User login
Individualized Melanoma Care
Melanoma has become more diverse in terms of the patients affected and the treatment course. Dr. Lorraine L. Rosamilia discusses individualized melanoma care from the early stages of diagnosis through treatment and follow-up. She explains clear margins from excisions and management based on tumor thickness. Coordinated care also is important to evaluate the best treatment course for the patient and favorable outcomes.
Melanoma has become more diverse in terms of the patients affected and the treatment course. Dr. Lorraine L. Rosamilia discusses individualized melanoma care from the early stages of diagnosis through treatment and follow-up. She explains clear margins from excisions and management based on tumor thickness. Coordinated care also is important to evaluate the best treatment course for the patient and favorable outcomes.
Melanoma has become more diverse in terms of the patients affected and the treatment course. Dr. Lorraine L. Rosamilia discusses individualized melanoma care from the early stages of diagnosis through treatment and follow-up. She explains clear margins from excisions and management based on tumor thickness. Coordinated care also is important to evaluate the best treatment course for the patient and favorable outcomes.
Individualizing Care for Melanoma Patients
What does your patient need to know at the first visit?
Melanoma patient visits and communications are complicated. When possible, you can preview the surgical and prognostic expectations with the patient when you see the lesion clinically for the first time, especially for a lesion that you’re sure is melanoma before it even hits the specimen cup. Sometimes you have to convey important biopsy and treatment information over the telephone, or sometimes you have the luxury of an extra clinic visit to discuss it with the patient and perhaps a family member in person.
During the first set of conversations, I explain the things that are tangible: the depth of the lesion, the relationship it has to prognosis, surgical options based on staging, further testing/referrals, and known risk factors. Then I wait. The digestion phase is critical before you hang up and schedule a surgery.
Probably the most important thing I tell patients is that melanoma is unpredictable. Not everyone has the same kind of “surgery,” we don’t say “remission,” we don’t have a certain “chemo” that we know will be best, and not everyone will need “CAT scans.” I tell patients that they’re stuck with me. Skin examinations, talk of sun protection, photography of nevi, upkeep of health maintenance visits with other specialists (eg, primary care physician, optometry/ophthalmology, gynecology, dentistry), and education of family members on melanoma risk factors will be the norm.
What are your go-to treatments?
In the past, surgical management was perhaps streamlined, but as we learn more about melanoma tumor biology, the less we can form generalizable guidelines for surgery, margin control, and lymph node perusal. Wide local excision is paramount, but the evidence that guidelines for 0.5-cm, 1-cm, and 2-cm margins or greater fit for all types of tumors is lacking, and many have challenged the recommended margins, particularly for lentigo maligna subtypes (Kunishige et al).
The 0.75-mm Breslow depth threshold guides my sentinel lymph node (SLN) discussion, with upstaging based on primary tumor mitotic index and/or ulceration. But SLN biopsy is always still a discussion and not a foregone conclusion because the risk-benefit ratio and prognostic/survival data are very difficult to dissect for some cases. Certainly, mention of SLN biopsy morbidity, including lymphedema, is warranted before the patient consents. Then there is further explanation that more invasive lymphadenectomy could occur based on SLN findings. If applicable in advanced cases, options for imaging and adjuvant therapy are introduced by us but then primarily led by oncologists.
Patients are generally pleased that the approach to melanoma surgery, reconstruction, SLN biopsy, and oncologic care is increasingly becoming (almost) harmoniously multidisciplinary. Larger institutions have streamlined their melanoma providers into shared clinics or at least regional networks—dermatologists, plastic/otolaryngology/oncologic surgeons, oncologists, and radiation oncologists, for instance—and simplified patient access based on individual tumor treatment plans. Dr. Christopher J. Miller at Penn Medicine (Philadelphia, Pennsylvania) is excelling at a collaborative surgical approach for melanoma between dermatologic surgery and specialties such as otolaryngology and also spearheading the Mohs micrographic surgery approach for primary melanoma as possible standard of care for some cases (Etzkorn et al).
Also evolving is the access to clinical trials and care coordination regarding the most novel metastatic melanoma therapies, which can now be a short drive away for most patients as the National Cancer Institute broadens its clinical trial reach.
After the diagnosis of melanoma is made, care is becoming much more individualized for surgery and beyond, but true morbidity and mortality benefit for the more complicated regimens has become anybody’s guess as this research field grossly and rapidly swells. Then these discussions with the patient become longer and more open-ended.
How do you keep patients compliant with treatment?
Initial surgery and multidisciplinary management requires meticulous communication between providers to ensure that pathology reports, surgical findings, imaging, wound care, and follow-up are transparent. The easier it is for patients to navigate the medical system(s), the more likely they are to comply. Voicemail, e-mails, text messages, and/or mailings are standard for different offices for reminders, but an old-fashioned telephone call goes a long way for a patient who has melanoma on his/her mind. Adherence to sun protection, health maintenance, and skin examination appointments is then the challenge.
Sun protection strategies were well-represented in the Cutis July issue’s Practical Pearls featuring Dr. Vincent A. DeLeo. My script for sunscreen-averse patients as I leave the room is “Take a mosey through the sunscreen aisle. You may be surprised at what you find.” And sporting goods stores are chock full of UPF (UV protection factor) clothing options. I’m not a scolder, but I do utilize the power of repetition/the squeaky wheel.
Patient delay or cancellation of skin checks requires other types of surveillance. Here at Geisinger Health System (State College, Pennsylvania), when melanoma patients check out, they are placed in a “priority appointment” slot, meaning that if they cancel, their names are automatically put on an electronic list that is compiled weekly by our schedulers to ensure that patients are called for another appointment. To avert telephone tag and delay of care, melanoma patients also get our direct nurses station extension and are encouraged to use our chart e-mail system to communicate with us if they notice a new or changing skin lesion.
What do you do if they refuse treatment?
Initial consent and compliance with melanoma wide local excision are rarely challenged by a well-informed patient, but what I find more common is delay in treatment and nonadherence to periodic skin examinations.
What resources do you recommend to patients for more information?
The National Comprehensive Cancer Network clini-cal practice guideline resource in oncology (melanoma) is the place to start, as their Web site is easily naviga-ble for patients and providers (http://www.nccn.org).
The American Academy of Dermatology (http://www.aad.org) and Skin Cancer Foundation (http://www.skincancer.org) Web sites also provide useful information, and there are always copies of their key melanoma and nevi surveillance pamphlets in our office.
Most recently, my melanoma patients have been inspired by another local patient and her cause: a 28-year-old woman with metastatic melanoma on a National Cancer Institute BRAF inhibitor clinical trial who plans to run an Ironman race this year (Thomason, Ames). Her motivation to maintain her baseline health and fitness while still reaching for this further remarkable goal gives fellow melanoma patients a source for enthusiasm and hope.
Inspiration and adherence come in all shapes and sizes for patients with melanoma. I find you need to throw multiple resources and strategies at them and see what sticks.
Suggested Readings
Ames C. Local runner fights cancer. WeAreCentralPA Web site. http://www.wearecentralpa.com/news/local-runner-fights -cancer#.VZw92-pJ2AU.mailto. Published June 30, 2015. Accessed August 14, 2015.
DeLeo VA. Patient compliance with photoprotection. Cutis. 2015;96:13-14.
Etzkorn JR, Sobanko JF, Elenitsas R, et al. Low recurrence rates for in situ and invasive melanomas using Mohs micrographic surgery with melanoma antigen recognized by T cells 1 (MART-1) immunostaining: tissue processing methodology to optimize pathologic staging and margin assessment. J Am Acad Dermatol. 2015;72:840-850.
Kunishige JH, Brodland DG, Zitelli JA. Larger surgical margins are required for lentigo maligna and other melanoma in situ. J Am Acad Dermatol. 2012;67:1069-1071.
NCCN clinical practice guidelines in oncology: melanoma. National Comprehensive Cancer Network Web site. http://www.nccn.org/professionals/physician_gls/pdf/melanoma.pdf. Updated March 11, 2015. Accessed August 14, 2015.
Thomason M. My story: April Salinas. Live In Our Skinz! Blog. http://blog.uvskinz.com/2015/01/30/my-story-april -salinas/#sthash.yvSRVglT.YGLDn95T.dpbs. Published January 30, 2015. Accessed August 14, 2015.
What does your patient need to know at the first visit?
Melanoma patient visits and communications are complicated. When possible, you can preview the surgical and prognostic expectations with the patient when you see the lesion clinically for the first time, especially for a lesion that you’re sure is melanoma before it even hits the specimen cup. Sometimes you have to convey important biopsy and treatment information over the telephone, or sometimes you have the luxury of an extra clinic visit to discuss it with the patient and perhaps a family member in person.
During the first set of conversations, I explain the things that are tangible: the depth of the lesion, the relationship it has to prognosis, surgical options based on staging, further testing/referrals, and known risk factors. Then I wait. The digestion phase is critical before you hang up and schedule a surgery.
Probably the most important thing I tell patients is that melanoma is unpredictable. Not everyone has the same kind of “surgery,” we don’t say “remission,” we don’t have a certain “chemo” that we know will be best, and not everyone will need “CAT scans.” I tell patients that they’re stuck with me. Skin examinations, talk of sun protection, photography of nevi, upkeep of health maintenance visits with other specialists (eg, primary care physician, optometry/ophthalmology, gynecology, dentistry), and education of family members on melanoma risk factors will be the norm.
What are your go-to treatments?
In the past, surgical management was perhaps streamlined, but as we learn more about melanoma tumor biology, the less we can form generalizable guidelines for surgery, margin control, and lymph node perusal. Wide local excision is paramount, but the evidence that guidelines for 0.5-cm, 1-cm, and 2-cm margins or greater fit for all types of tumors is lacking, and many have challenged the recommended margins, particularly for lentigo maligna subtypes (Kunishige et al).
The 0.75-mm Breslow depth threshold guides my sentinel lymph node (SLN) discussion, with upstaging based on primary tumor mitotic index and/or ulceration. But SLN biopsy is always still a discussion and not a foregone conclusion because the risk-benefit ratio and prognostic/survival data are very difficult to dissect for some cases. Certainly, mention of SLN biopsy morbidity, including lymphedema, is warranted before the patient consents. Then there is further explanation that more invasive lymphadenectomy could occur based on SLN findings. If applicable in advanced cases, options for imaging and adjuvant therapy are introduced by us but then primarily led by oncologists.
Patients are generally pleased that the approach to melanoma surgery, reconstruction, SLN biopsy, and oncologic care is increasingly becoming (almost) harmoniously multidisciplinary. Larger institutions have streamlined their melanoma providers into shared clinics or at least regional networks—dermatologists, plastic/otolaryngology/oncologic surgeons, oncologists, and radiation oncologists, for instance—and simplified patient access based on individual tumor treatment plans. Dr. Christopher J. Miller at Penn Medicine (Philadelphia, Pennsylvania) is excelling at a collaborative surgical approach for melanoma between dermatologic surgery and specialties such as otolaryngology and also spearheading the Mohs micrographic surgery approach for primary melanoma as possible standard of care for some cases (Etzkorn et al).
Also evolving is the access to clinical trials and care coordination regarding the most novel metastatic melanoma therapies, which can now be a short drive away for most patients as the National Cancer Institute broadens its clinical trial reach.
After the diagnosis of melanoma is made, care is becoming much more individualized for surgery and beyond, but true morbidity and mortality benefit for the more complicated regimens has become anybody’s guess as this research field grossly and rapidly swells. Then these discussions with the patient become longer and more open-ended.
How do you keep patients compliant with treatment?
Initial surgery and multidisciplinary management requires meticulous communication between providers to ensure that pathology reports, surgical findings, imaging, wound care, and follow-up are transparent. The easier it is for patients to navigate the medical system(s), the more likely they are to comply. Voicemail, e-mails, text messages, and/or mailings are standard for different offices for reminders, but an old-fashioned telephone call goes a long way for a patient who has melanoma on his/her mind. Adherence to sun protection, health maintenance, and skin examination appointments is then the challenge.
Sun protection strategies were well-represented in the Cutis July issue’s Practical Pearls featuring Dr. Vincent A. DeLeo. My script for sunscreen-averse patients as I leave the room is “Take a mosey through the sunscreen aisle. You may be surprised at what you find.” And sporting goods stores are chock full of UPF (UV protection factor) clothing options. I’m not a scolder, but I do utilize the power of repetition/the squeaky wheel.
Patient delay or cancellation of skin checks requires other types of surveillance. Here at Geisinger Health System (State College, Pennsylvania), when melanoma patients check out, they are placed in a “priority appointment” slot, meaning that if they cancel, their names are automatically put on an electronic list that is compiled weekly by our schedulers to ensure that patients are called for another appointment. To avert telephone tag and delay of care, melanoma patients also get our direct nurses station extension and are encouraged to use our chart e-mail system to communicate with us if they notice a new or changing skin lesion.
What do you do if they refuse treatment?
Initial consent and compliance with melanoma wide local excision are rarely challenged by a well-informed patient, but what I find more common is delay in treatment and nonadherence to periodic skin examinations.
What resources do you recommend to patients for more information?
The National Comprehensive Cancer Network clini-cal practice guideline resource in oncology (melanoma) is the place to start, as their Web site is easily naviga-ble for patients and providers (http://www.nccn.org).
The American Academy of Dermatology (http://www.aad.org) and Skin Cancer Foundation (http://www.skincancer.org) Web sites also provide useful information, and there are always copies of their key melanoma and nevi surveillance pamphlets in our office.
Most recently, my melanoma patients have been inspired by another local patient and her cause: a 28-year-old woman with metastatic melanoma on a National Cancer Institute BRAF inhibitor clinical trial who plans to run an Ironman race this year (Thomason, Ames). Her motivation to maintain her baseline health and fitness while still reaching for this further remarkable goal gives fellow melanoma patients a source for enthusiasm and hope.
Inspiration and adherence come in all shapes and sizes for patients with melanoma. I find you need to throw multiple resources and strategies at them and see what sticks.
What does your patient need to know at the first visit?
Melanoma patient visits and communications are complicated. When possible, you can preview the surgical and prognostic expectations with the patient when you see the lesion clinically for the first time, especially for a lesion that you’re sure is melanoma before it even hits the specimen cup. Sometimes you have to convey important biopsy and treatment information over the telephone, or sometimes you have the luxury of an extra clinic visit to discuss it with the patient and perhaps a family member in person.
During the first set of conversations, I explain the things that are tangible: the depth of the lesion, the relationship it has to prognosis, surgical options based on staging, further testing/referrals, and known risk factors. Then I wait. The digestion phase is critical before you hang up and schedule a surgery.
Probably the most important thing I tell patients is that melanoma is unpredictable. Not everyone has the same kind of “surgery,” we don’t say “remission,” we don’t have a certain “chemo” that we know will be best, and not everyone will need “CAT scans.” I tell patients that they’re stuck with me. Skin examinations, talk of sun protection, photography of nevi, upkeep of health maintenance visits with other specialists (eg, primary care physician, optometry/ophthalmology, gynecology, dentistry), and education of family members on melanoma risk factors will be the norm.
What are your go-to treatments?
In the past, surgical management was perhaps streamlined, but as we learn more about melanoma tumor biology, the less we can form generalizable guidelines for surgery, margin control, and lymph node perusal. Wide local excision is paramount, but the evidence that guidelines for 0.5-cm, 1-cm, and 2-cm margins or greater fit for all types of tumors is lacking, and many have challenged the recommended margins, particularly for lentigo maligna subtypes (Kunishige et al).
The 0.75-mm Breslow depth threshold guides my sentinel lymph node (SLN) discussion, with upstaging based on primary tumor mitotic index and/or ulceration. But SLN biopsy is always still a discussion and not a foregone conclusion because the risk-benefit ratio and prognostic/survival data are very difficult to dissect for some cases. Certainly, mention of SLN biopsy morbidity, including lymphedema, is warranted before the patient consents. Then there is further explanation that more invasive lymphadenectomy could occur based on SLN findings. If applicable in advanced cases, options for imaging and adjuvant therapy are introduced by us but then primarily led by oncologists.
Patients are generally pleased that the approach to melanoma surgery, reconstruction, SLN biopsy, and oncologic care is increasingly becoming (almost) harmoniously multidisciplinary. Larger institutions have streamlined their melanoma providers into shared clinics or at least regional networks—dermatologists, plastic/otolaryngology/oncologic surgeons, oncologists, and radiation oncologists, for instance—and simplified patient access based on individual tumor treatment plans. Dr. Christopher J. Miller at Penn Medicine (Philadelphia, Pennsylvania) is excelling at a collaborative surgical approach for melanoma between dermatologic surgery and specialties such as otolaryngology and also spearheading the Mohs micrographic surgery approach for primary melanoma as possible standard of care for some cases (Etzkorn et al).
Also evolving is the access to clinical trials and care coordination regarding the most novel metastatic melanoma therapies, which can now be a short drive away for most patients as the National Cancer Institute broadens its clinical trial reach.
After the diagnosis of melanoma is made, care is becoming much more individualized for surgery and beyond, but true morbidity and mortality benefit for the more complicated regimens has become anybody’s guess as this research field grossly and rapidly swells. Then these discussions with the patient become longer and more open-ended.
How do you keep patients compliant with treatment?
Initial surgery and multidisciplinary management requires meticulous communication between providers to ensure that pathology reports, surgical findings, imaging, wound care, and follow-up are transparent. The easier it is for patients to navigate the medical system(s), the more likely they are to comply. Voicemail, e-mails, text messages, and/or mailings are standard for different offices for reminders, but an old-fashioned telephone call goes a long way for a patient who has melanoma on his/her mind. Adherence to sun protection, health maintenance, and skin examination appointments is then the challenge.
Sun protection strategies were well-represented in the Cutis July issue’s Practical Pearls featuring Dr. Vincent A. DeLeo. My script for sunscreen-averse patients as I leave the room is “Take a mosey through the sunscreen aisle. You may be surprised at what you find.” And sporting goods stores are chock full of UPF (UV protection factor) clothing options. I’m not a scolder, but I do utilize the power of repetition/the squeaky wheel.
Patient delay or cancellation of skin checks requires other types of surveillance. Here at Geisinger Health System (State College, Pennsylvania), when melanoma patients check out, they are placed in a “priority appointment” slot, meaning that if they cancel, their names are automatically put on an electronic list that is compiled weekly by our schedulers to ensure that patients are called for another appointment. To avert telephone tag and delay of care, melanoma patients also get our direct nurses station extension and are encouraged to use our chart e-mail system to communicate with us if they notice a new or changing skin lesion.
What do you do if they refuse treatment?
Initial consent and compliance with melanoma wide local excision are rarely challenged by a well-informed patient, but what I find more common is delay in treatment and nonadherence to periodic skin examinations.
What resources do you recommend to patients for more information?
The National Comprehensive Cancer Network clini-cal practice guideline resource in oncology (melanoma) is the place to start, as their Web site is easily naviga-ble for patients and providers (http://www.nccn.org).
The American Academy of Dermatology (http://www.aad.org) and Skin Cancer Foundation (http://www.skincancer.org) Web sites also provide useful information, and there are always copies of their key melanoma and nevi surveillance pamphlets in our office.
Most recently, my melanoma patients have been inspired by another local patient and her cause: a 28-year-old woman with metastatic melanoma on a National Cancer Institute BRAF inhibitor clinical trial who plans to run an Ironman race this year (Thomason, Ames). Her motivation to maintain her baseline health and fitness while still reaching for this further remarkable goal gives fellow melanoma patients a source for enthusiasm and hope.
Inspiration and adherence come in all shapes and sizes for patients with melanoma. I find you need to throw multiple resources and strategies at them and see what sticks.
Suggested Readings
Ames C. Local runner fights cancer. WeAreCentralPA Web site. http://www.wearecentralpa.com/news/local-runner-fights -cancer#.VZw92-pJ2AU.mailto. Published June 30, 2015. Accessed August 14, 2015.
DeLeo VA. Patient compliance with photoprotection. Cutis. 2015;96:13-14.
Etzkorn JR, Sobanko JF, Elenitsas R, et al. Low recurrence rates for in situ and invasive melanomas using Mohs micrographic surgery with melanoma antigen recognized by T cells 1 (MART-1) immunostaining: tissue processing methodology to optimize pathologic staging and margin assessment. J Am Acad Dermatol. 2015;72:840-850.
Kunishige JH, Brodland DG, Zitelli JA. Larger surgical margins are required for lentigo maligna and other melanoma in situ. J Am Acad Dermatol. 2012;67:1069-1071.
NCCN clinical practice guidelines in oncology: melanoma. National Comprehensive Cancer Network Web site. http://www.nccn.org/professionals/physician_gls/pdf/melanoma.pdf. Updated March 11, 2015. Accessed August 14, 2015.
Thomason M. My story: April Salinas. Live In Our Skinz! Blog. http://blog.uvskinz.com/2015/01/30/my-story-april -salinas/#sthash.yvSRVglT.YGLDn95T.dpbs. Published January 30, 2015. Accessed August 14, 2015.
Suggested Readings
Ames C. Local runner fights cancer. WeAreCentralPA Web site. http://www.wearecentralpa.com/news/local-runner-fights -cancer#.VZw92-pJ2AU.mailto. Published June 30, 2015. Accessed August 14, 2015.
DeLeo VA. Patient compliance with photoprotection. Cutis. 2015;96:13-14.
Etzkorn JR, Sobanko JF, Elenitsas R, et al. Low recurrence rates for in situ and invasive melanomas using Mohs micrographic surgery with melanoma antigen recognized by T cells 1 (MART-1) immunostaining: tissue processing methodology to optimize pathologic staging and margin assessment. J Am Acad Dermatol. 2015;72:840-850.
Kunishige JH, Brodland DG, Zitelli JA. Larger surgical margins are required for lentigo maligna and other melanoma in situ. J Am Acad Dermatol. 2012;67:1069-1071.
NCCN clinical practice guidelines in oncology: melanoma. National Comprehensive Cancer Network Web site. http://www.nccn.org/professionals/physician_gls/pdf/melanoma.pdf. Updated March 11, 2015. Accessed August 14, 2015.
Thomason M. My story: April Salinas. Live In Our Skinz! Blog. http://blog.uvskinz.com/2015/01/30/my-story-april -salinas/#sthash.yvSRVglT.YGLDn95T.dpbs. Published January 30, 2015. Accessed August 14, 2015.
GM-CSF did not increase survival in high-risk melanoma
Neither adjuvant granulocyte macrophage colony-stimulating factor (GM-CSF) nor a peptide vaccination (PV) significantly improved relapse-free or overall survival in patients with high-risk stage III and IV melanoma who had already undergone surgical resection, according to a study published online Sept. 8 in the Journal of Clinical Oncology.
Findings from early clinical trials support the possible benefit of GM-CSF as adjuvant therapy for melanoma, said the authors, led by Dr. David H. Lawson of Winship Cancer Institute, Emory University, Atlanta. Some studies have also shown that combining cytokines, including GM-CSF, with melanoma vaccines yields augmented immunologic responses and clinically significant tumor responses.
However, the improvement in survival in the current study did not reach statistical significance. The median survival for patients who received GM-CSF was 69.6 months, compared to 59.3 months for the patients who received placebo. This represented a 17.4% improvement in patients who received GM-CSF, but it was less than the projected absolute increase of 13.3 months and the relative improvement of 33% required for significance (HR, 0.94; 95% repeated CI, 0.77 to 1.15; stratified log-rank P =.528).
The results for relapse-free survival also did not reach statistical significance. For patients treated with GM-CSF it was 11.4 months (95% CI,9.4 to 14.8 months) compared to 8.8 month in the placebo group (95% CI, 7.5 to 11.2 months), which was an increase of 2.6 months, or 30% (HR, 0.88; 95% CI,0.74 to 1.04; stratified log-rank P = .131).
Just over half of the cohort (53.3%) were HLA-A2–positive patients. This group was randomized to receive PV alone or with GM-CSF, or placebo. The median overall survival was 68.6 months (95% CI, 47.0 to 92.3 months) in patients who received PV and 63.3 months (95% CI,49.2 to 105.0 months) for those who received placebo (HR, 0.93; 95% CI0.71 to1.21; P = .598)
Also in this subgroup, the median relapse-free survival was only 1.7 months longer in patients who received PV than for those who got placebo (11.5 v 9.8 months, for a 17.3% improvement). This was also lower than what was expected (3 months; 33% improvement), and also did not reach statistical significance.
Even though this study did not support their hypotheses, “trials that test GM-CSF in patients with resected visceral melanoma metastases are worthy of consideration,” wrote the authors. “GM-CSF may find its greatest use in melanoma in combination with other agents” (J Clin Oncol. 2015 Sep 8. doi:10.1200/JCO.2015.62.0500).
The study was supported by grants from the Public Health Service, National Institutes of Health, and the National Cancer Institute. Several of the coauthors report financial relationships with industry.
Neither adjuvant granulocyte macrophage colony-stimulating factor (GM-CSF) nor a peptide vaccination (PV) significantly improved relapse-free or overall survival in patients with high-risk stage III and IV melanoma who had already undergone surgical resection, according to a study published online Sept. 8 in the Journal of Clinical Oncology.
Findings from early clinical trials support the possible benefit of GM-CSF as adjuvant therapy for melanoma, said the authors, led by Dr. David H. Lawson of Winship Cancer Institute, Emory University, Atlanta. Some studies have also shown that combining cytokines, including GM-CSF, with melanoma vaccines yields augmented immunologic responses and clinically significant tumor responses.
However, the improvement in survival in the current study did not reach statistical significance. The median survival for patients who received GM-CSF was 69.6 months, compared to 59.3 months for the patients who received placebo. This represented a 17.4% improvement in patients who received GM-CSF, but it was less than the projected absolute increase of 13.3 months and the relative improvement of 33% required for significance (HR, 0.94; 95% repeated CI, 0.77 to 1.15; stratified log-rank P =.528).
The results for relapse-free survival also did not reach statistical significance. For patients treated with GM-CSF it was 11.4 months (95% CI,9.4 to 14.8 months) compared to 8.8 month in the placebo group (95% CI, 7.5 to 11.2 months), which was an increase of 2.6 months, or 30% (HR, 0.88; 95% CI,0.74 to 1.04; stratified log-rank P = .131).
Just over half of the cohort (53.3%) were HLA-A2–positive patients. This group was randomized to receive PV alone or with GM-CSF, or placebo. The median overall survival was 68.6 months (95% CI, 47.0 to 92.3 months) in patients who received PV and 63.3 months (95% CI,49.2 to 105.0 months) for those who received placebo (HR, 0.93; 95% CI0.71 to1.21; P = .598)
Also in this subgroup, the median relapse-free survival was only 1.7 months longer in patients who received PV than for those who got placebo (11.5 v 9.8 months, for a 17.3% improvement). This was also lower than what was expected (3 months; 33% improvement), and also did not reach statistical significance.
Even though this study did not support their hypotheses, “trials that test GM-CSF in patients with resected visceral melanoma metastases are worthy of consideration,” wrote the authors. “GM-CSF may find its greatest use in melanoma in combination with other agents” (J Clin Oncol. 2015 Sep 8. doi:10.1200/JCO.2015.62.0500).
The study was supported by grants from the Public Health Service, National Institutes of Health, and the National Cancer Institute. Several of the coauthors report financial relationships with industry.
Neither adjuvant granulocyte macrophage colony-stimulating factor (GM-CSF) nor a peptide vaccination (PV) significantly improved relapse-free or overall survival in patients with high-risk stage III and IV melanoma who had already undergone surgical resection, according to a study published online Sept. 8 in the Journal of Clinical Oncology.
Findings from early clinical trials support the possible benefit of GM-CSF as adjuvant therapy for melanoma, said the authors, led by Dr. David H. Lawson of Winship Cancer Institute, Emory University, Atlanta. Some studies have also shown that combining cytokines, including GM-CSF, with melanoma vaccines yields augmented immunologic responses and clinically significant tumor responses.
However, the improvement in survival in the current study did not reach statistical significance. The median survival for patients who received GM-CSF was 69.6 months, compared to 59.3 months for the patients who received placebo. This represented a 17.4% improvement in patients who received GM-CSF, but it was less than the projected absolute increase of 13.3 months and the relative improvement of 33% required for significance (HR, 0.94; 95% repeated CI, 0.77 to 1.15; stratified log-rank P =.528).
The results for relapse-free survival also did not reach statistical significance. For patients treated with GM-CSF it was 11.4 months (95% CI,9.4 to 14.8 months) compared to 8.8 month in the placebo group (95% CI, 7.5 to 11.2 months), which was an increase of 2.6 months, or 30% (HR, 0.88; 95% CI,0.74 to 1.04; stratified log-rank P = .131).
Just over half of the cohort (53.3%) were HLA-A2–positive patients. This group was randomized to receive PV alone or with GM-CSF, or placebo. The median overall survival was 68.6 months (95% CI, 47.0 to 92.3 months) in patients who received PV and 63.3 months (95% CI,49.2 to 105.0 months) for those who received placebo (HR, 0.93; 95% CI0.71 to1.21; P = .598)
Also in this subgroup, the median relapse-free survival was only 1.7 months longer in patients who received PV than for those who got placebo (11.5 v 9.8 months, for a 17.3% improvement). This was also lower than what was expected (3 months; 33% improvement), and also did not reach statistical significance.
Even though this study did not support their hypotheses, “trials that test GM-CSF in patients with resected visceral melanoma metastases are worthy of consideration,” wrote the authors. “GM-CSF may find its greatest use in melanoma in combination with other agents” (J Clin Oncol. 2015 Sep 8. doi:10.1200/JCO.2015.62.0500).
The study was supported by grants from the Public Health Service, National Institutes of Health, and the National Cancer Institute. Several of the coauthors report financial relationships with industry.
FROM JOURNAL OF CLINICAL ONCOLOGY
Key clinical point: Adjuvant granulocyte macrophage colony-stimulating factor and peptide vaccination did not significantly improve relapse-free or overall survival in patients with high-risk resected melanoma.
Major finding: The median overall survival with GM-CSF versus placebo treatments was 69.6 months versus 59.3 months, and relapse-free survival was 11.4 months versus 8.8 months.
Data source: A study of 815 patients with high-risk resected melanoma who were randomly assigned to receive GM-CSF, PV, both, or placebo.
Disclosures: The study was supported by grants from the Public Health Service, National Institutes of Health, and the National Cancer Institute. Several of the coauthors report financial relationships with industry.
Melanoma regression a positive prognostic indicator
Histologic regression in melanoma is associated with a significantly lower risk of sentinel lymph node positivity and may serve as a prognostic factor when deciding whether to perform a sentinel lymph node biopsy, according to a study published online Sept. 2 in JAMA Dermatology.
Researchers conducted a meta-analysis of 14 studies in 10,098 patients who showed histologic regression of a primary melanoma and whose sentinel lymph node status was available. In the 14 studies combined, the analysis showed patients with histologic regression had a 44% lower likelihood of having a positive sentinel lymph node than patients without regression, reported Dr. Simone Ribero of the University of Turin (Italy), and coauthors (JAMA Dermatol. 2015 Sep 2. doi: 10.1001/jamadermatol.2015.2235).
This figure was even lower for patients enrolled in high-quality studies, compared with lower-quality studies, they said. In the nine studies considered high quality, patients with regression had a 52% lower likelihood of having a positive sentinel lymph node, while those enrolled in the five studies considered low quality had a 27% lower likelihood of having a positive sentinel lymph node.
The prognostic significance of regression in primary melanoma has been confounded by the fact that disappearance of part of the tumor can result in an underestimation of the original Breslow thickness. Some previous studies have found an increase in sentinel lymph node positivity associated with regression, while other studies suggested that regression was in fact a protective factor against sentinel lymph node metastasis.
Although the studies used different definitions of histologic regression and all were observational (counted among the limitations of the studies), the authors concluded that regression was prognostically favorable. “The results of this meta-analysis may be useful when deciding to offer SLNB [sentinel lymph node biopsy] to patients with regressions of melanomas,” they wrote, adding: “It may help clinicians make a final selection of the most appropriate patients for this procedure.”
The authors had no conflicts of interest to declare.
The finding that patients with histologic regression have half the risk for micrometastases in sentinel lymph node biopsy, compared with patients without regression gives a clear answer to the question of whether patients with thin melanoma and regression should undergo SLNB, and that answer is “No.”

|
Dr. Claus Garbe |
These findings underline that fact that prognosis of patients with melanoma does not depend only on the characteristics of the tumor itself, such as tumor thickness, ulceration, and mitosis, but also on the immune response of the host.
Dr. Claus Garbe is from the Department of Dermatology, University Hospital Tübingen (Germany). These comments are taken from an accompanying editorial (JAMA Dermatol. 2015 Sep 2. doi: 10.1001/jamadermatol.2015.2235). No conflicts of interest were declared.
The finding that patients with histologic regression have half the risk for micrometastases in sentinel lymph node biopsy, compared with patients without regression gives a clear answer to the question of whether patients with thin melanoma and regression should undergo SLNB, and that answer is “No.”

|
Dr. Claus Garbe |
These findings underline that fact that prognosis of patients with melanoma does not depend only on the characteristics of the tumor itself, such as tumor thickness, ulceration, and mitosis, but also on the immune response of the host.
Dr. Claus Garbe is from the Department of Dermatology, University Hospital Tübingen (Germany). These comments are taken from an accompanying editorial (JAMA Dermatol. 2015 Sep 2. doi: 10.1001/jamadermatol.2015.2235). No conflicts of interest were declared.
The finding that patients with histologic regression have half the risk for micrometastases in sentinel lymph node biopsy, compared with patients without regression gives a clear answer to the question of whether patients with thin melanoma and regression should undergo SLNB, and that answer is “No.”

|
Dr. Claus Garbe |
These findings underline that fact that prognosis of patients with melanoma does not depend only on the characteristics of the tumor itself, such as tumor thickness, ulceration, and mitosis, but also on the immune response of the host.
Dr. Claus Garbe is from the Department of Dermatology, University Hospital Tübingen (Germany). These comments are taken from an accompanying editorial (JAMA Dermatol. 2015 Sep 2. doi: 10.1001/jamadermatol.2015.2235). No conflicts of interest were declared.
Histologic regression in melanoma is associated with a significantly lower risk of sentinel lymph node positivity and may serve as a prognostic factor when deciding whether to perform a sentinel lymph node biopsy, according to a study published online Sept. 2 in JAMA Dermatology.
Researchers conducted a meta-analysis of 14 studies in 10,098 patients who showed histologic regression of a primary melanoma and whose sentinel lymph node status was available. In the 14 studies combined, the analysis showed patients with histologic regression had a 44% lower likelihood of having a positive sentinel lymph node than patients without regression, reported Dr. Simone Ribero of the University of Turin (Italy), and coauthors (JAMA Dermatol. 2015 Sep 2. doi: 10.1001/jamadermatol.2015.2235).
This figure was even lower for patients enrolled in high-quality studies, compared with lower-quality studies, they said. In the nine studies considered high quality, patients with regression had a 52% lower likelihood of having a positive sentinel lymph node, while those enrolled in the five studies considered low quality had a 27% lower likelihood of having a positive sentinel lymph node.
The prognostic significance of regression in primary melanoma has been confounded by the fact that disappearance of part of the tumor can result in an underestimation of the original Breslow thickness. Some previous studies have found an increase in sentinel lymph node positivity associated with regression, while other studies suggested that regression was in fact a protective factor against sentinel lymph node metastasis.
Although the studies used different definitions of histologic regression and all were observational (counted among the limitations of the studies), the authors concluded that regression was prognostically favorable. “The results of this meta-analysis may be useful when deciding to offer SLNB [sentinel lymph node biopsy] to patients with regressions of melanomas,” they wrote, adding: “It may help clinicians make a final selection of the most appropriate patients for this procedure.”
The authors had no conflicts of interest to declare.
Histologic regression in melanoma is associated with a significantly lower risk of sentinel lymph node positivity and may serve as a prognostic factor when deciding whether to perform a sentinel lymph node biopsy, according to a study published online Sept. 2 in JAMA Dermatology.
Researchers conducted a meta-analysis of 14 studies in 10,098 patients who showed histologic regression of a primary melanoma and whose sentinel lymph node status was available. In the 14 studies combined, the analysis showed patients with histologic regression had a 44% lower likelihood of having a positive sentinel lymph node than patients without regression, reported Dr. Simone Ribero of the University of Turin (Italy), and coauthors (JAMA Dermatol. 2015 Sep 2. doi: 10.1001/jamadermatol.2015.2235).
This figure was even lower for patients enrolled in high-quality studies, compared with lower-quality studies, they said. In the nine studies considered high quality, patients with regression had a 52% lower likelihood of having a positive sentinel lymph node, while those enrolled in the five studies considered low quality had a 27% lower likelihood of having a positive sentinel lymph node.
The prognostic significance of regression in primary melanoma has been confounded by the fact that disappearance of part of the tumor can result in an underestimation of the original Breslow thickness. Some previous studies have found an increase in sentinel lymph node positivity associated with regression, while other studies suggested that regression was in fact a protective factor against sentinel lymph node metastasis.
Although the studies used different definitions of histologic regression and all were observational (counted among the limitations of the studies), the authors concluded that regression was prognostically favorable. “The results of this meta-analysis may be useful when deciding to offer SLNB [sentinel lymph node biopsy] to patients with regressions of melanomas,” they wrote, adding: “It may help clinicians make a final selection of the most appropriate patients for this procedure.”
The authors had no conflicts of interest to declare.
FROM JAMA DERMATOLOGY
Key clinical point: Histologic regression in melanoma is associated with a significantly lower risk of sentinel lymph node positivity.
Major finding: Individuals with histologic regression in melanoma had a 44% lower likelihood of having a positive sentinel lymph node than patients without regression.
Data source: A meta-analysis evaluated 14 studies in 10,098 patients.
Disclosures: None of the authors had conflicts of interest to disclose.
Fewer moles linked with more aggressive melanoma
NEW YORK – Consistent with another recently published report but counterintuitive, patients with a high number of nevi prior to developing a melanoma have more favorable prognostic features than do those with fewer moles, according to data presented at the American Academy of Dermatology summer meeting.
Based on differences in numerous clinicopathologic features when patients with more than 50 nevi were compared with those with fewer than 50 nevi, “It is possible that there are different pathways that drive melanoma in these two patient groups, resulting in different degrees of aggressiveness,” reported Dr. Caroline Kim from the Pigmented Lesion Clinic and Cutaneous Oncology Program, Beth Israel Deaconess Medical Center, Harvard Medical School, Boston.
In this study of 281 melanoma patients evaluated over 2 years at Dr. Kim’s clinic, those with a high nevi count (89 patients) were compared with those with a lower nevi count (192 patients). The investigators also performed comparisons on those who had dysplastic versus nondysplastic nevi. Of differences between the groups, the initial melanoma grade was the most surprising.
In the greater-than-50-nevi group, 21.6% had either stage III (18.2%) or stage IV (3.4%) disease at diagnosis vs. 40.2% in the fewer-than-50-nevi group (31.2% and 9.0% with stage III and IV disease, respectively). Conversely, melanoma was diagnosed at stage I in 71.6% in the greater-than-50-nevi group vs. only 41.8% in the fewer-than-50-nevi group.
Among patients in the fewer-than-50-nevi group, ulceration was more common (25.1% vs. 10.3%; P = .017) as was nodular subtype (11.2% vs. 2.9%; P = .035). The group with the lower nevi count also had a higher mitotic rate (3.7 vs. 2.3 count/mm2) and a greater Breslow’s depth (1.78 vs. 1.21 mm; P = .010). Although not significantly different, the proportion of females was higher in the lower nevi count group (56.2% vs. 46.4%) and more patients in this group developed a melanoma in the head or neck (21.6% vs. 15.3%).
There are several potential explanations for the differences observed in this study. One is that the process that drives a high nevi count is different than that which produces lower numbers of nevi. According to Dr. Kim, some of the pathologic features in the greater-than-50-nevi group were more consistent with BRAF mutation patterns than the fewer-than- 50-nevi group. Another is that a high nevi count drives a more robust immune response against melanoma growth. Dr. Kim cited published studies that support both theories.
However, it is also possible that high nevi count increases likelihood of dermatologic care and vigilance for early detection of melanomas. This might be supported by the fact that the age of diagnosis was significantly younger in the greater-than-50-nevi count group (40.1 vs. 50.1 years; P less than .001). However, a younger age at diagnosis could also be a product of different pathways of disease development and growth.
The greater likelihood of more aggressive melanoma in patients with a history of fewer than 50 nevi than in those with greater than 50 nevi has also been supported by another large series that was recently published (Int J Cancer. 2015 Oct 1;137[7]:1691-8.) Cited by Dr. Kim as consistent with her own findings, this study of 2,184 melanoma cases compared 5- and 10-year survival in those who had greater than 50 nevi, which represented 31.3% of this population, to those with fewer. The greater survival at 5 years in the greater-than-50-nevi group (91.2% vs. 86.4%) and 10 years (87.2% vs. 79%) remained significant after adjusting “for all known melanoma prognostic factors,” according to the authors.
Further studies evaluating differences in patterns of melanoma growth in those with fewer than 50 nevi vs. those with more are needed, according to Dr. Kim. She acknowledged that one limitation of her study was that all cases were drawn from a single center. However, there is support for an independent study, and the findings, if verified, “have major implications for public health screening and education.” In particular, she suggested that those with lower nevi counts might deserve the type of attention already being directed at those with high nevi counts “as they may be paradoxically at risk for more aggressive melanomas.”
Dr. Kim reported financial relationships with Hoffman-LaRoche, Castle Biosciences, and Shape Pharmaceuticals.
This article was updated August 26, 2015.
NEW YORK – Consistent with another recently published report but counterintuitive, patients with a high number of nevi prior to developing a melanoma have more favorable prognostic features than do those with fewer moles, according to data presented at the American Academy of Dermatology summer meeting.
Based on differences in numerous clinicopathologic features when patients with more than 50 nevi were compared with those with fewer than 50 nevi, “It is possible that there are different pathways that drive melanoma in these two patient groups, resulting in different degrees of aggressiveness,” reported Dr. Caroline Kim from the Pigmented Lesion Clinic and Cutaneous Oncology Program, Beth Israel Deaconess Medical Center, Harvard Medical School, Boston.
In this study of 281 melanoma patients evaluated over 2 years at Dr. Kim’s clinic, those with a high nevi count (89 patients) were compared with those with a lower nevi count (192 patients). The investigators also performed comparisons on those who had dysplastic versus nondysplastic nevi. Of differences between the groups, the initial melanoma grade was the most surprising.
In the greater-than-50-nevi group, 21.6% had either stage III (18.2%) or stage IV (3.4%) disease at diagnosis vs. 40.2% in the fewer-than-50-nevi group (31.2% and 9.0% with stage III and IV disease, respectively). Conversely, melanoma was diagnosed at stage I in 71.6% in the greater-than-50-nevi group vs. only 41.8% in the fewer-than-50-nevi group.
Among patients in the fewer-than-50-nevi group, ulceration was more common (25.1% vs. 10.3%; P = .017) as was nodular subtype (11.2% vs. 2.9%; P = .035). The group with the lower nevi count also had a higher mitotic rate (3.7 vs. 2.3 count/mm2) and a greater Breslow’s depth (1.78 vs. 1.21 mm; P = .010). Although not significantly different, the proportion of females was higher in the lower nevi count group (56.2% vs. 46.4%) and more patients in this group developed a melanoma in the head or neck (21.6% vs. 15.3%).
There are several potential explanations for the differences observed in this study. One is that the process that drives a high nevi count is different than that which produces lower numbers of nevi. According to Dr. Kim, some of the pathologic features in the greater-than-50-nevi group were more consistent with BRAF mutation patterns than the fewer-than- 50-nevi group. Another is that a high nevi count drives a more robust immune response against melanoma growth. Dr. Kim cited published studies that support both theories.
However, it is also possible that high nevi count increases likelihood of dermatologic care and vigilance for early detection of melanomas. This might be supported by the fact that the age of diagnosis was significantly younger in the greater-than-50-nevi count group (40.1 vs. 50.1 years; P less than .001). However, a younger age at diagnosis could also be a product of different pathways of disease development and growth.
The greater likelihood of more aggressive melanoma in patients with a history of fewer than 50 nevi than in those with greater than 50 nevi has also been supported by another large series that was recently published (Int J Cancer. 2015 Oct 1;137[7]:1691-8.) Cited by Dr. Kim as consistent with her own findings, this study of 2,184 melanoma cases compared 5- and 10-year survival in those who had greater than 50 nevi, which represented 31.3% of this population, to those with fewer. The greater survival at 5 years in the greater-than-50-nevi group (91.2% vs. 86.4%) and 10 years (87.2% vs. 79%) remained significant after adjusting “for all known melanoma prognostic factors,” according to the authors.
Further studies evaluating differences in patterns of melanoma growth in those with fewer than 50 nevi vs. those with more are needed, according to Dr. Kim. She acknowledged that one limitation of her study was that all cases were drawn from a single center. However, there is support for an independent study, and the findings, if verified, “have major implications for public health screening and education.” In particular, she suggested that those with lower nevi counts might deserve the type of attention already being directed at those with high nevi counts “as they may be paradoxically at risk for more aggressive melanomas.”
Dr. Kim reported financial relationships with Hoffman-LaRoche, Castle Biosciences, and Shape Pharmaceuticals.
This article was updated August 26, 2015.
NEW YORK – Consistent with another recently published report but counterintuitive, patients with a high number of nevi prior to developing a melanoma have more favorable prognostic features than do those with fewer moles, according to data presented at the American Academy of Dermatology summer meeting.
Based on differences in numerous clinicopathologic features when patients with more than 50 nevi were compared with those with fewer than 50 nevi, “It is possible that there are different pathways that drive melanoma in these two patient groups, resulting in different degrees of aggressiveness,” reported Dr. Caroline Kim from the Pigmented Lesion Clinic and Cutaneous Oncology Program, Beth Israel Deaconess Medical Center, Harvard Medical School, Boston.
In this study of 281 melanoma patients evaluated over 2 years at Dr. Kim’s clinic, those with a high nevi count (89 patients) were compared with those with a lower nevi count (192 patients). The investigators also performed comparisons on those who had dysplastic versus nondysplastic nevi. Of differences between the groups, the initial melanoma grade was the most surprising.
In the greater-than-50-nevi group, 21.6% had either stage III (18.2%) or stage IV (3.4%) disease at diagnosis vs. 40.2% in the fewer-than-50-nevi group (31.2% and 9.0% with stage III and IV disease, respectively). Conversely, melanoma was diagnosed at stage I in 71.6% in the greater-than-50-nevi group vs. only 41.8% in the fewer-than-50-nevi group.
Among patients in the fewer-than-50-nevi group, ulceration was more common (25.1% vs. 10.3%; P = .017) as was nodular subtype (11.2% vs. 2.9%; P = .035). The group with the lower nevi count also had a higher mitotic rate (3.7 vs. 2.3 count/mm2) and a greater Breslow’s depth (1.78 vs. 1.21 mm; P = .010). Although not significantly different, the proportion of females was higher in the lower nevi count group (56.2% vs. 46.4%) and more patients in this group developed a melanoma in the head or neck (21.6% vs. 15.3%).
There are several potential explanations for the differences observed in this study. One is that the process that drives a high nevi count is different than that which produces lower numbers of nevi. According to Dr. Kim, some of the pathologic features in the greater-than-50-nevi group were more consistent with BRAF mutation patterns than the fewer-than- 50-nevi group. Another is that a high nevi count drives a more robust immune response against melanoma growth. Dr. Kim cited published studies that support both theories.
However, it is also possible that high nevi count increases likelihood of dermatologic care and vigilance for early detection of melanomas. This might be supported by the fact that the age of diagnosis was significantly younger in the greater-than-50-nevi count group (40.1 vs. 50.1 years; P less than .001). However, a younger age at diagnosis could also be a product of different pathways of disease development and growth.
The greater likelihood of more aggressive melanoma in patients with a history of fewer than 50 nevi than in those with greater than 50 nevi has also been supported by another large series that was recently published (Int J Cancer. 2015 Oct 1;137[7]:1691-8.) Cited by Dr. Kim as consistent with her own findings, this study of 2,184 melanoma cases compared 5- and 10-year survival in those who had greater than 50 nevi, which represented 31.3% of this population, to those with fewer. The greater survival at 5 years in the greater-than-50-nevi group (91.2% vs. 86.4%) and 10 years (87.2% vs. 79%) remained significant after adjusting “for all known melanoma prognostic factors,” according to the authors.
Further studies evaluating differences in patterns of melanoma growth in those with fewer than 50 nevi vs. those with more are needed, according to Dr. Kim. She acknowledged that one limitation of her study was that all cases were drawn from a single center. However, there is support for an independent study, and the findings, if verified, “have major implications for public health screening and education.” In particular, she suggested that those with lower nevi counts might deserve the type of attention already being directed at those with high nevi counts “as they may be paradoxically at risk for more aggressive melanomas.”
Dr. Kim reported financial relationships with Hoffman-LaRoche, Castle Biosciences, and Shape Pharmaceuticals.
This article was updated August 26, 2015.
AT THE AAD SUMMER ACADEMY 2015
Key clinical point: In patients with nevi who develop melanoma, malignancies appear to be more aggressive with a lower rather than a higher nevi count.
Major finding: Patients with fewer than 50 moles were more than twice as likely as patients with more than 50 moles to be diagnosed with stage III or IV disease.
Data source: Retrospective chart review.
Disclosures: Dr. Kim reported financial relationships with Hoffman-LaRoche, Castle Biosciences, and Shape Pharmaceuticals.
In skin lymphoma diagnosis, pathology must match clinical presentation
NEW YORK – Both dermatologists and pathologists can make the mistake of diagnosing a long list of pseudolymphomas as lymphomas, according to Dr. Antonio Subtil, who is an associate professor of dermatology and pathology at Yale University School of Medicine, New Haven, Connecticut.
The diagnosis is not trustworthy without agreement by a team of experts, he said. Most dermatologists do not have sufficient expertise in histopathology to interpret equivocal findings. Pathologists do not typically grasp subtleties in dermatologic presentations that may also be critical to reach an accurate diagnosis regardless of histopathology.
Mycosis fungoides (MF) represents almost 60% of cutaneous T-cell lymphomas (CTCL), but only a proportion of patients with MF have a classic presentation and the extensive array of other T-cell and B-cell lesions constitute a heterogeneous profile of lesions, according to Dr. Subtil, who spoke on skin lymphoma knowledge gaps at the summer meeting of the American Academy of Dermatology (AAD). The biases of the dermatologist and the pathologist in isolation can lead to misdiagnosis, which is the reason to insist on a clear correlation between pathologic and clinical findings.
Speaking as a dermatologist, Dr. Subtil cautioned that pathologists “do not understand our terminology” and are not facile in differentiating clinical descriptions of cutaneous lesions. Speaking as a pathologist, Dr. Subtil cautioned that “I cannot trust a pathological report by itself” without understanding the clinical context.
As an example, he recounted a case of an ulcerated nodule that proved to have a lymphoid infiltrate on biopsy. When cells in the infiltrate were found to be CD30+, the likely diagnosis appeared to be an anaplastic large cell lymphoma. Only further testing revealed molluscum contagiosum, which can have a similar appearance and also feature CD30+ cells. A course of unnecessary chemotherapy was narrowly averted.
A small sample of the pseudolymphomas that can fool the clinician include lymphomatoid drug eruption, cutaneous leishmaniasis, lymphomatoid lichenoid keratosis, and reactive lymphoid hyperplasia at the site of vaccination, according to Dr. Subtil. He outlined a series of challenges that should be recognized before settling on a diagnosis.
One of the most important of these challenges is the limited specificity and sensitivity of any clinical characteristic or diagnostic study in isolation, according to Dr. Subtil. Even tissue studies with polymerase chain reaction (PCR) are associated with false positive and false negative results and, like everything else, must be evaluated in the context of the pathological and clinical findings.
Another challenge to a definitive diagnosis is the overuse of the term “atypical” by both dermatologists and pathologists. Dr. Subtil called the propensity to use this label the “atypical syndrome” and cautioned that it may sometimes be an obstacle for seeking a more definitive description of the underlying pathology. Using a team approach, which Dr. Subtil suggested might also include a hematologist and an oncologist, to reach consensus about the diagnosis substantially reduces the risk of a misdiagnosis.
NEW YORK – Both dermatologists and pathologists can make the mistake of diagnosing a long list of pseudolymphomas as lymphomas, according to Dr. Antonio Subtil, who is an associate professor of dermatology and pathology at Yale University School of Medicine, New Haven, Connecticut.
The diagnosis is not trustworthy without agreement by a team of experts, he said. Most dermatologists do not have sufficient expertise in histopathology to interpret equivocal findings. Pathologists do not typically grasp subtleties in dermatologic presentations that may also be critical to reach an accurate diagnosis regardless of histopathology.
Mycosis fungoides (MF) represents almost 60% of cutaneous T-cell lymphomas (CTCL), but only a proportion of patients with MF have a classic presentation and the extensive array of other T-cell and B-cell lesions constitute a heterogeneous profile of lesions, according to Dr. Subtil, who spoke on skin lymphoma knowledge gaps at the summer meeting of the American Academy of Dermatology (AAD). The biases of the dermatologist and the pathologist in isolation can lead to misdiagnosis, which is the reason to insist on a clear correlation between pathologic and clinical findings.
Speaking as a dermatologist, Dr. Subtil cautioned that pathologists “do not understand our terminology” and are not facile in differentiating clinical descriptions of cutaneous lesions. Speaking as a pathologist, Dr. Subtil cautioned that “I cannot trust a pathological report by itself” without understanding the clinical context.
As an example, he recounted a case of an ulcerated nodule that proved to have a lymphoid infiltrate on biopsy. When cells in the infiltrate were found to be CD30+, the likely diagnosis appeared to be an anaplastic large cell lymphoma. Only further testing revealed molluscum contagiosum, which can have a similar appearance and also feature CD30+ cells. A course of unnecessary chemotherapy was narrowly averted.
A small sample of the pseudolymphomas that can fool the clinician include lymphomatoid drug eruption, cutaneous leishmaniasis, lymphomatoid lichenoid keratosis, and reactive lymphoid hyperplasia at the site of vaccination, according to Dr. Subtil. He outlined a series of challenges that should be recognized before settling on a diagnosis.
One of the most important of these challenges is the limited specificity and sensitivity of any clinical characteristic or diagnostic study in isolation, according to Dr. Subtil. Even tissue studies with polymerase chain reaction (PCR) are associated with false positive and false negative results and, like everything else, must be evaluated in the context of the pathological and clinical findings.
Another challenge to a definitive diagnosis is the overuse of the term “atypical” by both dermatologists and pathologists. Dr. Subtil called the propensity to use this label the “atypical syndrome” and cautioned that it may sometimes be an obstacle for seeking a more definitive description of the underlying pathology. Using a team approach, which Dr. Subtil suggested might also include a hematologist and an oncologist, to reach consensus about the diagnosis substantially reduces the risk of a misdiagnosis.
NEW YORK – Both dermatologists and pathologists can make the mistake of diagnosing a long list of pseudolymphomas as lymphomas, according to Dr. Antonio Subtil, who is an associate professor of dermatology and pathology at Yale University School of Medicine, New Haven, Connecticut.
The diagnosis is not trustworthy without agreement by a team of experts, he said. Most dermatologists do not have sufficient expertise in histopathology to interpret equivocal findings. Pathologists do not typically grasp subtleties in dermatologic presentations that may also be critical to reach an accurate diagnosis regardless of histopathology.
Mycosis fungoides (MF) represents almost 60% of cutaneous T-cell lymphomas (CTCL), but only a proportion of patients with MF have a classic presentation and the extensive array of other T-cell and B-cell lesions constitute a heterogeneous profile of lesions, according to Dr. Subtil, who spoke on skin lymphoma knowledge gaps at the summer meeting of the American Academy of Dermatology (AAD). The biases of the dermatologist and the pathologist in isolation can lead to misdiagnosis, which is the reason to insist on a clear correlation between pathologic and clinical findings.
Speaking as a dermatologist, Dr. Subtil cautioned that pathologists “do not understand our terminology” and are not facile in differentiating clinical descriptions of cutaneous lesions. Speaking as a pathologist, Dr. Subtil cautioned that “I cannot trust a pathological report by itself” without understanding the clinical context.
As an example, he recounted a case of an ulcerated nodule that proved to have a lymphoid infiltrate on biopsy. When cells in the infiltrate were found to be CD30+, the likely diagnosis appeared to be an anaplastic large cell lymphoma. Only further testing revealed molluscum contagiosum, which can have a similar appearance and also feature CD30+ cells. A course of unnecessary chemotherapy was narrowly averted.
A small sample of the pseudolymphomas that can fool the clinician include lymphomatoid drug eruption, cutaneous leishmaniasis, lymphomatoid lichenoid keratosis, and reactive lymphoid hyperplasia at the site of vaccination, according to Dr. Subtil. He outlined a series of challenges that should be recognized before settling on a diagnosis.
One of the most important of these challenges is the limited specificity and sensitivity of any clinical characteristic or diagnostic study in isolation, according to Dr. Subtil. Even tissue studies with polymerase chain reaction (PCR) are associated with false positive and false negative results and, like everything else, must be evaluated in the context of the pathological and clinical findings.
Another challenge to a definitive diagnosis is the overuse of the term “atypical” by both dermatologists and pathologists. Dr. Subtil called the propensity to use this label the “atypical syndrome” and cautioned that it may sometimes be an obstacle for seeking a more definitive description of the underlying pathology. Using a team approach, which Dr. Subtil suggested might also include a hematologist and an oncologist, to reach consensus about the diagnosis substantially reduces the risk of a misdiagnosis.
EXPERT ANALYSIS AT THE SUMMER MEETING OF THE AMERICAN ACADEMY OF DERMATOLOGY
Treatment options for lentigo maligna far from perfect
PARK CITY, UTAH – Dr. Glen M. Bowen has been working to improve the surgical treatment of lentigo maligna ever since he joined the Huntsman Cancer Institute at the University of Utah 16 years ago. A retrospective review from Memorial Sloan-Kettering Cancer Center found that on average, 7.1-mm margins are required to remove lentigo maligna (LM) (J. Am. Acad. Dermatol. 2008;58[1]:142-8).
“If you have an LM with a 10-mm diameter to begin with, 7.1-mm margins give you a final surgical diameter of 24.2 mm,” Dr. Bowen said at the annual meeting of the Pacific Dermatologic Association. “These are very morbid surgeries in cosmetically sensitive areas for a relatively low-risk tumor.”
The risk of LM progressing to an invasive melanoma is not known, but is estimated to range between 5% and 33%. Of 2,016 patients treated for LM at the Huntsman Cancer Institute, 522 have been treated with neo-adjuvant topical imiquimod 5% cream followed by a conservative staged excision with 2-mm margins with a recurrence rate of 2.3% during median follow-up of 5 years. Of their recurrences, about 20% recurred with invasion. All recurrences to date have been less than 1 mm in depth (stage IA), which has an estimated mortality risk of 5% at 5 years. “A 5% mortality rate of the 20% that recur with invasion of the 2.3% that recur after surgery yields a mortality risk of 0.023%,” Dr. Bowen said. “Due to the very low risk of actually dying from a recurrent LM, very large and morbid surgical defects strike me as a punishment that doesn’t fit the crime in terms of the cost-benefit ratio.”
He and his associates at the Huntsman Cancer Institute have observed some deaths in patients who presented with LM melanoma (invasive melanoma) but have not observed a single death in patients who presented with LM in situ that subsequently recurred. For these reasons, Dr. Bowen favors pretreating LM with imiquimod 5% cream followed by a conservative staged excision, a process that substantially decreases the size of the surgical defects.
His current treatment protocol involves a five-step process that begins with removing all of the visible lentigo maligna to rule out invasion, since 16% of LMs referred to him have harbored invasion when removed and are upgraded from stage 0 to IA. “I am not going to use a topical cream on an invasive melanoma,” he said. “After an excisional biopsy with minimal margins, I usually close the defect with a purse-string suture because it avoids removing standing cones and consequently enlarging the treatment area.”
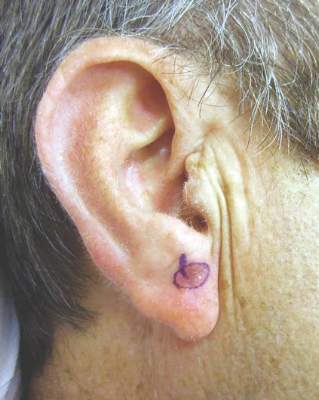
Second, he traces a template of the LM border on transparent plastic and places a tiny tattoo in the center of the biopsy site to enable pinpoint placement of the template at the time of surgery.
Third, he treats the site with imiquimod 5% cream Monday through Friday for 2-3 months and sees the patient monthly for dosage adjustments when needed. The fourth step involves enabling the site to recover for 2-6 months to allow for resolution of the inflammatory infiltrate. The final step involves re-excising around the original template with 2-mm margins for confirmation with the use of a negative control taken from an equally sun-exposed site taken some distance away from the LM. “Caucasians will have atypical junctional melanocytic hyperplasia (AJMH), which must be subtracted out as background,” he said. “Otherwise, if you hold a non–sun-exposed site as your standard for a negative margin, you will never stop cutting.”
Dr. Bowen likes to use frozen radial sections with routine staining with H and E and immunostaining with MART-1 (Melan-A) and SOX-10. Processing takes 2 hours, he continued, “so I put in relaxing sutures, which will stretch out nicely over 2 hours so I can usually close the defects primarily.”
In Dr. Bowen’s opinion, topical imiquimod as monotherapy for LM is not safe, since about 30% of patients treated with imiquimod will still harbor residual LM. “In our dataset, about 70% have no residual LM, 20% have residual LM in the center but negative perimeter margins, and 10% have LM touching a perimeter margin and require a second stage,” he said. “Taken together, 90% of patients pretreated with imiquimod will be cleared in one stage of surgery with 2 mm margins.”
Making the distinction between LM and AJMH common to chronically sun-damaged skin is no easy task. Dr. Bowen cited a concordance study between dermatopathologists interpreting staged excisional margins on permanent sections for LM where the concordance was only moderate at best. In this study, the use of a negative control improved the concordance rate on “difficult” cases from 46% to 76%; P = .001 (Arch Dermatol. 2003 May;139(5):595-604). “What we really need is a molecular marker that will tell us if a melanocyte is malignant or not,” he said. “All we have now are immunostains that tell you if it’s melanocyte but nothing more.” He went on to say that in multivariate analysis in two studies of the histologic features of LM, the only feature that consistently predicted the difference between LM and AJMH was the melanocyte density and its ratio to the negative control (Dermatol Surg. 2011;37(5):657-63 and J. Plast. Reconstr. Aesthet. Surg. 2014;67(10):1322-32). “The MART-1 immunostain is extremely sensitive, but it makes the slide somewhat muddy, so it’s hard to do an accurate cell count,” he said. “For that reason, we also use a SOX-10 immunostain which is very specific but not as sensitive. I believe that the truth lies somewhere in between those two immunostains in light of a positive control from our lab and a negative control from the patient.”
He concluded that the neoadjuvant use of imiquimod followed by a conservative staged excision “allows me to clear 90% of LM with a 2 mm margin with a recurrence rate of 2.3% in patients with a mean follow-up of 5-years or greater.”
Dr. Bowen reported having no financial disclosures.
PARK CITY, UTAH – Dr. Glen M. Bowen has been working to improve the surgical treatment of lentigo maligna ever since he joined the Huntsman Cancer Institute at the University of Utah 16 years ago. A retrospective review from Memorial Sloan-Kettering Cancer Center found that on average, 7.1-mm margins are required to remove lentigo maligna (LM) (J. Am. Acad. Dermatol. 2008;58[1]:142-8).
“If you have an LM with a 10-mm diameter to begin with, 7.1-mm margins give you a final surgical diameter of 24.2 mm,” Dr. Bowen said at the annual meeting of the Pacific Dermatologic Association. “These are very morbid surgeries in cosmetically sensitive areas for a relatively low-risk tumor.”
The risk of LM progressing to an invasive melanoma is not known, but is estimated to range between 5% and 33%. Of 2,016 patients treated for LM at the Huntsman Cancer Institute, 522 have been treated with neo-adjuvant topical imiquimod 5% cream followed by a conservative staged excision with 2-mm margins with a recurrence rate of 2.3% during median follow-up of 5 years. Of their recurrences, about 20% recurred with invasion. All recurrences to date have been less than 1 mm in depth (stage IA), which has an estimated mortality risk of 5% at 5 years. “A 5% mortality rate of the 20% that recur with invasion of the 2.3% that recur after surgery yields a mortality risk of 0.023%,” Dr. Bowen said. “Due to the very low risk of actually dying from a recurrent LM, very large and morbid surgical defects strike me as a punishment that doesn’t fit the crime in terms of the cost-benefit ratio.”
He and his associates at the Huntsman Cancer Institute have observed some deaths in patients who presented with LM melanoma (invasive melanoma) but have not observed a single death in patients who presented with LM in situ that subsequently recurred. For these reasons, Dr. Bowen favors pretreating LM with imiquimod 5% cream followed by a conservative staged excision, a process that substantially decreases the size of the surgical defects.
His current treatment protocol involves a five-step process that begins with removing all of the visible lentigo maligna to rule out invasion, since 16% of LMs referred to him have harbored invasion when removed and are upgraded from stage 0 to IA. “I am not going to use a topical cream on an invasive melanoma,” he said. “After an excisional biopsy with minimal margins, I usually close the defect with a purse-string suture because it avoids removing standing cones and consequently enlarging the treatment area.”
Second, he traces a template of the LM border on transparent plastic and places a tiny tattoo in the center of the biopsy site to enable pinpoint placement of the template at the time of surgery.
Third, he treats the site with imiquimod 5% cream Monday through Friday for 2-3 months and sees the patient monthly for dosage adjustments when needed. The fourth step involves enabling the site to recover for 2-6 months to allow for resolution of the inflammatory infiltrate. The final step involves re-excising around the original template with 2-mm margins for confirmation with the use of a negative control taken from an equally sun-exposed site taken some distance away from the LM. “Caucasians will have atypical junctional melanocytic hyperplasia (AJMH), which must be subtracted out as background,” he said. “Otherwise, if you hold a non–sun-exposed site as your standard for a negative margin, you will never stop cutting.”
Dr. Bowen likes to use frozen radial sections with routine staining with H and E and immunostaining with MART-1 (Melan-A) and SOX-10. Processing takes 2 hours, he continued, “so I put in relaxing sutures, which will stretch out nicely over 2 hours so I can usually close the defects primarily.”
In Dr. Bowen’s opinion, topical imiquimod as monotherapy for LM is not safe, since about 30% of patients treated with imiquimod will still harbor residual LM. “In our dataset, about 70% have no residual LM, 20% have residual LM in the center but negative perimeter margins, and 10% have LM touching a perimeter margin and require a second stage,” he said. “Taken together, 90% of patients pretreated with imiquimod will be cleared in one stage of surgery with 2 mm margins.”
Making the distinction between LM and AJMH common to chronically sun-damaged skin is no easy task. Dr. Bowen cited a concordance study between dermatopathologists interpreting staged excisional margins on permanent sections for LM where the concordance was only moderate at best. In this study, the use of a negative control improved the concordance rate on “difficult” cases from 46% to 76%; P = .001 (Arch Dermatol. 2003 May;139(5):595-604). “What we really need is a molecular marker that will tell us if a melanocyte is malignant or not,” he said. “All we have now are immunostains that tell you if it’s melanocyte but nothing more.” He went on to say that in multivariate analysis in two studies of the histologic features of LM, the only feature that consistently predicted the difference between LM and AJMH was the melanocyte density and its ratio to the negative control (Dermatol Surg. 2011;37(5):657-63 and J. Plast. Reconstr. Aesthet. Surg. 2014;67(10):1322-32). “The MART-1 immunostain is extremely sensitive, but it makes the slide somewhat muddy, so it’s hard to do an accurate cell count,” he said. “For that reason, we also use a SOX-10 immunostain which is very specific but not as sensitive. I believe that the truth lies somewhere in between those two immunostains in light of a positive control from our lab and a negative control from the patient.”
He concluded that the neoadjuvant use of imiquimod followed by a conservative staged excision “allows me to clear 90% of LM with a 2 mm margin with a recurrence rate of 2.3% in patients with a mean follow-up of 5-years or greater.”
Dr. Bowen reported having no financial disclosures.
PARK CITY, UTAH – Dr. Glen M. Bowen has been working to improve the surgical treatment of lentigo maligna ever since he joined the Huntsman Cancer Institute at the University of Utah 16 years ago. A retrospective review from Memorial Sloan-Kettering Cancer Center found that on average, 7.1-mm margins are required to remove lentigo maligna (LM) (J. Am. Acad. Dermatol. 2008;58[1]:142-8).
“If you have an LM with a 10-mm diameter to begin with, 7.1-mm margins give you a final surgical diameter of 24.2 mm,” Dr. Bowen said at the annual meeting of the Pacific Dermatologic Association. “These are very morbid surgeries in cosmetically sensitive areas for a relatively low-risk tumor.”
The risk of LM progressing to an invasive melanoma is not known, but is estimated to range between 5% and 33%. Of 2,016 patients treated for LM at the Huntsman Cancer Institute, 522 have been treated with neo-adjuvant topical imiquimod 5% cream followed by a conservative staged excision with 2-mm margins with a recurrence rate of 2.3% during median follow-up of 5 years. Of their recurrences, about 20% recurred with invasion. All recurrences to date have been less than 1 mm in depth (stage IA), which has an estimated mortality risk of 5% at 5 years. “A 5% mortality rate of the 20% that recur with invasion of the 2.3% that recur after surgery yields a mortality risk of 0.023%,” Dr. Bowen said. “Due to the very low risk of actually dying from a recurrent LM, very large and morbid surgical defects strike me as a punishment that doesn’t fit the crime in terms of the cost-benefit ratio.”
He and his associates at the Huntsman Cancer Institute have observed some deaths in patients who presented with LM melanoma (invasive melanoma) but have not observed a single death in patients who presented with LM in situ that subsequently recurred. For these reasons, Dr. Bowen favors pretreating LM with imiquimod 5% cream followed by a conservative staged excision, a process that substantially decreases the size of the surgical defects.
His current treatment protocol involves a five-step process that begins with removing all of the visible lentigo maligna to rule out invasion, since 16% of LMs referred to him have harbored invasion when removed and are upgraded from stage 0 to IA. “I am not going to use a topical cream on an invasive melanoma,” he said. “After an excisional biopsy with minimal margins, I usually close the defect with a purse-string suture because it avoids removing standing cones and consequently enlarging the treatment area.”
Second, he traces a template of the LM border on transparent plastic and places a tiny tattoo in the center of the biopsy site to enable pinpoint placement of the template at the time of surgery.
Third, he treats the site with imiquimod 5% cream Monday through Friday for 2-3 months and sees the patient monthly for dosage adjustments when needed. The fourth step involves enabling the site to recover for 2-6 months to allow for resolution of the inflammatory infiltrate. The final step involves re-excising around the original template with 2-mm margins for confirmation with the use of a negative control taken from an equally sun-exposed site taken some distance away from the LM. “Caucasians will have atypical junctional melanocytic hyperplasia (AJMH), which must be subtracted out as background,” he said. “Otherwise, if you hold a non–sun-exposed site as your standard for a negative margin, you will never stop cutting.”
Dr. Bowen likes to use frozen radial sections with routine staining with H and E and immunostaining with MART-1 (Melan-A) and SOX-10. Processing takes 2 hours, he continued, “so I put in relaxing sutures, which will stretch out nicely over 2 hours so I can usually close the defects primarily.”
In Dr. Bowen’s opinion, topical imiquimod as monotherapy for LM is not safe, since about 30% of patients treated with imiquimod will still harbor residual LM. “In our dataset, about 70% have no residual LM, 20% have residual LM in the center but negative perimeter margins, and 10% have LM touching a perimeter margin and require a second stage,” he said. “Taken together, 90% of patients pretreated with imiquimod will be cleared in one stage of surgery with 2 mm margins.”
Making the distinction between LM and AJMH common to chronically sun-damaged skin is no easy task. Dr. Bowen cited a concordance study between dermatopathologists interpreting staged excisional margins on permanent sections for LM where the concordance was only moderate at best. In this study, the use of a negative control improved the concordance rate on “difficult” cases from 46% to 76%; P = .001 (Arch Dermatol. 2003 May;139(5):595-604). “What we really need is a molecular marker that will tell us if a melanocyte is malignant or not,” he said. “All we have now are immunostains that tell you if it’s melanocyte but nothing more.” He went on to say that in multivariate analysis in two studies of the histologic features of LM, the only feature that consistently predicted the difference between LM and AJMH was the melanocyte density and its ratio to the negative control (Dermatol Surg. 2011;37(5):657-63 and J. Plast. Reconstr. Aesthet. Surg. 2014;67(10):1322-32). “The MART-1 immunostain is extremely sensitive, but it makes the slide somewhat muddy, so it’s hard to do an accurate cell count,” he said. “For that reason, we also use a SOX-10 immunostain which is very specific but not as sensitive. I believe that the truth lies somewhere in between those two immunostains in light of a positive control from our lab and a negative control from the patient.”
He concluded that the neoadjuvant use of imiquimod followed by a conservative staged excision “allows me to clear 90% of LM with a 2 mm margin with a recurrence rate of 2.3% in patients with a mean follow-up of 5-years or greater.”
Dr. Bowen reported having no financial disclosures.
EXPERT ANALYSIS from PDA 2015
Ingenol mebutate helped clear actinic keratoses
Cryosurgery followed by topical ingenol mebutate cleared extensive regions of actinic keratosis, which helped reveal residual squamous cell carcinomas, according to a report in the August issue of the Journal of Drugs in Dermatology.
The findings show that ingenol mebutate can clear multiple AKs and reduce the number of scarring biopsies required to identify SCCs, said Dr. Miriam S. Bettencourt, a dermatologist in group practice in Henderson, Nev. “In our dermatology clinic, many of the patients with a long history of AK who were treated with ingenol mebutate used sequentially after cryosurgery have achieved complete or partial clearance of AKs.”
Ingenol mebutate gel after cryosurgery cleared AKs more effectively than cryosurgery alone in a recent phase III trial (J Drugs Dermatol. 2014 Jun;13[6]741-7), Dr. Bettencourt noted. She described six men and one woman who each had at least 10 recurrent or hyperkeratotic AKs and previously had undergone cryosurgery. She treated all patients with cryosurgery, followed 2 weeks later by two or three once-daily applications of ingenol mebutate gel at strengths of 0.05% or 0.015%, respectively (J Drugs Dermatol. 2015 Aug;14[8];813-8). One course of ingenol mebutate gel cleared 50%-100% of AKs, Dr. Bettencourt said. She treated residual AKs with cryosurgery, and five patients also received at least one more course of ingenol mebutate to re-treat a partially cleared area or to treat a separate area. Shave biopsies of 10 residual suspicious lesions taken 3-8 months later all revealed invasive SCCs, which were treated with Mohs micrographic surgery (MMS). “These lesions may have been preexisting at the time of topical treatment but not readily recognized as suspicious in the heavily actinically damaged skin, in which suspected or small SCCs may be adjacent to or obscured by AKs,” she said. “Alternatively, these tumors may have been spontaneous new SCCs. In either case, we suggest that effective clearance of AKs from the palette of sun-damaged skin with ingenol mebutate permitted prompt recognition of these lesions as suspicious, and led to further diagnosis and treatment with MMS.”
All patients developed mild to moderate localized redness, flaking, and crusting starting on the second day of ingenol mebutate treatment and resolving within a week of finishing the course, Dr. Bettencourt said.
She reported that she had no relevant financial conflicts.
Cryosurgery followed by topical ingenol mebutate cleared extensive regions of actinic keratosis, which helped reveal residual squamous cell carcinomas, according to a report in the August issue of the Journal of Drugs in Dermatology.
The findings show that ingenol mebutate can clear multiple AKs and reduce the number of scarring biopsies required to identify SCCs, said Dr. Miriam S. Bettencourt, a dermatologist in group practice in Henderson, Nev. “In our dermatology clinic, many of the patients with a long history of AK who were treated with ingenol mebutate used sequentially after cryosurgery have achieved complete or partial clearance of AKs.”
Ingenol mebutate gel after cryosurgery cleared AKs more effectively than cryosurgery alone in a recent phase III trial (J Drugs Dermatol. 2014 Jun;13[6]741-7), Dr. Bettencourt noted. She described six men and one woman who each had at least 10 recurrent or hyperkeratotic AKs and previously had undergone cryosurgery. She treated all patients with cryosurgery, followed 2 weeks later by two or three once-daily applications of ingenol mebutate gel at strengths of 0.05% or 0.015%, respectively (J Drugs Dermatol. 2015 Aug;14[8];813-8). One course of ingenol mebutate gel cleared 50%-100% of AKs, Dr. Bettencourt said. She treated residual AKs with cryosurgery, and five patients also received at least one more course of ingenol mebutate to re-treat a partially cleared area or to treat a separate area. Shave biopsies of 10 residual suspicious lesions taken 3-8 months later all revealed invasive SCCs, which were treated with Mohs micrographic surgery (MMS). “These lesions may have been preexisting at the time of topical treatment but not readily recognized as suspicious in the heavily actinically damaged skin, in which suspected or small SCCs may be adjacent to or obscured by AKs,” she said. “Alternatively, these tumors may have been spontaneous new SCCs. In either case, we suggest that effective clearance of AKs from the palette of sun-damaged skin with ingenol mebutate permitted prompt recognition of these lesions as suspicious, and led to further diagnosis and treatment with MMS.”
All patients developed mild to moderate localized redness, flaking, and crusting starting on the second day of ingenol mebutate treatment and resolving within a week of finishing the course, Dr. Bettencourt said.
She reported that she had no relevant financial conflicts.
Cryosurgery followed by topical ingenol mebutate cleared extensive regions of actinic keratosis, which helped reveal residual squamous cell carcinomas, according to a report in the August issue of the Journal of Drugs in Dermatology.
The findings show that ingenol mebutate can clear multiple AKs and reduce the number of scarring biopsies required to identify SCCs, said Dr. Miriam S. Bettencourt, a dermatologist in group practice in Henderson, Nev. “In our dermatology clinic, many of the patients with a long history of AK who were treated with ingenol mebutate used sequentially after cryosurgery have achieved complete or partial clearance of AKs.”
Ingenol mebutate gel after cryosurgery cleared AKs more effectively than cryosurgery alone in a recent phase III trial (J Drugs Dermatol. 2014 Jun;13[6]741-7), Dr. Bettencourt noted. She described six men and one woman who each had at least 10 recurrent or hyperkeratotic AKs and previously had undergone cryosurgery. She treated all patients with cryosurgery, followed 2 weeks later by two or three once-daily applications of ingenol mebutate gel at strengths of 0.05% or 0.015%, respectively (J Drugs Dermatol. 2015 Aug;14[8];813-8). One course of ingenol mebutate gel cleared 50%-100% of AKs, Dr. Bettencourt said. She treated residual AKs with cryosurgery, and five patients also received at least one more course of ingenol mebutate to re-treat a partially cleared area or to treat a separate area. Shave biopsies of 10 residual suspicious lesions taken 3-8 months later all revealed invasive SCCs, which were treated with Mohs micrographic surgery (MMS). “These lesions may have been preexisting at the time of topical treatment but not readily recognized as suspicious in the heavily actinically damaged skin, in which suspected or small SCCs may be adjacent to or obscured by AKs,” she said. “Alternatively, these tumors may have been spontaneous new SCCs. In either case, we suggest that effective clearance of AKs from the palette of sun-damaged skin with ingenol mebutate permitted prompt recognition of these lesions as suspicious, and led to further diagnosis and treatment with MMS.”
All patients developed mild to moderate localized redness, flaking, and crusting starting on the second day of ingenol mebutate treatment and resolving within a week of finishing the course, Dr. Bettencourt said.
She reported that she had no relevant financial conflicts.
FROM THE JOURNAL OF DRUGS IN DERMATOLOGY
Key clinical point: Several courses of cryosurgery and ingenol mebutate helped clear actinic keratoses, helping a clinician identify residual squamous cell carcinomas.
Major finding: Lesion counts dropped by 50%-100% after cryosurgery followed by one to three courses of ingenol mebutate gel.
Data source: A case series of seven patients who had multiple AKs and 10 SCCs.
Disclosures: Dr. Bettencourt reported that she had no relevant financial conflicts.
Immune-related patterns of response present challenges
With the race to develop cancer immunotherapies escalating and new agents appearing in the clinic, oncologists’ decision-making toolbox will need to evolve.
“As immunotherapeutics become increasingly available to patients, clinicians face a major challenge in the evaluation of these novel drugs – the accurate determination of clinical efficacy,” physician-scientists recently wrote in a commentary published online in the Journal of Clinical Oncology.
Response evaluation criteria in solid tumors (RECIST) have typically driven oncologists’ decision making. Patients undergo scans and radiographic measurements to determine the extent of change in tumor size. Scan, treat, repeat is a mantra for advanced cancer patients, so much so some patients have sought to trademark the phrase for T-shirts. And significant tumor growth has traditionally signaled treatment failure.
But although some patients have responded to immune-targeted treatment with tumor shrinkage or stable disease that would be consistent with existing RECIST criteria, distinct immune-related patterns of response are also emerging, including pseudoprogression, said Dr. Victoria L. Chiou and Dr. Mauricio Burotto, both medical oncology fellows at the National Cancer Institute, in their commentary (Jour Clin Onc. 2015 Aug 10. doi: 10.1200/JCO.2015.61.6870).
Investigators first addressed this issue by proposing immune response criteria in 2009, based on data from ipilimumab phase II trials in patients with advanced melanoma. Dr. Jedd D. Wolchok of Memorial Sloan Kettering Cancer Center, New York, and his associates expanded on RECIST by adding two additional patterns of response: response after an increase in total tumor burden and response in the presence of new lesions. All patterns of response were associated with favorable survival, Dr. Wolchok and associates said (Clin Cancer Res 2009 Dec 1. doi: 10.1158/1078-0432.CCR-09-1624).
Subsequent trials in patients with advanced melanoma have confirmed that a subset of patients who are responders using immune response criteria would have been misclassified using RECIST alone.
In a study of patients with metastatic melanoma treated with nivolumab, 10% experienced distinct immune-related responses, according to Dr. Chiou and Dr. Burotto. In another study of patients with advanced melanoma, this one evaluating anti–PD-1 monoclonal antibody pembrolizumab, 6.7% of patients of the patients experienced pseudoprogression, and overall, 12% of patients were classified as responders or as having stable disease by immune response criteria but would have been classified as having progressive disease by RECIST.
It is time to expand on those first immune response criteria, Dr. Chiou and Dr. Burotto conclude. “Five years after the introduction of the immune response criteria, it is necessary to fully characterize the patterns of immune-related phenomena, to understand these patterns across multiple solid tumor types, and to evaluate how these guidelines are used in current clinical practice,” they wrote.
The Food and Drug Administration agrees. Though guidelines for industry were published in 2011 regarding cancer vaccines, there is no specific guidance on approval criteria for other cancer immunotherapies such as checkpoint inhibitors, Dr. Gideon Blumenthal, team leader in the FDA’s Office of Hematology and Oncology Products, Center for Drug Evaluation and Research, said in an interview.
“However, the FDA is actively looking at other metrics of response beyond conventional RECIST criteria, both to help industry in early ‘go/no-go’ decision making, as well as for helping in the design of later stages of trial design and to help inform approval decisions,” he said.
The FDA is interested in investigating internal data sets of immunotherapy trials, and in collaborating with external stakeholders to determine what the optimal endpoints for cancer immunotherapies will be. Median PFS and PFS hazard ratios do not appear to capture the overall survival benefit that patients derive from immunotherapies, particularly in early lung cancer trials with which he is most familiar, Dr. Blumenthal said.
“Overall survival remains the gold standard endpoint in oncology as it is a direct measure of clinical benefit and less subject to bias, but as more effective therapies come on line and patients live longer, overall survival will become more challenging to detect,” he said.
lnikolaides@frontlinemedcom.com
On Twitter @NikolaidesLaura
With the race to develop cancer immunotherapies escalating and new agents appearing in the clinic, oncologists’ decision-making toolbox will need to evolve.
“As immunotherapeutics become increasingly available to patients, clinicians face a major challenge in the evaluation of these novel drugs – the accurate determination of clinical efficacy,” physician-scientists recently wrote in a commentary published online in the Journal of Clinical Oncology.
Response evaluation criteria in solid tumors (RECIST) have typically driven oncologists’ decision making. Patients undergo scans and radiographic measurements to determine the extent of change in tumor size. Scan, treat, repeat is a mantra for advanced cancer patients, so much so some patients have sought to trademark the phrase for T-shirts. And significant tumor growth has traditionally signaled treatment failure.
But although some patients have responded to immune-targeted treatment with tumor shrinkage or stable disease that would be consistent with existing RECIST criteria, distinct immune-related patterns of response are also emerging, including pseudoprogression, said Dr. Victoria L. Chiou and Dr. Mauricio Burotto, both medical oncology fellows at the National Cancer Institute, in their commentary (Jour Clin Onc. 2015 Aug 10. doi: 10.1200/JCO.2015.61.6870).
Investigators first addressed this issue by proposing immune response criteria in 2009, based on data from ipilimumab phase II trials in patients with advanced melanoma. Dr. Jedd D. Wolchok of Memorial Sloan Kettering Cancer Center, New York, and his associates expanded on RECIST by adding two additional patterns of response: response after an increase in total tumor burden and response in the presence of new lesions. All patterns of response were associated with favorable survival, Dr. Wolchok and associates said (Clin Cancer Res 2009 Dec 1. doi: 10.1158/1078-0432.CCR-09-1624).
Subsequent trials in patients with advanced melanoma have confirmed that a subset of patients who are responders using immune response criteria would have been misclassified using RECIST alone.
In a study of patients with metastatic melanoma treated with nivolumab, 10% experienced distinct immune-related responses, according to Dr. Chiou and Dr. Burotto. In another study of patients with advanced melanoma, this one evaluating anti–PD-1 monoclonal antibody pembrolizumab, 6.7% of patients of the patients experienced pseudoprogression, and overall, 12% of patients were classified as responders or as having stable disease by immune response criteria but would have been classified as having progressive disease by RECIST.
It is time to expand on those first immune response criteria, Dr. Chiou and Dr. Burotto conclude. “Five years after the introduction of the immune response criteria, it is necessary to fully characterize the patterns of immune-related phenomena, to understand these patterns across multiple solid tumor types, and to evaluate how these guidelines are used in current clinical practice,” they wrote.
The Food and Drug Administration agrees. Though guidelines for industry were published in 2011 regarding cancer vaccines, there is no specific guidance on approval criteria for other cancer immunotherapies such as checkpoint inhibitors, Dr. Gideon Blumenthal, team leader in the FDA’s Office of Hematology and Oncology Products, Center for Drug Evaluation and Research, said in an interview.
“However, the FDA is actively looking at other metrics of response beyond conventional RECIST criteria, both to help industry in early ‘go/no-go’ decision making, as well as for helping in the design of later stages of trial design and to help inform approval decisions,” he said.
The FDA is interested in investigating internal data sets of immunotherapy trials, and in collaborating with external stakeholders to determine what the optimal endpoints for cancer immunotherapies will be. Median PFS and PFS hazard ratios do not appear to capture the overall survival benefit that patients derive from immunotherapies, particularly in early lung cancer trials with which he is most familiar, Dr. Blumenthal said.
“Overall survival remains the gold standard endpoint in oncology as it is a direct measure of clinical benefit and less subject to bias, but as more effective therapies come on line and patients live longer, overall survival will become more challenging to detect,” he said.
lnikolaides@frontlinemedcom.com
On Twitter @NikolaidesLaura
With the race to develop cancer immunotherapies escalating and new agents appearing in the clinic, oncologists’ decision-making toolbox will need to evolve.
“As immunotherapeutics become increasingly available to patients, clinicians face a major challenge in the evaluation of these novel drugs – the accurate determination of clinical efficacy,” physician-scientists recently wrote in a commentary published online in the Journal of Clinical Oncology.
Response evaluation criteria in solid tumors (RECIST) have typically driven oncologists’ decision making. Patients undergo scans and radiographic measurements to determine the extent of change in tumor size. Scan, treat, repeat is a mantra for advanced cancer patients, so much so some patients have sought to trademark the phrase for T-shirts. And significant tumor growth has traditionally signaled treatment failure.
But although some patients have responded to immune-targeted treatment with tumor shrinkage or stable disease that would be consistent with existing RECIST criteria, distinct immune-related patterns of response are also emerging, including pseudoprogression, said Dr. Victoria L. Chiou and Dr. Mauricio Burotto, both medical oncology fellows at the National Cancer Institute, in their commentary (Jour Clin Onc. 2015 Aug 10. doi: 10.1200/JCO.2015.61.6870).
Investigators first addressed this issue by proposing immune response criteria in 2009, based on data from ipilimumab phase II trials in patients with advanced melanoma. Dr. Jedd D. Wolchok of Memorial Sloan Kettering Cancer Center, New York, and his associates expanded on RECIST by adding two additional patterns of response: response after an increase in total tumor burden and response in the presence of new lesions. All patterns of response were associated with favorable survival, Dr. Wolchok and associates said (Clin Cancer Res 2009 Dec 1. doi: 10.1158/1078-0432.CCR-09-1624).
Subsequent trials in patients with advanced melanoma have confirmed that a subset of patients who are responders using immune response criteria would have been misclassified using RECIST alone.
In a study of patients with metastatic melanoma treated with nivolumab, 10% experienced distinct immune-related responses, according to Dr. Chiou and Dr. Burotto. In another study of patients with advanced melanoma, this one evaluating anti–PD-1 monoclonal antibody pembrolizumab, 6.7% of patients of the patients experienced pseudoprogression, and overall, 12% of patients were classified as responders or as having stable disease by immune response criteria but would have been classified as having progressive disease by RECIST.
It is time to expand on those first immune response criteria, Dr. Chiou and Dr. Burotto conclude. “Five years after the introduction of the immune response criteria, it is necessary to fully characterize the patterns of immune-related phenomena, to understand these patterns across multiple solid tumor types, and to evaluate how these guidelines are used in current clinical practice,” they wrote.
The Food and Drug Administration agrees. Though guidelines for industry were published in 2011 regarding cancer vaccines, there is no specific guidance on approval criteria for other cancer immunotherapies such as checkpoint inhibitors, Dr. Gideon Blumenthal, team leader in the FDA’s Office of Hematology and Oncology Products, Center for Drug Evaluation and Research, said in an interview.
“However, the FDA is actively looking at other metrics of response beyond conventional RECIST criteria, both to help industry in early ‘go/no-go’ decision making, as well as for helping in the design of later stages of trial design and to help inform approval decisions,” he said.
The FDA is interested in investigating internal data sets of immunotherapy trials, and in collaborating with external stakeholders to determine what the optimal endpoints for cancer immunotherapies will be. Median PFS and PFS hazard ratios do not appear to capture the overall survival benefit that patients derive from immunotherapies, particularly in early lung cancer trials with which he is most familiar, Dr. Blumenthal said.
“Overall survival remains the gold standard endpoint in oncology as it is a direct measure of clinical benefit and less subject to bias, but as more effective therapies come on line and patients live longer, overall survival will become more challenging to detect,” he said.
lnikolaides@frontlinemedcom.com
On Twitter @NikolaidesLaura
Indoor Tanning Is More Harmful Than Americans Believe
The Surgeon General has called on partners in prevention from various sectors to address skin cancer as a major public health problem. One of the main goals outlined in The Surgeon General’s Call to Action to Prevent Skin Cancer is to reduce harm from indoor tanning, which has been linked to increased risk for skin cancer, including melanoma, basal cell carcinoma, and squamous cell carcinoma.
Based on reports from the American Cancer Society, Centers for Disease Control and Prevention, Federal Trade Commission, Mayo Clinic, and US Food and Drug Administration, the following common myths about indoor tanning should be communicated to dermatology patients.
Myth: Indoor tanning will not increase your risk for skin cancer.
Fact: As many as 90% of melanomas are caused by UV exposure. Indoor tanning exposure to UVA and UVB radiation damages the skin and may lead to cancer. Melanoma is linked to severe sunburns, especially at a young age.
Myth: Indoor tanning is safer than tanning outdoors because it is a controlled dose of UV radiation.
Fact: Both indoor tanning and tanning outside are dangerous. Tanning beds may be more dangerous than the sun because they can be used at the same high intensity every day of the year, regardless of time of day, season, or cloud cover. Furthermore, the Surgeon General and US Food and Drug Administration report that an estimated 3000 Americans each year go to emergency departments with injuries caused by indoor tanning, including burns, eye injuries, immune suppression, and allergic reactions. Indoor tanning also causes premature skin aging.
Myth: A “base tan” protects your skin from sunburn.
Fact: Although many patients believe that a few sessions of indoor tanning will prevent them from burning in the sun, a tan does little to protect the skin from future UV exposure. In fact, the Centers for Disease Control and Prevention notes that people who tan indoors are more likely to report getting sunburned. The best way to protect the skin from sunburn is by using sun protection and avoiding indoor tanning.
Myth: Indoor tanning is a safe way to increase vitamin D levels.
Fact: It is important to get enough vitamin D; however, the safest way is through what you eat. Although UVB radiation helps the body produce vitamin D, patients do not need a tan to get that benefit. Ten to 15 minutes of unprotected natural sun exposure on the face and hands 2 to 3 times a week during the summer allows for a healthy dose of vitamin D. Dietary sources, such as low-fat milk, salmon, tuna, and fortified orange juice, are the safest way to get enough vitamin D.
Myth: Indoor tanning is approved by the government.
Fact: According to the Federal Trade Commission, no US government agency recommends the use of indoor tanning equipment. Tanning bed use by minors has been banned in many states, and efforts are ongoing to protect consumers younger than 18 years on local, state, and federal levels. In July 2009, the International Agency for Research on Cancer, part of the World Health Organization, moved tanning devices that emit UV radiation into the highest cancer risk category—carcinogenic to humans—concluding that they are more dangerous than previously thought.
Studies have consistently shown that indoor tanning increases a person’s risk of getting skin cancer and indoor tanning at a young age appears to be more strongly related to lifetime skin cancer risk. Patients should be reminded that every time they tan, they increase their risk of melanoma as well as premature skin aging and other skin cancers. Dermatologists should counsel patients on using sun protection and avoiding indoor tanning.
The Surgeon General has called on partners in prevention from various sectors to address skin cancer as a major public health problem. One of the main goals outlined in The Surgeon General’s Call to Action to Prevent Skin Cancer is to reduce harm from indoor tanning, which has been linked to increased risk for skin cancer, including melanoma, basal cell carcinoma, and squamous cell carcinoma.
Based on reports from the American Cancer Society, Centers for Disease Control and Prevention, Federal Trade Commission, Mayo Clinic, and US Food and Drug Administration, the following common myths about indoor tanning should be communicated to dermatology patients.
Myth: Indoor tanning will not increase your risk for skin cancer.
Fact: As many as 90% of melanomas are caused by UV exposure. Indoor tanning exposure to UVA and UVB radiation damages the skin and may lead to cancer. Melanoma is linked to severe sunburns, especially at a young age.
Myth: Indoor tanning is safer than tanning outdoors because it is a controlled dose of UV radiation.
Fact: Both indoor tanning and tanning outside are dangerous. Tanning beds may be more dangerous than the sun because they can be used at the same high intensity every day of the year, regardless of time of day, season, or cloud cover. Furthermore, the Surgeon General and US Food and Drug Administration report that an estimated 3000 Americans each year go to emergency departments with injuries caused by indoor tanning, including burns, eye injuries, immune suppression, and allergic reactions. Indoor tanning also causes premature skin aging.
Myth: A “base tan” protects your skin from sunburn.
Fact: Although many patients believe that a few sessions of indoor tanning will prevent them from burning in the sun, a tan does little to protect the skin from future UV exposure. In fact, the Centers for Disease Control and Prevention notes that people who tan indoors are more likely to report getting sunburned. The best way to protect the skin from sunburn is by using sun protection and avoiding indoor tanning.
Myth: Indoor tanning is a safe way to increase vitamin D levels.
Fact: It is important to get enough vitamin D; however, the safest way is through what you eat. Although UVB radiation helps the body produce vitamin D, patients do not need a tan to get that benefit. Ten to 15 minutes of unprotected natural sun exposure on the face and hands 2 to 3 times a week during the summer allows for a healthy dose of vitamin D. Dietary sources, such as low-fat milk, salmon, tuna, and fortified orange juice, are the safest way to get enough vitamin D.
Myth: Indoor tanning is approved by the government.
Fact: According to the Federal Trade Commission, no US government agency recommends the use of indoor tanning equipment. Tanning bed use by minors has been banned in many states, and efforts are ongoing to protect consumers younger than 18 years on local, state, and federal levels. In July 2009, the International Agency for Research on Cancer, part of the World Health Organization, moved tanning devices that emit UV radiation into the highest cancer risk category—carcinogenic to humans—concluding that they are more dangerous than previously thought.
Studies have consistently shown that indoor tanning increases a person’s risk of getting skin cancer and indoor tanning at a young age appears to be more strongly related to lifetime skin cancer risk. Patients should be reminded that every time they tan, they increase their risk of melanoma as well as premature skin aging and other skin cancers. Dermatologists should counsel patients on using sun protection and avoiding indoor tanning.
The Surgeon General has called on partners in prevention from various sectors to address skin cancer as a major public health problem. One of the main goals outlined in The Surgeon General’s Call to Action to Prevent Skin Cancer is to reduce harm from indoor tanning, which has been linked to increased risk for skin cancer, including melanoma, basal cell carcinoma, and squamous cell carcinoma.
Based on reports from the American Cancer Society, Centers for Disease Control and Prevention, Federal Trade Commission, Mayo Clinic, and US Food and Drug Administration, the following common myths about indoor tanning should be communicated to dermatology patients.
Myth: Indoor tanning will not increase your risk for skin cancer.
Fact: As many as 90% of melanomas are caused by UV exposure. Indoor tanning exposure to UVA and UVB radiation damages the skin and may lead to cancer. Melanoma is linked to severe sunburns, especially at a young age.
Myth: Indoor tanning is safer than tanning outdoors because it is a controlled dose of UV radiation.
Fact: Both indoor tanning and tanning outside are dangerous. Tanning beds may be more dangerous than the sun because they can be used at the same high intensity every day of the year, regardless of time of day, season, or cloud cover. Furthermore, the Surgeon General and US Food and Drug Administration report that an estimated 3000 Americans each year go to emergency departments with injuries caused by indoor tanning, including burns, eye injuries, immune suppression, and allergic reactions. Indoor tanning also causes premature skin aging.
Myth: A “base tan” protects your skin from sunburn.
Fact: Although many patients believe that a few sessions of indoor tanning will prevent them from burning in the sun, a tan does little to protect the skin from future UV exposure. In fact, the Centers for Disease Control and Prevention notes that people who tan indoors are more likely to report getting sunburned. The best way to protect the skin from sunburn is by using sun protection and avoiding indoor tanning.
Myth: Indoor tanning is a safe way to increase vitamin D levels.
Fact: It is important to get enough vitamin D; however, the safest way is through what you eat. Although UVB radiation helps the body produce vitamin D, patients do not need a tan to get that benefit. Ten to 15 minutes of unprotected natural sun exposure on the face and hands 2 to 3 times a week during the summer allows for a healthy dose of vitamin D. Dietary sources, such as low-fat milk, salmon, tuna, and fortified orange juice, are the safest way to get enough vitamin D.
Myth: Indoor tanning is approved by the government.
Fact: According to the Federal Trade Commission, no US government agency recommends the use of indoor tanning equipment. Tanning bed use by minors has been banned in many states, and efforts are ongoing to protect consumers younger than 18 years on local, state, and federal levels. In July 2009, the International Agency for Research on Cancer, part of the World Health Organization, moved tanning devices that emit UV radiation into the highest cancer risk category—carcinogenic to humans—concluding that they are more dangerous than previously thought.
Studies have consistently shown that indoor tanning increases a person’s risk of getting skin cancer and indoor tanning at a young age appears to be more strongly related to lifetime skin cancer risk. Patients should be reminded that every time they tan, they increase their risk of melanoma as well as premature skin aging and other skin cancers. Dermatologists should counsel patients on using sun protection and avoiding indoor tanning.