User login
Hospitals See Spike in Palliative Care Teams
The number of hospital palliative care teams has soared over the past decade, including a 138% increase at institutions with more than 50 beds, according to the Center to Advance Palliative Care (CAPC).
The center looked at data from the American Hospital Association’s Annual Survey and the National Palliative Care Registry from 2000 to 2009. Of the 2,489 hospitals studied, 1,568 (63%) reported having a palliative care team.
Uptake was greater at large hospitals. Among the 699 hospitals with more then 300 beds, 85% had teams, compared with 54% for those with 50-299 beds.
Among the 1,500 hospitals with fewer than 50 beds, only 22% reported having a team, but these smaller institutions were excluded from further analysis.
The rate was lowest in the South, with only 51% of hospitals reporting a team, compared with 73% in the Northeast. In addition, 72% of hospitals in the Midwest, and 68% of hospitals in the West said they had a team.
Palliative care has been a topic of controversy since the implementation of the Affordable Care Act, which contains a provision in Medicare that would reimburse doctors for counseling elderly patients on end-of-life issues and palliative care. Opponents to the provision argued that it would encourage doctors to withhold care from elderly patients.
At a policy summit in March, health experts voiced support for increasing palliative care, arguing that it improved the quality of life for elderly patients.
"The quality of a patient’s life goes up. The quality of the family’s life goes up. More patients die at home, which is where 80% of Americans say they’d like to die," said Dr. Allen S. Lichter, a panelist at the summit. "Now, when you do the cost analysis, it happens to save money. That’s the sprinkles on the icing on the top of the cake – that’s not the cake."
Dr. Lichter, chief executive officer of the American Society of Clinical Oncology,* added that physicians should focus on making patients comfortable in their last days, instead of offering "false hope for a cure."
"Palliative care teams are transforming the care of serious illness in this country because they address the fragmentation of the health care system and put control and choice back in the hands of the patient and family," said CAPC director Dr. Diane E. Meier in a statement. "Hospitals today recognize that palliative care is the key to delivering better quality, coordinated care to our sickest and most vulnerable patients."
Regardless of the controversy among the experts, a CAPC poll showed that 70% of Americans don’t know what palliative care is. The center conducted the poll through a combination of one-on-one interviews with caregivers, Internet focus groups, and a telephone survey of 800 adults aged 25 years and older. Currently, 90 million Americans are living with chronic conditions and that number will only increase as the baby boomers age, CAPC said.
*Correction, 8/18/2011: An earlier version of this story incorrectly identified Dr. Lichter's affiliation.
The number of hospital palliative care teams has soared over the past decade, including a 138% increase at institutions with more than 50 beds, according to the Center to Advance Palliative Care (CAPC).
The center looked at data from the American Hospital Association’s Annual Survey and the National Palliative Care Registry from 2000 to 2009. Of the 2,489 hospitals studied, 1,568 (63%) reported having a palliative care team.
Uptake was greater at large hospitals. Among the 699 hospitals with more then 300 beds, 85% had teams, compared with 54% for those with 50-299 beds.
Among the 1,500 hospitals with fewer than 50 beds, only 22% reported having a team, but these smaller institutions were excluded from further analysis.
The rate was lowest in the South, with only 51% of hospitals reporting a team, compared with 73% in the Northeast. In addition, 72% of hospitals in the Midwest, and 68% of hospitals in the West said they had a team.
Palliative care has been a topic of controversy since the implementation of the Affordable Care Act, which contains a provision in Medicare that would reimburse doctors for counseling elderly patients on end-of-life issues and palliative care. Opponents to the provision argued that it would encourage doctors to withhold care from elderly patients.
At a policy summit in March, health experts voiced support for increasing palliative care, arguing that it improved the quality of life for elderly patients.
"The quality of a patient’s life goes up. The quality of the family’s life goes up. More patients die at home, which is where 80% of Americans say they’d like to die," said Dr. Allen S. Lichter, a panelist at the summit. "Now, when you do the cost analysis, it happens to save money. That’s the sprinkles on the icing on the top of the cake – that’s not the cake."
Dr. Lichter, chief executive officer of the American Society of Clinical Oncology,* added that physicians should focus on making patients comfortable in their last days, instead of offering "false hope for a cure."
"Palliative care teams are transforming the care of serious illness in this country because they address the fragmentation of the health care system and put control and choice back in the hands of the patient and family," said CAPC director Dr. Diane E. Meier in a statement. "Hospitals today recognize that palliative care is the key to delivering better quality, coordinated care to our sickest and most vulnerable patients."
Regardless of the controversy among the experts, a CAPC poll showed that 70% of Americans don’t know what palliative care is. The center conducted the poll through a combination of one-on-one interviews with caregivers, Internet focus groups, and a telephone survey of 800 adults aged 25 years and older. Currently, 90 million Americans are living with chronic conditions and that number will only increase as the baby boomers age, CAPC said.
*Correction, 8/18/2011: An earlier version of this story incorrectly identified Dr. Lichter's affiliation.
The number of hospital palliative care teams has soared over the past decade, including a 138% increase at institutions with more than 50 beds, according to the Center to Advance Palliative Care (CAPC).
The center looked at data from the American Hospital Association’s Annual Survey and the National Palliative Care Registry from 2000 to 2009. Of the 2,489 hospitals studied, 1,568 (63%) reported having a palliative care team.
Uptake was greater at large hospitals. Among the 699 hospitals with more then 300 beds, 85% had teams, compared with 54% for those with 50-299 beds.
Among the 1,500 hospitals with fewer than 50 beds, only 22% reported having a team, but these smaller institutions were excluded from further analysis.
The rate was lowest in the South, with only 51% of hospitals reporting a team, compared with 73% in the Northeast. In addition, 72% of hospitals in the Midwest, and 68% of hospitals in the West said they had a team.
Palliative care has been a topic of controversy since the implementation of the Affordable Care Act, which contains a provision in Medicare that would reimburse doctors for counseling elderly patients on end-of-life issues and palliative care. Opponents to the provision argued that it would encourage doctors to withhold care from elderly patients.
At a policy summit in March, health experts voiced support for increasing palliative care, arguing that it improved the quality of life for elderly patients.
"The quality of a patient’s life goes up. The quality of the family’s life goes up. More patients die at home, which is where 80% of Americans say they’d like to die," said Dr. Allen S. Lichter, a panelist at the summit. "Now, when you do the cost analysis, it happens to save money. That’s the sprinkles on the icing on the top of the cake – that’s not the cake."
Dr. Lichter, chief executive officer of the American Society of Clinical Oncology,* added that physicians should focus on making patients comfortable in their last days, instead of offering "false hope for a cure."
"Palliative care teams are transforming the care of serious illness in this country because they address the fragmentation of the health care system and put control and choice back in the hands of the patient and family," said CAPC director Dr. Diane E. Meier in a statement. "Hospitals today recognize that palliative care is the key to delivering better quality, coordinated care to our sickest and most vulnerable patients."
Regardless of the controversy among the experts, a CAPC poll showed that 70% of Americans don’t know what palliative care is. The center conducted the poll through a combination of one-on-one interviews with caregivers, Internet focus groups, and a telephone survey of 800 adults aged 25 years and older. Currently, 90 million Americans are living with chronic conditions and that number will only increase as the baby boomers age, CAPC said.
*Correction, 8/18/2011: An earlier version of this story incorrectly identified Dr. Lichter's affiliation.
FROM THE CENTER TO ADVANCE PALLIATIVE CARE
Major Finding: 63% of hospitals in the U.S. with more then 50 beds have a palliative care team, a 138.3% increase since 2000.
Data Source: Data analysis of the American Hospital Association’s Annual Survey Database and the National Palliative Care Registry for 2000-2009
Disclosures: The authors reported no relevant financial disclosures.
Managing cancer pain: Frequently asked questions
Some 90% of patients with cancer experience pain during their illness.1 The pain usually worsens as the disease progresses, and patients may experience different types of pain.
Persistent pain decreases function, appetite, and sleep, induces fear, causes depression, and generally lowers the quality of life.2 Persistent pain is demoralizing and debilitating for patients and their caregivers.3
Adequate pain control is important to ensure that patients can function productively, maintain social relationships, and improve their quality of life.2 Yet 86% of practicing physicians surveyed believed that most cancer patients with pain were undermedicated,2 and most felt that pain management is unsuccessful in more than half of patients who seek help.3
The critical importance of pain management has been emphasized by the World Health Organization (WHO), by international and national professional organizations, and by government agencies. All practitioners who care for cancer patients need to be well educated in managing cancer pain, a key part of which is to educate patients about the process and what to expect. This results in better pain control.4
Although much has been written on the management of cancer pain in a referral setting, little has been published on how to manage it in primary care. In this article, we discuss common questions faced by generalists. We emphasize the use of opioids, perhaps the most challenging aspect of cancer pain management. We also discuss when consultation with a specialist in pain management or a palliative medicine specialist is especially helpful.
WHAT ARE THE DIFFERENT TYPES OF PAIN SYNDROMES?
Pain is classified in several ways1–6:
Nociceptive vs neuropathic. Nociceptive pain comprises somatic and visceral components and is the result of continued tissue injury.4 Neuropathic pain is due to injury to the peripheral and central nervous systems and occurs within an area of sensory or motor deficit.
Continuous vs intermittent. Continuous pain, even if controlled, can have breakthroughs, ie, flares of pain above the controlled baseline level. Intermittent pain is a pain flare without chronic baseline pain. Intermittent pain is further divided into incident pain (ie, on movement) and end-of-dose failure (ie, pain occurring just before the next scheduled opioid dose).5 Pain specialists continue to debate the meaning and the use of these terms.
Malignant vs nonmalignant. Cancer pain is multifactorial,1 being induced by the disease itself, by the treatment of cancer, and by pain unrelated to cancer or its treatment (eg, osteoarthritis or diabetic neuropathy).2
Familiarity with the causes and the types of pain, including pain related to cancer, is important, as this influences treatment decisions.
HOW IS PAIN ASSESSED?
The assessment of pain is vital in managing it.
Since pain is inherently subjective, the patient’s self-report is the gold standard.4 Characteristics of the pain along with a physical examination, laboratory testing, and imaging studies can define the pathophysiology of the pain and influence the decision to undertake further assessment or specific therapies.
Patients and physicians can use various scales, such as a visual analog scale, a numerical rating scale, a graphic scale, a verbal scale, a word descriptor scale, and a functional pain scale. A verbal scale can be used if the patient is alert, or a nonverbal scale if the patient has impaired cognition or speaks a different language. Intensity is the most common dimension evaluated in cancer pain, primarily via a numerical or visual analog scale. A numerical scale score of 0 to 10 has been found to be as effective as a visual analog scale (0 to 100 mm),7,8 and the numerical rating scale is generally preferred as a measure of pain intensity.9
There are no clear guidelines for selecting one scale over another.7 A clinically meaningful response (ie, meaningful to patients) is at least a two-point decrease on the 10-point numerical scale or a 13-mm decrease on the 100-mm visual analog scale. A decrease in the percentage of the pain relates to global improvement better than an absolute reduction on the numerical scale.
WHAT PROBLEMS ARE ENCOUNTERED IN MANAGING CANCER PAIN?
WHAT ARE THE DIFFERENT WAYS TO MANAGE CANCER PAIN?
Pain should be treated promptly and aggressively, because if untreated it can lead to delays in healing, changes in the central nervous system (eg, sensitization, plasticity), chronic stress, family stress, depression, job loss, and even suicide.12–14
Comprehensive pain management improves outcomes and includes the rational use of opioids and adjuvant analgesics, physical rehabilitation, cognitive behavioral (non-drug) therapies, family counseling, interventional procedures (kyphoplasty, nerve blocks, local injections, spinal analgesia), and complementary therapies such as acupuncture.12 Adjuvant analgesics include antidepressants, anticonvulsants, and local anesthetics.
HOW DO OPIOIDS RELIEVE CANCER PAIN?
Opioids bind to receptors in tissues throughout the body, including in the central and peripheral nervous systems15 and the digestive tract. The binding of an opioid to an opioid receptor—including mu, kappa, and delta receptors and orphan receptor-like ligand-1—initiates a cascade of intracellular reactions. Due to the nature of different interactions of opioids with each of these receptors, individuals vary in their response to opioids.15
WHAT ARE THE CHARACTERISTICS OF COMMON OPIOIDS?
- Step 1. Mild pain calls for a nonopioid analgesic with or without an adjuvant (more about adjuvants below).
- Step 2. Mild or moderate pain that persists or increases calls for a weak opioid such as codeine, tramadol (Ultram), or hydrocodone, with or without a nonopioid and with or without an adjuvant.
- Step 3. Severe pain calls for a strong opioid with or without a nonopioid, and with or without an adjuvant.
Morphine, the prototypical opioid, is well studied and versatile, as it can be given orally, parenterally, rectally, or intraspinally. It is readily available in the United States and Western Europe but not in some parts of the world, such as Asia and Africa. It is also cost-effective.
Hydromorphone (Dilaudid) is similar to morphine in terms of versatility, cost, and effectiveness in pain management. An extended-release form (Exalgo) is now available in the United States.
Oxycodone is readily available in both slow-release (eg, OxyContin) and immediate-release (eg, Oxy-IR) preparations and is also cost-effective. However, there is no parenteral formulation in the United States.
Methadone is inexpensive and can be used as a long-acting or an immediate-release opioid. However, it should be used with caution in patients with a prolonged QTc interval: in general, a QTc interval of 430 to 450 msec is not a contraindication, but there is a risk of torsades de pointes when the QTc is greater than 500 msec. The physician should also look for drug interactions when prescribing methadone, which is metabolized in the liver via the cytochrome P450 3A4 system. Methadone use can also lead to respiratory depression, prolonged QTc interval, and sudden death.
Buprenorphine can be used as a third- or fourth-tier opioid for patients with both kidney and liver failure. It can be given sublingually or parenterally. It may not be readily available, may not be covered by insurance, and is expensive.
Selecting an opioid to try first
The following are some general considerations when selecting an opioid to try first:
- Does the patient have a history of organ failure? Has the patient had a therapeutic response to, or adverse effects from, a particular opioid in the past?
- Which route would best fit the patient’s needs? (Oral is always preferable.)
- How often will breakthrough dosing be required? (In general, the breakthrough dose is administered at the drug’s half-life, but it can be administered between 1 and 4 hours.)
- How much will it cost? (Consider the cost, insurance coverage, and co-pays.)
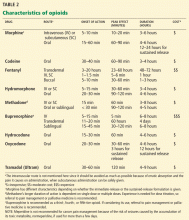
Table 2 shows different characteristics of commonly used opioids, including route of administration, onset of action, peak effect, and duration of action.1
WHAT ARE THE EQUIANALGESIC DOSES OF COMMONLY USED OPIOIDS?
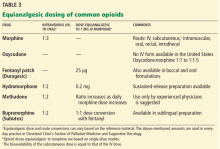
Table 3 lists equianalgesic doses and route conversions of commonly used opioids.18–20
WHAT ARE THE PRINCIPLES BEHIND OPIOID DOSING?
Table 4 shows important strategies for opioid dosing. An in-depth discussion of specific opioid dosing strategies is beyond the scope of this article.5
WHAT ARE THE COMMON ADVERSE EFFECTS OF OPIOIDS?
Adverse effects are among the most common reasons for failure of opioids to relieve pain. If these effects are not anticipated and treated prophylactically, patients may avoid taking their opioid drugs or may complain that they are “allergic” to them. In reality, true allergy to any of the opioids is rare. Patients comply better if they are taught to expect that most adverse effects are either preventable or manageable.21 A simple strategy includes reducing the opioid dose by 25% to 50%, using different opioids (“rotation”), changing the route of administration, and directly treating adverse effects.21,22
WHAT IS OPIOID ROTATION AND HOW IS IT DONE?
Opioid rotation involves changing to a different drug using the same administration route, with the aim of improving the analgesic response or reducing adverse effects.16 It may be useful in widening the therapeutic window, ie, establishing a more advantageous relationship between analgesia and toxicity.16 This strategy applies, for example, to patients who have an adverse reaction to morphine, and who may need rotation to fentanyl or methadone.
The major indication for switching opioids is poorly controlled pain with unacceptable adverse effects due to opioid toxicity, the rapid development of tolerance, refractory pain, or difficult pain syndromes.24 A recent prospective study showed that 42% of patients underwent opioid rotation, and the two most common reasons were inadequate analgesia and severe adverse effects.25 Opioid rotation resulted in relief of confusion (72%), nausea and vomiting (68%), and drowsiness (53%).25
Before trying opioid rotation, review the patient’s pain syndromes and the use of an adjuvant analgesic, and assess for evidence of opioid toxicity or contributing abnormal biochemical factors such as hydration status.24,26 Most opioids are mu-receptor agonists and may exhibit cross-tolerance, a phenomenon in which the alternative drug does not have the expected effects because of similar pharmacologic action of the first drug. Because the degree of cross-tolerance may change as opioid doses are escalated, it is advisable to proceed with caution when switching from one opioid to another in patients who are receiving very high doses. Opioid rotation generally would be ineffective if there is complete analgesic cross-tolerance between opioids.
The common equivalency conversion tables are based either on studies in patients who received low doses of opioids or on single-dose studies.16,24 By substituting opioids and using lower doses than expected according to the equivalency conversion tables (generally a 25% to 30% decrease), it is possible in most cases to reduce or relieve the symptoms of opioid toxicity and to manage patients highly tolerant to previous opioids while improving analgesia.24
Alternatives to opioid rotation are route conversion (oral to parenteral or spinal), addition of an adjuvant analgesic, and opioid dose reduction.
WHAT IS OPIOID TOXICITY AND HOW IS IT MANAGED?
Opioid overdose is commonly the result of an error in pain assessment, opioid prescribing, or dose administration. Opioid overdose classically presents as sedation or respiratory depression. The combination of coma, reduced respiratory rate, and pinpoint pupils is highly suggestive of opioid toxicity, and treatment should be initiated promptly.
This scenario, however, is the extreme example of opioid overdose, and it is rare when a patient is given the correct opioid dose titrated gradually over a period of time. The more common scenario is when a patient’s pain has finally been managed and the patient is resting comfortably with slow respirations. This would not warrant naloxone (Narcan) administration but rather close observation and monitoring of vital signs.
Naloxone has antagonist activity at all of the receptor sites.27 It is important to be alert for acute opioid withdrawal in patients taking high-dose opioids for a long time.27 There are no guidelines as to the route of administration and the dosing of naloxone. Table 6 summarizes the management of opioid overdose using naloxone.5
WHAT IS THE ROLE OF ADJUVANTS?
An adjuvant analgesic is any drug with a primary indication other than pain, but with analgesic properties in some painful conditions. Adjuvants are best used when a patient cannot obtain satisfactory pain relief from an opioid.28 Antidepressants, anticonvulsants, neuroleptics, antiarrhythmics, antihistamines, N-methyl-d-aspartate (NMDA) receptor antagonists, steroids, muscle relaxants, bisphosphonates, and radiopharmaceuticals can be adjuvant agents.29
Adjuvants are generally used to complement the analgesic effects of opioids to achieve optimal pain control with a minimum of adverse effects.28 The following scenarios should prompt the use of adjuvants in clinical practice28:
- The toxic limit of a primary pain medication has been reached.
- The therapeutic benefit of the primary pain medication has reached a plateau.
- The primary analgesic could not be used because of substance-abuse behavior, multiple organ failure, allergy, etc.
- The patient has multiple pain syndromes.
- The patient has additional symptoms unrelated to pain, eg, insomnia or depression.
Table 7 lists adjuvants with specific indications and points to remember when prescribing them.28,29
WHAT IS THE ROLE OF NSAIDs FOR CANCER PAIN?
Nonsteroidal anti-inflammatory drugs (NSAIDs) have a well-established role in treating cancer-related pain, either on their own for mild pain or in combination with opioids for moderate to severe pain, leading to additive analgesia. Using NSAIDs as adjuvants is common practice in certain cancer pain syndromes, such as malignant bone pain, although there is considerable variation in response.31
NSAIDs have long been known to inhibit peripheral prostaglandin synthesis, but recently they have also been suggested to have a central action. The central effect is related to NMDA receptor-induced activation of the nitric oxide system.31
NSAIDs have ceiling effects, and there is no therapeutic advantage to increasing the dose beyond that which is recommended.
Ketorolac (Toradol), indomethacin (Indocin), and diclofenac (Voltaren) have potent analgesic activity, whereas the “oxicam” NSAIDs show predominantly anti-inflammatory effects.30
No NSAID is clearly superior for a particular type of pain. Certain NSAIDs block the NMDA receptor and inhibit cyclo-oxygenase-1 and cyclo-oxygenase-2. There is a poor correlation between the analgesic effects of NSAIDs and cyclo-oxygenase inhibition. There is no evidence to support the use of selective cyclo-oxygenase-2 inhibitors for cancer pain, and these agents have no advantage over nonselective NSAIDs on the basis of limited gastrointestinal toxicity.32
In cancer pain, NSAIDs may delay the development of tolerance and allow lower doses of opioids to be used, with fewer central nervous system side effects.31,32 Despite the extensive use of NSAIDs, relatively few randomized studies have documented their efficacy in cancer pain compared with other chronic pain syndromes. Data on safe and effective doses from studies of nonmalignant pain may not apply to cancer pain, since cancer patients often have several serious conditions and are on multiple medications. In addition, the potential for adverse effects of NSAIDs (gastrointestinal bleeding, renal failure, thrombosis) may be greater in patients with advanced cancer.
In conclusion, NSAIDs may help if used judiciously in somatic pain and visceral pain, and perhaps even in neuropathic pain.31
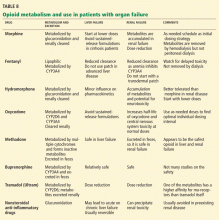
HOW IS CANCER PAIN MANAGED IN PATIENTS WITH ORGAN FAILURE?
- Opioids that can be used in liver failure or cirrhosis: morphine, hydromorphone, methadone, levorphanol, buprenorphine.
- Opioids that can be used in renal failure: methadone, fentanyl, and buprenorphine are safest; oxycodone and hydromorphone are moderately safe; morphine is the least safe.35,36
- Opioids that can be used in both kidney and liver failure: methadone, buprenorphine.
HOW CAN PROBLEMS RELATED TO SUBSTANCE ABUSE BE AVOIDED?
Substance abuse is less a problem in managing cancer pain than in chronic nonmalignant pain. Prescribing opioids safely is challenging, and very little has been published on substance abuse and the management of cancer pain. However, in the absence of practice guidelines, the best approach is to establish a dosing structure, control prescription refills, and monitor the patient.
Abuse is the misuse of an opioid via self-titration or altering the dosing schedule or route of administration. Patients who misuse opioids—ie, take them differently than prescribed—are not necessarily addicted.
Addiction is the abuse of a drug associated with psychological dependence, despite harm.
Diversion can occur without addiction and is done for financial gain, and this is the worst offense as it may harm others.
Pseudoaddiction is abnormal, demanding, often hostile behavior resulting from uncontrolled pain; once the pain is controlled, the behavior resolves.
Behaviors such as forging prescriptions, stealing or borrowing drugs, frequently “losing” prescriptions, and resisting changes to medication despite adverse effects are more predictive of addiction than are behaviors such as aggressive complaining about the need for more drugs, drug-hoarding, and unsanctioned dose escalations or other forms of noncompliance, as the latter three are more likely to indicate poorly controlled pain.37
Predictors of opioid abuse include a family history or a personal history of alcohol or drug abuse (including prescription drugs); a history of psychiatric illness (including anxiety disorder); male sex; nonwhite race; a history of driving under the influence of alcohol or drugs; a record of drug-related convictions; lost or stolen prescriptions; and using supplemental sources to obtain opioids.38 Socioeconomic status and disability level were not found to be significant predictors.38
Different scales are available to predict the risk of aberrant drug behavior in patients on chronic opioid therapy. Of the many available, the Screener and Opioid Assessment for Patients With Pain and the Current Opioid Misuse Measure assess all the key factors.38
After an assessment, the next step is monitoring. Unfortunately, no specific method has been validated. In one study, urine toxicology testing was more effective at identifying problems than monitoring patient behavior alone, and monitoring behavior alone would have resulted in missing about half of the patients with a problem.39 The same study showed that even in the absence of aberrant drug-related behavior based on predictors, a significant number of urine toxicology screens were positive.39
A negative urine screen for the patient’s opioid suggests diversion. The clinician should order a screen for the prescribed opioid because a general screen may not detect nonmorphine opioids. A general screen may detect polysubstance abuse, which is common in individuals with addiction.
Table 9 summarizes a strategy to manage opioid therapy in patients with history of substance abuse.40
WHAT IS THE ROLE OF COMPLEMENTARY AND ALTERNATIVE THERAPIES?
Complementary and alternative medicine therapies are commonly used by cancer patients, with an average prevalence rate of 31%.41–43 As the names suggest, they have been used both as an alternative to and as a complement to conventional medicine. Practitioners of complementary and alternative medicine emphasize its holistic, individualistic, empowering, and educational nature.
Patients do not routinely ask their physicians about these therapies,44 and physicians often have only a limited knowledge of them.45 Surveys of North American physicians showed that they view certain of these therapies as legitimate and effective.46,47
The role of complementary and alternative medicine in cancer pain has been the subject of debate, as relatively little is known about adverse effects and drug interactions. Nevertheless, the American Cancer Society and the National Comprehensive Cancer Network guidelines on cancer pain recommend nonpharmacologic treatment be added for patients who report a pain score of 4 or greater on a 10-point scale after analgesic adjustment.48,49
Most studies of complementary and alternative therapies for cancer pain are of poor quality, with significant shortcomings in methodology and study design and with no clear definition of outcomes.50
Acupuncture is probably the most studied of these therapies, but clinical trials so far have not shown it to be an effective adjunct analgesic for cancer pain.51 A placebo-controlled, blinded randomized trial using auricular acupuncture showed a pain score decrease of 36% from baseline at 2 months compared with controls.52
Studies involving cognitive therapy, supportive psychotherapy, and hypnosis showed modest benefit.53,54 Two trials involving relaxation and imagery reduced cancer pain compared with controls.55,56
Studies of massage therapy have shown mixed results; two studies reported a significant reduction in pain immediately after intervention, and no study found pain relief after 4 weeks.57–60 Studies involving Reiki and touch therapy were inconclusive.60,61
Music therapy has been used to treat patients physically, psychologically, socially, emotionally, and spiritually, with evidence still equivocal. A large prospective observational study involving 200 patients conducted by Gallagher et al62 showed pain was reduced by 30% after music therapy intervention. The same study showed a reduction in depression and anxiety.62 Music therapy could be used as a component of a multimodal approach to pain.
Herbal preparations are often used to treat cancer and symptoms by patients and naturalists. Some herbal medicines are known to cause toxicity in cancer patients. Examples are PC-SPES, mistletoe, and saw palmetto.63
At this juncture, there is some evidence that some complementary and alternative therapies can relieve cancer pain, and the most promising therapy seems to be related to mind-body medicine (eg, biofeedback, relaxation techniques). But before we can legitimately integrate these therapies into the management of cancer pain, we need large randomized controlled trials to determine if they are effective in patients on chronic high-dose opioids and if they decrease the need for opioids.
- Laird B, Colvin L, Fallon M. Management of cancer pain: basic principles and neuropathic cancer pain. Eur J Cancer 2008; 44:1078–1082.
- Chang HM. Cancer pain management. Med Clin North Am 1999; 83:711–736,
- Stannard C, Johnson M. Chronic pain management—can we do better? An interview-based survey in primary care. Curr Med Res Opin 2003; 19:703–706.
- Portenoy RK, Lesage P. Management of cancer pain. Lancet 1999; 353:1695–1700.
- Walsh D, Rivera NI, Davis MP, Lagman R, Legrand SB. Strategies for pain management: Cleveland Clinic Foundation guidelines for opioid dosing for cancer pain. Support Cancer Ther 2004; 1:157–164.
- Foley KM. Acute and chronic pain syndromes. In:Doyle D, Hanks G, Cherny N, Calman K, editors. Oxford Textbook of Palliative Medicine. 3rd ed. Oxford, UK: Oxford University Press; 2005:298–316.
- Jensen MP. The validity and reliability of pain measures in adults with cancer. J Pain 2003; 4:2–21.
- Jensen MP, Turner JA, Romano JM. What is the maximum number of levels needed in pain intensity measurement? Pain 1994; 58:387–392.
- Preston CC, Colman AM. Optimal number of response categories in rating scales: reliability, validity, discriminating power, and respondent p. Acta Psychol (Amst) 2000; 104:1–15.
- Peretti-Watel P, Bendiane MK, Obadia Y, Favre R, Lapiana JM, Moatti JP; South-Eastern France Palliative Care Group. The prescription of opioid analgesics to terminal cancer patients: impact of physicians’ general attitudes and contextual factors. Palliat Support Care 2003; 1:345–352.
- Jacobsen R, Liubarskiene Z, Møldrup C, Christrup L, Sjøgren P, Samsanaviciene J. Barriers to cancer pain management: a review of empirical research. Medicina (Kaunas) 2009; 45:427–433.
- Wiedemer NL, Harden PS, Arndt IO, Gallagher RM. The opioid renewal clinic: a primary care, managed approach to opioid therapy in chronic pain patients at risk for substance abuse. Pain Med 2007; 8:573–584.
- Rome HP, Rome JD. Limbically augmented pain syndrome (LAPS): kindling, corticolimbic sensitization, and the convergence of affective and sensory symptoms in chronic pain disorders. Pain Med 2000; 1:7–23.
- Fishbain DA, Rosomoff HL, Rosomoff RS. Drug abuse, dependence, and addiction in chronic pain patients. Clin J Pain 1992; 8:77–85.
- Murányi M, Radák Z. Pain and opioids. Orv Hetil 2008; 149:2363–2370.
- Vadalouca A, Moka E, Argyra E, Sikioti P, Siafaka I. Opioid rotation in patients with cancer: a review of the current literature. J Opioid Manag 2008; 4:213–250.
- Galvagno SM, Correll DJ, Narang S. Safe oral equianalgesic opioid dosing for patients with moderate-to-severe pain. www.hcplive.com/publications/Resident-and-Staff/2007/2007-04/2007-04_06. Accessed May 25, 2011.
- Walsh D. Pharmacological management of cancer pain. Semin Oncol 2000; 27:45–63.
- Shaheen PE, Walsh D, Lasheen W, Davis MP, Lagman RL. Opioid equianalgesic tables: are they all equally dangerous? J Pain Symptom Manage 2009; 38:409–417.
- Pereira J, Lawlor P, Vigano A, Dorgan M, Bruera E. Equianalgesic dose ratios for opioids. a critical review and proposals for long-term dosing. J Pain Symptom Manage 2001; 22:672–687.
- Harris JD. Management of expected and unexpected opioid-related side effects. Clin J Pain 2008; 24(suppl 10):S8–S13.
- Cherny N, Ripamonti C, Pereira J; Expert Working Group of the European Association of Palliative Care Network. Strategies to manage the adverse effects of oral morphine: an evidence-based report. J Clin Oncol 2001; 19:2542–2554.
- Harris JD, Kotob F. Management of opioid-related side effects. In:de Leon-Casasola OA, ed. Cancer Pain: Pharmacological, Interventional and Palliative Care. Philadelphia: Elsevier Inc; 2006:207–230.
- Mercadante S. Opioid rotation for cancer pain: rationale and clinical aspects. Cancer 1999; 86:1856–1866.
- Cheema B, Lagman RL, Walsh D, et al. A prospective study of opioid rotation in pain due to advanced cancer. J Cancer Pain & Symp Palliat 2006; 2:39–46.
- Schug SA, Zech D, Grond S, Jung H, Meuser T, Stobbe B. A long-term survey of morphine in cancer pain patients. J Pain Symptom Manage 1992; 7:259–266.
- Clarke SF, Dargan PI, Jones AL. Naloxone in opioid poisoning: walking the tightrope. Emerg Med J 2005; 22:612–616.
- Knotkova H, Pappagallo M. Adjuvant analgesics. Med Clin North Am 2007; 91:113–124.
- Lussier D, Huskey AG, Portenoy RK. Adjuvant analgesics in cancer pain management. Oncologist 2004; 9:571–591.
- Johnson JR, Burnell-Nugent M, Lossignol D, Ganae-Motan ED, Potts R, Fallon MT. Multicenter, double-blind, randomized, placebo-controlled, parallel-group study of the efficacy, safety, and tolerability of THC:CBD extract and THC extract in patients with intractable cancerrelated pain. J Pain Symptom Manage 2010; 39:167–179.
- Mercadante S. The use of anti-inflammatory drugs in cancer pain. Cancer Treat Rev 2001; 27:51–61.
- Davis MP, Walsh D, Lagman R, LeGrand SB. Controversies in pharmacotherapy of pain management. Lancet Oncol 2005; 6:696–704.
- Klotz U. Tramadol—the impact of its pharmacokinetic and pharmacodynamic properties on the clinical management of pain. Arzneimittelforschung 2003; 53:681–687.
- Davis MP, Lasheen W, Gamier P. Practical guide to opioids and their complications in managing cancer pain. What oncologists need to know. Oncology (Williston Park) 2007; 21:1229–1238.
- Dean M. Opioids in renal failure and dialysis patients. J Pain Symptom Manage 2004; 28:497–504.
- Davis MP. Buprenorphine in cancer pain. Support Care Cancer 2005; 13:878–887.
- Portenoy RK. Opioid therapy for chronic nonmalignant pain: a review of the critical issues. J Pain Symptom Manage 1996; 11:203–217.
- Turk DC, Swanson KS, Gatchel RJ. Predicting opioid misuse by chronic pain patients: a systematic review and literature synthesis. Clin J Pain 2008; 24:497–508.
- Katz NP, Sherburne S, Beach M, et al. Behavioral monitoring and urine toxicology testing in patients receiving long-term opioid therapy. Anesth Analg 2003; 97:1097–1102,
- Passik SD, Kirsh KL. Managing pain in patients with aberrant drug-taking behaviors. J Support Oncol 2005; 3:83–86.
- Ernst E, Cassileth BR. The prevalence of complementary/alternative medicine in cancer: a systematic review. Cancer 1998; 83:777–782.
- Eisenberg DM, Davis RB, Ettner SL, et al. Trends in alternative medicine use in the United States, 1990–1997: results of a follow-up national survey. JAMA 1998; 280:1569–1575.
- Richardson MA, Sanders T, Palmer JL, Greisinger A, Singletary SE. Complementary/alternative medicine use in a comprehensive cancer center and the implications for oncology. J Clin Oncol 2000; 18:2505–2514.
- Adler SR, Fosket JR. Disclosing complementary and alternative medicine use in the medical encounter: a qualitative study in women with breast cancer. J Fam Pract 1999; 48:453–458.
- Newell S, Sanson-Fisher RW. Australian oncologists’ self-reported knowledge and attitudes about non-traditional therapies used by cancer patients. Med J Aust 2000; 172:110–113.
- Berman BM, Singh BK, Lao L, Singh BB, Ferentz KS, Hartnoll SM. Physicians’ attitudes toward complementary or alternative medicine: a regional survey. J Am Board Fam Pract 1995; 8:361–366.
- Verhoef MJ, Sutherland LR. General practitioners’ assessment of and interest in alternative medicine in Canada. Soc Sci Med 1995; 41:511–515.
- American Cancer Society: Treatment guidelines for patients. Version 1. http://www.cancer.org/downloads/CRI/NCCN_pain.pdf.
- Benedetti C, Brock C, Cleeland C, et al; National Comprehensive Cancer Network. NCCN Practice Guidelines for Cancer Pain. Oncology (Williston Park) 2000; 14:135–150.
- Bardia A, Barton DL, Prokop LJ, Bauer BA, Moynihan TJ. Efficacy of complementary and alternative medicine therapies in relieving cancer pain: a systematic review. J Clin Oncol 2006; 24:5457–5464.
- Lee H, Schmidt K, Ernst E. Acupuncture for the relief of cancer-related pain—a systematic review. Eur J Pain 2005; 9:437–444.
- Alimi D, Rubino C, Pichard-Léandri E, Fermand-Brulé S, Dubreuil-Lemaire ML, Hill C. Analgesic effect of auricular acupuncture for cancer pain: a randomized, blinded, controlled trial. J Clin Oncol 2003; 21:4120–4126.
- Spiegel D, Bloom JR. Group therapy and hypnosis reduce metastatic breast carcinoma pain. Psychosom Med 1983; 45:333–339.
- Goodwin PJ, Leszcz M, Ennis M, et al. The effect of group psychosocial support on survival in metastatic breast cancer. N Engl J Med 2001; 345:1719–1726.
- Syrjala KL, Donaldson GW, Davis MW, Kippes ME, Carr JE. Relaxation and imagery and cognitive-behavioral training reduce pain during cancer treatment: a controlled clinical trial. Pain 1995; 63:189–198.
- Sloman R, Brown P, Aldana E, Chee E. The use of relaxation for the promotion of comfort and pain relief in persons with advanced cancer. Contemp Nurse 1994; 3:6–12.
- Weinrich SP, Weinrich MC. The effect of massage on pain in cancer patients. Appl Nurs Res 1990; 3:140–145.
- Wilkie DJ, Kampbell J, Cutshall S, et al. Effects of massage on pain intensity, analgesics and quality of life in patients with cancer pain: a pilot study of a randomized clinical trial conducted within hospice care delivery. Hosp J 2000; 15:31–53.
- Soden K, Vincent K, Craske S, Lucas C, Ashley S. A randomized controlled trial of aromatherapy massage in a hospice setting. Palliat Med 2004; 18:87–92.
- Post-White J, Kinney ME, Savik K, Gau JB, Wilcox C, Lerner I. Therapeutic massage and healing touch improve symptoms in cancer. Integr Cancer Ther 2003; 2:332–344.
- Olson K, Hanson J, Michaud M. A phase II trial of Reiki for the management of pain in advanced cancer patients. J Pain Symptom Manage 2003; 26:990–997.
- Gallagher LM, Lagman R, Walsh D, Davis MP, Legrand SB. The clinical effects of music therapy in palliative medicine. Support Care Cancer 2006; 14:859–866.
- Olaku O, White JD. Herbal therapy use by cancer patients: a literature review on case reports. Eur J Cancer 2011; 47:508–514.
Some 90% of patients with cancer experience pain during their illness.1 The pain usually worsens as the disease progresses, and patients may experience different types of pain.
Persistent pain decreases function, appetite, and sleep, induces fear, causes depression, and generally lowers the quality of life.2 Persistent pain is demoralizing and debilitating for patients and their caregivers.3
Adequate pain control is important to ensure that patients can function productively, maintain social relationships, and improve their quality of life.2 Yet 86% of practicing physicians surveyed believed that most cancer patients with pain were undermedicated,2 and most felt that pain management is unsuccessful in more than half of patients who seek help.3
The critical importance of pain management has been emphasized by the World Health Organization (WHO), by international and national professional organizations, and by government agencies. All practitioners who care for cancer patients need to be well educated in managing cancer pain, a key part of which is to educate patients about the process and what to expect. This results in better pain control.4
Although much has been written on the management of cancer pain in a referral setting, little has been published on how to manage it in primary care. In this article, we discuss common questions faced by generalists. We emphasize the use of opioids, perhaps the most challenging aspect of cancer pain management. We also discuss when consultation with a specialist in pain management or a palliative medicine specialist is especially helpful.
WHAT ARE THE DIFFERENT TYPES OF PAIN SYNDROMES?
Pain is classified in several ways1–6:
Nociceptive vs neuropathic. Nociceptive pain comprises somatic and visceral components and is the result of continued tissue injury.4 Neuropathic pain is due to injury to the peripheral and central nervous systems and occurs within an area of sensory or motor deficit.
Continuous vs intermittent. Continuous pain, even if controlled, can have breakthroughs, ie, flares of pain above the controlled baseline level. Intermittent pain is a pain flare without chronic baseline pain. Intermittent pain is further divided into incident pain (ie, on movement) and end-of-dose failure (ie, pain occurring just before the next scheduled opioid dose).5 Pain specialists continue to debate the meaning and the use of these terms.
Malignant vs nonmalignant. Cancer pain is multifactorial,1 being induced by the disease itself, by the treatment of cancer, and by pain unrelated to cancer or its treatment (eg, osteoarthritis or diabetic neuropathy).2
Familiarity with the causes and the types of pain, including pain related to cancer, is important, as this influences treatment decisions.
HOW IS PAIN ASSESSED?
The assessment of pain is vital in managing it.
Since pain is inherently subjective, the patient’s self-report is the gold standard.4 Characteristics of the pain along with a physical examination, laboratory testing, and imaging studies can define the pathophysiology of the pain and influence the decision to undertake further assessment or specific therapies.
Patients and physicians can use various scales, such as a visual analog scale, a numerical rating scale, a graphic scale, a verbal scale, a word descriptor scale, and a functional pain scale. A verbal scale can be used if the patient is alert, or a nonverbal scale if the patient has impaired cognition or speaks a different language. Intensity is the most common dimension evaluated in cancer pain, primarily via a numerical or visual analog scale. A numerical scale score of 0 to 10 has been found to be as effective as a visual analog scale (0 to 100 mm),7,8 and the numerical rating scale is generally preferred as a measure of pain intensity.9
There are no clear guidelines for selecting one scale over another.7 A clinically meaningful response (ie, meaningful to patients) is at least a two-point decrease on the 10-point numerical scale or a 13-mm decrease on the 100-mm visual analog scale. A decrease in the percentage of the pain relates to global improvement better than an absolute reduction on the numerical scale.
WHAT PROBLEMS ARE ENCOUNTERED IN MANAGING CANCER PAIN?
WHAT ARE THE DIFFERENT WAYS TO MANAGE CANCER PAIN?
Pain should be treated promptly and aggressively, because if untreated it can lead to delays in healing, changes in the central nervous system (eg, sensitization, plasticity), chronic stress, family stress, depression, job loss, and even suicide.12–14
Comprehensive pain management improves outcomes and includes the rational use of opioids and adjuvant analgesics, physical rehabilitation, cognitive behavioral (non-drug) therapies, family counseling, interventional procedures (kyphoplasty, nerve blocks, local injections, spinal analgesia), and complementary therapies such as acupuncture.12 Adjuvant analgesics include antidepressants, anticonvulsants, and local anesthetics.
HOW DO OPIOIDS RELIEVE CANCER PAIN?
Opioids bind to receptors in tissues throughout the body, including in the central and peripheral nervous systems15 and the digestive tract. The binding of an opioid to an opioid receptor—including mu, kappa, and delta receptors and orphan receptor-like ligand-1—initiates a cascade of intracellular reactions. Due to the nature of different interactions of opioids with each of these receptors, individuals vary in their response to opioids.15
WHAT ARE THE CHARACTERISTICS OF COMMON OPIOIDS?
- Step 1. Mild pain calls for a nonopioid analgesic with or without an adjuvant (more about adjuvants below).
- Step 2. Mild or moderate pain that persists or increases calls for a weak opioid such as codeine, tramadol (Ultram), or hydrocodone, with or without a nonopioid and with or without an adjuvant.
- Step 3. Severe pain calls for a strong opioid with or without a nonopioid, and with or without an adjuvant.
Morphine, the prototypical opioid, is well studied and versatile, as it can be given orally, parenterally, rectally, or intraspinally. It is readily available in the United States and Western Europe but not in some parts of the world, such as Asia and Africa. It is also cost-effective.
Hydromorphone (Dilaudid) is similar to morphine in terms of versatility, cost, and effectiveness in pain management. An extended-release form (Exalgo) is now available in the United States.
Oxycodone is readily available in both slow-release (eg, OxyContin) and immediate-release (eg, Oxy-IR) preparations and is also cost-effective. However, there is no parenteral formulation in the United States.
Methadone is inexpensive and can be used as a long-acting or an immediate-release opioid. However, it should be used with caution in patients with a prolonged QTc interval: in general, a QTc interval of 430 to 450 msec is not a contraindication, but there is a risk of torsades de pointes when the QTc is greater than 500 msec. The physician should also look for drug interactions when prescribing methadone, which is metabolized in the liver via the cytochrome P450 3A4 system. Methadone use can also lead to respiratory depression, prolonged QTc interval, and sudden death.
Buprenorphine can be used as a third- or fourth-tier opioid for patients with both kidney and liver failure. It can be given sublingually or parenterally. It may not be readily available, may not be covered by insurance, and is expensive.
Selecting an opioid to try first
The following are some general considerations when selecting an opioid to try first:
- Does the patient have a history of organ failure? Has the patient had a therapeutic response to, or adverse effects from, a particular opioid in the past?
- Which route would best fit the patient’s needs? (Oral is always preferable.)
- How often will breakthrough dosing be required? (In general, the breakthrough dose is administered at the drug’s half-life, but it can be administered between 1 and 4 hours.)
- How much will it cost? (Consider the cost, insurance coverage, and co-pays.)
Table 2 shows different characteristics of commonly used opioids, including route of administration, onset of action, peak effect, and duration of action.1
WHAT ARE THE EQUIANALGESIC DOSES OF COMMONLY USED OPIOIDS?
Table 3 lists equianalgesic doses and route conversions of commonly used opioids.18–20
WHAT ARE THE PRINCIPLES BEHIND OPIOID DOSING?
Table 4 shows important strategies for opioid dosing. An in-depth discussion of specific opioid dosing strategies is beyond the scope of this article.5
WHAT ARE THE COMMON ADVERSE EFFECTS OF OPIOIDS?
Adverse effects are among the most common reasons for failure of opioids to relieve pain. If these effects are not anticipated and treated prophylactically, patients may avoid taking their opioid drugs or may complain that they are “allergic” to them. In reality, true allergy to any of the opioids is rare. Patients comply better if they are taught to expect that most adverse effects are either preventable or manageable.21 A simple strategy includes reducing the opioid dose by 25% to 50%, using different opioids (“rotation”), changing the route of administration, and directly treating adverse effects.21,22
WHAT IS OPIOID ROTATION AND HOW IS IT DONE?
Opioid rotation involves changing to a different drug using the same administration route, with the aim of improving the analgesic response or reducing adverse effects.16 It may be useful in widening the therapeutic window, ie, establishing a more advantageous relationship between analgesia and toxicity.16 This strategy applies, for example, to patients who have an adverse reaction to morphine, and who may need rotation to fentanyl or methadone.
The major indication for switching opioids is poorly controlled pain with unacceptable adverse effects due to opioid toxicity, the rapid development of tolerance, refractory pain, or difficult pain syndromes.24 A recent prospective study showed that 42% of patients underwent opioid rotation, and the two most common reasons were inadequate analgesia and severe adverse effects.25 Opioid rotation resulted in relief of confusion (72%), nausea and vomiting (68%), and drowsiness (53%).25
Before trying opioid rotation, review the patient’s pain syndromes and the use of an adjuvant analgesic, and assess for evidence of opioid toxicity or contributing abnormal biochemical factors such as hydration status.24,26 Most opioids are mu-receptor agonists and may exhibit cross-tolerance, a phenomenon in which the alternative drug does not have the expected effects because of similar pharmacologic action of the first drug. Because the degree of cross-tolerance may change as opioid doses are escalated, it is advisable to proceed with caution when switching from one opioid to another in patients who are receiving very high doses. Opioid rotation generally would be ineffective if there is complete analgesic cross-tolerance between opioids.
The common equivalency conversion tables are based either on studies in patients who received low doses of opioids or on single-dose studies.16,24 By substituting opioids and using lower doses than expected according to the equivalency conversion tables (generally a 25% to 30% decrease), it is possible in most cases to reduce or relieve the symptoms of opioid toxicity and to manage patients highly tolerant to previous opioids while improving analgesia.24
Alternatives to opioid rotation are route conversion (oral to parenteral or spinal), addition of an adjuvant analgesic, and opioid dose reduction.
WHAT IS OPIOID TOXICITY AND HOW IS IT MANAGED?
Opioid overdose is commonly the result of an error in pain assessment, opioid prescribing, or dose administration. Opioid overdose classically presents as sedation or respiratory depression. The combination of coma, reduced respiratory rate, and pinpoint pupils is highly suggestive of opioid toxicity, and treatment should be initiated promptly.
This scenario, however, is the extreme example of opioid overdose, and it is rare when a patient is given the correct opioid dose titrated gradually over a period of time. The more common scenario is when a patient’s pain has finally been managed and the patient is resting comfortably with slow respirations. This would not warrant naloxone (Narcan) administration but rather close observation and monitoring of vital signs.
Naloxone has antagonist activity at all of the receptor sites.27 It is important to be alert for acute opioid withdrawal in patients taking high-dose opioids for a long time.27 There are no guidelines as to the route of administration and the dosing of naloxone. Table 6 summarizes the management of opioid overdose using naloxone.5
WHAT IS THE ROLE OF ADJUVANTS?
An adjuvant analgesic is any drug with a primary indication other than pain, but with analgesic properties in some painful conditions. Adjuvants are best used when a patient cannot obtain satisfactory pain relief from an opioid.28 Antidepressants, anticonvulsants, neuroleptics, antiarrhythmics, antihistamines, N-methyl-d-aspartate (NMDA) receptor antagonists, steroids, muscle relaxants, bisphosphonates, and radiopharmaceuticals can be adjuvant agents.29
Adjuvants are generally used to complement the analgesic effects of opioids to achieve optimal pain control with a minimum of adverse effects.28 The following scenarios should prompt the use of adjuvants in clinical practice28:
- The toxic limit of a primary pain medication has been reached.
- The therapeutic benefit of the primary pain medication has reached a plateau.
- The primary analgesic could not be used because of substance-abuse behavior, multiple organ failure, allergy, etc.
- The patient has multiple pain syndromes.
- The patient has additional symptoms unrelated to pain, eg, insomnia or depression.
Table 7 lists adjuvants with specific indications and points to remember when prescribing them.28,29
WHAT IS THE ROLE OF NSAIDs FOR CANCER PAIN?
Nonsteroidal anti-inflammatory drugs (NSAIDs) have a well-established role in treating cancer-related pain, either on their own for mild pain or in combination with opioids for moderate to severe pain, leading to additive analgesia. Using NSAIDs as adjuvants is common practice in certain cancer pain syndromes, such as malignant bone pain, although there is considerable variation in response.31
NSAIDs have long been known to inhibit peripheral prostaglandin synthesis, but recently they have also been suggested to have a central action. The central effect is related to NMDA receptor-induced activation of the nitric oxide system.31
NSAIDs have ceiling effects, and there is no therapeutic advantage to increasing the dose beyond that which is recommended.
Ketorolac (Toradol), indomethacin (Indocin), and diclofenac (Voltaren) have potent analgesic activity, whereas the “oxicam” NSAIDs show predominantly anti-inflammatory effects.30
No NSAID is clearly superior for a particular type of pain. Certain NSAIDs block the NMDA receptor and inhibit cyclo-oxygenase-1 and cyclo-oxygenase-2. There is a poor correlation between the analgesic effects of NSAIDs and cyclo-oxygenase inhibition. There is no evidence to support the use of selective cyclo-oxygenase-2 inhibitors for cancer pain, and these agents have no advantage over nonselective NSAIDs on the basis of limited gastrointestinal toxicity.32
In cancer pain, NSAIDs may delay the development of tolerance and allow lower doses of opioids to be used, with fewer central nervous system side effects.31,32 Despite the extensive use of NSAIDs, relatively few randomized studies have documented their efficacy in cancer pain compared with other chronic pain syndromes. Data on safe and effective doses from studies of nonmalignant pain may not apply to cancer pain, since cancer patients often have several serious conditions and are on multiple medications. In addition, the potential for adverse effects of NSAIDs (gastrointestinal bleeding, renal failure, thrombosis) may be greater in patients with advanced cancer.
In conclusion, NSAIDs may help if used judiciously in somatic pain and visceral pain, and perhaps even in neuropathic pain.31
HOW IS CANCER PAIN MANAGED IN PATIENTS WITH ORGAN FAILURE?
- Opioids that can be used in liver failure or cirrhosis: morphine, hydromorphone, methadone, levorphanol, buprenorphine.
- Opioids that can be used in renal failure: methadone, fentanyl, and buprenorphine are safest; oxycodone and hydromorphone are moderately safe; morphine is the least safe.35,36
- Opioids that can be used in both kidney and liver failure: methadone, buprenorphine.
HOW CAN PROBLEMS RELATED TO SUBSTANCE ABUSE BE AVOIDED?
Substance abuse is less a problem in managing cancer pain than in chronic nonmalignant pain. Prescribing opioids safely is challenging, and very little has been published on substance abuse and the management of cancer pain. However, in the absence of practice guidelines, the best approach is to establish a dosing structure, control prescription refills, and monitor the patient.
Abuse is the misuse of an opioid via self-titration or altering the dosing schedule or route of administration. Patients who misuse opioids—ie, take them differently than prescribed—are not necessarily addicted.
Addiction is the abuse of a drug associated with psychological dependence, despite harm.
Diversion can occur without addiction and is done for financial gain, and this is the worst offense as it may harm others.
Pseudoaddiction is abnormal, demanding, often hostile behavior resulting from uncontrolled pain; once the pain is controlled, the behavior resolves.
Behaviors such as forging prescriptions, stealing or borrowing drugs, frequently “losing” prescriptions, and resisting changes to medication despite adverse effects are more predictive of addiction than are behaviors such as aggressive complaining about the need for more drugs, drug-hoarding, and unsanctioned dose escalations or other forms of noncompliance, as the latter three are more likely to indicate poorly controlled pain.37
Predictors of opioid abuse include a family history or a personal history of alcohol or drug abuse (including prescription drugs); a history of psychiatric illness (including anxiety disorder); male sex; nonwhite race; a history of driving under the influence of alcohol or drugs; a record of drug-related convictions; lost or stolen prescriptions; and using supplemental sources to obtain opioids.38 Socioeconomic status and disability level were not found to be significant predictors.38
Different scales are available to predict the risk of aberrant drug behavior in patients on chronic opioid therapy. Of the many available, the Screener and Opioid Assessment for Patients With Pain and the Current Opioid Misuse Measure assess all the key factors.38
After an assessment, the next step is monitoring. Unfortunately, no specific method has been validated. In one study, urine toxicology testing was more effective at identifying problems than monitoring patient behavior alone, and monitoring behavior alone would have resulted in missing about half of the patients with a problem.39 The same study showed that even in the absence of aberrant drug-related behavior based on predictors, a significant number of urine toxicology screens were positive.39
A negative urine screen for the patient’s opioid suggests diversion. The clinician should order a screen for the prescribed opioid because a general screen may not detect nonmorphine opioids. A general screen may detect polysubstance abuse, which is common in individuals with addiction.
Table 9 summarizes a strategy to manage opioid therapy in patients with history of substance abuse.40
WHAT IS THE ROLE OF COMPLEMENTARY AND ALTERNATIVE THERAPIES?
Complementary and alternative medicine therapies are commonly used by cancer patients, with an average prevalence rate of 31%.41–43 As the names suggest, they have been used both as an alternative to and as a complement to conventional medicine. Practitioners of complementary and alternative medicine emphasize its holistic, individualistic, empowering, and educational nature.
Patients do not routinely ask their physicians about these therapies,44 and physicians often have only a limited knowledge of them.45 Surveys of North American physicians showed that they view certain of these therapies as legitimate and effective.46,47
The role of complementary and alternative medicine in cancer pain has been the subject of debate, as relatively little is known about adverse effects and drug interactions. Nevertheless, the American Cancer Society and the National Comprehensive Cancer Network guidelines on cancer pain recommend nonpharmacologic treatment be added for patients who report a pain score of 4 or greater on a 10-point scale after analgesic adjustment.48,49
Most studies of complementary and alternative therapies for cancer pain are of poor quality, with significant shortcomings in methodology and study design and with no clear definition of outcomes.50
Acupuncture is probably the most studied of these therapies, but clinical trials so far have not shown it to be an effective adjunct analgesic for cancer pain.51 A placebo-controlled, blinded randomized trial using auricular acupuncture showed a pain score decrease of 36% from baseline at 2 months compared with controls.52
Studies involving cognitive therapy, supportive psychotherapy, and hypnosis showed modest benefit.53,54 Two trials involving relaxation and imagery reduced cancer pain compared with controls.55,56
Studies of massage therapy have shown mixed results; two studies reported a significant reduction in pain immediately after intervention, and no study found pain relief after 4 weeks.57–60 Studies involving Reiki and touch therapy were inconclusive.60,61
Music therapy has been used to treat patients physically, psychologically, socially, emotionally, and spiritually, with evidence still equivocal. A large prospective observational study involving 200 patients conducted by Gallagher et al62 showed pain was reduced by 30% after music therapy intervention. The same study showed a reduction in depression and anxiety.62 Music therapy could be used as a component of a multimodal approach to pain.
Herbal preparations are often used to treat cancer and symptoms by patients and naturalists. Some herbal medicines are known to cause toxicity in cancer patients. Examples are PC-SPES, mistletoe, and saw palmetto.63
At this juncture, there is some evidence that some complementary and alternative therapies can relieve cancer pain, and the most promising therapy seems to be related to mind-body medicine (eg, biofeedback, relaxation techniques). But before we can legitimately integrate these therapies into the management of cancer pain, we need large randomized controlled trials to determine if they are effective in patients on chronic high-dose opioids and if they decrease the need for opioids.
Some 90% of patients with cancer experience pain during their illness.1 The pain usually worsens as the disease progresses, and patients may experience different types of pain.
Persistent pain decreases function, appetite, and sleep, induces fear, causes depression, and generally lowers the quality of life.2 Persistent pain is demoralizing and debilitating for patients and their caregivers.3
Adequate pain control is important to ensure that patients can function productively, maintain social relationships, and improve their quality of life.2 Yet 86% of practicing physicians surveyed believed that most cancer patients with pain were undermedicated,2 and most felt that pain management is unsuccessful in more than half of patients who seek help.3
The critical importance of pain management has been emphasized by the World Health Organization (WHO), by international and national professional organizations, and by government agencies. All practitioners who care for cancer patients need to be well educated in managing cancer pain, a key part of which is to educate patients about the process and what to expect. This results in better pain control.4
Although much has been written on the management of cancer pain in a referral setting, little has been published on how to manage it in primary care. In this article, we discuss common questions faced by generalists. We emphasize the use of opioids, perhaps the most challenging aspect of cancer pain management. We also discuss when consultation with a specialist in pain management or a palliative medicine specialist is especially helpful.
WHAT ARE THE DIFFERENT TYPES OF PAIN SYNDROMES?
Pain is classified in several ways1–6:
Nociceptive vs neuropathic. Nociceptive pain comprises somatic and visceral components and is the result of continued tissue injury.4 Neuropathic pain is due to injury to the peripheral and central nervous systems and occurs within an area of sensory or motor deficit.
Continuous vs intermittent. Continuous pain, even if controlled, can have breakthroughs, ie, flares of pain above the controlled baseline level. Intermittent pain is a pain flare without chronic baseline pain. Intermittent pain is further divided into incident pain (ie, on movement) and end-of-dose failure (ie, pain occurring just before the next scheduled opioid dose).5 Pain specialists continue to debate the meaning and the use of these terms.
Malignant vs nonmalignant. Cancer pain is multifactorial,1 being induced by the disease itself, by the treatment of cancer, and by pain unrelated to cancer or its treatment (eg, osteoarthritis or diabetic neuropathy).2
Familiarity with the causes and the types of pain, including pain related to cancer, is important, as this influences treatment decisions.
HOW IS PAIN ASSESSED?
The assessment of pain is vital in managing it.
Since pain is inherently subjective, the patient’s self-report is the gold standard.4 Characteristics of the pain along with a physical examination, laboratory testing, and imaging studies can define the pathophysiology of the pain and influence the decision to undertake further assessment or specific therapies.
Patients and physicians can use various scales, such as a visual analog scale, a numerical rating scale, a graphic scale, a verbal scale, a word descriptor scale, and a functional pain scale. A verbal scale can be used if the patient is alert, or a nonverbal scale if the patient has impaired cognition or speaks a different language. Intensity is the most common dimension evaluated in cancer pain, primarily via a numerical or visual analog scale. A numerical scale score of 0 to 10 has been found to be as effective as a visual analog scale (0 to 100 mm),7,8 and the numerical rating scale is generally preferred as a measure of pain intensity.9
There are no clear guidelines for selecting one scale over another.7 A clinically meaningful response (ie, meaningful to patients) is at least a two-point decrease on the 10-point numerical scale or a 13-mm decrease on the 100-mm visual analog scale. A decrease in the percentage of the pain relates to global improvement better than an absolute reduction on the numerical scale.
WHAT PROBLEMS ARE ENCOUNTERED IN MANAGING CANCER PAIN?
WHAT ARE THE DIFFERENT WAYS TO MANAGE CANCER PAIN?
Pain should be treated promptly and aggressively, because if untreated it can lead to delays in healing, changes in the central nervous system (eg, sensitization, plasticity), chronic stress, family stress, depression, job loss, and even suicide.12–14
Comprehensive pain management improves outcomes and includes the rational use of opioids and adjuvant analgesics, physical rehabilitation, cognitive behavioral (non-drug) therapies, family counseling, interventional procedures (kyphoplasty, nerve blocks, local injections, spinal analgesia), and complementary therapies such as acupuncture.12 Adjuvant analgesics include antidepressants, anticonvulsants, and local anesthetics.
HOW DO OPIOIDS RELIEVE CANCER PAIN?
Opioids bind to receptors in tissues throughout the body, including in the central and peripheral nervous systems15 and the digestive tract. The binding of an opioid to an opioid receptor—including mu, kappa, and delta receptors and orphan receptor-like ligand-1—initiates a cascade of intracellular reactions. Due to the nature of different interactions of opioids with each of these receptors, individuals vary in their response to opioids.15
WHAT ARE THE CHARACTERISTICS OF COMMON OPIOIDS?
- Step 1. Mild pain calls for a nonopioid analgesic with or without an adjuvant (more about adjuvants below).
- Step 2. Mild or moderate pain that persists or increases calls for a weak opioid such as codeine, tramadol (Ultram), or hydrocodone, with or without a nonopioid and with or without an adjuvant.
- Step 3. Severe pain calls for a strong opioid with or without a nonopioid, and with or without an adjuvant.
Morphine, the prototypical opioid, is well studied and versatile, as it can be given orally, parenterally, rectally, or intraspinally. It is readily available in the United States and Western Europe but not in some parts of the world, such as Asia and Africa. It is also cost-effective.
Hydromorphone (Dilaudid) is similar to morphine in terms of versatility, cost, and effectiveness in pain management. An extended-release form (Exalgo) is now available in the United States.
Oxycodone is readily available in both slow-release (eg, OxyContin) and immediate-release (eg, Oxy-IR) preparations and is also cost-effective. However, there is no parenteral formulation in the United States.
Methadone is inexpensive and can be used as a long-acting or an immediate-release opioid. However, it should be used with caution in patients with a prolonged QTc interval: in general, a QTc interval of 430 to 450 msec is not a contraindication, but there is a risk of torsades de pointes when the QTc is greater than 500 msec. The physician should also look for drug interactions when prescribing methadone, which is metabolized in the liver via the cytochrome P450 3A4 system. Methadone use can also lead to respiratory depression, prolonged QTc interval, and sudden death.
Buprenorphine can be used as a third- or fourth-tier opioid for patients with both kidney and liver failure. It can be given sublingually or parenterally. It may not be readily available, may not be covered by insurance, and is expensive.
Selecting an opioid to try first
The following are some general considerations when selecting an opioid to try first:
- Does the patient have a history of organ failure? Has the patient had a therapeutic response to, or adverse effects from, a particular opioid in the past?
- Which route would best fit the patient’s needs? (Oral is always preferable.)
- How often will breakthrough dosing be required? (In general, the breakthrough dose is administered at the drug’s half-life, but it can be administered between 1 and 4 hours.)
- How much will it cost? (Consider the cost, insurance coverage, and co-pays.)
Table 2 shows different characteristics of commonly used opioids, including route of administration, onset of action, peak effect, and duration of action.1
WHAT ARE THE EQUIANALGESIC DOSES OF COMMONLY USED OPIOIDS?
Table 3 lists equianalgesic doses and route conversions of commonly used opioids.18–20
WHAT ARE THE PRINCIPLES BEHIND OPIOID DOSING?
Table 4 shows important strategies for opioid dosing. An in-depth discussion of specific opioid dosing strategies is beyond the scope of this article.5
WHAT ARE THE COMMON ADVERSE EFFECTS OF OPIOIDS?
Adverse effects are among the most common reasons for failure of opioids to relieve pain. If these effects are not anticipated and treated prophylactically, patients may avoid taking their opioid drugs or may complain that they are “allergic” to them. In reality, true allergy to any of the opioids is rare. Patients comply better if they are taught to expect that most adverse effects are either preventable or manageable.21 A simple strategy includes reducing the opioid dose by 25% to 50%, using different opioids (“rotation”), changing the route of administration, and directly treating adverse effects.21,22
WHAT IS OPIOID ROTATION AND HOW IS IT DONE?
Opioid rotation involves changing to a different drug using the same administration route, with the aim of improving the analgesic response or reducing adverse effects.16 It may be useful in widening the therapeutic window, ie, establishing a more advantageous relationship between analgesia and toxicity.16 This strategy applies, for example, to patients who have an adverse reaction to morphine, and who may need rotation to fentanyl or methadone.
The major indication for switching opioids is poorly controlled pain with unacceptable adverse effects due to opioid toxicity, the rapid development of tolerance, refractory pain, or difficult pain syndromes.24 A recent prospective study showed that 42% of patients underwent opioid rotation, and the two most common reasons were inadequate analgesia and severe adverse effects.25 Opioid rotation resulted in relief of confusion (72%), nausea and vomiting (68%), and drowsiness (53%).25
Before trying opioid rotation, review the patient’s pain syndromes and the use of an adjuvant analgesic, and assess for evidence of opioid toxicity or contributing abnormal biochemical factors such as hydration status.24,26 Most opioids are mu-receptor agonists and may exhibit cross-tolerance, a phenomenon in which the alternative drug does not have the expected effects because of similar pharmacologic action of the first drug. Because the degree of cross-tolerance may change as opioid doses are escalated, it is advisable to proceed with caution when switching from one opioid to another in patients who are receiving very high doses. Opioid rotation generally would be ineffective if there is complete analgesic cross-tolerance between opioids.
The common equivalency conversion tables are based either on studies in patients who received low doses of opioids or on single-dose studies.16,24 By substituting opioids and using lower doses than expected according to the equivalency conversion tables (generally a 25% to 30% decrease), it is possible in most cases to reduce or relieve the symptoms of opioid toxicity and to manage patients highly tolerant to previous opioids while improving analgesia.24
Alternatives to opioid rotation are route conversion (oral to parenteral or spinal), addition of an adjuvant analgesic, and opioid dose reduction.
WHAT IS OPIOID TOXICITY AND HOW IS IT MANAGED?
Opioid overdose is commonly the result of an error in pain assessment, opioid prescribing, or dose administration. Opioid overdose classically presents as sedation or respiratory depression. The combination of coma, reduced respiratory rate, and pinpoint pupils is highly suggestive of opioid toxicity, and treatment should be initiated promptly.
This scenario, however, is the extreme example of opioid overdose, and it is rare when a patient is given the correct opioid dose titrated gradually over a period of time. The more common scenario is when a patient’s pain has finally been managed and the patient is resting comfortably with slow respirations. This would not warrant naloxone (Narcan) administration but rather close observation and monitoring of vital signs.
Naloxone has antagonist activity at all of the receptor sites.27 It is important to be alert for acute opioid withdrawal in patients taking high-dose opioids for a long time.27 There are no guidelines as to the route of administration and the dosing of naloxone. Table 6 summarizes the management of opioid overdose using naloxone.5
WHAT IS THE ROLE OF ADJUVANTS?
An adjuvant analgesic is any drug with a primary indication other than pain, but with analgesic properties in some painful conditions. Adjuvants are best used when a patient cannot obtain satisfactory pain relief from an opioid.28 Antidepressants, anticonvulsants, neuroleptics, antiarrhythmics, antihistamines, N-methyl-d-aspartate (NMDA) receptor antagonists, steroids, muscle relaxants, bisphosphonates, and radiopharmaceuticals can be adjuvant agents.29
Adjuvants are generally used to complement the analgesic effects of opioids to achieve optimal pain control with a minimum of adverse effects.28 The following scenarios should prompt the use of adjuvants in clinical practice28:
- The toxic limit of a primary pain medication has been reached.
- The therapeutic benefit of the primary pain medication has reached a plateau.
- The primary analgesic could not be used because of substance-abuse behavior, multiple organ failure, allergy, etc.
- The patient has multiple pain syndromes.
- The patient has additional symptoms unrelated to pain, eg, insomnia or depression.
Table 7 lists adjuvants with specific indications and points to remember when prescribing them.28,29
WHAT IS THE ROLE OF NSAIDs FOR CANCER PAIN?
Nonsteroidal anti-inflammatory drugs (NSAIDs) have a well-established role in treating cancer-related pain, either on their own for mild pain or in combination with opioids for moderate to severe pain, leading to additive analgesia. Using NSAIDs as adjuvants is common practice in certain cancer pain syndromes, such as malignant bone pain, although there is considerable variation in response.31
NSAIDs have long been known to inhibit peripheral prostaglandin synthesis, but recently they have also been suggested to have a central action. The central effect is related to NMDA receptor-induced activation of the nitric oxide system.31
NSAIDs have ceiling effects, and there is no therapeutic advantage to increasing the dose beyond that which is recommended.
Ketorolac (Toradol), indomethacin (Indocin), and diclofenac (Voltaren) have potent analgesic activity, whereas the “oxicam” NSAIDs show predominantly anti-inflammatory effects.30
No NSAID is clearly superior for a particular type of pain. Certain NSAIDs block the NMDA receptor and inhibit cyclo-oxygenase-1 and cyclo-oxygenase-2. There is a poor correlation between the analgesic effects of NSAIDs and cyclo-oxygenase inhibition. There is no evidence to support the use of selective cyclo-oxygenase-2 inhibitors for cancer pain, and these agents have no advantage over nonselective NSAIDs on the basis of limited gastrointestinal toxicity.32
In cancer pain, NSAIDs may delay the development of tolerance and allow lower doses of opioids to be used, with fewer central nervous system side effects.31,32 Despite the extensive use of NSAIDs, relatively few randomized studies have documented their efficacy in cancer pain compared with other chronic pain syndromes. Data on safe and effective doses from studies of nonmalignant pain may not apply to cancer pain, since cancer patients often have several serious conditions and are on multiple medications. In addition, the potential for adverse effects of NSAIDs (gastrointestinal bleeding, renal failure, thrombosis) may be greater in patients with advanced cancer.
In conclusion, NSAIDs may help if used judiciously in somatic pain and visceral pain, and perhaps even in neuropathic pain.31
HOW IS CANCER PAIN MANAGED IN PATIENTS WITH ORGAN FAILURE?
- Opioids that can be used in liver failure or cirrhosis: morphine, hydromorphone, methadone, levorphanol, buprenorphine.
- Opioids that can be used in renal failure: methadone, fentanyl, and buprenorphine are safest; oxycodone and hydromorphone are moderately safe; morphine is the least safe.35,36
- Opioids that can be used in both kidney and liver failure: methadone, buprenorphine.
HOW CAN PROBLEMS RELATED TO SUBSTANCE ABUSE BE AVOIDED?
Substance abuse is less a problem in managing cancer pain than in chronic nonmalignant pain. Prescribing opioids safely is challenging, and very little has been published on substance abuse and the management of cancer pain. However, in the absence of practice guidelines, the best approach is to establish a dosing structure, control prescription refills, and monitor the patient.
Abuse is the misuse of an opioid via self-titration or altering the dosing schedule or route of administration. Patients who misuse opioids—ie, take them differently than prescribed—are not necessarily addicted.
Addiction is the abuse of a drug associated with psychological dependence, despite harm.
Diversion can occur without addiction and is done for financial gain, and this is the worst offense as it may harm others.
Pseudoaddiction is abnormal, demanding, often hostile behavior resulting from uncontrolled pain; once the pain is controlled, the behavior resolves.
Behaviors such as forging prescriptions, stealing or borrowing drugs, frequently “losing” prescriptions, and resisting changes to medication despite adverse effects are more predictive of addiction than are behaviors such as aggressive complaining about the need for more drugs, drug-hoarding, and unsanctioned dose escalations or other forms of noncompliance, as the latter three are more likely to indicate poorly controlled pain.37
Predictors of opioid abuse include a family history or a personal history of alcohol or drug abuse (including prescription drugs); a history of psychiatric illness (including anxiety disorder); male sex; nonwhite race; a history of driving under the influence of alcohol or drugs; a record of drug-related convictions; lost or stolen prescriptions; and using supplemental sources to obtain opioids.38 Socioeconomic status and disability level were not found to be significant predictors.38
Different scales are available to predict the risk of aberrant drug behavior in patients on chronic opioid therapy. Of the many available, the Screener and Opioid Assessment for Patients With Pain and the Current Opioid Misuse Measure assess all the key factors.38
After an assessment, the next step is monitoring. Unfortunately, no specific method has been validated. In one study, urine toxicology testing was more effective at identifying problems than monitoring patient behavior alone, and monitoring behavior alone would have resulted in missing about half of the patients with a problem.39 The same study showed that even in the absence of aberrant drug-related behavior based on predictors, a significant number of urine toxicology screens were positive.39
A negative urine screen for the patient’s opioid suggests diversion. The clinician should order a screen for the prescribed opioid because a general screen may not detect nonmorphine opioids. A general screen may detect polysubstance abuse, which is common in individuals with addiction.
Table 9 summarizes a strategy to manage opioid therapy in patients with history of substance abuse.40
WHAT IS THE ROLE OF COMPLEMENTARY AND ALTERNATIVE THERAPIES?
Complementary and alternative medicine therapies are commonly used by cancer patients, with an average prevalence rate of 31%.41–43 As the names suggest, they have been used both as an alternative to and as a complement to conventional medicine. Practitioners of complementary and alternative medicine emphasize its holistic, individualistic, empowering, and educational nature.
Patients do not routinely ask their physicians about these therapies,44 and physicians often have only a limited knowledge of them.45 Surveys of North American physicians showed that they view certain of these therapies as legitimate and effective.46,47
The role of complementary and alternative medicine in cancer pain has been the subject of debate, as relatively little is known about adverse effects and drug interactions. Nevertheless, the American Cancer Society and the National Comprehensive Cancer Network guidelines on cancer pain recommend nonpharmacologic treatment be added for patients who report a pain score of 4 or greater on a 10-point scale after analgesic adjustment.48,49
Most studies of complementary and alternative therapies for cancer pain are of poor quality, with significant shortcomings in methodology and study design and with no clear definition of outcomes.50
Acupuncture is probably the most studied of these therapies, but clinical trials so far have not shown it to be an effective adjunct analgesic for cancer pain.51 A placebo-controlled, blinded randomized trial using auricular acupuncture showed a pain score decrease of 36% from baseline at 2 months compared with controls.52
Studies involving cognitive therapy, supportive psychotherapy, and hypnosis showed modest benefit.53,54 Two trials involving relaxation and imagery reduced cancer pain compared with controls.55,56
Studies of massage therapy have shown mixed results; two studies reported a significant reduction in pain immediately after intervention, and no study found pain relief after 4 weeks.57–60 Studies involving Reiki and touch therapy were inconclusive.60,61
Music therapy has been used to treat patients physically, psychologically, socially, emotionally, and spiritually, with evidence still equivocal. A large prospective observational study involving 200 patients conducted by Gallagher et al62 showed pain was reduced by 30% after music therapy intervention. The same study showed a reduction in depression and anxiety.62 Music therapy could be used as a component of a multimodal approach to pain.
Herbal preparations are often used to treat cancer and symptoms by patients and naturalists. Some herbal medicines are known to cause toxicity in cancer patients. Examples are PC-SPES, mistletoe, and saw palmetto.63
At this juncture, there is some evidence that some complementary and alternative therapies can relieve cancer pain, and the most promising therapy seems to be related to mind-body medicine (eg, biofeedback, relaxation techniques). But before we can legitimately integrate these therapies into the management of cancer pain, we need large randomized controlled trials to determine if they are effective in patients on chronic high-dose opioids and if they decrease the need for opioids.
- Laird B, Colvin L, Fallon M. Management of cancer pain: basic principles and neuropathic cancer pain. Eur J Cancer 2008; 44:1078–1082.
- Chang HM. Cancer pain management. Med Clin North Am 1999; 83:711–736,
- Stannard C, Johnson M. Chronic pain management—can we do better? An interview-based survey in primary care. Curr Med Res Opin 2003; 19:703–706.
- Portenoy RK, Lesage P. Management of cancer pain. Lancet 1999; 353:1695–1700.
- Walsh D, Rivera NI, Davis MP, Lagman R, Legrand SB. Strategies for pain management: Cleveland Clinic Foundation guidelines for opioid dosing for cancer pain. Support Cancer Ther 2004; 1:157–164.
- Foley KM. Acute and chronic pain syndromes. In:Doyle D, Hanks G, Cherny N, Calman K, editors. Oxford Textbook of Palliative Medicine. 3rd ed. Oxford, UK: Oxford University Press; 2005:298–316.
- Jensen MP. The validity and reliability of pain measures in adults with cancer. J Pain 2003; 4:2–21.
- Jensen MP, Turner JA, Romano JM. What is the maximum number of levels needed in pain intensity measurement? Pain 1994; 58:387–392.
- Preston CC, Colman AM. Optimal number of response categories in rating scales: reliability, validity, discriminating power, and respondent p. Acta Psychol (Amst) 2000; 104:1–15.
- Peretti-Watel P, Bendiane MK, Obadia Y, Favre R, Lapiana JM, Moatti JP; South-Eastern France Palliative Care Group. The prescription of opioid analgesics to terminal cancer patients: impact of physicians’ general attitudes and contextual factors. Palliat Support Care 2003; 1:345–352.
- Jacobsen R, Liubarskiene Z, Møldrup C, Christrup L, Sjøgren P, Samsanaviciene J. Barriers to cancer pain management: a review of empirical research. Medicina (Kaunas) 2009; 45:427–433.
- Wiedemer NL, Harden PS, Arndt IO, Gallagher RM. The opioid renewal clinic: a primary care, managed approach to opioid therapy in chronic pain patients at risk for substance abuse. Pain Med 2007; 8:573–584.
- Rome HP, Rome JD. Limbically augmented pain syndrome (LAPS): kindling, corticolimbic sensitization, and the convergence of affective and sensory symptoms in chronic pain disorders. Pain Med 2000; 1:7–23.
- Fishbain DA, Rosomoff HL, Rosomoff RS. Drug abuse, dependence, and addiction in chronic pain patients. Clin J Pain 1992; 8:77–85.
- Murányi M, Radák Z. Pain and opioids. Orv Hetil 2008; 149:2363–2370.
- Vadalouca A, Moka E, Argyra E, Sikioti P, Siafaka I. Opioid rotation in patients with cancer: a review of the current literature. J Opioid Manag 2008; 4:213–250.
- Galvagno SM, Correll DJ, Narang S. Safe oral equianalgesic opioid dosing for patients with moderate-to-severe pain. www.hcplive.com/publications/Resident-and-Staff/2007/2007-04/2007-04_06. Accessed May 25, 2011.
- Walsh D. Pharmacological management of cancer pain. Semin Oncol 2000; 27:45–63.
- Shaheen PE, Walsh D, Lasheen W, Davis MP, Lagman RL. Opioid equianalgesic tables: are they all equally dangerous? J Pain Symptom Manage 2009; 38:409–417.
- Pereira J, Lawlor P, Vigano A, Dorgan M, Bruera E. Equianalgesic dose ratios for opioids. a critical review and proposals for long-term dosing. J Pain Symptom Manage 2001; 22:672–687.
- Harris JD. Management of expected and unexpected opioid-related side effects. Clin J Pain 2008; 24(suppl 10):S8–S13.
- Cherny N, Ripamonti C, Pereira J; Expert Working Group of the European Association of Palliative Care Network. Strategies to manage the adverse effects of oral morphine: an evidence-based report. J Clin Oncol 2001; 19:2542–2554.
- Harris JD, Kotob F. Management of opioid-related side effects. In:de Leon-Casasola OA, ed. Cancer Pain: Pharmacological, Interventional and Palliative Care. Philadelphia: Elsevier Inc; 2006:207–230.
- Mercadante S. Opioid rotation for cancer pain: rationale and clinical aspects. Cancer 1999; 86:1856–1866.
- Cheema B, Lagman RL, Walsh D, et al. A prospective study of opioid rotation in pain due to advanced cancer. J Cancer Pain & Symp Palliat 2006; 2:39–46.
- Schug SA, Zech D, Grond S, Jung H, Meuser T, Stobbe B. A long-term survey of morphine in cancer pain patients. J Pain Symptom Manage 1992; 7:259–266.
- Clarke SF, Dargan PI, Jones AL. Naloxone in opioid poisoning: walking the tightrope. Emerg Med J 2005; 22:612–616.
- Knotkova H, Pappagallo M. Adjuvant analgesics. Med Clin North Am 2007; 91:113–124.
- Lussier D, Huskey AG, Portenoy RK. Adjuvant analgesics in cancer pain management. Oncologist 2004; 9:571–591.
- Johnson JR, Burnell-Nugent M, Lossignol D, Ganae-Motan ED, Potts R, Fallon MT. Multicenter, double-blind, randomized, placebo-controlled, parallel-group study of the efficacy, safety, and tolerability of THC:CBD extract and THC extract in patients with intractable cancerrelated pain. J Pain Symptom Manage 2010; 39:167–179.
- Mercadante S. The use of anti-inflammatory drugs in cancer pain. Cancer Treat Rev 2001; 27:51–61.
- Davis MP, Walsh D, Lagman R, LeGrand SB. Controversies in pharmacotherapy of pain management. Lancet Oncol 2005; 6:696–704.
- Klotz U. Tramadol—the impact of its pharmacokinetic and pharmacodynamic properties on the clinical management of pain. Arzneimittelforschung 2003; 53:681–687.
- Davis MP, Lasheen W, Gamier P. Practical guide to opioids and their complications in managing cancer pain. What oncologists need to know. Oncology (Williston Park) 2007; 21:1229–1238.
- Dean M. Opioids in renal failure and dialysis patients. J Pain Symptom Manage 2004; 28:497–504.
- Davis MP. Buprenorphine in cancer pain. Support Care Cancer 2005; 13:878–887.
- Portenoy RK. Opioid therapy for chronic nonmalignant pain: a review of the critical issues. J Pain Symptom Manage 1996; 11:203–217.
- Turk DC, Swanson KS, Gatchel RJ. Predicting opioid misuse by chronic pain patients: a systematic review and literature synthesis. Clin J Pain 2008; 24:497–508.
- Katz NP, Sherburne S, Beach M, et al. Behavioral monitoring and urine toxicology testing in patients receiving long-term opioid therapy. Anesth Analg 2003; 97:1097–1102,
- Passik SD, Kirsh KL. Managing pain in patients with aberrant drug-taking behaviors. J Support Oncol 2005; 3:83–86.
- Ernst E, Cassileth BR. The prevalence of complementary/alternative medicine in cancer: a systematic review. Cancer 1998; 83:777–782.
- Eisenberg DM, Davis RB, Ettner SL, et al. Trends in alternative medicine use in the United States, 1990–1997: results of a follow-up national survey. JAMA 1998; 280:1569–1575.
- Richardson MA, Sanders T, Palmer JL, Greisinger A, Singletary SE. Complementary/alternative medicine use in a comprehensive cancer center and the implications for oncology. J Clin Oncol 2000; 18:2505–2514.
- Adler SR, Fosket JR. Disclosing complementary and alternative medicine use in the medical encounter: a qualitative study in women with breast cancer. J Fam Pract 1999; 48:453–458.
- Newell S, Sanson-Fisher RW. Australian oncologists’ self-reported knowledge and attitudes about non-traditional therapies used by cancer patients. Med J Aust 2000; 172:110–113.
- Berman BM, Singh BK, Lao L, Singh BB, Ferentz KS, Hartnoll SM. Physicians’ attitudes toward complementary or alternative medicine: a regional survey. J Am Board Fam Pract 1995; 8:361–366.
- Verhoef MJ, Sutherland LR. General practitioners’ assessment of and interest in alternative medicine in Canada. Soc Sci Med 1995; 41:511–515.
- American Cancer Society: Treatment guidelines for patients. Version 1. http://www.cancer.org/downloads/CRI/NCCN_pain.pdf.
- Benedetti C, Brock C, Cleeland C, et al; National Comprehensive Cancer Network. NCCN Practice Guidelines for Cancer Pain. Oncology (Williston Park) 2000; 14:135–150.
- Bardia A, Barton DL, Prokop LJ, Bauer BA, Moynihan TJ. Efficacy of complementary and alternative medicine therapies in relieving cancer pain: a systematic review. J Clin Oncol 2006; 24:5457–5464.
- Lee H, Schmidt K, Ernst E. Acupuncture for the relief of cancer-related pain—a systematic review. Eur J Pain 2005; 9:437–444.
- Alimi D, Rubino C, Pichard-Léandri E, Fermand-Brulé S, Dubreuil-Lemaire ML, Hill C. Analgesic effect of auricular acupuncture for cancer pain: a randomized, blinded, controlled trial. J Clin Oncol 2003; 21:4120–4126.
- Spiegel D, Bloom JR. Group therapy and hypnosis reduce metastatic breast carcinoma pain. Psychosom Med 1983; 45:333–339.
- Goodwin PJ, Leszcz M, Ennis M, et al. The effect of group psychosocial support on survival in metastatic breast cancer. N Engl J Med 2001; 345:1719–1726.
- Syrjala KL, Donaldson GW, Davis MW, Kippes ME, Carr JE. Relaxation and imagery and cognitive-behavioral training reduce pain during cancer treatment: a controlled clinical trial. Pain 1995; 63:189–198.
- Sloman R, Brown P, Aldana E, Chee E. The use of relaxation for the promotion of comfort and pain relief in persons with advanced cancer. Contemp Nurse 1994; 3:6–12.
- Weinrich SP, Weinrich MC. The effect of massage on pain in cancer patients. Appl Nurs Res 1990; 3:140–145.
- Wilkie DJ, Kampbell J, Cutshall S, et al. Effects of massage on pain intensity, analgesics and quality of life in patients with cancer pain: a pilot study of a randomized clinical trial conducted within hospice care delivery. Hosp J 2000; 15:31–53.
- Soden K, Vincent K, Craske S, Lucas C, Ashley S. A randomized controlled trial of aromatherapy massage in a hospice setting. Palliat Med 2004; 18:87–92.
- Post-White J, Kinney ME, Savik K, Gau JB, Wilcox C, Lerner I. Therapeutic massage and healing touch improve symptoms in cancer. Integr Cancer Ther 2003; 2:332–344.
- Olson K, Hanson J, Michaud M. A phase II trial of Reiki for the management of pain in advanced cancer patients. J Pain Symptom Manage 2003; 26:990–997.
- Gallagher LM, Lagman R, Walsh D, Davis MP, Legrand SB. The clinical effects of music therapy in palliative medicine. Support Care Cancer 2006; 14:859–866.
- Olaku O, White JD. Herbal therapy use by cancer patients: a literature review on case reports. Eur J Cancer 2011; 47:508–514.
- Laird B, Colvin L, Fallon M. Management of cancer pain: basic principles and neuropathic cancer pain. Eur J Cancer 2008; 44:1078–1082.
- Chang HM. Cancer pain management. Med Clin North Am 1999; 83:711–736,
- Stannard C, Johnson M. Chronic pain management—can we do better? An interview-based survey in primary care. Curr Med Res Opin 2003; 19:703–706.
- Portenoy RK, Lesage P. Management of cancer pain. Lancet 1999; 353:1695–1700.
- Walsh D, Rivera NI, Davis MP, Lagman R, Legrand SB. Strategies for pain management: Cleveland Clinic Foundation guidelines for opioid dosing for cancer pain. Support Cancer Ther 2004; 1:157–164.
- Foley KM. Acute and chronic pain syndromes. In:Doyle D, Hanks G, Cherny N, Calman K, editors. Oxford Textbook of Palliative Medicine. 3rd ed. Oxford, UK: Oxford University Press; 2005:298–316.
- Jensen MP. The validity and reliability of pain measures in adults with cancer. J Pain 2003; 4:2–21.
- Jensen MP, Turner JA, Romano JM. What is the maximum number of levels needed in pain intensity measurement? Pain 1994; 58:387–392.
- Preston CC, Colman AM. Optimal number of response categories in rating scales: reliability, validity, discriminating power, and respondent p. Acta Psychol (Amst) 2000; 104:1–15.
- Peretti-Watel P, Bendiane MK, Obadia Y, Favre R, Lapiana JM, Moatti JP; South-Eastern France Palliative Care Group. The prescription of opioid analgesics to terminal cancer patients: impact of physicians’ general attitudes and contextual factors. Palliat Support Care 2003; 1:345–352.
- Jacobsen R, Liubarskiene Z, Møldrup C, Christrup L, Sjøgren P, Samsanaviciene J. Barriers to cancer pain management: a review of empirical research. Medicina (Kaunas) 2009; 45:427–433.
- Wiedemer NL, Harden PS, Arndt IO, Gallagher RM. The opioid renewal clinic: a primary care, managed approach to opioid therapy in chronic pain patients at risk for substance abuse. Pain Med 2007; 8:573–584.
- Rome HP, Rome JD. Limbically augmented pain syndrome (LAPS): kindling, corticolimbic sensitization, and the convergence of affective and sensory symptoms in chronic pain disorders. Pain Med 2000; 1:7–23.
- Fishbain DA, Rosomoff HL, Rosomoff RS. Drug abuse, dependence, and addiction in chronic pain patients. Clin J Pain 1992; 8:77–85.
- Murányi M, Radák Z. Pain and opioids. Orv Hetil 2008; 149:2363–2370.
- Vadalouca A, Moka E, Argyra E, Sikioti P, Siafaka I. Opioid rotation in patients with cancer: a review of the current literature. J Opioid Manag 2008; 4:213–250.
- Galvagno SM, Correll DJ, Narang S. Safe oral equianalgesic opioid dosing for patients with moderate-to-severe pain. www.hcplive.com/publications/Resident-and-Staff/2007/2007-04/2007-04_06. Accessed May 25, 2011.
- Walsh D. Pharmacological management of cancer pain. Semin Oncol 2000; 27:45–63.
- Shaheen PE, Walsh D, Lasheen W, Davis MP, Lagman RL. Opioid equianalgesic tables: are they all equally dangerous? J Pain Symptom Manage 2009; 38:409–417.
- Pereira J, Lawlor P, Vigano A, Dorgan M, Bruera E. Equianalgesic dose ratios for opioids. a critical review and proposals for long-term dosing. J Pain Symptom Manage 2001; 22:672–687.
- Harris JD. Management of expected and unexpected opioid-related side effects. Clin J Pain 2008; 24(suppl 10):S8–S13.
- Cherny N, Ripamonti C, Pereira J; Expert Working Group of the European Association of Palliative Care Network. Strategies to manage the adverse effects of oral morphine: an evidence-based report. J Clin Oncol 2001; 19:2542–2554.
- Harris JD, Kotob F. Management of opioid-related side effects. In:de Leon-Casasola OA, ed. Cancer Pain: Pharmacological, Interventional and Palliative Care. Philadelphia: Elsevier Inc; 2006:207–230.
- Mercadante S. Opioid rotation for cancer pain: rationale and clinical aspects. Cancer 1999; 86:1856–1866.
- Cheema B, Lagman RL, Walsh D, et al. A prospective study of opioid rotation in pain due to advanced cancer. J Cancer Pain & Symp Palliat 2006; 2:39–46.
- Schug SA, Zech D, Grond S, Jung H, Meuser T, Stobbe B. A long-term survey of morphine in cancer pain patients. J Pain Symptom Manage 1992; 7:259–266.
- Clarke SF, Dargan PI, Jones AL. Naloxone in opioid poisoning: walking the tightrope. Emerg Med J 2005; 22:612–616.
- Knotkova H, Pappagallo M. Adjuvant analgesics. Med Clin North Am 2007; 91:113–124.
- Lussier D, Huskey AG, Portenoy RK. Adjuvant analgesics in cancer pain management. Oncologist 2004; 9:571–591.
- Johnson JR, Burnell-Nugent M, Lossignol D, Ganae-Motan ED, Potts R, Fallon MT. Multicenter, double-blind, randomized, placebo-controlled, parallel-group study of the efficacy, safety, and tolerability of THC:CBD extract and THC extract in patients with intractable cancerrelated pain. J Pain Symptom Manage 2010; 39:167–179.
- Mercadante S. The use of anti-inflammatory drugs in cancer pain. Cancer Treat Rev 2001; 27:51–61.
- Davis MP, Walsh D, Lagman R, LeGrand SB. Controversies in pharmacotherapy of pain management. Lancet Oncol 2005; 6:696–704.
- Klotz U. Tramadol—the impact of its pharmacokinetic and pharmacodynamic properties on the clinical management of pain. Arzneimittelforschung 2003; 53:681–687.
- Davis MP, Lasheen W, Gamier P. Practical guide to opioids and their complications in managing cancer pain. What oncologists need to know. Oncology (Williston Park) 2007; 21:1229–1238.
- Dean M. Opioids in renal failure and dialysis patients. J Pain Symptom Manage 2004; 28:497–504.
- Davis MP. Buprenorphine in cancer pain. Support Care Cancer 2005; 13:878–887.
- Portenoy RK. Opioid therapy for chronic nonmalignant pain: a review of the critical issues. J Pain Symptom Manage 1996; 11:203–217.
- Turk DC, Swanson KS, Gatchel RJ. Predicting opioid misuse by chronic pain patients: a systematic review and literature synthesis. Clin J Pain 2008; 24:497–508.
- Katz NP, Sherburne S, Beach M, et al. Behavioral monitoring and urine toxicology testing in patients receiving long-term opioid therapy. Anesth Analg 2003; 97:1097–1102,
- Passik SD, Kirsh KL. Managing pain in patients with aberrant drug-taking behaviors. J Support Oncol 2005; 3:83–86.
- Ernst E, Cassileth BR. The prevalence of complementary/alternative medicine in cancer: a systematic review. Cancer 1998; 83:777–782.
- Eisenberg DM, Davis RB, Ettner SL, et al. Trends in alternative medicine use in the United States, 1990–1997: results of a follow-up national survey. JAMA 1998; 280:1569–1575.
- Richardson MA, Sanders T, Palmer JL, Greisinger A, Singletary SE. Complementary/alternative medicine use in a comprehensive cancer center and the implications for oncology. J Clin Oncol 2000; 18:2505–2514.
- Adler SR, Fosket JR. Disclosing complementary and alternative medicine use in the medical encounter: a qualitative study in women with breast cancer. J Fam Pract 1999; 48:453–458.
- Newell S, Sanson-Fisher RW. Australian oncologists’ self-reported knowledge and attitudes about non-traditional therapies used by cancer patients. Med J Aust 2000; 172:110–113.
- Berman BM, Singh BK, Lao L, Singh BB, Ferentz KS, Hartnoll SM. Physicians’ attitudes toward complementary or alternative medicine: a regional survey. J Am Board Fam Pract 1995; 8:361–366.
- Verhoef MJ, Sutherland LR. General practitioners’ assessment of and interest in alternative medicine in Canada. Soc Sci Med 1995; 41:511–515.
- American Cancer Society: Treatment guidelines for patients. Version 1. http://www.cancer.org/downloads/CRI/NCCN_pain.pdf.
- Benedetti C, Brock C, Cleeland C, et al; National Comprehensive Cancer Network. NCCN Practice Guidelines for Cancer Pain. Oncology (Williston Park) 2000; 14:135–150.
- Bardia A, Barton DL, Prokop LJ, Bauer BA, Moynihan TJ. Efficacy of complementary and alternative medicine therapies in relieving cancer pain: a systematic review. J Clin Oncol 2006; 24:5457–5464.
- Lee H, Schmidt K, Ernst E. Acupuncture for the relief of cancer-related pain—a systematic review. Eur J Pain 2005; 9:437–444.
- Alimi D, Rubino C, Pichard-Léandri E, Fermand-Brulé S, Dubreuil-Lemaire ML, Hill C. Analgesic effect of auricular acupuncture for cancer pain: a randomized, blinded, controlled trial. J Clin Oncol 2003; 21:4120–4126.
- Spiegel D, Bloom JR. Group therapy and hypnosis reduce metastatic breast carcinoma pain. Psychosom Med 1983; 45:333–339.
- Goodwin PJ, Leszcz M, Ennis M, et al. The effect of group psychosocial support on survival in metastatic breast cancer. N Engl J Med 2001; 345:1719–1726.
- Syrjala KL, Donaldson GW, Davis MW, Kippes ME, Carr JE. Relaxation and imagery and cognitive-behavioral training reduce pain during cancer treatment: a controlled clinical trial. Pain 1995; 63:189–198.
- Sloman R, Brown P, Aldana E, Chee E. The use of relaxation for the promotion of comfort and pain relief in persons with advanced cancer. Contemp Nurse 1994; 3:6–12.
- Weinrich SP, Weinrich MC. The effect of massage on pain in cancer patients. Appl Nurs Res 1990; 3:140–145.
- Wilkie DJ, Kampbell J, Cutshall S, et al. Effects of massage on pain intensity, analgesics and quality of life in patients with cancer pain: a pilot study of a randomized clinical trial conducted within hospice care delivery. Hosp J 2000; 15:31–53.
- Soden K, Vincent K, Craske S, Lucas C, Ashley S. A randomized controlled trial of aromatherapy massage in a hospice setting. Palliat Med 2004; 18:87–92.
- Post-White J, Kinney ME, Savik K, Gau JB, Wilcox C, Lerner I. Therapeutic massage and healing touch improve symptoms in cancer. Integr Cancer Ther 2003; 2:332–344.
- Olson K, Hanson J, Michaud M. A phase II trial of Reiki for the management of pain in advanced cancer patients. J Pain Symptom Manage 2003; 26:990–997.
- Gallagher LM, Lagman R, Walsh D, Davis MP, Legrand SB. The clinical effects of music therapy in palliative medicine. Support Care Cancer 2006; 14:859–866.
- Olaku O, White JD. Herbal therapy use by cancer patients: a literature review on case reports. Eur J Cancer 2011; 47:508–514.
KEY POINTS
- Opioids can be used effectively for the management of cancer pain, provided the physician has sufficient knowledge, education, and training.
- Adjuvants, if properly used, can help manage cancer pain more effectively.
- Complementary and alternative therapies look promising, but too little is known about them, so caution is advised when recommending them.
- Patients should be referred to a pain clinic if they have intractable pain or if they have severe side effects from opioid therapy.
- Overall improvement in patient satisfaction and quality of life can be noted when pain is effectively managed.
Matters of Life and Death
This year, New York became the second state to enact a law requiring health care providers to provide information on end-of-life options to terminally ill patients. California was the first; the Right to Know End-of-Life Options Act has been in effect there since January 1, 2009. New York’s Palliative Care Information Act took effect on February 9, 2011.
What do these laws mean, to whom do they apply—and in a larger context, is it possible to legislate one of the most difficult and intimate conversations a health care provider can have with a patient?
It’s the Law, But What Does That Mean?
First, the facts about these laws:
California’s Right to Know End-of-Life Options Act states that “when a health care provider … makes a diagnosis that a patient has a terminal illness, the health care provider shall, upon the patient’s request, provide the patient with comprehensive information and counseling regarding legal end-of-life options … and provide for the referral or transfer of a patient ... if the patient’s health care provider does not wish to comply with the patient’s request for information on end-of-life options.”
The law defines a “health care provider” as an attending physician or surgeon. However, it is further stipulated that the phrase “also means a nurse practitioner or physician assistant practicing in accordance with standardized procedures or protocols.”
New York’s Palliative Care Information Act states that “If a patient is diagnosed with a terminal illness or condition, the patient’s attending health care practitioner shall offer to provide the patient with information and counseling regarding palliative care and end-of-life options appropriate to the patient, including … prognosis, risk and benefits of the various options; and the patient’s legal rights to comprehensive pain and symptom management.”
The state Department of Health further clarifies that physicians and NPs are subject to the requirements of the law. PAs and other health care professionals may also, consistent with their scope of practice, provide information about these services but are not required to do so.
It is unclear how much of a difference these laws will make. In California, there has not been a noticeable flurry of concern or activity in response to the law; some clinicians haven’t even registered its existence on their radar. In a state facing an ongoing and major fiscal crisis, enforcing this type of law probably is not going to be a top priority.
New York’s law was passed despite protests from the medical society, which felt that parties that would be directly impacted by the law were not sufficiently consulted on its import and implementation. In May, two physicians from Maimonides Medical Center in Brooklyn wrote in the New England Journal of Medicine that “Physicians and health care organizations are struggling to understand what the law requires.”
Whether such laws are enforceable or effective in and of themselves, they do serve another purpose. They remind us of a largely unmet need in patient care: the need for an open and honest discussion about the end of life.
Shattering Hospice Myths
Medicine is about healing. Someone becomes sick, and the clinician strives to make him or her well again. But there are devastating diseases about which we don’t know everything, and for many of them, a full-blown cure is not yet available. Even where life can be prolonged, death is never completely vanquished. Everyone knows that, but it is not easy to accept.
“Because most people die in old age, these issues get a lot of attention in the geriatric medicine arena—but this is across the lifespan,” says Freddi I. Segal-Gidan, PA, PhD, Co-Director of the Alzheimer’s Research Center of California at Rancho Los Amigos National Rehabilitation Center in Downey. “Many people spend a lot of money in their last months of life because we are a very interventionist medical society. And we still believe that we can cure and save everybody.”
“It’s true that we can always do something more,” says Karen Ayers, ACNP, who works with Hospice of Humboldt in California. “But is it futile? Is it going to work? Or is it just going to flog somebody? That’s really the question.”
Americans have already proven that they are not ready for a discussion of the financial aspects of treatment for terminal illnesses and other chronic, life-shortening conditions. (Recall the “death panels” propaganda?) But the larger issue is how individual patients would prefer to die, if “in my sleep, at a very old age” is not possible.
Some are inclined to fight until the last moment; if two rounds of chemo didn’t do the job, well, let’s try a third. Others would prefer to be as comfortable as possible for the remaining days/weeks/months of their lives. Most do not want to spend the end of their lives hooked up to machines in a hospital.
The question is whether patients know that they have choices and really understand what the options are. There are often misperceptions that when a clinician brings up end-of-life care, it is because he or she has “given up on” the patient.
“People think when you talk about end-of-life care that means ‘We’re going to give you morphine until you go into oblivion, and then you die,’” says Debbie Miller-Saultz, DNP, FNP-BC, of the Department of Pain Medicine and Palliative Care at Beth-Israel Medical Center in New York City. “And people think the morphine is causing someone to die faster. But all of those myths are really quelled when you talk to people who have palliative care, because they are offered services in a way that is timely and that provides much more support. People aren’t distressed or living in pain.”
By way of evidence, Miller-Saultz cites a study published last August in the New England Journal of Medicine (2010;363:733-742). Researchers at Massachusetts General Hospital randomized patients with metastatic non–small cell lung cancer to receive either early palliative care integrated with standard oncologic care or standard oncologic care alone.
Patients who received early palliative care showed significant improvements in quality of life and mood, compared to their counterparts. Furthermore, while fewer patients in the palliative care group underwent aggressive end-of-life care (eg, chemotherapy within 14 days of death), median survival was higher for them than for patients in the oncologic care–only group (11.6 months vs 8.9 months).
Temel and colleagues said their results “offer great promise for alleviating distress in patients with metastatic disease and addressing critical concerns regarding the use of health care services at the end of life.”
A Tough Talk to Have
Despite such positive findings, hospice remains an underutilized resource; the average length of stay is less than two weeks. “The reality is, the hospice literature shows that people get into hospice too late,” says Segal-Gidan.
In New York State, the average length of stay in hospice is 11 days. “People have end-of-life issues and disease longer than 11 days,” says Miller-Saultz. “So that’s a big part of the problem.”
Clearly, this is part of the impetus for legislative efforts such as those in California and New York. The implication of these laws is that, left to their own devices, clinicians are not providing terminally ill patients with essential information about the services available to them. Physicians in particular have often been criticized by advocates of palliative care and hospice for pushing aggressive treatments and dismissing the other options as “not real medicine.”
Nurses’ training may make them more receptive to alternatives. “We’re not schooled to ‘cure,’ that everything you do is driven to cure,” Miller-Saultz says. “You palliate. You do pain and symptom management. To me, it’s really no different from what we do anyway.”
That may change as medical schools try to incorporate more on end-of-life care into their curricula. But knowing you need to have these conversations—and in some cases being mandated to have them—does not make the discussion any easier.
Ayers considers herself fortunate to live and work in a relatively isolated area of California, centered around Eureka. The medical/nursing professionals are used to working together and do a lot of community outreach. When the local St. Joseph’s Hospital surveyed patients and providers, for example, they discovered that patients felt they weren’t getting information about the end of their lives, and clinicians said the hospital was not the right setting to deliver that information.
“It was just too late, it was too chaotic, and the patients were too upset,” Ayers explains. “It wasn’t the right time to be having that conversation.”
From this finding came the Physician Communication Initiative (it was named before the NPs got involved). The group held a series of dinners at a physician’s house “to talk about these issues, what is tough about it, how do you do it, how do you work it into the conversation,” Ayers says, “and how can we do it better.”
The Only Certainty Is …
While not everyone lives in that kind of community, Ayers suggests clinicians in other areas reach out to local medical and nursing groups, hospice services, or other community groups that may have an interest in educating people about what palliative care and end-of-life services are available locally. Major organizations also have resources on their Web sites that may help clinicians prepare for conversations with patients (see box).
A lot depends on context. Segal-Gidan works with patients who have Alzheimer’s disease, dementia, or other neurologic problems. “I’m able to bring up end-of-life care in the context of, say, ‘You’ve had a stroke, and you’re more likely to have another one. We hope not, but if so, let’s talk about what you would want to happen,’” she says. “When people are elderly and they have multiple chronic illnesses or a degenerative disease, people realize they’re coming to the end of their life. And in some ways, these discussions are more likely to happen.”
Timing is another question. The New York law, for example, requires that information on palliative and end-of-life options be provided to patients whose life expectancy does not exceed six months. Can that really be predicted with any certainty?
“We can identify the person with Alzheimer’s disease or multiple sclerosis or cancer,” Segal-Gidan says, “but we can’t identify very well when they’re six months before death. And then it may be too late.” This is another likely reason why so many people spend so little time in hospice programs before they die.
It may make sense to encourage patients to consider the choices they would make sooner rather than later. But even if a plan of care has been established or an advanced directive has been completed, nothing is set in stone. People can change their minds; death is an emotional issue and even those who are capable of frank discussion in the abstract may feel differently when it becomes reality.
In their editorial, the doctors from Maimonides Medical Center described an elderly patient with gastrointestinal and lung cancer for whom a satisfactory care plan had been developed—or so they thought. When the patient’s condition started to decline, his daughter found that she no longer agreed with the plan.
“She had previously accepted the fact of her father’s impending death as a theoretical matter,” the doctors wrote, “but when she came face to face with the troubling reality of his deteriorating condition, her thoughts and feelings had changed.”
How patients will respond to treatment, how long they will survive, and how they will choose to end their lives—there is so much uncertainty. Yet who might have pressing need for these services typically is not part of it. As Segal-Gidan said, it is possible to identify patients with terminal illnesses or degenerative conditions (although the diagnosis sometimes comes much too late in the process). But what of the 32- or 45-year-old who survives a car accident but has a traumatic brain injury and can no longer problem-solve, process, or plan? Who will make the decisions for that person? Did he or she ever discuss with a spouse or parent what he or she would want?
“It’s when death occurs at younger ages, more suddenly or unexpectedly, or even after the course of an illness, that I think it doesn’t really get a lot of attention,” Segal-Gidan says. “The grief gets a lot of attention, but all of the things around it—preparing for it, acknowledging it as a possibility—aren’t talked about.”
It isn’t possible to predict death and it certainly isn’t possible to avoid it forever. If there is one thing that frustrates humans, it is that we do not have all the answers.
This year, New York became the second state to enact a law requiring health care providers to provide information on end-of-life options to terminally ill patients. California was the first; the Right to Know End-of-Life Options Act has been in effect there since January 1, 2009. New York’s Palliative Care Information Act took effect on February 9, 2011.
What do these laws mean, to whom do they apply—and in a larger context, is it possible to legislate one of the most difficult and intimate conversations a health care provider can have with a patient?
It’s the Law, But What Does That Mean?
First, the facts about these laws:
California’s Right to Know End-of-Life Options Act states that “when a health care provider … makes a diagnosis that a patient has a terminal illness, the health care provider shall, upon the patient’s request, provide the patient with comprehensive information and counseling regarding legal end-of-life options … and provide for the referral or transfer of a patient ... if the patient’s health care provider does not wish to comply with the patient’s request for information on end-of-life options.”
The law defines a “health care provider” as an attending physician or surgeon. However, it is further stipulated that the phrase “also means a nurse practitioner or physician assistant practicing in accordance with standardized procedures or protocols.”
New York’s Palliative Care Information Act states that “If a patient is diagnosed with a terminal illness or condition, the patient’s attending health care practitioner shall offer to provide the patient with information and counseling regarding palliative care and end-of-life options appropriate to the patient, including … prognosis, risk and benefits of the various options; and the patient’s legal rights to comprehensive pain and symptom management.”
The state Department of Health further clarifies that physicians and NPs are subject to the requirements of the law. PAs and other health care professionals may also, consistent with their scope of practice, provide information about these services but are not required to do so.
It is unclear how much of a difference these laws will make. In California, there has not been a noticeable flurry of concern or activity in response to the law; some clinicians haven’t even registered its existence on their radar. In a state facing an ongoing and major fiscal crisis, enforcing this type of law probably is not going to be a top priority.
New York’s law was passed despite protests from the medical society, which felt that parties that would be directly impacted by the law were not sufficiently consulted on its import and implementation. In May, two physicians from Maimonides Medical Center in Brooklyn wrote in the New England Journal of Medicine that “Physicians and health care organizations are struggling to understand what the law requires.”
Whether such laws are enforceable or effective in and of themselves, they do serve another purpose. They remind us of a largely unmet need in patient care: the need for an open and honest discussion about the end of life.
Shattering Hospice Myths
Medicine is about healing. Someone becomes sick, and the clinician strives to make him or her well again. But there are devastating diseases about which we don’t know everything, and for many of them, a full-blown cure is not yet available. Even where life can be prolonged, death is never completely vanquished. Everyone knows that, but it is not easy to accept.
“Because most people die in old age, these issues get a lot of attention in the geriatric medicine arena—but this is across the lifespan,” says Freddi I. Segal-Gidan, PA, PhD, Co-Director of the Alzheimer’s Research Center of California at Rancho Los Amigos National Rehabilitation Center in Downey. “Many people spend a lot of money in their last months of life because we are a very interventionist medical society. And we still believe that we can cure and save everybody.”
“It’s true that we can always do something more,” says Karen Ayers, ACNP, who works with Hospice of Humboldt in California. “But is it futile? Is it going to work? Or is it just going to flog somebody? That’s really the question.”
Americans have already proven that they are not ready for a discussion of the financial aspects of treatment for terminal illnesses and other chronic, life-shortening conditions. (Recall the “death panels” propaganda?) But the larger issue is how individual patients would prefer to die, if “in my sleep, at a very old age” is not possible.
Some are inclined to fight until the last moment; if two rounds of chemo didn’t do the job, well, let’s try a third. Others would prefer to be as comfortable as possible for the remaining days/weeks/months of their lives. Most do not want to spend the end of their lives hooked up to machines in a hospital.
The question is whether patients know that they have choices and really understand what the options are. There are often misperceptions that when a clinician brings up end-of-life care, it is because he or she has “given up on” the patient.
“People think when you talk about end-of-life care that means ‘We’re going to give you morphine until you go into oblivion, and then you die,’” says Debbie Miller-Saultz, DNP, FNP-BC, of the Department of Pain Medicine and Palliative Care at Beth-Israel Medical Center in New York City. “And people think the morphine is causing someone to die faster. But all of those myths are really quelled when you talk to people who have palliative care, because they are offered services in a way that is timely and that provides much more support. People aren’t distressed or living in pain.”
By way of evidence, Miller-Saultz cites a study published last August in the New England Journal of Medicine (2010;363:733-742). Researchers at Massachusetts General Hospital randomized patients with metastatic non–small cell lung cancer to receive either early palliative care integrated with standard oncologic care or standard oncologic care alone.
Patients who received early palliative care showed significant improvements in quality of life and mood, compared to their counterparts. Furthermore, while fewer patients in the palliative care group underwent aggressive end-of-life care (eg, chemotherapy within 14 days of death), median survival was higher for them than for patients in the oncologic care–only group (11.6 months vs 8.9 months).
Temel and colleagues said their results “offer great promise for alleviating distress in patients with metastatic disease and addressing critical concerns regarding the use of health care services at the end of life.”
A Tough Talk to Have
Despite such positive findings, hospice remains an underutilized resource; the average length of stay is less than two weeks. “The reality is, the hospice literature shows that people get into hospice too late,” says Segal-Gidan.
In New York State, the average length of stay in hospice is 11 days. “People have end-of-life issues and disease longer than 11 days,” says Miller-Saultz. “So that’s a big part of the problem.”
Clearly, this is part of the impetus for legislative efforts such as those in California and New York. The implication of these laws is that, left to their own devices, clinicians are not providing terminally ill patients with essential information about the services available to them. Physicians in particular have often been criticized by advocates of palliative care and hospice for pushing aggressive treatments and dismissing the other options as “not real medicine.”
Nurses’ training may make them more receptive to alternatives. “We’re not schooled to ‘cure,’ that everything you do is driven to cure,” Miller-Saultz says. “You palliate. You do pain and symptom management. To me, it’s really no different from what we do anyway.”
That may change as medical schools try to incorporate more on end-of-life care into their curricula. But knowing you need to have these conversations—and in some cases being mandated to have them—does not make the discussion any easier.
Ayers considers herself fortunate to live and work in a relatively isolated area of California, centered around Eureka. The medical/nursing professionals are used to working together and do a lot of community outreach. When the local St. Joseph’s Hospital surveyed patients and providers, for example, they discovered that patients felt they weren’t getting information about the end of their lives, and clinicians said the hospital was not the right setting to deliver that information.
“It was just too late, it was too chaotic, and the patients were too upset,” Ayers explains. “It wasn’t the right time to be having that conversation.”
From this finding came the Physician Communication Initiative (it was named before the NPs got involved). The group held a series of dinners at a physician’s house “to talk about these issues, what is tough about it, how do you do it, how do you work it into the conversation,” Ayers says, “and how can we do it better.”
The Only Certainty Is …
While not everyone lives in that kind of community, Ayers suggests clinicians in other areas reach out to local medical and nursing groups, hospice services, or other community groups that may have an interest in educating people about what palliative care and end-of-life services are available locally. Major organizations also have resources on their Web sites that may help clinicians prepare for conversations with patients (see box).
A lot depends on context. Segal-Gidan works with patients who have Alzheimer’s disease, dementia, or other neurologic problems. “I’m able to bring up end-of-life care in the context of, say, ‘You’ve had a stroke, and you’re more likely to have another one. We hope not, but if so, let’s talk about what you would want to happen,’” she says. “When people are elderly and they have multiple chronic illnesses or a degenerative disease, people realize they’re coming to the end of their life. And in some ways, these discussions are more likely to happen.”
Timing is another question. The New York law, for example, requires that information on palliative and end-of-life options be provided to patients whose life expectancy does not exceed six months. Can that really be predicted with any certainty?
“We can identify the person with Alzheimer’s disease or multiple sclerosis or cancer,” Segal-Gidan says, “but we can’t identify very well when they’re six months before death. And then it may be too late.” This is another likely reason why so many people spend so little time in hospice programs before they die.
It may make sense to encourage patients to consider the choices they would make sooner rather than later. But even if a plan of care has been established or an advanced directive has been completed, nothing is set in stone. People can change their minds; death is an emotional issue and even those who are capable of frank discussion in the abstract may feel differently when it becomes reality.
In their editorial, the doctors from Maimonides Medical Center described an elderly patient with gastrointestinal and lung cancer for whom a satisfactory care plan had been developed—or so they thought. When the patient’s condition started to decline, his daughter found that she no longer agreed with the plan.
“She had previously accepted the fact of her father’s impending death as a theoretical matter,” the doctors wrote, “but when she came face to face with the troubling reality of his deteriorating condition, her thoughts and feelings had changed.”
How patients will respond to treatment, how long they will survive, and how they will choose to end their lives—there is so much uncertainty. Yet who might have pressing need for these services typically is not part of it. As Segal-Gidan said, it is possible to identify patients with terminal illnesses or degenerative conditions (although the diagnosis sometimes comes much too late in the process). But what of the 32- or 45-year-old who survives a car accident but has a traumatic brain injury and can no longer problem-solve, process, or plan? Who will make the decisions for that person? Did he or she ever discuss with a spouse or parent what he or she would want?
“It’s when death occurs at younger ages, more suddenly or unexpectedly, or even after the course of an illness, that I think it doesn’t really get a lot of attention,” Segal-Gidan says. “The grief gets a lot of attention, but all of the things around it—preparing for it, acknowledging it as a possibility—aren’t talked about.”
It isn’t possible to predict death and it certainly isn’t possible to avoid it forever. If there is one thing that frustrates humans, it is that we do not have all the answers.
This year, New York became the second state to enact a law requiring health care providers to provide information on end-of-life options to terminally ill patients. California was the first; the Right to Know End-of-Life Options Act has been in effect there since January 1, 2009. New York’s Palliative Care Information Act took effect on February 9, 2011.
What do these laws mean, to whom do they apply—and in a larger context, is it possible to legislate one of the most difficult and intimate conversations a health care provider can have with a patient?
It’s the Law, But What Does That Mean?
First, the facts about these laws:
California’s Right to Know End-of-Life Options Act states that “when a health care provider … makes a diagnosis that a patient has a terminal illness, the health care provider shall, upon the patient’s request, provide the patient with comprehensive information and counseling regarding legal end-of-life options … and provide for the referral or transfer of a patient ... if the patient’s health care provider does not wish to comply with the patient’s request for information on end-of-life options.”
The law defines a “health care provider” as an attending physician or surgeon. However, it is further stipulated that the phrase “also means a nurse practitioner or physician assistant practicing in accordance with standardized procedures or protocols.”
New York’s Palliative Care Information Act states that “If a patient is diagnosed with a terminal illness or condition, the patient’s attending health care practitioner shall offer to provide the patient with information and counseling regarding palliative care and end-of-life options appropriate to the patient, including … prognosis, risk and benefits of the various options; and the patient’s legal rights to comprehensive pain and symptom management.”
The state Department of Health further clarifies that physicians and NPs are subject to the requirements of the law. PAs and other health care professionals may also, consistent with their scope of practice, provide information about these services but are not required to do so.
It is unclear how much of a difference these laws will make. In California, there has not been a noticeable flurry of concern or activity in response to the law; some clinicians haven’t even registered its existence on their radar. In a state facing an ongoing and major fiscal crisis, enforcing this type of law probably is not going to be a top priority.
New York’s law was passed despite protests from the medical society, which felt that parties that would be directly impacted by the law were not sufficiently consulted on its import and implementation. In May, two physicians from Maimonides Medical Center in Brooklyn wrote in the New England Journal of Medicine that “Physicians and health care organizations are struggling to understand what the law requires.”
Whether such laws are enforceable or effective in and of themselves, they do serve another purpose. They remind us of a largely unmet need in patient care: the need for an open and honest discussion about the end of life.
Shattering Hospice Myths
Medicine is about healing. Someone becomes sick, and the clinician strives to make him or her well again. But there are devastating diseases about which we don’t know everything, and for many of them, a full-blown cure is not yet available. Even where life can be prolonged, death is never completely vanquished. Everyone knows that, but it is not easy to accept.
“Because most people die in old age, these issues get a lot of attention in the geriatric medicine arena—but this is across the lifespan,” says Freddi I. Segal-Gidan, PA, PhD, Co-Director of the Alzheimer’s Research Center of California at Rancho Los Amigos National Rehabilitation Center in Downey. “Many people spend a lot of money in their last months of life because we are a very interventionist medical society. And we still believe that we can cure and save everybody.”
“It’s true that we can always do something more,” says Karen Ayers, ACNP, who works with Hospice of Humboldt in California. “But is it futile? Is it going to work? Or is it just going to flog somebody? That’s really the question.”
Americans have already proven that they are not ready for a discussion of the financial aspects of treatment for terminal illnesses and other chronic, life-shortening conditions. (Recall the “death panels” propaganda?) But the larger issue is how individual patients would prefer to die, if “in my sleep, at a very old age” is not possible.
Some are inclined to fight until the last moment; if two rounds of chemo didn’t do the job, well, let’s try a third. Others would prefer to be as comfortable as possible for the remaining days/weeks/months of their lives. Most do not want to spend the end of their lives hooked up to machines in a hospital.
The question is whether patients know that they have choices and really understand what the options are. There are often misperceptions that when a clinician brings up end-of-life care, it is because he or she has “given up on” the patient.
“People think when you talk about end-of-life care that means ‘We’re going to give you morphine until you go into oblivion, and then you die,’” says Debbie Miller-Saultz, DNP, FNP-BC, of the Department of Pain Medicine and Palliative Care at Beth-Israel Medical Center in New York City. “And people think the morphine is causing someone to die faster. But all of those myths are really quelled when you talk to people who have palliative care, because they are offered services in a way that is timely and that provides much more support. People aren’t distressed or living in pain.”
By way of evidence, Miller-Saultz cites a study published last August in the New England Journal of Medicine (2010;363:733-742). Researchers at Massachusetts General Hospital randomized patients with metastatic non–small cell lung cancer to receive either early palliative care integrated with standard oncologic care or standard oncologic care alone.
Patients who received early palliative care showed significant improvements in quality of life and mood, compared to their counterparts. Furthermore, while fewer patients in the palliative care group underwent aggressive end-of-life care (eg, chemotherapy within 14 days of death), median survival was higher for them than for patients in the oncologic care–only group (11.6 months vs 8.9 months).
Temel and colleagues said their results “offer great promise for alleviating distress in patients with metastatic disease and addressing critical concerns regarding the use of health care services at the end of life.”
A Tough Talk to Have
Despite such positive findings, hospice remains an underutilized resource; the average length of stay is less than two weeks. “The reality is, the hospice literature shows that people get into hospice too late,” says Segal-Gidan.
In New York State, the average length of stay in hospice is 11 days. “People have end-of-life issues and disease longer than 11 days,” says Miller-Saultz. “So that’s a big part of the problem.”
Clearly, this is part of the impetus for legislative efforts such as those in California and New York. The implication of these laws is that, left to their own devices, clinicians are not providing terminally ill patients with essential information about the services available to them. Physicians in particular have often been criticized by advocates of palliative care and hospice for pushing aggressive treatments and dismissing the other options as “not real medicine.”
Nurses’ training may make them more receptive to alternatives. “We’re not schooled to ‘cure,’ that everything you do is driven to cure,” Miller-Saultz says. “You palliate. You do pain and symptom management. To me, it’s really no different from what we do anyway.”
That may change as medical schools try to incorporate more on end-of-life care into their curricula. But knowing you need to have these conversations—and in some cases being mandated to have them—does not make the discussion any easier.
Ayers considers herself fortunate to live and work in a relatively isolated area of California, centered around Eureka. The medical/nursing professionals are used to working together and do a lot of community outreach. When the local St. Joseph’s Hospital surveyed patients and providers, for example, they discovered that patients felt they weren’t getting information about the end of their lives, and clinicians said the hospital was not the right setting to deliver that information.
“It was just too late, it was too chaotic, and the patients were too upset,” Ayers explains. “It wasn’t the right time to be having that conversation.”
From this finding came the Physician Communication Initiative (it was named before the NPs got involved). The group held a series of dinners at a physician’s house “to talk about these issues, what is tough about it, how do you do it, how do you work it into the conversation,” Ayers says, “and how can we do it better.”
The Only Certainty Is …
While not everyone lives in that kind of community, Ayers suggests clinicians in other areas reach out to local medical and nursing groups, hospice services, or other community groups that may have an interest in educating people about what palliative care and end-of-life services are available locally. Major organizations also have resources on their Web sites that may help clinicians prepare for conversations with patients (see box).
A lot depends on context. Segal-Gidan works with patients who have Alzheimer’s disease, dementia, or other neurologic problems. “I’m able to bring up end-of-life care in the context of, say, ‘You’ve had a stroke, and you’re more likely to have another one. We hope not, but if so, let’s talk about what you would want to happen,’” she says. “When people are elderly and they have multiple chronic illnesses or a degenerative disease, people realize they’re coming to the end of their life. And in some ways, these discussions are more likely to happen.”
Timing is another question. The New York law, for example, requires that information on palliative and end-of-life options be provided to patients whose life expectancy does not exceed six months. Can that really be predicted with any certainty?
“We can identify the person with Alzheimer’s disease or multiple sclerosis or cancer,” Segal-Gidan says, “but we can’t identify very well when they’re six months before death. And then it may be too late.” This is another likely reason why so many people spend so little time in hospice programs before they die.
It may make sense to encourage patients to consider the choices they would make sooner rather than later. But even if a plan of care has been established or an advanced directive has been completed, nothing is set in stone. People can change their minds; death is an emotional issue and even those who are capable of frank discussion in the abstract may feel differently when it becomes reality.
In their editorial, the doctors from Maimonides Medical Center described an elderly patient with gastrointestinal and lung cancer for whom a satisfactory care plan had been developed—or so they thought. When the patient’s condition started to decline, his daughter found that she no longer agreed with the plan.
“She had previously accepted the fact of her father’s impending death as a theoretical matter,” the doctors wrote, “but when she came face to face with the troubling reality of his deteriorating condition, her thoughts and feelings had changed.”
How patients will respond to treatment, how long they will survive, and how they will choose to end their lives—there is so much uncertainty. Yet who might have pressing need for these services typically is not part of it. As Segal-Gidan said, it is possible to identify patients with terminal illnesses or degenerative conditions (although the diagnosis sometimes comes much too late in the process). But what of the 32- or 45-year-old who survives a car accident but has a traumatic brain injury and can no longer problem-solve, process, or plan? Who will make the decisions for that person? Did he or she ever discuss with a spouse or parent what he or she would want?
“It’s when death occurs at younger ages, more suddenly or unexpectedly, or even after the course of an illness, that I think it doesn’t really get a lot of attention,” Segal-Gidan says. “The grief gets a lot of attention, but all of the things around it—preparing for it, acknowledging it as a possibility—aren’t talked about.”
It isn’t possible to predict death and it certainly isn’t possible to avoid it forever. If there is one thing that frustrates humans, it is that we do not have all the answers.
Thirteen Steps Advised to Prevent Delirium in Hospitalized Patients
Delirium is common but underrecognized in hospitalized patients, "a neglected condition relative to its frequency and serious consequences," and approximately one-third of cases are preventable, according to a report in the June 7 Annals of Internal Medicine.
The United Kingdom’s National Institute for Health and Clinical Excellence (NICE) has released a new clinical guideline for preventing delirium, which lists 13 recommendations that "could probably be easily accommodated in current care without incurring high costs." On the contrary, preventing delirium in hospitalized patients is expected to markedly cut health care costs, as well as to improve quality-adjusted life-year gains, compared with usual care, said Rachel O’Mahony, Ph.D., of the National Clinical Guideline Centre at the Royal College of Physicians, London, and her associates.
A multidisciplinary group of experts, including physicians, psychiatrists, specialist nurses, a home care manager, and patient representatives, reviewed the literature to find which prevention strategies were effective to compile the guidelines.
No single intervention was identified that significantly reduced hospital stay, placement in long-term care facilities, mortality, or duration or severity of delirium. Instead, multicomponent interventions provided the strongest evidence of improving these factors.
The recommendations include:
- Avoid changes in patient surroundings to prevent confusion and disorientation. This includes avoiding unnecessary room changes as well as changes in the personnel who provide care.
"Several moves within an acute care hospital are now common ... [from] emergency department to assessment unit to acute care ward and sometimes to post-acute care ward.
"Moving could make it difficult for a sick person on the brink of a delirium episode to maintain his or her orientation and contact with reality," Dr. O’Mahony and her colleagues said (Ann. Intern. Med. 2011;154:746-51).
- Provide appropriate lighting, clear signage, an easily visible 24-hour clock (to distinguish day from night in rooms without windows), and a calendar to help patients stay oriented to time and place.
- Reorient patients by explaining where they are and what your role is.
- Provide cognitively stimulating activities, such as encouraging patients to reminisce and facilitating visits from family and friends.
- Address dehydration and constipation, with intravenous fluids, if necessary, and manage fluid balance in patients with relevant comorbidities such as heart failure or kidney disease.
- Assess for hypoxia and optimize oxygen saturation.
- Actively assess for infection and treat it; employ infection-control procedures; and avoid unnecessary catheterization.
The presence of a bladder catheter is a known risk factor for delirium, the researchers noted.
- Address immobility by encouraging patients to walk as soon as possible, providing appropriate walking aids and ensuring they are available at all times, and encouraging range-of-motion exercises.
- Assess for pain, attending to nonverbal signs of pain, and manage it.
Although some clinicians are leery of inducing confusion by providing painkillers, pain itself is an independent risk factor for delirium, the investigators said.
- Review both the type and the number of medications.
- Address poor nutrition, and make sure that dentures fit properly in patients who have them.
- Address sensory impairment and resolve any reversible causes such as impacted ear wax or need for visual or hearing aids. Ensure such aids are in good working order.
- Promote good sleep patterns by avoiding procedures and minimizing ambient noise during sleeping hours.
"Some of these components are provided to some patients some of the time, but prevention of delirium requires that we do all of these things all the time to all of the patients who are at risk," Dr. O’Mahony and her associates said.
"It makes sense" to target these interventions to patients at highest risk of developing delirium. There are four such easy-to-identify groups, each with a greater than fivefold increase in risk for delirium: patients aged 65 [years] and older, patients with preexisting cognitive impairment, patients with severe illness, and patients with hip fracture, they noted.
"This enhanced approach goes beyond well-trained and prepared staff. It requires a health care system ... that supports comprehensive and reliable delivery of specific tasks," they researchers added.
The guideline was supported by the National Clinical Guideline Centre at the Royal College of Physicians; the U.K. Cochrane Centre and KSG-Trans; and the Bradford Institute for Health Research. None of the authors reported having any relevant conflicts of interest.
The full version of the guideline, including details about the methods used in its development, is available online. A synopsis is available at Annals of Internal Medicine.
Clinical guidelines should bring order to the dizzying array of diagnostic and therapeutic options facing physicians, but too often, the thousands of such guidelines published by "hundreds of groups in dozens of countries" instead can make the "chaos" even worse, according to Dr. Christine Laine, Dr. Darren B. Taichman, and Dr. Cynthia Mulrow.
The American College of Physicians has always presented summaries of the high-quality guidelines compiled by themselves and by the U.S. Preventive Services Task Force, and the organization now seeks "to publish thoughtful synopses of guidelines initially released in other venues but whose topics are highly relevant to the practice of internal medicine and its subspecialties.
"As the first example of this new venture," the ACP presents a synopsis of the NICE guideline for preventing delirium, because the methods for developing it came so close to meeting the Institute of Medicine’s "rigorous definition of trustworthy."
"We urge readers to gain familiarity with the IOM standards and consider them as they use guidelines to navigate the maze of available clinical choices," the editors noted.
Christine Laine, M.D., is editor in chief of the Annals of Internal Medicine, senior vice president of the American College of Physicians, and an internist at Jefferson Medical College, all in Philadelphia. Darren B. Taichman, M.D., Ph.D., is executive deputy editor of the Annals and director of the medical ICU at the University of Pennsylvania Medical Center, Philadelphia. Cynthia Mulrow, M.D., is senior deputy editor of the Annals. These remarks were taken from their editorial accompanying the NICE guideline (Ann. Intern. Med. 2011;154:774-5). No potential conflicts of interest were disclosed.
Clinical guidelines should bring order to the dizzying array of diagnostic and therapeutic options facing physicians, but too often, the thousands of such guidelines published by "hundreds of groups in dozens of countries" instead can make the "chaos" even worse, according to Dr. Christine Laine, Dr. Darren B. Taichman, and Dr. Cynthia Mulrow.
The American College of Physicians has always presented summaries of the high-quality guidelines compiled by themselves and by the U.S. Preventive Services Task Force, and the organization now seeks "to publish thoughtful synopses of guidelines initially released in other venues but whose topics are highly relevant to the practice of internal medicine and its subspecialties.
"As the first example of this new venture," the ACP presents a synopsis of the NICE guideline for preventing delirium, because the methods for developing it came so close to meeting the Institute of Medicine’s "rigorous definition of trustworthy."
"We urge readers to gain familiarity with the IOM standards and consider them as they use guidelines to navigate the maze of available clinical choices," the editors noted.
Christine Laine, M.D., is editor in chief of the Annals of Internal Medicine, senior vice president of the American College of Physicians, and an internist at Jefferson Medical College, all in Philadelphia. Darren B. Taichman, M.D., Ph.D., is executive deputy editor of the Annals and director of the medical ICU at the University of Pennsylvania Medical Center, Philadelphia. Cynthia Mulrow, M.D., is senior deputy editor of the Annals. These remarks were taken from their editorial accompanying the NICE guideline (Ann. Intern. Med. 2011;154:774-5). No potential conflicts of interest were disclosed.
Clinical guidelines should bring order to the dizzying array of diagnostic and therapeutic options facing physicians, but too often, the thousands of such guidelines published by "hundreds of groups in dozens of countries" instead can make the "chaos" even worse, according to Dr. Christine Laine, Dr. Darren B. Taichman, and Dr. Cynthia Mulrow.
The American College of Physicians has always presented summaries of the high-quality guidelines compiled by themselves and by the U.S. Preventive Services Task Force, and the organization now seeks "to publish thoughtful synopses of guidelines initially released in other venues but whose topics are highly relevant to the practice of internal medicine and its subspecialties.
"As the first example of this new venture," the ACP presents a synopsis of the NICE guideline for preventing delirium, because the methods for developing it came so close to meeting the Institute of Medicine’s "rigorous definition of trustworthy."
"We urge readers to gain familiarity with the IOM standards and consider them as they use guidelines to navigate the maze of available clinical choices," the editors noted.
Christine Laine, M.D., is editor in chief of the Annals of Internal Medicine, senior vice president of the American College of Physicians, and an internist at Jefferson Medical College, all in Philadelphia. Darren B. Taichman, M.D., Ph.D., is executive deputy editor of the Annals and director of the medical ICU at the University of Pennsylvania Medical Center, Philadelphia. Cynthia Mulrow, M.D., is senior deputy editor of the Annals. These remarks were taken from their editorial accompanying the NICE guideline (Ann. Intern. Med. 2011;154:774-5). No potential conflicts of interest were disclosed.
Delirium is common but underrecognized in hospitalized patients, "a neglected condition relative to its frequency and serious consequences," and approximately one-third of cases are preventable, according to a report in the June 7 Annals of Internal Medicine.
The United Kingdom’s National Institute for Health and Clinical Excellence (NICE) has released a new clinical guideline for preventing delirium, which lists 13 recommendations that "could probably be easily accommodated in current care without incurring high costs." On the contrary, preventing delirium in hospitalized patients is expected to markedly cut health care costs, as well as to improve quality-adjusted life-year gains, compared with usual care, said Rachel O’Mahony, Ph.D., of the National Clinical Guideline Centre at the Royal College of Physicians, London, and her associates.
A multidisciplinary group of experts, including physicians, psychiatrists, specialist nurses, a home care manager, and patient representatives, reviewed the literature to find which prevention strategies were effective to compile the guidelines.
No single intervention was identified that significantly reduced hospital stay, placement in long-term care facilities, mortality, or duration or severity of delirium. Instead, multicomponent interventions provided the strongest evidence of improving these factors.
The recommendations include:
- Avoid changes in patient surroundings to prevent confusion and disorientation. This includes avoiding unnecessary room changes as well as changes in the personnel who provide care.
"Several moves within an acute care hospital are now common ... [from] emergency department to assessment unit to acute care ward and sometimes to post-acute care ward.
"Moving could make it difficult for a sick person on the brink of a delirium episode to maintain his or her orientation and contact with reality," Dr. O’Mahony and her colleagues said (Ann. Intern. Med. 2011;154:746-51).
- Provide appropriate lighting, clear signage, an easily visible 24-hour clock (to distinguish day from night in rooms without windows), and a calendar to help patients stay oriented to time and place.
- Reorient patients by explaining where they are and what your role is.
- Provide cognitively stimulating activities, such as encouraging patients to reminisce and facilitating visits from family and friends.
- Address dehydration and constipation, with intravenous fluids, if necessary, and manage fluid balance in patients with relevant comorbidities such as heart failure or kidney disease.
- Assess for hypoxia and optimize oxygen saturation.
- Actively assess for infection and treat it; employ infection-control procedures; and avoid unnecessary catheterization.
The presence of a bladder catheter is a known risk factor for delirium, the researchers noted.
- Address immobility by encouraging patients to walk as soon as possible, providing appropriate walking aids and ensuring they are available at all times, and encouraging range-of-motion exercises.
- Assess for pain, attending to nonverbal signs of pain, and manage it.
Although some clinicians are leery of inducing confusion by providing painkillers, pain itself is an independent risk factor for delirium, the investigators said.
- Review both the type and the number of medications.
- Address poor nutrition, and make sure that dentures fit properly in patients who have them.
- Address sensory impairment and resolve any reversible causes such as impacted ear wax or need for visual or hearing aids. Ensure such aids are in good working order.
- Promote good sleep patterns by avoiding procedures and minimizing ambient noise during sleeping hours.
"Some of these components are provided to some patients some of the time, but prevention of delirium requires that we do all of these things all the time to all of the patients who are at risk," Dr. O’Mahony and her associates said.
"It makes sense" to target these interventions to patients at highest risk of developing delirium. There are four such easy-to-identify groups, each with a greater than fivefold increase in risk for delirium: patients aged 65 [years] and older, patients with preexisting cognitive impairment, patients with severe illness, and patients with hip fracture, they noted.
"This enhanced approach goes beyond well-trained and prepared staff. It requires a health care system ... that supports comprehensive and reliable delivery of specific tasks," they researchers added.
The guideline was supported by the National Clinical Guideline Centre at the Royal College of Physicians; the U.K. Cochrane Centre and KSG-Trans; and the Bradford Institute for Health Research. None of the authors reported having any relevant conflicts of interest.
The full version of the guideline, including details about the methods used in its development, is available online. A synopsis is available at Annals of Internal Medicine.
Delirium is common but underrecognized in hospitalized patients, "a neglected condition relative to its frequency and serious consequences," and approximately one-third of cases are preventable, according to a report in the June 7 Annals of Internal Medicine.
The United Kingdom’s National Institute for Health and Clinical Excellence (NICE) has released a new clinical guideline for preventing delirium, which lists 13 recommendations that "could probably be easily accommodated in current care without incurring high costs." On the contrary, preventing delirium in hospitalized patients is expected to markedly cut health care costs, as well as to improve quality-adjusted life-year gains, compared with usual care, said Rachel O’Mahony, Ph.D., of the National Clinical Guideline Centre at the Royal College of Physicians, London, and her associates.
A multidisciplinary group of experts, including physicians, psychiatrists, specialist nurses, a home care manager, and patient representatives, reviewed the literature to find which prevention strategies were effective to compile the guidelines.
No single intervention was identified that significantly reduced hospital stay, placement in long-term care facilities, mortality, or duration or severity of delirium. Instead, multicomponent interventions provided the strongest evidence of improving these factors.
The recommendations include:
- Avoid changes in patient surroundings to prevent confusion and disorientation. This includes avoiding unnecessary room changes as well as changes in the personnel who provide care.
"Several moves within an acute care hospital are now common ... [from] emergency department to assessment unit to acute care ward and sometimes to post-acute care ward.
"Moving could make it difficult for a sick person on the brink of a delirium episode to maintain his or her orientation and contact with reality," Dr. O’Mahony and her colleagues said (Ann. Intern. Med. 2011;154:746-51).
- Provide appropriate lighting, clear signage, an easily visible 24-hour clock (to distinguish day from night in rooms without windows), and a calendar to help patients stay oriented to time and place.
- Reorient patients by explaining where they are and what your role is.
- Provide cognitively stimulating activities, such as encouraging patients to reminisce and facilitating visits from family and friends.
- Address dehydration and constipation, with intravenous fluids, if necessary, and manage fluid balance in patients with relevant comorbidities such as heart failure or kidney disease.
- Assess for hypoxia and optimize oxygen saturation.
- Actively assess for infection and treat it; employ infection-control procedures; and avoid unnecessary catheterization.
The presence of a bladder catheter is a known risk factor for delirium, the researchers noted.
- Address immobility by encouraging patients to walk as soon as possible, providing appropriate walking aids and ensuring they are available at all times, and encouraging range-of-motion exercises.
- Assess for pain, attending to nonverbal signs of pain, and manage it.
Although some clinicians are leery of inducing confusion by providing painkillers, pain itself is an independent risk factor for delirium, the investigators said.
- Review both the type and the number of medications.
- Address poor nutrition, and make sure that dentures fit properly in patients who have them.
- Address sensory impairment and resolve any reversible causes such as impacted ear wax or need for visual or hearing aids. Ensure such aids are in good working order.
- Promote good sleep patterns by avoiding procedures and minimizing ambient noise during sleeping hours.
"Some of these components are provided to some patients some of the time, but prevention of delirium requires that we do all of these things all the time to all of the patients who are at risk," Dr. O’Mahony and her associates said.
"It makes sense" to target these interventions to patients at highest risk of developing delirium. There are four such easy-to-identify groups, each with a greater than fivefold increase in risk for delirium: patients aged 65 [years] and older, patients with preexisting cognitive impairment, patients with severe illness, and patients with hip fracture, they noted.
"This enhanced approach goes beyond well-trained and prepared staff. It requires a health care system ... that supports comprehensive and reliable delivery of specific tasks," they researchers added.
The guideline was supported by the National Clinical Guideline Centre at the Royal College of Physicians; the U.K. Cochrane Centre and KSG-Trans; and the Bradford Institute for Health Research. None of the authors reported having any relevant conflicts of interest.
The full version of the guideline, including details about the methods used in its development, is available online. A synopsis is available at Annals of Internal Medicine.
FROM THE ANNALS OF INTERNAL MEDICINE
Telling Patients Bad News Takes Practice, Skill, and Compassion
TAMPA – Sharing bad news with patients might not be easy, but it’s a skill physicians can learn and as important as knowing how to ready an EKG or an x-ray, James A. Avery, MD, CMD, said.
"What I am proposing is that giving bad news well is a fundamental long-term care physician skill, and competence in this area is critical," Dr. Avery said at this year’s AMDA – Dedicated to Long Term Medicine annual meeting.
"Giving bad news ... takes desire, courage, and practice," said Dr. Avery. "Patients deserve to get bad news delivered with compassion, hope, and integrity."
Plan ahead for the conversation; start with what the patient knows and wants to know; and develop a compassionate tone, said Dr. Avery, chief medical officer at Golden Living in Washington, a corporation that focuses on skilled nursing, assisted living, and rehabilitation therapy. Also, always provide an appropriate prognosis. "It’s your obligation to bring this up. Patients and families may be afraid to ask."
What can happen if the conversation is not done correctly? "If bad news is given poorly, it can rob hope and create distress, confusion, and anxiety. It can weaken the patient’s faith and set off a chain of events that adversely affects the survivors for years," said Dr. Avery.
"I was particularly bad at giving bad news at first," he said. A pulmonologist by training, he also worked for years in hospice care in both Florida and New York. He spoke with patients who transitioned to hospice from Memorial Sloan-Kettering Cancer Center, for example.
"I learned quickly that if I was going to give bad news, not to schedule the patient for midmorning on a Monday. It is too chaotic," Dr. Avery said. Schedule the patient for the first appointment after lunch or at the end of the day. Allow sufficient time and create a comfortable, private place with tissues available, he added.
Next, determine where each patient is in terms of understanding his or her illness. "Explore and ask," Dr. Avery said. Good questions include:
- Is there anyone else you want to have in the conversation?
- How do you understand what has happened to you medically?
- What have doctors told you about this illness?
- What do you think caused this illness?
"I cannot tell you how many patients with colon cancer thought they had it because they took too many antacids," Dr. Avery said. "Also, I had one woman with breast cancer who responded ‘Burger King.’ She had read an article that fatty foods caused breast cancer. She had guilt that she was leaving her family because she ate burgers instead of salads."
Also, determine how much the patient wants to know. "About 90% of patients want full information [about their condition], but everyone wants to know everything about treatment." Physicians also can be instrumental in allaying end-of-life fears, Dr. Avery said. Regardless of illness, most patients think some symptom is going to get worse and worse and crescendo in pain before they die. "How do people with [chronic obstructive pulmonary disease] die? Yes, the symptoms get worse, but with COPD, they get COPD narcosis, get sleepy, and drift away."
Intentionally develop and use a compassionate tone, Dr. Avery said. This is important because patients surveyed after they received bad news said the attitude of the person who spoke with them was the most important factor. The clarity of the message and privacy were also important, but they ranked far behind clinician attitude, he said.
Allow for silence. Let the message to sink in. "Give the patient plenty of time to react, respond, and ask questions." Also allow tears – "That can be a real problem for a lot of doctors."
A challenge for physicians is to be empathetic without breaking down completely, Dr. Avery said. When working in hospice care in New York, he frequently spent the day traveling by subway to clients’ residences. "Am I going to travel around weeping? No. You have to somehow try to meet where they are, but you cannot go there completely. It would be self-destructive."
"One reason physicians think they do not give bad news well is they fear their own response; that they will break down," Dr. Avery said. Try to determine the patient’s attitude and reflect it back to them. "This is what you do when things get emotional. And they will correct you if you’re wrong. If you say ‘You sound angry,’ they might say ‘No, I’m upset.’"
Another important thing to ask patients is "Have the doctors told you how long you have?" An accurate prognosis will help patients and family prepare, Dr. Avery said. "You have to tell them. If you don’t, they will seek a second opinion and/or leave the long-term care setting, because no one has told them." Less-experienced doctors and doctors who have had long and strong relationship with a patient can be especially poor at prognostication, he said.
Be completely honest and avoid stating a precise amount of time, such as "3 months." "I say, ‘It could be weeks instead of months,’ or, ‘It could be months instead of years.’ If they ask for a more precise prognosis, tell them it’s difficult to say, because it is," Dr. Avery said.
If you still do not feel comfortable giving a patient bad news, refer the patient to someone who does. "Call in hospice, call in palliative care. If you cannot give that bad news, you are obligated to do this," Dr. Avery said.
If your attitude is right and you’re speaking with a compassionate tone, what else should you keep in mind when giving a patient bad news?
Watch your body language, because about 90% of communication is nonverbal, Dr. Avery said. Make eye contact, for example.
Do not sound matter of fact. "Patients will say the doctor appears bored," Dr. Avery explained. At the same time, avoid rambling, he advised. A good way to do this is intentionally pause on a frequent basis. Develop a technique to slow yourself down. Dr. Avery said he silently counts backward from 10 to slow himself down, for example.
Provide information in small chunks. This is better than "the information dump," which is a tendency to disclose every detail to a patient when initial bad news is shared.
"I tell the patient she can raise her hand and stop me if it’s too much information at any point," he said.
Never say, ‘There is nothing more I can do for you," Dr. Avery advised. He said he often tells patients that there is nothing more he can do for their dementia or their cancer, "but there is a lot I can do for you as a person. ... Otherwise, you are referring to them as a lung cancer, and you’ve reduced them to an organ with a disease."
You also can admit the limitations of medicine, Dr. Avery said. "You can say, for example, ‘I wish we had more effective therapy for your condition,’ or ‘I wish I had a magic pill or magic wand I could use it to take away your cancer.’ "
Dr. Avery said that he had no relevant financial conflict of interest.
TAMPA – Sharing bad news with patients might not be easy, but it’s a skill physicians can learn and as important as knowing how to ready an EKG or an x-ray, James A. Avery, MD, CMD, said.
"What I am proposing is that giving bad news well is a fundamental long-term care physician skill, and competence in this area is critical," Dr. Avery said at this year’s AMDA – Dedicated to Long Term Medicine annual meeting.
"Giving bad news ... takes desire, courage, and practice," said Dr. Avery. "Patients deserve to get bad news delivered with compassion, hope, and integrity."
Plan ahead for the conversation; start with what the patient knows and wants to know; and develop a compassionate tone, said Dr. Avery, chief medical officer at Golden Living in Washington, a corporation that focuses on skilled nursing, assisted living, and rehabilitation therapy. Also, always provide an appropriate prognosis. "It’s your obligation to bring this up. Patients and families may be afraid to ask."
What can happen if the conversation is not done correctly? "If bad news is given poorly, it can rob hope and create distress, confusion, and anxiety. It can weaken the patient’s faith and set off a chain of events that adversely affects the survivors for years," said Dr. Avery.
"I was particularly bad at giving bad news at first," he said. A pulmonologist by training, he also worked for years in hospice care in both Florida and New York. He spoke with patients who transitioned to hospice from Memorial Sloan-Kettering Cancer Center, for example.
"I learned quickly that if I was going to give bad news, not to schedule the patient for midmorning on a Monday. It is too chaotic," Dr. Avery said. Schedule the patient for the first appointment after lunch or at the end of the day. Allow sufficient time and create a comfortable, private place with tissues available, he added.
Next, determine where each patient is in terms of understanding his or her illness. "Explore and ask," Dr. Avery said. Good questions include:
- Is there anyone else you want to have in the conversation?
- How do you understand what has happened to you medically?
- What have doctors told you about this illness?
- What do you think caused this illness?
"I cannot tell you how many patients with colon cancer thought they had it because they took too many antacids," Dr. Avery said. "Also, I had one woman with breast cancer who responded ‘Burger King.’ She had read an article that fatty foods caused breast cancer. She had guilt that she was leaving her family because she ate burgers instead of salads."
Also, determine how much the patient wants to know. "About 90% of patients want full information [about their condition], but everyone wants to know everything about treatment." Physicians also can be instrumental in allaying end-of-life fears, Dr. Avery said. Regardless of illness, most patients think some symptom is going to get worse and worse and crescendo in pain before they die. "How do people with [chronic obstructive pulmonary disease] die? Yes, the symptoms get worse, but with COPD, they get COPD narcosis, get sleepy, and drift away."
Intentionally develop and use a compassionate tone, Dr. Avery said. This is important because patients surveyed after they received bad news said the attitude of the person who spoke with them was the most important factor. The clarity of the message and privacy were also important, but they ranked far behind clinician attitude, he said.
Allow for silence. Let the message to sink in. "Give the patient plenty of time to react, respond, and ask questions." Also allow tears – "That can be a real problem for a lot of doctors."
A challenge for physicians is to be empathetic without breaking down completely, Dr. Avery said. When working in hospice care in New York, he frequently spent the day traveling by subway to clients’ residences. "Am I going to travel around weeping? No. You have to somehow try to meet where they are, but you cannot go there completely. It would be self-destructive."
"One reason physicians think they do not give bad news well is they fear their own response; that they will break down," Dr. Avery said. Try to determine the patient’s attitude and reflect it back to them. "This is what you do when things get emotional. And they will correct you if you’re wrong. If you say ‘You sound angry,’ they might say ‘No, I’m upset.’"
Another important thing to ask patients is "Have the doctors told you how long you have?" An accurate prognosis will help patients and family prepare, Dr. Avery said. "You have to tell them. If you don’t, they will seek a second opinion and/or leave the long-term care setting, because no one has told them." Less-experienced doctors and doctors who have had long and strong relationship with a patient can be especially poor at prognostication, he said.
Be completely honest and avoid stating a precise amount of time, such as "3 months." "I say, ‘It could be weeks instead of months,’ or, ‘It could be months instead of years.’ If they ask for a more precise prognosis, tell them it’s difficult to say, because it is," Dr. Avery said.
If you still do not feel comfortable giving a patient bad news, refer the patient to someone who does. "Call in hospice, call in palliative care. If you cannot give that bad news, you are obligated to do this," Dr. Avery said.
If your attitude is right and you’re speaking with a compassionate tone, what else should you keep in mind when giving a patient bad news?
Watch your body language, because about 90% of communication is nonverbal, Dr. Avery said. Make eye contact, for example.
Do not sound matter of fact. "Patients will say the doctor appears bored," Dr. Avery explained. At the same time, avoid rambling, he advised. A good way to do this is intentionally pause on a frequent basis. Develop a technique to slow yourself down. Dr. Avery said he silently counts backward from 10 to slow himself down, for example.
Provide information in small chunks. This is better than "the information dump," which is a tendency to disclose every detail to a patient when initial bad news is shared.
"I tell the patient she can raise her hand and stop me if it’s too much information at any point," he said.
Never say, ‘There is nothing more I can do for you," Dr. Avery advised. He said he often tells patients that there is nothing more he can do for their dementia or their cancer, "but there is a lot I can do for you as a person. ... Otherwise, you are referring to them as a lung cancer, and you’ve reduced them to an organ with a disease."
You also can admit the limitations of medicine, Dr. Avery said. "You can say, for example, ‘I wish we had more effective therapy for your condition,’ or ‘I wish I had a magic pill or magic wand I could use it to take away your cancer.’ "
Dr. Avery said that he had no relevant financial conflict of interest.
TAMPA – Sharing bad news with patients might not be easy, but it’s a skill physicians can learn and as important as knowing how to ready an EKG or an x-ray, James A. Avery, MD, CMD, said.
"What I am proposing is that giving bad news well is a fundamental long-term care physician skill, and competence in this area is critical," Dr. Avery said at this year’s AMDA – Dedicated to Long Term Medicine annual meeting.
"Giving bad news ... takes desire, courage, and practice," said Dr. Avery. "Patients deserve to get bad news delivered with compassion, hope, and integrity."
Plan ahead for the conversation; start with what the patient knows and wants to know; and develop a compassionate tone, said Dr. Avery, chief medical officer at Golden Living in Washington, a corporation that focuses on skilled nursing, assisted living, and rehabilitation therapy. Also, always provide an appropriate prognosis. "It’s your obligation to bring this up. Patients and families may be afraid to ask."
What can happen if the conversation is not done correctly? "If bad news is given poorly, it can rob hope and create distress, confusion, and anxiety. It can weaken the patient’s faith and set off a chain of events that adversely affects the survivors for years," said Dr. Avery.
"I was particularly bad at giving bad news at first," he said. A pulmonologist by training, he also worked for years in hospice care in both Florida and New York. He spoke with patients who transitioned to hospice from Memorial Sloan-Kettering Cancer Center, for example.
"I learned quickly that if I was going to give bad news, not to schedule the patient for midmorning on a Monday. It is too chaotic," Dr. Avery said. Schedule the patient for the first appointment after lunch or at the end of the day. Allow sufficient time and create a comfortable, private place with tissues available, he added.
Next, determine where each patient is in terms of understanding his or her illness. "Explore and ask," Dr. Avery said. Good questions include:
- Is there anyone else you want to have in the conversation?
- How do you understand what has happened to you medically?
- What have doctors told you about this illness?
- What do you think caused this illness?
"I cannot tell you how many patients with colon cancer thought they had it because they took too many antacids," Dr. Avery said. "Also, I had one woman with breast cancer who responded ‘Burger King.’ She had read an article that fatty foods caused breast cancer. She had guilt that she was leaving her family because she ate burgers instead of salads."
Also, determine how much the patient wants to know. "About 90% of patients want full information [about their condition], but everyone wants to know everything about treatment." Physicians also can be instrumental in allaying end-of-life fears, Dr. Avery said. Regardless of illness, most patients think some symptom is going to get worse and worse and crescendo in pain before they die. "How do people with [chronic obstructive pulmonary disease] die? Yes, the symptoms get worse, but with COPD, they get COPD narcosis, get sleepy, and drift away."
Intentionally develop and use a compassionate tone, Dr. Avery said. This is important because patients surveyed after they received bad news said the attitude of the person who spoke with them was the most important factor. The clarity of the message and privacy were also important, but they ranked far behind clinician attitude, he said.
Allow for silence. Let the message to sink in. "Give the patient plenty of time to react, respond, and ask questions." Also allow tears – "That can be a real problem for a lot of doctors."
A challenge for physicians is to be empathetic without breaking down completely, Dr. Avery said. When working in hospice care in New York, he frequently spent the day traveling by subway to clients’ residences. "Am I going to travel around weeping? No. You have to somehow try to meet where they are, but you cannot go there completely. It would be self-destructive."
"One reason physicians think they do not give bad news well is they fear their own response; that they will break down," Dr. Avery said. Try to determine the patient’s attitude and reflect it back to them. "This is what you do when things get emotional. And they will correct you if you’re wrong. If you say ‘You sound angry,’ they might say ‘No, I’m upset.’"
Another important thing to ask patients is "Have the doctors told you how long you have?" An accurate prognosis will help patients and family prepare, Dr. Avery said. "You have to tell them. If you don’t, they will seek a second opinion and/or leave the long-term care setting, because no one has told them." Less-experienced doctors and doctors who have had long and strong relationship with a patient can be especially poor at prognostication, he said.
Be completely honest and avoid stating a precise amount of time, such as "3 months." "I say, ‘It could be weeks instead of months,’ or, ‘It could be months instead of years.’ If they ask for a more precise prognosis, tell them it’s difficult to say, because it is," Dr. Avery said.
If you still do not feel comfortable giving a patient bad news, refer the patient to someone who does. "Call in hospice, call in palliative care. If you cannot give that bad news, you are obligated to do this," Dr. Avery said.
If your attitude is right and you’re speaking with a compassionate tone, what else should you keep in mind when giving a patient bad news?
Watch your body language, because about 90% of communication is nonverbal, Dr. Avery said. Make eye contact, for example.
Do not sound matter of fact. "Patients will say the doctor appears bored," Dr. Avery explained. At the same time, avoid rambling, he advised. A good way to do this is intentionally pause on a frequent basis. Develop a technique to slow yourself down. Dr. Avery said he silently counts backward from 10 to slow himself down, for example.
Provide information in small chunks. This is better than "the information dump," which is a tendency to disclose every detail to a patient when initial bad news is shared.
"I tell the patient she can raise her hand and stop me if it’s too much information at any point," he said.
Never say, ‘There is nothing more I can do for you," Dr. Avery advised. He said he often tells patients that there is nothing more he can do for their dementia or their cancer, "but there is a lot I can do for you as a person. ... Otherwise, you are referring to them as a lung cancer, and you’ve reduced them to an organ with a disease."
You also can admit the limitations of medicine, Dr. Avery said. "You can say, for example, ‘I wish we had more effective therapy for your condition,’ or ‘I wish I had a magic pill or magic wand I could use it to take away your cancer.’ "
Dr. Avery said that he had no relevant financial conflict of interest.
FROM THE AMDA -- DEDICATED TO LONG TERM CARE MEDICINE ANNUAL MEETING
Precedex May Provide Relief at End of Life When Other Drugs Fail
VANCOUVER, B.C. – Precedex (dexmedetomidine), a drug used for years by intensivists and anesthesiologists for sedation, is increasingly being recognized as a valuable palliative care tool, according to Dr. Ellen M. Flanagan.
The alpha-2 agonist is an opioid-sparing analgesic that does not depress respiratory function; an anxiolytic with benzodiazepine-like activity but less risk of delirium and disinhibition; and a sedative from which patients can be aroused, among other properties, said Dr. Flanagan, an anesthesiologist at Duke University, Durham, N.C., with a special interest in palliative and end-of-life care.
Precedex "is incredible. It has really got some pretty profound implications for those patients who are having horrible deaths [with] intractable pain, agitation, and delirium." When standard drugs are "not sufficient to control symptoms, Precedex could do the trick," she said at the annual meeting of the American Academy of Hospice and Palliative Care Medicine.
Intensive-care doctors and anesthesiologists use the drug routinely. More than 4.5 million vials have been administered since the Food and Drug Administration approved it in 1999 to sedate intubated and mechanically ventilated ICU patients. The drug received a second indication in 2008 for sedation of nonintubated patients before or during surgical and other procedures.
But "it’s a new drug to most palliative care physicians," Dr. Flanagan said.
Part of the reason is that Precedex has not been studied in palliative care patients. To date, case reports offer the strongest evidence of palliative efficacy, she said.
Dr. Flanagan and her colleagues hope to change the situation. Their investigational new drug application was approved by the FDA for a study in nine advanced cancer patients in the final week of life.
The team will assess the drug’s impact as an add-on to standard therapy for pain, agitation, delirium, communication capacity, dyspnea, vomiting, and oral secretions. The team is currently negotiating funding with Hospira, the drug’s maker, and other possible sources.
For now, the best dose, duration, and symptom targets for palliative care remain uncertain, she said.
Still, nurse practitioner Jennifer Gentry, palliative care services clinical coordinator at Duke and copresenter with Dr. Flanagan, said she has seen Precedex ease end-of-life suffering when other drugs failed to do so adequately, and enable reductions in opioid use so patients could be extubated and allowed to die in hospice or other comfortable settings, instead of the ICU.
Meanwhile, studies in other populations have shown good effect, Dr. Flanagan said.
In a study of postoperative pain control in 100 women following hysterectomy, women given Precedex required 29% less morphine and reported less pain and nausea on the first postoperative day. Sedation was similar between the morphine alone and morphine-plus-Precedex groups (Br. J. Anaesth. 2009;102:117-22).
In another trial of mechanically ventilated ICU patients, 54% of 244 patients given Precedex 0.2-1.4 mcg/kg per hour experienced delirium, but 76.6% of 122 given midazolam 0.02-0.1 mg/kg per hour became delirious. Precedex patients were able to be extubated a median of 1.9 days sooner, as well (JAMA 2009;301:489-99).
In general, the drug provides arousable sedation, which "I think is one of the most incredible things about it. Instead of just titrating morphine until the patient is unconscious, the patient can be sedated so they look like they’re sleeping, and you shake their shoulder and all of a sudden they are awake. When the family comes in and wants to talk, you may be able to arouse that patient" for meaningful communication, Dr. Flanagan said.
Precedex is expensive, however, at about $58/100 mcg, or about $600 per day. It’s coming off patent in 2013, so less-expensive generic formulations may soon be available, she said.
The drug’s side effects include dry mouth, hypotension, and bradycardia. For the study, "we did a lot of work with the FDA to try to set up parameters trying to figure out what [bradycardia and hypotension] might be due to drug and what might be due to the natural trajectory of death," Dr. Flanagan said.
Dr. Flanagan said she has no financial interests in Precedex.
VANCOUVER, B.C. – Precedex (dexmedetomidine), a drug used for years by intensivists and anesthesiologists for sedation, is increasingly being recognized as a valuable palliative care tool, according to Dr. Ellen M. Flanagan.
The alpha-2 agonist is an opioid-sparing analgesic that does not depress respiratory function; an anxiolytic with benzodiazepine-like activity but less risk of delirium and disinhibition; and a sedative from which patients can be aroused, among other properties, said Dr. Flanagan, an anesthesiologist at Duke University, Durham, N.C., with a special interest in palliative and end-of-life care.
Precedex "is incredible. It has really got some pretty profound implications for those patients who are having horrible deaths [with] intractable pain, agitation, and delirium." When standard drugs are "not sufficient to control symptoms, Precedex could do the trick," she said at the annual meeting of the American Academy of Hospice and Palliative Care Medicine.
Intensive-care doctors and anesthesiologists use the drug routinely. More than 4.5 million vials have been administered since the Food and Drug Administration approved it in 1999 to sedate intubated and mechanically ventilated ICU patients. The drug received a second indication in 2008 for sedation of nonintubated patients before or during surgical and other procedures.
But "it’s a new drug to most palliative care physicians," Dr. Flanagan said.
Part of the reason is that Precedex has not been studied in palliative care patients. To date, case reports offer the strongest evidence of palliative efficacy, she said.
Dr. Flanagan and her colleagues hope to change the situation. Their investigational new drug application was approved by the FDA for a study in nine advanced cancer patients in the final week of life.
The team will assess the drug’s impact as an add-on to standard therapy for pain, agitation, delirium, communication capacity, dyspnea, vomiting, and oral secretions. The team is currently negotiating funding with Hospira, the drug’s maker, and other possible sources.
For now, the best dose, duration, and symptom targets for palliative care remain uncertain, she said.
Still, nurse practitioner Jennifer Gentry, palliative care services clinical coordinator at Duke and copresenter with Dr. Flanagan, said she has seen Precedex ease end-of-life suffering when other drugs failed to do so adequately, and enable reductions in opioid use so patients could be extubated and allowed to die in hospice or other comfortable settings, instead of the ICU.
Meanwhile, studies in other populations have shown good effect, Dr. Flanagan said.
In a study of postoperative pain control in 100 women following hysterectomy, women given Precedex required 29% less morphine and reported less pain and nausea on the first postoperative day. Sedation was similar between the morphine alone and morphine-plus-Precedex groups (Br. J. Anaesth. 2009;102:117-22).
In another trial of mechanically ventilated ICU patients, 54% of 244 patients given Precedex 0.2-1.4 mcg/kg per hour experienced delirium, but 76.6% of 122 given midazolam 0.02-0.1 mg/kg per hour became delirious. Precedex patients were able to be extubated a median of 1.9 days sooner, as well (JAMA 2009;301:489-99).
In general, the drug provides arousable sedation, which "I think is one of the most incredible things about it. Instead of just titrating morphine until the patient is unconscious, the patient can be sedated so they look like they’re sleeping, and you shake their shoulder and all of a sudden they are awake. When the family comes in and wants to talk, you may be able to arouse that patient" for meaningful communication, Dr. Flanagan said.
Precedex is expensive, however, at about $58/100 mcg, or about $600 per day. It’s coming off patent in 2013, so less-expensive generic formulations may soon be available, she said.
The drug’s side effects include dry mouth, hypotension, and bradycardia. For the study, "we did a lot of work with the FDA to try to set up parameters trying to figure out what [bradycardia and hypotension] might be due to drug and what might be due to the natural trajectory of death," Dr. Flanagan said.
Dr. Flanagan said she has no financial interests in Precedex.
VANCOUVER, B.C. – Precedex (dexmedetomidine), a drug used for years by intensivists and anesthesiologists for sedation, is increasingly being recognized as a valuable palliative care tool, according to Dr. Ellen M. Flanagan.
The alpha-2 agonist is an opioid-sparing analgesic that does not depress respiratory function; an anxiolytic with benzodiazepine-like activity but less risk of delirium and disinhibition; and a sedative from which patients can be aroused, among other properties, said Dr. Flanagan, an anesthesiologist at Duke University, Durham, N.C., with a special interest in palliative and end-of-life care.
Precedex "is incredible. It has really got some pretty profound implications for those patients who are having horrible deaths [with] intractable pain, agitation, and delirium." When standard drugs are "not sufficient to control symptoms, Precedex could do the trick," she said at the annual meeting of the American Academy of Hospice and Palliative Care Medicine.
Intensive-care doctors and anesthesiologists use the drug routinely. More than 4.5 million vials have been administered since the Food and Drug Administration approved it in 1999 to sedate intubated and mechanically ventilated ICU patients. The drug received a second indication in 2008 for sedation of nonintubated patients before or during surgical and other procedures.
But "it’s a new drug to most palliative care physicians," Dr. Flanagan said.
Part of the reason is that Precedex has not been studied in palliative care patients. To date, case reports offer the strongest evidence of palliative efficacy, she said.
Dr. Flanagan and her colleagues hope to change the situation. Their investigational new drug application was approved by the FDA for a study in nine advanced cancer patients in the final week of life.
The team will assess the drug’s impact as an add-on to standard therapy for pain, agitation, delirium, communication capacity, dyspnea, vomiting, and oral secretions. The team is currently negotiating funding with Hospira, the drug’s maker, and other possible sources.
For now, the best dose, duration, and symptom targets for palliative care remain uncertain, she said.
Still, nurse practitioner Jennifer Gentry, palliative care services clinical coordinator at Duke and copresenter with Dr. Flanagan, said she has seen Precedex ease end-of-life suffering when other drugs failed to do so adequately, and enable reductions in opioid use so patients could be extubated and allowed to die in hospice or other comfortable settings, instead of the ICU.
Meanwhile, studies in other populations have shown good effect, Dr. Flanagan said.
In a study of postoperative pain control in 100 women following hysterectomy, women given Precedex required 29% less morphine and reported less pain and nausea on the first postoperative day. Sedation was similar between the morphine alone and morphine-plus-Precedex groups (Br. J. Anaesth. 2009;102:117-22).
In another trial of mechanically ventilated ICU patients, 54% of 244 patients given Precedex 0.2-1.4 mcg/kg per hour experienced delirium, but 76.6% of 122 given midazolam 0.02-0.1 mg/kg per hour became delirious. Precedex patients were able to be extubated a median of 1.9 days sooner, as well (JAMA 2009;301:489-99).
In general, the drug provides arousable sedation, which "I think is one of the most incredible things about it. Instead of just titrating morphine until the patient is unconscious, the patient can be sedated so they look like they’re sleeping, and you shake their shoulder and all of a sudden they are awake. When the family comes in and wants to talk, you may be able to arouse that patient" for meaningful communication, Dr. Flanagan said.
Precedex is expensive, however, at about $58/100 mcg, or about $600 per day. It’s coming off patent in 2013, so less-expensive generic formulations may soon be available, she said.
The drug’s side effects include dry mouth, hypotension, and bradycardia. For the study, "we did a lot of work with the FDA to try to set up parameters trying to figure out what [bradycardia and hypotension] might be due to drug and what might be due to the natural trajectory of death," Dr. Flanagan said.
Dr. Flanagan said she has no financial interests in Precedex.
FROM THE ANNUAL MEETING OF THE AMERICAN ACADEMY OF HOSPICE AND PALLIATIVE CARE MEDICINE
Higher Opioid Doses More Likely to Lead to Unintentional Overdose
Higher doses of opioids – the equivalent of 50 mg/dL or more of morphine – are more likely than are lower doses to lead to unintentional overdose, according to a report in the April 6 issue of JAMA.
However, the practice of adding a prescription for as-needed opioids to an existing prescription for regularly scheduled opioids, which allows patients to deal with acute exacerbations of pain, does not raise the risk of unintentional overdose in most patients, as some experts have feared, said Amy S.B. Bohnert, Ph.D., of the Department of Veterans Affairs and the University of Michigan, both in Ann Arbor, and her associates.
Citing a "troubling and dramatic" 124% increase in unintentional deaths from opioid overdose in the United States in recent years, the investigators examined the relationship between opioid-prescribing patterns and opioid-related deaths using a nationally representative sample of 155,434 cases in the VA’s National Patient Care Database.
The study subjects included patients taking opioids for cancer pain, chronic bodily pain, headache, neuropathy, and injuries; the prescribed opioid analgesics included codeine, morphine, oxycodone, hydrocodone, oxymorphone, and hydromorphone.
There were 750 deaths attributed to unintentional overdose, for an overall rate of 0.04% among all patients treated with opioids. The risk of overdose increased when the opioid dose was equivalent to 50 mg/dL of morphine, and there was a dose-response relationship in which increasing daily prescribed doses of opioids correlated with increasing risk of overdose death.
As an example, prescribed doses of 100 mg/dL or higher were associated with a hazard ratio of approximately 12.
Dr. Bohnert and her colleagues described their study as the first to assess concurrent prescribing of regularly scheduled opioids plus as-needed opioids. With the exception of cancer patients, participants treated with this strategy showed no excess risk of overdose. "Recent treatment guidelines have indicated that the long-term safety of this strategy for pain exacerbations has not been established, [but] in [this] study we did not find evidence of greater overdose risk associated with this treatment practice, after accounting for maximum daily dose and patient characteristics," they noted (JAMA 2011;305:1315-21).
The researchers also said theirs is the first study of this issue to include patients with cancer. The overall rate of opioid overdose was lower in cancer patients, compared with other subjects, but clinicians should be mindful that renal and liver impairment in this patient group can interfere with the metabolism of opioids and that high doses can place cancer patients at risk.
Cancer patients also were at increased risk of overdose if they were prescribed as-needed opioids alone, rather than regularly scheduled opioids. "Pain for patients with cancer can be particularly rapid in onset, unpredictable, and severe, and taking opioid doses as needed may result in [sudden] high doses being taken without the benefit of tolerance developed through a regularly scheduled opioid," the investigators said.
In a research letter accompanying this report, Dr. Nora D. Volkow, director of the National Institute on Drug Abuse, and her associates presented data from their study of prescription practices, which was aimed at identifying possible contributors to the high rate of opioid abuse. They drew on information in a national prescription database that included about 80 million prescriptions for opioid analgesics.
The main prescribers were general, family medicine, and osteopathic physicians, who accounted for 29% of these prescriptions, followed by internists (15%), dentists (8%), and orthopedic surgeons (8%).
Across all specialties, more than half of these prescriptions (56%, or 45 million) were given to patients who had already filled another opioid prescription during the preceding month. The data could not show whether such prescribing was "justified" or rather "suggests the need to improve information infrastructures that could enhance the safety of prescribed opioid analgesics and minimize diversion," Dr. Volkow and her colleagues said (JAMA 2011;305:1299-1301).
In addition, they found a fivefold increase in drug-treatment admissions for pharmaceutical opioids, from 19,941 to 121,091 in the past 10 years. Emergency department visits related to opioid overdose rose from 144,644 to 305,885 between 2004 and 2008. And deaths from unintentional opioid overdose have quadrupled, from approximately 3,000 in 1999 to 12,000 in 2007.
"Opioid overdose is now the second leading cause of unintentional death in the United States, second only to motor vehicle crashes," they noted.
The study by Dr. Bohnert and her colleagues was supported by the U.S. Department of Veterans Affairs’ Office of Mental Health Services and Office of Patient Care Services. One of Dr. Bohnert’s associates reported ties to Purdue Pharma. Dr. Volkow and her colleagues reported no disclosures.
Higher doses of opioids – the equivalent of 50 mg/dL or more of morphine – are more likely than are lower doses to lead to unintentional overdose, according to a report in the April 6 issue of JAMA.
However, the practice of adding a prescription for as-needed opioids to an existing prescription for regularly scheduled opioids, which allows patients to deal with acute exacerbations of pain, does not raise the risk of unintentional overdose in most patients, as some experts have feared, said Amy S.B. Bohnert, Ph.D., of the Department of Veterans Affairs and the University of Michigan, both in Ann Arbor, and her associates.
Citing a "troubling and dramatic" 124% increase in unintentional deaths from opioid overdose in the United States in recent years, the investigators examined the relationship between opioid-prescribing patterns and opioid-related deaths using a nationally representative sample of 155,434 cases in the VA’s National Patient Care Database.
The study subjects included patients taking opioids for cancer pain, chronic bodily pain, headache, neuropathy, and injuries; the prescribed opioid analgesics included codeine, morphine, oxycodone, hydrocodone, oxymorphone, and hydromorphone.
There were 750 deaths attributed to unintentional overdose, for an overall rate of 0.04% among all patients treated with opioids. The risk of overdose increased when the opioid dose was equivalent to 50 mg/dL of morphine, and there was a dose-response relationship in which increasing daily prescribed doses of opioids correlated with increasing risk of overdose death.
As an example, prescribed doses of 100 mg/dL or higher were associated with a hazard ratio of approximately 12.
Dr. Bohnert and her colleagues described their study as the first to assess concurrent prescribing of regularly scheduled opioids plus as-needed opioids. With the exception of cancer patients, participants treated with this strategy showed no excess risk of overdose. "Recent treatment guidelines have indicated that the long-term safety of this strategy for pain exacerbations has not been established, [but] in [this] study we did not find evidence of greater overdose risk associated with this treatment practice, after accounting for maximum daily dose and patient characteristics," they noted (JAMA 2011;305:1315-21).
The researchers also said theirs is the first study of this issue to include patients with cancer. The overall rate of opioid overdose was lower in cancer patients, compared with other subjects, but clinicians should be mindful that renal and liver impairment in this patient group can interfere with the metabolism of opioids and that high doses can place cancer patients at risk.
Cancer patients also were at increased risk of overdose if they were prescribed as-needed opioids alone, rather than regularly scheduled opioids. "Pain for patients with cancer can be particularly rapid in onset, unpredictable, and severe, and taking opioid doses as needed may result in [sudden] high doses being taken without the benefit of tolerance developed through a regularly scheduled opioid," the investigators said.
In a research letter accompanying this report, Dr. Nora D. Volkow, director of the National Institute on Drug Abuse, and her associates presented data from their study of prescription practices, which was aimed at identifying possible contributors to the high rate of opioid abuse. They drew on information in a national prescription database that included about 80 million prescriptions for opioid analgesics.
The main prescribers were general, family medicine, and osteopathic physicians, who accounted for 29% of these prescriptions, followed by internists (15%), dentists (8%), and orthopedic surgeons (8%).
Across all specialties, more than half of these prescriptions (56%, or 45 million) were given to patients who had already filled another opioid prescription during the preceding month. The data could not show whether such prescribing was "justified" or rather "suggests the need to improve information infrastructures that could enhance the safety of prescribed opioid analgesics and minimize diversion," Dr. Volkow and her colleagues said (JAMA 2011;305:1299-1301).
In addition, they found a fivefold increase in drug-treatment admissions for pharmaceutical opioids, from 19,941 to 121,091 in the past 10 years. Emergency department visits related to opioid overdose rose from 144,644 to 305,885 between 2004 and 2008. And deaths from unintentional opioid overdose have quadrupled, from approximately 3,000 in 1999 to 12,000 in 2007.
"Opioid overdose is now the second leading cause of unintentional death in the United States, second only to motor vehicle crashes," they noted.
The study by Dr. Bohnert and her colleagues was supported by the U.S. Department of Veterans Affairs’ Office of Mental Health Services and Office of Patient Care Services. One of Dr. Bohnert’s associates reported ties to Purdue Pharma. Dr. Volkow and her colleagues reported no disclosures.
Higher doses of opioids – the equivalent of 50 mg/dL or more of morphine – are more likely than are lower doses to lead to unintentional overdose, according to a report in the April 6 issue of JAMA.
However, the practice of adding a prescription for as-needed opioids to an existing prescription for regularly scheduled opioids, which allows patients to deal with acute exacerbations of pain, does not raise the risk of unintentional overdose in most patients, as some experts have feared, said Amy S.B. Bohnert, Ph.D., of the Department of Veterans Affairs and the University of Michigan, both in Ann Arbor, and her associates.
Citing a "troubling and dramatic" 124% increase in unintentional deaths from opioid overdose in the United States in recent years, the investigators examined the relationship between opioid-prescribing patterns and opioid-related deaths using a nationally representative sample of 155,434 cases in the VA’s National Patient Care Database.
The study subjects included patients taking opioids for cancer pain, chronic bodily pain, headache, neuropathy, and injuries; the prescribed opioid analgesics included codeine, morphine, oxycodone, hydrocodone, oxymorphone, and hydromorphone.
There were 750 deaths attributed to unintentional overdose, for an overall rate of 0.04% among all patients treated with opioids. The risk of overdose increased when the opioid dose was equivalent to 50 mg/dL of morphine, and there was a dose-response relationship in which increasing daily prescribed doses of opioids correlated with increasing risk of overdose death.
As an example, prescribed doses of 100 mg/dL or higher were associated with a hazard ratio of approximately 12.
Dr. Bohnert and her colleagues described their study as the first to assess concurrent prescribing of regularly scheduled opioids plus as-needed opioids. With the exception of cancer patients, participants treated with this strategy showed no excess risk of overdose. "Recent treatment guidelines have indicated that the long-term safety of this strategy for pain exacerbations has not been established, [but] in [this] study we did not find evidence of greater overdose risk associated with this treatment practice, after accounting for maximum daily dose and patient characteristics," they noted (JAMA 2011;305:1315-21).
The researchers also said theirs is the first study of this issue to include patients with cancer. The overall rate of opioid overdose was lower in cancer patients, compared with other subjects, but clinicians should be mindful that renal and liver impairment in this patient group can interfere with the metabolism of opioids and that high doses can place cancer patients at risk.
Cancer patients also were at increased risk of overdose if they were prescribed as-needed opioids alone, rather than regularly scheduled opioids. "Pain for patients with cancer can be particularly rapid in onset, unpredictable, and severe, and taking opioid doses as needed may result in [sudden] high doses being taken without the benefit of tolerance developed through a regularly scheduled opioid," the investigators said.
In a research letter accompanying this report, Dr. Nora D. Volkow, director of the National Institute on Drug Abuse, and her associates presented data from their study of prescription practices, which was aimed at identifying possible contributors to the high rate of opioid abuse. They drew on information in a national prescription database that included about 80 million prescriptions for opioid analgesics.
The main prescribers were general, family medicine, and osteopathic physicians, who accounted for 29% of these prescriptions, followed by internists (15%), dentists (8%), and orthopedic surgeons (8%).
Across all specialties, more than half of these prescriptions (56%, or 45 million) were given to patients who had already filled another opioid prescription during the preceding month. The data could not show whether such prescribing was "justified" or rather "suggests the need to improve information infrastructures that could enhance the safety of prescribed opioid analgesics and minimize diversion," Dr. Volkow and her colleagues said (JAMA 2011;305:1299-1301).
In addition, they found a fivefold increase in drug-treatment admissions for pharmaceutical opioids, from 19,941 to 121,091 in the past 10 years. Emergency department visits related to opioid overdose rose from 144,644 to 305,885 between 2004 and 2008. And deaths from unintentional opioid overdose have quadrupled, from approximately 3,000 in 1999 to 12,000 in 2007.
"Opioid overdose is now the second leading cause of unintentional death in the United States, second only to motor vehicle crashes," they noted.
The study by Dr. Bohnert and her colleagues was supported by the U.S. Department of Veterans Affairs’ Office of Mental Health Services and Office of Patient Care Services. One of Dr. Bohnert’s associates reported ties to Purdue Pharma. Dr. Volkow and her colleagues reported no disclosures.
FROM JAMA
DNRs, Older Demographic Can Skew Trauma Center Mortality Stats
DETROIT – Benchmarking trauma center performance using traditional measures fails to adequately reflect the aging face of trauma deaths today, a new study suggests.
Increased public awareness of advance directives and an aging population has shifted the demographics of trauma mortality, Dr. Glen A. Franklin said at the annual meeting of the Central Surgical Association.
"Traditional measures of trauma center quality based on injury severity or comorbidity indexing may not capture all the issues associated with mortality," he said.
Dr. Franklin and his colleagues at the University of Louisville in Kentucky studied the effects of advance directives and futile care on trauma center mortality by examining all trauma deaths in 2008-2009 to determine the proportion of patients whose outcome could have been changed by medical care.
Of the 5,433 patients treated, 347 died, resulting in a crude mortality of 6.4%. Advance directives were in place in 18% of patients at the time of admission, said Dr. Franklin, director of the university’s critical care/trauma fellowship program.
Care was withdrawn in 147 deaths (42%) at a median of 1.5 days. The median injury severity score was 25 for all patients who died (both those who did and those who did not have care withdrawn). Most deaths (66%) occurred early in the hospital course, within 48 hours or less, and the decision for withdrawal of care was made within that time frame in 24% of cases.
On univariate analysis, only age (69.5 years vs. 48 years) and the presence of an advance directive (69% vs. 31%) were significantly different between patients in whom care was or was not withdrawn. Injury severity score did not correlate with withdrawal of care (odds ratio 1.014), he said.
Patients with advance directives were significantly older than those without advance directives (72 years vs. 53 years) and were three times as likely to have care withdrawn (OR 3.11). The decision to withdraw care was made by family members in 140 cases (95%), by the patient in 6, and by power of attorney in 1.
Of the patients who died, 69 patients (20%) arrived at the trauma center in or rapidly progressed to cardiac arrest, and were placed in the futile-care category. A vigorous quality improvement review process indicated that medical care could not have been reasonably expected to impact survival in 62% (215 deaths), Dr. Franklin said. Only 12 deaths were considered potentially preventable.
When these factors are taken into account, the hospital’s overall crude death rate of 6.4% fell to 3.7% when adjusted for the 147 patients with care withdrawn (200 deaths remaining/5,433 cases) and further to 2.4% when adjusted for the 69 futile care patients (131 deaths remaining/5,433 cases).
According to information from the National Trauma Data Bank, trauma patient volume and injury severity are high at the University of Louisville, but so is the center’s mortality.
"It is this type of [nonadjusted] data that is making it out into the public sector, with really no ability for the layperson to interpret its meaning," Dr. Franklin said.
Risk-adjusted data will soon be available via the American College of Surgeons’ recently launched Trauma Quality Improvement Program (TQIP), he observed. The program mimics the National Surgical Quality Improvement Program (NSQIP) and looks at observed-to-expected mortality ratio in a risk-adjusted fashion. In a recent study using this approach, only 9 of 132 centers in the United States were identified as being above-average performers with regard to elderly trauma after adjustment for case mix (Ann. Surg. 2011;253:144-50), he added.
Dr. Franklin observed that societal, cultural, regional, religious, and racial factors can affect family perceptions of appropriate escalation of care and thus mortality outcomes. For example, emergency medical technicians in Kentucky cannot declare trauma patients dead, and paramedics can do so only under extreme circumstances such as decapitation or rigor mortis. As a result, deceased patients who arrive in the trauma center may be subjected to inappropriate resuscitation attempts, and ultimately figure in the trauma center’s mortality profile, he said. For another example, the decision to withdraw care may be made more conservatively when caring for patients in the Bible Belt, where religious beliefs are more likely to play a role in attitudes toward the end of life.
"We believe all of these factors need to be placed into the data set in some manner to affect trauma center performance, particularly as transparency becomes a key in the quality movement," he concluded.
Invited discussant Dr. Mark Hemmila of the University of Michigan, Ann Arbor, asked whether do-not-resuscitate (DNR) status prior to injury or surgery, data that are collected by both NSQIP and TQIP, might be a reasonable compromise for use as a covariate in risk adjustment of mortality data. He went on to ask whether trauma centers would collect this data accurately or whether there was potential to rig the system.
Dr. Franklin said that inclusion of DNR status or withdrawal of care in a data model could be problematic unless the terms are clearly defined beforehand. He noted, for example, that a DNR order may not be written in the chart for some patients, but that it is clear physicians are writing for comfort care measures when they extubate a patient, increase their pain medications, and move them out of a monitored setting. Although the potential exists to game the system and data entry needs to be reinforced, the data system itself is okay, according to Dr. Franklin.
Finally, it was noted by members of the audience that the medical community has not done a good job of educating the public on such issues as withdrawal of care, DNR orders, and palliative care, and that these issues will become more pressing as the population ages. When asked whether the researchers looked at race, Dr. Franklin said they did not, but that anecdotally, minorities were less likely to withdraw care.
Dr. Franklin and his coauthors disclosed no relevant conflicts of interest.
DETROIT – Benchmarking trauma center performance using traditional measures fails to adequately reflect the aging face of trauma deaths today, a new study suggests.
Increased public awareness of advance directives and an aging population has shifted the demographics of trauma mortality, Dr. Glen A. Franklin said at the annual meeting of the Central Surgical Association.
"Traditional measures of trauma center quality based on injury severity or comorbidity indexing may not capture all the issues associated with mortality," he said.
Dr. Franklin and his colleagues at the University of Louisville in Kentucky studied the effects of advance directives and futile care on trauma center mortality by examining all trauma deaths in 2008-2009 to determine the proportion of patients whose outcome could have been changed by medical care.
Of the 5,433 patients treated, 347 died, resulting in a crude mortality of 6.4%. Advance directives were in place in 18% of patients at the time of admission, said Dr. Franklin, director of the university’s critical care/trauma fellowship program.
Care was withdrawn in 147 deaths (42%) at a median of 1.5 days. The median injury severity score was 25 for all patients who died (both those who did and those who did not have care withdrawn). Most deaths (66%) occurred early in the hospital course, within 48 hours or less, and the decision for withdrawal of care was made within that time frame in 24% of cases.
On univariate analysis, only age (69.5 years vs. 48 years) and the presence of an advance directive (69% vs. 31%) were significantly different between patients in whom care was or was not withdrawn. Injury severity score did not correlate with withdrawal of care (odds ratio 1.014), he said.
Patients with advance directives were significantly older than those without advance directives (72 years vs. 53 years) and were three times as likely to have care withdrawn (OR 3.11). The decision to withdraw care was made by family members in 140 cases (95%), by the patient in 6, and by power of attorney in 1.
Of the patients who died, 69 patients (20%) arrived at the trauma center in or rapidly progressed to cardiac arrest, and were placed in the futile-care category. A vigorous quality improvement review process indicated that medical care could not have been reasonably expected to impact survival in 62% (215 deaths), Dr. Franklin said. Only 12 deaths were considered potentially preventable.
When these factors are taken into account, the hospital’s overall crude death rate of 6.4% fell to 3.7% when adjusted for the 147 patients with care withdrawn (200 deaths remaining/5,433 cases) and further to 2.4% when adjusted for the 69 futile care patients (131 deaths remaining/5,433 cases).
According to information from the National Trauma Data Bank, trauma patient volume and injury severity are high at the University of Louisville, but so is the center’s mortality.
"It is this type of [nonadjusted] data that is making it out into the public sector, with really no ability for the layperson to interpret its meaning," Dr. Franklin said.
Risk-adjusted data will soon be available via the American College of Surgeons’ recently launched Trauma Quality Improvement Program (TQIP), he observed. The program mimics the National Surgical Quality Improvement Program (NSQIP) and looks at observed-to-expected mortality ratio in a risk-adjusted fashion. In a recent study using this approach, only 9 of 132 centers in the United States were identified as being above-average performers with regard to elderly trauma after adjustment for case mix (Ann. Surg. 2011;253:144-50), he added.
Dr. Franklin observed that societal, cultural, regional, religious, and racial factors can affect family perceptions of appropriate escalation of care and thus mortality outcomes. For example, emergency medical technicians in Kentucky cannot declare trauma patients dead, and paramedics can do so only under extreme circumstances such as decapitation or rigor mortis. As a result, deceased patients who arrive in the trauma center may be subjected to inappropriate resuscitation attempts, and ultimately figure in the trauma center’s mortality profile, he said. For another example, the decision to withdraw care may be made more conservatively when caring for patients in the Bible Belt, where religious beliefs are more likely to play a role in attitudes toward the end of life.
"We believe all of these factors need to be placed into the data set in some manner to affect trauma center performance, particularly as transparency becomes a key in the quality movement," he concluded.
Invited discussant Dr. Mark Hemmila of the University of Michigan, Ann Arbor, asked whether do-not-resuscitate (DNR) status prior to injury or surgery, data that are collected by both NSQIP and TQIP, might be a reasonable compromise for use as a covariate in risk adjustment of mortality data. He went on to ask whether trauma centers would collect this data accurately or whether there was potential to rig the system.
Dr. Franklin said that inclusion of DNR status or withdrawal of care in a data model could be problematic unless the terms are clearly defined beforehand. He noted, for example, that a DNR order may not be written in the chart for some patients, but that it is clear physicians are writing for comfort care measures when they extubate a patient, increase their pain medications, and move them out of a monitored setting. Although the potential exists to game the system and data entry needs to be reinforced, the data system itself is okay, according to Dr. Franklin.
Finally, it was noted by members of the audience that the medical community has not done a good job of educating the public on such issues as withdrawal of care, DNR orders, and palliative care, and that these issues will become more pressing as the population ages. When asked whether the researchers looked at race, Dr. Franklin said they did not, but that anecdotally, minorities were less likely to withdraw care.
Dr. Franklin and his coauthors disclosed no relevant conflicts of interest.
DETROIT – Benchmarking trauma center performance using traditional measures fails to adequately reflect the aging face of trauma deaths today, a new study suggests.
Increased public awareness of advance directives and an aging population has shifted the demographics of trauma mortality, Dr. Glen A. Franklin said at the annual meeting of the Central Surgical Association.
"Traditional measures of trauma center quality based on injury severity or comorbidity indexing may not capture all the issues associated with mortality," he said.
Dr. Franklin and his colleagues at the University of Louisville in Kentucky studied the effects of advance directives and futile care on trauma center mortality by examining all trauma deaths in 2008-2009 to determine the proportion of patients whose outcome could have been changed by medical care.
Of the 5,433 patients treated, 347 died, resulting in a crude mortality of 6.4%. Advance directives were in place in 18% of patients at the time of admission, said Dr. Franklin, director of the university’s critical care/trauma fellowship program.
Care was withdrawn in 147 deaths (42%) at a median of 1.5 days. The median injury severity score was 25 for all patients who died (both those who did and those who did not have care withdrawn). Most deaths (66%) occurred early in the hospital course, within 48 hours or less, and the decision for withdrawal of care was made within that time frame in 24% of cases.
On univariate analysis, only age (69.5 years vs. 48 years) and the presence of an advance directive (69% vs. 31%) were significantly different between patients in whom care was or was not withdrawn. Injury severity score did not correlate with withdrawal of care (odds ratio 1.014), he said.
Patients with advance directives were significantly older than those without advance directives (72 years vs. 53 years) and were three times as likely to have care withdrawn (OR 3.11). The decision to withdraw care was made by family members in 140 cases (95%), by the patient in 6, and by power of attorney in 1.
Of the patients who died, 69 patients (20%) arrived at the trauma center in or rapidly progressed to cardiac arrest, and were placed in the futile-care category. A vigorous quality improvement review process indicated that medical care could not have been reasonably expected to impact survival in 62% (215 deaths), Dr. Franklin said. Only 12 deaths were considered potentially preventable.
When these factors are taken into account, the hospital’s overall crude death rate of 6.4% fell to 3.7% when adjusted for the 147 patients with care withdrawn (200 deaths remaining/5,433 cases) and further to 2.4% when adjusted for the 69 futile care patients (131 deaths remaining/5,433 cases).
According to information from the National Trauma Data Bank, trauma patient volume and injury severity are high at the University of Louisville, but so is the center’s mortality.
"It is this type of [nonadjusted] data that is making it out into the public sector, with really no ability for the layperson to interpret its meaning," Dr. Franklin said.
Risk-adjusted data will soon be available via the American College of Surgeons’ recently launched Trauma Quality Improvement Program (TQIP), he observed. The program mimics the National Surgical Quality Improvement Program (NSQIP) and looks at observed-to-expected mortality ratio in a risk-adjusted fashion. In a recent study using this approach, only 9 of 132 centers in the United States were identified as being above-average performers with regard to elderly trauma after adjustment for case mix (Ann. Surg. 2011;253:144-50), he added.
Dr. Franklin observed that societal, cultural, regional, religious, and racial factors can affect family perceptions of appropriate escalation of care and thus mortality outcomes. For example, emergency medical technicians in Kentucky cannot declare trauma patients dead, and paramedics can do so only under extreme circumstances such as decapitation or rigor mortis. As a result, deceased patients who arrive in the trauma center may be subjected to inappropriate resuscitation attempts, and ultimately figure in the trauma center’s mortality profile, he said. For another example, the decision to withdraw care may be made more conservatively when caring for patients in the Bible Belt, where religious beliefs are more likely to play a role in attitudes toward the end of life.
"We believe all of these factors need to be placed into the data set in some manner to affect trauma center performance, particularly as transparency becomes a key in the quality movement," he concluded.
Invited discussant Dr. Mark Hemmila of the University of Michigan, Ann Arbor, asked whether do-not-resuscitate (DNR) status prior to injury or surgery, data that are collected by both NSQIP and TQIP, might be a reasonable compromise for use as a covariate in risk adjustment of mortality data. He went on to ask whether trauma centers would collect this data accurately or whether there was potential to rig the system.
Dr. Franklin said that inclusion of DNR status or withdrawal of care in a data model could be problematic unless the terms are clearly defined beforehand. He noted, for example, that a DNR order may not be written in the chart for some patients, but that it is clear physicians are writing for comfort care measures when they extubate a patient, increase their pain medications, and move them out of a monitored setting. Although the potential exists to game the system and data entry needs to be reinforced, the data system itself is okay, according to Dr. Franklin.
Finally, it was noted by members of the audience that the medical community has not done a good job of educating the public on such issues as withdrawal of care, DNR orders, and palliative care, and that these issues will become more pressing as the population ages. When asked whether the researchers looked at race, Dr. Franklin said they did not, but that anecdotally, minorities were less likely to withdraw care.
Dr. Franklin and his coauthors disclosed no relevant conflicts of interest.
FROM THE ANNUAL MEETING OF THE CENTRAL SURGICAL ASSOCIATION
Chart All Possible Diagnoses to Improve Hospital Mortality Scores
VANCOUVER, B.C. - To improve hospital mortality scores, clinicians should include all possible diagnoses in patient charts, according to oncologist and palliative care specialist Dr. Thomas J. Smith.
U.S. News & World Report’s "Best Hospitals," the Centers for Medicaid and Medicare Services’ Hospital Compare, and other hospital rating programs compare hospital outcomes to national averages based on how sick patients are, said Dr. Smith, medical director of palliative care at Virginia Commonwealth University Massey Cancer Center in Richmond.
"You get counted on your observed mortality rate. They compare that to how many people are expected to die" according to the averages, he said at the annual meeting of the American Academy of Hospice and Palliative Care Medicine. Deaths in excess of the averages are considered unexpected and are assumed to be caused by poor quality care, which lowers hospitals in the mortality rankings.
By listing all possible diagnoses – especially at the end of life – "[you increase] your expected mortality, so that your observed-to-expected ratio improves. Even if you ignore improving mortality, if you change [how many people are expected to die] you’ll improve your ratio" and hospital standing, he said.
The reason is that hospital coders pull the diagnoses off patient charts and include them in claims submissions, the data upon which the rankings are based.
The approach is not about "gaming the system," Dr. Smith said. "You are adequately documenting the severity of the patients that you take care of" to ensure accurate mortality scores.
The key is to list diagnoses, not "medical thoughts," he said.
For example, " ‘lytic lesion of vertebra on spine films’ does not count. Write ‘bone mets’ [because] your coders can count that. Don’t write ‘neutrophils’ with an arrow going down. It doesn’t count. You have to write ‘neutropenia.’ If you write ‘admit for chemo,’ you can write ‘admit for chemo, dehydration, chronic blood loss anemia’ [as appropriate] and your severity index will go up a lot," said Dr. Smith, adding that each site of metastases should be listed because "you get points for each one."
Similarly, " ‘chest x-ray with pneumonitis status post radiation therapy’ does not count. You have to write ‘radiation pneumonitis.’ [And] don’t put ‘total protein and albumin low.’ Put ‘cachexia or malnutrition, moderate or severe.’ Don’t write ‘admit for pain control.’ Write ‘intractable pain form malignancy,’ and you have to say ‘lung cancer, breast cancer, bone cancer, liver cancer’ [accordingly]," he said.
Such diagnoses only have to be written once during the admission to count, he noted.
The approach worked for Massey Cancer Center’s palliative care unit.
"In about an 18- to 24-month period, we paid a lot attention to having most of the attendings and the fellows write out diagnostic statements, rather than medical thoughts," Dr. Smith said.
"When we started, our actual-vs.-expected mortality ratio was 150% higher than what it was supposed to be. We didn’t change the mortality rate one bit, but we changed the expected mortality. It came down to 55%, which is still high, but, heck, it’s a palliative care unit. A lot of people are sick and they’re going to die," he said.
"The APR-DRG [All Patient Refined Diagnosis Related Groups] severity of illness doubled just by paying attention to having those diagnostic statements put in," he said.
Dr. Smith gave an example of how the strategy also increases payments: An elderly person was admitted upon diagnosis of urosepsis, dehydration, and chronic obstructive pulmonary disease, but the patient’s condition allowed for more, including urinary tract infection; malnutrition; preexisting decubitus ulcer; and shock because the patient was a bit hypotensive.
With the additional diagnoses, the patient’s severity of illness (SOI) weighting increased from 0.5973 to 3.3739, resulting in a $5,613 payment increase.
"That’s a huge difference," Dr. Smith said.
He had a final tip for improving Hospital Compare mortality scores.
The CMS counts 30-day mortality, but the 30-day clock doesn’t start until 24 hours after admission. "So if you can enroll a Medicare patient in hospice within 24 hours of them hitting your emergency room door, then they don’t count towards your 30 day mortality," Dr. Smith said.
To help, "you can post somebody in the ER and make the ER aware that this is important, so that you can see those patients in the ER. Let your social workers know that the 24 shot-clock is ticking," Dr. Smith said.
The goal is to "accelerate the process of patients and families making choices. You can push that a little bit," he said.
Dr. Smith said he has no relevant disclosures.
What we write in the chart will have a direct impact on the DRG
applied to that episode of care, which will in turn affect the expected
length of stay. In addition, the more accurately we reflect the
patient's true condition - including all the patient diagnoses and
comorbidities - the higher the patient's expected mortality will be.
Accurate chart documentation is not gaming the system. It is an
essential piece of the system itself.
As far as ethics goes, intent
plays an important role. The medical literature already suggests that we
enroll patients into hospice too late. If the intent is to identify
hospice-appropriate patients early and to otherwise provide them with
the benefits of hospice longer by early enrollment, I think that is
fine. However, if the intent is to increase your exclusion pool by
recruiting questionable hospice patients and manipulating them into
enrolling into hospice - well then, that sort of speaks for itself,
doesn't it?
Franklin A. Michota, M.D., is the director of
academic affairs in the department of hospital medicine at the Cleveland
Clinic. He reported no relevant conflicts of interest.
What we write in the chart will have a direct impact on the DRG
applied to that episode of care, which will in turn affect the expected
length of stay. In addition, the more accurately we reflect the
patient's true condition - including all the patient diagnoses and
comorbidities - the higher the patient's expected mortality will be.
Accurate chart documentation is not gaming the system. It is an
essential piece of the system itself.
As far as ethics goes, intent
plays an important role. The medical literature already suggests that we
enroll patients into hospice too late. If the intent is to identify
hospice-appropriate patients early and to otherwise provide them with
the benefits of hospice longer by early enrollment, I think that is
fine. However, if the intent is to increase your exclusion pool by
recruiting questionable hospice patients and manipulating them into
enrolling into hospice - well then, that sort of speaks for itself,
doesn't it?
Franklin A. Michota, M.D., is the director of
academic affairs in the department of hospital medicine at the Cleveland
Clinic. He reported no relevant conflicts of interest.
What we write in the chart will have a direct impact on the DRG
applied to that episode of care, which will in turn affect the expected
length of stay. In addition, the more accurately we reflect the
patient's true condition - including all the patient diagnoses and
comorbidities - the higher the patient's expected mortality will be.
Accurate chart documentation is not gaming the system. It is an
essential piece of the system itself.
As far as ethics goes, intent
plays an important role. The medical literature already suggests that we
enroll patients into hospice too late. If the intent is to identify
hospice-appropriate patients early and to otherwise provide them with
the benefits of hospice longer by early enrollment, I think that is
fine. However, if the intent is to increase your exclusion pool by
recruiting questionable hospice patients and manipulating them into
enrolling into hospice - well then, that sort of speaks for itself,
doesn't it?
Franklin A. Michota, M.D., is the director of
academic affairs in the department of hospital medicine at the Cleveland
Clinic. He reported no relevant conflicts of interest.
VANCOUVER, B.C. - To improve hospital mortality scores, clinicians should include all possible diagnoses in patient charts, according to oncologist and palliative care specialist Dr. Thomas J. Smith.
U.S. News & World Report’s "Best Hospitals," the Centers for Medicaid and Medicare Services’ Hospital Compare, and other hospital rating programs compare hospital outcomes to national averages based on how sick patients are, said Dr. Smith, medical director of palliative care at Virginia Commonwealth University Massey Cancer Center in Richmond.
"You get counted on your observed mortality rate. They compare that to how many people are expected to die" according to the averages, he said at the annual meeting of the American Academy of Hospice and Palliative Care Medicine. Deaths in excess of the averages are considered unexpected and are assumed to be caused by poor quality care, which lowers hospitals in the mortality rankings.
By listing all possible diagnoses – especially at the end of life – "[you increase] your expected mortality, so that your observed-to-expected ratio improves. Even if you ignore improving mortality, if you change [how many people are expected to die] you’ll improve your ratio" and hospital standing, he said.
The reason is that hospital coders pull the diagnoses off patient charts and include them in claims submissions, the data upon which the rankings are based.
The approach is not about "gaming the system," Dr. Smith said. "You are adequately documenting the severity of the patients that you take care of" to ensure accurate mortality scores.
The key is to list diagnoses, not "medical thoughts," he said.
For example, " ‘lytic lesion of vertebra on spine films’ does not count. Write ‘bone mets’ [because] your coders can count that. Don’t write ‘neutrophils’ with an arrow going down. It doesn’t count. You have to write ‘neutropenia.’ If you write ‘admit for chemo,’ you can write ‘admit for chemo, dehydration, chronic blood loss anemia’ [as appropriate] and your severity index will go up a lot," said Dr. Smith, adding that each site of metastases should be listed because "you get points for each one."
Similarly, " ‘chest x-ray with pneumonitis status post radiation therapy’ does not count. You have to write ‘radiation pneumonitis.’ [And] don’t put ‘total protein and albumin low.’ Put ‘cachexia or malnutrition, moderate or severe.’ Don’t write ‘admit for pain control.’ Write ‘intractable pain form malignancy,’ and you have to say ‘lung cancer, breast cancer, bone cancer, liver cancer’ [accordingly]," he said.
Such diagnoses only have to be written once during the admission to count, he noted.
The approach worked for Massey Cancer Center’s palliative care unit.
"In about an 18- to 24-month period, we paid a lot attention to having most of the attendings and the fellows write out diagnostic statements, rather than medical thoughts," Dr. Smith said.
"When we started, our actual-vs.-expected mortality ratio was 150% higher than what it was supposed to be. We didn’t change the mortality rate one bit, but we changed the expected mortality. It came down to 55%, which is still high, but, heck, it’s a palliative care unit. A lot of people are sick and they’re going to die," he said.
"The APR-DRG [All Patient Refined Diagnosis Related Groups] severity of illness doubled just by paying attention to having those diagnostic statements put in," he said.
Dr. Smith gave an example of how the strategy also increases payments: An elderly person was admitted upon diagnosis of urosepsis, dehydration, and chronic obstructive pulmonary disease, but the patient’s condition allowed for more, including urinary tract infection; malnutrition; preexisting decubitus ulcer; and shock because the patient was a bit hypotensive.
With the additional diagnoses, the patient’s severity of illness (SOI) weighting increased from 0.5973 to 3.3739, resulting in a $5,613 payment increase.
"That’s a huge difference," Dr. Smith said.
He had a final tip for improving Hospital Compare mortality scores.
The CMS counts 30-day mortality, but the 30-day clock doesn’t start until 24 hours after admission. "So if you can enroll a Medicare patient in hospice within 24 hours of them hitting your emergency room door, then they don’t count towards your 30 day mortality," Dr. Smith said.
To help, "you can post somebody in the ER and make the ER aware that this is important, so that you can see those patients in the ER. Let your social workers know that the 24 shot-clock is ticking," Dr. Smith said.
The goal is to "accelerate the process of patients and families making choices. You can push that a little bit," he said.
Dr. Smith said he has no relevant disclosures.
VANCOUVER, B.C. - To improve hospital mortality scores, clinicians should include all possible diagnoses in patient charts, according to oncologist and palliative care specialist Dr. Thomas J. Smith.
U.S. News & World Report’s "Best Hospitals," the Centers for Medicaid and Medicare Services’ Hospital Compare, and other hospital rating programs compare hospital outcomes to national averages based on how sick patients are, said Dr. Smith, medical director of palliative care at Virginia Commonwealth University Massey Cancer Center in Richmond.
"You get counted on your observed mortality rate. They compare that to how many people are expected to die" according to the averages, he said at the annual meeting of the American Academy of Hospice and Palliative Care Medicine. Deaths in excess of the averages are considered unexpected and are assumed to be caused by poor quality care, which lowers hospitals in the mortality rankings.
By listing all possible diagnoses – especially at the end of life – "[you increase] your expected mortality, so that your observed-to-expected ratio improves. Even if you ignore improving mortality, if you change [how many people are expected to die] you’ll improve your ratio" and hospital standing, he said.
The reason is that hospital coders pull the diagnoses off patient charts and include them in claims submissions, the data upon which the rankings are based.
The approach is not about "gaming the system," Dr. Smith said. "You are adequately documenting the severity of the patients that you take care of" to ensure accurate mortality scores.
The key is to list diagnoses, not "medical thoughts," he said.
For example, " ‘lytic lesion of vertebra on spine films’ does not count. Write ‘bone mets’ [because] your coders can count that. Don’t write ‘neutrophils’ with an arrow going down. It doesn’t count. You have to write ‘neutropenia.’ If you write ‘admit for chemo,’ you can write ‘admit for chemo, dehydration, chronic blood loss anemia’ [as appropriate] and your severity index will go up a lot," said Dr. Smith, adding that each site of metastases should be listed because "you get points for each one."
Similarly, " ‘chest x-ray with pneumonitis status post radiation therapy’ does not count. You have to write ‘radiation pneumonitis.’ [And] don’t put ‘total protein and albumin low.’ Put ‘cachexia or malnutrition, moderate or severe.’ Don’t write ‘admit for pain control.’ Write ‘intractable pain form malignancy,’ and you have to say ‘lung cancer, breast cancer, bone cancer, liver cancer’ [accordingly]," he said.
Such diagnoses only have to be written once during the admission to count, he noted.
The approach worked for Massey Cancer Center’s palliative care unit.
"In about an 18- to 24-month period, we paid a lot attention to having most of the attendings and the fellows write out diagnostic statements, rather than medical thoughts," Dr. Smith said.
"When we started, our actual-vs.-expected mortality ratio was 150% higher than what it was supposed to be. We didn’t change the mortality rate one bit, but we changed the expected mortality. It came down to 55%, which is still high, but, heck, it’s a palliative care unit. A lot of people are sick and they’re going to die," he said.
"The APR-DRG [All Patient Refined Diagnosis Related Groups] severity of illness doubled just by paying attention to having those diagnostic statements put in," he said.
Dr. Smith gave an example of how the strategy also increases payments: An elderly person was admitted upon diagnosis of urosepsis, dehydration, and chronic obstructive pulmonary disease, but the patient’s condition allowed for more, including urinary tract infection; malnutrition; preexisting decubitus ulcer; and shock because the patient was a bit hypotensive.
With the additional diagnoses, the patient’s severity of illness (SOI) weighting increased from 0.5973 to 3.3739, resulting in a $5,613 payment increase.
"That’s a huge difference," Dr. Smith said.
He had a final tip for improving Hospital Compare mortality scores.
The CMS counts 30-day mortality, but the 30-day clock doesn’t start until 24 hours after admission. "So if you can enroll a Medicare patient in hospice within 24 hours of them hitting your emergency room door, then they don’t count towards your 30 day mortality," Dr. Smith said.
To help, "you can post somebody in the ER and make the ER aware that this is important, so that you can see those patients in the ER. Let your social workers know that the 24 shot-clock is ticking," Dr. Smith said.
The goal is to "accelerate the process of patients and families making choices. You can push that a little bit," he said.
Dr. Smith said he has no relevant disclosures.
FROM THE ANNUAL MEETING OF THE AMERICAN ACADEMY OF HOSPICE AND PALLIATIVE CARE MEDICINE
When Death is Sought: Preparing the Caregiver
Euthanasia “is never discussed openly, but the topic is out there and it’s terrifying,” Mrs. Ebert said during a roundtable discussion titled “The Many Faces and Challenges of Caregivers” led by veteran ABC news journalist Sam Donaldson at the annual conference of the National Comprehensive Cancer Network (NCCN) in Hollywood, Florida, last week. With respect to her husband, “here’s someone whose whole life was about speaking and sharing information, and he lost his ability to speak. He was also someone who loved food and eating with his friends and family and he wasn’t going to be able to eat.”
Up until that point, “I would have done anything for him. I didn’t know how to respond. I couldn’t go there,” Mrs. Pausch said. “I later learned that this is something that a lot of patients talk to their caregivers about, that it’s a normal part of the process, but it’s something that nobody tells you.”
—Diana Mahoney (on Twitter @DMPM1)
Euthanasia “is never discussed openly, but the topic is out there and it’s terrifying,” Mrs. Ebert said during a roundtable discussion titled “The Many Faces and Challenges of Caregivers” led by veteran ABC news journalist Sam Donaldson at the annual conference of the National Comprehensive Cancer Network (NCCN) in Hollywood, Florida, last week. With respect to her husband, “here’s someone whose whole life was about speaking and sharing information, and he lost his ability to speak. He was also someone who loved food and eating with his friends and family and he wasn’t going to be able to eat.”
Up until that point, “I would have done anything for him. I didn’t know how to respond. I couldn’t go there,” Mrs. Pausch said. “I later learned that this is something that a lot of patients talk to their caregivers about, that it’s a normal part of the process, but it’s something that nobody tells you.”
—Diana Mahoney (on Twitter @DMPM1)
Euthanasia “is never discussed openly, but the topic is out there and it’s terrifying,” Mrs. Ebert said during a roundtable discussion titled “The Many Faces and Challenges of Caregivers” led by veteran ABC news journalist Sam Donaldson at the annual conference of the National Comprehensive Cancer Network (NCCN) in Hollywood, Florida, last week. With respect to her husband, “here’s someone whose whole life was about speaking and sharing information, and he lost his ability to speak. He was also someone who loved food and eating with his friends and family and he wasn’t going to be able to eat.”
Up until that point, “I would have done anything for him. I didn’t know how to respond. I couldn’t go there,” Mrs. Pausch said. “I later learned that this is something that a lot of patients talk to their caregivers about, that it’s a normal part of the process, but it’s something that nobody tells you.”
—Diana Mahoney (on Twitter @DMPM1)