User login
Capmatinib active against NSCLC with MET exon 14 mutations
MUNICH – The experimental agent capmatinib was associated with a high response rate when used in the first line for patients with advanced non–small cell lung cancers bearing MET exon 14–skipping mutations, said investigators in the Geometry MONO-1 trial.
Among a cohort of 25 patients with treatment-naive, MET exon 14–mutated non–small cell lung cancer (NSCLC), the primary endpoint of overall response rate (ORR) as determined by blinded, independent reviewers was 72%.
In contrast, the ORR among 69 patients who had received one or more prior lines of therapy was 39.1%, reported Juergen Wolf, MD, of University Hospital Cologne (Germany).
“The differential benefit observed between patients treated in the first line and relapsed [settings] highlights the need of early diagnosis of this aberration, and prompt targeted treatment of this challenging patient population,” he said at the European Society for Medical Oncology Congress.
MET exon14–skipping mutations occur in approximately 3%-4% of NSCLC cases. The mutation is thought to be an oncogenic driver and has been shown to be a poor prognostic factor for patients with advanced NSCLC. Patients with this mutation have poor responses to conventional therapy and immune checkpoint inhibitors, even when their tumors have high levels of programmed death–ligand 1 (PD-L1) and high mutational burden, Dr. Wolf said.
Capmatinib (INC280) is an oral, reversible inhibitor of the MET receptor tyrosine kinase and is highly selective for MET, with particular affinity for MET exon 14 mutations. It is also capable of crossing the blood-brain barrier and has shown activity in the brain in preliminary studies.
The Geometry MONO-1 trial is a phase 2 study of capmatinib in patients with stage IIIB/IV NSCLC with tumors that demonstrate MET amplification and/or carry the MET exon 14 mutation. Three study cohorts of patients with MET amplification were closed for futility. Dr. Wolf reported results from two cohorts of patients with MET exon 14–skipping mutations regardless of gene copy number: one with treatment-naive patients and the other with patients being treated in the second or third line.
As noted, the ORR in 25 patients in the treatment-naive cohort after a median follow-up of 5.6 months was 72%, including 18 partial responses and no complete responses. In addition, six patients (24%) had stable disease, for a disease control rate of 96%.
In the pretreated cohort, however, there were no complete responses among 69 patients, and 27 patients (39.1%) had partial responses. In this cohort, an additional 26 patients (37.7%) had stable disease, for an ORR of 39.1% and disease-control rate of 78.3%.
Dr. Wolf also highlighted preliminary evidence of capmatinib activity in the brain. He noted that one patient, an 80-year-old woman with multiple untreated brain metastases as well as lesions in dermal lymph nodes, liver, and pleura, had complete resolution of brain metastases at the first postbaseline CT scan, 42 days after starting capmatinib. The duration of response was 11.3 months, at which point the patient discontinued the drug because of extracranial progressive disease.
Among all patients in all study cohorts (302) the most common grade 3 or 4 adverse events were peripheral edema, dyspnea, fatigue, nausea, vomiting, and decreased appetite. Adverse drug-related events (grade 3 or 4) included peripheral edema, nausea, vomiting, fatigue, and decreased appetite. In all, 10.3% of patients discontinued for adverse events suspected to be related to capmatinib.
Invited discussant James Chih-Hsin Yang, MD, PhD, from the National Taiwan University Hospital in Taipei, said that the study shows that the MET exon 14–skipping mutation is an oncogenic driver and that capmatinib is an effective tyrosine kinase inhibitor (TKI) for patients with NSCLC harboring this mutation.
Questions that still need to be answered, he said, include whether patients with the mutation are heterogeneous and may have differing response to TKIs, how long the duration of response is, how long it will take for resistance to capmatinib to occur, how it compares with other MET inhibitors, and if there are additional biomarkers that could help select patients for treatment with the novel agent.
The study was funded by Novartis. Dr. Wolf reported advisory board participation, institutional research support, and lecture fees from Novartis and others. Dr. Yang reported honoraria from advisory board participation and/or speaking from Novartis and others. His institution participated in the Geometry MONO-1 study, but he was not personally involved.
MUNICH – The experimental agent capmatinib was associated with a high response rate when used in the first line for patients with advanced non–small cell lung cancers bearing MET exon 14–skipping mutations, said investigators in the Geometry MONO-1 trial.
Among a cohort of 25 patients with treatment-naive, MET exon 14–mutated non–small cell lung cancer (NSCLC), the primary endpoint of overall response rate (ORR) as determined by blinded, independent reviewers was 72%.
In contrast, the ORR among 69 patients who had received one or more prior lines of therapy was 39.1%, reported Juergen Wolf, MD, of University Hospital Cologne (Germany).
“The differential benefit observed between patients treated in the first line and relapsed [settings] highlights the need of early diagnosis of this aberration, and prompt targeted treatment of this challenging patient population,” he said at the European Society for Medical Oncology Congress.
MET exon14–skipping mutations occur in approximately 3%-4% of NSCLC cases. The mutation is thought to be an oncogenic driver and has been shown to be a poor prognostic factor for patients with advanced NSCLC. Patients with this mutation have poor responses to conventional therapy and immune checkpoint inhibitors, even when their tumors have high levels of programmed death–ligand 1 (PD-L1) and high mutational burden, Dr. Wolf said.
Capmatinib (INC280) is an oral, reversible inhibitor of the MET receptor tyrosine kinase and is highly selective for MET, with particular affinity for MET exon 14 mutations. It is also capable of crossing the blood-brain barrier and has shown activity in the brain in preliminary studies.
The Geometry MONO-1 trial is a phase 2 study of capmatinib in patients with stage IIIB/IV NSCLC with tumors that demonstrate MET amplification and/or carry the MET exon 14 mutation. Three study cohorts of patients with MET amplification were closed for futility. Dr. Wolf reported results from two cohorts of patients with MET exon 14–skipping mutations regardless of gene copy number: one with treatment-naive patients and the other with patients being treated in the second or third line.
As noted, the ORR in 25 patients in the treatment-naive cohort after a median follow-up of 5.6 months was 72%, including 18 partial responses and no complete responses. In addition, six patients (24%) had stable disease, for a disease control rate of 96%.
In the pretreated cohort, however, there were no complete responses among 69 patients, and 27 patients (39.1%) had partial responses. In this cohort, an additional 26 patients (37.7%) had stable disease, for an ORR of 39.1% and disease-control rate of 78.3%.
Dr. Wolf also highlighted preliminary evidence of capmatinib activity in the brain. He noted that one patient, an 80-year-old woman with multiple untreated brain metastases as well as lesions in dermal lymph nodes, liver, and pleura, had complete resolution of brain metastases at the first postbaseline CT scan, 42 days after starting capmatinib. The duration of response was 11.3 months, at which point the patient discontinued the drug because of extracranial progressive disease.
Among all patients in all study cohorts (302) the most common grade 3 or 4 adverse events were peripheral edema, dyspnea, fatigue, nausea, vomiting, and decreased appetite. Adverse drug-related events (grade 3 or 4) included peripheral edema, nausea, vomiting, fatigue, and decreased appetite. In all, 10.3% of patients discontinued for adverse events suspected to be related to capmatinib.
Invited discussant James Chih-Hsin Yang, MD, PhD, from the National Taiwan University Hospital in Taipei, said that the study shows that the MET exon 14–skipping mutation is an oncogenic driver and that capmatinib is an effective tyrosine kinase inhibitor (TKI) for patients with NSCLC harboring this mutation.
Questions that still need to be answered, he said, include whether patients with the mutation are heterogeneous and may have differing response to TKIs, how long the duration of response is, how long it will take for resistance to capmatinib to occur, how it compares with other MET inhibitors, and if there are additional biomarkers that could help select patients for treatment with the novel agent.
The study was funded by Novartis. Dr. Wolf reported advisory board participation, institutional research support, and lecture fees from Novartis and others. Dr. Yang reported honoraria from advisory board participation and/or speaking from Novartis and others. His institution participated in the Geometry MONO-1 study, but he was not personally involved.
MUNICH – The experimental agent capmatinib was associated with a high response rate when used in the first line for patients with advanced non–small cell lung cancers bearing MET exon 14–skipping mutations, said investigators in the Geometry MONO-1 trial.
Among a cohort of 25 patients with treatment-naive, MET exon 14–mutated non–small cell lung cancer (NSCLC), the primary endpoint of overall response rate (ORR) as determined by blinded, independent reviewers was 72%.
In contrast, the ORR among 69 patients who had received one or more prior lines of therapy was 39.1%, reported Juergen Wolf, MD, of University Hospital Cologne (Germany).
“The differential benefit observed between patients treated in the first line and relapsed [settings] highlights the need of early diagnosis of this aberration, and prompt targeted treatment of this challenging patient population,” he said at the European Society for Medical Oncology Congress.
MET exon14–skipping mutations occur in approximately 3%-4% of NSCLC cases. The mutation is thought to be an oncogenic driver and has been shown to be a poor prognostic factor for patients with advanced NSCLC. Patients with this mutation have poor responses to conventional therapy and immune checkpoint inhibitors, even when their tumors have high levels of programmed death–ligand 1 (PD-L1) and high mutational burden, Dr. Wolf said.
Capmatinib (INC280) is an oral, reversible inhibitor of the MET receptor tyrosine kinase and is highly selective for MET, with particular affinity for MET exon 14 mutations. It is also capable of crossing the blood-brain barrier and has shown activity in the brain in preliminary studies.
The Geometry MONO-1 trial is a phase 2 study of capmatinib in patients with stage IIIB/IV NSCLC with tumors that demonstrate MET amplification and/or carry the MET exon 14 mutation. Three study cohorts of patients with MET amplification were closed for futility. Dr. Wolf reported results from two cohorts of patients with MET exon 14–skipping mutations regardless of gene copy number: one with treatment-naive patients and the other with patients being treated in the second or third line.
As noted, the ORR in 25 patients in the treatment-naive cohort after a median follow-up of 5.6 months was 72%, including 18 partial responses and no complete responses. In addition, six patients (24%) had stable disease, for a disease control rate of 96%.
In the pretreated cohort, however, there were no complete responses among 69 patients, and 27 patients (39.1%) had partial responses. In this cohort, an additional 26 patients (37.7%) had stable disease, for an ORR of 39.1% and disease-control rate of 78.3%.
Dr. Wolf also highlighted preliminary evidence of capmatinib activity in the brain. He noted that one patient, an 80-year-old woman with multiple untreated brain metastases as well as lesions in dermal lymph nodes, liver, and pleura, had complete resolution of brain metastases at the first postbaseline CT scan, 42 days after starting capmatinib. The duration of response was 11.3 months, at which point the patient discontinued the drug because of extracranial progressive disease.
Among all patients in all study cohorts (302) the most common grade 3 or 4 adverse events were peripheral edema, dyspnea, fatigue, nausea, vomiting, and decreased appetite. Adverse drug-related events (grade 3 or 4) included peripheral edema, nausea, vomiting, fatigue, and decreased appetite. In all, 10.3% of patients discontinued for adverse events suspected to be related to capmatinib.
Invited discussant James Chih-Hsin Yang, MD, PhD, from the National Taiwan University Hospital in Taipei, said that the study shows that the MET exon 14–skipping mutation is an oncogenic driver and that capmatinib is an effective tyrosine kinase inhibitor (TKI) for patients with NSCLC harboring this mutation.
Questions that still need to be answered, he said, include whether patients with the mutation are heterogeneous and may have differing response to TKIs, how long the duration of response is, how long it will take for resistance to capmatinib to occur, how it compares with other MET inhibitors, and if there are additional biomarkers that could help select patients for treatment with the novel agent.
The study was funded by Novartis. Dr. Wolf reported advisory board participation, institutional research support, and lecture fees from Novartis and others. Dr. Yang reported honoraria from advisory board participation and/or speaking from Novartis and others. His institution participated in the Geometry MONO-1 study, but he was not personally involved.
REPORTING FROM ESMO 2018
Key clinical point: Patients with non–small cell lung cancer bearing a MET exon 14–skipping mutation had high overall response rates to the MET inhibitor capmatinib.
Major finding: The overall response rate in treatment-naive patients was 72%.
Study details: A phase 2 trial with previously treated and untreated patients with advanced non–small cell lung cancers bearing MET exon 14–skipping mutations.
Disclosures: The study was funded by Novartis. Dr. Wolf reported advisory board participation and lecture fees from Novartis and others and institutional research support from Novartis and others. Dr. Yang reported honoraria from advisory board participation and/or speaking from Novartis and others. His institution participated in the Geometry MONO-1 study, but he was not personally involved.
Cigarette smoking at lowest level ever
“This new all-time low in cigarette smoking among U.S. adults is a tremendous public health accomplishment, and it demonstrates the importance of continued proven strategies to reduce smoking,” CDC Director Robert Redfield said in a written statement.
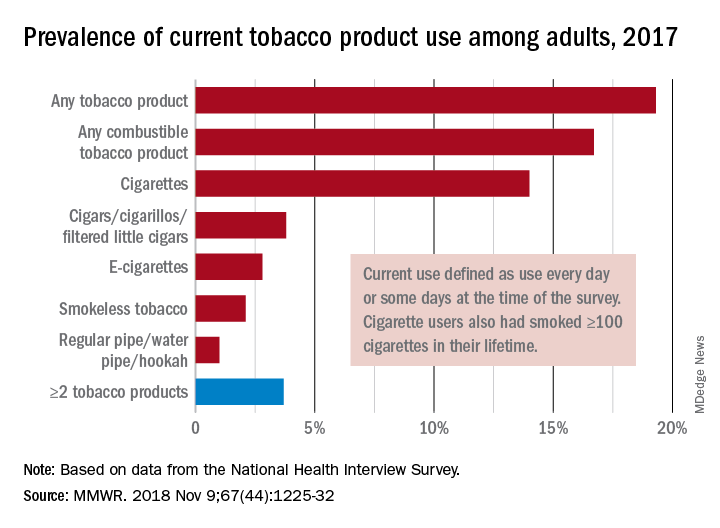
In 2017, 19.3% of adults aged 18 years and older – approximately 47.4 million Americans – reported current use of some type of tobacco product, and current use of combustible tobacco was 16.7%, Teresa W. Wang, PhD, of the CDC’s National Center for Chronic Disease Prevention and Health Promotion, Atlanta, and her associates reported in the Morbidity and Mortality Weekly Report. Current use was defined as use every day or some days, with an added requirement of at least 100 cigarettes in a lifetime added for cigarette smokers.
Data from the National Health Interview Survey showed that from 2016 to 2017, current use declined for any tobacco product, any combustible tobacco product, cigarettes, smokeless tobacco, and the combination of two or more tobacco products. The most common combination in 2017 was cigarettes and e-cigarettes, which was reported by 30.1% of the 9 million adults who used more than one product, Dr. Wang and her associates said.
Prevalence of current tobacco use was higher among men than women (24.8% vs. 14.2%), and adults aged 25-44 years (22.5%) had the highest level by age, followed by those aged 45-64 years (21.3%), 18-24 years (18.3%), and 65 years or older (11%). Use by race/ethnicity was highest among American Indian/Alaska Natives (29.8%), with the Midwest putting up the highest prevalence by region at 23.5%, they said.
“Although cigarette smoking among U.S. adults has declined considerably, tobacco products have evolved in recent years to include various combustible, noncombustible, and electronic products,” Dr. Wang and her associates wrote. “Implementation of evidence-based tobacco control interventions that address the diversity of tobacco products used by U.S. adults, in coordination with regulation of tobacco product manufacturing, marketing, and sales, can reduce tobacco-related disease and death in the United States.”
SOURCE: Wang TW et al. MMWR. 2018 Nov 9;67[44]:1225-32.

“This new all-time low in cigarette smoking among U.S. adults is a tremendous public health accomplishment, and it demonstrates the importance of continued proven strategies to reduce smoking,” CDC Director Robert Redfield said in a written statement.
In 2017, 19.3% of adults aged 18 years and older – approximately 47.4 million Americans – reported current use of some type of tobacco product, and current use of combustible tobacco was 16.7%, Teresa W. Wang, PhD, of the CDC’s National Center for Chronic Disease Prevention and Health Promotion, Atlanta, and her associates reported in the Morbidity and Mortality Weekly Report. Current use was defined as use every day or some days, with an added requirement of at least 100 cigarettes in a lifetime added for cigarette smokers.
Data from the National Health Interview Survey showed that from 2016 to 2017, current use declined for any tobacco product, any combustible tobacco product, cigarettes, smokeless tobacco, and the combination of two or more tobacco products. The most common combination in 2017 was cigarettes and e-cigarettes, which was reported by 30.1% of the 9 million adults who used more than one product, Dr. Wang and her associates said.
Prevalence of current tobacco use was higher among men than women (24.8% vs. 14.2%), and adults aged 25-44 years (22.5%) had the highest level by age, followed by those aged 45-64 years (21.3%), 18-24 years (18.3%), and 65 years or older (11%). Use by race/ethnicity was highest among American Indian/Alaska Natives (29.8%), with the Midwest putting up the highest prevalence by region at 23.5%, they said.
“Although cigarette smoking among U.S. adults has declined considerably, tobacco products have evolved in recent years to include various combustible, noncombustible, and electronic products,” Dr. Wang and her associates wrote. “Implementation of evidence-based tobacco control interventions that address the diversity of tobacco products used by U.S. adults, in coordination with regulation of tobacco product manufacturing, marketing, and sales, can reduce tobacco-related disease and death in the United States.”
SOURCE: Wang TW et al. MMWR. 2018 Nov 9;67[44]:1225-32.

“This new all-time low in cigarette smoking among U.S. adults is a tremendous public health accomplishment, and it demonstrates the importance of continued proven strategies to reduce smoking,” CDC Director Robert Redfield said in a written statement.
In 2017, 19.3% of adults aged 18 years and older – approximately 47.4 million Americans – reported current use of some type of tobacco product, and current use of combustible tobacco was 16.7%, Teresa W. Wang, PhD, of the CDC’s National Center for Chronic Disease Prevention and Health Promotion, Atlanta, and her associates reported in the Morbidity and Mortality Weekly Report. Current use was defined as use every day or some days, with an added requirement of at least 100 cigarettes in a lifetime added for cigarette smokers.
Data from the National Health Interview Survey showed that from 2016 to 2017, current use declined for any tobacco product, any combustible tobacco product, cigarettes, smokeless tobacco, and the combination of two or more tobacco products. The most common combination in 2017 was cigarettes and e-cigarettes, which was reported by 30.1% of the 9 million adults who used more than one product, Dr. Wang and her associates said.
Prevalence of current tobacco use was higher among men than women (24.8% vs. 14.2%), and adults aged 25-44 years (22.5%) had the highest level by age, followed by those aged 45-64 years (21.3%), 18-24 years (18.3%), and 65 years or older (11%). Use by race/ethnicity was highest among American Indian/Alaska Natives (29.8%), with the Midwest putting up the highest prevalence by region at 23.5%, they said.
“Although cigarette smoking among U.S. adults has declined considerably, tobacco products have evolved in recent years to include various combustible, noncombustible, and electronic products,” Dr. Wang and her associates wrote. “Implementation of evidence-based tobacco control interventions that address the diversity of tobacco products used by U.S. adults, in coordination with regulation of tobacco product manufacturing, marketing, and sales, can reduce tobacco-related disease and death in the United States.”
SOURCE: Wang TW et al. MMWR. 2018 Nov 9;67[44]:1225-32.
FROM MMWR
FDA approves lorlatinib as second line for ALK-positive advanced NSCLC
The Food and Drug Administration has granted accelerated approval to lorlatinib for patients with anaplastic lymphoma kinase (ALK)–positive metastatic non–small cell lung cancer (NSCLC) whose disease has progressed on crizotinib and at least one other ALK inhibitor for metastatic disease or whose disease has progressed on alectinib or ceritinib as the first ALK inhibitor therapy for metastatic disease.
Approval of the next-generation ALK inhibitor was based on an overall response rate of 48% – with 4% complete and 44% partial – in a subgroup of 215 patients with ALK-positive metastatic NSCLC enrolled in a nonrandomized, phase 2 trial, the FDA said in a press announcement. All patients had been previously treated with one or more ALK kinase inhibitors.
The median response duration was 12.5 months (95% confidence interval, 8.4-23.7) and the intracranial overall response rate in 89 patients with measurable lesions in the CNS was 60% (95% CI, 49-70) with 21% complete and 38% partial responses.
Common adverse reactions in patients receiving lorlatinib were edema, peripheral neuropathy, cognitive effects, dyspnea, fatigue, weight gain, arthralgia, mood effects, and diarrhea. The most common laboratory abnormalities were hypercholesterolemia and hypertriglyceridemia, the FDA said.
The recommended dose of lorlatinib, to be marketed as Lorbrena by Pfizer, is 100 mg orally once daily.
The Food and Drug Administration has granted accelerated approval to lorlatinib for patients with anaplastic lymphoma kinase (ALK)–positive metastatic non–small cell lung cancer (NSCLC) whose disease has progressed on crizotinib and at least one other ALK inhibitor for metastatic disease or whose disease has progressed on alectinib or ceritinib as the first ALK inhibitor therapy for metastatic disease.
Approval of the next-generation ALK inhibitor was based on an overall response rate of 48% – with 4% complete and 44% partial – in a subgroup of 215 patients with ALK-positive metastatic NSCLC enrolled in a nonrandomized, phase 2 trial, the FDA said in a press announcement. All patients had been previously treated with one or more ALK kinase inhibitors.
The median response duration was 12.5 months (95% confidence interval, 8.4-23.7) and the intracranial overall response rate in 89 patients with measurable lesions in the CNS was 60% (95% CI, 49-70) with 21% complete and 38% partial responses.
Common adverse reactions in patients receiving lorlatinib were edema, peripheral neuropathy, cognitive effects, dyspnea, fatigue, weight gain, arthralgia, mood effects, and diarrhea. The most common laboratory abnormalities were hypercholesterolemia and hypertriglyceridemia, the FDA said.
The recommended dose of lorlatinib, to be marketed as Lorbrena by Pfizer, is 100 mg orally once daily.
The Food and Drug Administration has granted accelerated approval to lorlatinib for patients with anaplastic lymphoma kinase (ALK)–positive metastatic non–small cell lung cancer (NSCLC) whose disease has progressed on crizotinib and at least one other ALK inhibitor for metastatic disease or whose disease has progressed on alectinib or ceritinib as the first ALK inhibitor therapy for metastatic disease.
Approval of the next-generation ALK inhibitor was based on an overall response rate of 48% – with 4% complete and 44% partial – in a subgroup of 215 patients with ALK-positive metastatic NSCLC enrolled in a nonrandomized, phase 2 trial, the FDA said in a press announcement. All patients had been previously treated with one or more ALK kinase inhibitors.
The median response duration was 12.5 months (95% confidence interval, 8.4-23.7) and the intracranial overall response rate in 89 patients with measurable lesions in the CNS was 60% (95% CI, 49-70) with 21% complete and 38% partial responses.
Common adverse reactions in patients receiving lorlatinib were edema, peripheral neuropathy, cognitive effects, dyspnea, fatigue, weight gain, arthralgia, mood effects, and diarrhea. The most common laboratory abnormalities were hypercholesterolemia and hypertriglyceridemia, the FDA said.
The recommended dose of lorlatinib, to be marketed as Lorbrena by Pfizer, is 100 mg orally once daily.
FDA expands approval of pembrolizumab in NSCLC
The Food and Drug Administration .
The drug is now approved for use in combination with carboplatin and either paclitaxel or nanoparticle albumin–bound (nab) paclitaxel for the first-line treatment of NSCLC, regardless of PD-L1 expression status.
This makes pembrolizumab the first anti-PD-1 therapy approved in the first-line setting both as monotherapy and in combination treatment for certain patients with metastatic NSCLC. All appropriate patients with metastatic squamous NSCLC or metastatic nonsquamous NSCLC and no EGFR or ALK mutations are now eligible to receive pembrolizumab-based treatment first-line.
The FDA’s approval is based on results from the phase 3 KEYNOTE-407 trial. This randomized, double-blind study enrolled patients with metastatic squamous NSCLC, regardless of tumor PD-L1 expression status, who had received no prior systemic treatment for metastatic disease.
Patients in the pembrolizumab arm (n = 278) received pembrolizumab and carboplatin every 3 weeks for four cycles, plus paclitaxel every 3 weeks for four cycles or nab-paclitaxel on days 1, 8, and 15 of every 3-week cycle for four cycles, followed by pembrolizumab every 3 weeks.
Patients in the control arm (n = 281) received the same regimen of carboplatin and paclitaxel/nab-paclitaxel, but placebo instead of pembrolizumab.
There was a significant improvement in overall response rate, progression-free survival, and overall survival in patients who received pembrolizumab.
The overall response rate was 58% in the pembrolizumab arm and 35% in the placebo arm (P = .0008). The median duration of response was 7.2 months and 4.9 months, respectively.
The median progression-free survival was 6.4 months in the pembrolizumab arm and 4.8 months in the placebo arm (P less than .0001). The median overall survival was 15.9 months and 11.3 months, respectively (P = .0017).
Safety data are available for the first 203 patients treated on the trial, 101 of them in the pembrolizumab arm.
Fifteen percent of patients discontinued pembrolizumab because of adverse events (AEs), and 43% of patients on pembrolizumab experienced AEs leading to dose interruption.
The most common AEs leading to dose interruption in the pembrolizumab arm were thrombocytopenia, neutropenia, anemia, asthenia, and diarrhea. The most frequent serious AEs in the pembrolizumab arm were febrile neutropenia, pneumonia, and urinary tract infection.
Additional details on this trial are available in the prescribing information, which can be found on the Keytruda website.
The Food and Drug Administration .
The drug is now approved for use in combination with carboplatin and either paclitaxel or nanoparticle albumin–bound (nab) paclitaxel for the first-line treatment of NSCLC, regardless of PD-L1 expression status.
This makes pembrolizumab the first anti-PD-1 therapy approved in the first-line setting both as monotherapy and in combination treatment for certain patients with metastatic NSCLC. All appropriate patients with metastatic squamous NSCLC or metastatic nonsquamous NSCLC and no EGFR or ALK mutations are now eligible to receive pembrolizumab-based treatment first-line.
The FDA’s approval is based on results from the phase 3 KEYNOTE-407 trial. This randomized, double-blind study enrolled patients with metastatic squamous NSCLC, regardless of tumor PD-L1 expression status, who had received no prior systemic treatment for metastatic disease.
Patients in the pembrolizumab arm (n = 278) received pembrolizumab and carboplatin every 3 weeks for four cycles, plus paclitaxel every 3 weeks for four cycles or nab-paclitaxel on days 1, 8, and 15 of every 3-week cycle for four cycles, followed by pembrolizumab every 3 weeks.
Patients in the control arm (n = 281) received the same regimen of carboplatin and paclitaxel/nab-paclitaxel, but placebo instead of pembrolizumab.
There was a significant improvement in overall response rate, progression-free survival, and overall survival in patients who received pembrolizumab.
The overall response rate was 58% in the pembrolizumab arm and 35% in the placebo arm (P = .0008). The median duration of response was 7.2 months and 4.9 months, respectively.
The median progression-free survival was 6.4 months in the pembrolizumab arm and 4.8 months in the placebo arm (P less than .0001). The median overall survival was 15.9 months and 11.3 months, respectively (P = .0017).
Safety data are available for the first 203 patients treated on the trial, 101 of them in the pembrolizumab arm.
Fifteen percent of patients discontinued pembrolizumab because of adverse events (AEs), and 43% of patients on pembrolizumab experienced AEs leading to dose interruption.
The most common AEs leading to dose interruption in the pembrolizumab arm were thrombocytopenia, neutropenia, anemia, asthenia, and diarrhea. The most frequent serious AEs in the pembrolizumab arm were febrile neutropenia, pneumonia, and urinary tract infection.
Additional details on this trial are available in the prescribing information, which can be found on the Keytruda website.
The Food and Drug Administration .
The drug is now approved for use in combination with carboplatin and either paclitaxel or nanoparticle albumin–bound (nab) paclitaxel for the first-line treatment of NSCLC, regardless of PD-L1 expression status.
This makes pembrolizumab the first anti-PD-1 therapy approved in the first-line setting both as monotherapy and in combination treatment for certain patients with metastatic NSCLC. All appropriate patients with metastatic squamous NSCLC or metastatic nonsquamous NSCLC and no EGFR or ALK mutations are now eligible to receive pembrolizumab-based treatment first-line.
The FDA’s approval is based on results from the phase 3 KEYNOTE-407 trial. This randomized, double-blind study enrolled patients with metastatic squamous NSCLC, regardless of tumor PD-L1 expression status, who had received no prior systemic treatment for metastatic disease.
Patients in the pembrolizumab arm (n = 278) received pembrolizumab and carboplatin every 3 weeks for four cycles, plus paclitaxel every 3 weeks for four cycles or nab-paclitaxel on days 1, 8, and 15 of every 3-week cycle for four cycles, followed by pembrolizumab every 3 weeks.
Patients in the control arm (n = 281) received the same regimen of carboplatin and paclitaxel/nab-paclitaxel, but placebo instead of pembrolizumab.
There was a significant improvement in overall response rate, progression-free survival, and overall survival in patients who received pembrolizumab.
The overall response rate was 58% in the pembrolizumab arm and 35% in the placebo arm (P = .0008). The median duration of response was 7.2 months and 4.9 months, respectively.
The median progression-free survival was 6.4 months in the pembrolizumab arm and 4.8 months in the placebo arm (P less than .0001). The median overall survival was 15.9 months and 11.3 months, respectively (P = .0017).
Safety data are available for the first 203 patients treated on the trial, 101 of them in the pembrolizumab arm.
Fifteen percent of patients discontinued pembrolizumab because of adverse events (AEs), and 43% of patients on pembrolizumab experienced AEs leading to dose interruption.
The most common AEs leading to dose interruption in the pembrolizumab arm were thrombocytopenia, neutropenia, anemia, asthenia, and diarrhea. The most frequent serious AEs in the pembrolizumab arm were febrile neutropenia, pneumonia, and urinary tract infection.
Additional details on this trial are available in the prescribing information, which can be found on the Keytruda website.
Smoking neglected in patients with PAD
Patients with claudication consulting a peripheral arterial disease provider are often active smokers, rarely receive evidence-based cessation interventions, and frequently relapse if they do quit, according to a report published online in the Journal of the American Heart Association.
More than one-third of patients with claudication consulting PAD specialists are active smokers, as seen in a data analysis of an international registry, wrote Krishna K. Patel, MD, of the department of cardiology, University of Missouri–Kansas City, and her colleagues.
The authors assessed 1,272 patients with PAD and new or worsening claudication who were enrolled at 16 vascular specialty clinics from 2011 to 2015 in the PORTRAIT (Patient-Centered Outcomes Related to Treatment Practices in Peripheral Arterial Disease: Investigating Trajectories) registry, (Clinicaltrials.gov: NCT01419080).
In-person interviews obtained smoking status from the patients and information on cessation interventions at baseline and at 3, 6, and 12 months. At baseline, 474 (37%) patients were active, 660 (52%) were former, and 138 (11%) were never smokers.
Among active smokers, only 16% were referred to cessation counseling, and only 11% were prescribed pharmacologic treatment.
At 3 months, the probability of quitting smoking was 21%. Those who kept smoking had a probability of quitting during the next 9 months that varied between 11% and 12% (P less than .001). The probability of relapse was high, with more than one-third of initial quitters (36%) resuming smoking, and at 12 months; 72% of all original smokers continued to smoke, according to the authors.
The high level of initial smoking and the failed efforts at attempting cessation are clinically important because cigarette smoking is the most important and modifiable risk factor for PAD, and patients with PAD who smoke have higher rates of disease progression, according to Dr. Patel and her colleagues.
“Few patients receive formal cessation interventions. The dynamic nature of these patients’ smoking practices also underscores the need for ongoing assessment of smoking, even among those who report that they have quit, and consistent offering of evidence-based cessation support. Future research should focus on identifying optimal strategies for implementing consistent cessation support,” the researchers concluded.
The study was funded by grants from the Netherlands Organization for Scientific Research and an unrestricted grant from W. L. Gore & Associates. One of the authors owns the copyright for a Peripheral Artery Questionnaire used in the study and serves as a consultant to United Healthcare, Bayer, and Novartis, with research grants from Abbot Vascular and Novartis. Another author is supported by an unrestricted research grant by Merck and Boston Scientific. The remaining authors reported having no disclosures.
SOURCE: Patel KR et al. J Am Heart Assoc. 2018;7:e010076. doi: 10.1161/JAHA.118.010076.
Patients with claudication consulting a peripheral arterial disease provider are often active smokers, rarely receive evidence-based cessation interventions, and frequently relapse if they do quit, according to a report published online in the Journal of the American Heart Association.
More than one-third of patients with claudication consulting PAD specialists are active smokers, as seen in a data analysis of an international registry, wrote Krishna K. Patel, MD, of the department of cardiology, University of Missouri–Kansas City, and her colleagues.
The authors assessed 1,272 patients with PAD and new or worsening claudication who were enrolled at 16 vascular specialty clinics from 2011 to 2015 in the PORTRAIT (Patient-Centered Outcomes Related to Treatment Practices in Peripheral Arterial Disease: Investigating Trajectories) registry, (Clinicaltrials.gov: NCT01419080).
In-person interviews obtained smoking status from the patients and information on cessation interventions at baseline and at 3, 6, and 12 months. At baseline, 474 (37%) patients were active, 660 (52%) were former, and 138 (11%) were never smokers.
Among active smokers, only 16% were referred to cessation counseling, and only 11% were prescribed pharmacologic treatment.
At 3 months, the probability of quitting smoking was 21%. Those who kept smoking had a probability of quitting during the next 9 months that varied between 11% and 12% (P less than .001). The probability of relapse was high, with more than one-third of initial quitters (36%) resuming smoking, and at 12 months; 72% of all original smokers continued to smoke, according to the authors.
The high level of initial smoking and the failed efforts at attempting cessation are clinically important because cigarette smoking is the most important and modifiable risk factor for PAD, and patients with PAD who smoke have higher rates of disease progression, according to Dr. Patel and her colleagues.
“Few patients receive formal cessation interventions. The dynamic nature of these patients’ smoking practices also underscores the need for ongoing assessment of smoking, even among those who report that they have quit, and consistent offering of evidence-based cessation support. Future research should focus on identifying optimal strategies for implementing consistent cessation support,” the researchers concluded.
The study was funded by grants from the Netherlands Organization for Scientific Research and an unrestricted grant from W. L. Gore & Associates. One of the authors owns the copyright for a Peripheral Artery Questionnaire used in the study and serves as a consultant to United Healthcare, Bayer, and Novartis, with research grants from Abbot Vascular and Novartis. Another author is supported by an unrestricted research grant by Merck and Boston Scientific. The remaining authors reported having no disclosures.
SOURCE: Patel KR et al. J Am Heart Assoc. 2018;7:e010076. doi: 10.1161/JAHA.118.010076.
Patients with claudication consulting a peripheral arterial disease provider are often active smokers, rarely receive evidence-based cessation interventions, and frequently relapse if they do quit, according to a report published online in the Journal of the American Heart Association.
More than one-third of patients with claudication consulting PAD specialists are active smokers, as seen in a data analysis of an international registry, wrote Krishna K. Patel, MD, of the department of cardiology, University of Missouri–Kansas City, and her colleagues.
The authors assessed 1,272 patients with PAD and new or worsening claudication who were enrolled at 16 vascular specialty clinics from 2011 to 2015 in the PORTRAIT (Patient-Centered Outcomes Related to Treatment Practices in Peripheral Arterial Disease: Investigating Trajectories) registry, (Clinicaltrials.gov: NCT01419080).
In-person interviews obtained smoking status from the patients and information on cessation interventions at baseline and at 3, 6, and 12 months. At baseline, 474 (37%) patients were active, 660 (52%) were former, and 138 (11%) were never smokers.
Among active smokers, only 16% were referred to cessation counseling, and only 11% were prescribed pharmacologic treatment.
At 3 months, the probability of quitting smoking was 21%. Those who kept smoking had a probability of quitting during the next 9 months that varied between 11% and 12% (P less than .001). The probability of relapse was high, with more than one-third of initial quitters (36%) resuming smoking, and at 12 months; 72% of all original smokers continued to smoke, according to the authors.
The high level of initial smoking and the failed efforts at attempting cessation are clinically important because cigarette smoking is the most important and modifiable risk factor for PAD, and patients with PAD who smoke have higher rates of disease progression, according to Dr. Patel and her colleagues.
“Few patients receive formal cessation interventions. The dynamic nature of these patients’ smoking practices also underscores the need for ongoing assessment of smoking, even among those who report that they have quit, and consistent offering of evidence-based cessation support. Future research should focus on identifying optimal strategies for implementing consistent cessation support,” the researchers concluded.
The study was funded by grants from the Netherlands Organization for Scientific Research and an unrestricted grant from W. L. Gore & Associates. One of the authors owns the copyright for a Peripheral Artery Questionnaire used in the study and serves as a consultant to United Healthcare, Bayer, and Novartis, with research grants from Abbot Vascular and Novartis. Another author is supported by an unrestricted research grant by Merck and Boston Scientific. The remaining authors reported having no disclosures.
SOURCE: Patel KR et al. J Am Heart Assoc. 2018;7:e010076. doi: 10.1161/JAHA.118.010076.
FROM THE JOURNAL OF THE AMERICAN HEART ASSOCIATION
Key clinical point: Patients with PAD are often smokers, rarely receive cessation interventions, and frequently relapse if they quit.
Major finding: Only 16% of active smokers received cessation counseling, and at 3 months, the probability of quitting smoking was 21%.
Study details: Interview study of 1,272 patients with PAD and new or worsening claudication who were enrolled in the PORTRAIT registry.
Disclosures: Study funding included an unrestricted grant from W. L. Gore & Associates. One author owns the copyright for the Peripheral Artery Questionnaire used in the study and has ties to several pharmaceutical companies. Another author is supported by an unrestricted corporate research grant. The remaining authors reported having no disclosures.
Source: Patel KR et al. J Am Heart Assoc. 2018;7:e010076. doi: 10.1161/JAHA.118.010076.
Checkpoint inhibitor seems safe and effective for patients with HIV
MUNICH – Patients with HIV who are treated with nivolumab, a programmed death-1 (PD-1) inhibitor, appear to have similar safety and efficacy outcomes compared with HIV-negative patients treated with the same agent, investigators found.
The retrospective study also showed that viral load and CD4 status were largely unchanged by immunotherapy, lead author Aurélien Gobert, MD, of Groupe Hospitalier Pitié Salpêtrière, Paris, reported at the European Society for Medical Oncology Congress.
HIV increases risks of certain cancer types, Dr. Gobert said in a press release. “These patients are at higher risk for a number of cancers: AIDS-defining forms, the diagnosis of which results in the categorization of a person as suffering from AIDS, but also various other types that they are two to three times more likely to develop than in the general population, such as anal, skin, head and neck, and lung cancer,” he said.
Despite the increased risks, few studies have evaluated cancer treatments for patients with HIV due to exclusions from most clinical trials. As HIV is an immune-based disease, concerns have arisen surrounding the safety and efficacy of using anti-neoplastic immunotherapies for HIV-positive patients. Considering that millions of people worldwide are HIV positive, research in this area can have real-world consequences.
Dr. Gobert and his colleagues analyzed data from CANCERVIH, a French national database of patients with cancer and HIV. Since May 2014, nivolumab has been administered to 20 patients. Nineteen had metastatic non–small-cell lung cancer and 1 had metastatic melanoma. At diagnosis, the median CD4 count was 338.5 per cubic millimeter. Seventeen patients had undetectable viral load, two had fewer than 40 copies per millimeter, and one patient’s viral load was unknown. Dr. Gobert described the population as “demographically homogenous,” with “most patients being males around 60 years old.”
Analysis showed that nivolumab had little impact on CD4 count or viral load. One patient had a decreased CD4 count and an increased viral load, but this occurred during an interruption to antiretroviral therapy, which clouds potential associations with nivolumab. No immune-related adverse events or deaths due to drug toxicity occurred. Efficacy was assessed in 17 patients: Four (24%) showed a partial response, 2 (12%) had stable disease, and 11 (64%) had disease progression.
“Based on these preliminary data, treatment with anti-PD-1 ... seems to be feasible in people with HIV,” Dr. Gobert reported. He added that “antiretroviral therapy should not be interrupted.”
In a comment for ESMO, John Haanen, PhD, of the Netherlands Cancer Institute, Amsterdam, said that the results “confirm those of other, smaller cohorts in showing that while on antiretroviral therapy, cancer patients living with HIV can safely receive anti-PD-1 treatment. The efficacy data also suggests that the overall response rate of HIV-positive patients seems to be similar to that of other cancer patients. These promising results need to be confirmed in larger studies – ideally, in a prospective clinical trial.”
Principal investigator Jean-Philippe Spano, MD, PhD, disclosed relationships with Gilead, Roche, BMS, and others.
SOURCE: Gobert et al. ESMO 2018, Abstract 1213P_PR.
MUNICH – Patients with HIV who are treated with nivolumab, a programmed death-1 (PD-1) inhibitor, appear to have similar safety and efficacy outcomes compared with HIV-negative patients treated with the same agent, investigators found.
The retrospective study also showed that viral load and CD4 status were largely unchanged by immunotherapy, lead author Aurélien Gobert, MD, of Groupe Hospitalier Pitié Salpêtrière, Paris, reported at the European Society for Medical Oncology Congress.
HIV increases risks of certain cancer types, Dr. Gobert said in a press release. “These patients are at higher risk for a number of cancers: AIDS-defining forms, the diagnosis of which results in the categorization of a person as suffering from AIDS, but also various other types that they are two to three times more likely to develop than in the general population, such as anal, skin, head and neck, and lung cancer,” he said.
Despite the increased risks, few studies have evaluated cancer treatments for patients with HIV due to exclusions from most clinical trials. As HIV is an immune-based disease, concerns have arisen surrounding the safety and efficacy of using anti-neoplastic immunotherapies for HIV-positive patients. Considering that millions of people worldwide are HIV positive, research in this area can have real-world consequences.
Dr. Gobert and his colleagues analyzed data from CANCERVIH, a French national database of patients with cancer and HIV. Since May 2014, nivolumab has been administered to 20 patients. Nineteen had metastatic non–small-cell lung cancer and 1 had metastatic melanoma. At diagnosis, the median CD4 count was 338.5 per cubic millimeter. Seventeen patients had undetectable viral load, two had fewer than 40 copies per millimeter, and one patient’s viral load was unknown. Dr. Gobert described the population as “demographically homogenous,” with “most patients being males around 60 years old.”
Analysis showed that nivolumab had little impact on CD4 count or viral load. One patient had a decreased CD4 count and an increased viral load, but this occurred during an interruption to antiretroviral therapy, which clouds potential associations with nivolumab. No immune-related adverse events or deaths due to drug toxicity occurred. Efficacy was assessed in 17 patients: Four (24%) showed a partial response, 2 (12%) had stable disease, and 11 (64%) had disease progression.
“Based on these preliminary data, treatment with anti-PD-1 ... seems to be feasible in people with HIV,” Dr. Gobert reported. He added that “antiretroviral therapy should not be interrupted.”
In a comment for ESMO, John Haanen, PhD, of the Netherlands Cancer Institute, Amsterdam, said that the results “confirm those of other, smaller cohorts in showing that while on antiretroviral therapy, cancer patients living with HIV can safely receive anti-PD-1 treatment. The efficacy data also suggests that the overall response rate of HIV-positive patients seems to be similar to that of other cancer patients. These promising results need to be confirmed in larger studies – ideally, in a prospective clinical trial.”
Principal investigator Jean-Philippe Spano, MD, PhD, disclosed relationships with Gilead, Roche, BMS, and others.
SOURCE: Gobert et al. ESMO 2018, Abstract 1213P_PR.
MUNICH – Patients with HIV who are treated with nivolumab, a programmed death-1 (PD-1) inhibitor, appear to have similar safety and efficacy outcomes compared with HIV-negative patients treated with the same agent, investigators found.
The retrospective study also showed that viral load and CD4 status were largely unchanged by immunotherapy, lead author Aurélien Gobert, MD, of Groupe Hospitalier Pitié Salpêtrière, Paris, reported at the European Society for Medical Oncology Congress.
HIV increases risks of certain cancer types, Dr. Gobert said in a press release. “These patients are at higher risk for a number of cancers: AIDS-defining forms, the diagnosis of which results in the categorization of a person as suffering from AIDS, but also various other types that they are two to three times more likely to develop than in the general population, such as anal, skin, head and neck, and lung cancer,” he said.
Despite the increased risks, few studies have evaluated cancer treatments for patients with HIV due to exclusions from most clinical trials. As HIV is an immune-based disease, concerns have arisen surrounding the safety and efficacy of using anti-neoplastic immunotherapies for HIV-positive patients. Considering that millions of people worldwide are HIV positive, research in this area can have real-world consequences.
Dr. Gobert and his colleagues analyzed data from CANCERVIH, a French national database of patients with cancer and HIV. Since May 2014, nivolumab has been administered to 20 patients. Nineteen had metastatic non–small-cell lung cancer and 1 had metastatic melanoma. At diagnosis, the median CD4 count was 338.5 per cubic millimeter. Seventeen patients had undetectable viral load, two had fewer than 40 copies per millimeter, and one patient’s viral load was unknown. Dr. Gobert described the population as “demographically homogenous,” with “most patients being males around 60 years old.”
Analysis showed that nivolumab had little impact on CD4 count or viral load. One patient had a decreased CD4 count and an increased viral load, but this occurred during an interruption to antiretroviral therapy, which clouds potential associations with nivolumab. No immune-related adverse events or deaths due to drug toxicity occurred. Efficacy was assessed in 17 patients: Four (24%) showed a partial response, 2 (12%) had stable disease, and 11 (64%) had disease progression.
“Based on these preliminary data, treatment with anti-PD-1 ... seems to be feasible in people with HIV,” Dr. Gobert reported. He added that “antiretroviral therapy should not be interrupted.”
In a comment for ESMO, John Haanen, PhD, of the Netherlands Cancer Institute, Amsterdam, said that the results “confirm those of other, smaller cohorts in showing that while on antiretroviral therapy, cancer patients living with HIV can safely receive anti-PD-1 treatment. The efficacy data also suggests that the overall response rate of HIV-positive patients seems to be similar to that of other cancer patients. These promising results need to be confirmed in larger studies – ideally, in a prospective clinical trial.”
Principal investigator Jean-Philippe Spano, MD, PhD, disclosed relationships with Gilead, Roche, BMS, and others.
SOURCE: Gobert et al. ESMO 2018, Abstract 1213P_PR.
REPORTING FROM ESMO 2018
Key clinical point: Patients with HIV who are treated with nivolumab, a programmed death-1 (PD-1) inhibitor, appear to have similar safety and efficacy outcomes compared with HIV-negative patients treated with the same agent.
Major finding: No immune-related adverse events or deaths due to drug toxicity occurred.
Study details: A retrospective analysis of 20 patients with HIV who received nivolumab immunotherapy.
Disclosures: Jean-Philippe Spano, principal investigator, disclosed relationships with Gilead, Roche, BMS, and others.
Source: Gobert et al. ESMO 2018, Abstract 1213P_PR.
PCI safely improves iPFS and OS in advanced NSCLC
TORONTO – Prophylactic cranial irradiation, which is standard-of-care practice in patients with small cell lung cancer, also appears to improve intracranial progression-free survival (iPFS) and overall survival in patients with stage IV non–small cell lung cancer (NSCLC), according to findings from a randomized study.
The cumulative incidence of brain metastases at 2 years was 22% in 41 patients who received prophylactic cranial irradiation (PCI), compared with 52% in 43 patients who received standard care with first- and second-generation tyrosine kinase inhibitors (TKIs) without PCI, Oscar Arrieta, MD, reported at the World Conference on Lung Cancer.
PCI was associated with lower odds of progression to the CNS (odds ratio, 0.16), Dr. Arrieta, of the National Cancer Institute of Mexico, Mexico City, said at the meeting sponsored by the International Association for the Study of Lung Cancer. Further, the relative risk for iPFS in patients with an epithelial growth factor receptor (EGFR) or anaplastic lymphoma kinase mutation (ALK), who comprised 70% of patients in both groups, was 0.29 with PCI.
Median overall survival in the groups was 42.8 months versus 25.9 months (HR, 0.48).
The burden of brain metastases can impact the quality and length of survival in patients with NSCLC, and because of an aging population and advances in detection and treatment of primary cancers, patients are living longer and thus are more likely to experience brain metastases, Dr. Arrieta said, noting that this is particularly true for patients at high risk, such as those with elevated carcinoembryonic antigen levels.
Although PCI is standard in small cell lung cancer, its role in NSCLC remains controversial because of concerns about neurologic morbidity and lack of overall survival benefit, he explained.
“The objective of this study was to determine if the use of PCI reduced the development of brain metastases and improved the survival in this population without impairing quality of life,” he said.
Study participants were patients with confirmed stage IV NSCLC and adenocarcinoma histology at high risk for developing brain metastasis. PCI in the treatment group was delivered at 25 Gy/10 fractions.
The findings suggest that in NSCLC with a high risk of developing brain metastases who are treated with a first- or second-generation TKI – particularly those with EGFR and ALK mutations – PCI increases iPFS, Dr. Arrieta said.
“The findings can be extrapolated to those treated with third-generation TKIs, which have higher CNS penetration,” he said, noting, however, that access to third-generation TKIs is limited in most developing countries and cost barriers are high.
Of note, the relatively low dose of PCI used in this study was not associated with significant differences in Mini-Mental State Examination or quality of life scores in the short-term. Long-term assessments are needed, he said, concluding that, while additional study is needed to confirm the findings, the results, including the overall survival benefit seen with PCI, “highlight the benefits of this approach, particularly among patients with a high risk of developing brain metastases.”
Invited discussant Nasser Hanna, MD, the Tom and Julie Wood Family Foundation Professor of Lung Cancer Clinical Research at Indiana University, Indianapolis, said the findings of this “interesting and important study,” are intriguing, but agreed that additional study is needed.
“The study is far too small ... to definitively make this conclusion [that PCI improves iPFS in this population]; I would not recommend PCI without confirmatory data from larger, randomized trials,” he said.
Dr. Arrieta reported advisory roles or provision of expert testimony for Eli Lilly, AstraZeneca, Boehringer Ingelheim, Roche, Merck, Takeda, and Bristol-Myers Squibb, and receipt of honoraria and/or research funding from AstraZeneca, Boehringer Ingelheim, Merck, Roche, and Bristol-Myers Squibb. Dr. Nasser reported receiving research grants from Bristol-Myers Squibb, AstraZeneca, Genentech, and Merck.
SOURCE: Arrieta O et al. WCLC 2018, Abstract MA08.02.
TORONTO – Prophylactic cranial irradiation, which is standard-of-care practice in patients with small cell lung cancer, also appears to improve intracranial progression-free survival (iPFS) and overall survival in patients with stage IV non–small cell lung cancer (NSCLC), according to findings from a randomized study.
The cumulative incidence of brain metastases at 2 years was 22% in 41 patients who received prophylactic cranial irradiation (PCI), compared with 52% in 43 patients who received standard care with first- and second-generation tyrosine kinase inhibitors (TKIs) without PCI, Oscar Arrieta, MD, reported at the World Conference on Lung Cancer.
PCI was associated with lower odds of progression to the CNS (odds ratio, 0.16), Dr. Arrieta, of the National Cancer Institute of Mexico, Mexico City, said at the meeting sponsored by the International Association for the Study of Lung Cancer. Further, the relative risk for iPFS in patients with an epithelial growth factor receptor (EGFR) or anaplastic lymphoma kinase mutation (ALK), who comprised 70% of patients in both groups, was 0.29 with PCI.
Median overall survival in the groups was 42.8 months versus 25.9 months (HR, 0.48).
The burden of brain metastases can impact the quality and length of survival in patients with NSCLC, and because of an aging population and advances in detection and treatment of primary cancers, patients are living longer and thus are more likely to experience brain metastases, Dr. Arrieta said, noting that this is particularly true for patients at high risk, such as those with elevated carcinoembryonic antigen levels.
Although PCI is standard in small cell lung cancer, its role in NSCLC remains controversial because of concerns about neurologic morbidity and lack of overall survival benefit, he explained.
“The objective of this study was to determine if the use of PCI reduced the development of brain metastases and improved the survival in this population without impairing quality of life,” he said.
Study participants were patients with confirmed stage IV NSCLC and adenocarcinoma histology at high risk for developing brain metastasis. PCI in the treatment group was delivered at 25 Gy/10 fractions.
The findings suggest that in NSCLC with a high risk of developing brain metastases who are treated with a first- or second-generation TKI – particularly those with EGFR and ALK mutations – PCI increases iPFS, Dr. Arrieta said.
“The findings can be extrapolated to those treated with third-generation TKIs, which have higher CNS penetration,” he said, noting, however, that access to third-generation TKIs is limited in most developing countries and cost barriers are high.
Of note, the relatively low dose of PCI used in this study was not associated with significant differences in Mini-Mental State Examination or quality of life scores in the short-term. Long-term assessments are needed, he said, concluding that, while additional study is needed to confirm the findings, the results, including the overall survival benefit seen with PCI, “highlight the benefits of this approach, particularly among patients with a high risk of developing brain metastases.”
Invited discussant Nasser Hanna, MD, the Tom and Julie Wood Family Foundation Professor of Lung Cancer Clinical Research at Indiana University, Indianapolis, said the findings of this “interesting and important study,” are intriguing, but agreed that additional study is needed.
“The study is far too small ... to definitively make this conclusion [that PCI improves iPFS in this population]; I would not recommend PCI without confirmatory data from larger, randomized trials,” he said.
Dr. Arrieta reported advisory roles or provision of expert testimony for Eli Lilly, AstraZeneca, Boehringer Ingelheim, Roche, Merck, Takeda, and Bristol-Myers Squibb, and receipt of honoraria and/or research funding from AstraZeneca, Boehringer Ingelheim, Merck, Roche, and Bristol-Myers Squibb. Dr. Nasser reported receiving research grants from Bristol-Myers Squibb, AstraZeneca, Genentech, and Merck.
SOURCE: Arrieta O et al. WCLC 2018, Abstract MA08.02.
TORONTO – Prophylactic cranial irradiation, which is standard-of-care practice in patients with small cell lung cancer, also appears to improve intracranial progression-free survival (iPFS) and overall survival in patients with stage IV non–small cell lung cancer (NSCLC), according to findings from a randomized study.
The cumulative incidence of brain metastases at 2 years was 22% in 41 patients who received prophylactic cranial irradiation (PCI), compared with 52% in 43 patients who received standard care with first- and second-generation tyrosine kinase inhibitors (TKIs) without PCI, Oscar Arrieta, MD, reported at the World Conference on Lung Cancer.
PCI was associated with lower odds of progression to the CNS (odds ratio, 0.16), Dr. Arrieta, of the National Cancer Institute of Mexico, Mexico City, said at the meeting sponsored by the International Association for the Study of Lung Cancer. Further, the relative risk for iPFS in patients with an epithelial growth factor receptor (EGFR) or anaplastic lymphoma kinase mutation (ALK), who comprised 70% of patients in both groups, was 0.29 with PCI.
Median overall survival in the groups was 42.8 months versus 25.9 months (HR, 0.48).
The burden of brain metastases can impact the quality and length of survival in patients with NSCLC, and because of an aging population and advances in detection and treatment of primary cancers, patients are living longer and thus are more likely to experience brain metastases, Dr. Arrieta said, noting that this is particularly true for patients at high risk, such as those with elevated carcinoembryonic antigen levels.
Although PCI is standard in small cell lung cancer, its role in NSCLC remains controversial because of concerns about neurologic morbidity and lack of overall survival benefit, he explained.
“The objective of this study was to determine if the use of PCI reduced the development of brain metastases and improved the survival in this population without impairing quality of life,” he said.
Study participants were patients with confirmed stage IV NSCLC and adenocarcinoma histology at high risk for developing brain metastasis. PCI in the treatment group was delivered at 25 Gy/10 fractions.
The findings suggest that in NSCLC with a high risk of developing brain metastases who are treated with a first- or second-generation TKI – particularly those with EGFR and ALK mutations – PCI increases iPFS, Dr. Arrieta said.
“The findings can be extrapolated to those treated with third-generation TKIs, which have higher CNS penetration,” he said, noting, however, that access to third-generation TKIs is limited in most developing countries and cost barriers are high.
Of note, the relatively low dose of PCI used in this study was not associated with significant differences in Mini-Mental State Examination or quality of life scores in the short-term. Long-term assessments are needed, he said, concluding that, while additional study is needed to confirm the findings, the results, including the overall survival benefit seen with PCI, “highlight the benefits of this approach, particularly among patients with a high risk of developing brain metastases.”
Invited discussant Nasser Hanna, MD, the Tom and Julie Wood Family Foundation Professor of Lung Cancer Clinical Research at Indiana University, Indianapolis, said the findings of this “interesting and important study,” are intriguing, but agreed that additional study is needed.
“The study is far too small ... to definitively make this conclusion [that PCI improves iPFS in this population]; I would not recommend PCI without confirmatory data from larger, randomized trials,” he said.
Dr. Arrieta reported advisory roles or provision of expert testimony for Eli Lilly, AstraZeneca, Boehringer Ingelheim, Roche, Merck, Takeda, and Bristol-Myers Squibb, and receipt of honoraria and/or research funding from AstraZeneca, Boehringer Ingelheim, Merck, Roche, and Bristol-Myers Squibb. Dr. Nasser reported receiving research grants from Bristol-Myers Squibb, AstraZeneca, Genentech, and Merck.
SOURCE: Arrieta O et al. WCLC 2018, Abstract MA08.02.
REPORTING FROM WCLC 2018
Key clinical point: Prophylactic cranial irradiation improves intracranial progression-free survival and overall survival in non–small cell lung cancer.
Major finding: Prophylactic cranial irradiation reduced the risk of CNS progression (odds ratio, 0.16).
Study details: A randomized study of 84 patients with non–small cell lung cancer.
Disclosures: Dr. Arrieta reported advisory roles or provision of expert testimony for Eli Lilly, AstraZeneca, Boehringer Ingelheim, Roche, Merck, Takeda, and Bristol-Myers Squibb, and receipt of honoraria and/or research funding from AstraZeneca, Boehringer Ingelheim, Merck, Roche, and Bristol-Myers Squibb. Dr. Nasser reported receiving research grants from Bristol-Myers Squibb, AstraZeneca, Genentech, and Merck.
Source: Arrieta O et al. WCLC 2018, Abstract MA08.02.
Neoadjuvant TKI for advanced NSCLC falls short
MUNICH – Neoadjuvant erlotinib for patients with stage IIIA (N2) non–small cell lung cancer (NSCLC) increased progression-free survival (PFS), compared with gemcitabine and cisplatin, according to results from the recent CTONG 1103 trial.
Despite beating chemotherapy, erlotinib, an epithelial growth factor receptor (EGFR)–mutant tyrosine kinase inhibitor (TKI), fell short of benchmarks set by adjuvant therapy, so it is unlikely that neoadjuvant erlotinib will see clinical use anytime soon.
Lead author Wen-Zhao Zhong, MD, PhD, of Guangdong Lung Cancer Institute and Guangdong General Hospital in Guangzhou, China, presenting at the European Society for Medical Oncology Congress, said that the recent findings support further investigation into biomarker-guided neoadjuvant therapy for stage IIIA (N2) NSCLC, as identifying patient subgroups could potentially improve outcomes.
Principal investigator Yi-Long Wu, MD, described the impetus for CTONG 1103 in an interview. “Recently, the CTONG 1104 trial showed for the first time that adjuvant EGFR-TKI gefitinib could improve disease-free survival ... compared to adjuvant chemotherapy ... in N1N2-resected NSCLC. This raises the possibility that EGFR-TKIs may play a beneficial role in the neoadjuvant setting for this subgroup,” he said.
CTONG 1103 is an ongoing, phase 2, open-label trial. Out of 386 patients screened, 72 were enrolled based on treatment naivety and EGFR mutation positivity (exon 19 or 21). Following randomization, patients received either erlotinib 150 mg daily for 42 days or gemcitabine 1,250 mg/m2 (days 1 and 8) and cisplatin 75 mg/m2 (day 1) every 3 weeks for two cycles. After surgery, patients in the erlotinib group continued therapy for 1 year, while patients in the chemotherapy cohort received two more cycles of treatment.
The primary endpoint was objective response rate, and secondary endpoints included PFS, pathological lymph node downstaging, overall survival, safety measures, and complete pathological response. The investigators also highlighted major pathological response (less than 10% viable cancer cells after preoperative therapy).
The results showed that about half of the patients receiving erlotinib had an objective response (54.1%), compared with approximately one-third in the chemotherapy group (34.3%); however, this difference was not statistically significant (P = .092). Erlotinib also provided a median PFS nearly twice that of chemotherapy (21.5 months vs. 11.9 months; P = .003) and more frequent lymph node downstaging (10.8% vs. 2.9%), but no patients achieved complete pathological response. The number of patients achieving major pathological response with erlotinib was limited but still more than chemotherapy (10.7% vs. 0%). The investigators are awaiting an overall survival rate.
Erlotinib showed similar adverse events to previous trials, most commonly, rash, diarrhea, cough, and oral ulcers, compared with chemotherapy, which was associated with GI issues, hematologic disturbances, and fatigue.
In response to these findings, Suresh S. Ramalingam, MD, invited discussant and deputy director of the Winship Cancer Institute of Emory University, Atlanta, first discussed relevant efficacy measures. “The best predictor of long-term outcomes in these patients is nodal downstaging,” Dr. Ramalingam said. In previous studies, “patients who had clearance of the nodes had the best outcomes, and that continues to be an important prognostic marker.”
While major pathological response is valuable, and previous studies have revealed prognostic value, evidence is too limited to suggest that this is equivalent with cure, and it should not be considered as significant as complete pathological response, he said.
Considering that “only 11% of the patients” treated with erlotinib had nodal downstaging and/or major pathological response, and none achieved complete pathological response, Dr. Ramalingam suggested that the results were modest at best.
“While erlotinib seems to be doing better than chemo, I feel that the chemo group here is underperforming, compared to historical controls,” Dr. Ramalingam said, noting higher benchmark objective response rates (61% vs. 34%) and complete pathological response rates (4% vs. 0%).
Instead of focusing on neoadjuvant studies, Dr. Ramalingam suggested that ongoing adjuvant trials (ALCHEMIST and ADAURA) hold more promise and are more likely to serve as inroads for TKIs like erlotinib.
“Adjuvant care has withstood the test of time,” Dr. Ramalingam said. “Neoadjuvant EGFR-TKI in N2 disease I don’t think is ready for center stage.”
CTONG 1103 was sponsored by CTONG and Roche. The authors reported financial affiliations with AstraZeneca, Pfizer, Roche, and others. Dr. Ramalingam reported compensation from Amgen, Bristol-Myers Squibb, Loxo Oncology, and others.
MUNICH – Neoadjuvant erlotinib for patients with stage IIIA (N2) non–small cell lung cancer (NSCLC) increased progression-free survival (PFS), compared with gemcitabine and cisplatin, according to results from the recent CTONG 1103 trial.
Despite beating chemotherapy, erlotinib, an epithelial growth factor receptor (EGFR)–mutant tyrosine kinase inhibitor (TKI), fell short of benchmarks set by adjuvant therapy, so it is unlikely that neoadjuvant erlotinib will see clinical use anytime soon.
Lead author Wen-Zhao Zhong, MD, PhD, of Guangdong Lung Cancer Institute and Guangdong General Hospital in Guangzhou, China, presenting at the European Society for Medical Oncology Congress, said that the recent findings support further investigation into biomarker-guided neoadjuvant therapy for stage IIIA (N2) NSCLC, as identifying patient subgroups could potentially improve outcomes.
Principal investigator Yi-Long Wu, MD, described the impetus for CTONG 1103 in an interview. “Recently, the CTONG 1104 trial showed for the first time that adjuvant EGFR-TKI gefitinib could improve disease-free survival ... compared to adjuvant chemotherapy ... in N1N2-resected NSCLC. This raises the possibility that EGFR-TKIs may play a beneficial role in the neoadjuvant setting for this subgroup,” he said.
CTONG 1103 is an ongoing, phase 2, open-label trial. Out of 386 patients screened, 72 were enrolled based on treatment naivety and EGFR mutation positivity (exon 19 or 21). Following randomization, patients received either erlotinib 150 mg daily for 42 days or gemcitabine 1,250 mg/m2 (days 1 and 8) and cisplatin 75 mg/m2 (day 1) every 3 weeks for two cycles. After surgery, patients in the erlotinib group continued therapy for 1 year, while patients in the chemotherapy cohort received two more cycles of treatment.
The primary endpoint was objective response rate, and secondary endpoints included PFS, pathological lymph node downstaging, overall survival, safety measures, and complete pathological response. The investigators also highlighted major pathological response (less than 10% viable cancer cells after preoperative therapy).
The results showed that about half of the patients receiving erlotinib had an objective response (54.1%), compared with approximately one-third in the chemotherapy group (34.3%); however, this difference was not statistically significant (P = .092). Erlotinib also provided a median PFS nearly twice that of chemotherapy (21.5 months vs. 11.9 months; P = .003) and more frequent lymph node downstaging (10.8% vs. 2.9%), but no patients achieved complete pathological response. The number of patients achieving major pathological response with erlotinib was limited but still more than chemotherapy (10.7% vs. 0%). The investigators are awaiting an overall survival rate.
Erlotinib showed similar adverse events to previous trials, most commonly, rash, diarrhea, cough, and oral ulcers, compared with chemotherapy, which was associated with GI issues, hematologic disturbances, and fatigue.
In response to these findings, Suresh S. Ramalingam, MD, invited discussant and deputy director of the Winship Cancer Institute of Emory University, Atlanta, first discussed relevant efficacy measures. “The best predictor of long-term outcomes in these patients is nodal downstaging,” Dr. Ramalingam said. In previous studies, “patients who had clearance of the nodes had the best outcomes, and that continues to be an important prognostic marker.”
While major pathological response is valuable, and previous studies have revealed prognostic value, evidence is too limited to suggest that this is equivalent with cure, and it should not be considered as significant as complete pathological response, he said.
Considering that “only 11% of the patients” treated with erlotinib had nodal downstaging and/or major pathological response, and none achieved complete pathological response, Dr. Ramalingam suggested that the results were modest at best.
“While erlotinib seems to be doing better than chemo, I feel that the chemo group here is underperforming, compared to historical controls,” Dr. Ramalingam said, noting higher benchmark objective response rates (61% vs. 34%) and complete pathological response rates (4% vs. 0%).
Instead of focusing on neoadjuvant studies, Dr. Ramalingam suggested that ongoing adjuvant trials (ALCHEMIST and ADAURA) hold more promise and are more likely to serve as inroads for TKIs like erlotinib.
“Adjuvant care has withstood the test of time,” Dr. Ramalingam said. “Neoadjuvant EGFR-TKI in N2 disease I don’t think is ready for center stage.”
CTONG 1103 was sponsored by CTONG and Roche. The authors reported financial affiliations with AstraZeneca, Pfizer, Roche, and others. Dr. Ramalingam reported compensation from Amgen, Bristol-Myers Squibb, Loxo Oncology, and others.
MUNICH – Neoadjuvant erlotinib for patients with stage IIIA (N2) non–small cell lung cancer (NSCLC) increased progression-free survival (PFS), compared with gemcitabine and cisplatin, according to results from the recent CTONG 1103 trial.
Despite beating chemotherapy, erlotinib, an epithelial growth factor receptor (EGFR)–mutant tyrosine kinase inhibitor (TKI), fell short of benchmarks set by adjuvant therapy, so it is unlikely that neoadjuvant erlotinib will see clinical use anytime soon.
Lead author Wen-Zhao Zhong, MD, PhD, of Guangdong Lung Cancer Institute and Guangdong General Hospital in Guangzhou, China, presenting at the European Society for Medical Oncology Congress, said that the recent findings support further investigation into biomarker-guided neoadjuvant therapy for stage IIIA (N2) NSCLC, as identifying patient subgroups could potentially improve outcomes.
Principal investigator Yi-Long Wu, MD, described the impetus for CTONG 1103 in an interview. “Recently, the CTONG 1104 trial showed for the first time that adjuvant EGFR-TKI gefitinib could improve disease-free survival ... compared to adjuvant chemotherapy ... in N1N2-resected NSCLC. This raises the possibility that EGFR-TKIs may play a beneficial role in the neoadjuvant setting for this subgroup,” he said.
CTONG 1103 is an ongoing, phase 2, open-label trial. Out of 386 patients screened, 72 were enrolled based on treatment naivety and EGFR mutation positivity (exon 19 or 21). Following randomization, patients received either erlotinib 150 mg daily for 42 days or gemcitabine 1,250 mg/m2 (days 1 and 8) and cisplatin 75 mg/m2 (day 1) every 3 weeks for two cycles. After surgery, patients in the erlotinib group continued therapy for 1 year, while patients in the chemotherapy cohort received two more cycles of treatment.
The primary endpoint was objective response rate, and secondary endpoints included PFS, pathological lymph node downstaging, overall survival, safety measures, and complete pathological response. The investigators also highlighted major pathological response (less than 10% viable cancer cells after preoperative therapy).
The results showed that about half of the patients receiving erlotinib had an objective response (54.1%), compared with approximately one-third in the chemotherapy group (34.3%); however, this difference was not statistically significant (P = .092). Erlotinib also provided a median PFS nearly twice that of chemotherapy (21.5 months vs. 11.9 months; P = .003) and more frequent lymph node downstaging (10.8% vs. 2.9%), but no patients achieved complete pathological response. The number of patients achieving major pathological response with erlotinib was limited but still more than chemotherapy (10.7% vs. 0%). The investigators are awaiting an overall survival rate.
Erlotinib showed similar adverse events to previous trials, most commonly, rash, diarrhea, cough, and oral ulcers, compared with chemotherapy, which was associated with GI issues, hematologic disturbances, and fatigue.
In response to these findings, Suresh S. Ramalingam, MD, invited discussant and deputy director of the Winship Cancer Institute of Emory University, Atlanta, first discussed relevant efficacy measures. “The best predictor of long-term outcomes in these patients is nodal downstaging,” Dr. Ramalingam said. In previous studies, “patients who had clearance of the nodes had the best outcomes, and that continues to be an important prognostic marker.”
While major pathological response is valuable, and previous studies have revealed prognostic value, evidence is too limited to suggest that this is equivalent with cure, and it should not be considered as significant as complete pathological response, he said.
Considering that “only 11% of the patients” treated with erlotinib had nodal downstaging and/or major pathological response, and none achieved complete pathological response, Dr. Ramalingam suggested that the results were modest at best.
“While erlotinib seems to be doing better than chemo, I feel that the chemo group here is underperforming, compared to historical controls,” Dr. Ramalingam said, noting higher benchmark objective response rates (61% vs. 34%) and complete pathological response rates (4% vs. 0%).
Instead of focusing on neoadjuvant studies, Dr. Ramalingam suggested that ongoing adjuvant trials (ALCHEMIST and ADAURA) hold more promise and are more likely to serve as inroads for TKIs like erlotinib.
“Adjuvant care has withstood the test of time,” Dr. Ramalingam said. “Neoadjuvant EGFR-TKI in N2 disease I don’t think is ready for center stage.”
CTONG 1103 was sponsored by CTONG and Roche. The authors reported financial affiliations with AstraZeneca, Pfizer, Roche, and others. Dr. Ramalingam reported compensation from Amgen, Bristol-Myers Squibb, Loxo Oncology, and others.
REPORTING FROM ESMO 2018
Key clinical point: Neoadjuvant erlotinib for patients with stage IIIA (N2) non–small cell lung cancer may increase progression-free survival, compared with gemcitabine and cisplatin, but clinical benefits fall short of current standards.
Major finding: Median progression-free survival for erlotinib was 21.5 months, compared with 11.9 months for gemcitabine and cisplatin (P = .003).
Study details: CTONG 1103 is an ongoing, phase 2, open-label study involving 72 patients with stage IIIA (N2) epithelial growth factor receptor–mutant non–small cell lung cancer
Disclosures: The study was sponsored by CTONG and Roche. The authors reported financial affiliations with AstraZeneca, Pfizer, Roche, and others. Dr. Ramalingam reported affiliations with Amgen, Bristol-Myers Squibb, Loxo Oncology, and others.
DETERRED trial: Concurrent atezolizumab, CRT shows promise in LA-NSCLC
TORONTO – (LA-NSCLC) in the phase 2 DETERRED trial.
In part 1 of the single-institution study, 10 patients underwent chemoradiation therapy (CRT) with low-dose carboplatin/paclitaxel followed by high-dose consolidation chemotherapy plus atezolizumab and atezolizumab maintenance for 1 year. Six patients in this group (60%) experienced grade 3 or higher adverse events (AEs). In part 2 of the study, 30 patients received concurrent atezolizumab and CRT followed by the same consolidation and maintenance used in part 1, and 17 (57%) experienced grade 3 or higher AEs, Steven H. Lin, MD, reported at the World Conference on Lung Cancer.
Grade 3 or higher AEs were associated with atezolizumab in 30% and 23% of patients in part 1 and part 2, respectively. In part 1 these included dyspnea, arthralgia, and a grade 5 tracheoesophageal fistula, and in part 2 included diarrhea, pneumonitis, nephritis, fatigue, respiratory failure, and heart failure in one patient each, and fatigue in three patients.
Grade 2 radiation pneumonitis was seen in two patients in each group, Dr. Lin of the University of Texas MD Anderson Cancer Center, Houston, said at the meeting, which was sponsored by the International Association for the Study of Lung Cancer.
Withdrawals caused by toxicity occurred in three and four patients in part 1 and 2, respectively.
Four patients in part 1 progressed with disease during atezolizumab maintenance and five have died from either tracheoesophageal fistula or grade 5 toxicity. In part 2, six have progressed and five have died, most caused by cancer progression, he said.
Preliminary survival data show a median progression-free survival of 20.1 months in part 1, whereas progression-free survival was not reached in part 2. Median overall survival at 1 year was 60% versus 77% in parts 1 and 2, respectively.
Consolidation immunotherapy with durvalumab after CRT has been the standard of care for LA-NSCLC as established by the phase 3 PACIFIC trial, but evidence from that trial also suggested timing of the start of immunotherapy may be important.
“If patients were randomized less than 14 days after starting durvalumab there was a trend, or suggestion, that there was potentially an improvement in progression-free survival, compared with patients who started durvalumab outside of this window,” Dr. Lin said, noting that this is also supported by some preclinical evidence showing that the effectiveness of immunotherapies may be enhanced when combined with concurrent CRT.
The DETERRED trial evaluated the safety and preliminary efficacy of this approach followed by consolidation full-dose carboplatin/paclitaxel with atezolizumab and maintenance atezolizumab for up to 1 year for LA-NSCLC.
Patients, who had a median age of 66.5 years, were enrolled between February 2016 and April 2018; 15% had stage II disease, 50% had stage IIIA, and 35% had stage IIIB. Most (58%) had adenocarcinoma.
In part 1, standard chemoradiation including low-dose carboplatin/paclitaxel was given for 6 weeks. After CRT completion, patients were given consolidated high-dose chemotherapy with carboplatin/paclitaxel and intravenous atezolizumab was started at that point at a dose of 1,200 mg every 3 weeks for up to 1 year from the first dose. Part 2 was initiated based on the safety data in part 1 showing no concerning toxicities. In part 2, atezolizumab was given concurrently with CRT followed by the same consolidated regimen and maintenance.
“So the take-home message from this study is that the concurrent immunotherapy with atezolizumab and chemoradiation therapy can be administered safely,” Dr. Lin said, adding that grade 3+ pneumonitis is low and not significantly increased with the addition of concurrent atezolizumab with CRT.
Early efficacy analyses also show promising results, but further follow-up is needed, he said.
Trials now being planned include a phase 3 trial comparing the DETERRED and PACIFIC regimens, a phase 1 study comparing durvalumab with radiation to replace chemotherapy in programmed death–ligand 1–high locoregionally advanced NSCLC, and a phase 1 study of nivolumab with radiation to replace chemotherapy in locoregionally advanced NSCLC, he noted.
The DETERRED trial was supported by Genentech. Dr. Lin has received research grants from STCube Pharmaceuticals, Hitachi Chemical Diagnostics, Genentech, New River Labs, and BeyondSpring Pharmaceuticals, and is an advisory board member for AstraZeneca and New River Labs.
SOURCE: Lin SH et al. WCLC 2018, Abstract OA01.06.
TORONTO – (LA-NSCLC) in the phase 2 DETERRED trial.
In part 1 of the single-institution study, 10 patients underwent chemoradiation therapy (CRT) with low-dose carboplatin/paclitaxel followed by high-dose consolidation chemotherapy plus atezolizumab and atezolizumab maintenance for 1 year. Six patients in this group (60%) experienced grade 3 or higher adverse events (AEs). In part 2 of the study, 30 patients received concurrent atezolizumab and CRT followed by the same consolidation and maintenance used in part 1, and 17 (57%) experienced grade 3 or higher AEs, Steven H. Lin, MD, reported at the World Conference on Lung Cancer.
Grade 3 or higher AEs were associated with atezolizumab in 30% and 23% of patients in part 1 and part 2, respectively. In part 1 these included dyspnea, arthralgia, and a grade 5 tracheoesophageal fistula, and in part 2 included diarrhea, pneumonitis, nephritis, fatigue, respiratory failure, and heart failure in one patient each, and fatigue in three patients.
Grade 2 radiation pneumonitis was seen in two patients in each group, Dr. Lin of the University of Texas MD Anderson Cancer Center, Houston, said at the meeting, which was sponsored by the International Association for the Study of Lung Cancer.
Withdrawals caused by toxicity occurred in three and four patients in part 1 and 2, respectively.
Four patients in part 1 progressed with disease during atezolizumab maintenance and five have died from either tracheoesophageal fistula or grade 5 toxicity. In part 2, six have progressed and five have died, most caused by cancer progression, he said.
Preliminary survival data show a median progression-free survival of 20.1 months in part 1, whereas progression-free survival was not reached in part 2. Median overall survival at 1 year was 60% versus 77% in parts 1 and 2, respectively.
Consolidation immunotherapy with durvalumab after CRT has been the standard of care for LA-NSCLC as established by the phase 3 PACIFIC trial, but evidence from that trial also suggested timing of the start of immunotherapy may be important.
“If patients were randomized less than 14 days after starting durvalumab there was a trend, or suggestion, that there was potentially an improvement in progression-free survival, compared with patients who started durvalumab outside of this window,” Dr. Lin said, noting that this is also supported by some preclinical evidence showing that the effectiveness of immunotherapies may be enhanced when combined with concurrent CRT.
The DETERRED trial evaluated the safety and preliminary efficacy of this approach followed by consolidation full-dose carboplatin/paclitaxel with atezolizumab and maintenance atezolizumab for up to 1 year for LA-NSCLC.
Patients, who had a median age of 66.5 years, were enrolled between February 2016 and April 2018; 15% had stage II disease, 50% had stage IIIA, and 35% had stage IIIB. Most (58%) had adenocarcinoma.
In part 1, standard chemoradiation including low-dose carboplatin/paclitaxel was given for 6 weeks. After CRT completion, patients were given consolidated high-dose chemotherapy with carboplatin/paclitaxel and intravenous atezolizumab was started at that point at a dose of 1,200 mg every 3 weeks for up to 1 year from the first dose. Part 2 was initiated based on the safety data in part 1 showing no concerning toxicities. In part 2, atezolizumab was given concurrently with CRT followed by the same consolidated regimen and maintenance.
“So the take-home message from this study is that the concurrent immunotherapy with atezolizumab and chemoradiation therapy can be administered safely,” Dr. Lin said, adding that grade 3+ pneumonitis is low and not significantly increased with the addition of concurrent atezolizumab with CRT.
Early efficacy analyses also show promising results, but further follow-up is needed, he said.
Trials now being planned include a phase 3 trial comparing the DETERRED and PACIFIC regimens, a phase 1 study comparing durvalumab with radiation to replace chemotherapy in programmed death–ligand 1–high locoregionally advanced NSCLC, and a phase 1 study of nivolumab with radiation to replace chemotherapy in locoregionally advanced NSCLC, he noted.
The DETERRED trial was supported by Genentech. Dr. Lin has received research grants from STCube Pharmaceuticals, Hitachi Chemical Diagnostics, Genentech, New River Labs, and BeyondSpring Pharmaceuticals, and is an advisory board member for AstraZeneca and New River Labs.
SOURCE: Lin SH et al. WCLC 2018, Abstract OA01.06.
TORONTO – (LA-NSCLC) in the phase 2 DETERRED trial.
In part 1 of the single-institution study, 10 patients underwent chemoradiation therapy (CRT) with low-dose carboplatin/paclitaxel followed by high-dose consolidation chemotherapy plus atezolizumab and atezolizumab maintenance for 1 year. Six patients in this group (60%) experienced grade 3 or higher adverse events (AEs). In part 2 of the study, 30 patients received concurrent atezolizumab and CRT followed by the same consolidation and maintenance used in part 1, and 17 (57%) experienced grade 3 or higher AEs, Steven H. Lin, MD, reported at the World Conference on Lung Cancer.
Grade 3 or higher AEs were associated with atezolizumab in 30% and 23% of patients in part 1 and part 2, respectively. In part 1 these included dyspnea, arthralgia, and a grade 5 tracheoesophageal fistula, and in part 2 included diarrhea, pneumonitis, nephritis, fatigue, respiratory failure, and heart failure in one patient each, and fatigue in three patients.
Grade 2 radiation pneumonitis was seen in two patients in each group, Dr. Lin of the University of Texas MD Anderson Cancer Center, Houston, said at the meeting, which was sponsored by the International Association for the Study of Lung Cancer.
Withdrawals caused by toxicity occurred in three and four patients in part 1 and 2, respectively.
Four patients in part 1 progressed with disease during atezolizumab maintenance and five have died from either tracheoesophageal fistula or grade 5 toxicity. In part 2, six have progressed and five have died, most caused by cancer progression, he said.
Preliminary survival data show a median progression-free survival of 20.1 months in part 1, whereas progression-free survival was not reached in part 2. Median overall survival at 1 year was 60% versus 77% in parts 1 and 2, respectively.
Consolidation immunotherapy with durvalumab after CRT has been the standard of care for LA-NSCLC as established by the phase 3 PACIFIC trial, but evidence from that trial also suggested timing of the start of immunotherapy may be important.
“If patients were randomized less than 14 days after starting durvalumab there was a trend, or suggestion, that there was potentially an improvement in progression-free survival, compared with patients who started durvalumab outside of this window,” Dr. Lin said, noting that this is also supported by some preclinical evidence showing that the effectiveness of immunotherapies may be enhanced when combined with concurrent CRT.
The DETERRED trial evaluated the safety and preliminary efficacy of this approach followed by consolidation full-dose carboplatin/paclitaxel with atezolizumab and maintenance atezolizumab for up to 1 year for LA-NSCLC.
Patients, who had a median age of 66.5 years, were enrolled between February 2016 and April 2018; 15% had stage II disease, 50% had stage IIIA, and 35% had stage IIIB. Most (58%) had adenocarcinoma.
In part 1, standard chemoradiation including low-dose carboplatin/paclitaxel was given for 6 weeks. After CRT completion, patients were given consolidated high-dose chemotherapy with carboplatin/paclitaxel and intravenous atezolizumab was started at that point at a dose of 1,200 mg every 3 weeks for up to 1 year from the first dose. Part 2 was initiated based on the safety data in part 1 showing no concerning toxicities. In part 2, atezolizumab was given concurrently with CRT followed by the same consolidated regimen and maintenance.
“So the take-home message from this study is that the concurrent immunotherapy with atezolizumab and chemoradiation therapy can be administered safely,” Dr. Lin said, adding that grade 3+ pneumonitis is low and not significantly increased with the addition of concurrent atezolizumab with CRT.
Early efficacy analyses also show promising results, but further follow-up is needed, he said.
Trials now being planned include a phase 3 trial comparing the DETERRED and PACIFIC regimens, a phase 1 study comparing durvalumab with radiation to replace chemotherapy in programmed death–ligand 1–high locoregionally advanced NSCLC, and a phase 1 study of nivolumab with radiation to replace chemotherapy in locoregionally advanced NSCLC, he noted.
The DETERRED trial was supported by Genentech. Dr. Lin has received research grants from STCube Pharmaceuticals, Hitachi Chemical Diagnostics, Genentech, New River Labs, and BeyondSpring Pharmaceuticals, and is an advisory board member for AstraZeneca and New River Labs.
SOURCE: Lin SH et al. WCLC 2018, Abstract OA01.06.
REPORTING FROM WCLC 2018
Key clinical point: Concurrent atezolizumab and chemoradiation therapy is safe and shows promising efficacy in locally advanced non–small cell lung cancer.
Major finding: A total of 60% and 57% of part 1 and 2 patients, respectively, experienced grade 3 or higher adverse events.
Study details: The phase 2 DETERRED trial of 40 patients.
Disclosures: The DETERRED trial was supported by Genentech. Dr. Lin has received research grants from STCube Pharmaceuticals, Hitachi Chemical Diagnostics, Genentech, New River Labs, and BeyondSpring Pharmaceuticals, and is an advisory board member for AstraZeneca and New River Labs.
Source: Lin SH et al. WCLC 2018, Abstract OA01.06.
ALTA-1L: Brigatinib beats crizotinib for PFS in ALK-positive NSCLC
TORONTO – in the first interim results from the multicenter, open-label, phase 3 ALTA-1L trial.
In fact, the primary endpoint of the study – blinded independent review committee–assessed progression-free survival (PFS) – was met at this first analysis. The next-generation ALK inhibitor brigatinib reduced the chance of progression or death by 51% as compared with crizotinib – the current first-line standard of care in this population, D. Ross Camidge, MD, reported at the World Conference on Lung Cancer.
“At a median follow-up of 9-11 months, 26% [of 137 patients] on the brigatinib arm and 46% [of 138 patients] on the crizotinib arm had experienced a PFS event. Median PFS was not reached for the brigatinib arm and was 9.8 months for the crizotinib arm; the hazard ratio for progression or death was 0.49 and highly statistically significant,” said Dr. Camidge, director of thoracic oncology and the Joyce Zeff Chair in Lung Cancer Research at the University of Colorado Cancer Center, Aurora, at the meeting sponsored by the International Association for the Study of Lung Cancer.
The findings were published simultaneously in the New England Journal of Medicine.
The 12-month PFS rate estimate was 67% for brigatinib and 43% for crizotinib, and the investigator-assessed hazard ratio for PFS was 0.45. Overall survival data are immature, Dr. Camidge added, noting that the PFS hazard ratios for those with versus those without prior chemotherapy were 0.35 and 0.55, respectively.
“PFS consistently favored brigatinib across all other subgroups,” he said. “Although the confidence intervals overlap, the effect size appears greater among those with baseline [CNS] disease than among those without [HR, 0.2 vs. 0.72].”
Data suggest that CNS progression among those with CNS disease at baseline tends to be an earlier event than either extra-CNS progression in general or CNS progression in those without baseline CNS disease. “Consequently, this first interim analysis may be preferentially emphasizing drug efficacy differences within the subgroup in whom the earlier progression events are occurring,” Dr. Camidge said.
As for overall objective responses, the rates were numerically higher with brigatinib versus crizotinib (70% vs. 60%), but were not statistically different in the two groups. “However, the median duration of confirmed responses was not reached for brigatinib and [was] 11.1 months with crizotinib, with the 12-month [probability] of sustained response being 75% for brigatinib and 41% for crizotinib,” he added.
Further, among those with measurable CNS lesions, brigatinib demonstrated a significantly higher intracranial response rate of 78% versus 29% (odds ratio, 10.42), and when those with nonmeasurable CNS disease were included in the intracranial response assessment, the odds ratio was 13.
Median intracranial PFS among those with CNS involvement at baseline was not reached in the brigatinib arm versus 5.6 months with crizotinib, for a highly statistically significant hazard ratio of 0.27. “Which, given that the median PFS for crizotinib in the overall population ... was 9.8 months, again emphasizes how CNS events in this subgroup tend to occur early,” he noted.
Study subjects had stage IIIB/IV ALK-positive non–small cell lung cancer (NSCLC) based on local ALK testing, Eastern Cooperative Oncology Group performance status of 0-2, no more than one prior systemic therapy for locally advanced/metastatic NSCLC, and no prior ALK inhibitor therapy, Dr. Camidge said, noting that asymptomatic, untreated brain metastases were allowed and crossover to the brigatinib arm was permitted for those with blinded independent review committee–assessed progression on crizotinib.
Those randomized to the brigatinib arm had a median age of 58 years and received 180 mg daily with a 7-day lead-in at 90 mg. Those randomized to the crizotinib group had a median age of 60 years and received 250 mg twice daily. Prior chemotherapy for advanced disease was received by 26% and 27% and brain metastasis was present in 29% and 30% of patients in the arms, respectively.
The most common treatment-emergent adverse events of grade 3 or higher in the brigatinib patients were laboratory abnormalities such as creatinine phosphokinase, lipase, and amylase increases, and the most common in the crizotinib group were gastrointestinal effects, transaminitis, bradycardia, peripheral edema, and vision disturbances.
Discontinuations caused by adverse events occurred in 12% and 9% of the brigatinib and crizotinib patients, respectively, he said. No clinical cases of pancreatitis occurred in either arm, there was no difference in the incidence of any grade myalgia or musculoskeletal pain between arms, and there was no grade 3 or greater myalgia or musculoskeletal pain reported.
Early-onset interstitial lung disease, however, “appears to be a unique side effect of brigatinib,” Dr. Camidge noted.
Although interstitial lung disease/pneumonitis occurred in both groups, onset at days 3-8 after treatment initiation occurred only with brigatinib, he said, adding that “it only occurred in 3% of cases – half the rate seen in the postcrizotinib setting.”
The PROFILE 1014 study established crizotinib as the standard first-line therapy for advanced ALK-rearranged NSCLC, showing superior PFS versus pemetrexed doublet chemotherapy (HR, 0.45), but median PFS with crizotinib in that trial was only 10.9 months, Dr. Camidge said.
The next-generation ALK inhibitor brigatinib, however, has demonstrated broad preclinical activity against ALK resistance mutations, has excellent CNS penetration, and is the only ALK inhibitor with marked activity against multiple epidermal growth factor receptor–mutant cell lines, he said. “It has already shown significant activity both within the CNS and extracranially in the postcrizotinib setting, where it has consistently demonstrated the longest reported median PFS [up to 16.7 months] of any licensed or experimental ALK inhibitor.”
The ALTA-1L study provides a head-to-head comparison of brigatinib and crizotinib in a real-world setting, and the findings demonstrate that “brigatinib represents a promising new treatment for inhibitor-naive, ALK-rearranged NSCLC,” he said.
In a press statement, Dr. Camidge further stated that, based on these findings, brigatinib is set to become a first-line treatment option for ALK-positive lung cancer, adding that, “even with only 9-11 months of follow-up, the efficacy of brigatinib is clearly superior to crizotinib. A lot of the initial difference is driven by an effect on brain metastases, which tend to be an earlier progression event. However, once differences in control of disease outside the brain have time to manifest, it is possible the PFS improvement may increase.”
Both Dr. Camidge and invited discussant Fiona Blackhall, PhD, noted that the tolerability of brigatinib may be “even better in the real world,” as most of the 29% of patients on the brigatinib arm with adverse event–related dose reductions had “paper toxicities” for which the clinical impact is not well understood.
The rate of dose reductions for adverse events with brigatinib was higher in ALTA-1L than that seen in earlier studies of the second-generation ALK inhibitor alectinib, said Dr. Blackhall, professor and chair in thoracic oncology at the University of Manchester (England).
“We need to be sure that these dose reductions are appropriate and not potentially compromising efficacy,” she added.
Dr. Blackhall also suggested that sequential use of crizotinib and other ALK inhibitors like brigatinib should be evaluated and that the potential impact of ALK inhibitor selection “on the spectrum and type of resistance mutations and mechanisms” should be considered.
“Brigatinib is a new first-line treatment option for patients with ALK-positive non–small cell lung cancer. ... But in the absence of direct comparison of next-generation ALK inhibitors, it is going to take some time before we can determine whether there is indeed – if there ever will be – a ‘best’ ALK inhibitor for our patients,” she said.
Dr. Camidge has received honoraria from Arrys/Kyn, AstraZeneca, Biothera, Celgene, Clovis Oncology, Daiichi Sankyo, G1 Therapeutics, Genoptix, Hansoh, Ignyta, Lycera, Mersana Therapeutics, Novartis, Orion, Revolution Medicines, Roche/Genentech, and Takeda, and has received research funding from ARIADTakeda. Dr. Blackhall reported research funding from Amgen, AstraZeneca, Novartis, and Pfizer, and has served has an advisory board member, consultant, or speaker for AbbVie, Boehringer Ingelheim, Cell Medica, Medivation, Merck, Regeneron, Roche, and Takeda.
SOURCE: Camidge DR et al. WCLC 2018, Abstract PL02.03.
TORONTO – in the first interim results from the multicenter, open-label, phase 3 ALTA-1L trial.
In fact, the primary endpoint of the study – blinded independent review committee–assessed progression-free survival (PFS) – was met at this first analysis. The next-generation ALK inhibitor brigatinib reduced the chance of progression or death by 51% as compared with crizotinib – the current first-line standard of care in this population, D. Ross Camidge, MD, reported at the World Conference on Lung Cancer.
“At a median follow-up of 9-11 months, 26% [of 137 patients] on the brigatinib arm and 46% [of 138 patients] on the crizotinib arm had experienced a PFS event. Median PFS was not reached for the brigatinib arm and was 9.8 months for the crizotinib arm; the hazard ratio for progression or death was 0.49 and highly statistically significant,” said Dr. Camidge, director of thoracic oncology and the Joyce Zeff Chair in Lung Cancer Research at the University of Colorado Cancer Center, Aurora, at the meeting sponsored by the International Association for the Study of Lung Cancer.
The findings were published simultaneously in the New England Journal of Medicine.
The 12-month PFS rate estimate was 67% for brigatinib and 43% for crizotinib, and the investigator-assessed hazard ratio for PFS was 0.45. Overall survival data are immature, Dr. Camidge added, noting that the PFS hazard ratios for those with versus those without prior chemotherapy were 0.35 and 0.55, respectively.
“PFS consistently favored brigatinib across all other subgroups,” he said. “Although the confidence intervals overlap, the effect size appears greater among those with baseline [CNS] disease than among those without [HR, 0.2 vs. 0.72].”
Data suggest that CNS progression among those with CNS disease at baseline tends to be an earlier event than either extra-CNS progression in general or CNS progression in those without baseline CNS disease. “Consequently, this first interim analysis may be preferentially emphasizing drug efficacy differences within the subgroup in whom the earlier progression events are occurring,” Dr. Camidge said.
As for overall objective responses, the rates were numerically higher with brigatinib versus crizotinib (70% vs. 60%), but were not statistically different in the two groups. “However, the median duration of confirmed responses was not reached for brigatinib and [was] 11.1 months with crizotinib, with the 12-month [probability] of sustained response being 75% for brigatinib and 41% for crizotinib,” he added.
Further, among those with measurable CNS lesions, brigatinib demonstrated a significantly higher intracranial response rate of 78% versus 29% (odds ratio, 10.42), and when those with nonmeasurable CNS disease were included in the intracranial response assessment, the odds ratio was 13.
Median intracranial PFS among those with CNS involvement at baseline was not reached in the brigatinib arm versus 5.6 months with crizotinib, for a highly statistically significant hazard ratio of 0.27. “Which, given that the median PFS for crizotinib in the overall population ... was 9.8 months, again emphasizes how CNS events in this subgroup tend to occur early,” he noted.
Study subjects had stage IIIB/IV ALK-positive non–small cell lung cancer (NSCLC) based on local ALK testing, Eastern Cooperative Oncology Group performance status of 0-2, no more than one prior systemic therapy for locally advanced/metastatic NSCLC, and no prior ALK inhibitor therapy, Dr. Camidge said, noting that asymptomatic, untreated brain metastases were allowed and crossover to the brigatinib arm was permitted for those with blinded independent review committee–assessed progression on crizotinib.
Those randomized to the brigatinib arm had a median age of 58 years and received 180 mg daily with a 7-day lead-in at 90 mg. Those randomized to the crizotinib group had a median age of 60 years and received 250 mg twice daily. Prior chemotherapy for advanced disease was received by 26% and 27% and brain metastasis was present in 29% and 30% of patients in the arms, respectively.
The most common treatment-emergent adverse events of grade 3 or higher in the brigatinib patients were laboratory abnormalities such as creatinine phosphokinase, lipase, and amylase increases, and the most common in the crizotinib group were gastrointestinal effects, transaminitis, bradycardia, peripheral edema, and vision disturbances.
Discontinuations caused by adverse events occurred in 12% and 9% of the brigatinib and crizotinib patients, respectively, he said. No clinical cases of pancreatitis occurred in either arm, there was no difference in the incidence of any grade myalgia or musculoskeletal pain between arms, and there was no grade 3 or greater myalgia or musculoskeletal pain reported.
Early-onset interstitial lung disease, however, “appears to be a unique side effect of brigatinib,” Dr. Camidge noted.
Although interstitial lung disease/pneumonitis occurred in both groups, onset at days 3-8 after treatment initiation occurred only with brigatinib, he said, adding that “it only occurred in 3% of cases – half the rate seen in the postcrizotinib setting.”
The PROFILE 1014 study established crizotinib as the standard first-line therapy for advanced ALK-rearranged NSCLC, showing superior PFS versus pemetrexed doublet chemotherapy (HR, 0.45), but median PFS with crizotinib in that trial was only 10.9 months, Dr. Camidge said.
The next-generation ALK inhibitor brigatinib, however, has demonstrated broad preclinical activity against ALK resistance mutations, has excellent CNS penetration, and is the only ALK inhibitor with marked activity against multiple epidermal growth factor receptor–mutant cell lines, he said. “It has already shown significant activity both within the CNS and extracranially in the postcrizotinib setting, where it has consistently demonstrated the longest reported median PFS [up to 16.7 months] of any licensed or experimental ALK inhibitor.”
The ALTA-1L study provides a head-to-head comparison of brigatinib and crizotinib in a real-world setting, and the findings demonstrate that “brigatinib represents a promising new treatment for inhibitor-naive, ALK-rearranged NSCLC,” he said.
In a press statement, Dr. Camidge further stated that, based on these findings, brigatinib is set to become a first-line treatment option for ALK-positive lung cancer, adding that, “even with only 9-11 months of follow-up, the efficacy of brigatinib is clearly superior to crizotinib. A lot of the initial difference is driven by an effect on brain metastases, which tend to be an earlier progression event. However, once differences in control of disease outside the brain have time to manifest, it is possible the PFS improvement may increase.”
Both Dr. Camidge and invited discussant Fiona Blackhall, PhD, noted that the tolerability of brigatinib may be “even better in the real world,” as most of the 29% of patients on the brigatinib arm with adverse event–related dose reductions had “paper toxicities” for which the clinical impact is not well understood.
The rate of dose reductions for adverse events with brigatinib was higher in ALTA-1L than that seen in earlier studies of the second-generation ALK inhibitor alectinib, said Dr. Blackhall, professor and chair in thoracic oncology at the University of Manchester (England).
“We need to be sure that these dose reductions are appropriate and not potentially compromising efficacy,” she added.
Dr. Blackhall also suggested that sequential use of crizotinib and other ALK inhibitors like brigatinib should be evaluated and that the potential impact of ALK inhibitor selection “on the spectrum and type of resistance mutations and mechanisms” should be considered.
“Brigatinib is a new first-line treatment option for patients with ALK-positive non–small cell lung cancer. ... But in the absence of direct comparison of next-generation ALK inhibitors, it is going to take some time before we can determine whether there is indeed – if there ever will be – a ‘best’ ALK inhibitor for our patients,” she said.
Dr. Camidge has received honoraria from Arrys/Kyn, AstraZeneca, Biothera, Celgene, Clovis Oncology, Daiichi Sankyo, G1 Therapeutics, Genoptix, Hansoh, Ignyta, Lycera, Mersana Therapeutics, Novartis, Orion, Revolution Medicines, Roche/Genentech, and Takeda, and has received research funding from ARIADTakeda. Dr. Blackhall reported research funding from Amgen, AstraZeneca, Novartis, and Pfizer, and has served has an advisory board member, consultant, or speaker for AbbVie, Boehringer Ingelheim, Cell Medica, Medivation, Merck, Regeneron, Roche, and Takeda.
SOURCE: Camidge DR et al. WCLC 2018, Abstract PL02.03.
TORONTO – in the first interim results from the multicenter, open-label, phase 3 ALTA-1L trial.
In fact, the primary endpoint of the study – blinded independent review committee–assessed progression-free survival (PFS) – was met at this first analysis. The next-generation ALK inhibitor brigatinib reduced the chance of progression or death by 51% as compared with crizotinib – the current first-line standard of care in this population, D. Ross Camidge, MD, reported at the World Conference on Lung Cancer.
“At a median follow-up of 9-11 months, 26% [of 137 patients] on the brigatinib arm and 46% [of 138 patients] on the crizotinib arm had experienced a PFS event. Median PFS was not reached for the brigatinib arm and was 9.8 months for the crizotinib arm; the hazard ratio for progression or death was 0.49 and highly statistically significant,” said Dr. Camidge, director of thoracic oncology and the Joyce Zeff Chair in Lung Cancer Research at the University of Colorado Cancer Center, Aurora, at the meeting sponsored by the International Association for the Study of Lung Cancer.
The findings were published simultaneously in the New England Journal of Medicine.
The 12-month PFS rate estimate was 67% for brigatinib and 43% for crizotinib, and the investigator-assessed hazard ratio for PFS was 0.45. Overall survival data are immature, Dr. Camidge added, noting that the PFS hazard ratios for those with versus those without prior chemotherapy were 0.35 and 0.55, respectively.
“PFS consistently favored brigatinib across all other subgroups,” he said. “Although the confidence intervals overlap, the effect size appears greater among those with baseline [CNS] disease than among those without [HR, 0.2 vs. 0.72].”
Data suggest that CNS progression among those with CNS disease at baseline tends to be an earlier event than either extra-CNS progression in general or CNS progression in those without baseline CNS disease. “Consequently, this first interim analysis may be preferentially emphasizing drug efficacy differences within the subgroup in whom the earlier progression events are occurring,” Dr. Camidge said.
As for overall objective responses, the rates were numerically higher with brigatinib versus crizotinib (70% vs. 60%), but were not statistically different in the two groups. “However, the median duration of confirmed responses was not reached for brigatinib and [was] 11.1 months with crizotinib, with the 12-month [probability] of sustained response being 75% for brigatinib and 41% for crizotinib,” he added.
Further, among those with measurable CNS lesions, brigatinib demonstrated a significantly higher intracranial response rate of 78% versus 29% (odds ratio, 10.42), and when those with nonmeasurable CNS disease were included in the intracranial response assessment, the odds ratio was 13.
Median intracranial PFS among those with CNS involvement at baseline was not reached in the brigatinib arm versus 5.6 months with crizotinib, for a highly statistically significant hazard ratio of 0.27. “Which, given that the median PFS for crizotinib in the overall population ... was 9.8 months, again emphasizes how CNS events in this subgroup tend to occur early,” he noted.
Study subjects had stage IIIB/IV ALK-positive non–small cell lung cancer (NSCLC) based on local ALK testing, Eastern Cooperative Oncology Group performance status of 0-2, no more than one prior systemic therapy for locally advanced/metastatic NSCLC, and no prior ALK inhibitor therapy, Dr. Camidge said, noting that asymptomatic, untreated brain metastases were allowed and crossover to the brigatinib arm was permitted for those with blinded independent review committee–assessed progression on crizotinib.
Those randomized to the brigatinib arm had a median age of 58 years and received 180 mg daily with a 7-day lead-in at 90 mg. Those randomized to the crizotinib group had a median age of 60 years and received 250 mg twice daily. Prior chemotherapy for advanced disease was received by 26% and 27% and brain metastasis was present in 29% and 30% of patients in the arms, respectively.
The most common treatment-emergent adverse events of grade 3 or higher in the brigatinib patients were laboratory abnormalities such as creatinine phosphokinase, lipase, and amylase increases, and the most common in the crizotinib group were gastrointestinal effects, transaminitis, bradycardia, peripheral edema, and vision disturbances.
Discontinuations caused by adverse events occurred in 12% and 9% of the brigatinib and crizotinib patients, respectively, he said. No clinical cases of pancreatitis occurred in either arm, there was no difference in the incidence of any grade myalgia or musculoskeletal pain between arms, and there was no grade 3 or greater myalgia or musculoskeletal pain reported.
Early-onset interstitial lung disease, however, “appears to be a unique side effect of brigatinib,” Dr. Camidge noted.
Although interstitial lung disease/pneumonitis occurred in both groups, onset at days 3-8 after treatment initiation occurred only with brigatinib, he said, adding that “it only occurred in 3% of cases – half the rate seen in the postcrizotinib setting.”
The PROFILE 1014 study established crizotinib as the standard first-line therapy for advanced ALK-rearranged NSCLC, showing superior PFS versus pemetrexed doublet chemotherapy (HR, 0.45), but median PFS with crizotinib in that trial was only 10.9 months, Dr. Camidge said.
The next-generation ALK inhibitor brigatinib, however, has demonstrated broad preclinical activity against ALK resistance mutations, has excellent CNS penetration, and is the only ALK inhibitor with marked activity against multiple epidermal growth factor receptor–mutant cell lines, he said. “It has already shown significant activity both within the CNS and extracranially in the postcrizotinib setting, where it has consistently demonstrated the longest reported median PFS [up to 16.7 months] of any licensed or experimental ALK inhibitor.”
The ALTA-1L study provides a head-to-head comparison of brigatinib and crizotinib in a real-world setting, and the findings demonstrate that “brigatinib represents a promising new treatment for inhibitor-naive, ALK-rearranged NSCLC,” he said.
In a press statement, Dr. Camidge further stated that, based on these findings, brigatinib is set to become a first-line treatment option for ALK-positive lung cancer, adding that, “even with only 9-11 months of follow-up, the efficacy of brigatinib is clearly superior to crizotinib. A lot of the initial difference is driven by an effect on brain metastases, which tend to be an earlier progression event. However, once differences in control of disease outside the brain have time to manifest, it is possible the PFS improvement may increase.”
Both Dr. Camidge and invited discussant Fiona Blackhall, PhD, noted that the tolerability of brigatinib may be “even better in the real world,” as most of the 29% of patients on the brigatinib arm with adverse event–related dose reductions had “paper toxicities” for which the clinical impact is not well understood.
The rate of dose reductions for adverse events with brigatinib was higher in ALTA-1L than that seen in earlier studies of the second-generation ALK inhibitor alectinib, said Dr. Blackhall, professor and chair in thoracic oncology at the University of Manchester (England).
“We need to be sure that these dose reductions are appropriate and not potentially compromising efficacy,” she added.
Dr. Blackhall also suggested that sequential use of crizotinib and other ALK inhibitors like brigatinib should be evaluated and that the potential impact of ALK inhibitor selection “on the spectrum and type of resistance mutations and mechanisms” should be considered.
“Brigatinib is a new first-line treatment option for patients with ALK-positive non–small cell lung cancer. ... But in the absence of direct comparison of next-generation ALK inhibitors, it is going to take some time before we can determine whether there is indeed – if there ever will be – a ‘best’ ALK inhibitor for our patients,” she said.
Dr. Camidge has received honoraria from Arrys/Kyn, AstraZeneca, Biothera, Celgene, Clovis Oncology, Daiichi Sankyo, G1 Therapeutics, Genoptix, Hansoh, Ignyta, Lycera, Mersana Therapeutics, Novartis, Orion, Revolution Medicines, Roche/Genentech, and Takeda, and has received research funding from ARIADTakeda. Dr. Blackhall reported research funding from Amgen, AstraZeneca, Novartis, and Pfizer, and has served has an advisory board member, consultant, or speaker for AbbVie, Boehringer Ingelheim, Cell Medica, Medivation, Merck, Regeneron, Roche, and Takeda.
SOURCE: Camidge DR et al. WCLC 2018, Abstract PL02.03.
REPORTING FROM WCLC 2018
Key clinical point: Brigatinib improves progression-free survival versus crizotinib in anaplastic lymphoma kinase–positive non–small cell lung cancer.
Major finding: Brigatinib reduced the chance of progression or death by 51% versus crizotinib.
Study details: The multicenter, open-label, phase 3 ALTA-1L trial of 275 patients.
Disclosures: Dr. Camidge has received honoraria from Arrys/Kyn, AstraZeneca, Biothera, Celgene, Clovis Oncology, Daiichi Sankyo, G1 Therapeutics, Genoptix, Hansoh, Ignyta, Lycera, Mersana Therapeutics, Novartis, Orion, Revolution Medicines, Roche/Genentech, and Takeda, and has received research funding from ARIAD/Takeda. Dr. Blackhall reported research funding from Amgen, AstraZeneca, Novartis, and Pfizer, and has served has an advisory board member, consultant, or speaker for AbbVie, Boehringer Ingelheim, Cell Medica, Medivation, Merck, Regeneron, Roche, and Takeda.
Source: Camidge DR et al. WCLC 2018, Abstract PL02.03.









