User login
To screen or not to screen children for hypertension?
In this issue of JFP, Smith et al recommend following guidelines from the American Academy of Pediatrics to annually screen children for hypertension (see page 220). This recommendation appears to be at odds with the recent US Preventive Services Task Force (USPSTF) statement that concluded there is insufficient evidence for screening children and adolescents for hypertension. But an “I” recommendation from the USPSTF is not the same as a “D” recommendation. “D” means don’t do it, because the evidence indicates that the harms outweigh the benefits. “I” means we don’t have enough evidence to weigh the harms and benefits, so it is up to you and your patients to decide what to do.
So whose recommendations should we follow?
Our decision should be based on a thorough understanding of the evidence, and that evidence is well summarized in the recent USPSTF report.1 The reviewers found no studies that evaluated the benefits and harms of screening children and adolescents for hypertension and no studies evaluating disease outcomes from treating hypertension in these patients.
There is, however, an association between elevated blood pressure in childhood and outcomes such as left ventricular hypertrophy and carotid intimal thickness.2 Some physicians contend that these “disease-oriented outcomes” are sufficient reason to identify and treat hypertension in children and adolescents.3 The USPSTF, however, requires a higher level of evidence that includes patient-oriented outcomes, such as a lower risk of congestive heart failure, renal failure, or death, before recommending treatment. Physicians and patients have to choose what level of evidence is sufficient to take action.
Dr. Smith comments: “As noted in their report, the USPSTF acknowledges that observational studies indicate an association between hypertension in childhood and hypertension in adulthood, but there have been no randomized trials to determine if treating hypertension in children and adolescents reduces risk of cardiovascular events. Although it is a cohort study, not a randomized trial, the ongoing i3C Consortium Outcomes Study4 may provide better information to guide decision-making for children and adolescents with elevated blood pressure.”
What we can all agree on is that, when hypertension is identified in a child or adolescent, it is important to determine if there is a treatable cause of elevated blood pressure such as coarctation of the aorta or renal disease. It is also important to address risk factors for elevated blood pressure and cardiovascular disease, such as obesity, poor dietary habits, and smoking. The treatment is lifestyle modification with diet, exercise, and smoking cessation.
- USPSTF: High blood pressure in children and adolescents: screening. Accessed June 2, 2021. https://uspreventiveservicestaskforce.org/uspstf/recommendation/blood-pressure-in-children-and-adolescents-hypertension-screening
- Yang L, Magnussen CG, Yang L, et al. Elevated blood pressure in childhood or adolescence and cardiovascular outcomes in adulthood: a systematic review. Hypertension. 2020;75:948–955. doi: 10.1161/hypertensionaha.119.14168
- Falkner B, Lurbe E. The USPSTF call to inaction on blood pressure screening in children and adolescents. Pediatr Nephrol. 2021;36:1327-1329. doi: 10.1007/s00467-021-04926-y
- Sinaiko AR, Jacobs DR Jr, Woo JG, et al. The International Childhood Cardiovascular Cohort (i3C) consortium outcomes study of childhood cardiovascular risk factors and adult cardiovascular morbidity and mortality: Design and recruitment. Contemp Clin Trials. 2018;69:55-64. doi: 10.1016/j.cct.2018.04.009
In this issue of JFP, Smith et al recommend following guidelines from the American Academy of Pediatrics to annually screen children for hypertension (see page 220). This recommendation appears to be at odds with the recent US Preventive Services Task Force (USPSTF) statement that concluded there is insufficient evidence for screening children and adolescents for hypertension. But an “I” recommendation from the USPSTF is not the same as a “D” recommendation. “D” means don’t do it, because the evidence indicates that the harms outweigh the benefits. “I” means we don’t have enough evidence to weigh the harms and benefits, so it is up to you and your patients to decide what to do.
So whose recommendations should we follow?
Our decision should be based on a thorough understanding of the evidence, and that evidence is well summarized in the recent USPSTF report.1 The reviewers found no studies that evaluated the benefits and harms of screening children and adolescents for hypertension and no studies evaluating disease outcomes from treating hypertension in these patients.
There is, however, an association between elevated blood pressure in childhood and outcomes such as left ventricular hypertrophy and carotid intimal thickness.2 Some physicians contend that these “disease-oriented outcomes” are sufficient reason to identify and treat hypertension in children and adolescents.3 The USPSTF, however, requires a higher level of evidence that includes patient-oriented outcomes, such as a lower risk of congestive heart failure, renal failure, or death, before recommending treatment. Physicians and patients have to choose what level of evidence is sufficient to take action.
Dr. Smith comments: “As noted in their report, the USPSTF acknowledges that observational studies indicate an association between hypertension in childhood and hypertension in adulthood, but there have been no randomized trials to determine if treating hypertension in children and adolescents reduces risk of cardiovascular events. Although it is a cohort study, not a randomized trial, the ongoing i3C Consortium Outcomes Study4 may provide better information to guide decision-making for children and adolescents with elevated blood pressure.”
What we can all agree on is that, when hypertension is identified in a child or adolescent, it is important to determine if there is a treatable cause of elevated blood pressure such as coarctation of the aorta or renal disease. It is also important to address risk factors for elevated blood pressure and cardiovascular disease, such as obesity, poor dietary habits, and smoking. The treatment is lifestyle modification with diet, exercise, and smoking cessation.
In this issue of JFP, Smith et al recommend following guidelines from the American Academy of Pediatrics to annually screen children for hypertension (see page 220). This recommendation appears to be at odds with the recent US Preventive Services Task Force (USPSTF) statement that concluded there is insufficient evidence for screening children and adolescents for hypertension. But an “I” recommendation from the USPSTF is not the same as a “D” recommendation. “D” means don’t do it, because the evidence indicates that the harms outweigh the benefits. “I” means we don’t have enough evidence to weigh the harms and benefits, so it is up to you and your patients to decide what to do.
So whose recommendations should we follow?
Our decision should be based on a thorough understanding of the evidence, and that evidence is well summarized in the recent USPSTF report.1 The reviewers found no studies that evaluated the benefits and harms of screening children and adolescents for hypertension and no studies evaluating disease outcomes from treating hypertension in these patients.
There is, however, an association between elevated blood pressure in childhood and outcomes such as left ventricular hypertrophy and carotid intimal thickness.2 Some physicians contend that these “disease-oriented outcomes” are sufficient reason to identify and treat hypertension in children and adolescents.3 The USPSTF, however, requires a higher level of evidence that includes patient-oriented outcomes, such as a lower risk of congestive heart failure, renal failure, or death, before recommending treatment. Physicians and patients have to choose what level of evidence is sufficient to take action.
Dr. Smith comments: “As noted in their report, the USPSTF acknowledges that observational studies indicate an association between hypertension in childhood and hypertension in adulthood, but there have been no randomized trials to determine if treating hypertension in children and adolescents reduces risk of cardiovascular events. Although it is a cohort study, not a randomized trial, the ongoing i3C Consortium Outcomes Study4 may provide better information to guide decision-making for children and adolescents with elevated blood pressure.”
What we can all agree on is that, when hypertension is identified in a child or adolescent, it is important to determine if there is a treatable cause of elevated blood pressure such as coarctation of the aorta or renal disease. It is also important to address risk factors for elevated blood pressure and cardiovascular disease, such as obesity, poor dietary habits, and smoking. The treatment is lifestyle modification with diet, exercise, and smoking cessation.
- USPSTF: High blood pressure in children and adolescents: screening. Accessed June 2, 2021. https://uspreventiveservicestaskforce.org/uspstf/recommendation/blood-pressure-in-children-and-adolescents-hypertension-screening
- Yang L, Magnussen CG, Yang L, et al. Elevated blood pressure in childhood or adolescence and cardiovascular outcomes in adulthood: a systematic review. Hypertension. 2020;75:948–955. doi: 10.1161/hypertensionaha.119.14168
- Falkner B, Lurbe E. The USPSTF call to inaction on blood pressure screening in children and adolescents. Pediatr Nephrol. 2021;36:1327-1329. doi: 10.1007/s00467-021-04926-y
- Sinaiko AR, Jacobs DR Jr, Woo JG, et al. The International Childhood Cardiovascular Cohort (i3C) consortium outcomes study of childhood cardiovascular risk factors and adult cardiovascular morbidity and mortality: Design and recruitment. Contemp Clin Trials. 2018;69:55-64. doi: 10.1016/j.cct.2018.04.009
- USPSTF: High blood pressure in children and adolescents: screening. Accessed June 2, 2021. https://uspreventiveservicestaskforce.org/uspstf/recommendation/blood-pressure-in-children-and-adolescents-hypertension-screening
- Yang L, Magnussen CG, Yang L, et al. Elevated blood pressure in childhood or adolescence and cardiovascular outcomes in adulthood: a systematic review. Hypertension. 2020;75:948–955. doi: 10.1161/hypertensionaha.119.14168
- Falkner B, Lurbe E. The USPSTF call to inaction on blood pressure screening in children and adolescents. Pediatr Nephrol. 2021;36:1327-1329. doi: 10.1007/s00467-021-04926-y
- Sinaiko AR, Jacobs DR Jr, Woo JG, et al. The International Childhood Cardiovascular Cohort (i3C) consortium outcomes study of childhood cardiovascular risk factors and adult cardiovascular morbidity and mortality: Design and recruitment. Contemp Clin Trials. 2018;69:55-64. doi: 10.1016/j.cct.2018.04.009
Bariatric surgery tied to 22% lower 5-year stroke risk
Patients with obesity who underwent bariatric surgery had 46% lower odds of stroke 1 year later, similar odds of stroke 3 years later, and 22% lower odds of stroke 5 years later, compared with matched control patients, in new research.
Michael D. Williams, MD, presented the study findings (abstract A002) at the annual meeting of the American Society for Metabolic & Bariatric Surgery.
The findings are “very good news,” even though the protection against stroke declined further out from the surgery, John D. Scott, MD, scientific program chair of the ASMBS meeting, told this news organization.
The investigators matched more than 56,000 patients with obesity who had bariatric surgery with an equal number of similar patients who did not have this surgery, from a large national insurance database, in what they believe is the largest study of this to date.
“Any intervention that decreases your risk of [cardiovascular] events is good news,” said Dr. Scott, a clinical professor of surgery at the University of South Carolina, Greenville, and metabolic and bariatric surgery director at Prisma Health in Greenville, S.C. “And having a 22%-45% chance of reduction in stroke risk is a very worthwhile intervention.”
Asked how this would change the way clinicians inform patients of what to expect from bariatric surgery, he said: “I would advise patients that studies like this show that surgery would not increase your risk of having a stroke.
“This is consistent with many studies that show that the risks of all macrovascular events decrease after the comorbidity reductions seen after surgery.”
According to Dr. Scott, “the next steps might include a prospective randomized trial of medical treatment versus surgery alone for [cardiovascular]/stroke outcomes, but this is unlikely.”
Similarly, Dr. Williams told this news organization that “I would tell [patients] that surgery is an effective and durable method for weight loss. It also can improve comorbid conditions, particularly diabetes and hypertension.”
Even with this study, “I’m not sure it’s appropriate to say that bariatric surgery will reduce the risk of stroke,” he cautioned.
“However, as we continue to investigate the effects of bariatric surgery, this study contributes to the greater body of knowledge that suggests that reduction in ischemic stroke risk is yet another benefit of bariatric surgery.”
The assigned discussant, Corrigan L. McBride, MD, MBA wanted to know if the lower odds ratio at 1 year might be because preoperative patient selection might eliminate patients at high risk of poor cardiovascular outcomes.
Dr. Williams, a resident at Rush Medical College, Chicago, replied that it is difficult to eliminate potential selection bias, despite best efforts, but this study shows that he can tell patients: “Having surgery is not going to increases your risk of stroke.”
“This is an important study,” Dr. McBride, professor and chief of minimally invasive surgery and bariatric surgery, University of Nebraska Medical Center, Omaha, told this news organization.
“It is the first large study to show a decreased [or no increased] risk of stroke 1, 3, and 5 years after bariatric surgery compared to matched patients, and it had enough data to look at stroke as a standalone endpoint,” Dr. McBride said. “It is important too, for patients and their physicians to understand that there is a lower chance of them having a stroke if they have surgery than if they do not.”
‘Important,’ ‘good news’ for stroke risk after bariatric surgery
The impact of bariatric surgery on remission of type 2 diabetes is well known, Dr. Williams noted, and other studies have reported how bariatric surgery affects the risk of major adverse cardiovascular events – a composite of stroke, myocardial infarction, coronary artery disease, and all-cause death – including a study presented in the same meeting session.
However, a very large sample size is needed to be able to demonstrate the effect of bariatric surgery on stroke, since stroke is a rare event.
The researchers analyzed data from the Mariner (PearlDiver.) all-payer insurance national claims database of patients in the United States.
They matched 56,514 patients with a body mass index over 35 kg/m2 and comorbidities or a BMI of more than 40 who underwent sleeve gastrectomy or Roux-en-Y gastric bypass during 2010-2019 with 56,514 control patients who did not undergo bariatric surgery.
A year after bariatric surgery, patients in that group had a lower stroke rate than patients in the control group (0.6% vs. 1.2%), and they had close to 50% lower odds of having a stroke (odds ratio, 0.54; 95% CI, 0.47-0.61).
Three years after bariatric surgery, there were 44,948 patients in each group; the rate of stroke was 2.1% in the surgery group and 2.2% in the control group, and there was no significant difference in the odds of having a stroke (OR, 0.96; 95% CI, 0.91-1.00).
Five years after bariatric surgery, there were 27,619 patients in each group; the stroke rate was lower in the bariatric surgery group than in the control group (2.8% vs 3.6%), but reduced odds of stroke was not as great as after 1 year (OR, 0.78; 95% CI, 0.65-0.90).
Dr. Williams has no relevant financial disclosures. Dr. McBride and Dr. Scott disclosed that they are speakers/trainers/faculty advisers for Gore. Dr. Scott is also a consultant for C-SATS (part of Johnson & Johnson).
Patients with obesity who underwent bariatric surgery had 46% lower odds of stroke 1 year later, similar odds of stroke 3 years later, and 22% lower odds of stroke 5 years later, compared with matched control patients, in new research.
Michael D. Williams, MD, presented the study findings (abstract A002) at the annual meeting of the American Society for Metabolic & Bariatric Surgery.
The findings are “very good news,” even though the protection against stroke declined further out from the surgery, John D. Scott, MD, scientific program chair of the ASMBS meeting, told this news organization.
The investigators matched more than 56,000 patients with obesity who had bariatric surgery with an equal number of similar patients who did not have this surgery, from a large national insurance database, in what they believe is the largest study of this to date.
“Any intervention that decreases your risk of [cardiovascular] events is good news,” said Dr. Scott, a clinical professor of surgery at the University of South Carolina, Greenville, and metabolic and bariatric surgery director at Prisma Health in Greenville, S.C. “And having a 22%-45% chance of reduction in stroke risk is a very worthwhile intervention.”
Asked how this would change the way clinicians inform patients of what to expect from bariatric surgery, he said: “I would advise patients that studies like this show that surgery would not increase your risk of having a stroke.
“This is consistent with many studies that show that the risks of all macrovascular events decrease after the comorbidity reductions seen after surgery.”
According to Dr. Scott, “the next steps might include a prospective randomized trial of medical treatment versus surgery alone for [cardiovascular]/stroke outcomes, but this is unlikely.”
Similarly, Dr. Williams told this news organization that “I would tell [patients] that surgery is an effective and durable method for weight loss. It also can improve comorbid conditions, particularly diabetes and hypertension.”
Even with this study, “I’m not sure it’s appropriate to say that bariatric surgery will reduce the risk of stroke,” he cautioned.
“However, as we continue to investigate the effects of bariatric surgery, this study contributes to the greater body of knowledge that suggests that reduction in ischemic stroke risk is yet another benefit of bariatric surgery.”
The assigned discussant, Corrigan L. McBride, MD, MBA wanted to know if the lower odds ratio at 1 year might be because preoperative patient selection might eliminate patients at high risk of poor cardiovascular outcomes.
Dr. Williams, a resident at Rush Medical College, Chicago, replied that it is difficult to eliminate potential selection bias, despite best efforts, but this study shows that he can tell patients: “Having surgery is not going to increases your risk of stroke.”
“This is an important study,” Dr. McBride, professor and chief of minimally invasive surgery and bariatric surgery, University of Nebraska Medical Center, Omaha, told this news organization.
“It is the first large study to show a decreased [or no increased] risk of stroke 1, 3, and 5 years after bariatric surgery compared to matched patients, and it had enough data to look at stroke as a standalone endpoint,” Dr. McBride said. “It is important too, for patients and their physicians to understand that there is a lower chance of them having a stroke if they have surgery than if they do not.”
‘Important,’ ‘good news’ for stroke risk after bariatric surgery
The impact of bariatric surgery on remission of type 2 diabetes is well known, Dr. Williams noted, and other studies have reported how bariatric surgery affects the risk of major adverse cardiovascular events – a composite of stroke, myocardial infarction, coronary artery disease, and all-cause death – including a study presented in the same meeting session.
However, a very large sample size is needed to be able to demonstrate the effect of bariatric surgery on stroke, since stroke is a rare event.
The researchers analyzed data from the Mariner (PearlDiver.) all-payer insurance national claims database of patients in the United States.
They matched 56,514 patients with a body mass index over 35 kg/m2 and comorbidities or a BMI of more than 40 who underwent sleeve gastrectomy or Roux-en-Y gastric bypass during 2010-2019 with 56,514 control patients who did not undergo bariatric surgery.
A year after bariatric surgery, patients in that group had a lower stroke rate than patients in the control group (0.6% vs. 1.2%), and they had close to 50% lower odds of having a stroke (odds ratio, 0.54; 95% CI, 0.47-0.61).
Three years after bariatric surgery, there were 44,948 patients in each group; the rate of stroke was 2.1% in the surgery group and 2.2% in the control group, and there was no significant difference in the odds of having a stroke (OR, 0.96; 95% CI, 0.91-1.00).
Five years after bariatric surgery, there were 27,619 patients in each group; the stroke rate was lower in the bariatric surgery group than in the control group (2.8% vs 3.6%), but reduced odds of stroke was not as great as after 1 year (OR, 0.78; 95% CI, 0.65-0.90).
Dr. Williams has no relevant financial disclosures. Dr. McBride and Dr. Scott disclosed that they are speakers/trainers/faculty advisers for Gore. Dr. Scott is also a consultant for C-SATS (part of Johnson & Johnson).
Patients with obesity who underwent bariatric surgery had 46% lower odds of stroke 1 year later, similar odds of stroke 3 years later, and 22% lower odds of stroke 5 years later, compared with matched control patients, in new research.
Michael D. Williams, MD, presented the study findings (abstract A002) at the annual meeting of the American Society for Metabolic & Bariatric Surgery.
The findings are “very good news,” even though the protection against stroke declined further out from the surgery, John D. Scott, MD, scientific program chair of the ASMBS meeting, told this news organization.
The investigators matched more than 56,000 patients with obesity who had bariatric surgery with an equal number of similar patients who did not have this surgery, from a large national insurance database, in what they believe is the largest study of this to date.
“Any intervention that decreases your risk of [cardiovascular] events is good news,” said Dr. Scott, a clinical professor of surgery at the University of South Carolina, Greenville, and metabolic and bariatric surgery director at Prisma Health in Greenville, S.C. “And having a 22%-45% chance of reduction in stroke risk is a very worthwhile intervention.”
Asked how this would change the way clinicians inform patients of what to expect from bariatric surgery, he said: “I would advise patients that studies like this show that surgery would not increase your risk of having a stroke.
“This is consistent with many studies that show that the risks of all macrovascular events decrease after the comorbidity reductions seen after surgery.”
According to Dr. Scott, “the next steps might include a prospective randomized trial of medical treatment versus surgery alone for [cardiovascular]/stroke outcomes, but this is unlikely.”
Similarly, Dr. Williams told this news organization that “I would tell [patients] that surgery is an effective and durable method for weight loss. It also can improve comorbid conditions, particularly diabetes and hypertension.”
Even with this study, “I’m not sure it’s appropriate to say that bariatric surgery will reduce the risk of stroke,” he cautioned.
“However, as we continue to investigate the effects of bariatric surgery, this study contributes to the greater body of knowledge that suggests that reduction in ischemic stroke risk is yet another benefit of bariatric surgery.”
The assigned discussant, Corrigan L. McBride, MD, MBA wanted to know if the lower odds ratio at 1 year might be because preoperative patient selection might eliminate patients at high risk of poor cardiovascular outcomes.
Dr. Williams, a resident at Rush Medical College, Chicago, replied that it is difficult to eliminate potential selection bias, despite best efforts, but this study shows that he can tell patients: “Having surgery is not going to increases your risk of stroke.”
“This is an important study,” Dr. McBride, professor and chief of minimally invasive surgery and bariatric surgery, University of Nebraska Medical Center, Omaha, told this news organization.
“It is the first large study to show a decreased [or no increased] risk of stroke 1, 3, and 5 years after bariatric surgery compared to matched patients, and it had enough data to look at stroke as a standalone endpoint,” Dr. McBride said. “It is important too, for patients and their physicians to understand that there is a lower chance of them having a stroke if they have surgery than if they do not.”
‘Important,’ ‘good news’ for stroke risk after bariatric surgery
The impact of bariatric surgery on remission of type 2 diabetes is well known, Dr. Williams noted, and other studies have reported how bariatric surgery affects the risk of major adverse cardiovascular events – a composite of stroke, myocardial infarction, coronary artery disease, and all-cause death – including a study presented in the same meeting session.
However, a very large sample size is needed to be able to demonstrate the effect of bariatric surgery on stroke, since stroke is a rare event.
The researchers analyzed data from the Mariner (PearlDiver.) all-payer insurance national claims database of patients in the United States.
They matched 56,514 patients with a body mass index over 35 kg/m2 and comorbidities or a BMI of more than 40 who underwent sleeve gastrectomy or Roux-en-Y gastric bypass during 2010-2019 with 56,514 control patients who did not undergo bariatric surgery.
A year after bariatric surgery, patients in that group had a lower stroke rate than patients in the control group (0.6% vs. 1.2%), and they had close to 50% lower odds of having a stroke (odds ratio, 0.54; 95% CI, 0.47-0.61).
Three years after bariatric surgery, there were 44,948 patients in each group; the rate of stroke was 2.1% in the surgery group and 2.2% in the control group, and there was no significant difference in the odds of having a stroke (OR, 0.96; 95% CI, 0.91-1.00).
Five years after bariatric surgery, there were 27,619 patients in each group; the stroke rate was lower in the bariatric surgery group than in the control group (2.8% vs 3.6%), but reduced odds of stroke was not as great as after 1 year (OR, 0.78; 95% CI, 0.65-0.90).
Dr. Williams has no relevant financial disclosures. Dr. McBride and Dr. Scott disclosed that they are speakers/trainers/faculty advisers for Gore. Dr. Scott is also a consultant for C-SATS (part of Johnson & Johnson).
FROM ASMBS 2021
Medically suspect criterion can determine bariatric surgery coverage
A delaying tactic used by some U.S. health insurers to limit coverage of bariatric surgery does not jibe with the clinical experience at one U.S. center with 461 patients who underwent primary or revisional bariatric surgery.
The tactic applies to patients with a baseline body mass index (BMI) of 35-39 kg/m2 who usually also need at least one comorbidity to qualify for insurance coverage for bariatric surgery, and specifically to the subgroup for whom hypertension is the qualifying comorbidity.
Some insurers limit surgery coverage to patients with hypertension who fail to reach their goal blood pressure on agents from three different drug classes, a policy that is “extremely frustrating and dangerous,” said Yannis Raftopoulos, MD, PhD, in his presentation at the annual meeting of the American Society for Metabolic and Bariatric Surgery.
Using number of antihypertensive drugs ‘is not correct’
“Using the number of antihypertensive medications to justify surgery is not correct because blood pressure control is not [always] better when patients take two or three medications, compared with when they are taking one. This harms patients because the more severe their hypertension, the worse their control,” said Dr. Raftopoulos, director of the weight management program at Holyoke (Mass.) Medical Center.
He presented findings from a retrospective study of 461 patients who underwent either sleeve gastrectomy or laparoscopic Roux-en-Y gastric bypass at his center, including 213 (46%) diagnosed with hypertension at the time of their surgery. Within this group were 68 patients with a BMI of 35-39, which meant that they could get insurance coverage for bariatric surgery only if they also had a relevant comorbidity such as hypertension, diabetes, or severe sleep apnea.
Among these patients, 36 (17% of those with hypertension) had only hypertension as their relevant comorbidity and would not have qualified for bariatric surgery under the strictest criteria applied by some insurers that require patients to remain hypertensive despite treatment with at least three different antihypertensive medications. (These 36 patients underwent bariatric surgery because their insurance coverage did not have this restriction.)
The analyses Dr. Raftopoulos presented also documented the rate of hypertension resolution among patients in the series who had hypertension at baseline and 1-year follow-up results. Among 65 patients on one antihypertensive drug at baseline, 43 (66%) had complete resolution of their hypertension after 1 year, defined as blood pressure of less than 130/90 mm Hg while completely off antihypertensive treatment. In contrast, among 55 patients on two antihypertensive medications at baseline, 28 (51%) had complete resolution after 1 year, and among 24 patients on three or more antihypertensive medications at baseline, 3 (13%) had complete resolution 1 year after bariatric surgery, he reported.
“Patients who were treated with one oral antihypertensive medication preoperatively had a higher likelihood of postoperative hypertension resolution,” concluded Dr. Raftopoulos.
Restricting access to bariatric surgery to patients with a BMI of less than 40 based on the preoperative intensity of their antihypertensive treatment “is not supported by our data, and can be potentially harmful,” he declared.
“This study was the result of discussions about this problem with multiple insurers in my area,” he added. “This affects a good number of patients.”
Waiting for hypertension to become less treatable
The results Dr. Raftopoulos presented “are not surprising, because they confirm the hypothesis that earlier intervention in the course of a disease like hypertension is more likely to be successful,” commented Bruce D. Schirmer, MD, a professor of surgery at the University of Virginia, Charlottesville, and designated discussant for the report.
The policy followed by some health insurers to delay coverage for bariatric surgery until patients fail three medications “forces patients with more treatable hypertension to wait until their disease worsens and becomes less treatable before they can receive appropriate treatment,” he said.
Dr. Schirmer attributed the motivation for this approach to a “despicable” and “reprehensible” reason: “Actuarial calculations that show paying for curative therapy is not cost effective in the short term. The duration of a patient’s policy may not be long enough to yield a positive financial outcome, so it becomes more appropriate to deny optimal care and have patients become sicker from their disease.”
“I applaud the authors for accumulating the data that point out this unfortunate rule of some insurance companies,” Dr. Schirmer added.
The practice is comparable with an insurer requiring that a patient’s cancer must be metastatic before allowing coverage for treatment, commented Ann M. Rogers, MD, professor and director of the Penn State University surgical weight loss program in Hershey, Penn., and a moderator of the session.
Dr. Raftopoulos, Dr. Schirmer, and Dr. Rogers had no disclosures.
A delaying tactic used by some U.S. health insurers to limit coverage of bariatric surgery does not jibe with the clinical experience at one U.S. center with 461 patients who underwent primary or revisional bariatric surgery.
The tactic applies to patients with a baseline body mass index (BMI) of 35-39 kg/m2 who usually also need at least one comorbidity to qualify for insurance coverage for bariatric surgery, and specifically to the subgroup for whom hypertension is the qualifying comorbidity.
Some insurers limit surgery coverage to patients with hypertension who fail to reach their goal blood pressure on agents from three different drug classes, a policy that is “extremely frustrating and dangerous,” said Yannis Raftopoulos, MD, PhD, in his presentation at the annual meeting of the American Society for Metabolic and Bariatric Surgery.
Using number of antihypertensive drugs ‘is not correct’
“Using the number of antihypertensive medications to justify surgery is not correct because blood pressure control is not [always] better when patients take two or three medications, compared with when they are taking one. This harms patients because the more severe their hypertension, the worse their control,” said Dr. Raftopoulos, director of the weight management program at Holyoke (Mass.) Medical Center.
He presented findings from a retrospective study of 461 patients who underwent either sleeve gastrectomy or laparoscopic Roux-en-Y gastric bypass at his center, including 213 (46%) diagnosed with hypertension at the time of their surgery. Within this group were 68 patients with a BMI of 35-39, which meant that they could get insurance coverage for bariatric surgery only if they also had a relevant comorbidity such as hypertension, diabetes, or severe sleep apnea.
Among these patients, 36 (17% of those with hypertension) had only hypertension as their relevant comorbidity and would not have qualified for bariatric surgery under the strictest criteria applied by some insurers that require patients to remain hypertensive despite treatment with at least three different antihypertensive medications. (These 36 patients underwent bariatric surgery because their insurance coverage did not have this restriction.)
The analyses Dr. Raftopoulos presented also documented the rate of hypertension resolution among patients in the series who had hypertension at baseline and 1-year follow-up results. Among 65 patients on one antihypertensive drug at baseline, 43 (66%) had complete resolution of their hypertension after 1 year, defined as blood pressure of less than 130/90 mm Hg while completely off antihypertensive treatment. In contrast, among 55 patients on two antihypertensive medications at baseline, 28 (51%) had complete resolution after 1 year, and among 24 patients on three or more antihypertensive medications at baseline, 3 (13%) had complete resolution 1 year after bariatric surgery, he reported.
“Patients who were treated with one oral antihypertensive medication preoperatively had a higher likelihood of postoperative hypertension resolution,” concluded Dr. Raftopoulos.
Restricting access to bariatric surgery to patients with a BMI of less than 40 based on the preoperative intensity of their antihypertensive treatment “is not supported by our data, and can be potentially harmful,” he declared.
“This study was the result of discussions about this problem with multiple insurers in my area,” he added. “This affects a good number of patients.”
Waiting for hypertension to become less treatable
The results Dr. Raftopoulos presented “are not surprising, because they confirm the hypothesis that earlier intervention in the course of a disease like hypertension is more likely to be successful,” commented Bruce D. Schirmer, MD, a professor of surgery at the University of Virginia, Charlottesville, and designated discussant for the report.
The policy followed by some health insurers to delay coverage for bariatric surgery until patients fail three medications “forces patients with more treatable hypertension to wait until their disease worsens and becomes less treatable before they can receive appropriate treatment,” he said.
Dr. Schirmer attributed the motivation for this approach to a “despicable” and “reprehensible” reason: “Actuarial calculations that show paying for curative therapy is not cost effective in the short term. The duration of a patient’s policy may not be long enough to yield a positive financial outcome, so it becomes more appropriate to deny optimal care and have patients become sicker from their disease.”
“I applaud the authors for accumulating the data that point out this unfortunate rule of some insurance companies,” Dr. Schirmer added.
The practice is comparable with an insurer requiring that a patient’s cancer must be metastatic before allowing coverage for treatment, commented Ann M. Rogers, MD, professor and director of the Penn State University surgical weight loss program in Hershey, Penn., and a moderator of the session.
Dr. Raftopoulos, Dr. Schirmer, and Dr. Rogers had no disclosures.
A delaying tactic used by some U.S. health insurers to limit coverage of bariatric surgery does not jibe with the clinical experience at one U.S. center with 461 patients who underwent primary or revisional bariatric surgery.
The tactic applies to patients with a baseline body mass index (BMI) of 35-39 kg/m2 who usually also need at least one comorbidity to qualify for insurance coverage for bariatric surgery, and specifically to the subgroup for whom hypertension is the qualifying comorbidity.
Some insurers limit surgery coverage to patients with hypertension who fail to reach their goal blood pressure on agents from three different drug classes, a policy that is “extremely frustrating and dangerous,” said Yannis Raftopoulos, MD, PhD, in his presentation at the annual meeting of the American Society for Metabolic and Bariatric Surgery.
Using number of antihypertensive drugs ‘is not correct’
“Using the number of antihypertensive medications to justify surgery is not correct because blood pressure control is not [always] better when patients take two or three medications, compared with when they are taking one. This harms patients because the more severe their hypertension, the worse their control,” said Dr. Raftopoulos, director of the weight management program at Holyoke (Mass.) Medical Center.
He presented findings from a retrospective study of 461 patients who underwent either sleeve gastrectomy or laparoscopic Roux-en-Y gastric bypass at his center, including 213 (46%) diagnosed with hypertension at the time of their surgery. Within this group were 68 patients with a BMI of 35-39, which meant that they could get insurance coverage for bariatric surgery only if they also had a relevant comorbidity such as hypertension, diabetes, or severe sleep apnea.
Among these patients, 36 (17% of those with hypertension) had only hypertension as their relevant comorbidity and would not have qualified for bariatric surgery under the strictest criteria applied by some insurers that require patients to remain hypertensive despite treatment with at least three different antihypertensive medications. (These 36 patients underwent bariatric surgery because their insurance coverage did not have this restriction.)
The analyses Dr. Raftopoulos presented also documented the rate of hypertension resolution among patients in the series who had hypertension at baseline and 1-year follow-up results. Among 65 patients on one antihypertensive drug at baseline, 43 (66%) had complete resolution of their hypertension after 1 year, defined as blood pressure of less than 130/90 mm Hg while completely off antihypertensive treatment. In contrast, among 55 patients on two antihypertensive medications at baseline, 28 (51%) had complete resolution after 1 year, and among 24 patients on three or more antihypertensive medications at baseline, 3 (13%) had complete resolution 1 year after bariatric surgery, he reported.
“Patients who were treated with one oral antihypertensive medication preoperatively had a higher likelihood of postoperative hypertension resolution,” concluded Dr. Raftopoulos.
Restricting access to bariatric surgery to patients with a BMI of less than 40 based on the preoperative intensity of their antihypertensive treatment “is not supported by our data, and can be potentially harmful,” he declared.
“This study was the result of discussions about this problem with multiple insurers in my area,” he added. “This affects a good number of patients.”
Waiting for hypertension to become less treatable
The results Dr. Raftopoulos presented “are not surprising, because they confirm the hypothesis that earlier intervention in the course of a disease like hypertension is more likely to be successful,” commented Bruce D. Schirmer, MD, a professor of surgery at the University of Virginia, Charlottesville, and designated discussant for the report.
The policy followed by some health insurers to delay coverage for bariatric surgery until patients fail three medications “forces patients with more treatable hypertension to wait until their disease worsens and becomes less treatable before they can receive appropriate treatment,” he said.
Dr. Schirmer attributed the motivation for this approach to a “despicable” and “reprehensible” reason: “Actuarial calculations that show paying for curative therapy is not cost effective in the short term. The duration of a patient’s policy may not be long enough to yield a positive financial outcome, so it becomes more appropriate to deny optimal care and have patients become sicker from their disease.”
“I applaud the authors for accumulating the data that point out this unfortunate rule of some insurance companies,” Dr. Schirmer added.
The practice is comparable with an insurer requiring that a patient’s cancer must be metastatic before allowing coverage for treatment, commented Ann M. Rogers, MD, professor and director of the Penn State University surgical weight loss program in Hershey, Penn., and a moderator of the session.
Dr. Raftopoulos, Dr. Schirmer, and Dr. Rogers had no disclosures.
FROM ASMBS 2021
Bariatric surgery cuts insulin needs in type 1 diabetes with severe obesity
While bariatric surgery does nothing to directly improve the disease of patients with type 1 diabetes, it can work indirectly by moderating severe obesity and improving insulin sensitivity to cut the total insulin needs of patients with type 1 diabetes and obesity, based on a single-center, retrospective chart review of 38 U.S. patients.
Two years following their bariatric surgery, these 38 patients with confirmed type 1 diabetes and an average body mass index of 43 kg/m2 before surgery saw their average daily insulin requirement nearly halved, dropping from 118 units/day to 60 units/day, a significant decrease, Brian J. Dessify, DO, said in a presentation at the annual meeting of the American Society for Metabolic and Bariatric Surgery.
Another measure of this effect showed that the percentage of patients who required more than one drug for treating their hyperglycemia fell from 66% before surgery to 52% 2 years after surgery, a change that was not statistically significant, said Dr. Dessify, a bariatric surgeon at Geisinger Medical Center in Danville, Pa.
Appropriate for patients with ‘double diabetes’
These results “provide good evidence for [using] bariatric surgery” in people with both obesity and type 1 diabetes,” he concluded. This includes people with what Dr. Dessify called “double diabetes,” meaning that they do not make endogenous insulin, and are also resistant to the effects of exogenous insulin and hence have features of both type 2 and type 1 diabetes.
“This is a really important study,” commented Ali Aminian, MD, director of the Bariatric and Metabolic Institute of the Cleveland Clinic. “For patients with type 1 diabetes, the primary goal of bariatric surgery is weight loss and improvement of obesity-related comorbidities. Patients with type 2 diabetes can be a candidate for bariatric surgery regardless of their weight,” Dr. Aminian said as designated discussant for the report.
“The goal of bariatric surgery in patients with type 1 diabetes is to promote sensitivity to the exogenous insulin they receive,” agreed Julie Kim, MD, a bariatric surgeon at Mount Auburn Hospital in Waltham, Mass., and a second discussant for the report. Patients with double diabetes “are probably a subclass of patients [with type 1 diabetes] who might benefit even more from bariatric surgery.”
Using gastric sleeves to avoid diabetic ketoacidosis
Dr. Aminian also noted that “at the Cleveland Clinic we consider a sleeve gastrectomy the procedure of choice” for patients with type 1 diabetes or type 2 diabetes with insulin insufficiency “unless the patient has an absolute contraindication” because of the increased risk for diabetic ketoacidosis in these patients “undergoing any surgery, including bariatric surgery.” Patients with insulin insufficiency “require intensive diabetes and insulin management preoperatively to reduce their risk for developing diabetic ketoacidosis,” and using a sleeve rather than bypass generally results in “more reliable absorption of carbohydrates and nutrients” while also reducing the risk for hypoglycemia, Dr. Aminian said.
In the series reported by Dr. Dessify, 33 patients underwent gastric bypass and 5 had sleeve gastrectomy. The decision to use bypass usually stemmed from its “marginal” improvement in weight loss, compared with a sleeve procedure, and an overall preference at Geisinger for bypass procedures. Dr. Dessify added that he had not yet run a comprehensive assessment of diabetic ketoacidosis complications among patients in his reported series.
Those 38 patients underwent their bariatric procedure during 2002-2019, constituting fewer than 1% of the 4,549 total bariatric surgeries done at Geisinger during that period. The 38 patients with type 1 diabetes averaged 41 years of age, 33 (87%) were women, and 37 (97%) were White. Dr. Dessify and associates undertook this review “to help provide supporting evidence for using bariatric surgery in people with obesity and type 1 diabetes,” he noted.
Dr. Dessify, Dr. Aminian, and Dr. Kim had no disclosures.
While bariatric surgery does nothing to directly improve the disease of patients with type 1 diabetes, it can work indirectly by moderating severe obesity and improving insulin sensitivity to cut the total insulin needs of patients with type 1 diabetes and obesity, based on a single-center, retrospective chart review of 38 U.S. patients.
Two years following their bariatric surgery, these 38 patients with confirmed type 1 diabetes and an average body mass index of 43 kg/m2 before surgery saw their average daily insulin requirement nearly halved, dropping from 118 units/day to 60 units/day, a significant decrease, Brian J. Dessify, DO, said in a presentation at the annual meeting of the American Society for Metabolic and Bariatric Surgery.
Another measure of this effect showed that the percentage of patients who required more than one drug for treating their hyperglycemia fell from 66% before surgery to 52% 2 years after surgery, a change that was not statistically significant, said Dr. Dessify, a bariatric surgeon at Geisinger Medical Center in Danville, Pa.
Appropriate for patients with ‘double diabetes’
These results “provide good evidence for [using] bariatric surgery” in people with both obesity and type 1 diabetes,” he concluded. This includes people with what Dr. Dessify called “double diabetes,” meaning that they do not make endogenous insulin, and are also resistant to the effects of exogenous insulin and hence have features of both type 2 and type 1 diabetes.
“This is a really important study,” commented Ali Aminian, MD, director of the Bariatric and Metabolic Institute of the Cleveland Clinic. “For patients with type 1 diabetes, the primary goal of bariatric surgery is weight loss and improvement of obesity-related comorbidities. Patients with type 2 diabetes can be a candidate for bariatric surgery regardless of their weight,” Dr. Aminian said as designated discussant for the report.
“The goal of bariatric surgery in patients with type 1 diabetes is to promote sensitivity to the exogenous insulin they receive,” agreed Julie Kim, MD, a bariatric surgeon at Mount Auburn Hospital in Waltham, Mass., and a second discussant for the report. Patients with double diabetes “are probably a subclass of patients [with type 1 diabetes] who might benefit even more from bariatric surgery.”
Using gastric sleeves to avoid diabetic ketoacidosis
Dr. Aminian also noted that “at the Cleveland Clinic we consider a sleeve gastrectomy the procedure of choice” for patients with type 1 diabetes or type 2 diabetes with insulin insufficiency “unless the patient has an absolute contraindication” because of the increased risk for diabetic ketoacidosis in these patients “undergoing any surgery, including bariatric surgery.” Patients with insulin insufficiency “require intensive diabetes and insulin management preoperatively to reduce their risk for developing diabetic ketoacidosis,” and using a sleeve rather than bypass generally results in “more reliable absorption of carbohydrates and nutrients” while also reducing the risk for hypoglycemia, Dr. Aminian said.
In the series reported by Dr. Dessify, 33 patients underwent gastric bypass and 5 had sleeve gastrectomy. The decision to use bypass usually stemmed from its “marginal” improvement in weight loss, compared with a sleeve procedure, and an overall preference at Geisinger for bypass procedures. Dr. Dessify added that he had not yet run a comprehensive assessment of diabetic ketoacidosis complications among patients in his reported series.
Those 38 patients underwent their bariatric procedure during 2002-2019, constituting fewer than 1% of the 4,549 total bariatric surgeries done at Geisinger during that period. The 38 patients with type 1 diabetes averaged 41 years of age, 33 (87%) were women, and 37 (97%) were White. Dr. Dessify and associates undertook this review “to help provide supporting evidence for using bariatric surgery in people with obesity and type 1 diabetes,” he noted.
Dr. Dessify, Dr. Aminian, and Dr. Kim had no disclosures.
While bariatric surgery does nothing to directly improve the disease of patients with type 1 diabetes, it can work indirectly by moderating severe obesity and improving insulin sensitivity to cut the total insulin needs of patients with type 1 diabetes and obesity, based on a single-center, retrospective chart review of 38 U.S. patients.
Two years following their bariatric surgery, these 38 patients with confirmed type 1 diabetes and an average body mass index of 43 kg/m2 before surgery saw their average daily insulin requirement nearly halved, dropping from 118 units/day to 60 units/day, a significant decrease, Brian J. Dessify, DO, said in a presentation at the annual meeting of the American Society for Metabolic and Bariatric Surgery.
Another measure of this effect showed that the percentage of patients who required more than one drug for treating their hyperglycemia fell from 66% before surgery to 52% 2 years after surgery, a change that was not statistically significant, said Dr. Dessify, a bariatric surgeon at Geisinger Medical Center in Danville, Pa.
Appropriate for patients with ‘double diabetes’
These results “provide good evidence for [using] bariatric surgery” in people with both obesity and type 1 diabetes,” he concluded. This includes people with what Dr. Dessify called “double diabetes,” meaning that they do not make endogenous insulin, and are also resistant to the effects of exogenous insulin and hence have features of both type 2 and type 1 diabetes.
“This is a really important study,” commented Ali Aminian, MD, director of the Bariatric and Metabolic Institute of the Cleveland Clinic. “For patients with type 1 diabetes, the primary goal of bariatric surgery is weight loss and improvement of obesity-related comorbidities. Patients with type 2 diabetes can be a candidate for bariatric surgery regardless of their weight,” Dr. Aminian said as designated discussant for the report.
“The goal of bariatric surgery in patients with type 1 diabetes is to promote sensitivity to the exogenous insulin they receive,” agreed Julie Kim, MD, a bariatric surgeon at Mount Auburn Hospital in Waltham, Mass., and a second discussant for the report. Patients with double diabetes “are probably a subclass of patients [with type 1 diabetes] who might benefit even more from bariatric surgery.”
Using gastric sleeves to avoid diabetic ketoacidosis
Dr. Aminian also noted that “at the Cleveland Clinic we consider a sleeve gastrectomy the procedure of choice” for patients with type 1 diabetes or type 2 diabetes with insulin insufficiency “unless the patient has an absolute contraindication” because of the increased risk for diabetic ketoacidosis in these patients “undergoing any surgery, including bariatric surgery.” Patients with insulin insufficiency “require intensive diabetes and insulin management preoperatively to reduce their risk for developing diabetic ketoacidosis,” and using a sleeve rather than bypass generally results in “more reliable absorption of carbohydrates and nutrients” while also reducing the risk for hypoglycemia, Dr. Aminian said.
In the series reported by Dr. Dessify, 33 patients underwent gastric bypass and 5 had sleeve gastrectomy. The decision to use bypass usually stemmed from its “marginal” improvement in weight loss, compared with a sleeve procedure, and an overall preference at Geisinger for bypass procedures. Dr. Dessify added that he had not yet run a comprehensive assessment of diabetic ketoacidosis complications among patients in his reported series.
Those 38 patients underwent their bariatric procedure during 2002-2019, constituting fewer than 1% of the 4,549 total bariatric surgeries done at Geisinger during that period. The 38 patients with type 1 diabetes averaged 41 years of age, 33 (87%) were women, and 37 (97%) were White. Dr. Dessify and associates undertook this review “to help provide supporting evidence for using bariatric surgery in people with obesity and type 1 diabetes,” he noted.
Dr. Dessify, Dr. Aminian, and Dr. Kim had no disclosures.
FROM ASMBS 2021
Getting hypertension under control in the youngest of patients
Hypertension and elevated blood pressure (BP) in children and adolescents correlate to hypertension in adults, insofar as complications and medical therapy increase with age.1,2 Untreated, hypertension in children and adolescents can result in multiple harmful physiologic changes, including left ventricular hypertrophy, left atrial enlargement, diastolic dysfunction, arterial stiffening, endothelial dysfunction, and neurocognitive deficits.3-5
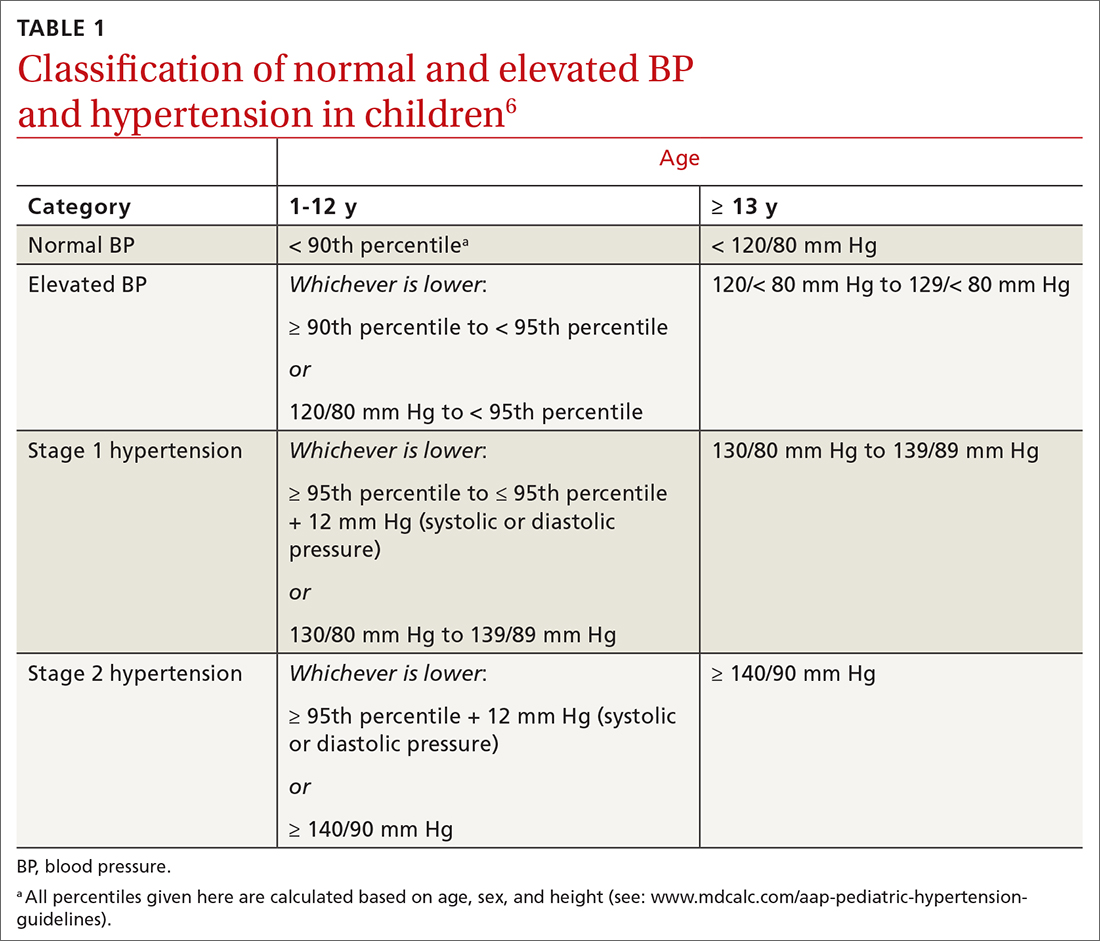
In 2017, the American Academy of Pediatrics (AAP) published clinical practice guidelines for the diagnosis and management of elevated BP and hypertension in children and adolescentsa (TABLE 16). Applying the definition of elevated BP set out in these guidelines yielded a 13% prevalence of hypertension in a cohort of subjects 10 to 18 years of age with comorbid obesity and diabetes mellitus (DM). AAP guideline definitions also improved the sensitivity for identifying hypertensive end-organ damage.7
As the prevalence of hypertension increases, screening for and accurate diagnosis of this condition in children are becoming more important. Recognition and management remain a vital part of primary care. In this article, we review the updated guidance on diagnosis and treatment, including lifestyle modification and pharmacotherapy.
First step: Identifying hypertension
Risk factors
Risk factors for pediatric hypertension are similar to those in adults. These include obesity (body mass index ≥ 95th percentile for age), types 1 and 2 DM, elevated sodium intake, sleep-disordered breathing, and chronic kidney disease (CKD). Some risk factors, such as premature birth and coarctation of the aorta, are specific to the pediatric population.8-14 Pediatric obesity strongly correlates with both pediatric and adult hypertension, and accelerated weight gain might increase the risk of elevated BP in adulthood.15,16

Intervening early to mitigate or eliminate some of these modifiable risk factors can prevent or treat hypertension.17 Alternatively, having been breastfed as an infant has been reliably shown to reduce the risk of elevated BP in children.13
Recommendations for screening and measuring BP
The optimal age to start measuring BP is not clearly defined. AAP recommends measurement:
- annually in all children ≥ 3 years of age
- at every encounter in patients who have a specific comorbid condition, including obesity, DM, renal disease, and aortic-arch abnormalities (obstruction and coarctation) and in those who are taking medication known to increase BP.6
Protocol. Measure BP in the right arm for consistency and comparison with reference values. The width of the cuff bladder should be at least 40%, and the length, 80% to 100%, of arm circumference. Position the cuff bladder midway between the olecranon and acromion. Obtain the measurement in a quiet and comfortable environment after the patient has rested for 3 to 5 minutes. The patient should be seated, preferably with feet on the floor; elbows should be supported at the level of the heart.
Continue to: When an initial reading...
When an initial reading is elevated, whether by oscillometric or auscultatory measurement, 2 more auscultatory BP measurements should be taken during the same visit; these measurements are averaged to determine the BP category.18
TABLE 16 defines BP categories based on age, sex, and height. We recommend using the free resource MD Calc (www.mdcalc.com/aap-pediatric-hypertension-guidelines) to assist in calculating the BP category.
TABLE 26 describes the timing of follow-up based on the initial BP reading and diagnosis.
Ambulatory BP monitoring (ABPM) is a validated device that measures BP every 20 to 30 minutes throughout the day and night. ABPM should be performed initially in all patients with persistently elevated BP and routinely in children and adolescents with a high-risk comorbidity (TABLE 26). Note: Insurance coverage of ABPM is limited.
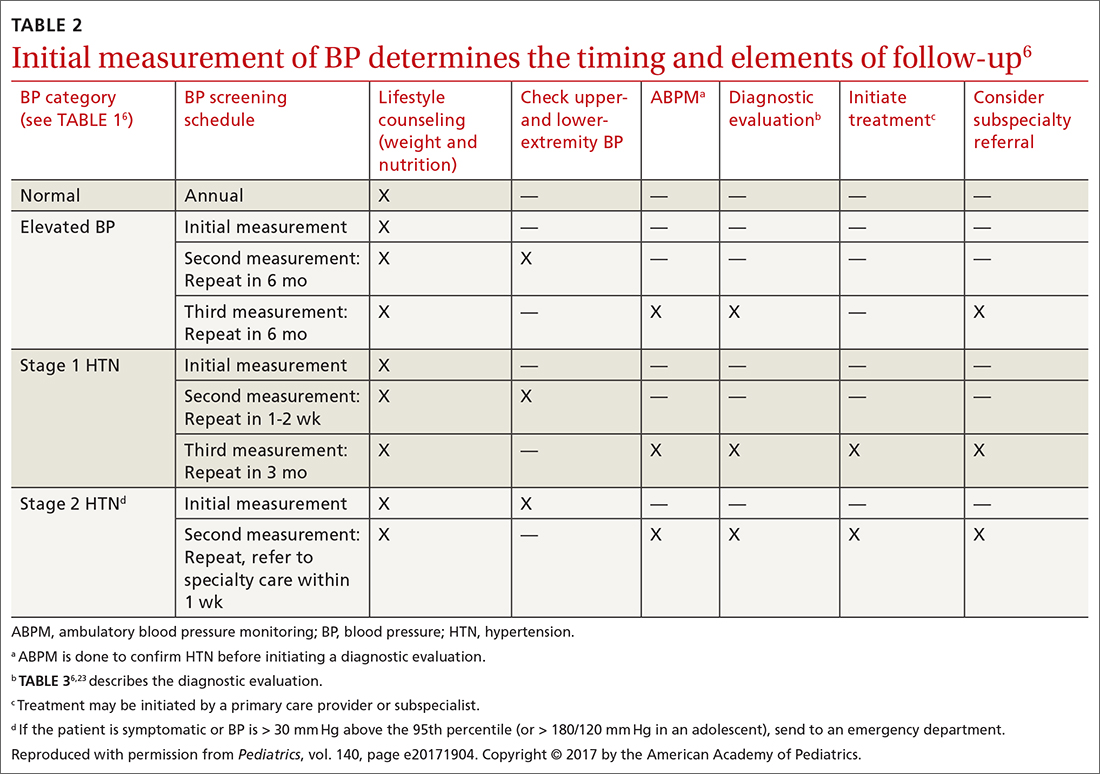
ABPM is also used to diagnose so-called white-coat hypertension, defined as BP ≥ 95th percentile for age, sex, and height in the clinic setting but < 95th percentile during ABPM. This phenomenon can be challenging to diagnose.
Continue to: Home monitoring
Home monitoring. Do not use home BP monitoring to establish a diagnosis of hypertension, although one of these devices can be used as an adjunct to office and ambulatory BP monitoring after the diagnosis has been made.6
Evaluating hypertension in children and adolescents
Once a diagnosis of hypertension has been made, undertake a thorough history, physical examination, and diagnostic testing to evaluate for possible causes, comorbidities, and any evidence of end-organ damage.
Comprehensive history. Pertinent aspects include perinatal, nutritional, physical activity, psychosocial, family, medication—and of course, medical—histories.6
Maternal elevated BP or hypertension is related to an offspring’s elevated BP in childhood and adolescence.19 Other pertinent aspects of the perinatal history include complications of pregnancy, gestational age, birth weight, and neonatal complications.6
Nutritional and physical activity histories can highlight contributing factors in the development of hypertension and can be a guide to recommending lifestyle modifications.6 Sodium intake, which influences BP, should be part of the nutritional history.20
Continue to: Important aspects...
Important aspects of the psychosocial history include feelings of depression or anxiety, bullying, and body perception. Children older than 10 years should be asked about smoking, alcohol, and other substance use.
The family history should include notation of first- and second-degree relatives with hypertension.6
Inquire about medications that can raise BP, including oral contraceptives, which are commonly prescribed in this population.21,22
The physical exam should include measured height and weight, with calculation of the body mass index percentile for age; of note, obesity is strongly associated with hypertension, and poor growth might signal underlying chronic disease. Once elevated BP has been confirmed, the exam should include measurement of BP in both arms and in a leg (TABLE 26). BP that is lower in the leg than in the arms (in any given patient, BP readings in the legs are usually higher than in the arms), or weak or absent femoral pulses, suggest coarctation of the aorta.6
Focus the balance of the physical exam on physical findings that suggest secondary causes of hypertension or evidence of end-organ damage.
Continue to: Testing
Testing. TABLE 36,23 summarizes the diagnostic testing recommended for all children and for specific populations; TABLE 26 indicates when to obtain diagnostic testing.
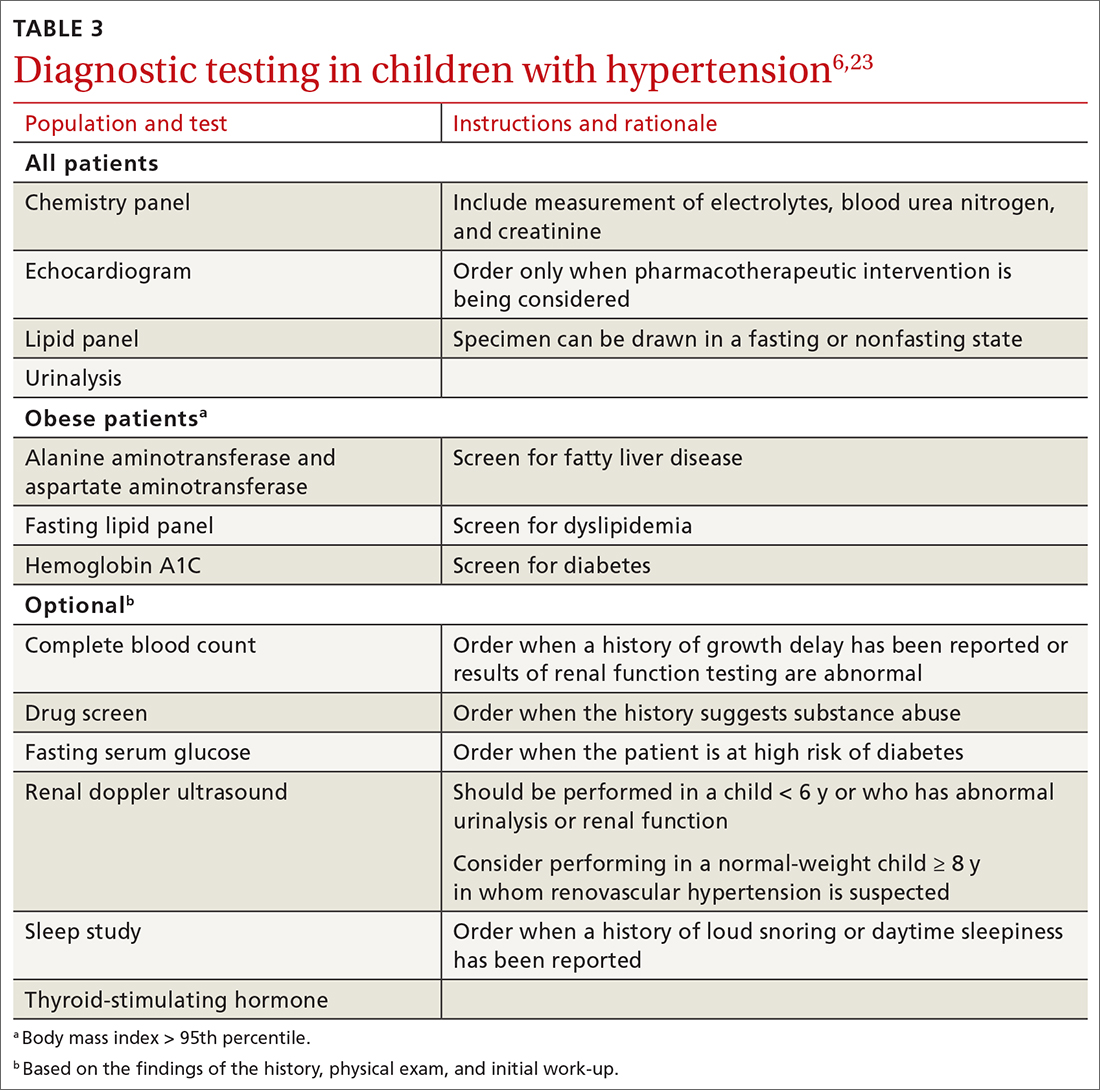
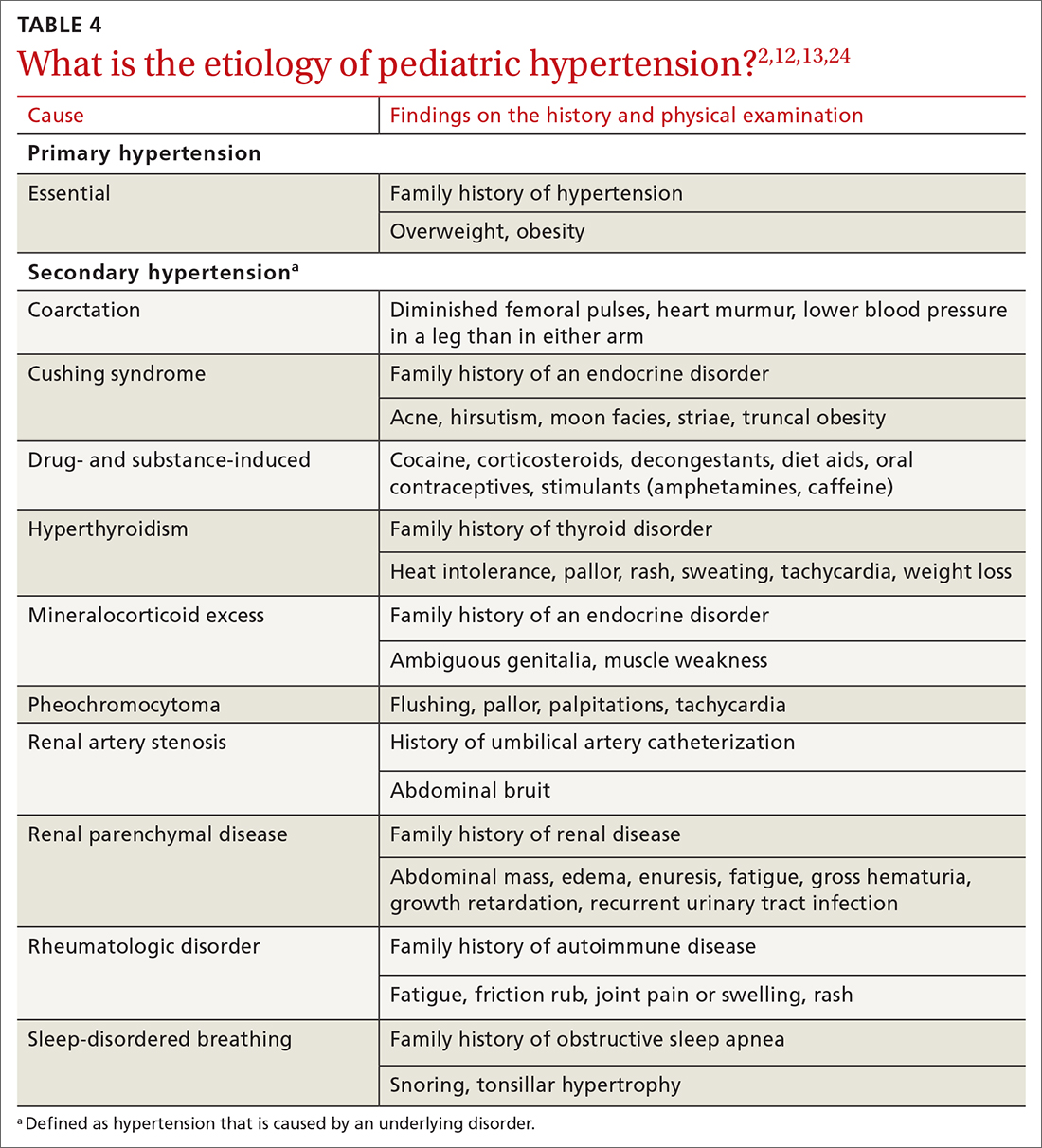
TABLE 42,12,13,24 outlines the basis of primary and of secondary hypertension and common historical and physical findings that suggest a secondary cause.
Mapping out the treatment plan
Pediatric hypertension should be treated in patients with stage 1 or higher hypertension.6 This threshold for therapy is based on evidence that reducing BP below a goal of (1) the 90th percentile (calculated based on age, sex, and height) in children up to 12 years of age or (2) of < 130/80 mm Hg for children ≥ 13 years reduces short- and long-term morbidity and mortality.5,6,25
Choice of initial treatment depends on the severity of BP elevation and the presence of comorbidities (FIGURE6,20,25-28). The initial, fundamental treatment recommendation is lifestyle modification,6,29 including regular physical exercise, a change in nutritional habits, weight loss (because obesity is a common comorbid condition), elimination of tobacco and substance use, and stress reduction.25,26 Medications can be used as well, along with other treatments for specific causes of secondary hypertension.
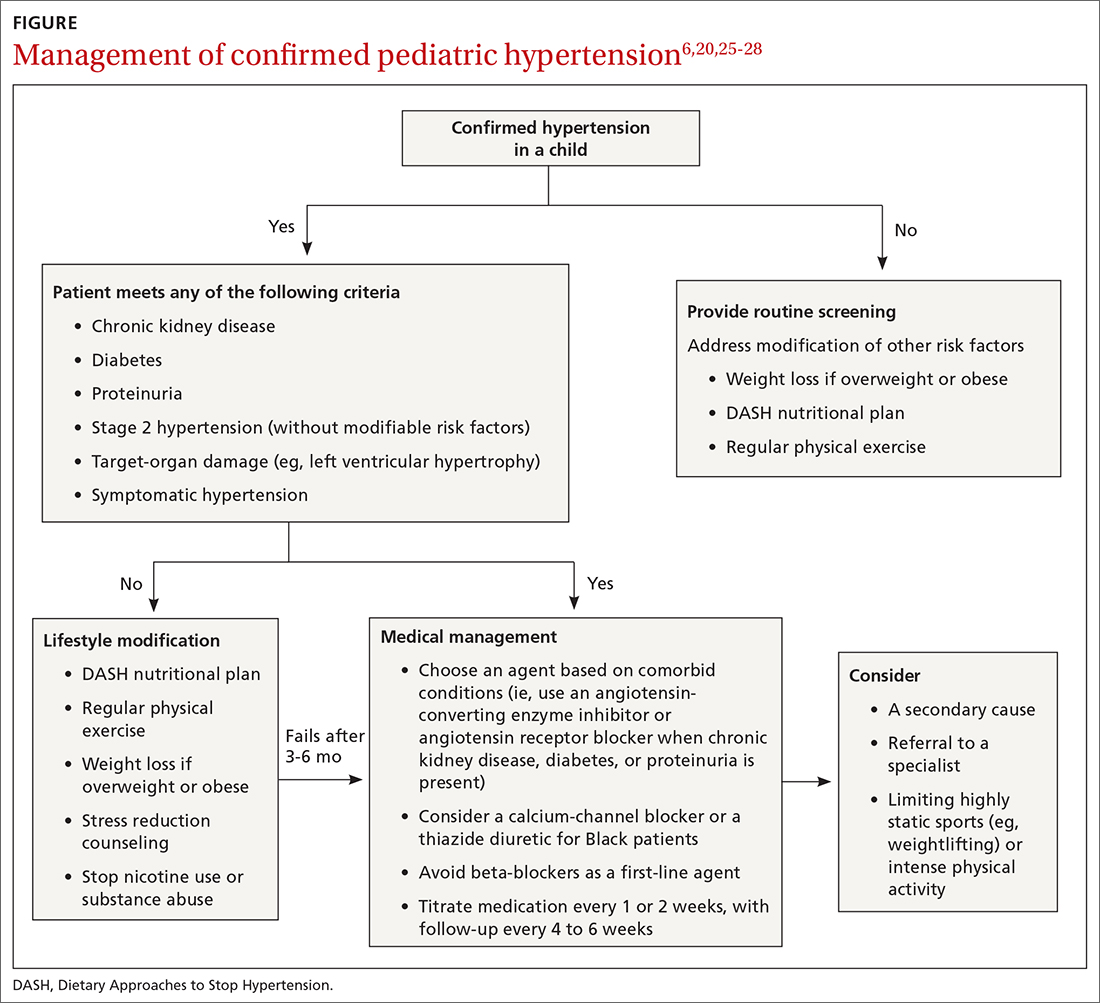
Referral to a specialist can be considered if consultation for assistance with treatment is preferred (TABLE 26) or if the patient has:
- treatment-resistant hypertension
- stage 2 hypertension that is not quickly responsive to initial treatment
- an identified secondary cause of hypertension.
Continue to: Lifestyle modification can make a big difference
Lifestyle modification can make a big difference
Exercise. “Regular” physical exercise for children to reduce BP is defined as ≥ 30 to 60 minutes of active play daily.6,29 Studies have shown significant improvement not only in BP but also in other cardiovascular disease risk parameters with regular physical exercise.27 A study found that the reduction in systolic BP is, on average, approximately 6 mm Hg with physical activity alone.30
Nutrition. DASH—Dietary Approaches to Stop Hypertension—is an evidence-based program to reduce BP. This nutritional guideline focuses on a diet rich in natural foods, including fruits, vegetables, minimally processed carbohydrates and whole grains, and low-fat dairy and meats. It also emphasizes the importance of avoiding foods high in processed sugars and reducing sodium intake.31 Higher-than-recommended sodium intake, based on age and sex (and established as part of dietary recommendations for children on the US Department of Health and Human Services’ website health.gov) directly correlates with the risk of prehypertension and hypertension—especially in overweight and obese children.20,32 DASH has been shown to reliably reduce the incidence of hypertension in children; other studies have supported increased intake of fruits, vegetables, and legumes as strategies to reduce BP.33,34
Other interventions. Techniques to improve adherence to exercise and nutritional modifications for children include motivational interviewing, community programs and education, and family counseling.27,35 A recent study showed that a community-based lifestyle modification program that is focused on weight loss in obese children resulted in a significant reduction in BP values at higher stages of obesity.36 There is evidence that techniques such as controlled breathing and meditation can reduce BP.37 Last, screening and counseling to encourage tobacco and substance use discontinuation are recommended for children and adolescents to improve health outcomes.25
Proceed with pharmacotherapy when these criteria are met
Medical therapy is recommended when certain criteria are met, although this decision should be individualized and made in agreement by the treating physician, patient, and family. These criteria (FIGURE6,20,25-28) are6,29:
- once a diagnosis of stage 1 hypertension has been established, failure to meet a BP goal after 3 to 6 months of attempting lifestyle modifications
- stage 2 hypertension without a modifiable risk factor, such as obesity
- any stage of hypertension with comorbid CKD, DM, or proteinuria
- target-organ damage, such as left ventricular hypertrophy
- symptomatic hypertension.6,29
There are circumstances in which one or another specific antihypertensive agent is recommended for children; however, for most patients with primary hypertension, the following classes are recommended for first-line use6,22:
- angiotensin-converting enzyme (ACE) inhibitors
- angiotensin receptor blockers (ARBs)
- calcium-channel blockers (CCBs)
- thiazide diuretics.
Continue to: For a child with known CKD...
For a child with known CKD, DM, or proteinuria, an ACE inhibitor or ARB is beneficial as first-line therapy.38 Because ACE inhibitors and ARBs have teratogenic effects, however, a thorough review of fertility status is recommended for female patients before any of these agents are started. CCBs and thiazides are typically recommended as first-line agents for Black patients.6,28 Beta-blockers are typically avoided in the first line because of their adverse effect profile.
Most antihypertensive medications can be titrated every 1 or 2 weeks; the patient’s BP can be monitored with a home BP cuff to track the effect of titration. In general, the patient should be seen for follow-up every 4 to 6 weeks for a BP recheck and review of medication tolerance and adverse effects. Once the treatment goal is achieved, it is reasonable to have the patient return every 3 to 6 months to reassess the treatment plan.
If the BP goal is difficult to achieve despite titration of medication and lifestyle changes, consider repeat ABPM assessment, a specialty referral, or both. It is reasonable for children who have been started on medication and have adhered to lifestyle modifications to practice a “step-down” approach to discontinuing medication; this approach can also be considered once any secondary cause has been corrected. Any target-organ abnormalities identified at diagnosis (eg, proteinuria, CKD, left ventricular hypertrophy) need to be reexamined at follow-up.6
Restrict activities—or not?
There is evidence that a child with stage 1 or well-controlled stage 2 hypertension without evidence of end-organ damage should not have restrictions on sports or activity. However, in uncontrolled stage 2 hypertension or when evidence of target end-organ damage is present, you should advise against participation in highly competitive sports and highly static sports (eg, weightlifting, wrestling), based on expert opinion6,25 (FIGURE6,20,25-28).
aAAP guidelines on the management of pediatric hypertension vary from those of the US Preventive Services Task Force. See the Practice Alert, “A review of the latest USPSTF recommendations,” in the May 2021 issue.
CORRESPONDENCE
Dustin K. Smith, MD, Family Medicine Department, 2080 Child Street, Jacksonville, FL, 32214; dustinksmith@yahoo.com
1. Theodore RF, Broadbent J, Nagin D, et al. Childhood to early-midlife systolic blood pressure trajectories: early-life predictors, effect modifiers, and adult cardiovascular outcomes. Hypertension. 2015;66:1108-1115. doi: 10.1161/HYPERTENSIONAHA.115.05831
2. Lurbe E, Agabiti-Rosei E, Cruickshank JK, et al. 2016 European Society of Hypertension guidelines for the management of high blood pressure in children and adolescents. J Hypertens. 2016;34:1887-1920. doi: 10.1097/HJH.0000000000001039
3. Weaver DJ, Mitsnefes MM. Effects of systemic hypertension on the cardiovascular system. Prog Pediatr Cardiol. 2016;41:59-65. https://doi.org/10.1016/j.ppedcard.2015.11.005
4. Ippisch HM, Daniels SR. Hypertension in overweight and obese children. Prog Pediatr Cardiol. 2008;25:177-182. doi: org/10.1016/j.ppedcard.2008.05.002
5. Urbina EM, Lande MB, Hooper SR, et al. Target organ abnormalities in pediatric hypertension. J Pediatr. 2018;202:14-22. doi: 10.1016/j.jpeds.2018.07.026
6. Flynn JT, Kaelber DC, Baker-Smith CM, et al; . Clinical practice guideline for screening and management of high blood pressure in children and adolescents. Pediatrics. 2017;140:e20171904. doi: 10.1542/peds.2017-1904
7. Khoury M, Khoury PR, Dolan LM, et al. Clinical implications of the revised AAP pediatric hypertension guidelines. Pediatrics. 2018;142:e20180245. doi: 10.1542/peds.2018-0245
8. Falkner B, Gidding SS, Ramirez-Garnica G, et al. The relationship of body mass index and blood pressure in primary care pediatric patients. J Pediatr. 2006;148:195-200. doi: 10.1016/j.jpeds.2005.10.030
9. Rodriguez BL, Dabelea D, Liese AD, et al; SEARCH Study Group. Prevalence and correlates of elevated blood pressure in youth with diabetes mellitus: the SEARCH for diabetes in youth study. J Pediatr. 2010;157:245-251.e1. doi: 10.1016/j.jpeds.2010.02.021
10. Shay CM, Ning H, Daniels SR, et al. Status of cardiovascular health in US adolescents: prevalence estimates from the National Health and Nutrition Examination Surveys (NHANES) 2005-2010. Circulation. 2013;127:1369-1376. doi: 10.1161/CIRCULATIONAHA.113.001559
11. Archbold KH, Vasquez MM, Goodwin JL, et al. Effects of sleep patterns and obesity on increases in blood pressure in a 5-year period: report from the Tucson Children’s Assessment of Sleep Apnea Study. J Pediatr. 2012;161:26-30. doi: 10.1016/j.jpeds.2011.12.034
12. Flynn JT, Mitsnefes M, Pierce C, et al; . Blood pressure in children with chronic kidney disease: a report from the Chronic Kidney Disease in Children study. Hypertension. 2008;52:631-637. doi: 10.1161/HYPERTENSIONAHA.108.110635
13. Martin RM, Ness AR, Gunnell D, et al; ALSPAC Study Team. Does breast-feeding in infancy lower blood pressure in childhood? The Avon Longitudinal Study of Parents and Children (ALSPAC). Circulation. 2004;109:1259-1266. doi: 10.1161/01.CIR.0000118468.76447.CE
14. Brickner ME, Hillis LD, Lange RA. Congenital heart disease in adults. N Engl J Med. 2000;342:256-263. doi: 10.1056/NEJM200001273420407
15. Chen X, Wang Y. Tracking of blood pressure from childhood to adulthood: a systematic review and meta-regression analysis. Circulation. 2008;117:3171-3180. doi: 10.1161/CIRCULATIONAHA.107.730366
16. Sun SS, Grave GD, Siervogel RM, et al. Systolic blood pressure in childhood predicts hypertension and metabolic syndrome later in life. Pediatrics. 2007;119:237-246. doi: 10.1542/peds.2006-2543
17. Parker ED, Sinaiko AR, Kharbanda EO, et al. Change in weight status and development of hypertension. Pediatrics. 2016; 137:e20151662. doi: 10.1542/peds.2015-1662
18. Pickering TG, Hall JE, Appel LJ, et al; . Recommendations for blood pressure measurement in humans and experimental animals: Part 1: blood pressure measurement in humans: a statement for professionals from the Subcommittee of Professional and Public Education of the American Heart Association Council on High Blood Pressure Research. Hypertension. 2005;45:142-161. doi: 10.1161/01.HYP.0000150859.47929.8e
19. Staley JR, Bradley J, Silverwood RJ, et al. Associations of blood pressure in pregnancy with offspring blood pressure trajectories during childhood and adolescence: findings from a prospective study. J Am Heart Assoc. 2015;4:e001422. doi: 10.1161/JAHA.114.001422
20. Yang Q, Zhang Z, Zuklina EV, et al. Sodium intake and blood pressure among US children and adolescents. Pediatrics. 2012;130:611-619. doi: 10.1542/peds.2011-3870
21. Le-Ha C, Beilin LJ, Burrows S, et al. Oral contraceptive use in girls and alcohol consumption in boys are associated with increased blood pressure in late adolescence. Eur J Prev Cardiol. 2013;20:947-955. doi: 10.1177/2047487312452966
22. Samuels JA, Franco K, Wan F, Sorof JM. Effect of stimulants on 24-h ambulatory blood pressure in children with ADHD: a double-blind, randomized, cross-over trial. Pediatr Nephrol. 2006;21:92-95. doi: 10.1007/s00467-005-2051-1
23. Wiesen J, Adkins M, Fortune S, et al. Evaluation of pediatric patients with mild-to-moderate hypertension: yield of diagnostic testing. Pediatrics. 2008;122:e988-993. doi: 10.1542/peds.2008-0365
24. Kapur G, Ahmed M, Pan C, et al. Secondary hypertension in overweight and stage 1 hypertensive children: a Midwest Pediatric Nephrology Consortium report. J Clin Hypertens (Greenwich). 2010;12:34-39. doi: 10.1111/j.1751-7176.2009.00195.x
25. Anyaegbu EI, Dharnidharka VR. Hypertension in the teenager. Pediatr Clin North Am. 2014;61:131-151. doi: 10.1016/j.pcl.2013.09.011
26. Gandhi B, Cheek S, Campo JV. Anxiety in the pediatric medical setting. Child Adolesc Psychiatr Clin N Am. 2012;21:643-653. doi: 10.1016/j.chc.2012.05.013
27. Farpour-Lambert NJ, Aggoun Y, Marchand LM, et al. Physical activity reduces systemic blood pressure and improves early markers of atherosclerosis in pre-pubertal obese children. J Am Coll Cardiol. 2009;54:2396-2406. doi: 10.1016/j.jacc.2009.08.030
28. Li JS, Baker-Smith CM, Smith PB, et al. Racial differences in blood pressure response to angiotensin-converting enzyme inhibitors in children: a meta-analysis. Clin Pharmacol Ther. 2008;84:315-319. doi: 10.1038/clpt.2008.113
29. Singer PS. Updates on hypertension and new guidelines. Adv Pediatr. 2019;66:177-187. doi: 10.1016/j.yapd.2019.03.009
30. Torrance B, McGuire KA, Lewanczuk R, et al. Overweight, physical activity and high blood pressure in children: a review of the literature. Vasc Health Risk Manag. 2007;3:139-149.
31. DASH eating plan. National Heart, Lung, and Blood Institute. Accessed April 26, 2021. www.nhlbi.nih.gov/health-topics/dash-eating-plan
32. Nutritional goals for age-sex groups based on dietary reference intakes and dietary guidelines recommendations (Appendix 7). In: US Department of Agriculture. Dietary guidelines for Americans, 2015-2020. 8th ed. December 2015;97-98. Accessed April 26, 2021. https://health.gov/sites/default/files/2019-09/2015-2020_Dietary_Guidelines.pdf
33. Asghari G, Yuzbashian E, Mirmiran P, et al. Dietary Approaches to Stop Hypertension (DASH) dietary pattern is associated with reduced incidence of metabolic syndrome in children and adolescents. J Pediatr. 2016;174:178-184.e1. doi: 10.1016/j.jpeds.2016.03.077
34. Damasceno MMC, de Araújo MFM, de Freitas RWJF, et al. The association between blood pressure in adolescents and the consumption of fruits, vegetables and fruit juice–an exploratory study. J Clin Nurs. 2011;20:1553-1560. doi: 10.1111/j.1365-2702.2010.03608.x
35. Anderson KL. A review of the prevention and medical management of childhood obesity. Child Adolesc Psychiatr Clin N Am. 2018;27:63-76. doi: 10.1016/j.chc.2017.08.003
36. Kumar S, King EC, Christison, et al; POWER Work Group. Health outcomes of youth in clinical pediatric weight management programs in POWER. J Pediatr. 2019;208:57-65.e4. doi: 10.1016/j.jpeds.2018.12.049
37. Gregoski MJ, Barnes VA, Tingen MS, et al. Breathing awareness meditation and LifeSkills® Training programs influence upon ambulatory blood pressure and sodium excretion among African American adolescents. J Adolesc Health. 2011;48:59-64. doi: 10.1016/j.jadohealth.2010.05.019
38. Escape Trial Group; E, Trivelli A, Picca S, et al. Strict blood-pressure control and progression of renal failure in children. N Engl J Med. 2009;361:1639-1650. doi: 10.1056/NEJMoa0902066
Hypertension and elevated blood pressure (BP) in children and adolescents correlate to hypertension in adults, insofar as complications and medical therapy increase with age.1,2 Untreated, hypertension in children and adolescents can result in multiple harmful physiologic changes, including left ventricular hypertrophy, left atrial enlargement, diastolic dysfunction, arterial stiffening, endothelial dysfunction, and neurocognitive deficits.3-5
In 2017, the American Academy of Pediatrics (AAP) published clinical practice guidelines for the diagnosis and management of elevated BP and hypertension in children and adolescentsa (TABLE 16). Applying the definition of elevated BP set out in these guidelines yielded a 13% prevalence of hypertension in a cohort of subjects 10 to 18 years of age with comorbid obesity and diabetes mellitus (DM). AAP guideline definitions also improved the sensitivity for identifying hypertensive end-organ damage.7

As the prevalence of hypertension increases, screening for and accurate diagnosis of this condition in children are becoming more important. Recognition and management remain a vital part of primary care. In this article, we review the updated guidance on diagnosis and treatment, including lifestyle modification and pharmacotherapy.
First step: Identifying hypertension
Risk factors
Risk factors for pediatric hypertension are similar to those in adults. These include obesity (body mass index ≥ 95th percentile for age), types 1 and 2 DM, elevated sodium intake, sleep-disordered breathing, and chronic kidney disease (CKD). Some risk factors, such as premature birth and coarctation of the aorta, are specific to the pediatric population.8-14 Pediatric obesity strongly correlates with both pediatric and adult hypertension, and accelerated weight gain might increase the risk of elevated BP in adulthood.15,16

Intervening early to mitigate or eliminate some of these modifiable risk factors can prevent or treat hypertension.17 Alternatively, having been breastfed as an infant has been reliably shown to reduce the risk of elevated BP in children.13
Recommendations for screening and measuring BP
The optimal age to start measuring BP is not clearly defined. AAP recommends measurement:
- annually in all children ≥ 3 years of age
- at every encounter in patients who have a specific comorbid condition, including obesity, DM, renal disease, and aortic-arch abnormalities (obstruction and coarctation) and in those who are taking medication known to increase BP.6
Protocol. Measure BP in the right arm for consistency and comparison with reference values. The width of the cuff bladder should be at least 40%, and the length, 80% to 100%, of arm circumference. Position the cuff bladder midway between the olecranon and acromion. Obtain the measurement in a quiet and comfortable environment after the patient has rested for 3 to 5 minutes. The patient should be seated, preferably with feet on the floor; elbows should be supported at the level of the heart.
Continue to: When an initial reading...
When an initial reading is elevated, whether by oscillometric or auscultatory measurement, 2 more auscultatory BP measurements should be taken during the same visit; these measurements are averaged to determine the BP category.18
TABLE 16 defines BP categories based on age, sex, and height. We recommend using the free resource MD Calc (www.mdcalc.com/aap-pediatric-hypertension-guidelines) to assist in calculating the BP category.
TABLE 26 describes the timing of follow-up based on the initial BP reading and diagnosis.
Ambulatory BP monitoring (ABPM) is a validated device that measures BP every 20 to 30 minutes throughout the day and night. ABPM should be performed initially in all patients with persistently elevated BP and routinely in children and adolescents with a high-risk comorbidity (TABLE 26). Note: Insurance coverage of ABPM is limited.

ABPM is also used to diagnose so-called white-coat hypertension, defined as BP ≥ 95th percentile for age, sex, and height in the clinic setting but < 95th percentile during ABPM. This phenomenon can be challenging to diagnose.
Continue to: Home monitoring
Home monitoring. Do not use home BP monitoring to establish a diagnosis of hypertension, although one of these devices can be used as an adjunct to office and ambulatory BP monitoring after the diagnosis has been made.6
Evaluating hypertension in children and adolescents
Once a diagnosis of hypertension has been made, undertake a thorough history, physical examination, and diagnostic testing to evaluate for possible causes, comorbidities, and any evidence of end-organ damage.
Comprehensive history. Pertinent aspects include perinatal, nutritional, physical activity, psychosocial, family, medication—and of course, medical—histories.6
Maternal elevated BP or hypertension is related to an offspring’s elevated BP in childhood and adolescence.19 Other pertinent aspects of the perinatal history include complications of pregnancy, gestational age, birth weight, and neonatal complications.6
Nutritional and physical activity histories can highlight contributing factors in the development of hypertension and can be a guide to recommending lifestyle modifications.6 Sodium intake, which influences BP, should be part of the nutritional history.20
Continue to: Important aspects...
Important aspects of the psychosocial history include feelings of depression or anxiety, bullying, and body perception. Children older than 10 years should be asked about smoking, alcohol, and other substance use.
The family history should include notation of first- and second-degree relatives with hypertension.6
Inquire about medications that can raise BP, including oral contraceptives, which are commonly prescribed in this population.21,22
The physical exam should include measured height and weight, with calculation of the body mass index percentile for age; of note, obesity is strongly associated with hypertension, and poor growth might signal underlying chronic disease. Once elevated BP has been confirmed, the exam should include measurement of BP in both arms and in a leg (TABLE 26). BP that is lower in the leg than in the arms (in any given patient, BP readings in the legs are usually higher than in the arms), or weak or absent femoral pulses, suggest coarctation of the aorta.6
Focus the balance of the physical exam on physical findings that suggest secondary causes of hypertension or evidence of end-organ damage.
Continue to: Testing
Testing. TABLE 36,23 summarizes the diagnostic testing recommended for all children and for specific populations; TABLE 26 indicates when to obtain diagnostic testing.

TABLE 42,12,13,24 outlines the basis of primary and of secondary hypertension and common historical and physical findings that suggest a secondary cause.

Mapping out the treatment plan
Pediatric hypertension should be treated in patients with stage 1 or higher hypertension.6 This threshold for therapy is based on evidence that reducing BP below a goal of (1) the 90th percentile (calculated based on age, sex, and height) in children up to 12 years of age or (2) of < 130/80 mm Hg for children ≥ 13 years reduces short- and long-term morbidity and mortality.5,6,25
Choice of initial treatment depends on the severity of BP elevation and the presence of comorbidities (FIGURE6,20,25-28). The initial, fundamental treatment recommendation is lifestyle modification,6,29 including regular physical exercise, a change in nutritional habits, weight loss (because obesity is a common comorbid condition), elimination of tobacco and substance use, and stress reduction.25,26 Medications can be used as well, along with other treatments for specific causes of secondary hypertension.

Referral to a specialist can be considered if consultation for assistance with treatment is preferred (TABLE 26) or if the patient has:
- treatment-resistant hypertension
- stage 2 hypertension that is not quickly responsive to initial treatment
- an identified secondary cause of hypertension.
Continue to: Lifestyle modification can make a big difference
Lifestyle modification can make a big difference
Exercise. “Regular” physical exercise for children to reduce BP is defined as ≥ 30 to 60 minutes of active play daily.6,29 Studies have shown significant improvement not only in BP but also in other cardiovascular disease risk parameters with regular physical exercise.27 A study found that the reduction in systolic BP is, on average, approximately 6 mm Hg with physical activity alone.30
Nutrition. DASH—Dietary Approaches to Stop Hypertension—is an evidence-based program to reduce BP. This nutritional guideline focuses on a diet rich in natural foods, including fruits, vegetables, minimally processed carbohydrates and whole grains, and low-fat dairy and meats. It also emphasizes the importance of avoiding foods high in processed sugars and reducing sodium intake.31 Higher-than-recommended sodium intake, based on age and sex (and established as part of dietary recommendations for children on the US Department of Health and Human Services’ website health.gov) directly correlates with the risk of prehypertension and hypertension—especially in overweight and obese children.20,32 DASH has been shown to reliably reduce the incidence of hypertension in children; other studies have supported increased intake of fruits, vegetables, and legumes as strategies to reduce BP.33,34
Other interventions. Techniques to improve adherence to exercise and nutritional modifications for children include motivational interviewing, community programs and education, and family counseling.27,35 A recent study showed that a community-based lifestyle modification program that is focused on weight loss in obese children resulted in a significant reduction in BP values at higher stages of obesity.36 There is evidence that techniques such as controlled breathing and meditation can reduce BP.37 Last, screening and counseling to encourage tobacco and substance use discontinuation are recommended for children and adolescents to improve health outcomes.25
Proceed with pharmacotherapy when these criteria are met
Medical therapy is recommended when certain criteria are met, although this decision should be individualized and made in agreement by the treating physician, patient, and family. These criteria (FIGURE6,20,25-28) are6,29:
- once a diagnosis of stage 1 hypertension has been established, failure to meet a BP goal after 3 to 6 months of attempting lifestyle modifications
- stage 2 hypertension without a modifiable risk factor, such as obesity
- any stage of hypertension with comorbid CKD, DM, or proteinuria
- target-organ damage, such as left ventricular hypertrophy
- symptomatic hypertension.6,29
There are circumstances in which one or another specific antihypertensive agent is recommended for children; however, for most patients with primary hypertension, the following classes are recommended for first-line use6,22:
- angiotensin-converting enzyme (ACE) inhibitors
- angiotensin receptor blockers (ARBs)
- calcium-channel blockers (CCBs)
- thiazide diuretics.
Continue to: For a child with known CKD...
For a child with known CKD, DM, or proteinuria, an ACE inhibitor or ARB is beneficial as first-line therapy.38 Because ACE inhibitors and ARBs have teratogenic effects, however, a thorough review of fertility status is recommended for female patients before any of these agents are started. CCBs and thiazides are typically recommended as first-line agents for Black patients.6,28 Beta-blockers are typically avoided in the first line because of their adverse effect profile.
Most antihypertensive medications can be titrated every 1 or 2 weeks; the patient’s BP can be monitored with a home BP cuff to track the effect of titration. In general, the patient should be seen for follow-up every 4 to 6 weeks for a BP recheck and review of medication tolerance and adverse effects. Once the treatment goal is achieved, it is reasonable to have the patient return every 3 to 6 months to reassess the treatment plan.
If the BP goal is difficult to achieve despite titration of medication and lifestyle changes, consider repeat ABPM assessment, a specialty referral, or both. It is reasonable for children who have been started on medication and have adhered to lifestyle modifications to practice a “step-down” approach to discontinuing medication; this approach can also be considered once any secondary cause has been corrected. Any target-organ abnormalities identified at diagnosis (eg, proteinuria, CKD, left ventricular hypertrophy) need to be reexamined at follow-up.6
Restrict activities—or not?
There is evidence that a child with stage 1 or well-controlled stage 2 hypertension without evidence of end-organ damage should not have restrictions on sports or activity. However, in uncontrolled stage 2 hypertension or when evidence of target end-organ damage is present, you should advise against participation in highly competitive sports and highly static sports (eg, weightlifting, wrestling), based on expert opinion6,25 (FIGURE6,20,25-28).
aAAP guidelines on the management of pediatric hypertension vary from those of the US Preventive Services Task Force. See the Practice Alert, “A review of the latest USPSTF recommendations,” in the May 2021 issue.
CORRESPONDENCE
Dustin K. Smith, MD, Family Medicine Department, 2080 Child Street, Jacksonville, FL, 32214; dustinksmith@yahoo.com
Hypertension and elevated blood pressure (BP) in children and adolescents correlate to hypertension in adults, insofar as complications and medical therapy increase with age.1,2 Untreated, hypertension in children and adolescents can result in multiple harmful physiologic changes, including left ventricular hypertrophy, left atrial enlargement, diastolic dysfunction, arterial stiffening, endothelial dysfunction, and neurocognitive deficits.3-5
In 2017, the American Academy of Pediatrics (AAP) published clinical practice guidelines for the diagnosis and management of elevated BP and hypertension in children and adolescentsa (TABLE 16). Applying the definition of elevated BP set out in these guidelines yielded a 13% prevalence of hypertension in a cohort of subjects 10 to 18 years of age with comorbid obesity and diabetes mellitus (DM). AAP guideline definitions also improved the sensitivity for identifying hypertensive end-organ damage.7

As the prevalence of hypertension increases, screening for and accurate diagnosis of this condition in children are becoming more important. Recognition and management remain a vital part of primary care. In this article, we review the updated guidance on diagnosis and treatment, including lifestyle modification and pharmacotherapy.
First step: Identifying hypertension
Risk factors
Risk factors for pediatric hypertension are similar to those in adults. These include obesity (body mass index ≥ 95th percentile for age), types 1 and 2 DM, elevated sodium intake, sleep-disordered breathing, and chronic kidney disease (CKD). Some risk factors, such as premature birth and coarctation of the aorta, are specific to the pediatric population.8-14 Pediatric obesity strongly correlates with both pediatric and adult hypertension, and accelerated weight gain might increase the risk of elevated BP in adulthood.15,16

Intervening early to mitigate or eliminate some of these modifiable risk factors can prevent or treat hypertension.17 Alternatively, having been breastfed as an infant has been reliably shown to reduce the risk of elevated BP in children.13
Recommendations for screening and measuring BP
The optimal age to start measuring BP is not clearly defined. AAP recommends measurement:
- annually in all children ≥ 3 years of age
- at every encounter in patients who have a specific comorbid condition, including obesity, DM, renal disease, and aortic-arch abnormalities (obstruction and coarctation) and in those who are taking medication known to increase BP.6
Protocol. Measure BP in the right arm for consistency and comparison with reference values. The width of the cuff bladder should be at least 40%, and the length, 80% to 100%, of arm circumference. Position the cuff bladder midway between the olecranon and acromion. Obtain the measurement in a quiet and comfortable environment after the patient has rested for 3 to 5 minutes. The patient should be seated, preferably with feet on the floor; elbows should be supported at the level of the heart.
Continue to: When an initial reading...
When an initial reading is elevated, whether by oscillometric or auscultatory measurement, 2 more auscultatory BP measurements should be taken during the same visit; these measurements are averaged to determine the BP category.18
TABLE 16 defines BP categories based on age, sex, and height. We recommend using the free resource MD Calc (www.mdcalc.com/aap-pediatric-hypertension-guidelines) to assist in calculating the BP category.
TABLE 26 describes the timing of follow-up based on the initial BP reading and diagnosis.
Ambulatory BP monitoring (ABPM) is a validated device that measures BP every 20 to 30 minutes throughout the day and night. ABPM should be performed initially in all patients with persistently elevated BP and routinely in children and adolescents with a high-risk comorbidity (TABLE 26). Note: Insurance coverage of ABPM is limited.

ABPM is also used to diagnose so-called white-coat hypertension, defined as BP ≥ 95th percentile for age, sex, and height in the clinic setting but < 95th percentile during ABPM. This phenomenon can be challenging to diagnose.
Continue to: Home monitoring
Home monitoring. Do not use home BP monitoring to establish a diagnosis of hypertension, although one of these devices can be used as an adjunct to office and ambulatory BP monitoring after the diagnosis has been made.6
Evaluating hypertension in children and adolescents
Once a diagnosis of hypertension has been made, undertake a thorough history, physical examination, and diagnostic testing to evaluate for possible causes, comorbidities, and any evidence of end-organ damage.
Comprehensive history. Pertinent aspects include perinatal, nutritional, physical activity, psychosocial, family, medication—and of course, medical—histories.6
Maternal elevated BP or hypertension is related to an offspring’s elevated BP in childhood and adolescence.19 Other pertinent aspects of the perinatal history include complications of pregnancy, gestational age, birth weight, and neonatal complications.6
Nutritional and physical activity histories can highlight contributing factors in the development of hypertension and can be a guide to recommending lifestyle modifications.6 Sodium intake, which influences BP, should be part of the nutritional history.20
Continue to: Important aspects...
Important aspects of the psychosocial history include feelings of depression or anxiety, bullying, and body perception. Children older than 10 years should be asked about smoking, alcohol, and other substance use.
The family history should include notation of first- and second-degree relatives with hypertension.6
Inquire about medications that can raise BP, including oral contraceptives, which are commonly prescribed in this population.21,22
The physical exam should include measured height and weight, with calculation of the body mass index percentile for age; of note, obesity is strongly associated with hypertension, and poor growth might signal underlying chronic disease. Once elevated BP has been confirmed, the exam should include measurement of BP in both arms and in a leg (TABLE 26). BP that is lower in the leg than in the arms (in any given patient, BP readings in the legs are usually higher than in the arms), or weak or absent femoral pulses, suggest coarctation of the aorta.6
Focus the balance of the physical exam on physical findings that suggest secondary causes of hypertension or evidence of end-organ damage.
Continue to: Testing
Testing. TABLE 36,23 summarizes the diagnostic testing recommended for all children and for specific populations; TABLE 26 indicates when to obtain diagnostic testing.

TABLE 42,12,13,24 outlines the basis of primary and of secondary hypertension and common historical and physical findings that suggest a secondary cause.

Mapping out the treatment plan
Pediatric hypertension should be treated in patients with stage 1 or higher hypertension.6 This threshold for therapy is based on evidence that reducing BP below a goal of (1) the 90th percentile (calculated based on age, sex, and height) in children up to 12 years of age or (2) of < 130/80 mm Hg for children ≥ 13 years reduces short- and long-term morbidity and mortality.5,6,25
Choice of initial treatment depends on the severity of BP elevation and the presence of comorbidities (FIGURE6,20,25-28). The initial, fundamental treatment recommendation is lifestyle modification,6,29 including regular physical exercise, a change in nutritional habits, weight loss (because obesity is a common comorbid condition), elimination of tobacco and substance use, and stress reduction.25,26 Medications can be used as well, along with other treatments for specific causes of secondary hypertension.

Referral to a specialist can be considered if consultation for assistance with treatment is preferred (TABLE 26) or if the patient has:
- treatment-resistant hypertension
- stage 2 hypertension that is not quickly responsive to initial treatment
- an identified secondary cause of hypertension.
Continue to: Lifestyle modification can make a big difference
Lifestyle modification can make a big difference
Exercise. “Regular” physical exercise for children to reduce BP is defined as ≥ 30 to 60 minutes of active play daily.6,29 Studies have shown significant improvement not only in BP but also in other cardiovascular disease risk parameters with regular physical exercise.27 A study found that the reduction in systolic BP is, on average, approximately 6 mm Hg with physical activity alone.30
Nutrition. DASH—Dietary Approaches to Stop Hypertension—is an evidence-based program to reduce BP. This nutritional guideline focuses on a diet rich in natural foods, including fruits, vegetables, minimally processed carbohydrates and whole grains, and low-fat dairy and meats. It also emphasizes the importance of avoiding foods high in processed sugars and reducing sodium intake.31 Higher-than-recommended sodium intake, based on age and sex (and established as part of dietary recommendations for children on the US Department of Health and Human Services’ website health.gov) directly correlates with the risk of prehypertension and hypertension—especially in overweight and obese children.20,32 DASH has been shown to reliably reduce the incidence of hypertension in children; other studies have supported increased intake of fruits, vegetables, and legumes as strategies to reduce BP.33,34
Other interventions. Techniques to improve adherence to exercise and nutritional modifications for children include motivational interviewing, community programs and education, and family counseling.27,35 A recent study showed that a community-based lifestyle modification program that is focused on weight loss in obese children resulted in a significant reduction in BP values at higher stages of obesity.36 There is evidence that techniques such as controlled breathing and meditation can reduce BP.37 Last, screening and counseling to encourage tobacco and substance use discontinuation are recommended for children and adolescents to improve health outcomes.25
Proceed with pharmacotherapy when these criteria are met
Medical therapy is recommended when certain criteria are met, although this decision should be individualized and made in agreement by the treating physician, patient, and family. These criteria (FIGURE6,20,25-28) are6,29:
- once a diagnosis of stage 1 hypertension has been established, failure to meet a BP goal after 3 to 6 months of attempting lifestyle modifications
- stage 2 hypertension without a modifiable risk factor, such as obesity
- any stage of hypertension with comorbid CKD, DM, or proteinuria
- target-organ damage, such as left ventricular hypertrophy
- symptomatic hypertension.6,29
There are circumstances in which one or another specific antihypertensive agent is recommended for children; however, for most patients with primary hypertension, the following classes are recommended for first-line use6,22:
- angiotensin-converting enzyme (ACE) inhibitors
- angiotensin receptor blockers (ARBs)
- calcium-channel blockers (CCBs)
- thiazide diuretics.
Continue to: For a child with known CKD...
For a child with known CKD, DM, or proteinuria, an ACE inhibitor or ARB is beneficial as first-line therapy.38 Because ACE inhibitors and ARBs have teratogenic effects, however, a thorough review of fertility status is recommended for female patients before any of these agents are started. CCBs and thiazides are typically recommended as first-line agents for Black patients.6,28 Beta-blockers are typically avoided in the first line because of their adverse effect profile.
Most antihypertensive medications can be titrated every 1 or 2 weeks; the patient’s BP can be monitored with a home BP cuff to track the effect of titration. In general, the patient should be seen for follow-up every 4 to 6 weeks for a BP recheck and review of medication tolerance and adverse effects. Once the treatment goal is achieved, it is reasonable to have the patient return every 3 to 6 months to reassess the treatment plan.
If the BP goal is difficult to achieve despite titration of medication and lifestyle changes, consider repeat ABPM assessment, a specialty referral, or both. It is reasonable for children who have been started on medication and have adhered to lifestyle modifications to practice a “step-down” approach to discontinuing medication; this approach can also be considered once any secondary cause has been corrected. Any target-organ abnormalities identified at diagnosis (eg, proteinuria, CKD, left ventricular hypertrophy) need to be reexamined at follow-up.6
Restrict activities—or not?
There is evidence that a child with stage 1 or well-controlled stage 2 hypertension without evidence of end-organ damage should not have restrictions on sports or activity. However, in uncontrolled stage 2 hypertension or when evidence of target end-organ damage is present, you should advise against participation in highly competitive sports and highly static sports (eg, weightlifting, wrestling), based on expert opinion6,25 (FIGURE6,20,25-28).
aAAP guidelines on the management of pediatric hypertension vary from those of the US Preventive Services Task Force. See the Practice Alert, “A review of the latest USPSTF recommendations,” in the May 2021 issue.
CORRESPONDENCE
Dustin K. Smith, MD, Family Medicine Department, 2080 Child Street, Jacksonville, FL, 32214; dustinksmith@yahoo.com
1. Theodore RF, Broadbent J, Nagin D, et al. Childhood to early-midlife systolic blood pressure trajectories: early-life predictors, effect modifiers, and adult cardiovascular outcomes. Hypertension. 2015;66:1108-1115. doi: 10.1161/HYPERTENSIONAHA.115.05831
2. Lurbe E, Agabiti-Rosei E, Cruickshank JK, et al. 2016 European Society of Hypertension guidelines for the management of high blood pressure in children and adolescents. J Hypertens. 2016;34:1887-1920. doi: 10.1097/HJH.0000000000001039
3. Weaver DJ, Mitsnefes MM. Effects of systemic hypertension on the cardiovascular system. Prog Pediatr Cardiol. 2016;41:59-65. https://doi.org/10.1016/j.ppedcard.2015.11.005
4. Ippisch HM, Daniels SR. Hypertension in overweight and obese children. Prog Pediatr Cardiol. 2008;25:177-182. doi: org/10.1016/j.ppedcard.2008.05.002
5. Urbina EM, Lande MB, Hooper SR, et al. Target organ abnormalities in pediatric hypertension. J Pediatr. 2018;202:14-22. doi: 10.1016/j.jpeds.2018.07.026
6. Flynn JT, Kaelber DC, Baker-Smith CM, et al; . Clinical practice guideline for screening and management of high blood pressure in children and adolescents. Pediatrics. 2017;140:e20171904. doi: 10.1542/peds.2017-1904
7. Khoury M, Khoury PR, Dolan LM, et al. Clinical implications of the revised AAP pediatric hypertension guidelines. Pediatrics. 2018;142:e20180245. doi: 10.1542/peds.2018-0245
8. Falkner B, Gidding SS, Ramirez-Garnica G, et al. The relationship of body mass index and blood pressure in primary care pediatric patients. J Pediatr. 2006;148:195-200. doi: 10.1016/j.jpeds.2005.10.030
9. Rodriguez BL, Dabelea D, Liese AD, et al; SEARCH Study Group. Prevalence and correlates of elevated blood pressure in youth with diabetes mellitus: the SEARCH for diabetes in youth study. J Pediatr. 2010;157:245-251.e1. doi: 10.1016/j.jpeds.2010.02.021
10. Shay CM, Ning H, Daniels SR, et al. Status of cardiovascular health in US adolescents: prevalence estimates from the National Health and Nutrition Examination Surveys (NHANES) 2005-2010. Circulation. 2013;127:1369-1376. doi: 10.1161/CIRCULATIONAHA.113.001559
11. Archbold KH, Vasquez MM, Goodwin JL, et al. Effects of sleep patterns and obesity on increases in blood pressure in a 5-year period: report from the Tucson Children’s Assessment of Sleep Apnea Study. J Pediatr. 2012;161:26-30. doi: 10.1016/j.jpeds.2011.12.034
12. Flynn JT, Mitsnefes M, Pierce C, et al; . Blood pressure in children with chronic kidney disease: a report from the Chronic Kidney Disease in Children study. Hypertension. 2008;52:631-637. doi: 10.1161/HYPERTENSIONAHA.108.110635
13. Martin RM, Ness AR, Gunnell D, et al; ALSPAC Study Team. Does breast-feeding in infancy lower blood pressure in childhood? The Avon Longitudinal Study of Parents and Children (ALSPAC). Circulation. 2004;109:1259-1266. doi: 10.1161/01.CIR.0000118468.76447.CE
14. Brickner ME, Hillis LD, Lange RA. Congenital heart disease in adults. N Engl J Med. 2000;342:256-263. doi: 10.1056/NEJM200001273420407
15. Chen X, Wang Y. Tracking of blood pressure from childhood to adulthood: a systematic review and meta-regression analysis. Circulation. 2008;117:3171-3180. doi: 10.1161/CIRCULATIONAHA.107.730366
16. Sun SS, Grave GD, Siervogel RM, et al. Systolic blood pressure in childhood predicts hypertension and metabolic syndrome later in life. Pediatrics. 2007;119:237-246. doi: 10.1542/peds.2006-2543
17. Parker ED, Sinaiko AR, Kharbanda EO, et al. Change in weight status and development of hypertension. Pediatrics. 2016; 137:e20151662. doi: 10.1542/peds.2015-1662
18. Pickering TG, Hall JE, Appel LJ, et al; . Recommendations for blood pressure measurement in humans and experimental animals: Part 1: blood pressure measurement in humans: a statement for professionals from the Subcommittee of Professional and Public Education of the American Heart Association Council on High Blood Pressure Research. Hypertension. 2005;45:142-161. doi: 10.1161/01.HYP.0000150859.47929.8e
19. Staley JR, Bradley J, Silverwood RJ, et al. Associations of blood pressure in pregnancy with offspring blood pressure trajectories during childhood and adolescence: findings from a prospective study. J Am Heart Assoc. 2015;4:e001422. doi: 10.1161/JAHA.114.001422
20. Yang Q, Zhang Z, Zuklina EV, et al. Sodium intake and blood pressure among US children and adolescents. Pediatrics. 2012;130:611-619. doi: 10.1542/peds.2011-3870
21. Le-Ha C, Beilin LJ, Burrows S, et al. Oral contraceptive use in girls and alcohol consumption in boys are associated with increased blood pressure in late adolescence. Eur J Prev Cardiol. 2013;20:947-955. doi: 10.1177/2047487312452966
22. Samuels JA, Franco K, Wan F, Sorof JM. Effect of stimulants on 24-h ambulatory blood pressure in children with ADHD: a double-blind, randomized, cross-over trial. Pediatr Nephrol. 2006;21:92-95. doi: 10.1007/s00467-005-2051-1
23. Wiesen J, Adkins M, Fortune S, et al. Evaluation of pediatric patients with mild-to-moderate hypertension: yield of diagnostic testing. Pediatrics. 2008;122:e988-993. doi: 10.1542/peds.2008-0365
24. Kapur G, Ahmed M, Pan C, et al. Secondary hypertension in overweight and stage 1 hypertensive children: a Midwest Pediatric Nephrology Consortium report. J Clin Hypertens (Greenwich). 2010;12:34-39. doi: 10.1111/j.1751-7176.2009.00195.x
25. Anyaegbu EI, Dharnidharka VR. Hypertension in the teenager. Pediatr Clin North Am. 2014;61:131-151. doi: 10.1016/j.pcl.2013.09.011
26. Gandhi B, Cheek S, Campo JV. Anxiety in the pediatric medical setting. Child Adolesc Psychiatr Clin N Am. 2012;21:643-653. doi: 10.1016/j.chc.2012.05.013
27. Farpour-Lambert NJ, Aggoun Y, Marchand LM, et al. Physical activity reduces systemic blood pressure and improves early markers of atherosclerosis in pre-pubertal obese children. J Am Coll Cardiol. 2009;54:2396-2406. doi: 10.1016/j.jacc.2009.08.030
28. Li JS, Baker-Smith CM, Smith PB, et al. Racial differences in blood pressure response to angiotensin-converting enzyme inhibitors in children: a meta-analysis. Clin Pharmacol Ther. 2008;84:315-319. doi: 10.1038/clpt.2008.113
29. Singer PS. Updates on hypertension and new guidelines. Adv Pediatr. 2019;66:177-187. doi: 10.1016/j.yapd.2019.03.009
30. Torrance B, McGuire KA, Lewanczuk R, et al. Overweight, physical activity and high blood pressure in children: a review of the literature. Vasc Health Risk Manag. 2007;3:139-149.
31. DASH eating plan. National Heart, Lung, and Blood Institute. Accessed April 26, 2021. www.nhlbi.nih.gov/health-topics/dash-eating-plan
32. Nutritional goals for age-sex groups based on dietary reference intakes and dietary guidelines recommendations (Appendix 7). In: US Department of Agriculture. Dietary guidelines for Americans, 2015-2020. 8th ed. December 2015;97-98. Accessed April 26, 2021. https://health.gov/sites/default/files/2019-09/2015-2020_Dietary_Guidelines.pdf
33. Asghari G, Yuzbashian E, Mirmiran P, et al. Dietary Approaches to Stop Hypertension (DASH) dietary pattern is associated with reduced incidence of metabolic syndrome in children and adolescents. J Pediatr. 2016;174:178-184.e1. doi: 10.1016/j.jpeds.2016.03.077
34. Damasceno MMC, de Araújo MFM, de Freitas RWJF, et al. The association between blood pressure in adolescents and the consumption of fruits, vegetables and fruit juice–an exploratory study. J Clin Nurs. 2011;20:1553-1560. doi: 10.1111/j.1365-2702.2010.03608.x
35. Anderson KL. A review of the prevention and medical management of childhood obesity. Child Adolesc Psychiatr Clin N Am. 2018;27:63-76. doi: 10.1016/j.chc.2017.08.003
36. Kumar S, King EC, Christison, et al; POWER Work Group. Health outcomes of youth in clinical pediatric weight management programs in POWER. J Pediatr. 2019;208:57-65.e4. doi: 10.1016/j.jpeds.2018.12.049
37. Gregoski MJ, Barnes VA, Tingen MS, et al. Breathing awareness meditation and LifeSkills® Training programs influence upon ambulatory blood pressure and sodium excretion among African American adolescents. J Adolesc Health. 2011;48:59-64. doi: 10.1016/j.jadohealth.2010.05.019
38. Escape Trial Group; E, Trivelli A, Picca S, et al. Strict blood-pressure control and progression of renal failure in children. N Engl J Med. 2009;361:1639-1650. doi: 10.1056/NEJMoa0902066
1. Theodore RF, Broadbent J, Nagin D, et al. Childhood to early-midlife systolic blood pressure trajectories: early-life predictors, effect modifiers, and adult cardiovascular outcomes. Hypertension. 2015;66:1108-1115. doi: 10.1161/HYPERTENSIONAHA.115.05831
2. Lurbe E, Agabiti-Rosei E, Cruickshank JK, et al. 2016 European Society of Hypertension guidelines for the management of high blood pressure in children and adolescents. J Hypertens. 2016;34:1887-1920. doi: 10.1097/HJH.0000000000001039
3. Weaver DJ, Mitsnefes MM. Effects of systemic hypertension on the cardiovascular system. Prog Pediatr Cardiol. 2016;41:59-65. https://doi.org/10.1016/j.ppedcard.2015.11.005
4. Ippisch HM, Daniels SR. Hypertension in overweight and obese children. Prog Pediatr Cardiol. 2008;25:177-182. doi: org/10.1016/j.ppedcard.2008.05.002
5. Urbina EM, Lande MB, Hooper SR, et al. Target organ abnormalities in pediatric hypertension. J Pediatr. 2018;202:14-22. doi: 10.1016/j.jpeds.2018.07.026
6. Flynn JT, Kaelber DC, Baker-Smith CM, et al; . Clinical practice guideline for screening and management of high blood pressure in children and adolescents. Pediatrics. 2017;140:e20171904. doi: 10.1542/peds.2017-1904
7. Khoury M, Khoury PR, Dolan LM, et al. Clinical implications of the revised AAP pediatric hypertension guidelines. Pediatrics. 2018;142:e20180245. doi: 10.1542/peds.2018-0245
8. Falkner B, Gidding SS, Ramirez-Garnica G, et al. The relationship of body mass index and blood pressure in primary care pediatric patients. J Pediatr. 2006;148:195-200. doi: 10.1016/j.jpeds.2005.10.030
9. Rodriguez BL, Dabelea D, Liese AD, et al; SEARCH Study Group. Prevalence and correlates of elevated blood pressure in youth with diabetes mellitus: the SEARCH for diabetes in youth study. J Pediatr. 2010;157:245-251.e1. doi: 10.1016/j.jpeds.2010.02.021
10. Shay CM, Ning H, Daniels SR, et al. Status of cardiovascular health in US adolescents: prevalence estimates from the National Health and Nutrition Examination Surveys (NHANES) 2005-2010. Circulation. 2013;127:1369-1376. doi: 10.1161/CIRCULATIONAHA.113.001559
11. Archbold KH, Vasquez MM, Goodwin JL, et al. Effects of sleep patterns and obesity on increases in blood pressure in a 5-year period: report from the Tucson Children’s Assessment of Sleep Apnea Study. J Pediatr. 2012;161:26-30. doi: 10.1016/j.jpeds.2011.12.034
12. Flynn JT, Mitsnefes M, Pierce C, et al; . Blood pressure in children with chronic kidney disease: a report from the Chronic Kidney Disease in Children study. Hypertension. 2008;52:631-637. doi: 10.1161/HYPERTENSIONAHA.108.110635
13. Martin RM, Ness AR, Gunnell D, et al; ALSPAC Study Team. Does breast-feeding in infancy lower blood pressure in childhood? The Avon Longitudinal Study of Parents and Children (ALSPAC). Circulation. 2004;109:1259-1266. doi: 10.1161/01.CIR.0000118468.76447.CE
14. Brickner ME, Hillis LD, Lange RA. Congenital heart disease in adults. N Engl J Med. 2000;342:256-263. doi: 10.1056/NEJM200001273420407
15. Chen X, Wang Y. Tracking of blood pressure from childhood to adulthood: a systematic review and meta-regression analysis. Circulation. 2008;117:3171-3180. doi: 10.1161/CIRCULATIONAHA.107.730366
16. Sun SS, Grave GD, Siervogel RM, et al. Systolic blood pressure in childhood predicts hypertension and metabolic syndrome later in life. Pediatrics. 2007;119:237-246. doi: 10.1542/peds.2006-2543
17. Parker ED, Sinaiko AR, Kharbanda EO, et al. Change in weight status and development of hypertension. Pediatrics. 2016; 137:e20151662. doi: 10.1542/peds.2015-1662
18. Pickering TG, Hall JE, Appel LJ, et al; . Recommendations for blood pressure measurement in humans and experimental animals: Part 1: blood pressure measurement in humans: a statement for professionals from the Subcommittee of Professional and Public Education of the American Heart Association Council on High Blood Pressure Research. Hypertension. 2005;45:142-161. doi: 10.1161/01.HYP.0000150859.47929.8e
19. Staley JR, Bradley J, Silverwood RJ, et al. Associations of blood pressure in pregnancy with offspring blood pressure trajectories during childhood and adolescence: findings from a prospective study. J Am Heart Assoc. 2015;4:e001422. doi: 10.1161/JAHA.114.001422
20. Yang Q, Zhang Z, Zuklina EV, et al. Sodium intake and blood pressure among US children and adolescents. Pediatrics. 2012;130:611-619. doi: 10.1542/peds.2011-3870
21. Le-Ha C, Beilin LJ, Burrows S, et al. Oral contraceptive use in girls and alcohol consumption in boys are associated with increased blood pressure in late adolescence. Eur J Prev Cardiol. 2013;20:947-955. doi: 10.1177/2047487312452966
22. Samuels JA, Franco K, Wan F, Sorof JM. Effect of stimulants on 24-h ambulatory blood pressure in children with ADHD: a double-blind, randomized, cross-over trial. Pediatr Nephrol. 2006;21:92-95. doi: 10.1007/s00467-005-2051-1
23. Wiesen J, Adkins M, Fortune S, et al. Evaluation of pediatric patients with mild-to-moderate hypertension: yield of diagnostic testing. Pediatrics. 2008;122:e988-993. doi: 10.1542/peds.2008-0365
24. Kapur G, Ahmed M, Pan C, et al. Secondary hypertension in overweight and stage 1 hypertensive children: a Midwest Pediatric Nephrology Consortium report. J Clin Hypertens (Greenwich). 2010;12:34-39. doi: 10.1111/j.1751-7176.2009.00195.x
25. Anyaegbu EI, Dharnidharka VR. Hypertension in the teenager. Pediatr Clin North Am. 2014;61:131-151. doi: 10.1016/j.pcl.2013.09.011
26. Gandhi B, Cheek S, Campo JV. Anxiety in the pediatric medical setting. Child Adolesc Psychiatr Clin N Am. 2012;21:643-653. doi: 10.1016/j.chc.2012.05.013
27. Farpour-Lambert NJ, Aggoun Y, Marchand LM, et al. Physical activity reduces systemic blood pressure and improves early markers of atherosclerosis in pre-pubertal obese children. J Am Coll Cardiol. 2009;54:2396-2406. doi: 10.1016/j.jacc.2009.08.030
28. Li JS, Baker-Smith CM, Smith PB, et al. Racial differences in blood pressure response to angiotensin-converting enzyme inhibitors in children: a meta-analysis. Clin Pharmacol Ther. 2008;84:315-319. doi: 10.1038/clpt.2008.113
29. Singer PS. Updates on hypertension and new guidelines. Adv Pediatr. 2019;66:177-187. doi: 10.1016/j.yapd.2019.03.009
30. Torrance B, McGuire KA, Lewanczuk R, et al. Overweight, physical activity and high blood pressure in children: a review of the literature. Vasc Health Risk Manag. 2007;3:139-149.
31. DASH eating plan. National Heart, Lung, and Blood Institute. Accessed April 26, 2021. www.nhlbi.nih.gov/health-topics/dash-eating-plan
32. Nutritional goals for age-sex groups based on dietary reference intakes and dietary guidelines recommendations (Appendix 7). In: US Department of Agriculture. Dietary guidelines for Americans, 2015-2020. 8th ed. December 2015;97-98. Accessed April 26, 2021. https://health.gov/sites/default/files/2019-09/2015-2020_Dietary_Guidelines.pdf
33. Asghari G, Yuzbashian E, Mirmiran P, et al. Dietary Approaches to Stop Hypertension (DASH) dietary pattern is associated with reduced incidence of metabolic syndrome in children and adolescents. J Pediatr. 2016;174:178-184.e1. doi: 10.1016/j.jpeds.2016.03.077
34. Damasceno MMC, de Araújo MFM, de Freitas RWJF, et al. The association between blood pressure in adolescents and the consumption of fruits, vegetables and fruit juice–an exploratory study. J Clin Nurs. 2011;20:1553-1560. doi: 10.1111/j.1365-2702.2010.03608.x
35. Anderson KL. A review of the prevention and medical management of childhood obesity. Child Adolesc Psychiatr Clin N Am. 2018;27:63-76. doi: 10.1016/j.chc.2017.08.003
36. Kumar S, King EC, Christison, et al; POWER Work Group. Health outcomes of youth in clinical pediatric weight management programs in POWER. J Pediatr. 2019;208:57-65.e4. doi: 10.1016/j.jpeds.2018.12.049
37. Gregoski MJ, Barnes VA, Tingen MS, et al. Breathing awareness meditation and LifeSkills® Training programs influence upon ambulatory blood pressure and sodium excretion among African American adolescents. J Adolesc Health. 2011;48:59-64. doi: 10.1016/j.jadohealth.2010.05.019
38. Escape Trial Group; E, Trivelli A, Picca S, et al. Strict blood-pressure control and progression of renal failure in children. N Engl J Med. 2009;361:1639-1650. doi: 10.1056/NEJMoa0902066
PRACTICE RECOMMENDATIONS
› Measure the blood pressure (BP) of all children 3 years and older annually; those who have a specific comorbid condition (eg, obesity, diabetes, renal disease, or an aortic-arch abnormality) or who are taking medication known to elevate BP should have their BP checked at every health care visit. C
› Encourage lifestyle modification as the initial treatment for elevated BP or hypertension in children. A
› Utilize pharmacotherapy for (1) children with stage 1 hypertension who have failed to meet BP goals after 3 to 6 months of lifestyle modification and (2) children with stage 2 hypertension who do not have a modifiable risk factor, such as obesity. C
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
Bariatric surgery’s cardiovascular benefit extends to 7 years
Patients with obesity who had bariatric surgery had a lower risk of having a major adverse cardiovascular event (MACE) or dying from all causes during a median 7-year follow-up, compared with similar patients who did not undergo surgery.
These findings, from a province-wide retrospective cohort study from Quebec, follow two recent, slightly shorter similar trials.
Now we need a large randomized clinical trial (RCT), experts say, to definitively establish cardiovascular and mortality benefits in people with obesity who have metabolic/bariatric surgery. And such a trial is just beginning.
Philippe Bouchard, MD, a general surgery resident from McGill University in Montreal presented the Quebec study in a top papers session at the annual meeting of the American Society for Metabolic & Bariatric Surgery.
The findings showed that, among obese patients with metabolic syndrome, bariatric/metabolic surgery is associated with a sustained decrease in the incidence of MACE and all-cause mortality of at least 5 years, Dr. Bouchard said.
“The results of this population-based observational study should be validated in randomized controlled trials,” he concluded.
In the meantime, “we believe our study adds to the body of evidence in mainly two ways,” Dr. Bouchard told this news organization in an email.
It has a longer follow-up than recent observational studies, “a median of 7 years, compared to 3.9 years in a study from the Cleveland Clinic, and 4.6 years in one from Ontario, he said.
“This allows us to [estimate] an absolute risk reduction of MACE of 5.11% at 10 years,” he added. This is a smaller risk reduction than the roughly 40% risk reduction seen in the other two studies, possibly because of selection bias, Dr. Bouchard speculated.
“Second, most of the larger cohorts are heavily weighted on Roux-en-Y gastric bypass,” he continued. In contrast, their study included diverse procedures, including sleeve gastrectomy, duodenal switch, and adjustable gastric banding.
“Given the rise in popularity of a derivative of the duodenal switch – the single-anastomosis duodenal-ileal bypass with sleeve gastrectomy (SADi-S) – we believe this information is timely and relevant to clinicians,” Dr. Bouchard said.
RCT on the subject is coming
“I totally agree that we need a large randomized controlled trial of bariatric surgery versus optimal medical therapy to conclusively establish” the impact of bariatric surgery on cardiovascular outcomes, said the assigned discussant, Mehran Anvari, MD. And their research group is just about to begin one.
In the absence of RCT data, clinicians “may currently not refer [eligible] patients for bariatric surgery because of the high risk they pose,” said Dr. Anvari, professor and director of the Centre for Minimal Access Surgery of McMaster University, Hamilton, Ont., and senior author in the Ontario study.
Furthermore, an important point is that the current trial extended the follow-up to 7 years, he told this news organization in an email.
That study included patients with diabetes and hypertension, he added, whereas his group included patients with a history of cardiovascular disease and/or heart failure.
“We hope these studies encourage general practitioners and cardiologists to consider bariatric surgery as a viable treatment option to prevent and reduce the risk of MACE in the obese patients [body mass index >35 kg/m2] with significant cardiovascular disease,” he said.
“We have embarked on a pilot RCT among bariatric centers of excellence in Ontario,” Dr. Anvari added, which showed the feasibility and safety of such a study.
He estimates that the RCT will need to recruit 2,000 patients to demonstrate the safety and effectiveness of bariatric surgery in reducing MACE and cardiac and all-cause mortality among patients with existing cardiovascular disease.
This “will require international collaboration,” he added, “and our group is currently establishing collaboration with sites in North America, Europe, and Australia to conduct such a study.”
Patients matched for age, sex, number of comorbidities
Quebec has a single public health care system that covers the cost of bariatric surgery for eligible patients; that is, those with a BMI greater than 35 kg/m2 and comorbidities or a BMI greater than 40 kg/m2.
Using this provincial health care database, which covers over 97% of the population, the researchers identified 3,637 patients with diabetes and/or hypertension who had bariatric surgery during 2007-2012.
They matched the surgery patients with 5,420 control patients with obesity who lived in the same geographic region and had a similar age, sex, and number of Charlson Comorbidity Index comorbidities, but did not undergo bariatric surgery.
The patients had a mean age of 50 and 64% were women.
Half had zero to one comorbidities, a quarter had two comorbidities, and another quarter had at least three comorbidities.
Most patients in the surgery group had type 2 diabetes (70%) and 50% had hypertension, whereas in the control group, most patients had hypertension (82%) and 41% had diabetes.
The most common type of bariatric surgery was adjustable gastric banding (42% of patients), followed by duodenal switch (24%), sleeve gastrectomy (23%), and Roux-en-Y gastric bypass (11%).
The primary outcome was the incidence of MACE, defined as coronary artery events (including myocardial infarction, percutaneous coronary intervention, and coronary artery bypass graft), stroke, heart failure, and all-cause mortality,
After a median follow-up of 7-11 years, fewer patients in the surgical group than in the control group had MACE (20% vs. 25%) or died from all causes (4.1% vs. 6.3%, both statistically significant at P < .01)
Similarly, significantly fewer patients in the surgical group than in the control group had a coronary artery event or heart failure (each P < .01).
However, there were no significant between-group difference in the rate of stroke, possibly because of the small number of strokes.
The risk of MACE was 17% lower in the group that had bariatric surgery than in the control group (adjusted hazard ratio, 0.83; 95% confidence interval, 0.78-0.89), after adjusting for age, sex, and number of comorbidities.
In subgroup analysis, patients who had adjustable gastric banding, Roux-en-Y gastric bypass, or duodenal switch had a significantly lower risk of MACE than control patients.
The risk of MACE was similar in patients who had sleeve gastrectomy and in control patients.
However, these subgroup results need to be interpreted with caution since the surgery and control patients in each surgery type subgroup were not matched for age, sex, and comorbidities, said Dr. Bouchard.
He acknowledged that study limitations include a lack of information about the patients’ BMI, weight, medications, and glycemic control (hemoglobin A1c).
Dr. Bouchard and Dr. Anvari have no relevant financial disclosures.
Patients with obesity who had bariatric surgery had a lower risk of having a major adverse cardiovascular event (MACE) or dying from all causes during a median 7-year follow-up, compared with similar patients who did not undergo surgery.
These findings, from a province-wide retrospective cohort study from Quebec, follow two recent, slightly shorter similar trials.
Now we need a large randomized clinical trial (RCT), experts say, to definitively establish cardiovascular and mortality benefits in people with obesity who have metabolic/bariatric surgery. And such a trial is just beginning.
Philippe Bouchard, MD, a general surgery resident from McGill University in Montreal presented the Quebec study in a top papers session at the annual meeting of the American Society for Metabolic & Bariatric Surgery.
The findings showed that, among obese patients with metabolic syndrome, bariatric/metabolic surgery is associated with a sustained decrease in the incidence of MACE and all-cause mortality of at least 5 years, Dr. Bouchard said.
“The results of this population-based observational study should be validated in randomized controlled trials,” he concluded.
In the meantime, “we believe our study adds to the body of evidence in mainly two ways,” Dr. Bouchard told this news organization in an email.
It has a longer follow-up than recent observational studies, “a median of 7 years, compared to 3.9 years in a study from the Cleveland Clinic, and 4.6 years in one from Ontario, he said.
“This allows us to [estimate] an absolute risk reduction of MACE of 5.11% at 10 years,” he added. This is a smaller risk reduction than the roughly 40% risk reduction seen in the other two studies, possibly because of selection bias, Dr. Bouchard speculated.
“Second, most of the larger cohorts are heavily weighted on Roux-en-Y gastric bypass,” he continued. In contrast, their study included diverse procedures, including sleeve gastrectomy, duodenal switch, and adjustable gastric banding.
“Given the rise in popularity of a derivative of the duodenal switch – the single-anastomosis duodenal-ileal bypass with sleeve gastrectomy (SADi-S) – we believe this information is timely and relevant to clinicians,” Dr. Bouchard said.
RCT on the subject is coming
“I totally agree that we need a large randomized controlled trial of bariatric surgery versus optimal medical therapy to conclusively establish” the impact of bariatric surgery on cardiovascular outcomes, said the assigned discussant, Mehran Anvari, MD. And their research group is just about to begin one.
In the absence of RCT data, clinicians “may currently not refer [eligible] patients for bariatric surgery because of the high risk they pose,” said Dr. Anvari, professor and director of the Centre for Minimal Access Surgery of McMaster University, Hamilton, Ont., and senior author in the Ontario study.
Furthermore, an important point is that the current trial extended the follow-up to 7 years, he told this news organization in an email.
That study included patients with diabetes and hypertension, he added, whereas his group included patients with a history of cardiovascular disease and/or heart failure.
“We hope these studies encourage general practitioners and cardiologists to consider bariatric surgery as a viable treatment option to prevent and reduce the risk of MACE in the obese patients [body mass index >35 kg/m2] with significant cardiovascular disease,” he said.
“We have embarked on a pilot RCT among bariatric centers of excellence in Ontario,” Dr. Anvari added, which showed the feasibility and safety of such a study.
He estimates that the RCT will need to recruit 2,000 patients to demonstrate the safety and effectiveness of bariatric surgery in reducing MACE and cardiac and all-cause mortality among patients with existing cardiovascular disease.
This “will require international collaboration,” he added, “and our group is currently establishing collaboration with sites in North America, Europe, and Australia to conduct such a study.”
Patients matched for age, sex, number of comorbidities
Quebec has a single public health care system that covers the cost of bariatric surgery for eligible patients; that is, those with a BMI greater than 35 kg/m2 and comorbidities or a BMI greater than 40 kg/m2.
Using this provincial health care database, which covers over 97% of the population, the researchers identified 3,637 patients with diabetes and/or hypertension who had bariatric surgery during 2007-2012.
They matched the surgery patients with 5,420 control patients with obesity who lived in the same geographic region and had a similar age, sex, and number of Charlson Comorbidity Index comorbidities, but did not undergo bariatric surgery.
The patients had a mean age of 50 and 64% were women.
Half had zero to one comorbidities, a quarter had two comorbidities, and another quarter had at least three comorbidities.
Most patients in the surgery group had type 2 diabetes (70%) and 50% had hypertension, whereas in the control group, most patients had hypertension (82%) and 41% had diabetes.
The most common type of bariatric surgery was adjustable gastric banding (42% of patients), followed by duodenal switch (24%), sleeve gastrectomy (23%), and Roux-en-Y gastric bypass (11%).
The primary outcome was the incidence of MACE, defined as coronary artery events (including myocardial infarction, percutaneous coronary intervention, and coronary artery bypass graft), stroke, heart failure, and all-cause mortality,
After a median follow-up of 7-11 years, fewer patients in the surgical group than in the control group had MACE (20% vs. 25%) or died from all causes (4.1% vs. 6.3%, both statistically significant at P < .01)
Similarly, significantly fewer patients in the surgical group than in the control group had a coronary artery event or heart failure (each P < .01).
However, there were no significant between-group difference in the rate of stroke, possibly because of the small number of strokes.
The risk of MACE was 17% lower in the group that had bariatric surgery than in the control group (adjusted hazard ratio, 0.83; 95% confidence interval, 0.78-0.89), after adjusting for age, sex, and number of comorbidities.
In subgroup analysis, patients who had adjustable gastric banding, Roux-en-Y gastric bypass, or duodenal switch had a significantly lower risk of MACE than control patients.
The risk of MACE was similar in patients who had sleeve gastrectomy and in control patients.
However, these subgroup results need to be interpreted with caution since the surgery and control patients in each surgery type subgroup were not matched for age, sex, and comorbidities, said Dr. Bouchard.
He acknowledged that study limitations include a lack of information about the patients’ BMI, weight, medications, and glycemic control (hemoglobin A1c).
Dr. Bouchard and Dr. Anvari have no relevant financial disclosures.
Patients with obesity who had bariatric surgery had a lower risk of having a major adverse cardiovascular event (MACE) or dying from all causes during a median 7-year follow-up, compared with similar patients who did not undergo surgery.
These findings, from a province-wide retrospective cohort study from Quebec, follow two recent, slightly shorter similar trials.
Now we need a large randomized clinical trial (RCT), experts say, to definitively establish cardiovascular and mortality benefits in people with obesity who have metabolic/bariatric surgery. And such a trial is just beginning.
Philippe Bouchard, MD, a general surgery resident from McGill University in Montreal presented the Quebec study in a top papers session at the annual meeting of the American Society for Metabolic & Bariatric Surgery.
The findings showed that, among obese patients with metabolic syndrome, bariatric/metabolic surgery is associated with a sustained decrease in the incidence of MACE and all-cause mortality of at least 5 years, Dr. Bouchard said.
“The results of this population-based observational study should be validated in randomized controlled trials,” he concluded.
In the meantime, “we believe our study adds to the body of evidence in mainly two ways,” Dr. Bouchard told this news organization in an email.
It has a longer follow-up than recent observational studies, “a median of 7 years, compared to 3.9 years in a study from the Cleveland Clinic, and 4.6 years in one from Ontario, he said.
“This allows us to [estimate] an absolute risk reduction of MACE of 5.11% at 10 years,” he added. This is a smaller risk reduction than the roughly 40% risk reduction seen in the other two studies, possibly because of selection bias, Dr. Bouchard speculated.
“Second, most of the larger cohorts are heavily weighted on Roux-en-Y gastric bypass,” he continued. In contrast, their study included diverse procedures, including sleeve gastrectomy, duodenal switch, and adjustable gastric banding.
“Given the rise in popularity of a derivative of the duodenal switch – the single-anastomosis duodenal-ileal bypass with sleeve gastrectomy (SADi-S) – we believe this information is timely and relevant to clinicians,” Dr. Bouchard said.
RCT on the subject is coming
“I totally agree that we need a large randomized controlled trial of bariatric surgery versus optimal medical therapy to conclusively establish” the impact of bariatric surgery on cardiovascular outcomes, said the assigned discussant, Mehran Anvari, MD. And their research group is just about to begin one.
In the absence of RCT data, clinicians “may currently not refer [eligible] patients for bariatric surgery because of the high risk they pose,” said Dr. Anvari, professor and director of the Centre for Minimal Access Surgery of McMaster University, Hamilton, Ont., and senior author in the Ontario study.
Furthermore, an important point is that the current trial extended the follow-up to 7 years, he told this news organization in an email.
That study included patients with diabetes and hypertension, he added, whereas his group included patients with a history of cardiovascular disease and/or heart failure.
“We hope these studies encourage general practitioners and cardiologists to consider bariatric surgery as a viable treatment option to prevent and reduce the risk of MACE in the obese patients [body mass index >35 kg/m2] with significant cardiovascular disease,” he said.
“We have embarked on a pilot RCT among bariatric centers of excellence in Ontario,” Dr. Anvari added, which showed the feasibility and safety of such a study.
He estimates that the RCT will need to recruit 2,000 patients to demonstrate the safety and effectiveness of bariatric surgery in reducing MACE and cardiac and all-cause mortality among patients with existing cardiovascular disease.
This “will require international collaboration,” he added, “and our group is currently establishing collaboration with sites in North America, Europe, and Australia to conduct such a study.”
Patients matched for age, sex, number of comorbidities
Quebec has a single public health care system that covers the cost of bariatric surgery for eligible patients; that is, those with a BMI greater than 35 kg/m2 and comorbidities or a BMI greater than 40 kg/m2.
Using this provincial health care database, which covers over 97% of the population, the researchers identified 3,637 patients with diabetes and/or hypertension who had bariatric surgery during 2007-2012.
They matched the surgery patients with 5,420 control patients with obesity who lived in the same geographic region and had a similar age, sex, and number of Charlson Comorbidity Index comorbidities, but did not undergo bariatric surgery.
The patients had a mean age of 50 and 64% were women.
Half had zero to one comorbidities, a quarter had two comorbidities, and another quarter had at least three comorbidities.
Most patients in the surgery group had type 2 diabetes (70%) and 50% had hypertension, whereas in the control group, most patients had hypertension (82%) and 41% had diabetes.
The most common type of bariatric surgery was adjustable gastric banding (42% of patients), followed by duodenal switch (24%), sleeve gastrectomy (23%), and Roux-en-Y gastric bypass (11%).
The primary outcome was the incidence of MACE, defined as coronary artery events (including myocardial infarction, percutaneous coronary intervention, and coronary artery bypass graft), stroke, heart failure, and all-cause mortality,
After a median follow-up of 7-11 years, fewer patients in the surgical group than in the control group had MACE (20% vs. 25%) or died from all causes (4.1% vs. 6.3%, both statistically significant at P < .01)
Similarly, significantly fewer patients in the surgical group than in the control group had a coronary artery event or heart failure (each P < .01).
However, there were no significant between-group difference in the rate of stroke, possibly because of the small number of strokes.
The risk of MACE was 17% lower in the group that had bariatric surgery than in the control group (adjusted hazard ratio, 0.83; 95% confidence interval, 0.78-0.89), after adjusting for age, sex, and number of comorbidities.
In subgroup analysis, patients who had adjustable gastric banding, Roux-en-Y gastric bypass, or duodenal switch had a significantly lower risk of MACE than control patients.
The risk of MACE was similar in patients who had sleeve gastrectomy and in control patients.
However, these subgroup results need to be interpreted with caution since the surgery and control patients in each surgery type subgroup were not matched for age, sex, and comorbidities, said Dr. Bouchard.
He acknowledged that study limitations include a lack of information about the patients’ BMI, weight, medications, and glycemic control (hemoglobin A1c).
Dr. Bouchard and Dr. Anvari have no relevant financial disclosures.
FROM ASMBS 2021
Healthy with obesity? The latest study casts doubt
compared with people without obesity and or adverse metabolic profiles, new research suggests.
The latest data on this controversial subject come from an analysis of nearly 400,000 people in the U.K. Biobank. Although the data also showed that metabolically healthy obesity poses less risk than “metabolically unhealthy” obesity, the risk of progression from healthy to unhealthy within 3-5 years was high.
“People with metabolically healthy obesity are not ‘healthy’ as they are at higher risk of atherosclerotic cardiovascular disease [ASCVD], heart failure, and respiratory diseases, compared with nonobese people with a normal metabolic profile. As such, weight management could be beneficial to all people with obesity irrespective of metabolic profile,” Ziyi Zhou and colleagues wrote in their report, published June 10, 2021, in Diabetologia.
Moreover, they advised avoiding the term metabolically healthy obesity entirely in clinical medicine “as it is misleading, and different strategies for risk stratification should be explored.”
In interviews, two experts provided somewhat different takes on the study and the overall subject.
‘Lifestyle should be explored with every single patient regardless of their weight’
Yoni Freedhoff, MD, medical director of the Bariatric Medical Institute, Ottawa, said “clinicians and patients need to be aware that obesity increases a person’s risk of various medical problems, and in turn this might lead to more frequent screening. This increased screening might be analogous to that of a person with a strong familial history of cancer who of course we would never describe as being ‘unhealthy’ as a consequence of their increased risk.”
In addition to screening, “lifestyle should be explored with every single patient regardless of their weight, and if a person’s weight is not affecting their health or their quality of life, a clinician need only let the patient know that, were they to want to discuss weight management options in the future, that they’d be there for them,” said Dr. Freedhoff.
‘Metabolically healthy obesity’ has had many definitions
Matthias Schulze, DrPH, head of the molecular epidemiology at the German Institute of Human Nutrition, Potsdam, and professor at the University of Potsdam, pointed out that the way metabolically healthy obesity is defined and the outcomes assessed make a difference.
In the current study, the term is defined as having a body mass index of at least 30 kg/m2 and at least four of six metabolically healthy criteria: blood pressure, C-reactive protein, triacylglycerols, LDL cholesterol, HDL cholesterol, and hemoglobin A1c.
In May 2021, Dr. Schulze and associates reported in JAMA Network Open on a different definition that they found to identify individuals who do not have an increased risk of cardiovascular disease death and total mortality. Interestingly, they also used the U.K. Biobank as their validation cohort.
“We derived a new definition of metabolic health ... that is different from those used in [the current] article. Importantly, we included a measure of body fat distribution, waist-to-hip ratio. On the other side, we investigated only mortality outcomes and we can therefore not exclude the possibility that other outcomes may still be related. [For example], a higher diabetes risk may still be present among those we have defined as having metabolically healthy obesity.”
Dr. Schulze also said that several previous studies and meta-analyses have suggested that “previous common definitions of metabolically healthy obesity do not identify a subgroup without risk, or being at risk comparable to normal-weight metabolically healthy. Thus, this study confirms this conclusion. [But] this doesn’t rule out that there are better ways of defining subgroups.”
Clinically, he said “given that we investigated only mortality, we cannot conclude that our ‘metabolically healthy obesity’ group doesn’t require intervention.”
Higher rates of diabetes, ASCVD, heart failure, death
The current population-based study included 381,363 U.K. Biobank participants who were followed up for a median 11.2 years. Overall, about 55% did not have obesity or metabolic abnormalities, 9% had metabolically healthy obesity, 20% were metabolically unhealthy but did not have obesity, and 16% had metabolically unhealthy obesity as defined by the investigators.
The investigators adjusted the data for several potential confounders, including age, sex, ethnicity, education, socioeconomic status, smoking status, physical activity, and dietary factors.
Compared with individuals without obesity or metabolic abnormalities, those with metabolically healthy obesity had significantly higher rates of incident diabetes (hazard ratio, 4.32), ASCVD (HR, 1.18), myocardial infarction (HR, 1.23), stroke (HR, 1.10), heart failure (HR, 1.76), respiratory diseases (HR, 1.28), and chronic obstructive pulmonary disease (HR, 1.19).
In general, rates of cardiovascular and respiratory outcomes were highest in metabolically unhealthy obesity, followed by those without obesity but with metabolic abnormalities and those with metabolically healthy obesity. However, for incident and fatal heart failure and incident respiratory diseases, those with metabolically healthy obesity had higher rates than did those without obesity but with metabolic abnormalities.
Compared with those without obesity or metabolic abnormalities, those with metabolically healthy obesity had significantly higher all-cause mortality rates (HR, 1.22). And, compared with those without obesity (regardless of metabolic status) at baseline, those with metabolically healthy obesity were significantly more likely to have diabetes (HR, 2.06), heart failure (HR, 1.6), and respiratory diseases (HR, 1.2), but not ASCVD. The association was also significant for all-cause and heart failure mortality (HR, 1.12 and 1.44, respectively), but not for other causes of death.
Progression from metabolically healthy to unhealthy is common
Among 8,512 participants for whom longitudinal data were available for a median of 4.4 years, half of those with metabolically healthy obesity remained in that category, 20% no longer had obesity, and more than a quarter transitioned to metabolically unhealthy obesity. Compared with those without obesity or metabolic abnormalities throughout, those who transitioned from metabolically healthy to metabolically unhealthy had significantly higher rates of incident ASCVD (HR, 2.46) and all-cause mortality (HR, 3.07).
But those who remained in the metabolically healthy obesity category throughout did not have significantly increased risks for the adverse outcomes measured.
Ms. Zhou and colleagues noted that the data demonstrate heterogeneity among people with obesity, which offers the potential to stratify risk based on prognosis. For example, “people with [metabolically unhealthy obesity] were at a higher risk of mortality and morbidity than everyone else, and thus they should be prioritized for intervention.”
However, they add, “Obesity is associated with a wide range of diseases, and using a single label or categorical risk algorithm is unlikely to be effective compared with prediction algorithms based on disease-specific and continuous risk markers.”
Ms. Zhou has no disclosures. One coauthor has relationships with numerous pharmaceutical companies; the rest have none. Dr. Freedhoff has served as a director, officer, partner, employee, adviser, consultant, or trustee for the Bariatric Medical Institute and Constant Health. He is a speaker or a member of a speakers bureau for Obesity Canada and Novo Nordisk, received research grant from Novo Nordisk, and received income of at least $250 from WebMD, CTV, and Random House. Dr/ Schulze has received grants from German Federal Ministry of Education and Research.
compared with people without obesity and or adverse metabolic profiles, new research suggests.
The latest data on this controversial subject come from an analysis of nearly 400,000 people in the U.K. Biobank. Although the data also showed that metabolically healthy obesity poses less risk than “metabolically unhealthy” obesity, the risk of progression from healthy to unhealthy within 3-5 years was high.
“People with metabolically healthy obesity are not ‘healthy’ as they are at higher risk of atherosclerotic cardiovascular disease [ASCVD], heart failure, and respiratory diseases, compared with nonobese people with a normal metabolic profile. As such, weight management could be beneficial to all people with obesity irrespective of metabolic profile,” Ziyi Zhou and colleagues wrote in their report, published June 10, 2021, in Diabetologia.
Moreover, they advised avoiding the term metabolically healthy obesity entirely in clinical medicine “as it is misleading, and different strategies for risk stratification should be explored.”
In interviews, two experts provided somewhat different takes on the study and the overall subject.
‘Lifestyle should be explored with every single patient regardless of their weight’
Yoni Freedhoff, MD, medical director of the Bariatric Medical Institute, Ottawa, said “clinicians and patients need to be aware that obesity increases a person’s risk of various medical problems, and in turn this might lead to more frequent screening. This increased screening might be analogous to that of a person with a strong familial history of cancer who of course we would never describe as being ‘unhealthy’ as a consequence of their increased risk.”
In addition to screening, “lifestyle should be explored with every single patient regardless of their weight, and if a person’s weight is not affecting their health or their quality of life, a clinician need only let the patient know that, were they to want to discuss weight management options in the future, that they’d be there for them,” said Dr. Freedhoff.
‘Metabolically healthy obesity’ has had many definitions
Matthias Schulze, DrPH, head of the molecular epidemiology at the German Institute of Human Nutrition, Potsdam, and professor at the University of Potsdam, pointed out that the way metabolically healthy obesity is defined and the outcomes assessed make a difference.
In the current study, the term is defined as having a body mass index of at least 30 kg/m2 and at least four of six metabolically healthy criteria: blood pressure, C-reactive protein, triacylglycerols, LDL cholesterol, HDL cholesterol, and hemoglobin A1c.
In May 2021, Dr. Schulze and associates reported in JAMA Network Open on a different definition that they found to identify individuals who do not have an increased risk of cardiovascular disease death and total mortality. Interestingly, they also used the U.K. Biobank as their validation cohort.
“We derived a new definition of metabolic health ... that is different from those used in [the current] article. Importantly, we included a measure of body fat distribution, waist-to-hip ratio. On the other side, we investigated only mortality outcomes and we can therefore not exclude the possibility that other outcomes may still be related. [For example], a higher diabetes risk may still be present among those we have defined as having metabolically healthy obesity.”
Dr. Schulze also said that several previous studies and meta-analyses have suggested that “previous common definitions of metabolically healthy obesity do not identify a subgroup without risk, or being at risk comparable to normal-weight metabolically healthy. Thus, this study confirms this conclusion. [But] this doesn’t rule out that there are better ways of defining subgroups.”
Clinically, he said “given that we investigated only mortality, we cannot conclude that our ‘metabolically healthy obesity’ group doesn’t require intervention.”
Higher rates of diabetes, ASCVD, heart failure, death
The current population-based study included 381,363 U.K. Biobank participants who were followed up for a median 11.2 years. Overall, about 55% did not have obesity or metabolic abnormalities, 9% had metabolically healthy obesity, 20% were metabolically unhealthy but did not have obesity, and 16% had metabolically unhealthy obesity as defined by the investigators.
The investigators adjusted the data for several potential confounders, including age, sex, ethnicity, education, socioeconomic status, smoking status, physical activity, and dietary factors.
Compared with individuals without obesity or metabolic abnormalities, those with metabolically healthy obesity had significantly higher rates of incident diabetes (hazard ratio, 4.32), ASCVD (HR, 1.18), myocardial infarction (HR, 1.23), stroke (HR, 1.10), heart failure (HR, 1.76), respiratory diseases (HR, 1.28), and chronic obstructive pulmonary disease (HR, 1.19).
In general, rates of cardiovascular and respiratory outcomes were highest in metabolically unhealthy obesity, followed by those without obesity but with metabolic abnormalities and those with metabolically healthy obesity. However, for incident and fatal heart failure and incident respiratory diseases, those with metabolically healthy obesity had higher rates than did those without obesity but with metabolic abnormalities.
Compared with those without obesity or metabolic abnormalities, those with metabolically healthy obesity had significantly higher all-cause mortality rates (HR, 1.22). And, compared with those without obesity (regardless of metabolic status) at baseline, those with metabolically healthy obesity were significantly more likely to have diabetes (HR, 2.06), heart failure (HR, 1.6), and respiratory diseases (HR, 1.2), but not ASCVD. The association was also significant for all-cause and heart failure mortality (HR, 1.12 and 1.44, respectively), but not for other causes of death.
Progression from metabolically healthy to unhealthy is common
Among 8,512 participants for whom longitudinal data were available for a median of 4.4 years, half of those with metabolically healthy obesity remained in that category, 20% no longer had obesity, and more than a quarter transitioned to metabolically unhealthy obesity. Compared with those without obesity or metabolic abnormalities throughout, those who transitioned from metabolically healthy to metabolically unhealthy had significantly higher rates of incident ASCVD (HR, 2.46) and all-cause mortality (HR, 3.07).
But those who remained in the metabolically healthy obesity category throughout did not have significantly increased risks for the adverse outcomes measured.
Ms. Zhou and colleagues noted that the data demonstrate heterogeneity among people with obesity, which offers the potential to stratify risk based on prognosis. For example, “people with [metabolically unhealthy obesity] were at a higher risk of mortality and morbidity than everyone else, and thus they should be prioritized for intervention.”
However, they add, “Obesity is associated with a wide range of diseases, and using a single label or categorical risk algorithm is unlikely to be effective compared with prediction algorithms based on disease-specific and continuous risk markers.”
Ms. Zhou has no disclosures. One coauthor has relationships with numerous pharmaceutical companies; the rest have none. Dr. Freedhoff has served as a director, officer, partner, employee, adviser, consultant, or trustee for the Bariatric Medical Institute and Constant Health. He is a speaker or a member of a speakers bureau for Obesity Canada and Novo Nordisk, received research grant from Novo Nordisk, and received income of at least $250 from WebMD, CTV, and Random House. Dr/ Schulze has received grants from German Federal Ministry of Education and Research.
compared with people without obesity and or adverse metabolic profiles, new research suggests.
The latest data on this controversial subject come from an analysis of nearly 400,000 people in the U.K. Biobank. Although the data also showed that metabolically healthy obesity poses less risk than “metabolically unhealthy” obesity, the risk of progression from healthy to unhealthy within 3-5 years was high.
“People with metabolically healthy obesity are not ‘healthy’ as they are at higher risk of atherosclerotic cardiovascular disease [ASCVD], heart failure, and respiratory diseases, compared with nonobese people with a normal metabolic profile. As such, weight management could be beneficial to all people with obesity irrespective of metabolic profile,” Ziyi Zhou and colleagues wrote in their report, published June 10, 2021, in Diabetologia.
Moreover, they advised avoiding the term metabolically healthy obesity entirely in clinical medicine “as it is misleading, and different strategies for risk stratification should be explored.”
In interviews, two experts provided somewhat different takes on the study and the overall subject.
‘Lifestyle should be explored with every single patient regardless of their weight’
Yoni Freedhoff, MD, medical director of the Bariatric Medical Institute, Ottawa, said “clinicians and patients need to be aware that obesity increases a person’s risk of various medical problems, and in turn this might lead to more frequent screening. This increased screening might be analogous to that of a person with a strong familial history of cancer who of course we would never describe as being ‘unhealthy’ as a consequence of their increased risk.”
In addition to screening, “lifestyle should be explored with every single patient regardless of their weight, and if a person’s weight is not affecting their health or their quality of life, a clinician need only let the patient know that, were they to want to discuss weight management options in the future, that they’d be there for them,” said Dr. Freedhoff.
‘Metabolically healthy obesity’ has had many definitions
Matthias Schulze, DrPH, head of the molecular epidemiology at the German Institute of Human Nutrition, Potsdam, and professor at the University of Potsdam, pointed out that the way metabolically healthy obesity is defined and the outcomes assessed make a difference.
In the current study, the term is defined as having a body mass index of at least 30 kg/m2 and at least four of six metabolically healthy criteria: blood pressure, C-reactive protein, triacylglycerols, LDL cholesterol, HDL cholesterol, and hemoglobin A1c.
In May 2021, Dr. Schulze and associates reported in JAMA Network Open on a different definition that they found to identify individuals who do not have an increased risk of cardiovascular disease death and total mortality. Interestingly, they also used the U.K. Biobank as their validation cohort.
“We derived a new definition of metabolic health ... that is different from those used in [the current] article. Importantly, we included a measure of body fat distribution, waist-to-hip ratio. On the other side, we investigated only mortality outcomes and we can therefore not exclude the possibility that other outcomes may still be related. [For example], a higher diabetes risk may still be present among those we have defined as having metabolically healthy obesity.”
Dr. Schulze also said that several previous studies and meta-analyses have suggested that “previous common definitions of metabolically healthy obesity do not identify a subgroup without risk, or being at risk comparable to normal-weight metabolically healthy. Thus, this study confirms this conclusion. [But] this doesn’t rule out that there are better ways of defining subgroups.”
Clinically, he said “given that we investigated only mortality, we cannot conclude that our ‘metabolically healthy obesity’ group doesn’t require intervention.”
Higher rates of diabetes, ASCVD, heart failure, death
The current population-based study included 381,363 U.K. Biobank participants who were followed up for a median 11.2 years. Overall, about 55% did not have obesity or metabolic abnormalities, 9% had metabolically healthy obesity, 20% were metabolically unhealthy but did not have obesity, and 16% had metabolically unhealthy obesity as defined by the investigators.
The investigators adjusted the data for several potential confounders, including age, sex, ethnicity, education, socioeconomic status, smoking status, physical activity, and dietary factors.
Compared with individuals without obesity or metabolic abnormalities, those with metabolically healthy obesity had significantly higher rates of incident diabetes (hazard ratio, 4.32), ASCVD (HR, 1.18), myocardial infarction (HR, 1.23), stroke (HR, 1.10), heart failure (HR, 1.76), respiratory diseases (HR, 1.28), and chronic obstructive pulmonary disease (HR, 1.19).
In general, rates of cardiovascular and respiratory outcomes were highest in metabolically unhealthy obesity, followed by those without obesity but with metabolic abnormalities and those with metabolically healthy obesity. However, for incident and fatal heart failure and incident respiratory diseases, those with metabolically healthy obesity had higher rates than did those without obesity but with metabolic abnormalities.
Compared with those without obesity or metabolic abnormalities, those with metabolically healthy obesity had significantly higher all-cause mortality rates (HR, 1.22). And, compared with those without obesity (regardless of metabolic status) at baseline, those with metabolically healthy obesity were significantly more likely to have diabetes (HR, 2.06), heart failure (HR, 1.6), and respiratory diseases (HR, 1.2), but not ASCVD. The association was also significant for all-cause and heart failure mortality (HR, 1.12 and 1.44, respectively), but not for other causes of death.
Progression from metabolically healthy to unhealthy is common
Among 8,512 participants for whom longitudinal data were available for a median of 4.4 years, half of those with metabolically healthy obesity remained in that category, 20% no longer had obesity, and more than a quarter transitioned to metabolically unhealthy obesity. Compared with those without obesity or metabolic abnormalities throughout, those who transitioned from metabolically healthy to metabolically unhealthy had significantly higher rates of incident ASCVD (HR, 2.46) and all-cause mortality (HR, 3.07).
But those who remained in the metabolically healthy obesity category throughout did not have significantly increased risks for the adverse outcomes measured.
Ms. Zhou and colleagues noted that the data demonstrate heterogeneity among people with obesity, which offers the potential to stratify risk based on prognosis. For example, “people with [metabolically unhealthy obesity] were at a higher risk of mortality and morbidity than everyone else, and thus they should be prioritized for intervention.”
However, they add, “Obesity is associated with a wide range of diseases, and using a single label or categorical risk algorithm is unlikely to be effective compared with prediction algorithms based on disease-specific and continuous risk markers.”
Ms. Zhou has no disclosures. One coauthor has relationships with numerous pharmaceutical companies; the rest have none. Dr. Freedhoff has served as a director, officer, partner, employee, adviser, consultant, or trustee for the Bariatric Medical Institute and Constant Health. He is a speaker or a member of a speakers bureau for Obesity Canada and Novo Nordisk, received research grant from Novo Nordisk, and received income of at least $250 from WebMD, CTV, and Random House. Dr/ Schulze has received grants from German Federal Ministry of Education and Research.
FROM DIABETOLOGIA
Bariatric surgery tied to fewer HFpEF hospitalizations
Patients who underwent metabolic and bariatric surgery had fewer than half the number of hospitalizations for both acute and chronic episodes of heart failure with preserved ejection fraction (HFpEF) in a retrospective analysis of more than 2 million Americans collected in a national database.
In a multivariate analysis that adjusted for several variables patients without a history of bariatric surgery had three- to fivefold more hospitalizations for acute events involving HFpEF, and more than double the rate of hospitalizations for chronic HFpEF events, David R. Funes, MD, said at the annual meeting of the American Society for Metabolic and Bariatric Surgery.
While this analysis has the limitations of being retrospective, observational, and entirely reliant on procedure codes to define medical histories and outcomes, it had the advantage of using a large database designed to represent the U.S. adult population, said Dr. Funes, a bariatric surgeon at the Cleveland Clinic in Weston, Fla.
HFpEF effects could ‘extend’ surgery’s use
The report “adds an important article to the literature where there is a true void in trying to discern the effect of bariatric surgery on HFpEF,” commented Tammy L. Kindel, MD, PhD, director of the bariatric surgery program at the Medical College of Wisconsin, Milwaukee, and designated discussant for the report. “Minimal studies [up to now] demonstrate that weight loss in any form can modify diastolic dysfunction in patients with HFpEF. Studies that investigate the impact of bariatric surgery on clinical outcomes in patients with HFpEF are probably the most important for extending use of metabolic surgery,” Dr. Kindel said.
She added that “one of the most difficult parts of studying HFpEF” is making a firm diagnosis that often involves excluding other potential causes. She also questioned Dr. Funes about his confidence that his analysis correctly identified patients only with HFpEF. Dr. Funes replied that the diagnostic codes his team used allowed for a clear distinction between patients identified with HFpEF and those with heart failure with reduced ejection fraction, but he also admitted that his study’s complete reliance on these codes introduced a limitation to the analysis.
Including patients with diastolic dysfunction as well as HFpEF
The study used data collected during 2010-2015 by the National Inpatient Sample, run by the U.S. Department of Health & Human Services in a case-control analysis that included 296,041 patients who had undergone some form of bariatric surgery and 2,004,804 people with no history of bariatric surgery selected as controls on the basis of their obesity.
The absolute numbers showed that, during the observation period, the incidence of acute HFpEF hospitalizations was 0.19% among those with prior bariatric surgery and 0.86% among those with no surgery, and the incidence of chronic heart failure hospitalizations was 0.01% among people with prior bariatric surgery and 0.05% among those without prior surgery. Dr. Funes said. He noted that, during the period studied patients, with HFpEF were usually identified as having diastolic heart failure, an older name for the same disease.
In multivariate analyses that adjusted for age, sex, race, hypertension, diabetes, smoking, and coronary artery disease, people without prior bariatric surgery and with hypertension had a 2.8-fold increased rate of acute hospitalizations for HFpEF, while those without hypertension or prior bariatric surgery had a 5.2-fold increased rate. In addition, control patients, regardless of hypertension status, had a 2.9-fold increased rate of hospitalizations for chronic HFpEF events. All these differences were statistically significant.
Dr. Funes also reported results from additional analyses that focused on a roughly 68,000-patient subgroup of those included in the study who had a history of coronary artery disease, including about 62,000 with no prior bariatric surgery and nearly 6,000 people with prior bariatric surgery. In a multivariate analysis of this subgroup, people without prior bariatric surgery had a 2.65-fold increased rate of hospitalization for a HFpEF event (either acute or chronic), compared with those who had undergone bariatric surgery.
Dr. Funes and associates and Dr. Kindel had no relevant disclosures.
Patients who underwent metabolic and bariatric surgery had fewer than half the number of hospitalizations for both acute and chronic episodes of heart failure with preserved ejection fraction (HFpEF) in a retrospective analysis of more than 2 million Americans collected in a national database.
In a multivariate analysis that adjusted for several variables patients without a history of bariatric surgery had three- to fivefold more hospitalizations for acute events involving HFpEF, and more than double the rate of hospitalizations for chronic HFpEF events, David R. Funes, MD, said at the annual meeting of the American Society for Metabolic and Bariatric Surgery.
While this analysis has the limitations of being retrospective, observational, and entirely reliant on procedure codes to define medical histories and outcomes, it had the advantage of using a large database designed to represent the U.S. adult population, said Dr. Funes, a bariatric surgeon at the Cleveland Clinic in Weston, Fla.
HFpEF effects could ‘extend’ surgery’s use
The report “adds an important article to the literature where there is a true void in trying to discern the effect of bariatric surgery on HFpEF,” commented Tammy L. Kindel, MD, PhD, director of the bariatric surgery program at the Medical College of Wisconsin, Milwaukee, and designated discussant for the report. “Minimal studies [up to now] demonstrate that weight loss in any form can modify diastolic dysfunction in patients with HFpEF. Studies that investigate the impact of bariatric surgery on clinical outcomes in patients with HFpEF are probably the most important for extending use of metabolic surgery,” Dr. Kindel said.
She added that “one of the most difficult parts of studying HFpEF” is making a firm diagnosis that often involves excluding other potential causes. She also questioned Dr. Funes about his confidence that his analysis correctly identified patients only with HFpEF. Dr. Funes replied that the diagnostic codes his team used allowed for a clear distinction between patients identified with HFpEF and those with heart failure with reduced ejection fraction, but he also admitted that his study’s complete reliance on these codes introduced a limitation to the analysis.
Including patients with diastolic dysfunction as well as HFpEF
The study used data collected during 2010-2015 by the National Inpatient Sample, run by the U.S. Department of Health & Human Services in a case-control analysis that included 296,041 patients who had undergone some form of bariatric surgery and 2,004,804 people with no history of bariatric surgery selected as controls on the basis of their obesity.
The absolute numbers showed that, during the observation period, the incidence of acute HFpEF hospitalizations was 0.19% among those with prior bariatric surgery and 0.86% among those with no surgery, and the incidence of chronic heart failure hospitalizations was 0.01% among people with prior bariatric surgery and 0.05% among those without prior surgery. Dr. Funes said. He noted that, during the period studied patients, with HFpEF were usually identified as having diastolic heart failure, an older name for the same disease.
In multivariate analyses that adjusted for age, sex, race, hypertension, diabetes, smoking, and coronary artery disease, people without prior bariatric surgery and with hypertension had a 2.8-fold increased rate of acute hospitalizations for HFpEF, while those without hypertension or prior bariatric surgery had a 5.2-fold increased rate. In addition, control patients, regardless of hypertension status, had a 2.9-fold increased rate of hospitalizations for chronic HFpEF events. All these differences were statistically significant.
Dr. Funes also reported results from additional analyses that focused on a roughly 68,000-patient subgroup of those included in the study who had a history of coronary artery disease, including about 62,000 with no prior bariatric surgery and nearly 6,000 people with prior bariatric surgery. In a multivariate analysis of this subgroup, people without prior bariatric surgery had a 2.65-fold increased rate of hospitalization for a HFpEF event (either acute or chronic), compared with those who had undergone bariatric surgery.
Dr. Funes and associates and Dr. Kindel had no relevant disclosures.
Patients who underwent metabolic and bariatric surgery had fewer than half the number of hospitalizations for both acute and chronic episodes of heart failure with preserved ejection fraction (HFpEF) in a retrospective analysis of more than 2 million Americans collected in a national database.
In a multivariate analysis that adjusted for several variables patients without a history of bariatric surgery had three- to fivefold more hospitalizations for acute events involving HFpEF, and more than double the rate of hospitalizations for chronic HFpEF events, David R. Funes, MD, said at the annual meeting of the American Society for Metabolic and Bariatric Surgery.
While this analysis has the limitations of being retrospective, observational, and entirely reliant on procedure codes to define medical histories and outcomes, it had the advantage of using a large database designed to represent the U.S. adult population, said Dr. Funes, a bariatric surgeon at the Cleveland Clinic in Weston, Fla.
HFpEF effects could ‘extend’ surgery’s use
The report “adds an important article to the literature where there is a true void in trying to discern the effect of bariatric surgery on HFpEF,” commented Tammy L. Kindel, MD, PhD, director of the bariatric surgery program at the Medical College of Wisconsin, Milwaukee, and designated discussant for the report. “Minimal studies [up to now] demonstrate that weight loss in any form can modify diastolic dysfunction in patients with HFpEF. Studies that investigate the impact of bariatric surgery on clinical outcomes in patients with HFpEF are probably the most important for extending use of metabolic surgery,” Dr. Kindel said.
She added that “one of the most difficult parts of studying HFpEF” is making a firm diagnosis that often involves excluding other potential causes. She also questioned Dr. Funes about his confidence that his analysis correctly identified patients only with HFpEF. Dr. Funes replied that the diagnostic codes his team used allowed for a clear distinction between patients identified with HFpEF and those with heart failure with reduced ejection fraction, but he also admitted that his study’s complete reliance on these codes introduced a limitation to the analysis.
Including patients with diastolic dysfunction as well as HFpEF
The study used data collected during 2010-2015 by the National Inpatient Sample, run by the U.S. Department of Health & Human Services in a case-control analysis that included 296,041 patients who had undergone some form of bariatric surgery and 2,004,804 people with no history of bariatric surgery selected as controls on the basis of their obesity.
The absolute numbers showed that, during the observation period, the incidence of acute HFpEF hospitalizations was 0.19% among those with prior bariatric surgery and 0.86% among those with no surgery, and the incidence of chronic heart failure hospitalizations was 0.01% among people with prior bariatric surgery and 0.05% among those without prior surgery. Dr. Funes said. He noted that, during the period studied patients, with HFpEF were usually identified as having diastolic heart failure, an older name for the same disease.
In multivariate analyses that adjusted for age, sex, race, hypertension, diabetes, smoking, and coronary artery disease, people without prior bariatric surgery and with hypertension had a 2.8-fold increased rate of acute hospitalizations for HFpEF, while those without hypertension or prior bariatric surgery had a 5.2-fold increased rate. In addition, control patients, regardless of hypertension status, had a 2.9-fold increased rate of hospitalizations for chronic HFpEF events. All these differences were statistically significant.
Dr. Funes also reported results from additional analyses that focused on a roughly 68,000-patient subgroup of those included in the study who had a history of coronary artery disease, including about 62,000 with no prior bariatric surgery and nearly 6,000 people with prior bariatric surgery. In a multivariate analysis of this subgroup, people without prior bariatric surgery had a 2.65-fold increased rate of hospitalization for a HFpEF event (either acute or chronic), compared with those who had undergone bariatric surgery.
Dr. Funes and associates and Dr. Kindel had no relevant disclosures.
FROM ASMBS 2021
FDA: More metformin extended-release tablets recalled
Two lots of metformin HCl extended-release tablets have been recalled by Viona Pharmaceuticals because unacceptable levels of nitrosodimethylamine (NDMA), a likely carcinogen, were found in the 750-mg tablets.
According to a June 11 alert from the Food and Drug Administration, the affected lot numbers are M915601 and M915602.
This generic product was made by Cadila Healthcare, Ahmedabad, India, in November 2019 with an expiration date of October 2021, and distributed throughout the United States. The pill is white to off-white, capsule-shaped, uncoated tablets, debossed with “Z”, “C” on one side and “20” on the other side.
No adverse events related to the lots involved in the recall have been reported, the FDA said. It also recommends that clinicians continue to prescribe metformin when clinically appropriate.
In late 2019, the FDA announced it had become aware of NDMA in some metformin products in other countries. The agency immediately began testing to determine whether the metformin in the U.S. supply was at risk, as part of the ongoing investigation into nitrosamine impurities across medication types, which included recalls of hypertension and heartburn medications within the past 3 years.
In February 2020, the FDA reported that they hadn’t found NDMA levels that exceeded the acceptable daily intake. But starting in May 2020, voluntary recalls by, numerous manufacturers have been announced as levels of the compound exceeded that cutoff.
Two lots of metformin HCl extended-release tablets have been recalled by Viona Pharmaceuticals because unacceptable levels of nitrosodimethylamine (NDMA), a likely carcinogen, were found in the 750-mg tablets.
According to a June 11 alert from the Food and Drug Administration, the affected lot numbers are M915601 and M915602.
This generic product was made by Cadila Healthcare, Ahmedabad, India, in November 2019 with an expiration date of October 2021, and distributed throughout the United States. The pill is white to off-white, capsule-shaped, uncoated tablets, debossed with “Z”, “C” on one side and “20” on the other side.
No adverse events related to the lots involved in the recall have been reported, the FDA said. It also recommends that clinicians continue to prescribe metformin when clinically appropriate.
In late 2019, the FDA announced it had become aware of NDMA in some metformin products in other countries. The agency immediately began testing to determine whether the metformin in the U.S. supply was at risk, as part of the ongoing investigation into nitrosamine impurities across medication types, which included recalls of hypertension and heartburn medications within the past 3 years.
In February 2020, the FDA reported that they hadn’t found NDMA levels that exceeded the acceptable daily intake. But starting in May 2020, voluntary recalls by, numerous manufacturers have been announced as levels of the compound exceeded that cutoff.
Two lots of metformin HCl extended-release tablets have been recalled by Viona Pharmaceuticals because unacceptable levels of nitrosodimethylamine (NDMA), a likely carcinogen, were found in the 750-mg tablets.
According to a June 11 alert from the Food and Drug Administration, the affected lot numbers are M915601 and M915602.
This generic product was made by Cadila Healthcare, Ahmedabad, India, in November 2019 with an expiration date of October 2021, and distributed throughout the United States. The pill is white to off-white, capsule-shaped, uncoated tablets, debossed with “Z”, “C” on one side and “20” on the other side.
No adverse events related to the lots involved in the recall have been reported, the FDA said. It also recommends that clinicians continue to prescribe metformin when clinically appropriate.
In late 2019, the FDA announced it had become aware of NDMA in some metformin products in other countries. The agency immediately began testing to determine whether the metformin in the U.S. supply was at risk, as part of the ongoing investigation into nitrosamine impurities across medication types, which included recalls of hypertension and heartburn medications within the past 3 years.
In February 2020, the FDA reported that they hadn’t found NDMA levels that exceeded the acceptable daily intake. But starting in May 2020, voluntary recalls by, numerous manufacturers have been announced as levels of the compound exceeded that cutoff.
FROM THE FOOD AND DRUG ADMINISTRATION
Screaming for screens: Digital well-being in the 2020s
Charlie is a 15-year-old male whose medical history includes overweight and autism spectrum disorder. While his autism symptoms are stable and he is doing fairly well in school, your sense is that he is underperforming and unhappy. His screening for anxiety and depression is not outstanding and you wonder whether to leave well enough alone.
Historically, pediatrician queries about media use happen in a minority of visits,1 overcrowded by the multitude of screening and acute care needs, let alone the pressures of electronic health record prompts, billing, and documentation. Yet the COVID-19 pandemic has emphasized what was already getting louder: screen life is becoming a ubiquitous, increasing, and normative function of child development. Digital well-being exhibits bidirectional interactions with most of the core indicators of child health: sleep, nutrition, safety, mood, relationships, and many other aspects of physical and mental health.1
The pandemic unveiled the blessings and curses of digital life by shifting many into remote work and school situations where screen time became both necessary and uncontrollable. Reeling with changes in employment, health, finances, and more, families struggled to forge a new screen-life balance that could bridge academic, professional, and recreational use.
Research has wavered in producing a verdict on the effects of screen time, in part because of limitations in methodology and follow-up time,2 and exacerbated by the quickly changing nature of screen use. Screen time may put youth at risk for obesity and behavior problems,3 but the latter may be mediated in part by loss of sleep because of late-night digital activity.4 While survey data at the population level show little link between screen time and well-being impairments,5 zooming in on individuals may tell a different story. Twenge and Campbell show light use of digital media (compared with nonuse) is associated with greater well-being while heavy use is associated with lower well-being and a higher risk for depression and suicidal behavior – especially in girls.6,7 Largely cross-sectional data show a small detriment to psychological well-being associated with digital technology, though this may be bidirectional and does not clearly differentiate types of technology.2
Recent neuroscience suggests that, compared with active play, sedentary screen time after school reduced impulse control and increased brain activity in regions associated with craving.8 This may explain some of the link between screen time and obesity. Brain imaging of preschoolers showed that greater screen time correlated with lower reading readiness as well as less integrity of white-matter tracts involved in language and executive function,9 whereas nurturing home reading practices were protective for language development and white matter integrity.10
Returning to the care of Charlie, providers may benefit from taking time to reflect on their own digital environment. What does the patient-side view of your office look like? Many offices use telephone reminders and patient portals, fill prescriptions electronically, and have waiting rooms with WiFi or devices for children’s use. Office visits share space with providers’ desktops, laptops, and smartphones, with EMRs guiding the visit. EMRs may come home for evening documentation. How does this affect provider digital well-being? How do you start the conversation with families about digital well-being?
The American Academy of Pediatrics recommends media screening be incorporated into routine pediatric care, with several tools available to support this. Adapting the HEADSSS model for psychosocial check-ins, Clark and colleagues propose an additional “S” to capture screen time.11 Their model queries which apps and social media are used, quantity of use, effects on self-confidence, and whether cyberbullying or sexting are occurring. Smartphones themselves provide an eye-opening and accessible dataset, with built-in features (for example, Screen Time for iOS) tracking not just daily duration of use, but also how frequently the phone is picked up and which apps get more use. Screening may be followed by motivational coaching, emphasizing nonjudgment, curiosity, empathy, and flexibility — for patient and provider.12
In Charlie’s case, screening reveals heavy use of social Internet games that connect him with like-minded peers. While he describes an inclusiveness and level of socialization that he has not found outside the home, the quantity of use is interfering with sleep, schoolwork, and physical activity.
Significant problematic Internet use may lead to intervention or referral – addictive behaviors and mental health symptoms may warrant connection with mental health providers. Cyberbullying or unsafe behaviors may additionally benefit from parental and school-based support. There is early and limited evidence that psychological and educational interventions may be of benefit for problematic Internet use.13
When digital life is not so dramatically affecting well-being, providers may begin by working with families on a media use plan. The AAP offers its own website to support this. Other well-researched and well-designed sites include Digital Wellness Lab For Parents, with developmentally staged information and plentiful research, and Common Sense Media, which reviews apps, movies, and more; plus they have a knowledge/advice section under “Parents Need to Know.” Keep in mind that digital media can also support youth in managing psychiatric problems, e.g., a digital intervention promoting positive psychology practices looked very helpful for young people with psychosis.14
For Charlie, a health coaching approach is adopted. Using Gabrielli’s TECH parenting rubric,15 Charlie’s parents are coached to make space to talk about and coview media and apps, as well as creating a Family Media Use Plan for everyone – parents included. Alongside setting limits on screen time; health promotion activities like exercise, reading, and schoolwork are also rewarded with extra screen time. When Charlie returns 3 months later, the family reports that, in recognition of their collective digital overload, they preserved dinnertime and after 10 p.m. as screen-free downtime. While they still have concerns about Charlie’s gaming and social life, his sleep is somewhat improved and family tension is lower.
Attention to digital well-being stands to benefit provider and patient alike, and over time may gain from the scaffolding of handouts, standardized assessments, and health coaching providers that may be in place to support other important domains like sleep hygiene, food security, and parenting.
Dr. Rosenfeld is assistant professor, University of Vermont, Vermont Center for Children, Youth, and Families, Burlington. He has no relevant disclosures.
References
1. Chassiakos YR et al. Pediatrics. 2016;138(5)e20162593.
2. Orben A. Soc Psychiatry Psych Epi. 2020;55(4):407.
3. Fang K et al. Child Care Health Dev. 2019;45(5):744-53.
4. Janssen X et al. Sleep Med Rev. 2020;49:101226.
5. George MJ et al. J Ped. 2020;219:180.
6. Twenge JM and Campbell WK. Psychiatry Q. 2019;90(2):311-31.
7. Twenge JM and Martin GN. J Adolesc. 2020;79:91.
8. Efraim M et al. Brain Imaging Behav. 2021;15(1):177-89.
9. Hutton JS et al. JAMA Pediatr. 2020;174(1):e193869.
10. Hutton JS et al. Acta Paediatr. 2020;109(7):1376-86.
11. Clark DL et al. Pediatrics. 2018;141(6).
12. Jericho M and Elliot A. Clin Child Psychol Psychiatry. 2020;25(3):662.
13. Malinauskas R and Malinauskine V. J Behav Addict. 2019;8(4):613.
14. Lim MH et al. Soc Psychiatry Psychiatr Epi. 2020;55(7):877-89.
15. Gabrielli J et al. Pediatrics. 2018;142(1)e20173718.
Charlie is a 15-year-old male whose medical history includes overweight and autism spectrum disorder. While his autism symptoms are stable and he is doing fairly well in school, your sense is that he is underperforming and unhappy. His screening for anxiety and depression is not outstanding and you wonder whether to leave well enough alone.
Historically, pediatrician queries about media use happen in a minority of visits,1 overcrowded by the multitude of screening and acute care needs, let alone the pressures of electronic health record prompts, billing, and documentation. Yet the COVID-19 pandemic has emphasized what was already getting louder: screen life is becoming a ubiquitous, increasing, and normative function of child development. Digital well-being exhibits bidirectional interactions with most of the core indicators of child health: sleep, nutrition, safety, mood, relationships, and many other aspects of physical and mental health.1
The pandemic unveiled the blessings and curses of digital life by shifting many into remote work and school situations where screen time became both necessary and uncontrollable. Reeling with changes in employment, health, finances, and more, families struggled to forge a new screen-life balance that could bridge academic, professional, and recreational use.
Research has wavered in producing a verdict on the effects of screen time, in part because of limitations in methodology and follow-up time,2 and exacerbated by the quickly changing nature of screen use. Screen time may put youth at risk for obesity and behavior problems,3 but the latter may be mediated in part by loss of sleep because of late-night digital activity.4 While survey data at the population level show little link between screen time and well-being impairments,5 zooming in on individuals may tell a different story. Twenge and Campbell show light use of digital media (compared with nonuse) is associated with greater well-being while heavy use is associated with lower well-being and a higher risk for depression and suicidal behavior – especially in girls.6,7 Largely cross-sectional data show a small detriment to psychological well-being associated with digital technology, though this may be bidirectional and does not clearly differentiate types of technology.2
Recent neuroscience suggests that, compared with active play, sedentary screen time after school reduced impulse control and increased brain activity in regions associated with craving.8 This may explain some of the link between screen time and obesity. Brain imaging of preschoolers showed that greater screen time correlated with lower reading readiness as well as less integrity of white-matter tracts involved in language and executive function,9 whereas nurturing home reading practices were protective for language development and white matter integrity.10
Returning to the care of Charlie, providers may benefit from taking time to reflect on their own digital environment. What does the patient-side view of your office look like? Many offices use telephone reminders and patient portals, fill prescriptions electronically, and have waiting rooms with WiFi or devices for children’s use. Office visits share space with providers’ desktops, laptops, and smartphones, with EMRs guiding the visit. EMRs may come home for evening documentation. How does this affect provider digital well-being? How do you start the conversation with families about digital well-being?
The American Academy of Pediatrics recommends media screening be incorporated into routine pediatric care, with several tools available to support this. Adapting the HEADSSS model for psychosocial check-ins, Clark and colleagues propose an additional “S” to capture screen time.11 Their model queries which apps and social media are used, quantity of use, effects on self-confidence, and whether cyberbullying or sexting are occurring. Smartphones themselves provide an eye-opening and accessible dataset, with built-in features (for example, Screen Time for iOS) tracking not just daily duration of use, but also how frequently the phone is picked up and which apps get more use. Screening may be followed by motivational coaching, emphasizing nonjudgment, curiosity, empathy, and flexibility — for patient and provider.12
In Charlie’s case, screening reveals heavy use of social Internet games that connect him with like-minded peers. While he describes an inclusiveness and level of socialization that he has not found outside the home, the quantity of use is interfering with sleep, schoolwork, and physical activity.
Significant problematic Internet use may lead to intervention or referral – addictive behaviors and mental health symptoms may warrant connection with mental health providers. Cyberbullying or unsafe behaviors may additionally benefit from parental and school-based support. There is early and limited evidence that psychological and educational interventions may be of benefit for problematic Internet use.13
When digital life is not so dramatically affecting well-being, providers may begin by working with families on a media use plan. The AAP offers its own website to support this. Other well-researched and well-designed sites include Digital Wellness Lab For Parents, with developmentally staged information and plentiful research, and Common Sense Media, which reviews apps, movies, and more; plus they have a knowledge/advice section under “Parents Need to Know.” Keep in mind that digital media can also support youth in managing psychiatric problems, e.g., a digital intervention promoting positive psychology practices looked very helpful for young people with psychosis.14
For Charlie, a health coaching approach is adopted. Using Gabrielli’s TECH parenting rubric,15 Charlie’s parents are coached to make space to talk about and coview media and apps, as well as creating a Family Media Use Plan for everyone – parents included. Alongside setting limits on screen time; health promotion activities like exercise, reading, and schoolwork are also rewarded with extra screen time. When Charlie returns 3 months later, the family reports that, in recognition of their collective digital overload, they preserved dinnertime and after 10 p.m. as screen-free downtime. While they still have concerns about Charlie’s gaming and social life, his sleep is somewhat improved and family tension is lower.
Attention to digital well-being stands to benefit provider and patient alike, and over time may gain from the scaffolding of handouts, standardized assessments, and health coaching providers that may be in place to support other important domains like sleep hygiene, food security, and parenting.
Dr. Rosenfeld is assistant professor, University of Vermont, Vermont Center for Children, Youth, and Families, Burlington. He has no relevant disclosures.
References
1. Chassiakos YR et al. Pediatrics. 2016;138(5)e20162593.
2. Orben A. Soc Psychiatry Psych Epi. 2020;55(4):407.
3. Fang K et al. Child Care Health Dev. 2019;45(5):744-53.
4. Janssen X et al. Sleep Med Rev. 2020;49:101226.
5. George MJ et al. J Ped. 2020;219:180.
6. Twenge JM and Campbell WK. Psychiatry Q. 2019;90(2):311-31.
7. Twenge JM and Martin GN. J Adolesc. 2020;79:91.
8. Efraim M et al. Brain Imaging Behav. 2021;15(1):177-89.
9. Hutton JS et al. JAMA Pediatr. 2020;174(1):e193869.
10. Hutton JS et al. Acta Paediatr. 2020;109(7):1376-86.
11. Clark DL et al. Pediatrics. 2018;141(6).
12. Jericho M and Elliot A. Clin Child Psychol Psychiatry. 2020;25(3):662.
13. Malinauskas R and Malinauskine V. J Behav Addict. 2019;8(4):613.
14. Lim MH et al. Soc Psychiatry Psychiatr Epi. 2020;55(7):877-89.
15. Gabrielli J et al. Pediatrics. 2018;142(1)e20173718.
Charlie is a 15-year-old male whose medical history includes overweight and autism spectrum disorder. While his autism symptoms are stable and he is doing fairly well in school, your sense is that he is underperforming and unhappy. His screening for anxiety and depression is not outstanding and you wonder whether to leave well enough alone.
Historically, pediatrician queries about media use happen in a minority of visits,1 overcrowded by the multitude of screening and acute care needs, let alone the pressures of electronic health record prompts, billing, and documentation. Yet the COVID-19 pandemic has emphasized what was already getting louder: screen life is becoming a ubiquitous, increasing, and normative function of child development. Digital well-being exhibits bidirectional interactions with most of the core indicators of child health: sleep, nutrition, safety, mood, relationships, and many other aspects of physical and mental health.1
The pandemic unveiled the blessings and curses of digital life by shifting many into remote work and school situations where screen time became both necessary and uncontrollable. Reeling with changes in employment, health, finances, and more, families struggled to forge a new screen-life balance that could bridge academic, professional, and recreational use.
Research has wavered in producing a verdict on the effects of screen time, in part because of limitations in methodology and follow-up time,2 and exacerbated by the quickly changing nature of screen use. Screen time may put youth at risk for obesity and behavior problems,3 but the latter may be mediated in part by loss of sleep because of late-night digital activity.4 While survey data at the population level show little link between screen time and well-being impairments,5 zooming in on individuals may tell a different story. Twenge and Campbell show light use of digital media (compared with nonuse) is associated with greater well-being while heavy use is associated with lower well-being and a higher risk for depression and suicidal behavior – especially in girls.6,7 Largely cross-sectional data show a small detriment to psychological well-being associated with digital technology, though this may be bidirectional and does not clearly differentiate types of technology.2
Recent neuroscience suggests that, compared with active play, sedentary screen time after school reduced impulse control and increased brain activity in regions associated with craving.8 This may explain some of the link between screen time and obesity. Brain imaging of preschoolers showed that greater screen time correlated with lower reading readiness as well as less integrity of white-matter tracts involved in language and executive function,9 whereas nurturing home reading practices were protective for language development and white matter integrity.10
Returning to the care of Charlie, providers may benefit from taking time to reflect on their own digital environment. What does the patient-side view of your office look like? Many offices use telephone reminders and patient portals, fill prescriptions electronically, and have waiting rooms with WiFi or devices for children’s use. Office visits share space with providers’ desktops, laptops, and smartphones, with EMRs guiding the visit. EMRs may come home for evening documentation. How does this affect provider digital well-being? How do you start the conversation with families about digital well-being?
The American Academy of Pediatrics recommends media screening be incorporated into routine pediatric care, with several tools available to support this. Adapting the HEADSSS model for psychosocial check-ins, Clark and colleagues propose an additional “S” to capture screen time.11 Their model queries which apps and social media are used, quantity of use, effects on self-confidence, and whether cyberbullying or sexting are occurring. Smartphones themselves provide an eye-opening and accessible dataset, with built-in features (for example, Screen Time for iOS) tracking not just daily duration of use, but also how frequently the phone is picked up and which apps get more use. Screening may be followed by motivational coaching, emphasizing nonjudgment, curiosity, empathy, and flexibility — for patient and provider.12
In Charlie’s case, screening reveals heavy use of social Internet games that connect him with like-minded peers. While he describes an inclusiveness and level of socialization that he has not found outside the home, the quantity of use is interfering with sleep, schoolwork, and physical activity.
Significant problematic Internet use may lead to intervention or referral – addictive behaviors and mental health symptoms may warrant connection with mental health providers. Cyberbullying or unsafe behaviors may additionally benefit from parental and school-based support. There is early and limited evidence that psychological and educational interventions may be of benefit for problematic Internet use.13
When digital life is not so dramatically affecting well-being, providers may begin by working with families on a media use plan. The AAP offers its own website to support this. Other well-researched and well-designed sites include Digital Wellness Lab For Parents, with developmentally staged information and plentiful research, and Common Sense Media, which reviews apps, movies, and more; plus they have a knowledge/advice section under “Parents Need to Know.” Keep in mind that digital media can also support youth in managing psychiatric problems, e.g., a digital intervention promoting positive psychology practices looked very helpful for young people with psychosis.14
For Charlie, a health coaching approach is adopted. Using Gabrielli’s TECH parenting rubric,15 Charlie’s parents are coached to make space to talk about and coview media and apps, as well as creating a Family Media Use Plan for everyone – parents included. Alongside setting limits on screen time; health promotion activities like exercise, reading, and schoolwork are also rewarded with extra screen time. When Charlie returns 3 months later, the family reports that, in recognition of their collective digital overload, they preserved dinnertime and after 10 p.m. as screen-free downtime. While they still have concerns about Charlie’s gaming and social life, his sleep is somewhat improved and family tension is lower.
Attention to digital well-being stands to benefit provider and patient alike, and over time may gain from the scaffolding of handouts, standardized assessments, and health coaching providers that may be in place to support other important domains like sleep hygiene, food security, and parenting.
Dr. Rosenfeld is assistant professor, University of Vermont, Vermont Center for Children, Youth, and Families, Burlington. He has no relevant disclosures.
References
1. Chassiakos YR et al. Pediatrics. 2016;138(5)e20162593.
2. Orben A. Soc Psychiatry Psych Epi. 2020;55(4):407.
3. Fang K et al. Child Care Health Dev. 2019;45(5):744-53.
4. Janssen X et al. Sleep Med Rev. 2020;49:101226.
5. George MJ et al. J Ped. 2020;219:180.
6. Twenge JM and Campbell WK. Psychiatry Q. 2019;90(2):311-31.
7. Twenge JM and Martin GN. J Adolesc. 2020;79:91.
8. Efraim M et al. Brain Imaging Behav. 2021;15(1):177-89.
9. Hutton JS et al. JAMA Pediatr. 2020;174(1):e193869.
10. Hutton JS et al. Acta Paediatr. 2020;109(7):1376-86.
11. Clark DL et al. Pediatrics. 2018;141(6).
12. Jericho M and Elliot A. Clin Child Psychol Psychiatry. 2020;25(3):662.
13. Malinauskas R and Malinauskine V. J Behav Addict. 2019;8(4):613.
14. Lim MH et al. Soc Psychiatry Psychiatr Epi. 2020;55(7):877-89.
15. Gabrielli J et al. Pediatrics. 2018;142(1)e20173718.