User login
FDA approves nivolumab-ipilimumab combination for melanoma
Nivolumab, a programmed death receptor-1 (PD-1)–blocking antibody, has been approved for use in combination with ipilimumab for treating people with BRAF V600 wild-type, unresectable or metastatic melanoma.
Approval was based on demonstration of an increase in the objective response rate, prolonged response durations, and improvement in progression-free survival (PFS) in an international, multicenter, double-blind, randomized, two-arm, active-controlled trial in patients who were previously untreated for unresectable or metastatic, BRAF V600 wild-type melanoma, according to a Sept. 30 statement issued by the Food and Drug Administration.
Nivolumab, marketed as Opdivo Injection by Bristol-Myers Squibb, was approved in 2014 for the treatment of patients with unresectable or metastatic melanoma and disease progression following ipilimumab therapy and, if BRAF V600 mutation positive, a BRAF inhibitor. Ipilimumab, approved in 2011 and marketed as Yervoy by BMS, is a human cytotoxic T-lymphocyte antigen 4 (CTLA-4)–blocking antibody indicated for treating unresectable or metastatic melanoma.
“Combined nivolumab (anti–PD-1) and ipilimumab (anti–CTLA-4) mediated inhibition results in enhanced T-cell function that is greater than the effects of either antibody alone, and results in improved antitumor responses in metastatic melanoma,” the updated nivolumab prescribing information states.
This is the first FDA approval of a cancer treatment regimen that includes two immuno-oncology agents, according to the company’s statement announcing the approval.
The phase II CheckMate 069 study enrolled 142 patients, including 109 patients with BRAF V600 wild-type melanoma, randomized in a 2:1 ratio to receive the combination or ipilimumab plus placebo; their median age was 66 years; 84% had an ECOG performance score of 0, and 15% had a score of 1. The overall response rate was 60% among those on the combination therapy vs. 11% among those on ipilimumab alone – a 49% improvement (P less than .001), according to the FDA.
Nine of the 43 patients with an objective response (ranging from 3 to 7 months) in the combination therapy group “have progressed after response, died, or received subsequent therapy,” the FDA said. Among the 34 patients who continued to have a response at the last analysis, 14 had responses that had lasted from at least 6 months to less than 9 months. The remaining 20 patients had responses that had lasted at least 9 months.
The estimated median PFS was 8.9 months in the combination group vs. 4.7 months in the ipilimumab-only arm, a 60% reduced risk (P less than .002)
Serious adverse reactions, adverse reactions resulting in permanent discontinuation or a delayed dose, and grade 3 or 4 adverse reaction were higher among those treated with the combination. Colitis, diarrhea, pyrexia, and pneumonitis were the most common serious adverse reactions among those on the combination.
The recommended dose and schedule is as follows, according to the FDA: 1 mg/kg of nivolumab administered as an IV infusion over 60 minutes, followed by ipilimumab on the same day, every 3 weeks for four doses. “The recommended subsequent dose of nivolumab, as a single agent, is 3 mg/kg as an intravenous infusion over 60 minutes every 2 weeks until disease progression or unacceptable toxicity.”
Both indications for nivolumab were approved as accelerated approvals, based on the “tumor response rate and durability of response. Continued approval for these indications may be contingent upon verification and description of clinical benefit in the confirmatory trials,” according to the prescribing information.
Serious adverse events associated with this therapy should be reported to the FDA’s MedWatch program at 800-332-1088 or www.fda.gov/medwatch.
Nivolumab, a programmed death receptor-1 (PD-1)–blocking antibody, has been approved for use in combination with ipilimumab for treating people with BRAF V600 wild-type, unresectable or metastatic melanoma.
Approval was based on demonstration of an increase in the objective response rate, prolonged response durations, and improvement in progression-free survival (PFS) in an international, multicenter, double-blind, randomized, two-arm, active-controlled trial in patients who were previously untreated for unresectable or metastatic, BRAF V600 wild-type melanoma, according to a Sept. 30 statement issued by the Food and Drug Administration.
Nivolumab, marketed as Opdivo Injection by Bristol-Myers Squibb, was approved in 2014 for the treatment of patients with unresectable or metastatic melanoma and disease progression following ipilimumab therapy and, if BRAF V600 mutation positive, a BRAF inhibitor. Ipilimumab, approved in 2011 and marketed as Yervoy by BMS, is a human cytotoxic T-lymphocyte antigen 4 (CTLA-4)–blocking antibody indicated for treating unresectable or metastatic melanoma.
“Combined nivolumab (anti–PD-1) and ipilimumab (anti–CTLA-4) mediated inhibition results in enhanced T-cell function that is greater than the effects of either antibody alone, and results in improved antitumor responses in metastatic melanoma,” the updated nivolumab prescribing information states.
This is the first FDA approval of a cancer treatment regimen that includes two immuno-oncology agents, according to the company’s statement announcing the approval.
The phase II CheckMate 069 study enrolled 142 patients, including 109 patients with BRAF V600 wild-type melanoma, randomized in a 2:1 ratio to receive the combination or ipilimumab plus placebo; their median age was 66 years; 84% had an ECOG performance score of 0, and 15% had a score of 1. The overall response rate was 60% among those on the combination therapy vs. 11% among those on ipilimumab alone – a 49% improvement (P less than .001), according to the FDA.
Nine of the 43 patients with an objective response (ranging from 3 to 7 months) in the combination therapy group “have progressed after response, died, or received subsequent therapy,” the FDA said. Among the 34 patients who continued to have a response at the last analysis, 14 had responses that had lasted from at least 6 months to less than 9 months. The remaining 20 patients had responses that had lasted at least 9 months.
The estimated median PFS was 8.9 months in the combination group vs. 4.7 months in the ipilimumab-only arm, a 60% reduced risk (P less than .002)
Serious adverse reactions, adverse reactions resulting in permanent discontinuation or a delayed dose, and grade 3 or 4 adverse reaction were higher among those treated with the combination. Colitis, diarrhea, pyrexia, and pneumonitis were the most common serious adverse reactions among those on the combination.
The recommended dose and schedule is as follows, according to the FDA: 1 mg/kg of nivolumab administered as an IV infusion over 60 minutes, followed by ipilimumab on the same day, every 3 weeks for four doses. “The recommended subsequent dose of nivolumab, as a single agent, is 3 mg/kg as an intravenous infusion over 60 minutes every 2 weeks until disease progression or unacceptable toxicity.”
Both indications for nivolumab were approved as accelerated approvals, based on the “tumor response rate and durability of response. Continued approval for these indications may be contingent upon verification and description of clinical benefit in the confirmatory trials,” according to the prescribing information.
Serious adverse events associated with this therapy should be reported to the FDA’s MedWatch program at 800-332-1088 or www.fda.gov/medwatch.
Nivolumab, a programmed death receptor-1 (PD-1)–blocking antibody, has been approved for use in combination with ipilimumab for treating people with BRAF V600 wild-type, unresectable or metastatic melanoma.
Approval was based on demonstration of an increase in the objective response rate, prolonged response durations, and improvement in progression-free survival (PFS) in an international, multicenter, double-blind, randomized, two-arm, active-controlled trial in patients who were previously untreated for unresectable or metastatic, BRAF V600 wild-type melanoma, according to a Sept. 30 statement issued by the Food and Drug Administration.
Nivolumab, marketed as Opdivo Injection by Bristol-Myers Squibb, was approved in 2014 for the treatment of patients with unresectable or metastatic melanoma and disease progression following ipilimumab therapy and, if BRAF V600 mutation positive, a BRAF inhibitor. Ipilimumab, approved in 2011 and marketed as Yervoy by BMS, is a human cytotoxic T-lymphocyte antigen 4 (CTLA-4)–blocking antibody indicated for treating unresectable or metastatic melanoma.
“Combined nivolumab (anti–PD-1) and ipilimumab (anti–CTLA-4) mediated inhibition results in enhanced T-cell function that is greater than the effects of either antibody alone, and results in improved antitumor responses in metastatic melanoma,” the updated nivolumab prescribing information states.
This is the first FDA approval of a cancer treatment regimen that includes two immuno-oncology agents, according to the company’s statement announcing the approval.
The phase II CheckMate 069 study enrolled 142 patients, including 109 patients with BRAF V600 wild-type melanoma, randomized in a 2:1 ratio to receive the combination or ipilimumab plus placebo; their median age was 66 years; 84% had an ECOG performance score of 0, and 15% had a score of 1. The overall response rate was 60% among those on the combination therapy vs. 11% among those on ipilimumab alone – a 49% improvement (P less than .001), according to the FDA.
Nine of the 43 patients with an objective response (ranging from 3 to 7 months) in the combination therapy group “have progressed after response, died, or received subsequent therapy,” the FDA said. Among the 34 patients who continued to have a response at the last analysis, 14 had responses that had lasted from at least 6 months to less than 9 months. The remaining 20 patients had responses that had lasted at least 9 months.
The estimated median PFS was 8.9 months in the combination group vs. 4.7 months in the ipilimumab-only arm, a 60% reduced risk (P less than .002)
Serious adverse reactions, adverse reactions resulting in permanent discontinuation or a delayed dose, and grade 3 or 4 adverse reaction were higher among those treated with the combination. Colitis, diarrhea, pyrexia, and pneumonitis were the most common serious adverse reactions among those on the combination.
The recommended dose and schedule is as follows, according to the FDA: 1 mg/kg of nivolumab administered as an IV infusion over 60 minutes, followed by ipilimumab on the same day, every 3 weeks for four doses. “The recommended subsequent dose of nivolumab, as a single agent, is 3 mg/kg as an intravenous infusion over 60 minutes every 2 weeks until disease progression or unacceptable toxicity.”
Both indications for nivolumab were approved as accelerated approvals, based on the “tumor response rate and durability of response. Continued approval for these indications may be contingent upon verification and description of clinical benefit in the confirmatory trials,” according to the prescribing information.
Serious adverse events associated with this therapy should be reported to the FDA’s MedWatch program at 800-332-1088 or www.fda.gov/medwatch.
Topical fluorouracil shows long-term benefit for actinic keratoses
One course of topical fluorouracil cream reduced the need for localized treatments and the number of actinic keratoses (AK) over a mean follow-up of 2.6 years in the Veterans Affairs Keratinocyte Carcinoma Chemoprevention (VAKCC) trial.
These results “indicate that treating a patient with a single course of fluorouracil would reduce the subsequent number of spot treatments and benefit care of patients with multiple AKs for longer than 2 years,” concluded Dr. Hyemin Pomerantz of the department of dermatoepidemiology at the Providence (R.I.) VA Medical Center and his coauthors. Previous studies on treating AKs with topical fluorouracil have followed up participants for less than 6 months, they pointed out (JAMA Dermatol. 2015;9:952-60).
In the randomized, double-blinded, placebo-controlled study, conducted from 2009 to 2011 at 12 VA dermatology clinics, participants received topical fluorouracil cream, 5%, or a vehicle control cream, applied twice a day for 4 weeks, and were followed up for a mean of 2.6 years. There were no significant differences in the baseline characteristics of the 468 participants randomized to receive fluorouracil cream and the 464 participants randomized to receive the control cream.
The mean total AK count on the face and ears in both groups was about 11. At 6 months, the mean number of AKs per participant had dropped to 3 in the fluorouracil group (a 73% reduction from baseline) vs. a mean of 8.1 in the control group, a 24% reduction from baseline (P less than .001). Over the study period, those treated with fluorouracil had significantly fewer AKs. Over the entire study, there was not a significant difference in the number of hypertrophic AKs between the two groups, although the number of hypertrophic AKs was lower in the treatment group at 6 months.
During the study period, more participants treated with fluorouracil had complete clearance of AKs on the face and ears, compared with the control group (P less than .001). Finally, the fluorouracil group required 2 spot treatments per visit per participant vs. 3.9 in the control group (P less than .001).
“Our findings highlight the long-term efficacy of topical fluorouracil cream in treating and preventing AKs. A single course of topical fluorouracil cream, 5%, treatment led to a sustained reduction of the number of AKs and subsequent AK treatments,” in the population of patients at high risk for basal cell and squamous cell carcinomas, the authors wrote.
The study was supported by the Office of Research and Development Cooperative Studies Program at the Department of Veterans Affairs. Three of the authors reported several disclosures, including serving as a consultant to several pharmaceutical companies; the others, including the lead author, had no disclosures.
One course of topical fluorouracil cream reduced the need for localized treatments and the number of actinic keratoses (AK) over a mean follow-up of 2.6 years in the Veterans Affairs Keratinocyte Carcinoma Chemoprevention (VAKCC) trial.
These results “indicate that treating a patient with a single course of fluorouracil would reduce the subsequent number of spot treatments and benefit care of patients with multiple AKs for longer than 2 years,” concluded Dr. Hyemin Pomerantz of the department of dermatoepidemiology at the Providence (R.I.) VA Medical Center and his coauthors. Previous studies on treating AKs with topical fluorouracil have followed up participants for less than 6 months, they pointed out (JAMA Dermatol. 2015;9:952-60).
In the randomized, double-blinded, placebo-controlled study, conducted from 2009 to 2011 at 12 VA dermatology clinics, participants received topical fluorouracil cream, 5%, or a vehicle control cream, applied twice a day for 4 weeks, and were followed up for a mean of 2.6 years. There were no significant differences in the baseline characteristics of the 468 participants randomized to receive fluorouracil cream and the 464 participants randomized to receive the control cream.
The mean total AK count on the face and ears in both groups was about 11. At 6 months, the mean number of AKs per participant had dropped to 3 in the fluorouracil group (a 73% reduction from baseline) vs. a mean of 8.1 in the control group, a 24% reduction from baseline (P less than .001). Over the study period, those treated with fluorouracil had significantly fewer AKs. Over the entire study, there was not a significant difference in the number of hypertrophic AKs between the two groups, although the number of hypertrophic AKs was lower in the treatment group at 6 months.
During the study period, more participants treated with fluorouracil had complete clearance of AKs on the face and ears, compared with the control group (P less than .001). Finally, the fluorouracil group required 2 spot treatments per visit per participant vs. 3.9 in the control group (P less than .001).
“Our findings highlight the long-term efficacy of topical fluorouracil cream in treating and preventing AKs. A single course of topical fluorouracil cream, 5%, treatment led to a sustained reduction of the number of AKs and subsequent AK treatments,” in the population of patients at high risk for basal cell and squamous cell carcinomas, the authors wrote.
The study was supported by the Office of Research and Development Cooperative Studies Program at the Department of Veterans Affairs. Three of the authors reported several disclosures, including serving as a consultant to several pharmaceutical companies; the others, including the lead author, had no disclosures.
One course of topical fluorouracil cream reduced the need for localized treatments and the number of actinic keratoses (AK) over a mean follow-up of 2.6 years in the Veterans Affairs Keratinocyte Carcinoma Chemoprevention (VAKCC) trial.
These results “indicate that treating a patient with a single course of fluorouracil would reduce the subsequent number of spot treatments and benefit care of patients with multiple AKs for longer than 2 years,” concluded Dr. Hyemin Pomerantz of the department of dermatoepidemiology at the Providence (R.I.) VA Medical Center and his coauthors. Previous studies on treating AKs with topical fluorouracil have followed up participants for less than 6 months, they pointed out (JAMA Dermatol. 2015;9:952-60).
In the randomized, double-blinded, placebo-controlled study, conducted from 2009 to 2011 at 12 VA dermatology clinics, participants received topical fluorouracil cream, 5%, or a vehicle control cream, applied twice a day for 4 weeks, and were followed up for a mean of 2.6 years. There were no significant differences in the baseline characteristics of the 468 participants randomized to receive fluorouracil cream and the 464 participants randomized to receive the control cream.
The mean total AK count on the face and ears in both groups was about 11. At 6 months, the mean number of AKs per participant had dropped to 3 in the fluorouracil group (a 73% reduction from baseline) vs. a mean of 8.1 in the control group, a 24% reduction from baseline (P less than .001). Over the study period, those treated with fluorouracil had significantly fewer AKs. Over the entire study, there was not a significant difference in the number of hypertrophic AKs between the two groups, although the number of hypertrophic AKs was lower in the treatment group at 6 months.
During the study period, more participants treated with fluorouracil had complete clearance of AKs on the face and ears, compared with the control group (P less than .001). Finally, the fluorouracil group required 2 spot treatments per visit per participant vs. 3.9 in the control group (P less than .001).
“Our findings highlight the long-term efficacy of topical fluorouracil cream in treating and preventing AKs. A single course of topical fluorouracil cream, 5%, treatment led to a sustained reduction of the number of AKs and subsequent AK treatments,” in the population of patients at high risk for basal cell and squamous cell carcinomas, the authors wrote.
The study was supported by the Office of Research and Development Cooperative Studies Program at the Department of Veterans Affairs. Three of the authors reported several disclosures, including serving as a consultant to several pharmaceutical companies; the others, including the lead author, had no disclosures.
FROM JAMA DERMATOLOGY
Key clinical point:One course of topical fluorouracil cream, 5%, decreased the need for localized treatment and the number of actinic keratoses long term.
Major finding: Participants whose AKs were treated with a course of fluorouracil cream, 5%, had significantly fewer AKs and required fewer treatments, compared with the control group, for over 2 years.
Data source: The randomized, double-blind study compared the effect of a course of topical fluorouracil cream with a vehicle cream on the number of AKs and other measures, over a mean follow-up of 2.6 years, in 932 patients treated at 12 VA dermatology clinics.
Disclosures: The study was supported by the Office of Research and Development Cooperative Studies Program at the Department of Veterans Affairs. Three of the authors reported several disclosures, including serving as a consultant to several pharmaceutical companies; the others had no disclosures.
Topical resiquimod effective for early-stage cutaneous T-cell lymphoma
Topical resiquimod was effective and well tolerated in patients with early-stage cutaneous T-cell lymphoma (CTCL), in some cases inducing regression in both treated and untreated lesions, according to researchers.
The mean number of prior unsuccessful therapies among the patients was 6, yet the majority of patients (11 of 12) experienced significant improvement, and 2 patients had complete clinical responses with no evidence of disease after treatment. One patient, despite a 15-year history of disease and 11 unsuccessful treatments, experienced a complete resolution of both treated and untreated skin lesions.
The open-label, phase I trial evaluated 12 patients with early-stage CTCL. Patients experienced minor adverse effects (all grade 1), which were primarily skin irritation. The trial evaluated 0.03% and 0.06% resiquimod, with complete and more rapid responses occurring at the higher dose. Both doses were equally well tolerated.
“These studies support further trials of this medication in early-stage, skin-limited CTCL and suggest resiquimod might also be useful as an adjuvant therapy in the treatment of more advanced CTCL,” wrote Dr. Alain Rook of the Department of Dermatology and the Center for Clinical Biostatistics and Epidemiology, Perelman School of Medicine, Philadelphia, and colleagues.
Arising from T cells that traffic to the skin, CTCLs are non-Hodgkin lymphomas whose only potential cure is stem cell transplantation. Studies suggest that host antitumor immunity plays an important role in the disease, and in this study, high responders showed recruitment and expansion of benign T-cell clones and activation of T cells and natural killer cells in the skin.
In the absence of cell-surface markers to distinguish malignant from benign T cells in the lesion, the team used high throughput screening of the T-cell receptor–beta gene to quantify malignant cells and monitor response to therapy. Of the 10 patients with identified malignant cells, biopsied lesions showed that most had reduction of malignant T-cell clones and 3 had complete eradication. The results may not reflect responses in nonbiopsied lesions.
Resiquimod recruits T cells and other immune cells to the skin, causing inflammation that the researchers observed persisted after complete or nearly complete malignant T-cell eradication. Study results suggested that activation of CD4+ cells and expansion of tumor-specific T cells is critical for effectiveness of resiquimod.
Topical resiquimod was effective and well tolerated in patients with early-stage cutaneous T-cell lymphoma (CTCL), in some cases inducing regression in both treated and untreated lesions, according to researchers.
The mean number of prior unsuccessful therapies among the patients was 6, yet the majority of patients (11 of 12) experienced significant improvement, and 2 patients had complete clinical responses with no evidence of disease after treatment. One patient, despite a 15-year history of disease and 11 unsuccessful treatments, experienced a complete resolution of both treated and untreated skin lesions.
The open-label, phase I trial evaluated 12 patients with early-stage CTCL. Patients experienced minor adverse effects (all grade 1), which were primarily skin irritation. The trial evaluated 0.03% and 0.06% resiquimod, with complete and more rapid responses occurring at the higher dose. Both doses were equally well tolerated.
“These studies support further trials of this medication in early-stage, skin-limited CTCL and suggest resiquimod might also be useful as an adjuvant therapy in the treatment of more advanced CTCL,” wrote Dr. Alain Rook of the Department of Dermatology and the Center for Clinical Biostatistics and Epidemiology, Perelman School of Medicine, Philadelphia, and colleagues.
Arising from T cells that traffic to the skin, CTCLs are non-Hodgkin lymphomas whose only potential cure is stem cell transplantation. Studies suggest that host antitumor immunity plays an important role in the disease, and in this study, high responders showed recruitment and expansion of benign T-cell clones and activation of T cells and natural killer cells in the skin.
In the absence of cell-surface markers to distinguish malignant from benign T cells in the lesion, the team used high throughput screening of the T-cell receptor–beta gene to quantify malignant cells and monitor response to therapy. Of the 10 patients with identified malignant cells, biopsied lesions showed that most had reduction of malignant T-cell clones and 3 had complete eradication. The results may not reflect responses in nonbiopsied lesions.
Resiquimod recruits T cells and other immune cells to the skin, causing inflammation that the researchers observed persisted after complete or nearly complete malignant T-cell eradication. Study results suggested that activation of CD4+ cells and expansion of tumor-specific T cells is critical for effectiveness of resiquimod.
Topical resiquimod was effective and well tolerated in patients with early-stage cutaneous T-cell lymphoma (CTCL), in some cases inducing regression in both treated and untreated lesions, according to researchers.
The mean number of prior unsuccessful therapies among the patients was 6, yet the majority of patients (11 of 12) experienced significant improvement, and 2 patients had complete clinical responses with no evidence of disease after treatment. One patient, despite a 15-year history of disease and 11 unsuccessful treatments, experienced a complete resolution of both treated and untreated skin lesions.
The open-label, phase I trial evaluated 12 patients with early-stage CTCL. Patients experienced minor adverse effects (all grade 1), which were primarily skin irritation. The trial evaluated 0.03% and 0.06% resiquimod, with complete and more rapid responses occurring at the higher dose. Both doses were equally well tolerated.
“These studies support further trials of this medication in early-stage, skin-limited CTCL and suggest resiquimod might also be useful as an adjuvant therapy in the treatment of more advanced CTCL,” wrote Dr. Alain Rook of the Department of Dermatology and the Center for Clinical Biostatistics and Epidemiology, Perelman School of Medicine, Philadelphia, and colleagues.
Arising from T cells that traffic to the skin, CTCLs are non-Hodgkin lymphomas whose only potential cure is stem cell transplantation. Studies suggest that host antitumor immunity plays an important role in the disease, and in this study, high responders showed recruitment and expansion of benign T-cell clones and activation of T cells and natural killer cells in the skin.
In the absence of cell-surface markers to distinguish malignant from benign T cells in the lesion, the team used high throughput screening of the T-cell receptor–beta gene to quantify malignant cells and monitor response to therapy. Of the 10 patients with identified malignant cells, biopsied lesions showed that most had reduction of malignant T-cell clones and 3 had complete eradication. The results may not reflect responses in nonbiopsied lesions.
Resiquimod recruits T cells and other immune cells to the skin, causing inflammation that the researchers observed persisted after complete or nearly complete malignant T-cell eradication. Study results suggested that activation of CD4+ cells and expansion of tumor-specific T cells is critical for effectiveness of resiquimod.
FROM BLOOD
Key clinical point: Topical resiquimod was effective and well tolerated in patients with early-stage cutaneous T-cell lymphoma (CTCL).
Major finding: In total, 11 of 12 patients had significant improvement: 2 had resolution of all evidence of disease, and 9 experienced improvement greater than or equal to 50%.
Data source: The open-label, phase I trial evaluated 12 patients with early stage CTCL.
Disclosures: Dr. Rook and one coauthor have patents pending on HTS in cutaneous lymphoma. His coauthor is employed by Adaptive Biotechnologies.
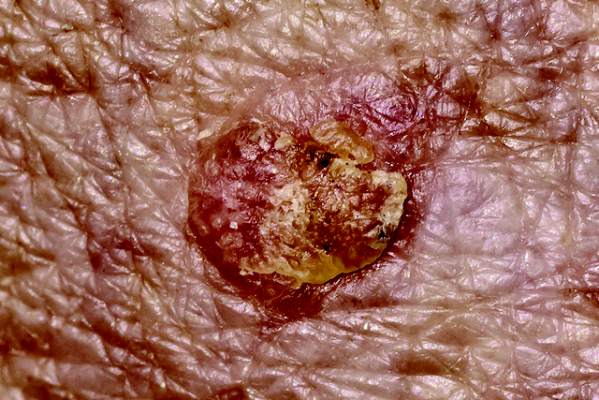
Natalizumab-Associated Melanoma: A Report of 139 Cases From the Southern Network on Adverse Reactions (SONAR)
Background: Natalizumab is an effective immunosuppressive therapy for multiple sclerosis (MS) that received its initial FDA approval in 2004. Its most notable toxicity is progressive multifocal leukoencephalopathy (PML), an opportunistic infection that is the focus of an FDA-mandated registry, TOUCH. The Southern Network on Adverse Reactions identified a fatal case of natalizumab-associated urethral melanoma and undertook an extensive evaluation of all cases of natalizumab-associated melanoma included in the FDA’s Adverse Event Reporting System (FAERS) (between 2005 and 2014).
Methods: Patient characteristics were determined from the FAERS reports and put into a comprehensive dataset. The quality of these reports was assessed based on a 15-point scale of various clinical, demographic, and pharmacy components. Cases were identified as being reported through the TOUCH system, using TOUCH information but reporting outside the system, not using TOUCH at all in the U.S. or not U.S. Quality scores and their components within the 4 Touch groups were statistically compared across all attributes using the mathematical programming-based statistical methodology univariate optimal discriminant analysis.
Results: The mean patient age at the time of diagnosis of melanoma was 46 years (SD 11). Seventeen patients were diagnosed with cutaneous melanoma developing in nonsun-exposed areas. We found that cases reported through the TOUCH registry were of lower quality (mean score 7.7) compared with others that reported outside of the U.S. (mean score 8.5, P < .008). Those cases reported through the TOUCH system were less likely to report the site of melanoma (P < .019) and have overall lower clinical scores (P < .04) compared with reports using TOUCH information but reporting outside of the system.
Conclusions: Our findings suggest that in the U.S., the TOUCH Registry should be expanded to require clinicians to report details of natalizumab-associated melanoma, an opportunistic illness that frequently develops in immunocompromised persons. Also, the FDA-approved product label for natalizumab should be revised to include information on occurrences of melanoma among natalizumab-treated MS patients, particularly those who have cutaneous nevi prior to natalizumab initiation. Natalizumab-treated MS patients and their physicians should be vigilant for changes in nevi appearances and development of new cutaneous lesions (particularly in nonsun-exposed cutaneous areas).
Background: Natalizumab is an effective immunosuppressive therapy for multiple sclerosis (MS) that received its initial FDA approval in 2004. Its most notable toxicity is progressive multifocal leukoencephalopathy (PML), an opportunistic infection that is the focus of an FDA-mandated registry, TOUCH. The Southern Network on Adverse Reactions identified a fatal case of natalizumab-associated urethral melanoma and undertook an extensive evaluation of all cases of natalizumab-associated melanoma included in the FDA’s Adverse Event Reporting System (FAERS) (between 2005 and 2014).
Methods: Patient characteristics were determined from the FAERS reports and put into a comprehensive dataset. The quality of these reports was assessed based on a 15-point scale of various clinical, demographic, and pharmacy components. Cases were identified as being reported through the TOUCH system, using TOUCH information but reporting outside the system, not using TOUCH at all in the U.S. or not U.S. Quality scores and their components within the 4 Touch groups were statistically compared across all attributes using the mathematical programming-based statistical methodology univariate optimal discriminant analysis.
Results: The mean patient age at the time of diagnosis of melanoma was 46 years (SD 11). Seventeen patients were diagnosed with cutaneous melanoma developing in nonsun-exposed areas. We found that cases reported through the TOUCH registry were of lower quality (mean score 7.7) compared with others that reported outside of the U.S. (mean score 8.5, P < .008). Those cases reported through the TOUCH system were less likely to report the site of melanoma (P < .019) and have overall lower clinical scores (P < .04) compared with reports using TOUCH information but reporting outside of the system.
Conclusions: Our findings suggest that in the U.S., the TOUCH Registry should be expanded to require clinicians to report details of natalizumab-associated melanoma, an opportunistic illness that frequently develops in immunocompromised persons. Also, the FDA-approved product label for natalizumab should be revised to include information on occurrences of melanoma among natalizumab-treated MS patients, particularly those who have cutaneous nevi prior to natalizumab initiation. Natalizumab-treated MS patients and their physicians should be vigilant for changes in nevi appearances and development of new cutaneous lesions (particularly in nonsun-exposed cutaneous areas).
Background: Natalizumab is an effective immunosuppressive therapy for multiple sclerosis (MS) that received its initial FDA approval in 2004. Its most notable toxicity is progressive multifocal leukoencephalopathy (PML), an opportunistic infection that is the focus of an FDA-mandated registry, TOUCH. The Southern Network on Adverse Reactions identified a fatal case of natalizumab-associated urethral melanoma and undertook an extensive evaluation of all cases of natalizumab-associated melanoma included in the FDA’s Adverse Event Reporting System (FAERS) (between 2005 and 2014).
Methods: Patient characteristics were determined from the FAERS reports and put into a comprehensive dataset. The quality of these reports was assessed based on a 15-point scale of various clinical, demographic, and pharmacy components. Cases were identified as being reported through the TOUCH system, using TOUCH information but reporting outside the system, not using TOUCH at all in the U.S. or not U.S. Quality scores and their components within the 4 Touch groups were statistically compared across all attributes using the mathematical programming-based statistical methodology univariate optimal discriminant analysis.
Results: The mean patient age at the time of diagnosis of melanoma was 46 years (SD 11). Seventeen patients were diagnosed with cutaneous melanoma developing in nonsun-exposed areas. We found that cases reported through the TOUCH registry were of lower quality (mean score 7.7) compared with others that reported outside of the U.S. (mean score 8.5, P < .008). Those cases reported through the TOUCH system were less likely to report the site of melanoma (P < .019) and have overall lower clinical scores (P < .04) compared with reports using TOUCH information but reporting outside of the system.
Conclusions: Our findings suggest that in the U.S., the TOUCH Registry should be expanded to require clinicians to report details of natalizumab-associated melanoma, an opportunistic illness that frequently develops in immunocompromised persons. Also, the FDA-approved product label for natalizumab should be revised to include information on occurrences of melanoma among natalizumab-treated MS patients, particularly those who have cutaneous nevi prior to natalizumab initiation. Natalizumab-treated MS patients and their physicians should be vigilant for changes in nevi appearances and development of new cutaneous lesions (particularly in nonsun-exposed cutaneous areas).
New cancer cases to reach nearly 2.4 million in 2035 – AACR
There will be nearly 2.4 million new cases of cancer in the U.S. by 2035, according to an estimate from the American Association for Cancer Research.
In its Cancer Progress Report 2015, AACR notes that new cases in the current year are estimated at nearly 1.7 million.
“Given that cancer is primarily a disease of aging and that the portion of the U.S. population age 65 and older is expected to double in size by 2060, it is anticipated that the number of new cancer cases diagnosed each year in the United States will increase dramatically,” the report states.
“Also contributing to the projected increase are the continual use of cigarettes by 18% of U.S. adults, and high rates of obesity and physical inactivity, both of which are linked to an increased risk for several types of cancer.”
The spike in new cases comes as death rates are trending downward. From 2002 to 2011, overall cancer death rates declined per year by 1.8% for U.S. men, 1.4% for U.S. women, and 2.1% for 0- to 19-year-olds, though the overall number of cancer deaths is expected to rise. Nearly 600,000 people living in the United States are expected to die from cancer in 2015.
The new diagnoses are expected to drive up the cost of cancer.
“In the United States alone, it is estimated that the direct medical costs of cancer care will rise to $156 billion in 2020, from nearly $125 billion in 2010,” AACR says in the report. “When these costs are compared to the total NCI budget for fiscal year 2015, which is just $5 billion, it is clear that research that spurs lifesaving progress against cancer is a wise national investment.”
To that end, AACR is calling on Congress and the White House to boost funding for both the National Institutes of Health and the National Cancer Institute by at least 7%, which would result in funding levels in fiscal year 2020 for NIH and NCI of $42.5 billion and $7 billion, respectively. AACR is also calling for a similar 7% increase the Food and Drug Administration’s budget to help drive private biomedical research.
There will be nearly 2.4 million new cases of cancer in the U.S. by 2035, according to an estimate from the American Association for Cancer Research.
In its Cancer Progress Report 2015, AACR notes that new cases in the current year are estimated at nearly 1.7 million.
“Given that cancer is primarily a disease of aging and that the portion of the U.S. population age 65 and older is expected to double in size by 2060, it is anticipated that the number of new cancer cases diagnosed each year in the United States will increase dramatically,” the report states.
“Also contributing to the projected increase are the continual use of cigarettes by 18% of U.S. adults, and high rates of obesity and physical inactivity, both of which are linked to an increased risk for several types of cancer.”
The spike in new cases comes as death rates are trending downward. From 2002 to 2011, overall cancer death rates declined per year by 1.8% for U.S. men, 1.4% for U.S. women, and 2.1% for 0- to 19-year-olds, though the overall number of cancer deaths is expected to rise. Nearly 600,000 people living in the United States are expected to die from cancer in 2015.
The new diagnoses are expected to drive up the cost of cancer.
“In the United States alone, it is estimated that the direct medical costs of cancer care will rise to $156 billion in 2020, from nearly $125 billion in 2010,” AACR says in the report. “When these costs are compared to the total NCI budget for fiscal year 2015, which is just $5 billion, it is clear that research that spurs lifesaving progress against cancer is a wise national investment.”
To that end, AACR is calling on Congress and the White House to boost funding for both the National Institutes of Health and the National Cancer Institute by at least 7%, which would result in funding levels in fiscal year 2020 for NIH and NCI of $42.5 billion and $7 billion, respectively. AACR is also calling for a similar 7% increase the Food and Drug Administration’s budget to help drive private biomedical research.
There will be nearly 2.4 million new cases of cancer in the U.S. by 2035, according to an estimate from the American Association for Cancer Research.
In its Cancer Progress Report 2015, AACR notes that new cases in the current year are estimated at nearly 1.7 million.
“Given that cancer is primarily a disease of aging and that the portion of the U.S. population age 65 and older is expected to double in size by 2060, it is anticipated that the number of new cancer cases diagnosed each year in the United States will increase dramatically,” the report states.
“Also contributing to the projected increase are the continual use of cigarettes by 18% of U.S. adults, and high rates of obesity and physical inactivity, both of which are linked to an increased risk for several types of cancer.”
The spike in new cases comes as death rates are trending downward. From 2002 to 2011, overall cancer death rates declined per year by 1.8% for U.S. men, 1.4% for U.S. women, and 2.1% for 0- to 19-year-olds, though the overall number of cancer deaths is expected to rise. Nearly 600,000 people living in the United States are expected to die from cancer in 2015.
The new diagnoses are expected to drive up the cost of cancer.
“In the United States alone, it is estimated that the direct medical costs of cancer care will rise to $156 billion in 2020, from nearly $125 billion in 2010,” AACR says in the report. “When these costs are compared to the total NCI budget for fiscal year 2015, which is just $5 billion, it is clear that research that spurs lifesaving progress against cancer is a wise national investment.”
To that end, AACR is calling on Congress and the White House to boost funding for both the National Institutes of Health and the National Cancer Institute by at least 7%, which would result in funding levels in fiscal year 2020 for NIH and NCI of $42.5 billion and $7 billion, respectively. AACR is also calling for a similar 7% increase the Food and Drug Administration’s budget to help drive private biomedical research.
FROM AACR CANCER PROGRESS REPORT 2015
Acne and Melanoma: What to Do With the Reported Connection?
Dermatologists have become accustomed to reading about the associations of dermatologic disease with extracutaneous comorbidities (psoriasis certainly takes the lead). One may see the headline “Study finds increased risk for melanoma in female acne patients” and say “Sure, why not?” However, before we all jump on the association bandwagon, let’s better appreciate this finding.
A study published online January 8 in Cancer by Zhang et al followed 99,128 female nurses in the Nurses’ Health Study II cohort for 20 years. This cohort has been utilized for numerous prospective studies over the year. Even after adjusting for known risk factors, investigators discovered that women with a history of severe cystic teenage acne had a hazard ratio of 1.44 for melanoma. The authors replicated the association with an independent melanoma case-control study of 930 cases and 1026 controls, finding an odds ratio of 1.27. They also found that individuals with teenage acne were more likely to have nevi (52.7% vs 50.1% in the cohort study; 55.2% vs 45.1% in the control study).
These data points ultimately led the team to conclude that acne may serve as an independent risk factor for melanoma, attributing androgens in female acne as a possible and plausible explanation due to their known effect on telomere elongation; melanocytes with longer telomere lengths have more opportunity to develop mutations, which could lead to malignant transformation, as the extended length ultimately delays initiation of cellular senescence. The longer these cells are “awake,” more moles can form, which means more room for trouble.
What’s the issue?
The size of this cohort certainly gives credibility to the data and statistics presented. Although the study is powered very well by the numbers, it is a unique cohort because all participants were nurses, narrowing down the demographics to some degree given general patterns, behaviors, and backgrounds when it comes to this group, an issue that has been previously raised with using this cohort. That said, more research is certainly warranted to elucidate the proposed mechanism and further clarify the association.
From a purely clinical standpoint, this paper is powerful ammo that can be used in our war against skin cancer. This very large cohort probably does not follow the American Academy of Dermatology guidelines for sun protection, skin cancer prevention, and surveillance. It could be a nice tidbit for patients at the end of your spiel on acne and then work in the photoprotection discussion, something we haven’t been the best at according to a recent study published in JAMA Dermatology (JAMA Dermatol. 2014;150:51-55)! Would it be such a bad thing if this paper helped us encourage all women with moderate to severe acne to undertake more effective sun-safe behaviors and to visit their dermatologist every year for total-body skin examinations?
Dermatologists have become accustomed to reading about the associations of dermatologic disease with extracutaneous comorbidities (psoriasis certainly takes the lead). One may see the headline “Study finds increased risk for melanoma in female acne patients” and say “Sure, why not?” However, before we all jump on the association bandwagon, let’s better appreciate this finding.
A study published online January 8 in Cancer by Zhang et al followed 99,128 female nurses in the Nurses’ Health Study II cohort for 20 years. This cohort has been utilized for numerous prospective studies over the year. Even after adjusting for known risk factors, investigators discovered that women with a history of severe cystic teenage acne had a hazard ratio of 1.44 for melanoma. The authors replicated the association with an independent melanoma case-control study of 930 cases and 1026 controls, finding an odds ratio of 1.27. They also found that individuals with teenage acne were more likely to have nevi (52.7% vs 50.1% in the cohort study; 55.2% vs 45.1% in the control study).
These data points ultimately led the team to conclude that acne may serve as an independent risk factor for melanoma, attributing androgens in female acne as a possible and plausible explanation due to their known effect on telomere elongation; melanocytes with longer telomere lengths have more opportunity to develop mutations, which could lead to malignant transformation, as the extended length ultimately delays initiation of cellular senescence. The longer these cells are “awake,” more moles can form, which means more room for trouble.
What’s the issue?
The size of this cohort certainly gives credibility to the data and statistics presented. Although the study is powered very well by the numbers, it is a unique cohort because all participants were nurses, narrowing down the demographics to some degree given general patterns, behaviors, and backgrounds when it comes to this group, an issue that has been previously raised with using this cohort. That said, more research is certainly warranted to elucidate the proposed mechanism and further clarify the association.
From a purely clinical standpoint, this paper is powerful ammo that can be used in our war against skin cancer. This very large cohort probably does not follow the American Academy of Dermatology guidelines for sun protection, skin cancer prevention, and surveillance. It could be a nice tidbit for patients at the end of your spiel on acne and then work in the photoprotection discussion, something we haven’t been the best at according to a recent study published in JAMA Dermatology (JAMA Dermatol. 2014;150:51-55)! Would it be such a bad thing if this paper helped us encourage all women with moderate to severe acne to undertake more effective sun-safe behaviors and to visit their dermatologist every year for total-body skin examinations?
Dermatologists have become accustomed to reading about the associations of dermatologic disease with extracutaneous comorbidities (psoriasis certainly takes the lead). One may see the headline “Study finds increased risk for melanoma in female acne patients” and say “Sure, why not?” However, before we all jump on the association bandwagon, let’s better appreciate this finding.
A study published online January 8 in Cancer by Zhang et al followed 99,128 female nurses in the Nurses’ Health Study II cohort for 20 years. This cohort has been utilized for numerous prospective studies over the year. Even after adjusting for known risk factors, investigators discovered that women with a history of severe cystic teenage acne had a hazard ratio of 1.44 for melanoma. The authors replicated the association with an independent melanoma case-control study of 930 cases and 1026 controls, finding an odds ratio of 1.27. They also found that individuals with teenage acne were more likely to have nevi (52.7% vs 50.1% in the cohort study; 55.2% vs 45.1% in the control study).
These data points ultimately led the team to conclude that acne may serve as an independent risk factor for melanoma, attributing androgens in female acne as a possible and plausible explanation due to their known effect on telomere elongation; melanocytes with longer telomere lengths have more opportunity to develop mutations, which could lead to malignant transformation, as the extended length ultimately delays initiation of cellular senescence. The longer these cells are “awake,” more moles can form, which means more room for trouble.
What’s the issue?
The size of this cohort certainly gives credibility to the data and statistics presented. Although the study is powered very well by the numbers, it is a unique cohort because all participants were nurses, narrowing down the demographics to some degree given general patterns, behaviors, and backgrounds when it comes to this group, an issue that has been previously raised with using this cohort. That said, more research is certainly warranted to elucidate the proposed mechanism and further clarify the association.
From a purely clinical standpoint, this paper is powerful ammo that can be used in our war against skin cancer. This very large cohort probably does not follow the American Academy of Dermatology guidelines for sun protection, skin cancer prevention, and surveillance. It could be a nice tidbit for patients at the end of your spiel on acne and then work in the photoprotection discussion, something we haven’t been the best at according to a recent study published in JAMA Dermatology (JAMA Dermatol. 2014;150:51-55)! Would it be such a bad thing if this paper helped us encourage all women with moderate to severe acne to undertake more effective sun-safe behaviors and to visit their dermatologist every year for total-body skin examinations?
Sunscreens with DNA repair enzymes might lessen AK progression
Patients with actinic keratosis who used UPF 50 sunscreen containing DNA repair enzymes improved significantly more on two measures of malignant progression than did those who used sunscreen alone, according to research published in the Journal of Drugs in Dermatology.
At 6 months, improvements in field cancerization and levels of cyclobutane pyrimidine dimers were significantly greater (P less than .001) for the sunscreen-plus-enzymes group compared with sunscreen-only patients, wrote Dr. Mauro Carducci of Centro Ortopedico di Quadrante in Omegna, Italy, and his associates.
The study is the first of its type to directly compare the clinical effects of two such topicals, the investigators wrote. The findings set the stage for longer, larger trials that are powered to assess the risk of progression to squamous cell carcinoma, they added.
For the study, 28 patients with AK were randomly assigned to use SPF 50 sunscreen alone or a formula that contained 1% photolyase from Anacystis nidulans and 1% endonuclease from Micrococcus luteus. Patients applied 2 mg/cm2 of sunscreen to treatment areas that contained 4-10 AKs. They were not allowed to use other topicals during the trial or for 2 weeks beforehand.
All of the patients were white and aged older than 65 years; three-quarters were men. The investigators used fluorescence diagnostics with methylaminolaevulinate to measure field cancerization, and analyzed skin biopsies to quantify CPD levels (J Drugs Dermatol. 2015;14[9]:986-90.).
Hyperkeratosis improved the same amount in both groups at month 6, according to the researchers. But field cancerizations dropped 29% from baseline in the sunscreen-plus-enzymes group, compared with a 10% decrease with sunscreen alone (P less than .0001). Likewise, CPD levels fell 61% from baseline in the sunscreen-plus-enzymes group compared with a 35% drop with sunscreen alone (P less than .0001).
Despite those significant differences, the study was not powered to detect differences in the risk of transformation to SCC, the researchers cautioned.
Biodue S.p.A. provided the methyl aminolevulinate used in the study. Dr. Enzo Emanuele, the study’s senior author, is a major shareholder of Living Research S.A.S., a privately held biomedical research organization that provided funding for the work. The other researchers reported no conflicts of interest.
Patients with actinic keratosis who used UPF 50 sunscreen containing DNA repair enzymes improved significantly more on two measures of malignant progression than did those who used sunscreen alone, according to research published in the Journal of Drugs in Dermatology.
At 6 months, improvements in field cancerization and levels of cyclobutane pyrimidine dimers were significantly greater (P less than .001) for the sunscreen-plus-enzymes group compared with sunscreen-only patients, wrote Dr. Mauro Carducci of Centro Ortopedico di Quadrante in Omegna, Italy, and his associates.
The study is the first of its type to directly compare the clinical effects of two such topicals, the investigators wrote. The findings set the stage for longer, larger trials that are powered to assess the risk of progression to squamous cell carcinoma, they added.
For the study, 28 patients with AK were randomly assigned to use SPF 50 sunscreen alone or a formula that contained 1% photolyase from Anacystis nidulans and 1% endonuclease from Micrococcus luteus. Patients applied 2 mg/cm2 of sunscreen to treatment areas that contained 4-10 AKs. They were not allowed to use other topicals during the trial or for 2 weeks beforehand.
All of the patients were white and aged older than 65 years; three-quarters were men. The investigators used fluorescence diagnostics with methylaminolaevulinate to measure field cancerization, and analyzed skin biopsies to quantify CPD levels (J Drugs Dermatol. 2015;14[9]:986-90.).
Hyperkeratosis improved the same amount in both groups at month 6, according to the researchers. But field cancerizations dropped 29% from baseline in the sunscreen-plus-enzymes group, compared with a 10% decrease with sunscreen alone (P less than .0001). Likewise, CPD levels fell 61% from baseline in the sunscreen-plus-enzymes group compared with a 35% drop with sunscreen alone (P less than .0001).
Despite those significant differences, the study was not powered to detect differences in the risk of transformation to SCC, the researchers cautioned.
Biodue S.p.A. provided the methyl aminolevulinate used in the study. Dr. Enzo Emanuele, the study’s senior author, is a major shareholder of Living Research S.A.S., a privately held biomedical research organization that provided funding for the work. The other researchers reported no conflicts of interest.
Patients with actinic keratosis who used UPF 50 sunscreen containing DNA repair enzymes improved significantly more on two measures of malignant progression than did those who used sunscreen alone, according to research published in the Journal of Drugs in Dermatology.
At 6 months, improvements in field cancerization and levels of cyclobutane pyrimidine dimers were significantly greater (P less than .001) for the sunscreen-plus-enzymes group compared with sunscreen-only patients, wrote Dr. Mauro Carducci of Centro Ortopedico di Quadrante in Omegna, Italy, and his associates.
The study is the first of its type to directly compare the clinical effects of two such topicals, the investigators wrote. The findings set the stage for longer, larger trials that are powered to assess the risk of progression to squamous cell carcinoma, they added.
For the study, 28 patients with AK were randomly assigned to use SPF 50 sunscreen alone or a formula that contained 1% photolyase from Anacystis nidulans and 1% endonuclease from Micrococcus luteus. Patients applied 2 mg/cm2 of sunscreen to treatment areas that contained 4-10 AKs. They were not allowed to use other topicals during the trial or for 2 weeks beforehand.
All of the patients were white and aged older than 65 years; three-quarters were men. The investigators used fluorescence diagnostics with methylaminolaevulinate to measure field cancerization, and analyzed skin biopsies to quantify CPD levels (J Drugs Dermatol. 2015;14[9]:986-90.).
Hyperkeratosis improved the same amount in both groups at month 6, according to the researchers. But field cancerizations dropped 29% from baseline in the sunscreen-plus-enzymes group, compared with a 10% decrease with sunscreen alone (P less than .0001). Likewise, CPD levels fell 61% from baseline in the sunscreen-plus-enzymes group compared with a 35% drop with sunscreen alone (P less than .0001).
Despite those significant differences, the study was not powered to detect differences in the risk of transformation to SCC, the researchers cautioned.
Biodue S.p.A. provided the methyl aminolevulinate used in the study. Dr. Enzo Emanuele, the study’s senior author, is a major shareholder of Living Research S.A.S., a privately held biomedical research organization that provided funding for the work. The other researchers reported no conflicts of interest.
FROM THE JOURNAL OF DRUGS IN DERMATOLOGY
Key clinical point:Sunscreen containing DNA repair enzymes might prevent malignant progression of actinic keratosis better than sunscreen alone.
Major finding: Field cancerization and cyclobutane pyrimidine dimer levels improved significantly more with sunscreen plus enzymes than with sunscreen only (P less than .0001 for each).
Data source: Six-month randomized trial of 28 patients with actinic keratosis.
Disclosures: Biodue S.p.A. provided the methyl aminolevulinate used in the study. Dr. Enzo Emanuele, the study’s senior author, is a major shareholder of Living Research S.A.S., a privately held biomedical research organization that provided funding for the work. The other researchers reported no conflicts of interest.
Management of dysplastic nevi varies widely
PARK CITY, UTAH – When you ask clinicians why they elect to reexcise dysplastic nevi, you’re likely to get a variety of answers, according to Dr. Douglas Grossman.
“Sometimes they have no reason all,” Dr. Grossman, professor of dermatology at the University of Utah, Salt Lake City, said at the annual meeting of the Pacific Dermatologic Association. “Sometimes the answer is ‘to prevent recurrence.’ Sometimes it’s to ensure complete removal of the lesion, to confirm the diagnosis, to rule out melanoma, or for ‘therapeutic’ purposes.”
Dysplastic nevus margins are often positive, “because the melanocytes in these lesions tend to extend up to 2 mm beyond clinical margins,” Dr. Grossman said. “I always try to remove the entire clinical lesion as well as 1-2 mm around the lesion to avoid positive margins.”
A survey of 101 Chicago Dermatological Society members found wide variation in clinical practice concerning how clinicians manage dysplastic nevi if the margins are clear vs. positive, and based on the degree of atypia (Arch Dermatol. 2012;148[2]:259-60). The greatest quantitative shift in decision making (from observe to reexcise) was seen for dysplastic nevi with moderate dysplasia. Specifically, the decision to reexcise dysplastic nevi with moderate dysplasia ranged from 9% (for clear margins) to 81% (for positive margins) of respondents. “The margin status is driving the management,” said Dr. Grossman, who was not involved with the study.
In his opinion, two “unspoken fears” are also impacting the way clinicians treat dysplastic nevi. The first is that the nevus will recur in the scar years later and be indistinguishable from melanoma – the so-called pseudomelanoma phenomenon. The second fear is that residual nevus cells left behind will transform to melanoma. Evidence for these two possibilities, however, is lacking, Dr. Grossman said. According to a large study of pseudomelanoma phenomena, recurrence was rare and easily distinguishable from cases of melanoma with regression (Modern Pathol. 2009;22:611-7). “Almost all of them had recurred within a year, so if a nevus is going to recur, it’s usually going to recur within that first year,” he said.
As for the issue of recurrence, Dr. Grossman led a research team that evaluated 271 nevus biopsy sites in 115 patients (J Am Acad Dermatol. 2010;62[4]:591-6). At greater than 2 years of follow-up they observed a recurrence rate of 3.6% for dysplastic nevi, compared with 3.3% for nondysplastic nevi, “which is similar,” he said. In terms of melanoma development, one study of 28 incompletely removed dysplastic nevi found that no melanomas developed during 5 years of follow-up (Am J Dermatopathol. 1985;7 Suppl:93-7).
Dr. Grossman referenced four cases in the medical literature in which the diagnosis was changed upon reexcision from dysplastic nevus to melanoma. “The question here is whether the initial biopsy or the reexcision was the correct diagnosis,” he said. “Development of melanoma at the site of a previously biopsied dysplastic nevus is exceedingly rare, and has not been described beyond a few case reports. The most likely explanation for how this could occur would be sampling error. If you have a melanoma arising at the site, maybe it was melanoma to start with, and the diagnosis was missed on the original biopsy. It could also be the case of pseudomelanoma phenomenon where it’s a nevus to start with but it comes back looking more atypical, so it’s incorrectly diagnosed as melanoma. A final possibility, which is the least likely, is that the few nevus cells left behind transformed into melanoma.”
According to a recent consensus statement from the Pigmented Lesion Subcommittee of the Melanoma Prevention Working Group, mild/moderate dysplastic nevi with clear margins do not need reexcision (JAMA Dermatol. 2015;151[2]:212-8). The working group also recommends that mild dysplastic nevi with positive margins may be safely observed and that observation for moderately dysplastic nevi with positive margins “may be a reasonable option.”
Dr. Grossman reported having no financial disclosures.
PARK CITY, UTAH – When you ask clinicians why they elect to reexcise dysplastic nevi, you’re likely to get a variety of answers, according to Dr. Douglas Grossman.
“Sometimes they have no reason all,” Dr. Grossman, professor of dermatology at the University of Utah, Salt Lake City, said at the annual meeting of the Pacific Dermatologic Association. “Sometimes the answer is ‘to prevent recurrence.’ Sometimes it’s to ensure complete removal of the lesion, to confirm the diagnosis, to rule out melanoma, or for ‘therapeutic’ purposes.”
Dysplastic nevus margins are often positive, “because the melanocytes in these lesions tend to extend up to 2 mm beyond clinical margins,” Dr. Grossman said. “I always try to remove the entire clinical lesion as well as 1-2 mm around the lesion to avoid positive margins.”
A survey of 101 Chicago Dermatological Society members found wide variation in clinical practice concerning how clinicians manage dysplastic nevi if the margins are clear vs. positive, and based on the degree of atypia (Arch Dermatol. 2012;148[2]:259-60). The greatest quantitative shift in decision making (from observe to reexcise) was seen for dysplastic nevi with moderate dysplasia. Specifically, the decision to reexcise dysplastic nevi with moderate dysplasia ranged from 9% (for clear margins) to 81% (for positive margins) of respondents. “The margin status is driving the management,” said Dr. Grossman, who was not involved with the study.
In his opinion, two “unspoken fears” are also impacting the way clinicians treat dysplastic nevi. The first is that the nevus will recur in the scar years later and be indistinguishable from melanoma – the so-called pseudomelanoma phenomenon. The second fear is that residual nevus cells left behind will transform to melanoma. Evidence for these two possibilities, however, is lacking, Dr. Grossman said. According to a large study of pseudomelanoma phenomena, recurrence was rare and easily distinguishable from cases of melanoma with regression (Modern Pathol. 2009;22:611-7). “Almost all of them had recurred within a year, so if a nevus is going to recur, it’s usually going to recur within that first year,” he said.
As for the issue of recurrence, Dr. Grossman led a research team that evaluated 271 nevus biopsy sites in 115 patients (J Am Acad Dermatol. 2010;62[4]:591-6). At greater than 2 years of follow-up they observed a recurrence rate of 3.6% for dysplastic nevi, compared with 3.3% for nondysplastic nevi, “which is similar,” he said. In terms of melanoma development, one study of 28 incompletely removed dysplastic nevi found that no melanomas developed during 5 years of follow-up (Am J Dermatopathol. 1985;7 Suppl:93-7).
Dr. Grossman referenced four cases in the medical literature in which the diagnosis was changed upon reexcision from dysplastic nevus to melanoma. “The question here is whether the initial biopsy or the reexcision was the correct diagnosis,” he said. “Development of melanoma at the site of a previously biopsied dysplastic nevus is exceedingly rare, and has not been described beyond a few case reports. The most likely explanation for how this could occur would be sampling error. If you have a melanoma arising at the site, maybe it was melanoma to start with, and the diagnosis was missed on the original biopsy. It could also be the case of pseudomelanoma phenomenon where it’s a nevus to start with but it comes back looking more atypical, so it’s incorrectly diagnosed as melanoma. A final possibility, which is the least likely, is that the few nevus cells left behind transformed into melanoma.”
According to a recent consensus statement from the Pigmented Lesion Subcommittee of the Melanoma Prevention Working Group, mild/moderate dysplastic nevi with clear margins do not need reexcision (JAMA Dermatol. 2015;151[2]:212-8). The working group also recommends that mild dysplastic nevi with positive margins may be safely observed and that observation for moderately dysplastic nevi with positive margins “may be a reasonable option.”
Dr. Grossman reported having no financial disclosures.
PARK CITY, UTAH – When you ask clinicians why they elect to reexcise dysplastic nevi, you’re likely to get a variety of answers, according to Dr. Douglas Grossman.
“Sometimes they have no reason all,” Dr. Grossman, professor of dermatology at the University of Utah, Salt Lake City, said at the annual meeting of the Pacific Dermatologic Association. “Sometimes the answer is ‘to prevent recurrence.’ Sometimes it’s to ensure complete removal of the lesion, to confirm the diagnosis, to rule out melanoma, or for ‘therapeutic’ purposes.”
Dysplastic nevus margins are often positive, “because the melanocytes in these lesions tend to extend up to 2 mm beyond clinical margins,” Dr. Grossman said. “I always try to remove the entire clinical lesion as well as 1-2 mm around the lesion to avoid positive margins.”
A survey of 101 Chicago Dermatological Society members found wide variation in clinical practice concerning how clinicians manage dysplastic nevi if the margins are clear vs. positive, and based on the degree of atypia (Arch Dermatol. 2012;148[2]:259-60). The greatest quantitative shift in decision making (from observe to reexcise) was seen for dysplastic nevi with moderate dysplasia. Specifically, the decision to reexcise dysplastic nevi with moderate dysplasia ranged from 9% (for clear margins) to 81% (for positive margins) of respondents. “The margin status is driving the management,” said Dr. Grossman, who was not involved with the study.
In his opinion, two “unspoken fears” are also impacting the way clinicians treat dysplastic nevi. The first is that the nevus will recur in the scar years later and be indistinguishable from melanoma – the so-called pseudomelanoma phenomenon. The second fear is that residual nevus cells left behind will transform to melanoma. Evidence for these two possibilities, however, is lacking, Dr. Grossman said. According to a large study of pseudomelanoma phenomena, recurrence was rare and easily distinguishable from cases of melanoma with regression (Modern Pathol. 2009;22:611-7). “Almost all of them had recurred within a year, so if a nevus is going to recur, it’s usually going to recur within that first year,” he said.
As for the issue of recurrence, Dr. Grossman led a research team that evaluated 271 nevus biopsy sites in 115 patients (J Am Acad Dermatol. 2010;62[4]:591-6). At greater than 2 years of follow-up they observed a recurrence rate of 3.6% for dysplastic nevi, compared with 3.3% for nondysplastic nevi, “which is similar,” he said. In terms of melanoma development, one study of 28 incompletely removed dysplastic nevi found that no melanomas developed during 5 years of follow-up (Am J Dermatopathol. 1985;7 Suppl:93-7).
Dr. Grossman referenced four cases in the medical literature in which the diagnosis was changed upon reexcision from dysplastic nevus to melanoma. “The question here is whether the initial biopsy or the reexcision was the correct diagnosis,” he said. “Development of melanoma at the site of a previously biopsied dysplastic nevus is exceedingly rare, and has not been described beyond a few case reports. The most likely explanation for how this could occur would be sampling error. If you have a melanoma arising at the site, maybe it was melanoma to start with, and the diagnosis was missed on the original biopsy. It could also be the case of pseudomelanoma phenomenon where it’s a nevus to start with but it comes back looking more atypical, so it’s incorrectly diagnosed as melanoma. A final possibility, which is the least likely, is that the few nevus cells left behind transformed into melanoma.”
According to a recent consensus statement from the Pigmented Lesion Subcommittee of the Melanoma Prevention Working Group, mild/moderate dysplastic nevi with clear margins do not need reexcision (JAMA Dermatol. 2015;151[2]:212-8). The working group also recommends that mild dysplastic nevi with positive margins may be safely observed and that observation for moderately dysplastic nevi with positive margins “may be a reasonable option.”
Dr. Grossman reported having no financial disclosures.
EXPERT ANALYSIS FROM PDA 2015
An Analysis of the Clinical Trial Landscape for Cutaneous Melanoma
The incidence of cutaneous melanoma, the deadliest form of skin cancer, has been steadily increasing over the last several decades.1 Currently, there are 73,870 new diagnoses of melanoma anticipated in the United States in 2015 alone.2 Many cases of melanoma are caught at early, actionable, and curable stages thanks in part to patient education and screening by dermatologists.3 However, until recently, few options existed for the treatment of locally advanced and metastatic melanomas, with a median survival rate of less than 1 year.4
Clinical trials represent the most reliable method for advancing treatment and improving outcomes for patients with disease; however, patient accrual and access to clinical trials remain formidable barriers. Studies have suggested that patients in rural areas perceive both an increased distance to clinical trial sites and a lack of awareness of available trials compared to their urban counterparts. Additionally, studies have shown that provider awareness of actively enrolling clinical trials in their respective fields is a key determinate in patient enrollment.5 Finally, insufficient funding and lack of collaboration has resulted in many small phase 1 or phase 2 single-center trials, which are less likely to quickly impact clinical care.6 Increased awareness of the ClinicalTrials.gov registry, a publicly available and easily accessible database, can facilitate referral, enrollment, and collaboration among physicians, patients, and researchers alike.
Using the ClinicalTrials.gov database, we sought to analyze the clinical trial landscape for cutaneous melanoma to understand the current state of melanoma research, future direction, and potential barriers that may impede success.
Methods
The primary objective was to provide a snapshot of the melanoma clinical research landscape from 2005 to 2013, including the number of registered trials, phase distribution, recruitment status, location of trials, type of intervention, and disease state being studied. Secondary objectives included describing patterns of clinical trial distribution within the United States in the context of melanoma mortality and examining changing trends in interventions studied in trials over time.
ClinicalTrials.gov is a comprehensive online registry of clinical trials conducted in the United States and abroad that is maintained by the National Library of Medicine.7 Although the initiative was launched in 2000, the registry became effectively comprehensive in September 2005 when the International Committee of Medical Journal Editors declared prospective registration of clinical trials as a prerequisite for publication. The US Food and Drug Administration followed suit in September 2007, expanding the requirements for registration and declaring penalties for parties who did not comply.8 Each registered trial can be found through searchable keywords, and each study page contains details of study design, principal investigators, and inclusion and exclusion criteria, as well as contact information for enrollment.
Study Selection
Clinical trials registered between September 15, 2005, and December 31, 2013, were evaluated; a total of 138,312 trials were found to be registered on ClinicalTrials.gov during that time period. We limited our study selection to interventional studies, which were filtered by topic to yield only those pertaining to melanoma patients. To minimize reporting bias, trials registered prior to the implementation of the International Committee of Medical Journal Editors’ reporting requirements were excluded. To focus specifically on the landscape of trials in cutaneous melanoma, trials investigating multiple advanced malignancies, uveal or ocular melanoma, and mucosal melanoma were manually excluded.
Study Variables
Information on each clinical trial was extracted from ClinicalTrials.gov. Each trial was manually reviewed by an investigator to determine the disease state and type of intervention being studied. Studies investigating multiple modalities concurrently were classified as “other.”
Data Analysis
Study variables were first analyzed among the entire cohort as a whole. Using each trial location and a python script based on open-source code, the number of actively recruiting melanoma trials in each US county was identified and mapped. County-level, melanoma-specific mortality data from 2001 to 2010 was extracted from the Centers for Disease Control and Prevention’s WONDER (Wide-ranging Online Data for Epidemiologic Research) mortality database (wonder.cdc.gov). Finally, to analyze changing trends in cutaneous melanoma investigation, trials were grouped into 3 categories based on the date they were received on ClinicalTrials.gov: (1) 2005-2007, (2) 2008-2010, and (3) 2011-2013. Disease state and type of intervention were analyzed and compared among each group using the χ2 statistic.
Results
Of the 138,312 trials registered on ClinicalTrials.gov between September 15, 2005, and December 31, 2013, only 931 were identified as interventional studies pertaining to melanoma patients. Of these, 154 were excluded because of a focus on uveal, ocular, or mucosal melanoma or because of the inclusion of participants with multiple types of advanced malignancies. The final analysis included 777 trials specifically focusing on cutaneous melanoma.
Characteristics of these 777 trials were varied. Many interventions were in the early stages of development, with 339 (44%) trials classified as phase 0, phase 1, or phase 1/phase 2; 306 (39%) as phase 2; and 71 (9%) as nonpharmacologic (nonphase) trials. Only 58 trials (8%) were classified as phase 3 or phase 4. The majority of the trials were actively recruiting (225 [29%]), active but not yet recruiting (172 [22%]), or completed (255 [33%]); however, 98 trials (13%) had been suspended, terminated, or withdrawn. Additionally, 22 trials (3%) were not yet recruiting and 5 (<1%) were classified as “other” because they did not have a recruitment status listed.
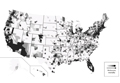
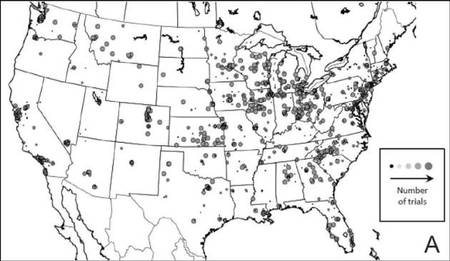
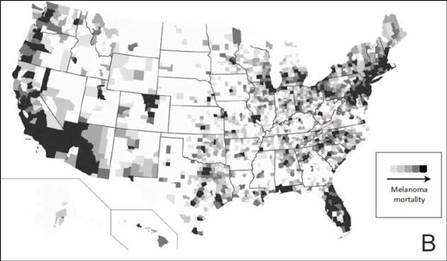
The distribution of actively enrolling clinical trials corresponds to major metropolitan areas within the Northeast, Upper Midwest, and Coastal California (Figure 1A). Figure 1B demonstrates the melanoma-specific mortality across the United States. Areas in the Southwest and Florida shared some of the greatest disease burden.
|
| Figure 1. Geographical representation of US clinical trial enrollment with the number of actively recruiting trials for each unique US zip code presented. The circle size corresponds to the number of trials. The largest circles indicate more than 5 trials within a given zip code (A). County-level melanoma-specific mortality data are presented for 2001 to 2010 (B). Darkest areas represent the highest numbers of melanoma deaths.
|
The disease state and type of intervention for all the included trials are summarized in Figure 2. The vast majority of trials (633/777 [82%]) enrolled participants with metastatic melanoma. Unlike many other tumor types, only 64 (8%) trials enrolled patients specifically in the adjuvant setting. Most trials focused on targeted (175 [23%]), immune (180 [23%]), and vaccine (117 [15%]) therapy.
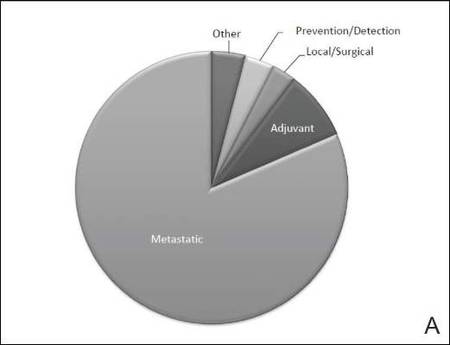
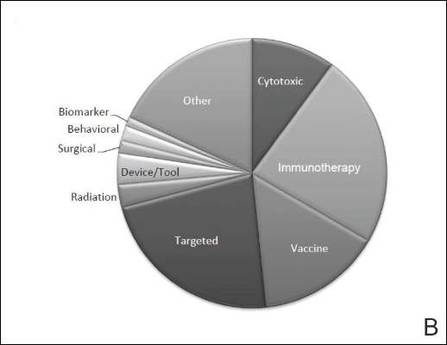
|
| Figure 2. Trial distribution stratified by disease state (A) and type of intervention (B). Trial distribution is shown for 777 interventional clinical trials including melanoma patients. The majority of clinical trials involved patients with metastatic melanoma. The majority of trials investigated targeted therapy, immunotherapy, and vaccine therapy. |
We subsequently analyzed changes in trial characteristics over time. We noted a decrease in the number of trials investigating cytotoxic and vaccine-based therapies, and increasing numbers of trials investigating immunotherapy (P=.041). Between 2005 and 2007, 14% (27/201) of all trials investigated cytotoxic therapies compared to just 7% (20/294) of trials between 2011 and 2013. With the approval of ipilimumab, 29% (85/294) of all clinical trials between 2011 and 2013 investigated immunotherapies, which comprised only 18% (37/201) of clinical trials between 2005 and 2007. The majority of trials continued to enroll patients in the metastatic setting where outcomes remain poor. Importantly, only 6% (49/777) of all clinical trials have focused on prevention, early detection, and local management of melanoma, which has remained constant over time.
Comment
Cutaneous melanoma remains an area of active investigation, interdisciplinary collaboration, and great promise. The ClinicalTrials.gov registry serves not only to increase transparency among interested parties but also as a rich resource to study the clinical research landscape as demonstrated in this study.
Greater understanding of the underlying genetic and immunogenic properties of melanoma tissues has led to the US Food and Drug Administration approval of several novel agents to treat metastatic disease. BRAF inhibitors such as vemurafenib and dabrafenib target more than 50% of all melanoma tumors harboring mutations in the BRAF gene and have shown unparalleled efficacy in clinical trials; however, durability of response and adverse effects still remain a concern.4,9-11 Ipilimumab, a CTLA-4 inhibitor, enhances antitumor immunity and demonstrated improved survival in clinical trials.12,13 Nivolumab, a fully human IgG4 programmed death 1 (PD-1) immune-checkpoint inhibitor antibody, also demonstrated improved overall and progression-free survival.14 Finally, trametinib, a MEK inhibitor, used in combination with BRAF inhibitors has demonstrated improved response over BRAF inhibitors alone.15
Although traditional cytotoxic chemotherapy was one of the few available treatment options before 2011, response was infrequent.16 Our data indicate that the melanoma research landscape has shifted to follow advances in targeted therapy and immunotherapy. We noted a decrease in the study of cytotoxic chemotherapy in metastatic melanoma, with a compensatory increase in immunotherapy trials and a continued commitment to targeted therapy. Further, with the approval of BRAF inhibitors, CTLA-4 inhibitors, and PD-1 inhibitors for metastatic disease, some have pushed to move these agents into the adjuvant setting to prevent micrometastases from evolving into clinically significant disease.17 Early results from EORTC (European Organisation for Research and Treatment of Cancer) 18071 comparing adjuvant ipilimumab to placebo demonstrated a 26.1-month versus 17.1-month improvement in relapse-free survival, respectively.18 However, this finding has important implications for clinical dermatologists. Patients treated with BRAF inhibitors are at increased risk for keratoacanthomas, invasive squamous cell carcinomas, and secondary primary melanomas.19,20 Caring for these patients requires increased vigilance and collaboration between dermatologists and oncologists.
Our study also highlights the dynamic nature of the field. For example, novel vaccine therapies have demonstrated promise in the metastatic/ unresectable tumor setting, with some herpes simplex virus–based vaccines generating durable antitumor immune responses in patients with melanoma.21 Combination therapy with CTLA-4 and PD-1 inhibitors has demonstrated improved objective response rates and progression-free survival over monotherapy.22 As the status of actively recruiting trials changes on a regular basis, we encourage physicians to access ClinicalTrials.gov to find details and contact information for actively recruiting trials and results on completed trials.
Early detection and management, however, still remain our primary option for cure, and the role of community dermatologists cannot be overstated.23 Patients with stage I and stage II disease have excellent long-term survival rates, yet only 6% of all clinical trials in cutaneous melanoma have focused on patient education, disease prevention, early detection, and local management. With an increasing incidence of melanoma among an aging population, the disease burden remains of substantial concern.24 Optimizing disease prevention, appropriate screening, and early detection are critical roles for dermatologists.
Finally, our data offer some insight into accrual barriers often faced by clinical trials. Actively enrolling clinical trials cluster within major metropolitan areas, presumably with large academic medical centers; however, areas in the southwestern United States and Florida, for example, have some of the highest burden of disease, likely secondary to sun exposure and aging populations.25 Integration of community dermatologists and oncologists may decrease both actual and patient-perceived barriers to care, which requires further exploration.6
Conclusion
Melanoma incidence and disease burden is increasing, and the field of melanoma research is incredibly dynamic. Going forward, we believe dermatologists will continue to play a critical role both in primary disease prevention and detection as well as in detection of secondary treatment-related skin toxicities. ClinicalTrials.gov is an invaluable resource to keep interested parties informed, foster collaboration, identify potential barriers to success, and suggest future directions.
1. DeSantis CE, Lin CC, Mariotto AB, et al. Cancer treatment and survivorship statistics, 2014. CA Cancer J Clin. 2014;64:252-271.
2. American Cancer Society. Cancer Facts and Figures 2015. American Cancer Society Web site. http://www.cancer.org/acs/groups/content/@editorial/documents/document/acspc-044552.pdf. Accessed August 26, 2015.
3. Cheng MY, Moreau JF, McGuire ST, et al. Melanoma depth in patients with an established dermatologist. J Am Acad Dermatol. 2014;70:841-846.
4. Jang S, Atkins MB. Which drug, and when, for patients with BRAF-mutant melanoma? Lancet Oncol. 2013;14:e60-e69.
5. Kim SH, Tanner A, Friedman DB, et al. Barriers to clinical trial participation: a comparison of rural and urban communities in South Carolina. J Community Health. 2014;39:562-571.
6. Gregg JR, Horn L, Davidson MA, et al. Patient enrollment onto clinical trials: the role of physician knowledge. J Cancer Educ. 2014;29:74-79.
7. Galsky MD, Hendricks R, Svatek R, et al. Critical analysis of contemporary clinical research in muscle-invasive and metastatic urothelial cancer: a report from the
Bladder Cancer Advocacy Network Clinical Trials Working Group. Cancer. 2013;119:1994-1998.
8. Zarin DA, Tse T, Williams RJ, et al. The ClinicalTrials.gov results database—update and key issues. N Engl J Med. 2011;364:852-860.
9. Luke JJ, Hodi FS. Ipilimumab, vemurafenib, dabrafenib, and trametinib: synergistic competitors in the clinical management of BRAF mutant malignant melanoma. Oncologist. 2013;18:717-725.
10. McArthur GA, Chapman PB, Robert C, et al. Safety and efficacy of vemurafenib in BRAF(V600E) and BRAF(V600K) mutation-positive melanoma (BRIM-3): extended follow-up of a phase 3, randomised, open-label study. Lancet Oncol. 2014;15:323-332.
11. Swaika A, Crozier JA, Joseph RW. Vemurafenib: an evidence-based review of its clinical utility in the treatment of metastatic melanoma. Drug Des Devel Ther. 2014;8:775-787.
12. Lacouture ME, Wolchok JD, Yosipovitch G, et al. Ipilimumab in patients with cancer and the management of dermatologic adverse events. J Am Acad Dermatol. 2014;71:161-169.
13. Hodi FS, O’Day SJ, McDermott DF, et al. Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med. 2010;363:711-723.
14. Robert C, Long G, Brady B, et al. Nivolumab in previously untreated melanoma without BRAF mutation. N Engl J Med. 2015;372:320-330.
15. Robert C, Karaszewska B, Schachter J, et al. Improved overall survival in melanoma with combined dabrafenib and trametinib. N Engl J Med. 2015;372:30-39.
16. Espinosa E, Berrocal A, López Martin JA, et al. Advances in cutaneous melanoma. Clin Transl Oncol. 2012;14:325-332.
17. Chapman PB. Treating metastatic melanoma in 2014: what just happened and what is next? Am Soc Clin Oncol Educ Book. 2014:16-19.
18. Eggermont A, Chiarion-Sileni V, Grob JJ, et al. Adjuvant ipilimumab versus placebo after complete resection of high-risk stage III melanoma (EORTC 18071): a randomised, double-blind, phase 3 trial. Lancet Oncol. 2015;16:522-530.
19. Curry JL, Tetzlaff MT, Nicholson K, et al. Histological features associated with vemurafenib-induced skin toxicities: examination of 141 cutaneous lesions biopsied during therapy. Am J Dermatopathol. 2014; 36:557-561.
20. Perier-Muzet M, Thomas L, Poulalhon N, et al. Melanoma patients under vemurafenib: prospective follow-up of melanocytic lesions by digital dermoscopy. J Invest Dermatol. 2014;134:1351-1358.
21. Ross MI, Andtbacka RI, Puzanov I, et al. Patterns of durable response with intralesional talimogene laherparepvec (T-VEC): results from a phase III trial in patients with stage IIIb-IV melanoma. Paper presented at: ASCO Annual Meeting; June 2, 2014; Boston, MA.
22. Postow MA, Chesney J, Pavlick AC, et al. Novolumab and ipilimumab versus ipilimumab in untreated melanoma. N Engl J Med. 2015;372:2006-2017.
23. Gorantla VC, Kirkwood JM. State of melanoma: an historic overview of a field in transition. Hematol Oncol Clin North Am. 2014;28:415-435.
24. Coit DG, Olszanski AJ. Progress in the management of melanoma in 2013. J Natl Compr Canc Netw. 2013; 11(5 suppl):645-648.
25. Watson M, Johnson CJ, Chen VW, et al. Melanoma surveillance in the United States: overview of methods. J Am Acad Dermatol. 2011;65(5, suppl 1):S6-S16.
The incidence of cutaneous melanoma, the deadliest form of skin cancer, has been steadily increasing over the last several decades.1 Currently, there are 73,870 new diagnoses of melanoma anticipated in the United States in 2015 alone.2 Many cases of melanoma are caught at early, actionable, and curable stages thanks in part to patient education and screening by dermatologists.3 However, until recently, few options existed for the treatment of locally advanced and metastatic melanomas, with a median survival rate of less than 1 year.4
Clinical trials represent the most reliable method for advancing treatment and improving outcomes for patients with disease; however, patient accrual and access to clinical trials remain formidable barriers. Studies have suggested that patients in rural areas perceive both an increased distance to clinical trial sites and a lack of awareness of available trials compared to their urban counterparts. Additionally, studies have shown that provider awareness of actively enrolling clinical trials in their respective fields is a key determinate in patient enrollment.5 Finally, insufficient funding and lack of collaboration has resulted in many small phase 1 or phase 2 single-center trials, which are less likely to quickly impact clinical care.6 Increased awareness of the ClinicalTrials.gov registry, a publicly available and easily accessible database, can facilitate referral, enrollment, and collaboration among physicians, patients, and researchers alike.
Using the ClinicalTrials.gov database, we sought to analyze the clinical trial landscape for cutaneous melanoma to understand the current state of melanoma research, future direction, and potential barriers that may impede success.
Methods
The primary objective was to provide a snapshot of the melanoma clinical research landscape from 2005 to 2013, including the number of registered trials, phase distribution, recruitment status, location of trials, type of intervention, and disease state being studied. Secondary objectives included describing patterns of clinical trial distribution within the United States in the context of melanoma mortality and examining changing trends in interventions studied in trials over time.
ClinicalTrials.gov is a comprehensive online registry of clinical trials conducted in the United States and abroad that is maintained by the National Library of Medicine.7 Although the initiative was launched in 2000, the registry became effectively comprehensive in September 2005 when the International Committee of Medical Journal Editors declared prospective registration of clinical trials as a prerequisite for publication. The US Food and Drug Administration followed suit in September 2007, expanding the requirements for registration and declaring penalties for parties who did not comply.8 Each registered trial can be found through searchable keywords, and each study page contains details of study design, principal investigators, and inclusion and exclusion criteria, as well as contact information for enrollment.
Study Selection
Clinical trials registered between September 15, 2005, and December 31, 2013, were evaluated; a total of 138,312 trials were found to be registered on ClinicalTrials.gov during that time period. We limited our study selection to interventional studies, which were filtered by topic to yield only those pertaining to melanoma patients. To minimize reporting bias, trials registered prior to the implementation of the International Committee of Medical Journal Editors’ reporting requirements were excluded. To focus specifically on the landscape of trials in cutaneous melanoma, trials investigating multiple advanced malignancies, uveal or ocular melanoma, and mucosal melanoma were manually excluded.
Study Variables
Information on each clinical trial was extracted from ClinicalTrials.gov. Each trial was manually reviewed by an investigator to determine the disease state and type of intervention being studied. Studies investigating multiple modalities concurrently were classified as “other.”
Data Analysis
Study variables were first analyzed among the entire cohort as a whole. Using each trial location and a python script based on open-source code, the number of actively recruiting melanoma trials in each US county was identified and mapped. County-level, melanoma-specific mortality data from 2001 to 2010 was extracted from the Centers for Disease Control and Prevention’s WONDER (Wide-ranging Online Data for Epidemiologic Research) mortality database (wonder.cdc.gov). Finally, to analyze changing trends in cutaneous melanoma investigation, trials were grouped into 3 categories based on the date they were received on ClinicalTrials.gov: (1) 2005-2007, (2) 2008-2010, and (3) 2011-2013. Disease state and type of intervention were analyzed and compared among each group using the χ2 statistic.
Results
Of the 138,312 trials registered on ClinicalTrials.gov between September 15, 2005, and December 31, 2013, only 931 were identified as interventional studies pertaining to melanoma patients. Of these, 154 were excluded because of a focus on uveal, ocular, or mucosal melanoma or because of the inclusion of participants with multiple types of advanced malignancies. The final analysis included 777 trials specifically focusing on cutaneous melanoma.
Characteristics of these 777 trials were varied. Many interventions were in the early stages of development, with 339 (44%) trials classified as phase 0, phase 1, or phase 1/phase 2; 306 (39%) as phase 2; and 71 (9%) as nonpharmacologic (nonphase) trials. Only 58 trials (8%) were classified as phase 3 or phase 4. The majority of the trials were actively recruiting (225 [29%]), active but not yet recruiting (172 [22%]), or completed (255 [33%]); however, 98 trials (13%) had been suspended, terminated, or withdrawn. Additionally, 22 trials (3%) were not yet recruiting and 5 (<1%) were classified as “other” because they did not have a recruitment status listed.
The distribution of actively enrolling clinical trials corresponds to major metropolitan areas within the Northeast, Upper Midwest, and Coastal California (Figure 1A). Figure 1B demonstrates the melanoma-specific mortality across the United States. Areas in the Southwest and Florida shared some of the greatest disease burden.


|
| Figure 1. Geographical representation of US clinical trial enrollment with the number of actively recruiting trials for each unique US zip code presented. The circle size corresponds to the number of trials. The largest circles indicate more than 5 trials within a given zip code (A). County-level melanoma-specific mortality data are presented for 2001 to 2010 (B). Darkest areas represent the highest numbers of melanoma deaths.
|
The disease state and type of intervention for all the included trials are summarized in Figure 2. The vast majority of trials (633/777 [82%]) enrolled participants with metastatic melanoma. Unlike many other tumor types, only 64 (8%) trials enrolled patients specifically in the adjuvant setting. Most trials focused on targeted (175 [23%]), immune (180 [23%]), and vaccine (117 [15%]) therapy.


|
| Figure 2. Trial distribution stratified by disease state (A) and type of intervention (B). Trial distribution is shown for 777 interventional clinical trials including melanoma patients. The majority of clinical trials involved patients with metastatic melanoma. The majority of trials investigated targeted therapy, immunotherapy, and vaccine therapy. |
We subsequently analyzed changes in trial characteristics over time. We noted a decrease in the number of trials investigating cytotoxic and vaccine-based therapies, and increasing numbers of trials investigating immunotherapy (P=.041). Between 2005 and 2007, 14% (27/201) of all trials investigated cytotoxic therapies compared to just 7% (20/294) of trials between 2011 and 2013. With the approval of ipilimumab, 29% (85/294) of all clinical trials between 2011 and 2013 investigated immunotherapies, which comprised only 18% (37/201) of clinical trials between 2005 and 2007. The majority of trials continued to enroll patients in the metastatic setting where outcomes remain poor. Importantly, only 6% (49/777) of all clinical trials have focused on prevention, early detection, and local management of melanoma, which has remained constant over time.
Comment
Cutaneous melanoma remains an area of active investigation, interdisciplinary collaboration, and great promise. The ClinicalTrials.gov registry serves not only to increase transparency among interested parties but also as a rich resource to study the clinical research landscape as demonstrated in this study.
Greater understanding of the underlying genetic and immunogenic properties of melanoma tissues has led to the US Food and Drug Administration approval of several novel agents to treat metastatic disease. BRAF inhibitors such as vemurafenib and dabrafenib target more than 50% of all melanoma tumors harboring mutations in the BRAF gene and have shown unparalleled efficacy in clinical trials; however, durability of response and adverse effects still remain a concern.4,9-11 Ipilimumab, a CTLA-4 inhibitor, enhances antitumor immunity and demonstrated improved survival in clinical trials.12,13 Nivolumab, a fully human IgG4 programmed death 1 (PD-1) immune-checkpoint inhibitor antibody, also demonstrated improved overall and progression-free survival.14 Finally, trametinib, a MEK inhibitor, used in combination with BRAF inhibitors has demonstrated improved response over BRAF inhibitors alone.15
Although traditional cytotoxic chemotherapy was one of the few available treatment options before 2011, response was infrequent.16 Our data indicate that the melanoma research landscape has shifted to follow advances in targeted therapy and immunotherapy. We noted a decrease in the study of cytotoxic chemotherapy in metastatic melanoma, with a compensatory increase in immunotherapy trials and a continued commitment to targeted therapy. Further, with the approval of BRAF inhibitors, CTLA-4 inhibitors, and PD-1 inhibitors for metastatic disease, some have pushed to move these agents into the adjuvant setting to prevent micrometastases from evolving into clinically significant disease.17 Early results from EORTC (European Organisation for Research and Treatment of Cancer) 18071 comparing adjuvant ipilimumab to placebo demonstrated a 26.1-month versus 17.1-month improvement in relapse-free survival, respectively.18 However, this finding has important implications for clinical dermatologists. Patients treated with BRAF inhibitors are at increased risk for keratoacanthomas, invasive squamous cell carcinomas, and secondary primary melanomas.19,20 Caring for these patients requires increased vigilance and collaboration between dermatologists and oncologists.
Our study also highlights the dynamic nature of the field. For example, novel vaccine therapies have demonstrated promise in the metastatic/ unresectable tumor setting, with some herpes simplex virus–based vaccines generating durable antitumor immune responses in patients with melanoma.21 Combination therapy with CTLA-4 and PD-1 inhibitors has demonstrated improved objective response rates and progression-free survival over monotherapy.22 As the status of actively recruiting trials changes on a regular basis, we encourage physicians to access ClinicalTrials.gov to find details and contact information for actively recruiting trials and results on completed trials.
Early detection and management, however, still remain our primary option for cure, and the role of community dermatologists cannot be overstated.23 Patients with stage I and stage II disease have excellent long-term survival rates, yet only 6% of all clinical trials in cutaneous melanoma have focused on patient education, disease prevention, early detection, and local management. With an increasing incidence of melanoma among an aging population, the disease burden remains of substantial concern.24 Optimizing disease prevention, appropriate screening, and early detection are critical roles for dermatologists.
Finally, our data offer some insight into accrual barriers often faced by clinical trials. Actively enrolling clinical trials cluster within major metropolitan areas, presumably with large academic medical centers; however, areas in the southwestern United States and Florida, for example, have some of the highest burden of disease, likely secondary to sun exposure and aging populations.25 Integration of community dermatologists and oncologists may decrease both actual and patient-perceived barriers to care, which requires further exploration.6
Conclusion
Melanoma incidence and disease burden is increasing, and the field of melanoma research is incredibly dynamic. Going forward, we believe dermatologists will continue to play a critical role both in primary disease prevention and detection as well as in detection of secondary treatment-related skin toxicities. ClinicalTrials.gov is an invaluable resource to keep interested parties informed, foster collaboration, identify potential barriers to success, and suggest future directions.
The incidence of cutaneous melanoma, the deadliest form of skin cancer, has been steadily increasing over the last several decades.1 Currently, there are 73,870 new diagnoses of melanoma anticipated in the United States in 2015 alone.2 Many cases of melanoma are caught at early, actionable, and curable stages thanks in part to patient education and screening by dermatologists.3 However, until recently, few options existed for the treatment of locally advanced and metastatic melanomas, with a median survival rate of less than 1 year.4
Clinical trials represent the most reliable method for advancing treatment and improving outcomes for patients with disease; however, patient accrual and access to clinical trials remain formidable barriers. Studies have suggested that patients in rural areas perceive both an increased distance to clinical trial sites and a lack of awareness of available trials compared to their urban counterparts. Additionally, studies have shown that provider awareness of actively enrolling clinical trials in their respective fields is a key determinate in patient enrollment.5 Finally, insufficient funding and lack of collaboration has resulted in many small phase 1 or phase 2 single-center trials, which are less likely to quickly impact clinical care.6 Increased awareness of the ClinicalTrials.gov registry, a publicly available and easily accessible database, can facilitate referral, enrollment, and collaboration among physicians, patients, and researchers alike.
Using the ClinicalTrials.gov database, we sought to analyze the clinical trial landscape for cutaneous melanoma to understand the current state of melanoma research, future direction, and potential barriers that may impede success.
Methods
The primary objective was to provide a snapshot of the melanoma clinical research landscape from 2005 to 2013, including the number of registered trials, phase distribution, recruitment status, location of trials, type of intervention, and disease state being studied. Secondary objectives included describing patterns of clinical trial distribution within the United States in the context of melanoma mortality and examining changing trends in interventions studied in trials over time.
ClinicalTrials.gov is a comprehensive online registry of clinical trials conducted in the United States and abroad that is maintained by the National Library of Medicine.7 Although the initiative was launched in 2000, the registry became effectively comprehensive in September 2005 when the International Committee of Medical Journal Editors declared prospective registration of clinical trials as a prerequisite for publication. The US Food and Drug Administration followed suit in September 2007, expanding the requirements for registration and declaring penalties for parties who did not comply.8 Each registered trial can be found through searchable keywords, and each study page contains details of study design, principal investigators, and inclusion and exclusion criteria, as well as contact information for enrollment.
Study Selection
Clinical trials registered between September 15, 2005, and December 31, 2013, were evaluated; a total of 138,312 trials were found to be registered on ClinicalTrials.gov during that time period. We limited our study selection to interventional studies, which were filtered by topic to yield only those pertaining to melanoma patients. To minimize reporting bias, trials registered prior to the implementation of the International Committee of Medical Journal Editors’ reporting requirements were excluded. To focus specifically on the landscape of trials in cutaneous melanoma, trials investigating multiple advanced malignancies, uveal or ocular melanoma, and mucosal melanoma were manually excluded.
Study Variables
Information on each clinical trial was extracted from ClinicalTrials.gov. Each trial was manually reviewed by an investigator to determine the disease state and type of intervention being studied. Studies investigating multiple modalities concurrently were classified as “other.”
Data Analysis
Study variables were first analyzed among the entire cohort as a whole. Using each trial location and a python script based on open-source code, the number of actively recruiting melanoma trials in each US county was identified and mapped. County-level, melanoma-specific mortality data from 2001 to 2010 was extracted from the Centers for Disease Control and Prevention’s WONDER (Wide-ranging Online Data for Epidemiologic Research) mortality database (wonder.cdc.gov). Finally, to analyze changing trends in cutaneous melanoma investigation, trials were grouped into 3 categories based on the date they were received on ClinicalTrials.gov: (1) 2005-2007, (2) 2008-2010, and (3) 2011-2013. Disease state and type of intervention were analyzed and compared among each group using the χ2 statistic.
Results
Of the 138,312 trials registered on ClinicalTrials.gov between September 15, 2005, and December 31, 2013, only 931 were identified as interventional studies pertaining to melanoma patients. Of these, 154 were excluded because of a focus on uveal, ocular, or mucosal melanoma or because of the inclusion of participants with multiple types of advanced malignancies. The final analysis included 777 trials specifically focusing on cutaneous melanoma.
Characteristics of these 777 trials were varied. Many interventions were in the early stages of development, with 339 (44%) trials classified as phase 0, phase 1, or phase 1/phase 2; 306 (39%) as phase 2; and 71 (9%) as nonpharmacologic (nonphase) trials. Only 58 trials (8%) were classified as phase 3 or phase 4. The majority of the trials were actively recruiting (225 [29%]), active but not yet recruiting (172 [22%]), or completed (255 [33%]); however, 98 trials (13%) had been suspended, terminated, or withdrawn. Additionally, 22 trials (3%) were not yet recruiting and 5 (<1%) were classified as “other” because they did not have a recruitment status listed.
The distribution of actively enrolling clinical trials corresponds to major metropolitan areas within the Northeast, Upper Midwest, and Coastal California (Figure 1A). Figure 1B demonstrates the melanoma-specific mortality across the United States. Areas in the Southwest and Florida shared some of the greatest disease burden.


|
| Figure 1. Geographical representation of US clinical trial enrollment with the number of actively recruiting trials for each unique US zip code presented. The circle size corresponds to the number of trials. The largest circles indicate more than 5 trials within a given zip code (A). County-level melanoma-specific mortality data are presented for 2001 to 2010 (B). Darkest areas represent the highest numbers of melanoma deaths.
|
The disease state and type of intervention for all the included trials are summarized in Figure 2. The vast majority of trials (633/777 [82%]) enrolled participants with metastatic melanoma. Unlike many other tumor types, only 64 (8%) trials enrolled patients specifically in the adjuvant setting. Most trials focused on targeted (175 [23%]), immune (180 [23%]), and vaccine (117 [15%]) therapy.


|
| Figure 2. Trial distribution stratified by disease state (A) and type of intervention (B). Trial distribution is shown for 777 interventional clinical trials including melanoma patients. The majority of clinical trials involved patients with metastatic melanoma. The majority of trials investigated targeted therapy, immunotherapy, and vaccine therapy. |
We subsequently analyzed changes in trial characteristics over time. We noted a decrease in the number of trials investigating cytotoxic and vaccine-based therapies, and increasing numbers of trials investigating immunotherapy (P=.041). Between 2005 and 2007, 14% (27/201) of all trials investigated cytotoxic therapies compared to just 7% (20/294) of trials between 2011 and 2013. With the approval of ipilimumab, 29% (85/294) of all clinical trials between 2011 and 2013 investigated immunotherapies, which comprised only 18% (37/201) of clinical trials between 2005 and 2007. The majority of trials continued to enroll patients in the metastatic setting where outcomes remain poor. Importantly, only 6% (49/777) of all clinical trials have focused on prevention, early detection, and local management of melanoma, which has remained constant over time.
Comment
Cutaneous melanoma remains an area of active investigation, interdisciplinary collaboration, and great promise. The ClinicalTrials.gov registry serves not only to increase transparency among interested parties but also as a rich resource to study the clinical research landscape as demonstrated in this study.
Greater understanding of the underlying genetic and immunogenic properties of melanoma tissues has led to the US Food and Drug Administration approval of several novel agents to treat metastatic disease. BRAF inhibitors such as vemurafenib and dabrafenib target more than 50% of all melanoma tumors harboring mutations in the BRAF gene and have shown unparalleled efficacy in clinical trials; however, durability of response and adverse effects still remain a concern.4,9-11 Ipilimumab, a CTLA-4 inhibitor, enhances antitumor immunity and demonstrated improved survival in clinical trials.12,13 Nivolumab, a fully human IgG4 programmed death 1 (PD-1) immune-checkpoint inhibitor antibody, also demonstrated improved overall and progression-free survival.14 Finally, trametinib, a MEK inhibitor, used in combination with BRAF inhibitors has demonstrated improved response over BRAF inhibitors alone.15
Although traditional cytotoxic chemotherapy was one of the few available treatment options before 2011, response was infrequent.16 Our data indicate that the melanoma research landscape has shifted to follow advances in targeted therapy and immunotherapy. We noted a decrease in the study of cytotoxic chemotherapy in metastatic melanoma, with a compensatory increase in immunotherapy trials and a continued commitment to targeted therapy. Further, with the approval of BRAF inhibitors, CTLA-4 inhibitors, and PD-1 inhibitors for metastatic disease, some have pushed to move these agents into the adjuvant setting to prevent micrometastases from evolving into clinically significant disease.17 Early results from EORTC (European Organisation for Research and Treatment of Cancer) 18071 comparing adjuvant ipilimumab to placebo demonstrated a 26.1-month versus 17.1-month improvement in relapse-free survival, respectively.18 However, this finding has important implications for clinical dermatologists. Patients treated with BRAF inhibitors are at increased risk for keratoacanthomas, invasive squamous cell carcinomas, and secondary primary melanomas.19,20 Caring for these patients requires increased vigilance and collaboration between dermatologists and oncologists.
Our study also highlights the dynamic nature of the field. For example, novel vaccine therapies have demonstrated promise in the metastatic/ unresectable tumor setting, with some herpes simplex virus–based vaccines generating durable antitumor immune responses in patients with melanoma.21 Combination therapy with CTLA-4 and PD-1 inhibitors has demonstrated improved objective response rates and progression-free survival over monotherapy.22 As the status of actively recruiting trials changes on a regular basis, we encourage physicians to access ClinicalTrials.gov to find details and contact information for actively recruiting trials and results on completed trials.
Early detection and management, however, still remain our primary option for cure, and the role of community dermatologists cannot be overstated.23 Patients with stage I and stage II disease have excellent long-term survival rates, yet only 6% of all clinical trials in cutaneous melanoma have focused on patient education, disease prevention, early detection, and local management. With an increasing incidence of melanoma among an aging population, the disease burden remains of substantial concern.24 Optimizing disease prevention, appropriate screening, and early detection are critical roles for dermatologists.
Finally, our data offer some insight into accrual barriers often faced by clinical trials. Actively enrolling clinical trials cluster within major metropolitan areas, presumably with large academic medical centers; however, areas in the southwestern United States and Florida, for example, have some of the highest burden of disease, likely secondary to sun exposure and aging populations.25 Integration of community dermatologists and oncologists may decrease both actual and patient-perceived barriers to care, which requires further exploration.6
Conclusion
Melanoma incidence and disease burden is increasing, and the field of melanoma research is incredibly dynamic. Going forward, we believe dermatologists will continue to play a critical role both in primary disease prevention and detection as well as in detection of secondary treatment-related skin toxicities. ClinicalTrials.gov is an invaluable resource to keep interested parties informed, foster collaboration, identify potential barriers to success, and suggest future directions.
1. DeSantis CE, Lin CC, Mariotto AB, et al. Cancer treatment and survivorship statistics, 2014. CA Cancer J Clin. 2014;64:252-271.
2. American Cancer Society. Cancer Facts and Figures 2015. American Cancer Society Web site. http://www.cancer.org/acs/groups/content/@editorial/documents/document/acspc-044552.pdf. Accessed August 26, 2015.
3. Cheng MY, Moreau JF, McGuire ST, et al. Melanoma depth in patients with an established dermatologist. J Am Acad Dermatol. 2014;70:841-846.
4. Jang S, Atkins MB. Which drug, and when, for patients with BRAF-mutant melanoma? Lancet Oncol. 2013;14:e60-e69.
5. Kim SH, Tanner A, Friedman DB, et al. Barriers to clinical trial participation: a comparison of rural and urban communities in South Carolina. J Community Health. 2014;39:562-571.
6. Gregg JR, Horn L, Davidson MA, et al. Patient enrollment onto clinical trials: the role of physician knowledge. J Cancer Educ. 2014;29:74-79.
7. Galsky MD, Hendricks R, Svatek R, et al. Critical analysis of contemporary clinical research in muscle-invasive and metastatic urothelial cancer: a report from the
Bladder Cancer Advocacy Network Clinical Trials Working Group. Cancer. 2013;119:1994-1998.
8. Zarin DA, Tse T, Williams RJ, et al. The ClinicalTrials.gov results database—update and key issues. N Engl J Med. 2011;364:852-860.
9. Luke JJ, Hodi FS. Ipilimumab, vemurafenib, dabrafenib, and trametinib: synergistic competitors in the clinical management of BRAF mutant malignant melanoma. Oncologist. 2013;18:717-725.
10. McArthur GA, Chapman PB, Robert C, et al. Safety and efficacy of vemurafenib in BRAF(V600E) and BRAF(V600K) mutation-positive melanoma (BRIM-3): extended follow-up of a phase 3, randomised, open-label study. Lancet Oncol. 2014;15:323-332.
11. Swaika A, Crozier JA, Joseph RW. Vemurafenib: an evidence-based review of its clinical utility in the treatment of metastatic melanoma. Drug Des Devel Ther. 2014;8:775-787.
12. Lacouture ME, Wolchok JD, Yosipovitch G, et al. Ipilimumab in patients with cancer and the management of dermatologic adverse events. J Am Acad Dermatol. 2014;71:161-169.
13. Hodi FS, O’Day SJ, McDermott DF, et al. Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med. 2010;363:711-723.
14. Robert C, Long G, Brady B, et al. Nivolumab in previously untreated melanoma without BRAF mutation. N Engl J Med. 2015;372:320-330.
15. Robert C, Karaszewska B, Schachter J, et al. Improved overall survival in melanoma with combined dabrafenib and trametinib. N Engl J Med. 2015;372:30-39.
16. Espinosa E, Berrocal A, López Martin JA, et al. Advances in cutaneous melanoma. Clin Transl Oncol. 2012;14:325-332.
17. Chapman PB. Treating metastatic melanoma in 2014: what just happened and what is next? Am Soc Clin Oncol Educ Book. 2014:16-19.
18. Eggermont A, Chiarion-Sileni V, Grob JJ, et al. Adjuvant ipilimumab versus placebo after complete resection of high-risk stage III melanoma (EORTC 18071): a randomised, double-blind, phase 3 trial. Lancet Oncol. 2015;16:522-530.
19. Curry JL, Tetzlaff MT, Nicholson K, et al. Histological features associated with vemurafenib-induced skin toxicities: examination of 141 cutaneous lesions biopsied during therapy. Am J Dermatopathol. 2014; 36:557-561.
20. Perier-Muzet M, Thomas L, Poulalhon N, et al. Melanoma patients under vemurafenib: prospective follow-up of melanocytic lesions by digital dermoscopy. J Invest Dermatol. 2014;134:1351-1358.
21. Ross MI, Andtbacka RI, Puzanov I, et al. Patterns of durable response with intralesional talimogene laherparepvec (T-VEC): results from a phase III trial in patients with stage IIIb-IV melanoma. Paper presented at: ASCO Annual Meeting; June 2, 2014; Boston, MA.
22. Postow MA, Chesney J, Pavlick AC, et al. Novolumab and ipilimumab versus ipilimumab in untreated melanoma. N Engl J Med. 2015;372:2006-2017.
23. Gorantla VC, Kirkwood JM. State of melanoma: an historic overview of a field in transition. Hematol Oncol Clin North Am. 2014;28:415-435.
24. Coit DG, Olszanski AJ. Progress in the management of melanoma in 2013. J Natl Compr Canc Netw. 2013; 11(5 suppl):645-648.
25. Watson M, Johnson CJ, Chen VW, et al. Melanoma surveillance in the United States: overview of methods. J Am Acad Dermatol. 2011;65(5, suppl 1):S6-S16.
1. DeSantis CE, Lin CC, Mariotto AB, et al. Cancer treatment and survivorship statistics, 2014. CA Cancer J Clin. 2014;64:252-271.
2. American Cancer Society. Cancer Facts and Figures 2015. American Cancer Society Web site. http://www.cancer.org/acs/groups/content/@editorial/documents/document/acspc-044552.pdf. Accessed August 26, 2015.
3. Cheng MY, Moreau JF, McGuire ST, et al. Melanoma depth in patients with an established dermatologist. J Am Acad Dermatol. 2014;70:841-846.
4. Jang S, Atkins MB. Which drug, and when, for patients with BRAF-mutant melanoma? Lancet Oncol. 2013;14:e60-e69.
5. Kim SH, Tanner A, Friedman DB, et al. Barriers to clinical trial participation: a comparison of rural and urban communities in South Carolina. J Community Health. 2014;39:562-571.
6. Gregg JR, Horn L, Davidson MA, et al. Patient enrollment onto clinical trials: the role of physician knowledge. J Cancer Educ. 2014;29:74-79.
7. Galsky MD, Hendricks R, Svatek R, et al. Critical analysis of contemporary clinical research in muscle-invasive and metastatic urothelial cancer: a report from the
Bladder Cancer Advocacy Network Clinical Trials Working Group. Cancer. 2013;119:1994-1998.
8. Zarin DA, Tse T, Williams RJ, et al. The ClinicalTrials.gov results database—update and key issues. N Engl J Med. 2011;364:852-860.
9. Luke JJ, Hodi FS. Ipilimumab, vemurafenib, dabrafenib, and trametinib: synergistic competitors in the clinical management of BRAF mutant malignant melanoma. Oncologist. 2013;18:717-725.
10. McArthur GA, Chapman PB, Robert C, et al. Safety and efficacy of vemurafenib in BRAF(V600E) and BRAF(V600K) mutation-positive melanoma (BRIM-3): extended follow-up of a phase 3, randomised, open-label study. Lancet Oncol. 2014;15:323-332.
11. Swaika A, Crozier JA, Joseph RW. Vemurafenib: an evidence-based review of its clinical utility in the treatment of metastatic melanoma. Drug Des Devel Ther. 2014;8:775-787.
12. Lacouture ME, Wolchok JD, Yosipovitch G, et al. Ipilimumab in patients with cancer and the management of dermatologic adverse events. J Am Acad Dermatol. 2014;71:161-169.
13. Hodi FS, O’Day SJ, McDermott DF, et al. Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med. 2010;363:711-723.
14. Robert C, Long G, Brady B, et al. Nivolumab in previously untreated melanoma without BRAF mutation. N Engl J Med. 2015;372:320-330.
15. Robert C, Karaszewska B, Schachter J, et al. Improved overall survival in melanoma with combined dabrafenib and trametinib. N Engl J Med. 2015;372:30-39.
16. Espinosa E, Berrocal A, López Martin JA, et al. Advances in cutaneous melanoma. Clin Transl Oncol. 2012;14:325-332.
17. Chapman PB. Treating metastatic melanoma in 2014: what just happened and what is next? Am Soc Clin Oncol Educ Book. 2014:16-19.
18. Eggermont A, Chiarion-Sileni V, Grob JJ, et al. Adjuvant ipilimumab versus placebo after complete resection of high-risk stage III melanoma (EORTC 18071): a randomised, double-blind, phase 3 trial. Lancet Oncol. 2015;16:522-530.
19. Curry JL, Tetzlaff MT, Nicholson K, et al. Histological features associated with vemurafenib-induced skin toxicities: examination of 141 cutaneous lesions biopsied during therapy. Am J Dermatopathol. 2014; 36:557-561.
20. Perier-Muzet M, Thomas L, Poulalhon N, et al. Melanoma patients under vemurafenib: prospective follow-up of melanocytic lesions by digital dermoscopy. J Invest Dermatol. 2014;134:1351-1358.
21. Ross MI, Andtbacka RI, Puzanov I, et al. Patterns of durable response with intralesional talimogene laherparepvec (T-VEC): results from a phase III trial in patients with stage IIIb-IV melanoma. Paper presented at: ASCO Annual Meeting; June 2, 2014; Boston, MA.
22. Postow MA, Chesney J, Pavlick AC, et al. Novolumab and ipilimumab versus ipilimumab in untreated melanoma. N Engl J Med. 2015;372:2006-2017.
23. Gorantla VC, Kirkwood JM. State of melanoma: an historic overview of a field in transition. Hematol Oncol Clin North Am. 2014;28:415-435.
24. Coit DG, Olszanski AJ. Progress in the management of melanoma in 2013. J Natl Compr Canc Netw. 2013; 11(5 suppl):645-648.
25. Watson M, Johnson CJ, Chen VW, et al. Melanoma surveillance in the United States: overview of methods. J Am Acad Dermatol. 2011;65(5, suppl 1):S6-S16.
Practice Points
- The landscape of melanoma clinical trial research has shifted to follow advances in targeted therapy and immunotherapy.
- With these new treatments there is an increased risk for nonmelanoma skin toxicities requiring increased vigilance and collaboration between dermatologists and oncologists.
- Physicians are encouraged to use ClinicalTrials.gov to find details and contact information for actively recruiting clinical trials and results on completed trials.
Physician Skin Examinations for Melanoma Screening
In the United States an estimated 73,870 new cases of melanoma will be diagnosed in 2015.1 Although melanoma accounts for less than 2% of all US skin cancer cases, it is responsible for the vast majority of skin cancer deaths. From 2007 to 2011, melanoma mortality rates decreased by 2.6% per year in individuals younger than 50 years but increased by 0.6% per year among those 50 years and older.1 Reports of the direct annual treatment costs for melanoma in the United States have ranged from $44.9 million for Medicare recipients with existing cases of melanoma to $932.5 million for newly diagnosed melanomas across all age groups.2
Melanoma survival rates are inversely related to tumor thickness at the time of diagnosis.3 Melanoma can be cured if caught early and properly treated. Secondary preventative measures include physician skin examinations (PSEs), which may increase the likelihood of detecting melanomas in earlier stages, thereby potentially increasing survival rates and quality of life as well as decreasing treatment costs. Physician skin examinations are performed in the physician’s office and are safe, noninvasive, and painless. Patients with suspicious lesions should subsequently undergo a skin biopsy, which is a low-risk procedure. False-positives from biopsies do not lead to extreme patient morbidity, and false-negatives will hopefully be detected at a subsequent visit.
There is a lack of consensus regarding recommendations for PSEs for skin cancer screening. Due to a lack of randomized controlled trials on the effects of skin cancer screening on patient morbidity and mortality, the US Preventive Services Task Force (USPSTF) has concluded that there is insufficient evidence to recommend for or against such screening4; however, other organizations including the American Cancer Society and the American Academy of Dermatology recommend periodic skin cancer screening examinations.1,5 In a rapidly changing health care climate and with the rollout of the Patient Protection and Affordable Care Act, a USPSTF recommendation for skin screening with PSEs for skin cancer would have a large impact on clinical practice in the United States.
This article provides a systematic review of the current domestic and international data regarding the impact of PSEs on melanoma tumor thickness at the time of diagnosis as well as mortality from melanoma.
Methods
Search Strategy
A systematic search of PubMed articles indexed for MEDLINE and Embase for studies related to melanoma and PSEs was performed for the period from each database’s inception to November 8, 2014. One of the authors (S.L.M.) designed a broad search strategy with assistance from a medical librarian who had expertise in searching research bibliographies. Articles were excluded if they had a cross-sectional study design or were editorials or review articles. Search terms included skin neoplasm, skin cancer, or melanoma in combination with any of the following: skin examination, mass screening, screening, and secondary prevention.
Study Selection
All published studies reporting outcomes and correlations with PSEs and cutaneous melanoma in adult patients were screened. If multiple studies were published describing the same study, follow-up studies were included for data extraction, but the original study was the primary resource. Observational studies were a focus in this review, as these types of studies are much more common in this subject area.
One of the authors (S.L.M.) screened the titles and abstracts of identified studies for eligibility. If the reviewer considered a study potentially eligible based on the abstract review, a full-text review was carried out. The reference lists of eligible studies were manually searched to identify additional studies.
Data Extraction, Quality Assessment, and Data Synthesis
Data items to be extracted were agreed on before search implementation and were extracted by one investigator (S.L.M.) following criteria developed by review of the Cochrane Handbook for Systematic Reviews of Interventions.6 Study population, design, sample size, and outcomes were extracted. Risk of bias of individual articles was evaluated using a tool developed from the RTI item bank (RTI International) for determining the risk of bias and precision of eligible observational studies.7 Studies ultimately were classified into 3 categories based on the risk of bias: (1) low risk of bias, (2) medium risk of bias, and (3) high risk of bias. The strength of evidence of included studies was evaluated by the following items: risk of bias, consistency, directness, precision, and overall conclusion. Data from the included studies was synthesized qualitatively in a narrative format. This review adhered to guidelines in the Cochrane Handbook for Systematic Reviews of Interventions6 and the PRISMA (preferred reporting items for systematic reviews and meta-analyses) guidelines.8
Results
A total of 705 titles were screened, 98 abstracts were assessed for eligibility, 42 full-text reviews were carried out, and 5 eligible studies were identified (Figure 1). Five observational studies were included in the final review. A summary of the results is presented in Table 1.
Included studies were assessed for several types of biases, including selection bias, attrition bias, detection bias, performance bias, and response bias. The judgments were given for each domain (Table 2). There was heterogeneity in study design, reporting of total-body skin examination methods, and reporting of outcomes among all 5 studies. All 5 studies were assessed as having a medium risk of bias.
Physician Skin Examination Impact
One article by Berwick et al9 reanalyzed data from a 1996 study10 and provided no significant evidence regarding the benefits of PSEs in the reduction of melanoma mortality. Data for 650 patients with newly diagnosed melanomas were obtained from the Connecticut Tumor Registry, a site for the National Cancer Institute’s Surveillance, Epidemiology, and End Results (SEER) program, along with 549 age- and sex-frequency matched controls from the general population.10 Participants were followed biannually for a mean of 5.4 years. Of the original 650 case patients, 122 were excluded from the study with reasons provided. Physician skin examination was defined as a positive response to the following questionnaire item: “[Before your recent biopsy] did the doctor examine your skin during any of your visits?”9 Data analysis showed no significant association between PSE and death from melanoma. Upon univariate analysis, the hazard ratio for physician screening was 0.7 (95% confidence interval [CI], 0.4-1.3).9
The SCREEN (Skin Cancer Research to Provide Evidence for Effectiveness of Screening in Northern Germany) project, which was undertaken in Schleswig-Holstein, Germany, is the world’s largest systematic population-based skin cancer screening program.15 The participation rate was
19% (N=360,288) of the eligible population (citizens aged ≥20 years with statutory health insurance). Screening was a 2-step process performed by trained physicians: initial general practitioner whole-body skin examination followed by referral to a dermatologist for evaluation of suspicious skin findings. Five years after the SCREEN program was conducted, melanoma mortality declined by 47% per 100,000 men and by 49% per 100,000 women. The annual percentage change in the most recent 10-year period (2000-2009) was 7.5% (95% CI, –14.0 to –0.5; P<.05) for men and 7.1% for women (95% CI,
–10.5 to –2.9; P<.05). Simultaneously, the melanoma mortality rates in the 4 unscreened adjacent regions and the rest of Germany were stable, significantly (P<.05) different from the decline in mortality observed in Schleswig-Holstein.15
A community-based, prospective cohort study investigated the impact of an employee melanoma screening program at the Lawrence Livermore National Laboratory (Livermore, California) (1984-1996) demonstrated an impact on melanoma thickness and mortality rates.12 The cohort (approximately 5100 participants) was followed over 3 phases of surveillance: (1) preawareness (1969-1975), (2) early awareness of increased melanoma risk (1976-1984), and (3) screening program (1984-1996). The screening program encouraged employees to self-examine their skin for “suggestive lesions”; if a suggestive lesion was found, a full-body skin examination was performed by a physician. After being evaluated, participants with melanoma, dysplastic nevi, 50 or more moles, or a family history of melanoma were offered a periodic full-body examination every 3 to 24 months, often with
full-body photography and dermoscopy. Physician skin screening resulted in a reduction in crude incidence of thicker melanomas (defined as
>0.75 mm) during the 3 study phases. Compared with the early-awareness period (phase 2), a 69% reduction in the diagnosis of thick melanomas was reported in the screening program period (phase 3)(P=.0001). During the screening period, no eligible melanoma deaths occurred in the study population, whereas the expected number of deaths was 3.39 (P=.034) based on observed melanoma mortality in 5 San Francisco/Oakland Bay–area counties in California as reported to the SEER program from 1984 to 1996.12
The strongest evidence for reduced thickness of melanomas detected via PSEs was reported in a population-based, case-control study by Aitken et al14 of all residents in Queensland, Australia, aged 20 to 75 years with a histologically confirmed first primary invasive cutaneous melanoma diagnosed between January 2000 and December 2003. Whole-body PSE in the 3 years before diagnosis was inversely associated with tumor thickness at diagnosis (χ2=44.37; P<.001), including a 14% lower risk of diagnosis of a thick melanoma (>0.75 mm)(odds ratio [OR], 0.86; 95% CI, 0.75-0.98) and a 40% lower risk of diagnosis of a melanoma that was 3 mm or larger (OR, 0.60; 95% CI, 0.43-0.83). The investigators applied melanoma thickness-specific survival estimates to the thickness distribution of the screened and unscreened cases in their sample to estimate melanoma deaths within 5 and 10 years of diagnosis. Compared to the unscreened cases, they estimated that the screened cases would have 26% fewer melanoma deaths within 5 years of diagnosis and 23% fewer deaths within 10 years.14
Another prospective cohort study in Queensland was designed to detect a 20% reduction in mortality from melanoma during a 15-year intervention period in communities that received a screening program.11 A total of 44 communities (aggregate population, 560,000 adults aged ≥30 years) were randomized into intervention or control groups to receive a community-based melanoma screening program for 3 years versus usual medical care.Overall, thinner melanomas were identified in communities with the screening program versus neighboring communities without it.11 Of the 33 melanomas found through the screening program, 39% (13/33) were in situ lesions, 55% (18/33) were thin (<1 mm) invasive lesions, and 6% (2/33) were 1-mm thick or greater.16 Within the population of Queensland during the period from 1999 through 2002, 36% were in situ lesions, 48% were invasive thin melanomas, and 16% were invasive melanomas 1-mm thick or more, indicating that melanomas found through screening were thinner or less advanced.17
Comment
Our review identified 5 studies describing the impact of PSEs for melanoma screening on tumor thickness at diagnosis and melanoma mortality. Key findings are highlighted in Figure 2. Our findings suggest that PSEs are associated with a decline in melanoma tumor thickness and melanoma-specific mortality. Our findings are qualitatively similar to prior reviews that supported the use of PSEs to detect thinner melanomas and improve mortality outcomes.18-20
The greatest evidence for population-based screening programs was provided by the SCREEN study. This landmark study documented that screening programs utilizing primary care physicians (PCPs) and dermatologists can lead to a reduction in melanoma mortality.15 Findings from the study led to the countrywide expansion of the screening program in 2008, leading to 45 million Germans eligible for skin cancer screenings every 2 years.21 Nearly two-thirds of dermatologists (N=1348) were satisfied with routine PSE and 83% perceived a better quality of health care for skin with the 2008 expansion.22
Data suggest that physician-detected melanomas through PSEs or routine physical examinations are thinner at the time of diagnosis than those found by patients or their partners.14,23-26 Terushkin and Halpern20 analyzed 9 worldwide studies encompassing more than 7500 patients and found that physician-detected melanomas were 0.55 mm thinner than those detected by patients or their significant others. The workplace screening and education program reviewed herein also reported a reduction in thicker melanomas and melanoma mortality during the study period.12
Not all Americans have a regular dermatologist. As such, educating PCPs in skin cancer detection has been a recent area of study. The premise is that the skin examination can be integrated into routine physical examinations conducted by PCPs. The previously discussed studies, particularly Aitken et al,14 Schneider et al,12 and Katalinic et al,15 as well as the SCREEN program studies,15 suggest that integration of the skin examination into the routine physical examination may be a feasible method to reduce melanoma thickness and mortality. Furthermore, the SCREEN study15 identified participants with risk factors for melanoma, finding that approximately half of men and women (N=360,288) had at least one melanoma risk factor, which suggests that it may be more practical to design screening practices around high-risk participants.
Several studies were excluded from our analysis on the basis of study design, including cross-sectional observational studies; however, it is worth briefly commenting on the findings of the excluded studies here, as they add to the body of literature. A community-based, multi-institutional study of 566 adults with invasive melanoma assessed the role of PSEs in the year prior to diagnosis by interviewing participants in clinic within 3 months of melanoma diagnosis.24 Patients who underwent full-body PSE in the year prior to diagnosis were more than 2 times more likely to have thinner (≤1 mm) melanomas (OR, 2.51; 95% CI, 1.62-3.87]). Notably, men older than 60 years appeared to benefit the most from this practice; men in this age group contributed greatly to the observed effect, likely because they had 4 times the odds of a thinner melanoma (OR, 4.09; 95% CI, 1.88-8.89]). Thinner melanomas also were associated with an age of 60 years or younger, female sex, and higher education level.24
Pollitt et al27 analyzed the association between prediagnosis Medicaid enrollment status and melanoma tumor thickness. The study found that men and women who intermittently enrolled in Medicaid or were not enrolled until the month of diagnosis had an increased chance of late-stage melanoma when compared to other patients. Patients who continuously enrolled during the year prior to diagnosis had lower odds for thicker melanomas, suggesting that these patients had greater access to screening examinations.27
Roetzheim et al28 analyzed data from the SEER-Medicare linked dataset to investigate patterns of dermatologist and PCP visits in the 2 years before melanoma diagnosis. Medicare beneficiaries seeing both a dermatologist and a PCP prior to melanoma diagnosis had greater odds of a thinner melanoma and lower melanoma mortality compared to patients without such visits.28
Durbec et al29 conducted a retrospective, population-based study of 650 patients in France who were seen by a dermatologist for melanoma. The thinnest melanomas were reported in patients seeing a dermatologist for prospective follow-up of nevi or consulting a dermatologist for other diseases. Patients referred to a dermatologist by PCPs tended to be older and had the highest frequency of thick (>3 mm), nodular, and/or ulcerated melanomas,29 which could be interpreted as a need for greater PCP education in melanoma screening.
Rates of skin examinations have been increasing since the year 2000, both overall and among high-risk groups as reported by a recent study on skin cancer screening trends. Prevalence of having at least one total-body skin examination increased from 14.5% in 2000 to 16.5% in 2005 to 19.8% in 2010 (P<.0001).30 One study revealed a practice gap in which more than 3 in 10 PCPs and 1 in 10 dermatologists reported not screening more than half their high-risk patients for skin cancer.31 The major obstacle to narrowing the identified practice gap involves establishing a national strategy to screen high-risk individuals for skin cancer and requires partnerships among patients, PCPs, specialists, policy makers, and government sponsors.
Lack of evidence that screening for skin cancer with PSEs reduces overall mortality does not mean there is a lack of lifesaving potential of screenings. The resources required to execute a randomized controlled trial with adequate power are vast, as the USPSTF estimated 800,000 participants would be needed.4 Barriers to conducting a randomized clinical trial for skin cancer screening include the large sample size required, prolonged follow-up, and various ethical issues such as withholding screening for a cancer that is potentially curable in early stages. Lessons from screenings for breast and prostate cancers have taught us that such randomized controlled trials assessing cancer screening are costly and do not always produce definitive answers.32
Conclusion
Although proof of improved health outcomes from randomized controlled trials is still required, there is evidence to support targeted screening programs for the detection of thinner melanomas and, by proxy, reduced melanoma mortality. Amidst the health care climate change and payment reform, recommendations from national organizations on melanoma screenings are paramount. Clinicians should continue to offer regular skin examinations as the body of evidence continues to grow in support of PSEs for melanoma screening.
Acknowledgments—We are grateful to Mary Butler, PhD, and Robert Kane, MD, both from Minneapolis, Minnesota, for their guidance and consultation.
1. American Cancer Society. Cancer Facts & Figures 2015. Atlanta, GA: American Cancer Society; 2015. http: //www.cancer.org/Research/CancerFactsStatistics/cancer factsfigures2015/cancer-facts-and-figures-2015. Accessed July 6, 2015.
2. Guy G Jr, Ekwueme D, Tangka F, et al. Melanoma treatment costs: a systematic review of the literature, 1990-2011. Am J Prev. 2012;43:537-545.
3. Margolis D, Halpern A, Rebbeck T, et al. Validation of a melanoma prognostic model. Arch Dermatol. 1998;134:1597-1601.
4. Wolff T, Tai E, Miller T. Screening for skin cancer: an update of the evidence for the U.S. Preventative Services Task Force. Ann Intern Med. 2009;150:194-198.
5. American Academy of Dermatology. Melanoma
Monday. http://www.aad.org/spot-skin-cancer
/community-programs-events/melanoma-monday. Accessed August 19, 2015.
6. Higgins JPT, Green S, eds. Cochrane Handbook for
Systematic Reviews of Interventions Version 5.1.0 [updated March 2011]. The Cochrane Collaboration, 2011. http:
//www.cochrane-handbook.org. Updated March 2011. Accessed November 10, 2014.
7. Viswanathan M, Berkman N. Development of the RTI item bank on risk of bias and precision of observational studies. J Clin Epidemiol. 2012;65:163-178.
8. Moher D, Liberati A, Tetzlaff J, et al; PRISMA group. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement [published online ahead of print July 23, 2009]. J Clin Epidemiol. 2009;62:1006-1012.
9. Berwick M, Armstrong B, Ben-Porat L. Sun exposure and mortality from melanoma. J Natl Cancer Inst. 2005;97:195-199.
10. Berwick M, Begg C, Fine J, et al. Screening for cutaneous melanoma by skin self-examination. J Natl Cancer Inst. 1996;88:17-23.
11. Aitken J, Elwood J, Lowe J, et al. A randomised trial of population screening for melanoma. J Med Screen. 2002;9:33-37.
12. Schneider J, Moore D, Mendelsohn M. Screening program reduced melanoma mortality at the Lawrence Livermore National Laboratory, 1984 to 1996. J Am Acad Dermatol. 2008;58:741-749.
13. Expert Health Data Programming Inc. Health data software and health statistics. Available from: http: //www.ehdp.com. Accessed April 1, 2001. Cited by: Schneider J, Moore D, Mendelsohn M. Screening program reduced melanoma mortality at the Lawrence Livermore National Laboratory, 1984 to 1996. J Am Acad Dermatol. 2008;58:741-749.
14. Aitken J, Elwood M, Baade P, et al. Clinical whole-body skin examination reduces the incidence of thick melanomas. Int J Cancer. 2010;126:450-458.
15. Katalinic A, Waldmann A, Weinstock M, et al. Does skin cancer screening save lives? an observational study comparing trends in melanoma mortality in regions with and without screening. Cancer. 2012;118:5395-5402.
16. Aitken J, Janda M, Elwood M, et al. Clinical outcomes from skin screening clinics within a community-based melanoma screening program. J Am Acad Dermatol. 2006;54:105-114.
17. Coory M, Baade P, Aitken JF, et al. Trends for in-situ and invasive melanoma in Queensland, Australia, 1982 to 2002. Cancer Causes Control. 2006;17:21-27.
18. Mayer JE, Swetter SM, Fu T, et al. Screening, early detection, education, and trends for melanoma: current status (2007-2013) and future directions: part II. screening, education, and future directions. J Am Acad Dermatol. 2014;71:611.e1-611.e10; quiz, 621-622.
19. Curiel-Lewandrowski C, Chen S, Swetter S, et al. Screening and prevention measures for melanoma: is there a survival advantage? Curr Oncol Rep. 2012;14:458-467.
20. Terushkin V, Halpern A. Melanoma early detection. Hematol Oncol Clin North Am. 2009;23:481-500.
21. Geller A, Greinert R, Sinclair C, et al. A nationwide population-based skin cancer screening in Germany: proceedings of the first meeting of the International Task Force on Skin Cancer Screening and Prevention (September 24 and 25, 2009) [published online ahead of print April 8, 2010]. Cancer Epidemiol. 2010;34:355-358.
22. Kornek T, Schafer I, Reusch M, et al. Routine skin cancer screening in Germany: four years of experience from the dermatologists’ perspective. Dermatology. 2012;225:289-293.
23. De Giorgi V, Grazzini M, Rossari S, et al. Is skin self-examination for cutaneous melanoma detection still adequate? a retrospective study. Dermatology. 2012;225:31-36.
24. Swetter S, Johnson T, Miller D, et al. Melanoma in middle-aged and older men: a multi-institutional survey study of factors related to tumor thickness. Arch Dermatol. 2009;145:397-404.
25. Kantor J, Kantor D. Routine dermatologist-performed full-body skin examination and early melanoma detection. Arch Dermatol. 2009;145:873-876.
26. Kovalyshyn I, Dusza S, Siamas K, et al. The impact of physician screening on melanoma detection. Arch Dermatol. 2011;147:1269-1275.
27. Pollitt R, Clarke C, Shema S, et al. California Medicaid enrollment and melanoma stage at diagnosis: a population-based study. Am J Prev Med. 2008;35:7-13.
28. Roetzheim R, Lee J, Ferrante J, et al. The influence of dermatologist and primary care physician visits on melanoma outcomes among Medicare beneficiaries. J Am Board Fam Med. 2013;26:637-647.
29. Durbec F, Vitry F, Granel-Brocard F, et al. The role of circumstances of diagnosis and access to dermatological care in early diagnosis of cutaneous melanoma: a population-based study in France. Arch Dermatol. 2010;146:240-246.
30. Lakhani N, Saraiya M, Thompson T, et al. Total body skin examination for skin cancer screening among U.S. adults from 2000 to 2010. Prev Med. 2014;61:75-80.
31. Oliveria SA, Heneghan MK, Cushman LF, et al. Skin cancer screening by dermatologists, family practitioners, and internists: barriers and facilitating factors. Arch Dermatol. 2011;147:39-44.
32. Bigby M. Why the evidence for skin cancer screening is insufficient: lessons from prostate cancer screening. Arch Dermatol. 2010;146:322-324.
In the United States an estimated 73,870 new cases of melanoma will be diagnosed in 2015.1 Although melanoma accounts for less than 2% of all US skin cancer cases, it is responsible for the vast majority of skin cancer deaths. From 2007 to 2011, melanoma mortality rates decreased by 2.6% per year in individuals younger than 50 years but increased by 0.6% per year among those 50 years and older.1 Reports of the direct annual treatment costs for melanoma in the United States have ranged from $44.9 million for Medicare recipients with existing cases of melanoma to $932.5 million for newly diagnosed melanomas across all age groups.2
Melanoma survival rates are inversely related to tumor thickness at the time of diagnosis.3 Melanoma can be cured if caught early and properly treated. Secondary preventative measures include physician skin examinations (PSEs), which may increase the likelihood of detecting melanomas in earlier stages, thereby potentially increasing survival rates and quality of life as well as decreasing treatment costs. Physician skin examinations are performed in the physician’s office and are safe, noninvasive, and painless. Patients with suspicious lesions should subsequently undergo a skin biopsy, which is a low-risk procedure. False-positives from biopsies do not lead to extreme patient morbidity, and false-negatives will hopefully be detected at a subsequent visit.
There is a lack of consensus regarding recommendations for PSEs for skin cancer screening. Due to a lack of randomized controlled trials on the effects of skin cancer screening on patient morbidity and mortality, the US Preventive Services Task Force (USPSTF) has concluded that there is insufficient evidence to recommend for or against such screening4; however, other organizations including the American Cancer Society and the American Academy of Dermatology recommend periodic skin cancer screening examinations.1,5 In a rapidly changing health care climate and with the rollout of the Patient Protection and Affordable Care Act, a USPSTF recommendation for skin screening with PSEs for skin cancer would have a large impact on clinical practice in the United States.
This article provides a systematic review of the current domestic and international data regarding the impact of PSEs on melanoma tumor thickness at the time of diagnosis as well as mortality from melanoma.
Methods
Search Strategy
A systematic search of PubMed articles indexed for MEDLINE and Embase for studies related to melanoma and PSEs was performed for the period from each database’s inception to November 8, 2014. One of the authors (S.L.M.) designed a broad search strategy with assistance from a medical librarian who had expertise in searching research bibliographies. Articles were excluded if they had a cross-sectional study design or were editorials or review articles. Search terms included skin neoplasm, skin cancer, or melanoma in combination with any of the following: skin examination, mass screening, screening, and secondary prevention.
Study Selection
All published studies reporting outcomes and correlations with PSEs and cutaneous melanoma in adult patients were screened. If multiple studies were published describing the same study, follow-up studies were included for data extraction, but the original study was the primary resource. Observational studies were a focus in this review, as these types of studies are much more common in this subject area.
One of the authors (S.L.M.) screened the titles and abstracts of identified studies for eligibility. If the reviewer considered a study potentially eligible based on the abstract review, a full-text review was carried out. The reference lists of eligible studies were manually searched to identify additional studies.
Data Extraction, Quality Assessment, and Data Synthesis
Data items to be extracted were agreed on before search implementation and were extracted by one investigator (S.L.M.) following criteria developed by review of the Cochrane Handbook for Systematic Reviews of Interventions.6 Study population, design, sample size, and outcomes were extracted. Risk of bias of individual articles was evaluated using a tool developed from the RTI item bank (RTI International) for determining the risk of bias and precision of eligible observational studies.7 Studies ultimately were classified into 3 categories based on the risk of bias: (1) low risk of bias, (2) medium risk of bias, and (3) high risk of bias. The strength of evidence of included studies was evaluated by the following items: risk of bias, consistency, directness, precision, and overall conclusion. Data from the included studies was synthesized qualitatively in a narrative format. This review adhered to guidelines in the Cochrane Handbook for Systematic Reviews of Interventions6 and the PRISMA (preferred reporting items for systematic reviews and meta-analyses) guidelines.8
Results
A total of 705 titles were screened, 98 abstracts were assessed for eligibility, 42 full-text reviews were carried out, and 5 eligible studies were identified (Figure 1). Five observational studies were included in the final review. A summary of the results is presented in Table 1.
Included studies were assessed for several types of biases, including selection bias, attrition bias, detection bias, performance bias, and response bias. The judgments were given for each domain (Table 2). There was heterogeneity in study design, reporting of total-body skin examination methods, and reporting of outcomes among all 5 studies. All 5 studies were assessed as having a medium risk of bias.
Physician Skin Examination Impact
One article by Berwick et al9 reanalyzed data from a 1996 study10 and provided no significant evidence regarding the benefits of PSEs in the reduction of melanoma mortality. Data for 650 patients with newly diagnosed melanomas were obtained from the Connecticut Tumor Registry, a site for the National Cancer Institute’s Surveillance, Epidemiology, and End Results (SEER) program, along with 549 age- and sex-frequency matched controls from the general population.10 Participants were followed biannually for a mean of 5.4 years. Of the original 650 case patients, 122 were excluded from the study with reasons provided. Physician skin examination was defined as a positive response to the following questionnaire item: “[Before your recent biopsy] did the doctor examine your skin during any of your visits?”9 Data analysis showed no significant association between PSE and death from melanoma. Upon univariate analysis, the hazard ratio for physician screening was 0.7 (95% confidence interval [CI], 0.4-1.3).9
The SCREEN (Skin Cancer Research to Provide Evidence for Effectiveness of Screening in Northern Germany) project, which was undertaken in Schleswig-Holstein, Germany, is the world’s largest systematic population-based skin cancer screening program.15 The participation rate was
19% (N=360,288) of the eligible population (citizens aged ≥20 years with statutory health insurance). Screening was a 2-step process performed by trained physicians: initial general practitioner whole-body skin examination followed by referral to a dermatologist for evaluation of suspicious skin findings. Five years after the SCREEN program was conducted, melanoma mortality declined by 47% per 100,000 men and by 49% per 100,000 women. The annual percentage change in the most recent 10-year period (2000-2009) was 7.5% (95% CI, –14.0 to –0.5; P<.05) for men and 7.1% for women (95% CI,
–10.5 to –2.9; P<.05). Simultaneously, the melanoma mortality rates in the 4 unscreened adjacent regions and the rest of Germany were stable, significantly (P<.05) different from the decline in mortality observed in Schleswig-Holstein.15
A community-based, prospective cohort study investigated the impact of an employee melanoma screening program at the Lawrence Livermore National Laboratory (Livermore, California) (1984-1996) demonstrated an impact on melanoma thickness and mortality rates.12 The cohort (approximately 5100 participants) was followed over 3 phases of surveillance: (1) preawareness (1969-1975), (2) early awareness of increased melanoma risk (1976-1984), and (3) screening program (1984-1996). The screening program encouraged employees to self-examine their skin for “suggestive lesions”; if a suggestive lesion was found, a full-body skin examination was performed by a physician. After being evaluated, participants with melanoma, dysplastic nevi, 50 or more moles, or a family history of melanoma were offered a periodic full-body examination every 3 to 24 months, often with
full-body photography and dermoscopy. Physician skin screening resulted in a reduction in crude incidence of thicker melanomas (defined as
>0.75 mm) during the 3 study phases. Compared with the early-awareness period (phase 2), a 69% reduction in the diagnosis of thick melanomas was reported in the screening program period (phase 3)(P=.0001). During the screening period, no eligible melanoma deaths occurred in the study population, whereas the expected number of deaths was 3.39 (P=.034) based on observed melanoma mortality in 5 San Francisco/Oakland Bay–area counties in California as reported to the SEER program from 1984 to 1996.12
The strongest evidence for reduced thickness of melanomas detected via PSEs was reported in a population-based, case-control study by Aitken et al14 of all residents in Queensland, Australia, aged 20 to 75 years with a histologically confirmed first primary invasive cutaneous melanoma diagnosed between January 2000 and December 2003. Whole-body PSE in the 3 years before diagnosis was inversely associated with tumor thickness at diagnosis (χ2=44.37; P<.001), including a 14% lower risk of diagnosis of a thick melanoma (>0.75 mm)(odds ratio [OR], 0.86; 95% CI, 0.75-0.98) and a 40% lower risk of diagnosis of a melanoma that was 3 mm or larger (OR, 0.60; 95% CI, 0.43-0.83). The investigators applied melanoma thickness-specific survival estimates to the thickness distribution of the screened and unscreened cases in their sample to estimate melanoma deaths within 5 and 10 years of diagnosis. Compared to the unscreened cases, they estimated that the screened cases would have 26% fewer melanoma deaths within 5 years of diagnosis and 23% fewer deaths within 10 years.14
Another prospective cohort study in Queensland was designed to detect a 20% reduction in mortality from melanoma during a 15-year intervention period in communities that received a screening program.11 A total of 44 communities (aggregate population, 560,000 adults aged ≥30 years) were randomized into intervention or control groups to receive a community-based melanoma screening program for 3 years versus usual medical care.Overall, thinner melanomas were identified in communities with the screening program versus neighboring communities without it.11 Of the 33 melanomas found through the screening program, 39% (13/33) were in situ lesions, 55% (18/33) were thin (<1 mm) invasive lesions, and 6% (2/33) were 1-mm thick or greater.16 Within the population of Queensland during the period from 1999 through 2002, 36% were in situ lesions, 48% were invasive thin melanomas, and 16% were invasive melanomas 1-mm thick or more, indicating that melanomas found through screening were thinner or less advanced.17
Comment
Our review identified 5 studies describing the impact of PSEs for melanoma screening on tumor thickness at diagnosis and melanoma mortality. Key findings are highlighted in Figure 2. Our findings suggest that PSEs are associated with a decline in melanoma tumor thickness and melanoma-specific mortality. Our findings are qualitatively similar to prior reviews that supported the use of PSEs to detect thinner melanomas and improve mortality outcomes.18-20
The greatest evidence for population-based screening programs was provided by the SCREEN study. This landmark study documented that screening programs utilizing primary care physicians (PCPs) and dermatologists can lead to a reduction in melanoma mortality.15 Findings from the study led to the countrywide expansion of the screening program in 2008, leading to 45 million Germans eligible for skin cancer screenings every 2 years.21 Nearly two-thirds of dermatologists (N=1348) were satisfied with routine PSE and 83% perceived a better quality of health care for skin with the 2008 expansion.22
Data suggest that physician-detected melanomas through PSEs or routine physical examinations are thinner at the time of diagnosis than those found by patients or their partners.14,23-26 Terushkin and Halpern20 analyzed 9 worldwide studies encompassing more than 7500 patients and found that physician-detected melanomas were 0.55 mm thinner than those detected by patients or their significant others. The workplace screening and education program reviewed herein also reported a reduction in thicker melanomas and melanoma mortality during the study period.12
Not all Americans have a regular dermatologist. As such, educating PCPs in skin cancer detection has been a recent area of study. The premise is that the skin examination can be integrated into routine physical examinations conducted by PCPs. The previously discussed studies, particularly Aitken et al,14 Schneider et al,12 and Katalinic et al,15 as well as the SCREEN program studies,15 suggest that integration of the skin examination into the routine physical examination may be a feasible method to reduce melanoma thickness and mortality. Furthermore, the SCREEN study15 identified participants with risk factors for melanoma, finding that approximately half of men and women (N=360,288) had at least one melanoma risk factor, which suggests that it may be more practical to design screening practices around high-risk participants.
Several studies were excluded from our analysis on the basis of study design, including cross-sectional observational studies; however, it is worth briefly commenting on the findings of the excluded studies here, as they add to the body of literature. A community-based, multi-institutional study of 566 adults with invasive melanoma assessed the role of PSEs in the year prior to diagnosis by interviewing participants in clinic within 3 months of melanoma diagnosis.24 Patients who underwent full-body PSE in the year prior to diagnosis were more than 2 times more likely to have thinner (≤1 mm) melanomas (OR, 2.51; 95% CI, 1.62-3.87]). Notably, men older than 60 years appeared to benefit the most from this practice; men in this age group contributed greatly to the observed effect, likely because they had 4 times the odds of a thinner melanoma (OR, 4.09; 95% CI, 1.88-8.89]). Thinner melanomas also were associated with an age of 60 years or younger, female sex, and higher education level.24
Pollitt et al27 analyzed the association between prediagnosis Medicaid enrollment status and melanoma tumor thickness. The study found that men and women who intermittently enrolled in Medicaid or were not enrolled until the month of diagnosis had an increased chance of late-stage melanoma when compared to other patients. Patients who continuously enrolled during the year prior to diagnosis had lower odds for thicker melanomas, suggesting that these patients had greater access to screening examinations.27
Roetzheim et al28 analyzed data from the SEER-Medicare linked dataset to investigate patterns of dermatologist and PCP visits in the 2 years before melanoma diagnosis. Medicare beneficiaries seeing both a dermatologist and a PCP prior to melanoma diagnosis had greater odds of a thinner melanoma and lower melanoma mortality compared to patients without such visits.28
Durbec et al29 conducted a retrospective, population-based study of 650 patients in France who were seen by a dermatologist for melanoma. The thinnest melanomas were reported in patients seeing a dermatologist for prospective follow-up of nevi or consulting a dermatologist for other diseases. Patients referred to a dermatologist by PCPs tended to be older and had the highest frequency of thick (>3 mm), nodular, and/or ulcerated melanomas,29 which could be interpreted as a need for greater PCP education in melanoma screening.
Rates of skin examinations have been increasing since the year 2000, both overall and among high-risk groups as reported by a recent study on skin cancer screening trends. Prevalence of having at least one total-body skin examination increased from 14.5% in 2000 to 16.5% in 2005 to 19.8% in 2010 (P<.0001).30 One study revealed a practice gap in which more than 3 in 10 PCPs and 1 in 10 dermatologists reported not screening more than half their high-risk patients for skin cancer.31 The major obstacle to narrowing the identified practice gap involves establishing a national strategy to screen high-risk individuals for skin cancer and requires partnerships among patients, PCPs, specialists, policy makers, and government sponsors.
Lack of evidence that screening for skin cancer with PSEs reduces overall mortality does not mean there is a lack of lifesaving potential of screenings. The resources required to execute a randomized controlled trial with adequate power are vast, as the USPSTF estimated 800,000 participants would be needed.4 Barriers to conducting a randomized clinical trial for skin cancer screening include the large sample size required, prolonged follow-up, and various ethical issues such as withholding screening for a cancer that is potentially curable in early stages. Lessons from screenings for breast and prostate cancers have taught us that such randomized controlled trials assessing cancer screening are costly and do not always produce definitive answers.32
Conclusion
Although proof of improved health outcomes from randomized controlled trials is still required, there is evidence to support targeted screening programs for the detection of thinner melanomas and, by proxy, reduced melanoma mortality. Amidst the health care climate change and payment reform, recommendations from national organizations on melanoma screenings are paramount. Clinicians should continue to offer regular skin examinations as the body of evidence continues to grow in support of PSEs for melanoma screening.
Acknowledgments—We are grateful to Mary Butler, PhD, and Robert Kane, MD, both from Minneapolis, Minnesota, for their guidance and consultation.
In the United States an estimated 73,870 new cases of melanoma will be diagnosed in 2015.1 Although melanoma accounts for less than 2% of all US skin cancer cases, it is responsible for the vast majority of skin cancer deaths. From 2007 to 2011, melanoma mortality rates decreased by 2.6% per year in individuals younger than 50 years but increased by 0.6% per year among those 50 years and older.1 Reports of the direct annual treatment costs for melanoma in the United States have ranged from $44.9 million for Medicare recipients with existing cases of melanoma to $932.5 million for newly diagnosed melanomas across all age groups.2
Melanoma survival rates are inversely related to tumor thickness at the time of diagnosis.3 Melanoma can be cured if caught early and properly treated. Secondary preventative measures include physician skin examinations (PSEs), which may increase the likelihood of detecting melanomas in earlier stages, thereby potentially increasing survival rates and quality of life as well as decreasing treatment costs. Physician skin examinations are performed in the physician’s office and are safe, noninvasive, and painless. Patients with suspicious lesions should subsequently undergo a skin biopsy, which is a low-risk procedure. False-positives from biopsies do not lead to extreme patient morbidity, and false-negatives will hopefully be detected at a subsequent visit.
There is a lack of consensus regarding recommendations for PSEs for skin cancer screening. Due to a lack of randomized controlled trials on the effects of skin cancer screening on patient morbidity and mortality, the US Preventive Services Task Force (USPSTF) has concluded that there is insufficient evidence to recommend for or against such screening4; however, other organizations including the American Cancer Society and the American Academy of Dermatology recommend periodic skin cancer screening examinations.1,5 In a rapidly changing health care climate and with the rollout of the Patient Protection and Affordable Care Act, a USPSTF recommendation for skin screening with PSEs for skin cancer would have a large impact on clinical practice in the United States.
This article provides a systematic review of the current domestic and international data regarding the impact of PSEs on melanoma tumor thickness at the time of diagnosis as well as mortality from melanoma.
Methods
Search Strategy
A systematic search of PubMed articles indexed for MEDLINE and Embase for studies related to melanoma and PSEs was performed for the period from each database’s inception to November 8, 2014. One of the authors (S.L.M.) designed a broad search strategy with assistance from a medical librarian who had expertise in searching research bibliographies. Articles were excluded if they had a cross-sectional study design or were editorials or review articles. Search terms included skin neoplasm, skin cancer, or melanoma in combination with any of the following: skin examination, mass screening, screening, and secondary prevention.
Study Selection
All published studies reporting outcomes and correlations with PSEs and cutaneous melanoma in adult patients were screened. If multiple studies were published describing the same study, follow-up studies were included for data extraction, but the original study was the primary resource. Observational studies were a focus in this review, as these types of studies are much more common in this subject area.
One of the authors (S.L.M.) screened the titles and abstracts of identified studies for eligibility. If the reviewer considered a study potentially eligible based on the abstract review, a full-text review was carried out. The reference lists of eligible studies were manually searched to identify additional studies.
Data Extraction, Quality Assessment, and Data Synthesis
Data items to be extracted were agreed on before search implementation and were extracted by one investigator (S.L.M.) following criteria developed by review of the Cochrane Handbook for Systematic Reviews of Interventions.6 Study population, design, sample size, and outcomes were extracted. Risk of bias of individual articles was evaluated using a tool developed from the RTI item bank (RTI International) for determining the risk of bias and precision of eligible observational studies.7 Studies ultimately were classified into 3 categories based on the risk of bias: (1) low risk of bias, (2) medium risk of bias, and (3) high risk of bias. The strength of evidence of included studies was evaluated by the following items: risk of bias, consistency, directness, precision, and overall conclusion. Data from the included studies was synthesized qualitatively in a narrative format. This review adhered to guidelines in the Cochrane Handbook for Systematic Reviews of Interventions6 and the PRISMA (preferred reporting items for systematic reviews and meta-analyses) guidelines.8
Results
A total of 705 titles were screened, 98 abstracts were assessed for eligibility, 42 full-text reviews were carried out, and 5 eligible studies were identified (Figure 1). Five observational studies were included in the final review. A summary of the results is presented in Table 1.
Included studies were assessed for several types of biases, including selection bias, attrition bias, detection bias, performance bias, and response bias. The judgments were given for each domain (Table 2). There was heterogeneity in study design, reporting of total-body skin examination methods, and reporting of outcomes among all 5 studies. All 5 studies were assessed as having a medium risk of bias.
Physician Skin Examination Impact
One article by Berwick et al9 reanalyzed data from a 1996 study10 and provided no significant evidence regarding the benefits of PSEs in the reduction of melanoma mortality. Data for 650 patients with newly diagnosed melanomas were obtained from the Connecticut Tumor Registry, a site for the National Cancer Institute’s Surveillance, Epidemiology, and End Results (SEER) program, along with 549 age- and sex-frequency matched controls from the general population.10 Participants were followed biannually for a mean of 5.4 years. Of the original 650 case patients, 122 were excluded from the study with reasons provided. Physician skin examination was defined as a positive response to the following questionnaire item: “[Before your recent biopsy] did the doctor examine your skin during any of your visits?”9 Data analysis showed no significant association between PSE and death from melanoma. Upon univariate analysis, the hazard ratio for physician screening was 0.7 (95% confidence interval [CI], 0.4-1.3).9
The SCREEN (Skin Cancer Research to Provide Evidence for Effectiveness of Screening in Northern Germany) project, which was undertaken in Schleswig-Holstein, Germany, is the world’s largest systematic population-based skin cancer screening program.15 The participation rate was
19% (N=360,288) of the eligible population (citizens aged ≥20 years with statutory health insurance). Screening was a 2-step process performed by trained physicians: initial general practitioner whole-body skin examination followed by referral to a dermatologist for evaluation of suspicious skin findings. Five years after the SCREEN program was conducted, melanoma mortality declined by 47% per 100,000 men and by 49% per 100,000 women. The annual percentage change in the most recent 10-year period (2000-2009) was 7.5% (95% CI, –14.0 to –0.5; P<.05) for men and 7.1% for women (95% CI,
–10.5 to –2.9; P<.05). Simultaneously, the melanoma mortality rates in the 4 unscreened adjacent regions and the rest of Germany were stable, significantly (P<.05) different from the decline in mortality observed in Schleswig-Holstein.15
A community-based, prospective cohort study investigated the impact of an employee melanoma screening program at the Lawrence Livermore National Laboratory (Livermore, California) (1984-1996) demonstrated an impact on melanoma thickness and mortality rates.12 The cohort (approximately 5100 participants) was followed over 3 phases of surveillance: (1) preawareness (1969-1975), (2) early awareness of increased melanoma risk (1976-1984), and (3) screening program (1984-1996). The screening program encouraged employees to self-examine their skin for “suggestive lesions”; if a suggestive lesion was found, a full-body skin examination was performed by a physician. After being evaluated, participants with melanoma, dysplastic nevi, 50 or more moles, or a family history of melanoma were offered a periodic full-body examination every 3 to 24 months, often with
full-body photography and dermoscopy. Physician skin screening resulted in a reduction in crude incidence of thicker melanomas (defined as
>0.75 mm) during the 3 study phases. Compared with the early-awareness period (phase 2), a 69% reduction in the diagnosis of thick melanomas was reported in the screening program period (phase 3)(P=.0001). During the screening period, no eligible melanoma deaths occurred in the study population, whereas the expected number of deaths was 3.39 (P=.034) based on observed melanoma mortality in 5 San Francisco/Oakland Bay–area counties in California as reported to the SEER program from 1984 to 1996.12
The strongest evidence for reduced thickness of melanomas detected via PSEs was reported in a population-based, case-control study by Aitken et al14 of all residents in Queensland, Australia, aged 20 to 75 years with a histologically confirmed first primary invasive cutaneous melanoma diagnosed between January 2000 and December 2003. Whole-body PSE in the 3 years before diagnosis was inversely associated with tumor thickness at diagnosis (χ2=44.37; P<.001), including a 14% lower risk of diagnosis of a thick melanoma (>0.75 mm)(odds ratio [OR], 0.86; 95% CI, 0.75-0.98) and a 40% lower risk of diagnosis of a melanoma that was 3 mm or larger (OR, 0.60; 95% CI, 0.43-0.83). The investigators applied melanoma thickness-specific survival estimates to the thickness distribution of the screened and unscreened cases in their sample to estimate melanoma deaths within 5 and 10 years of diagnosis. Compared to the unscreened cases, they estimated that the screened cases would have 26% fewer melanoma deaths within 5 years of diagnosis and 23% fewer deaths within 10 years.14
Another prospective cohort study in Queensland was designed to detect a 20% reduction in mortality from melanoma during a 15-year intervention period in communities that received a screening program.11 A total of 44 communities (aggregate population, 560,000 adults aged ≥30 years) were randomized into intervention or control groups to receive a community-based melanoma screening program for 3 years versus usual medical care.Overall, thinner melanomas were identified in communities with the screening program versus neighboring communities without it.11 Of the 33 melanomas found through the screening program, 39% (13/33) were in situ lesions, 55% (18/33) were thin (<1 mm) invasive lesions, and 6% (2/33) were 1-mm thick or greater.16 Within the population of Queensland during the period from 1999 through 2002, 36% were in situ lesions, 48% were invasive thin melanomas, and 16% were invasive melanomas 1-mm thick or more, indicating that melanomas found through screening were thinner or less advanced.17
Comment
Our review identified 5 studies describing the impact of PSEs for melanoma screening on tumor thickness at diagnosis and melanoma mortality. Key findings are highlighted in Figure 2. Our findings suggest that PSEs are associated with a decline in melanoma tumor thickness and melanoma-specific mortality. Our findings are qualitatively similar to prior reviews that supported the use of PSEs to detect thinner melanomas and improve mortality outcomes.18-20
The greatest evidence for population-based screening programs was provided by the SCREEN study. This landmark study documented that screening programs utilizing primary care physicians (PCPs) and dermatologists can lead to a reduction in melanoma mortality.15 Findings from the study led to the countrywide expansion of the screening program in 2008, leading to 45 million Germans eligible for skin cancer screenings every 2 years.21 Nearly two-thirds of dermatologists (N=1348) were satisfied with routine PSE and 83% perceived a better quality of health care for skin with the 2008 expansion.22
Data suggest that physician-detected melanomas through PSEs or routine physical examinations are thinner at the time of diagnosis than those found by patients or their partners.14,23-26 Terushkin and Halpern20 analyzed 9 worldwide studies encompassing more than 7500 patients and found that physician-detected melanomas were 0.55 mm thinner than those detected by patients or their significant others. The workplace screening and education program reviewed herein also reported a reduction in thicker melanomas and melanoma mortality during the study period.12
Not all Americans have a regular dermatologist. As such, educating PCPs in skin cancer detection has been a recent area of study. The premise is that the skin examination can be integrated into routine physical examinations conducted by PCPs. The previously discussed studies, particularly Aitken et al,14 Schneider et al,12 and Katalinic et al,15 as well as the SCREEN program studies,15 suggest that integration of the skin examination into the routine physical examination may be a feasible method to reduce melanoma thickness and mortality. Furthermore, the SCREEN study15 identified participants with risk factors for melanoma, finding that approximately half of men and women (N=360,288) had at least one melanoma risk factor, which suggests that it may be more practical to design screening practices around high-risk participants.
Several studies were excluded from our analysis on the basis of study design, including cross-sectional observational studies; however, it is worth briefly commenting on the findings of the excluded studies here, as they add to the body of literature. A community-based, multi-institutional study of 566 adults with invasive melanoma assessed the role of PSEs in the year prior to diagnosis by interviewing participants in clinic within 3 months of melanoma diagnosis.24 Patients who underwent full-body PSE in the year prior to diagnosis were more than 2 times more likely to have thinner (≤1 mm) melanomas (OR, 2.51; 95% CI, 1.62-3.87]). Notably, men older than 60 years appeared to benefit the most from this practice; men in this age group contributed greatly to the observed effect, likely because they had 4 times the odds of a thinner melanoma (OR, 4.09; 95% CI, 1.88-8.89]). Thinner melanomas also were associated with an age of 60 years or younger, female sex, and higher education level.24
Pollitt et al27 analyzed the association between prediagnosis Medicaid enrollment status and melanoma tumor thickness. The study found that men and women who intermittently enrolled in Medicaid or were not enrolled until the month of diagnosis had an increased chance of late-stage melanoma when compared to other patients. Patients who continuously enrolled during the year prior to diagnosis had lower odds for thicker melanomas, suggesting that these patients had greater access to screening examinations.27
Roetzheim et al28 analyzed data from the SEER-Medicare linked dataset to investigate patterns of dermatologist and PCP visits in the 2 years before melanoma diagnosis. Medicare beneficiaries seeing both a dermatologist and a PCP prior to melanoma diagnosis had greater odds of a thinner melanoma and lower melanoma mortality compared to patients without such visits.28
Durbec et al29 conducted a retrospective, population-based study of 650 patients in France who were seen by a dermatologist for melanoma. The thinnest melanomas were reported in patients seeing a dermatologist for prospective follow-up of nevi or consulting a dermatologist for other diseases. Patients referred to a dermatologist by PCPs tended to be older and had the highest frequency of thick (>3 mm), nodular, and/or ulcerated melanomas,29 which could be interpreted as a need for greater PCP education in melanoma screening.
Rates of skin examinations have been increasing since the year 2000, both overall and among high-risk groups as reported by a recent study on skin cancer screening trends. Prevalence of having at least one total-body skin examination increased from 14.5% in 2000 to 16.5% in 2005 to 19.8% in 2010 (P<.0001).30 One study revealed a practice gap in which more than 3 in 10 PCPs and 1 in 10 dermatologists reported not screening more than half their high-risk patients for skin cancer.31 The major obstacle to narrowing the identified practice gap involves establishing a national strategy to screen high-risk individuals for skin cancer and requires partnerships among patients, PCPs, specialists, policy makers, and government sponsors.
Lack of evidence that screening for skin cancer with PSEs reduces overall mortality does not mean there is a lack of lifesaving potential of screenings. The resources required to execute a randomized controlled trial with adequate power are vast, as the USPSTF estimated 800,000 participants would be needed.4 Barriers to conducting a randomized clinical trial for skin cancer screening include the large sample size required, prolonged follow-up, and various ethical issues such as withholding screening for a cancer that is potentially curable in early stages. Lessons from screenings for breast and prostate cancers have taught us that such randomized controlled trials assessing cancer screening are costly and do not always produce definitive answers.32
Conclusion
Although proof of improved health outcomes from randomized controlled trials is still required, there is evidence to support targeted screening programs for the detection of thinner melanomas and, by proxy, reduced melanoma mortality. Amidst the health care climate change and payment reform, recommendations from national organizations on melanoma screenings are paramount. Clinicians should continue to offer regular skin examinations as the body of evidence continues to grow in support of PSEs for melanoma screening.
Acknowledgments—We are grateful to Mary Butler, PhD, and Robert Kane, MD, both from Minneapolis, Minnesota, for their guidance and consultation.
1. American Cancer Society. Cancer Facts & Figures 2015. Atlanta, GA: American Cancer Society; 2015. http: //www.cancer.org/Research/CancerFactsStatistics/cancer factsfigures2015/cancer-facts-and-figures-2015. Accessed July 6, 2015.
2. Guy G Jr, Ekwueme D, Tangka F, et al. Melanoma treatment costs: a systematic review of the literature, 1990-2011. Am J Prev. 2012;43:537-545.
3. Margolis D, Halpern A, Rebbeck T, et al. Validation of a melanoma prognostic model. Arch Dermatol. 1998;134:1597-1601.
4. Wolff T, Tai E, Miller T. Screening for skin cancer: an update of the evidence for the U.S. Preventative Services Task Force. Ann Intern Med. 2009;150:194-198.
5. American Academy of Dermatology. Melanoma
Monday. http://www.aad.org/spot-skin-cancer
/community-programs-events/melanoma-monday. Accessed August 19, 2015.
6. Higgins JPT, Green S, eds. Cochrane Handbook for
Systematic Reviews of Interventions Version 5.1.0 [updated March 2011]. The Cochrane Collaboration, 2011. http:
//www.cochrane-handbook.org. Updated March 2011. Accessed November 10, 2014.
7. Viswanathan M, Berkman N. Development of the RTI item bank on risk of bias and precision of observational studies. J Clin Epidemiol. 2012;65:163-178.
8. Moher D, Liberati A, Tetzlaff J, et al; PRISMA group. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement [published online ahead of print July 23, 2009]. J Clin Epidemiol. 2009;62:1006-1012.
9. Berwick M, Armstrong B, Ben-Porat L. Sun exposure and mortality from melanoma. J Natl Cancer Inst. 2005;97:195-199.
10. Berwick M, Begg C, Fine J, et al. Screening for cutaneous melanoma by skin self-examination. J Natl Cancer Inst. 1996;88:17-23.
11. Aitken J, Elwood J, Lowe J, et al. A randomised trial of population screening for melanoma. J Med Screen. 2002;9:33-37.
12. Schneider J, Moore D, Mendelsohn M. Screening program reduced melanoma mortality at the Lawrence Livermore National Laboratory, 1984 to 1996. J Am Acad Dermatol. 2008;58:741-749.
13. Expert Health Data Programming Inc. Health data software and health statistics. Available from: http: //www.ehdp.com. Accessed April 1, 2001. Cited by: Schneider J, Moore D, Mendelsohn M. Screening program reduced melanoma mortality at the Lawrence Livermore National Laboratory, 1984 to 1996. J Am Acad Dermatol. 2008;58:741-749.
14. Aitken J, Elwood M, Baade P, et al. Clinical whole-body skin examination reduces the incidence of thick melanomas. Int J Cancer. 2010;126:450-458.
15. Katalinic A, Waldmann A, Weinstock M, et al. Does skin cancer screening save lives? an observational study comparing trends in melanoma mortality in regions with and without screening. Cancer. 2012;118:5395-5402.
16. Aitken J, Janda M, Elwood M, et al. Clinical outcomes from skin screening clinics within a community-based melanoma screening program. J Am Acad Dermatol. 2006;54:105-114.
17. Coory M, Baade P, Aitken JF, et al. Trends for in-situ and invasive melanoma in Queensland, Australia, 1982 to 2002. Cancer Causes Control. 2006;17:21-27.
18. Mayer JE, Swetter SM, Fu T, et al. Screening, early detection, education, and trends for melanoma: current status (2007-2013) and future directions: part II. screening, education, and future directions. J Am Acad Dermatol. 2014;71:611.e1-611.e10; quiz, 621-622.
19. Curiel-Lewandrowski C, Chen S, Swetter S, et al. Screening and prevention measures for melanoma: is there a survival advantage? Curr Oncol Rep. 2012;14:458-467.
20. Terushkin V, Halpern A. Melanoma early detection. Hematol Oncol Clin North Am. 2009;23:481-500.
21. Geller A, Greinert R, Sinclair C, et al. A nationwide population-based skin cancer screening in Germany: proceedings of the first meeting of the International Task Force on Skin Cancer Screening and Prevention (September 24 and 25, 2009) [published online ahead of print April 8, 2010]. Cancer Epidemiol. 2010;34:355-358.
22. Kornek T, Schafer I, Reusch M, et al. Routine skin cancer screening in Germany: four years of experience from the dermatologists’ perspective. Dermatology. 2012;225:289-293.
23. De Giorgi V, Grazzini M, Rossari S, et al. Is skin self-examination for cutaneous melanoma detection still adequate? a retrospective study. Dermatology. 2012;225:31-36.
24. Swetter S, Johnson T, Miller D, et al. Melanoma in middle-aged and older men: a multi-institutional survey study of factors related to tumor thickness. Arch Dermatol. 2009;145:397-404.
25. Kantor J, Kantor D. Routine dermatologist-performed full-body skin examination and early melanoma detection. Arch Dermatol. 2009;145:873-876.
26. Kovalyshyn I, Dusza S, Siamas K, et al. The impact of physician screening on melanoma detection. Arch Dermatol. 2011;147:1269-1275.
27. Pollitt R, Clarke C, Shema S, et al. California Medicaid enrollment and melanoma stage at diagnosis: a population-based study. Am J Prev Med. 2008;35:7-13.
28. Roetzheim R, Lee J, Ferrante J, et al. The influence of dermatologist and primary care physician visits on melanoma outcomes among Medicare beneficiaries. J Am Board Fam Med. 2013;26:637-647.
29. Durbec F, Vitry F, Granel-Brocard F, et al. The role of circumstances of diagnosis and access to dermatological care in early diagnosis of cutaneous melanoma: a population-based study in France. Arch Dermatol. 2010;146:240-246.
30. Lakhani N, Saraiya M, Thompson T, et al. Total body skin examination for skin cancer screening among U.S. adults from 2000 to 2010. Prev Med. 2014;61:75-80.
31. Oliveria SA, Heneghan MK, Cushman LF, et al. Skin cancer screening by dermatologists, family practitioners, and internists: barriers and facilitating factors. Arch Dermatol. 2011;147:39-44.
32. Bigby M. Why the evidence for skin cancer screening is insufficient: lessons from prostate cancer screening. Arch Dermatol. 2010;146:322-324.
1. American Cancer Society. Cancer Facts & Figures 2015. Atlanta, GA: American Cancer Society; 2015. http: //www.cancer.org/Research/CancerFactsStatistics/cancer factsfigures2015/cancer-facts-and-figures-2015. Accessed July 6, 2015.
2. Guy G Jr, Ekwueme D, Tangka F, et al. Melanoma treatment costs: a systematic review of the literature, 1990-2011. Am J Prev. 2012;43:537-545.
3. Margolis D, Halpern A, Rebbeck T, et al. Validation of a melanoma prognostic model. Arch Dermatol. 1998;134:1597-1601.
4. Wolff T, Tai E, Miller T. Screening for skin cancer: an update of the evidence for the U.S. Preventative Services Task Force. Ann Intern Med. 2009;150:194-198.
5. American Academy of Dermatology. Melanoma
Monday. http://www.aad.org/spot-skin-cancer
/community-programs-events/melanoma-monday. Accessed August 19, 2015.
6. Higgins JPT, Green S, eds. Cochrane Handbook for
Systematic Reviews of Interventions Version 5.1.0 [updated March 2011]. The Cochrane Collaboration, 2011. http:
//www.cochrane-handbook.org. Updated March 2011. Accessed November 10, 2014.
7. Viswanathan M, Berkman N. Development of the RTI item bank on risk of bias and precision of observational studies. J Clin Epidemiol. 2012;65:163-178.
8. Moher D, Liberati A, Tetzlaff J, et al; PRISMA group. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement [published online ahead of print July 23, 2009]. J Clin Epidemiol. 2009;62:1006-1012.
9. Berwick M, Armstrong B, Ben-Porat L. Sun exposure and mortality from melanoma. J Natl Cancer Inst. 2005;97:195-199.
10. Berwick M, Begg C, Fine J, et al. Screening for cutaneous melanoma by skin self-examination. J Natl Cancer Inst. 1996;88:17-23.
11. Aitken J, Elwood J, Lowe J, et al. A randomised trial of population screening for melanoma. J Med Screen. 2002;9:33-37.
12. Schneider J, Moore D, Mendelsohn M. Screening program reduced melanoma mortality at the Lawrence Livermore National Laboratory, 1984 to 1996. J Am Acad Dermatol. 2008;58:741-749.
13. Expert Health Data Programming Inc. Health data software and health statistics. Available from: http: //www.ehdp.com. Accessed April 1, 2001. Cited by: Schneider J, Moore D, Mendelsohn M. Screening program reduced melanoma mortality at the Lawrence Livermore National Laboratory, 1984 to 1996. J Am Acad Dermatol. 2008;58:741-749.
14. Aitken J, Elwood M, Baade P, et al. Clinical whole-body skin examination reduces the incidence of thick melanomas. Int J Cancer. 2010;126:450-458.
15. Katalinic A, Waldmann A, Weinstock M, et al. Does skin cancer screening save lives? an observational study comparing trends in melanoma mortality in regions with and without screening. Cancer. 2012;118:5395-5402.
16. Aitken J, Janda M, Elwood M, et al. Clinical outcomes from skin screening clinics within a community-based melanoma screening program. J Am Acad Dermatol. 2006;54:105-114.
17. Coory M, Baade P, Aitken JF, et al. Trends for in-situ and invasive melanoma in Queensland, Australia, 1982 to 2002. Cancer Causes Control. 2006;17:21-27.
18. Mayer JE, Swetter SM, Fu T, et al. Screening, early detection, education, and trends for melanoma: current status (2007-2013) and future directions: part II. screening, education, and future directions. J Am Acad Dermatol. 2014;71:611.e1-611.e10; quiz, 621-622.
19. Curiel-Lewandrowski C, Chen S, Swetter S, et al. Screening and prevention measures for melanoma: is there a survival advantage? Curr Oncol Rep. 2012;14:458-467.
20. Terushkin V, Halpern A. Melanoma early detection. Hematol Oncol Clin North Am. 2009;23:481-500.
21. Geller A, Greinert R, Sinclair C, et al. A nationwide population-based skin cancer screening in Germany: proceedings of the first meeting of the International Task Force on Skin Cancer Screening and Prevention (September 24 and 25, 2009) [published online ahead of print April 8, 2010]. Cancer Epidemiol. 2010;34:355-358.
22. Kornek T, Schafer I, Reusch M, et al. Routine skin cancer screening in Germany: four years of experience from the dermatologists’ perspective. Dermatology. 2012;225:289-293.
23. De Giorgi V, Grazzini M, Rossari S, et al. Is skin self-examination for cutaneous melanoma detection still adequate? a retrospective study. Dermatology. 2012;225:31-36.
24. Swetter S, Johnson T, Miller D, et al. Melanoma in middle-aged and older men: a multi-institutional survey study of factors related to tumor thickness. Arch Dermatol. 2009;145:397-404.
25. Kantor J, Kantor D. Routine dermatologist-performed full-body skin examination and early melanoma detection. Arch Dermatol. 2009;145:873-876.
26. Kovalyshyn I, Dusza S, Siamas K, et al. The impact of physician screening on melanoma detection. Arch Dermatol. 2011;147:1269-1275.
27. Pollitt R, Clarke C, Shema S, et al. California Medicaid enrollment and melanoma stage at diagnosis: a population-based study. Am J Prev Med. 2008;35:7-13.
28. Roetzheim R, Lee J, Ferrante J, et al. The influence of dermatologist and primary care physician visits on melanoma outcomes among Medicare beneficiaries. J Am Board Fam Med. 2013;26:637-647.
29. Durbec F, Vitry F, Granel-Brocard F, et al. The role of circumstances of diagnosis and access to dermatological care in early diagnosis of cutaneous melanoma: a population-based study in France. Arch Dermatol. 2010;146:240-246.
30. Lakhani N, Saraiya M, Thompson T, et al. Total body skin examination for skin cancer screening among U.S. adults from 2000 to 2010. Prev Med. 2014;61:75-80.
31. Oliveria SA, Heneghan MK, Cushman LF, et al. Skin cancer screening by dermatologists, family practitioners, and internists: barriers and facilitating factors. Arch Dermatol. 2011;147:39-44.
32. Bigby M. Why the evidence for skin cancer screening is insufficient: lessons from prostate cancer screening. Arch Dermatol. 2010;146:322-324.
Practice Points
- Current guidelines regarding melanoma screening are inconsistent.
- There is a growing pool of evidence supporting screening to improve melanoma outcomes.