User login
CMS loosens clinician scope-of-practice, telehealth rules for COVID-19 crisis
To boost the capacity of frontline clinicians and facilities to fight COVID-19, the Centers for Medicare & Medicaid Services (CMS) on Thursday announced it is temporarily suspending rules to allow physicians to provide telehealth services across state lines, and will permit midlevel practitioners to provide as much care as their state licenses allow.
Physicians can now care for patients at rural hospitals across state lines via phone, radio, or online communications without having to be physically present.
“Remotely located physicians, coordinating with nurse practitioners at rural hospitals, will provide staffs at such facilities additional flexibility to meet the needs of their patients,” a CMS news release said.
At skilled nursing facilities, nurse practitioners will now be able to perform some medical exams that doctors normally conduct on Medicare patients, whether they are COVID-19-related or not, CMS said.
Occupational therapists from home health agencies can now perform initial assessments on certain homebound patients, allowing home health services to start sooner and freeing home health nurses to do more direct patient care.
In addition, hospice nurses will be relieved of hospice aide in-service training tasks so they can spend more time with patients.
“It’s all hands on deck during this crisis,” said CMS Administrator Seema Verma in the press release. “All frontline medical professionals need to be able to work at the highest level they were trained for. CMS is making sure there are no regulatory obstacles to increasing the medical workforce to handle the patient surge during the COVID-19 pandemic.”
The announcement did not directly address the question of whether CMS’ new telemedicine and scope-of-practice policies override state laws. The agency said, “CMS sets and enforces essential quality and safety standards that supplement state scope-of-practice and licensure laws for healthcare workers. CMS has continuously examined its regulations to identify areas where federal requirements may be more stringent than state laws and requirements.”
On March 20, Vice President Pence announced that physicians would be allowed to practice across state lines during the COVID-19 crisis, as reported by Medscape Medical News. Until now, however, CMS had not changed its regulations to allow doctors to conduct telehealth consultations in states other than the ones in which they are licensed.
Other Changes
As part of other rule changes to support the healthcare workforce, CMS said on March 30 that it will pay for more than 80 additional services when furnished via telehealth.
These include emergency department visits, initial skilled nursing facility and discharge visits, and home visits. In addition, the agency said it would cover phone visits with Medicare beneficiaries.
Moreover, while virtual “check-in” visits had previously been limited to established patients, CMS said that doctors would be able to provide these services to both new and established patients.
Among its other regulatory changes in recent weeks, CMS has also temporarily:
- Permitted physicians whose privileges will expire to continue practicing at a hospital, and allowed new physicians to begin working prior to full hospital medical staff/governing body review and approval
- Lifted regulatory requirements regarding hospital personnel qualified to perform specific respiratory care procedures, allowing these professionals to operate to the fullest extent of their licensure
- Waived federal minimum personnel qualifications for clinical nurse specialists, nurse practitioners, and physician assistants so they can work at rural hospitals as long as they meet state licensure requirements
- Allowed physicians and nonphysician practitioners to use telehealth to care for patients at long-term care facilities, rather than having to treat patients at those facilities in person
This article first appeared on Medscape.com.
To boost the capacity of frontline clinicians and facilities to fight COVID-19, the Centers for Medicare & Medicaid Services (CMS) on Thursday announced it is temporarily suspending rules to allow physicians to provide telehealth services across state lines, and will permit midlevel practitioners to provide as much care as their state licenses allow.
Physicians can now care for patients at rural hospitals across state lines via phone, radio, or online communications without having to be physically present.
“Remotely located physicians, coordinating with nurse practitioners at rural hospitals, will provide staffs at such facilities additional flexibility to meet the needs of their patients,” a CMS news release said.
At skilled nursing facilities, nurse practitioners will now be able to perform some medical exams that doctors normally conduct on Medicare patients, whether they are COVID-19-related or not, CMS said.
Occupational therapists from home health agencies can now perform initial assessments on certain homebound patients, allowing home health services to start sooner and freeing home health nurses to do more direct patient care.
In addition, hospice nurses will be relieved of hospice aide in-service training tasks so they can spend more time with patients.
“It’s all hands on deck during this crisis,” said CMS Administrator Seema Verma in the press release. “All frontline medical professionals need to be able to work at the highest level they were trained for. CMS is making sure there are no regulatory obstacles to increasing the medical workforce to handle the patient surge during the COVID-19 pandemic.”
The announcement did not directly address the question of whether CMS’ new telemedicine and scope-of-practice policies override state laws. The agency said, “CMS sets and enforces essential quality and safety standards that supplement state scope-of-practice and licensure laws for healthcare workers. CMS has continuously examined its regulations to identify areas where federal requirements may be more stringent than state laws and requirements.”
On March 20, Vice President Pence announced that physicians would be allowed to practice across state lines during the COVID-19 crisis, as reported by Medscape Medical News. Until now, however, CMS had not changed its regulations to allow doctors to conduct telehealth consultations in states other than the ones in which they are licensed.
Other Changes
As part of other rule changes to support the healthcare workforce, CMS said on March 30 that it will pay for more than 80 additional services when furnished via telehealth.
These include emergency department visits, initial skilled nursing facility and discharge visits, and home visits. In addition, the agency said it would cover phone visits with Medicare beneficiaries.
Moreover, while virtual “check-in” visits had previously been limited to established patients, CMS said that doctors would be able to provide these services to both new and established patients.
Among its other regulatory changes in recent weeks, CMS has also temporarily:
- Permitted physicians whose privileges will expire to continue practicing at a hospital, and allowed new physicians to begin working prior to full hospital medical staff/governing body review and approval
- Lifted regulatory requirements regarding hospital personnel qualified to perform specific respiratory care procedures, allowing these professionals to operate to the fullest extent of their licensure
- Waived federal minimum personnel qualifications for clinical nurse specialists, nurse practitioners, and physician assistants so they can work at rural hospitals as long as they meet state licensure requirements
- Allowed physicians and nonphysician practitioners to use telehealth to care for patients at long-term care facilities, rather than having to treat patients at those facilities in person
This article first appeared on Medscape.com.
To boost the capacity of frontline clinicians and facilities to fight COVID-19, the Centers for Medicare & Medicaid Services (CMS) on Thursday announced it is temporarily suspending rules to allow physicians to provide telehealth services across state lines, and will permit midlevel practitioners to provide as much care as their state licenses allow.
Physicians can now care for patients at rural hospitals across state lines via phone, radio, or online communications without having to be physically present.
“Remotely located physicians, coordinating with nurse practitioners at rural hospitals, will provide staffs at such facilities additional flexibility to meet the needs of their patients,” a CMS news release said.
At skilled nursing facilities, nurse practitioners will now be able to perform some medical exams that doctors normally conduct on Medicare patients, whether they are COVID-19-related or not, CMS said.
Occupational therapists from home health agencies can now perform initial assessments on certain homebound patients, allowing home health services to start sooner and freeing home health nurses to do more direct patient care.
In addition, hospice nurses will be relieved of hospice aide in-service training tasks so they can spend more time with patients.
“It’s all hands on deck during this crisis,” said CMS Administrator Seema Verma in the press release. “All frontline medical professionals need to be able to work at the highest level they were trained for. CMS is making sure there are no regulatory obstacles to increasing the medical workforce to handle the patient surge during the COVID-19 pandemic.”
The announcement did not directly address the question of whether CMS’ new telemedicine and scope-of-practice policies override state laws. The agency said, “CMS sets and enforces essential quality and safety standards that supplement state scope-of-practice and licensure laws for healthcare workers. CMS has continuously examined its regulations to identify areas where federal requirements may be more stringent than state laws and requirements.”
On March 20, Vice President Pence announced that physicians would be allowed to practice across state lines during the COVID-19 crisis, as reported by Medscape Medical News. Until now, however, CMS had not changed its regulations to allow doctors to conduct telehealth consultations in states other than the ones in which they are licensed.
Other Changes
As part of other rule changes to support the healthcare workforce, CMS said on March 30 that it will pay for more than 80 additional services when furnished via telehealth.
These include emergency department visits, initial skilled nursing facility and discharge visits, and home visits. In addition, the agency said it would cover phone visits with Medicare beneficiaries.
Moreover, while virtual “check-in” visits had previously been limited to established patients, CMS said that doctors would be able to provide these services to both new and established patients.
Among its other regulatory changes in recent weeks, CMS has also temporarily:
- Permitted physicians whose privileges will expire to continue practicing at a hospital, and allowed new physicians to begin working prior to full hospital medical staff/governing body review and approval
- Lifted regulatory requirements regarding hospital personnel qualified to perform specific respiratory care procedures, allowing these professionals to operate to the fullest extent of their licensure
- Waived federal minimum personnel qualifications for clinical nurse specialists, nurse practitioners, and physician assistants so they can work at rural hospitals as long as they meet state licensure requirements
- Allowed physicians and nonphysician practitioners to use telehealth to care for patients at long-term care facilities, rather than having to treat patients at those facilities in person
This article first appeared on Medscape.com.
What do early remdesivir data suggest?
New data on the investigational antiviral drug remdesivir (Gilead) suggest clinical improvement in 36 of 53 patients (68%) hospitalized for severe COVID-19, according to a new study published online April 10 in the New England Journal of Medicine.
But experts are warning that these data come from compassionate use in a wide variety of patients, with no randomization and no control group.
“It is impossible to know the outcome for this relatively small group of patients had they not received remdesivir,” commented Stephen Griffin, PhD, associate professor at the University of Leeds School of Medicine, United Kingdom, who was not involved with the study.
“As the authors point out, a randomized clinical trial is necessary to determine the true effectiveness of this drug,” Griffin added in comments he provided to the Science Media Centre in London. Such trials are underway.
“The data from this paper are almost uninterpretable,” said Stephen Evans, MSc, FRCP, professor of pharmacoepidemiology, London School of Hygiene & Tropical Medicine, who provided comments to the Science Media Centre.
Evans notes that the authors describe multiple caveats that limit interpretation of the results, including the small sample size, the relatively short follow-up, missing data, no follow-up on eight patients, and lack of a randomized control group.
Meanwhile, Josh Farkas, MD, who writes the PulmCrit blog, details his criticisms in a piece entitled, “Eleven reasons the NEJM paper on remdesivir reveals nothing.” Beyond the issues the authors list, he points out several more, including cherry picking of patients. “Remdesivir was aggressively sought-after by thousands of patients with COVID-19,” he writes. “Of these patients, 61 ended up receiving the drug. Why did these patients receive medication, out of scores of patients applying to receive it?”
Also, there are no follow-up data for 8 of the 61 patients who received an initial dose of the drug, leaving 53 for the published analysis, continues Farkas, who is an assistant professor of pulmonary and critical care medicine at the University of Vermont in Burlington.
“What happened to these patients? Did they die from anaphylaxis? Did they get well, sign out against medical advice, and go party? This is unknown — but I’m worried that these patients actually didn’t fare so well,” Farkas writes.
Farkas, like Evans and Griffin, concludes that the data are largely unusable. “Until [a randomized controlled trial] is performed, further compassionate use of remdesivir probably isn’t justified,” he writes.
Data from Compassionate Use Program
The data in the NEJM article come from a compassionate use program set up by Gilead. The company says it has provided emergency access to remdesivir for several hundred patients in the United States, Europe, and Japan.
The authors, led by Jonathan Grein, MD, from Cedars–Sinai Medical Center, Los Angeles, California, report on 61 patients who received remdesivir as part of this program.
The authors, several of whom are employees of Gilead, note that data on 8 patients could not be analyzed (including 7 patients with no posttreatment data and 1 with a dosing error).
Of the 53 patients whose data were included, 22 were in the United States, 22 in Europe or Canada, and 9 in Japan.
These were patients hospitalized with COVID-19 who had confirmed SARS-CoV-2 infection and had an oxygen saturation of 94% or less while they were breathing ambient air, or who were receiving oxygen support.
Patients received a 10-day course of remdesivir, consisting of 200 mg administered intravenously on day 1, followed by 100 mg daily for the remaining 9 days of treatment.
At baseline, 30 patients (57%) were receiving mechanical ventilation and 4 (8%) were receiving extracorporeal membrane oxygenation.
During a median follow-up of 18 days, 36 patients (68%) had an improvement in oxygen-support class, including 17 (57%) of 30 patients receiving mechanical ventilation who were extubated.
A total of 25 patients (47%) were discharged, and 7 patients (13%) died; mortality was 18% (6 of 34) among patients receiving invasive ventilation and 5% (1 of 19) among those not receiving invasive ventilation.
While the authors acknowledge limitations of the data they collected, they nevertheless comment that “comparisons with contemporaneous cohorts from the literature, in whom general care is expected to be consistent with that of our cohort, suggest that remdesivir may have clinical benefit in patients with severe COVID-19.”
“Currently there is no proven treatment for COVID-19. We cannot draw definitive conclusions from these data, but the observations from this group of hospitalized patients who received remdesivir are hopeful,” said Grein in a Cedars–Sinai press release. “We look forward to the results of controlled clinical trials to potentially validate these findings.”
Experts are not convinced, however.
“The drug was being used in patients who were severely ill, but reporting on 61 out of several hundred makes it clear that generalizations about the efficacy and safety must be treated with great caution,” said Evans. “There is some evidence suggesting efficacy, but we simply do not know what would have happened to these patients had they not been given the drug.”
“I would say it’s impossible to discern whether there is a treatment effect or not,” said Duncan Richards, MA, DM, FRCP, clinical pharmacologist and professor of clinical therapeutics, University of Oxford, UK. “This is in part due to the mixed patient population, ranging from those needing low dose oxygen, who are more likely to survive anyway, to much more severe cases ... [who] show a much more mixed picture.”
“There are ongoing large international randomized controlled trials with remdesivir — we really need to see those data, “ he said in comments to Science Media Centre. “Safe and effective treatments for COVID-19 are critically needed and should be expedited wherever possible, but it’s important not to compromise on the quality of the research.”
Multiple coauthors are employees of Gilead, the company developing remdesivir. Griffin, Evans, and Farkas have disclosed no relevant financial relationships. Richards consults for GlaxoSmithKline in the field of drug safety. GSK does not manufacture any of the products mentioned.
N Engl J Med. 2020 Apr 10. Full text.
This article first appeared on Medscape.com.
New data on the investigational antiviral drug remdesivir (Gilead) suggest clinical improvement in 36 of 53 patients (68%) hospitalized for severe COVID-19, according to a new study published online April 10 in the New England Journal of Medicine.
But experts are warning that these data come from compassionate use in a wide variety of patients, with no randomization and no control group.
“It is impossible to know the outcome for this relatively small group of patients had they not received remdesivir,” commented Stephen Griffin, PhD, associate professor at the University of Leeds School of Medicine, United Kingdom, who was not involved with the study.
“As the authors point out, a randomized clinical trial is necessary to determine the true effectiveness of this drug,” Griffin added in comments he provided to the Science Media Centre in London. Such trials are underway.
“The data from this paper are almost uninterpretable,” said Stephen Evans, MSc, FRCP, professor of pharmacoepidemiology, London School of Hygiene & Tropical Medicine, who provided comments to the Science Media Centre.
Evans notes that the authors describe multiple caveats that limit interpretation of the results, including the small sample size, the relatively short follow-up, missing data, no follow-up on eight patients, and lack of a randomized control group.
Meanwhile, Josh Farkas, MD, who writes the PulmCrit blog, details his criticisms in a piece entitled, “Eleven reasons the NEJM paper on remdesivir reveals nothing.” Beyond the issues the authors list, he points out several more, including cherry picking of patients. “Remdesivir was aggressively sought-after by thousands of patients with COVID-19,” he writes. “Of these patients, 61 ended up receiving the drug. Why did these patients receive medication, out of scores of patients applying to receive it?”
Also, there are no follow-up data for 8 of the 61 patients who received an initial dose of the drug, leaving 53 for the published analysis, continues Farkas, who is an assistant professor of pulmonary and critical care medicine at the University of Vermont in Burlington.
“What happened to these patients? Did they die from anaphylaxis? Did they get well, sign out against medical advice, and go party? This is unknown — but I’m worried that these patients actually didn’t fare so well,” Farkas writes.
Farkas, like Evans and Griffin, concludes that the data are largely unusable. “Until [a randomized controlled trial] is performed, further compassionate use of remdesivir probably isn’t justified,” he writes.
Data from Compassionate Use Program
The data in the NEJM article come from a compassionate use program set up by Gilead. The company says it has provided emergency access to remdesivir for several hundred patients in the United States, Europe, and Japan.
The authors, led by Jonathan Grein, MD, from Cedars–Sinai Medical Center, Los Angeles, California, report on 61 patients who received remdesivir as part of this program.
The authors, several of whom are employees of Gilead, note that data on 8 patients could not be analyzed (including 7 patients with no posttreatment data and 1 with a dosing error).
Of the 53 patients whose data were included, 22 were in the United States, 22 in Europe or Canada, and 9 in Japan.
These were patients hospitalized with COVID-19 who had confirmed SARS-CoV-2 infection and had an oxygen saturation of 94% or less while they were breathing ambient air, or who were receiving oxygen support.
Patients received a 10-day course of remdesivir, consisting of 200 mg administered intravenously on day 1, followed by 100 mg daily for the remaining 9 days of treatment.
At baseline, 30 patients (57%) were receiving mechanical ventilation and 4 (8%) were receiving extracorporeal membrane oxygenation.
During a median follow-up of 18 days, 36 patients (68%) had an improvement in oxygen-support class, including 17 (57%) of 30 patients receiving mechanical ventilation who were extubated.
A total of 25 patients (47%) were discharged, and 7 patients (13%) died; mortality was 18% (6 of 34) among patients receiving invasive ventilation and 5% (1 of 19) among those not receiving invasive ventilation.
While the authors acknowledge limitations of the data they collected, they nevertheless comment that “comparisons with contemporaneous cohorts from the literature, in whom general care is expected to be consistent with that of our cohort, suggest that remdesivir may have clinical benefit in patients with severe COVID-19.”
“Currently there is no proven treatment for COVID-19. We cannot draw definitive conclusions from these data, but the observations from this group of hospitalized patients who received remdesivir are hopeful,” said Grein in a Cedars–Sinai press release. “We look forward to the results of controlled clinical trials to potentially validate these findings.”
Experts are not convinced, however.
“The drug was being used in patients who were severely ill, but reporting on 61 out of several hundred makes it clear that generalizations about the efficacy and safety must be treated with great caution,” said Evans. “There is some evidence suggesting efficacy, but we simply do not know what would have happened to these patients had they not been given the drug.”
“I would say it’s impossible to discern whether there is a treatment effect or not,” said Duncan Richards, MA, DM, FRCP, clinical pharmacologist and professor of clinical therapeutics, University of Oxford, UK. “This is in part due to the mixed patient population, ranging from those needing low dose oxygen, who are more likely to survive anyway, to much more severe cases ... [who] show a much more mixed picture.”
“There are ongoing large international randomized controlled trials with remdesivir — we really need to see those data, “ he said in comments to Science Media Centre. “Safe and effective treatments for COVID-19 are critically needed and should be expedited wherever possible, but it’s important not to compromise on the quality of the research.”
Multiple coauthors are employees of Gilead, the company developing remdesivir. Griffin, Evans, and Farkas have disclosed no relevant financial relationships. Richards consults for GlaxoSmithKline in the field of drug safety. GSK does not manufacture any of the products mentioned.
N Engl J Med. 2020 Apr 10. Full text.
This article first appeared on Medscape.com.
New data on the investigational antiviral drug remdesivir (Gilead) suggest clinical improvement in 36 of 53 patients (68%) hospitalized for severe COVID-19, according to a new study published online April 10 in the New England Journal of Medicine.
But experts are warning that these data come from compassionate use in a wide variety of patients, with no randomization and no control group.
“It is impossible to know the outcome for this relatively small group of patients had they not received remdesivir,” commented Stephen Griffin, PhD, associate professor at the University of Leeds School of Medicine, United Kingdom, who was not involved with the study.
“As the authors point out, a randomized clinical trial is necessary to determine the true effectiveness of this drug,” Griffin added in comments he provided to the Science Media Centre in London. Such trials are underway.
“The data from this paper are almost uninterpretable,” said Stephen Evans, MSc, FRCP, professor of pharmacoepidemiology, London School of Hygiene & Tropical Medicine, who provided comments to the Science Media Centre.
Evans notes that the authors describe multiple caveats that limit interpretation of the results, including the small sample size, the relatively short follow-up, missing data, no follow-up on eight patients, and lack of a randomized control group.
Meanwhile, Josh Farkas, MD, who writes the PulmCrit blog, details his criticisms in a piece entitled, “Eleven reasons the NEJM paper on remdesivir reveals nothing.” Beyond the issues the authors list, he points out several more, including cherry picking of patients. “Remdesivir was aggressively sought-after by thousands of patients with COVID-19,” he writes. “Of these patients, 61 ended up receiving the drug. Why did these patients receive medication, out of scores of patients applying to receive it?”
Also, there are no follow-up data for 8 of the 61 patients who received an initial dose of the drug, leaving 53 for the published analysis, continues Farkas, who is an assistant professor of pulmonary and critical care medicine at the University of Vermont in Burlington.
“What happened to these patients? Did they die from anaphylaxis? Did they get well, sign out against medical advice, and go party? This is unknown — but I’m worried that these patients actually didn’t fare so well,” Farkas writes.
Farkas, like Evans and Griffin, concludes that the data are largely unusable. “Until [a randomized controlled trial] is performed, further compassionate use of remdesivir probably isn’t justified,” he writes.
Data from Compassionate Use Program
The data in the NEJM article come from a compassionate use program set up by Gilead. The company says it has provided emergency access to remdesivir for several hundred patients in the United States, Europe, and Japan.
The authors, led by Jonathan Grein, MD, from Cedars–Sinai Medical Center, Los Angeles, California, report on 61 patients who received remdesivir as part of this program.
The authors, several of whom are employees of Gilead, note that data on 8 patients could not be analyzed (including 7 patients with no posttreatment data and 1 with a dosing error).
Of the 53 patients whose data were included, 22 were in the United States, 22 in Europe or Canada, and 9 in Japan.
These were patients hospitalized with COVID-19 who had confirmed SARS-CoV-2 infection and had an oxygen saturation of 94% or less while they were breathing ambient air, or who were receiving oxygen support.
Patients received a 10-day course of remdesivir, consisting of 200 mg administered intravenously on day 1, followed by 100 mg daily for the remaining 9 days of treatment.
At baseline, 30 patients (57%) were receiving mechanical ventilation and 4 (8%) were receiving extracorporeal membrane oxygenation.
During a median follow-up of 18 days, 36 patients (68%) had an improvement in oxygen-support class, including 17 (57%) of 30 patients receiving mechanical ventilation who were extubated.
A total of 25 patients (47%) were discharged, and 7 patients (13%) died; mortality was 18% (6 of 34) among patients receiving invasive ventilation and 5% (1 of 19) among those not receiving invasive ventilation.
While the authors acknowledge limitations of the data they collected, they nevertheless comment that “comparisons with contemporaneous cohorts from the literature, in whom general care is expected to be consistent with that of our cohort, suggest that remdesivir may have clinical benefit in patients with severe COVID-19.”
“Currently there is no proven treatment for COVID-19. We cannot draw definitive conclusions from these data, but the observations from this group of hospitalized patients who received remdesivir are hopeful,” said Grein in a Cedars–Sinai press release. “We look forward to the results of controlled clinical trials to potentially validate these findings.”
Experts are not convinced, however.
“The drug was being used in patients who were severely ill, but reporting on 61 out of several hundred makes it clear that generalizations about the efficacy and safety must be treated with great caution,” said Evans. “There is some evidence suggesting efficacy, but we simply do not know what would have happened to these patients had they not been given the drug.”
“I would say it’s impossible to discern whether there is a treatment effect or not,” said Duncan Richards, MA, DM, FRCP, clinical pharmacologist and professor of clinical therapeutics, University of Oxford, UK. “This is in part due to the mixed patient population, ranging from those needing low dose oxygen, who are more likely to survive anyway, to much more severe cases ... [who] show a much more mixed picture.”
“There are ongoing large international randomized controlled trials with remdesivir — we really need to see those data, “ he said in comments to Science Media Centre. “Safe and effective treatments for COVID-19 are critically needed and should be expedited wherever possible, but it’s important not to compromise on the quality of the research.”
Multiple coauthors are employees of Gilead, the company developing remdesivir. Griffin, Evans, and Farkas have disclosed no relevant financial relationships. Richards consults for GlaxoSmithKline in the field of drug safety. GSK does not manufacture any of the products mentioned.
N Engl J Med. 2020 Apr 10. Full text.
This article first appeared on Medscape.com.
Remdesivir tops list of promising COVID-19 treatments in review of nearly 300 trials
, according to authors of a recent review covering nearly 300 active clinical treatment trials underway for the disease.
Remdesivir, which has potent in vitro activity against the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), is not approved by the Food and Drug Administration and is currently being tested in randomized trials, according to the review authors, led by James M. Sanders, PhD, of the department of pharmacy at University of Texas Southwestern Medical Center in Dallas.
By contrast, oseltamivir has not demonstrated efficacy against the virus, corticosteroids are not recommended, and promising data from a small French hydroxychloroquine study are balanced by “several major limitations” including small sample size and exclusion of early dropouts from the analysis, among others, Dr. Sanders and coauthors said in their report.
“These limitations coupled with concerns of additive cardiotoxicity with combination therapy [i.e., hydroxychloroquine with azithromycin] do not support adoption of this regimen without additional studies,” the researchers wrote. Their report is in JAMA.
Dr. Sanders and colleagues identified 291 COVID-19–specific studies listed in ClinicalTrials.gov through April 2, including 29 placebo-controlled trials.
This might represent just a sliver of the treatments that could combat COVID-19, according to the researchers, who said more than 3,000 small-molecule drug candidates with potential activity against human coronaviruses have been identified.
“This large amount of potential agents will hopefully yield more candidate therapeutics in the race to find effective treatments or preventive strategies against COVID-19,” said Dr. Sanders and coauthors.
Remdesivir for COVID-19
Remdesivir, an investigational nucleotide analog, is one promising agent because of its broad-spectrum and potent activity against SARS-CoV-2 and other novel coronaviruses, they said, adding that phase 1 trials demonstrated the drug was well tolerated without observed liver or kidney toxicity.
There have been “successful” case reports of remdesivir use in COVID-19, and at least five ongoing clinical trials are evaluating the drug’s safety and antiviral activity in this disease. Among those studies is a National Institutes of Health–sponsored adaptive, randomized, placebo-controlled trial that will provide data on the use of remdesivir versus supportive care.
“As the results from randomized controlled trials are anticipated, inclusion of this agent for treatment of COVID-19 may be considered,” Dr. Sanders and colleagues wrote in their report. To date, remdesivir remains investigational and needs to be obtained via compassionate use, through expanded access, or by participating in a clinical trial, they added.
Hydroxychloroquine and chloroquine
Among the published hydroxychloroquine studies is a “promising” 36-patient open-label nonrandomized French study, in which the antimalarial agent given every 8 hours improved virologic clearance by day 6 versus controls (70% vs. 12.5%, respectively), the review authors said. Moreover, viral clearance was 100% for 6 patients who received hydroxychloroquine plus azithromycin, compared to 57% (8 of 14) for patients treated with hydroxychloroquine alone. However, that study had several important limitations, including the small sample size, variable viral loads at baseline between groups, and a lack of safety and clinical outcomes reporting, according to the investigators. Moreover, six patients in the hydroxychloroquine group were taken out of the analysis because of early treatment stoppage due to medical intolerance or critical illness, the authors noted.
One prospective study including 30 patients in China demonstrated no difference in virologic outcomes for patients randomized to hydroxychloroquine plus standard of care versus standard of care alone, they added. There is also a case series of more than 100 patients with COVID-19 that reportedly improved viral clearance and reduced disease progression, though they said results haven’t been published or presented beyond a news briefing in China.
Randomized, controlled trials of chloroquine and hydroxychloroquine for COVID-19 treatment are underway, and studies are planned or enrolling to look at chloroquine prophylaxis in health care personnel and hydroxychloroquine for postexposure prophylaxis, authors said.
In results from one of those randomized trials, just reported, a higher dose of chloroquine was associated with a cardiac adverse event and an increased mortality risk, leading to the closure of that study arm. In the parallel, double-blinded, phase IIb clinical trial, patients in Brazil with SARS-CoV-2 infection received low or high doses of chloroquine plus ceftriaxone and azithromycin. According to the preprint publication, a higher rate of heart rate–corrected QT interval (QTc) prolongation and a “trend toward higher lethality” was observed in the high-dose group, leading investigators to “strongly recommend” the higher dose be abandoned.
“No apparent benefit of chloroquine was seen regarding lethality in our patients so far, but we will still enroll patients in the low chloroquine dose group to complete the originally planned sample size,” said investigators of the study, which at the time of the report had enrolled 81 out of an anticipated 440 patients.
Other COVID-19 pharmacologic therapies under study
Treatments of note in the review included the following:
- Tocilizumab. This monoclonal antibody IL-6 receptor antagonist, approved by the FDA for treatment of rheumatoid arthritis and for cytokine release syndrome related to chimeric antigen receptor (CAR) T-cell therapy, has yielded success in small series of patients with severe cases of COVID-19, according to authors. In one 21-patient report, 91% had clinical improvement, usually after a single dose. In China, tocilizumab is included in COVID-19 treatment guidelines, and several randomized clinical trials are underway in China including patients with COVID-19 with severe pneumonia.
- Immunoglobulin therapy. Antibodies from recovered COVID-19 patients could help with free virus and infected cell immune clearance, the authors said, adding that further studies are warranted beyond a few small published case series that suggest promise. Furthermore, on March 24 the FDA released guidance for screening donors for COVID-19 convalescent plasma and on emergency investigational new drug applications based on this modality.
- Lopinavir/ritonavir. Despite demonstrated in vitro activity against other novel coronaviruses, there is no published in vitro data for lopinavir/ritonavir in SARS-CoV-2, and likely a “limited role” for this combination anticipated in treating COVID-19, according to the review authors. In an open-label randomized clinical trial published in the New England Journal of Medicine (2020 Mar 18. doi: 10.1056/NEJMoa2001282), there were no differences in clinical improvement, viral clearance, or mortality for antiviral treatment versus standard care. Delayed treatment initiation may explain the ineffectiveness, though a subgroup analysis didn’t show a shorter time to clinical improvement for those who got the treatment earlier.
- Ribavirin. Likewise, this antiviral medication has efficacy and safety data suggesting “limited value” for treatment of COVID-19. Treatment of SARS yielded “inconclusive results” for ribavirin, which was also associated with substantial toxicity that included hemolytic anemia in 60% of SARS patients.
- Oseltamivir. While it may treat influenza, it has no documented activity against SARS-CoV-2 in vitro: “This agent has no role in the management of COVID-19 once influenza has been excluded,” said Dr. Sanders and coauthors.
- Corticosteroids. They could decrease inflammatory responses in the lung, but they could also lead to delays in viral clearance and increases in secondary infection risk. Guidelines for COVID-19 say to avoid corticosteroids, and the authors of the review concur, saying that potential harms and lack of proven benefit mean they usually should not be used outside of a randomized clinical trial setting.
- Vaccines. Clearly, vaccines represent the “most effective long-term strategy” to prevent future COVID-19 outbreaks, though at least 12-18 months would be required until vaccines can be widely deployed, authors said.
Dr. Sanders reported no potential conflicts. Senior author James B. Cutrell, MD, also of the University of Texas Southwestern Medical Center, reported nonfinancial support from Gilead and Regeneron outside of the study. No other authors reported disclosures.
, according to authors of a recent review covering nearly 300 active clinical treatment trials underway for the disease.
Remdesivir, which has potent in vitro activity against the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), is not approved by the Food and Drug Administration and is currently being tested in randomized trials, according to the review authors, led by James M. Sanders, PhD, of the department of pharmacy at University of Texas Southwestern Medical Center in Dallas.
By contrast, oseltamivir has not demonstrated efficacy against the virus, corticosteroids are not recommended, and promising data from a small French hydroxychloroquine study are balanced by “several major limitations” including small sample size and exclusion of early dropouts from the analysis, among others, Dr. Sanders and coauthors said in their report.
“These limitations coupled with concerns of additive cardiotoxicity with combination therapy [i.e., hydroxychloroquine with azithromycin] do not support adoption of this regimen without additional studies,” the researchers wrote. Their report is in JAMA.
Dr. Sanders and colleagues identified 291 COVID-19–specific studies listed in ClinicalTrials.gov through April 2, including 29 placebo-controlled trials.
This might represent just a sliver of the treatments that could combat COVID-19, according to the researchers, who said more than 3,000 small-molecule drug candidates with potential activity against human coronaviruses have been identified.
“This large amount of potential agents will hopefully yield more candidate therapeutics in the race to find effective treatments or preventive strategies against COVID-19,” said Dr. Sanders and coauthors.
Remdesivir for COVID-19
Remdesivir, an investigational nucleotide analog, is one promising agent because of its broad-spectrum and potent activity against SARS-CoV-2 and other novel coronaviruses, they said, adding that phase 1 trials demonstrated the drug was well tolerated without observed liver or kidney toxicity.
There have been “successful” case reports of remdesivir use in COVID-19, and at least five ongoing clinical trials are evaluating the drug’s safety and antiviral activity in this disease. Among those studies is a National Institutes of Health–sponsored adaptive, randomized, placebo-controlled trial that will provide data on the use of remdesivir versus supportive care.
“As the results from randomized controlled trials are anticipated, inclusion of this agent for treatment of COVID-19 may be considered,” Dr. Sanders and colleagues wrote in their report. To date, remdesivir remains investigational and needs to be obtained via compassionate use, through expanded access, or by participating in a clinical trial, they added.
Hydroxychloroquine and chloroquine
Among the published hydroxychloroquine studies is a “promising” 36-patient open-label nonrandomized French study, in which the antimalarial agent given every 8 hours improved virologic clearance by day 6 versus controls (70% vs. 12.5%, respectively), the review authors said. Moreover, viral clearance was 100% for 6 patients who received hydroxychloroquine plus azithromycin, compared to 57% (8 of 14) for patients treated with hydroxychloroquine alone. However, that study had several important limitations, including the small sample size, variable viral loads at baseline between groups, and a lack of safety and clinical outcomes reporting, according to the investigators. Moreover, six patients in the hydroxychloroquine group were taken out of the analysis because of early treatment stoppage due to medical intolerance or critical illness, the authors noted.
One prospective study including 30 patients in China demonstrated no difference in virologic outcomes for patients randomized to hydroxychloroquine plus standard of care versus standard of care alone, they added. There is also a case series of more than 100 patients with COVID-19 that reportedly improved viral clearance and reduced disease progression, though they said results haven’t been published or presented beyond a news briefing in China.
Randomized, controlled trials of chloroquine and hydroxychloroquine for COVID-19 treatment are underway, and studies are planned or enrolling to look at chloroquine prophylaxis in health care personnel and hydroxychloroquine for postexposure prophylaxis, authors said.
In results from one of those randomized trials, just reported, a higher dose of chloroquine was associated with a cardiac adverse event and an increased mortality risk, leading to the closure of that study arm. In the parallel, double-blinded, phase IIb clinical trial, patients in Brazil with SARS-CoV-2 infection received low or high doses of chloroquine plus ceftriaxone and azithromycin. According to the preprint publication, a higher rate of heart rate–corrected QT interval (QTc) prolongation and a “trend toward higher lethality” was observed in the high-dose group, leading investigators to “strongly recommend” the higher dose be abandoned.
“No apparent benefit of chloroquine was seen regarding lethality in our patients so far, but we will still enroll patients in the low chloroquine dose group to complete the originally planned sample size,” said investigators of the study, which at the time of the report had enrolled 81 out of an anticipated 440 patients.
Other COVID-19 pharmacologic therapies under study
Treatments of note in the review included the following:
- Tocilizumab. This monoclonal antibody IL-6 receptor antagonist, approved by the FDA for treatment of rheumatoid arthritis and for cytokine release syndrome related to chimeric antigen receptor (CAR) T-cell therapy, has yielded success in small series of patients with severe cases of COVID-19, according to authors. In one 21-patient report, 91% had clinical improvement, usually after a single dose. In China, tocilizumab is included in COVID-19 treatment guidelines, and several randomized clinical trials are underway in China including patients with COVID-19 with severe pneumonia.
- Immunoglobulin therapy. Antibodies from recovered COVID-19 patients could help with free virus and infected cell immune clearance, the authors said, adding that further studies are warranted beyond a few small published case series that suggest promise. Furthermore, on March 24 the FDA released guidance for screening donors for COVID-19 convalescent plasma and on emergency investigational new drug applications based on this modality.
- Lopinavir/ritonavir. Despite demonstrated in vitro activity against other novel coronaviruses, there is no published in vitro data for lopinavir/ritonavir in SARS-CoV-2, and likely a “limited role” for this combination anticipated in treating COVID-19, according to the review authors. In an open-label randomized clinical trial published in the New England Journal of Medicine (2020 Mar 18. doi: 10.1056/NEJMoa2001282), there were no differences in clinical improvement, viral clearance, or mortality for antiviral treatment versus standard care. Delayed treatment initiation may explain the ineffectiveness, though a subgroup analysis didn’t show a shorter time to clinical improvement for those who got the treatment earlier.
- Ribavirin. Likewise, this antiviral medication has efficacy and safety data suggesting “limited value” for treatment of COVID-19. Treatment of SARS yielded “inconclusive results” for ribavirin, which was also associated with substantial toxicity that included hemolytic anemia in 60% of SARS patients.
- Oseltamivir. While it may treat influenza, it has no documented activity against SARS-CoV-2 in vitro: “This agent has no role in the management of COVID-19 once influenza has been excluded,” said Dr. Sanders and coauthors.
- Corticosteroids. They could decrease inflammatory responses in the lung, but they could also lead to delays in viral clearance and increases in secondary infection risk. Guidelines for COVID-19 say to avoid corticosteroids, and the authors of the review concur, saying that potential harms and lack of proven benefit mean they usually should not be used outside of a randomized clinical trial setting.
- Vaccines. Clearly, vaccines represent the “most effective long-term strategy” to prevent future COVID-19 outbreaks, though at least 12-18 months would be required until vaccines can be widely deployed, authors said.
Dr. Sanders reported no potential conflicts. Senior author James B. Cutrell, MD, also of the University of Texas Southwestern Medical Center, reported nonfinancial support from Gilead and Regeneron outside of the study. No other authors reported disclosures.
, according to authors of a recent review covering nearly 300 active clinical treatment trials underway for the disease.
Remdesivir, which has potent in vitro activity against the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), is not approved by the Food and Drug Administration and is currently being tested in randomized trials, according to the review authors, led by James M. Sanders, PhD, of the department of pharmacy at University of Texas Southwestern Medical Center in Dallas.
By contrast, oseltamivir has not demonstrated efficacy against the virus, corticosteroids are not recommended, and promising data from a small French hydroxychloroquine study are balanced by “several major limitations” including small sample size and exclusion of early dropouts from the analysis, among others, Dr. Sanders and coauthors said in their report.
“These limitations coupled with concerns of additive cardiotoxicity with combination therapy [i.e., hydroxychloroquine with azithromycin] do not support adoption of this regimen without additional studies,” the researchers wrote. Their report is in JAMA.
Dr. Sanders and colleagues identified 291 COVID-19–specific studies listed in ClinicalTrials.gov through April 2, including 29 placebo-controlled trials.
This might represent just a sliver of the treatments that could combat COVID-19, according to the researchers, who said more than 3,000 small-molecule drug candidates with potential activity against human coronaviruses have been identified.
“This large amount of potential agents will hopefully yield more candidate therapeutics in the race to find effective treatments or preventive strategies against COVID-19,” said Dr. Sanders and coauthors.
Remdesivir for COVID-19
Remdesivir, an investigational nucleotide analog, is one promising agent because of its broad-spectrum and potent activity against SARS-CoV-2 and other novel coronaviruses, they said, adding that phase 1 trials demonstrated the drug was well tolerated without observed liver or kidney toxicity.
There have been “successful” case reports of remdesivir use in COVID-19, and at least five ongoing clinical trials are evaluating the drug’s safety and antiviral activity in this disease. Among those studies is a National Institutes of Health–sponsored adaptive, randomized, placebo-controlled trial that will provide data on the use of remdesivir versus supportive care.
“As the results from randomized controlled trials are anticipated, inclusion of this agent for treatment of COVID-19 may be considered,” Dr. Sanders and colleagues wrote in their report. To date, remdesivir remains investigational and needs to be obtained via compassionate use, through expanded access, or by participating in a clinical trial, they added.
Hydroxychloroquine and chloroquine
Among the published hydroxychloroquine studies is a “promising” 36-patient open-label nonrandomized French study, in which the antimalarial agent given every 8 hours improved virologic clearance by day 6 versus controls (70% vs. 12.5%, respectively), the review authors said. Moreover, viral clearance was 100% for 6 patients who received hydroxychloroquine plus azithromycin, compared to 57% (8 of 14) for patients treated with hydroxychloroquine alone. However, that study had several important limitations, including the small sample size, variable viral loads at baseline between groups, and a lack of safety and clinical outcomes reporting, according to the investigators. Moreover, six patients in the hydroxychloroquine group were taken out of the analysis because of early treatment stoppage due to medical intolerance or critical illness, the authors noted.
One prospective study including 30 patients in China demonstrated no difference in virologic outcomes for patients randomized to hydroxychloroquine plus standard of care versus standard of care alone, they added. There is also a case series of more than 100 patients with COVID-19 that reportedly improved viral clearance and reduced disease progression, though they said results haven’t been published or presented beyond a news briefing in China.
Randomized, controlled trials of chloroquine and hydroxychloroquine for COVID-19 treatment are underway, and studies are planned or enrolling to look at chloroquine prophylaxis in health care personnel and hydroxychloroquine for postexposure prophylaxis, authors said.
In results from one of those randomized trials, just reported, a higher dose of chloroquine was associated with a cardiac adverse event and an increased mortality risk, leading to the closure of that study arm. In the parallel, double-blinded, phase IIb clinical trial, patients in Brazil with SARS-CoV-2 infection received low or high doses of chloroquine plus ceftriaxone and azithromycin. According to the preprint publication, a higher rate of heart rate–corrected QT interval (QTc) prolongation and a “trend toward higher lethality” was observed in the high-dose group, leading investigators to “strongly recommend” the higher dose be abandoned.
“No apparent benefit of chloroquine was seen regarding lethality in our patients so far, but we will still enroll patients in the low chloroquine dose group to complete the originally planned sample size,” said investigators of the study, which at the time of the report had enrolled 81 out of an anticipated 440 patients.
Other COVID-19 pharmacologic therapies under study
Treatments of note in the review included the following:
- Tocilizumab. This monoclonal antibody IL-6 receptor antagonist, approved by the FDA for treatment of rheumatoid arthritis and for cytokine release syndrome related to chimeric antigen receptor (CAR) T-cell therapy, has yielded success in small series of patients with severe cases of COVID-19, according to authors. In one 21-patient report, 91% had clinical improvement, usually after a single dose. In China, tocilizumab is included in COVID-19 treatment guidelines, and several randomized clinical trials are underway in China including patients with COVID-19 with severe pneumonia.
- Immunoglobulin therapy. Antibodies from recovered COVID-19 patients could help with free virus and infected cell immune clearance, the authors said, adding that further studies are warranted beyond a few small published case series that suggest promise. Furthermore, on March 24 the FDA released guidance for screening donors for COVID-19 convalescent plasma and on emergency investigational new drug applications based on this modality.
- Lopinavir/ritonavir. Despite demonstrated in vitro activity against other novel coronaviruses, there is no published in vitro data for lopinavir/ritonavir in SARS-CoV-2, and likely a “limited role” for this combination anticipated in treating COVID-19, according to the review authors. In an open-label randomized clinical trial published in the New England Journal of Medicine (2020 Mar 18. doi: 10.1056/NEJMoa2001282), there were no differences in clinical improvement, viral clearance, or mortality for antiviral treatment versus standard care. Delayed treatment initiation may explain the ineffectiveness, though a subgroup analysis didn’t show a shorter time to clinical improvement for those who got the treatment earlier.
- Ribavirin. Likewise, this antiviral medication has efficacy and safety data suggesting “limited value” for treatment of COVID-19. Treatment of SARS yielded “inconclusive results” for ribavirin, which was also associated with substantial toxicity that included hemolytic anemia in 60% of SARS patients.
- Oseltamivir. While it may treat influenza, it has no documented activity against SARS-CoV-2 in vitro: “This agent has no role in the management of COVID-19 once influenza has been excluded,” said Dr. Sanders and coauthors.
- Corticosteroids. They could decrease inflammatory responses in the lung, but they could also lead to delays in viral clearance and increases in secondary infection risk. Guidelines for COVID-19 say to avoid corticosteroids, and the authors of the review concur, saying that potential harms and lack of proven benefit mean they usually should not be used outside of a randomized clinical trial setting.
- Vaccines. Clearly, vaccines represent the “most effective long-term strategy” to prevent future COVID-19 outbreaks, though at least 12-18 months would be required until vaccines can be widely deployed, authors said.
Dr. Sanders reported no potential conflicts. Senior author James B. Cutrell, MD, also of the University of Texas Southwestern Medical Center, reported nonfinancial support from Gilead and Regeneron outside of the study. No other authors reported disclosures.
FROM JAMA
Troponins touted as ‘ally’ in COVID-19 triage, but message is nuanced
, cardiologists in the United Kingdom advise in a recently published viewpoint.
The tests can be used to “inform the triage of patients to critical care, guide the use of supportive treatments, and facilitate targeted cardiac investigations in those most likely to benefit,” Nicholas Mills, MD, PhD, University of Edinburgh, United Kingdom, told theheart.org | Medscape Cardiology. He is senior author on the viewpoint published online April 6 in the journal Circulation.
Older adults and those with a history of underlying cardiovascular disease appear to be at greatest risk of dying from COVID-19. “From early reports it is clear that elevated cardiac troponin concentrations predict in-hospital mortality,” said Mills.
In a recent report on hospitalized patients with COVID-19 in Wuhan, China, for example, cardiac injury (hs-cTn above the 99th-percentile upper reference limit) was seen in 1 in 5 patients and was an independent predictor of dying in the hospital. Mortality was 10-fold higher in those with cardiac injury on presentation.
Elevated cardiac troponin in the setting of COVID-19, Mills said, “may reflect illness severity with myocardial injury arising due to myocardial oxygen supply–demand imbalance. Or it may be due to direct cardiac involvement through viral myocarditis or stress cardiomyopathy, or where the prothrombotic and proinflammatory state is precipitating acute coronary syndromes.”
In their viewpoint, the authors note that circulating cTn is a marker of myocardial injury, “including but not limited to myocardial infarction or myocarditis, and the clinical relevance of this distinction has never been so clear.”
Therefore, the consequence of not measuring cardiac troponin may be to “ignore the plethora of ischemic and nonischemic causes” of myocardial injury related to COVID-19. “Clinicians who have used troponin measurement as a binary test for myocardial infarction independent of clinical context and those who consider an elevated cardiac troponin concentration to be a mandate for invasive coronary angiography must recalibrate,” they write.
“Rather than encouraging avoidance of troponin testing, we must harness the unheralded engagement from the cardiovascular community due to COVID-19 to better understand the utility of this essential biomarker and to educate clinicians on its interpretation and implications for prognosis and clinical decision making.”
Based on “same logic” as recent ACC guidance
The viewpoint was to some extent a response to a recent informal guidance from the American College of Cardiology (ACC) that advised caution in use of troponin and natriuretic peptide tests in patients with COVID-19.
Even so, that ACC guidance and the new viewpoint in Circulation are based on the “same logic,” James Januzzi Jr, MD, Massachusetts General Hospital, Boston, told theheart.org | Medscape Cardiology. Both documents:
- Point out that troponins are frequently abnormal in patients with severe cases of COVID-19
- Caution that clinicians should not equate an abnormal hs-cTn with acute myocardial infarction
- Note that, in most cases, hs-cTn elevations are a result of noncoronary mechanisms
- Recognize the potential risk to caregivers and the continued unchecked spread of SARS-CoV-2 related to downstream testing that might not be needed
“The Circulation opinion piece states that clinicians often use troponin as a binary test for myocardial infarction and a mandate for downstream testing, suggesting clinicians will need to recalibrate that approach, something I agree with and which is the central message of the ACC position,” Januzzi said.
Probably the biggest difference between the two documents, he said, is in the Circulation authors’ apparent enthusiasm to use hs-cTn as a tool to judge disease severity in patients with COVID-19.
It’s been known for more than a decade that myocardial injury is “an important risk predictor” in critical illness, Januzzi explained. “So the link between cardiac injury and outcomes in critical illness is nothing new. The difference is the fact we are seeing so many patients with COVID-19 all at once, and the authors suggest that using troponin might help in triage decision making.”
“There may be [such] a role here, but the data have not been systematically collected, and whether troponin truly adds something beyond information already available at the bedside — for example, does it add anything not already obvious at the bedside? — has not yet been conclusively proven,” Januzzi cautioned.
“As well, there are no prospective data supporting troponin as a trigger for ICU triage or for deciding on specific treatments.”
Positive cTn status “common” in COVID-19 patients
In his experience, Barry Cohen, MD, Morristown Medical Center, New Jersey, told theheart.org | Medscape Cardiology, that positive cTn status is “common in COVID-19 patients and appears to have prognostic value, not only in type 1 MI due to atherothrombotic disease (related to a proinflammatory and prothrombotic state), but more frequently type 2 MI (supply–demand mismatch), viral myocarditis, coronary microvascular ischemia, stress cardiomyopathy or tachyarrhythmias.”
Moreover, Cohen said, hs-cTn “has identified patients at increased risk for ventilation support (invasive and noninvasive), acute respiratory distress syndrome, acute kidney injury, and mortality.”
Echoing both the ACC document and the Circulation report, Cohen also said hs-cTn measurements “appear to help risk stratify COVID-19 patients, but clearly do not mean that a troponin-positive patient needs to go to the cath lab and be treated as having acute coronary syndrome. Only a minority of these patients require this intervention.”
Mills discloses receiving honoraria from Abbott Diagnostics, Roche Diagnostics, Siemens Healthineers, and LumiraDx. Januzzi has previously disclosed receiving personal fees from the American College of Cardiology, Pfizer, Merck, AbbVie, Amgen, Boehringer Ingelheim, and Takeda; grants and personal fees from Novartis, Roche, Abbott, and Janssen; and grants from Singulex and Prevencio. Cohen has disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
, cardiologists in the United Kingdom advise in a recently published viewpoint.
The tests can be used to “inform the triage of patients to critical care, guide the use of supportive treatments, and facilitate targeted cardiac investigations in those most likely to benefit,” Nicholas Mills, MD, PhD, University of Edinburgh, United Kingdom, told theheart.org | Medscape Cardiology. He is senior author on the viewpoint published online April 6 in the journal Circulation.
Older adults and those with a history of underlying cardiovascular disease appear to be at greatest risk of dying from COVID-19. “From early reports it is clear that elevated cardiac troponin concentrations predict in-hospital mortality,” said Mills.
In a recent report on hospitalized patients with COVID-19 in Wuhan, China, for example, cardiac injury (hs-cTn above the 99th-percentile upper reference limit) was seen in 1 in 5 patients and was an independent predictor of dying in the hospital. Mortality was 10-fold higher in those with cardiac injury on presentation.
Elevated cardiac troponin in the setting of COVID-19, Mills said, “may reflect illness severity with myocardial injury arising due to myocardial oxygen supply–demand imbalance. Or it may be due to direct cardiac involvement through viral myocarditis or stress cardiomyopathy, or where the prothrombotic and proinflammatory state is precipitating acute coronary syndromes.”
In their viewpoint, the authors note that circulating cTn is a marker of myocardial injury, “including but not limited to myocardial infarction or myocarditis, and the clinical relevance of this distinction has never been so clear.”
Therefore, the consequence of not measuring cardiac troponin may be to “ignore the plethora of ischemic and nonischemic causes” of myocardial injury related to COVID-19. “Clinicians who have used troponin measurement as a binary test for myocardial infarction independent of clinical context and those who consider an elevated cardiac troponin concentration to be a mandate for invasive coronary angiography must recalibrate,” they write.
“Rather than encouraging avoidance of troponin testing, we must harness the unheralded engagement from the cardiovascular community due to COVID-19 to better understand the utility of this essential biomarker and to educate clinicians on its interpretation and implications for prognosis and clinical decision making.”
Based on “same logic” as recent ACC guidance
The viewpoint was to some extent a response to a recent informal guidance from the American College of Cardiology (ACC) that advised caution in use of troponin and natriuretic peptide tests in patients with COVID-19.
Even so, that ACC guidance and the new viewpoint in Circulation are based on the “same logic,” James Januzzi Jr, MD, Massachusetts General Hospital, Boston, told theheart.org | Medscape Cardiology. Both documents:
- Point out that troponins are frequently abnormal in patients with severe cases of COVID-19
- Caution that clinicians should not equate an abnormal hs-cTn with acute myocardial infarction
- Note that, in most cases, hs-cTn elevations are a result of noncoronary mechanisms
- Recognize the potential risk to caregivers and the continued unchecked spread of SARS-CoV-2 related to downstream testing that might not be needed
“The Circulation opinion piece states that clinicians often use troponin as a binary test for myocardial infarction and a mandate for downstream testing, suggesting clinicians will need to recalibrate that approach, something I agree with and which is the central message of the ACC position,” Januzzi said.
Probably the biggest difference between the two documents, he said, is in the Circulation authors’ apparent enthusiasm to use hs-cTn as a tool to judge disease severity in patients with COVID-19.
It’s been known for more than a decade that myocardial injury is “an important risk predictor” in critical illness, Januzzi explained. “So the link between cardiac injury and outcomes in critical illness is nothing new. The difference is the fact we are seeing so many patients with COVID-19 all at once, and the authors suggest that using troponin might help in triage decision making.”
“There may be [such] a role here, but the data have not been systematically collected, and whether troponin truly adds something beyond information already available at the bedside — for example, does it add anything not already obvious at the bedside? — has not yet been conclusively proven,” Januzzi cautioned.
“As well, there are no prospective data supporting troponin as a trigger for ICU triage or for deciding on specific treatments.”
Positive cTn status “common” in COVID-19 patients
In his experience, Barry Cohen, MD, Morristown Medical Center, New Jersey, told theheart.org | Medscape Cardiology, that positive cTn status is “common in COVID-19 patients and appears to have prognostic value, not only in type 1 MI due to atherothrombotic disease (related to a proinflammatory and prothrombotic state), but more frequently type 2 MI (supply–demand mismatch), viral myocarditis, coronary microvascular ischemia, stress cardiomyopathy or tachyarrhythmias.”
Moreover, Cohen said, hs-cTn “has identified patients at increased risk for ventilation support (invasive and noninvasive), acute respiratory distress syndrome, acute kidney injury, and mortality.”
Echoing both the ACC document and the Circulation report, Cohen also said hs-cTn measurements “appear to help risk stratify COVID-19 patients, but clearly do not mean that a troponin-positive patient needs to go to the cath lab and be treated as having acute coronary syndrome. Only a minority of these patients require this intervention.”
Mills discloses receiving honoraria from Abbott Diagnostics, Roche Diagnostics, Siemens Healthineers, and LumiraDx. Januzzi has previously disclosed receiving personal fees from the American College of Cardiology, Pfizer, Merck, AbbVie, Amgen, Boehringer Ingelheim, and Takeda; grants and personal fees from Novartis, Roche, Abbott, and Janssen; and grants from Singulex and Prevencio. Cohen has disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
, cardiologists in the United Kingdom advise in a recently published viewpoint.
The tests can be used to “inform the triage of patients to critical care, guide the use of supportive treatments, and facilitate targeted cardiac investigations in those most likely to benefit,” Nicholas Mills, MD, PhD, University of Edinburgh, United Kingdom, told theheart.org | Medscape Cardiology. He is senior author on the viewpoint published online April 6 in the journal Circulation.
Older adults and those with a history of underlying cardiovascular disease appear to be at greatest risk of dying from COVID-19. “From early reports it is clear that elevated cardiac troponin concentrations predict in-hospital mortality,” said Mills.
In a recent report on hospitalized patients with COVID-19 in Wuhan, China, for example, cardiac injury (hs-cTn above the 99th-percentile upper reference limit) was seen in 1 in 5 patients and was an independent predictor of dying in the hospital. Mortality was 10-fold higher in those with cardiac injury on presentation.
Elevated cardiac troponin in the setting of COVID-19, Mills said, “may reflect illness severity with myocardial injury arising due to myocardial oxygen supply–demand imbalance. Or it may be due to direct cardiac involvement through viral myocarditis or stress cardiomyopathy, or where the prothrombotic and proinflammatory state is precipitating acute coronary syndromes.”
In their viewpoint, the authors note that circulating cTn is a marker of myocardial injury, “including but not limited to myocardial infarction or myocarditis, and the clinical relevance of this distinction has never been so clear.”
Therefore, the consequence of not measuring cardiac troponin may be to “ignore the plethora of ischemic and nonischemic causes” of myocardial injury related to COVID-19. “Clinicians who have used troponin measurement as a binary test for myocardial infarction independent of clinical context and those who consider an elevated cardiac troponin concentration to be a mandate for invasive coronary angiography must recalibrate,” they write.
“Rather than encouraging avoidance of troponin testing, we must harness the unheralded engagement from the cardiovascular community due to COVID-19 to better understand the utility of this essential biomarker and to educate clinicians on its interpretation and implications for prognosis and clinical decision making.”
Based on “same logic” as recent ACC guidance
The viewpoint was to some extent a response to a recent informal guidance from the American College of Cardiology (ACC) that advised caution in use of troponin and natriuretic peptide tests in patients with COVID-19.
Even so, that ACC guidance and the new viewpoint in Circulation are based on the “same logic,” James Januzzi Jr, MD, Massachusetts General Hospital, Boston, told theheart.org | Medscape Cardiology. Both documents:
- Point out that troponins are frequently abnormal in patients with severe cases of COVID-19
- Caution that clinicians should not equate an abnormal hs-cTn with acute myocardial infarction
- Note that, in most cases, hs-cTn elevations are a result of noncoronary mechanisms
- Recognize the potential risk to caregivers and the continued unchecked spread of SARS-CoV-2 related to downstream testing that might not be needed
“The Circulation opinion piece states that clinicians often use troponin as a binary test for myocardial infarction and a mandate for downstream testing, suggesting clinicians will need to recalibrate that approach, something I agree with and which is the central message of the ACC position,” Januzzi said.
Probably the biggest difference between the two documents, he said, is in the Circulation authors’ apparent enthusiasm to use hs-cTn as a tool to judge disease severity in patients with COVID-19.
It’s been known for more than a decade that myocardial injury is “an important risk predictor” in critical illness, Januzzi explained. “So the link between cardiac injury and outcomes in critical illness is nothing new. The difference is the fact we are seeing so many patients with COVID-19 all at once, and the authors suggest that using troponin might help in triage decision making.”
“There may be [such] a role here, but the data have not been systematically collected, and whether troponin truly adds something beyond information already available at the bedside — for example, does it add anything not already obvious at the bedside? — has not yet been conclusively proven,” Januzzi cautioned.
“As well, there are no prospective data supporting troponin as a trigger for ICU triage or for deciding on specific treatments.”
Positive cTn status “common” in COVID-19 patients
In his experience, Barry Cohen, MD, Morristown Medical Center, New Jersey, told theheart.org | Medscape Cardiology, that positive cTn status is “common in COVID-19 patients and appears to have prognostic value, not only in type 1 MI due to atherothrombotic disease (related to a proinflammatory and prothrombotic state), but more frequently type 2 MI (supply–demand mismatch), viral myocarditis, coronary microvascular ischemia, stress cardiomyopathy or tachyarrhythmias.”
Moreover, Cohen said, hs-cTn “has identified patients at increased risk for ventilation support (invasive and noninvasive), acute respiratory distress syndrome, acute kidney injury, and mortality.”
Echoing both the ACC document and the Circulation report, Cohen also said hs-cTn measurements “appear to help risk stratify COVID-19 patients, but clearly do not mean that a troponin-positive patient needs to go to the cath lab and be treated as having acute coronary syndrome. Only a minority of these patients require this intervention.”
Mills discloses receiving honoraria from Abbott Diagnostics, Roche Diagnostics, Siemens Healthineers, and LumiraDx. Januzzi has previously disclosed receiving personal fees from the American College of Cardiology, Pfizer, Merck, AbbVie, Amgen, Boehringer Ingelheim, and Takeda; grants and personal fees from Novartis, Roche, Abbott, and Janssen; and grants from Singulex and Prevencio. Cohen has disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
COVID-19 pandemic brings unexpected pediatric consequences
As physicians and advanced practitioners, we have been preparing to face COVID-19 – anticipating increasing volumes of patients with fevers, cough, and shortness of breath, and potential surges in emergency departments (EDs) and primary care offices. Fortunately, while COVID-19 has demonstrated more mild symptoms in pediatric patients, the heightened public health fears and mandated social isolation have created some unforeseen consequences for pediatric patients. This article presents cases encountered over the course of 2 weeks in our ED that shed light on the unexpected ramifications of living in the time of a pandemic. These encounters should remind us as providers to be diligent and thorough in giving guidance to families during a time when face-to-face medicine has become increasingly difficult and limited.
These stories have been modified to protect patient confidentiality.
Case 1
A 2-week-old full-term infant arrived in the ED after having a fever for 48 hours. The patient’s mother reported that she had called the pediatrician yesterday to ask for advice on treating the fever and was instructed to give acetaminophen and bring the infant into the ED for testing.
When we asked mom why she did not bring the infant in yesterday, she stated that the fever went down with acetaminophen, and the baby was drinking well and urinating normally. Mostly, she was afraid to bring the child into the ED given concern for COVID-19; however, when the fever persisted today, she came in. During the work-up, the infant was noted to have focal seizures and was ultimately diagnosed with bacterial meningitis.
Takeaway: Families may be hesitant to follow pediatrician’s advice to seek medical attention at an ED or doctor’s office because of the fear of being exposed to COVID-19.
- If something is urgent or emergent, be sure to stress the importance to families that the advice is non-negotiable for their child’s health.
- Attempt to call ahead for patients who might be more vulnerable in waiting rooms or overcrowded hospitals.
Case 2
A 5-month-old baby presented to the ED with new-onset seizures. Immediate bedside blood work performed demonstrated a normal blood glucose, but the baby was profoundly hyponatremic. Upon asking the mother if the baby has had any vomiting, diarrhea, or difficulty tolerating feeds, she says that she has been diluting formula because all the stores were out of formula. Today, she gave the baby plain water because they were completely out of formula.
Takeaway: With economists estimating unemployment rates in the United States at 13% at press time (the worst since the Great Depression), many families may lack resources to purchase necessities.
- Even if families have the ability to purchase necessities, they may be difficult to find or unavailable (e.g., formula, medications, diapers).
- Consider reaching out to patients in your practice to ask about their ability to find essentials and with advice on what to do if they run out of formula or diapers, or who they should contact if they cannot refill a medication.
- Are you in a position to speak with your mayor or local council to ensure there are regulations on the hoarding of essential items?
- In a time when breast milk or formula is not available for children younger than 1 year of age, what will you recommend for families? There are no current American Academy of Pediatrics’ guidelines.
Case 3
A school-aged girl was helping her mother sanitize the home during the COVID-19 pandemic. She had her gloves on, her commercial antiseptic cleaner ready to go, but it was not spraying. She turned the bottle around to check the nozzle and sprayed herself in the eyes. The family presented to the ED for alkaline burn to her eyes, which required copious irrigation.
Takeaway: Children are spending more time in the house with access to button batteries, choking hazards, and cleaning supplies.
- Cleaning products can cause chemical burns. These products should not be used by young children.
Case 4
A school-aged boy arrived via emergency medical services (EMS) for altered mental status. He told his father he was feeling dizzy and then lost consciousness. EMS noticed that he had some tonic movements of his lower extremities, and when he arrived in the ED, he had eye deviation and was unresponsive.
Work-up ultimately demonstrated that this patient had a seizure and a dangerously elevated ethanol level from drinking an entire bottle of hand sanitizer. Hand sanitizer may contain high concentrations of ethyl alcohol or isopropyl alcohol, which when ingested can cause intoxication or poisoning.
Takeaway: Many products that we may view as harmless can be toxic if ingested in large amounts.
- Consider making a list of products that families may have acquired and have around the home during this COVID-19 pandemic and instruct families to make sure dangerous items (e.g., acetaminophen, aspirin, hand sanitizer, lighters, firearms, batteries) are locked up and/or out of reach of children.
- Make sure families know the Poison Control phone number (800-222-1222).
Case 5
An adolescent female currently being treated with immunosuppressants arrived from home with fever. Her medical history revealed that the patient’s guardian recently passed away from suspected COVID-19. The patient was tested and is herself found to be positive for COVID-19. The patient is currently being cared for by relatives who also live in the same home. They require extensive education and teaching regarding the patient’s medication regimen, while also dealing with the loss of their loved one and the fear of personal exposure.
Takeaway: Communicate with families – especially those with special health care needs – about issues of guardianship in case a child’s primary caretaker falls ill.
- Discuss with families about having easily accessible lists of medications and medical conditions.
- Involve social work and child life specialists to help children and their families deal with life-altering changes and losses suffered during this time, as well as fears related to mortality and exposure.
Case 6
A 3-year-old boy arrived covered in bruises and complaining of stomachache. While the mother denies any known abuse, she states that her significant other has been getting more and more “worked up having to deal with the child’s behavior all day every day.” The preschool the child previously attended has closed due to the pandemic.
Takeaway: Abuse is more common when the parents perceive that there is little community support and when families feel a lack of connection to the community.1 Huang et al. examined the relationship between the economy and nonaccidental trauma, showing a doubling in the rate of nonaccidental head trauma during economic recession.2
- Allow families to know that they are not alone and that child care is difficult
- Offer advice on what caretakers can do if they feel alone or at their mental or physical limit.
- Provide strategies on your practice’s website if a situation at home becomes tense and strained.
Case 7
An adolescent female arrived to the ED with increased suicidality. She normally follows with her psychiatrist once a month and her therapist once a week. Since the beginning of COVID-19 restrictions, she has been using telemedicine for her therapy visits. While previously doing well, she reports that her suicidal ideations have worsened because of feeling isolated from her friends now that school is out and she is not allowed to see them. Although compliant with her medications, her thoughts have increased to the point where she has to be admitted to inpatient psychiatry.
Takeaway: Anxiety, depression, and suicide may increase in a down economy. After the 2008 global economic crisis, rates of suicide drastically increased.3
- Recognize the limitations of telemedicine (technology limitations, patient cooperation, etc.)
- Social isolation may contribute to worsening mental health
- Know when to advise patients to seek in-person evaluation and care for medical and mental health concerns.
Pediatricians are at the forefront of preventative medicine. Families rely on pediatricians for trustworthy and accurate anticipatory guidance, a need that is only heightened during times of local and national stress. The social isolation, fear, and lack of resources accompanying this pandemic have serious consequences for our families. What can you and your practice do to keep children safe in the time of COVID-19?
Dr. Angelica DesPain is a pediatric emergency medicine fellow at Children’s National Hospital in Washington. Dr. Rachel Hatcliffe is an attending physician at the hospital. Neither physician had any relevant financial disclosures. Email Dr. DesPain and/or Dr. Hatcliffe at pdnews@mdedge.com.
References
1. Child Dev. 1978;49:604-16.
2. J Neurosurg Pediatr 2011 Aug;8(2):171-6.
3. BMJ 2013;347:f5239.
As physicians and advanced practitioners, we have been preparing to face COVID-19 – anticipating increasing volumes of patients with fevers, cough, and shortness of breath, and potential surges in emergency departments (EDs) and primary care offices. Fortunately, while COVID-19 has demonstrated more mild symptoms in pediatric patients, the heightened public health fears and mandated social isolation have created some unforeseen consequences for pediatric patients. This article presents cases encountered over the course of 2 weeks in our ED that shed light on the unexpected ramifications of living in the time of a pandemic. These encounters should remind us as providers to be diligent and thorough in giving guidance to families during a time when face-to-face medicine has become increasingly difficult and limited.
These stories have been modified to protect patient confidentiality.
Case 1
A 2-week-old full-term infant arrived in the ED after having a fever for 48 hours. The patient’s mother reported that she had called the pediatrician yesterday to ask for advice on treating the fever and was instructed to give acetaminophen and bring the infant into the ED for testing.
When we asked mom why she did not bring the infant in yesterday, she stated that the fever went down with acetaminophen, and the baby was drinking well and urinating normally. Mostly, she was afraid to bring the child into the ED given concern for COVID-19; however, when the fever persisted today, she came in. During the work-up, the infant was noted to have focal seizures and was ultimately diagnosed with bacterial meningitis.
Takeaway: Families may be hesitant to follow pediatrician’s advice to seek medical attention at an ED or doctor’s office because of the fear of being exposed to COVID-19.
- If something is urgent or emergent, be sure to stress the importance to families that the advice is non-negotiable for their child’s health.
- Attempt to call ahead for patients who might be more vulnerable in waiting rooms or overcrowded hospitals.
Case 2
A 5-month-old baby presented to the ED with new-onset seizures. Immediate bedside blood work performed demonstrated a normal blood glucose, but the baby was profoundly hyponatremic. Upon asking the mother if the baby has had any vomiting, diarrhea, or difficulty tolerating feeds, she says that she has been diluting formula because all the stores were out of formula. Today, she gave the baby plain water because they were completely out of formula.
Takeaway: With economists estimating unemployment rates in the United States at 13% at press time (the worst since the Great Depression), many families may lack resources to purchase necessities.
- Even if families have the ability to purchase necessities, they may be difficult to find or unavailable (e.g., formula, medications, diapers).
- Consider reaching out to patients in your practice to ask about their ability to find essentials and with advice on what to do if they run out of formula or diapers, or who they should contact if they cannot refill a medication.
- Are you in a position to speak with your mayor or local council to ensure there are regulations on the hoarding of essential items?
- In a time when breast milk or formula is not available for children younger than 1 year of age, what will you recommend for families? There are no current American Academy of Pediatrics’ guidelines.
Case 3
A school-aged girl was helping her mother sanitize the home during the COVID-19 pandemic. She had her gloves on, her commercial antiseptic cleaner ready to go, but it was not spraying. She turned the bottle around to check the nozzle and sprayed herself in the eyes. The family presented to the ED for alkaline burn to her eyes, which required copious irrigation.
Takeaway: Children are spending more time in the house with access to button batteries, choking hazards, and cleaning supplies.
- Cleaning products can cause chemical burns. These products should not be used by young children.
Case 4
A school-aged boy arrived via emergency medical services (EMS) for altered mental status. He told his father he was feeling dizzy and then lost consciousness. EMS noticed that he had some tonic movements of his lower extremities, and when he arrived in the ED, he had eye deviation and was unresponsive.
Work-up ultimately demonstrated that this patient had a seizure and a dangerously elevated ethanol level from drinking an entire bottle of hand sanitizer. Hand sanitizer may contain high concentrations of ethyl alcohol or isopropyl alcohol, which when ingested can cause intoxication or poisoning.
Takeaway: Many products that we may view as harmless can be toxic if ingested in large amounts.
- Consider making a list of products that families may have acquired and have around the home during this COVID-19 pandemic and instruct families to make sure dangerous items (e.g., acetaminophen, aspirin, hand sanitizer, lighters, firearms, batteries) are locked up and/or out of reach of children.
- Make sure families know the Poison Control phone number (800-222-1222).
Case 5
An adolescent female currently being treated with immunosuppressants arrived from home with fever. Her medical history revealed that the patient’s guardian recently passed away from suspected COVID-19. The patient was tested and is herself found to be positive for COVID-19. The patient is currently being cared for by relatives who also live in the same home. They require extensive education and teaching regarding the patient’s medication regimen, while also dealing with the loss of their loved one and the fear of personal exposure.
Takeaway: Communicate with families – especially those with special health care needs – about issues of guardianship in case a child’s primary caretaker falls ill.
- Discuss with families about having easily accessible lists of medications and medical conditions.
- Involve social work and child life specialists to help children and their families deal with life-altering changes and losses suffered during this time, as well as fears related to mortality and exposure.
Case 6
A 3-year-old boy arrived covered in bruises and complaining of stomachache. While the mother denies any known abuse, she states that her significant other has been getting more and more “worked up having to deal with the child’s behavior all day every day.” The preschool the child previously attended has closed due to the pandemic.
Takeaway: Abuse is more common when the parents perceive that there is little community support and when families feel a lack of connection to the community.1 Huang et al. examined the relationship between the economy and nonaccidental trauma, showing a doubling in the rate of nonaccidental head trauma during economic recession.2
- Allow families to know that they are not alone and that child care is difficult
- Offer advice on what caretakers can do if they feel alone or at their mental or physical limit.
- Provide strategies on your practice’s website if a situation at home becomes tense and strained.
Case 7
An adolescent female arrived to the ED with increased suicidality. She normally follows with her psychiatrist once a month and her therapist once a week. Since the beginning of COVID-19 restrictions, she has been using telemedicine for her therapy visits. While previously doing well, she reports that her suicidal ideations have worsened because of feeling isolated from her friends now that school is out and she is not allowed to see them. Although compliant with her medications, her thoughts have increased to the point where she has to be admitted to inpatient psychiatry.
Takeaway: Anxiety, depression, and suicide may increase in a down economy. After the 2008 global economic crisis, rates of suicide drastically increased.3
- Recognize the limitations of telemedicine (technology limitations, patient cooperation, etc.)
- Social isolation may contribute to worsening mental health
- Know when to advise patients to seek in-person evaluation and care for medical and mental health concerns.
Pediatricians are at the forefront of preventative medicine. Families rely on pediatricians for trustworthy and accurate anticipatory guidance, a need that is only heightened during times of local and national stress. The social isolation, fear, and lack of resources accompanying this pandemic have serious consequences for our families. What can you and your practice do to keep children safe in the time of COVID-19?
Dr. Angelica DesPain is a pediatric emergency medicine fellow at Children’s National Hospital in Washington. Dr. Rachel Hatcliffe is an attending physician at the hospital. Neither physician had any relevant financial disclosures. Email Dr. DesPain and/or Dr. Hatcliffe at pdnews@mdedge.com.
References
1. Child Dev. 1978;49:604-16.
2. J Neurosurg Pediatr 2011 Aug;8(2):171-6.
3. BMJ 2013;347:f5239.
As physicians and advanced practitioners, we have been preparing to face COVID-19 – anticipating increasing volumes of patients with fevers, cough, and shortness of breath, and potential surges in emergency departments (EDs) and primary care offices. Fortunately, while COVID-19 has demonstrated more mild symptoms in pediatric patients, the heightened public health fears and mandated social isolation have created some unforeseen consequences for pediatric patients. This article presents cases encountered over the course of 2 weeks in our ED that shed light on the unexpected ramifications of living in the time of a pandemic. These encounters should remind us as providers to be diligent and thorough in giving guidance to families during a time when face-to-face medicine has become increasingly difficult and limited.
These stories have been modified to protect patient confidentiality.
Case 1
A 2-week-old full-term infant arrived in the ED after having a fever for 48 hours. The patient’s mother reported that she had called the pediatrician yesterday to ask for advice on treating the fever and was instructed to give acetaminophen and bring the infant into the ED for testing.
When we asked mom why she did not bring the infant in yesterday, she stated that the fever went down with acetaminophen, and the baby was drinking well and urinating normally. Mostly, she was afraid to bring the child into the ED given concern for COVID-19; however, when the fever persisted today, she came in. During the work-up, the infant was noted to have focal seizures and was ultimately diagnosed with bacterial meningitis.
Takeaway: Families may be hesitant to follow pediatrician’s advice to seek medical attention at an ED or doctor’s office because of the fear of being exposed to COVID-19.
- If something is urgent or emergent, be sure to stress the importance to families that the advice is non-negotiable for their child’s health.
- Attempt to call ahead for patients who might be more vulnerable in waiting rooms or overcrowded hospitals.
Case 2
A 5-month-old baby presented to the ED with new-onset seizures. Immediate bedside blood work performed demonstrated a normal blood glucose, but the baby was profoundly hyponatremic. Upon asking the mother if the baby has had any vomiting, diarrhea, or difficulty tolerating feeds, she says that she has been diluting formula because all the stores were out of formula. Today, she gave the baby plain water because they were completely out of formula.
Takeaway: With economists estimating unemployment rates in the United States at 13% at press time (the worst since the Great Depression), many families may lack resources to purchase necessities.
- Even if families have the ability to purchase necessities, they may be difficult to find or unavailable (e.g., formula, medications, diapers).
- Consider reaching out to patients in your practice to ask about their ability to find essentials and with advice on what to do if they run out of formula or diapers, or who they should contact if they cannot refill a medication.
- Are you in a position to speak with your mayor or local council to ensure there are regulations on the hoarding of essential items?
- In a time when breast milk or formula is not available for children younger than 1 year of age, what will you recommend for families? There are no current American Academy of Pediatrics’ guidelines.
Case 3
A school-aged girl was helping her mother sanitize the home during the COVID-19 pandemic. She had her gloves on, her commercial antiseptic cleaner ready to go, but it was not spraying. She turned the bottle around to check the nozzle and sprayed herself in the eyes. The family presented to the ED for alkaline burn to her eyes, which required copious irrigation.
Takeaway: Children are spending more time in the house with access to button batteries, choking hazards, and cleaning supplies.
- Cleaning products can cause chemical burns. These products should not be used by young children.
Case 4
A school-aged boy arrived via emergency medical services (EMS) for altered mental status. He told his father he was feeling dizzy and then lost consciousness. EMS noticed that he had some tonic movements of his lower extremities, and when he arrived in the ED, he had eye deviation and was unresponsive.
Work-up ultimately demonstrated that this patient had a seizure and a dangerously elevated ethanol level from drinking an entire bottle of hand sanitizer. Hand sanitizer may contain high concentrations of ethyl alcohol or isopropyl alcohol, which when ingested can cause intoxication or poisoning.
Takeaway: Many products that we may view as harmless can be toxic if ingested in large amounts.
- Consider making a list of products that families may have acquired and have around the home during this COVID-19 pandemic and instruct families to make sure dangerous items (e.g., acetaminophen, aspirin, hand sanitizer, lighters, firearms, batteries) are locked up and/or out of reach of children.
- Make sure families know the Poison Control phone number (800-222-1222).
Case 5
An adolescent female currently being treated with immunosuppressants arrived from home with fever. Her medical history revealed that the patient’s guardian recently passed away from suspected COVID-19. The patient was tested and is herself found to be positive for COVID-19. The patient is currently being cared for by relatives who also live in the same home. They require extensive education and teaching regarding the patient’s medication regimen, while also dealing with the loss of their loved one and the fear of personal exposure.
Takeaway: Communicate with families – especially those with special health care needs – about issues of guardianship in case a child’s primary caretaker falls ill.
- Discuss with families about having easily accessible lists of medications and medical conditions.
- Involve social work and child life specialists to help children and their families deal with life-altering changes and losses suffered during this time, as well as fears related to mortality and exposure.
Case 6
A 3-year-old boy arrived covered in bruises and complaining of stomachache. While the mother denies any known abuse, she states that her significant other has been getting more and more “worked up having to deal with the child’s behavior all day every day.” The preschool the child previously attended has closed due to the pandemic.
Takeaway: Abuse is more common when the parents perceive that there is little community support and when families feel a lack of connection to the community.1 Huang et al. examined the relationship between the economy and nonaccidental trauma, showing a doubling in the rate of nonaccidental head trauma during economic recession.2
- Allow families to know that they are not alone and that child care is difficult
- Offer advice on what caretakers can do if they feel alone or at their mental or physical limit.
- Provide strategies on your practice’s website if a situation at home becomes tense and strained.
Case 7
An adolescent female arrived to the ED with increased suicidality. She normally follows with her psychiatrist once a month and her therapist once a week. Since the beginning of COVID-19 restrictions, she has been using telemedicine for her therapy visits. While previously doing well, she reports that her suicidal ideations have worsened because of feeling isolated from her friends now that school is out and she is not allowed to see them. Although compliant with her medications, her thoughts have increased to the point where she has to be admitted to inpatient psychiatry.
Takeaway: Anxiety, depression, and suicide may increase in a down economy. After the 2008 global economic crisis, rates of suicide drastically increased.3
- Recognize the limitations of telemedicine (technology limitations, patient cooperation, etc.)
- Social isolation may contribute to worsening mental health
- Know when to advise patients to seek in-person evaluation and care for medical and mental health concerns.
Pediatricians are at the forefront of preventative medicine. Families rely on pediatricians for trustworthy and accurate anticipatory guidance, a need that is only heightened during times of local and national stress. The social isolation, fear, and lack of resources accompanying this pandemic have serious consequences for our families. What can you and your practice do to keep children safe in the time of COVID-19?
Dr. Angelica DesPain is a pediatric emergency medicine fellow at Children’s National Hospital in Washington. Dr. Rachel Hatcliffe is an attending physician at the hospital. Neither physician had any relevant financial disclosures. Email Dr. DesPain and/or Dr. Hatcliffe at pdnews@mdedge.com.
References
1. Child Dev. 1978;49:604-16.
2. J Neurosurg Pediatr 2011 Aug;8(2):171-6.
3. BMJ 2013;347:f5239.
ASH tackles COVID-19 with hematology-related FAQ, promotes new registries
The American Society of Hematology has committed a portion of its website to providing continually updated information addressing specific hematologic disorders in relation to COVID-19.
“As the world grapples with the novel coronavirus, ASH believes that we can help each other be as knowledgeable and prepared as possible,” wrote the society’s president, Stephanie J. Lee, MD, MPH.
On its website, ASH provides relevant COVID-19 information in a series of FAQ divided into malignant and nonmalignant hematologic diseases and disorders. In the malignant category, the various lymphomas and leukemias are individually addressed, as well as other conditions such as myelodysplastic syndromes, myeloproliferative neoplasms, and multiple myeloma. In the nonmalignant category, ASH has provided FAQ on aplastic anemia, thalassemia, sickle cell disease, pulmonary embolism, venous thromboembolism/anticoagulation, coagulopathy, and immune as well as thrombotic thrombocytopenic purpura.
In addition to the continually updated series of relevant FAQ, as part of its response to the pandemic ASH is promoting two unique COVID-19 registries for physicians: the ASH Research Collaborative’s (ASH RC) Data Hub COVID-19 Registry and the Surveillance Epidemiology of Coronavirus (COVID-19) Under Research Exclusion Sickle Cell Disease (SECURE-SCD) Registry.
“The ASH Research Collaborative’s (ASH RC) Data Hub launched the COVID-19 Registry and is currently capturing data on people who test positive for COVID-19 and have been or are currently being treated for hematologic malignancy,” according to the website. The intention is to provide “near real-time observational data summaries,” which will hopefully provide useful information to clinicians treating hematologic malignancies in patients in the midst of the COVID-19 pandemic.
The registry allows clinicians to enter their own cases in a specified format to allow data analysis on clinical practice and patient outcomes that will be aggregated to provide rapid insights for clinicians to help them care for their patients, according to ASH.
The second registry specifically deals with COVID-19 cases in patients with sickle cell disease. It also allows clinicians to add cases with a similar intention of aggregating data to provide near real-time insights into patient care. “We are asking providers caring for these patients to report all of their cases of COVID-19 to this registry,” according to the registry website. The registry is for reporting COVID-19 cases in sickle cell disease patients “after sufficient time has passed to observe the disease course through resolution of acute illness and/or death.”
ASH also provides more generalized information for hematology practitioners dealing with COVID-19 on the topics of conducting their practice and using telemedicine, among others.
Correction, April 15, 2020: This story originally said incorrectly that ASH developed the 2 new registries. The registries are merely being promoted on the ASH website.
The American Society of Hematology has committed a portion of its website to providing continually updated information addressing specific hematologic disorders in relation to COVID-19.
“As the world grapples with the novel coronavirus, ASH believes that we can help each other be as knowledgeable and prepared as possible,” wrote the society’s president, Stephanie J. Lee, MD, MPH.
On its website, ASH provides relevant COVID-19 information in a series of FAQ divided into malignant and nonmalignant hematologic diseases and disorders. In the malignant category, the various lymphomas and leukemias are individually addressed, as well as other conditions such as myelodysplastic syndromes, myeloproliferative neoplasms, and multiple myeloma. In the nonmalignant category, ASH has provided FAQ on aplastic anemia, thalassemia, sickle cell disease, pulmonary embolism, venous thromboembolism/anticoagulation, coagulopathy, and immune as well as thrombotic thrombocytopenic purpura.
In addition to the continually updated series of relevant FAQ, as part of its response to the pandemic ASH is promoting two unique COVID-19 registries for physicians: the ASH Research Collaborative’s (ASH RC) Data Hub COVID-19 Registry and the Surveillance Epidemiology of Coronavirus (COVID-19) Under Research Exclusion Sickle Cell Disease (SECURE-SCD) Registry.
“The ASH Research Collaborative’s (ASH RC) Data Hub launched the COVID-19 Registry and is currently capturing data on people who test positive for COVID-19 and have been or are currently being treated for hematologic malignancy,” according to the website. The intention is to provide “near real-time observational data summaries,” which will hopefully provide useful information to clinicians treating hematologic malignancies in patients in the midst of the COVID-19 pandemic.
The registry allows clinicians to enter their own cases in a specified format to allow data analysis on clinical practice and patient outcomes that will be aggregated to provide rapid insights for clinicians to help them care for their patients, according to ASH.
The second registry specifically deals with COVID-19 cases in patients with sickle cell disease. It also allows clinicians to add cases with a similar intention of aggregating data to provide near real-time insights into patient care. “We are asking providers caring for these patients to report all of their cases of COVID-19 to this registry,” according to the registry website. The registry is for reporting COVID-19 cases in sickle cell disease patients “after sufficient time has passed to observe the disease course through resolution of acute illness and/or death.”
ASH also provides more generalized information for hematology practitioners dealing with COVID-19 on the topics of conducting their practice and using telemedicine, among others.
Correction, April 15, 2020: This story originally said incorrectly that ASH developed the 2 new registries. The registries are merely being promoted on the ASH website.
The American Society of Hematology has committed a portion of its website to providing continually updated information addressing specific hematologic disorders in relation to COVID-19.
“As the world grapples with the novel coronavirus, ASH believes that we can help each other be as knowledgeable and prepared as possible,” wrote the society’s president, Stephanie J. Lee, MD, MPH.
On its website, ASH provides relevant COVID-19 information in a series of FAQ divided into malignant and nonmalignant hematologic diseases and disorders. In the malignant category, the various lymphomas and leukemias are individually addressed, as well as other conditions such as myelodysplastic syndromes, myeloproliferative neoplasms, and multiple myeloma. In the nonmalignant category, ASH has provided FAQ on aplastic anemia, thalassemia, sickle cell disease, pulmonary embolism, venous thromboembolism/anticoagulation, coagulopathy, and immune as well as thrombotic thrombocytopenic purpura.
In addition to the continually updated series of relevant FAQ, as part of its response to the pandemic ASH is promoting two unique COVID-19 registries for physicians: the ASH Research Collaborative’s (ASH RC) Data Hub COVID-19 Registry and the Surveillance Epidemiology of Coronavirus (COVID-19) Under Research Exclusion Sickle Cell Disease (SECURE-SCD) Registry.
“The ASH Research Collaborative’s (ASH RC) Data Hub launched the COVID-19 Registry and is currently capturing data on people who test positive for COVID-19 and have been or are currently being treated for hematologic malignancy,” according to the website. The intention is to provide “near real-time observational data summaries,” which will hopefully provide useful information to clinicians treating hematologic malignancies in patients in the midst of the COVID-19 pandemic.
The registry allows clinicians to enter their own cases in a specified format to allow data analysis on clinical practice and patient outcomes that will be aggregated to provide rapid insights for clinicians to help them care for their patients, according to ASH.
The second registry specifically deals with COVID-19 cases in patients with sickle cell disease. It also allows clinicians to add cases with a similar intention of aggregating data to provide near real-time insights into patient care. “We are asking providers caring for these patients to report all of their cases of COVID-19 to this registry,” according to the registry website. The registry is for reporting COVID-19 cases in sickle cell disease patients “after sufficient time has passed to observe the disease course through resolution of acute illness and/or death.”
ASH also provides more generalized information for hematology practitioners dealing with COVID-19 on the topics of conducting their practice and using telemedicine, among others.
Correction, April 15, 2020: This story originally said incorrectly that ASH developed the 2 new registries. The registries are merely being promoted on the ASH website.
COVID-19 causes financial woes for GI practices
On a typical clinic day, Will Bulsiewicz, MD, a Charleston, S.C.–based gastroenterologist, used to see 22 patients, while other days were filled with up to 16 procedures.
Since COVID-19 however, things have vastly changed. Dr. Bulsiewicz now visits with all clinic patients through telehealth, and the volume has dipped to between zero and six patients per day. His three-doctor practice has also experienced a more than 90% reduction in endoscopy volume.
“Naturally, this has been devastating,” Dr. Bulsiewicz said in an interview. “Our practice was started in 1984, and we had a business model that we used for the history of our practice. That practice model was upended in a matter of 2 weeks.”
Dr. Bulsiewicz is far from alone. Community GI practices across the country are experiencing similar financial distress in the face of COVID-19. In addition to a decrease in patient referrals, the Centers for Medicare & Medicaid Services has requested that all elective esophagogastroduodenoscopies, colonoscopies, endoscopies, surgeries, and procedures be delayed during the coronavirus outbreak to conserve critical equipment and limit virus exposure. The guidance aligns with recent recommendations issued by American Gastroenterological Association, American Association for the Study of Liver Diseases, American College of Gastroenterology, and American Society for Gastrointestinal Endoscopy. The lack of patients has led to plummeting revenue for many GI practices and resulted in layoffs, reduced hours, and limited salaries in order to keep practices afloat.
“We’ve had to make drastic changes in the way we work,” said Rajeev Jain, MD, AGAF, a Dallas-based gastroenterologist. “The way private practices are economically set up, they don’t have large reserves of capital or liquidity. We’re not like Apple or these big companies that have these massive cushions. It’s one thing when you have a downturn in the economy and less people come to get care, but when you have a complete shutdown, your revenue stream to pay your bills is literally dried up.”
Dr. Jain’s practice is part of Texas Digestive Disease Consultants (TDDC), which provides GI care for patients in Texas and Louisiana. TDDC is part of GI Alliance, a private equity–based consolidation of practices that includes several states and more than 350 GIs. The management services organization is a collaboration between the PE firm and the partner physicians. Since the COVID-19 outbreak, Dr. Jain said his practice has seen a dramatic drop in patients. Normally, Dr. Jain would perform between 25 and 30 outpatient scopes over the course of 2 days, he said. On a recent Monday, he performed two procedures. To preserve cash flow, Dr. Jain said he and his senior partners are not taking an income right now. Some employees were recently furloughed and laid off.
“I never in my life thought that I would have to lay off people because of an economic issue,” Dr. Jain said. “That’s psychological strain that as a physician owner you feel because these are people that you work with on a day-to-day basis and you don’t want them suffering either. That’s been a tough thing.
James S. Leavitt, MD, said his 17-physician center in Miami, Fla., has furloughed about half its staff. The center is part of Gastro Health, a private equity firm–based medical group with more than 250 providers in four states. Dr. Leavitt, president and chief clinical officer for Gastro Health, said his center has gone from about 150 patients per day to 5 or fewer, while procedures have dropped from more than 100 a day to maybe 5.
Having partnered with a private equity firm, however, Dr. Leavitt believes his practice is bettered situated to manage the health crisis and address financial challenges.
“It’s made us better prepared to weather the storm. We have a very high-powered, sophisticated administration and much broader base and access to capital. [For example,] we had a lot of depth in management so that we could roll out a robust televisit program in a week in four states with over 250 doctors.”
From a business standpoint, however, certain goals for the company are on hold, he said, such as closing on potential acquisitions.
Telemedicine works well for many patients, particularly for follow-up patients and for patients who have an established relationship with Dr. Leavitt, he said. There are limitations of course, he noted.
“If I were a dermatologist, maybe I could see the skin rash, but you can’t examine the patient,” he said. “There are certain things you can’t do. If a patient has significant abdominal pain, a televisit isn’t the greatest.”
That’s why Dr. Leavitt’s care center remains open for the handful of patients who must be seen in-person, he said. Those patients are screened beforehand and their temperatures taken before treatment.
Dr. Bulsiewicz’s practice made the transition to telehealth after never having used the modality before COVID-19.
“This was a scramble,” said Dr. Bulsiewicz, who posts about COVID-19 on social media. “We started from zero knowledge to implementation in less than a week.”
Overall, the switch went smoothly, but Dr. Bulsiewicz said reimbursement challenges come with telehealth.
“The billing is not the same,” he said. “You’re doing the same work or more, and you’re taking a reduced fee because of the antiquated fee structure that is forcing you to apply the typical rules of an office encounter.”
He hopes CMS will alter the reimbursement schedule to temporarily pay on par with traditional evaluation and management codes based on medical complexity as opposed to documentation of physical exam. CMS has already expanded Medicare telehealth coverage to cover a wider range of health care services in light of the COVID-19 crisis and also broadened the range of communication tools that can be used, according to a March announcement.
In the meantime, many practices have applied for financial assistance programs. The AGA recently pushed the government for additional assistance to help struggling practices.
Dr. Jain hopes these assistance programs roll out quickly.
“If these don’t get out there quick enough and big enough, we are going to see a massive wave of loss of independent practices and/or consolidation,” he said. “I fear a death to small, independent practices because they’re not going to have the financial wherewithal to tolerate this for too long.”
On a typical clinic day, Will Bulsiewicz, MD, a Charleston, S.C.–based gastroenterologist, used to see 22 patients, while other days were filled with up to 16 procedures.
Since COVID-19 however, things have vastly changed. Dr. Bulsiewicz now visits with all clinic patients through telehealth, and the volume has dipped to between zero and six patients per day. His three-doctor practice has also experienced a more than 90% reduction in endoscopy volume.
“Naturally, this has been devastating,” Dr. Bulsiewicz said in an interview. “Our practice was started in 1984, and we had a business model that we used for the history of our practice. That practice model was upended in a matter of 2 weeks.”
Dr. Bulsiewicz is far from alone. Community GI practices across the country are experiencing similar financial distress in the face of COVID-19. In addition to a decrease in patient referrals, the Centers for Medicare & Medicaid Services has requested that all elective esophagogastroduodenoscopies, colonoscopies, endoscopies, surgeries, and procedures be delayed during the coronavirus outbreak to conserve critical equipment and limit virus exposure. The guidance aligns with recent recommendations issued by American Gastroenterological Association, American Association for the Study of Liver Diseases, American College of Gastroenterology, and American Society for Gastrointestinal Endoscopy. The lack of patients has led to plummeting revenue for many GI practices and resulted in layoffs, reduced hours, and limited salaries in order to keep practices afloat.
“We’ve had to make drastic changes in the way we work,” said Rajeev Jain, MD, AGAF, a Dallas-based gastroenterologist. “The way private practices are economically set up, they don’t have large reserves of capital or liquidity. We’re not like Apple or these big companies that have these massive cushions. It’s one thing when you have a downturn in the economy and less people come to get care, but when you have a complete shutdown, your revenue stream to pay your bills is literally dried up.”
Dr. Jain’s practice is part of Texas Digestive Disease Consultants (TDDC), which provides GI care for patients in Texas and Louisiana. TDDC is part of GI Alliance, a private equity–based consolidation of practices that includes several states and more than 350 GIs. The management services organization is a collaboration between the PE firm and the partner physicians. Since the COVID-19 outbreak, Dr. Jain said his practice has seen a dramatic drop in patients. Normally, Dr. Jain would perform between 25 and 30 outpatient scopes over the course of 2 days, he said. On a recent Monday, he performed two procedures. To preserve cash flow, Dr. Jain said he and his senior partners are not taking an income right now. Some employees were recently furloughed and laid off.
“I never in my life thought that I would have to lay off people because of an economic issue,” Dr. Jain said. “That’s psychological strain that as a physician owner you feel because these are people that you work with on a day-to-day basis and you don’t want them suffering either. That’s been a tough thing.
James S. Leavitt, MD, said his 17-physician center in Miami, Fla., has furloughed about half its staff. The center is part of Gastro Health, a private equity firm–based medical group with more than 250 providers in four states. Dr. Leavitt, president and chief clinical officer for Gastro Health, said his center has gone from about 150 patients per day to 5 or fewer, while procedures have dropped from more than 100 a day to maybe 5.
Having partnered with a private equity firm, however, Dr. Leavitt believes his practice is bettered situated to manage the health crisis and address financial challenges.
“It’s made us better prepared to weather the storm. We have a very high-powered, sophisticated administration and much broader base and access to capital. [For example,] we had a lot of depth in management so that we could roll out a robust televisit program in a week in four states with over 250 doctors.”
From a business standpoint, however, certain goals for the company are on hold, he said, such as closing on potential acquisitions.
Telemedicine works well for many patients, particularly for follow-up patients and for patients who have an established relationship with Dr. Leavitt, he said. There are limitations of course, he noted.
“If I were a dermatologist, maybe I could see the skin rash, but you can’t examine the patient,” he said. “There are certain things you can’t do. If a patient has significant abdominal pain, a televisit isn’t the greatest.”
That’s why Dr. Leavitt’s care center remains open for the handful of patients who must be seen in-person, he said. Those patients are screened beforehand and their temperatures taken before treatment.
Dr. Bulsiewicz’s practice made the transition to telehealth after never having used the modality before COVID-19.
“This was a scramble,” said Dr. Bulsiewicz, who posts about COVID-19 on social media. “We started from zero knowledge to implementation in less than a week.”
Overall, the switch went smoothly, but Dr. Bulsiewicz said reimbursement challenges come with telehealth.
“The billing is not the same,” he said. “You’re doing the same work or more, and you’re taking a reduced fee because of the antiquated fee structure that is forcing you to apply the typical rules of an office encounter.”
He hopes CMS will alter the reimbursement schedule to temporarily pay on par with traditional evaluation and management codes based on medical complexity as opposed to documentation of physical exam. CMS has already expanded Medicare telehealth coverage to cover a wider range of health care services in light of the COVID-19 crisis and also broadened the range of communication tools that can be used, according to a March announcement.
In the meantime, many practices have applied for financial assistance programs. The AGA recently pushed the government for additional assistance to help struggling practices.
Dr. Jain hopes these assistance programs roll out quickly.
“If these don’t get out there quick enough and big enough, we are going to see a massive wave of loss of independent practices and/or consolidation,” he said. “I fear a death to small, independent practices because they’re not going to have the financial wherewithal to tolerate this for too long.”
On a typical clinic day, Will Bulsiewicz, MD, a Charleston, S.C.–based gastroenterologist, used to see 22 patients, while other days were filled with up to 16 procedures.
Since COVID-19 however, things have vastly changed. Dr. Bulsiewicz now visits with all clinic patients through telehealth, and the volume has dipped to between zero and six patients per day. His three-doctor practice has also experienced a more than 90% reduction in endoscopy volume.
“Naturally, this has been devastating,” Dr. Bulsiewicz said in an interview. “Our practice was started in 1984, and we had a business model that we used for the history of our practice. That practice model was upended in a matter of 2 weeks.”
Dr. Bulsiewicz is far from alone. Community GI practices across the country are experiencing similar financial distress in the face of COVID-19. In addition to a decrease in patient referrals, the Centers for Medicare & Medicaid Services has requested that all elective esophagogastroduodenoscopies, colonoscopies, endoscopies, surgeries, and procedures be delayed during the coronavirus outbreak to conserve critical equipment and limit virus exposure. The guidance aligns with recent recommendations issued by American Gastroenterological Association, American Association for the Study of Liver Diseases, American College of Gastroenterology, and American Society for Gastrointestinal Endoscopy. The lack of patients has led to plummeting revenue for many GI practices and resulted in layoffs, reduced hours, and limited salaries in order to keep practices afloat.
“We’ve had to make drastic changes in the way we work,” said Rajeev Jain, MD, AGAF, a Dallas-based gastroenterologist. “The way private practices are economically set up, they don’t have large reserves of capital or liquidity. We’re not like Apple or these big companies that have these massive cushions. It’s one thing when you have a downturn in the economy and less people come to get care, but when you have a complete shutdown, your revenue stream to pay your bills is literally dried up.”
Dr. Jain’s practice is part of Texas Digestive Disease Consultants (TDDC), which provides GI care for patients in Texas and Louisiana. TDDC is part of GI Alliance, a private equity–based consolidation of practices that includes several states and more than 350 GIs. The management services organization is a collaboration between the PE firm and the partner physicians. Since the COVID-19 outbreak, Dr. Jain said his practice has seen a dramatic drop in patients. Normally, Dr. Jain would perform between 25 and 30 outpatient scopes over the course of 2 days, he said. On a recent Monday, he performed two procedures. To preserve cash flow, Dr. Jain said he and his senior partners are not taking an income right now. Some employees were recently furloughed and laid off.
“I never in my life thought that I would have to lay off people because of an economic issue,” Dr. Jain said. “That’s psychological strain that as a physician owner you feel because these are people that you work with on a day-to-day basis and you don’t want them suffering either. That’s been a tough thing.
James S. Leavitt, MD, said his 17-physician center in Miami, Fla., has furloughed about half its staff. The center is part of Gastro Health, a private equity firm–based medical group with more than 250 providers in four states. Dr. Leavitt, president and chief clinical officer for Gastro Health, said his center has gone from about 150 patients per day to 5 or fewer, while procedures have dropped from more than 100 a day to maybe 5.
Having partnered with a private equity firm, however, Dr. Leavitt believes his practice is bettered situated to manage the health crisis and address financial challenges.
“It’s made us better prepared to weather the storm. We have a very high-powered, sophisticated administration and much broader base and access to capital. [For example,] we had a lot of depth in management so that we could roll out a robust televisit program in a week in four states with over 250 doctors.”
From a business standpoint, however, certain goals for the company are on hold, he said, such as closing on potential acquisitions.
Telemedicine works well for many patients, particularly for follow-up patients and for patients who have an established relationship with Dr. Leavitt, he said. There are limitations of course, he noted.
“If I were a dermatologist, maybe I could see the skin rash, but you can’t examine the patient,” he said. “There are certain things you can’t do. If a patient has significant abdominal pain, a televisit isn’t the greatest.”
That’s why Dr. Leavitt’s care center remains open for the handful of patients who must be seen in-person, he said. Those patients are screened beforehand and their temperatures taken before treatment.
Dr. Bulsiewicz’s practice made the transition to telehealth after never having used the modality before COVID-19.
“This was a scramble,” said Dr. Bulsiewicz, who posts about COVID-19 on social media. “We started from zero knowledge to implementation in less than a week.”
Overall, the switch went smoothly, but Dr. Bulsiewicz said reimbursement challenges come with telehealth.
“The billing is not the same,” he said. “You’re doing the same work or more, and you’re taking a reduced fee because of the antiquated fee structure that is forcing you to apply the typical rules of an office encounter.”
He hopes CMS will alter the reimbursement schedule to temporarily pay on par with traditional evaluation and management codes based on medical complexity as opposed to documentation of physical exam. CMS has already expanded Medicare telehealth coverage to cover a wider range of health care services in light of the COVID-19 crisis and also broadened the range of communication tools that can be used, according to a March announcement.
In the meantime, many practices have applied for financial assistance programs. The AGA recently pushed the government for additional assistance to help struggling practices.
Dr. Jain hopes these assistance programs roll out quickly.
“If these don’t get out there quick enough and big enough, we are going to see a massive wave of loss of independent practices and/or consolidation,” he said. “I fear a death to small, independent practices because they’re not going to have the financial wherewithal to tolerate this for too long.”
Managing gynecologic cancers during the COVID-19 pandemic
To manage patients with gynecologic cancers, oncologists in the United States and Europe are recommending reducing outpatient visits, delaying surgeries, prolonging chemotherapy regimens, and generally trying to keep cancer patients away from those who have tested positive for COVID-19.
“We recognize that, in this special situation, we must continue to provide our gynecologic oncology patients with the highest quality of medical services,” Pedro T. Ramirez, MD, of the University of Texas MD Anderson Cancer Center in Houston and associates wrote in an editorial published in the International Journal of Gynecological Cancer.
At the same time, the authors added, the safety of patients, their families, and medical staff needs to be assured.
Dr. Ramirez and colleagues’ editorial includes recommendations on how to optimize the care of patients with gynecologic cancers while prioritizing safety and minimizing the burden to the healthcare system. The group’s recommendations outline when surgery, radiotherapy, and other treatments might be safely postponed and when they need to proceed out of urgency.
Some authors of the editorial also described their experiences with COVID-19 during a webinar on managing patients with advanced ovarian cancer, which was hosted by the European Society of Gynaecological Oncology (ESGO).
A lack of resources
In Spain, health resources “are collapsed” by the pandemic, editorial author Luis Chiva, MD, said during the webinar.
At his institution, the Clínica Universidad de Navarra in Madrid, 98% of the 1,500 intensive care beds were occupied by COVID-19 patients at the end of March. So the hope was to be able to refer their patients to other communities where there may still be some capacity.
Another problem in Spain is the high percentage of health workers infected with SARS-CoV-2, the virus behind COVID-19. More than 15,000 health workers were recently reported to be sick or self-isolating, which is around 14% of the health care workforce in the country.
Dr. Chiva noted that this puts those treating gynecologic cancers in a difficult position. On the one hand, surgery to remove a high-risk ovarian mass should not be delayed, but the majority of hospitals in Spain simply cannot perform this type of surgery during the pandemic.
“Unfortunately, due to this specific situation, almost, I would say in 80%-90% of hospitals, we are only able to carry out emergency surgical procedures,” Dr. Chiva said. That’s general emergency procedures such as appendectomies, removing blockages, and dealing with hemorrhages, not gynecologic surgeries. “It’s almost impossible to schedule the typical oncological cases,” he said.
Even with the Hospital IFEMA now set up at the Feria de Madrid, which is usually used to host large-scale events, there are “minimal options for performing standard oncological surgery,” Dr. Chiva said. He estimated that just 5% of hospitals in Spain are able to perform oncologic surgeries as normal, with maybe 15% able to offer surgery without the backup of postsurgical intensive care.
‘Ring-fencing’
“This is really an unusual time for us,” commented Jonathan Ledermann, MD, vice president of ESGO and a professor of medical oncology at University College London, who moderated the webinar.
“This is affecting the way in which we diagnose our patients and have access to care,” he said. “It compromises the way in which we treat patients. We have to adjust our treatment pathways. We have to look at the risks of coronavirus infection in cancer patients and how we manage patients in a socially distancing environment. We also need to think about managing gynecological oncology departments in the face of disease amongst staff, the risks of transmission, and the reduced clinical service.”
Dr. Ledermann noted that “ring-fencing” a few hospitals to deal only with patients free of COVID-19 might be a way forward. This approach has been used in Northern Italy and was recently started in London.
“We try to divide and have separate access between COVID-positive and -negative patients,” said Anna Fagotti, MD, an assistant professor at Fondazione Policlinico Universitario Agostino Gemelli IRCCS in Rome and another coauthor of the editorial.
“We are trying to divide the work flow of patients and try to ensure treatment to cancer patients as much as we can,” she explained. “This means that it’s a very difficult situation, and, every time, you have to deal with the number of places available as some places have been taken by other patients from the emergency room. We are still trying to have a number of beds and intensive care unit beds available for our patients.”
Setting up dedicated hospitals is a good idea, but it has to be done before the “tsunami” of cases hits and there are no more intensive care beds or ventilators, according to Antonio González-Martín, MD, of Clínica Universidad de Navarra in Madrid, another coauthor of the editorial.
Limiting hospital visits
Strategies to limit the number of times patients need to come into hospital for appointments and treatment is key to getting through the pandemic, Sandro Pignata, MD, of Instituto Nazionale Tumori IRCCS Fondazione G. Pascale in Naples, Italy, said during the webinar.
“It will be imperative to explore options that reduce the number of procedures or surgical interventions that may be associated with prolonged operative time, risk of major blood loss, necessitating blood products, risk of infection to the medical personnel, or admission to intensive care units,” Dr. Ramirez and colleagues wrote in their editorial.
“In considering management of disease, we must recognize that, in many centers, access to routine visits and surgery may be either completely restricted or significantly reduced. We must, therefore, consider options that may still offer our patients a treatment plan that addresses their disease while at the same time limiting risk of exposure,” the authors wrote.
The authors declared no competing interests or specific funding in relation to their work, and the webinar participants had no conflicts of interest.
SOURCE: Ramirez PT et al. Int J Gynecol Cancer. 2020 Mar 27. doi: 10.1136/ijgc-2020-001419.
To manage patients with gynecologic cancers, oncologists in the United States and Europe are recommending reducing outpatient visits, delaying surgeries, prolonging chemotherapy regimens, and generally trying to keep cancer patients away from those who have tested positive for COVID-19.
“We recognize that, in this special situation, we must continue to provide our gynecologic oncology patients with the highest quality of medical services,” Pedro T. Ramirez, MD, of the University of Texas MD Anderson Cancer Center in Houston and associates wrote in an editorial published in the International Journal of Gynecological Cancer.
At the same time, the authors added, the safety of patients, their families, and medical staff needs to be assured.
Dr. Ramirez and colleagues’ editorial includes recommendations on how to optimize the care of patients with gynecologic cancers while prioritizing safety and minimizing the burden to the healthcare system. The group’s recommendations outline when surgery, radiotherapy, and other treatments might be safely postponed and when they need to proceed out of urgency.
Some authors of the editorial also described their experiences with COVID-19 during a webinar on managing patients with advanced ovarian cancer, which was hosted by the European Society of Gynaecological Oncology (ESGO).
A lack of resources
In Spain, health resources “are collapsed” by the pandemic, editorial author Luis Chiva, MD, said during the webinar.
At his institution, the Clínica Universidad de Navarra in Madrid, 98% of the 1,500 intensive care beds were occupied by COVID-19 patients at the end of March. So the hope was to be able to refer their patients to other communities where there may still be some capacity.
Another problem in Spain is the high percentage of health workers infected with SARS-CoV-2, the virus behind COVID-19. More than 15,000 health workers were recently reported to be sick or self-isolating, which is around 14% of the health care workforce in the country.
Dr. Chiva noted that this puts those treating gynecologic cancers in a difficult position. On the one hand, surgery to remove a high-risk ovarian mass should not be delayed, but the majority of hospitals in Spain simply cannot perform this type of surgery during the pandemic.
“Unfortunately, due to this specific situation, almost, I would say in 80%-90% of hospitals, we are only able to carry out emergency surgical procedures,” Dr. Chiva said. That’s general emergency procedures such as appendectomies, removing blockages, and dealing with hemorrhages, not gynecologic surgeries. “It’s almost impossible to schedule the typical oncological cases,” he said.
Even with the Hospital IFEMA now set up at the Feria de Madrid, which is usually used to host large-scale events, there are “minimal options for performing standard oncological surgery,” Dr. Chiva said. He estimated that just 5% of hospitals in Spain are able to perform oncologic surgeries as normal, with maybe 15% able to offer surgery without the backup of postsurgical intensive care.
‘Ring-fencing’
“This is really an unusual time for us,” commented Jonathan Ledermann, MD, vice president of ESGO and a professor of medical oncology at University College London, who moderated the webinar.
“This is affecting the way in which we diagnose our patients and have access to care,” he said. “It compromises the way in which we treat patients. We have to adjust our treatment pathways. We have to look at the risks of coronavirus infection in cancer patients and how we manage patients in a socially distancing environment. We also need to think about managing gynecological oncology departments in the face of disease amongst staff, the risks of transmission, and the reduced clinical service.”
Dr. Ledermann noted that “ring-fencing” a few hospitals to deal only with patients free of COVID-19 might be a way forward. This approach has been used in Northern Italy and was recently started in London.
“We try to divide and have separate access between COVID-positive and -negative patients,” said Anna Fagotti, MD, an assistant professor at Fondazione Policlinico Universitario Agostino Gemelli IRCCS in Rome and another coauthor of the editorial.
“We are trying to divide the work flow of patients and try to ensure treatment to cancer patients as much as we can,” she explained. “This means that it’s a very difficult situation, and, every time, you have to deal with the number of places available as some places have been taken by other patients from the emergency room. We are still trying to have a number of beds and intensive care unit beds available for our patients.”
Setting up dedicated hospitals is a good idea, but it has to be done before the “tsunami” of cases hits and there are no more intensive care beds or ventilators, according to Antonio González-Martín, MD, of Clínica Universidad de Navarra in Madrid, another coauthor of the editorial.
Limiting hospital visits
Strategies to limit the number of times patients need to come into hospital for appointments and treatment is key to getting through the pandemic, Sandro Pignata, MD, of Instituto Nazionale Tumori IRCCS Fondazione G. Pascale in Naples, Italy, said during the webinar.
“It will be imperative to explore options that reduce the number of procedures or surgical interventions that may be associated with prolonged operative time, risk of major blood loss, necessitating blood products, risk of infection to the medical personnel, or admission to intensive care units,” Dr. Ramirez and colleagues wrote in their editorial.
“In considering management of disease, we must recognize that, in many centers, access to routine visits and surgery may be either completely restricted or significantly reduced. We must, therefore, consider options that may still offer our patients a treatment plan that addresses their disease while at the same time limiting risk of exposure,” the authors wrote.
The authors declared no competing interests or specific funding in relation to their work, and the webinar participants had no conflicts of interest.
SOURCE: Ramirez PT et al. Int J Gynecol Cancer. 2020 Mar 27. doi: 10.1136/ijgc-2020-001419.
To manage patients with gynecologic cancers, oncologists in the United States and Europe are recommending reducing outpatient visits, delaying surgeries, prolonging chemotherapy regimens, and generally trying to keep cancer patients away from those who have tested positive for COVID-19.
“We recognize that, in this special situation, we must continue to provide our gynecologic oncology patients with the highest quality of medical services,” Pedro T. Ramirez, MD, of the University of Texas MD Anderson Cancer Center in Houston and associates wrote in an editorial published in the International Journal of Gynecological Cancer.
At the same time, the authors added, the safety of patients, their families, and medical staff needs to be assured.
Dr. Ramirez and colleagues’ editorial includes recommendations on how to optimize the care of patients with gynecologic cancers while prioritizing safety and minimizing the burden to the healthcare system. The group’s recommendations outline when surgery, radiotherapy, and other treatments might be safely postponed and when they need to proceed out of urgency.
Some authors of the editorial also described their experiences with COVID-19 during a webinar on managing patients with advanced ovarian cancer, which was hosted by the European Society of Gynaecological Oncology (ESGO).
A lack of resources
In Spain, health resources “are collapsed” by the pandemic, editorial author Luis Chiva, MD, said during the webinar.
At his institution, the Clínica Universidad de Navarra in Madrid, 98% of the 1,500 intensive care beds were occupied by COVID-19 patients at the end of March. So the hope was to be able to refer their patients to other communities where there may still be some capacity.
Another problem in Spain is the high percentage of health workers infected with SARS-CoV-2, the virus behind COVID-19. More than 15,000 health workers were recently reported to be sick or self-isolating, which is around 14% of the health care workforce in the country.
Dr. Chiva noted that this puts those treating gynecologic cancers in a difficult position. On the one hand, surgery to remove a high-risk ovarian mass should not be delayed, but the majority of hospitals in Spain simply cannot perform this type of surgery during the pandemic.
“Unfortunately, due to this specific situation, almost, I would say in 80%-90% of hospitals, we are only able to carry out emergency surgical procedures,” Dr. Chiva said. That’s general emergency procedures such as appendectomies, removing blockages, and dealing with hemorrhages, not gynecologic surgeries. “It’s almost impossible to schedule the typical oncological cases,” he said.
Even with the Hospital IFEMA now set up at the Feria de Madrid, which is usually used to host large-scale events, there are “minimal options for performing standard oncological surgery,” Dr. Chiva said. He estimated that just 5% of hospitals in Spain are able to perform oncologic surgeries as normal, with maybe 15% able to offer surgery without the backup of postsurgical intensive care.
‘Ring-fencing’
“This is really an unusual time for us,” commented Jonathan Ledermann, MD, vice president of ESGO and a professor of medical oncology at University College London, who moderated the webinar.
“This is affecting the way in which we diagnose our patients and have access to care,” he said. “It compromises the way in which we treat patients. We have to adjust our treatment pathways. We have to look at the risks of coronavirus infection in cancer patients and how we manage patients in a socially distancing environment. We also need to think about managing gynecological oncology departments in the face of disease amongst staff, the risks of transmission, and the reduced clinical service.”
Dr. Ledermann noted that “ring-fencing” a few hospitals to deal only with patients free of COVID-19 might be a way forward. This approach has been used in Northern Italy and was recently started in London.
“We try to divide and have separate access between COVID-positive and -negative patients,” said Anna Fagotti, MD, an assistant professor at Fondazione Policlinico Universitario Agostino Gemelli IRCCS in Rome and another coauthor of the editorial.
“We are trying to divide the work flow of patients and try to ensure treatment to cancer patients as much as we can,” she explained. “This means that it’s a very difficult situation, and, every time, you have to deal with the number of places available as some places have been taken by other patients from the emergency room. We are still trying to have a number of beds and intensive care unit beds available for our patients.”
Setting up dedicated hospitals is a good idea, but it has to be done before the “tsunami” of cases hits and there are no more intensive care beds or ventilators, according to Antonio González-Martín, MD, of Clínica Universidad de Navarra in Madrid, another coauthor of the editorial.
Limiting hospital visits
Strategies to limit the number of times patients need to come into hospital for appointments and treatment is key to getting through the pandemic, Sandro Pignata, MD, of Instituto Nazionale Tumori IRCCS Fondazione G. Pascale in Naples, Italy, said during the webinar.
“It will be imperative to explore options that reduce the number of procedures or surgical interventions that may be associated with prolonged operative time, risk of major blood loss, necessitating blood products, risk of infection to the medical personnel, or admission to intensive care units,” Dr. Ramirez and colleagues wrote in their editorial.
“In considering management of disease, we must recognize that, in many centers, access to routine visits and surgery may be either completely restricted or significantly reduced. We must, therefore, consider options that may still offer our patients a treatment plan that addresses their disease while at the same time limiting risk of exposure,” the authors wrote.
The authors declared no competing interests or specific funding in relation to their work, and the webinar participants had no conflicts of interest.
SOURCE: Ramirez PT et al. Int J Gynecol Cancer. 2020 Mar 27. doi: 10.1136/ijgc-2020-001419.
FROM THE INTERNATIONAL JOURNAL OF GYNECOLOGICAL CANCER
CMS implements temporary regulatory changes to aid COVID-19 response
The Centers for Medicare & Medicaid Services has announced a wide range of temporary regulatory moves aimed at helping hospitals and health systems handle the surge of COVID-19 patients.
“We are waiving a wide and unprecedented range of regulatory requirements to equip the American health care system with maximum flexibility to deal with an influx of cases,” CMS Administrator Seema Verma said during a March 30 conference call with reporters. “Many health care systems may not need these waivers and they shouldn’t use them if the situation doesn’t warrant it. But the flexibilities are there if it does. At a time of crisis, no regulatory barriers should stand in the way of patient care.”
Among the changes is an expansion of the venues in which health care systems and hospitals can provide services.
Federal regulations call for hospitals to provide services within their own buildings, raising concerns as to whether there will be enough capacity to handle the anticipated COVID-19 caseload.
“Under CMS’s temporary new rules, hospitals will be able to transfer patients to outside facilities, such as ambulatory surgery centers, inpatient rehabilitation hospitals, hotels, and dormitories, while still receiving hospital payments under Medicare,” the CMS stated in a fact sheet highlighting the regulatory changes.
Ambulatory surgery centers will have the option to contract with local health care systems to provide hospital services or they can enroll and bill as hospitals during the emergency, the fact sheet noted. They will be able to perform hospital services such as cancer procedures, trauma surgeries, and other essential surgeries.
CMS also is waiving the limit on the number of beds a doctor-owned hospital can have.
For Medicare patients who may be homebound, CMS will now pay for a laboratory technician to make a home visit to collect a specimen for COVID-19 testing, and hospitals will be able to conduct testing in homes or other community-based settings under certain circumstances.
In addition, CMS is taking actions aimed at expanding the health care workforce.
For instance, the agency is issuing a “blanket waiver” that allows hospitals to provide benefits to medical staff, including multiple daily meals, laundry service for personal clothing, or child care services while the staff is at the hospital providing patient care, according to the fact sheet. Teaching hospitals will also receive more flexibility in using residents to provide health care services under the virtual direction of a teaching physician, who may be available through audio/video technology.
CMS also is temporarily eliminating paperwork requirements and allowing greater use of verbal orders so that clinicians can spend more time on direct patient care.
Another change announced deals with the Merit-based Incentive Payment System (MIPS) track of the Quality Payment Program. Clinicians affected by the pandemic can request reweighting of the MIPS performance categories for the 2019 performance year, which will allow clinicians who are unable to submit MIPS data in the current submission period to request reweighting and receive a neutral payment adjustment in the 2021 payment year.
CMS also added an option for calendar year 2020 in the improvement activity category. Clinicians will receive credit if they are participating in a clinical trial that uses a drug or biological product to treat a COVID-19 patient and they report the findings to a clinical trial repository or registry.
On the device/equipment side, Medicare will cover respiratory-related devices and equipment “for any medical reason determined by clinicians,” according to the fact sheet, rather than only under certain circumstances.
And on the telehealth side, CMS is expanding the number of services that it will pay for via telehealth by more than 80, including ED visits, initial nursing facility and discharge visits, and home visits, which must be provided by a clinician that is allowed to provide telehealth. CMS will allow the use of commonly available interactive apps with audio and video, as well as audio-only phones.
CMS noted that providers can report telehealth for new and established patients, even if the billing code is specific for established patients only. CMS also has removed requirements regarding documentation of medical history and/or physical examination in the medical records during the COVID-19 crisis to help facilitate the use of telehealth for evaluation and management visits.
To help practices financially, providers who participate in Medicare fee-for-service will be able to request up to a 100% advance on their Medicare payments for a 3-month period. Repayment begins 120 days after the advance payment is received and must be paid within 210 days of the payment. Repayment will be automatically deducted from claims processed.
The agency also included new exceptions to the Stark Law. Some examples include the ability for hospitals to pay above or below fair market value to rent equipment or receive services from physicians and the allowance of health care providers to provide financial assistance to each other to ensure continuity of operations.
*This story was updated on 4/17/2020.
The Centers for Medicare & Medicaid Services has announced a wide range of temporary regulatory moves aimed at helping hospitals and health systems handle the surge of COVID-19 patients.
“We are waiving a wide and unprecedented range of regulatory requirements to equip the American health care system with maximum flexibility to deal with an influx of cases,” CMS Administrator Seema Verma said during a March 30 conference call with reporters. “Many health care systems may not need these waivers and they shouldn’t use them if the situation doesn’t warrant it. But the flexibilities are there if it does. At a time of crisis, no regulatory barriers should stand in the way of patient care.”
Among the changes is an expansion of the venues in which health care systems and hospitals can provide services.
Federal regulations call for hospitals to provide services within their own buildings, raising concerns as to whether there will be enough capacity to handle the anticipated COVID-19 caseload.
“Under CMS’s temporary new rules, hospitals will be able to transfer patients to outside facilities, such as ambulatory surgery centers, inpatient rehabilitation hospitals, hotels, and dormitories, while still receiving hospital payments under Medicare,” the CMS stated in a fact sheet highlighting the regulatory changes.
Ambulatory surgery centers will have the option to contract with local health care systems to provide hospital services or they can enroll and bill as hospitals during the emergency, the fact sheet noted. They will be able to perform hospital services such as cancer procedures, trauma surgeries, and other essential surgeries.
CMS also is waiving the limit on the number of beds a doctor-owned hospital can have.
For Medicare patients who may be homebound, CMS will now pay for a laboratory technician to make a home visit to collect a specimen for COVID-19 testing, and hospitals will be able to conduct testing in homes or other community-based settings under certain circumstances.
In addition, CMS is taking actions aimed at expanding the health care workforce.
For instance, the agency is issuing a “blanket waiver” that allows hospitals to provide benefits to medical staff, including multiple daily meals, laundry service for personal clothing, or child care services while the staff is at the hospital providing patient care, according to the fact sheet. Teaching hospitals will also receive more flexibility in using residents to provide health care services under the virtual direction of a teaching physician, who may be available through audio/video technology.
CMS also is temporarily eliminating paperwork requirements and allowing greater use of verbal orders so that clinicians can spend more time on direct patient care.
Another change announced deals with the Merit-based Incentive Payment System (MIPS) track of the Quality Payment Program. Clinicians affected by the pandemic can request reweighting of the MIPS performance categories for the 2019 performance year, which will allow clinicians who are unable to submit MIPS data in the current submission period to request reweighting and receive a neutral payment adjustment in the 2021 payment year.
CMS also added an option for calendar year 2020 in the improvement activity category. Clinicians will receive credit if they are participating in a clinical trial that uses a drug or biological product to treat a COVID-19 patient and they report the findings to a clinical trial repository or registry.
On the device/equipment side, Medicare will cover respiratory-related devices and equipment “for any medical reason determined by clinicians,” according to the fact sheet, rather than only under certain circumstances.
And on the telehealth side, CMS is expanding the number of services that it will pay for via telehealth by more than 80, including ED visits, initial nursing facility and discharge visits, and home visits, which must be provided by a clinician that is allowed to provide telehealth. CMS will allow the use of commonly available interactive apps with audio and video, as well as audio-only phones.
CMS noted that providers can report telehealth for new and established patients, even if the billing code is specific for established patients only. CMS also has removed requirements regarding documentation of medical history and/or physical examination in the medical records during the COVID-19 crisis to help facilitate the use of telehealth for evaluation and management visits.
To help practices financially, providers who participate in Medicare fee-for-service will be able to request up to a 100% advance on their Medicare payments for a 3-month period. Repayment begins 120 days after the advance payment is received and must be paid within 210 days of the payment. Repayment will be automatically deducted from claims processed.
The agency also included new exceptions to the Stark Law. Some examples include the ability for hospitals to pay above or below fair market value to rent equipment or receive services from physicians and the allowance of health care providers to provide financial assistance to each other to ensure continuity of operations.
*This story was updated on 4/17/2020.
The Centers for Medicare & Medicaid Services has announced a wide range of temporary regulatory moves aimed at helping hospitals and health systems handle the surge of COVID-19 patients.
“We are waiving a wide and unprecedented range of regulatory requirements to equip the American health care system with maximum flexibility to deal with an influx of cases,” CMS Administrator Seema Verma said during a March 30 conference call with reporters. “Many health care systems may not need these waivers and they shouldn’t use them if the situation doesn’t warrant it. But the flexibilities are there if it does. At a time of crisis, no regulatory barriers should stand in the way of patient care.”
Among the changes is an expansion of the venues in which health care systems and hospitals can provide services.
Federal regulations call for hospitals to provide services within their own buildings, raising concerns as to whether there will be enough capacity to handle the anticipated COVID-19 caseload.
“Under CMS’s temporary new rules, hospitals will be able to transfer patients to outside facilities, such as ambulatory surgery centers, inpatient rehabilitation hospitals, hotels, and dormitories, while still receiving hospital payments under Medicare,” the CMS stated in a fact sheet highlighting the regulatory changes.
Ambulatory surgery centers will have the option to contract with local health care systems to provide hospital services or they can enroll and bill as hospitals during the emergency, the fact sheet noted. They will be able to perform hospital services such as cancer procedures, trauma surgeries, and other essential surgeries.
CMS also is waiving the limit on the number of beds a doctor-owned hospital can have.
For Medicare patients who may be homebound, CMS will now pay for a laboratory technician to make a home visit to collect a specimen for COVID-19 testing, and hospitals will be able to conduct testing in homes or other community-based settings under certain circumstances.
In addition, CMS is taking actions aimed at expanding the health care workforce.
For instance, the agency is issuing a “blanket waiver” that allows hospitals to provide benefits to medical staff, including multiple daily meals, laundry service for personal clothing, or child care services while the staff is at the hospital providing patient care, according to the fact sheet. Teaching hospitals will also receive more flexibility in using residents to provide health care services under the virtual direction of a teaching physician, who may be available through audio/video technology.
CMS also is temporarily eliminating paperwork requirements and allowing greater use of verbal orders so that clinicians can spend more time on direct patient care.
Another change announced deals with the Merit-based Incentive Payment System (MIPS) track of the Quality Payment Program. Clinicians affected by the pandemic can request reweighting of the MIPS performance categories for the 2019 performance year, which will allow clinicians who are unable to submit MIPS data in the current submission period to request reweighting and receive a neutral payment adjustment in the 2021 payment year.
CMS also added an option for calendar year 2020 in the improvement activity category. Clinicians will receive credit if they are participating in a clinical trial that uses a drug or biological product to treat a COVID-19 patient and they report the findings to a clinical trial repository or registry.
On the device/equipment side, Medicare will cover respiratory-related devices and equipment “for any medical reason determined by clinicians,” according to the fact sheet, rather than only under certain circumstances.
And on the telehealth side, CMS is expanding the number of services that it will pay for via telehealth by more than 80, including ED visits, initial nursing facility and discharge visits, and home visits, which must be provided by a clinician that is allowed to provide telehealth. CMS will allow the use of commonly available interactive apps with audio and video, as well as audio-only phones.
CMS noted that providers can report telehealth for new and established patients, even if the billing code is specific for established patients only. CMS also has removed requirements regarding documentation of medical history and/or physical examination in the medical records during the COVID-19 crisis to help facilitate the use of telehealth for evaluation and management visits.
To help practices financially, providers who participate in Medicare fee-for-service will be able to request up to a 100% advance on their Medicare payments for a 3-month period. Repayment begins 120 days after the advance payment is received and must be paid within 210 days of the payment. Repayment will be automatically deducted from claims processed.
The agency also included new exceptions to the Stark Law. Some examples include the ability for hospitals to pay above or below fair market value to rent equipment or receive services from physicians and the allowance of health care providers to provide financial assistance to each other to ensure continuity of operations.
*This story was updated on 4/17/2020.
COVID-19: Managing resource crunch and ethical challenges
COVID-19 has been a watershed event in medical history of epic proportions. With this fast-spreading pandemic stretching resources at health care institutions, practical considerations for management of a disease about which we are still learning has been a huge challenge.
Although many guidelines have been made available by medical societies and experts worldwide, there appear to be very few which throw light on management in a resource-poor setup. The hospitalist, as a front-line provider, is likely expected to lead the planning and management of resources in order to deliver appropriate care.
As per American Hospital Association data, there are 2,704 community hospitals that can deliver ICU care in the United States. There are 534,964 acute care beds with 96,596 ICU beds. Additionally, there are 25,157 step-down beds and 1,183 burn unit beds. Of the 2,704 hospitals, 74% are in metropolitan areas (> 50,000 population), 17% (464) are in micropolitan areas (10,000-49,999 population), and the remaining 9% (244) are in rural areas. Only 7% (36,453) of hospital beds and 5% (4715) of ICU beds are in micropolitan areas. Two percent of acute care hospital beds and 1% of ICU beds are in rural areas. Although the US has the highest per capita number of ICU beds in the world, this may not be sufficient as these are concentrated in highly populated metropolitan areas.
Infrastructure and human power resource augmentation will be important. Infrastructure can be ramped up by:
- Canceling elective procedures
- Using the operating room and perioperative room ventilators and beds
- Servicing and using older functioning hospitals, medical wards, and ventilators.
As ventilators are expected to be in short supply, while far from ideal, other resources may include using ventilators from the Strategic National Stockpile, renting from vendors, and using state-owned stockpiles. Use of non-invasive ventilators, such as CPAP (continuous positive airway pressure), BiPAP (bi-level positive airway pressure), and HFNC (high-flow nasal cannula) may be considered in addition to full-featured ventilators. Rapidly manufacturing new ventilators with government direction is also being undertaken.
Although estimates vary based on the model used, about 1 million people are expected to need ventilatory support. However, in addition to infrastructural shortcomings, trained persons to care for these patients are lacking. Approximately 48% of acute care hospitals have no intensivists, and there are only 28,808 intensivists as per 2015 AHA data. In order to increase the amount of skilled manpower needed to staff ICUs, a model from the Society of Critical Care Medicine’s Fundamental Disaster Management Program can be adopted. This involves an intensivist overseeing four different teams, with each team caring for 24 patients. Each team is led by a non-ICU physician or an ICU advanced practice provider (APP) who in turn cares for the patient with respiratory therapists, pharmacists, ICU nurses, and other non-ICU health professionals.
It is essential that infrastructure and human power be augmented and optimized, as well as contingency plans, including triage based on ethical and legal considerations, put in place if demand overwhelms capacity.
Lack of PPE and fear among health care staff
There have been widespread reports in the media, and several anecdotal reports, about severe shortages of personal protective equipment (PPE), and as a result, an increase in fear and anxiety among frontline health care workers.
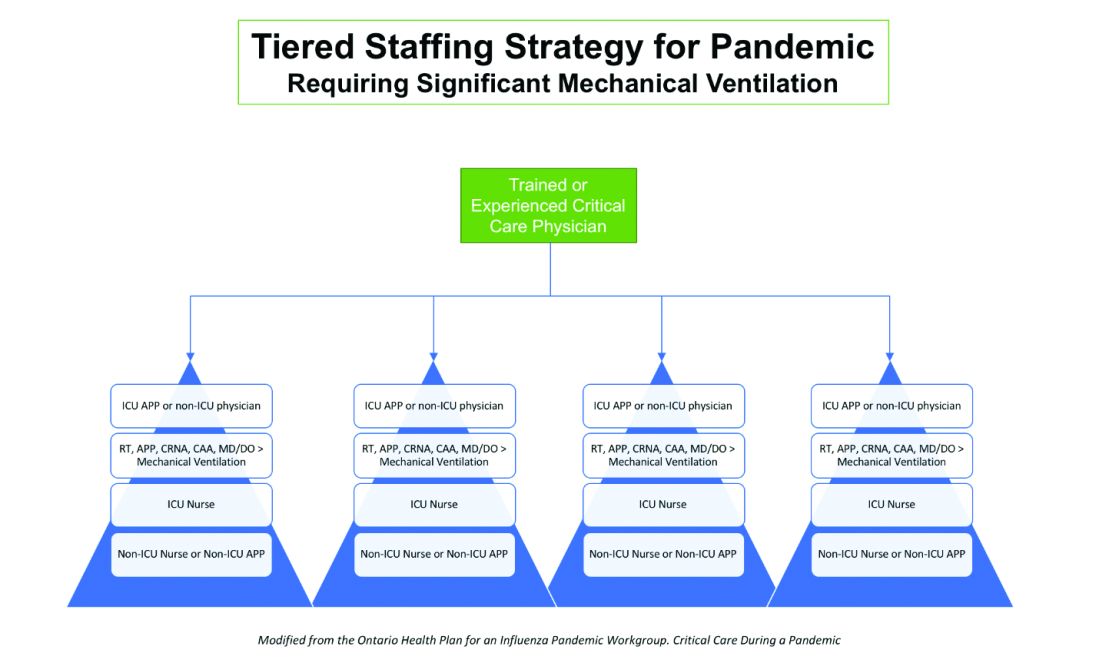
In addition, there also have been reports about hospital administrators disciplining medical and nursing staff for voicing their concerns about PPE shortages to the general public and the media. This likely stems from the narrow “optics” and public relations concerns of health care facilities.
It is evident that the size and magnitude of the COVID-19 pandemic was grossly underestimated, and preparations were inadequate. But according to past surveys of health care workers, a good number of them believe that medical and nursing staff have a duty to deliver care to sick people even if it exposes them to personal danger.
Given the special skills and privileges that health care professionals possess, they do have a moral and ethical responsibility to take care of sick patients even if a danger to themselves exists. However, society also has a responsibility to provide for the safety of these health care workers by supplying them with appropriate safety gear. Given the unprecedented nature of this pandemic, it is obvious that federal and state governments, public health officials, and hospital administrators (along with health care professionals) are still learning how to appropriately respond to the challenge.
It would be reasonable and appropriate for everyone concerned to understand and acknowledge that there is a shortage of PPE, and while arranging for this to be replenished, undertake and implement measures that maximize the safety of all health care workers. An open forum, mutually agreed-upon policy and procedures, along with mechanisms to address concerns should be formulated.
In addition, health care workers who test positive for COVID-19 can be a source of infection for other health care workers and non-infected patients. Therefore, health care workers have the responsibility of reporting their symptoms, the right to have themselves tested, and they must follow agreed-upon procedures that would limit their ability to infect other people, including mandated absenteeism from work. Every individual has a right to safety at the workplace and this right cannot be compromised, as otherwise this will lead to a suboptimal response to the pandemic. The government, hospital administrators, and health care workers will need to come together and work cohesively.
Ethical issues surrounding resource allocation
At the time of hospital admission, any suspected or confirmed COVID-19 patient should have his or her wishes recorded with the admitting team regarding the goals of care and code status. During any critical illness, goals evolve depending on discussions, reflections of the patient with family, and clinical response to therapy. A patient who does not want any kind of life support obviously should not be offered an ICU level of care.
On the other hand, in the event of resources becoming overwhelmed by demand as can be expected during this pandemic, careful ethical considerations will need to be applied.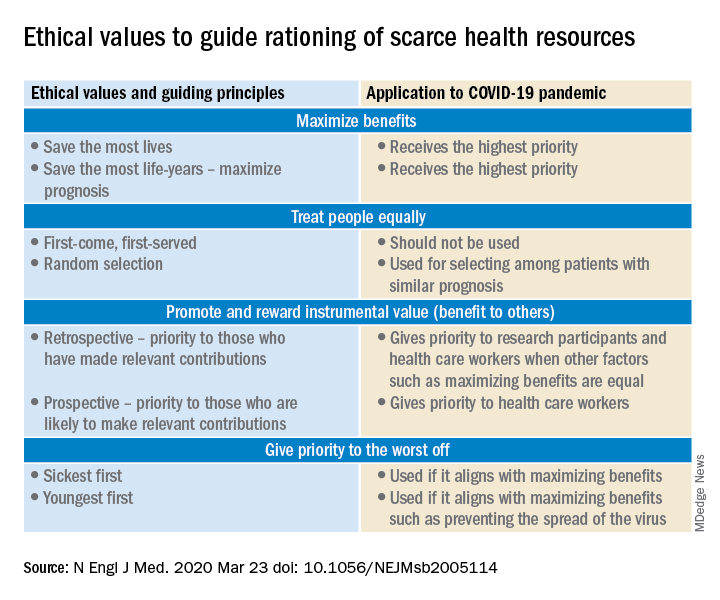
A carefully crafted transparent ethical framework, with a clear understanding of the decision-making process, that involves all stakeholders – including government entities, public health officials, health care workers, ethics specialists, and members of the community – is essential. Ideally, allocation of resources should be made according to a well-written plan, by a triage team that can include a nontreating physician, bioethicists, legal personnel, and religious representatives. It should not be left to the front-line treating physician, who is unlikely to be trained to make these decisions and who has an ethical responsibility to advocate for the patient under his care.
Ethical principles that deserve consideration
The “principle of utility” provides the maximum possible benefit to the maximum number of people. It should not only save the greatest number of lives but also maximize improvements in individuals’ posttreatment length of life.
The “principle of equity” requires that resources are allocated on a nondiscriminatory basis with a fair distribution of benefits and burdens. When conflicts arise between these two principles, a balanced approach likely will help when handled with a transparent decision-making process, with decisions to be applied consistently. Most experts would agree on not only saving more lives but also in preserving more years of life.
The distribution of medical resources should not be based on age or disability. Frailty and functional status are important considerations; however, priority is to be given to sicker patients who have lesser comorbidities and are also likely to survive longer. This could entail that younger, healthier patients will access scarce resources based on the principle of maximizing benefits.
Another consideration is “preservation of functioning of the society.” Those individuals who are important for providing important public services, health care services, and the functioning of other key aspects of society can be considered for prioritization of resources. While this may not satisfy the classic utilitarian principle of doing maximum good to the maximum number of people, it will help to continue augmenting the fight against the pandemic because of the critical role that such individuals play.
For patients with a similar prognosis, the principle of equality comes into play, and distribution should be done by way of random allocation, like a lottery. Distribution based on the principle of “first come, first served” is unlikely to be a fair process, as it would likely discriminate against patients based on their ability to access care.
Care should also be taken not to discriminate among people who have COVID-19 and non–COVID-19 health conditions that require medical care. Distribution should never be done based on an individual’s political influence, celebrity, or financial status, as occurred in the early days of the pandemic regarding access to testing.
Resuscitation or not?
Should a COVID-19 positive patient be offered CPR in case of cardiac arrest? The concern is that CPR is a high-level aerosolizing procedure and PPE is in short supply with the worsening of the pandemic. This will depend more on local policies and resource availability, along with goals of care that have to be determined at the time of admission and subsequent conversations.
The American Hospital Association has issued a general guideline and as more data become available, we can have more informed discussions with patients and families. At this point, all due precautions that prevent the infection of health care personnel are applicable.
Ethical considerations often do not have answers that are a universal fit, and the challenge is always to promote the best interest of the patient with a balance of judiciously utilizing scarce community resources.
Although many states have had discussions and some even have written policies, they have never been implemented. The organization and application of a judicious ethical “crisis level of care” is extremely challenging and likely to test the foundation and fabric of the society.
Dr. Jain is senior associate consultant, hospital & critical care medicine, at Mayo Clinic in Mankato, Minn. He is a board-certified internal medicine, pulmonary, and critical care physician, and has special interests in rural medicine and ethical issues involving critical care medicine. Dr. Tirupathi is the medical director of keystone infectious diseases/HIV in Chambersburg and is currently chair of infection prevention at Wellspan Chambersburg and Waynesboro Hospitals, all in Pennsylvania. Dr. Palabindala is hospital medicine division chief at the University of Mississippi Medical Center, Jackson, and a member of the editorial advisory board for The Hospitalist.
Sources
1. United States Resource Availability for COVID-19. SCCM Blog.
2. Intensive care medicine: Triage in case of bottlenecks. l
3. Interim Guidance for Healthcare Providers during COVID-19 Outbreak.
4. Emanuel EJ et al. Fair allocation of scarce medical resources in the time of Covid-19. N Engl J Med 2020 Mar 23.doi: 10.1056/NEJMsb2005114.
5. Devnani M et al. Planning and response to the influenza A (H1N1) pandemic: Ethics, equity and justice. Indian J Med Ethics. 2011 Oct-Dec;8(4):237-40.
6. Alexander C and Wynia M. Ready and willing? Physicians’ sense of preparedness for bioterrorism. Health Aff (Millwood). 2003 Sep-Oct;22(5):189-97.
7. Damery S et al. Healthcare workers’ perceptions of the duty to work during an influenza pandemic. J Med Ethics. 2010 Jan;36(1):12-8.
COVID-19 has been a watershed event in medical history of epic proportions. With this fast-spreading pandemic stretching resources at health care institutions, practical considerations for management of a disease about which we are still learning has been a huge challenge.
Although many guidelines have been made available by medical societies and experts worldwide, there appear to be very few which throw light on management in a resource-poor setup. The hospitalist, as a front-line provider, is likely expected to lead the planning and management of resources in order to deliver appropriate care.
As per American Hospital Association data, there are 2,704 community hospitals that can deliver ICU care in the United States. There are 534,964 acute care beds with 96,596 ICU beds. Additionally, there are 25,157 step-down beds and 1,183 burn unit beds. Of the 2,704 hospitals, 74% are in metropolitan areas (> 50,000 population), 17% (464) are in micropolitan areas (10,000-49,999 population), and the remaining 9% (244) are in rural areas. Only 7% (36,453) of hospital beds and 5% (4715) of ICU beds are in micropolitan areas. Two percent of acute care hospital beds and 1% of ICU beds are in rural areas. Although the US has the highest per capita number of ICU beds in the world, this may not be sufficient as these are concentrated in highly populated metropolitan areas.
Infrastructure and human power resource augmentation will be important. Infrastructure can be ramped up by:
- Canceling elective procedures
- Using the operating room and perioperative room ventilators and beds
- Servicing and using older functioning hospitals, medical wards, and ventilators.
As ventilators are expected to be in short supply, while far from ideal, other resources may include using ventilators from the Strategic National Stockpile, renting from vendors, and using state-owned stockpiles. Use of non-invasive ventilators, such as CPAP (continuous positive airway pressure), BiPAP (bi-level positive airway pressure), and HFNC (high-flow nasal cannula) may be considered in addition to full-featured ventilators. Rapidly manufacturing new ventilators with government direction is also being undertaken.
Although estimates vary based on the model used, about 1 million people are expected to need ventilatory support. However, in addition to infrastructural shortcomings, trained persons to care for these patients are lacking. Approximately 48% of acute care hospitals have no intensivists, and there are only 28,808 intensivists as per 2015 AHA data. In order to increase the amount of skilled manpower needed to staff ICUs, a model from the Society of Critical Care Medicine’s Fundamental Disaster Management Program can be adopted. This involves an intensivist overseeing four different teams, with each team caring for 24 patients. Each team is led by a non-ICU physician or an ICU advanced practice provider (APP) who in turn cares for the patient with respiratory therapists, pharmacists, ICU nurses, and other non-ICU health professionals.
It is essential that infrastructure and human power be augmented and optimized, as well as contingency plans, including triage based on ethical and legal considerations, put in place if demand overwhelms capacity.
Lack of PPE and fear among health care staff
There have been widespread reports in the media, and several anecdotal reports, about severe shortages of personal protective equipment (PPE), and as a result, an increase in fear and anxiety among frontline health care workers.

In addition, there also have been reports about hospital administrators disciplining medical and nursing staff for voicing their concerns about PPE shortages to the general public and the media. This likely stems from the narrow “optics” and public relations concerns of health care facilities.
It is evident that the size and magnitude of the COVID-19 pandemic was grossly underestimated, and preparations were inadequate. But according to past surveys of health care workers, a good number of them believe that medical and nursing staff have a duty to deliver care to sick people even if it exposes them to personal danger.
Given the special skills and privileges that health care professionals possess, they do have a moral and ethical responsibility to take care of sick patients even if a danger to themselves exists. However, society also has a responsibility to provide for the safety of these health care workers by supplying them with appropriate safety gear. Given the unprecedented nature of this pandemic, it is obvious that federal and state governments, public health officials, and hospital administrators (along with health care professionals) are still learning how to appropriately respond to the challenge.
It would be reasonable and appropriate for everyone concerned to understand and acknowledge that there is a shortage of PPE, and while arranging for this to be replenished, undertake and implement measures that maximize the safety of all health care workers. An open forum, mutually agreed-upon policy and procedures, along with mechanisms to address concerns should be formulated.
In addition, health care workers who test positive for COVID-19 can be a source of infection for other health care workers and non-infected patients. Therefore, health care workers have the responsibility of reporting their symptoms, the right to have themselves tested, and they must follow agreed-upon procedures that would limit their ability to infect other people, including mandated absenteeism from work. Every individual has a right to safety at the workplace and this right cannot be compromised, as otherwise this will lead to a suboptimal response to the pandemic. The government, hospital administrators, and health care workers will need to come together and work cohesively.
Ethical issues surrounding resource allocation
At the time of hospital admission, any suspected or confirmed COVID-19 patient should have his or her wishes recorded with the admitting team regarding the goals of care and code status. During any critical illness, goals evolve depending on discussions, reflections of the patient with family, and clinical response to therapy. A patient who does not want any kind of life support obviously should not be offered an ICU level of care.
On the other hand, in the event of resources becoming overwhelmed by demand as can be expected during this pandemic, careful ethical considerations will need to be applied.
A carefully crafted transparent ethical framework, with a clear understanding of the decision-making process, that involves all stakeholders – including government entities, public health officials, health care workers, ethics specialists, and members of the community – is essential. Ideally, allocation of resources should be made according to a well-written plan, by a triage team that can include a nontreating physician, bioethicists, legal personnel, and religious representatives. It should not be left to the front-line treating physician, who is unlikely to be trained to make these decisions and who has an ethical responsibility to advocate for the patient under his care.
Ethical principles that deserve consideration
The “principle of utility” provides the maximum possible benefit to the maximum number of people. It should not only save the greatest number of lives but also maximize improvements in individuals’ posttreatment length of life.
The “principle of equity” requires that resources are allocated on a nondiscriminatory basis with a fair distribution of benefits and burdens. When conflicts arise between these two principles, a balanced approach likely will help when handled with a transparent decision-making process, with decisions to be applied consistently. Most experts would agree on not only saving more lives but also in preserving more years of life.
The distribution of medical resources should not be based on age or disability. Frailty and functional status are important considerations; however, priority is to be given to sicker patients who have lesser comorbidities and are also likely to survive longer. This could entail that younger, healthier patients will access scarce resources based on the principle of maximizing benefits.
Another consideration is “preservation of functioning of the society.” Those individuals who are important for providing important public services, health care services, and the functioning of other key aspects of society can be considered for prioritization of resources. While this may not satisfy the classic utilitarian principle of doing maximum good to the maximum number of people, it will help to continue augmenting the fight against the pandemic because of the critical role that such individuals play.
For patients with a similar prognosis, the principle of equality comes into play, and distribution should be done by way of random allocation, like a lottery. Distribution based on the principle of “first come, first served” is unlikely to be a fair process, as it would likely discriminate against patients based on their ability to access care.
Care should also be taken not to discriminate among people who have COVID-19 and non–COVID-19 health conditions that require medical care. Distribution should never be done based on an individual’s political influence, celebrity, or financial status, as occurred in the early days of the pandemic regarding access to testing.
Resuscitation or not?
Should a COVID-19 positive patient be offered CPR in case of cardiac arrest? The concern is that CPR is a high-level aerosolizing procedure and PPE is in short supply with the worsening of the pandemic. This will depend more on local policies and resource availability, along with goals of care that have to be determined at the time of admission and subsequent conversations.
The American Hospital Association has issued a general guideline and as more data become available, we can have more informed discussions with patients and families. At this point, all due precautions that prevent the infection of health care personnel are applicable.
Ethical considerations often do not have answers that are a universal fit, and the challenge is always to promote the best interest of the patient with a balance of judiciously utilizing scarce community resources.
Although many states have had discussions and some even have written policies, they have never been implemented. The organization and application of a judicious ethical “crisis level of care” is extremely challenging and likely to test the foundation and fabric of the society.
Dr. Jain is senior associate consultant, hospital & critical care medicine, at Mayo Clinic in Mankato, Minn. He is a board-certified internal medicine, pulmonary, and critical care physician, and has special interests in rural medicine and ethical issues involving critical care medicine. Dr. Tirupathi is the medical director of keystone infectious diseases/HIV in Chambersburg and is currently chair of infection prevention at Wellspan Chambersburg and Waynesboro Hospitals, all in Pennsylvania. Dr. Palabindala is hospital medicine division chief at the University of Mississippi Medical Center, Jackson, and a member of the editorial advisory board for The Hospitalist.
Sources
1. United States Resource Availability for COVID-19. SCCM Blog.
2. Intensive care medicine: Triage in case of bottlenecks. l
3. Interim Guidance for Healthcare Providers during COVID-19 Outbreak.
4. Emanuel EJ et al. Fair allocation of scarce medical resources in the time of Covid-19. N Engl J Med 2020 Mar 23.doi: 10.1056/NEJMsb2005114.
5. Devnani M et al. Planning and response to the influenza A (H1N1) pandemic: Ethics, equity and justice. Indian J Med Ethics. 2011 Oct-Dec;8(4):237-40.
6. Alexander C and Wynia M. Ready and willing? Physicians’ sense of preparedness for bioterrorism. Health Aff (Millwood). 2003 Sep-Oct;22(5):189-97.
7. Damery S et al. Healthcare workers’ perceptions of the duty to work during an influenza pandemic. J Med Ethics. 2010 Jan;36(1):12-8.
COVID-19 has been a watershed event in medical history of epic proportions. With this fast-spreading pandemic stretching resources at health care institutions, practical considerations for management of a disease about which we are still learning has been a huge challenge.
Although many guidelines have been made available by medical societies and experts worldwide, there appear to be very few which throw light on management in a resource-poor setup. The hospitalist, as a front-line provider, is likely expected to lead the planning and management of resources in order to deliver appropriate care.
As per American Hospital Association data, there are 2,704 community hospitals that can deliver ICU care in the United States. There are 534,964 acute care beds with 96,596 ICU beds. Additionally, there are 25,157 step-down beds and 1,183 burn unit beds. Of the 2,704 hospitals, 74% are in metropolitan areas (> 50,000 population), 17% (464) are in micropolitan areas (10,000-49,999 population), and the remaining 9% (244) are in rural areas. Only 7% (36,453) of hospital beds and 5% (4715) of ICU beds are in micropolitan areas. Two percent of acute care hospital beds and 1% of ICU beds are in rural areas. Although the US has the highest per capita number of ICU beds in the world, this may not be sufficient as these are concentrated in highly populated metropolitan areas.
Infrastructure and human power resource augmentation will be important. Infrastructure can be ramped up by:
- Canceling elective procedures
- Using the operating room and perioperative room ventilators and beds
- Servicing and using older functioning hospitals, medical wards, and ventilators.
As ventilators are expected to be in short supply, while far from ideal, other resources may include using ventilators from the Strategic National Stockpile, renting from vendors, and using state-owned stockpiles. Use of non-invasive ventilators, such as CPAP (continuous positive airway pressure), BiPAP (bi-level positive airway pressure), and HFNC (high-flow nasal cannula) may be considered in addition to full-featured ventilators. Rapidly manufacturing new ventilators with government direction is also being undertaken.
Although estimates vary based on the model used, about 1 million people are expected to need ventilatory support. However, in addition to infrastructural shortcomings, trained persons to care for these patients are lacking. Approximately 48% of acute care hospitals have no intensivists, and there are only 28,808 intensivists as per 2015 AHA data. In order to increase the amount of skilled manpower needed to staff ICUs, a model from the Society of Critical Care Medicine’s Fundamental Disaster Management Program can be adopted. This involves an intensivist overseeing four different teams, with each team caring for 24 patients. Each team is led by a non-ICU physician or an ICU advanced practice provider (APP) who in turn cares for the patient with respiratory therapists, pharmacists, ICU nurses, and other non-ICU health professionals.
It is essential that infrastructure and human power be augmented and optimized, as well as contingency plans, including triage based on ethical and legal considerations, put in place if demand overwhelms capacity.
Lack of PPE and fear among health care staff
There have been widespread reports in the media, and several anecdotal reports, about severe shortages of personal protective equipment (PPE), and as a result, an increase in fear and anxiety among frontline health care workers.

In addition, there also have been reports about hospital administrators disciplining medical and nursing staff for voicing their concerns about PPE shortages to the general public and the media. This likely stems from the narrow “optics” and public relations concerns of health care facilities.
It is evident that the size and magnitude of the COVID-19 pandemic was grossly underestimated, and preparations were inadequate. But according to past surveys of health care workers, a good number of them believe that medical and nursing staff have a duty to deliver care to sick people even if it exposes them to personal danger.
Given the special skills and privileges that health care professionals possess, they do have a moral and ethical responsibility to take care of sick patients even if a danger to themselves exists. However, society also has a responsibility to provide for the safety of these health care workers by supplying them with appropriate safety gear. Given the unprecedented nature of this pandemic, it is obvious that federal and state governments, public health officials, and hospital administrators (along with health care professionals) are still learning how to appropriately respond to the challenge.
It would be reasonable and appropriate for everyone concerned to understand and acknowledge that there is a shortage of PPE, and while arranging for this to be replenished, undertake and implement measures that maximize the safety of all health care workers. An open forum, mutually agreed-upon policy and procedures, along with mechanisms to address concerns should be formulated.
In addition, health care workers who test positive for COVID-19 can be a source of infection for other health care workers and non-infected patients. Therefore, health care workers have the responsibility of reporting their symptoms, the right to have themselves tested, and they must follow agreed-upon procedures that would limit their ability to infect other people, including mandated absenteeism from work. Every individual has a right to safety at the workplace and this right cannot be compromised, as otherwise this will lead to a suboptimal response to the pandemic. The government, hospital administrators, and health care workers will need to come together and work cohesively.
Ethical issues surrounding resource allocation
At the time of hospital admission, any suspected or confirmed COVID-19 patient should have his or her wishes recorded with the admitting team regarding the goals of care and code status. During any critical illness, goals evolve depending on discussions, reflections of the patient with family, and clinical response to therapy. A patient who does not want any kind of life support obviously should not be offered an ICU level of care.
On the other hand, in the event of resources becoming overwhelmed by demand as can be expected during this pandemic, careful ethical considerations will need to be applied.
A carefully crafted transparent ethical framework, with a clear understanding of the decision-making process, that involves all stakeholders – including government entities, public health officials, health care workers, ethics specialists, and members of the community – is essential. Ideally, allocation of resources should be made according to a well-written plan, by a triage team that can include a nontreating physician, bioethicists, legal personnel, and religious representatives. It should not be left to the front-line treating physician, who is unlikely to be trained to make these decisions and who has an ethical responsibility to advocate for the patient under his care.
Ethical principles that deserve consideration
The “principle of utility” provides the maximum possible benefit to the maximum number of people. It should not only save the greatest number of lives but also maximize improvements in individuals’ posttreatment length of life.
The “principle of equity” requires that resources are allocated on a nondiscriminatory basis with a fair distribution of benefits and burdens. When conflicts arise between these two principles, a balanced approach likely will help when handled with a transparent decision-making process, with decisions to be applied consistently. Most experts would agree on not only saving more lives but also in preserving more years of life.
The distribution of medical resources should not be based on age or disability. Frailty and functional status are important considerations; however, priority is to be given to sicker patients who have lesser comorbidities and are also likely to survive longer. This could entail that younger, healthier patients will access scarce resources based on the principle of maximizing benefits.
Another consideration is “preservation of functioning of the society.” Those individuals who are important for providing important public services, health care services, and the functioning of other key aspects of society can be considered for prioritization of resources. While this may not satisfy the classic utilitarian principle of doing maximum good to the maximum number of people, it will help to continue augmenting the fight against the pandemic because of the critical role that such individuals play.
For patients with a similar prognosis, the principle of equality comes into play, and distribution should be done by way of random allocation, like a lottery. Distribution based on the principle of “first come, first served” is unlikely to be a fair process, as it would likely discriminate against patients based on their ability to access care.
Care should also be taken not to discriminate among people who have COVID-19 and non–COVID-19 health conditions that require medical care. Distribution should never be done based on an individual’s political influence, celebrity, or financial status, as occurred in the early days of the pandemic regarding access to testing.
Resuscitation or not?
Should a COVID-19 positive patient be offered CPR in case of cardiac arrest? The concern is that CPR is a high-level aerosolizing procedure and PPE is in short supply with the worsening of the pandemic. This will depend more on local policies and resource availability, along with goals of care that have to be determined at the time of admission and subsequent conversations.
The American Hospital Association has issued a general guideline and as more data become available, we can have more informed discussions with patients and families. At this point, all due precautions that prevent the infection of health care personnel are applicable.
Ethical considerations often do not have answers that are a universal fit, and the challenge is always to promote the best interest of the patient with a balance of judiciously utilizing scarce community resources.
Although many states have had discussions and some even have written policies, they have never been implemented. The organization and application of a judicious ethical “crisis level of care” is extremely challenging and likely to test the foundation and fabric of the society.
Dr. Jain is senior associate consultant, hospital & critical care medicine, at Mayo Clinic in Mankato, Minn. He is a board-certified internal medicine, pulmonary, and critical care physician, and has special interests in rural medicine and ethical issues involving critical care medicine. Dr. Tirupathi is the medical director of keystone infectious diseases/HIV in Chambersburg and is currently chair of infection prevention at Wellspan Chambersburg and Waynesboro Hospitals, all in Pennsylvania. Dr. Palabindala is hospital medicine division chief at the University of Mississippi Medical Center, Jackson, and a member of the editorial advisory board for The Hospitalist.
Sources
1. United States Resource Availability for COVID-19. SCCM Blog.
2. Intensive care medicine: Triage in case of bottlenecks. l
3. Interim Guidance for Healthcare Providers during COVID-19 Outbreak.
4. Emanuel EJ et al. Fair allocation of scarce medical resources in the time of Covid-19. N Engl J Med 2020 Mar 23.doi: 10.1056/NEJMsb2005114.
5. Devnani M et al. Planning and response to the influenza A (H1N1) pandemic: Ethics, equity and justice. Indian J Med Ethics. 2011 Oct-Dec;8(4):237-40.
6. Alexander C and Wynia M. Ready and willing? Physicians’ sense of preparedness for bioterrorism. Health Aff (Millwood). 2003 Sep-Oct;22(5):189-97.
7. Damery S et al. Healthcare workers’ perceptions of the duty to work during an influenza pandemic. J Med Ethics. 2010 Jan;36(1):12-8.