User login
Treat ulcerative colitis ‘to target’
LAS VEGAS – In ulcerative colitis, symptoms aren’t everything. That’s the opinion of Stephen B. Hanauer, MD, medical director of Northwestern University’s Digestive Health Center, Chicago, who discussed the condition and the methods physicians use to assess patient improvement, at the annual meeting of the American College of Gastroenterology.
Many gastroenterologists accept patient reports of symptom improvement, but Dr. Hanauer advocates for a follow-up endoscopy, even when patients are making good progress.
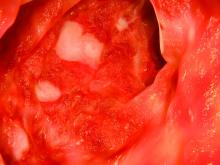
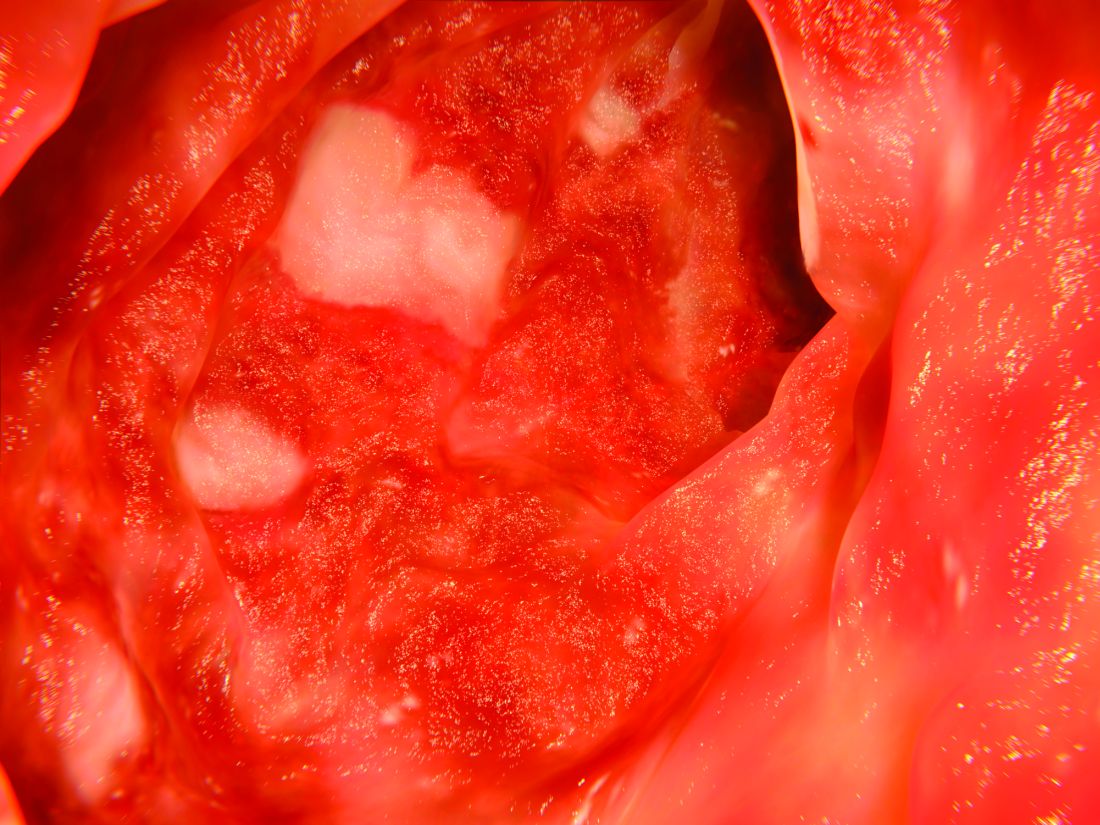
Even in patients with improved symptoms, many endoscopic evaluations show inflammation and troubling histology that is in turn associated with worse quality of life and greater odds of hospitalizations and surgery (Dig Liver Dis. 2013 Dec;45:969-77).
So a better outcome is histological healing, characterized by the absence of neutrophils. A study presented at Digestive Disease Week® in 2015 (abstract 592) showed better relapse-free survival after histologic remission. “The deeper the remission that we achieve, the better the outcomes are going to be,” said Dr. Hanauer.
In fact, patients who report that they are asymptomatic may still be experiencing disease. “Many chronic patients accept a certain degree of symptoms as a new normal,” Dr. Hanauer said. Patients accept living with frequent nighttime awakenings to evacuate, or more frequent bowel movements. Further treatment could reduce or eliminate such problems. “There are subtle things you can improve upon, to convince them that the new normal is not the acceptable normal,” said Dr. Hanauer.
He suggests an endoscopy 3-12 months after patients report symptom abatement, although he admits that cost and other considerations can be barriers.
When an endoscopy is performed, it can lead to a conundrum if it shows residual inflammation despite a lack of symptoms. The physician might consider switching to a different class of drug, but that will usually raise cost and introduce new risks of adverse events. “We are doing that in some settings. For example, in postoperative Crohn’s disease, where patients have no active disease, we advocate prophylactically administering agents to prevent recurrence. So we are willing to do that in certain subsets of patients, but we have clinical trials to show the benefits of that,” said Dr. Hanauer. Unfortunately, there are no clinical trials demonstrating the benefits of such an approach in ulcerative colitis.
Biomarkers offer potential assistance for physicians who want to “treat to target,” as Dr. Hanauer put it, to achieve biological remission. Calprotectin – a protein secreted by neutrophils that mark the presence of these cells – are measurable in feces, and the levels correlate well with ulcerative colitis disease activity (Inflamm Bowel Dis. 2012 Nov;18:2011-7).
Dr. Hanauer’s advice to go beyond clinical remission is already occurring at the Food and Drug Administration, which no longer accepts clinical remission findings in testing new drugs for inflammatory bowel disease. “They are requiring patient-reported outcomes accompanied by endoscopic evidence of healing. I’m advocating what’s going on on a regulatory basis be applied on a clinical basis,” said Dr. Hanauer.
Dr. Hanauer had no relevant financial disclosures.
LAS VEGAS – In ulcerative colitis, symptoms aren’t everything. That’s the opinion of Stephen B. Hanauer, MD, medical director of Northwestern University’s Digestive Health Center, Chicago, who discussed the condition and the methods physicians use to assess patient improvement, at the annual meeting of the American College of Gastroenterology.
Many gastroenterologists accept patient reports of symptom improvement, but Dr. Hanauer advocates for a follow-up endoscopy, even when patients are making good progress.
Even in patients with improved symptoms, many endoscopic evaluations show inflammation and troubling histology that is in turn associated with worse quality of life and greater odds of hospitalizations and surgery (Dig Liver Dis. 2013 Dec;45:969-77).
So a better outcome is histological healing, characterized by the absence of neutrophils. A study presented at Digestive Disease Week® in 2015 (abstract 592) showed better relapse-free survival after histologic remission. “The deeper the remission that we achieve, the better the outcomes are going to be,” said Dr. Hanauer.
In fact, patients who report that they are asymptomatic may still be experiencing disease. “Many chronic patients accept a certain degree of symptoms as a new normal,” Dr. Hanauer said. Patients accept living with frequent nighttime awakenings to evacuate, or more frequent bowel movements. Further treatment could reduce or eliminate such problems. “There are subtle things you can improve upon, to convince them that the new normal is not the acceptable normal,” said Dr. Hanauer.
He suggests an endoscopy 3-12 months after patients report symptom abatement, although he admits that cost and other considerations can be barriers.
When an endoscopy is performed, it can lead to a conundrum if it shows residual inflammation despite a lack of symptoms. The physician might consider switching to a different class of drug, but that will usually raise cost and introduce new risks of adverse events. “We are doing that in some settings. For example, in postoperative Crohn’s disease, where patients have no active disease, we advocate prophylactically administering agents to prevent recurrence. So we are willing to do that in certain subsets of patients, but we have clinical trials to show the benefits of that,” said Dr. Hanauer. Unfortunately, there are no clinical trials demonstrating the benefits of such an approach in ulcerative colitis.
Biomarkers offer potential assistance for physicians who want to “treat to target,” as Dr. Hanauer put it, to achieve biological remission. Calprotectin – a protein secreted by neutrophils that mark the presence of these cells – are measurable in feces, and the levels correlate well with ulcerative colitis disease activity (Inflamm Bowel Dis. 2012 Nov;18:2011-7).
Dr. Hanauer’s advice to go beyond clinical remission is already occurring at the Food and Drug Administration, which no longer accepts clinical remission findings in testing new drugs for inflammatory bowel disease. “They are requiring patient-reported outcomes accompanied by endoscopic evidence of healing. I’m advocating what’s going on on a regulatory basis be applied on a clinical basis,” said Dr. Hanauer.
Dr. Hanauer had no relevant financial disclosures.
LAS VEGAS – In ulcerative colitis, symptoms aren’t everything. That’s the opinion of Stephen B. Hanauer, MD, medical director of Northwestern University’s Digestive Health Center, Chicago, who discussed the condition and the methods physicians use to assess patient improvement, at the annual meeting of the American College of Gastroenterology.
Many gastroenterologists accept patient reports of symptom improvement, but Dr. Hanauer advocates for a follow-up endoscopy, even when patients are making good progress.
Even in patients with improved symptoms, many endoscopic evaluations show inflammation and troubling histology that is in turn associated with worse quality of life and greater odds of hospitalizations and surgery (Dig Liver Dis. 2013 Dec;45:969-77).
So a better outcome is histological healing, characterized by the absence of neutrophils. A study presented at Digestive Disease Week® in 2015 (abstract 592) showed better relapse-free survival after histologic remission. “The deeper the remission that we achieve, the better the outcomes are going to be,” said Dr. Hanauer.
In fact, patients who report that they are asymptomatic may still be experiencing disease. “Many chronic patients accept a certain degree of symptoms as a new normal,” Dr. Hanauer said. Patients accept living with frequent nighttime awakenings to evacuate, or more frequent bowel movements. Further treatment could reduce or eliminate such problems. “There are subtle things you can improve upon, to convince them that the new normal is not the acceptable normal,” said Dr. Hanauer.
He suggests an endoscopy 3-12 months after patients report symptom abatement, although he admits that cost and other considerations can be barriers.
When an endoscopy is performed, it can lead to a conundrum if it shows residual inflammation despite a lack of symptoms. The physician might consider switching to a different class of drug, but that will usually raise cost and introduce new risks of adverse events. “We are doing that in some settings. For example, in postoperative Crohn’s disease, where patients have no active disease, we advocate prophylactically administering agents to prevent recurrence. So we are willing to do that in certain subsets of patients, but we have clinical trials to show the benefits of that,” said Dr. Hanauer. Unfortunately, there are no clinical trials demonstrating the benefits of such an approach in ulcerative colitis.
Biomarkers offer potential assistance for physicians who want to “treat to target,” as Dr. Hanauer put it, to achieve biological remission. Calprotectin – a protein secreted by neutrophils that mark the presence of these cells – are measurable in feces, and the levels correlate well with ulcerative colitis disease activity (Inflamm Bowel Dis. 2012 Nov;18:2011-7).
Dr. Hanauer’s advice to go beyond clinical remission is already occurring at the Food and Drug Administration, which no longer accepts clinical remission findings in testing new drugs for inflammatory bowel disease. “They are requiring patient-reported outcomes accompanied by endoscopic evidence of healing. I’m advocating what’s going on on a regulatory basis be applied on a clinical basis,” said Dr. Hanauer.
Dr. Hanauer had no relevant financial disclosures.
EXPERT ANALYSIS FROM ACG 2016
Healthy relatives of IBD patients have intestinal dysbiosis
Healthy first-degree relatives of children and adolescents with inflammatory bowel disease had intestinal dysbiosis and an altered intestinal metabolome that correlated with one another and with the disease state, researchers reported in the November issue of Cellular and Molecular Gastroenterology and Hepatology.
These findings suggest the existence of a “high-risk microbiome/metabolome” that may increase susceptibility to inflammatory bowel disease (IBD), Jonathan Jacobs, MD, of the University of California, Los Angeles, reported with his associates. Among the healthy relatives with dysbiosis, nearly one-third also had elevated levels of fecal calprotectin, a marker of intestinal inflammation that has been previously reported in family members of patients with ulcerative colitis, the researchers noted. “If validated, prospective identification of [high-risk individuals] creates the opportunity to prevent disease development by targeting the dysbiosis and/or its metabolic consequences in the intestine,” they wrote.
Participants in the study fell into one of two “operational taxonomic units” based on their microbial communities, the researchers said. The second taxonomic unit not only was associated with IBD, “but irrespective of disease status, had lower microbial diversity and characteristic shifts in microbial composition, including increased Enterobacteriaceae, consistent with dysbiosis.” Among 21 families in the study, 19 had at least one member in this group. Furthermore, tests of an independent pediatric cohort revealed this same taxonomic group characterized by low microbial diversity and associated with IBD.
Participants also fell into two distinct metabotypes, one of which was associated with IBD and had increased levels of bile acids and amino acid derivatives previously linked to Crohn’s disease, as well as elevated levels of taurine, tryptophan, serinyl tryptophan, and adrenic acid, an omega-6 fatty acid, the researchers said. Taxonomic units and metabolic groups correlated highly with one another among both IBD patients and healthy relatives (P = 3 × 10-8), suggesting a “functional relationship between the microbiome and the metabolome,” the investigators said. Additionally, although most healthy relatives had normal fecal calprotectin levels, those in the second operational taxonomic unit had about sixfold greater odds of elevated calprotectin, compared with those in the first group (P = .05).
“The existence of an IBD-associated operational taxonomic unit type and metabotype in healthy relatives suggests that dysbiosis with its associated metabolic products may be a preexisting trait that precedes the acquisition of [inflammatory bowel] disease,” the investigators concluded. “Validating this interpretation will require prospective longitudinal studies to assess the incidence of IBD in individuals stratified by operational taxonomic type, metabotype, and calprotectin, and to assess the stability of operational taxonomic unit types and metabotypes across time and diet.”
The research was supported by the Helmsley Charitable Trust, the Crohn’s and Colitis Foundation of America, the Fineberg Foundation, the United States Public Health Service, the National Institutes of Health, the Cedars-Sinai F. Widjaja Foundation Inflammatory Bowel and Immunobiology Research Institute, the European Union, and the Joshua L. and Lisa Z. Greer Chair in Inflammatory Bowel Disease Genetics. The investigators had no conflicts of interest.
Numerous studies have established an association between intestinal dysbiosis and inflammatory bowel disease, and data from mouse models of colitis show that IBD-associated microbiota can exacerbate ongoing inflammation. Still, it is not clear that dysbiosis is either necessary or sufficient to trigger IBD – it could be that the microbiome must first be shaped by an inflammatory state on the host’s side before it can achieve a pathogenic signature and contribute to disease. New findings reported here speak to this question by demonstrating the presence of an IBD-like intestinal microbiome signature in a high-risk population, healthy first-degree relatives of IBD patients. The observation of dysbiosis in at-risk but asymptomatic individuals is an important step toward understanding the sequence of disease onset, raising the provocative possibility that, at least in some cases, dysbiosis is a predisease state, and could potentially be an instigator. The key next step will be longitudinal studies testing the predictive power of these findings. If healthy relatives with the IBD-associated microbiome signatures proceed to develop disease at an increased rate versus relatives with normal gut flora, these signatures would have potential clinical utility either as a screening and risk-assessment tool, or possibly as a target for preventative treatment.
Mark R. Frey, PhD, is assistant professor of pediatrics and biochemistry & molecular medicine at the Saban Research Institute at Children’s Hospital Los Angeles, University of Southern California, Los Angeles. He has no conflicts of interest to disclose.
Numerous studies have established an association between intestinal dysbiosis and inflammatory bowel disease, and data from mouse models of colitis show that IBD-associated microbiota can exacerbate ongoing inflammation. Still, it is not clear that dysbiosis is either necessary or sufficient to trigger IBD – it could be that the microbiome must first be shaped by an inflammatory state on the host’s side before it can achieve a pathogenic signature and contribute to disease. New findings reported here speak to this question by demonstrating the presence of an IBD-like intestinal microbiome signature in a high-risk population, healthy first-degree relatives of IBD patients. The observation of dysbiosis in at-risk but asymptomatic individuals is an important step toward understanding the sequence of disease onset, raising the provocative possibility that, at least in some cases, dysbiosis is a predisease state, and could potentially be an instigator. The key next step will be longitudinal studies testing the predictive power of these findings. If healthy relatives with the IBD-associated microbiome signatures proceed to develop disease at an increased rate versus relatives with normal gut flora, these signatures would have potential clinical utility either as a screening and risk-assessment tool, or possibly as a target for preventative treatment.
Mark R. Frey, PhD, is assistant professor of pediatrics and biochemistry & molecular medicine at the Saban Research Institute at Children’s Hospital Los Angeles, University of Southern California, Los Angeles. He has no conflicts of interest to disclose.
Numerous studies have established an association between intestinal dysbiosis and inflammatory bowel disease, and data from mouse models of colitis show that IBD-associated microbiota can exacerbate ongoing inflammation. Still, it is not clear that dysbiosis is either necessary or sufficient to trigger IBD – it could be that the microbiome must first be shaped by an inflammatory state on the host’s side before it can achieve a pathogenic signature and contribute to disease. New findings reported here speak to this question by demonstrating the presence of an IBD-like intestinal microbiome signature in a high-risk population, healthy first-degree relatives of IBD patients. The observation of dysbiosis in at-risk but asymptomatic individuals is an important step toward understanding the sequence of disease onset, raising the provocative possibility that, at least in some cases, dysbiosis is a predisease state, and could potentially be an instigator. The key next step will be longitudinal studies testing the predictive power of these findings. If healthy relatives with the IBD-associated microbiome signatures proceed to develop disease at an increased rate versus relatives with normal gut flora, these signatures would have potential clinical utility either as a screening and risk-assessment tool, or possibly as a target for preventative treatment.
Mark R. Frey, PhD, is assistant professor of pediatrics and biochemistry & molecular medicine at the Saban Research Institute at Children’s Hospital Los Angeles, University of Southern California, Los Angeles. He has no conflicts of interest to disclose.
Healthy first-degree relatives of children and adolescents with inflammatory bowel disease had intestinal dysbiosis and an altered intestinal metabolome that correlated with one another and with the disease state, researchers reported in the November issue of Cellular and Molecular Gastroenterology and Hepatology.
These findings suggest the existence of a “high-risk microbiome/metabolome” that may increase susceptibility to inflammatory bowel disease (IBD), Jonathan Jacobs, MD, of the University of California, Los Angeles, reported with his associates. Among the healthy relatives with dysbiosis, nearly one-third also had elevated levels of fecal calprotectin, a marker of intestinal inflammation that has been previously reported in family members of patients with ulcerative colitis, the researchers noted. “If validated, prospective identification of [high-risk individuals] creates the opportunity to prevent disease development by targeting the dysbiosis and/or its metabolic consequences in the intestine,” they wrote.
Participants in the study fell into one of two “operational taxonomic units” based on their microbial communities, the researchers said. The second taxonomic unit not only was associated with IBD, “but irrespective of disease status, had lower microbial diversity and characteristic shifts in microbial composition, including increased Enterobacteriaceae, consistent with dysbiosis.” Among 21 families in the study, 19 had at least one member in this group. Furthermore, tests of an independent pediatric cohort revealed this same taxonomic group characterized by low microbial diversity and associated with IBD.
Participants also fell into two distinct metabotypes, one of which was associated with IBD and had increased levels of bile acids and amino acid derivatives previously linked to Crohn’s disease, as well as elevated levels of taurine, tryptophan, serinyl tryptophan, and adrenic acid, an omega-6 fatty acid, the researchers said. Taxonomic units and metabolic groups correlated highly with one another among both IBD patients and healthy relatives (P = 3 × 10-8), suggesting a “functional relationship between the microbiome and the metabolome,” the investigators said. Additionally, although most healthy relatives had normal fecal calprotectin levels, those in the second operational taxonomic unit had about sixfold greater odds of elevated calprotectin, compared with those in the first group (P = .05).
“The existence of an IBD-associated operational taxonomic unit type and metabotype in healthy relatives suggests that dysbiosis with its associated metabolic products may be a preexisting trait that precedes the acquisition of [inflammatory bowel] disease,” the investigators concluded. “Validating this interpretation will require prospective longitudinal studies to assess the incidence of IBD in individuals stratified by operational taxonomic type, metabotype, and calprotectin, and to assess the stability of operational taxonomic unit types and metabotypes across time and diet.”
The research was supported by the Helmsley Charitable Trust, the Crohn’s and Colitis Foundation of America, the Fineberg Foundation, the United States Public Health Service, the National Institutes of Health, the Cedars-Sinai F. Widjaja Foundation Inflammatory Bowel and Immunobiology Research Institute, the European Union, and the Joshua L. and Lisa Z. Greer Chair in Inflammatory Bowel Disease Genetics. The investigators had no conflicts of interest.
Healthy first-degree relatives of children and adolescents with inflammatory bowel disease had intestinal dysbiosis and an altered intestinal metabolome that correlated with one another and with the disease state, researchers reported in the November issue of Cellular and Molecular Gastroenterology and Hepatology.
These findings suggest the existence of a “high-risk microbiome/metabolome” that may increase susceptibility to inflammatory bowel disease (IBD), Jonathan Jacobs, MD, of the University of California, Los Angeles, reported with his associates. Among the healthy relatives with dysbiosis, nearly one-third also had elevated levels of fecal calprotectin, a marker of intestinal inflammation that has been previously reported in family members of patients with ulcerative colitis, the researchers noted. “If validated, prospective identification of [high-risk individuals] creates the opportunity to prevent disease development by targeting the dysbiosis and/or its metabolic consequences in the intestine,” they wrote.
Participants in the study fell into one of two “operational taxonomic units” based on their microbial communities, the researchers said. The second taxonomic unit not only was associated with IBD, “but irrespective of disease status, had lower microbial diversity and characteristic shifts in microbial composition, including increased Enterobacteriaceae, consistent with dysbiosis.” Among 21 families in the study, 19 had at least one member in this group. Furthermore, tests of an independent pediatric cohort revealed this same taxonomic group characterized by low microbial diversity and associated with IBD.
Participants also fell into two distinct metabotypes, one of which was associated with IBD and had increased levels of bile acids and amino acid derivatives previously linked to Crohn’s disease, as well as elevated levels of taurine, tryptophan, serinyl tryptophan, and adrenic acid, an omega-6 fatty acid, the researchers said. Taxonomic units and metabolic groups correlated highly with one another among both IBD patients and healthy relatives (P = 3 × 10-8), suggesting a “functional relationship between the microbiome and the metabolome,” the investigators said. Additionally, although most healthy relatives had normal fecal calprotectin levels, those in the second operational taxonomic unit had about sixfold greater odds of elevated calprotectin, compared with those in the first group (P = .05).
“The existence of an IBD-associated operational taxonomic unit type and metabotype in healthy relatives suggests that dysbiosis with its associated metabolic products may be a preexisting trait that precedes the acquisition of [inflammatory bowel] disease,” the investigators concluded. “Validating this interpretation will require prospective longitudinal studies to assess the incidence of IBD in individuals stratified by operational taxonomic type, metabotype, and calprotectin, and to assess the stability of operational taxonomic unit types and metabotypes across time and diet.”
The research was supported by the Helmsley Charitable Trust, the Crohn’s and Colitis Foundation of America, the Fineberg Foundation, the United States Public Health Service, the National Institutes of Health, the Cedars-Sinai F. Widjaja Foundation Inflammatory Bowel and Immunobiology Research Institute, the European Union, and the Joshua L. and Lisa Z. Greer Chair in Inflammatory Bowel Disease Genetics. The investigators had no conflicts of interest.
Key clinical point: Healthy first-degree relatives of patients with remitted inflammatory bowel disease can have intestinal dysbiosis and an altered intestinal metabolome that may signify subclinical inflammation or a “pre-IBD” state.
Major finding: Nineteen of 21 families had at least one member whose microbial taxonomy and metabolome correlated with IBD.
Data source: A cross-sectional study of 21 pediatric and adolescent probands with inflammatory bowel disease, nine parents and six siblings with IBD, and 54 healthy first-degree relatives.
Disclosures: The research was supported by the Helmsley Charitable Trust, the Crohn’s and Colitis Foundation of America, the Fineberg Foundation, the United States Public Health Service, the National Institutes of Health, the Cedars-Sinai F. Widjaja Foundation Inflammatory Bowel and Immunobiology Research Institute, the European Union, and the Joshua L. and Lisa Z. Greer Chair in Inflammatory Bowel Disease Genetics. The investigators had no conflicts of interest.
VIDEO: Duodenal bulb sampling barely increased celiac yield in low-probability cohort
Separate sampling of the duodenal bulb increased detection of celiac disease by only 0.1% when endoscopy patients had a low pretest probability of celiac disease, according to research published in the November issue of Clinical Gastroenterology and Hepatology.
Duodenal bulb histology did reveal other abnormal findings, such as chronic peptic duodenitis, gastric heterotopia, and Brunner gland hyperplasia, wrote Samantha Stoven, MD, of Mayo Clinic, Rochester, Minn., and her associates. These findings did not seem to impede the identification of celiac disease, but their clinical implications were unclear, the researchers noted.
Most studies of the diagnostic yield of duodenal bulb specimens have been performed in patients with known celiac disease or positive serology. In past studies of these high-probability cohorts, duodenal bulb sampling increased the diagnostic yield of celiac disease anywhere from 1.8% to 18%, but whether and how that finding translates to low-probability cohorts is unclear, the researchers said. Therefore, they retrospectively analyzed data from 679 endoscopy patients who had both duodenal bulb and small bowel biopsies collected at three Mayo Clinic sites in 2011. These sites are “open access,” meaning that patients can be referred for endoscopy without the approval of a gastroenterologist.
The average age of the patients was 50 years, and 63% were female. They were most commonly referred for duodenal biopsy because of chronic dyspepsia (46% of patients), diarrhea (35%), or nausea (17%). Patients with either known celiac disease or positive serology were excluded from the study (Clin Gastroenterol Hepatol. 2016 Mar 7. doi: 10.1016/j.cgh.2016.02.026). A total of 265 patients (39%) had abnormal duodenal histology, which was most often diagnosed as chronic peptic duodenitis, the researchers said. Histologic abnormalities usually involved the duodenal bulb (36% of cases), not the distal duodenum (15%; P less than .0001). However, among the 16 patients (2%) found to have celiac disease, just one patient had disease only in the duodenal bulb. Thus, duodenal bulb sampling increased the diagnostic yield of celiac disease by only 0.1% when considering the overall cohort. The patient with celiac disease limited to the bulb was a 46-year-old female presenting with diarrhea and anemia who had normal serologies but a permissive human leukocyte antigen test. Her duodenal bulb had villous atrophy and more than 25 intraepithelial lymphocytes per 100 epithelial cells, while her distal duodenum was normal.
Among the 85% of patients who had normal distal duodenums, 28% had abnormal bulb histology, most often chronic peptic duodenitis, active chronic peptic duodenitis, or gastric heterotopia, the researchers said. Among the 59% of patients whose celiac serology before endoscopy was truly unknown, only two (0.5%) had histologic changes consistent with celiac disease, which in both cases were located in the distal duodenum.
SOURCE: American Gastroenterological Association
“Individual sampling of the duodenal bulb in patients with either negative or unknown celiac serologic status can be considered in practices where expert gastrointestinal pathologists are present and there is agreement that both samples can be submitted in the same bottle, or there is not a separate charge for the additional container. Further studies may be needed to assess the diagnostic yield of separate bulb biopsies for celiac detection in all comers.”
An American College of Gastroenterology Junior Faculty Development Award helped support the work. Senior author Joseph A. Murray, MD, disclosed ties to Alba Therapeutics, Alvine Pharmaceuticals, AMAG Pharmaceuticals, and several other corporate entities. The remaining authors had no disclosures.
Histologic diagnosis of celiac disease has traditionally relied upon endoscopic biopsies from the second and third portions of the duodenum. However, several recent studies indicate that duodenal bulb biopsies may show changes of celiac disease, despite normal histology in the more distal duodenum.
In their study, Dr. Stoven and her colleagues evaluated the diagnostic utility of endoscopic duodenal bulb biopsy in patients with a low probability for celiac disease. A new diagnosis of celiac disease was made in 16 of their 679 patients (2.4%). Only one patient showed villous atrophy of the duodenal bulb with normal histology of the more distal duodenum. Although a diagnosis of celiac disease was made, the case was atypical not only because distal duodenal biopsies were normal but also because multiple celiac serology tests were negative, raising the possibility of nonceliac villous atrophy. Thus, the added diagnostic yield of duodenal bulb biopsies in this low-risk population was extremely low (0.15% at most).
Ciaran P. Kelly, MD, AGAF, professor of medicine, Harvard Medical School, director Celiac Center, Beth Israel Deaconess Medical Center, Boston, has acted as a scientific adviser to companies including Celimmune, Cour Pharmaceuticals, ImmunogenX, and Takeda; he also acts as principal investigator on a research grant on celiac disease supported by Aptalis.
Histologic diagnosis of celiac disease has traditionally relied upon endoscopic biopsies from the second and third portions of the duodenum. However, several recent studies indicate that duodenal bulb biopsies may show changes of celiac disease, despite normal histology in the more distal duodenum.
In their study, Dr. Stoven and her colleagues evaluated the diagnostic utility of endoscopic duodenal bulb biopsy in patients with a low probability for celiac disease. A new diagnosis of celiac disease was made in 16 of their 679 patients (2.4%). Only one patient showed villous atrophy of the duodenal bulb with normal histology of the more distal duodenum. Although a diagnosis of celiac disease was made, the case was atypical not only because distal duodenal biopsies were normal but also because multiple celiac serology tests were negative, raising the possibility of nonceliac villous atrophy. Thus, the added diagnostic yield of duodenal bulb biopsies in this low-risk population was extremely low (0.15% at most).
Ciaran P. Kelly, MD, AGAF, professor of medicine, Harvard Medical School, director Celiac Center, Beth Israel Deaconess Medical Center, Boston, has acted as a scientific adviser to companies including Celimmune, Cour Pharmaceuticals, ImmunogenX, and Takeda; he also acts as principal investigator on a research grant on celiac disease supported by Aptalis.
Histologic diagnosis of celiac disease has traditionally relied upon endoscopic biopsies from the second and third portions of the duodenum. However, several recent studies indicate that duodenal bulb biopsies may show changes of celiac disease, despite normal histology in the more distal duodenum.
In their study, Dr. Stoven and her colleagues evaluated the diagnostic utility of endoscopic duodenal bulb biopsy in patients with a low probability for celiac disease. A new diagnosis of celiac disease was made in 16 of their 679 patients (2.4%). Only one patient showed villous atrophy of the duodenal bulb with normal histology of the more distal duodenum. Although a diagnosis of celiac disease was made, the case was atypical not only because distal duodenal biopsies were normal but also because multiple celiac serology tests were negative, raising the possibility of nonceliac villous atrophy. Thus, the added diagnostic yield of duodenal bulb biopsies in this low-risk population was extremely low (0.15% at most).
Ciaran P. Kelly, MD, AGAF, professor of medicine, Harvard Medical School, director Celiac Center, Beth Israel Deaconess Medical Center, Boston, has acted as a scientific adviser to companies including Celimmune, Cour Pharmaceuticals, ImmunogenX, and Takeda; he also acts as principal investigator on a research grant on celiac disease supported by Aptalis.
Separate sampling of the duodenal bulb increased detection of celiac disease by only 0.1% when endoscopy patients had a low pretest probability of celiac disease, according to research published in the November issue of Clinical Gastroenterology and Hepatology.
Duodenal bulb histology did reveal other abnormal findings, such as chronic peptic duodenitis, gastric heterotopia, and Brunner gland hyperplasia, wrote Samantha Stoven, MD, of Mayo Clinic, Rochester, Minn., and her associates. These findings did not seem to impede the identification of celiac disease, but their clinical implications were unclear, the researchers noted.
Most studies of the diagnostic yield of duodenal bulb specimens have been performed in patients with known celiac disease or positive serology. In past studies of these high-probability cohorts, duodenal bulb sampling increased the diagnostic yield of celiac disease anywhere from 1.8% to 18%, but whether and how that finding translates to low-probability cohorts is unclear, the researchers said. Therefore, they retrospectively analyzed data from 679 endoscopy patients who had both duodenal bulb and small bowel biopsies collected at three Mayo Clinic sites in 2011. These sites are “open access,” meaning that patients can be referred for endoscopy without the approval of a gastroenterologist.
The average age of the patients was 50 years, and 63% were female. They were most commonly referred for duodenal biopsy because of chronic dyspepsia (46% of patients), diarrhea (35%), or nausea (17%). Patients with either known celiac disease or positive serology were excluded from the study (Clin Gastroenterol Hepatol. 2016 Mar 7. doi: 10.1016/j.cgh.2016.02.026). A total of 265 patients (39%) had abnormal duodenal histology, which was most often diagnosed as chronic peptic duodenitis, the researchers said. Histologic abnormalities usually involved the duodenal bulb (36% of cases), not the distal duodenum (15%; P less than .0001). However, among the 16 patients (2%) found to have celiac disease, just one patient had disease only in the duodenal bulb. Thus, duodenal bulb sampling increased the diagnostic yield of celiac disease by only 0.1% when considering the overall cohort. The patient with celiac disease limited to the bulb was a 46-year-old female presenting with diarrhea and anemia who had normal serologies but a permissive human leukocyte antigen test. Her duodenal bulb had villous atrophy and more than 25 intraepithelial lymphocytes per 100 epithelial cells, while her distal duodenum was normal.
Among the 85% of patients who had normal distal duodenums, 28% had abnormal bulb histology, most often chronic peptic duodenitis, active chronic peptic duodenitis, or gastric heterotopia, the researchers said. Among the 59% of patients whose celiac serology before endoscopy was truly unknown, only two (0.5%) had histologic changes consistent with celiac disease, which in both cases were located in the distal duodenum.
SOURCE: American Gastroenterological Association
“Individual sampling of the duodenal bulb in patients with either negative or unknown celiac serologic status can be considered in practices where expert gastrointestinal pathologists are present and there is agreement that both samples can be submitted in the same bottle, or there is not a separate charge for the additional container. Further studies may be needed to assess the diagnostic yield of separate bulb biopsies for celiac detection in all comers.”
An American College of Gastroenterology Junior Faculty Development Award helped support the work. Senior author Joseph A. Murray, MD, disclosed ties to Alba Therapeutics, Alvine Pharmaceuticals, AMAG Pharmaceuticals, and several other corporate entities. The remaining authors had no disclosures.
Separate sampling of the duodenal bulb increased detection of celiac disease by only 0.1% when endoscopy patients had a low pretest probability of celiac disease, according to research published in the November issue of Clinical Gastroenterology and Hepatology.
Duodenal bulb histology did reveal other abnormal findings, such as chronic peptic duodenitis, gastric heterotopia, and Brunner gland hyperplasia, wrote Samantha Stoven, MD, of Mayo Clinic, Rochester, Minn., and her associates. These findings did not seem to impede the identification of celiac disease, but their clinical implications were unclear, the researchers noted.
Most studies of the diagnostic yield of duodenal bulb specimens have been performed in patients with known celiac disease or positive serology. In past studies of these high-probability cohorts, duodenal bulb sampling increased the diagnostic yield of celiac disease anywhere from 1.8% to 18%, but whether and how that finding translates to low-probability cohorts is unclear, the researchers said. Therefore, they retrospectively analyzed data from 679 endoscopy patients who had both duodenal bulb and small bowel biopsies collected at three Mayo Clinic sites in 2011. These sites are “open access,” meaning that patients can be referred for endoscopy without the approval of a gastroenterologist.
The average age of the patients was 50 years, and 63% were female. They were most commonly referred for duodenal biopsy because of chronic dyspepsia (46% of patients), diarrhea (35%), or nausea (17%). Patients with either known celiac disease or positive serology were excluded from the study (Clin Gastroenterol Hepatol. 2016 Mar 7. doi: 10.1016/j.cgh.2016.02.026). A total of 265 patients (39%) had abnormal duodenal histology, which was most often diagnosed as chronic peptic duodenitis, the researchers said. Histologic abnormalities usually involved the duodenal bulb (36% of cases), not the distal duodenum (15%; P less than .0001). However, among the 16 patients (2%) found to have celiac disease, just one patient had disease only in the duodenal bulb. Thus, duodenal bulb sampling increased the diagnostic yield of celiac disease by only 0.1% when considering the overall cohort. The patient with celiac disease limited to the bulb was a 46-year-old female presenting with diarrhea and anemia who had normal serologies but a permissive human leukocyte antigen test. Her duodenal bulb had villous atrophy and more than 25 intraepithelial lymphocytes per 100 epithelial cells, while her distal duodenum was normal.
Among the 85% of patients who had normal distal duodenums, 28% had abnormal bulb histology, most often chronic peptic duodenitis, active chronic peptic duodenitis, or gastric heterotopia, the researchers said. Among the 59% of patients whose celiac serology before endoscopy was truly unknown, only two (0.5%) had histologic changes consistent with celiac disease, which in both cases were located in the distal duodenum.
SOURCE: American Gastroenterological Association
“Individual sampling of the duodenal bulb in patients with either negative or unknown celiac serologic status can be considered in practices where expert gastrointestinal pathologists are present and there is agreement that both samples can be submitted in the same bottle, or there is not a separate charge for the additional container. Further studies may be needed to assess the diagnostic yield of separate bulb biopsies for celiac detection in all comers.”
An American College of Gastroenterology Junior Faculty Development Award helped support the work. Senior author Joseph A. Murray, MD, disclosed ties to Alba Therapeutics, Alvine Pharmaceuticals, AMAG Pharmaceuticals, and several other corporate entities. The remaining authors had no disclosures.
FROM CLINICAL GASTROENTEROLOGY AND HEPATOLOGY
Key clinical point: Separate sampling of the duodenal bulb increased detection of celiac disease by only 0.1% when endoscopy patients had a low pretest probability of celiac disease.
Major finding: One (0.1%) patient had celiac disease limited to the duodenal bulb.
Data source: A multicenter retrospective study of 679 patients without celiac disease or positive serology from whom duodenal bulb and small bowel biopsies were collected during endoscopy.
Disclosures: An American College of Gastroenterology Junior Faculty Development Award helped support the work. Senior author Joseph A. Murray, MD, disclosed ties to Alba Therapeutics, Alvine Pharmaceuticals, AMAG Pharmaceuticals, and several other corporate entities. The remaining authors had no disclosures.
Plecanatide safe, effective for chronic constipation
LAS VEGAS – Two new studies suggest that the peptide plecanatide is safe and effective in the treatment of chronic idiopathic constipation. The drug had a low rate of diarrhea, about 7.1%.
The research, presented in two posters at the annual meeting of the American College of Gastroenterology, includes a pooled efficacy and safety analysis from two previous phase III clinical trials, as well as an open-label extension study.
Though there is no way to be sure in the absence of head-to-head studies, plecanatide could have a better tolerability profile than the existing constipation drugs like lubiprostone (Amitiza) and linaclotide (Linzess).
The drug is a derivative of uroguanylin, a peptide found in the gastrointestinal tract. Like the native peptide, plecanatide stimulates digestive fluid movement in the proximal small intestine, which in turn encourages regular bowel function. “I think because it is so much closer to the innate uroguanylin molecule, it will have better tolerability,” said Dr. Rao in an interview.
The open-label study followed 2,370 patients who received 3-mg or 6-mg doses of plecanatide once daily for up to 72 weeks. The most common adverse events were diarrhea (7.1%) and urinary tract infection (2.2%), and these were the only adverse events that occurred at a frequency above 2%.
About 5% of patients discontinued due to adverse events, 3.1% because of diarrhea. Patients were also asked to score their satisfaction with the drug and their willingness to continue on it, and the median values to those answers corresponded to quite satisfied and quite likely to continue.
The other study was a pooled analysis of two previously presented double-blind, placebo-controlled phase III trials of plecanatide. These studies included 2,791 patients with chronic idiopathic constipation who were treated over the course of 12 weeks, with 3- and 6-mg doses. Both groups showed significant improvements in the rate of durable overall complete spontaneous bowel movements: 20.5% in the 3-mg group and 19.8% in the 6-mg group, compared with 11.5% in the placebo group (P less than .001 for both comparisons).
Patients experienced improvements as early as the first week of treatment (31.6% in the 3-mg group versus 16.1% in placebo, P less than .001), and improvements were maintained through the end of the treatment period. There were also significant improvements in secondary endpoints, including stool consistency, straining, and bloating.
Adverse events occurred in 30.6% of subjects taking the 3-mg dose and 31.1% of those taking 6 mg, compared with 28.7% in the placebo group. As with the long-term study, the most common adverse event was diarrhea (4.6% in 3 mg and 5.1% in 6 mg, compared with 1.3% in placebo). Of those in the 3-mg group, 4.1% discontinued, as did 4.5% in the 6-mg group, and 2.2% in the placebo group.
“I think these are very exciting results. They clearly show a benefit of plecanatide in patients with chronic constipation. These are really methodologically rigorous, large clinical trials that should provide doctors and patients with confidence that the drug will provide benefits,” commented William D. Chey, MD, a professor of medicine at the University of Michigan, Ann Arbor.
Plecanatide is also being developed for irritable bowel syndrome with constipation, and has finished recruitment for two phase III clinical trials, which Synergy expects to report on later this year.
Dr. Rao is a member of an advisory committee for Forest Laboratories, Hollister, In Control Medical, Ironwood, Sucampo, and Vibrant. Dr. Chey is a consultant for Ironwood and Synergy.
AGA Resource
AGA offers information for your patients about constipation at http://www.gastro.org/patient-care/conditions-diseases/constipation.
LAS VEGAS – Two new studies suggest that the peptide plecanatide is safe and effective in the treatment of chronic idiopathic constipation. The drug had a low rate of diarrhea, about 7.1%.
The research, presented in two posters at the annual meeting of the American College of Gastroenterology, includes a pooled efficacy and safety analysis from two previous phase III clinical trials, as well as an open-label extension study.
Though there is no way to be sure in the absence of head-to-head studies, plecanatide could have a better tolerability profile than the existing constipation drugs like lubiprostone (Amitiza) and linaclotide (Linzess).
The drug is a derivative of uroguanylin, a peptide found in the gastrointestinal tract. Like the native peptide, plecanatide stimulates digestive fluid movement in the proximal small intestine, which in turn encourages regular bowel function. “I think because it is so much closer to the innate uroguanylin molecule, it will have better tolerability,” said Dr. Rao in an interview.
The open-label study followed 2,370 patients who received 3-mg or 6-mg doses of plecanatide once daily for up to 72 weeks. The most common adverse events were diarrhea (7.1%) and urinary tract infection (2.2%), and these were the only adverse events that occurred at a frequency above 2%.
About 5% of patients discontinued due to adverse events, 3.1% because of diarrhea. Patients were also asked to score their satisfaction with the drug and their willingness to continue on it, and the median values to those answers corresponded to quite satisfied and quite likely to continue.
The other study was a pooled analysis of two previously presented double-blind, placebo-controlled phase III trials of plecanatide. These studies included 2,791 patients with chronic idiopathic constipation who were treated over the course of 12 weeks, with 3- and 6-mg doses. Both groups showed significant improvements in the rate of durable overall complete spontaneous bowel movements: 20.5% in the 3-mg group and 19.8% in the 6-mg group, compared with 11.5% in the placebo group (P less than .001 for both comparisons).
Patients experienced improvements as early as the first week of treatment (31.6% in the 3-mg group versus 16.1% in placebo, P less than .001), and improvements were maintained through the end of the treatment period. There were also significant improvements in secondary endpoints, including stool consistency, straining, and bloating.
Adverse events occurred in 30.6% of subjects taking the 3-mg dose and 31.1% of those taking 6 mg, compared with 28.7% in the placebo group. As with the long-term study, the most common adverse event was diarrhea (4.6% in 3 mg and 5.1% in 6 mg, compared with 1.3% in placebo). Of those in the 3-mg group, 4.1% discontinued, as did 4.5% in the 6-mg group, and 2.2% in the placebo group.
“I think these are very exciting results. They clearly show a benefit of plecanatide in patients with chronic constipation. These are really methodologically rigorous, large clinical trials that should provide doctors and patients with confidence that the drug will provide benefits,” commented William D. Chey, MD, a professor of medicine at the University of Michigan, Ann Arbor.
Plecanatide is also being developed for irritable bowel syndrome with constipation, and has finished recruitment for two phase III clinical trials, which Synergy expects to report on later this year.
Dr. Rao is a member of an advisory committee for Forest Laboratories, Hollister, In Control Medical, Ironwood, Sucampo, and Vibrant. Dr. Chey is a consultant for Ironwood and Synergy.
AGA Resource
AGA offers information for your patients about constipation at http://www.gastro.org/patient-care/conditions-diseases/constipation.
LAS VEGAS – Two new studies suggest that the peptide plecanatide is safe and effective in the treatment of chronic idiopathic constipation. The drug had a low rate of diarrhea, about 7.1%.
The research, presented in two posters at the annual meeting of the American College of Gastroenterology, includes a pooled efficacy and safety analysis from two previous phase III clinical trials, as well as an open-label extension study.
Though there is no way to be sure in the absence of head-to-head studies, plecanatide could have a better tolerability profile than the existing constipation drugs like lubiprostone (Amitiza) and linaclotide (Linzess).
The drug is a derivative of uroguanylin, a peptide found in the gastrointestinal tract. Like the native peptide, plecanatide stimulates digestive fluid movement in the proximal small intestine, which in turn encourages regular bowel function. “I think because it is so much closer to the innate uroguanylin molecule, it will have better tolerability,” said Dr. Rao in an interview.
The open-label study followed 2,370 patients who received 3-mg or 6-mg doses of plecanatide once daily for up to 72 weeks. The most common adverse events were diarrhea (7.1%) and urinary tract infection (2.2%), and these were the only adverse events that occurred at a frequency above 2%.
About 5% of patients discontinued due to adverse events, 3.1% because of diarrhea. Patients were also asked to score their satisfaction with the drug and their willingness to continue on it, and the median values to those answers corresponded to quite satisfied and quite likely to continue.
The other study was a pooled analysis of two previously presented double-blind, placebo-controlled phase III trials of plecanatide. These studies included 2,791 patients with chronic idiopathic constipation who were treated over the course of 12 weeks, with 3- and 6-mg doses. Both groups showed significant improvements in the rate of durable overall complete spontaneous bowel movements: 20.5% in the 3-mg group and 19.8% in the 6-mg group, compared with 11.5% in the placebo group (P less than .001 for both comparisons).
Patients experienced improvements as early as the first week of treatment (31.6% in the 3-mg group versus 16.1% in placebo, P less than .001), and improvements were maintained through the end of the treatment period. There were also significant improvements in secondary endpoints, including stool consistency, straining, and bloating.
Adverse events occurred in 30.6% of subjects taking the 3-mg dose and 31.1% of those taking 6 mg, compared with 28.7% in the placebo group. As with the long-term study, the most common adverse event was diarrhea (4.6% in 3 mg and 5.1% in 6 mg, compared with 1.3% in placebo). Of those in the 3-mg group, 4.1% discontinued, as did 4.5% in the 6-mg group, and 2.2% in the placebo group.
“I think these are very exciting results. They clearly show a benefit of plecanatide in patients with chronic constipation. These are really methodologically rigorous, large clinical trials that should provide doctors and patients with confidence that the drug will provide benefits,” commented William D. Chey, MD, a professor of medicine at the University of Michigan, Ann Arbor.
Plecanatide is also being developed for irritable bowel syndrome with constipation, and has finished recruitment for two phase III clinical trials, which Synergy expects to report on later this year.
Dr. Rao is a member of an advisory committee for Forest Laboratories, Hollister, In Control Medical, Ironwood, Sucampo, and Vibrant. Dr. Chey is a consultant for Ironwood and Synergy.
AGA Resource
AGA offers information for your patients about constipation at http://www.gastro.org/patient-care/conditions-diseases/constipation.
AT ACG 2016
Key clinical point: Plecanatide is safe and effective in the treatment of idiopathic chronic constipation.
Major finding: A long-term study and an analysis of two phase III clinical trials show the drug is effective at reducing constipation and has low rates of adverse events and discontinuation.
Data source: Open-label extension trial and randomized, placebo-controlled, clinical trials.
Disclosures: Dr. Rao is a member of an advisory committee for Forest Laboratories, Hollister, In Control Medical, Ironwood, Sucampo, and Vibrant. Dr. Chey is a consultant for Ironwood.
Molecular subtypes of Crohn’s may shape treatment approach
Researchers have identified two distinct molecular subclasses of Crohn’s disease, based on gene expression and phenotype, which could help with better tailoring of treatment.
Crohn’s disease is a chronic inflammatory disorder with a diverse clinical presentation and pattern of progression, yet the molecular and genetic factors underlying the disease are not well understood.
This complicates treatment – which currently relies on doctors’ subjective clinical classification – and makes it difficult to develop an evidence-based personalized approach.
Writing in the Oct. 14 online edition of Gut, Matthew Weiser, a graduate student in the genetics department at the University of North Carolina at Chapel Hill and his coauthors report the results of a genetic and molecular analysis of noninflamed colon tissue from a cohort of 21 adult patients with Crohn’s disease and from healthy controls.
This analysis revealed two distinct molecular phenotypes in colon tissue, which they labeled as “colon-like” and “ileum-like.” They found significant differences in cellular metabolism and immune pathways (Gut. 2016 Oct 14. doi: 10.1136/gutjnl-2016-312518).
In the colon-like subtype, the tissue samples – which were all taken from the colon – showed gene expression patterns that were typical of cells from the colon. However, in the ileum-like subtype, the gene expression patterns in these colon cells more closely resembled the gene expression of ileum cells.
They also saw significant differences in other molecular traits, such as chromatin accessibility, and pathways involved in lipid and xenobiotic metabolism.
“Furthermore, chromatin accessibility data suggest these subclasses exist due to stable molecular transformations of the genomic architecture in colon tissue cells, and not transient differences due to external cellular signaling,” researchers reported.
To rule out the possibility that these differences might be from treatment history, researchers conducted a similar analysis using samples from 201 treatment-naive pediatric patients with Crohn’s disease and found the same molecular and genetic patterns.
“Together, these data strongly suggest that the colon-like and ileum-like molecular signatures define two forms of [Crohn’s disease] present, regardless of tissue sampling location, patient age, or treatment status.”
The researchers also examined the impact the two subtypes might have on the clinical disease and therefore the therapeutic approach taken. In the cohort of treatment-naive pediatric patients, they found that patients with the more colon-like Crohn’s disease were more likely to have both colon and ileum involvement, have deep ulcers, and show more macroscopic inflammation than those with more ileum-like Crohn’s disease.
They also found that adults in the colon-like disease subclass were the only ones who were likely to have rectal disease or require a colectomy, although they noted that the sample size was small.
More patients with the ileum-like disease showed no inflammation and were also more likely to have involvement of the colon only.
“Although our sample size is small, these data suggest that molecular subtypes of [Crohn’s disease] can stratify patients into clinically distinct and relevant subgroups and may prospectively identify those more likely to require intensive medical therapy,” the authors wrote.
“As a first step, molecular stratifications of archived patient tissue and serum from major clinical trials could be performed in the context of response to biological and microbial therapies for [Crohn’s disease].”
The authors pointed out that they did not study samples from both intestinal regions in each patient but suggested that their findings should motivate future studies in larger cohorts using matched tissue samples from both the colon and ileum.
“These data emphasize the need to continue and expand these studies over time to incorporate the evolving clinical phenotype in both adult and pediatric patients, and the need to study both tissues in the same patient.”
The study was supported by the National Institute of Environmental Health Sciences, National Institute of Diabetes and Digestive and Kidney Diseases, American Gastroenterological Association, Broad Medical Research Program, Crohn’s and Colitis Foundation of America, International Human Microbiome Consortium, UNC Team Translational Science Award, and Helmsley Charitable Trust SHARE 2, Project 3. No conflicts of interest were declared.
Researchers have identified two distinct molecular subclasses of Crohn’s disease, based on gene expression and phenotype, which could help with better tailoring of treatment.
Crohn’s disease is a chronic inflammatory disorder with a diverse clinical presentation and pattern of progression, yet the molecular and genetic factors underlying the disease are not well understood.
This complicates treatment – which currently relies on doctors’ subjective clinical classification – and makes it difficult to develop an evidence-based personalized approach.
Writing in the Oct. 14 online edition of Gut, Matthew Weiser, a graduate student in the genetics department at the University of North Carolina at Chapel Hill and his coauthors report the results of a genetic and molecular analysis of noninflamed colon tissue from a cohort of 21 adult patients with Crohn’s disease and from healthy controls.
This analysis revealed two distinct molecular phenotypes in colon tissue, which they labeled as “colon-like” and “ileum-like.” They found significant differences in cellular metabolism and immune pathways (Gut. 2016 Oct 14. doi: 10.1136/gutjnl-2016-312518).
In the colon-like subtype, the tissue samples – which were all taken from the colon – showed gene expression patterns that were typical of cells from the colon. However, in the ileum-like subtype, the gene expression patterns in these colon cells more closely resembled the gene expression of ileum cells.
They also saw significant differences in other molecular traits, such as chromatin accessibility, and pathways involved in lipid and xenobiotic metabolism.
“Furthermore, chromatin accessibility data suggest these subclasses exist due to stable molecular transformations of the genomic architecture in colon tissue cells, and not transient differences due to external cellular signaling,” researchers reported.
To rule out the possibility that these differences might be from treatment history, researchers conducted a similar analysis using samples from 201 treatment-naive pediatric patients with Crohn’s disease and found the same molecular and genetic patterns.
“Together, these data strongly suggest that the colon-like and ileum-like molecular signatures define two forms of [Crohn’s disease] present, regardless of tissue sampling location, patient age, or treatment status.”
The researchers also examined the impact the two subtypes might have on the clinical disease and therefore the therapeutic approach taken. In the cohort of treatment-naive pediatric patients, they found that patients with the more colon-like Crohn’s disease were more likely to have both colon and ileum involvement, have deep ulcers, and show more macroscopic inflammation than those with more ileum-like Crohn’s disease.
They also found that adults in the colon-like disease subclass were the only ones who were likely to have rectal disease or require a colectomy, although they noted that the sample size was small.
More patients with the ileum-like disease showed no inflammation and were also more likely to have involvement of the colon only.
“Although our sample size is small, these data suggest that molecular subtypes of [Crohn’s disease] can stratify patients into clinically distinct and relevant subgroups and may prospectively identify those more likely to require intensive medical therapy,” the authors wrote.
“As a first step, molecular stratifications of archived patient tissue and serum from major clinical trials could be performed in the context of response to biological and microbial therapies for [Crohn’s disease].”
The authors pointed out that they did not study samples from both intestinal regions in each patient but suggested that their findings should motivate future studies in larger cohorts using matched tissue samples from both the colon and ileum.
“These data emphasize the need to continue and expand these studies over time to incorporate the evolving clinical phenotype in both adult and pediatric patients, and the need to study both tissues in the same patient.”
The study was supported by the National Institute of Environmental Health Sciences, National Institute of Diabetes and Digestive and Kidney Diseases, American Gastroenterological Association, Broad Medical Research Program, Crohn’s and Colitis Foundation of America, International Human Microbiome Consortium, UNC Team Translational Science Award, and Helmsley Charitable Trust SHARE 2, Project 3. No conflicts of interest were declared.
Researchers have identified two distinct molecular subclasses of Crohn’s disease, based on gene expression and phenotype, which could help with better tailoring of treatment.
Crohn’s disease is a chronic inflammatory disorder with a diverse clinical presentation and pattern of progression, yet the molecular and genetic factors underlying the disease are not well understood.
This complicates treatment – which currently relies on doctors’ subjective clinical classification – and makes it difficult to develop an evidence-based personalized approach.
Writing in the Oct. 14 online edition of Gut, Matthew Weiser, a graduate student in the genetics department at the University of North Carolina at Chapel Hill and his coauthors report the results of a genetic and molecular analysis of noninflamed colon tissue from a cohort of 21 adult patients with Crohn’s disease and from healthy controls.
This analysis revealed two distinct molecular phenotypes in colon tissue, which they labeled as “colon-like” and “ileum-like.” They found significant differences in cellular metabolism and immune pathways (Gut. 2016 Oct 14. doi: 10.1136/gutjnl-2016-312518).
In the colon-like subtype, the tissue samples – which were all taken from the colon – showed gene expression patterns that were typical of cells from the colon. However, in the ileum-like subtype, the gene expression patterns in these colon cells more closely resembled the gene expression of ileum cells.
They also saw significant differences in other molecular traits, such as chromatin accessibility, and pathways involved in lipid and xenobiotic metabolism.
“Furthermore, chromatin accessibility data suggest these subclasses exist due to stable molecular transformations of the genomic architecture in colon tissue cells, and not transient differences due to external cellular signaling,” researchers reported.
To rule out the possibility that these differences might be from treatment history, researchers conducted a similar analysis using samples from 201 treatment-naive pediatric patients with Crohn’s disease and found the same molecular and genetic patterns.
“Together, these data strongly suggest that the colon-like and ileum-like molecular signatures define two forms of [Crohn’s disease] present, regardless of tissue sampling location, patient age, or treatment status.”
The researchers also examined the impact the two subtypes might have on the clinical disease and therefore the therapeutic approach taken. In the cohort of treatment-naive pediatric patients, they found that patients with the more colon-like Crohn’s disease were more likely to have both colon and ileum involvement, have deep ulcers, and show more macroscopic inflammation than those with more ileum-like Crohn’s disease.
They also found that adults in the colon-like disease subclass were the only ones who were likely to have rectal disease or require a colectomy, although they noted that the sample size was small.
More patients with the ileum-like disease showed no inflammation and were also more likely to have involvement of the colon only.
“Although our sample size is small, these data suggest that molecular subtypes of [Crohn’s disease] can stratify patients into clinically distinct and relevant subgroups and may prospectively identify those more likely to require intensive medical therapy,” the authors wrote.
“As a first step, molecular stratifications of archived patient tissue and serum from major clinical trials could be performed in the context of response to biological and microbial therapies for [Crohn’s disease].”
The authors pointed out that they did not study samples from both intestinal regions in each patient but suggested that their findings should motivate future studies in larger cohorts using matched tissue samples from both the colon and ileum.
“These data emphasize the need to continue and expand these studies over time to incorporate the evolving clinical phenotype in both adult and pediatric patients, and the need to study both tissues in the same patient.”
The study was supported by the National Institute of Environmental Health Sciences, National Institute of Diabetes and Digestive and Kidney Diseases, American Gastroenterological Association, Broad Medical Research Program, Crohn’s and Colitis Foundation of America, International Human Microbiome Consortium, UNC Team Translational Science Award, and Helmsley Charitable Trust SHARE 2, Project 3. No conflicts of interest were declared.
FROM GUT
Key clinical point: Researchers have identified two distinct molecular subclasses of Crohn’s disease, based on gene expression and phenotype, which could help with better tailoring of treatment.
Major finding: Molecular and genetic analysis has revealed a “colon-like” subtype and “ileum-like” subtype of Crohn’s disease, with distinct molecular profiles that correspond with clinical features.
Data source: Molecular and genetic analysis of samples from 21 adults with Crohn’s disease and 201 treatment-naive pediatric Crohn’s disease patients.
Disclosures: The study was supported by the National Institute of Environmental Health Sciences, National Institute of Diabetes and Digestive and Kidney Diseases, American Gastroenterological Association, Broad Medical Research Program, Crohn’s and Colitis Foundation of America, International Human Microbiome Consortium, UNC Team Translational Science Award, and Helmsley Charitable Trust SHARE 2, Project 3. No conflicts of interest were declared.
IBD medical homes dramatically cut ED visits, hospitalizations
LAS VEGAS – A collaborative care approach to patients with inflammatory bowel disease (IBD) dramatically reduced emergency department visits and hospitalizations and also boosted quality of life.
The IBD patient-centered medical home employs open-access scheduling, remote monitoring, and telemedicine. A patient’s team includes a social worker, a nurse practitioner, a dietitian, a psychiatrist, and other specialists. The gastroenterologist acts as a primary care physician, according to Miguel Regueiro, MD, AGAF, medical director of the IBD Center at the University of Pittsburgh Medical Center, who presented the outcomes at the annual meeting of the American College of Gastroenterology.
Patients were eligible if at least 25% of their health care expenditures in the prior year were related to Crohn’s disease or ulcerative colitis.
The team enrolled 308 patients in the first year, with 290 remaining by the end of the year. Quality of life, as measured by the Short Inflammatory Bowel Disease Questionnaire, rose from a mean score of 7.1 to 9.1 points by the end of the study. There was a 51.9% decrease in ED visits (322 to 155), and a 53.1% reduction in hospitalizations (160 to 75), compared with the previous year.
Among patients who had been in the program for at least 6 months, ED visits dropped by 47% (197 to 116; P = .001) and hospitalizations dropped by 44% (100 to 56; P less than .005).
Physicians must also rise to the challenge. “The majority of gastroenterologists wouldn’t want to do this because it puts responsibility on them for the whole patient rather than just the disease,” said Dr. Regueiro.
But that discomfort shouldn’t sway them from making necessary changes. Rising health care costs could force specialists into new roles. “The way I look at it, if we don’t do something like this, the insurance companies will tell us what to do,” said Dr. Regueiro.
Dr. Regueiro is a member of advisory boards for AbbVie, Jansen, Takeda, UCB, and Pfizer. Dr. Quigley reported no conflicts of interest.
LAS VEGAS – A collaborative care approach to patients with inflammatory bowel disease (IBD) dramatically reduced emergency department visits and hospitalizations and also boosted quality of life.
The IBD patient-centered medical home employs open-access scheduling, remote monitoring, and telemedicine. A patient’s team includes a social worker, a nurse practitioner, a dietitian, a psychiatrist, and other specialists. The gastroenterologist acts as a primary care physician, according to Miguel Regueiro, MD, AGAF, medical director of the IBD Center at the University of Pittsburgh Medical Center, who presented the outcomes at the annual meeting of the American College of Gastroenterology.
Patients were eligible if at least 25% of their health care expenditures in the prior year were related to Crohn’s disease or ulcerative colitis.
The team enrolled 308 patients in the first year, with 290 remaining by the end of the year. Quality of life, as measured by the Short Inflammatory Bowel Disease Questionnaire, rose from a mean score of 7.1 to 9.1 points by the end of the study. There was a 51.9% decrease in ED visits (322 to 155), and a 53.1% reduction in hospitalizations (160 to 75), compared with the previous year.
Among patients who had been in the program for at least 6 months, ED visits dropped by 47% (197 to 116; P = .001) and hospitalizations dropped by 44% (100 to 56; P less than .005).
Physicians must also rise to the challenge. “The majority of gastroenterologists wouldn’t want to do this because it puts responsibility on them for the whole patient rather than just the disease,” said Dr. Regueiro.
But that discomfort shouldn’t sway them from making necessary changes. Rising health care costs could force specialists into new roles. “The way I look at it, if we don’t do something like this, the insurance companies will tell us what to do,” said Dr. Regueiro.
Dr. Regueiro is a member of advisory boards for AbbVie, Jansen, Takeda, UCB, and Pfizer. Dr. Quigley reported no conflicts of interest.
LAS VEGAS – A collaborative care approach to patients with inflammatory bowel disease (IBD) dramatically reduced emergency department visits and hospitalizations and also boosted quality of life.
The IBD patient-centered medical home employs open-access scheduling, remote monitoring, and telemedicine. A patient’s team includes a social worker, a nurse practitioner, a dietitian, a psychiatrist, and other specialists. The gastroenterologist acts as a primary care physician, according to Miguel Regueiro, MD, AGAF, medical director of the IBD Center at the University of Pittsburgh Medical Center, who presented the outcomes at the annual meeting of the American College of Gastroenterology.
Patients were eligible if at least 25% of their health care expenditures in the prior year were related to Crohn’s disease or ulcerative colitis.
The team enrolled 308 patients in the first year, with 290 remaining by the end of the year. Quality of life, as measured by the Short Inflammatory Bowel Disease Questionnaire, rose from a mean score of 7.1 to 9.1 points by the end of the study. There was a 51.9% decrease in ED visits (322 to 155), and a 53.1% reduction in hospitalizations (160 to 75), compared with the previous year.
Among patients who had been in the program for at least 6 months, ED visits dropped by 47% (197 to 116; P = .001) and hospitalizations dropped by 44% (100 to 56; P less than .005).
Physicians must also rise to the challenge. “The majority of gastroenterologists wouldn’t want to do this because it puts responsibility on them for the whole patient rather than just the disease,” said Dr. Regueiro.
But that discomfort shouldn’t sway them from making necessary changes. Rising health care costs could force specialists into new roles. “The way I look at it, if we don’t do something like this, the insurance companies will tell us what to do,” said Dr. Regueiro.
Dr. Regueiro is a member of advisory boards for AbbVie, Jansen, Takeda, UCB, and Pfizer. Dr. Quigley reported no conflicts of interest.
AT ACG 2016
Key clinical point:
Major finding: ED visits were cut by 51.9% and hospitalizations by 53.1% compared to the previous year.
Data source: Observational study.
Disclosures: Dr. Regueiro is a member of advisory boards for AbbVie, Jansen, Takeda, UCB, and Pfizer. Dr. Quigley reported no conflicts of interest.
Vedolizumab shows safety, efficacy for pediatric IBD
MONTREAL – Vedolizumab, 2 years out from its entry onto the U.S. market as an option for treating adults with inflammatory bowel disease, also has shown early safety and efficacy in 52 pediatric patients with ulcerative colitis or Crohn’s disease.
Retrospective review of a patients series assembled from three U.S. centers showed 13 (76%) ulcerative colitis patients in remission among 17 treated and followed for 14 weeks, the study’s primary outcome. Among 24 Crohn’s disease patients treated and followed for 14 weeks, 10 (42%) had remissions, Namita Singh, MD, said at the World Congress of Pediatric Gastroenterology, Hepatology, and Nutrition.
Treatment with vedolizumab (Entyvio), an anti-integrin with highly gut-specific activity that limits its systemic effects, was especially potent for the five patients in the series who were naive to treatment with an anti–tumor necrosis factor (TNF) agent. All five patients were in remission at 14 weeks, said Dr. Singh, a pediatric gastroenterologist at Cedars-Sinai Medical Center, Los Angeles.
“From this and other data it seems like there is a potential role for vedolizumab as first-line treatment” for selected patients, Dr. Singh said in an interview. “The attraction of vedolizumab is its gut selectivity.”
The American Gastroenterological Association lists vedolizumab as an equal alternative to an anti-TNF agent in its recommended algorithm for treating ulcerative colitis, but vedolizumab is not mentioned in the association’s posted guidance for treating Crohn’s disease. ”Prospective studies are needed” to generate more definitive evidence on how to use vedolizumab, she acknowledged, but added that pediatric gastroenterological societies “need to come up with guidelines on where to place vedolizumab.”Dr. Singh said she discusses with patients and their families the potential risks and benefits of the various treatment options available when inflammatory bowel disease (IBD) doesn’t respond to mesalamine: an anti-TNF agent, an immunomodulator like methotrexate or azathioprine, or vedolizumab. One important issue when deciding which drug class to try first is the cost of treatment and whether it will be covered by insurance.
The combined series included pediatric IBD patients less than 18 years old, with an actual median age of just under 15 years. They had been diagnosed with ulcerative colitis or Crohn’s disease for a median of 3 years. The 30 Crohn’s disease patients had received a median of two anti-TNF agents prior to starting vedolizumab, while the ulcerative colitis patients had received a median of one anti-TNF drug before vedolizumab. Three-quarters of the patients received the adult dose of 300 mg per infusion. A fifth of the patients received a dosage of 6 mg/kg, and the remaining patients received 5 mg/kg.
Although the 24 Crohn’s disease patients followed through 14 weeks of therapy had a 42% remission rate, the remission rate jumped sharply to more than 70% among the 11 patients followed to 30 weeks.
One limitation to vedolizumab is its relatively slow onset of action, slower than anti-TNF agents, which makes vedolizumab less suitable for patients with acute, severe colitis, although short-term improvement of acute colitis can be achieved with a corticosteroid when starting a patient on vedolizumab, Dr. Singh said. A trial currently underway is collecting prospective data on vedolizumab in pediatric IBD patients that could result in pediatric labeling for the drug, she added.
Until those data are available, current experience suggests vedolizumab “is good for ulcerative colitis patients,” she said. “Our data encourage us to continue” offering vedolizumab to selected pediatric IBD patients.
Dr. Singh has been a consultant to Janssen and Prometheus and received grant support from Janssen.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
MONTREAL – Vedolizumab, 2 years out from its entry onto the U.S. market as an option for treating adults with inflammatory bowel disease, also has shown early safety and efficacy in 52 pediatric patients with ulcerative colitis or Crohn’s disease.
Retrospective review of a patients series assembled from three U.S. centers showed 13 (76%) ulcerative colitis patients in remission among 17 treated and followed for 14 weeks, the study’s primary outcome. Among 24 Crohn’s disease patients treated and followed for 14 weeks, 10 (42%) had remissions, Namita Singh, MD, said at the World Congress of Pediatric Gastroenterology, Hepatology, and Nutrition.
Treatment with vedolizumab (Entyvio), an anti-integrin with highly gut-specific activity that limits its systemic effects, was especially potent for the five patients in the series who were naive to treatment with an anti–tumor necrosis factor (TNF) agent. All five patients were in remission at 14 weeks, said Dr. Singh, a pediatric gastroenterologist at Cedars-Sinai Medical Center, Los Angeles.
“From this and other data it seems like there is a potential role for vedolizumab as first-line treatment” for selected patients, Dr. Singh said in an interview. “The attraction of vedolizumab is its gut selectivity.”
The American Gastroenterological Association lists vedolizumab as an equal alternative to an anti-TNF agent in its recommended algorithm for treating ulcerative colitis, but vedolizumab is not mentioned in the association’s posted guidance for treating Crohn’s disease. ”Prospective studies are needed” to generate more definitive evidence on how to use vedolizumab, she acknowledged, but added that pediatric gastroenterological societies “need to come up with guidelines on where to place vedolizumab.”Dr. Singh said she discusses with patients and their families the potential risks and benefits of the various treatment options available when inflammatory bowel disease (IBD) doesn’t respond to mesalamine: an anti-TNF agent, an immunomodulator like methotrexate or azathioprine, or vedolizumab. One important issue when deciding which drug class to try first is the cost of treatment and whether it will be covered by insurance.
The combined series included pediatric IBD patients less than 18 years old, with an actual median age of just under 15 years. They had been diagnosed with ulcerative colitis or Crohn’s disease for a median of 3 years. The 30 Crohn’s disease patients had received a median of two anti-TNF agents prior to starting vedolizumab, while the ulcerative colitis patients had received a median of one anti-TNF drug before vedolizumab. Three-quarters of the patients received the adult dose of 300 mg per infusion. A fifth of the patients received a dosage of 6 mg/kg, and the remaining patients received 5 mg/kg.
Although the 24 Crohn’s disease patients followed through 14 weeks of therapy had a 42% remission rate, the remission rate jumped sharply to more than 70% among the 11 patients followed to 30 weeks.
One limitation to vedolizumab is its relatively slow onset of action, slower than anti-TNF agents, which makes vedolizumab less suitable for patients with acute, severe colitis, although short-term improvement of acute colitis can be achieved with a corticosteroid when starting a patient on vedolizumab, Dr. Singh said. A trial currently underway is collecting prospective data on vedolizumab in pediatric IBD patients that could result in pediatric labeling for the drug, she added.
Until those data are available, current experience suggests vedolizumab “is good for ulcerative colitis patients,” she said. “Our data encourage us to continue” offering vedolizumab to selected pediatric IBD patients.
Dr. Singh has been a consultant to Janssen and Prometheus and received grant support from Janssen.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
MONTREAL – Vedolizumab, 2 years out from its entry onto the U.S. market as an option for treating adults with inflammatory bowel disease, also has shown early safety and efficacy in 52 pediatric patients with ulcerative colitis or Crohn’s disease.
Retrospective review of a patients series assembled from three U.S. centers showed 13 (76%) ulcerative colitis patients in remission among 17 treated and followed for 14 weeks, the study’s primary outcome. Among 24 Crohn’s disease patients treated and followed for 14 weeks, 10 (42%) had remissions, Namita Singh, MD, said at the World Congress of Pediatric Gastroenterology, Hepatology, and Nutrition.
Treatment with vedolizumab (Entyvio), an anti-integrin with highly gut-specific activity that limits its systemic effects, was especially potent for the five patients in the series who were naive to treatment with an anti–tumor necrosis factor (TNF) agent. All five patients were in remission at 14 weeks, said Dr. Singh, a pediatric gastroenterologist at Cedars-Sinai Medical Center, Los Angeles.
“From this and other data it seems like there is a potential role for vedolizumab as first-line treatment” for selected patients, Dr. Singh said in an interview. “The attraction of vedolizumab is its gut selectivity.”
The American Gastroenterological Association lists vedolizumab as an equal alternative to an anti-TNF agent in its recommended algorithm for treating ulcerative colitis, but vedolizumab is not mentioned in the association’s posted guidance for treating Crohn’s disease. ”Prospective studies are needed” to generate more definitive evidence on how to use vedolizumab, she acknowledged, but added that pediatric gastroenterological societies “need to come up with guidelines on where to place vedolizumab.”Dr. Singh said she discusses with patients and their families the potential risks and benefits of the various treatment options available when inflammatory bowel disease (IBD) doesn’t respond to mesalamine: an anti-TNF agent, an immunomodulator like methotrexate or azathioprine, or vedolizumab. One important issue when deciding which drug class to try first is the cost of treatment and whether it will be covered by insurance.
The combined series included pediatric IBD patients less than 18 years old, with an actual median age of just under 15 years. They had been diagnosed with ulcerative colitis or Crohn’s disease for a median of 3 years. The 30 Crohn’s disease patients had received a median of two anti-TNF agents prior to starting vedolizumab, while the ulcerative colitis patients had received a median of one anti-TNF drug before vedolizumab. Three-quarters of the patients received the adult dose of 300 mg per infusion. A fifth of the patients received a dosage of 6 mg/kg, and the remaining patients received 5 mg/kg.
Although the 24 Crohn’s disease patients followed through 14 weeks of therapy had a 42% remission rate, the remission rate jumped sharply to more than 70% among the 11 patients followed to 30 weeks.
One limitation to vedolizumab is its relatively slow onset of action, slower than anti-TNF agents, which makes vedolizumab less suitable for patients with acute, severe colitis, although short-term improvement of acute colitis can be achieved with a corticosteroid when starting a patient on vedolizumab, Dr. Singh said. A trial currently underway is collecting prospective data on vedolizumab in pediatric IBD patients that could result in pediatric labeling for the drug, she added.
Until those data are available, current experience suggests vedolizumab “is good for ulcerative colitis patients,” she said. “Our data encourage us to continue” offering vedolizumab to selected pediatric IBD patients.
Dr. Singh has been a consultant to Janssen and Prometheus and received grant support from Janssen.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
AT WCPGHAN 2016
Key clinical point:
Major finding: Fourteen weeks of treatment with vedolizumab produced remission in 76% of ulcerative colitis patients and 42% of Crohn’s disease patients.
Data source: Retrospective review of 52 pediatric patients with inflammatory bowel disease at seen at any of three U.S. centers.
Disclosures: Dr. Singh has been a consultant to Janssen and Prometheus and received grant support from Janssen.
Pregabalin reduces pain in IBS patients
LAS VEGAS – Pregabalin reduced abdominal pain in patients with irritable bowel syndrome (IBS) and moderate to severe abdominal pain, according to a study presented at the annual meeting of the American College of Gastroenterology.
Antispasmodics and neuromodulators are commonly used to treat such patients, but a significant number don’t respond to these agents, and opioids carry risks of addiction.
The drug makes sense for IBS patients experiencing significant pain, according to Yuri Saito, MD, of the department of medicine and a consultant in the division of gastroenterology at the Mayo Clinic. Rochester, Minn., who presented the research. She noted that pregabalin is approved by the Food and Drug Administration for fibromyalgia, which occurs in many IBS patients. IBS patients also experience frequent anxiety, which can exacerbate symptoms. Pregabalin is not approved for anxiety, but is often prescribed off label. “We thought there were multiple reasons why pregabalin would potentially be effective in IBS,” Dr. Saito said in an interview.
Patients taking pregabalin (n = 41) had lower Bowel Symptom Scale (BSS) pain scores than did placebo (n = 44; 25 vs. 42; P =.008) and lower overall BSS severity scores at weeks 9-12 (26 vs. 42; P =.009). BSS diarrhea scores were lower in pregabalin (17 vs. 32; P = .049), as were bloating BSS scores (29 vs. 44; P =.016).
The study focused on patients with moderate to severe pain, who had experienced three or more pain attacks in a month, and at least one attack during a 2-week screening period. The pregabalin dosage began at 75 mg twice per day and was ramped up to 225 mg twice per day. That dosage was maintained from day 7 through week 12.
Somewhat disappointingly, the researchers found no difference in quality of life measures, but the presence of fibromyalgia may have complicated those measures, Dr. Gerson said.
Thirty-two percent of subjects in the pregabalin arm experienced dizziness, compared with 5% in the placebo group (P =.01). Other side effects included blurred vision (15% vs. 2%; P =.05) and feeling high or tipsy (10% vs. 0%; P =.05).
The results are encouraging and provide an additional treatment option. “I think it’s probably useful, but mainly in patients with diarrhea-prominent IBS,” said Dr. Gerson.
Dr. Saito was more effusive: “The take-home message is that, for patients with moderate to severe pain who have not responded to antispasmodics or other neuromodulators, Pregabalin may be useful as an alternate modality.”
Dr. Saito is an adviser or board member with Commonwealth Labs, Salix, and Synergy. Dr. Gerson is on Allergan’s speakers bureau.
LAS VEGAS – Pregabalin reduced abdominal pain in patients with irritable bowel syndrome (IBS) and moderate to severe abdominal pain, according to a study presented at the annual meeting of the American College of Gastroenterology.
Antispasmodics and neuromodulators are commonly used to treat such patients, but a significant number don’t respond to these agents, and opioids carry risks of addiction.
The drug makes sense for IBS patients experiencing significant pain, according to Yuri Saito, MD, of the department of medicine and a consultant in the division of gastroenterology at the Mayo Clinic. Rochester, Minn., who presented the research. She noted that pregabalin is approved by the Food and Drug Administration for fibromyalgia, which occurs in many IBS patients. IBS patients also experience frequent anxiety, which can exacerbate symptoms. Pregabalin is not approved for anxiety, but is often prescribed off label. “We thought there were multiple reasons why pregabalin would potentially be effective in IBS,” Dr. Saito said in an interview.
Patients taking pregabalin (n = 41) had lower Bowel Symptom Scale (BSS) pain scores than did placebo (n = 44; 25 vs. 42; P =.008) and lower overall BSS severity scores at weeks 9-12 (26 vs. 42; P =.009). BSS diarrhea scores were lower in pregabalin (17 vs. 32; P = .049), as were bloating BSS scores (29 vs. 44; P =.016).
The study focused on patients with moderate to severe pain, who had experienced three or more pain attacks in a month, and at least one attack during a 2-week screening period. The pregabalin dosage began at 75 mg twice per day and was ramped up to 225 mg twice per day. That dosage was maintained from day 7 through week 12.
Somewhat disappointingly, the researchers found no difference in quality of life measures, but the presence of fibromyalgia may have complicated those measures, Dr. Gerson said.
Thirty-two percent of subjects in the pregabalin arm experienced dizziness, compared with 5% in the placebo group (P =.01). Other side effects included blurred vision (15% vs. 2%; P =.05) and feeling high or tipsy (10% vs. 0%; P =.05).
The results are encouraging and provide an additional treatment option. “I think it’s probably useful, but mainly in patients with diarrhea-prominent IBS,” said Dr. Gerson.
Dr. Saito was more effusive: “The take-home message is that, for patients with moderate to severe pain who have not responded to antispasmodics or other neuromodulators, Pregabalin may be useful as an alternate modality.”
Dr. Saito is an adviser or board member with Commonwealth Labs, Salix, and Synergy. Dr. Gerson is on Allergan’s speakers bureau.
LAS VEGAS – Pregabalin reduced abdominal pain in patients with irritable bowel syndrome (IBS) and moderate to severe abdominal pain, according to a study presented at the annual meeting of the American College of Gastroenterology.
Antispasmodics and neuromodulators are commonly used to treat such patients, but a significant number don’t respond to these agents, and opioids carry risks of addiction.
The drug makes sense for IBS patients experiencing significant pain, according to Yuri Saito, MD, of the department of medicine and a consultant in the division of gastroenterology at the Mayo Clinic. Rochester, Minn., who presented the research. She noted that pregabalin is approved by the Food and Drug Administration for fibromyalgia, which occurs in many IBS patients. IBS patients also experience frequent anxiety, which can exacerbate symptoms. Pregabalin is not approved for anxiety, but is often prescribed off label. “We thought there were multiple reasons why pregabalin would potentially be effective in IBS,” Dr. Saito said in an interview.
Patients taking pregabalin (n = 41) had lower Bowel Symptom Scale (BSS) pain scores than did placebo (n = 44; 25 vs. 42; P =.008) and lower overall BSS severity scores at weeks 9-12 (26 vs. 42; P =.009). BSS diarrhea scores were lower in pregabalin (17 vs. 32; P = .049), as were bloating BSS scores (29 vs. 44; P =.016).
The study focused on patients with moderate to severe pain, who had experienced three or more pain attacks in a month, and at least one attack during a 2-week screening period. The pregabalin dosage began at 75 mg twice per day and was ramped up to 225 mg twice per day. That dosage was maintained from day 7 through week 12.
Somewhat disappointingly, the researchers found no difference in quality of life measures, but the presence of fibromyalgia may have complicated those measures, Dr. Gerson said.
Thirty-two percent of subjects in the pregabalin arm experienced dizziness, compared with 5% in the placebo group (P =.01). Other side effects included blurred vision (15% vs. 2%; P =.05) and feeling high or tipsy (10% vs. 0%; P =.05).
The results are encouraging and provide an additional treatment option. “I think it’s probably useful, but mainly in patients with diarrhea-prominent IBS,” said Dr. Gerson.
Dr. Saito was more effusive: “The take-home message is that, for patients with moderate to severe pain who have not responded to antispasmodics or other neuromodulators, Pregabalin may be useful as an alternate modality.”
Dr. Saito is an adviser or board member with Commonwealth Labs, Salix, and Synergy. Dr. Gerson is on Allergan’s speakers bureau.
AT ACG 2016
Key clinical point:
Major finding: In a pilot study, pregabalin reduced pain scores and diarrhea in patients with IBS and moderate to severe abdominal pain.
Data source: A randomized, placebo controlled clinical trial.
Disclosures: The study was funded by Pfizer. Dr. Saito is an adviser or board member with Commonwealth Labs, Salix, and Synergy. Dr. Gerson is on Allergan’s speakers bureau.
Tryptase gene variant linked to GI, joint, and skin symptoms
Researchers have identified a genetic variant associated with inherited elevated basal serum tryptase levels and linked to a distinct group of comorbid multisystem complaints.
These features, including cutaneous flushing, certain chronic pain disorders, autonomic dysfunction, and gastrointestinal dysmotility, have been reported in association with genetic disorders or joint hypermobility syndromes such as Ehlers-Danlos syndrome type III (hypermobility type, EDS III) and often follow a dominant inheritance pattern in affected families, providing a reason to look into a genetic basis for these patient characteristics, according to Jonathan J. Lyons, MD, of the National Institute of Allergy and Infectious Diseases, and his coauthors. The researchers reported their findings Oct. 17 in Nature Genetics.
The researchers recruited 96 individuals from 35 families with a syndrome of elevated basal serum tryptase levels and multiple comorbid symptoms following an autosomal dominant pattern of inheritance.
These symptoms included gastrointestinal dysmotility such as irritable bowel syndrome or chronic gastroesophageal reflux, connective tissue abnormalities such as joint hypermobility, congenital skeletal abnormalities, retained primary dentition, symptoms suggestive of autonomic dysfunction such as postural orthostatic tachycardia syndrome, and elevated composite autonomic symptom scores. Other symptoms included recurrent cutaneous flushing and pruritus – often associated with urticaria and complaints of sleep disruption – and systemic reaction to stinging insects.
Using exome and genome sequencing followed by linkage analysis, researchers identified duplications and triplications within the TPSAB1 gene encoding alpha-tryptase (Nat Genet. 2016 Oct 17. doi: 10.1038/ng.3696). Further analysis found elevated alpha-tryptase/beta-tryptase ratios among affected family members and suggested that multiple copies of the alpha-tryptase sequence were inherited together.
To confirm the finding, researchers examined genetic data from a cohort of healthy unrelated volunteers in the National Human Genome Research Institute ClinSeq cohort, which identified 125 samples with partially enriched duplication of alpha tryptase–encoding sequence using a common haplotype.
Of these, three individuals had single-allele duplications of the alpha tryptase–encoding sequence and also presented with similar symptoms to the original cohort: cutaneous flushing, itching, or hives, systemic venom reactions, irritable bowel syndrome, retained primary dentition, and elevated autonomic symptom scores.
“We have found that this phenotype is most frequently inherited in an autosomal dominant manner and that, when this occurs, it is exclusively associated with increased copy number on a single allele of alpha tryptase–encoding sequence in the TPSAB1 gene, a genetic trait we have termed hereditary alpha-tryptasemia,” the researchers reported. “The families studied in our initial cohort likely represent the most severe phenotypes among individuals affected with hereditary alpha-tryptasemia, owing in part to the lack of detection of triplication of alpha tryptase–encoding sequence in unselected populations, which we have tentatively designated as hereditary alpha-tryptasemia syndrome.”
The authors suggested that part of the clinical presentation of this syndrome included symptoms that may be associated clinically with mast cell mediator release. In the context of elevated basal serum tryptase levels, this might prompt a doctor to investigate for clonal mast cell disease, which would include bone-marrow biopsy.
However, given that such an investigation would be challenging, and given that elevated tryptase levels are not uncommon in the generally population, they suggested tryptase genotyping may be warranted.
The study was supported by the National Institute of Allergy and Infectious Diseases, the ARTrust/the Mastocytosis Society, and the National Human Genome Research Institute. One author declared royalties associated with the tryptase UniCAP assay, and consulting fees from Genentech. Another author declared an advisory position and royalties from private industry.
Researchers have identified a genetic variant associated with inherited elevated basal serum tryptase levels and linked to a distinct group of comorbid multisystem complaints.
These features, including cutaneous flushing, certain chronic pain disorders, autonomic dysfunction, and gastrointestinal dysmotility, have been reported in association with genetic disorders or joint hypermobility syndromes such as Ehlers-Danlos syndrome type III (hypermobility type, EDS III) and often follow a dominant inheritance pattern in affected families, providing a reason to look into a genetic basis for these patient characteristics, according to Jonathan J. Lyons, MD, of the National Institute of Allergy and Infectious Diseases, and his coauthors. The researchers reported their findings Oct. 17 in Nature Genetics.
The researchers recruited 96 individuals from 35 families with a syndrome of elevated basal serum tryptase levels and multiple comorbid symptoms following an autosomal dominant pattern of inheritance.
These symptoms included gastrointestinal dysmotility such as irritable bowel syndrome or chronic gastroesophageal reflux, connective tissue abnormalities such as joint hypermobility, congenital skeletal abnormalities, retained primary dentition, symptoms suggestive of autonomic dysfunction such as postural orthostatic tachycardia syndrome, and elevated composite autonomic symptom scores. Other symptoms included recurrent cutaneous flushing and pruritus – often associated with urticaria and complaints of sleep disruption – and systemic reaction to stinging insects.
Using exome and genome sequencing followed by linkage analysis, researchers identified duplications and triplications within the TPSAB1 gene encoding alpha-tryptase (Nat Genet. 2016 Oct 17. doi: 10.1038/ng.3696). Further analysis found elevated alpha-tryptase/beta-tryptase ratios among affected family members and suggested that multiple copies of the alpha-tryptase sequence were inherited together.
To confirm the finding, researchers examined genetic data from a cohort of healthy unrelated volunteers in the National Human Genome Research Institute ClinSeq cohort, which identified 125 samples with partially enriched duplication of alpha tryptase–encoding sequence using a common haplotype.
Of these, three individuals had single-allele duplications of the alpha tryptase–encoding sequence and also presented with similar symptoms to the original cohort: cutaneous flushing, itching, or hives, systemic venom reactions, irritable bowel syndrome, retained primary dentition, and elevated autonomic symptom scores.
“We have found that this phenotype is most frequently inherited in an autosomal dominant manner and that, when this occurs, it is exclusively associated with increased copy number on a single allele of alpha tryptase–encoding sequence in the TPSAB1 gene, a genetic trait we have termed hereditary alpha-tryptasemia,” the researchers reported. “The families studied in our initial cohort likely represent the most severe phenotypes among individuals affected with hereditary alpha-tryptasemia, owing in part to the lack of detection of triplication of alpha tryptase–encoding sequence in unselected populations, which we have tentatively designated as hereditary alpha-tryptasemia syndrome.”
The authors suggested that part of the clinical presentation of this syndrome included symptoms that may be associated clinically with mast cell mediator release. In the context of elevated basal serum tryptase levels, this might prompt a doctor to investigate for clonal mast cell disease, which would include bone-marrow biopsy.
However, given that such an investigation would be challenging, and given that elevated tryptase levels are not uncommon in the generally population, they suggested tryptase genotyping may be warranted.
The study was supported by the National Institute of Allergy and Infectious Diseases, the ARTrust/the Mastocytosis Society, and the National Human Genome Research Institute. One author declared royalties associated with the tryptase UniCAP assay, and consulting fees from Genentech. Another author declared an advisory position and royalties from private industry.
Researchers have identified a genetic variant associated with inherited elevated basal serum tryptase levels and linked to a distinct group of comorbid multisystem complaints.
These features, including cutaneous flushing, certain chronic pain disorders, autonomic dysfunction, and gastrointestinal dysmotility, have been reported in association with genetic disorders or joint hypermobility syndromes such as Ehlers-Danlos syndrome type III (hypermobility type, EDS III) and often follow a dominant inheritance pattern in affected families, providing a reason to look into a genetic basis for these patient characteristics, according to Jonathan J. Lyons, MD, of the National Institute of Allergy and Infectious Diseases, and his coauthors. The researchers reported their findings Oct. 17 in Nature Genetics.
The researchers recruited 96 individuals from 35 families with a syndrome of elevated basal serum tryptase levels and multiple comorbid symptoms following an autosomal dominant pattern of inheritance.
These symptoms included gastrointestinal dysmotility such as irritable bowel syndrome or chronic gastroesophageal reflux, connective tissue abnormalities such as joint hypermobility, congenital skeletal abnormalities, retained primary dentition, symptoms suggestive of autonomic dysfunction such as postural orthostatic tachycardia syndrome, and elevated composite autonomic symptom scores. Other symptoms included recurrent cutaneous flushing and pruritus – often associated with urticaria and complaints of sleep disruption – and systemic reaction to stinging insects.
Using exome and genome sequencing followed by linkage analysis, researchers identified duplications and triplications within the TPSAB1 gene encoding alpha-tryptase (Nat Genet. 2016 Oct 17. doi: 10.1038/ng.3696). Further analysis found elevated alpha-tryptase/beta-tryptase ratios among affected family members and suggested that multiple copies of the alpha-tryptase sequence were inherited together.
To confirm the finding, researchers examined genetic data from a cohort of healthy unrelated volunteers in the National Human Genome Research Institute ClinSeq cohort, which identified 125 samples with partially enriched duplication of alpha tryptase–encoding sequence using a common haplotype.
Of these, three individuals had single-allele duplications of the alpha tryptase–encoding sequence and also presented with similar symptoms to the original cohort: cutaneous flushing, itching, or hives, systemic venom reactions, irritable bowel syndrome, retained primary dentition, and elevated autonomic symptom scores.
“We have found that this phenotype is most frequently inherited in an autosomal dominant manner and that, when this occurs, it is exclusively associated with increased copy number on a single allele of alpha tryptase–encoding sequence in the TPSAB1 gene, a genetic trait we have termed hereditary alpha-tryptasemia,” the researchers reported. “The families studied in our initial cohort likely represent the most severe phenotypes among individuals affected with hereditary alpha-tryptasemia, owing in part to the lack of detection of triplication of alpha tryptase–encoding sequence in unselected populations, which we have tentatively designated as hereditary alpha-tryptasemia syndrome.”
The authors suggested that part of the clinical presentation of this syndrome included symptoms that may be associated clinically with mast cell mediator release. In the context of elevated basal serum tryptase levels, this might prompt a doctor to investigate for clonal mast cell disease, which would include bone-marrow biopsy.
However, given that such an investigation would be challenging, and given that elevated tryptase levels are not uncommon in the generally population, they suggested tryptase genotyping may be warranted.
The study was supported by the National Institute of Allergy and Infectious Diseases, the ARTrust/the Mastocytosis Society, and the National Human Genome Research Institute. One author declared royalties associated with the tryptase UniCAP assay, and consulting fees from Genentech. Another author declared an advisory position and royalties from private industry.
FROM NATURE GENETICS
Key clinical point:
Major finding: Increased copy number on a single allele of alpha tryptase–encoding sequence in the TPSAB1 gene is associated with elevated basal serum tryptase and a collection of symptoms including irritable bowel syndrome, joint hypermobility, and autonomic dysfunction.
Data source: Study of 96 individuals from 35 families with a syndrome of elevated basal serum tryptase levels and multiple comorbid symptoms.
Disclosures: The study was supported by the National Institute of Allergy and Infectious Diseases, the ARTrust/the Mastocytosis Society, and the National Human Genome Research Institute. One author declared royalties associated with the tryptase UniCAP assay, and consulting fees from Genentech. Another author declared an advisory position and royalties from private industry.
UC patients who failed infliximab gain from vedolizumab
Vedolizumab (Entyvio) was much more effective than placebo as a treatment for ulcerative colitis (UC) in a range of patients, researchers found in a follow-up analysis of trial data.
Patients who’d previously failed TNF-antagonist therapy with infliximab, however, fared worse at 6 and 52 weeks than those who hadn’t tried the treatment. Still, Brian G. Feagan, MD, of Western University, London, Ont., and fellow researchers report that vedolizumab had a “consistent benefit” in patients with moderate to severe active UC, regardless of whether their experience with TNF-antagonist therapy was poor or nonexistent. Remission rates at 52 weeks reached as high as 46.9%.
According to background information in the study, tumor necrosis factor–alpha (TNF) antagonist therapy has significantly improved treatment for ulcerative colitis, but many patients fail to respond. There is also a risk of infection.
The new study, published online Sept. 14 in Clinical Gastroenterology and Hepatology (2016. doi: 10.1016/j.cgh.2016.08.044), offers prespecified exploratory and post hoc analyses of vedolizumab vs. placebo in sets of UC patients from a phase III trial.
In a blinded group, 374 patients were randomly assigned to receive intravenous vedolizumab or placebo on days 1 and 15; 521 other patients in an open-label group received intravenous vedolizumab in the same fashion.
Patients in both groups who responded to vedolizumab at 6 weeks were randomly assigned to one of three groups: They continued vedolizumab every 8 weeks or every 4 weeks, or they received a placebo at week 6 for as many as 46 weeks. Patients who initially received the placebo either continued taking it over the maintenance period or stopped; those who didn’t respond to vedolizumab continued to get it every 4 weeks through week 52.
The new study focuses on 464 patients had not taken a TNF antagonist previously and 367 who’d failed TNF-antagonist therapy.
At week 6, 53.1% of the vedolizumab patients who hadn’t tried a TNF antagonist saw a clinical response, compared with 26.3% of counterparts who took a placebo (absolute difference, 26.4%; 95% confidence interval, 12.4-40.4; relative risk, 2.0; 95% CI, 1.3-3.0).
Among patients who had failed TNF antagonists, 39.0% saw a clinical response on vedolizumab at 6 weeks, and 20.6% saw one on placebo (absolute difference, 18.1%; 95% CI, 2.8-33.5; RR, 1.9; 95% CI, 1.1-3.2.). All those who’d failed TNF antagonists had taken infliximab because it was the only drug of its type available at the time of enrollment.
At week 52, the percentage of TNF antagonist–naive patients who were in remission was 46.9% in those on vedolizumab and 19.0% in those on placebo (absolute difference: 28.0%; 95% CI, 14.9-41.1; RR, 2.5; 95% CI, 1.5-4.0.) Among those who’d failed TNF-antagonist therapy, 36.1% of those on vedolizumab reached remission, and just 5.3% of those on placebo did (absolute difference, 29.5%; 95% CI, 12.8-46.1; RR, 6.6; 95% CI, 1.7-26.5.)
“These analyses show that vedolizumab had a consistent benefit for inducing and maintaining clinical response and remission in both TNF-naive and TNF-failure patients with moderately to severely active UC,” the authors write. “Further, no increased rates of serious infections were observed with vedolizumab treatment relative to placebo in either subgroup.”
The study authors caution against comparing the efficacy statistics in this study to the results of trials of TNF antagonists. While it’s “alluring” to make the comparison, they write, “We would caution against using such an approach to determine relative efficacy, given the confounding effects of differences in patient populations, outcome definitions, and, notably, trial design.”
They note that a study comparing the safety and efficacy of vedolizumab to adalimumab (Humira) in ulcerative colitis is underway (NCT02497469) – it’s now recruiting patients – and “the results of such studies will be critical in informing payers regarding the relative value of these agents.”
What’s next? The researchers note that TNF-antagonist alternatives to infliximab now exist. “Randomized trials should be performed comparing vedolizumab to the use of a second TNF antagonist in TNF -antagonist failure patients with adequate serum drug concentrations,” they write.
Millennium Pharmaceuticals (Takeda Pharmaceuticals) funded the clinical studies. Some of the study authors are employees of Takeda. Other study authors have received support from Takeda, Millennium, or both.
Vedolizumab (Entyvio) was much more effective than placebo as a treatment for ulcerative colitis (UC) in a range of patients, researchers found in a follow-up analysis of trial data.
Patients who’d previously failed TNF-antagonist therapy with infliximab, however, fared worse at 6 and 52 weeks than those who hadn’t tried the treatment. Still, Brian G. Feagan, MD, of Western University, London, Ont., and fellow researchers report that vedolizumab had a “consistent benefit” in patients with moderate to severe active UC, regardless of whether their experience with TNF-antagonist therapy was poor or nonexistent. Remission rates at 52 weeks reached as high as 46.9%.
According to background information in the study, tumor necrosis factor–alpha (TNF) antagonist therapy has significantly improved treatment for ulcerative colitis, but many patients fail to respond. There is also a risk of infection.
The new study, published online Sept. 14 in Clinical Gastroenterology and Hepatology (2016. doi: 10.1016/j.cgh.2016.08.044), offers prespecified exploratory and post hoc analyses of vedolizumab vs. placebo in sets of UC patients from a phase III trial.
In a blinded group, 374 patients were randomly assigned to receive intravenous vedolizumab or placebo on days 1 and 15; 521 other patients in an open-label group received intravenous vedolizumab in the same fashion.
Patients in both groups who responded to vedolizumab at 6 weeks were randomly assigned to one of three groups: They continued vedolizumab every 8 weeks or every 4 weeks, or they received a placebo at week 6 for as many as 46 weeks. Patients who initially received the placebo either continued taking it over the maintenance period or stopped; those who didn’t respond to vedolizumab continued to get it every 4 weeks through week 52.
The new study focuses on 464 patients had not taken a TNF antagonist previously and 367 who’d failed TNF-antagonist therapy.
At week 6, 53.1% of the vedolizumab patients who hadn’t tried a TNF antagonist saw a clinical response, compared with 26.3% of counterparts who took a placebo (absolute difference, 26.4%; 95% confidence interval, 12.4-40.4; relative risk, 2.0; 95% CI, 1.3-3.0).
Among patients who had failed TNF antagonists, 39.0% saw a clinical response on vedolizumab at 6 weeks, and 20.6% saw one on placebo (absolute difference, 18.1%; 95% CI, 2.8-33.5; RR, 1.9; 95% CI, 1.1-3.2.). All those who’d failed TNF antagonists had taken infliximab because it was the only drug of its type available at the time of enrollment.
At week 52, the percentage of TNF antagonist–naive patients who were in remission was 46.9% in those on vedolizumab and 19.0% in those on placebo (absolute difference: 28.0%; 95% CI, 14.9-41.1; RR, 2.5; 95% CI, 1.5-4.0.) Among those who’d failed TNF-antagonist therapy, 36.1% of those on vedolizumab reached remission, and just 5.3% of those on placebo did (absolute difference, 29.5%; 95% CI, 12.8-46.1; RR, 6.6; 95% CI, 1.7-26.5.)
“These analyses show that vedolizumab had a consistent benefit for inducing and maintaining clinical response and remission in both TNF-naive and TNF-failure patients with moderately to severely active UC,” the authors write. “Further, no increased rates of serious infections were observed with vedolizumab treatment relative to placebo in either subgroup.”
The study authors caution against comparing the efficacy statistics in this study to the results of trials of TNF antagonists. While it’s “alluring” to make the comparison, they write, “We would caution against using such an approach to determine relative efficacy, given the confounding effects of differences in patient populations, outcome definitions, and, notably, trial design.”
They note that a study comparing the safety and efficacy of vedolizumab to adalimumab (Humira) in ulcerative colitis is underway (NCT02497469) – it’s now recruiting patients – and “the results of such studies will be critical in informing payers regarding the relative value of these agents.”
What’s next? The researchers note that TNF-antagonist alternatives to infliximab now exist. “Randomized trials should be performed comparing vedolizumab to the use of a second TNF antagonist in TNF -antagonist failure patients with adequate serum drug concentrations,” they write.
Millennium Pharmaceuticals (Takeda Pharmaceuticals) funded the clinical studies. Some of the study authors are employees of Takeda. Other study authors have received support from Takeda, Millennium, or both.
Vedolizumab (Entyvio) was much more effective than placebo as a treatment for ulcerative colitis (UC) in a range of patients, researchers found in a follow-up analysis of trial data.
Patients who’d previously failed TNF-antagonist therapy with infliximab, however, fared worse at 6 and 52 weeks than those who hadn’t tried the treatment. Still, Brian G. Feagan, MD, of Western University, London, Ont., and fellow researchers report that vedolizumab had a “consistent benefit” in patients with moderate to severe active UC, regardless of whether their experience with TNF-antagonist therapy was poor or nonexistent. Remission rates at 52 weeks reached as high as 46.9%.
According to background information in the study, tumor necrosis factor–alpha (TNF) antagonist therapy has significantly improved treatment for ulcerative colitis, but many patients fail to respond. There is also a risk of infection.
The new study, published online Sept. 14 in Clinical Gastroenterology and Hepatology (2016. doi: 10.1016/j.cgh.2016.08.044), offers prespecified exploratory and post hoc analyses of vedolizumab vs. placebo in sets of UC patients from a phase III trial.
In a blinded group, 374 patients were randomly assigned to receive intravenous vedolizumab or placebo on days 1 and 15; 521 other patients in an open-label group received intravenous vedolizumab in the same fashion.
Patients in both groups who responded to vedolizumab at 6 weeks were randomly assigned to one of three groups: They continued vedolizumab every 8 weeks or every 4 weeks, or they received a placebo at week 6 for as many as 46 weeks. Patients who initially received the placebo either continued taking it over the maintenance period or stopped; those who didn’t respond to vedolizumab continued to get it every 4 weeks through week 52.
The new study focuses on 464 patients had not taken a TNF antagonist previously and 367 who’d failed TNF-antagonist therapy.
At week 6, 53.1% of the vedolizumab patients who hadn’t tried a TNF antagonist saw a clinical response, compared with 26.3% of counterparts who took a placebo (absolute difference, 26.4%; 95% confidence interval, 12.4-40.4; relative risk, 2.0; 95% CI, 1.3-3.0).
Among patients who had failed TNF antagonists, 39.0% saw a clinical response on vedolizumab at 6 weeks, and 20.6% saw one on placebo (absolute difference, 18.1%; 95% CI, 2.8-33.5; RR, 1.9; 95% CI, 1.1-3.2.). All those who’d failed TNF antagonists had taken infliximab because it was the only drug of its type available at the time of enrollment.
At week 52, the percentage of TNF antagonist–naive patients who were in remission was 46.9% in those on vedolizumab and 19.0% in those on placebo (absolute difference: 28.0%; 95% CI, 14.9-41.1; RR, 2.5; 95% CI, 1.5-4.0.) Among those who’d failed TNF-antagonist therapy, 36.1% of those on vedolizumab reached remission, and just 5.3% of those on placebo did (absolute difference, 29.5%; 95% CI, 12.8-46.1; RR, 6.6; 95% CI, 1.7-26.5.)
“These analyses show that vedolizumab had a consistent benefit for inducing and maintaining clinical response and remission in both TNF-naive and TNF-failure patients with moderately to severely active UC,” the authors write. “Further, no increased rates of serious infections were observed with vedolizumab treatment relative to placebo in either subgroup.”
The study authors caution against comparing the efficacy statistics in this study to the results of trials of TNF antagonists. While it’s “alluring” to make the comparison, they write, “We would caution against using such an approach to determine relative efficacy, given the confounding effects of differences in patient populations, outcome definitions, and, notably, trial design.”
They note that a study comparing the safety and efficacy of vedolizumab to adalimumab (Humira) in ulcerative colitis is underway (NCT02497469) – it’s now recruiting patients – and “the results of such studies will be critical in informing payers regarding the relative value of these agents.”
What’s next? The researchers note that TNF-antagonist alternatives to infliximab now exist. “Randomized trials should be performed comparing vedolizumab to the use of a second TNF antagonist in TNF -antagonist failure patients with adequate serum drug concentrations,” they write.
Millennium Pharmaceuticals (Takeda Pharmaceuticals) funded the clinical studies. Some of the study authors are employees of Takeda. Other study authors have received support from Takeda, Millennium, or both.
Key clinical point: , but they didn’t fare as well as those who never tried TNF-antagonist therapy.
Major finding: Of UC patients who took vedolizumab for 52 weeks, 46.9% of those who’d never tried TNF-antagonist therapy were in remission, compared with 36.1% of those who failed infliximab.
Data source: Post hoc analysis of 831 patients with moderate to severe UC in a multicenter, randomized, phase III trial of vedolizumab vs. placebo.
Disclosures: Millennium Pharmaceuticals (Takeda Pharmaceuticals) funded the clinical studies. Some of the study authors are employees of Takeda. Other study authors have received support from Takeda, Millennium, or both.