User login
FDA approves ustekinumab for adult Crohn’s disease
The Food and Drug Administration has approved ustekinumab (Stelara) for the treatment of moderately to severely active Crohn’s disease in certain adults.
Specifically, the new approval for the human monoclonal antibody, which was previously approved for the treatment of psoriasis and psoriatic arthritis, is for Crohn’s disease patients aged 18 years or older who fail or cannot tolerate treatment with immunomodulators or corticosteroids but who never failed treatment with a tumor necrosis factor (TNF) blocker, or who fail or cannot tolerate treatment with one or more TNF blockers, according to a statement from the drug’s maker, Janssen Biotech.
“Crohn’s disease is a complex condition to treat, and not all therapies work for every patient. The FDA approval of Stelara represents an important advancement in treating patients with Crohn’s disease, as this therapy offers an alternate mechanism of action to induce and maintain clinical remission over time,” study investigator William J. Sandborn, MD, of the University of California, San Diego, said in the statement.
AGA Resource
AGA offers an IBD Clinical Service Line that provides tools to help you become more efficient, understand quality standards and improve the process of care for patients. Learn more at http://www.gastro.org/patient-care/conditions-diseases/ibd.
The Food and Drug Administration has approved ustekinumab (Stelara) for the treatment of moderately to severely active Crohn’s disease in certain adults.
Specifically, the new approval for the human monoclonal antibody, which was previously approved for the treatment of psoriasis and psoriatic arthritis, is for Crohn’s disease patients aged 18 years or older who fail or cannot tolerate treatment with immunomodulators or corticosteroids but who never failed treatment with a tumor necrosis factor (TNF) blocker, or who fail or cannot tolerate treatment with one or more TNF blockers, according to a statement from the drug’s maker, Janssen Biotech.
“Crohn’s disease is a complex condition to treat, and not all therapies work for every patient. The FDA approval of Stelara represents an important advancement in treating patients with Crohn’s disease, as this therapy offers an alternate mechanism of action to induce and maintain clinical remission over time,” study investigator William J. Sandborn, MD, of the University of California, San Diego, said in the statement.
AGA Resource
AGA offers an IBD Clinical Service Line that provides tools to help you become more efficient, understand quality standards and improve the process of care for patients. Learn more at http://www.gastro.org/patient-care/conditions-diseases/ibd.
The Food and Drug Administration has approved ustekinumab (Stelara) for the treatment of moderately to severely active Crohn’s disease in certain adults.
Specifically, the new approval for the human monoclonal antibody, which was previously approved for the treatment of psoriasis and psoriatic arthritis, is for Crohn’s disease patients aged 18 years or older who fail or cannot tolerate treatment with immunomodulators or corticosteroids but who never failed treatment with a tumor necrosis factor (TNF) blocker, or who fail or cannot tolerate treatment with one or more TNF blockers, according to a statement from the drug’s maker, Janssen Biotech.
“Crohn’s disease is a complex condition to treat, and not all therapies work for every patient. The FDA approval of Stelara represents an important advancement in treating patients with Crohn’s disease, as this therapy offers an alternate mechanism of action to induce and maintain clinical remission over time,” study investigator William J. Sandborn, MD, of the University of California, San Diego, said in the statement.
AGA Resource
AGA offers an IBD Clinical Service Line that provides tools to help you become more efficient, understand quality standards and improve the process of care for patients. Learn more at http://www.gastro.org/patient-care/conditions-diseases/ibd.
Biologics did not up risk of serious infections in IBD
Treating inflammatory bowel disease with biologic therapies increases the risk of opportunistic infections but not the risk of serious infections, based on a systematic review and meta-analysis reported in the October issue of Clinical Gastroenterology and Hepatology.
Contrary to common belief, infection risk seemed similar with the integrin and anti–tumor necrosis factor classes, said Stefanos Bonovas, MD, MSc, PhD, of Humanitas Clinical and Research Center, Milan, with his associates. Clinicians and patients can use these findings to better weigh the risks and benefits of biologic therapies for IBD, although “studies in real-world settings, national and international registries, and clinical audits may serve as complementary data sources to further assess biologic treatments’ comparative and long-term safety profiles,” the researchers added.
Biologics can effectively manage IBD but raise concerns about infection and malignancy. To examine these risks in adults with IBD, the researchers systematically searched PubMed, Embase, Scopus, the Cochrane IBD Group Specialized Trials Register, the World Health Organization International Clinical Trials Registry Platform, and clinicaltrials.gov through March 2016 for randomized, placebo-controlled or head-to-head trials of approved IBD therapies, including adalimumab, certolizumab, golimumab, infliximab, natalizumab, and vedolizumab. After excluding systematic reviews, uncontrolled trials, and secondary analyses, 49 trials of 14,590 patients remained for meta-analysis, the investigators said (Clin Gastroenterol Hepatol. 2016 May 4. doi: 10.1016/j.cgh.2016.04.039).
Biologic therapy conferred a “moderate increase” in the likelihood of any infection, with an odds ratio of 1.19 when compared with patients who did not receive biologics (95% confidence interval, 1.10-1.29), the researchers said. The risk of opportunistic infections was somewhat higher (OR, 1.90; 95% CI, 1.21-3.01). However, biologic therapy did not significantly heighten the risk of serious infections, which most studies defined as infections leading to hospitalization, intravenous antibiotic treatment, or death (OR, 0.89; 95% CI, 0.71-1.12). “On the contrary, biologics appeared to significantly reduce risk of serious infections in studies with low risk of bias (OR, 0.56; 95% CI, 0.35-0.90),” the investigators wrote. They did not find an increased risk of malignancy with biologic therapy (OR, 0.90; 95% CI, 0.54-1.50), “but [these] data were insufficient in terms of exposure and follow-up times,” they added.
Comparisons of individual agents and classes also did not reveal any significant associations with the risk of infection, the investigators noted. They did acknowledge several limitations. None of the trials were head-to-head comparisons between biologics, and many were industry funded. Estimates of comparative harm were based on indirect comparisons, and therefore merited cautious interpretation. Because the trials were carried out for regulatory purposes, they enrolled highly selected and homogeneous cohorts of IBD patients, leading to underrepresentation of high-risk and elderly individuals, the researchers noted. “Finally, the exposure and follow-up times were up to 24 months, a time period that is considered sufficient when analyzing infectious adverse effects but wholly insufficient for cancer outcomes. With this in mind, we must look to large register-based cohort studies to enhance our understanding of the biologics-cancer association, despite the biases inherent in observational study designs.”
Dr. Bonovas had no disclosures, while three coauthors reported relationships with a number of pharmaceutical companies.
Treating inflammatory bowel disease with biologic therapies increases the risk of opportunistic infections but not the risk of serious infections, based on a systematic review and meta-analysis reported in the October issue of Clinical Gastroenterology and Hepatology.
Contrary to common belief, infection risk seemed similar with the integrin and anti–tumor necrosis factor classes, said Stefanos Bonovas, MD, MSc, PhD, of Humanitas Clinical and Research Center, Milan, with his associates. Clinicians and patients can use these findings to better weigh the risks and benefits of biologic therapies for IBD, although “studies in real-world settings, national and international registries, and clinical audits may serve as complementary data sources to further assess biologic treatments’ comparative and long-term safety profiles,” the researchers added.
Biologics can effectively manage IBD but raise concerns about infection and malignancy. To examine these risks in adults with IBD, the researchers systematically searched PubMed, Embase, Scopus, the Cochrane IBD Group Specialized Trials Register, the World Health Organization International Clinical Trials Registry Platform, and clinicaltrials.gov through March 2016 for randomized, placebo-controlled or head-to-head trials of approved IBD therapies, including adalimumab, certolizumab, golimumab, infliximab, natalizumab, and vedolizumab. After excluding systematic reviews, uncontrolled trials, and secondary analyses, 49 trials of 14,590 patients remained for meta-analysis, the investigators said (Clin Gastroenterol Hepatol. 2016 May 4. doi: 10.1016/j.cgh.2016.04.039).
Biologic therapy conferred a “moderate increase” in the likelihood of any infection, with an odds ratio of 1.19 when compared with patients who did not receive biologics (95% confidence interval, 1.10-1.29), the researchers said. The risk of opportunistic infections was somewhat higher (OR, 1.90; 95% CI, 1.21-3.01). However, biologic therapy did not significantly heighten the risk of serious infections, which most studies defined as infections leading to hospitalization, intravenous antibiotic treatment, or death (OR, 0.89; 95% CI, 0.71-1.12). “On the contrary, biologics appeared to significantly reduce risk of serious infections in studies with low risk of bias (OR, 0.56; 95% CI, 0.35-0.90),” the investigators wrote. They did not find an increased risk of malignancy with biologic therapy (OR, 0.90; 95% CI, 0.54-1.50), “but [these] data were insufficient in terms of exposure and follow-up times,” they added.
Comparisons of individual agents and classes also did not reveal any significant associations with the risk of infection, the investigators noted. They did acknowledge several limitations. None of the trials were head-to-head comparisons between biologics, and many were industry funded. Estimates of comparative harm were based on indirect comparisons, and therefore merited cautious interpretation. Because the trials were carried out for regulatory purposes, they enrolled highly selected and homogeneous cohorts of IBD patients, leading to underrepresentation of high-risk and elderly individuals, the researchers noted. “Finally, the exposure and follow-up times were up to 24 months, a time period that is considered sufficient when analyzing infectious adverse effects but wholly insufficient for cancer outcomes. With this in mind, we must look to large register-based cohort studies to enhance our understanding of the biologics-cancer association, despite the biases inherent in observational study designs.”
Dr. Bonovas had no disclosures, while three coauthors reported relationships with a number of pharmaceutical companies.
Treating inflammatory bowel disease with biologic therapies increases the risk of opportunistic infections but not the risk of serious infections, based on a systematic review and meta-analysis reported in the October issue of Clinical Gastroenterology and Hepatology.
Contrary to common belief, infection risk seemed similar with the integrin and anti–tumor necrosis factor classes, said Stefanos Bonovas, MD, MSc, PhD, of Humanitas Clinical and Research Center, Milan, with his associates. Clinicians and patients can use these findings to better weigh the risks and benefits of biologic therapies for IBD, although “studies in real-world settings, national and international registries, and clinical audits may serve as complementary data sources to further assess biologic treatments’ comparative and long-term safety profiles,” the researchers added.
Biologics can effectively manage IBD but raise concerns about infection and malignancy. To examine these risks in adults with IBD, the researchers systematically searched PubMed, Embase, Scopus, the Cochrane IBD Group Specialized Trials Register, the World Health Organization International Clinical Trials Registry Platform, and clinicaltrials.gov through March 2016 for randomized, placebo-controlled or head-to-head trials of approved IBD therapies, including adalimumab, certolizumab, golimumab, infliximab, natalizumab, and vedolizumab. After excluding systematic reviews, uncontrolled trials, and secondary analyses, 49 trials of 14,590 patients remained for meta-analysis, the investigators said (Clin Gastroenterol Hepatol. 2016 May 4. doi: 10.1016/j.cgh.2016.04.039).
Biologic therapy conferred a “moderate increase” in the likelihood of any infection, with an odds ratio of 1.19 when compared with patients who did not receive biologics (95% confidence interval, 1.10-1.29), the researchers said. The risk of opportunistic infections was somewhat higher (OR, 1.90; 95% CI, 1.21-3.01). However, biologic therapy did not significantly heighten the risk of serious infections, which most studies defined as infections leading to hospitalization, intravenous antibiotic treatment, or death (OR, 0.89; 95% CI, 0.71-1.12). “On the contrary, biologics appeared to significantly reduce risk of serious infections in studies with low risk of bias (OR, 0.56; 95% CI, 0.35-0.90),” the investigators wrote. They did not find an increased risk of malignancy with biologic therapy (OR, 0.90; 95% CI, 0.54-1.50), “but [these] data were insufficient in terms of exposure and follow-up times,” they added.
Comparisons of individual agents and classes also did not reveal any significant associations with the risk of infection, the investigators noted. They did acknowledge several limitations. None of the trials were head-to-head comparisons between biologics, and many were industry funded. Estimates of comparative harm were based on indirect comparisons, and therefore merited cautious interpretation. Because the trials were carried out for regulatory purposes, they enrolled highly selected and homogeneous cohorts of IBD patients, leading to underrepresentation of high-risk and elderly individuals, the researchers noted. “Finally, the exposure and follow-up times were up to 24 months, a time period that is considered sufficient when analyzing infectious adverse effects but wholly insufficient for cancer outcomes. With this in mind, we must look to large register-based cohort studies to enhance our understanding of the biologics-cancer association, despite the biases inherent in observational study designs.”
Dr. Bonovas had no disclosures, while three coauthors reported relationships with a number of pharmaceutical companies.
FROM CLINICAL GASTROENTEROLOGY AND HEPATOLOGY
Key clinical point: Biologic therapies for inflammatory bowel disease significantly increase the risk of opportunistic infections but not serious infections.
Major finding: Biologic therapy increased the odds of opportunistic infection about 1.9-fold, but did not significantly increase the odds of infections leading to hospitalization, intravenous antibiotic treatment, or death.
Data source: A systematic review and meta-analysis of 49 randomized controlled trials of 14,590 patients with inflammatory bowel disease.
Disclosures: The investigators did not report external funding sources. Dr. Bonovas had no disclosures, while three coauthors reported relationships with a number of pharmaceutical companies.
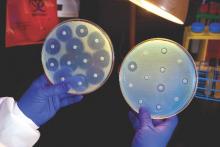
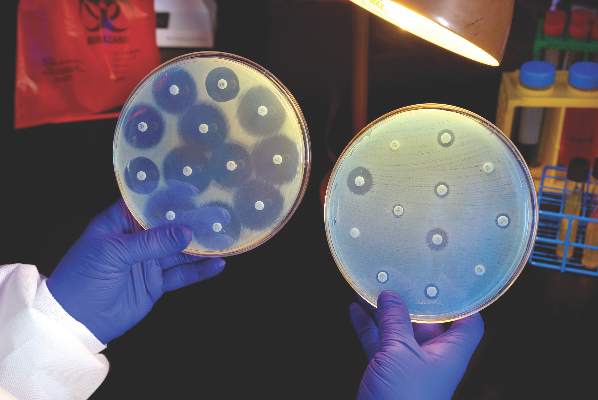
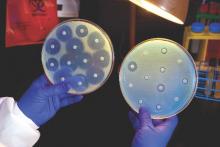
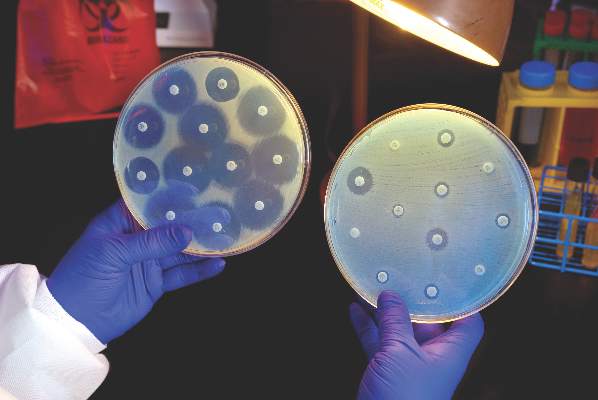
Ceftazidime-avibactam stands up to CRE, but resistance a problem
Intravenous ceftazidime-avibactam successfully treated 59% of carbapenem-resistant Enterobacteriaceae (CRE) infections, and 76% of patients remained alive at 30 days, according to a retrospective cohort study published in Clinical Infectious Diseases.
Those rates resemble previous reports of treatment with in vitro active agents, while the rate of acute kidney injury was about a third lower, said Ryan K. Shields, PharmD, of the University of Pittsburgh, and his associates. But 8% of CRE infections developed ceftazidime-avibactam resistance, which accounted for about a third of microbiological failures, the researchers said. “It is incumbent upon health care providers to share their clinical experiences with ceftazidime-avibactam and other new beta-lactamase inhibitors, so these agents can be used most effectively for the longest period of time,” they added.
Ceftazidime-avibactam (Avycaz) is a novel beta-lactam/beta-lactamase inhibitor combination approved by the Food and Drug Administration in 2015 for complicated intra-abdominal and urinary tract infections. It was hoped that the newly approved combination would prove safer and more effective than previously developed agents that showed in vitro activity against CRE, such as colistin, gentamicin, and tigecycline, the researchers noted.
They described CRE-infected patients treated with ceftazidime-avibactam (median, 14 days; range, 4-71 days) between April 2015 and February 2016. The average age of the patients was 64 years (range 26-78 years), and 57% were men. Infections included ventilator or health care–associated pneumonia, primary bacteremia, intra-abdominal infection, skin and soft tissue infections, pyelonephritis, mediastinitis, subdural empyema/ventriculitis, and purulent tracheobronchitis. All the CRE isolates were susceptible to ceftazidime-avibactam at baseline. In all, 70% of patients received ceftazidime-avibactam as monotherapy, while 30% received it in combination with intravenous or inhaled gentamicin, intravenous or intrathecal colistin, or tigecycline (Clin Infect Dis. 2016 Sep 13. doi: 10.1093/cid/ciw636).
A total of 28 (76%) patients were alive at 30 days and 62% were alive at 90 days, the investigators said. They calculated a 59% rate of clinical success, defined as absence of recurrence within 30 days of onset, resolution of signs and symptoms, and sterilization of site-specific cultures within 7 days of treatment. Combination therapy did not improve the chances of clinical success, they noted. Among the 15 clinical failures, 9 patients died, 4 developed recurrent CRE infections, and 2 did not clinically improve. Clinical success was less likely when patients needed continuous renal replacement therapy (17% vs. 68% for other patients; P = .03) or had higher Sequential Organ Failure Assessment (SOFA) scores (average, 5.2 in clinical successes vs. 8.8 in clinical failures; P = .047). In addition, 10% of patients developed acute kidney injury within 7 days of starting treatment, which was “considerably lower than the approximately 30% rate we previously reported with carbapenem-colistin or aminoglycoside-based combinations,” the investigators said.
The sample size was too small to definitively answer questions about whether combination regimens can overcome resistance, improve outcomes, or effectively treat specific types of CRE infection, the researchers noted. “Nevertheless, we can conclude that ceftazidime-avibactam offers an important advance in the treatment of CRE infections,” they wrote. “The development of resistance after as few as 10 days of therapy is troubling, and treatment failures and deaths in a significant minority of patients highlight the need for more agents with activity against CRE.”
The University of Pittsburgh Medical Center and the National Institutes of Health provided funding. One coauthor disclosed ties to Meiji, Shionogi, Tetraphase, Achaogen, Merck, and The Medicines Company. The other authors had no disclosures.
In the movie “Jaws,” after confidently setting out with an experienced shark hunter, upon catching his first glimpse of the predator, Chief Brody famously uttered, “We’re gonna need a bigger boat.” Similarly, we rejoiced at our triumph when ceftazidime-avibactam became available to treat our patients infected with [Klebsiella pneumoniae carbapenemase]-producing bacteria, and confidently set out to combat this killer. But like Chief Brody, we appear to have underestimated our foe; we too need a “bigger boat.”
We must not let the past repeat itself; hubris about the sudden availability of effective antibiotics has led to overconfidence and complacency among the medical and microbiological communities on several prior occasions in the last 80 years, with serious societal consequences. Shields and colleagues have provided us with a sobering reminder that there is no endpoint in our struggle against microbes. They will never stop adapting to what we conceive of to combat them, and in turn we must never stop conceiving of new ways to stay one step ahead.
Brad Spellberg, MD, is at Los Angeles County–USC Medical Center in Los Angeles. He disclosed ties to Cempra, The Medicines Company, MedImmune/AstraZeneca, PTC Therapeutics, Entasis, Tetraphase, Merck, Genentech, Dipexium, Motif, BioAIM, and Synthetic Biologics. He has received grants from AstraZeneca, Merck, Melinta, Steris, NIH, and Veterans Affairs Merit Review. These comments are from an editorial (Clin Infect Dis. 2016 Sept 13. doi: 10.1093/cid/ciw639).
In the movie “Jaws,” after confidently setting out with an experienced shark hunter, upon catching his first glimpse of the predator, Chief Brody famously uttered, “We’re gonna need a bigger boat.” Similarly, we rejoiced at our triumph when ceftazidime-avibactam became available to treat our patients infected with [Klebsiella pneumoniae carbapenemase]-producing bacteria, and confidently set out to combat this killer. But like Chief Brody, we appear to have underestimated our foe; we too need a “bigger boat.”
We must not let the past repeat itself; hubris about the sudden availability of effective antibiotics has led to overconfidence and complacency among the medical and microbiological communities on several prior occasions in the last 80 years, with serious societal consequences. Shields and colleagues have provided us with a sobering reminder that there is no endpoint in our struggle against microbes. They will never stop adapting to what we conceive of to combat them, and in turn we must never stop conceiving of new ways to stay one step ahead.
Brad Spellberg, MD, is at Los Angeles County–USC Medical Center in Los Angeles. He disclosed ties to Cempra, The Medicines Company, MedImmune/AstraZeneca, PTC Therapeutics, Entasis, Tetraphase, Merck, Genentech, Dipexium, Motif, BioAIM, and Synthetic Biologics. He has received grants from AstraZeneca, Merck, Melinta, Steris, NIH, and Veterans Affairs Merit Review. These comments are from an editorial (Clin Infect Dis. 2016 Sept 13. doi: 10.1093/cid/ciw639).
In the movie “Jaws,” after confidently setting out with an experienced shark hunter, upon catching his first glimpse of the predator, Chief Brody famously uttered, “We’re gonna need a bigger boat.” Similarly, we rejoiced at our triumph when ceftazidime-avibactam became available to treat our patients infected with [Klebsiella pneumoniae carbapenemase]-producing bacteria, and confidently set out to combat this killer. But like Chief Brody, we appear to have underestimated our foe; we too need a “bigger boat.”
We must not let the past repeat itself; hubris about the sudden availability of effective antibiotics has led to overconfidence and complacency among the medical and microbiological communities on several prior occasions in the last 80 years, with serious societal consequences. Shields and colleagues have provided us with a sobering reminder that there is no endpoint in our struggle against microbes. They will never stop adapting to what we conceive of to combat them, and in turn we must never stop conceiving of new ways to stay one step ahead.
Brad Spellberg, MD, is at Los Angeles County–USC Medical Center in Los Angeles. He disclosed ties to Cempra, The Medicines Company, MedImmune/AstraZeneca, PTC Therapeutics, Entasis, Tetraphase, Merck, Genentech, Dipexium, Motif, BioAIM, and Synthetic Biologics. He has received grants from AstraZeneca, Merck, Melinta, Steris, NIH, and Veterans Affairs Merit Review. These comments are from an editorial (Clin Infect Dis. 2016 Sept 13. doi: 10.1093/cid/ciw639).
Intravenous ceftazidime-avibactam successfully treated 59% of carbapenem-resistant Enterobacteriaceae (CRE) infections, and 76% of patients remained alive at 30 days, according to a retrospective cohort study published in Clinical Infectious Diseases.
Those rates resemble previous reports of treatment with in vitro active agents, while the rate of acute kidney injury was about a third lower, said Ryan K. Shields, PharmD, of the University of Pittsburgh, and his associates. But 8% of CRE infections developed ceftazidime-avibactam resistance, which accounted for about a third of microbiological failures, the researchers said. “It is incumbent upon health care providers to share their clinical experiences with ceftazidime-avibactam and other new beta-lactamase inhibitors, so these agents can be used most effectively for the longest period of time,” they added.
Ceftazidime-avibactam (Avycaz) is a novel beta-lactam/beta-lactamase inhibitor combination approved by the Food and Drug Administration in 2015 for complicated intra-abdominal and urinary tract infections. It was hoped that the newly approved combination would prove safer and more effective than previously developed agents that showed in vitro activity against CRE, such as colistin, gentamicin, and tigecycline, the researchers noted.
They described CRE-infected patients treated with ceftazidime-avibactam (median, 14 days; range, 4-71 days) between April 2015 and February 2016. The average age of the patients was 64 years (range 26-78 years), and 57% were men. Infections included ventilator or health care–associated pneumonia, primary bacteremia, intra-abdominal infection, skin and soft tissue infections, pyelonephritis, mediastinitis, subdural empyema/ventriculitis, and purulent tracheobronchitis. All the CRE isolates were susceptible to ceftazidime-avibactam at baseline. In all, 70% of patients received ceftazidime-avibactam as monotherapy, while 30% received it in combination with intravenous or inhaled gentamicin, intravenous or intrathecal colistin, or tigecycline (Clin Infect Dis. 2016 Sep 13. doi: 10.1093/cid/ciw636).
A total of 28 (76%) patients were alive at 30 days and 62% were alive at 90 days, the investigators said. They calculated a 59% rate of clinical success, defined as absence of recurrence within 30 days of onset, resolution of signs and symptoms, and sterilization of site-specific cultures within 7 days of treatment. Combination therapy did not improve the chances of clinical success, they noted. Among the 15 clinical failures, 9 patients died, 4 developed recurrent CRE infections, and 2 did not clinically improve. Clinical success was less likely when patients needed continuous renal replacement therapy (17% vs. 68% for other patients; P = .03) or had higher Sequential Organ Failure Assessment (SOFA) scores (average, 5.2 in clinical successes vs. 8.8 in clinical failures; P = .047). In addition, 10% of patients developed acute kidney injury within 7 days of starting treatment, which was “considerably lower than the approximately 30% rate we previously reported with carbapenem-colistin or aminoglycoside-based combinations,” the investigators said.
The sample size was too small to definitively answer questions about whether combination regimens can overcome resistance, improve outcomes, or effectively treat specific types of CRE infection, the researchers noted. “Nevertheless, we can conclude that ceftazidime-avibactam offers an important advance in the treatment of CRE infections,” they wrote. “The development of resistance after as few as 10 days of therapy is troubling, and treatment failures and deaths in a significant minority of patients highlight the need for more agents with activity against CRE.”
The University of Pittsburgh Medical Center and the National Institutes of Health provided funding. One coauthor disclosed ties to Meiji, Shionogi, Tetraphase, Achaogen, Merck, and The Medicines Company. The other authors had no disclosures.
Intravenous ceftazidime-avibactam successfully treated 59% of carbapenem-resistant Enterobacteriaceae (CRE) infections, and 76% of patients remained alive at 30 days, according to a retrospective cohort study published in Clinical Infectious Diseases.
Those rates resemble previous reports of treatment with in vitro active agents, while the rate of acute kidney injury was about a third lower, said Ryan K. Shields, PharmD, of the University of Pittsburgh, and his associates. But 8% of CRE infections developed ceftazidime-avibactam resistance, which accounted for about a third of microbiological failures, the researchers said. “It is incumbent upon health care providers to share their clinical experiences with ceftazidime-avibactam and other new beta-lactamase inhibitors, so these agents can be used most effectively for the longest period of time,” they added.
Ceftazidime-avibactam (Avycaz) is a novel beta-lactam/beta-lactamase inhibitor combination approved by the Food and Drug Administration in 2015 for complicated intra-abdominal and urinary tract infections. It was hoped that the newly approved combination would prove safer and more effective than previously developed agents that showed in vitro activity against CRE, such as colistin, gentamicin, and tigecycline, the researchers noted.
They described CRE-infected patients treated with ceftazidime-avibactam (median, 14 days; range, 4-71 days) between April 2015 and February 2016. The average age of the patients was 64 years (range 26-78 years), and 57% were men. Infections included ventilator or health care–associated pneumonia, primary bacteremia, intra-abdominal infection, skin and soft tissue infections, pyelonephritis, mediastinitis, subdural empyema/ventriculitis, and purulent tracheobronchitis. All the CRE isolates were susceptible to ceftazidime-avibactam at baseline. In all, 70% of patients received ceftazidime-avibactam as monotherapy, while 30% received it in combination with intravenous or inhaled gentamicin, intravenous or intrathecal colistin, or tigecycline (Clin Infect Dis. 2016 Sep 13. doi: 10.1093/cid/ciw636).
A total of 28 (76%) patients were alive at 30 days and 62% were alive at 90 days, the investigators said. They calculated a 59% rate of clinical success, defined as absence of recurrence within 30 days of onset, resolution of signs and symptoms, and sterilization of site-specific cultures within 7 days of treatment. Combination therapy did not improve the chances of clinical success, they noted. Among the 15 clinical failures, 9 patients died, 4 developed recurrent CRE infections, and 2 did not clinically improve. Clinical success was less likely when patients needed continuous renal replacement therapy (17% vs. 68% for other patients; P = .03) or had higher Sequential Organ Failure Assessment (SOFA) scores (average, 5.2 in clinical successes vs. 8.8 in clinical failures; P = .047). In addition, 10% of patients developed acute kidney injury within 7 days of starting treatment, which was “considerably lower than the approximately 30% rate we previously reported with carbapenem-colistin or aminoglycoside-based combinations,” the investigators said.
The sample size was too small to definitively answer questions about whether combination regimens can overcome resistance, improve outcomes, or effectively treat specific types of CRE infection, the researchers noted. “Nevertheless, we can conclude that ceftazidime-avibactam offers an important advance in the treatment of CRE infections,” they wrote. “The development of resistance after as few as 10 days of therapy is troubling, and treatment failures and deaths in a significant minority of patients highlight the need for more agents with activity against CRE.”
The University of Pittsburgh Medical Center and the National Institutes of Health provided funding. One coauthor disclosed ties to Meiji, Shionogi, Tetraphase, Achaogen, Merck, and The Medicines Company. The other authors had no disclosures.
FROM CLINICAL INFECTIOUS DISEASES
Key clinical point: Ceftazidime-avibactam effectively treated carbapenem-resistant Enterobacteriaceae (CRE) infections, but resistance emerged rapidly and in some cases led to microbiological failure.
Major finding: The rate of clinical success was 59%; 10% of patients developed acute kidney injury within 7 days of starting treatment; 8% developed resistance.
Data source: A single-center retrospective study of 37 patients with carbapenem-resistant Enterobacteriaceae infections treated with ceftazidime-avibactam.
Disclosures: The University of Pittsburgh Medical Center and the National Institutes of Health provided funding. A coauthor disclosed ties to Meiji, Shionogi, Tetraphase, Achaogen, Merck, and The Medicines Company. The other authors had no disclosures.
CSF2RB mutation, common in Ashkenazim, linked to Crohn’s
A frameshift mutation in the CSF2RB gene that is common in Ashkenazi Jews has been linked to Crohn’s disease, according to a report in Gastroenterology.
The Ashkenazi Jewish population has a four- to sevenfold higher prevalence of Crohn’s disease than does the general population. To identify possible genetic mutations associated with the disease, researchers performed exome sequencing of samples from 1,477 Ashkenazi Jewish patients with Crohn’s disease and 2,614 Ashkenazi Jewish control subjects who didn’t have the disorder. All the study participants were enrolled at medical centers throughout North America, Europe, and Israel, noted Ling-Shiang Chuang, PhD, a postdoctoral fellow in genetics and genomic sciences, Mount Sinai Medical Center, New York, and his associates.
They genotyped 224 frameshift mutations and identified one in the colony-stimulating factor 2–receptor beta common subunit (CSF2RB) gene that was significantly associated with Crohn’s disease. They validated this association in a replication cohort of 1,515 Ashkenazi Jewish patients with Crohn’s disease and 7,052 health Ashkenazi control subjects, observing nearly identical allele frequencies and odds ratios as in the discovery cohort.
Since the CSF2RB gene encodes for the receptor for GM-CSF (granulocyte-macrophage colony-stimulating factor) cytokines, the mutation would be expected to reduce GM-CSF signaling in monocytes obtained from carriers. The investigators found this was true when they examined monocytes taken from intestinal-tissue samples and peripheral-blood samples from affected patients.
“The present findings of a loss-of-function frameshift variant in the CSF2RB gene argue for a primary pathogenic role of decreased GM-CSF signaling in driving a subset of Crohn’s disease cases. As many as 30% of cases have increased levels of anti-GM-CSF antibodies, which can neutralize GM-CSF activity, indicating that impaired GM-CSF signaling plays a major role in Crohn’s disease,” Dr. Chuang and his associates wrote (Gastroenterol. 2016;151:710-23.e2. doi: 10.1053/j.gastro.2016.06.045).
Future research may identify subgroups of Crohn’s patients in which treatments that target GM-CSF signaling may be effective, and it may also point the way to novel therapies for the broader population of patients with Crohn’s disease, they added.
This study was supported by the National Institutes of Health, the Inflammatory Bowel Disease Genetics Consortium, the Genetic Research Center at Mount Sinai Medical Center, New York’s Crohn’s Foundation, and several others. Dr. Chuang and his associates reported having no relevant financial disclosures.
Genome-wide association studies (GWASs) have identified over 200 DNA mutations (mainly single nucleotide polymorphisms, SNPs) contributing to inflammatory bowel disease. However, their causality in IBD remains unknown. In the post-GWAS era, functional characterization and mechanistic elucidation of these SNPs is a major challenge. SNPs impacting the protein-coding genes can drastically alter protein function and play an important role in molecular pathogenesis. However, most SNPs are in noncoding genes and their impact on gene regulation and disease outcome remains largely unknown. This study reveals a frameshift mutation in the CSF2RB gene associated with Crohn’s disease in an Ashkenazi Jewish population. A frameshift mutation results in the deletion or insertion in a DNA sequence that shifts the way the sequence is transcribed into RNA. The most well described frameshift mutation in Crohn’s disease with clinical relevance is in the NOD2 gene, which results in impaired immune response to microbial stimuli. Because GWASs show association not causality, the biological consequence of the CSF2RB frameshift mutation in intestinal macrophages leads to a decrease in its response to granulocyte-monocyte stimulating factor (GM-CSF) providing a potential mechanistic role for the mutation in Crohn’s disease pathogenesis in a disease-relevant cell and site. This study will pave the way for future important studies that reveal the impact of SNPs on Crohn’s disease development, prognosis, and eventually response to therapy.
Shehzad Z. Sheikh MD, PhD, assistant professor of medicine, department of medicine, division of gastroenterology and hepatology, University of North Carolina, Chapel Hill.
Genome-wide association studies (GWASs) have identified over 200 DNA mutations (mainly single nucleotide polymorphisms, SNPs) contributing to inflammatory bowel disease. However, their causality in IBD remains unknown. In the post-GWAS era, functional characterization and mechanistic elucidation of these SNPs is a major challenge. SNPs impacting the protein-coding genes can drastically alter protein function and play an important role in molecular pathogenesis. However, most SNPs are in noncoding genes and their impact on gene regulation and disease outcome remains largely unknown. This study reveals a frameshift mutation in the CSF2RB gene associated with Crohn’s disease in an Ashkenazi Jewish population. A frameshift mutation results in the deletion or insertion in a DNA sequence that shifts the way the sequence is transcribed into RNA. The most well described frameshift mutation in Crohn’s disease with clinical relevance is in the NOD2 gene, which results in impaired immune response to microbial stimuli. Because GWASs show association not causality, the biological consequence of the CSF2RB frameshift mutation in intestinal macrophages leads to a decrease in its response to granulocyte-monocyte stimulating factor (GM-CSF) providing a potential mechanistic role for the mutation in Crohn’s disease pathogenesis in a disease-relevant cell and site. This study will pave the way for future important studies that reveal the impact of SNPs on Crohn’s disease development, prognosis, and eventually response to therapy.
Shehzad Z. Sheikh MD, PhD, assistant professor of medicine, department of medicine, division of gastroenterology and hepatology, University of North Carolina, Chapel Hill.
Genome-wide association studies (GWASs) have identified over 200 DNA mutations (mainly single nucleotide polymorphisms, SNPs) contributing to inflammatory bowel disease. However, their causality in IBD remains unknown. In the post-GWAS era, functional characterization and mechanistic elucidation of these SNPs is a major challenge. SNPs impacting the protein-coding genes can drastically alter protein function and play an important role in molecular pathogenesis. However, most SNPs are in noncoding genes and their impact on gene regulation and disease outcome remains largely unknown. This study reveals a frameshift mutation in the CSF2RB gene associated with Crohn’s disease in an Ashkenazi Jewish population. A frameshift mutation results in the deletion or insertion in a DNA sequence that shifts the way the sequence is transcribed into RNA. The most well described frameshift mutation in Crohn’s disease with clinical relevance is in the NOD2 gene, which results in impaired immune response to microbial stimuli. Because GWASs show association not causality, the biological consequence of the CSF2RB frameshift mutation in intestinal macrophages leads to a decrease in its response to granulocyte-monocyte stimulating factor (GM-CSF) providing a potential mechanistic role for the mutation in Crohn’s disease pathogenesis in a disease-relevant cell and site. This study will pave the way for future important studies that reveal the impact of SNPs on Crohn’s disease development, prognosis, and eventually response to therapy.
Shehzad Z. Sheikh MD, PhD, assistant professor of medicine, department of medicine, division of gastroenterology and hepatology, University of North Carolina, Chapel Hill.
A frameshift mutation in the CSF2RB gene that is common in Ashkenazi Jews has been linked to Crohn’s disease, according to a report in Gastroenterology.
The Ashkenazi Jewish population has a four- to sevenfold higher prevalence of Crohn’s disease than does the general population. To identify possible genetic mutations associated with the disease, researchers performed exome sequencing of samples from 1,477 Ashkenazi Jewish patients with Crohn’s disease and 2,614 Ashkenazi Jewish control subjects who didn’t have the disorder. All the study participants were enrolled at medical centers throughout North America, Europe, and Israel, noted Ling-Shiang Chuang, PhD, a postdoctoral fellow in genetics and genomic sciences, Mount Sinai Medical Center, New York, and his associates.
They genotyped 224 frameshift mutations and identified one in the colony-stimulating factor 2–receptor beta common subunit (CSF2RB) gene that was significantly associated with Crohn’s disease. They validated this association in a replication cohort of 1,515 Ashkenazi Jewish patients with Crohn’s disease and 7,052 health Ashkenazi control subjects, observing nearly identical allele frequencies and odds ratios as in the discovery cohort.
Since the CSF2RB gene encodes for the receptor for GM-CSF (granulocyte-macrophage colony-stimulating factor) cytokines, the mutation would be expected to reduce GM-CSF signaling in monocytes obtained from carriers. The investigators found this was true when they examined monocytes taken from intestinal-tissue samples and peripheral-blood samples from affected patients.
“The present findings of a loss-of-function frameshift variant in the CSF2RB gene argue for a primary pathogenic role of decreased GM-CSF signaling in driving a subset of Crohn’s disease cases. As many as 30% of cases have increased levels of anti-GM-CSF antibodies, which can neutralize GM-CSF activity, indicating that impaired GM-CSF signaling plays a major role in Crohn’s disease,” Dr. Chuang and his associates wrote (Gastroenterol. 2016;151:710-23.e2. doi: 10.1053/j.gastro.2016.06.045).
Future research may identify subgroups of Crohn’s patients in which treatments that target GM-CSF signaling may be effective, and it may also point the way to novel therapies for the broader population of patients with Crohn’s disease, they added.
This study was supported by the National Institutes of Health, the Inflammatory Bowel Disease Genetics Consortium, the Genetic Research Center at Mount Sinai Medical Center, New York’s Crohn’s Foundation, and several others. Dr. Chuang and his associates reported having no relevant financial disclosures.
A frameshift mutation in the CSF2RB gene that is common in Ashkenazi Jews has been linked to Crohn’s disease, according to a report in Gastroenterology.
The Ashkenazi Jewish population has a four- to sevenfold higher prevalence of Crohn’s disease than does the general population. To identify possible genetic mutations associated with the disease, researchers performed exome sequencing of samples from 1,477 Ashkenazi Jewish patients with Crohn’s disease and 2,614 Ashkenazi Jewish control subjects who didn’t have the disorder. All the study participants were enrolled at medical centers throughout North America, Europe, and Israel, noted Ling-Shiang Chuang, PhD, a postdoctoral fellow in genetics and genomic sciences, Mount Sinai Medical Center, New York, and his associates.
They genotyped 224 frameshift mutations and identified one in the colony-stimulating factor 2–receptor beta common subunit (CSF2RB) gene that was significantly associated with Crohn’s disease. They validated this association in a replication cohort of 1,515 Ashkenazi Jewish patients with Crohn’s disease and 7,052 health Ashkenazi control subjects, observing nearly identical allele frequencies and odds ratios as in the discovery cohort.
Since the CSF2RB gene encodes for the receptor for GM-CSF (granulocyte-macrophage colony-stimulating factor) cytokines, the mutation would be expected to reduce GM-CSF signaling in monocytes obtained from carriers. The investigators found this was true when they examined monocytes taken from intestinal-tissue samples and peripheral-blood samples from affected patients.
“The present findings of a loss-of-function frameshift variant in the CSF2RB gene argue for a primary pathogenic role of decreased GM-CSF signaling in driving a subset of Crohn’s disease cases. As many as 30% of cases have increased levels of anti-GM-CSF antibodies, which can neutralize GM-CSF activity, indicating that impaired GM-CSF signaling plays a major role in Crohn’s disease,” Dr. Chuang and his associates wrote (Gastroenterol. 2016;151:710-23.e2. doi: 10.1053/j.gastro.2016.06.045).
Future research may identify subgroups of Crohn’s patients in which treatments that target GM-CSF signaling may be effective, and it may also point the way to novel therapies for the broader population of patients with Crohn’s disease, they added.
This study was supported by the National Institutes of Health, the Inflammatory Bowel Disease Genetics Consortium, the Genetic Research Center at Mount Sinai Medical Center, New York’s Crohn’s Foundation, and several others. Dr. Chuang and his associates reported having no relevant financial disclosures.
FROM GASTROENTEROLOGY
Key clinical point: A frameshift mutation in the CSF2RB gene that is common in Ashkenazi Jews has been linked to Crohn’s disease.
Major finding: Genotyping of 224 frameshift mutations identified one in the colony-stimulating factor 2–receptor common subunit (CSF2RB) gene that was significantly associated with Crohn’s disease.
Data source: Exome sequencing and genotype analyses of samples from 1,477 Ashkenazi Jewish people with Crohn’s disease and 2,614 without it.
Disclosures: This study was supported by the National Institutes of Health, the Inflammatory Bowel Disease Genetics Consortium, the Genetic Research Center at Mount Sinai Medical Center, New York’s Crohn’s Foundation, and several others. Dr. Chuang and his associates reported having no relevant financial disclosures.
FDA approves biosimilar adalimumab
There are “no clinically meaningful differences” between Amgen’s biosimilar adalimumab (Amjevita) and AbbVie’s branded product Humira, the Food and Drug Administration noted it its Sept. 23 announcement of Amjevita’s approval.
Although Amjevita (adalimumab-atto) is expected to cost less than Humira, Amgen has not released price information or a launch date pending ongoing litigation with AbbVie over intellectual property rights, an Amgen spokeswoman said.
The products carry an identical black box warning of tuberculosis and other serious infections, as well as lymphoma and other malignancies “reported in children and adolescent patients treated with [tumor necrosis factor] blockers including adalimumab.” As with Humira, “the most common expected adverse reactions with Amjevita are infections and injection site reactions,” the FDA said. Both products are approved in 20 mg/0.4 mL and 40 mg/0.8 mL prefilled injections, but Humira also has a 10 mg/0.2 mL option.
Amjevita was unanimously recommended for approval by an FDA review panel in July. Although “the biosimilar pathway is still a new frontier,” it’s likely to “enhance access to treatment for patients with serious medical conditions,” Janet Woodcock, MD, director of the FDA’s Center for Drug Evaluation and Research, said in the agency statement.
The approval follows the FDA’s approval of biosimilar infliximab (Inflectra) in April 2016 and biosimilar etanercept (Erelzi) in August 2016. Inflectra has not hit the U.S. market yet, but the European experience with biosimilar infliximab – generally positive – may give an indication of how Amjevita will fare in the United States. It’s perhaps a third or more less expensive than the original product (Remicade) and often used for new starts. There is uncertainty, however, about switching patients already established on Remicade, especially when it’s forced by cost issues.
Interchangeability is a concern in the United States as well. The FDA is working on the issue but has not yet released guidance, and the agency was careful to note in its statement that Amjevita was “approved as a biosimilar, not as an interchangeable product.” Biosimilar adalimumab, meanwhile, is under review in Europe, according to an Amgen statement.
The FDA approved Amjevita after reviewing structural and functional characteristics, pharmacokinetics and pharmacodynamics data, clinical immunogenicity data, and other clinical safety and effectiveness data that demonstrated similarity to Humira, including two phase III trials for plaque psoriasis and rheumatoid arthritis.
An AbbVie spokesperson said the company “anticipated Amgen’s product would be approved,” but noted the ongoing litigation.
There are “no clinically meaningful differences” between Amgen’s biosimilar adalimumab (Amjevita) and AbbVie’s branded product Humira, the Food and Drug Administration noted it its Sept. 23 announcement of Amjevita’s approval.
Although Amjevita (adalimumab-atto) is expected to cost less than Humira, Amgen has not released price information or a launch date pending ongoing litigation with AbbVie over intellectual property rights, an Amgen spokeswoman said.
The products carry an identical black box warning of tuberculosis and other serious infections, as well as lymphoma and other malignancies “reported in children and adolescent patients treated with [tumor necrosis factor] blockers including adalimumab.” As with Humira, “the most common expected adverse reactions with Amjevita are infections and injection site reactions,” the FDA said. Both products are approved in 20 mg/0.4 mL and 40 mg/0.8 mL prefilled injections, but Humira also has a 10 mg/0.2 mL option.
Amjevita was unanimously recommended for approval by an FDA review panel in July. Although “the biosimilar pathway is still a new frontier,” it’s likely to “enhance access to treatment for patients with serious medical conditions,” Janet Woodcock, MD, director of the FDA’s Center for Drug Evaluation and Research, said in the agency statement.
The approval follows the FDA’s approval of biosimilar infliximab (Inflectra) in April 2016 and biosimilar etanercept (Erelzi) in August 2016. Inflectra has not hit the U.S. market yet, but the European experience with biosimilar infliximab – generally positive – may give an indication of how Amjevita will fare in the United States. It’s perhaps a third or more less expensive than the original product (Remicade) and often used for new starts. There is uncertainty, however, about switching patients already established on Remicade, especially when it’s forced by cost issues.
Interchangeability is a concern in the United States as well. The FDA is working on the issue but has not yet released guidance, and the agency was careful to note in its statement that Amjevita was “approved as a biosimilar, not as an interchangeable product.” Biosimilar adalimumab, meanwhile, is under review in Europe, according to an Amgen statement.
The FDA approved Amjevita after reviewing structural and functional characteristics, pharmacokinetics and pharmacodynamics data, clinical immunogenicity data, and other clinical safety and effectiveness data that demonstrated similarity to Humira, including two phase III trials for plaque psoriasis and rheumatoid arthritis.
An AbbVie spokesperson said the company “anticipated Amgen’s product would be approved,” but noted the ongoing litigation.
There are “no clinically meaningful differences” between Amgen’s biosimilar adalimumab (Amjevita) and AbbVie’s branded product Humira, the Food and Drug Administration noted it its Sept. 23 announcement of Amjevita’s approval.
Although Amjevita (adalimumab-atto) is expected to cost less than Humira, Amgen has not released price information or a launch date pending ongoing litigation with AbbVie over intellectual property rights, an Amgen spokeswoman said.
The products carry an identical black box warning of tuberculosis and other serious infections, as well as lymphoma and other malignancies “reported in children and adolescent patients treated with [tumor necrosis factor] blockers including adalimumab.” As with Humira, “the most common expected adverse reactions with Amjevita are infections and injection site reactions,” the FDA said. Both products are approved in 20 mg/0.4 mL and 40 mg/0.8 mL prefilled injections, but Humira also has a 10 mg/0.2 mL option.
Amjevita was unanimously recommended for approval by an FDA review panel in July. Although “the biosimilar pathway is still a new frontier,” it’s likely to “enhance access to treatment for patients with serious medical conditions,” Janet Woodcock, MD, director of the FDA’s Center for Drug Evaluation and Research, said in the agency statement.
The approval follows the FDA’s approval of biosimilar infliximab (Inflectra) in April 2016 and biosimilar etanercept (Erelzi) in August 2016. Inflectra has not hit the U.S. market yet, but the European experience with biosimilar infliximab – generally positive – may give an indication of how Amjevita will fare in the United States. It’s perhaps a third or more less expensive than the original product (Remicade) and often used for new starts. There is uncertainty, however, about switching patients already established on Remicade, especially when it’s forced by cost issues.
Interchangeability is a concern in the United States as well. The FDA is working on the issue but has not yet released guidance, and the agency was careful to note in its statement that Amjevita was “approved as a biosimilar, not as an interchangeable product.” Biosimilar adalimumab, meanwhile, is under review in Europe, according to an Amgen statement.
The FDA approved Amjevita after reviewing structural and functional characteristics, pharmacokinetics and pharmacodynamics data, clinical immunogenicity data, and other clinical safety and effectiveness data that demonstrated similarity to Humira, including two phase III trials for plaque psoriasis and rheumatoid arthritis.
An AbbVie spokesperson said the company “anticipated Amgen’s product would be approved,” but noted the ongoing litigation.
Gluten-free adherence triples while celiac disease prevalence remains stable
The number of people adhering to a gluten-free diet more than tripled between 2009 and 2014, despite the fact that the prevalence of celiac disease has remained largely stable over the same period, according to data from the National Health and Nutrition Examination Survey.
Hyun-seok Kim, MD, MPH, and colleagues from Rutgers New Jersey Medical School noted that there is a popular trend of people choosing gluten-free diets, which exceeds the numbers that would be solely attributable to an increasing prevalence of celiac disease.
In a report published online Sept. 6 in JAMA Internal Medicine, the researchers noted that of 22,278 persons aged 6 years or older for whom data were available on celiac disease status and gluten-free diet status, 106 (0.69%) had a diagnosis of celiac disease, and 213 (1.08%) followed a gluten-free diet but did not have celiac disease.
At a U.S. population level, this would correspond to an estimated 1.76 million individuals with celiac disease, and 2.7 million individuals without celiac disease who follow a gluten-free diet.
The prevalence of celiac disease ranged from 0.70% during 2009-2010, to 0.77% during 2011-2012, and 0.58% during 2013-2014 (JAMA Intern Med. 2016 Sept 6. doi: 10.1001/jamainternmed.2016.5254).
In contrast, the prevalence of a gluten-free diet without celiac disease increased from 0.52% during 2009-2010 to 0.99% during 2011-2012 and 1.69% during 2013-2014, although the increase was even greater among non-Hispanic whites.
“The two trends may be related because gluten consumption has been identified as a risk factor of celiac disease, such that steady or even decreasing gluten consumption may be contributing to a plateau in celiac disease,” they reported.
The authors suggested that there were a number of reasons why individuals without celiac disease might choose to follow a gluten-free diet. “The public perception is that gluten-free diets are healthier and may provide benefits to nonspecific gastrointestinal symptoms,” they wrote, pointing out that gluten-free products are now also more widely available in supermarkets and online.
“There is also an increasing number of individuals with self-diagnosed gluten sensitivity but not the typical enteropathic or serologic features of celiac disease who have improved gastrointestinal health after avoidance of gluten-containing products.”
They stressed that the numbers of individuals in the survey with celiac disease or adhering to a gluten-free diet were relatively small, and that a diagnosis of celiac disease was not confirmed by intestinal biopsy, relying instead on serological tests and prior diagnosis by a health professional.
No conflicts of interest were declared.
Part of what may be driving this gluten-free diet trend is simply a belief, fueled by marketing and media, that these foods are healthier. However, surveys suggest that many individuals who adhere to a gluten-free diet believe that the exclusion of gluten has resulted in subjective health benefits from weight loss to reduced symptoms of inflammation and gastrointestinal distress.
Because a gluten-free diet may have negative social, financial, and health repercussions, it is important for clinicians to understand whether, in most cases, it is the elimination of the protein gluten that is responsible for symptom improvement or whether following a gluten-free diet is simply a marker of other dietary choices that are creating positive effects.
Although the choice to be gluten free may be driven in part by marketing and a misperception that gluten free is healthier, it is important that this choice not be dismissed as an unfounded trend except for those with celiac disease and wheat allergy.
Dr. Daphne Miller is from the department of family and community medicine at the University of California, San Francisco. The comments are taken from an editorial (JAMA Intern Med. 2016 Sept 6. doi: 10.1001/jamainternmed.2016.5271). No conflicts of interest were declared.
Part of what may be driving this gluten-free diet trend is simply a belief, fueled by marketing and media, that these foods are healthier. However, surveys suggest that many individuals who adhere to a gluten-free diet believe that the exclusion of gluten has resulted in subjective health benefits from weight loss to reduced symptoms of inflammation and gastrointestinal distress.
Because a gluten-free diet may have negative social, financial, and health repercussions, it is important for clinicians to understand whether, in most cases, it is the elimination of the protein gluten that is responsible for symptom improvement or whether following a gluten-free diet is simply a marker of other dietary choices that are creating positive effects.
Although the choice to be gluten free may be driven in part by marketing and a misperception that gluten free is healthier, it is important that this choice not be dismissed as an unfounded trend except for those with celiac disease and wheat allergy.
Dr. Daphne Miller is from the department of family and community medicine at the University of California, San Francisco. The comments are taken from an editorial (JAMA Intern Med. 2016 Sept 6. doi: 10.1001/jamainternmed.2016.5271). No conflicts of interest were declared.
Part of what may be driving this gluten-free diet trend is simply a belief, fueled by marketing and media, that these foods are healthier. However, surveys suggest that many individuals who adhere to a gluten-free diet believe that the exclusion of gluten has resulted in subjective health benefits from weight loss to reduced symptoms of inflammation and gastrointestinal distress.
Because a gluten-free diet may have negative social, financial, and health repercussions, it is important for clinicians to understand whether, in most cases, it is the elimination of the protein gluten that is responsible for symptom improvement or whether following a gluten-free diet is simply a marker of other dietary choices that are creating positive effects.
Although the choice to be gluten free may be driven in part by marketing and a misperception that gluten free is healthier, it is important that this choice not be dismissed as an unfounded trend except for those with celiac disease and wheat allergy.
Dr. Daphne Miller is from the department of family and community medicine at the University of California, San Francisco. The comments are taken from an editorial (JAMA Intern Med. 2016 Sept 6. doi: 10.1001/jamainternmed.2016.5271). No conflicts of interest were declared.
The number of people adhering to a gluten-free diet more than tripled between 2009 and 2014, despite the fact that the prevalence of celiac disease has remained largely stable over the same period, according to data from the National Health and Nutrition Examination Survey.
Hyun-seok Kim, MD, MPH, and colleagues from Rutgers New Jersey Medical School noted that there is a popular trend of people choosing gluten-free diets, which exceeds the numbers that would be solely attributable to an increasing prevalence of celiac disease.
In a report published online Sept. 6 in JAMA Internal Medicine, the researchers noted that of 22,278 persons aged 6 years or older for whom data were available on celiac disease status and gluten-free diet status, 106 (0.69%) had a diagnosis of celiac disease, and 213 (1.08%) followed a gluten-free diet but did not have celiac disease.
At a U.S. population level, this would correspond to an estimated 1.76 million individuals with celiac disease, and 2.7 million individuals without celiac disease who follow a gluten-free diet.
The prevalence of celiac disease ranged from 0.70% during 2009-2010, to 0.77% during 2011-2012, and 0.58% during 2013-2014 (JAMA Intern Med. 2016 Sept 6. doi: 10.1001/jamainternmed.2016.5254).
In contrast, the prevalence of a gluten-free diet without celiac disease increased from 0.52% during 2009-2010 to 0.99% during 2011-2012 and 1.69% during 2013-2014, although the increase was even greater among non-Hispanic whites.
“The two trends may be related because gluten consumption has been identified as a risk factor of celiac disease, such that steady or even decreasing gluten consumption may be contributing to a plateau in celiac disease,” they reported.
The authors suggested that there were a number of reasons why individuals without celiac disease might choose to follow a gluten-free diet. “The public perception is that gluten-free diets are healthier and may provide benefits to nonspecific gastrointestinal symptoms,” they wrote, pointing out that gluten-free products are now also more widely available in supermarkets and online.
“There is also an increasing number of individuals with self-diagnosed gluten sensitivity but not the typical enteropathic or serologic features of celiac disease who have improved gastrointestinal health after avoidance of gluten-containing products.”
They stressed that the numbers of individuals in the survey with celiac disease or adhering to a gluten-free diet were relatively small, and that a diagnosis of celiac disease was not confirmed by intestinal biopsy, relying instead on serological tests and prior diagnosis by a health professional.
No conflicts of interest were declared.
The number of people adhering to a gluten-free diet more than tripled between 2009 and 2014, despite the fact that the prevalence of celiac disease has remained largely stable over the same period, according to data from the National Health and Nutrition Examination Survey.
Hyun-seok Kim, MD, MPH, and colleagues from Rutgers New Jersey Medical School noted that there is a popular trend of people choosing gluten-free diets, which exceeds the numbers that would be solely attributable to an increasing prevalence of celiac disease.
In a report published online Sept. 6 in JAMA Internal Medicine, the researchers noted that of 22,278 persons aged 6 years or older for whom data were available on celiac disease status and gluten-free diet status, 106 (0.69%) had a diagnosis of celiac disease, and 213 (1.08%) followed a gluten-free diet but did not have celiac disease.
At a U.S. population level, this would correspond to an estimated 1.76 million individuals with celiac disease, and 2.7 million individuals without celiac disease who follow a gluten-free diet.
The prevalence of celiac disease ranged from 0.70% during 2009-2010, to 0.77% during 2011-2012, and 0.58% during 2013-2014 (JAMA Intern Med. 2016 Sept 6. doi: 10.1001/jamainternmed.2016.5254).
In contrast, the prevalence of a gluten-free diet without celiac disease increased from 0.52% during 2009-2010 to 0.99% during 2011-2012 and 1.69% during 2013-2014, although the increase was even greater among non-Hispanic whites.
“The two trends may be related because gluten consumption has been identified as a risk factor of celiac disease, such that steady or even decreasing gluten consumption may be contributing to a plateau in celiac disease,” they reported.
The authors suggested that there were a number of reasons why individuals without celiac disease might choose to follow a gluten-free diet. “The public perception is that gluten-free diets are healthier and may provide benefits to nonspecific gastrointestinal symptoms,” they wrote, pointing out that gluten-free products are now also more widely available in supermarkets and online.
“There is also an increasing number of individuals with self-diagnosed gluten sensitivity but not the typical enteropathic or serologic features of celiac disease who have improved gastrointestinal health after avoidance of gluten-containing products.”
They stressed that the numbers of individuals in the survey with celiac disease or adhering to a gluten-free diet were relatively small, and that a diagnosis of celiac disease was not confirmed by intestinal biopsy, relying instead on serological tests and prior diagnosis by a health professional.
No conflicts of interest were declared.
FROM JAMA INTERNAL MEDICINE
Key clinical point: The number of people adhering to a gluten-free diet more than tripled between 2009 and 2014, while the prevalence of celiac disease has remained largely stable over the same period.
Major finding: The prevalence of celiac disease ranged from 0.70% during 2009-2010 to 0.58% in 2013-2014, while the prevalence of a gluten-free diet without celiac disease increased from 0.52% in 2009-2010 to 1.69% during 2013-2014.
Data source: Analysis of data from 22,278 participants in the National Health and Nutrition Examination Survey.
Disclosures: No conflicts of interest were disclosed.
Hospitals increase CRE risk when they share patients
The more hospitals share patients, the more likely they are to have a problem with carbapenem-resistant Enterobacteriaceae (CRE), especially if long-term acute care hospitals (LTACHs) are in the mix, according to a state-wide investigation from Illinois.
Greater hospital centrality was independently associated with higher rates overall, and sharing four or more patients with a long-term acute care hospital (LTACH) in the 3-month study window doubled the rate of CRE cases.
Although it’s possible that was because of chance (P = 0.11), the link between LTACHs and CRE “is consistent with prior analyses that have shown the central role LTACHs have in” spreading the organism, said the researchers, led by Michael Ray of the Illinois Department of Public Health (Clin Infect Dis. 2016 Aug 2. pii: ciw461).
Patients often spend weeks in LTACH facilities for ongoing, serious health problems. The severity of illness, long stay, and sometimes chronic antibiotic use increase the risk of CRE exposure, and the team found that many LTACH patients are colonized.
“These findings have immediate public health implications. … Early interventions should be focused on the most connected facilities, as well as those with strong connections to LTACHs.” When one hospital has an outbreak, facilities that share its patients need to swing into action screening new admissions and taking other steps to prevent regional spread, the team said.
Meanwhile, “state-wide patient-sharing data, which are now increasingly available through sources like the Healthcare Cost and Utilization Project, provide an important way to assess hospital risk of CRE exposure based on its position in regional patient-sharing networks,” they noted. “Public health can play a critical role in identifying tightly connected hospitals and educating personnel at such facilities about their risk and need for enhanced infection control interventions.”
The team came to their conclusions after linking Illinois’ drug-resistant organisms registry with admissions data for 185 hospitals. About half reported at least one CRE case over 3 months, with a mean of 3.5 cases per hospital.
There was an average of 64 patient-sharing connections per facility, with a minimum of one connection and a maximum of 145 connections. Each additional patient two hospitals shared corresponded to a 3% increase in the CRE rate in urban facilities and a 6% increase in rural ones. The investigators didn’t explain the discrepancy, except to note that rural areas don’t have LTACHs.
Almost two-thirds of hospitals reporting CRE were in Chicago-area counties; almost half had shared at least one patient with an LTACH, and 21% had shared four or more.
CRE cases were an average of 64 years old, and equally distributed between men and women and black and white patients.
The Centers for Disease Control and Prevention funded the work. The authors had no disclosures.
The more hospitals share patients, the more likely they are to have a problem with carbapenem-resistant Enterobacteriaceae (CRE), especially if long-term acute care hospitals (LTACHs) are in the mix, according to a state-wide investigation from Illinois.
Greater hospital centrality was independently associated with higher rates overall, and sharing four or more patients with a long-term acute care hospital (LTACH) in the 3-month study window doubled the rate of CRE cases.
Although it’s possible that was because of chance (P = 0.11), the link between LTACHs and CRE “is consistent with prior analyses that have shown the central role LTACHs have in” spreading the organism, said the researchers, led by Michael Ray of the Illinois Department of Public Health (Clin Infect Dis. 2016 Aug 2. pii: ciw461).
Patients often spend weeks in LTACH facilities for ongoing, serious health problems. The severity of illness, long stay, and sometimes chronic antibiotic use increase the risk of CRE exposure, and the team found that many LTACH patients are colonized.
“These findings have immediate public health implications. … Early interventions should be focused on the most connected facilities, as well as those with strong connections to LTACHs.” When one hospital has an outbreak, facilities that share its patients need to swing into action screening new admissions and taking other steps to prevent regional spread, the team said.
Meanwhile, “state-wide patient-sharing data, which are now increasingly available through sources like the Healthcare Cost and Utilization Project, provide an important way to assess hospital risk of CRE exposure based on its position in regional patient-sharing networks,” they noted. “Public health can play a critical role in identifying tightly connected hospitals and educating personnel at such facilities about their risk and need for enhanced infection control interventions.”
The team came to their conclusions after linking Illinois’ drug-resistant organisms registry with admissions data for 185 hospitals. About half reported at least one CRE case over 3 months, with a mean of 3.5 cases per hospital.
There was an average of 64 patient-sharing connections per facility, with a minimum of one connection and a maximum of 145 connections. Each additional patient two hospitals shared corresponded to a 3% increase in the CRE rate in urban facilities and a 6% increase in rural ones. The investigators didn’t explain the discrepancy, except to note that rural areas don’t have LTACHs.
Almost two-thirds of hospitals reporting CRE were in Chicago-area counties; almost half had shared at least one patient with an LTACH, and 21% had shared four or more.
CRE cases were an average of 64 years old, and equally distributed between men and women and black and white patients.
The Centers for Disease Control and Prevention funded the work. The authors had no disclosures.
The more hospitals share patients, the more likely they are to have a problem with carbapenem-resistant Enterobacteriaceae (CRE), especially if long-term acute care hospitals (LTACHs) are in the mix, according to a state-wide investigation from Illinois.
Greater hospital centrality was independently associated with higher rates overall, and sharing four or more patients with a long-term acute care hospital (LTACH) in the 3-month study window doubled the rate of CRE cases.
Although it’s possible that was because of chance (P = 0.11), the link between LTACHs and CRE “is consistent with prior analyses that have shown the central role LTACHs have in” spreading the organism, said the researchers, led by Michael Ray of the Illinois Department of Public Health (Clin Infect Dis. 2016 Aug 2. pii: ciw461).
Patients often spend weeks in LTACH facilities for ongoing, serious health problems. The severity of illness, long stay, and sometimes chronic antibiotic use increase the risk of CRE exposure, and the team found that many LTACH patients are colonized.
“These findings have immediate public health implications. … Early interventions should be focused on the most connected facilities, as well as those with strong connections to LTACHs.” When one hospital has an outbreak, facilities that share its patients need to swing into action screening new admissions and taking other steps to prevent regional spread, the team said.
Meanwhile, “state-wide patient-sharing data, which are now increasingly available through sources like the Healthcare Cost and Utilization Project, provide an important way to assess hospital risk of CRE exposure based on its position in regional patient-sharing networks,” they noted. “Public health can play a critical role in identifying tightly connected hospitals and educating personnel at such facilities about their risk and need for enhanced infection control interventions.”
The team came to their conclusions after linking Illinois’ drug-resistant organisms registry with admissions data for 185 hospitals. About half reported at least one CRE case over 3 months, with a mean of 3.5 cases per hospital.
There was an average of 64 patient-sharing connections per facility, with a minimum of one connection and a maximum of 145 connections. Each additional patient two hospitals shared corresponded to a 3% increase in the CRE rate in urban facilities and a 6% increase in rural ones. The investigators didn’t explain the discrepancy, except to note that rural areas don’t have LTACHs.
Almost two-thirds of hospitals reporting CRE were in Chicago-area counties; almost half had shared at least one patient with an LTACH, and 21% had shared four or more.
CRE cases were an average of 64 years old, and equally distributed between men and women and black and white patients.
The Centers for Disease Control and Prevention funded the work. The authors had no disclosures.
FROM CLINICAL INFECTIOUS DISEASES
Key clinical point: The more hospitals share patients, the more likely they are to have a problem with CRE, especially if long-term acute care hospitals are in the mix.
Major finding: Sharing four or more patients with a long-term acute care hospital in the 3-month study window doubled the rate of CRE cases (P = 0.11).
Data source: 185 Illinois hospitals.
Disclosures: The Centers for Disease Control and Prevention funded the work. The authors had no disclosures.
European guideline for diagnosing C. difficile infection updated
The European Society of Clinical Microbiology and Infectious Diseases has updated its clinical guideline for diagnosing Clostridium difficile infection, according to a report in Clinical Microbiology and Infection.
“Our aim is to not only improve diagnosis of C. difficile infection, but also to standardize the diagnostic process across Europe to allow for improved surveillance of the disease,” Ed J. Kuijper, MD, of the Centre for Infectious Diseases, Leiden (the Netherlands) University Medical Centre and lead investigator for the new guideline, said in a press statement.
The Society released its first guideline regarding C. difficile diagnosis in 2009, but it required revision because many new diagnostic tests have become commercially available since then. The updated guideline focuses on diagnosing patients of all ages with diarrhea who are suspected of having C. difficile infection and is intended for use by medical microbiologists, gastroenterologists, infectious disease specialists, and infection control practitioners, said Monique J.T. Crobach, MD, who is also of Leiden University and is first author of the guideline, and her associates (Clin Microbiol Infect. 2016;22:S63-81).
They performed a comprehensive meta-analysis of 56 studies that compared 24 commercially available diagnostic assays against either of the current “gold standard” tests, the cell cytotoxicity neutralization assay or the toxigenic culture. Forty-one of these studies were published after 2009. Based on their findings, Dr. Crobach and her associates formulated 16 recommendations and suggestions, noting the quality of evidence supporting each one.
The updated guideline strongly recommends against using any single diagnostic assay to diagnose C. difficile infection, regardless of the technology on which it is based. Instead, diagnosis should rest on clinical signs and symptoms together with a two-step algorithm starting with either a nucleic acid amplification test or a glutamate dehydrogenase enzyme immunoassay. Samples with positive results on this initial test should be tested further with a toxin A/B enzyme immunoassay.
An alternative algorithm is to initially test samples with both a glutamate dehydrogenase and a toxin A/B enzyme immunoassay. The guideline spells out what actions to take in the event of concordant positive results, concordant negative results, or discordant results.
Testing for C. difficile should not be limited to samples with a specific request from a physician. All unformed stool samples from patients aged 3 years and older should be tested for the organism. In contrast, formed stool samples shouldn’t be tested for C. difficile unless the patient has paralytic ileus.
The guideline also addresses repeat testing. Performing a repeat test after an initial negative result during the same diarrheal episode may be useful in selected cases with ongoing clinical suspicion during an epidemic situation, or even in cases with high clinical suspicion during endemic situations. But repeat testing after an initial positive result generally is not recommended. Repeat testing to assess whether the patient is cured is definitely not recommended.
However, the decision to treat patients for C. difficile infection is a clinical one and may be justified even if all laboratory results are negative, Dr. Crobach and her associates noted.
The European Society of Clinical Microbiology and Infectious Diseases has updated its clinical guideline for diagnosing Clostridium difficile infection, according to a report in Clinical Microbiology and Infection.
“Our aim is to not only improve diagnosis of C. difficile infection, but also to standardize the diagnostic process across Europe to allow for improved surveillance of the disease,” Ed J. Kuijper, MD, of the Centre for Infectious Diseases, Leiden (the Netherlands) University Medical Centre and lead investigator for the new guideline, said in a press statement.
The Society released its first guideline regarding C. difficile diagnosis in 2009, but it required revision because many new diagnostic tests have become commercially available since then. The updated guideline focuses on diagnosing patients of all ages with diarrhea who are suspected of having C. difficile infection and is intended for use by medical microbiologists, gastroenterologists, infectious disease specialists, and infection control practitioners, said Monique J.T. Crobach, MD, who is also of Leiden University and is first author of the guideline, and her associates (Clin Microbiol Infect. 2016;22:S63-81).
They performed a comprehensive meta-analysis of 56 studies that compared 24 commercially available diagnostic assays against either of the current “gold standard” tests, the cell cytotoxicity neutralization assay or the toxigenic culture. Forty-one of these studies were published after 2009. Based on their findings, Dr. Crobach and her associates formulated 16 recommendations and suggestions, noting the quality of evidence supporting each one.
The updated guideline strongly recommends against using any single diagnostic assay to diagnose C. difficile infection, regardless of the technology on which it is based. Instead, diagnosis should rest on clinical signs and symptoms together with a two-step algorithm starting with either a nucleic acid amplification test or a glutamate dehydrogenase enzyme immunoassay. Samples with positive results on this initial test should be tested further with a toxin A/B enzyme immunoassay.
An alternative algorithm is to initially test samples with both a glutamate dehydrogenase and a toxin A/B enzyme immunoassay. The guideline spells out what actions to take in the event of concordant positive results, concordant negative results, or discordant results.
Testing for C. difficile should not be limited to samples with a specific request from a physician. All unformed stool samples from patients aged 3 years and older should be tested for the organism. In contrast, formed stool samples shouldn’t be tested for C. difficile unless the patient has paralytic ileus.
The guideline also addresses repeat testing. Performing a repeat test after an initial negative result during the same diarrheal episode may be useful in selected cases with ongoing clinical suspicion during an epidemic situation, or even in cases with high clinical suspicion during endemic situations. But repeat testing after an initial positive result generally is not recommended. Repeat testing to assess whether the patient is cured is definitely not recommended.
However, the decision to treat patients for C. difficile infection is a clinical one and may be justified even if all laboratory results are negative, Dr. Crobach and her associates noted.
The European Society of Clinical Microbiology and Infectious Diseases has updated its clinical guideline for diagnosing Clostridium difficile infection, according to a report in Clinical Microbiology and Infection.
“Our aim is to not only improve diagnosis of C. difficile infection, but also to standardize the diagnostic process across Europe to allow for improved surveillance of the disease,” Ed J. Kuijper, MD, of the Centre for Infectious Diseases, Leiden (the Netherlands) University Medical Centre and lead investigator for the new guideline, said in a press statement.
The Society released its first guideline regarding C. difficile diagnosis in 2009, but it required revision because many new diagnostic tests have become commercially available since then. The updated guideline focuses on diagnosing patients of all ages with diarrhea who are suspected of having C. difficile infection and is intended for use by medical microbiologists, gastroenterologists, infectious disease specialists, and infection control practitioners, said Monique J.T. Crobach, MD, who is also of Leiden University and is first author of the guideline, and her associates (Clin Microbiol Infect. 2016;22:S63-81).
They performed a comprehensive meta-analysis of 56 studies that compared 24 commercially available diagnostic assays against either of the current “gold standard” tests, the cell cytotoxicity neutralization assay or the toxigenic culture. Forty-one of these studies were published after 2009. Based on their findings, Dr. Crobach and her associates formulated 16 recommendations and suggestions, noting the quality of evidence supporting each one.
The updated guideline strongly recommends against using any single diagnostic assay to diagnose C. difficile infection, regardless of the technology on which it is based. Instead, diagnosis should rest on clinical signs and symptoms together with a two-step algorithm starting with either a nucleic acid amplification test or a glutamate dehydrogenase enzyme immunoassay. Samples with positive results on this initial test should be tested further with a toxin A/B enzyme immunoassay.
An alternative algorithm is to initially test samples with both a glutamate dehydrogenase and a toxin A/B enzyme immunoassay. The guideline spells out what actions to take in the event of concordant positive results, concordant negative results, or discordant results.
Testing for C. difficile should not be limited to samples with a specific request from a physician. All unformed stool samples from patients aged 3 years and older should be tested for the organism. In contrast, formed stool samples shouldn’t be tested for C. difficile unless the patient has paralytic ileus.
The guideline also addresses repeat testing. Performing a repeat test after an initial negative result during the same diarrheal episode may be useful in selected cases with ongoing clinical suspicion during an epidemic situation, or even in cases with high clinical suspicion during endemic situations. But repeat testing after an initial positive result generally is not recommended. Repeat testing to assess whether the patient is cured is definitely not recommended.
However, the decision to treat patients for C. difficile infection is a clinical one and may be justified even if all laboratory results are negative, Dr. Crobach and her associates noted.
FROM CLINICAL MICROBIOLOGY AND INFECTION
Key clinical point: The European Society of Clinical Microbiology and Infectious Diseases updated its guideline for diagnosing C. difficile infection.
Major finding: No single diagnostic assay should be used to diagnose C. difficile infection; diagnosis should be based on clinical signs and symptoms together with a two-step algorithm starting with either a nucleic acid amplification test or a glutamate dehydrogenase enzyme immunoassay.
Data source: A meta-analysis of 56 studies comparing the efficacy of 24 laboratory assays for diagnosing C. difficile, and a compilation of testing guidelines.
Disclosures: The meta-analysis was supported by the European Society of Clinical Microbiology and Infectious Diseases. Dr. Crobach and her associates reported having no relevant financial disclosures.
Inflammatory bowel disease
For the Spring Postgraduate Course session on inflammatory bowel disease (IBD), we were fortunate to secure four of the best educators on IBD in the American Gastroenterological Association.
William Sandborn, MD, of the University of California, San Diego, led the session with an overview of the immunomodulators and biologics that are employed for the treatment of Crohn’s disease and ulcerative colitis in 2016, including the thiopurines, methotrexate, calcineurin inhibitors, anti–tumor necrosis factor (TNF) agents, anti-integrins, and upcoming therapies such as ustekinumab (anti-interleukins 12 and 23) and tofacitinib (Janus kinase antagonist).
The evidence base for thiopurine monotherapy in IBD is weaker than we had once assumed, and the combination of infliximab and azathioprine remains the gold standard in efficacy with respect to steroid-free remission. Methotrexate remains a reasonable option as an immunomodulator, especially for patients who can’t tolerate thiopurines or who are risk averse. Cyclosporine has a niche indication for the treatment of acute severe colitis, and is at least equivalent to infliximab for that indication.
We have a total of four anti-TNF agents approved for the treatment of moderate to severe Crohn’s disease and ulcerative colitis, including infliximab, adalimumab, certolizumab pegol (Crohn’s only), and golimumab (ulcerative colitis only). The infliximab biosimilar CT-P13 is approved for IBD in several countries. The anti-integrin, vedolizumab, is approved for both moderate to severe ulcerative colitis and Crohn’s disease. Ustekinumab was shown to be more effective than placebo in inducing clinical response and remission in moderate to severe Crohn’s disease patients who were either refractory/intolerant to or naive to anti-TNF therapy. Tofacitinib was recently shown in two trials to be more effective than placebo in inducing clinical remission and mucosal healing in patients with moderate to severe ulcerative colitis.
Maria Abreu, MD, from the University of Miami highlighted the fact that although our current therapies are far more effective than previous ones, they can be problematic, and dose adjustment is often needed. We should tailor our treatment plan based on our assessment of the patient’s risk for intestinal complications or surgery – patients with multiple risk factors such as young age at diagnosis, extensive involvement, deep ulcers, previous surgeries, or penetrating/stricturing behavior at baseline should be treated more aggressively, perhaps with a top-down approach. In general, biologics will be more effective when used in combination with immunomodulators. Monotherapy can be considered in patients with low inflammatory burden, few risk factors for more severe disease, or in those at high risk for complications from combination therapy.
At least one way in which concomitant immunomodulator therapy exerts its beneficial effect is by increasing trough levels of the biologic. One recent study of the relationship between adalimumab levels and endoscopic inflammation suggested that the median adalimumab trough level associated with mucosal healing was 13.5 mcg/mL. Patients with low drug levels and no or low levels of anti-drug antibodies should undergo dose escalation. Those with absent drug levels and high levels of antibodies should switch to a different agent. If the drug level is considered adequate and yet inflammation is still present, consideration should be given to switching to a biologic with a different mechanism of action. In some patients with low levels of antibodies who are on monotherapy, there may be a role for adding an immunomodulator in an attempt to suppress antibody formation. The importance of vedolizumab and ustekinumab as biologics with different mechanisms of action was also highlighted. An exciting class of biologics currently under development is the anti–IL-23 class, including drugs such as risankizumab and MEDI-2070.
David T. Rubin, MD, from the University of Chicago reviewed the various complications associated with IBD and its therapies. Disease-related complications include clinical flares, bowel obstruction, fistula/abscess formation, intestinal cancer, the need for surgery (sometimes more than one), and extraintestinal manifestations. More accurate diagnosis and earlier effective therapies may help to reduce the risk of these complications. Certain infections such as herpes zoster, pneumonia, and Clostridium difficile infection appear to occur more commonly in IBD patients. The TREAT registry showed that the use of corticosteroids is associated with an increased risk of serious infections and mortality, and the use of infliximab is associated with an increased risk of serious infection. C. difficile infection in particular can be problematic, and should be treated aggressively. Thiopurines appear to increase the risk of lymphoma and nonmelanoma skin cancer. Lymphoma risk quickly reverts to baseline after thiopurines are discontinued. Overall, no increased risk of cancer can be demonstrated among patients on anti-TNF therapy, and patients with a history of cancer do not have an increased risk of recurrent or new cancers on anti-TNF therapy. There may be an association between anti-TNF use and melanoma, although this bears further study.
Miguel Regueiro, MD, from the University of Pittsburgh wrapped up the session with a talk on managing postoperative IBD patients. Although the ileal pouch–anal anastomosis (IPAA) broadened access to proctocolectomy, it is associated with several disorders, including pelvic sepsis, pouchitis, Crohn’s disease of the ileoanal pouch, cuffitis, and irritable pouch syndrome. At least 50% of IPAA patients will develop at least one episode of pouchitis. Most patients will respond to ciprofloxacin or metronidazole, but a small percentage will develop chronic pouchitis. Ileocecal resection for the complications of Crohn’s disease generally results in immediate improvements in quality of life, but unfortunately, most patients will develop endoscopic and eventually clinical recurrence. At 6-12 months after surgery, a Rutgeerts score of i2 or higher (at least five aphthous lesions in the neoterminal ileum) is associated with a high risk of clinical recurrence, and this finding can be used as a trigger for escalation of therapy.
Infliximab was associated with a markedly decreased risk of endoscopic recurrence in a small investigator-initiated trial, and subsequent studies have strongly suggested that infliximab and adalimumab reduce the risk of recurrence. Although the primary endpoint of clinical recurrence was not met in the large multicenter PREVENT trial, infliximab was shown to significantly decrease endoscopic recurrence. The POCER study showed that a slightly less proactive approach of watchful waiting in lower-risk patients and making decisions about therapy at the 6-month colonoscopy was a reasonable one. The risk factors one must consider in stratifying postop patients include early age at surgery, short time between diagnosis and surgery, cigarette smoking, penetrating disease behavior, and a history of previous resections.
Dr. Loftus is professor of medicine in the division of gastroenterology and hepatology, Mayo Clinic, Rochester, Minn. He has consulted for and has received research support from Janssen, UCB, Takeda, and AbbVie.
This is a summary provided by the moderator of one of the spring postgraduate course sessions held at DDW 2016.
For the Spring Postgraduate Course session on inflammatory bowel disease (IBD), we were fortunate to secure four of the best educators on IBD in the American Gastroenterological Association.
William Sandborn, MD, of the University of California, San Diego, led the session with an overview of the immunomodulators and biologics that are employed for the treatment of Crohn’s disease and ulcerative colitis in 2016, including the thiopurines, methotrexate, calcineurin inhibitors, anti–tumor necrosis factor (TNF) agents, anti-integrins, and upcoming therapies such as ustekinumab (anti-interleukins 12 and 23) and tofacitinib (Janus kinase antagonist).
The evidence base for thiopurine monotherapy in IBD is weaker than we had once assumed, and the combination of infliximab and azathioprine remains the gold standard in efficacy with respect to steroid-free remission. Methotrexate remains a reasonable option as an immunomodulator, especially for patients who can’t tolerate thiopurines or who are risk averse. Cyclosporine has a niche indication for the treatment of acute severe colitis, and is at least equivalent to infliximab for that indication.
We have a total of four anti-TNF agents approved for the treatment of moderate to severe Crohn’s disease and ulcerative colitis, including infliximab, adalimumab, certolizumab pegol (Crohn’s only), and golimumab (ulcerative colitis only). The infliximab biosimilar CT-P13 is approved for IBD in several countries. The anti-integrin, vedolizumab, is approved for both moderate to severe ulcerative colitis and Crohn’s disease. Ustekinumab was shown to be more effective than placebo in inducing clinical response and remission in moderate to severe Crohn’s disease patients who were either refractory/intolerant to or naive to anti-TNF therapy. Tofacitinib was recently shown in two trials to be more effective than placebo in inducing clinical remission and mucosal healing in patients with moderate to severe ulcerative colitis.
Maria Abreu, MD, from the University of Miami highlighted the fact that although our current therapies are far more effective than previous ones, they can be problematic, and dose adjustment is often needed. We should tailor our treatment plan based on our assessment of the patient’s risk for intestinal complications or surgery – patients with multiple risk factors such as young age at diagnosis, extensive involvement, deep ulcers, previous surgeries, or penetrating/stricturing behavior at baseline should be treated more aggressively, perhaps with a top-down approach. In general, biologics will be more effective when used in combination with immunomodulators. Monotherapy can be considered in patients with low inflammatory burden, few risk factors for more severe disease, or in those at high risk for complications from combination therapy.
At least one way in which concomitant immunomodulator therapy exerts its beneficial effect is by increasing trough levels of the biologic. One recent study of the relationship between adalimumab levels and endoscopic inflammation suggested that the median adalimumab trough level associated with mucosal healing was 13.5 mcg/mL. Patients with low drug levels and no or low levels of anti-drug antibodies should undergo dose escalation. Those with absent drug levels and high levels of antibodies should switch to a different agent. If the drug level is considered adequate and yet inflammation is still present, consideration should be given to switching to a biologic with a different mechanism of action. In some patients with low levels of antibodies who are on monotherapy, there may be a role for adding an immunomodulator in an attempt to suppress antibody formation. The importance of vedolizumab and ustekinumab as biologics with different mechanisms of action was also highlighted. An exciting class of biologics currently under development is the anti–IL-23 class, including drugs such as risankizumab and MEDI-2070.
David T. Rubin, MD, from the University of Chicago reviewed the various complications associated with IBD and its therapies. Disease-related complications include clinical flares, bowel obstruction, fistula/abscess formation, intestinal cancer, the need for surgery (sometimes more than one), and extraintestinal manifestations. More accurate diagnosis and earlier effective therapies may help to reduce the risk of these complications. Certain infections such as herpes zoster, pneumonia, and Clostridium difficile infection appear to occur more commonly in IBD patients. The TREAT registry showed that the use of corticosteroids is associated with an increased risk of serious infections and mortality, and the use of infliximab is associated with an increased risk of serious infection. C. difficile infection in particular can be problematic, and should be treated aggressively. Thiopurines appear to increase the risk of lymphoma and nonmelanoma skin cancer. Lymphoma risk quickly reverts to baseline after thiopurines are discontinued. Overall, no increased risk of cancer can be demonstrated among patients on anti-TNF therapy, and patients with a history of cancer do not have an increased risk of recurrent or new cancers on anti-TNF therapy. There may be an association between anti-TNF use and melanoma, although this bears further study.
Miguel Regueiro, MD, from the University of Pittsburgh wrapped up the session with a talk on managing postoperative IBD patients. Although the ileal pouch–anal anastomosis (IPAA) broadened access to proctocolectomy, it is associated with several disorders, including pelvic sepsis, pouchitis, Crohn’s disease of the ileoanal pouch, cuffitis, and irritable pouch syndrome. At least 50% of IPAA patients will develop at least one episode of pouchitis. Most patients will respond to ciprofloxacin or metronidazole, but a small percentage will develop chronic pouchitis. Ileocecal resection for the complications of Crohn’s disease generally results in immediate improvements in quality of life, but unfortunately, most patients will develop endoscopic and eventually clinical recurrence. At 6-12 months after surgery, a Rutgeerts score of i2 or higher (at least five aphthous lesions in the neoterminal ileum) is associated with a high risk of clinical recurrence, and this finding can be used as a trigger for escalation of therapy.
Infliximab was associated with a markedly decreased risk of endoscopic recurrence in a small investigator-initiated trial, and subsequent studies have strongly suggested that infliximab and adalimumab reduce the risk of recurrence. Although the primary endpoint of clinical recurrence was not met in the large multicenter PREVENT trial, infliximab was shown to significantly decrease endoscopic recurrence. The POCER study showed that a slightly less proactive approach of watchful waiting in lower-risk patients and making decisions about therapy at the 6-month colonoscopy was a reasonable one. The risk factors one must consider in stratifying postop patients include early age at surgery, short time between diagnosis and surgery, cigarette smoking, penetrating disease behavior, and a history of previous resections.
Dr. Loftus is professor of medicine in the division of gastroenterology and hepatology, Mayo Clinic, Rochester, Minn. He has consulted for and has received research support from Janssen, UCB, Takeda, and AbbVie.
This is a summary provided by the moderator of one of the spring postgraduate course sessions held at DDW 2016.
For the Spring Postgraduate Course session on inflammatory bowel disease (IBD), we were fortunate to secure four of the best educators on IBD in the American Gastroenterological Association.
William Sandborn, MD, of the University of California, San Diego, led the session with an overview of the immunomodulators and biologics that are employed for the treatment of Crohn’s disease and ulcerative colitis in 2016, including the thiopurines, methotrexate, calcineurin inhibitors, anti–tumor necrosis factor (TNF) agents, anti-integrins, and upcoming therapies such as ustekinumab (anti-interleukins 12 and 23) and tofacitinib (Janus kinase antagonist).
The evidence base for thiopurine monotherapy in IBD is weaker than we had once assumed, and the combination of infliximab and azathioprine remains the gold standard in efficacy with respect to steroid-free remission. Methotrexate remains a reasonable option as an immunomodulator, especially for patients who can’t tolerate thiopurines or who are risk averse. Cyclosporine has a niche indication for the treatment of acute severe colitis, and is at least equivalent to infliximab for that indication.
We have a total of four anti-TNF agents approved for the treatment of moderate to severe Crohn’s disease and ulcerative colitis, including infliximab, adalimumab, certolizumab pegol (Crohn’s only), and golimumab (ulcerative colitis only). The infliximab biosimilar CT-P13 is approved for IBD in several countries. The anti-integrin, vedolizumab, is approved for both moderate to severe ulcerative colitis and Crohn’s disease. Ustekinumab was shown to be more effective than placebo in inducing clinical response and remission in moderate to severe Crohn’s disease patients who were either refractory/intolerant to or naive to anti-TNF therapy. Tofacitinib was recently shown in two trials to be more effective than placebo in inducing clinical remission and mucosal healing in patients with moderate to severe ulcerative colitis.
Maria Abreu, MD, from the University of Miami highlighted the fact that although our current therapies are far more effective than previous ones, they can be problematic, and dose adjustment is often needed. We should tailor our treatment plan based on our assessment of the patient’s risk for intestinal complications or surgery – patients with multiple risk factors such as young age at diagnosis, extensive involvement, deep ulcers, previous surgeries, or penetrating/stricturing behavior at baseline should be treated more aggressively, perhaps with a top-down approach. In general, biologics will be more effective when used in combination with immunomodulators. Monotherapy can be considered in patients with low inflammatory burden, few risk factors for more severe disease, or in those at high risk for complications from combination therapy.
At least one way in which concomitant immunomodulator therapy exerts its beneficial effect is by increasing trough levels of the biologic. One recent study of the relationship between adalimumab levels and endoscopic inflammation suggested that the median adalimumab trough level associated with mucosal healing was 13.5 mcg/mL. Patients with low drug levels and no or low levels of anti-drug antibodies should undergo dose escalation. Those with absent drug levels and high levels of antibodies should switch to a different agent. If the drug level is considered adequate and yet inflammation is still present, consideration should be given to switching to a biologic with a different mechanism of action. In some patients with low levels of antibodies who are on monotherapy, there may be a role for adding an immunomodulator in an attempt to suppress antibody formation. The importance of vedolizumab and ustekinumab as biologics with different mechanisms of action was also highlighted. An exciting class of biologics currently under development is the anti–IL-23 class, including drugs such as risankizumab and MEDI-2070.
David T. Rubin, MD, from the University of Chicago reviewed the various complications associated with IBD and its therapies. Disease-related complications include clinical flares, bowel obstruction, fistula/abscess formation, intestinal cancer, the need for surgery (sometimes more than one), and extraintestinal manifestations. More accurate diagnosis and earlier effective therapies may help to reduce the risk of these complications. Certain infections such as herpes zoster, pneumonia, and Clostridium difficile infection appear to occur more commonly in IBD patients. The TREAT registry showed that the use of corticosteroids is associated with an increased risk of serious infections and mortality, and the use of infliximab is associated with an increased risk of serious infection. C. difficile infection in particular can be problematic, and should be treated aggressively. Thiopurines appear to increase the risk of lymphoma and nonmelanoma skin cancer. Lymphoma risk quickly reverts to baseline after thiopurines are discontinued. Overall, no increased risk of cancer can be demonstrated among patients on anti-TNF therapy, and patients with a history of cancer do not have an increased risk of recurrent or new cancers on anti-TNF therapy. There may be an association between anti-TNF use and melanoma, although this bears further study.
Miguel Regueiro, MD, from the University of Pittsburgh wrapped up the session with a talk on managing postoperative IBD patients. Although the ileal pouch–anal anastomosis (IPAA) broadened access to proctocolectomy, it is associated with several disorders, including pelvic sepsis, pouchitis, Crohn’s disease of the ileoanal pouch, cuffitis, and irritable pouch syndrome. At least 50% of IPAA patients will develop at least one episode of pouchitis. Most patients will respond to ciprofloxacin or metronidazole, but a small percentage will develop chronic pouchitis. Ileocecal resection for the complications of Crohn’s disease generally results in immediate improvements in quality of life, but unfortunately, most patients will develop endoscopic and eventually clinical recurrence. At 6-12 months after surgery, a Rutgeerts score of i2 or higher (at least five aphthous lesions in the neoterminal ileum) is associated with a high risk of clinical recurrence, and this finding can be used as a trigger for escalation of therapy.
Infliximab was associated with a markedly decreased risk of endoscopic recurrence in a small investigator-initiated trial, and subsequent studies have strongly suggested that infliximab and adalimumab reduce the risk of recurrence. Although the primary endpoint of clinical recurrence was not met in the large multicenter PREVENT trial, infliximab was shown to significantly decrease endoscopic recurrence. The POCER study showed that a slightly less proactive approach of watchful waiting in lower-risk patients and making decisions about therapy at the 6-month colonoscopy was a reasonable one. The risk factors one must consider in stratifying postop patients include early age at surgery, short time between diagnosis and surgery, cigarette smoking, penetrating disease behavior, and a history of previous resections.
Dr. Loftus is professor of medicine in the division of gastroenterology and hepatology, Mayo Clinic, Rochester, Minn. He has consulted for and has received research support from Janssen, UCB, Takeda, and AbbVie.
This is a summary provided by the moderator of one of the spring postgraduate course sessions held at DDW 2016.
Strange bedfellows: FMT and esophageal disease
Fecal microbiota transplant (FMT) and esophageal disease were strange bedfellows in a series of informative lectures by world experts in their respective fields.
FMT breaks the cycle of resistant Clostridium difficile infection, where resistant bacteria overwhelm innate gut flora following antibiotic use. FMT introduces competition for nutrients and microbiota-derived bacteriocins targeting resistant bacteria, restores secondary bile salt metabolism, and stimulates mucosal immunity against resistant bacteria. The potential for FMT to benefit diseases other than recurrent C. difficile infection continues to be explored.
Eosinophilic esophagitis (EoE) is characterized clinically by dysphagia and food impaction, and pathologically by eosinophil-dominant inflammation (at least 15 eosinophils/high-power field on biopsy). Esophageal subepithelial fibrosis contributes to a narrow caliber, poorly distensible esophagus. Alternative causes of esophageal eosinophilia, particularly that induced by reflux, need to be excluded. Proton pump inhibitor (PPI) response does not distinguish EoE from reflux disease, but management starts with these drugs. Swallowed topical steroids, and the six-food elimination diet are alternative effective therapies. Biologic agents are being evaluated as future therapeutic options.
High-resolution manometry (HRM) has improved acquisition and display of esophageal pressure data, simplifying interpretation using three software tools, integrated relaxation pressure, distal contractile integral, and distal latency. Stationary impedance with HRM (high-resolution impedance manometry, HRIM) provides further gains in esophageal bolus transit assessment. Automated impedance manometry analysis, esophageal impedance integral ratio, bolus flow time, and functional lumen imaging probe–derived metrics add to esophageal physiologic assessments and esophageal function testing.
Rare disorders such as esophageal lichen planus can manifest with dysphagia and esophageal inflammation and strictures; these disorders are managed with immunosuppressive agents and cautious endoscopic dilation. Rumination (regurgitation of recently ingested food) and belching (with aerophagy) mimic reflux disease. These are distinguished using HRIM and pH-impedance monitoring, and treated with diaphragmatic breathing. Early postfundoplication dysphagia is common and responds to dilation; peptic strictures or slipped fundoplication needing wrap revision can cause late dysphagia. Scleroderma esophagus can be difficult to differentiate from advanced achalasia. Pneumatic dilation can benefit postmyotomy dysphagia.
Low-grade dysplasia within Barrett’s esophagus has a variable natural history, primarily from overdiagnosis and interobserver variation; many patients are down-staged upon review. Progression to high-grade dysplasia or intramucosal cancer (0.5%-1.7% per annum) is higher with confirmed low-grade dysplasia. Consequently, guidelines recommend review by expert pathologists and surveillance after 6-12 months. Evidence for radiofrequency ablation in carefully selected patients continues to grow. Radiofrequency ablation eliminates dysplasia and reduces the risk of progression to high-grade dysplasia. Better markers for diagnosis and prognosis continue to be studied.
Dr. Gyawali is professor of medicine, division of gastroenterology, Washington University in St. Louis. He has consulted for and received speaking fees or research funding from Medtronic, Torax, Ironwood, and Allergan.
This is a summary provided by the moderator of one of the spring postgraduate course sessions held at DDW 2016.
Fecal microbiota transplant (FMT) and esophageal disease were strange bedfellows in a series of informative lectures by world experts in their respective fields.
FMT breaks the cycle of resistant Clostridium difficile infection, where resistant bacteria overwhelm innate gut flora following antibiotic use. FMT introduces competition for nutrients and microbiota-derived bacteriocins targeting resistant bacteria, restores secondary bile salt metabolism, and stimulates mucosal immunity against resistant bacteria. The potential for FMT to benefit diseases other than recurrent C. difficile infection continues to be explored.
Eosinophilic esophagitis (EoE) is characterized clinically by dysphagia and food impaction, and pathologically by eosinophil-dominant inflammation (at least 15 eosinophils/high-power field on biopsy). Esophageal subepithelial fibrosis contributes to a narrow caliber, poorly distensible esophagus. Alternative causes of esophageal eosinophilia, particularly that induced by reflux, need to be excluded. Proton pump inhibitor (PPI) response does not distinguish EoE from reflux disease, but management starts with these drugs. Swallowed topical steroids, and the six-food elimination diet are alternative effective therapies. Biologic agents are being evaluated as future therapeutic options.
High-resolution manometry (HRM) has improved acquisition and display of esophageal pressure data, simplifying interpretation using three software tools, integrated relaxation pressure, distal contractile integral, and distal latency. Stationary impedance with HRM (high-resolution impedance manometry, HRIM) provides further gains in esophageal bolus transit assessment. Automated impedance manometry analysis, esophageal impedance integral ratio, bolus flow time, and functional lumen imaging probe–derived metrics add to esophageal physiologic assessments and esophageal function testing.
Rare disorders such as esophageal lichen planus can manifest with dysphagia and esophageal inflammation and strictures; these disorders are managed with immunosuppressive agents and cautious endoscopic dilation. Rumination (regurgitation of recently ingested food) and belching (with aerophagy) mimic reflux disease. These are distinguished using HRIM and pH-impedance monitoring, and treated with diaphragmatic breathing. Early postfundoplication dysphagia is common and responds to dilation; peptic strictures or slipped fundoplication needing wrap revision can cause late dysphagia. Scleroderma esophagus can be difficult to differentiate from advanced achalasia. Pneumatic dilation can benefit postmyotomy dysphagia.
Low-grade dysplasia within Barrett’s esophagus has a variable natural history, primarily from overdiagnosis and interobserver variation; many patients are down-staged upon review. Progression to high-grade dysplasia or intramucosal cancer (0.5%-1.7% per annum) is higher with confirmed low-grade dysplasia. Consequently, guidelines recommend review by expert pathologists and surveillance after 6-12 months. Evidence for radiofrequency ablation in carefully selected patients continues to grow. Radiofrequency ablation eliminates dysplasia and reduces the risk of progression to high-grade dysplasia. Better markers for diagnosis and prognosis continue to be studied.
Dr. Gyawali is professor of medicine, division of gastroenterology, Washington University in St. Louis. He has consulted for and received speaking fees or research funding from Medtronic, Torax, Ironwood, and Allergan.
This is a summary provided by the moderator of one of the spring postgraduate course sessions held at DDW 2016.
Fecal microbiota transplant (FMT) and esophageal disease were strange bedfellows in a series of informative lectures by world experts in their respective fields.
FMT breaks the cycle of resistant Clostridium difficile infection, where resistant bacteria overwhelm innate gut flora following antibiotic use. FMT introduces competition for nutrients and microbiota-derived bacteriocins targeting resistant bacteria, restores secondary bile salt metabolism, and stimulates mucosal immunity against resistant bacteria. The potential for FMT to benefit diseases other than recurrent C. difficile infection continues to be explored.
Eosinophilic esophagitis (EoE) is characterized clinically by dysphagia and food impaction, and pathologically by eosinophil-dominant inflammation (at least 15 eosinophils/high-power field on biopsy). Esophageal subepithelial fibrosis contributes to a narrow caliber, poorly distensible esophagus. Alternative causes of esophageal eosinophilia, particularly that induced by reflux, need to be excluded. Proton pump inhibitor (PPI) response does not distinguish EoE from reflux disease, but management starts with these drugs. Swallowed topical steroids, and the six-food elimination diet are alternative effective therapies. Biologic agents are being evaluated as future therapeutic options.
High-resolution manometry (HRM) has improved acquisition and display of esophageal pressure data, simplifying interpretation using three software tools, integrated relaxation pressure, distal contractile integral, and distal latency. Stationary impedance with HRM (high-resolution impedance manometry, HRIM) provides further gains in esophageal bolus transit assessment. Automated impedance manometry analysis, esophageal impedance integral ratio, bolus flow time, and functional lumen imaging probe–derived metrics add to esophageal physiologic assessments and esophageal function testing.
Rare disorders such as esophageal lichen planus can manifest with dysphagia and esophageal inflammation and strictures; these disorders are managed with immunosuppressive agents and cautious endoscopic dilation. Rumination (regurgitation of recently ingested food) and belching (with aerophagy) mimic reflux disease. These are distinguished using HRIM and pH-impedance monitoring, and treated with diaphragmatic breathing. Early postfundoplication dysphagia is common and responds to dilation; peptic strictures or slipped fundoplication needing wrap revision can cause late dysphagia. Scleroderma esophagus can be difficult to differentiate from advanced achalasia. Pneumatic dilation can benefit postmyotomy dysphagia.
Low-grade dysplasia within Barrett’s esophagus has a variable natural history, primarily from overdiagnosis and interobserver variation; many patients are down-staged upon review. Progression to high-grade dysplasia or intramucosal cancer (0.5%-1.7% per annum) is higher with confirmed low-grade dysplasia. Consequently, guidelines recommend review by expert pathologists and surveillance after 6-12 months. Evidence for radiofrequency ablation in carefully selected patients continues to grow. Radiofrequency ablation eliminates dysplasia and reduces the risk of progression to high-grade dysplasia. Better markers for diagnosis and prognosis continue to be studied.
Dr. Gyawali is professor of medicine, division of gastroenterology, Washington University in St. Louis. He has consulted for and received speaking fees or research funding from Medtronic, Torax, Ironwood, and Allergan.
This is a summary provided by the moderator of one of the spring postgraduate course sessions held at DDW 2016.