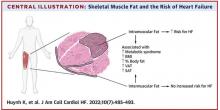
User login
Transplanted pig hearts functioned normally in deceased persons on ventilator support
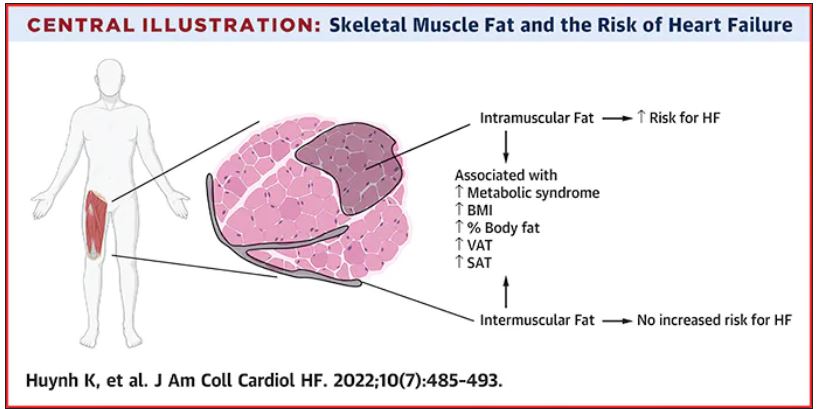
A team of surgeons successfully transplanted genetically engineered pig hearts into two recently deceased people whose bodies were being maintained on ventilatory support – not in the hope of restoring life, but as a proof-of-concept experiment in xenotransplantation that could eventually help to ease the critical shortage of donor organs.
The surgeries were performed on June 16 and July 6, 2022, using porcine hearts from animals genetically engineered to prevent organ rejection and promote adaptive immunity by human recipients
without utilizing unapproved devices or techniques or medications,” said Nader Moazami, MD, surgical director of heart transplantation and chief of the division of heart and lung transplantation and mechanical circulatory support at NYU Langone Health, New York.
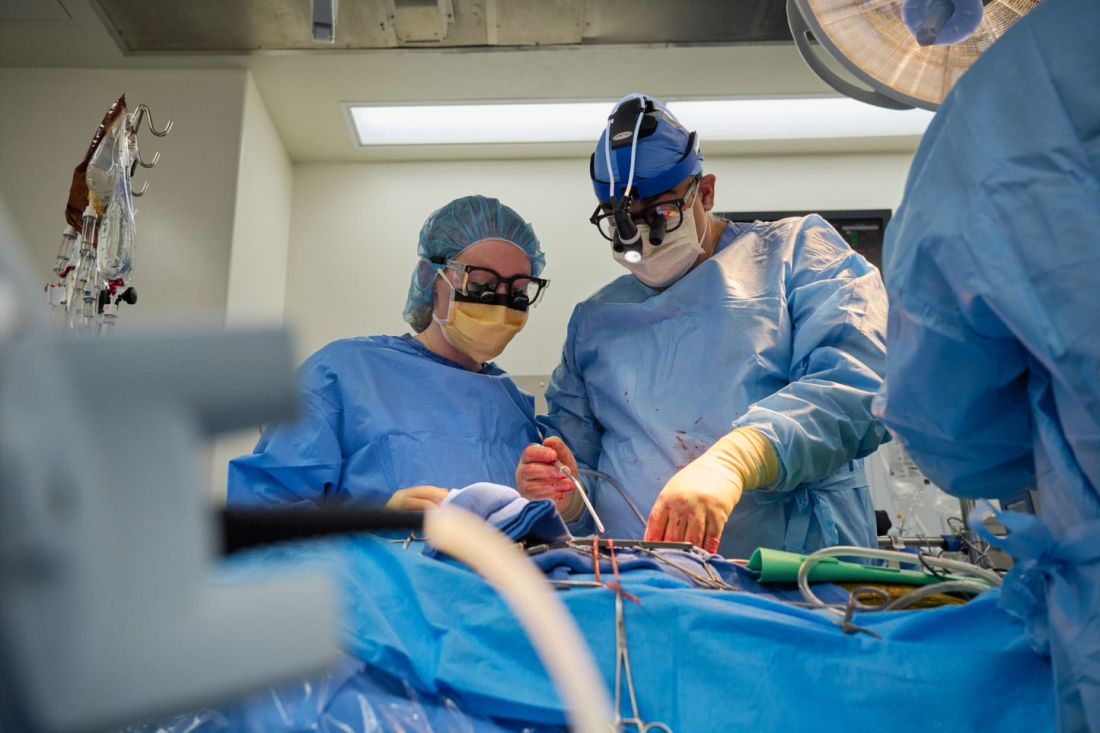
Through 72 hours of postoperative monitoring “we evaluated the heart for functionality and the heart function was completely normal with excellent contractility,” he said at a press briefing announcing early results of the experimental program.
He acknowledged that for the first of the two procedures some surgical modification of the pig heart was required, primarily because of size differences between the donor and recipient.
“Nevertheless, we learned a tremendous amount from the first operation, and when that experience was translated into the second operation it even performed better,” he said.
Alex Reyentovich, MD, medical director of heart transplantation and director of the NYU Langone advanced heart failure program noted that “there are 6 million individuals with heart failure in the United States. About 100,000 of those individuals have end-stage heart failure, and we only do about 3,500 heart transplants a year in the United States, so we have a tremendous deficiency in organs, and there are many people dying waiting for a heart.”
Infection protocols
To date there has been only one xenotransplant of a genetically modified pig heart into a living human recipient, David Bennett Sr., age 57. The surgery, performed at the University of Maryland in January 2022, was initially successful, with the patient able to sit up in bed a few days after the procedure, and the heart performing like a “rock star” according to transplant surgeon Bartley Griffith, MD.
However, Mr. Bennett died 2 months after the procedure from compromise of the organ by an as yet undetermined cause, of which one may have been the heart's infection by porcine cytomegalovirus (CMV).
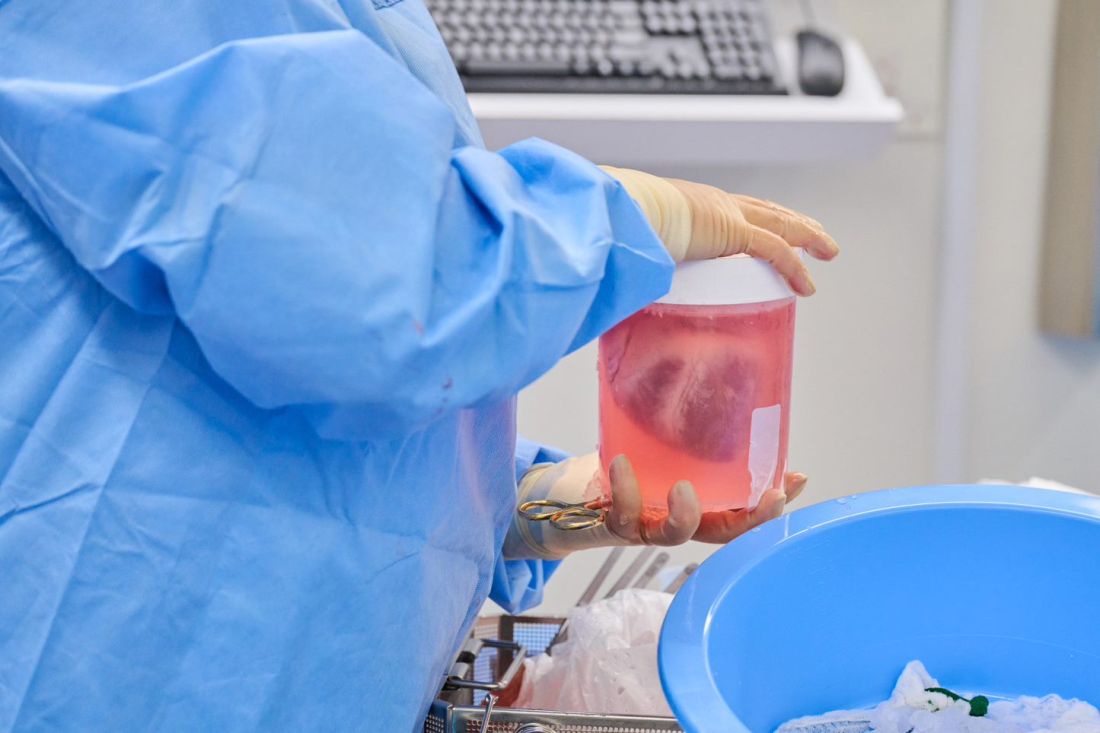
The NYU team, mindful of this potential setback, used more sensitive assays to screen the donor organs for porcine CMV, and implemented protocols to prevent and to monitor for potential zoonotic transmission of porcine endogenous retrovirus.
The procedure used a dedicated operating room and equipment that will not be used for clinical procedures, the team emphasized.
An organ transplant specialist who was not involved in the study commented that there can be unwelcome surprises even with the most rigorous infection prophylaxis protocols.
“I think these are important steps, but they don’t resolve the question of infectious risk. Sometimes viruses or latent infections are only manifested later,” said Jay A. Fishman, MD, associate director of the Massachusetts General Hospital Transplant Center and director of the transplant infectious diseases and compromised host program at the hospital, which is in Boston.
“I think these are important steps, but as you may recall from the Maryland heart transplant experience, when porcine cytomegalovirus was activated, it was a long way into that patient’s course, and so we just don’t know whether something would have been reactivated later,” he said in an interview.
Dr. Fishman noted that experience with xenotransplantation at the University of Maryland and other centers has suggested that immunosuppressive regimens used for human-to-human transplants may not be suited for animal-to-human grafts.
The hearts were taken from pigs genetically modified with knockouts of four porcine genes to prevent rejection – including a gene for a growth hormone that would otherwise cause the heart to continue to expand in the recipient’s chest – and with the addition of six human transgenes encoding for expression of proteins regulating biologic pathways that might be disrupted by incompatibilities across species.
Vietnam veteran
The organ recipients were recently deceased patients who had expressed the clear wish to be organ donors but whose organs were for clinical reasons unsuitable for transplant.
The first recipient was Lawrence Kelly, a Vietnam War veteran and welder who died from heart failure at the age of 72.
“He was an organ donor, and would be so happy to know how much his contribution to this research will help people like him with this heart disease. He was a hero his whole life, and he went out a hero,” said Alice Michael, Mr. Kelly’s partner of 33 years, who also spoke at the briefing.
“It was, I think, one of the most incredible things to see a pig heart pounding away and beating inside the chest of a human being,” said Robert A. Montgomery, MD, DPhil, director of the NYU Transplant Institute, and himself a heart transplant recipient.
Dr. Fishman said he had no relevant conflicts of interest.
This article was updated on 7/12/22 and 7/14/22.
A team of surgeons successfully transplanted genetically engineered pig hearts into two recently deceased people whose bodies were being maintained on ventilatory support – not in the hope of restoring life, but as a proof-of-concept experiment in xenotransplantation that could eventually help to ease the critical shortage of donor organs.
The surgeries were performed on June 16 and July 6, 2022, using porcine hearts from animals genetically engineered to prevent organ rejection and promote adaptive immunity by human recipients
without utilizing unapproved devices or techniques or medications,” said Nader Moazami, MD, surgical director of heart transplantation and chief of the division of heart and lung transplantation and mechanical circulatory support at NYU Langone Health, New York.

Through 72 hours of postoperative monitoring “we evaluated the heart for functionality and the heart function was completely normal with excellent contractility,” he said at a press briefing announcing early results of the experimental program.
He acknowledged that for the first of the two procedures some surgical modification of the pig heart was required, primarily because of size differences between the donor and recipient.
“Nevertheless, we learned a tremendous amount from the first operation, and when that experience was translated into the second operation it even performed better,” he said.
Alex Reyentovich, MD, medical director of heart transplantation and director of the NYU Langone advanced heart failure program noted that “there are 6 million individuals with heart failure in the United States. About 100,000 of those individuals have end-stage heart failure, and we only do about 3,500 heart transplants a year in the United States, so we have a tremendous deficiency in organs, and there are many people dying waiting for a heart.”
Infection protocols
To date there has been only one xenotransplant of a genetically modified pig heart into a living human recipient, David Bennett Sr., age 57. The surgery, performed at the University of Maryland in January 2022, was initially successful, with the patient able to sit up in bed a few days after the procedure, and the heart performing like a “rock star” according to transplant surgeon Bartley Griffith, MD.
However, Mr. Bennett died 2 months after the procedure from compromise of the organ by an as yet undetermined cause, of which one may have been the heart's infection by porcine cytomegalovirus (CMV).

The NYU team, mindful of this potential setback, used more sensitive assays to screen the donor organs for porcine CMV, and implemented protocols to prevent and to monitor for potential zoonotic transmission of porcine endogenous retrovirus.
The procedure used a dedicated operating room and equipment that will not be used for clinical procedures, the team emphasized.
An organ transplant specialist who was not involved in the study commented that there can be unwelcome surprises even with the most rigorous infection prophylaxis protocols.
“I think these are important steps, but they don’t resolve the question of infectious risk. Sometimes viruses or latent infections are only manifested later,” said Jay A. Fishman, MD, associate director of the Massachusetts General Hospital Transplant Center and director of the transplant infectious diseases and compromised host program at the hospital, which is in Boston.
“I think these are important steps, but as you may recall from the Maryland heart transplant experience, when porcine cytomegalovirus was activated, it was a long way into that patient’s course, and so we just don’t know whether something would have been reactivated later,” he said in an interview.
Dr. Fishman noted that experience with xenotransplantation at the University of Maryland and other centers has suggested that immunosuppressive regimens used for human-to-human transplants may not be suited for animal-to-human grafts.
The hearts were taken from pigs genetically modified with knockouts of four porcine genes to prevent rejection – including a gene for a growth hormone that would otherwise cause the heart to continue to expand in the recipient’s chest – and with the addition of six human transgenes encoding for expression of proteins regulating biologic pathways that might be disrupted by incompatibilities across species.
Vietnam veteran
The organ recipients were recently deceased patients who had expressed the clear wish to be organ donors but whose organs were for clinical reasons unsuitable for transplant.
The first recipient was Lawrence Kelly, a Vietnam War veteran and welder who died from heart failure at the age of 72.
“He was an organ donor, and would be so happy to know how much his contribution to this research will help people like him with this heart disease. He was a hero his whole life, and he went out a hero,” said Alice Michael, Mr. Kelly’s partner of 33 years, who also spoke at the briefing.
“It was, I think, one of the most incredible things to see a pig heart pounding away and beating inside the chest of a human being,” said Robert A. Montgomery, MD, DPhil, director of the NYU Transplant Institute, and himself a heart transplant recipient.
Dr. Fishman said he had no relevant conflicts of interest.
This article was updated on 7/12/22 and 7/14/22.
A team of surgeons successfully transplanted genetically engineered pig hearts into two recently deceased people whose bodies were being maintained on ventilatory support – not in the hope of restoring life, but as a proof-of-concept experiment in xenotransplantation that could eventually help to ease the critical shortage of donor organs.
The surgeries were performed on June 16 and July 6, 2022, using porcine hearts from animals genetically engineered to prevent organ rejection and promote adaptive immunity by human recipients
without utilizing unapproved devices or techniques or medications,” said Nader Moazami, MD, surgical director of heart transplantation and chief of the division of heart and lung transplantation and mechanical circulatory support at NYU Langone Health, New York.

Through 72 hours of postoperative monitoring “we evaluated the heart for functionality and the heart function was completely normal with excellent contractility,” he said at a press briefing announcing early results of the experimental program.
He acknowledged that for the first of the two procedures some surgical modification of the pig heart was required, primarily because of size differences between the donor and recipient.
“Nevertheless, we learned a tremendous amount from the first operation, and when that experience was translated into the second operation it even performed better,” he said.
Alex Reyentovich, MD, medical director of heart transplantation and director of the NYU Langone advanced heart failure program noted that “there are 6 million individuals with heart failure in the United States. About 100,000 of those individuals have end-stage heart failure, and we only do about 3,500 heart transplants a year in the United States, so we have a tremendous deficiency in organs, and there are many people dying waiting for a heart.”
Infection protocols
To date there has been only one xenotransplant of a genetically modified pig heart into a living human recipient, David Bennett Sr., age 57. The surgery, performed at the University of Maryland in January 2022, was initially successful, with the patient able to sit up in bed a few days after the procedure, and the heart performing like a “rock star” according to transplant surgeon Bartley Griffith, MD.
However, Mr. Bennett died 2 months after the procedure from compromise of the organ by an as yet undetermined cause, of which one may have been the heart's infection by porcine cytomegalovirus (CMV).

The NYU team, mindful of this potential setback, used more sensitive assays to screen the donor organs for porcine CMV, and implemented protocols to prevent and to monitor for potential zoonotic transmission of porcine endogenous retrovirus.
The procedure used a dedicated operating room and equipment that will not be used for clinical procedures, the team emphasized.
An organ transplant specialist who was not involved in the study commented that there can be unwelcome surprises even with the most rigorous infection prophylaxis protocols.
“I think these are important steps, but they don’t resolve the question of infectious risk. Sometimes viruses or latent infections are only manifested later,” said Jay A. Fishman, MD, associate director of the Massachusetts General Hospital Transplant Center and director of the transplant infectious diseases and compromised host program at the hospital, which is in Boston.
“I think these are important steps, but as you may recall from the Maryland heart transplant experience, when porcine cytomegalovirus was activated, it was a long way into that patient’s course, and so we just don’t know whether something would have been reactivated later,” he said in an interview.
Dr. Fishman noted that experience with xenotransplantation at the University of Maryland and other centers has suggested that immunosuppressive regimens used for human-to-human transplants may not be suited for animal-to-human grafts.
The hearts were taken from pigs genetically modified with knockouts of four porcine genes to prevent rejection – including a gene for a growth hormone that would otherwise cause the heart to continue to expand in the recipient’s chest – and with the addition of six human transgenes encoding for expression of proteins regulating biologic pathways that might be disrupted by incompatibilities across species.
Vietnam veteran
The organ recipients were recently deceased patients who had expressed the clear wish to be organ donors but whose organs were for clinical reasons unsuitable for transplant.
The first recipient was Lawrence Kelly, a Vietnam War veteran and welder who died from heart failure at the age of 72.
“He was an organ donor, and would be so happy to know how much his contribution to this research will help people like him with this heart disease. He was a hero his whole life, and he went out a hero,” said Alice Michael, Mr. Kelly’s partner of 33 years, who also spoke at the briefing.
“It was, I think, one of the most incredible things to see a pig heart pounding away and beating inside the chest of a human being,” said Robert A. Montgomery, MD, DPhil, director of the NYU Transplant Institute, and himself a heart transplant recipient.
Dr. Fishman said he had no relevant conflicts of interest.
This article was updated on 7/12/22 and 7/14/22.
PCOS ups risk of heart complications during delivery period
Pregnant women with polycystic ovary syndrome (PCOS) appear to be at significantly increased risk of experiencing cardiac complications while hospitalized during and after delivery.
An estimated 5 million women of childbearing age in the United States have PCOS, a hormone disorder linked to infertility. PCOS is also known to contribute to the development of cardiometabolic abnormalities like high cholesterol and high blood pressure, which are associated with acute cardiovascular complications during delivery.
But a study, published online in the Journal of the American Heart Association, found that even after accounting for pre-eclampsia, age, comorbidities, and race, PCOS was linked to a 76% increased risk for heart failure, a 79% higher risk of a weakened heart, and an 82% increased risk of having blood clots in the hours and days around giving birth in hospital settings, compared with women without PCOS.
“Perhaps women need a closer follow-up during their pregnancy,” said Erin Michos, MD, MHS, associate director of preventive cardiology at Johns Hopkins Medicine, Baltimore, and a co-author of the study. “They’re counseled about the difficulties of getting pregnant, but what about when they get pregnant?”
Hospitalizations of women with PCOS were also associated with longer stays (3 vs. 2 days) and higher costs ($4,901 vs. $3616; P < .01), compared with women without PCOS.
Over the 17-year analysis period, the number of women with PCOS rose from 569 per 100,000 deliveries to 15,349 per 100,000 deliveries. The researchers attributed the increase in part to greater awareness and diagnosis of the disorder. Dr. Michos and her colleagues used the National Inpatient Sample, managed by the Agency for Healthcare Research and Quality, to pull claims data for women who gave birth in hospitals between 2002 and 2019.
Solutions?
Dr. Michos said there may be more prevention work from og.gyns. to both educate patients about their heart risks during the delivery process and also to refer them to relevant cardiac specialists.
“These women may seek out a gynecologist because of the symptoms, perhaps irregular menses, but along with that should come counseling of the long-term cardiovascular complication,” Dr. Michos said. “And after a pregnancy there should be a good handoff to a primary care provider, so they get a cardiovascular assessment.”
Lifestyle management before, during, and after pregnancy can help prevent the onset of the long-term consequences of cardiac complications during delivery, according to Valerie Baker, MD, director of the division of reproductive endocrinology and infertility at Hopkins Medicine, and her colleagues in a viewpoint published in the journal Fertility and Sterility.
“Once women with PCOS are identified by screening to be at higher risk for [cardiovascular disease], the foundational approach should be lifestyle management followed by statin therapy,” Dr. Baker’s group wrote. “These interventions should include dietary management and physical activity, especially for those who are prediabetic.”
The current study came on the heels of a June 14 meta-analysis by Dr. Michos’ group that found that women with PCOS may be twice as likely as those without PCOS to have coronary artery calcification, a precursor to atherosclerosis and a sign of the early onset of cardiovascular disease.
“We shouldn’t assume that all women of reproductive age are low risk,” Dr. Michos said. “This is the window of time that we can reshape the trajectory early in life.”
The study was supported by the Amato Fund for Women’s Cardiovascular Health research at Johns Hopkins University and through grant support from the American Heart Association (940166). Dr. Michos reported advisory board participation for AstraZeneca, Amarin, Novartis, Novo Nordisk, Bayer, Boehringer Ingelheim, Esperion, and Pfizer. Study coauthor Michael Honigberg, MD, reported consulting fees from CRISPR Therapeutics, unrelated to the present work. The remaining authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Pregnant women with polycystic ovary syndrome (PCOS) appear to be at significantly increased risk of experiencing cardiac complications while hospitalized during and after delivery.
An estimated 5 million women of childbearing age in the United States have PCOS, a hormone disorder linked to infertility. PCOS is also known to contribute to the development of cardiometabolic abnormalities like high cholesterol and high blood pressure, which are associated with acute cardiovascular complications during delivery.
But a study, published online in the Journal of the American Heart Association, found that even after accounting for pre-eclampsia, age, comorbidities, and race, PCOS was linked to a 76% increased risk for heart failure, a 79% higher risk of a weakened heart, and an 82% increased risk of having blood clots in the hours and days around giving birth in hospital settings, compared with women without PCOS.
“Perhaps women need a closer follow-up during their pregnancy,” said Erin Michos, MD, MHS, associate director of preventive cardiology at Johns Hopkins Medicine, Baltimore, and a co-author of the study. “They’re counseled about the difficulties of getting pregnant, but what about when they get pregnant?”
Hospitalizations of women with PCOS were also associated with longer stays (3 vs. 2 days) and higher costs ($4,901 vs. $3616; P < .01), compared with women without PCOS.
Over the 17-year analysis period, the number of women with PCOS rose from 569 per 100,000 deliveries to 15,349 per 100,000 deliveries. The researchers attributed the increase in part to greater awareness and diagnosis of the disorder. Dr. Michos and her colleagues used the National Inpatient Sample, managed by the Agency for Healthcare Research and Quality, to pull claims data for women who gave birth in hospitals between 2002 and 2019.
Solutions?
Dr. Michos said there may be more prevention work from og.gyns. to both educate patients about their heart risks during the delivery process and also to refer them to relevant cardiac specialists.
“These women may seek out a gynecologist because of the symptoms, perhaps irregular menses, but along with that should come counseling of the long-term cardiovascular complication,” Dr. Michos said. “And after a pregnancy there should be a good handoff to a primary care provider, so they get a cardiovascular assessment.”
Lifestyle management before, during, and after pregnancy can help prevent the onset of the long-term consequences of cardiac complications during delivery, according to Valerie Baker, MD, director of the division of reproductive endocrinology and infertility at Hopkins Medicine, and her colleagues in a viewpoint published in the journal Fertility and Sterility.
“Once women with PCOS are identified by screening to be at higher risk for [cardiovascular disease], the foundational approach should be lifestyle management followed by statin therapy,” Dr. Baker’s group wrote. “These interventions should include dietary management and physical activity, especially for those who are prediabetic.”
The current study came on the heels of a June 14 meta-analysis by Dr. Michos’ group that found that women with PCOS may be twice as likely as those without PCOS to have coronary artery calcification, a precursor to atherosclerosis and a sign of the early onset of cardiovascular disease.
“We shouldn’t assume that all women of reproductive age are low risk,” Dr. Michos said. “This is the window of time that we can reshape the trajectory early in life.”
The study was supported by the Amato Fund for Women’s Cardiovascular Health research at Johns Hopkins University and through grant support from the American Heart Association (940166). Dr. Michos reported advisory board participation for AstraZeneca, Amarin, Novartis, Novo Nordisk, Bayer, Boehringer Ingelheim, Esperion, and Pfizer. Study coauthor Michael Honigberg, MD, reported consulting fees from CRISPR Therapeutics, unrelated to the present work. The remaining authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Pregnant women with polycystic ovary syndrome (PCOS) appear to be at significantly increased risk of experiencing cardiac complications while hospitalized during and after delivery.
An estimated 5 million women of childbearing age in the United States have PCOS, a hormone disorder linked to infertility. PCOS is also known to contribute to the development of cardiometabolic abnormalities like high cholesterol and high blood pressure, which are associated with acute cardiovascular complications during delivery.
But a study, published online in the Journal of the American Heart Association, found that even after accounting for pre-eclampsia, age, comorbidities, and race, PCOS was linked to a 76% increased risk for heart failure, a 79% higher risk of a weakened heart, and an 82% increased risk of having blood clots in the hours and days around giving birth in hospital settings, compared with women without PCOS.
“Perhaps women need a closer follow-up during their pregnancy,” said Erin Michos, MD, MHS, associate director of preventive cardiology at Johns Hopkins Medicine, Baltimore, and a co-author of the study. “They’re counseled about the difficulties of getting pregnant, but what about when they get pregnant?”
Hospitalizations of women with PCOS were also associated with longer stays (3 vs. 2 days) and higher costs ($4,901 vs. $3616; P < .01), compared with women without PCOS.
Over the 17-year analysis period, the number of women with PCOS rose from 569 per 100,000 deliveries to 15,349 per 100,000 deliveries. The researchers attributed the increase in part to greater awareness and diagnosis of the disorder. Dr. Michos and her colleagues used the National Inpatient Sample, managed by the Agency for Healthcare Research and Quality, to pull claims data for women who gave birth in hospitals between 2002 and 2019.
Solutions?
Dr. Michos said there may be more prevention work from og.gyns. to both educate patients about their heart risks during the delivery process and also to refer them to relevant cardiac specialists.
“These women may seek out a gynecologist because of the symptoms, perhaps irregular menses, but along with that should come counseling of the long-term cardiovascular complication,” Dr. Michos said. “And after a pregnancy there should be a good handoff to a primary care provider, so they get a cardiovascular assessment.”
Lifestyle management before, during, and after pregnancy can help prevent the onset of the long-term consequences of cardiac complications during delivery, according to Valerie Baker, MD, director of the division of reproductive endocrinology and infertility at Hopkins Medicine, and her colleagues in a viewpoint published in the journal Fertility and Sterility.
“Once women with PCOS are identified by screening to be at higher risk for [cardiovascular disease], the foundational approach should be lifestyle management followed by statin therapy,” Dr. Baker’s group wrote. “These interventions should include dietary management and physical activity, especially for those who are prediabetic.”
The current study came on the heels of a June 14 meta-analysis by Dr. Michos’ group that found that women with PCOS may be twice as likely as those without PCOS to have coronary artery calcification, a precursor to atherosclerosis and a sign of the early onset of cardiovascular disease.
“We shouldn’t assume that all women of reproductive age are low risk,” Dr. Michos said. “This is the window of time that we can reshape the trajectory early in life.”
The study was supported by the Amato Fund for Women’s Cardiovascular Health research at Johns Hopkins University and through grant support from the American Heart Association (940166). Dr. Michos reported advisory board participation for AstraZeneca, Amarin, Novartis, Novo Nordisk, Bayer, Boehringer Ingelheim, Esperion, and Pfizer. Study coauthor Michael Honigberg, MD, reported consulting fees from CRISPR Therapeutics, unrelated to the present work. The remaining authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Adding salt to food linked to higher risk of premature death
in a new study.
In the study of more than 500,000 people, compared with those who never or rarely added salt, those who always added salt to their food had a 28% increased risk of dying prematurely (defined as death before the age of 75 years).
Results also showed that adding salt to food was linked to a lower life expectancy. At the age of 50 years, life expectancy was reduced by 1.5 years in women and by 2.28 years in men who always added salt to their food, compared with those who never or rarely did.
However, these increased risks appeared to be attenuated with increasing intakes of high-potassium foods (vegetables and fruits).
The study was published online in the European Heart Journal.
“As far as we are aware, this is the first study to analyze adding salt to meals as a unique measurement for dietary sodium intake. Such a measure is less likely affected by other dietary components, especially potassium intake,” senior author Lu Qi, MD, Tulane University, New Orleans, told this news organization.
“Our study provides supportive evidence from a novel perspective to show the adverse effects of high sodium intake on human health, which is still a controversial topic. Our findings support the advice that reduction of salt intake by reducing the salt added to meals may benefit health and improve life expectancy. Our results also suggest that high intakes of fruits and vegetables are beneficial regarding lowering the adverse effects of salt,” he added.
Link between dietary salt and health is subject of longstanding debate
The researchers explained that the relationship between dietary salt intake and health remains a subject of longstanding debate, with previous studies on the association between sodium intake and mortality having shown conflicting results.
They attributed the inconsistent results to the low accuracy of sodium measurement, noting that sodium intake varies widely from day to day, but the majority of previous studies have largely relied on a single day’s urine collection or dietary survey for estimating the sodium intake, which is inadequate to assess an individual’s usual consumption levels.
They also pointed out that it is difficult to separate the contributions of intakes of sodium and potassium to health based on current methods for measuring dietary sodium and potassium, and this may confound the association between sodium intake and health outcomes.
They noted that the hypothesis that a high-potassium intake may attenuate the adverse association of high-sodium intake with health outcomes has been proposed for many years, but studies assessing the interaction between sodium intake and potassium intake on the risk of mortality are scarce.
Adding salt to food at the table is a common eating behavior directly related to an individual’s long-term preference for salty tasting foods and habitual salt intake, the authors said, adding that commonly used table salt contains 97%-99% sodium chloride, minimizing the potential confounding effects of other dietary factors including potassium. “Therefore, adding salt to foods provides a unique assessment to evaluate the association between habitual sodium intake and mortality.”
UK Biobank study
For the current study Dr. Qi and colleagues analyzed data from 501,379 people taking part in the UK Biobank study. When joining the study between 2006 and 2010, the participants were asked whether they added salt to their foods never/rarely, sometimes, usually or always. Participants were then followed for a median of 9 years.
After adjustment for sex, age, race, smoking, moderate drinking, body mass index, physical activity, Townsend deprivation index, high cholesterol, chronic kidney disease, diabetes, cardiovascular disease, and cancer, results showed an increasing risk of all-cause premature mortality rose with increasing frequency of adding salt to foods.
The adjusted hazard ratios, compared with those who never or rarely added salt, were 1.02 (95% CI, 0.99-1.06) for those who added salt sometimes, 1.07 (95% CI, 1.02-1.11) for those who usually added salt, and 1.28 (95% CI, 1.20-1.35) for those who always added salt.
The researchers also estimated the lower survival time caused by the high frequency of adding salt to foods. At age 50, women who always added salt to foods had an average 1.50 fewer years of life expectancy, and men who always added salt had an average 2.28 fewer years of life expectancy, as compared with their counterparts who never/rarely added salt to foods.
For cause-specific premature mortality, results showed that higher frequency of adding salt to foods was significantly associated with a higher risk of cardiovascular mortality and cancer mortality, but not for dementia mortality or respiratory mortality. For the subtypes of cardiovascular mortality, adding salt to foods was significantly associated with higher risk of stroke mortality but not coronary heart disease mortality.
Other analyses suggested that the association of adding salt to foods with an increased risk of premature mortality appeared to be attenuated with increasing intake of food high in potassium (fruits and vegetables).
The authors point out that the amounts of discretionary sodium intake (the salt used at the table or in home cooking) have been largely overlooked in previous studies, even though adding salt to foods accounts for a considerable proportion of total sodium intake (6%-20%) in Western diets.
“Our findings also support the notion that even a modest reduction in sodium intake is likely to result in substantial health benefits, especially when it is achieved in the general population,” they conclude.
Conflicting information from different studies
But the current findings seem to directly contradict those from another recent study by Messerli and colleagues showing higher sodium intake correlates with improved life expectancy.
Addressing these contradictory results, Dr. Qi commented: “The study of Messerli et al. is based on an ecological design, in which the analysis is performed on country average sodium intake, rather than at the individual level. This type of ecological study has several major limitations, such as the lack of individuals’ sodium intake, uncontrolled confounding, and the cross-sectional nature. Typically, ecological studies are not considered useful for testing hypothesis in epidemiological studies.”
Dr. Qi noted that, in contrast, his current study analyzes individuals’ exposure, and has a prospective design. “Our findings are supported by previous large-scale observational studies and clinical trials which show the high intake of sodium may adversely affect chronic diseases such as cardiovascular disease and hypertension.” =
Lead author of the ecological study, Franz Messerli, MD, Bern (Switzerland) University Hospital, however, was not convinced by the findings from Dr. Qi’s study.
“The difference in 24-hour sodium intake between those who never/rarely added salt and those who always did is a minuscule 0.17 g. It is highly unlikely that such negligible quantity has any impact on blood pressure, not to mention cardiovascular mortality or life expectancy,” he commented in an interview.
He also pointed out that, in Dr. Qi’s study, people who added salt more frequently also consumed more red meat and processed meat, as well as less fish and less fruit and vegetables. “I would suggest that the bad habit of adding salt at the table is simply a powerful marker for an unhealthy diet.”
“There is no question that an excessive salt intake is harmful in hypertensive patients and increases the risk of stroke. But 0.17 g is not going to make any difference,” Dr. Messerli added.
What is the optimum level?
In an editorial accompanying the study by Dr. Qi and colleagues in the European Heart Journal, Annika Rosengren, MD, PhD, Sahlgrenska University Hospital, Gothenburg, Sweden, noted that guidelines recommend a salt intake below 5 g, or about a teaspoon, per day. But few individuals meet this recommendation.
Because several recent studies show a U- or J-shaped association between salt and atherosclerotic cardiovascular disease, reducing salt intake across the whole population may not be universally beneficial, Dr. Rosengren said.
“So far, what the collective evidence about salt seems to indicate is that healthy people consuming what constitutes normal levels of ordinary salt need not worry too much about their salt intake,” she wrote.
Instead, she advised a diet rich in fruit and vegetables should be a priority to counterbalance potentially harmful effects of salt, and for many other reasons.
And she added that people at high risk, such as those with hypertension who have a high salt intake, are probably well advised to cut down, and not adding extra salt to already prepared foods is one way of achieving this. However, at the individual level, the optimal salt consumption range, or the “sweet spot” remains to be determined.
“Not adding extra salt to food is unlikely to be harmful and could contribute to strategies to lower population blood pressure levels,” Dr. Rosengren concluded.
A version of this article first appeared on Medscape.com.
in a new study.
In the study of more than 500,000 people, compared with those who never or rarely added salt, those who always added salt to their food had a 28% increased risk of dying prematurely (defined as death before the age of 75 years).
Results also showed that adding salt to food was linked to a lower life expectancy. At the age of 50 years, life expectancy was reduced by 1.5 years in women and by 2.28 years in men who always added salt to their food, compared with those who never or rarely did.
However, these increased risks appeared to be attenuated with increasing intakes of high-potassium foods (vegetables and fruits).
The study was published online in the European Heart Journal.
“As far as we are aware, this is the first study to analyze adding salt to meals as a unique measurement for dietary sodium intake. Such a measure is less likely affected by other dietary components, especially potassium intake,” senior author Lu Qi, MD, Tulane University, New Orleans, told this news organization.
“Our study provides supportive evidence from a novel perspective to show the adverse effects of high sodium intake on human health, which is still a controversial topic. Our findings support the advice that reduction of salt intake by reducing the salt added to meals may benefit health and improve life expectancy. Our results also suggest that high intakes of fruits and vegetables are beneficial regarding lowering the adverse effects of salt,” he added.
Link between dietary salt and health is subject of longstanding debate
The researchers explained that the relationship between dietary salt intake and health remains a subject of longstanding debate, with previous studies on the association between sodium intake and mortality having shown conflicting results.
They attributed the inconsistent results to the low accuracy of sodium measurement, noting that sodium intake varies widely from day to day, but the majority of previous studies have largely relied on a single day’s urine collection or dietary survey for estimating the sodium intake, which is inadequate to assess an individual’s usual consumption levels.
They also pointed out that it is difficult to separate the contributions of intakes of sodium and potassium to health based on current methods for measuring dietary sodium and potassium, and this may confound the association between sodium intake and health outcomes.
They noted that the hypothesis that a high-potassium intake may attenuate the adverse association of high-sodium intake with health outcomes has been proposed for many years, but studies assessing the interaction between sodium intake and potassium intake on the risk of mortality are scarce.
Adding salt to food at the table is a common eating behavior directly related to an individual’s long-term preference for salty tasting foods and habitual salt intake, the authors said, adding that commonly used table salt contains 97%-99% sodium chloride, minimizing the potential confounding effects of other dietary factors including potassium. “Therefore, adding salt to foods provides a unique assessment to evaluate the association between habitual sodium intake and mortality.”
UK Biobank study
For the current study Dr. Qi and colleagues analyzed data from 501,379 people taking part in the UK Biobank study. When joining the study between 2006 and 2010, the participants were asked whether they added salt to their foods never/rarely, sometimes, usually or always. Participants were then followed for a median of 9 years.
After adjustment for sex, age, race, smoking, moderate drinking, body mass index, physical activity, Townsend deprivation index, high cholesterol, chronic kidney disease, diabetes, cardiovascular disease, and cancer, results showed an increasing risk of all-cause premature mortality rose with increasing frequency of adding salt to foods.
The adjusted hazard ratios, compared with those who never or rarely added salt, were 1.02 (95% CI, 0.99-1.06) for those who added salt sometimes, 1.07 (95% CI, 1.02-1.11) for those who usually added salt, and 1.28 (95% CI, 1.20-1.35) for those who always added salt.
The researchers also estimated the lower survival time caused by the high frequency of adding salt to foods. At age 50, women who always added salt to foods had an average 1.50 fewer years of life expectancy, and men who always added salt had an average 2.28 fewer years of life expectancy, as compared with their counterparts who never/rarely added salt to foods.
For cause-specific premature mortality, results showed that higher frequency of adding salt to foods was significantly associated with a higher risk of cardiovascular mortality and cancer mortality, but not for dementia mortality or respiratory mortality. For the subtypes of cardiovascular mortality, adding salt to foods was significantly associated with higher risk of stroke mortality but not coronary heart disease mortality.
Other analyses suggested that the association of adding salt to foods with an increased risk of premature mortality appeared to be attenuated with increasing intake of food high in potassium (fruits and vegetables).
The authors point out that the amounts of discretionary sodium intake (the salt used at the table or in home cooking) have been largely overlooked in previous studies, even though adding salt to foods accounts for a considerable proportion of total sodium intake (6%-20%) in Western diets.
“Our findings also support the notion that even a modest reduction in sodium intake is likely to result in substantial health benefits, especially when it is achieved in the general population,” they conclude.
Conflicting information from different studies
But the current findings seem to directly contradict those from another recent study by Messerli and colleagues showing higher sodium intake correlates with improved life expectancy.
Addressing these contradictory results, Dr. Qi commented: “The study of Messerli et al. is based on an ecological design, in which the analysis is performed on country average sodium intake, rather than at the individual level. This type of ecological study has several major limitations, such as the lack of individuals’ sodium intake, uncontrolled confounding, and the cross-sectional nature. Typically, ecological studies are not considered useful for testing hypothesis in epidemiological studies.”
Dr. Qi noted that, in contrast, his current study analyzes individuals’ exposure, and has a prospective design. “Our findings are supported by previous large-scale observational studies and clinical trials which show the high intake of sodium may adversely affect chronic diseases such as cardiovascular disease and hypertension.” =
Lead author of the ecological study, Franz Messerli, MD, Bern (Switzerland) University Hospital, however, was not convinced by the findings from Dr. Qi’s study.
“The difference in 24-hour sodium intake between those who never/rarely added salt and those who always did is a minuscule 0.17 g. It is highly unlikely that such negligible quantity has any impact on blood pressure, not to mention cardiovascular mortality or life expectancy,” he commented in an interview.
He also pointed out that, in Dr. Qi’s study, people who added salt more frequently also consumed more red meat and processed meat, as well as less fish and less fruit and vegetables. “I would suggest that the bad habit of adding salt at the table is simply a powerful marker for an unhealthy diet.”
“There is no question that an excessive salt intake is harmful in hypertensive patients and increases the risk of stroke. But 0.17 g is not going to make any difference,” Dr. Messerli added.
What is the optimum level?
In an editorial accompanying the study by Dr. Qi and colleagues in the European Heart Journal, Annika Rosengren, MD, PhD, Sahlgrenska University Hospital, Gothenburg, Sweden, noted that guidelines recommend a salt intake below 5 g, or about a teaspoon, per day. But few individuals meet this recommendation.
Because several recent studies show a U- or J-shaped association between salt and atherosclerotic cardiovascular disease, reducing salt intake across the whole population may not be universally beneficial, Dr. Rosengren said.
“So far, what the collective evidence about salt seems to indicate is that healthy people consuming what constitutes normal levels of ordinary salt need not worry too much about their salt intake,” she wrote.
Instead, she advised a diet rich in fruit and vegetables should be a priority to counterbalance potentially harmful effects of salt, and for many other reasons.
And she added that people at high risk, such as those with hypertension who have a high salt intake, are probably well advised to cut down, and not adding extra salt to already prepared foods is one way of achieving this. However, at the individual level, the optimal salt consumption range, or the “sweet spot” remains to be determined.
“Not adding extra salt to food is unlikely to be harmful and could contribute to strategies to lower population blood pressure levels,” Dr. Rosengren concluded.
A version of this article first appeared on Medscape.com.
in a new study.
In the study of more than 500,000 people, compared with those who never or rarely added salt, those who always added salt to their food had a 28% increased risk of dying prematurely (defined as death before the age of 75 years).
Results also showed that adding salt to food was linked to a lower life expectancy. At the age of 50 years, life expectancy was reduced by 1.5 years in women and by 2.28 years in men who always added salt to their food, compared with those who never or rarely did.
However, these increased risks appeared to be attenuated with increasing intakes of high-potassium foods (vegetables and fruits).
The study was published online in the European Heart Journal.
“As far as we are aware, this is the first study to analyze adding salt to meals as a unique measurement for dietary sodium intake. Such a measure is less likely affected by other dietary components, especially potassium intake,” senior author Lu Qi, MD, Tulane University, New Orleans, told this news organization.
“Our study provides supportive evidence from a novel perspective to show the adverse effects of high sodium intake on human health, which is still a controversial topic. Our findings support the advice that reduction of salt intake by reducing the salt added to meals may benefit health and improve life expectancy. Our results also suggest that high intakes of fruits and vegetables are beneficial regarding lowering the adverse effects of salt,” he added.
Link between dietary salt and health is subject of longstanding debate
The researchers explained that the relationship between dietary salt intake and health remains a subject of longstanding debate, with previous studies on the association between sodium intake and mortality having shown conflicting results.
They attributed the inconsistent results to the low accuracy of sodium measurement, noting that sodium intake varies widely from day to day, but the majority of previous studies have largely relied on a single day’s urine collection or dietary survey for estimating the sodium intake, which is inadequate to assess an individual’s usual consumption levels.
They also pointed out that it is difficult to separate the contributions of intakes of sodium and potassium to health based on current methods for measuring dietary sodium and potassium, and this may confound the association between sodium intake and health outcomes.
They noted that the hypothesis that a high-potassium intake may attenuate the adverse association of high-sodium intake with health outcomes has been proposed for many years, but studies assessing the interaction between sodium intake and potassium intake on the risk of mortality are scarce.
Adding salt to food at the table is a common eating behavior directly related to an individual’s long-term preference for salty tasting foods and habitual salt intake, the authors said, adding that commonly used table salt contains 97%-99% sodium chloride, minimizing the potential confounding effects of other dietary factors including potassium. “Therefore, adding salt to foods provides a unique assessment to evaluate the association between habitual sodium intake and mortality.”
UK Biobank study
For the current study Dr. Qi and colleagues analyzed data from 501,379 people taking part in the UK Biobank study. When joining the study between 2006 and 2010, the participants were asked whether they added salt to their foods never/rarely, sometimes, usually or always. Participants were then followed for a median of 9 years.
After adjustment for sex, age, race, smoking, moderate drinking, body mass index, physical activity, Townsend deprivation index, high cholesterol, chronic kidney disease, diabetes, cardiovascular disease, and cancer, results showed an increasing risk of all-cause premature mortality rose with increasing frequency of adding salt to foods.
The adjusted hazard ratios, compared with those who never or rarely added salt, were 1.02 (95% CI, 0.99-1.06) for those who added salt sometimes, 1.07 (95% CI, 1.02-1.11) for those who usually added salt, and 1.28 (95% CI, 1.20-1.35) for those who always added salt.
The researchers also estimated the lower survival time caused by the high frequency of adding salt to foods. At age 50, women who always added salt to foods had an average 1.50 fewer years of life expectancy, and men who always added salt had an average 2.28 fewer years of life expectancy, as compared with their counterparts who never/rarely added salt to foods.
For cause-specific premature mortality, results showed that higher frequency of adding salt to foods was significantly associated with a higher risk of cardiovascular mortality and cancer mortality, but not for dementia mortality or respiratory mortality. For the subtypes of cardiovascular mortality, adding salt to foods was significantly associated with higher risk of stroke mortality but not coronary heart disease mortality.
Other analyses suggested that the association of adding salt to foods with an increased risk of premature mortality appeared to be attenuated with increasing intake of food high in potassium (fruits and vegetables).
The authors point out that the amounts of discretionary sodium intake (the salt used at the table or in home cooking) have been largely overlooked in previous studies, even though adding salt to foods accounts for a considerable proportion of total sodium intake (6%-20%) in Western diets.
“Our findings also support the notion that even a modest reduction in sodium intake is likely to result in substantial health benefits, especially when it is achieved in the general population,” they conclude.
Conflicting information from different studies
But the current findings seem to directly contradict those from another recent study by Messerli and colleagues showing higher sodium intake correlates with improved life expectancy.
Addressing these contradictory results, Dr. Qi commented: “The study of Messerli et al. is based on an ecological design, in which the analysis is performed on country average sodium intake, rather than at the individual level. This type of ecological study has several major limitations, such as the lack of individuals’ sodium intake, uncontrolled confounding, and the cross-sectional nature. Typically, ecological studies are not considered useful for testing hypothesis in epidemiological studies.”
Dr. Qi noted that, in contrast, his current study analyzes individuals’ exposure, and has a prospective design. “Our findings are supported by previous large-scale observational studies and clinical trials which show the high intake of sodium may adversely affect chronic diseases such as cardiovascular disease and hypertension.” =
Lead author of the ecological study, Franz Messerli, MD, Bern (Switzerland) University Hospital, however, was not convinced by the findings from Dr. Qi’s study.
“The difference in 24-hour sodium intake between those who never/rarely added salt and those who always did is a minuscule 0.17 g. It is highly unlikely that such negligible quantity has any impact on blood pressure, not to mention cardiovascular mortality or life expectancy,” he commented in an interview.
He also pointed out that, in Dr. Qi’s study, people who added salt more frequently also consumed more red meat and processed meat, as well as less fish and less fruit and vegetables. “I would suggest that the bad habit of adding salt at the table is simply a powerful marker for an unhealthy diet.”
“There is no question that an excessive salt intake is harmful in hypertensive patients and increases the risk of stroke. But 0.17 g is not going to make any difference,” Dr. Messerli added.
What is the optimum level?
In an editorial accompanying the study by Dr. Qi and colleagues in the European Heart Journal, Annika Rosengren, MD, PhD, Sahlgrenska University Hospital, Gothenburg, Sweden, noted that guidelines recommend a salt intake below 5 g, or about a teaspoon, per day. But few individuals meet this recommendation.
Because several recent studies show a U- or J-shaped association between salt and atherosclerotic cardiovascular disease, reducing salt intake across the whole population may not be universally beneficial, Dr. Rosengren said.
“So far, what the collective evidence about salt seems to indicate is that healthy people consuming what constitutes normal levels of ordinary salt need not worry too much about their salt intake,” she wrote.
Instead, she advised a diet rich in fruit and vegetables should be a priority to counterbalance potentially harmful effects of salt, and for many other reasons.
And she added that people at high risk, such as those with hypertension who have a high salt intake, are probably well advised to cut down, and not adding extra salt to already prepared foods is one way of achieving this. However, at the individual level, the optimal salt consumption range, or the “sweet spot” remains to be determined.
“Not adding extra salt to food is unlikely to be harmful and could contribute to strategies to lower population blood pressure levels,” Dr. Rosengren concluded.
A version of this article first appeared on Medscape.com.
FROM THE EUROPEAN HEART JOURNAL
Doc releases song after racist massacre in Buffalo
Physician-musician Cleveland Francis, MD, responded to the recent mass shooting in Buffalo, New York, which left 10 dead, in the only way he knew how. He wrote and recorded a song to honor the victims as “a plea to the other side to recognize us as people,” the Black cardiologist told this news organization.
He couldn’t sleep after the shooting, and “this song was just in my head.” In the 1990s, Dr. Francis took a 3-year sabbatical from medicine to perform and tour as a country singer. He leveraged his Nashville connections to get “Buffalo” produced and recorded.
Acclaimed artist James Threalkill created the accompanying art, titled “The Heavenly Escort of the Buffalo 10,” after listening to a scratch demo.
Dr. Francis doesn’t want people to overlook the massacre as just another gun violence incident because this was “overt hate-crime racism,” he said.
According to the affidavit submitted by FBI agent Christopher J. Dlugokinski, the suspect’s “motive for the mass shooting was to prevent Black people from replacing White people and eliminating the White race, and to inspire others to commit similar attacks.”
Dr. Francis views the Buffalo shooting as distinct from cases like the murder of George Floyd that involved crime or police. It immediately made him think of the Mother Emanuel Church shooting in Charleston, South Carolina. “Having a black skin is now a death warrant,” he said.
The song is also an appeal for White people to fight racism. Dr. Francis is concerned about young men caught up in white supremacy and suggests that we be more alert to children or grandchildren who disconnect from their families, spend time on the dark web, and access guns. The lyrics deliberately don’t mention guns because Dr. Francis wanted to stay out of that debate. “I just sang: ‘What else do I have to do to prove to you that I’m human too?’ ”
Despite his country credentials, Dr. Francis wrote “Buffalo” as a Gospel song because that genre “connects with Black people more and because that civil rights movement was through the church with Dr. Martin Luther King,” he explained. Although he sings all styles of music, the song is performed by Nashville-based singer Michael Lusk so that it’s not a “Cleve Francis thing,” he said, referring to his stage name.
Songwriter Norman Kerner collaborated on the song. The music was produced and recorded by David Thein and mixed by Bob Bullock of Nashville, who Dr. Francis had worked with when he was an artist on Capitol Records.
They sent the video and artwork to the Mayor of Buffalo, Byron Brown, but have yet to hear back. Dr. Francis hopes it could be part of their healing, noting that some people used the song in their Juneteenth celebrations.
The Louisiana native grew up during segregation and was one of two Black students in the Medical College of Virginia class of 1973. After completing his cardiology fellowship, no one would hire him, so Dr. Francis set up his own practice in Northern Virginia. He now works at Inova Heart and Vascular Institute in Alexandria, Va. He remains optimistic about race relations in America and would love a Black pop or Gospel star to record “Buffalo” and bring it to a wider audience.
Dr. Francis is a regular blogger for Medscape. His contribution to country music is recognized in the National Museum of African American History and Culture in Washington, DC. You can find more of his music on YouTube.
A version of this article first appeared on Medscape.com.
Physician-musician Cleveland Francis, MD, responded to the recent mass shooting in Buffalo, New York, which left 10 dead, in the only way he knew how. He wrote and recorded a song to honor the victims as “a plea to the other side to recognize us as people,” the Black cardiologist told this news organization.
He couldn’t sleep after the shooting, and “this song was just in my head.” In the 1990s, Dr. Francis took a 3-year sabbatical from medicine to perform and tour as a country singer. He leveraged his Nashville connections to get “Buffalo” produced and recorded.
Acclaimed artist James Threalkill created the accompanying art, titled “The Heavenly Escort of the Buffalo 10,” after listening to a scratch demo.
Dr. Francis doesn’t want people to overlook the massacre as just another gun violence incident because this was “overt hate-crime racism,” he said.
According to the affidavit submitted by FBI agent Christopher J. Dlugokinski, the suspect’s “motive for the mass shooting was to prevent Black people from replacing White people and eliminating the White race, and to inspire others to commit similar attacks.”
Dr. Francis views the Buffalo shooting as distinct from cases like the murder of George Floyd that involved crime or police. It immediately made him think of the Mother Emanuel Church shooting in Charleston, South Carolina. “Having a black skin is now a death warrant,” he said.
The song is also an appeal for White people to fight racism. Dr. Francis is concerned about young men caught up in white supremacy and suggests that we be more alert to children or grandchildren who disconnect from their families, spend time on the dark web, and access guns. The lyrics deliberately don’t mention guns because Dr. Francis wanted to stay out of that debate. “I just sang: ‘What else do I have to do to prove to you that I’m human too?’ ”
Despite his country credentials, Dr. Francis wrote “Buffalo” as a Gospel song because that genre “connects with Black people more and because that civil rights movement was through the church with Dr. Martin Luther King,” he explained. Although he sings all styles of music, the song is performed by Nashville-based singer Michael Lusk so that it’s not a “Cleve Francis thing,” he said, referring to his stage name.
Songwriter Norman Kerner collaborated on the song. The music was produced and recorded by David Thein and mixed by Bob Bullock of Nashville, who Dr. Francis had worked with when he was an artist on Capitol Records.
They sent the video and artwork to the Mayor of Buffalo, Byron Brown, but have yet to hear back. Dr. Francis hopes it could be part of their healing, noting that some people used the song in their Juneteenth celebrations.
The Louisiana native grew up during segregation and was one of two Black students in the Medical College of Virginia class of 1973. After completing his cardiology fellowship, no one would hire him, so Dr. Francis set up his own practice in Northern Virginia. He now works at Inova Heart and Vascular Institute in Alexandria, Va. He remains optimistic about race relations in America and would love a Black pop or Gospel star to record “Buffalo” and bring it to a wider audience.
Dr. Francis is a regular blogger for Medscape. His contribution to country music is recognized in the National Museum of African American History and Culture in Washington, DC. You can find more of his music on YouTube.
A version of this article first appeared on Medscape.com.
Physician-musician Cleveland Francis, MD, responded to the recent mass shooting in Buffalo, New York, which left 10 dead, in the only way he knew how. He wrote and recorded a song to honor the victims as “a plea to the other side to recognize us as people,” the Black cardiologist told this news organization.
He couldn’t sleep after the shooting, and “this song was just in my head.” In the 1990s, Dr. Francis took a 3-year sabbatical from medicine to perform and tour as a country singer. He leveraged his Nashville connections to get “Buffalo” produced and recorded.
Acclaimed artist James Threalkill created the accompanying art, titled “The Heavenly Escort of the Buffalo 10,” after listening to a scratch demo.
Dr. Francis doesn’t want people to overlook the massacre as just another gun violence incident because this was “overt hate-crime racism,” he said.
According to the affidavit submitted by FBI agent Christopher J. Dlugokinski, the suspect’s “motive for the mass shooting was to prevent Black people from replacing White people and eliminating the White race, and to inspire others to commit similar attacks.”
Dr. Francis views the Buffalo shooting as distinct from cases like the murder of George Floyd that involved crime or police. It immediately made him think of the Mother Emanuel Church shooting in Charleston, South Carolina. “Having a black skin is now a death warrant,” he said.
The song is also an appeal for White people to fight racism. Dr. Francis is concerned about young men caught up in white supremacy and suggests that we be more alert to children or grandchildren who disconnect from their families, spend time on the dark web, and access guns. The lyrics deliberately don’t mention guns because Dr. Francis wanted to stay out of that debate. “I just sang: ‘What else do I have to do to prove to you that I’m human too?’ ”
Despite his country credentials, Dr. Francis wrote “Buffalo” as a Gospel song because that genre “connects with Black people more and because that civil rights movement was through the church with Dr. Martin Luther King,” he explained. Although he sings all styles of music, the song is performed by Nashville-based singer Michael Lusk so that it’s not a “Cleve Francis thing,” he said, referring to his stage name.
Songwriter Norman Kerner collaborated on the song. The music was produced and recorded by David Thein and mixed by Bob Bullock of Nashville, who Dr. Francis had worked with when he was an artist on Capitol Records.
They sent the video and artwork to the Mayor of Buffalo, Byron Brown, but have yet to hear back. Dr. Francis hopes it could be part of their healing, noting that some people used the song in their Juneteenth celebrations.
The Louisiana native grew up during segregation and was one of two Black students in the Medical College of Virginia class of 1973. After completing his cardiology fellowship, no one would hire him, so Dr. Francis set up his own practice in Northern Virginia. He now works at Inova Heart and Vascular Institute in Alexandria, Va. He remains optimistic about race relations in America and would love a Black pop or Gospel star to record “Buffalo” and bring it to a wider audience.
Dr. Francis is a regular blogger for Medscape. His contribution to country music is recognized in the National Museum of African American History and Culture in Washington, DC. You can find more of his music on YouTube.
A version of this article first appeared on Medscape.com.
New AHA checklist: Only one in five adults has optimal heart health
About 80% of American adults have low to moderate cardiovascular (CV) health based on the American Heart Association checklist for optimal heart health, which now includes healthy sleep as an essential component for heart health.
With the addition of sleep, “Life’s Essential 8” replaces the AHA’s “Life’s Simple 7” checklist.
“The new metric of sleep duration reflects the latest research findings: Sleep impacts overall health, and people who have healthier sleep patterns manage health factors such as weight, blood pressure, or risk for type 2 diabetes more effectively,” AHA President Donald M. Lloyd-Jones, MD, said in a news release.
“In addition, advances in ways to measure sleep, such as with wearable devices, now offer people the ability to reliably and routinely monitor their sleep habits at home,” said Dr. Lloyd-Jones, chair of the department of preventive medicine at Northwestern University in Chicago.
The AHA Presidential Advisory – Life’s Essential 8: Updating and Enhancing the American Heart Association’s Construct on Cardiovascular Health – was published online in the journal Circulation.
A companion paper published simultaneously in Circulation reports the first study using Life’s Essential 8.
Overall, the results show that CV health of the U.S. population is “suboptimal, and we see important differences across age and sociodemographic groups,” Dr. Lloyd-Jones said.
Refining Life’s Simple 7
The AHA first defined the seven metrics for optimal CV health in 2010. After 12 years and more than 2,400 scientific papers on the topic, new discoveries in CV health and ways to measure it provided an opportunity to revisit each health component in more detail and provide updates as needed, the AHA explains.
“We felt it was the right time to conduct a comprehensive review of the latest research to refine the existing metrics and consider any new metrics that add value to assessing cardiovascular health for all people,” Dr. Lloyd-Jones said.
Four of the original metrics have been redefined for consistency with newer clinical guidelines or compatibility with new measurement tools, and the scoring system can now also be applied to anyone ages 2 and older. Here is a snapshot of Life’s Essential 8 metrics, including updates.
1. Diet (updated)
The tool includes a new guide to assess diet quality for adults and children at the individual and population level. At the population level, dietary assessment is based on daily intake of elements in the Dietary Approaches to Stop Hypertension (DASH) eating pattern. For individuals, the Mediterranean Eating Pattern for Americans (MEPA) is used to assess and monitor cardiovascular health.
2. Physical activity (no changes)
Physical activity continues to be measured by the total number of minutes of moderate or vigorous physical activity per week, as defined by the U.S. Physical Activity Guidelines for Americans (2nd edition). The optimal level is 150 minutes (2.5 hours) of moderate physical activity or more per week or 75 minutes per week of vigorous-intensity physical activity for adults; 420 minutes (7 hours) or more per week for children ages 6 and older; and age-specific modifications for younger children.
3. Nicotine exposure (updated)
Use of inhaled nicotine-delivery systems, which includes e-cigarettes or vaping devices, has been added since the previous metric monitored only traditional, combustible cigarettes. This reflects use by adults and youth and their implications on long-term health. Second-hand smoke exposure for children and adults has also been added.
4. Sleep duration (new)
Sleep duration is associated with CV health. Measured by average hours of sleep per night, the ideal level is 7-9 hours daily for adults. Ideal daily sleep ranges for children are 10-16 hours per 24 hours for ages 5 and younger; 9-12 hours for ages 6-12 years; and 8-10 hours for ages 13-18 years.
5. Body mass index (no changes)
The AHA acknowledges that body mass index (BMI) is an imperfect metric. Yet, because it’s easily calculated and widely available, BMI continues as a “reasonable” gauge to assess weight categories that may lead to health problems. BMI of 18.5-24.9 is associated with the highest levels of CV health. The AHA notes that BMI ranges and the subsequent health risks associated with them may differ among people from diverse racial or ethnic backgrounds or ancestry. This aligns with the World Health Organization recommendations to adjust BMI ranges for people of Asian or Pacific Islander ancestry because recent evidence indicates their risk of conditions such as CVD or type 2 diabetes is higher at a lower BMI.
6. Blood lipids (updated)
The metric for blood lipids (cholesterol and triglycerides) is updated to use non-HDL cholesterol as the preferred number to monitor, rather than total cholesterol. This shift is made because non-HDL cholesterol can be measured without fasting beforehand (thereby increasing its availability at any time of day and implementation at more appointments) and reliably calculated among all people.
7. Blood glucose (updated)
This metric is expanded to include the option of hemoglobin A1c readings or blood glucose levels for people with or without type 1 or 2 diabetes or prediabetes.
8. Blood pressure (no changes)
Blood pressure criteria remain unchanged from 2017 guidance that established levels less than 120/80 mm Hg as optimal, and defined hypertension as 130-139 mm Hg systolic pressure or 80-89 mm Hg diastolic pressure.
‘Concerning’ new data
Results of the first study using Life’s Essential 8 show that the overall CV health of the U.S. population is “well below ideal,” with 80% of adults scoring at a low or moderate level, the researchers report.
Data for the analysis came from 2013-2018 U.S. National Health and Nutrition Examination surveys (NHANES) of more than 13,500 adults aged 20-79 years and nearly 9,900 children aged 2-19 years. Among the key findings:
- The average CV health score based on Life’s Essential 8 was 64.7 for adults and 65.5 for children – in the moderate range on the 0-100 scale.
- Only 0.45% of adults had a perfect score of 100; 20% had high CV health (score of 80 or higher), 63% moderate (score of 50-79), and 18% had low CV health (score of less than 50).
- Adult women had higher average CV health scores (67) compared with men (62.5).
- In general, adults scored lowest in the areas of diet, physical activity, and BMI.
- CV health scores were generally lower at older ages.
- Non-Hispanic Asian Americans had a higher average CV health score than other racial/ethnic groups. Non-Hispanic Whites had the second highest average CV health score, followed, in order, by Hispanic (other than Mexican), Mexican, and non-Hispanic Blacks.
- Children’s diet scores were low, at an average of 40.6.
- Adult sociodemographic groups varied notably in CV health scores for diet, nicotine exposure, blood glucose, and blood pressure.
“These data represent the first look at the cardiovascular health of the U.S. population using the AHA’s new Life’s Essential 8 scoring algorithm,” Dr. Lloyd-Jones said.
“Life’s Essential 8 is a major step forward in our ability to identify when cardiovascular health can be preserved and when it is suboptimal. It should energize efforts to improve cardiovascular health for all people and at every life stage,” Dr. Lloyd-Jones added.
“Analyses like this can help policymakers, communities, clinicians, and the public to understand the opportunities to intervene to improve and maintain optimal cardiovascular health across the life course,” he said.
This research had no commercial funding. The authors have no reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
About 80% of American adults have low to moderate cardiovascular (CV) health based on the American Heart Association checklist for optimal heart health, which now includes healthy sleep as an essential component for heart health.
With the addition of sleep, “Life’s Essential 8” replaces the AHA’s “Life’s Simple 7” checklist.
“The new metric of sleep duration reflects the latest research findings: Sleep impacts overall health, and people who have healthier sleep patterns manage health factors such as weight, blood pressure, or risk for type 2 diabetes more effectively,” AHA President Donald M. Lloyd-Jones, MD, said in a news release.
“In addition, advances in ways to measure sleep, such as with wearable devices, now offer people the ability to reliably and routinely monitor their sleep habits at home,” said Dr. Lloyd-Jones, chair of the department of preventive medicine at Northwestern University in Chicago.
The AHA Presidential Advisory – Life’s Essential 8: Updating and Enhancing the American Heart Association’s Construct on Cardiovascular Health – was published online in the journal Circulation.
A companion paper published simultaneously in Circulation reports the first study using Life’s Essential 8.
Overall, the results show that CV health of the U.S. population is “suboptimal, and we see important differences across age and sociodemographic groups,” Dr. Lloyd-Jones said.
Refining Life’s Simple 7
The AHA first defined the seven metrics for optimal CV health in 2010. After 12 years and more than 2,400 scientific papers on the topic, new discoveries in CV health and ways to measure it provided an opportunity to revisit each health component in more detail and provide updates as needed, the AHA explains.
“We felt it was the right time to conduct a comprehensive review of the latest research to refine the existing metrics and consider any new metrics that add value to assessing cardiovascular health for all people,” Dr. Lloyd-Jones said.
Four of the original metrics have been redefined for consistency with newer clinical guidelines or compatibility with new measurement tools, and the scoring system can now also be applied to anyone ages 2 and older. Here is a snapshot of Life’s Essential 8 metrics, including updates.
1. Diet (updated)
The tool includes a new guide to assess diet quality for adults and children at the individual and population level. At the population level, dietary assessment is based on daily intake of elements in the Dietary Approaches to Stop Hypertension (DASH) eating pattern. For individuals, the Mediterranean Eating Pattern for Americans (MEPA) is used to assess and monitor cardiovascular health.
2. Physical activity (no changes)
Physical activity continues to be measured by the total number of minutes of moderate or vigorous physical activity per week, as defined by the U.S. Physical Activity Guidelines for Americans (2nd edition). The optimal level is 150 minutes (2.5 hours) of moderate physical activity or more per week or 75 minutes per week of vigorous-intensity physical activity for adults; 420 minutes (7 hours) or more per week for children ages 6 and older; and age-specific modifications for younger children.
3. Nicotine exposure (updated)
Use of inhaled nicotine-delivery systems, which includes e-cigarettes or vaping devices, has been added since the previous metric monitored only traditional, combustible cigarettes. This reflects use by adults and youth and their implications on long-term health. Second-hand smoke exposure for children and adults has also been added.
4. Sleep duration (new)
Sleep duration is associated with CV health. Measured by average hours of sleep per night, the ideal level is 7-9 hours daily for adults. Ideal daily sleep ranges for children are 10-16 hours per 24 hours for ages 5 and younger; 9-12 hours for ages 6-12 years; and 8-10 hours for ages 13-18 years.
5. Body mass index (no changes)
The AHA acknowledges that body mass index (BMI) is an imperfect metric. Yet, because it’s easily calculated and widely available, BMI continues as a “reasonable” gauge to assess weight categories that may lead to health problems. BMI of 18.5-24.9 is associated with the highest levels of CV health. The AHA notes that BMI ranges and the subsequent health risks associated with them may differ among people from diverse racial or ethnic backgrounds or ancestry. This aligns with the World Health Organization recommendations to adjust BMI ranges for people of Asian or Pacific Islander ancestry because recent evidence indicates their risk of conditions such as CVD or type 2 diabetes is higher at a lower BMI.
6. Blood lipids (updated)
The metric for blood lipids (cholesterol and triglycerides) is updated to use non-HDL cholesterol as the preferred number to monitor, rather than total cholesterol. This shift is made because non-HDL cholesterol can be measured without fasting beforehand (thereby increasing its availability at any time of day and implementation at more appointments) and reliably calculated among all people.
7. Blood glucose (updated)
This metric is expanded to include the option of hemoglobin A1c readings or blood glucose levels for people with or without type 1 or 2 diabetes or prediabetes.
8. Blood pressure (no changes)
Blood pressure criteria remain unchanged from 2017 guidance that established levels less than 120/80 mm Hg as optimal, and defined hypertension as 130-139 mm Hg systolic pressure or 80-89 mm Hg diastolic pressure.
‘Concerning’ new data
Results of the first study using Life’s Essential 8 show that the overall CV health of the U.S. population is “well below ideal,” with 80% of adults scoring at a low or moderate level, the researchers report.
Data for the analysis came from 2013-2018 U.S. National Health and Nutrition Examination surveys (NHANES) of more than 13,500 adults aged 20-79 years and nearly 9,900 children aged 2-19 years. Among the key findings:
- The average CV health score based on Life’s Essential 8 was 64.7 for adults and 65.5 for children – in the moderate range on the 0-100 scale.
- Only 0.45% of adults had a perfect score of 100; 20% had high CV health (score of 80 or higher), 63% moderate (score of 50-79), and 18% had low CV health (score of less than 50).
- Adult women had higher average CV health scores (67) compared with men (62.5).
- In general, adults scored lowest in the areas of diet, physical activity, and BMI.
- CV health scores were generally lower at older ages.
- Non-Hispanic Asian Americans had a higher average CV health score than other racial/ethnic groups. Non-Hispanic Whites had the second highest average CV health score, followed, in order, by Hispanic (other than Mexican), Mexican, and non-Hispanic Blacks.
- Children’s diet scores were low, at an average of 40.6.
- Adult sociodemographic groups varied notably in CV health scores for diet, nicotine exposure, blood glucose, and blood pressure.
“These data represent the first look at the cardiovascular health of the U.S. population using the AHA’s new Life’s Essential 8 scoring algorithm,” Dr. Lloyd-Jones said.
“Life’s Essential 8 is a major step forward in our ability to identify when cardiovascular health can be preserved and when it is suboptimal. It should energize efforts to improve cardiovascular health for all people and at every life stage,” Dr. Lloyd-Jones added.
“Analyses like this can help policymakers, communities, clinicians, and the public to understand the opportunities to intervene to improve and maintain optimal cardiovascular health across the life course,” he said.
This research had no commercial funding. The authors have no reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
About 80% of American adults have low to moderate cardiovascular (CV) health based on the American Heart Association checklist for optimal heart health, which now includes healthy sleep as an essential component for heart health.
With the addition of sleep, “Life’s Essential 8” replaces the AHA’s “Life’s Simple 7” checklist.
“The new metric of sleep duration reflects the latest research findings: Sleep impacts overall health, and people who have healthier sleep patterns manage health factors such as weight, blood pressure, or risk for type 2 diabetes more effectively,” AHA President Donald M. Lloyd-Jones, MD, said in a news release.
“In addition, advances in ways to measure sleep, such as with wearable devices, now offer people the ability to reliably and routinely monitor their sleep habits at home,” said Dr. Lloyd-Jones, chair of the department of preventive medicine at Northwestern University in Chicago.
The AHA Presidential Advisory – Life’s Essential 8: Updating and Enhancing the American Heart Association’s Construct on Cardiovascular Health – was published online in the journal Circulation.
A companion paper published simultaneously in Circulation reports the first study using Life’s Essential 8.
Overall, the results show that CV health of the U.S. population is “suboptimal, and we see important differences across age and sociodemographic groups,” Dr. Lloyd-Jones said.
Refining Life’s Simple 7
The AHA first defined the seven metrics for optimal CV health in 2010. After 12 years and more than 2,400 scientific papers on the topic, new discoveries in CV health and ways to measure it provided an opportunity to revisit each health component in more detail and provide updates as needed, the AHA explains.
“We felt it was the right time to conduct a comprehensive review of the latest research to refine the existing metrics and consider any new metrics that add value to assessing cardiovascular health for all people,” Dr. Lloyd-Jones said.
Four of the original metrics have been redefined for consistency with newer clinical guidelines or compatibility with new measurement tools, and the scoring system can now also be applied to anyone ages 2 and older. Here is a snapshot of Life’s Essential 8 metrics, including updates.
1. Diet (updated)
The tool includes a new guide to assess diet quality for adults and children at the individual and population level. At the population level, dietary assessment is based on daily intake of elements in the Dietary Approaches to Stop Hypertension (DASH) eating pattern. For individuals, the Mediterranean Eating Pattern for Americans (MEPA) is used to assess and monitor cardiovascular health.
2. Physical activity (no changes)
Physical activity continues to be measured by the total number of minutes of moderate or vigorous physical activity per week, as defined by the U.S. Physical Activity Guidelines for Americans (2nd edition). The optimal level is 150 minutes (2.5 hours) of moderate physical activity or more per week or 75 minutes per week of vigorous-intensity physical activity for adults; 420 minutes (7 hours) or more per week for children ages 6 and older; and age-specific modifications for younger children.
3. Nicotine exposure (updated)
Use of inhaled nicotine-delivery systems, which includes e-cigarettes or vaping devices, has been added since the previous metric monitored only traditional, combustible cigarettes. This reflects use by adults and youth and their implications on long-term health. Second-hand smoke exposure for children and adults has also been added.
4. Sleep duration (new)
Sleep duration is associated with CV health. Measured by average hours of sleep per night, the ideal level is 7-9 hours daily for adults. Ideal daily sleep ranges for children are 10-16 hours per 24 hours for ages 5 and younger; 9-12 hours for ages 6-12 years; and 8-10 hours for ages 13-18 years.
5. Body mass index (no changes)
The AHA acknowledges that body mass index (BMI) is an imperfect metric. Yet, because it’s easily calculated and widely available, BMI continues as a “reasonable” gauge to assess weight categories that may lead to health problems. BMI of 18.5-24.9 is associated with the highest levels of CV health. The AHA notes that BMI ranges and the subsequent health risks associated with them may differ among people from diverse racial or ethnic backgrounds or ancestry. This aligns with the World Health Organization recommendations to adjust BMI ranges for people of Asian or Pacific Islander ancestry because recent evidence indicates their risk of conditions such as CVD or type 2 diabetes is higher at a lower BMI.
6. Blood lipids (updated)
The metric for blood lipids (cholesterol and triglycerides) is updated to use non-HDL cholesterol as the preferred number to monitor, rather than total cholesterol. This shift is made because non-HDL cholesterol can be measured without fasting beforehand (thereby increasing its availability at any time of day and implementation at more appointments) and reliably calculated among all people.
7. Blood glucose (updated)
This metric is expanded to include the option of hemoglobin A1c readings or blood glucose levels for people with or without type 1 or 2 diabetes or prediabetes.
8. Blood pressure (no changes)
Blood pressure criteria remain unchanged from 2017 guidance that established levels less than 120/80 mm Hg as optimal, and defined hypertension as 130-139 mm Hg systolic pressure or 80-89 mm Hg diastolic pressure.
‘Concerning’ new data
Results of the first study using Life’s Essential 8 show that the overall CV health of the U.S. population is “well below ideal,” with 80% of adults scoring at a low or moderate level, the researchers report.
Data for the analysis came from 2013-2018 U.S. National Health and Nutrition Examination surveys (NHANES) of more than 13,500 adults aged 20-79 years and nearly 9,900 children aged 2-19 years. Among the key findings:
- The average CV health score based on Life’s Essential 8 was 64.7 for adults and 65.5 for children – in the moderate range on the 0-100 scale.
- Only 0.45% of adults had a perfect score of 100; 20% had high CV health (score of 80 or higher), 63% moderate (score of 50-79), and 18% had low CV health (score of less than 50).
- Adult women had higher average CV health scores (67) compared with men (62.5).
- In general, adults scored lowest in the areas of diet, physical activity, and BMI.
- CV health scores were generally lower at older ages.
- Non-Hispanic Asian Americans had a higher average CV health score than other racial/ethnic groups. Non-Hispanic Whites had the second highest average CV health score, followed, in order, by Hispanic (other than Mexican), Mexican, and non-Hispanic Blacks.
- Children’s diet scores were low, at an average of 40.6.
- Adult sociodemographic groups varied notably in CV health scores for diet, nicotine exposure, blood glucose, and blood pressure.
“These data represent the first look at the cardiovascular health of the U.S. population using the AHA’s new Life’s Essential 8 scoring algorithm,” Dr. Lloyd-Jones said.
“Life’s Essential 8 is a major step forward in our ability to identify when cardiovascular health can be preserved and when it is suboptimal. It should energize efforts to improve cardiovascular health for all people and at every life stage,” Dr. Lloyd-Jones added.
“Analyses like this can help policymakers, communities, clinicians, and the public to understand the opportunities to intervene to improve and maintain optimal cardiovascular health across the life course,” he said.
This research had no commercial funding. The authors have no reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM CIRCULATION
Heart attack care not equal for women and people of color
Radiating chest pain, shortness of breath, nausea, lightheadedness. Everyone knows the telltale signs of a myocardial infarction. Yet a new study shows that despite this widespread recognition, heart attacks aren’t attended to quickly across the board. Historically, the study says, women and people of color wait longer to access emergency care for a heart attack.
Researchers from the University of California, San Francisco published these findings in the Annals of Emergency Medicine. The study used the Office of Statewide Health Planning and Development dataset to gather information on 453,136 cases of heart attack in California between 2005 and 2015. They found that over time, differences in timely treatment between the demographics narrowed, but the gap still existed.
The study defined timely treatment as receiving care for a heart attack within 3 days of admission to a hospital. Women and people of color were found to wait 3 days or more to receive care than their White male counterparts. A disparity of this sort can cause ripples of health effects across society, ripples that doctors should be aware of, says lead author Juan Carlos Montoy, MD. Dr. Montoy was “sadly surprised by our findings that disparities for women and for Black patients only decreased slightly or not at all over time.”
In the study, the team separated the dataset between the two primary types of heart attack: ST-segment elevation myocardial infarction (STEMI), caused by blood vessel blockage, and non–ST-segment elevation myocardial infarction (NSTEMI), caused by a narrowing or temporary blockage of the artery.
Regardless of the type of heart attack, the standard first step in treatment is a coronary angiogram. After finding out where blood flow is disrupted using the angiogram, a physician can proceed with treatment.
But when looking back, the team found that it took a while for many patients to receive this first step in treatment. In 2005, 50% of men and 35.7% of women with STEMI and 45% of men and 33.1% of women with NSTEMI had a timely angiography. In the same year, 46% of White patients and 31.2% of Black patients with STEMI underwent timely angiography.
By 2015, timely treatment increased across the board, but there were still discrepancies, with 76.7% of men and 66.8% of women with STEMI undergoing timely angiography and 56.3% of men and 45.9% of women with NSTEMI undergoing timely angiography. Also in 2015, 75.2% of White patients and 69.2% of Black patients underwent timely angiography for STEMI.
Although differences in care decreased between the demographics, the gap still exists. Whereas this dataset only extends to 2015, this trend may still persist today, says Robert Glatter, MD, an emergency medicine physician at Lenox Hill Hospital, New York, who was not involved in the study. Therefore, physicians need to consider this bias when treating patients. “The bottom line is that we continue to have much work to do to achieve equality in managing not only medical conditions but treating people who have them equally,” Dr. Glatter said.
“Raising awareness of ongoing inequality in care related to gender and ethnic disparities is critical to drive change in our institutions,” he emphasized. “We simply cannot accept the status quo.”
The study was funded by the National Heart, Lung, and Blood Institute of the National Institutes of Health. Dr. Glatter and the authors declared no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Radiating chest pain, shortness of breath, nausea, lightheadedness. Everyone knows the telltale signs of a myocardial infarction. Yet a new study shows that despite this widespread recognition, heart attacks aren’t attended to quickly across the board. Historically, the study says, women and people of color wait longer to access emergency care for a heart attack.
Researchers from the University of California, San Francisco published these findings in the Annals of Emergency Medicine. The study used the Office of Statewide Health Planning and Development dataset to gather information on 453,136 cases of heart attack in California between 2005 and 2015. They found that over time, differences in timely treatment between the demographics narrowed, but the gap still existed.
The study defined timely treatment as receiving care for a heart attack within 3 days of admission to a hospital. Women and people of color were found to wait 3 days or more to receive care than their White male counterparts. A disparity of this sort can cause ripples of health effects across society, ripples that doctors should be aware of, says lead author Juan Carlos Montoy, MD. Dr. Montoy was “sadly surprised by our findings that disparities for women and for Black patients only decreased slightly or not at all over time.”
In the study, the team separated the dataset between the two primary types of heart attack: ST-segment elevation myocardial infarction (STEMI), caused by blood vessel blockage, and non–ST-segment elevation myocardial infarction (NSTEMI), caused by a narrowing or temporary blockage of the artery.
Regardless of the type of heart attack, the standard first step in treatment is a coronary angiogram. After finding out where blood flow is disrupted using the angiogram, a physician can proceed with treatment.
But when looking back, the team found that it took a while for many patients to receive this first step in treatment. In 2005, 50% of men and 35.7% of women with STEMI and 45% of men and 33.1% of women with NSTEMI had a timely angiography. In the same year, 46% of White patients and 31.2% of Black patients with STEMI underwent timely angiography.
By 2015, timely treatment increased across the board, but there were still discrepancies, with 76.7% of men and 66.8% of women with STEMI undergoing timely angiography and 56.3% of men and 45.9% of women with NSTEMI undergoing timely angiography. Also in 2015, 75.2% of White patients and 69.2% of Black patients underwent timely angiography for STEMI.
Although differences in care decreased between the demographics, the gap still exists. Whereas this dataset only extends to 2015, this trend may still persist today, says Robert Glatter, MD, an emergency medicine physician at Lenox Hill Hospital, New York, who was not involved in the study. Therefore, physicians need to consider this bias when treating patients. “The bottom line is that we continue to have much work to do to achieve equality in managing not only medical conditions but treating people who have them equally,” Dr. Glatter said.
“Raising awareness of ongoing inequality in care related to gender and ethnic disparities is critical to drive change in our institutions,” he emphasized. “We simply cannot accept the status quo.”
The study was funded by the National Heart, Lung, and Blood Institute of the National Institutes of Health. Dr. Glatter and the authors declared no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Radiating chest pain, shortness of breath, nausea, lightheadedness. Everyone knows the telltale signs of a myocardial infarction. Yet a new study shows that despite this widespread recognition, heart attacks aren’t attended to quickly across the board. Historically, the study says, women and people of color wait longer to access emergency care for a heart attack.
Researchers from the University of California, San Francisco published these findings in the Annals of Emergency Medicine. The study used the Office of Statewide Health Planning and Development dataset to gather information on 453,136 cases of heart attack in California between 2005 and 2015. They found that over time, differences in timely treatment between the demographics narrowed, but the gap still existed.
The study defined timely treatment as receiving care for a heart attack within 3 days of admission to a hospital. Women and people of color were found to wait 3 days or more to receive care than their White male counterparts. A disparity of this sort can cause ripples of health effects across society, ripples that doctors should be aware of, says lead author Juan Carlos Montoy, MD. Dr. Montoy was “sadly surprised by our findings that disparities for women and for Black patients only decreased slightly or not at all over time.”
In the study, the team separated the dataset between the two primary types of heart attack: ST-segment elevation myocardial infarction (STEMI), caused by blood vessel blockage, and non–ST-segment elevation myocardial infarction (NSTEMI), caused by a narrowing or temporary blockage of the artery.
Regardless of the type of heart attack, the standard first step in treatment is a coronary angiogram. After finding out where blood flow is disrupted using the angiogram, a physician can proceed with treatment.
But when looking back, the team found that it took a while for many patients to receive this first step in treatment. In 2005, 50% of men and 35.7% of women with STEMI and 45% of men and 33.1% of women with NSTEMI had a timely angiography. In the same year, 46% of White patients and 31.2% of Black patients with STEMI underwent timely angiography.
By 2015, timely treatment increased across the board, but there were still discrepancies, with 76.7% of men and 66.8% of women with STEMI undergoing timely angiography and 56.3% of men and 45.9% of women with NSTEMI undergoing timely angiography. Also in 2015, 75.2% of White patients and 69.2% of Black patients underwent timely angiography for STEMI.
Although differences in care decreased between the demographics, the gap still exists. Whereas this dataset only extends to 2015, this trend may still persist today, says Robert Glatter, MD, an emergency medicine physician at Lenox Hill Hospital, New York, who was not involved in the study. Therefore, physicians need to consider this bias when treating patients. “The bottom line is that we continue to have much work to do to achieve equality in managing not only medical conditions but treating people who have them equally,” Dr. Glatter said.
“Raising awareness of ongoing inequality in care related to gender and ethnic disparities is critical to drive change in our institutions,” he emphasized. “We simply cannot accept the status quo.”
The study was funded by the National Heart, Lung, and Blood Institute of the National Institutes of Health. Dr. Glatter and the authors declared no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM ANNALS OF EMERGENCY MEDICINE
New biomarker data add to concerns over REDUCE-IT trial
A new analysis of the REDUCE-IT study has reignited concerns that the benefit shown by the high-dose fish oil product in the study, icosapent ethyl (Vascepa, Amarin), may have been related to harms caused by the placebo mineral oil.
Results show that allocation to icosapent ethyl had minimal effects on a series of biomarkers associated with atherosclerotic disease, whereas levels of these biomarkers increased among those allocated to mineral oil.
At 12 months, the median percent increases from baseline in the mineral oil group were 1.5% for homocysteine, 2.2% for lipoprotein(a), 10.9% for oxidized low-density-lipoprotein (LDL) cholesterol, 16.2% for interleukin (IL)-6, 18.5% for lipoprotein-associated phospholipase A2, 21.9% for high-sensitivity C-reactive protein (hsCRP), and 28.9% for IL-1β. The changes were similar at 24 months. However, in the icosapent ethyl group, there were minimal changes in these biomarkers at 12 and 24 months.
The study was published online in Circulation.
The authors, led by Paul Ridker, MD, Brigham & Women’s Hospital, Boston, do not voice much opinion on what the results mean, concluding that “the effect of these findings on the interpretation of the REDUCE-IT trial results remains unclear and will require further investigation.”
They also say that a second icosapent ethyl trial using a nonmineral oil comparator “would help resolve ongoing controversy.”
However, the authors are a mixed group; Dr. Ridker and some of his coauthors were not part of the original REDUCE-IT trial, whereas other coauthors were members of the REDUCE-IT steering committee, and one was an employee of Amarin.
Lead investigator of the REDUCE-IT trial, Deepak Bhatt, MD, also from Brigham & Women’s Hospital, who is the senior author of the current study, played down the new findings, saying they did not offer much new incremental information on mechanistic insight.
“These are small degrees of biomarker increases on an absolute level we are seeing with the placebo that would not be expected to produce harm,” Dr. Bhatt told this news organization. He also said the Circulation peer-review process had removed some of the discussion, which could lead to some “misinterpretation” of the authors’ views.
Dr. Ridker was unavailable for further comment.
The publication of this study has inevitably poured fuel on the fire regarding the controversy that has long dogged the REDUCE-IT trial, with questions about the large reduction in event rates seen with icosapent ethyl without an obvious mechanistic explanation.
‘Smoking gun’
One of the loudest critics of the study, Steve Nissen, MD, Cleveland Clinic, described the new findings as “the closest thing I’ve seen to a smoking gun in medicine for a long, long time.”
“The result of this new analysis shows that mineral oil increases virtually every inflammatory and lipid marker that they measured,” he commented.
“There are a lot of theories, but the bottom line is that something really bad happened in the mineral-oil group, which makes icosapent ethyl look efficacious. In my view, this needs to be reviewed by the FDA for consideration of removing the label claim for cardiovascular benefit.”
Other experts in the field not directly involved in the study voiced concern about these new findings, adding to calls for another trial.
In a Twitter thread on the issue, Harlan Krumholz, MD, describes the Circulation publication as “an exceptionally important article,” adding that it is “time to rethink this drug.”
“My point is ... once you know you have non-neutral comparator and the effect on risk biomarkers is far from trivial ... then you have introduced substantial uncertainty about the trial result, as conveyed by the authors ... and no one can say what would happen with a neutral comparator,” Dr. Krumholz writes.
In an accompanying editorial in Circulation, Robert Harrington, MD, professor of medicine at Stanford (Calif.) University, concludes that “the hard reality is that we are left with uncertainties and the questions raised by use of the mineral oil as placebo can only be answered by another randomized controlled trial.”
“My main points are that the chosen placebo was not inert (an essential characteristic for an active control–placebo comparison), that the biomarker data show elevations in multiple markers involved with inflammatory pathways associated with atherosclerosis, and that these data create enough uncertainty in the trial interpretation that the best recourse to answer the criticisms is to do another trial with a truly inert placebo,” Dr. Harrington said in an interview.
He added that Dr. Bhatt’s point that the changes in biomarkers are too small to really matter clinically may be right. “But for me, the uncertainties mean that I have less confidence in the true magnitude of the treatment effect than I would have if there were no changes in the inflammatory markers.”
In Circulation, the authors say it is unclear why multiple biomarkers increased over time among REDUCE-IT participants allocated to mineral oil. They note that no substantive changes in these biomarkers were observed in the placebo groups over periods of 3-5 years in other trials, including JUPITER, CIRT, CANTOS, SPIRE, and the STRENGTH trials, which evaluated a different high-dose omega-3 oil product but used corn oil as the placebo.
“The core design of REDUCE-IT does not make it possible to resolve convincingly whether any adverse effects associated with mineral oil use as a comparator may have affected clinical outcomes,” they write.
They point out that regulatory agencies evaluating REDUCE-IT estimated that approximately 3% of the net clinical benefit observed with icosapent ethyl might have been a consequence of adverse biomarker effects on LDL cholesterol and hsCRP attributable to mineral oil. But in the context of an overall 25% relative risk reduction in first events and a 30% reduction in total ischemic events observed, a potential bias of this magnitude, even if doubled in size, would be unlikely to fully attenuate the overall benefit of icosapent ethyl observed.
They add that they are not aware of a method to assess what the potential magnitude might be of a combination of the multiple effects.
New data do not change the debate
“We did a large, well-powered randomized trial, and this paper shouldn’t change anything in how that trial should be interpreted,” Dr. Bhatt said in an interview.
He claims the new biomarkers evaluated in the study are correlated with LDL and CRP, data which have already been reported and analyzed so have limited relevance.
“It’s not really independent biomarker information; this is what we would expect to see when we see small increases in LDL and CRP. So, I don’t think this new information fundamentally changes the debate,” he said.
Dr. Bhatt also pointed out that the study highlights relative increases rather than absolute increases in the biomarkers, making it seem more alarming than is actually the case.
“The paper makes it seem like that there are large increases in these other biomarkers, but the values reported are relative increases and the absolute increases were actually rather small. In many cases, the changes reported are less than the lower limit of quantification of the assay used,” he noted.
He added: “Even if one is unable to get around the placebo issue in the REDUCE-IT trial, there will always be the JELIS trial – a randomized trial with no placebo showing a 19% relative risk reduction. While the biomarker data may be interesting, what really matters in the end is clinical events. And significant reductions in two independent trials should be enough.”
Dr. Bhatt says the REDUCE-IT steering committee does not believe another trial is needed. “Maybe a different population would be good – such as primary prevention, patients without elevated triglycerides – but just repeating REDUCE- IT with a different placebo would be a waste of resources,” he commented.
But Dr. Nissen refuted Dr. Bhatt’s claims.
“These biomarkers are not in the same pathways as LDL and CRP, and these are not small increases. In the CANTOS trial, a monoclonal antibody against interleukin-1β beta showed a significant benefit. The increase in interleukin-1β now reported in REDUCE-IT is exactly the opposite of CANTOS,” he pointed out.
“The FDA did not know about these additional biomarkers when it reviewed the data on LDL and CRP. Now we have new information. It needs to be looked at again,” Dr. Nissen added.
Funding for the study was provided by Amarin Pharma. Dr. Bhatt was the lead investigator of the REDUCE-IT trial. Dr. Nissen was the lead investigator the STRENGTH trial. Further disclosures of the authors can be found in Circulation.
A version of this article first appeared on Medscape.com.
A new analysis of the REDUCE-IT study has reignited concerns that the benefit shown by the high-dose fish oil product in the study, icosapent ethyl (Vascepa, Amarin), may have been related to harms caused by the placebo mineral oil.
Results show that allocation to icosapent ethyl had minimal effects on a series of biomarkers associated with atherosclerotic disease, whereas levels of these biomarkers increased among those allocated to mineral oil.
At 12 months, the median percent increases from baseline in the mineral oil group were 1.5% for homocysteine, 2.2% for lipoprotein(a), 10.9% for oxidized low-density-lipoprotein (LDL) cholesterol, 16.2% for interleukin (IL)-6, 18.5% for lipoprotein-associated phospholipase A2, 21.9% for high-sensitivity C-reactive protein (hsCRP), and 28.9% for IL-1β. The changes were similar at 24 months. However, in the icosapent ethyl group, there were minimal changes in these biomarkers at 12 and 24 months.
The study was published online in Circulation.
The authors, led by Paul Ridker, MD, Brigham & Women’s Hospital, Boston, do not voice much opinion on what the results mean, concluding that “the effect of these findings on the interpretation of the REDUCE-IT trial results remains unclear and will require further investigation.”
They also say that a second icosapent ethyl trial using a nonmineral oil comparator “would help resolve ongoing controversy.”
However, the authors are a mixed group; Dr. Ridker and some of his coauthors were not part of the original REDUCE-IT trial, whereas other coauthors were members of the REDUCE-IT steering committee, and one was an employee of Amarin.
Lead investigator of the REDUCE-IT trial, Deepak Bhatt, MD, also from Brigham & Women’s Hospital, who is the senior author of the current study, played down the new findings, saying they did not offer much new incremental information on mechanistic insight.
“These are small degrees of biomarker increases on an absolute level we are seeing with the placebo that would not be expected to produce harm,” Dr. Bhatt told this news organization. He also said the Circulation peer-review process had removed some of the discussion, which could lead to some “misinterpretation” of the authors’ views.
Dr. Ridker was unavailable for further comment.
The publication of this study has inevitably poured fuel on the fire regarding the controversy that has long dogged the REDUCE-IT trial, with questions about the large reduction in event rates seen with icosapent ethyl without an obvious mechanistic explanation.
‘Smoking gun’
One of the loudest critics of the study, Steve Nissen, MD, Cleveland Clinic, described the new findings as “the closest thing I’ve seen to a smoking gun in medicine for a long, long time.”
“The result of this new analysis shows that mineral oil increases virtually every inflammatory and lipid marker that they measured,” he commented.
“There are a lot of theories, but the bottom line is that something really bad happened in the mineral-oil group, which makes icosapent ethyl look efficacious. In my view, this needs to be reviewed by the FDA for consideration of removing the label claim for cardiovascular benefit.”
Other experts in the field not directly involved in the study voiced concern about these new findings, adding to calls for another trial.
In a Twitter thread on the issue, Harlan Krumholz, MD, describes the Circulation publication as “an exceptionally important article,” adding that it is “time to rethink this drug.”
“My point is ... once you know you have non-neutral comparator and the effect on risk biomarkers is far from trivial ... then you have introduced substantial uncertainty about the trial result, as conveyed by the authors ... and no one can say what would happen with a neutral comparator,” Dr. Krumholz writes.
In an accompanying editorial in Circulation, Robert Harrington, MD, professor of medicine at Stanford (Calif.) University, concludes that “the hard reality is that we are left with uncertainties and the questions raised by use of the mineral oil as placebo can only be answered by another randomized controlled trial.”
“My main points are that the chosen placebo was not inert (an essential characteristic for an active control–placebo comparison), that the biomarker data show elevations in multiple markers involved with inflammatory pathways associated with atherosclerosis, and that these data create enough uncertainty in the trial interpretation that the best recourse to answer the criticisms is to do another trial with a truly inert placebo,” Dr. Harrington said in an interview.
He added that Dr. Bhatt’s point that the changes in biomarkers are too small to really matter clinically may be right. “But for me, the uncertainties mean that I have less confidence in the true magnitude of the treatment effect than I would have if there were no changes in the inflammatory markers.”
In Circulation, the authors say it is unclear why multiple biomarkers increased over time among REDUCE-IT participants allocated to mineral oil. They note that no substantive changes in these biomarkers were observed in the placebo groups over periods of 3-5 years in other trials, including JUPITER, CIRT, CANTOS, SPIRE, and the STRENGTH trials, which evaluated a different high-dose omega-3 oil product but used corn oil as the placebo.
“The core design of REDUCE-IT does not make it possible to resolve convincingly whether any adverse effects associated with mineral oil use as a comparator may have affected clinical outcomes,” they write.
They point out that regulatory agencies evaluating REDUCE-IT estimated that approximately 3% of the net clinical benefit observed with icosapent ethyl might have been a consequence of adverse biomarker effects on LDL cholesterol and hsCRP attributable to mineral oil. But in the context of an overall 25% relative risk reduction in first events and a 30% reduction in total ischemic events observed, a potential bias of this magnitude, even if doubled in size, would be unlikely to fully attenuate the overall benefit of icosapent ethyl observed.
They add that they are not aware of a method to assess what the potential magnitude might be of a combination of the multiple effects.
New data do not change the debate
“We did a large, well-powered randomized trial, and this paper shouldn’t change anything in how that trial should be interpreted,” Dr. Bhatt said in an interview.
He claims the new biomarkers evaluated in the study are correlated with LDL and CRP, data which have already been reported and analyzed so have limited relevance.
“It’s not really independent biomarker information; this is what we would expect to see when we see small increases in LDL and CRP. So, I don’t think this new information fundamentally changes the debate,” he said.
Dr. Bhatt also pointed out that the study highlights relative increases rather than absolute increases in the biomarkers, making it seem more alarming than is actually the case.
“The paper makes it seem like that there are large increases in these other biomarkers, but the values reported are relative increases and the absolute increases were actually rather small. In many cases, the changes reported are less than the lower limit of quantification of the assay used,” he noted.
He added: “Even if one is unable to get around the placebo issue in the REDUCE-IT trial, there will always be the JELIS trial – a randomized trial with no placebo showing a 19% relative risk reduction. While the biomarker data may be interesting, what really matters in the end is clinical events. And significant reductions in two independent trials should be enough.”
Dr. Bhatt says the REDUCE-IT steering committee does not believe another trial is needed. “Maybe a different population would be good – such as primary prevention, patients without elevated triglycerides – but just repeating REDUCE- IT with a different placebo would be a waste of resources,” he commented.
But Dr. Nissen refuted Dr. Bhatt’s claims.
“These biomarkers are not in the same pathways as LDL and CRP, and these are not small increases. In the CANTOS trial, a monoclonal antibody against interleukin-1β beta showed a significant benefit. The increase in interleukin-1β now reported in REDUCE-IT is exactly the opposite of CANTOS,” he pointed out.
“The FDA did not know about these additional biomarkers when it reviewed the data on LDL and CRP. Now we have new information. It needs to be looked at again,” Dr. Nissen added.
Funding for the study was provided by Amarin Pharma. Dr. Bhatt was the lead investigator of the REDUCE-IT trial. Dr. Nissen was the lead investigator the STRENGTH trial. Further disclosures of the authors can be found in Circulation.
A version of this article first appeared on Medscape.com.
A new analysis of the REDUCE-IT study has reignited concerns that the benefit shown by the high-dose fish oil product in the study, icosapent ethyl (Vascepa, Amarin), may have been related to harms caused by the placebo mineral oil.
Results show that allocation to icosapent ethyl had minimal effects on a series of biomarkers associated with atherosclerotic disease, whereas levels of these biomarkers increased among those allocated to mineral oil.
At 12 months, the median percent increases from baseline in the mineral oil group were 1.5% for homocysteine, 2.2% for lipoprotein(a), 10.9% for oxidized low-density-lipoprotein (LDL) cholesterol, 16.2% for interleukin (IL)-6, 18.5% for lipoprotein-associated phospholipase A2, 21.9% for high-sensitivity C-reactive protein (hsCRP), and 28.9% for IL-1β. The changes were similar at 24 months. However, in the icosapent ethyl group, there were minimal changes in these biomarkers at 12 and 24 months.
The study was published online in Circulation.
The authors, led by Paul Ridker, MD, Brigham & Women’s Hospital, Boston, do not voice much opinion on what the results mean, concluding that “the effect of these findings on the interpretation of the REDUCE-IT trial results remains unclear and will require further investigation.”
They also say that a second icosapent ethyl trial using a nonmineral oil comparator “would help resolve ongoing controversy.”
However, the authors are a mixed group; Dr. Ridker and some of his coauthors were not part of the original REDUCE-IT trial, whereas other coauthors were members of the REDUCE-IT steering committee, and one was an employee of Amarin.
Lead investigator of the REDUCE-IT trial, Deepak Bhatt, MD, also from Brigham & Women’s Hospital, who is the senior author of the current study, played down the new findings, saying they did not offer much new incremental information on mechanistic insight.
“These are small degrees of biomarker increases on an absolute level we are seeing with the placebo that would not be expected to produce harm,” Dr. Bhatt told this news organization. He also said the Circulation peer-review process had removed some of the discussion, which could lead to some “misinterpretation” of the authors’ views.
Dr. Ridker was unavailable for further comment.
The publication of this study has inevitably poured fuel on the fire regarding the controversy that has long dogged the REDUCE-IT trial, with questions about the large reduction in event rates seen with icosapent ethyl without an obvious mechanistic explanation.
‘Smoking gun’
One of the loudest critics of the study, Steve Nissen, MD, Cleveland Clinic, described the new findings as “the closest thing I’ve seen to a smoking gun in medicine for a long, long time.”
“The result of this new analysis shows that mineral oil increases virtually every inflammatory and lipid marker that they measured,” he commented.
“There are a lot of theories, but the bottom line is that something really bad happened in the mineral-oil group, which makes icosapent ethyl look efficacious. In my view, this needs to be reviewed by the FDA for consideration of removing the label claim for cardiovascular benefit.”
Other experts in the field not directly involved in the study voiced concern about these new findings, adding to calls for another trial.
In a Twitter thread on the issue, Harlan Krumholz, MD, describes the Circulation publication as “an exceptionally important article,” adding that it is “time to rethink this drug.”
“My point is ... once you know you have non-neutral comparator and the effect on risk biomarkers is far from trivial ... then you have introduced substantial uncertainty about the trial result, as conveyed by the authors ... and no one can say what would happen with a neutral comparator,” Dr. Krumholz writes.
In an accompanying editorial in Circulation, Robert Harrington, MD, professor of medicine at Stanford (Calif.) University, concludes that “the hard reality is that we are left with uncertainties and the questions raised by use of the mineral oil as placebo can only be answered by another randomized controlled trial.”
“My main points are that the chosen placebo was not inert (an essential characteristic for an active control–placebo comparison), that the biomarker data show elevations in multiple markers involved with inflammatory pathways associated with atherosclerosis, and that these data create enough uncertainty in the trial interpretation that the best recourse to answer the criticisms is to do another trial with a truly inert placebo,” Dr. Harrington said in an interview.
He added that Dr. Bhatt’s point that the changes in biomarkers are too small to really matter clinically may be right. “But for me, the uncertainties mean that I have less confidence in the true magnitude of the treatment effect than I would have if there were no changes in the inflammatory markers.”
In Circulation, the authors say it is unclear why multiple biomarkers increased over time among REDUCE-IT participants allocated to mineral oil. They note that no substantive changes in these biomarkers were observed in the placebo groups over periods of 3-5 years in other trials, including JUPITER, CIRT, CANTOS, SPIRE, and the STRENGTH trials, which evaluated a different high-dose omega-3 oil product but used corn oil as the placebo.
“The core design of REDUCE-IT does not make it possible to resolve convincingly whether any adverse effects associated with mineral oil use as a comparator may have affected clinical outcomes,” they write.
They point out that regulatory agencies evaluating REDUCE-IT estimated that approximately 3% of the net clinical benefit observed with icosapent ethyl might have been a consequence of adverse biomarker effects on LDL cholesterol and hsCRP attributable to mineral oil. But in the context of an overall 25% relative risk reduction in first events and a 30% reduction in total ischemic events observed, a potential bias of this magnitude, even if doubled in size, would be unlikely to fully attenuate the overall benefit of icosapent ethyl observed.
They add that they are not aware of a method to assess what the potential magnitude might be of a combination of the multiple effects.
New data do not change the debate
“We did a large, well-powered randomized trial, and this paper shouldn’t change anything in how that trial should be interpreted,” Dr. Bhatt said in an interview.
He claims the new biomarkers evaluated in the study are correlated with LDL and CRP, data which have already been reported and analyzed so have limited relevance.
“It’s not really independent biomarker information; this is what we would expect to see when we see small increases in LDL and CRP. So, I don’t think this new information fundamentally changes the debate,” he said.
Dr. Bhatt also pointed out that the study highlights relative increases rather than absolute increases in the biomarkers, making it seem more alarming than is actually the case.
“The paper makes it seem like that there are large increases in these other biomarkers, but the values reported are relative increases and the absolute increases were actually rather small. In many cases, the changes reported are less than the lower limit of quantification of the assay used,” he noted.
He added: “Even if one is unable to get around the placebo issue in the REDUCE-IT trial, there will always be the JELIS trial – a randomized trial with no placebo showing a 19% relative risk reduction. While the biomarker data may be interesting, what really matters in the end is clinical events. And significant reductions in two independent trials should be enough.”
Dr. Bhatt says the REDUCE-IT steering committee does not believe another trial is needed. “Maybe a different population would be good – such as primary prevention, patients without elevated triglycerides – but just repeating REDUCE- IT with a different placebo would be a waste of resources,” he commented.
But Dr. Nissen refuted Dr. Bhatt’s claims.
“These biomarkers are not in the same pathways as LDL and CRP, and these are not small increases. In the CANTOS trial, a monoclonal antibody against interleukin-1β beta showed a significant benefit. The increase in interleukin-1β now reported in REDUCE-IT is exactly the opposite of CANTOS,” he pointed out.
“The FDA did not know about these additional biomarkers when it reviewed the data on LDL and CRP. Now we have new information. It needs to be looked at again,” Dr. Nissen added.
Funding for the study was provided by Amarin Pharma. Dr. Bhatt was the lead investigator of the REDUCE-IT trial. Dr. Nissen was the lead investigator the STRENGTH trial. Further disclosures of the authors can be found in Circulation.
A version of this article first appeared on Medscape.com.
FROM CIRCULATION
Nordic walking bests other workouts on functional outcome in CVD
Nordic walking was significantly better at improving functional capacity than were moderate- to vigorous-intensity continuous training and high-intensity interval training (HIIT) in a single-center randomized controlled trial.
Participants who did Nordic walking saw better improvements in functional capacity, measured via the 6-minute walk test distances, than did individuals doing either of the other exercise strategies (interaction effect, P = .010).
From baseline to 26 weeks, the average changes in 6-minute walk test distance were 55.6 m and 59.9 m for moderate- to vigorous-intensity continuous training and HIIT, respectively, but 94.2 m in the Nordic walking group, reported Tasuku Terada, PhD, University of Ottawa Heart Institute, Ontario, and colleagues.
Previous research looked at these results at the end of a 12-week supervised exercise intervention and showed that although all three strategies were safe and had positive effects on physical and mental health in these patients, Nordic walking had a better effect in raising the 6-minute walk test scores than did moderate- to vigorous-intensity continuous training and HIIT, the researchers noted.
“This study is a follow-up on the previous study to show that Nordic walking had greater sustained effects even after the observation phase,” from 12 to 26 weeks, Dr. Terada said in an interview.
“Exercise is a medicine to improve the health of patients, but unfortunately, sometimes it is not as often utilized,” Dr. Terada told this news organization.
Giving patients additional exercise modalities is beneficial because not everyone likes HIIT workouts or long continuous walking, Dr. Terada said. “So, if that’s the case, we can recommend Nordic walking as another type of exercise and expect a similar or good impact in functional capacity.”
The results were published online in the Canadian Journal of Cardiology.
“I think it honestly supports the idea that, as many other studies show, physical activity and exercise improve functional capacity no matter how you measure it and have beneficial effects on mental health and quality of life and particularly depression as well,” Carl “Chip” Lavie, MD, University of Queensland, New Orleans, who coauthored an editorial accompanying the publication, said in an interview.
“Clinicians need to get patients to do the type of exercise that they are going to do. A lot of people ask what’s the best exercise, and the best exercise is one that the person is going to do,” Dr. Lavie said.
Nordic walking is an enhanced form of walking that engages the upper and lower body musculatures, noted Dr. Lavie.
“With regard to Nordic walking, I think that now adds an additional option that many people wouldn’t have thought about. For many of the patients that have issues that are musculoskeletal, issues with posture, gait, or balance, using the poles can be a way to allow them to walk much better and increase their speed, and as they do that, they become fitter,” Dr. Lavie continued.
Moreover, these findings support the use of Nordic walking in cardiac rehabilitation programs, the editorialists noted.
Cardiac rehabilitation
The study examined patients with coronary artery disease who underwent cardiac revascularization. They were then referred by their physicians to cardiac rehabilitation.
Participants were randomly assigned to one of the following intervention groups: Nordic walking (n = 30), moderate- to vigorous-intensity continuous training (n = 27), and HIIT (n = 29) for a 12-week period. There was then an additional 14-week observation period after the exercise program. Mean age was 60 years across the intervention groups.
The research team analyzed the extent of participants’ depression with Beck Depression Inventory–II, quality of life with Short Form–36 and HeartQoL, and functional capacity with a 6-minute walk test. They assessed functional capacity, depression, and quality of life at baseline, 12 weeks, and 26 weeks.
Using linear mixed models with extended measures, the study authors evaluated sustained effects, which were between week 12 and week 26, and prolonged effects, which were between baseline and week 26.
From baseline to 26 weeks, participants saw significantly better outcomes in quality of life, depression symptoms, and 6-minute walk test (P < .05).
Physical quality of life and 6-minute walk test distance rose significantly between weeks 12 and 26 (P < .05).
Notably, at week 26, all training groups achieved the minimal clinical threshold difference of 54 m, although participants in the Nordic walking cohort demonstrated significantly greater improvement in outcomes.
Other data indicated the following:
- From baseline to week 12, physical activity levels rose significantly, and this improvement was sustained through the observation period.
- During the observation period, mental component summary significantly declined while physical component summary outcomes improved.
- After completion of cardiac rehabilitation, functional capacity continued to increase significantly.
- Moderate- to vigorous-intensity continuous training, HIIT, and Nordic walking had positive and significant prolonged effects on depression symptoms and general and disease-specific quality of life, with no differences in the extent of improvements between exercise types.
Some limitations of the study include the fact that women comprised a small portion of the study group, which limits the generalizability of these data, the cohort was recruited from a single medical facility, and there was a short follow-up time, the researchers noted.
“Further research is warranted to investigate the efficacy and integration of Nordic walking into home-based exercise after supervised cardiac rehabilitation for maintenance of physical and mental health,” the editorialists concluded.
Dr. Terada, Dr. Lavie, and Dr. Taylor reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Nordic walking was significantly better at improving functional capacity than were moderate- to vigorous-intensity continuous training and high-intensity interval training (HIIT) in a single-center randomized controlled trial.
Participants who did Nordic walking saw better improvements in functional capacity, measured via the 6-minute walk test distances, than did individuals doing either of the other exercise strategies (interaction effect, P = .010).
From baseline to 26 weeks, the average changes in 6-minute walk test distance were 55.6 m and 59.9 m for moderate- to vigorous-intensity continuous training and HIIT, respectively, but 94.2 m in the Nordic walking group, reported Tasuku Terada, PhD, University of Ottawa Heart Institute, Ontario, and colleagues.
Previous research looked at these results at the end of a 12-week supervised exercise intervention and showed that although all three strategies were safe and had positive effects on physical and mental health in these patients, Nordic walking had a better effect in raising the 6-minute walk test scores than did moderate- to vigorous-intensity continuous training and HIIT, the researchers noted.
“This study is a follow-up on the previous study to show that Nordic walking had greater sustained effects even after the observation phase,” from 12 to 26 weeks, Dr. Terada said in an interview.
“Exercise is a medicine to improve the health of patients, but unfortunately, sometimes it is not as often utilized,” Dr. Terada told this news organization.
Giving patients additional exercise modalities is beneficial because not everyone likes HIIT workouts or long continuous walking, Dr. Terada said. “So, if that’s the case, we can recommend Nordic walking as another type of exercise and expect a similar or good impact in functional capacity.”
The results were published online in the Canadian Journal of Cardiology.
“I think it honestly supports the idea that, as many other studies show, physical activity and exercise improve functional capacity no matter how you measure it and have beneficial effects on mental health and quality of life and particularly depression as well,” Carl “Chip” Lavie, MD, University of Queensland, New Orleans, who coauthored an editorial accompanying the publication, said in an interview.
“Clinicians need to get patients to do the type of exercise that they are going to do. A lot of people ask what’s the best exercise, and the best exercise is one that the person is going to do,” Dr. Lavie said.
Nordic walking is an enhanced form of walking that engages the upper and lower body musculatures, noted Dr. Lavie.
“With regard to Nordic walking, I think that now adds an additional option that many people wouldn’t have thought about. For many of the patients that have issues that are musculoskeletal, issues with posture, gait, or balance, using the poles can be a way to allow them to walk much better and increase their speed, and as they do that, they become fitter,” Dr. Lavie continued.
Moreover, these findings support the use of Nordic walking in cardiac rehabilitation programs, the editorialists noted.
Cardiac rehabilitation
The study examined patients with coronary artery disease who underwent cardiac revascularization. They were then referred by their physicians to cardiac rehabilitation.
Participants were randomly assigned to one of the following intervention groups: Nordic walking (n = 30), moderate- to vigorous-intensity continuous training (n = 27), and HIIT (n = 29) for a 12-week period. There was then an additional 14-week observation period after the exercise program. Mean age was 60 years across the intervention groups.
The research team analyzed the extent of participants’ depression with Beck Depression Inventory–II, quality of life with Short Form–36 and HeartQoL, and functional capacity with a 6-minute walk test. They assessed functional capacity, depression, and quality of life at baseline, 12 weeks, and 26 weeks.
Using linear mixed models with extended measures, the study authors evaluated sustained effects, which were between week 12 and week 26, and prolonged effects, which were between baseline and week 26.
From baseline to 26 weeks, participants saw significantly better outcomes in quality of life, depression symptoms, and 6-minute walk test (P < .05).
Physical quality of life and 6-minute walk test distance rose significantly between weeks 12 and 26 (P < .05).
Notably, at week 26, all training groups achieved the minimal clinical threshold difference of 54 m, although participants in the Nordic walking cohort demonstrated significantly greater improvement in outcomes.
Other data indicated the following:
- From baseline to week 12, physical activity levels rose significantly, and this improvement was sustained through the observation period.
- During the observation period, mental component summary significantly declined while physical component summary outcomes improved.
- After completion of cardiac rehabilitation, functional capacity continued to increase significantly.
- Moderate- to vigorous-intensity continuous training, HIIT, and Nordic walking had positive and significant prolonged effects on depression symptoms and general and disease-specific quality of life, with no differences in the extent of improvements between exercise types.
Some limitations of the study include the fact that women comprised a small portion of the study group, which limits the generalizability of these data, the cohort was recruited from a single medical facility, and there was a short follow-up time, the researchers noted.
“Further research is warranted to investigate the efficacy and integration of Nordic walking into home-based exercise after supervised cardiac rehabilitation for maintenance of physical and mental health,” the editorialists concluded.
Dr. Terada, Dr. Lavie, and Dr. Taylor reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Nordic walking was significantly better at improving functional capacity than were moderate- to vigorous-intensity continuous training and high-intensity interval training (HIIT) in a single-center randomized controlled trial.
Participants who did Nordic walking saw better improvements in functional capacity, measured via the 6-minute walk test distances, than did individuals doing either of the other exercise strategies (interaction effect, P = .010).
From baseline to 26 weeks, the average changes in 6-minute walk test distance were 55.6 m and 59.9 m for moderate- to vigorous-intensity continuous training and HIIT, respectively, but 94.2 m in the Nordic walking group, reported Tasuku Terada, PhD, University of Ottawa Heart Institute, Ontario, and colleagues.
Previous research looked at these results at the end of a 12-week supervised exercise intervention and showed that although all three strategies were safe and had positive effects on physical and mental health in these patients, Nordic walking had a better effect in raising the 6-minute walk test scores than did moderate- to vigorous-intensity continuous training and HIIT, the researchers noted.
“This study is a follow-up on the previous study to show that Nordic walking had greater sustained effects even after the observation phase,” from 12 to 26 weeks, Dr. Terada said in an interview.
“Exercise is a medicine to improve the health of patients, but unfortunately, sometimes it is not as often utilized,” Dr. Terada told this news organization.
Giving patients additional exercise modalities is beneficial because not everyone likes HIIT workouts or long continuous walking, Dr. Terada said. “So, if that’s the case, we can recommend Nordic walking as another type of exercise and expect a similar or good impact in functional capacity.”
The results were published online in the Canadian Journal of Cardiology.
“I think it honestly supports the idea that, as many other studies show, physical activity and exercise improve functional capacity no matter how you measure it and have beneficial effects on mental health and quality of life and particularly depression as well,” Carl “Chip” Lavie, MD, University of Queensland, New Orleans, who coauthored an editorial accompanying the publication, said in an interview.
“Clinicians need to get patients to do the type of exercise that they are going to do. A lot of people ask what’s the best exercise, and the best exercise is one that the person is going to do,” Dr. Lavie said.
Nordic walking is an enhanced form of walking that engages the upper and lower body musculatures, noted Dr. Lavie.
“With regard to Nordic walking, I think that now adds an additional option that many people wouldn’t have thought about. For many of the patients that have issues that are musculoskeletal, issues with posture, gait, or balance, using the poles can be a way to allow them to walk much better and increase their speed, and as they do that, they become fitter,” Dr. Lavie continued.
Moreover, these findings support the use of Nordic walking in cardiac rehabilitation programs, the editorialists noted.
Cardiac rehabilitation
The study examined patients with coronary artery disease who underwent cardiac revascularization. They were then referred by their physicians to cardiac rehabilitation.
Participants were randomly assigned to one of the following intervention groups: Nordic walking (n = 30), moderate- to vigorous-intensity continuous training (n = 27), and HIIT (n = 29) for a 12-week period. There was then an additional 14-week observation period after the exercise program. Mean age was 60 years across the intervention groups.
The research team analyzed the extent of participants’ depression with Beck Depression Inventory–II, quality of life with Short Form–36 and HeartQoL, and functional capacity with a 6-minute walk test. They assessed functional capacity, depression, and quality of life at baseline, 12 weeks, and 26 weeks.
Using linear mixed models with extended measures, the study authors evaluated sustained effects, which were between week 12 and week 26, and prolonged effects, which were between baseline and week 26.
From baseline to 26 weeks, participants saw significantly better outcomes in quality of life, depression symptoms, and 6-minute walk test (P < .05).
Physical quality of life and 6-minute walk test distance rose significantly between weeks 12 and 26 (P < .05).
Notably, at week 26, all training groups achieved the minimal clinical threshold difference of 54 m, although participants in the Nordic walking cohort demonstrated significantly greater improvement in outcomes.
Other data indicated the following:
- From baseline to week 12, physical activity levels rose significantly, and this improvement was sustained through the observation period.
- During the observation period, mental component summary significantly declined while physical component summary outcomes improved.
- After completion of cardiac rehabilitation, functional capacity continued to increase significantly.
- Moderate- to vigorous-intensity continuous training, HIIT, and Nordic walking had positive and significant prolonged effects on depression symptoms and general and disease-specific quality of life, with no differences in the extent of improvements between exercise types.
Some limitations of the study include the fact that women comprised a small portion of the study group, which limits the generalizability of these data, the cohort was recruited from a single medical facility, and there was a short follow-up time, the researchers noted.
“Further research is warranted to investigate the efficacy and integration of Nordic walking into home-based exercise after supervised cardiac rehabilitation for maintenance of physical and mental health,” the editorialists concluded.
Dr. Terada, Dr. Lavie, and Dr. Taylor reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM THE CANADIAN JOURNAL OF CARDIOLOGY
Early cardiac rehab as effective as later start after sternotomy
Cardiac rehabilitation (CR) started 2 weeks after sternotomy for a cardiac procedure was noninferior to usual care, in which CR starts 6 weeks after the procedure, with a greater improvement in 6-minute walk test outcomes, a randomized study suggests.
There was no difference in adverse events between groups, although the researchers pointed out that the study was not powered specifically for safety outcomes.
“Cardiac surgical techniques have evolved significantly over the last 60 years, leading to improved survival and shorter hospital stays,” Gordon McGregor, PhD, University of Warwick, Coventry, England, told this news organization. “However, sternal precautions and rehabilitation guidelines have not changed accordingly. There has never been a guideline based on empirical evidence to support rehabilitation professionals working with cardiac surgery patients after median sternotomy.”
“By adopting a progressive individualized approach,” he added, “cardiac surgery sternotomy patients can start cardiac rehabilitation up to 4 weeks earlier than current guidance, and thus potentially complete their recovery sooner.”
Results of the Early Initiation of Poststernotomy Cardiac Rehabilitation Exercise Training study were published online in JAMA Cardiology.
In the study, Dr. McGregor and colleagues randomly assigned 158 patients (mean age, 63 years; 84% men) to 8 weeks of 1-hour, twice-weekly supervised CR exercise training starting 2 weeks (early) or 6 weeks (usual care) after sternotomy.
The primary outcome was change in the 6-minute walk test distance from baseline to 10 or 14 weeks after sternotomy, respectively, and 12 months after randomization.
For usual care, training followed British standards: a warm-up with light cardiovascular and mobility exercises; continuous moderate-intensity cardiovascular exercise; a cooldown; functional exercises using resistance machines and free weights; and upper-body exercises designed to prevent sternal and leg wound pain and complications.
There are no specific outpatient CR exercise guidelines for early CR, so study participants followed an individualized exercise program for the first 2-3 weeks after surgery, starting with light mobility and moderate-intensity cardiovascular training when they could do those exercises with minimal discomfort. They then progressed to current British standards, as per usual care.
Forty patients were lost to follow-up, largely because of the pandemic; about half the participants in each group were included in the primary analysis.
Early CR was not inferior to usual care, the authors wrote. The mean change in 6-minute walk distance from baseline to completion of CR was 28 meters greater in the early group than in the usual-care group, and was achieved 4 weeks earlier in the recovery timeline.
Secondary outcomes (functional fitness and quality of life) improved in both groups and between-group differences were not statistically significant, indicating the noninferiority of early CR, the authors noted.
Safety not proven
There were more adverse events in the early group than in the usual-care group (58 vs. 46) and more serious adverse events (18 vs. 14), but fewer deaths (1 vs. 2).
Although there was no between-group difference in the likelihood of having an adverse or serious adverse event, Dr. McGregor acknowledged that the study was “not powered specifically for safety outcomes.” He added that “there is the potential to run a very large multination definitive superiority [randomized, controlled trial] with safety as the primary outcome; however, a very large sample would be required.”
Meanwhile, he said, “we can say with some degree of certainty that early CR was likely as safe as usual-care CR. In the United Kingdom, we work closely with the British Association for Cardiovascular Prevention and Rehabilitation and the Association of Chartered Physiotherapists in Cardiovascular Rehabilitation, who will incorporate our findings in their guidelines and training courses.”
Questions remain
Asked to comment on the study, John Larry, MD, medical director of cardiology and cardiac rehabilitation at the Ohio State University Wexner Medical Center East Hospital, Columbus, said: “For those under time pressure to return to work, [early CR] could be an advantage to allow more rehab time and improved stamina prior to their return-to-work date.”
That said, he noted, “we typically delay any significant upper-body training activities for 8-10 weeks to avoid impact on healing of the sternum. Thus ... starting sooner would limit the amount of time a patient would have to engage in any upper-body resistance training. Many lose upper body strength after surgery, so this is an important part of the recovery/rehab process.”
Matthew Tomey, MD, director of the cardiac intensive care unit, Mount Sinai Morningside, New York, advised “caution” when interpreting the findings, stating that “there was no evident difference in the primary outcome measure of functional capacity by 14 weeks, and the trial was not designed to directly assess impact on either social functioning or economic productivity.”
“I would be interested to [see] more comprehensive data on safety in a larger, more diverse sample of postoperative patients,” he said, “as well as evidence to indicate clear advantage of an earlier start for patient-centered outcomes specifically after cardiac surgery.
“Perhaps the greatest challenges to full realization of the benefits of CR in practice have been gaps in referral and gaps in enrollment,” he added. “It is incumbent upon us as clinicians to counsel our patients and to provide appropriate referrals.”
The study was supported by the Medical and Life Sciences Research Fund and the Jeremy Pilcher Memorial Fund. No conflicts of interest were reported.
A version of this article first appeared on Medscape.com.
Cardiac rehabilitation (CR) started 2 weeks after sternotomy for a cardiac procedure was noninferior to usual care, in which CR starts 6 weeks after the procedure, with a greater improvement in 6-minute walk test outcomes, a randomized study suggests.
There was no difference in adverse events between groups, although the researchers pointed out that the study was not powered specifically for safety outcomes.
“Cardiac surgical techniques have evolved significantly over the last 60 years, leading to improved survival and shorter hospital stays,” Gordon McGregor, PhD, University of Warwick, Coventry, England, told this news organization. “However, sternal precautions and rehabilitation guidelines have not changed accordingly. There has never been a guideline based on empirical evidence to support rehabilitation professionals working with cardiac surgery patients after median sternotomy.”
“By adopting a progressive individualized approach,” he added, “cardiac surgery sternotomy patients can start cardiac rehabilitation up to 4 weeks earlier than current guidance, and thus potentially complete their recovery sooner.”
Results of the Early Initiation of Poststernotomy Cardiac Rehabilitation Exercise Training study were published online in JAMA Cardiology.
In the study, Dr. McGregor and colleagues randomly assigned 158 patients (mean age, 63 years; 84% men) to 8 weeks of 1-hour, twice-weekly supervised CR exercise training starting 2 weeks (early) or 6 weeks (usual care) after sternotomy.
The primary outcome was change in the 6-minute walk test distance from baseline to 10 or 14 weeks after sternotomy, respectively, and 12 months after randomization.
For usual care, training followed British standards: a warm-up with light cardiovascular and mobility exercises; continuous moderate-intensity cardiovascular exercise; a cooldown; functional exercises using resistance machines and free weights; and upper-body exercises designed to prevent sternal and leg wound pain and complications.
There are no specific outpatient CR exercise guidelines for early CR, so study participants followed an individualized exercise program for the first 2-3 weeks after surgery, starting with light mobility and moderate-intensity cardiovascular training when they could do those exercises with minimal discomfort. They then progressed to current British standards, as per usual care.
Forty patients were lost to follow-up, largely because of the pandemic; about half the participants in each group were included in the primary analysis.
Early CR was not inferior to usual care, the authors wrote. The mean change in 6-minute walk distance from baseline to completion of CR was 28 meters greater in the early group than in the usual-care group, and was achieved 4 weeks earlier in the recovery timeline.
Secondary outcomes (functional fitness and quality of life) improved in both groups and between-group differences were not statistically significant, indicating the noninferiority of early CR, the authors noted.
Safety not proven
There were more adverse events in the early group than in the usual-care group (58 vs. 46) and more serious adverse events (18 vs. 14), but fewer deaths (1 vs. 2).
Although there was no between-group difference in the likelihood of having an adverse or serious adverse event, Dr. McGregor acknowledged that the study was “not powered specifically for safety outcomes.” He added that “there is the potential to run a very large multination definitive superiority [randomized, controlled trial] with safety as the primary outcome; however, a very large sample would be required.”
Meanwhile, he said, “we can say with some degree of certainty that early CR was likely as safe as usual-care CR. In the United Kingdom, we work closely with the British Association for Cardiovascular Prevention and Rehabilitation and the Association of Chartered Physiotherapists in Cardiovascular Rehabilitation, who will incorporate our findings in their guidelines and training courses.”
Questions remain
Asked to comment on the study, John Larry, MD, medical director of cardiology and cardiac rehabilitation at the Ohio State University Wexner Medical Center East Hospital, Columbus, said: “For those under time pressure to return to work, [early CR] could be an advantage to allow more rehab time and improved stamina prior to their return-to-work date.”
That said, he noted, “we typically delay any significant upper-body training activities for 8-10 weeks to avoid impact on healing of the sternum. Thus ... starting sooner would limit the amount of time a patient would have to engage in any upper-body resistance training. Many lose upper body strength after surgery, so this is an important part of the recovery/rehab process.”
Matthew Tomey, MD, director of the cardiac intensive care unit, Mount Sinai Morningside, New York, advised “caution” when interpreting the findings, stating that “there was no evident difference in the primary outcome measure of functional capacity by 14 weeks, and the trial was not designed to directly assess impact on either social functioning or economic productivity.”
“I would be interested to [see] more comprehensive data on safety in a larger, more diverse sample of postoperative patients,” he said, “as well as evidence to indicate clear advantage of an earlier start for patient-centered outcomes specifically after cardiac surgery.
“Perhaps the greatest challenges to full realization of the benefits of CR in practice have been gaps in referral and gaps in enrollment,” he added. “It is incumbent upon us as clinicians to counsel our patients and to provide appropriate referrals.”
The study was supported by the Medical and Life Sciences Research Fund and the Jeremy Pilcher Memorial Fund. No conflicts of interest were reported.
A version of this article first appeared on Medscape.com.
Cardiac rehabilitation (CR) started 2 weeks after sternotomy for a cardiac procedure was noninferior to usual care, in which CR starts 6 weeks after the procedure, with a greater improvement in 6-minute walk test outcomes, a randomized study suggests.
There was no difference in adverse events between groups, although the researchers pointed out that the study was not powered specifically for safety outcomes.
“Cardiac surgical techniques have evolved significantly over the last 60 years, leading to improved survival and shorter hospital stays,” Gordon McGregor, PhD, University of Warwick, Coventry, England, told this news organization. “However, sternal precautions and rehabilitation guidelines have not changed accordingly. There has never been a guideline based on empirical evidence to support rehabilitation professionals working with cardiac surgery patients after median sternotomy.”
“By adopting a progressive individualized approach,” he added, “cardiac surgery sternotomy patients can start cardiac rehabilitation up to 4 weeks earlier than current guidance, and thus potentially complete their recovery sooner.”
Results of the Early Initiation of Poststernotomy Cardiac Rehabilitation Exercise Training study were published online in JAMA Cardiology.
In the study, Dr. McGregor and colleagues randomly assigned 158 patients (mean age, 63 years; 84% men) to 8 weeks of 1-hour, twice-weekly supervised CR exercise training starting 2 weeks (early) or 6 weeks (usual care) after sternotomy.
The primary outcome was change in the 6-minute walk test distance from baseline to 10 or 14 weeks after sternotomy, respectively, and 12 months after randomization.
For usual care, training followed British standards: a warm-up with light cardiovascular and mobility exercises; continuous moderate-intensity cardiovascular exercise; a cooldown; functional exercises using resistance machines and free weights; and upper-body exercises designed to prevent sternal and leg wound pain and complications.
There are no specific outpatient CR exercise guidelines for early CR, so study participants followed an individualized exercise program for the first 2-3 weeks after surgery, starting with light mobility and moderate-intensity cardiovascular training when they could do those exercises with minimal discomfort. They then progressed to current British standards, as per usual care.
Forty patients were lost to follow-up, largely because of the pandemic; about half the participants in each group were included in the primary analysis.
Early CR was not inferior to usual care, the authors wrote. The mean change in 6-minute walk distance from baseline to completion of CR was 28 meters greater in the early group than in the usual-care group, and was achieved 4 weeks earlier in the recovery timeline.
Secondary outcomes (functional fitness and quality of life) improved in both groups and between-group differences were not statistically significant, indicating the noninferiority of early CR, the authors noted.
Safety not proven
There were more adverse events in the early group than in the usual-care group (58 vs. 46) and more serious adverse events (18 vs. 14), but fewer deaths (1 vs. 2).
Although there was no between-group difference in the likelihood of having an adverse or serious adverse event, Dr. McGregor acknowledged that the study was “not powered specifically for safety outcomes.” He added that “there is the potential to run a very large multination definitive superiority [randomized, controlled trial] with safety as the primary outcome; however, a very large sample would be required.”
Meanwhile, he said, “we can say with some degree of certainty that early CR was likely as safe as usual-care CR. In the United Kingdom, we work closely with the British Association for Cardiovascular Prevention and Rehabilitation and the Association of Chartered Physiotherapists in Cardiovascular Rehabilitation, who will incorporate our findings in their guidelines and training courses.”
Questions remain
Asked to comment on the study, John Larry, MD, medical director of cardiology and cardiac rehabilitation at the Ohio State University Wexner Medical Center East Hospital, Columbus, said: “For those under time pressure to return to work, [early CR] could be an advantage to allow more rehab time and improved stamina prior to their return-to-work date.”
That said, he noted, “we typically delay any significant upper-body training activities for 8-10 weeks to avoid impact on healing of the sternum. Thus ... starting sooner would limit the amount of time a patient would have to engage in any upper-body resistance training. Many lose upper body strength after surgery, so this is an important part of the recovery/rehab process.”
Matthew Tomey, MD, director of the cardiac intensive care unit, Mount Sinai Morningside, New York, advised “caution” when interpreting the findings, stating that “there was no evident difference in the primary outcome measure of functional capacity by 14 weeks, and the trial was not designed to directly assess impact on either social functioning or economic productivity.”
“I would be interested to [see] more comprehensive data on safety in a larger, more diverse sample of postoperative patients,” he said, “as well as evidence to indicate clear advantage of an earlier start for patient-centered outcomes specifically after cardiac surgery.
“Perhaps the greatest challenges to full realization of the benefits of CR in practice have been gaps in referral and gaps in enrollment,” he added. “It is incumbent upon us as clinicians to counsel our patients and to provide appropriate referrals.”
The study was supported by the Medical and Life Sciences Research Fund and the Jeremy Pilcher Memorial Fund. No conflicts of interest were reported.
A version of this article first appeared on Medscape.com.
FROM JAMA CARDIOLOGY
Thigh muscle fat predicts risk of developing heart failure
in a new study. The association was independent of other cardiometabolic risk factors and measures of adiposity such as body mass index.
The observation raises the possibility of new avenues of research aimed at modifying intramuscular fat levels as a strategy to reduce the risk of developing heart failure.
The study was published online in JACC: Heart Failure.
The authors, led by Kevin Huynh, MD, University of Texas Southwestern Medical Center, Dallas, explained that obesity is a known risk for heart failure, and has been incorporated into risk calculators for heart failure.
However, obesity is a complex and heterogeneous disease with substantial regional variability of adipose deposition in body tissues, they noted. For example, variability in visceral adipose tissue and subcutaneous adipose tissue has been shown to have a differential impact on both cardiovascular risk factors and clinical cardiovascular disease outcomes.
The fat deposition around and within nonadipose tissues (termed “ectopic fat”), such as skeletal muscle, is also a known risk factor for cardiovascular disease, independent of adiposity. However, the impact of peripheral skeletal muscle fat deposition on heart failure risk is not as well studied.
The researchers noted that ectopic fat in skeletal muscle can be measured through imaging and categorized as either intermuscular or intramuscular fat according to the location of muscle fat around or within skeletal muscle, respectively.
The researchers conducted the current study to characterize the association of both intermuscular and intramuscular fat deposition with heart failure risk in a large cohort of older adults.
They used data from 2,399 individuals aged 70-79 years without heart failure at baseline who participated in the Health ABC (Health, Aging and Body Composition) study. Measures of intramuscular and intermuscular fat in the thigh were determined by CT, and the participants were followed for an average of 12 years.
During the follow-up period, there were 485 incident heart failure events. Higher sex-specific tertiles of intramuscular and intermuscular fat were each associated with heart failure risk.
After multivariable adjustment for age, sex, race, education, blood pressure, fasting blood sugar, current smoking, prevalent coronary disease, and creatinine, higher intramuscular fat, but not intermuscular fat, was significantly associated with higher risk for heart failure.
Individuals in the highest tertile of intramuscular fat had a 34% increased risk of developing heart failure, compared with those in the lowest tertile. This finding was independent of other cardiometabolic risk factors, measures of adiposity including body mass index and percent fat, muscle strength, and muscle mass.
The association was slightly attenuated when adjusted for inflammatory markers, suggesting that inflammation may be a contributor.
The association between higher intramuscular fat and heart failure appeared specific to higher risk of incident heart failure with reduced ejection fraction, but not with heart failure with preserved ejection fraction.
The researchers noted that skeletal muscle is a pivotal endocrine organ in addition to the role it plays in the production of mechanical power.
They pointed out that there are differences in the biology of intermuscular and intramuscular fat deposition, and that excess intramuscular fat deposition is a result of dysregulated lipid metabolism and is associated with insulin resistance (a known risk factor for the development of heart failure), inflammation, and muscle wasting conditions.
They concluded that, in patients with heart failure, alterations in skeletal muscle function are most likely affected by multiple contributors, including inflammation, oxidative stress, and neurohormonal factors. “As these factors are also implicated in the pathogenesis of heart failure, intramuscular fat deposition may indicate a biological milieu that increases the risk of heart failure.”
New approaches to reduce heart failure risk?
In an accompanying editorial, Salvatore Carbone, PhD, Virginia Commonwealth University, Richmond, said the findings of the study are “exceptionally novel,” providing novel evidence that noncardiac body composition compartments, particularly intramuscular adipose tissue, can predict the risk for heart failure in a diverse population of older adults.
He called for further research to understand the mechanisms involved and to assess if this risk factor can be effectively modified to reduce the risk of developing heart failure.
Dr. Carbone reported that intramuscular adipose tissue can be influenced by dietary fat intake and can be worsened by accumulation of saturated fatty acids, which also contribute to insulin resistance.
He noted that saturated fatty acid–induced insulin resistance in the skeletal muscle appears to be mediated by proinflammatory pathways within the skeletal muscle itself, which can be reversed by monounsaturated fatty acids, like oleic acid, that can be found in the largest amount in food like olive oil, canola oil, and avocados, among others.
He added that sodium-glucose transporter 2 inhibitors, drugs used in the treatment of diabetes that have also been shown to prevent heart failure in individuals at risk, can also improve the composition of intramuscular adipose tissue by reducing its content of saturated fatty acids and increase the content of monosaturated fatty acids.
The study results suggest that the quality of intramuscular adipose tissue might also play an important role and could be targeted by therapeutic strategies, he commented.
Dr. Carbone concluded that “studies testing novel modalities of exercise training, intentional weight loss, diet quality improvements with and without weight loss (i.e., increase of dietary monounsaturated fatty acids, such as oleic acid), as well as pharmacological anti-inflammatory strategies should be encouraged in this population to test whether the reduction in intramuscular adipose tissue or improvements of its quality can ultimately reduce the risk for heart failure in this population.”
This research was supported by the National Institute on Aging and the National Institute of Nursing Research. Dr. Huynh and Dr. Carbone disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
in a new study. The association was independent of other cardiometabolic risk factors and measures of adiposity such as body mass index.
The observation raises the possibility of new avenues of research aimed at modifying intramuscular fat levels as a strategy to reduce the risk of developing heart failure.
The study was published online in JACC: Heart Failure.
The authors, led by Kevin Huynh, MD, University of Texas Southwestern Medical Center, Dallas, explained that obesity is a known risk for heart failure, and has been incorporated into risk calculators for heart failure.
However, obesity is a complex and heterogeneous disease with substantial regional variability of adipose deposition in body tissues, they noted. For example, variability in visceral adipose tissue and subcutaneous adipose tissue has been shown to have a differential impact on both cardiovascular risk factors and clinical cardiovascular disease outcomes.
The fat deposition around and within nonadipose tissues (termed “ectopic fat”), such as skeletal muscle, is also a known risk factor for cardiovascular disease, independent of adiposity. However, the impact of peripheral skeletal muscle fat deposition on heart failure risk is not as well studied.
The researchers noted that ectopic fat in skeletal muscle can be measured through imaging and categorized as either intermuscular or intramuscular fat according to the location of muscle fat around or within skeletal muscle, respectively.
The researchers conducted the current study to characterize the association of both intermuscular and intramuscular fat deposition with heart failure risk in a large cohort of older adults.
They used data from 2,399 individuals aged 70-79 years without heart failure at baseline who participated in the Health ABC (Health, Aging and Body Composition) study. Measures of intramuscular and intermuscular fat in the thigh were determined by CT, and the participants were followed for an average of 12 years.
During the follow-up period, there were 485 incident heart failure events. Higher sex-specific tertiles of intramuscular and intermuscular fat were each associated with heart failure risk.
After multivariable adjustment for age, sex, race, education, blood pressure, fasting blood sugar, current smoking, prevalent coronary disease, and creatinine, higher intramuscular fat, but not intermuscular fat, was significantly associated with higher risk for heart failure.
Individuals in the highest tertile of intramuscular fat had a 34% increased risk of developing heart failure, compared with those in the lowest tertile. This finding was independent of other cardiometabolic risk factors, measures of adiposity including body mass index and percent fat, muscle strength, and muscle mass.
The association was slightly attenuated when adjusted for inflammatory markers, suggesting that inflammation may be a contributor.
The association between higher intramuscular fat and heart failure appeared specific to higher risk of incident heart failure with reduced ejection fraction, but not with heart failure with preserved ejection fraction.
The researchers noted that skeletal muscle is a pivotal endocrine organ in addition to the role it plays in the production of mechanical power.
They pointed out that there are differences in the biology of intermuscular and intramuscular fat deposition, and that excess intramuscular fat deposition is a result of dysregulated lipid metabolism and is associated with insulin resistance (a known risk factor for the development of heart failure), inflammation, and muscle wasting conditions.
They concluded that, in patients with heart failure, alterations in skeletal muscle function are most likely affected by multiple contributors, including inflammation, oxidative stress, and neurohormonal factors. “As these factors are also implicated in the pathogenesis of heart failure, intramuscular fat deposition may indicate a biological milieu that increases the risk of heart failure.”
New approaches to reduce heart failure risk?
In an accompanying editorial, Salvatore Carbone, PhD, Virginia Commonwealth University, Richmond, said the findings of the study are “exceptionally novel,” providing novel evidence that noncardiac body composition compartments, particularly intramuscular adipose tissue, can predict the risk for heart failure in a diverse population of older adults.
He called for further research to understand the mechanisms involved and to assess if this risk factor can be effectively modified to reduce the risk of developing heart failure.
Dr. Carbone reported that intramuscular adipose tissue can be influenced by dietary fat intake and can be worsened by accumulation of saturated fatty acids, which also contribute to insulin resistance.
He noted that saturated fatty acid–induced insulin resistance in the skeletal muscle appears to be mediated by proinflammatory pathways within the skeletal muscle itself, which can be reversed by monounsaturated fatty acids, like oleic acid, that can be found in the largest amount in food like olive oil, canola oil, and avocados, among others.
He added that sodium-glucose transporter 2 inhibitors, drugs used in the treatment of diabetes that have also been shown to prevent heart failure in individuals at risk, can also improve the composition of intramuscular adipose tissue by reducing its content of saturated fatty acids and increase the content of monosaturated fatty acids.
The study results suggest that the quality of intramuscular adipose tissue might also play an important role and could be targeted by therapeutic strategies, he commented.
Dr. Carbone concluded that “studies testing novel modalities of exercise training, intentional weight loss, diet quality improvements with and without weight loss (i.e., increase of dietary monounsaturated fatty acids, such as oleic acid), as well as pharmacological anti-inflammatory strategies should be encouraged in this population to test whether the reduction in intramuscular adipose tissue or improvements of its quality can ultimately reduce the risk for heart failure in this population.”
This research was supported by the National Institute on Aging and the National Institute of Nursing Research. Dr. Huynh and Dr. Carbone disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
in a new study. The association was independent of other cardiometabolic risk factors and measures of adiposity such as body mass index.
The observation raises the possibility of new avenues of research aimed at modifying intramuscular fat levels as a strategy to reduce the risk of developing heart failure.
The study was published online in JACC: Heart Failure.
The authors, led by Kevin Huynh, MD, University of Texas Southwestern Medical Center, Dallas, explained that obesity is a known risk for heart failure, and has been incorporated into risk calculators for heart failure.
However, obesity is a complex and heterogeneous disease with substantial regional variability of adipose deposition in body tissues, they noted. For example, variability in visceral adipose tissue and subcutaneous adipose tissue has been shown to have a differential impact on both cardiovascular risk factors and clinical cardiovascular disease outcomes.
The fat deposition around and within nonadipose tissues (termed “ectopic fat”), such as skeletal muscle, is also a known risk factor for cardiovascular disease, independent of adiposity. However, the impact of peripheral skeletal muscle fat deposition on heart failure risk is not as well studied.
The researchers noted that ectopic fat in skeletal muscle can be measured through imaging and categorized as either intermuscular or intramuscular fat according to the location of muscle fat around or within skeletal muscle, respectively.
The researchers conducted the current study to characterize the association of both intermuscular and intramuscular fat deposition with heart failure risk in a large cohort of older adults.
They used data from 2,399 individuals aged 70-79 years without heart failure at baseline who participated in the Health ABC (Health, Aging and Body Composition) study. Measures of intramuscular and intermuscular fat in the thigh were determined by CT, and the participants were followed for an average of 12 years.
During the follow-up period, there were 485 incident heart failure events. Higher sex-specific tertiles of intramuscular and intermuscular fat were each associated with heart failure risk.
After multivariable adjustment for age, sex, race, education, blood pressure, fasting blood sugar, current smoking, prevalent coronary disease, and creatinine, higher intramuscular fat, but not intermuscular fat, was significantly associated with higher risk for heart failure.
Individuals in the highest tertile of intramuscular fat had a 34% increased risk of developing heart failure, compared with those in the lowest tertile. This finding was independent of other cardiometabolic risk factors, measures of adiposity including body mass index and percent fat, muscle strength, and muscle mass.
The association was slightly attenuated when adjusted for inflammatory markers, suggesting that inflammation may be a contributor.
The association between higher intramuscular fat and heart failure appeared specific to higher risk of incident heart failure with reduced ejection fraction, but not with heart failure with preserved ejection fraction.
The researchers noted that skeletal muscle is a pivotal endocrine organ in addition to the role it plays in the production of mechanical power.
They pointed out that there are differences in the biology of intermuscular and intramuscular fat deposition, and that excess intramuscular fat deposition is a result of dysregulated lipid metabolism and is associated with insulin resistance (a known risk factor for the development of heart failure), inflammation, and muscle wasting conditions.
They concluded that, in patients with heart failure, alterations in skeletal muscle function are most likely affected by multiple contributors, including inflammation, oxidative stress, and neurohormonal factors. “As these factors are also implicated in the pathogenesis of heart failure, intramuscular fat deposition may indicate a biological milieu that increases the risk of heart failure.”
New approaches to reduce heart failure risk?
In an accompanying editorial, Salvatore Carbone, PhD, Virginia Commonwealth University, Richmond, said the findings of the study are “exceptionally novel,” providing novel evidence that noncardiac body composition compartments, particularly intramuscular adipose tissue, can predict the risk for heart failure in a diverse population of older adults.
He called for further research to understand the mechanisms involved and to assess if this risk factor can be effectively modified to reduce the risk of developing heart failure.
Dr. Carbone reported that intramuscular adipose tissue can be influenced by dietary fat intake and can be worsened by accumulation of saturated fatty acids, which also contribute to insulin resistance.
He noted that saturated fatty acid–induced insulin resistance in the skeletal muscle appears to be mediated by proinflammatory pathways within the skeletal muscle itself, which can be reversed by monounsaturated fatty acids, like oleic acid, that can be found in the largest amount in food like olive oil, canola oil, and avocados, among others.
He added that sodium-glucose transporter 2 inhibitors, drugs used in the treatment of diabetes that have also been shown to prevent heart failure in individuals at risk, can also improve the composition of intramuscular adipose tissue by reducing its content of saturated fatty acids and increase the content of monosaturated fatty acids.
The study results suggest that the quality of intramuscular adipose tissue might also play an important role and could be targeted by therapeutic strategies, he commented.
Dr. Carbone concluded that “studies testing novel modalities of exercise training, intentional weight loss, diet quality improvements with and without weight loss (i.e., increase of dietary monounsaturated fatty acids, such as oleic acid), as well as pharmacological anti-inflammatory strategies should be encouraged in this population to test whether the reduction in intramuscular adipose tissue or improvements of its quality can ultimately reduce the risk for heart failure in this population.”
This research was supported by the National Institute on Aging and the National Institute of Nursing Research. Dr. Huynh and Dr. Carbone disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM JACC: HEART FAILURE