User login
Investigational CDK4/6 inhibitor shows activity, less toxicity
Abemaciclib, a CDK4/6 inhibitor, showed durable clinical activity when given as continuous single-agent therapy to patients with advanced cancer, including breast cancer and non–small-cell lung cancer, according to investigators.
Neutropenia was rarely observed in patients treated with abemaciclib, the toxicity observed in some patients who receive the only Food and Drug Administration–approved CDK4/6 inhibitor, palbociclib.
“Abemaciclib is a small-molecule inhibitor of CDK4 and CDK6 that is structurally distinct from other dual inhibitors (such as palbociclib and ribociclib) and notably exhibits greater selectivity for CDK4 compared with CDK6,” wrote Dr. Amita Patnaik of South Texas Accelerated Research Therapeutics and her associates (Cancer Discov. 2016 May 23. doi: 10.1158/2159-8290.CD-16-0095).
Furthermore, preclinical models indicate that the drug can cross the blood-brain barrier, suggesting potential efficacy against primary and metastatic tumors involving the central nervous system, they said.
A total of 225 patients with various types of advanced cancers were enrolled in this multicohort phase I study (dose escalation, n = 33; single-agent abemaciclib therapy for breast cancer, n = 47; non–small-cell lung cancer, n = 68; glioblastoma, n = 17; melanoma, n = 26; colorectal cancer, n = 15; abemaciclib plus fulvestrant combination therapy for hormone receptor–positive breast cancer, n = 19). Abemaciclib was given orally to all patients.
Neither dose-limiting toxicity nor maximum tolerated dose was reached in patients treated at levels of 50 mg, 100 mg, 150 mg, or 225 mg once daily. The maximum tolerated dose was 200 mg for patients treated with abemaciclib twice daily.
In the single-agent breast cancer cohort, the disease control rate was 81% for hormone receptor–positive (HR-positive) tumors, 33% for HR-negative tumors, 100% for HR-positive HER2-positive tumors, 72% for HR-positive HER2-negative tumors, and 70% overall. The response rate was 31% for HR-positive tumors, 0% for HR-negative tumors, 36% for HR-positive HER2-positive tumors, 28% for HR-positive HER2-negative tumors, and 23% overall.
The overall response rate was 21% for breast cancer patients receiving abemaciclib plus fulvestrant.
Among the 68 patients with non–small-cell lung cancer, 2 had a partial response and 31 had stable disease. Of the 26 patients with melanoma, 1 had a partial response and 6 had stable disease. Of the 17 patients with glioblastoma, 3 had stable disease.
Overall, there were no study-related deaths. Diarrhea, nausea, and fatigue were the most common adverse events; all were reversible.
Neutropenia was observed in 39 patients (23% of 173 patients in the single-agent tumor-specific cohort) – 2 were grade 4 events. Grade 3 neutropenia occurred in 6 patients (32% of 19 patients with HR-positive breast cancer receiving combination therapy with abemaciclib plus fulvestrant).
“Previous reports have identified neutropenia as an adverse event associated with dual inhibition of CDK4 and CDK6. However, abemaciclib given as a single agent on a continuous schedule in the tumor-specific cohorts was associated with an acceptable incidence of investigator-reported grade 3 (9%, 16 of 173 patients) or grade 4 (1%, 2 of 173 patients) neutropenia,” wrote the investigators.
“In summary, the results of this clinical trial demonstrate the safety and antitumor activity of abemaciclib as a single agent and support its further development both as monotherapy and in rational combinations. Furthermore, these findings validate CDK4 and CDK6 as anticancer drug targets and translate preclinical predictions regarding therapeutic targeting of cell-cycle derangements in cancer into clinical efficacy,”they wrote.
Eli Lilly funded the study. Eight investigators reported serving in advisory roles, having ownership or stock interest in, or receiving financial compensation from multiple companies including Eli Lilly.
On Twitter @JessCraig_OP
Abemaciclib, a CDK4/6 inhibitor, showed durable clinical activity when given as continuous single-agent therapy to patients with advanced cancer, including breast cancer and non–small-cell lung cancer, according to investigators.
Neutropenia was rarely observed in patients treated with abemaciclib, the toxicity observed in some patients who receive the only Food and Drug Administration–approved CDK4/6 inhibitor, palbociclib.
“Abemaciclib is a small-molecule inhibitor of CDK4 and CDK6 that is structurally distinct from other dual inhibitors (such as palbociclib and ribociclib) and notably exhibits greater selectivity for CDK4 compared with CDK6,” wrote Dr. Amita Patnaik of South Texas Accelerated Research Therapeutics and her associates (Cancer Discov. 2016 May 23. doi: 10.1158/2159-8290.CD-16-0095).
Furthermore, preclinical models indicate that the drug can cross the blood-brain barrier, suggesting potential efficacy against primary and metastatic tumors involving the central nervous system, they said.
A total of 225 patients with various types of advanced cancers were enrolled in this multicohort phase I study (dose escalation, n = 33; single-agent abemaciclib therapy for breast cancer, n = 47; non–small-cell lung cancer, n = 68; glioblastoma, n = 17; melanoma, n = 26; colorectal cancer, n = 15; abemaciclib plus fulvestrant combination therapy for hormone receptor–positive breast cancer, n = 19). Abemaciclib was given orally to all patients.
Neither dose-limiting toxicity nor maximum tolerated dose was reached in patients treated at levels of 50 mg, 100 mg, 150 mg, or 225 mg once daily. The maximum tolerated dose was 200 mg for patients treated with abemaciclib twice daily.
In the single-agent breast cancer cohort, the disease control rate was 81% for hormone receptor–positive (HR-positive) tumors, 33% for HR-negative tumors, 100% for HR-positive HER2-positive tumors, 72% for HR-positive HER2-negative tumors, and 70% overall. The response rate was 31% for HR-positive tumors, 0% for HR-negative tumors, 36% for HR-positive HER2-positive tumors, 28% for HR-positive HER2-negative tumors, and 23% overall.
The overall response rate was 21% for breast cancer patients receiving abemaciclib plus fulvestrant.
Among the 68 patients with non–small-cell lung cancer, 2 had a partial response and 31 had stable disease. Of the 26 patients with melanoma, 1 had a partial response and 6 had stable disease. Of the 17 patients with glioblastoma, 3 had stable disease.
Overall, there were no study-related deaths. Diarrhea, nausea, and fatigue were the most common adverse events; all were reversible.
Neutropenia was observed in 39 patients (23% of 173 patients in the single-agent tumor-specific cohort) – 2 were grade 4 events. Grade 3 neutropenia occurred in 6 patients (32% of 19 patients with HR-positive breast cancer receiving combination therapy with abemaciclib plus fulvestrant).
“Previous reports have identified neutropenia as an adverse event associated with dual inhibition of CDK4 and CDK6. However, abemaciclib given as a single agent on a continuous schedule in the tumor-specific cohorts was associated with an acceptable incidence of investigator-reported grade 3 (9%, 16 of 173 patients) or grade 4 (1%, 2 of 173 patients) neutropenia,” wrote the investigators.
“In summary, the results of this clinical trial demonstrate the safety and antitumor activity of abemaciclib as a single agent and support its further development both as monotherapy and in rational combinations. Furthermore, these findings validate CDK4 and CDK6 as anticancer drug targets and translate preclinical predictions regarding therapeutic targeting of cell-cycle derangements in cancer into clinical efficacy,”they wrote.
Eli Lilly funded the study. Eight investigators reported serving in advisory roles, having ownership or stock interest in, or receiving financial compensation from multiple companies including Eli Lilly.
On Twitter @JessCraig_OP
Abemaciclib, a CDK4/6 inhibitor, showed durable clinical activity when given as continuous single-agent therapy to patients with advanced cancer, including breast cancer and non–small-cell lung cancer, according to investigators.
Neutropenia was rarely observed in patients treated with abemaciclib, the toxicity observed in some patients who receive the only Food and Drug Administration–approved CDK4/6 inhibitor, palbociclib.
“Abemaciclib is a small-molecule inhibitor of CDK4 and CDK6 that is structurally distinct from other dual inhibitors (such as palbociclib and ribociclib) and notably exhibits greater selectivity for CDK4 compared with CDK6,” wrote Dr. Amita Patnaik of South Texas Accelerated Research Therapeutics and her associates (Cancer Discov. 2016 May 23. doi: 10.1158/2159-8290.CD-16-0095).
Furthermore, preclinical models indicate that the drug can cross the blood-brain barrier, suggesting potential efficacy against primary and metastatic tumors involving the central nervous system, they said.
A total of 225 patients with various types of advanced cancers were enrolled in this multicohort phase I study (dose escalation, n = 33; single-agent abemaciclib therapy for breast cancer, n = 47; non–small-cell lung cancer, n = 68; glioblastoma, n = 17; melanoma, n = 26; colorectal cancer, n = 15; abemaciclib plus fulvestrant combination therapy for hormone receptor–positive breast cancer, n = 19). Abemaciclib was given orally to all patients.
Neither dose-limiting toxicity nor maximum tolerated dose was reached in patients treated at levels of 50 mg, 100 mg, 150 mg, or 225 mg once daily. The maximum tolerated dose was 200 mg for patients treated with abemaciclib twice daily.
In the single-agent breast cancer cohort, the disease control rate was 81% for hormone receptor–positive (HR-positive) tumors, 33% for HR-negative tumors, 100% for HR-positive HER2-positive tumors, 72% for HR-positive HER2-negative tumors, and 70% overall. The response rate was 31% for HR-positive tumors, 0% for HR-negative tumors, 36% for HR-positive HER2-positive tumors, 28% for HR-positive HER2-negative tumors, and 23% overall.
The overall response rate was 21% for breast cancer patients receiving abemaciclib plus fulvestrant.
Among the 68 patients with non–small-cell lung cancer, 2 had a partial response and 31 had stable disease. Of the 26 patients with melanoma, 1 had a partial response and 6 had stable disease. Of the 17 patients with glioblastoma, 3 had stable disease.
Overall, there were no study-related deaths. Diarrhea, nausea, and fatigue were the most common adverse events; all were reversible.
Neutropenia was observed in 39 patients (23% of 173 patients in the single-agent tumor-specific cohort) – 2 were grade 4 events. Grade 3 neutropenia occurred in 6 patients (32% of 19 patients with HR-positive breast cancer receiving combination therapy with abemaciclib plus fulvestrant).
“Previous reports have identified neutropenia as an adverse event associated with dual inhibition of CDK4 and CDK6. However, abemaciclib given as a single agent on a continuous schedule in the tumor-specific cohorts was associated with an acceptable incidence of investigator-reported grade 3 (9%, 16 of 173 patients) or grade 4 (1%, 2 of 173 patients) neutropenia,” wrote the investigators.
“In summary, the results of this clinical trial demonstrate the safety and antitumor activity of abemaciclib as a single agent and support its further development both as monotherapy and in rational combinations. Furthermore, these findings validate CDK4 and CDK6 as anticancer drug targets and translate preclinical predictions regarding therapeutic targeting of cell-cycle derangements in cancer into clinical efficacy,”they wrote.
Eli Lilly funded the study. Eight investigators reported serving in advisory roles, having ownership or stock interest in, or receiving financial compensation from multiple companies including Eli Lilly.
On Twitter @JessCraig_OP
FROM CANCER DISCOVERY
Key clinical point: A phase I trial indicates that abemaciclib is safe and shows activity in treating patients with advanced breast and other cancers.
Major finding: In the single-agent breast cancer cohort, the overall disease control rate was 70%. Incidence of neutropenia was 9% for grade 3 and 1% for grade 4.
Data source: A multicenter phase I dose-escalation and tumor-specific cohort study of 225 patients with advanced cancers.
Disclosures: Eli Lilly funded the study. Eight investigators reported serving in advisory roles, having ownership or stock interest in, or receiving financial compensation from multiple companies including Eli Lilly.
Gene expression test predicts melanoma metastasis
ORLANDO – A commercially available melanoma gene expression test had a negative predictive value of 98% for metastasis, but also correctly identified patients at very high risk of disease spread.
In a retrospective study, the DecisionDx-Melanoma test, which examines 28 risk genes, showed that patients stratified as high risk with the test were 22 times more likely to develop metastatic disease than were those stratified as low risk, Dr. Bradley Greenhaw said at the annual meeting of the American College of Mohs Surgery. The test classifies patients with stage I and stage II melanoma as having a low risk (class 1) or a high risk (class 2) of metastasis within 5 years.
“Our mean follow-up time is less than half of the 5-year risk this test is able to predict,” said Dr. Greenhaw, a Mohs surgeon in Tupelo, Miss. “So if the test is performing as it’s supposed to, there would be even more events – you would see this gap widen even more as time passes. Seeing this with our limited follow-up period is pretty significant.”
The test also added valuable prognostic information to the American Joint Committee on Cancer (AJCC) staging method, he said. “For example, based on AJCC staging alone, we would expect to see a 5-year metastatic rate of 5%-10% for stage I melanomas. We saw less than a 1% rate. That is a pretty significant negative predictive value.”
The genes in the panel were selected from those examined in eight different genetic risk studies of cutaneous melanomas. The panel divides tumors into the high- and low-risk groups. A validation study published last year concluded that the 5-year disease-free survival rates were 97% for class 1 tumors and 31% for class 2 tumors (Clin Cancer Res 2015;21[1];175-83).
In the retrospective study, Dr. Greenhaw and his associates examined the test’s accuracy in a cohort of 256 patients treated at his center. The mean follow-up time was 23 months, although some patients have completed 5 years of follow-up.
Of the tumor blocks analyzed, 214 were class 1 and 42 were class 2. He noted significant differences between the class 1 and class 2 tumors, including mean patient age (66 years vs. 74 years, respectively), mean Breslow’s depth (0.7 mm vs. 2.3 mm), mitotic rate (0.8 vs. 3.3), and ulceration (4% vs. 41%).
Over the mean 2-year follow-up time, three class 1 tumors and 10 class 2 tumors metastasized (1.4% vs. 23.8%). The test’s negative predictive value was 98%; class 2 tumors were 22 times more likely to metastasize. “About 77% of metastatic tumors were correctly identified as high risk by this test,” Dr. Greenhaw said in an interview. “For comparison, more than 80% were correctly identified in the validation study, so our study was in line with it.”
The test showed a link between metastatic risk and ulceration at presentation. Of the class 1 tumors, 4% were ulcerated, compared to 41% of class 2 tumors; class 2 tumors were 16 times more likely to be ulcerated. Among the ulcerated lesions, metastasis occurred in 11% of class 1 and 41% of class 2 tumors.
Dr. Greenhaw also compared the gene test’s characterization of tumors to the AJCC staging.
Of the 214 class 1 tumors, 94% were stage IA or IB. Among these, there was no metastasis. Of the class 2 tumors, 17% were stage IA and 26% were stage IB. Interestingly, Dr. Greenhaw said, despite the high-risk genetic signature, none of these tumors metastasized. “This could have been because although the gene expression showed them to be associated with a high risk of metastases, the tumors were completely excised prior to the metastatic event, or because our follow-up is still relatively short,” he noted.
In the class 1 group, 4% of the tumors were stage IIA and 2% were stage IIB. Of these 13 tumors, two metastasized. “This 15% metastatic rate is a lower rate than we would predict by the AJCC staging,” Dr. Greenhaw said.
In the class 2 groups, 24% were stage IIA, 26% were stage IIB, and 7% were stage IIC. Of these 24 tumors, 10 metastasized. “This is a higher rate than the AJCC staging would predict, and it occurred rather quickly, with our abbreviated follow-up period,” Dr. Greenhaw noted.
Based on these findings, Dr. Greenhaw suggested a clinical management algorithm:
• All patients with invasive melanoma should be offered the gene expression profiling.
• Those with class 1 tumors have a high chance of cure and a low risk of metastasis. They can be reasonably managed with clinical skin and nodal exams every 6 months for 2 years, and then annually.
• Those with class 2 tumors have a much higher risk of metastatic disease. They should receive clinical skin and nodal exams every 3 months for 2 years, then every 6-12 months for 5 years. After that, a yearly exam should suffice.
Tests like this are an important advance in managing melanoma, Dr. Greenhaw said. “I think this is where the future of melanoma prognosis is. The keys are found in the genes and DNA of these tumors. These tests are being used in other types of cancer and I think it’s where we need to go in our field as well.”
Dr. Greenhaw has no financial disclosures to report.
ORLANDO – A commercially available melanoma gene expression test had a negative predictive value of 98% for metastasis, but also correctly identified patients at very high risk of disease spread.
In a retrospective study, the DecisionDx-Melanoma test, which examines 28 risk genes, showed that patients stratified as high risk with the test were 22 times more likely to develop metastatic disease than were those stratified as low risk, Dr. Bradley Greenhaw said at the annual meeting of the American College of Mohs Surgery. The test classifies patients with stage I and stage II melanoma as having a low risk (class 1) or a high risk (class 2) of metastasis within 5 years.
“Our mean follow-up time is less than half of the 5-year risk this test is able to predict,” said Dr. Greenhaw, a Mohs surgeon in Tupelo, Miss. “So if the test is performing as it’s supposed to, there would be even more events – you would see this gap widen even more as time passes. Seeing this with our limited follow-up period is pretty significant.”
The test also added valuable prognostic information to the American Joint Committee on Cancer (AJCC) staging method, he said. “For example, based on AJCC staging alone, we would expect to see a 5-year metastatic rate of 5%-10% for stage I melanomas. We saw less than a 1% rate. That is a pretty significant negative predictive value.”
The genes in the panel were selected from those examined in eight different genetic risk studies of cutaneous melanomas. The panel divides tumors into the high- and low-risk groups. A validation study published last year concluded that the 5-year disease-free survival rates were 97% for class 1 tumors and 31% for class 2 tumors (Clin Cancer Res 2015;21[1];175-83).
In the retrospective study, Dr. Greenhaw and his associates examined the test’s accuracy in a cohort of 256 patients treated at his center. The mean follow-up time was 23 months, although some patients have completed 5 years of follow-up.
Of the tumor blocks analyzed, 214 were class 1 and 42 were class 2. He noted significant differences between the class 1 and class 2 tumors, including mean patient age (66 years vs. 74 years, respectively), mean Breslow’s depth (0.7 mm vs. 2.3 mm), mitotic rate (0.8 vs. 3.3), and ulceration (4% vs. 41%).
Over the mean 2-year follow-up time, three class 1 tumors and 10 class 2 tumors metastasized (1.4% vs. 23.8%). The test’s negative predictive value was 98%; class 2 tumors were 22 times more likely to metastasize. “About 77% of metastatic tumors were correctly identified as high risk by this test,” Dr. Greenhaw said in an interview. “For comparison, more than 80% were correctly identified in the validation study, so our study was in line with it.”
The test showed a link between metastatic risk and ulceration at presentation. Of the class 1 tumors, 4% were ulcerated, compared to 41% of class 2 tumors; class 2 tumors were 16 times more likely to be ulcerated. Among the ulcerated lesions, metastasis occurred in 11% of class 1 and 41% of class 2 tumors.
Dr. Greenhaw also compared the gene test’s characterization of tumors to the AJCC staging.
Of the 214 class 1 tumors, 94% were stage IA or IB. Among these, there was no metastasis. Of the class 2 tumors, 17% were stage IA and 26% were stage IB. Interestingly, Dr. Greenhaw said, despite the high-risk genetic signature, none of these tumors metastasized. “This could have been because although the gene expression showed them to be associated with a high risk of metastases, the tumors were completely excised prior to the metastatic event, or because our follow-up is still relatively short,” he noted.
In the class 1 group, 4% of the tumors were stage IIA and 2% were stage IIB. Of these 13 tumors, two metastasized. “This 15% metastatic rate is a lower rate than we would predict by the AJCC staging,” Dr. Greenhaw said.
In the class 2 groups, 24% were stage IIA, 26% were stage IIB, and 7% were stage IIC. Of these 24 tumors, 10 metastasized. “This is a higher rate than the AJCC staging would predict, and it occurred rather quickly, with our abbreviated follow-up period,” Dr. Greenhaw noted.
Based on these findings, Dr. Greenhaw suggested a clinical management algorithm:
• All patients with invasive melanoma should be offered the gene expression profiling.
• Those with class 1 tumors have a high chance of cure and a low risk of metastasis. They can be reasonably managed with clinical skin and nodal exams every 6 months for 2 years, and then annually.
• Those with class 2 tumors have a much higher risk of metastatic disease. They should receive clinical skin and nodal exams every 3 months for 2 years, then every 6-12 months for 5 years. After that, a yearly exam should suffice.
Tests like this are an important advance in managing melanoma, Dr. Greenhaw said. “I think this is where the future of melanoma prognosis is. The keys are found in the genes and DNA of these tumors. These tests are being used in other types of cancer and I think it’s where we need to go in our field as well.”
Dr. Greenhaw has no financial disclosures to report.
ORLANDO – A commercially available melanoma gene expression test had a negative predictive value of 98% for metastasis, but also correctly identified patients at very high risk of disease spread.
In a retrospective study, the DecisionDx-Melanoma test, which examines 28 risk genes, showed that patients stratified as high risk with the test were 22 times more likely to develop metastatic disease than were those stratified as low risk, Dr. Bradley Greenhaw said at the annual meeting of the American College of Mohs Surgery. The test classifies patients with stage I and stage II melanoma as having a low risk (class 1) or a high risk (class 2) of metastasis within 5 years.
“Our mean follow-up time is less than half of the 5-year risk this test is able to predict,” said Dr. Greenhaw, a Mohs surgeon in Tupelo, Miss. “So if the test is performing as it’s supposed to, there would be even more events – you would see this gap widen even more as time passes. Seeing this with our limited follow-up period is pretty significant.”
The test also added valuable prognostic information to the American Joint Committee on Cancer (AJCC) staging method, he said. “For example, based on AJCC staging alone, we would expect to see a 5-year metastatic rate of 5%-10% for stage I melanomas. We saw less than a 1% rate. That is a pretty significant negative predictive value.”
The genes in the panel were selected from those examined in eight different genetic risk studies of cutaneous melanomas. The panel divides tumors into the high- and low-risk groups. A validation study published last year concluded that the 5-year disease-free survival rates were 97% for class 1 tumors and 31% for class 2 tumors (Clin Cancer Res 2015;21[1];175-83).
In the retrospective study, Dr. Greenhaw and his associates examined the test’s accuracy in a cohort of 256 patients treated at his center. The mean follow-up time was 23 months, although some patients have completed 5 years of follow-up.
Of the tumor blocks analyzed, 214 were class 1 and 42 were class 2. He noted significant differences between the class 1 and class 2 tumors, including mean patient age (66 years vs. 74 years, respectively), mean Breslow’s depth (0.7 mm vs. 2.3 mm), mitotic rate (0.8 vs. 3.3), and ulceration (4% vs. 41%).
Over the mean 2-year follow-up time, three class 1 tumors and 10 class 2 tumors metastasized (1.4% vs. 23.8%). The test’s negative predictive value was 98%; class 2 tumors were 22 times more likely to metastasize. “About 77% of metastatic tumors were correctly identified as high risk by this test,” Dr. Greenhaw said in an interview. “For comparison, more than 80% were correctly identified in the validation study, so our study was in line with it.”
The test showed a link between metastatic risk and ulceration at presentation. Of the class 1 tumors, 4% were ulcerated, compared to 41% of class 2 tumors; class 2 tumors were 16 times more likely to be ulcerated. Among the ulcerated lesions, metastasis occurred in 11% of class 1 and 41% of class 2 tumors.
Dr. Greenhaw also compared the gene test’s characterization of tumors to the AJCC staging.
Of the 214 class 1 tumors, 94% were stage IA or IB. Among these, there was no metastasis. Of the class 2 tumors, 17% were stage IA and 26% were stage IB. Interestingly, Dr. Greenhaw said, despite the high-risk genetic signature, none of these tumors metastasized. “This could have been because although the gene expression showed them to be associated with a high risk of metastases, the tumors were completely excised prior to the metastatic event, or because our follow-up is still relatively short,” he noted.
In the class 1 group, 4% of the tumors were stage IIA and 2% were stage IIB. Of these 13 tumors, two metastasized. “This 15% metastatic rate is a lower rate than we would predict by the AJCC staging,” Dr. Greenhaw said.
In the class 2 groups, 24% were stage IIA, 26% were stage IIB, and 7% were stage IIC. Of these 24 tumors, 10 metastasized. “This is a higher rate than the AJCC staging would predict, and it occurred rather quickly, with our abbreviated follow-up period,” Dr. Greenhaw noted.
Based on these findings, Dr. Greenhaw suggested a clinical management algorithm:
• All patients with invasive melanoma should be offered the gene expression profiling.
• Those with class 1 tumors have a high chance of cure and a low risk of metastasis. They can be reasonably managed with clinical skin and nodal exams every 6 months for 2 years, and then annually.
• Those with class 2 tumors have a much higher risk of metastatic disease. They should receive clinical skin and nodal exams every 3 months for 2 years, then every 6-12 months for 5 years. After that, a yearly exam should suffice.
Tests like this are an important advance in managing melanoma, Dr. Greenhaw said. “I think this is where the future of melanoma prognosis is. The keys are found in the genes and DNA of these tumors. These tests are being used in other types of cancer and I think it’s where we need to go in our field as well.”
Dr. Greenhaw has no financial disclosures to report.
AT THE ACMS ANNUAL MEETING
Key clinical point: A commercially available gene expression test can help predict metastasis in early melanoma cases.
Major finding: The test had a 98% negative predictive value.
Data source: A retrospective study that evaluated the gene expression test in 256 patients with melanoma.
Disclosures: Dr. Greenhaw has no financial interest in the test.
TBI prior to adoptive cell transfer added harm, no benefit
The addition of total body irradiation to nonmyeloablative chemotherapy prior to adoptive cell transfer does not provide any response or survival benefits but increases the risk of developing thrombotic microangiopathy (TMA), investigators reported.
Sequential clinical studies in the Surgery Branch of the National Cancer Institute had suggested that adding total body irradiation (TBI) to the preparative regimen for adoptive cell transfer could increase the complete response rate.
To evaluate in a randomized trial whether the addition of TBI to the nonmyeloablating (NMA) chemotherapy preparative regimen did indeed increase the complete response rate, 101 patients with metastatic melanoma with at least two measurable lesions were randomly assigned to receive only NMA chemotherapy or NMA chemotherapy and TBI. Patients in both treatment groups then underwent adoptive cell transfer and received intravenous infusions of tumor-infiltrating lymphocytes and high-dose IL-2. Only patients who received both NMA chemotherapy and TBI received CD34+ hematopoietic stem cells.
Complete response (CR) rate was 24% in both experimental arms (NMA: 12 of 51 patients, 95% CI 13% to 37%; NMA and TBI: 12 of 50 patients, 95% CI 13% to 38%). Progression-free survival and overall survival were similarly identical for the two experimental groups, Dr. Stephanie Goff of the National Cancer Institute and her associates reported (J Clin Oncol. 2016 May 23. doi: 10.1200/JCO.2016.66.7220).
The NMA plus TBI group had a high incidence of late-onset TMA (27%), complications that led to one treatment-related death.
“Addition of TBI did not increase CR rates or impact overall survival, but did introduce a new toxicity in the form of TMA. The nonmyeloablative chemotherapy regimen thus seemed to provide sufficient lymphodepletion for successful adoptive transfer without the need to add TBI,” Dr. Goff and associates concluded.
The Center for Cancer Research at the National Cancer Institute, the Dr. Miriam and Sheldon G. Adelson Medical Research Foundation, and Lion Biotechnologies funded the study. Four investigators disclosed financial ties with various companies. The other investigators had no disclosures to report.
On Twitter @JessCraig_OP
The addition of total body irradiation to nonmyeloablative chemotherapy prior to adoptive cell transfer does not provide any response or survival benefits but increases the risk of developing thrombotic microangiopathy (TMA), investigators reported.
Sequential clinical studies in the Surgery Branch of the National Cancer Institute had suggested that adding total body irradiation (TBI) to the preparative regimen for adoptive cell transfer could increase the complete response rate.
To evaluate in a randomized trial whether the addition of TBI to the nonmyeloablating (NMA) chemotherapy preparative regimen did indeed increase the complete response rate, 101 patients with metastatic melanoma with at least two measurable lesions were randomly assigned to receive only NMA chemotherapy or NMA chemotherapy and TBI. Patients in both treatment groups then underwent adoptive cell transfer and received intravenous infusions of tumor-infiltrating lymphocytes and high-dose IL-2. Only patients who received both NMA chemotherapy and TBI received CD34+ hematopoietic stem cells.
Complete response (CR) rate was 24% in both experimental arms (NMA: 12 of 51 patients, 95% CI 13% to 37%; NMA and TBI: 12 of 50 patients, 95% CI 13% to 38%). Progression-free survival and overall survival were similarly identical for the two experimental groups, Dr. Stephanie Goff of the National Cancer Institute and her associates reported (J Clin Oncol. 2016 May 23. doi: 10.1200/JCO.2016.66.7220).
The NMA plus TBI group had a high incidence of late-onset TMA (27%), complications that led to one treatment-related death.
“Addition of TBI did not increase CR rates or impact overall survival, but did introduce a new toxicity in the form of TMA. The nonmyeloablative chemotherapy regimen thus seemed to provide sufficient lymphodepletion for successful adoptive transfer without the need to add TBI,” Dr. Goff and associates concluded.
The Center for Cancer Research at the National Cancer Institute, the Dr. Miriam and Sheldon G. Adelson Medical Research Foundation, and Lion Biotechnologies funded the study. Four investigators disclosed financial ties with various companies. The other investigators had no disclosures to report.
On Twitter @JessCraig_OP
The addition of total body irradiation to nonmyeloablative chemotherapy prior to adoptive cell transfer does not provide any response or survival benefits but increases the risk of developing thrombotic microangiopathy (TMA), investigators reported.
Sequential clinical studies in the Surgery Branch of the National Cancer Institute had suggested that adding total body irradiation (TBI) to the preparative regimen for adoptive cell transfer could increase the complete response rate.
To evaluate in a randomized trial whether the addition of TBI to the nonmyeloablating (NMA) chemotherapy preparative regimen did indeed increase the complete response rate, 101 patients with metastatic melanoma with at least two measurable lesions were randomly assigned to receive only NMA chemotherapy or NMA chemotherapy and TBI. Patients in both treatment groups then underwent adoptive cell transfer and received intravenous infusions of tumor-infiltrating lymphocytes and high-dose IL-2. Only patients who received both NMA chemotherapy and TBI received CD34+ hematopoietic stem cells.
Complete response (CR) rate was 24% in both experimental arms (NMA: 12 of 51 patients, 95% CI 13% to 37%; NMA and TBI: 12 of 50 patients, 95% CI 13% to 38%). Progression-free survival and overall survival were similarly identical for the two experimental groups, Dr. Stephanie Goff of the National Cancer Institute and her associates reported (J Clin Oncol. 2016 May 23. doi: 10.1200/JCO.2016.66.7220).
The NMA plus TBI group had a high incidence of late-onset TMA (27%), complications that led to one treatment-related death.
“Addition of TBI did not increase CR rates or impact overall survival, but did introduce a new toxicity in the form of TMA. The nonmyeloablative chemotherapy regimen thus seemed to provide sufficient lymphodepletion for successful adoptive transfer without the need to add TBI,” Dr. Goff and associates concluded.
The Center for Cancer Research at the National Cancer Institute, the Dr. Miriam and Sheldon G. Adelson Medical Research Foundation, and Lion Biotechnologies funded the study. Four investigators disclosed financial ties with various companies. The other investigators had no disclosures to report.
On Twitter @JessCraig_OP
FROM JOURNAL OF CLINICAL ONCOLOGY
Key clinical point: No benefit but potential harm is observed when TBI is added to NMA chemotherapy prior to adoptive cell transfer.
Major finding: Complete response (CR) rate was 24% in both experimental arms (NMA: 12 of 51 patients, 95% CI 13% to 37%; NMA and TBI: 12 of 50 patients, 95% CI 13% to 38%). Progression-free survival and overall survival were also similar for the two experimental groups. A total of 27% of patients in the NMA plus TBI group experienced thrombotic microangiopathy.
Data source: Randomized clinical trial of 101 patients with metastatic melanoma.
Disclosures: The Center for Cancer Research at the National Cancer Institute, the Dr. Miriam and Sheldon G. Adelson Medical Research Foundation, and Lion Biotechnologies funded the study. Four investigators disclosed financial ties with various companies. The other investigators had no disclosures to report.
Sentinel node biopsies may be useful in head and neck squamous cell carcinoma
ORLANDO – Sentinel node biopsies may be a useful staging tool for patients with cutaneous squamous cell carcinomas of the head and neck.
These patients – especially those with compromised immune systems – appear to be at sufficiently high risk of metastasis to justify the procedure, Dr. Jonathan Lopez said at the annual meeting of the American College of Mohs Surgery.
“We found that sentinel lymph node biopsy in our clinic had a 91% negative predictive value for local recurrence, nodal recurrence, and disease-specific death. It provides valuable prognostic information for patients at increased risk of nodal metastasis,” said Dr. Lopez, a dermatology resident at the Mayo Clinic, Rochester, Minn.
He and his associates conducted a chart review of 24 patients treated at the Mayo Clinic from 2000 to 2014 for a cutaneous squamous cell carcinoma (SCC) of the head or neck. Of these, 11 patients were immunosuppressed. Five had undergone a kidney transplant and three a lung transplant. One patient had Hodgkin’s lymphoma, one had cutaneous lymphocytic leukemia, and one, metastatic urothelial carcinoma.
Before sentinel node biopsy, eight patients had a wide local excision; 12 were treated with Mohs micrographic surgery only; and four had a Mohs procedure followed by resection for better margins.
The biopsies identified two patients with nodal disease, but failed to identify a third who had it, Dr. Lopez said.
Patient No. 1 had a primary SCC on the nasal tip that was stage 2, according to the American Joint Committee on Cancer (AJCC) staging system, and 2b according to the Brigham and Women’s Hospital (BWH) system. He had undergone a prior double lung transplant and his lymph node dissection showed no nodal metastasis. He declined radiotherapy and died within 2 months of the biopsy, of unclear causes that were not related to his skin cancer.
Patient No. 2 had a primary lesion on the right cheek, and a history of kidney transplant. His cancer was stage 2 by the AJCC system and 2b by the BWH system. His lymph node dissection of the right parotid and neck was negative. At last follow-up of 3.5 years, he was cancer free. However, Dr. Lopez noted, the patient died at 4 years’ follow-up of unknown causes.
The final patient had a primary lesion on the right conchal bowl. It was a stage 2 cancer by the AJCC system and 2a by the BWH system. His sentinel node biopsy was negative. However, the otolaryngologist who performed the biopsy also took seven superficial parotid nodes and one of those was positive. This patient had no recurrence at the last visit, 1.5 years after the biopsy.
The sentinel node biopsies were negative in the 21 other patients. Of these, 14 had no evidence of recurrence at a mean of 3 years’ follow-up after the sentinel lymph node biopsy. Two developed local recurrence and two others, both of whom had a history of multiple squamous cell carcinomas, developed nodal spread and died of metastatic disease. Three have died of causes unrelated to their cancer.
Dr. Lopez had no financial disclosures.
ORLANDO – Sentinel node biopsies may be a useful staging tool for patients with cutaneous squamous cell carcinomas of the head and neck.
These patients – especially those with compromised immune systems – appear to be at sufficiently high risk of metastasis to justify the procedure, Dr. Jonathan Lopez said at the annual meeting of the American College of Mohs Surgery.
“We found that sentinel lymph node biopsy in our clinic had a 91% negative predictive value for local recurrence, nodal recurrence, and disease-specific death. It provides valuable prognostic information for patients at increased risk of nodal metastasis,” said Dr. Lopez, a dermatology resident at the Mayo Clinic, Rochester, Minn.
He and his associates conducted a chart review of 24 patients treated at the Mayo Clinic from 2000 to 2014 for a cutaneous squamous cell carcinoma (SCC) of the head or neck. Of these, 11 patients were immunosuppressed. Five had undergone a kidney transplant and three a lung transplant. One patient had Hodgkin’s lymphoma, one had cutaneous lymphocytic leukemia, and one, metastatic urothelial carcinoma.
Before sentinel node biopsy, eight patients had a wide local excision; 12 were treated with Mohs micrographic surgery only; and four had a Mohs procedure followed by resection for better margins.
The biopsies identified two patients with nodal disease, but failed to identify a third who had it, Dr. Lopez said.
Patient No. 1 had a primary SCC on the nasal tip that was stage 2, according to the American Joint Committee on Cancer (AJCC) staging system, and 2b according to the Brigham and Women’s Hospital (BWH) system. He had undergone a prior double lung transplant and his lymph node dissection showed no nodal metastasis. He declined radiotherapy and died within 2 months of the biopsy, of unclear causes that were not related to his skin cancer.
Patient No. 2 had a primary lesion on the right cheek, and a history of kidney transplant. His cancer was stage 2 by the AJCC system and 2b by the BWH system. His lymph node dissection of the right parotid and neck was negative. At last follow-up of 3.5 years, he was cancer free. However, Dr. Lopez noted, the patient died at 4 years’ follow-up of unknown causes.
The final patient had a primary lesion on the right conchal bowl. It was a stage 2 cancer by the AJCC system and 2a by the BWH system. His sentinel node biopsy was negative. However, the otolaryngologist who performed the biopsy also took seven superficial parotid nodes and one of those was positive. This patient had no recurrence at the last visit, 1.5 years after the biopsy.
The sentinel node biopsies were negative in the 21 other patients. Of these, 14 had no evidence of recurrence at a mean of 3 years’ follow-up after the sentinel lymph node biopsy. Two developed local recurrence and two others, both of whom had a history of multiple squamous cell carcinomas, developed nodal spread and died of metastatic disease. Three have died of causes unrelated to their cancer.
Dr. Lopez had no financial disclosures.
ORLANDO – Sentinel node biopsies may be a useful staging tool for patients with cutaneous squamous cell carcinomas of the head and neck.
These patients – especially those with compromised immune systems – appear to be at sufficiently high risk of metastasis to justify the procedure, Dr. Jonathan Lopez said at the annual meeting of the American College of Mohs Surgery.
“We found that sentinel lymph node biopsy in our clinic had a 91% negative predictive value for local recurrence, nodal recurrence, and disease-specific death. It provides valuable prognostic information for patients at increased risk of nodal metastasis,” said Dr. Lopez, a dermatology resident at the Mayo Clinic, Rochester, Minn.
He and his associates conducted a chart review of 24 patients treated at the Mayo Clinic from 2000 to 2014 for a cutaneous squamous cell carcinoma (SCC) of the head or neck. Of these, 11 patients were immunosuppressed. Five had undergone a kidney transplant and three a lung transplant. One patient had Hodgkin’s lymphoma, one had cutaneous lymphocytic leukemia, and one, metastatic urothelial carcinoma.
Before sentinel node biopsy, eight patients had a wide local excision; 12 were treated with Mohs micrographic surgery only; and four had a Mohs procedure followed by resection for better margins.
The biopsies identified two patients with nodal disease, but failed to identify a third who had it, Dr. Lopez said.
Patient No. 1 had a primary SCC on the nasal tip that was stage 2, according to the American Joint Committee on Cancer (AJCC) staging system, and 2b according to the Brigham and Women’s Hospital (BWH) system. He had undergone a prior double lung transplant and his lymph node dissection showed no nodal metastasis. He declined radiotherapy and died within 2 months of the biopsy, of unclear causes that were not related to his skin cancer.
Patient No. 2 had a primary lesion on the right cheek, and a history of kidney transplant. His cancer was stage 2 by the AJCC system and 2b by the BWH system. His lymph node dissection of the right parotid and neck was negative. At last follow-up of 3.5 years, he was cancer free. However, Dr. Lopez noted, the patient died at 4 years’ follow-up of unknown causes.
The final patient had a primary lesion on the right conchal bowl. It was a stage 2 cancer by the AJCC system and 2a by the BWH system. His sentinel node biopsy was negative. However, the otolaryngologist who performed the biopsy also took seven superficial parotid nodes and one of those was positive. This patient had no recurrence at the last visit, 1.5 years after the biopsy.
The sentinel node biopsies were negative in the 21 other patients. Of these, 14 had no evidence of recurrence at a mean of 3 years’ follow-up after the sentinel lymph node biopsy. Two developed local recurrence and two others, both of whom had a history of multiple squamous cell carcinomas, developed nodal spread and died of metastatic disease. Three have died of causes unrelated to their cancer.
Dr. Lopez had no financial disclosures.
AT THE ACMS ANNUAL MEETING
Key clinical point: Sentinel node biopsies identified nodal spread in some patients with cutaneous SCC of the head and neck
Major finding: The procedure had a 91% negative predictive value for nodal spread and disease-specific death.
Data source: The retrospective chart review comprised of 24 patients, treated at the Mayo Clinic for cutaneous SCC of the head and neck from 2000 to 2014.
Disclosures: Dr. Lopez had no financial disclosures.
Matrilin-2 protein distinguished BCCs from benign tumors in study
ORLANDO – Matrilin-2 – a matrix protein found in peritumoral stroma – reliably distinguished invasive basal cell carcinoma from the often difficult-to-distinguish basaloid follicular hamartoma (BFH), in a study that evaluated the protein as a marker in this setting.
The protein marked 41 of 42 cancers and none of the hamartomas, Dr. Renato Goreshi reported at the annual meeting of the American College of Mohs Surgery. The one cancer it failed to identify was a superficial basal cell tumor – a finding that makes sense, since dermal fibroblasts appear to secrete matrilin-2 as a response to invasive skin tumors, said Dr. Goreshi of the Roger Williams Cancer Center, Providence, R.I.
Mohs surgery typically employs hematoxylin and eosin staining to delineate tumor boundary. But, Dr. Goreshi said, that stain doesn’t always reliably differentiate adnexal tumors from basal cell carcinomas. “Basaloid follicular hamartoma can be particularly difficult to distinguish from basal cell carcinoma,” he said.
BFH typically presents as individual or linearly arranged, small skin-colored to brown papules or plaques, or as multiple lesions in a generalized distribution on the face, scalp, and occasionally, the trunk (Arch Pathol Lab Med. 2010 Aug;134[8]:1215-9). These are often stable for many years. The differential diagnosis includes basal cell carcinoma and trichoepithelioma.
BFH sometimes occurs near a BCC, although there are no data on how often this happens.
Dr. Goreshi cited a 2007 case report of a young woman that illustrates this problem. The patient presented with a basal cell carcinoma on the side of her nose. The adjacent BFH was unrecognized, however. She underwent a multiple-stage Mohs that was unnecessarily extended because tumor margins included sections of the BFH.
“The lesion was interpreted as malignancy by both the Mohs surgeon and the dermatopathologist, but was later determined to have been a hamartoma. This highlights the importance of finding an effective marker,” Dr. Goreshi said.
He and his fellowship director, Dr. Satori Iwamoto, chief of Mohs micrographic surgery at Roger Williams, looked for a reliable way to differentiate these tumors, capitalizing on the invasive nature of BCC. The peritumoral stroma plays a role in tumor growth and invasion. It involves fibroblasts, inflammatory and endothelial cells, and extracellular matrix proteins. Matrilin-2, which is involved in the formation of filamentous networks, was a promising candidate and the initial investigations looked good, said Dr. Goreshi said.
Their confirmatory study comprised 42 BCC and seven BFH sections that were obtained during Mohs surgery. All were stained for matrilin-2 and scored for location and intensity of staining by two reviewers. The investigators also conducted flow cytometry to determine the source of the protein.
The BCC set consisted of 11 morpheaform/infiltrative BCCs, 25 nodular BCCs, and 6 superficial BCCs. With the exception of one superficial lesion, all of these stained positive for matrilin-2 in the peritumoral stroma. None of the BFH sections stained positive for the protein, however. Flow cytometry determined that the protein was coming from dermal fibroblasts in the stroma.
This is actually a key point, Dr. Goreshi noted. “Matrilin-2 is not acting as a conventional tumor marker would, but as a marker of invasion.”
This was again played out in the variation of staining intensity in the tumor subtypes. It was most intense around the infiltrative subtypes. There was also adnexal staining, but it was significantly less than what was seen in the peritumoral stroma. There was virtually no staining in or around the hamartoma.
Staining was not as intense around the superficial BCC subtypes. In fact, it was not significantly different from what was seen in the adnexal structures. Again, however, there was no staining in or around the hamartoma.
“Now we are looking at the staining patterns of other lesions, including melanoma and squamous cell carcinoma, and trying to figure out why the dermal fibroblasts are secreting matrilin-2,” Dr. Goreshi said.
The study was the winner of the 2016 Theodore Tromovitch Award, presented for original research conducted by a fellow-in-training during his or her year of training.
Neither Dr. Goreshi nor Dr. Iwamoto had any relevant financial disclosures.
ORLANDO – Matrilin-2 – a matrix protein found in peritumoral stroma – reliably distinguished invasive basal cell carcinoma from the often difficult-to-distinguish basaloid follicular hamartoma (BFH), in a study that evaluated the protein as a marker in this setting.
The protein marked 41 of 42 cancers and none of the hamartomas, Dr. Renato Goreshi reported at the annual meeting of the American College of Mohs Surgery. The one cancer it failed to identify was a superficial basal cell tumor – a finding that makes sense, since dermal fibroblasts appear to secrete matrilin-2 as a response to invasive skin tumors, said Dr. Goreshi of the Roger Williams Cancer Center, Providence, R.I.
Mohs surgery typically employs hematoxylin and eosin staining to delineate tumor boundary. But, Dr. Goreshi said, that stain doesn’t always reliably differentiate adnexal tumors from basal cell carcinomas. “Basaloid follicular hamartoma can be particularly difficult to distinguish from basal cell carcinoma,” he said.
BFH typically presents as individual or linearly arranged, small skin-colored to brown papules or plaques, or as multiple lesions in a generalized distribution on the face, scalp, and occasionally, the trunk (Arch Pathol Lab Med. 2010 Aug;134[8]:1215-9). These are often stable for many years. The differential diagnosis includes basal cell carcinoma and trichoepithelioma.
BFH sometimes occurs near a BCC, although there are no data on how often this happens.
Dr. Goreshi cited a 2007 case report of a young woman that illustrates this problem. The patient presented with a basal cell carcinoma on the side of her nose. The adjacent BFH was unrecognized, however. She underwent a multiple-stage Mohs that was unnecessarily extended because tumor margins included sections of the BFH.
“The lesion was interpreted as malignancy by both the Mohs surgeon and the dermatopathologist, but was later determined to have been a hamartoma. This highlights the importance of finding an effective marker,” Dr. Goreshi said.
He and his fellowship director, Dr. Satori Iwamoto, chief of Mohs micrographic surgery at Roger Williams, looked for a reliable way to differentiate these tumors, capitalizing on the invasive nature of BCC. The peritumoral stroma plays a role in tumor growth and invasion. It involves fibroblasts, inflammatory and endothelial cells, and extracellular matrix proteins. Matrilin-2, which is involved in the formation of filamentous networks, was a promising candidate and the initial investigations looked good, said Dr. Goreshi said.
Their confirmatory study comprised 42 BCC and seven BFH sections that were obtained during Mohs surgery. All were stained for matrilin-2 and scored for location and intensity of staining by two reviewers. The investigators also conducted flow cytometry to determine the source of the protein.
The BCC set consisted of 11 morpheaform/infiltrative BCCs, 25 nodular BCCs, and 6 superficial BCCs. With the exception of one superficial lesion, all of these stained positive for matrilin-2 in the peritumoral stroma. None of the BFH sections stained positive for the protein, however. Flow cytometry determined that the protein was coming from dermal fibroblasts in the stroma.
This is actually a key point, Dr. Goreshi noted. “Matrilin-2 is not acting as a conventional tumor marker would, but as a marker of invasion.”
This was again played out in the variation of staining intensity in the tumor subtypes. It was most intense around the infiltrative subtypes. There was also adnexal staining, but it was significantly less than what was seen in the peritumoral stroma. There was virtually no staining in or around the hamartoma.
Staining was not as intense around the superficial BCC subtypes. In fact, it was not significantly different from what was seen in the adnexal structures. Again, however, there was no staining in or around the hamartoma.
“Now we are looking at the staining patterns of other lesions, including melanoma and squamous cell carcinoma, and trying to figure out why the dermal fibroblasts are secreting matrilin-2,” Dr. Goreshi said.
The study was the winner of the 2016 Theodore Tromovitch Award, presented for original research conducted by a fellow-in-training during his or her year of training.
Neither Dr. Goreshi nor Dr. Iwamoto had any relevant financial disclosures.
ORLANDO – Matrilin-2 – a matrix protein found in peritumoral stroma – reliably distinguished invasive basal cell carcinoma from the often difficult-to-distinguish basaloid follicular hamartoma (BFH), in a study that evaluated the protein as a marker in this setting.
The protein marked 41 of 42 cancers and none of the hamartomas, Dr. Renato Goreshi reported at the annual meeting of the American College of Mohs Surgery. The one cancer it failed to identify was a superficial basal cell tumor – a finding that makes sense, since dermal fibroblasts appear to secrete matrilin-2 as a response to invasive skin tumors, said Dr. Goreshi of the Roger Williams Cancer Center, Providence, R.I.
Mohs surgery typically employs hematoxylin and eosin staining to delineate tumor boundary. But, Dr. Goreshi said, that stain doesn’t always reliably differentiate adnexal tumors from basal cell carcinomas. “Basaloid follicular hamartoma can be particularly difficult to distinguish from basal cell carcinoma,” he said.
BFH typically presents as individual or linearly arranged, small skin-colored to brown papules or plaques, or as multiple lesions in a generalized distribution on the face, scalp, and occasionally, the trunk (Arch Pathol Lab Med. 2010 Aug;134[8]:1215-9). These are often stable for many years. The differential diagnosis includes basal cell carcinoma and trichoepithelioma.
BFH sometimes occurs near a BCC, although there are no data on how often this happens.
Dr. Goreshi cited a 2007 case report of a young woman that illustrates this problem. The patient presented with a basal cell carcinoma on the side of her nose. The adjacent BFH was unrecognized, however. She underwent a multiple-stage Mohs that was unnecessarily extended because tumor margins included sections of the BFH.
“The lesion was interpreted as malignancy by both the Mohs surgeon and the dermatopathologist, but was later determined to have been a hamartoma. This highlights the importance of finding an effective marker,” Dr. Goreshi said.
He and his fellowship director, Dr. Satori Iwamoto, chief of Mohs micrographic surgery at Roger Williams, looked for a reliable way to differentiate these tumors, capitalizing on the invasive nature of BCC. The peritumoral stroma plays a role in tumor growth and invasion. It involves fibroblasts, inflammatory and endothelial cells, and extracellular matrix proteins. Matrilin-2, which is involved in the formation of filamentous networks, was a promising candidate and the initial investigations looked good, said Dr. Goreshi said.
Their confirmatory study comprised 42 BCC and seven BFH sections that were obtained during Mohs surgery. All were stained for matrilin-2 and scored for location and intensity of staining by two reviewers. The investigators also conducted flow cytometry to determine the source of the protein.
The BCC set consisted of 11 morpheaform/infiltrative BCCs, 25 nodular BCCs, and 6 superficial BCCs. With the exception of one superficial lesion, all of these stained positive for matrilin-2 in the peritumoral stroma. None of the BFH sections stained positive for the protein, however. Flow cytometry determined that the protein was coming from dermal fibroblasts in the stroma.
This is actually a key point, Dr. Goreshi noted. “Matrilin-2 is not acting as a conventional tumor marker would, but as a marker of invasion.”
This was again played out in the variation of staining intensity in the tumor subtypes. It was most intense around the infiltrative subtypes. There was also adnexal staining, but it was significantly less than what was seen in the peritumoral stroma. There was virtually no staining in or around the hamartoma.
Staining was not as intense around the superficial BCC subtypes. In fact, it was not significantly different from what was seen in the adnexal structures. Again, however, there was no staining in or around the hamartoma.
“Now we are looking at the staining patterns of other lesions, including melanoma and squamous cell carcinoma, and trying to figure out why the dermal fibroblasts are secreting matrilin-2,” Dr. Goreshi said.
The study was the winner of the 2016 Theodore Tromovitch Award, presented for original research conducted by a fellow-in-training during his or her year of training.
Neither Dr. Goreshi nor Dr. Iwamoto had any relevant financial disclosures.
AT THE ACMS ANNUAL MEETING
Key clinical point: Matrilin-2 is the first marker of tumor invasion to be used in skin cancers.
Major finding: The protein bound to 41 of 42 BCCs, and to none of the hamartoma lesions studied, reliably distinguishing the two.
Data source: 42 frozen section BCCs and seven basaloid follicular hamartomas.
Disclosures: Neither Dr. Goreshi nor Dr. Iwamoto had any relevant financial disclosures.
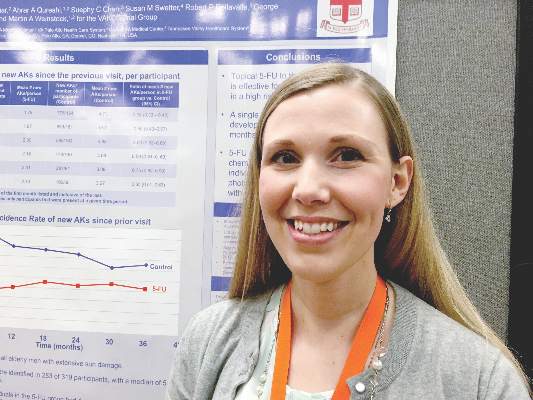
For preventing AKs, 5-FU beats placebo for up to 3 years
SCOTTSDALE – A single course of topical 5-fluorouracil (5-FU) prevented 62% more actinic keratoses than placebo, and this chemopreventive effect persisted for up to 3 years, according to an analysis of the Veterans Affairs Keratinocyte Carcinoma Chemoprevention Trial (VAKCCT) trial.
Other studies have shown that 5-FU effectively treats precancerous AKs, but have not examined whether 5-FU can prevent AKs, Dr. Joanna Walker said in an interview at the annual meeting of the Society for Investigative Dermatology.
Clinicians should consider preventive 5-FU in patients who are at high risk for basal cell and squamous cell carcinomas, especially if a skin check reveals multiple AKs, said Dr. Walker of the department of dermatology, Brown University, Providence, RI.
The VAKCCT was a randomized, double-blind, placebo-controlled study conducted at 12 Veterans Affairs dermatology clinics. The 319 patients in the analysis were nearly all elderly men with extensive sun damage, with a total of 2,386 AKs at baseline, for an average of five lesions per patient. Patients also had a history of at least two keratinocyte carcinomas in the past 5 years, including at least one lesion on the face or ears, and no recent history of 5-FU exposure.
The clinically and demographically similar study arms were randomized to either 5% topical 5-FU cream or a vehicle control cream, applied twice daily for 2-4 weeks. Both groups received cryotherapy for existing AKs, and were given free SPF 30 sunscreen. At each 6-month follow-up visit, the researchers counted existing AKs and new lesions.
At month 6, the treatment group had 62% fewer new AKs than the placebo group (average per patient, 1.78 and 4.73, respectively), a statistically significant difference. At months 12, 18, 24, 30, and 36, respectively, the treatment group had 50%, 40%, 41%, 25%, and 35% fewer new AKs than the placebo group, and these differences all were statistically significant. Furthermore, at month 6, only 56% of treated patients had at least one new AK, compared with 78% of the control group (incidence rate ratio, 0.72; 95% confidence interval, 0.54-0.95).
This chemopreventive effect remained significant for 24 months, the investigators reported. “Individuals with at least five AKs at the time of 5-FU treatment had an even more dramatic reduction in new AKs,” Dr. Walker noted. “There is now high-quality evidence supporting the use of topical 5-FU for AK chemoprevention. I think this is important information that we can take back to the clinic when we are trying to convince our patients to go through a course of 5-FU.”
The rate of new AKs in the placebo group fell during the first 2.5 years of the study and then stabilized. “For both groups, there was a dramatic increase in the use of sunscreen during the trial, and we hypothesized that the decrease in AKs in the control group was due to increased use of sun-protective measures,” Dr. Walker said.
The research was funded by the Cooperative Studies Program of the U.S. Department of Veterans Affairs. Dr. Walker had no disclosures.
SCOTTSDALE – A single course of topical 5-fluorouracil (5-FU) prevented 62% more actinic keratoses than placebo, and this chemopreventive effect persisted for up to 3 years, according to an analysis of the Veterans Affairs Keratinocyte Carcinoma Chemoprevention Trial (VAKCCT) trial.
Other studies have shown that 5-FU effectively treats precancerous AKs, but have not examined whether 5-FU can prevent AKs, Dr. Joanna Walker said in an interview at the annual meeting of the Society for Investigative Dermatology.
Clinicians should consider preventive 5-FU in patients who are at high risk for basal cell and squamous cell carcinomas, especially if a skin check reveals multiple AKs, said Dr. Walker of the department of dermatology, Brown University, Providence, RI.
The VAKCCT was a randomized, double-blind, placebo-controlled study conducted at 12 Veterans Affairs dermatology clinics. The 319 patients in the analysis were nearly all elderly men with extensive sun damage, with a total of 2,386 AKs at baseline, for an average of five lesions per patient. Patients also had a history of at least two keratinocyte carcinomas in the past 5 years, including at least one lesion on the face or ears, and no recent history of 5-FU exposure.
The clinically and demographically similar study arms were randomized to either 5% topical 5-FU cream or a vehicle control cream, applied twice daily for 2-4 weeks. Both groups received cryotherapy for existing AKs, and were given free SPF 30 sunscreen. At each 6-month follow-up visit, the researchers counted existing AKs and new lesions.
At month 6, the treatment group had 62% fewer new AKs than the placebo group (average per patient, 1.78 and 4.73, respectively), a statistically significant difference. At months 12, 18, 24, 30, and 36, respectively, the treatment group had 50%, 40%, 41%, 25%, and 35% fewer new AKs than the placebo group, and these differences all were statistically significant. Furthermore, at month 6, only 56% of treated patients had at least one new AK, compared with 78% of the control group (incidence rate ratio, 0.72; 95% confidence interval, 0.54-0.95).
This chemopreventive effect remained significant for 24 months, the investigators reported. “Individuals with at least five AKs at the time of 5-FU treatment had an even more dramatic reduction in new AKs,” Dr. Walker noted. “There is now high-quality evidence supporting the use of topical 5-FU for AK chemoprevention. I think this is important information that we can take back to the clinic when we are trying to convince our patients to go through a course of 5-FU.”
The rate of new AKs in the placebo group fell during the first 2.5 years of the study and then stabilized. “For both groups, there was a dramatic increase in the use of sunscreen during the trial, and we hypothesized that the decrease in AKs in the control group was due to increased use of sun-protective measures,” Dr. Walker said.
The research was funded by the Cooperative Studies Program of the U.S. Department of Veterans Affairs. Dr. Walker had no disclosures.
SCOTTSDALE – A single course of topical 5-fluorouracil (5-FU) prevented 62% more actinic keratoses than placebo, and this chemopreventive effect persisted for up to 3 years, according to an analysis of the Veterans Affairs Keratinocyte Carcinoma Chemoprevention Trial (VAKCCT) trial.
Other studies have shown that 5-FU effectively treats precancerous AKs, but have not examined whether 5-FU can prevent AKs, Dr. Joanna Walker said in an interview at the annual meeting of the Society for Investigative Dermatology.
Clinicians should consider preventive 5-FU in patients who are at high risk for basal cell and squamous cell carcinomas, especially if a skin check reveals multiple AKs, said Dr. Walker of the department of dermatology, Brown University, Providence, RI.
The VAKCCT was a randomized, double-blind, placebo-controlled study conducted at 12 Veterans Affairs dermatology clinics. The 319 patients in the analysis were nearly all elderly men with extensive sun damage, with a total of 2,386 AKs at baseline, for an average of five lesions per patient. Patients also had a history of at least two keratinocyte carcinomas in the past 5 years, including at least one lesion on the face or ears, and no recent history of 5-FU exposure.
The clinically and demographically similar study arms were randomized to either 5% topical 5-FU cream or a vehicle control cream, applied twice daily for 2-4 weeks. Both groups received cryotherapy for existing AKs, and were given free SPF 30 sunscreen. At each 6-month follow-up visit, the researchers counted existing AKs and new lesions.
At month 6, the treatment group had 62% fewer new AKs than the placebo group (average per patient, 1.78 and 4.73, respectively), a statistically significant difference. At months 12, 18, 24, 30, and 36, respectively, the treatment group had 50%, 40%, 41%, 25%, and 35% fewer new AKs than the placebo group, and these differences all were statistically significant. Furthermore, at month 6, only 56% of treated patients had at least one new AK, compared with 78% of the control group (incidence rate ratio, 0.72; 95% confidence interval, 0.54-0.95).
This chemopreventive effect remained significant for 24 months, the investigators reported. “Individuals with at least five AKs at the time of 5-FU treatment had an even more dramatic reduction in new AKs,” Dr. Walker noted. “There is now high-quality evidence supporting the use of topical 5-FU for AK chemoprevention. I think this is important information that we can take back to the clinic when we are trying to convince our patients to go through a course of 5-FU.”
The rate of new AKs in the placebo group fell during the first 2.5 years of the study and then stabilized. “For both groups, there was a dramatic increase in the use of sunscreen during the trial, and we hypothesized that the decrease in AKs in the control group was due to increased use of sun-protective measures,” Dr. Walker said.
The research was funded by the Cooperative Studies Program of the U.S. Department of Veterans Affairs. Dr. Walker had no disclosures.
AT THE 2016 SID ANNUAL MEETING
Key clinical point: One course of topical 5-fluorouracil was effective and durable in preventing new actinic keratoses in high-risk patients.
Major finding: At month 6, the treatment group had 62% fewer new AKs than the placebo group, and the difference remained significant at month 36.
Data source: The double-blind controlled study evaluated 5-FU vs. a vehicle cream in 319 veterans, most of whom were elderly men.
Disclosures: The study was funded by the Cooperative Studies Program of the U.S. Department of Veterans Affairs. Dr. Walker had no disclosures.
Liquid biopsy–based test detects BRAF mutations
Analysis of plasma-derived cell-free DNA can identify BRAF mutations as accurately as formalin-fixed paraffin-embedded tumor sample analysis, investigators report.
Of 160 patients with advanced solid tumors, BRAF V600 mutations were detected in 62 (39%) archival formalin-fixed paraffin-embedded (FFPE) tumor samples and 47 (29%) plasma cell-free (cf) DNA samples. The two methods had overall agreement in 141 patients (88%; kappa coefficient, 0.74; SE, 0.06; 95% confidence interval, 0.63-0.85), Dr. Filip Janku of the University of Texas and his associates reported (Mol Cancer Ther. 2016 May 20. doi: 10.1158/1535-7163.MCT-15-0712).
BRAF mutations are prevalent in many types of advanced cancers, and targeting these mutations has demonstrated efficacy, but current assessment of BRAF mutations is limited by a lack of tissue samples and complicated by constantly changing mutation status.
cfDNA is secreted into the circulation by tumor cells and cells in the tumor microenvironment that are undergoing apoptosis or necroptosis and might serve as an alternate source for determining BRAF mutation.
“Collecting plasma cfDNA is a minimally invasive procedure that can be repeated at multiple times for diagnostic and disease-monitoring purposes,” Dr. Janku and associates said.
The investigators analyzed plasma samples from 160 patients with advanced solid tumors – most commonly colorectal cancer and melanoma –who had available archival FFPE tumor samples. cfDNA was isolated and extracted from whole blood and analyzed using the Idylla BRAF Mutation Test, a fully integrated real-time polymerase chain reaction–based test with a turnaround time of about 90 minutes.
Dr. Janku and associates found that a higher amount of mutant cfDNA was associated with shorter overall survival. Mean overall survival for patients with a BRAF-mutant cfDNA percentage of less than 2% (10.7 months; 95% CI, 9-12.4) was significantly longer than patients with a BRAF-mutant cfDNA percentage of greater than 2% (4.4 months; 95% CI, 3.2-5.6). Similar results were observed using different percentage cutoffs.
“Our study shows that the Idylla system can detect BRAF V600 mutations in plasma-derived cfDNA from patients with advanced cancers and has acceptable concordance (baseline, 88%; any time point, 90%); sensitivity (baseline, 73%; any time, 77%); and specificity (baseline, 98%; any time, 98%), compared with CLIA-certified laboratory testing of FFPE tumor tissue obtained at different times during routine care,” investigators wrote.
This study was supported by Biocartis, the Elsa U. Pardee Foundation, and the Sidney Kimmel Foundation for Cancer Research. Five of the investigators reported serving in an advisory role for, having ownership interest in, or receiving financial compensation or honoraria from multiple companies. The other investigators reported having no disclosures.
On Twitter @JessCraig_OP
Analysis of plasma-derived cell-free DNA can identify BRAF mutations as accurately as formalin-fixed paraffin-embedded tumor sample analysis, investigators report.
Of 160 patients with advanced solid tumors, BRAF V600 mutations were detected in 62 (39%) archival formalin-fixed paraffin-embedded (FFPE) tumor samples and 47 (29%) plasma cell-free (cf) DNA samples. The two methods had overall agreement in 141 patients (88%; kappa coefficient, 0.74; SE, 0.06; 95% confidence interval, 0.63-0.85), Dr. Filip Janku of the University of Texas and his associates reported (Mol Cancer Ther. 2016 May 20. doi: 10.1158/1535-7163.MCT-15-0712).
BRAF mutations are prevalent in many types of advanced cancers, and targeting these mutations has demonstrated efficacy, but current assessment of BRAF mutations is limited by a lack of tissue samples and complicated by constantly changing mutation status.
cfDNA is secreted into the circulation by tumor cells and cells in the tumor microenvironment that are undergoing apoptosis or necroptosis and might serve as an alternate source for determining BRAF mutation.
“Collecting plasma cfDNA is a minimally invasive procedure that can be repeated at multiple times for diagnostic and disease-monitoring purposes,” Dr. Janku and associates said.
The investigators analyzed plasma samples from 160 patients with advanced solid tumors – most commonly colorectal cancer and melanoma –who had available archival FFPE tumor samples. cfDNA was isolated and extracted from whole blood and analyzed using the Idylla BRAF Mutation Test, a fully integrated real-time polymerase chain reaction–based test with a turnaround time of about 90 minutes.
Dr. Janku and associates found that a higher amount of mutant cfDNA was associated with shorter overall survival. Mean overall survival for patients with a BRAF-mutant cfDNA percentage of less than 2% (10.7 months; 95% CI, 9-12.4) was significantly longer than patients with a BRAF-mutant cfDNA percentage of greater than 2% (4.4 months; 95% CI, 3.2-5.6). Similar results were observed using different percentage cutoffs.
“Our study shows that the Idylla system can detect BRAF V600 mutations in plasma-derived cfDNA from patients with advanced cancers and has acceptable concordance (baseline, 88%; any time point, 90%); sensitivity (baseline, 73%; any time, 77%); and specificity (baseline, 98%; any time, 98%), compared with CLIA-certified laboratory testing of FFPE tumor tissue obtained at different times during routine care,” investigators wrote.
This study was supported by Biocartis, the Elsa U. Pardee Foundation, and the Sidney Kimmel Foundation for Cancer Research. Five of the investigators reported serving in an advisory role for, having ownership interest in, or receiving financial compensation or honoraria from multiple companies. The other investigators reported having no disclosures.
On Twitter @JessCraig_OP
Analysis of plasma-derived cell-free DNA can identify BRAF mutations as accurately as formalin-fixed paraffin-embedded tumor sample analysis, investigators report.
Of 160 patients with advanced solid tumors, BRAF V600 mutations were detected in 62 (39%) archival formalin-fixed paraffin-embedded (FFPE) tumor samples and 47 (29%) plasma cell-free (cf) DNA samples. The two methods had overall agreement in 141 patients (88%; kappa coefficient, 0.74; SE, 0.06; 95% confidence interval, 0.63-0.85), Dr. Filip Janku of the University of Texas and his associates reported (Mol Cancer Ther. 2016 May 20. doi: 10.1158/1535-7163.MCT-15-0712).
BRAF mutations are prevalent in many types of advanced cancers, and targeting these mutations has demonstrated efficacy, but current assessment of BRAF mutations is limited by a lack of tissue samples and complicated by constantly changing mutation status.
cfDNA is secreted into the circulation by tumor cells and cells in the tumor microenvironment that are undergoing apoptosis or necroptosis and might serve as an alternate source for determining BRAF mutation.
“Collecting plasma cfDNA is a minimally invasive procedure that can be repeated at multiple times for diagnostic and disease-monitoring purposes,” Dr. Janku and associates said.
The investigators analyzed plasma samples from 160 patients with advanced solid tumors – most commonly colorectal cancer and melanoma –who had available archival FFPE tumor samples. cfDNA was isolated and extracted from whole blood and analyzed using the Idylla BRAF Mutation Test, a fully integrated real-time polymerase chain reaction–based test with a turnaround time of about 90 minutes.
Dr. Janku and associates found that a higher amount of mutant cfDNA was associated with shorter overall survival. Mean overall survival for patients with a BRAF-mutant cfDNA percentage of less than 2% (10.7 months; 95% CI, 9-12.4) was significantly longer than patients with a BRAF-mutant cfDNA percentage of greater than 2% (4.4 months; 95% CI, 3.2-5.6). Similar results were observed using different percentage cutoffs.
“Our study shows that the Idylla system can detect BRAF V600 mutations in plasma-derived cfDNA from patients with advanced cancers and has acceptable concordance (baseline, 88%; any time point, 90%); sensitivity (baseline, 73%; any time, 77%); and specificity (baseline, 98%; any time, 98%), compared with CLIA-certified laboratory testing of FFPE tumor tissue obtained at different times during routine care,” investigators wrote.
This study was supported by Biocartis, the Elsa U. Pardee Foundation, and the Sidney Kimmel Foundation for Cancer Research. Five of the investigators reported serving in an advisory role for, having ownership interest in, or receiving financial compensation or honoraria from multiple companies. The other investigators reported having no disclosures.
On Twitter @JessCraig_OP
FROM MOLECULAR CANCER THERAPEUTICS
Key clinical point: Analysis of plasma-derived cfDNA can identify BRAF mutations as accurately as FFPE tumor sample analysis.
Major finding: Concordance between mutation analysis of FFPE tumor samples and plasma cfDNA was in overall agreement (88%; kappa, 0.74; SE, 0.06; 95% CI, 0.63-0.85).
Data source: Analysis of FFPE tumor samples and plasma-derived cfDNA for 160 patients with advanced cancers.
Disclosures: This study was supported by Biocartis, the Elsa U. Pardee Foundation, and the Sidney Kimmel Foundation for Cancer Research. Five of the investigators reported serving in an advisory role for, having ownership interest in, or receiving financial compensation or honoraria from multiple companies. The other investigators reported having no disclosures.
Pembrolizumab benefit holds long-term for some melanoma patients
The anti–PD-1 immunotherapy pembrolizumab increases long-term survival in some patients with advanced melanoma, according to updated results of KEYNOTE-001.
Among the 655 patients studied in the phase 1b trial, the 3-year overall survival rate for advanced melanoma patients treated with pembrolizumab was 40%, Dr. Caroline Robert reported in a presscast leading up to the annual meeting of the American Society of Clinical Oncology.
Median overall survival was 24.4 months. Before 2011, patients with advanced melanoma had a median overall survival of less than one year, said Dr. Robert, head of the dermatology unit at the Institut Gustave-Roussy in Paris.
Study participants received pembrolizumab at either 2 or 10 mg/kg every 3 weeks. During the trial, 2 mg/kg every 3 weeks was determined to be the optimal dosing regimen. Patients remained on the treatment until disease progression, intolerable toxicity, or investigator decision. Eight percent of patients stopped treatment due to drug-related symptoms, and there were no drug-related deaths.
Survival rates differed slightly based on prior melanoma treatment. Of the 655 patients in the study, 75% had received previous treatments; patients who had not received prior treatment had slightly higher survival at 45%.
Overall response rate was 33%. Responses were durable as 73% of patients had a response rate of two or more years. Ninety-five patients had a complete response, and 61 of those patients stopped treatment following the complete response.
“I really hope for a cure for these people,” Dr. Robert said.
Pembrolizumab was generally well tolerated with the most common adverse events being fatigue (40%), itchiness (28%), and rash (23%).
ASCO spokesperson and moderator, Dr. Don Dizon, said the results of this study are “incredibly exciting given they came from a phase I trial. This is a new treatment and the longest follow-up of people [with melanoma] who have received pembrolizumab.” Dr. Dizon hopes to “potentially see a cure [for] melanoma” given the reported durability and response rate to the drug.
On Twitter @JessCraig_OP
The anti–PD-1 immunotherapy pembrolizumab increases long-term survival in some patients with advanced melanoma, according to updated results of KEYNOTE-001.
Among the 655 patients studied in the phase 1b trial, the 3-year overall survival rate for advanced melanoma patients treated with pembrolizumab was 40%, Dr. Caroline Robert reported in a presscast leading up to the annual meeting of the American Society of Clinical Oncology.
Median overall survival was 24.4 months. Before 2011, patients with advanced melanoma had a median overall survival of less than one year, said Dr. Robert, head of the dermatology unit at the Institut Gustave-Roussy in Paris.
Study participants received pembrolizumab at either 2 or 10 mg/kg every 3 weeks. During the trial, 2 mg/kg every 3 weeks was determined to be the optimal dosing regimen. Patients remained on the treatment until disease progression, intolerable toxicity, or investigator decision. Eight percent of patients stopped treatment due to drug-related symptoms, and there were no drug-related deaths.
Survival rates differed slightly based on prior melanoma treatment. Of the 655 patients in the study, 75% had received previous treatments; patients who had not received prior treatment had slightly higher survival at 45%.
Overall response rate was 33%. Responses were durable as 73% of patients had a response rate of two or more years. Ninety-five patients had a complete response, and 61 of those patients stopped treatment following the complete response.
“I really hope for a cure for these people,” Dr. Robert said.
Pembrolizumab was generally well tolerated with the most common adverse events being fatigue (40%), itchiness (28%), and rash (23%).
ASCO spokesperson and moderator, Dr. Don Dizon, said the results of this study are “incredibly exciting given they came from a phase I trial. This is a new treatment and the longest follow-up of people [with melanoma] who have received pembrolizumab.” Dr. Dizon hopes to “potentially see a cure [for] melanoma” given the reported durability and response rate to the drug.
On Twitter @JessCraig_OP
The anti–PD-1 immunotherapy pembrolizumab increases long-term survival in some patients with advanced melanoma, according to updated results of KEYNOTE-001.
Among the 655 patients studied in the phase 1b trial, the 3-year overall survival rate for advanced melanoma patients treated with pembrolizumab was 40%, Dr. Caroline Robert reported in a presscast leading up to the annual meeting of the American Society of Clinical Oncology.
Median overall survival was 24.4 months. Before 2011, patients with advanced melanoma had a median overall survival of less than one year, said Dr. Robert, head of the dermatology unit at the Institut Gustave-Roussy in Paris.
Study participants received pembrolizumab at either 2 or 10 mg/kg every 3 weeks. During the trial, 2 mg/kg every 3 weeks was determined to be the optimal dosing regimen. Patients remained on the treatment until disease progression, intolerable toxicity, or investigator decision. Eight percent of patients stopped treatment due to drug-related symptoms, and there were no drug-related deaths.
Survival rates differed slightly based on prior melanoma treatment. Of the 655 patients in the study, 75% had received previous treatments; patients who had not received prior treatment had slightly higher survival at 45%.
Overall response rate was 33%. Responses were durable as 73% of patients had a response rate of two or more years. Ninety-five patients had a complete response, and 61 of those patients stopped treatment following the complete response.
“I really hope for a cure for these people,” Dr. Robert said.
Pembrolizumab was generally well tolerated with the most common adverse events being fatigue (40%), itchiness (28%), and rash (23%).
ASCO spokesperson and moderator, Dr. Don Dizon, said the results of this study are “incredibly exciting given they came from a phase I trial. This is a new treatment and the longest follow-up of people [with melanoma] who have received pembrolizumab.” Dr. Dizon hopes to “potentially see a cure [for] melanoma” given the reported durability and response rate to the drug.
On Twitter @JessCraig_OP
FROM THE 2016 ASCO ANNUAL MEETING
Key clinical point: Pembrolizumab increases long-term survival in some patients with advanced melanoma.
Major finding: The 3-year overall survival rate for advanced melanoma patients treated with pembrolizumab was 40%. The median overall survival was 24.4 months.
Data source: A phase Ib clinical trial of 655 patients with advanced melanoma.
Disclosures: This study was funded by Merck. All of the investigators reported serving in advisory/consultatory roles for, having stock in, or receiving financial compensation or honoraria from several companies.
Early Detection of Melanoma in Men
Men do not know as much about skin cancer prevention and detection techniques as women, according to a recent survey conducted by the American Academy of Dermatology (AAD). This lack of knowledge may delay or prevent early diagnosis and treatment of melanoma and other nonmelanoma skin cancers in this patient population.
The survey results showed that only 56% of men versus 76% of women know there is no such thing as a healthy tan, and only 54% of men versus 70% of women know that getting a base tan is not a healthy way to protect skin from the sun. Furthermore, only 56% of men surveyed were aware that skin cancer could occur on areas of the skin not typically exposed to the sun compared to 65% of women.
“While our survey results indicate that men don’t know as much about skin cancer prevention and detection as women, men over 50 have a higher risk of developing melanoma, so it’s especially important for them to be vigilant about protecting and monitoring their skin,” said AAD President Abel Torres, MD, JD.
May is skin cancer awareness month and the AAD is encouraging patients to make sure their skin is “Looking Good in 2016” by using sun protection and regularly examining skin for signs of skin cancer. The campaign features a public service announcement encouraging men to check their skin for signs of skin cancer and find a partner to help. The AAD also released a new infographic with tips on performing a skin cancer self-examination that dermatologists can share with patients to promote early detection of skin cancer.
Dermatologist intervention in catching skin cancers when they are easier to treat also is key. At the 74th Annual Meeting of the American Academy of Dermatology in Washington, DC, Dr. Orit Markowitz discussed noninvasive imaging tools that can help dermatologists diagnose skin cancers earlier. She noted that even when a lesion looks very small, tools such as dermoscopy can reveal features that indicate it already has depth and therefore may progress to a more serious malignancy. Early detection is particularly crucial in cases of rare aggressive tumors such as amelanotic melanoma. “If something is very pink clinically and then suddenly has pigmentation dermoscopically, you really have to be considering biopsying that lesion because you may be looking at an early amelanotic melanoma,” Dr. Markowitz explained. By the time the lesion develops more obvious clinical features suggesting malignancy, the tumor progression may be far more advanced.
Men do not know as much about skin cancer prevention and detection techniques as women, according to a recent survey conducted by the American Academy of Dermatology (AAD). This lack of knowledge may delay or prevent early diagnosis and treatment of melanoma and other nonmelanoma skin cancers in this patient population.
The survey results showed that only 56% of men versus 76% of women know there is no such thing as a healthy tan, and only 54% of men versus 70% of women know that getting a base tan is not a healthy way to protect skin from the sun. Furthermore, only 56% of men surveyed were aware that skin cancer could occur on areas of the skin not typically exposed to the sun compared to 65% of women.
“While our survey results indicate that men don’t know as much about skin cancer prevention and detection as women, men over 50 have a higher risk of developing melanoma, so it’s especially important for them to be vigilant about protecting and monitoring their skin,” said AAD President Abel Torres, MD, JD.
May is skin cancer awareness month and the AAD is encouraging patients to make sure their skin is “Looking Good in 2016” by using sun protection and regularly examining skin for signs of skin cancer. The campaign features a public service announcement encouraging men to check their skin for signs of skin cancer and find a partner to help. The AAD also released a new infographic with tips on performing a skin cancer self-examination that dermatologists can share with patients to promote early detection of skin cancer.
Dermatologist intervention in catching skin cancers when they are easier to treat also is key. At the 74th Annual Meeting of the American Academy of Dermatology in Washington, DC, Dr. Orit Markowitz discussed noninvasive imaging tools that can help dermatologists diagnose skin cancers earlier. She noted that even when a lesion looks very small, tools such as dermoscopy can reveal features that indicate it already has depth and therefore may progress to a more serious malignancy. Early detection is particularly crucial in cases of rare aggressive tumors such as amelanotic melanoma. “If something is very pink clinically and then suddenly has pigmentation dermoscopically, you really have to be considering biopsying that lesion because you may be looking at an early amelanotic melanoma,” Dr. Markowitz explained. By the time the lesion develops more obvious clinical features suggesting malignancy, the tumor progression may be far more advanced.
Men do not know as much about skin cancer prevention and detection techniques as women, according to a recent survey conducted by the American Academy of Dermatology (AAD). This lack of knowledge may delay or prevent early diagnosis and treatment of melanoma and other nonmelanoma skin cancers in this patient population.
The survey results showed that only 56% of men versus 76% of women know there is no such thing as a healthy tan, and only 54% of men versus 70% of women know that getting a base tan is not a healthy way to protect skin from the sun. Furthermore, only 56% of men surveyed were aware that skin cancer could occur on areas of the skin not typically exposed to the sun compared to 65% of women.
“While our survey results indicate that men don’t know as much about skin cancer prevention and detection as women, men over 50 have a higher risk of developing melanoma, so it’s especially important for them to be vigilant about protecting and monitoring their skin,” said AAD President Abel Torres, MD, JD.
May is skin cancer awareness month and the AAD is encouraging patients to make sure their skin is “Looking Good in 2016” by using sun protection and regularly examining skin for signs of skin cancer. The campaign features a public service announcement encouraging men to check their skin for signs of skin cancer and find a partner to help. The AAD also released a new infographic with tips on performing a skin cancer self-examination that dermatologists can share with patients to promote early detection of skin cancer.
Dermatologist intervention in catching skin cancers when they are easier to treat also is key. At the 74th Annual Meeting of the American Academy of Dermatology in Washington, DC, Dr. Orit Markowitz discussed noninvasive imaging tools that can help dermatologists diagnose skin cancers earlier. She noted that even when a lesion looks very small, tools such as dermoscopy can reveal features that indicate it already has depth and therefore may progress to a more serious malignancy. Early detection is particularly crucial in cases of rare aggressive tumors such as amelanotic melanoma. “If something is very pink clinically and then suddenly has pigmentation dermoscopically, you really have to be considering biopsying that lesion because you may be looking at an early amelanotic melanoma,” Dr. Markowitz explained. By the time the lesion develops more obvious clinical features suggesting malignancy, the tumor progression may be far more advanced.
Mohs surgeons, dermatopathologists nearly always agree on frozen section analysis
ORLANDO – Mohs fellowship–trained surgeons are highly skilled at reading frozen section slides obtained during surgery, agreeing with the dermatopathologist’s assessment more than 99% of the time, according to a review of more than 4,000 cases.
Among 4,145 slides in five datasets, only 28 were discordant. The concordance of surgeons’ and dermatopathologists’ assessments was quite consistent, ranging from 95%-of 99.7%.
“There is absolutely no detriment to our interpretation of these frozen section slides,” Dr. James Highsmith said at the annual meeting of the American College of Mohs Surgery. “These numbers give credence as to why we have these good cure rates. We can say with 99% accuracy that yes, we are removing these tumors from our patients.”
Dr. Highsmith, a Mohs surgeon in Birmingham, Ala., reviewed 10 years of his own data (1,720 cases), as well as four published cohorts comprising another 2,425 cases. All of the studies examined the concordance of frozen section assessments between fellowship-trained Mohs surgeons and the dermatopathologists working with them. All of the series reported concordance, discordance, sensitivity, and specificity.
His own database is the largest one yet compiled, spanning the longest period of time. Of 1,720 cases, there were eight discrepancies: one in which the surgeon identified tumor that was not supported by dermatopathology, and seven in which the surgeon missed tumor that dermatopathology confirmed was present. The concordance rate was 99.5%. This worked out to a 99.4% sensitivity and 99.8% specificity, Dr. Highsmith said.
Results were similar with the other four studies.
The largest was a 2009 retrospective review of 1,156 slides collected over 10 years; they were part of a preexisting randomized, blinded study. Of these, 32 slides (2.8%) were discordant between the Mohs surgeon and the dermatopathologist, for a 99.7% concordance rate (J Am Acad Dermatol. 2009 Jan;[1]: 94-8).
The second largest cohort was published in 1989. It comprised 1,000 slides that were evaluated by the Mohs surgeon and a general pathologist; disputed slides were read by two other Mohs surgeons, two pathologists, and a dermatopathologist. The overall concordance rate was 98.9% (J Am Acad Dermatol. 1989;20[4]:670-4).
In 2013, a Mohs surgeon published a set of 170 slides that had been randomly selected from his cases as part of a quality assurance audit. There was one discordant interpretation, for an overall concordance rate of 99.4% (Dermatol Surg. 2013;39[11]:1648-52).
The smallest study, published in 2011, reviewed 99 slides assessed by a single Mohs surgeon. The concordance rate with dermatopathology was 95%.
An overall analysis of these four cohorts determined a sensitivity of 99.4%, specificity of 99.2% and negative predictive value of 99.2%, Dr. Highsmith said.
“When a fellowship-trained Mohs surgeon looks at a slide and says there’s no more tumor present, then there’s a greater-than-99% chance that is accurate,” he said.
Dr. Highsmith had no financial disclosures.
ORLANDO – Mohs fellowship–trained surgeons are highly skilled at reading frozen section slides obtained during surgery, agreeing with the dermatopathologist’s assessment more than 99% of the time, according to a review of more than 4,000 cases.
Among 4,145 slides in five datasets, only 28 were discordant. The concordance of surgeons’ and dermatopathologists’ assessments was quite consistent, ranging from 95%-of 99.7%.
“There is absolutely no detriment to our interpretation of these frozen section slides,” Dr. James Highsmith said at the annual meeting of the American College of Mohs Surgery. “These numbers give credence as to why we have these good cure rates. We can say with 99% accuracy that yes, we are removing these tumors from our patients.”
Dr. Highsmith, a Mohs surgeon in Birmingham, Ala., reviewed 10 years of his own data (1,720 cases), as well as four published cohorts comprising another 2,425 cases. All of the studies examined the concordance of frozen section assessments between fellowship-trained Mohs surgeons and the dermatopathologists working with them. All of the series reported concordance, discordance, sensitivity, and specificity.
His own database is the largest one yet compiled, spanning the longest period of time. Of 1,720 cases, there were eight discrepancies: one in which the surgeon identified tumor that was not supported by dermatopathology, and seven in which the surgeon missed tumor that dermatopathology confirmed was present. The concordance rate was 99.5%. This worked out to a 99.4% sensitivity and 99.8% specificity, Dr. Highsmith said.
Results were similar with the other four studies.
The largest was a 2009 retrospective review of 1,156 slides collected over 10 years; they were part of a preexisting randomized, blinded study. Of these, 32 slides (2.8%) were discordant between the Mohs surgeon and the dermatopathologist, for a 99.7% concordance rate (J Am Acad Dermatol. 2009 Jan;[1]: 94-8).
The second largest cohort was published in 1989. It comprised 1,000 slides that were evaluated by the Mohs surgeon and a general pathologist; disputed slides were read by two other Mohs surgeons, two pathologists, and a dermatopathologist. The overall concordance rate was 98.9% (J Am Acad Dermatol. 1989;20[4]:670-4).
In 2013, a Mohs surgeon published a set of 170 slides that had been randomly selected from his cases as part of a quality assurance audit. There was one discordant interpretation, for an overall concordance rate of 99.4% (Dermatol Surg. 2013;39[11]:1648-52).
The smallest study, published in 2011, reviewed 99 slides assessed by a single Mohs surgeon. The concordance rate with dermatopathology was 95%.
An overall analysis of these four cohorts determined a sensitivity of 99.4%, specificity of 99.2% and negative predictive value of 99.2%, Dr. Highsmith said.
“When a fellowship-trained Mohs surgeon looks at a slide and says there’s no more tumor present, then there’s a greater-than-99% chance that is accurate,” he said.
Dr. Highsmith had no financial disclosures.
ORLANDO – Mohs fellowship–trained surgeons are highly skilled at reading frozen section slides obtained during surgery, agreeing with the dermatopathologist’s assessment more than 99% of the time, according to a review of more than 4,000 cases.
Among 4,145 slides in five datasets, only 28 were discordant. The concordance of surgeons’ and dermatopathologists’ assessments was quite consistent, ranging from 95%-of 99.7%.
“There is absolutely no detriment to our interpretation of these frozen section slides,” Dr. James Highsmith said at the annual meeting of the American College of Mohs Surgery. “These numbers give credence as to why we have these good cure rates. We can say with 99% accuracy that yes, we are removing these tumors from our patients.”
Dr. Highsmith, a Mohs surgeon in Birmingham, Ala., reviewed 10 years of his own data (1,720 cases), as well as four published cohorts comprising another 2,425 cases. All of the studies examined the concordance of frozen section assessments between fellowship-trained Mohs surgeons and the dermatopathologists working with them. All of the series reported concordance, discordance, sensitivity, and specificity.
His own database is the largest one yet compiled, spanning the longest period of time. Of 1,720 cases, there were eight discrepancies: one in which the surgeon identified tumor that was not supported by dermatopathology, and seven in which the surgeon missed tumor that dermatopathology confirmed was present. The concordance rate was 99.5%. This worked out to a 99.4% sensitivity and 99.8% specificity, Dr. Highsmith said.
Results were similar with the other four studies.
The largest was a 2009 retrospective review of 1,156 slides collected over 10 years; they were part of a preexisting randomized, blinded study. Of these, 32 slides (2.8%) were discordant between the Mohs surgeon and the dermatopathologist, for a 99.7% concordance rate (J Am Acad Dermatol. 2009 Jan;[1]: 94-8).
The second largest cohort was published in 1989. It comprised 1,000 slides that were evaluated by the Mohs surgeon and a general pathologist; disputed slides were read by two other Mohs surgeons, two pathologists, and a dermatopathologist. The overall concordance rate was 98.9% (J Am Acad Dermatol. 1989;20[4]:670-4).
In 2013, a Mohs surgeon published a set of 170 slides that had been randomly selected from his cases as part of a quality assurance audit. There was one discordant interpretation, for an overall concordance rate of 99.4% (Dermatol Surg. 2013;39[11]:1648-52).
The smallest study, published in 2011, reviewed 99 slides assessed by a single Mohs surgeon. The concordance rate with dermatopathology was 95%.
An overall analysis of these four cohorts determined a sensitivity of 99.4%, specificity of 99.2% and negative predictive value of 99.2%, Dr. Highsmith said.
“When a fellowship-trained Mohs surgeon looks at a slide and says there’s no more tumor present, then there’s a greater-than-99% chance that is accurate,” he said.
Dr. Highsmith had no financial disclosures.
AT THE ACMS ANNUAL MEETING
Key clinical point: Fellowship-trained Mohs surgeons and dermatopathologists almost always agree on slide interpretation.
Major finding: The concordance rate between surgeons and dermatopathologists was over 99% in a pooled review of studies.
Data source: A review of 4,145 frozen section slides analyzed for concordance rates between Mohs surgeons and dermatopathologists.
Disclosures: Dr. James Highsmith had no financial disclosures.