User login
Fauci: Cautious optimism for COVID-19 vaccine by end of 2020
with distribution of first doses possible before the end of the year, according to Anthony S. Fauci, MD, director, National Institute of Allergy and Infectious Diseases, Bethesda, Md.
“Given the rate of infection that’s going on in this country, and the distribution of the clinical trial sites involving tens of thousands of volunteers, we project that we will have an answer as to whether or not we have a safe and effective vaccine by November or December,” Dr. Fauci said today in his virtual keynote address during the annual meeting of the American College of Chest Physicians.
“It may come earlier -- this month, in October,” he added in his remarks. “That is unlikely – it is more likely that we’ll have an answer in November and December.”
If that timing does come to pass, Dr. Fauci said, it’s possible that distribution of doses could start at the end of the year, continuing throughout the beginning and middle of 2021.
Although there are no guarantees, Dr. Fauci said he is “cautiously optimistic” regarding the timeline.
He said that his optimism is based in part on animal studies and phase 1 data that demonstrate robust neutralizing antibody responses to a vaccine that are equivalent to, if not greater than, natural infection with the SARS-CoV-2 virus that causes COVID-19.
Rapid development gives reason for hope
Ryan C. Maves, MD, FCCP, a critical care and infectious disease specialist at Naval Medical Center San Diego, said there is reason to be hopeful that a vaccine will be available by the end of the calendar year. He cautioned, however, that this timing is based on the assumption that one of the vaccines will be proven safe and effective very soon.
“We’re lucky to have multiple phase 3 trials using multiple vaccine technologies in different platforms,” Dr. Maves said in a panel discussion following Dr. Fauci’s remarks. “I think the odds are very high that one of them will be effective.”
“I’m hoping that multiple vaccines will be effective,” Dr. Maves added. “Then we’ll be in a good position of determining which is the best of several good options, as a society and as a world.”
COVID-19 vaccine development over the past year has been remarkably fast, especially given the previous record set by the mumps vaccine, which took about four years to go from initial steps to rollout, Dr. Maves noted.
Dr. Fauci said the federal government has taken a “strategic approach” to the COVID-19 vaccine that includes direct involvement in the research and development of six different vaccine candidates, five of which are now in phase 3 trials.
As part of that strategic approach, the study protocols are harmonized to have a common data and safety monitoring board, common primary and secondary endpoints, and an independent statistical group to determine correlates of protection, Dr. Fauci said.
Prioritizing COVID-19 vaccine distribution
Who gets COVID-19 vaccine first will be a challenge for governmental organizations as well as bioethicists, who have proposed different strategies for fairly prioritizing different groups for access.
Reaching communities of color will be an important consideration for prioritization, according to Dr. Maves, given the disproportionate burden of disease on Black and Hispanic individuals, among other such populations.
COVID-19–related hospitalization rates have been substantially higher in communities of color, Dr. Fauci said in his keynote address. Age-adjusted hospitalization rates for Hispanic/Latinx and Black populations are 375 to 368 per 100,000, respectively, compared with just 82 per 100,000 for White non-Hispanics, according to data from the Centers for Disease Control and Prevention.
Outreach to those communities should include building trust in those populations that they will benefit from a safe and effective vaccine, and making sure that the vaccine is available to those communities as quickly as possible, Dr. Maves said.
Dr. Fauci and Dr. Maves provided no disclosures related to their presentations.
with distribution of first doses possible before the end of the year, according to Anthony S. Fauci, MD, director, National Institute of Allergy and Infectious Diseases, Bethesda, Md.
“Given the rate of infection that’s going on in this country, and the distribution of the clinical trial sites involving tens of thousands of volunteers, we project that we will have an answer as to whether or not we have a safe and effective vaccine by November or December,” Dr. Fauci said today in his virtual keynote address during the annual meeting of the American College of Chest Physicians.
“It may come earlier -- this month, in October,” he added in his remarks. “That is unlikely – it is more likely that we’ll have an answer in November and December.”
If that timing does come to pass, Dr. Fauci said, it’s possible that distribution of doses could start at the end of the year, continuing throughout the beginning and middle of 2021.
Although there are no guarantees, Dr. Fauci said he is “cautiously optimistic” regarding the timeline.
He said that his optimism is based in part on animal studies and phase 1 data that demonstrate robust neutralizing antibody responses to a vaccine that are equivalent to, if not greater than, natural infection with the SARS-CoV-2 virus that causes COVID-19.
Rapid development gives reason for hope
Ryan C. Maves, MD, FCCP, a critical care and infectious disease specialist at Naval Medical Center San Diego, said there is reason to be hopeful that a vaccine will be available by the end of the calendar year. He cautioned, however, that this timing is based on the assumption that one of the vaccines will be proven safe and effective very soon.
“We’re lucky to have multiple phase 3 trials using multiple vaccine technologies in different platforms,” Dr. Maves said in a panel discussion following Dr. Fauci’s remarks. “I think the odds are very high that one of them will be effective.”
“I’m hoping that multiple vaccines will be effective,” Dr. Maves added. “Then we’ll be in a good position of determining which is the best of several good options, as a society and as a world.”
COVID-19 vaccine development over the past year has been remarkably fast, especially given the previous record set by the mumps vaccine, which took about four years to go from initial steps to rollout, Dr. Maves noted.
Dr. Fauci said the federal government has taken a “strategic approach” to the COVID-19 vaccine that includes direct involvement in the research and development of six different vaccine candidates, five of which are now in phase 3 trials.
As part of that strategic approach, the study protocols are harmonized to have a common data and safety monitoring board, common primary and secondary endpoints, and an independent statistical group to determine correlates of protection, Dr. Fauci said.
Prioritizing COVID-19 vaccine distribution
Who gets COVID-19 vaccine first will be a challenge for governmental organizations as well as bioethicists, who have proposed different strategies for fairly prioritizing different groups for access.
Reaching communities of color will be an important consideration for prioritization, according to Dr. Maves, given the disproportionate burden of disease on Black and Hispanic individuals, among other such populations.
COVID-19–related hospitalization rates have been substantially higher in communities of color, Dr. Fauci said in his keynote address. Age-adjusted hospitalization rates for Hispanic/Latinx and Black populations are 375 to 368 per 100,000, respectively, compared with just 82 per 100,000 for White non-Hispanics, according to data from the Centers for Disease Control and Prevention.
Outreach to those communities should include building trust in those populations that they will benefit from a safe and effective vaccine, and making sure that the vaccine is available to those communities as quickly as possible, Dr. Maves said.
Dr. Fauci and Dr. Maves provided no disclosures related to their presentations.
with distribution of first doses possible before the end of the year, according to Anthony S. Fauci, MD, director, National Institute of Allergy and Infectious Diseases, Bethesda, Md.
“Given the rate of infection that’s going on in this country, and the distribution of the clinical trial sites involving tens of thousands of volunteers, we project that we will have an answer as to whether or not we have a safe and effective vaccine by November or December,” Dr. Fauci said today in his virtual keynote address during the annual meeting of the American College of Chest Physicians.
“It may come earlier -- this month, in October,” he added in his remarks. “That is unlikely – it is more likely that we’ll have an answer in November and December.”
If that timing does come to pass, Dr. Fauci said, it’s possible that distribution of doses could start at the end of the year, continuing throughout the beginning and middle of 2021.
Although there are no guarantees, Dr. Fauci said he is “cautiously optimistic” regarding the timeline.
He said that his optimism is based in part on animal studies and phase 1 data that demonstrate robust neutralizing antibody responses to a vaccine that are equivalent to, if not greater than, natural infection with the SARS-CoV-2 virus that causes COVID-19.
Rapid development gives reason for hope
Ryan C. Maves, MD, FCCP, a critical care and infectious disease specialist at Naval Medical Center San Diego, said there is reason to be hopeful that a vaccine will be available by the end of the calendar year. He cautioned, however, that this timing is based on the assumption that one of the vaccines will be proven safe and effective very soon.
“We’re lucky to have multiple phase 3 trials using multiple vaccine technologies in different platforms,” Dr. Maves said in a panel discussion following Dr. Fauci’s remarks. “I think the odds are very high that one of them will be effective.”
“I’m hoping that multiple vaccines will be effective,” Dr. Maves added. “Then we’ll be in a good position of determining which is the best of several good options, as a society and as a world.”
COVID-19 vaccine development over the past year has been remarkably fast, especially given the previous record set by the mumps vaccine, which took about four years to go from initial steps to rollout, Dr. Maves noted.
Dr. Fauci said the federal government has taken a “strategic approach” to the COVID-19 vaccine that includes direct involvement in the research and development of six different vaccine candidates, five of which are now in phase 3 trials.
As part of that strategic approach, the study protocols are harmonized to have a common data and safety monitoring board, common primary and secondary endpoints, and an independent statistical group to determine correlates of protection, Dr. Fauci said.
Prioritizing COVID-19 vaccine distribution
Who gets COVID-19 vaccine first will be a challenge for governmental organizations as well as bioethicists, who have proposed different strategies for fairly prioritizing different groups for access.
Reaching communities of color will be an important consideration for prioritization, according to Dr. Maves, given the disproportionate burden of disease on Black and Hispanic individuals, among other such populations.
COVID-19–related hospitalization rates have been substantially higher in communities of color, Dr. Fauci said in his keynote address. Age-adjusted hospitalization rates for Hispanic/Latinx and Black populations are 375 to 368 per 100,000, respectively, compared with just 82 per 100,000 for White non-Hispanics, according to data from the Centers for Disease Control and Prevention.
Outreach to those communities should include building trust in those populations that they will benefit from a safe and effective vaccine, and making sure that the vaccine is available to those communities as quickly as possible, Dr. Maves said.
Dr. Fauci and Dr. Maves provided no disclosures related to their presentations.
FROM CHEST 2020
NACMI: Clear benefit with PCI in STEMI COVID-19 patients
Patients with COVID-19 who present with ST-segment elevation MI (STEMI) represent a unique, high-risk population with greater risks for in-hospital death and stroke, according to initial results from the North American COVID-19 ST-Segment Elevation Myocardial Infarction Registry (NACMI).
Although COVID-19–confirmed patients were less likely to undergo angiography than patients under investigation (PUI) for COVID-19 or historical STEMI activation controls, 71% underwent primary percutaneous coronary intervention (PCI).
“Primary PCI is preferable and feasible in COVID-19–positive patients, with door-to-balloon times similar to PUI or COVID-negative patients, and that supports the updated COVID-specific STEMI guidelines,” study cochair Timothy D. Henry, MD, said in a late-breaking clinical science session at TCT 2020, the Transcatheter Cardiovascular Therapeutics virtual annual meeting.
The multisociety COVID-specific guidelines were initially issued in April, endorsing PCI as the standard of care and allowing for consideration of fibrinolysis-based therapy at non-PCI capable hospitals.
Five previous publications on a total of 174 COVID-19 patients with ST-elevation have shown there are more frequent in-hospital STEMI presentations, more cases without a clear culprit lesion, more thrombotic lesions and microthrombi, and higher mortality, ranging from 12% to 72%. Still, there has been considerable controversy over exactly what to do when COVID-19 patients with ST elevation reach the cath lab, he said at the meeting sponsored by the Cardiovascular Research Foundation.
NACMI represents the largest experience with ST-elevation patients and is a unique collaboration between the Society for Cardiovascular Angiography and Interventions, Canadian Association of Interventional Cardiology, American College of Cardiology, and Midwest STEMI Consortium, noted Dr. Henry, who is medical director of the Lindner Center for Research and Education at the Christ Hospital, Cincinnati.
The registry enrolled any COVID-19–positive patient or person under investigation older than 18 years with ST-segment elevation or new-onset left bundle branch block on electrocardiogram with a clinical correlate of myocardial ischemia such as chest pain, dyspnea, cardiac arrest, shock, or mechanical ventilation. There were no exclusion criteria.
Data from 171 patients with confirmed COVID-19 and 423 PUI from 64 sites were then propensity-matched to a control population from the Midwest STEMI Consortium, a prospective, multicenter registry of consecutive STEMI patients.
The three groups were similar in sex and age but there was a striking difference in race, with 27% of African American and 24% of Hispanic patients COVID-confirmed, compared with 11% and 6% in the PUI group and 4% and 1% in the control group. Likewise, there was a significant increase in diabetes (44% vs. 33% vs. 20%), which has been reported previously with influenza.
COVID-19–positive patients, as compared with PUI and controls, were significantly more likely to present with cardiogenic shock before PCI (20% vs. 14% vs. 5%), but not cardiac arrest (12% vs. 17% vs. 11%), and to have lower left ventricular ejection fractions (45% vs. 45% vs. 50%).
They also presented with more atypical symptoms than PUI patients, particularly infiltrates on chest x-ray (49% vs. 17%) and dyspnea (58% vs. 38%). Data were not available for these outcomes among historic controls.
Importantly, 21% of the COVID-19 patients did not undergo angiography, compared with 5% of PUI patients and 0% of controls (P < .001), “which is much higher than we would expect or have suspected,” Dr. Henry said. Thrombolytic use was very uncommon in those undergoing angiography, likely as a result of the guidelines.
Very surprisingly, there were no differences in door-to-balloon times between the COVID-positive, PUI, and control groups despite the ongoing pandemic (80 min vs. 78 min vs. 86 min).
But there was clear worsening in in-hospital mortality in COVID-19–positive patients (32% vs. 12% and 6%; P < .001), as well as in-hospital stroke (3.4% vs. 2% vs. 0.6%) that reached statistical significance only when compared with historical controls (P = .039). Total length of stay was twice as long in COVID-confirmed patients as in both PUI and controls (6 days vs. 3 days; P < .001).
Following the formal presentation, invited discussant Philippe Gabriel Steg, MD, Imperial College London, said the researchers have provided a great service in reporting the data so quickly but noted that an ongoing French registry of events before, during, and after the first COVID-19 wave has not seen an increased death rate.
“Can you tease out whether the increased death rate is related to cardiovascular deaths or to COVID-related pneumonias, shocks, ARDSs [acute respiratory distress syndromes], and so on and so forth? Because our impression – and that’s what we’ve published in Lancet Public Health – is that the cardiovascular morality rate doesn’t seem that affected by COVID.”
Dr. Henry replied that these are early data but “I will tell you that patients who did get PCI had a mortality rate that was only around 12% or 13%, and the patients who did not undergo angiography or were treated with medical therapy had higher mortality. Now, of course, that’s selected and we need to do a much better matching and look at that, but that’s our goal and we will have that information,” he said.
During a press briefing on the study, discussant Renu Virmani, MD, president and founder of CVPath Institute, noted that, in their analysis of 40 autopsy cases from Bergamot, Italy, small intramyocardial microthrombi were seen in nine patients, whereas epicardial microthrombi were seen in only three or four.
“Some of the cases are being taken as being related to coronary disease but may be more thrombotic than anything else,” she said. “I think there’s a combination, and that’s why the outcomes are so poor. You didn’t show us TIMI flow but that’s something to think about: Was TIMI flow different in the patients who died because you have very high mortality? I think we need to get to the bottom of what is the underlying cause of that thrombosis.”
Future topics of interest include ethnic and regional/country differences; time-to-treatment including chest pain onset-to-arrival; transfer, in-hospital, and no-culprit patients; changes over time during the pandemic; and eventually 1-year outcomes, Dr. Henry said.
Press briefing moderator Ajay Kirtane, MD, director of the cardiac catheterization labs at NewYork-Presbyterian/Columbia University Irving, New York, remarked that “a lot of times people will pooh-pooh observational data, but this is exactly the type of data that we need to try to be able to gather information about what our practices are, how they fit. And I think many of us around the world will see these data, and it will echo their own experience.”
The study was funded by the Society for Cardiovascular Angiography and Interventions and the Canadian Association of Interventional Cardiology. Dr. Henry has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Patients with COVID-19 who present with ST-segment elevation MI (STEMI) represent a unique, high-risk population with greater risks for in-hospital death and stroke, according to initial results from the North American COVID-19 ST-Segment Elevation Myocardial Infarction Registry (NACMI).
Although COVID-19–confirmed patients were less likely to undergo angiography than patients under investigation (PUI) for COVID-19 or historical STEMI activation controls, 71% underwent primary percutaneous coronary intervention (PCI).
“Primary PCI is preferable and feasible in COVID-19–positive patients, with door-to-balloon times similar to PUI or COVID-negative patients, and that supports the updated COVID-specific STEMI guidelines,” study cochair Timothy D. Henry, MD, said in a late-breaking clinical science session at TCT 2020, the Transcatheter Cardiovascular Therapeutics virtual annual meeting.
The multisociety COVID-specific guidelines were initially issued in April, endorsing PCI as the standard of care and allowing for consideration of fibrinolysis-based therapy at non-PCI capable hospitals.
Five previous publications on a total of 174 COVID-19 patients with ST-elevation have shown there are more frequent in-hospital STEMI presentations, more cases without a clear culprit lesion, more thrombotic lesions and microthrombi, and higher mortality, ranging from 12% to 72%. Still, there has been considerable controversy over exactly what to do when COVID-19 patients with ST elevation reach the cath lab, he said at the meeting sponsored by the Cardiovascular Research Foundation.
NACMI represents the largest experience with ST-elevation patients and is a unique collaboration between the Society for Cardiovascular Angiography and Interventions, Canadian Association of Interventional Cardiology, American College of Cardiology, and Midwest STEMI Consortium, noted Dr. Henry, who is medical director of the Lindner Center for Research and Education at the Christ Hospital, Cincinnati.
The registry enrolled any COVID-19–positive patient or person under investigation older than 18 years with ST-segment elevation or new-onset left bundle branch block on electrocardiogram with a clinical correlate of myocardial ischemia such as chest pain, dyspnea, cardiac arrest, shock, or mechanical ventilation. There were no exclusion criteria.
Data from 171 patients with confirmed COVID-19 and 423 PUI from 64 sites were then propensity-matched to a control population from the Midwest STEMI Consortium, a prospective, multicenter registry of consecutive STEMI patients.
The three groups were similar in sex and age but there was a striking difference in race, with 27% of African American and 24% of Hispanic patients COVID-confirmed, compared with 11% and 6% in the PUI group and 4% and 1% in the control group. Likewise, there was a significant increase in diabetes (44% vs. 33% vs. 20%), which has been reported previously with influenza.
COVID-19–positive patients, as compared with PUI and controls, were significantly more likely to present with cardiogenic shock before PCI (20% vs. 14% vs. 5%), but not cardiac arrest (12% vs. 17% vs. 11%), and to have lower left ventricular ejection fractions (45% vs. 45% vs. 50%).
They also presented with more atypical symptoms than PUI patients, particularly infiltrates on chest x-ray (49% vs. 17%) and dyspnea (58% vs. 38%). Data were not available for these outcomes among historic controls.
Importantly, 21% of the COVID-19 patients did not undergo angiography, compared with 5% of PUI patients and 0% of controls (P < .001), “which is much higher than we would expect or have suspected,” Dr. Henry said. Thrombolytic use was very uncommon in those undergoing angiography, likely as a result of the guidelines.
Very surprisingly, there were no differences in door-to-balloon times between the COVID-positive, PUI, and control groups despite the ongoing pandemic (80 min vs. 78 min vs. 86 min).
But there was clear worsening in in-hospital mortality in COVID-19–positive patients (32% vs. 12% and 6%; P < .001), as well as in-hospital stroke (3.4% vs. 2% vs. 0.6%) that reached statistical significance only when compared with historical controls (P = .039). Total length of stay was twice as long in COVID-confirmed patients as in both PUI and controls (6 days vs. 3 days; P < .001).
Following the formal presentation, invited discussant Philippe Gabriel Steg, MD, Imperial College London, said the researchers have provided a great service in reporting the data so quickly but noted that an ongoing French registry of events before, during, and after the first COVID-19 wave has not seen an increased death rate.
“Can you tease out whether the increased death rate is related to cardiovascular deaths or to COVID-related pneumonias, shocks, ARDSs [acute respiratory distress syndromes], and so on and so forth? Because our impression – and that’s what we’ve published in Lancet Public Health – is that the cardiovascular morality rate doesn’t seem that affected by COVID.”
Dr. Henry replied that these are early data but “I will tell you that patients who did get PCI had a mortality rate that was only around 12% or 13%, and the patients who did not undergo angiography or were treated with medical therapy had higher mortality. Now, of course, that’s selected and we need to do a much better matching and look at that, but that’s our goal and we will have that information,” he said.
During a press briefing on the study, discussant Renu Virmani, MD, president and founder of CVPath Institute, noted that, in their analysis of 40 autopsy cases from Bergamot, Italy, small intramyocardial microthrombi were seen in nine patients, whereas epicardial microthrombi were seen in only three or four.
“Some of the cases are being taken as being related to coronary disease but may be more thrombotic than anything else,” she said. “I think there’s a combination, and that’s why the outcomes are so poor. You didn’t show us TIMI flow but that’s something to think about: Was TIMI flow different in the patients who died because you have very high mortality? I think we need to get to the bottom of what is the underlying cause of that thrombosis.”
Future topics of interest include ethnic and regional/country differences; time-to-treatment including chest pain onset-to-arrival; transfer, in-hospital, and no-culprit patients; changes over time during the pandemic; and eventually 1-year outcomes, Dr. Henry said.
Press briefing moderator Ajay Kirtane, MD, director of the cardiac catheterization labs at NewYork-Presbyterian/Columbia University Irving, New York, remarked that “a lot of times people will pooh-pooh observational data, but this is exactly the type of data that we need to try to be able to gather information about what our practices are, how they fit. And I think many of us around the world will see these data, and it will echo their own experience.”
The study was funded by the Society for Cardiovascular Angiography and Interventions and the Canadian Association of Interventional Cardiology. Dr. Henry has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Patients with COVID-19 who present with ST-segment elevation MI (STEMI) represent a unique, high-risk population with greater risks for in-hospital death and stroke, according to initial results from the North American COVID-19 ST-Segment Elevation Myocardial Infarction Registry (NACMI).
Although COVID-19–confirmed patients were less likely to undergo angiography than patients under investigation (PUI) for COVID-19 or historical STEMI activation controls, 71% underwent primary percutaneous coronary intervention (PCI).
“Primary PCI is preferable and feasible in COVID-19–positive patients, with door-to-balloon times similar to PUI or COVID-negative patients, and that supports the updated COVID-specific STEMI guidelines,” study cochair Timothy D. Henry, MD, said in a late-breaking clinical science session at TCT 2020, the Transcatheter Cardiovascular Therapeutics virtual annual meeting.
The multisociety COVID-specific guidelines were initially issued in April, endorsing PCI as the standard of care and allowing for consideration of fibrinolysis-based therapy at non-PCI capable hospitals.
Five previous publications on a total of 174 COVID-19 patients with ST-elevation have shown there are more frequent in-hospital STEMI presentations, more cases without a clear culprit lesion, more thrombotic lesions and microthrombi, and higher mortality, ranging from 12% to 72%. Still, there has been considerable controversy over exactly what to do when COVID-19 patients with ST elevation reach the cath lab, he said at the meeting sponsored by the Cardiovascular Research Foundation.
NACMI represents the largest experience with ST-elevation patients and is a unique collaboration between the Society for Cardiovascular Angiography and Interventions, Canadian Association of Interventional Cardiology, American College of Cardiology, and Midwest STEMI Consortium, noted Dr. Henry, who is medical director of the Lindner Center for Research and Education at the Christ Hospital, Cincinnati.
The registry enrolled any COVID-19–positive patient or person under investigation older than 18 years with ST-segment elevation or new-onset left bundle branch block on electrocardiogram with a clinical correlate of myocardial ischemia such as chest pain, dyspnea, cardiac arrest, shock, or mechanical ventilation. There were no exclusion criteria.
Data from 171 patients with confirmed COVID-19 and 423 PUI from 64 sites were then propensity-matched to a control population from the Midwest STEMI Consortium, a prospective, multicenter registry of consecutive STEMI patients.
The three groups were similar in sex and age but there was a striking difference in race, with 27% of African American and 24% of Hispanic patients COVID-confirmed, compared with 11% and 6% in the PUI group and 4% and 1% in the control group. Likewise, there was a significant increase in diabetes (44% vs. 33% vs. 20%), which has been reported previously with influenza.
COVID-19–positive patients, as compared with PUI and controls, were significantly more likely to present with cardiogenic shock before PCI (20% vs. 14% vs. 5%), but not cardiac arrest (12% vs. 17% vs. 11%), and to have lower left ventricular ejection fractions (45% vs. 45% vs. 50%).
They also presented with more atypical symptoms than PUI patients, particularly infiltrates on chest x-ray (49% vs. 17%) and dyspnea (58% vs. 38%). Data were not available for these outcomes among historic controls.
Importantly, 21% of the COVID-19 patients did not undergo angiography, compared with 5% of PUI patients and 0% of controls (P < .001), “which is much higher than we would expect or have suspected,” Dr. Henry said. Thrombolytic use was very uncommon in those undergoing angiography, likely as a result of the guidelines.
Very surprisingly, there were no differences in door-to-balloon times between the COVID-positive, PUI, and control groups despite the ongoing pandemic (80 min vs. 78 min vs. 86 min).
But there was clear worsening in in-hospital mortality in COVID-19–positive patients (32% vs. 12% and 6%; P < .001), as well as in-hospital stroke (3.4% vs. 2% vs. 0.6%) that reached statistical significance only when compared with historical controls (P = .039). Total length of stay was twice as long in COVID-confirmed patients as in both PUI and controls (6 days vs. 3 days; P < .001).
Following the formal presentation, invited discussant Philippe Gabriel Steg, MD, Imperial College London, said the researchers have provided a great service in reporting the data so quickly but noted that an ongoing French registry of events before, during, and after the first COVID-19 wave has not seen an increased death rate.
“Can you tease out whether the increased death rate is related to cardiovascular deaths or to COVID-related pneumonias, shocks, ARDSs [acute respiratory distress syndromes], and so on and so forth? Because our impression – and that’s what we’ve published in Lancet Public Health – is that the cardiovascular morality rate doesn’t seem that affected by COVID.”
Dr. Henry replied that these are early data but “I will tell you that patients who did get PCI had a mortality rate that was only around 12% or 13%, and the patients who did not undergo angiography or were treated with medical therapy had higher mortality. Now, of course, that’s selected and we need to do a much better matching and look at that, but that’s our goal and we will have that information,” he said.
During a press briefing on the study, discussant Renu Virmani, MD, president and founder of CVPath Institute, noted that, in their analysis of 40 autopsy cases from Bergamot, Italy, small intramyocardial microthrombi were seen in nine patients, whereas epicardial microthrombi were seen in only three or four.
“Some of the cases are being taken as being related to coronary disease but may be more thrombotic than anything else,” she said. “I think there’s a combination, and that’s why the outcomes are so poor. You didn’t show us TIMI flow but that’s something to think about: Was TIMI flow different in the patients who died because you have very high mortality? I think we need to get to the bottom of what is the underlying cause of that thrombosis.”
Future topics of interest include ethnic and regional/country differences; time-to-treatment including chest pain onset-to-arrival; transfer, in-hospital, and no-culprit patients; changes over time during the pandemic; and eventually 1-year outcomes, Dr. Henry said.
Press briefing moderator Ajay Kirtane, MD, director of the cardiac catheterization labs at NewYork-Presbyterian/Columbia University Irving, New York, remarked that “a lot of times people will pooh-pooh observational data, but this is exactly the type of data that we need to try to be able to gather information about what our practices are, how they fit. And I think many of us around the world will see these data, and it will echo their own experience.”
The study was funded by the Society for Cardiovascular Angiography and Interventions and the Canadian Association of Interventional Cardiology. Dr. Henry has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
The unsteady state
As the COVID-19 pandemic continues to chug along, some communities feel it slowing to a pace at which they might feel comfortable about a return to, if not quite “business as usual,” at least “business as sort of normal-ish.” They are ready to accept a level of disease that signals they have reached a steady state. However, in other communities, the virus has picked up speed and is threatening to overwhelm the medical infrastructure. If you are in one of those fortunate and skillfully managed states in which folks are beginning to talk seriously, but with little evidence, that it is time to return to normal, it is probably far too early.
Eons ago in pandemic terms, the World Health Organization in Thailand published a list of criteria to aid in determining when a community could consider lifting the limits that seemed to have been effective in halting transmission of the virus (“Transitioning to and maintaining a steady state of low-level or no transmission,” WHO, Thailand, 2020 Apr 18). While much more has been learned about the behavior of the virus since the spring of 2020, the criteria from the WHO in Thailand are worth considering.
Here is my summary of their criteria for returning to normalcy. First, virus transmission is controlled to the point that only sporadic cases and small clusters exist, and that all of these are traceable in origin. Second, health care and public health systems are in place with sufficient capacities to manage a shift from detection to treatment should the case load increase dramatically; this capacity should include detection, testing, isolation, and quarantine. Third, outbreaks in high-risk populations such as nursing homes have been minimized. Fourth, workplace prevention strategies are in place and have been demonstrated to be effective. Fifth, risk of imported cases is at manageable levels. Finally, communities are engaged.
It is hard to argue with the rationale behind each of these criteria. However, the United States is not Thailand, and just thinking about how this country would go about meeting those criteria provides a window into some of the reasons why we have done so poorly and will continue to be challenged in dealing with the pandemic.
First, notice that the criteria make no mention of a vaccine. One gets the sense that from the top down our country is banking too heavily on the effectiveness and widespread delivery of a vaccine. Even if and when a vaccine is developed and delivered, all of these criteria still must be met and kept in mind for a future pandemic.
Second, the criteria call for an effective health care system, but it is abundantly clear that the United States does not have a cohesive health care system and probably won’t for the foreseeable future. The best we can hope for is individual states cobbling together their own systems, which may in turn serve as examples for those states who haven’t had the foresight. We have had a public health system of sorts, but its credibility and effectiveness has been neutered to the point that again we must rely on each state’s ability to see through the haze and create it’s own systems for detection, testing, tracking, isolating, and quarantining – often with little help in materiel support from the federal government. The sliver of good news is that, after a bit of a stumbling start, detecting and limiting the importation of cases from abroad is being addressed.
We continue to hear and see evidence that there are segments of the population who are not engaged in the activities that we have learned are necessary to stabilize the pandemic. My sense is that those people represent a very small minority. But, it is probably large enough to make the route to a steady state on a national level long and painful. This unfortunately is to be expected in a country that was built on a framework of personal freedoms. The best you can hope for in achieving a steady state is to live in one of the states that seems to be achieving the fine balance between personal freedoms and the common good.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at pdnews@mdedge.com.
As the COVID-19 pandemic continues to chug along, some communities feel it slowing to a pace at which they might feel comfortable about a return to, if not quite “business as usual,” at least “business as sort of normal-ish.” They are ready to accept a level of disease that signals they have reached a steady state. However, in other communities, the virus has picked up speed and is threatening to overwhelm the medical infrastructure. If you are in one of those fortunate and skillfully managed states in which folks are beginning to talk seriously, but with little evidence, that it is time to return to normal, it is probably far too early.
Eons ago in pandemic terms, the World Health Organization in Thailand published a list of criteria to aid in determining when a community could consider lifting the limits that seemed to have been effective in halting transmission of the virus (“Transitioning to and maintaining a steady state of low-level or no transmission,” WHO, Thailand, 2020 Apr 18). While much more has been learned about the behavior of the virus since the spring of 2020, the criteria from the WHO in Thailand are worth considering.
Here is my summary of their criteria for returning to normalcy. First, virus transmission is controlled to the point that only sporadic cases and small clusters exist, and that all of these are traceable in origin. Second, health care and public health systems are in place with sufficient capacities to manage a shift from detection to treatment should the case load increase dramatically; this capacity should include detection, testing, isolation, and quarantine. Third, outbreaks in high-risk populations such as nursing homes have been minimized. Fourth, workplace prevention strategies are in place and have been demonstrated to be effective. Fifth, risk of imported cases is at manageable levels. Finally, communities are engaged.
It is hard to argue with the rationale behind each of these criteria. However, the United States is not Thailand, and just thinking about how this country would go about meeting those criteria provides a window into some of the reasons why we have done so poorly and will continue to be challenged in dealing with the pandemic.
First, notice that the criteria make no mention of a vaccine. One gets the sense that from the top down our country is banking too heavily on the effectiveness and widespread delivery of a vaccine. Even if and when a vaccine is developed and delivered, all of these criteria still must be met and kept in mind for a future pandemic.
Second, the criteria call for an effective health care system, but it is abundantly clear that the United States does not have a cohesive health care system and probably won’t for the foreseeable future. The best we can hope for is individual states cobbling together their own systems, which may in turn serve as examples for those states who haven’t had the foresight. We have had a public health system of sorts, but its credibility and effectiveness has been neutered to the point that again we must rely on each state’s ability to see through the haze and create it’s own systems for detection, testing, tracking, isolating, and quarantining – often with little help in materiel support from the federal government. The sliver of good news is that, after a bit of a stumbling start, detecting and limiting the importation of cases from abroad is being addressed.
We continue to hear and see evidence that there are segments of the population who are not engaged in the activities that we have learned are necessary to stabilize the pandemic. My sense is that those people represent a very small minority. But, it is probably large enough to make the route to a steady state on a national level long and painful. This unfortunately is to be expected in a country that was built on a framework of personal freedoms. The best you can hope for in achieving a steady state is to live in one of the states that seems to be achieving the fine balance between personal freedoms and the common good.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at pdnews@mdedge.com.
As the COVID-19 pandemic continues to chug along, some communities feel it slowing to a pace at which they might feel comfortable about a return to, if not quite “business as usual,” at least “business as sort of normal-ish.” They are ready to accept a level of disease that signals they have reached a steady state. However, in other communities, the virus has picked up speed and is threatening to overwhelm the medical infrastructure. If you are in one of those fortunate and skillfully managed states in which folks are beginning to talk seriously, but with little evidence, that it is time to return to normal, it is probably far too early.
Eons ago in pandemic terms, the World Health Organization in Thailand published a list of criteria to aid in determining when a community could consider lifting the limits that seemed to have been effective in halting transmission of the virus (“Transitioning to and maintaining a steady state of low-level or no transmission,” WHO, Thailand, 2020 Apr 18). While much more has been learned about the behavior of the virus since the spring of 2020, the criteria from the WHO in Thailand are worth considering.
Here is my summary of their criteria for returning to normalcy. First, virus transmission is controlled to the point that only sporadic cases and small clusters exist, and that all of these are traceable in origin. Second, health care and public health systems are in place with sufficient capacities to manage a shift from detection to treatment should the case load increase dramatically; this capacity should include detection, testing, isolation, and quarantine. Third, outbreaks in high-risk populations such as nursing homes have been minimized. Fourth, workplace prevention strategies are in place and have been demonstrated to be effective. Fifth, risk of imported cases is at manageable levels. Finally, communities are engaged.
It is hard to argue with the rationale behind each of these criteria. However, the United States is not Thailand, and just thinking about how this country would go about meeting those criteria provides a window into some of the reasons why we have done so poorly and will continue to be challenged in dealing with the pandemic.
First, notice that the criteria make no mention of a vaccine. One gets the sense that from the top down our country is banking too heavily on the effectiveness and widespread delivery of a vaccine. Even if and when a vaccine is developed and delivered, all of these criteria still must be met and kept in mind for a future pandemic.
Second, the criteria call for an effective health care system, but it is abundantly clear that the United States does not have a cohesive health care system and probably won’t for the foreseeable future. The best we can hope for is individual states cobbling together their own systems, which may in turn serve as examples for those states who haven’t had the foresight. We have had a public health system of sorts, but its credibility and effectiveness has been neutered to the point that again we must rely on each state’s ability to see through the haze and create it’s own systems for detection, testing, tracking, isolating, and quarantining – often with little help in materiel support from the federal government. The sliver of good news is that, after a bit of a stumbling start, detecting and limiting the importation of cases from abroad is being addressed.
We continue to hear and see evidence that there are segments of the population who are not engaged in the activities that we have learned are necessary to stabilize the pandemic. My sense is that those people represent a very small minority. But, it is probably large enough to make the route to a steady state on a national level long and painful. This unfortunately is to be expected in a country that was built on a framework of personal freedoms. The best you can hope for in achieving a steady state is to live in one of the states that seems to be achieving the fine balance between personal freedoms and the common good.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at pdnews@mdedge.com.
Diarrhea prevalent among COVID-19 patients with IBD
Diarrhea affected one in every five patients with inflammatory bowel disease (IBD) and COVID-19, compared with only 7%-10% of all patients with COVID-19 in prior studies, researchers reported in Clinical Gastroenterology and Hepatology.
In a systematic review and meta-analysis of 23 studies incorporating data from 449 patients with IBD and COVID-19, their most common symptoms were fever (affecting 48.3% of patients), cough (46.5%), and diarrhea (20.5%), and diarrhea was approximately twice as prevalent as dyspnea, nausea, abdominal pain, and fatigue, wrote Ferdinando D’Amico of Humanitas University in Milan and his associates. “[S]ymptoms experienced by IBD patients with COVID-19 are similar to those occurring in the general population, except for a higher percentage of diarrhea,” they wrote. This increased prevalence might result from IBD itself or from inflammatory effects of viral gut tropism, they noted. “Currently, the diagnostic–therapeutic approach does not differ between IBD and non-IBD patients, but further studies are needed to evaluate whether fecal research of viral RNA and treatment with IBD drugs may play a role in the management of COVID-19 patients.”
To characterize the clinical presentation and course of patients with IBD and COVID-19, the researchers searched PubMed, Embase, Web of Science, and MedRxiv through July 29, 2020, for keywords related to COVID-19, Crohn’s disease, ulcerative colitis, and IBD. They identified 23 studies presenting clinical data from adults or children with a confirmed IBD diagnosis and least one case of COVID-19. Among 243,760 patients with IBD, 1,028 patients had COVID-19 infection, including 509 patients with Crohn’s disease, 428 patients with ulcerative colitis, 49 patients with indeterminate colitis, and 42 patients for whom the IBD subtype was not recorded.
In all, 0.4% of patients with IBD had COVID-19. Nearly all had been diagnosed by polymerase chain reaction of nasopharyngeal swabs, and approximately 40% also had received chest CT scans. Most were male (56.5%), and 43.5% were older than 65 years. Patients were receiving a wide range of IBD therapies, most commonly anti–tumor necrosis factor (TNF) agents, mesalamine, thiopurine (alone or in combination with biologics), vedolizumab, ustekinumab, steroids, methotrexate, and tofacitinib. Results from six studies indicated that patients with IBD were significantly more likely to be diagnosed with COVID-19 if they were older than 66 years (odds ratio, 21.3) or had other comorbidities (OR, 1.24). The most commonly used drugs for managing COVID-19 were hydroxychloroquine, lopinavir/ritonavir, steroids, antibiotics, chloroquine, tofacitinib, and infliximab.
A total of 30.6% of patients with IBD and COVID-19 were hospitalized, 11.4% stayed in the ICU, 3.7% required mechanical ventilation, and 3.8% died from COVID-19. Significant risk factors for death from COVID-19 included older age, active IBD, and a Charlson Comorbidity Index score above 1. Similarly, risk factors for severe COVID-19 included older age, having two or more comorbidities, receiving systemic steroids, and receiving mesalamine/sulfasalazine. In one study, a recent (3-month) history of corticosteroid treatment was associated with a 60% increase in the risk for severe COVID-19. Other immune-mediated therapies did not show this association. Patients with ulcerative colitis were significantly more likely to be seen in the ED or hospitalized, compared with patients with other forms of IBD (adjusted OR, 12.7).
No funding sources were disclosed. Dr. D’Amico reported having no conflicts of interest. Two coinvestigators disclosed ties to AbbVie, MSD, Schering-Plough, UCB Pharma, and several other pharmaceutical companies.
SOURCE: D’Amico F et al. Clin Gastroenterol Hepatol. 2020 Aug 7. doi: 10.1016/j.cgh.2020.08.003.
Diarrhea is one of the hallmark features in inflammatory bowel disease (IBD). The systematic review and meta-analysis by D’Amico and colleagues highlights an increased prevalence of diarrhea in IBD patients with COVID-19. We have learned that SARS-CoV-2 enters the gastrointestinal tract through angiotensin converting enzyme 2, which has been found in absorptive enterocytes of the ileum and colon. The subsequent invasion can cause a change in intestinal microbiota (dysbiosis) and trigger diarrhea. Prior studies also reported SARS-CoV-2 being isolated in the duodenum and rectum while showing RNA shedding in approximately 40% of patients. Clinicians may now face the diagnostic challenge of distinguishing the cause of diarrhea as an exacerbation from underlying IBD versus viral superinfection. The authors astutely hypothesized that having access to fecal polymerase chain reaction tests may be particularly useful to guiding clinical treatment decisions.
Lukasz Kwapisz, MD, FRCPC, is assistant professor of medicine and gastroenterology at Baylor College of Medicine, Houston. He has no conflicts of interest.
Diarrhea is one of the hallmark features in inflammatory bowel disease (IBD). The systematic review and meta-analysis by D’Amico and colleagues highlights an increased prevalence of diarrhea in IBD patients with COVID-19. We have learned that SARS-CoV-2 enters the gastrointestinal tract through angiotensin converting enzyme 2, which has been found in absorptive enterocytes of the ileum and colon. The subsequent invasion can cause a change in intestinal microbiota (dysbiosis) and trigger diarrhea. Prior studies also reported SARS-CoV-2 being isolated in the duodenum and rectum while showing RNA shedding in approximately 40% of patients. Clinicians may now face the diagnostic challenge of distinguishing the cause of diarrhea as an exacerbation from underlying IBD versus viral superinfection. The authors astutely hypothesized that having access to fecal polymerase chain reaction tests may be particularly useful to guiding clinical treatment decisions.
Lukasz Kwapisz, MD, FRCPC, is assistant professor of medicine and gastroenterology at Baylor College of Medicine, Houston. He has no conflicts of interest.
Diarrhea is one of the hallmark features in inflammatory bowel disease (IBD). The systematic review and meta-analysis by D’Amico and colleagues highlights an increased prevalence of diarrhea in IBD patients with COVID-19. We have learned that SARS-CoV-2 enters the gastrointestinal tract through angiotensin converting enzyme 2, which has been found in absorptive enterocytes of the ileum and colon. The subsequent invasion can cause a change in intestinal microbiota (dysbiosis) and trigger diarrhea. Prior studies also reported SARS-CoV-2 being isolated in the duodenum and rectum while showing RNA shedding in approximately 40% of patients. Clinicians may now face the diagnostic challenge of distinguishing the cause of diarrhea as an exacerbation from underlying IBD versus viral superinfection. The authors astutely hypothesized that having access to fecal polymerase chain reaction tests may be particularly useful to guiding clinical treatment decisions.
Lukasz Kwapisz, MD, FRCPC, is assistant professor of medicine and gastroenterology at Baylor College of Medicine, Houston. He has no conflicts of interest.
Diarrhea affected one in every five patients with inflammatory bowel disease (IBD) and COVID-19, compared with only 7%-10% of all patients with COVID-19 in prior studies, researchers reported in Clinical Gastroenterology and Hepatology.
In a systematic review and meta-analysis of 23 studies incorporating data from 449 patients with IBD and COVID-19, their most common symptoms were fever (affecting 48.3% of patients), cough (46.5%), and diarrhea (20.5%), and diarrhea was approximately twice as prevalent as dyspnea, nausea, abdominal pain, and fatigue, wrote Ferdinando D’Amico of Humanitas University in Milan and his associates. “[S]ymptoms experienced by IBD patients with COVID-19 are similar to those occurring in the general population, except for a higher percentage of diarrhea,” they wrote. This increased prevalence might result from IBD itself or from inflammatory effects of viral gut tropism, they noted. “Currently, the diagnostic–therapeutic approach does not differ between IBD and non-IBD patients, but further studies are needed to evaluate whether fecal research of viral RNA and treatment with IBD drugs may play a role in the management of COVID-19 patients.”
To characterize the clinical presentation and course of patients with IBD and COVID-19, the researchers searched PubMed, Embase, Web of Science, and MedRxiv through July 29, 2020, for keywords related to COVID-19, Crohn’s disease, ulcerative colitis, and IBD. They identified 23 studies presenting clinical data from adults or children with a confirmed IBD diagnosis and least one case of COVID-19. Among 243,760 patients with IBD, 1,028 patients had COVID-19 infection, including 509 patients with Crohn’s disease, 428 patients with ulcerative colitis, 49 patients with indeterminate colitis, and 42 patients for whom the IBD subtype was not recorded.
In all, 0.4% of patients with IBD had COVID-19. Nearly all had been diagnosed by polymerase chain reaction of nasopharyngeal swabs, and approximately 40% also had received chest CT scans. Most were male (56.5%), and 43.5% were older than 65 years. Patients were receiving a wide range of IBD therapies, most commonly anti–tumor necrosis factor (TNF) agents, mesalamine, thiopurine (alone or in combination with biologics), vedolizumab, ustekinumab, steroids, methotrexate, and tofacitinib. Results from six studies indicated that patients with IBD were significantly more likely to be diagnosed with COVID-19 if they were older than 66 years (odds ratio, 21.3) or had other comorbidities (OR, 1.24). The most commonly used drugs for managing COVID-19 were hydroxychloroquine, lopinavir/ritonavir, steroids, antibiotics, chloroquine, tofacitinib, and infliximab.
A total of 30.6% of patients with IBD and COVID-19 were hospitalized, 11.4% stayed in the ICU, 3.7% required mechanical ventilation, and 3.8% died from COVID-19. Significant risk factors for death from COVID-19 included older age, active IBD, and a Charlson Comorbidity Index score above 1. Similarly, risk factors for severe COVID-19 included older age, having two or more comorbidities, receiving systemic steroids, and receiving mesalamine/sulfasalazine. In one study, a recent (3-month) history of corticosteroid treatment was associated with a 60% increase in the risk for severe COVID-19. Other immune-mediated therapies did not show this association. Patients with ulcerative colitis were significantly more likely to be seen in the ED or hospitalized, compared with patients with other forms of IBD (adjusted OR, 12.7).
No funding sources were disclosed. Dr. D’Amico reported having no conflicts of interest. Two coinvestigators disclosed ties to AbbVie, MSD, Schering-Plough, UCB Pharma, and several other pharmaceutical companies.
SOURCE: D’Amico F et al. Clin Gastroenterol Hepatol. 2020 Aug 7. doi: 10.1016/j.cgh.2020.08.003.
Diarrhea affected one in every five patients with inflammatory bowel disease (IBD) and COVID-19, compared with only 7%-10% of all patients with COVID-19 in prior studies, researchers reported in Clinical Gastroenterology and Hepatology.
In a systematic review and meta-analysis of 23 studies incorporating data from 449 patients with IBD and COVID-19, their most common symptoms were fever (affecting 48.3% of patients), cough (46.5%), and diarrhea (20.5%), and diarrhea was approximately twice as prevalent as dyspnea, nausea, abdominal pain, and fatigue, wrote Ferdinando D’Amico of Humanitas University in Milan and his associates. “[S]ymptoms experienced by IBD patients with COVID-19 are similar to those occurring in the general population, except for a higher percentage of diarrhea,” they wrote. This increased prevalence might result from IBD itself or from inflammatory effects of viral gut tropism, they noted. “Currently, the diagnostic–therapeutic approach does not differ between IBD and non-IBD patients, but further studies are needed to evaluate whether fecal research of viral RNA and treatment with IBD drugs may play a role in the management of COVID-19 patients.”
To characterize the clinical presentation and course of patients with IBD and COVID-19, the researchers searched PubMed, Embase, Web of Science, and MedRxiv through July 29, 2020, for keywords related to COVID-19, Crohn’s disease, ulcerative colitis, and IBD. They identified 23 studies presenting clinical data from adults or children with a confirmed IBD diagnosis and least one case of COVID-19. Among 243,760 patients with IBD, 1,028 patients had COVID-19 infection, including 509 patients with Crohn’s disease, 428 patients with ulcerative colitis, 49 patients with indeterminate colitis, and 42 patients for whom the IBD subtype was not recorded.
In all, 0.4% of patients with IBD had COVID-19. Nearly all had been diagnosed by polymerase chain reaction of nasopharyngeal swabs, and approximately 40% also had received chest CT scans. Most were male (56.5%), and 43.5% were older than 65 years. Patients were receiving a wide range of IBD therapies, most commonly anti–tumor necrosis factor (TNF) agents, mesalamine, thiopurine (alone or in combination with biologics), vedolizumab, ustekinumab, steroids, methotrexate, and tofacitinib. Results from six studies indicated that patients with IBD were significantly more likely to be diagnosed with COVID-19 if they were older than 66 years (odds ratio, 21.3) or had other comorbidities (OR, 1.24). The most commonly used drugs for managing COVID-19 were hydroxychloroquine, lopinavir/ritonavir, steroids, antibiotics, chloroquine, tofacitinib, and infliximab.
A total of 30.6% of patients with IBD and COVID-19 were hospitalized, 11.4% stayed in the ICU, 3.7% required mechanical ventilation, and 3.8% died from COVID-19. Significant risk factors for death from COVID-19 included older age, active IBD, and a Charlson Comorbidity Index score above 1. Similarly, risk factors for severe COVID-19 included older age, having two or more comorbidities, receiving systemic steroids, and receiving mesalamine/sulfasalazine. In one study, a recent (3-month) history of corticosteroid treatment was associated with a 60% increase in the risk for severe COVID-19. Other immune-mediated therapies did not show this association. Patients with ulcerative colitis were significantly more likely to be seen in the ED or hospitalized, compared with patients with other forms of IBD (adjusted OR, 12.7).
No funding sources were disclosed. Dr. D’Amico reported having no conflicts of interest. Two coinvestigators disclosed ties to AbbVie, MSD, Schering-Plough, UCB Pharma, and several other pharmaceutical companies.
SOURCE: D’Amico F et al. Clin Gastroenterol Hepatol. 2020 Aug 7. doi: 10.1016/j.cgh.2020.08.003.
FROM CLINICAL GASTROENTEROLOGY AND HEPATOLOGY
COVID-19: A second wave of mental illness 'imminent'
The mental health consequences of COVID-19 deaths are likely to overwhelm an already tattered U.S. mental health system, leading to a lack of access, particularly for the most vulnerable, experts warn.
“A second wave of devastation is imminent, attributable to mental health consequences of COVID-19,” write Naomi Simon, MD, and coauthors with the department of psychiatry, New York University.
In a Viewpoint article published in JAMA on Oct. 12, physicians offer some sobering statistics.
Since February 2020, COVID-19 has taken the lives of more than 214,000 Americans. The number of deaths currently attributed to the virus is nearly four times the number of Americans killed during the Vietnam War. The magnitude of death over a short period is a tragedy on a “historic scale,” wrote Dr. Simon and colleagues.
The surge in mental health problems related to COVID-19 deaths will bring further challenges to individuals, families, and communities, including a spike in deaths from suicide and drug overdoses, they warned.
It’s important to consider, they noted, that each COVID-19 death leaves an estimated nine family members bereaved, which is projected to lead to an estimated 2 million bereaved individuals in the United States.
The necessary social distancing and quarantine measures implemented to fight the virus have amplified emotional turmoil and have disrupted the ability of personal support networks and communities to come together and grieve.
“Of central concern is the transformation of normal grief and distress into prolonged grief and major depressive disorder and symptoms of posttraumatic stress disorder,” Simon and colleagues said.
“Once established, these conditions can become chronic with additional comorbidities such as substance use disorders. Prolonged grief affects approximately 10% of bereaved individuals, but this is likely an underestimate for grief related to deaths from COVID-19,” they wrote.
As with the first COVID-19 wave, the mental health wave will disproportionately affect Black persons, Hispanic persons, older adults, persons in lower socioeconomic groups of all races and ethnicities, and healthcare workers, they note.
The psychological risks for health care and other essential workers are of particular concern, they say. “Supporting the mental health of these and other essential workforce is critical to readiness for managing recurrent waves of the pandemic,” they stated.
How will the United States manage this impending wave of mental health problems?
“The solution will require increased funding for mental health; widespread screening to identify individuals at highest risk including suicide risk; availability of primary care clinicians and mental health professionals trained to treat those with prolonged grief, depression, traumatic stress, and substance abuse; and a diligent focus on families and communities to creatively restore the approaches by which they have managed tragedy and loss over generations,” the authors wrote.
“History has shown that societies recover from such devastation when leaders and members are joined by a shared purpose, acting in a unified way to facilitate recovery. In such societies, there is a shared understanding that its members must care for one another because the loss of one is a loss for all. Above all, this shared understanding must be restored,” they concluded.
Dr. Simon has received personal fees from Vanda Pharmaceuticals Inc, MGH Psychiatry Academy, Axovant Sciences, Springworks, Praxis Therapeutics, Aptinyx, Genomind, and Wiley (deputy editor, Depression and Anxiety). Saxe has received royalties from Guilford Press for the book Trauma Systems Therapy for Children and Teens (2016). Marmar serves on the scientific advisory board and owns equity in Receptor Life Sciences and serves on the PTSD advisory board for Otsuka Pharmaceutical.
A version of this article originally appeared on Medscape.com.
The mental health consequences of COVID-19 deaths are likely to overwhelm an already tattered U.S. mental health system, leading to a lack of access, particularly for the most vulnerable, experts warn.
“A second wave of devastation is imminent, attributable to mental health consequences of COVID-19,” write Naomi Simon, MD, and coauthors with the department of psychiatry, New York University.
In a Viewpoint article published in JAMA on Oct. 12, physicians offer some sobering statistics.
Since February 2020, COVID-19 has taken the lives of more than 214,000 Americans. The number of deaths currently attributed to the virus is nearly four times the number of Americans killed during the Vietnam War. The magnitude of death over a short period is a tragedy on a “historic scale,” wrote Dr. Simon and colleagues.
The surge in mental health problems related to COVID-19 deaths will bring further challenges to individuals, families, and communities, including a spike in deaths from suicide and drug overdoses, they warned.
It’s important to consider, they noted, that each COVID-19 death leaves an estimated nine family members bereaved, which is projected to lead to an estimated 2 million bereaved individuals in the United States.
The necessary social distancing and quarantine measures implemented to fight the virus have amplified emotional turmoil and have disrupted the ability of personal support networks and communities to come together and grieve.
“Of central concern is the transformation of normal grief and distress into prolonged grief and major depressive disorder and symptoms of posttraumatic stress disorder,” Simon and colleagues said.
“Once established, these conditions can become chronic with additional comorbidities such as substance use disorders. Prolonged grief affects approximately 10% of bereaved individuals, but this is likely an underestimate for grief related to deaths from COVID-19,” they wrote.
As with the first COVID-19 wave, the mental health wave will disproportionately affect Black persons, Hispanic persons, older adults, persons in lower socioeconomic groups of all races and ethnicities, and healthcare workers, they note.
The psychological risks for health care and other essential workers are of particular concern, they say. “Supporting the mental health of these and other essential workforce is critical to readiness for managing recurrent waves of the pandemic,” they stated.
How will the United States manage this impending wave of mental health problems?
“The solution will require increased funding for mental health; widespread screening to identify individuals at highest risk including suicide risk; availability of primary care clinicians and mental health professionals trained to treat those with prolonged grief, depression, traumatic stress, and substance abuse; and a diligent focus on families and communities to creatively restore the approaches by which they have managed tragedy and loss over generations,” the authors wrote.
“History has shown that societies recover from such devastation when leaders and members are joined by a shared purpose, acting in a unified way to facilitate recovery. In such societies, there is a shared understanding that its members must care for one another because the loss of one is a loss for all. Above all, this shared understanding must be restored,” they concluded.
Dr. Simon has received personal fees from Vanda Pharmaceuticals Inc, MGH Psychiatry Academy, Axovant Sciences, Springworks, Praxis Therapeutics, Aptinyx, Genomind, and Wiley (deputy editor, Depression and Anxiety). Saxe has received royalties from Guilford Press for the book Trauma Systems Therapy for Children and Teens (2016). Marmar serves on the scientific advisory board and owns equity in Receptor Life Sciences and serves on the PTSD advisory board for Otsuka Pharmaceutical.
A version of this article originally appeared on Medscape.com.
The mental health consequences of COVID-19 deaths are likely to overwhelm an already tattered U.S. mental health system, leading to a lack of access, particularly for the most vulnerable, experts warn.
“A second wave of devastation is imminent, attributable to mental health consequences of COVID-19,” write Naomi Simon, MD, and coauthors with the department of psychiatry, New York University.
In a Viewpoint article published in JAMA on Oct. 12, physicians offer some sobering statistics.
Since February 2020, COVID-19 has taken the lives of more than 214,000 Americans. The number of deaths currently attributed to the virus is nearly four times the number of Americans killed during the Vietnam War. The magnitude of death over a short period is a tragedy on a “historic scale,” wrote Dr. Simon and colleagues.
The surge in mental health problems related to COVID-19 deaths will bring further challenges to individuals, families, and communities, including a spike in deaths from suicide and drug overdoses, they warned.
It’s important to consider, they noted, that each COVID-19 death leaves an estimated nine family members bereaved, which is projected to lead to an estimated 2 million bereaved individuals in the United States.
The necessary social distancing and quarantine measures implemented to fight the virus have amplified emotional turmoil and have disrupted the ability of personal support networks and communities to come together and grieve.
“Of central concern is the transformation of normal grief and distress into prolonged grief and major depressive disorder and symptoms of posttraumatic stress disorder,” Simon and colleagues said.
“Once established, these conditions can become chronic with additional comorbidities such as substance use disorders. Prolonged grief affects approximately 10% of bereaved individuals, but this is likely an underestimate for grief related to deaths from COVID-19,” they wrote.
As with the first COVID-19 wave, the mental health wave will disproportionately affect Black persons, Hispanic persons, older adults, persons in lower socioeconomic groups of all races and ethnicities, and healthcare workers, they note.
The psychological risks for health care and other essential workers are of particular concern, they say. “Supporting the mental health of these and other essential workforce is critical to readiness for managing recurrent waves of the pandemic,” they stated.
How will the United States manage this impending wave of mental health problems?
“The solution will require increased funding for mental health; widespread screening to identify individuals at highest risk including suicide risk; availability of primary care clinicians and mental health professionals trained to treat those with prolonged grief, depression, traumatic stress, and substance abuse; and a diligent focus on families and communities to creatively restore the approaches by which they have managed tragedy and loss over generations,” the authors wrote.
“History has shown that societies recover from such devastation when leaders and members are joined by a shared purpose, acting in a unified way to facilitate recovery. In such societies, there is a shared understanding that its members must care for one another because the loss of one is a loss for all. Above all, this shared understanding must be restored,” they concluded.
Dr. Simon has received personal fees from Vanda Pharmaceuticals Inc, MGH Psychiatry Academy, Axovant Sciences, Springworks, Praxis Therapeutics, Aptinyx, Genomind, and Wiley (deputy editor, Depression and Anxiety). Saxe has received royalties from Guilford Press for the book Trauma Systems Therapy for Children and Teens (2016). Marmar serves on the scientific advisory board and owns equity in Receptor Life Sciences and serves on the PTSD advisory board for Otsuka Pharmaceutical.
A version of this article originally appeared on Medscape.com.
Blood group O linked to decreased risk of SARS-CoV-2 infection
Blood group O was associated with a decreased risk for contracting SARS-CoV-2 infection, according to the results of large retrospective analysis of the Danish population.
Researchers Mike Bogetofte Barnkob, MD, of the Department of Clinical Immunology, Odense (Denmark) University Hospital, and colleagues performed a retrospective cohort analysis of all Danish individuals with a known ABO blood group who were tested for SARS-CoV-2 between Feb. 27, 2020, and July 30, 2020.
Of the 841,327 people tested, ABO and RhD blood groups could be identified for 473,654 individuals. ABO and RhD data from 2,204,742 (38% of the entire Danish population) were used as a reference, according to the online report in Blood Advances.
The primary outcome was status of ABO and RhD blood groups and test results for SARS-CoV-2. The secondary outcomes followed were hospitalization and death from COVID-19.
Reduced prevalence
The study found that ABO blood groups varied significantly between patients and the reference group, with only 38.41% (95% confidence interval, 37.30%-39.50%) of the patients belonging to blood group O, compared with 41.70% (95% CI, 41.60%-41.80%) in the controls, corresponding to a relative risk of 0.87 (95% CI, 0.83-0.91) for acquiring COVID-19.
There was a slight, but statistically significant, difference in blood group distribution between the SARS-CoV-22 individuals and the reference population (P < .001), according to the authors.
Among the SARS-CoV-2 individuals, fewer group O individuals were found (P < .001); while more A, B, and AB individuals were seen (P < .001, P = .011, and P = .091, respectively). There was no significant difference seen among A, B, and AB blood groups (P = .30). The RR for contracting SARS-CoV-2 were 1.09 (95% CI, 1.04-1.14) for A group individuals; 1.06 (95% CI, 0.99-1.14) for B group; and 1.15 (95% CI, 1.03-1.27) for AB group, respectively.
There was no difference found in the RhD group between positive test cases and the reference population (P = .15). In addition, there was no statistical difference (all P > .40) between ABO blood groups and clinical severity of COVID-19 for nonhospitalized patients versus hospitalized patients or for deceased patients versus living patients, the researchers added.
Possible causes
The authors speculated on two possible causes of the lower prevalence of SARS-CoV-2 infection in the blood group O population. The first is that anti-A and anti-B antibodies may have an effect on neutralizing SARS-CoV viruses and that anti-A and anti-B are present on mucosal surfaces in some individuals lacking the corresponding ABO blood group. The second is that the association between ABO blood groups and levels of von Willebrand factor, which is higher in non-O individuals and is tied to an increased likelihood of arterial and venous thrombosis, could have an indirect or unknown impact on susceptibility to infection, according to the authors.
“Given the known increased risk of thrombosis in non-O individuals and the evolving central role for thrombosis in the pathogenesis of COVID-19, it is important to explore this aspect more closely in larger patient cohorts (e.g., by examining ABO blood type and viral load, the severity of symptoms, and the long-term effects following COVID-19),” the researchers concluded.
One author reported receiving fees from Bristol Myers Squibb, Novartis, and Roche. The remaining authors reported they had no competing financial interests.
SOURCE: Barnkob MB et al. Blood Adv. 2020 Oct 14. doi: 10.1182/bloodadvances.2020002657.
Blood group O was associated with a decreased risk for contracting SARS-CoV-2 infection, according to the results of large retrospective analysis of the Danish population.
Researchers Mike Bogetofte Barnkob, MD, of the Department of Clinical Immunology, Odense (Denmark) University Hospital, and colleagues performed a retrospective cohort analysis of all Danish individuals with a known ABO blood group who were tested for SARS-CoV-2 between Feb. 27, 2020, and July 30, 2020.
Of the 841,327 people tested, ABO and RhD blood groups could be identified for 473,654 individuals. ABO and RhD data from 2,204,742 (38% of the entire Danish population) were used as a reference, according to the online report in Blood Advances.
The primary outcome was status of ABO and RhD blood groups and test results for SARS-CoV-2. The secondary outcomes followed were hospitalization and death from COVID-19.
Reduced prevalence
The study found that ABO blood groups varied significantly between patients and the reference group, with only 38.41% (95% confidence interval, 37.30%-39.50%) of the patients belonging to blood group O, compared with 41.70% (95% CI, 41.60%-41.80%) in the controls, corresponding to a relative risk of 0.87 (95% CI, 0.83-0.91) for acquiring COVID-19.
There was a slight, but statistically significant, difference in blood group distribution between the SARS-CoV-22 individuals and the reference population (P < .001), according to the authors.
Among the SARS-CoV-2 individuals, fewer group O individuals were found (P < .001); while more A, B, and AB individuals were seen (P < .001, P = .011, and P = .091, respectively). There was no significant difference seen among A, B, and AB blood groups (P = .30). The RR for contracting SARS-CoV-2 were 1.09 (95% CI, 1.04-1.14) for A group individuals; 1.06 (95% CI, 0.99-1.14) for B group; and 1.15 (95% CI, 1.03-1.27) for AB group, respectively.
There was no difference found in the RhD group between positive test cases and the reference population (P = .15). In addition, there was no statistical difference (all P > .40) between ABO blood groups and clinical severity of COVID-19 for nonhospitalized patients versus hospitalized patients or for deceased patients versus living patients, the researchers added.
Possible causes
The authors speculated on two possible causes of the lower prevalence of SARS-CoV-2 infection in the blood group O population. The first is that anti-A and anti-B antibodies may have an effect on neutralizing SARS-CoV viruses and that anti-A and anti-B are present on mucosal surfaces in some individuals lacking the corresponding ABO blood group. The second is that the association between ABO blood groups and levels of von Willebrand factor, which is higher in non-O individuals and is tied to an increased likelihood of arterial and venous thrombosis, could have an indirect or unknown impact on susceptibility to infection, according to the authors.
“Given the known increased risk of thrombosis in non-O individuals and the evolving central role for thrombosis in the pathogenesis of COVID-19, it is important to explore this aspect more closely in larger patient cohorts (e.g., by examining ABO blood type and viral load, the severity of symptoms, and the long-term effects following COVID-19),” the researchers concluded.
One author reported receiving fees from Bristol Myers Squibb, Novartis, and Roche. The remaining authors reported they had no competing financial interests.
SOURCE: Barnkob MB et al. Blood Adv. 2020 Oct 14. doi: 10.1182/bloodadvances.2020002657.
Blood group O was associated with a decreased risk for contracting SARS-CoV-2 infection, according to the results of large retrospective analysis of the Danish population.
Researchers Mike Bogetofte Barnkob, MD, of the Department of Clinical Immunology, Odense (Denmark) University Hospital, and colleagues performed a retrospective cohort analysis of all Danish individuals with a known ABO blood group who were tested for SARS-CoV-2 between Feb. 27, 2020, and July 30, 2020.
Of the 841,327 people tested, ABO and RhD blood groups could be identified for 473,654 individuals. ABO and RhD data from 2,204,742 (38% of the entire Danish population) were used as a reference, according to the online report in Blood Advances.
The primary outcome was status of ABO and RhD blood groups and test results for SARS-CoV-2. The secondary outcomes followed were hospitalization and death from COVID-19.
Reduced prevalence
The study found that ABO blood groups varied significantly between patients and the reference group, with only 38.41% (95% confidence interval, 37.30%-39.50%) of the patients belonging to blood group O, compared with 41.70% (95% CI, 41.60%-41.80%) in the controls, corresponding to a relative risk of 0.87 (95% CI, 0.83-0.91) for acquiring COVID-19.
There was a slight, but statistically significant, difference in blood group distribution between the SARS-CoV-22 individuals and the reference population (P < .001), according to the authors.
Among the SARS-CoV-2 individuals, fewer group O individuals were found (P < .001); while more A, B, and AB individuals were seen (P < .001, P = .011, and P = .091, respectively). There was no significant difference seen among A, B, and AB blood groups (P = .30). The RR for contracting SARS-CoV-2 were 1.09 (95% CI, 1.04-1.14) for A group individuals; 1.06 (95% CI, 0.99-1.14) for B group; and 1.15 (95% CI, 1.03-1.27) for AB group, respectively.
There was no difference found in the RhD group between positive test cases and the reference population (P = .15). In addition, there was no statistical difference (all P > .40) between ABO blood groups and clinical severity of COVID-19 for nonhospitalized patients versus hospitalized patients or for deceased patients versus living patients, the researchers added.
Possible causes
The authors speculated on two possible causes of the lower prevalence of SARS-CoV-2 infection in the blood group O population. The first is that anti-A and anti-B antibodies may have an effect on neutralizing SARS-CoV viruses and that anti-A and anti-B are present on mucosal surfaces in some individuals lacking the corresponding ABO blood group. The second is that the association between ABO blood groups and levels of von Willebrand factor, which is higher in non-O individuals and is tied to an increased likelihood of arterial and venous thrombosis, could have an indirect or unknown impact on susceptibility to infection, according to the authors.
“Given the known increased risk of thrombosis in non-O individuals and the evolving central role for thrombosis in the pathogenesis of COVID-19, it is important to explore this aspect more closely in larger patient cohorts (e.g., by examining ABO blood type and viral load, the severity of symptoms, and the long-term effects following COVID-19),” the researchers concluded.
One author reported receiving fees from Bristol Myers Squibb, Novartis, and Roche. The remaining authors reported they had no competing financial interests.
SOURCE: Barnkob MB et al. Blood Adv. 2020 Oct 14. doi: 10.1182/bloodadvances.2020002657.
FROM BLOOD ADVANCES
Being HIV positive increases risk of death from COVID-19
compared with people without HIV.
A comparison of outcomes of people with HIV to people without HIV who were hospitalized in the United Kingdom with COVID-19 from Jan. 17 to June 4 showed that HIV-positive status was associated with a 63% increased risk of day 28 mortality.
This was especially true for HIV+ patients younger than 70 years of age, said Anna Maria Geretti, MD, PhD, professor of virology and infectious diseases, University of Liverpool, England.
The results are from an analysis of data from the ISARIC World Health Organization (WHO) Clinical Characterisation Protocol (UK) study, and were presented at the HIV Glasgow annual meeting, held virtually this year because of the pandemic.
“We investigated whether HIV status could be important in COVID-19 outcomes because there was anxiety on the part of our patients, and we wanted to gather some evidence-based information in order to help guide them,” Dr. Geretti said in an interview.
“ISARIC is an international protocol and the UK is one of the nations participating. We applied for access to its very large database, which connects data from all patients who are hospitalized with either known or suspected COVID-19. We wanted to see specifically how the presentation and outcomes of patients with HIV compared with the rest of the population without HIV. It afforded us an ideal opportunity to start to answer this question, and this is our first analysis in what will be an ongoing process. Importantly, we showed that there is a need to really look more carefully at the population with HIV,” she said.
Out of a total of 47,539 patients in the database, 115 (0.24%) had confirmed HIV-positive status, and 103 of those 115, or 89.6%, had a record of being on antiretroviral therapy.
On admission, the patients with HIV were younger, with a median age of 55 compared with 74 for patients without HIV (P < .001). They also had a higher prevalence of obesity, moderate to severe liver disease, higher lymphocyte counts and C-reactive protein, as well as more systemic symptoms.
There were no differences in respiratory rate, need for oxygen, or prevalence of chest infiltrates.
The cumulative incidence of mortality at day 28 was 25.2% in HIV-positive patients compared with 32.1% in HIV-negative patients (P = .12).
But when the researchers looked more closely, they noticed that the mortality rate was actually higher in younger HIV+ patients compared with HIV-negative patients.
Stratified by age, 28-day mortality was significantly higher in HIV+ patients aged <50 years (P =.004); and those aged 50 to 59 years (P = .05).
“So below the age of 70, the risk of mortality was double in people with HIV. The people with HIV who died often had diabetes with complications and also more frequent obesity, but this was not the only explanation,” Dr. Geretti said. “There is something to do with the HIV status per se.”
Next steps will be to expand the data set and repeat the analysis with an additional 100 patients “at least” she said.
The researchers also hope to zero in on what about being HIV+ is increasing the mortality risk from COVID-19.
“Right now we need greater numbers and we hope that the research community will be stimulated to take a closer look at this information, and merge other data so that we can strengthen confidence in the data and tease out what factors are causing this increased risk for mortality,” Dr. Geretti said.
She also emphasized that all patients admitted to hospital with COVID-19 should be asked about their HIV status.
“It is important that the HIV status be recorded if we want to increase our ability to understand how HIV impacts survival,” she stressed. “In our experience we found that most of the hospital records were not doing that. Since HIV+ patients seem to be at increased risk, HIV status should be factored into the clinical management. Ask patients if they are HIV+, and if it is not known, then do a test. That would be good practice.”
Dr. Geretti reported no relevant financial relationships. The work was supported by grants from the National Institute of Health Research, the Medical Research Council, the Wellcome Trust, the Department for International Development, and the Bill and Melinda Gates Foundation.
A version of this article originally appeared on Medscape.com.
compared with people without HIV.
A comparison of outcomes of people with HIV to people without HIV who were hospitalized in the United Kingdom with COVID-19 from Jan. 17 to June 4 showed that HIV-positive status was associated with a 63% increased risk of day 28 mortality.
This was especially true for HIV+ patients younger than 70 years of age, said Anna Maria Geretti, MD, PhD, professor of virology and infectious diseases, University of Liverpool, England.
The results are from an analysis of data from the ISARIC World Health Organization (WHO) Clinical Characterisation Protocol (UK) study, and were presented at the HIV Glasgow annual meeting, held virtually this year because of the pandemic.
“We investigated whether HIV status could be important in COVID-19 outcomes because there was anxiety on the part of our patients, and we wanted to gather some evidence-based information in order to help guide them,” Dr. Geretti said in an interview.
“ISARIC is an international protocol and the UK is one of the nations participating. We applied for access to its very large database, which connects data from all patients who are hospitalized with either known or suspected COVID-19. We wanted to see specifically how the presentation and outcomes of patients with HIV compared with the rest of the population without HIV. It afforded us an ideal opportunity to start to answer this question, and this is our first analysis in what will be an ongoing process. Importantly, we showed that there is a need to really look more carefully at the population with HIV,” she said.
Out of a total of 47,539 patients in the database, 115 (0.24%) had confirmed HIV-positive status, and 103 of those 115, or 89.6%, had a record of being on antiretroviral therapy.
On admission, the patients with HIV were younger, with a median age of 55 compared with 74 for patients without HIV (P < .001). They also had a higher prevalence of obesity, moderate to severe liver disease, higher lymphocyte counts and C-reactive protein, as well as more systemic symptoms.
There were no differences in respiratory rate, need for oxygen, or prevalence of chest infiltrates.
The cumulative incidence of mortality at day 28 was 25.2% in HIV-positive patients compared with 32.1% in HIV-negative patients (P = .12).
But when the researchers looked more closely, they noticed that the mortality rate was actually higher in younger HIV+ patients compared with HIV-negative patients.
Stratified by age, 28-day mortality was significantly higher in HIV+ patients aged <50 years (P =.004); and those aged 50 to 59 years (P = .05).
“So below the age of 70, the risk of mortality was double in people with HIV. The people with HIV who died often had diabetes with complications and also more frequent obesity, but this was not the only explanation,” Dr. Geretti said. “There is something to do with the HIV status per se.”
Next steps will be to expand the data set and repeat the analysis with an additional 100 patients “at least” she said.
The researchers also hope to zero in on what about being HIV+ is increasing the mortality risk from COVID-19.
“Right now we need greater numbers and we hope that the research community will be stimulated to take a closer look at this information, and merge other data so that we can strengthen confidence in the data and tease out what factors are causing this increased risk for mortality,” Dr. Geretti said.
She also emphasized that all patients admitted to hospital with COVID-19 should be asked about their HIV status.
“It is important that the HIV status be recorded if we want to increase our ability to understand how HIV impacts survival,” she stressed. “In our experience we found that most of the hospital records were not doing that. Since HIV+ patients seem to be at increased risk, HIV status should be factored into the clinical management. Ask patients if they are HIV+, and if it is not known, then do a test. That would be good practice.”
Dr. Geretti reported no relevant financial relationships. The work was supported by grants from the National Institute of Health Research, the Medical Research Council, the Wellcome Trust, the Department for International Development, and the Bill and Melinda Gates Foundation.
A version of this article originally appeared on Medscape.com.
compared with people without HIV.
A comparison of outcomes of people with HIV to people without HIV who were hospitalized in the United Kingdom with COVID-19 from Jan. 17 to June 4 showed that HIV-positive status was associated with a 63% increased risk of day 28 mortality.
This was especially true for HIV+ patients younger than 70 years of age, said Anna Maria Geretti, MD, PhD, professor of virology and infectious diseases, University of Liverpool, England.
The results are from an analysis of data from the ISARIC World Health Organization (WHO) Clinical Characterisation Protocol (UK) study, and were presented at the HIV Glasgow annual meeting, held virtually this year because of the pandemic.
“We investigated whether HIV status could be important in COVID-19 outcomes because there was anxiety on the part of our patients, and we wanted to gather some evidence-based information in order to help guide them,” Dr. Geretti said in an interview.
“ISARIC is an international protocol and the UK is one of the nations participating. We applied for access to its very large database, which connects data from all patients who are hospitalized with either known or suspected COVID-19. We wanted to see specifically how the presentation and outcomes of patients with HIV compared with the rest of the population without HIV. It afforded us an ideal opportunity to start to answer this question, and this is our first analysis in what will be an ongoing process. Importantly, we showed that there is a need to really look more carefully at the population with HIV,” she said.
Out of a total of 47,539 patients in the database, 115 (0.24%) had confirmed HIV-positive status, and 103 of those 115, or 89.6%, had a record of being on antiretroviral therapy.
On admission, the patients with HIV were younger, with a median age of 55 compared with 74 for patients without HIV (P < .001). They also had a higher prevalence of obesity, moderate to severe liver disease, higher lymphocyte counts and C-reactive protein, as well as more systemic symptoms.
There were no differences in respiratory rate, need for oxygen, or prevalence of chest infiltrates.
The cumulative incidence of mortality at day 28 was 25.2% in HIV-positive patients compared with 32.1% in HIV-negative patients (P = .12).
But when the researchers looked more closely, they noticed that the mortality rate was actually higher in younger HIV+ patients compared with HIV-negative patients.
Stratified by age, 28-day mortality was significantly higher in HIV+ patients aged <50 years (P =.004); and those aged 50 to 59 years (P = .05).
“So below the age of 70, the risk of mortality was double in people with HIV. The people with HIV who died often had diabetes with complications and also more frequent obesity, but this was not the only explanation,” Dr. Geretti said. “There is something to do with the HIV status per se.”
Next steps will be to expand the data set and repeat the analysis with an additional 100 patients “at least” she said.
The researchers also hope to zero in on what about being HIV+ is increasing the mortality risk from COVID-19.
“Right now we need greater numbers and we hope that the research community will be stimulated to take a closer look at this information, and merge other data so that we can strengthen confidence in the data and tease out what factors are causing this increased risk for mortality,” Dr. Geretti said.
She also emphasized that all patients admitted to hospital with COVID-19 should be asked about their HIV status.
“It is important that the HIV status be recorded if we want to increase our ability to understand how HIV impacts survival,” she stressed. “In our experience we found that most of the hospital records were not doing that. Since HIV+ patients seem to be at increased risk, HIV status should be factored into the clinical management. Ask patients if they are HIV+, and if it is not known, then do a test. That would be good practice.”
Dr. Geretti reported no relevant financial relationships. The work was supported by grants from the National Institute of Health Research, the Medical Research Council, the Wellcome Trust, the Department for International Development, and the Bill and Melinda Gates Foundation.
A version of this article originally appeared on Medscape.com.
Delayed cancer screening could cause increase in deaths, study says
Delays in colorectal cancer screening due to the COVID-19 pandemic could lead to higher rates of advanced-stage cancer and death, according to a new study.
When compared with a delay of less than three months, the longer delay seen this year may result in an 11.9% increase in death rates.
“Across the globe, health care systems are facing serious difficulties while dealing with COVID-19, and it is imperative that support is given to the public and patients throughout the crisis, including for high-impact diseases such as colorectal cancer,” Luigi Ricciardiello, the lead study author and a professor at the University of Bologna in Italy, said in a statement.
Ricciardiello and colleagues presented their research on Monday at UEG Week Virtual 2020, an international conference for gastroenterologists. The study will be published in the UEG Journal .
The researchers created a model to forecast the effects of delayed cancer screening during 2020. A “moderate” delay of 7-12 months caused a 3% increase in advanced-stage colon cancer, and a long delay of more than 12 months caused a 7% increase in advanced cancer.
Based on a survival rate of 5 years for stage 3 or stage 4 colorectal cancer, the death rate would increase nearly 12% when screening is delayed for more than a year, as compared with less than three months of delay.
The research team found similar results when forecasting advanced-stage cancer and deaths earlier this year. In a paper published in Clinical Gastroenterology and Hepatology in early September, they projected that deaths could increase 12% if screening is delayed for more than a year.
Throughout the pandemic, screening programs have been delayed in many countries, particularly across Europe.
“Healthcare authorities need to act urgently on how they reorganise activities during COVID-19, without compromising the diagnosis of other high-impact diseases like this research shows,” Ricciardiello said.
United European Gastroenterology, a professional medical organization for digestive health specialists, has called for policymakers to implement colon cancer screening programs across the European Union. Annually, more than 375,000 new cases are diagnosed across the EU, and more than 170,000 people die from colorectal cancer, according to a UEG report.
“Early-stage diagnosis of colorectal cancer is crucial — it’s far easier to treat and enhances optimal patient outcomes,” Ricciardiello said. “It is therefore essential that vital diagnosis tools, like screening programmes, continue and help to prevent mortality rates from rising even further.”
This article first appeared on Medscape.com.
Delays in colorectal cancer screening due to the COVID-19 pandemic could lead to higher rates of advanced-stage cancer and death, according to a new study.
When compared with a delay of less than three months, the longer delay seen this year may result in an 11.9% increase in death rates.
“Across the globe, health care systems are facing serious difficulties while dealing with COVID-19, and it is imperative that support is given to the public and patients throughout the crisis, including for high-impact diseases such as colorectal cancer,” Luigi Ricciardiello, the lead study author and a professor at the University of Bologna in Italy, said in a statement.
Ricciardiello and colleagues presented their research on Monday at UEG Week Virtual 2020, an international conference for gastroenterologists. The study will be published in the UEG Journal .
The researchers created a model to forecast the effects of delayed cancer screening during 2020. A “moderate” delay of 7-12 months caused a 3% increase in advanced-stage colon cancer, and a long delay of more than 12 months caused a 7% increase in advanced cancer.
Based on a survival rate of 5 years for stage 3 or stage 4 colorectal cancer, the death rate would increase nearly 12% when screening is delayed for more than a year, as compared with less than three months of delay.
The research team found similar results when forecasting advanced-stage cancer and deaths earlier this year. In a paper published in Clinical Gastroenterology and Hepatology in early September, they projected that deaths could increase 12% if screening is delayed for more than a year.
Throughout the pandemic, screening programs have been delayed in many countries, particularly across Europe.
“Healthcare authorities need to act urgently on how they reorganise activities during COVID-19, without compromising the diagnosis of other high-impact diseases like this research shows,” Ricciardiello said.
United European Gastroenterology, a professional medical organization for digestive health specialists, has called for policymakers to implement colon cancer screening programs across the European Union. Annually, more than 375,000 new cases are diagnosed across the EU, and more than 170,000 people die from colorectal cancer, according to a UEG report.
“Early-stage diagnosis of colorectal cancer is crucial — it’s far easier to treat and enhances optimal patient outcomes,” Ricciardiello said. “It is therefore essential that vital diagnosis tools, like screening programmes, continue and help to prevent mortality rates from rising even further.”
This article first appeared on Medscape.com.
Delays in colorectal cancer screening due to the COVID-19 pandemic could lead to higher rates of advanced-stage cancer and death, according to a new study.
When compared with a delay of less than three months, the longer delay seen this year may result in an 11.9% increase in death rates.
“Across the globe, health care systems are facing serious difficulties while dealing with COVID-19, and it is imperative that support is given to the public and patients throughout the crisis, including for high-impact diseases such as colorectal cancer,” Luigi Ricciardiello, the lead study author and a professor at the University of Bologna in Italy, said in a statement.
Ricciardiello and colleagues presented their research on Monday at UEG Week Virtual 2020, an international conference for gastroenterologists. The study will be published in the UEG Journal .
The researchers created a model to forecast the effects of delayed cancer screening during 2020. A “moderate” delay of 7-12 months caused a 3% increase in advanced-stage colon cancer, and a long delay of more than 12 months caused a 7% increase in advanced cancer.
Based on a survival rate of 5 years for stage 3 or stage 4 colorectal cancer, the death rate would increase nearly 12% when screening is delayed for more than a year, as compared with less than three months of delay.
The research team found similar results when forecasting advanced-stage cancer and deaths earlier this year. In a paper published in Clinical Gastroenterology and Hepatology in early September, they projected that deaths could increase 12% if screening is delayed for more than a year.
Throughout the pandemic, screening programs have been delayed in many countries, particularly across Europe.
“Healthcare authorities need to act urgently on how they reorganise activities during COVID-19, without compromising the diagnosis of other high-impact diseases like this research shows,” Ricciardiello said.
United European Gastroenterology, a professional medical organization for digestive health specialists, has called for policymakers to implement colon cancer screening programs across the European Union. Annually, more than 375,000 new cases are diagnosed across the EU, and more than 170,000 people die from colorectal cancer, according to a UEG report.
“Early-stage diagnosis of colorectal cancer is crucial — it’s far easier to treat and enhances optimal patient outcomes,” Ricciardiello said. “It is therefore essential that vital diagnosis tools, like screening programmes, continue and help to prevent mortality rates from rising even further.”
This article first appeared on Medscape.com.
What’s in a number? 697,633 children with COVID-19
according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.
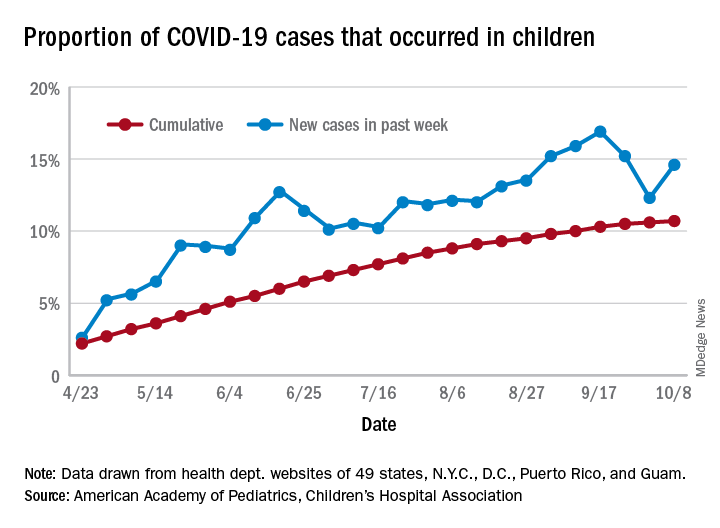
For the week, 14.6% of all COVID-19 cases reported in the United States occurred in children, after 2 consecutive weeks of declines that saw the proportion drop from 16.9% to 12.3%. The cumulative rate of child cases for the entire pandemic is 10.7%, with total child cases in the United States now up to 697,633 and cases among all ages at just over 6.5 million, the AAP and the CHA said Oct. 12 in their weekly COVID-19 report.
Nationally, there were 927 cases reported per 100,000 children as of Oct. 8, with rates at the state level varying from 176 per 100,000 in Vermont to 2,221 per 100,000 in North Dakota. Two other states were over 2,000 cases per 100,000 children: Tennessee (2,155) and South Carolina (2,116), based on data from the health departments of 49 states (New York does not report age distribution), as well as the District of Columbia, New York City, Puerto Rico, and Guam.
Severe illness continues to be rare in children, and national (25 states and New York City) hospitalization rates dropped in the last week. The proportion of hospitalizations occurring in children slipped from a pandemic high of 1.8% the previous week to 1.7% during the week of Oct. 8, and the rate of hospitalizations for children with COVID-19 was down to 1.4% from 1.6% the week before and 1.9% on Sept. 3, the AAP and the CHA said.
Mortality data from 42 states and New York City also show a decline. For the third consecutive week, children represented just 0.06% of all COVID-19 deaths in the United States, down from a high of 0.07% on Sept. 17. Only 0.02% of all cases in children have resulted in death, and that figure has been dropping since early June, when it reached 0.06%, according to the AAP/CHA report. As of Oct. 8, there have been 115 total deaths reported in children.
according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.

For the week, 14.6% of all COVID-19 cases reported in the United States occurred in children, after 2 consecutive weeks of declines that saw the proportion drop from 16.9% to 12.3%. The cumulative rate of child cases for the entire pandemic is 10.7%, with total child cases in the United States now up to 697,633 and cases among all ages at just over 6.5 million, the AAP and the CHA said Oct. 12 in their weekly COVID-19 report.
Nationally, there were 927 cases reported per 100,000 children as of Oct. 8, with rates at the state level varying from 176 per 100,000 in Vermont to 2,221 per 100,000 in North Dakota. Two other states were over 2,000 cases per 100,000 children: Tennessee (2,155) and South Carolina (2,116), based on data from the health departments of 49 states (New York does not report age distribution), as well as the District of Columbia, New York City, Puerto Rico, and Guam.
Severe illness continues to be rare in children, and national (25 states and New York City) hospitalization rates dropped in the last week. The proportion of hospitalizations occurring in children slipped from a pandemic high of 1.8% the previous week to 1.7% during the week of Oct. 8, and the rate of hospitalizations for children with COVID-19 was down to 1.4% from 1.6% the week before and 1.9% on Sept. 3, the AAP and the CHA said.
Mortality data from 42 states and New York City also show a decline. For the third consecutive week, children represented just 0.06% of all COVID-19 deaths in the United States, down from a high of 0.07% on Sept. 17. Only 0.02% of all cases in children have resulted in death, and that figure has been dropping since early June, when it reached 0.06%, according to the AAP/CHA report. As of Oct. 8, there have been 115 total deaths reported in children.
according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.

For the week, 14.6% of all COVID-19 cases reported in the United States occurred in children, after 2 consecutive weeks of declines that saw the proportion drop from 16.9% to 12.3%. The cumulative rate of child cases for the entire pandemic is 10.7%, with total child cases in the United States now up to 697,633 and cases among all ages at just over 6.5 million, the AAP and the CHA said Oct. 12 in their weekly COVID-19 report.
Nationally, there were 927 cases reported per 100,000 children as of Oct. 8, with rates at the state level varying from 176 per 100,000 in Vermont to 2,221 per 100,000 in North Dakota. Two other states were over 2,000 cases per 100,000 children: Tennessee (2,155) and South Carolina (2,116), based on data from the health departments of 49 states (New York does not report age distribution), as well as the District of Columbia, New York City, Puerto Rico, and Guam.
Severe illness continues to be rare in children, and national (25 states and New York City) hospitalization rates dropped in the last week. The proportion of hospitalizations occurring in children slipped from a pandemic high of 1.8% the previous week to 1.7% during the week of Oct. 8, and the rate of hospitalizations for children with COVID-19 was down to 1.4% from 1.6% the week before and 1.9% on Sept. 3, the AAP and the CHA said.
Mortality data from 42 states and New York City also show a decline. For the third consecutive week, children represented just 0.06% of all COVID-19 deaths in the United States, down from a high of 0.07% on Sept. 17. Only 0.02% of all cases in children have resulted in death, and that figure has been dropping since early June, when it reached 0.06%, according to the AAP/CHA report. As of Oct. 8, there have been 115 total deaths reported in children.
COVID-19 pandemic amplifies uncertainty for immigrant hospitalists
H1-B visa program needs improvement
Statistics tell the tale of immigrants in the American health care workforce in broad strokes. In an interview, though, one hospitalist shared the particulars of his professional and personal journey since arriving in the United States from India 15 years ago.
Mihir Patel, MD, MPH, FHM, came to the United States in 2005 to complete a Master’s in Public Health. Fifteen years later, he is still waiting for the green card that signifies U.S. permanent residency status. The paperwork for the application, he said, was completed in 2012. Since then, he’s been renewing his H-1B visa every three years, and he has no expectation that anything will change soon.
“If you are from India, which has a significant backlog of green cards – up to 50 years…you just wait forever,” he said. “Many people even die waiting for their green card to arrive.”
Arriving on a student visa, Dr. Patel completed his MPH in 2008 and began an internal medicine residency that same year, holding a J-1 visa for the 3 years of his US residency program.
“Post-residency, I started working in a rural hospital in an underserved area of northeast Tennessee as a hospitalist,” thus completing the 3 years of service in a rural underserved area that’s a requirement for J-1 visa holders, said Dr. Patel. “I loved this rural community hospital so much that I ended up staying there for 6 years. During my work at this rural hospital, I was able to enjoy the autonomy of managing a small ICU, doing both critical care procedures and management of intubated critical patients while working as a hospitalist,” he said. Dr. Patel served as chief of staff at the hospital for two years, and also served on the board of directors for his 400-physician medical group.
“I was a proud member of this rural community – Rogersville,” said Dr. Patel. Although he and his wife, who was completing her hospitalist residency, lived in Johnson City, Tenn., “I did not mind driving 120 miles round trip every day to go to my small-town hospital for 6 years,” he said.
Spending this time in rural Tennessee allowed Dr. Patel to finish the requirements necessary for the Physician National Interest Waiver and submit his application for permanent residency. The waiver, though, doesn’t give him priority status in the waiting list for permanent residency status.
After a stint in northern California to be closer to extended family, the pull of “beautiful northeast Tennesse and the rural community” was too strong, so Dr. Patel and his family moved back to Johnson City in 2018.
Now, Dr. Patel is a hospitalist at Ballad Health System in Johnson City. He is the corporate director of Ballad’s telemedicine program and is now also the medical director of the COVID-19 Strike Team. He co-founded and is president of the Blue Ridge Chapter of the Society of Hospital Medicine. Under another H-1B visa, Dr. Patel works part-time from home as a telehospitalist, covering six hospitals in 4 different states.
Even in ordinary circumstances, the H-1B visa comes with constraints. Although Dr. Patel’s 6-year old daughter was born in the U.S. and is a citizen, Dr. Patel and his wife have to reapply for their visas every 3 years. “If we travel outside the U.S., we have to get our visas stamped. We cannot change jobs easily due to fear of visa denial, especially with the recent political environment,” said Dr. Patel. “It feels like we are essential health care workers but non-essential immigrants.”
Having recently completed a physician executive MBA program, Dr. Patel said he’d like to start a business of his own using Lean health care principles and telemedicine to improve rural health care. “But while on an H-1B I cannot do anything outside my sponsored employment,” he said.
Ideally, health care organizations would have high flexibility in how and where staff are deployed when a surge of COVID-19 patients hits. Dr. Patel made the point that visa restrictions can make this much harder: “During this COVID crisis, this restriction can cause significant negative impact for small rural hospitals, where local physicians are quarantined and available physicians are on a visa who cannot legally work outside their primary facilities – even though they are willing to work,” he said. “One cannot even work using telemedicine in the same health system, if that is not specifically mentioned during H-1B petition filling. More than 15,000 physicians who are struck by the green card backlog are in the same situation all over U.S.,” he added.
These constraints, though, pale before the consequences of a worst-case pandemic scenario for an immigrant family, where the physician – the primary visa-holder – becomes disabled or dies. In this case, dependent family members must self-deport. “In addition, there would not be any disability or Social Security benefits for the physician or dependents, as they are not citizens or green card holders and they cannot legally stay in the US,” noted Dr. Patel. “Any hospitalist working during the COVID-19 pandemic can have this fate due to our high exposure risk.”
Reauthorizing the H1-B visa program
SHM has been advocating to improve the H1-B visa system for years, Dr. Patel said, The Fairness for High Skilled Immigrants Act passed the U.S. House of Representatives with bipartisan support, and the Society is advocating for its passage in the Senate.
The Fairness for High-Skilled Immigrants Act (S. 386) simplifies the employment-based immigration system by removing per-country caps, converting the employment-based immigration system into a “first-come, first serve” system that does not discriminate on country of origin. The act will also help alleviate the decades-long green card and permanent residency application backlogs.
Dr. Patel emphasized the importance of action by Congress to reauthorize the physician visa waiver program and expediting physician permanent residency. “This is a crisis and we are all physicians who are ready to serve, regardless of our country of origin. Please let us help this great nation by giving us freedom from visa restrictions and providing security for our families.
“During wartime, all frontline soldiers are naturalized and given citizenship by presidential mandate; this is more than war and we are not asking for citizenship – but at least give us a green card which we have already satisfied all requirements for. If not now, then when?” he asked.
H1-B visa program needs improvement
H1-B visa program needs improvement
Statistics tell the tale of immigrants in the American health care workforce in broad strokes. In an interview, though, one hospitalist shared the particulars of his professional and personal journey since arriving in the United States from India 15 years ago.
Mihir Patel, MD, MPH, FHM, came to the United States in 2005 to complete a Master’s in Public Health. Fifteen years later, he is still waiting for the green card that signifies U.S. permanent residency status. The paperwork for the application, he said, was completed in 2012. Since then, he’s been renewing his H-1B visa every three years, and he has no expectation that anything will change soon.
“If you are from India, which has a significant backlog of green cards – up to 50 years…you just wait forever,” he said. “Many people even die waiting for their green card to arrive.”
Arriving on a student visa, Dr. Patel completed his MPH in 2008 and began an internal medicine residency that same year, holding a J-1 visa for the 3 years of his US residency program.
“Post-residency, I started working in a rural hospital in an underserved area of northeast Tennessee as a hospitalist,” thus completing the 3 years of service in a rural underserved area that’s a requirement for J-1 visa holders, said Dr. Patel. “I loved this rural community hospital so much that I ended up staying there for 6 years. During my work at this rural hospital, I was able to enjoy the autonomy of managing a small ICU, doing both critical care procedures and management of intubated critical patients while working as a hospitalist,” he said. Dr. Patel served as chief of staff at the hospital for two years, and also served on the board of directors for his 400-physician medical group.
“I was a proud member of this rural community – Rogersville,” said Dr. Patel. Although he and his wife, who was completing her hospitalist residency, lived in Johnson City, Tenn., “I did not mind driving 120 miles round trip every day to go to my small-town hospital for 6 years,” he said.
Spending this time in rural Tennessee allowed Dr. Patel to finish the requirements necessary for the Physician National Interest Waiver and submit his application for permanent residency. The waiver, though, doesn’t give him priority status in the waiting list for permanent residency status.
After a stint in northern California to be closer to extended family, the pull of “beautiful northeast Tennesse and the rural community” was too strong, so Dr. Patel and his family moved back to Johnson City in 2018.
Now, Dr. Patel is a hospitalist at Ballad Health System in Johnson City. He is the corporate director of Ballad’s telemedicine program and is now also the medical director of the COVID-19 Strike Team. He co-founded and is president of the Blue Ridge Chapter of the Society of Hospital Medicine. Under another H-1B visa, Dr. Patel works part-time from home as a telehospitalist, covering six hospitals in 4 different states.
Even in ordinary circumstances, the H-1B visa comes with constraints. Although Dr. Patel’s 6-year old daughter was born in the U.S. and is a citizen, Dr. Patel and his wife have to reapply for their visas every 3 years. “If we travel outside the U.S., we have to get our visas stamped. We cannot change jobs easily due to fear of visa denial, especially with the recent political environment,” said Dr. Patel. “It feels like we are essential health care workers but non-essential immigrants.”
Having recently completed a physician executive MBA program, Dr. Patel said he’d like to start a business of his own using Lean health care principles and telemedicine to improve rural health care. “But while on an H-1B I cannot do anything outside my sponsored employment,” he said.
Ideally, health care organizations would have high flexibility in how and where staff are deployed when a surge of COVID-19 patients hits. Dr. Patel made the point that visa restrictions can make this much harder: “During this COVID crisis, this restriction can cause significant negative impact for small rural hospitals, where local physicians are quarantined and available physicians are on a visa who cannot legally work outside their primary facilities – even though they are willing to work,” he said. “One cannot even work using telemedicine in the same health system, if that is not specifically mentioned during H-1B petition filling. More than 15,000 physicians who are struck by the green card backlog are in the same situation all over U.S.,” he added.
These constraints, though, pale before the consequences of a worst-case pandemic scenario for an immigrant family, where the physician – the primary visa-holder – becomes disabled or dies. In this case, dependent family members must self-deport. “In addition, there would not be any disability or Social Security benefits for the physician or dependents, as they are not citizens or green card holders and they cannot legally stay in the US,” noted Dr. Patel. “Any hospitalist working during the COVID-19 pandemic can have this fate due to our high exposure risk.”
Reauthorizing the H1-B visa program
SHM has been advocating to improve the H1-B visa system for years, Dr. Patel said, The Fairness for High Skilled Immigrants Act passed the U.S. House of Representatives with bipartisan support, and the Society is advocating for its passage in the Senate.
The Fairness for High-Skilled Immigrants Act (S. 386) simplifies the employment-based immigration system by removing per-country caps, converting the employment-based immigration system into a “first-come, first serve” system that does not discriminate on country of origin. The act will also help alleviate the decades-long green card and permanent residency application backlogs.
Dr. Patel emphasized the importance of action by Congress to reauthorize the physician visa waiver program and expediting physician permanent residency. “This is a crisis and we are all physicians who are ready to serve, regardless of our country of origin. Please let us help this great nation by giving us freedom from visa restrictions and providing security for our families.
“During wartime, all frontline soldiers are naturalized and given citizenship by presidential mandate; this is more than war and we are not asking for citizenship – but at least give us a green card which we have already satisfied all requirements for. If not now, then when?” he asked.
Statistics tell the tale of immigrants in the American health care workforce in broad strokes. In an interview, though, one hospitalist shared the particulars of his professional and personal journey since arriving in the United States from India 15 years ago.
Mihir Patel, MD, MPH, FHM, came to the United States in 2005 to complete a Master’s in Public Health. Fifteen years later, he is still waiting for the green card that signifies U.S. permanent residency status. The paperwork for the application, he said, was completed in 2012. Since then, he’s been renewing his H-1B visa every three years, and he has no expectation that anything will change soon.
“If you are from India, which has a significant backlog of green cards – up to 50 years…you just wait forever,” he said. “Many people even die waiting for their green card to arrive.”
Arriving on a student visa, Dr. Patel completed his MPH in 2008 and began an internal medicine residency that same year, holding a J-1 visa for the 3 years of his US residency program.
“Post-residency, I started working in a rural hospital in an underserved area of northeast Tennessee as a hospitalist,” thus completing the 3 years of service in a rural underserved area that’s a requirement for J-1 visa holders, said Dr. Patel. “I loved this rural community hospital so much that I ended up staying there for 6 years. During my work at this rural hospital, I was able to enjoy the autonomy of managing a small ICU, doing both critical care procedures and management of intubated critical patients while working as a hospitalist,” he said. Dr. Patel served as chief of staff at the hospital for two years, and also served on the board of directors for his 400-physician medical group.
“I was a proud member of this rural community – Rogersville,” said Dr. Patel. Although he and his wife, who was completing her hospitalist residency, lived in Johnson City, Tenn., “I did not mind driving 120 miles round trip every day to go to my small-town hospital for 6 years,” he said.
Spending this time in rural Tennessee allowed Dr. Patel to finish the requirements necessary for the Physician National Interest Waiver and submit his application for permanent residency. The waiver, though, doesn’t give him priority status in the waiting list for permanent residency status.
After a stint in northern California to be closer to extended family, the pull of “beautiful northeast Tennesse and the rural community” was too strong, so Dr. Patel and his family moved back to Johnson City in 2018.
Now, Dr. Patel is a hospitalist at Ballad Health System in Johnson City. He is the corporate director of Ballad’s telemedicine program and is now also the medical director of the COVID-19 Strike Team. He co-founded and is president of the Blue Ridge Chapter of the Society of Hospital Medicine. Under another H-1B visa, Dr. Patel works part-time from home as a telehospitalist, covering six hospitals in 4 different states.
Even in ordinary circumstances, the H-1B visa comes with constraints. Although Dr. Patel’s 6-year old daughter was born in the U.S. and is a citizen, Dr. Patel and his wife have to reapply for their visas every 3 years. “If we travel outside the U.S., we have to get our visas stamped. We cannot change jobs easily due to fear of visa denial, especially with the recent political environment,” said Dr. Patel. “It feels like we are essential health care workers but non-essential immigrants.”
Having recently completed a physician executive MBA program, Dr. Patel said he’d like to start a business of his own using Lean health care principles and telemedicine to improve rural health care. “But while on an H-1B I cannot do anything outside my sponsored employment,” he said.
Ideally, health care organizations would have high flexibility in how and where staff are deployed when a surge of COVID-19 patients hits. Dr. Patel made the point that visa restrictions can make this much harder: “During this COVID crisis, this restriction can cause significant negative impact for small rural hospitals, where local physicians are quarantined and available physicians are on a visa who cannot legally work outside their primary facilities – even though they are willing to work,” he said. “One cannot even work using telemedicine in the same health system, if that is not specifically mentioned during H-1B petition filling. More than 15,000 physicians who are struck by the green card backlog are in the same situation all over U.S.,” he added.
These constraints, though, pale before the consequences of a worst-case pandemic scenario for an immigrant family, where the physician – the primary visa-holder – becomes disabled or dies. In this case, dependent family members must self-deport. “In addition, there would not be any disability or Social Security benefits for the physician or dependents, as they are not citizens or green card holders and they cannot legally stay in the US,” noted Dr. Patel. “Any hospitalist working during the COVID-19 pandemic can have this fate due to our high exposure risk.”
Reauthorizing the H1-B visa program
SHM has been advocating to improve the H1-B visa system for years, Dr. Patel said, The Fairness for High Skilled Immigrants Act passed the U.S. House of Representatives with bipartisan support, and the Society is advocating for its passage in the Senate.
The Fairness for High-Skilled Immigrants Act (S. 386) simplifies the employment-based immigration system by removing per-country caps, converting the employment-based immigration system into a “first-come, first serve” system that does not discriminate on country of origin. The act will also help alleviate the decades-long green card and permanent residency application backlogs.
Dr. Patel emphasized the importance of action by Congress to reauthorize the physician visa waiver program and expediting physician permanent residency. “This is a crisis and we are all physicians who are ready to serve, regardless of our country of origin. Please let us help this great nation by giving us freedom from visa restrictions and providing security for our families.
“During wartime, all frontline soldiers are naturalized and given citizenship by presidential mandate; this is more than war and we are not asking for citizenship – but at least give us a green card which we have already satisfied all requirements for. If not now, then when?” he asked.