User login
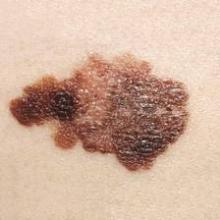
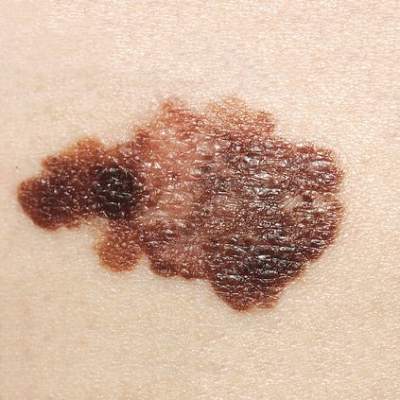
Intermittent interferon falls short for stage III melanoma patients
For patients with resected stage III melanoma, intermittent intravenous high-dose IFN–alpha-2b (iHDI) is not superior to conventional 1-year high-dose IFN–alpha-2b therapy, according to results from a phase III study. Relapse-free survival was significantly inferior with the intermittent regimen.
“These data preclude speculation about a possible equivalence of the regimens. Taken together, the approved schedules for adjuvant IFN [interferon] treatment of patients with melanoma are likely to remain the standard of care until new approaches, such as with ipilimumab or other drugs, show durable improvements in RFS or OS and acceptable safety profiles,” wrote Dr. Peter Mohr of Elbe-Klinikum Buxtehude, Germany, and his colleagues (J Clin. Onc. 2015 Oct. 26. doi: 10.1200/JCO.2015.59.6932).
The prospective, multicenter, phase III randomized trial included 649 patients with stage III melanoma who received either three courses of iHDI or the conventional HDI 1-year regimen.After a median follow-up time of 55 months, distant metastasis-free survival and overall survival for the two treatment schedules were similar. The 5-year distant metastasis-free survival for iHDI, compared with HDI, was 49.2% vs. 53.1%; 5-year overall survival was 62.9% vs. 64.1%, respectively. However, relapse-free survival was significantly worse for iHDI than for HDI (hazard ratio, 1.27; 95% confidence interval, 1.02-1.59; P = .03).
Adverse events (AEs) were similar for the two arms, except the HDI group experienced more anemia than the iHDI group (48.1% vs. 30.9%; P less than .001) and neutropenia of grade 3 or higher was more frequent in the iHDI group (38.9% vs. 30.1%; P = .004). The intermittent schedule resulted in fewer cumulative AEs, particularly grade 3 or 4 fatigue. Fatigue is an expected but often debilitating AE of HDI therapy, the investigators noted.
Reduced fatigue was associated with 40% less early discontinuation with iHDI than with standard HDI. In the iHDI group, quality of life measures returned to or exceeded baseline values within 4 weeks of administration. However, improvements in quality of life should be weighed against potential loss of efficacy: at least 50% of patients would tolerate moderate or severe IFN–alpha-2b toxicity for improvement in 5-year, disease-free survival, the investigators said.
For patients with resected stage III melanoma, intermittent intravenous high-dose IFN–alpha-2b (iHDI) is not superior to conventional 1-year high-dose IFN–alpha-2b therapy, according to results from a phase III study. Relapse-free survival was significantly inferior with the intermittent regimen.
“These data preclude speculation about a possible equivalence of the regimens. Taken together, the approved schedules for adjuvant IFN [interferon] treatment of patients with melanoma are likely to remain the standard of care until new approaches, such as with ipilimumab or other drugs, show durable improvements in RFS or OS and acceptable safety profiles,” wrote Dr. Peter Mohr of Elbe-Klinikum Buxtehude, Germany, and his colleagues (J Clin. Onc. 2015 Oct. 26. doi: 10.1200/JCO.2015.59.6932).
The prospective, multicenter, phase III randomized trial included 649 patients with stage III melanoma who received either three courses of iHDI or the conventional HDI 1-year regimen.After a median follow-up time of 55 months, distant metastasis-free survival and overall survival for the two treatment schedules were similar. The 5-year distant metastasis-free survival for iHDI, compared with HDI, was 49.2% vs. 53.1%; 5-year overall survival was 62.9% vs. 64.1%, respectively. However, relapse-free survival was significantly worse for iHDI than for HDI (hazard ratio, 1.27; 95% confidence interval, 1.02-1.59; P = .03).
Adverse events (AEs) were similar for the two arms, except the HDI group experienced more anemia than the iHDI group (48.1% vs. 30.9%; P less than .001) and neutropenia of grade 3 or higher was more frequent in the iHDI group (38.9% vs. 30.1%; P = .004). The intermittent schedule resulted in fewer cumulative AEs, particularly grade 3 or 4 fatigue. Fatigue is an expected but often debilitating AE of HDI therapy, the investigators noted.
Reduced fatigue was associated with 40% less early discontinuation with iHDI than with standard HDI. In the iHDI group, quality of life measures returned to or exceeded baseline values within 4 weeks of administration. However, improvements in quality of life should be weighed against potential loss of efficacy: at least 50% of patients would tolerate moderate or severe IFN–alpha-2b toxicity for improvement in 5-year, disease-free survival, the investigators said.
For patients with resected stage III melanoma, intermittent intravenous high-dose IFN–alpha-2b (iHDI) is not superior to conventional 1-year high-dose IFN–alpha-2b therapy, according to results from a phase III study. Relapse-free survival was significantly inferior with the intermittent regimen.
“These data preclude speculation about a possible equivalence of the regimens. Taken together, the approved schedules for adjuvant IFN [interferon] treatment of patients with melanoma are likely to remain the standard of care until new approaches, such as with ipilimumab or other drugs, show durable improvements in RFS or OS and acceptable safety profiles,” wrote Dr. Peter Mohr of Elbe-Klinikum Buxtehude, Germany, and his colleagues (J Clin. Onc. 2015 Oct. 26. doi: 10.1200/JCO.2015.59.6932).
The prospective, multicenter, phase III randomized trial included 649 patients with stage III melanoma who received either three courses of iHDI or the conventional HDI 1-year regimen.After a median follow-up time of 55 months, distant metastasis-free survival and overall survival for the two treatment schedules were similar. The 5-year distant metastasis-free survival for iHDI, compared with HDI, was 49.2% vs. 53.1%; 5-year overall survival was 62.9% vs. 64.1%, respectively. However, relapse-free survival was significantly worse for iHDI than for HDI (hazard ratio, 1.27; 95% confidence interval, 1.02-1.59; P = .03).
Adverse events (AEs) were similar for the two arms, except the HDI group experienced more anemia than the iHDI group (48.1% vs. 30.9%; P less than .001) and neutropenia of grade 3 or higher was more frequent in the iHDI group (38.9% vs. 30.1%; P = .004). The intermittent schedule resulted in fewer cumulative AEs, particularly grade 3 or 4 fatigue. Fatigue is an expected but often debilitating AE of HDI therapy, the investigators noted.
Reduced fatigue was associated with 40% less early discontinuation with iHDI than with standard HDI. In the iHDI group, quality of life measures returned to or exceeded baseline values within 4 weeks of administration. However, improvements in quality of life should be weighed against potential loss of efficacy: at least 50% of patients would tolerate moderate or severe IFN–alpha-2b toxicity for improvement in 5-year, disease-free survival, the investigators said.
Key clinical point: Distant metastasis-free survival and overall survival were similar in patients with stage III melanoma treated with intermittent high-dose intravenous IFN–alpha-2b (iHDI) vs. standard 1-year high-dose IFN–alpha-2b (HDI), but the difference for relapse-free survival was significant (HR, 1.27; P = .03), favoring standard HDI.
Major finding: The 5-year distant metastasis-free survival for iHDI, compared with HDI was 49.2% vs. 53.1%; 5-year overall survival was 62.9% vs. 64.1%, respectively. Relapse-free survival was significantly worse for iHDI than HDI (HR, 1.27; 95% CI, 1.02-1.59; P = .03).
Data source: The prospective, multicenter, phase III trial randomized 649 patients with stage III melanoma to receive either three courses of iHDI or the conventional HDI 1-year regimen.
Disclosures: Dr. Mohr reported financial relationships with Merck, Bristol-Myers Squibb, Roche, GlaxoSmithKline, LEO Pharma, Novartis, Merck Sharp & Dohme, and Roche. Several of his coauthors reported having ties to industry sources.
Nonmelanoma skin cancer initially misdiagnosed in 36% of small cohort
In a small sample study, 10 (36%) of 28 children and young adults diagnosed with nonmelanoma skin cancer were given a misdiagnosis initially, suggesting that young patients with NMSC risk factors may require heightened monitoring from health care providers, according to a study published in Journal of the American Academy of Dermatology (2015 doi: 10.1016/j.jaad.2015.08.007).
In their efforts to identify potential risk factors and gaps in care associated with NMSC in pediatric populations, lead author Hasan Khosravi of Harvard Medical School, Boston, and his associates examined records from 28 patients and 182 occurrences of NMSC, collected from Boston Children’s Hospital between 1993 and 2014.
Thirteen (46%) of the 28 pediatric NMSC patients had a history of prolonged immunosuppression, radiation therapy, chemotherapy, voriconazole use, or a combination of these. Among these 28 patients, 19 were diagnosed with basal cell carcinoma (BCC), 7 were diagnosed with squamous cell carcinoma (SCC), and 2 were diagnosed with both BCC and SCC.
The authors noted significant delays in the initial diagnosis in both types of cancer; the mean number of days from time of lesion onset to diagnosis was 667 for SCC and 1,176 for BCC. When misdiagnosed, carcinomas were incorrectly identified as viral wart and graft-versus-host disease for SCC, and psoriasis, acrochordon, wart, nevus, and atypical nevus for BCC.
Even so, interventions were effective in the pediatric population, the authors noted.
“Although most of our patients developed subsequent NMSC after their initial diagnosis, the majority of cases were treated successfully with surgical excision, without recurrence or spread of disease. This suggests that interventions in children and young adults that involve prevention of subsequent disease may be most impactful,” they wrote.
The researchers had no conflicts to declare.
In a small sample study, 10 (36%) of 28 children and young adults diagnosed with nonmelanoma skin cancer were given a misdiagnosis initially, suggesting that young patients with NMSC risk factors may require heightened monitoring from health care providers, according to a study published in Journal of the American Academy of Dermatology (2015 doi: 10.1016/j.jaad.2015.08.007).
In their efforts to identify potential risk factors and gaps in care associated with NMSC in pediatric populations, lead author Hasan Khosravi of Harvard Medical School, Boston, and his associates examined records from 28 patients and 182 occurrences of NMSC, collected from Boston Children’s Hospital between 1993 and 2014.
Thirteen (46%) of the 28 pediatric NMSC patients had a history of prolonged immunosuppression, radiation therapy, chemotherapy, voriconazole use, or a combination of these. Among these 28 patients, 19 were diagnosed with basal cell carcinoma (BCC), 7 were diagnosed with squamous cell carcinoma (SCC), and 2 were diagnosed with both BCC and SCC.
The authors noted significant delays in the initial diagnosis in both types of cancer; the mean number of days from time of lesion onset to diagnosis was 667 for SCC and 1,176 for BCC. When misdiagnosed, carcinomas were incorrectly identified as viral wart and graft-versus-host disease for SCC, and psoriasis, acrochordon, wart, nevus, and atypical nevus for BCC.
Even so, interventions were effective in the pediatric population, the authors noted.
“Although most of our patients developed subsequent NMSC after their initial diagnosis, the majority of cases were treated successfully with surgical excision, without recurrence or spread of disease. This suggests that interventions in children and young adults that involve prevention of subsequent disease may be most impactful,” they wrote.
The researchers had no conflicts to declare.
In a small sample study, 10 (36%) of 28 children and young adults diagnosed with nonmelanoma skin cancer were given a misdiagnosis initially, suggesting that young patients with NMSC risk factors may require heightened monitoring from health care providers, according to a study published in Journal of the American Academy of Dermatology (2015 doi: 10.1016/j.jaad.2015.08.007).
In their efforts to identify potential risk factors and gaps in care associated with NMSC in pediatric populations, lead author Hasan Khosravi of Harvard Medical School, Boston, and his associates examined records from 28 patients and 182 occurrences of NMSC, collected from Boston Children’s Hospital between 1993 and 2014.
Thirteen (46%) of the 28 pediatric NMSC patients had a history of prolonged immunosuppression, radiation therapy, chemotherapy, voriconazole use, or a combination of these. Among these 28 patients, 19 were diagnosed with basal cell carcinoma (BCC), 7 were diagnosed with squamous cell carcinoma (SCC), and 2 were diagnosed with both BCC and SCC.
The authors noted significant delays in the initial diagnosis in both types of cancer; the mean number of days from time of lesion onset to diagnosis was 667 for SCC and 1,176 for BCC. When misdiagnosed, carcinomas were incorrectly identified as viral wart and graft-versus-host disease for SCC, and psoriasis, acrochordon, wart, nevus, and atypical nevus for BCC.
Even so, interventions were effective in the pediatric population, the authors noted.
“Although most of our patients developed subsequent NMSC after their initial diagnosis, the majority of cases were treated successfully with surgical excision, without recurrence or spread of disease. This suggests that interventions in children and young adults that involve prevention of subsequent disease may be most impactful,” they wrote.
The researchers had no conflicts to declare.
FROM JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
New systemic therapies altering management of melanoma brain metastases
SEATTLE – Targeted therapies for melanoma are improving outcomes for patients with brain metastases and changing management of this dreaded complication, according to Dr. John A. Thompson, codirector of the Melanoma Clinic at the Seattle Cancer Care Alliance and a professor in the medical oncology division at the University of Washington, both in Seattle.
“We are in a new era of treatment of brain metastasis. It’s no longer just surgery and radiation therapy, but a multidisciplinary approach involving medical oncology, radiation oncology, and surgery,” he told attendees of the World Cutaneous Malignancies Congress.
Without question, surgery and radiation therapy techniques have advanced over time, and these modalities remain helpful in achieving local control of disease. Additionally, greater experience with them has improved patient selection.
For example, research has identified several predictors of benefit from stereotactic radiosurgery, such as fewer brain metastases and better performance status (J Neurosurg. 2011;114:769-79). But overall survival was fairly poor given disease elsewhere in the body. “Here’s where I think the new developments in systemic therapy may hopefully be able to start improving this curve,” Dr. Thompson said.
Toxicity of some forms of radiation therapy also remains problematic. For example, a recent randomized phase III trial in patients with up to three brain metastases showed that addition of whole-brain radiation therapy to radiosurgery improved intracranial tumor control, but at the expense of more rapid cognitive decline (ASCO 2015. Abstract LBA4). And there was no gain in overall survival.
“The changes that have happened in systemic therapy over the past 5-10 years have been very exciting,” Dr. Thompson said. By way of example, he pointed to a phase II trial that tested ipilimumab (Yervoy), an antibody to the T-cell receptor cytotoxic T lymphocyte–associated antigen 4 (CTLA4), as induction therapy and then maintenance therapy among patients with melanoma who had brain metastases (Lancet Oncol. 2012;13:459-65).
The subset of patients who were neurologically stable and not on systemic steroids had a disease control rate of 24%, with some having objective responses, including ones in the CNS; the value was lower, at 10%, among patients who were neurologically symptomatic and on steroids, indicating more advanced disease. The 2-year overall survival rate was 26%.
“I think this is encouraging if you compare [these patients] to historical control patients with brain metastases,” said Dr. Thompson, who disclosed that he performs contracted research with Agensys, BMS, Merck, Novartis, and Pfizer.
Another phase II trial, CheckMate 204, which is currently enrolling patients, is taking the concept further, testing the combination of ipilimumab and nivolumab (Opdivo), an antibody to the cell surface receptor programmed death-1 (PD-1), as induction therapy followed by nivolumab maintenance therapy among patients with melanoma who have brain metastases. “This is I think a high-priority trial,” he asserted.
Discussing some cases from his own practice, Dr. Thompson described patient-tailored integration of the new systemic therapies with surgery and radiation therapy.
Oncologists must often deal with toxicities of these therapies as well, such as the colitis and hypophysitis related to ipilimumab therapy, and the development of resistance over time, Dr. Thompson acknowledged.
Nonetheless, the multimodality approach has led to regression and elimination of brain metastases, and even allowed some patients to achieve remission, he reported.
SEATTLE – Targeted therapies for melanoma are improving outcomes for patients with brain metastases and changing management of this dreaded complication, according to Dr. John A. Thompson, codirector of the Melanoma Clinic at the Seattle Cancer Care Alliance and a professor in the medical oncology division at the University of Washington, both in Seattle.
“We are in a new era of treatment of brain metastasis. It’s no longer just surgery and radiation therapy, but a multidisciplinary approach involving medical oncology, radiation oncology, and surgery,” he told attendees of the World Cutaneous Malignancies Congress.
Without question, surgery and radiation therapy techniques have advanced over time, and these modalities remain helpful in achieving local control of disease. Additionally, greater experience with them has improved patient selection.
For example, research has identified several predictors of benefit from stereotactic radiosurgery, such as fewer brain metastases and better performance status (J Neurosurg. 2011;114:769-79). But overall survival was fairly poor given disease elsewhere in the body. “Here’s where I think the new developments in systemic therapy may hopefully be able to start improving this curve,” Dr. Thompson said.
Toxicity of some forms of radiation therapy also remains problematic. For example, a recent randomized phase III trial in patients with up to three brain metastases showed that addition of whole-brain radiation therapy to radiosurgery improved intracranial tumor control, but at the expense of more rapid cognitive decline (ASCO 2015. Abstract LBA4). And there was no gain in overall survival.
“The changes that have happened in systemic therapy over the past 5-10 years have been very exciting,” Dr. Thompson said. By way of example, he pointed to a phase II trial that tested ipilimumab (Yervoy), an antibody to the T-cell receptor cytotoxic T lymphocyte–associated antigen 4 (CTLA4), as induction therapy and then maintenance therapy among patients with melanoma who had brain metastases (Lancet Oncol. 2012;13:459-65).
The subset of patients who were neurologically stable and not on systemic steroids had a disease control rate of 24%, with some having objective responses, including ones in the CNS; the value was lower, at 10%, among patients who were neurologically symptomatic and on steroids, indicating more advanced disease. The 2-year overall survival rate was 26%.
“I think this is encouraging if you compare [these patients] to historical control patients with brain metastases,” said Dr. Thompson, who disclosed that he performs contracted research with Agensys, BMS, Merck, Novartis, and Pfizer.
Another phase II trial, CheckMate 204, which is currently enrolling patients, is taking the concept further, testing the combination of ipilimumab and nivolumab (Opdivo), an antibody to the cell surface receptor programmed death-1 (PD-1), as induction therapy followed by nivolumab maintenance therapy among patients with melanoma who have brain metastases. “This is I think a high-priority trial,” he asserted.
Discussing some cases from his own practice, Dr. Thompson described patient-tailored integration of the new systemic therapies with surgery and radiation therapy.
Oncologists must often deal with toxicities of these therapies as well, such as the colitis and hypophysitis related to ipilimumab therapy, and the development of resistance over time, Dr. Thompson acknowledged.
Nonetheless, the multimodality approach has led to regression and elimination of brain metastases, and even allowed some patients to achieve remission, he reported.
SEATTLE – Targeted therapies for melanoma are improving outcomes for patients with brain metastases and changing management of this dreaded complication, according to Dr. John A. Thompson, codirector of the Melanoma Clinic at the Seattle Cancer Care Alliance and a professor in the medical oncology division at the University of Washington, both in Seattle.
“We are in a new era of treatment of brain metastasis. It’s no longer just surgery and radiation therapy, but a multidisciplinary approach involving medical oncology, radiation oncology, and surgery,” he told attendees of the World Cutaneous Malignancies Congress.
Without question, surgery and radiation therapy techniques have advanced over time, and these modalities remain helpful in achieving local control of disease. Additionally, greater experience with them has improved patient selection.
For example, research has identified several predictors of benefit from stereotactic radiosurgery, such as fewer brain metastases and better performance status (J Neurosurg. 2011;114:769-79). But overall survival was fairly poor given disease elsewhere in the body. “Here’s where I think the new developments in systemic therapy may hopefully be able to start improving this curve,” Dr. Thompson said.
Toxicity of some forms of radiation therapy also remains problematic. For example, a recent randomized phase III trial in patients with up to three brain metastases showed that addition of whole-brain radiation therapy to radiosurgery improved intracranial tumor control, but at the expense of more rapid cognitive decline (ASCO 2015. Abstract LBA4). And there was no gain in overall survival.
“The changes that have happened in systemic therapy over the past 5-10 years have been very exciting,” Dr. Thompson said. By way of example, he pointed to a phase II trial that tested ipilimumab (Yervoy), an antibody to the T-cell receptor cytotoxic T lymphocyte–associated antigen 4 (CTLA4), as induction therapy and then maintenance therapy among patients with melanoma who had brain metastases (Lancet Oncol. 2012;13:459-65).
The subset of patients who were neurologically stable and not on systemic steroids had a disease control rate of 24%, with some having objective responses, including ones in the CNS; the value was lower, at 10%, among patients who were neurologically symptomatic and on steroids, indicating more advanced disease. The 2-year overall survival rate was 26%.
“I think this is encouraging if you compare [these patients] to historical control patients with brain metastases,” said Dr. Thompson, who disclosed that he performs contracted research with Agensys, BMS, Merck, Novartis, and Pfizer.
Another phase II trial, CheckMate 204, which is currently enrolling patients, is taking the concept further, testing the combination of ipilimumab and nivolumab (Opdivo), an antibody to the cell surface receptor programmed death-1 (PD-1), as induction therapy followed by nivolumab maintenance therapy among patients with melanoma who have brain metastases. “This is I think a high-priority trial,” he asserted.
Discussing some cases from his own practice, Dr. Thompson described patient-tailored integration of the new systemic therapies with surgery and radiation therapy.
Oncologists must often deal with toxicities of these therapies as well, such as the colitis and hypophysitis related to ipilimumab therapy, and the development of resistance over time, Dr. Thompson acknowledged.
Nonetheless, the multimodality approach has led to regression and elimination of brain metastases, and even allowed some patients to achieve remission, he reported.
EXPERT ANALYSIS FROM THE WORLD CUTANEOUS MALIGNANCIES CONGRESS
EADV: Prophylactic photodynamic therapy benefits transplant recipients
COPENHAGEN – Twice-yearly prophylactic photodynamic therapy for primary prevention of actinic keratoses and squamous cell carcinomas is a novel and effective strategy that addresses the problem of accelerated photocarcinogenesis in organ transplant recipients, according to an interim analysis of a multinational, randomized, controlled trial.
“The overall aim is to prevent squamous cell carcinoma development. Photodynamic therapy is well established for secondary prevention of further AKs, and these very early data show that it can also be used for primary prevention in very high-risk patients,” Dr. Katrine Togsverd-Bo said at the annual congress of the European Academy of Dermatology and Venereology.
Accelerated carcinogenesis on sun-exposed skin is a major concern in organ transplant recipients (OTRs). They experience early onset of multiple AKs, with field cancerization and up to a 100-fold increased risk of squamous cell carcinomas (SCCs). Moreover, their SCCs are at substantially greater risk of metastasis than SCCs occurring in the general population, noted Dr. Togsverd-Bo of Bispebjerg Hospital and the University of Copenhagen.
She presented an interim analysis of an ongoing 5-year prospective randomized trial in 50 renal transplant recipients at academic dermatology centers in Copenhagen, Oslo, and Gothenburg, Sweden. All participants had clinically normal-appearing skin at baseline, with no history of AKs or SCCs. They are undergoing twice-yearly, split-side photodynamic therapy (PDT) on the face, forearm, and hand, with the opposite side serving as the untreated control.
To date, 25 patients have completed 3 years of the study. At 3 years of prospective follow-up by blinded evaluators, 50% of patients had AKs on their untreated side, compared with 26% on the prophylactic PDT side. The collective number of AKs on untreated skin was 43, compared with just 8 AKs on PDT-treated skin. Seven patients had AKs only on their untreated side, six had AKs on both sides, and none had any AKs only on their PDT-treated side.
The twice-yearly prophylactic PDT regimen consists of a 3-hour application of 20% methyl aminolevulinate as a photosensitizer followed by applications of a conventional LED light at 37 J/cm2.
Dr. Togsverd-Bo reported having no financial conflicts regarding her study.
COPENHAGEN – Twice-yearly prophylactic photodynamic therapy for primary prevention of actinic keratoses and squamous cell carcinomas is a novel and effective strategy that addresses the problem of accelerated photocarcinogenesis in organ transplant recipients, according to an interim analysis of a multinational, randomized, controlled trial.
“The overall aim is to prevent squamous cell carcinoma development. Photodynamic therapy is well established for secondary prevention of further AKs, and these very early data show that it can also be used for primary prevention in very high-risk patients,” Dr. Katrine Togsverd-Bo said at the annual congress of the European Academy of Dermatology and Venereology.
Accelerated carcinogenesis on sun-exposed skin is a major concern in organ transplant recipients (OTRs). They experience early onset of multiple AKs, with field cancerization and up to a 100-fold increased risk of squamous cell carcinomas (SCCs). Moreover, their SCCs are at substantially greater risk of metastasis than SCCs occurring in the general population, noted Dr. Togsverd-Bo of Bispebjerg Hospital and the University of Copenhagen.
She presented an interim analysis of an ongoing 5-year prospective randomized trial in 50 renal transplant recipients at academic dermatology centers in Copenhagen, Oslo, and Gothenburg, Sweden. All participants had clinically normal-appearing skin at baseline, with no history of AKs or SCCs. They are undergoing twice-yearly, split-side photodynamic therapy (PDT) on the face, forearm, and hand, with the opposite side serving as the untreated control.
To date, 25 patients have completed 3 years of the study. At 3 years of prospective follow-up by blinded evaluators, 50% of patients had AKs on their untreated side, compared with 26% on the prophylactic PDT side. The collective number of AKs on untreated skin was 43, compared with just 8 AKs on PDT-treated skin. Seven patients had AKs only on their untreated side, six had AKs on both sides, and none had any AKs only on their PDT-treated side.
The twice-yearly prophylactic PDT regimen consists of a 3-hour application of 20% methyl aminolevulinate as a photosensitizer followed by applications of a conventional LED light at 37 J/cm2.
Dr. Togsverd-Bo reported having no financial conflicts regarding her study.
COPENHAGEN – Twice-yearly prophylactic photodynamic therapy for primary prevention of actinic keratoses and squamous cell carcinomas is a novel and effective strategy that addresses the problem of accelerated photocarcinogenesis in organ transplant recipients, according to an interim analysis of a multinational, randomized, controlled trial.
“The overall aim is to prevent squamous cell carcinoma development. Photodynamic therapy is well established for secondary prevention of further AKs, and these very early data show that it can also be used for primary prevention in very high-risk patients,” Dr. Katrine Togsverd-Bo said at the annual congress of the European Academy of Dermatology and Venereology.
Accelerated carcinogenesis on sun-exposed skin is a major concern in organ transplant recipients (OTRs). They experience early onset of multiple AKs, with field cancerization and up to a 100-fold increased risk of squamous cell carcinomas (SCCs). Moreover, their SCCs are at substantially greater risk of metastasis than SCCs occurring in the general population, noted Dr. Togsverd-Bo of Bispebjerg Hospital and the University of Copenhagen.
She presented an interim analysis of an ongoing 5-year prospective randomized trial in 50 renal transplant recipients at academic dermatology centers in Copenhagen, Oslo, and Gothenburg, Sweden. All participants had clinically normal-appearing skin at baseline, with no history of AKs or SCCs. They are undergoing twice-yearly, split-side photodynamic therapy (PDT) on the face, forearm, and hand, with the opposite side serving as the untreated control.
To date, 25 patients have completed 3 years of the study. At 3 years of prospective follow-up by blinded evaluators, 50% of patients had AKs on their untreated side, compared with 26% on the prophylactic PDT side. The collective number of AKs on untreated skin was 43, compared with just 8 AKs on PDT-treated skin. Seven patients had AKs only on their untreated side, six had AKs on both sides, and none had any AKs only on their PDT-treated side.
The twice-yearly prophylactic PDT regimen consists of a 3-hour application of 20% methyl aminolevulinate as a photosensitizer followed by applications of a conventional LED light at 37 J/cm2.
Dr. Togsverd-Bo reported having no financial conflicts regarding her study.
AT THE EADV CONGRESS
Key clinical point: Prophylactic photodynamic therapy is a new and effective strategy for primary prevention of actinic keratoses and squamous cell carcinomas in organ transplant recipients.
Major finding: At 3 years of follow-up, 25 renal transplant recipients collectively had 8 actinic keratoses on the side of their face, forearms, and hands treated with twice-yearly prophylactic photodynamic therapy, compared with 43 AKs on the untreated control side.
Data source: This is an interim 3-year analysis from an ongoing 5-year prospective multinational, randomized, controlled trial involving 50 renal transplant recipients.
Disclosures: The presenter reported having no financial conflicts regarding this ongoing study.
A Novel Method of Skin Closure for Aging or Fragile Skin
Patients who have been on steroids, aspirin, or anticoagulants or who are elderly may have a fragile outer skin layer that is similar to parchment paper, which may be challenging for surgeons. In these patients, the epidermal layer is thin and translucent; when a surgeon cuts through this thin layer, the tissue beneath shows minimal dermis and poor-quality fat with weakened tissue support. When undergoing excisional surgery, there is no strong tissue to help the closure sutures remain intact. Surgeons may struggle with skin tears around the sutures and dehiscence on suture removal.
This article describes a novel approach to skin closure in patients with aging or thin skin using a polyethylene film with an acrylate adhesive in the excision area to aid in maintaining skin integrity throughout the healing process following surgery.
Closure Technique
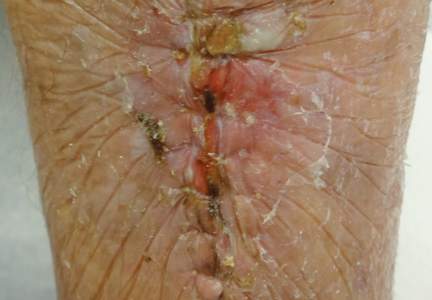
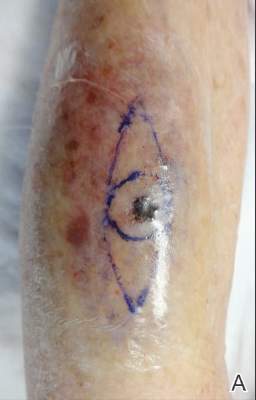
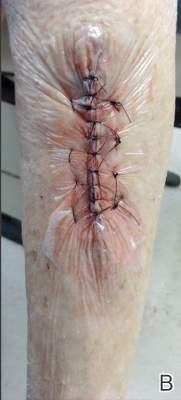
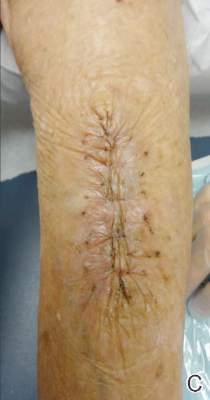
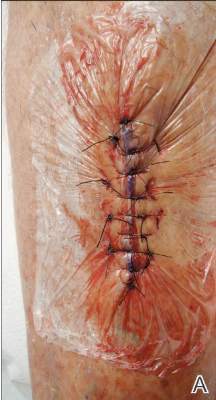
First, the skin area is cleansed with a sterilizing soap preparation. A sterile marking pen then is used to outline the excision area. A 10×12-cm layer of polyethylene film is then attached to the excision site. Excision of the tumor is performed by cutting through the film in the marked area (Figure 1A), and closure is performed by suturing the wound edges through the polyethylene film while the area is still covered with the film (Figure 1B). The sutures can be left in for 2 weeks or longer if necessary. The patient should be instructed not to remove the film or perform any extensive cleansing of the treatment area. Antibiotics should be administered, as the polyethylene film maintains its sterile integrity for 7 days only. Because sutures are on the surface of the film, they are easily accessed for removal. Figure 1C shows the excision site after removal of the sutures and polyethylene film on the left tibia of a 95-year-old woman. Adhesive butterfly closures can be applied to strengthen the excision area after suture removal and prevent dehiscence.
|
Figure 1. The excision site was marked after polyethylene adhesive film was applied to a squamous cell carcinoma on the left tibia of 95-year-old woman (A). Closure was performed by suturing the wound edges through the polyethylene film (B). The excision site appeared to have no dehiscence or signs of infection after removal of the sutures and polyethylene film (C). |
Case Reports
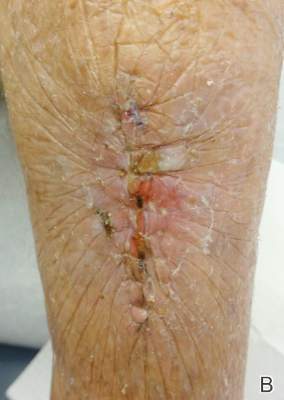
Twelve procedures for skin cancer excision were conducted in 10 patients using polyethylene adhesive film as a surgical aid due to extremely poor quality of the epidermis. The tumors were all squamous cell carcinomas and were located on the arms and legs. Patients were aged 73 to 95 years. Figure 2 demonstrates an example of excision of a squamous cell carcinoma on the left tibia of an 82-year-old man with prior dehiscence and infection after leg surgeries. Good results were achieved using the closure technique described here, along with prophylactic antibiotics.
|
Figure 2. A squamous cell carcinoma excision site on the left tibia of an 82-year-old man that had been covered with polyethylene adhesive film prior to excision (A) and 17 days following removal of the sutures and film (B). |
One patient had complications from a Staphylococcus infection because antibiotics were not administered. The patient had prior infections with other surgeries. Antibiotics were given 4 days after surgery. The infection was cleared and the polyethylene film was retained for a total of 12 days.
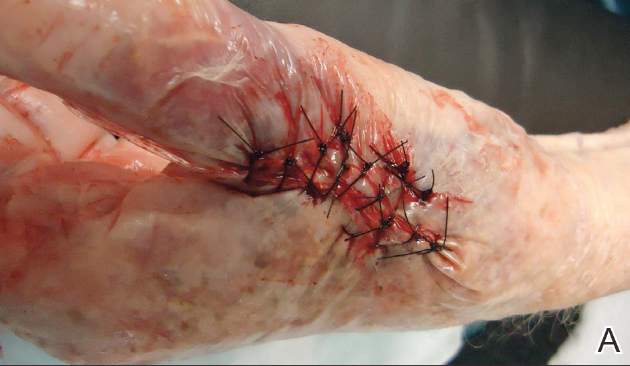
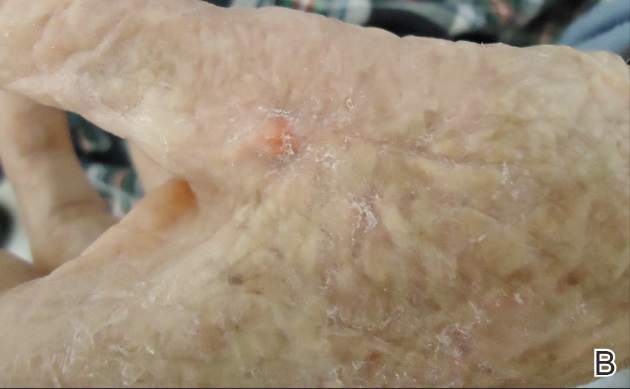
Sutures were removed after 14 days for excision sites on the arms and 17 days for excision sites on the legs. All excision sites healed without dehiscence with a cosmetically acceptable scar. Figure 3A shows a completed excision on the left hand of a 92-year-old man, and Figure 3B is the result 5 weeks after excision.
|
Figure 3. A squamous cell carcinoma excision site on the left thumb of a 92-year-old man that had been covered with polyethylene adhesive film prior to exci- sion (A). No visible scarring or dehiscence was noted 5 weeks after excision, following removal of the sutures and film (B). |
None of the patients reported discomfort from the polyethylene film remaining on the skin following surgery, though postoperative care required extra caution when dressing so as not to disturb or compromise the film. Patients were advised about postoperative care and were instructed not to remove the dressing. They were all given antibiotics as a necessary adjunct to maintain a lessened bacteria burden imposed by an impervious layer of acrylate adhesive. Complications resulted from failure to immediately provide antibiotics to 1 patient. The polyethylene film did not hinder healing or postoperative results.
Comment
Various techniques for handling fragile skin during surgery have been described in the literature. Fomon et al1 discussed aging skin as it relates to plastic surgery. Foster and Chan2 described a skin support technique for closing elliptical incisions in patients with fragile skin. Mazzurco and Krach3 discussed the use of a hydrocolloid dressing to aid in the closure of surgical wounds in patients with fragile skin.
The closure method described here was found to be particularly helpful when used as an adjunct to surgery in patients with fragile skin that lacked a suitable dermis. The polyethylene adhesive film helped to hold the sutures more securely. This method is cost-effective and is associated with a high level of patient satisfaction. For the surgeon, this technique may aid in dealing with difficult surgical situations and helps prevent wound complications in elderly patients or those with fragile skin.
1. Fomon S, Bell JW, Schattner A. Aging skin, a surgical challenge. AMA Arch Otolaryngol. 1955;61:554-562.
2. Foster RS, Chan J. The Fixomull skin support method for wound closure in patients with fragile skin. Australas J Dermatol. 2011;52:209-211.
3. Mazzurco JD, Krach KJ. Use of a hydrocolloid dressing to aid in the closure of surgical wounds in patients with fragile skin. J Am Acad Dermatol. 2012;66:335-336.
Patients who have been on steroids, aspirin, or anticoagulants or who are elderly may have a fragile outer skin layer that is similar to parchment paper, which may be challenging for surgeons. In these patients, the epidermal layer is thin and translucent; when a surgeon cuts through this thin layer, the tissue beneath shows minimal dermis and poor-quality fat with weakened tissue support. When undergoing excisional surgery, there is no strong tissue to help the closure sutures remain intact. Surgeons may struggle with skin tears around the sutures and dehiscence on suture removal.
This article describes a novel approach to skin closure in patients with aging or thin skin using a polyethylene film with an acrylate adhesive in the excision area to aid in maintaining skin integrity throughout the healing process following surgery.
Closure Technique
First, the skin area is cleansed with a sterilizing soap preparation. A sterile marking pen then is used to outline the excision area. A 10×12-cm layer of polyethylene film is then attached to the excision site. Excision of the tumor is performed by cutting through the film in the marked area (Figure 1A), and closure is performed by suturing the wound edges through the polyethylene film while the area is still covered with the film (Figure 1B). The sutures can be left in for 2 weeks or longer if necessary. The patient should be instructed not to remove the film or perform any extensive cleansing of the treatment area. Antibiotics should be administered, as the polyethylene film maintains its sterile integrity for 7 days only. Because sutures are on the surface of the film, they are easily accessed for removal. Figure 1C shows the excision site after removal of the sutures and polyethylene film on the left tibia of a 95-year-old woman. Adhesive butterfly closures can be applied to strengthen the excision area after suture removal and prevent dehiscence.



|
Figure 1. The excision site was marked after polyethylene adhesive film was applied to a squamous cell carcinoma on the left tibia of 95-year-old woman (A). Closure was performed by suturing the wound edges through the polyethylene film (B). The excision site appeared to have no dehiscence or signs of infection after removal of the sutures and polyethylene film (C). |
Case Reports
Twelve procedures for skin cancer excision were conducted in 10 patients using polyethylene adhesive film as a surgical aid due to extremely poor quality of the epidermis. The tumors were all squamous cell carcinomas and were located on the arms and legs. Patients were aged 73 to 95 years. Figure 2 demonstrates an example of excision of a squamous cell carcinoma on the left tibia of an 82-year-old man with prior dehiscence and infection after leg surgeries. Good results were achieved using the closure technique described here, along with prophylactic antibiotics.


|
Figure 2. A squamous cell carcinoma excision site on the left tibia of an 82-year-old man that had been covered with polyethylene adhesive film prior to excision (A) and 17 days following removal of the sutures and film (B). |
One patient had complications from a Staphylococcus infection because antibiotics were not administered. The patient had prior infections with other surgeries. Antibiotics were given 4 days after surgery. The infection was cleared and the polyethylene film was retained for a total of 12 days.
Sutures were removed after 14 days for excision sites on the arms and 17 days for excision sites on the legs. All excision sites healed without dehiscence with a cosmetically acceptable scar. Figure 3A shows a completed excision on the left hand of a 92-year-old man, and Figure 3B is the result 5 weeks after excision.


|
Figure 3. A squamous cell carcinoma excision site on the left thumb of a 92-year-old man that had been covered with polyethylene adhesive film prior to exci- sion (A). No visible scarring or dehiscence was noted 5 weeks after excision, following removal of the sutures and film (B). |
None of the patients reported discomfort from the polyethylene film remaining on the skin following surgery, though postoperative care required extra caution when dressing so as not to disturb or compromise the film. Patients were advised about postoperative care and were instructed not to remove the dressing. They were all given antibiotics as a necessary adjunct to maintain a lessened bacteria burden imposed by an impervious layer of acrylate adhesive. Complications resulted from failure to immediately provide antibiotics to 1 patient. The polyethylene film did not hinder healing or postoperative results.
Comment
Various techniques for handling fragile skin during surgery have been described in the literature. Fomon et al1 discussed aging skin as it relates to plastic surgery. Foster and Chan2 described a skin support technique for closing elliptical incisions in patients with fragile skin. Mazzurco and Krach3 discussed the use of a hydrocolloid dressing to aid in the closure of surgical wounds in patients with fragile skin.
The closure method described here was found to be particularly helpful when used as an adjunct to surgery in patients with fragile skin that lacked a suitable dermis. The polyethylene adhesive film helped to hold the sutures more securely. This method is cost-effective and is associated with a high level of patient satisfaction. For the surgeon, this technique may aid in dealing with difficult surgical situations and helps prevent wound complications in elderly patients or those with fragile skin.
Patients who have been on steroids, aspirin, or anticoagulants or who are elderly may have a fragile outer skin layer that is similar to parchment paper, which may be challenging for surgeons. In these patients, the epidermal layer is thin and translucent; when a surgeon cuts through this thin layer, the tissue beneath shows minimal dermis and poor-quality fat with weakened tissue support. When undergoing excisional surgery, there is no strong tissue to help the closure sutures remain intact. Surgeons may struggle with skin tears around the sutures and dehiscence on suture removal.
This article describes a novel approach to skin closure in patients with aging or thin skin using a polyethylene film with an acrylate adhesive in the excision area to aid in maintaining skin integrity throughout the healing process following surgery.
Closure Technique
First, the skin area is cleansed with a sterilizing soap preparation. A sterile marking pen then is used to outline the excision area. A 10×12-cm layer of polyethylene film is then attached to the excision site. Excision of the tumor is performed by cutting through the film in the marked area (Figure 1A), and closure is performed by suturing the wound edges through the polyethylene film while the area is still covered with the film (Figure 1B). The sutures can be left in for 2 weeks or longer if necessary. The patient should be instructed not to remove the film or perform any extensive cleansing of the treatment area. Antibiotics should be administered, as the polyethylene film maintains its sterile integrity for 7 days only. Because sutures are on the surface of the film, they are easily accessed for removal. Figure 1C shows the excision site after removal of the sutures and polyethylene film on the left tibia of a 95-year-old woman. Adhesive butterfly closures can be applied to strengthen the excision area after suture removal and prevent dehiscence.



|
Figure 1. The excision site was marked after polyethylene adhesive film was applied to a squamous cell carcinoma on the left tibia of 95-year-old woman (A). Closure was performed by suturing the wound edges through the polyethylene film (B). The excision site appeared to have no dehiscence or signs of infection after removal of the sutures and polyethylene film (C). |
Case Reports
Twelve procedures for skin cancer excision were conducted in 10 patients using polyethylene adhesive film as a surgical aid due to extremely poor quality of the epidermis. The tumors were all squamous cell carcinomas and were located on the arms and legs. Patients were aged 73 to 95 years. Figure 2 demonstrates an example of excision of a squamous cell carcinoma on the left tibia of an 82-year-old man with prior dehiscence and infection after leg surgeries. Good results were achieved using the closure technique described here, along with prophylactic antibiotics.


|
Figure 2. A squamous cell carcinoma excision site on the left tibia of an 82-year-old man that had been covered with polyethylene adhesive film prior to excision (A) and 17 days following removal of the sutures and film (B). |
One patient had complications from a Staphylococcus infection because antibiotics were not administered. The patient had prior infections with other surgeries. Antibiotics were given 4 days after surgery. The infection was cleared and the polyethylene film was retained for a total of 12 days.
Sutures were removed after 14 days for excision sites on the arms and 17 days for excision sites on the legs. All excision sites healed without dehiscence with a cosmetically acceptable scar. Figure 3A shows a completed excision on the left hand of a 92-year-old man, and Figure 3B is the result 5 weeks after excision.


|
Figure 3. A squamous cell carcinoma excision site on the left thumb of a 92-year-old man that had been covered with polyethylene adhesive film prior to exci- sion (A). No visible scarring or dehiscence was noted 5 weeks after excision, following removal of the sutures and film (B). |
None of the patients reported discomfort from the polyethylene film remaining on the skin following surgery, though postoperative care required extra caution when dressing so as not to disturb or compromise the film. Patients were advised about postoperative care and were instructed not to remove the dressing. They were all given antibiotics as a necessary adjunct to maintain a lessened bacteria burden imposed by an impervious layer of acrylate adhesive. Complications resulted from failure to immediately provide antibiotics to 1 patient. The polyethylene film did not hinder healing or postoperative results.
Comment
Various techniques for handling fragile skin during surgery have been described in the literature. Fomon et al1 discussed aging skin as it relates to plastic surgery. Foster and Chan2 described a skin support technique for closing elliptical incisions in patients with fragile skin. Mazzurco and Krach3 discussed the use of a hydrocolloid dressing to aid in the closure of surgical wounds in patients with fragile skin.
The closure method described here was found to be particularly helpful when used as an adjunct to surgery in patients with fragile skin that lacked a suitable dermis. The polyethylene adhesive film helped to hold the sutures more securely. This method is cost-effective and is associated with a high level of patient satisfaction. For the surgeon, this technique may aid in dealing with difficult surgical situations and helps prevent wound complications in elderly patients or those with fragile skin.
1. Fomon S, Bell JW, Schattner A. Aging skin, a surgical challenge. AMA Arch Otolaryngol. 1955;61:554-562.
2. Foster RS, Chan J. The Fixomull skin support method for wound closure in patients with fragile skin. Australas J Dermatol. 2011;52:209-211.
3. Mazzurco JD, Krach KJ. Use of a hydrocolloid dressing to aid in the closure of surgical wounds in patients with fragile skin. J Am Acad Dermatol. 2012;66:335-336.
1. Fomon S, Bell JW, Schattner A. Aging skin, a surgical challenge. AMA Arch Otolaryngol. 1955;61:554-562.
2. Foster RS, Chan J. The Fixomull skin support method for wound closure in patients with fragile skin. Australas J Dermatol. 2011;52:209-211.
3. Mazzurco JD, Krach KJ. Use of a hydrocolloid dressing to aid in the closure of surgical wounds in patients with fragile skin. J Am Acad Dermatol. 2012;66:335-336.
Practice Points
- A novel method of skin closure using a polyethylene film with an acrylate adhesive can aid in strengthening suture integrity and preventing skin tears.
- Dehiscence of excision sites in patients with aging or fragile skin can be prevented.
- This closure technique promotes healing and efficient scar formation.
Merkel cell carcinoma responds to immunotherapy
VIENNA – High response rates to pembrolizumab and to intratumoral delivery of plasmid interleukin-12 (IL-12) were observed among patients with advanced Merkel cell carcinoma (MCC), based on studies presented at the 2015 European Cancer Congress.
There is strong rationale for immunotherapy in MCC. For one thing, the Merkel cell polyomavirus, which is expressed in 80% of tumors, serves as a powerful antigen for stimulating an immune response. Secondly, MCC tumors often express the ligand for the programmed death protein (PD-1), creating an opportunity for PD-1 pathway blockade with PD-1 inhibitors such as pembrolizumab, according to the investigators.
Responses in 10 of 14 given pembrolizumab
Dr. Paul Nghiem of the University of Washington in Seattle, led an open-label, single-arm, multicenter phase II trial of pembrolizumab, which is approved for metastatic melanoma and which could become the first systemic therapy for unresectable or metastatic MCC (Abstract 22LBA).
In the study, which opened in January 2015, there are 24 evaluable patients treated with pembrolizumab 2 mg/kg every 3 weeks for up to 2 years. Fourteen had at least one posttreatment scan; 10 of these 14 patients (71%) have responded, 2 had complete responses. One patient had stable disease and three progressed.
“Responses were rapid, and appear more durable than we see with chemotherapy,” Dr. Nghiem said.
The drug was generally well tolerated, with an adverse event profile similar to what has been seen in melanoma. One patient experienced grade 4 myocarditis after one dose and one developed grade 4 transaminase elevation after two doses. Both improved after discontinuing pembrolizumab and starting corticosteroids.
“Strikingly, despite receiving only one or two doses, both patients had profound and ongoing responses to pembrolizumab,” Dr. Nghiem observed.
Many subjects had “profound shrinkage of tumor that has not rebounded,” he noted. “Pembrolizumab looks to be very favorable in terms of durability of response. With chemotherapy, at 90 days half of our patients have gone off treatment.”
The researchers are considering expanding the study population, and may include a chemotherapy-relapsed cohort. They are also analyzing biomarkers, and hope to evaluate response according to virus-positive versus virus-negative status.
Dr. Caroline Robert of the Institut Gustave Roussy in Villejuif, France, commented on the findings, first noting the difficulty of treating patients for whom there are no approved therapies. Median overall survival is less than 10 months, she said, and clinicians have lacked clinical trials to enroll patients.
“MCC patients represent a high medical need,” she said. Although the results with pembrolizumab are early, “they are very promising.”
Intratumoral plasmid IL-12
Dr. Shailender Bhatia of the University of Washington in Seattle, described a different immunotherapeutic approach in an MCC population (Abstract 504).
IL-12, which regulates adaptive type-1 immunity, has demonstrated antitumor efficacy in MCC but is associated with severe toxicities when administered systemically. Local IL-12 delivery to the tumor microenvironment “may provide adequate cytokine concentration in the vicinity of tumor antigens, and therefore improve efficacy while sparing systemic toxicity,” he said.
Intratumoral IL-12 plasmid DNA (pIL-12) injection with electroporation (which uses pulsed electricity to open pores in cell membranes) has shown antitumor activity in melanoma, and might also work in MCC, Dr. Bhatia and his colleagues hypothesized. The concept is one of promoting tumor inflammation and thereby a systemic antitumor immune response. This would be reflected, and measured, by increased IL-12 protein expression in the tumor microenvironment, which became this study’s primary endpoint.
“To the best of our knowledge, this study represents the first prospective clinical trial of immunotherapy in advanced MCC,” Dr. Bhatia noted.
Beginning in January 2012, the study enrolled 15 patients with MCC and superficial injectable tumors. Patients received injections of pIL-12 on days 1, 5, and 8; 2 weeks later, on day 22, their lesions were biopsied. The 3-person cohort with localized MCC (stage IIIb) then underwent definitive surgery and/or radiation therapy starting in week 4, while the 12 with distant metastatic disease (stage IV) could receive additional treatment cycles (maximum of 4) at least 6 weeks apart.
The treatment was well tolerated, with most adverse events – primarily pain and local inflammatory reaction – being mild and transient. No patients discontinued because of toxicity.
The primary endpoint, sustained local expression of IL-12 protein on day 22, was observed in 79% of patients. In paired biopsy samples, comparing baseline to day 22 levels, IL-12 protein expression increased by almost 2-fold to more than 3,000-fold.
In addition, enrichment of Merkel cell polyomavirus-specific CD8-positive T cells were found in the tumor infiltrating lymphocytes (TILs) of treated and distant tumors in some patients.
“Additionally, the pIL-12/electroporation treatment led to objective clinical responses in metastatic MCC,” Dr. Bhatia reported.
Among the three patients with locally advanced disease, one had a pathologic complete response and remains free of recurrence more than 6 months later. Another patient has been recurrence free for more than 3 years. The third patient was recurrence free for 9 months before developing progressive disease.
Among the 12 patients with metastatic disease, 3 responded to treatment and 1 achieved stable disease, while 8 (52%) progressed.
The injections led to regression not only of treated lesions, but also of clearly distinct noninjected MCC tumors. The proportion of treated lesions with major (more than 30%) regression was 44%. Among 10 patients with at least one distant lesion, 30% of noninjected distant lesions regressed.
“We believe this approach warrants further exploration in MCC, perhaps in combination with emerging systemic therapies, such as anti-PD-1/PD-L1 agents,” Dr. Bhatia said.
Dr. Nghiem and Dr. Bhatia had no relevant disclosures. In the IL-12 study, some investigators have financial relationships with OncoSec Medical, which manufactures the study drug.
VIENNA – High response rates to pembrolizumab and to intratumoral delivery of plasmid interleukin-12 (IL-12) were observed among patients with advanced Merkel cell carcinoma (MCC), based on studies presented at the 2015 European Cancer Congress.
There is strong rationale for immunotherapy in MCC. For one thing, the Merkel cell polyomavirus, which is expressed in 80% of tumors, serves as a powerful antigen for stimulating an immune response. Secondly, MCC tumors often express the ligand for the programmed death protein (PD-1), creating an opportunity for PD-1 pathway blockade with PD-1 inhibitors such as pembrolizumab, according to the investigators.
Responses in 10 of 14 given pembrolizumab
Dr. Paul Nghiem of the University of Washington in Seattle, led an open-label, single-arm, multicenter phase II trial of pembrolizumab, which is approved for metastatic melanoma and which could become the first systemic therapy for unresectable or metastatic MCC (Abstract 22LBA).
In the study, which opened in January 2015, there are 24 evaluable patients treated with pembrolizumab 2 mg/kg every 3 weeks for up to 2 years. Fourteen had at least one posttreatment scan; 10 of these 14 patients (71%) have responded, 2 had complete responses. One patient had stable disease and three progressed.
“Responses were rapid, and appear more durable than we see with chemotherapy,” Dr. Nghiem said.
The drug was generally well tolerated, with an adverse event profile similar to what has been seen in melanoma. One patient experienced grade 4 myocarditis after one dose and one developed grade 4 transaminase elevation after two doses. Both improved after discontinuing pembrolizumab and starting corticosteroids.
“Strikingly, despite receiving only one or two doses, both patients had profound and ongoing responses to pembrolizumab,” Dr. Nghiem observed.
Many subjects had “profound shrinkage of tumor that has not rebounded,” he noted. “Pembrolizumab looks to be very favorable in terms of durability of response. With chemotherapy, at 90 days half of our patients have gone off treatment.”
The researchers are considering expanding the study population, and may include a chemotherapy-relapsed cohort. They are also analyzing biomarkers, and hope to evaluate response according to virus-positive versus virus-negative status.
Dr. Caroline Robert of the Institut Gustave Roussy in Villejuif, France, commented on the findings, first noting the difficulty of treating patients for whom there are no approved therapies. Median overall survival is less than 10 months, she said, and clinicians have lacked clinical trials to enroll patients.
“MCC patients represent a high medical need,” she said. Although the results with pembrolizumab are early, “they are very promising.”
Intratumoral plasmid IL-12
Dr. Shailender Bhatia of the University of Washington in Seattle, described a different immunotherapeutic approach in an MCC population (Abstract 504).
IL-12, which regulates adaptive type-1 immunity, has demonstrated antitumor efficacy in MCC but is associated with severe toxicities when administered systemically. Local IL-12 delivery to the tumor microenvironment “may provide adequate cytokine concentration in the vicinity of tumor antigens, and therefore improve efficacy while sparing systemic toxicity,” he said.
Intratumoral IL-12 plasmid DNA (pIL-12) injection with electroporation (which uses pulsed electricity to open pores in cell membranes) has shown antitumor activity in melanoma, and might also work in MCC, Dr. Bhatia and his colleagues hypothesized. The concept is one of promoting tumor inflammation and thereby a systemic antitumor immune response. This would be reflected, and measured, by increased IL-12 protein expression in the tumor microenvironment, which became this study’s primary endpoint.
“To the best of our knowledge, this study represents the first prospective clinical trial of immunotherapy in advanced MCC,” Dr. Bhatia noted.
Beginning in January 2012, the study enrolled 15 patients with MCC and superficial injectable tumors. Patients received injections of pIL-12 on days 1, 5, and 8; 2 weeks later, on day 22, their lesions were biopsied. The 3-person cohort with localized MCC (stage IIIb) then underwent definitive surgery and/or radiation therapy starting in week 4, while the 12 with distant metastatic disease (stage IV) could receive additional treatment cycles (maximum of 4) at least 6 weeks apart.
The treatment was well tolerated, with most adverse events – primarily pain and local inflammatory reaction – being mild and transient. No patients discontinued because of toxicity.
The primary endpoint, sustained local expression of IL-12 protein on day 22, was observed in 79% of patients. In paired biopsy samples, comparing baseline to day 22 levels, IL-12 protein expression increased by almost 2-fold to more than 3,000-fold.
In addition, enrichment of Merkel cell polyomavirus-specific CD8-positive T cells were found in the tumor infiltrating lymphocytes (TILs) of treated and distant tumors in some patients.
“Additionally, the pIL-12/electroporation treatment led to objective clinical responses in metastatic MCC,” Dr. Bhatia reported.
Among the three patients with locally advanced disease, one had a pathologic complete response and remains free of recurrence more than 6 months later. Another patient has been recurrence free for more than 3 years. The third patient was recurrence free for 9 months before developing progressive disease.
Among the 12 patients with metastatic disease, 3 responded to treatment and 1 achieved stable disease, while 8 (52%) progressed.
The injections led to regression not only of treated lesions, but also of clearly distinct noninjected MCC tumors. The proportion of treated lesions with major (more than 30%) regression was 44%. Among 10 patients with at least one distant lesion, 30% of noninjected distant lesions regressed.
“We believe this approach warrants further exploration in MCC, perhaps in combination with emerging systemic therapies, such as anti-PD-1/PD-L1 agents,” Dr. Bhatia said.
Dr. Nghiem and Dr. Bhatia had no relevant disclosures. In the IL-12 study, some investigators have financial relationships with OncoSec Medical, which manufactures the study drug.
VIENNA – High response rates to pembrolizumab and to intratumoral delivery of plasmid interleukin-12 (IL-12) were observed among patients with advanced Merkel cell carcinoma (MCC), based on studies presented at the 2015 European Cancer Congress.
There is strong rationale for immunotherapy in MCC. For one thing, the Merkel cell polyomavirus, which is expressed in 80% of tumors, serves as a powerful antigen for stimulating an immune response. Secondly, MCC tumors often express the ligand for the programmed death protein (PD-1), creating an opportunity for PD-1 pathway blockade with PD-1 inhibitors such as pembrolizumab, according to the investigators.
Responses in 10 of 14 given pembrolizumab
Dr. Paul Nghiem of the University of Washington in Seattle, led an open-label, single-arm, multicenter phase II trial of pembrolizumab, which is approved for metastatic melanoma and which could become the first systemic therapy for unresectable or metastatic MCC (Abstract 22LBA).
In the study, which opened in January 2015, there are 24 evaluable patients treated with pembrolizumab 2 mg/kg every 3 weeks for up to 2 years. Fourteen had at least one posttreatment scan; 10 of these 14 patients (71%) have responded, 2 had complete responses. One patient had stable disease and three progressed.
“Responses were rapid, and appear more durable than we see with chemotherapy,” Dr. Nghiem said.
The drug was generally well tolerated, with an adverse event profile similar to what has been seen in melanoma. One patient experienced grade 4 myocarditis after one dose and one developed grade 4 transaminase elevation after two doses. Both improved after discontinuing pembrolizumab and starting corticosteroids.
“Strikingly, despite receiving only one or two doses, both patients had profound and ongoing responses to pembrolizumab,” Dr. Nghiem observed.
Many subjects had “profound shrinkage of tumor that has not rebounded,” he noted. “Pembrolizumab looks to be very favorable in terms of durability of response. With chemotherapy, at 90 days half of our patients have gone off treatment.”
The researchers are considering expanding the study population, and may include a chemotherapy-relapsed cohort. They are also analyzing biomarkers, and hope to evaluate response according to virus-positive versus virus-negative status.
Dr. Caroline Robert of the Institut Gustave Roussy in Villejuif, France, commented on the findings, first noting the difficulty of treating patients for whom there are no approved therapies. Median overall survival is less than 10 months, she said, and clinicians have lacked clinical trials to enroll patients.
“MCC patients represent a high medical need,” she said. Although the results with pembrolizumab are early, “they are very promising.”
Intratumoral plasmid IL-12
Dr. Shailender Bhatia of the University of Washington in Seattle, described a different immunotherapeutic approach in an MCC population (Abstract 504).
IL-12, which regulates adaptive type-1 immunity, has demonstrated antitumor efficacy in MCC but is associated with severe toxicities when administered systemically. Local IL-12 delivery to the tumor microenvironment “may provide adequate cytokine concentration in the vicinity of tumor antigens, and therefore improve efficacy while sparing systemic toxicity,” he said.
Intratumoral IL-12 plasmid DNA (pIL-12) injection with electroporation (which uses pulsed electricity to open pores in cell membranes) has shown antitumor activity in melanoma, and might also work in MCC, Dr. Bhatia and his colleagues hypothesized. The concept is one of promoting tumor inflammation and thereby a systemic antitumor immune response. This would be reflected, and measured, by increased IL-12 protein expression in the tumor microenvironment, which became this study’s primary endpoint.
“To the best of our knowledge, this study represents the first prospective clinical trial of immunotherapy in advanced MCC,” Dr. Bhatia noted.
Beginning in January 2012, the study enrolled 15 patients with MCC and superficial injectable tumors. Patients received injections of pIL-12 on days 1, 5, and 8; 2 weeks later, on day 22, their lesions were biopsied. The 3-person cohort with localized MCC (stage IIIb) then underwent definitive surgery and/or radiation therapy starting in week 4, while the 12 with distant metastatic disease (stage IV) could receive additional treatment cycles (maximum of 4) at least 6 weeks apart.
The treatment was well tolerated, with most adverse events – primarily pain and local inflammatory reaction – being mild and transient. No patients discontinued because of toxicity.
The primary endpoint, sustained local expression of IL-12 protein on day 22, was observed in 79% of patients. In paired biopsy samples, comparing baseline to day 22 levels, IL-12 protein expression increased by almost 2-fold to more than 3,000-fold.
In addition, enrichment of Merkel cell polyomavirus-specific CD8-positive T cells were found in the tumor infiltrating lymphocytes (TILs) of treated and distant tumors in some patients.
“Additionally, the pIL-12/electroporation treatment led to objective clinical responses in metastatic MCC,” Dr. Bhatia reported.
Among the three patients with locally advanced disease, one had a pathologic complete response and remains free of recurrence more than 6 months later. Another patient has been recurrence free for more than 3 years. The third patient was recurrence free for 9 months before developing progressive disease.
Among the 12 patients with metastatic disease, 3 responded to treatment and 1 achieved stable disease, while 8 (52%) progressed.
The injections led to regression not only of treated lesions, but also of clearly distinct noninjected MCC tumors. The proportion of treated lesions with major (more than 30%) regression was 44%. Among 10 patients with at least one distant lesion, 30% of noninjected distant lesions regressed.
“We believe this approach warrants further exploration in MCC, perhaps in combination with emerging systemic therapies, such as anti-PD-1/PD-L1 agents,” Dr. Bhatia said.
Dr. Nghiem and Dr. Bhatia had no relevant disclosures. In the IL-12 study, some investigators have financial relationships with OncoSec Medical, which manufactures the study drug.
AT THE EUROPEAN CANCER CONGRESS 2015
Key clinical point: Merkel cell carcinoma may respond well to immunotherapeutic approaches.
Major finding: Patients had robust and durable responses to the PD-1 inhibitor pembrolizumab and to intratumoral IL-12 injections.
Data source: Single-arm open label studies; pembrolizumab study included 24 patients. IL-2 study included 15 patients.
Disclosures: Dr. Nghiem reported no conflicts of interest. In the IL-12 study, some investigators have financial relationships with OncoSec Medical, which manufactures the study drug. Dr. Bhatia had no relevant disclosures.
Nab-paclitaxel marginally superior to dacarbazine for metastatic melanoma
In chemotherapy-naive patients with metastatic melanoma, progression-free survival (PFS) was significantly longer with nab-paclitaxel, compared with dacarbazine, according to results from a phase III trial.
Median PFS for the nab-paclitaxel arm vs. the dacarbazine arm was 4.8 vs. 2.5 months, respectively (hazard ratio, 0.792; 95% confidence interval, 0.631-0.992; P = .044). Overall survival was similar in the two arms: 12.6 months for nab-paclitaxel vs. 10.5 months for dacarbazine (P = .27). The nab-paclitaxel group had an increased, but not significant, overall response rate (15% vs. 11%) and a significantly improved disease control rate (P = .004). The disease control rate reflects the number of complete responses plus partial responses plus stable disease for 16 or more weeks.
Nab-paclitaxel benefited patients regardless of BRAF mutation status, noted Dr. Evan Hersh, professor of medicine at the University of Arizona, Tucson, and colleagues.
“Additionally, in a post hoc analysis of this trial, nab-paclitaxel was shown to benefit a subgroup of patients with low or absent TILs [tumor-infiltrating lymphocytes], a poor prognostic factor in melanoma,” they wrote (Ann Oncol 2015 Sep 26. doi: 10.1093/annonc/mdv324).
The most common treatment-related adverse events of grade 3 or greater were neuropathy (25% vs. 0%), neutropenia (20% vs. 10%), and leukopenia (12% vs. 7%) for the nab-paclitaxel and dacarbazine arms, respectively. The median onset of grade 3 or greater peripheral neuropathy was 101 days after treatment began.
Between April 2009 and June 2011, the multicenter, phase III, randomized, controlled trial enrolled 529 adults with stage IV malignant melanoma who had no prior cytotoxic therapy.
A majority of participants from both treatment arms underwent post-study therapy, including newer agents such as BRAF inhibitors and ipilimumab. This may account for the diminished treatment affect of nab-paclitaxel on overall survival, compared with the more significant benefit in PFS observed early in the study.
A biomarker analysis found no correlation between tumor expression of the albumin-binding protein SPARC and PFS with nab-paclitaxel treatment. SPARC was hypothesized to enrich nab-paclitaxel in the tumor microenvironment thereby increasing its efficacy.
The study by Hersh et al. was conceived prior to the success of immune checkpoint inhibitors and targeted therapies for melanoma. The treatment landscape has changed rapidly, and the current role of chemotherapy, and nab-paclitaxel specifically, is uncertain.
In the last 5 years, four classes of therapies have significantly improved overall survival over dacarbazine: ipilimumab (HR, 0.72; P less than .001), vemurafenib (HR, 0.37; P less than .001), trametinib (HR, 0.54; P = .01), and nivolumab (HR, 0.42; P less than .001).
For the large majority of patients, chemotherapy would not be considered for the first line of therapy. Patients typically considered for chemotherapy would have failed or would have been intolerant to ipilimumab and an anti-PD1 agent, as well as BRAF/MEK inhibitors if the tumors were BRAF-mutation positive. In these heavily pretreated patients, however, toxicities associated with chemotherapy may outweigh benefits.
In patients with a history of autoimmune disease, some have suggested checkpoint inhibitors may exacerbate these conditions. However, recent retrospective studies suggest that ipilimumab is safe for patients with autoimmune disorders such as inflammatory bowel disease and rheumatoid arthritis.
The current study did not include quality of life data, and given the lack of improvement in overall survival, the superiority of nab-paclitaxel over dacarbazine has not been shown definitively.
Chemotherapy may have a role in combination with targeted or immunotherapeutic agents. Checkpoint inhibitors may be more active in patients with baseline tumor-infiltrating lymphocytes (TILs) and tumor PDL1 expression. Chemotherapy may induce TILs in tumors where the cells are low or absent, and nab-paclitaxel may be a good candidate to explore such combinations.
It remains to be determined if chemotherapy offers greater benefit than the many other strategies under consideration. This brings to the forefront the urgent need for preclinical models to enable comparisons of combination strategies.
Dr. Matteo Carlino is a clinical senior lecturer at the Sydney Medical School, University of Sydney and a medical oncologist at Westmead Hospital and Blacktown Hospital, Australia. Dr. Georgina Long is an associate professor and medical oncologist at Melanoma Institute Australia, University of Sydney. These remarks were part of an editorial accompanying the report (Ann Oncol. 2015 Sep 15. doi: 10.1093/annonc/mdv361). Dr. Carlino and Dr. Long reported having no disclosures.
The study by Hersh et al. was conceived prior to the success of immune checkpoint inhibitors and targeted therapies for melanoma. The treatment landscape has changed rapidly, and the current role of chemotherapy, and nab-paclitaxel specifically, is uncertain.
In the last 5 years, four classes of therapies have significantly improved overall survival over dacarbazine: ipilimumab (HR, 0.72; P less than .001), vemurafenib (HR, 0.37; P less than .001), trametinib (HR, 0.54; P = .01), and nivolumab (HR, 0.42; P less than .001).
For the large majority of patients, chemotherapy would not be considered for the first line of therapy. Patients typically considered for chemotherapy would have failed or would have been intolerant to ipilimumab and an anti-PD1 agent, as well as BRAF/MEK inhibitors if the tumors were BRAF-mutation positive. In these heavily pretreated patients, however, toxicities associated with chemotherapy may outweigh benefits.
In patients with a history of autoimmune disease, some have suggested checkpoint inhibitors may exacerbate these conditions. However, recent retrospective studies suggest that ipilimumab is safe for patients with autoimmune disorders such as inflammatory bowel disease and rheumatoid arthritis.
The current study did not include quality of life data, and given the lack of improvement in overall survival, the superiority of nab-paclitaxel over dacarbazine has not been shown definitively.
Chemotherapy may have a role in combination with targeted or immunotherapeutic agents. Checkpoint inhibitors may be more active in patients with baseline tumor-infiltrating lymphocytes (TILs) and tumor PDL1 expression. Chemotherapy may induce TILs in tumors where the cells are low or absent, and nab-paclitaxel may be a good candidate to explore such combinations.
It remains to be determined if chemotherapy offers greater benefit than the many other strategies under consideration. This brings to the forefront the urgent need for preclinical models to enable comparisons of combination strategies.
Dr. Matteo Carlino is a clinical senior lecturer at the Sydney Medical School, University of Sydney and a medical oncologist at Westmead Hospital and Blacktown Hospital, Australia. Dr. Georgina Long is an associate professor and medical oncologist at Melanoma Institute Australia, University of Sydney. These remarks were part of an editorial accompanying the report (Ann Oncol. 2015 Sep 15. doi: 10.1093/annonc/mdv361). Dr. Carlino and Dr. Long reported having no disclosures.
The study by Hersh et al. was conceived prior to the success of immune checkpoint inhibitors and targeted therapies for melanoma. The treatment landscape has changed rapidly, and the current role of chemotherapy, and nab-paclitaxel specifically, is uncertain.
In the last 5 years, four classes of therapies have significantly improved overall survival over dacarbazine: ipilimumab (HR, 0.72; P less than .001), vemurafenib (HR, 0.37; P less than .001), trametinib (HR, 0.54; P = .01), and nivolumab (HR, 0.42; P less than .001).
For the large majority of patients, chemotherapy would not be considered for the first line of therapy. Patients typically considered for chemotherapy would have failed or would have been intolerant to ipilimumab and an anti-PD1 agent, as well as BRAF/MEK inhibitors if the tumors were BRAF-mutation positive. In these heavily pretreated patients, however, toxicities associated with chemotherapy may outweigh benefits.
In patients with a history of autoimmune disease, some have suggested checkpoint inhibitors may exacerbate these conditions. However, recent retrospective studies suggest that ipilimumab is safe for patients with autoimmune disorders such as inflammatory bowel disease and rheumatoid arthritis.
The current study did not include quality of life data, and given the lack of improvement in overall survival, the superiority of nab-paclitaxel over dacarbazine has not been shown definitively.
Chemotherapy may have a role in combination with targeted or immunotherapeutic agents. Checkpoint inhibitors may be more active in patients with baseline tumor-infiltrating lymphocytes (TILs) and tumor PDL1 expression. Chemotherapy may induce TILs in tumors where the cells are low or absent, and nab-paclitaxel may be a good candidate to explore such combinations.
It remains to be determined if chemotherapy offers greater benefit than the many other strategies under consideration. This brings to the forefront the urgent need for preclinical models to enable comparisons of combination strategies.
Dr. Matteo Carlino is a clinical senior lecturer at the Sydney Medical School, University of Sydney and a medical oncologist at Westmead Hospital and Blacktown Hospital, Australia. Dr. Georgina Long is an associate professor and medical oncologist at Melanoma Institute Australia, University of Sydney. These remarks were part of an editorial accompanying the report (Ann Oncol. 2015 Sep 15. doi: 10.1093/annonc/mdv361). Dr. Carlino and Dr. Long reported having no disclosures.
In chemotherapy-naive patients with metastatic melanoma, progression-free survival (PFS) was significantly longer with nab-paclitaxel, compared with dacarbazine, according to results from a phase III trial.
Median PFS for the nab-paclitaxel arm vs. the dacarbazine arm was 4.8 vs. 2.5 months, respectively (hazard ratio, 0.792; 95% confidence interval, 0.631-0.992; P = .044). Overall survival was similar in the two arms: 12.6 months for nab-paclitaxel vs. 10.5 months for dacarbazine (P = .27). The nab-paclitaxel group had an increased, but not significant, overall response rate (15% vs. 11%) and a significantly improved disease control rate (P = .004). The disease control rate reflects the number of complete responses plus partial responses plus stable disease for 16 or more weeks.
Nab-paclitaxel benefited patients regardless of BRAF mutation status, noted Dr. Evan Hersh, professor of medicine at the University of Arizona, Tucson, and colleagues.
“Additionally, in a post hoc analysis of this trial, nab-paclitaxel was shown to benefit a subgroup of patients with low or absent TILs [tumor-infiltrating lymphocytes], a poor prognostic factor in melanoma,” they wrote (Ann Oncol 2015 Sep 26. doi: 10.1093/annonc/mdv324).
The most common treatment-related adverse events of grade 3 or greater were neuropathy (25% vs. 0%), neutropenia (20% vs. 10%), and leukopenia (12% vs. 7%) for the nab-paclitaxel and dacarbazine arms, respectively. The median onset of grade 3 or greater peripheral neuropathy was 101 days after treatment began.
Between April 2009 and June 2011, the multicenter, phase III, randomized, controlled trial enrolled 529 adults with stage IV malignant melanoma who had no prior cytotoxic therapy.
A majority of participants from both treatment arms underwent post-study therapy, including newer agents such as BRAF inhibitors and ipilimumab. This may account for the diminished treatment affect of nab-paclitaxel on overall survival, compared with the more significant benefit in PFS observed early in the study.
A biomarker analysis found no correlation between tumor expression of the albumin-binding protein SPARC and PFS with nab-paclitaxel treatment. SPARC was hypothesized to enrich nab-paclitaxel in the tumor microenvironment thereby increasing its efficacy.
In chemotherapy-naive patients with metastatic melanoma, progression-free survival (PFS) was significantly longer with nab-paclitaxel, compared with dacarbazine, according to results from a phase III trial.
Median PFS for the nab-paclitaxel arm vs. the dacarbazine arm was 4.8 vs. 2.5 months, respectively (hazard ratio, 0.792; 95% confidence interval, 0.631-0.992; P = .044). Overall survival was similar in the two arms: 12.6 months for nab-paclitaxel vs. 10.5 months for dacarbazine (P = .27). The nab-paclitaxel group had an increased, but not significant, overall response rate (15% vs. 11%) and a significantly improved disease control rate (P = .004). The disease control rate reflects the number of complete responses plus partial responses plus stable disease for 16 or more weeks.
Nab-paclitaxel benefited patients regardless of BRAF mutation status, noted Dr. Evan Hersh, professor of medicine at the University of Arizona, Tucson, and colleagues.
“Additionally, in a post hoc analysis of this trial, nab-paclitaxel was shown to benefit a subgroup of patients with low or absent TILs [tumor-infiltrating lymphocytes], a poor prognostic factor in melanoma,” they wrote (Ann Oncol 2015 Sep 26. doi: 10.1093/annonc/mdv324).
The most common treatment-related adverse events of grade 3 or greater were neuropathy (25% vs. 0%), neutropenia (20% vs. 10%), and leukopenia (12% vs. 7%) for the nab-paclitaxel and dacarbazine arms, respectively. The median onset of grade 3 or greater peripheral neuropathy was 101 days after treatment began.
Between April 2009 and June 2011, the multicenter, phase III, randomized, controlled trial enrolled 529 adults with stage IV malignant melanoma who had no prior cytotoxic therapy.
A majority of participants from both treatment arms underwent post-study therapy, including newer agents such as BRAF inhibitors and ipilimumab. This may account for the diminished treatment affect of nab-paclitaxel on overall survival, compared with the more significant benefit in PFS observed early in the study.
A biomarker analysis found no correlation between tumor expression of the albumin-binding protein SPARC and PFS with nab-paclitaxel treatment. SPARC was hypothesized to enrich nab-paclitaxel in the tumor microenvironment thereby increasing its efficacy.
FROM ANNALS OF ONCOLOGY
Key clinical point: Nab-pacilitaxel had significantly longer PFS than dacarbazine in chemotherapy-naive patients with metastatic melanoma.
Major finding: Median PFS in the nab-paclitaxel vs. dacarbazine arms was 4.8 vs. 2.5 months, respectively (HR, 0.792; 95% CI, 0.631-0.992; P = .044).
Data source: The phase III, randomized, controlled trial enrolled 529 patients between April 2009 and June 2011.
Disclosures: Dr. Hersh reported research funding from Celgene. His coauthors reported financial ties to several industry sources.
Erythematous Scaly Patch on the Jawline
The Diagnosis: Amelanotic Melanoma In Situ
Histopathology revealed a broad asymmetric melanocytic proliferation at the dermoepidermal junction, consisting both of singly dispersed cells as well as randomly positioned nests (Figure 1). The single cells demonstrated junctional confluence and extension along adnexal structures highlighted by melan-A stain (Figure 2). The melanocytes were markedly atypical with enlarged and hyperchromatic nuclei containing multiple nucleoli. No dermal involvement was seen. There was papillary dermal fibrosis and an active host lymphocytic response. Based on these findings, a diagnosis of amelanotic melanoma in situ was made.
|
Figure 1. Histopathology revealed confluence of atypical melanocytes at the dermoepidermal junction and pagetoid scatter of melanocytes to the spinous layer (A)(H&E, original magnification ×4). Higher-power magnification highlighted the atypia of the individual melanocytes (B)(H&E, original magnification ×10). |
Subsequent scouting punch biopsies at the superior, anterior, and posterior aspects of the lesion were performed (Figure 3). All 3 revealed a similar nested and single cell proliferation at the dermoepidermal junction, confirming residual amelanotic melanoma in situ. The patient was referred to the otolaryngology department and underwent wide local excision with 5-mm margins and reconstructive repair.
Amelanotic melanoma comprises 2% to 8% of cutaneous melanomas. It is more common in fair-skinned elderly women with an average age of diagnosis of 61.8 years. Because features typically associated with melanoma such as asymmetry, border irregularity, and color variegation often are absent, amelanotic melanoma represents a notable diagnostic challenge for clinicians. Lesions can present nonspecifically as erythematous macules, papules, patches, or plaques and can have associated pruritus and scale.1,2
Clinical misdiagnoses for amelanotic melanoma include Bowen disease, basal cell carcinoma, actinic keratosis, lichenoid keratosis, intradermal nevus, dermatofibroma, inflamed seborrheic keratosis, nummular dermatitis, pyogenic granuloma, and granuloma annulare.1-6 There have been few case reports of amelanotic melanoma in situ, with most being the lentigo maligna variant that were initially clinically diagnosed as superficial basal cell carcinoma, Bowen disease, or dermatitis.7,8 In one case report, an amelanotic lentigo maligna was incidentally discovered after performing a mapping shave biopsy on what was normal-appearing skin.9
Dermoscopic evidence of vascular structures in lesions, including the presence of dotted vessels, milky red areas, and/or serpentine (linear irregular) vessels, may be the only clues to suggest amelanotic melanoma before biopsy. However, these findings are nonspecific and can be seen in other benign and malignant skin conditions.2
Complete surgical excision is the standard treatment of amelanotic melanoma in situ given its potential for invasion. However, the lack of pigment can make margins difficult to define. Because of its ability to detect disease beyond visual margins, Mohs micrographic surgery may have better cure rates than conventional excision.4 Prognosis for amelanotic melanoma is the same as other melanomas of equal thickness and location, though delay in diagnosis can adversely affect outcomes. Furthermore, amelanotic melanoma in situ can rapidly progress to invasive melanoma.3,5 Thus it is important to maintain clinical suspicion for amelanotic melanoma in fair-skinned elderly women presenting with a persistent or recurring erythematous scaly lesion on sun-exposed skin.
- Rahbari H, Nabai H, Mehregan AH, et al. Amelanotic lentigo maligna melanoma: a diagnostic conundrum— presentation of four new cases. Cancer. 1996;77:2052-2057.
- Jaimes N, Braun RP, Thomas L, et al. Clinical and dermoscopic characteristics of amelanotic melanomas that are not of the nodular subtype. J Eur Acad Dermatol Venereol. 2012;26:591-596.
- Koch SE, Lange JR. Amelanotic melanoma: the great masquerader. J Am Acad Dermatol. 2000;42:731-734.
- Conrad N, Jackson B, Goldberg L. Amelanotic lentigo maligna melanoma: a unique case presentation. Dermatol Surg. 1999;25:408-411.
- Cliff S, Otter M, Holden CA. Amelanotic lentigo maligna melanoma of the face: a case report and review of the literature. Clin Exp Dermatol. 1997;22:177-179.
- Dalton SR, Fillman EP, Altman CE, et al. Atypical junctional melanocytic proliferations in benign lichenoid keratosis. Hum Pathol. 2003;34:706-709.
- Paver K, Stewart M, Kossard S, et al. Amelanotic lentigo maligna. Australas J Dermatol. 1981;22:106-108.
- Lewis JE. Lentigo maligna presenting as an eczematous lesion. Cutis. 1987;40:357-359.
- Perera E, Mellick N, Teng P, et al. A clinically invisible melanoma. Australas J Dermatol. 2014;55:e58-e59.
The Diagnosis: Amelanotic Melanoma In Situ
Histopathology revealed a broad asymmetric melanocytic proliferation at the dermoepidermal junction, consisting both of singly dispersed cells as well as randomly positioned nests (Figure 1). The single cells demonstrated junctional confluence and extension along adnexal structures highlighted by melan-A stain (Figure 2). The melanocytes were markedly atypical with enlarged and hyperchromatic nuclei containing multiple nucleoli. No dermal involvement was seen. There was papillary dermal fibrosis and an active host lymphocytic response. Based on these findings, a diagnosis of amelanotic melanoma in situ was made.


|
Figure 1. Histopathology revealed confluence of atypical melanocytes at the dermoepidermal junction and pagetoid scatter of melanocytes to the spinous layer (A)(H&E, original magnification ×4). Higher-power magnification highlighted the atypia of the individual melanocytes (B)(H&E, original magnification ×10). |
Subsequent scouting punch biopsies at the superior, anterior, and posterior aspects of the lesion were performed (Figure 3). All 3 revealed a similar nested and single cell proliferation at the dermoepidermal junction, confirming residual amelanotic melanoma in situ. The patient was referred to the otolaryngology department and underwent wide local excision with 5-mm margins and reconstructive repair.
Amelanotic melanoma comprises 2% to 8% of cutaneous melanomas. It is more common in fair-skinned elderly women with an average age of diagnosis of 61.8 years. Because features typically associated with melanoma such as asymmetry, border irregularity, and color variegation often are absent, amelanotic melanoma represents a notable diagnostic challenge for clinicians. Lesions can present nonspecifically as erythematous macules, papules, patches, or plaques and can have associated pruritus and scale.1,2
Clinical misdiagnoses for amelanotic melanoma include Bowen disease, basal cell carcinoma, actinic keratosis, lichenoid keratosis, intradermal nevus, dermatofibroma, inflamed seborrheic keratosis, nummular dermatitis, pyogenic granuloma, and granuloma annulare.1-6 There have been few case reports of amelanotic melanoma in situ, with most being the lentigo maligna variant that were initially clinically diagnosed as superficial basal cell carcinoma, Bowen disease, or dermatitis.7,8 In one case report, an amelanotic lentigo maligna was incidentally discovered after performing a mapping shave biopsy on what was normal-appearing skin.9
Dermoscopic evidence of vascular structures in lesions, including the presence of dotted vessels, milky red areas, and/or serpentine (linear irregular) vessels, may be the only clues to suggest amelanotic melanoma before biopsy. However, these findings are nonspecific and can be seen in other benign and malignant skin conditions.2
Complete surgical excision is the standard treatment of amelanotic melanoma in situ given its potential for invasion. However, the lack of pigment can make margins difficult to define. Because of its ability to detect disease beyond visual margins, Mohs micrographic surgery may have better cure rates than conventional excision.4 Prognosis for amelanotic melanoma is the same as other melanomas of equal thickness and location, though delay in diagnosis can adversely affect outcomes. Furthermore, amelanotic melanoma in situ can rapidly progress to invasive melanoma.3,5 Thus it is important to maintain clinical suspicion for amelanotic melanoma in fair-skinned elderly women presenting with a persistent or recurring erythematous scaly lesion on sun-exposed skin.
The Diagnosis: Amelanotic Melanoma In Situ
Histopathology revealed a broad asymmetric melanocytic proliferation at the dermoepidermal junction, consisting both of singly dispersed cells as well as randomly positioned nests (Figure 1). The single cells demonstrated junctional confluence and extension along adnexal structures highlighted by melan-A stain (Figure 2). The melanocytes were markedly atypical with enlarged and hyperchromatic nuclei containing multiple nucleoli. No dermal involvement was seen. There was papillary dermal fibrosis and an active host lymphocytic response. Based on these findings, a diagnosis of amelanotic melanoma in situ was made.


|
Figure 1. Histopathology revealed confluence of atypical melanocytes at the dermoepidermal junction and pagetoid scatter of melanocytes to the spinous layer (A)(H&E, original magnification ×4). Higher-power magnification highlighted the atypia of the individual melanocytes (B)(H&E, original magnification ×10). |
Subsequent scouting punch biopsies at the superior, anterior, and posterior aspects of the lesion were performed (Figure 3). All 3 revealed a similar nested and single cell proliferation at the dermoepidermal junction, confirming residual amelanotic melanoma in situ. The patient was referred to the otolaryngology department and underwent wide local excision with 5-mm margins and reconstructive repair.
Amelanotic melanoma comprises 2% to 8% of cutaneous melanomas. It is more common in fair-skinned elderly women with an average age of diagnosis of 61.8 years. Because features typically associated with melanoma such as asymmetry, border irregularity, and color variegation often are absent, amelanotic melanoma represents a notable diagnostic challenge for clinicians. Lesions can present nonspecifically as erythematous macules, papules, patches, or plaques and can have associated pruritus and scale.1,2
Clinical misdiagnoses for amelanotic melanoma include Bowen disease, basal cell carcinoma, actinic keratosis, lichenoid keratosis, intradermal nevus, dermatofibroma, inflamed seborrheic keratosis, nummular dermatitis, pyogenic granuloma, and granuloma annulare.1-6 There have been few case reports of amelanotic melanoma in situ, with most being the lentigo maligna variant that were initially clinically diagnosed as superficial basal cell carcinoma, Bowen disease, or dermatitis.7,8 In one case report, an amelanotic lentigo maligna was incidentally discovered after performing a mapping shave biopsy on what was normal-appearing skin.9
Dermoscopic evidence of vascular structures in lesions, including the presence of dotted vessels, milky red areas, and/or serpentine (linear irregular) vessels, may be the only clues to suggest amelanotic melanoma before biopsy. However, these findings are nonspecific and can be seen in other benign and malignant skin conditions.2
Complete surgical excision is the standard treatment of amelanotic melanoma in situ given its potential for invasion. However, the lack of pigment can make margins difficult to define. Because of its ability to detect disease beyond visual margins, Mohs micrographic surgery may have better cure rates than conventional excision.4 Prognosis for amelanotic melanoma is the same as other melanomas of equal thickness and location, though delay in diagnosis can adversely affect outcomes. Furthermore, amelanotic melanoma in situ can rapidly progress to invasive melanoma.3,5 Thus it is important to maintain clinical suspicion for amelanotic melanoma in fair-skinned elderly women presenting with a persistent or recurring erythematous scaly lesion on sun-exposed skin.
- Rahbari H, Nabai H, Mehregan AH, et al. Amelanotic lentigo maligna melanoma: a diagnostic conundrum— presentation of four new cases. Cancer. 1996;77:2052-2057.
- Jaimes N, Braun RP, Thomas L, et al. Clinical and dermoscopic characteristics of amelanotic melanomas that are not of the nodular subtype. J Eur Acad Dermatol Venereol. 2012;26:591-596.
- Koch SE, Lange JR. Amelanotic melanoma: the great masquerader. J Am Acad Dermatol. 2000;42:731-734.
- Conrad N, Jackson B, Goldberg L. Amelanotic lentigo maligna melanoma: a unique case presentation. Dermatol Surg. 1999;25:408-411.
- Cliff S, Otter M, Holden CA. Amelanotic lentigo maligna melanoma of the face: a case report and review of the literature. Clin Exp Dermatol. 1997;22:177-179.
- Dalton SR, Fillman EP, Altman CE, et al. Atypical junctional melanocytic proliferations in benign lichenoid keratosis. Hum Pathol. 2003;34:706-709.
- Paver K, Stewart M, Kossard S, et al. Amelanotic lentigo maligna. Australas J Dermatol. 1981;22:106-108.
- Lewis JE. Lentigo maligna presenting as an eczematous lesion. Cutis. 1987;40:357-359.
- Perera E, Mellick N, Teng P, et al. A clinically invisible melanoma. Australas J Dermatol. 2014;55:e58-e59.
- Rahbari H, Nabai H, Mehregan AH, et al. Amelanotic lentigo maligna melanoma: a diagnostic conundrum— presentation of four new cases. Cancer. 1996;77:2052-2057.
- Jaimes N, Braun RP, Thomas L, et al. Clinical and dermoscopic characteristics of amelanotic melanomas that are not of the nodular subtype. J Eur Acad Dermatol Venereol. 2012;26:591-596.
- Koch SE, Lange JR. Amelanotic melanoma: the great masquerader. J Am Acad Dermatol. 2000;42:731-734.
- Conrad N, Jackson B, Goldberg L. Amelanotic lentigo maligna melanoma: a unique case presentation. Dermatol Surg. 1999;25:408-411.
- Cliff S, Otter M, Holden CA. Amelanotic lentigo maligna melanoma of the face: a case report and review of the literature. Clin Exp Dermatol. 1997;22:177-179.
- Dalton SR, Fillman EP, Altman CE, et al. Atypical junctional melanocytic proliferations in benign lichenoid keratosis. Hum Pathol. 2003;34:706-709.
- Paver K, Stewart M, Kossard S, et al. Amelanotic lentigo maligna. Australas J Dermatol. 1981;22:106-108.
- Lewis JE. Lentigo maligna presenting as an eczematous lesion. Cutis. 1987;40:357-359.
- Perera E, Mellick N, Teng P, et al. A clinically invisible melanoma. Australas J Dermatol. 2014;55:e58-e59.

Atypia of the individual melanocytes.
Melan-A stain highlighted the density and confluence of melanocytes within the epidermis.
Three scouting punch biopsies were performed along the periphery of the lesion.
A 70-year-old white woman with a history of basal cell carcinoma presented with a 2.7×1.9-cm ill-defined, erythematous, scaly patch along the left side of the jawline. Ten months prior to presentation, the lesion appeared as a grayish macule that was clinically diagnosed as a pigmented actinic keratosis and was treated with cryotherapy with resolution noted at 6-month follow-up. Differential diagnosis of the current lesion included actinic keratosis, lichenoid keratosis, and superficial basal cell carcinoma. A shave biopsy was performed.
Children with multiple congenital melanocytic nevi should have CNS MRI
For children born with multiple congenital melanocytic nevi, a single MRI of the central nervous system within the first year of life was an effective way to screen for future outcomes, according to Dr. Regula Waelchli of Great Ormond Street Hospital for Children, London, and her associates.
Of the 271 children screened, 21% had an abnormal CNS MRI. An abnormal MRI increased the odds of negative future outcomes significantly. The most likely outcome was requirement for neurosurgery with an odds ratio of 71, followed by seizure with an OR of 13.4, and neurodevelopmental problems with an OR of 3.
The most common abnormality was intraparenchymal melanosis, present in most of the children with abnormal MRIs. However, if the only abnormality present was intraparenchymal melanosis, as it was for more than half of the abnormal group, no routine follow-up MRI was necessary and neurosurgery wasn’t required.
In patients with normal MRIs, seizures were temporary and/or easy to control with a single medication, and neurodevelopmental issues were mild, the investigators found.
While only those patients with an abnormality other than intraparenchymal melanosis require repeat MRIs, “any change in neurological status at any age should always trigger a repeat MRI, independent of the initial MRI findings,” Dr. Waelchli and her associates concluded.
Find the full study in the British Journal of Dermatology (doi: 10.1111/bjd.13898).
For children born with multiple congenital melanocytic nevi, a single MRI of the central nervous system within the first year of life was an effective way to screen for future outcomes, according to Dr. Regula Waelchli of Great Ormond Street Hospital for Children, London, and her associates.
Of the 271 children screened, 21% had an abnormal CNS MRI. An abnormal MRI increased the odds of negative future outcomes significantly. The most likely outcome was requirement for neurosurgery with an odds ratio of 71, followed by seizure with an OR of 13.4, and neurodevelopmental problems with an OR of 3.
The most common abnormality was intraparenchymal melanosis, present in most of the children with abnormal MRIs. However, if the only abnormality present was intraparenchymal melanosis, as it was for more than half of the abnormal group, no routine follow-up MRI was necessary and neurosurgery wasn’t required.
In patients with normal MRIs, seizures were temporary and/or easy to control with a single medication, and neurodevelopmental issues were mild, the investigators found.
While only those patients with an abnormality other than intraparenchymal melanosis require repeat MRIs, “any change in neurological status at any age should always trigger a repeat MRI, independent of the initial MRI findings,” Dr. Waelchli and her associates concluded.
Find the full study in the British Journal of Dermatology (doi: 10.1111/bjd.13898).
For children born with multiple congenital melanocytic nevi, a single MRI of the central nervous system within the first year of life was an effective way to screen for future outcomes, according to Dr. Regula Waelchli of Great Ormond Street Hospital for Children, London, and her associates.
Of the 271 children screened, 21% had an abnormal CNS MRI. An abnormal MRI increased the odds of negative future outcomes significantly. The most likely outcome was requirement for neurosurgery with an odds ratio of 71, followed by seizure with an OR of 13.4, and neurodevelopmental problems with an OR of 3.
The most common abnormality was intraparenchymal melanosis, present in most of the children with abnormal MRIs. However, if the only abnormality present was intraparenchymal melanosis, as it was for more than half of the abnormal group, no routine follow-up MRI was necessary and neurosurgery wasn’t required.
In patients with normal MRIs, seizures were temporary and/or easy to control with a single medication, and neurodevelopmental issues were mild, the investigators found.
While only those patients with an abnormality other than intraparenchymal melanosis require repeat MRIs, “any change in neurological status at any age should always trigger a repeat MRI, independent of the initial MRI findings,” Dr. Waelchli and her associates concluded.
Find the full study in the British Journal of Dermatology (doi: 10.1111/bjd.13898).
Children with multiple congenital melanocytic nevi should have CNS MRI
For children born with multiple congenital melanocytic nevi, a single MRI of the central nervous system within the first year of life was an effective way to screen for future outcomes, according to Dr. Regula Waelchli of Great Ormond Street Hospital for Children, London, and her associates.
Of the 271 children screened, 21% had an abnormal CNS MRI. An abnormal MRI increased the odds of negative future outcomes significantly. The most likely outcome was requirement for neurosurgery with an odds ratio of 71, followed by seizure with an OR of 13.4, and neurodevelopmental problems with an OR of 3.
The most common abnormality was intraparenchymal melanosis, present in most of the children with abnormal MRIs. However, if the only abnormality present was intraparenchymal melanosis, as it was for more than half of the abnormal group, no routine follow-up MRI was necessary and neurosurgery wasn’t required.
In patients with normal MRIs, seizures were temporary and/or easy to control with a single medication, and neurodevelopmental issues were mild, the investigators found.
While only those patients with an abnormality other than intraparenchymal melanosis require repeat MRIs, “any change in neurological status at any age should always trigger a repeat MRI, independent of the initial MRI findings,” Dr. Waelchli and her associates concluded.
Find the full study in the British Journal of Dermatology (doi: 10.1111/bjd.13898).
For children born with multiple congenital melanocytic nevi, a single MRI of the central nervous system within the first year of life was an effective way to screen for future outcomes, according to Dr. Regula Waelchli of Great Ormond Street Hospital for Children, London, and her associates.
Of the 271 children screened, 21% had an abnormal CNS MRI. An abnormal MRI increased the odds of negative future outcomes significantly. The most likely outcome was requirement for neurosurgery with an odds ratio of 71, followed by seizure with an OR of 13.4, and neurodevelopmental problems with an OR of 3.
The most common abnormality was intraparenchymal melanosis, present in most of the children with abnormal MRIs. However, if the only abnormality present was intraparenchymal melanosis, as it was for more than half of the abnormal group, no routine follow-up MRI was necessary and neurosurgery wasn’t required.
In patients with normal MRIs, seizures were temporary and/or easy to control with a single medication, and neurodevelopmental issues were mild, the investigators found.
While only those patients with an abnormality other than intraparenchymal melanosis require repeat MRIs, “any change in neurological status at any age should always trigger a repeat MRI, independent of the initial MRI findings,” Dr. Waelchli and her associates concluded.
Find the full study in the British Journal of Dermatology (doi: 10.1111/bjd.13898).
For children born with multiple congenital melanocytic nevi, a single MRI of the central nervous system within the first year of life was an effective way to screen for future outcomes, according to Dr. Regula Waelchli of Great Ormond Street Hospital for Children, London, and her associates.
Of the 271 children screened, 21% had an abnormal CNS MRI. An abnormal MRI increased the odds of negative future outcomes significantly. The most likely outcome was requirement for neurosurgery with an odds ratio of 71, followed by seizure with an OR of 13.4, and neurodevelopmental problems with an OR of 3.
The most common abnormality was intraparenchymal melanosis, present in most of the children with abnormal MRIs. However, if the only abnormality present was intraparenchymal melanosis, as it was for more than half of the abnormal group, no routine follow-up MRI was necessary and neurosurgery wasn’t required.
In patients with normal MRIs, seizures were temporary and/or easy to control with a single medication, and neurodevelopmental issues were mild, the investigators found.
While only those patients with an abnormality other than intraparenchymal melanosis require repeat MRIs, “any change in neurological status at any age should always trigger a repeat MRI, independent of the initial MRI findings,” Dr. Waelchli and her associates concluded.
Find the full study in the British Journal of Dermatology (doi: 10.1111/bjd.13898).