User login
‘Profound human toll’ in excess deaths from COVID-19 calculated in two studies
However, additional deaths could be indirectly related because people avoided emergency care during the pandemic, new research shows.
Deaths linked to COVID-19 varied by state and phase of the pandemic, as reported in a study from researchers at Virginia Commonwealth University and Yale University that was published online October 12 in JAMA.
Another study published online simultaneously in JAMA took more of an international perspective. Investigators from the University of Pennsylvania and Harvard University found that in America there were more excess deaths and there was higher all-cause mortality during the pandemic than in 18 other countries.
Although the ongoing number of deaths attributable to COVID-19 continues to garner attention, there can be a lag of weeks or months in how long it takes some public health agencies to update their figures.
“For the public at large, the take-home message is twofold: that the number of deaths caused by the pandemic exceeds publicly reported COVID-19 death counts by 20% and that states that reopened or lifted restrictions early suffered a protracted surge in excess deaths that extended into the summer,” lead author of the US-focused study, Steven H. Woolf, MD, MPH, told Medscape Medical News.
The take-away for physicians is in the bigger picture – it is likely that the COVID-19 pandemic is responsible for deaths from other conditions as well. “Surges in COVID-19 were accompanied by an increase in deaths attributed to other causes, such as heart disease and Alzheimer’s disease and dementia,” said Woolf, director emeritus and senior adviser at the Center on Society and Health and professor in the Department of Family Medicine and Population Health at the Virginia Commonwealth University School of Medicine in Richmond, Virginia.
The investigators identified 225,530 excess US deaths in the 5 months from March to July. They report that 67% were directly attributable to COVID-19.
Deaths linked to COVID-19 included those in which the disease was listed as an underlying or contributing cause. US total death rates are “remarkably consistent” year after year, and the investigators calculated a 20% overall jump in mortality.
The study included data from the National Center for Health Statistics and the US Census Bureau for 48 states and the District of Columbia. Connecticut and North Carolina were excluded because of missing data.
Woolf and colleagues also found statistically higher rates of deaths from two other causes, heart disease and Alzheimer’s disease/dementia.
Altered states
New York, New Jersey, Massachusetts, Louisiana, Arizona, Mississippi, Maryland, Delaware, Rhode Island, and Michigan had the highest per capita excess death rates. Three states experienced the shortest epidemics during the study period: New York, New Jersey, and Massachusetts.
Some lessons could be learned by looking at how individual states managed large numbers of people with COVID-19. “Although we suspected that states that reopened early might have put themselves at risk of a pandemic surge, the consistency with which that occurred and the devastating numbers of deaths they suffered was a surprise,” Woolf said.
“The goal of our study is not to look in the rearview mirror and lament what happened months ago but to learn the lesson going forward: Our country will be unable to take control of this pandemic without more robust efforts to control community spread,” Woolf said. “Our study found that states that did this well, such as New York and New Jersey, experienced large surges but bent the curve and were back to baseline in less than 10 weeks.
“If we could do this as a country, countless lives could be saved.”
A global perspective
The United States experienced high mortality linked to COVID-19, as well as high all-cause mortality, compared with 18 other countries, as reported in the study by University of Pennsylvania and Harvard University researchers.
The United States ranked third, with 72 deaths per 100,000 people, among countries with moderate or high mortality. Although perhaps not surprising given the state of SARS-CoV-2 infection across the United States, a question remains as to what extent the relatively high mortality rate is linked to early outbreaks vs “poor long-term response,” the researchers note.
Alyssa Bilinski, MSc, and lead author Ezekiel J. Emanuel, MD, PhD, chair of the Department of Medical Ethics and Health Policy at the University of Pennsylvania Perelman School of Medicine in Philadelphia, calculated the difference in COVID-19 deaths among countries through Sept. 19, 2020. On this date, the United States reported a total 198,589 COVID-19 deaths.
They calculated that, if the US death rates were similar to those in Australia, the United States would have experienced 187,661 fewer COVID-19 deaths. If similar to those of Canada, there would have been 117,622 fewer deaths in the United States.
The US death rate was lower than six other countries with high COVID-19 mortality in the early spring, including Belgium, Spain, and the United Kingdom. However, after May 10, the per capita mortality rate in the United States exceeded the others.
Between May 10 and Sept. 19, the death rate in Italy was 9.1 per 100,000, vs 36.9 per 100,000.
“After the first peak in early spring, US death rates from COVID-19 and from all causes remained higher than even countries with high COVID-19 mortality,” the researchers note. “This may have been a result of several factors, including weak public health infrastructure and a decentralized, inconsistent US response to the pandemic.”
“Mortifying and motivating”
Woolf and colleagues estimate that more than 225,000 excess deaths occurred in recent months; this represents a 20% increase over expected deaths, note Harvey V. Fineberg, MD, PhD, of the Gordon and Betty Moore Foundation, in an accompanying editorial in JAMA.
“Importantly, a condition such as COVID-19 can contribute both directly and indirectly to excess mortality,” he writes.
Although the direct contribution to the mortality rates by those infected is straightforward, “the indirect contribution may relate to circumstances or choices due to the COVID-19 pandemic: for example, a patient who develops symptoms of a stroke is too concerned about COVID-19 to go to the emergency department, and a potentially reversible condition becomes fatal.”
Fineberg notes that “a general indication of the death toll from COVID-19 and the excess deaths related to the pandemic, as presented by Woolf et al, are sufficiently mortifying and motivating.”
“Profound human toll”
“The importance of the estimate by Woolf et al – which suggests that for the entirety of 2020, more than 400,000 excess deaths will occur – cannot be overstated, because it accounts for what could be declines in some causes of death, like motor vehicle crashes, but increases in others, like myocardial infarction,” write Howard Bauchner, MD, editor in chief of JAMA, and Phil B. Fontanarosa, MD, MBA, executive editor of JAMA, in another accompanying editorial.
“These deaths reflect a true measure of the human cost of the Great Pandemic of 2020,” they add.
The study from Emanuel and Bilinski was notable for calculating the excess COVID-19 and all-cause mortality to Sept. 2020, they note. “After the initial peak in early spring, US death rates from COVID-19 and from all causes remained higher than rates in countries with high COVID-19 mortality.”
“Few people will forget the Great Pandemic of 2020, where and how they lived, how it substantially changed their lives, and for many, the profound human toll it has taken,” Bauchner and Fontanarosa write.
The study by Woolf and colleagues was supported by National Center for Advancing Translational Sciences, the National Institute on Aging, and the National Institute of Allergy and Infectious Diseases. The study by Bilinski and Emanuel was partially funded by the Colton Foundation. Woolf, Emanuel, Fineberg, Bauchner, and Fontanarosa have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
However, additional deaths could be indirectly related because people avoided emergency care during the pandemic, new research shows.
Deaths linked to COVID-19 varied by state and phase of the pandemic, as reported in a study from researchers at Virginia Commonwealth University and Yale University that was published online October 12 in JAMA.
Another study published online simultaneously in JAMA took more of an international perspective. Investigators from the University of Pennsylvania and Harvard University found that in America there were more excess deaths and there was higher all-cause mortality during the pandemic than in 18 other countries.
Although the ongoing number of deaths attributable to COVID-19 continues to garner attention, there can be a lag of weeks or months in how long it takes some public health agencies to update their figures.
“For the public at large, the take-home message is twofold: that the number of deaths caused by the pandemic exceeds publicly reported COVID-19 death counts by 20% and that states that reopened or lifted restrictions early suffered a protracted surge in excess deaths that extended into the summer,” lead author of the US-focused study, Steven H. Woolf, MD, MPH, told Medscape Medical News.
The take-away for physicians is in the bigger picture – it is likely that the COVID-19 pandemic is responsible for deaths from other conditions as well. “Surges in COVID-19 were accompanied by an increase in deaths attributed to other causes, such as heart disease and Alzheimer’s disease and dementia,” said Woolf, director emeritus and senior adviser at the Center on Society and Health and professor in the Department of Family Medicine and Population Health at the Virginia Commonwealth University School of Medicine in Richmond, Virginia.
The investigators identified 225,530 excess US deaths in the 5 months from March to July. They report that 67% were directly attributable to COVID-19.
Deaths linked to COVID-19 included those in which the disease was listed as an underlying or contributing cause. US total death rates are “remarkably consistent” year after year, and the investigators calculated a 20% overall jump in mortality.
The study included data from the National Center for Health Statistics and the US Census Bureau for 48 states and the District of Columbia. Connecticut and North Carolina were excluded because of missing data.
Woolf and colleagues also found statistically higher rates of deaths from two other causes, heart disease and Alzheimer’s disease/dementia.
Altered states
New York, New Jersey, Massachusetts, Louisiana, Arizona, Mississippi, Maryland, Delaware, Rhode Island, and Michigan had the highest per capita excess death rates. Three states experienced the shortest epidemics during the study period: New York, New Jersey, and Massachusetts.
Some lessons could be learned by looking at how individual states managed large numbers of people with COVID-19. “Although we suspected that states that reopened early might have put themselves at risk of a pandemic surge, the consistency with which that occurred and the devastating numbers of deaths they suffered was a surprise,” Woolf said.
“The goal of our study is not to look in the rearview mirror and lament what happened months ago but to learn the lesson going forward: Our country will be unable to take control of this pandemic without more robust efforts to control community spread,” Woolf said. “Our study found that states that did this well, such as New York and New Jersey, experienced large surges but bent the curve and were back to baseline in less than 10 weeks.
“If we could do this as a country, countless lives could be saved.”
A global perspective
The United States experienced high mortality linked to COVID-19, as well as high all-cause mortality, compared with 18 other countries, as reported in the study by University of Pennsylvania and Harvard University researchers.
The United States ranked third, with 72 deaths per 100,000 people, among countries with moderate or high mortality. Although perhaps not surprising given the state of SARS-CoV-2 infection across the United States, a question remains as to what extent the relatively high mortality rate is linked to early outbreaks vs “poor long-term response,” the researchers note.
Alyssa Bilinski, MSc, and lead author Ezekiel J. Emanuel, MD, PhD, chair of the Department of Medical Ethics and Health Policy at the University of Pennsylvania Perelman School of Medicine in Philadelphia, calculated the difference in COVID-19 deaths among countries through Sept. 19, 2020. On this date, the United States reported a total 198,589 COVID-19 deaths.
They calculated that, if the US death rates were similar to those in Australia, the United States would have experienced 187,661 fewer COVID-19 deaths. If similar to those of Canada, there would have been 117,622 fewer deaths in the United States.
The US death rate was lower than six other countries with high COVID-19 mortality in the early spring, including Belgium, Spain, and the United Kingdom. However, after May 10, the per capita mortality rate in the United States exceeded the others.
Between May 10 and Sept. 19, the death rate in Italy was 9.1 per 100,000, vs 36.9 per 100,000.
“After the first peak in early spring, US death rates from COVID-19 and from all causes remained higher than even countries with high COVID-19 mortality,” the researchers note. “This may have been a result of several factors, including weak public health infrastructure and a decentralized, inconsistent US response to the pandemic.”
“Mortifying and motivating”
Woolf and colleagues estimate that more than 225,000 excess deaths occurred in recent months; this represents a 20% increase over expected deaths, note Harvey V. Fineberg, MD, PhD, of the Gordon and Betty Moore Foundation, in an accompanying editorial in JAMA.
“Importantly, a condition such as COVID-19 can contribute both directly and indirectly to excess mortality,” he writes.
Although the direct contribution to the mortality rates by those infected is straightforward, “the indirect contribution may relate to circumstances or choices due to the COVID-19 pandemic: for example, a patient who develops symptoms of a stroke is too concerned about COVID-19 to go to the emergency department, and a potentially reversible condition becomes fatal.”
Fineberg notes that “a general indication of the death toll from COVID-19 and the excess deaths related to the pandemic, as presented by Woolf et al, are sufficiently mortifying and motivating.”
“Profound human toll”
“The importance of the estimate by Woolf et al – which suggests that for the entirety of 2020, more than 400,000 excess deaths will occur – cannot be overstated, because it accounts for what could be declines in some causes of death, like motor vehicle crashes, but increases in others, like myocardial infarction,” write Howard Bauchner, MD, editor in chief of JAMA, and Phil B. Fontanarosa, MD, MBA, executive editor of JAMA, in another accompanying editorial.
“These deaths reflect a true measure of the human cost of the Great Pandemic of 2020,” they add.
The study from Emanuel and Bilinski was notable for calculating the excess COVID-19 and all-cause mortality to Sept. 2020, they note. “After the initial peak in early spring, US death rates from COVID-19 and from all causes remained higher than rates in countries with high COVID-19 mortality.”
“Few people will forget the Great Pandemic of 2020, where and how they lived, how it substantially changed their lives, and for many, the profound human toll it has taken,” Bauchner and Fontanarosa write.
The study by Woolf and colleagues was supported by National Center for Advancing Translational Sciences, the National Institute on Aging, and the National Institute of Allergy and Infectious Diseases. The study by Bilinski and Emanuel was partially funded by the Colton Foundation. Woolf, Emanuel, Fineberg, Bauchner, and Fontanarosa have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
However, additional deaths could be indirectly related because people avoided emergency care during the pandemic, new research shows.
Deaths linked to COVID-19 varied by state and phase of the pandemic, as reported in a study from researchers at Virginia Commonwealth University and Yale University that was published online October 12 in JAMA.
Another study published online simultaneously in JAMA took more of an international perspective. Investigators from the University of Pennsylvania and Harvard University found that in America there were more excess deaths and there was higher all-cause mortality during the pandemic than in 18 other countries.
Although the ongoing number of deaths attributable to COVID-19 continues to garner attention, there can be a lag of weeks or months in how long it takes some public health agencies to update their figures.
“For the public at large, the take-home message is twofold: that the number of deaths caused by the pandemic exceeds publicly reported COVID-19 death counts by 20% and that states that reopened or lifted restrictions early suffered a protracted surge in excess deaths that extended into the summer,” lead author of the US-focused study, Steven H. Woolf, MD, MPH, told Medscape Medical News.
The take-away for physicians is in the bigger picture – it is likely that the COVID-19 pandemic is responsible for deaths from other conditions as well. “Surges in COVID-19 were accompanied by an increase in deaths attributed to other causes, such as heart disease and Alzheimer’s disease and dementia,” said Woolf, director emeritus and senior adviser at the Center on Society and Health and professor in the Department of Family Medicine and Population Health at the Virginia Commonwealth University School of Medicine in Richmond, Virginia.
The investigators identified 225,530 excess US deaths in the 5 months from March to July. They report that 67% were directly attributable to COVID-19.
Deaths linked to COVID-19 included those in which the disease was listed as an underlying or contributing cause. US total death rates are “remarkably consistent” year after year, and the investigators calculated a 20% overall jump in mortality.
The study included data from the National Center for Health Statistics and the US Census Bureau for 48 states and the District of Columbia. Connecticut and North Carolina were excluded because of missing data.
Woolf and colleagues also found statistically higher rates of deaths from two other causes, heart disease and Alzheimer’s disease/dementia.
Altered states
New York, New Jersey, Massachusetts, Louisiana, Arizona, Mississippi, Maryland, Delaware, Rhode Island, and Michigan had the highest per capita excess death rates. Three states experienced the shortest epidemics during the study period: New York, New Jersey, and Massachusetts.
Some lessons could be learned by looking at how individual states managed large numbers of people with COVID-19. “Although we suspected that states that reopened early might have put themselves at risk of a pandemic surge, the consistency with which that occurred and the devastating numbers of deaths they suffered was a surprise,” Woolf said.
“The goal of our study is not to look in the rearview mirror and lament what happened months ago but to learn the lesson going forward: Our country will be unable to take control of this pandemic without more robust efforts to control community spread,” Woolf said. “Our study found that states that did this well, such as New York and New Jersey, experienced large surges but bent the curve and were back to baseline in less than 10 weeks.
“If we could do this as a country, countless lives could be saved.”
A global perspective
The United States experienced high mortality linked to COVID-19, as well as high all-cause mortality, compared with 18 other countries, as reported in the study by University of Pennsylvania and Harvard University researchers.
The United States ranked third, with 72 deaths per 100,000 people, among countries with moderate or high mortality. Although perhaps not surprising given the state of SARS-CoV-2 infection across the United States, a question remains as to what extent the relatively high mortality rate is linked to early outbreaks vs “poor long-term response,” the researchers note.
Alyssa Bilinski, MSc, and lead author Ezekiel J. Emanuel, MD, PhD, chair of the Department of Medical Ethics and Health Policy at the University of Pennsylvania Perelman School of Medicine in Philadelphia, calculated the difference in COVID-19 deaths among countries through Sept. 19, 2020. On this date, the United States reported a total 198,589 COVID-19 deaths.
They calculated that, if the US death rates were similar to those in Australia, the United States would have experienced 187,661 fewer COVID-19 deaths. If similar to those of Canada, there would have been 117,622 fewer deaths in the United States.
The US death rate was lower than six other countries with high COVID-19 mortality in the early spring, including Belgium, Spain, and the United Kingdom. However, after May 10, the per capita mortality rate in the United States exceeded the others.
Between May 10 and Sept. 19, the death rate in Italy was 9.1 per 100,000, vs 36.9 per 100,000.
“After the first peak in early spring, US death rates from COVID-19 and from all causes remained higher than even countries with high COVID-19 mortality,” the researchers note. “This may have been a result of several factors, including weak public health infrastructure and a decentralized, inconsistent US response to the pandemic.”
“Mortifying and motivating”
Woolf and colleagues estimate that more than 225,000 excess deaths occurred in recent months; this represents a 20% increase over expected deaths, note Harvey V. Fineberg, MD, PhD, of the Gordon and Betty Moore Foundation, in an accompanying editorial in JAMA.
“Importantly, a condition such as COVID-19 can contribute both directly and indirectly to excess mortality,” he writes.
Although the direct contribution to the mortality rates by those infected is straightforward, “the indirect contribution may relate to circumstances or choices due to the COVID-19 pandemic: for example, a patient who develops symptoms of a stroke is too concerned about COVID-19 to go to the emergency department, and a potentially reversible condition becomes fatal.”
Fineberg notes that “a general indication of the death toll from COVID-19 and the excess deaths related to the pandemic, as presented by Woolf et al, are sufficiently mortifying and motivating.”
“Profound human toll”
“The importance of the estimate by Woolf et al – which suggests that for the entirety of 2020, more than 400,000 excess deaths will occur – cannot be overstated, because it accounts for what could be declines in some causes of death, like motor vehicle crashes, but increases in others, like myocardial infarction,” write Howard Bauchner, MD, editor in chief of JAMA, and Phil B. Fontanarosa, MD, MBA, executive editor of JAMA, in another accompanying editorial.
“These deaths reflect a true measure of the human cost of the Great Pandemic of 2020,” they add.
The study from Emanuel and Bilinski was notable for calculating the excess COVID-19 and all-cause mortality to Sept. 2020, they note. “After the initial peak in early spring, US death rates from COVID-19 and from all causes remained higher than rates in countries with high COVID-19 mortality.”
“Few people will forget the Great Pandemic of 2020, where and how they lived, how it substantially changed their lives, and for many, the profound human toll it has taken,” Bauchner and Fontanarosa write.
The study by Woolf and colleagues was supported by National Center for Advancing Translational Sciences, the National Institute on Aging, and the National Institute of Allergy and Infectious Diseases. The study by Bilinski and Emanuel was partially funded by the Colton Foundation. Woolf, Emanuel, Fineberg, Bauchner, and Fontanarosa have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Fourteen-day sports hiatus recommended for children after COVID-19
Children should not return to sports for 14 days after exposure to COVID-19, and those with moderate symptoms should undergo an electrocardiogram before returning, according to the American Academy of Pediatrics.
said Susannah Briskin, MD, a pediatric sports medicine specialist at Rainbow Babies and Children’s Hospital in Cleveland.
“There has been emerging evidence about cases of myocarditis occurring in athletes, including athletes who are asymptomatic with COVID-19,” she said in an interview.
The update aligns the AAP recommendations with those from the American College of Cardiologists, she added.
Recent imaging studies have turned up signs of myocarditis in athletes recovering from mild or asymptomatic cases of COVID-19 and have prompted calls for clearer guidelines about imaging studies and return to play.
Viral myocarditis poses a risk to athletes because it can lead to potentially fatal arrhythmias, Dr. Briskin said.
Although children benefit from participating in sports, these activities also put them at risk of contracting COVID-19 and spreading it to others, the guidance noted.
To balance the risks and benefits, the academy proposed guidelines that vary depending on the severity of the presentation.
In the first category are patients with a severe presentation (hypotension, arrhythmias, need for intubation or extracorporeal membrane oxygenation support, kidney or cardiac failure) or with multisystem inflammatory syndrome. Clinicians should treat these patients as though they have myocarditis. Patients should be restricted from engaging in sports and other exercise for 3-6 months, the guidance stated.
The primary care physician and “appropriate pediatric medical subspecialist, preferably in consultation with a pediatric cardiologist,” should clear them before they return to activities. In examining patients for return to play, clinicians should focus on cardiac symptoms, including chest pain, shortness of breath, fatigue, palpitations, or syncope, the guidance said.
In another category are patients with cardiac symptoms, those with concerning findings on examination, and those with moderate symptoms of COVID-19, including prolonged fever. These patients should undergo an ECG and possibly be referred to a pediatric cardiologist, the guidelines said. These symptoms must be absent for at least 14 days before these patients can return to sports, and the athletes should obtain clearance from their primary care physicians before they resume.
In a third category are patients who have been infected with SARS-CoV-2 or who have had close contact with someone who was infected but who have not themselves experienced symptoms. These athletes should refrain from sports for at least 14 days, the guidelines said.
Children who don’t fall into any of these categories should not be tested for the virus or antibodies to it before participation in sports, the academy said.
The guidelines don’t vary depending on the sport. But the academy has issued separate guidance for parents and guardians to help them evaluate the risk for COVID-19 transmission by sport.
Athletes participating in “sports that have greater amount of contact time or proximity to people would be at higher risk for contracting COVID-19,” Dr. Briskin said. “But I think that’s all fairly common sense, given the recommendations for non–sport-related activity just in terms of social distancing and masking.”
The new guidance called on sports organizers to minimize contact by, for example, modifying drills and conditioning. It recommended that athletes wear masks except during vigorous exercise or when participating in water sports, as well as in other circumstances in which the mask could become a safety hazard.
They also recommended using handwashing stations or hand sanitizer, avoiding contact with shared surfaces, and avoiding small rooms and areas with poor ventilation.
Dr. Briskin disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Children should not return to sports for 14 days after exposure to COVID-19, and those with moderate symptoms should undergo an electrocardiogram before returning, according to the American Academy of Pediatrics.
said Susannah Briskin, MD, a pediatric sports medicine specialist at Rainbow Babies and Children’s Hospital in Cleveland.
“There has been emerging evidence about cases of myocarditis occurring in athletes, including athletes who are asymptomatic with COVID-19,” she said in an interview.
The update aligns the AAP recommendations with those from the American College of Cardiologists, she added.
Recent imaging studies have turned up signs of myocarditis in athletes recovering from mild or asymptomatic cases of COVID-19 and have prompted calls for clearer guidelines about imaging studies and return to play.
Viral myocarditis poses a risk to athletes because it can lead to potentially fatal arrhythmias, Dr. Briskin said.
Although children benefit from participating in sports, these activities also put them at risk of contracting COVID-19 and spreading it to others, the guidance noted.
To balance the risks and benefits, the academy proposed guidelines that vary depending on the severity of the presentation.
In the first category are patients with a severe presentation (hypotension, arrhythmias, need for intubation or extracorporeal membrane oxygenation support, kidney or cardiac failure) or with multisystem inflammatory syndrome. Clinicians should treat these patients as though they have myocarditis. Patients should be restricted from engaging in sports and other exercise for 3-6 months, the guidance stated.
The primary care physician and “appropriate pediatric medical subspecialist, preferably in consultation with a pediatric cardiologist,” should clear them before they return to activities. In examining patients for return to play, clinicians should focus on cardiac symptoms, including chest pain, shortness of breath, fatigue, palpitations, or syncope, the guidance said.
In another category are patients with cardiac symptoms, those with concerning findings on examination, and those with moderate symptoms of COVID-19, including prolonged fever. These patients should undergo an ECG and possibly be referred to a pediatric cardiologist, the guidelines said. These symptoms must be absent for at least 14 days before these patients can return to sports, and the athletes should obtain clearance from their primary care physicians before they resume.
In a third category are patients who have been infected with SARS-CoV-2 or who have had close contact with someone who was infected but who have not themselves experienced symptoms. These athletes should refrain from sports for at least 14 days, the guidelines said.
Children who don’t fall into any of these categories should not be tested for the virus or antibodies to it before participation in sports, the academy said.
The guidelines don’t vary depending on the sport. But the academy has issued separate guidance for parents and guardians to help them evaluate the risk for COVID-19 transmission by sport.
Athletes participating in “sports that have greater amount of contact time or proximity to people would be at higher risk for contracting COVID-19,” Dr. Briskin said. “But I think that’s all fairly common sense, given the recommendations for non–sport-related activity just in terms of social distancing and masking.”
The new guidance called on sports organizers to minimize contact by, for example, modifying drills and conditioning. It recommended that athletes wear masks except during vigorous exercise or when participating in water sports, as well as in other circumstances in which the mask could become a safety hazard.
They also recommended using handwashing stations or hand sanitizer, avoiding contact with shared surfaces, and avoiding small rooms and areas with poor ventilation.
Dr. Briskin disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Children should not return to sports for 14 days after exposure to COVID-19, and those with moderate symptoms should undergo an electrocardiogram before returning, according to the American Academy of Pediatrics.
said Susannah Briskin, MD, a pediatric sports medicine specialist at Rainbow Babies and Children’s Hospital in Cleveland.
“There has been emerging evidence about cases of myocarditis occurring in athletes, including athletes who are asymptomatic with COVID-19,” she said in an interview.
The update aligns the AAP recommendations with those from the American College of Cardiologists, she added.
Recent imaging studies have turned up signs of myocarditis in athletes recovering from mild or asymptomatic cases of COVID-19 and have prompted calls for clearer guidelines about imaging studies and return to play.
Viral myocarditis poses a risk to athletes because it can lead to potentially fatal arrhythmias, Dr. Briskin said.
Although children benefit from participating in sports, these activities also put them at risk of contracting COVID-19 and spreading it to others, the guidance noted.
To balance the risks and benefits, the academy proposed guidelines that vary depending on the severity of the presentation.
In the first category are patients with a severe presentation (hypotension, arrhythmias, need for intubation or extracorporeal membrane oxygenation support, kidney or cardiac failure) or with multisystem inflammatory syndrome. Clinicians should treat these patients as though they have myocarditis. Patients should be restricted from engaging in sports and other exercise for 3-6 months, the guidance stated.
The primary care physician and “appropriate pediatric medical subspecialist, preferably in consultation with a pediatric cardiologist,” should clear them before they return to activities. In examining patients for return to play, clinicians should focus on cardiac symptoms, including chest pain, shortness of breath, fatigue, palpitations, or syncope, the guidance said.
In another category are patients with cardiac symptoms, those with concerning findings on examination, and those with moderate symptoms of COVID-19, including prolonged fever. These patients should undergo an ECG and possibly be referred to a pediatric cardiologist, the guidelines said. These symptoms must be absent for at least 14 days before these patients can return to sports, and the athletes should obtain clearance from their primary care physicians before they resume.
In a third category are patients who have been infected with SARS-CoV-2 or who have had close contact with someone who was infected but who have not themselves experienced symptoms. These athletes should refrain from sports for at least 14 days, the guidelines said.
Children who don’t fall into any of these categories should not be tested for the virus or antibodies to it before participation in sports, the academy said.
The guidelines don’t vary depending on the sport. But the academy has issued separate guidance for parents and guardians to help them evaluate the risk for COVID-19 transmission by sport.
Athletes participating in “sports that have greater amount of contact time or proximity to people would be at higher risk for contracting COVID-19,” Dr. Briskin said. “But I think that’s all fairly common sense, given the recommendations for non–sport-related activity just in terms of social distancing and masking.”
The new guidance called on sports organizers to minimize contact by, for example, modifying drills and conditioning. It recommended that athletes wear masks except during vigorous exercise or when participating in water sports, as well as in other circumstances in which the mask could become a safety hazard.
They also recommended using handwashing stations or hand sanitizer, avoiding contact with shared surfaces, and avoiding small rooms and areas with poor ventilation.
Dr. Briskin disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Learning about “No”
To say that the pandemic has dropped us into uncharted territory is an understatement of unmeasurable proportions. Every day we learn more about it, and every day that new information brings us new challenges. COVID-19 is playing by its own set of rules. To keep pace with it societies have been forced to adapt to them, and members of those societies have had to realize that these new rules must be obeyed or be prepared to suffer the consequences.
I’m not sure exactly when it happened but gradually over my 7 and a half decades on this planet it appears that following the rules and understanding the value of “No” have become concepts to be ignored and left to gather dust in the attics and basements of our society. The tug of war between well-considered rules and the often misinterpreted concept of freedom has been ebbing and flowing since Eve plucked a forbidden apple off that tree.
In some parts of the world, the twin skills of saying and responding to “No” have become lost arts. I think it is not by chance that, of the four books I have written for parents, the one titled “How to Say No to Your Toddler” has become the most widely distributed, having been translated into Italian, Polish, and Russian. It is only slightly comforting to learn that at least some parents understand that creating rules can be important, but realize they aren’t quite sure how go about it.
As it has become clear that social distancing and mask wearing are associated with curtailing the spread of COVID-19, state and local governments have had to bone up on their long-forgotten No-saying skills. This relearning process has been particularly painful for school administrators who may have been warned that “You’ll never be able to get first and second graders to wear masks” or that “College students just won’t obey the rules.”
Both of these cautions are based on observations by educators with years of experience and certainly have a ring of truth to them. But could it be that these pessimistic predictions reflect a society in which parents and educators have lost the talent for crafting sensible rules and linking them to enforceable and rational consequences?
As colleges throughout the country have reopened using a variety of learning and residential strategies, there have been numerous incidents that validate the gloomy predictions of student misbehavior. Smaller schools seem to be having less difficulty, which is not surprising given their relative ease in fostering a sense of community. Many schools have been forced to rollback their plans for in-person learning because students have failed to follow some very simple but unpopular rules.
In a swift and decisive response to student misbehavior, Northeastern University in Boston dismissed 11 first-year students and will not refund their tuition when officials discovered a prohibited social gathering in one of the resident facilities (“Northeastern Dismisses 11 Students for Gathering in Violation of COVID-19 Policies,” by Ian Thomsen, News at Northwestern). This response seemed to have come as a surprise to many students and parents around the country who have become accustomed a diet of warnings and minor sanctions.
Whether this action by Northeastern will trigger similar responses by other universities remains to be seen. But we can hope that it sets an example of how learning about “No” can be an important part of one’s education.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at pdnews@mdedge.com.
To say that the pandemic has dropped us into uncharted territory is an understatement of unmeasurable proportions. Every day we learn more about it, and every day that new information brings us new challenges. COVID-19 is playing by its own set of rules. To keep pace with it societies have been forced to adapt to them, and members of those societies have had to realize that these new rules must be obeyed or be prepared to suffer the consequences.
I’m not sure exactly when it happened but gradually over my 7 and a half decades on this planet it appears that following the rules and understanding the value of “No” have become concepts to be ignored and left to gather dust in the attics and basements of our society. The tug of war between well-considered rules and the often misinterpreted concept of freedom has been ebbing and flowing since Eve plucked a forbidden apple off that tree.
In some parts of the world, the twin skills of saying and responding to “No” have become lost arts. I think it is not by chance that, of the four books I have written for parents, the one titled “How to Say No to Your Toddler” has become the most widely distributed, having been translated into Italian, Polish, and Russian. It is only slightly comforting to learn that at least some parents understand that creating rules can be important, but realize they aren’t quite sure how go about it.
As it has become clear that social distancing and mask wearing are associated with curtailing the spread of COVID-19, state and local governments have had to bone up on their long-forgotten No-saying skills. This relearning process has been particularly painful for school administrators who may have been warned that “You’ll never be able to get first and second graders to wear masks” or that “College students just won’t obey the rules.”
Both of these cautions are based on observations by educators with years of experience and certainly have a ring of truth to them. But could it be that these pessimistic predictions reflect a society in which parents and educators have lost the talent for crafting sensible rules and linking them to enforceable and rational consequences?
As colleges throughout the country have reopened using a variety of learning and residential strategies, there have been numerous incidents that validate the gloomy predictions of student misbehavior. Smaller schools seem to be having less difficulty, which is not surprising given their relative ease in fostering a sense of community. Many schools have been forced to rollback their plans for in-person learning because students have failed to follow some very simple but unpopular rules.
In a swift and decisive response to student misbehavior, Northeastern University in Boston dismissed 11 first-year students and will not refund their tuition when officials discovered a prohibited social gathering in one of the resident facilities (“Northeastern Dismisses 11 Students for Gathering in Violation of COVID-19 Policies,” by Ian Thomsen, News at Northwestern). This response seemed to have come as a surprise to many students and parents around the country who have become accustomed a diet of warnings and minor sanctions.
Whether this action by Northeastern will trigger similar responses by other universities remains to be seen. But we can hope that it sets an example of how learning about “No” can be an important part of one’s education.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at pdnews@mdedge.com.
To say that the pandemic has dropped us into uncharted territory is an understatement of unmeasurable proportions. Every day we learn more about it, and every day that new information brings us new challenges. COVID-19 is playing by its own set of rules. To keep pace with it societies have been forced to adapt to them, and members of those societies have had to realize that these new rules must be obeyed or be prepared to suffer the consequences.
I’m not sure exactly when it happened but gradually over my 7 and a half decades on this planet it appears that following the rules and understanding the value of “No” have become concepts to be ignored and left to gather dust in the attics and basements of our society. The tug of war between well-considered rules and the often misinterpreted concept of freedom has been ebbing and flowing since Eve plucked a forbidden apple off that tree.
In some parts of the world, the twin skills of saying and responding to “No” have become lost arts. I think it is not by chance that, of the four books I have written for parents, the one titled “How to Say No to Your Toddler” has become the most widely distributed, having been translated into Italian, Polish, and Russian. It is only slightly comforting to learn that at least some parents understand that creating rules can be important, but realize they aren’t quite sure how go about it.
As it has become clear that social distancing and mask wearing are associated with curtailing the spread of COVID-19, state and local governments have had to bone up on their long-forgotten No-saying skills. This relearning process has been particularly painful for school administrators who may have been warned that “You’ll never be able to get first and second graders to wear masks” or that “College students just won’t obey the rules.”
Both of these cautions are based on observations by educators with years of experience and certainly have a ring of truth to them. But could it be that these pessimistic predictions reflect a society in which parents and educators have lost the talent for crafting sensible rules and linking them to enforceable and rational consequences?
As colleges throughout the country have reopened using a variety of learning and residential strategies, there have been numerous incidents that validate the gloomy predictions of student misbehavior. Smaller schools seem to be having less difficulty, which is not surprising given their relative ease in fostering a sense of community. Many schools have been forced to rollback their plans for in-person learning because students have failed to follow some very simple but unpopular rules.
In a swift and decisive response to student misbehavior, Northeastern University in Boston dismissed 11 first-year students and will not refund their tuition when officials discovered a prohibited social gathering in one of the resident facilities (“Northeastern Dismisses 11 Students for Gathering in Violation of COVID-19 Policies,” by Ian Thomsen, News at Northwestern). This response seemed to have come as a surprise to many students and parents around the country who have become accustomed a diet of warnings and minor sanctions.
Whether this action by Northeastern will trigger similar responses by other universities remains to be seen. But we can hope that it sets an example of how learning about “No” can be an important part of one’s education.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at pdnews@mdedge.com.
Prescribe Halloween safety by region, current conditions
Halloween is fast approaching and retail stores are fully stocked with costumes and candy. Physician dialog is beginning to shift from school access toward how to counsel patients and families on COVID-19 safety around Halloween. advised pediatrician Shelly Vaziri Flais, MD.

Halloween “is going to look very different this year, especially in urban and rural settings, according to Dr. Flais, who is a spokesperson for the American Academy of Pediatrics and assistant professor of clinical pediatrics at Northwestern University, Chicago. The notion that trick-or-treating automatically involves physically distancing is a misconception. Urban celebrations frequently see many people gathering on the streets, and that will be even more likely in a pandemic year when people have been separated for long periods of time.
For pediatricians advising families on COVID-19 safety measures to follow while celebrating Halloween, it’s not going to be a one-size-fits-all approach, said Dr. Flais, who practices pediatrics at Pediatric Health Associates in Naperville, Ill.
The goal for physicians across the board should be “to ensure that we aren’t so cautious that we drive folks to do things that are higher risk,” she said in an interview. “We are now 6-7 months into the pandemic and the public is growing weary of laying low, so it is important for physicians to not recommend safety measures that are too restrictive.”
The balance pediatricians will need to strike in advising their patients is tricky at best. So in dispensing advice, it is important to make sure that it has a benefit to the overall population, cautioned Dr. Flais. Activities such as hosting independently organized, heavily packed indoor gatherings where people are eating, drinking, and not wearing masks is not going to be beneficial for the masses.
“We’re all lucky that we have technology. We’ve gotten used to doing virtual hugs and activities on Zoom,” she said, adding that she has already seen some really creative ideas on social media for enjoying a COVID-conscious Halloween, including a festive candy chute created by an Ohio family that is perfect for distributing candy while minimizing physical contact.
In an AAP press release, Dr. Flais noted that “this is a good time to teach children the importance of protecting not just ourselves but each other.” How we choose to manage our safety and the safety of our children “can have a ripple effect on our family members.” It is possible to make safe, responsible choices when celebrating and still create magical memories for our children.
Francis E. Rushton Jr., MD, of the University of South Carolina, Columbia, said in an interview, “ I certainly support the AAP recommendations. Because of the way COVID-19 virus is spread, I would emphasize with my patients that the No. 1 thing to do is to enforce facial mask wearing while out trick-or-treating.
“I would also err on the side of safety if my child was showing any signs of illness and find an alternative method of celebrating Halloween that would not involve close contact with other individuals,” said Dr. Rushton, who is a member of the Pediatric News editorial advisory board.
AAP-recommended Do’s and Don’ts for celebrating Halloween
DO:
- Avoid large gatherings.
- Maintain 6 feet distance.
- Wear cloth masks and wash hands often.
- Use hand sanitizer before and after visiting pumpkin patches and apple orchards.
DON’T:
- Wear painted cloth masks since paints can contain toxins that should not be breathed.
- Use a costume mask unless it has layers of breathable fabric snugly covering mouth and nose.
- Wear cloth mask under costume mask.
- Attend indoor parties or haunted houses.
CDC safety considerations (supplemental to state and local safety laws)
- Assess current cases and overall spread in your community before making any plans.
- Choose outdoor venues or indoor facilities that are well ventilated.
- Consider the length of the event, how many are attending, where they are coming from, and how they behave before and during the event.
- If you are awaiting test results, have COVID-19 symptoms, or have been exposed to COVID-19, stay home.
- If you are at higher risk, avoid large gatherings and limit exposure to anyone you do not live with.
- Make available to others masks, 60% or greater alcohol-based hand sanitizer, and tissues.
- Avoid touching your nose, eyes, and mouth.
- For a complete set of Centers for Disease Control and Prevention COVID safety recommendations go here.
Suggested safe, fun activities
- Use Zoom and other chat programs to share costumes, play games, and watch festive movies.
- Participate in socially distanced outdoor community events at local parks, zoos, etc.
- Attend haunted forests and corn mazes. Maintain more than 6 feet of distance around screaming patrons.
- Decorate pumpkins.
- Cook a Halloween-themed meal.
- If trick-or-treating has been canceled, try a scavenger hunt in the house or yard.
- When handing out treats, wear gloves and mask. Consider prepackaging treat bags. Line up visitors 6 feet apart and discourage gatherings around entranceways.
- Wipe down all goodies received and consider quarantining them for a few days.
- Always wash hands before and after trick-or-treating and when handling treats.
Halloween is fast approaching and retail stores are fully stocked with costumes and candy. Physician dialog is beginning to shift from school access toward how to counsel patients and families on COVID-19 safety around Halloween. advised pediatrician Shelly Vaziri Flais, MD.

Halloween “is going to look very different this year, especially in urban and rural settings, according to Dr. Flais, who is a spokesperson for the American Academy of Pediatrics and assistant professor of clinical pediatrics at Northwestern University, Chicago. The notion that trick-or-treating automatically involves physically distancing is a misconception. Urban celebrations frequently see many people gathering on the streets, and that will be even more likely in a pandemic year when people have been separated for long periods of time.
For pediatricians advising families on COVID-19 safety measures to follow while celebrating Halloween, it’s not going to be a one-size-fits-all approach, said Dr. Flais, who practices pediatrics at Pediatric Health Associates in Naperville, Ill.
The goal for physicians across the board should be “to ensure that we aren’t so cautious that we drive folks to do things that are higher risk,” she said in an interview. “We are now 6-7 months into the pandemic and the public is growing weary of laying low, so it is important for physicians to not recommend safety measures that are too restrictive.”
The balance pediatricians will need to strike in advising their patients is tricky at best. So in dispensing advice, it is important to make sure that it has a benefit to the overall population, cautioned Dr. Flais. Activities such as hosting independently organized, heavily packed indoor gatherings where people are eating, drinking, and not wearing masks is not going to be beneficial for the masses.
“We’re all lucky that we have technology. We’ve gotten used to doing virtual hugs and activities on Zoom,” she said, adding that she has already seen some really creative ideas on social media for enjoying a COVID-conscious Halloween, including a festive candy chute created by an Ohio family that is perfect for distributing candy while minimizing physical contact.
In an AAP press release, Dr. Flais noted that “this is a good time to teach children the importance of protecting not just ourselves but each other.” How we choose to manage our safety and the safety of our children “can have a ripple effect on our family members.” It is possible to make safe, responsible choices when celebrating and still create magical memories for our children.
Francis E. Rushton Jr., MD, of the University of South Carolina, Columbia, said in an interview, “ I certainly support the AAP recommendations. Because of the way COVID-19 virus is spread, I would emphasize with my patients that the No. 1 thing to do is to enforce facial mask wearing while out trick-or-treating.
“I would also err on the side of safety if my child was showing any signs of illness and find an alternative method of celebrating Halloween that would not involve close contact with other individuals,” said Dr. Rushton, who is a member of the Pediatric News editorial advisory board.
AAP-recommended Do’s and Don’ts for celebrating Halloween
DO:
- Avoid large gatherings.
- Maintain 6 feet distance.
- Wear cloth masks and wash hands often.
- Use hand sanitizer before and after visiting pumpkin patches and apple orchards.
DON’T:
- Wear painted cloth masks since paints can contain toxins that should not be breathed.
- Use a costume mask unless it has layers of breathable fabric snugly covering mouth and nose.
- Wear cloth mask under costume mask.
- Attend indoor parties or haunted houses.
CDC safety considerations (supplemental to state and local safety laws)
- Assess current cases and overall spread in your community before making any plans.
- Choose outdoor venues or indoor facilities that are well ventilated.
- Consider the length of the event, how many are attending, where they are coming from, and how they behave before and during the event.
- If you are awaiting test results, have COVID-19 symptoms, or have been exposed to COVID-19, stay home.
- If you are at higher risk, avoid large gatherings and limit exposure to anyone you do not live with.
- Make available to others masks, 60% or greater alcohol-based hand sanitizer, and tissues.
- Avoid touching your nose, eyes, and mouth.
- For a complete set of Centers for Disease Control and Prevention COVID safety recommendations go here.
Suggested safe, fun activities
- Use Zoom and other chat programs to share costumes, play games, and watch festive movies.
- Participate in socially distanced outdoor community events at local parks, zoos, etc.
- Attend haunted forests and corn mazes. Maintain more than 6 feet of distance around screaming patrons.
- Decorate pumpkins.
- Cook a Halloween-themed meal.
- If trick-or-treating has been canceled, try a scavenger hunt in the house or yard.
- When handing out treats, wear gloves and mask. Consider prepackaging treat bags. Line up visitors 6 feet apart and discourage gatherings around entranceways.
- Wipe down all goodies received and consider quarantining them for a few days.
- Always wash hands before and after trick-or-treating and when handling treats.
Halloween is fast approaching and retail stores are fully stocked with costumes and candy. Physician dialog is beginning to shift from school access toward how to counsel patients and families on COVID-19 safety around Halloween. advised pediatrician Shelly Vaziri Flais, MD.

Halloween “is going to look very different this year, especially in urban and rural settings, according to Dr. Flais, who is a spokesperson for the American Academy of Pediatrics and assistant professor of clinical pediatrics at Northwestern University, Chicago. The notion that trick-or-treating automatically involves physically distancing is a misconception. Urban celebrations frequently see many people gathering on the streets, and that will be even more likely in a pandemic year when people have been separated for long periods of time.
For pediatricians advising families on COVID-19 safety measures to follow while celebrating Halloween, it’s not going to be a one-size-fits-all approach, said Dr. Flais, who practices pediatrics at Pediatric Health Associates in Naperville, Ill.
The goal for physicians across the board should be “to ensure that we aren’t so cautious that we drive folks to do things that are higher risk,” she said in an interview. “We are now 6-7 months into the pandemic and the public is growing weary of laying low, so it is important for physicians to not recommend safety measures that are too restrictive.”
The balance pediatricians will need to strike in advising their patients is tricky at best. So in dispensing advice, it is important to make sure that it has a benefit to the overall population, cautioned Dr. Flais. Activities such as hosting independently organized, heavily packed indoor gatherings where people are eating, drinking, and not wearing masks is not going to be beneficial for the masses.
“We’re all lucky that we have technology. We’ve gotten used to doing virtual hugs and activities on Zoom,” she said, adding that she has already seen some really creative ideas on social media for enjoying a COVID-conscious Halloween, including a festive candy chute created by an Ohio family that is perfect for distributing candy while minimizing physical contact.
In an AAP press release, Dr. Flais noted that “this is a good time to teach children the importance of protecting not just ourselves but each other.” How we choose to manage our safety and the safety of our children “can have a ripple effect on our family members.” It is possible to make safe, responsible choices when celebrating and still create magical memories for our children.
Francis E. Rushton Jr., MD, of the University of South Carolina, Columbia, said in an interview, “ I certainly support the AAP recommendations. Because of the way COVID-19 virus is spread, I would emphasize with my patients that the No. 1 thing to do is to enforce facial mask wearing while out trick-or-treating.
“I would also err on the side of safety if my child was showing any signs of illness and find an alternative method of celebrating Halloween that would not involve close contact with other individuals,” said Dr. Rushton, who is a member of the Pediatric News editorial advisory board.
AAP-recommended Do’s and Don’ts for celebrating Halloween
DO:
- Avoid large gatherings.
- Maintain 6 feet distance.
- Wear cloth masks and wash hands often.
- Use hand sanitizer before and after visiting pumpkin patches and apple orchards.
DON’T:
- Wear painted cloth masks since paints can contain toxins that should not be breathed.
- Use a costume mask unless it has layers of breathable fabric snugly covering mouth and nose.
- Wear cloth mask under costume mask.
- Attend indoor parties or haunted houses.
CDC safety considerations (supplemental to state and local safety laws)
- Assess current cases and overall spread in your community before making any plans.
- Choose outdoor venues or indoor facilities that are well ventilated.
- Consider the length of the event, how many are attending, where they are coming from, and how they behave before and during the event.
- If you are awaiting test results, have COVID-19 symptoms, or have been exposed to COVID-19, stay home.
- If you are at higher risk, avoid large gatherings and limit exposure to anyone you do not live with.
- Make available to others masks, 60% or greater alcohol-based hand sanitizer, and tissues.
- Avoid touching your nose, eyes, and mouth.
- For a complete set of Centers for Disease Control and Prevention COVID safety recommendations go here.
Suggested safe, fun activities
- Use Zoom and other chat programs to share costumes, play games, and watch festive movies.
- Participate in socially distanced outdoor community events at local parks, zoos, etc.
- Attend haunted forests and corn mazes. Maintain more than 6 feet of distance around screaming patrons.
- Decorate pumpkins.
- Cook a Halloween-themed meal.
- If trick-or-treating has been canceled, try a scavenger hunt in the house or yard.
- When handing out treats, wear gloves and mask. Consider prepackaging treat bags. Line up visitors 6 feet apart and discourage gatherings around entranceways.
- Wipe down all goodies received and consider quarantining them for a few days.
- Always wash hands before and after trick-or-treating and when handling treats.
Prospects and challenges for the upcoming influenza season
The 2020-2021 influenza season is shaping up to be challenging. Its likely concurrence with the ongoing severe acute respiratory syndrome-coronavirus 2 (SARS-coV-2) pandemic (COVID-19) will pose diagnostic and therapeutic dilemmas and could overload the hospital system. But there could also be potential synergies in preventing morbidity and mortality from each disease.
A consistent pattern overthe past few influenza seasons
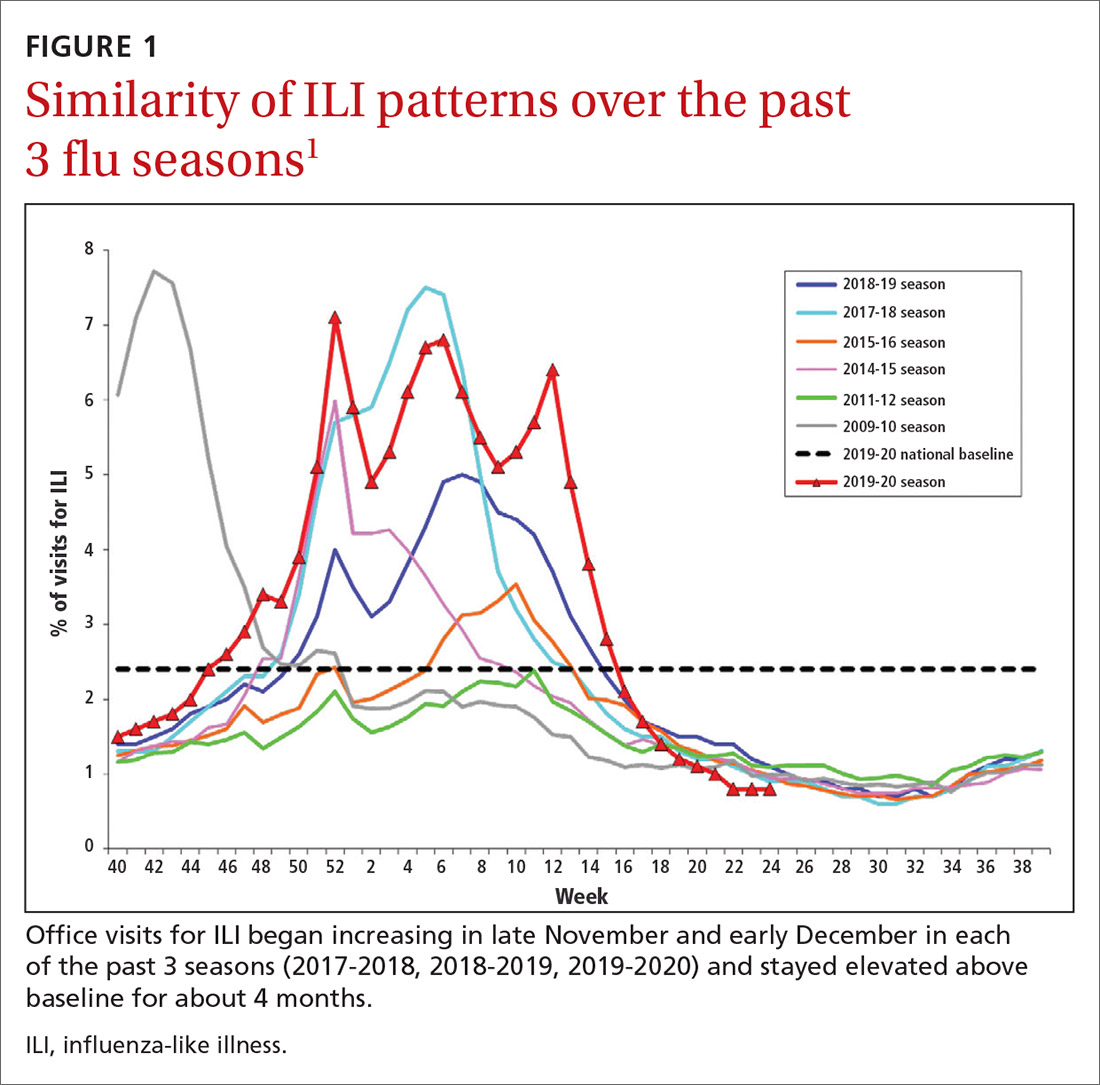
During the 2019-2020 flu season, there were an estimated 410,000 to 740,000 hospitalizations and 24,000 to 62,000 deaths attributed to influenza.1 As seen in FIGURE 1, office visits for influenza-like illness (ILI) began to increase in late November and early December in each of the last 3 years (2017-2018, 2018-2019, 2019-2020) and stayed elevated above baseline for about 4 months each season.1
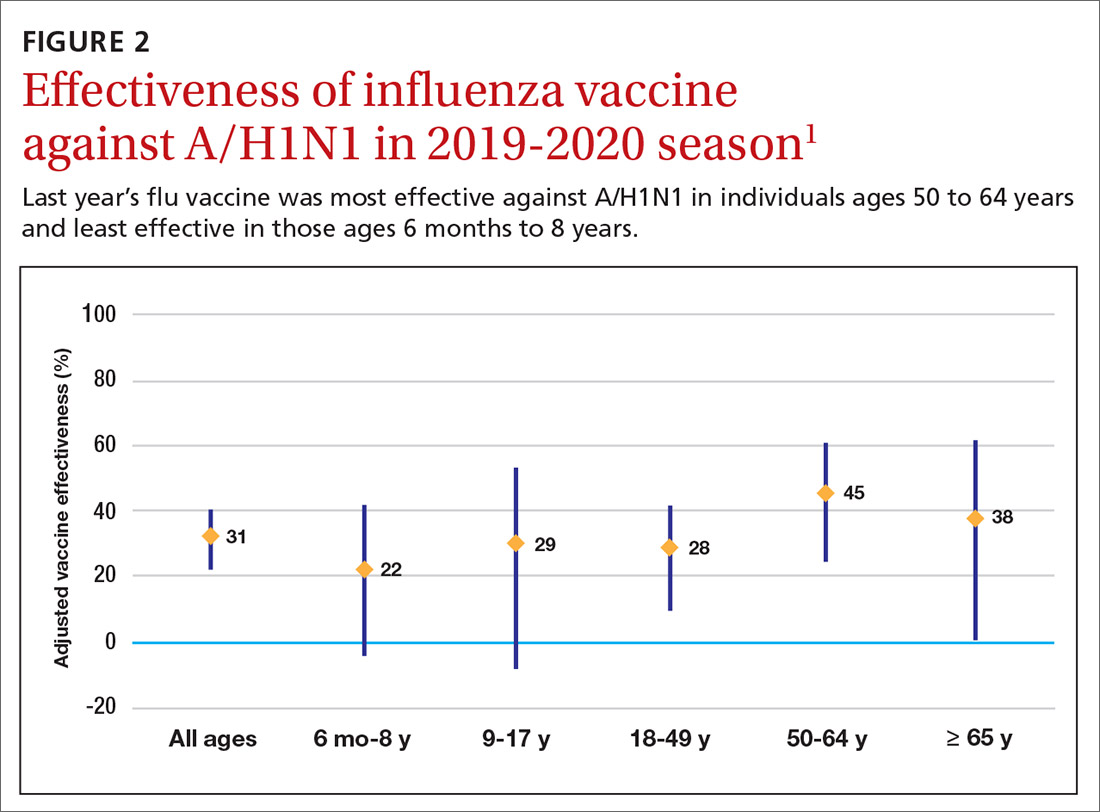
The effectiveness of influenza vaccine during the 2019-2020 season is being estimated using the US Flu Vaccine Effectiveness Network, which has close to 9000 enrollees. Overall, it appears the vaccine was 39% effective against medically attended influenza, with a higher effectiveness against influenza B (44%) than against A/H1N1 (31%). Effectiveness against influenza B was similar in all age groups, but effectiveness against A/H1N1 was highest for those ages 50 to 64 years (45%) and lowest for those ages 6 months through 8 years (22%), although 95% confidence intervals overlapped for all age groups (FIGURE 2). These preliminary effectiveness rates were presented at the summer meeting of the Advisory Committee on Immunization Practices (ACIP).1
Influenza vaccine safety data for 2019-2020 were based on the Vaccine Adverse Event Reporting System (VAERS), a passive surveillance system, and on the Vaccine Safety Datalink (VSD) system, an active surveillance system involving close to 6 million doses administered at VSD sites. No safety concerns were identified for any of the different vaccine types. Both the VAERS and VSD surveillance systems have been described in more detail in a previous Practice Alert.2
Recommendations for 2020-2021
The composition of the influenza vaccines for this year’s flu season will be different for 3 of the 4 antigens: A/H1N1, A/H2N2 and B/Victoria.3 The antigens included in the influenza vaccines each year are decided on in the spring, based on surveillance of circulating strains around the world. The effectiveness of the vaccine each year largely depends on how well the strains included in the vaccine match those circulating in the United States during the influenza season.
The main immunization recommendation for preventing morbidity and mortality from influenza has not changed: All individuals ages 6 months and older without a contraindication should receive an influenza vaccine.4 The Centers for Disease Control and Prevention (CDC) recommends that patients receive the vaccine by the end of October.4 This includes the second dose for those children younger than 9 years who need 2 doses—ie, those who have received fewer than 2 doses of influenza vaccine prior to July 2020. Vaccination should continue through the end of the season for anyone who has not received a 2020-2021 influenza vaccine.
Two new influenza vaccine products are available for use in those ages 65 years and older: Fluzone high-dose quadrivalent and Fluad Quadrivalent (adjuvanted).4 Both of these products were available last year as trivalent options. Currently no specific vaccine product is listed as preferred by ACIP for those ages 65 and older.
Continue to: New vaccine contraindications
New vaccine contraindications. Four medical conditions have been added to the list of contraindications for quadrivalent live, attenuated influenza vaccine (LAIV4): cochlear implant, cerebrospinal fluid leak, asplenia (anatomic and functional), and sickle cell anemia.4 In addition, those who receive LAIV4 should not be prescribed an influenza antiviral until 2 weeks after receiving the vaccine. And the vaccine should not be administered for 48 hours after receipt of oseltamivir or zanamivir, 5 days after peramivir, and 17 days after baloxavir marboxil.4 This is to prevent possible antiviral inactivation of the live attenuated influenza viruses in the vaccine.
For those who have a history of severe allergic reaction to eggs, there are now 2 egg-free options: cell-culture-based inactivated vaccine (ccIIV4) and recombinant influenza vaccine (RIV4).3,4 Urticaria alone is not considered a severe reaction. If neither of these egg-free options is available, a vaccine may still be administered in a medical setting supervised by a provider who is able to manage a severe allergic reaction (which rarely occurs).
All vaccine products available for the upcoming influenza season are listed and described on the CDC Web site, as is a summary of related recommendations.4 Particular attention should be paid to the dose of vaccine administered, as it differs by product for those ages 6 through 35 months of age and those ages 65 years and older.
Use of antiviral medications
Four antiviral medications are now available for treating influenza (3 neuraminidase inhibitors and 1 endonuclease inhibitor), and there are 2 agents for preventing influenza, both neuraminidase inhibitors (TABLE 1).5 The CDC recommends treating with antivirals as soon as possible if individuals with confirmed or suspected influenza require hospitalization; have severe, complicated, or progressive illness; or are at high risk for complications. Use antivirals based on clinical judgment if previously healthy individuals do not have severe complications and are not at increased risk for complications, and only if the medication can be started within 48 hours of symptom onset.
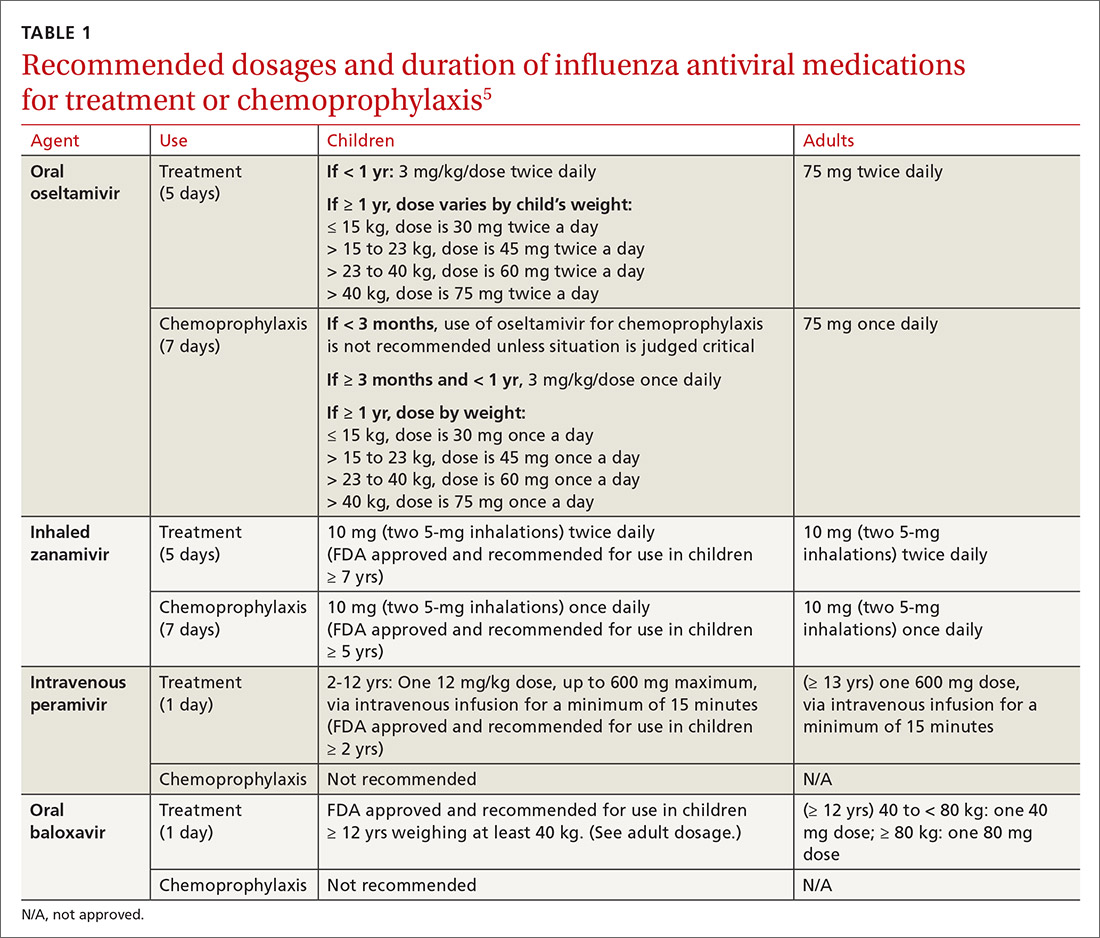
The CDC discourages widespread use of antivirals to prevent influenza, either pre- or postexposure, although it specifies certain situations in which usage would be acceptable (TABLE 2).5 There is some concern that widespread use could lead to the emergence of drug-resistant strains and that using postexposure dosing could lead to suboptimal treatment if influenza infection occurred before the start of prophylaxis. If postexposure antivirals are prescribed, they should be started within 48 hours of exposure and continued for 7 days after the last exposure.

Continue to: A potential perfect storm
A potential perfect storm: Concurrence of influenza and SARS-coV-19
While we have vaccines and antivirals to prevent influenza, and have effective antivirals for treatment, no prevention or treatment options exist for COVID-19, except, possibly, dexamethasone to reduce mortality among those seriously ill.6 The concurrence of influenza and COVID-19 will present unique challenges for the health care system.
Action steps. Keep abreast of the incidences of circulating SARS-coV-19 and influenza viruses in your community. The similar signs and symptoms of these 2 infectious agents will complicate diagnosis. Rapid, or point-of-care, tests for influenza are widely available, but their accuracy varies and not all tests detect both influenza A and B. The CDC lists approved point-of-care tests at www.cdc.gov/flu/professionals/diagnosis/table-ridt.html and advises on how to interpret these test results when influenza is and is not circulating in the community, at www.cdc.gov/flu/professionals/diagnosis/clinician_guidance_ridt.htm.
Clinical practice advice for both conditions should be implemented when any patient presents with ILI:7
- Most patients who are not seriously ill and have no conditions that place them at high risk for adverse outcomes can be treated symptomatically at home.
- Those with ILI should be tested for both influenza virus and SARS-CoV-2 if testing is available. It is possible to be co-infected.
- Sick patients should self-isolate at home for the duration of their symptoms.
- If others live in the house, the sick person should stay in a separate room and wear a mask. Everyone in the house should cover coughs and sneezes (if not wearing a mask), dispose of used tissues in a trash can (rather than leaving them on night stands and countertops), and wash hands frequently.
- All household members should be vaccinated against influenza. Those who are unvaccinated, and those at high risk who have been recently vaccinated, can consider influenza antiviral prophylaxis. If the sick family member is confirmed to have COVID-19, with no co-existing influenza, anti-influenza antiviral prophylaxis may be discontinued.
- Clinical infection control practices should be the same for anyone presenting with ILI.7 Enhanced clinic-based infection control practices to prevent spread of SARS-CoV-2 are listed in TABLE 3.8
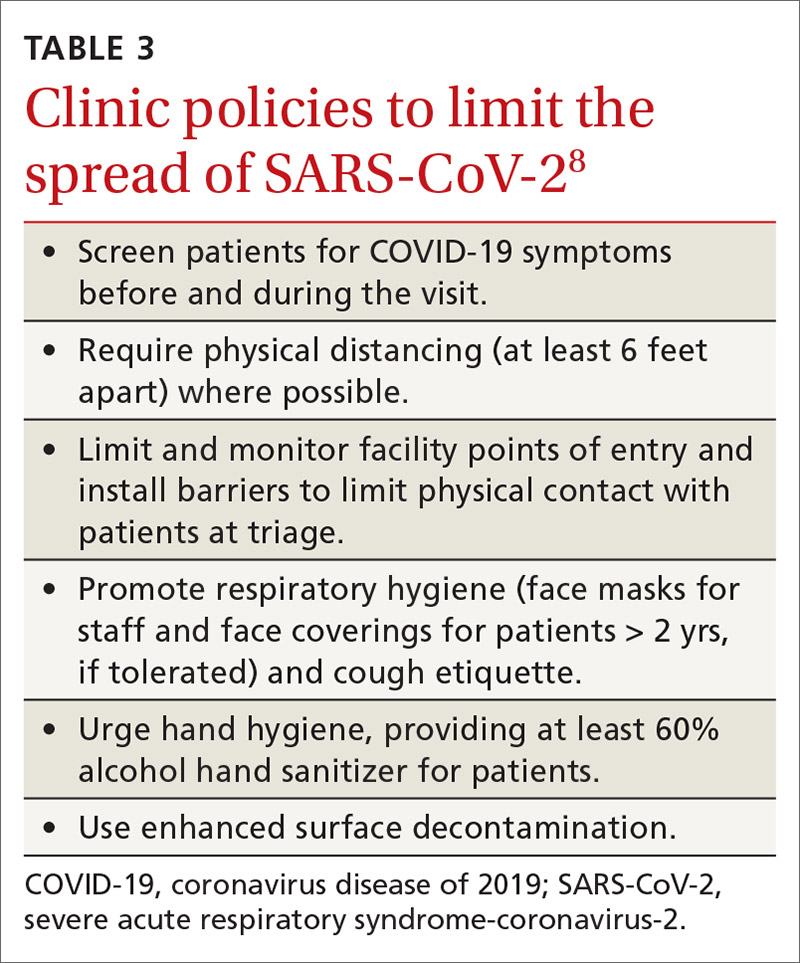
Since there currently are no preventive medications proven to work for COVID-19, the main clinical decision physicians will have to make when a patient presents with ILI is whether to use antivirals to treat those who are at risk for complications based on the result of rapid, on-site influenza testing, or clinical presentation, or both. In this situation, knowledge of which viruses are circulating at high rates in the community could be valuable.
Milder season or perfect storm? The society-wide interventions that have been encouraged (although not mandated everywhere) to prevent community spread of SARS-CoV-2 should help prevent the community spread of influenza as well, and, if adhered to, may lead to a milder influenza season than would otherwise have occurred. However, given the uncertainties, the combination of influenza and coronavirus could present a perfect storm for the health care system and result in higher-than-normal morbidity and mortality from ILI and pneumonia overall.
Continue to: The possibility that one or more vaccines...
The possibility that one or more vaccines to prevent COVID-19 may be available in late 2020 or early 2021 offers hope. However, in current testing, the vaccine is not being given simultaneously with the influenza vaccine. If the potential for adverse interaction exists between the vaccines, it is important that influenza vaccine be given by mid- to late-October to avoid such an interaction if and when the new SARS-CoV-2 vaccine becomes available. Individuals who have symptoms of COVID-19 should not be vaccinated with influenza vaccine until they are considered noninfectious.
Encourage influenza vaccination. The COVID-19 pandemic may make it difficult to achieve desired community influenza vaccine levels because of decreased visits to medical facilities for preventive care, possible lower insurance coverage due to loss of employment, and a decrease in worksite mass vaccination programs. This makes it important for family physicians to encourage and offer influenza vaccines at their clinical sites.
Several evidence-based practices have been shown to improve vaccine uptake. Examples of such practices include patient reminder and recall systems that provide feedback to clinicians about rates of vaccination among patients, and establishing standing orders for vaccine administration that allow other health care providers to assess a patient’s immunization status and administer vaccinations according to a protocol.9 Finally, the CDC provides a video on how to recommend influenza vaccine to those who may be resistant (www.cdc.gov/vaccines/howirecommend/adult-vacc-videos.html).
SIDEBAR
CDC influenza resources
Point-of-care tests that detect both influenza A and B viruses approved by the CDC
www.cdc.gov/flu/professionals/diagnosis/table-ridt.html
Advice on how to interpret the test results
www.cdc.gov/flu/professionals/diagnosis/clinician_guidance_ridt.htm
How to recommend influenza vaccine to reluctant patients
www.cdc.gov/vaccines/howirecommend/adult-vacc-videos.html
CDC, Centers for Disease Control and Prevention.
1. Grohskopf L. Influenza work groups: updates, considerations, and proposed recommendations for the 2020-2021 season. Presented at the ACIP meeting June 24, 2020. www.youtube.com/watch?v=W1SV2DSJsaQ&list=PLvrp9iOILTQb6D9e1YZWpbUvzfptNMKx2&index=8&t=0s. [Time stamp: 1:26:48] Accessed Septemeber 29, 2020.
2. Campos-Outcalt D. Facts to help you keep pace with the vaccine conversation. J Fam Pract. 2019;68:341-346.
3. Grohskopf L, Alyanak E, Broder KR, et al. Prevention and control of seasonal influenza with vaccines: recommendations of the Advisory Committee on Immunization Practices—United States, 2020-21 Influenza Season. MMWR Recomm Rep. 2020;69:1-24.
4. Prevention and Control of Seasonal Influenza with Vaccines: Recommendations of the Advisory Committee on Immunization Practices (ACIP)—United States, 2020-21 Summary of Recommendations. www.cdc.gov/flu/pdf/professionals/acip/acip-2020-21-summary-of-recommendations.pdf. Accessed September 29, 2020.
5. CDC. Influenza antiviral medications: summary for clinicians. www.cdc.gov/flu/professionals/antivirals/summary-clinicians.htm. Accessed September 29, 2020.
6. NIH. COVID-19 treatment guidelines. Corticosteroids. www.covid19treatmentguidelines.nih.gov/immune-based-therapy/immunomodulators/corticosteroids/. Accessed September 29, 2020.
7. CDC. Infection control. www.cdc.gov/infectioncontrol/. Accessed September 29, 2020.
8. CDC. Interim infection prevention and control recommendations for healthcare personnel during the coronavirus disease 2019 (COVID-19) pandemic. www.cdc.gov/coronavirus/2019-ncov/hcp/infection-control.html. Accessed September 29, 2020.
9. HHS. CPSTF findings for increasing vaccination. www.thecommunityguide.org/content/task-force-findings-increasing-vaccination. Accessed September 29, 2020.
The 2020-2021 influenza season is shaping up to be challenging. Its likely concurrence with the ongoing severe acute respiratory syndrome-coronavirus 2 (SARS-coV-2) pandemic (COVID-19) will pose diagnostic and therapeutic dilemmas and could overload the hospital system. But there could also be potential synergies in preventing morbidity and mortality from each disease.
A consistent pattern overthe past few influenza seasons
During the 2019-2020 flu season, there were an estimated 410,000 to 740,000 hospitalizations and 24,000 to 62,000 deaths attributed to influenza.1 As seen in FIGURE 1, office visits for influenza-like illness (ILI) began to increase in late November and early December in each of the last 3 years (2017-2018, 2018-2019, 2019-2020) and stayed elevated above baseline for about 4 months each season.1

The effectiveness of influenza vaccine during the 2019-2020 season is being estimated using the US Flu Vaccine Effectiveness Network, which has close to 9000 enrollees. Overall, it appears the vaccine was 39% effective against medically attended influenza, with a higher effectiveness against influenza B (44%) than against A/H1N1 (31%). Effectiveness against influenza B was similar in all age groups, but effectiveness against A/H1N1 was highest for those ages 50 to 64 years (45%) and lowest for those ages 6 months through 8 years (22%), although 95% confidence intervals overlapped for all age groups (FIGURE 2). These preliminary effectiveness rates were presented at the summer meeting of the Advisory Committee on Immunization Practices (ACIP).1

Influenza vaccine safety data for 2019-2020 were based on the Vaccine Adverse Event Reporting System (VAERS), a passive surveillance system, and on the Vaccine Safety Datalink (VSD) system, an active surveillance system involving close to 6 million doses administered at VSD sites. No safety concerns were identified for any of the different vaccine types. Both the VAERS and VSD surveillance systems have been described in more detail in a previous Practice Alert.2
Recommendations for 2020-2021
The composition of the influenza vaccines for this year’s flu season will be different for 3 of the 4 antigens: A/H1N1, A/H2N2 and B/Victoria.3 The antigens included in the influenza vaccines each year are decided on in the spring, based on surveillance of circulating strains around the world. The effectiveness of the vaccine each year largely depends on how well the strains included in the vaccine match those circulating in the United States during the influenza season.
The main immunization recommendation for preventing morbidity and mortality from influenza has not changed: All individuals ages 6 months and older without a contraindication should receive an influenza vaccine.4 The Centers for Disease Control and Prevention (CDC) recommends that patients receive the vaccine by the end of October.4 This includes the second dose for those children younger than 9 years who need 2 doses—ie, those who have received fewer than 2 doses of influenza vaccine prior to July 2020. Vaccination should continue through the end of the season for anyone who has not received a 2020-2021 influenza vaccine.
Two new influenza vaccine products are available for use in those ages 65 years and older: Fluzone high-dose quadrivalent and Fluad Quadrivalent (adjuvanted).4 Both of these products were available last year as trivalent options. Currently no specific vaccine product is listed as preferred by ACIP for those ages 65 and older.
Continue to: New vaccine contraindications
New vaccine contraindications. Four medical conditions have been added to the list of contraindications for quadrivalent live, attenuated influenza vaccine (LAIV4): cochlear implant, cerebrospinal fluid leak, asplenia (anatomic and functional), and sickle cell anemia.4 In addition, those who receive LAIV4 should not be prescribed an influenza antiviral until 2 weeks after receiving the vaccine. And the vaccine should not be administered for 48 hours after receipt of oseltamivir or zanamivir, 5 days after peramivir, and 17 days after baloxavir marboxil.4 This is to prevent possible antiviral inactivation of the live attenuated influenza viruses in the vaccine.
For those who have a history of severe allergic reaction to eggs, there are now 2 egg-free options: cell-culture-based inactivated vaccine (ccIIV4) and recombinant influenza vaccine (RIV4).3,4 Urticaria alone is not considered a severe reaction. If neither of these egg-free options is available, a vaccine may still be administered in a medical setting supervised by a provider who is able to manage a severe allergic reaction (which rarely occurs).
All vaccine products available for the upcoming influenza season are listed and described on the CDC Web site, as is a summary of related recommendations.4 Particular attention should be paid to the dose of vaccine administered, as it differs by product for those ages 6 through 35 months of age and those ages 65 years and older.
Use of antiviral medications
Four antiviral medications are now available for treating influenza (3 neuraminidase inhibitors and 1 endonuclease inhibitor), and there are 2 agents for preventing influenza, both neuraminidase inhibitors (TABLE 1).5 The CDC recommends treating with antivirals as soon as possible if individuals with confirmed or suspected influenza require hospitalization; have severe, complicated, or progressive illness; or are at high risk for complications. Use antivirals based on clinical judgment if previously healthy individuals do not have severe complications and are not at increased risk for complications, and only if the medication can be started within 48 hours of symptom onset.

The CDC discourages widespread use of antivirals to prevent influenza, either pre- or postexposure, although it specifies certain situations in which usage would be acceptable (TABLE 2).5 There is some concern that widespread use could lead to the emergence of drug-resistant strains and that using postexposure dosing could lead to suboptimal treatment if influenza infection occurred before the start of prophylaxis. If postexposure antivirals are prescribed, they should be started within 48 hours of exposure and continued for 7 days after the last exposure.

Continue to: A potential perfect storm
A potential perfect storm: Concurrence of influenza and SARS-coV-19
While we have vaccines and antivirals to prevent influenza, and have effective antivirals for treatment, no prevention or treatment options exist for COVID-19, except, possibly, dexamethasone to reduce mortality among those seriously ill.6 The concurrence of influenza and COVID-19 will present unique challenges for the health care system.
Action steps. Keep abreast of the incidences of circulating SARS-coV-19 and influenza viruses in your community. The similar signs and symptoms of these 2 infectious agents will complicate diagnosis. Rapid, or point-of-care, tests for influenza are widely available, but their accuracy varies and not all tests detect both influenza A and B. The CDC lists approved point-of-care tests at www.cdc.gov/flu/professionals/diagnosis/table-ridt.html and advises on how to interpret these test results when influenza is and is not circulating in the community, at www.cdc.gov/flu/professionals/diagnosis/clinician_guidance_ridt.htm.
Clinical practice advice for both conditions should be implemented when any patient presents with ILI:7
- Most patients who are not seriously ill and have no conditions that place them at high risk for adverse outcomes can be treated symptomatically at home.
- Those with ILI should be tested for both influenza virus and SARS-CoV-2 if testing is available. It is possible to be co-infected.
- Sick patients should self-isolate at home for the duration of their symptoms.
- If others live in the house, the sick person should stay in a separate room and wear a mask. Everyone in the house should cover coughs and sneezes (if not wearing a mask), dispose of used tissues in a trash can (rather than leaving them on night stands and countertops), and wash hands frequently.
- All household members should be vaccinated against influenza. Those who are unvaccinated, and those at high risk who have been recently vaccinated, can consider influenza antiviral prophylaxis. If the sick family member is confirmed to have COVID-19, with no co-existing influenza, anti-influenza antiviral prophylaxis may be discontinued.
- Clinical infection control practices should be the same for anyone presenting with ILI.7 Enhanced clinic-based infection control practices to prevent spread of SARS-CoV-2 are listed in TABLE 3.8

Since there currently are no preventive medications proven to work for COVID-19, the main clinical decision physicians will have to make when a patient presents with ILI is whether to use antivirals to treat those who are at risk for complications based on the result of rapid, on-site influenza testing, or clinical presentation, or both. In this situation, knowledge of which viruses are circulating at high rates in the community could be valuable.
Milder season or perfect storm? The society-wide interventions that have been encouraged (although not mandated everywhere) to prevent community spread of SARS-CoV-2 should help prevent the community spread of influenza as well, and, if adhered to, may lead to a milder influenza season than would otherwise have occurred. However, given the uncertainties, the combination of influenza and coronavirus could present a perfect storm for the health care system and result in higher-than-normal morbidity and mortality from ILI and pneumonia overall.
Continue to: The possibility that one or more vaccines...
The possibility that one or more vaccines to prevent COVID-19 may be available in late 2020 or early 2021 offers hope. However, in current testing, the vaccine is not being given simultaneously with the influenza vaccine. If the potential for adverse interaction exists between the vaccines, it is important that influenza vaccine be given by mid- to late-October to avoid such an interaction if and when the new SARS-CoV-2 vaccine becomes available. Individuals who have symptoms of COVID-19 should not be vaccinated with influenza vaccine until they are considered noninfectious.
Encourage influenza vaccination. The COVID-19 pandemic may make it difficult to achieve desired community influenza vaccine levels because of decreased visits to medical facilities for preventive care, possible lower insurance coverage due to loss of employment, and a decrease in worksite mass vaccination programs. This makes it important for family physicians to encourage and offer influenza vaccines at their clinical sites.
Several evidence-based practices have been shown to improve vaccine uptake. Examples of such practices include patient reminder and recall systems that provide feedback to clinicians about rates of vaccination among patients, and establishing standing orders for vaccine administration that allow other health care providers to assess a patient’s immunization status and administer vaccinations according to a protocol.9 Finally, the CDC provides a video on how to recommend influenza vaccine to those who may be resistant (www.cdc.gov/vaccines/howirecommend/adult-vacc-videos.html).
SIDEBAR
CDC influenza resources
Point-of-care tests that detect both influenza A and B viruses approved by the CDC
www.cdc.gov/flu/professionals/diagnosis/table-ridt.html
Advice on how to interpret the test results
www.cdc.gov/flu/professionals/diagnosis/clinician_guidance_ridt.htm
How to recommend influenza vaccine to reluctant patients
www.cdc.gov/vaccines/howirecommend/adult-vacc-videos.html
CDC, Centers for Disease Control and Prevention.
The 2020-2021 influenza season is shaping up to be challenging. Its likely concurrence with the ongoing severe acute respiratory syndrome-coronavirus 2 (SARS-coV-2) pandemic (COVID-19) will pose diagnostic and therapeutic dilemmas and could overload the hospital system. But there could also be potential synergies in preventing morbidity and mortality from each disease.
A consistent pattern overthe past few influenza seasons
During the 2019-2020 flu season, there were an estimated 410,000 to 740,000 hospitalizations and 24,000 to 62,000 deaths attributed to influenza.1 As seen in FIGURE 1, office visits for influenza-like illness (ILI) began to increase in late November and early December in each of the last 3 years (2017-2018, 2018-2019, 2019-2020) and stayed elevated above baseline for about 4 months each season.1

The effectiveness of influenza vaccine during the 2019-2020 season is being estimated using the US Flu Vaccine Effectiveness Network, which has close to 9000 enrollees. Overall, it appears the vaccine was 39% effective against medically attended influenza, with a higher effectiveness against influenza B (44%) than against A/H1N1 (31%). Effectiveness against influenza B was similar in all age groups, but effectiveness against A/H1N1 was highest for those ages 50 to 64 years (45%) and lowest for those ages 6 months through 8 years (22%), although 95% confidence intervals overlapped for all age groups (FIGURE 2). These preliminary effectiveness rates were presented at the summer meeting of the Advisory Committee on Immunization Practices (ACIP).1

Influenza vaccine safety data for 2019-2020 were based on the Vaccine Adverse Event Reporting System (VAERS), a passive surveillance system, and on the Vaccine Safety Datalink (VSD) system, an active surveillance system involving close to 6 million doses administered at VSD sites. No safety concerns were identified for any of the different vaccine types. Both the VAERS and VSD surveillance systems have been described in more detail in a previous Practice Alert.2
Recommendations for 2020-2021
The composition of the influenza vaccines for this year’s flu season will be different for 3 of the 4 antigens: A/H1N1, A/H2N2 and B/Victoria.3 The antigens included in the influenza vaccines each year are decided on in the spring, based on surveillance of circulating strains around the world. The effectiveness of the vaccine each year largely depends on how well the strains included in the vaccine match those circulating in the United States during the influenza season.
The main immunization recommendation for preventing morbidity and mortality from influenza has not changed: All individuals ages 6 months and older without a contraindication should receive an influenza vaccine.4 The Centers for Disease Control and Prevention (CDC) recommends that patients receive the vaccine by the end of October.4 This includes the second dose for those children younger than 9 years who need 2 doses—ie, those who have received fewer than 2 doses of influenza vaccine prior to July 2020. Vaccination should continue through the end of the season for anyone who has not received a 2020-2021 influenza vaccine.
Two new influenza vaccine products are available for use in those ages 65 years and older: Fluzone high-dose quadrivalent and Fluad Quadrivalent (adjuvanted).4 Both of these products were available last year as trivalent options. Currently no specific vaccine product is listed as preferred by ACIP for those ages 65 and older.
Continue to: New vaccine contraindications
New vaccine contraindications. Four medical conditions have been added to the list of contraindications for quadrivalent live, attenuated influenza vaccine (LAIV4): cochlear implant, cerebrospinal fluid leak, asplenia (anatomic and functional), and sickle cell anemia.4 In addition, those who receive LAIV4 should not be prescribed an influenza antiviral until 2 weeks after receiving the vaccine. And the vaccine should not be administered for 48 hours after receipt of oseltamivir or zanamivir, 5 days after peramivir, and 17 days after baloxavir marboxil.4 This is to prevent possible antiviral inactivation of the live attenuated influenza viruses in the vaccine.
For those who have a history of severe allergic reaction to eggs, there are now 2 egg-free options: cell-culture-based inactivated vaccine (ccIIV4) and recombinant influenza vaccine (RIV4).3,4 Urticaria alone is not considered a severe reaction. If neither of these egg-free options is available, a vaccine may still be administered in a medical setting supervised by a provider who is able to manage a severe allergic reaction (which rarely occurs).
All vaccine products available for the upcoming influenza season are listed and described on the CDC Web site, as is a summary of related recommendations.4 Particular attention should be paid to the dose of vaccine administered, as it differs by product for those ages 6 through 35 months of age and those ages 65 years and older.
Use of antiviral medications
Four antiviral medications are now available for treating influenza (3 neuraminidase inhibitors and 1 endonuclease inhibitor), and there are 2 agents for preventing influenza, both neuraminidase inhibitors (TABLE 1).5 The CDC recommends treating with antivirals as soon as possible if individuals with confirmed or suspected influenza require hospitalization; have severe, complicated, or progressive illness; or are at high risk for complications. Use antivirals based on clinical judgment if previously healthy individuals do not have severe complications and are not at increased risk for complications, and only if the medication can be started within 48 hours of symptom onset.

The CDC discourages widespread use of antivirals to prevent influenza, either pre- or postexposure, although it specifies certain situations in which usage would be acceptable (TABLE 2).5 There is some concern that widespread use could lead to the emergence of drug-resistant strains and that using postexposure dosing could lead to suboptimal treatment if influenza infection occurred before the start of prophylaxis. If postexposure antivirals are prescribed, they should be started within 48 hours of exposure and continued for 7 days after the last exposure.

Continue to: A potential perfect storm
A potential perfect storm: Concurrence of influenza and SARS-coV-19
While we have vaccines and antivirals to prevent influenza, and have effective antivirals for treatment, no prevention or treatment options exist for COVID-19, except, possibly, dexamethasone to reduce mortality among those seriously ill.6 The concurrence of influenza and COVID-19 will present unique challenges for the health care system.
Action steps. Keep abreast of the incidences of circulating SARS-coV-19 and influenza viruses in your community. The similar signs and symptoms of these 2 infectious agents will complicate diagnosis. Rapid, or point-of-care, tests for influenza are widely available, but their accuracy varies and not all tests detect both influenza A and B. The CDC lists approved point-of-care tests at www.cdc.gov/flu/professionals/diagnosis/table-ridt.html and advises on how to interpret these test results when influenza is and is not circulating in the community, at www.cdc.gov/flu/professionals/diagnosis/clinician_guidance_ridt.htm.
Clinical practice advice for both conditions should be implemented when any patient presents with ILI:7
- Most patients who are not seriously ill and have no conditions that place them at high risk for adverse outcomes can be treated symptomatically at home.
- Those with ILI should be tested for both influenza virus and SARS-CoV-2 if testing is available. It is possible to be co-infected.
- Sick patients should self-isolate at home for the duration of their symptoms.
- If others live in the house, the sick person should stay in a separate room and wear a mask. Everyone in the house should cover coughs and sneezes (if not wearing a mask), dispose of used tissues in a trash can (rather than leaving them on night stands and countertops), and wash hands frequently.
- All household members should be vaccinated against influenza. Those who are unvaccinated, and those at high risk who have been recently vaccinated, can consider influenza antiviral prophylaxis. If the sick family member is confirmed to have COVID-19, with no co-existing influenza, anti-influenza antiviral prophylaxis may be discontinued.
- Clinical infection control practices should be the same for anyone presenting with ILI.7 Enhanced clinic-based infection control practices to prevent spread of SARS-CoV-2 are listed in TABLE 3.8

Since there currently are no preventive medications proven to work for COVID-19, the main clinical decision physicians will have to make when a patient presents with ILI is whether to use antivirals to treat those who are at risk for complications based on the result of rapid, on-site influenza testing, or clinical presentation, or both. In this situation, knowledge of which viruses are circulating at high rates in the community could be valuable.
Milder season or perfect storm? The society-wide interventions that have been encouraged (although not mandated everywhere) to prevent community spread of SARS-CoV-2 should help prevent the community spread of influenza as well, and, if adhered to, may lead to a milder influenza season than would otherwise have occurred. However, given the uncertainties, the combination of influenza and coronavirus could present a perfect storm for the health care system and result in higher-than-normal morbidity and mortality from ILI and pneumonia overall.
Continue to: The possibility that one or more vaccines...
The possibility that one or more vaccines to prevent COVID-19 may be available in late 2020 or early 2021 offers hope. However, in current testing, the vaccine is not being given simultaneously with the influenza vaccine. If the potential for adverse interaction exists between the vaccines, it is important that influenza vaccine be given by mid- to late-October to avoid such an interaction if and when the new SARS-CoV-2 vaccine becomes available. Individuals who have symptoms of COVID-19 should not be vaccinated with influenza vaccine until they are considered noninfectious.
Encourage influenza vaccination. The COVID-19 pandemic may make it difficult to achieve desired community influenza vaccine levels because of decreased visits to medical facilities for preventive care, possible lower insurance coverage due to loss of employment, and a decrease in worksite mass vaccination programs. This makes it important for family physicians to encourage and offer influenza vaccines at their clinical sites.
Several evidence-based practices have been shown to improve vaccine uptake. Examples of such practices include patient reminder and recall systems that provide feedback to clinicians about rates of vaccination among patients, and establishing standing orders for vaccine administration that allow other health care providers to assess a patient’s immunization status and administer vaccinations according to a protocol.9 Finally, the CDC provides a video on how to recommend influenza vaccine to those who may be resistant (www.cdc.gov/vaccines/howirecommend/adult-vacc-videos.html).
SIDEBAR
CDC influenza resources
Point-of-care tests that detect both influenza A and B viruses approved by the CDC
www.cdc.gov/flu/professionals/diagnosis/table-ridt.html
Advice on how to interpret the test results
www.cdc.gov/flu/professionals/diagnosis/clinician_guidance_ridt.htm
How to recommend influenza vaccine to reluctant patients
www.cdc.gov/vaccines/howirecommend/adult-vacc-videos.html
CDC, Centers for Disease Control and Prevention.
1. Grohskopf L. Influenza work groups: updates, considerations, and proposed recommendations for the 2020-2021 season. Presented at the ACIP meeting June 24, 2020. www.youtube.com/watch?v=W1SV2DSJsaQ&list=PLvrp9iOILTQb6D9e1YZWpbUvzfptNMKx2&index=8&t=0s. [Time stamp: 1:26:48] Accessed Septemeber 29, 2020.
2. Campos-Outcalt D. Facts to help you keep pace with the vaccine conversation. J Fam Pract. 2019;68:341-346.
3. Grohskopf L, Alyanak E, Broder KR, et al. Prevention and control of seasonal influenza with vaccines: recommendations of the Advisory Committee on Immunization Practices—United States, 2020-21 Influenza Season. MMWR Recomm Rep. 2020;69:1-24.
4. Prevention and Control of Seasonal Influenza with Vaccines: Recommendations of the Advisory Committee on Immunization Practices (ACIP)—United States, 2020-21 Summary of Recommendations. www.cdc.gov/flu/pdf/professionals/acip/acip-2020-21-summary-of-recommendations.pdf. Accessed September 29, 2020.
5. CDC. Influenza antiviral medications: summary for clinicians. www.cdc.gov/flu/professionals/antivirals/summary-clinicians.htm. Accessed September 29, 2020.
6. NIH. COVID-19 treatment guidelines. Corticosteroids. www.covid19treatmentguidelines.nih.gov/immune-based-therapy/immunomodulators/corticosteroids/. Accessed September 29, 2020.
7. CDC. Infection control. www.cdc.gov/infectioncontrol/. Accessed September 29, 2020.
8. CDC. Interim infection prevention and control recommendations for healthcare personnel during the coronavirus disease 2019 (COVID-19) pandemic. www.cdc.gov/coronavirus/2019-ncov/hcp/infection-control.html. Accessed September 29, 2020.
9. HHS. CPSTF findings for increasing vaccination. www.thecommunityguide.org/content/task-force-findings-increasing-vaccination. Accessed September 29, 2020.
1. Grohskopf L. Influenza work groups: updates, considerations, and proposed recommendations for the 2020-2021 season. Presented at the ACIP meeting June 24, 2020. www.youtube.com/watch?v=W1SV2DSJsaQ&list=PLvrp9iOILTQb6D9e1YZWpbUvzfptNMKx2&index=8&t=0s. [Time stamp: 1:26:48] Accessed Septemeber 29, 2020.
2. Campos-Outcalt D. Facts to help you keep pace with the vaccine conversation. J Fam Pract. 2019;68:341-346.
3. Grohskopf L, Alyanak E, Broder KR, et al. Prevention and control of seasonal influenza with vaccines: recommendations of the Advisory Committee on Immunization Practices—United States, 2020-21 Influenza Season. MMWR Recomm Rep. 2020;69:1-24.
4. Prevention and Control of Seasonal Influenza with Vaccines: Recommendations of the Advisory Committee on Immunization Practices (ACIP)—United States, 2020-21 Summary of Recommendations. www.cdc.gov/flu/pdf/professionals/acip/acip-2020-21-summary-of-recommendations.pdf. Accessed September 29, 2020.
5. CDC. Influenza antiviral medications: summary for clinicians. www.cdc.gov/flu/professionals/antivirals/summary-clinicians.htm. Accessed September 29, 2020.
6. NIH. COVID-19 treatment guidelines. Corticosteroids. www.covid19treatmentguidelines.nih.gov/immune-based-therapy/immunomodulators/corticosteroids/. Accessed September 29, 2020.
7. CDC. Infection control. www.cdc.gov/infectioncontrol/. Accessed September 29, 2020.
8. CDC. Interim infection prevention and control recommendations for healthcare personnel during the coronavirus disease 2019 (COVID-19) pandemic. www.cdc.gov/coronavirus/2019-ncov/hcp/infection-control.html. Accessed September 29, 2020.
9. HHS. CPSTF findings for increasing vaccination. www.thecommunityguide.org/content/task-force-findings-increasing-vaccination. Accessed September 29, 2020.
Returning to competition
As we continue to stumble around trying to find our way out of the COVID-19 pandemic, it has become clear that the journey has been a never-ending continuum of exercises in risk/benefit assessment. The population always has sorted itself into a bell-shaped curve from those who are risk averse to those who revel in risk taking. And, of course, with a paucity of facts on which we can base our assessment of risk, the discussion often shifts to our gut feelings about the benefits.
When faced with the question of when it is time for children to return to in-person schooling, there seems to be reasonably good agreement about the benefits of face-to-face learning. The level of risk is still to be determined.
When it comes to the issue of when to return to competitive school sports, the risks are equally indeterminate but there is less agreement on the benefits. This lack of uniformity reflects a long-standing dichotomy between those parents and students with a passion for competitive sports and those who see them as nonessential. This existential tug-of-war has gone on in almost every school system I am aware of when the school budget comes up for a vote.
The debate about a return to competitive sports on a collegiate and professional level unfortunately is colored by enormous revenues from media contracts, which means that high school and middle schools can’t look to what are essentially businesses for guidance. The delay created confusion, fluctuating angst and disappointment, but the end product made some sense. Volleyball (indoor) and football were indefinitely delayed. Heavy breathing between competitors separated by a couple of feet and protected only by a flimsy net or helmet cage seems like a risk not worth taking – at least until we have more information.
Other sports were allowed to start with restrictions based on existing social distancing mandates which include no locker rooms and no fans. Some rules such as no throw-ins for soccer didn’t make sense given what we are learning about the virus. But, for the most part, the compromises should result in a chance to reap the benefits of competition for the students whose families are willing to expose them to the yet to be fully determined risks.
There has been some grumbling from parents who see the no-fans mandate as a step too far. Until we know more about the risk of group gatherings outdoors, having no fans, including parents and grandparents, makes sense. In fact, to me it is a step long overdue and a rare sliver of silver lining to the pandemic. Competitive youth sports are for the kids. They are not meant to be entertainment events. Too often children are exposed to parental pressure (voiced and unvoiced) about their “performance” on the field. Neither my younger sister nor I can remember our parents going to any of my away football games in high school or any of my lacrosse games in college. I never felt the loss.
Will I miss watching my grandchildren compete? Of course I will miss it badly. However, giving kids some space to learn and enjoy the competition for itself in an atmosphere free of parental over-involvement will be a breath of fresh air. Something we need badly during this pandemic.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Email him at pdnews@mdedge.com.
As we continue to stumble around trying to find our way out of the COVID-19 pandemic, it has become clear that the journey has been a never-ending continuum of exercises in risk/benefit assessment. The population always has sorted itself into a bell-shaped curve from those who are risk averse to those who revel in risk taking. And, of course, with a paucity of facts on which we can base our assessment of risk, the discussion often shifts to our gut feelings about the benefits.
When faced with the question of when it is time for children to return to in-person schooling, there seems to be reasonably good agreement about the benefits of face-to-face learning. The level of risk is still to be determined.
When it comes to the issue of when to return to competitive school sports, the risks are equally indeterminate but there is less agreement on the benefits. This lack of uniformity reflects a long-standing dichotomy between those parents and students with a passion for competitive sports and those who see them as nonessential. This existential tug-of-war has gone on in almost every school system I am aware of when the school budget comes up for a vote.
The debate about a return to competitive sports on a collegiate and professional level unfortunately is colored by enormous revenues from media contracts, which means that high school and middle schools can’t look to what are essentially businesses for guidance. The delay created confusion, fluctuating angst and disappointment, but the end product made some sense. Volleyball (indoor) and football were indefinitely delayed. Heavy breathing between competitors separated by a couple of feet and protected only by a flimsy net or helmet cage seems like a risk not worth taking – at least until we have more information.
Other sports were allowed to start with restrictions based on existing social distancing mandates which include no locker rooms and no fans. Some rules such as no throw-ins for soccer didn’t make sense given what we are learning about the virus. But, for the most part, the compromises should result in a chance to reap the benefits of competition for the students whose families are willing to expose them to the yet to be fully determined risks.
There has been some grumbling from parents who see the no-fans mandate as a step too far. Until we know more about the risk of group gatherings outdoors, having no fans, including parents and grandparents, makes sense. In fact, to me it is a step long overdue and a rare sliver of silver lining to the pandemic. Competitive youth sports are for the kids. They are not meant to be entertainment events. Too often children are exposed to parental pressure (voiced and unvoiced) about their “performance” on the field. Neither my younger sister nor I can remember our parents going to any of my away football games in high school or any of my lacrosse games in college. I never felt the loss.
Will I miss watching my grandchildren compete? Of course I will miss it badly. However, giving kids some space to learn and enjoy the competition for itself in an atmosphere free of parental over-involvement will be a breath of fresh air. Something we need badly during this pandemic.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Email him at pdnews@mdedge.com.
As we continue to stumble around trying to find our way out of the COVID-19 pandemic, it has become clear that the journey has been a never-ending continuum of exercises in risk/benefit assessment. The population always has sorted itself into a bell-shaped curve from those who are risk averse to those who revel in risk taking. And, of course, with a paucity of facts on which we can base our assessment of risk, the discussion often shifts to our gut feelings about the benefits.
When faced with the question of when it is time for children to return to in-person schooling, there seems to be reasonably good agreement about the benefits of face-to-face learning. The level of risk is still to be determined.
When it comes to the issue of when to return to competitive school sports, the risks are equally indeterminate but there is less agreement on the benefits. This lack of uniformity reflects a long-standing dichotomy between those parents and students with a passion for competitive sports and those who see them as nonessential. This existential tug-of-war has gone on in almost every school system I am aware of when the school budget comes up for a vote.
The debate about a return to competitive sports on a collegiate and professional level unfortunately is colored by enormous revenues from media contracts, which means that high school and middle schools can’t look to what are essentially businesses for guidance. The delay created confusion, fluctuating angst and disappointment, but the end product made some sense. Volleyball (indoor) and football were indefinitely delayed. Heavy breathing between competitors separated by a couple of feet and protected only by a flimsy net or helmet cage seems like a risk not worth taking – at least until we have more information.
Other sports were allowed to start with restrictions based on existing social distancing mandates which include no locker rooms and no fans. Some rules such as no throw-ins for soccer didn’t make sense given what we are learning about the virus. But, for the most part, the compromises should result in a chance to reap the benefits of competition for the students whose families are willing to expose them to the yet to be fully determined risks.
There has been some grumbling from parents who see the no-fans mandate as a step too far. Until we know more about the risk of group gatherings outdoors, having no fans, including parents and grandparents, makes sense. In fact, to me it is a step long overdue and a rare sliver of silver lining to the pandemic. Competitive youth sports are for the kids. They are not meant to be entertainment events. Too often children are exposed to parental pressure (voiced and unvoiced) about their “performance” on the field. Neither my younger sister nor I can remember our parents going to any of my away football games in high school or any of my lacrosse games in college. I never felt the loss.
Will I miss watching my grandchildren compete? Of course I will miss it badly. However, giving kids some space to learn and enjoy the competition for itself in an atmosphere free of parental over-involvement will be a breath of fresh air. Something we need badly during this pandemic.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Email him at pdnews@mdedge.com.
Remdesivir effective, well-tolerated in final trial report
Drug beats placebo across multiple endpoints in COVID-19 patients
In May 2020, remdesivir received Food and Drug Administration approval for emergency treatment of severe COVID-19 on the basis of a preliminary report on this trial. In August 2020, the FDA expanded the indication to include all hospitalized adult and pediatric patients with suspected or laboratory-confirmed COVID-19 infection irrespective of severity.
“Our findings were consistent with the findings of the preliminary report: a 10-day course of remdesivir was superior to placebo in the treatment of hospitalized patients with COVID-19,” reported a team of investigators led by John H. Beigel, MD, of the Division of Microbiology and Infectious Diseases at the National Institute of Allergy and Infectious Diseases, in the New England Journal of Medicine.
The drug’s broadened indication was not based on the ACTT-1 trial, according to Dr. Beigel. “Other data have demonstrated that remdesivir shortens recovery in patients with lower acuity. In our study, evidence of pneumonia was an enrollment requirement,” he explained in an interview.
In the newly published final ACTT-1 data, the median time to recovery was 10 days for those on active therapy versus 15 days for those randomized to placebo. With a rate ratio of 1.29 (P less than .001), this translated to a recovery that was about one third faster.
In this final report, remdesivir’s significant advantage over placebo regarding the trial’s primary endpoint was reinforced by efficacy on multiple secondary endpoints.
This benefits on multiple secondary endpoints included a 50% greater odds ratio (OR, 1.5; 95% CI, 1.2-1.9) of significant clinical improvement by day 15 after adjustment for baseline severity, a shorter initial length of hospital stay (12 vs. 17 days) and fewer days on oxygen supplementation (13 vs. 21 days) for the subgroup of patients on oxygen at enrollment.
Although the numerically lower mortality in the remdesivir arm (6.75 vs. 11.9%) did not reach statistical significance, Dr. Beigel said, “mortality was moving in the same direction as the other key endpoints.”
According to the study investigators, the types of rates of adverse events on remdesivir, which inhibits viral replication, “were generally similar in the remdesivir and placebo groups.”
In ACTT-1, 1,062 patients were randomized to remdesivir (200 mg loading dose followed by 100 mg daily for up to 9 days) or placebo. Patients were enrolled at study sites in North America, Europe, and Asia.
The data of ACTT-1 confirm a benefit from remdesivir in hospitalized COVID-19 patients with severe disease, but Dr. Beigel said he agrees with the current FDA indication that supports treatment in any hospitalized COVID-19 patient.
“We saw bigger benefits in patients with more severe infections. The benefits are not as large in patients with mild disease, but I think remdesivir should be considered in any hospitalized patient,” Dr. Beigel said.
This point of view is shared.
“I would give this drug to anyone in the hospital infected with COVID-19 assuming there was an ample supply and no need for rationing,” said Donna E. Sweet, MD, professor of internal medicine, University of Kansas, Wichita. She noted that this study has implications for hospital and hospital staff, as well as for patients.
“This type of reduction in recovery time means a reduction in potential exposures to hospital staff, a reduced need for PPE [personal protective equipment], and it will free up beds in the ICU [intensive care unit],” said Dr. Sweet, who also serves as an editorial advisory board member for Internal Medicine News.
An infectious disease specialist at the University of Minnesota also considers remdesivir to have an important role for conserving resources that deserves emphasis.
The reduction in time to recovery “is of benefit to the health system by maintaining hospital bed capacity,” said David R. Boulware, MD, professor of medicine at the University of Minnesota, Minneapolis.
According to his reading of the available data, including those from ACTT-1, the benefit appears to be greatest in those with a moderate degree of illness, which he defined as “sick enough to be hospitalized and require oxygen, yet not severely sick [and] requiring a ventilator or [extracorporeal membrane oxygenation].”
This does not preclude a benefit in those with more severe or milder disease, but patients with mild disease “are likely to recover regardless – or despite – whatever therapy they receive,” he said.
Dr. Beigel, the principal investigator of this trial, reports no potential conflicts of interest.
SOURCE: Beigel JH et al. N Engl J Med. 2020 Oct 8. doi: 10.1056/NEJMoa2007764.
Drug beats placebo across multiple endpoints in COVID-19 patients
Drug beats placebo across multiple endpoints in COVID-19 patients
In May 2020, remdesivir received Food and Drug Administration approval for emergency treatment of severe COVID-19 on the basis of a preliminary report on this trial. In August 2020, the FDA expanded the indication to include all hospitalized adult and pediatric patients with suspected or laboratory-confirmed COVID-19 infection irrespective of severity.
“Our findings were consistent with the findings of the preliminary report: a 10-day course of remdesivir was superior to placebo in the treatment of hospitalized patients with COVID-19,” reported a team of investigators led by John H. Beigel, MD, of the Division of Microbiology and Infectious Diseases at the National Institute of Allergy and Infectious Diseases, in the New England Journal of Medicine.
The drug’s broadened indication was not based on the ACTT-1 trial, according to Dr. Beigel. “Other data have demonstrated that remdesivir shortens recovery in patients with lower acuity. In our study, evidence of pneumonia was an enrollment requirement,” he explained in an interview.
In the newly published final ACTT-1 data, the median time to recovery was 10 days for those on active therapy versus 15 days for those randomized to placebo. With a rate ratio of 1.29 (P less than .001), this translated to a recovery that was about one third faster.
In this final report, remdesivir’s significant advantage over placebo regarding the trial’s primary endpoint was reinforced by efficacy on multiple secondary endpoints.
This benefits on multiple secondary endpoints included a 50% greater odds ratio (OR, 1.5; 95% CI, 1.2-1.9) of significant clinical improvement by day 15 after adjustment for baseline severity, a shorter initial length of hospital stay (12 vs. 17 days) and fewer days on oxygen supplementation (13 vs. 21 days) for the subgroup of patients on oxygen at enrollment.
Although the numerically lower mortality in the remdesivir arm (6.75 vs. 11.9%) did not reach statistical significance, Dr. Beigel said, “mortality was moving in the same direction as the other key endpoints.”
According to the study investigators, the types of rates of adverse events on remdesivir, which inhibits viral replication, “were generally similar in the remdesivir and placebo groups.”
In ACTT-1, 1,062 patients were randomized to remdesivir (200 mg loading dose followed by 100 mg daily for up to 9 days) or placebo. Patients were enrolled at study sites in North America, Europe, and Asia.
The data of ACTT-1 confirm a benefit from remdesivir in hospitalized COVID-19 patients with severe disease, but Dr. Beigel said he agrees with the current FDA indication that supports treatment in any hospitalized COVID-19 patient.
“We saw bigger benefits in patients with more severe infections. The benefits are not as large in patients with mild disease, but I think remdesivir should be considered in any hospitalized patient,” Dr. Beigel said.
This point of view is shared.
“I would give this drug to anyone in the hospital infected with COVID-19 assuming there was an ample supply and no need for rationing,” said Donna E. Sweet, MD, professor of internal medicine, University of Kansas, Wichita. She noted that this study has implications for hospital and hospital staff, as well as for patients.
“This type of reduction in recovery time means a reduction in potential exposures to hospital staff, a reduced need for PPE [personal protective equipment], and it will free up beds in the ICU [intensive care unit],” said Dr. Sweet, who also serves as an editorial advisory board member for Internal Medicine News.
An infectious disease specialist at the University of Minnesota also considers remdesivir to have an important role for conserving resources that deserves emphasis.
The reduction in time to recovery “is of benefit to the health system by maintaining hospital bed capacity,” said David R. Boulware, MD, professor of medicine at the University of Minnesota, Minneapolis.
According to his reading of the available data, including those from ACTT-1, the benefit appears to be greatest in those with a moderate degree of illness, which he defined as “sick enough to be hospitalized and require oxygen, yet not severely sick [and] requiring a ventilator or [extracorporeal membrane oxygenation].”
This does not preclude a benefit in those with more severe or milder disease, but patients with mild disease “are likely to recover regardless – or despite – whatever therapy they receive,” he said.
Dr. Beigel, the principal investigator of this trial, reports no potential conflicts of interest.
SOURCE: Beigel JH et al. N Engl J Med. 2020 Oct 8. doi: 10.1056/NEJMoa2007764.
In May 2020, remdesivir received Food and Drug Administration approval for emergency treatment of severe COVID-19 on the basis of a preliminary report on this trial. In August 2020, the FDA expanded the indication to include all hospitalized adult and pediatric patients with suspected or laboratory-confirmed COVID-19 infection irrespective of severity.
“Our findings were consistent with the findings of the preliminary report: a 10-day course of remdesivir was superior to placebo in the treatment of hospitalized patients with COVID-19,” reported a team of investigators led by John H. Beigel, MD, of the Division of Microbiology and Infectious Diseases at the National Institute of Allergy and Infectious Diseases, in the New England Journal of Medicine.
The drug’s broadened indication was not based on the ACTT-1 trial, according to Dr. Beigel. “Other data have demonstrated that remdesivir shortens recovery in patients with lower acuity. In our study, evidence of pneumonia was an enrollment requirement,” he explained in an interview.
In the newly published final ACTT-1 data, the median time to recovery was 10 days for those on active therapy versus 15 days for those randomized to placebo. With a rate ratio of 1.29 (P less than .001), this translated to a recovery that was about one third faster.
In this final report, remdesivir’s significant advantage over placebo regarding the trial’s primary endpoint was reinforced by efficacy on multiple secondary endpoints.
This benefits on multiple secondary endpoints included a 50% greater odds ratio (OR, 1.5; 95% CI, 1.2-1.9) of significant clinical improvement by day 15 after adjustment for baseline severity, a shorter initial length of hospital stay (12 vs. 17 days) and fewer days on oxygen supplementation (13 vs. 21 days) for the subgroup of patients on oxygen at enrollment.
Although the numerically lower mortality in the remdesivir arm (6.75 vs. 11.9%) did not reach statistical significance, Dr. Beigel said, “mortality was moving in the same direction as the other key endpoints.”
According to the study investigators, the types of rates of adverse events on remdesivir, which inhibits viral replication, “were generally similar in the remdesivir and placebo groups.”
In ACTT-1, 1,062 patients were randomized to remdesivir (200 mg loading dose followed by 100 mg daily for up to 9 days) or placebo. Patients were enrolled at study sites in North America, Europe, and Asia.
The data of ACTT-1 confirm a benefit from remdesivir in hospitalized COVID-19 patients with severe disease, but Dr. Beigel said he agrees with the current FDA indication that supports treatment in any hospitalized COVID-19 patient.
“We saw bigger benefits in patients with more severe infections. The benefits are not as large in patients with mild disease, but I think remdesivir should be considered in any hospitalized patient,” Dr. Beigel said.
This point of view is shared.
“I would give this drug to anyone in the hospital infected with COVID-19 assuming there was an ample supply and no need for rationing,” said Donna E. Sweet, MD, professor of internal medicine, University of Kansas, Wichita. She noted that this study has implications for hospital and hospital staff, as well as for patients.
“This type of reduction in recovery time means a reduction in potential exposures to hospital staff, a reduced need for PPE [personal protective equipment], and it will free up beds in the ICU [intensive care unit],” said Dr. Sweet, who also serves as an editorial advisory board member for Internal Medicine News.
An infectious disease specialist at the University of Minnesota also considers remdesivir to have an important role for conserving resources that deserves emphasis.
The reduction in time to recovery “is of benefit to the health system by maintaining hospital bed capacity,” said David R. Boulware, MD, professor of medicine at the University of Minnesota, Minneapolis.
According to his reading of the available data, including those from ACTT-1, the benefit appears to be greatest in those with a moderate degree of illness, which he defined as “sick enough to be hospitalized and require oxygen, yet not severely sick [and] requiring a ventilator or [extracorporeal membrane oxygenation].”
This does not preclude a benefit in those with more severe or milder disease, but patients with mild disease “are likely to recover regardless – or despite – whatever therapy they receive,” he said.
Dr. Beigel, the principal investigator of this trial, reports no potential conflicts of interest.
SOURCE: Beigel JH et al. N Engl J Med. 2020 Oct 8. doi: 10.1056/NEJMoa2007764.
More data on impact of corticosteroids on COVID-19 mortality in patients with COPD
, a study of almost 1 million individuals in the United Kingdom has shown.
Patients with chronic obstructive pulmonary disease or asthma who used ICS on a regular basis were more likely to die from COVID-19 than COPD or asthma patients who were prescribed non-ICS therapies, reported co-lead author Anna Schultze, PhD, of London School of Hygiene & Tropical Medicine and colleagues.
Of note, the increased risk of death among ICS users likely stemmed from greater severity of preexisting chronic respiratory conditions, instead of directly from ICS usage, which has little apparent impact on COVID-19 mortality, the investigators wrote in Lancet Respiratory Medicine.
These findings conflict with a hypothesis proposed early in the pandemic: that ICS may protect individuals from SARS-CoV-2 infection and poor outcomes with COVID-19.
According to Megan Conroy, MD, of the department of internal medicine at the Ohio State University Wexner Medical Center, Columbus, this hypothesis was based on some unexpected epidemiological findings.
“In general, we tend to think people with underlying lung disease – like COPD or asthma – to be at higher risk for severe forms of lower respiratory tract infections,” Dr. Conroy said. “Somewhat surprisingly, early data in the pandemic showed patients with COPD and asthma [were] underrepresented [among patients with COVID] when compared to the prevalence of these diseases in the population.”
This raised the possibility of an incidental protective effect from regular ICS therapy, which “had some strong theoretic pathophysiologic basis,” Dr. Conroy said, referring to research that demonstrated ICS-mediated downregulation of SARS-CoV-2 entry receptors ACE2 and TMPRSS2.
Dr. Schultze and colleagues noted that investigators for two ongoing randomized controlled trials (NCT04331054, NCT04330586) are studying ICS as an intervention for COVID-19; but neither trial includes individuals already taking ICS for chronic respiratory disease.
The present observational study therefore aimed to assess mortality risk within this population. Data were drawn from electronic health records and a U.K. national mortality database, with follow-up ranging from March 1 to May 6, 2020. Eligibility required a relevant prescription within 4 months of first follow-up. In the COPD group, patients were prescribed a long-acting beta agonist plus a long-acting muscarinic antagonist (LABA–LAMA), LABA alone, LABA plus ICS, LABA–LAMA plus ICS, or ICS alone (if prescribed LABA within 4 months).
In the asthma group, patients received low/medium-dose ICS, high-dose ICS, or a short-acting beta agonist (SABA) alone. Patients with COPD were at least 35 years of age, while those with asthma were 18 years or older. Hazard ratios were adjusted for a variety of covariates, including respiratory disease–exacerbation history, age, sex, body mass index, hypertension, diabetes, and others.
These eligibility criteria returned 148,557 patients with COPD and 818,490 with asthma.
Patients with COPD who were prescribed ICS plus LABA-LAMA or ICS plus LABA had an increased risk of COVID-19-related death, compared with those who did not receive ICS (adjusted hazard ratio, 1.39; 95% confidence interval, 1.10-1.76). Separate analyses of patients who received a triple combination (LABA–LAMA plus ICS) versus those who took a dual combination (LABA plus ICS) showed that triple-combination therapy was significantly associated with increased COVID-19-related mortality (aHR, 1.43; 95% CI, 1.12-1.83), while dual-combination therapy was less so (aHR, 1.29; 95% CI, 0.96-1.74). Non–COVID-19–related mortality was significantly increased for all COPD patients who were prescribed ICS, with or without adjustment for covariates.
Asthma patients prescribed high-dose ICS instead of SABA alone had a slightly greater risk of COVID-19–related death, based on an adjusted hazard ratio of 1.55 (95% CI, 1.10-2.18). Those with asthma who received low/medium–dose ICS demonstrated a slight trend toward increased mortality risk, but this was not significant (aHR, 1.14; 95% CI, 0.85-1.54). ICS usage in the asthma group was not linked with a significant increase in non–COVID-19–related death.
“In summary, we found no evidence of a beneficial effect of regular ICS use among people with COPD and asthma on COVID-19–related mortality,” the investigators concluded.
In agreement with the investigators, Dr. Conroy said that the increased mortality rate among ICS users should not be misconstrued as a medication-related risk.
“While the study found that those with COPD or asthma taking ICS and high-dose ICS were at an increased risk of death, this could easily be explained by the likelihood that those are the patients who are more likely to have more severe underlying lung disease,” Dr. Conroy said. “While this observational study did attempt to control for exacerbation history, the ability to do so by electronic health records data is certainly imperfect.”
With this in mind, patients with chronic respiratory disease should be encouraged to adhere to their usual treatment regimen, Dr. Conroy added.
“There isn’t evidence to increase or decrease medications just because of the pandemic,” she said. “A patient with asthma or COPD should continue to take the medications that are needed to achieve good control of their lung disease.”
The study was funded by the U.K. Medical Research Council. The investigators reported additional relationships with the Wellcome Trust, the Good Thinking Foundation, the Laura and John Arnold Foundation, and others. Dr. Conroy reported no conflicts of interest.
SOURCE: Schultze A et al. Lancet Respir Med. 2020 Sep 24. doi: 10.1016/ S2213-2600(20)30415-X.
, a study of almost 1 million individuals in the United Kingdom has shown.
Patients with chronic obstructive pulmonary disease or asthma who used ICS on a regular basis were more likely to die from COVID-19 than COPD or asthma patients who were prescribed non-ICS therapies, reported co-lead author Anna Schultze, PhD, of London School of Hygiene & Tropical Medicine and colleagues.
Of note, the increased risk of death among ICS users likely stemmed from greater severity of preexisting chronic respiratory conditions, instead of directly from ICS usage, which has little apparent impact on COVID-19 mortality, the investigators wrote in Lancet Respiratory Medicine.
These findings conflict with a hypothesis proposed early in the pandemic: that ICS may protect individuals from SARS-CoV-2 infection and poor outcomes with COVID-19.
According to Megan Conroy, MD, of the department of internal medicine at the Ohio State University Wexner Medical Center, Columbus, this hypothesis was based on some unexpected epidemiological findings.
“In general, we tend to think people with underlying lung disease – like COPD or asthma – to be at higher risk for severe forms of lower respiratory tract infections,” Dr. Conroy said. “Somewhat surprisingly, early data in the pandemic showed patients with COPD and asthma [were] underrepresented [among patients with COVID] when compared to the prevalence of these diseases in the population.”
This raised the possibility of an incidental protective effect from regular ICS therapy, which “had some strong theoretic pathophysiologic basis,” Dr. Conroy said, referring to research that demonstrated ICS-mediated downregulation of SARS-CoV-2 entry receptors ACE2 and TMPRSS2.
Dr. Schultze and colleagues noted that investigators for two ongoing randomized controlled trials (NCT04331054, NCT04330586) are studying ICS as an intervention for COVID-19; but neither trial includes individuals already taking ICS for chronic respiratory disease.
The present observational study therefore aimed to assess mortality risk within this population. Data were drawn from electronic health records and a U.K. national mortality database, with follow-up ranging from March 1 to May 6, 2020. Eligibility required a relevant prescription within 4 months of first follow-up. In the COPD group, patients were prescribed a long-acting beta agonist plus a long-acting muscarinic antagonist (LABA–LAMA), LABA alone, LABA plus ICS, LABA–LAMA plus ICS, or ICS alone (if prescribed LABA within 4 months).
In the asthma group, patients received low/medium-dose ICS, high-dose ICS, or a short-acting beta agonist (SABA) alone. Patients with COPD were at least 35 years of age, while those with asthma were 18 years or older. Hazard ratios were adjusted for a variety of covariates, including respiratory disease–exacerbation history, age, sex, body mass index, hypertension, diabetes, and others.
These eligibility criteria returned 148,557 patients with COPD and 818,490 with asthma.
Patients with COPD who were prescribed ICS plus LABA-LAMA or ICS plus LABA had an increased risk of COVID-19-related death, compared with those who did not receive ICS (adjusted hazard ratio, 1.39; 95% confidence interval, 1.10-1.76). Separate analyses of patients who received a triple combination (LABA–LAMA plus ICS) versus those who took a dual combination (LABA plus ICS) showed that triple-combination therapy was significantly associated with increased COVID-19-related mortality (aHR, 1.43; 95% CI, 1.12-1.83), while dual-combination therapy was less so (aHR, 1.29; 95% CI, 0.96-1.74). Non–COVID-19–related mortality was significantly increased for all COPD patients who were prescribed ICS, with or without adjustment for covariates.
Asthma patients prescribed high-dose ICS instead of SABA alone had a slightly greater risk of COVID-19–related death, based on an adjusted hazard ratio of 1.55 (95% CI, 1.10-2.18). Those with asthma who received low/medium–dose ICS demonstrated a slight trend toward increased mortality risk, but this was not significant (aHR, 1.14; 95% CI, 0.85-1.54). ICS usage in the asthma group was not linked with a significant increase in non–COVID-19–related death.
“In summary, we found no evidence of a beneficial effect of regular ICS use among people with COPD and asthma on COVID-19–related mortality,” the investigators concluded.
In agreement with the investigators, Dr. Conroy said that the increased mortality rate among ICS users should not be misconstrued as a medication-related risk.
“While the study found that those with COPD or asthma taking ICS and high-dose ICS were at an increased risk of death, this could easily be explained by the likelihood that those are the patients who are more likely to have more severe underlying lung disease,” Dr. Conroy said. “While this observational study did attempt to control for exacerbation history, the ability to do so by electronic health records data is certainly imperfect.”
With this in mind, patients with chronic respiratory disease should be encouraged to adhere to their usual treatment regimen, Dr. Conroy added.
“There isn’t evidence to increase or decrease medications just because of the pandemic,” she said. “A patient with asthma or COPD should continue to take the medications that are needed to achieve good control of their lung disease.”
The study was funded by the U.K. Medical Research Council. The investigators reported additional relationships with the Wellcome Trust, the Good Thinking Foundation, the Laura and John Arnold Foundation, and others. Dr. Conroy reported no conflicts of interest.
SOURCE: Schultze A et al. Lancet Respir Med. 2020 Sep 24. doi: 10.1016/ S2213-2600(20)30415-X.
, a study of almost 1 million individuals in the United Kingdom has shown.
Patients with chronic obstructive pulmonary disease or asthma who used ICS on a regular basis were more likely to die from COVID-19 than COPD or asthma patients who were prescribed non-ICS therapies, reported co-lead author Anna Schultze, PhD, of London School of Hygiene & Tropical Medicine and colleagues.
Of note, the increased risk of death among ICS users likely stemmed from greater severity of preexisting chronic respiratory conditions, instead of directly from ICS usage, which has little apparent impact on COVID-19 mortality, the investigators wrote in Lancet Respiratory Medicine.
These findings conflict with a hypothesis proposed early in the pandemic: that ICS may protect individuals from SARS-CoV-2 infection and poor outcomes with COVID-19.
According to Megan Conroy, MD, of the department of internal medicine at the Ohio State University Wexner Medical Center, Columbus, this hypothesis was based on some unexpected epidemiological findings.
“In general, we tend to think people with underlying lung disease – like COPD or asthma – to be at higher risk for severe forms of lower respiratory tract infections,” Dr. Conroy said. “Somewhat surprisingly, early data in the pandemic showed patients with COPD and asthma [were] underrepresented [among patients with COVID] when compared to the prevalence of these diseases in the population.”
This raised the possibility of an incidental protective effect from regular ICS therapy, which “had some strong theoretic pathophysiologic basis,” Dr. Conroy said, referring to research that demonstrated ICS-mediated downregulation of SARS-CoV-2 entry receptors ACE2 and TMPRSS2.
Dr. Schultze and colleagues noted that investigators for two ongoing randomized controlled trials (NCT04331054, NCT04330586) are studying ICS as an intervention for COVID-19; but neither trial includes individuals already taking ICS for chronic respiratory disease.
The present observational study therefore aimed to assess mortality risk within this population. Data were drawn from electronic health records and a U.K. national mortality database, with follow-up ranging from March 1 to May 6, 2020. Eligibility required a relevant prescription within 4 months of first follow-up. In the COPD group, patients were prescribed a long-acting beta agonist plus a long-acting muscarinic antagonist (LABA–LAMA), LABA alone, LABA plus ICS, LABA–LAMA plus ICS, or ICS alone (if prescribed LABA within 4 months).
In the asthma group, patients received low/medium-dose ICS, high-dose ICS, or a short-acting beta agonist (SABA) alone. Patients with COPD were at least 35 years of age, while those with asthma were 18 years or older. Hazard ratios were adjusted for a variety of covariates, including respiratory disease–exacerbation history, age, sex, body mass index, hypertension, diabetes, and others.
These eligibility criteria returned 148,557 patients with COPD and 818,490 with asthma.
Patients with COPD who were prescribed ICS plus LABA-LAMA or ICS plus LABA had an increased risk of COVID-19-related death, compared with those who did not receive ICS (adjusted hazard ratio, 1.39; 95% confidence interval, 1.10-1.76). Separate analyses of patients who received a triple combination (LABA–LAMA plus ICS) versus those who took a dual combination (LABA plus ICS) showed that triple-combination therapy was significantly associated with increased COVID-19-related mortality (aHR, 1.43; 95% CI, 1.12-1.83), while dual-combination therapy was less so (aHR, 1.29; 95% CI, 0.96-1.74). Non–COVID-19–related mortality was significantly increased for all COPD patients who were prescribed ICS, with or without adjustment for covariates.
Asthma patients prescribed high-dose ICS instead of SABA alone had a slightly greater risk of COVID-19–related death, based on an adjusted hazard ratio of 1.55 (95% CI, 1.10-2.18). Those with asthma who received low/medium–dose ICS demonstrated a slight trend toward increased mortality risk, but this was not significant (aHR, 1.14; 95% CI, 0.85-1.54). ICS usage in the asthma group was not linked with a significant increase in non–COVID-19–related death.
“In summary, we found no evidence of a beneficial effect of regular ICS use among people with COPD and asthma on COVID-19–related mortality,” the investigators concluded.
In agreement with the investigators, Dr. Conroy said that the increased mortality rate among ICS users should not be misconstrued as a medication-related risk.
“While the study found that those with COPD or asthma taking ICS and high-dose ICS were at an increased risk of death, this could easily be explained by the likelihood that those are the patients who are more likely to have more severe underlying lung disease,” Dr. Conroy said. “While this observational study did attempt to control for exacerbation history, the ability to do so by electronic health records data is certainly imperfect.”
With this in mind, patients with chronic respiratory disease should be encouraged to adhere to their usual treatment regimen, Dr. Conroy added.
“There isn’t evidence to increase or decrease medications just because of the pandemic,” she said. “A patient with asthma or COPD should continue to take the medications that are needed to achieve good control of their lung disease.”
The study was funded by the U.K. Medical Research Council. The investigators reported additional relationships with the Wellcome Trust, the Good Thinking Foundation, the Laura and John Arnold Foundation, and others. Dr. Conroy reported no conflicts of interest.
SOURCE: Schultze A et al. Lancet Respir Med. 2020 Sep 24. doi: 10.1016/ S2213-2600(20)30415-X.
FROM LANCET RESPIRATORY MEDICINE
COVID-19 vaccine hesitancy ‘somewhat understandable,’ expert says
“I worry that vaccines are going to be sold like magic powder that we sprinkle across the land and make the virus go away,” Paul Offit, MD, said at the virtual American Academy of Pediatrics (AAP) 2020 National Conference. “That’s not true.”
according to Dr. Offit, director of the Vaccine Education Center and an attending physician in the Division of Infectious Diseases at Children’s Hospital of Philadelphia.
“I think we can get a vaccine that’s 75%-80% effective at preventing mild to moderate disease, but that means one of every four people can still get moderate to severe disease,” Dr. Offit continued.
And that’s if there is high uptake of the vaccine, which may not be the case. Recent polls have suggested there is considerable concern about the pending vaccines.
“It’s somewhat understandable,” Dr. Offitt acknowledged, especially given the “frightening” language used to describe vaccine development. Terms such as “warp speed” may suggest that haste might trump safety considerations. Before COVID-19, the fastest vaccine ever developed was for mumps, he said, with the virus isolated in 1963 and a commercial product available in 1967.
Addressing hesitancy in clinics
In a wide-ranging livestream plenary presentation, Dr. Offit, coinventor of a rotavirus vaccine, shed light on SARS-CoV-2 vaccine development and his impressions of vaccine hesitancy among patients and families. He also offered advice for how to reassure those skeptical of the safety and efficacy of any SARS-COV-2 vaccine, given the accelerated development process.
With more than 180 different vaccines in various stages of investigation, Dr. Offit called the effort to develop COVID-19 vaccines “unprecedented.” Part of that is a result of governments relieving pharmaceutical companies of much of the typical financial risk – which often climbs to hundreds of millions of dollars – by underwriting the costs of vaccine development to battle the pandemic-inducing virus, he said.
But this very swiftness is also stoking antivaccine sentiment. Dr. Offit, part of vaccine advisory groups for the National Institutes of Health and U.S. Food and Drug Administration, cited recent research reporting nearly half of American adults definitely or probably would not get a COVID-19 vaccine if it were available today.
“One way you convince skeptics is with data presented in a clear, compassionate, and compelling way,” he said.
“The other group is vaccine cynics, who are basically conspiracy theorists who believe pharmaceutical companies control the world, the government, the medical establishment. I think there’s no talking them down from this.”
Numerous strategies are being used in COVID-19 vaccine development, he noted, including messenger RNA, DNA, viral vectors, purified protein, and whole killed virus. Dr. Offit believes any candidates approved for distribution will likely be in the range of 75% effective at preventing mild to moderate symptoms.
But clinicians should be ready to face immediate questions of safety. “Even if this vaccination is given to 20,000 [trial participants] safely, that’s not 20 million,” Dr. Offit said. “Anyone could reasonably ask questions about if it causes rare, serious side effects.
“The good news is, there are systems in place,” such as adverse event reporting systems, to identify rare events, even those that occur in one in a million vaccine recipients. Reminding patients of that continued surveillance can be reassuring.
Another reassuring point is that COVID-19 vaccine trial participants have included people from many diverse populations, he said. But children, notably absent so far, should be added to trials immediately, Dr. Offit contends.
“This is going to be important when you consider strategies to get children universally back into school,” he said, which is a “critical issue” from both learning and wellness standpoints. “It breaks my heart that we’ve been unable to do this when other countries have.”
Transparency will be paramount
While presenting data transparently to patients is key in helping them accept COVID-19 vaccination, Dr. Offit said, he also believes “telling stories” can be just as effective, if not more so. When the varicella vaccine was approved in 1995, he said, the “uptake the first few years was pretty miserable” until public service messaging emphasized that some children die from chickenpox.
“Fear works,” he said. “You always worry about pushback of something being oversold, but hopefully we’re scared enough about this virus” to convince people that vaccination is wise. “I do think personal stories carry weight on both sides,” Dr. Offit said.
Mark Sawyer, MD, of University of California San Diego School of Medicine and Rady Children’s Hospital in San Diego, California, said Offit’s presentation offered important takeaways for clinicians about how to broach the topic of COVID-19 vaccination with patients and families.
“We need to communicate clearly and transparently to patients about what we do and don’t know” about the vaccines, Dr. Sawyer said in an interview. “We will know if they have common side effects, but we will not know about very rare side effects until we have used the vaccines for a while.
“We will know how well the vaccine works over the short-term, but we won’t know over the long term,” added Dr. Sawyer, a member of the AAP Committee on Infectious Diseases.
“We can reassure the community that SARS-CoV-2 vaccines are being evaluated in trials in the same way and with the same thoroughness as other vaccines have been,” he said. “That should give people confidence that shortcuts are not being taken with regard to safety and effectiveness evaluations.”
Dr. Offit and Dr. Sawyer have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
“I worry that vaccines are going to be sold like magic powder that we sprinkle across the land and make the virus go away,” Paul Offit, MD, said at the virtual American Academy of Pediatrics (AAP) 2020 National Conference. “That’s not true.”
according to Dr. Offit, director of the Vaccine Education Center and an attending physician in the Division of Infectious Diseases at Children’s Hospital of Philadelphia.
“I think we can get a vaccine that’s 75%-80% effective at preventing mild to moderate disease, but that means one of every four people can still get moderate to severe disease,” Dr. Offit continued.
And that’s if there is high uptake of the vaccine, which may not be the case. Recent polls have suggested there is considerable concern about the pending vaccines.
“It’s somewhat understandable,” Dr. Offitt acknowledged, especially given the “frightening” language used to describe vaccine development. Terms such as “warp speed” may suggest that haste might trump safety considerations. Before COVID-19, the fastest vaccine ever developed was for mumps, he said, with the virus isolated in 1963 and a commercial product available in 1967.
Addressing hesitancy in clinics
In a wide-ranging livestream plenary presentation, Dr. Offit, coinventor of a rotavirus vaccine, shed light on SARS-CoV-2 vaccine development and his impressions of vaccine hesitancy among patients and families. He also offered advice for how to reassure those skeptical of the safety and efficacy of any SARS-COV-2 vaccine, given the accelerated development process.
With more than 180 different vaccines in various stages of investigation, Dr. Offit called the effort to develop COVID-19 vaccines “unprecedented.” Part of that is a result of governments relieving pharmaceutical companies of much of the typical financial risk – which often climbs to hundreds of millions of dollars – by underwriting the costs of vaccine development to battle the pandemic-inducing virus, he said.
But this very swiftness is also stoking antivaccine sentiment. Dr. Offit, part of vaccine advisory groups for the National Institutes of Health and U.S. Food and Drug Administration, cited recent research reporting nearly half of American adults definitely or probably would not get a COVID-19 vaccine if it were available today.
“One way you convince skeptics is with data presented in a clear, compassionate, and compelling way,” he said.
“The other group is vaccine cynics, who are basically conspiracy theorists who believe pharmaceutical companies control the world, the government, the medical establishment. I think there’s no talking them down from this.”
Numerous strategies are being used in COVID-19 vaccine development, he noted, including messenger RNA, DNA, viral vectors, purified protein, and whole killed virus. Dr. Offit believes any candidates approved for distribution will likely be in the range of 75% effective at preventing mild to moderate symptoms.
But clinicians should be ready to face immediate questions of safety. “Even if this vaccination is given to 20,000 [trial participants] safely, that’s not 20 million,” Dr. Offit said. “Anyone could reasonably ask questions about if it causes rare, serious side effects.
“The good news is, there are systems in place,” such as adverse event reporting systems, to identify rare events, even those that occur in one in a million vaccine recipients. Reminding patients of that continued surveillance can be reassuring.
Another reassuring point is that COVID-19 vaccine trial participants have included people from many diverse populations, he said. But children, notably absent so far, should be added to trials immediately, Dr. Offit contends.
“This is going to be important when you consider strategies to get children universally back into school,” he said, which is a “critical issue” from both learning and wellness standpoints. “It breaks my heart that we’ve been unable to do this when other countries have.”
Transparency will be paramount
While presenting data transparently to patients is key in helping them accept COVID-19 vaccination, Dr. Offit said, he also believes “telling stories” can be just as effective, if not more so. When the varicella vaccine was approved in 1995, he said, the “uptake the first few years was pretty miserable” until public service messaging emphasized that some children die from chickenpox.
“Fear works,” he said. “You always worry about pushback of something being oversold, but hopefully we’re scared enough about this virus” to convince people that vaccination is wise. “I do think personal stories carry weight on both sides,” Dr. Offit said.
Mark Sawyer, MD, of University of California San Diego School of Medicine and Rady Children’s Hospital in San Diego, California, said Offit’s presentation offered important takeaways for clinicians about how to broach the topic of COVID-19 vaccination with patients and families.
“We need to communicate clearly and transparently to patients about what we do and don’t know” about the vaccines, Dr. Sawyer said in an interview. “We will know if they have common side effects, but we will not know about very rare side effects until we have used the vaccines for a while.
“We will know how well the vaccine works over the short-term, but we won’t know over the long term,” added Dr. Sawyer, a member of the AAP Committee on Infectious Diseases.
“We can reassure the community that SARS-CoV-2 vaccines are being evaluated in trials in the same way and with the same thoroughness as other vaccines have been,” he said. “That should give people confidence that shortcuts are not being taken with regard to safety and effectiveness evaluations.”
Dr. Offit and Dr. Sawyer have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
“I worry that vaccines are going to be sold like magic powder that we sprinkle across the land and make the virus go away,” Paul Offit, MD, said at the virtual American Academy of Pediatrics (AAP) 2020 National Conference. “That’s not true.”
according to Dr. Offit, director of the Vaccine Education Center and an attending physician in the Division of Infectious Diseases at Children’s Hospital of Philadelphia.
“I think we can get a vaccine that’s 75%-80% effective at preventing mild to moderate disease, but that means one of every four people can still get moderate to severe disease,” Dr. Offit continued.
And that’s if there is high uptake of the vaccine, which may not be the case. Recent polls have suggested there is considerable concern about the pending vaccines.
“It’s somewhat understandable,” Dr. Offitt acknowledged, especially given the “frightening” language used to describe vaccine development. Terms such as “warp speed” may suggest that haste might trump safety considerations. Before COVID-19, the fastest vaccine ever developed was for mumps, he said, with the virus isolated in 1963 and a commercial product available in 1967.
Addressing hesitancy in clinics
In a wide-ranging livestream plenary presentation, Dr. Offit, coinventor of a rotavirus vaccine, shed light on SARS-CoV-2 vaccine development and his impressions of vaccine hesitancy among patients and families. He also offered advice for how to reassure those skeptical of the safety and efficacy of any SARS-COV-2 vaccine, given the accelerated development process.
With more than 180 different vaccines in various stages of investigation, Dr. Offit called the effort to develop COVID-19 vaccines “unprecedented.” Part of that is a result of governments relieving pharmaceutical companies of much of the typical financial risk – which often climbs to hundreds of millions of dollars – by underwriting the costs of vaccine development to battle the pandemic-inducing virus, he said.
But this very swiftness is also stoking antivaccine sentiment. Dr. Offit, part of vaccine advisory groups for the National Institutes of Health and U.S. Food and Drug Administration, cited recent research reporting nearly half of American adults definitely or probably would not get a COVID-19 vaccine if it were available today.
“One way you convince skeptics is with data presented in a clear, compassionate, and compelling way,” he said.
“The other group is vaccine cynics, who are basically conspiracy theorists who believe pharmaceutical companies control the world, the government, the medical establishment. I think there’s no talking them down from this.”
Numerous strategies are being used in COVID-19 vaccine development, he noted, including messenger RNA, DNA, viral vectors, purified protein, and whole killed virus. Dr. Offit believes any candidates approved for distribution will likely be in the range of 75% effective at preventing mild to moderate symptoms.
But clinicians should be ready to face immediate questions of safety. “Even if this vaccination is given to 20,000 [trial participants] safely, that’s not 20 million,” Dr. Offit said. “Anyone could reasonably ask questions about if it causes rare, serious side effects.
“The good news is, there are systems in place,” such as adverse event reporting systems, to identify rare events, even those that occur in one in a million vaccine recipients. Reminding patients of that continued surveillance can be reassuring.
Another reassuring point is that COVID-19 vaccine trial participants have included people from many diverse populations, he said. But children, notably absent so far, should be added to trials immediately, Dr. Offit contends.
“This is going to be important when you consider strategies to get children universally back into school,” he said, which is a “critical issue” from both learning and wellness standpoints. “It breaks my heart that we’ve been unable to do this when other countries have.”
Transparency will be paramount
While presenting data transparently to patients is key in helping them accept COVID-19 vaccination, Dr. Offit said, he also believes “telling stories” can be just as effective, if not more so. When the varicella vaccine was approved in 1995, he said, the “uptake the first few years was pretty miserable” until public service messaging emphasized that some children die from chickenpox.
“Fear works,” he said. “You always worry about pushback of something being oversold, but hopefully we’re scared enough about this virus” to convince people that vaccination is wise. “I do think personal stories carry weight on both sides,” Dr. Offit said.
Mark Sawyer, MD, of University of California San Diego School of Medicine and Rady Children’s Hospital in San Diego, California, said Offit’s presentation offered important takeaways for clinicians about how to broach the topic of COVID-19 vaccination with patients and families.
“We need to communicate clearly and transparently to patients about what we do and don’t know” about the vaccines, Dr. Sawyer said in an interview. “We will know if they have common side effects, but we will not know about very rare side effects until we have used the vaccines for a while.
“We will know how well the vaccine works over the short-term, but we won’t know over the long term,” added Dr. Sawyer, a member of the AAP Committee on Infectious Diseases.
“We can reassure the community that SARS-CoV-2 vaccines are being evaluated in trials in the same way and with the same thoroughness as other vaccines have been,” he said. “That should give people confidence that shortcuts are not being taken with regard to safety and effectiveness evaluations.”
Dr. Offit and Dr. Sawyer have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Flexibility, innovation key to practice management during pandemic
Practice management is the responsibility of every pediatrician, and leadership is more important than ever in a crisis like the COVID-19 pandemic.
“Ultimately you have a critical role in ensuring that your practice remains sustainable so that you can continue to deliver great care,” Sue Kressly, MD, a retired pediatrician from Warrington, Pa., said at the virtual annual meeting of the American Academy of Pediatrics. “None of us escaped some impact of the COVID crisis, and many of us are going to experience lasting change.”
Dr. Kressly and Suzanne Berman, MD, a pediatrician in central Tennessee, presented a live online session on how the pandemic is affecting practices and how pediatricians can effectively address those challenges.
Three ways a crisis impacts practices
“When a practice experiences a crisis, it really exposes what your practice is made of, for better or for worse,” Dr. Berman said. “The COVID crisis has been profound and broad and long enough to really stress the core tensile strength of practices along at least three axes.” Those are staffing, financial health, and partnerships.
It’s a normal human response to enter survival mode during a crisis, so staff management becomes more important than ever. Some things to consider are whether you have a truly collaborative team culture in your practice and whether you’re really listening to the staff’s struggles and suggestions.
“Staffing challenges can be very difficult,” Dr. Berman said. “Permitting staff to work from home is the single biggest thing you can do when staff needs to self-isolate.”
Financially, most medical practices have adequate cash on hand not to have to pay close attention to the numbers, Dr. Kressly said, but if physicians are looking at their books for the first time during a crisis, they have no way of knowing what their baseline expectations should be or how much to worry about their finances. It’s important to understand your practice’s or department’s budget.
Jesse Hackell, MD, a private practice pediatrician in a suburb of New York City and vice president of the New York AAP Chapter 3, attended the session and appreciated this point on finances.
“In order to provide good quality care to kids, you need to be financially successful because otherwise you’ll close your doors,” Dr. Hackell said in an interview. “It’s making yourself available to be able to provide care.”
Stressors among partners during a crisis arise from responding to the challenges of the crisis, such as who should be impacted by pay cuts or furloughs, how to account for overhead, how to distribute revenue and how to divide the work equitably. Other issues include how to protect higher risk providers fairly and how to shift schedules or case load based on unforeseen events, including quarantining.
“There is no ‘fair’ in a crisis,” Dr. Berman said. “We must use the equity paradigm to be sure everyone has what they need to survive and have the best outcome possible.”
The speakers also discussed the importance of a practice’s situation before the pandemic began, a point that resonated with attendee Jason Terk, MD, a pediatrician who practices in a large pediatric health care system near Fort Worth, Texas.
“Just like the pandemic impacts the health of people in different ways based upon their baseline health, the pandemic impacts practices in different ways based on the practice’s baseline health,” he said in an interview. “If you had good operations, a good culture, good communication and all those other good indicators of practice health before, then you stood a much better chance of surviving the pandemic as a practice than practices that had weaknesses before.”
The size of a practice did not necessarily predict the impact of the crisis, Dr. Berman said. Rather, practices with good patient engagement, active recall programs, and good fiscal planning did better.
Dr. Hackell said. “We had never seen anything like this before,” he said in an interview. “From the start we had no idea what was going to work. Try something and see if it works. If it fails, try something else. We were all operating blind here.”
The focus of most practices in the spring was on well visits, chronic care follow-up, and telehealth. Going into fall and winter, innovation will be necessary to provide appropriate care for all children while keeping in mind that the choices pediatricians make will have long-lasting implications for their staff and patients. The speakers stressed the importance of communication and transparency within the office team and to patients and the community.
Dr. Hackell appreciated the speakers’ point that kids need care, and pediatricians need to meet that need.
“Kids need well care and immunizations, and kids get sick and need sick care,” he said. “Parents need a lot more reassurance during times like this. We need to be able to provide that care and be sure that we do it safely. To give the right care at the right time in the right location is key.”
Making practice adaptations
In balancing risk and access to care, Dr. Kressly described the importance of multiple interventions, including managing some patients out of the office and making physical changes, such as putting in physical barriers and eliminating waiting rooms.
“Many practices are highly focused on PPE [personal protective equipment],” Dr. Kressly said, but even Centers for Disease Control and Prevention guidance emphasizes that PPE is the last line of defense. “There are many things we can do to protect our teams and our patients, and we know that not one single adaption is going to be 100% effective. But like the Swiss cheese model indicates, when you layer all of these efforts on top of one another, many defenses allow for the protection of the majority of people.”
Other changes include restricting office visitors to one per patient, implementing social distancing, requiring visitors to wear masks, and considering alternate locations for visits, including car and parking lot visits.
“No idea is too crazy, and some of the best ideas come from your staff,” Dr. Kressly said. She also recommended asking families where they feel most comfortable meeting.
“Don’t make any assumptions about where they want to be seen, but ask and together decide where the patient can most safely and effectively be given appropriate care,” she said.
Dr. Kressly also noted the new CPT code, 99072, that can be used to bill for “additional supplies, materials, and clinical staff time over and above those usually included in an office visit or other nonfacility service(s), when performed during a public health emergency as defined by law, due to respiratory-transmitted infectious disease.”
Pediatricians should think of ways they can remove barriers to access, such as adjusting no-show cancellation penalties and adjusting practice policies as needed when things change. “Avoid creating a culture where families do not disclose all information for fear of not being seen,” Dr. Kressly said.
A slower pace because of delays and hiccups is also normal at this time, Dr. Berman said. “If you feel like you’re just not as efficient as you were prior to COVID, it’s not just you,” she said. “It’s true. Everyone has to grapple with new things now. It takes longer.”
Things that add time include remote check-in and paperwork, more time to don and doff PPE and disinfect, dealing with technology failures, adjusting to new procedures or policies, and the general mental fatigue of adhering to PPE best practices. Patience is vital during this time, Dr. Berman said.
Several ways to improve efficiency include cutting out unnecessary steps, using standing orders and Advance Beneficiary Notice of Noncoverage (ABNs) for flu vaccinations, keeping credit card numbers on file for contactless payment, and considering the clinical and financial value of lab testing before ordering it.
“Effective triage helps patient satisfaction, access to care, and efficiency of your office workload,” Dr. Kressly said. “Use technology where it’s appropriate, but then add people where it’s needed. Connections to caring people matter even more in a time of crisis.”
The speakers also highlighted the importance of early flu vaccinations.
“One of the single biggest things you can do for value in COVID is to get your flu vaccine numbers up,” Dr. Berman said. “Severely reducing the burden of influenza will help flatten the curve, it will reduce febrile respiratory illness, and it will protect your most fragile patients.”
Two ways to do that include flu clinics and making a strong push for immunizations during the first 8 weeks after getting the vaccines. Dr Berman shared numbers from two practices showing how many more total immunizations were done in the practice that began vaccinating in early August versus early September.
A crisis is an opportunity
The speakers closed on an optimistic note that emphasized the opportunities that can grow out of the challenges presented by the pandemic, a point Dr Terk elaborated on.
“One of the most important things is realizing how we can potentially use a crisis to transform our practices,” Dr. Terk said in the interview. “As had been said before, a crisis is a terrible thing to waste. Those practices that have the gumption to innovate and find different ways to improve the way they provide care are probably going to be in better shape as we go forward.”
Critical to that success is taking risks, he added.
“When you’re innovating, failure has to be something you are permissive of because if you’re risk-averse and failure-averse, you’re not going to have the opportunity to grow and innovate, and this is another opportunity to innovate,” Dr. Terk said.
He also stressed the value of learning from one another. “We need to help each other by sharing our good practices, and on the flip side, be open to learning from each other,” he said. “Those pediatricians who are struggling need to be open-minded and open-hearted to understanding how we can operate our practices better and know that the things we think are barriers we can’t change are probably things we probably haven’t allowed ourselves to think about changing.”
Dr. Kressly and Dr. Berman recommended several specific actions for pediatricians to take:
- Creating a SWOT (Strengths, Weaknesses, Opportunities, and Threats) analysis regarding your practice management response to the pandemic.
- Articulating three goals for improving your understanding or the implementation of management in your practice.
- Creating a working group to identify and implement ways to improve clinical work flow and communication strategies.
“Now is the time to meaningfully address disparities of access to appropriate health care and the impact of social determinants of health,” Dr. Kressly said. It’s also an opportunity to build meaningful relationships with patient families based on trust, science, and “true shared decision-making with health literacy in mind.”
Dr Kressly is the medical director of and owns shares in Office Practicum. Dr. Berman is the assistant medical director of and owns shares in Office Practicum, and is the owner of Script Doctor LLC. Dr. Terk and Dr. Hackell had no relevant financial disclosures.
Practice management is the responsibility of every pediatrician, and leadership is more important than ever in a crisis like the COVID-19 pandemic.
“Ultimately you have a critical role in ensuring that your practice remains sustainable so that you can continue to deliver great care,” Sue Kressly, MD, a retired pediatrician from Warrington, Pa., said at the virtual annual meeting of the American Academy of Pediatrics. “None of us escaped some impact of the COVID crisis, and many of us are going to experience lasting change.”
Dr. Kressly and Suzanne Berman, MD, a pediatrician in central Tennessee, presented a live online session on how the pandemic is affecting practices and how pediatricians can effectively address those challenges.
Three ways a crisis impacts practices
“When a practice experiences a crisis, it really exposes what your practice is made of, for better or for worse,” Dr. Berman said. “The COVID crisis has been profound and broad and long enough to really stress the core tensile strength of practices along at least three axes.” Those are staffing, financial health, and partnerships.
It’s a normal human response to enter survival mode during a crisis, so staff management becomes more important than ever. Some things to consider are whether you have a truly collaborative team culture in your practice and whether you’re really listening to the staff’s struggles and suggestions.
“Staffing challenges can be very difficult,” Dr. Berman said. “Permitting staff to work from home is the single biggest thing you can do when staff needs to self-isolate.”
Financially, most medical practices have adequate cash on hand not to have to pay close attention to the numbers, Dr. Kressly said, but if physicians are looking at their books for the first time during a crisis, they have no way of knowing what their baseline expectations should be or how much to worry about their finances. It’s important to understand your practice’s or department’s budget.
Jesse Hackell, MD, a private practice pediatrician in a suburb of New York City and vice president of the New York AAP Chapter 3, attended the session and appreciated this point on finances.
“In order to provide good quality care to kids, you need to be financially successful because otherwise you’ll close your doors,” Dr. Hackell said in an interview. “It’s making yourself available to be able to provide care.”
Stressors among partners during a crisis arise from responding to the challenges of the crisis, such as who should be impacted by pay cuts or furloughs, how to account for overhead, how to distribute revenue and how to divide the work equitably. Other issues include how to protect higher risk providers fairly and how to shift schedules or case load based on unforeseen events, including quarantining.
“There is no ‘fair’ in a crisis,” Dr. Berman said. “We must use the equity paradigm to be sure everyone has what they need to survive and have the best outcome possible.”
The speakers also discussed the importance of a practice’s situation before the pandemic began, a point that resonated with attendee Jason Terk, MD, a pediatrician who practices in a large pediatric health care system near Fort Worth, Texas.
“Just like the pandemic impacts the health of people in different ways based upon their baseline health, the pandemic impacts practices in different ways based on the practice’s baseline health,” he said in an interview. “If you had good operations, a good culture, good communication and all those other good indicators of practice health before, then you stood a much better chance of surviving the pandemic as a practice than practices that had weaknesses before.”
The size of a practice did not necessarily predict the impact of the crisis, Dr. Berman said. Rather, practices with good patient engagement, active recall programs, and good fiscal planning did better.
Dr. Hackell said. “We had never seen anything like this before,” he said in an interview. “From the start we had no idea what was going to work. Try something and see if it works. If it fails, try something else. We were all operating blind here.”
The focus of most practices in the spring was on well visits, chronic care follow-up, and telehealth. Going into fall and winter, innovation will be necessary to provide appropriate care for all children while keeping in mind that the choices pediatricians make will have long-lasting implications for their staff and patients. The speakers stressed the importance of communication and transparency within the office team and to patients and the community.
Dr. Hackell appreciated the speakers’ point that kids need care, and pediatricians need to meet that need.
“Kids need well care and immunizations, and kids get sick and need sick care,” he said. “Parents need a lot more reassurance during times like this. We need to be able to provide that care and be sure that we do it safely. To give the right care at the right time in the right location is key.”
Making practice adaptations
In balancing risk and access to care, Dr. Kressly described the importance of multiple interventions, including managing some patients out of the office and making physical changes, such as putting in physical barriers and eliminating waiting rooms.
“Many practices are highly focused on PPE [personal protective equipment],” Dr. Kressly said, but even Centers for Disease Control and Prevention guidance emphasizes that PPE is the last line of defense. “There are many things we can do to protect our teams and our patients, and we know that not one single adaption is going to be 100% effective. But like the Swiss cheese model indicates, when you layer all of these efforts on top of one another, many defenses allow for the protection of the majority of people.”
Other changes include restricting office visitors to one per patient, implementing social distancing, requiring visitors to wear masks, and considering alternate locations for visits, including car and parking lot visits.
“No idea is too crazy, and some of the best ideas come from your staff,” Dr. Kressly said. She also recommended asking families where they feel most comfortable meeting.
“Don’t make any assumptions about where they want to be seen, but ask and together decide where the patient can most safely and effectively be given appropriate care,” she said.
Dr. Kressly also noted the new CPT code, 99072, that can be used to bill for “additional supplies, materials, and clinical staff time over and above those usually included in an office visit or other nonfacility service(s), when performed during a public health emergency as defined by law, due to respiratory-transmitted infectious disease.”
Pediatricians should think of ways they can remove barriers to access, such as adjusting no-show cancellation penalties and adjusting practice policies as needed when things change. “Avoid creating a culture where families do not disclose all information for fear of not being seen,” Dr. Kressly said.
A slower pace because of delays and hiccups is also normal at this time, Dr. Berman said. “If you feel like you’re just not as efficient as you were prior to COVID, it’s not just you,” she said. “It’s true. Everyone has to grapple with new things now. It takes longer.”
Things that add time include remote check-in and paperwork, more time to don and doff PPE and disinfect, dealing with technology failures, adjusting to new procedures or policies, and the general mental fatigue of adhering to PPE best practices. Patience is vital during this time, Dr. Berman said.
Several ways to improve efficiency include cutting out unnecessary steps, using standing orders and Advance Beneficiary Notice of Noncoverage (ABNs) for flu vaccinations, keeping credit card numbers on file for contactless payment, and considering the clinical and financial value of lab testing before ordering it.
“Effective triage helps patient satisfaction, access to care, and efficiency of your office workload,” Dr. Kressly said. “Use technology where it’s appropriate, but then add people where it’s needed. Connections to caring people matter even more in a time of crisis.”
The speakers also highlighted the importance of early flu vaccinations.
“One of the single biggest things you can do for value in COVID is to get your flu vaccine numbers up,” Dr. Berman said. “Severely reducing the burden of influenza will help flatten the curve, it will reduce febrile respiratory illness, and it will protect your most fragile patients.”
Two ways to do that include flu clinics and making a strong push for immunizations during the first 8 weeks after getting the vaccines. Dr Berman shared numbers from two practices showing how many more total immunizations were done in the practice that began vaccinating in early August versus early September.
A crisis is an opportunity
The speakers closed on an optimistic note that emphasized the opportunities that can grow out of the challenges presented by the pandemic, a point Dr Terk elaborated on.
“One of the most important things is realizing how we can potentially use a crisis to transform our practices,” Dr. Terk said in the interview. “As had been said before, a crisis is a terrible thing to waste. Those practices that have the gumption to innovate and find different ways to improve the way they provide care are probably going to be in better shape as we go forward.”
Critical to that success is taking risks, he added.
“When you’re innovating, failure has to be something you are permissive of because if you’re risk-averse and failure-averse, you’re not going to have the opportunity to grow and innovate, and this is another opportunity to innovate,” Dr. Terk said.
He also stressed the value of learning from one another. “We need to help each other by sharing our good practices, and on the flip side, be open to learning from each other,” he said. “Those pediatricians who are struggling need to be open-minded and open-hearted to understanding how we can operate our practices better and know that the things we think are barriers we can’t change are probably things we probably haven’t allowed ourselves to think about changing.”
Dr. Kressly and Dr. Berman recommended several specific actions for pediatricians to take:
- Creating a SWOT (Strengths, Weaknesses, Opportunities, and Threats) analysis regarding your practice management response to the pandemic.
- Articulating three goals for improving your understanding or the implementation of management in your practice.
- Creating a working group to identify and implement ways to improve clinical work flow and communication strategies.
“Now is the time to meaningfully address disparities of access to appropriate health care and the impact of social determinants of health,” Dr. Kressly said. It’s also an opportunity to build meaningful relationships with patient families based on trust, science, and “true shared decision-making with health literacy in mind.”
Dr Kressly is the medical director of and owns shares in Office Practicum. Dr. Berman is the assistant medical director of and owns shares in Office Practicum, and is the owner of Script Doctor LLC. Dr. Terk and Dr. Hackell had no relevant financial disclosures.
Practice management is the responsibility of every pediatrician, and leadership is more important than ever in a crisis like the COVID-19 pandemic.
“Ultimately you have a critical role in ensuring that your practice remains sustainable so that you can continue to deliver great care,” Sue Kressly, MD, a retired pediatrician from Warrington, Pa., said at the virtual annual meeting of the American Academy of Pediatrics. “None of us escaped some impact of the COVID crisis, and many of us are going to experience lasting change.”
Dr. Kressly and Suzanne Berman, MD, a pediatrician in central Tennessee, presented a live online session on how the pandemic is affecting practices and how pediatricians can effectively address those challenges.
Three ways a crisis impacts practices
“When a practice experiences a crisis, it really exposes what your practice is made of, for better or for worse,” Dr. Berman said. “The COVID crisis has been profound and broad and long enough to really stress the core tensile strength of practices along at least three axes.” Those are staffing, financial health, and partnerships.
It’s a normal human response to enter survival mode during a crisis, so staff management becomes more important than ever. Some things to consider are whether you have a truly collaborative team culture in your practice and whether you’re really listening to the staff’s struggles and suggestions.
“Staffing challenges can be very difficult,” Dr. Berman said. “Permitting staff to work from home is the single biggest thing you can do when staff needs to self-isolate.”
Financially, most medical practices have adequate cash on hand not to have to pay close attention to the numbers, Dr. Kressly said, but if physicians are looking at their books for the first time during a crisis, they have no way of knowing what their baseline expectations should be or how much to worry about their finances. It’s important to understand your practice’s or department’s budget.
Jesse Hackell, MD, a private practice pediatrician in a suburb of New York City and vice president of the New York AAP Chapter 3, attended the session and appreciated this point on finances.
“In order to provide good quality care to kids, you need to be financially successful because otherwise you’ll close your doors,” Dr. Hackell said in an interview. “It’s making yourself available to be able to provide care.”
Stressors among partners during a crisis arise from responding to the challenges of the crisis, such as who should be impacted by pay cuts or furloughs, how to account for overhead, how to distribute revenue and how to divide the work equitably. Other issues include how to protect higher risk providers fairly and how to shift schedules or case load based on unforeseen events, including quarantining.
“There is no ‘fair’ in a crisis,” Dr. Berman said. “We must use the equity paradigm to be sure everyone has what they need to survive and have the best outcome possible.”
The speakers also discussed the importance of a practice’s situation before the pandemic began, a point that resonated with attendee Jason Terk, MD, a pediatrician who practices in a large pediatric health care system near Fort Worth, Texas.
“Just like the pandemic impacts the health of people in different ways based upon their baseline health, the pandemic impacts practices in different ways based on the practice’s baseline health,” he said in an interview. “If you had good operations, a good culture, good communication and all those other good indicators of practice health before, then you stood a much better chance of surviving the pandemic as a practice than practices that had weaknesses before.”
The size of a practice did not necessarily predict the impact of the crisis, Dr. Berman said. Rather, practices with good patient engagement, active recall programs, and good fiscal planning did better.
Dr. Hackell said. “We had never seen anything like this before,” he said in an interview. “From the start we had no idea what was going to work. Try something and see if it works. If it fails, try something else. We were all operating blind here.”
The focus of most practices in the spring was on well visits, chronic care follow-up, and telehealth. Going into fall and winter, innovation will be necessary to provide appropriate care for all children while keeping in mind that the choices pediatricians make will have long-lasting implications for their staff and patients. The speakers stressed the importance of communication and transparency within the office team and to patients and the community.
Dr. Hackell appreciated the speakers’ point that kids need care, and pediatricians need to meet that need.
“Kids need well care and immunizations, and kids get sick and need sick care,” he said. “Parents need a lot more reassurance during times like this. We need to be able to provide that care and be sure that we do it safely. To give the right care at the right time in the right location is key.”
Making practice adaptations
In balancing risk and access to care, Dr. Kressly described the importance of multiple interventions, including managing some patients out of the office and making physical changes, such as putting in physical barriers and eliminating waiting rooms.
“Many practices are highly focused on PPE [personal protective equipment],” Dr. Kressly said, but even Centers for Disease Control and Prevention guidance emphasizes that PPE is the last line of defense. “There are many things we can do to protect our teams and our patients, and we know that not one single adaption is going to be 100% effective. But like the Swiss cheese model indicates, when you layer all of these efforts on top of one another, many defenses allow for the protection of the majority of people.”
Other changes include restricting office visitors to one per patient, implementing social distancing, requiring visitors to wear masks, and considering alternate locations for visits, including car and parking lot visits.
“No idea is too crazy, and some of the best ideas come from your staff,” Dr. Kressly said. She also recommended asking families where they feel most comfortable meeting.
“Don’t make any assumptions about where they want to be seen, but ask and together decide where the patient can most safely and effectively be given appropriate care,” she said.
Dr. Kressly also noted the new CPT code, 99072, that can be used to bill for “additional supplies, materials, and clinical staff time over and above those usually included in an office visit or other nonfacility service(s), when performed during a public health emergency as defined by law, due to respiratory-transmitted infectious disease.”
Pediatricians should think of ways they can remove barriers to access, such as adjusting no-show cancellation penalties and adjusting practice policies as needed when things change. “Avoid creating a culture where families do not disclose all information for fear of not being seen,” Dr. Kressly said.
A slower pace because of delays and hiccups is also normal at this time, Dr. Berman said. “If you feel like you’re just not as efficient as you were prior to COVID, it’s not just you,” she said. “It’s true. Everyone has to grapple with new things now. It takes longer.”
Things that add time include remote check-in and paperwork, more time to don and doff PPE and disinfect, dealing with technology failures, adjusting to new procedures or policies, and the general mental fatigue of adhering to PPE best practices. Patience is vital during this time, Dr. Berman said.
Several ways to improve efficiency include cutting out unnecessary steps, using standing orders and Advance Beneficiary Notice of Noncoverage (ABNs) for flu vaccinations, keeping credit card numbers on file for contactless payment, and considering the clinical and financial value of lab testing before ordering it.
“Effective triage helps patient satisfaction, access to care, and efficiency of your office workload,” Dr. Kressly said. “Use technology where it’s appropriate, but then add people where it’s needed. Connections to caring people matter even more in a time of crisis.”
The speakers also highlighted the importance of early flu vaccinations.
“One of the single biggest things you can do for value in COVID is to get your flu vaccine numbers up,” Dr. Berman said. “Severely reducing the burden of influenza will help flatten the curve, it will reduce febrile respiratory illness, and it will protect your most fragile patients.”
Two ways to do that include flu clinics and making a strong push for immunizations during the first 8 weeks after getting the vaccines. Dr Berman shared numbers from two practices showing how many more total immunizations were done in the practice that began vaccinating in early August versus early September.
A crisis is an opportunity
The speakers closed on an optimistic note that emphasized the opportunities that can grow out of the challenges presented by the pandemic, a point Dr Terk elaborated on.
“One of the most important things is realizing how we can potentially use a crisis to transform our practices,” Dr. Terk said in the interview. “As had been said before, a crisis is a terrible thing to waste. Those practices that have the gumption to innovate and find different ways to improve the way they provide care are probably going to be in better shape as we go forward.”
Critical to that success is taking risks, he added.
“When you’re innovating, failure has to be something you are permissive of because if you’re risk-averse and failure-averse, you’re not going to have the opportunity to grow and innovate, and this is another opportunity to innovate,” Dr. Terk said.
He also stressed the value of learning from one another. “We need to help each other by sharing our good practices, and on the flip side, be open to learning from each other,” he said. “Those pediatricians who are struggling need to be open-minded and open-hearted to understanding how we can operate our practices better and know that the things we think are barriers we can’t change are probably things we probably haven’t allowed ourselves to think about changing.”
Dr. Kressly and Dr. Berman recommended several specific actions for pediatricians to take:
- Creating a SWOT (Strengths, Weaknesses, Opportunities, and Threats) analysis regarding your practice management response to the pandemic.
- Articulating three goals for improving your understanding or the implementation of management in your practice.
- Creating a working group to identify and implement ways to improve clinical work flow and communication strategies.
“Now is the time to meaningfully address disparities of access to appropriate health care and the impact of social determinants of health,” Dr. Kressly said. It’s also an opportunity to build meaningful relationships with patient families based on trust, science, and “true shared decision-making with health literacy in mind.”
Dr Kressly is the medical director of and owns shares in Office Practicum. Dr. Berman is the assistant medical director of and owns shares in Office Practicum, and is the owner of Script Doctor LLC. Dr. Terk and Dr. Hackell had no relevant financial disclosures.
FROM AAP 2020