or p16 data for the cohort represents significant limitations that may have impacted interpretation of results. Clinical outcome measures and cause of death assessment were limited in this national database, affecting the ability to draw conclusions regarding the impact of rurality, area deprivation, and travel time on outcomes.
DISCLOSURES: Chad Brenner reported holding several patents related to the development and use of circulating tumor DNA tests in patients with HNSCC. Jose P. Zevallos disclosed being the founder, equity shareholder, and board member of Droplet Biosciences and Echogenesis Therapeutics, serving as chief scientific advisor and shareholder of Vine Medical, and acting as a consultant for Merck and Johnson & Johnson. Matthew E. Spector reported serving as a consultant for Hologic. Kristen L. Zayan, Jennifer L. McCoy, Monique Y. Boudreaux-Kelly, Zachary Hahn, John Hotchkiss, and Jessica H. Maxwell declared no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
Does Marital Status Affect Cancer Risk?
Adults who have never been married had a higher cancer risk than their married or previously married peers, with patterns observed across many major cancer types and particularly strong for cancers linked to infections, smoking, and reproductive factors, new data suggest.
The findings are based on a large, population-based cancer registry analysis of more than 4 million cases, making it the largest study of its kind in the US.
First author Paulo Pinheiro, PhD, cautioned, however, that the study does not suggest that marriage itself is protective.
"As with any observational study, we cannot establish causation, and unmeasured factors may contribute to the associations,” said Pinheiro, with Sylvester Comprehensive Cancer Center, University of Miami Health System, in Miami.
Marital status may, however, help identify groups with different patterns of cancer risk, which likely reflect social and lifestyle behaviors rather than a direct causal effect, Pinheiro explained.
Married individuals, for instance, are less likely to smoke — a known cancer risk factor — and more likely to have children and undergo cancer screening, which can influence cancer incidence through reproductive effects and screening, including earlier detection and removal of precancerous lesions.
"Marital status is therefore best understood as a marker of those accumulated factors," Pinheiro said.
The study was published online on April 8 in Cancer Research Communications.
Filling a Data Gap
Marriage has consistently been associated with earlier cancer diagnosis and improved survival among those with cancer, but its relationship to cancer incidence remains less clear.
To address that gap, researchers analyzed data from 12 US states that included demographic and cancer information for more than 4.2 million cancer cases diagnosed between 2015 and 2022.
The analysis included more than 500 million person-years at risk in adults 30 years or older, representing an annual population of more than 62 million. The never-married group comprised about 19% of the total population — 22% were men and 17% were women.
Compared with ever-married individuals, never-married men and women had higher cancer incidence across many major cancer types, racial and ethnic groups, and age groups.
Overall, cancer rates were about 68% higher in never-married men and 85% higher in never-married women compared with their ever-married counterparts (incidence rate ratios [IRRs], 1.68 and 1.85, respectively).
Never-married Black men had the highest overall cancer rates (1600 per 100,000), whereas married Black men had significantly lower rates than married White men (752.6 vs 836.2 per 100,000), suggesting complex interactions between marital status and structural factors, the researchers noted.
Site-specific patterns revealed clues to potential mechanisms linking marital status and cancer.
Compared with ever-married individuals, never-married people had the highest excess risks for human papillomavirus-related cancers — about five times higher for anal cancer in men (IRR, 5.04) and approaching three times higher for cervical cancer in women (IRR, 2.64).
Other strong associations between never-married individuals and cancer risk were observed for smoking-related cancers, including lung (IRR, 2.1 for both men and women) and esophageal cancers (IRR, 2.4 in men and 2.7 in women), and malignancies including liver (IRR, 2.3 for both men and women), bladder (IRR, 2.3 women only), and colorectal (IRR, 2.1 women only) cancers.
Among women, the higher incidence of ovarian and uterine cancers (IRR, 2.4 for both) among the never-married group supports the influence of reproductive mechanisms, such as giving birth, on cancer risk.
The association between marital status and cancer risk was weaker for breast, prostate, and thyroid cancers (with IRRs < 2), suggesting potentially less modifiable etiologies.
Overall, “methodologically, it is quite robust, particularly in its clear framing of ever- vs never-married individuals and the use of standardized incidence rates and regression modeling,” Pinheiro said.
The analysis did not adjust for individual-level risk factors such as smoking, diet, physical activity, or alcohol use — factors that may partly explain the observed associations.
Adjusting for these lifestyle and health behavior factors at the individual level would require detailed information on these behaviors, and “data at that level simply do not exist at a national scale,” Pinheiro said. It would also “obscure the real-world pattern we are trying to measure.”
Gilbert Welch, MD, noted that adjusting for these individual-level cancer risk factors “would certainly attenuate the associations.”
“That said, it wouldn’t be crazy to suggest marriage drives some of these risk factors,” said Welch, general internist and senior investigator at the Center for Surgery and Public Health, Brigham and Women’s Hospital, Boston. Married couples benefit from combined incomes and shared expenses, and “may well help support individuals in making healthy choices (like not smoking).”
But, he added, “it would be crazy to suggest that the reason to get married is to lower cancer risk.”
The authors flagged a study limitation — the fact that ever-married status lumps together people who are currently married, divorced, and widowed, and these groups may have different risk profiles. Additionally, “individuals in strained or abusive marriages may not experience protective social benefits,” while those in long-term cohabiting relationships classified as never-married may experience high levels of support, the authors wrote.
Overall, though, Pinheiro clarified that the main finding is “not about marriage as a causal agent, but about identifying a large population group, the never-married, with a consistently higher cancer burden that has been largely overlooked in public health practice and cancer prevention efforts.”
Linda Waite, professor, Department of Sociology, University of Chicago, who wasn’t involved in the study, wasn’t surprised by the findings. For men, not having a spouse may “disadvantage” them in ways that might increase cancer risk.
Unmarried men are more likely to drink and smoke heavily, which increase cancer risk, she said. A spouse may also influence health awareness and decisions, such as noticing suspicious symptoms, pushing their partner to see a doctor, or helping manage their partner’s care.
Plus, “for both men and women, having a spouse may improve medical care by giving each partner a companion for medical appointments and another person to help manage risks of disease,” Waite said.
The study had no commercial funding. Pinheiro and Waite had no relevant disclosures.
A version of this article first appeared on Medscape.com.
Adults who have never been married had a higher cancer risk than their married or previously married peers, with patterns observed across many major cancer types and particularly strong for cancers linked to infections, smoking, and reproductive factors, new data suggest.
The findings are based on a large, population-based cancer registry analysis of more than 4 million cases, making it the largest study of its kind in the US.
First author Paulo Pinheiro, PhD, cautioned, however, that the study does not suggest that marriage itself is protective.
"As with any observational study, we cannot establish causation, and unmeasured factors may contribute to the associations,” said Pinheiro, with Sylvester Comprehensive Cancer Center, University of Miami Health System, in Miami.
Marital status may, however, help identify groups with different patterns of cancer risk, which likely reflect social and lifestyle behaviors rather than a direct causal effect, Pinheiro explained.
Married individuals, for instance, are less likely to smoke — a known cancer risk factor — and more likely to have children and undergo cancer screening, which can influence cancer incidence through reproductive effects and screening, including earlier detection and removal of precancerous lesions.
"Marital status is therefore best understood as a marker of those accumulated factors," Pinheiro said.
The study was published online on April 8 in Cancer Research Communications.
Filling a Data Gap
Marriage has consistently been associated with earlier cancer diagnosis and improved survival among those with cancer, but its relationship to cancer incidence remains less clear.
To address that gap, researchers analyzed data from 12 US states that included demographic and cancer information for more than 4.2 million cancer cases diagnosed between 2015 and 2022.
The analysis included more than 500 million person-years at risk in adults 30 years or older, representing an annual population of more than 62 million. The never-married group comprised about 19% of the total population — 22% were men and 17% were women.
Compared with ever-married individuals, never-married men and women had higher cancer incidence across many major cancer types, racial and ethnic groups, and age groups.
Overall, cancer rates were about 68% higher in never-married men and 85% higher in never-married women compared with their ever-married counterparts (incidence rate ratios [IRRs], 1.68 and 1.85, respectively).
Never-married Black men had the highest overall cancer rates (1600 per 100,000), whereas married Black men had significantly lower rates than married White men (752.6 vs 836.2 per 100,000), suggesting complex interactions between marital status and structural factors, the researchers noted.
Site-specific patterns revealed clues to potential mechanisms linking marital status and cancer.
Compared with ever-married individuals, never-married people had the highest excess risks for human papillomavirus-related cancers — about five times higher for anal cancer in men (IRR, 5.04) and approaching three times higher for cervical cancer in women (IRR, 2.64).
Other strong associations between never-married individuals and cancer risk were observed for smoking-related cancers, including lung (IRR, 2.1 for both men and women) and esophageal cancers (IRR, 2.4 in men and 2.7 in women), and malignancies including liver (IRR, 2.3 for both men and women), bladder (IRR, 2.3 women only), and colorectal (IRR, 2.1 women only) cancers.
Among women, the higher incidence of ovarian and uterine cancers (IRR, 2.4 for both) among the never-married group supports the influence of reproductive mechanisms, such as giving birth, on cancer risk.
The association between marital status and cancer risk was weaker for breast, prostate, and thyroid cancers (with IRRs < 2), suggesting potentially less modifiable etiologies.
Overall, “methodologically, it is quite robust, particularly in its clear framing of ever- vs never-married individuals and the use of standardized incidence rates and regression modeling,” Pinheiro said.
The analysis did not adjust for individual-level risk factors such as smoking, diet, physical activity, or alcohol use — factors that may partly explain the observed associations.
Adjusting for these lifestyle and health behavior factors at the individual level would require detailed information on these behaviors, and “data at that level simply do not exist at a national scale,” Pinheiro said. It would also “obscure the real-world pattern we are trying to measure.”
Gilbert Welch, MD, noted that adjusting for these individual-level cancer risk factors “would certainly attenuate the associations.”
“That said, it wouldn’t be crazy to suggest marriage drives some of these risk factors,” said Welch, general internist and senior investigator at the Center for Surgery and Public Health, Brigham and Women’s Hospital, Boston. Married couples benefit from combined incomes and shared expenses, and “may well help support individuals in making healthy choices (like not smoking).”
But, he added, “it would be crazy to suggest that the reason to get married is to lower cancer risk.”
The authors flagged a study limitation — the fact that ever-married status lumps together people who are currently married, divorced, and widowed, and these groups may have different risk profiles. Additionally, “individuals in strained or abusive marriages may not experience protective social benefits,” while those in long-term cohabiting relationships classified as never-married may experience high levels of support, the authors wrote.
Overall, though, Pinheiro clarified that the main finding is “not about marriage as a causal agent, but about identifying a large population group, the never-married, with a consistently higher cancer burden that has been largely overlooked in public health practice and cancer prevention efforts.”
Linda Waite, professor, Department of Sociology, University of Chicago, who wasn’t involved in the study, wasn’t surprised by the findings. For men, not having a spouse may “disadvantage” them in ways that might increase cancer risk.
Unmarried men are more likely to drink and smoke heavily, which increase cancer risk, she said. A spouse may also influence health awareness and decisions, such as noticing suspicious symptoms, pushing their partner to see a doctor, or helping manage their partner’s care.
Plus, “for both men and women, having a spouse may improve medical care by giving each partner a companion for medical appointments and another person to help manage risks of disease,” Waite said.
The study had no commercial funding. Pinheiro and Waite had no relevant disclosures.
A version of this article first appeared on Medscape.com.
Adults who have never been married had a higher cancer risk than their married or previously married peers, with patterns observed across many major cancer types and particularly strong for cancers linked to infections, smoking, and reproductive factors, new data suggest.
The findings are based on a large, population-based cancer registry analysis of more than 4 million cases, making it the largest study of its kind in the US.
First author Paulo Pinheiro, PhD, cautioned, however, that the study does not suggest that marriage itself is protective.
"As with any observational study, we cannot establish causation, and unmeasured factors may contribute to the associations,” said Pinheiro, with Sylvester Comprehensive Cancer Center, University of Miami Health System, in Miami.
Marital status may, however, help identify groups with different patterns of cancer risk, which likely reflect social and lifestyle behaviors rather than a direct causal effect, Pinheiro explained.
Married individuals, for instance, are less likely to smoke — a known cancer risk factor — and more likely to have children and undergo cancer screening, which can influence cancer incidence through reproductive effects and screening, including earlier detection and removal of precancerous lesions.
"Marital status is therefore best understood as a marker of those accumulated factors," Pinheiro said.
The study was published online on April 8 in Cancer Research Communications.
Filling a Data Gap
Marriage has consistently been associated with earlier cancer diagnosis and improved survival among those with cancer, but its relationship to cancer incidence remains less clear.
To address that gap, researchers analyzed data from 12 US states that included demographic and cancer information for more than 4.2 million cancer cases diagnosed between 2015 and 2022.
The analysis included more than 500 million person-years at risk in adults 30 years or older, representing an annual population of more than 62 million. The never-married group comprised about 19% of the total population — 22% were men and 17% were women.
Compared with ever-married individuals, never-married men and women had higher cancer incidence across many major cancer types, racial and ethnic groups, and age groups.
Overall, cancer rates were about 68% higher in never-married men and 85% higher in never-married women compared with their ever-married counterparts (incidence rate ratios [IRRs], 1.68 and 1.85, respectively).
Never-married Black men had the highest overall cancer rates (1600 per 100,000), whereas married Black men had significantly lower rates than married White men (752.6 vs 836.2 per 100,000), suggesting complex interactions between marital status and structural factors, the researchers noted.
Site-specific patterns revealed clues to potential mechanisms linking marital status and cancer.
Compared with ever-married individuals, never-married people had the highest excess risks for human papillomavirus-related cancers — about five times higher for anal cancer in men (IRR, 5.04) and approaching three times higher for cervical cancer in women (IRR, 2.64).
Other strong associations between never-married individuals and cancer risk were observed for smoking-related cancers, including lung (IRR, 2.1 for both men and women) and esophageal cancers (IRR, 2.4 in men and 2.7 in women), and malignancies including liver (IRR, 2.3 for both men and women), bladder (IRR, 2.3 women only), and colorectal (IRR, 2.1 women only) cancers.
Among women, the higher incidence of ovarian and uterine cancers (IRR, 2.4 for both) among the never-married group supports the influence of reproductive mechanisms, such as giving birth, on cancer risk.
The association between marital status and cancer risk was weaker for breast, prostate, and thyroid cancers (with IRRs < 2), suggesting potentially less modifiable etiologies.
Overall, “methodologically, it is quite robust, particularly in its clear framing of ever- vs never-married individuals and the use of standardized incidence rates and regression modeling,” Pinheiro said.
The analysis did not adjust for individual-level risk factors such as smoking, diet, physical activity, or alcohol use — factors that may partly explain the observed associations.
Adjusting for these lifestyle and health behavior factors at the individual level would require detailed information on these behaviors, and “data at that level simply do not exist at a national scale,” Pinheiro said. It would also “obscure the real-world pattern we are trying to measure.”
Gilbert Welch, MD, noted that adjusting for these individual-level cancer risk factors “would certainly attenuate the associations.”
“That said, it wouldn’t be crazy to suggest marriage drives some of these risk factors,” said Welch, general internist and senior investigator at the Center for Surgery and Public Health, Brigham and Women’s Hospital, Boston. Married couples benefit from combined incomes and shared expenses, and “may well help support individuals in making healthy choices (like not smoking).”
But, he added, “it would be crazy to suggest that the reason to get married is to lower cancer risk.”
The authors flagged a study limitation — the fact that ever-married status lumps together people who are currently married, divorced, and widowed, and these groups may have different risk profiles. Additionally, “individuals in strained or abusive marriages may not experience protective social benefits,” while those in long-term cohabiting relationships classified as never-married may experience high levels of support, the authors wrote.
Overall, though, Pinheiro clarified that the main finding is “not about marriage as a causal agent, but about identifying a large population group, the never-married, with a consistently higher cancer burden that has been largely overlooked in public health practice and cancer prevention efforts.”
Linda Waite, professor, Department of Sociology, University of Chicago, who wasn’t involved in the study, wasn’t surprised by the findings. For men, not having a spouse may “disadvantage” them in ways that might increase cancer risk.
Unmarried men are more likely to drink and smoke heavily, which increase cancer risk, she said. A spouse may also influence health awareness and decisions, such as noticing suspicious symptoms, pushing their partner to see a doctor, or helping manage their partner’s care.
Plus, “for both men and women, having a spouse may improve medical care by giving each partner a companion for medical appointments and another person to help manage risks of disease,” Waite said.
The study had no commercial funding. Pinheiro and Waite had no relevant disclosures.
A version of this article first appeared on Medscape.com.
VHA CRC Screening Has Blind Spots, Disparities
TOPLINE:
More than 1 in 8 colorectal cancer (CRC) cases among veterans occur outside the standard screening age of 50-75 years or those with high-risk personal or family history. High-risk patients face > 6 times the risk for CRC compared with average-risk patients aged 50-75 years who are up to date with screening, while Black patients have > 50% higher risk compared with White patients.
METHODOLOGY:
Researchers conducted a case-control analysis using Veterans Health Administration (VHA) Corporate Data Warehouse data from 2012-2018 at 2 sites: Veterans Affairs (VA) New York Harbor Health Care System and VA Puget Sound Health Care System.
Participants included 3714 cases among veterans with CRC matched to 14,856 controls (4:1), with matching on age (± 3 years), sex, and facility site; each control was used once.
Screening categories included 5 groups by age (50-75 years vs < 50 years or > 75 years), screening up-to-date status, and high-risk status (inflammatory bowel disease, hereditary cancer syndromes, or family history).
CRC screening was considered up to date if US Preventive Services Task Force-recommended tests were completed on time (colonoscopy ≤ 10 years; guaiac-based fecal occult blood test or fecal immunochemical test ≤ 1 year).
TAKEAWAY:
Compared with category 1 (age 50-75 years and up-to-date with screening), CRC was associated with category 4 (age < 50 years or > 75 years and not up to date) (odds ratio [OR], 1.40; 95% CI, 1.11-1.78), and category 5 (high risk) (OR, 6.23; 95% CI, 5.06-7.66).
Race and comorbidity associations included higher CRC risk for Black vs White patients (OR, 1.54; 95% CI, 1.37-1.73), and higher CRC risk with diabetes (OR, 1.65; 95% CI, 1.51-1.81) and alcohol use disorder (OR, 1.53; 95% CI, 1.35-1.73).
Among 3714 CRC cases, 71.1% occurred in individuals aged 50-75 years not up to date with screening.
A total of 12.5% of CRC cases occurred in people outside age 50-75 or with high-risk personal or family history, suggesting that conventional screening-adherence metrics may miss a clinically relevant minority.
IN PRACTICE:
“The conventional measure of CRC screening, focused on average-risk individuals aged 50 to 75, does not reflect screening status in an important minority of CRC patients," the authors wrote.
SOURCE:
The study was led by researchers at NYU Grossman School of Medicine and Veterans Affairs New York Harbor Health Care System, and published online July 9, 2026 in Medicine.
LIMITATIONS:
The study population consisted predominantly of male veterans (97.1%), who tend to be older and have more comorbidities compared with the US population, which may limit the generalizability of findings to other populations. Researchers defined screening status cross-sectionally relative to a single point in time rather than assessing longitudinal screening adherence, which may not fully capture the consistency of screening over time that is likely important for defining CRC risk. Veterans may receive screening at non-VA medical facilities, potentially leading to incomplete documentation of screening status and important covariates such as race, ethnicity, and comorbidities. The possibility of residual confounding cannot be excluded despite adjustment for multiple risk factors in the analysis.
DISCLOSURES:
This study received support from NIH grant K08 CA230162 and the AGA Caroline Craig Augustyn & Damian Augustyn Award in Digestive Cancer, both awarded to Peter S. Liang. Liang disclosed receiving research support from Freenome and serving on the advisory boards for Guardant Health and Natera. The remaining authors reported no funding or conflicts of interest to disclose.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
TOPLINE:
More than 1 in 8 colorectal cancer (CRC) cases among veterans occur outside the standard screening age of 50-75 years or those with high-risk personal or family history. High-risk patients face > 6 times the risk for CRC compared with average-risk patients aged 50-75 years who are up to date with screening, while Black patients have > 50% higher risk compared with White patients.
METHODOLOGY:
Researchers conducted a case-control analysis using Veterans Health Administration (VHA) Corporate Data Warehouse data from 2012-2018 at 2 sites: Veterans Affairs (VA) New York Harbor Health Care System and VA Puget Sound Health Care System.
Participants included 3714 cases among veterans with CRC matched to 14,856 controls (4:1), with matching on age (± 3 years), sex, and facility site; each control was used once.
Screening categories included 5 groups by age (50-75 years vs < 50 years or > 75 years), screening up-to-date status, and high-risk status (inflammatory bowel disease, hereditary cancer syndromes, or family history).
CRC screening was considered up to date if US Preventive Services Task Force-recommended tests were completed on time (colonoscopy ≤ 10 years; guaiac-based fecal occult blood test or fecal immunochemical test ≤ 1 year).
TAKEAWAY:
Compared with category 1 (age 50-75 years and up-to-date with screening), CRC was associated with category 4 (age < 50 years or > 75 years and not up to date) (odds ratio [OR], 1.40; 95% CI, 1.11-1.78), and category 5 (high risk) (OR, 6.23; 95% CI, 5.06-7.66).
Race and comorbidity associations included higher CRC risk for Black vs White patients (OR, 1.54; 95% CI, 1.37-1.73), and higher CRC risk with diabetes (OR, 1.65; 95% CI, 1.51-1.81) and alcohol use disorder (OR, 1.53; 95% CI, 1.35-1.73).
Among 3714 CRC cases, 71.1% occurred in individuals aged 50-75 years not up to date with screening.
A total of 12.5% of CRC cases occurred in people outside age 50-75 or with high-risk personal or family history, suggesting that conventional screening-adherence metrics may miss a clinically relevant minority.
IN PRACTICE:
“The conventional measure of CRC screening, focused on average-risk individuals aged 50 to 75, does not reflect screening status in an important minority of CRC patients," the authors wrote.
SOURCE:
The study was led by researchers at NYU Grossman School of Medicine and Veterans Affairs New York Harbor Health Care System, and published online July 9, 2026 in Medicine.
LIMITATIONS:
The study population consisted predominantly of male veterans (97.1%), who tend to be older and have more comorbidities compared with the US population, which may limit the generalizability of findings to other populations. Researchers defined screening status cross-sectionally relative to a single point in time rather than assessing longitudinal screening adherence, which may not fully capture the consistency of screening over time that is likely important for defining CRC risk. Veterans may receive screening at non-VA medical facilities, potentially leading to incomplete documentation of screening status and important covariates such as race, ethnicity, and comorbidities. The possibility of residual confounding cannot be excluded despite adjustment for multiple risk factors in the analysis.
DISCLOSURES:
This study received support from NIH grant K08 CA230162 and the AGA Caroline Craig Augustyn & Damian Augustyn Award in Digestive Cancer, both awarded to Peter S. Liang. Liang disclosed receiving research support from Freenome and serving on the advisory boards for Guardant Health and Natera. The remaining authors reported no funding or conflicts of interest to disclose.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
TOPLINE:
More than 1 in 8 colorectal cancer (CRC) cases among veterans occur outside the standard screening age of 50-75 years or those with high-risk personal or family history. High-risk patients face > 6 times the risk for CRC compared with average-risk patients aged 50-75 years who are up to date with screening, while Black patients have > 50% higher risk compared with White patients.
METHODOLOGY:
Researchers conducted a case-control analysis using Veterans Health Administration (VHA) Corporate Data Warehouse data from 2012-2018 at 2 sites: Veterans Affairs (VA) New York Harbor Health Care System and VA Puget Sound Health Care System.
Participants included 3714 cases among veterans with CRC matched to 14,856 controls (4:1), with matching on age (± 3 years), sex, and facility site; each control was used once.
Screening categories included 5 groups by age (50-75 years vs < 50 years or > 75 years), screening up-to-date status, and high-risk status (inflammatory bowel disease, hereditary cancer syndromes, or family history).
CRC screening was considered up to date if US Preventive Services Task Force-recommended tests were completed on time (colonoscopy ≤ 10 years; guaiac-based fecal occult blood test or fecal immunochemical test ≤ 1 year).
TAKEAWAY:
Compared with category 1 (age 50-75 years and up-to-date with screening), CRC was associated with category 4 (age < 50 years or > 75 years and not up to date) (odds ratio [OR], 1.40; 95% CI, 1.11-1.78), and category 5 (high risk) (OR, 6.23; 95% CI, 5.06-7.66).
Race and comorbidity associations included higher CRC risk for Black vs White patients (OR, 1.54; 95% CI, 1.37-1.73), and higher CRC risk with diabetes (OR, 1.65; 95% CI, 1.51-1.81) and alcohol use disorder (OR, 1.53; 95% CI, 1.35-1.73).
Among 3714 CRC cases, 71.1% occurred in individuals aged 50-75 years not up to date with screening.
A total of 12.5% of CRC cases occurred in people outside age 50-75 or with high-risk personal or family history, suggesting that conventional screening-adherence metrics may miss a clinically relevant minority.
IN PRACTICE:
“The conventional measure of CRC screening, focused on average-risk individuals aged 50 to 75, does not reflect screening status in an important minority of CRC patients," the authors wrote.
SOURCE:
The study was led by researchers at NYU Grossman School of Medicine and Veterans Affairs New York Harbor Health Care System, and published online July 9, 2026 in Medicine.
LIMITATIONS:
The study population consisted predominantly of male veterans (97.1%), who tend to be older and have more comorbidities compared with the US population, which may limit the generalizability of findings to other populations. Researchers defined screening status cross-sectionally relative to a single point in time rather than assessing longitudinal screening adherence, which may not fully capture the consistency of screening over time that is likely important for defining CRC risk. Veterans may receive screening at non-VA medical facilities, potentially leading to incomplete documentation of screening status and important covariates such as race, ethnicity, and comorbidities. The possibility of residual confounding cannot be excluded despite adjustment for multiple risk factors in the analysis.
DISCLOSURES:
This study received support from NIH grant K08 CA230162 and the AGA Caroline Craig Augustyn & Damian Augustyn Award in Digestive Cancer, both awarded to Peter S. Liang. Liang disclosed receiving research support from Freenome and serving on the advisory boards for Guardant Health and Natera. The remaining authors reported no funding or conflicts of interest to disclose.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
Hidradenitis Suppurativa Associated With Elevated Risks for Multiple Cancer Types
Hidradenitis Suppurativa Associated With Elevated Risks for Multiple Cancer Types
TOPLINE:
In a meta-analysis, patients with hidradenitis suppurativa (HS) faced a more than 80% higher risk for cancer overall than the general population, with particularly elevated risks for gastrointestinal, head and neck, hematologic, and respiratory system cancers.
METHODOLOGY:
- Researchers conducted a meta-analysis including 11 studies from PubMed, Embase, and Web of Science databases published between 2001 and 2024; these studies examined the risk for cancer in patients with HS compared with that in the general population.
- These studies included 624,721 patients diagnosed with HS (mean age, 33.6-43.8 years) and 393,691,636 control individuals from the general population.
- Researchers performed an inverse variance-weighted random-effects analysis to calculate pooled odds ratios (ORs) for cancer overall and specific cancer subtypes.
- Cancer types were categorized into 11 groups for subgroup analysis: bone and soft tissue cancers, breast cancer, central nervous system cancers, endocrine-related cancers, gastrointestinal cancers, head and neck cancers, hematologic cancers, respiratory system cancers, skin cancers, urogenital cancers, and unspecified cancers.
TAKEAWAY:
- Patients with HS demonstrated a significantly higher risk for cancer overall than control individuals (crude OR, 1.82; P = .018).
- Patients with HS showed an increased risk for gastrointestinal cancers (crude OR, 1.61; P = .0002), head and neck cancers (crude OR, 2.41; P = .00001), hematologic cancers (crude OR, 1.71; P = .00005), and respiratory system cancers (crude OR, 1.81; P = .04).
- Patients with HS demonstrated significantly elevated risks for both Hodgkin lymphoma (OR, 2.44; P = .0001) and non-Hodgkin lymphoma (OR, 1.15; P = .012).
- A non-significant increased risk for skin cancer was observed in patients with HS (crude OR, 1.48; P = .08). No increased risks for bone and soft tissue cancers, central nervous system cancers, breast cancer, or urogenital cancers were observed in patients with HS.
IN PRACTICE:
"HS was associated with an increased overall risk of cancer, including several specific subtypes, compared with controls," the authors wrote, suggesting that "studies are adjusting for confounders and assess long-term associations between HS and cancer risk are highly needed to investigate which factors contribute to this cancer risk."
SOURCE:
This study was led by Daniel Isufi, Department of Dermatology and Allergy, Copenhagen University Hospital-Herlev and Gentofte Hospital, University of Copenhagen, Copenhagen, Denmark. It was published online on March 11, 2026, in Dermatology and Therapy.
LIMITATIONS:
Limited data on cancer subtypes hindered meta-analyses of rare cancers, and the lack of reporting on anti‑inflammatory treatment and disease severity prevented subgroup analyses. Most studies originated from North America, introducing potential geographic bias. No study reported BMI, and ethnicity was poorly documented. Only few studies adjusted for key confounders (smoking, obesity, and alcohol intake), limiting the determination of whether the increased risk for cancer was due to HS itself or shared lifestyle and metabolic factors.
DISCLOSURES:
This study did not receive any funding or sponsorship. Two authors reported receiving research grant funding from the LEO Foundation and having other ties with various other sources.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
TOPLINE:
In a meta-analysis, patients with hidradenitis suppurativa (HS) faced a more than 80% higher risk for cancer overall than the general population, with particularly elevated risks for gastrointestinal, head and neck, hematologic, and respiratory system cancers.
METHODOLOGY:
- Researchers conducted a meta-analysis including 11 studies from PubMed, Embase, and Web of Science databases published between 2001 and 2024; these studies examined the risk for cancer in patients with HS compared with that in the general population.
- These studies included 624,721 patients diagnosed with HS (mean age, 33.6-43.8 years) and 393,691,636 control individuals from the general population.
- Researchers performed an inverse variance-weighted random-effects analysis to calculate pooled odds ratios (ORs) for cancer overall and specific cancer subtypes.
- Cancer types were categorized into 11 groups for subgroup analysis: bone and soft tissue cancers, breast cancer, central nervous system cancers, endocrine-related cancers, gastrointestinal cancers, head and neck cancers, hematologic cancers, respiratory system cancers, skin cancers, urogenital cancers, and unspecified cancers.
TAKEAWAY:
- Patients with HS demonstrated a significantly higher risk for cancer overall than control individuals (crude OR, 1.82; P = .018).
- Patients with HS showed an increased risk for gastrointestinal cancers (crude OR, 1.61; P = .0002), head and neck cancers (crude OR, 2.41; P = .00001), hematologic cancers (crude OR, 1.71; P = .00005), and respiratory system cancers (crude OR, 1.81; P = .04).
- Patients with HS demonstrated significantly elevated risks for both Hodgkin lymphoma (OR, 2.44; P = .0001) and non-Hodgkin lymphoma (OR, 1.15; P = .012).
- A non-significant increased risk for skin cancer was observed in patients with HS (crude OR, 1.48; P = .08). No increased risks for bone and soft tissue cancers, central nervous system cancers, breast cancer, or urogenital cancers were observed in patients with HS.
IN PRACTICE:
"HS was associated with an increased overall risk of cancer, including several specific subtypes, compared with controls," the authors wrote, suggesting that "studies are adjusting for confounders and assess long-term associations between HS and cancer risk are highly needed to investigate which factors contribute to this cancer risk."
SOURCE:
This study was led by Daniel Isufi, Department of Dermatology and Allergy, Copenhagen University Hospital-Herlev and Gentofte Hospital, University of Copenhagen, Copenhagen, Denmark. It was published online on March 11, 2026, in Dermatology and Therapy.
LIMITATIONS:
Limited data on cancer subtypes hindered meta-analyses of rare cancers, and the lack of reporting on anti‑inflammatory treatment and disease severity prevented subgroup analyses. Most studies originated from North America, introducing potential geographic bias. No study reported BMI, and ethnicity was poorly documented. Only few studies adjusted for key confounders (smoking, obesity, and alcohol intake), limiting the determination of whether the increased risk for cancer was due to HS itself or shared lifestyle and metabolic factors.
DISCLOSURES:
This study did not receive any funding or sponsorship. Two authors reported receiving research grant funding from the LEO Foundation and having other ties with various other sources.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
TOPLINE:
In a meta-analysis, patients with hidradenitis suppurativa (HS) faced a more than 80% higher risk for cancer overall than the general population, with particularly elevated risks for gastrointestinal, head and neck, hematologic, and respiratory system cancers.
METHODOLOGY:
- Researchers conducted a meta-analysis including 11 studies from PubMed, Embase, and Web of Science databases published between 2001 and 2024; these studies examined the risk for cancer in patients with HS compared with that in the general population.
- These studies included 624,721 patients diagnosed with HS (mean age, 33.6-43.8 years) and 393,691,636 control individuals from the general population.
- Researchers performed an inverse variance-weighted random-effects analysis to calculate pooled odds ratios (ORs) for cancer overall and specific cancer subtypes.
- Cancer types were categorized into 11 groups for subgroup analysis: bone and soft tissue cancers, breast cancer, central nervous system cancers, endocrine-related cancers, gastrointestinal cancers, head and neck cancers, hematologic cancers, respiratory system cancers, skin cancers, urogenital cancers, and unspecified cancers.
TAKEAWAY:
- Patients with HS demonstrated a significantly higher risk for cancer overall than control individuals (crude OR, 1.82; P = .018).
- Patients with HS showed an increased risk for gastrointestinal cancers (crude OR, 1.61; P = .0002), head and neck cancers (crude OR, 2.41; P = .00001), hematologic cancers (crude OR, 1.71; P = .00005), and respiratory system cancers (crude OR, 1.81; P = .04).
- Patients with HS demonstrated significantly elevated risks for both Hodgkin lymphoma (OR, 2.44; P = .0001) and non-Hodgkin lymphoma (OR, 1.15; P = .012).
- A non-significant increased risk for skin cancer was observed in patients with HS (crude OR, 1.48; P = .08). No increased risks for bone and soft tissue cancers, central nervous system cancers, breast cancer, or urogenital cancers were observed in patients with HS.
IN PRACTICE:
"HS was associated with an increased overall risk of cancer, including several specific subtypes, compared with controls," the authors wrote, suggesting that "studies are adjusting for confounders and assess long-term associations between HS and cancer risk are highly needed to investigate which factors contribute to this cancer risk."
SOURCE:
This study was led by Daniel Isufi, Department of Dermatology and Allergy, Copenhagen University Hospital-Herlev and Gentofte Hospital, University of Copenhagen, Copenhagen, Denmark. It was published online on March 11, 2026, in Dermatology and Therapy.
LIMITATIONS:
Limited data on cancer subtypes hindered meta-analyses of rare cancers, and the lack of reporting on anti‑inflammatory treatment and disease severity prevented subgroup analyses. Most studies originated from North America, introducing potential geographic bias. No study reported BMI, and ethnicity was poorly documented. Only few studies adjusted for key confounders (smoking, obesity, and alcohol intake), limiting the determination of whether the increased risk for cancer was due to HS itself or shared lifestyle and metabolic factors.
DISCLOSURES:
This study did not receive any funding or sponsorship. Two authors reported receiving research grant funding from the LEO Foundation and having other ties with various other sources.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
Hidradenitis Suppurativa Associated With Elevated Risks for Multiple Cancer Types
Hidradenitis Suppurativa Associated With Elevated Risks for Multiple Cancer Types
“Colon Age” Tool Evaluates Early CRC Risk in Male Vets
TOPLINE: Interviews with 23 male veterans (aged 35-49 years) at average-risk for colorectal cancer (CRC) and 8 primary care practitioners (PCPs) found broad acceptability of the Colon Age concept, with 96% of patients agreeing to calculation. PCPs describe its potential use to support screening discussions (fecal immunochemical test [FIT] vs colonoscopy) but emphasize workflow barriers, requesting electronic medical record integration and “time neutral” implementation.
METHODOLOGY:
Researchers conducted semistructured qualitative interviews with 31 participants (23 male veteran patients aged 35-49 years and 8 PCPs) at the Richard L. Roudebush Veterans Affairs Medical Center between June and September 2022.
Patients were eligible if they were at average risk for CRC, had no prior screening (colonoscopy or fecal immunochemical test [FIT]), no inflammatory bowel disease, and no significant family history of CRC.
Interviews explored participants' experiences with CRC screening, understanding of the Colon Age tool, and perceived clinical use.
Audio-recorded interviews were transcribed, deidentified, and analyzed using the constant comparison method with open and focused coding phases until saturation was reached.
TAKEAWAY:
Among 23 male veteran patients (mean age 47 years), 96% agreed to have their Colon Age calculated; 68% had a Colon Age below their biological age, 14% higher than their biological age, and 18% equal to their biological age.
Patients accepted the Colon Age concept, finding it easy to understand and helpful for being informed about their health, though most were unaware of screening options beyond colonoscopy prior to the interview.
The 8 PCPs (mean age 53 years, 50% female, mean 29 years in practice) interviewed found the tool acceptable and useful for screening conversations, improving uptake, and facilitating shared decision-making, particularly in gray zone cases where screening decisions are unclear.
PCPs emphasized the need for the tool to be integrated into the electronic medical record system and expressed concerns about time commitment, consistency with practice guidelines, and the validation process, stating they would only use the tool if it were time neutral and evidence-based.
IN PRACTICE: “Although the age at which to begin colorectal cancer screening in the US was lowered to 45 years in 2018, uptake of screening in persons aged 45 to 49 has been slow,” wrote the authors of the study.
SOURCE:The study was led by researchers at the VA Center for Health Information and Communication. It was published online on July 15 in BMC Primary Care.
LIMITATIONS: The study was conducted at a single VA medical center in the Midwest and all patient participants were male, which may limit generalizability to nonveteran patients, female patients, and non-VA clinicians. The Colon Age tool has limitations, as it was based on a risk prediction model with modest discrimination, and the linkage to screening recommendations was based on arbitrary Surveillance, Epidemiology and End Results thresholds chosen by the tool developers. Additionally, the qualitative nature of the study with a small sample size may not capture the full range of perspectives across diverse health care settings and patient populations.
DISCLOSURES: The primary author received support from Health Services Research and Development, Veterans Administration. Funding for this project was provided by Richard L. Roudebush VA Medical Center Indianapolis, Indiana Center for Health Information, and Communication COIN funds. The authors reported no relevant conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
TOPLINE: Interviews with 23 male veterans (aged 35-49 years) at average-risk for colorectal cancer (CRC) and 8 primary care practitioners (PCPs) found broad acceptability of the Colon Age concept, with 96% of patients agreeing to calculation. PCPs describe its potential use to support screening discussions (fecal immunochemical test [FIT] vs colonoscopy) but emphasize workflow barriers, requesting electronic medical record integration and “time neutral” implementation.
METHODOLOGY:
Researchers conducted semistructured qualitative interviews with 31 participants (23 male veteran patients aged 35-49 years and 8 PCPs) at the Richard L. Roudebush Veterans Affairs Medical Center between June and September 2022.
Patients were eligible if they were at average risk for CRC, had no prior screening (colonoscopy or fecal immunochemical test [FIT]), no inflammatory bowel disease, and no significant family history of CRC.
Interviews explored participants' experiences with CRC screening, understanding of the Colon Age tool, and perceived clinical use.
Audio-recorded interviews were transcribed, deidentified, and analyzed using the constant comparison method with open and focused coding phases until saturation was reached.
TAKEAWAY:
Among 23 male veteran patients (mean age 47 years), 96% agreed to have their Colon Age calculated; 68% had a Colon Age below their biological age, 14% higher than their biological age, and 18% equal to their biological age.
Patients accepted the Colon Age concept, finding it easy to understand and helpful for being informed about their health, though most were unaware of screening options beyond colonoscopy prior to the interview.
The 8 PCPs (mean age 53 years, 50% female, mean 29 years in practice) interviewed found the tool acceptable and useful for screening conversations, improving uptake, and facilitating shared decision-making, particularly in gray zone cases where screening decisions are unclear.
PCPs emphasized the need for the tool to be integrated into the electronic medical record system and expressed concerns about time commitment, consistency with practice guidelines, and the validation process, stating they would only use the tool if it were time neutral and evidence-based.
IN PRACTICE: “Although the age at which to begin colorectal cancer screening in the US was lowered to 45 years in 2018, uptake of screening in persons aged 45 to 49 has been slow,” wrote the authors of the study.
SOURCE:The study was led by researchers at the VA Center for Health Information and Communication. It was published online on July 15 in BMC Primary Care.
LIMITATIONS: The study was conducted at a single VA medical center in the Midwest and all patient participants were male, which may limit generalizability to nonveteran patients, female patients, and non-VA clinicians. The Colon Age tool has limitations, as it was based on a risk prediction model with modest discrimination, and the linkage to screening recommendations was based on arbitrary Surveillance, Epidemiology and End Results thresholds chosen by the tool developers. Additionally, the qualitative nature of the study with a small sample size may not capture the full range of perspectives across diverse health care settings and patient populations.
DISCLOSURES: The primary author received support from Health Services Research and Development, Veterans Administration. Funding for this project was provided by Richard L. Roudebush VA Medical Center Indianapolis, Indiana Center for Health Information, and Communication COIN funds. The authors reported no relevant conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
TOPLINE: Interviews with 23 male veterans (aged 35-49 years) at average-risk for colorectal cancer (CRC) and 8 primary care practitioners (PCPs) found broad acceptability of the Colon Age concept, with 96% of patients agreeing to calculation. PCPs describe its potential use to support screening discussions (fecal immunochemical test [FIT] vs colonoscopy) but emphasize workflow barriers, requesting electronic medical record integration and “time neutral” implementation.
METHODOLOGY:
Researchers conducted semistructured qualitative interviews with 31 participants (23 male veteran patients aged 35-49 years and 8 PCPs) at the Richard L. Roudebush Veterans Affairs Medical Center between June and September 2022.
Patients were eligible if they were at average risk for CRC, had no prior screening (colonoscopy or fecal immunochemical test [FIT]), no inflammatory bowel disease, and no significant family history of CRC.
Interviews explored participants' experiences with CRC screening, understanding of the Colon Age tool, and perceived clinical use.
Audio-recorded interviews were transcribed, deidentified, and analyzed using the constant comparison method with open and focused coding phases until saturation was reached.
TAKEAWAY:
Among 23 male veteran patients (mean age 47 years), 96% agreed to have their Colon Age calculated; 68% had a Colon Age below their biological age, 14% higher than their biological age, and 18% equal to their biological age.
Patients accepted the Colon Age concept, finding it easy to understand and helpful for being informed about their health, though most were unaware of screening options beyond colonoscopy prior to the interview.
The 8 PCPs (mean age 53 years, 50% female, mean 29 years in practice) interviewed found the tool acceptable and useful for screening conversations, improving uptake, and facilitating shared decision-making, particularly in gray zone cases where screening decisions are unclear.
PCPs emphasized the need for the tool to be integrated into the electronic medical record system and expressed concerns about time commitment, consistency with practice guidelines, and the validation process, stating they would only use the tool if it were time neutral and evidence-based.
IN PRACTICE: “Although the age at which to begin colorectal cancer screening in the US was lowered to 45 years in 2018, uptake of screening in persons aged 45 to 49 has been slow,” wrote the authors of the study.
SOURCE:The study was led by researchers at the VA Center for Health Information and Communication. It was published online on July 15 in BMC Primary Care.
LIMITATIONS: The study was conducted at a single VA medical center in the Midwest and all patient participants were male, which may limit generalizability to nonveteran patients, female patients, and non-VA clinicians. The Colon Age tool has limitations, as it was based on a risk prediction model with modest discrimination, and the linkage to screening recommendations was based on arbitrary Surveillance, Epidemiology and End Results thresholds chosen by the tool developers. Additionally, the qualitative nature of the study with a small sample size may not capture the full range of perspectives across diverse health care settings and patient populations.
DISCLOSURES: The primary author received support from Health Services Research and Development, Veterans Administration. Funding for this project was provided by Richard L. Roudebush VA Medical Center Indianapolis, Indiana Center for Health Information, and Communication COIN funds. The authors reported no relevant conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
Veteran Testicular Cancer Survivors Face High Mental Health Burden
Anxiety, depression, and suicide rates are elevated for veterans who are survivors of testicular cancer (TC) compared with veterans without cancer, a retrospective analysis finds.
Over 5 years, the cumulative incidence of anxiety and depression was 53.4% in veterans with TC vs 35.0% in matched controls (P < .001; hazard ratio [HR], 1.66), reported Aditya Bagrodia, MD, professor of urology and radiation oncology at the University of California San Diego, et al in Cancer Medicine. The cumulative incidence of suicidality was 5.0% and 0.1%, respectively (P < .001; HR, 22.99).
“More than half of men with testicular cancer contend with these diagnoses,” Bagrodia told Federal Practitioner. “There are risk factors, including chemotherapy, being single or divorced, or unemployed.”
Patients in these groups warrant aggressive screening and intervention, Bagrodia said. TC is the most common cancer in men in the military and the most common malignancy in men aged 18 to 45 years, Bagrodia said: “The vast majority of men who have testicular cancer are curable.”
Patients, however, face an intense burden.
“One theme that comes up consistently from patients and caregivers is centered around mental health impact, brain fog, anxiety, depression, and difficulty concentrating,” Bagrodia said. “We wanted to dig into this a little bit further. The idea is to shed light on how common these diagnoses are on these young cancer survivors and intervene so we could positively impact their quality of life.”
The study analyzed 2022 patients with TC and 6375 matched controls enrolled at the US Department of Veterans Affairs (VA) from 1990 through 2016. In the cancer cohort, the mean age at diagnosis was 42.46 years, and ages ranged from 18 to 88 years; 89.7% of patients were White, 6.0% were Black, 2.4% were other race, 1.2% were Asian/Pacific Islander, and 0.7% were Native; 6.2% were Hispanic; and 19.9% were diagnosed between 1990 and 1999.
Factors linked to higher rates of anxiety/depression among patients with TC included divorce (HR 1.15, P = .044), unemployment (HR 1.68, P < .001), and receipt of chemotherapy (HR 1.20, P < .001).
The incidence of de novo anxiety/depression was 30.1% for patients with TC vs 16.7% for controls (P < .001), and the incidence of de novo suicidality was 2.4% for patients and 0.1% for controls.
“These are men who are going to beat their cancer and go on to live for decades and decades,” Bagrodia said. “We found that the impact of a diagnosis and chemotherapy can persist beyond the initial time frame.”
It’s not clear, however, why chemotherapy boosts the risk, Bagrodia said. Clinicians who treat patients with TC should let them know that anxiety, depression, and suicidality are common and treatable concerns.
“We've got some wonderful support services, therapy, and medications that can help out with those diagnoses,” Bagrodia said.
The study authors noted limitations such as the retrospective study design and limited consideration of factors that may affect mental health.
“Additionally, the baseline rates of anxiety, depression, and suicidality are high in the VA population, which may limit ability to apply results to the civilian population,” Bagrodia said.
Genitourinary oncologist Philippe Spiess, MD, of Moffitt Cancer Center in Tampa, praised the study in an interview, saying it provides stronger evidence than previous research.
"It's not only about screening but surveillance, because you never know what kind of challenges they have in their lives,” Spiess told Federal Practitioner, emphasizing the need for clinicians to continue to monitor patients. “They're young, they're vulnerable. Don’t assume they're going to get help somewhere else. You need to be that source that facilitates it.”
No funding is reported. Bagrodia and other authors have no disclosures. Spiess has no disclosures.
Anxiety, depression, and suicide rates are elevated for veterans who are survivors of testicular cancer (TC) compared with veterans without cancer, a retrospective analysis finds.
Over 5 years, the cumulative incidence of anxiety and depression was 53.4% in veterans with TC vs 35.0% in matched controls (P < .001; hazard ratio [HR], 1.66), reported Aditya Bagrodia, MD, professor of urology and radiation oncology at the University of California San Diego, et al in Cancer Medicine. The cumulative incidence of suicidality was 5.0% and 0.1%, respectively (P < .001; HR, 22.99).
“More than half of men with testicular cancer contend with these diagnoses,” Bagrodia told Federal Practitioner. “There are risk factors, including chemotherapy, being single or divorced, or unemployed.”
Patients in these groups warrant aggressive screening and intervention, Bagrodia said. TC is the most common cancer in men in the military and the most common malignancy in men aged 18 to 45 years, Bagrodia said: “The vast majority of men who have testicular cancer are curable.”
Patients, however, face an intense burden.
“One theme that comes up consistently from patients and caregivers is centered around mental health impact, brain fog, anxiety, depression, and difficulty concentrating,” Bagrodia said. “We wanted to dig into this a little bit further. The idea is to shed light on how common these diagnoses are on these young cancer survivors and intervene so we could positively impact their quality of life.”
The study analyzed 2022 patients with TC and 6375 matched controls enrolled at the US Department of Veterans Affairs (VA) from 1990 through 2016. In the cancer cohort, the mean age at diagnosis was 42.46 years, and ages ranged from 18 to 88 years; 89.7% of patients were White, 6.0% were Black, 2.4% were other race, 1.2% were Asian/Pacific Islander, and 0.7% were Native; 6.2% were Hispanic; and 19.9% were diagnosed between 1990 and 1999.
Factors linked to higher rates of anxiety/depression among patients with TC included divorce (HR 1.15, P = .044), unemployment (HR 1.68, P < .001), and receipt of chemotherapy (HR 1.20, P < .001).
The incidence of de novo anxiety/depression was 30.1% for patients with TC vs 16.7% for controls (P < .001), and the incidence of de novo suicidality was 2.4% for patients and 0.1% for controls.
“These are men who are going to beat their cancer and go on to live for decades and decades,” Bagrodia said. “We found that the impact of a diagnosis and chemotherapy can persist beyond the initial time frame.”
It’s not clear, however, why chemotherapy boosts the risk, Bagrodia said. Clinicians who treat patients with TC should let them know that anxiety, depression, and suicidality are common and treatable concerns.
“We've got some wonderful support services, therapy, and medications that can help out with those diagnoses,” Bagrodia said.
The study authors noted limitations such as the retrospective study design and limited consideration of factors that may affect mental health.
“Additionally, the baseline rates of anxiety, depression, and suicidality are high in the VA population, which may limit ability to apply results to the civilian population,” Bagrodia said.
Genitourinary oncologist Philippe Spiess, MD, of Moffitt Cancer Center in Tampa, praised the study in an interview, saying it provides stronger evidence than previous research.
"It's not only about screening but surveillance, because you never know what kind of challenges they have in their lives,” Spiess told Federal Practitioner, emphasizing the need for clinicians to continue to monitor patients. “They're young, they're vulnerable. Don’t assume they're going to get help somewhere else. You need to be that source that facilitates it.”
No funding is reported. Bagrodia and other authors have no disclosures. Spiess has no disclosures.
Anxiety, depression, and suicide rates are elevated for veterans who are survivors of testicular cancer (TC) compared with veterans without cancer, a retrospective analysis finds.
Over 5 years, the cumulative incidence of anxiety and depression was 53.4% in veterans with TC vs 35.0% in matched controls (P < .001; hazard ratio [HR], 1.66), reported Aditya Bagrodia, MD, professor of urology and radiation oncology at the University of California San Diego, et al in Cancer Medicine. The cumulative incidence of suicidality was 5.0% and 0.1%, respectively (P < .001; HR, 22.99).
“More than half of men with testicular cancer contend with these diagnoses,” Bagrodia told Federal Practitioner. “There are risk factors, including chemotherapy, being single or divorced, or unemployed.”
Patients in these groups warrant aggressive screening and intervention, Bagrodia said. TC is the most common cancer in men in the military and the most common malignancy in men aged 18 to 45 years, Bagrodia said: “The vast majority of men who have testicular cancer are curable.”
Patients, however, face an intense burden.
“One theme that comes up consistently from patients and caregivers is centered around mental health impact, brain fog, anxiety, depression, and difficulty concentrating,” Bagrodia said. “We wanted to dig into this a little bit further. The idea is to shed light on how common these diagnoses are on these young cancer survivors and intervene so we could positively impact their quality of life.”
The study analyzed 2022 patients with TC and 6375 matched controls enrolled at the US Department of Veterans Affairs (VA) from 1990 through 2016. In the cancer cohort, the mean age at diagnosis was 42.46 years, and ages ranged from 18 to 88 years; 89.7% of patients were White, 6.0% were Black, 2.4% were other race, 1.2% were Asian/Pacific Islander, and 0.7% were Native; 6.2% were Hispanic; and 19.9% were diagnosed between 1990 and 1999.
Factors linked to higher rates of anxiety/depression among patients with TC included divorce (HR 1.15, P = .044), unemployment (HR 1.68, P < .001), and receipt of chemotherapy (HR 1.20, P < .001).
The incidence of de novo anxiety/depression was 30.1% for patients with TC vs 16.7% for controls (P < .001), and the incidence of de novo suicidality was 2.4% for patients and 0.1% for controls.
“These are men who are going to beat their cancer and go on to live for decades and decades,” Bagrodia said. “We found that the impact of a diagnosis and chemotherapy can persist beyond the initial time frame.”
It’s not clear, however, why chemotherapy boosts the risk, Bagrodia said. Clinicians who treat patients with TC should let them know that anxiety, depression, and suicidality are common and treatable concerns.
“We've got some wonderful support services, therapy, and medications that can help out with those diagnoses,” Bagrodia said.
The study authors noted limitations such as the retrospective study design and limited consideration of factors that may affect mental health.
“Additionally, the baseline rates of anxiety, depression, and suicidality are high in the VA population, which may limit ability to apply results to the civilian population,” Bagrodia said.
Genitourinary oncologist Philippe Spiess, MD, of Moffitt Cancer Center in Tampa, praised the study in an interview, saying it provides stronger evidence than previous research.
"It's not only about screening but surveillance, because you never know what kind of challenges they have in their lives,” Spiess told Federal Practitioner, emphasizing the need for clinicians to continue to monitor patients. “They're young, they're vulnerable. Don’t assume they're going to get help somewhere else. You need to be that source that facilitates it.”
No funding is reported. Bagrodia and other authors have no disclosures. Spiess has no disclosures.