User login
Think of pediatric morphea as a systemic, chronic disease, expert advises
INDIANAPOLIS – In the opinion of Elena Pope, MD, MSc,
“There is no correlation between the extent and activity of skin lesions and the presence, severity, and activity of extracutaneous manifestations,” Dr. Pope, professor of pediatrics at the University of Toronto and division head of pediatric dermatology at the Hospital for Sick Children in Toronto, said during the annual meeting of the Society for Pediatric Dermatology. “Treatment needs to be tailored to the extent of cutaneous manifestations, and I think we need to be aware of and address the impact on patients’ quality of life,” she added. There is also a need for more research “on targeted and better-tolerated therapies to put a stop to the progression of disease.”
Congenital morphea is a form of localized scleroderma that presents at birth but can be confused with port wine stain. Results from a multicenter retrospective review of 25 cases conducted by Dr. Pope and colleagues found that the median age at diagnosis was 2.9 years and 76% had linear-type lesions. In addition, 48% had extracutaneous involvement (all of these patients had linear morphea), most commonly of the central nervous system.
“It’s important to realize these lesions may become active over time,” Dr. Pope said. “In my experience, there are two different courses. Either you have innocuous lesions when the patients are born and they may become active around 3-4 years of age, or you have early intrauterine involvement, with lesions inactive at birth but with potential for significant damage in utero.”
She cautioned against treating a suspected port wine stain lesion with laser until congenital morphea is ruled out. “I’m aware of at least one lawsuit of a child where someone used a laser in a child who had progression with significant sclerosis,” she said. “The parents assumed it was the use of the laser that led to the progression, not the actual disease.”
Extracutaneous manifestations are common in morphea patients. A multicenter study of 750 patients with juvenile scleroderma found that 22% had extracutaneous manifestations. Almost half of patients (47%) had arthritis, but 17% had neurologic findings such as seizures and headaches, 9% had vascular manifestations, and 8% had uveitis. Subsequent studies found that neurological disease affects between 11% and 19% of cases, especially in those involving the head and neck.
“There is a wide range of manifestations from headache and neuropsychiatric changes to brain atrophy, seizures, and CNS cavernoma,” Dr. Pope said. “There also can be orthodental involvement such as malocclusion. It’s important to do a brain MRI, eye exam for uveitis, and don’t forget the orthodental assessment.”
She recalled a 10-year-old boy who presented to the Hospital for Sick Children with tissue loss on the forehead and eyebrow and eyelashes. He had no other congenital morphea symptoms and the MRI was normal, but the eye exam revealed uveitis. “It’s important to remember that uveitis is asymptomatic, so unless you look for it, you’re not going to find it,” she said.
According to unpublished data in 42 congenital morphea patients with lesions limited to the head and neck, who underwent MRI imaging at the Hospital for Sick Children, 57% had CNS changes that were ipsilateral in 68% of cases. “White matter changes were the most common, and to our surprise, there were patients who had progressive CNS disease, including CNS vasculitis, new lesions, and enhancement of prior stable lesions,” Dr. Pope said.
She recalled the case of an 8-year-old boy who presented to the hospital with intractable seizures. Upon completion of the MRI, one of the radiologists noted that the imaging showed subtle thinning of the forehead, and he was referred to Dr. Pope and colleagues for assessment. In the span of 4 years, despite aggressive treatment, the boy’s CNS disease progressed. “There was more enhancement, more tissue loss, his seizures are very hard to control, and he has many neurodevelopmental changes,” she recalled. “What I learned from this case is that skin activity does not correlate with imaging. Don’t assume that just because the skin is burnt out that the CNS will be the same. Also, the extent of skin disease does not predict involvement or progression of the CNS.”
Linear lesions on the lower extremities are a harbinger of orthopedic complications, which can occur in about half of patients. Joint contractures in this subset of patients are seen in about 81% of cases, while other sequelae can include arthritis, limb atrophy, leg-leg discrepancy, and angular deformity. “About 14% of patients require intervention,” Dr. Pope said. “In terms of working those patients up, you need to do an MRI and assess the extent of muscle and fascial involvement. Early physiotherapy and an orthopedic evaluation are also recommended.”
As for possible markers of morphea, antinuclear antibody is positive in 22%-68% of cases and correlates with disease severity, extracutaneous manifestations, and disease flare-up. Antihistone antibodies (AHA) are positive in about 47% of cases, “and that tends to correlate with the extent of skin and muscle involvement,” Dr. Pope said. “Anti–double-stranded DNA correlates with extent of disease, but the only known biomarker to date that correlates with disease activity is CXCL9/10. This has been documented in the skin as well as in the blood. So, this marker may help us determine if the patient needs to be treated or not.”
Treatments
For treatment of active localized disease, topical medications are helpful in some cases. Options include topical steroids, calcipotriol with or without betamethasone, imiquimod, and tacrolimus. “In my experience the combination of calcipotriol with betamethasone is best,” she said. “It really shuts down the activity fairly soon, and you can scale down to calcipotriol alone. I don’t find imiquimod very helpful for active lesions, although it has a role for inactive lesions.”
For patients with linear or generalized/mixed disease, “the combination of methotrexate and corticosteroids or methotrexate alone is probably the way to go,” Dr. Pope said. “The addition of steroids really depends on where the lesion is and how worried you are about other problems.”
According to the best available literature, 88% of patients should respond to treatment with methotrexate (MTX) and/or steroids within 3-6 months, and 74% within 3 months. “If they don’t, you have to wonder if the patient’s taking the medication, or you need to think about other alternative treatments,” she said. “Complete remission is possible in most of the patients, and the longer you treat the more you will see that. On average, most of us treat patients for about 3 years, but there are treatment failures as well. This can occur in up to 16% of patients.”
As for second-line treatment agents for congenital morphea, clinicians often turn to mycophenolate mofetil (MMF). Results from a retrospective longitudinal study of juvenile localized scleroderma patients found that after a mean of 9 years 91% of patients on MMF and 100% of patients on MTX had inactive disease. “There were no differences in relapse rates, although MMF seems to have a more sustained long-term effect and overall is better tolerated,” said Dr. Pope, who was not involved with the study. “However, it’s more immunosuppressive than MTX, which is important, especially in the era of COVID-19. You also need to think about the potential for more hematological suppression with MMF use.” If standard therapy fails, there is anecdotal data supporting the use of abatacept (which suppresses the T-cell activity in affected patients), tofacitinib (which inhibits transforming growth factor–beta), or dupilumab (which inhibits interleukin-4).
Dr. Pope emphasized the effect congenital morphea has on quality of life. Remarks from patients with facial morphea and their parents who participated in a focus group on the topic organized by the Hospital for Sick Children included, “You just want to stay inside because you are afraid of what people will say,” “They laugh at her. They make fun of her, and it’s terrible,” and “MTX makes me feel weird. I would throw up, feel dizzy.”
“You have to take that into consideration, because we cannot make the treatment worse than the disease,” Dr. Pope said. “There are many domains where patients could be affected, including skin symptoms, physical functioning, body image and social support, side effects of medication, and presence of extracutaneous manifestations. Predictors of poor quality of life include female sex and involvement of hands and feet.”
Dr. Pope disclosed that she has received grants/research support from AbbVie, Centocor, and Amgen. She has also received consulting fees from AbbVie, Sanofi, Novartis, Boehringer-Ingelheim, Phoenix, Amryt Pharma, and Timber Pharmaceuticals.
INDIANAPOLIS – In the opinion of Elena Pope, MD, MSc,
“There is no correlation between the extent and activity of skin lesions and the presence, severity, and activity of extracutaneous manifestations,” Dr. Pope, professor of pediatrics at the University of Toronto and division head of pediatric dermatology at the Hospital for Sick Children in Toronto, said during the annual meeting of the Society for Pediatric Dermatology. “Treatment needs to be tailored to the extent of cutaneous manifestations, and I think we need to be aware of and address the impact on patients’ quality of life,” she added. There is also a need for more research “on targeted and better-tolerated therapies to put a stop to the progression of disease.”
Congenital morphea is a form of localized scleroderma that presents at birth but can be confused with port wine stain. Results from a multicenter retrospective review of 25 cases conducted by Dr. Pope and colleagues found that the median age at diagnosis was 2.9 years and 76% had linear-type lesions. In addition, 48% had extracutaneous involvement (all of these patients had linear morphea), most commonly of the central nervous system.
“It’s important to realize these lesions may become active over time,” Dr. Pope said. “In my experience, there are two different courses. Either you have innocuous lesions when the patients are born and they may become active around 3-4 years of age, or you have early intrauterine involvement, with lesions inactive at birth but with potential for significant damage in utero.”
She cautioned against treating a suspected port wine stain lesion with laser until congenital morphea is ruled out. “I’m aware of at least one lawsuit of a child where someone used a laser in a child who had progression with significant sclerosis,” she said. “The parents assumed it was the use of the laser that led to the progression, not the actual disease.”
Extracutaneous manifestations are common in morphea patients. A multicenter study of 750 patients with juvenile scleroderma found that 22% had extracutaneous manifestations. Almost half of patients (47%) had arthritis, but 17% had neurologic findings such as seizures and headaches, 9% had vascular manifestations, and 8% had uveitis. Subsequent studies found that neurological disease affects between 11% and 19% of cases, especially in those involving the head and neck.
“There is a wide range of manifestations from headache and neuropsychiatric changes to brain atrophy, seizures, and CNS cavernoma,” Dr. Pope said. “There also can be orthodental involvement such as malocclusion. It’s important to do a brain MRI, eye exam for uveitis, and don’t forget the orthodental assessment.”
She recalled a 10-year-old boy who presented to the Hospital for Sick Children with tissue loss on the forehead and eyebrow and eyelashes. He had no other congenital morphea symptoms and the MRI was normal, but the eye exam revealed uveitis. “It’s important to remember that uveitis is asymptomatic, so unless you look for it, you’re not going to find it,” she said.
According to unpublished data in 42 congenital morphea patients with lesions limited to the head and neck, who underwent MRI imaging at the Hospital for Sick Children, 57% had CNS changes that were ipsilateral in 68% of cases. “White matter changes were the most common, and to our surprise, there were patients who had progressive CNS disease, including CNS vasculitis, new lesions, and enhancement of prior stable lesions,” Dr. Pope said.
She recalled the case of an 8-year-old boy who presented to the hospital with intractable seizures. Upon completion of the MRI, one of the radiologists noted that the imaging showed subtle thinning of the forehead, and he was referred to Dr. Pope and colleagues for assessment. In the span of 4 years, despite aggressive treatment, the boy’s CNS disease progressed. “There was more enhancement, more tissue loss, his seizures are very hard to control, and he has many neurodevelopmental changes,” she recalled. “What I learned from this case is that skin activity does not correlate with imaging. Don’t assume that just because the skin is burnt out that the CNS will be the same. Also, the extent of skin disease does not predict involvement or progression of the CNS.”
Linear lesions on the lower extremities are a harbinger of orthopedic complications, which can occur in about half of patients. Joint contractures in this subset of patients are seen in about 81% of cases, while other sequelae can include arthritis, limb atrophy, leg-leg discrepancy, and angular deformity. “About 14% of patients require intervention,” Dr. Pope said. “In terms of working those patients up, you need to do an MRI and assess the extent of muscle and fascial involvement. Early physiotherapy and an orthopedic evaluation are also recommended.”
As for possible markers of morphea, antinuclear antibody is positive in 22%-68% of cases and correlates with disease severity, extracutaneous manifestations, and disease flare-up. Antihistone antibodies (AHA) are positive in about 47% of cases, “and that tends to correlate with the extent of skin and muscle involvement,” Dr. Pope said. “Anti–double-stranded DNA correlates with extent of disease, but the only known biomarker to date that correlates with disease activity is CXCL9/10. This has been documented in the skin as well as in the blood. So, this marker may help us determine if the patient needs to be treated or not.”
Treatments
For treatment of active localized disease, topical medications are helpful in some cases. Options include topical steroids, calcipotriol with or without betamethasone, imiquimod, and tacrolimus. “In my experience the combination of calcipotriol with betamethasone is best,” she said. “It really shuts down the activity fairly soon, and you can scale down to calcipotriol alone. I don’t find imiquimod very helpful for active lesions, although it has a role for inactive lesions.”
For patients with linear or generalized/mixed disease, “the combination of methotrexate and corticosteroids or methotrexate alone is probably the way to go,” Dr. Pope said. “The addition of steroids really depends on where the lesion is and how worried you are about other problems.”
According to the best available literature, 88% of patients should respond to treatment with methotrexate (MTX) and/or steroids within 3-6 months, and 74% within 3 months. “If they don’t, you have to wonder if the patient’s taking the medication, or you need to think about other alternative treatments,” she said. “Complete remission is possible in most of the patients, and the longer you treat the more you will see that. On average, most of us treat patients for about 3 years, but there are treatment failures as well. This can occur in up to 16% of patients.”
As for second-line treatment agents for congenital morphea, clinicians often turn to mycophenolate mofetil (MMF). Results from a retrospective longitudinal study of juvenile localized scleroderma patients found that after a mean of 9 years 91% of patients on MMF and 100% of patients on MTX had inactive disease. “There were no differences in relapse rates, although MMF seems to have a more sustained long-term effect and overall is better tolerated,” said Dr. Pope, who was not involved with the study. “However, it’s more immunosuppressive than MTX, which is important, especially in the era of COVID-19. You also need to think about the potential for more hematological suppression with MMF use.” If standard therapy fails, there is anecdotal data supporting the use of abatacept (which suppresses the T-cell activity in affected patients), tofacitinib (which inhibits transforming growth factor–beta), or dupilumab (which inhibits interleukin-4).
Dr. Pope emphasized the effect congenital morphea has on quality of life. Remarks from patients with facial morphea and their parents who participated in a focus group on the topic organized by the Hospital for Sick Children included, “You just want to stay inside because you are afraid of what people will say,” “They laugh at her. They make fun of her, and it’s terrible,” and “MTX makes me feel weird. I would throw up, feel dizzy.”
“You have to take that into consideration, because we cannot make the treatment worse than the disease,” Dr. Pope said. “There are many domains where patients could be affected, including skin symptoms, physical functioning, body image and social support, side effects of medication, and presence of extracutaneous manifestations. Predictors of poor quality of life include female sex and involvement of hands and feet.”
Dr. Pope disclosed that she has received grants/research support from AbbVie, Centocor, and Amgen. She has also received consulting fees from AbbVie, Sanofi, Novartis, Boehringer-Ingelheim, Phoenix, Amryt Pharma, and Timber Pharmaceuticals.
INDIANAPOLIS – In the opinion of Elena Pope, MD, MSc,
“There is no correlation between the extent and activity of skin lesions and the presence, severity, and activity of extracutaneous manifestations,” Dr. Pope, professor of pediatrics at the University of Toronto and division head of pediatric dermatology at the Hospital for Sick Children in Toronto, said during the annual meeting of the Society for Pediatric Dermatology. “Treatment needs to be tailored to the extent of cutaneous manifestations, and I think we need to be aware of and address the impact on patients’ quality of life,” she added. There is also a need for more research “on targeted and better-tolerated therapies to put a stop to the progression of disease.”
Congenital morphea is a form of localized scleroderma that presents at birth but can be confused with port wine stain. Results from a multicenter retrospective review of 25 cases conducted by Dr. Pope and colleagues found that the median age at diagnosis was 2.9 years and 76% had linear-type lesions. In addition, 48% had extracutaneous involvement (all of these patients had linear morphea), most commonly of the central nervous system.
“It’s important to realize these lesions may become active over time,” Dr. Pope said. “In my experience, there are two different courses. Either you have innocuous lesions when the patients are born and they may become active around 3-4 years of age, or you have early intrauterine involvement, with lesions inactive at birth but with potential for significant damage in utero.”
She cautioned against treating a suspected port wine stain lesion with laser until congenital morphea is ruled out. “I’m aware of at least one lawsuit of a child where someone used a laser in a child who had progression with significant sclerosis,” she said. “The parents assumed it was the use of the laser that led to the progression, not the actual disease.”
Extracutaneous manifestations are common in morphea patients. A multicenter study of 750 patients with juvenile scleroderma found that 22% had extracutaneous manifestations. Almost half of patients (47%) had arthritis, but 17% had neurologic findings such as seizures and headaches, 9% had vascular manifestations, and 8% had uveitis. Subsequent studies found that neurological disease affects between 11% and 19% of cases, especially in those involving the head and neck.
“There is a wide range of manifestations from headache and neuropsychiatric changes to brain atrophy, seizures, and CNS cavernoma,” Dr. Pope said. “There also can be orthodental involvement such as malocclusion. It’s important to do a brain MRI, eye exam for uveitis, and don’t forget the orthodental assessment.”
She recalled a 10-year-old boy who presented to the Hospital for Sick Children with tissue loss on the forehead and eyebrow and eyelashes. He had no other congenital morphea symptoms and the MRI was normal, but the eye exam revealed uveitis. “It’s important to remember that uveitis is asymptomatic, so unless you look for it, you’re not going to find it,” she said.
According to unpublished data in 42 congenital morphea patients with lesions limited to the head and neck, who underwent MRI imaging at the Hospital for Sick Children, 57% had CNS changes that were ipsilateral in 68% of cases. “White matter changes were the most common, and to our surprise, there were patients who had progressive CNS disease, including CNS vasculitis, new lesions, and enhancement of prior stable lesions,” Dr. Pope said.
She recalled the case of an 8-year-old boy who presented to the hospital with intractable seizures. Upon completion of the MRI, one of the radiologists noted that the imaging showed subtle thinning of the forehead, and he was referred to Dr. Pope and colleagues for assessment. In the span of 4 years, despite aggressive treatment, the boy’s CNS disease progressed. “There was more enhancement, more tissue loss, his seizures are very hard to control, and he has many neurodevelopmental changes,” she recalled. “What I learned from this case is that skin activity does not correlate with imaging. Don’t assume that just because the skin is burnt out that the CNS will be the same. Also, the extent of skin disease does not predict involvement or progression of the CNS.”
Linear lesions on the lower extremities are a harbinger of orthopedic complications, which can occur in about half of patients. Joint contractures in this subset of patients are seen in about 81% of cases, while other sequelae can include arthritis, limb atrophy, leg-leg discrepancy, and angular deformity. “About 14% of patients require intervention,” Dr. Pope said. “In terms of working those patients up, you need to do an MRI and assess the extent of muscle and fascial involvement. Early physiotherapy and an orthopedic evaluation are also recommended.”
As for possible markers of morphea, antinuclear antibody is positive in 22%-68% of cases and correlates with disease severity, extracutaneous manifestations, and disease flare-up. Antihistone antibodies (AHA) are positive in about 47% of cases, “and that tends to correlate with the extent of skin and muscle involvement,” Dr. Pope said. “Anti–double-stranded DNA correlates with extent of disease, but the only known biomarker to date that correlates with disease activity is CXCL9/10. This has been documented in the skin as well as in the blood. So, this marker may help us determine if the patient needs to be treated or not.”
Treatments
For treatment of active localized disease, topical medications are helpful in some cases. Options include topical steroids, calcipotriol with or without betamethasone, imiquimod, and tacrolimus. “In my experience the combination of calcipotriol with betamethasone is best,” she said. “It really shuts down the activity fairly soon, and you can scale down to calcipotriol alone. I don’t find imiquimod very helpful for active lesions, although it has a role for inactive lesions.”
For patients with linear or generalized/mixed disease, “the combination of methotrexate and corticosteroids or methotrexate alone is probably the way to go,” Dr. Pope said. “The addition of steroids really depends on where the lesion is and how worried you are about other problems.”
According to the best available literature, 88% of patients should respond to treatment with methotrexate (MTX) and/or steroids within 3-6 months, and 74% within 3 months. “If they don’t, you have to wonder if the patient’s taking the medication, or you need to think about other alternative treatments,” she said. “Complete remission is possible in most of the patients, and the longer you treat the more you will see that. On average, most of us treat patients for about 3 years, but there are treatment failures as well. This can occur in up to 16% of patients.”
As for second-line treatment agents for congenital morphea, clinicians often turn to mycophenolate mofetil (MMF). Results from a retrospective longitudinal study of juvenile localized scleroderma patients found that after a mean of 9 years 91% of patients on MMF and 100% of patients on MTX had inactive disease. “There were no differences in relapse rates, although MMF seems to have a more sustained long-term effect and overall is better tolerated,” said Dr. Pope, who was not involved with the study. “However, it’s more immunosuppressive than MTX, which is important, especially in the era of COVID-19. You also need to think about the potential for more hematological suppression with MMF use.” If standard therapy fails, there is anecdotal data supporting the use of abatacept (which suppresses the T-cell activity in affected patients), tofacitinib (which inhibits transforming growth factor–beta), or dupilumab (which inhibits interleukin-4).
Dr. Pope emphasized the effect congenital morphea has on quality of life. Remarks from patients with facial morphea and their parents who participated in a focus group on the topic organized by the Hospital for Sick Children included, “You just want to stay inside because you are afraid of what people will say,” “They laugh at her. They make fun of her, and it’s terrible,” and “MTX makes me feel weird. I would throw up, feel dizzy.”
“You have to take that into consideration, because we cannot make the treatment worse than the disease,” Dr. Pope said. “There are many domains where patients could be affected, including skin symptoms, physical functioning, body image and social support, side effects of medication, and presence of extracutaneous manifestations. Predictors of poor quality of life include female sex and involvement of hands and feet.”
Dr. Pope disclosed that she has received grants/research support from AbbVie, Centocor, and Amgen. She has also received consulting fees from AbbVie, Sanofi, Novartis, Boehringer-Ingelheim, Phoenix, Amryt Pharma, and Timber Pharmaceuticals.
AT SPD 2022
No more ‘escape hatch’: Post Roe, new worries about meds linked to birth defects
As states ban or limit abortion in the wake of the demise of Roe v. Wade, physicians are turning their attention to widely-used drugs that can cause birth defects. At issue: Should these drugs still be prescribed to women of childbearing age if they don’t have the option of terminating their pregnancies?
“Doctors are going to understandably be terrified that a patient may become pregnant using a teratogen that they have prescribed,” said University of Pittsburgh rheumatologist Mehret Birru Talabi, MD, PhD, who works in a state where the future of abortion rights is uncertain. “While this was a feared outcome before Roe v. Wade was overturned, abortion provided an escape hatch by which women could avoid having to continue a pregnancy and potentially raise a child with congenital anomalies. I believe that prescribing is going to become much more defensive and conservative. Some clinicians may choose not to prescribe these medications to patients who have childbearing potential, even if they don’t have much risk for pregnancy.”
Other physicians expressed similar concerns in interviews. Duke University, Durham, N.C., rheumatologist Megan E. B. Clowse, MD, MPH, fears that physicians will be wary of prescribing a variety of medications – including new ones for which there are few pregnancy data – if abortion is unavailable. “Women who receive these new or teratogenic medications will likely lose their reproductive autonomy and be forced to choose between having sexual relationships with men, obtaining procedures that make them permanently sterile, or using contraception that may cause intolerable side effects,” she said. “I am very concerned that young women with rheumatic disease will now be left with active disease resulting in joint damage and renal failure.”
Abortion is now banned in at least six states, according to The New York Times. That number may rise to 16 as more restrictions become law. Another five states aren’t expected to ban abortion soon but have implemented gestational age limits on abortion or are expected to adopt them. In another nine states, courts or lawmakers will decide whether abortion remains legal.
Only 20 states and the District of Columbia have firm abortion protections in place.
Numerous drugs are considered teratogens, which means they may cause birth defects. Thalidomide is the most infamous, but there are many more, including several used in rheumatology, dermatology, and gastroenterology. Among the most widely used teratogenic medications are the acne drugs isotretinoin and methotrexate, which are used to treat a variety of conditions, such as cancer, rheumatoid arthritis, and psoriasis.
Dr. Clowse, who helps manage an industry-supported website devoted to reproductive care for women with lupus (www.LupusPregnancy.org), noted that several drugs linked to birth defects and pregnancy loss are commonly prescribed in rheumatology.
“Methotrexate is the most common medication and has been the cornerstone of rheumatoid arthritis [treatment] for at least two decades,” she said. “Mycophenolate is our best medication to treat lupus nephritis, which is inflammation in the kidneys caused by lupus. This is a common complication for young women with lupus, and all of our guideline-recommended treatment regimens include a medication that causes pregnancy loss and birth defects, either mycophenolate or cyclophosphamide.”
Rheumatologists also prescribe a large number of new drugs for which there are few data about pregnancy risks. “It typically takes about two decades to have sufficient data about the safety of our medications,” she said.
Reflecting the sensitivity of the topic, Dr. Clowse made clear that her opinions don’t represent the views of her institution. She works in North Carolina, where the fate of abortion rights is uncertain, according to The New York Times.
What about alternatives? “The short answer is that some of these medications work really well and sometimes much better than the nonteratogenic alternatives,” said Dr. Birru Talabi. “I’m worried about methotrexate. It has been used to induce abortions but is primarily used in the United States as a highly effective treatment for cancer as well as a myriad of rheumatic diseases. If legislators try to restrict access to methotrexate, we may see increasing disability and even death among people who need this medication but cannot access it.”
Rheumatologists aren’t the only physicians who are worrying about the fates of their patients in a new era of abortion restrictions. Gastroenterologist Sunanda Kane, MD, MSPH, of the Mayo Clinic, Rochester, Minn., said several teratogenic medications are used in her field to treat constipation, viral hepatitis, and inflammatory bowel disease.
“When treating women of childbearing age, there are usually alternatives. If we do prescribe a medication with a high teratogenic potential, we counsel and document that we have discussed two forms of birth control to avoid pregnancy. We usually do not prescribe a drug with teratogenic potential with the ‘out’ being an abortion if a pregnancy does occur,” she said. However, “if abortion is not even on the table as an option, we may be much less likely to prescribe these medications. This will be particularly true in patients who clearly do not have the means to travel to have an abortion in any situation.”
Abortion is expected to remain legal in Minnesota, where Dr. Kane practices, but it may be restricted or banned in nearby Wisconsin, depending on the state legislature. None of her patients have had abortions after becoming pregnant while taking the medications, she said, although she “did have a patient who because of her religious faith did not have an abortion after exposure and ended up with a stillbirth.”
The crackdown on abortion won’t just pose risks to patients who take potentially dangerous medications, physicians said. Dr. Kane said pregnancy itself is a significant risk for patients with “very active, uncontrolled gastrointestinal conditions where a pregnancy could be harmful to the mother’s health or result in offspring that are very unhealthy.” These include decompensated cirrhosis, uncontrolled Crohn’s disease or ulcerative colitis, refractory gastroparesis, uncontrolled celiac sprue, and chronic pancreatitis, she said.
“There have been times when after shared decisionmaking, a patient with very active inflammatory bowel disease has decided to terminate the pregnancy because of her own ongoing health issues,” she said. “Not having this option will potentially lead to disastrous results.”
Dr. Clowse, the Duke University rheumatologist, echoed Dr. Kane’s concerns about women who are too sick to bear children. “The removal of abortion rights puts the lives and quality of life for women with rheumatic disease at risk. For patients with lupus and other systemic rheumatic disease, pregnancy can be medically catastrophic, leading to permanent harm and even death to the woman and her offspring. I am worried that women in these conditions will die without lifesaving pregnancy terminations, due to worries about the legal consequences for their physicians.”
The U.S. Supreme Court’s ruling that overturned Roe v. Wade has also raised the prospect that the court could ultimately allow birth control to be restricted or outlawed.
While the ruling states that “nothing in this opinion should be understood to cast doubt on precedents that do not concern abortion,” Justice Clarence Thomas wrote a concurrence in which he said that the court should reconsider a 1960s ruling that forbids the banning of contraceptives. Republicans have dismissed concerns about bans being allowed, although Democrats, including the president and vice president, starkly warn that they could happen.
“If we as providers have to be concerned that there will be an unplanned pregnancy because of the lack of access to contraception,” Dr. Kane said, “this will have significant downstream consequences to the kind of care we can provide and might just drive some providers to not give care to female patients at all given this concern.”
The physicians quoted in this article report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
As states ban or limit abortion in the wake of the demise of Roe v. Wade, physicians are turning their attention to widely-used drugs that can cause birth defects. At issue: Should these drugs still be prescribed to women of childbearing age if they don’t have the option of terminating their pregnancies?
“Doctors are going to understandably be terrified that a patient may become pregnant using a teratogen that they have prescribed,” said University of Pittsburgh rheumatologist Mehret Birru Talabi, MD, PhD, who works in a state where the future of abortion rights is uncertain. “While this was a feared outcome before Roe v. Wade was overturned, abortion provided an escape hatch by which women could avoid having to continue a pregnancy and potentially raise a child with congenital anomalies. I believe that prescribing is going to become much more defensive and conservative. Some clinicians may choose not to prescribe these medications to patients who have childbearing potential, even if they don’t have much risk for pregnancy.”
Other physicians expressed similar concerns in interviews. Duke University, Durham, N.C., rheumatologist Megan E. B. Clowse, MD, MPH, fears that physicians will be wary of prescribing a variety of medications – including new ones for which there are few pregnancy data – if abortion is unavailable. “Women who receive these new or teratogenic medications will likely lose their reproductive autonomy and be forced to choose between having sexual relationships with men, obtaining procedures that make them permanently sterile, or using contraception that may cause intolerable side effects,” she said. “I am very concerned that young women with rheumatic disease will now be left with active disease resulting in joint damage and renal failure.”
Abortion is now banned in at least six states, according to The New York Times. That number may rise to 16 as more restrictions become law. Another five states aren’t expected to ban abortion soon but have implemented gestational age limits on abortion or are expected to adopt them. In another nine states, courts or lawmakers will decide whether abortion remains legal.
Only 20 states and the District of Columbia have firm abortion protections in place.
Numerous drugs are considered teratogens, which means they may cause birth defects. Thalidomide is the most infamous, but there are many more, including several used in rheumatology, dermatology, and gastroenterology. Among the most widely used teratogenic medications are the acne drugs isotretinoin and methotrexate, which are used to treat a variety of conditions, such as cancer, rheumatoid arthritis, and psoriasis.
Dr. Clowse, who helps manage an industry-supported website devoted to reproductive care for women with lupus (www.LupusPregnancy.org), noted that several drugs linked to birth defects and pregnancy loss are commonly prescribed in rheumatology.
“Methotrexate is the most common medication and has been the cornerstone of rheumatoid arthritis [treatment] for at least two decades,” she said. “Mycophenolate is our best medication to treat lupus nephritis, which is inflammation in the kidneys caused by lupus. This is a common complication for young women with lupus, and all of our guideline-recommended treatment regimens include a medication that causes pregnancy loss and birth defects, either mycophenolate or cyclophosphamide.”
Rheumatologists also prescribe a large number of new drugs for which there are few data about pregnancy risks. “It typically takes about two decades to have sufficient data about the safety of our medications,” she said.
Reflecting the sensitivity of the topic, Dr. Clowse made clear that her opinions don’t represent the views of her institution. She works in North Carolina, where the fate of abortion rights is uncertain, according to The New York Times.
What about alternatives? “The short answer is that some of these medications work really well and sometimes much better than the nonteratogenic alternatives,” said Dr. Birru Talabi. “I’m worried about methotrexate. It has been used to induce abortions but is primarily used in the United States as a highly effective treatment for cancer as well as a myriad of rheumatic diseases. If legislators try to restrict access to methotrexate, we may see increasing disability and even death among people who need this medication but cannot access it.”
Rheumatologists aren’t the only physicians who are worrying about the fates of their patients in a new era of abortion restrictions. Gastroenterologist Sunanda Kane, MD, MSPH, of the Mayo Clinic, Rochester, Minn., said several teratogenic medications are used in her field to treat constipation, viral hepatitis, and inflammatory bowel disease.
“When treating women of childbearing age, there are usually alternatives. If we do prescribe a medication with a high teratogenic potential, we counsel and document that we have discussed two forms of birth control to avoid pregnancy. We usually do not prescribe a drug with teratogenic potential with the ‘out’ being an abortion if a pregnancy does occur,” she said. However, “if abortion is not even on the table as an option, we may be much less likely to prescribe these medications. This will be particularly true in patients who clearly do not have the means to travel to have an abortion in any situation.”
Abortion is expected to remain legal in Minnesota, where Dr. Kane practices, but it may be restricted or banned in nearby Wisconsin, depending on the state legislature. None of her patients have had abortions after becoming pregnant while taking the medications, she said, although she “did have a patient who because of her religious faith did not have an abortion after exposure and ended up with a stillbirth.”
The crackdown on abortion won’t just pose risks to patients who take potentially dangerous medications, physicians said. Dr. Kane said pregnancy itself is a significant risk for patients with “very active, uncontrolled gastrointestinal conditions where a pregnancy could be harmful to the mother’s health or result in offspring that are very unhealthy.” These include decompensated cirrhosis, uncontrolled Crohn’s disease or ulcerative colitis, refractory gastroparesis, uncontrolled celiac sprue, and chronic pancreatitis, she said.
“There have been times when after shared decisionmaking, a patient with very active inflammatory bowel disease has decided to terminate the pregnancy because of her own ongoing health issues,” she said. “Not having this option will potentially lead to disastrous results.”
Dr. Clowse, the Duke University rheumatologist, echoed Dr. Kane’s concerns about women who are too sick to bear children. “The removal of abortion rights puts the lives and quality of life for women with rheumatic disease at risk. For patients with lupus and other systemic rheumatic disease, pregnancy can be medically catastrophic, leading to permanent harm and even death to the woman and her offspring. I am worried that women in these conditions will die without lifesaving pregnancy terminations, due to worries about the legal consequences for their physicians.”
The U.S. Supreme Court’s ruling that overturned Roe v. Wade has also raised the prospect that the court could ultimately allow birth control to be restricted or outlawed.
While the ruling states that “nothing in this opinion should be understood to cast doubt on precedents that do not concern abortion,” Justice Clarence Thomas wrote a concurrence in which he said that the court should reconsider a 1960s ruling that forbids the banning of contraceptives. Republicans have dismissed concerns about bans being allowed, although Democrats, including the president and vice president, starkly warn that they could happen.
“If we as providers have to be concerned that there will be an unplanned pregnancy because of the lack of access to contraception,” Dr. Kane said, “this will have significant downstream consequences to the kind of care we can provide and might just drive some providers to not give care to female patients at all given this concern.”
The physicians quoted in this article report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
As states ban or limit abortion in the wake of the demise of Roe v. Wade, physicians are turning their attention to widely-used drugs that can cause birth defects. At issue: Should these drugs still be prescribed to women of childbearing age if they don’t have the option of terminating their pregnancies?
“Doctors are going to understandably be terrified that a patient may become pregnant using a teratogen that they have prescribed,” said University of Pittsburgh rheumatologist Mehret Birru Talabi, MD, PhD, who works in a state where the future of abortion rights is uncertain. “While this was a feared outcome before Roe v. Wade was overturned, abortion provided an escape hatch by which women could avoid having to continue a pregnancy and potentially raise a child with congenital anomalies. I believe that prescribing is going to become much more defensive and conservative. Some clinicians may choose not to prescribe these medications to patients who have childbearing potential, even if they don’t have much risk for pregnancy.”
Other physicians expressed similar concerns in interviews. Duke University, Durham, N.C., rheumatologist Megan E. B. Clowse, MD, MPH, fears that physicians will be wary of prescribing a variety of medications – including new ones for which there are few pregnancy data – if abortion is unavailable. “Women who receive these new or teratogenic medications will likely lose their reproductive autonomy and be forced to choose between having sexual relationships with men, obtaining procedures that make them permanently sterile, or using contraception that may cause intolerable side effects,” she said. “I am very concerned that young women with rheumatic disease will now be left with active disease resulting in joint damage and renal failure.”
Abortion is now banned in at least six states, according to The New York Times. That number may rise to 16 as more restrictions become law. Another five states aren’t expected to ban abortion soon but have implemented gestational age limits on abortion or are expected to adopt them. In another nine states, courts or lawmakers will decide whether abortion remains legal.
Only 20 states and the District of Columbia have firm abortion protections in place.
Numerous drugs are considered teratogens, which means they may cause birth defects. Thalidomide is the most infamous, but there are many more, including several used in rheumatology, dermatology, and gastroenterology. Among the most widely used teratogenic medications are the acne drugs isotretinoin and methotrexate, which are used to treat a variety of conditions, such as cancer, rheumatoid arthritis, and psoriasis.
Dr. Clowse, who helps manage an industry-supported website devoted to reproductive care for women with lupus (www.LupusPregnancy.org), noted that several drugs linked to birth defects and pregnancy loss are commonly prescribed in rheumatology.
“Methotrexate is the most common medication and has been the cornerstone of rheumatoid arthritis [treatment] for at least two decades,” she said. “Mycophenolate is our best medication to treat lupus nephritis, which is inflammation in the kidneys caused by lupus. This is a common complication for young women with lupus, and all of our guideline-recommended treatment regimens include a medication that causes pregnancy loss and birth defects, either mycophenolate or cyclophosphamide.”
Rheumatologists also prescribe a large number of new drugs for which there are few data about pregnancy risks. “It typically takes about two decades to have sufficient data about the safety of our medications,” she said.
Reflecting the sensitivity of the topic, Dr. Clowse made clear that her opinions don’t represent the views of her institution. She works in North Carolina, where the fate of abortion rights is uncertain, according to The New York Times.
What about alternatives? “The short answer is that some of these medications work really well and sometimes much better than the nonteratogenic alternatives,” said Dr. Birru Talabi. “I’m worried about methotrexate. It has been used to induce abortions but is primarily used in the United States as a highly effective treatment for cancer as well as a myriad of rheumatic diseases. If legislators try to restrict access to methotrexate, we may see increasing disability and even death among people who need this medication but cannot access it.”
Rheumatologists aren’t the only physicians who are worrying about the fates of their patients in a new era of abortion restrictions. Gastroenterologist Sunanda Kane, MD, MSPH, of the Mayo Clinic, Rochester, Minn., said several teratogenic medications are used in her field to treat constipation, viral hepatitis, and inflammatory bowel disease.
“When treating women of childbearing age, there are usually alternatives. If we do prescribe a medication with a high teratogenic potential, we counsel and document that we have discussed two forms of birth control to avoid pregnancy. We usually do not prescribe a drug with teratogenic potential with the ‘out’ being an abortion if a pregnancy does occur,” she said. However, “if abortion is not even on the table as an option, we may be much less likely to prescribe these medications. This will be particularly true in patients who clearly do not have the means to travel to have an abortion in any situation.”
Abortion is expected to remain legal in Minnesota, where Dr. Kane practices, but it may be restricted or banned in nearby Wisconsin, depending on the state legislature. None of her patients have had abortions after becoming pregnant while taking the medications, she said, although she “did have a patient who because of her religious faith did not have an abortion after exposure and ended up with a stillbirth.”
The crackdown on abortion won’t just pose risks to patients who take potentially dangerous medications, physicians said. Dr. Kane said pregnancy itself is a significant risk for patients with “very active, uncontrolled gastrointestinal conditions where a pregnancy could be harmful to the mother’s health or result in offspring that are very unhealthy.” These include decompensated cirrhosis, uncontrolled Crohn’s disease or ulcerative colitis, refractory gastroparesis, uncontrolled celiac sprue, and chronic pancreatitis, she said.
“There have been times when after shared decisionmaking, a patient with very active inflammatory bowel disease has decided to terminate the pregnancy because of her own ongoing health issues,” she said. “Not having this option will potentially lead to disastrous results.”
Dr. Clowse, the Duke University rheumatologist, echoed Dr. Kane’s concerns about women who are too sick to bear children. “The removal of abortion rights puts the lives and quality of life for women with rheumatic disease at risk. For patients with lupus and other systemic rheumatic disease, pregnancy can be medically catastrophic, leading to permanent harm and even death to the woman and her offspring. I am worried that women in these conditions will die without lifesaving pregnancy terminations, due to worries about the legal consequences for their physicians.”
The U.S. Supreme Court’s ruling that overturned Roe v. Wade has also raised the prospect that the court could ultimately allow birth control to be restricted or outlawed.
While the ruling states that “nothing in this opinion should be understood to cast doubt on precedents that do not concern abortion,” Justice Clarence Thomas wrote a concurrence in which he said that the court should reconsider a 1960s ruling that forbids the banning of contraceptives. Republicans have dismissed concerns about bans being allowed, although Democrats, including the president and vice president, starkly warn that they could happen.
“If we as providers have to be concerned that there will be an unplanned pregnancy because of the lack of access to contraception,” Dr. Kane said, “this will have significant downstream consequences to the kind of care we can provide and might just drive some providers to not give care to female patients at all given this concern.”
The physicians quoted in this article report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Autoimmune disease linked to better late-stage breast cancer survival
CHICAGO – Comorbid autoimmune disease is associated with a greater chance of survival among women with stage IV breast cancer, according to a retrospective study presented at the annual meeting of the American Society of Clinical Oncology.
“It’s counterintuitive that, if you have two diseases instead of one, that you live longer, so then we had to scratch our heads a little bit and think about why these people are living longer,” said lead author Demitrios Dedousis, MD, University Hospitals, Case Medical Center, Cleveland.
Dr. Dedousis and colleagues conducted a retrospective analysis of patients from Surveillance, Epidemiology, and End Results–Medicare databases between 2007 and 2014 with breast cancer. The study included data from 137,324 patients diagnosed between 2007 and 2012, before the widespread use of immunotherapy. 27% of patients had an autoimmune disease, most commonly rheumatoid arthritis (23%), psoriasis (2.4%), and systemic lupus erythematosus (1.1%).
When all patients were included in the analysis, those with autoimmune disorders had slightly longer survival times, but these weren’t clinically significant. A subanalysis found a greater difference in survival.
The association appears more pronounced in metastatic cancer. Patients with stage 4 breast cancer and autoimmune disease had a longer mean overall survival (36 months vs. 30 months; hazard ratio, 1.46; P < .0001. Cancer-specific survival: HR, 1.39; P < .0001). Patients with autoimmune disease and stage 1-3 breast cancer had lower overall survival (P < .0001, P < 0.0001, and P = 0.026 respectively), compared with patients without autoimmune disease.
“What we thought was happening is that the lack of increased survival in stages 1 through 3 was hiding the increase in survival among the stage IV patients when looking at the overall cohort,” Dr. Dedousis said.
The retrospective nature of the study makes it impossible to draw any firm conclusions about causation. It could be that patients who have already been diagnosed with an autoimmune disease are more vigilant about going to health checkups. “There are other possible explanations, but the one that’s most interesting to us is that their immune system is involved in fighting the cancer. Our study certainly didn’t prove that, but it’s suggesting that’s a possibility,” Dr. Dedousis said.
He and his coauthors anticipate conducting similar studies in other cancers to see if there are similar relationships. Some preliminary work has already suggested something similar in lung cancer. “I think demonstrating this in a few kinds of cancer goes part of the way towards showing that this is a real biological phenomenon,” he said.
Another research avenue is to examine the immune systems and pathology specimens in patients with both an autoimmune disease and cancer to see if there is a greater immune response within the tumor. If so, that could suggest new immunotherapy strategies.
Another possibility is to look at the specific immune pathways within “protective” autoimmune conditions. “For the sake of argument, if we find a particular autoimmune condition is improving survival across multiple kinds of cancers, we could look at those pathways that are specifically involved in that autoimmune condition. It might help us identify a target for drug development,” Dr. Dedousis said.
Asked why a potential benefit might be more apparent in late-stage disease, he suggested that, in early-stage breast cancer, surgery and other treatments may be so effective that the immune system’s role only rarely makes a difference. It could play a larger role in late-stage disease when there are less effective therapies. It could also be that the immune system doesn’t recognize the cancer until it has spread beyond the regional lymph nodes on its way to metastasizing.
According to the National Cancer Institute, 10%-30% of people with cancer also have an autoimmune disease.
Dr. Dedousis has no relevant financial disclosures.
CHICAGO – Comorbid autoimmune disease is associated with a greater chance of survival among women with stage IV breast cancer, according to a retrospective study presented at the annual meeting of the American Society of Clinical Oncology.
“It’s counterintuitive that, if you have two diseases instead of one, that you live longer, so then we had to scratch our heads a little bit and think about why these people are living longer,” said lead author Demitrios Dedousis, MD, University Hospitals, Case Medical Center, Cleveland.
Dr. Dedousis and colleagues conducted a retrospective analysis of patients from Surveillance, Epidemiology, and End Results–Medicare databases between 2007 and 2014 with breast cancer. The study included data from 137,324 patients diagnosed between 2007 and 2012, before the widespread use of immunotherapy. 27% of patients had an autoimmune disease, most commonly rheumatoid arthritis (23%), psoriasis (2.4%), and systemic lupus erythematosus (1.1%).
When all patients were included in the analysis, those with autoimmune disorders had slightly longer survival times, but these weren’t clinically significant. A subanalysis found a greater difference in survival.
The association appears more pronounced in metastatic cancer. Patients with stage 4 breast cancer and autoimmune disease had a longer mean overall survival (36 months vs. 30 months; hazard ratio, 1.46; P < .0001. Cancer-specific survival: HR, 1.39; P < .0001). Patients with autoimmune disease and stage 1-3 breast cancer had lower overall survival (P < .0001, P < 0.0001, and P = 0.026 respectively), compared with patients without autoimmune disease.
“What we thought was happening is that the lack of increased survival in stages 1 through 3 was hiding the increase in survival among the stage IV patients when looking at the overall cohort,” Dr. Dedousis said.
The retrospective nature of the study makes it impossible to draw any firm conclusions about causation. It could be that patients who have already been diagnosed with an autoimmune disease are more vigilant about going to health checkups. “There are other possible explanations, but the one that’s most interesting to us is that their immune system is involved in fighting the cancer. Our study certainly didn’t prove that, but it’s suggesting that’s a possibility,” Dr. Dedousis said.
He and his coauthors anticipate conducting similar studies in other cancers to see if there are similar relationships. Some preliminary work has already suggested something similar in lung cancer. “I think demonstrating this in a few kinds of cancer goes part of the way towards showing that this is a real biological phenomenon,” he said.
Another research avenue is to examine the immune systems and pathology specimens in patients with both an autoimmune disease and cancer to see if there is a greater immune response within the tumor. If so, that could suggest new immunotherapy strategies.
Another possibility is to look at the specific immune pathways within “protective” autoimmune conditions. “For the sake of argument, if we find a particular autoimmune condition is improving survival across multiple kinds of cancers, we could look at those pathways that are specifically involved in that autoimmune condition. It might help us identify a target for drug development,” Dr. Dedousis said.
Asked why a potential benefit might be more apparent in late-stage disease, he suggested that, in early-stage breast cancer, surgery and other treatments may be so effective that the immune system’s role only rarely makes a difference. It could play a larger role in late-stage disease when there are less effective therapies. It could also be that the immune system doesn’t recognize the cancer until it has spread beyond the regional lymph nodes on its way to metastasizing.
According to the National Cancer Institute, 10%-30% of people with cancer also have an autoimmune disease.
Dr. Dedousis has no relevant financial disclosures.
CHICAGO – Comorbid autoimmune disease is associated with a greater chance of survival among women with stage IV breast cancer, according to a retrospective study presented at the annual meeting of the American Society of Clinical Oncology.
“It’s counterintuitive that, if you have two diseases instead of one, that you live longer, so then we had to scratch our heads a little bit and think about why these people are living longer,” said lead author Demitrios Dedousis, MD, University Hospitals, Case Medical Center, Cleveland.
Dr. Dedousis and colleagues conducted a retrospective analysis of patients from Surveillance, Epidemiology, and End Results–Medicare databases between 2007 and 2014 with breast cancer. The study included data from 137,324 patients diagnosed between 2007 and 2012, before the widespread use of immunotherapy. 27% of patients had an autoimmune disease, most commonly rheumatoid arthritis (23%), psoriasis (2.4%), and systemic lupus erythematosus (1.1%).
When all patients were included in the analysis, those with autoimmune disorders had slightly longer survival times, but these weren’t clinically significant. A subanalysis found a greater difference in survival.
The association appears more pronounced in metastatic cancer. Patients with stage 4 breast cancer and autoimmune disease had a longer mean overall survival (36 months vs. 30 months; hazard ratio, 1.46; P < .0001. Cancer-specific survival: HR, 1.39; P < .0001). Patients with autoimmune disease and stage 1-3 breast cancer had lower overall survival (P < .0001, P < 0.0001, and P = 0.026 respectively), compared with patients without autoimmune disease.
“What we thought was happening is that the lack of increased survival in stages 1 through 3 was hiding the increase in survival among the stage IV patients when looking at the overall cohort,” Dr. Dedousis said.
The retrospective nature of the study makes it impossible to draw any firm conclusions about causation. It could be that patients who have already been diagnosed with an autoimmune disease are more vigilant about going to health checkups. “There are other possible explanations, but the one that’s most interesting to us is that their immune system is involved in fighting the cancer. Our study certainly didn’t prove that, but it’s suggesting that’s a possibility,” Dr. Dedousis said.
He and his coauthors anticipate conducting similar studies in other cancers to see if there are similar relationships. Some preliminary work has already suggested something similar in lung cancer. “I think demonstrating this in a few kinds of cancer goes part of the way towards showing that this is a real biological phenomenon,” he said.
Another research avenue is to examine the immune systems and pathology specimens in patients with both an autoimmune disease and cancer to see if there is a greater immune response within the tumor. If so, that could suggest new immunotherapy strategies.
Another possibility is to look at the specific immune pathways within “protective” autoimmune conditions. “For the sake of argument, if we find a particular autoimmune condition is improving survival across multiple kinds of cancers, we could look at those pathways that are specifically involved in that autoimmune condition. It might help us identify a target for drug development,” Dr. Dedousis said.
Asked why a potential benefit might be more apparent in late-stage disease, he suggested that, in early-stage breast cancer, surgery and other treatments may be so effective that the immune system’s role only rarely makes a difference. It could play a larger role in late-stage disease when there are less effective therapies. It could also be that the immune system doesn’t recognize the cancer until it has spread beyond the regional lymph nodes on its way to metastasizing.
According to the National Cancer Institute, 10%-30% of people with cancer also have an autoimmune disease.
Dr. Dedousis has no relevant financial disclosures.
AT ASCO 2022
Deucravacitinib and orelabrutinib perform well in early lupus trials
Deucravacitinib and orelabrutinib – two novel oral drugs under investigation for the treatment of systemic lupus erythematosus (SLE) – have performed well in early clinical trials reported as late-breaking abstracts at the annual European Congress of Rheumatology.
In the phase 2 PAISLEY study, up to 58% of patients treated with deucravacitinib versus 34% of placebo-treated patients met the primary study endpoint of an SLE Responder Index-4 (SRI-4) after 38 weeks of treatment. Deucravacitinib also “achieved or meaningfully improved” all of the secondary endpoints set out in the 363-patient trial and was reported to have a safety and tolerability profile that was generally similar to placebo.
“Deucravacitinib shows promise as a novel therapy for SLE and warrants further investigation in phase 3 trials,” said Eric F. Morand, MD, PhD, a clinical rheumatologist and head of the School of Clinical Sciences at Monash University in Melbourne.
In a separate, ongoing phase 1b/2a study designed to evaluate orelabrutinib as a potential treatment for SLE, no safety concerns were seen with the investigational drug, along with “trending efficacy,” that supports “further studies in larger and longer-term trials,” according to the study’s investigators.
“What sets these two new drugs apart from currently available targeted therapies are their mode of action,” said Md Yuzaiful Md Yusof, MBChB, PhD, who was not involved in either study.
“The results from the PAISLEY study are promising, and it’s good to see the patients recruited were of diverse ethnicity [50%–60% were White],” added Dr. Md Yusof, a senior research fellow within the Leeds (England) Institute of Rheumatic and Musculoskeletal Medicine and a consultant rheumatologist at Leeds Teaching Hospitals NHS Trust.
He noted that the placebo rate was also low: “This could be contributed to by keeping the background prednisolone dose low, which is often a challenge in designing SLE trials.”
Deucravacitinib – the distant cousin of the JAK family?
“Deucravacitinib is a compound you might not have heard of before,” Dr. Morand said.
“It’s an inhibitor of a kinase called TYK2, which, broadly speaking, is a member of JAK [Janus kinase] family,” he explained in an interview. TYK2 regulates signal transduction downstream of receptors for interleukin (IL)-23 and IL-12 pathways and the type I interferon family.
“It’s a very finite set of cytokine signals” that are being blocked with deucravacitinib, he said, adding that this means it’s more directly targeting SLE pathogenic mechanisms than perhaps other JAK inhibitor compounds.
“It also means that it shouldn’t have some of the downsides of the other JAK inhibitors,” Dr. Morand said, “such as hematopoietic side effects, including cytopenias.”
The phase 2 PAISLEY study
This study involved 363 patients with moderate to severe, active SLE were recruited and randomized to receive placebo (n = 90) or one of three doses of deucravacitinib: 3 mg twice daily (n = 91), 6 mg twice daily (n = 93), or 12 mg once daily (n = 89). Most patients were also taking multiple background therapies, but this was similar across the four treatment arms.
The SRI-4 primary endpoint after 38 weeks of treatment was met by 34.4% of patients who received placebo, but 58.2% of those treated with deucravacitinib 3 mg twice daily (P = .0006 versus placebo), 49.5% (P = .021) of those treated with 6 mg twice daily, and 44.9% (P = .078) treated with 12 mg once daily.
“All secondary outcome measures were achieved or meaningfully improved at week 48, including SRI-4, BICLA [British Isles Lupus Assessment Group-based Composite Lupus Assessment], low-level disease activity state [LLDAS], reduction in skin disease and reduction in arthritis,” Dr. Morand said.
In addition, early biomarker results showed reductions in double-stranded DNA titers and increases in serum C4 complement with deucravacitinib across the duration of the study.
In discussion, Dr. Morand was asked about the seemingly negative or inverse dose response seen in the trial, with the best results seen with the 3-mg twice daily dose, then lower effects seen with two higher doses.
“Our analysis is that it’s not an inverse dose response, but rather a flat dose response above the 3-mg [twice daily] dose,” he said, noting that there was a higher dropout rate because of adverse effects in the 12-mg once daily group and those participants were recorded as nonresponders.
“We think what we’ve seen here is that 3 mg twice daily is a sufficient dose and there was no additional therapeutic gain above that.”
Rates of adverse events (AEs), serious AEs, and AEs of interest were overall fairly similar between deucravacitinib and placebo groups. The most common side effects seen with deucravacitinib were upper respiratory tract infection, nasopharyngitis, headache, and urinary tract infection. Skin reactions, such as acne, rash, and pruritis, among others, were more common in deucravacitinib- than in placebo-treated patients.
Importantly, Dr. Morand noted that there were no major cardiac events or thrombotic events and no deaths seen in the study. There was no signal for an increase in serious or opportunistic infections, including herpes zoster. There was no effect on common laboratory parameters.
“These are very encouraging results for patients with SLE,” Albert Roy, executive director of Lupus Therapeutics, said in a press release issued by the Lupus Research Alliance.
“We are honored to have played a role in this exciting work by helping to conduct this clinical trial through our Lupus Clinical Investigators Network of renowned North American academic centers.”
In an interview, he added: “We’re cautiously optimistic. Hopefully, if it continues to progress through phase 3, it’ll be the first oral agent that would be approved for lupus, notwithstanding prednisone and Plaquenil [hydroxychloroquine], back in the 50s.”
Orelabrutinib phase 2 study in SLE
Another approach to oral route of administration under investigation in SLE is the use of orelabrutinib, an irreversible inhibitor of Bruton’s tyrosine kinase (BTK) that was approved in China in December 2020 for the treatment of certain lymphomas and leukemias.
The rationale for testing it in SLE comes from two preclinical studies that had suggested a possible benefit in reducing disease activity, explained Zhanguo Li, MD, PhD, professor at Peking University People’s Hospital in Beijing. He presented the results of an ongoing randomized, double-blind, placebo-controlled, phase Ib/IIa dose-finding study comparing three different doses of orelabrutinib (50, 80, and 100 mg, once daily) to placebo.
As in the deucravacitinib trial, the SRI-4 was used to assess the potential efficacy of orelabrutinib, although in a much smaller patient population (n = 92) and at a shorter time point (12 weeks). Results showed an 11%-20% difference between the percentage of patients who met SRI-4 response criteria with orelabrutinib and those on placebo, at a respective 46.5%, 53.3%, 56.3% and 35.7%.
SLE Disease Activity Index (SLEDAI) scores showed a similar benefit of orelabrutinib over placebo, with 54%-63% and 30% of patients, respectively, achieving a score of 8 or more.
Adverse event rates were similar to those of placebo with most events being of mild or moderate nature. Three patients treated with orelabrutinib experienced serious adverse events, of which one was grade 3, but there were no reported deaths.
Pharmacokinetic and pharmacodynamic data showed a dose effect, and nearly complete occupancy of BTK was achieved at all dose levels for 24 hours, consistent with once-daily dosing.
“BTK plays an important role in B-cell regulation, thus B-cell and myeloid-cell blockade through BTK inhibition is an interesting potential new target for SLE,” Dr. Md Yusof said.
“Data from this early dose-ranging trial is encouraging. No major safety signal apart from mild reduction in lymphocyte and white cell counts,” he added.
“There are still plenty of challenges ahead for this drug’s development, particularly as none of the BTK inhibitors have yet to succeed in phase 3 trials in rheumatic and musculoskeletal diseases,” Dr. Md Yusof said.
Early days for both agents
While both seem currently promising, it’s very early days for deucravacitinib and orelabrutinib as possible new agents for SLE.
Aside from SLE, deucravacitinib is being tested across multiple immune-mediated diseases. This includes psoriasis, where two phase 3 trials – POETYK PSO-1 and POETYK PSO-2 – have already been completed, and psoriatic arthritis, where a phase 2 trial has been reported; all with positive results.
Phase 3 testing of deucravacitinib will go ahead and recruitment may start toward the end of this year, but it’ll take years to complete the studies, Dr. Morand said. Even if the trials prove positive, neither agent is going to be available for clinical use for several years.
A case in point is anifrolumab (Saphnelo), which Dr. Morand was involved in assessing. Despite gaining approval in the United States and across much of the world, the drug still going through reimbursement processes.
“The trial data, and lots of post hoc analysis, show clearly that it’s a major step forward in treating lupus,” he said in an interview, but “access is limited in most places, so hands-on experience with that new treatment is still limited for most clinicians.”
As for all the other new targeted approaches under investigation, “although there’s a lot of trial activity, there’s still a couple of years away before any of the current trials deliver new treatment. That’s if they provide positive findings. Indeed, there have been numerous agents that have shown promise at phase 2 but then fall at the final phase 3 hurdle, including baricitinib, which Dr. Morand reported on in a separate poster presentation.
Phase 3 data proved disappointing: “Results are not sufficiently positive for that to go forward,” he said, adding that “transitioning from a successful phase 2 to a successful phase 3 is challenging, and many products have failed.”
Dr. Morand added: “It’s a very exciting time to be in lupus research, and there’s a lot of optimism about the future. But when I go back to my clinic tomorrow, I treat my patients exactly the same as I did last week and last year.”
It’s yet to be seen if deucravacitinib will fulfill its early promise, but it’s off to an impressive start. A positive for patients is that it’s an oral drug, with the potential to improve access to treatment across the world where getting infusions may be an issue.
“These are some of the most exciting data that I’ve seen at the phase 2 level in terms of effect size across all the readouts that are used,” Dr. Morand said. “There’s no guesswork here; it worked across all the measures. That’s very reassuring.”
The PAISLEY study was sponsored by Bristol-Myers Squibb. Dr. Morand has acted as a consultant to the company and received research support for the conduct of the trial. He disclosed acting as a consultant or receiving research funding from AbbVie, Amgen, AstraZeneca, Biogen, Eli Lilly, EMD Serono, Janssen, Genentech, Servier, Novartis, and UCB. Mr. Roy is the executive director of Lupus Therapeutics, which manages the Lupus Clinical Investigators Network based in North America. Lupus Therapeutics is the clinical trials arm of the Lupus Research Alliance, a nongovernmental, nonprofit funder of lupus research worldwide. The orelabrutinib study was sponsored by InnoCare Pharma. Dr. Li is the principal investigator for the trial but had no conflicts of interest to declare. Dr. Md Yusof disclosed receiving consultancy fees from Aurinia Pharmaceuticals.
Deucravacitinib and orelabrutinib – two novel oral drugs under investigation for the treatment of systemic lupus erythematosus (SLE) – have performed well in early clinical trials reported as late-breaking abstracts at the annual European Congress of Rheumatology.
In the phase 2 PAISLEY study, up to 58% of patients treated with deucravacitinib versus 34% of placebo-treated patients met the primary study endpoint of an SLE Responder Index-4 (SRI-4) after 38 weeks of treatment. Deucravacitinib also “achieved or meaningfully improved” all of the secondary endpoints set out in the 363-patient trial and was reported to have a safety and tolerability profile that was generally similar to placebo.
“Deucravacitinib shows promise as a novel therapy for SLE and warrants further investigation in phase 3 trials,” said Eric F. Morand, MD, PhD, a clinical rheumatologist and head of the School of Clinical Sciences at Monash University in Melbourne.
In a separate, ongoing phase 1b/2a study designed to evaluate orelabrutinib as a potential treatment for SLE, no safety concerns were seen with the investigational drug, along with “trending efficacy,” that supports “further studies in larger and longer-term trials,” according to the study’s investigators.
“What sets these two new drugs apart from currently available targeted therapies are their mode of action,” said Md Yuzaiful Md Yusof, MBChB, PhD, who was not involved in either study.
“The results from the PAISLEY study are promising, and it’s good to see the patients recruited were of diverse ethnicity [50%–60% were White],” added Dr. Md Yusof, a senior research fellow within the Leeds (England) Institute of Rheumatic and Musculoskeletal Medicine and a consultant rheumatologist at Leeds Teaching Hospitals NHS Trust.
He noted that the placebo rate was also low: “This could be contributed to by keeping the background prednisolone dose low, which is often a challenge in designing SLE trials.”
Deucravacitinib – the distant cousin of the JAK family?
“Deucravacitinib is a compound you might not have heard of before,” Dr. Morand said.
“It’s an inhibitor of a kinase called TYK2, which, broadly speaking, is a member of JAK [Janus kinase] family,” he explained in an interview. TYK2 regulates signal transduction downstream of receptors for interleukin (IL)-23 and IL-12 pathways and the type I interferon family.
“It’s a very finite set of cytokine signals” that are being blocked with deucravacitinib, he said, adding that this means it’s more directly targeting SLE pathogenic mechanisms than perhaps other JAK inhibitor compounds.
“It also means that it shouldn’t have some of the downsides of the other JAK inhibitors,” Dr. Morand said, “such as hematopoietic side effects, including cytopenias.”
The phase 2 PAISLEY study
This study involved 363 patients with moderate to severe, active SLE were recruited and randomized to receive placebo (n = 90) or one of three doses of deucravacitinib: 3 mg twice daily (n = 91), 6 mg twice daily (n = 93), or 12 mg once daily (n = 89). Most patients were also taking multiple background therapies, but this was similar across the four treatment arms.
The SRI-4 primary endpoint after 38 weeks of treatment was met by 34.4% of patients who received placebo, but 58.2% of those treated with deucravacitinib 3 mg twice daily (P = .0006 versus placebo), 49.5% (P = .021) of those treated with 6 mg twice daily, and 44.9% (P = .078) treated with 12 mg once daily.
“All secondary outcome measures were achieved or meaningfully improved at week 48, including SRI-4, BICLA [British Isles Lupus Assessment Group-based Composite Lupus Assessment], low-level disease activity state [LLDAS], reduction in skin disease and reduction in arthritis,” Dr. Morand said.
In addition, early biomarker results showed reductions in double-stranded DNA titers and increases in serum C4 complement with deucravacitinib across the duration of the study.
In discussion, Dr. Morand was asked about the seemingly negative or inverse dose response seen in the trial, with the best results seen with the 3-mg twice daily dose, then lower effects seen with two higher doses.
“Our analysis is that it’s not an inverse dose response, but rather a flat dose response above the 3-mg [twice daily] dose,” he said, noting that there was a higher dropout rate because of adverse effects in the 12-mg once daily group and those participants were recorded as nonresponders.
“We think what we’ve seen here is that 3 mg twice daily is a sufficient dose and there was no additional therapeutic gain above that.”
Rates of adverse events (AEs), serious AEs, and AEs of interest were overall fairly similar between deucravacitinib and placebo groups. The most common side effects seen with deucravacitinib were upper respiratory tract infection, nasopharyngitis, headache, and urinary tract infection. Skin reactions, such as acne, rash, and pruritis, among others, were more common in deucravacitinib- than in placebo-treated patients.
Importantly, Dr. Morand noted that there were no major cardiac events or thrombotic events and no deaths seen in the study. There was no signal for an increase in serious or opportunistic infections, including herpes zoster. There was no effect on common laboratory parameters.
“These are very encouraging results for patients with SLE,” Albert Roy, executive director of Lupus Therapeutics, said in a press release issued by the Lupus Research Alliance.
“We are honored to have played a role in this exciting work by helping to conduct this clinical trial through our Lupus Clinical Investigators Network of renowned North American academic centers.”
In an interview, he added: “We’re cautiously optimistic. Hopefully, if it continues to progress through phase 3, it’ll be the first oral agent that would be approved for lupus, notwithstanding prednisone and Plaquenil [hydroxychloroquine], back in the 50s.”
Orelabrutinib phase 2 study in SLE
Another approach to oral route of administration under investigation in SLE is the use of orelabrutinib, an irreversible inhibitor of Bruton’s tyrosine kinase (BTK) that was approved in China in December 2020 for the treatment of certain lymphomas and leukemias.
The rationale for testing it in SLE comes from two preclinical studies that had suggested a possible benefit in reducing disease activity, explained Zhanguo Li, MD, PhD, professor at Peking University People’s Hospital in Beijing. He presented the results of an ongoing randomized, double-blind, placebo-controlled, phase Ib/IIa dose-finding study comparing three different doses of orelabrutinib (50, 80, and 100 mg, once daily) to placebo.
As in the deucravacitinib trial, the SRI-4 was used to assess the potential efficacy of orelabrutinib, although in a much smaller patient population (n = 92) and at a shorter time point (12 weeks). Results showed an 11%-20% difference between the percentage of patients who met SRI-4 response criteria with orelabrutinib and those on placebo, at a respective 46.5%, 53.3%, 56.3% and 35.7%.
SLE Disease Activity Index (SLEDAI) scores showed a similar benefit of orelabrutinib over placebo, with 54%-63% and 30% of patients, respectively, achieving a score of 8 or more.
Adverse event rates were similar to those of placebo with most events being of mild or moderate nature. Three patients treated with orelabrutinib experienced serious adverse events, of which one was grade 3, but there were no reported deaths.
Pharmacokinetic and pharmacodynamic data showed a dose effect, and nearly complete occupancy of BTK was achieved at all dose levels for 24 hours, consistent with once-daily dosing.
“BTK plays an important role in B-cell regulation, thus B-cell and myeloid-cell blockade through BTK inhibition is an interesting potential new target for SLE,” Dr. Md Yusof said.
“Data from this early dose-ranging trial is encouraging. No major safety signal apart from mild reduction in lymphocyte and white cell counts,” he added.
“There are still plenty of challenges ahead for this drug’s development, particularly as none of the BTK inhibitors have yet to succeed in phase 3 trials in rheumatic and musculoskeletal diseases,” Dr. Md Yusof said.
Early days for both agents
While both seem currently promising, it’s very early days for deucravacitinib and orelabrutinib as possible new agents for SLE.
Aside from SLE, deucravacitinib is being tested across multiple immune-mediated diseases. This includes psoriasis, where two phase 3 trials – POETYK PSO-1 and POETYK PSO-2 – have already been completed, and psoriatic arthritis, where a phase 2 trial has been reported; all with positive results.
Phase 3 testing of deucravacitinib will go ahead and recruitment may start toward the end of this year, but it’ll take years to complete the studies, Dr. Morand said. Even if the trials prove positive, neither agent is going to be available for clinical use for several years.
A case in point is anifrolumab (Saphnelo), which Dr. Morand was involved in assessing. Despite gaining approval in the United States and across much of the world, the drug still going through reimbursement processes.
“The trial data, and lots of post hoc analysis, show clearly that it’s a major step forward in treating lupus,” he said in an interview, but “access is limited in most places, so hands-on experience with that new treatment is still limited for most clinicians.”
As for all the other new targeted approaches under investigation, “although there’s a lot of trial activity, there’s still a couple of years away before any of the current trials deliver new treatment. That’s if they provide positive findings. Indeed, there have been numerous agents that have shown promise at phase 2 but then fall at the final phase 3 hurdle, including baricitinib, which Dr. Morand reported on in a separate poster presentation.
Phase 3 data proved disappointing: “Results are not sufficiently positive for that to go forward,” he said, adding that “transitioning from a successful phase 2 to a successful phase 3 is challenging, and many products have failed.”
Dr. Morand added: “It’s a very exciting time to be in lupus research, and there’s a lot of optimism about the future. But when I go back to my clinic tomorrow, I treat my patients exactly the same as I did last week and last year.”
It’s yet to be seen if deucravacitinib will fulfill its early promise, but it’s off to an impressive start. A positive for patients is that it’s an oral drug, with the potential to improve access to treatment across the world where getting infusions may be an issue.
“These are some of the most exciting data that I’ve seen at the phase 2 level in terms of effect size across all the readouts that are used,” Dr. Morand said. “There’s no guesswork here; it worked across all the measures. That’s very reassuring.”
The PAISLEY study was sponsored by Bristol-Myers Squibb. Dr. Morand has acted as a consultant to the company and received research support for the conduct of the trial. He disclosed acting as a consultant or receiving research funding from AbbVie, Amgen, AstraZeneca, Biogen, Eli Lilly, EMD Serono, Janssen, Genentech, Servier, Novartis, and UCB. Mr. Roy is the executive director of Lupus Therapeutics, which manages the Lupus Clinical Investigators Network based in North America. Lupus Therapeutics is the clinical trials arm of the Lupus Research Alliance, a nongovernmental, nonprofit funder of lupus research worldwide. The orelabrutinib study was sponsored by InnoCare Pharma. Dr. Li is the principal investigator for the trial but had no conflicts of interest to declare. Dr. Md Yusof disclosed receiving consultancy fees from Aurinia Pharmaceuticals.
Deucravacitinib and orelabrutinib – two novel oral drugs under investigation for the treatment of systemic lupus erythematosus (SLE) – have performed well in early clinical trials reported as late-breaking abstracts at the annual European Congress of Rheumatology.
In the phase 2 PAISLEY study, up to 58% of patients treated with deucravacitinib versus 34% of placebo-treated patients met the primary study endpoint of an SLE Responder Index-4 (SRI-4) after 38 weeks of treatment. Deucravacitinib also “achieved or meaningfully improved” all of the secondary endpoints set out in the 363-patient trial and was reported to have a safety and tolerability profile that was generally similar to placebo.
“Deucravacitinib shows promise as a novel therapy for SLE and warrants further investigation in phase 3 trials,” said Eric F. Morand, MD, PhD, a clinical rheumatologist and head of the School of Clinical Sciences at Monash University in Melbourne.
In a separate, ongoing phase 1b/2a study designed to evaluate orelabrutinib as a potential treatment for SLE, no safety concerns were seen with the investigational drug, along with “trending efficacy,” that supports “further studies in larger and longer-term trials,” according to the study’s investigators.
“What sets these two new drugs apart from currently available targeted therapies are their mode of action,” said Md Yuzaiful Md Yusof, MBChB, PhD, who was not involved in either study.
“The results from the PAISLEY study are promising, and it’s good to see the patients recruited were of diverse ethnicity [50%–60% were White],” added Dr. Md Yusof, a senior research fellow within the Leeds (England) Institute of Rheumatic and Musculoskeletal Medicine and a consultant rheumatologist at Leeds Teaching Hospitals NHS Trust.
He noted that the placebo rate was also low: “This could be contributed to by keeping the background prednisolone dose low, which is often a challenge in designing SLE trials.”
Deucravacitinib – the distant cousin of the JAK family?
“Deucravacitinib is a compound you might not have heard of before,” Dr. Morand said.
“It’s an inhibitor of a kinase called TYK2, which, broadly speaking, is a member of JAK [Janus kinase] family,” he explained in an interview. TYK2 regulates signal transduction downstream of receptors for interleukin (IL)-23 and IL-12 pathways and the type I interferon family.
“It’s a very finite set of cytokine signals” that are being blocked with deucravacitinib, he said, adding that this means it’s more directly targeting SLE pathogenic mechanisms than perhaps other JAK inhibitor compounds.
“It also means that it shouldn’t have some of the downsides of the other JAK inhibitors,” Dr. Morand said, “such as hematopoietic side effects, including cytopenias.”
The phase 2 PAISLEY study
This study involved 363 patients with moderate to severe, active SLE were recruited and randomized to receive placebo (n = 90) or one of three doses of deucravacitinib: 3 mg twice daily (n = 91), 6 mg twice daily (n = 93), or 12 mg once daily (n = 89). Most patients were also taking multiple background therapies, but this was similar across the four treatment arms.
The SRI-4 primary endpoint after 38 weeks of treatment was met by 34.4% of patients who received placebo, but 58.2% of those treated with deucravacitinib 3 mg twice daily (P = .0006 versus placebo), 49.5% (P = .021) of those treated with 6 mg twice daily, and 44.9% (P = .078) treated with 12 mg once daily.
“All secondary outcome measures were achieved or meaningfully improved at week 48, including SRI-4, BICLA [British Isles Lupus Assessment Group-based Composite Lupus Assessment], low-level disease activity state [LLDAS], reduction in skin disease and reduction in arthritis,” Dr. Morand said.
In addition, early biomarker results showed reductions in double-stranded DNA titers and increases in serum C4 complement with deucravacitinib across the duration of the study.
In discussion, Dr. Morand was asked about the seemingly negative or inverse dose response seen in the trial, with the best results seen with the 3-mg twice daily dose, then lower effects seen with two higher doses.
“Our analysis is that it’s not an inverse dose response, but rather a flat dose response above the 3-mg [twice daily] dose,” he said, noting that there was a higher dropout rate because of adverse effects in the 12-mg once daily group and those participants were recorded as nonresponders.
“We think what we’ve seen here is that 3 mg twice daily is a sufficient dose and there was no additional therapeutic gain above that.”
Rates of adverse events (AEs), serious AEs, and AEs of interest were overall fairly similar between deucravacitinib and placebo groups. The most common side effects seen with deucravacitinib were upper respiratory tract infection, nasopharyngitis, headache, and urinary tract infection. Skin reactions, such as acne, rash, and pruritis, among others, were more common in deucravacitinib- than in placebo-treated patients.
Importantly, Dr. Morand noted that there were no major cardiac events or thrombotic events and no deaths seen in the study. There was no signal for an increase in serious or opportunistic infections, including herpes zoster. There was no effect on common laboratory parameters.
“These are very encouraging results for patients with SLE,” Albert Roy, executive director of Lupus Therapeutics, said in a press release issued by the Lupus Research Alliance.
“We are honored to have played a role in this exciting work by helping to conduct this clinical trial through our Lupus Clinical Investigators Network of renowned North American academic centers.”
In an interview, he added: “We’re cautiously optimistic. Hopefully, if it continues to progress through phase 3, it’ll be the first oral agent that would be approved for lupus, notwithstanding prednisone and Plaquenil [hydroxychloroquine], back in the 50s.”
Orelabrutinib phase 2 study in SLE
Another approach to oral route of administration under investigation in SLE is the use of orelabrutinib, an irreversible inhibitor of Bruton’s tyrosine kinase (BTK) that was approved in China in December 2020 for the treatment of certain lymphomas and leukemias.
The rationale for testing it in SLE comes from two preclinical studies that had suggested a possible benefit in reducing disease activity, explained Zhanguo Li, MD, PhD, professor at Peking University People’s Hospital in Beijing. He presented the results of an ongoing randomized, double-blind, placebo-controlled, phase Ib/IIa dose-finding study comparing three different doses of orelabrutinib (50, 80, and 100 mg, once daily) to placebo.
As in the deucravacitinib trial, the SRI-4 was used to assess the potential efficacy of orelabrutinib, although in a much smaller patient population (n = 92) and at a shorter time point (12 weeks). Results showed an 11%-20% difference between the percentage of patients who met SRI-4 response criteria with orelabrutinib and those on placebo, at a respective 46.5%, 53.3%, 56.3% and 35.7%.
SLE Disease Activity Index (SLEDAI) scores showed a similar benefit of orelabrutinib over placebo, with 54%-63% and 30% of patients, respectively, achieving a score of 8 or more.
Adverse event rates were similar to those of placebo with most events being of mild or moderate nature. Three patients treated with orelabrutinib experienced serious adverse events, of which one was grade 3, but there were no reported deaths.
Pharmacokinetic and pharmacodynamic data showed a dose effect, and nearly complete occupancy of BTK was achieved at all dose levels for 24 hours, consistent with once-daily dosing.
“BTK plays an important role in B-cell regulation, thus B-cell and myeloid-cell blockade through BTK inhibition is an interesting potential new target for SLE,” Dr. Md Yusof said.
“Data from this early dose-ranging trial is encouraging. No major safety signal apart from mild reduction in lymphocyte and white cell counts,” he added.
“There are still plenty of challenges ahead for this drug’s development, particularly as none of the BTK inhibitors have yet to succeed in phase 3 trials in rheumatic and musculoskeletal diseases,” Dr. Md Yusof said.
Early days for both agents
While both seem currently promising, it’s very early days for deucravacitinib and orelabrutinib as possible new agents for SLE.
Aside from SLE, deucravacitinib is being tested across multiple immune-mediated diseases. This includes psoriasis, where two phase 3 trials – POETYK PSO-1 and POETYK PSO-2 – have already been completed, and psoriatic arthritis, where a phase 2 trial has been reported; all with positive results.
Phase 3 testing of deucravacitinib will go ahead and recruitment may start toward the end of this year, but it’ll take years to complete the studies, Dr. Morand said. Even if the trials prove positive, neither agent is going to be available for clinical use for several years.
A case in point is anifrolumab (Saphnelo), which Dr. Morand was involved in assessing. Despite gaining approval in the United States and across much of the world, the drug still going through reimbursement processes.
“The trial data, and lots of post hoc analysis, show clearly that it’s a major step forward in treating lupus,” he said in an interview, but “access is limited in most places, so hands-on experience with that new treatment is still limited for most clinicians.”
As for all the other new targeted approaches under investigation, “although there’s a lot of trial activity, there’s still a couple of years away before any of the current trials deliver new treatment. That’s if they provide positive findings. Indeed, there have been numerous agents that have shown promise at phase 2 but then fall at the final phase 3 hurdle, including baricitinib, which Dr. Morand reported on in a separate poster presentation.
Phase 3 data proved disappointing: “Results are not sufficiently positive for that to go forward,” he said, adding that “transitioning from a successful phase 2 to a successful phase 3 is challenging, and many products have failed.”
Dr. Morand added: “It’s a very exciting time to be in lupus research, and there’s a lot of optimism about the future. But when I go back to my clinic tomorrow, I treat my patients exactly the same as I did last week and last year.”
It’s yet to be seen if deucravacitinib will fulfill its early promise, but it’s off to an impressive start. A positive for patients is that it’s an oral drug, with the potential to improve access to treatment across the world where getting infusions may be an issue.
“These are some of the most exciting data that I’ve seen at the phase 2 level in terms of effect size across all the readouts that are used,” Dr. Morand said. “There’s no guesswork here; it worked across all the measures. That’s very reassuring.”
The PAISLEY study was sponsored by Bristol-Myers Squibb. Dr. Morand has acted as a consultant to the company and received research support for the conduct of the trial. He disclosed acting as a consultant or receiving research funding from AbbVie, Amgen, AstraZeneca, Biogen, Eli Lilly, EMD Serono, Janssen, Genentech, Servier, Novartis, and UCB. Mr. Roy is the executive director of Lupus Therapeutics, which manages the Lupus Clinical Investigators Network based in North America. Lupus Therapeutics is the clinical trials arm of the Lupus Research Alliance, a nongovernmental, nonprofit funder of lupus research worldwide. The orelabrutinib study was sponsored by InnoCare Pharma. Dr. Li is the principal investigator for the trial but had no conflicts of interest to declare. Dr. Md Yusof disclosed receiving consultancy fees from Aurinia Pharmaceuticals.
FROM THE EULAR 2022 CONGRESS
Stopping immunosuppressives in lupus nephritis isn’t noninferior to continuing
COPENHAGEN – Discontinuing maintenance immunosuppressive therapy (IST) in patients with proliferative lupus nephritis in remission proved less effective than continuing it in terms of the rates of renal relapse and severe systemic lupus erythematosus (SLE) flare, results from the WIN-Lupus trial show.
Lead researcher Noemie Jourde-Chiche, MD, assistant professor at Aix-Marseille (France) University, presented the results at the annual European Congress of Rheumatology.
The randomized, controlled trial aimed to determine the optimal duration of maintenance IST for proliferative lupus nephritis, by asking whether discontinuation of such therapy after 2-3 years was noninferior to IST continuation for 2 more years.
“This is the first randomized IST trial in proliferative lupus nephritis,” Dr. Jourde-Chiche reported. “We found that noninferiority of IST discontinuation was not demonstrated, and those who discontinued had a higher risk of SLE flares, but the majority of patients who discontinued did not experience flare.”
Regarding the incidence of renal relapse, no statistically significant difference was found between patients who continued versus those who discontinued IST.
Rheumatologist Christophe Richez, MD, of the Groupe Hospitalier Pellegrin-CHU de Bordeaux (France) welcomed the trial. “The work does not find a significant difference but suggests that with more power, the difference would have been significant,” he said in an interview.
He added that a significant number of patients refused to enter the study for several reasons, including scheduling a pregnancy and fear of relapse after stopping the treatment. “This in itself shows that we need this type of study to know if we can stop the treatment to plan a pregnancy or if a relay treatment is necessary. These data also mean we can now better inform our patients about the benefit-risk [profile] of continuing treatment or not.”
Dr. Richez, who was not involved in the trial, noted that the data provide information that strongly suggest more research is needed to better determine which patients are at risk of relapse, and consequently, for which patients is discontinuation possible. “There’s also a need for further analysis of tolerance to the immunosuppressive drugs according to the strategy.”
Finally, he referred to the issue around therapeutic adherence that is often faced with management of lupus. “The data from this study will allow us to better explain to patients the need to continue or discontinue their treatments, including hydroxychloroquine, and also the possibility of decreasing the immunosuppressive drugs to half-dose.”
Maintenance therapy in lupus nephritis: To continue or not?
Conducted in 28 French centers, participants had class III or IV lupus nephritis with active lesions and had previously received induction IST of cyclophosphamide or mycophenolate mofetil with hydroxychloroquine and glucocorticoids. Maintenance IST was azathioprine or mycophenolate mofetil, hydroxychloroquine, and possibly low-dose glucocorticoids (below 10 mg/day).
A total of 96 patients were randomized into two groups: continuation of maintenance therapy (azathioprine or mycophenolate mofetil for 2-3 more years, hydroxychloroquine, and possibly low-dose glucocorticoids (below 10 mg/day), or discontinuation of maintenance therapy (azathioprine or mycophenolate mofetil) over 3 months.
Both study arms were similar with a mean age of 36-37 years, and 82%-86% of patients were female, 59%-67% were White. They had a mean disease duration of 7-9 years, and 72%-80% had experienced their first flare of proliferative nephritis. A total of 54%-65% had received cyclophosphamide induction therapy, and 75%-81% were on mycophenolate mofetil maintenance therapy that had been ongoing for a mean of 2.8 years. Tests of patients’ kidney function revealed a mean serum creatinine of 67-72 micromol/L and estimated glomerular filtration rate (eGFR) of 94-101 mL/min per 1.73 m2. The mean SLE Disease Activity Index score was around 2.
Follow-up visits were conducted every 3 months for 2 years, and the trial had a primary end point of renal relapse rate at 24 months (confirmed by kidney biopsy), while secondary endpoints included rate of severe SLE flares, survival without renal relapse, or severe flare and adverse events, among others.
Patients were excluded if they were not taking hydroxychloroquine, had extrarenal SLE, an eGFR less than 30 mL/min per 1.73 m2 or stage VI disease. After some participants did not finish the trial because of pregnancy, wish for pregnancy, or adverse events, data from a total of 40 in the IST continuation group and 44 in the discontinuation group were analyzed.
Noninferiority of discontinuation versus continuation not shown
A total of 12.5% in the continuation arm experienced renal relapse over 24 months, compared with 27.3% in the discontinuation arm (P = .079).
“The endpoint of noninferiority of discontinuation of immunosuppressive therapy was not shown, but no statistically significant difference was found between groups for renal relapse,” Dr. Jourde-Chiche said.
Severe SLE flares (renal or extrarenal) occurred significantly less often over 24 months in the continuation arm at 12.5% versus 31.8% in the discontinuation arm (P = .034).
However, Dr. Jourde-Chiche pointed out that “the majority of patients who discontinued treatment did not experience a flare.”
No differences were seen between groups with respect to any secondary endpoints. “Fortunately, no patients died or reached end-stage renal disease, and overall, the adverse events did not differ between groups,” she reported.
The study identified several risk factors for renal relapse, among which were low complement component 3 and higher baseline urinary protein to creatinine ratio.
Longer immunosuppressive therapy prediscontinuation leads to better results
Gabriella Moroni, MD, of the nephrology unit at Ospedale Maggiore Policlinico, Milan, authored a review of studies that attempted to interrupt glucocorticoids and other immunosuppressive agents in lupus nephritis and in SLE. Her review concluded that “the available data suggest that therapy withdrawal is feasible at least in patients enjoying a complete clinical remission after a prolonged therapy.”
Asked to comment on the French study, Dr. Moroni said the trial was very welcome. “In our long-term experience, we have stopped IST in 73 patients with lupus nephritis, followed for a mean of 23 years. Of these, 32 never reassumed therapy and 20 had at least one flare.”
Dr. Moroni noted that those participants who did not experience flares had significantly longer IST and longer remission before discontinuation and took hydroxychloroquine more frequently.
“We feel that, to prevent severe flares, firstly, lupus nephritis should be in complete remission for at least 3 years, and secondly, patients should have received IST for at least 5 years before discontinuation; immunosuppressive drugs should be tapered off very slowly and after strict clinical surveillance, and finally, hydroxychloroquine can prevent extrarenal flares.
Dr. Jourde-Chiche reported serving on a speakers bureau for Vifor Pharma and receiving grant/research support from Fresenius Medical Care. Some coauthors reported financial ties to many pharmaceutical companies. Dr. Richez said he has received fees for lectures or boards from GlaxoSmithKline, AstraZeneca, and Novartis. Dr. Moroni had no relevant disclosures.
COPENHAGEN – Discontinuing maintenance immunosuppressive therapy (IST) in patients with proliferative lupus nephritis in remission proved less effective than continuing it in terms of the rates of renal relapse and severe systemic lupus erythematosus (SLE) flare, results from the WIN-Lupus trial show.
Lead researcher Noemie Jourde-Chiche, MD, assistant professor at Aix-Marseille (France) University, presented the results at the annual European Congress of Rheumatology.
The randomized, controlled trial aimed to determine the optimal duration of maintenance IST for proliferative lupus nephritis, by asking whether discontinuation of such therapy after 2-3 years was noninferior to IST continuation for 2 more years.
“This is the first randomized IST trial in proliferative lupus nephritis,” Dr. Jourde-Chiche reported. “We found that noninferiority of IST discontinuation was not demonstrated, and those who discontinued had a higher risk of SLE flares, but the majority of patients who discontinued did not experience flare.”
Regarding the incidence of renal relapse, no statistically significant difference was found between patients who continued versus those who discontinued IST.
Rheumatologist Christophe Richez, MD, of the Groupe Hospitalier Pellegrin-CHU de Bordeaux (France) welcomed the trial. “The work does not find a significant difference but suggests that with more power, the difference would have been significant,” he said in an interview.
He added that a significant number of patients refused to enter the study for several reasons, including scheduling a pregnancy and fear of relapse after stopping the treatment. “This in itself shows that we need this type of study to know if we can stop the treatment to plan a pregnancy or if a relay treatment is necessary. These data also mean we can now better inform our patients about the benefit-risk [profile] of continuing treatment or not.”
Dr. Richez, who was not involved in the trial, noted that the data provide information that strongly suggest more research is needed to better determine which patients are at risk of relapse, and consequently, for which patients is discontinuation possible. “There’s also a need for further analysis of tolerance to the immunosuppressive drugs according to the strategy.”
Finally, he referred to the issue around therapeutic adherence that is often faced with management of lupus. “The data from this study will allow us to better explain to patients the need to continue or discontinue their treatments, including hydroxychloroquine, and also the possibility of decreasing the immunosuppressive drugs to half-dose.”
Maintenance therapy in lupus nephritis: To continue or not?
Conducted in 28 French centers, participants had class III or IV lupus nephritis with active lesions and had previously received induction IST of cyclophosphamide or mycophenolate mofetil with hydroxychloroquine and glucocorticoids. Maintenance IST was azathioprine or mycophenolate mofetil, hydroxychloroquine, and possibly low-dose glucocorticoids (below 10 mg/day).
A total of 96 patients were randomized into two groups: continuation of maintenance therapy (azathioprine or mycophenolate mofetil for 2-3 more years, hydroxychloroquine, and possibly low-dose glucocorticoids (below 10 mg/day), or discontinuation of maintenance therapy (azathioprine or mycophenolate mofetil) over 3 months.
Both study arms were similar with a mean age of 36-37 years, and 82%-86% of patients were female, 59%-67% were White. They had a mean disease duration of 7-9 years, and 72%-80% had experienced their first flare of proliferative nephritis. A total of 54%-65% had received cyclophosphamide induction therapy, and 75%-81% were on mycophenolate mofetil maintenance therapy that had been ongoing for a mean of 2.8 years. Tests of patients’ kidney function revealed a mean serum creatinine of 67-72 micromol/L and estimated glomerular filtration rate (eGFR) of 94-101 mL/min per 1.73 m2. The mean SLE Disease Activity Index score was around 2.
Follow-up visits were conducted every 3 months for 2 years, and the trial had a primary end point of renal relapse rate at 24 months (confirmed by kidney biopsy), while secondary endpoints included rate of severe SLE flares, survival without renal relapse, or severe flare and adverse events, among others.
Patients were excluded if they were not taking hydroxychloroquine, had extrarenal SLE, an eGFR less than 30 mL/min per 1.73 m2 or stage VI disease. After some participants did not finish the trial because of pregnancy, wish for pregnancy, or adverse events, data from a total of 40 in the IST continuation group and 44 in the discontinuation group were analyzed.
Noninferiority of discontinuation versus continuation not shown
A total of 12.5% in the continuation arm experienced renal relapse over 24 months, compared with 27.3% in the discontinuation arm (P = .079).
“The endpoint of noninferiority of discontinuation of immunosuppressive therapy was not shown, but no statistically significant difference was found between groups for renal relapse,” Dr. Jourde-Chiche said.
Severe SLE flares (renal or extrarenal) occurred significantly less often over 24 months in the continuation arm at 12.5% versus 31.8% in the discontinuation arm (P = .034).
However, Dr. Jourde-Chiche pointed out that “the majority of patients who discontinued treatment did not experience a flare.”
No differences were seen between groups with respect to any secondary endpoints. “Fortunately, no patients died or reached end-stage renal disease, and overall, the adverse events did not differ between groups,” she reported.
The study identified several risk factors for renal relapse, among which were low complement component 3 and higher baseline urinary protein to creatinine ratio.
Longer immunosuppressive therapy prediscontinuation leads to better results
Gabriella Moroni, MD, of the nephrology unit at Ospedale Maggiore Policlinico, Milan, authored a review of studies that attempted to interrupt glucocorticoids and other immunosuppressive agents in lupus nephritis and in SLE. Her review concluded that “the available data suggest that therapy withdrawal is feasible at least in patients enjoying a complete clinical remission after a prolonged therapy.”
Asked to comment on the French study, Dr. Moroni said the trial was very welcome. “In our long-term experience, we have stopped IST in 73 patients with lupus nephritis, followed for a mean of 23 years. Of these, 32 never reassumed therapy and 20 had at least one flare.”
Dr. Moroni noted that those participants who did not experience flares had significantly longer IST and longer remission before discontinuation and took hydroxychloroquine more frequently.
“We feel that, to prevent severe flares, firstly, lupus nephritis should be in complete remission for at least 3 years, and secondly, patients should have received IST for at least 5 years before discontinuation; immunosuppressive drugs should be tapered off very slowly and after strict clinical surveillance, and finally, hydroxychloroquine can prevent extrarenal flares.
Dr. Jourde-Chiche reported serving on a speakers bureau for Vifor Pharma and receiving grant/research support from Fresenius Medical Care. Some coauthors reported financial ties to many pharmaceutical companies. Dr. Richez said he has received fees for lectures or boards from GlaxoSmithKline, AstraZeneca, and Novartis. Dr. Moroni had no relevant disclosures.
COPENHAGEN – Discontinuing maintenance immunosuppressive therapy (IST) in patients with proliferative lupus nephritis in remission proved less effective than continuing it in terms of the rates of renal relapse and severe systemic lupus erythematosus (SLE) flare, results from the WIN-Lupus trial show.
Lead researcher Noemie Jourde-Chiche, MD, assistant professor at Aix-Marseille (France) University, presented the results at the annual European Congress of Rheumatology.
The randomized, controlled trial aimed to determine the optimal duration of maintenance IST for proliferative lupus nephritis, by asking whether discontinuation of such therapy after 2-3 years was noninferior to IST continuation for 2 more years.
“This is the first randomized IST trial in proliferative lupus nephritis,” Dr. Jourde-Chiche reported. “We found that noninferiority of IST discontinuation was not demonstrated, and those who discontinued had a higher risk of SLE flares, but the majority of patients who discontinued did not experience flare.”
Regarding the incidence of renal relapse, no statistically significant difference was found between patients who continued versus those who discontinued IST.
Rheumatologist Christophe Richez, MD, of the Groupe Hospitalier Pellegrin-CHU de Bordeaux (France) welcomed the trial. “The work does not find a significant difference but suggests that with more power, the difference would have been significant,” he said in an interview.
He added that a significant number of patients refused to enter the study for several reasons, including scheduling a pregnancy and fear of relapse after stopping the treatment. “This in itself shows that we need this type of study to know if we can stop the treatment to plan a pregnancy or if a relay treatment is necessary. These data also mean we can now better inform our patients about the benefit-risk [profile] of continuing treatment or not.”
Dr. Richez, who was not involved in the trial, noted that the data provide information that strongly suggest more research is needed to better determine which patients are at risk of relapse, and consequently, for which patients is discontinuation possible. “There’s also a need for further analysis of tolerance to the immunosuppressive drugs according to the strategy.”
Finally, he referred to the issue around therapeutic adherence that is often faced with management of lupus. “The data from this study will allow us to better explain to patients the need to continue or discontinue their treatments, including hydroxychloroquine, and also the possibility of decreasing the immunosuppressive drugs to half-dose.”
Maintenance therapy in lupus nephritis: To continue or not?
Conducted in 28 French centers, participants had class III or IV lupus nephritis with active lesions and had previously received induction IST of cyclophosphamide or mycophenolate mofetil with hydroxychloroquine and glucocorticoids. Maintenance IST was azathioprine or mycophenolate mofetil, hydroxychloroquine, and possibly low-dose glucocorticoids (below 10 mg/day).
A total of 96 patients were randomized into two groups: continuation of maintenance therapy (azathioprine or mycophenolate mofetil for 2-3 more years, hydroxychloroquine, and possibly low-dose glucocorticoids (below 10 mg/day), or discontinuation of maintenance therapy (azathioprine or mycophenolate mofetil) over 3 months.
Both study arms were similar with a mean age of 36-37 years, and 82%-86% of patients were female, 59%-67% were White. They had a mean disease duration of 7-9 years, and 72%-80% had experienced their first flare of proliferative nephritis. A total of 54%-65% had received cyclophosphamide induction therapy, and 75%-81% were on mycophenolate mofetil maintenance therapy that had been ongoing for a mean of 2.8 years. Tests of patients’ kidney function revealed a mean serum creatinine of 67-72 micromol/L and estimated glomerular filtration rate (eGFR) of 94-101 mL/min per 1.73 m2. The mean SLE Disease Activity Index score was around 2.
Follow-up visits were conducted every 3 months for 2 years, and the trial had a primary end point of renal relapse rate at 24 months (confirmed by kidney biopsy), while secondary endpoints included rate of severe SLE flares, survival without renal relapse, or severe flare and adverse events, among others.
Patients were excluded if they were not taking hydroxychloroquine, had extrarenal SLE, an eGFR less than 30 mL/min per 1.73 m2 or stage VI disease. After some participants did not finish the trial because of pregnancy, wish for pregnancy, or adverse events, data from a total of 40 in the IST continuation group and 44 in the discontinuation group were analyzed.
Noninferiority of discontinuation versus continuation not shown
A total of 12.5% in the continuation arm experienced renal relapse over 24 months, compared with 27.3% in the discontinuation arm (P = .079).
“The endpoint of noninferiority of discontinuation of immunosuppressive therapy was not shown, but no statistically significant difference was found between groups for renal relapse,” Dr. Jourde-Chiche said.
Severe SLE flares (renal or extrarenal) occurred significantly less often over 24 months in the continuation arm at 12.5% versus 31.8% in the discontinuation arm (P = .034).
However, Dr. Jourde-Chiche pointed out that “the majority of patients who discontinued treatment did not experience a flare.”
No differences were seen between groups with respect to any secondary endpoints. “Fortunately, no patients died or reached end-stage renal disease, and overall, the adverse events did not differ between groups,” she reported.
The study identified several risk factors for renal relapse, among which were low complement component 3 and higher baseline urinary protein to creatinine ratio.
Longer immunosuppressive therapy prediscontinuation leads to better results
Gabriella Moroni, MD, of the nephrology unit at Ospedale Maggiore Policlinico, Milan, authored a review of studies that attempted to interrupt glucocorticoids and other immunosuppressive agents in lupus nephritis and in SLE. Her review concluded that “the available data suggest that therapy withdrawal is feasible at least in patients enjoying a complete clinical remission after a prolonged therapy.”
Asked to comment on the French study, Dr. Moroni said the trial was very welcome. “In our long-term experience, we have stopped IST in 73 patients with lupus nephritis, followed for a mean of 23 years. Of these, 32 never reassumed therapy and 20 had at least one flare.”
Dr. Moroni noted that those participants who did not experience flares had significantly longer IST and longer remission before discontinuation and took hydroxychloroquine more frequently.
“We feel that, to prevent severe flares, firstly, lupus nephritis should be in complete remission for at least 3 years, and secondly, patients should have received IST for at least 5 years before discontinuation; immunosuppressive drugs should be tapered off very slowly and after strict clinical surveillance, and finally, hydroxychloroquine can prevent extrarenal flares.
Dr. Jourde-Chiche reported serving on a speakers bureau for Vifor Pharma and receiving grant/research support from Fresenius Medical Care. Some coauthors reported financial ties to many pharmaceutical companies. Dr. Richez said he has received fees for lectures or boards from GlaxoSmithKline, AstraZeneca, and Novartis. Dr. Moroni had no relevant disclosures.
AT THE EULAR 2022 CONGRESS
Think it’s ILD? Tell it to the machines
SAN FRANCISCO – Interstitial lung disease is a difficult diagnosis to make, but a combination of artificial intelligence (AI) techniques and automated language processing could help clinicians identify the early signs of ILD and start patients on therapy, investigators say.
For example, applying an AI algorithm to spirometry readings taken from patients whose data were registered in the UK Biobank identified 27% as having ILD, and of this group, 66% had ostensibly normal lung function on spirometry but were later diagnosed with ILD, reported Marko Topalovic, PhD, from the AI company ArtiQ in Leuven, Belgium, at the American Thoracic Society’s international conference.
“A diagnosis of ILD is very challenging, so you have patients who are going to be misdiagnosed or have a very late diagnosis, so we aimed to apply our AI algorithm on spirometry to see whether we could detect ILD much earlier,” he said in an interview conducted during a poster discussion session.
AI detected ILD up to 6.8 years before a clinician’s diagnosis, Dr. Topalovic said.
Reading between the lines
In a separate study, investigators at the University of California, Davis, used language analysis software to scour electronic health records for words indicative of early ILD, and found that the technique dramatically shortened the median time to a pulmonary referral, compared with historical controls.
“This is a language processing program that can essentially look through the radiology reports and look for the key words that often describe interstitial lung disease, like traction, honeycomb, fibrotic, etc. With those studies being flagged, an actual pulmonologist will then further review the scan, and see whether it meets criteria for one of the interstitial lung diseases,” lead author William Leon, MD, a resident in the department of internal medicine at the University of California, Davis, said in an interview.
“We then sent the primary care doctor a message to say: ‘Hey, this patient has ILD. You need to send them to a pulmonologist,’ ” he added.
Putting it together
Philip L. Molyneaux, MRCP (UK), MBBS, BS (Hons), from Imperial College London, who comoderated the session but was not involved in the studies, speculated that combining these and other, nontechnical interventions also discussed could help to improve diagnosis of ILD and allow clinicians to prescribe therapy earlier in the disease course.
“What’s going to give you the biggest impact for patients? Everyone working individually is coming up with great advances, and if you put them all together it’s going to provide much greater benefit for our patients,” he said in an interview.
AI Spirometry details
In collaboration with colleagues at the Laboratory of Respiratory Disease at University Hospital in Leuven, Dr. Topalovic applied AI to results of spirometry performed prior to diagnosis of ILD among 109 patients registered in the UK Biobank, a repository of information on more than 500,000 volunteers.
The patients selected had ILD listed as their cause of death, had spirometry performed up to 7 years before their deaths, and did not receive a diagnosis of ILD on the day of the index spirometry.
In all 73% of patients were men, 27% women, with an average age of 64.6 years. A large majority of the sample (77.15%) had a history of smoking, and 60 of the patients (55%) died within one year of an ILD diagnosis.
The investigators plugged the spirometry data and each patients demographic information – including gender, age, height, weight, race, and smoking status – into the AI clinical decision support program, which yielded a statistical probability for each subject of having normal lung function, asthma, COPD, ILD, another obstructive disease, or another unidentifiable respiratory disease.
In 29 patients (27%) the software listed ILD as the highest probability, and of this group 19 patients (66%) had normal lung function according to standard interpretation guidelines.
Spirometry parameters among patients identified as having probable ILD were different from those where ILD was not detected. For example, forced vital capacity (FVC) was 76% of predicted among patients with likely ILD versus 87% of predicted in those who had a diagnosis later (P = .003). Similar differences were seen in the forced expiratory volume in 1 second to FVC ratio, at 0.82 vs. 0.75, respectively (P = .007).
There were no differences in mortality or in median time between spirometry and clinician diagnosis between the groups.
Language processing details
Dr. Leon and colleagues used a language analysis software package to review CT chest reports. Reports were flagged if they contained the words traction, honeycomb, fibrotic, fibrosis, reticular, or reticulation.
The CT scan accompanying each flagged reported was reviewed by a pulmonologist for the presence of ILD, and scans with ILD identified were referred to pulmonary specialists. The results of 2,198 prospective scans followed by prospective screening were compared with those of 1,690 historical controls seen in 2015 and 2016.
The investigators found that 85 incident cases of ILD were identified in the historical controls, compared with 143 in the prospective cohort, leading to 38 and 120 pulmonary referrals, respectively.
For the primary outcome of median time from CT to pulmonary referral, the authors found that it was 1.27 months for the prospective cohort, compared with not reached (censored after 18 months) in historical controls.
The hazard ratio for a pulmonary referral in the prospective versus historical cohort was 2.79, an association that was strengthened after adjusting for sex, age, race, smoking pack-years, cough, crackles, and dyspnea (HR, 4.54; both comparisons significant according to confidence intervals).
The studies were internally funded. Dr. Topalovic is CEO and cofounder of ArtiQ. Dr. Leon and Dr. Molyneaux reported no relevant conflicts of interest.
SAN FRANCISCO – Interstitial lung disease is a difficult diagnosis to make, but a combination of artificial intelligence (AI) techniques and automated language processing could help clinicians identify the early signs of ILD and start patients on therapy, investigators say.
For example, applying an AI algorithm to spirometry readings taken from patients whose data were registered in the UK Biobank identified 27% as having ILD, and of this group, 66% had ostensibly normal lung function on spirometry but were later diagnosed with ILD, reported Marko Topalovic, PhD, from the AI company ArtiQ in Leuven, Belgium, at the American Thoracic Society’s international conference.
“A diagnosis of ILD is very challenging, so you have patients who are going to be misdiagnosed or have a very late diagnosis, so we aimed to apply our AI algorithm on spirometry to see whether we could detect ILD much earlier,” he said in an interview conducted during a poster discussion session.
AI detected ILD up to 6.8 years before a clinician’s diagnosis, Dr. Topalovic said.
Reading between the lines
In a separate study, investigators at the University of California, Davis, used language analysis software to scour electronic health records for words indicative of early ILD, and found that the technique dramatically shortened the median time to a pulmonary referral, compared with historical controls.
“This is a language processing program that can essentially look through the radiology reports and look for the key words that often describe interstitial lung disease, like traction, honeycomb, fibrotic, etc. With those studies being flagged, an actual pulmonologist will then further review the scan, and see whether it meets criteria for one of the interstitial lung diseases,” lead author William Leon, MD, a resident in the department of internal medicine at the University of California, Davis, said in an interview.
“We then sent the primary care doctor a message to say: ‘Hey, this patient has ILD. You need to send them to a pulmonologist,’ ” he added.
Putting it together
Philip L. Molyneaux, MRCP (UK), MBBS, BS (Hons), from Imperial College London, who comoderated the session but was not involved in the studies, speculated that combining these and other, nontechnical interventions also discussed could help to improve diagnosis of ILD and allow clinicians to prescribe therapy earlier in the disease course.
“What’s going to give you the biggest impact for patients? Everyone working individually is coming up with great advances, and if you put them all together it’s going to provide much greater benefit for our patients,” he said in an interview.
AI Spirometry details
In collaboration with colleagues at the Laboratory of Respiratory Disease at University Hospital in Leuven, Dr. Topalovic applied AI to results of spirometry performed prior to diagnosis of ILD among 109 patients registered in the UK Biobank, a repository of information on more than 500,000 volunteers.
The patients selected had ILD listed as their cause of death, had spirometry performed up to 7 years before their deaths, and did not receive a diagnosis of ILD on the day of the index spirometry.
In all 73% of patients were men, 27% women, with an average age of 64.6 years. A large majority of the sample (77.15%) had a history of smoking, and 60 of the patients (55%) died within one year of an ILD diagnosis.
The investigators plugged the spirometry data and each patients demographic information – including gender, age, height, weight, race, and smoking status – into the AI clinical decision support program, which yielded a statistical probability for each subject of having normal lung function, asthma, COPD, ILD, another obstructive disease, or another unidentifiable respiratory disease.
In 29 patients (27%) the software listed ILD as the highest probability, and of this group 19 patients (66%) had normal lung function according to standard interpretation guidelines.
Spirometry parameters among patients identified as having probable ILD were different from those where ILD was not detected. For example, forced vital capacity (FVC) was 76% of predicted among patients with likely ILD versus 87% of predicted in those who had a diagnosis later (P = .003). Similar differences were seen in the forced expiratory volume in 1 second to FVC ratio, at 0.82 vs. 0.75, respectively (P = .007).
There were no differences in mortality or in median time between spirometry and clinician diagnosis between the groups.
Language processing details
Dr. Leon and colleagues used a language analysis software package to review CT chest reports. Reports were flagged if they contained the words traction, honeycomb, fibrotic, fibrosis, reticular, or reticulation.
The CT scan accompanying each flagged reported was reviewed by a pulmonologist for the presence of ILD, and scans with ILD identified were referred to pulmonary specialists. The results of 2,198 prospective scans followed by prospective screening were compared with those of 1,690 historical controls seen in 2015 and 2016.
The investigators found that 85 incident cases of ILD were identified in the historical controls, compared with 143 in the prospective cohort, leading to 38 and 120 pulmonary referrals, respectively.
For the primary outcome of median time from CT to pulmonary referral, the authors found that it was 1.27 months for the prospective cohort, compared with not reached (censored after 18 months) in historical controls.
The hazard ratio for a pulmonary referral in the prospective versus historical cohort was 2.79, an association that was strengthened after adjusting for sex, age, race, smoking pack-years, cough, crackles, and dyspnea (HR, 4.54; both comparisons significant according to confidence intervals).
The studies were internally funded. Dr. Topalovic is CEO and cofounder of ArtiQ. Dr. Leon and Dr. Molyneaux reported no relevant conflicts of interest.
SAN FRANCISCO – Interstitial lung disease is a difficult diagnosis to make, but a combination of artificial intelligence (AI) techniques and automated language processing could help clinicians identify the early signs of ILD and start patients on therapy, investigators say.
For example, applying an AI algorithm to spirometry readings taken from patients whose data were registered in the UK Biobank identified 27% as having ILD, and of this group, 66% had ostensibly normal lung function on spirometry but were later diagnosed with ILD, reported Marko Topalovic, PhD, from the AI company ArtiQ in Leuven, Belgium, at the American Thoracic Society’s international conference.
“A diagnosis of ILD is very challenging, so you have patients who are going to be misdiagnosed or have a very late diagnosis, so we aimed to apply our AI algorithm on spirometry to see whether we could detect ILD much earlier,” he said in an interview conducted during a poster discussion session.
AI detected ILD up to 6.8 years before a clinician’s diagnosis, Dr. Topalovic said.
Reading between the lines
In a separate study, investigators at the University of California, Davis, used language analysis software to scour electronic health records for words indicative of early ILD, and found that the technique dramatically shortened the median time to a pulmonary referral, compared with historical controls.
“This is a language processing program that can essentially look through the radiology reports and look for the key words that often describe interstitial lung disease, like traction, honeycomb, fibrotic, etc. With those studies being flagged, an actual pulmonologist will then further review the scan, and see whether it meets criteria for one of the interstitial lung diseases,” lead author William Leon, MD, a resident in the department of internal medicine at the University of California, Davis, said in an interview.
“We then sent the primary care doctor a message to say: ‘Hey, this patient has ILD. You need to send them to a pulmonologist,’ ” he added.
Putting it together
Philip L. Molyneaux, MRCP (UK), MBBS, BS (Hons), from Imperial College London, who comoderated the session but was not involved in the studies, speculated that combining these and other, nontechnical interventions also discussed could help to improve diagnosis of ILD and allow clinicians to prescribe therapy earlier in the disease course.
“What’s going to give you the biggest impact for patients? Everyone working individually is coming up with great advances, and if you put them all together it’s going to provide much greater benefit for our patients,” he said in an interview.
AI Spirometry details
In collaboration with colleagues at the Laboratory of Respiratory Disease at University Hospital in Leuven, Dr. Topalovic applied AI to results of spirometry performed prior to diagnosis of ILD among 109 patients registered in the UK Biobank, a repository of information on more than 500,000 volunteers.
The patients selected had ILD listed as their cause of death, had spirometry performed up to 7 years before their deaths, and did not receive a diagnosis of ILD on the day of the index spirometry.
In all 73% of patients were men, 27% women, with an average age of 64.6 years. A large majority of the sample (77.15%) had a history of smoking, and 60 of the patients (55%) died within one year of an ILD diagnosis.
The investigators plugged the spirometry data and each patients demographic information – including gender, age, height, weight, race, and smoking status – into the AI clinical decision support program, which yielded a statistical probability for each subject of having normal lung function, asthma, COPD, ILD, another obstructive disease, or another unidentifiable respiratory disease.
In 29 patients (27%) the software listed ILD as the highest probability, and of this group 19 patients (66%) had normal lung function according to standard interpretation guidelines.
Spirometry parameters among patients identified as having probable ILD were different from those where ILD was not detected. For example, forced vital capacity (FVC) was 76% of predicted among patients with likely ILD versus 87% of predicted in those who had a diagnosis later (P = .003). Similar differences were seen in the forced expiratory volume in 1 second to FVC ratio, at 0.82 vs. 0.75, respectively (P = .007).
There were no differences in mortality or in median time between spirometry and clinician diagnosis between the groups.
Language processing details
Dr. Leon and colleagues used a language analysis software package to review CT chest reports. Reports were flagged if they contained the words traction, honeycomb, fibrotic, fibrosis, reticular, or reticulation.
The CT scan accompanying each flagged reported was reviewed by a pulmonologist for the presence of ILD, and scans with ILD identified were referred to pulmonary specialists. The results of 2,198 prospective scans followed by prospective screening were compared with those of 1,690 historical controls seen in 2015 and 2016.
The investigators found that 85 incident cases of ILD were identified in the historical controls, compared with 143 in the prospective cohort, leading to 38 and 120 pulmonary referrals, respectively.
For the primary outcome of median time from CT to pulmonary referral, the authors found that it was 1.27 months for the prospective cohort, compared with not reached (censored after 18 months) in historical controls.
The hazard ratio for a pulmonary referral in the prospective versus historical cohort was 2.79, an association that was strengthened after adjusting for sex, age, race, smoking pack-years, cough, crackles, and dyspnea (HR, 4.54; both comparisons significant according to confidence intervals).
The studies were internally funded. Dr. Topalovic is CEO and cofounder of ArtiQ. Dr. Leon and Dr. Molyneaux reported no relevant conflicts of interest.
AT ATS 2022
Lupus mutation may unlock targeted drugs for patient subset
Scientists have confirmed that a receptor long suspected to be linked to lupus is, in fact, a major driver of the autoimmune disease for at least some subset of patients, according to a study recently published in Nature. Researchers discovered the crucial role of toll-like receptor 7 (TLR7) because of a rare mutation in a pediatric patient with systemic lupus erythematosus (SLE) who had a particularly severe presentation.
“Sometimes it’s valuable to find these very severe cases where there is one mutation that has a strong effect because if we understand how those mutations work, the lessons we learn can generally tell us about disease mechanisms,” explained senior author Carola G. Vinuesa, MD, PhD, of the Centre for Personalised Immunology at Australian National University in Canberra and The Francis Crick Institute in London.

“It’s quite difficult to find one mutation that can alone cause the entire disease,” Dr. Vinuesa added, but what it reveals about how the disease develops may lead to more effective targeted therapies than the immune suppressants most often used to treat lupus currently.
The mutation they found was in the TLR7 gene that encodes the TLR7 protein. TLR7 is a receptor used by immune cells to identify viral RNA so they can fight off viral infections, including COVID-19. But if the body’s own genetic material binds to TLR7 in susceptible individuals, it can lead to an overproduction of type 1 interferons, which are cytokines that trigger or exacerbate the immune reactions that lead to lupus symptoms. The TLR7 gene occurs on the X chromosome, which may explain men’s greater susceptibility to COVID-19 and the greater incidence of lupus in women, who have two X chromosomes instead of the one that men have, Dr. Vinuesa said.
Previous research had shown an association between TLR7 and lupus, but this new study is the first to provide definitive proof that a TLR7 mutation by itself can directly cause human lupus. After discovering the variant in the patient, Dr. Vinuesa’s team used CRISPR to edit the genome of a mouse model and introduce the same mutation the patient had. “And they developed full-blown disease, just with this one single base-pair substitution – 1 letter in the 3 billion letters of the genome,” Dr. Vinuesa said. “It tells us that these receptors are not just there to recognize viral RNA, that in some circumstances, they could be triggered by our own nucleic acids.”
One pathway among many?
The finding does not mean that every lupus patient has this mutation, which remains rare, but suggests that overactivity in this receptor already reported in many lupus patients may be causally related to disease, Dr. Vinuesa said.
Noa Schwartz, MD, an assistant professor of medicine at Albert Einstein College of Medicine, New York, and director of the Montefiore-Einstein Institute for Lupus Care and Research, said in an interview that lupus is thought of as a syndrome, a collection of different but similar diseases that don’t necessarily have a single cause. But finding a single gene mutation that could potentially lead to lupus is an important piece of the puzzle, said Dr. Schwartz, who was not involved in the study. Based on past research in mice models, “we’ve hypothesized that TLR7 is important in humans as well, but this is the last nail in the coffin.”
One of the key questions this finding has prompted is how many patients’ disease results from TLR7 activity. “Because of the evidence from Ignacio Sanz’s group demonstrating TLR7 overactivity in a significant fraction of SLE patients, we believe that it is probably going to be pretty important,” Dr. Vinuesa said. “My feeling is that it is going to be quite a central pathway in lupus pathogenesis, if not the central pathway.”
Dr. Schwartz was more cautious, noting that it is probably important for a subset of patients but may “have a limited effect on the general lupus population.” While it’s not yet clear how large that subset is, it is possible it will include people with cutaneous lupus, those with primarily dermatologic symptoms.
“Hydroxychloroquine works particularly well for cutaneous manifestations of lupus, and one of the ways that works is by inhibiting TLR7 and TLR9, so this [finding] potentially matters for skin disease and lupus, but it’s very early,” Dr. Schwartz said. If it does turn out that TLR7 activity is particularly associated with cutaneous lupus, it may mean therapies with fewer side effects, she said. “Specifically for cutaneous lupus, the concept of suppressing the entire immune system for skin illness sometimes feels, especially to patients, very extreme, so they are [patients] who directed therapy could be so especially relevant for.”
Laura Lewandowski, MD, an assistant clinical investigator and head of the lupus genomics and global health disparities unit at the National Institute of Arthritis and Musculoskeletal and Skin Disease, described this study as particularly remarkable in the way it revealed the mechanism leading to lupus symptoms.
“As whole genome sequencing becomes faster and less expensive, more and more people are employing them in their studies,” most of which report changes in certain genes, Dr. Lewandowski said. “One of the most striking findings about this paper was that they took it to the next step and did a really elegant study on the exact way this gain-of-function TLR7 mutation leads to the autoimmunity that we see in lupus. The detail of mechanism in this paper is really unique.”
A step toward personalized medicine
Dr. Lewandowski is part of a team that recently presented a poster related to genomic sequencing in lupus patients at the annual meeting of the Childhood Arthritis and Rheumatology Research Alliance. Her study reported on the whole genome sequencing of patients with childhood-onset SLE who were already enrolled in the CARRA Lupus Registry. Children with lupus may be more likely than adults to have rare genetic variants, so a registry of childhood-onset SLE patients with fully sequenced genomes provides an opportunity to look for single-gene mutations specifically linked to lupus, said Dr. Lewandowski, who has recently begun a research collaboration with Dr. Vinuesa.
“As we move forward and more and more patients are included in these studies, we will understand a little bit more about the genetic architecture of patients who have rare variations leading to disease, or even common variations,” Dr. Lewandowski said about the intersection between her research and Dr. Vinuesa’s study. The more data they gather, the more they can explore the possible interactions of rare and common variants that play a role in SLE as well as what environmental triggers, such as viral infection or pollution exposure, might tip someone into having an autoimmune disease. “We’re just starting to peek under the hood,” Dr. Lewandowski said.
If further research can reveal the relative contribution of genetics to the disease and what those genetic drivers are, it may allow for greater precision in therapies and “ultimately improve the quality of life for our patients, the ultimate goal of all of these studies,” Dr. Lewandowski said.
Drugs that target TLR7 already exist for other indications, and clinical trials have already begun to see if these TLR7 inhibitors benefit lupus patients.
“If the clinical trials work, this will be quite a nice, targeted therapy with potentially much less side effects than other therapies on the market at the moment,” Dr. Vinuesa said. She is cautiously hopeful, saying it’s likely to make an impact on lupus treatment, but it’s too early to say precisely how much.
“It allows us to understand the disease mechanisms a little bit better and to try and assess what percentage of patients’ disease can be explained by overactivity in this receptor,” Dr. Vinuesa said. She thinks it’s possible that TLR7 over activation may be relevant to other systemic autoimmune diseases as well, such as Sjögren’s syndrome, rheumatoid arthritis, or juvenile dermatomyositis, but it will take more studies to find out.
“Right now, we have medicines that broadly inhibit the immune system and aren’t as targeted, but we have a lot more clinical and scientific work to do before we move this field forward for lupus patients,” Dr. Lewandowski said. “This is one case where they were able to find the exact molecular defect, and it’s not the end of the path of precision medicine — it’s the beginning.”
Dr. Vinuesa, Dr. Schwartz, and Dr. Lewandowski reported no disclosures.
A version of this article first appeared on Medscape.com.
Scientists have confirmed that a receptor long suspected to be linked to lupus is, in fact, a major driver of the autoimmune disease for at least some subset of patients, according to a study recently published in Nature. Researchers discovered the crucial role of toll-like receptor 7 (TLR7) because of a rare mutation in a pediatric patient with systemic lupus erythematosus (SLE) who had a particularly severe presentation.
“Sometimes it’s valuable to find these very severe cases where there is one mutation that has a strong effect because if we understand how those mutations work, the lessons we learn can generally tell us about disease mechanisms,” explained senior author Carola G. Vinuesa, MD, PhD, of the Centre for Personalised Immunology at Australian National University in Canberra and The Francis Crick Institute in London.

“It’s quite difficult to find one mutation that can alone cause the entire disease,” Dr. Vinuesa added, but what it reveals about how the disease develops may lead to more effective targeted therapies than the immune suppressants most often used to treat lupus currently.
The mutation they found was in the TLR7 gene that encodes the TLR7 protein. TLR7 is a receptor used by immune cells to identify viral RNA so they can fight off viral infections, including COVID-19. But if the body’s own genetic material binds to TLR7 in susceptible individuals, it can lead to an overproduction of type 1 interferons, which are cytokines that trigger or exacerbate the immune reactions that lead to lupus symptoms. The TLR7 gene occurs on the X chromosome, which may explain men’s greater susceptibility to COVID-19 and the greater incidence of lupus in women, who have two X chromosomes instead of the one that men have, Dr. Vinuesa said.
Previous research had shown an association between TLR7 and lupus, but this new study is the first to provide definitive proof that a TLR7 mutation by itself can directly cause human lupus. After discovering the variant in the patient, Dr. Vinuesa’s team used CRISPR to edit the genome of a mouse model and introduce the same mutation the patient had. “And they developed full-blown disease, just with this one single base-pair substitution – 1 letter in the 3 billion letters of the genome,” Dr. Vinuesa said. “It tells us that these receptors are not just there to recognize viral RNA, that in some circumstances, they could be triggered by our own nucleic acids.”
One pathway among many?
The finding does not mean that every lupus patient has this mutation, which remains rare, but suggests that overactivity in this receptor already reported in many lupus patients may be causally related to disease, Dr. Vinuesa said.
Noa Schwartz, MD, an assistant professor of medicine at Albert Einstein College of Medicine, New York, and director of the Montefiore-Einstein Institute for Lupus Care and Research, said in an interview that lupus is thought of as a syndrome, a collection of different but similar diseases that don’t necessarily have a single cause. But finding a single gene mutation that could potentially lead to lupus is an important piece of the puzzle, said Dr. Schwartz, who was not involved in the study. Based on past research in mice models, “we’ve hypothesized that TLR7 is important in humans as well, but this is the last nail in the coffin.”
One of the key questions this finding has prompted is how many patients’ disease results from TLR7 activity. “Because of the evidence from Ignacio Sanz’s group demonstrating TLR7 overactivity in a significant fraction of SLE patients, we believe that it is probably going to be pretty important,” Dr. Vinuesa said. “My feeling is that it is going to be quite a central pathway in lupus pathogenesis, if not the central pathway.”
Dr. Schwartz was more cautious, noting that it is probably important for a subset of patients but may “have a limited effect on the general lupus population.” While it’s not yet clear how large that subset is, it is possible it will include people with cutaneous lupus, those with primarily dermatologic symptoms.
“Hydroxychloroquine works particularly well for cutaneous manifestations of lupus, and one of the ways that works is by inhibiting TLR7 and TLR9, so this [finding] potentially matters for skin disease and lupus, but it’s very early,” Dr. Schwartz said. If it does turn out that TLR7 activity is particularly associated with cutaneous lupus, it may mean therapies with fewer side effects, she said. “Specifically for cutaneous lupus, the concept of suppressing the entire immune system for skin illness sometimes feels, especially to patients, very extreme, so they are [patients] who directed therapy could be so especially relevant for.”
Laura Lewandowski, MD, an assistant clinical investigator and head of the lupus genomics and global health disparities unit at the National Institute of Arthritis and Musculoskeletal and Skin Disease, described this study as particularly remarkable in the way it revealed the mechanism leading to lupus symptoms.
“As whole genome sequencing becomes faster and less expensive, more and more people are employing them in their studies,” most of which report changes in certain genes, Dr. Lewandowski said. “One of the most striking findings about this paper was that they took it to the next step and did a really elegant study on the exact way this gain-of-function TLR7 mutation leads to the autoimmunity that we see in lupus. The detail of mechanism in this paper is really unique.”
A step toward personalized medicine
Dr. Lewandowski is part of a team that recently presented a poster related to genomic sequencing in lupus patients at the annual meeting of the Childhood Arthritis and Rheumatology Research Alliance. Her study reported on the whole genome sequencing of patients with childhood-onset SLE who were already enrolled in the CARRA Lupus Registry. Children with lupus may be more likely than adults to have rare genetic variants, so a registry of childhood-onset SLE patients with fully sequenced genomes provides an opportunity to look for single-gene mutations specifically linked to lupus, said Dr. Lewandowski, who has recently begun a research collaboration with Dr. Vinuesa.
“As we move forward and more and more patients are included in these studies, we will understand a little bit more about the genetic architecture of patients who have rare variations leading to disease, or even common variations,” Dr. Lewandowski said about the intersection between her research and Dr. Vinuesa’s study. The more data they gather, the more they can explore the possible interactions of rare and common variants that play a role in SLE as well as what environmental triggers, such as viral infection or pollution exposure, might tip someone into having an autoimmune disease. “We’re just starting to peek under the hood,” Dr. Lewandowski said.
If further research can reveal the relative contribution of genetics to the disease and what those genetic drivers are, it may allow for greater precision in therapies and “ultimately improve the quality of life for our patients, the ultimate goal of all of these studies,” Dr. Lewandowski said.
Drugs that target TLR7 already exist for other indications, and clinical trials have already begun to see if these TLR7 inhibitors benefit lupus patients.
“If the clinical trials work, this will be quite a nice, targeted therapy with potentially much less side effects than other therapies on the market at the moment,” Dr. Vinuesa said. She is cautiously hopeful, saying it’s likely to make an impact on lupus treatment, but it’s too early to say precisely how much.
“It allows us to understand the disease mechanisms a little bit better and to try and assess what percentage of patients’ disease can be explained by overactivity in this receptor,” Dr. Vinuesa said. She thinks it’s possible that TLR7 over activation may be relevant to other systemic autoimmune diseases as well, such as Sjögren’s syndrome, rheumatoid arthritis, or juvenile dermatomyositis, but it will take more studies to find out.
“Right now, we have medicines that broadly inhibit the immune system and aren’t as targeted, but we have a lot more clinical and scientific work to do before we move this field forward for lupus patients,” Dr. Lewandowski said. “This is one case where they were able to find the exact molecular defect, and it’s not the end of the path of precision medicine — it’s the beginning.”
Dr. Vinuesa, Dr. Schwartz, and Dr. Lewandowski reported no disclosures.
A version of this article first appeared on Medscape.com.
Scientists have confirmed that a receptor long suspected to be linked to lupus is, in fact, a major driver of the autoimmune disease for at least some subset of patients, according to a study recently published in Nature. Researchers discovered the crucial role of toll-like receptor 7 (TLR7) because of a rare mutation in a pediatric patient with systemic lupus erythematosus (SLE) who had a particularly severe presentation.
“Sometimes it’s valuable to find these very severe cases where there is one mutation that has a strong effect because if we understand how those mutations work, the lessons we learn can generally tell us about disease mechanisms,” explained senior author Carola G. Vinuesa, MD, PhD, of the Centre for Personalised Immunology at Australian National University in Canberra and The Francis Crick Institute in London.

“It’s quite difficult to find one mutation that can alone cause the entire disease,” Dr. Vinuesa added, but what it reveals about how the disease develops may lead to more effective targeted therapies than the immune suppressants most often used to treat lupus currently.
The mutation they found was in the TLR7 gene that encodes the TLR7 protein. TLR7 is a receptor used by immune cells to identify viral RNA so they can fight off viral infections, including COVID-19. But if the body’s own genetic material binds to TLR7 in susceptible individuals, it can lead to an overproduction of type 1 interferons, which are cytokines that trigger or exacerbate the immune reactions that lead to lupus symptoms. The TLR7 gene occurs on the X chromosome, which may explain men’s greater susceptibility to COVID-19 and the greater incidence of lupus in women, who have two X chromosomes instead of the one that men have, Dr. Vinuesa said.
Previous research had shown an association between TLR7 and lupus, but this new study is the first to provide definitive proof that a TLR7 mutation by itself can directly cause human lupus. After discovering the variant in the patient, Dr. Vinuesa’s team used CRISPR to edit the genome of a mouse model and introduce the same mutation the patient had. “And they developed full-blown disease, just with this one single base-pair substitution – 1 letter in the 3 billion letters of the genome,” Dr. Vinuesa said. “It tells us that these receptors are not just there to recognize viral RNA, that in some circumstances, they could be triggered by our own nucleic acids.”
One pathway among many?
The finding does not mean that every lupus patient has this mutation, which remains rare, but suggests that overactivity in this receptor already reported in many lupus patients may be causally related to disease, Dr. Vinuesa said.
Noa Schwartz, MD, an assistant professor of medicine at Albert Einstein College of Medicine, New York, and director of the Montefiore-Einstein Institute for Lupus Care and Research, said in an interview that lupus is thought of as a syndrome, a collection of different but similar diseases that don’t necessarily have a single cause. But finding a single gene mutation that could potentially lead to lupus is an important piece of the puzzle, said Dr. Schwartz, who was not involved in the study. Based on past research in mice models, “we’ve hypothesized that TLR7 is important in humans as well, but this is the last nail in the coffin.”
One of the key questions this finding has prompted is how many patients’ disease results from TLR7 activity. “Because of the evidence from Ignacio Sanz’s group demonstrating TLR7 overactivity in a significant fraction of SLE patients, we believe that it is probably going to be pretty important,” Dr. Vinuesa said. “My feeling is that it is going to be quite a central pathway in lupus pathogenesis, if not the central pathway.”
Dr. Schwartz was more cautious, noting that it is probably important for a subset of patients but may “have a limited effect on the general lupus population.” While it’s not yet clear how large that subset is, it is possible it will include people with cutaneous lupus, those with primarily dermatologic symptoms.
“Hydroxychloroquine works particularly well for cutaneous manifestations of lupus, and one of the ways that works is by inhibiting TLR7 and TLR9, so this [finding] potentially matters for skin disease and lupus, but it’s very early,” Dr. Schwartz said. If it does turn out that TLR7 activity is particularly associated with cutaneous lupus, it may mean therapies with fewer side effects, she said. “Specifically for cutaneous lupus, the concept of suppressing the entire immune system for skin illness sometimes feels, especially to patients, very extreme, so they are [patients] who directed therapy could be so especially relevant for.”
Laura Lewandowski, MD, an assistant clinical investigator and head of the lupus genomics and global health disparities unit at the National Institute of Arthritis and Musculoskeletal and Skin Disease, described this study as particularly remarkable in the way it revealed the mechanism leading to lupus symptoms.
“As whole genome sequencing becomes faster and less expensive, more and more people are employing them in their studies,” most of which report changes in certain genes, Dr. Lewandowski said. “One of the most striking findings about this paper was that they took it to the next step and did a really elegant study on the exact way this gain-of-function TLR7 mutation leads to the autoimmunity that we see in lupus. The detail of mechanism in this paper is really unique.”
A step toward personalized medicine
Dr. Lewandowski is part of a team that recently presented a poster related to genomic sequencing in lupus patients at the annual meeting of the Childhood Arthritis and Rheumatology Research Alliance. Her study reported on the whole genome sequencing of patients with childhood-onset SLE who were already enrolled in the CARRA Lupus Registry. Children with lupus may be more likely than adults to have rare genetic variants, so a registry of childhood-onset SLE patients with fully sequenced genomes provides an opportunity to look for single-gene mutations specifically linked to lupus, said Dr. Lewandowski, who has recently begun a research collaboration with Dr. Vinuesa.
“As we move forward and more and more patients are included in these studies, we will understand a little bit more about the genetic architecture of patients who have rare variations leading to disease, or even common variations,” Dr. Lewandowski said about the intersection between her research and Dr. Vinuesa’s study. The more data they gather, the more they can explore the possible interactions of rare and common variants that play a role in SLE as well as what environmental triggers, such as viral infection or pollution exposure, might tip someone into having an autoimmune disease. “We’re just starting to peek under the hood,” Dr. Lewandowski said.
If further research can reveal the relative contribution of genetics to the disease and what those genetic drivers are, it may allow for greater precision in therapies and “ultimately improve the quality of life for our patients, the ultimate goal of all of these studies,” Dr. Lewandowski said.
Drugs that target TLR7 already exist for other indications, and clinical trials have already begun to see if these TLR7 inhibitors benefit lupus patients.
“If the clinical trials work, this will be quite a nice, targeted therapy with potentially much less side effects than other therapies on the market at the moment,” Dr. Vinuesa said. She is cautiously hopeful, saying it’s likely to make an impact on lupus treatment, but it’s too early to say precisely how much.
“It allows us to understand the disease mechanisms a little bit better and to try and assess what percentage of patients’ disease can be explained by overactivity in this receptor,” Dr. Vinuesa said. She thinks it’s possible that TLR7 over activation may be relevant to other systemic autoimmune diseases as well, such as Sjögren’s syndrome, rheumatoid arthritis, or juvenile dermatomyositis, but it will take more studies to find out.
“Right now, we have medicines that broadly inhibit the immune system and aren’t as targeted, but we have a lot more clinical and scientific work to do before we move this field forward for lupus patients,” Dr. Lewandowski said. “This is one case where they were able to find the exact molecular defect, and it’s not the end of the path of precision medicine — it’s the beginning.”
Dr. Vinuesa, Dr. Schwartz, and Dr. Lewandowski reported no disclosures.
A version of this article first appeared on Medscape.com.
FROM NATURE
High maternal, fetal morbidity rates in SLE pregnancies
COPENHAGEN – Pregnant women with systemic lupus erythematosus (SLE) are at significantly higher risk of requiring transfusion, developing a cerebrovascular disorder, or developing acute renal failure than pregnant women without SLE, a review of data from an American national sample indicates.
Pregnant women with SLE also have a twofold-higher risk for premature delivery, and a threefold risk of having a fetus with intrauterine growth restriction than their pregnant counterparts without SLE, reported Bella Mehta, MBBS, MS, MD, a rheumatologist at the Hospital for Special Surgery in New York.
“Severe maternal morbidity and fetal morbidity still remain high, but this work can help inform physicians and counsel patients for pregnancy planning and management,” she said at the annual European Congress of Rheumatology.
Although in-hospital maternal and fetal mortality rates for women with SLE have declined over the past 2 decades, the same cannot be said for morbidities, prompting the investigators to conduct a study to determine the proportion of fetal and maternal morbidity in SLE deliveries, compared with non-SLE deliveries over a decade.
Inpatient Sample
Dr. Mehta and colleagues studied retrospective data on 40 million delivery-related admissions from the National Inpatient Sample database. Of these patients, 51,161 had a diagnosis of SLE.
They identified all delivery-related hospital admissions for patients with and without SLE from 2008 through 2017 using diagnostic codes.
The researchers looked at fetal morbidity indicators, including preterm delivery and intrauterine growth restriction, and used the Centers for Disease Control and Prevention standard definition of severe maternal morbidity as “unexpected outcomes of labor and delivery that result in significant short- or long- term consequences to a woman’s health.”
They identified 21 severe maternal morbidity outcomes, including blood transfusion requirements, acute renal failure, eclampsia and disseminated intravascular coagulation, cardiovascular and peripheral vascular disorders, and general medical issues (hysterectomy, shock, sepsis, adult respiratory distress syndrome, severe anesthesia complications, temporary tracheostomy, and ventilation).
Study results
Women with SLE were slightly older at the time of delivery (mean age, 30.05 vs. 29.19 years) and had more comorbidities, according to the Elixhauser Comorbidity Scale, with 97.84% of women in this group having one to four comorbidities, compared with 19.4% of women without SLE.
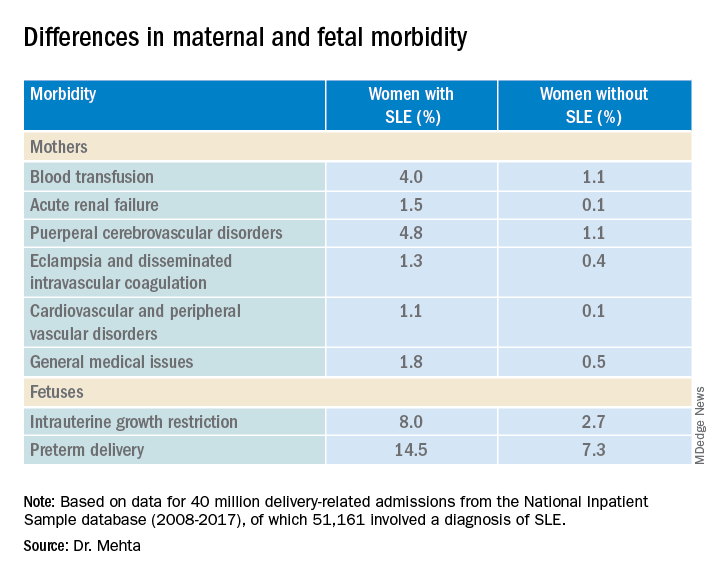
Dr. Mehta acknowledged that the study was limited by the inability to capture outpatient deliveries, although she noted that only about 1.3% of deliveries in the United States occur outside the inpatient setting.
In addition, she noted that the database does not include information on lupus disease activity, Apgar scores, SLE flares, the presence of nephritis, antiphospholipid or anti-Ro/SSA antibodies, or medication use.
A rheumatologist who was not involved in the study said in an interview that the data from this study are in line with those in other recently published studies.
“The problem is that these data were not corrected for further disease activity or drugs,” said Frauke Förger, MD, professor of rheumatology and immunology at the University of Bern (Switzerland), who comoderated the oral abstract session where the data were presented.
She said prospective studies that adjusted for factors such as SLE disease activity and medication use will be required to give clinicians a better understanding of how to manage pregnancies in women with SLE.
The study was supported by an award from Weill Cornell Medicine. Dr. Mehta and Dr. Förger reported no relevant financial disclosures.
A version of this article first appeared on Medscape.com.
COPENHAGEN – Pregnant women with systemic lupus erythematosus (SLE) are at significantly higher risk of requiring transfusion, developing a cerebrovascular disorder, or developing acute renal failure than pregnant women without SLE, a review of data from an American national sample indicates.
Pregnant women with SLE also have a twofold-higher risk for premature delivery, and a threefold risk of having a fetus with intrauterine growth restriction than their pregnant counterparts without SLE, reported Bella Mehta, MBBS, MS, MD, a rheumatologist at the Hospital for Special Surgery in New York.
“Severe maternal morbidity and fetal morbidity still remain high, but this work can help inform physicians and counsel patients for pregnancy planning and management,” she said at the annual European Congress of Rheumatology.
Although in-hospital maternal and fetal mortality rates for women with SLE have declined over the past 2 decades, the same cannot be said for morbidities, prompting the investigators to conduct a study to determine the proportion of fetal and maternal morbidity in SLE deliveries, compared with non-SLE deliveries over a decade.
Inpatient Sample
Dr. Mehta and colleagues studied retrospective data on 40 million delivery-related admissions from the National Inpatient Sample database. Of these patients, 51,161 had a diagnosis of SLE.
They identified all delivery-related hospital admissions for patients with and without SLE from 2008 through 2017 using diagnostic codes.
The researchers looked at fetal morbidity indicators, including preterm delivery and intrauterine growth restriction, and used the Centers for Disease Control and Prevention standard definition of severe maternal morbidity as “unexpected outcomes of labor and delivery that result in significant short- or long- term consequences to a woman’s health.”
They identified 21 severe maternal morbidity outcomes, including blood transfusion requirements, acute renal failure, eclampsia and disseminated intravascular coagulation, cardiovascular and peripheral vascular disorders, and general medical issues (hysterectomy, shock, sepsis, adult respiratory distress syndrome, severe anesthesia complications, temporary tracheostomy, and ventilation).
Study results
Women with SLE were slightly older at the time of delivery (mean age, 30.05 vs. 29.19 years) and had more comorbidities, according to the Elixhauser Comorbidity Scale, with 97.84% of women in this group having one to four comorbidities, compared with 19.4% of women without SLE.

Dr. Mehta acknowledged that the study was limited by the inability to capture outpatient deliveries, although she noted that only about 1.3% of deliveries in the United States occur outside the inpatient setting.
In addition, she noted that the database does not include information on lupus disease activity, Apgar scores, SLE flares, the presence of nephritis, antiphospholipid or anti-Ro/SSA antibodies, or medication use.
A rheumatologist who was not involved in the study said in an interview that the data from this study are in line with those in other recently published studies.
“The problem is that these data were not corrected for further disease activity or drugs,” said Frauke Förger, MD, professor of rheumatology and immunology at the University of Bern (Switzerland), who comoderated the oral abstract session where the data were presented.
She said prospective studies that adjusted for factors such as SLE disease activity and medication use will be required to give clinicians a better understanding of how to manage pregnancies in women with SLE.
The study was supported by an award from Weill Cornell Medicine. Dr. Mehta and Dr. Förger reported no relevant financial disclosures.
A version of this article first appeared on Medscape.com.
COPENHAGEN – Pregnant women with systemic lupus erythematosus (SLE) are at significantly higher risk of requiring transfusion, developing a cerebrovascular disorder, or developing acute renal failure than pregnant women without SLE, a review of data from an American national sample indicates.
Pregnant women with SLE also have a twofold-higher risk for premature delivery, and a threefold risk of having a fetus with intrauterine growth restriction than their pregnant counterparts without SLE, reported Bella Mehta, MBBS, MS, MD, a rheumatologist at the Hospital for Special Surgery in New York.
“Severe maternal morbidity and fetal morbidity still remain high, but this work can help inform physicians and counsel patients for pregnancy planning and management,” she said at the annual European Congress of Rheumatology.
Although in-hospital maternal and fetal mortality rates for women with SLE have declined over the past 2 decades, the same cannot be said for morbidities, prompting the investigators to conduct a study to determine the proportion of fetal and maternal morbidity in SLE deliveries, compared with non-SLE deliveries over a decade.
Inpatient Sample
Dr. Mehta and colleagues studied retrospective data on 40 million delivery-related admissions from the National Inpatient Sample database. Of these patients, 51,161 had a diagnosis of SLE.
They identified all delivery-related hospital admissions for patients with and without SLE from 2008 through 2017 using diagnostic codes.
The researchers looked at fetal morbidity indicators, including preterm delivery and intrauterine growth restriction, and used the Centers for Disease Control and Prevention standard definition of severe maternal morbidity as “unexpected outcomes of labor and delivery that result in significant short- or long- term consequences to a woman’s health.”
They identified 21 severe maternal morbidity outcomes, including blood transfusion requirements, acute renal failure, eclampsia and disseminated intravascular coagulation, cardiovascular and peripheral vascular disorders, and general medical issues (hysterectomy, shock, sepsis, adult respiratory distress syndrome, severe anesthesia complications, temporary tracheostomy, and ventilation).
Study results
Women with SLE were slightly older at the time of delivery (mean age, 30.05 vs. 29.19 years) and had more comorbidities, according to the Elixhauser Comorbidity Scale, with 97.84% of women in this group having one to four comorbidities, compared with 19.4% of women without SLE.

Dr. Mehta acknowledged that the study was limited by the inability to capture outpatient deliveries, although she noted that only about 1.3% of deliveries in the United States occur outside the inpatient setting.
In addition, she noted that the database does not include information on lupus disease activity, Apgar scores, SLE flares, the presence of nephritis, antiphospholipid or anti-Ro/SSA antibodies, or medication use.
A rheumatologist who was not involved in the study said in an interview that the data from this study are in line with those in other recently published studies.
“The problem is that these data were not corrected for further disease activity or drugs,” said Frauke Förger, MD, professor of rheumatology and immunology at the University of Bern (Switzerland), who comoderated the oral abstract session where the data were presented.
She said prospective studies that adjusted for factors such as SLE disease activity and medication use will be required to give clinicians a better understanding of how to manage pregnancies in women with SLE.
The study was supported by an award from Weill Cornell Medicine. Dr. Mehta and Dr. Förger reported no relevant financial disclosures.
A version of this article first appeared on Medscape.com.
AT THE EULAR 2022 CONGRESS
Updated EULAR recommendations for AAV include new drugs, practices
The
The 2022 revision – which was unveiled at the annual European Congress of Rheumatology – includes guidance on using new drugs, such as avacopan (Tavneos) and mepolizumab (Nucala), as well as revised recommendations on the use of rituximab and glucocorticosteroids.

The overhaul also contains specific recommendations for treating eosinophilic granulomatosis with polyangiitis (EGPA), separating it out from granulomatosis with polyangiitis (GPA) and microscopic polyangiitis (MPA) for the first time.
“Until now, EGPA has usually been managed in the same way as [the] other diseases,” Bernhard Hellmich, MD, of the University of Tübingen (Germany) said in an interview ahead of his presentation at the congress.
“But we now have data on each type specifically, so there is good reason to make separate recommendations,” he added.
Indeed, so much new data has become available in the past few years there are only three recommendations that remain unchanged from the previous iteration published in 2016.
Since then, “several high-impact studies in AAV have been published and the results of these studies required an update of the existing recommendations,” Dr. Hellmich said.
Developed in record time – just 7 months from start to finish – the process of updating the recommendations on AAV followed EULAR’s standard operating procedures. An important step in this process is to perform a systemic literature review. Perhaps crucially, and in contrast to the first U.S. vasculitis guidelines published only in 2021, the most recent literature search was able to include data on avacopan, which was approved for use in Europe in January as an adjunctive treatment for adults with severe active GPA and MPA.
The results of the literature review were reported separately at the EULAR 2022 Congress, with separate presentations highlighting the data behind the amended treatment and diagnostic and follow-up procedure recommendations.
Highlights of the changes
A key change is the introduction of four overarching principles, which weren’t included in the previous update, said Dr. Hellmich.
“We moved some of the existing recommendations with low level of evidence to overarching principles,” he added, stating that the first general principle was that all patients should be offered “the best care which must be based on shared decision-making between the patient and the physician considering efficacy, safety, and costs.”
The second principle states that patients should have access to education that covers the prognosis and impact of AAV, including recognizing warning symptoms and treatment options.
The third focuses on screening for adverse effects and comorbidities, recommending that patients are given appropriate prophylaxis and lifestyle advice.
Finally, the fourth general principle recognizes that AAV is a rare group of heterogenous and potentially life-threatening diseases that need multidisciplinary care, with access to specific vasculitis expertise.
New recommendations
Of the 17 recommendations made, 6 are completely new, including one on ANCA testing in patients who are suspected of having AAV.
“We recommend testing for both PR3- and MPO-ANCA using a high-quality antigen-specific assay as the primary method of testing,” Dr. Hellmich said. This is based on strong new evidence that antigen-specific assays have superior diagnostic accuracy, compared with indirect immunofluorescence.
“We also want to emphasize that ANCA testing should be done in patients with signs and symptoms in order to minimize the risk of false-positive results,” Dr. Hellmich said.
Also new is the recommendation to use oral steroids to induce remission in GPA/MPA, followed by a stepwise reduction in the dose, aiming for a dose of not more than 5 mg prednisolone per day by 4-5 months of treatment.
“Glucocorticoids are very effective, but also are the major trigger of infections in AAV,” said Dr. Hellmich. This is important since infections are a major driver of early mortality in AAV.
“Another possibility to reduce glucocorticoid exposure is avacopan,” he said. It’s recommended to be used in combination with rituximab or cyclophosphamide for remission induction in GPA/MPA as a strategy to basically “get rid of steroids.”
Indeed, “for patients who really have a high burden of glucocorticoid-associated adverse effects, especially relapsing patients, I think it would make sense just to give avacopan and no steroids,” Dr. Hellmich said.
Other new recommendations concern remission induction and maintenance therapy in new-onset EGPA. Regarding the latter, the choice of treatment depends on whether there is an organ- or life-threatening situation, with methotrexate, azathioprine, mepolizumab, or rituximab all recommended equally, or if there is no organ- or life-threatening situation, then mepolizumab is preferred.
Revised and unchanged recommendations
Eight of the recommendations have been revised, with rituximab being placed more prominently as a treatment in some. For remission induction in GPA and MPA with organ- or life threatening disease, rituximab is now the preferred option for relapsing disease. Rituximab also replaced methotrexate as the preferred option for maintaining remission, although methotrexate and azathioprine can still be considered as alternatives.
Another changed statement is on the duration of maintenance treatment in GPA and MPA, which now advocates 1-2 years of treatment after achieving remission. Longer therapy might be needed in relapsing cases, but the benefits and risks need to be carefully considered and patient preferences taken into account.
Prophylaxis against pneumonia and other infections is still recommended, with the revised guidance noting that patients receiving cyclophosphamide, rituximab, or high-dose steroids, should be treated with trimethoprim-sulfamethoxazole (co-trimoxazole).
“There are retrospective data in the AAV population that the administration of co-trimoxazole reduces not only the incidence of pneumocystis, but also of other infections. So, this is important recommendation for clinical practice,” Dr. Hellmich said.
Summing up
“For a rare disease group, I think this is very good progress,” said Dr. Hellmich, but “there are still many open questions, so we have a long research agenda.”
There is purposefully no recommendation on COVID-19, however, as “the conditions that impact COVID outcomes change rapidly and any recommendation made now is likely to be outdated soon; the AAV recommendations are intended to last for at least a couple of years.”
In a press release issued by the German Society for Rheumatology, which was unrelated to Dr. Hellmich’s talk, experts commented on vasculitis guidelines generally, noting that there has been a move toward using biologic therapies such as rituximab and mepolizumab as a new standard of therapy.
DGRh President and chief physician at the Immanuel Hospital in Berlin Andreas Krause, MD, observed that “cyclophosphamide, which was used in the past and which inhibits blood formation in the bone marrow and can lead to infertility, can now often be dispensed with.”
Julia Holle, MD, of Rheumazentrum Schleswig-Holstein Mitte in Neumünster, Germany, was also quoted in the press release, saying that, “for patients, the successful use of biologics and the reduction in the glucocorticoid dose is important progress.”
Dr. Holle was involved in the development of revised European guidelines. She is also the lead author of a recent publication on treatment of vasculitis on available evidence. Dr. Hellmich acknowledged having ties to multiple pharma companies, acting as speaker, consultant, or both to Abbvie, Amgen, AstraZeneca, Boehringer Ingelheim, Bristol-Myers Squibb, Chugai, GlaxoSmithKline, InflaRx, Merck Sharp & Dohme, Novartis, Pfizer, Roche, and Vifor.
The
The 2022 revision – which was unveiled at the annual European Congress of Rheumatology – includes guidance on using new drugs, such as avacopan (Tavneos) and mepolizumab (Nucala), as well as revised recommendations on the use of rituximab and glucocorticosteroids.

The overhaul also contains specific recommendations for treating eosinophilic granulomatosis with polyangiitis (EGPA), separating it out from granulomatosis with polyangiitis (GPA) and microscopic polyangiitis (MPA) for the first time.
“Until now, EGPA has usually been managed in the same way as [the] other diseases,” Bernhard Hellmich, MD, of the University of Tübingen (Germany) said in an interview ahead of his presentation at the congress.
“But we now have data on each type specifically, so there is good reason to make separate recommendations,” he added.
Indeed, so much new data has become available in the past few years there are only three recommendations that remain unchanged from the previous iteration published in 2016.
Since then, “several high-impact studies in AAV have been published and the results of these studies required an update of the existing recommendations,” Dr. Hellmich said.
Developed in record time – just 7 months from start to finish – the process of updating the recommendations on AAV followed EULAR’s standard operating procedures. An important step in this process is to perform a systemic literature review. Perhaps crucially, and in contrast to the first U.S. vasculitis guidelines published only in 2021, the most recent literature search was able to include data on avacopan, which was approved for use in Europe in January as an adjunctive treatment for adults with severe active GPA and MPA.
The results of the literature review were reported separately at the EULAR 2022 Congress, with separate presentations highlighting the data behind the amended treatment and diagnostic and follow-up procedure recommendations.
Highlights of the changes
A key change is the introduction of four overarching principles, which weren’t included in the previous update, said Dr. Hellmich.
“We moved some of the existing recommendations with low level of evidence to overarching principles,” he added, stating that the first general principle was that all patients should be offered “the best care which must be based on shared decision-making between the patient and the physician considering efficacy, safety, and costs.”
The second principle states that patients should have access to education that covers the prognosis and impact of AAV, including recognizing warning symptoms and treatment options.
The third focuses on screening for adverse effects and comorbidities, recommending that patients are given appropriate prophylaxis and lifestyle advice.
Finally, the fourth general principle recognizes that AAV is a rare group of heterogenous and potentially life-threatening diseases that need multidisciplinary care, with access to specific vasculitis expertise.
New recommendations
Of the 17 recommendations made, 6 are completely new, including one on ANCA testing in patients who are suspected of having AAV.
“We recommend testing for both PR3- and MPO-ANCA using a high-quality antigen-specific assay as the primary method of testing,” Dr. Hellmich said. This is based on strong new evidence that antigen-specific assays have superior diagnostic accuracy, compared with indirect immunofluorescence.
“We also want to emphasize that ANCA testing should be done in patients with signs and symptoms in order to minimize the risk of false-positive results,” Dr. Hellmich said.
Also new is the recommendation to use oral steroids to induce remission in GPA/MPA, followed by a stepwise reduction in the dose, aiming for a dose of not more than 5 mg prednisolone per day by 4-5 months of treatment.
“Glucocorticoids are very effective, but also are the major trigger of infections in AAV,” said Dr. Hellmich. This is important since infections are a major driver of early mortality in AAV.
“Another possibility to reduce glucocorticoid exposure is avacopan,” he said. It’s recommended to be used in combination with rituximab or cyclophosphamide for remission induction in GPA/MPA as a strategy to basically “get rid of steroids.”
Indeed, “for patients who really have a high burden of glucocorticoid-associated adverse effects, especially relapsing patients, I think it would make sense just to give avacopan and no steroids,” Dr. Hellmich said.
Other new recommendations concern remission induction and maintenance therapy in new-onset EGPA. Regarding the latter, the choice of treatment depends on whether there is an organ- or life-threatening situation, with methotrexate, azathioprine, mepolizumab, or rituximab all recommended equally, or if there is no organ- or life-threatening situation, then mepolizumab is preferred.
Revised and unchanged recommendations
Eight of the recommendations have been revised, with rituximab being placed more prominently as a treatment in some. For remission induction in GPA and MPA with organ- or life threatening disease, rituximab is now the preferred option for relapsing disease. Rituximab also replaced methotrexate as the preferred option for maintaining remission, although methotrexate and azathioprine can still be considered as alternatives.
Another changed statement is on the duration of maintenance treatment in GPA and MPA, which now advocates 1-2 years of treatment after achieving remission. Longer therapy might be needed in relapsing cases, but the benefits and risks need to be carefully considered and patient preferences taken into account.
Prophylaxis against pneumonia and other infections is still recommended, with the revised guidance noting that patients receiving cyclophosphamide, rituximab, or high-dose steroids, should be treated with trimethoprim-sulfamethoxazole (co-trimoxazole).
“There are retrospective data in the AAV population that the administration of co-trimoxazole reduces not only the incidence of pneumocystis, but also of other infections. So, this is important recommendation for clinical practice,” Dr. Hellmich said.
Summing up
“For a rare disease group, I think this is very good progress,” said Dr. Hellmich, but “there are still many open questions, so we have a long research agenda.”
There is purposefully no recommendation on COVID-19, however, as “the conditions that impact COVID outcomes change rapidly and any recommendation made now is likely to be outdated soon; the AAV recommendations are intended to last for at least a couple of years.”
In a press release issued by the German Society for Rheumatology, which was unrelated to Dr. Hellmich’s talk, experts commented on vasculitis guidelines generally, noting that there has been a move toward using biologic therapies such as rituximab and mepolizumab as a new standard of therapy.
DGRh President and chief physician at the Immanuel Hospital in Berlin Andreas Krause, MD, observed that “cyclophosphamide, which was used in the past and which inhibits blood formation in the bone marrow and can lead to infertility, can now often be dispensed with.”
Julia Holle, MD, of Rheumazentrum Schleswig-Holstein Mitte in Neumünster, Germany, was also quoted in the press release, saying that, “for patients, the successful use of biologics and the reduction in the glucocorticoid dose is important progress.”
Dr. Holle was involved in the development of revised European guidelines. She is also the lead author of a recent publication on treatment of vasculitis on available evidence. Dr. Hellmich acknowledged having ties to multiple pharma companies, acting as speaker, consultant, or both to Abbvie, Amgen, AstraZeneca, Boehringer Ingelheim, Bristol-Myers Squibb, Chugai, GlaxoSmithKline, InflaRx, Merck Sharp & Dohme, Novartis, Pfizer, Roche, and Vifor.
The
The 2022 revision – which was unveiled at the annual European Congress of Rheumatology – includes guidance on using new drugs, such as avacopan (Tavneos) and mepolizumab (Nucala), as well as revised recommendations on the use of rituximab and glucocorticosteroids.

The overhaul also contains specific recommendations for treating eosinophilic granulomatosis with polyangiitis (EGPA), separating it out from granulomatosis with polyangiitis (GPA) and microscopic polyangiitis (MPA) for the first time.
“Until now, EGPA has usually been managed in the same way as [the] other diseases,” Bernhard Hellmich, MD, of the University of Tübingen (Germany) said in an interview ahead of his presentation at the congress.
“But we now have data on each type specifically, so there is good reason to make separate recommendations,” he added.
Indeed, so much new data has become available in the past few years there are only three recommendations that remain unchanged from the previous iteration published in 2016.
Since then, “several high-impact studies in AAV have been published and the results of these studies required an update of the existing recommendations,” Dr. Hellmich said.
Developed in record time – just 7 months from start to finish – the process of updating the recommendations on AAV followed EULAR’s standard operating procedures. An important step in this process is to perform a systemic literature review. Perhaps crucially, and in contrast to the first U.S. vasculitis guidelines published only in 2021, the most recent literature search was able to include data on avacopan, which was approved for use in Europe in January as an adjunctive treatment for adults with severe active GPA and MPA.
The results of the literature review were reported separately at the EULAR 2022 Congress, with separate presentations highlighting the data behind the amended treatment and diagnostic and follow-up procedure recommendations.
Highlights of the changes
A key change is the introduction of four overarching principles, which weren’t included in the previous update, said Dr. Hellmich.
“We moved some of the existing recommendations with low level of evidence to overarching principles,” he added, stating that the first general principle was that all patients should be offered “the best care which must be based on shared decision-making between the patient and the physician considering efficacy, safety, and costs.”
The second principle states that patients should have access to education that covers the prognosis and impact of AAV, including recognizing warning symptoms and treatment options.
The third focuses on screening for adverse effects and comorbidities, recommending that patients are given appropriate prophylaxis and lifestyle advice.
Finally, the fourth general principle recognizes that AAV is a rare group of heterogenous and potentially life-threatening diseases that need multidisciplinary care, with access to specific vasculitis expertise.
New recommendations
Of the 17 recommendations made, 6 are completely new, including one on ANCA testing in patients who are suspected of having AAV.
“We recommend testing for both PR3- and MPO-ANCA using a high-quality antigen-specific assay as the primary method of testing,” Dr. Hellmich said. This is based on strong new evidence that antigen-specific assays have superior diagnostic accuracy, compared with indirect immunofluorescence.
“We also want to emphasize that ANCA testing should be done in patients with signs and symptoms in order to minimize the risk of false-positive results,” Dr. Hellmich said.
Also new is the recommendation to use oral steroids to induce remission in GPA/MPA, followed by a stepwise reduction in the dose, aiming for a dose of not more than 5 mg prednisolone per day by 4-5 months of treatment.
“Glucocorticoids are very effective, but also are the major trigger of infections in AAV,” said Dr. Hellmich. This is important since infections are a major driver of early mortality in AAV.
“Another possibility to reduce glucocorticoid exposure is avacopan,” he said. It’s recommended to be used in combination with rituximab or cyclophosphamide for remission induction in GPA/MPA as a strategy to basically “get rid of steroids.”
Indeed, “for patients who really have a high burden of glucocorticoid-associated adverse effects, especially relapsing patients, I think it would make sense just to give avacopan and no steroids,” Dr. Hellmich said.
Other new recommendations concern remission induction and maintenance therapy in new-onset EGPA. Regarding the latter, the choice of treatment depends on whether there is an organ- or life-threatening situation, with methotrexate, azathioprine, mepolizumab, or rituximab all recommended equally, or if there is no organ- or life-threatening situation, then mepolizumab is preferred.
Revised and unchanged recommendations
Eight of the recommendations have been revised, with rituximab being placed more prominently as a treatment in some. For remission induction in GPA and MPA with organ- or life threatening disease, rituximab is now the preferred option for relapsing disease. Rituximab also replaced methotrexate as the preferred option for maintaining remission, although methotrexate and azathioprine can still be considered as alternatives.
Another changed statement is on the duration of maintenance treatment in GPA and MPA, which now advocates 1-2 years of treatment after achieving remission. Longer therapy might be needed in relapsing cases, but the benefits and risks need to be carefully considered and patient preferences taken into account.
Prophylaxis against pneumonia and other infections is still recommended, with the revised guidance noting that patients receiving cyclophosphamide, rituximab, or high-dose steroids, should be treated with trimethoprim-sulfamethoxazole (co-trimoxazole).
“There are retrospective data in the AAV population that the administration of co-trimoxazole reduces not only the incidence of pneumocystis, but also of other infections. So, this is important recommendation for clinical practice,” Dr. Hellmich said.
Summing up
“For a rare disease group, I think this is very good progress,” said Dr. Hellmich, but “there are still many open questions, so we have a long research agenda.”
There is purposefully no recommendation on COVID-19, however, as “the conditions that impact COVID outcomes change rapidly and any recommendation made now is likely to be outdated soon; the AAV recommendations are intended to last for at least a couple of years.”
In a press release issued by the German Society for Rheumatology, which was unrelated to Dr. Hellmich’s talk, experts commented on vasculitis guidelines generally, noting that there has been a move toward using biologic therapies such as rituximab and mepolizumab as a new standard of therapy.
DGRh President and chief physician at the Immanuel Hospital in Berlin Andreas Krause, MD, observed that “cyclophosphamide, which was used in the past and which inhibits blood formation in the bone marrow and can lead to infertility, can now often be dispensed with.”
Julia Holle, MD, of Rheumazentrum Schleswig-Holstein Mitte in Neumünster, Germany, was also quoted in the press release, saying that, “for patients, the successful use of biologics and the reduction in the glucocorticoid dose is important progress.”
Dr. Holle was involved in the development of revised European guidelines. She is also the lead author of a recent publication on treatment of vasculitis on available evidence. Dr. Hellmich acknowledged having ties to multiple pharma companies, acting as speaker, consultant, or both to Abbvie, Amgen, AstraZeneca, Boehringer Ingelheim, Bristol-Myers Squibb, Chugai, GlaxoSmithKline, InflaRx, Merck Sharp & Dohme, Novartis, Pfizer, Roche, and Vifor.
FROM THE EULAR 2022 CONGRESS
Abortion debate may affect Rx decisions for pregnant women
Obstetrician Beverly Gray, MD, is already seeing the effects of the Roe v. Wade abortion debate in her North Carolina practice.
The state allows abortion but requires that women get counseling with a qualified health professional 72 hours before the procedure. “Aside from that, we still have patients asking for more efficacious contraceptive methods just in case,” said Dr. Gray, residency director and division director for women’s community and population health and associate professor for obstetrics and gynecology at Duke University, Durham, N.C.
Patients and staff in her clinic have also been approaching her about tubal ligation. “They’re asking about additional birth control methods because they’re concerned about what’s going to happen” with the challenge to the historic Roe v. Wade decision in the Supreme Court and subsequent actions in the states to restrict or ban abortion, she said.
This has implications not just for abortion but for medications known to affect pregnancy. “What I’m really worried about is physicians will be withholding medicine because they’re concerned about teratogenic effects,” said Dr. Gray.
With more states issuing restrictions on abortion, doctors are worried that patients needing certain drugs to maintain their lupus flares, cancer, or other diseases may decide not to take them in the event they accidentally become pregnant. If the drug is known to affect the fetus, the fear is a patient who lives in a state with abortion restrictions will no longer have the option to terminate a pregnancy.
Instead, a scenario may arise in which the patient – and their physician – may opt not to treat at all with an otherwise lifesaving medication, experts told this news organization.
The U.S. landscape on abortion restrictions
A leaked draft of a U.S. Supreme Court opinion on Mississippi’s 15-week abortion ban has sent the medical community into a tailspin. The case, Dobbs v. Jackson Women’s Health Organization, challenges the 1973 Roe v. Wade decision that affirms the constitutional right to abortion. It’s anticipated the high court will decide on the case in June.
Although the upcoming decision is subject to change, the draft indicated the high court would uphold the Mississippi ban. This would essentially overturn the 1973 ruling. An earlier Supreme Court decision allowing a Texas law banning abortion at 6 weeks suggests the court may already be heading in this direction. At the state level, legislatures have been moving on divergent paths – some taking steps to preserve abortion rights, others initiating restrictions.
More than 100 abortion restrictions in 19 states took effect in 2021, according to the Guttmacher Institute, which tracks such metrics. In 2022, “two key themes are anti-abortion policymakers’ continued pursuit of various types of abortion bans and restrictions on medication abortion,” the institute reported.
Forty-six states and the District of Columbia have introduced 2,025 restrictions or proactive measures on sexual and reproductive health and rights so far this year. The latest tally from Guttmacher, updated in late May, revealed that 11 states so far have enacted 42 abortion restrictions. A total of 6 states (Arizona, Florida, Idaho, Kentucky, Oklahoma, and Wyoming) have issued nine bans on abortion.
Comparatively, 11 states have enacted 19 protective abortion measures.
Twenty-two states have introduced 117 restrictions on medication abortions, which account for 54% of U.S. abortions. This includes seven measures that would ban medication abortion outright, according to Guttmacher. Kentucky and South Dakota collectively have enacted 14 restrictions on medication abortion, as well as provisions that ban mailing of abortion pills.
Chilling effect on prescribing
Some physicians anticipate that drugs such as the “morning-after” pill (levonorgestrel) will become less available as restrictions go into effect, since these are medications designed to prevent pregnancy.*
However, the ongoing effort to put a lid on abortion measures has prompted concerns about a trickle-down effect on other medications that are otherwise life-changing or lifesaving to patients but pose a risk to the fetus.
Several drugs are well documented to affect fetal growth and development of the fetus, ranging from mild, transitory effects to severe, permanent birth defects, said Ronald G. Grifka, MD, chief medical officer of University of Michigan Health-West and clinical professor of pediatrics at the University of Michigan Medical School, Ann Arbor. “As new medications are developed, we will need heightened attention to make sure they are safe for the fetus,” he added.
Certain teratogenic medications are associated with a high risk of abortion even though this isn’t their primary use, noted Christina Chambers, PhD, MPH, co-director of the Center for Better Beginnings and associate director with the Altman Clinical & Translational Research Institute at the University of California, San Diego.
“I don’t think anyone would intentionally take these drugs to induce spontaneous abortion. But if the drugs pose a risk for it, I can see how the laws might be stretched” to include them, said Dr. Chambers.
Methotrexate, a medication for autoimmune disorders, has a high risk of spontaneous abortion. So do acne medications such as isotretinoin.
Patients are usually told they’re not supposed to get pregnant on these drugs because there’s a high risk of pregnancy loss and risk of malformations and potential learning problems in the fetus. But many pregnancies aren’t planned, said Dr. Chambers. “Patients may forget about the side effects or think their birth control will protect them. And the next time they refill the medication, they may not hear about the warnings again.”
With a restrictive abortion law or ban in effect, a woman might think: “I won’t take this drug because if there’s any potential that I might get pregnant, I won’t have the option to abort an at-risk pregnancy.” Women and their doctors, for that matter, don’t want to put themselves in this position, said Dr. Chambers.
Rheumatologist Megan Clowse, MD, who prescribes several medications that potentially cause major birth defects and pregnancy loss, worries about the ramifications of these accumulating bans.
“Methotrexate has been a leading drug for us for decades for rheumatoid arthritis. Mycophenolate is a vital drug for lupus,” said Dr. Clowse, associate professor of medicine at Duke University’s division of rheumatology and immunology.
Both methotrexate and mycophenolate pose about a 40% risk of pregnancy loss and significantly increase the risk for birth defects. “I’m definitely concerned that there might be doctors or women who elect not to use those medications in women of reproductive age because of the potential risk for pregnancy and absence of abortion rights,” said Dr. Clowse.
These situations might force women to use contraceptives they don’t want to use, such as hormonal implants or intrauterine devices, she added. Another side effect is that women and their partners may decide to abstain from sex.
The iPLEDGE factor
Some rheumatology drugs like lenalidomide (Revlimid) require a valid negative pregnancy test in a lab every month. Similarly, the iPLEDGE Risk Evaluation and Mitigation Strategy seeks to reduce the teratogenicity of isotretinoin by requiring two types of birth control and regular pregnancy tests by users.
For isotretinoin specifically, abortion restrictions “could lead to increased adherence to pregnancy prevention measures which are already stringent in iPLEDGE. But on the other hand, it could lead to reduced willingness of physicians to prescribe or patients to take the medication,” said Dr. Chambers.
With programs like iPLEDGE in effect, the rate of pregnancies and abortions that occur in dermatology are relatively low, said Jenny Murase, MD, associate clinical professor of dermatology at the University of California, San Francisco.
Nevertheless, as a physician who regularly prescribes medications like isotretinoin in women of childbearing age, “it’s terrifying to me that a woman wouldn’t have the option to terminate the pregnancy if a teratogenic effect from the medication caused a severe birth defect,” said Dr. Murase.
Dermatologists use other teratogenic medications such as thalidomide, mycophenolate mofetil, and methotrexate for chronic dermatologic disease like psoriasis and atopic dermatitis.
The situation is especially tricky for dermatologists since most patients – about 80% – never discuss their pregnancy with their specialist prior to pregnancy initiation. Dr. Murase recalls when a patient with chronic plaque psoriasis on methotrexate in her late 40s became pregnant and had an abortion even before Dr. Murase became aware of the pregnancy.
Because dermatologists routinely prescribe long-term medications for chronic diseases like acne, psoriasis, and atopic dermatitis, it is important to have a conversation regarding the risks and benefits of long-term medication should a pregnancy occur in any woman of childbearing age, she said.
Fewer women in clinical trials?
Abortion restrictions could possibly discourage women of reproductive age to participate in a clinical trial for a new medication, said Dr. Chambers.
A female patient with a chronic disease who’s randomized to receive a new medication may be required to use certain types of birth control because of unknown potential adverse effects the drug may have on the fetus. But in some cases, accidental pregnancies happen.
The participant in the trial may say, “I don’t know enough about the safety of this drug in pregnancy, and I’ve already taken it. I want to terminate the pregnancy,” said Dr. Chambers. Thinking ahead, a woman may decide not to do the trial to avoid the risk of getting pregnant and not having the option to terminate the pregnancy.
This could apply to new drugs such as antiviral treatments, or medications for severe chronic disease that typically have no clinical trial data in pregnancy prior to initial release into the market.
Women may start taking the drug without thinking about getting pregnant, then realize there are no safety data and become concerned about its effects on a future pregnancy.
The question is: Will abortion restrictions have a chilling effect on these new drugs as well? Patients and their doctors may decide not to try it until more data are available. “I can see where abortion restrictions would change the risk or benefit calculation in thinking about what you do or don’t prescribe or take during reproductive age,” said Dr. Chambers.
The upside of restrictions?
If there’s a positive side to these developments with abortion bans, it may encourage women taking new medications or joining clinical trials to think even more carefully about adherence to effective contraception, said Dr. Chambers.
Some methods are more effective than others, she emphasized. “When you have an unplanned pregnancy, it could mean that the method you used wasn’t optimal or you weren’t using it as recommended.” A goal moving forward is to encourage more thoughtful use of highly effective contraceptives, thus reducing the number of unplanned pregnancies, she added.
If patients are taking methotrexate, “the time to think about pregnancy is before getting pregnant so you can switch to a drug that’s compatible with pregnancy,” she said.
This whole thought process regarding pregnancy planning could work toward useful health goals, said Dr. Chambers. “Nobody thinks termination is the preferred method, but planning ahead should involve a discussion of what works best for the patient.”
Patients do have other choices, said Dr. Grifka. “Fortunately, there are many commonly prescribed medications which cross the placenta and have no ill effects on the fetus.”
Talking to patients about choices
Dr. Clowse, who spends a lot of time training rheumatologists, encourages them to have conversations with patients about pregnancy planning. It’s a lot to manage, getting the right drug to a female patient with chronic illness, especially in this current climate of abortion upheaval, she noted.
Her approach is to have an open and honest conversation with patients about their concerns and fears, what the realities are, and what the potential future options are for certain rheumatology drugs in the United States.
Some women who see what’s happening across the country may become so risk averse that they may choose to die rather than take a lifesaving drug that poses certain risks under new restrictions.
“I think that’s tragic,” said Dr. Clowse.
To help their patients, Dr. Gray believes physicians across specialties should better educate themselves about physiology in pregnancy and how to counsel patients on the impact of not taking medications in pregnancy.
In her view, it’s almost coercive to say to a patient, “You really need to have effective contraception if I’m going to give you this lifesaving or quality-of-life-improving medication.”
When confronting such scenarios, Dr. Gray doesn’t think physicians need to change how they counsel patients about contraception. “I don’t think we should be putting pressure on patients to consider other permanent methods just because there’s a lack of abortion options.”
Patients will eventually make those decisions for themselves, she said. “They’re going to want a more efficacious method because they’re worried about not having access to abortion if they get pregnant.”
Dr. Gray reports being a site principal investigator for a phase 3 trial for VeraCept IUD, funded by Sebela Pharmaceuticals. Dr. Clowse reports receiving research funding and doing consulting for GlaxoSmithKline.
*Correction, 6/2/2022: A previous version of this article misstated the intended use of drugs such as the “morning-after” pill (levonorgestrel). They are taken to prevent unintended pregnancy.
A version of this article first appeared on Medscape.com .
Obstetrician Beverly Gray, MD, is already seeing the effects of the Roe v. Wade abortion debate in her North Carolina practice.
The state allows abortion but requires that women get counseling with a qualified health professional 72 hours before the procedure. “Aside from that, we still have patients asking for more efficacious contraceptive methods just in case,” said Dr. Gray, residency director and division director for women’s community and population health and associate professor for obstetrics and gynecology at Duke University, Durham, N.C.
Patients and staff in her clinic have also been approaching her about tubal ligation. “They’re asking about additional birth control methods because they’re concerned about what’s going to happen” with the challenge to the historic Roe v. Wade decision in the Supreme Court and subsequent actions in the states to restrict or ban abortion, she said.
This has implications not just for abortion but for medications known to affect pregnancy. “What I’m really worried about is physicians will be withholding medicine because they’re concerned about teratogenic effects,” said Dr. Gray.
With more states issuing restrictions on abortion, doctors are worried that patients needing certain drugs to maintain their lupus flares, cancer, or other diseases may decide not to take them in the event they accidentally become pregnant. If the drug is known to affect the fetus, the fear is a patient who lives in a state with abortion restrictions will no longer have the option to terminate a pregnancy.
Instead, a scenario may arise in which the patient – and their physician – may opt not to treat at all with an otherwise lifesaving medication, experts told this news organization.
The U.S. landscape on abortion restrictions
A leaked draft of a U.S. Supreme Court opinion on Mississippi’s 15-week abortion ban has sent the medical community into a tailspin. The case, Dobbs v. Jackson Women’s Health Organization, challenges the 1973 Roe v. Wade decision that affirms the constitutional right to abortion. It’s anticipated the high court will decide on the case in June.
Although the upcoming decision is subject to change, the draft indicated the high court would uphold the Mississippi ban. This would essentially overturn the 1973 ruling. An earlier Supreme Court decision allowing a Texas law banning abortion at 6 weeks suggests the court may already be heading in this direction. At the state level, legislatures have been moving on divergent paths – some taking steps to preserve abortion rights, others initiating restrictions.
More than 100 abortion restrictions in 19 states took effect in 2021, according to the Guttmacher Institute, which tracks such metrics. In 2022, “two key themes are anti-abortion policymakers’ continued pursuit of various types of abortion bans and restrictions on medication abortion,” the institute reported.
Forty-six states and the District of Columbia have introduced 2,025 restrictions or proactive measures on sexual and reproductive health and rights so far this year. The latest tally from Guttmacher, updated in late May, revealed that 11 states so far have enacted 42 abortion restrictions. A total of 6 states (Arizona, Florida, Idaho, Kentucky, Oklahoma, and Wyoming) have issued nine bans on abortion.
Comparatively, 11 states have enacted 19 protective abortion measures.
Twenty-two states have introduced 117 restrictions on medication abortions, which account for 54% of U.S. abortions. This includes seven measures that would ban medication abortion outright, according to Guttmacher. Kentucky and South Dakota collectively have enacted 14 restrictions on medication abortion, as well as provisions that ban mailing of abortion pills.
Chilling effect on prescribing
Some physicians anticipate that drugs such as the “morning-after” pill (levonorgestrel) will become less available as restrictions go into effect, since these are medications designed to prevent pregnancy.*
However, the ongoing effort to put a lid on abortion measures has prompted concerns about a trickle-down effect on other medications that are otherwise life-changing or lifesaving to patients but pose a risk to the fetus.
Several drugs are well documented to affect fetal growth and development of the fetus, ranging from mild, transitory effects to severe, permanent birth defects, said Ronald G. Grifka, MD, chief medical officer of University of Michigan Health-West and clinical professor of pediatrics at the University of Michigan Medical School, Ann Arbor. “As new medications are developed, we will need heightened attention to make sure they are safe for the fetus,” he added.
Certain teratogenic medications are associated with a high risk of abortion even though this isn’t their primary use, noted Christina Chambers, PhD, MPH, co-director of the Center for Better Beginnings and associate director with the Altman Clinical & Translational Research Institute at the University of California, San Diego.
“I don’t think anyone would intentionally take these drugs to induce spontaneous abortion. But if the drugs pose a risk for it, I can see how the laws might be stretched” to include them, said Dr. Chambers.
Methotrexate, a medication for autoimmune disorders, has a high risk of spontaneous abortion. So do acne medications such as isotretinoin.
Patients are usually told they’re not supposed to get pregnant on these drugs because there’s a high risk of pregnancy loss and risk of malformations and potential learning problems in the fetus. But many pregnancies aren’t planned, said Dr. Chambers. “Patients may forget about the side effects or think their birth control will protect them. And the next time they refill the medication, they may not hear about the warnings again.”
With a restrictive abortion law or ban in effect, a woman might think: “I won’t take this drug because if there’s any potential that I might get pregnant, I won’t have the option to abort an at-risk pregnancy.” Women and their doctors, for that matter, don’t want to put themselves in this position, said Dr. Chambers.
Rheumatologist Megan Clowse, MD, who prescribes several medications that potentially cause major birth defects and pregnancy loss, worries about the ramifications of these accumulating bans.
“Methotrexate has been a leading drug for us for decades for rheumatoid arthritis. Mycophenolate is a vital drug for lupus,” said Dr. Clowse, associate professor of medicine at Duke University’s division of rheumatology and immunology.
Both methotrexate and mycophenolate pose about a 40% risk of pregnancy loss and significantly increase the risk for birth defects. “I’m definitely concerned that there might be doctors or women who elect not to use those medications in women of reproductive age because of the potential risk for pregnancy and absence of abortion rights,” said Dr. Clowse.
These situations might force women to use contraceptives they don’t want to use, such as hormonal implants or intrauterine devices, she added. Another side effect is that women and their partners may decide to abstain from sex.
The iPLEDGE factor
Some rheumatology drugs like lenalidomide (Revlimid) require a valid negative pregnancy test in a lab every month. Similarly, the iPLEDGE Risk Evaluation and Mitigation Strategy seeks to reduce the teratogenicity of isotretinoin by requiring two types of birth control and regular pregnancy tests by users.
For isotretinoin specifically, abortion restrictions “could lead to increased adherence to pregnancy prevention measures which are already stringent in iPLEDGE. But on the other hand, it could lead to reduced willingness of physicians to prescribe or patients to take the medication,” said Dr. Chambers.
With programs like iPLEDGE in effect, the rate of pregnancies and abortions that occur in dermatology are relatively low, said Jenny Murase, MD, associate clinical professor of dermatology at the University of California, San Francisco.
Nevertheless, as a physician who regularly prescribes medications like isotretinoin in women of childbearing age, “it’s terrifying to me that a woman wouldn’t have the option to terminate the pregnancy if a teratogenic effect from the medication caused a severe birth defect,” said Dr. Murase.
Dermatologists use other teratogenic medications such as thalidomide, mycophenolate mofetil, and methotrexate for chronic dermatologic disease like psoriasis and atopic dermatitis.
The situation is especially tricky for dermatologists since most patients – about 80% – never discuss their pregnancy with their specialist prior to pregnancy initiation. Dr. Murase recalls when a patient with chronic plaque psoriasis on methotrexate in her late 40s became pregnant and had an abortion even before Dr. Murase became aware of the pregnancy.
Because dermatologists routinely prescribe long-term medications for chronic diseases like acne, psoriasis, and atopic dermatitis, it is important to have a conversation regarding the risks and benefits of long-term medication should a pregnancy occur in any woman of childbearing age, she said.
Fewer women in clinical trials?
Abortion restrictions could possibly discourage women of reproductive age to participate in a clinical trial for a new medication, said Dr. Chambers.
A female patient with a chronic disease who’s randomized to receive a new medication may be required to use certain types of birth control because of unknown potential adverse effects the drug may have on the fetus. But in some cases, accidental pregnancies happen.
The participant in the trial may say, “I don’t know enough about the safety of this drug in pregnancy, and I’ve already taken it. I want to terminate the pregnancy,” said Dr. Chambers. Thinking ahead, a woman may decide not to do the trial to avoid the risk of getting pregnant and not having the option to terminate the pregnancy.
This could apply to new drugs such as antiviral treatments, or medications for severe chronic disease that typically have no clinical trial data in pregnancy prior to initial release into the market.
Women may start taking the drug without thinking about getting pregnant, then realize there are no safety data and become concerned about its effects on a future pregnancy.
The question is: Will abortion restrictions have a chilling effect on these new drugs as well? Patients and their doctors may decide not to try it until more data are available. “I can see where abortion restrictions would change the risk or benefit calculation in thinking about what you do or don’t prescribe or take during reproductive age,” said Dr. Chambers.
The upside of restrictions?
If there’s a positive side to these developments with abortion bans, it may encourage women taking new medications or joining clinical trials to think even more carefully about adherence to effective contraception, said Dr. Chambers.
Some methods are more effective than others, she emphasized. “When you have an unplanned pregnancy, it could mean that the method you used wasn’t optimal or you weren’t using it as recommended.” A goal moving forward is to encourage more thoughtful use of highly effective contraceptives, thus reducing the number of unplanned pregnancies, she added.
If patients are taking methotrexate, “the time to think about pregnancy is before getting pregnant so you can switch to a drug that’s compatible with pregnancy,” she said.
This whole thought process regarding pregnancy planning could work toward useful health goals, said Dr. Chambers. “Nobody thinks termination is the preferred method, but planning ahead should involve a discussion of what works best for the patient.”
Patients do have other choices, said Dr. Grifka. “Fortunately, there are many commonly prescribed medications which cross the placenta and have no ill effects on the fetus.”
Talking to patients about choices
Dr. Clowse, who spends a lot of time training rheumatologists, encourages them to have conversations with patients about pregnancy planning. It’s a lot to manage, getting the right drug to a female patient with chronic illness, especially in this current climate of abortion upheaval, she noted.
Her approach is to have an open and honest conversation with patients about their concerns and fears, what the realities are, and what the potential future options are for certain rheumatology drugs in the United States.
Some women who see what’s happening across the country may become so risk averse that they may choose to die rather than take a lifesaving drug that poses certain risks under new restrictions.
“I think that’s tragic,” said Dr. Clowse.
To help their patients, Dr. Gray believes physicians across specialties should better educate themselves about physiology in pregnancy and how to counsel patients on the impact of not taking medications in pregnancy.
In her view, it’s almost coercive to say to a patient, “You really need to have effective contraception if I’m going to give you this lifesaving or quality-of-life-improving medication.”
When confronting such scenarios, Dr. Gray doesn’t think physicians need to change how they counsel patients about contraception. “I don’t think we should be putting pressure on patients to consider other permanent methods just because there’s a lack of abortion options.”
Patients will eventually make those decisions for themselves, she said. “They’re going to want a more efficacious method because they’re worried about not having access to abortion if they get pregnant.”
Dr. Gray reports being a site principal investigator for a phase 3 trial for VeraCept IUD, funded by Sebela Pharmaceuticals. Dr. Clowse reports receiving research funding and doing consulting for GlaxoSmithKline.
*Correction, 6/2/2022: A previous version of this article misstated the intended use of drugs such as the “morning-after” pill (levonorgestrel). They are taken to prevent unintended pregnancy.
A version of this article first appeared on Medscape.com .
Obstetrician Beverly Gray, MD, is already seeing the effects of the Roe v. Wade abortion debate in her North Carolina practice.
The state allows abortion but requires that women get counseling with a qualified health professional 72 hours before the procedure. “Aside from that, we still have patients asking for more efficacious contraceptive methods just in case,” said Dr. Gray, residency director and division director for women’s community and population health and associate professor for obstetrics and gynecology at Duke University, Durham, N.C.
Patients and staff in her clinic have also been approaching her about tubal ligation. “They’re asking about additional birth control methods because they’re concerned about what’s going to happen” with the challenge to the historic Roe v. Wade decision in the Supreme Court and subsequent actions in the states to restrict or ban abortion, she said.
This has implications not just for abortion but for medications known to affect pregnancy. “What I’m really worried about is physicians will be withholding medicine because they’re concerned about teratogenic effects,” said Dr. Gray.
With more states issuing restrictions on abortion, doctors are worried that patients needing certain drugs to maintain their lupus flares, cancer, or other diseases may decide not to take them in the event they accidentally become pregnant. If the drug is known to affect the fetus, the fear is a patient who lives in a state with abortion restrictions will no longer have the option to terminate a pregnancy.
Instead, a scenario may arise in which the patient – and their physician – may opt not to treat at all with an otherwise lifesaving medication, experts told this news organization.
The U.S. landscape on abortion restrictions
A leaked draft of a U.S. Supreme Court opinion on Mississippi’s 15-week abortion ban has sent the medical community into a tailspin. The case, Dobbs v. Jackson Women’s Health Organization, challenges the 1973 Roe v. Wade decision that affirms the constitutional right to abortion. It’s anticipated the high court will decide on the case in June.
Although the upcoming decision is subject to change, the draft indicated the high court would uphold the Mississippi ban. This would essentially overturn the 1973 ruling. An earlier Supreme Court decision allowing a Texas law banning abortion at 6 weeks suggests the court may already be heading in this direction. At the state level, legislatures have been moving on divergent paths – some taking steps to preserve abortion rights, others initiating restrictions.
More than 100 abortion restrictions in 19 states took effect in 2021, according to the Guttmacher Institute, which tracks such metrics. In 2022, “two key themes are anti-abortion policymakers’ continued pursuit of various types of abortion bans and restrictions on medication abortion,” the institute reported.
Forty-six states and the District of Columbia have introduced 2,025 restrictions or proactive measures on sexual and reproductive health and rights so far this year. The latest tally from Guttmacher, updated in late May, revealed that 11 states so far have enacted 42 abortion restrictions. A total of 6 states (Arizona, Florida, Idaho, Kentucky, Oklahoma, and Wyoming) have issued nine bans on abortion.
Comparatively, 11 states have enacted 19 protective abortion measures.
Twenty-two states have introduced 117 restrictions on medication abortions, which account for 54% of U.S. abortions. This includes seven measures that would ban medication abortion outright, according to Guttmacher. Kentucky and South Dakota collectively have enacted 14 restrictions on medication abortion, as well as provisions that ban mailing of abortion pills.
Chilling effect on prescribing
Some physicians anticipate that drugs such as the “morning-after” pill (levonorgestrel) will become less available as restrictions go into effect, since these are medications designed to prevent pregnancy.*
However, the ongoing effort to put a lid on abortion measures has prompted concerns about a trickle-down effect on other medications that are otherwise life-changing or lifesaving to patients but pose a risk to the fetus.
Several drugs are well documented to affect fetal growth and development of the fetus, ranging from mild, transitory effects to severe, permanent birth defects, said Ronald G. Grifka, MD, chief medical officer of University of Michigan Health-West and clinical professor of pediatrics at the University of Michigan Medical School, Ann Arbor. “As new medications are developed, we will need heightened attention to make sure they are safe for the fetus,” he added.
Certain teratogenic medications are associated with a high risk of abortion even though this isn’t their primary use, noted Christina Chambers, PhD, MPH, co-director of the Center for Better Beginnings and associate director with the Altman Clinical & Translational Research Institute at the University of California, San Diego.
“I don’t think anyone would intentionally take these drugs to induce spontaneous abortion. But if the drugs pose a risk for it, I can see how the laws might be stretched” to include them, said Dr. Chambers.
Methotrexate, a medication for autoimmune disorders, has a high risk of spontaneous abortion. So do acne medications such as isotretinoin.
Patients are usually told they’re not supposed to get pregnant on these drugs because there’s a high risk of pregnancy loss and risk of malformations and potential learning problems in the fetus. But many pregnancies aren’t planned, said Dr. Chambers. “Patients may forget about the side effects or think their birth control will protect them. And the next time they refill the medication, they may not hear about the warnings again.”
With a restrictive abortion law or ban in effect, a woman might think: “I won’t take this drug because if there’s any potential that I might get pregnant, I won’t have the option to abort an at-risk pregnancy.” Women and their doctors, for that matter, don’t want to put themselves in this position, said Dr. Chambers.
Rheumatologist Megan Clowse, MD, who prescribes several medications that potentially cause major birth defects and pregnancy loss, worries about the ramifications of these accumulating bans.
“Methotrexate has been a leading drug for us for decades for rheumatoid arthritis. Mycophenolate is a vital drug for lupus,” said Dr. Clowse, associate professor of medicine at Duke University’s division of rheumatology and immunology.
Both methotrexate and mycophenolate pose about a 40% risk of pregnancy loss and significantly increase the risk for birth defects. “I’m definitely concerned that there might be doctors or women who elect not to use those medications in women of reproductive age because of the potential risk for pregnancy and absence of abortion rights,” said Dr. Clowse.
These situations might force women to use contraceptives they don’t want to use, such as hormonal implants or intrauterine devices, she added. Another side effect is that women and their partners may decide to abstain from sex.
The iPLEDGE factor
Some rheumatology drugs like lenalidomide (Revlimid) require a valid negative pregnancy test in a lab every month. Similarly, the iPLEDGE Risk Evaluation and Mitigation Strategy seeks to reduce the teratogenicity of isotretinoin by requiring two types of birth control and regular pregnancy tests by users.
For isotretinoin specifically, abortion restrictions “could lead to increased adherence to pregnancy prevention measures which are already stringent in iPLEDGE. But on the other hand, it could lead to reduced willingness of physicians to prescribe or patients to take the medication,” said Dr. Chambers.
With programs like iPLEDGE in effect, the rate of pregnancies and abortions that occur in dermatology are relatively low, said Jenny Murase, MD, associate clinical professor of dermatology at the University of California, San Francisco.
Nevertheless, as a physician who regularly prescribes medications like isotretinoin in women of childbearing age, “it’s terrifying to me that a woman wouldn’t have the option to terminate the pregnancy if a teratogenic effect from the medication caused a severe birth defect,” said Dr. Murase.
Dermatologists use other teratogenic medications such as thalidomide, mycophenolate mofetil, and methotrexate for chronic dermatologic disease like psoriasis and atopic dermatitis.
The situation is especially tricky for dermatologists since most patients – about 80% – never discuss their pregnancy with their specialist prior to pregnancy initiation. Dr. Murase recalls when a patient with chronic plaque psoriasis on methotrexate in her late 40s became pregnant and had an abortion even before Dr. Murase became aware of the pregnancy.
Because dermatologists routinely prescribe long-term medications for chronic diseases like acne, psoriasis, and atopic dermatitis, it is important to have a conversation regarding the risks and benefits of long-term medication should a pregnancy occur in any woman of childbearing age, she said.
Fewer women in clinical trials?
Abortion restrictions could possibly discourage women of reproductive age to participate in a clinical trial for a new medication, said Dr. Chambers.
A female patient with a chronic disease who’s randomized to receive a new medication may be required to use certain types of birth control because of unknown potential adverse effects the drug may have on the fetus. But in some cases, accidental pregnancies happen.
The participant in the trial may say, “I don’t know enough about the safety of this drug in pregnancy, and I’ve already taken it. I want to terminate the pregnancy,” said Dr. Chambers. Thinking ahead, a woman may decide not to do the trial to avoid the risk of getting pregnant and not having the option to terminate the pregnancy.
This could apply to new drugs such as antiviral treatments, or medications for severe chronic disease that typically have no clinical trial data in pregnancy prior to initial release into the market.
Women may start taking the drug without thinking about getting pregnant, then realize there are no safety data and become concerned about its effects on a future pregnancy.
The question is: Will abortion restrictions have a chilling effect on these new drugs as well? Patients and their doctors may decide not to try it until more data are available. “I can see where abortion restrictions would change the risk or benefit calculation in thinking about what you do or don’t prescribe or take during reproductive age,” said Dr. Chambers.
The upside of restrictions?
If there’s a positive side to these developments with abortion bans, it may encourage women taking new medications or joining clinical trials to think even more carefully about adherence to effective contraception, said Dr. Chambers.
Some methods are more effective than others, she emphasized. “When you have an unplanned pregnancy, it could mean that the method you used wasn’t optimal or you weren’t using it as recommended.” A goal moving forward is to encourage more thoughtful use of highly effective contraceptives, thus reducing the number of unplanned pregnancies, she added.
If patients are taking methotrexate, “the time to think about pregnancy is before getting pregnant so you can switch to a drug that’s compatible with pregnancy,” she said.
This whole thought process regarding pregnancy planning could work toward useful health goals, said Dr. Chambers. “Nobody thinks termination is the preferred method, but planning ahead should involve a discussion of what works best for the patient.”
Patients do have other choices, said Dr. Grifka. “Fortunately, there are many commonly prescribed medications which cross the placenta and have no ill effects on the fetus.”
Talking to patients about choices
Dr. Clowse, who spends a lot of time training rheumatologists, encourages them to have conversations with patients about pregnancy planning. It’s a lot to manage, getting the right drug to a female patient with chronic illness, especially in this current climate of abortion upheaval, she noted.
Her approach is to have an open and honest conversation with patients about their concerns and fears, what the realities are, and what the potential future options are for certain rheumatology drugs in the United States.
Some women who see what’s happening across the country may become so risk averse that they may choose to die rather than take a lifesaving drug that poses certain risks under new restrictions.
“I think that’s tragic,” said Dr. Clowse.
To help their patients, Dr. Gray believes physicians across specialties should better educate themselves about physiology in pregnancy and how to counsel patients on the impact of not taking medications in pregnancy.
In her view, it’s almost coercive to say to a patient, “You really need to have effective contraception if I’m going to give you this lifesaving or quality-of-life-improving medication.”
When confronting such scenarios, Dr. Gray doesn’t think physicians need to change how they counsel patients about contraception. “I don’t think we should be putting pressure on patients to consider other permanent methods just because there’s a lack of abortion options.”
Patients will eventually make those decisions for themselves, she said. “They’re going to want a more efficacious method because they’re worried about not having access to abortion if they get pregnant.”
Dr. Gray reports being a site principal investigator for a phase 3 trial for VeraCept IUD, funded by Sebela Pharmaceuticals. Dr. Clowse reports receiving research funding and doing consulting for GlaxoSmithKline.
*Correction, 6/2/2022: A previous version of this article misstated the intended use of drugs such as the “morning-after” pill (levonorgestrel). They are taken to prevent unintended pregnancy.
A version of this article first appeared on Medscape.com .


















