User login
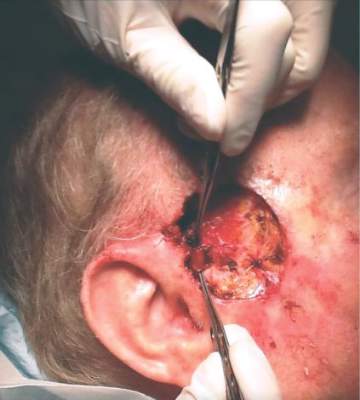
En bloc excision may be viable amputation alternative for nail melanoma
ORLANDO – En bloc excision can cure melanoma in situ of the nail apparatus without amputation, while also preserving function and quality of life, according to the results of a retrospective study of patients treated with this approach.
At 3 years’ follow-up, there were no melanoma recurrences in any of 29 patients treated with the digit-sparing excision, Dr. Thomas Knackstedt said at the annual meeting of the American College of Mohs Surgery. Follow-up telephone interviews found that patients retained full function of the affected finger, and reported virtually no quality of life impairment.
“Because treatment without disarticulation or amputation can provide these excellent long-term cure rates in nail apparatus melanoma in situ, it should be the first-line treatment whenever possible,” said Dr. Knackstedt, a Mohs surgeon in East Greenwich, R.I.
Although similar excellent outcomes have been reported with wide local excision, Dr. Knackstedt said it remains a controversial treatment for nail apparatus melanoma in situ (NAMis).
He reviewed his institution’s experience with treating 29 cases of NAMis with en bloc excision: removal of nail folds, matrix, bed, hyponychium, and margins. All of the surgeries were performed between 2005 and 2015. The current mean follow-up time is 35.5 months for clinic visits and 39 months for telephone contact. The longest follow-up is 103 months. There have been no melanoma recurrences.
The patients were a mean of 39 years old. Melanoma appeared most often on the thumb (seven patients) and hallux (six). The average duration of the lesion was almost 5 years, although that varied from 6 months to 13 years.
Twenty-four patients (83%) presented with longitudinal melanonychia, with an average diameter of 4.1 mm. Four patients (14%) had complete melanonychia, and one patient had erythronychia.
Most of the cases were repaired with a full-thickness skin graft. One was left to heal by second intention, and one case was repaired by a dorsal metacarpal artery flap.
There were two wound infections, one case of delayed graft necrosis, seven nail spicules, and two cysts. There were no cases of tendon injury. One patient complained about temperature sensitivity in the affected finger. There were no cases of disease recurrence, although one patient with a nail spicule did show disease persistence. The spicule was excised, and that patient remains disease free 5 years later, Dr. Knackstedt said.
The telephone survey collected data from 23 patients (79%). Patients were unanimous in reporting that they were “very satisfied” with their treatment. In an 11-question quality of life survey, 78% said that the surgery affected their quality of life “only a little” or “not at all.” On a 10-point Likert scale with 10 being most affected by the surgery, the mean rating was 1.3.
“In light of these results, and when we consider that even a distal joint amputation can reduce the functionality of a finger by 40%-80%, we conclude that en bloc excision must be considered as a viable alternative to amputation for NAMis,” Dr. Knackstedt said.
He had no relevant financial disclosures.
ORLANDO – En bloc excision can cure melanoma in situ of the nail apparatus without amputation, while also preserving function and quality of life, according to the results of a retrospective study of patients treated with this approach.
At 3 years’ follow-up, there were no melanoma recurrences in any of 29 patients treated with the digit-sparing excision, Dr. Thomas Knackstedt said at the annual meeting of the American College of Mohs Surgery. Follow-up telephone interviews found that patients retained full function of the affected finger, and reported virtually no quality of life impairment.
“Because treatment without disarticulation or amputation can provide these excellent long-term cure rates in nail apparatus melanoma in situ, it should be the first-line treatment whenever possible,” said Dr. Knackstedt, a Mohs surgeon in East Greenwich, R.I.
Although similar excellent outcomes have been reported with wide local excision, Dr. Knackstedt said it remains a controversial treatment for nail apparatus melanoma in situ (NAMis).
He reviewed his institution’s experience with treating 29 cases of NAMis with en bloc excision: removal of nail folds, matrix, bed, hyponychium, and margins. All of the surgeries were performed between 2005 and 2015. The current mean follow-up time is 35.5 months for clinic visits and 39 months for telephone contact. The longest follow-up is 103 months. There have been no melanoma recurrences.
The patients were a mean of 39 years old. Melanoma appeared most often on the thumb (seven patients) and hallux (six). The average duration of the lesion was almost 5 years, although that varied from 6 months to 13 years.
Twenty-four patients (83%) presented with longitudinal melanonychia, with an average diameter of 4.1 mm. Four patients (14%) had complete melanonychia, and one patient had erythronychia.
Most of the cases were repaired with a full-thickness skin graft. One was left to heal by second intention, and one case was repaired by a dorsal metacarpal artery flap.
There were two wound infections, one case of delayed graft necrosis, seven nail spicules, and two cysts. There were no cases of tendon injury. One patient complained about temperature sensitivity in the affected finger. There were no cases of disease recurrence, although one patient with a nail spicule did show disease persistence. The spicule was excised, and that patient remains disease free 5 years later, Dr. Knackstedt said.
The telephone survey collected data from 23 patients (79%). Patients were unanimous in reporting that they were “very satisfied” with their treatment. In an 11-question quality of life survey, 78% said that the surgery affected their quality of life “only a little” or “not at all.” On a 10-point Likert scale with 10 being most affected by the surgery, the mean rating was 1.3.
“In light of these results, and when we consider that even a distal joint amputation can reduce the functionality of a finger by 40%-80%, we conclude that en bloc excision must be considered as a viable alternative to amputation for NAMis,” Dr. Knackstedt said.
He had no relevant financial disclosures.
ORLANDO – En bloc excision can cure melanoma in situ of the nail apparatus without amputation, while also preserving function and quality of life, according to the results of a retrospective study of patients treated with this approach.
At 3 years’ follow-up, there were no melanoma recurrences in any of 29 patients treated with the digit-sparing excision, Dr. Thomas Knackstedt said at the annual meeting of the American College of Mohs Surgery. Follow-up telephone interviews found that patients retained full function of the affected finger, and reported virtually no quality of life impairment.
“Because treatment without disarticulation or amputation can provide these excellent long-term cure rates in nail apparatus melanoma in situ, it should be the first-line treatment whenever possible,” said Dr. Knackstedt, a Mohs surgeon in East Greenwich, R.I.
Although similar excellent outcomes have been reported with wide local excision, Dr. Knackstedt said it remains a controversial treatment for nail apparatus melanoma in situ (NAMis).
He reviewed his institution’s experience with treating 29 cases of NAMis with en bloc excision: removal of nail folds, matrix, bed, hyponychium, and margins. All of the surgeries were performed between 2005 and 2015. The current mean follow-up time is 35.5 months for clinic visits and 39 months for telephone contact. The longest follow-up is 103 months. There have been no melanoma recurrences.
The patients were a mean of 39 years old. Melanoma appeared most often on the thumb (seven patients) and hallux (six). The average duration of the lesion was almost 5 years, although that varied from 6 months to 13 years.
Twenty-four patients (83%) presented with longitudinal melanonychia, with an average diameter of 4.1 mm. Four patients (14%) had complete melanonychia, and one patient had erythronychia.
Most of the cases were repaired with a full-thickness skin graft. One was left to heal by second intention, and one case was repaired by a dorsal metacarpal artery flap.
There were two wound infections, one case of delayed graft necrosis, seven nail spicules, and two cysts. There were no cases of tendon injury. One patient complained about temperature sensitivity in the affected finger. There were no cases of disease recurrence, although one patient with a nail spicule did show disease persistence. The spicule was excised, and that patient remains disease free 5 years later, Dr. Knackstedt said.
The telephone survey collected data from 23 patients (79%). Patients were unanimous in reporting that they were “very satisfied” with their treatment. In an 11-question quality of life survey, 78% said that the surgery affected their quality of life “only a little” or “not at all.” On a 10-point Likert scale with 10 being most affected by the surgery, the mean rating was 1.3.
“In light of these results, and when we consider that even a distal joint amputation can reduce the functionality of a finger by 40%-80%, we conclude that en bloc excision must be considered as a viable alternative to amputation for NAMis,” Dr. Knackstedt said.
He had no relevant financial disclosures.
AT THE ACMS ANNUAL MEETING
Key clinical point: En bloc removal of the nail apparatus effected good clinical and quality of life outcomes in nail melanoma.
Major finding: There have been no recurrences of melanoma over a mean follow-up of 39 months.
Data source: A review evaluated 29 cases of nail apparatus melanoma in situ treated with en bloc excision at one institution.
Disclosures: Dr. Knackstedt had no relevant financial disclosures.
What’s Less Noticeable: A Straight Scar or a Zigzag Scar?
One of the determinants of a successful surgical outcome is the perception, on the part of the patient, of the cosmesis of a scar. The use of Z-plasty is an accepted means by which to break a scar up into smaller geometric segments. In some instances, a Z-plasty is used for scar revision to elongate a scar that may be pulling. However, a study published online in JAMA Facial Plastic Surgery on April 7 mentions the lack of studies measuring the perception of these scars among the normal population after surgery.
Ratnarathorn et al designed a prospective Internet-based survey with a goal of 580 responses to give a power of 90%. The survey was distributed to a diverse sample of the US population. Using editing software, Ratnarathorn et al superimposed a mature linear scar and a mature zigzag scar onto the faces of standardized headshots from 4 individuals (2 males, 2 females). Each individual had 1 image of the linear scar and 1 image of the zigzag scars superimposed onto each of 3 anatomical areas—forehead (flat surface), cheek (convex surface), and temple (concave surface)—yielding 24 images for the respondents to assess.
A 24.5% (n=876) response rate was achieved with 3575 surveys distributed. Of the 876 respondents, 810 (92.5%) completed the survey (46.1% male, 53.9% female). Respondents were asked to rate the scars on a scale of 1 to 10 (1=normal skin; 10=worst scar imaginable).
Results were statistically significantly lower (better) for the linear scars compared to the zigzag scars in all 3 anatomic areas and across both male and female groups with a mean score of 2.9 versus 4.5 (P<.001). A multivariable regression model of respondent age, sex, educational level, and income showed no statistically significant effect on the rating of the scars.
What’s the issue?
This study highlights some interesting points. Coming from an academic practice, we oftentimes find ourselves teaching residents a variety of skin closure techniques to deal with defects from skin cancer excisions. It is both challenging and fun to design complex closures; however, we must keep in mind what is in the best interest of the patient. One of the points I try to emphasize is that we must understand that there are no true straight lines on the face. In fact, when scars from procedures appear as geometric shapes on the face, our eyes tend to be drawn to them. For this reason, it often is best to use curvilinear lines wherever possible. Ratnarathorn et al highlights that point exactly. More studies of this nature are needed to assess what is perceived as a successful outcome, by both physicians and patients.
As you follow your patients for the long-term, have you noticed that you perform more or fewer zigzag scars?
One of the determinants of a successful surgical outcome is the perception, on the part of the patient, of the cosmesis of a scar. The use of Z-plasty is an accepted means by which to break a scar up into smaller geometric segments. In some instances, a Z-plasty is used for scar revision to elongate a scar that may be pulling. However, a study published online in JAMA Facial Plastic Surgery on April 7 mentions the lack of studies measuring the perception of these scars among the normal population after surgery.
Ratnarathorn et al designed a prospective Internet-based survey with a goal of 580 responses to give a power of 90%. The survey was distributed to a diverse sample of the US population. Using editing software, Ratnarathorn et al superimposed a mature linear scar and a mature zigzag scar onto the faces of standardized headshots from 4 individuals (2 males, 2 females). Each individual had 1 image of the linear scar and 1 image of the zigzag scars superimposed onto each of 3 anatomical areas—forehead (flat surface), cheek (convex surface), and temple (concave surface)—yielding 24 images for the respondents to assess.
A 24.5% (n=876) response rate was achieved with 3575 surveys distributed. Of the 876 respondents, 810 (92.5%) completed the survey (46.1% male, 53.9% female). Respondents were asked to rate the scars on a scale of 1 to 10 (1=normal skin; 10=worst scar imaginable).
Results were statistically significantly lower (better) for the linear scars compared to the zigzag scars in all 3 anatomic areas and across both male and female groups with a mean score of 2.9 versus 4.5 (P<.001). A multivariable regression model of respondent age, sex, educational level, and income showed no statistically significant effect on the rating of the scars.
What’s the issue?
This study highlights some interesting points. Coming from an academic practice, we oftentimes find ourselves teaching residents a variety of skin closure techniques to deal with defects from skin cancer excisions. It is both challenging and fun to design complex closures; however, we must keep in mind what is in the best interest of the patient. One of the points I try to emphasize is that we must understand that there are no true straight lines on the face. In fact, when scars from procedures appear as geometric shapes on the face, our eyes tend to be drawn to them. For this reason, it often is best to use curvilinear lines wherever possible. Ratnarathorn et al highlights that point exactly. More studies of this nature are needed to assess what is perceived as a successful outcome, by both physicians and patients.
As you follow your patients for the long-term, have you noticed that you perform more or fewer zigzag scars?
One of the determinants of a successful surgical outcome is the perception, on the part of the patient, of the cosmesis of a scar. The use of Z-plasty is an accepted means by which to break a scar up into smaller geometric segments. In some instances, a Z-plasty is used for scar revision to elongate a scar that may be pulling. However, a study published online in JAMA Facial Plastic Surgery on April 7 mentions the lack of studies measuring the perception of these scars among the normal population after surgery.
Ratnarathorn et al designed a prospective Internet-based survey with a goal of 580 responses to give a power of 90%. The survey was distributed to a diverse sample of the US population. Using editing software, Ratnarathorn et al superimposed a mature linear scar and a mature zigzag scar onto the faces of standardized headshots from 4 individuals (2 males, 2 females). Each individual had 1 image of the linear scar and 1 image of the zigzag scars superimposed onto each of 3 anatomical areas—forehead (flat surface), cheek (convex surface), and temple (concave surface)—yielding 24 images for the respondents to assess.
A 24.5% (n=876) response rate was achieved with 3575 surveys distributed. Of the 876 respondents, 810 (92.5%) completed the survey (46.1% male, 53.9% female). Respondents were asked to rate the scars on a scale of 1 to 10 (1=normal skin; 10=worst scar imaginable).
Results were statistically significantly lower (better) for the linear scars compared to the zigzag scars in all 3 anatomic areas and across both male and female groups with a mean score of 2.9 versus 4.5 (P<.001). A multivariable regression model of respondent age, sex, educational level, and income showed no statistically significant effect on the rating of the scars.
What’s the issue?
This study highlights some interesting points. Coming from an academic practice, we oftentimes find ourselves teaching residents a variety of skin closure techniques to deal with defects from skin cancer excisions. It is both challenging and fun to design complex closures; however, we must keep in mind what is in the best interest of the patient. One of the points I try to emphasize is that we must understand that there are no true straight lines on the face. In fact, when scars from procedures appear as geometric shapes on the face, our eyes tend to be drawn to them. For this reason, it often is best to use curvilinear lines wherever possible. Ratnarathorn et al highlights that point exactly. More studies of this nature are needed to assess what is perceived as a successful outcome, by both physicians and patients.
As you follow your patients for the long-term, have you noticed that you perform more or fewer zigzag scars?
Search is on for cases of aggressive, ruxolitinib-associated skin cancers
ORLANDO – The hematologic cancer drug ruxolitinib seems to be associated with cases of aggressive nonmelanoma skin cancer.
After treating a very aggressive squamous cell carcinoma in a 55-year-old man treated with ruxolitinib for polycythemia vera, and hearing firsthand of three other similar cases, Dr. Fiona Zwald is collecting additional data on the association. She intends to publish these cases in a monograph as a warning to dermatologists, hematologists, oncologists, and other physicians who manage patients with hematologic malignancies, she said at the annual meeting of the American College of Mohs Surgery.
The prescribing information for ruxolitinib (Jakafi, Incyte Pharmaceuticals; Jakavi, Novartis) was updated in 2014 to warn that patients taking the drug face an increased risk of nonmelanoma skin cancers. The label also recommends that physicians inspect the skin regularly and urge patients to be alert for and report any new or changing lesions.
Despite the warnings and recommendations, cases are occurring – and some are quite serious, said Dr. Zwald, a Mohs surgeon in Atlanta.
“People should know this is actually happening. If you have experience with this medication, please let us know so we can compile this report. We are trying to assess the number of skin cancers before and after initiating this medication,” she said.
Ruxolitinib is an inhibitor of Janus kinase with a special affinity for the JAK1 and JAK2 subtypes. Like other cytokine-signaling molecules, their function depends on cell context; it may inhibit cell growth in one setting, and, in another, stimulate it. Ruxolitinib was initially approved in 2011 for the treatment of intermediate- and high-risk myelofibrosis, including primary myelofibrosis, post–polycythemia vera myelofibrosis, and post–essential thrombocythemia myelofibrosis.
In 2014, indications for ruxolitinib were expanded to include treatment of patients with polycythemia vera who have had an inadequate response to or are intolerant of hydroxyurea.
Dr. Zwald’s patient had a 10-year history of polycythemia vera. He was initially well controlled on the standard hydroxyurea treatment. In the meantime, he began working as a caddy at a major U.S. golf club. He developed many facial squamous cell carcinomas that were treated with excision and radiation. A year before he presented to Dr. Zwald, he stopped responding to hydroxyurea and was placed on ruxolitinib.
The patient presented with a 4-cm ulcerated lesion over part of his right temple and to the right helical crus; the lesion had developed over 3 months. Dr. Zwald consulted with the patient’s medical oncologist; treatment with ruxolitinib continued, albeit at a reduced dosage in light of recent events.
She performed Mohs surgery on the patient. It was a challenging case, she said, not the least because adequate anesthesia could not be achieved with local anesthetic. Preoperative staging showed no nodal spread.
“He did, unfortunately demonstrate a large, indurated mass located over one branch of the superficial temporal artery. At the helical crus there was an area of bound-down, fixed tumor. Knowing that I would not be able to fully resect this, I passed him on to the operating room,” Dr. Zwald said. “This tumor was found to extend down to the parotid capsule, but margins were clear.” The surgical defect was successfully repaired with a split-thickness skin graft.
The tumor recurred about 3 months later, and the patient underwent another surgery.
“This time we could not get clear surgical margins, and the tumor was approaching the external auditory meatus. Surgery was abandoned due to fears of complications to that area,” she said.
She presented the case at tumor board, during which she and her colleagues discussed adjuvant radiation. They initially abandoned this idea because he had already had so much radiation to his face. After the second surgery, they decide to proceed with radiation. “The next conversation we have will be whether to add another adjuvant therapy to treatment.”
She sent out the case and requests for feedback to the International Transplant Skin Cancer Collaborative, an 800-member consortium of dermatologists and Mohs surgeons who take care of transplant patients. She received information on three additional cases of aggressive squamous cell carcinoma (SCC) associated with ruxolitinib treatment:
• A patient with myelodysplastic syndrome with aggressive scalp SCC with cutaneous metastases.
• A patient with undifferentiated pleomorphic sarcoma of the scalp, several cutaneous SCCs.
• A patient with a myelodysplastic syndrome with in-transit metastases and explosive cutaneous SCCs. The patient has had the ruxolitinib dose reduced and may be switched to capecitabine.
Dr. Zwald noted that her patient was at risk for aggressive skin cancers for reasons in addition to ruxolitinib treatment.
“He was already immunosuppressed from his malignancy. He was on hydroxyurea, a drug that’s a cumulative phototoxin, and he’s out in the sun playing golf every day, and then was put on ruxolitinib. But the question we face now is how to try and stop this medication so we can get better treatment for him which will, of course, be very difficult.”
To contribute to Dr. Zwald’s case series, please email her at Fiona.Zwald@gmail.com.
She had no relevant financial disclosures.
On Twitter @Alz_Gal
ORLANDO – The hematologic cancer drug ruxolitinib seems to be associated with cases of aggressive nonmelanoma skin cancer.
After treating a very aggressive squamous cell carcinoma in a 55-year-old man treated with ruxolitinib for polycythemia vera, and hearing firsthand of three other similar cases, Dr. Fiona Zwald is collecting additional data on the association. She intends to publish these cases in a monograph as a warning to dermatologists, hematologists, oncologists, and other physicians who manage patients with hematologic malignancies, she said at the annual meeting of the American College of Mohs Surgery.
The prescribing information for ruxolitinib (Jakafi, Incyte Pharmaceuticals; Jakavi, Novartis) was updated in 2014 to warn that patients taking the drug face an increased risk of nonmelanoma skin cancers. The label also recommends that physicians inspect the skin regularly and urge patients to be alert for and report any new or changing lesions.
Despite the warnings and recommendations, cases are occurring – and some are quite serious, said Dr. Zwald, a Mohs surgeon in Atlanta.
“People should know this is actually happening. If you have experience with this medication, please let us know so we can compile this report. We are trying to assess the number of skin cancers before and after initiating this medication,” she said.
Ruxolitinib is an inhibitor of Janus kinase with a special affinity for the JAK1 and JAK2 subtypes. Like other cytokine-signaling molecules, their function depends on cell context; it may inhibit cell growth in one setting, and, in another, stimulate it. Ruxolitinib was initially approved in 2011 for the treatment of intermediate- and high-risk myelofibrosis, including primary myelofibrosis, post–polycythemia vera myelofibrosis, and post–essential thrombocythemia myelofibrosis.
In 2014, indications for ruxolitinib were expanded to include treatment of patients with polycythemia vera who have had an inadequate response to or are intolerant of hydroxyurea.
Dr. Zwald’s patient had a 10-year history of polycythemia vera. He was initially well controlled on the standard hydroxyurea treatment. In the meantime, he began working as a caddy at a major U.S. golf club. He developed many facial squamous cell carcinomas that were treated with excision and radiation. A year before he presented to Dr. Zwald, he stopped responding to hydroxyurea and was placed on ruxolitinib.
The patient presented with a 4-cm ulcerated lesion over part of his right temple and to the right helical crus; the lesion had developed over 3 months. Dr. Zwald consulted with the patient’s medical oncologist; treatment with ruxolitinib continued, albeit at a reduced dosage in light of recent events.
She performed Mohs surgery on the patient. It was a challenging case, she said, not the least because adequate anesthesia could not be achieved with local anesthetic. Preoperative staging showed no nodal spread.
“He did, unfortunately demonstrate a large, indurated mass located over one branch of the superficial temporal artery. At the helical crus there was an area of bound-down, fixed tumor. Knowing that I would not be able to fully resect this, I passed him on to the operating room,” Dr. Zwald said. “This tumor was found to extend down to the parotid capsule, but margins were clear.” The surgical defect was successfully repaired with a split-thickness skin graft.
The tumor recurred about 3 months later, and the patient underwent another surgery.
“This time we could not get clear surgical margins, and the tumor was approaching the external auditory meatus. Surgery was abandoned due to fears of complications to that area,” she said.
She presented the case at tumor board, during which she and her colleagues discussed adjuvant radiation. They initially abandoned this idea because he had already had so much radiation to his face. After the second surgery, they decide to proceed with radiation. “The next conversation we have will be whether to add another adjuvant therapy to treatment.”
She sent out the case and requests for feedback to the International Transplant Skin Cancer Collaborative, an 800-member consortium of dermatologists and Mohs surgeons who take care of transplant patients. She received information on three additional cases of aggressive squamous cell carcinoma (SCC) associated with ruxolitinib treatment:
• A patient with myelodysplastic syndrome with aggressive scalp SCC with cutaneous metastases.
• A patient with undifferentiated pleomorphic sarcoma of the scalp, several cutaneous SCCs.
• A patient with a myelodysplastic syndrome with in-transit metastases and explosive cutaneous SCCs. The patient has had the ruxolitinib dose reduced and may be switched to capecitabine.
Dr. Zwald noted that her patient was at risk for aggressive skin cancers for reasons in addition to ruxolitinib treatment.
“He was already immunosuppressed from his malignancy. He was on hydroxyurea, a drug that’s a cumulative phototoxin, and he’s out in the sun playing golf every day, and then was put on ruxolitinib. But the question we face now is how to try and stop this medication so we can get better treatment for him which will, of course, be very difficult.”
To contribute to Dr. Zwald’s case series, please email her at Fiona.Zwald@gmail.com.
She had no relevant financial disclosures.
On Twitter @Alz_Gal
ORLANDO – The hematologic cancer drug ruxolitinib seems to be associated with cases of aggressive nonmelanoma skin cancer.
After treating a very aggressive squamous cell carcinoma in a 55-year-old man treated with ruxolitinib for polycythemia vera, and hearing firsthand of three other similar cases, Dr. Fiona Zwald is collecting additional data on the association. She intends to publish these cases in a monograph as a warning to dermatologists, hematologists, oncologists, and other physicians who manage patients with hematologic malignancies, she said at the annual meeting of the American College of Mohs Surgery.
The prescribing information for ruxolitinib (Jakafi, Incyte Pharmaceuticals; Jakavi, Novartis) was updated in 2014 to warn that patients taking the drug face an increased risk of nonmelanoma skin cancers. The label also recommends that physicians inspect the skin regularly and urge patients to be alert for and report any new or changing lesions.
Despite the warnings and recommendations, cases are occurring – and some are quite serious, said Dr. Zwald, a Mohs surgeon in Atlanta.
“People should know this is actually happening. If you have experience with this medication, please let us know so we can compile this report. We are trying to assess the number of skin cancers before and after initiating this medication,” she said.
Ruxolitinib is an inhibitor of Janus kinase with a special affinity for the JAK1 and JAK2 subtypes. Like other cytokine-signaling molecules, their function depends on cell context; it may inhibit cell growth in one setting, and, in another, stimulate it. Ruxolitinib was initially approved in 2011 for the treatment of intermediate- and high-risk myelofibrosis, including primary myelofibrosis, post–polycythemia vera myelofibrosis, and post–essential thrombocythemia myelofibrosis.
In 2014, indications for ruxolitinib were expanded to include treatment of patients with polycythemia vera who have had an inadequate response to or are intolerant of hydroxyurea.
Dr. Zwald’s patient had a 10-year history of polycythemia vera. He was initially well controlled on the standard hydroxyurea treatment. In the meantime, he began working as a caddy at a major U.S. golf club. He developed many facial squamous cell carcinomas that were treated with excision and radiation. A year before he presented to Dr. Zwald, he stopped responding to hydroxyurea and was placed on ruxolitinib.
The patient presented with a 4-cm ulcerated lesion over part of his right temple and to the right helical crus; the lesion had developed over 3 months. Dr. Zwald consulted with the patient’s medical oncologist; treatment with ruxolitinib continued, albeit at a reduced dosage in light of recent events.
She performed Mohs surgery on the patient. It was a challenging case, she said, not the least because adequate anesthesia could not be achieved with local anesthetic. Preoperative staging showed no nodal spread.
“He did, unfortunately demonstrate a large, indurated mass located over one branch of the superficial temporal artery. At the helical crus there was an area of bound-down, fixed tumor. Knowing that I would not be able to fully resect this, I passed him on to the operating room,” Dr. Zwald said. “This tumor was found to extend down to the parotid capsule, but margins were clear.” The surgical defect was successfully repaired with a split-thickness skin graft.
The tumor recurred about 3 months later, and the patient underwent another surgery.
“This time we could not get clear surgical margins, and the tumor was approaching the external auditory meatus. Surgery was abandoned due to fears of complications to that area,” she said.
She presented the case at tumor board, during which she and her colleagues discussed adjuvant radiation. They initially abandoned this idea because he had already had so much radiation to his face. After the second surgery, they decide to proceed with radiation. “The next conversation we have will be whether to add another adjuvant therapy to treatment.”
She sent out the case and requests for feedback to the International Transplant Skin Cancer Collaborative, an 800-member consortium of dermatologists and Mohs surgeons who take care of transplant patients. She received information on three additional cases of aggressive squamous cell carcinoma (SCC) associated with ruxolitinib treatment:
• A patient with myelodysplastic syndrome with aggressive scalp SCC with cutaneous metastases.
• A patient with undifferentiated pleomorphic sarcoma of the scalp, several cutaneous SCCs.
• A patient with a myelodysplastic syndrome with in-transit metastases and explosive cutaneous SCCs. The patient has had the ruxolitinib dose reduced and may be switched to capecitabine.
Dr. Zwald noted that her patient was at risk for aggressive skin cancers for reasons in addition to ruxolitinib treatment.
“He was already immunosuppressed from his malignancy. He was on hydroxyurea, a drug that’s a cumulative phototoxin, and he’s out in the sun playing golf every day, and then was put on ruxolitinib. But the question we face now is how to try and stop this medication so we can get better treatment for him which will, of course, be very difficult.”
To contribute to Dr. Zwald’s case series, please email her at Fiona.Zwald@gmail.com.
She had no relevant financial disclosures.
On Twitter @Alz_Gal
AT THE ACMS ANNUAL MEETING
Mohs with CK-7 staining: 98% 5-year cure rate for extramammary Paget disease
ORLANDO – Mohs surgery with cytokeratin-7 immunohistochemistry staining effected complete removal of extramammary Paget disease and resulted in a 5-year, 95% recurrence-free cure rate.
The results are significantly better than the often-cited 77% cure rate seen with Mohs surgery alone, Dr. Ali Alexander Damavandy said at the annual meeting of the American College of Mohs Surgery.
“These are statistically significant and clinically substantial results,” said Dr. Damavandy, a procedural dermatology fellow at the University of Pennsylvania, Philadelphia. “With this method you can tell a patient that in 5 years, he has a 95% chance of still not having the tumor. The high recurrence-free rate we have seen supports the view that Mohs surgery with cytokeratin-7 [CK-7] immunohistochemistry should be considered the curative treatment of choice for both primary and recurrent extramammary Paget disease of the skin.”
Dr. Damavandy’s retrospective review of 49 cases may seem small, he said, but it is “far and away the biggest cohort that exists, and the only substantial cohort with immunohistochemistry staining of CK-7.” The 5-year follow-up is also a strength of the study, he added.
Extramammary Paget disease (EMPD) remains an “extraordinarily hard tumor to treat. It typically presents with widespread intraepithelial islands of disease that are not clinically apparent. Local recurrence rates range from 33% to 60%, “leaving a lot of room for improvement,” Dr. Damavandy said.
“The explanations for recurrence are twofold,” he added. “First there is difficulty visualizing tumor cells in frozen sections. Second, it is a multifocal tumor that grows in a discontinuous fashion.”
Although one simply has to deal with the tumor spread, improvements can be made in reading the histopathology. Immunohistochemistry staining with CK-7 allows highly accurate visualization of individual Paget tumor cells, which express a very high level of the protein.
“Our hypothesis was that combining this improved tumor cell visualization with Mohs total excision would really make a dent in the recurrence rates of this disease,” Dr. Damavandy said.
He conducted a review of patients who were treated at four practices from 2004 to 2012. The cohort comprised 49 patients with 62 tumors; 43 were primary and the rest were recurrences. The majority (90%) occurred in the urogenital and anogenital region; the rest were axial or facial. All were treated with Mohs microsurgery enhanced by CK-7 staining of frozen sections. Recurrences were biopsy proven.
The typical Mohs technique for EMPD is to delineate a tumor-free area peripherally, and then excise the central tumor and tumor-bearing islands. The procedure can be long and arduous. Many cases are multiday efforts with large areas of excision.
“These can be very difficult wounds to manage even in a hospital. It’s resource-intense surgery,” Dr. Damavandy said.
The mean margin necessary to clear the lesions in this cohort was 2.6 cm. A 4-cm margin cleared almost 80%; 10% required a 7-cm margin.
“If we were going to make a recommendation to a nonMohs surgeon, it would be that 7 cm are needed to clear 98% of the tumors. That seems to me not reasonable for these anatomically complex and delicate regions. At the same time, the most commonly reported margin in the literature is 2 cm. In this cohort that would only have cleared 40% of the tumors.”
A mean of 3.4 Mohs stages were needed to obtain clear margins, although the range was wide (1-14 stages). Surgery took a mean of 1.5 days (range 1-5 days). Four patients could not be cleared and needed multidisciplinary care. These included two with vaginal invasion and two with invasion of the anal canal.
By 5 years, the overall tumor-free survival rate was 97%; the tumor-free survival rate was slightly better for primary than recurrent tumors (98% vs. 95%). When these rates were compared with Mohs without CK-7 staining, “primary Mohs doesn’t compare very well,” Dr. Damavandy said. At 5 years, the literature-reported recurrence rate for primary tumors treated with Mohs alone was 16% vs. Dr. Damavandy’s report of 2.3% for Mohs plus CK-7 staining. For recurrent tumors, the recurrence rates were 50% vs. 5.3%.
Two tumors recurred; the mean time to recurrence was 31 months. One recurrence was of a primary tumor treated by peripheral Mohs surgery, which was an uncommon approach in this cohort. This patient had a deep recurrence at 16 months.
The other case was a recurrence of a vulvar tumor that extended into the vaginal epithelium and across the cervix. Despite a vaginectomy and vaginal hysterectomy with clear margins, the patient had a recurrence at 46 months.
The patients were retreated and at last follow-up were free of disease.
Dr. Damavandy had no financial disclosures.
ORLANDO – Mohs surgery with cytokeratin-7 immunohistochemistry staining effected complete removal of extramammary Paget disease and resulted in a 5-year, 95% recurrence-free cure rate.
The results are significantly better than the often-cited 77% cure rate seen with Mohs surgery alone, Dr. Ali Alexander Damavandy said at the annual meeting of the American College of Mohs Surgery.
“These are statistically significant and clinically substantial results,” said Dr. Damavandy, a procedural dermatology fellow at the University of Pennsylvania, Philadelphia. “With this method you can tell a patient that in 5 years, he has a 95% chance of still not having the tumor. The high recurrence-free rate we have seen supports the view that Mohs surgery with cytokeratin-7 [CK-7] immunohistochemistry should be considered the curative treatment of choice for both primary and recurrent extramammary Paget disease of the skin.”
Dr. Damavandy’s retrospective review of 49 cases may seem small, he said, but it is “far and away the biggest cohort that exists, and the only substantial cohort with immunohistochemistry staining of CK-7.” The 5-year follow-up is also a strength of the study, he added.
Extramammary Paget disease (EMPD) remains an “extraordinarily hard tumor to treat. It typically presents with widespread intraepithelial islands of disease that are not clinically apparent. Local recurrence rates range from 33% to 60%, “leaving a lot of room for improvement,” Dr. Damavandy said.
“The explanations for recurrence are twofold,” he added. “First there is difficulty visualizing tumor cells in frozen sections. Second, it is a multifocal tumor that grows in a discontinuous fashion.”
Although one simply has to deal with the tumor spread, improvements can be made in reading the histopathology. Immunohistochemistry staining with CK-7 allows highly accurate visualization of individual Paget tumor cells, which express a very high level of the protein.
“Our hypothesis was that combining this improved tumor cell visualization with Mohs total excision would really make a dent in the recurrence rates of this disease,” Dr. Damavandy said.
He conducted a review of patients who were treated at four practices from 2004 to 2012. The cohort comprised 49 patients with 62 tumors; 43 were primary and the rest were recurrences. The majority (90%) occurred in the urogenital and anogenital region; the rest were axial or facial. All were treated with Mohs microsurgery enhanced by CK-7 staining of frozen sections. Recurrences were biopsy proven.
The typical Mohs technique for EMPD is to delineate a tumor-free area peripherally, and then excise the central tumor and tumor-bearing islands. The procedure can be long and arduous. Many cases are multiday efforts with large areas of excision.
“These can be very difficult wounds to manage even in a hospital. It’s resource-intense surgery,” Dr. Damavandy said.
The mean margin necessary to clear the lesions in this cohort was 2.6 cm. A 4-cm margin cleared almost 80%; 10% required a 7-cm margin.
“If we were going to make a recommendation to a nonMohs surgeon, it would be that 7 cm are needed to clear 98% of the tumors. That seems to me not reasonable for these anatomically complex and delicate regions. At the same time, the most commonly reported margin in the literature is 2 cm. In this cohort that would only have cleared 40% of the tumors.”
A mean of 3.4 Mohs stages were needed to obtain clear margins, although the range was wide (1-14 stages). Surgery took a mean of 1.5 days (range 1-5 days). Four patients could not be cleared and needed multidisciplinary care. These included two with vaginal invasion and two with invasion of the anal canal.
By 5 years, the overall tumor-free survival rate was 97%; the tumor-free survival rate was slightly better for primary than recurrent tumors (98% vs. 95%). When these rates were compared with Mohs without CK-7 staining, “primary Mohs doesn’t compare very well,” Dr. Damavandy said. At 5 years, the literature-reported recurrence rate for primary tumors treated with Mohs alone was 16% vs. Dr. Damavandy’s report of 2.3% for Mohs plus CK-7 staining. For recurrent tumors, the recurrence rates were 50% vs. 5.3%.
Two tumors recurred; the mean time to recurrence was 31 months. One recurrence was of a primary tumor treated by peripheral Mohs surgery, which was an uncommon approach in this cohort. This patient had a deep recurrence at 16 months.
The other case was a recurrence of a vulvar tumor that extended into the vaginal epithelium and across the cervix. Despite a vaginectomy and vaginal hysterectomy with clear margins, the patient had a recurrence at 46 months.
The patients were retreated and at last follow-up were free of disease.
Dr. Damavandy had no financial disclosures.
ORLANDO – Mohs surgery with cytokeratin-7 immunohistochemistry staining effected complete removal of extramammary Paget disease and resulted in a 5-year, 95% recurrence-free cure rate.
The results are significantly better than the often-cited 77% cure rate seen with Mohs surgery alone, Dr. Ali Alexander Damavandy said at the annual meeting of the American College of Mohs Surgery.
“These are statistically significant and clinically substantial results,” said Dr. Damavandy, a procedural dermatology fellow at the University of Pennsylvania, Philadelphia. “With this method you can tell a patient that in 5 years, he has a 95% chance of still not having the tumor. The high recurrence-free rate we have seen supports the view that Mohs surgery with cytokeratin-7 [CK-7] immunohistochemistry should be considered the curative treatment of choice for both primary and recurrent extramammary Paget disease of the skin.”
Dr. Damavandy’s retrospective review of 49 cases may seem small, he said, but it is “far and away the biggest cohort that exists, and the only substantial cohort with immunohistochemistry staining of CK-7.” The 5-year follow-up is also a strength of the study, he added.
Extramammary Paget disease (EMPD) remains an “extraordinarily hard tumor to treat. It typically presents with widespread intraepithelial islands of disease that are not clinically apparent. Local recurrence rates range from 33% to 60%, “leaving a lot of room for improvement,” Dr. Damavandy said.
“The explanations for recurrence are twofold,” he added. “First there is difficulty visualizing tumor cells in frozen sections. Second, it is a multifocal tumor that grows in a discontinuous fashion.”
Although one simply has to deal with the tumor spread, improvements can be made in reading the histopathology. Immunohistochemistry staining with CK-7 allows highly accurate visualization of individual Paget tumor cells, which express a very high level of the protein.
“Our hypothesis was that combining this improved tumor cell visualization with Mohs total excision would really make a dent in the recurrence rates of this disease,” Dr. Damavandy said.
He conducted a review of patients who were treated at four practices from 2004 to 2012. The cohort comprised 49 patients with 62 tumors; 43 were primary and the rest were recurrences. The majority (90%) occurred in the urogenital and anogenital region; the rest were axial or facial. All were treated with Mohs microsurgery enhanced by CK-7 staining of frozen sections. Recurrences were biopsy proven.
The typical Mohs technique for EMPD is to delineate a tumor-free area peripherally, and then excise the central tumor and tumor-bearing islands. The procedure can be long and arduous. Many cases are multiday efforts with large areas of excision.
“These can be very difficult wounds to manage even in a hospital. It’s resource-intense surgery,” Dr. Damavandy said.
The mean margin necessary to clear the lesions in this cohort was 2.6 cm. A 4-cm margin cleared almost 80%; 10% required a 7-cm margin.
“If we were going to make a recommendation to a nonMohs surgeon, it would be that 7 cm are needed to clear 98% of the tumors. That seems to me not reasonable for these anatomically complex and delicate regions. At the same time, the most commonly reported margin in the literature is 2 cm. In this cohort that would only have cleared 40% of the tumors.”
A mean of 3.4 Mohs stages were needed to obtain clear margins, although the range was wide (1-14 stages). Surgery took a mean of 1.5 days (range 1-5 days). Four patients could not be cleared and needed multidisciplinary care. These included two with vaginal invasion and two with invasion of the anal canal.
By 5 years, the overall tumor-free survival rate was 97%; the tumor-free survival rate was slightly better for primary than recurrent tumors (98% vs. 95%). When these rates were compared with Mohs without CK-7 staining, “primary Mohs doesn’t compare very well,” Dr. Damavandy said. At 5 years, the literature-reported recurrence rate for primary tumors treated with Mohs alone was 16% vs. Dr. Damavandy’s report of 2.3% for Mohs plus CK-7 staining. For recurrent tumors, the recurrence rates were 50% vs. 5.3%.
Two tumors recurred; the mean time to recurrence was 31 months. One recurrence was of a primary tumor treated by peripheral Mohs surgery, which was an uncommon approach in this cohort. This patient had a deep recurrence at 16 months.
The other case was a recurrence of a vulvar tumor that extended into the vaginal epithelium and across the cervix. Despite a vaginectomy and vaginal hysterectomy with clear margins, the patient had a recurrence at 46 months.
The patients were retreated and at last follow-up were free of disease.
Dr. Damavandy had no financial disclosures.
AT THE ACMS ANNUAL MEETING
Key clinical point: Mohs surgery enhanced with cytokeratin-7 immunohistochemistry appears to be very effective for extamammaory Paget disease.
Major finding: The 5-year recurrence rate was less than 5%.
Data source: The case review comprised 49 patients with 62 tumors.
Disclosures: Dr. Damavandy had no financial disclosures
Detecting morphologically difficult-to-diagnose melanomas
WAIKOLOA, HAWAII – Nodular melanoma is one of a handful of morphologic subtypes of melanoma that are difficult to diagnose in a timely way because they lack the conventional features associated with melanoma. But certain clinical and dermoscopic clues are helpful in avoiding misdiagnosis, Dr. Ashfaq A. Marghoob said at the Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
He provided tips on how to recognize nodular melanoma and other difficult-to-diagnose morphologic subtypes, including desmoplastic, amelanotic, nevoid, and epidermotropic metastatic melanoma.
These diagnostically challenging melanoma subtypes have several things in common: clinically, they don’t look like melanomas, and dermoscopically, they display specific tell-tale features that speak of malignancy, including atypical polymorphous blood vessels and crystalline structures. Dermoscopy can be a big help in deciding whether to biopsy a clinically unremarkable-looking lesion, observed Dr. Marghoob of Memorial Sloan Kettering Cancer Center in New York.
Here’s the rundown on how to avoid delayed diagnosis of these melanomas:
Nodular melanoma: The big challenge posed by nodular melanoma is that it grows at a very high rate, a median of 0.5 mm per month, “so it’s really not a subtype of melanoma that’s amenable to screening,” the dermatologist said.
“Think about this: A patient comes in for screening today and you see nothing on their skin. Then a month from today they start a nodular melanoma. Their next appointment with you is a year from now. By the time they come back, that nodular melanoma is going to be a life-threatening cancer,” he explained. “It requires patient engagement in self-examination to bring these lesions to our attention, and we have to be perceptive enough to see these patients in a timely manner when they call. Otherwise these are melanomas that are really the killer tumors.”
Nodular melanomas often lack the “ABCD” features associated with most melanomas: asymmetry, border irregularity, color, and diameter greater than one-quarter of an inch. Instead, the lesions may exhibit the modified ABCDs described in pediatric melanoma: amelanotic, bump showing symmetry, color homogeneity in a shade of either pink or blue-black, and de novo, meaning the lesion didn’t arise in association with a nevus, Dr. Marghoob continued.
Roughly 60% of nodular melanomas are pigmented, the other 40% amelanotic. Dermoscopically, pigmented nodular melanomas are characterized by a greater degree of symmetry than that seen with other melanomas. Atypical vessels that exhibit tortuosity, a cork screw pattern, or an irregular hairpin shape are prominently visible. The lesions are multicolored, often blue-black, and may exhibit a blue-white veil. Notably absent are regression structures, streaks, and networks.
An amelanotic nodular melanoma also features prominent atypical vessels, symmetry, and sometimes crystalline structures, which appear as shiny white lines. Comma vessels and arborizing vessels are absent.
A more detailed description of the dermoscopic findings in nodular melanoma is available in a study coauthored by Dr. Marghoob (JAMA Dermatol. 2013;149[6]:699-709).
Desmoplastic melanoma: Seventy percent of desmoplastic melanomas are amelanotic. They often present as an unremarkable firm subcutaneous papule or nodule. Indeed, desmoplastic melanomas often have such a banal clinical appearance that in one-third of cases, melanoma is not even in the differential diagnosis.
“You’re thinking cyst, lipoma, dermatofibroma ... something other than a malignancy,” Dr. Marghoob said.
Suspicion is raised by a benign-looking lesion that’s symptomatic and/or exhibiting growth on chronically sun-damaged skin in an older patient. A scar with no history of trauma that’s located on chronically sun-damaged skin is another tipoff. Also, desmoplastic melanoma can arise in association with a lentigo maligna component.
“It is this knowledge of the association between lentigo maligna and desmoplastic melanoma that for the first time has enabled us to diagnose desmoplastic melanomas that are thin – not 5 or 10 mm, but more like 0.6 mm in thickness,” Dr. Marghoob said.
The main dermoscopic clue is the presence of atypical vascular structures.
For a fuller account of the dermoscopic characteristics of desmoplastic melanomas, Dr. Marghoob recommended a study he coauthored (JAMA Dermatol. 2013;149[4]:413-21).
Amelanotic melanoma: An indication of how difficult these cancers are to diagnose was provided by the international Genes, Environment, and Melanoma Study of nearly 3,500 invasive melanomas, 8% of which were amelanotic. They were at a more advanced tumor stage at diagnosis. As a result, the mortality risk during a median 7.6 years of follow-up was an adjusted twofold greater for patients with amelanotic as compared with pigmented melanomas (JAMA Dermatol. 2014;150[12]:1306-314).
Dr. Marghoob coauthored a retrospective review of 20 consecutively diagnosed amelanotic melanomas. The lesions lacked any of the classic ABCD features. Most were symmetric, oval to round in shape, had regular borders, and were pink in color. Dermoscopically, a consistent finding was a polymorphous vascular pattern (J Eur Acad Dermatol Venereol. 2012;26[5]:591-6).
In an earlier five-continent study comparing dermoscopic features of 222 amelanotic melanomas, 105 pigmented melanomas, and 170 benign melanocytic lesions, investigators came up with a simple model that distinguished melanomas from nonmelanomas with 70% sensitivity and 56% specificity. The take-home message: Amelanotic melanomas often feature more than one shade of pink upon dermoscopic examination, along with dotted and linear irregular vessels and predominant central vessels (Arch Dermatol. 2008;144[9]:1120-7).
Nevoid melanoma: Clinically they often resemble an intradermal nevus, and they’re often amelanotic. Dermoscopically, however, an intradermal nevus displays a mammillated surface, with each individual mammillated area being associated with an isolated comma or hairpin vessel. In contrast, the surface of a nevoid melanoma is more undulating in appearance and lacks the classic vascular pattern seen in intradermal nevi. If there’s pigmentation present in a nevoid melanoma it will be distributed heterogeneously. The dermoscopic finding of irregular blood vessels and/or crystalline structures tips the balance in favor of biopsy.
Dr. Marghoob referred dermatologists interested in the dermoscopic features of nevoid melanoma to a recent study by members of the International Dermoscopy Society; he was one of the authors (Br J Dermatol. 2015;172[4]:961-7).
Epidermotropic metastatic melanoma: These lesions lack the ABCDs, are often amelanotic, and look like nondescript papules. If such a lesion is new, especially in a patient with a history of melanoma, think epidermotropic metastatic melanoma.
Spanish dermatologists have described five different dermoscopic patterns of melanoma metastases in an informative retrospective study: blue nevus-like, angioma-like, nevus-like, vascular, and unspecific. The vascular type is the most common form, characterized by amelanotic papules 2-3 mm in diameter and tortuous corkscrew vessels within the lesions (Br J Dermatol. 2013;169[1]:91-9).
Dr. Marghoob reported having no financial conflicts of interest regarding his presentation. SDEF and this news organization are owned by the same parent company.
WAIKOLOA, HAWAII – Nodular melanoma is one of a handful of morphologic subtypes of melanoma that are difficult to diagnose in a timely way because they lack the conventional features associated with melanoma. But certain clinical and dermoscopic clues are helpful in avoiding misdiagnosis, Dr. Ashfaq A. Marghoob said at the Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
He provided tips on how to recognize nodular melanoma and other difficult-to-diagnose morphologic subtypes, including desmoplastic, amelanotic, nevoid, and epidermotropic metastatic melanoma.
These diagnostically challenging melanoma subtypes have several things in common: clinically, they don’t look like melanomas, and dermoscopically, they display specific tell-tale features that speak of malignancy, including atypical polymorphous blood vessels and crystalline structures. Dermoscopy can be a big help in deciding whether to biopsy a clinically unremarkable-looking lesion, observed Dr. Marghoob of Memorial Sloan Kettering Cancer Center in New York.
Here’s the rundown on how to avoid delayed diagnosis of these melanomas:
Nodular melanoma: The big challenge posed by nodular melanoma is that it grows at a very high rate, a median of 0.5 mm per month, “so it’s really not a subtype of melanoma that’s amenable to screening,” the dermatologist said.
“Think about this: A patient comes in for screening today and you see nothing on their skin. Then a month from today they start a nodular melanoma. Their next appointment with you is a year from now. By the time they come back, that nodular melanoma is going to be a life-threatening cancer,” he explained. “It requires patient engagement in self-examination to bring these lesions to our attention, and we have to be perceptive enough to see these patients in a timely manner when they call. Otherwise these are melanomas that are really the killer tumors.”
Nodular melanomas often lack the “ABCD” features associated with most melanomas: asymmetry, border irregularity, color, and diameter greater than one-quarter of an inch. Instead, the lesions may exhibit the modified ABCDs described in pediatric melanoma: amelanotic, bump showing symmetry, color homogeneity in a shade of either pink or blue-black, and de novo, meaning the lesion didn’t arise in association with a nevus, Dr. Marghoob continued.
Roughly 60% of nodular melanomas are pigmented, the other 40% amelanotic. Dermoscopically, pigmented nodular melanomas are characterized by a greater degree of symmetry than that seen with other melanomas. Atypical vessels that exhibit tortuosity, a cork screw pattern, or an irregular hairpin shape are prominently visible. The lesions are multicolored, often blue-black, and may exhibit a blue-white veil. Notably absent are regression structures, streaks, and networks.
An amelanotic nodular melanoma also features prominent atypical vessels, symmetry, and sometimes crystalline structures, which appear as shiny white lines. Comma vessels and arborizing vessels are absent.
A more detailed description of the dermoscopic findings in nodular melanoma is available in a study coauthored by Dr. Marghoob (JAMA Dermatol. 2013;149[6]:699-709).
Desmoplastic melanoma: Seventy percent of desmoplastic melanomas are amelanotic. They often present as an unremarkable firm subcutaneous papule or nodule. Indeed, desmoplastic melanomas often have such a banal clinical appearance that in one-third of cases, melanoma is not even in the differential diagnosis.
“You’re thinking cyst, lipoma, dermatofibroma ... something other than a malignancy,” Dr. Marghoob said.
Suspicion is raised by a benign-looking lesion that’s symptomatic and/or exhibiting growth on chronically sun-damaged skin in an older patient. A scar with no history of trauma that’s located on chronically sun-damaged skin is another tipoff. Also, desmoplastic melanoma can arise in association with a lentigo maligna component.
“It is this knowledge of the association between lentigo maligna and desmoplastic melanoma that for the first time has enabled us to diagnose desmoplastic melanomas that are thin – not 5 or 10 mm, but more like 0.6 mm in thickness,” Dr. Marghoob said.
The main dermoscopic clue is the presence of atypical vascular structures.
For a fuller account of the dermoscopic characteristics of desmoplastic melanomas, Dr. Marghoob recommended a study he coauthored (JAMA Dermatol. 2013;149[4]:413-21).
Amelanotic melanoma: An indication of how difficult these cancers are to diagnose was provided by the international Genes, Environment, and Melanoma Study of nearly 3,500 invasive melanomas, 8% of which were amelanotic. They were at a more advanced tumor stage at diagnosis. As a result, the mortality risk during a median 7.6 years of follow-up was an adjusted twofold greater for patients with amelanotic as compared with pigmented melanomas (JAMA Dermatol. 2014;150[12]:1306-314).
Dr. Marghoob coauthored a retrospective review of 20 consecutively diagnosed amelanotic melanomas. The lesions lacked any of the classic ABCD features. Most were symmetric, oval to round in shape, had regular borders, and were pink in color. Dermoscopically, a consistent finding was a polymorphous vascular pattern (J Eur Acad Dermatol Venereol. 2012;26[5]:591-6).
In an earlier five-continent study comparing dermoscopic features of 222 amelanotic melanomas, 105 pigmented melanomas, and 170 benign melanocytic lesions, investigators came up with a simple model that distinguished melanomas from nonmelanomas with 70% sensitivity and 56% specificity. The take-home message: Amelanotic melanomas often feature more than one shade of pink upon dermoscopic examination, along with dotted and linear irregular vessels and predominant central vessels (Arch Dermatol. 2008;144[9]:1120-7).
Nevoid melanoma: Clinically they often resemble an intradermal nevus, and they’re often amelanotic. Dermoscopically, however, an intradermal nevus displays a mammillated surface, with each individual mammillated area being associated with an isolated comma or hairpin vessel. In contrast, the surface of a nevoid melanoma is more undulating in appearance and lacks the classic vascular pattern seen in intradermal nevi. If there’s pigmentation present in a nevoid melanoma it will be distributed heterogeneously. The dermoscopic finding of irregular blood vessels and/or crystalline structures tips the balance in favor of biopsy.
Dr. Marghoob referred dermatologists interested in the dermoscopic features of nevoid melanoma to a recent study by members of the International Dermoscopy Society; he was one of the authors (Br J Dermatol. 2015;172[4]:961-7).
Epidermotropic metastatic melanoma: These lesions lack the ABCDs, are often amelanotic, and look like nondescript papules. If such a lesion is new, especially in a patient with a history of melanoma, think epidermotropic metastatic melanoma.
Spanish dermatologists have described five different dermoscopic patterns of melanoma metastases in an informative retrospective study: blue nevus-like, angioma-like, nevus-like, vascular, and unspecific. The vascular type is the most common form, characterized by amelanotic papules 2-3 mm in diameter and tortuous corkscrew vessels within the lesions (Br J Dermatol. 2013;169[1]:91-9).
Dr. Marghoob reported having no financial conflicts of interest regarding his presentation. SDEF and this news organization are owned by the same parent company.
WAIKOLOA, HAWAII – Nodular melanoma is one of a handful of morphologic subtypes of melanoma that are difficult to diagnose in a timely way because they lack the conventional features associated with melanoma. But certain clinical and dermoscopic clues are helpful in avoiding misdiagnosis, Dr. Ashfaq A. Marghoob said at the Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
He provided tips on how to recognize nodular melanoma and other difficult-to-diagnose morphologic subtypes, including desmoplastic, amelanotic, nevoid, and epidermotropic metastatic melanoma.
These diagnostically challenging melanoma subtypes have several things in common: clinically, they don’t look like melanomas, and dermoscopically, they display specific tell-tale features that speak of malignancy, including atypical polymorphous blood vessels and crystalline structures. Dermoscopy can be a big help in deciding whether to biopsy a clinically unremarkable-looking lesion, observed Dr. Marghoob of Memorial Sloan Kettering Cancer Center in New York.
Here’s the rundown on how to avoid delayed diagnosis of these melanomas:
Nodular melanoma: The big challenge posed by nodular melanoma is that it grows at a very high rate, a median of 0.5 mm per month, “so it’s really not a subtype of melanoma that’s amenable to screening,” the dermatologist said.
“Think about this: A patient comes in for screening today and you see nothing on their skin. Then a month from today they start a nodular melanoma. Their next appointment with you is a year from now. By the time they come back, that nodular melanoma is going to be a life-threatening cancer,” he explained. “It requires patient engagement in self-examination to bring these lesions to our attention, and we have to be perceptive enough to see these patients in a timely manner when they call. Otherwise these are melanomas that are really the killer tumors.”
Nodular melanomas often lack the “ABCD” features associated with most melanomas: asymmetry, border irregularity, color, and diameter greater than one-quarter of an inch. Instead, the lesions may exhibit the modified ABCDs described in pediatric melanoma: amelanotic, bump showing symmetry, color homogeneity in a shade of either pink or blue-black, and de novo, meaning the lesion didn’t arise in association with a nevus, Dr. Marghoob continued.
Roughly 60% of nodular melanomas are pigmented, the other 40% amelanotic. Dermoscopically, pigmented nodular melanomas are characterized by a greater degree of symmetry than that seen with other melanomas. Atypical vessels that exhibit tortuosity, a cork screw pattern, or an irregular hairpin shape are prominently visible. The lesions are multicolored, often blue-black, and may exhibit a blue-white veil. Notably absent are regression structures, streaks, and networks.
An amelanotic nodular melanoma also features prominent atypical vessels, symmetry, and sometimes crystalline structures, which appear as shiny white lines. Comma vessels and arborizing vessels are absent.
A more detailed description of the dermoscopic findings in nodular melanoma is available in a study coauthored by Dr. Marghoob (JAMA Dermatol. 2013;149[6]:699-709).
Desmoplastic melanoma: Seventy percent of desmoplastic melanomas are amelanotic. They often present as an unremarkable firm subcutaneous papule or nodule. Indeed, desmoplastic melanomas often have such a banal clinical appearance that in one-third of cases, melanoma is not even in the differential diagnosis.
“You’re thinking cyst, lipoma, dermatofibroma ... something other than a malignancy,” Dr. Marghoob said.
Suspicion is raised by a benign-looking lesion that’s symptomatic and/or exhibiting growth on chronically sun-damaged skin in an older patient. A scar with no history of trauma that’s located on chronically sun-damaged skin is another tipoff. Also, desmoplastic melanoma can arise in association with a lentigo maligna component.
“It is this knowledge of the association between lentigo maligna and desmoplastic melanoma that for the first time has enabled us to diagnose desmoplastic melanomas that are thin – not 5 or 10 mm, but more like 0.6 mm in thickness,” Dr. Marghoob said.
The main dermoscopic clue is the presence of atypical vascular structures.
For a fuller account of the dermoscopic characteristics of desmoplastic melanomas, Dr. Marghoob recommended a study he coauthored (JAMA Dermatol. 2013;149[4]:413-21).
Amelanotic melanoma: An indication of how difficult these cancers are to diagnose was provided by the international Genes, Environment, and Melanoma Study of nearly 3,500 invasive melanomas, 8% of which were amelanotic. They were at a more advanced tumor stage at diagnosis. As a result, the mortality risk during a median 7.6 years of follow-up was an adjusted twofold greater for patients with amelanotic as compared with pigmented melanomas (JAMA Dermatol. 2014;150[12]:1306-314).
Dr. Marghoob coauthored a retrospective review of 20 consecutively diagnosed amelanotic melanomas. The lesions lacked any of the classic ABCD features. Most were symmetric, oval to round in shape, had regular borders, and were pink in color. Dermoscopically, a consistent finding was a polymorphous vascular pattern (J Eur Acad Dermatol Venereol. 2012;26[5]:591-6).
In an earlier five-continent study comparing dermoscopic features of 222 amelanotic melanomas, 105 pigmented melanomas, and 170 benign melanocytic lesions, investigators came up with a simple model that distinguished melanomas from nonmelanomas with 70% sensitivity and 56% specificity. The take-home message: Amelanotic melanomas often feature more than one shade of pink upon dermoscopic examination, along with dotted and linear irregular vessels and predominant central vessels (Arch Dermatol. 2008;144[9]:1120-7).
Nevoid melanoma: Clinically they often resemble an intradermal nevus, and they’re often amelanotic. Dermoscopically, however, an intradermal nevus displays a mammillated surface, with each individual mammillated area being associated with an isolated comma or hairpin vessel. In contrast, the surface of a nevoid melanoma is more undulating in appearance and lacks the classic vascular pattern seen in intradermal nevi. If there’s pigmentation present in a nevoid melanoma it will be distributed heterogeneously. The dermoscopic finding of irregular blood vessels and/or crystalline structures tips the balance in favor of biopsy.
Dr. Marghoob referred dermatologists interested in the dermoscopic features of nevoid melanoma to a recent study by members of the International Dermoscopy Society; he was one of the authors (Br J Dermatol. 2015;172[4]:961-7).
Epidermotropic metastatic melanoma: These lesions lack the ABCDs, are often amelanotic, and look like nondescript papules. If such a lesion is new, especially in a patient with a history of melanoma, think epidermotropic metastatic melanoma.
Spanish dermatologists have described five different dermoscopic patterns of melanoma metastases in an informative retrospective study: blue nevus-like, angioma-like, nevus-like, vascular, and unspecific. The vascular type is the most common form, characterized by amelanotic papules 2-3 mm in diameter and tortuous corkscrew vessels within the lesions (Br J Dermatol. 2013;169[1]:91-9).
Dr. Marghoob reported having no financial conflicts of interest regarding his presentation. SDEF and this news organization are owned by the same parent company.
EXPERT ANALYSIS FROM SDEF HAWAII DERMATOLOGY SEMINAR
Patient-related factors are key to nevus suspiciousness
WAIKOLOA, HAWAII – Some melanomas can be diagnosed from the examination room doorway. Others are evident only upon close visual inspection. But the most challenging cases require integration of information regarding the lesion’s dermoscopic pattern in the context of the clues provided by key patient-specific characteristics, according to Dr. Michael A. Marchetti.
“Consider a melanoma diagnosis when a lesion is wrong for the patient’s age, the anatomic location, or skin type, or if it deviates from the patient’s expected signature pattern of nevi,” noted Dr. Marchetti, a dermatologist at Memorial Sloan-Kettering Cancer Center in New York.
A landmark study that provides guidance in this area was conducted several years ago at dermatology centers in four European countries and at Memorial Sloan-Kettering. The investigators enrolled 480 consecutive children and adults with a total of 5,481 melanocytic nevi. The study showcased significant age- and anatomic site–related differences in the distribution of nevi categorized according to dermoscopic subgroups (Arch Dermatol. 2011;147[6]:663-70).
For example, nevi characterized dermoscopically by a peripheral rim of globules were found to be vastly more common during the first 3 decades of life than later. Based upon this study and other data, Dr. Marchetti’s suggested management strategy for peripheral globular nevus with no other concerning features in patients up to age 30 is reassurance that the lesion is not going to be a problem and doesn’t require careful monitoring or biopsy. In a patient beyond age 30, however, frequent monitoring or biopsy is appropriate – and the older the patient with a peripheral globular nevus, the lower the threshold for biopsy.
A peripheral globular nevus is typically a nevus in its radial growth phase, which can continue for years before the nevus enters senescence. The same is true of a nevus that exhibits a Spitzoid starbust pattern on dermoscopy. It is growing radially, and the pretest probability that it’s malignant is highly age dependent.
“In a child, it’s very likely to be a Spitz nevus. In an adult, it’s very likely to be a melanoma. Based upon morphology alone, you really can’t make a distinction between these lesions,” Dr. Marchetti said.
The importance of age in differentiating these starburst lesions was underscored in a recent Italian study involving 384 symmetric, dermoscopically Spitzoid-looking lesions in patients aged 12 or older. Histopathologically, 13.3% of the lesions were diagnosed as melanoma. The probability increased with advancing age, reaching 50% or more after age 50 years. The investigators concluded that the only safe strategy to avoid missing a melanoma is to excise all Spitzoid-looking lesions in patients aged 12 years or older (J Am Acad Dermatol. 2015;72[1]:47-53).
“That concurs with our practice at Memorial Sloan-Kettering. We tend to biopsy all lesions with this particular morphology because you just can’t tell,” Dr. Marchetti said at the Hawaii Dermatology Seminar provided by Global Academy for Medical Education/Skin Disease Education Foundation.
Nevi with a globular dermoscopic pattern occur mostly in children and mainly on the upper back. A globular lesion that develops in an adult or on an extremity is “wrong,” and therefore biopsy should be seriously considered.
In contrast, nevi classified dermoscopically as reticular occur on the trunk and extremities with similar frequency, he continued.
Age and anatomic location are also important considerations in deciding whether a lesion might be a case of nested melanoma of the elderly. This is a form of melanoma first described only a few years ago. It takes the form of a large flat pigmented lesion showing a dermoscopic pattern of irregular globules throughout in a patient over aged 60 years. Notably, three-quarters of cases of nested melanoma of the elderly occur on the extremities (JAMA Dermatol. 2013;149[8]:941-5).
Skin type can raise or diminish the concern that a pigmented lesion is a melanoma. Patients with a lighter skin type tend to have light brown nevi with a patchy reticular network and central hypopigmentation; dark nevi are relatively uncommon in lighter-skinned patients and thus stand out as suspicious outliers. The flip side is also true: Patients with darker skin types tend to have dark nevi with central hyperpigmentation.
A nevus that deviates from a patient’s signature pattern is often referred to as the ugly duckling sign of melanoma.
“It’s the context provided by the background nevi which informs you if there’s an outlier. This can be helpful not only in detecting melanoma, but also in reducing unnecessary biopsies. If a lesion you’re wondering about looks dermoscopically like the patient’s other nevi, that’s reassuring,” Dr. Marchetti said.
He reported having no financial conflicts regarding his presentation. SDEF and this news organization are owned by the same parent company.
WAIKOLOA, HAWAII – Some melanomas can be diagnosed from the examination room doorway. Others are evident only upon close visual inspection. But the most challenging cases require integration of information regarding the lesion’s dermoscopic pattern in the context of the clues provided by key patient-specific characteristics, according to Dr. Michael A. Marchetti.
“Consider a melanoma diagnosis when a lesion is wrong for the patient’s age, the anatomic location, or skin type, or if it deviates from the patient’s expected signature pattern of nevi,” noted Dr. Marchetti, a dermatologist at Memorial Sloan-Kettering Cancer Center in New York.
A landmark study that provides guidance in this area was conducted several years ago at dermatology centers in four European countries and at Memorial Sloan-Kettering. The investigators enrolled 480 consecutive children and adults with a total of 5,481 melanocytic nevi. The study showcased significant age- and anatomic site–related differences in the distribution of nevi categorized according to dermoscopic subgroups (Arch Dermatol. 2011;147[6]:663-70).
For example, nevi characterized dermoscopically by a peripheral rim of globules were found to be vastly more common during the first 3 decades of life than later. Based upon this study and other data, Dr. Marchetti’s suggested management strategy for peripheral globular nevus with no other concerning features in patients up to age 30 is reassurance that the lesion is not going to be a problem and doesn’t require careful monitoring or biopsy. In a patient beyond age 30, however, frequent monitoring or biopsy is appropriate – and the older the patient with a peripheral globular nevus, the lower the threshold for biopsy.
A peripheral globular nevus is typically a nevus in its radial growth phase, which can continue for years before the nevus enters senescence. The same is true of a nevus that exhibits a Spitzoid starbust pattern on dermoscopy. It is growing radially, and the pretest probability that it’s malignant is highly age dependent.
“In a child, it’s very likely to be a Spitz nevus. In an adult, it’s very likely to be a melanoma. Based upon morphology alone, you really can’t make a distinction between these lesions,” Dr. Marchetti said.
The importance of age in differentiating these starburst lesions was underscored in a recent Italian study involving 384 symmetric, dermoscopically Spitzoid-looking lesions in patients aged 12 or older. Histopathologically, 13.3% of the lesions were diagnosed as melanoma. The probability increased with advancing age, reaching 50% or more after age 50 years. The investigators concluded that the only safe strategy to avoid missing a melanoma is to excise all Spitzoid-looking lesions in patients aged 12 years or older (J Am Acad Dermatol. 2015;72[1]:47-53).
“That concurs with our practice at Memorial Sloan-Kettering. We tend to biopsy all lesions with this particular morphology because you just can’t tell,” Dr. Marchetti said at the Hawaii Dermatology Seminar provided by Global Academy for Medical Education/Skin Disease Education Foundation.
Nevi with a globular dermoscopic pattern occur mostly in children and mainly on the upper back. A globular lesion that develops in an adult or on an extremity is “wrong,” and therefore biopsy should be seriously considered.
In contrast, nevi classified dermoscopically as reticular occur on the trunk and extremities with similar frequency, he continued.
Age and anatomic location are also important considerations in deciding whether a lesion might be a case of nested melanoma of the elderly. This is a form of melanoma first described only a few years ago. It takes the form of a large flat pigmented lesion showing a dermoscopic pattern of irregular globules throughout in a patient over aged 60 years. Notably, three-quarters of cases of nested melanoma of the elderly occur on the extremities (JAMA Dermatol. 2013;149[8]:941-5).
Skin type can raise or diminish the concern that a pigmented lesion is a melanoma. Patients with a lighter skin type tend to have light brown nevi with a patchy reticular network and central hypopigmentation; dark nevi are relatively uncommon in lighter-skinned patients and thus stand out as suspicious outliers. The flip side is also true: Patients with darker skin types tend to have dark nevi with central hyperpigmentation.
A nevus that deviates from a patient’s signature pattern is often referred to as the ugly duckling sign of melanoma.
“It’s the context provided by the background nevi which informs you if there’s an outlier. This can be helpful not only in detecting melanoma, but also in reducing unnecessary biopsies. If a lesion you’re wondering about looks dermoscopically like the patient’s other nevi, that’s reassuring,” Dr. Marchetti said.
He reported having no financial conflicts regarding his presentation. SDEF and this news organization are owned by the same parent company.
WAIKOLOA, HAWAII – Some melanomas can be diagnosed from the examination room doorway. Others are evident only upon close visual inspection. But the most challenging cases require integration of information regarding the lesion’s dermoscopic pattern in the context of the clues provided by key patient-specific characteristics, according to Dr. Michael A. Marchetti.
“Consider a melanoma diagnosis when a lesion is wrong for the patient’s age, the anatomic location, or skin type, or if it deviates from the patient’s expected signature pattern of nevi,” noted Dr. Marchetti, a dermatologist at Memorial Sloan-Kettering Cancer Center in New York.
A landmark study that provides guidance in this area was conducted several years ago at dermatology centers in four European countries and at Memorial Sloan-Kettering. The investigators enrolled 480 consecutive children and adults with a total of 5,481 melanocytic nevi. The study showcased significant age- and anatomic site–related differences in the distribution of nevi categorized according to dermoscopic subgroups (Arch Dermatol. 2011;147[6]:663-70).
For example, nevi characterized dermoscopically by a peripheral rim of globules were found to be vastly more common during the first 3 decades of life than later. Based upon this study and other data, Dr. Marchetti’s suggested management strategy for peripheral globular nevus with no other concerning features in patients up to age 30 is reassurance that the lesion is not going to be a problem and doesn’t require careful monitoring or biopsy. In a patient beyond age 30, however, frequent monitoring or biopsy is appropriate – and the older the patient with a peripheral globular nevus, the lower the threshold for biopsy.
A peripheral globular nevus is typically a nevus in its radial growth phase, which can continue for years before the nevus enters senescence. The same is true of a nevus that exhibits a Spitzoid starbust pattern on dermoscopy. It is growing radially, and the pretest probability that it’s malignant is highly age dependent.
“In a child, it’s very likely to be a Spitz nevus. In an adult, it’s very likely to be a melanoma. Based upon morphology alone, you really can’t make a distinction between these lesions,” Dr. Marchetti said.
The importance of age in differentiating these starburst lesions was underscored in a recent Italian study involving 384 symmetric, dermoscopically Spitzoid-looking lesions in patients aged 12 or older. Histopathologically, 13.3% of the lesions were diagnosed as melanoma. The probability increased with advancing age, reaching 50% or more after age 50 years. The investigators concluded that the only safe strategy to avoid missing a melanoma is to excise all Spitzoid-looking lesions in patients aged 12 years or older (J Am Acad Dermatol. 2015;72[1]:47-53).
“That concurs with our practice at Memorial Sloan-Kettering. We tend to biopsy all lesions with this particular morphology because you just can’t tell,” Dr. Marchetti said at the Hawaii Dermatology Seminar provided by Global Academy for Medical Education/Skin Disease Education Foundation.
Nevi with a globular dermoscopic pattern occur mostly in children and mainly on the upper back. A globular lesion that develops in an adult or on an extremity is “wrong,” and therefore biopsy should be seriously considered.
In contrast, nevi classified dermoscopically as reticular occur on the trunk and extremities with similar frequency, he continued.
Age and anatomic location are also important considerations in deciding whether a lesion might be a case of nested melanoma of the elderly. This is a form of melanoma first described only a few years ago. It takes the form of a large flat pigmented lesion showing a dermoscopic pattern of irregular globules throughout in a patient over aged 60 years. Notably, three-quarters of cases of nested melanoma of the elderly occur on the extremities (JAMA Dermatol. 2013;149[8]:941-5).
Skin type can raise or diminish the concern that a pigmented lesion is a melanoma. Patients with a lighter skin type tend to have light brown nevi with a patchy reticular network and central hypopigmentation; dark nevi are relatively uncommon in lighter-skinned patients and thus stand out as suspicious outliers. The flip side is also true: Patients with darker skin types tend to have dark nevi with central hyperpigmentation.
A nevus that deviates from a patient’s signature pattern is often referred to as the ugly duckling sign of melanoma.
“It’s the context provided by the background nevi which informs you if there’s an outlier. This can be helpful not only in detecting melanoma, but also in reducing unnecessary biopsies. If a lesion you’re wondering about looks dermoscopically like the patient’s other nevi, that’s reassuring,” Dr. Marchetti said.
He reported having no financial conflicts regarding his presentation. SDEF and this news organization are owned by the same parent company.
EXPERT ANALYSIS FROM SDEF HAWAII DERMATOLOGY SEMINAR
Merkel cell carcinoma responds to first-line pembrolizumab
First-line therapy with pembrolizumab is associated with frequent and durable responses in patients with advanced Merkel cell carcinoma (MCC), according to a study presented at the annual meeting of the American Association for Cancer Research.
The objective response rate wad 56% among 25 patients with MCC who had received at least one dose of pembrolizumab in a phase II study, Dr. Paul Nghiem said at a press conference during the meeting.
This rare but aggressive skin cancer has been notoriously hard to treat with typical chemotherapy of platinum compounds and etoposide, with half of tumors progressing by 3 months and 90% progressing by 10 months. MCC is about three times more likely to kill a patient compared with melanoma.
Given that about 40% of the 2,000 cases per year develop advanced disease, pembrolizumab has the potential to be an advance over chemotherapy although at this point no therapy is approved by the Food and Drug Administration for MCC advanced disease. The disease may be caused by ultraviolet light exposure in older individuals or with immune suppression. In about 80% of cases, Merkel cell polyomavirus (MCPyV) is present, but the virus is ubiquitous in the environment, with widespread exposure in the population without ill effects for most people.
Pembrolizumab acts to block the interaction of PD-1 on tumor-specific or MCPyV-specific T cells with its ligand, PD-L1, on tumor cells or antigen-presenting cells, thus allowing the T cells to recognize the tumors and enhance tumor cell killing.
In a phase II multicenter, single-arm, open-label trial, patients with unresectable or metastatic disease who had never before received any systemic therapy received pembrolizumab 2 mg/kg IV every 3 weeks for up to 2 years. Tumor evaluations were done every 9 or 12 weeks with a median follow-up of 7.6 months.
Of the 25 patients who had received at least one dose and had at least one radiological assessment, there was an objective response rate of 56% (14/25), including 4 complete, 10 partial, and 1 unconfirmed partial response. One patient had stable disease, and nine had progressive disease.
“By the first scan at 3 months most patients have found where they were going to go,” said Dr. Nghiem, professor of medicine in dermatology at the University of Washington, Seattle. “Several had failed early, but by the time of the first scan at 3 months there were often very profound and complete responses, and a lot of the partial responses just remain very steady over time.”
With the caveat that this was not a randomized trial, he said the progression-free survival (PFS) among responders was 67% at 6 months and the median PFS was 9 months, compared to a historical median overall survival with chemotherapy of 9.5 months after metastatic diagnosis. Among responders, 86% continue to have excellent disease control more than 6 months after starting therapy.
Objective responses in this small study appeared to be associated with positive viral status of the tumors. Among patients with virus-positive tumors, 62% (10/16) had objective responses vs. 44% (4/9) in whom the tumors were virus negative. “That was not a statistically significant difference … but maybe suggests, like head and neck [cancers], there may be a better story there for virus positive,” Dr. Nghiem said. PD-L1 expression in the tumors did not predict responses.
In response to a question during the news conference about the high response rate compared to responses to therapy of most other solid tumors, Dr. Nghiem pointed out, “Historically, this has been a very immune-associated cancer … If you had CD8 T cells infiltrating into this cancer at any reasonable number, among 300 patients not one died of the disease.” Unleashing the immune system in this trial through PD-1 blockade may therefore help to explain the good outcomes.
Adverse events were managed with corticosteroid treatment and discontinuing the drug. Two patients who had severe drug-related toxicities improved after steroids and stopping the drug. But, nonetheless, they have ongoing antitumor responses several months after stopping pembrolizumab.
The investigators are currently expanding the trial and recruiting more patients.
The study was simultaneously published online in the New England Journal of Medicine (2016 April 19. doi: 10.1056/NEJMoa1603702).
The study was supported in part by Merck. Dr. Nghiem is a consultant to EMD Serono and has grant/research support from Bristol-Myers Squibb.
First-line therapy with pembrolizumab is associated with frequent and durable responses in patients with advanced Merkel cell carcinoma (MCC), according to a study presented at the annual meeting of the American Association for Cancer Research.
The objective response rate wad 56% among 25 patients with MCC who had received at least one dose of pembrolizumab in a phase II study, Dr. Paul Nghiem said at a press conference during the meeting.
This rare but aggressive skin cancer has been notoriously hard to treat with typical chemotherapy of platinum compounds and etoposide, with half of tumors progressing by 3 months and 90% progressing by 10 months. MCC is about three times more likely to kill a patient compared with melanoma.
Given that about 40% of the 2,000 cases per year develop advanced disease, pembrolizumab has the potential to be an advance over chemotherapy although at this point no therapy is approved by the Food and Drug Administration for MCC advanced disease. The disease may be caused by ultraviolet light exposure in older individuals or with immune suppression. In about 80% of cases, Merkel cell polyomavirus (MCPyV) is present, but the virus is ubiquitous in the environment, with widespread exposure in the population without ill effects for most people.
Pembrolizumab acts to block the interaction of PD-1 on tumor-specific or MCPyV-specific T cells with its ligand, PD-L1, on tumor cells or antigen-presenting cells, thus allowing the T cells to recognize the tumors and enhance tumor cell killing.
In a phase II multicenter, single-arm, open-label trial, patients with unresectable or metastatic disease who had never before received any systemic therapy received pembrolizumab 2 mg/kg IV every 3 weeks for up to 2 years. Tumor evaluations were done every 9 or 12 weeks with a median follow-up of 7.6 months.
Of the 25 patients who had received at least one dose and had at least one radiological assessment, there was an objective response rate of 56% (14/25), including 4 complete, 10 partial, and 1 unconfirmed partial response. One patient had stable disease, and nine had progressive disease.
“By the first scan at 3 months most patients have found where they were going to go,” said Dr. Nghiem, professor of medicine in dermatology at the University of Washington, Seattle. “Several had failed early, but by the time of the first scan at 3 months there were often very profound and complete responses, and a lot of the partial responses just remain very steady over time.”
With the caveat that this was not a randomized trial, he said the progression-free survival (PFS) among responders was 67% at 6 months and the median PFS was 9 months, compared to a historical median overall survival with chemotherapy of 9.5 months after metastatic diagnosis. Among responders, 86% continue to have excellent disease control more than 6 months after starting therapy.
Objective responses in this small study appeared to be associated with positive viral status of the tumors. Among patients with virus-positive tumors, 62% (10/16) had objective responses vs. 44% (4/9) in whom the tumors were virus negative. “That was not a statistically significant difference … but maybe suggests, like head and neck [cancers], there may be a better story there for virus positive,” Dr. Nghiem said. PD-L1 expression in the tumors did not predict responses.
In response to a question during the news conference about the high response rate compared to responses to therapy of most other solid tumors, Dr. Nghiem pointed out, “Historically, this has been a very immune-associated cancer … If you had CD8 T cells infiltrating into this cancer at any reasonable number, among 300 patients not one died of the disease.” Unleashing the immune system in this trial through PD-1 blockade may therefore help to explain the good outcomes.
Adverse events were managed with corticosteroid treatment and discontinuing the drug. Two patients who had severe drug-related toxicities improved after steroids and stopping the drug. But, nonetheless, they have ongoing antitumor responses several months after stopping pembrolizumab.
The investigators are currently expanding the trial and recruiting more patients.
The study was simultaneously published online in the New England Journal of Medicine (2016 April 19. doi: 10.1056/NEJMoa1603702).
The study was supported in part by Merck. Dr. Nghiem is a consultant to EMD Serono and has grant/research support from Bristol-Myers Squibb.
First-line therapy with pembrolizumab is associated with frequent and durable responses in patients with advanced Merkel cell carcinoma (MCC), according to a study presented at the annual meeting of the American Association for Cancer Research.
The objective response rate wad 56% among 25 patients with MCC who had received at least one dose of pembrolizumab in a phase II study, Dr. Paul Nghiem said at a press conference during the meeting.
This rare but aggressive skin cancer has been notoriously hard to treat with typical chemotherapy of platinum compounds and etoposide, with half of tumors progressing by 3 months and 90% progressing by 10 months. MCC is about three times more likely to kill a patient compared with melanoma.
Given that about 40% of the 2,000 cases per year develop advanced disease, pembrolizumab has the potential to be an advance over chemotherapy although at this point no therapy is approved by the Food and Drug Administration for MCC advanced disease. The disease may be caused by ultraviolet light exposure in older individuals or with immune suppression. In about 80% of cases, Merkel cell polyomavirus (MCPyV) is present, but the virus is ubiquitous in the environment, with widespread exposure in the population without ill effects for most people.
Pembrolizumab acts to block the interaction of PD-1 on tumor-specific or MCPyV-specific T cells with its ligand, PD-L1, on tumor cells or antigen-presenting cells, thus allowing the T cells to recognize the tumors and enhance tumor cell killing.
In a phase II multicenter, single-arm, open-label trial, patients with unresectable or metastatic disease who had never before received any systemic therapy received pembrolizumab 2 mg/kg IV every 3 weeks for up to 2 years. Tumor evaluations were done every 9 or 12 weeks with a median follow-up of 7.6 months.
Of the 25 patients who had received at least one dose and had at least one radiological assessment, there was an objective response rate of 56% (14/25), including 4 complete, 10 partial, and 1 unconfirmed partial response. One patient had stable disease, and nine had progressive disease.
“By the first scan at 3 months most patients have found where they were going to go,” said Dr. Nghiem, professor of medicine in dermatology at the University of Washington, Seattle. “Several had failed early, but by the time of the first scan at 3 months there were often very profound and complete responses, and a lot of the partial responses just remain very steady over time.”
With the caveat that this was not a randomized trial, he said the progression-free survival (PFS) among responders was 67% at 6 months and the median PFS was 9 months, compared to a historical median overall survival with chemotherapy of 9.5 months after metastatic diagnosis. Among responders, 86% continue to have excellent disease control more than 6 months after starting therapy.
Objective responses in this small study appeared to be associated with positive viral status of the tumors. Among patients with virus-positive tumors, 62% (10/16) had objective responses vs. 44% (4/9) in whom the tumors were virus negative. “That was not a statistically significant difference … but maybe suggests, like head and neck [cancers], there may be a better story there for virus positive,” Dr. Nghiem said. PD-L1 expression in the tumors did not predict responses.
In response to a question during the news conference about the high response rate compared to responses to therapy of most other solid tumors, Dr. Nghiem pointed out, “Historically, this has been a very immune-associated cancer … If you had CD8 T cells infiltrating into this cancer at any reasonable number, among 300 patients not one died of the disease.” Unleashing the immune system in this trial through PD-1 blockade may therefore help to explain the good outcomes.
Adverse events were managed with corticosteroid treatment and discontinuing the drug. Two patients who had severe drug-related toxicities improved after steroids and stopping the drug. But, nonetheless, they have ongoing antitumor responses several months after stopping pembrolizumab.
The investigators are currently expanding the trial and recruiting more patients.
The study was simultaneously published online in the New England Journal of Medicine (2016 April 19. doi: 10.1056/NEJMoa1603702).
The study was supported in part by Merck. Dr. Nghiem is a consultant to EMD Serono and has grant/research support from Bristol-Myers Squibb.
FROM THE AACR ANNUAL MEETING
Key clinical point: Rare Merkel cell carcinoma responds well to immunotherapy with pembrolizumab.
Major finding: The response rate was 56%, all occurring by 3 months.
Data source: Phase II multicenter, single-arm, open-label trial of 25 patients (NCT02267603).
Disclosures: The study was supported in part by Merck. Dr. Nghiem is a consultant to EMD Serono and has grant/research support from Bristol-Myers Squibb.
Pembrolizumab: 33% response rate in advanced melanoma
Immunotherapy with the PD-1 inhibitor pembrolizumab yielded a 33% overall response rate, a 35% progression-free survival rate at 1 year, and a median overall survival of 23 months in 655 patients with advanced melanoma, according to a report published online in JAMA.
The agent generally was well tolerated, with grade 3 or 4 treatment-related toxicities occurring in 14% of patients. Only 4% of patients discontinued pembrolizumab because of adverse events, including colitis, pyrexia, pneumonitis, and thyroid abnormalities, said Dr. Antoni Ribas of University of California, Los Angeles, and his associates.
The investigators analyzed data pooled from 8 open-label phase I cohorts, some of which have been reported previously, in this manufacturer-sponsored study. All the participants were adults (median age, 61 years) with advanced unresectable melanoma who resided in Australia, Canada, France, and the U.S. This pooled study population was heterogeneous, as the various cohorts had different eligibility criteria; some patients were treatment-naïve, some had received ipilimumab, and some were treated concomitantly with BRAF or MEK inhibitors.
The estimated median duration of response was 28.2 months, and 44% of patients who responded to pembrolizumab had a response duration of longer than 1 year. Approximately half of the patients were still alive at 2 years.
Treatment benefit was consistent regardless of whether or not patients had previously received ipilimumab and regardless of the dose or schedule of pembrolizumab they were given. Patients who were treatment-naïve showed the greatest response to pembrolizumab: an overall response rate of 45%, a 1-year progression-free survival rate of 52%, and a median overall survival of 31 months, the investigators said (JAMA. 2016 Apr 19. doi: 10.1001/jama.2016.4059). “Collectively, these data suggest that the majority of patients with melanoma treated with pembrolizumab will experience lasting objective responses,” they added.
The encouraging results of Ribas et al add to the burgeoning literature on the potential of PD-1 blockade to improve outcomes in metastatic melanoma. One finding in particular – the fact that half of the patients in these cohorts were alive at 2 years – is noteworthy because the median overall survival in most previous clinical trials of the disease was consistently shorter than 1 year.
However, the results also highlight the limitations of PD-1 blockade. Median progression-free survival was only 4 months, and the 1-year progression-free survival rate was only 35%. This suggests that the host immune system fails to adequately control the malignancy in two-thirds of patients within the first year. And even among treatment responders, disease progression occurred in 26%.
Dr. Shailender Bhatia and Dr. John A. Thompson are at the University of Washington and the Fred Hutchinson Cancer Research Center, both in Seattle. Dr. Bhatia and Dr. Thompson made these remarks in an editorial accompanying Dr. Ribas’s report (JAMA. 2016 Apr 19;315:1573-4).
The encouraging results of Ribas et al add to the burgeoning literature on the potential of PD-1 blockade to improve outcomes in metastatic melanoma. One finding in particular – the fact that half of the patients in these cohorts were alive at 2 years – is noteworthy because the median overall survival in most previous clinical trials of the disease was consistently shorter than 1 year.
However, the results also highlight the limitations of PD-1 blockade. Median progression-free survival was only 4 months, and the 1-year progression-free survival rate was only 35%. This suggests that the host immune system fails to adequately control the malignancy in two-thirds of patients within the first year. And even among treatment responders, disease progression occurred in 26%.
Dr. Shailender Bhatia and Dr. John A. Thompson are at the University of Washington and the Fred Hutchinson Cancer Research Center, both in Seattle. Dr. Bhatia and Dr. Thompson made these remarks in an editorial accompanying Dr. Ribas’s report (JAMA. 2016 Apr 19;315:1573-4).
The encouraging results of Ribas et al add to the burgeoning literature on the potential of PD-1 blockade to improve outcomes in metastatic melanoma. One finding in particular – the fact that half of the patients in these cohorts were alive at 2 years – is noteworthy because the median overall survival in most previous clinical trials of the disease was consistently shorter than 1 year.
However, the results also highlight the limitations of PD-1 blockade. Median progression-free survival was only 4 months, and the 1-year progression-free survival rate was only 35%. This suggests that the host immune system fails to adequately control the malignancy in two-thirds of patients within the first year. And even among treatment responders, disease progression occurred in 26%.
Dr. Shailender Bhatia and Dr. John A. Thompson are at the University of Washington and the Fred Hutchinson Cancer Research Center, both in Seattle. Dr. Bhatia and Dr. Thompson made these remarks in an editorial accompanying Dr. Ribas’s report (JAMA. 2016 Apr 19;315:1573-4).
Immunotherapy with the PD-1 inhibitor pembrolizumab yielded a 33% overall response rate, a 35% progression-free survival rate at 1 year, and a median overall survival of 23 months in 655 patients with advanced melanoma, according to a report published online in JAMA.
The agent generally was well tolerated, with grade 3 or 4 treatment-related toxicities occurring in 14% of patients. Only 4% of patients discontinued pembrolizumab because of adverse events, including colitis, pyrexia, pneumonitis, and thyroid abnormalities, said Dr. Antoni Ribas of University of California, Los Angeles, and his associates.
The investigators analyzed data pooled from 8 open-label phase I cohorts, some of which have been reported previously, in this manufacturer-sponsored study. All the participants were adults (median age, 61 years) with advanced unresectable melanoma who resided in Australia, Canada, France, and the U.S. This pooled study population was heterogeneous, as the various cohorts had different eligibility criteria; some patients were treatment-naïve, some had received ipilimumab, and some were treated concomitantly with BRAF or MEK inhibitors.
The estimated median duration of response was 28.2 months, and 44% of patients who responded to pembrolizumab had a response duration of longer than 1 year. Approximately half of the patients were still alive at 2 years.
Treatment benefit was consistent regardless of whether or not patients had previously received ipilimumab and regardless of the dose or schedule of pembrolizumab they were given. Patients who were treatment-naïve showed the greatest response to pembrolizumab: an overall response rate of 45%, a 1-year progression-free survival rate of 52%, and a median overall survival of 31 months, the investigators said (JAMA. 2016 Apr 19. doi: 10.1001/jama.2016.4059). “Collectively, these data suggest that the majority of patients with melanoma treated with pembrolizumab will experience lasting objective responses,” they added.
Immunotherapy with the PD-1 inhibitor pembrolizumab yielded a 33% overall response rate, a 35% progression-free survival rate at 1 year, and a median overall survival of 23 months in 655 patients with advanced melanoma, according to a report published online in JAMA.
The agent generally was well tolerated, with grade 3 or 4 treatment-related toxicities occurring in 14% of patients. Only 4% of patients discontinued pembrolizumab because of adverse events, including colitis, pyrexia, pneumonitis, and thyroid abnormalities, said Dr. Antoni Ribas of University of California, Los Angeles, and his associates.
The investigators analyzed data pooled from 8 open-label phase I cohorts, some of which have been reported previously, in this manufacturer-sponsored study. All the participants were adults (median age, 61 years) with advanced unresectable melanoma who resided in Australia, Canada, France, and the U.S. This pooled study population was heterogeneous, as the various cohorts had different eligibility criteria; some patients were treatment-naïve, some had received ipilimumab, and some were treated concomitantly with BRAF or MEK inhibitors.
The estimated median duration of response was 28.2 months, and 44% of patients who responded to pembrolizumab had a response duration of longer than 1 year. Approximately half of the patients were still alive at 2 years.
Treatment benefit was consistent regardless of whether or not patients had previously received ipilimumab and regardless of the dose or schedule of pembrolizumab they were given. Patients who were treatment-naïve showed the greatest response to pembrolizumab: an overall response rate of 45%, a 1-year progression-free survival rate of 52%, and a median overall survival of 31 months, the investigators said (JAMA. 2016 Apr 19. doi: 10.1001/jama.2016.4059). “Collectively, these data suggest that the majority of patients with melanoma treated with pembrolizumab will experience lasting objective responses,” they added.
FROM JAMA
Key clinical point: Immunotherapy with pembrolizumab yielded a 33% overall response rate, a 35% 1-year progression-free survival rate, and a median overall survival of 23 months in 655 patients with advanced melanoma.
Major finding: The estimated median duration of response was 28.2 months, and half of the study population was still alive at 2 years.
Data source: A manufacturer-sponsored pooled analysis of data from 8 open-label phase I cohorts involving 655 patients followed for a median of 15 months.
Disclosures: This study was supported by Merck, maker of pembrolizumab, which also participated in the design and conduct of the study; the collection, analysis, and interpretation of the data; and preparation of the report. Dr. Ribas reported owning stock in several pharmaceutical companies. His associates reported ties to numerous industry sources.
Engineered helper T cells targeting MAGE-A3 produced some clinical responses
CD4+ T cells genetically engineered to recognize MAGE-A3, a protein found on many types of tumor cells but not on normal cells, were safely given to patients with metastatic cancers, and some of the patients had clinical responses.
Whereas the majority of current adoptive T-cell therapies have used genetically modified unpurified T cells or CD8+ (cytotoxic) T cells, Yong-Chen William Lu, Ph.D., of the surgery branch of the National Cancer Institute conducted a phase I trial of CD4+ (helper) T cells to evaluate their contribution to tumor killing. Preclinical and clinical studies indicated that CD4+ T cells can induce tumor regression.
Dr. Lu explained the individualized process of making the T cells for infusion, during a news conference at the annual meeting of the American Association for Cancer Research. First, T cells are harvested from the peripheral blood of a patient, from which CD4+ T cells are isolated. Using a retroviral vector, scientists insert the gene for a T-cell receptor that recognizes MAGE-A3 in conjunction with the HLA molecule DPB1*0401. In this way, the cells can recognize the tumor antigen in anyone of that HLA type. Then the cells carrying the engineered T-cell receptor are expanded in vitro and infused into the patient.
DPB1*0401 was chosen as the HLA genotype because “it is present in 60% of the Caucasian population. It is the highest one,” Dr. Lu said. MAGE-A3 is expressed on cells during fetal development and is later lost on normal tissues. But several tumor types, including some melanomas and some urothelial, esophageal, and cervical cancers reexpress this protein, making it a good target for immunotherapy.
The trial included patients of the HLA DPB1*0401 type with metastatic cancer that is MAGE-A3 positive. Patients had received at least one first-line treatment for metastatic disease and had been nonresponders or had had a recurrence of the tumor.
This phase I trial involved 14 patients in nine different dose cohorts, receiving from 10 million engineered T cells at the lowest dose to 78-103 billion cells in the highest cohort. Three patients in the middle and highest dosing cohorts achieved partial responses, one each with cervical, esophageal, and urothelial tumors. The partial response lasted 4 months for the patient with esophageal cancer and is ongoing at 7 months for the urothelial and 15 months for the cervical cancer patients.
In response to a question from the audience about adverse effects, Dr. Lu said patients had short-term elevations of circulating cytokines, “which only lasted for a few days,” and a fever lasting 1-2 weeks.
In summary, he said, “We think this first [CD4+] engineered T-cells therapy targeting MAGE-A3 is safe and has shown early clinical response.” Phase II trials are now beginning.
The approach of this immunotherapy method is different from that involved in using checkpoint inhibitors, “which essentially release the brakes of the immune system so the T cells can do their job of attacking the cancer,” commented Dr. Louis Weiner, director of the Georgetown Lombardi Cancer Center in Washington, D.C. “This is actually not trying to release brakes but essentially press on the accelerator, if you will, by using an expanded tumor-specific population of T cells that will then be able to hone in and attack the cancer.” He envisioned a time in the future when this approach could be combined with checkpoint inhibitors for enhanced antitumor effects.
Dr, Weiner also noted that the T cells with receptors engineered to recognize MAGE-A3 are more specific for tumors, compared with approaches using chimeric antigen receptor T cells to treat certain hematologic malignancies. In that setting the chimeric antigen receptor T cells recognize and eliminate, for example, not only malignant B cells bearing a pan-B cell marker but also all normal B cells.
However, in the question and answer period, Dr. Lu said one problem with his approach is the possibility that not all cells in a tumor may express MAGE-A3.
There was no commercial funding of the study. Dr. Lu had no relevant financial disclosures. Dr. Weiner is an adviser, review panel member, or consultant, or has other relationships, with numerous pharmaceutical companies.
CD4+ T cells genetically engineered to recognize MAGE-A3, a protein found on many types of tumor cells but not on normal cells, were safely given to patients with metastatic cancers, and some of the patients had clinical responses.
Whereas the majority of current adoptive T-cell therapies have used genetically modified unpurified T cells or CD8+ (cytotoxic) T cells, Yong-Chen William Lu, Ph.D., of the surgery branch of the National Cancer Institute conducted a phase I trial of CD4+ (helper) T cells to evaluate their contribution to tumor killing. Preclinical and clinical studies indicated that CD4+ T cells can induce tumor regression.
Dr. Lu explained the individualized process of making the T cells for infusion, during a news conference at the annual meeting of the American Association for Cancer Research. First, T cells are harvested from the peripheral blood of a patient, from which CD4+ T cells are isolated. Using a retroviral vector, scientists insert the gene for a T-cell receptor that recognizes MAGE-A3 in conjunction with the HLA molecule DPB1*0401. In this way, the cells can recognize the tumor antigen in anyone of that HLA type. Then the cells carrying the engineered T-cell receptor are expanded in vitro and infused into the patient.
DPB1*0401 was chosen as the HLA genotype because “it is present in 60% of the Caucasian population. It is the highest one,” Dr. Lu said. MAGE-A3 is expressed on cells during fetal development and is later lost on normal tissues. But several tumor types, including some melanomas and some urothelial, esophageal, and cervical cancers reexpress this protein, making it a good target for immunotherapy.
The trial included patients of the HLA DPB1*0401 type with metastatic cancer that is MAGE-A3 positive. Patients had received at least one first-line treatment for metastatic disease and had been nonresponders or had had a recurrence of the tumor.
This phase I trial involved 14 patients in nine different dose cohorts, receiving from 10 million engineered T cells at the lowest dose to 78-103 billion cells in the highest cohort. Three patients in the middle and highest dosing cohorts achieved partial responses, one each with cervical, esophageal, and urothelial tumors. The partial response lasted 4 months for the patient with esophageal cancer and is ongoing at 7 months for the urothelial and 15 months for the cervical cancer patients.
In response to a question from the audience about adverse effects, Dr. Lu said patients had short-term elevations of circulating cytokines, “which only lasted for a few days,” and a fever lasting 1-2 weeks.
In summary, he said, “We think this first [CD4+] engineered T-cells therapy targeting MAGE-A3 is safe and has shown early clinical response.” Phase II trials are now beginning.
The approach of this immunotherapy method is different from that involved in using checkpoint inhibitors, “which essentially release the brakes of the immune system so the T cells can do their job of attacking the cancer,” commented Dr. Louis Weiner, director of the Georgetown Lombardi Cancer Center in Washington, D.C. “This is actually not trying to release brakes but essentially press on the accelerator, if you will, by using an expanded tumor-specific population of T cells that will then be able to hone in and attack the cancer.” He envisioned a time in the future when this approach could be combined with checkpoint inhibitors for enhanced antitumor effects.
Dr, Weiner also noted that the T cells with receptors engineered to recognize MAGE-A3 are more specific for tumors, compared with approaches using chimeric antigen receptor T cells to treat certain hematologic malignancies. In that setting the chimeric antigen receptor T cells recognize and eliminate, for example, not only malignant B cells bearing a pan-B cell marker but also all normal B cells.
However, in the question and answer period, Dr. Lu said one problem with his approach is the possibility that not all cells in a tumor may express MAGE-A3.
There was no commercial funding of the study. Dr. Lu had no relevant financial disclosures. Dr. Weiner is an adviser, review panel member, or consultant, or has other relationships, with numerous pharmaceutical companies.
CD4+ T cells genetically engineered to recognize MAGE-A3, a protein found on many types of tumor cells but not on normal cells, were safely given to patients with metastatic cancers, and some of the patients had clinical responses.
Whereas the majority of current adoptive T-cell therapies have used genetically modified unpurified T cells or CD8+ (cytotoxic) T cells, Yong-Chen William Lu, Ph.D., of the surgery branch of the National Cancer Institute conducted a phase I trial of CD4+ (helper) T cells to evaluate their contribution to tumor killing. Preclinical and clinical studies indicated that CD4+ T cells can induce tumor regression.
Dr. Lu explained the individualized process of making the T cells for infusion, during a news conference at the annual meeting of the American Association for Cancer Research. First, T cells are harvested from the peripheral blood of a patient, from which CD4+ T cells are isolated. Using a retroviral vector, scientists insert the gene for a T-cell receptor that recognizes MAGE-A3 in conjunction with the HLA molecule DPB1*0401. In this way, the cells can recognize the tumor antigen in anyone of that HLA type. Then the cells carrying the engineered T-cell receptor are expanded in vitro and infused into the patient.
DPB1*0401 was chosen as the HLA genotype because “it is present in 60% of the Caucasian population. It is the highest one,” Dr. Lu said. MAGE-A3 is expressed on cells during fetal development and is later lost on normal tissues. But several tumor types, including some melanomas and some urothelial, esophageal, and cervical cancers reexpress this protein, making it a good target for immunotherapy.
The trial included patients of the HLA DPB1*0401 type with metastatic cancer that is MAGE-A3 positive. Patients had received at least one first-line treatment for metastatic disease and had been nonresponders or had had a recurrence of the tumor.
This phase I trial involved 14 patients in nine different dose cohorts, receiving from 10 million engineered T cells at the lowest dose to 78-103 billion cells in the highest cohort. Three patients in the middle and highest dosing cohorts achieved partial responses, one each with cervical, esophageal, and urothelial tumors. The partial response lasted 4 months for the patient with esophageal cancer and is ongoing at 7 months for the urothelial and 15 months for the cervical cancer patients.
In response to a question from the audience about adverse effects, Dr. Lu said patients had short-term elevations of circulating cytokines, “which only lasted for a few days,” and a fever lasting 1-2 weeks.
In summary, he said, “We think this first [CD4+] engineered T-cells therapy targeting MAGE-A3 is safe and has shown early clinical response.” Phase II trials are now beginning.
The approach of this immunotherapy method is different from that involved in using checkpoint inhibitors, “which essentially release the brakes of the immune system so the T cells can do their job of attacking the cancer,” commented Dr. Louis Weiner, director of the Georgetown Lombardi Cancer Center in Washington, D.C. “This is actually not trying to release brakes but essentially press on the accelerator, if you will, by using an expanded tumor-specific population of T cells that will then be able to hone in and attack the cancer.” He envisioned a time in the future when this approach could be combined with checkpoint inhibitors for enhanced antitumor effects.
Dr, Weiner also noted that the T cells with receptors engineered to recognize MAGE-A3 are more specific for tumors, compared with approaches using chimeric antigen receptor T cells to treat certain hematologic malignancies. In that setting the chimeric antigen receptor T cells recognize and eliminate, for example, not only malignant B cells bearing a pan-B cell marker but also all normal B cells.
However, in the question and answer period, Dr. Lu said one problem with his approach is the possibility that not all cells in a tumor may express MAGE-A3.
There was no commercial funding of the study. Dr. Lu had no relevant financial disclosures. Dr. Weiner is an adviser, review panel member, or consultant, or has other relationships, with numerous pharmaceutical companies.
FROM THE AACR ANNUAL MEETING
Key clinical point: Engineered helper T cells safely target tumors and produce clinical responses.
Major finding: Three of 14 patients with different tumor types had partial tumor responses when given T cells with receptors engineered to recognize a common tumor antigen, MAGE-A3.
Data source: A phase I dose-escalating study of 14 patients.
Disclosures: There was no commercial funding of the study. Dr. Lu had no relevant financial disclosures. Dr. Weiner is an adviser, review panel member, or consultant, or has other relationships, with numerous pharmaceutical companies.
Study found long-term benefit of nivolumab for advanced melanoma patients
One-third of a group of heavily pretreated patients with advanced melanoma who received nivolumab monotherapy were alive 5 years after starting the drug, a phase I trial showed. Survival plateaued at 34% by 48 months, after which time disease control was maintained.
“What it does demonstrate is the importance of the durability of clinical benefit to patients [who] now we’re measuring in terms of years, as well as the memory aspect of how the immunologic memory can translate into benefit,” said Dr. Stephen Hodi, director of the melanoma center at Dana-Farber Cancer Institute in Boston during a news conference at the annual meeting of the American Association for Cancer Research.
Nivolumab is an immune checkpoint inhibitor that blocks PD-1, allowing enhanced antitumor immune responses. It is approved as a first-line agent for advanced melanoma, alone or in combination with ipilimumab. The approved dose for monotherapy is 3 mg/kg every 2 weeks.
The study randomized 107 patients (median age, 61 years; 67% male) to one of five doses ranging from 0.1 mg/kg to 10 mg/kg IV given every 2 weeks for up to 96 weeks. The patients had advanced melanoma at any site and had been on one to five lines of systemic therapy prior to entering the trial but could not have received any immune checkpoint inhibitors. They had treated and stable brain metastases and an ECOG (Eastern Cooperative Oncology Group) performance status of 0, 1, or 2. The minimum follow-up was 45 months.
Among all the patients, the median overall survival was 17.3 months (95% confidence interval, 12.5-37.8 months), and for those patients receiving 3-mg/kg doses, the median survival was 20.3 months (95% CI, 7.2 months and no upper confidence level reached). The Kaplan-Meier survival curves for all patients and those receiving the 3-mg/kg dose began to level off at about 36 months, and were essentially flat at 34% for all patients as a group from 48 months out to 5 years.
Five patients who achieved disease control but later had progressive disease were retreated with their original doses of nivolumab after being off the drug for more than 100 days. “In all five patients, disease control was obtained again, and this disease control was again durable,” Dr. Hodi reported.
Nivolumab monotherapy was safe and well tolerated. No study deaths or new safety signals were observed. The most common drug-related adverse effects were any grade of fatigue (29.9% of all patients; 47.1% of patients at the 3-mg/kg dose) and any grade of rash (23.4% and 11.8%, respectively). Although there were some grade 3-4 adverse events, adverse reactions led to study discontinuation for only 10.3% of the total patient group and 5.9% of patients on the 3-mg/kg dose.
Calling the study findings “very compelling,” conference moderator Dr. Louis Weiner, director of the Georgetown Lombardi Comprehensive Cancer Center in Washington, D.C., identified what he said is a key point of the study – the length of benefit. “What distinguishes immunotherapy at this point when it’s effective from other forms of cancer treatment is the durability of the benefit,” he said. “People who have good responses really seem to be protected against the disease returning in many cases.”
He asked Dr. Hodi if he would expect even better disease control results if nivolumab were combined with a different checkpoint inhibitor, such as ipilimumab. Dr. Hodi replied that it is reasonable to think that agents working through different mechanisms of action should produce even better results.
A reporter in the audience asked if it was Dr. Hodi’s practice to test tumors for PD-1 expression before using nivolumab. He replied that he and his colleagues do not routinely test for PD-1 because there have been observations that some tumors that test negative for the biomarker do respond to the drug.
One-third of a group of heavily pretreated patients with advanced melanoma who received nivolumab monotherapy were alive 5 years after starting the drug, a phase I trial showed. Survival plateaued at 34% by 48 months, after which time disease control was maintained.
“What it does demonstrate is the importance of the durability of clinical benefit to patients [who] now we’re measuring in terms of years, as well as the memory aspect of how the immunologic memory can translate into benefit,” said Dr. Stephen Hodi, director of the melanoma center at Dana-Farber Cancer Institute in Boston during a news conference at the annual meeting of the American Association for Cancer Research.
Nivolumab is an immune checkpoint inhibitor that blocks PD-1, allowing enhanced antitumor immune responses. It is approved as a first-line agent for advanced melanoma, alone or in combination with ipilimumab. The approved dose for monotherapy is 3 mg/kg every 2 weeks.
The study randomized 107 patients (median age, 61 years; 67% male) to one of five doses ranging from 0.1 mg/kg to 10 mg/kg IV given every 2 weeks for up to 96 weeks. The patients had advanced melanoma at any site and had been on one to five lines of systemic therapy prior to entering the trial but could not have received any immune checkpoint inhibitors. They had treated and stable brain metastases and an ECOG (Eastern Cooperative Oncology Group) performance status of 0, 1, or 2. The minimum follow-up was 45 months.
Among all the patients, the median overall survival was 17.3 months (95% confidence interval, 12.5-37.8 months), and for those patients receiving 3-mg/kg doses, the median survival was 20.3 months (95% CI, 7.2 months and no upper confidence level reached). The Kaplan-Meier survival curves for all patients and those receiving the 3-mg/kg dose began to level off at about 36 months, and were essentially flat at 34% for all patients as a group from 48 months out to 5 years.
Five patients who achieved disease control but later had progressive disease were retreated with their original doses of nivolumab after being off the drug for more than 100 days. “In all five patients, disease control was obtained again, and this disease control was again durable,” Dr. Hodi reported.
Nivolumab monotherapy was safe and well tolerated. No study deaths or new safety signals were observed. The most common drug-related adverse effects were any grade of fatigue (29.9% of all patients; 47.1% of patients at the 3-mg/kg dose) and any grade of rash (23.4% and 11.8%, respectively). Although there were some grade 3-4 adverse events, adverse reactions led to study discontinuation for only 10.3% of the total patient group and 5.9% of patients on the 3-mg/kg dose.
Calling the study findings “very compelling,” conference moderator Dr. Louis Weiner, director of the Georgetown Lombardi Comprehensive Cancer Center in Washington, D.C., identified what he said is a key point of the study – the length of benefit. “What distinguishes immunotherapy at this point when it’s effective from other forms of cancer treatment is the durability of the benefit,” he said. “People who have good responses really seem to be protected against the disease returning in many cases.”
He asked Dr. Hodi if he would expect even better disease control results if nivolumab were combined with a different checkpoint inhibitor, such as ipilimumab. Dr. Hodi replied that it is reasonable to think that agents working through different mechanisms of action should produce even better results.
A reporter in the audience asked if it was Dr. Hodi’s practice to test tumors for PD-1 expression before using nivolumab. He replied that he and his colleagues do not routinely test for PD-1 because there have been observations that some tumors that test negative for the biomarker do respond to the drug.
One-third of a group of heavily pretreated patients with advanced melanoma who received nivolumab monotherapy were alive 5 years after starting the drug, a phase I trial showed. Survival plateaued at 34% by 48 months, after which time disease control was maintained.
“What it does demonstrate is the importance of the durability of clinical benefit to patients [who] now we’re measuring in terms of years, as well as the memory aspect of how the immunologic memory can translate into benefit,” said Dr. Stephen Hodi, director of the melanoma center at Dana-Farber Cancer Institute in Boston during a news conference at the annual meeting of the American Association for Cancer Research.
Nivolumab is an immune checkpoint inhibitor that blocks PD-1, allowing enhanced antitumor immune responses. It is approved as a first-line agent for advanced melanoma, alone or in combination with ipilimumab. The approved dose for monotherapy is 3 mg/kg every 2 weeks.
The study randomized 107 patients (median age, 61 years; 67% male) to one of five doses ranging from 0.1 mg/kg to 10 mg/kg IV given every 2 weeks for up to 96 weeks. The patients had advanced melanoma at any site and had been on one to five lines of systemic therapy prior to entering the trial but could not have received any immune checkpoint inhibitors. They had treated and stable brain metastases and an ECOG (Eastern Cooperative Oncology Group) performance status of 0, 1, or 2. The minimum follow-up was 45 months.
Among all the patients, the median overall survival was 17.3 months (95% confidence interval, 12.5-37.8 months), and for those patients receiving 3-mg/kg doses, the median survival was 20.3 months (95% CI, 7.2 months and no upper confidence level reached). The Kaplan-Meier survival curves for all patients and those receiving the 3-mg/kg dose began to level off at about 36 months, and were essentially flat at 34% for all patients as a group from 48 months out to 5 years.
Five patients who achieved disease control but later had progressive disease were retreated with their original doses of nivolumab after being off the drug for more than 100 days. “In all five patients, disease control was obtained again, and this disease control was again durable,” Dr. Hodi reported.
Nivolumab monotherapy was safe and well tolerated. No study deaths or new safety signals were observed. The most common drug-related adverse effects were any grade of fatigue (29.9% of all patients; 47.1% of patients at the 3-mg/kg dose) and any grade of rash (23.4% and 11.8%, respectively). Although there were some grade 3-4 adverse events, adverse reactions led to study discontinuation for only 10.3% of the total patient group and 5.9% of patients on the 3-mg/kg dose.
Calling the study findings “very compelling,” conference moderator Dr. Louis Weiner, director of the Georgetown Lombardi Comprehensive Cancer Center in Washington, D.C., identified what he said is a key point of the study – the length of benefit. “What distinguishes immunotherapy at this point when it’s effective from other forms of cancer treatment is the durability of the benefit,” he said. “People who have good responses really seem to be protected against the disease returning in many cases.”
He asked Dr. Hodi if he would expect even better disease control results if nivolumab were combined with a different checkpoint inhibitor, such as ipilimumab. Dr. Hodi replied that it is reasonable to think that agents working through different mechanisms of action should produce even better results.
A reporter in the audience asked if it was Dr. Hodi’s practice to test tumors for PD-1 expression before using nivolumab. He replied that he and his colleagues do not routinely test for PD-1 because there have been observations that some tumors that test negative for the biomarker do respond to the drug.
FROM THE AACR ANNUAL MEETING
Key clinical point: Heavily pretreated patients with advanced melanoma had long-term responses on nivolumab monotherapy.
Major finding: Nivolumab monotherapy resulted in a 34% overall survival rate at 5 years for melanoma patients who had received extensive prior therapy. Data source: A phase I randomized dose-ranging study involving 107 patients with advanced melanoma.
Disclosures: The study was funded by Bristol-Myers Squibb. Dr. Hodi has received grant/research support from and is a nonpaid consultant to Bristol-Myers. Dr. Weiner is an adviser, review panel member, or consultant or has other relationships with numerous pharmaceutical companies.