User login
‘This food will kill you, that food will save you’
Not sure if you’ve heard the news, but eating a single hot dog will apparently cost you 36 minutes of healthy life. My first thought when hearing this was of course the same as everyone else’s: Poor Joey Chestnut, multiyear winner of Nathan’s annual hot dog–eating contest.
He won this year’s contest with 76 hot dogs, which puts his total number of competition-consumed hot dogs at 1,089 – which cost him, it would seem, 27.2 days of healthy life. Unless, of course, every hot dog he inhaled came with a bun hosting two portions of sesame seeds, which in turn would buy him 50 extra minutes of life (25 minutes per portion, you see) and would consequently have extended his life by 10.6 days.
Clearly, the obvious solution here is to ensure that all hot dog buns have two portions of sesame seeds on them moving forward; that way, hot dogs can transition from being poisonous killers to antiaging medicine.
The other solution, albeit less exciting, perhaps, is for researchers to stop studying single foods’ impacts on health, and/or for journals to stop publishing them, and/or for the media to stop promoting them – because they are all as ridiculously useless as the example above highlighting findings from a newly published study in Nature Food, entitled “Small targeted dietary changes can yield substantial gains for human health and the environment.”
While no doubt we would all love for diet and health to be so well understood that we could choose specific single foods (knowing that they would prolong our lives) while avoiding single foods that would shorten it, there’s this unfortunate truth that the degree of confounding among food alone is staggering. People eat thousands of different foods in thousands of different dietary combinations. Moreover, most (all?) research conducted on dietary impacts of single foods on health don’t actually track consumption of those specific foods over time, let alone their interactions with all other foods consumed, but rather at moments in time.
In the case of the “hot dogs will kill you unless there are sesame seeds on your bun” article, for example, the researchers utilized one solitary dietary recall session upon which to base their ridiculously specific, ridiculous conclusions.
People’s diets also change over time for various reasons, and of course people themselves are very different. You might imagine that people whose diets are rich in chicken wings, sugared soda, and hot dogs will have markedly different lifestyles and demographics than those whose diets are rich in walnuts, sashimi, and avocados.
So why do we keep seeing studies like this being published? Is it because they’re basically clickbait catnip for journals and newspapers, and in our publish-or-perish attention-seeking world, that means they not only get a pass but they get a press release? Is it because peer review is broken and everyone knows it? Is it because as a society, we’re frogs who have been steeping for decades in the ever-heated pot of nutritional nonsense, and consequently don’t think to question it?
I don’t know the answer to any of those questions, but one thing I do know: Studies on single foods’ impact on life length are pointless, impossible, and idiotic, and people who share them noncritically should be forever shunned – or at the very least, forever ignored.
Yoni Freedhoff, MD, is an associate professor of family medicine at the University of Ottawa and medical director of the Bariatric Medical Institute, a nonsurgical weight-management center.
A version of this article first appeared on Medscape.com.
Not sure if you’ve heard the news, but eating a single hot dog will apparently cost you 36 minutes of healthy life. My first thought when hearing this was of course the same as everyone else’s: Poor Joey Chestnut, multiyear winner of Nathan’s annual hot dog–eating contest.
He won this year’s contest with 76 hot dogs, which puts his total number of competition-consumed hot dogs at 1,089 – which cost him, it would seem, 27.2 days of healthy life. Unless, of course, every hot dog he inhaled came with a bun hosting two portions of sesame seeds, which in turn would buy him 50 extra minutes of life (25 minutes per portion, you see) and would consequently have extended his life by 10.6 days.
Clearly, the obvious solution here is to ensure that all hot dog buns have two portions of sesame seeds on them moving forward; that way, hot dogs can transition from being poisonous killers to antiaging medicine.
The other solution, albeit less exciting, perhaps, is for researchers to stop studying single foods’ impacts on health, and/or for journals to stop publishing them, and/or for the media to stop promoting them – because they are all as ridiculously useless as the example above highlighting findings from a newly published study in Nature Food, entitled “Small targeted dietary changes can yield substantial gains for human health and the environment.”
While no doubt we would all love for diet and health to be so well understood that we could choose specific single foods (knowing that they would prolong our lives) while avoiding single foods that would shorten it, there’s this unfortunate truth that the degree of confounding among food alone is staggering. People eat thousands of different foods in thousands of different dietary combinations. Moreover, most (all?) research conducted on dietary impacts of single foods on health don’t actually track consumption of those specific foods over time, let alone their interactions with all other foods consumed, but rather at moments in time.
In the case of the “hot dogs will kill you unless there are sesame seeds on your bun” article, for example, the researchers utilized one solitary dietary recall session upon which to base their ridiculously specific, ridiculous conclusions.
People’s diets also change over time for various reasons, and of course people themselves are very different. You might imagine that people whose diets are rich in chicken wings, sugared soda, and hot dogs will have markedly different lifestyles and demographics than those whose diets are rich in walnuts, sashimi, and avocados.
So why do we keep seeing studies like this being published? Is it because they’re basically clickbait catnip for journals and newspapers, and in our publish-or-perish attention-seeking world, that means they not only get a pass but they get a press release? Is it because peer review is broken and everyone knows it? Is it because as a society, we’re frogs who have been steeping for decades in the ever-heated pot of nutritional nonsense, and consequently don’t think to question it?
I don’t know the answer to any of those questions, but one thing I do know: Studies on single foods’ impact on life length are pointless, impossible, and idiotic, and people who share them noncritically should be forever shunned – or at the very least, forever ignored.
Yoni Freedhoff, MD, is an associate professor of family medicine at the University of Ottawa and medical director of the Bariatric Medical Institute, a nonsurgical weight-management center.
A version of this article first appeared on Medscape.com.
Not sure if you’ve heard the news, but eating a single hot dog will apparently cost you 36 minutes of healthy life. My first thought when hearing this was of course the same as everyone else’s: Poor Joey Chestnut, multiyear winner of Nathan’s annual hot dog–eating contest.
He won this year’s contest with 76 hot dogs, which puts his total number of competition-consumed hot dogs at 1,089 – which cost him, it would seem, 27.2 days of healthy life. Unless, of course, every hot dog he inhaled came with a bun hosting two portions of sesame seeds, which in turn would buy him 50 extra minutes of life (25 minutes per portion, you see) and would consequently have extended his life by 10.6 days.
Clearly, the obvious solution here is to ensure that all hot dog buns have two portions of sesame seeds on them moving forward; that way, hot dogs can transition from being poisonous killers to antiaging medicine.
The other solution, albeit less exciting, perhaps, is for researchers to stop studying single foods’ impacts on health, and/or for journals to stop publishing them, and/or for the media to stop promoting them – because they are all as ridiculously useless as the example above highlighting findings from a newly published study in Nature Food, entitled “Small targeted dietary changes can yield substantial gains for human health and the environment.”
While no doubt we would all love for diet and health to be so well understood that we could choose specific single foods (knowing that they would prolong our lives) while avoiding single foods that would shorten it, there’s this unfortunate truth that the degree of confounding among food alone is staggering. People eat thousands of different foods in thousands of different dietary combinations. Moreover, most (all?) research conducted on dietary impacts of single foods on health don’t actually track consumption of those specific foods over time, let alone their interactions with all other foods consumed, but rather at moments in time.
In the case of the “hot dogs will kill you unless there are sesame seeds on your bun” article, for example, the researchers utilized one solitary dietary recall session upon which to base their ridiculously specific, ridiculous conclusions.
People’s diets also change over time for various reasons, and of course people themselves are very different. You might imagine that people whose diets are rich in chicken wings, sugared soda, and hot dogs will have markedly different lifestyles and demographics than those whose diets are rich in walnuts, sashimi, and avocados.
So why do we keep seeing studies like this being published? Is it because they’re basically clickbait catnip for journals and newspapers, and in our publish-or-perish attention-seeking world, that means they not only get a pass but they get a press release? Is it because peer review is broken and everyone knows it? Is it because as a society, we’re frogs who have been steeping for decades in the ever-heated pot of nutritional nonsense, and consequently don’t think to question it?
I don’t know the answer to any of those questions, but one thing I do know: Studies on single foods’ impact on life length are pointless, impossible, and idiotic, and people who share them noncritically should be forever shunned – or at the very least, forever ignored.
Yoni Freedhoff, MD, is an associate professor of family medicine at the University of Ottawa and medical director of the Bariatric Medical Institute, a nonsurgical weight-management center.
A version of this article first appeared on Medscape.com.
EMPEROR-Preserved spouts torrent of reports on empagliflozin treatment of HFpEF
The featured report from the 6,000-patient EMPEROR-Preserved trial at the virtual annual congress of the European Society of Cardiology drew lots of attention for its headline finding: the first unequivocal demonstration that a medication, empagliflozin, can significantly reduce the rate of cardiovascular death and hospitalization for heart failure in patients with heart failure with preserved ejection fraction (HFpEF, a left ventricular ejection fraction of more than 40%), with the details simultaneously published online.
But at the same time, the EMPEROR-Preserved investigators released four additional reports with a lot more outcome analyses that also deserve some attention.
The puzzling neutral effect on renal events
Perhaps the most surprising and complicated set of findings among the main EMPEROR-Preserved outcomes involved renal outcomes.
The trial’s primary outcome was the combined rate of cardiovascular death or hospitalization for heart failure (HHF), and the results showed that treatment with empagliflozin (Jardiance) for a median of 26 months on top of standard treatment for patients with HFpEF led to a significant 21% relative risk reduction, compared with placebo-treated patients.
The trial had two prespecified secondary outcomes. One was the total number of HHF, which dropped by a significant 27%, compared with placebo. The second was the mean change in slope of estimated glomerular filtration rate (eGFR) on an annualized basis, and the empagliflozin regimen reduced the cumulative annual deficit, compared with placebo by an average of 1.36 mL/min per 1.73 m2, a significant difference.
This preservation of renal function was consistent with results from many prior studies of empagliflozin and all of the other U.S.-approved agents from the sodium-glucose cotransporter 2 inhibitor class. Preservation of renal function and a reduction in renal events has become a hallmark property of all agents in the SGLT2 inhibitor class both in patients with type 2 diabetes, as well as in those without diabetes but with heart failure with reduced ejection fraction (HFrEF) or with chronic kidney disease.
EMPEROR-Preserved threw a wrench into what had been an unbroken history of renal protection by SGLT2 inhibitors. That happened when a prespecified endpoint of the study – a composite renal outcome defined as time to first occurrence of chronic dialysis, renal transplantation, a sustained reduction of at least 40% in eGFR, or a sustained drop in eGFR of more than 10 or 15 mL/min per 1.73 m2 from baseline – yielded an unexpected neutral finding.
For this composite renal outcome, EMPEROR-Preserved showed a nonsignificant 5% reduction, compared with placebo, a result that both differed from what had been seen in essentially all the other SGLT2 inhibitor trials that had looked at this, but which also seemed at odds with the observed significant preservation of renal function that seemed substantial enough to produce a clinically meaningful benefit.
Renal effects blunted in HFpEF
The immediate upshot was a letter published by several EMPEROR-Preserved investigators that spelled out this discrepancy and came to the jolting conclusion that “eGFR slope analysis has limitations as a surrogate for predicting the effect of drugs on renal outcomes in patients with heart failure.”
The same authors, along with some additional associates, also published a second letter that noted a further unexpected twist with the renal outcome: “In prior large-scale clinical trials, the effect of SGLT2 inhibitors on heart failure and renal outcomes had consistently tracked together,” they noted, but in this case it didn’t, a discordance they said was “extraordinarily puzzling”.
This led the study’s leaders to reanalyze the renal outcomes using a different definition, one that Milton Packer, MD, who helped design the trial and oversaw several of its analyses, called “a more conventional definition of renal events,” during his presentation of these findings at the congress. The researchers swapped out a 40% drop from baseline eGFR as an event and replaced it with a 50% decline, a change designed to screen out less severe, and often transient, reductions in kidney function that have less lasting impact on health. They also added an additional component to the composite endpoint, renal death. A revised analysis using this new renal composite outcome appeared in the European Journal of Heart Failure letter.
This change cut the total number of renal events tallied in the trial nearly in half, down to 112, and showed a more robust decline in renal events with empagliflozin treatment compared with the initial analysis, although the drop remained nonsignificant. The revised analysis also showed that the overall, nonsignificant 22% relative reduction in renal events in patients on empagliflozin, compared with placebo, dwindled down to completely nonexistent in the tertile of patients with a left ventricular ejection fraction of 60% or greater. In this tertile the hazard ratio actually showed a nonsignificant point estimate of a 24% increased rate of renal events on empagliflozin, with the caveat that this subgroup now included a total of just 40 total events between the two treatment arms. (Each of the two other tertiles also had roughly the same number of total events.)
The biggest effect on renal-event reduction was in the tertile of patients with an ejection fraction of 41%-49%, in which empagliflozin treatment was linked with a significant 59% cut in renal events, compared with placebo. The analysis also showed significant heterogeneity in thus outcome between this subgroup and the other two tertiles that had higher ejection fractions and showed reduced rates of protection by empagliflozin against renal events.
This apparent blunting of a renal effect despite preservation of renal function seemed to mimic the blunting of the primary cardiovascular outcome effect that also appeared in patients with ejection fractions in the 60%-65% range or above.
“If we knew what blunted the effect of empagliflozin on heart failure outcomes at higher ejection fraction levels, we think the same explanation may also apply to the blunting of effect on renal outcomes, but right now we do not know the answer to either question,” Dr. Packer said in an interview. He’s suggested that one possibility is that many of the enrolled patients identified as having HFpEF, but with these high ejection fractions may have not actually had HFpEF, and their signs and symptoms may have instead resulted from atrial fibrillation.
“Many patients with an ejection fraction of 60%-65% and above had atrial fibrillation,” he noted, with a prevalence at enrollment in this subgroup of about 50%. Atrial fibrillation can cause dyspnea, a hallmark symptom leading to diagnosis of heart failure, and it also increases levels of N-terminal of the prohormone brain natriuretic peptide, a metric that served as a gatekeeper for entry into the trial. “Essentially, we are saying that many of the criteria that we specified to ensure that patients had heart failure probably did not work very well in patients with an ejection fraction of 65% or greater,” said Dr. Packer, a cardiologist at Baylor University Medical Center in Dallas. “We need to figure out who these patients are.”
Some experts not involved with the study voiced skepticism that the renal findings reflected a real issue.
“I’m quite optimistic that in the long-term the effect on eGFR will translate into renal protection,” said Rudolf A. de Boer, MD, PhD, a professor of translational cardiology at University Medical Center Groningen (the Netherlands), and designated discussant at the congress for the presentation by Dr. Packer.
John J.V. McMurray, MD, a professor of cardiology and a heart failure specialist at Glasgow University, speculated that the unexpected renal outcomes data may relate to the initial decline in renal function produced by treatment with SGLT2 inhibitors despite their longer-term enhancement of renal protection.
“If you use a treatment that protects the kidneys in the long-term but causes an initial dip in eGFR, more patients receiving that treatment will have an early ‘event,’ ” he noted in an interview. He also cautioned about the dangers of subgroup analyses that dice the study population into small cohorts.
“Trials are powered to look at the effect of treatment in the overall population. Everything else is exploratory, underpowered, and subject to the play of chance,” Dr. McMurray stressed.
Counting additional cardiovascular disease events allows more analyses
A third auxiliary report from the EMPEROR-Preserved investigators performed several prespecified analyses that depended on adding additional cardiovascular disease endpoints to the core tallies of cardiovascular death or HHF – such as emergent, urgent, and outpatient events that reflected worsening heart failure – and also included information on diuretic and vasopressor use because of worsening heart failure. The increased event numbers allowed the researchers to perform 30 additional analyses included in this report, according to the count kept by Dr. Packer who was the lead author.
He highlighted several of the additional results in this paper that documented benefits from empagliflozin treatment, compared with placebo:
- A significant 29% reduction in the need for admission to a cardiac care unit or intensive care unit during an HHF.
- A nonsignificant 33% reduction in the need for intravenous vasopressors or positive inotropic drugs during HHF.
- A significantly increased rate of patients achieving a higher New York Heart Association functional class. For example, after the first year of treatment patients who received empagliflozin had a 37% higher rate of functional class improvement, compared with patients who received placebo.
Dr. McMurray had his own list of key takeaways from this paper, including:
- Among patients who needed hospitalization, “those treated with empagliflozin were less sick than those in the placebo group.”
- In addition to reducing HHF empagliflozin treatment also reduced episodes of outpatient worsening as reflected by their receipt of intensified diuretic treatment, which occurred a significant 27% less often, compared with patients on placebo.
- Treatment with empagliflozin also linked with a significant 39% relative reduction in emergency or urgent-care visits that required intravenous therapy.
Empagliflozin’s performance relative to sacubitril/valsartan
The fourth additional report focused on a post hoc, cross-trial comparison of the results from EMPEROR-Preserved and from another recent trial that, like EMPEROR-Preserved, assessed in patients with HFpEF a drug previously proven to work quite well in patients with HFrEF. The comparator drug was sacubitril/valsartan (Entresto), which underwent testing in patients with HFpEF in the PARAGON-HF trial.
The primary outcome of PARAGON-HF, which randomized 4,822 patients, was reduction in cardiovascular death and in total HHF. This dropped by a relative 13%, compared with placebo, during a median of 35 months, a between-group difference that came close to but did not achieve significance (P = .06). Despite this limitation, the Food and Drug Administration in February 2021 loosened the indication for using sacubitril/valsartan in patients with heart failure and a “below normal” ejection fraction, a category that can include many patients considered to have HFpEF.
Although the researchers who ran this analysis, including Dr. Packer, who was the first author, admitted that “comparison of effect sizes across trials is fraught with difficulties,” they nonetheless concluded from their analysis that “for all outcomes that included HHF the effect size was larger for empagliflozin than for sacubitril/valsartan.”
Dr. McMurray, a lead instigator for PARAGON-HF, said there was little to take away from this analysis.
“The patient populations were different, and sacubitril/valsartan was compared against an active therapy, valsartan,” while in EMPEROR-Preserved empagliflozin compared against placebo. “Most of us believe that sacubitril/valsartan and SGLT2 inhibitors work in different but complementary ways, and their benefits are additive. You would want patients with HFpEF or HFrEF to take both,” he said in an interview.
Dr. Packer agreed with that approach and added that he would probably also prescribe a third agent, spironolactone, to many patients with HFpEF.
EMPEROR-Preserved was sponsored by Boehringer Ingelheim and Eli Lilly, which jointly market empagliflozin (Jardiance). PARAGON-HF was sponsored by Novartis, which markets sacubitril/valsartan (Entresto). Dr. Packer has received consulting fees from Boehringer Ingelheim and from numerous other companies. Dr. de Boer has research contracts with Boehringer Ingelheim as well as from Abbott, AstraZeneca, Cardior, Ionis, Novo Nordisk, and Roche, and he has been a consultant to Novartis as well as to Abbott, AstraZeneca, Gayer, and Roche. Dr. McMurray led trials of sacubitril/valsartan sponsored by Novartis, and his institution has received compensation for his participation in studies sponsored by Abbvie, AstraZeneca, Cardurion, DalCor, GlaxoSmithKline, Pfizer, and Theracos.
The featured report from the 6,000-patient EMPEROR-Preserved trial at the virtual annual congress of the European Society of Cardiology drew lots of attention for its headline finding: the first unequivocal demonstration that a medication, empagliflozin, can significantly reduce the rate of cardiovascular death and hospitalization for heart failure in patients with heart failure with preserved ejection fraction (HFpEF, a left ventricular ejection fraction of more than 40%), with the details simultaneously published online.
But at the same time, the EMPEROR-Preserved investigators released four additional reports with a lot more outcome analyses that also deserve some attention.
The puzzling neutral effect on renal events
Perhaps the most surprising and complicated set of findings among the main EMPEROR-Preserved outcomes involved renal outcomes.
The trial’s primary outcome was the combined rate of cardiovascular death or hospitalization for heart failure (HHF), and the results showed that treatment with empagliflozin (Jardiance) for a median of 26 months on top of standard treatment for patients with HFpEF led to a significant 21% relative risk reduction, compared with placebo-treated patients.
The trial had two prespecified secondary outcomes. One was the total number of HHF, which dropped by a significant 27%, compared with placebo. The second was the mean change in slope of estimated glomerular filtration rate (eGFR) on an annualized basis, and the empagliflozin regimen reduced the cumulative annual deficit, compared with placebo by an average of 1.36 mL/min per 1.73 m2, a significant difference.
This preservation of renal function was consistent with results from many prior studies of empagliflozin and all of the other U.S.-approved agents from the sodium-glucose cotransporter 2 inhibitor class. Preservation of renal function and a reduction in renal events has become a hallmark property of all agents in the SGLT2 inhibitor class both in patients with type 2 diabetes, as well as in those without diabetes but with heart failure with reduced ejection fraction (HFrEF) or with chronic kidney disease.
EMPEROR-Preserved threw a wrench into what had been an unbroken history of renal protection by SGLT2 inhibitors. That happened when a prespecified endpoint of the study – a composite renal outcome defined as time to first occurrence of chronic dialysis, renal transplantation, a sustained reduction of at least 40% in eGFR, or a sustained drop in eGFR of more than 10 or 15 mL/min per 1.73 m2 from baseline – yielded an unexpected neutral finding.
For this composite renal outcome, EMPEROR-Preserved showed a nonsignificant 5% reduction, compared with placebo, a result that both differed from what had been seen in essentially all the other SGLT2 inhibitor trials that had looked at this, but which also seemed at odds with the observed significant preservation of renal function that seemed substantial enough to produce a clinically meaningful benefit.
Renal effects blunted in HFpEF
The immediate upshot was a letter published by several EMPEROR-Preserved investigators that spelled out this discrepancy and came to the jolting conclusion that “eGFR slope analysis has limitations as a surrogate for predicting the effect of drugs on renal outcomes in patients with heart failure.”
The same authors, along with some additional associates, also published a second letter that noted a further unexpected twist with the renal outcome: “In prior large-scale clinical trials, the effect of SGLT2 inhibitors on heart failure and renal outcomes had consistently tracked together,” they noted, but in this case it didn’t, a discordance they said was “extraordinarily puzzling”.
This led the study’s leaders to reanalyze the renal outcomes using a different definition, one that Milton Packer, MD, who helped design the trial and oversaw several of its analyses, called “a more conventional definition of renal events,” during his presentation of these findings at the congress. The researchers swapped out a 40% drop from baseline eGFR as an event and replaced it with a 50% decline, a change designed to screen out less severe, and often transient, reductions in kidney function that have less lasting impact on health. They also added an additional component to the composite endpoint, renal death. A revised analysis using this new renal composite outcome appeared in the European Journal of Heart Failure letter.
This change cut the total number of renal events tallied in the trial nearly in half, down to 112, and showed a more robust decline in renal events with empagliflozin treatment compared with the initial analysis, although the drop remained nonsignificant. The revised analysis also showed that the overall, nonsignificant 22% relative reduction in renal events in patients on empagliflozin, compared with placebo, dwindled down to completely nonexistent in the tertile of patients with a left ventricular ejection fraction of 60% or greater. In this tertile the hazard ratio actually showed a nonsignificant point estimate of a 24% increased rate of renal events on empagliflozin, with the caveat that this subgroup now included a total of just 40 total events between the two treatment arms. (Each of the two other tertiles also had roughly the same number of total events.)
The biggest effect on renal-event reduction was in the tertile of patients with an ejection fraction of 41%-49%, in which empagliflozin treatment was linked with a significant 59% cut in renal events, compared with placebo. The analysis also showed significant heterogeneity in thus outcome between this subgroup and the other two tertiles that had higher ejection fractions and showed reduced rates of protection by empagliflozin against renal events.
This apparent blunting of a renal effect despite preservation of renal function seemed to mimic the blunting of the primary cardiovascular outcome effect that also appeared in patients with ejection fractions in the 60%-65% range or above.
“If we knew what blunted the effect of empagliflozin on heart failure outcomes at higher ejection fraction levels, we think the same explanation may also apply to the blunting of effect on renal outcomes, but right now we do not know the answer to either question,” Dr. Packer said in an interview. He’s suggested that one possibility is that many of the enrolled patients identified as having HFpEF, but with these high ejection fractions may have not actually had HFpEF, and their signs and symptoms may have instead resulted from atrial fibrillation.
“Many patients with an ejection fraction of 60%-65% and above had atrial fibrillation,” he noted, with a prevalence at enrollment in this subgroup of about 50%. Atrial fibrillation can cause dyspnea, a hallmark symptom leading to diagnosis of heart failure, and it also increases levels of N-terminal of the prohormone brain natriuretic peptide, a metric that served as a gatekeeper for entry into the trial. “Essentially, we are saying that many of the criteria that we specified to ensure that patients had heart failure probably did not work very well in patients with an ejection fraction of 65% or greater,” said Dr. Packer, a cardiologist at Baylor University Medical Center in Dallas. “We need to figure out who these patients are.”
Some experts not involved with the study voiced skepticism that the renal findings reflected a real issue.
“I’m quite optimistic that in the long-term the effect on eGFR will translate into renal protection,” said Rudolf A. de Boer, MD, PhD, a professor of translational cardiology at University Medical Center Groningen (the Netherlands), and designated discussant at the congress for the presentation by Dr. Packer.
John J.V. McMurray, MD, a professor of cardiology and a heart failure specialist at Glasgow University, speculated that the unexpected renal outcomes data may relate to the initial decline in renal function produced by treatment with SGLT2 inhibitors despite their longer-term enhancement of renal protection.
“If you use a treatment that protects the kidneys in the long-term but causes an initial dip in eGFR, more patients receiving that treatment will have an early ‘event,’ ” he noted in an interview. He also cautioned about the dangers of subgroup analyses that dice the study population into small cohorts.
“Trials are powered to look at the effect of treatment in the overall population. Everything else is exploratory, underpowered, and subject to the play of chance,” Dr. McMurray stressed.
Counting additional cardiovascular disease events allows more analyses
A third auxiliary report from the EMPEROR-Preserved investigators performed several prespecified analyses that depended on adding additional cardiovascular disease endpoints to the core tallies of cardiovascular death or HHF – such as emergent, urgent, and outpatient events that reflected worsening heart failure – and also included information on diuretic and vasopressor use because of worsening heart failure. The increased event numbers allowed the researchers to perform 30 additional analyses included in this report, according to the count kept by Dr. Packer who was the lead author.
He highlighted several of the additional results in this paper that documented benefits from empagliflozin treatment, compared with placebo:
- A significant 29% reduction in the need for admission to a cardiac care unit or intensive care unit during an HHF.
- A nonsignificant 33% reduction in the need for intravenous vasopressors or positive inotropic drugs during HHF.
- A significantly increased rate of patients achieving a higher New York Heart Association functional class. For example, after the first year of treatment patients who received empagliflozin had a 37% higher rate of functional class improvement, compared with patients who received placebo.
Dr. McMurray had his own list of key takeaways from this paper, including:
- Among patients who needed hospitalization, “those treated with empagliflozin were less sick than those in the placebo group.”
- In addition to reducing HHF empagliflozin treatment also reduced episodes of outpatient worsening as reflected by their receipt of intensified diuretic treatment, which occurred a significant 27% less often, compared with patients on placebo.
- Treatment with empagliflozin also linked with a significant 39% relative reduction in emergency or urgent-care visits that required intravenous therapy.
Empagliflozin’s performance relative to sacubitril/valsartan
The fourth additional report focused on a post hoc, cross-trial comparison of the results from EMPEROR-Preserved and from another recent trial that, like EMPEROR-Preserved, assessed in patients with HFpEF a drug previously proven to work quite well in patients with HFrEF. The comparator drug was sacubitril/valsartan (Entresto), which underwent testing in patients with HFpEF in the PARAGON-HF trial.
The primary outcome of PARAGON-HF, which randomized 4,822 patients, was reduction in cardiovascular death and in total HHF. This dropped by a relative 13%, compared with placebo, during a median of 35 months, a between-group difference that came close to but did not achieve significance (P = .06). Despite this limitation, the Food and Drug Administration in February 2021 loosened the indication for using sacubitril/valsartan in patients with heart failure and a “below normal” ejection fraction, a category that can include many patients considered to have HFpEF.
Although the researchers who ran this analysis, including Dr. Packer, who was the first author, admitted that “comparison of effect sizes across trials is fraught with difficulties,” they nonetheless concluded from their analysis that “for all outcomes that included HHF the effect size was larger for empagliflozin than for sacubitril/valsartan.”
Dr. McMurray, a lead instigator for PARAGON-HF, said there was little to take away from this analysis.
“The patient populations were different, and sacubitril/valsartan was compared against an active therapy, valsartan,” while in EMPEROR-Preserved empagliflozin compared against placebo. “Most of us believe that sacubitril/valsartan and SGLT2 inhibitors work in different but complementary ways, and their benefits are additive. You would want patients with HFpEF or HFrEF to take both,” he said in an interview.
Dr. Packer agreed with that approach and added that he would probably also prescribe a third agent, spironolactone, to many patients with HFpEF.
EMPEROR-Preserved was sponsored by Boehringer Ingelheim and Eli Lilly, which jointly market empagliflozin (Jardiance). PARAGON-HF was sponsored by Novartis, which markets sacubitril/valsartan (Entresto). Dr. Packer has received consulting fees from Boehringer Ingelheim and from numerous other companies. Dr. de Boer has research contracts with Boehringer Ingelheim as well as from Abbott, AstraZeneca, Cardior, Ionis, Novo Nordisk, and Roche, and he has been a consultant to Novartis as well as to Abbott, AstraZeneca, Gayer, and Roche. Dr. McMurray led trials of sacubitril/valsartan sponsored by Novartis, and his institution has received compensation for his participation in studies sponsored by Abbvie, AstraZeneca, Cardurion, DalCor, GlaxoSmithKline, Pfizer, and Theracos.
The featured report from the 6,000-patient EMPEROR-Preserved trial at the virtual annual congress of the European Society of Cardiology drew lots of attention for its headline finding: the first unequivocal demonstration that a medication, empagliflozin, can significantly reduce the rate of cardiovascular death and hospitalization for heart failure in patients with heart failure with preserved ejection fraction (HFpEF, a left ventricular ejection fraction of more than 40%), with the details simultaneously published online.
But at the same time, the EMPEROR-Preserved investigators released four additional reports with a lot more outcome analyses that also deserve some attention.
The puzzling neutral effect on renal events
Perhaps the most surprising and complicated set of findings among the main EMPEROR-Preserved outcomes involved renal outcomes.
The trial’s primary outcome was the combined rate of cardiovascular death or hospitalization for heart failure (HHF), and the results showed that treatment with empagliflozin (Jardiance) for a median of 26 months on top of standard treatment for patients with HFpEF led to a significant 21% relative risk reduction, compared with placebo-treated patients.
The trial had two prespecified secondary outcomes. One was the total number of HHF, which dropped by a significant 27%, compared with placebo. The second was the mean change in slope of estimated glomerular filtration rate (eGFR) on an annualized basis, and the empagliflozin regimen reduced the cumulative annual deficit, compared with placebo by an average of 1.36 mL/min per 1.73 m2, a significant difference.
This preservation of renal function was consistent with results from many prior studies of empagliflozin and all of the other U.S.-approved agents from the sodium-glucose cotransporter 2 inhibitor class. Preservation of renal function and a reduction in renal events has become a hallmark property of all agents in the SGLT2 inhibitor class both in patients with type 2 diabetes, as well as in those without diabetes but with heart failure with reduced ejection fraction (HFrEF) or with chronic kidney disease.
EMPEROR-Preserved threw a wrench into what had been an unbroken history of renal protection by SGLT2 inhibitors. That happened when a prespecified endpoint of the study – a composite renal outcome defined as time to first occurrence of chronic dialysis, renal transplantation, a sustained reduction of at least 40% in eGFR, or a sustained drop in eGFR of more than 10 or 15 mL/min per 1.73 m2 from baseline – yielded an unexpected neutral finding.
For this composite renal outcome, EMPEROR-Preserved showed a nonsignificant 5% reduction, compared with placebo, a result that both differed from what had been seen in essentially all the other SGLT2 inhibitor trials that had looked at this, but which also seemed at odds with the observed significant preservation of renal function that seemed substantial enough to produce a clinically meaningful benefit.
Renal effects blunted in HFpEF
The immediate upshot was a letter published by several EMPEROR-Preserved investigators that spelled out this discrepancy and came to the jolting conclusion that “eGFR slope analysis has limitations as a surrogate for predicting the effect of drugs on renal outcomes in patients with heart failure.”
The same authors, along with some additional associates, also published a second letter that noted a further unexpected twist with the renal outcome: “In prior large-scale clinical trials, the effect of SGLT2 inhibitors on heart failure and renal outcomes had consistently tracked together,” they noted, but in this case it didn’t, a discordance they said was “extraordinarily puzzling”.
This led the study’s leaders to reanalyze the renal outcomes using a different definition, one that Milton Packer, MD, who helped design the trial and oversaw several of its analyses, called “a more conventional definition of renal events,” during his presentation of these findings at the congress. The researchers swapped out a 40% drop from baseline eGFR as an event and replaced it with a 50% decline, a change designed to screen out less severe, and often transient, reductions in kidney function that have less lasting impact on health. They also added an additional component to the composite endpoint, renal death. A revised analysis using this new renal composite outcome appeared in the European Journal of Heart Failure letter.
This change cut the total number of renal events tallied in the trial nearly in half, down to 112, and showed a more robust decline in renal events with empagliflozin treatment compared with the initial analysis, although the drop remained nonsignificant. The revised analysis also showed that the overall, nonsignificant 22% relative reduction in renal events in patients on empagliflozin, compared with placebo, dwindled down to completely nonexistent in the tertile of patients with a left ventricular ejection fraction of 60% or greater. In this tertile the hazard ratio actually showed a nonsignificant point estimate of a 24% increased rate of renal events on empagliflozin, with the caveat that this subgroup now included a total of just 40 total events between the two treatment arms. (Each of the two other tertiles also had roughly the same number of total events.)
The biggest effect on renal-event reduction was in the tertile of patients with an ejection fraction of 41%-49%, in which empagliflozin treatment was linked with a significant 59% cut in renal events, compared with placebo. The analysis also showed significant heterogeneity in thus outcome between this subgroup and the other two tertiles that had higher ejection fractions and showed reduced rates of protection by empagliflozin against renal events.
This apparent blunting of a renal effect despite preservation of renal function seemed to mimic the blunting of the primary cardiovascular outcome effect that also appeared in patients with ejection fractions in the 60%-65% range or above.
“If we knew what blunted the effect of empagliflozin on heart failure outcomes at higher ejection fraction levels, we think the same explanation may also apply to the blunting of effect on renal outcomes, but right now we do not know the answer to either question,” Dr. Packer said in an interview. He’s suggested that one possibility is that many of the enrolled patients identified as having HFpEF, but with these high ejection fractions may have not actually had HFpEF, and their signs and symptoms may have instead resulted from atrial fibrillation.
“Many patients with an ejection fraction of 60%-65% and above had atrial fibrillation,” he noted, with a prevalence at enrollment in this subgroup of about 50%. Atrial fibrillation can cause dyspnea, a hallmark symptom leading to diagnosis of heart failure, and it also increases levels of N-terminal of the prohormone brain natriuretic peptide, a metric that served as a gatekeeper for entry into the trial. “Essentially, we are saying that many of the criteria that we specified to ensure that patients had heart failure probably did not work very well in patients with an ejection fraction of 65% or greater,” said Dr. Packer, a cardiologist at Baylor University Medical Center in Dallas. “We need to figure out who these patients are.”
Some experts not involved with the study voiced skepticism that the renal findings reflected a real issue.
“I’m quite optimistic that in the long-term the effect on eGFR will translate into renal protection,” said Rudolf A. de Boer, MD, PhD, a professor of translational cardiology at University Medical Center Groningen (the Netherlands), and designated discussant at the congress for the presentation by Dr. Packer.
John J.V. McMurray, MD, a professor of cardiology and a heart failure specialist at Glasgow University, speculated that the unexpected renal outcomes data may relate to the initial decline in renal function produced by treatment with SGLT2 inhibitors despite their longer-term enhancement of renal protection.
“If you use a treatment that protects the kidneys in the long-term but causes an initial dip in eGFR, more patients receiving that treatment will have an early ‘event,’ ” he noted in an interview. He also cautioned about the dangers of subgroup analyses that dice the study population into small cohorts.
“Trials are powered to look at the effect of treatment in the overall population. Everything else is exploratory, underpowered, and subject to the play of chance,” Dr. McMurray stressed.
Counting additional cardiovascular disease events allows more analyses
A third auxiliary report from the EMPEROR-Preserved investigators performed several prespecified analyses that depended on adding additional cardiovascular disease endpoints to the core tallies of cardiovascular death or HHF – such as emergent, urgent, and outpatient events that reflected worsening heart failure – and also included information on diuretic and vasopressor use because of worsening heart failure. The increased event numbers allowed the researchers to perform 30 additional analyses included in this report, according to the count kept by Dr. Packer who was the lead author.
He highlighted several of the additional results in this paper that documented benefits from empagliflozin treatment, compared with placebo:
- A significant 29% reduction in the need for admission to a cardiac care unit or intensive care unit during an HHF.
- A nonsignificant 33% reduction in the need for intravenous vasopressors or positive inotropic drugs during HHF.
- A significantly increased rate of patients achieving a higher New York Heart Association functional class. For example, after the first year of treatment patients who received empagliflozin had a 37% higher rate of functional class improvement, compared with patients who received placebo.
Dr. McMurray had his own list of key takeaways from this paper, including:
- Among patients who needed hospitalization, “those treated with empagliflozin were less sick than those in the placebo group.”
- In addition to reducing HHF empagliflozin treatment also reduced episodes of outpatient worsening as reflected by their receipt of intensified diuretic treatment, which occurred a significant 27% less often, compared with patients on placebo.
- Treatment with empagliflozin also linked with a significant 39% relative reduction in emergency or urgent-care visits that required intravenous therapy.
Empagliflozin’s performance relative to sacubitril/valsartan
The fourth additional report focused on a post hoc, cross-trial comparison of the results from EMPEROR-Preserved and from another recent trial that, like EMPEROR-Preserved, assessed in patients with HFpEF a drug previously proven to work quite well in patients with HFrEF. The comparator drug was sacubitril/valsartan (Entresto), which underwent testing in patients with HFpEF in the PARAGON-HF trial.
The primary outcome of PARAGON-HF, which randomized 4,822 patients, was reduction in cardiovascular death and in total HHF. This dropped by a relative 13%, compared with placebo, during a median of 35 months, a between-group difference that came close to but did not achieve significance (P = .06). Despite this limitation, the Food and Drug Administration in February 2021 loosened the indication for using sacubitril/valsartan in patients with heart failure and a “below normal” ejection fraction, a category that can include many patients considered to have HFpEF.
Although the researchers who ran this analysis, including Dr. Packer, who was the first author, admitted that “comparison of effect sizes across trials is fraught with difficulties,” they nonetheless concluded from their analysis that “for all outcomes that included HHF the effect size was larger for empagliflozin than for sacubitril/valsartan.”
Dr. McMurray, a lead instigator for PARAGON-HF, said there was little to take away from this analysis.
“The patient populations were different, and sacubitril/valsartan was compared against an active therapy, valsartan,” while in EMPEROR-Preserved empagliflozin compared against placebo. “Most of us believe that sacubitril/valsartan and SGLT2 inhibitors work in different but complementary ways, and their benefits are additive. You would want patients with HFpEF or HFrEF to take both,” he said in an interview.
Dr. Packer agreed with that approach and added that he would probably also prescribe a third agent, spironolactone, to many patients with HFpEF.
EMPEROR-Preserved was sponsored by Boehringer Ingelheim and Eli Lilly, which jointly market empagliflozin (Jardiance). PARAGON-HF was sponsored by Novartis, which markets sacubitril/valsartan (Entresto). Dr. Packer has received consulting fees from Boehringer Ingelheim and from numerous other companies. Dr. de Boer has research contracts with Boehringer Ingelheim as well as from Abbott, AstraZeneca, Cardior, Ionis, Novo Nordisk, and Roche, and he has been a consultant to Novartis as well as to Abbott, AstraZeneca, Gayer, and Roche. Dr. McMurray led trials of sacubitril/valsartan sponsored by Novartis, and his institution has received compensation for his participation in studies sponsored by Abbvie, AstraZeneca, Cardurion, DalCor, GlaxoSmithKline, Pfizer, and Theracos.
FROM ESC 2021
Alcohol ups risk for atrial fibrillation episode hours later
Consuming alcohol increases the risk for an atrial fibrillation (AF) episode hours later, according to a study published online Aug. 30 in Annals of Internal Medicine.
Past research has associated long-term alcohol consumption with the development of AF, and abstinence from alcohol has been associated with a lower overall AF burden. However, lead study author Greg Marcus, MD, a cardioelectrophysiolgist at the University of California, San Francisco, noted that many patients say that alcohol is a trigger for discrete AF episodes.
To test whether that was possible, the researchers enrolled 100 patients who had a history of AF events and who consumed at least one drink per month. Participants wore a transdermal alcohol sensor and an ambulatory, single-lead electrocardiogram device for 4 weeks. They were instructed to press a button on the electrocardiogram device each time they consumed a standard alcoholic beverage. In addition, blood samples were tested for phosphatidylethanol (PEth) at the participants’ 2-week and 4-week visits. PEth is a phospholipid formed in the blood after alcohol intake. It remains in the blood for up to 4 weeks after alcohol consumption.
The study findings confirmed what the patients had reported. The odds of an AF episode were 38% greater with every 0.1% increase in peak blood alcohol concentration over the previous 12 hours (odds ratio [OR], 1.38; 95% confidence interval, 1.04-1.83; P = .024). Moreover, an episode of AF was associated with twofold greater odds (OR, 2.02; 95% CI, 1.38-3.17) of having consumed one alcoholic drink in the past 4 hours. It was associated with more than threefold greater odds of having consumed two or more drinks (OR, 3.58; 95% CI, 1.63-7.89).
“The major takeaway is, among atrial fibrillation patients, consuming alcohol substantially heightened their risk for any given atrial fibrillation event in the subsequent few hours,” Dr. Marcus said. “The more alcohol consumed, the higher that risk.”
The acute effect of alcohol on these arrhythmias also means that modifying alcohol consumption could immediately benefit some patients. “These data combined with other evidence suggest that recommending minimizing or completely eliminating alcohol will likely be helpful to them,” Dr. Marcus said.
The study’s reliance on wearables and sensors was impressive, said Mariann R. Piano, PhD, director of the Center for Research Development and Scholarship, Vanderbilt University, Nashville, Tenn. Often, these types of studies are “self-reported and confounded by recall bias,” she said. But this study passively documented arrhythmia events and blood alcohol level without any patient input. The additional measures of alcohol consumption were used to validate the blood alcohol sensor.
The study’s focus on patients with a history of AF highlighted a high-risk patient group, according to Dr. Piano, who coauthored an editorial about the study. However, the findings may not be applicable to the general population.
Dr. Marcus said alcohol’s role in causing these types of arrhythmias is probably a matter of degree. AF patients are more prone to events than is the general population and are therefore more sensitive to alcohol, he said. But excessive alcohol consumption could increase the chance of AF in the general population.
The study is not without its limitations, however. For instance, “it would have been really ideal if we knew what that blood alcohol was” before an episode, Dr. Piano said. The number of drinks is a good start, but two drinks can affect persons differently, depending on their weight and height. Also, baseline PEth values suggest that patients had been drinking before the study, she said. Ideally, patients could have been asked to abstain from alcohol for a period before the study to determine a negative baseline PEth value and minimize the effects of previous drinking on AF episodes.
Moving forward, this research should inform how clinicians care for their AF patients, both experts agree. “We need to talk to patients about how much they drink,” Dr. Piano said. In addition, patients should be advised to closely monitor what they’re drinking.
“This definitely sharpens the focus of the importance of a thorough alcohol history when we see an atrial fibrillation patient and to counsel them to reduce or eliminate alcohol, even among those that don’t have alcohol use disorders,” Dr. Marcus said.
Preliminary results of the study were presented as a late-breaking clinical trials presentation at the American College of Cardiology meeting in May.
Dr. Marcus has received grants from Baylis, Jawbone, and Eight Sleep and has received personal fees from InCarda and Johnson & Johnson. Coauthors have received personal fees from VivaLNK, Huba Pharmaceuticals, Johnson & Johnson, and Merck and grants from Samsung and Amgen Inc. The editorialists have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Consuming alcohol increases the risk for an atrial fibrillation (AF) episode hours later, according to a study published online Aug. 30 in Annals of Internal Medicine.
Past research has associated long-term alcohol consumption with the development of AF, and abstinence from alcohol has been associated with a lower overall AF burden. However, lead study author Greg Marcus, MD, a cardioelectrophysiolgist at the University of California, San Francisco, noted that many patients say that alcohol is a trigger for discrete AF episodes.
To test whether that was possible, the researchers enrolled 100 patients who had a history of AF events and who consumed at least one drink per month. Participants wore a transdermal alcohol sensor and an ambulatory, single-lead electrocardiogram device for 4 weeks. They were instructed to press a button on the electrocardiogram device each time they consumed a standard alcoholic beverage. In addition, blood samples were tested for phosphatidylethanol (PEth) at the participants’ 2-week and 4-week visits. PEth is a phospholipid formed in the blood after alcohol intake. It remains in the blood for up to 4 weeks after alcohol consumption.
The study findings confirmed what the patients had reported. The odds of an AF episode were 38% greater with every 0.1% increase in peak blood alcohol concentration over the previous 12 hours (odds ratio [OR], 1.38; 95% confidence interval, 1.04-1.83; P = .024). Moreover, an episode of AF was associated with twofold greater odds (OR, 2.02; 95% CI, 1.38-3.17) of having consumed one alcoholic drink in the past 4 hours. It was associated with more than threefold greater odds of having consumed two or more drinks (OR, 3.58; 95% CI, 1.63-7.89).
“The major takeaway is, among atrial fibrillation patients, consuming alcohol substantially heightened their risk for any given atrial fibrillation event in the subsequent few hours,” Dr. Marcus said. “The more alcohol consumed, the higher that risk.”
The acute effect of alcohol on these arrhythmias also means that modifying alcohol consumption could immediately benefit some patients. “These data combined with other evidence suggest that recommending minimizing or completely eliminating alcohol will likely be helpful to them,” Dr. Marcus said.
The study’s reliance on wearables and sensors was impressive, said Mariann R. Piano, PhD, director of the Center for Research Development and Scholarship, Vanderbilt University, Nashville, Tenn. Often, these types of studies are “self-reported and confounded by recall bias,” she said. But this study passively documented arrhythmia events and blood alcohol level without any patient input. The additional measures of alcohol consumption were used to validate the blood alcohol sensor.
The study’s focus on patients with a history of AF highlighted a high-risk patient group, according to Dr. Piano, who coauthored an editorial about the study. However, the findings may not be applicable to the general population.
Dr. Marcus said alcohol’s role in causing these types of arrhythmias is probably a matter of degree. AF patients are more prone to events than is the general population and are therefore more sensitive to alcohol, he said. But excessive alcohol consumption could increase the chance of AF in the general population.
The study is not without its limitations, however. For instance, “it would have been really ideal if we knew what that blood alcohol was” before an episode, Dr. Piano said. The number of drinks is a good start, but two drinks can affect persons differently, depending on their weight and height. Also, baseline PEth values suggest that patients had been drinking before the study, she said. Ideally, patients could have been asked to abstain from alcohol for a period before the study to determine a negative baseline PEth value and minimize the effects of previous drinking on AF episodes.
Moving forward, this research should inform how clinicians care for their AF patients, both experts agree. “We need to talk to patients about how much they drink,” Dr. Piano said. In addition, patients should be advised to closely monitor what they’re drinking.
“This definitely sharpens the focus of the importance of a thorough alcohol history when we see an atrial fibrillation patient and to counsel them to reduce or eliminate alcohol, even among those that don’t have alcohol use disorders,” Dr. Marcus said.
Preliminary results of the study were presented as a late-breaking clinical trials presentation at the American College of Cardiology meeting in May.
Dr. Marcus has received grants from Baylis, Jawbone, and Eight Sleep and has received personal fees from InCarda and Johnson & Johnson. Coauthors have received personal fees from VivaLNK, Huba Pharmaceuticals, Johnson & Johnson, and Merck and grants from Samsung and Amgen Inc. The editorialists have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Consuming alcohol increases the risk for an atrial fibrillation (AF) episode hours later, according to a study published online Aug. 30 in Annals of Internal Medicine.
Past research has associated long-term alcohol consumption with the development of AF, and abstinence from alcohol has been associated with a lower overall AF burden. However, lead study author Greg Marcus, MD, a cardioelectrophysiolgist at the University of California, San Francisco, noted that many patients say that alcohol is a trigger for discrete AF episodes.
To test whether that was possible, the researchers enrolled 100 patients who had a history of AF events and who consumed at least one drink per month. Participants wore a transdermal alcohol sensor and an ambulatory, single-lead electrocardiogram device for 4 weeks. They were instructed to press a button on the electrocardiogram device each time they consumed a standard alcoholic beverage. In addition, blood samples were tested for phosphatidylethanol (PEth) at the participants’ 2-week and 4-week visits. PEth is a phospholipid formed in the blood after alcohol intake. It remains in the blood for up to 4 weeks after alcohol consumption.
The study findings confirmed what the patients had reported. The odds of an AF episode were 38% greater with every 0.1% increase in peak blood alcohol concentration over the previous 12 hours (odds ratio [OR], 1.38; 95% confidence interval, 1.04-1.83; P = .024). Moreover, an episode of AF was associated with twofold greater odds (OR, 2.02; 95% CI, 1.38-3.17) of having consumed one alcoholic drink in the past 4 hours. It was associated with more than threefold greater odds of having consumed two or more drinks (OR, 3.58; 95% CI, 1.63-7.89).
“The major takeaway is, among atrial fibrillation patients, consuming alcohol substantially heightened their risk for any given atrial fibrillation event in the subsequent few hours,” Dr. Marcus said. “The more alcohol consumed, the higher that risk.”
The acute effect of alcohol on these arrhythmias also means that modifying alcohol consumption could immediately benefit some patients. “These data combined with other evidence suggest that recommending minimizing or completely eliminating alcohol will likely be helpful to them,” Dr. Marcus said.
The study’s reliance on wearables and sensors was impressive, said Mariann R. Piano, PhD, director of the Center for Research Development and Scholarship, Vanderbilt University, Nashville, Tenn. Often, these types of studies are “self-reported and confounded by recall bias,” she said. But this study passively documented arrhythmia events and blood alcohol level without any patient input. The additional measures of alcohol consumption were used to validate the blood alcohol sensor.
The study’s focus on patients with a history of AF highlighted a high-risk patient group, according to Dr. Piano, who coauthored an editorial about the study. However, the findings may not be applicable to the general population.
Dr. Marcus said alcohol’s role in causing these types of arrhythmias is probably a matter of degree. AF patients are more prone to events than is the general population and are therefore more sensitive to alcohol, he said. But excessive alcohol consumption could increase the chance of AF in the general population.
The study is not without its limitations, however. For instance, “it would have been really ideal if we knew what that blood alcohol was” before an episode, Dr. Piano said. The number of drinks is a good start, but two drinks can affect persons differently, depending on their weight and height. Also, baseline PEth values suggest that patients had been drinking before the study, she said. Ideally, patients could have been asked to abstain from alcohol for a period before the study to determine a negative baseline PEth value and minimize the effects of previous drinking on AF episodes.
Moving forward, this research should inform how clinicians care for their AF patients, both experts agree. “We need to talk to patients about how much they drink,” Dr. Piano said. In addition, patients should be advised to closely monitor what they’re drinking.
“This definitely sharpens the focus of the importance of a thorough alcohol history when we see an atrial fibrillation patient and to counsel them to reduce or eliminate alcohol, even among those that don’t have alcohol use disorders,” Dr. Marcus said.
Preliminary results of the study were presented as a late-breaking clinical trials presentation at the American College of Cardiology meeting in May.
Dr. Marcus has received grants from Baylis, Jawbone, and Eight Sleep and has received personal fees from InCarda and Johnson & Johnson. Coauthors have received personal fees from VivaLNK, Huba Pharmaceuticals, Johnson & Johnson, and Merck and grants from Samsung and Amgen Inc. The editorialists have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
ACST-2: Carotid stenting, surgery on par in asymptomatic patients
Carotid artery stenting (CAS) and carotid endarterectomy (CEA) provided comparable outcomes over time in asymptomatic patients receiving good medical therapy in the largest trial to date of what to do with severe carotid artery narrowing that is yet to cause a stroke.
Among more than 3,600 patients, stenting and surgery performed by experienced physicians involved a 1.0% risk for causing disabling stroke or death within 30 days.
The annual rate of fatal or disabling strokes was about 0.5% with either procedure over an average 5 years’ follow-up – essentially halving the annual stroke risk had neither procedure been performed, according to Alison Halliday, MD, principal investigator of the Asymptomatic Carotid Surgery Trial-2 (ACST-2).
The results were reported Aug. 29 in a Hot Line session at the virtual annual congress of the European Society of Cardiology and published simultaneously online in The Lancet.
Session chair Gilles Montalescot, MD, Sorbonne University, Paris, noted that ACST-2 doubled the number of randomly assigned patients with asymptomatic carotid stenosis studied in previous trials, “so, a huge contribution to the evidence base in this field and apparently good news for both revascularization techniques.”
Thirty-day and 5-year outcomes
The trial was conducted in 33 countries between January 2008 and December 2020, enrolling 3,625 patients (70% were male; mean age, 70 years) with carotid stenosis of at least 60% on ultrasonography, in whom stenting or surgery was suitable but both the doctor and patient were “substantially uncertain” which procedure to prefer.
Among the 1,811 patients assigned to stenting, 87% underwent the procedure at a median of 14 days; 6% crossed over to surgery, typically because of a highly calcified lesion or a more tortuous carotid than anticipated; and 6% had no intervention.
Among the 1,814 patients assigned to surgery, 92% had the procedure at a median of 14 days; 3% crossed over to stenting, typically because of patient or doctor preference or reluctance to undergo general anesthesia; and 4% had no intervention.
Patients without complications who had stenting stayed on average 1 day less than did those undergoing surgery.
During an earlier press briefing, Dr. Halliday highlighted the need for procedural competency and said doctors had to submit a record of their CEA or CAS experience and, consistent with current guidelines, had to demonstrate an independently verified stroke or death rate of 6% or less for symptomatic patients and 3% or lower for asymptomatic patients.
The results showed the 30-day risk for death, myocardial infarction (MI), or any stroke was 3.9% with carotid stenting and 3.2% with surgery (P = .26).
But with stenting, there was a slightly higher risk for procedural nondisabling strokes (48 vs. 29; P = .03), including 15 strokes vs. 5 strokes, respectively, that left patients with no residual symptoms. This is “consistent with large, recent nationally representative registry data,” observed Dr. Halliday, of the University of Oxford (England).
For those undergoing surgery, cranial nerve palsies were reported in 5.4% vs. no patients undergoing stenting.
At 5 years, the nonprocedural fatal or disabling stroke rate was 2.5% in each group (rate ratio [RR], 0.98; P = .91), with any nonprocedural stroke occurring in 5.3% of patients with stenting vs. 4.5% with surgery (RR, 1.16; P = .33).
The investigators performed a meta-analysis combining the ACST-2 results with those of eight prior trials (four in asymptomatic and four in symptomatic patients) that yielded a similar nonsignificant result for any nonprocedural stroke (RR, 1.11; P = .21).
Based on the results from ACST-2 plus the major trials, stenting and surgery involve “similar risks and similar benefits,” Dr. Halliday concluded.
Discussant Marco Roffi, MD, University Hospital of Geneva, said, “In centers with documented expertise, carotid artery stenting should be offered as an alternative to carotid endarterectomy in patients with asymptomatic stenosis and suitable anatomy.”
While the trial provides “good news” for patients, he pointed out that a reduction in the sample size from 5,000 to 3,625 limited the statistical power and that enrollment over a long period of time may have introduced confounders, such as changes in equipment technique, and medical therapy.
Also, many centers enrolled few patients, raising the concern over low-volume centers and operators, Dr. Roffi said. “We know that 8% of the centers enrolled 39% of the patients,” and “information on the credentialing and experience of the interventionalists was limited.”
Further, a lack of systematic MI assessment may have favored the surgery group, and more recent developments in stenting with the potential of reducing periprocedural stroke were rarely used, such as proximal emboli protection in only 15% and double-layer stents in 11%.
Friedhelm Beyersdorf, MD, University Hospital of Freiburg, Germany, said that, as a vascular surgeon, he finds it understandable that there might be a higher incidence of nonfatal strokes when treating carotid stenosis with stents, given the vulnerability of these lesions.
“Nevertheless, the main conclusion from the entire study is that carotid artery treatment is extremely safe, it has to be done in order to avoid strokes, and, obviously, there seems to be an advantage for surgery in terms of nondisabling stroke,” he said.
Session chair Dr. Montalescot, however, said that what the study cannot address – and what was the subject of many online audience comments – is whether either intervention should be performed in these patients.
Unlike earlier trials comparing interventions to medical therapy, Dr. Halliday said ACST-2 enrolled patients for whom the decision had been made that revascularization was needed. In addition, 99%-100% were receiving antithrombotic therapy at baseline, 85%-90% were receiving antihypertensives, and about 85% were taking statins.
Longer-term follow-up should provide a better picture of the nonprocedural stroke risk, with patients asked annually about exactly what medications and doses they are taking, she said.
“We will have an enormous list of exactly what’s gone on and the intensity of that therapy, which is, of course, much more intense than when we carried out our first trial. But these were people in whom a procedure was thought to be necessary,” she noted.
When asked during the press conference which procedure she would choose, Dr. Halliday, a surgeon, observed that patient preference is important but that the nature of the lesion itself often determines the optimal choice.
“If you know the competence of the people doing it is equal, then the less invasive procedure – providing it has good long-term viability, and that’s why we’re following for 10 years – is the more important,” she added.
The study was funded by the UK Medical Research Council and Health Technology Assessment Programme. Dr. Halliday reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Carotid artery stenting (CAS) and carotid endarterectomy (CEA) provided comparable outcomes over time in asymptomatic patients receiving good medical therapy in the largest trial to date of what to do with severe carotid artery narrowing that is yet to cause a stroke.
Among more than 3,600 patients, stenting and surgery performed by experienced physicians involved a 1.0% risk for causing disabling stroke or death within 30 days.
The annual rate of fatal or disabling strokes was about 0.5% with either procedure over an average 5 years’ follow-up – essentially halving the annual stroke risk had neither procedure been performed, according to Alison Halliday, MD, principal investigator of the Asymptomatic Carotid Surgery Trial-2 (ACST-2).
The results were reported Aug. 29 in a Hot Line session at the virtual annual congress of the European Society of Cardiology and published simultaneously online in The Lancet.
Session chair Gilles Montalescot, MD, Sorbonne University, Paris, noted that ACST-2 doubled the number of randomly assigned patients with asymptomatic carotid stenosis studied in previous trials, “so, a huge contribution to the evidence base in this field and apparently good news for both revascularization techniques.”
Thirty-day and 5-year outcomes
The trial was conducted in 33 countries between January 2008 and December 2020, enrolling 3,625 patients (70% were male; mean age, 70 years) with carotid stenosis of at least 60% on ultrasonography, in whom stenting or surgery was suitable but both the doctor and patient were “substantially uncertain” which procedure to prefer.
Among the 1,811 patients assigned to stenting, 87% underwent the procedure at a median of 14 days; 6% crossed over to surgery, typically because of a highly calcified lesion or a more tortuous carotid than anticipated; and 6% had no intervention.
Among the 1,814 patients assigned to surgery, 92% had the procedure at a median of 14 days; 3% crossed over to stenting, typically because of patient or doctor preference or reluctance to undergo general anesthesia; and 4% had no intervention.
Patients without complications who had stenting stayed on average 1 day less than did those undergoing surgery.
During an earlier press briefing, Dr. Halliday highlighted the need for procedural competency and said doctors had to submit a record of their CEA or CAS experience and, consistent with current guidelines, had to demonstrate an independently verified stroke or death rate of 6% or less for symptomatic patients and 3% or lower for asymptomatic patients.
The results showed the 30-day risk for death, myocardial infarction (MI), or any stroke was 3.9% with carotid stenting and 3.2% with surgery (P = .26).
But with stenting, there was a slightly higher risk for procedural nondisabling strokes (48 vs. 29; P = .03), including 15 strokes vs. 5 strokes, respectively, that left patients with no residual symptoms. This is “consistent with large, recent nationally representative registry data,” observed Dr. Halliday, of the University of Oxford (England).
For those undergoing surgery, cranial nerve palsies were reported in 5.4% vs. no patients undergoing stenting.
At 5 years, the nonprocedural fatal or disabling stroke rate was 2.5% in each group (rate ratio [RR], 0.98; P = .91), with any nonprocedural stroke occurring in 5.3% of patients with stenting vs. 4.5% with surgery (RR, 1.16; P = .33).
The investigators performed a meta-analysis combining the ACST-2 results with those of eight prior trials (four in asymptomatic and four in symptomatic patients) that yielded a similar nonsignificant result for any nonprocedural stroke (RR, 1.11; P = .21).
Based on the results from ACST-2 plus the major trials, stenting and surgery involve “similar risks and similar benefits,” Dr. Halliday concluded.
Discussant Marco Roffi, MD, University Hospital of Geneva, said, “In centers with documented expertise, carotid artery stenting should be offered as an alternative to carotid endarterectomy in patients with asymptomatic stenosis and suitable anatomy.”
While the trial provides “good news” for patients, he pointed out that a reduction in the sample size from 5,000 to 3,625 limited the statistical power and that enrollment over a long period of time may have introduced confounders, such as changes in equipment technique, and medical therapy.
Also, many centers enrolled few patients, raising the concern over low-volume centers and operators, Dr. Roffi said. “We know that 8% of the centers enrolled 39% of the patients,” and “information on the credentialing and experience of the interventionalists was limited.”
Further, a lack of systematic MI assessment may have favored the surgery group, and more recent developments in stenting with the potential of reducing periprocedural stroke were rarely used, such as proximal emboli protection in only 15% and double-layer stents in 11%.
Friedhelm Beyersdorf, MD, University Hospital of Freiburg, Germany, said that, as a vascular surgeon, he finds it understandable that there might be a higher incidence of nonfatal strokes when treating carotid stenosis with stents, given the vulnerability of these lesions.
“Nevertheless, the main conclusion from the entire study is that carotid artery treatment is extremely safe, it has to be done in order to avoid strokes, and, obviously, there seems to be an advantage for surgery in terms of nondisabling stroke,” he said.
Session chair Dr. Montalescot, however, said that what the study cannot address – and what was the subject of many online audience comments – is whether either intervention should be performed in these patients.
Unlike earlier trials comparing interventions to medical therapy, Dr. Halliday said ACST-2 enrolled patients for whom the decision had been made that revascularization was needed. In addition, 99%-100% were receiving antithrombotic therapy at baseline, 85%-90% were receiving antihypertensives, and about 85% were taking statins.
Longer-term follow-up should provide a better picture of the nonprocedural stroke risk, with patients asked annually about exactly what medications and doses they are taking, she said.
“We will have an enormous list of exactly what’s gone on and the intensity of that therapy, which is, of course, much more intense than when we carried out our first trial. But these were people in whom a procedure was thought to be necessary,” she noted.
When asked during the press conference which procedure she would choose, Dr. Halliday, a surgeon, observed that patient preference is important but that the nature of the lesion itself often determines the optimal choice.
“If you know the competence of the people doing it is equal, then the less invasive procedure – providing it has good long-term viability, and that’s why we’re following for 10 years – is the more important,” she added.
The study was funded by the UK Medical Research Council and Health Technology Assessment Programme. Dr. Halliday reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Carotid artery stenting (CAS) and carotid endarterectomy (CEA) provided comparable outcomes over time in asymptomatic patients receiving good medical therapy in the largest trial to date of what to do with severe carotid artery narrowing that is yet to cause a stroke.
Among more than 3,600 patients, stenting and surgery performed by experienced physicians involved a 1.0% risk for causing disabling stroke or death within 30 days.
The annual rate of fatal or disabling strokes was about 0.5% with either procedure over an average 5 years’ follow-up – essentially halving the annual stroke risk had neither procedure been performed, according to Alison Halliday, MD, principal investigator of the Asymptomatic Carotid Surgery Trial-2 (ACST-2).
The results were reported Aug. 29 in a Hot Line session at the virtual annual congress of the European Society of Cardiology and published simultaneously online in The Lancet.
Session chair Gilles Montalescot, MD, Sorbonne University, Paris, noted that ACST-2 doubled the number of randomly assigned patients with asymptomatic carotid stenosis studied in previous trials, “so, a huge contribution to the evidence base in this field and apparently good news for both revascularization techniques.”
Thirty-day and 5-year outcomes
The trial was conducted in 33 countries between January 2008 and December 2020, enrolling 3,625 patients (70% were male; mean age, 70 years) with carotid stenosis of at least 60% on ultrasonography, in whom stenting or surgery was suitable but both the doctor and patient were “substantially uncertain” which procedure to prefer.
Among the 1,811 patients assigned to stenting, 87% underwent the procedure at a median of 14 days; 6% crossed over to surgery, typically because of a highly calcified lesion or a more tortuous carotid than anticipated; and 6% had no intervention.
Among the 1,814 patients assigned to surgery, 92% had the procedure at a median of 14 days; 3% crossed over to stenting, typically because of patient or doctor preference or reluctance to undergo general anesthesia; and 4% had no intervention.
Patients without complications who had stenting stayed on average 1 day less than did those undergoing surgery.
During an earlier press briefing, Dr. Halliday highlighted the need for procedural competency and said doctors had to submit a record of their CEA or CAS experience and, consistent with current guidelines, had to demonstrate an independently verified stroke or death rate of 6% or less for symptomatic patients and 3% or lower for asymptomatic patients.
The results showed the 30-day risk for death, myocardial infarction (MI), or any stroke was 3.9% with carotid stenting and 3.2% with surgery (P = .26).
But with stenting, there was a slightly higher risk for procedural nondisabling strokes (48 vs. 29; P = .03), including 15 strokes vs. 5 strokes, respectively, that left patients with no residual symptoms. This is “consistent with large, recent nationally representative registry data,” observed Dr. Halliday, of the University of Oxford (England).
For those undergoing surgery, cranial nerve palsies were reported in 5.4% vs. no patients undergoing stenting.
At 5 years, the nonprocedural fatal or disabling stroke rate was 2.5% in each group (rate ratio [RR], 0.98; P = .91), with any nonprocedural stroke occurring in 5.3% of patients with stenting vs. 4.5% with surgery (RR, 1.16; P = .33).
The investigators performed a meta-analysis combining the ACST-2 results with those of eight prior trials (four in asymptomatic and four in symptomatic patients) that yielded a similar nonsignificant result for any nonprocedural stroke (RR, 1.11; P = .21).
Based on the results from ACST-2 plus the major trials, stenting and surgery involve “similar risks and similar benefits,” Dr. Halliday concluded.
Discussant Marco Roffi, MD, University Hospital of Geneva, said, “In centers with documented expertise, carotid artery stenting should be offered as an alternative to carotid endarterectomy in patients with asymptomatic stenosis and suitable anatomy.”
While the trial provides “good news” for patients, he pointed out that a reduction in the sample size from 5,000 to 3,625 limited the statistical power and that enrollment over a long period of time may have introduced confounders, such as changes in equipment technique, and medical therapy.
Also, many centers enrolled few patients, raising the concern over low-volume centers and operators, Dr. Roffi said. “We know that 8% of the centers enrolled 39% of the patients,” and “information on the credentialing and experience of the interventionalists was limited.”
Further, a lack of systematic MI assessment may have favored the surgery group, and more recent developments in stenting with the potential of reducing periprocedural stroke were rarely used, such as proximal emboli protection in only 15% and double-layer stents in 11%.
Friedhelm Beyersdorf, MD, University Hospital of Freiburg, Germany, said that, as a vascular surgeon, he finds it understandable that there might be a higher incidence of nonfatal strokes when treating carotid stenosis with stents, given the vulnerability of these lesions.
“Nevertheless, the main conclusion from the entire study is that carotid artery treatment is extremely safe, it has to be done in order to avoid strokes, and, obviously, there seems to be an advantage for surgery in terms of nondisabling stroke,” he said.
Session chair Dr. Montalescot, however, said that what the study cannot address – and what was the subject of many online audience comments – is whether either intervention should be performed in these patients.
Unlike earlier trials comparing interventions to medical therapy, Dr. Halliday said ACST-2 enrolled patients for whom the decision had been made that revascularization was needed. In addition, 99%-100% were receiving antithrombotic therapy at baseline, 85%-90% were receiving antihypertensives, and about 85% were taking statins.
Longer-term follow-up should provide a better picture of the nonprocedural stroke risk, with patients asked annually about exactly what medications and doses they are taking, she said.
“We will have an enormous list of exactly what’s gone on and the intensity of that therapy, which is, of course, much more intense than when we carried out our first trial. But these were people in whom a procedure was thought to be necessary,” she noted.
When asked during the press conference which procedure she would choose, Dr. Halliday, a surgeon, observed that patient preference is important but that the nature of the lesion itself often determines the optimal choice.
“If you know the competence of the people doing it is equal, then the less invasive procedure – providing it has good long-term viability, and that’s why we’re following for 10 years – is the more important,” she added.
The study was funded by the UK Medical Research Council and Health Technology Assessment Programme. Dr. Halliday reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Angiography can wait for cardiac arrest without ST-elevation
A protocol of immediate angiography provided no mortality benefit over a strategy or delayed or more selective angiography among patients resuscitated from out-of-hospital cardiac arrest and without ST-segment elevation, new randomized results show.
“Among patients with resuscitated out-of-hospital cardiac arrest of possible cardiac origin, with shockable and nonshockable arrest rhythm and no ST-elevation, a strategy of immediate, unselected coronary angiography was not found to be beneficial over a delayed and selective approach with regard to the 30-day risk of all-cause death,” concluded principal investigator Steffen Desch, MD, University of Leipzig (Germany) Heart Center.
The results support previous results of the Coronary Angiography after Cardiac Arrest (COACT) trial, in patients with shockable rhythms, which also showed no differences in clinical outcomes between immediate and delayed coronary angiography at both 90 days and 1 year, he noted.
“What the clinicians wanted to know is, is it really necessary to get up at 3 a.m. in the morning to perform a coronary angiography on these patients, and that’s certainly out,” Dr. Desch said in an interview. “So, there’s really no room for this strategy anymore. You can take your time and wait a day or 2.”
These findings, from the TOMAHAWK trial, were presented Aug. 29 at the annual congress of the European Society of Cardiology and simultaneously published online in the New England Journal of Medicine.
Larger group without ST-segment elevation
Prognosis after out-of-hospital cardiac arrest is extremely poor, with an overall survival rate of less than 10%, Dr. Desch noted. “Actually, only 20% make it to the hospital; the vast majority of these patients die out in the field, so there’s really a great need in improving treatment.”
Acute coronary syndrome accounts for up to 60% of out-of-hospital arrests in which a cardiac cause has been identified, the authors wrote in their report. ST-segment elevation on postresuscitation electrocardiography “has good positive predictive value” for acute coronary lesions triggering the arrest, but in the far larger subgroup of patients without ST-segment elevation, “the spectrum of underlying causes is considerably broader and includes both cardiac and noncardiac causes.”
In patients with myocardial infarction, early revascularization would prevent negative consequences of myocardial injury, but unselected early coronary angiography would put patients not having an MI at unnecessary risk for procedural complications or delay in the diagnosis of the actual cause of their arrest, they noted.
In this trial, the researchers randomly assigned 554 patients from 31 sites in Germany and Denmark who were successfully resuscitated after cardiac arrest of possible cardiac origin to immediate transfer for coronary angiography or to initial intensive care assessment with delayed or selective angiography after a minimum delay of at least 1 day.
In the end, the average delay in this arm was 2 days, Dr. Desch noted. If the clinical course indicated that a coronary cause was unlikely, angiography might not be performed at all in this group.
No patient had ST-segment elevation on postresuscitation electrocardiography. The primary endpoint was death from any cause at 30 days; secondary end points were death from any cause or severe neurologic deficit at 30 days.
Results showed that 95% of patients in the immediate angiography group actually underwent the procedure, compared with 62% of those in the delayed group, a finding that was “logical” given the study design, he said.
At 30 days, 54% of patients in the immediate angiography group and 46% in the delayed group had died, a nonsignificant difference (P = .06). Because the researchers had performed an interim analysis, Dr. Desch explained, the final P value for significance in this trial was not .05, but rather .034, to account for multiple comparisons.
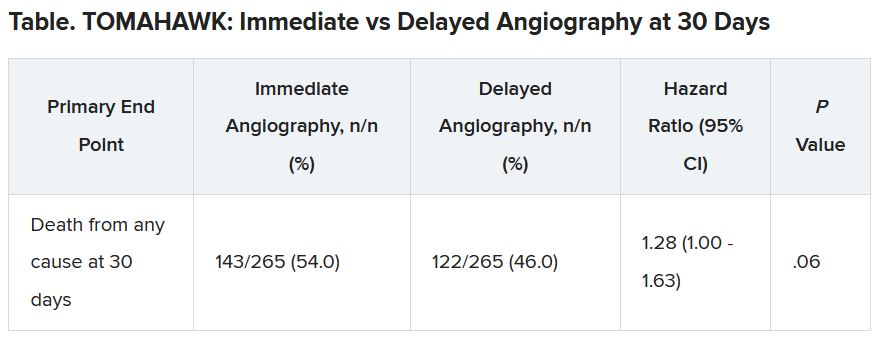
The secondary end point of death from any cause or severe neurologic deficit at 30 days “was actually nominally significant in favor of the delayed group,” he said. “So, this is not corrected for multiple testing, it’s just a hypothesis that’s in the room, but it’s certainly worthy of discussion that the immediate strategy might actually cause harm.”
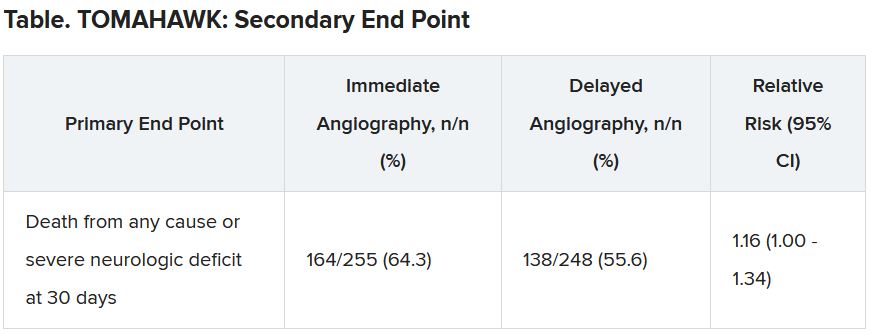
There was no difference between the groups in peak release of myocardial enzymes, or any other safety end points, including bleeding, stroke, or renal failure, Dr. Desch said.
Further analyses showed no large differences between subgroups, including age, diabetes, first monitored rhythm, confirmed MI as the trigger of the arrest, sex, and the time from cardiac arrest to the return of spontaneous circulation, he noted.
Opportunity to minimize harm
Discussant for the results during the presentation was Susanna Price, MBBS, PhD, Royal Brompton Hospital, London.
Dr. Price concluded: “What this means for me, is it gives me information that’s useful regarding the opportunity to minimize harm, which is a lot of what critical care is about, so we don’t necessarily now have to move these patients very acutely when they’ve just come in through the ED [emergency department]. It has implications for resource utilization, but also implications for mobilizing patients around the hospital during COVID-19.”
It’s also important to note that coronary angiography was still carried out in certain patients, “so we still have to have that dialogue with our interventional cardiologists for certain patients who may need to go to the cath lab, and what it should now allow us to do is give appropriate focus to how to manage these patients when they come in to the ED or to our ICUs [intensive care units],” she said.
Dr. Price added, though, that perhaps “the most important slide” in the presentation was that showing 90% of these patients had a witnessed cardiac arrest, “and yet a third of these patients, 168 of them, had no bystander CPR at all.”
She pointed to the “chain of survival” after cardiac arrest, of which Charles D. Deakin, MD, University Hospital Southampton (England), wrote that “not all links are equal.”
“Early recognition and calling for help, early CPR, early defibrillation where appropriate are very, very important, and we need to be addressing all of these, as well as what happens in the cath lab and after admission,” Dr. Price said.
This research was funded by the German Center for Cardiovascular Research. Dr. Desch and Dr. Price reported no relevant disclosures.
A version of this article first appeared on Medscape.com.
A protocol of immediate angiography provided no mortality benefit over a strategy or delayed or more selective angiography among patients resuscitated from out-of-hospital cardiac arrest and without ST-segment elevation, new randomized results show.
“Among patients with resuscitated out-of-hospital cardiac arrest of possible cardiac origin, with shockable and nonshockable arrest rhythm and no ST-elevation, a strategy of immediate, unselected coronary angiography was not found to be beneficial over a delayed and selective approach with regard to the 30-day risk of all-cause death,” concluded principal investigator Steffen Desch, MD, University of Leipzig (Germany) Heart Center.
The results support previous results of the Coronary Angiography after Cardiac Arrest (COACT) trial, in patients with shockable rhythms, which also showed no differences in clinical outcomes between immediate and delayed coronary angiography at both 90 days and 1 year, he noted.
“What the clinicians wanted to know is, is it really necessary to get up at 3 a.m. in the morning to perform a coronary angiography on these patients, and that’s certainly out,” Dr. Desch said in an interview. “So, there’s really no room for this strategy anymore. You can take your time and wait a day or 2.”
These findings, from the TOMAHAWK trial, were presented Aug. 29 at the annual congress of the European Society of Cardiology and simultaneously published online in the New England Journal of Medicine.
Larger group without ST-segment elevation
Prognosis after out-of-hospital cardiac arrest is extremely poor, with an overall survival rate of less than 10%, Dr. Desch noted. “Actually, only 20% make it to the hospital; the vast majority of these patients die out in the field, so there’s really a great need in improving treatment.”
Acute coronary syndrome accounts for up to 60% of out-of-hospital arrests in which a cardiac cause has been identified, the authors wrote in their report. ST-segment elevation on postresuscitation electrocardiography “has good positive predictive value” for acute coronary lesions triggering the arrest, but in the far larger subgroup of patients without ST-segment elevation, “the spectrum of underlying causes is considerably broader and includes both cardiac and noncardiac causes.”
In patients with myocardial infarction, early revascularization would prevent negative consequences of myocardial injury, but unselected early coronary angiography would put patients not having an MI at unnecessary risk for procedural complications or delay in the diagnosis of the actual cause of their arrest, they noted.
In this trial, the researchers randomly assigned 554 patients from 31 sites in Germany and Denmark who were successfully resuscitated after cardiac arrest of possible cardiac origin to immediate transfer for coronary angiography or to initial intensive care assessment with delayed or selective angiography after a minimum delay of at least 1 day.
In the end, the average delay in this arm was 2 days, Dr. Desch noted. If the clinical course indicated that a coronary cause was unlikely, angiography might not be performed at all in this group.
No patient had ST-segment elevation on postresuscitation electrocardiography. The primary endpoint was death from any cause at 30 days; secondary end points were death from any cause or severe neurologic deficit at 30 days.
Results showed that 95% of patients in the immediate angiography group actually underwent the procedure, compared with 62% of those in the delayed group, a finding that was “logical” given the study design, he said.
At 30 days, 54% of patients in the immediate angiography group and 46% in the delayed group had died, a nonsignificant difference (P = .06). Because the researchers had performed an interim analysis, Dr. Desch explained, the final P value for significance in this trial was not .05, but rather .034, to account for multiple comparisons.

The secondary end point of death from any cause or severe neurologic deficit at 30 days “was actually nominally significant in favor of the delayed group,” he said. “So, this is not corrected for multiple testing, it’s just a hypothesis that’s in the room, but it’s certainly worthy of discussion that the immediate strategy might actually cause harm.”

There was no difference between the groups in peak release of myocardial enzymes, or any other safety end points, including bleeding, stroke, or renal failure, Dr. Desch said.
Further analyses showed no large differences between subgroups, including age, diabetes, first monitored rhythm, confirmed MI as the trigger of the arrest, sex, and the time from cardiac arrest to the return of spontaneous circulation, he noted.
Opportunity to minimize harm
Discussant for the results during the presentation was Susanna Price, MBBS, PhD, Royal Brompton Hospital, London.
Dr. Price concluded: “What this means for me, is it gives me information that’s useful regarding the opportunity to minimize harm, which is a lot of what critical care is about, so we don’t necessarily now have to move these patients very acutely when they’ve just come in through the ED [emergency department]. It has implications for resource utilization, but also implications for mobilizing patients around the hospital during COVID-19.”
It’s also important to note that coronary angiography was still carried out in certain patients, “so we still have to have that dialogue with our interventional cardiologists for certain patients who may need to go to the cath lab, and what it should now allow us to do is give appropriate focus to how to manage these patients when they come in to the ED or to our ICUs [intensive care units],” she said.
Dr. Price added, though, that perhaps “the most important slide” in the presentation was that showing 90% of these patients had a witnessed cardiac arrest, “and yet a third of these patients, 168 of them, had no bystander CPR at all.”
She pointed to the “chain of survival” after cardiac arrest, of which Charles D. Deakin, MD, University Hospital Southampton (England), wrote that “not all links are equal.”
“Early recognition and calling for help, early CPR, early defibrillation where appropriate are very, very important, and we need to be addressing all of these, as well as what happens in the cath lab and after admission,” Dr. Price said.
This research was funded by the German Center for Cardiovascular Research. Dr. Desch and Dr. Price reported no relevant disclosures.
A version of this article first appeared on Medscape.com.
A protocol of immediate angiography provided no mortality benefit over a strategy or delayed or more selective angiography among patients resuscitated from out-of-hospital cardiac arrest and without ST-segment elevation, new randomized results show.
“Among patients with resuscitated out-of-hospital cardiac arrest of possible cardiac origin, with shockable and nonshockable arrest rhythm and no ST-elevation, a strategy of immediate, unselected coronary angiography was not found to be beneficial over a delayed and selective approach with regard to the 30-day risk of all-cause death,” concluded principal investigator Steffen Desch, MD, University of Leipzig (Germany) Heart Center.
The results support previous results of the Coronary Angiography after Cardiac Arrest (COACT) trial, in patients with shockable rhythms, which also showed no differences in clinical outcomes between immediate and delayed coronary angiography at both 90 days and 1 year, he noted.
“What the clinicians wanted to know is, is it really necessary to get up at 3 a.m. in the morning to perform a coronary angiography on these patients, and that’s certainly out,” Dr. Desch said in an interview. “So, there’s really no room for this strategy anymore. You can take your time and wait a day or 2.”
These findings, from the TOMAHAWK trial, were presented Aug. 29 at the annual congress of the European Society of Cardiology and simultaneously published online in the New England Journal of Medicine.
Larger group without ST-segment elevation
Prognosis after out-of-hospital cardiac arrest is extremely poor, with an overall survival rate of less than 10%, Dr. Desch noted. “Actually, only 20% make it to the hospital; the vast majority of these patients die out in the field, so there’s really a great need in improving treatment.”
Acute coronary syndrome accounts for up to 60% of out-of-hospital arrests in which a cardiac cause has been identified, the authors wrote in their report. ST-segment elevation on postresuscitation electrocardiography “has good positive predictive value” for acute coronary lesions triggering the arrest, but in the far larger subgroup of patients without ST-segment elevation, “the spectrum of underlying causes is considerably broader and includes both cardiac and noncardiac causes.”
In patients with myocardial infarction, early revascularization would prevent negative consequences of myocardial injury, but unselected early coronary angiography would put patients not having an MI at unnecessary risk for procedural complications or delay in the diagnosis of the actual cause of their arrest, they noted.
In this trial, the researchers randomly assigned 554 patients from 31 sites in Germany and Denmark who were successfully resuscitated after cardiac arrest of possible cardiac origin to immediate transfer for coronary angiography or to initial intensive care assessment with delayed or selective angiography after a minimum delay of at least 1 day.
In the end, the average delay in this arm was 2 days, Dr. Desch noted. If the clinical course indicated that a coronary cause was unlikely, angiography might not be performed at all in this group.
No patient had ST-segment elevation on postresuscitation electrocardiography. The primary endpoint was death from any cause at 30 days; secondary end points were death from any cause or severe neurologic deficit at 30 days.
Results showed that 95% of patients in the immediate angiography group actually underwent the procedure, compared with 62% of those in the delayed group, a finding that was “logical” given the study design, he said.
At 30 days, 54% of patients in the immediate angiography group and 46% in the delayed group had died, a nonsignificant difference (P = .06). Because the researchers had performed an interim analysis, Dr. Desch explained, the final P value for significance in this trial was not .05, but rather .034, to account for multiple comparisons.

The secondary end point of death from any cause or severe neurologic deficit at 30 days “was actually nominally significant in favor of the delayed group,” he said. “So, this is not corrected for multiple testing, it’s just a hypothesis that’s in the room, but it’s certainly worthy of discussion that the immediate strategy might actually cause harm.”

There was no difference between the groups in peak release of myocardial enzymes, or any other safety end points, including bleeding, stroke, or renal failure, Dr. Desch said.
Further analyses showed no large differences between subgroups, including age, diabetes, first monitored rhythm, confirmed MI as the trigger of the arrest, sex, and the time from cardiac arrest to the return of spontaneous circulation, he noted.
Opportunity to minimize harm
Discussant for the results during the presentation was Susanna Price, MBBS, PhD, Royal Brompton Hospital, London.
Dr. Price concluded: “What this means for me, is it gives me information that’s useful regarding the opportunity to minimize harm, which is a lot of what critical care is about, so we don’t necessarily now have to move these patients very acutely when they’ve just come in through the ED [emergency department]. It has implications for resource utilization, but also implications for mobilizing patients around the hospital during COVID-19.”
It’s also important to note that coronary angiography was still carried out in certain patients, “so we still have to have that dialogue with our interventional cardiologists for certain patients who may need to go to the cath lab, and what it should now allow us to do is give appropriate focus to how to manage these patients when they come in to the ED or to our ICUs [intensive care units],” she said.
Dr. Price added, though, that perhaps “the most important slide” in the presentation was that showing 90% of these patients had a witnessed cardiac arrest, “and yet a third of these patients, 168 of them, had no bystander CPR at all.”
She pointed to the “chain of survival” after cardiac arrest, of which Charles D. Deakin, MD, University Hospital Southampton (England), wrote that “not all links are equal.”
“Early recognition and calling for help, early CPR, early defibrillation where appropriate are very, very important, and we need to be addressing all of these, as well as what happens in the cath lab and after admission,” Dr. Price said.
This research was funded by the German Center for Cardiovascular Research. Dr. Desch and Dr. Price reported no relevant disclosures.
A version of this article first appeared on Medscape.com.
Although inconclusive, CV safety study of cancer therapy attracts attention
The first global trial to compare the cardiovascular (CV) safety of two therapies for prostate cancer proved inconclusive because of inadequate enrollment and events, but the study is a harbinger of growth in the emerging specialty of cardio-oncology, according to experts.
“Many new cancer agents have extended patient survival, yet some of these agents have significant potential cardiovascular toxicity,” said Renato D. Lopes, MD, in presenting a study at the annual congress of the European Society of Cardiology.
In the context of improving survival in patients with or at risk for both cancer and cardiovascular disease, he suggested that the prostate cancer study he led could be “a model for interdisciplinary collaboration” needed to address the relative and sometimes competing risks of these disease states.
This point was seconded by several pioneers in cardio-oncology who participated in the discussion of the results of the trial, called PRONOUNCE.
“We know many drugs in oncology increase cardiovascular risk, so these are the types of trials we need,” according Thomas M. Suter, MD, who leads the cardio-oncology service at the University Hospital, Berne, Switzerland. He was the ESC-invited discussant for PRONOUNCE.
More than 100 centers in 12 countries involved
In PRONOUNCE, 545 patients with prostate cancer and established atherosclerotic cardiovascular disease were randomized to degarelix, a gonadotropin-releasing hormone antagonist, or leuprolide, a GnRH agonist. The patients were enrolled at 113 participating centers in 12 countries. All of the patients had an indication for an androgen-deprivation therapy (ADT).
In numerous previous studies, “ADT has been associated with higher CV morbidity and mortality, particularly in men with preexisting CV disease,” explained Dr. Lopes, but the relative cardiovascular safety of GnRH agonists relative to GnRH antagonists has been “controversial.”
The PRONOUNCE study was designed to resolve this issue, but the study was terminated early because of slow enrollment (not related to the COVID-19 pandemic). The planned enrollment was 900 patients.
In addition, the rate of major adverse cardiovascular events (MACE), defined as myocardial infarction, stroke, or death, was lower over the course of follow-up than anticipated in the study design.
No significant difference on primary endpoint
At the end of 12 months, MACE occurred in 11 (4.1%) of patients randomized to leuprolide and 15 (5.5%) of those randomized to degarelix. The greater hazard ratio for MACE in the degarelix group did not approach statistical significance (hazard ratio, 1.28; P = .53).
As a result, the question of the relative CV safety of these drugs “remains unresolved,” according to Dr. Lopes, professor of medicine at Duke University Medical Center, Durham, N.C.
This does not diminish the need to answer this question. In the addition to the fact that cancer is a malignancy primarily of advancing age when CV disease is prevalent – the mean age in this study was 73 years and 44% were over age 75 – it is often an indolent disease with long periods of survival, according to Dr. Lopes. About half of prostate cancer patients have concomitant CV disease, and about half will receive ADT at some point in their treatment.
In patients receiving ADT, leuprolide is far more commonly used than GnRH antagonists, which are offered in only about 4% of patients, according to data cited by Dr. Lopes. The underlying hypothesis of this study was that leuprolide is associated with greater CV risk, which might have been relevant to a risk-benefit calculation, if the hypothesis had been confirmed.
Cancer drugs can increase CV risk
Based on experimental data, “there is concern the leuprolide is involved in plaque destabilization,” said Dr. Lopes, but he noted that ADTs in general are associated with adverse metabolic changes, including increases in LDL cholesterol, insulin resistance, and body fat, all of which could be relevant to CV risk.
It is the improving rates of survival for prostate cancer as well for other types of cancer that have increased attention to the potential for cancer drugs to increase CV risk, another major cause of early mortality. For these competing risks, objective data are needed to evaluate a relative risk-to-benefit ratio for treatment choices.
This dilemma led the ESC to recently establish its Council on Cardio-Oncology, and many centers around the world are also creating interdisciplinary groups to guide treatment choices for patients with both diseases.
“You will certainly get a lot of referrals,” said Rudolf de Boer, MD, professor of translational cardiology, University Medical Center, Groningen, Netherlands. Basing his remark on his own experience starting a cardio-oncology clinic at his institution, he called this work challenging and agreed that the need for objective data is urgent.
“We need data to provide common ground on which to judge relative risks,” Dr. de Boer said. He also praised the PRONOUNCE investigators for their efforts even if the data failed to answer the question posed.
The PRONOUNCE results were published online in Circulation at the time of Dr. Lopes’s presentation.
The study received funding from Ferring Pharmaceuticals. Dr. Lopes reports financial relationships with Bristol-Myers Squibb, GlaxoSmithKline, Medtronic, Pfizer, and Sanofi. Dr. Suter reports financial relationships with Boehringer Ingelheim, GlaxoSmithKline, and Roche. Dr. de Boer reports financial relationships with AstraZeneca, Abbott, Bristol-Myers Squibb, Novartis, Novo Nordisk, and Roche.
The first global trial to compare the cardiovascular (CV) safety of two therapies for prostate cancer proved inconclusive because of inadequate enrollment and events, but the study is a harbinger of growth in the emerging specialty of cardio-oncology, according to experts.
“Many new cancer agents have extended patient survival, yet some of these agents have significant potential cardiovascular toxicity,” said Renato D. Lopes, MD, in presenting a study at the annual congress of the European Society of Cardiology.
In the context of improving survival in patients with or at risk for both cancer and cardiovascular disease, he suggested that the prostate cancer study he led could be “a model for interdisciplinary collaboration” needed to address the relative and sometimes competing risks of these disease states.
This point was seconded by several pioneers in cardio-oncology who participated in the discussion of the results of the trial, called PRONOUNCE.
“We know many drugs in oncology increase cardiovascular risk, so these are the types of trials we need,” according Thomas M. Suter, MD, who leads the cardio-oncology service at the University Hospital, Berne, Switzerland. He was the ESC-invited discussant for PRONOUNCE.
More than 100 centers in 12 countries involved
In PRONOUNCE, 545 patients with prostate cancer and established atherosclerotic cardiovascular disease were randomized to degarelix, a gonadotropin-releasing hormone antagonist, or leuprolide, a GnRH agonist. The patients were enrolled at 113 participating centers in 12 countries. All of the patients had an indication for an androgen-deprivation therapy (ADT).
In numerous previous studies, “ADT has been associated with higher CV morbidity and mortality, particularly in men with preexisting CV disease,” explained Dr. Lopes, but the relative cardiovascular safety of GnRH agonists relative to GnRH antagonists has been “controversial.”
The PRONOUNCE study was designed to resolve this issue, but the study was terminated early because of slow enrollment (not related to the COVID-19 pandemic). The planned enrollment was 900 patients.
In addition, the rate of major adverse cardiovascular events (MACE), defined as myocardial infarction, stroke, or death, was lower over the course of follow-up than anticipated in the study design.
No significant difference on primary endpoint
At the end of 12 months, MACE occurred in 11 (4.1%) of patients randomized to leuprolide and 15 (5.5%) of those randomized to degarelix. The greater hazard ratio for MACE in the degarelix group did not approach statistical significance (hazard ratio, 1.28; P = .53).
As a result, the question of the relative CV safety of these drugs “remains unresolved,” according to Dr. Lopes, professor of medicine at Duke University Medical Center, Durham, N.C.
This does not diminish the need to answer this question. In the addition to the fact that cancer is a malignancy primarily of advancing age when CV disease is prevalent – the mean age in this study was 73 years and 44% were over age 75 – it is often an indolent disease with long periods of survival, according to Dr. Lopes. About half of prostate cancer patients have concomitant CV disease, and about half will receive ADT at some point in their treatment.
In patients receiving ADT, leuprolide is far more commonly used than GnRH antagonists, which are offered in only about 4% of patients, according to data cited by Dr. Lopes. The underlying hypothesis of this study was that leuprolide is associated with greater CV risk, which might have been relevant to a risk-benefit calculation, if the hypothesis had been confirmed.
Cancer drugs can increase CV risk
Based on experimental data, “there is concern the leuprolide is involved in plaque destabilization,” said Dr. Lopes, but he noted that ADTs in general are associated with adverse metabolic changes, including increases in LDL cholesterol, insulin resistance, and body fat, all of which could be relevant to CV risk.
It is the improving rates of survival for prostate cancer as well for other types of cancer that have increased attention to the potential for cancer drugs to increase CV risk, another major cause of early mortality. For these competing risks, objective data are needed to evaluate a relative risk-to-benefit ratio for treatment choices.
This dilemma led the ESC to recently establish its Council on Cardio-Oncology, and many centers around the world are also creating interdisciplinary groups to guide treatment choices for patients with both diseases.
“You will certainly get a lot of referrals,” said Rudolf de Boer, MD, professor of translational cardiology, University Medical Center, Groningen, Netherlands. Basing his remark on his own experience starting a cardio-oncology clinic at his institution, he called this work challenging and agreed that the need for objective data is urgent.
“We need data to provide common ground on which to judge relative risks,” Dr. de Boer said. He also praised the PRONOUNCE investigators for their efforts even if the data failed to answer the question posed.
The PRONOUNCE results were published online in Circulation at the time of Dr. Lopes’s presentation.
The study received funding from Ferring Pharmaceuticals. Dr. Lopes reports financial relationships with Bristol-Myers Squibb, GlaxoSmithKline, Medtronic, Pfizer, and Sanofi. Dr. Suter reports financial relationships with Boehringer Ingelheim, GlaxoSmithKline, and Roche. Dr. de Boer reports financial relationships with AstraZeneca, Abbott, Bristol-Myers Squibb, Novartis, Novo Nordisk, and Roche.
The first global trial to compare the cardiovascular (CV) safety of two therapies for prostate cancer proved inconclusive because of inadequate enrollment and events, but the study is a harbinger of growth in the emerging specialty of cardio-oncology, according to experts.
“Many new cancer agents have extended patient survival, yet some of these agents have significant potential cardiovascular toxicity,” said Renato D. Lopes, MD, in presenting a study at the annual congress of the European Society of Cardiology.
In the context of improving survival in patients with or at risk for both cancer and cardiovascular disease, he suggested that the prostate cancer study he led could be “a model for interdisciplinary collaboration” needed to address the relative and sometimes competing risks of these disease states.
This point was seconded by several pioneers in cardio-oncology who participated in the discussion of the results of the trial, called PRONOUNCE.
“We know many drugs in oncology increase cardiovascular risk, so these are the types of trials we need,” according Thomas M. Suter, MD, who leads the cardio-oncology service at the University Hospital, Berne, Switzerland. He was the ESC-invited discussant for PRONOUNCE.
More than 100 centers in 12 countries involved
In PRONOUNCE, 545 patients with prostate cancer and established atherosclerotic cardiovascular disease were randomized to degarelix, a gonadotropin-releasing hormone antagonist, or leuprolide, a GnRH agonist. The patients were enrolled at 113 participating centers in 12 countries. All of the patients had an indication for an androgen-deprivation therapy (ADT).
In numerous previous studies, “ADT has been associated with higher CV morbidity and mortality, particularly in men with preexisting CV disease,” explained Dr. Lopes, but the relative cardiovascular safety of GnRH agonists relative to GnRH antagonists has been “controversial.”
The PRONOUNCE study was designed to resolve this issue, but the study was terminated early because of slow enrollment (not related to the COVID-19 pandemic). The planned enrollment was 900 patients.
In addition, the rate of major adverse cardiovascular events (MACE), defined as myocardial infarction, stroke, or death, was lower over the course of follow-up than anticipated in the study design.
No significant difference on primary endpoint
At the end of 12 months, MACE occurred in 11 (4.1%) of patients randomized to leuprolide and 15 (5.5%) of those randomized to degarelix. The greater hazard ratio for MACE in the degarelix group did not approach statistical significance (hazard ratio, 1.28; P = .53).
As a result, the question of the relative CV safety of these drugs “remains unresolved,” according to Dr. Lopes, professor of medicine at Duke University Medical Center, Durham, N.C.
This does not diminish the need to answer this question. In the addition to the fact that cancer is a malignancy primarily of advancing age when CV disease is prevalent – the mean age in this study was 73 years and 44% were over age 75 – it is often an indolent disease with long periods of survival, according to Dr. Lopes. About half of prostate cancer patients have concomitant CV disease, and about half will receive ADT at some point in their treatment.
In patients receiving ADT, leuprolide is far more commonly used than GnRH antagonists, which are offered in only about 4% of patients, according to data cited by Dr. Lopes. The underlying hypothesis of this study was that leuprolide is associated with greater CV risk, which might have been relevant to a risk-benefit calculation, if the hypothesis had been confirmed.
Cancer drugs can increase CV risk
Based on experimental data, “there is concern the leuprolide is involved in plaque destabilization,” said Dr. Lopes, but he noted that ADTs in general are associated with adverse metabolic changes, including increases in LDL cholesterol, insulin resistance, and body fat, all of which could be relevant to CV risk.
It is the improving rates of survival for prostate cancer as well for other types of cancer that have increased attention to the potential for cancer drugs to increase CV risk, another major cause of early mortality. For these competing risks, objective data are needed to evaluate a relative risk-to-benefit ratio for treatment choices.
This dilemma led the ESC to recently establish its Council on Cardio-Oncology, and many centers around the world are also creating interdisciplinary groups to guide treatment choices for patients with both diseases.
“You will certainly get a lot of referrals,” said Rudolf de Boer, MD, professor of translational cardiology, University Medical Center, Groningen, Netherlands. Basing his remark on his own experience starting a cardio-oncology clinic at his institution, he called this work challenging and agreed that the need for objective data is urgent.
“We need data to provide common ground on which to judge relative risks,” Dr. de Boer said. He also praised the PRONOUNCE investigators for their efforts even if the data failed to answer the question posed.
The PRONOUNCE results were published online in Circulation at the time of Dr. Lopes’s presentation.
The study received funding from Ferring Pharmaceuticals. Dr. Lopes reports financial relationships with Bristol-Myers Squibb, GlaxoSmithKline, Medtronic, Pfizer, and Sanofi. Dr. Suter reports financial relationships with Boehringer Ingelheim, GlaxoSmithKline, and Roche. Dr. de Boer reports financial relationships with AstraZeneca, Abbott, Bristol-Myers Squibb, Novartis, Novo Nordisk, and Roche.
FROM ESC 2021
Walnuts lowered LDL cholesterol in healthy seniors: WAHA study
Benefit sustained over 2 years
Healthy elderly people who ate a walnut-supplemented diet for 2 years showed significant reductions in LDL cholesterol, according to a randomized study that used imaging to evaluate lipid changes.
“Regularly eating walnuts will lower your LDL cholesterol and improve the quality of LDL particles, rendering them less prone to enter the arterial wall and build up atherosclerosis, and this will occur without unwanted weight gain in spite of the high-fat – healthy vegetable fat, though – content of walnuts,” Emilio Ros, MD, PhD, senior author of the Walnuts and Healthy Aging (WAHA) study, said in an interview.
WAHA is a parallel-group, randomized, controlled trial that followed 636 patients over 2 years at centers in Loma Linda, Calif., and Barcelona. They were randomly assigned to either a walnut-free or walnut-supplemented diet, and every 2 months they were underwent nuclear magnetic resonance spectroscopy and recorded their compliance, toleration, medication changes, and body weight.
The researchers reported “significantly decreased” total cholesterol, LDL cholesterol and intermediate-density lipoprotein cholesterol, along with reductions in total LDL cholesterol particles and small LDL cholesterol particle number in patients on a walnut-supplemented diet, compared with controls. However, triglycerides and HDL cholesterol were unaffected.
Study results
The study reported mean reductions in the following lipid categories among what the researchers called the “walnut group”:
- Total cholesterol, –8.5 mg/dL (95% confidence interval, –11.2 to –5.4), a 4.4% mean reduction.
- LDL-C, –4.3 mg/dL (95% CI, –6.6 to –1.6), a 3.6% reduction.
- Intermediate-density lipoprotein cholesterol, –1.3 mg/dL (95% CI, –1.5 to –1.0] for a 16.8% reduction.
- Total LDL cholesterol particles, a reduction of 4.3%.
- Small LDL cholesterol particle number, a 6.1% decrease.
“WAHA is the largest and longest randomized nut trial to date, which overcomes power limitations of former trials, smaller and of shorter duration,” said Dr. Ros, of the lipid clinic, endocrinology, nutrition service at the Hospital Clinic Villarroel at the University of Barcelona. He noted that studies he has participated in have already shown that the walnut-supplemented diet had beneficial effects on blood pressure, systemic inflammation and endothelial function.
Other strengths of the study, he said, were that it recruited patients from two distinct locations and that it retained 90% of participants over 2 years (the study started out with 708 participants).
Christie Ballantyne, MD, concurred that the size and duration of the study are worth noting. “People always have questions about what they should eat,” said Dr. Ballantyne, chief of cardiology at Baylor College of Medicine and director of cardiovascular disease prevention at Methodist DeBakey Heart Center, both in Houston, said in an interview. “It’s very difficult to do nutritional studies. Most studies are small and short term, so it’s unusual to have study that’s large with 2 years of follow-up. This is a larger-than-usual study.”
Potential study limitations
Dr. Ros did acknowledge some limitations of the study. Participants weren’t blinded, the feeding setting wasn’t controlled, and participants were generally healthy and had average normal lipid profiles because they were taking statins, which may explain the modest lipid improvements in the study. “LDL cholesterol lowering by nuts is related to baseline levels, hence the reduction observed in our study was modest,” he said.
Additionally, because the study group was elderly, the results don’t apply to younger people. “Yet,” Dr. Ros added, “we know from many prior studies that nuts in general and walnuts in particular will lower blood cholesterol regardless of age.”
Dr. Ballantyne noted that the use of nuclear magnetic resonance spectroscopy to evaluate lipid levels involves a methodology that isn’t as systematic or as standardized as the typical lipid profile, and that different systems use proprietary software to interpret results. “That’s a little bit of an issue,” he said. “What exactly do the numbers mean?”
Overall, though, Dr. Ballantyne said the study is a significant addition to the literature. “The study is large, well done, and it confirms the benefits of something we’ve been telling patients is a good choice. It’s useful because there’s so much noise. It is important because there’s tremendous confusion and misinformation about what’s really healthy to eat.”
The study was supported by a grant from the California Walnut Commission (CWC), from which Dr. Ros and two coauthors received research funding through their institutions. Dr. Ros also reported receiving compensation from CWC and serves on a CWC advisory council. The other authors have no relationships to disclose. The WAHA study was supported by a grant from the CWC, from which Dr. Ros and some coinvestigators have received research funding through their institutions. Dr. Ballantyne has no relevant relationships to disclose.
Benefit sustained over 2 years
Benefit sustained over 2 years
Healthy elderly people who ate a walnut-supplemented diet for 2 years showed significant reductions in LDL cholesterol, according to a randomized study that used imaging to evaluate lipid changes.
“Regularly eating walnuts will lower your LDL cholesterol and improve the quality of LDL particles, rendering them less prone to enter the arterial wall and build up atherosclerosis, and this will occur without unwanted weight gain in spite of the high-fat – healthy vegetable fat, though – content of walnuts,” Emilio Ros, MD, PhD, senior author of the Walnuts and Healthy Aging (WAHA) study, said in an interview.
WAHA is a parallel-group, randomized, controlled trial that followed 636 patients over 2 years at centers in Loma Linda, Calif., and Barcelona. They were randomly assigned to either a walnut-free or walnut-supplemented diet, and every 2 months they were underwent nuclear magnetic resonance spectroscopy and recorded their compliance, toleration, medication changes, and body weight.
The researchers reported “significantly decreased” total cholesterol, LDL cholesterol and intermediate-density lipoprotein cholesterol, along with reductions in total LDL cholesterol particles and small LDL cholesterol particle number in patients on a walnut-supplemented diet, compared with controls. However, triglycerides and HDL cholesterol were unaffected.
Study results
The study reported mean reductions in the following lipid categories among what the researchers called the “walnut group”:
- Total cholesterol, –8.5 mg/dL (95% confidence interval, –11.2 to –5.4), a 4.4% mean reduction.
- LDL-C, –4.3 mg/dL (95% CI, –6.6 to –1.6), a 3.6% reduction.
- Intermediate-density lipoprotein cholesterol, –1.3 mg/dL (95% CI, –1.5 to –1.0] for a 16.8% reduction.
- Total LDL cholesterol particles, a reduction of 4.3%.
- Small LDL cholesterol particle number, a 6.1% decrease.
“WAHA is the largest and longest randomized nut trial to date, which overcomes power limitations of former trials, smaller and of shorter duration,” said Dr. Ros, of the lipid clinic, endocrinology, nutrition service at the Hospital Clinic Villarroel at the University of Barcelona. He noted that studies he has participated in have already shown that the walnut-supplemented diet had beneficial effects on blood pressure, systemic inflammation and endothelial function.
Other strengths of the study, he said, were that it recruited patients from two distinct locations and that it retained 90% of participants over 2 years (the study started out with 708 participants).
Christie Ballantyne, MD, concurred that the size and duration of the study are worth noting. “People always have questions about what they should eat,” said Dr. Ballantyne, chief of cardiology at Baylor College of Medicine and director of cardiovascular disease prevention at Methodist DeBakey Heart Center, both in Houston, said in an interview. “It’s very difficult to do nutritional studies. Most studies are small and short term, so it’s unusual to have study that’s large with 2 years of follow-up. This is a larger-than-usual study.”
Potential study limitations
Dr. Ros did acknowledge some limitations of the study. Participants weren’t blinded, the feeding setting wasn’t controlled, and participants were generally healthy and had average normal lipid profiles because they were taking statins, which may explain the modest lipid improvements in the study. “LDL cholesterol lowering by nuts is related to baseline levels, hence the reduction observed in our study was modest,” he said.
Additionally, because the study group was elderly, the results don’t apply to younger people. “Yet,” Dr. Ros added, “we know from many prior studies that nuts in general and walnuts in particular will lower blood cholesterol regardless of age.”
Dr. Ballantyne noted that the use of nuclear magnetic resonance spectroscopy to evaluate lipid levels involves a methodology that isn’t as systematic or as standardized as the typical lipid profile, and that different systems use proprietary software to interpret results. “That’s a little bit of an issue,” he said. “What exactly do the numbers mean?”
Overall, though, Dr. Ballantyne said the study is a significant addition to the literature. “The study is large, well done, and it confirms the benefits of something we’ve been telling patients is a good choice. It’s useful because there’s so much noise. It is important because there’s tremendous confusion and misinformation about what’s really healthy to eat.”
The study was supported by a grant from the California Walnut Commission (CWC), from which Dr. Ros and two coauthors received research funding through their institutions. Dr. Ros also reported receiving compensation from CWC and serves on a CWC advisory council. The other authors have no relationships to disclose. The WAHA study was supported by a grant from the CWC, from which Dr. Ros and some coinvestigators have received research funding through their institutions. Dr. Ballantyne has no relevant relationships to disclose.
Healthy elderly people who ate a walnut-supplemented diet for 2 years showed significant reductions in LDL cholesterol, according to a randomized study that used imaging to evaluate lipid changes.
“Regularly eating walnuts will lower your LDL cholesterol and improve the quality of LDL particles, rendering them less prone to enter the arterial wall and build up atherosclerosis, and this will occur without unwanted weight gain in spite of the high-fat – healthy vegetable fat, though – content of walnuts,” Emilio Ros, MD, PhD, senior author of the Walnuts and Healthy Aging (WAHA) study, said in an interview.
WAHA is a parallel-group, randomized, controlled trial that followed 636 patients over 2 years at centers in Loma Linda, Calif., and Barcelona. They were randomly assigned to either a walnut-free or walnut-supplemented diet, and every 2 months they were underwent nuclear magnetic resonance spectroscopy and recorded their compliance, toleration, medication changes, and body weight.
The researchers reported “significantly decreased” total cholesterol, LDL cholesterol and intermediate-density lipoprotein cholesterol, along with reductions in total LDL cholesterol particles and small LDL cholesterol particle number in patients on a walnut-supplemented diet, compared with controls. However, triglycerides and HDL cholesterol were unaffected.
Study results
The study reported mean reductions in the following lipid categories among what the researchers called the “walnut group”:
- Total cholesterol, –8.5 mg/dL (95% confidence interval, –11.2 to –5.4), a 4.4% mean reduction.
- LDL-C, –4.3 mg/dL (95% CI, –6.6 to –1.6), a 3.6% reduction.
- Intermediate-density lipoprotein cholesterol, –1.3 mg/dL (95% CI, –1.5 to –1.0] for a 16.8% reduction.
- Total LDL cholesterol particles, a reduction of 4.3%.
- Small LDL cholesterol particle number, a 6.1% decrease.
“WAHA is the largest and longest randomized nut trial to date, which overcomes power limitations of former trials, smaller and of shorter duration,” said Dr. Ros, of the lipid clinic, endocrinology, nutrition service at the Hospital Clinic Villarroel at the University of Barcelona. He noted that studies he has participated in have already shown that the walnut-supplemented diet had beneficial effects on blood pressure, systemic inflammation and endothelial function.
Other strengths of the study, he said, were that it recruited patients from two distinct locations and that it retained 90% of participants over 2 years (the study started out with 708 participants).
Christie Ballantyne, MD, concurred that the size and duration of the study are worth noting. “People always have questions about what they should eat,” said Dr. Ballantyne, chief of cardiology at Baylor College of Medicine and director of cardiovascular disease prevention at Methodist DeBakey Heart Center, both in Houston, said in an interview. “It’s very difficult to do nutritional studies. Most studies are small and short term, so it’s unusual to have study that’s large with 2 years of follow-up. This is a larger-than-usual study.”
Potential study limitations
Dr. Ros did acknowledge some limitations of the study. Participants weren’t blinded, the feeding setting wasn’t controlled, and participants were generally healthy and had average normal lipid profiles because they were taking statins, which may explain the modest lipid improvements in the study. “LDL cholesterol lowering by nuts is related to baseline levels, hence the reduction observed in our study was modest,” he said.
Additionally, because the study group was elderly, the results don’t apply to younger people. “Yet,” Dr. Ros added, “we know from many prior studies that nuts in general and walnuts in particular will lower blood cholesterol regardless of age.”
Dr. Ballantyne noted that the use of nuclear magnetic resonance spectroscopy to evaluate lipid levels involves a methodology that isn’t as systematic or as standardized as the typical lipid profile, and that different systems use proprietary software to interpret results. “That’s a little bit of an issue,” he said. “What exactly do the numbers mean?”
Overall, though, Dr. Ballantyne said the study is a significant addition to the literature. “The study is large, well done, and it confirms the benefits of something we’ve been telling patients is a good choice. It’s useful because there’s so much noise. It is important because there’s tremendous confusion and misinformation about what’s really healthy to eat.”
The study was supported by a grant from the California Walnut Commission (CWC), from which Dr. Ros and two coauthors received research funding through their institutions. Dr. Ros also reported receiving compensation from CWC and serves on a CWC advisory council. The other authors have no relationships to disclose. The WAHA study was supported by a grant from the CWC, from which Dr. Ros and some coinvestigators have received research funding through their institutions. Dr. Ballantyne has no relevant relationships to disclose.
FROM CIRCULATION
LOOP trial undercuts value of long-term continuous ECG screening for AFib
Perhaps short, asymptomatic bouts of atrial fibrillation (AFib) that show up on long-term, continuous monitoring aren’t worth hunting for just so oral anticoagulation (OAC) can be started, even in elderly people with other stroke risk factors.
That’s a potential message from a randomized trial that tested an AFib screening strategy relying on an implantable loop recorder (ILR) in older adults without AFib but with other stroke risk factors who were invited to participate. OAC was recommended to any participant found with even a short bout of the arrhythmia (that is, any lasting 6 minutes or longer).
More than three times as many in the monitoring group compared to a standard-care cohort were found to have AFib, and nearly all were put on OAC. In fact, monitored participants were almost three times as likely to be put on OAC (P < .0001) compared with controls.
But it didn’t make any apparent difference to outcomes. The risk for stroke or systemic embolism did not significantly differ between the two groups over more than 5 years in the trial of about 6,000 participants, called LOOP.
“This result was seen despite a high proportion of atrial fibrillation detection, and a high acceptance of anticoagulation therapy, and might imply that not all atrial fibrillation is worth screening for, and not all screen-detected atrial fibrillation merits anticoagulation,” contend the authors of the LOOP report, simultaneously published in The Lancet and presented Aug. 29 at the virtual European Society of Cardiology (ESC) Congress 2021.
“The rates of bleeding were modest, despite the low threshold for anticoagulation,” and was not significantly different between the two groups, Jesper H. Svendsen, MD, DMSc, Copenhagen University Hospital, Denmark, said at a media briefing before his presentation of the trial at the congress. He is lead author on the Lancet report.
At least 6 minutes of AFib was identified in more than 30% of the ILR-monitored patients, and about 90% of those were started on OAC, Dr. Svendsen observed.
But one take-home message from LOOP, he said in an interview, is that “short-lasting episodes” of AFib do not necessarily pose an untoward risk for stroke compared with AFib revealed by intermittent monitoring, which “primarily identifies longer-lasting atrial fibrillation episodes. So short-lasting episodes are probably not as serious as long-lasting.”
The LOOP trial “teaches us that perhaps short-lasting asymptomatic episodes may not benefit from being screened or found,” said Stefan James, MD, PhD, Uppsala University, Sweden. However, that may not be the case when the monitored individual is symptomatic or has longer-lasting AFib episodes, he said in an interview. “But certainly, this study teaches us that we need to understand much better the relationship between short episodes versus symptoms versus medical outcomes.”
In LOOP, 6,004 people aged 70-90 years without AFib but with at least one other stroke risk factor, which could include hypertension, diabetes, a history of stroke, or heart failure, were implanted with an ILR, the Reveal LINQ (Medtronic).
They were randomly assigned at four centers in Denmark to a monitoring group or a usual care group in a 1:3 ratio. Overwhelmingly, most had hypertension. Almost half the population were women.
OAC was recommended for all persons in the monitoring group who showed an episode of AFib lasting at least 6 minutes.
Atrial fibrillation was diagnosed in 31.8% of the 1,501 participants in the monitored group and 12.2% of the 4,503 assigned to usual care, for a hazard ratio (HR) of 3.17 (95% confidence interval, 2.81-3.59; P < .0001).
OAC was started in 29.7% of monitored participants and 13.1% of the control cohort, for an HR of 2.72 (95% CI, 2.41-3.08; P < .0001).
There were 315 strokes and three systemic arterial embolisms observed in the entire trial, for primary endpoint rates of 4.5% in the ILR monitoring group and 5.6% in the control group (HR, 0.80; 95% CI, 0.61-1.05; P = .11). Adding transient ischemic attack (TIA) or cardiovascular death to the endpoint did not make for a significant difference. The rates of major bleeding were 4.3% and 3.5%, respectively (P = .11).
“In general, the findings were consistent across subgroups,” including by age, sex, diabetes and heart failure status, stroke history, antiplatelet therapy, renal function, and even CHA2DS2–VASc score, Dr. Svendsen noted.
But, he said, participants in the highest tertile for baseline systolic blood pressure (BP), at least 157 mm Hg, “seemed to benefit from being screened,” with a 49% reduction in risk for the primary endpoint (P = .0066). The interaction between systolic BP and outcome was significant (P = .007).
Only 9.3% of participants in LOOP did not have a baseline diagnosis of hypertension and so had to have another risk factor to enroll, the published report notes. However, the significant interaction with systolic BP “suggests that patients with dysregulated hypertension could benefit from this type of screening and concomitant anticoagulation.”
“There is a tight association between our primary endpoint and hypertension,” Dr. Svendsen said in an interview. “But I think it’s very important to say that this subgroup analysis is only hypothesis-generating.”
An editorial accompanying the LOOP publication suggests, in line with Dr. Svendsen’s proposal, that “shorter atrial fibrillation episodes found by long-term ILRs might not have the same stroke risk as atrial fibrillation detected through single-timepoint or less intense monitoring.”
If much of the paroxysmal AFib observed in LOOP and other studies with similar monitoring methods “is not the actual cause of stroke and is instead predominantly a risk marker, further research is warranted to establish whether a different screening focus and treatment paradigm are required to prevent stroke and other vascular brain injury related to atrial fibrillation,” wrote editorialists Ben Freedman, MBBS, PhD, and Nicole Lowres, BPhty, PhD, University of Sydney, Australia.
LOOP was partially supported by Medtronic. Dr. Svendsen is a member of Medtronic advisory boards and has received speaker honoraria and research grants from Medtronic in relation to this work and outside the submitted work. Disclosures for the other authors are in the report. Dr. Freedman reports grants to the Heart Research Institute, speakers fees and nonfinancial support from the Bristol-Myers Squibb–Pfizer Alliance, speakers fees and nonfinancial support from Daiichi Sankyo, nonfinancial support from AliveCor, and speakers fees and nonfinancial support from Omron unrelated to the topic of the editorial but related to atrial fibrillation and screening for atrial fibrillation. Dr. Lowres reports grants to the Heart Research Institute from the Bristol-Myers Squibb–Pfizer Alliance unrelated to the topic of the editorial but related to atrial fibrillation and screening for atrial fibrillation.
A version of this article first appeared on Medscape.com.
Perhaps short, asymptomatic bouts of atrial fibrillation (AFib) that show up on long-term, continuous monitoring aren’t worth hunting for just so oral anticoagulation (OAC) can be started, even in elderly people with other stroke risk factors.
That’s a potential message from a randomized trial that tested an AFib screening strategy relying on an implantable loop recorder (ILR) in older adults without AFib but with other stroke risk factors who were invited to participate. OAC was recommended to any participant found with even a short bout of the arrhythmia (that is, any lasting 6 minutes or longer).
More than three times as many in the monitoring group compared to a standard-care cohort were found to have AFib, and nearly all were put on OAC. In fact, monitored participants were almost three times as likely to be put on OAC (P < .0001) compared with controls.
But it didn’t make any apparent difference to outcomes. The risk for stroke or systemic embolism did not significantly differ between the two groups over more than 5 years in the trial of about 6,000 participants, called LOOP.
“This result was seen despite a high proportion of atrial fibrillation detection, and a high acceptance of anticoagulation therapy, and might imply that not all atrial fibrillation is worth screening for, and not all screen-detected atrial fibrillation merits anticoagulation,” contend the authors of the LOOP report, simultaneously published in The Lancet and presented Aug. 29 at the virtual European Society of Cardiology (ESC) Congress 2021.
“The rates of bleeding were modest, despite the low threshold for anticoagulation,” and was not significantly different between the two groups, Jesper H. Svendsen, MD, DMSc, Copenhagen University Hospital, Denmark, said at a media briefing before his presentation of the trial at the congress. He is lead author on the Lancet report.
At least 6 minutes of AFib was identified in more than 30% of the ILR-monitored patients, and about 90% of those were started on OAC, Dr. Svendsen observed.
But one take-home message from LOOP, he said in an interview, is that “short-lasting episodes” of AFib do not necessarily pose an untoward risk for stroke compared with AFib revealed by intermittent monitoring, which “primarily identifies longer-lasting atrial fibrillation episodes. So short-lasting episodes are probably not as serious as long-lasting.”
The LOOP trial “teaches us that perhaps short-lasting asymptomatic episodes may not benefit from being screened or found,” said Stefan James, MD, PhD, Uppsala University, Sweden. However, that may not be the case when the monitored individual is symptomatic or has longer-lasting AFib episodes, he said in an interview. “But certainly, this study teaches us that we need to understand much better the relationship between short episodes versus symptoms versus medical outcomes.”
In LOOP, 6,004 people aged 70-90 years without AFib but with at least one other stroke risk factor, which could include hypertension, diabetes, a history of stroke, or heart failure, were implanted with an ILR, the Reveal LINQ (Medtronic).
They were randomly assigned at four centers in Denmark to a monitoring group or a usual care group in a 1:3 ratio. Overwhelmingly, most had hypertension. Almost half the population were women.
OAC was recommended for all persons in the monitoring group who showed an episode of AFib lasting at least 6 minutes.
Atrial fibrillation was diagnosed in 31.8% of the 1,501 participants in the monitored group and 12.2% of the 4,503 assigned to usual care, for a hazard ratio (HR) of 3.17 (95% confidence interval, 2.81-3.59; P < .0001).
OAC was started in 29.7% of monitored participants and 13.1% of the control cohort, for an HR of 2.72 (95% CI, 2.41-3.08; P < .0001).
There were 315 strokes and three systemic arterial embolisms observed in the entire trial, for primary endpoint rates of 4.5% in the ILR monitoring group and 5.6% in the control group (HR, 0.80; 95% CI, 0.61-1.05; P = .11). Adding transient ischemic attack (TIA) or cardiovascular death to the endpoint did not make for a significant difference. The rates of major bleeding were 4.3% and 3.5%, respectively (P = .11).
“In general, the findings were consistent across subgroups,” including by age, sex, diabetes and heart failure status, stroke history, antiplatelet therapy, renal function, and even CHA2DS2–VASc score, Dr. Svendsen noted.
But, he said, participants in the highest tertile for baseline systolic blood pressure (BP), at least 157 mm Hg, “seemed to benefit from being screened,” with a 49% reduction in risk for the primary endpoint (P = .0066). The interaction between systolic BP and outcome was significant (P = .007).
Only 9.3% of participants in LOOP did not have a baseline diagnosis of hypertension and so had to have another risk factor to enroll, the published report notes. However, the significant interaction with systolic BP “suggests that patients with dysregulated hypertension could benefit from this type of screening and concomitant anticoagulation.”
“There is a tight association between our primary endpoint and hypertension,” Dr. Svendsen said in an interview. “But I think it’s very important to say that this subgroup analysis is only hypothesis-generating.”
An editorial accompanying the LOOP publication suggests, in line with Dr. Svendsen’s proposal, that “shorter atrial fibrillation episodes found by long-term ILRs might not have the same stroke risk as atrial fibrillation detected through single-timepoint or less intense monitoring.”
If much of the paroxysmal AFib observed in LOOP and other studies with similar monitoring methods “is not the actual cause of stroke and is instead predominantly a risk marker, further research is warranted to establish whether a different screening focus and treatment paradigm are required to prevent stroke and other vascular brain injury related to atrial fibrillation,” wrote editorialists Ben Freedman, MBBS, PhD, and Nicole Lowres, BPhty, PhD, University of Sydney, Australia.
LOOP was partially supported by Medtronic. Dr. Svendsen is a member of Medtronic advisory boards and has received speaker honoraria and research grants from Medtronic in relation to this work and outside the submitted work. Disclosures for the other authors are in the report. Dr. Freedman reports grants to the Heart Research Institute, speakers fees and nonfinancial support from the Bristol-Myers Squibb–Pfizer Alliance, speakers fees and nonfinancial support from Daiichi Sankyo, nonfinancial support from AliveCor, and speakers fees and nonfinancial support from Omron unrelated to the topic of the editorial but related to atrial fibrillation and screening for atrial fibrillation. Dr. Lowres reports grants to the Heart Research Institute from the Bristol-Myers Squibb–Pfizer Alliance unrelated to the topic of the editorial but related to atrial fibrillation and screening for atrial fibrillation.
A version of this article first appeared on Medscape.com.
Perhaps short, asymptomatic bouts of atrial fibrillation (AFib) that show up on long-term, continuous monitoring aren’t worth hunting for just so oral anticoagulation (OAC) can be started, even in elderly people with other stroke risk factors.
That’s a potential message from a randomized trial that tested an AFib screening strategy relying on an implantable loop recorder (ILR) in older adults without AFib but with other stroke risk factors who were invited to participate. OAC was recommended to any participant found with even a short bout of the arrhythmia (that is, any lasting 6 minutes or longer).
More than three times as many in the monitoring group compared to a standard-care cohort were found to have AFib, and nearly all were put on OAC. In fact, monitored participants were almost three times as likely to be put on OAC (P < .0001) compared with controls.
But it didn’t make any apparent difference to outcomes. The risk for stroke or systemic embolism did not significantly differ between the two groups over more than 5 years in the trial of about 6,000 participants, called LOOP.
“This result was seen despite a high proportion of atrial fibrillation detection, and a high acceptance of anticoagulation therapy, and might imply that not all atrial fibrillation is worth screening for, and not all screen-detected atrial fibrillation merits anticoagulation,” contend the authors of the LOOP report, simultaneously published in The Lancet and presented Aug. 29 at the virtual European Society of Cardiology (ESC) Congress 2021.
“The rates of bleeding were modest, despite the low threshold for anticoagulation,” and was not significantly different between the two groups, Jesper H. Svendsen, MD, DMSc, Copenhagen University Hospital, Denmark, said at a media briefing before his presentation of the trial at the congress. He is lead author on the Lancet report.
At least 6 minutes of AFib was identified in more than 30% of the ILR-monitored patients, and about 90% of those were started on OAC, Dr. Svendsen observed.
But one take-home message from LOOP, he said in an interview, is that “short-lasting episodes” of AFib do not necessarily pose an untoward risk for stroke compared with AFib revealed by intermittent monitoring, which “primarily identifies longer-lasting atrial fibrillation episodes. So short-lasting episodes are probably not as serious as long-lasting.”
The LOOP trial “teaches us that perhaps short-lasting asymptomatic episodes may not benefit from being screened or found,” said Stefan James, MD, PhD, Uppsala University, Sweden. However, that may not be the case when the monitored individual is symptomatic or has longer-lasting AFib episodes, he said in an interview. “But certainly, this study teaches us that we need to understand much better the relationship between short episodes versus symptoms versus medical outcomes.”
In LOOP, 6,004 people aged 70-90 years without AFib but with at least one other stroke risk factor, which could include hypertension, diabetes, a history of stroke, or heart failure, were implanted with an ILR, the Reveal LINQ (Medtronic).
They were randomly assigned at four centers in Denmark to a monitoring group or a usual care group in a 1:3 ratio. Overwhelmingly, most had hypertension. Almost half the population were women.
OAC was recommended for all persons in the monitoring group who showed an episode of AFib lasting at least 6 minutes.
Atrial fibrillation was diagnosed in 31.8% of the 1,501 participants in the monitored group and 12.2% of the 4,503 assigned to usual care, for a hazard ratio (HR) of 3.17 (95% confidence interval, 2.81-3.59; P < .0001).
OAC was started in 29.7% of monitored participants and 13.1% of the control cohort, for an HR of 2.72 (95% CI, 2.41-3.08; P < .0001).
There were 315 strokes and three systemic arterial embolisms observed in the entire trial, for primary endpoint rates of 4.5% in the ILR monitoring group and 5.6% in the control group (HR, 0.80; 95% CI, 0.61-1.05; P = .11). Adding transient ischemic attack (TIA) or cardiovascular death to the endpoint did not make for a significant difference. The rates of major bleeding were 4.3% and 3.5%, respectively (P = .11).
“In general, the findings were consistent across subgroups,” including by age, sex, diabetes and heart failure status, stroke history, antiplatelet therapy, renal function, and even CHA2DS2–VASc score, Dr. Svendsen noted.
But, he said, participants in the highest tertile for baseline systolic blood pressure (BP), at least 157 mm Hg, “seemed to benefit from being screened,” with a 49% reduction in risk for the primary endpoint (P = .0066). The interaction between systolic BP and outcome was significant (P = .007).
Only 9.3% of participants in LOOP did not have a baseline diagnosis of hypertension and so had to have another risk factor to enroll, the published report notes. However, the significant interaction with systolic BP “suggests that patients with dysregulated hypertension could benefit from this type of screening and concomitant anticoagulation.”
“There is a tight association between our primary endpoint and hypertension,” Dr. Svendsen said in an interview. “But I think it’s very important to say that this subgroup analysis is only hypothesis-generating.”
An editorial accompanying the LOOP publication suggests, in line with Dr. Svendsen’s proposal, that “shorter atrial fibrillation episodes found by long-term ILRs might not have the same stroke risk as atrial fibrillation detected through single-timepoint or less intense monitoring.”
If much of the paroxysmal AFib observed in LOOP and other studies with similar monitoring methods “is not the actual cause of stroke and is instead predominantly a risk marker, further research is warranted to establish whether a different screening focus and treatment paradigm are required to prevent stroke and other vascular brain injury related to atrial fibrillation,” wrote editorialists Ben Freedman, MBBS, PhD, and Nicole Lowres, BPhty, PhD, University of Sydney, Australia.
LOOP was partially supported by Medtronic. Dr. Svendsen is a member of Medtronic advisory boards and has received speaker honoraria and research grants from Medtronic in relation to this work and outside the submitted work. Disclosures for the other authors are in the report. Dr. Freedman reports grants to the Heart Research Institute, speakers fees and nonfinancial support from the Bristol-Myers Squibb–Pfizer Alliance, speakers fees and nonfinancial support from Daiichi Sankyo, nonfinancial support from AliveCor, and speakers fees and nonfinancial support from Omron unrelated to the topic of the editorial but related to atrial fibrillation and screening for atrial fibrillation. Dr. Lowres reports grants to the Heart Research Institute from the Bristol-Myers Squibb–Pfizer Alliance unrelated to the topic of the editorial but related to atrial fibrillation and screening for atrial fibrillation.
A version of this article first appeared on Medscape.com.
SSaSS: Salt substitute shows clear reduction in stroke, CV events, death
Switching from regular salt to a low-sodium salt substitute has major public health benefits, including a reduction in stroke, cardiovascular events, and death, a new landmark study shows.
The Salt Substitute and Stroke Study (SSaSS) was conducted in 21,000 people with a history of stroke or high blood pressure in rural China, with half of them using a lower-sodium salt substitute instead of regular salt.
Results showed that after 5 years, those using the salt substitute had a 14% reduction in stroke, a 13% reduction in major cardiovascular events, and a 12% reduction in death. These benefits were achieved without any apparent adverse effects.
The trial was presented by Bruce Neal, MB, George Institute for Global Health, Sydney, Australia, on Aug. 29 at the virtual European Society of Cardiology (ESC) Congress 2021. They were simultaneously published online in the New England Journal of Medicine.
“This is one of the largest dietary intervention trials ever conducted and has shown very clear evidence of protection against stroke, cardiovascular events, and premature death, with no adverse effects with a very simple and low-cost intervention,” Dr. Neal concluded. “This is a very easy thing to work into the diet. You just replace regular salt with a substitute that looks and tastes almost identical,” he added.
Addressing the issue of whether these results are generalizable to other populations, Dr. Neal said, “We believe the results are relevant to everyone who eats salt.
“The way the body manages sodium and potassium and their association with blood pressure is highly consistent across different populations,” he said. “Almost everyone, with the exception of a few people with serious kidney disease, should be avoiding salt or switching to a salt substitute and expect to see some benefit of this.”
Commentators at the ESC presentation lauded the study as “magnificent,” with “extraordinary” results and “very powerful implications.”
Designated discussant, hypertension expert Bryan Williams, MD, University College London, said the SSaSS was “probably the most important study with regards to public health that we will see.” He described the reductions in stroke, cardiovascular events, and death as “extraordinary for such a simple intervention.”
Dr. Williams added: “Those who have doubted the benefits of salt restriction must now admit they were wrong. The debate stops here. The data are in. Global health interventions to implement these findings must now begin.”
He also highlighted the large number of events in the trial. “This was a large, pragmatic, long-duration study in a high-risk population, and with 5,000 cardiovascular events it gives enormous power to show benefits.”
Chair of the ESC session, Barbara Casadei, MD, DPhil, John Radcliffe Hospital, Oxford (England), said the SSaSS “will change the way we think about salt and be remembered for years to come.”
Noting that the benefits were seen in all subgroups across the study, Bertram Pitt, MD, University of Michigan, Ann Arbor, was particularly excited about the stroke reduction seen in patients with diabetes, noting that several recent trials of new diabetes drugs have not managed to show a reduction in stroke.
“For patients with diabetes, this is a really important intervention,” he stated.
However, an editorial accompanying the NEJM publication gave a somewhat less enthusiastic response to the study than the ESC commentators.
Julie R. Ingelfinger, MD, deputy editor of the journal, points out that serial monitoring of potassium levels was not performed in the trial, so it is possible that hyperkalemic episodes were not detected, and persons with a history of medical conditions that may be associated with hyperkalemia were not studied.
She also noted that because the salt substitute was distributed to families, it would have been instructive to have data on the household members without risk factors, but no such data were obtained.
“Overall, the SSaSS provides some intriguing hints, but wider effectiveness is hard to predict, given limited generalizability,” she concluded.
Cluster-randomized trial
The SSaSS was an open-label, cluster-randomized trial involving 20,995 people from 600 villages in rural China who had a history of stroke or were 60 years of age or older and had uncontrolled hypertension. Patients with a history of severe kidney disease and those taking potassium supplements or potassium-sparing diuretics were excluded.
They were randomly assigned in a 1:1 ratio to the intervention group, in which the participants used a salt substitute (roughly 75% sodium chloride and 25% potassium chloride), or to the control group, in which the participants continued to use regular salt (100% sodium chloride).
Results showed that after a mean follow-up of 4.74 years, systolic blood pressure was reduced by 3.3 mm Hg in the salt substitute group.
The rate of stroke, the primary endpoint, was 29.14 events per 1,000 person-years in the salt substitute group vs. 33.65 events per 1,000 person-years with regular salt (rate ratio, 0.86; 95% confidence interval, 0.77-0.96; P = .006).
The rates of major cardiovascular events were 49.09 events per 1,000 person-years in the salt substitute group vs. 56.29 events per 1,000 person-years in those using regular salt (rate ratio, 0.87; 95% CI, 0.80-0.94; P < .001).
And the rate of death was 39.28 events per 1,000 person-years with the salt substitute vs. 44.61 events per 1,000 person-years with regular salt (rate ratio, 0.88; 95% CI, 0.82-0.95; P < .001).
The rate of serious adverse events attributed to hyperkalemia was not significantly higher with the salt substitute than with regular salt (3.35 events vs. 3.30 events per 1,000 person-years; rate ratio, 1.04; 95% CI, 0.80-1.37; P = .76).
Dr. Neal reported that 7%-8% of the control group started using salt substitute over the study period, so these results have likely underestimated the true effect of switching to a salt substitute product.
Noting that about 10 million cardiovascular events occur each year in China, he said the study results suggested that using salt substitute instead of regular salt could prevent about 10% of these events.
Food manufacturers must make changes
Dr. Neal acknowledged that a limitation of the study was the fact it was conducted in a single country, which would raise issues of generalizability. But he said he believes the results are generalizable to other populations.
Those who would get the most benefit from switching to a salt substitute are those who consume large amounts of discretionary salt – salt added at home at the time of cooking for preservation of food or seasoning. “This is salt that is easy to replace with salt substitute,” Dr. Neal noted.
“There are more than 5 billion people in the world that consume more than 50% of their salt intake as discretionary salt – mainly in the developing world. These people would expect to get significant health benefits from a switch to salt substitute.”
He pointed out that salt substitute is low cost and is easy to manufacture. “Salt substitutes cost around 50% more than regular salt, but this translates into just a dollar or two per person per year to make the switch.”
Dr. Neal said the results also apply to higher-income countries but must be implemented by governments and food manufactures, as most salt in these countries comes from processed foods.
“This study provides strong evidence to take to the food industry,” he concluded. “We would like to see food manufacturers switch to using salt substitute and for salt substitute products to be widely available on supermarket shelves. We also urge governments to take action to promote use of salt substitutes over regular salt. This could take the form of taxing regular salt or subsidies for use of salt substitutes.”
The SSaSS was supported by grants from the National Health and Medical Research Council of Australia. Dr. Neal reports no disclosures. Dr. Ingelfinger is employed by the New England Journal of Medicine as deputy editor.
A version of this article first appeared on Medscape.com.
Switching from regular salt to a low-sodium salt substitute has major public health benefits, including a reduction in stroke, cardiovascular events, and death, a new landmark study shows.
The Salt Substitute and Stroke Study (SSaSS) was conducted in 21,000 people with a history of stroke or high blood pressure in rural China, with half of them using a lower-sodium salt substitute instead of regular salt.
Results showed that after 5 years, those using the salt substitute had a 14% reduction in stroke, a 13% reduction in major cardiovascular events, and a 12% reduction in death. These benefits were achieved without any apparent adverse effects.
The trial was presented by Bruce Neal, MB, George Institute for Global Health, Sydney, Australia, on Aug. 29 at the virtual European Society of Cardiology (ESC) Congress 2021. They were simultaneously published online in the New England Journal of Medicine.
“This is one of the largest dietary intervention trials ever conducted and has shown very clear evidence of protection against stroke, cardiovascular events, and premature death, with no adverse effects with a very simple and low-cost intervention,” Dr. Neal concluded. “This is a very easy thing to work into the diet. You just replace regular salt with a substitute that looks and tastes almost identical,” he added.
Addressing the issue of whether these results are generalizable to other populations, Dr. Neal said, “We believe the results are relevant to everyone who eats salt.
“The way the body manages sodium and potassium and their association with blood pressure is highly consistent across different populations,” he said. “Almost everyone, with the exception of a few people with serious kidney disease, should be avoiding salt or switching to a salt substitute and expect to see some benefit of this.”
Commentators at the ESC presentation lauded the study as “magnificent,” with “extraordinary” results and “very powerful implications.”
Designated discussant, hypertension expert Bryan Williams, MD, University College London, said the SSaSS was “probably the most important study with regards to public health that we will see.” He described the reductions in stroke, cardiovascular events, and death as “extraordinary for such a simple intervention.”
Dr. Williams added: “Those who have doubted the benefits of salt restriction must now admit they were wrong. The debate stops here. The data are in. Global health interventions to implement these findings must now begin.”
He also highlighted the large number of events in the trial. “This was a large, pragmatic, long-duration study in a high-risk population, and with 5,000 cardiovascular events it gives enormous power to show benefits.”
Chair of the ESC session, Barbara Casadei, MD, DPhil, John Radcliffe Hospital, Oxford (England), said the SSaSS “will change the way we think about salt and be remembered for years to come.”
Noting that the benefits were seen in all subgroups across the study, Bertram Pitt, MD, University of Michigan, Ann Arbor, was particularly excited about the stroke reduction seen in patients with diabetes, noting that several recent trials of new diabetes drugs have not managed to show a reduction in stroke.
“For patients with diabetes, this is a really important intervention,” he stated.
However, an editorial accompanying the NEJM publication gave a somewhat less enthusiastic response to the study than the ESC commentators.
Julie R. Ingelfinger, MD, deputy editor of the journal, points out that serial monitoring of potassium levels was not performed in the trial, so it is possible that hyperkalemic episodes were not detected, and persons with a history of medical conditions that may be associated with hyperkalemia were not studied.
She also noted that because the salt substitute was distributed to families, it would have been instructive to have data on the household members without risk factors, but no such data were obtained.
“Overall, the SSaSS provides some intriguing hints, but wider effectiveness is hard to predict, given limited generalizability,” she concluded.
Cluster-randomized trial
The SSaSS was an open-label, cluster-randomized trial involving 20,995 people from 600 villages in rural China who had a history of stroke or were 60 years of age or older and had uncontrolled hypertension. Patients with a history of severe kidney disease and those taking potassium supplements or potassium-sparing diuretics were excluded.
They were randomly assigned in a 1:1 ratio to the intervention group, in which the participants used a salt substitute (roughly 75% sodium chloride and 25% potassium chloride), or to the control group, in which the participants continued to use regular salt (100% sodium chloride).
Results showed that after a mean follow-up of 4.74 years, systolic blood pressure was reduced by 3.3 mm Hg in the salt substitute group.
The rate of stroke, the primary endpoint, was 29.14 events per 1,000 person-years in the salt substitute group vs. 33.65 events per 1,000 person-years with regular salt (rate ratio, 0.86; 95% confidence interval, 0.77-0.96; P = .006).
The rates of major cardiovascular events were 49.09 events per 1,000 person-years in the salt substitute group vs. 56.29 events per 1,000 person-years in those using regular salt (rate ratio, 0.87; 95% CI, 0.80-0.94; P < .001).
And the rate of death was 39.28 events per 1,000 person-years with the salt substitute vs. 44.61 events per 1,000 person-years with regular salt (rate ratio, 0.88; 95% CI, 0.82-0.95; P < .001).
The rate of serious adverse events attributed to hyperkalemia was not significantly higher with the salt substitute than with regular salt (3.35 events vs. 3.30 events per 1,000 person-years; rate ratio, 1.04; 95% CI, 0.80-1.37; P = .76).
Dr. Neal reported that 7%-8% of the control group started using salt substitute over the study period, so these results have likely underestimated the true effect of switching to a salt substitute product.
Noting that about 10 million cardiovascular events occur each year in China, he said the study results suggested that using salt substitute instead of regular salt could prevent about 10% of these events.
Food manufacturers must make changes
Dr. Neal acknowledged that a limitation of the study was the fact it was conducted in a single country, which would raise issues of generalizability. But he said he believes the results are generalizable to other populations.
Those who would get the most benefit from switching to a salt substitute are those who consume large amounts of discretionary salt – salt added at home at the time of cooking for preservation of food or seasoning. “This is salt that is easy to replace with salt substitute,” Dr. Neal noted.
“There are more than 5 billion people in the world that consume more than 50% of their salt intake as discretionary salt – mainly in the developing world. These people would expect to get significant health benefits from a switch to salt substitute.”
He pointed out that salt substitute is low cost and is easy to manufacture. “Salt substitutes cost around 50% more than regular salt, but this translates into just a dollar or two per person per year to make the switch.”
Dr. Neal said the results also apply to higher-income countries but must be implemented by governments and food manufactures, as most salt in these countries comes from processed foods.
“This study provides strong evidence to take to the food industry,” he concluded. “We would like to see food manufacturers switch to using salt substitute and for salt substitute products to be widely available on supermarket shelves. We also urge governments to take action to promote use of salt substitutes over regular salt. This could take the form of taxing regular salt or subsidies for use of salt substitutes.”
The SSaSS was supported by grants from the National Health and Medical Research Council of Australia. Dr. Neal reports no disclosures. Dr. Ingelfinger is employed by the New England Journal of Medicine as deputy editor.
A version of this article first appeared on Medscape.com.
Switching from regular salt to a low-sodium salt substitute has major public health benefits, including a reduction in stroke, cardiovascular events, and death, a new landmark study shows.
The Salt Substitute and Stroke Study (SSaSS) was conducted in 21,000 people with a history of stroke or high blood pressure in rural China, with half of them using a lower-sodium salt substitute instead of regular salt.
Results showed that after 5 years, those using the salt substitute had a 14% reduction in stroke, a 13% reduction in major cardiovascular events, and a 12% reduction in death. These benefits were achieved without any apparent adverse effects.
The trial was presented by Bruce Neal, MB, George Institute for Global Health, Sydney, Australia, on Aug. 29 at the virtual European Society of Cardiology (ESC) Congress 2021. They were simultaneously published online in the New England Journal of Medicine.
“This is one of the largest dietary intervention trials ever conducted and has shown very clear evidence of protection against stroke, cardiovascular events, and premature death, with no adverse effects with a very simple and low-cost intervention,” Dr. Neal concluded. “This is a very easy thing to work into the diet. You just replace regular salt with a substitute that looks and tastes almost identical,” he added.
Addressing the issue of whether these results are generalizable to other populations, Dr. Neal said, “We believe the results are relevant to everyone who eats salt.
“The way the body manages sodium and potassium and their association with blood pressure is highly consistent across different populations,” he said. “Almost everyone, with the exception of a few people with serious kidney disease, should be avoiding salt or switching to a salt substitute and expect to see some benefit of this.”
Commentators at the ESC presentation lauded the study as “magnificent,” with “extraordinary” results and “very powerful implications.”
Designated discussant, hypertension expert Bryan Williams, MD, University College London, said the SSaSS was “probably the most important study with regards to public health that we will see.” He described the reductions in stroke, cardiovascular events, and death as “extraordinary for such a simple intervention.”
Dr. Williams added: “Those who have doubted the benefits of salt restriction must now admit they were wrong. The debate stops here. The data are in. Global health interventions to implement these findings must now begin.”
He also highlighted the large number of events in the trial. “This was a large, pragmatic, long-duration study in a high-risk population, and with 5,000 cardiovascular events it gives enormous power to show benefits.”
Chair of the ESC session, Barbara Casadei, MD, DPhil, John Radcliffe Hospital, Oxford (England), said the SSaSS “will change the way we think about salt and be remembered for years to come.”
Noting that the benefits were seen in all subgroups across the study, Bertram Pitt, MD, University of Michigan, Ann Arbor, was particularly excited about the stroke reduction seen in patients with diabetes, noting that several recent trials of new diabetes drugs have not managed to show a reduction in stroke.
“For patients with diabetes, this is a really important intervention,” he stated.
However, an editorial accompanying the NEJM publication gave a somewhat less enthusiastic response to the study than the ESC commentators.
Julie R. Ingelfinger, MD, deputy editor of the journal, points out that serial monitoring of potassium levels was not performed in the trial, so it is possible that hyperkalemic episodes were not detected, and persons with a history of medical conditions that may be associated with hyperkalemia were not studied.
She also noted that because the salt substitute was distributed to families, it would have been instructive to have data on the household members without risk factors, but no such data were obtained.
“Overall, the SSaSS provides some intriguing hints, but wider effectiveness is hard to predict, given limited generalizability,” she concluded.
Cluster-randomized trial
The SSaSS was an open-label, cluster-randomized trial involving 20,995 people from 600 villages in rural China who had a history of stroke or were 60 years of age or older and had uncontrolled hypertension. Patients with a history of severe kidney disease and those taking potassium supplements or potassium-sparing diuretics were excluded.
They were randomly assigned in a 1:1 ratio to the intervention group, in which the participants used a salt substitute (roughly 75% sodium chloride and 25% potassium chloride), or to the control group, in which the participants continued to use regular salt (100% sodium chloride).
Results showed that after a mean follow-up of 4.74 years, systolic blood pressure was reduced by 3.3 mm Hg in the salt substitute group.
The rate of stroke, the primary endpoint, was 29.14 events per 1,000 person-years in the salt substitute group vs. 33.65 events per 1,000 person-years with regular salt (rate ratio, 0.86; 95% confidence interval, 0.77-0.96; P = .006).
The rates of major cardiovascular events were 49.09 events per 1,000 person-years in the salt substitute group vs. 56.29 events per 1,000 person-years in those using regular salt (rate ratio, 0.87; 95% CI, 0.80-0.94; P < .001).
And the rate of death was 39.28 events per 1,000 person-years with the salt substitute vs. 44.61 events per 1,000 person-years with regular salt (rate ratio, 0.88; 95% CI, 0.82-0.95; P < .001).
The rate of serious adverse events attributed to hyperkalemia was not significantly higher with the salt substitute than with regular salt (3.35 events vs. 3.30 events per 1,000 person-years; rate ratio, 1.04; 95% CI, 0.80-1.37; P = .76).
Dr. Neal reported that 7%-8% of the control group started using salt substitute over the study period, so these results have likely underestimated the true effect of switching to a salt substitute product.
Noting that about 10 million cardiovascular events occur each year in China, he said the study results suggested that using salt substitute instead of regular salt could prevent about 10% of these events.
Food manufacturers must make changes
Dr. Neal acknowledged that a limitation of the study was the fact it was conducted in a single country, which would raise issues of generalizability. But he said he believes the results are generalizable to other populations.
Those who would get the most benefit from switching to a salt substitute are those who consume large amounts of discretionary salt – salt added at home at the time of cooking for preservation of food or seasoning. “This is salt that is easy to replace with salt substitute,” Dr. Neal noted.
“There are more than 5 billion people in the world that consume more than 50% of their salt intake as discretionary salt – mainly in the developing world. These people would expect to get significant health benefits from a switch to salt substitute.”
He pointed out that salt substitute is low cost and is easy to manufacture. “Salt substitutes cost around 50% more than regular salt, but this translates into just a dollar or two per person per year to make the switch.”
Dr. Neal said the results also apply to higher-income countries but must be implemented by governments and food manufactures, as most salt in these countries comes from processed foods.
“This study provides strong evidence to take to the food industry,” he concluded. “We would like to see food manufacturers switch to using salt substitute and for salt substitute products to be widely available on supermarket shelves. We also urge governments to take action to promote use of salt substitutes over regular salt. This could take the form of taxing regular salt or subsidies for use of salt substitutes.”
The SSaSS was supported by grants from the National Health and Medical Research Council of Australia. Dr. Neal reports no disclosures. Dr. Ingelfinger is employed by the New England Journal of Medicine as deputy editor.
A version of this article first appeared on Medscape.com.
MASTER DAPT: 1 month DAPT enough after high-bleeding-risk PCI
Another trial has added to the movement toward shortening the duration of dual antiplatelet therapy (DAPT) after percutaneous coronary intervention (PCI).
In the MASTER DAPT trial involving patients at high risk for bleeding who had undergone implantation of a biodegradable-polymer sirolimus-eluting stent, switching from DAPT to single antiplatelet therapy at a median of 34 days after PCI was noninferior to the continuation of DAPT treatment for a median duration of 193 days with regard to the incidence of major adverse cardiac or cerebral events, and was associated with a lower incidence of major or clinically relevant bleeding.
The results of the study were presented by Marco Valgimigli, MD, Cardiocentro Ticino Institute, Lugano, Switzerland, on Aug. 28 at the virtual European Society of Cardiology (ESC) Congress 2021. They were simultaneously published online in the New England Journal of Medicine.
“It has been suggested in previous studies that if patients are at high bleeding risk, then they do not seem to derive ischemic benefit from prolonging DAPT, they just get the increased bleeding risk,” Dr. Valgimigli said. “But this has never been prospectively tested until now.”
He pointed out that patients at high bleeding risk are a large group, representing up to 40% of patients undergoing PCI, and the MASTER DAPT trial included “all-comer” high-bleeding-risk patients with no selection based on ischemic risk.
The trial was very well received by commentators at the ESC Hot Line presentation.
Chair of the session, Roxana Mehran, MD, Icahn School of Medicine at Mount Sinai, New York, described the trial as “practice-changing.”
And Robert Byrne, MD, Mater Private Hospital, Dublin, added: “This is a standout trial. We have become more comfortable with abbreviated DAPT in high-bleeding-risk patients, but definite evidence for this has been lacking until now. This study tells us that just 1 month of DAPT appears to be safe in that there was no increase in ischemic complications and there was a clear reduction in bleeding.”
The MASTER DAPT study involved 4,579 patients at high bleeding risk who had undergone implantation of a biodegradable-polymer sirolimus-eluting coronary stent (Ultimaster, Terumo). Around half the patients had PCI for acute coronary syndrome (ACS) and half had it electively. One month after PCI they were randomly assigned to discontinue DAPT immediately (abbreviated therapy) or to continue it for at least 2 additional months (standard therapy).
The three co-primary outcomes were net adverse clinical events (a composite of death from any cause, myocardial infarction, stroke, or major bleeding), major adverse cardiac or cerebral events (a composite of death from any cause, myocardial infarction, or stroke), and major or clinically relevant nonmajor bleeding, all assessed cumulatively at 335 days. The first two outcomes were assessed for noninferiority in the per-protocol population, and the third outcome for superiority in the intention-to-treat population.
Dual antiplatelet therapy consisted of aspirin plus a P2Y12 inhibitor. The choices of the type of P2Y12 inhibitor for DAPT and the type of monotherapy after the discontinuation of DAPT were at the discretion of the investigator. Clopidogrel was the most popular choice, used as monotherapy in 54% of the patients in the abbreviated-therapy group and as part of DAPT in 79% of patients in the standard-therapy group.
Results showed that net adverse clinical events occurred in 7.5% of the abbreviated-therapy group and in 7.7% of the standard-therapy group (difference, –0.23 percentage points; 95% confidence interval, –1.80 to 1.33 percentage points; P < .001 for noninferiority).
Major adverse cardiac or cerebral events occurred in 6.1% of the abbreviated-therapy group and 5.9% of standard therapy group (difference, 0.11 percentage points; 95% CI, –1.29 to 1.51 percentage points; P = .001 for noninferiority).
Reduction in bleeding driven by BARC-2
Major bleeding or clinically relevant nonmajor bleeding occurred in 6.5% in the abbreviated-therapy group and in 9.4% in the standard-therapy group (difference, –2.82 percentage points; 95% CI, –4.40 to –1.24 percentage points; P < .001 for superiority).
“This is a highly statistically significant reduction in bleeding giving a number needed to treat of 35,” Dr. Valgimigli said.
The lower risk for bleeding in the abbreviated-therapy group was mainly due to the lower incidence of clinically relevant nonmajor bleeding events (BARC type 2) in this group than in the standard-therapy group (4.5% vs. 6.8%).
During the discussion, Dr. Byrne pointed out that the most serious type of bleeding (BARC type 3-5) was not reduced in the abbreviated DAPT group.
Dr. Valgimigli responded that the investigators were surprised about that because previous studies indicated that this most serious bleeding would be reduced, but he suggested that this may be explained by the standard group receiving 3-6 months of DAPT rather than a year or more in previous studies. “Having said that, BARC-2 bleeding is not a trivial event,” he added.
Can results be applied to other stents?
Dr. Byrne also questioned whether the results can be applied to patients receiving other types of stents – not just Ultimaster, which is not available everywhere. Dr. Valgimigli highlighted the low rate of stent thrombosis seen with the Ultimaster stent and said, “I would be scared to assume these results are reproducible with other stents.”
But Dr. Mehran challenged this view, saying, “I’m not so sure about that. I think we can probably extrapolate.”
In an interview, Dr. Mehran added: “I think this is one of the much-needed studies in our field. For the first time, we have a randomized trial on duration of DAPT in high-bleeding-risk patients. The study was inclusive, and enrolled truly high-bleeding-risk patients, including those on oral anticoagulants.
“These results show that, although high-bleeding-risk patients are at high risk of ischemic events, just 1 month of DAPT works well for them regardless, by reducing bleeding, net adverse clinical events, and without increasing ischemic events,” she concluded.
In an editorial accompanying the publication, E. Magnus Ohman, MB, from Duke University, Durham, N.C., pointed out the wide CIs in the results, which he said introduced some uncertainly to the findings.
But he concluded that: “The findings of Dr. Valgimigli and colleagues are important and move us toward a shorter and simpler antithrombotic strategy after PCI.”
In an interview, Dr. Ohman pointed out that the Ultimaster stent is not available in the United States. “We have to think about whether this stent would perform differently to other third- or fourth-generation stents. I wouldn’t have thought so, but it is hard to say for sure.
“All in all, we are looking at shorter periods of DAPT now after PCI. Several trials have now suggested that is the way to go. The forthcoming U.S. PCI guidelines should put all the studies together and come up with recommendations on different patient groups,” he concluded.
Several commentators said they would like to see the data on the patients receiving oral anticoagulants in the study before making firm conclusions on how to translate the results into clinical practice. “This is such an important group. It is difficult to interpret the results without this data,” Dr. Ohman noted. Patients receiving oral anticoagulants, who made up 36% of the study population, will be the subject of a separate report to be presented at the ESC meeting.
The MASTER DAPT trial was supported by Terumo. Dr. Valgimigli reports research grants from Terumo, Abbott, and SMT and consulting or speaker fees from Terumo, Abbott, Daiichi Sankyo, Chiesi, Vesalio, Vifor, Avimedica, Medtronic, Boston Scientific, and AstraZeneca. Dr. Ohman reports grants from Abiomed, grants from Chiesi USA, personal fees from Cara Therapeutics, Genentech, Imbria, Impulse Dynamics, Milestone Pharmaceuticals, XyloCor, Cytokinetics, Dispersol, Otsuka, Pfizer, Cytosorbents, Neurocrine, and Paradigm, outside the submitted work.
A version of this article first appeared on Medscape.com.
Another trial has added to the movement toward shortening the duration of dual antiplatelet therapy (DAPT) after percutaneous coronary intervention (PCI).
In the MASTER DAPT trial involving patients at high risk for bleeding who had undergone implantation of a biodegradable-polymer sirolimus-eluting stent, switching from DAPT to single antiplatelet therapy at a median of 34 days after PCI was noninferior to the continuation of DAPT treatment for a median duration of 193 days with regard to the incidence of major adverse cardiac or cerebral events, and was associated with a lower incidence of major or clinically relevant bleeding.
The results of the study were presented by Marco Valgimigli, MD, Cardiocentro Ticino Institute, Lugano, Switzerland, on Aug. 28 at the virtual European Society of Cardiology (ESC) Congress 2021. They were simultaneously published online in the New England Journal of Medicine.
“It has been suggested in previous studies that if patients are at high bleeding risk, then they do not seem to derive ischemic benefit from prolonging DAPT, they just get the increased bleeding risk,” Dr. Valgimigli said. “But this has never been prospectively tested until now.”
He pointed out that patients at high bleeding risk are a large group, representing up to 40% of patients undergoing PCI, and the MASTER DAPT trial included “all-comer” high-bleeding-risk patients with no selection based on ischemic risk.
The trial was very well received by commentators at the ESC Hot Line presentation.
Chair of the session, Roxana Mehran, MD, Icahn School of Medicine at Mount Sinai, New York, described the trial as “practice-changing.”
And Robert Byrne, MD, Mater Private Hospital, Dublin, added: “This is a standout trial. We have become more comfortable with abbreviated DAPT in high-bleeding-risk patients, but definite evidence for this has been lacking until now. This study tells us that just 1 month of DAPT appears to be safe in that there was no increase in ischemic complications and there was a clear reduction in bleeding.”
The MASTER DAPT study involved 4,579 patients at high bleeding risk who had undergone implantation of a biodegradable-polymer sirolimus-eluting coronary stent (Ultimaster, Terumo). Around half the patients had PCI for acute coronary syndrome (ACS) and half had it electively. One month after PCI they were randomly assigned to discontinue DAPT immediately (abbreviated therapy) or to continue it for at least 2 additional months (standard therapy).
The three co-primary outcomes were net adverse clinical events (a composite of death from any cause, myocardial infarction, stroke, or major bleeding), major adverse cardiac or cerebral events (a composite of death from any cause, myocardial infarction, or stroke), and major or clinically relevant nonmajor bleeding, all assessed cumulatively at 335 days. The first two outcomes were assessed for noninferiority in the per-protocol population, and the third outcome for superiority in the intention-to-treat population.
Dual antiplatelet therapy consisted of aspirin plus a P2Y12 inhibitor. The choices of the type of P2Y12 inhibitor for DAPT and the type of monotherapy after the discontinuation of DAPT were at the discretion of the investigator. Clopidogrel was the most popular choice, used as monotherapy in 54% of the patients in the abbreviated-therapy group and as part of DAPT in 79% of patients in the standard-therapy group.
Results showed that net adverse clinical events occurred in 7.5% of the abbreviated-therapy group and in 7.7% of the standard-therapy group (difference, –0.23 percentage points; 95% confidence interval, –1.80 to 1.33 percentage points; P < .001 for noninferiority).
Major adverse cardiac or cerebral events occurred in 6.1% of the abbreviated-therapy group and 5.9% of standard therapy group (difference, 0.11 percentage points; 95% CI, –1.29 to 1.51 percentage points; P = .001 for noninferiority).
Reduction in bleeding driven by BARC-2
Major bleeding or clinically relevant nonmajor bleeding occurred in 6.5% in the abbreviated-therapy group and in 9.4% in the standard-therapy group (difference, –2.82 percentage points; 95% CI, –4.40 to –1.24 percentage points; P < .001 for superiority).
“This is a highly statistically significant reduction in bleeding giving a number needed to treat of 35,” Dr. Valgimigli said.
The lower risk for bleeding in the abbreviated-therapy group was mainly due to the lower incidence of clinically relevant nonmajor bleeding events (BARC type 2) in this group than in the standard-therapy group (4.5% vs. 6.8%).
During the discussion, Dr. Byrne pointed out that the most serious type of bleeding (BARC type 3-5) was not reduced in the abbreviated DAPT group.
Dr. Valgimigli responded that the investigators were surprised about that because previous studies indicated that this most serious bleeding would be reduced, but he suggested that this may be explained by the standard group receiving 3-6 months of DAPT rather than a year or more in previous studies. “Having said that, BARC-2 bleeding is not a trivial event,” he added.
Can results be applied to other stents?
Dr. Byrne also questioned whether the results can be applied to patients receiving other types of stents – not just Ultimaster, which is not available everywhere. Dr. Valgimigli highlighted the low rate of stent thrombosis seen with the Ultimaster stent and said, “I would be scared to assume these results are reproducible with other stents.”
But Dr. Mehran challenged this view, saying, “I’m not so sure about that. I think we can probably extrapolate.”
In an interview, Dr. Mehran added: “I think this is one of the much-needed studies in our field. For the first time, we have a randomized trial on duration of DAPT in high-bleeding-risk patients. The study was inclusive, and enrolled truly high-bleeding-risk patients, including those on oral anticoagulants.
“These results show that, although high-bleeding-risk patients are at high risk of ischemic events, just 1 month of DAPT works well for them regardless, by reducing bleeding, net adverse clinical events, and without increasing ischemic events,” she concluded.
In an editorial accompanying the publication, E. Magnus Ohman, MB, from Duke University, Durham, N.C., pointed out the wide CIs in the results, which he said introduced some uncertainly to the findings.
But he concluded that: “The findings of Dr. Valgimigli and colleagues are important and move us toward a shorter and simpler antithrombotic strategy after PCI.”
In an interview, Dr. Ohman pointed out that the Ultimaster stent is not available in the United States. “We have to think about whether this stent would perform differently to other third- or fourth-generation stents. I wouldn’t have thought so, but it is hard to say for sure.
“All in all, we are looking at shorter periods of DAPT now after PCI. Several trials have now suggested that is the way to go. The forthcoming U.S. PCI guidelines should put all the studies together and come up with recommendations on different patient groups,” he concluded.
Several commentators said they would like to see the data on the patients receiving oral anticoagulants in the study before making firm conclusions on how to translate the results into clinical practice. “This is such an important group. It is difficult to interpret the results without this data,” Dr. Ohman noted. Patients receiving oral anticoagulants, who made up 36% of the study population, will be the subject of a separate report to be presented at the ESC meeting.
The MASTER DAPT trial was supported by Terumo. Dr. Valgimigli reports research grants from Terumo, Abbott, and SMT and consulting or speaker fees from Terumo, Abbott, Daiichi Sankyo, Chiesi, Vesalio, Vifor, Avimedica, Medtronic, Boston Scientific, and AstraZeneca. Dr. Ohman reports grants from Abiomed, grants from Chiesi USA, personal fees from Cara Therapeutics, Genentech, Imbria, Impulse Dynamics, Milestone Pharmaceuticals, XyloCor, Cytokinetics, Dispersol, Otsuka, Pfizer, Cytosorbents, Neurocrine, and Paradigm, outside the submitted work.
A version of this article first appeared on Medscape.com.
Another trial has added to the movement toward shortening the duration of dual antiplatelet therapy (DAPT) after percutaneous coronary intervention (PCI).
In the MASTER DAPT trial involving patients at high risk for bleeding who had undergone implantation of a biodegradable-polymer sirolimus-eluting stent, switching from DAPT to single antiplatelet therapy at a median of 34 days after PCI was noninferior to the continuation of DAPT treatment for a median duration of 193 days with regard to the incidence of major adverse cardiac or cerebral events, and was associated with a lower incidence of major or clinically relevant bleeding.
The results of the study were presented by Marco Valgimigli, MD, Cardiocentro Ticino Institute, Lugano, Switzerland, on Aug. 28 at the virtual European Society of Cardiology (ESC) Congress 2021. They were simultaneously published online in the New England Journal of Medicine.
“It has been suggested in previous studies that if patients are at high bleeding risk, then they do not seem to derive ischemic benefit from prolonging DAPT, they just get the increased bleeding risk,” Dr. Valgimigli said. “But this has never been prospectively tested until now.”
He pointed out that patients at high bleeding risk are a large group, representing up to 40% of patients undergoing PCI, and the MASTER DAPT trial included “all-comer” high-bleeding-risk patients with no selection based on ischemic risk.
The trial was very well received by commentators at the ESC Hot Line presentation.
Chair of the session, Roxana Mehran, MD, Icahn School of Medicine at Mount Sinai, New York, described the trial as “practice-changing.”
And Robert Byrne, MD, Mater Private Hospital, Dublin, added: “This is a standout trial. We have become more comfortable with abbreviated DAPT in high-bleeding-risk patients, but definite evidence for this has been lacking until now. This study tells us that just 1 month of DAPT appears to be safe in that there was no increase in ischemic complications and there was a clear reduction in bleeding.”
The MASTER DAPT study involved 4,579 patients at high bleeding risk who had undergone implantation of a biodegradable-polymer sirolimus-eluting coronary stent (Ultimaster, Terumo). Around half the patients had PCI for acute coronary syndrome (ACS) and half had it electively. One month after PCI they were randomly assigned to discontinue DAPT immediately (abbreviated therapy) or to continue it for at least 2 additional months (standard therapy).
The three co-primary outcomes were net adverse clinical events (a composite of death from any cause, myocardial infarction, stroke, or major bleeding), major adverse cardiac or cerebral events (a composite of death from any cause, myocardial infarction, or stroke), and major or clinically relevant nonmajor bleeding, all assessed cumulatively at 335 days. The first two outcomes were assessed for noninferiority in the per-protocol population, and the third outcome for superiority in the intention-to-treat population.
Dual antiplatelet therapy consisted of aspirin plus a P2Y12 inhibitor. The choices of the type of P2Y12 inhibitor for DAPT and the type of monotherapy after the discontinuation of DAPT were at the discretion of the investigator. Clopidogrel was the most popular choice, used as monotherapy in 54% of the patients in the abbreviated-therapy group and as part of DAPT in 79% of patients in the standard-therapy group.
Results showed that net adverse clinical events occurred in 7.5% of the abbreviated-therapy group and in 7.7% of the standard-therapy group (difference, –0.23 percentage points; 95% confidence interval, –1.80 to 1.33 percentage points; P < .001 for noninferiority).
Major adverse cardiac or cerebral events occurred in 6.1% of the abbreviated-therapy group and 5.9% of standard therapy group (difference, 0.11 percentage points; 95% CI, –1.29 to 1.51 percentage points; P = .001 for noninferiority).
Reduction in bleeding driven by BARC-2
Major bleeding or clinically relevant nonmajor bleeding occurred in 6.5% in the abbreviated-therapy group and in 9.4% in the standard-therapy group (difference, –2.82 percentage points; 95% CI, –4.40 to –1.24 percentage points; P < .001 for superiority).
“This is a highly statistically significant reduction in bleeding giving a number needed to treat of 35,” Dr. Valgimigli said.
The lower risk for bleeding in the abbreviated-therapy group was mainly due to the lower incidence of clinically relevant nonmajor bleeding events (BARC type 2) in this group than in the standard-therapy group (4.5% vs. 6.8%).
During the discussion, Dr. Byrne pointed out that the most serious type of bleeding (BARC type 3-5) was not reduced in the abbreviated DAPT group.
Dr. Valgimigli responded that the investigators were surprised about that because previous studies indicated that this most serious bleeding would be reduced, but he suggested that this may be explained by the standard group receiving 3-6 months of DAPT rather than a year or more in previous studies. “Having said that, BARC-2 bleeding is not a trivial event,” he added.
Can results be applied to other stents?
Dr. Byrne also questioned whether the results can be applied to patients receiving other types of stents – not just Ultimaster, which is not available everywhere. Dr. Valgimigli highlighted the low rate of stent thrombosis seen with the Ultimaster stent and said, “I would be scared to assume these results are reproducible with other stents.”
But Dr. Mehran challenged this view, saying, “I’m not so sure about that. I think we can probably extrapolate.”
In an interview, Dr. Mehran added: “I think this is one of the much-needed studies in our field. For the first time, we have a randomized trial on duration of DAPT in high-bleeding-risk patients. The study was inclusive, and enrolled truly high-bleeding-risk patients, including those on oral anticoagulants.
“These results show that, although high-bleeding-risk patients are at high risk of ischemic events, just 1 month of DAPT works well for them regardless, by reducing bleeding, net adverse clinical events, and without increasing ischemic events,” she concluded.
In an editorial accompanying the publication, E. Magnus Ohman, MB, from Duke University, Durham, N.C., pointed out the wide CIs in the results, which he said introduced some uncertainly to the findings.
But he concluded that: “The findings of Dr. Valgimigli and colleagues are important and move us toward a shorter and simpler antithrombotic strategy after PCI.”
In an interview, Dr. Ohman pointed out that the Ultimaster stent is not available in the United States. “We have to think about whether this stent would perform differently to other third- or fourth-generation stents. I wouldn’t have thought so, but it is hard to say for sure.
“All in all, we are looking at shorter periods of DAPT now after PCI. Several trials have now suggested that is the way to go. The forthcoming U.S. PCI guidelines should put all the studies together and come up with recommendations on different patient groups,” he concluded.
Several commentators said they would like to see the data on the patients receiving oral anticoagulants in the study before making firm conclusions on how to translate the results into clinical practice. “This is such an important group. It is difficult to interpret the results without this data,” Dr. Ohman noted. Patients receiving oral anticoagulants, who made up 36% of the study population, will be the subject of a separate report to be presented at the ESC meeting.
The MASTER DAPT trial was supported by Terumo. Dr. Valgimigli reports research grants from Terumo, Abbott, and SMT and consulting or speaker fees from Terumo, Abbott, Daiichi Sankyo, Chiesi, Vesalio, Vifor, Avimedica, Medtronic, Boston Scientific, and AstraZeneca. Dr. Ohman reports grants from Abiomed, grants from Chiesi USA, personal fees from Cara Therapeutics, Genentech, Imbria, Impulse Dynamics, Milestone Pharmaceuticals, XyloCor, Cytokinetics, Dispersol, Otsuka, Pfizer, Cytosorbents, Neurocrine, and Paradigm, outside the submitted work.
A version of this article first appeared on Medscape.com.