User login
Outpatient diverticulitis therapy cures patients, saves money
Discharging patients with uncomplicated diverticulitis from the emergency department on antibiotics was just as effective as hospitalization – and cost about a third as much, based on a study conducted in Spain.
Dr. Sebastiano Biondi and his colleagues conducted their randomized, parallel-group study at five hospitals. The cohort consisted of 132 adults, mean age 56 years, who presented to the emergency department with localized abdominal pain. All had an abdominal CT scan and received intravenous antibiotics, either an initial IV infusion of 1 g of amoxicillin per 125 mg of clavulanic acid, or, in the case of those with a penicillin allergy, ciprofloxacin 200 mg and metronidazole 500 mg.
Half of the patients were then assigned to either inpatient or outpatient antibiotic treatment. The admitted patients continued to receive IV antibiotics and fluids for 36-48 hours until they tolerated oral feeding. The outpatients were discharged on oral amoxicillin and clavulanic acid (875 mg per 125 mg every 8 hours) or, in those with penicillin allergy, the combination of ciprofloxacin (500 mg every 12 hours) and metronidazole (500 mg every 8 hours).
The study’s main endpoint was treatment failure rate (persistence, increase, or recurrence of abdominal pain and/or fever; inflammatory bowel obstruction; or the need for radiological abscess drainage or immediate surgery due to complicated diverticulitis). Patients were followed every day for 5 days, and then interviewed on day 14. Before the final follow-up at 60 days, patients underwent a colonoscopy to rule out malignancy.
Seven patients (5%) were readmitted due to treatment failure: 4 (6%) in the inpatient group and three (4.5%) in the outpatient group. The difference was not statistically significant. No one needed emergency surgery as part of readmission, and there were no deaths.
A quality of life assessment found that hospitalized patients reported a significantly higher level of physical health during the first 2 weeks of treatment, but that difference disappeared after 14 days.
Total treatment costs were significantly less in the outpatient group, with an average savings of $1,840 per patient ($895 for outpatient care compared with $2,735 for inpatient care). Almost all of that cost difference was due to hospital bed cost, with an average stay of 4 days.
"The outpatient protocol of this study is applicable to a selected group of patients with uncomplicated diverticulitis," Dr. Biondi reported in the January issue of Annals of Surgery (doi: 10.1097/SLA.0b013e3182965a11).
Widespread adoption of outpatient treatment could have a profound effect on the cost of treating diverticulitis, an ever more common gastrointestinal problem, said Dr. Biondi of the University of Barcelona and his coauthors. "According to data from the National Hospital Discharge Survey, diverticular disease is responsible for 314,000 hospital admissions per year in the United States, and the estimated annual cost in 1998 was about $2.6 billion."
The Spanish Ministry of Health funded the trial. The authors had no financial conflicts.
Discharging patients with uncomplicated diverticulitis from the emergency department on antibiotics was just as effective as hospitalization – and cost about a third as much, based on a study conducted in Spain.
Dr. Sebastiano Biondi and his colleagues conducted their randomized, parallel-group study at five hospitals. The cohort consisted of 132 adults, mean age 56 years, who presented to the emergency department with localized abdominal pain. All had an abdominal CT scan and received intravenous antibiotics, either an initial IV infusion of 1 g of amoxicillin per 125 mg of clavulanic acid, or, in the case of those with a penicillin allergy, ciprofloxacin 200 mg and metronidazole 500 mg.
Half of the patients were then assigned to either inpatient or outpatient antibiotic treatment. The admitted patients continued to receive IV antibiotics and fluids for 36-48 hours until they tolerated oral feeding. The outpatients were discharged on oral amoxicillin and clavulanic acid (875 mg per 125 mg every 8 hours) or, in those with penicillin allergy, the combination of ciprofloxacin (500 mg every 12 hours) and metronidazole (500 mg every 8 hours).
The study’s main endpoint was treatment failure rate (persistence, increase, or recurrence of abdominal pain and/or fever; inflammatory bowel obstruction; or the need for radiological abscess drainage or immediate surgery due to complicated diverticulitis). Patients were followed every day for 5 days, and then interviewed on day 14. Before the final follow-up at 60 days, patients underwent a colonoscopy to rule out malignancy.
Seven patients (5%) were readmitted due to treatment failure: 4 (6%) in the inpatient group and three (4.5%) in the outpatient group. The difference was not statistically significant. No one needed emergency surgery as part of readmission, and there were no deaths.
A quality of life assessment found that hospitalized patients reported a significantly higher level of physical health during the first 2 weeks of treatment, but that difference disappeared after 14 days.
Total treatment costs were significantly less in the outpatient group, with an average savings of $1,840 per patient ($895 for outpatient care compared with $2,735 for inpatient care). Almost all of that cost difference was due to hospital bed cost, with an average stay of 4 days.
"The outpatient protocol of this study is applicable to a selected group of patients with uncomplicated diverticulitis," Dr. Biondi reported in the January issue of Annals of Surgery (doi: 10.1097/SLA.0b013e3182965a11).
Widespread adoption of outpatient treatment could have a profound effect on the cost of treating diverticulitis, an ever more common gastrointestinal problem, said Dr. Biondi of the University of Barcelona and his coauthors. "According to data from the National Hospital Discharge Survey, diverticular disease is responsible for 314,000 hospital admissions per year in the United States, and the estimated annual cost in 1998 was about $2.6 billion."
The Spanish Ministry of Health funded the trial. The authors had no financial conflicts.
Discharging patients with uncomplicated diverticulitis from the emergency department on antibiotics was just as effective as hospitalization – and cost about a third as much, based on a study conducted in Spain.
Dr. Sebastiano Biondi and his colleagues conducted their randomized, parallel-group study at five hospitals. The cohort consisted of 132 adults, mean age 56 years, who presented to the emergency department with localized abdominal pain. All had an abdominal CT scan and received intravenous antibiotics, either an initial IV infusion of 1 g of amoxicillin per 125 mg of clavulanic acid, or, in the case of those with a penicillin allergy, ciprofloxacin 200 mg and metronidazole 500 mg.
Half of the patients were then assigned to either inpatient or outpatient antibiotic treatment. The admitted patients continued to receive IV antibiotics and fluids for 36-48 hours until they tolerated oral feeding. The outpatients were discharged on oral amoxicillin and clavulanic acid (875 mg per 125 mg every 8 hours) or, in those with penicillin allergy, the combination of ciprofloxacin (500 mg every 12 hours) and metronidazole (500 mg every 8 hours).
The study’s main endpoint was treatment failure rate (persistence, increase, or recurrence of abdominal pain and/or fever; inflammatory bowel obstruction; or the need for radiological abscess drainage or immediate surgery due to complicated diverticulitis). Patients were followed every day for 5 days, and then interviewed on day 14. Before the final follow-up at 60 days, patients underwent a colonoscopy to rule out malignancy.
Seven patients (5%) were readmitted due to treatment failure: 4 (6%) in the inpatient group and three (4.5%) in the outpatient group. The difference was not statistically significant. No one needed emergency surgery as part of readmission, and there were no deaths.
A quality of life assessment found that hospitalized patients reported a significantly higher level of physical health during the first 2 weeks of treatment, but that difference disappeared after 14 days.
Total treatment costs were significantly less in the outpatient group, with an average savings of $1,840 per patient ($895 for outpatient care compared with $2,735 for inpatient care). Almost all of that cost difference was due to hospital bed cost, with an average stay of 4 days.
"The outpatient protocol of this study is applicable to a selected group of patients with uncomplicated diverticulitis," Dr. Biondi reported in the January issue of Annals of Surgery (doi: 10.1097/SLA.0b013e3182965a11).
Widespread adoption of outpatient treatment could have a profound effect on the cost of treating diverticulitis, an ever more common gastrointestinal problem, said Dr. Biondi of the University of Barcelona and his coauthors. "According to data from the National Hospital Discharge Survey, diverticular disease is responsible for 314,000 hospital admissions per year in the United States, and the estimated annual cost in 1998 was about $2.6 billion."
The Spanish Ministry of Health funded the trial. The authors had no financial conflicts.
FROM ANNALS OF SURGERY
Major finding: Compared with inpatients, those with uncomplicated diverticulitis treated as outpatients had similarly low rates of treatment failure (4.5% vs. 6%), with a cost savings of $1,840 for each outpatient treatment.
Data source: The randomized, parallel-group study comprised 132 patients.
Disclosures: The Spanish Ministry of Health funded the study. The authors had no financial conflicts.
FDA advisers back vedolizumab approval to treat moderate to severe inflammatory bowel disease
SILVER SPRING, MD. – The benefits of vedolizumab, an integrin antagonist, outweigh the potential for progressive multifocal encephalopathy and other possible risks as a treatment for moderate to severely active Crohn’s disease and ulcerative colitis, and should be approved, according to two Food and Drug Administration advisory panels.
At a meeting of the FDA’s Gastrointestinal Drugs and Drug Safety and Risk Management advisory committees, the panels unanimously supported approval of vedolizumab for both indications, although they were less confident about the efficacy data of vedolizumab for induction in Crohn’s disease. They also strongly recommended a postmarketing program to monitor safety issues after approval, including progressive multifocal encephalopathy (PML), other infections, and other types of adverse events such as autoimmune hepatitis.
Vedolizumab is a monoclonal antibody that binds exclusively to the alpha 4 beta 7 integrin, "a key mediator of gastrointestinal inflammation," according to the manufacturer, Takeda Pharmaceuticals USA. It is administered intravenously at 0, 2, and 6 weeks, followed by once every 8 weeks for maintenance therapy. No cases of PML have been reported in more than 3,100 patients treated with vedolizumab worldwide, including 906 treated for 3 or more years, according to Takeda. But because it is thought to disrupt integrin function, like natalizumab (Tysabri), which is approved for multiple sclerosis and Crohn’s disease and is associated with an increased risk of PML, the potential for PML with vedolizumab therapy was the main safety issue raised by the FDA. Natalizumab is approved with a Risk Evaluation and Mitigation Strategy (REMS) that addresses the risk of PML, the usually fatal demyelinating CNS infection.
The phase III studies evaluating vedolizumab in patients with Crohn’s disease and ulcerative colitis were similarly designed, randomizing patients to vedolizumab or placebo in 6-week induction studies, and randomizing patients who responded to vedolizumab induction therapy to vedolizumab or placebo in 52-week maintenance studies.
In a Crohn’s disease induction and maintenance study of 368 patients, 14.5% of those on vedolizumab had achieved a clinical remission at week 6, vs. almost 7% of those on placebo, a statistically significant difference. At week 52, 39% of those who were treated every 8 weeks as maintenance therapy had a clinical remission, vs. almost 22% of those on placebo, also a significant difference. But in a second induction study of 315 patients, mostly treatment-refractory patients, there was not a significant difference in 6-week clinical remission rates between those on vedolizumab (15%) and those on placebo (12%).
The panelists voted 12 to 9 that these data supported efficacy for Crohn’s disease induction, and voted 20 to 0, with one abstention, that the data supported approval of the maintenance indication, agreeing the maintenance data were robust. Most (14) agreed that the benefits outweighed the risks to support approval of patients with Crohn’s who have failed treatment with steroids or immunosuppressants or tumor necrosis factor (TNF)–alpha blockers, the indication proposed by the manufacturer. Six panelists supported approval, but for the narrower use, in patients who had failed treatment with immunosuppressants or TNF-alpha antagonists, an indication that would not include patients who had failed steroids only. (The remaining panelist abstained.)
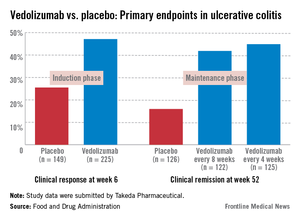
In the induction and maintenance trial of 374 patients with ulcerative colitis, the clinical response rate at 6 weeks was 47% among those on vedolizumab, vs. 25.5% among those on placebo, a statistically significant difference. In this study, almost 42% of those on vedolizumab every 8 weeks as maintenance therapy had achieved clinical remission at week 52, vs. 16% of those on placebo, also a statistically significant difference.
All panel members supported approval for the ulcerative colitis indication, based on the benefits and risks, but differed on the wording of the indication, with 13 panelists supporting approval for patients with ulcerative colitis who have failed steroids or immunosuppressants or TNF-alpha antagonists. The remaining eight panelists supported approval for patients who have failed immunosuppressants or TNF-alpha antagonists, which would not include an indication for patients who had failed treatment with steroids only.
The overall infection rate was higher among the patients treated with vedolizumab than among those on placebo in these studies (primarily upper respiratory tract infections and pharyngitis), which did not appear to be related to the number of infusions or concomitant immunosuppressive treatment. The rates of serious infections (3%-4%) were similar among those on vedolizumab and placebo. There were four patients with serious hepatic adverse events – acute hepatitis – that resolved. The 12 deaths among those on vedolizumab were not related to the drug, and there was no apparent increase in malignancies among treated patients, according to the FDA reviewers.
In a unanimous 21-0 vote, the panel agreed that Takeda had "adequately characterized the potential risk of PML" to support approval, but panelists recommended postmarketing follow-up for PML and other serious adverse events in treated patients.
The FDA is expected to make a decision on approval for the ulcerative colitis indication by Feb. 18, 2014, and for the Crohn’s disease indication by June 18, 2014.
The FDA usually follows the recommendations of its advisory panels. Members of FDA panels have usually been cleared of conflicts related to the product under review; occasionally, a panelist is given a waiver, but not at this meeting.
If approved, Takeda plans to market vedolizumab as Entyvio.
SILVER SPRING, MD. – The benefits of vedolizumab, an integrin antagonist, outweigh the potential for progressive multifocal encephalopathy and other possible risks as a treatment for moderate to severely active Crohn’s disease and ulcerative colitis, and should be approved, according to two Food and Drug Administration advisory panels.
At a meeting of the FDA’s Gastrointestinal Drugs and Drug Safety and Risk Management advisory committees, the panels unanimously supported approval of vedolizumab for both indications, although they were less confident about the efficacy data of vedolizumab for induction in Crohn’s disease. They also strongly recommended a postmarketing program to monitor safety issues after approval, including progressive multifocal encephalopathy (PML), other infections, and other types of adverse events such as autoimmune hepatitis.

Vedolizumab is a monoclonal antibody that binds exclusively to the alpha 4 beta 7 integrin, "a key mediator of gastrointestinal inflammation," according to the manufacturer, Takeda Pharmaceuticals USA. It is administered intravenously at 0, 2, and 6 weeks, followed by once every 8 weeks for maintenance therapy. No cases of PML have been reported in more than 3,100 patients treated with vedolizumab worldwide, including 906 treated for 3 or more years, according to Takeda. But because it is thought to disrupt integrin function, like natalizumab (Tysabri), which is approved for multiple sclerosis and Crohn’s disease and is associated with an increased risk of PML, the potential for PML with vedolizumab therapy was the main safety issue raised by the FDA. Natalizumab is approved with a Risk Evaluation and Mitigation Strategy (REMS) that addresses the risk of PML, the usually fatal demyelinating CNS infection.
The phase III studies evaluating vedolizumab in patients with Crohn’s disease and ulcerative colitis were similarly designed, randomizing patients to vedolizumab or placebo in 6-week induction studies, and randomizing patients who responded to vedolizumab induction therapy to vedolizumab or placebo in 52-week maintenance studies.
In a Crohn’s disease induction and maintenance study of 368 patients, 14.5% of those on vedolizumab had achieved a clinical remission at week 6, vs. almost 7% of those on placebo, a statistically significant difference. At week 52, 39% of those who were treated every 8 weeks as maintenance therapy had a clinical remission, vs. almost 22% of those on placebo, also a significant difference. But in a second induction study of 315 patients, mostly treatment-refractory patients, there was not a significant difference in 6-week clinical remission rates between those on vedolizumab (15%) and those on placebo (12%).
The panelists voted 12 to 9 that these data supported efficacy for Crohn’s disease induction, and voted 20 to 0, with one abstention, that the data supported approval of the maintenance indication, agreeing the maintenance data were robust. Most (14) agreed that the benefits outweighed the risks to support approval of patients with Crohn’s who have failed treatment with steroids or immunosuppressants or tumor necrosis factor (TNF)–alpha blockers, the indication proposed by the manufacturer. Six panelists supported approval, but for the narrower use, in patients who had failed treatment with immunosuppressants or TNF-alpha antagonists, an indication that would not include patients who had failed steroids only. (The remaining panelist abstained.)
In the induction and maintenance trial of 374 patients with ulcerative colitis, the clinical response rate at 6 weeks was 47% among those on vedolizumab, vs. 25.5% among those on placebo, a statistically significant difference. In this study, almost 42% of those on vedolizumab every 8 weeks as maintenance therapy had achieved clinical remission at week 52, vs. 16% of those on placebo, also a statistically significant difference.
All panel members supported approval for the ulcerative colitis indication, based on the benefits and risks, but differed on the wording of the indication, with 13 panelists supporting approval for patients with ulcerative colitis who have failed steroids or immunosuppressants or TNF-alpha antagonists. The remaining eight panelists supported approval for patients who have failed immunosuppressants or TNF-alpha antagonists, which would not include an indication for patients who had failed treatment with steroids only.
The overall infection rate was higher among the patients treated with vedolizumab than among those on placebo in these studies (primarily upper respiratory tract infections and pharyngitis), which did not appear to be related to the number of infusions or concomitant immunosuppressive treatment. The rates of serious infections (3%-4%) were similar among those on vedolizumab and placebo. There were four patients with serious hepatic adverse events – acute hepatitis – that resolved. The 12 deaths among those on vedolizumab were not related to the drug, and there was no apparent increase in malignancies among treated patients, according to the FDA reviewers.
In a unanimous 21-0 vote, the panel agreed that Takeda had "adequately characterized the potential risk of PML" to support approval, but panelists recommended postmarketing follow-up for PML and other serious adverse events in treated patients.
The FDA is expected to make a decision on approval for the ulcerative colitis indication by Feb. 18, 2014, and for the Crohn’s disease indication by June 18, 2014.
The FDA usually follows the recommendations of its advisory panels. Members of FDA panels have usually been cleared of conflicts related to the product under review; occasionally, a panelist is given a waiver, but not at this meeting.
If approved, Takeda plans to market vedolizumab as Entyvio.
SILVER SPRING, MD. – The benefits of vedolizumab, an integrin antagonist, outweigh the potential for progressive multifocal encephalopathy and other possible risks as a treatment for moderate to severely active Crohn’s disease and ulcerative colitis, and should be approved, according to two Food and Drug Administration advisory panels.
At a meeting of the FDA’s Gastrointestinal Drugs and Drug Safety and Risk Management advisory committees, the panels unanimously supported approval of vedolizumab for both indications, although they were less confident about the efficacy data of vedolizumab for induction in Crohn’s disease. They also strongly recommended a postmarketing program to monitor safety issues after approval, including progressive multifocal encephalopathy (PML), other infections, and other types of adverse events such as autoimmune hepatitis.

Vedolizumab is a monoclonal antibody that binds exclusively to the alpha 4 beta 7 integrin, "a key mediator of gastrointestinal inflammation," according to the manufacturer, Takeda Pharmaceuticals USA. It is administered intravenously at 0, 2, and 6 weeks, followed by once every 8 weeks for maintenance therapy. No cases of PML have been reported in more than 3,100 patients treated with vedolizumab worldwide, including 906 treated for 3 or more years, according to Takeda. But because it is thought to disrupt integrin function, like natalizumab (Tysabri), which is approved for multiple sclerosis and Crohn’s disease and is associated with an increased risk of PML, the potential for PML with vedolizumab therapy was the main safety issue raised by the FDA. Natalizumab is approved with a Risk Evaluation and Mitigation Strategy (REMS) that addresses the risk of PML, the usually fatal demyelinating CNS infection.
The phase III studies evaluating vedolizumab in patients with Crohn’s disease and ulcerative colitis were similarly designed, randomizing patients to vedolizumab or placebo in 6-week induction studies, and randomizing patients who responded to vedolizumab induction therapy to vedolizumab or placebo in 52-week maintenance studies.
In a Crohn’s disease induction and maintenance study of 368 patients, 14.5% of those on vedolizumab had achieved a clinical remission at week 6, vs. almost 7% of those on placebo, a statistically significant difference. At week 52, 39% of those who were treated every 8 weeks as maintenance therapy had a clinical remission, vs. almost 22% of those on placebo, also a significant difference. But in a second induction study of 315 patients, mostly treatment-refractory patients, there was not a significant difference in 6-week clinical remission rates between those on vedolizumab (15%) and those on placebo (12%).
The panelists voted 12 to 9 that these data supported efficacy for Crohn’s disease induction, and voted 20 to 0, with one abstention, that the data supported approval of the maintenance indication, agreeing the maintenance data were robust. Most (14) agreed that the benefits outweighed the risks to support approval of patients with Crohn’s who have failed treatment with steroids or immunosuppressants or tumor necrosis factor (TNF)–alpha blockers, the indication proposed by the manufacturer. Six panelists supported approval, but for the narrower use, in patients who had failed treatment with immunosuppressants or TNF-alpha antagonists, an indication that would not include patients who had failed steroids only. (The remaining panelist abstained.)
In the induction and maintenance trial of 374 patients with ulcerative colitis, the clinical response rate at 6 weeks was 47% among those on vedolizumab, vs. 25.5% among those on placebo, a statistically significant difference. In this study, almost 42% of those on vedolizumab every 8 weeks as maintenance therapy had achieved clinical remission at week 52, vs. 16% of those on placebo, also a statistically significant difference.
All panel members supported approval for the ulcerative colitis indication, based on the benefits and risks, but differed on the wording of the indication, with 13 panelists supporting approval for patients with ulcerative colitis who have failed steroids or immunosuppressants or TNF-alpha antagonists. The remaining eight panelists supported approval for patients who have failed immunosuppressants or TNF-alpha antagonists, which would not include an indication for patients who had failed treatment with steroids only.
The overall infection rate was higher among the patients treated with vedolizumab than among those on placebo in these studies (primarily upper respiratory tract infections and pharyngitis), which did not appear to be related to the number of infusions or concomitant immunosuppressive treatment. The rates of serious infections (3%-4%) were similar among those on vedolizumab and placebo. There were four patients with serious hepatic adverse events – acute hepatitis – that resolved. The 12 deaths among those on vedolizumab were not related to the drug, and there was no apparent increase in malignancies among treated patients, according to the FDA reviewers.
In a unanimous 21-0 vote, the panel agreed that Takeda had "adequately characterized the potential risk of PML" to support approval, but panelists recommended postmarketing follow-up for PML and other serious adverse events in treated patients.
The FDA is expected to make a decision on approval for the ulcerative colitis indication by Feb. 18, 2014, and for the Crohn’s disease indication by June 18, 2014.
The FDA usually follows the recommendations of its advisory panels. Members of FDA panels have usually been cleared of conflicts related to the product under review; occasionally, a panelist is given a waiver, but not at this meeting.
If approved, Takeda plans to market vedolizumab as Entyvio.
AT AN FDA ADVISORY COMMITTEE MEETING
Thrombosis precautions in IBD not met in two-thirds of high-risk cases
HOLLYWOOD, FLA. – The relative risk of thromboembolic events is greater in inpatient inflammatory bowel disease patients than in the general population, but prophylactic treatment is still not standard, according to a speaker at a conference on inflammatory bowel diseases.
"It’s a relatively rare problem, only about 1 to 1.5 percent," said Dr. Athos Bousvaros, of Boston Children’s Hospital. "So, why worry about it so much? Because it really is a major source of morbidity in the IBD population. It usually happens in the sick patients, the ones at risk for strokes; and it usually happens at the worst time, when you’re thinking about colectomy."
However, Dr. Bousvaros said only about a third of IBD patients at risk for a thromboembolic event are given prophylactic treatment in the inpatient setting, especially in severe colitis, and it is generally recommended.
Although pharmacologic prophylaxis is included in the American College of Gastroenterology guidelines, Dr. Bousvaros cited a recent study that found only 35% of gastroenterologists in the United States actually do so (J. Clin. Gastroenterol. 2013;47:e1-e6).
"In the inpatient setting, especially in severe colitis, [prophylaxis] is generally recommended," Dr. Bousvaros said. "It is included in the AGA [American Gastroenterological Association] physician performance measure set." In the outpatient setting, data do not support it, he said.
Relative risk high
While the absolute risk is low, the relative risk of a venous thromboembolic event is six times greater in IBD, particularly in patients aged 20 years or less, said Dr. Bousvaros, citing a cohort study that used Danish administrative data (Gut 2011;60:937-43). "It’s mainly patients with flares, and mainly those with colitis, either Crohn’s or severe ulcerative colitis," Dr. Bousvaros said.
In a prospective study of about 2,800 IBD patients (mean age, 42 years) recruited over 2.5 years, matched with non-IBD controls, and followed for several years, about 4% developed de novo venous thromboembolism (Gastroenterol. 2010;139:779-787.e1). Dr. Bousvaros emphasized that IBD was an independent risk factor for VTE recurrence in the study. "They were typically treated with long-standing prophylaxis. And if any anticoagulation was involved, the risk of recurrence was high," he said at the meeting, which was sponsored by the Crohn’s & Colitis Foundation of America.
High-risk criteria
Overall, the relative risk for VTE was found by a just-published meta-analysis of more than 200,000 IBD patients to be 2.4 for deep vein thrombosis, 2.5 for pulmonary embolism, 1.3 for ischemic heart disease, and 3.4 for mesenteric ischemia (J. Crohns Colitis 2013 Oct 29 [doi: 10.1016/j.crohns.2013.09.021]). Dr. Bousvaros said the investigators did not find an increased risk for arterial thromboembolic events in IBD, but that VTEs "were highly significant in this population."
Patients with IBD should be considered high risk for VTE if they are being treated in hospital for severe colitis and have a personal or family history of thrombosis, have known thrombophilia, have been taking oral contraceptives, have a history of smoking, are obese, or have had a PICC line.
"Any of those makes you a particularly high-risk patient," Dr. Bousvaros said.
Dr. Bousvaros disclosed he has consulting relationships with Cubist, Dyax, and Millennium.
HOLLYWOOD, FLA. – The relative risk of thromboembolic events is greater in inpatient inflammatory bowel disease patients than in the general population, but prophylactic treatment is still not standard, according to a speaker at a conference on inflammatory bowel diseases.
"It’s a relatively rare problem, only about 1 to 1.5 percent," said Dr. Athos Bousvaros, of Boston Children’s Hospital. "So, why worry about it so much? Because it really is a major source of morbidity in the IBD population. It usually happens in the sick patients, the ones at risk for strokes; and it usually happens at the worst time, when you’re thinking about colectomy."
However, Dr. Bousvaros said only about a third of IBD patients at risk for a thromboembolic event are given prophylactic treatment in the inpatient setting, especially in severe colitis, and it is generally recommended.
Although pharmacologic prophylaxis is included in the American College of Gastroenterology guidelines, Dr. Bousvaros cited a recent study that found only 35% of gastroenterologists in the United States actually do so (J. Clin. Gastroenterol. 2013;47:e1-e6).
"In the inpatient setting, especially in severe colitis, [prophylaxis] is generally recommended," Dr. Bousvaros said. "It is included in the AGA [American Gastroenterological Association] physician performance measure set." In the outpatient setting, data do not support it, he said.
Relative risk high
While the absolute risk is low, the relative risk of a venous thromboembolic event is six times greater in IBD, particularly in patients aged 20 years or less, said Dr. Bousvaros, citing a cohort study that used Danish administrative data (Gut 2011;60:937-43). "It’s mainly patients with flares, and mainly those with colitis, either Crohn’s or severe ulcerative colitis," Dr. Bousvaros said.
In a prospective study of about 2,800 IBD patients (mean age, 42 years) recruited over 2.5 years, matched with non-IBD controls, and followed for several years, about 4% developed de novo venous thromboembolism (Gastroenterol. 2010;139:779-787.e1). Dr. Bousvaros emphasized that IBD was an independent risk factor for VTE recurrence in the study. "They were typically treated with long-standing prophylaxis. And if any anticoagulation was involved, the risk of recurrence was high," he said at the meeting, which was sponsored by the Crohn’s & Colitis Foundation of America.
High-risk criteria
Overall, the relative risk for VTE was found by a just-published meta-analysis of more than 200,000 IBD patients to be 2.4 for deep vein thrombosis, 2.5 for pulmonary embolism, 1.3 for ischemic heart disease, and 3.4 for mesenteric ischemia (J. Crohns Colitis 2013 Oct 29 [doi: 10.1016/j.crohns.2013.09.021]). Dr. Bousvaros said the investigators did not find an increased risk for arterial thromboembolic events in IBD, but that VTEs "were highly significant in this population."
Patients with IBD should be considered high risk for VTE if they are being treated in hospital for severe colitis and have a personal or family history of thrombosis, have known thrombophilia, have been taking oral contraceptives, have a history of smoking, are obese, or have had a PICC line.
"Any of those makes you a particularly high-risk patient," Dr. Bousvaros said.
Dr. Bousvaros disclosed he has consulting relationships with Cubist, Dyax, and Millennium.
HOLLYWOOD, FLA. – The relative risk of thromboembolic events is greater in inpatient inflammatory bowel disease patients than in the general population, but prophylactic treatment is still not standard, according to a speaker at a conference on inflammatory bowel diseases.
"It’s a relatively rare problem, only about 1 to 1.5 percent," said Dr. Athos Bousvaros, of Boston Children’s Hospital. "So, why worry about it so much? Because it really is a major source of morbidity in the IBD population. It usually happens in the sick patients, the ones at risk for strokes; and it usually happens at the worst time, when you’re thinking about colectomy."
However, Dr. Bousvaros said only about a third of IBD patients at risk for a thromboembolic event are given prophylactic treatment in the inpatient setting, especially in severe colitis, and it is generally recommended.
Although pharmacologic prophylaxis is included in the American College of Gastroenterology guidelines, Dr. Bousvaros cited a recent study that found only 35% of gastroenterologists in the United States actually do so (J. Clin. Gastroenterol. 2013;47:e1-e6).
"In the inpatient setting, especially in severe colitis, [prophylaxis] is generally recommended," Dr. Bousvaros said. "It is included in the AGA [American Gastroenterological Association] physician performance measure set." In the outpatient setting, data do not support it, he said.
Relative risk high
While the absolute risk is low, the relative risk of a venous thromboembolic event is six times greater in IBD, particularly in patients aged 20 years or less, said Dr. Bousvaros, citing a cohort study that used Danish administrative data (Gut 2011;60:937-43). "It’s mainly patients with flares, and mainly those with colitis, either Crohn’s or severe ulcerative colitis," Dr. Bousvaros said.
In a prospective study of about 2,800 IBD patients (mean age, 42 years) recruited over 2.5 years, matched with non-IBD controls, and followed for several years, about 4% developed de novo venous thromboembolism (Gastroenterol. 2010;139:779-787.e1). Dr. Bousvaros emphasized that IBD was an independent risk factor for VTE recurrence in the study. "They were typically treated with long-standing prophylaxis. And if any anticoagulation was involved, the risk of recurrence was high," he said at the meeting, which was sponsored by the Crohn’s & Colitis Foundation of America.
High-risk criteria
Overall, the relative risk for VTE was found by a just-published meta-analysis of more than 200,000 IBD patients to be 2.4 for deep vein thrombosis, 2.5 for pulmonary embolism, 1.3 for ischemic heart disease, and 3.4 for mesenteric ischemia (J. Crohns Colitis 2013 Oct 29 [doi: 10.1016/j.crohns.2013.09.021]). Dr. Bousvaros said the investigators did not find an increased risk for arterial thromboembolic events in IBD, but that VTEs "were highly significant in this population."
Patients with IBD should be considered high risk for VTE if they are being treated in hospital for severe colitis and have a personal or family history of thrombosis, have known thrombophilia, have been taking oral contraceptives, have a history of smoking, are obese, or have had a PICC line.
"Any of those makes you a particularly high-risk patient," Dr. Bousvaros said.
Dr. Bousvaros disclosed he has consulting relationships with Cubist, Dyax, and Millennium.
EXPERT ANALYSIS FROM 2013 ADVANCES IN IBD
Thiopurine use ups risk of skin cancer for ulcerative colitis patients
SAN DIEGO – Current thiopurine use is an independent risk factor for nonmelanoma skin cancer in patients with ulcerative colitis, according to a study reported at the annual meeting of the American College of Gastroenterology.
In the retrospective cohort study of more than 14,000 U.S. veterans with ulcerative colitis, current users of thiopurines were more than twice as likely as never users to receive a diagnosis of squamous or basal cell skin cancer. The excess risk, however, disappeared after stopping thiopurines.
"It is crucial to educate physicians and patients about the risk of nonmelanoma skin cancer and possible preventive measures," asserted Dr. Ali Abbas, an internal medicine resident at * University of Florida, Gainesville.
Session comoderator Dr. Stephen B. Hanauer of the University of Chicago noted, "The current quality measures in [inflammatory bowel disease] include a number of measures related to biologic therapy – immunization/vaccination, [tuberculosis] testing, etc. ... Do you believe that yearly skin examination should be added? Is there enough evidence that this should be added to the quality indicators?"
"I think now we have enough evidence to recommend regular skin examination for those on long-term thiopurines," Dr. Abbas replied, while adding that the reversibility of risk after stopping remains controversial.
Dr. Hanauer further noted that number of health care visits also predicted nonmelanoma skin cancer risk. "I would suspect that, while patients are on thiopurines, they are getting more intensive visits, so can you dissociate those factors?"
Multivariate analyses took into account the number of visits, according to Dr. Abbas. "Even after exclusion of this confounding effect, we had a two times increase of the risk," he said. "If you compare our hazard ratio with previously reported hazard ratios, it’s lower – most of them reported were 4 to 5. So I am assuming that we kind of excluded the effect of the detection bias and we present a more independent effect of thiopurines on risk of nonmelanoma skin cancer."
"Do you know what the lag time would be then from exposure to the development of a nonmelanoma? So is it biologically plausible to understand that this would be reversible in patients who stop therapy?" asked Dr. David T. Rubin, codirector of the inflammatory bowel disease center at the University of Chicago, the other session comoderator.
The investigators did not calculate lag time, Dr. Abbas replied. "The biological origin is just the interaction between the UV [ultraviolet] radiation on the skin and the damage that the thiopurine causes to the DNA will prevent the repair. So from a biological point of view, I think it’s understandable to conclude that, once the insult to the DNA or the repair mechanism is gone, the UV radiation effect will also be low."
"Well, I think there is another biologic plausibility," Dr. Hanauer suggested, "which is the potential for viral infections such as [human papillomavirus] contributing to skin cancers, and the known effect of thiopurines on viruses. So certainly, UV is a very strong association, but we are also familiar with individuals who have skin warts."
"You do need to understand lag time between exposure and subsequent neoplasia before you conclude that when you are off therapy, you don’t need screening anymore," Dr. Rubin said.
Upcoming analyses will stratify patients according to the location of skin cancer, for example, looking at anogenital skin cancers separately, Dr. Abbas replied.
In the study, the investigators reviewed pharmacy benefits records for 14,527 veterans with an ulcerative colitis diagnosis who were seen in the Veterans Affairs health care system between 2001 and 2011.
They were 59 years old on average at baseline; 94% were male and 77% were white. Their median follow-up was 8.1 years, according to Dr. Abbas.
Overall, 23% of the patients used thiopurines, for a median of 1.6 years during follow-up, and the median duration of follow-up after stopping these medications was 3.6 years. The median duration of follow-up among thiopurine-unexposed patients was 6.7 years.
Main results showed that the incidence of nonmelanoma skin cancer was 3.7 per 1,000 person-years among unexposed patients, 8.4 per 1,000 person-years during thiopurine use, and 3.0 per 1,000 person-years after stopping thiopurines.
In a multivariate model, patients had a significantly higher risk of nonmelanoma skin cancer while taking thiopurines when compared with never-users (hazard ratio, 2.1). There also was a nonsignificant trend toward reduced risk after stopping, as compared with never users (0.7).
Other factors associated with higher risk included older age, male sex, white race/ethnicity, living in zones with high UV exposure, and more frequent use of the VA health care system.
In stratified analyses, the incidence of nonmelanoma skin cancer during thiopurine use increased with patient age from younger than 40 years to 40-65 years to older than 65 years (0.6, 9.1, and 12.2 per 1,000 person-years); was greater among patients living in high-UV zones versus low- or medium-UV zones (10.3 vs. 6.0 per 1,000 person-years); and increased with number of VA visits annually from fewer than six, to 6-12, and then more than 12 (1.2, 9.4, and 12.3 per 1,000 person-years).
Finally, the rate rose with the cumulative duration of exposure to thiopurines. It increased steadily during the first 2 years of use, stabilized through the fourth year of use, and rose sharply in the fifth year of use to 13.6 per 1,000 person-years.
Dr. Abbas disclosed no relevant conflicts of interest.
*Correction 12/10/13: A previous version of this article incorrectly reported Dr. Ali Abbas' university affiliation. This version has been updated.
SAN DIEGO – Current thiopurine use is an independent risk factor for nonmelanoma skin cancer in patients with ulcerative colitis, according to a study reported at the annual meeting of the American College of Gastroenterology.
In the retrospective cohort study of more than 14,000 U.S. veterans with ulcerative colitis, current users of thiopurines were more than twice as likely as never users to receive a diagnosis of squamous or basal cell skin cancer. The excess risk, however, disappeared after stopping thiopurines.
"It is crucial to educate physicians and patients about the risk of nonmelanoma skin cancer and possible preventive measures," asserted Dr. Ali Abbas, an internal medicine resident at * University of Florida, Gainesville.
Session comoderator Dr. Stephen B. Hanauer of the University of Chicago noted, "The current quality measures in [inflammatory bowel disease] include a number of measures related to biologic therapy – immunization/vaccination, [tuberculosis] testing, etc. ... Do you believe that yearly skin examination should be added? Is there enough evidence that this should be added to the quality indicators?"
"I think now we have enough evidence to recommend regular skin examination for those on long-term thiopurines," Dr. Abbas replied, while adding that the reversibility of risk after stopping remains controversial.
Dr. Hanauer further noted that number of health care visits also predicted nonmelanoma skin cancer risk. "I would suspect that, while patients are on thiopurines, they are getting more intensive visits, so can you dissociate those factors?"
Multivariate analyses took into account the number of visits, according to Dr. Abbas. "Even after exclusion of this confounding effect, we had a two times increase of the risk," he said. "If you compare our hazard ratio with previously reported hazard ratios, it’s lower – most of them reported were 4 to 5. So I am assuming that we kind of excluded the effect of the detection bias and we present a more independent effect of thiopurines on risk of nonmelanoma skin cancer."
"Do you know what the lag time would be then from exposure to the development of a nonmelanoma? So is it biologically plausible to understand that this would be reversible in patients who stop therapy?" asked Dr. David T. Rubin, codirector of the inflammatory bowel disease center at the University of Chicago, the other session comoderator.
The investigators did not calculate lag time, Dr. Abbas replied. "The biological origin is just the interaction between the UV [ultraviolet] radiation on the skin and the damage that the thiopurine causes to the DNA will prevent the repair. So from a biological point of view, I think it’s understandable to conclude that, once the insult to the DNA or the repair mechanism is gone, the UV radiation effect will also be low."
"Well, I think there is another biologic plausibility," Dr. Hanauer suggested, "which is the potential for viral infections such as [human papillomavirus] contributing to skin cancers, and the known effect of thiopurines on viruses. So certainly, UV is a very strong association, but we are also familiar with individuals who have skin warts."
"You do need to understand lag time between exposure and subsequent neoplasia before you conclude that when you are off therapy, you don’t need screening anymore," Dr. Rubin said.
Upcoming analyses will stratify patients according to the location of skin cancer, for example, looking at anogenital skin cancers separately, Dr. Abbas replied.
In the study, the investigators reviewed pharmacy benefits records for 14,527 veterans with an ulcerative colitis diagnosis who were seen in the Veterans Affairs health care system between 2001 and 2011.
They were 59 years old on average at baseline; 94% were male and 77% were white. Their median follow-up was 8.1 years, according to Dr. Abbas.
Overall, 23% of the patients used thiopurines, for a median of 1.6 years during follow-up, and the median duration of follow-up after stopping these medications was 3.6 years. The median duration of follow-up among thiopurine-unexposed patients was 6.7 years.
Main results showed that the incidence of nonmelanoma skin cancer was 3.7 per 1,000 person-years among unexposed patients, 8.4 per 1,000 person-years during thiopurine use, and 3.0 per 1,000 person-years after stopping thiopurines.
In a multivariate model, patients had a significantly higher risk of nonmelanoma skin cancer while taking thiopurines when compared with never-users (hazard ratio, 2.1). There also was a nonsignificant trend toward reduced risk after stopping, as compared with never users (0.7).
Other factors associated with higher risk included older age, male sex, white race/ethnicity, living in zones with high UV exposure, and more frequent use of the VA health care system.
In stratified analyses, the incidence of nonmelanoma skin cancer during thiopurine use increased with patient age from younger than 40 years to 40-65 years to older than 65 years (0.6, 9.1, and 12.2 per 1,000 person-years); was greater among patients living in high-UV zones versus low- or medium-UV zones (10.3 vs. 6.0 per 1,000 person-years); and increased with number of VA visits annually from fewer than six, to 6-12, and then more than 12 (1.2, 9.4, and 12.3 per 1,000 person-years).
Finally, the rate rose with the cumulative duration of exposure to thiopurines. It increased steadily during the first 2 years of use, stabilized through the fourth year of use, and rose sharply in the fifth year of use to 13.6 per 1,000 person-years.
Dr. Abbas disclosed no relevant conflicts of interest.
*Correction 12/10/13: A previous version of this article incorrectly reported Dr. Ali Abbas' university affiliation. This version has been updated.
SAN DIEGO – Current thiopurine use is an independent risk factor for nonmelanoma skin cancer in patients with ulcerative colitis, according to a study reported at the annual meeting of the American College of Gastroenterology.
In the retrospective cohort study of more than 14,000 U.S. veterans with ulcerative colitis, current users of thiopurines were more than twice as likely as never users to receive a diagnosis of squamous or basal cell skin cancer. The excess risk, however, disappeared after stopping thiopurines.
"It is crucial to educate physicians and patients about the risk of nonmelanoma skin cancer and possible preventive measures," asserted Dr. Ali Abbas, an internal medicine resident at * University of Florida, Gainesville.
Session comoderator Dr. Stephen B. Hanauer of the University of Chicago noted, "The current quality measures in [inflammatory bowel disease] include a number of measures related to biologic therapy – immunization/vaccination, [tuberculosis] testing, etc. ... Do you believe that yearly skin examination should be added? Is there enough evidence that this should be added to the quality indicators?"
"I think now we have enough evidence to recommend regular skin examination for those on long-term thiopurines," Dr. Abbas replied, while adding that the reversibility of risk after stopping remains controversial.
Dr. Hanauer further noted that number of health care visits also predicted nonmelanoma skin cancer risk. "I would suspect that, while patients are on thiopurines, they are getting more intensive visits, so can you dissociate those factors?"
Multivariate analyses took into account the number of visits, according to Dr. Abbas. "Even after exclusion of this confounding effect, we had a two times increase of the risk," he said. "If you compare our hazard ratio with previously reported hazard ratios, it’s lower – most of them reported were 4 to 5. So I am assuming that we kind of excluded the effect of the detection bias and we present a more independent effect of thiopurines on risk of nonmelanoma skin cancer."
"Do you know what the lag time would be then from exposure to the development of a nonmelanoma? So is it biologically plausible to understand that this would be reversible in patients who stop therapy?" asked Dr. David T. Rubin, codirector of the inflammatory bowel disease center at the University of Chicago, the other session comoderator.
The investigators did not calculate lag time, Dr. Abbas replied. "The biological origin is just the interaction between the UV [ultraviolet] radiation on the skin and the damage that the thiopurine causes to the DNA will prevent the repair. So from a biological point of view, I think it’s understandable to conclude that, once the insult to the DNA or the repair mechanism is gone, the UV radiation effect will also be low."
"Well, I think there is another biologic plausibility," Dr. Hanauer suggested, "which is the potential for viral infections such as [human papillomavirus] contributing to skin cancers, and the known effect of thiopurines on viruses. So certainly, UV is a very strong association, but we are also familiar with individuals who have skin warts."
"You do need to understand lag time between exposure and subsequent neoplasia before you conclude that when you are off therapy, you don’t need screening anymore," Dr. Rubin said.
Upcoming analyses will stratify patients according to the location of skin cancer, for example, looking at anogenital skin cancers separately, Dr. Abbas replied.
In the study, the investigators reviewed pharmacy benefits records for 14,527 veterans with an ulcerative colitis diagnosis who were seen in the Veterans Affairs health care system between 2001 and 2011.
They were 59 years old on average at baseline; 94% were male and 77% were white. Their median follow-up was 8.1 years, according to Dr. Abbas.
Overall, 23% of the patients used thiopurines, for a median of 1.6 years during follow-up, and the median duration of follow-up after stopping these medications was 3.6 years. The median duration of follow-up among thiopurine-unexposed patients was 6.7 years.
Main results showed that the incidence of nonmelanoma skin cancer was 3.7 per 1,000 person-years among unexposed patients, 8.4 per 1,000 person-years during thiopurine use, and 3.0 per 1,000 person-years after stopping thiopurines.
In a multivariate model, patients had a significantly higher risk of nonmelanoma skin cancer while taking thiopurines when compared with never-users (hazard ratio, 2.1). There also was a nonsignificant trend toward reduced risk after stopping, as compared with never users (0.7).
Other factors associated with higher risk included older age, male sex, white race/ethnicity, living in zones with high UV exposure, and more frequent use of the VA health care system.
In stratified analyses, the incidence of nonmelanoma skin cancer during thiopurine use increased with patient age from younger than 40 years to 40-65 years to older than 65 years (0.6, 9.1, and 12.2 per 1,000 person-years); was greater among patients living in high-UV zones versus low- or medium-UV zones (10.3 vs. 6.0 per 1,000 person-years); and increased with number of VA visits annually from fewer than six, to 6-12, and then more than 12 (1.2, 9.4, and 12.3 per 1,000 person-years).
Finally, the rate rose with the cumulative duration of exposure to thiopurines. It increased steadily during the first 2 years of use, stabilized through the fourth year of use, and rose sharply in the fifth year of use to 13.6 per 1,000 person-years.
Dr. Abbas disclosed no relevant conflicts of interest.
*Correction 12/10/13: A previous version of this article incorrectly reported Dr. Ali Abbas' university affiliation. This version has been updated.
AT THE ACG ANNUAL MEETING
Major Finding: Current users of thiopurines had more than twice the risk of nonmelanoma skin cancer as never users (hazard ratio, 2.1), but former users did not have an elevation of risk.
Data Source: A nationwide retrospective cohort study of 14,527 veterans with ulcerative colitis.
Disclosures: Dr. Abbas disclosed no relevant conflicts of interest.
New diverticulosis data challenge long-held beliefs
Not only is there no link between low-fiber diets and diverticulosis, but the incidence of diverticulitis is not nearly as common as was previously believed.
Those are the conclusions of two new studies in the December issue of Clinical Gastroenterology and Hepatology, both of which challenge long-held beliefs about the causes of these conditions.
In the first study, Dr. Anne F. Peery of the University of North Carolina at Chapel Hill, and her colleagues looked at 539 patients with colonic diverticula and 1,569 controls, all culled from the Vitamin D and Calcium Polyp Prevention Study, a double-blind, placebo-controlled trial of vitamin D and/or calcium for the prevention of colonic adenomas (doi:10.1016/j.cgh.2013.06.033).
Patients with a self-reported history of diverticulosis or diverticulitis were excluded, as were cases with a history of colon resection, inflammatory bowel disease, or familial history of colon cancer. Most cases (88%) had descending or sigmoid colon diverticula, and these patients were significantly older and more likely to be male than were the controls.
According to Dr. Peery and colleagues, there was no difference between cases and controls in terms of mean dietary fiber intake (14.8 g per day versus 15.3 g per day, P = .2) and reported supplemental fiber intake (5% versus 5%, P = .7).
Nor was there any significant link when investigators compared the highest quartile of fiber intake (mean, 25 g/day) to the lowest (mean, 8 g/day) (odds ratio = 0.96; 95% confidence interval, 0.71-1.30).
Finally, the investigators found no associations between dietary fiber intake by subtype (for instance, beans, grains, fruits, and vegetables) and diverticulosis.
"Forty years ago, Dr. Neil Painter popularized the hypothesis that inadequate dietary fiber intake and constipation were the cause of sigmoid diverticulosis," wrote Dr. Peery. However, "Although the fiber hypothesis is conceptually attractive and widely accepted, it has not been rigorously examined."
And while Dr. Peery’s data were based on a food frequency questionnaire – which could be subject to measurement bias – she added that "the mean total fiber intake in the highest quartile was 25 g, versus 8 g in the lowest.
"This wide range makes it unlikely that homogeneity of intake accounts for the null association of fiber with the presence of diverticula," she wrote.
A second study by Dr. Kamyar Shahedi of the University of California Los Angeles/Veteran’s Affairs Center for Outcomes Research and Education, also sought evidence for the commonly held belief that up to 25% of patients with diverticulosis will develop diverticulitis.
Dr. Shahedi and colleagues performed a retrospective survival analysis of 2,222 patients from the Veteran’s Affairs Greater Los Angeles Healthcare System with colonic diverticulosis and a median follow-up of 6.75 years (doi:10.1016/j.cgh.2013.06.020). Patients were excluded if they had any ICD-9 code for diverticulitis or documentation of diverticulitis in the medical record notes at any point before the index date of diverticulosis.
When the researchers looked only at imaging-confirmed or surgical specimen–confirmed cases, just 23 patients (1%) developed acute diverticulitis during the study period, Dr. Shahedi found. This jumped to 95 patients (4.3%) when clinical diagnoses were also used, for an incidence of 6 cases per 1,000 patient years.
Looking at predictors for progression, the authors found that only age was related to the development of diverticulitis, with every year of age at diverticulosis detection conferring a 2.4% lower hazard of developing diverticulitis.
According to the authors, the "widely cited figures" that up to a quarter of patients with diverticulosis will develop acute diverticulitis is based on data collected before the time of routine colon screening. "Therefore, the true denominator of individuals harboring diverticulosis was not accounted for in these calculations," they concluded.
And while their retrospective study does leave room for the possibility that cases were missed, "Future series or patient registries may better standardize the definition of diverticulitis in a prospective cohort," wrote the investigators.
In the meantime, prevalence data such as these "may help to reframe discussions with patients regarding their probability of developing clinically significant diverticulitis."
However, even as these two findings change the way providers counsel patients about the cause and impact of diverticula, a third study, also in December’s issue of Clinical Gastroenterology and Hepatology, adds another wrinkle: Patients who do develop diverticulitis are at increased risk for a diagnosis of irritable bowel syndrome later on.
Dr. Erica Cohen of the VA Greater Los Angeles Healthcare System, and colleagues looked at 1,105 chart-confirmed cases of diverticulitis, identified retrospectively from the same dataset used by Dr. Shahedi (doi:10.1016/j.cgh.2013.03.007).
All cases were matched with controls seen on the same day, the mean follow-up period was 6.3 years, and patients with pre-existing IBS or functional bowel diagnoses were excluded from the study.
The primary outcome was a new IBS diagnosis after the index diverticulitis attack (for cases) or enrollment date (for controls). Ultimately, Dr. Cohen found 24 cases of newly diagnosed IBS during the study period: 20 among diverticulitis cases, and 4 among controls. That translated to a hazard ratio of 4.7 among cases compared with controls, even after adjustment for age, sex, ethnicity, race, inpatient versus outpatient status, and comorbidity score (95% CI, 1.6 –14.0; P = .006).
Dr. Cohen offered several possible explanations for the association between diverticulitis and new diagnosis of IBS.
"Inflammation may alter gastrointestinal reflexes, amplify visceral sensitivity, render the bowel more susceptible to negative effects of microbiota, and alter motility in IBS," she said.
"Another putative mechanism of chronic diverticular disease involves shifts in intestinal microbiota leading to chronic inflammation, similar to theoretical models of IBS," Dr. Cohen said.
"Future research should identify demographic and clinical predictors of post-diverticulitis irritable bowel syndrome and evaluate its incidence in prospective studies to better determine whether the link is causal or merely associative," she concluded.
Finally, a fourth study could help researchers reduce the risk of the painful inflammatory condition: Among diverticulosis patients, higher levels of serum vitamin D were associated significantly with a lower risk of diverticulitis.
In her analysis, also published in the December issue of Clinical Gastroenterology and Hepatology, Dr. Lillias H. Maguire and colleagues identified 9,116 diverticulosis patients and 922 diverticulitis patients from the Partners Healthcare Research Patient Data Registry (doi:10.1016/j.cgh.2013.07.035). All patients had at least one prediagnostic serum vitamin D level on record between 1993 and 2012.
Dr. Maguire of Massachusetts General Hospital, Boston, found that patients with uncomplicated diverticulosis had mean levels of 29.1 ng/mL, versus 25.3 ng/mL among the diverticulitis patients (P less than .0001).
A sensitivity analysis that compared the mean prediagnostic values between cases and controls who had more than one reported vitamin D level yielded similarly significant results: The mean vitamin D level of uncomplicated diverticulosis was 33.0 ng/mL, compared with 28.1 ng/mL for acute diverticulitis patients (P less than .0001), 28.8 ng/mL for complicated diverticulitis patients (P = .002), 23.9 ng/mL for surgical diverticulitis cases (P less than .0001), and 25.5 ng/mL for recurrent diverticulitis patients (P less than .0001).
Indeed, "Compared with patients with acute diverticulitis without other sequelae, patients in the subgroups who developed abscess, required surgery, or had recurrent attacks were observed to have lower prediagnostic levels of vitamin D."
These differences between diverticulitis subgroups did not reach significance except in the cohort of patients who required surgery, who had the lowest levels of all.
"Taken together with prior studies showing an inverse association of 25(OH)D and risk of colonic cancer and inflammatory bowel disease, these results highlight the potential importance of vitamin D in the maintenance of colonic health," the investigators wrote.
"Additional studies in cohorts with more detailed information on potential confounders of this association are warranted," they added.
Dr. Peery, whose study looked at fiber intake among diverticulosis patients, and her collaborators reported having no disclosures, and stated that they received funding from the National Institutes of Health.
Dr. Shahedi, who assessed the incidence of diverticulitis, disclosed that three coinvestigators are employees of Shire Pharmaceuticals, which sponsored their study. Other investigators disclosed ties to Amgen and Ironwood Pharmaceuticals.
Dr. Cohen’s coinvestigators, who studied the prevalence of IBS following diverticulitis, disclosed ties to Ironwood Pharmaceuticals, Prometheus, Takeda Pharmaceuticals, Amgen, Ritter Pharmaceuticals, and Shire. Two investigators were employees of Shire, which funded the research.
Finally, the coinvestigators of Dr. Maguire, who looked at vitamin D levels, reported ties to Shire, Bayer Health, Pfizer, Millennium Pharmaceuticals, and Pozen. They were funded by grants from the American College of Gastroenterology as well as the National Institutes of Health.
Among the widely held beliefs of both lay and medical communities are that a) diverticulosis is associated with constipation and low consumption of dietary fiber, b) individuals with diverticulosis should eat neither seeds nor nuts, and c) diverticulitis will occur in up to 25% of individuals over their lifetimes. Certitude is not the same as correctness and facts do not always support our most cherished beliefs. This is highlighted by four recent studies published in the December issue of Clinical Gastroenterology and Hepatology.
A cross-sectional study by Peery et al. showed that the first of these beliefs to not be true and also found that nonwhite subjects had a 26% lower risk than did whites even after adjustment for risk factors. This suggests that earlier studies demonstrating a low prevalence of diverticulosis in African populations may have reflected, in part, racial differences rather than dietary issues.
A large study by Shahedi et al., of mostly male patients with incidental diverticulosis found by colonoscopy, suggests that the risk of developing diverticulitis has been vastly overestimated. This may be indicative of the higher mix of asymptomatic diverticulosis discovered during screening colonoscopy, in contrast with earlier studies in which imaging was often performed on symptomatic patients.
Equally intriguing is the biologically plausible finding by Maguire et al., that higher serum levels of vitamin D may reduce the risk of diverticulitis. Screening for and correcting vitamin D deficiencies are widely accepted practices and easy to implement. It might also be mentioned that popcorn consumption was associated with a decreased incidence in diverticulitis according to a study published 5 years ago (JAMA 2008;300:907-14), a finding still not fully appreciated by either the lay or medical community.
Lastly, Cohen and colleagues provide evidence for an increased risk of developing irritable bowel syndrome after acute diverticulitis. Although these data can be conceptualized as similar to postinfectious IBS, this does not imply causality. Previous studies have suggested that treatment with antibiotics increases functional abdominal symptoms, including IBS (Am. J. Gastroenterol. 2002;97:104-8). Nevertheless, diverticulitis may have functional GI consequences beyond the acute event.
Despite what we think we know, many questions of clinical importance about diverticulosis remain to be answered (Am. J. Gastroenterol. 2012;107:1486-93).
Dr. Arnold Wald is professor of medicine in the division of gastroenterology and hepatology, University of Wisconsin School of Medicine and Public Health. He had no relevant conflicts of interest.
Among the widely held beliefs of both lay and medical communities are that a) diverticulosis is associated with constipation and low consumption of dietary fiber, b) individuals with diverticulosis should eat neither seeds nor nuts, and c) diverticulitis will occur in up to 25% of individuals over their lifetimes. Certitude is not the same as correctness and facts do not always support our most cherished beliefs. This is highlighted by four recent studies published in the December issue of Clinical Gastroenterology and Hepatology.
A cross-sectional study by Peery et al. showed that the first of these beliefs to not be true and also found that nonwhite subjects had a 26% lower risk than did whites even after adjustment for risk factors. This suggests that earlier studies demonstrating a low prevalence of diverticulosis in African populations may have reflected, in part, racial differences rather than dietary issues.
A large study by Shahedi et al., of mostly male patients with incidental diverticulosis found by colonoscopy, suggests that the risk of developing diverticulitis has been vastly overestimated. This may be indicative of the higher mix of asymptomatic diverticulosis discovered during screening colonoscopy, in contrast with earlier studies in which imaging was often performed on symptomatic patients.
Equally intriguing is the biologically plausible finding by Maguire et al., that higher serum levels of vitamin D may reduce the risk of diverticulitis. Screening for and correcting vitamin D deficiencies are widely accepted practices and easy to implement. It might also be mentioned that popcorn consumption was associated with a decreased incidence in diverticulitis according to a study published 5 years ago (JAMA 2008;300:907-14), a finding still not fully appreciated by either the lay or medical community.
Lastly, Cohen and colleagues provide evidence for an increased risk of developing irritable bowel syndrome after acute diverticulitis. Although these data can be conceptualized as similar to postinfectious IBS, this does not imply causality. Previous studies have suggested that treatment with antibiotics increases functional abdominal symptoms, including IBS (Am. J. Gastroenterol. 2002;97:104-8). Nevertheless, diverticulitis may have functional GI consequences beyond the acute event.
Despite what we think we know, many questions of clinical importance about diverticulosis remain to be answered (Am. J. Gastroenterol. 2012;107:1486-93).
Dr. Arnold Wald is professor of medicine in the division of gastroenterology and hepatology, University of Wisconsin School of Medicine and Public Health. He had no relevant conflicts of interest.
Among the widely held beliefs of both lay and medical communities are that a) diverticulosis is associated with constipation and low consumption of dietary fiber, b) individuals with diverticulosis should eat neither seeds nor nuts, and c) diverticulitis will occur in up to 25% of individuals over their lifetimes. Certitude is not the same as correctness and facts do not always support our most cherished beliefs. This is highlighted by four recent studies published in the December issue of Clinical Gastroenterology and Hepatology.
A cross-sectional study by Peery et al. showed that the first of these beliefs to not be true and also found that nonwhite subjects had a 26% lower risk than did whites even after adjustment for risk factors. This suggests that earlier studies demonstrating a low prevalence of diverticulosis in African populations may have reflected, in part, racial differences rather than dietary issues.
A large study by Shahedi et al., of mostly male patients with incidental diverticulosis found by colonoscopy, suggests that the risk of developing diverticulitis has been vastly overestimated. This may be indicative of the higher mix of asymptomatic diverticulosis discovered during screening colonoscopy, in contrast with earlier studies in which imaging was often performed on symptomatic patients.
Equally intriguing is the biologically plausible finding by Maguire et al., that higher serum levels of vitamin D may reduce the risk of diverticulitis. Screening for and correcting vitamin D deficiencies are widely accepted practices and easy to implement. It might also be mentioned that popcorn consumption was associated with a decreased incidence in diverticulitis according to a study published 5 years ago (JAMA 2008;300:907-14), a finding still not fully appreciated by either the lay or medical community.
Lastly, Cohen and colleagues provide evidence for an increased risk of developing irritable bowel syndrome after acute diverticulitis. Although these data can be conceptualized as similar to postinfectious IBS, this does not imply causality. Previous studies have suggested that treatment with antibiotics increases functional abdominal symptoms, including IBS (Am. J. Gastroenterol. 2002;97:104-8). Nevertheless, diverticulitis may have functional GI consequences beyond the acute event.
Despite what we think we know, many questions of clinical importance about diverticulosis remain to be answered (Am. J. Gastroenterol. 2012;107:1486-93).
Dr. Arnold Wald is professor of medicine in the division of gastroenterology and hepatology, University of Wisconsin School of Medicine and Public Health. He had no relevant conflicts of interest.
Not only is there no link between low-fiber diets and diverticulosis, but the incidence of diverticulitis is not nearly as common as was previously believed.
Those are the conclusions of two new studies in the December issue of Clinical Gastroenterology and Hepatology, both of which challenge long-held beliefs about the causes of these conditions.
In the first study, Dr. Anne F. Peery of the University of North Carolina at Chapel Hill, and her colleagues looked at 539 patients with colonic diverticula and 1,569 controls, all culled from the Vitamin D and Calcium Polyp Prevention Study, a double-blind, placebo-controlled trial of vitamin D and/or calcium for the prevention of colonic adenomas (doi:10.1016/j.cgh.2013.06.033).
Patients with a self-reported history of diverticulosis or diverticulitis were excluded, as were cases with a history of colon resection, inflammatory bowel disease, or familial history of colon cancer. Most cases (88%) had descending or sigmoid colon diverticula, and these patients were significantly older and more likely to be male than were the controls.
According to Dr. Peery and colleagues, there was no difference between cases and controls in terms of mean dietary fiber intake (14.8 g per day versus 15.3 g per day, P = .2) and reported supplemental fiber intake (5% versus 5%, P = .7).
Nor was there any significant link when investigators compared the highest quartile of fiber intake (mean, 25 g/day) to the lowest (mean, 8 g/day) (odds ratio = 0.96; 95% confidence interval, 0.71-1.30).
Finally, the investigators found no associations between dietary fiber intake by subtype (for instance, beans, grains, fruits, and vegetables) and diverticulosis.
"Forty years ago, Dr. Neil Painter popularized the hypothesis that inadequate dietary fiber intake and constipation were the cause of sigmoid diverticulosis," wrote Dr. Peery. However, "Although the fiber hypothesis is conceptually attractive and widely accepted, it has not been rigorously examined."
And while Dr. Peery’s data were based on a food frequency questionnaire – which could be subject to measurement bias – she added that "the mean total fiber intake in the highest quartile was 25 g, versus 8 g in the lowest.
"This wide range makes it unlikely that homogeneity of intake accounts for the null association of fiber with the presence of diverticula," she wrote.
A second study by Dr. Kamyar Shahedi of the University of California Los Angeles/Veteran’s Affairs Center for Outcomes Research and Education, also sought evidence for the commonly held belief that up to 25% of patients with diverticulosis will develop diverticulitis.
Dr. Shahedi and colleagues performed a retrospective survival analysis of 2,222 patients from the Veteran’s Affairs Greater Los Angeles Healthcare System with colonic diverticulosis and a median follow-up of 6.75 years (doi:10.1016/j.cgh.2013.06.020). Patients were excluded if they had any ICD-9 code for diverticulitis or documentation of diverticulitis in the medical record notes at any point before the index date of diverticulosis.
When the researchers looked only at imaging-confirmed or surgical specimen–confirmed cases, just 23 patients (1%) developed acute diverticulitis during the study period, Dr. Shahedi found. This jumped to 95 patients (4.3%) when clinical diagnoses were also used, for an incidence of 6 cases per 1,000 patient years.
Looking at predictors for progression, the authors found that only age was related to the development of diverticulitis, with every year of age at diverticulosis detection conferring a 2.4% lower hazard of developing diverticulitis.
According to the authors, the "widely cited figures" that up to a quarter of patients with diverticulosis will develop acute diverticulitis is based on data collected before the time of routine colon screening. "Therefore, the true denominator of individuals harboring diverticulosis was not accounted for in these calculations," they concluded.
And while their retrospective study does leave room for the possibility that cases were missed, "Future series or patient registries may better standardize the definition of diverticulitis in a prospective cohort," wrote the investigators.
In the meantime, prevalence data such as these "may help to reframe discussions with patients regarding their probability of developing clinically significant diverticulitis."
However, even as these two findings change the way providers counsel patients about the cause and impact of diverticula, a third study, also in December’s issue of Clinical Gastroenterology and Hepatology, adds another wrinkle: Patients who do develop diverticulitis are at increased risk for a diagnosis of irritable bowel syndrome later on.
Dr. Erica Cohen of the VA Greater Los Angeles Healthcare System, and colleagues looked at 1,105 chart-confirmed cases of diverticulitis, identified retrospectively from the same dataset used by Dr. Shahedi (doi:10.1016/j.cgh.2013.03.007).
All cases were matched with controls seen on the same day, the mean follow-up period was 6.3 years, and patients with pre-existing IBS or functional bowel diagnoses were excluded from the study.
The primary outcome was a new IBS diagnosis after the index diverticulitis attack (for cases) or enrollment date (for controls). Ultimately, Dr. Cohen found 24 cases of newly diagnosed IBS during the study period: 20 among diverticulitis cases, and 4 among controls. That translated to a hazard ratio of 4.7 among cases compared with controls, even after adjustment for age, sex, ethnicity, race, inpatient versus outpatient status, and comorbidity score (95% CI, 1.6 –14.0; P = .006).
Dr. Cohen offered several possible explanations for the association between diverticulitis and new diagnosis of IBS.
"Inflammation may alter gastrointestinal reflexes, amplify visceral sensitivity, render the bowel more susceptible to negative effects of microbiota, and alter motility in IBS," she said.
"Another putative mechanism of chronic diverticular disease involves shifts in intestinal microbiota leading to chronic inflammation, similar to theoretical models of IBS," Dr. Cohen said.
"Future research should identify demographic and clinical predictors of post-diverticulitis irritable bowel syndrome and evaluate its incidence in prospective studies to better determine whether the link is causal or merely associative," she concluded.
Finally, a fourth study could help researchers reduce the risk of the painful inflammatory condition: Among diverticulosis patients, higher levels of serum vitamin D were associated significantly with a lower risk of diverticulitis.
In her analysis, also published in the December issue of Clinical Gastroenterology and Hepatology, Dr. Lillias H. Maguire and colleagues identified 9,116 diverticulosis patients and 922 diverticulitis patients from the Partners Healthcare Research Patient Data Registry (doi:10.1016/j.cgh.2013.07.035). All patients had at least one prediagnostic serum vitamin D level on record between 1993 and 2012.
Dr. Maguire of Massachusetts General Hospital, Boston, found that patients with uncomplicated diverticulosis had mean levels of 29.1 ng/mL, versus 25.3 ng/mL among the diverticulitis patients (P less than .0001).
A sensitivity analysis that compared the mean prediagnostic values between cases and controls who had more than one reported vitamin D level yielded similarly significant results: The mean vitamin D level of uncomplicated diverticulosis was 33.0 ng/mL, compared with 28.1 ng/mL for acute diverticulitis patients (P less than .0001), 28.8 ng/mL for complicated diverticulitis patients (P = .002), 23.9 ng/mL for surgical diverticulitis cases (P less than .0001), and 25.5 ng/mL for recurrent diverticulitis patients (P less than .0001).
Indeed, "Compared with patients with acute diverticulitis without other sequelae, patients in the subgroups who developed abscess, required surgery, or had recurrent attacks were observed to have lower prediagnostic levels of vitamin D."
These differences between diverticulitis subgroups did not reach significance except in the cohort of patients who required surgery, who had the lowest levels of all.
"Taken together with prior studies showing an inverse association of 25(OH)D and risk of colonic cancer and inflammatory bowel disease, these results highlight the potential importance of vitamin D in the maintenance of colonic health," the investigators wrote.
"Additional studies in cohorts with more detailed information on potential confounders of this association are warranted," they added.
Dr. Peery, whose study looked at fiber intake among diverticulosis patients, and her collaborators reported having no disclosures, and stated that they received funding from the National Institutes of Health.
Dr. Shahedi, who assessed the incidence of diverticulitis, disclosed that three coinvestigators are employees of Shire Pharmaceuticals, which sponsored their study. Other investigators disclosed ties to Amgen and Ironwood Pharmaceuticals.
Dr. Cohen’s coinvestigators, who studied the prevalence of IBS following diverticulitis, disclosed ties to Ironwood Pharmaceuticals, Prometheus, Takeda Pharmaceuticals, Amgen, Ritter Pharmaceuticals, and Shire. Two investigators were employees of Shire, which funded the research.
Finally, the coinvestigators of Dr. Maguire, who looked at vitamin D levels, reported ties to Shire, Bayer Health, Pfizer, Millennium Pharmaceuticals, and Pozen. They were funded by grants from the American College of Gastroenterology as well as the National Institutes of Health.
Not only is there no link between low-fiber diets and diverticulosis, but the incidence of diverticulitis is not nearly as common as was previously believed.
Those are the conclusions of two new studies in the December issue of Clinical Gastroenterology and Hepatology, both of which challenge long-held beliefs about the causes of these conditions.
In the first study, Dr. Anne F. Peery of the University of North Carolina at Chapel Hill, and her colleagues looked at 539 patients with colonic diverticula and 1,569 controls, all culled from the Vitamin D and Calcium Polyp Prevention Study, a double-blind, placebo-controlled trial of vitamin D and/or calcium for the prevention of colonic adenomas (doi:10.1016/j.cgh.2013.06.033).
Patients with a self-reported history of diverticulosis or diverticulitis were excluded, as were cases with a history of colon resection, inflammatory bowel disease, or familial history of colon cancer. Most cases (88%) had descending or sigmoid colon diverticula, and these patients were significantly older and more likely to be male than were the controls.
According to Dr. Peery and colleagues, there was no difference between cases and controls in terms of mean dietary fiber intake (14.8 g per day versus 15.3 g per day, P = .2) and reported supplemental fiber intake (5% versus 5%, P = .7).
Nor was there any significant link when investigators compared the highest quartile of fiber intake (mean, 25 g/day) to the lowest (mean, 8 g/day) (odds ratio = 0.96; 95% confidence interval, 0.71-1.30).
Finally, the investigators found no associations between dietary fiber intake by subtype (for instance, beans, grains, fruits, and vegetables) and diverticulosis.
"Forty years ago, Dr. Neil Painter popularized the hypothesis that inadequate dietary fiber intake and constipation were the cause of sigmoid diverticulosis," wrote Dr. Peery. However, "Although the fiber hypothesis is conceptually attractive and widely accepted, it has not been rigorously examined."
And while Dr. Peery’s data were based on a food frequency questionnaire – which could be subject to measurement bias – she added that "the mean total fiber intake in the highest quartile was 25 g, versus 8 g in the lowest.
"This wide range makes it unlikely that homogeneity of intake accounts for the null association of fiber with the presence of diverticula," she wrote.
A second study by Dr. Kamyar Shahedi of the University of California Los Angeles/Veteran’s Affairs Center for Outcomes Research and Education, also sought evidence for the commonly held belief that up to 25% of patients with diverticulosis will develop diverticulitis.
Dr. Shahedi and colleagues performed a retrospective survival analysis of 2,222 patients from the Veteran’s Affairs Greater Los Angeles Healthcare System with colonic diverticulosis and a median follow-up of 6.75 years (doi:10.1016/j.cgh.2013.06.020). Patients were excluded if they had any ICD-9 code for diverticulitis or documentation of diverticulitis in the medical record notes at any point before the index date of diverticulosis.
When the researchers looked only at imaging-confirmed or surgical specimen–confirmed cases, just 23 patients (1%) developed acute diverticulitis during the study period, Dr. Shahedi found. This jumped to 95 patients (4.3%) when clinical diagnoses were also used, for an incidence of 6 cases per 1,000 patient years.
Looking at predictors for progression, the authors found that only age was related to the development of diverticulitis, with every year of age at diverticulosis detection conferring a 2.4% lower hazard of developing diverticulitis.
According to the authors, the "widely cited figures" that up to a quarter of patients with diverticulosis will develop acute diverticulitis is based on data collected before the time of routine colon screening. "Therefore, the true denominator of individuals harboring diverticulosis was not accounted for in these calculations," they concluded.
And while their retrospective study does leave room for the possibility that cases were missed, "Future series or patient registries may better standardize the definition of diverticulitis in a prospective cohort," wrote the investigators.
In the meantime, prevalence data such as these "may help to reframe discussions with patients regarding their probability of developing clinically significant diverticulitis."
However, even as these two findings change the way providers counsel patients about the cause and impact of diverticula, a third study, also in December’s issue of Clinical Gastroenterology and Hepatology, adds another wrinkle: Patients who do develop diverticulitis are at increased risk for a diagnosis of irritable bowel syndrome later on.
Dr. Erica Cohen of the VA Greater Los Angeles Healthcare System, and colleagues looked at 1,105 chart-confirmed cases of diverticulitis, identified retrospectively from the same dataset used by Dr. Shahedi (doi:10.1016/j.cgh.2013.03.007).
All cases were matched with controls seen on the same day, the mean follow-up period was 6.3 years, and patients with pre-existing IBS or functional bowel diagnoses were excluded from the study.
The primary outcome was a new IBS diagnosis after the index diverticulitis attack (for cases) or enrollment date (for controls). Ultimately, Dr. Cohen found 24 cases of newly diagnosed IBS during the study period: 20 among diverticulitis cases, and 4 among controls. That translated to a hazard ratio of 4.7 among cases compared with controls, even after adjustment for age, sex, ethnicity, race, inpatient versus outpatient status, and comorbidity score (95% CI, 1.6 –14.0; P = .006).
Dr. Cohen offered several possible explanations for the association between diverticulitis and new diagnosis of IBS.
"Inflammation may alter gastrointestinal reflexes, amplify visceral sensitivity, render the bowel more susceptible to negative effects of microbiota, and alter motility in IBS," she said.
"Another putative mechanism of chronic diverticular disease involves shifts in intestinal microbiota leading to chronic inflammation, similar to theoretical models of IBS," Dr. Cohen said.
"Future research should identify demographic and clinical predictors of post-diverticulitis irritable bowel syndrome and evaluate its incidence in prospective studies to better determine whether the link is causal or merely associative," she concluded.
Finally, a fourth study could help researchers reduce the risk of the painful inflammatory condition: Among diverticulosis patients, higher levels of serum vitamin D were associated significantly with a lower risk of diverticulitis.
In her analysis, also published in the December issue of Clinical Gastroenterology and Hepatology, Dr. Lillias H. Maguire and colleagues identified 9,116 diverticulosis patients and 922 diverticulitis patients from the Partners Healthcare Research Patient Data Registry (doi:10.1016/j.cgh.2013.07.035). All patients had at least one prediagnostic serum vitamin D level on record between 1993 and 2012.
Dr. Maguire of Massachusetts General Hospital, Boston, found that patients with uncomplicated diverticulosis had mean levels of 29.1 ng/mL, versus 25.3 ng/mL among the diverticulitis patients (P less than .0001).
A sensitivity analysis that compared the mean prediagnostic values between cases and controls who had more than one reported vitamin D level yielded similarly significant results: The mean vitamin D level of uncomplicated diverticulosis was 33.0 ng/mL, compared with 28.1 ng/mL for acute diverticulitis patients (P less than .0001), 28.8 ng/mL for complicated diverticulitis patients (P = .002), 23.9 ng/mL for surgical diverticulitis cases (P less than .0001), and 25.5 ng/mL for recurrent diverticulitis patients (P less than .0001).
Indeed, "Compared with patients with acute diverticulitis without other sequelae, patients in the subgroups who developed abscess, required surgery, or had recurrent attacks were observed to have lower prediagnostic levels of vitamin D."
These differences between diverticulitis subgroups did not reach significance except in the cohort of patients who required surgery, who had the lowest levels of all.
"Taken together with prior studies showing an inverse association of 25(OH)D and risk of colonic cancer and inflammatory bowel disease, these results highlight the potential importance of vitamin D in the maintenance of colonic health," the investigators wrote.
"Additional studies in cohorts with more detailed information on potential confounders of this association are warranted," they added.
Dr. Peery, whose study looked at fiber intake among diverticulosis patients, and her collaborators reported having no disclosures, and stated that they received funding from the National Institutes of Health.
Dr. Shahedi, who assessed the incidence of diverticulitis, disclosed that three coinvestigators are employees of Shire Pharmaceuticals, which sponsored their study. Other investigators disclosed ties to Amgen and Ironwood Pharmaceuticals.
Dr. Cohen’s coinvestigators, who studied the prevalence of IBS following diverticulitis, disclosed ties to Ironwood Pharmaceuticals, Prometheus, Takeda Pharmaceuticals, Amgen, Ritter Pharmaceuticals, and Shire. Two investigators were employees of Shire, which funded the research.
Finally, the coinvestigators of Dr. Maguire, who looked at vitamin D levels, reported ties to Shire, Bayer Health, Pfizer, Millennium Pharmaceuticals, and Pozen. They were funded by grants from the American College of Gastroenterology as well as the National Institutes of Health.
FROM CLINICAL GASTROENTEROLOGY AND HEPATOLOGY
Major finding: The development of diverticulosis is not likely related to dietary fiber intake, and the progression of diverticulosis to diverticulitis may be less common than is now thought. On the other hand, patients who do develop diverticulitis may be at higher risk for irritable bowel syndrome, and ensuring adequate levels of serum vitamin D may help prevent diverticulitis altogether.
Data source: Four studies on diverticulosis and diverticulitis.
Disclosures: Dr. Peery and her collaborators stated that they received funding from the National Institutes of Health. Dr. Shahedi disclosed that three coinvestigators are employees of Shire Pharmaceuticals, which sponsored their study. Other investigators disclosed ties to Amgen and Ironwood Pharmaceuticals. Dr. Cohen’s coinvestigators disclosed ties to Ironwood Pharmaceuticals, Prometheus, Takeda Pharmaceuticals, Amgen, Ritter Pharmaceuticals, and Shire. Two investigators were employees of Shire, which funded their research. Dr. Maguire’s coinvestigators reported ties to Shire, Bayer Health, Pfizer, Millennium Pharmaceuticals, and Pozen. They stated that they were funded by grants from the American College of Gastroenterology and the National Institutes of Health.
Fecal transplant is cost effective for treating recurrent C. difficile
SAN DIEGO – Fecal microbiota transplantation by colonoscopy is cost-effective when used as the initial treatment for recurrent Clostridium difficile infection, new data show.
A team led by Dr. Gauree Konijeti, a gastroenterology fellow at Massachusetts General Hospital in Boston, constructed decision analytic models of various treatment strategies in a hypothetical cohort of patients with a first, mild to moderate recurrence of C. difficile infection.
Relative to vancomycin, fecal microbiota transplantation (FMT) delivered by colonoscopy had an incremental cost-effectiveness ratio (ICER) of about $38,000 per quality-adjusted life-year gained, placing it well within the conventional willingness-to-pay threshold of $50,000, she reported at the annual meeting of the American College of Gastroenterology.
Additionally, FMT colonoscopy was more effective and less costly than both metronidazole (Flagyl) and fidaxomicin (Dificid).
However, FMT using other modes of delivery – either enema or duodenal infusion through esophagogastroduodenoscopy – was not cost-effective because of its relatively lower cure rates.
"A strategy consisting of first-line treatment with FMT colonoscopy for an initial recurrence of C. difficile appeared cost-effective at conventional willingness-to-pay thresholds," Dr. Konijeti commented.
"Guidelines should consider earlier use of FMT in the treatment of C. difficile infection, and future studies should incorporate FMT for comparative effectiveness," she recommended.
A session attendee asked, "What are your thoughts on the future of noncolonoscopic delivery methods?"
"One is if we can increase the infusion cure rates for enema and duodenal infusion, or even nasogastric infusion, those would be cost-effective. Right now they are on the order of 81%, compared to colonoscopic delivery, which is closer to the 93%-95% range," Dr. Konijeti replied.
"The other thought is that there has been a study of a fecal transplant pill that was recently presented at ID Week, where they used fresh donor feces from related donors and encapsulated a concentrated form of bacteria into these pills, and then gave about 20 pills to patients in a case series. They showed about a 100% efficacy rate, with only one recurrence in the setting of antibiotics," she said. "So I think we are in an era where we have the opportunity to deliver FMT via a variety of strategies, but we need to find more standardized ways of doing it and then optimize the efficacy."
Giving some background to the research, Dr. Konijeti noted that C. difficile infection has become increasingly challenging to manage. Emergence of the 027 strain has led to lower cure rates and higher rates of resistance. Today, up to one-third of patients have a recurrence after an initial infection, and up to two-thirds of that group go on to have yet more recurrences.
"FMT has emerged as a highly effective therapy because of high cure rates and low rates of recurrence," she commented.
The investigators studied four competing treatment strategies – vancomycin, metronidazole, fidaxomicin, and FMT – for treatment of a first, mild to moderate recurrence of C. difficile infection in a hypothetical cohort of patients having a median age of 65 years.
The models used various subsequent treatments in the event of a second and third recurrence, and the time horizon was 6 months. A key assumption was that payers would be willing to pay up to $50,000 per quality-adjusted life-year gained.
Base-case results showed that FMT colonoscopy was the most cost-effective strategy relative to vancomycin, with an ICER of $38,382 per quality-adjusted life-year gained, and was much more effective than both metronidazole and fidaxomicin.
However, in sensitivity analyses, FMT delivered by duodenal infusion or enema was not superior to other strategies.
Additional analyses tinkering with various model components showed that FMT colonoscopy was the most cost-effective strategy as long as its cure rate exceeded 93.8%, its cost was less than $2,324, or the probability of a post-treatment recurrence was less than 10%.
When the investigators explored thresholds for other treatment strategies, they found vancomycin would be the most cost-effective if its post-treatment recurrence rate were less than 33.9% (vs. 35.5% in the base case); fidaxomicin if its cost dropped to less than $1,539 (vs. $2,800 in the base case); and FMT by duodenal infusion or enema if the cure rate with one-time infusion hit 89.4% and 88.8%, respectively (vs. 81.3% and 81.5%).
Finally, when analyses assumed that FMT was not available, vancomycin was the most cost-effective strategy.
Dr. Konijeti disclosed no relevant conflicts of interest.
SAN DIEGO – Fecal microbiota transplantation by colonoscopy is cost-effective when used as the initial treatment for recurrent Clostridium difficile infection, new data show.
A team led by Dr. Gauree Konijeti, a gastroenterology fellow at Massachusetts General Hospital in Boston, constructed decision analytic models of various treatment strategies in a hypothetical cohort of patients with a first, mild to moderate recurrence of C. difficile infection.
Relative to vancomycin, fecal microbiota transplantation (FMT) delivered by colonoscopy had an incremental cost-effectiveness ratio (ICER) of about $38,000 per quality-adjusted life-year gained, placing it well within the conventional willingness-to-pay threshold of $50,000, she reported at the annual meeting of the American College of Gastroenterology.
Additionally, FMT colonoscopy was more effective and less costly than both metronidazole (Flagyl) and fidaxomicin (Dificid).
However, FMT using other modes of delivery – either enema or duodenal infusion through esophagogastroduodenoscopy – was not cost-effective because of its relatively lower cure rates.
"A strategy consisting of first-line treatment with FMT colonoscopy for an initial recurrence of C. difficile appeared cost-effective at conventional willingness-to-pay thresholds," Dr. Konijeti commented.
"Guidelines should consider earlier use of FMT in the treatment of C. difficile infection, and future studies should incorporate FMT for comparative effectiveness," she recommended.
A session attendee asked, "What are your thoughts on the future of noncolonoscopic delivery methods?"
"One is if we can increase the infusion cure rates for enema and duodenal infusion, or even nasogastric infusion, those would be cost-effective. Right now they are on the order of 81%, compared to colonoscopic delivery, which is closer to the 93%-95% range," Dr. Konijeti replied.
"The other thought is that there has been a study of a fecal transplant pill that was recently presented at ID Week, where they used fresh donor feces from related donors and encapsulated a concentrated form of bacteria into these pills, and then gave about 20 pills to patients in a case series. They showed about a 100% efficacy rate, with only one recurrence in the setting of antibiotics," she said. "So I think we are in an era where we have the opportunity to deliver FMT via a variety of strategies, but we need to find more standardized ways of doing it and then optimize the efficacy."
Giving some background to the research, Dr. Konijeti noted that C. difficile infection has become increasingly challenging to manage. Emergence of the 027 strain has led to lower cure rates and higher rates of resistance. Today, up to one-third of patients have a recurrence after an initial infection, and up to two-thirds of that group go on to have yet more recurrences.
"FMT has emerged as a highly effective therapy because of high cure rates and low rates of recurrence," she commented.
The investigators studied four competing treatment strategies – vancomycin, metronidazole, fidaxomicin, and FMT – for treatment of a first, mild to moderate recurrence of C. difficile infection in a hypothetical cohort of patients having a median age of 65 years.
The models used various subsequent treatments in the event of a second and third recurrence, and the time horizon was 6 months. A key assumption was that payers would be willing to pay up to $50,000 per quality-adjusted life-year gained.
Base-case results showed that FMT colonoscopy was the most cost-effective strategy relative to vancomycin, with an ICER of $38,382 per quality-adjusted life-year gained, and was much more effective than both metronidazole and fidaxomicin.
However, in sensitivity analyses, FMT delivered by duodenal infusion or enema was not superior to other strategies.
Additional analyses tinkering with various model components showed that FMT colonoscopy was the most cost-effective strategy as long as its cure rate exceeded 93.8%, its cost was less than $2,324, or the probability of a post-treatment recurrence was less than 10%.
When the investigators explored thresholds for other treatment strategies, they found vancomycin would be the most cost-effective if its post-treatment recurrence rate were less than 33.9% (vs. 35.5% in the base case); fidaxomicin if its cost dropped to less than $1,539 (vs. $2,800 in the base case); and FMT by duodenal infusion or enema if the cure rate with one-time infusion hit 89.4% and 88.8%, respectively (vs. 81.3% and 81.5%).
Finally, when analyses assumed that FMT was not available, vancomycin was the most cost-effective strategy.
Dr. Konijeti disclosed no relevant conflicts of interest.
SAN DIEGO – Fecal microbiota transplantation by colonoscopy is cost-effective when used as the initial treatment for recurrent Clostridium difficile infection, new data show.
A team led by Dr. Gauree Konijeti, a gastroenterology fellow at Massachusetts General Hospital in Boston, constructed decision analytic models of various treatment strategies in a hypothetical cohort of patients with a first, mild to moderate recurrence of C. difficile infection.
Relative to vancomycin, fecal microbiota transplantation (FMT) delivered by colonoscopy had an incremental cost-effectiveness ratio (ICER) of about $38,000 per quality-adjusted life-year gained, placing it well within the conventional willingness-to-pay threshold of $50,000, she reported at the annual meeting of the American College of Gastroenterology.
Additionally, FMT colonoscopy was more effective and less costly than both metronidazole (Flagyl) and fidaxomicin (Dificid).
However, FMT using other modes of delivery – either enema or duodenal infusion through esophagogastroduodenoscopy – was not cost-effective because of its relatively lower cure rates.
"A strategy consisting of first-line treatment with FMT colonoscopy for an initial recurrence of C. difficile appeared cost-effective at conventional willingness-to-pay thresholds," Dr. Konijeti commented.
"Guidelines should consider earlier use of FMT in the treatment of C. difficile infection, and future studies should incorporate FMT for comparative effectiveness," she recommended.
A session attendee asked, "What are your thoughts on the future of noncolonoscopic delivery methods?"
"One is if we can increase the infusion cure rates for enema and duodenal infusion, or even nasogastric infusion, those would be cost-effective. Right now they are on the order of 81%, compared to colonoscopic delivery, which is closer to the 93%-95% range," Dr. Konijeti replied.
"The other thought is that there has been a study of a fecal transplant pill that was recently presented at ID Week, where they used fresh donor feces from related donors and encapsulated a concentrated form of bacteria into these pills, and then gave about 20 pills to patients in a case series. They showed about a 100% efficacy rate, with only one recurrence in the setting of antibiotics," she said. "So I think we are in an era where we have the opportunity to deliver FMT via a variety of strategies, but we need to find more standardized ways of doing it and then optimize the efficacy."
Giving some background to the research, Dr. Konijeti noted that C. difficile infection has become increasingly challenging to manage. Emergence of the 027 strain has led to lower cure rates and higher rates of resistance. Today, up to one-third of patients have a recurrence after an initial infection, and up to two-thirds of that group go on to have yet more recurrences.
"FMT has emerged as a highly effective therapy because of high cure rates and low rates of recurrence," she commented.
The investigators studied four competing treatment strategies – vancomycin, metronidazole, fidaxomicin, and FMT – for treatment of a first, mild to moderate recurrence of C. difficile infection in a hypothetical cohort of patients having a median age of 65 years.
The models used various subsequent treatments in the event of a second and third recurrence, and the time horizon was 6 months. A key assumption was that payers would be willing to pay up to $50,000 per quality-adjusted life-year gained.
Base-case results showed that FMT colonoscopy was the most cost-effective strategy relative to vancomycin, with an ICER of $38,382 per quality-adjusted life-year gained, and was much more effective than both metronidazole and fidaxomicin.
However, in sensitivity analyses, FMT delivered by duodenal infusion or enema was not superior to other strategies.
Additional analyses tinkering with various model components showed that FMT colonoscopy was the most cost-effective strategy as long as its cure rate exceeded 93.8%, its cost was less than $2,324, or the probability of a post-treatment recurrence was less than 10%.
When the investigators explored thresholds for other treatment strategies, they found vancomycin would be the most cost-effective if its post-treatment recurrence rate were less than 33.9% (vs. 35.5% in the base case); fidaxomicin if its cost dropped to less than $1,539 (vs. $2,800 in the base case); and FMT by duodenal infusion or enema if the cure rate with one-time infusion hit 89.4% and 88.8%, respectively (vs. 81.3% and 81.5%).
Finally, when analyses assumed that FMT was not available, vancomycin was the most cost-effective strategy.
Dr. Konijeti disclosed no relevant conflicts of interest.
AT THE ACG ANNUAL MEETING
Major finding: Fecal microbiota transplantation by colonoscopy had an ICER of $38,382 per quality-adjusted life-year gained relative to vancomycin treatment, and it dominated both metronidazole and fidaxomicin.
Data source: A decision analytic modeling study among patients with a first, mild to moderate recurrence of C. difficile infection.
Disclosures: Dr. Konijeti disclosed no relevant conflicts of interest.
Anti-vinculin antibody assay could be answer for diagnosis of IBS
SAN DIEGO – A blood test for antibodies to vinculin, a protein involved in nerve cell migration, may allow objective diagnosis of irritable bowel syndrome, a condition historically diagnosed clinically, after a thorough workup excludes other possibilities.
Investigators led by Dr. Mark Pimentel, director of the GI Motility Program at the Cedars-Sinai Medical Center in Los Angeles, performed a multicenter validation study of the test among patients with irritable bowel syndrome (IBS) or inflammatory bowel disease (IBD), and healthy individuals.
Study results, reported at the annual meeting of the American College of Gastroenterology, showed that the anti-vinculin antibody test had a positive predictive value of at least 90% for distinguishing IBS from IBD.
And when analyses also took into account antibodies to cytolethal distending toxin B (CdtB) – a toxin produced by bacteria commonly associated with food poisoning – the positive predictive value was at least 94%.
"Elevated anti-vinculin antibodies are specific for IBS compared to IBD, and an increase in anti-vinculin antibodies with respect to anti-CdtB increases that specificity," Dr. Pimentel said, summing up the findings. "This may be the first serum diagnostic biomarker that can discriminate IBS from IBD, and it would help avoid unnecessary tests."
Additionally, the findings lend support to a pathogenic mechanism for postinfectious IBS suggested by a rodent model, whereby bacterial gastroenteritis gives rise to autoimmunity against vinculin in the digestive tract.
The assay may be useful in IBD research too, he noted. "One of the problems with IBD studies is those patients who don’t respond to therapies, and maybe they have IBS and they don’t have IBD. Maybe this test could be used to screen those patients out before the study begins."
A session attendee expressed reservations about the study, noting that some analyses compared IBS patients with healthy individuals, and that positive predictive values may not be the best statistic given the composition of the study population.
"We don’t need a test to tell us someone that has no symptoms versus someone that does. So this is the start of your validation, not the end of it," he said. "If you apply this to the population right now, I’ve done some calculations, your positive predictive value would be about 20%. So it’s not that great in clinical practice. ... I’m sure you will develop this more and it will get better, but right now, I don’t think this is ready for prime time."
"First, you can use a likelihood ratio, which accounts for the volumes of patients. ... Our likelihood ratio is between 3 and 4, which I hope gives you more confidence in it," Dr. Pimentel replied. "The second thing is that the patients who arrive in a doctor’s office are not healthy: They are going to have IBD or IBS or something else if they have diarrhea in the clinic."
In a related press briefing, Dr. Brian E. Lacy, of the Dartmouth-Hitchcock Medical Center, Lebanon, N.H., commented, "This is an incredibly important topic, when we are talking about prevalence rates of IBS – a conservative rate is 12% to 15% – and when you are talking about spending $20 billion to $30 billion a year diagnosing and treating IBS."
"For many patients, IBS is a diagnosis of exclusion; they undergo a battery of unnecessary tests which are usually fruitless because this is a functional bowel disorder," he added. "To possibly have a diagnostic test – a blood test – that could confidently make the diagnosis of IBS to me would be incredibly important. And I think for the community primary care providers, family practice doctors, who are not confident at diagnosing IBS, to have somebody say, ‘This is a great test, and we can not only make the diagnosis, but exclude or maybe improve our ability to exclude the patients with IBD,’ that would be incredibly important."
The test may also have implications for treatment, according to Dr. Pimentel. "Another question is, could this antibody test predict who will respond to antibiotics, or does it predict bacterial overgrowth or other treatable aspects of IBS?" he explained.
Finally, such a test would help validate IBS as a legitimate medical condition. "IBS is a very, very difficult illness because nobody understands it, and it kind of gets the short end of the stick because it is viewed as a lifestyle disorder instead of a legitimate disease," he commented. "So what I’d like to do in my career is to make IBS a real disease, not just a syndrome as it’s been for at least the last 2 decades."
Press briefing moderator Dr. Michael E. Cox of the Mercy Medical Center in Baltimore, said, "The $64,000 question is, when would this possibly be ready for prime time?"
"We are validating this antibody every day," Dr. Pimentel replied, although as yet, no companies are collaborating in developing the assay. "When it will be ready for prime time, I’m not sure."
In the study, the investigators assayed serum samples from 162 prospectively identified patients who met Rome III criteria for IBS, 30 patients with active IBD who were not receiving biologic agents, and 26 consecutive healthy individuals.
Across groups, about 70% of patients were female, with no significant differences in the sex and age distributions.
Results showed that the anti-vinculin antibody optical density (OD) reading was higher in patients with IBS than in patients with IBD (P less than .01) and healthy individuals (P less than .01), reported Dr. Pimentel.
Meanwhile, the anti-CdtB antibody OD reading was higher in the patients with IBD than in the patients with IBS (P = .02).
For distinguishing IBS from IBD, an anti-vinculin antibody OD reading exceeding 0.8 had a sensitivity of 43%, a specificity of 73%, and a positive predictive value of 90%.
There is a good rationale for simultaneously looking at anti-CdtB and anti-vinculin, according to Dr. Pimentel: In the model of postinfectious IBS, anti-vinculin antibodies persist over time, whereas anti-CdtB antibodies decline.
And indeed, in the study, the difference between the two OD readings (anti-vinculin minus anti-CdtB) was higher in the patients with IBS than in both the patients with IBD (P less than .0001) and the healthy individuals (P less than .001).
For distinguishing IBS from IBD, a difference exceeding 0.2 had a sensitivity of 41%, a specificity of 88%, and a positive predictive value of 94%.
A confounding issue is that about 10% of patients with IBD also have IBS, Dr. Pimentel noted. But a model taking this into account showed high positive predictive values for an anti-vinculin antibody OD reading exceeding 0.8 (92%) and for a difference between the OD readings of anti-vinculin and anti-CdtB exceeding 0.2 (97%).
Dr. Pimentel disclosed no relevant conflicts of interest.
SAN DIEGO – A blood test for antibodies to vinculin, a protein involved in nerve cell migration, may allow objective diagnosis of irritable bowel syndrome, a condition historically diagnosed clinically, after a thorough workup excludes other possibilities.
Investigators led by Dr. Mark Pimentel, director of the GI Motility Program at the Cedars-Sinai Medical Center in Los Angeles, performed a multicenter validation study of the test among patients with irritable bowel syndrome (IBS) or inflammatory bowel disease (IBD), and healthy individuals.
Study results, reported at the annual meeting of the American College of Gastroenterology, showed that the anti-vinculin antibody test had a positive predictive value of at least 90% for distinguishing IBS from IBD.
And when analyses also took into account antibodies to cytolethal distending toxin B (CdtB) – a toxin produced by bacteria commonly associated with food poisoning – the positive predictive value was at least 94%.
"Elevated anti-vinculin antibodies are specific for IBS compared to IBD, and an increase in anti-vinculin antibodies with respect to anti-CdtB increases that specificity," Dr. Pimentel said, summing up the findings. "This may be the first serum diagnostic biomarker that can discriminate IBS from IBD, and it would help avoid unnecessary tests."
Additionally, the findings lend support to a pathogenic mechanism for postinfectious IBS suggested by a rodent model, whereby bacterial gastroenteritis gives rise to autoimmunity against vinculin in the digestive tract.
The assay may be useful in IBD research too, he noted. "One of the problems with IBD studies is those patients who don’t respond to therapies, and maybe they have IBS and they don’t have IBD. Maybe this test could be used to screen those patients out before the study begins."
A session attendee expressed reservations about the study, noting that some analyses compared IBS patients with healthy individuals, and that positive predictive values may not be the best statistic given the composition of the study population.
"We don’t need a test to tell us someone that has no symptoms versus someone that does. So this is the start of your validation, not the end of it," he said. "If you apply this to the population right now, I’ve done some calculations, your positive predictive value would be about 20%. So it’s not that great in clinical practice. ... I’m sure you will develop this more and it will get better, but right now, I don’t think this is ready for prime time."
"First, you can use a likelihood ratio, which accounts for the volumes of patients. ... Our likelihood ratio is between 3 and 4, which I hope gives you more confidence in it," Dr. Pimentel replied. "The second thing is that the patients who arrive in a doctor’s office are not healthy: They are going to have IBD or IBS or something else if they have diarrhea in the clinic."
In a related press briefing, Dr. Brian E. Lacy, of the Dartmouth-Hitchcock Medical Center, Lebanon, N.H., commented, "This is an incredibly important topic, when we are talking about prevalence rates of IBS – a conservative rate is 12% to 15% – and when you are talking about spending $20 billion to $30 billion a year diagnosing and treating IBS."
"For many patients, IBS is a diagnosis of exclusion; they undergo a battery of unnecessary tests which are usually fruitless because this is a functional bowel disorder," he added. "To possibly have a diagnostic test – a blood test – that could confidently make the diagnosis of IBS to me would be incredibly important. And I think for the community primary care providers, family practice doctors, who are not confident at diagnosing IBS, to have somebody say, ‘This is a great test, and we can not only make the diagnosis, but exclude or maybe improve our ability to exclude the patients with IBD,’ that would be incredibly important."
The test may also have implications for treatment, according to Dr. Pimentel. "Another question is, could this antibody test predict who will respond to antibiotics, or does it predict bacterial overgrowth or other treatable aspects of IBS?" he explained.
Finally, such a test would help validate IBS as a legitimate medical condition. "IBS is a very, very difficult illness because nobody understands it, and it kind of gets the short end of the stick because it is viewed as a lifestyle disorder instead of a legitimate disease," he commented. "So what I’d like to do in my career is to make IBS a real disease, not just a syndrome as it’s been for at least the last 2 decades."
Press briefing moderator Dr. Michael E. Cox of the Mercy Medical Center in Baltimore, said, "The $64,000 question is, when would this possibly be ready for prime time?"
"We are validating this antibody every day," Dr. Pimentel replied, although as yet, no companies are collaborating in developing the assay. "When it will be ready for prime time, I’m not sure."
In the study, the investigators assayed serum samples from 162 prospectively identified patients who met Rome III criteria for IBS, 30 patients with active IBD who were not receiving biologic agents, and 26 consecutive healthy individuals.
Across groups, about 70% of patients were female, with no significant differences in the sex and age distributions.
Results showed that the anti-vinculin antibody optical density (OD) reading was higher in patients with IBS than in patients with IBD (P less than .01) and healthy individuals (P less than .01), reported Dr. Pimentel.
Meanwhile, the anti-CdtB antibody OD reading was higher in the patients with IBD than in the patients with IBS (P = .02).
For distinguishing IBS from IBD, an anti-vinculin antibody OD reading exceeding 0.8 had a sensitivity of 43%, a specificity of 73%, and a positive predictive value of 90%.
There is a good rationale for simultaneously looking at anti-CdtB and anti-vinculin, according to Dr. Pimentel: In the model of postinfectious IBS, anti-vinculin antibodies persist over time, whereas anti-CdtB antibodies decline.
And indeed, in the study, the difference between the two OD readings (anti-vinculin minus anti-CdtB) was higher in the patients with IBS than in both the patients with IBD (P less than .0001) and the healthy individuals (P less than .001).
For distinguishing IBS from IBD, a difference exceeding 0.2 had a sensitivity of 41%, a specificity of 88%, and a positive predictive value of 94%.
A confounding issue is that about 10% of patients with IBD also have IBS, Dr. Pimentel noted. But a model taking this into account showed high positive predictive values for an anti-vinculin antibody OD reading exceeding 0.8 (92%) and for a difference between the OD readings of anti-vinculin and anti-CdtB exceeding 0.2 (97%).
Dr. Pimentel disclosed no relevant conflicts of interest.
SAN DIEGO – A blood test for antibodies to vinculin, a protein involved in nerve cell migration, may allow objective diagnosis of irritable bowel syndrome, a condition historically diagnosed clinically, after a thorough workup excludes other possibilities.
Investigators led by Dr. Mark Pimentel, director of the GI Motility Program at the Cedars-Sinai Medical Center in Los Angeles, performed a multicenter validation study of the test among patients with irritable bowel syndrome (IBS) or inflammatory bowel disease (IBD), and healthy individuals.
Study results, reported at the annual meeting of the American College of Gastroenterology, showed that the anti-vinculin antibody test had a positive predictive value of at least 90% for distinguishing IBS from IBD.
And when analyses also took into account antibodies to cytolethal distending toxin B (CdtB) – a toxin produced by bacteria commonly associated with food poisoning – the positive predictive value was at least 94%.
"Elevated anti-vinculin antibodies are specific for IBS compared to IBD, and an increase in anti-vinculin antibodies with respect to anti-CdtB increases that specificity," Dr. Pimentel said, summing up the findings. "This may be the first serum diagnostic biomarker that can discriminate IBS from IBD, and it would help avoid unnecessary tests."
Additionally, the findings lend support to a pathogenic mechanism for postinfectious IBS suggested by a rodent model, whereby bacterial gastroenteritis gives rise to autoimmunity against vinculin in the digestive tract.
The assay may be useful in IBD research too, he noted. "One of the problems with IBD studies is those patients who don’t respond to therapies, and maybe they have IBS and they don’t have IBD. Maybe this test could be used to screen those patients out before the study begins."
A session attendee expressed reservations about the study, noting that some analyses compared IBS patients with healthy individuals, and that positive predictive values may not be the best statistic given the composition of the study population.
"We don’t need a test to tell us someone that has no symptoms versus someone that does. So this is the start of your validation, not the end of it," he said. "If you apply this to the population right now, I’ve done some calculations, your positive predictive value would be about 20%. So it’s not that great in clinical practice. ... I’m sure you will develop this more and it will get better, but right now, I don’t think this is ready for prime time."
"First, you can use a likelihood ratio, which accounts for the volumes of patients. ... Our likelihood ratio is between 3 and 4, which I hope gives you more confidence in it," Dr. Pimentel replied. "The second thing is that the patients who arrive in a doctor’s office are not healthy: They are going to have IBD or IBS or something else if they have diarrhea in the clinic."
In a related press briefing, Dr. Brian E. Lacy, of the Dartmouth-Hitchcock Medical Center, Lebanon, N.H., commented, "This is an incredibly important topic, when we are talking about prevalence rates of IBS – a conservative rate is 12% to 15% – and when you are talking about spending $20 billion to $30 billion a year diagnosing and treating IBS."
"For many patients, IBS is a diagnosis of exclusion; they undergo a battery of unnecessary tests which are usually fruitless because this is a functional bowel disorder," he added. "To possibly have a diagnostic test – a blood test – that could confidently make the diagnosis of IBS to me would be incredibly important. And I think for the community primary care providers, family practice doctors, who are not confident at diagnosing IBS, to have somebody say, ‘This is a great test, and we can not only make the diagnosis, but exclude or maybe improve our ability to exclude the patients with IBD,’ that would be incredibly important."
The test may also have implications for treatment, according to Dr. Pimentel. "Another question is, could this antibody test predict who will respond to antibiotics, or does it predict bacterial overgrowth or other treatable aspects of IBS?" he explained.
Finally, such a test would help validate IBS as a legitimate medical condition. "IBS is a very, very difficult illness because nobody understands it, and it kind of gets the short end of the stick because it is viewed as a lifestyle disorder instead of a legitimate disease," he commented. "So what I’d like to do in my career is to make IBS a real disease, not just a syndrome as it’s been for at least the last 2 decades."
Press briefing moderator Dr. Michael E. Cox of the Mercy Medical Center in Baltimore, said, "The $64,000 question is, when would this possibly be ready for prime time?"
"We are validating this antibody every day," Dr. Pimentel replied, although as yet, no companies are collaborating in developing the assay. "When it will be ready for prime time, I’m not sure."
In the study, the investigators assayed serum samples from 162 prospectively identified patients who met Rome III criteria for IBS, 30 patients with active IBD who were not receiving biologic agents, and 26 consecutive healthy individuals.
Across groups, about 70% of patients were female, with no significant differences in the sex and age distributions.
Results showed that the anti-vinculin antibody optical density (OD) reading was higher in patients with IBS than in patients with IBD (P less than .01) and healthy individuals (P less than .01), reported Dr. Pimentel.
Meanwhile, the anti-CdtB antibody OD reading was higher in the patients with IBD than in the patients with IBS (P = .02).
For distinguishing IBS from IBD, an anti-vinculin antibody OD reading exceeding 0.8 had a sensitivity of 43%, a specificity of 73%, and a positive predictive value of 90%.
There is a good rationale for simultaneously looking at anti-CdtB and anti-vinculin, according to Dr. Pimentel: In the model of postinfectious IBS, anti-vinculin antibodies persist over time, whereas anti-CdtB antibodies decline.
And indeed, in the study, the difference between the two OD readings (anti-vinculin minus anti-CdtB) was higher in the patients with IBS than in both the patients with IBD (P less than .0001) and the healthy individuals (P less than .001).
For distinguishing IBS from IBD, a difference exceeding 0.2 had a sensitivity of 41%, a specificity of 88%, and a positive predictive value of 94%.
A confounding issue is that about 10% of patients with IBD also have IBS, Dr. Pimentel noted. But a model taking this into account showed high positive predictive values for an anti-vinculin antibody OD reading exceeding 0.8 (92%) and for a difference between the OD readings of anti-vinculin and anti-CdtB exceeding 0.2 (97%).
Dr. Pimentel disclosed no relevant conflicts of interest.
AT THE ACG ANNUAL MEETING
Major finding: For distinguishing IBS from IBD, anti-vinculin antibodies had a positive predictive value of at least 90% alone and at least 94% when combined with anti-CdtB antibodies.
Data source: A cross-sectional study of 162 patients with IBS, 30 patients with active IBD, and 26 healthy individuals.
Disclosures: Dr. Pimentel disclosed no relevant conflicts of interest.
Utility of three endoscopic IBD activity scores highlighted in two studies
A reduction of at least 50% in either of two different Crohn’s disease endoscopic activity scores after 26 weeks of treatment predicted those patients likely to be off of corticosteroid therapy after about 1 year of treatment, with a sensitivity approaching 75%, reported Dr. Marc Ferrante and his coinvestigators.
The study provides evidence that this cutoff could be used as a reliable predictor of clinical response, but it needs to be studied further, according to the authors, who are members of the International Organization for the Study of Inflammatory Bowel Diseases. The study was published in the Novemberissue of Gastroenterology.
Another study, published in the same issue of Gastroenterology, evaluated the reliability of the Ulcerative Colitis Endoscopic Index of Severity (UCEIS), an instrument recently created to assess the endoscopic severity of UC. This study randomized 25 investigators to assess and score 28 endoscopic videos of patients with UC – without knowledge of clinical features in most cases. The results indicated that the UCEIS provides a "satisfactory" level of intrainvestigator and interinvestigator agreement and that it is "a reliable instrument for measuring the endoscopic disease activity of UC," according to Dr. Simon Travis, of the translational gastroenterology unit at John Radcliffe Hospital, Oxford, England, and his coinvestigators.
The version of the UCEIS that provides a score from 0 to 8, based on findings of vascular pattern, bleeding, and erosions/ulcers, is the "favored version" but needs to be validated further, they concluded (Gastroenterol. 2013 July 29 [doi: 10.1053/j.gastro.2013.07.024]).
In the Crohn’s disease (CD) study, which used data on 172 patients enrolled in SONIC (Study of Biologic and Immunomodulator Naive Patients in Crohn’s Disease), Dr. Ferrante, of the department of gastroenterology at University Hospitals Leuven, Belgium, and his colleagues identified a cutoff point in two endoscopic response scores after 26 weeks of treatment that was predictive of clinical response at 50 weeks.
At baseline, the patients had endoscopic lesions and CD Activity Index (CDAI) scores of 220-450 points (median, 277). Their median age was 34 years, and they had been diagnosed with CD for a median of 2.5 years. The patients were dependent on corticosteroids and were randomized to treatment with infliximab (Remicade) infusions and/or oral azathioprine, and were followed to 50 weeks. The primary endpoint of this study was corticosteroid-free clinical remission (CFREM) at week 26, and one of the secondary endpoints was complete healing of mucosal ulcerations.
The investigators evaluated different cutoff points of endoscopic responses for two methods used to score the severity of endoscopic lesions: SES-CD (Simple Endoscopic Score for CD) and CDEIS (CD Endoscopic Index of Severity).
Almost half of the patients achieved mucosal healing at the 26th week.
A decrease from baseline in the SES-CD of at least 50% at week 26 "appeared to be the best discriminative cutoff value" for predicting patients most likely to be in clinical remission (off steroids at week 50). This degree of SES-CD response was met by 112 (65%) patients, with 74% sensitivity and 48% specificity for predicting CFREM at week 50.
For the CDEIS values, the best cutoff was at least a 45% drop from baseline at week 26 for predicting patients most likely to achieve CFREM at week 50, with 75% sensitivity and 45% specificity.
Because of the "subtle" difference between the two measures (50% and 45% cutoffs), "we propose a definition of endoscopic response with a decrease in baseline in CDEIS of at least 50% at week 26 for both tests," the authors wrote. (For CDEIS, the 50% cutoff had a sensitivity of 73% and a specificity of 46%.)
"As such, this endoscopic endpoint could serve as a reliable predictor of the midterm clinical outcome of therapies," Dr. Ferrante and his coauthors concluded. A 50% reduction in endoscopic activity from baseline also has potential for use as an endpoint in studies evaluating pharmacotherapy or treatment strategies "to show healing capacity," they wrote.
These cutoff values should be validated in a prospective study, which should also look at correlations between the endoscopic response and "disease-modifying long-term outcomes," such as a sustained clinical response or surgery, they added.
In the study that evaluated the reliability of the UCEIS, 25 investigators from North America and Europe were randomized to assess 28 of 57 sigmoidoscopy videos of patients with UC, which included some duplicates to evaluate intraobserver reliability. Clinical details such as the number of stools per day were provided for only two of the videos. They were also trained.
The UCEIS provides a score of 0 to 8 based on the sum of three descriptors: vascular pattern (normal, patchy obliteration, or obliterated), bleeding (none, mucosal, luminal mild, luminal moderate, or severe), and erosions and ulcers (none, erosions, superficial ulcer, or deep ulcer). Almost 700 video evaluations were performed.
The results included a satisfactory level of intrainvestigator and interinvestigator reliability, indicating that the UCEIS is simple to use and also "reliably evaluates the overall endoscopic severity of UC and accounts for 88% of the variance between endoscopists," the authors concluded.
The study was the first step in the validation of the UCEIS and has "confirmed the reliability of the UCEIS, even if further validation is needed to establish thresholds for remission, the clinical relevance of different UCEIS scores, and responsiveness of the UCEIS to change in disease status," they noted.
The CD study was partly funded by Janssen Biotech; Dr. Ferrante was supported by the Belgian Society of Gastrointestinal Endoscopy and by Funds for Scientific Research, Belgium. The authors disclosed serving on advisory committees and review panels of, or serving as consultants and speakers for, multiple pharmaceutical manufacturers, as well as receiving grants or research support from the companies, including Janssen and Abbott. Several authors were employees of Janssen Biotech and Janssen Biologics, the manufacturer of infliximab. One author had no disclosures.
The UCEIS study was funded by Warner Chilcott, and one author was an employee of the company. The authors disclosed serving as consultants, advisers, and/or speakers for, and receiving research grants from, multiple pharmaceutical companies. Several authors had no disclosures.
The research field of measurements is fundamental for improving management strategies in inflammatory bowel diseases (IBDs) since the process can progress no faster than its key measured variables. In recent years, there has been a growing interest in mucosal healing as a goal of treatment in both CD and UC, since it seems to serve as a surrogate marker for long-term well-being and reduced complications (and this is our true ultimate goal of treatment).
However, the paucity of data on how to best measure mucosal healing limits our ability to treat patients to this target. Is mucosal healing best measured by macroscopic appearance of the mucosa? If so, how can we accurately quantify the degree of inflammation, and what cutoff scores are associated with our ultimate goal? Perhaps other markers of mucosal healing are superior, such as histology, calprotectin (which may be more sensitive for microscopic inflammation), or magnetic resonance (MR) enterography, which measures the entire thickness of the bowel wall and the entire length of the small bowel. Even after determining the best measurement tool and its cutoff values, change over time (i.e., responsiveness) and reliability must be determined before its implementation in treatment algorithms.
In the November issue of Gastroenterology, Ferrante et al. showed in the robust SONIC data set that a reduction of 50% in the CDEIS (CD Endoscopic Index of Severity) and the simplified SES-CD (Simple Endoscopic Score for CD), or mucosal healing by week 26, predicts to some extent 1-year clinical remission in CD. In the second study, Travis et al. elegantly report on the reasonable reliability and initial validity of the only validated endoscopic score in UC, the UCEIS (Ulcerative Colitis Endoscopic Index of Severity) score. Both studies present important data that advance our knowledge one step further toward the "treat to target" concept in IBD.
However, the two studies also highlight the shortcomings of our endoscopic definition of mucosal healing and their imperfect reliability, validity, and prediction power. Ferrante et al. should be applauded for reporting diagnostic utility statistics and not only P values in evaluating prediction power. Modest sensitivity/specificity of 74%-75%/45%-48% indicate that mucosal healing can be regarded as another important predictive variable in the decision-making process but not yet alone as the gold standard that mandates treatment escalation.
The studies facilitate significantly our evaluation of endoscopic mucosal healing, but as the authors of both studies rightly indicate, more studies are needed to further define our moving "target" before implementing it in IBD treatment algorithms.
Dr. Dan Turner is head of the pediatric gastroenterology and nutrition unit, Hebrew University of Jerusalem, Shaare Zedek Medical Center. He has no relevant conflicts of interest.
The research field of measurements is fundamental for improving management strategies in inflammatory bowel diseases (IBDs) since the process can progress no faster than its key measured variables. In recent years, there has been a growing interest in mucosal healing as a goal of treatment in both CD and UC, since it seems to serve as a surrogate marker for long-term well-being and reduced complications (and this is our true ultimate goal of treatment).
However, the paucity of data on how to best measure mucosal healing limits our ability to treat patients to this target. Is mucosal healing best measured by macroscopic appearance of the mucosa? If so, how can we accurately quantify the degree of inflammation, and what cutoff scores are associated with our ultimate goal? Perhaps other markers of mucosal healing are superior, such as histology, calprotectin (which may be more sensitive for microscopic inflammation), or magnetic resonance (MR) enterography, which measures the entire thickness of the bowel wall and the entire length of the small bowel. Even after determining the best measurement tool and its cutoff values, change over time (i.e., responsiveness) and reliability must be determined before its implementation in treatment algorithms.
In the November issue of Gastroenterology, Ferrante et al. showed in the robust SONIC data set that a reduction of 50% in the CDEIS (CD Endoscopic Index of Severity) and the simplified SES-CD (Simple Endoscopic Score for CD), or mucosal healing by week 26, predicts to some extent 1-year clinical remission in CD. In the second study, Travis et al. elegantly report on the reasonable reliability and initial validity of the only validated endoscopic score in UC, the UCEIS (Ulcerative Colitis Endoscopic Index of Severity) score. Both studies present important data that advance our knowledge one step further toward the "treat to target" concept in IBD.
However, the two studies also highlight the shortcomings of our endoscopic definition of mucosal healing and their imperfect reliability, validity, and prediction power. Ferrante et al. should be applauded for reporting diagnostic utility statistics and not only P values in evaluating prediction power. Modest sensitivity/specificity of 74%-75%/45%-48% indicate that mucosal healing can be regarded as another important predictive variable in the decision-making process but not yet alone as the gold standard that mandates treatment escalation.
The studies facilitate significantly our evaluation of endoscopic mucosal healing, but as the authors of both studies rightly indicate, more studies are needed to further define our moving "target" before implementing it in IBD treatment algorithms.
Dr. Dan Turner is head of the pediatric gastroenterology and nutrition unit, Hebrew University of Jerusalem, Shaare Zedek Medical Center. He has no relevant conflicts of interest.
The research field of measurements is fundamental for improving management strategies in inflammatory bowel diseases (IBDs) since the process can progress no faster than its key measured variables. In recent years, there has been a growing interest in mucosal healing as a goal of treatment in both CD and UC, since it seems to serve as a surrogate marker for long-term well-being and reduced complications (and this is our true ultimate goal of treatment).
However, the paucity of data on how to best measure mucosal healing limits our ability to treat patients to this target. Is mucosal healing best measured by macroscopic appearance of the mucosa? If so, how can we accurately quantify the degree of inflammation, and what cutoff scores are associated with our ultimate goal? Perhaps other markers of mucosal healing are superior, such as histology, calprotectin (which may be more sensitive for microscopic inflammation), or magnetic resonance (MR) enterography, which measures the entire thickness of the bowel wall and the entire length of the small bowel. Even after determining the best measurement tool and its cutoff values, change over time (i.e., responsiveness) and reliability must be determined before its implementation in treatment algorithms.
In the November issue of Gastroenterology, Ferrante et al. showed in the robust SONIC data set that a reduction of 50% in the CDEIS (CD Endoscopic Index of Severity) and the simplified SES-CD (Simple Endoscopic Score for CD), or mucosal healing by week 26, predicts to some extent 1-year clinical remission in CD. In the second study, Travis et al. elegantly report on the reasonable reliability and initial validity of the only validated endoscopic score in UC, the UCEIS (Ulcerative Colitis Endoscopic Index of Severity) score. Both studies present important data that advance our knowledge one step further toward the "treat to target" concept in IBD.
However, the two studies also highlight the shortcomings of our endoscopic definition of mucosal healing and their imperfect reliability, validity, and prediction power. Ferrante et al. should be applauded for reporting diagnostic utility statistics and not only P values in evaluating prediction power. Modest sensitivity/specificity of 74%-75%/45%-48% indicate that mucosal healing can be regarded as another important predictive variable in the decision-making process but not yet alone as the gold standard that mandates treatment escalation.
The studies facilitate significantly our evaluation of endoscopic mucosal healing, but as the authors of both studies rightly indicate, more studies are needed to further define our moving "target" before implementing it in IBD treatment algorithms.
Dr. Dan Turner is head of the pediatric gastroenterology and nutrition unit, Hebrew University of Jerusalem, Shaare Zedek Medical Center. He has no relevant conflicts of interest.
A reduction of at least 50% in either of two different Crohn’s disease endoscopic activity scores after 26 weeks of treatment predicted those patients likely to be off of corticosteroid therapy after about 1 year of treatment, with a sensitivity approaching 75%, reported Dr. Marc Ferrante and his coinvestigators.
The study provides evidence that this cutoff could be used as a reliable predictor of clinical response, but it needs to be studied further, according to the authors, who are members of the International Organization for the Study of Inflammatory Bowel Diseases. The study was published in the Novemberissue of Gastroenterology.
Another study, published in the same issue of Gastroenterology, evaluated the reliability of the Ulcerative Colitis Endoscopic Index of Severity (UCEIS), an instrument recently created to assess the endoscopic severity of UC. This study randomized 25 investigators to assess and score 28 endoscopic videos of patients with UC – without knowledge of clinical features in most cases. The results indicated that the UCEIS provides a "satisfactory" level of intrainvestigator and interinvestigator agreement and that it is "a reliable instrument for measuring the endoscopic disease activity of UC," according to Dr. Simon Travis, of the translational gastroenterology unit at John Radcliffe Hospital, Oxford, England, and his coinvestigators.
The version of the UCEIS that provides a score from 0 to 8, based on findings of vascular pattern, bleeding, and erosions/ulcers, is the "favored version" but needs to be validated further, they concluded (Gastroenterol. 2013 July 29 [doi: 10.1053/j.gastro.2013.07.024]).
In the Crohn’s disease (CD) study, which used data on 172 patients enrolled in SONIC (Study of Biologic and Immunomodulator Naive Patients in Crohn’s Disease), Dr. Ferrante, of the department of gastroenterology at University Hospitals Leuven, Belgium, and his colleagues identified a cutoff point in two endoscopic response scores after 26 weeks of treatment that was predictive of clinical response at 50 weeks.
At baseline, the patients had endoscopic lesions and CD Activity Index (CDAI) scores of 220-450 points (median, 277). Their median age was 34 years, and they had been diagnosed with CD for a median of 2.5 years. The patients were dependent on corticosteroids and were randomized to treatment with infliximab (Remicade) infusions and/or oral azathioprine, and were followed to 50 weeks. The primary endpoint of this study was corticosteroid-free clinical remission (CFREM) at week 26, and one of the secondary endpoints was complete healing of mucosal ulcerations.
The investigators evaluated different cutoff points of endoscopic responses for two methods used to score the severity of endoscopic lesions: SES-CD (Simple Endoscopic Score for CD) and CDEIS (CD Endoscopic Index of Severity).
Almost half of the patients achieved mucosal healing at the 26th week.
A decrease from baseline in the SES-CD of at least 50% at week 26 "appeared to be the best discriminative cutoff value" for predicting patients most likely to be in clinical remission (off steroids at week 50). This degree of SES-CD response was met by 112 (65%) patients, with 74% sensitivity and 48% specificity for predicting CFREM at week 50.
For the CDEIS values, the best cutoff was at least a 45% drop from baseline at week 26 for predicting patients most likely to achieve CFREM at week 50, with 75% sensitivity and 45% specificity.
Because of the "subtle" difference between the two measures (50% and 45% cutoffs), "we propose a definition of endoscopic response with a decrease in baseline in CDEIS of at least 50% at week 26 for both tests," the authors wrote. (For CDEIS, the 50% cutoff had a sensitivity of 73% and a specificity of 46%.)
"As such, this endoscopic endpoint could serve as a reliable predictor of the midterm clinical outcome of therapies," Dr. Ferrante and his coauthors concluded. A 50% reduction in endoscopic activity from baseline also has potential for use as an endpoint in studies evaluating pharmacotherapy or treatment strategies "to show healing capacity," they wrote.
These cutoff values should be validated in a prospective study, which should also look at correlations between the endoscopic response and "disease-modifying long-term outcomes," such as a sustained clinical response or surgery, they added.
In the study that evaluated the reliability of the UCEIS, 25 investigators from North America and Europe were randomized to assess 28 of 57 sigmoidoscopy videos of patients with UC, which included some duplicates to evaluate intraobserver reliability. Clinical details such as the number of stools per day were provided for only two of the videos. They were also trained.
The UCEIS provides a score of 0 to 8 based on the sum of three descriptors: vascular pattern (normal, patchy obliteration, or obliterated), bleeding (none, mucosal, luminal mild, luminal moderate, or severe), and erosions and ulcers (none, erosions, superficial ulcer, or deep ulcer). Almost 700 video evaluations were performed.
The results included a satisfactory level of intrainvestigator and interinvestigator reliability, indicating that the UCEIS is simple to use and also "reliably evaluates the overall endoscopic severity of UC and accounts for 88% of the variance between endoscopists," the authors concluded.
The study was the first step in the validation of the UCEIS and has "confirmed the reliability of the UCEIS, even if further validation is needed to establish thresholds for remission, the clinical relevance of different UCEIS scores, and responsiveness of the UCEIS to change in disease status," they noted.
The CD study was partly funded by Janssen Biotech; Dr. Ferrante was supported by the Belgian Society of Gastrointestinal Endoscopy and by Funds for Scientific Research, Belgium. The authors disclosed serving on advisory committees and review panels of, or serving as consultants and speakers for, multiple pharmaceutical manufacturers, as well as receiving grants or research support from the companies, including Janssen and Abbott. Several authors were employees of Janssen Biotech and Janssen Biologics, the manufacturer of infliximab. One author had no disclosures.
The UCEIS study was funded by Warner Chilcott, and one author was an employee of the company. The authors disclosed serving as consultants, advisers, and/or speakers for, and receiving research grants from, multiple pharmaceutical companies. Several authors had no disclosures.
A reduction of at least 50% in either of two different Crohn’s disease endoscopic activity scores after 26 weeks of treatment predicted those patients likely to be off of corticosteroid therapy after about 1 year of treatment, with a sensitivity approaching 75%, reported Dr. Marc Ferrante and his coinvestigators.
The study provides evidence that this cutoff could be used as a reliable predictor of clinical response, but it needs to be studied further, according to the authors, who are members of the International Organization for the Study of Inflammatory Bowel Diseases. The study was published in the Novemberissue of Gastroenterology.
Another study, published in the same issue of Gastroenterology, evaluated the reliability of the Ulcerative Colitis Endoscopic Index of Severity (UCEIS), an instrument recently created to assess the endoscopic severity of UC. This study randomized 25 investigators to assess and score 28 endoscopic videos of patients with UC – without knowledge of clinical features in most cases. The results indicated that the UCEIS provides a "satisfactory" level of intrainvestigator and interinvestigator agreement and that it is "a reliable instrument for measuring the endoscopic disease activity of UC," according to Dr. Simon Travis, of the translational gastroenterology unit at John Radcliffe Hospital, Oxford, England, and his coinvestigators.
The version of the UCEIS that provides a score from 0 to 8, based on findings of vascular pattern, bleeding, and erosions/ulcers, is the "favored version" but needs to be validated further, they concluded (Gastroenterol. 2013 July 29 [doi: 10.1053/j.gastro.2013.07.024]).
In the Crohn’s disease (CD) study, which used data on 172 patients enrolled in SONIC (Study of Biologic and Immunomodulator Naive Patients in Crohn’s Disease), Dr. Ferrante, of the department of gastroenterology at University Hospitals Leuven, Belgium, and his colleagues identified a cutoff point in two endoscopic response scores after 26 weeks of treatment that was predictive of clinical response at 50 weeks.
At baseline, the patients had endoscopic lesions and CD Activity Index (CDAI) scores of 220-450 points (median, 277). Their median age was 34 years, and they had been diagnosed with CD for a median of 2.5 years. The patients were dependent on corticosteroids and were randomized to treatment with infliximab (Remicade) infusions and/or oral azathioprine, and were followed to 50 weeks. The primary endpoint of this study was corticosteroid-free clinical remission (CFREM) at week 26, and one of the secondary endpoints was complete healing of mucosal ulcerations.
The investigators evaluated different cutoff points of endoscopic responses for two methods used to score the severity of endoscopic lesions: SES-CD (Simple Endoscopic Score for CD) and CDEIS (CD Endoscopic Index of Severity).
Almost half of the patients achieved mucosal healing at the 26th week.
A decrease from baseline in the SES-CD of at least 50% at week 26 "appeared to be the best discriminative cutoff value" for predicting patients most likely to be in clinical remission (off steroids at week 50). This degree of SES-CD response was met by 112 (65%) patients, with 74% sensitivity and 48% specificity for predicting CFREM at week 50.
For the CDEIS values, the best cutoff was at least a 45% drop from baseline at week 26 for predicting patients most likely to achieve CFREM at week 50, with 75% sensitivity and 45% specificity.
Because of the "subtle" difference between the two measures (50% and 45% cutoffs), "we propose a definition of endoscopic response with a decrease in baseline in CDEIS of at least 50% at week 26 for both tests," the authors wrote. (For CDEIS, the 50% cutoff had a sensitivity of 73% and a specificity of 46%.)
"As such, this endoscopic endpoint could serve as a reliable predictor of the midterm clinical outcome of therapies," Dr. Ferrante and his coauthors concluded. A 50% reduction in endoscopic activity from baseline also has potential for use as an endpoint in studies evaluating pharmacotherapy or treatment strategies "to show healing capacity," they wrote.
These cutoff values should be validated in a prospective study, which should also look at correlations between the endoscopic response and "disease-modifying long-term outcomes," such as a sustained clinical response or surgery, they added.
In the study that evaluated the reliability of the UCEIS, 25 investigators from North America and Europe were randomized to assess 28 of 57 sigmoidoscopy videos of patients with UC, which included some duplicates to evaluate intraobserver reliability. Clinical details such as the number of stools per day were provided for only two of the videos. They were also trained.
The UCEIS provides a score of 0 to 8 based on the sum of three descriptors: vascular pattern (normal, patchy obliteration, or obliterated), bleeding (none, mucosal, luminal mild, luminal moderate, or severe), and erosions and ulcers (none, erosions, superficial ulcer, or deep ulcer). Almost 700 video evaluations were performed.
The results included a satisfactory level of intrainvestigator and interinvestigator reliability, indicating that the UCEIS is simple to use and also "reliably evaluates the overall endoscopic severity of UC and accounts for 88% of the variance between endoscopists," the authors concluded.
The study was the first step in the validation of the UCEIS and has "confirmed the reliability of the UCEIS, even if further validation is needed to establish thresholds for remission, the clinical relevance of different UCEIS scores, and responsiveness of the UCEIS to change in disease status," they noted.
The CD study was partly funded by Janssen Biotech; Dr. Ferrante was supported by the Belgian Society of Gastrointestinal Endoscopy and by Funds for Scientific Research, Belgium. The authors disclosed serving on advisory committees and review panels of, or serving as consultants and speakers for, multiple pharmaceutical manufacturers, as well as receiving grants or research support from the companies, including Janssen and Abbott. Several authors were employees of Janssen Biotech and Janssen Biologics, the manufacturer of infliximab. One author had no disclosures.
The UCEIS study was funded by Warner Chilcott, and one author was an employee of the company. The authors disclosed serving as consultants, advisers, and/or speakers for, and receiving research grants from, multiple pharmaceutical companies. Several authors had no disclosures.
FROM GASTROENTEROLOGY
Common analgesics linked to flares of Crohn’s disease
SAN DIEGO – Commonly used pain medications increase the risk of exacerbations of Crohn’s disease, judging from the findings of a prospective cohort study of nearly 800 patients with inflammatory bowel disease initially in remission.
Patients with Crohn’s disease who used nonsteroidal anti-inflammatory drugs (NSAIDs) at least five times monthly were 65% more likely to have active disease 6 months later, investigators reported at the annual meeting of the American College of Gastroenterology. Unexpectedly, any use of acetaminophen was also an independent risk factor in this group.
"A lot of our patients in remission are taking these medications. We need to ask detailed histories about this," she added. It is noteworthy that no such effect was seen in ulcerative colitis patients. "There might be a dose/response issue here. We have a lot of young women obviously with inflammatory bowel disease who take ibuprofen for menstrual cramping." When taken fewer than five times monthly, these drugs do not seem to have an effect, she noted.
"Implications may be that the requirement for any pain medication while in remission may actually be a marker of occult ongoing disease. Occult disease itself may be the risk factor for disease activity," commented lead investigator Dr. Millie Long, an assistant professor at the University of North Carolina, Chapel Hill.
"It’s also theoretically possible that a mechanism common to both drugs may be associated with increased disease activity," she added. For example, both NSAIDs and acetaminophen inhibit cyclooxygenase-3.
"Further prospective studies are needed with objective inflammatory outcomes to better assess the risks of these pain medications and better counsel patients," Dr. Long maintained.
One audience member noted that the study could not tease apart whether analgesic use was the cause or the result of an exacerbation.
"I would argue that we specifically limited this to a population of patients in remission by validated disease activity indices at baseline. ... We also had data on the quality of life of these individuals, the Shortened Inflammatory Bowel Disease questionnaire. These were individuals who felt well," Dr. Long replied. "That said, they could have been taking their pain medication for gut pain, I agree. They could have been taking it for arthritis, they could have been taking it for a headache."
Session comoderator Dr. Miguel D. Regueiro, a gastroenterologist at the University of Pittsburgh, asked, "In your practice, will you allow Crohn’s patients to take an NSAID three or four times a month?"
"I do, particularly for my young women who really need it for menstrual cramping. I don’t think that one time a week – and obviously these data support that – is going to cause a problem," Dr. Long replied. "I do feel these data as well as the Bonner prospective cohort data [Inflamm. Bowel Dis. 2004;10:751-57] show that high-dose NSAIDs may be bad news in patients with Crohn’s disease."
The investigators conducted the study among Crohn’s and Colitis Foundation of America (CCFA) Partners, an Internet-based cohort of patients with inflammatory bowel disease. The patients completed online surveys at baseline and 6 months, answering questions about disease activity and average number of times per month they used various pain medications.
Analyses were based on 791 patients who were in remission at baseline. About 75% had Crohn’s disease, reported Dr. Long. The mean duration of inflammatory bowel disease was approximately 14 years.
Overall, 43% of patients reported using any NSAIDs, and 19% reported using them at least five times monthly. Additionally, 65% reported any use of acetaminophen.
At follow-up, about a fifth of patients overall had experienced an exacerbation and now had active disease, defined as a simplified Crohn’s Disease Activity Index (sCDAI) score of at least 150 or a Simple Clinical Colitis Activity Index (SCCAI) score of greater than 2.
In the cohort as a whole, the proportion of patients with an exacerbation did not vary significantly according NSAID use, but it was higher among acetaminophen users versus nonusers (20% vs. 14%; P = .03).
Analyses stratified by type of disease showed that patients with Crohn’s disease were more likely to have an exacerbation if they used NSAIDs at least five times monthly versus less often, and if they used acetaminophen at all. In contrast, patients with ulcerative colitis were not affected.
In multivariate analyses that controlled for potential confounders, including use of other medications, NSAID use five or more times monthly was an independent risk factor for exacerbation in the entire cohort (relative risk, 1.46) and in the subset with Crohn’s disease (RR, 1.65).
Any use of acetaminophen was also an independent risk factor, both in the entire cohort (RR, 1.41) and in the subset with Crohn’s disease (RR, 1.72).
Dr. Long disclosed no relevant conflicts of interest.
SAN DIEGO – Commonly used pain medications increase the risk of exacerbations of Crohn’s disease, judging from the findings of a prospective cohort study of nearly 800 patients with inflammatory bowel disease initially in remission.
Patients with Crohn’s disease who used nonsteroidal anti-inflammatory drugs (NSAIDs) at least five times monthly were 65% more likely to have active disease 6 months later, investigators reported at the annual meeting of the American College of Gastroenterology. Unexpectedly, any use of acetaminophen was also an independent risk factor in this group.
"A lot of our patients in remission are taking these medications. We need to ask detailed histories about this," she added. It is noteworthy that no such effect was seen in ulcerative colitis patients. "There might be a dose/response issue here. We have a lot of young women obviously with inflammatory bowel disease who take ibuprofen for menstrual cramping." When taken fewer than five times monthly, these drugs do not seem to have an effect, she noted.
"Implications may be that the requirement for any pain medication while in remission may actually be a marker of occult ongoing disease. Occult disease itself may be the risk factor for disease activity," commented lead investigator Dr. Millie Long, an assistant professor at the University of North Carolina, Chapel Hill.
"It’s also theoretically possible that a mechanism common to both drugs may be associated with increased disease activity," she added. For example, both NSAIDs and acetaminophen inhibit cyclooxygenase-3.
"Further prospective studies are needed with objective inflammatory outcomes to better assess the risks of these pain medications and better counsel patients," Dr. Long maintained.
One audience member noted that the study could not tease apart whether analgesic use was the cause or the result of an exacerbation.
"I would argue that we specifically limited this to a population of patients in remission by validated disease activity indices at baseline. ... We also had data on the quality of life of these individuals, the Shortened Inflammatory Bowel Disease questionnaire. These were individuals who felt well," Dr. Long replied. "That said, they could have been taking their pain medication for gut pain, I agree. They could have been taking it for arthritis, they could have been taking it for a headache."
Session comoderator Dr. Miguel D. Regueiro, a gastroenterologist at the University of Pittsburgh, asked, "In your practice, will you allow Crohn’s patients to take an NSAID three or four times a month?"
"I do, particularly for my young women who really need it for menstrual cramping. I don’t think that one time a week – and obviously these data support that – is going to cause a problem," Dr. Long replied. "I do feel these data as well as the Bonner prospective cohort data [Inflamm. Bowel Dis. 2004;10:751-57] show that high-dose NSAIDs may be bad news in patients with Crohn’s disease."
The investigators conducted the study among Crohn’s and Colitis Foundation of America (CCFA) Partners, an Internet-based cohort of patients with inflammatory bowel disease. The patients completed online surveys at baseline and 6 months, answering questions about disease activity and average number of times per month they used various pain medications.
Analyses were based on 791 patients who were in remission at baseline. About 75% had Crohn’s disease, reported Dr. Long. The mean duration of inflammatory bowel disease was approximately 14 years.
Overall, 43% of patients reported using any NSAIDs, and 19% reported using them at least five times monthly. Additionally, 65% reported any use of acetaminophen.
At follow-up, about a fifth of patients overall had experienced an exacerbation and now had active disease, defined as a simplified Crohn’s Disease Activity Index (sCDAI) score of at least 150 or a Simple Clinical Colitis Activity Index (SCCAI) score of greater than 2.
In the cohort as a whole, the proportion of patients with an exacerbation did not vary significantly according NSAID use, but it was higher among acetaminophen users versus nonusers (20% vs. 14%; P = .03).
Analyses stratified by type of disease showed that patients with Crohn’s disease were more likely to have an exacerbation if they used NSAIDs at least five times monthly versus less often, and if they used acetaminophen at all. In contrast, patients with ulcerative colitis were not affected.
In multivariate analyses that controlled for potential confounders, including use of other medications, NSAID use five or more times monthly was an independent risk factor for exacerbation in the entire cohort (relative risk, 1.46) and in the subset with Crohn’s disease (RR, 1.65).
Any use of acetaminophen was also an independent risk factor, both in the entire cohort (RR, 1.41) and in the subset with Crohn’s disease (RR, 1.72).
Dr. Long disclosed no relevant conflicts of interest.
SAN DIEGO – Commonly used pain medications increase the risk of exacerbations of Crohn’s disease, judging from the findings of a prospective cohort study of nearly 800 patients with inflammatory bowel disease initially in remission.
Patients with Crohn’s disease who used nonsteroidal anti-inflammatory drugs (NSAIDs) at least five times monthly were 65% more likely to have active disease 6 months later, investigators reported at the annual meeting of the American College of Gastroenterology. Unexpectedly, any use of acetaminophen was also an independent risk factor in this group.
"A lot of our patients in remission are taking these medications. We need to ask detailed histories about this," she added. It is noteworthy that no such effect was seen in ulcerative colitis patients. "There might be a dose/response issue here. We have a lot of young women obviously with inflammatory bowel disease who take ibuprofen for menstrual cramping." When taken fewer than five times monthly, these drugs do not seem to have an effect, she noted.
"Implications may be that the requirement for any pain medication while in remission may actually be a marker of occult ongoing disease. Occult disease itself may be the risk factor for disease activity," commented lead investigator Dr. Millie Long, an assistant professor at the University of North Carolina, Chapel Hill.
"It’s also theoretically possible that a mechanism common to both drugs may be associated with increased disease activity," she added. For example, both NSAIDs and acetaminophen inhibit cyclooxygenase-3.
"Further prospective studies are needed with objective inflammatory outcomes to better assess the risks of these pain medications and better counsel patients," Dr. Long maintained.
One audience member noted that the study could not tease apart whether analgesic use was the cause or the result of an exacerbation.
"I would argue that we specifically limited this to a population of patients in remission by validated disease activity indices at baseline. ... We also had data on the quality of life of these individuals, the Shortened Inflammatory Bowel Disease questionnaire. These were individuals who felt well," Dr. Long replied. "That said, they could have been taking their pain medication for gut pain, I agree. They could have been taking it for arthritis, they could have been taking it for a headache."
Session comoderator Dr. Miguel D. Regueiro, a gastroenterologist at the University of Pittsburgh, asked, "In your practice, will you allow Crohn’s patients to take an NSAID three or four times a month?"
"I do, particularly for my young women who really need it for menstrual cramping. I don’t think that one time a week – and obviously these data support that – is going to cause a problem," Dr. Long replied. "I do feel these data as well as the Bonner prospective cohort data [Inflamm. Bowel Dis. 2004;10:751-57] show that high-dose NSAIDs may be bad news in patients with Crohn’s disease."
The investigators conducted the study among Crohn’s and Colitis Foundation of America (CCFA) Partners, an Internet-based cohort of patients with inflammatory bowel disease. The patients completed online surveys at baseline and 6 months, answering questions about disease activity and average number of times per month they used various pain medications.
Analyses were based on 791 patients who were in remission at baseline. About 75% had Crohn’s disease, reported Dr. Long. The mean duration of inflammatory bowel disease was approximately 14 years.
Overall, 43% of patients reported using any NSAIDs, and 19% reported using them at least five times monthly. Additionally, 65% reported any use of acetaminophen.
At follow-up, about a fifth of patients overall had experienced an exacerbation and now had active disease, defined as a simplified Crohn’s Disease Activity Index (sCDAI) score of at least 150 or a Simple Clinical Colitis Activity Index (SCCAI) score of greater than 2.
In the cohort as a whole, the proportion of patients with an exacerbation did not vary significantly according NSAID use, but it was higher among acetaminophen users versus nonusers (20% vs. 14%; P = .03).
Analyses stratified by type of disease showed that patients with Crohn’s disease were more likely to have an exacerbation if they used NSAIDs at least five times monthly versus less often, and if they used acetaminophen at all. In contrast, patients with ulcerative colitis were not affected.
In multivariate analyses that controlled for potential confounders, including use of other medications, NSAID use five or more times monthly was an independent risk factor for exacerbation in the entire cohort (relative risk, 1.46) and in the subset with Crohn’s disease (RR, 1.65).
Any use of acetaminophen was also an independent risk factor, both in the entire cohort (RR, 1.41) and in the subset with Crohn’s disease (RR, 1.72).
Dr. Long disclosed no relevant conflicts of interest.
AT THE ACG ANNUAL MEETING
Major finding: Patients with Crohn’s disease were 65% more likely to have an exacerbation if they used NSAIDs five or more times per month and 72% more likely if they used acetaminophen at all.
Data source: A prospective cohort study of 791 patients with inflammatory bowel disease who were in remission at baseline.
Disclosures: Dr. Long disclosed no relevant conflicts of interest.
AGA’s vice president highlights IBS patient care
Functional GI and motility disorders are arguably one of the most relevant groups of disorders bringing patients to gastroenterologists. This area offers potential to reinvigorate the role of GIs; however, progress in this field has been difficult, and the speed bumps have included focus on symptom-based diagnosis rather than understanding of patho-physiological mechanisms, excessive regulatory focus on patient response outcomes, and lack of funding.
Michael Camilleri, M.D., AGAF, vice president of the AGA Institute from Mayo Clinic, has been at the forefront of applied, patient-oriented gastroenterology research over the past 25 years. Thanks to funding he received from the AGA Research Foundation at critical points in his career, Dr. Camilleri has developed validated measurements that shape how clinicians diagnose and care for patients with gastroparesis, dyspepsia, IBS, chronic constipation, and diarrhea. His research has also helped to develop the drugs that are used widely in practice today.
These findings have brought on a renaissance in IBS patient care; however, in order to continue to move forward, we must understand the interrelationships of the diverse traits of these functional GI diseases. This understanding is essential if we are to have an impact on the diseases that we are trying to cure sometimes, to relieve often and to comfort always.
Dr. Camilleri discusses this renaissance in gastroenterology in a new video for the AGA Research Foundation. To watch the video, visit www.gastro.org/foundation.
Functional GI and motility disorders are arguably one of the most relevant groups of disorders bringing patients to gastroenterologists. This area offers potential to reinvigorate the role of GIs; however, progress in this field has been difficult, and the speed bumps have included focus on symptom-based diagnosis rather than understanding of patho-physiological mechanisms, excessive regulatory focus on patient response outcomes, and lack of funding.
Michael Camilleri, M.D., AGAF, vice president of the AGA Institute from Mayo Clinic, has been at the forefront of applied, patient-oriented gastroenterology research over the past 25 years. Thanks to funding he received from the AGA Research Foundation at critical points in his career, Dr. Camilleri has developed validated measurements that shape how clinicians diagnose and care for patients with gastroparesis, dyspepsia, IBS, chronic constipation, and diarrhea. His research has also helped to develop the drugs that are used widely in practice today.
These findings have brought on a renaissance in IBS patient care; however, in order to continue to move forward, we must understand the interrelationships of the diverse traits of these functional GI diseases. This understanding is essential if we are to have an impact on the diseases that we are trying to cure sometimes, to relieve often and to comfort always.
Dr. Camilleri discusses this renaissance in gastroenterology in a new video for the AGA Research Foundation. To watch the video, visit www.gastro.org/foundation.
Functional GI and motility disorders are arguably one of the most relevant groups of disorders bringing patients to gastroenterologists. This area offers potential to reinvigorate the role of GIs; however, progress in this field has been difficult, and the speed bumps have included focus on symptom-based diagnosis rather than understanding of patho-physiological mechanisms, excessive regulatory focus on patient response outcomes, and lack of funding.
Michael Camilleri, M.D., AGAF, vice president of the AGA Institute from Mayo Clinic, has been at the forefront of applied, patient-oriented gastroenterology research over the past 25 years. Thanks to funding he received from the AGA Research Foundation at critical points in his career, Dr. Camilleri has developed validated measurements that shape how clinicians diagnose and care for patients with gastroparesis, dyspepsia, IBS, chronic constipation, and diarrhea. His research has also helped to develop the drugs that are used widely in practice today.
These findings have brought on a renaissance in IBS patient care; however, in order to continue to move forward, we must understand the interrelationships of the diverse traits of these functional GI diseases. This understanding is essential if we are to have an impact on the diseases that we are trying to cure sometimes, to relieve often and to comfort always.
Dr. Camilleri discusses this renaissance in gastroenterology in a new video for the AGA Research Foundation. To watch the video, visit www.gastro.org/foundation.