User login
Telehealth Pulmonary Rehabilitation for Patients With Severe Chronic Obstructive Pulmonary Disease
According to World Health Organization estimates, 65 million people have moderate-to-severe chronic obstructive pulmonary disease (COPD) globally, and > 20 million patients with COPD are living in the US.1 COPD is a progressive respiratory disease with a poor prognosis and a significant cause of morbidity and mortality in the US, especially within the Veterans Health Administration (VHA).2 The prevalence of COPD is higher in veterans than it is in the general population. COPD prevalence in the adult US population has been estimated to be between 5% and 15%, whereas in veterans, prevalence estimates have ranged from about 5% to 43%.3-5
COPD is associated with disabling dyspnea, muscle weakness, exercise intolerance, morbidity, and mortality. These symptoms and complications gradually and progressively compromise mobility, ability to perform daily functions, and decrease quality of life (QOL). Dyspnea, fatigue, and discomfort are the principal symptoms that negatively impact exercise tolerance.6,7 Therefore, patients often intentionally limit their activities to avoid these uncomfortable feelings and adopt a more sedentary behavior. As the disease progresses, individuals with COPD will gradually need assistance in performing activities of daily living, which eventually leads to functional dependence.
Pulmonary rehabilitation (PR) is an essential component of the management of symptomatic patients with COPD. PR is an evidence-based, multidisciplinary, comprehensive intervention that includes exercise and education for patients with chronic respiratory disease.8 The key benefits of PR are clinical improvements in dyspnea, physical capacity, QOL, and reduced disability in patients with COPD and other respiratory diseases.9-11 PR was found to improve respiratory health in veterans with COPD and decrease respiratory-related health care utilization.12
Despite the known benefits of PR, many patients with chronic respiratory diseases are not referred or do not have access to rehabilitation. Also, uptake of PR is low due to patient frailty, transportation issues, and other health care access problems.13-15 Unfortunately, in the US health care system, access to PR and other nonpharmacologic treatments can be challenging due to a shortage of available PR programs, limited physician referral to existing programs, and lack of family and social support.16
There are only a few accredited PR programs in VHA facilities, and they tend to be located in urban areas.12,17 Many patients have limited access to the PR programs due to geographic distance to the programs and transportation challenges (eg, limited ability to drive, cost of transportation). Moreover, veterans with COPD are likely to have limited mobility or are homebound due to experiencing shortness of breath with minimal exertion. Given the clear benefits of PR and the increasing impact of COPD on morbidity and mortality of the patients with COPD, strategies to improve the access and capacity of PR are needed. VA telehealth services allow for distribution of health care services in different geographic locations by providing access for the veterans who live in rural and highly rural areas. The most recent implementation of VA Video Connect (VVC) by the VHA provides a new avenue for clinicians to deliver much needed medical care into the veterans’ home.
COPD Telehealth Program
In this article, we describe the processes for developing and delivering an in-home, interactive, supervised PR program for veterans with severe COPD through VA telehealth service. The program consists of 18 sessions delivered over 6 weeks by a licensed physical therapist (PT) and a respiratory therapist (RT). The aims of the telehealth PR are to improve exercise tolerance, reduce dyspnea and fatigue, improve QOL, improve accessibility, and decrease costs and transportation burdens for patients with COPD. The program was developed, implemented and delivered by an interdisciplinary team, including a pulmonologist, PT, RT, physiatrist, and nonclinical supporting staff.
Patient Assessment
To be eligible to participate in the program the patient must: (1) have a forced expiratory volume (FEV1) < 60%; ( 2) be medically stable and be receiving optimal medical management; (3) have no severe cognitive impairments; (4) be able to use a computer and e-mail; (5) be able to ambulate with or without a walking device; (6) be willing to enroll in a smoking cessation program or to stop smoking; (7) be willing to participate without prolonged interruption; and (8) have all visual and auditory impairments corrected with medical devices.
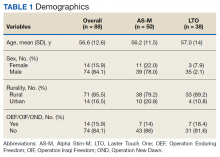
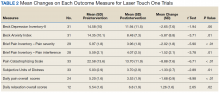
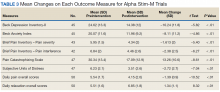
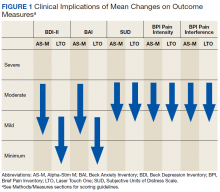
After referral and enrollment, patients receive medical and physical examinations by the PR team, including a pulmonologist, a PT, and a RT, to ensure that the patients are medically stable to undergo rehabilitation and to develop a tailored exercise program while being mindful of the comorbidities, limitations, and precautions, (eg, loss of balance, risk of fall, limited range of motion). The preprogram assessment includes a pulmonary function test, arterial blood gas test, Montreal Cognitive Assessment, Modified Medical Research Council Scale, St. George Respiratory Questionnaire, the COPD Assessment Test, Patient Health Questionnaire-9,Generalized Anxiety Disorder Assessment-7, Epworth Sleepiness Scale, Katz Index of Independence of Activities of Daily Living, medications and inhaler use, oxygen use, breathing pattern, coughing, 6-minute walk test, Modified Borg Dyspnea Scale, grip strength, 5 Times Sit to Stand Test, manual muscle test, gait measure, Timed Up & Go test, clinical balance tests, range of motion, flexibility, sensation, pain, and fall history.18-32 Educational needs (eg, respiratory hygiene, nutrition, infection control, sleep, disease/symptom management) also are evaluated.
This thorough assessment is performed in a face-to-face outpatient visit. During the program participation, a physiatrist may be consulted for additional needs (eg, wheelchair assessment, home safety evaluation/ modifications, and mobility/disability issues). After completing the 6-week program, patients are scheduled for the postprogram evaluation in a face-to-face outpatient visit with the clinicians.
Equipment
Both clinician and the patient are equipped with a computer with Wi-Fi connectivity, a webcam, and a microphone. Patients are provided an exercise pictorial booklet, an exercise compact disk (audio and video), small exercise apparatuses (eg, assorted colors of resistance bands, hand grip exerciser, hand putty, ergometer, harmonica, and pedometer), incentive spirometer, pulse oximeter, cough assistive device (as needed), blood pressure monitor, COPD information booklets, and a diary to use at home during the program.33
Technology Preparation
Prior to starting the telehealth program, the patient is contacted 1 or 2 days before the first session for technical preparation and familiarization of the VA telehealth connection process. Either the PT or RT provides step-by-step instructions for the patient to practice connecting through VVC during this preparatory phone call. The patient also practices using the computer webcam, speaker, and microphone; checks the telehealth scheduling e-mail; and learns how to solve possible common technical issues (eg, adjusting volume and position of webcam). The patient is asked to set up a table close to the computer and to place all exercise apparatuses and respiratory devices on the table surface.
Program Delivery
A secure online VVC is used for connection during the telehealth session. The patient received an e-mail from the telehealth scheduling system with a link for VVC before each session. During the 6-week program, each telehealth session is conducted by a PT and a RT concurrently for 120 minutes, 3 days per week. The PT provides exercises for the patient to attempt, and the RT provides breathing training and monitoring during the session. After a successful connection to VVC, the therapist verifies the patient’s identity and confirms patient consent for the telehealth session.
After this check-in process, the patient performs a self-measure of resting blood pressure (BP), heart rate, respiratory rate, and blood oxygen saturation and reports to the therapists. During the exercise session, fatigue/exertion, dyspnea (Modified Borg Dyspnea Scale; Borg CR10 Scale), BP, heart rate, oxygen saturation, and other clinical symptoms and responses to exercise are monitored by the therapists, using both patient-reported measures and clinical observation by the therapists.34,35 Any medical emergency during the session is reported immediately to the pulmonologist for further management.
Structure
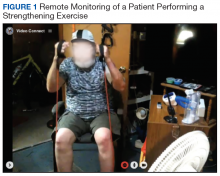
Prior to each exercise session, exercise precautions, fall prevention, good posture, pursed-lip breathing, pacing, and coordinated breathing are discussed with the patient. The PT demonstrates stretching and warm-up exercises in front of the webcam for the patient to follow. Then the patient performs all exercises in view of the webcam during the session (Figure 1). A RT monitors breathing patterns and corrects with verbal instructions if not properly performed.
Loss of skeletal muscle mass and cachexia are highly prevalent comorbidities of COPD and have been associated with breathlessness, functional limitation, and poor prognosis.36 To address these comorbidities, our program consists of progressive strengthening, aerobic, balance, and flexibility exercises. Resistance bands and tubes are used for strengthening exercises. Callisthenic exercises (eg, chair squat, chair stand, knee marching, bridging, single limb stances, and lunge) are used for progressive strengthening and balance exercises. Progression of strengthening and balance exercises are done through increasing the volume of exercise (ie, numbers of sets and repetitions) and increased load and level of difficulty based on the patient’s progress and comorbidity. The exercise program focuses on strengthening muscles, especially large muscle groups, to improve overall muscle strength and performance of functional activities.37
Arm/pedal ergometer and daily walking are used for daily aerobic exercise. In a study of patients with COPD by the PAC-COPD Study Group, step counter use was found to increase physical activity and improve exercise capacity, which supports its use in COPD management.38 During program participation, the patient is asked to wear a pedometer to monitor the number of steps taken per day and to report step data to the therapists during the telehealth session. The pedometer stores the previous 41 calendar days of data and displays the most recent 7 calendar days of data.
The patient is encouraged to set a realistic daily step goal. The general program goal is to increase at least 1000 steps per day. However, this goal can be adjusted depending on the patient’s health status and comorbid conditions. The PAC-COPD Study Group found that for every additional 1000 daily steps at low intensity, COPD hospitalization risk decreased by 20%.39 A magnitude of 2000 steps or about 1 mile of walking per day was found to be associated with increased physical activity and health benefits in the general population.40
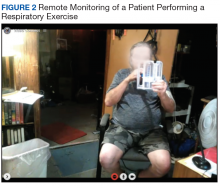
Respiratory muscle training and breathing exercise are provided by the RT, using breathing and incentive spirometer techniques (Figure 2). Huff coughing, diaphragmatic deep breathing, and pursed-lip breathing are instructed by the RT during the session. Effective coughing technique with a cough assistive device is also provided during breathing training if needed.
Patient Education
In patients with COPD, there are numerous positive health benefits associated with education, including assisting the patients to become active participants in the PR program leading to satisfying outcomes; assisting the patients to better understand the lung health, disease processes, physical and psychological changes that occur with COPD; assisting the patients to explore coping strategies for those changes; building lifelong behavioral changes; and developing the self-management skills for sustainability. Through the educational process, patients with COPD can become more skilled at collaborative self-management and improve adherence to their treatment plan, which in turn can result in a reduction in hospital admissions and reduced health care costs.8,41
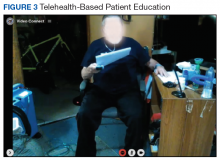
Education is provided with every session after the patient completes the exercise. Patients are required to record their COPD symptoms, daily activity, home exercise program, sleep, food intake, and additional physical or social activity in their COPD diary and to report during the session (Figure 3). A COPD diary assists patients in self-monitoring their COPD symptoms and provides the therapists with information about clinical changes, behavioral changes, and/or specific unmet needs for education. Several topics related to COPD are included in the education session: lung or respiratory disease/condition and self-management; smoking cessation; physical activity; energy-conserving techniques; breathing and coughing techniques; smoking cessation; nutrition/healthy eating and weight counseling; sex and intimacy; psychological counseling and/or group support; emergency planning (eg, medical, travel, and inclement weather); correct use of inhaler and medications; home oxygen; sleep and sleep hygiene; palliative care and advanced directive; infection control; and sputum clearance.42,43
Program Maintenance
After successfully completing the 6-week program, patients are referred to the VA TeleMOVE! Program or MOVE! Weight Management Program for continuous, long-term management of weight, nutrition, physical activity/exercise, and social activity needs or goals. The patients are scheduled for monthly follow-up phone visits for 6 months with the telerehabilitation team for enforcing sustainability. The phone call visit consists of reviewing breathing techniques, exercise program, physical activity, education, encouragement, and addressing any issues that arise during the self-maintained period.
Limitations
There are several issues of concern and precautions when delivering PR through telehealth into the home. First, the patient performs exercises independently without being manually guarded by the therapists. Risk of falls are a major concern due to impaired balance, poor vision, and other possible unusual physiologic responses to exercise (eg, drop in BP, dizziness, loss of balance). The area in front of the computer needs to be cleared of fall hazards (ie, area rug, wires, objects on the floor). The patient also needs to be educated on self-measurements of BP and oxygen saturation and reports to the therapists. The therapists provide detailed instructions on how to obtain these measures correctly; otherwise, the values may not be valid for a clinical judgment during the exercise session or for other clinical management. In a home environment, there is a limited use of exercise apparatuses. For this program, we only used resistance bands/tubes, small arm/leg ergometer, hand grip, and hand putty for the exercise program. We feel that dumbbell and weight plates are not suitable due to a possible risk of injury if the patient accidently drops them.
Advanced balance training is not suitable due to an increased risk for falls. Without the presence of the PT, level of challenge/difficulty is somewhat limited for this telehealth supervision exercise program. In addition, visual and audio quality are necessary for the session. The patient and the therapists need to see each other clearly to ensure correct methods and forms of each exercise. Furthermore, rehearsal of technical skills with the therapists is very important because this population is older and often has limited computer skills. Any technical difficulty or failure can lead to undesirable situations (eg, anxiety episodes, worries, shortness of breath, upset), which compromise exercise performance during the session. Finally, a phone is needed as an alternative in case of a poor VVC connection.
Conclusion
COPD symptoms and complications greatly affect patients’ ability to perform daily activities, decrease QOL and functional ability, and result in extensive use of health services. Many patients have limited access to a PR program at hospitals or rehabilitation centers due to health conditions, lack of transportation, and/or family support. This home-based, interactive telehealth PR program can break down the geographic barriers, solve poor program accessibility, potentially increase the utilization of PR, and reduce the cost and travel required by the patients.
Acknowledgments
The Telehealth Pulmonary Rehabilitation Program was originally funded by the Veterans Health Administration VA ACCESS Program (AS, CL, HKH). We thank all the veterans for their time and effort in participating in this newly developed rehabilitation program.
1. World Health Organization. Chronic obstructive pulmonary disease (COPD). http://www.who.int/news-room/fact-sheets/detail/chronic-obstructive-pulmonary-disease-(copd). Published December 1, 2017. Accessed August 7, 2019.
2. Yu W, Ravelo A, Wagner TH, et al. Prevalence and costs of chronic conditions in the VA health care system. Med Care Res Rev. 2003;60(suppl 3):146S-167S.
3. Doney B, Hnizdo E, Dillon CF, et al. Prevalence of airflow obstruction in U.S. adults aged 40-79 years: NHANES data 1988-1994 and 2007-2010. COPD. 2015;12(4):355-365.
4. Murphy DE, Chaudhry Z, Almoosa KF, Panos RJ. High prevalence of chronic obstructive pulmonary disease among veterans in the urban midwest. Mil Med. 2011;176(5):552-560.
5. Cypel YS, Hines SE, Davey VJ, Eber SM, Schneiderman AI. Self-reported physician-diagnosed chronic obstructive pulmonary disease and spirometry patterns in Vietnam Era US Army Chemical Corps veterans: a retrospective cohort study. Am J Ind Med. 2018;61(10):802-814.
6. Rochester CL. Exercise training in chronic obstructive pulmonary disease. J Rehabil Res Dev. 2003;40(5)(suppl 2):59-80.
7. Cortopassi F, Gurung P, Pinto-Plata V. Chronic obstructive pulmonary disease in elderly patients. Clin Geriatr Med. 2017;33(4):539-552.
8. Spruit MA, Singh SJ, Garvey C, et al; ATS/ERS Task Force on Pulmonary Rehabilitation. An official American Thoracic Society/European Respiratory Society statement: key concepts and advances in pulmonary rehabilitation. Am J Respir Crit Care Med. 2013;188(8):e13-e64.
9. Robinson H, Williams V, Curtis F, Bridle C, Jones AW. Facilitators and barriers to physical activity following pulmonary rehabilitation in COPD: a systematic review of qualitative studies. NPJ Prim Care Respir Med. 2018;28(1):19.
10. McCarthy B, Casey D, Devane D, Murphy K, Murphy E, Lacasse Y. Pulmonary rehabilitation for chronic obstructive pulmonary disease. Cochrane Database Syst Rev. 2015;(2):CD003793.
11. Ries AL, Bauldoff GS, Carlin BW, et al. Pulmonary rehabilitation: joint AACP/AACVPR evidence-based clinical practice guidelines. Chest. 2007;131(suppl 5):4S-42S.
12. Major S, Moreno M, Shelton J, Panos RJ. Veterans with chronic obstructive pulmonary disease achieve clinically relevant improvements in respiratory health after pulmonary rehabilitation. J Cardiopulm Rehabil Prev. 2014;34(6):420-429.
13. Liu Y, Dickerson T, Early F, Fuld J, Clarkson PJ. Understanding influences on the uptake of pulmonary rehabilitation in the East of England: an inclusive design/mixed methods study protocol. BMJ Open. 2018;8(4):e020750.
14. Harris D, Hayter M, Allender S. Factors affecting the offer of pulmonary rehabilitation to patients with chronic obstructive pulmonary disease by primary care professionals: a qualitative study. Prim Health Care Res Dev. 2008;9(4):280-290.
15. Mathar H, Fastholm P, Hansen IR, Larsen NS. Why do patients with COPD decline rehabilitation. Scand J Caring Sci. 2016;30(3):432-441.
16. Han MK, Martinez CH, Au DH, et al. Meeting the challenge of COPD care delivery in the USA: a multiprovider perspective. Lancet Respir Med. 2016;4(6):473-526.
17. American Association of Cardiovascular and Pulmonary Rehabilitation (AACVPR). Online searchable program directory. https://www.aacvpr.org/Resources/Program-Directory Accessed July 19, 2018.
18. Nasreddine ZS, Phillips NA, Bédirian V, et al. The Montreal Cognitive Assessment, MoCA: a brief screening tool for mild cognitive impairment. J Am Geriatr Soc. 2005;53(4):695-699.
19. Fletcher CM, Elmes PC, Fairbairn AS, Wood CH. The significance of respiratory symptoms and the diagnosis of chronic bronchitis in a working population. Br Med J. 1959;2(5147):257-266.
20. O’Donnell DE, Aaron S, Bourbeau J, et al. Canadian Thoracic Society recommendations for management of chronic obstructive pulmonary disease—2007 update. Can Respir J. 2007;14(suppl B):5B-32B.
21. Jones PW, Quirk FH, Baveystock CM. The St George’s Respiratory Questionnaire. Respir Med. 1991;85(suppl B):25-31.
22. Jones PW, Harding G, Berry P, Wiklund I, Chen WH, Kline Leidy N. Development and first validation of the COPD Assessment Test. Eur Respir J. 2009;34(3):648-654.
23. Kroenke K, Spitzer RL, Williams JB. The PHQ-9: validity of a brief depression severity measure. J Gen Intern Med. 2001;16(9):606-613.
24. Spitzer RL, Kroenke K, Williams JBW, Löwe B. A brief measure for assessing generalized anxiety disorder: the GAD-7. Arch Intern Med. 2006;166(10):1092-1097.
25. Johns MW. A new method for measuring daytime sleepiness: the Epworth sleepiness scale. Sleep. 1991;14(6):540-545.
26. Katz S. Assessing self-maintenance: activities of daily living, mobility and instrumental activities of daily living. J Am Geriatr Soc. 1983;31(12):721-727.
27. Holland AE, Spruit MA, Troosters T, et al. An official European Respiratory Society/American Thoracic Society technical standard: field walking tests in chronic respiratory disease. Eur Respir J. 2014;44(6):1428-1446.
28. Mahler DA, Horowitz MB. Perception of breathlessness during exercise in patients with respiratory disease. Med Sci Sports Exerc. 1994;26(9):1078-1081.
29. Liao WC, Wang CH, Yu SY, Chen LY, Wang CY. Grip strength measurement in older adults in Taiwan: a comparison of three testing positions. Australas J Ageing. 2014;33(4):278-282.
30. Buatois S, Miljkovic D, Manckoundia P, et al. Five times sit to stand test is a predictor of recurrent falls in healthy community-living subjects aged 65 and older. J Am Geriatr Soc. 2008;56(8):1575-1577.
31. Bryant MS, Workman CD, Jackson GR. Multidirectional walk test in persons with Parkinson’s disease: a validity study. Int J Rehabil Res. 2015;38(1):88-91.
32. Podsiadlo D, Richardson S. The timed “Up & Go”: a test of basic functional mobility for frail elderly persons. J Am Geriatr Soc. 1991;39(2):142-148.
33. University of Nebraska Medical Center. Timed Up and Go (TUG) Test. https://www.unmc.edu/media/intmed/geriatrics/nebgec/pdf/frailelderlyjuly09/toolkits/timedupandgo_w_norms.pdf. Accessed August 13, 2019.
34. Borg GA. Psychophysical bases of perceived exertion. Med Sci Sports Exerc. 1982;14(5):377-381.
35. Mahler DA, Horowitz MB. Clinical evaluation of exertional dyspnea. Clin Chest Med. 1994;15(2):259-269.
36. Dudgeon D, Baracos VE. Physiological and functional failure in chronic obstructive pulmonary disease, congestive heart failure and cancer: a debilitating intersection of sarcopenia, cachexia and breathlessness. Curr Opin Support Palliat Care. 2016;10(3):236-241.
37. Lee AL, Holland AE. Time to adapt exercise training regimens in pulmonary rehabilitation—a review of the literature. Int J Chron Obstruct Pulmon Dis. 2014;9:1275-1288.
38. Qiu S, Cai X, Wang X, et al. Using step counters to promote physical activity and exercise capacity in patients with chronic obstructive pulmonary disease: a meta-analysis. Ther Adv Respir Dis. 2018;12:1753466618787386.
39. Donaire-Gonzalez D, Gimeno-Santos E, Balcells E, et al; PAC-COPD Study Group. Benefits of physical activity on COPD hospitalization depend on intensity. Eur Respir J. 2015;46(5):1281-1289.
40. Bravata DM, Smith-Spangler C, Sundaram V, et al. Using pedometers to increase physical activity and improve health: a systematic review. JAMA. 2007;298(19):2296-2304.
41. Zwerink M, Brusse-Keizer M, van der Valk PD, et al. Self-management for patients with chronic obstructive pulmonary disease. Cochrane Database Syst Rev. 2014;19(3):CD002990.
42. Wilson JS, O’Neill B, Reilly J, MacMahon J, Bradley JM. Education in pulmonary rehabilitation: the patient’s perspective. Arch Phys Med Rehabil. 2007;88(12):1704-1709.
43. Bourbeau J, Nault D, Dang-Tan T. Self-management and behaviour modification in COPD. Patient Educ Couns. 2004;52(3):271-277.
According to World Health Organization estimates, 65 million people have moderate-to-severe chronic obstructive pulmonary disease (COPD) globally, and > 20 million patients with COPD are living in the US.1 COPD is a progressive respiratory disease with a poor prognosis and a significant cause of morbidity and mortality in the US, especially within the Veterans Health Administration (VHA).2 The prevalence of COPD is higher in veterans than it is in the general population. COPD prevalence in the adult US population has been estimated to be between 5% and 15%, whereas in veterans, prevalence estimates have ranged from about 5% to 43%.3-5
COPD is associated with disabling dyspnea, muscle weakness, exercise intolerance, morbidity, and mortality. These symptoms and complications gradually and progressively compromise mobility, ability to perform daily functions, and decrease quality of life (QOL). Dyspnea, fatigue, and discomfort are the principal symptoms that negatively impact exercise tolerance.6,7 Therefore, patients often intentionally limit their activities to avoid these uncomfortable feelings and adopt a more sedentary behavior. As the disease progresses, individuals with COPD will gradually need assistance in performing activities of daily living, which eventually leads to functional dependence.
Pulmonary rehabilitation (PR) is an essential component of the management of symptomatic patients with COPD. PR is an evidence-based, multidisciplinary, comprehensive intervention that includes exercise and education for patients with chronic respiratory disease.8 The key benefits of PR are clinical improvements in dyspnea, physical capacity, QOL, and reduced disability in patients with COPD and other respiratory diseases.9-11 PR was found to improve respiratory health in veterans with COPD and decrease respiratory-related health care utilization.12
Despite the known benefits of PR, many patients with chronic respiratory diseases are not referred or do not have access to rehabilitation. Also, uptake of PR is low due to patient frailty, transportation issues, and other health care access problems.13-15 Unfortunately, in the US health care system, access to PR and other nonpharmacologic treatments can be challenging due to a shortage of available PR programs, limited physician referral to existing programs, and lack of family and social support.16
There are only a few accredited PR programs in VHA facilities, and they tend to be located in urban areas.12,17 Many patients have limited access to the PR programs due to geographic distance to the programs and transportation challenges (eg, limited ability to drive, cost of transportation). Moreover, veterans with COPD are likely to have limited mobility or are homebound due to experiencing shortness of breath with minimal exertion. Given the clear benefits of PR and the increasing impact of COPD on morbidity and mortality of the patients with COPD, strategies to improve the access and capacity of PR are needed. VA telehealth services allow for distribution of health care services in different geographic locations by providing access for the veterans who live in rural and highly rural areas. The most recent implementation of VA Video Connect (VVC) by the VHA provides a new avenue for clinicians to deliver much needed medical care into the veterans’ home.
COPD Telehealth Program
In this article, we describe the processes for developing and delivering an in-home, interactive, supervised PR program for veterans with severe COPD through VA telehealth service. The program consists of 18 sessions delivered over 6 weeks by a licensed physical therapist (PT) and a respiratory therapist (RT). The aims of the telehealth PR are to improve exercise tolerance, reduce dyspnea and fatigue, improve QOL, improve accessibility, and decrease costs and transportation burdens for patients with COPD. The program was developed, implemented and delivered by an interdisciplinary team, including a pulmonologist, PT, RT, physiatrist, and nonclinical supporting staff.
Patient Assessment
To be eligible to participate in the program the patient must: (1) have a forced expiratory volume (FEV1) < 60%; ( 2) be medically stable and be receiving optimal medical management; (3) have no severe cognitive impairments; (4) be able to use a computer and e-mail; (5) be able to ambulate with or without a walking device; (6) be willing to enroll in a smoking cessation program or to stop smoking; (7) be willing to participate without prolonged interruption; and (8) have all visual and auditory impairments corrected with medical devices.
After referral and enrollment, patients receive medical and physical examinations by the PR team, including a pulmonologist, a PT, and a RT, to ensure that the patients are medically stable to undergo rehabilitation and to develop a tailored exercise program while being mindful of the comorbidities, limitations, and precautions, (eg, loss of balance, risk of fall, limited range of motion). The preprogram assessment includes a pulmonary function test, arterial blood gas test, Montreal Cognitive Assessment, Modified Medical Research Council Scale, St. George Respiratory Questionnaire, the COPD Assessment Test, Patient Health Questionnaire-9,Generalized Anxiety Disorder Assessment-7, Epworth Sleepiness Scale, Katz Index of Independence of Activities of Daily Living, medications and inhaler use, oxygen use, breathing pattern, coughing, 6-minute walk test, Modified Borg Dyspnea Scale, grip strength, 5 Times Sit to Stand Test, manual muscle test, gait measure, Timed Up & Go test, clinical balance tests, range of motion, flexibility, sensation, pain, and fall history.18-32 Educational needs (eg, respiratory hygiene, nutrition, infection control, sleep, disease/symptom management) also are evaluated.
This thorough assessment is performed in a face-to-face outpatient visit. During the program participation, a physiatrist may be consulted for additional needs (eg, wheelchair assessment, home safety evaluation/ modifications, and mobility/disability issues). After completing the 6-week program, patients are scheduled for the postprogram evaluation in a face-to-face outpatient visit with the clinicians.
Equipment
Both clinician and the patient are equipped with a computer with Wi-Fi connectivity, a webcam, and a microphone. Patients are provided an exercise pictorial booklet, an exercise compact disk (audio and video), small exercise apparatuses (eg, assorted colors of resistance bands, hand grip exerciser, hand putty, ergometer, harmonica, and pedometer), incentive spirometer, pulse oximeter, cough assistive device (as needed), blood pressure monitor, COPD information booklets, and a diary to use at home during the program.33
Technology Preparation
Prior to starting the telehealth program, the patient is contacted 1 or 2 days before the first session for technical preparation and familiarization of the VA telehealth connection process. Either the PT or RT provides step-by-step instructions for the patient to practice connecting through VVC during this preparatory phone call. The patient also practices using the computer webcam, speaker, and microphone; checks the telehealth scheduling e-mail; and learns how to solve possible common technical issues (eg, adjusting volume and position of webcam). The patient is asked to set up a table close to the computer and to place all exercise apparatuses and respiratory devices on the table surface.
Program Delivery
A secure online VVC is used for connection during the telehealth session. The patient received an e-mail from the telehealth scheduling system with a link for VVC before each session. During the 6-week program, each telehealth session is conducted by a PT and a RT concurrently for 120 minutes, 3 days per week. The PT provides exercises for the patient to attempt, and the RT provides breathing training and monitoring during the session. After a successful connection to VVC, the therapist verifies the patient’s identity and confirms patient consent for the telehealth session.
After this check-in process, the patient performs a self-measure of resting blood pressure (BP), heart rate, respiratory rate, and blood oxygen saturation and reports to the therapists. During the exercise session, fatigue/exertion, dyspnea (Modified Borg Dyspnea Scale; Borg CR10 Scale), BP, heart rate, oxygen saturation, and other clinical symptoms and responses to exercise are monitored by the therapists, using both patient-reported measures and clinical observation by the therapists.34,35 Any medical emergency during the session is reported immediately to the pulmonologist for further management.
Structure
Prior to each exercise session, exercise precautions, fall prevention, good posture, pursed-lip breathing, pacing, and coordinated breathing are discussed with the patient. The PT demonstrates stretching and warm-up exercises in front of the webcam for the patient to follow. Then the patient performs all exercises in view of the webcam during the session (Figure 1). A RT monitors breathing patterns and corrects with verbal instructions if not properly performed.
Loss of skeletal muscle mass and cachexia are highly prevalent comorbidities of COPD and have been associated with breathlessness, functional limitation, and poor prognosis.36 To address these comorbidities, our program consists of progressive strengthening, aerobic, balance, and flexibility exercises. Resistance bands and tubes are used for strengthening exercises. Callisthenic exercises (eg, chair squat, chair stand, knee marching, bridging, single limb stances, and lunge) are used for progressive strengthening and balance exercises. Progression of strengthening and balance exercises are done through increasing the volume of exercise (ie, numbers of sets and repetitions) and increased load and level of difficulty based on the patient’s progress and comorbidity. The exercise program focuses on strengthening muscles, especially large muscle groups, to improve overall muscle strength and performance of functional activities.37
Arm/pedal ergometer and daily walking are used for daily aerobic exercise. In a study of patients with COPD by the PAC-COPD Study Group, step counter use was found to increase physical activity and improve exercise capacity, which supports its use in COPD management.38 During program participation, the patient is asked to wear a pedometer to monitor the number of steps taken per day and to report step data to the therapists during the telehealth session. The pedometer stores the previous 41 calendar days of data and displays the most recent 7 calendar days of data.
The patient is encouraged to set a realistic daily step goal. The general program goal is to increase at least 1000 steps per day. However, this goal can be adjusted depending on the patient’s health status and comorbid conditions. The PAC-COPD Study Group found that for every additional 1000 daily steps at low intensity, COPD hospitalization risk decreased by 20%.39 A magnitude of 2000 steps or about 1 mile of walking per day was found to be associated with increased physical activity and health benefits in the general population.40
Respiratory muscle training and breathing exercise are provided by the RT, using breathing and incentive spirometer techniques (Figure 2). Huff coughing, diaphragmatic deep breathing, and pursed-lip breathing are instructed by the RT during the session. Effective coughing technique with a cough assistive device is also provided during breathing training if needed.
Patient Education
In patients with COPD, there are numerous positive health benefits associated with education, including assisting the patients to become active participants in the PR program leading to satisfying outcomes; assisting the patients to better understand the lung health, disease processes, physical and psychological changes that occur with COPD; assisting the patients to explore coping strategies for those changes; building lifelong behavioral changes; and developing the self-management skills for sustainability. Through the educational process, patients with COPD can become more skilled at collaborative self-management and improve adherence to their treatment plan, which in turn can result in a reduction in hospital admissions and reduced health care costs.8,41
Education is provided with every session after the patient completes the exercise. Patients are required to record their COPD symptoms, daily activity, home exercise program, sleep, food intake, and additional physical or social activity in their COPD diary and to report during the session (Figure 3). A COPD diary assists patients in self-monitoring their COPD symptoms and provides the therapists with information about clinical changes, behavioral changes, and/or specific unmet needs for education. Several topics related to COPD are included in the education session: lung or respiratory disease/condition and self-management; smoking cessation; physical activity; energy-conserving techniques; breathing and coughing techniques; smoking cessation; nutrition/healthy eating and weight counseling; sex and intimacy; psychological counseling and/or group support; emergency planning (eg, medical, travel, and inclement weather); correct use of inhaler and medications; home oxygen; sleep and sleep hygiene; palliative care and advanced directive; infection control; and sputum clearance.42,43
Program Maintenance
After successfully completing the 6-week program, patients are referred to the VA TeleMOVE! Program or MOVE! Weight Management Program for continuous, long-term management of weight, nutrition, physical activity/exercise, and social activity needs or goals. The patients are scheduled for monthly follow-up phone visits for 6 months with the telerehabilitation team for enforcing sustainability. The phone call visit consists of reviewing breathing techniques, exercise program, physical activity, education, encouragement, and addressing any issues that arise during the self-maintained period.
Limitations
There are several issues of concern and precautions when delivering PR through telehealth into the home. First, the patient performs exercises independently without being manually guarded by the therapists. Risk of falls are a major concern due to impaired balance, poor vision, and other possible unusual physiologic responses to exercise (eg, drop in BP, dizziness, loss of balance). The area in front of the computer needs to be cleared of fall hazards (ie, area rug, wires, objects on the floor). The patient also needs to be educated on self-measurements of BP and oxygen saturation and reports to the therapists. The therapists provide detailed instructions on how to obtain these measures correctly; otherwise, the values may not be valid for a clinical judgment during the exercise session or for other clinical management. In a home environment, there is a limited use of exercise apparatuses. For this program, we only used resistance bands/tubes, small arm/leg ergometer, hand grip, and hand putty for the exercise program. We feel that dumbbell and weight plates are not suitable due to a possible risk of injury if the patient accidently drops them.
Advanced balance training is not suitable due to an increased risk for falls. Without the presence of the PT, level of challenge/difficulty is somewhat limited for this telehealth supervision exercise program. In addition, visual and audio quality are necessary for the session. The patient and the therapists need to see each other clearly to ensure correct methods and forms of each exercise. Furthermore, rehearsal of technical skills with the therapists is very important because this population is older and often has limited computer skills. Any technical difficulty or failure can lead to undesirable situations (eg, anxiety episodes, worries, shortness of breath, upset), which compromise exercise performance during the session. Finally, a phone is needed as an alternative in case of a poor VVC connection.
Conclusion
COPD symptoms and complications greatly affect patients’ ability to perform daily activities, decrease QOL and functional ability, and result in extensive use of health services. Many patients have limited access to a PR program at hospitals or rehabilitation centers due to health conditions, lack of transportation, and/or family support. This home-based, interactive telehealth PR program can break down the geographic barriers, solve poor program accessibility, potentially increase the utilization of PR, and reduce the cost and travel required by the patients.
Acknowledgments
The Telehealth Pulmonary Rehabilitation Program was originally funded by the Veterans Health Administration VA ACCESS Program (AS, CL, HKH). We thank all the veterans for their time and effort in participating in this newly developed rehabilitation program.
According to World Health Organization estimates, 65 million people have moderate-to-severe chronic obstructive pulmonary disease (COPD) globally, and > 20 million patients with COPD are living in the US.1 COPD is a progressive respiratory disease with a poor prognosis and a significant cause of morbidity and mortality in the US, especially within the Veterans Health Administration (VHA).2 The prevalence of COPD is higher in veterans than it is in the general population. COPD prevalence in the adult US population has been estimated to be between 5% and 15%, whereas in veterans, prevalence estimates have ranged from about 5% to 43%.3-5
COPD is associated with disabling dyspnea, muscle weakness, exercise intolerance, morbidity, and mortality. These symptoms and complications gradually and progressively compromise mobility, ability to perform daily functions, and decrease quality of life (QOL). Dyspnea, fatigue, and discomfort are the principal symptoms that negatively impact exercise tolerance.6,7 Therefore, patients often intentionally limit their activities to avoid these uncomfortable feelings and adopt a more sedentary behavior. As the disease progresses, individuals with COPD will gradually need assistance in performing activities of daily living, which eventually leads to functional dependence.
Pulmonary rehabilitation (PR) is an essential component of the management of symptomatic patients with COPD. PR is an evidence-based, multidisciplinary, comprehensive intervention that includes exercise and education for patients with chronic respiratory disease.8 The key benefits of PR are clinical improvements in dyspnea, physical capacity, QOL, and reduced disability in patients with COPD and other respiratory diseases.9-11 PR was found to improve respiratory health in veterans with COPD and decrease respiratory-related health care utilization.12
Despite the known benefits of PR, many patients with chronic respiratory diseases are not referred or do not have access to rehabilitation. Also, uptake of PR is low due to patient frailty, transportation issues, and other health care access problems.13-15 Unfortunately, in the US health care system, access to PR and other nonpharmacologic treatments can be challenging due to a shortage of available PR programs, limited physician referral to existing programs, and lack of family and social support.16
There are only a few accredited PR programs in VHA facilities, and they tend to be located in urban areas.12,17 Many patients have limited access to the PR programs due to geographic distance to the programs and transportation challenges (eg, limited ability to drive, cost of transportation). Moreover, veterans with COPD are likely to have limited mobility or are homebound due to experiencing shortness of breath with minimal exertion. Given the clear benefits of PR and the increasing impact of COPD on morbidity and mortality of the patients with COPD, strategies to improve the access and capacity of PR are needed. VA telehealth services allow for distribution of health care services in different geographic locations by providing access for the veterans who live in rural and highly rural areas. The most recent implementation of VA Video Connect (VVC) by the VHA provides a new avenue for clinicians to deliver much needed medical care into the veterans’ home.
COPD Telehealth Program
In this article, we describe the processes for developing and delivering an in-home, interactive, supervised PR program for veterans with severe COPD through VA telehealth service. The program consists of 18 sessions delivered over 6 weeks by a licensed physical therapist (PT) and a respiratory therapist (RT). The aims of the telehealth PR are to improve exercise tolerance, reduce dyspnea and fatigue, improve QOL, improve accessibility, and decrease costs and transportation burdens for patients with COPD. The program was developed, implemented and delivered by an interdisciplinary team, including a pulmonologist, PT, RT, physiatrist, and nonclinical supporting staff.
Patient Assessment
To be eligible to participate in the program the patient must: (1) have a forced expiratory volume (FEV1) < 60%; ( 2) be medically stable and be receiving optimal medical management; (3) have no severe cognitive impairments; (4) be able to use a computer and e-mail; (5) be able to ambulate with or without a walking device; (6) be willing to enroll in a smoking cessation program or to stop smoking; (7) be willing to participate without prolonged interruption; and (8) have all visual and auditory impairments corrected with medical devices.
After referral and enrollment, patients receive medical and physical examinations by the PR team, including a pulmonologist, a PT, and a RT, to ensure that the patients are medically stable to undergo rehabilitation and to develop a tailored exercise program while being mindful of the comorbidities, limitations, and precautions, (eg, loss of balance, risk of fall, limited range of motion). The preprogram assessment includes a pulmonary function test, arterial blood gas test, Montreal Cognitive Assessment, Modified Medical Research Council Scale, St. George Respiratory Questionnaire, the COPD Assessment Test, Patient Health Questionnaire-9,Generalized Anxiety Disorder Assessment-7, Epworth Sleepiness Scale, Katz Index of Independence of Activities of Daily Living, medications and inhaler use, oxygen use, breathing pattern, coughing, 6-minute walk test, Modified Borg Dyspnea Scale, grip strength, 5 Times Sit to Stand Test, manual muscle test, gait measure, Timed Up & Go test, clinical balance tests, range of motion, flexibility, sensation, pain, and fall history.18-32 Educational needs (eg, respiratory hygiene, nutrition, infection control, sleep, disease/symptom management) also are evaluated.
This thorough assessment is performed in a face-to-face outpatient visit. During the program participation, a physiatrist may be consulted for additional needs (eg, wheelchair assessment, home safety evaluation/ modifications, and mobility/disability issues). After completing the 6-week program, patients are scheduled for the postprogram evaluation in a face-to-face outpatient visit with the clinicians.
Equipment
Both clinician and the patient are equipped with a computer with Wi-Fi connectivity, a webcam, and a microphone. Patients are provided an exercise pictorial booklet, an exercise compact disk (audio and video), small exercise apparatuses (eg, assorted colors of resistance bands, hand grip exerciser, hand putty, ergometer, harmonica, and pedometer), incentive spirometer, pulse oximeter, cough assistive device (as needed), blood pressure monitor, COPD information booklets, and a diary to use at home during the program.33
Technology Preparation
Prior to starting the telehealth program, the patient is contacted 1 or 2 days before the first session for technical preparation and familiarization of the VA telehealth connection process. Either the PT or RT provides step-by-step instructions for the patient to practice connecting through VVC during this preparatory phone call. The patient also practices using the computer webcam, speaker, and microphone; checks the telehealth scheduling e-mail; and learns how to solve possible common technical issues (eg, adjusting volume and position of webcam). The patient is asked to set up a table close to the computer and to place all exercise apparatuses and respiratory devices on the table surface.
Program Delivery
A secure online VVC is used for connection during the telehealth session. The patient received an e-mail from the telehealth scheduling system with a link for VVC before each session. During the 6-week program, each telehealth session is conducted by a PT and a RT concurrently for 120 minutes, 3 days per week. The PT provides exercises for the patient to attempt, and the RT provides breathing training and monitoring during the session. After a successful connection to VVC, the therapist verifies the patient’s identity and confirms patient consent for the telehealth session.
After this check-in process, the patient performs a self-measure of resting blood pressure (BP), heart rate, respiratory rate, and blood oxygen saturation and reports to the therapists. During the exercise session, fatigue/exertion, dyspnea (Modified Borg Dyspnea Scale; Borg CR10 Scale), BP, heart rate, oxygen saturation, and other clinical symptoms and responses to exercise are monitored by the therapists, using both patient-reported measures and clinical observation by the therapists.34,35 Any medical emergency during the session is reported immediately to the pulmonologist for further management.
Structure
Prior to each exercise session, exercise precautions, fall prevention, good posture, pursed-lip breathing, pacing, and coordinated breathing are discussed with the patient. The PT demonstrates stretching and warm-up exercises in front of the webcam for the patient to follow. Then the patient performs all exercises in view of the webcam during the session (Figure 1). A RT monitors breathing patterns and corrects with verbal instructions if not properly performed.
Loss of skeletal muscle mass and cachexia are highly prevalent comorbidities of COPD and have been associated with breathlessness, functional limitation, and poor prognosis.36 To address these comorbidities, our program consists of progressive strengthening, aerobic, balance, and flexibility exercises. Resistance bands and tubes are used for strengthening exercises. Callisthenic exercises (eg, chair squat, chair stand, knee marching, bridging, single limb stances, and lunge) are used for progressive strengthening and balance exercises. Progression of strengthening and balance exercises are done through increasing the volume of exercise (ie, numbers of sets and repetitions) and increased load and level of difficulty based on the patient’s progress and comorbidity. The exercise program focuses on strengthening muscles, especially large muscle groups, to improve overall muscle strength and performance of functional activities.37
Arm/pedal ergometer and daily walking are used for daily aerobic exercise. In a study of patients with COPD by the PAC-COPD Study Group, step counter use was found to increase physical activity and improve exercise capacity, which supports its use in COPD management.38 During program participation, the patient is asked to wear a pedometer to monitor the number of steps taken per day and to report step data to the therapists during the telehealth session. The pedometer stores the previous 41 calendar days of data and displays the most recent 7 calendar days of data.
The patient is encouraged to set a realistic daily step goal. The general program goal is to increase at least 1000 steps per day. However, this goal can be adjusted depending on the patient’s health status and comorbid conditions. The PAC-COPD Study Group found that for every additional 1000 daily steps at low intensity, COPD hospitalization risk decreased by 20%.39 A magnitude of 2000 steps or about 1 mile of walking per day was found to be associated with increased physical activity and health benefits in the general population.40
Respiratory muscle training and breathing exercise are provided by the RT, using breathing and incentive spirometer techniques (Figure 2). Huff coughing, diaphragmatic deep breathing, and pursed-lip breathing are instructed by the RT during the session. Effective coughing technique with a cough assistive device is also provided during breathing training if needed.
Patient Education
In patients with COPD, there are numerous positive health benefits associated with education, including assisting the patients to become active participants in the PR program leading to satisfying outcomes; assisting the patients to better understand the lung health, disease processes, physical and psychological changes that occur with COPD; assisting the patients to explore coping strategies for those changes; building lifelong behavioral changes; and developing the self-management skills for sustainability. Through the educational process, patients with COPD can become more skilled at collaborative self-management and improve adherence to their treatment plan, which in turn can result in a reduction in hospital admissions and reduced health care costs.8,41
Education is provided with every session after the patient completes the exercise. Patients are required to record their COPD symptoms, daily activity, home exercise program, sleep, food intake, and additional physical or social activity in their COPD diary and to report during the session (Figure 3). A COPD diary assists patients in self-monitoring their COPD symptoms and provides the therapists with information about clinical changes, behavioral changes, and/or specific unmet needs for education. Several topics related to COPD are included in the education session: lung or respiratory disease/condition and self-management; smoking cessation; physical activity; energy-conserving techniques; breathing and coughing techniques; smoking cessation; nutrition/healthy eating and weight counseling; sex and intimacy; psychological counseling and/or group support; emergency planning (eg, medical, travel, and inclement weather); correct use of inhaler and medications; home oxygen; sleep and sleep hygiene; palliative care and advanced directive; infection control; and sputum clearance.42,43
Program Maintenance
After successfully completing the 6-week program, patients are referred to the VA TeleMOVE! Program or MOVE! Weight Management Program for continuous, long-term management of weight, nutrition, physical activity/exercise, and social activity needs or goals. The patients are scheduled for monthly follow-up phone visits for 6 months with the telerehabilitation team for enforcing sustainability. The phone call visit consists of reviewing breathing techniques, exercise program, physical activity, education, encouragement, and addressing any issues that arise during the self-maintained period.
Limitations
There are several issues of concern and precautions when delivering PR through telehealth into the home. First, the patient performs exercises independently without being manually guarded by the therapists. Risk of falls are a major concern due to impaired balance, poor vision, and other possible unusual physiologic responses to exercise (eg, drop in BP, dizziness, loss of balance). The area in front of the computer needs to be cleared of fall hazards (ie, area rug, wires, objects on the floor). The patient also needs to be educated on self-measurements of BP and oxygen saturation and reports to the therapists. The therapists provide detailed instructions on how to obtain these measures correctly; otherwise, the values may not be valid for a clinical judgment during the exercise session or for other clinical management. In a home environment, there is a limited use of exercise apparatuses. For this program, we only used resistance bands/tubes, small arm/leg ergometer, hand grip, and hand putty for the exercise program. We feel that dumbbell and weight plates are not suitable due to a possible risk of injury if the patient accidently drops them.
Advanced balance training is not suitable due to an increased risk for falls. Without the presence of the PT, level of challenge/difficulty is somewhat limited for this telehealth supervision exercise program. In addition, visual and audio quality are necessary for the session. The patient and the therapists need to see each other clearly to ensure correct methods and forms of each exercise. Furthermore, rehearsal of technical skills with the therapists is very important because this population is older and often has limited computer skills. Any technical difficulty or failure can lead to undesirable situations (eg, anxiety episodes, worries, shortness of breath, upset), which compromise exercise performance during the session. Finally, a phone is needed as an alternative in case of a poor VVC connection.
Conclusion
COPD symptoms and complications greatly affect patients’ ability to perform daily activities, decrease QOL and functional ability, and result in extensive use of health services. Many patients have limited access to a PR program at hospitals or rehabilitation centers due to health conditions, lack of transportation, and/or family support. This home-based, interactive telehealth PR program can break down the geographic barriers, solve poor program accessibility, potentially increase the utilization of PR, and reduce the cost and travel required by the patients.
Acknowledgments
The Telehealth Pulmonary Rehabilitation Program was originally funded by the Veterans Health Administration VA ACCESS Program (AS, CL, HKH). We thank all the veterans for their time and effort in participating in this newly developed rehabilitation program.
1. World Health Organization. Chronic obstructive pulmonary disease (COPD). http://www.who.int/news-room/fact-sheets/detail/chronic-obstructive-pulmonary-disease-(copd). Published December 1, 2017. Accessed August 7, 2019.
2. Yu W, Ravelo A, Wagner TH, et al. Prevalence and costs of chronic conditions in the VA health care system. Med Care Res Rev. 2003;60(suppl 3):146S-167S.
3. Doney B, Hnizdo E, Dillon CF, et al. Prevalence of airflow obstruction in U.S. adults aged 40-79 years: NHANES data 1988-1994 and 2007-2010. COPD. 2015;12(4):355-365.
4. Murphy DE, Chaudhry Z, Almoosa KF, Panos RJ. High prevalence of chronic obstructive pulmonary disease among veterans in the urban midwest. Mil Med. 2011;176(5):552-560.
5. Cypel YS, Hines SE, Davey VJ, Eber SM, Schneiderman AI. Self-reported physician-diagnosed chronic obstructive pulmonary disease and spirometry patterns in Vietnam Era US Army Chemical Corps veterans: a retrospective cohort study. Am J Ind Med. 2018;61(10):802-814.
6. Rochester CL. Exercise training in chronic obstructive pulmonary disease. J Rehabil Res Dev. 2003;40(5)(suppl 2):59-80.
7. Cortopassi F, Gurung P, Pinto-Plata V. Chronic obstructive pulmonary disease in elderly patients. Clin Geriatr Med. 2017;33(4):539-552.
8. Spruit MA, Singh SJ, Garvey C, et al; ATS/ERS Task Force on Pulmonary Rehabilitation. An official American Thoracic Society/European Respiratory Society statement: key concepts and advances in pulmonary rehabilitation. Am J Respir Crit Care Med. 2013;188(8):e13-e64.
9. Robinson H, Williams V, Curtis F, Bridle C, Jones AW. Facilitators and barriers to physical activity following pulmonary rehabilitation in COPD: a systematic review of qualitative studies. NPJ Prim Care Respir Med. 2018;28(1):19.
10. McCarthy B, Casey D, Devane D, Murphy K, Murphy E, Lacasse Y. Pulmonary rehabilitation for chronic obstructive pulmonary disease. Cochrane Database Syst Rev. 2015;(2):CD003793.
11. Ries AL, Bauldoff GS, Carlin BW, et al. Pulmonary rehabilitation: joint AACP/AACVPR evidence-based clinical practice guidelines. Chest. 2007;131(suppl 5):4S-42S.
12. Major S, Moreno M, Shelton J, Panos RJ. Veterans with chronic obstructive pulmonary disease achieve clinically relevant improvements in respiratory health after pulmonary rehabilitation. J Cardiopulm Rehabil Prev. 2014;34(6):420-429.
13. Liu Y, Dickerson T, Early F, Fuld J, Clarkson PJ. Understanding influences on the uptake of pulmonary rehabilitation in the East of England: an inclusive design/mixed methods study protocol. BMJ Open. 2018;8(4):e020750.
14. Harris D, Hayter M, Allender S. Factors affecting the offer of pulmonary rehabilitation to patients with chronic obstructive pulmonary disease by primary care professionals: a qualitative study. Prim Health Care Res Dev. 2008;9(4):280-290.
15. Mathar H, Fastholm P, Hansen IR, Larsen NS. Why do patients with COPD decline rehabilitation. Scand J Caring Sci. 2016;30(3):432-441.
16. Han MK, Martinez CH, Au DH, et al. Meeting the challenge of COPD care delivery in the USA: a multiprovider perspective. Lancet Respir Med. 2016;4(6):473-526.
17. American Association of Cardiovascular and Pulmonary Rehabilitation (AACVPR). Online searchable program directory. https://www.aacvpr.org/Resources/Program-Directory Accessed July 19, 2018.
18. Nasreddine ZS, Phillips NA, Bédirian V, et al. The Montreal Cognitive Assessment, MoCA: a brief screening tool for mild cognitive impairment. J Am Geriatr Soc. 2005;53(4):695-699.
19. Fletcher CM, Elmes PC, Fairbairn AS, Wood CH. The significance of respiratory symptoms and the diagnosis of chronic bronchitis in a working population. Br Med J. 1959;2(5147):257-266.
20. O’Donnell DE, Aaron S, Bourbeau J, et al. Canadian Thoracic Society recommendations for management of chronic obstructive pulmonary disease—2007 update. Can Respir J. 2007;14(suppl B):5B-32B.
21. Jones PW, Quirk FH, Baveystock CM. The St George’s Respiratory Questionnaire. Respir Med. 1991;85(suppl B):25-31.
22. Jones PW, Harding G, Berry P, Wiklund I, Chen WH, Kline Leidy N. Development and first validation of the COPD Assessment Test. Eur Respir J. 2009;34(3):648-654.
23. Kroenke K, Spitzer RL, Williams JB. The PHQ-9: validity of a brief depression severity measure. J Gen Intern Med. 2001;16(9):606-613.
24. Spitzer RL, Kroenke K, Williams JBW, Löwe B. A brief measure for assessing generalized anxiety disorder: the GAD-7. Arch Intern Med. 2006;166(10):1092-1097.
25. Johns MW. A new method for measuring daytime sleepiness: the Epworth sleepiness scale. Sleep. 1991;14(6):540-545.
26. Katz S. Assessing self-maintenance: activities of daily living, mobility and instrumental activities of daily living. J Am Geriatr Soc. 1983;31(12):721-727.
27. Holland AE, Spruit MA, Troosters T, et al. An official European Respiratory Society/American Thoracic Society technical standard: field walking tests in chronic respiratory disease. Eur Respir J. 2014;44(6):1428-1446.
28. Mahler DA, Horowitz MB. Perception of breathlessness during exercise in patients with respiratory disease. Med Sci Sports Exerc. 1994;26(9):1078-1081.
29. Liao WC, Wang CH, Yu SY, Chen LY, Wang CY. Grip strength measurement in older adults in Taiwan: a comparison of three testing positions. Australas J Ageing. 2014;33(4):278-282.
30. Buatois S, Miljkovic D, Manckoundia P, et al. Five times sit to stand test is a predictor of recurrent falls in healthy community-living subjects aged 65 and older. J Am Geriatr Soc. 2008;56(8):1575-1577.
31. Bryant MS, Workman CD, Jackson GR. Multidirectional walk test in persons with Parkinson’s disease: a validity study. Int J Rehabil Res. 2015;38(1):88-91.
32. Podsiadlo D, Richardson S. The timed “Up & Go”: a test of basic functional mobility for frail elderly persons. J Am Geriatr Soc. 1991;39(2):142-148.
33. University of Nebraska Medical Center. Timed Up and Go (TUG) Test. https://www.unmc.edu/media/intmed/geriatrics/nebgec/pdf/frailelderlyjuly09/toolkits/timedupandgo_w_norms.pdf. Accessed August 13, 2019.
34. Borg GA. Psychophysical bases of perceived exertion. Med Sci Sports Exerc. 1982;14(5):377-381.
35. Mahler DA, Horowitz MB. Clinical evaluation of exertional dyspnea. Clin Chest Med. 1994;15(2):259-269.
36. Dudgeon D, Baracos VE. Physiological and functional failure in chronic obstructive pulmonary disease, congestive heart failure and cancer: a debilitating intersection of sarcopenia, cachexia and breathlessness. Curr Opin Support Palliat Care. 2016;10(3):236-241.
37. Lee AL, Holland AE. Time to adapt exercise training regimens in pulmonary rehabilitation—a review of the literature. Int J Chron Obstruct Pulmon Dis. 2014;9:1275-1288.
38. Qiu S, Cai X, Wang X, et al. Using step counters to promote physical activity and exercise capacity in patients with chronic obstructive pulmonary disease: a meta-analysis. Ther Adv Respir Dis. 2018;12:1753466618787386.
39. Donaire-Gonzalez D, Gimeno-Santos E, Balcells E, et al; PAC-COPD Study Group. Benefits of physical activity on COPD hospitalization depend on intensity. Eur Respir J. 2015;46(5):1281-1289.
40. Bravata DM, Smith-Spangler C, Sundaram V, et al. Using pedometers to increase physical activity and improve health: a systematic review. JAMA. 2007;298(19):2296-2304.
41. Zwerink M, Brusse-Keizer M, van der Valk PD, et al. Self-management for patients with chronic obstructive pulmonary disease. Cochrane Database Syst Rev. 2014;19(3):CD002990.
42. Wilson JS, O’Neill B, Reilly J, MacMahon J, Bradley JM. Education in pulmonary rehabilitation: the patient’s perspective. Arch Phys Med Rehabil. 2007;88(12):1704-1709.
43. Bourbeau J, Nault D, Dang-Tan T. Self-management and behaviour modification in COPD. Patient Educ Couns. 2004;52(3):271-277.
1. World Health Organization. Chronic obstructive pulmonary disease (COPD). http://www.who.int/news-room/fact-sheets/detail/chronic-obstructive-pulmonary-disease-(copd). Published December 1, 2017. Accessed August 7, 2019.
2. Yu W, Ravelo A, Wagner TH, et al. Prevalence and costs of chronic conditions in the VA health care system. Med Care Res Rev. 2003;60(suppl 3):146S-167S.
3. Doney B, Hnizdo E, Dillon CF, et al. Prevalence of airflow obstruction in U.S. adults aged 40-79 years: NHANES data 1988-1994 and 2007-2010. COPD. 2015;12(4):355-365.
4. Murphy DE, Chaudhry Z, Almoosa KF, Panos RJ. High prevalence of chronic obstructive pulmonary disease among veterans in the urban midwest. Mil Med. 2011;176(5):552-560.
5. Cypel YS, Hines SE, Davey VJ, Eber SM, Schneiderman AI. Self-reported physician-diagnosed chronic obstructive pulmonary disease and spirometry patterns in Vietnam Era US Army Chemical Corps veterans: a retrospective cohort study. Am J Ind Med. 2018;61(10):802-814.
6. Rochester CL. Exercise training in chronic obstructive pulmonary disease. J Rehabil Res Dev. 2003;40(5)(suppl 2):59-80.
7. Cortopassi F, Gurung P, Pinto-Plata V. Chronic obstructive pulmonary disease in elderly patients. Clin Geriatr Med. 2017;33(4):539-552.
8. Spruit MA, Singh SJ, Garvey C, et al; ATS/ERS Task Force on Pulmonary Rehabilitation. An official American Thoracic Society/European Respiratory Society statement: key concepts and advances in pulmonary rehabilitation. Am J Respir Crit Care Med. 2013;188(8):e13-e64.
9. Robinson H, Williams V, Curtis F, Bridle C, Jones AW. Facilitators and barriers to physical activity following pulmonary rehabilitation in COPD: a systematic review of qualitative studies. NPJ Prim Care Respir Med. 2018;28(1):19.
10. McCarthy B, Casey D, Devane D, Murphy K, Murphy E, Lacasse Y. Pulmonary rehabilitation for chronic obstructive pulmonary disease. Cochrane Database Syst Rev. 2015;(2):CD003793.
11. Ries AL, Bauldoff GS, Carlin BW, et al. Pulmonary rehabilitation: joint AACP/AACVPR evidence-based clinical practice guidelines. Chest. 2007;131(suppl 5):4S-42S.
12. Major S, Moreno M, Shelton J, Panos RJ. Veterans with chronic obstructive pulmonary disease achieve clinically relevant improvements in respiratory health after pulmonary rehabilitation. J Cardiopulm Rehabil Prev. 2014;34(6):420-429.
13. Liu Y, Dickerson T, Early F, Fuld J, Clarkson PJ. Understanding influences on the uptake of pulmonary rehabilitation in the East of England: an inclusive design/mixed methods study protocol. BMJ Open. 2018;8(4):e020750.
14. Harris D, Hayter M, Allender S. Factors affecting the offer of pulmonary rehabilitation to patients with chronic obstructive pulmonary disease by primary care professionals: a qualitative study. Prim Health Care Res Dev. 2008;9(4):280-290.
15. Mathar H, Fastholm P, Hansen IR, Larsen NS. Why do patients with COPD decline rehabilitation. Scand J Caring Sci. 2016;30(3):432-441.
16. Han MK, Martinez CH, Au DH, et al. Meeting the challenge of COPD care delivery in the USA: a multiprovider perspective. Lancet Respir Med. 2016;4(6):473-526.
17. American Association of Cardiovascular and Pulmonary Rehabilitation (AACVPR). Online searchable program directory. https://www.aacvpr.org/Resources/Program-Directory Accessed July 19, 2018.
18. Nasreddine ZS, Phillips NA, Bédirian V, et al. The Montreal Cognitive Assessment, MoCA: a brief screening tool for mild cognitive impairment. J Am Geriatr Soc. 2005;53(4):695-699.
19. Fletcher CM, Elmes PC, Fairbairn AS, Wood CH. The significance of respiratory symptoms and the diagnosis of chronic bronchitis in a working population. Br Med J. 1959;2(5147):257-266.
20. O’Donnell DE, Aaron S, Bourbeau J, et al. Canadian Thoracic Society recommendations for management of chronic obstructive pulmonary disease—2007 update. Can Respir J. 2007;14(suppl B):5B-32B.
21. Jones PW, Quirk FH, Baveystock CM. The St George’s Respiratory Questionnaire. Respir Med. 1991;85(suppl B):25-31.
22. Jones PW, Harding G, Berry P, Wiklund I, Chen WH, Kline Leidy N. Development and first validation of the COPD Assessment Test. Eur Respir J. 2009;34(3):648-654.
23. Kroenke K, Spitzer RL, Williams JB. The PHQ-9: validity of a brief depression severity measure. J Gen Intern Med. 2001;16(9):606-613.
24. Spitzer RL, Kroenke K, Williams JBW, Löwe B. A brief measure for assessing generalized anxiety disorder: the GAD-7. Arch Intern Med. 2006;166(10):1092-1097.
25. Johns MW. A new method for measuring daytime sleepiness: the Epworth sleepiness scale. Sleep. 1991;14(6):540-545.
26. Katz S. Assessing self-maintenance: activities of daily living, mobility and instrumental activities of daily living. J Am Geriatr Soc. 1983;31(12):721-727.
27. Holland AE, Spruit MA, Troosters T, et al. An official European Respiratory Society/American Thoracic Society technical standard: field walking tests in chronic respiratory disease. Eur Respir J. 2014;44(6):1428-1446.
28. Mahler DA, Horowitz MB. Perception of breathlessness during exercise in patients with respiratory disease. Med Sci Sports Exerc. 1994;26(9):1078-1081.
29. Liao WC, Wang CH, Yu SY, Chen LY, Wang CY. Grip strength measurement in older adults in Taiwan: a comparison of three testing positions. Australas J Ageing. 2014;33(4):278-282.
30. Buatois S, Miljkovic D, Manckoundia P, et al. Five times sit to stand test is a predictor of recurrent falls in healthy community-living subjects aged 65 and older. J Am Geriatr Soc. 2008;56(8):1575-1577.
31. Bryant MS, Workman CD, Jackson GR. Multidirectional walk test in persons with Parkinson’s disease: a validity study. Int J Rehabil Res. 2015;38(1):88-91.
32. Podsiadlo D, Richardson S. The timed “Up & Go”: a test of basic functional mobility for frail elderly persons. J Am Geriatr Soc. 1991;39(2):142-148.
33. University of Nebraska Medical Center. Timed Up and Go (TUG) Test. https://www.unmc.edu/media/intmed/geriatrics/nebgec/pdf/frailelderlyjuly09/toolkits/timedupandgo_w_norms.pdf. Accessed August 13, 2019.
34. Borg GA. Psychophysical bases of perceived exertion. Med Sci Sports Exerc. 1982;14(5):377-381.
35. Mahler DA, Horowitz MB. Clinical evaluation of exertional dyspnea. Clin Chest Med. 1994;15(2):259-269.
36. Dudgeon D, Baracos VE. Physiological and functional failure in chronic obstructive pulmonary disease, congestive heart failure and cancer: a debilitating intersection of sarcopenia, cachexia and breathlessness. Curr Opin Support Palliat Care. 2016;10(3):236-241.
37. Lee AL, Holland AE. Time to adapt exercise training regimens in pulmonary rehabilitation—a review of the literature. Int J Chron Obstruct Pulmon Dis. 2014;9:1275-1288.
38. Qiu S, Cai X, Wang X, et al. Using step counters to promote physical activity and exercise capacity in patients with chronic obstructive pulmonary disease: a meta-analysis. Ther Adv Respir Dis. 2018;12:1753466618787386.
39. Donaire-Gonzalez D, Gimeno-Santos E, Balcells E, et al; PAC-COPD Study Group. Benefits of physical activity on COPD hospitalization depend on intensity. Eur Respir J. 2015;46(5):1281-1289.
40. Bravata DM, Smith-Spangler C, Sundaram V, et al. Using pedometers to increase physical activity and improve health: a systematic review. JAMA. 2007;298(19):2296-2304.
41. Zwerink M, Brusse-Keizer M, van der Valk PD, et al. Self-management for patients with chronic obstructive pulmonary disease. Cochrane Database Syst Rev. 2014;19(3):CD002990.
42. Wilson JS, O’Neill B, Reilly J, MacMahon J, Bradley JM. Education in pulmonary rehabilitation: the patient’s perspective. Arch Phys Med Rehabil. 2007;88(12):1704-1709.
43. Bourbeau J, Nault D, Dang-Tan T. Self-management and behaviour modification in COPD. Patient Educ Couns. 2004;52(3):271-277.
Addressing the Shortage of Physician Assistants in Medicine Clerkship Sites
The Federal Bureau of Labor Statistics projects 37% job growth for physician assistants (PAs) from 2016 to 2026, much greater than the average for all other occupations as well as for other medical professions.1 This growth has been accompanied by increased enrollment in medical (doctor of medicine [MD], doctor of osteopathic medicine) and nurse practitioner (NP) schools.2 Clinical teaching sites serve a crucial function in the training of all clinical disciplines. These sites provide hands-on and experiential learning in medical settings, necessary components for learners practicing to become clinicians. Significant PA program expansion has led to increased demand for clinical training, creating competition for sites and a shortage of willing and well-trained preceptors.3
This challenge has been recognized by PA program directors. In the Joint Report of the 2013 Multi-Discipline Clerkship/Clinical Training Site Survey, PA program directors expressed concern about the adequacy of clinical opportunities for students, increased difficulty developing new core sites, and preserving existing core sites. In addition, they noted that a shortage of clinical sites was one of the greatest barriers to the PA programs’ sustained growth and success.4
Program directors also indicated difficulty securing clinical training sites in internal medicine (IM) and high rates of attrition of medicine clinical preceptors for their students.5 The reasons are multifold: increasing clinical demands, time, teaching competence, lack of experience, academic affiliation, lack of reimbursement, or compensation. Moreover, there is a declining number of PAs who work in primary care compared with specialty and subspecialty care, limiting the availability of clinical training preceptors in medicine and primary care.6-8 According to the American Academy of PAs (AAPA) census and salary survey data, the percentage of PAs working in the primary care specialties (ie, family medicine, IM, and general pediatrics) has decreased from > 47% in 1995 to 24% in 2017.9 As such, there is a need to broaden the educational landscape to provide more high-quality training sites in IM.
The postacute health care setting may address this training need. It offers a unique clinical opportunity to expose learners to a broad range of disease complexity and clinical acuity, as the percentage of patients discharged from hospitals to postacute care (PAC) has increased and care shifts from the hospital to the PAC setting.10,11 The longer PAC length of stay also enables learners to follow patients longitudinally over several weeks and experience interprofessional team-based care. In addition, the PAC setting offers learners the ability to acquire the necessary skills for smooth and effective transitions of care. This setting has been extensively used for trainees of nursing, pharmacy, physical therapy (PT) and occupational therapy (OT), speech-language pathology, psychology, and social work (SW), but few programs have used the PAC setting as clerkship sites for IM rotations for PA students. To address this need for IM sites, the VA Boston Healthcare System (VABHS), in conjunction with the Boston University School of Medicine Physician Assistant Program, developed a novel medicine clinical clerkship site for physician assistants in the PAC unit of the community living center (CLC) at VABHS. This report describes the program structure, curriculum, and participant evaluation results.
Clinical Clerkship Program
VABHS CLC is a 110-bed facility comprising 3 units: a 65-bed PAC unit, a 15-bed closed hospice/palliative care unit, and a 30-bed long-term care unit. The service is staffed continuously with physicians, PAs, and NPs. A majority of patients are admitted from the acute care hospital of VABHS (West Roxbury campus) and other regional VA facilities. The CLC offers dynamic services, including phlebotomy, general radiology, IV diuretics and antibiotics, wound care, and subacute PT, OT, and speech-language pathology rehabilitation. The CLC serves as a venue for transitioning patients from acute inpatient care to home. The patient population is often elderly, with multiple active comorbidities and variable medical literacy, adherence, and follow-up.
The CLC provides a diverse interprofessional learning environment, offering core IM rotations for first-year psychiatry residents, oral and maxillofacial surgery residents, and PA students. The CLC also has expanded as a clinical site both for transitions-in-care IM resident curricula and electives as well as a geriatrics fellowship. In addition, the site offers rotations for NPs, nursing, pharmacy, physical and occupational therapies, speech-language pathology, psychology, and SW.
The Boston University School of Medicine Physician Assistant Program was founded in 2015 as a master’s degree program completed over 28 months. The first 12 months are didactic, and the following 16 months are clinical training with 14 months of rotations (2 IM, family medicine, pediatrics, emergency medicine, general surgery, obstetrics and gynecology, psychiatry, neurology, and 5 elective rotations), and 2 months for a thesis. The program has about 30 students per year and 4 clerkship sites for IM.
Program Description
The VABHS medicine clerkship hosts 1 to 2 PA students for 4-week blocks in the PAC unit of the CLC. Each student rotates on both PA and MD teams. Students follow 3 to 4 patients and participate fully in their care from admission to discharge; they prepare daily presentations and participate in medical management, family meetings, chart documentation, and care coordination with the interprofessional team. Students are provided a physical examination checklist and feedback form, and they are expected to track findings and record feedback and goals with their supervising preceptor weekly. They also make formal case presentations and participate in monthly medicine didactic rounds available to all VABHS IM students and trainees via videoconference.
In addition, beginning in July 2017, all PA students in the CLC began to participate in a 4-week Interprofessional Curriculum in Transitional Care. The curriculum includes 14 didactic lectures taught by 16 interprofessional faculty, including medicine, geriatric, and palliative care physicians; PAs; social workers; physical and occupational therapists; pharmacists; and a geriatric psychologist. The didactics include topics on the interprofessional team, the care continuum, teams and teamwork, interdisciplinary coordination of care, components of effective transitions in care, medication reconciliation, approaching difficult conversations, advance care planning, and quality improvement. The goal of the curriculum is to provide learners the knowledge, skills, and dispositions necessary for high-quality transitional care and interprofessional practice as well as specific training for effective and safe transfers of care between clinical settings. Although PA students are the main participants in this curriculum, all other learners in the PAC unit are also invited to attend the lectures.
The unique attributes of this training site include direct interaction with supervising PAs and physicians, rather than experiencing the traditional teaching hierarchy (with interns, residents, fellows); observation of the natural progression of disease of both acute care and primary care issues due to the longer length of stay (2 to 6 weeks, where the typical student will see the same patient 7 to 10 times during their rotation); exposure to a host of medically complex patients offering a multitude of clinical scenarios and abnormal physical exam findings; exposure to a hospice/palliative care ward and end-of-life care; and interaction within an interprofessional training environment of nursing, pharmacy, PT, OT, speech-language pathology, psychology, and SW trainees.
Program Evaluation
At the end of rotations continuously through the year, PA students electronically complete a site evaluation from the Boston University School of Medicine Physician Assistant Program. The evaluation consists of 14 questions: 6 about site quality and 8 about instruction quality. The questions are answered on a 5-point Likert scale. Also included are 2 open-ended response questions that ask what they liked about the rotation and what they felt could be improved. Results are anonymous, de-identified and blinded both to the program as well as the clerkship site. Results are aggregated and provided to program sites annually. Responses are converted to a dichotomous variable, where any good or excellent response (4 or 5) is considered positive and any neutral or below (3, 2, 1) is considered a nonpositive response.
Results
The clerkship site has been operational since June 22, 2015. There have been 59 students who participated in the rotation. A different scale in these evaluations was used between June 22, 2015, and September 13, 2015. Therefore, 7 responses were excluded from the analysis, leaving 52 usable evaluations. The responses were analyzed both in total (for the CLC as well as other IM rotation sites) and by individual clerkship year to look for any trends over time: September 14, 2015, through April 24, 2016; April 25, 2016, through April 28, 2017; and May 1, 2017, through March 1, 2018 (Table).
Site evaluations showed high satisfaction regarding the quality of the physical environment as well as the learning environment. Students endorsed the PAC unit having resources and physical space for them, such as a desk and computer, opportunity for participation in patient care, and parking (100%; n = 52). Site evaluations revealed high satisfaction with the quality of teaching and faculty encouragement and support of their learning (100%; n = 52). The evaluations revealed that bedside teaching was strong (94%; n = 49). The students reported high satisfaction with the volume of patients provided (92%; n = 48) as well as the diversity of diagnoses (92%; n = 48).
There were fewer positive responses in the first 2 years of the rotation with regard to formal lectures (50% and 67%; 7/14 and 16/24, respectively). In the third year of the rotation, students had a much higher satisfaction rate (93%; 13/14). This increased satisfaction was associated with the development and incorporation of the Interprofessional Curriculum in Transitional Care in 2017.
Discussion
Access to high-quality PA student clerkship sites has become a pressing issue in recent years because of increased competition for sites and a shortage of willing and well-trained preceptors. There has been marked growth in schools and enrollment across all medical professions. The Accreditation Review Commission on Education for the PA (ARC-PA) reported that the total number of accredited entry-level PA programs in 2018 was 246, with 58 new accredited programs projected by 2022.12 The Joint Report of the 2013 Multi-Discipline Clerkship/Clinical Training Site Survey reported a 66% increase in first-year enrollment in PA programs from 2002 to 2012.5 Programs must implement alternative strategies to attract clinical sites (eg, academic appointments, increased clinical resources to training sites) or face continued challenges with recruiting training sites for their students. Postacute care may be a natural extension to expand the footprint for clinical sites for these programs, augmenting acute inpatient and outpatient rotations. This implementation would increase the pool of clinical training sites and preceptors.
The experience with this novel training site, based on PA student feedback and evaluations, has been positive, and the postacute setting can provide students with high-quality IM clinical experiences. Students report adequate patient volume and diversity. In addition, evaluations are comparable with that of other IM site rotations the students experience. Qualitative feedback has emphasized the value of following patients over longer periods; eg, weeks vs days (as in acute care) enabling students to build relationships with patients as well as observe a richer clinical spectrum of disease over a less compressed period. “Patients have complex issues, so from a medical standpoint it challenges you to think of new ways to manage their care,” commented a representative student. “It is really beneficial that you can follow them over time.”
Furthermore, in response to student feedback on didactics, an interprofessional curriculum was developed to add formal structure as well as to create a curriculum in care transitions. This curriculum provided a unique opportunity for PA students to receive formal instruction on areas of particular relevance for transitional care (eg, care continuum, end of life issues, and care transitions). The curriculum also allows the interprofessional faculty a unique and enjoyable opportunity for interprofessional collaboration.
The 1 month PAC rotation is augmented with inpatient IM and outpatient family medicine rotations, consequently giving exposure to the full continuum of care. The PAC setting provides learners multifaceted benefits: the opportunity to strengthen and develop the knowledge, attitudes, and skills necessary for IM; increased understanding of other professions by observing and interacting as a team caring for a patient over a longer period as opposed to the acute care setting; the ability to perform effective, efficient, and safe transfer between clinical settings; and broad exposure to transitional care. As a result, the PAC rotation enhances but does not replace the necessary and essential rotations of inpatient and outpatient medicine.
Moreover, this rotation provides unique and core IM training for PA students. Our site focuses on interprofessional collaboration, emphasizing the importance of team-based care, an essential concept in modern day medicine. Formal exposure to other care specialties, such as PT and OT, SW, and mental health, is essential for students to appreciate clinical medicine and a patient’s physical and mental experience over the course of a disease and clinical state. In addition, the physical exam checklist ensures that students are exposed to the full spectrum of IM examination findings during their rotation. Finally, weekly feedback forms require students to ask and receive concrete feedback from their supervising providers.
Limitations
The generalizability of this model requires careful consideration. VABHS is a tertiary care integrated health care system, enabling students to learn from patients moving through multiple care transitions in a single health care system. In addition, other settings may not have the staffing or clinical volume to sustain such a model. All PAC clinical faculty teach voluntarily, and local leadership has set expectations for all clinicians to participate in teaching of trainees and PA students. Evaluations also note less diversity in the patient population, a challenge that some VA facilities face. This issue could be addressed by ensuring that students also have IM rotations at other inpatient medical facilities. A more balanced experience, where students reap the positive benefits of PAC but do not lose exposure to a diverse patient pool, could result. Furthermore, some of the perceived positive impacts also may be related to professional and personal attributes of the teaching clinicians rather than to the PAC setting.
Conclusion
PAC settings can be effective training sites for medicine clerkships for PA students and can provide high-quality training in IM as PA programs continue to expand. This setting offers students exposure to interprofessional, team-based care and the opportunity to care for patients with a broad range of disease complexity. Learning is further enhanced by the ability to follow patients longitudinally over their disease course as well as to work directly with teaching faculty and other interprofessional health care professionals. Evaluations of this novel clerkship experience have shown high levels of student satisfaction in knowledge growth, clinical skills, bedside teaching, and mentorship.
Acknowledgments
We thank Juman Hijab for her critical role in establishing and maintaining the clerkship. We thank Steven Simon, Matt Russell, and Thomas Parrino for their leadership and guidance in establishing and maintaining the clerkship. We thank the Boston University School of Medicine Physician Assistant Program Director Mary Warner for her support and guidance in creating and supporting the clerkship. In addition, we thank the interprofessional education faculty for their dedicated involvement in teaching, including Stephanie Saunders, Lindsay Lefers, Jessica Rawlins, Lindsay Brennan, Angela Viani, Eric Charette, Nicole O’Neil, Susan Nathan, Jordana Meyerson, Shivani Jindal, Wei Shen, Amy Hanson, Gilda Cain, and Kate Hinrichs.
1. US Department of Labor, Bureau of Labor Statistics. Occupational outlook handbook: physician assistants. https://www.bls.gov/ooh/healthcare/physician-assistants.htm. Updated June 18, 2019. Accessed August 13, 2019.
2. Association of American Medical Colleges. 2019 update: the complexities of physician supply and demand: projections from 2017 to 2032. https://aamc-black.global.ssl.fastly.net/production/media/filer_public/31/13/3113ee5c-a038-4c16-89af-294a69826650/2019_update_-_the_complexities_of_physician_supply_and_demand_-_projections_from_2017-2032.pdf. Published April 2019. Accessed August 15, 2019.
3. Glicken AD, Miller AA. Physician assistants: from pipeline to practice. Acad Med. 2013;88(12):1883-1889.
4. Erikson C, Hamann R, Levitan T, Pankow S, Stanley J, Whatley M. Recruiting and maintaining US clinical training sites: joint report of the 2013 multi-discipline clerkship/clinical training site survey. https://paeaonline.org/wp-content/uploads/2015/10/Recruiting-and-Maintaining-U.S.-Clinical-Training-Sites.pdf. Accessed August 13, 2019.
5. Physician Assistant Education Association. By the numbers: 30th annual report on physician assistant educational programs. 2015. http://paeaonline.org/wp-content/uploads/2016/12/2015-by-the-numbers-program-report-30.pdf. Published 2015. Accessed August 15, 2019.
6. Morgan P, Himmerick KA, Leach B, Dieter P, Everett C. Scarcity of primary care positions may divert physician assistants into specialty practice. Med Care Res Rev. 2017;74(1):109-122.
7. Coplan B, Cawley J, Stoehr J. Physician assistants in primary care: trends and characteristics. Ann Fam Med. 2013;11(1):75-79.
8. Morgan P, Leach B, Himmerick K, Everett C. Job openings for PAs by specialty. JAAPA. 2018;31(1):45-47.
9. American Academy of Physician Assistants. 2017 AAPA Salary Report. Alexandria, VA; 2017.
10. Barnett ML, Grabowski DC, Mehrotra A. Home-to-home time—measuring what matters to patients and payers. N Engl J Med. 2017;377(1):4-6.
11. Werner RM, Konetzka RT. Trends in post-acute care use among Medicare beneficiaries: 2000 to 2015. JAMA. 2018;319(15):1616-1617.
12. Accreditation Review Commission on Education for the Physician Assistant. http://www.arc-pa.org/accreditation/accredited-programs. Accessed May 10, 2019.
The Federal Bureau of Labor Statistics projects 37% job growth for physician assistants (PAs) from 2016 to 2026, much greater than the average for all other occupations as well as for other medical professions.1 This growth has been accompanied by increased enrollment in medical (doctor of medicine [MD], doctor of osteopathic medicine) and nurse practitioner (NP) schools.2 Clinical teaching sites serve a crucial function in the training of all clinical disciplines. These sites provide hands-on and experiential learning in medical settings, necessary components for learners practicing to become clinicians. Significant PA program expansion has led to increased demand for clinical training, creating competition for sites and a shortage of willing and well-trained preceptors.3
This challenge has been recognized by PA program directors. In the Joint Report of the 2013 Multi-Discipline Clerkship/Clinical Training Site Survey, PA program directors expressed concern about the adequacy of clinical opportunities for students, increased difficulty developing new core sites, and preserving existing core sites. In addition, they noted that a shortage of clinical sites was one of the greatest barriers to the PA programs’ sustained growth and success.4
Program directors also indicated difficulty securing clinical training sites in internal medicine (IM) and high rates of attrition of medicine clinical preceptors for their students.5 The reasons are multifold: increasing clinical demands, time, teaching competence, lack of experience, academic affiliation, lack of reimbursement, or compensation. Moreover, there is a declining number of PAs who work in primary care compared with specialty and subspecialty care, limiting the availability of clinical training preceptors in medicine and primary care.6-8 According to the American Academy of PAs (AAPA) census and salary survey data, the percentage of PAs working in the primary care specialties (ie, family medicine, IM, and general pediatrics) has decreased from > 47% in 1995 to 24% in 2017.9 As such, there is a need to broaden the educational landscape to provide more high-quality training sites in IM.
The postacute health care setting may address this training need. It offers a unique clinical opportunity to expose learners to a broad range of disease complexity and clinical acuity, as the percentage of patients discharged from hospitals to postacute care (PAC) has increased and care shifts from the hospital to the PAC setting.10,11 The longer PAC length of stay also enables learners to follow patients longitudinally over several weeks and experience interprofessional team-based care. In addition, the PAC setting offers learners the ability to acquire the necessary skills for smooth and effective transitions of care. This setting has been extensively used for trainees of nursing, pharmacy, physical therapy (PT) and occupational therapy (OT), speech-language pathology, psychology, and social work (SW), but few programs have used the PAC setting as clerkship sites for IM rotations for PA students. To address this need for IM sites, the VA Boston Healthcare System (VABHS), in conjunction with the Boston University School of Medicine Physician Assistant Program, developed a novel medicine clinical clerkship site for physician assistants in the PAC unit of the community living center (CLC) at VABHS. This report describes the program structure, curriculum, and participant evaluation results.
Clinical Clerkship Program
VABHS CLC is a 110-bed facility comprising 3 units: a 65-bed PAC unit, a 15-bed closed hospice/palliative care unit, and a 30-bed long-term care unit. The service is staffed continuously with physicians, PAs, and NPs. A majority of patients are admitted from the acute care hospital of VABHS (West Roxbury campus) and other regional VA facilities. The CLC offers dynamic services, including phlebotomy, general radiology, IV diuretics and antibiotics, wound care, and subacute PT, OT, and speech-language pathology rehabilitation. The CLC serves as a venue for transitioning patients from acute inpatient care to home. The patient population is often elderly, with multiple active comorbidities and variable medical literacy, adherence, and follow-up.
The CLC provides a diverse interprofessional learning environment, offering core IM rotations for first-year psychiatry residents, oral and maxillofacial surgery residents, and PA students. The CLC also has expanded as a clinical site both for transitions-in-care IM resident curricula and electives as well as a geriatrics fellowship. In addition, the site offers rotations for NPs, nursing, pharmacy, physical and occupational therapies, speech-language pathology, psychology, and SW.
The Boston University School of Medicine Physician Assistant Program was founded in 2015 as a master’s degree program completed over 28 months. The first 12 months are didactic, and the following 16 months are clinical training with 14 months of rotations (2 IM, family medicine, pediatrics, emergency medicine, general surgery, obstetrics and gynecology, psychiatry, neurology, and 5 elective rotations), and 2 months for a thesis. The program has about 30 students per year and 4 clerkship sites for IM.
Program Description
The VABHS medicine clerkship hosts 1 to 2 PA students for 4-week blocks in the PAC unit of the CLC. Each student rotates on both PA and MD teams. Students follow 3 to 4 patients and participate fully in their care from admission to discharge; they prepare daily presentations and participate in medical management, family meetings, chart documentation, and care coordination with the interprofessional team. Students are provided a physical examination checklist and feedback form, and they are expected to track findings and record feedback and goals with their supervising preceptor weekly. They also make formal case presentations and participate in monthly medicine didactic rounds available to all VABHS IM students and trainees via videoconference.
In addition, beginning in July 2017, all PA students in the CLC began to participate in a 4-week Interprofessional Curriculum in Transitional Care. The curriculum includes 14 didactic lectures taught by 16 interprofessional faculty, including medicine, geriatric, and palliative care physicians; PAs; social workers; physical and occupational therapists; pharmacists; and a geriatric psychologist. The didactics include topics on the interprofessional team, the care continuum, teams and teamwork, interdisciplinary coordination of care, components of effective transitions in care, medication reconciliation, approaching difficult conversations, advance care planning, and quality improvement. The goal of the curriculum is to provide learners the knowledge, skills, and dispositions necessary for high-quality transitional care and interprofessional practice as well as specific training for effective and safe transfers of care between clinical settings. Although PA students are the main participants in this curriculum, all other learners in the PAC unit are also invited to attend the lectures.
The unique attributes of this training site include direct interaction with supervising PAs and physicians, rather than experiencing the traditional teaching hierarchy (with interns, residents, fellows); observation of the natural progression of disease of both acute care and primary care issues due to the longer length of stay (2 to 6 weeks, where the typical student will see the same patient 7 to 10 times during their rotation); exposure to a host of medically complex patients offering a multitude of clinical scenarios and abnormal physical exam findings; exposure to a hospice/palliative care ward and end-of-life care; and interaction within an interprofessional training environment of nursing, pharmacy, PT, OT, speech-language pathology, psychology, and SW trainees.
Program Evaluation
At the end of rotations continuously through the year, PA students electronically complete a site evaluation from the Boston University School of Medicine Physician Assistant Program. The evaluation consists of 14 questions: 6 about site quality and 8 about instruction quality. The questions are answered on a 5-point Likert scale. Also included are 2 open-ended response questions that ask what they liked about the rotation and what they felt could be improved. Results are anonymous, de-identified and blinded both to the program as well as the clerkship site. Results are aggregated and provided to program sites annually. Responses are converted to a dichotomous variable, where any good or excellent response (4 or 5) is considered positive and any neutral or below (3, 2, 1) is considered a nonpositive response.
Results
The clerkship site has been operational since June 22, 2015. There have been 59 students who participated in the rotation. A different scale in these evaluations was used between June 22, 2015, and September 13, 2015. Therefore, 7 responses were excluded from the analysis, leaving 52 usable evaluations. The responses were analyzed both in total (for the CLC as well as other IM rotation sites) and by individual clerkship year to look for any trends over time: September 14, 2015, through April 24, 2016; April 25, 2016, through April 28, 2017; and May 1, 2017, through March 1, 2018 (Table).
Site evaluations showed high satisfaction regarding the quality of the physical environment as well as the learning environment. Students endorsed the PAC unit having resources and physical space for them, such as a desk and computer, opportunity for participation in patient care, and parking (100%; n = 52). Site evaluations revealed high satisfaction with the quality of teaching and faculty encouragement and support of their learning (100%; n = 52). The evaluations revealed that bedside teaching was strong (94%; n = 49). The students reported high satisfaction with the volume of patients provided (92%; n = 48) as well as the diversity of diagnoses (92%; n = 48).
There were fewer positive responses in the first 2 years of the rotation with regard to formal lectures (50% and 67%; 7/14 and 16/24, respectively). In the third year of the rotation, students had a much higher satisfaction rate (93%; 13/14). This increased satisfaction was associated with the development and incorporation of the Interprofessional Curriculum in Transitional Care in 2017.
Discussion
Access to high-quality PA student clerkship sites has become a pressing issue in recent years because of increased competition for sites and a shortage of willing and well-trained preceptors. There has been marked growth in schools and enrollment across all medical professions. The Accreditation Review Commission on Education for the PA (ARC-PA) reported that the total number of accredited entry-level PA programs in 2018 was 246, with 58 new accredited programs projected by 2022.12 The Joint Report of the 2013 Multi-Discipline Clerkship/Clinical Training Site Survey reported a 66% increase in first-year enrollment in PA programs from 2002 to 2012.5 Programs must implement alternative strategies to attract clinical sites (eg, academic appointments, increased clinical resources to training sites) or face continued challenges with recruiting training sites for their students. Postacute care may be a natural extension to expand the footprint for clinical sites for these programs, augmenting acute inpatient and outpatient rotations. This implementation would increase the pool of clinical training sites and preceptors.
The experience with this novel training site, based on PA student feedback and evaluations, has been positive, and the postacute setting can provide students with high-quality IM clinical experiences. Students report adequate patient volume and diversity. In addition, evaluations are comparable with that of other IM site rotations the students experience. Qualitative feedback has emphasized the value of following patients over longer periods; eg, weeks vs days (as in acute care) enabling students to build relationships with patients as well as observe a richer clinical spectrum of disease over a less compressed period. “Patients have complex issues, so from a medical standpoint it challenges you to think of new ways to manage their care,” commented a representative student. “It is really beneficial that you can follow them over time.”
Furthermore, in response to student feedback on didactics, an interprofessional curriculum was developed to add formal structure as well as to create a curriculum in care transitions. This curriculum provided a unique opportunity for PA students to receive formal instruction on areas of particular relevance for transitional care (eg, care continuum, end of life issues, and care transitions). The curriculum also allows the interprofessional faculty a unique and enjoyable opportunity for interprofessional collaboration.
The 1 month PAC rotation is augmented with inpatient IM and outpatient family medicine rotations, consequently giving exposure to the full continuum of care. The PAC setting provides learners multifaceted benefits: the opportunity to strengthen and develop the knowledge, attitudes, and skills necessary for IM; increased understanding of other professions by observing and interacting as a team caring for a patient over a longer period as opposed to the acute care setting; the ability to perform effective, efficient, and safe transfer between clinical settings; and broad exposure to transitional care. As a result, the PAC rotation enhances but does not replace the necessary and essential rotations of inpatient and outpatient medicine.
Moreover, this rotation provides unique and core IM training for PA students. Our site focuses on interprofessional collaboration, emphasizing the importance of team-based care, an essential concept in modern day medicine. Formal exposure to other care specialties, such as PT and OT, SW, and mental health, is essential for students to appreciate clinical medicine and a patient’s physical and mental experience over the course of a disease and clinical state. In addition, the physical exam checklist ensures that students are exposed to the full spectrum of IM examination findings during their rotation. Finally, weekly feedback forms require students to ask and receive concrete feedback from their supervising providers.
Limitations
The generalizability of this model requires careful consideration. VABHS is a tertiary care integrated health care system, enabling students to learn from patients moving through multiple care transitions in a single health care system. In addition, other settings may not have the staffing or clinical volume to sustain such a model. All PAC clinical faculty teach voluntarily, and local leadership has set expectations for all clinicians to participate in teaching of trainees and PA students. Evaluations also note less diversity in the patient population, a challenge that some VA facilities face. This issue could be addressed by ensuring that students also have IM rotations at other inpatient medical facilities. A more balanced experience, where students reap the positive benefits of PAC but do not lose exposure to a diverse patient pool, could result. Furthermore, some of the perceived positive impacts also may be related to professional and personal attributes of the teaching clinicians rather than to the PAC setting.
Conclusion
PAC settings can be effective training sites for medicine clerkships for PA students and can provide high-quality training in IM as PA programs continue to expand. This setting offers students exposure to interprofessional, team-based care and the opportunity to care for patients with a broad range of disease complexity. Learning is further enhanced by the ability to follow patients longitudinally over their disease course as well as to work directly with teaching faculty and other interprofessional health care professionals. Evaluations of this novel clerkship experience have shown high levels of student satisfaction in knowledge growth, clinical skills, bedside teaching, and mentorship.
Acknowledgments
We thank Juman Hijab for her critical role in establishing and maintaining the clerkship. We thank Steven Simon, Matt Russell, and Thomas Parrino for their leadership and guidance in establishing and maintaining the clerkship. We thank the Boston University School of Medicine Physician Assistant Program Director Mary Warner for her support and guidance in creating and supporting the clerkship. In addition, we thank the interprofessional education faculty for their dedicated involvement in teaching, including Stephanie Saunders, Lindsay Lefers, Jessica Rawlins, Lindsay Brennan, Angela Viani, Eric Charette, Nicole O’Neil, Susan Nathan, Jordana Meyerson, Shivani Jindal, Wei Shen, Amy Hanson, Gilda Cain, and Kate Hinrichs.
The Federal Bureau of Labor Statistics projects 37% job growth for physician assistants (PAs) from 2016 to 2026, much greater than the average for all other occupations as well as for other medical professions.1 This growth has been accompanied by increased enrollment in medical (doctor of medicine [MD], doctor of osteopathic medicine) and nurse practitioner (NP) schools.2 Clinical teaching sites serve a crucial function in the training of all clinical disciplines. These sites provide hands-on and experiential learning in medical settings, necessary components for learners practicing to become clinicians. Significant PA program expansion has led to increased demand for clinical training, creating competition for sites and a shortage of willing and well-trained preceptors.3
This challenge has been recognized by PA program directors. In the Joint Report of the 2013 Multi-Discipline Clerkship/Clinical Training Site Survey, PA program directors expressed concern about the adequacy of clinical opportunities for students, increased difficulty developing new core sites, and preserving existing core sites. In addition, they noted that a shortage of clinical sites was one of the greatest barriers to the PA programs’ sustained growth and success.4
Program directors also indicated difficulty securing clinical training sites in internal medicine (IM) and high rates of attrition of medicine clinical preceptors for their students.5 The reasons are multifold: increasing clinical demands, time, teaching competence, lack of experience, academic affiliation, lack of reimbursement, or compensation. Moreover, there is a declining number of PAs who work in primary care compared with specialty and subspecialty care, limiting the availability of clinical training preceptors in medicine and primary care.6-8 According to the American Academy of PAs (AAPA) census and salary survey data, the percentage of PAs working in the primary care specialties (ie, family medicine, IM, and general pediatrics) has decreased from > 47% in 1995 to 24% in 2017.9 As such, there is a need to broaden the educational landscape to provide more high-quality training sites in IM.
The postacute health care setting may address this training need. It offers a unique clinical opportunity to expose learners to a broad range of disease complexity and clinical acuity, as the percentage of patients discharged from hospitals to postacute care (PAC) has increased and care shifts from the hospital to the PAC setting.10,11 The longer PAC length of stay also enables learners to follow patients longitudinally over several weeks and experience interprofessional team-based care. In addition, the PAC setting offers learners the ability to acquire the necessary skills for smooth and effective transitions of care. This setting has been extensively used for trainees of nursing, pharmacy, physical therapy (PT) and occupational therapy (OT), speech-language pathology, psychology, and social work (SW), but few programs have used the PAC setting as clerkship sites for IM rotations for PA students. To address this need for IM sites, the VA Boston Healthcare System (VABHS), in conjunction with the Boston University School of Medicine Physician Assistant Program, developed a novel medicine clinical clerkship site for physician assistants in the PAC unit of the community living center (CLC) at VABHS. This report describes the program structure, curriculum, and participant evaluation results.
Clinical Clerkship Program
VABHS CLC is a 110-bed facility comprising 3 units: a 65-bed PAC unit, a 15-bed closed hospice/palliative care unit, and a 30-bed long-term care unit. The service is staffed continuously with physicians, PAs, and NPs. A majority of patients are admitted from the acute care hospital of VABHS (West Roxbury campus) and other regional VA facilities. The CLC offers dynamic services, including phlebotomy, general radiology, IV diuretics and antibiotics, wound care, and subacute PT, OT, and speech-language pathology rehabilitation. The CLC serves as a venue for transitioning patients from acute inpatient care to home. The patient population is often elderly, with multiple active comorbidities and variable medical literacy, adherence, and follow-up.
The CLC provides a diverse interprofessional learning environment, offering core IM rotations for first-year psychiatry residents, oral and maxillofacial surgery residents, and PA students. The CLC also has expanded as a clinical site both for transitions-in-care IM resident curricula and electives as well as a geriatrics fellowship. In addition, the site offers rotations for NPs, nursing, pharmacy, physical and occupational therapies, speech-language pathology, psychology, and SW.
The Boston University School of Medicine Physician Assistant Program was founded in 2015 as a master’s degree program completed over 28 months. The first 12 months are didactic, and the following 16 months are clinical training with 14 months of rotations (2 IM, family medicine, pediatrics, emergency medicine, general surgery, obstetrics and gynecology, psychiatry, neurology, and 5 elective rotations), and 2 months for a thesis. The program has about 30 students per year and 4 clerkship sites for IM.
Program Description
The VABHS medicine clerkship hosts 1 to 2 PA students for 4-week blocks in the PAC unit of the CLC. Each student rotates on both PA and MD teams. Students follow 3 to 4 patients and participate fully in their care from admission to discharge; they prepare daily presentations and participate in medical management, family meetings, chart documentation, and care coordination with the interprofessional team. Students are provided a physical examination checklist and feedback form, and they are expected to track findings and record feedback and goals with their supervising preceptor weekly. They also make formal case presentations and participate in monthly medicine didactic rounds available to all VABHS IM students and trainees via videoconference.
In addition, beginning in July 2017, all PA students in the CLC began to participate in a 4-week Interprofessional Curriculum in Transitional Care. The curriculum includes 14 didactic lectures taught by 16 interprofessional faculty, including medicine, geriatric, and palliative care physicians; PAs; social workers; physical and occupational therapists; pharmacists; and a geriatric psychologist. The didactics include topics on the interprofessional team, the care continuum, teams and teamwork, interdisciplinary coordination of care, components of effective transitions in care, medication reconciliation, approaching difficult conversations, advance care planning, and quality improvement. The goal of the curriculum is to provide learners the knowledge, skills, and dispositions necessary for high-quality transitional care and interprofessional practice as well as specific training for effective and safe transfers of care between clinical settings. Although PA students are the main participants in this curriculum, all other learners in the PAC unit are also invited to attend the lectures.
The unique attributes of this training site include direct interaction with supervising PAs and physicians, rather than experiencing the traditional teaching hierarchy (with interns, residents, fellows); observation of the natural progression of disease of both acute care and primary care issues due to the longer length of stay (2 to 6 weeks, where the typical student will see the same patient 7 to 10 times during their rotation); exposure to a host of medically complex patients offering a multitude of clinical scenarios and abnormal physical exam findings; exposure to a hospice/palliative care ward and end-of-life care; and interaction within an interprofessional training environment of nursing, pharmacy, PT, OT, speech-language pathology, psychology, and SW trainees.
Program Evaluation
At the end of rotations continuously through the year, PA students electronically complete a site evaluation from the Boston University School of Medicine Physician Assistant Program. The evaluation consists of 14 questions: 6 about site quality and 8 about instruction quality. The questions are answered on a 5-point Likert scale. Also included are 2 open-ended response questions that ask what they liked about the rotation and what they felt could be improved. Results are anonymous, de-identified and blinded both to the program as well as the clerkship site. Results are aggregated and provided to program sites annually. Responses are converted to a dichotomous variable, where any good or excellent response (4 or 5) is considered positive and any neutral or below (3, 2, 1) is considered a nonpositive response.
Results
The clerkship site has been operational since June 22, 2015. There have been 59 students who participated in the rotation. A different scale in these evaluations was used between June 22, 2015, and September 13, 2015. Therefore, 7 responses were excluded from the analysis, leaving 52 usable evaluations. The responses were analyzed both in total (for the CLC as well as other IM rotation sites) and by individual clerkship year to look for any trends over time: September 14, 2015, through April 24, 2016; April 25, 2016, through April 28, 2017; and May 1, 2017, through March 1, 2018 (Table).
Site evaluations showed high satisfaction regarding the quality of the physical environment as well as the learning environment. Students endorsed the PAC unit having resources and physical space for them, such as a desk and computer, opportunity for participation in patient care, and parking (100%; n = 52). Site evaluations revealed high satisfaction with the quality of teaching and faculty encouragement and support of their learning (100%; n = 52). The evaluations revealed that bedside teaching was strong (94%; n = 49). The students reported high satisfaction with the volume of patients provided (92%; n = 48) as well as the diversity of diagnoses (92%; n = 48).
There were fewer positive responses in the first 2 years of the rotation with regard to formal lectures (50% and 67%; 7/14 and 16/24, respectively). In the third year of the rotation, students had a much higher satisfaction rate (93%; 13/14). This increased satisfaction was associated with the development and incorporation of the Interprofessional Curriculum in Transitional Care in 2017.
Discussion
Access to high-quality PA student clerkship sites has become a pressing issue in recent years because of increased competition for sites and a shortage of willing and well-trained preceptors. There has been marked growth in schools and enrollment across all medical professions. The Accreditation Review Commission on Education for the PA (ARC-PA) reported that the total number of accredited entry-level PA programs in 2018 was 246, with 58 new accredited programs projected by 2022.12 The Joint Report of the 2013 Multi-Discipline Clerkship/Clinical Training Site Survey reported a 66% increase in first-year enrollment in PA programs from 2002 to 2012.5 Programs must implement alternative strategies to attract clinical sites (eg, academic appointments, increased clinical resources to training sites) or face continued challenges with recruiting training sites for their students. Postacute care may be a natural extension to expand the footprint for clinical sites for these programs, augmenting acute inpatient and outpatient rotations. This implementation would increase the pool of clinical training sites and preceptors.
The experience with this novel training site, based on PA student feedback and evaluations, has been positive, and the postacute setting can provide students with high-quality IM clinical experiences. Students report adequate patient volume and diversity. In addition, evaluations are comparable with that of other IM site rotations the students experience. Qualitative feedback has emphasized the value of following patients over longer periods; eg, weeks vs days (as in acute care) enabling students to build relationships with patients as well as observe a richer clinical spectrum of disease over a less compressed period. “Patients have complex issues, so from a medical standpoint it challenges you to think of new ways to manage their care,” commented a representative student. “It is really beneficial that you can follow them over time.”
Furthermore, in response to student feedback on didactics, an interprofessional curriculum was developed to add formal structure as well as to create a curriculum in care transitions. This curriculum provided a unique opportunity for PA students to receive formal instruction on areas of particular relevance for transitional care (eg, care continuum, end of life issues, and care transitions). The curriculum also allows the interprofessional faculty a unique and enjoyable opportunity for interprofessional collaboration.
The 1 month PAC rotation is augmented with inpatient IM and outpatient family medicine rotations, consequently giving exposure to the full continuum of care. The PAC setting provides learners multifaceted benefits: the opportunity to strengthen and develop the knowledge, attitudes, and skills necessary for IM; increased understanding of other professions by observing and interacting as a team caring for a patient over a longer period as opposed to the acute care setting; the ability to perform effective, efficient, and safe transfer between clinical settings; and broad exposure to transitional care. As a result, the PAC rotation enhances but does not replace the necessary and essential rotations of inpatient and outpatient medicine.
Moreover, this rotation provides unique and core IM training for PA students. Our site focuses on interprofessional collaboration, emphasizing the importance of team-based care, an essential concept in modern day medicine. Formal exposure to other care specialties, such as PT and OT, SW, and mental health, is essential for students to appreciate clinical medicine and a patient’s physical and mental experience over the course of a disease and clinical state. In addition, the physical exam checklist ensures that students are exposed to the full spectrum of IM examination findings during their rotation. Finally, weekly feedback forms require students to ask and receive concrete feedback from their supervising providers.
Limitations
The generalizability of this model requires careful consideration. VABHS is a tertiary care integrated health care system, enabling students to learn from patients moving through multiple care transitions in a single health care system. In addition, other settings may not have the staffing or clinical volume to sustain such a model. All PAC clinical faculty teach voluntarily, and local leadership has set expectations for all clinicians to participate in teaching of trainees and PA students. Evaluations also note less diversity in the patient population, a challenge that some VA facilities face. This issue could be addressed by ensuring that students also have IM rotations at other inpatient medical facilities. A more balanced experience, where students reap the positive benefits of PAC but do not lose exposure to a diverse patient pool, could result. Furthermore, some of the perceived positive impacts also may be related to professional and personal attributes of the teaching clinicians rather than to the PAC setting.
Conclusion
PAC settings can be effective training sites for medicine clerkships for PA students and can provide high-quality training in IM as PA programs continue to expand. This setting offers students exposure to interprofessional, team-based care and the opportunity to care for patients with a broad range of disease complexity. Learning is further enhanced by the ability to follow patients longitudinally over their disease course as well as to work directly with teaching faculty and other interprofessional health care professionals. Evaluations of this novel clerkship experience have shown high levels of student satisfaction in knowledge growth, clinical skills, bedside teaching, and mentorship.
Acknowledgments
We thank Juman Hijab for her critical role in establishing and maintaining the clerkship. We thank Steven Simon, Matt Russell, and Thomas Parrino for their leadership and guidance in establishing and maintaining the clerkship. We thank the Boston University School of Medicine Physician Assistant Program Director Mary Warner for her support and guidance in creating and supporting the clerkship. In addition, we thank the interprofessional education faculty for their dedicated involvement in teaching, including Stephanie Saunders, Lindsay Lefers, Jessica Rawlins, Lindsay Brennan, Angela Viani, Eric Charette, Nicole O’Neil, Susan Nathan, Jordana Meyerson, Shivani Jindal, Wei Shen, Amy Hanson, Gilda Cain, and Kate Hinrichs.
1. US Department of Labor, Bureau of Labor Statistics. Occupational outlook handbook: physician assistants. https://www.bls.gov/ooh/healthcare/physician-assistants.htm. Updated June 18, 2019. Accessed August 13, 2019.
2. Association of American Medical Colleges. 2019 update: the complexities of physician supply and demand: projections from 2017 to 2032. https://aamc-black.global.ssl.fastly.net/production/media/filer_public/31/13/3113ee5c-a038-4c16-89af-294a69826650/2019_update_-_the_complexities_of_physician_supply_and_demand_-_projections_from_2017-2032.pdf. Published April 2019. Accessed August 15, 2019.
3. Glicken AD, Miller AA. Physician assistants: from pipeline to practice. Acad Med. 2013;88(12):1883-1889.
4. Erikson C, Hamann R, Levitan T, Pankow S, Stanley J, Whatley M. Recruiting and maintaining US clinical training sites: joint report of the 2013 multi-discipline clerkship/clinical training site survey. https://paeaonline.org/wp-content/uploads/2015/10/Recruiting-and-Maintaining-U.S.-Clinical-Training-Sites.pdf. Accessed August 13, 2019.
5. Physician Assistant Education Association. By the numbers: 30th annual report on physician assistant educational programs. 2015. http://paeaonline.org/wp-content/uploads/2016/12/2015-by-the-numbers-program-report-30.pdf. Published 2015. Accessed August 15, 2019.
6. Morgan P, Himmerick KA, Leach B, Dieter P, Everett C. Scarcity of primary care positions may divert physician assistants into specialty practice. Med Care Res Rev. 2017;74(1):109-122.
7. Coplan B, Cawley J, Stoehr J. Physician assistants in primary care: trends and characteristics. Ann Fam Med. 2013;11(1):75-79.
8. Morgan P, Leach B, Himmerick K, Everett C. Job openings for PAs by specialty. JAAPA. 2018;31(1):45-47.
9. American Academy of Physician Assistants. 2017 AAPA Salary Report. Alexandria, VA; 2017.
10. Barnett ML, Grabowski DC, Mehrotra A. Home-to-home time—measuring what matters to patients and payers. N Engl J Med. 2017;377(1):4-6.
11. Werner RM, Konetzka RT. Trends in post-acute care use among Medicare beneficiaries: 2000 to 2015. JAMA. 2018;319(15):1616-1617.
12. Accreditation Review Commission on Education for the Physician Assistant. http://www.arc-pa.org/accreditation/accredited-programs. Accessed May 10, 2019.
1. US Department of Labor, Bureau of Labor Statistics. Occupational outlook handbook: physician assistants. https://www.bls.gov/ooh/healthcare/physician-assistants.htm. Updated June 18, 2019. Accessed August 13, 2019.
2. Association of American Medical Colleges. 2019 update: the complexities of physician supply and demand: projections from 2017 to 2032. https://aamc-black.global.ssl.fastly.net/production/media/filer_public/31/13/3113ee5c-a038-4c16-89af-294a69826650/2019_update_-_the_complexities_of_physician_supply_and_demand_-_projections_from_2017-2032.pdf. Published April 2019. Accessed August 15, 2019.
3. Glicken AD, Miller AA. Physician assistants: from pipeline to practice. Acad Med. 2013;88(12):1883-1889.
4. Erikson C, Hamann R, Levitan T, Pankow S, Stanley J, Whatley M. Recruiting and maintaining US clinical training sites: joint report of the 2013 multi-discipline clerkship/clinical training site survey. https://paeaonline.org/wp-content/uploads/2015/10/Recruiting-and-Maintaining-U.S.-Clinical-Training-Sites.pdf. Accessed August 13, 2019.
5. Physician Assistant Education Association. By the numbers: 30th annual report on physician assistant educational programs. 2015. http://paeaonline.org/wp-content/uploads/2016/12/2015-by-the-numbers-program-report-30.pdf. Published 2015. Accessed August 15, 2019.
6. Morgan P, Himmerick KA, Leach B, Dieter P, Everett C. Scarcity of primary care positions may divert physician assistants into specialty practice. Med Care Res Rev. 2017;74(1):109-122.
7. Coplan B, Cawley J, Stoehr J. Physician assistants in primary care: trends and characteristics. Ann Fam Med. 2013;11(1):75-79.
8. Morgan P, Leach B, Himmerick K, Everett C. Job openings for PAs by specialty. JAAPA. 2018;31(1):45-47.
9. American Academy of Physician Assistants. 2017 AAPA Salary Report. Alexandria, VA; 2017.
10. Barnett ML, Grabowski DC, Mehrotra A. Home-to-home time—measuring what matters to patients and payers. N Engl J Med. 2017;377(1):4-6.
11. Werner RM, Konetzka RT. Trends in post-acute care use among Medicare beneficiaries: 2000 to 2015. JAMA. 2018;319(15):1616-1617.
12. Accreditation Review Commission on Education for the Physician Assistant. http://www.arc-pa.org/accreditation/accredited-programs. Accessed May 10, 2019.
Multispecialty Opioid Risk Reduction Program Targeting Chronic Pain and Addiction Management in Veterans
Chronic pain significantly affects 100 million Americans.1,2 Pain accounts for $560 to $635 billion in annual financial costs to society, including health care costs and loss of productivity (ie, days missed from work, hours of work lost, and lower wages).2,3 Although pain prevalence exceeds other chronic diseases, such as diabetes mellitus, cancer, and heart disease, it lacks a sufficient body of evidence-based research and guidelines on the underlying mechanisms, valid methods of assessment, and comparative effectiveness of treatments to effectively implement into clinical practice.2,4 Prevention and treatment of pain are often delayed, inaccessible, or inadequate.2 Primary care providers (PCPs) are most often sought for pain management and treat about 52% of chronic pain patients.2,3,5 Veterans are especially vulnerable to chronic pain and are at risk for inadequate treatment.2
Background
There is an epidemic of drug abuse and mortality from opioid prescription medication.6 In the US, rates of overdose deaths from prescription opioids were 6.1 per 100,000 for men and 4.2 per 100,000 for women in 2017. Opioids were involved in 47,600 overdose deaths in 2017, accounting for 67.8% of all drug overdose deaths.7
A large number of patients on long-term opioids have preexisting substance use disorders and/or psychiatric disease, further complicating chronic pain management.8-10 Prescription opioid use has been the precursor for about 80% of people who are now heroin addicts.11 Iatrogenic addiction from prescription medications isn’t easily captured by standard addiction criteria. Consequently, in patients who are on opioid therapy for prolonged periods, separating complex opioid dependence from addiction is difficult.12 Improved addiction screening and risk mitigation strategies are needed along with aggressive treatment monitoring to curb the opioid epidemic.
Opioid Management in Primary Care
The majority of opioid medications are prescribed by PCPs, which is magnified in the US Department of Veterans Affairs (VA) health care system due to the high prevalence of service-related injuries.3,13 The VA is at the forefront of addressing the complexities of opioid addiction through several initiatives.14 The ability to offer the frequent visits needed to safely manage patients prescribed opioids and the integration of mental health and addiction treatment are often lacking in non-VA primary care clinics. Therefore, a key to solving the opioid crisis is developing these capabilities so they can be delivered within the primary care setting. There is substantial evidence in support of nonopioid alternatives to chronic pain management, including other pharmacologic approaches, exercise, physical therapy, acupuncture, weight loss, smoking cessation, chiropractic care, cognitive behavioral therapy (CBT), and other integrative health modalities.
A 2009 VA directive mandated the development of a comprehensive, integrated, systemwide approach to pain management.15 The VA Stepped-Care Biopsychosocial Model for Pain Management is dependent on timely access to secondary consultation from pain medicine, behavioral health, physical medicine, and other specialty consultation.15
History of VHA SCAN-ECHO Model
The Specialty Care Access Network–Extension for Community Health Outcomes (SCAN-ECHO) is a Veterans Health Administration (VHA) adaptation of a program that originated at the University of Mexico.16,17 The SCAN-ECHO model uses a multisite videoconferencing network to provide specialty care consultations to PCPs and patient aligned care teams (PACTs). During the 60- to 90-minute weekly sessions, case presentations are analyzed in real time so that over time, the PCPs gain knowledge, competency, and confidence in learning how to handle complex chronic conditions.
Since its implementation, the SCAN-ECHO program has been adopted across the VHA in a variety of specialties. One program, the SCAN-ECHO for Pain Management (SCAN-ECHO-PM) was implemented in 7 VHA networks in 31 states, spanning 47 medical centers and 148 community-based outpatient clinics (CBOCs).18 The SCAN-ECHO-PM program successfully conducted 257 multidisciplinary pain consultations between 2011 and 2013, resulting in increased initiation of nonopioid medications.18
The aim of this article is to describe the implementation of a multicomponent primary care-based pain clinic with a fully integrated mental health service and addiction service at the North Florida/South Georgia Veterans Health System (NF/SGVHS). A practiced-based intervention of the biopsychosocial model with robust patient engagement has guided the development of the NF/SGVHS pain clinic (Figure 1).4,19
Pain CLinic
NF/SGVHS comprises the Malcom Randall and Lake City VA medical centers (VAMCs) hospitals, 3 satellite outpatient clinics, and 8 CBOCs. Spanning 33 counties in North Florida and 19 counties in South Georgia, the NF/SGVHS serves more than 140,000 patients. In 2010, the Malcom Randall VAMC established a multidisciplinary primary care pain clinic to manage veterans at high-risk for noncancer chronic pain and addiction. The noncancer pain policy was revised after garnering support from stakeholders who treat chronic pain, including the chiefs of psychiatry, rehabilitation medicine, neurosurgery, psychology, interventional pain, pharmacy, nursing, addiction medicine, and primary care. The clinic is staffed by primary care physicians trained in internal medicine and family medicine and is structured with 1-hour first visits, and 30-minute follow-up visits to allow enough time for comprehensive evaluation while meeting the needs for close follow-up support.
All physicians in the clinic have buprenorphine prescribing credentials to aid in the management of opioid addiction, as some patients feel more comfortable receiving addiction treatment in a primary care setting. The multimodal care model consists of several services that include addiction psychiatrists, interventional pain specialists, pain psychologists, and pain pharmacologists who coordinate the care to the veterans. The addiction psychiatrists offer a full range of services with inpatient residential and outpatient programs. Through recurring meetings with primary care pain clinic staff, the addiction psychiatrists are available to discuss use of buprenorphine and arrange follow-up for patients with complex pain addiction. There is ongoing collaboration to develop the best care plan that meets the patient’s needs for chronic pain, addiction, and/or mental health issues. The interventional pain service has 3 fellowship-trained pain care providers who deliver comprehensive evaluation, pharmacologic recommendations, and a full range of interventional and complementary therapies with an emphasis on objective functional improvement. Pain care providers offer alternatives to patients who are being weaned from opioids and support the multidisciplinary patient engagement model.
The pain psychology program, established in 2011, delivers CBT to 5 onsite locations and 5 telehealth locations. The service includes an advanced CBT program and a couples CBT program. The pharmacy pain fellowship program provides staff for an outpatient e-consult pain management service and an inpatient pharmacy consult service. Harnessing pain specialty pharmacists, the pharmacy service addresses pharmacokinetic issues, urine drug screen (UDS) results, opioid tapering and discharge planning for pain, addiction and mental health needs. The NF/SGVHS Primary Care Pain Clinic was established to support PCPs who did not feel comfortable managing chronic pain patients. These patients were typically on high-dose opioid therapy (> 100-mg morphine equivalent daily doses [MEDDs]); patients with a history of opioid addiction; patients with an addiction to opioids combined with benzodiazepines; and patients with comorbid medical issues (eg, sleep apnea), which complicated their management. The process of addressing opioid safety in these complex pain patients can be labor intensive and generally cannot be accomplished in a brief visit in a primary care setting where many other medical problems often need to be addressed.
Most patients on high-dose opioids are fearful of any changes in their medications. The difficult conversation regarding opioid safety is a lengthy one and frequently will occur over multiple visits. In addition, safely tapering opioids requires frequent follow-up to provide psychological support and to address withdrawal and mental health issues that may arise. As opioids are tapered, the clinic reinforces improved pain care through a multimodal biopsychosocial model. All veterans receiving pain care outside the VA are monitored annually to assure they are receiving evidence-based pain care as defined by the biopsychosocial model.
Education
Since 2011, the NF/SGVHS SCAN-ECHO pain and addiction educational forum has created > 50 hours of approved annual continuing medical education (CME) on pain management and addiction for PCPs. Initially, the 1-hour weekly educational audioconferences presented a pain management case along with related topics and involved specialists from interventional pain, physical therapy, psychiatry, nursing, neurology, and psychology departments. In 2013, in conjunction with the VA SCAN-ECHO program of Hunter Holmes McGuire VAMC in Richmond, Virginia, and Walter Reed National Military Medical Center in Bethesda, Maryland, the audioconference was expanded to 2 days each week with additional topics on addiction management. Residency and fellowship rotations were developed that specifically targeted fellows from psychiatry, pharmacology, and interventional pain departments.
Currently, an 8-session pain school is delivered onsite and at 7 telehealth locations. The school is a collaborative effort involving interventional pain, psychology, pharmacy, nutrition, and the primary care pain clinic staff. As the cornerstone of the program, the pain school stresses the biopsychosocial patient engagement model.
Program Evaluation
The VA is equipped with multiple telehealth service networks that allow for the delivery of programs, such as the pain school, a pain psychology program, and a yoga program, onsite or offsite. The VA Computerized Patient Record System (CPRS) manages electronic health records, allowing for rapid chart review and e-consults. The NF/SGVHS Pain Management Program provides about 1500 e-consults yearly. The CPRS includes templates with pain metrics to help PCPs deliver pain care more efficiently and evaluate performance measures. This system also allows for the capture of data to track improvements in the care of the veterans served.
From 2012 to 2017, more than 5000 NF/SGVHS patients were weaned from opioids. Overall, there was an 87% reduction in patients receiving opioids ( ≥ 100-mg MEDDs) within the NF/SGVHS, which is significantly more than the 49% seen nationally across the VHA (Figure 2). Percent reduction was calculated by taking the difference in number of patients receiving opioids in 2012 and 2017, dividing by the number of patients receiving opioids in 2012 and multiplying by 100. The largest proportion of opioid dose reductions for NF/SGVHS and VHA patients, respectively, were seen in 300-mg to 399-mg MEDDs (95% vs 67%, respectively); followed by ≥ 400-mg MEDDs (94% vs 71%, respectively); 200-mg to 299-mg MEDDs (91% vs 58%, respectively); and 100-mg to 199-mg MEDDs (84% vs 40%, respectively). When examining NF/SGVHS trends over time, there has been a consistent decline in patients prescribed opioids (18 223 in 2012 compared with 12 877 in 2017) with similar trends in benzodiazepine-opioid combination therapy (2694 in 2012 compared with 833 in 2017) (Figure 3).
Similar declines are seen when patients are stratified by the MEDD (Figure 4). From 2012 to 2017, 92% of the patients were successfully tapered off doses ≥ 400-mg MEDD (2012, n = 72; 2017, n = 6), and tapered off 300-mg to 399-mg MEDD (2012, n = 107; 2017, n = 5); 95% were tapered off 200-mg to 299-mg MEDD (2012, n = 262; 2017, n = 22); and 86% were tapered off 100-mg to 199-mg MEDD (2012, n = 876; 2017; n = 127).
Conclusion
Successful integration of primary care with mental health and addiction services is paramount to aggressively taper patients with chronic pain from opioids. There is evidence that drug dependence and chronic pain should be treated like other chronic illness.20 Both chronic pain and addiction can be treated with a multidimensional self-management approach. In view of the high incidence of mental health and addiction associated with opioid use, it makes sense that an integrated, 1-stop pain and addiction clinic that understands and addresses both issues is more likely to improve patient outcomes.
Acknowledgments
This material is the result of work supported by the resources and facilities at the North Florida/South Georgia Veterans Health System, Geriatric Research Education Clinical Center in Gainesville, Florida.
1. Dueñas M, Ojeda B, Salazar A, Mico JA, Failde I. A review of chronic pain impact on patients, their social environment and the health care system. J Pain Res. 2016;9:457-467.
2. Institute of Medicine (US) Committee on Advancing Pain Research, Care, and Education. Relieving Pain in America: A Blueprint for Transforming Prevention, Care, Education, and Research. Washington, DC: Institute of Medicine; 2011.
3. Breuer B, Cruciani R, Portenoy RK. Pain management by primary care physicians, pain physicians, chiropractors, and acupuncturists: a national survey. South Med J. 2010;103(8):738-747.
4. Gatchel RJ, McGeary DD, McGeary CA, Lippe B. Interdisciplinary chronic pain management: past, present, and future. Am Psychol. 2014;69(2):119-130.
5. Meghani SH, Polomano RC, Tait RC, Vallerand AH, Anderson KO, Gallagher RM. Advancing a national agenda to eliminate disparities in pain care: directions for health policy, education, practice, and research. Pain Med. 2012;13(1):5-28.
6. McHugh RK, Nielsen S, Weiss RD. Prescription drug abuse: from epidemiology to public policy. J Subst Abuse Treat. 2015;48(1):1-7.
7. Scholl L, Seth P, Kariisa M, Wilson N, Baldwin G. Drug and opioid-involved overdose deaths-United States, 2013-2017. MMWR Morb Mortal Wkly Rep. 2018;67(5152):1419-1427.
8. Edlund MJ, Martin BC, Devries A, Fan MY, Braden JB, Sullivan MD. Trends in use of opioids for chronic noncancer pain among individuals with mental health and substance use disorders: the TROUP study. Clin J Pain. 2010;26(1):1-8.
9. Højsted J, Sjøgren P. Addiction to opioids in chronic pain patients: a literature review. Eur J Pain. 2007;11(5):490-518.
10. Seal KH, Shi Y, Cohen G, et al. Association of mental health disorders with prescription opioids and high-risk opioid use in US veterans of Iraq and Afghanistan. JAMA. 2012;307(9):940-947.
11. Kolodny A, Courtwright DT, Hwang CS, et al. The prescription opioid and heroin crisis: a public health approach to an epidemic of addiction. Annu Rev Public Health. 2015;36:559-574.
12. Ballantyne JC, Sullivan MD, Kolodny A. Opioid dependence vs addiction: a distinction without a difference? Arch Intern Med. 2012;172(17):1342-1343.
13. Levy B, Paulozzi L, Mack KA, Jones CM. Trends in opioid analgesic-prescribing rates by specialty, U.S., 2007-2012. Am J Prev Med. 2015;49(3):409-413.
14. Gellad WF, Good CB, Shulkin DJ. Addressing the opioid epidemic in the United States: lessons from the Department of Veterans Affairs. JAMA Intern Med. 2017;177(5):611-612.
15. US Department of Veterans Affairs. Veteran Health Administration Directive 2009-053, Pain Management. https://www.va.gov/painmanagement/docs/vha09paindirective.pdf. Published October 28, 2009. Accessed August 19, 2019.
16. Arora S, Geppert CM, Kalishman S, et al. Academic health center management of chronic diseases through knowledge networks: Project ECHO. Acad Med. 2007;82(2):154-160.
17. Kirsh S, Su GL, Sales A, Jain R. Access to outpatient specialty care: solutions from an integrated health care system. Am J Med Qual. 2015;30(1):88-90.
18. Frank JW, Carey EP, Fagan KM, et al. Evaluation of a telementoring intervention for pain management in the Veterans Health Administration. Pain Med. 2015;16(6):1090-1100.
19. Fillingim RB. Individual differences in pain: understanding the mosaic that makes pain personal. Pain. 2017;158 (suppl 1):S11-S18.
20. McLellan AT, Lewis DC, O’Brien CP, Kleber HD. Drug dependence, a chronic medical illness: implications for treatment, insurance, and outcomes evaluation. JAMA. 2000;284(13):1689-1695.
Chronic pain significantly affects 100 million Americans.1,2 Pain accounts for $560 to $635 billion in annual financial costs to society, including health care costs and loss of productivity (ie, days missed from work, hours of work lost, and lower wages).2,3 Although pain prevalence exceeds other chronic diseases, such as diabetes mellitus, cancer, and heart disease, it lacks a sufficient body of evidence-based research and guidelines on the underlying mechanisms, valid methods of assessment, and comparative effectiveness of treatments to effectively implement into clinical practice.2,4 Prevention and treatment of pain are often delayed, inaccessible, or inadequate.2 Primary care providers (PCPs) are most often sought for pain management and treat about 52% of chronic pain patients.2,3,5 Veterans are especially vulnerable to chronic pain and are at risk for inadequate treatment.2
Background
There is an epidemic of drug abuse and mortality from opioid prescription medication.6 In the US, rates of overdose deaths from prescription opioids were 6.1 per 100,000 for men and 4.2 per 100,000 for women in 2017. Opioids were involved in 47,600 overdose deaths in 2017, accounting for 67.8% of all drug overdose deaths.7
A large number of patients on long-term opioids have preexisting substance use disorders and/or psychiatric disease, further complicating chronic pain management.8-10 Prescription opioid use has been the precursor for about 80% of people who are now heroin addicts.11 Iatrogenic addiction from prescription medications isn’t easily captured by standard addiction criteria. Consequently, in patients who are on opioid therapy for prolonged periods, separating complex opioid dependence from addiction is difficult.12 Improved addiction screening and risk mitigation strategies are needed along with aggressive treatment monitoring to curb the opioid epidemic.
Opioid Management in Primary Care
The majority of opioid medications are prescribed by PCPs, which is magnified in the US Department of Veterans Affairs (VA) health care system due to the high prevalence of service-related injuries.3,13 The VA is at the forefront of addressing the complexities of opioid addiction through several initiatives.14 The ability to offer the frequent visits needed to safely manage patients prescribed opioids and the integration of mental health and addiction treatment are often lacking in non-VA primary care clinics. Therefore, a key to solving the opioid crisis is developing these capabilities so they can be delivered within the primary care setting. There is substantial evidence in support of nonopioid alternatives to chronic pain management, including other pharmacologic approaches, exercise, physical therapy, acupuncture, weight loss, smoking cessation, chiropractic care, cognitive behavioral therapy (CBT), and other integrative health modalities.
A 2009 VA directive mandated the development of a comprehensive, integrated, systemwide approach to pain management.15 The VA Stepped-Care Biopsychosocial Model for Pain Management is dependent on timely access to secondary consultation from pain medicine, behavioral health, physical medicine, and other specialty consultation.15
History of VHA SCAN-ECHO Model
The Specialty Care Access Network–Extension for Community Health Outcomes (SCAN-ECHO) is a Veterans Health Administration (VHA) adaptation of a program that originated at the University of Mexico.16,17 The SCAN-ECHO model uses a multisite videoconferencing network to provide specialty care consultations to PCPs and patient aligned care teams (PACTs). During the 60- to 90-minute weekly sessions, case presentations are analyzed in real time so that over time, the PCPs gain knowledge, competency, and confidence in learning how to handle complex chronic conditions.
Since its implementation, the SCAN-ECHO program has been adopted across the VHA in a variety of specialties. One program, the SCAN-ECHO for Pain Management (SCAN-ECHO-PM) was implemented in 7 VHA networks in 31 states, spanning 47 medical centers and 148 community-based outpatient clinics (CBOCs).18 The SCAN-ECHO-PM program successfully conducted 257 multidisciplinary pain consultations between 2011 and 2013, resulting in increased initiation of nonopioid medications.18
The aim of this article is to describe the implementation of a multicomponent primary care-based pain clinic with a fully integrated mental health service and addiction service at the North Florida/South Georgia Veterans Health System (NF/SGVHS). A practiced-based intervention of the biopsychosocial model with robust patient engagement has guided the development of the NF/SGVHS pain clinic (Figure 1).4,19
Pain CLinic
NF/SGVHS comprises the Malcom Randall and Lake City VA medical centers (VAMCs) hospitals, 3 satellite outpatient clinics, and 8 CBOCs. Spanning 33 counties in North Florida and 19 counties in South Georgia, the NF/SGVHS serves more than 140,000 patients. In 2010, the Malcom Randall VAMC established a multidisciplinary primary care pain clinic to manage veterans at high-risk for noncancer chronic pain and addiction. The noncancer pain policy was revised after garnering support from stakeholders who treat chronic pain, including the chiefs of psychiatry, rehabilitation medicine, neurosurgery, psychology, interventional pain, pharmacy, nursing, addiction medicine, and primary care. The clinic is staffed by primary care physicians trained in internal medicine and family medicine and is structured with 1-hour first visits, and 30-minute follow-up visits to allow enough time for comprehensive evaluation while meeting the needs for close follow-up support.
All physicians in the clinic have buprenorphine prescribing credentials to aid in the management of opioid addiction, as some patients feel more comfortable receiving addiction treatment in a primary care setting. The multimodal care model consists of several services that include addiction psychiatrists, interventional pain specialists, pain psychologists, and pain pharmacologists who coordinate the care to the veterans. The addiction psychiatrists offer a full range of services with inpatient residential and outpatient programs. Through recurring meetings with primary care pain clinic staff, the addiction psychiatrists are available to discuss use of buprenorphine and arrange follow-up for patients with complex pain addiction. There is ongoing collaboration to develop the best care plan that meets the patient’s needs for chronic pain, addiction, and/or mental health issues. The interventional pain service has 3 fellowship-trained pain care providers who deliver comprehensive evaluation, pharmacologic recommendations, and a full range of interventional and complementary therapies with an emphasis on objective functional improvement. Pain care providers offer alternatives to patients who are being weaned from opioids and support the multidisciplinary patient engagement model.
The pain psychology program, established in 2011, delivers CBT to 5 onsite locations and 5 telehealth locations. The service includes an advanced CBT program and a couples CBT program. The pharmacy pain fellowship program provides staff for an outpatient e-consult pain management service and an inpatient pharmacy consult service. Harnessing pain specialty pharmacists, the pharmacy service addresses pharmacokinetic issues, urine drug screen (UDS) results, opioid tapering and discharge planning for pain, addiction and mental health needs. The NF/SGVHS Primary Care Pain Clinic was established to support PCPs who did not feel comfortable managing chronic pain patients. These patients were typically on high-dose opioid therapy (> 100-mg morphine equivalent daily doses [MEDDs]); patients with a history of opioid addiction; patients with an addiction to opioids combined with benzodiazepines; and patients with comorbid medical issues (eg, sleep apnea), which complicated their management. The process of addressing opioid safety in these complex pain patients can be labor intensive and generally cannot be accomplished in a brief visit in a primary care setting where many other medical problems often need to be addressed.
Most patients on high-dose opioids are fearful of any changes in their medications. The difficult conversation regarding opioid safety is a lengthy one and frequently will occur over multiple visits. In addition, safely tapering opioids requires frequent follow-up to provide psychological support and to address withdrawal and mental health issues that may arise. As opioids are tapered, the clinic reinforces improved pain care through a multimodal biopsychosocial model. All veterans receiving pain care outside the VA are monitored annually to assure they are receiving evidence-based pain care as defined by the biopsychosocial model.
Education
Since 2011, the NF/SGVHS SCAN-ECHO pain and addiction educational forum has created > 50 hours of approved annual continuing medical education (CME) on pain management and addiction for PCPs. Initially, the 1-hour weekly educational audioconferences presented a pain management case along with related topics and involved specialists from interventional pain, physical therapy, psychiatry, nursing, neurology, and psychology departments. In 2013, in conjunction with the VA SCAN-ECHO program of Hunter Holmes McGuire VAMC in Richmond, Virginia, and Walter Reed National Military Medical Center in Bethesda, Maryland, the audioconference was expanded to 2 days each week with additional topics on addiction management. Residency and fellowship rotations were developed that specifically targeted fellows from psychiatry, pharmacology, and interventional pain departments.
Currently, an 8-session pain school is delivered onsite and at 7 telehealth locations. The school is a collaborative effort involving interventional pain, psychology, pharmacy, nutrition, and the primary care pain clinic staff. As the cornerstone of the program, the pain school stresses the biopsychosocial patient engagement model.
Program Evaluation
The VA is equipped with multiple telehealth service networks that allow for the delivery of programs, such as the pain school, a pain psychology program, and a yoga program, onsite or offsite. The VA Computerized Patient Record System (CPRS) manages electronic health records, allowing for rapid chart review and e-consults. The NF/SGVHS Pain Management Program provides about 1500 e-consults yearly. The CPRS includes templates with pain metrics to help PCPs deliver pain care more efficiently and evaluate performance measures. This system also allows for the capture of data to track improvements in the care of the veterans served.
From 2012 to 2017, more than 5000 NF/SGVHS patients were weaned from opioids. Overall, there was an 87% reduction in patients receiving opioids ( ≥ 100-mg MEDDs) within the NF/SGVHS, which is significantly more than the 49% seen nationally across the VHA (Figure 2). Percent reduction was calculated by taking the difference in number of patients receiving opioids in 2012 and 2017, dividing by the number of patients receiving opioids in 2012 and multiplying by 100. The largest proportion of opioid dose reductions for NF/SGVHS and VHA patients, respectively, were seen in 300-mg to 399-mg MEDDs (95% vs 67%, respectively); followed by ≥ 400-mg MEDDs (94% vs 71%, respectively); 200-mg to 299-mg MEDDs (91% vs 58%, respectively); and 100-mg to 199-mg MEDDs (84% vs 40%, respectively). When examining NF/SGVHS trends over time, there has been a consistent decline in patients prescribed opioids (18 223 in 2012 compared with 12 877 in 2017) with similar trends in benzodiazepine-opioid combination therapy (2694 in 2012 compared with 833 in 2017) (Figure 3).
Similar declines are seen when patients are stratified by the MEDD (Figure 4). From 2012 to 2017, 92% of the patients were successfully tapered off doses ≥ 400-mg MEDD (2012, n = 72; 2017, n = 6), and tapered off 300-mg to 399-mg MEDD (2012, n = 107; 2017, n = 5); 95% were tapered off 200-mg to 299-mg MEDD (2012, n = 262; 2017, n = 22); and 86% were tapered off 100-mg to 199-mg MEDD (2012, n = 876; 2017; n = 127).
Conclusion
Successful integration of primary care with mental health and addiction services is paramount to aggressively taper patients with chronic pain from opioids. There is evidence that drug dependence and chronic pain should be treated like other chronic illness.20 Both chronic pain and addiction can be treated with a multidimensional self-management approach. In view of the high incidence of mental health and addiction associated with opioid use, it makes sense that an integrated, 1-stop pain and addiction clinic that understands and addresses both issues is more likely to improve patient outcomes.
Acknowledgments
This material is the result of work supported by the resources and facilities at the North Florida/South Georgia Veterans Health System, Geriatric Research Education Clinical Center in Gainesville, Florida.
Chronic pain significantly affects 100 million Americans.1,2 Pain accounts for $560 to $635 billion in annual financial costs to society, including health care costs and loss of productivity (ie, days missed from work, hours of work lost, and lower wages).2,3 Although pain prevalence exceeds other chronic diseases, such as diabetes mellitus, cancer, and heart disease, it lacks a sufficient body of evidence-based research and guidelines on the underlying mechanisms, valid methods of assessment, and comparative effectiveness of treatments to effectively implement into clinical practice.2,4 Prevention and treatment of pain are often delayed, inaccessible, or inadequate.2 Primary care providers (PCPs) are most often sought for pain management and treat about 52% of chronic pain patients.2,3,5 Veterans are especially vulnerable to chronic pain and are at risk for inadequate treatment.2
Background
There is an epidemic of drug abuse and mortality from opioid prescription medication.6 In the US, rates of overdose deaths from prescription opioids were 6.1 per 100,000 for men and 4.2 per 100,000 for women in 2017. Opioids were involved in 47,600 overdose deaths in 2017, accounting for 67.8% of all drug overdose deaths.7
A large number of patients on long-term opioids have preexisting substance use disorders and/or psychiatric disease, further complicating chronic pain management.8-10 Prescription opioid use has been the precursor for about 80% of people who are now heroin addicts.11 Iatrogenic addiction from prescription medications isn’t easily captured by standard addiction criteria. Consequently, in patients who are on opioid therapy for prolonged periods, separating complex opioid dependence from addiction is difficult.12 Improved addiction screening and risk mitigation strategies are needed along with aggressive treatment monitoring to curb the opioid epidemic.
Opioid Management in Primary Care
The majority of opioid medications are prescribed by PCPs, which is magnified in the US Department of Veterans Affairs (VA) health care system due to the high prevalence of service-related injuries.3,13 The VA is at the forefront of addressing the complexities of opioid addiction through several initiatives.14 The ability to offer the frequent visits needed to safely manage patients prescribed opioids and the integration of mental health and addiction treatment are often lacking in non-VA primary care clinics. Therefore, a key to solving the opioid crisis is developing these capabilities so they can be delivered within the primary care setting. There is substantial evidence in support of nonopioid alternatives to chronic pain management, including other pharmacologic approaches, exercise, physical therapy, acupuncture, weight loss, smoking cessation, chiropractic care, cognitive behavioral therapy (CBT), and other integrative health modalities.
A 2009 VA directive mandated the development of a comprehensive, integrated, systemwide approach to pain management.15 The VA Stepped-Care Biopsychosocial Model for Pain Management is dependent on timely access to secondary consultation from pain medicine, behavioral health, physical medicine, and other specialty consultation.15
History of VHA SCAN-ECHO Model
The Specialty Care Access Network–Extension for Community Health Outcomes (SCAN-ECHO) is a Veterans Health Administration (VHA) adaptation of a program that originated at the University of Mexico.16,17 The SCAN-ECHO model uses a multisite videoconferencing network to provide specialty care consultations to PCPs and patient aligned care teams (PACTs). During the 60- to 90-minute weekly sessions, case presentations are analyzed in real time so that over time, the PCPs gain knowledge, competency, and confidence in learning how to handle complex chronic conditions.
Since its implementation, the SCAN-ECHO program has been adopted across the VHA in a variety of specialties. One program, the SCAN-ECHO for Pain Management (SCAN-ECHO-PM) was implemented in 7 VHA networks in 31 states, spanning 47 medical centers and 148 community-based outpatient clinics (CBOCs).18 The SCAN-ECHO-PM program successfully conducted 257 multidisciplinary pain consultations between 2011 and 2013, resulting in increased initiation of nonopioid medications.18
The aim of this article is to describe the implementation of a multicomponent primary care-based pain clinic with a fully integrated mental health service and addiction service at the North Florida/South Georgia Veterans Health System (NF/SGVHS). A practiced-based intervention of the biopsychosocial model with robust patient engagement has guided the development of the NF/SGVHS pain clinic (Figure 1).4,19
Pain CLinic
NF/SGVHS comprises the Malcom Randall and Lake City VA medical centers (VAMCs) hospitals, 3 satellite outpatient clinics, and 8 CBOCs. Spanning 33 counties in North Florida and 19 counties in South Georgia, the NF/SGVHS serves more than 140,000 patients. In 2010, the Malcom Randall VAMC established a multidisciplinary primary care pain clinic to manage veterans at high-risk for noncancer chronic pain and addiction. The noncancer pain policy was revised after garnering support from stakeholders who treat chronic pain, including the chiefs of psychiatry, rehabilitation medicine, neurosurgery, psychology, interventional pain, pharmacy, nursing, addiction medicine, and primary care. The clinic is staffed by primary care physicians trained in internal medicine and family medicine and is structured with 1-hour first visits, and 30-minute follow-up visits to allow enough time for comprehensive evaluation while meeting the needs for close follow-up support.
All physicians in the clinic have buprenorphine prescribing credentials to aid in the management of opioid addiction, as some patients feel more comfortable receiving addiction treatment in a primary care setting. The multimodal care model consists of several services that include addiction psychiatrists, interventional pain specialists, pain psychologists, and pain pharmacologists who coordinate the care to the veterans. The addiction psychiatrists offer a full range of services with inpatient residential and outpatient programs. Through recurring meetings with primary care pain clinic staff, the addiction psychiatrists are available to discuss use of buprenorphine and arrange follow-up for patients with complex pain addiction. There is ongoing collaboration to develop the best care plan that meets the patient’s needs for chronic pain, addiction, and/or mental health issues. The interventional pain service has 3 fellowship-trained pain care providers who deliver comprehensive evaluation, pharmacologic recommendations, and a full range of interventional and complementary therapies with an emphasis on objective functional improvement. Pain care providers offer alternatives to patients who are being weaned from opioids and support the multidisciplinary patient engagement model.
The pain psychology program, established in 2011, delivers CBT to 5 onsite locations and 5 telehealth locations. The service includes an advanced CBT program and a couples CBT program. The pharmacy pain fellowship program provides staff for an outpatient e-consult pain management service and an inpatient pharmacy consult service. Harnessing pain specialty pharmacists, the pharmacy service addresses pharmacokinetic issues, urine drug screen (UDS) results, opioid tapering and discharge planning for pain, addiction and mental health needs. The NF/SGVHS Primary Care Pain Clinic was established to support PCPs who did not feel comfortable managing chronic pain patients. These patients were typically on high-dose opioid therapy (> 100-mg morphine equivalent daily doses [MEDDs]); patients with a history of opioid addiction; patients with an addiction to opioids combined with benzodiazepines; and patients with comorbid medical issues (eg, sleep apnea), which complicated their management. The process of addressing opioid safety in these complex pain patients can be labor intensive and generally cannot be accomplished in a brief visit in a primary care setting where many other medical problems often need to be addressed.
Most patients on high-dose opioids are fearful of any changes in their medications. The difficult conversation regarding opioid safety is a lengthy one and frequently will occur over multiple visits. In addition, safely tapering opioids requires frequent follow-up to provide psychological support and to address withdrawal and mental health issues that may arise. As opioids are tapered, the clinic reinforces improved pain care through a multimodal biopsychosocial model. All veterans receiving pain care outside the VA are monitored annually to assure they are receiving evidence-based pain care as defined by the biopsychosocial model.
Education
Since 2011, the NF/SGVHS SCAN-ECHO pain and addiction educational forum has created > 50 hours of approved annual continuing medical education (CME) on pain management and addiction for PCPs. Initially, the 1-hour weekly educational audioconferences presented a pain management case along with related topics and involved specialists from interventional pain, physical therapy, psychiatry, nursing, neurology, and psychology departments. In 2013, in conjunction with the VA SCAN-ECHO program of Hunter Holmes McGuire VAMC in Richmond, Virginia, and Walter Reed National Military Medical Center in Bethesda, Maryland, the audioconference was expanded to 2 days each week with additional topics on addiction management. Residency and fellowship rotations were developed that specifically targeted fellows from psychiatry, pharmacology, and interventional pain departments.
Currently, an 8-session pain school is delivered onsite and at 7 telehealth locations. The school is a collaborative effort involving interventional pain, psychology, pharmacy, nutrition, and the primary care pain clinic staff. As the cornerstone of the program, the pain school stresses the biopsychosocial patient engagement model.
Program Evaluation
The VA is equipped with multiple telehealth service networks that allow for the delivery of programs, such as the pain school, a pain psychology program, and a yoga program, onsite or offsite. The VA Computerized Patient Record System (CPRS) manages electronic health records, allowing for rapid chart review and e-consults. The NF/SGVHS Pain Management Program provides about 1500 e-consults yearly. The CPRS includes templates with pain metrics to help PCPs deliver pain care more efficiently and evaluate performance measures. This system also allows for the capture of data to track improvements in the care of the veterans served.
From 2012 to 2017, more than 5000 NF/SGVHS patients were weaned from opioids. Overall, there was an 87% reduction in patients receiving opioids ( ≥ 100-mg MEDDs) within the NF/SGVHS, which is significantly more than the 49% seen nationally across the VHA (Figure 2). Percent reduction was calculated by taking the difference in number of patients receiving opioids in 2012 and 2017, dividing by the number of patients receiving opioids in 2012 and multiplying by 100. The largest proportion of opioid dose reductions for NF/SGVHS and VHA patients, respectively, were seen in 300-mg to 399-mg MEDDs (95% vs 67%, respectively); followed by ≥ 400-mg MEDDs (94% vs 71%, respectively); 200-mg to 299-mg MEDDs (91% vs 58%, respectively); and 100-mg to 199-mg MEDDs (84% vs 40%, respectively). When examining NF/SGVHS trends over time, there has been a consistent decline in patients prescribed opioids (18 223 in 2012 compared with 12 877 in 2017) with similar trends in benzodiazepine-opioid combination therapy (2694 in 2012 compared with 833 in 2017) (Figure 3).
Similar declines are seen when patients are stratified by the MEDD (Figure 4). From 2012 to 2017, 92% of the patients were successfully tapered off doses ≥ 400-mg MEDD (2012, n = 72; 2017, n = 6), and tapered off 300-mg to 399-mg MEDD (2012, n = 107; 2017, n = 5); 95% were tapered off 200-mg to 299-mg MEDD (2012, n = 262; 2017, n = 22); and 86% were tapered off 100-mg to 199-mg MEDD (2012, n = 876; 2017; n = 127).
Conclusion
Successful integration of primary care with mental health and addiction services is paramount to aggressively taper patients with chronic pain from opioids. There is evidence that drug dependence and chronic pain should be treated like other chronic illness.20 Both chronic pain and addiction can be treated with a multidimensional self-management approach. In view of the high incidence of mental health and addiction associated with opioid use, it makes sense that an integrated, 1-stop pain and addiction clinic that understands and addresses both issues is more likely to improve patient outcomes.
Acknowledgments
This material is the result of work supported by the resources and facilities at the North Florida/South Georgia Veterans Health System, Geriatric Research Education Clinical Center in Gainesville, Florida.
1. Dueñas M, Ojeda B, Salazar A, Mico JA, Failde I. A review of chronic pain impact on patients, their social environment and the health care system. J Pain Res. 2016;9:457-467.
2. Institute of Medicine (US) Committee on Advancing Pain Research, Care, and Education. Relieving Pain in America: A Blueprint for Transforming Prevention, Care, Education, and Research. Washington, DC: Institute of Medicine; 2011.
3. Breuer B, Cruciani R, Portenoy RK. Pain management by primary care physicians, pain physicians, chiropractors, and acupuncturists: a national survey. South Med J. 2010;103(8):738-747.
4. Gatchel RJ, McGeary DD, McGeary CA, Lippe B. Interdisciplinary chronic pain management: past, present, and future. Am Psychol. 2014;69(2):119-130.
5. Meghani SH, Polomano RC, Tait RC, Vallerand AH, Anderson KO, Gallagher RM. Advancing a national agenda to eliminate disparities in pain care: directions for health policy, education, practice, and research. Pain Med. 2012;13(1):5-28.
6. McHugh RK, Nielsen S, Weiss RD. Prescription drug abuse: from epidemiology to public policy. J Subst Abuse Treat. 2015;48(1):1-7.
7. Scholl L, Seth P, Kariisa M, Wilson N, Baldwin G. Drug and opioid-involved overdose deaths-United States, 2013-2017. MMWR Morb Mortal Wkly Rep. 2018;67(5152):1419-1427.
8. Edlund MJ, Martin BC, Devries A, Fan MY, Braden JB, Sullivan MD. Trends in use of opioids for chronic noncancer pain among individuals with mental health and substance use disorders: the TROUP study. Clin J Pain. 2010;26(1):1-8.
9. Højsted J, Sjøgren P. Addiction to opioids in chronic pain patients: a literature review. Eur J Pain. 2007;11(5):490-518.
10. Seal KH, Shi Y, Cohen G, et al. Association of mental health disorders with prescription opioids and high-risk opioid use in US veterans of Iraq and Afghanistan. JAMA. 2012;307(9):940-947.
11. Kolodny A, Courtwright DT, Hwang CS, et al. The prescription opioid and heroin crisis: a public health approach to an epidemic of addiction. Annu Rev Public Health. 2015;36:559-574.
12. Ballantyne JC, Sullivan MD, Kolodny A. Opioid dependence vs addiction: a distinction without a difference? Arch Intern Med. 2012;172(17):1342-1343.
13. Levy B, Paulozzi L, Mack KA, Jones CM. Trends in opioid analgesic-prescribing rates by specialty, U.S., 2007-2012. Am J Prev Med. 2015;49(3):409-413.
14. Gellad WF, Good CB, Shulkin DJ. Addressing the opioid epidemic in the United States: lessons from the Department of Veterans Affairs. JAMA Intern Med. 2017;177(5):611-612.
15. US Department of Veterans Affairs. Veteran Health Administration Directive 2009-053, Pain Management. https://www.va.gov/painmanagement/docs/vha09paindirective.pdf. Published October 28, 2009. Accessed August 19, 2019.
16. Arora S, Geppert CM, Kalishman S, et al. Academic health center management of chronic diseases through knowledge networks: Project ECHO. Acad Med. 2007;82(2):154-160.
17. Kirsh S, Su GL, Sales A, Jain R. Access to outpatient specialty care: solutions from an integrated health care system. Am J Med Qual. 2015;30(1):88-90.
18. Frank JW, Carey EP, Fagan KM, et al. Evaluation of a telementoring intervention for pain management in the Veterans Health Administration. Pain Med. 2015;16(6):1090-1100.
19. Fillingim RB. Individual differences in pain: understanding the mosaic that makes pain personal. Pain. 2017;158 (suppl 1):S11-S18.
20. McLellan AT, Lewis DC, O’Brien CP, Kleber HD. Drug dependence, a chronic medical illness: implications for treatment, insurance, and outcomes evaluation. JAMA. 2000;284(13):1689-1695.
1. Dueñas M, Ojeda B, Salazar A, Mico JA, Failde I. A review of chronic pain impact on patients, their social environment and the health care system. J Pain Res. 2016;9:457-467.
2. Institute of Medicine (US) Committee on Advancing Pain Research, Care, and Education. Relieving Pain in America: A Blueprint for Transforming Prevention, Care, Education, and Research. Washington, DC: Institute of Medicine; 2011.
3. Breuer B, Cruciani R, Portenoy RK. Pain management by primary care physicians, pain physicians, chiropractors, and acupuncturists: a national survey. South Med J. 2010;103(8):738-747.
4. Gatchel RJ, McGeary DD, McGeary CA, Lippe B. Interdisciplinary chronic pain management: past, present, and future. Am Psychol. 2014;69(2):119-130.
5. Meghani SH, Polomano RC, Tait RC, Vallerand AH, Anderson KO, Gallagher RM. Advancing a national agenda to eliminate disparities in pain care: directions for health policy, education, practice, and research. Pain Med. 2012;13(1):5-28.
6. McHugh RK, Nielsen S, Weiss RD. Prescription drug abuse: from epidemiology to public policy. J Subst Abuse Treat. 2015;48(1):1-7.
7. Scholl L, Seth P, Kariisa M, Wilson N, Baldwin G. Drug and opioid-involved overdose deaths-United States, 2013-2017. MMWR Morb Mortal Wkly Rep. 2018;67(5152):1419-1427.
8. Edlund MJ, Martin BC, Devries A, Fan MY, Braden JB, Sullivan MD. Trends in use of opioids for chronic noncancer pain among individuals with mental health and substance use disorders: the TROUP study. Clin J Pain. 2010;26(1):1-8.
9. Højsted J, Sjøgren P. Addiction to opioids in chronic pain patients: a literature review. Eur J Pain. 2007;11(5):490-518.
10. Seal KH, Shi Y, Cohen G, et al. Association of mental health disorders with prescription opioids and high-risk opioid use in US veterans of Iraq and Afghanistan. JAMA. 2012;307(9):940-947.
11. Kolodny A, Courtwright DT, Hwang CS, et al. The prescription opioid and heroin crisis: a public health approach to an epidemic of addiction. Annu Rev Public Health. 2015;36:559-574.
12. Ballantyne JC, Sullivan MD, Kolodny A. Opioid dependence vs addiction: a distinction without a difference? Arch Intern Med. 2012;172(17):1342-1343.
13. Levy B, Paulozzi L, Mack KA, Jones CM. Trends in opioid analgesic-prescribing rates by specialty, U.S., 2007-2012. Am J Prev Med. 2015;49(3):409-413.
14. Gellad WF, Good CB, Shulkin DJ. Addressing the opioid epidemic in the United States: lessons from the Department of Veterans Affairs. JAMA Intern Med. 2017;177(5):611-612.
15. US Department of Veterans Affairs. Veteran Health Administration Directive 2009-053, Pain Management. https://www.va.gov/painmanagement/docs/vha09paindirective.pdf. Published October 28, 2009. Accessed August 19, 2019.
16. Arora S, Geppert CM, Kalishman S, et al. Academic health center management of chronic diseases through knowledge networks: Project ECHO. Acad Med. 2007;82(2):154-160.
17. Kirsh S, Su GL, Sales A, Jain R. Access to outpatient specialty care: solutions from an integrated health care system. Am J Med Qual. 2015;30(1):88-90.
18. Frank JW, Carey EP, Fagan KM, et al. Evaluation of a telementoring intervention for pain management in the Veterans Health Administration. Pain Med. 2015;16(6):1090-1100.
19. Fillingim RB. Individual differences in pain: understanding the mosaic that makes pain personal. Pain. 2017;158 (suppl 1):S11-S18.
20. McLellan AT, Lewis DC, O’Brien CP, Kleber HD. Drug dependence, a chronic medical illness: implications for treatment, insurance, and outcomes evaluation. JAMA. 2000;284(13):1689-1695.
Use of Mobile Messaging System for Self-Management of Chemotherapy Symptoms in Patients with Advanced Cancer (FULL)
Cancer and cancer-related treatment can cause a myriad of adverse effects.1,2 Early identification and management of these symptoms is paramount to the success of cancer treatment completion; however, clinic and telephonic strategies for addressing symptoms often result in delays in care.1 New strategies for patient engagement in the management of cancer and treatment-related symptoms are needed.
The use of online self-management tools can result in improvement in symptoms, reduce cancer symptom distress, improve quality-of-life, and improve medication adherence.3-9 A meta-analysis concluded that online interventions showed promise, but optimizing interventions would require additional research.10 Another meta-analysis found that online self-management was effective in managing several symptoms.11 An e-health method of collecting patient self-reported symptoms has been found to be acceptable to patients and feasible for use.12-14 We postulated that a mobile text messaging strategy may be an effective modality for augmenting symptom management for cancer patients in real time.
In the US Departmant of Veterans Affairs (VA), “Annie,” a self-care tool utilizing a text-messaging system has been implemented. Annie was developed modeling “Flo,” a messaging system in the United Kingdom that has been used for case management of chronic obstructive pulmonary disease, heart failure, stress incontinence, asthma, as a medication reminder tool, and to provide support for weight loss or post-operatively.15-17 Using Annie in the US, veterans have the ability to receive and track health information. Use of the Annie program has demonstrated improved continuous positive airway pressure monitor utilization in veterans with traumatic brain injury.18 Other uses within the Veterans Health Administration (VHA) include assisting patients with anger management, liver disease, anxiety, asthma, diabetes, HIV, hypertension, weight loss, and smoking cessation.
Methods
The Hematology/Oncology division of the Minneapolis VA Healthcare System (MVAHCS) is a tertiary care facility that administers about 260 new chemotherapy regimens annually. The MVAHCS interdisciplinary hematology/oncology group initiated a quality improvement project to determine the feasibility, acceptability, and experience of tailoring the Annie tool for self-management of cancer symptoms. The group consisted of 2 physicians, 3 advanced practice registered nurses, 1 physician assistant, 2 registered nurses, and 2 Annie program team members.
We first created a symptom management pilot protocol as a result of multidisciplinary team discussions. Examples of discussion points for consideration included, but were not limited to, timing of texts, amount of information to ask for and provide, what potential symptoms to consider, and which patient population to pilot first.
The initial protocol was agreed upon and is as follows: Patients were sent text messages twice daily Monday through Friday, and asked to rate 2 symptoms per day, using a severity scale of 0 to 4 (absent, mild, moderate, severe, or disabling): nausea/vomiting, mouth sores, fatigue (Figure 1), trouble breathing, appetite, constipation, diarrhea (Figure 2), numbness/tingling, pain. In addition, patients were asked whether they had had a fever or not. Based on their response to the symptom inquiries, the patient received an automated text response. The text may have provided positive affirmation that they were doing well, given them advice for home management, referred them to an educational hyperlink, asked them to call a direct number to the clinic, or instructed them to report directly to the emergency department (ED). Patients could input a particular symptom on any day, even if they were not specifically asked about that symptom on that day. Patients also were instructed to text, only if it was not an inconvenience to them, as we wanted the intervention to be helpful and not a burden.
Results
Through screening new patient consults or those referred for chemotherapy education, 15 male veterans enrolled in the symptom monitoring program over an 8 month period. There were additional patients who were not offered the program or chose not to participate; often due to not having texting capabilities on their phone or not liking the texting feature. The majority of those who participated in the program (n = 14) were enrolled at the start of Cycle 1; the other patient was enrolled at the start of Cycle 2. Patients were enrolled an average of 89 days (range 8-204). Average response rate was 84.2% (range 30-100%).
Although symptoms were not reviewed in real time, we reviewed responses to determine the utilization of the instructions given for the program. No veteran had 0 symptoms reported. There were numerous occurrences of a score of 1 or 2. Many of these patients had baseline symptoms due to their underlying cancer. A score of 3 or 4 on the system prompted the patient to call the clinic or go to the ED. Seven patients (some with multiple occurrences) were prompted to call; only 4 of these made the follow-up call to the clinic. All were offered a same day visit, but each declined. Only 1 patient reported a symptom on a day not prompted for that symptom. Symptoms that were reported are listed in order of frequency: fatigue, appetite loss, numbness, pain, mouth sore, and breathing difficulty. There were no visits to the ED.
Program Evaluation
An evaluation was conducted 30 to 60 days after program enrollment. We elicited feedback to determine who was reading and responding to the text message: the patient, a family member, or a caregiver; whether they found the prompts helpful and took action; how they felt about the number of texts; if they felt the program was helpful; and any other feedback that would improve the program. In general, the patients (8) answered the texts independently. In 4 cases, the spouse answered the texts, and 3 patients answered the texts together with their spouses. Most patients (11) found the amount of texting to be “just right.” However, 3 found it to be too many texts and 1 didn’t find the amount of texting to be enough.
Three veterans did not have enough symptoms to feel the program was of benefit to them, but they did feel it would have been helpful if they had been more symptomatic. One veteran recalled taking loperamide as needed, as a result of prompting. No veterans felt as though the texting feature was difficult to use; and overall, were very positive about the program. Several appreciated receiving messages that validated when they were doing well, and they felt empowered by self-management. One of the spouses was a registered nurse and found the information too basic to be of use.
Discussion
Initial evaluation of the program via survey found no technology challenges. Patients have been very positive about the program including ease of use, appreciation of messages that validated when they were doing well, empowerment of self-management, and some utilization of the texting advice for symptom management. Educational hyperlinks for constipation, fatigue, diarrhea, and nausea/vomiting were added after this evaluation, and patients felt that these additions provided a higher level of education.
Staff time for this intervention was minimal. A nurse navigator offered the texting program to the patient during chemotherapy education, along with some instructions, which generally took about 5 minutes. One of the Annie program staff enrolled the patient. From that point forward, this was a self-management tool, beyond checking to ensure that the patient was successful in starting the program and evaluating use for the purposes of this quality improvement project. This self-management tool did not replace any other mechanism that a patient would normally have in our department for seeking help for symptoms. The MVAHSC typical process for symptom management is to have patients call a 24/7 nurse line. If the triage nurse feels the symptoms are related to the patient’s cancer or cancer treatment, they are referred to the physician assistant who is assigned to take those calls and has the option to see the patient the same day. Patients could continue to call the nurse line or speak with providers at the next appointment at their discretion.
Conclusion
Although Annie has the option of using either text messaging or a mobile application, this project only utilized text messaging. The study by Basch and colleagues was the closest randomized trial we could identify to compare to our quality improvement intervention.5 The 2 main, distinct differences were that Basch and colleagues utilized online monitoring; and nurses were utilized to screen and intervene on responses, as appropriate.
The ability of our program to text patients without the use of an application or tablet, may enable more patients to participate due to ease of use. There would be no increased in expected workload for clinical staff, and may lead to decreased call burden. Since our program is automated, while still providing patients with the option to call and speak with a staff member as needed, this is a cost-effective, first-line option for symptom management for those experiencing cancer-related symptoms. We believe this text messaging tool can have system wide use and benefit throughout the VHA.
1. Bruera E, Dev R. Overview of managing common non-pain symptoms in palliative care. https://www.uptodate.com/contents/overview-of-managing-common-non-pain-symptoms-in-palliative-care. Updated June 12, 2019. Accessed July 18, 2019.
2. Pirschel C. The crucial role of symptom management in cancer care. https://voice.ons.org/news-and-views/the-crucial-role-of-symptom-management-in-cancer-care. Published December 14, 2017. Accessed July 18, 2019.
3. Adam R, Burton CD, Bond CM, de Bruin M, Murchie P. Can patient-reported measurements of pain be used to improve cancer pain management? A systematic review and meta-analysis. BMJ Support Palliat Care. 2017;7(4):373-382.
4. Basch E, Deal AM, Kris MG, et al. Symptom monitoring with patient-reported outcomes during routine cancer treatment: a randomized controlled trial. J Clin Oncol. 2016;34(6):557-565.
5. Berry DL, Blonquist TM, Patel RA, Halpenny B, McReynolds J. Exposure to a patient-centered, Web-based intervention for managing cancer symptom and quality of life issues: Impact on symptom distress. J Med Internet Res. 2015;3(7):e136.
6. Kolb NA, Smith AG, Singleton JR, et al. Chemotherapy-related neuropathic symptom management: a randomized trial of an automated symptom-monitoring system paired with nurse practitioner follow-up. Support Care Cancer. 2018;26(5):1607-1615
7. Kamdar MM, Centi AJ, Fischer N, Jetwani K. A randomized controlled trial of a novel artificial-intelligence based smartphone application to optimize the management of cancer-related pain. Presented at: 2018 Palliative and Supportive Care in Oncology Symposium; November 16-17, 2018; San Diego, CA.
8. Mooney KH, Beck SL, Wong B, et al. Automated home monitoring and management of patient-reported symptoms during chemotherapy: results of the symptom care at home RCT. Cancer Med. 2017;6(3):537-546.
9. Spoelstra SL, Given CW, Sikorskii A, et al. Proof of concept of a mobile health short message service text message intervention that promotes adherence to oral anticancer agent medications: a randomized controlled trial. Telemed J E Health. 2016;22(6):497-506.
10. Fridriksdottir N, Gunnarsdottir S, Zoëga S, Ingadottir B, Hafsteinsdottir EJG. Effects of web-based interventions on cancer patients’ symptoms: review of randomized trials. Support Care Cancer. 2018;26(2):3370-351.
11. Kim AR, Park HA. Web-based self-management support intervention for cancer survivors: a systematic review and meta-analysis. Stud Health Technol Inform. 2015;216:142-147.
12. Girgis A, Durcinoska I, Levesque JV, et al; PROMPT-Care Program Group. eHealth system for collecting and utilizing patient reported outcome measures for personalized treatment and care (PROMPT-Care) among cancer patients: mixed methods approach to evaluate feasibility and acceptability. J Med Internet Res. 2017;19(10):e330.
13. Moradian S, Krzyzanowska MK, Maguire R, et al. Usability evaluation of a mobile phone-based system for remote monitoring and management of chemotherapy-related side effects in cancer patients: Mixed methods study. JMIR Cancer. 2018;4(2): e10932.
14. Voruganti T, Grunfeld E, Jamieson T, et al. My team of care study: a pilot randomized controlled trial of a web-based communication tool for collaborative care in patients with advanced cancer. J Med Internet Res. 2017;19(7):e219.
15. The Health Foundation. Overview of Florence simple telehealth text messaging system. https://www.health.org.uk/article/overview-of-the-florence-simple-telehealth-text-messaging-system. Accessed July 31, 2019.
16. Bragg DD, Edis H, Clark S, Parsons SL, Perumpalath B…Maxwell-Armstrong CA. Development of a telehealth monitoring service after colorectal surgery: a feasibility study. 2017;9(9):193-199.
17. O’Connell P. Annie-the VA’s self-care game changer. http://www.simple.uk.net/home/blog/blogcontent/annie-thevasself-caregamechanger. Published April 21, 2016. Accessed August 2, 2019.
18. Kataria L, Sundahl, C, Skalina L, et al. Text message reminders and intensive education improves positive airway pressure compliance and cognition in veterans with traumatic brain injury and obstructive sleep apnea: ANNIE pilot study (P1.097). Neurology, 2018; 90(suppl 15):P1.097.
Cancer and cancer-related treatment can cause a myriad of adverse effects.1,2 Early identification and management of these symptoms is paramount to the success of cancer treatment completion; however, clinic and telephonic strategies for addressing symptoms often result in delays in care.1 New strategies for patient engagement in the management of cancer and treatment-related symptoms are needed.
The use of online self-management tools can result in improvement in symptoms, reduce cancer symptom distress, improve quality-of-life, and improve medication adherence.3-9 A meta-analysis concluded that online interventions showed promise, but optimizing interventions would require additional research.10 Another meta-analysis found that online self-management was effective in managing several symptoms.11 An e-health method of collecting patient self-reported symptoms has been found to be acceptable to patients and feasible for use.12-14 We postulated that a mobile text messaging strategy may be an effective modality for augmenting symptom management for cancer patients in real time.
In the US Departmant of Veterans Affairs (VA), “Annie,” a self-care tool utilizing a text-messaging system has been implemented. Annie was developed modeling “Flo,” a messaging system in the United Kingdom that has been used for case management of chronic obstructive pulmonary disease, heart failure, stress incontinence, asthma, as a medication reminder tool, and to provide support for weight loss or post-operatively.15-17 Using Annie in the US, veterans have the ability to receive and track health information. Use of the Annie program has demonstrated improved continuous positive airway pressure monitor utilization in veterans with traumatic brain injury.18 Other uses within the Veterans Health Administration (VHA) include assisting patients with anger management, liver disease, anxiety, asthma, diabetes, HIV, hypertension, weight loss, and smoking cessation.
Methods
The Hematology/Oncology division of the Minneapolis VA Healthcare System (MVAHCS) is a tertiary care facility that administers about 260 new chemotherapy regimens annually. The MVAHCS interdisciplinary hematology/oncology group initiated a quality improvement project to determine the feasibility, acceptability, and experience of tailoring the Annie tool for self-management of cancer symptoms. The group consisted of 2 physicians, 3 advanced practice registered nurses, 1 physician assistant, 2 registered nurses, and 2 Annie program team members.
We first created a symptom management pilot protocol as a result of multidisciplinary team discussions. Examples of discussion points for consideration included, but were not limited to, timing of texts, amount of information to ask for and provide, what potential symptoms to consider, and which patient population to pilot first.
The initial protocol was agreed upon and is as follows: Patients were sent text messages twice daily Monday through Friday, and asked to rate 2 symptoms per day, using a severity scale of 0 to 4 (absent, mild, moderate, severe, or disabling): nausea/vomiting, mouth sores, fatigue (Figure 1), trouble breathing, appetite, constipation, diarrhea (Figure 2), numbness/tingling, pain. In addition, patients were asked whether they had had a fever or not. Based on their response to the symptom inquiries, the patient received an automated text response. The text may have provided positive affirmation that they were doing well, given them advice for home management, referred them to an educational hyperlink, asked them to call a direct number to the clinic, or instructed them to report directly to the emergency department (ED). Patients could input a particular symptom on any day, even if they were not specifically asked about that symptom on that day. Patients also were instructed to text, only if it was not an inconvenience to them, as we wanted the intervention to be helpful and not a burden.
Results
Through screening new patient consults or those referred for chemotherapy education, 15 male veterans enrolled in the symptom monitoring program over an 8 month period. There were additional patients who were not offered the program or chose not to participate; often due to not having texting capabilities on their phone or not liking the texting feature. The majority of those who participated in the program (n = 14) were enrolled at the start of Cycle 1; the other patient was enrolled at the start of Cycle 2. Patients were enrolled an average of 89 days (range 8-204). Average response rate was 84.2% (range 30-100%).
Although symptoms were not reviewed in real time, we reviewed responses to determine the utilization of the instructions given for the program. No veteran had 0 symptoms reported. There were numerous occurrences of a score of 1 or 2. Many of these patients had baseline symptoms due to their underlying cancer. A score of 3 or 4 on the system prompted the patient to call the clinic or go to the ED. Seven patients (some with multiple occurrences) were prompted to call; only 4 of these made the follow-up call to the clinic. All were offered a same day visit, but each declined. Only 1 patient reported a symptom on a day not prompted for that symptom. Symptoms that were reported are listed in order of frequency: fatigue, appetite loss, numbness, pain, mouth sore, and breathing difficulty. There were no visits to the ED.
Program Evaluation
An evaluation was conducted 30 to 60 days after program enrollment. We elicited feedback to determine who was reading and responding to the text message: the patient, a family member, or a caregiver; whether they found the prompts helpful and took action; how they felt about the number of texts; if they felt the program was helpful; and any other feedback that would improve the program. In general, the patients (8) answered the texts independently. In 4 cases, the spouse answered the texts, and 3 patients answered the texts together with their spouses. Most patients (11) found the amount of texting to be “just right.” However, 3 found it to be too many texts and 1 didn’t find the amount of texting to be enough.
Three veterans did not have enough symptoms to feel the program was of benefit to them, but they did feel it would have been helpful if they had been more symptomatic. One veteran recalled taking loperamide as needed, as a result of prompting. No veterans felt as though the texting feature was difficult to use; and overall, were very positive about the program. Several appreciated receiving messages that validated when they were doing well, and they felt empowered by self-management. One of the spouses was a registered nurse and found the information too basic to be of use.
Discussion
Initial evaluation of the program via survey found no technology challenges. Patients have been very positive about the program including ease of use, appreciation of messages that validated when they were doing well, empowerment of self-management, and some utilization of the texting advice for symptom management. Educational hyperlinks for constipation, fatigue, diarrhea, and nausea/vomiting were added after this evaluation, and patients felt that these additions provided a higher level of education.
Staff time for this intervention was minimal. A nurse navigator offered the texting program to the patient during chemotherapy education, along with some instructions, which generally took about 5 minutes. One of the Annie program staff enrolled the patient. From that point forward, this was a self-management tool, beyond checking to ensure that the patient was successful in starting the program and evaluating use for the purposes of this quality improvement project. This self-management tool did not replace any other mechanism that a patient would normally have in our department for seeking help for symptoms. The MVAHSC typical process for symptom management is to have patients call a 24/7 nurse line. If the triage nurse feels the symptoms are related to the patient’s cancer or cancer treatment, they are referred to the physician assistant who is assigned to take those calls and has the option to see the patient the same day. Patients could continue to call the nurse line or speak with providers at the next appointment at their discretion.
Conclusion
Although Annie has the option of using either text messaging or a mobile application, this project only utilized text messaging. The study by Basch and colleagues was the closest randomized trial we could identify to compare to our quality improvement intervention.5 The 2 main, distinct differences were that Basch and colleagues utilized online monitoring; and nurses were utilized to screen and intervene on responses, as appropriate.
The ability of our program to text patients without the use of an application or tablet, may enable more patients to participate due to ease of use. There would be no increased in expected workload for clinical staff, and may lead to decreased call burden. Since our program is automated, while still providing patients with the option to call and speak with a staff member as needed, this is a cost-effective, first-line option for symptom management for those experiencing cancer-related symptoms. We believe this text messaging tool can have system wide use and benefit throughout the VHA.
Cancer and cancer-related treatment can cause a myriad of adverse effects.1,2 Early identification and management of these symptoms is paramount to the success of cancer treatment completion; however, clinic and telephonic strategies for addressing symptoms often result in delays in care.1 New strategies for patient engagement in the management of cancer and treatment-related symptoms are needed.
The use of online self-management tools can result in improvement in symptoms, reduce cancer symptom distress, improve quality-of-life, and improve medication adherence.3-9 A meta-analysis concluded that online interventions showed promise, but optimizing interventions would require additional research.10 Another meta-analysis found that online self-management was effective in managing several symptoms.11 An e-health method of collecting patient self-reported symptoms has been found to be acceptable to patients and feasible for use.12-14 We postulated that a mobile text messaging strategy may be an effective modality for augmenting symptom management for cancer patients in real time.
In the US Departmant of Veterans Affairs (VA), “Annie,” a self-care tool utilizing a text-messaging system has been implemented. Annie was developed modeling “Flo,” a messaging system in the United Kingdom that has been used for case management of chronic obstructive pulmonary disease, heart failure, stress incontinence, asthma, as a medication reminder tool, and to provide support for weight loss or post-operatively.15-17 Using Annie in the US, veterans have the ability to receive and track health information. Use of the Annie program has demonstrated improved continuous positive airway pressure monitor utilization in veterans with traumatic brain injury.18 Other uses within the Veterans Health Administration (VHA) include assisting patients with anger management, liver disease, anxiety, asthma, diabetes, HIV, hypertension, weight loss, and smoking cessation.
Methods
The Hematology/Oncology division of the Minneapolis VA Healthcare System (MVAHCS) is a tertiary care facility that administers about 260 new chemotherapy regimens annually. The MVAHCS interdisciplinary hematology/oncology group initiated a quality improvement project to determine the feasibility, acceptability, and experience of tailoring the Annie tool for self-management of cancer symptoms. The group consisted of 2 physicians, 3 advanced practice registered nurses, 1 physician assistant, 2 registered nurses, and 2 Annie program team members.
We first created a symptom management pilot protocol as a result of multidisciplinary team discussions. Examples of discussion points for consideration included, but were not limited to, timing of texts, amount of information to ask for and provide, what potential symptoms to consider, and which patient population to pilot first.
The initial protocol was agreed upon and is as follows: Patients were sent text messages twice daily Monday through Friday, and asked to rate 2 symptoms per day, using a severity scale of 0 to 4 (absent, mild, moderate, severe, or disabling): nausea/vomiting, mouth sores, fatigue (Figure 1), trouble breathing, appetite, constipation, diarrhea (Figure 2), numbness/tingling, pain. In addition, patients were asked whether they had had a fever or not. Based on their response to the symptom inquiries, the patient received an automated text response. The text may have provided positive affirmation that they were doing well, given them advice for home management, referred them to an educational hyperlink, asked them to call a direct number to the clinic, or instructed them to report directly to the emergency department (ED). Patients could input a particular symptom on any day, even if they were not specifically asked about that symptom on that day. Patients also were instructed to text, only if it was not an inconvenience to them, as we wanted the intervention to be helpful and not a burden.
Results
Through screening new patient consults or those referred for chemotherapy education, 15 male veterans enrolled in the symptom monitoring program over an 8 month period. There were additional patients who were not offered the program or chose not to participate; often due to not having texting capabilities on their phone or not liking the texting feature. The majority of those who participated in the program (n = 14) were enrolled at the start of Cycle 1; the other patient was enrolled at the start of Cycle 2. Patients were enrolled an average of 89 days (range 8-204). Average response rate was 84.2% (range 30-100%).
Although symptoms were not reviewed in real time, we reviewed responses to determine the utilization of the instructions given for the program. No veteran had 0 symptoms reported. There were numerous occurrences of a score of 1 or 2. Many of these patients had baseline symptoms due to their underlying cancer. A score of 3 or 4 on the system prompted the patient to call the clinic or go to the ED. Seven patients (some with multiple occurrences) were prompted to call; only 4 of these made the follow-up call to the clinic. All were offered a same day visit, but each declined. Only 1 patient reported a symptom on a day not prompted for that symptom. Symptoms that were reported are listed in order of frequency: fatigue, appetite loss, numbness, pain, mouth sore, and breathing difficulty. There were no visits to the ED.
Program Evaluation
An evaluation was conducted 30 to 60 days after program enrollment. We elicited feedback to determine who was reading and responding to the text message: the patient, a family member, or a caregiver; whether they found the prompts helpful and took action; how they felt about the number of texts; if they felt the program was helpful; and any other feedback that would improve the program. In general, the patients (8) answered the texts independently. In 4 cases, the spouse answered the texts, and 3 patients answered the texts together with their spouses. Most patients (11) found the amount of texting to be “just right.” However, 3 found it to be too many texts and 1 didn’t find the amount of texting to be enough.
Three veterans did not have enough symptoms to feel the program was of benefit to them, but they did feel it would have been helpful if they had been more symptomatic. One veteran recalled taking loperamide as needed, as a result of prompting. No veterans felt as though the texting feature was difficult to use; and overall, were very positive about the program. Several appreciated receiving messages that validated when they were doing well, and they felt empowered by self-management. One of the spouses was a registered nurse and found the information too basic to be of use.
Discussion
Initial evaluation of the program via survey found no technology challenges. Patients have been very positive about the program including ease of use, appreciation of messages that validated when they were doing well, empowerment of self-management, and some utilization of the texting advice for symptom management. Educational hyperlinks for constipation, fatigue, diarrhea, and nausea/vomiting were added after this evaluation, and patients felt that these additions provided a higher level of education.
Staff time for this intervention was minimal. A nurse navigator offered the texting program to the patient during chemotherapy education, along with some instructions, which generally took about 5 minutes. One of the Annie program staff enrolled the patient. From that point forward, this was a self-management tool, beyond checking to ensure that the patient was successful in starting the program and evaluating use for the purposes of this quality improvement project. This self-management tool did not replace any other mechanism that a patient would normally have in our department for seeking help for symptoms. The MVAHSC typical process for symptom management is to have patients call a 24/7 nurse line. If the triage nurse feels the symptoms are related to the patient’s cancer or cancer treatment, they are referred to the physician assistant who is assigned to take those calls and has the option to see the patient the same day. Patients could continue to call the nurse line or speak with providers at the next appointment at their discretion.
Conclusion
Although Annie has the option of using either text messaging or a mobile application, this project only utilized text messaging. The study by Basch and colleagues was the closest randomized trial we could identify to compare to our quality improvement intervention.5 The 2 main, distinct differences were that Basch and colleagues utilized online monitoring; and nurses were utilized to screen and intervene on responses, as appropriate.
The ability of our program to text patients without the use of an application or tablet, may enable more patients to participate due to ease of use. There would be no increased in expected workload for clinical staff, and may lead to decreased call burden. Since our program is automated, while still providing patients with the option to call and speak with a staff member as needed, this is a cost-effective, first-line option for symptom management for those experiencing cancer-related symptoms. We believe this text messaging tool can have system wide use and benefit throughout the VHA.
1. Bruera E, Dev R. Overview of managing common non-pain symptoms in palliative care. https://www.uptodate.com/contents/overview-of-managing-common-non-pain-symptoms-in-palliative-care. Updated June 12, 2019. Accessed July 18, 2019.
2. Pirschel C. The crucial role of symptom management in cancer care. https://voice.ons.org/news-and-views/the-crucial-role-of-symptom-management-in-cancer-care. Published December 14, 2017. Accessed July 18, 2019.
3. Adam R, Burton CD, Bond CM, de Bruin M, Murchie P. Can patient-reported measurements of pain be used to improve cancer pain management? A systematic review and meta-analysis. BMJ Support Palliat Care. 2017;7(4):373-382.
4. Basch E, Deal AM, Kris MG, et al. Symptom monitoring with patient-reported outcomes during routine cancer treatment: a randomized controlled trial. J Clin Oncol. 2016;34(6):557-565.
5. Berry DL, Blonquist TM, Patel RA, Halpenny B, McReynolds J. Exposure to a patient-centered, Web-based intervention for managing cancer symptom and quality of life issues: Impact on symptom distress. J Med Internet Res. 2015;3(7):e136.
6. Kolb NA, Smith AG, Singleton JR, et al. Chemotherapy-related neuropathic symptom management: a randomized trial of an automated symptom-monitoring system paired with nurse practitioner follow-up. Support Care Cancer. 2018;26(5):1607-1615
7. Kamdar MM, Centi AJ, Fischer N, Jetwani K. A randomized controlled trial of a novel artificial-intelligence based smartphone application to optimize the management of cancer-related pain. Presented at: 2018 Palliative and Supportive Care in Oncology Symposium; November 16-17, 2018; San Diego, CA.
8. Mooney KH, Beck SL, Wong B, et al. Automated home monitoring and management of patient-reported symptoms during chemotherapy: results of the symptom care at home RCT. Cancer Med. 2017;6(3):537-546.
9. Spoelstra SL, Given CW, Sikorskii A, et al. Proof of concept of a mobile health short message service text message intervention that promotes adherence to oral anticancer agent medications: a randomized controlled trial. Telemed J E Health. 2016;22(6):497-506.
10. Fridriksdottir N, Gunnarsdottir S, Zoëga S, Ingadottir B, Hafsteinsdottir EJG. Effects of web-based interventions on cancer patients’ symptoms: review of randomized trials. Support Care Cancer. 2018;26(2):3370-351.
11. Kim AR, Park HA. Web-based self-management support intervention for cancer survivors: a systematic review and meta-analysis. Stud Health Technol Inform. 2015;216:142-147.
12. Girgis A, Durcinoska I, Levesque JV, et al; PROMPT-Care Program Group. eHealth system for collecting and utilizing patient reported outcome measures for personalized treatment and care (PROMPT-Care) among cancer patients: mixed methods approach to evaluate feasibility and acceptability. J Med Internet Res. 2017;19(10):e330.
13. Moradian S, Krzyzanowska MK, Maguire R, et al. Usability evaluation of a mobile phone-based system for remote monitoring and management of chemotherapy-related side effects in cancer patients: Mixed methods study. JMIR Cancer. 2018;4(2): e10932.
14. Voruganti T, Grunfeld E, Jamieson T, et al. My team of care study: a pilot randomized controlled trial of a web-based communication tool for collaborative care in patients with advanced cancer. J Med Internet Res. 2017;19(7):e219.
15. The Health Foundation. Overview of Florence simple telehealth text messaging system. https://www.health.org.uk/article/overview-of-the-florence-simple-telehealth-text-messaging-system. Accessed July 31, 2019.
16. Bragg DD, Edis H, Clark S, Parsons SL, Perumpalath B…Maxwell-Armstrong CA. Development of a telehealth monitoring service after colorectal surgery: a feasibility study. 2017;9(9):193-199.
17. O’Connell P. Annie-the VA’s self-care game changer. http://www.simple.uk.net/home/blog/blogcontent/annie-thevasself-caregamechanger. Published April 21, 2016. Accessed August 2, 2019.
18. Kataria L, Sundahl, C, Skalina L, et al. Text message reminders and intensive education improves positive airway pressure compliance and cognition in veterans with traumatic brain injury and obstructive sleep apnea: ANNIE pilot study (P1.097). Neurology, 2018; 90(suppl 15):P1.097.
1. Bruera E, Dev R. Overview of managing common non-pain symptoms in palliative care. https://www.uptodate.com/contents/overview-of-managing-common-non-pain-symptoms-in-palliative-care. Updated June 12, 2019. Accessed July 18, 2019.
2. Pirschel C. The crucial role of symptom management in cancer care. https://voice.ons.org/news-and-views/the-crucial-role-of-symptom-management-in-cancer-care. Published December 14, 2017. Accessed July 18, 2019.
3. Adam R, Burton CD, Bond CM, de Bruin M, Murchie P. Can patient-reported measurements of pain be used to improve cancer pain management? A systematic review and meta-analysis. BMJ Support Palliat Care. 2017;7(4):373-382.
4. Basch E, Deal AM, Kris MG, et al. Symptom monitoring with patient-reported outcomes during routine cancer treatment: a randomized controlled trial. J Clin Oncol. 2016;34(6):557-565.
5. Berry DL, Blonquist TM, Patel RA, Halpenny B, McReynolds J. Exposure to a patient-centered, Web-based intervention for managing cancer symptom and quality of life issues: Impact on symptom distress. J Med Internet Res. 2015;3(7):e136.
6. Kolb NA, Smith AG, Singleton JR, et al. Chemotherapy-related neuropathic symptom management: a randomized trial of an automated symptom-monitoring system paired with nurse practitioner follow-up. Support Care Cancer. 2018;26(5):1607-1615
7. Kamdar MM, Centi AJ, Fischer N, Jetwani K. A randomized controlled trial of a novel artificial-intelligence based smartphone application to optimize the management of cancer-related pain. Presented at: 2018 Palliative and Supportive Care in Oncology Symposium; November 16-17, 2018; San Diego, CA.
8. Mooney KH, Beck SL, Wong B, et al. Automated home monitoring and management of patient-reported symptoms during chemotherapy: results of the symptom care at home RCT. Cancer Med. 2017;6(3):537-546.
9. Spoelstra SL, Given CW, Sikorskii A, et al. Proof of concept of a mobile health short message service text message intervention that promotes adherence to oral anticancer agent medications: a randomized controlled trial. Telemed J E Health. 2016;22(6):497-506.
10. Fridriksdottir N, Gunnarsdottir S, Zoëga S, Ingadottir B, Hafsteinsdottir EJG. Effects of web-based interventions on cancer patients’ symptoms: review of randomized trials. Support Care Cancer. 2018;26(2):3370-351.
11. Kim AR, Park HA. Web-based self-management support intervention for cancer survivors: a systematic review and meta-analysis. Stud Health Technol Inform. 2015;216:142-147.
12. Girgis A, Durcinoska I, Levesque JV, et al; PROMPT-Care Program Group. eHealth system for collecting and utilizing patient reported outcome measures for personalized treatment and care (PROMPT-Care) among cancer patients: mixed methods approach to evaluate feasibility and acceptability. J Med Internet Res. 2017;19(10):e330.
13. Moradian S, Krzyzanowska MK, Maguire R, et al. Usability evaluation of a mobile phone-based system for remote monitoring and management of chemotherapy-related side effects in cancer patients: Mixed methods study. JMIR Cancer. 2018;4(2): e10932.
14. Voruganti T, Grunfeld E, Jamieson T, et al. My team of care study: a pilot randomized controlled trial of a web-based communication tool for collaborative care in patients with advanced cancer. J Med Internet Res. 2017;19(7):e219.
15. The Health Foundation. Overview of Florence simple telehealth text messaging system. https://www.health.org.uk/article/overview-of-the-florence-simple-telehealth-text-messaging-system. Accessed July 31, 2019.
16. Bragg DD, Edis H, Clark S, Parsons SL, Perumpalath B…Maxwell-Armstrong CA. Development of a telehealth monitoring service after colorectal surgery: a feasibility study. 2017;9(9):193-199.
17. O’Connell P. Annie-the VA’s self-care game changer. http://www.simple.uk.net/home/blog/blogcontent/annie-thevasself-caregamechanger. Published April 21, 2016. Accessed August 2, 2019.
18. Kataria L, Sundahl, C, Skalina L, et al. Text message reminders and intensive education improves positive airway pressure compliance and cognition in veterans with traumatic brain injury and obstructive sleep apnea: ANNIE pilot study (P1.097). Neurology, 2018; 90(suppl 15):P1.097.
A Novel Pharmaceutical Care Model for High-Risk Patients
Nonadherence is a significant problem that has a negative impact on both patients and public health. Patients with multiple diseases often have complicated medication regimens, which can be difficult for them to manage. Unfortunately, nonadherence in these high-risk patients can have drastic consequences, including disease progression, hospitalization, and death, resulting in billions of dollars in unnecessary costs nationwide.1,2 The Wheel Model of Pharmaceutical Care (Figure) is a novel care model developed at the Gallup Indian Medical Center (GIMC) in New Mexico to address these problems by positioning pharmacy as a proactive service. The Wheel Model of Pharmaceutical Care was designed to improve adherence and patient outcomes and to encourage communication among the patient, pharmacists, prescribers, and other health care team members.
Pharmacists are central to managing patients’ medication therapies and coordinating communication among the health care providers (HCPs).1,3 Medication therapy management (MTM), a required component of Medicare Part D plans, helps ensure appropriate drug use and reduce the risk of adverse events.3 Since pharmacists receive prescriptions from all of the patient’s HCPs, patients may see pharmacists more often than they see any other HCP. GIMC is currently piloting a new clinic, the Medication Optimization, Synchronization, and Adherence Improvement Clinic (MOSAIC), that was created to implement the Wheel Model of Pharmaceutical Care. MOSAIC aims to provide proactive pharmacy services and continuous MTM to high-risk patients and will enable the effectiveness of this new pharmaceutical care model to be assessed.
Methods
Studies have identified certain populations who are at an increased risk for nonadherence: the elderly, patients with complex or extensive medication regimens, patients with multiple chronic medical conditions, substance misusers, certain ethnicities, patients of lower socioeconomic status, patients with limited literacy, and the homeless.2,4 Federal regulations require that Medicare Part D plans target beneficiaries who meet specific criteria for MTM programs. Under these rules, plans must target beneficiaries with ≥ 3 chronic diseases and ≥ 8 chronic medications, although plans also may include patients with fewer medications and diseases.3 Although the Wheel Model of Pharmaceutical Care is postulated to be an accurate model for the ideal care of all patients, initial implementation should be targeted toward populations who are likely to benefit the most from intervention. For these reasons, elderly Native American patients who have ≥ 2 chronic diseases and who take ≥ 5 chronic medications were targeted for initial enrollment in MOSAIC at GIMC.
Overview
In MOSAIC, pharmacists act as the hub of the pharmaceutical care wheel. Pharmacists work to ensure optimization of the patient’s comprehensive, integrated care plan—the rim of the wheel. As a part of this optimization process, MOSAIC pharmacists facilitate synchronization of the patient’s prescriptions to a monthly or quarterly target fill date. The patient’s current medication therapy is organized, and pharmacists track which medications are due to be filled instead of depending on the patient to request each prescription refill. This process effectively changes pharmacy from a requested service to a provided service.
Pharmacists also monitor the air in the tire to promote adherence. This is accomplished by providing efficient monthly or quarterly telephone or in-person consultations, which helps the patient better understand his or her comprehensive, integrated care plan. MOSAIC eliminates the possibility of nonadherence due to running out of refills. Specialized packaging, such as pill boxes or blister packs, can also improve adherence for certain patients.
MOSAIC ensures that pharmacists stay connected with the spokes, which represent a patient’s numerous prescribers, and close communication loops. Pharmacists can make prescribers aware of potential gaps or overlaps in treatment and assist them in the optimization and development of the patient’s comprehensive, integrated care plan. Pharmacists also make sure that the patient’s medication profile is current and accurate in the electronic health record (EHR). Any pertinent information discovered during MOSAIC encounters, such as abnormal laboratory results or changes in medications or disease, is documented in an EHR note. The patient’s prescribers are made aware of this information by tagging them as additional signers to the note in the EHR.
Keeping patients—the tires—healthy will ensure smooth operation of the vehicle and have a positive impact on public health. MOSAIC is expected to not only improve individual patient outcomes, but also decrease health care costs for patients and society due to nonadherence, suboptimal regimens, stockpiled home medications, and preventable hospital admissions.
Traditionally, pharmacy has been a requested service: A patient requests each of their prescriptions to be refilled, and the pharmacy fills the prescription. Ideally, pharmacy must become a provided service, with pharmacists keeping track of when a patient’s medications are due to be filled and actively looking for medication therapy optimization opportunities. This is accomplished by synchronizing the patient’s medications to the same monthly or quarterly fill date; screening for any potentially inappropriate medications, including high-risk medications in elderly patients, duplications, and omissions; verifying any medication changes with the patient each fill; and then providing all needed medications to the patient at a scheduled time.
To facilitate this process, custom software was developed for MOSAIC. In addition, a collaborative practice agreement (CPA) was drafted that allowed MOSAIC pharmacists to make certain medication therapy optimizations on behalf of the patient’s primary care provider. As part of this CPA, pharmacists also may order and act on certain laboratory tests, which helps to monitor disease progression, ensure safe medication use, and meet Government Performance and Results Act (GPRA) measures. As a novel model of pharmaceutical care, the effects of this approach are not yet known; however, research suggests that increased communication among HCPs and patient-centered approaches to care are beneficial to patient outcomes, adherence, and public health.1,5
Investigated Outcomes
As patients continue to enroll in MOSAIC, the effectiveness of the clinic will be evaluated. Specifically, quality of life, patient and HCP satisfaction with the program, adherence metrics, hospitalization rates, and all-cause mortality will be assessed for patients enrolled in MOSAIC as well as similar patients who are not enrolled in MOSAIC. Also, pharmacists will log all recommended medication therapy interventions so that the optimization component of MOSAIC may be quantified. GPRA measures and the financial implications of the interventions made by MOSAIC will also be evaluated.
Discussion
There are a number of factors, such as MTM services and interprofessional care teams, that research has shown to independently improve patient outcomes, adherence, or public health. By synthesizing these factors, a completely new approach—the Wheel Model of Pharmaceutical Care—was developed. This model presents a radical departure from traditional, requested-service practices and posits pharmacy as a provided service instead. Although the ideas of MTM and interprofessional care teams are not new, there has never been a practical way to truly integrate community pharmacists into the patient care team or to ensure adequate communication among all of the patient’s HCPs. The Wheel Model of Pharmaceutical Care includes public health as one of its core components and provides a framework for pharmacies to meaningfully impact health outcomes for patients.
The Wheel Model of Pharmaceutical Care was designed to minimize the likelihood of nonadherence. Despite this, patients might willfully choose to be nonadherent, forget to take their medications, or neglect to pick up their medications. Additionally, in health care systems where patients must pay for their medications, prescription drug costs might be a barrier to adherence.
When nonadherence is suspected, the Wheel Model of Pharmaceutical Care directs pharmacists in MOSAIC to take action. First, the underlying cause of the nonadherence must be determined. For example, if a patient is nonadherent because of an adverse drug reaction, a therapy change may be indicated. If a patient is nonadherent due to apathy toward their health or therapy, the patient may benefit from education about their condition and treatment options; thus, the patient can make shared, informed decisions and feel more actively involved with his or her health. If a patients is nonadherent due to forgetfulness, adherence packaging dispense methods should be considered as an alternative to traditional vials. Depending on the services offered by a given pharmacy, adherence packaging options may include blister packs, pill boxes, or strips prepared by robotic dispensing systems. The use of medication reminders, whether in the form of a smartphone application or a simple alarm clock, should be discussed with the patient. If the patient does not pick up their medications on time, a pharmacist can contact the patient to determine why the medications were not picked up and to assess any nonadherence. In this case, mail order pharmacy services, if available, should be offered to patients as a more convenient option.
The medication regimen optimization component of MOSAIC helps reduce the workload of primary care providers and allows pharmacists to act autonomously based on clinical judgment, within the scope of the CPA. This can prevent delays in care caused by no refills remaining on a prescription. The laboratory monitoring component allows pharmacists to track diseases and take action if necessary, which should have a favorable impact on GPRA measures. Medication optimizations can reduce wasted resources by identifying cost-saving formulary alternatives, potentially inappropriate medications, and suboptimal doses.
Since many Indian Health Service beneficiaries do not have private insurance and therefore do not generate third-party reimbursements for services and care provided by GIMC, keeping patients healthy and out of the hospital is a top priority. As more patients are enrolled in MOSAIC, the program is expected to have a favorable impact on pharmacy workload and workflow as well. Prescriptions are anticipated and filled in advance, which decreases the amount of patients calling and presenting to the pharmacy for same-day refill requests. Scheduling when MOSAIC patients’ medications are to be filled and dispensed creates a predictable workload that allows the pharmacy staff to be managed more efficiently.
Conclusion
Adherence is the responsibility of the patient, but the Wheel Model of Pharmaceutical Care aims to provide pharmacists with a framework to monitor and encourage adherence in their patients. By taking this patient-centered approach, MOSAIC is expected to improve outcomes and decrease hospitalizations for high-risk patients who simply need a little extra help with their medications.
1. Bosworth HB, Granger BB, Mendys P, et al. Medication adherence: a call for action. Am Heart J. 2011;162(3):412-424.
2. Vlasnik JJ, Aliotta SL, DeLor B. Medication adherence: factors influencing compliance with prescribed medication plans. Case Manager. 2005;16(2):47-51.
3. Drug utilization management, quality assurance, and medication therapy management programs (MTMPs). Fed Regist. 2012;77(71):2207-22175. To be codified at 42 CFR § 423.153.
4. Thiruchselvam T, Naglie G, Moineddin R, et al. Risk factors for medication nonadherence in older adults with cognitive impairment who live alone. Int J Geriatr Psychiatry. 2012;27(12):1275-1282.
5. Liddy C, Blazkho V, Mill K. Challenges of self-management when living with multiple chronic conditions: systematic review of the qualitative literature. Can Fam Physician. 2014;60(12):1123-1133.
Nonadherence is a significant problem that has a negative impact on both patients and public health. Patients with multiple diseases often have complicated medication regimens, which can be difficult for them to manage. Unfortunately, nonadherence in these high-risk patients can have drastic consequences, including disease progression, hospitalization, and death, resulting in billions of dollars in unnecessary costs nationwide.1,2 The Wheel Model of Pharmaceutical Care (Figure) is a novel care model developed at the Gallup Indian Medical Center (GIMC) in New Mexico to address these problems by positioning pharmacy as a proactive service. The Wheel Model of Pharmaceutical Care was designed to improve adherence and patient outcomes and to encourage communication among the patient, pharmacists, prescribers, and other health care team members.
Pharmacists are central to managing patients’ medication therapies and coordinating communication among the health care providers (HCPs).1,3 Medication therapy management (MTM), a required component of Medicare Part D plans, helps ensure appropriate drug use and reduce the risk of adverse events.3 Since pharmacists receive prescriptions from all of the patient’s HCPs, patients may see pharmacists more often than they see any other HCP. GIMC is currently piloting a new clinic, the Medication Optimization, Synchronization, and Adherence Improvement Clinic (MOSAIC), that was created to implement the Wheel Model of Pharmaceutical Care. MOSAIC aims to provide proactive pharmacy services and continuous MTM to high-risk patients and will enable the effectiveness of this new pharmaceutical care model to be assessed.
Methods
Studies have identified certain populations who are at an increased risk for nonadherence: the elderly, patients with complex or extensive medication regimens, patients with multiple chronic medical conditions, substance misusers, certain ethnicities, patients of lower socioeconomic status, patients with limited literacy, and the homeless.2,4 Federal regulations require that Medicare Part D plans target beneficiaries who meet specific criteria for MTM programs. Under these rules, plans must target beneficiaries with ≥ 3 chronic diseases and ≥ 8 chronic medications, although plans also may include patients with fewer medications and diseases.3 Although the Wheel Model of Pharmaceutical Care is postulated to be an accurate model for the ideal care of all patients, initial implementation should be targeted toward populations who are likely to benefit the most from intervention. For these reasons, elderly Native American patients who have ≥ 2 chronic diseases and who take ≥ 5 chronic medications were targeted for initial enrollment in MOSAIC at GIMC.
Overview
In MOSAIC, pharmacists act as the hub of the pharmaceutical care wheel. Pharmacists work to ensure optimization of the patient’s comprehensive, integrated care plan—the rim of the wheel. As a part of this optimization process, MOSAIC pharmacists facilitate synchronization of the patient’s prescriptions to a monthly or quarterly target fill date. The patient’s current medication therapy is organized, and pharmacists track which medications are due to be filled instead of depending on the patient to request each prescription refill. This process effectively changes pharmacy from a requested service to a provided service.
Pharmacists also monitor the air in the tire to promote adherence. This is accomplished by providing efficient monthly or quarterly telephone or in-person consultations, which helps the patient better understand his or her comprehensive, integrated care plan. MOSAIC eliminates the possibility of nonadherence due to running out of refills. Specialized packaging, such as pill boxes or blister packs, can also improve adherence for certain patients.
MOSAIC ensures that pharmacists stay connected with the spokes, which represent a patient’s numerous prescribers, and close communication loops. Pharmacists can make prescribers aware of potential gaps or overlaps in treatment and assist them in the optimization and development of the patient’s comprehensive, integrated care plan. Pharmacists also make sure that the patient’s medication profile is current and accurate in the electronic health record (EHR). Any pertinent information discovered during MOSAIC encounters, such as abnormal laboratory results or changes in medications or disease, is documented in an EHR note. The patient’s prescribers are made aware of this information by tagging them as additional signers to the note in the EHR.
Keeping patients—the tires—healthy will ensure smooth operation of the vehicle and have a positive impact on public health. MOSAIC is expected to not only improve individual patient outcomes, but also decrease health care costs for patients and society due to nonadherence, suboptimal regimens, stockpiled home medications, and preventable hospital admissions.
Traditionally, pharmacy has been a requested service: A patient requests each of their prescriptions to be refilled, and the pharmacy fills the prescription. Ideally, pharmacy must become a provided service, with pharmacists keeping track of when a patient’s medications are due to be filled and actively looking for medication therapy optimization opportunities. This is accomplished by synchronizing the patient’s medications to the same monthly or quarterly fill date; screening for any potentially inappropriate medications, including high-risk medications in elderly patients, duplications, and omissions; verifying any medication changes with the patient each fill; and then providing all needed medications to the patient at a scheduled time.
To facilitate this process, custom software was developed for MOSAIC. In addition, a collaborative practice agreement (CPA) was drafted that allowed MOSAIC pharmacists to make certain medication therapy optimizations on behalf of the patient’s primary care provider. As part of this CPA, pharmacists also may order and act on certain laboratory tests, which helps to monitor disease progression, ensure safe medication use, and meet Government Performance and Results Act (GPRA) measures. As a novel model of pharmaceutical care, the effects of this approach are not yet known; however, research suggests that increased communication among HCPs and patient-centered approaches to care are beneficial to patient outcomes, adherence, and public health.1,5
Investigated Outcomes
As patients continue to enroll in MOSAIC, the effectiveness of the clinic will be evaluated. Specifically, quality of life, patient and HCP satisfaction with the program, adherence metrics, hospitalization rates, and all-cause mortality will be assessed for patients enrolled in MOSAIC as well as similar patients who are not enrolled in MOSAIC. Also, pharmacists will log all recommended medication therapy interventions so that the optimization component of MOSAIC may be quantified. GPRA measures and the financial implications of the interventions made by MOSAIC will also be evaluated.
Discussion
There are a number of factors, such as MTM services and interprofessional care teams, that research has shown to independently improve patient outcomes, adherence, or public health. By synthesizing these factors, a completely new approach—the Wheel Model of Pharmaceutical Care—was developed. This model presents a radical departure from traditional, requested-service practices and posits pharmacy as a provided service instead. Although the ideas of MTM and interprofessional care teams are not new, there has never been a practical way to truly integrate community pharmacists into the patient care team or to ensure adequate communication among all of the patient’s HCPs. The Wheel Model of Pharmaceutical Care includes public health as one of its core components and provides a framework for pharmacies to meaningfully impact health outcomes for patients.
The Wheel Model of Pharmaceutical Care was designed to minimize the likelihood of nonadherence. Despite this, patients might willfully choose to be nonadherent, forget to take their medications, or neglect to pick up their medications. Additionally, in health care systems where patients must pay for their medications, prescription drug costs might be a barrier to adherence.
When nonadherence is suspected, the Wheel Model of Pharmaceutical Care directs pharmacists in MOSAIC to take action. First, the underlying cause of the nonadherence must be determined. For example, if a patient is nonadherent because of an adverse drug reaction, a therapy change may be indicated. If a patient is nonadherent due to apathy toward their health or therapy, the patient may benefit from education about their condition and treatment options; thus, the patient can make shared, informed decisions and feel more actively involved with his or her health. If a patients is nonadherent due to forgetfulness, adherence packaging dispense methods should be considered as an alternative to traditional vials. Depending on the services offered by a given pharmacy, adherence packaging options may include blister packs, pill boxes, or strips prepared by robotic dispensing systems. The use of medication reminders, whether in the form of a smartphone application or a simple alarm clock, should be discussed with the patient. If the patient does not pick up their medications on time, a pharmacist can contact the patient to determine why the medications were not picked up and to assess any nonadherence. In this case, mail order pharmacy services, if available, should be offered to patients as a more convenient option.
The medication regimen optimization component of MOSAIC helps reduce the workload of primary care providers and allows pharmacists to act autonomously based on clinical judgment, within the scope of the CPA. This can prevent delays in care caused by no refills remaining on a prescription. The laboratory monitoring component allows pharmacists to track diseases and take action if necessary, which should have a favorable impact on GPRA measures. Medication optimizations can reduce wasted resources by identifying cost-saving formulary alternatives, potentially inappropriate medications, and suboptimal doses.
Since many Indian Health Service beneficiaries do not have private insurance and therefore do not generate third-party reimbursements for services and care provided by GIMC, keeping patients healthy and out of the hospital is a top priority. As more patients are enrolled in MOSAIC, the program is expected to have a favorable impact on pharmacy workload and workflow as well. Prescriptions are anticipated and filled in advance, which decreases the amount of patients calling and presenting to the pharmacy for same-day refill requests. Scheduling when MOSAIC patients’ medications are to be filled and dispensed creates a predictable workload that allows the pharmacy staff to be managed more efficiently.
Conclusion
Adherence is the responsibility of the patient, but the Wheel Model of Pharmaceutical Care aims to provide pharmacists with a framework to monitor and encourage adherence in their patients. By taking this patient-centered approach, MOSAIC is expected to improve outcomes and decrease hospitalizations for high-risk patients who simply need a little extra help with their medications.
Nonadherence is a significant problem that has a negative impact on both patients and public health. Patients with multiple diseases often have complicated medication regimens, which can be difficult for them to manage. Unfortunately, nonadherence in these high-risk patients can have drastic consequences, including disease progression, hospitalization, and death, resulting in billions of dollars in unnecessary costs nationwide.1,2 The Wheel Model of Pharmaceutical Care (Figure) is a novel care model developed at the Gallup Indian Medical Center (GIMC) in New Mexico to address these problems by positioning pharmacy as a proactive service. The Wheel Model of Pharmaceutical Care was designed to improve adherence and patient outcomes and to encourage communication among the patient, pharmacists, prescribers, and other health care team members.
Pharmacists are central to managing patients’ medication therapies and coordinating communication among the health care providers (HCPs).1,3 Medication therapy management (MTM), a required component of Medicare Part D plans, helps ensure appropriate drug use and reduce the risk of adverse events.3 Since pharmacists receive prescriptions from all of the patient’s HCPs, patients may see pharmacists more often than they see any other HCP. GIMC is currently piloting a new clinic, the Medication Optimization, Synchronization, and Adherence Improvement Clinic (MOSAIC), that was created to implement the Wheel Model of Pharmaceutical Care. MOSAIC aims to provide proactive pharmacy services and continuous MTM to high-risk patients and will enable the effectiveness of this new pharmaceutical care model to be assessed.
Methods
Studies have identified certain populations who are at an increased risk for nonadherence: the elderly, patients with complex or extensive medication regimens, patients with multiple chronic medical conditions, substance misusers, certain ethnicities, patients of lower socioeconomic status, patients with limited literacy, and the homeless.2,4 Federal regulations require that Medicare Part D plans target beneficiaries who meet specific criteria for MTM programs. Under these rules, plans must target beneficiaries with ≥ 3 chronic diseases and ≥ 8 chronic medications, although plans also may include patients with fewer medications and diseases.3 Although the Wheel Model of Pharmaceutical Care is postulated to be an accurate model for the ideal care of all patients, initial implementation should be targeted toward populations who are likely to benefit the most from intervention. For these reasons, elderly Native American patients who have ≥ 2 chronic diseases and who take ≥ 5 chronic medications were targeted for initial enrollment in MOSAIC at GIMC.
Overview
In MOSAIC, pharmacists act as the hub of the pharmaceutical care wheel. Pharmacists work to ensure optimization of the patient’s comprehensive, integrated care plan—the rim of the wheel. As a part of this optimization process, MOSAIC pharmacists facilitate synchronization of the patient’s prescriptions to a monthly or quarterly target fill date. The patient’s current medication therapy is organized, and pharmacists track which medications are due to be filled instead of depending on the patient to request each prescription refill. This process effectively changes pharmacy from a requested service to a provided service.
Pharmacists also monitor the air in the tire to promote adherence. This is accomplished by providing efficient monthly or quarterly telephone or in-person consultations, which helps the patient better understand his or her comprehensive, integrated care plan. MOSAIC eliminates the possibility of nonadherence due to running out of refills. Specialized packaging, such as pill boxes or blister packs, can also improve adherence for certain patients.
MOSAIC ensures that pharmacists stay connected with the spokes, which represent a patient’s numerous prescribers, and close communication loops. Pharmacists can make prescribers aware of potential gaps or overlaps in treatment and assist them in the optimization and development of the patient’s comprehensive, integrated care plan. Pharmacists also make sure that the patient’s medication profile is current and accurate in the electronic health record (EHR). Any pertinent information discovered during MOSAIC encounters, such as abnormal laboratory results or changes in medications or disease, is documented in an EHR note. The patient’s prescribers are made aware of this information by tagging them as additional signers to the note in the EHR.
Keeping patients—the tires—healthy will ensure smooth operation of the vehicle and have a positive impact on public health. MOSAIC is expected to not only improve individual patient outcomes, but also decrease health care costs for patients and society due to nonadherence, suboptimal regimens, stockpiled home medications, and preventable hospital admissions.
Traditionally, pharmacy has been a requested service: A patient requests each of their prescriptions to be refilled, and the pharmacy fills the prescription. Ideally, pharmacy must become a provided service, with pharmacists keeping track of when a patient’s medications are due to be filled and actively looking for medication therapy optimization opportunities. This is accomplished by synchronizing the patient’s medications to the same monthly or quarterly fill date; screening for any potentially inappropriate medications, including high-risk medications in elderly patients, duplications, and omissions; verifying any medication changes with the patient each fill; and then providing all needed medications to the patient at a scheduled time.
To facilitate this process, custom software was developed for MOSAIC. In addition, a collaborative practice agreement (CPA) was drafted that allowed MOSAIC pharmacists to make certain medication therapy optimizations on behalf of the patient’s primary care provider. As part of this CPA, pharmacists also may order and act on certain laboratory tests, which helps to monitor disease progression, ensure safe medication use, and meet Government Performance and Results Act (GPRA) measures. As a novel model of pharmaceutical care, the effects of this approach are not yet known; however, research suggests that increased communication among HCPs and patient-centered approaches to care are beneficial to patient outcomes, adherence, and public health.1,5
Investigated Outcomes
As patients continue to enroll in MOSAIC, the effectiveness of the clinic will be evaluated. Specifically, quality of life, patient and HCP satisfaction with the program, adherence metrics, hospitalization rates, and all-cause mortality will be assessed for patients enrolled in MOSAIC as well as similar patients who are not enrolled in MOSAIC. Also, pharmacists will log all recommended medication therapy interventions so that the optimization component of MOSAIC may be quantified. GPRA measures and the financial implications of the interventions made by MOSAIC will also be evaluated.
Discussion
There are a number of factors, such as MTM services and interprofessional care teams, that research has shown to independently improve patient outcomes, adherence, or public health. By synthesizing these factors, a completely new approach—the Wheel Model of Pharmaceutical Care—was developed. This model presents a radical departure from traditional, requested-service practices and posits pharmacy as a provided service instead. Although the ideas of MTM and interprofessional care teams are not new, there has never been a practical way to truly integrate community pharmacists into the patient care team or to ensure adequate communication among all of the patient’s HCPs. The Wheel Model of Pharmaceutical Care includes public health as one of its core components and provides a framework for pharmacies to meaningfully impact health outcomes for patients.
The Wheel Model of Pharmaceutical Care was designed to minimize the likelihood of nonadherence. Despite this, patients might willfully choose to be nonadherent, forget to take their medications, or neglect to pick up their medications. Additionally, in health care systems where patients must pay for their medications, prescription drug costs might be a barrier to adherence.
When nonadherence is suspected, the Wheel Model of Pharmaceutical Care directs pharmacists in MOSAIC to take action. First, the underlying cause of the nonadherence must be determined. For example, if a patient is nonadherent because of an adverse drug reaction, a therapy change may be indicated. If a patient is nonadherent due to apathy toward their health or therapy, the patient may benefit from education about their condition and treatment options; thus, the patient can make shared, informed decisions and feel more actively involved with his or her health. If a patients is nonadherent due to forgetfulness, adherence packaging dispense methods should be considered as an alternative to traditional vials. Depending on the services offered by a given pharmacy, adherence packaging options may include blister packs, pill boxes, or strips prepared by robotic dispensing systems. The use of medication reminders, whether in the form of a smartphone application or a simple alarm clock, should be discussed with the patient. If the patient does not pick up their medications on time, a pharmacist can contact the patient to determine why the medications were not picked up and to assess any nonadherence. In this case, mail order pharmacy services, if available, should be offered to patients as a more convenient option.
The medication regimen optimization component of MOSAIC helps reduce the workload of primary care providers and allows pharmacists to act autonomously based on clinical judgment, within the scope of the CPA. This can prevent delays in care caused by no refills remaining on a prescription. The laboratory monitoring component allows pharmacists to track diseases and take action if necessary, which should have a favorable impact on GPRA measures. Medication optimizations can reduce wasted resources by identifying cost-saving formulary alternatives, potentially inappropriate medications, and suboptimal doses.
Since many Indian Health Service beneficiaries do not have private insurance and therefore do not generate third-party reimbursements for services and care provided by GIMC, keeping patients healthy and out of the hospital is a top priority. As more patients are enrolled in MOSAIC, the program is expected to have a favorable impact on pharmacy workload and workflow as well. Prescriptions are anticipated and filled in advance, which decreases the amount of patients calling and presenting to the pharmacy for same-day refill requests. Scheduling when MOSAIC patients’ medications are to be filled and dispensed creates a predictable workload that allows the pharmacy staff to be managed more efficiently.
Conclusion
Adherence is the responsibility of the patient, but the Wheel Model of Pharmaceutical Care aims to provide pharmacists with a framework to monitor and encourage adherence in their patients. By taking this patient-centered approach, MOSAIC is expected to improve outcomes and decrease hospitalizations for high-risk patients who simply need a little extra help with their medications.
1. Bosworth HB, Granger BB, Mendys P, et al. Medication adherence: a call for action. Am Heart J. 2011;162(3):412-424.
2. Vlasnik JJ, Aliotta SL, DeLor B. Medication adherence: factors influencing compliance with prescribed medication plans. Case Manager. 2005;16(2):47-51.
3. Drug utilization management, quality assurance, and medication therapy management programs (MTMPs). Fed Regist. 2012;77(71):2207-22175. To be codified at 42 CFR § 423.153.
4. Thiruchselvam T, Naglie G, Moineddin R, et al. Risk factors for medication nonadherence in older adults with cognitive impairment who live alone. Int J Geriatr Psychiatry. 2012;27(12):1275-1282.
5. Liddy C, Blazkho V, Mill K. Challenges of self-management when living with multiple chronic conditions: systematic review of the qualitative literature. Can Fam Physician. 2014;60(12):1123-1133.
1. Bosworth HB, Granger BB, Mendys P, et al. Medication adherence: a call for action. Am Heart J. 2011;162(3):412-424.
2. Vlasnik JJ, Aliotta SL, DeLor B. Medication adherence: factors influencing compliance with prescribed medication plans. Case Manager. 2005;16(2):47-51.
3. Drug utilization management, quality assurance, and medication therapy management programs (MTMPs). Fed Regist. 2012;77(71):2207-22175. To be codified at 42 CFR § 423.153.
4. Thiruchselvam T, Naglie G, Moineddin R, et al. Risk factors for medication nonadherence in older adults with cognitive impairment who live alone. Int J Geriatr Psychiatry. 2012;27(12):1275-1282.
5. Liddy C, Blazkho V, Mill K. Challenges of self-management when living with multiple chronic conditions: systematic review of the qualitative literature. Can Fam Physician. 2014;60(12):1123-1133.
Improved Patient Outcomes and Reduced Wait Times: Transforming a VA Outpatient Substance Use Disorder Program
Substance use disorders (SUDs) are an increasing public health concern in the US. The 2015 National Survey on Drug Use and Health indicated that 27 million people (8% of the US population) reported current use of recreational drugs or misuse of alcohol or prescription medications.1 The 2013 National Survey on Drug Use and Health indicated that 1.5 million veterans (roughly 6.6%) met the criteria for a SUD.2 More than 50% of patients awaiting entry into a SUD treatment program will never achieve admission due, in part, to long wait times.3-5
National attention has been focused on increasing veteran access to quality treatment based on evidence-based practices (EBPs). Several national legislative measures and treatment protocols have been implemented: the Uniform Mental Health Services in US Department of Veterans Affairs (VA) medical centers and clinics; Veterans Access, Choice, and Accountability Act (2014); Cognitive Behavioral Therapy for Substance Use Disorders (CBT-SUD) Training Program; and the Psychotropic Drug Safety Initiative (PDSI).6-8 Consistent with these directives and in line with American Society of Addiction Medicine (ASAM) and Substance Abuse and Mental Health Services Administration (SAMHSA) guidelines for medication-assisted therapies (MAT),the James A. Haley Veterans’ Hospital (JAHVH) Mental Health and Behavioral Sciences Service (MH&BSS) Substance Use Disorders Service (SUDS) in Tampa, Florida, implemented an evidence-based, treatment-on-demand model of care.9-11
Meeting SUD Treatment Needs
What does the new supervisor of a clinical program do when a 24-employee outpatient VA Alcohol and Drug Addiction Treatment Program (ADATP) has an average 33-day wait time for treatment with 54% of patients lost to care between initial evaluation and admission?12 Patients lacked consistent access to SUD pharmacotherapy. The national VA clinical performance indicators were substandard and there are no additional resources available to apply to the program.
At JAHVH the program supervisor enlisted hospital leadership to support program redesign. The redesign sought to improve operational efficiency and eliminate patient wait time; adopt national standards for assessment and treatment developed by ASAM; implement strictly evidence-based psychotherapeutic treatments; educate program psychiatrists about evidence-based psychopharmacologic treatments and hold them accountable for patient adherence; streamline documentation templates; free clinical providers from nonclinical tasks; create an inpatient addiction consult team to diagnose and treat chronic hospitalized patients with SUDs; ensure continuity of care; and, standardize consistent, objective measures of patient response to treatment to track the program’s effectiveness.
In this article, the authors provide an explanation of the clinical, theoretical foundation and the practical steps taken to design and implement this transformation. They then describe the lessons learned, hoping that their process will serve as a model for those in similar situations.
Program Redesign
July 1, 2015, a new program supervisor was hired and began a 2-month evaluation and analysis of the program with input from leadership, staff, and hospital/community stakeholders. September 1, the monthlong process of developing the redesign began. On September 30 the plan was presented to, and approved by, MH&BSS leadership. October was spent preparing for change with an implementation date of November 2 selected. On November 2, 2015, the complete redesign was implemented.
Needs Assessment
A needs assessment yielded improvement opportunities in program structure (levels of care); clinical content; staff and resource allocation, including clinical workflow and management systems. Staff identified philosophical and practical variance in the program, often leading to confusion for patients and clinicians and potentially resulting in disparate quality care and patient outcomes. Recommendations for addressing these needs included incorporating ASAM guidelines for assignment to clinically appropriate levels of care, implementation of consistent EBPs for SUD and comorbid conditions,9 and emphasis on staff training and development to champion evidence-based program philosophy and service delivery.
The assessment determined that the average waitlist time was 33 days, and patients were required to abstain from substance or alcohol use prior to admission to the Intensive Outpatient Program. If a waitlisted patient relapsed, she or he was removed from the waitlist and denied admission. A study conducted at JAHVH reported that 54% of waitlisted patients in this clinic (prior to November 2, 2015) never were admitted to the program.12 Access to care was considered a significant issue.
Program Implementation
September was spent developing a comprehensive redesign of the SUD clinic. The vision included incorporating all ASAM levels of care; creating an evidence-based, treatment-on-demand model of care; and, securing the support of MH&BSS leadership team, staff, and patients for the redesign. The supervisory clinician interviewed staff both individually and as a group. Clinicians were provided extensive training on EBP for SUDs, including psychotherapies, psychosocial treatments, and psychopharmacologic interventions. A journal club was started with staff-generated topics that offered articles sharing current research, EBPs, and psychotherapeutic techniques, continuing education on substances, and management of coexisting diagnoses. Clinicians increased the frequency of SUD in-service trainings. Psychiatrists provided several Grand Rounds to the MH&BSS service. All counselors were assigned to 1 of the program’s 3 clinical psychologists for individual weekly clinical supervision.
By providing all staff with current, evidence-based, clinically relevant treatment information and emphasizing its relationship to successful patient outcomes, program leadership energized staff support. Staff were encouraged to perform at the top of their scope of practice and engage in training and consultation. Each staff member was delegated a role in the process to inspire buy-in.
Preparation for the Shift
October was spent preparing for a seamless, one-day implementation of proposed changes, including implementation of updated clinical policies, procedures, and document templates (rewritten to include only clinically appropriate information required by VA policy or the Joint Commission); streamlined staff schedules; and utilization of staff-developed and research/policy-driven EBP handbook. Finally, the Brief Addiction Monitor (BAM) was selected as objective criteria to consistently assess patient progress in treatment, and staff were instructed to use this measure at regular intervals and for all levels of care.
Emphasis was placed on ongoing fortification of staff and patient support for the reorganization. For example, the Addiction Severity Index, though not required by policy, was historically used, adding 90 minutes to the evaluation and admission session. Staff agreed to remove this measure to improve clinician availability. Staff were also empowered to rename the redesigned program, and chose Substance Use Disorders Service (SUDS).
Process Changes
To achieve same-day access to clinical care, program leadership created a daily morning orientation group. Patients are scheduled or may attend as a walk-in. The orientation’s purpose is to explain what services are available and to offer each patient an opportunity for immediate evaluation and treatment. Staff schedules were modified to provide patient evaluation appointment slots immediately following orientation. The number of immediate evaluation slots was initially assessed by analyzing the demand for treatment over the previous 6 months, determining the daily mean, and setting the number of slots to accommodate 3 standard deviations above the daily mean. If a patient in a daily orientation group expresses a willingness to engage in treatment, he or she is immediately evaluated by a counselor during a 90-minute session and seen by a psychiatrist to determine whether pharmacologic treatment would be appropriate. If needed, the medication is prescribed that day. The primary purpose of the patient’s initial clinical evaluation is to determine the most appropriate level of care based on ASAM criteria. Also available were 90-minute afternoon evaluation appointments with psychiatrists for patients who walk into the clinic after the morning orientation group had ended.
Prior to the redesign, clinic psychiatrists were minimally prescribing evidence-based pharmacotherapy for sobriety support. At the time of redesign, only 8% of patients diagnosed with opioid use disorders (OUDs) were prescribed buprenorphine/naloxone or naltrexone. Just 1.9% of patients diagnosed with alcohol use disorder (AUD) were prescribed naltrexone or acamprosate. With the redesign, access to these medications has significantly expanded.
All templates were redesigned to ensure consistent documentation. This change decreased the overall provider task burden, and explicitly supported the use of ASAM multidimensional criteria and the Brief Addiction Monitor (BAM) to identify a pretreatment baseline score and track each patient’s clinical progress.13 Evidence-based written curricula were standardized for individual and group psychotherapies to reduce provider and programmatic variation.
The redesign creates distinct levels of care based on ASAM criteria, including harm reduction, ambulatory detoxification, outpatient group and individual psychotherapy, an evidence-based Intensive Outpatient Program (IOP), and aftercare. Application of the ASAM standards has allowed clinicians to make accurate placement decisions that best meet individual patient needs and to serve as effective stewards even with limited treatment and financial resources. Although JAHVH does not have a residential SUD program, procedures were developed to refer veterans to community-based residential treatment programs when appropriate.
Group Therapies
With the redesign, SUDS was no longer exclusively a 12-step program; however, it still supported and recognized the value of this approach for some patients. A psychologist periodically audits group sessions to prevent drift from that group’s curriculum. Counselors are assigned to weekly hour-long clinical supervision sessions with a psychologist to review patient care and reinforce the application of evidence-based individualized treatment.
After reviewing empirical literature and VA directives, CBT-SUD was adopted. It encompasses individual and group interventions, such as motivational interviewing (MI), contingency management (CM), and medication-assisted therapies as primary therapeutic treatment modalities, all of which have demonstrated efficacy as measured by length of sobriety postintervention.9,14,15
Clinical Staff Improvements
Staff were reorganized into 3 interdisciplinary treatment teams. A weekly team meeting is scheduled to coordinate care and discuss the treatment of complex patients. Clinical staff focus has shifted from case-management to diagnosis and treatment; now patients are referred to their primary care team’s social worker for case management services. Allowing clinical staff to focus solely on the diagnosis and clinical treatment of SUDs has significantly enhanced productivity and morale.
Staff receive training in the newly adopted interventions during brief monthly refresher courses provided by inhouse psychologists. Additional training includes participation in local and national SUD teleconferences and onsite meetings with experts in harm reduction and motivational interventions. During the transition, clinicians were encouraged to attend staff resiliency training. Continuing education was available to the SUDS psychiatrists and all inpatient and outpatient psychiatrists at JAHVH. Recently, this educational initiative was expanded to include all primary care and inpatient internal medicine physicians.
Implementation
On November 2, 2015, all planned programmatic changes were simultaneously implemented. On that day, clinician and patient schedules changed, the new EBP curriculum was administered, the use of streamlined documentation procedures began, and daily orientation groups followed by same-day evaluations were initiated.
The pretreatment sobriety requirement was eliminated as a barrier to care, and the program began to use a harm-reduction treatment track as recommended by ASAM guidelines. Patients with urgent or emergent medical or psychiatric problems were immediately assessed by SUDS health care providers and treated in the clinic or transported to the emergency department. Previously unavailable, patient access to ambulatory detoxification was initiated. The prescription of buprenorphine/naloxone for the treatment of OUD treatments increased from 1 prescriber to all 3.
Three months after program reorganization, the leadership reviewed overall workflow, conducted patient satisfaction surveys, and evaluated facility use and productivity. To address patient needs and facilitate optimal use of space, the number of same-day evaluation slots was reduced while the number of individual therapy slots was increased.
Staff meet in workgroups to discuss EBPs and further refine content with feedback from the supervisory clinician and team psychologists who routinely audit group therapy sessions. Staff report ongoing benefit from weekly supervision with a clinical psychologist. An inpatient addiction consultation team that uses existing manpower and resources has been developed.
Program Goals and Outcomes
The SUDS program serves more patients at multiple levels of standardized care with 2 fewer full-time positions. One counselor and one advanced practice registered nurse were reallocated to different programs within the JAHVH VA mental health clinic. Following a review of all program clinic profiles in the VA’s Computerized Patient Record System (CPRS) for utilization, accuracy, and necessity, and allowing for accurate program data capture, the transition resulted in a reduction of distinct clinics from 114 to 67 (-58.7%). In fiscal year 2018, review of CPRS yielded 19,786 total visits (3,645 unique visits).
Eliminate Patient Wait Tme
Patient wait time, as measured in CPRS from date of initial evaluation to date of treatment was reduced from an average of 33 days to 0 within 2 weeks of program implementation. A review of CPRS data also indicated that preadmission attrition dropped from 54% to < 1%; all patients desiring treatment are assigned a counselor and treatment is initiated the same day.
Adopt ASAM Criteria
After the redesign, patients have received more appropriate care based on individualized treatment plans. Due to the implementation of a fluid and supportive model, patients can move through levels of care as clinical need dictates rather than failing treatment and having to reengage. Staff receive ongoing education on the use of ASAM. Evaluation and treatment plan templates now reflect assignment to level of care rationale using ASAM guidelines.
Use of Evidence-Based Psychotherapeutic Treatments
More consistent, coordinated, and effective psychotherapies have improved patient care. The program’s previous issues with patients receiving conflicting treatment guidance from different providers has been resolved. Duplicate and ineffective treatments, including multiple readmissions to the IOP level of care, overemphasis of abstinence-based modalities for patients in active use, and referrals to inpatient SUD care under the assumption that “higher level of care is better” have ceased through staff education, leadership support, and appropriate staffing and communication. Review of patient advocate complaints tracked by and resolved by the service demonstrated an 80% decrease in patient advocate complaints regarding SUD clinic services.
Implement Evidence-Based Psychopharmacologic Treatments
The pharmacotherapy education initiative yielded tangible benefits and is likely a significant contributor to the program’s improved clinical outcomes. Prescription of pharmacotherapy for patients with OUD has risen from 8% to 25.1% in eligible patients. Appropriate medication prescription for the treatment of AUD has risen from 1.9% to 9.8% in eligible patients. These data are reflected in the VA Pharmaceutical Drug Safety Initiative (PDSI) dashboard.
Streamline Documentation
Significantly reducing the charting burden was likely a significant contributor to increased provider productivity and improved patient outcomes. Regular meetings between SUDS leadership and clinical informatics ensure that standardized note templates meet hospital policy and gather all necessary accreditation information.
Improve Employee Morale
Increased staff morale is indicated by a noticeable reduction in employee sick days; a decrease of > 20% (over the same time period the previous year), per the VA electronic timekeeping system, during the first 6 months following the November 2 program implementation.
SUDS Inpatient Addiction Consult Team
In January of 2017, SUDS began an inpatient medicine consultation service to offer evaluation, pharmacotherapy, and supportive counseling to patients diagnosed with SUDs who had been admitted to inpatient medical and surgical services. This team includes existing SUDS staff members reallocated to the inpatient service, is led by a SUDS psychiatrist, and includes 3 multidisciplinary clinicians with extensive training in assessment, diagnosis, and treatment planning of SUDs and comorbid conditions. Prior to implementation, the SUDS inpatient addiction consult team met with hospital leadership and attending physicians for inpatient medicine and psychiatry physicians.
To access the SUDS inpatient addiction consult team, physicians request a consult. Patients are offered an evaluation and are assigned to a level of care with orders for outpatient appointments with a counselor and psychiatrist within 7 days of hospital discharge. Medication-assisted treatment for chronic SUDs is implemented while patients remain admitted to the inpatient medical service. In fiscal year 2018, the SUDS inpatient addiction consult team performed 1,428 inpatient evaluations.
Consistent Treatment Outcome Measures
The BAM is a clinical tool designed to measure patient outcomes in substance use disorders.13 Its 17-item scale measures substance use risk factors that may lead to relapse, and protective factors that are recovery-oriented behaviors that help prevent relapse. It demonstrates sensitivity to change and has excellent test-retest reliability. The BAM has been in use in the addictions treatment program since 2011 but was previously administered only after admission to the IOP and again after a 30- to 90-day follow-up period. Since the program redesign, all SUDS patients are administered the BAM at their initial evaluation and at each individual appointment thereafter. The initial BAM assessment encompasses the previous 30 days; this 30-day version is also used for monthly follow-ups. For BAM assessments that occur within 30 days from the time of the last evaluation, a 7-day version is used. Prior to the redesign, about 24% of patients received a follow-up 30-day BAM assessment.12 Per CPRS review of veterans participating in continued treatment, the rate rose to 100% 3 months after the redesign.
When program staff compared preredesign and postredesign BAM data, they detected significant clinical differences. Data demonstrate a 22.2% improvement in protective factors, including patient confidence in their ability to remain abstinent; engaging in self-help activities, such as attending Alcoholics Anonymous meetings; engaging in organized spiritual activities; going to school, working, or volunteering; securing a regular income; and time spent with friends or family who are supportive of recovery.
The data also show a marked reduction in substance use at follow-up points in treatment and a corresponding decrease in risk factors. One item of the BAM assesses patient level of satisfaction with their treatment. Since the redesign, patients report that they are “considerably” satisfied with their SUD treatment.
Currently, program staff are conducting a review of BAM scores by level of care to further parse the impact of various treatments and best target patient need using measurement-based care and EBP, such as contingency management, which provides small monetary incentives when patients maintain clean urine drug screens.16 In addition, the program plans to achieve more uniformity in BAM assessment intervals at all levels of care, and possibly also integrate BAM data into SUD group therapies. Correlation of the BAM scores to other metrics, such as readmission to inpatient medicine, relapse, urine drug screen, or critical laboratory values, will provide additional insight into impact of programmatic changes.
Discussion
Feedback from other clinics and services within the hospital has been very positive. Some providers have reported that they appreciate the ease and availability of access to SUDS. Additionally, patients engaged in treatment prior to the redesign have been contacted for an updated evaluation and assignment to a counselor and appropriate level of care. From the staff’s perspective, the shift to immediate access to care has allowed a more streamlined process with fewer hurdles for patient admission. Staff report that they now feel empowered to meet the needs of veterans in a comprehensive, same-day fashion.
The success of our redesign was contingent on internal and external sta
The successful implementation of these changes has revealed several important elements regarding patient care. The first lesson was that improving access and integrating best practices is possible without additional resources, outside monies, or disruption to patient services. With the support of MH&BSS leadership, the program streamlined existing processes and used both staff and clinic resources more efficiently.
The second lesson involved the importance of continually reviewing and revising standard operating procedures to match the needs of the current patient population. Policies and procedures that once were viewed as potential barriers to change have been replaced with a more flexible approach and willingness to evolve.
As a result, far fewer patients have been lost to treatment. The time and resources that staff historically dedicated to nonclinical patient care are now redirected to immediate service provision. This increase in operational efficiency and treatment efficacy has resulted in a boost to staff morale, even during a time of immense change and increased productivity. Program staff are now able to personally witness the significant changes in their patients’ lives and feel a sense of pride at being a member of a hard-working team that provides the highest quality of substance use treatment. This is critical to job satisfaction and meets the VA mission to provide timely, effective, and evidence-based treatments to patients.
Conclusion
JAHVH strives to continue to provide the highest quality of SUD treatment available. Future directions aim to streamline clinic operations by constantly monitoring and reviewing workloads, while also considering patient feedback. A continuous review of EBP is part of our clinic’s culture. Program leadership endeavors to promote an open environment where providers can share their triumphs and frustrations and foster a team approach to problem solving. Further plans include expanding the range of treatment levels offered by developing a residential SUD treatment facility.
1. Substance Abuse and Mental Health Services Administration. 2015 National Survey on Drug Use and Health: Summary of the Effects of the 2015 NSDUH Questionnaire Redesign: Implications for Data Users. https://www.samhsa.gov/data/sites/default/files/NSDUH-TrendBreak-2015.pdf. Published June 2016. Accessed June 12, 2019.
2. Substance Abuse and Mental Health Services Administration. Results from the 2013 National Survey on Drug Use and Health: Summary of National Findings. NSDUH Series H-48, HHS Publication No. (SMA) 14-4863. Rockville, MD: Substance Abuse and Mental Health Services Administration; 2014.
3. Donovan DM, Rosengren DB, Downey L, Cox GB, Sloan PDSKL. Attrition prevention with individuals awaiting publicly funded drug treatment. Addiction. 2001;96(8):1149-1160.
4. Hser Y, Maglione M, Polinsky ML, Anglin MD. Predicting treatment entry among treatment-seeking drug abusers. J Subst Abuse Treatment. 1997;15(3):213-220.
5. Stark MJ, Campbell BK, Brinkerhoff CV. “Hello, may we help you?” A study of attrition prevention at the time of the first phone contact with substance-abusing clients. Am J Drug Alcohol Abuse. 1990;16:67-76.
6. US Department of Veterans Affairs. Uniform Mental Health Services in VA Medical Centers and Clinics. VHA Handbook 1160.01. https://www.va.gov/vhapublications/ViewPublication.asp?pub_ID=1762. Updated November 2015. Accessed December 12, 2017.
7. Veterans Access, Choice and Accountability Act of 2014, 2 USC § 933.
8. DeMarce JM, Gnys M, Raffa SD, Karlin, BE. Cognitive Behavioral Therapy for Substance Use Disorders Among Veterans: Therapist Manual. Washington, DC: US Department of Veterans Affairs; 2014.
9. Mee-Lee D, Shulman GD, Fishman MJ, Gastfriend DR, Miller MM, eds. The ASAM Criteria: Treatment Criteria for Addictive, Substance-Related, and Co-Occurring Conditions. 3rd ed. Carson City, NV: The Change Companies; 2013.
10. Substance Abuse and Mental Health Services Administration and National Institute on Alcohol Abuse and Alcoholism. Medication for the Treatment of Alcohol Use Disorder: A Brief Guide. HHS Publication No. (SMA) 15-4907. Rockville, MD: Substance Abuse and Mental Health Services Administration; 2015.
11. Substance Abuse and Mental Health Services Administration and National Institute on Alcohol Abuse and Alcoholism. Medication for Opioid Use Disorder – Full Document. HHS Publication No. (SMA) 18-5063FULLDOC. Rockville, MD: Substance Abuse and Mental Health Services Administration; 2018.
12. Winn JL, Shealy SE, Kropp GJ, Felkins-Dohm D, Gonzales-Nolas C, Francis E. Housing assistance and case management: Improving access to substance use disorder treatment for homeless veterans. Psychological Serv. 2013;10(2):233-240.
13. Cacciola JS, Alterman AI, DePhilippis D, et al. Development and initial evaluation of the Brief Addiction Monitor (BAM). J Subst Abuse Treatment. 2013;44(3):256-263.
14. McHugh RK, Hearon BA, Otto MW. Cognitive-behavioral therapy for substance use disorders, Psychiatr Clinics North Am. 2010;33:511–525.
15. Karlin, BE, Cross, G. From the laboratory to the therapy room: national dissemination and implementation of evidence-based psychotherapies in the U.S. Department of Veterans Affairs health care system. Am Psychol. 2014;69:19-33.
16. DePhilippis D, Petry NM, Bonn-Miller MO, Rosenbach SB, McKay JR. The national implementation of contingency management (CM) in the Department of Veterans Affairs: attendance at CM sessions and substance use outcomes, Drug Alcohol Dependence. 2018;185:367-373.
Substance use disorders (SUDs) are an increasing public health concern in the US. The 2015 National Survey on Drug Use and Health indicated that 27 million people (8% of the US population) reported current use of recreational drugs or misuse of alcohol or prescription medications.1 The 2013 National Survey on Drug Use and Health indicated that 1.5 million veterans (roughly 6.6%) met the criteria for a SUD.2 More than 50% of patients awaiting entry into a SUD treatment program will never achieve admission due, in part, to long wait times.3-5
National attention has been focused on increasing veteran access to quality treatment based on evidence-based practices (EBPs). Several national legislative measures and treatment protocols have been implemented: the Uniform Mental Health Services in US Department of Veterans Affairs (VA) medical centers and clinics; Veterans Access, Choice, and Accountability Act (2014); Cognitive Behavioral Therapy for Substance Use Disorders (CBT-SUD) Training Program; and the Psychotropic Drug Safety Initiative (PDSI).6-8 Consistent with these directives and in line with American Society of Addiction Medicine (ASAM) and Substance Abuse and Mental Health Services Administration (SAMHSA) guidelines for medication-assisted therapies (MAT),the James A. Haley Veterans’ Hospital (JAHVH) Mental Health and Behavioral Sciences Service (MH&BSS) Substance Use Disorders Service (SUDS) in Tampa, Florida, implemented an evidence-based, treatment-on-demand model of care.9-11
Meeting SUD Treatment Needs
What does the new supervisor of a clinical program do when a 24-employee outpatient VA Alcohol and Drug Addiction Treatment Program (ADATP) has an average 33-day wait time for treatment with 54% of patients lost to care between initial evaluation and admission?12 Patients lacked consistent access to SUD pharmacotherapy. The national VA clinical performance indicators were substandard and there are no additional resources available to apply to the program.
At JAHVH the program supervisor enlisted hospital leadership to support program redesign. The redesign sought to improve operational efficiency and eliminate patient wait time; adopt national standards for assessment and treatment developed by ASAM; implement strictly evidence-based psychotherapeutic treatments; educate program psychiatrists about evidence-based psychopharmacologic treatments and hold them accountable for patient adherence; streamline documentation templates; free clinical providers from nonclinical tasks; create an inpatient addiction consult team to diagnose and treat chronic hospitalized patients with SUDs; ensure continuity of care; and, standardize consistent, objective measures of patient response to treatment to track the program’s effectiveness.
In this article, the authors provide an explanation of the clinical, theoretical foundation and the practical steps taken to design and implement this transformation. They then describe the lessons learned, hoping that their process will serve as a model for those in similar situations.
Program Redesign
July 1, 2015, a new program supervisor was hired and began a 2-month evaluation and analysis of the program with input from leadership, staff, and hospital/community stakeholders. September 1, the monthlong process of developing the redesign began. On September 30 the plan was presented to, and approved by, MH&BSS leadership. October was spent preparing for change with an implementation date of November 2 selected. On November 2, 2015, the complete redesign was implemented.
Needs Assessment
A needs assessment yielded improvement opportunities in program structure (levels of care); clinical content; staff and resource allocation, including clinical workflow and management systems. Staff identified philosophical and practical variance in the program, often leading to confusion for patients and clinicians and potentially resulting in disparate quality care and patient outcomes. Recommendations for addressing these needs included incorporating ASAM guidelines for assignment to clinically appropriate levels of care, implementation of consistent EBPs for SUD and comorbid conditions,9 and emphasis on staff training and development to champion evidence-based program philosophy and service delivery.
The assessment determined that the average waitlist time was 33 days, and patients were required to abstain from substance or alcohol use prior to admission to the Intensive Outpatient Program. If a waitlisted patient relapsed, she or he was removed from the waitlist and denied admission. A study conducted at JAHVH reported that 54% of waitlisted patients in this clinic (prior to November 2, 2015) never were admitted to the program.12 Access to care was considered a significant issue.
Program Implementation
September was spent developing a comprehensive redesign of the SUD clinic. The vision included incorporating all ASAM levels of care; creating an evidence-based, treatment-on-demand model of care; and, securing the support of MH&BSS leadership team, staff, and patients for the redesign. The supervisory clinician interviewed staff both individually and as a group. Clinicians were provided extensive training on EBP for SUDs, including psychotherapies, psychosocial treatments, and psychopharmacologic interventions. A journal club was started with staff-generated topics that offered articles sharing current research, EBPs, and psychotherapeutic techniques, continuing education on substances, and management of coexisting diagnoses. Clinicians increased the frequency of SUD in-service trainings. Psychiatrists provided several Grand Rounds to the MH&BSS service. All counselors were assigned to 1 of the program’s 3 clinical psychologists for individual weekly clinical supervision.
By providing all staff with current, evidence-based, clinically relevant treatment information and emphasizing its relationship to successful patient outcomes, program leadership energized staff support. Staff were encouraged to perform at the top of their scope of practice and engage in training and consultation. Each staff member was delegated a role in the process to inspire buy-in.
Preparation for the Shift
October was spent preparing for a seamless, one-day implementation of proposed changes, including implementation of updated clinical policies, procedures, and document templates (rewritten to include only clinically appropriate information required by VA policy or the Joint Commission); streamlined staff schedules; and utilization of staff-developed and research/policy-driven EBP handbook. Finally, the Brief Addiction Monitor (BAM) was selected as objective criteria to consistently assess patient progress in treatment, and staff were instructed to use this measure at regular intervals and for all levels of care.
Emphasis was placed on ongoing fortification of staff and patient support for the reorganization. For example, the Addiction Severity Index, though not required by policy, was historically used, adding 90 minutes to the evaluation and admission session. Staff agreed to remove this measure to improve clinician availability. Staff were also empowered to rename the redesigned program, and chose Substance Use Disorders Service (SUDS).
Process Changes
To achieve same-day access to clinical care, program leadership created a daily morning orientation group. Patients are scheduled or may attend as a walk-in. The orientation’s purpose is to explain what services are available and to offer each patient an opportunity for immediate evaluation and treatment. Staff schedules were modified to provide patient evaluation appointment slots immediately following orientation. The number of immediate evaluation slots was initially assessed by analyzing the demand for treatment over the previous 6 months, determining the daily mean, and setting the number of slots to accommodate 3 standard deviations above the daily mean. If a patient in a daily orientation group expresses a willingness to engage in treatment, he or she is immediately evaluated by a counselor during a 90-minute session and seen by a psychiatrist to determine whether pharmacologic treatment would be appropriate. If needed, the medication is prescribed that day. The primary purpose of the patient’s initial clinical evaluation is to determine the most appropriate level of care based on ASAM criteria. Also available were 90-minute afternoon evaluation appointments with psychiatrists for patients who walk into the clinic after the morning orientation group had ended.
Prior to the redesign, clinic psychiatrists were minimally prescribing evidence-based pharmacotherapy for sobriety support. At the time of redesign, only 8% of patients diagnosed with opioid use disorders (OUDs) were prescribed buprenorphine/naloxone or naltrexone. Just 1.9% of patients diagnosed with alcohol use disorder (AUD) were prescribed naltrexone or acamprosate. With the redesign, access to these medications has significantly expanded.
All templates were redesigned to ensure consistent documentation. This change decreased the overall provider task burden, and explicitly supported the use of ASAM multidimensional criteria and the Brief Addiction Monitor (BAM) to identify a pretreatment baseline score and track each patient’s clinical progress.13 Evidence-based written curricula were standardized for individual and group psychotherapies to reduce provider and programmatic variation.
The redesign creates distinct levels of care based on ASAM criteria, including harm reduction, ambulatory detoxification, outpatient group and individual psychotherapy, an evidence-based Intensive Outpatient Program (IOP), and aftercare. Application of the ASAM standards has allowed clinicians to make accurate placement decisions that best meet individual patient needs and to serve as effective stewards even with limited treatment and financial resources. Although JAHVH does not have a residential SUD program, procedures were developed to refer veterans to community-based residential treatment programs when appropriate.
Group Therapies
With the redesign, SUDS was no longer exclusively a 12-step program; however, it still supported and recognized the value of this approach for some patients. A psychologist periodically audits group sessions to prevent drift from that group’s curriculum. Counselors are assigned to weekly hour-long clinical supervision sessions with a psychologist to review patient care and reinforce the application of evidence-based individualized treatment.
After reviewing empirical literature and VA directives, CBT-SUD was adopted. It encompasses individual and group interventions, such as motivational interviewing (MI), contingency management (CM), and medication-assisted therapies as primary therapeutic treatment modalities, all of which have demonstrated efficacy as measured by length of sobriety postintervention.9,14,15
Clinical Staff Improvements
Staff were reorganized into 3 interdisciplinary treatment teams. A weekly team meeting is scheduled to coordinate care and discuss the treatment of complex patients. Clinical staff focus has shifted from case-management to diagnosis and treatment; now patients are referred to their primary care team’s social worker for case management services. Allowing clinical staff to focus solely on the diagnosis and clinical treatment of SUDs has significantly enhanced productivity and morale.
Staff receive training in the newly adopted interventions during brief monthly refresher courses provided by inhouse psychologists. Additional training includes participation in local and national SUD teleconferences and onsite meetings with experts in harm reduction and motivational interventions. During the transition, clinicians were encouraged to attend staff resiliency training. Continuing education was available to the SUDS psychiatrists and all inpatient and outpatient psychiatrists at JAHVH. Recently, this educational initiative was expanded to include all primary care and inpatient internal medicine physicians.
Implementation
On November 2, 2015, all planned programmatic changes were simultaneously implemented. On that day, clinician and patient schedules changed, the new EBP curriculum was administered, the use of streamlined documentation procedures began, and daily orientation groups followed by same-day evaluations were initiated.
The pretreatment sobriety requirement was eliminated as a barrier to care, and the program began to use a harm-reduction treatment track as recommended by ASAM guidelines. Patients with urgent or emergent medical or psychiatric problems were immediately assessed by SUDS health care providers and treated in the clinic or transported to the emergency department. Previously unavailable, patient access to ambulatory detoxification was initiated. The prescription of buprenorphine/naloxone for the treatment of OUD treatments increased from 1 prescriber to all 3.
Three months after program reorganization, the leadership reviewed overall workflow, conducted patient satisfaction surveys, and evaluated facility use and productivity. To address patient needs and facilitate optimal use of space, the number of same-day evaluation slots was reduced while the number of individual therapy slots was increased.
Staff meet in workgroups to discuss EBPs and further refine content with feedback from the supervisory clinician and team psychologists who routinely audit group therapy sessions. Staff report ongoing benefit from weekly supervision with a clinical psychologist. An inpatient addiction consultation team that uses existing manpower and resources has been developed.
Program Goals and Outcomes
The SUDS program serves more patients at multiple levels of standardized care with 2 fewer full-time positions. One counselor and one advanced practice registered nurse were reallocated to different programs within the JAHVH VA mental health clinic. Following a review of all program clinic profiles in the VA’s Computerized Patient Record System (CPRS) for utilization, accuracy, and necessity, and allowing for accurate program data capture, the transition resulted in a reduction of distinct clinics from 114 to 67 (-58.7%). In fiscal year 2018, review of CPRS yielded 19,786 total visits (3,645 unique visits).
Eliminate Patient Wait Tme
Patient wait time, as measured in CPRS from date of initial evaluation to date of treatment was reduced from an average of 33 days to 0 within 2 weeks of program implementation. A review of CPRS data also indicated that preadmission attrition dropped from 54% to < 1%; all patients desiring treatment are assigned a counselor and treatment is initiated the same day.
Adopt ASAM Criteria
After the redesign, patients have received more appropriate care based on individualized treatment plans. Due to the implementation of a fluid and supportive model, patients can move through levels of care as clinical need dictates rather than failing treatment and having to reengage. Staff receive ongoing education on the use of ASAM. Evaluation and treatment plan templates now reflect assignment to level of care rationale using ASAM guidelines.
Use of Evidence-Based Psychotherapeutic Treatments
More consistent, coordinated, and effective psychotherapies have improved patient care. The program’s previous issues with patients receiving conflicting treatment guidance from different providers has been resolved. Duplicate and ineffective treatments, including multiple readmissions to the IOP level of care, overemphasis of abstinence-based modalities for patients in active use, and referrals to inpatient SUD care under the assumption that “higher level of care is better” have ceased through staff education, leadership support, and appropriate staffing and communication. Review of patient advocate complaints tracked by and resolved by the service demonstrated an 80% decrease in patient advocate complaints regarding SUD clinic services.
Implement Evidence-Based Psychopharmacologic Treatments
The pharmacotherapy education initiative yielded tangible benefits and is likely a significant contributor to the program’s improved clinical outcomes. Prescription of pharmacotherapy for patients with OUD has risen from 8% to 25.1% in eligible patients. Appropriate medication prescription for the treatment of AUD has risen from 1.9% to 9.8% in eligible patients. These data are reflected in the VA Pharmaceutical Drug Safety Initiative (PDSI) dashboard.
Streamline Documentation
Significantly reducing the charting burden was likely a significant contributor to increased provider productivity and improved patient outcomes. Regular meetings between SUDS leadership and clinical informatics ensure that standardized note templates meet hospital policy and gather all necessary accreditation information.
Improve Employee Morale
Increased staff morale is indicated by a noticeable reduction in employee sick days; a decrease of > 20% (over the same time period the previous year), per the VA electronic timekeeping system, during the first 6 months following the November 2 program implementation.
SUDS Inpatient Addiction Consult Team
In January of 2017, SUDS began an inpatient medicine consultation service to offer evaluation, pharmacotherapy, and supportive counseling to patients diagnosed with SUDs who had been admitted to inpatient medical and surgical services. This team includes existing SUDS staff members reallocated to the inpatient service, is led by a SUDS psychiatrist, and includes 3 multidisciplinary clinicians with extensive training in assessment, diagnosis, and treatment planning of SUDs and comorbid conditions. Prior to implementation, the SUDS inpatient addiction consult team met with hospital leadership and attending physicians for inpatient medicine and psychiatry physicians.
To access the SUDS inpatient addiction consult team, physicians request a consult. Patients are offered an evaluation and are assigned to a level of care with orders for outpatient appointments with a counselor and psychiatrist within 7 days of hospital discharge. Medication-assisted treatment for chronic SUDs is implemented while patients remain admitted to the inpatient medical service. In fiscal year 2018, the SUDS inpatient addiction consult team performed 1,428 inpatient evaluations.
Consistent Treatment Outcome Measures
The BAM is a clinical tool designed to measure patient outcomes in substance use disorders.13 Its 17-item scale measures substance use risk factors that may lead to relapse, and protective factors that are recovery-oriented behaviors that help prevent relapse. It demonstrates sensitivity to change and has excellent test-retest reliability. The BAM has been in use in the addictions treatment program since 2011 but was previously administered only after admission to the IOP and again after a 30- to 90-day follow-up period. Since the program redesign, all SUDS patients are administered the BAM at their initial evaluation and at each individual appointment thereafter. The initial BAM assessment encompasses the previous 30 days; this 30-day version is also used for monthly follow-ups. For BAM assessments that occur within 30 days from the time of the last evaluation, a 7-day version is used. Prior to the redesign, about 24% of patients received a follow-up 30-day BAM assessment.12 Per CPRS review of veterans participating in continued treatment, the rate rose to 100% 3 months after the redesign.
When program staff compared preredesign and postredesign BAM data, they detected significant clinical differences. Data demonstrate a 22.2% improvement in protective factors, including patient confidence in their ability to remain abstinent; engaging in self-help activities, such as attending Alcoholics Anonymous meetings; engaging in organized spiritual activities; going to school, working, or volunteering; securing a regular income; and time spent with friends or family who are supportive of recovery.
The data also show a marked reduction in substance use at follow-up points in treatment and a corresponding decrease in risk factors. One item of the BAM assesses patient level of satisfaction with their treatment. Since the redesign, patients report that they are “considerably” satisfied with their SUD treatment.
Currently, program staff are conducting a review of BAM scores by level of care to further parse the impact of various treatments and best target patient need using measurement-based care and EBP, such as contingency management, which provides small monetary incentives when patients maintain clean urine drug screens.16 In addition, the program plans to achieve more uniformity in BAM assessment intervals at all levels of care, and possibly also integrate BAM data into SUD group therapies. Correlation of the BAM scores to other metrics, such as readmission to inpatient medicine, relapse, urine drug screen, or critical laboratory values, will provide additional insight into impact of programmatic changes.
Discussion
Feedback from other clinics and services within the hospital has been very positive. Some providers have reported that they appreciate the ease and availability of access to SUDS. Additionally, patients engaged in treatment prior to the redesign have been contacted for an updated evaluation and assignment to a counselor and appropriate level of care. From the staff’s perspective, the shift to immediate access to care has allowed a more streamlined process with fewer hurdles for patient admission. Staff report that they now feel empowered to meet the needs of veterans in a comprehensive, same-day fashion.
The success of our redesign was contingent on internal and external sta
The successful implementation of these changes has revealed several important elements regarding patient care. The first lesson was that improving access and integrating best practices is possible without additional resources, outside monies, or disruption to patient services. With the support of MH&BSS leadership, the program streamlined existing processes and used both staff and clinic resources more efficiently.
The second lesson involved the importance of continually reviewing and revising standard operating procedures to match the needs of the current patient population. Policies and procedures that once were viewed as potential barriers to change have been replaced with a more flexible approach and willingness to evolve.
As a result, far fewer patients have been lost to treatment. The time and resources that staff historically dedicated to nonclinical patient care are now redirected to immediate service provision. This increase in operational efficiency and treatment efficacy has resulted in a boost to staff morale, even during a time of immense change and increased productivity. Program staff are now able to personally witness the significant changes in their patients’ lives and feel a sense of pride at being a member of a hard-working team that provides the highest quality of substance use treatment. This is critical to job satisfaction and meets the VA mission to provide timely, effective, and evidence-based treatments to patients.
Conclusion
JAHVH strives to continue to provide the highest quality of SUD treatment available. Future directions aim to streamline clinic operations by constantly monitoring and reviewing workloads, while also considering patient feedback. A continuous review of EBP is part of our clinic’s culture. Program leadership endeavors to promote an open environment where providers can share their triumphs and frustrations and foster a team approach to problem solving. Further plans include expanding the range of treatment levels offered by developing a residential SUD treatment facility.
Substance use disorders (SUDs) are an increasing public health concern in the US. The 2015 National Survey on Drug Use and Health indicated that 27 million people (8% of the US population) reported current use of recreational drugs or misuse of alcohol or prescription medications.1 The 2013 National Survey on Drug Use and Health indicated that 1.5 million veterans (roughly 6.6%) met the criteria for a SUD.2 More than 50% of patients awaiting entry into a SUD treatment program will never achieve admission due, in part, to long wait times.3-5
National attention has been focused on increasing veteran access to quality treatment based on evidence-based practices (EBPs). Several national legislative measures and treatment protocols have been implemented: the Uniform Mental Health Services in US Department of Veterans Affairs (VA) medical centers and clinics; Veterans Access, Choice, and Accountability Act (2014); Cognitive Behavioral Therapy for Substance Use Disorders (CBT-SUD) Training Program; and the Psychotropic Drug Safety Initiative (PDSI).6-8 Consistent with these directives and in line with American Society of Addiction Medicine (ASAM) and Substance Abuse and Mental Health Services Administration (SAMHSA) guidelines for medication-assisted therapies (MAT),the James A. Haley Veterans’ Hospital (JAHVH) Mental Health and Behavioral Sciences Service (MH&BSS) Substance Use Disorders Service (SUDS) in Tampa, Florida, implemented an evidence-based, treatment-on-demand model of care.9-11
Meeting SUD Treatment Needs
What does the new supervisor of a clinical program do when a 24-employee outpatient VA Alcohol and Drug Addiction Treatment Program (ADATP) has an average 33-day wait time for treatment with 54% of patients lost to care between initial evaluation and admission?12 Patients lacked consistent access to SUD pharmacotherapy. The national VA clinical performance indicators were substandard and there are no additional resources available to apply to the program.
At JAHVH the program supervisor enlisted hospital leadership to support program redesign. The redesign sought to improve operational efficiency and eliminate patient wait time; adopt national standards for assessment and treatment developed by ASAM; implement strictly evidence-based psychotherapeutic treatments; educate program psychiatrists about evidence-based psychopharmacologic treatments and hold them accountable for patient adherence; streamline documentation templates; free clinical providers from nonclinical tasks; create an inpatient addiction consult team to diagnose and treat chronic hospitalized patients with SUDs; ensure continuity of care; and, standardize consistent, objective measures of patient response to treatment to track the program’s effectiveness.
In this article, the authors provide an explanation of the clinical, theoretical foundation and the practical steps taken to design and implement this transformation. They then describe the lessons learned, hoping that their process will serve as a model for those in similar situations.
Program Redesign
July 1, 2015, a new program supervisor was hired and began a 2-month evaluation and analysis of the program with input from leadership, staff, and hospital/community stakeholders. September 1, the monthlong process of developing the redesign began. On September 30 the plan was presented to, and approved by, MH&BSS leadership. October was spent preparing for change with an implementation date of November 2 selected. On November 2, 2015, the complete redesign was implemented.
Needs Assessment
A needs assessment yielded improvement opportunities in program structure (levels of care); clinical content; staff and resource allocation, including clinical workflow and management systems. Staff identified philosophical and practical variance in the program, often leading to confusion for patients and clinicians and potentially resulting in disparate quality care and patient outcomes. Recommendations for addressing these needs included incorporating ASAM guidelines for assignment to clinically appropriate levels of care, implementation of consistent EBPs for SUD and comorbid conditions,9 and emphasis on staff training and development to champion evidence-based program philosophy and service delivery.
The assessment determined that the average waitlist time was 33 days, and patients were required to abstain from substance or alcohol use prior to admission to the Intensive Outpatient Program. If a waitlisted patient relapsed, she or he was removed from the waitlist and denied admission. A study conducted at JAHVH reported that 54% of waitlisted patients in this clinic (prior to November 2, 2015) never were admitted to the program.12 Access to care was considered a significant issue.
Program Implementation
September was spent developing a comprehensive redesign of the SUD clinic. The vision included incorporating all ASAM levels of care; creating an evidence-based, treatment-on-demand model of care; and, securing the support of MH&BSS leadership team, staff, and patients for the redesign. The supervisory clinician interviewed staff both individually and as a group. Clinicians were provided extensive training on EBP for SUDs, including psychotherapies, psychosocial treatments, and psychopharmacologic interventions. A journal club was started with staff-generated topics that offered articles sharing current research, EBPs, and psychotherapeutic techniques, continuing education on substances, and management of coexisting diagnoses. Clinicians increased the frequency of SUD in-service trainings. Psychiatrists provided several Grand Rounds to the MH&BSS service. All counselors were assigned to 1 of the program’s 3 clinical psychologists for individual weekly clinical supervision.
By providing all staff with current, evidence-based, clinically relevant treatment information and emphasizing its relationship to successful patient outcomes, program leadership energized staff support. Staff were encouraged to perform at the top of their scope of practice and engage in training and consultation. Each staff member was delegated a role in the process to inspire buy-in.
Preparation for the Shift
October was spent preparing for a seamless, one-day implementation of proposed changes, including implementation of updated clinical policies, procedures, and document templates (rewritten to include only clinically appropriate information required by VA policy or the Joint Commission); streamlined staff schedules; and utilization of staff-developed and research/policy-driven EBP handbook. Finally, the Brief Addiction Monitor (BAM) was selected as objective criteria to consistently assess patient progress in treatment, and staff were instructed to use this measure at regular intervals and for all levels of care.
Emphasis was placed on ongoing fortification of staff and patient support for the reorganization. For example, the Addiction Severity Index, though not required by policy, was historically used, adding 90 minutes to the evaluation and admission session. Staff agreed to remove this measure to improve clinician availability. Staff were also empowered to rename the redesigned program, and chose Substance Use Disorders Service (SUDS).
Process Changes
To achieve same-day access to clinical care, program leadership created a daily morning orientation group. Patients are scheduled or may attend as a walk-in. The orientation’s purpose is to explain what services are available and to offer each patient an opportunity for immediate evaluation and treatment. Staff schedules were modified to provide patient evaluation appointment slots immediately following orientation. The number of immediate evaluation slots was initially assessed by analyzing the demand for treatment over the previous 6 months, determining the daily mean, and setting the number of slots to accommodate 3 standard deviations above the daily mean. If a patient in a daily orientation group expresses a willingness to engage in treatment, he or she is immediately evaluated by a counselor during a 90-minute session and seen by a psychiatrist to determine whether pharmacologic treatment would be appropriate. If needed, the medication is prescribed that day. The primary purpose of the patient’s initial clinical evaluation is to determine the most appropriate level of care based on ASAM criteria. Also available were 90-minute afternoon evaluation appointments with psychiatrists for patients who walk into the clinic after the morning orientation group had ended.
Prior to the redesign, clinic psychiatrists were minimally prescribing evidence-based pharmacotherapy for sobriety support. At the time of redesign, only 8% of patients diagnosed with opioid use disorders (OUDs) were prescribed buprenorphine/naloxone or naltrexone. Just 1.9% of patients diagnosed with alcohol use disorder (AUD) were prescribed naltrexone or acamprosate. With the redesign, access to these medications has significantly expanded.
All templates were redesigned to ensure consistent documentation. This change decreased the overall provider task burden, and explicitly supported the use of ASAM multidimensional criteria and the Brief Addiction Monitor (BAM) to identify a pretreatment baseline score and track each patient’s clinical progress.13 Evidence-based written curricula were standardized for individual and group psychotherapies to reduce provider and programmatic variation.
The redesign creates distinct levels of care based on ASAM criteria, including harm reduction, ambulatory detoxification, outpatient group and individual psychotherapy, an evidence-based Intensive Outpatient Program (IOP), and aftercare. Application of the ASAM standards has allowed clinicians to make accurate placement decisions that best meet individual patient needs and to serve as effective stewards even with limited treatment and financial resources. Although JAHVH does not have a residential SUD program, procedures were developed to refer veterans to community-based residential treatment programs when appropriate.
Group Therapies
With the redesign, SUDS was no longer exclusively a 12-step program; however, it still supported and recognized the value of this approach for some patients. A psychologist periodically audits group sessions to prevent drift from that group’s curriculum. Counselors are assigned to weekly hour-long clinical supervision sessions with a psychologist to review patient care and reinforce the application of evidence-based individualized treatment.
After reviewing empirical literature and VA directives, CBT-SUD was adopted. It encompasses individual and group interventions, such as motivational interviewing (MI), contingency management (CM), and medication-assisted therapies as primary therapeutic treatment modalities, all of which have demonstrated efficacy as measured by length of sobriety postintervention.9,14,15
Clinical Staff Improvements
Staff were reorganized into 3 interdisciplinary treatment teams. A weekly team meeting is scheduled to coordinate care and discuss the treatment of complex patients. Clinical staff focus has shifted from case-management to diagnosis and treatment; now patients are referred to their primary care team’s social worker for case management services. Allowing clinical staff to focus solely on the diagnosis and clinical treatment of SUDs has significantly enhanced productivity and morale.
Staff receive training in the newly adopted interventions during brief monthly refresher courses provided by inhouse psychologists. Additional training includes participation in local and national SUD teleconferences and onsite meetings with experts in harm reduction and motivational interventions. During the transition, clinicians were encouraged to attend staff resiliency training. Continuing education was available to the SUDS psychiatrists and all inpatient and outpatient psychiatrists at JAHVH. Recently, this educational initiative was expanded to include all primary care and inpatient internal medicine physicians.
Implementation
On November 2, 2015, all planned programmatic changes were simultaneously implemented. On that day, clinician and patient schedules changed, the new EBP curriculum was administered, the use of streamlined documentation procedures began, and daily orientation groups followed by same-day evaluations were initiated.
The pretreatment sobriety requirement was eliminated as a barrier to care, and the program began to use a harm-reduction treatment track as recommended by ASAM guidelines. Patients with urgent or emergent medical or psychiatric problems were immediately assessed by SUDS health care providers and treated in the clinic or transported to the emergency department. Previously unavailable, patient access to ambulatory detoxification was initiated. The prescription of buprenorphine/naloxone for the treatment of OUD treatments increased from 1 prescriber to all 3.
Three months after program reorganization, the leadership reviewed overall workflow, conducted patient satisfaction surveys, and evaluated facility use and productivity. To address patient needs and facilitate optimal use of space, the number of same-day evaluation slots was reduced while the number of individual therapy slots was increased.
Staff meet in workgroups to discuss EBPs and further refine content with feedback from the supervisory clinician and team psychologists who routinely audit group therapy sessions. Staff report ongoing benefit from weekly supervision with a clinical psychologist. An inpatient addiction consultation team that uses existing manpower and resources has been developed.
Program Goals and Outcomes
The SUDS program serves more patients at multiple levels of standardized care with 2 fewer full-time positions. One counselor and one advanced practice registered nurse were reallocated to different programs within the JAHVH VA mental health clinic. Following a review of all program clinic profiles in the VA’s Computerized Patient Record System (CPRS) for utilization, accuracy, and necessity, and allowing for accurate program data capture, the transition resulted in a reduction of distinct clinics from 114 to 67 (-58.7%). In fiscal year 2018, review of CPRS yielded 19,786 total visits (3,645 unique visits).
Eliminate Patient Wait Tme
Patient wait time, as measured in CPRS from date of initial evaluation to date of treatment was reduced from an average of 33 days to 0 within 2 weeks of program implementation. A review of CPRS data also indicated that preadmission attrition dropped from 54% to < 1%; all patients desiring treatment are assigned a counselor and treatment is initiated the same day.
Adopt ASAM Criteria
After the redesign, patients have received more appropriate care based on individualized treatment plans. Due to the implementation of a fluid and supportive model, patients can move through levels of care as clinical need dictates rather than failing treatment and having to reengage. Staff receive ongoing education on the use of ASAM. Evaluation and treatment plan templates now reflect assignment to level of care rationale using ASAM guidelines.
Use of Evidence-Based Psychotherapeutic Treatments
More consistent, coordinated, and effective psychotherapies have improved patient care. The program’s previous issues with patients receiving conflicting treatment guidance from different providers has been resolved. Duplicate and ineffective treatments, including multiple readmissions to the IOP level of care, overemphasis of abstinence-based modalities for patients in active use, and referrals to inpatient SUD care under the assumption that “higher level of care is better” have ceased through staff education, leadership support, and appropriate staffing and communication. Review of patient advocate complaints tracked by and resolved by the service demonstrated an 80% decrease in patient advocate complaints regarding SUD clinic services.
Implement Evidence-Based Psychopharmacologic Treatments
The pharmacotherapy education initiative yielded tangible benefits and is likely a significant contributor to the program’s improved clinical outcomes. Prescription of pharmacotherapy for patients with OUD has risen from 8% to 25.1% in eligible patients. Appropriate medication prescription for the treatment of AUD has risen from 1.9% to 9.8% in eligible patients. These data are reflected in the VA Pharmaceutical Drug Safety Initiative (PDSI) dashboard.
Streamline Documentation
Significantly reducing the charting burden was likely a significant contributor to increased provider productivity and improved patient outcomes. Regular meetings between SUDS leadership and clinical informatics ensure that standardized note templates meet hospital policy and gather all necessary accreditation information.
Improve Employee Morale
Increased staff morale is indicated by a noticeable reduction in employee sick days; a decrease of > 20% (over the same time period the previous year), per the VA electronic timekeeping system, during the first 6 months following the November 2 program implementation.
SUDS Inpatient Addiction Consult Team
In January of 2017, SUDS began an inpatient medicine consultation service to offer evaluation, pharmacotherapy, and supportive counseling to patients diagnosed with SUDs who had been admitted to inpatient medical and surgical services. This team includes existing SUDS staff members reallocated to the inpatient service, is led by a SUDS psychiatrist, and includes 3 multidisciplinary clinicians with extensive training in assessment, diagnosis, and treatment planning of SUDs and comorbid conditions. Prior to implementation, the SUDS inpatient addiction consult team met with hospital leadership and attending physicians for inpatient medicine and psychiatry physicians.
To access the SUDS inpatient addiction consult team, physicians request a consult. Patients are offered an evaluation and are assigned to a level of care with orders for outpatient appointments with a counselor and psychiatrist within 7 days of hospital discharge. Medication-assisted treatment for chronic SUDs is implemented while patients remain admitted to the inpatient medical service. In fiscal year 2018, the SUDS inpatient addiction consult team performed 1,428 inpatient evaluations.
Consistent Treatment Outcome Measures
The BAM is a clinical tool designed to measure patient outcomes in substance use disorders.13 Its 17-item scale measures substance use risk factors that may lead to relapse, and protective factors that are recovery-oriented behaviors that help prevent relapse. It demonstrates sensitivity to change and has excellent test-retest reliability. The BAM has been in use in the addictions treatment program since 2011 but was previously administered only after admission to the IOP and again after a 30- to 90-day follow-up period. Since the program redesign, all SUDS patients are administered the BAM at their initial evaluation and at each individual appointment thereafter. The initial BAM assessment encompasses the previous 30 days; this 30-day version is also used for monthly follow-ups. For BAM assessments that occur within 30 days from the time of the last evaluation, a 7-day version is used. Prior to the redesign, about 24% of patients received a follow-up 30-day BAM assessment.12 Per CPRS review of veterans participating in continued treatment, the rate rose to 100% 3 months after the redesign.
When program staff compared preredesign and postredesign BAM data, they detected significant clinical differences. Data demonstrate a 22.2% improvement in protective factors, including patient confidence in their ability to remain abstinent; engaging in self-help activities, such as attending Alcoholics Anonymous meetings; engaging in organized spiritual activities; going to school, working, or volunteering; securing a regular income; and time spent with friends or family who are supportive of recovery.
The data also show a marked reduction in substance use at follow-up points in treatment and a corresponding decrease in risk factors. One item of the BAM assesses patient level of satisfaction with their treatment. Since the redesign, patients report that they are “considerably” satisfied with their SUD treatment.
Currently, program staff are conducting a review of BAM scores by level of care to further parse the impact of various treatments and best target patient need using measurement-based care and EBP, such as contingency management, which provides small monetary incentives when patients maintain clean urine drug screens.16 In addition, the program plans to achieve more uniformity in BAM assessment intervals at all levels of care, and possibly also integrate BAM data into SUD group therapies. Correlation of the BAM scores to other metrics, such as readmission to inpatient medicine, relapse, urine drug screen, or critical laboratory values, will provide additional insight into impact of programmatic changes.
Discussion
Feedback from other clinics and services within the hospital has been very positive. Some providers have reported that they appreciate the ease and availability of access to SUDS. Additionally, patients engaged in treatment prior to the redesign have been contacted for an updated evaluation and assignment to a counselor and appropriate level of care. From the staff’s perspective, the shift to immediate access to care has allowed a more streamlined process with fewer hurdles for patient admission. Staff report that they now feel empowered to meet the needs of veterans in a comprehensive, same-day fashion.
The success of our redesign was contingent on internal and external sta
The successful implementation of these changes has revealed several important elements regarding patient care. The first lesson was that improving access and integrating best practices is possible without additional resources, outside monies, or disruption to patient services. With the support of MH&BSS leadership, the program streamlined existing processes and used both staff and clinic resources more efficiently.
The second lesson involved the importance of continually reviewing and revising standard operating procedures to match the needs of the current patient population. Policies and procedures that once were viewed as potential barriers to change have been replaced with a more flexible approach and willingness to evolve.
As a result, far fewer patients have been lost to treatment. The time and resources that staff historically dedicated to nonclinical patient care are now redirected to immediate service provision. This increase in operational efficiency and treatment efficacy has resulted in a boost to staff morale, even during a time of immense change and increased productivity. Program staff are now able to personally witness the significant changes in their patients’ lives and feel a sense of pride at being a member of a hard-working team that provides the highest quality of substance use treatment. This is critical to job satisfaction and meets the VA mission to provide timely, effective, and evidence-based treatments to patients.
Conclusion
JAHVH strives to continue to provide the highest quality of SUD treatment available. Future directions aim to streamline clinic operations by constantly monitoring and reviewing workloads, while also considering patient feedback. A continuous review of EBP is part of our clinic’s culture. Program leadership endeavors to promote an open environment where providers can share their triumphs and frustrations and foster a team approach to problem solving. Further plans include expanding the range of treatment levels offered by developing a residential SUD treatment facility.
1. Substance Abuse and Mental Health Services Administration. 2015 National Survey on Drug Use and Health: Summary of the Effects of the 2015 NSDUH Questionnaire Redesign: Implications for Data Users. https://www.samhsa.gov/data/sites/default/files/NSDUH-TrendBreak-2015.pdf. Published June 2016. Accessed June 12, 2019.
2. Substance Abuse and Mental Health Services Administration. Results from the 2013 National Survey on Drug Use and Health: Summary of National Findings. NSDUH Series H-48, HHS Publication No. (SMA) 14-4863. Rockville, MD: Substance Abuse and Mental Health Services Administration; 2014.
3. Donovan DM, Rosengren DB, Downey L, Cox GB, Sloan PDSKL. Attrition prevention with individuals awaiting publicly funded drug treatment. Addiction. 2001;96(8):1149-1160.
4. Hser Y, Maglione M, Polinsky ML, Anglin MD. Predicting treatment entry among treatment-seeking drug abusers. J Subst Abuse Treatment. 1997;15(3):213-220.
5. Stark MJ, Campbell BK, Brinkerhoff CV. “Hello, may we help you?” A study of attrition prevention at the time of the first phone contact with substance-abusing clients. Am J Drug Alcohol Abuse. 1990;16:67-76.
6. US Department of Veterans Affairs. Uniform Mental Health Services in VA Medical Centers and Clinics. VHA Handbook 1160.01. https://www.va.gov/vhapublications/ViewPublication.asp?pub_ID=1762. Updated November 2015. Accessed December 12, 2017.
7. Veterans Access, Choice and Accountability Act of 2014, 2 USC § 933.
8. DeMarce JM, Gnys M, Raffa SD, Karlin, BE. Cognitive Behavioral Therapy for Substance Use Disorders Among Veterans: Therapist Manual. Washington, DC: US Department of Veterans Affairs; 2014.
9. Mee-Lee D, Shulman GD, Fishman MJ, Gastfriend DR, Miller MM, eds. The ASAM Criteria: Treatment Criteria for Addictive, Substance-Related, and Co-Occurring Conditions. 3rd ed. Carson City, NV: The Change Companies; 2013.
10. Substance Abuse and Mental Health Services Administration and National Institute on Alcohol Abuse and Alcoholism. Medication for the Treatment of Alcohol Use Disorder: A Brief Guide. HHS Publication No. (SMA) 15-4907. Rockville, MD: Substance Abuse and Mental Health Services Administration; 2015.
11. Substance Abuse and Mental Health Services Administration and National Institute on Alcohol Abuse and Alcoholism. Medication for Opioid Use Disorder – Full Document. HHS Publication No. (SMA) 18-5063FULLDOC. Rockville, MD: Substance Abuse and Mental Health Services Administration; 2018.
12. Winn JL, Shealy SE, Kropp GJ, Felkins-Dohm D, Gonzales-Nolas C, Francis E. Housing assistance and case management: Improving access to substance use disorder treatment for homeless veterans. Psychological Serv. 2013;10(2):233-240.
13. Cacciola JS, Alterman AI, DePhilippis D, et al. Development and initial evaluation of the Brief Addiction Monitor (BAM). J Subst Abuse Treatment. 2013;44(3):256-263.
14. McHugh RK, Hearon BA, Otto MW. Cognitive-behavioral therapy for substance use disorders, Psychiatr Clinics North Am. 2010;33:511–525.
15. Karlin, BE, Cross, G. From the laboratory to the therapy room: national dissemination and implementation of evidence-based psychotherapies in the U.S. Department of Veterans Affairs health care system. Am Psychol. 2014;69:19-33.
16. DePhilippis D, Petry NM, Bonn-Miller MO, Rosenbach SB, McKay JR. The national implementation of contingency management (CM) in the Department of Veterans Affairs: attendance at CM sessions and substance use outcomes, Drug Alcohol Dependence. 2018;185:367-373.
1. Substance Abuse and Mental Health Services Administration. 2015 National Survey on Drug Use and Health: Summary of the Effects of the 2015 NSDUH Questionnaire Redesign: Implications for Data Users. https://www.samhsa.gov/data/sites/default/files/NSDUH-TrendBreak-2015.pdf. Published June 2016. Accessed June 12, 2019.
2. Substance Abuse and Mental Health Services Administration. Results from the 2013 National Survey on Drug Use and Health: Summary of National Findings. NSDUH Series H-48, HHS Publication No. (SMA) 14-4863. Rockville, MD: Substance Abuse and Mental Health Services Administration; 2014.
3. Donovan DM, Rosengren DB, Downey L, Cox GB, Sloan PDSKL. Attrition prevention with individuals awaiting publicly funded drug treatment. Addiction. 2001;96(8):1149-1160.
4. Hser Y, Maglione M, Polinsky ML, Anglin MD. Predicting treatment entry among treatment-seeking drug abusers. J Subst Abuse Treatment. 1997;15(3):213-220.
5. Stark MJ, Campbell BK, Brinkerhoff CV. “Hello, may we help you?” A study of attrition prevention at the time of the first phone contact with substance-abusing clients. Am J Drug Alcohol Abuse. 1990;16:67-76.
6. US Department of Veterans Affairs. Uniform Mental Health Services in VA Medical Centers and Clinics. VHA Handbook 1160.01. https://www.va.gov/vhapublications/ViewPublication.asp?pub_ID=1762. Updated November 2015. Accessed December 12, 2017.
7. Veterans Access, Choice and Accountability Act of 2014, 2 USC § 933.
8. DeMarce JM, Gnys M, Raffa SD, Karlin, BE. Cognitive Behavioral Therapy for Substance Use Disorders Among Veterans: Therapist Manual. Washington, DC: US Department of Veterans Affairs; 2014.
9. Mee-Lee D, Shulman GD, Fishman MJ, Gastfriend DR, Miller MM, eds. The ASAM Criteria: Treatment Criteria for Addictive, Substance-Related, and Co-Occurring Conditions. 3rd ed. Carson City, NV: The Change Companies; 2013.
10. Substance Abuse and Mental Health Services Administration and National Institute on Alcohol Abuse and Alcoholism. Medication for the Treatment of Alcohol Use Disorder: A Brief Guide. HHS Publication No. (SMA) 15-4907. Rockville, MD: Substance Abuse and Mental Health Services Administration; 2015.
11. Substance Abuse and Mental Health Services Administration and National Institute on Alcohol Abuse and Alcoholism. Medication for Opioid Use Disorder – Full Document. HHS Publication No. (SMA) 18-5063FULLDOC. Rockville, MD: Substance Abuse and Mental Health Services Administration; 2018.
12. Winn JL, Shealy SE, Kropp GJ, Felkins-Dohm D, Gonzales-Nolas C, Francis E. Housing assistance and case management: Improving access to substance use disorder treatment for homeless veterans. Psychological Serv. 2013;10(2):233-240.
13. Cacciola JS, Alterman AI, DePhilippis D, et al. Development and initial evaluation of the Brief Addiction Monitor (BAM). J Subst Abuse Treatment. 2013;44(3):256-263.
14. McHugh RK, Hearon BA, Otto MW. Cognitive-behavioral therapy for substance use disorders, Psychiatr Clinics North Am. 2010;33:511–525.
15. Karlin, BE, Cross, G. From the laboratory to the therapy room: national dissemination and implementation of evidence-based psychotherapies in the U.S. Department of Veterans Affairs health care system. Am Psychol. 2014;69:19-33.
16. DePhilippis D, Petry NM, Bonn-Miller MO, Rosenbach SB, McKay JR. The national implementation of contingency management (CM) in the Department of Veterans Affairs: attendance at CM sessions and substance use outcomes, Drug Alcohol Dependence. 2018;185:367-373.
Development of a Program to Support VA Community Living Centers’ Quality Improvement
US Department of Veterans Affairs (VA) Community Living Centers (CLCs) provide a dynamic array of long- and short-term health and rehabilitative services in a person-centered environment designed to meet the individual needs of veteran residents. The VA Office of Geriatrics and Extended Care (GEC) manages CLCs as part of its commitment to “optimizing the health and well-being of veterans with multiple chronic conditions, life-limiting illness, frailty or disability associated with chronic disease, aging or injury.”1
CLCs are home to veterans who require short stays before going home, as well as those who require longer or permanent domicile. CLCs also are home to several special populations of veterans, including those with spinal cord injury and those who choose palliative or hospice care. CLCs have embraced cultural transformation, creating therapeutic environments that function as real homes, with the kitchen at the center, and daily activities scheduled around the veterans’ preferences. Data about CLC quality are now available to the public, highlighting the important role of support for and continual refinement to quality improvement (QI) processes in the CLC system. 2,3
CONCERT Program
High-functioning teams are critical to achieving improvement in such processes.4 In fiscal year (FY) 2017, GEC launched a national center to engage and support CLC staff in creating high-functioning, relationship-based teams through specific QI practices, thereby aiming to improve veteran experience and quality of care. The center, known as the CLCs’ Ongoing National Center for Enhancing Resources and Training (CONCERT), is based on extensive VA-funded research in CLCs5-7 and builds on existing, evidence-based literature emphasizing the importance of strengths-based learning, collaborative problem solving, and structured observation.8-13 The CONCERT mission is to support CLCs in ongoing QI efforts, providing guidance, training, and resources. This article summarizes the previous research on which CONCERT is based and describes its current activities, which focus on implementing a national team-based quality improvement initiative.
Earlier VA-funded CLC research included a VA Office of Patient Centered Care and Cultural Transformation local innovation project and 2 VA Office of Research and Development-funded research studies. The local innovation project focused on strengthening staff leadership and relational skills in 1 CLC by engaging leaders and staff in collaborative work to reduce stress. The goal was to build high-functioning team skills through shared projects that created positive work experiences and reduced job-related stress while also improving veteran experience and quality of care.14,15 Over the course of a year, 2 national consultants in nursing home quality improvement worked with CLC leadership and staff, including conducting nine 4-day site visits. Using an approach designed to foster development of high-functioning teams, individual CLC neighborhoods (ie, units) developed and implemented neighborhood-initiated, neighborhood-based pilot projects, such as an individualized finger foods dining option for residents with dementia who became distressed when sitting at a table during a meal. Outcomes of these projects included improved staff communication and staff satisfaction, particularly psychological safety.
In the concurrently conducted pilot research study, a research team comprehensively assessed the person-centered care efforts of 3 CLCs prior to their construction of Green House-type (small house) homes. This mixed-methods study included more than 50 qualitative interviews conducted with VA medical center leadership and CLC staff and residents. Researchers also administered online employee surveys and conducted site visits, including more than 60 hours of direct observation of CLC life and team functioning. The local institutional review boards approved all study procedures, and researchers notified local unions.
Analyses highlighted 2 important aspects of person-centered care not captured by then-existing measurement instruments: the type, quality, and number of staff/resident interactions and the type, quality, and level of resident engagement. The team therefore developed a structured, systematic, observation-based instrument to measure these concepts.5 But while researchers found this instrument useful, it was too complex to be used by CLC staff for QI.
LOCK Quality Improvement
A later and larger research study addressed this issue. In the study, researchers worked with CLC staff to convert the complex observation-based research instrument into several structured tools that were easier for CLC staff to use.6 The researchers then incorporated their experience with the prior local innovation project and designed and implemented a QI program, which operationalized an evidence-based bundle of practices to implement the new tools in 6 CLCs. Researchers called the bundle of practices “LOCK”: (1) Learn from the bright spots; (2) Observe; (3) Collaborate in huddles; and (4) Keep it bite-sized.
Learn from the bright spots. Studies on strengths-based learning indicate that recognizing and sharing positive instances of ideal practice helps provide clear direction regarding what needs to be done differently to achieve success. Identifying and learning from outlying instances of successful practice encourages staff to continue those behaviors and gives staff tangible examples of how they may improve.16-19 That is, concentrating on instances where a negative outcome was at risk of occurring but did not occur (ie, a positive outlier or “bright spot”) enables staff to analyze what facilitated the success and design and pilot strategies to replicate it.
Observe. Human factors engineering is built on the principle that integrated approaches for studying work systems can identify areas for improvement.8 Observation is a key tool in this approach. A recent review of 69 studies that used observation to assess clinical performance found it useful in identifying factors affecting quality and safety.9
Collaborate in huddles. A necessary component to overcoming barriers to successful QI is having high-functioning teams effectively coordinate work. In the theory of relational coordination, this is operationalized as high-quality interactions (frequent, timely, and accurate communication) and high-quality relationships (share knowledge, shared goals, and mutual respect).10,11 Improved relational coordination can lead to higher quality of care outcomes and job satisfaction by enabling individuals to manage their tasks with less delay, more rapid and effective responses, fewer errors, and less wasted effort.12
Keep it bite-sized. Regular practice of a new behavior is one of the keys to making that new behavior part of an automatic routine (ie, a habit). To be successfully integrated into staff work routines, QI initiatives must be perceived as congruent with and easily integrated into care goals and workplace practices. Quick, focused, team-building and solution-oriented QI initiatives, therefore, have the greatest chance of success, particularly if staff feel they have little time for participating in new initiatives.13
Researchers designed the 4 LOCK practices to be interrelated and build on one another, creating a bundle to be used together to help facilitate positive change in resident/staff interactions and resident engagement.7 For 6 months, researchers studied the 6 CLCs’ use of the new structured observation tools as part of the LOCK-based QI program. The participating CLCs had such success in improving staff interactions with residents and residents’ engagement in CLC life that GEC, under the CONCERT umbrella, rolled out the LOCK bundle of practices to CLCs nationwide.20
CONCERT’s current activities focus on helping CLCs implement the LOCK bundle nationwide as a relational coordination-based national QI initiative designed to improve quality of care and staff satisfaction. The CONCERT team began this implementation in FY 2017 using a train-the-trainer approach through a staggered veterans integrated service network (VISN) rollout. Each CLC sent 2 leaders to a VISN-wide training program at a host CLC site (the host site was able to have more participants attend). Afterward, the CONCERT team provided individualized phone support to help CLCs implement the program. A VA Pulse (intranet-based social media portal) site hosts all training materials, program videos, an active blog, community discussions, etc.
In FY 2018, the program shifted to a VISN-based support system, with a CONCERT team member assigned to each VISN and VISN-based webinars to facilitate information exchange, collaboration, and group learning. In FY 2018, the CONCERT team also conducted site visits to selected CLCs with strong implementation success records to learn about program facilitators and to disseminate the lessons learned. Spanning FYs 2018 and 2019, the CONCERT team also supports historically low-performing CLCs through a series of rapid-cycle learning intensives based on the Institute for Healthcare Improvement breakthrough collaborative series model for accelerated and sustained QI.21 These incorporate in-person or virtual learning sessions, in which participants learn about and share effective practices, and between-session learning assignments, to facilitate the piloting, implementation, and sustainment of system changes. As part of the CONCERT continuous QI process, the CONCERT team closely monitors the impact of the program and continues to pilot, adapt, and change practices as it learns more about how best to help CLCs improve.
Conclusion
A key CONCERT principle is that health care systems create health care outcomes. The CONCERT team uses the theory of relational coordination to support implementation of the LOCK bundle of practices to help CLCs change their systems to achieve high performance. Through implementation of the LOCK bundle of practices, CLC staff develop, pilot, and spread new systems for communication, teamwork, and collaborative problem solving, as well as developing skills to participate effectively in these systems. CONCERT represents just 1 way VA supports CLCs in their continual journeys toward ever-improved quality of veteran care.
Acknowledgments
The authors thank Barbara Frank and Cathie Brady for their contributions to the development of the CONCERT program.
1. US Department of Veterans Affairs, Geriatrics and Extended Care Services (GEC). https://www.va.gov/GERIATRICS/index.asp. Updated February 25, 2019. Accessed April 9, 2019.
2. US Department of Veterans Affairs. https://www.accesstocare.va.gov/CNH/Statemap. Accessed April 10, 2019.
3. US Department of Veterans Affairs. https://www.va.gov/QUALITYOFCARE/apps/aspire/clcsurvey.aspx/. U
4. Gittell JH, Weinberg D, Pfefferle S, Bishop C. Impact of relational coordination on job satisfaction and quality outcomes: a study of nursing homes. Hum Resour Manag. 2008;18(2):154-170
5. Snow AL, Dodson, ML, Palmer JA, et al. Development of a new systematic observation tool of nursing home resident and staff engagement and relationship. Gerontologist. 2018;58(2):e15-e24.
6. Hartmann CW, Palmer JA, Mills WL, et al. Adaptation of a nursing home culture change research instrument for frontline staff quality improvement use. Psychol Serv. 2017;14(3):337-346.
7. Mills WL, Pimentel CB, Palmer JA, et al. Applying a theory-driven framework to guide quality improvement efforts in nursing homes: the LOCK model. Gerontologist. 2018;58(3):598-605.
8. Caravon P, Hundt AS, Karsh B, et al. Work system design for patient safety: the SEIPS model. Quality & Safety in Health Care. 2006;15(suppl 1), i50-i58.
9. Yanes AF, McElroy LM, Abecassis ZA, Holl J, Woods D, Ladner DP. Observation for assessment of clinician performance: a narrative review. BMJ Qual Saf. 2016;25(1):46-55.
10. Gittell JH. Supervisory span, relational coordination and flight departure performance: a reassessment of postbureaucracy theory. Organ Sci. 2011;12(4):468-483.
11. Gittell JH. New Directions for Relational Coordination Theory. In Spreitzer GM, Cameron KS, eds. The Oxford Handbook of Positive Organizational Scholarship. Oxford University Press: New York; 2012:400-411.
12. Weinberg DB, Lusenhop RW, Gittell JH, Kautz CM. Coordination between formal providers and informal caregivers. Health Care Manage Rev. 2007;32(2):140-149.
13. Phillips J, Hebish LJ, Mann S, Ching JM, Blackmore CC. Engaging frontline leaders and staff in real-time improvement. Jt Comm J Qual Patient Saf. 2016;42(4):170-183.
14. Farrell D, Brady C, Frank B. Meeting the Leadership Challenge in Long-Term Care: What You Do Matters. Health Professions Press: Baltimore, MD; 2011.
15. Brady C, Farrell D, Frank B. A Long-Term Leaders’ Guide to High Performance: Doing Better Together. Health Professions Press: Baltimore, MD; 2018.
16. Bradley EH, Curry LA, Ramanadhan S, Rowe L, Nembhard IM, Krumholz HM. Research in action: using positive deviance to improve quality of health care. Implement Sci. 2009;4:25.
17. Marsh DR, Schroeder DG, Dearden KA, Sternin J, Sternin M. The power of positive deviance. BMJ. 2004; 329(7475):1177-1179.
18. Vogt K, Johnson F, Fraser V, et al. An innovative, strengths-based, peer mentoring approach to professional development for registered dietitians. Can J Diet Pract Res. 2015;76(4):185-189.
19. Beckett P, Field J, Molloy L, Yu N, Holmes D, Pile E. Practice what you preach: developing person-centered culture in inpatient mental health settings through strengths-based, transformational leadership. Issues Ment Health Nurs. 2013;34(8):595-601.
20. Hartmann CW, Mills WL, Pimentel CB, et al. Impact of intervention to improve nursing home resident-staff interactions and engagement. Gerontologist. 2018;58(4):e291-e301.
21. Institute for Healthcare Improvement. The breakthrough series: IHI’s collaborative model for achieving breakthrough improvement. http://www.ihi.org/resources/Pages/IHIWhitePapers/TheBreakthroughSeriesIHIsCollaborativeModelforAchievingBreakthroughImprovement.aspx. Published 2003. Accessed April 9, 2019.
US Department of Veterans Affairs (VA) Community Living Centers (CLCs) provide a dynamic array of long- and short-term health and rehabilitative services in a person-centered environment designed to meet the individual needs of veteran residents. The VA Office of Geriatrics and Extended Care (GEC) manages CLCs as part of its commitment to “optimizing the health and well-being of veterans with multiple chronic conditions, life-limiting illness, frailty or disability associated with chronic disease, aging or injury.”1
CLCs are home to veterans who require short stays before going home, as well as those who require longer or permanent domicile. CLCs also are home to several special populations of veterans, including those with spinal cord injury and those who choose palliative or hospice care. CLCs have embraced cultural transformation, creating therapeutic environments that function as real homes, with the kitchen at the center, and daily activities scheduled around the veterans’ preferences. Data about CLC quality are now available to the public, highlighting the important role of support for and continual refinement to quality improvement (QI) processes in the CLC system. 2,3
CONCERT Program
High-functioning teams are critical to achieving improvement in such processes.4 In fiscal year (FY) 2017, GEC launched a national center to engage and support CLC staff in creating high-functioning, relationship-based teams through specific QI practices, thereby aiming to improve veteran experience and quality of care. The center, known as the CLCs’ Ongoing National Center for Enhancing Resources and Training (CONCERT), is based on extensive VA-funded research in CLCs5-7 and builds on existing, evidence-based literature emphasizing the importance of strengths-based learning, collaborative problem solving, and structured observation.8-13 The CONCERT mission is to support CLCs in ongoing QI efforts, providing guidance, training, and resources. This article summarizes the previous research on which CONCERT is based and describes its current activities, which focus on implementing a national team-based quality improvement initiative.
Earlier VA-funded CLC research included a VA Office of Patient Centered Care and Cultural Transformation local innovation project and 2 VA Office of Research and Development-funded research studies. The local innovation project focused on strengthening staff leadership and relational skills in 1 CLC by engaging leaders and staff in collaborative work to reduce stress. The goal was to build high-functioning team skills through shared projects that created positive work experiences and reduced job-related stress while also improving veteran experience and quality of care.14,15 Over the course of a year, 2 national consultants in nursing home quality improvement worked with CLC leadership and staff, including conducting nine 4-day site visits. Using an approach designed to foster development of high-functioning teams, individual CLC neighborhoods (ie, units) developed and implemented neighborhood-initiated, neighborhood-based pilot projects, such as an individualized finger foods dining option for residents with dementia who became distressed when sitting at a table during a meal. Outcomes of these projects included improved staff communication and staff satisfaction, particularly psychological safety.
In the concurrently conducted pilot research study, a research team comprehensively assessed the person-centered care efforts of 3 CLCs prior to their construction of Green House-type (small house) homes. This mixed-methods study included more than 50 qualitative interviews conducted with VA medical center leadership and CLC staff and residents. Researchers also administered online employee surveys and conducted site visits, including more than 60 hours of direct observation of CLC life and team functioning. The local institutional review boards approved all study procedures, and researchers notified local unions.
Analyses highlighted 2 important aspects of person-centered care not captured by then-existing measurement instruments: the type, quality, and number of staff/resident interactions and the type, quality, and level of resident engagement. The team therefore developed a structured, systematic, observation-based instrument to measure these concepts.5 But while researchers found this instrument useful, it was too complex to be used by CLC staff for QI.
LOCK Quality Improvement
A later and larger research study addressed this issue. In the study, researchers worked with CLC staff to convert the complex observation-based research instrument into several structured tools that were easier for CLC staff to use.6 The researchers then incorporated their experience with the prior local innovation project and designed and implemented a QI program, which operationalized an evidence-based bundle of practices to implement the new tools in 6 CLCs. Researchers called the bundle of practices “LOCK”: (1) Learn from the bright spots; (2) Observe; (3) Collaborate in huddles; and (4) Keep it bite-sized.
Learn from the bright spots. Studies on strengths-based learning indicate that recognizing and sharing positive instances of ideal practice helps provide clear direction regarding what needs to be done differently to achieve success. Identifying and learning from outlying instances of successful practice encourages staff to continue those behaviors and gives staff tangible examples of how they may improve.16-19 That is, concentrating on instances where a negative outcome was at risk of occurring but did not occur (ie, a positive outlier or “bright spot”) enables staff to analyze what facilitated the success and design and pilot strategies to replicate it.
Observe. Human factors engineering is built on the principle that integrated approaches for studying work systems can identify areas for improvement.8 Observation is a key tool in this approach. A recent review of 69 studies that used observation to assess clinical performance found it useful in identifying factors affecting quality and safety.9
Collaborate in huddles. A necessary component to overcoming barriers to successful QI is having high-functioning teams effectively coordinate work. In the theory of relational coordination, this is operationalized as high-quality interactions (frequent, timely, and accurate communication) and high-quality relationships (share knowledge, shared goals, and mutual respect).10,11 Improved relational coordination can lead to higher quality of care outcomes and job satisfaction by enabling individuals to manage their tasks with less delay, more rapid and effective responses, fewer errors, and less wasted effort.12
Keep it bite-sized. Regular practice of a new behavior is one of the keys to making that new behavior part of an automatic routine (ie, a habit). To be successfully integrated into staff work routines, QI initiatives must be perceived as congruent with and easily integrated into care goals and workplace practices. Quick, focused, team-building and solution-oriented QI initiatives, therefore, have the greatest chance of success, particularly if staff feel they have little time for participating in new initiatives.13
Researchers designed the 4 LOCK practices to be interrelated and build on one another, creating a bundle to be used together to help facilitate positive change in resident/staff interactions and resident engagement.7 For 6 months, researchers studied the 6 CLCs’ use of the new structured observation tools as part of the LOCK-based QI program. The participating CLCs had such success in improving staff interactions with residents and residents’ engagement in CLC life that GEC, under the CONCERT umbrella, rolled out the LOCK bundle of practices to CLCs nationwide.20
CONCERT’s current activities focus on helping CLCs implement the LOCK bundle nationwide as a relational coordination-based national QI initiative designed to improve quality of care and staff satisfaction. The CONCERT team began this implementation in FY 2017 using a train-the-trainer approach through a staggered veterans integrated service network (VISN) rollout. Each CLC sent 2 leaders to a VISN-wide training program at a host CLC site (the host site was able to have more participants attend). Afterward, the CONCERT team provided individualized phone support to help CLCs implement the program. A VA Pulse (intranet-based social media portal) site hosts all training materials, program videos, an active blog, community discussions, etc.
In FY 2018, the program shifted to a VISN-based support system, with a CONCERT team member assigned to each VISN and VISN-based webinars to facilitate information exchange, collaboration, and group learning. In FY 2018, the CONCERT team also conducted site visits to selected CLCs with strong implementation success records to learn about program facilitators and to disseminate the lessons learned. Spanning FYs 2018 and 2019, the CONCERT team also supports historically low-performing CLCs through a series of rapid-cycle learning intensives based on the Institute for Healthcare Improvement breakthrough collaborative series model for accelerated and sustained QI.21 These incorporate in-person or virtual learning sessions, in which participants learn about and share effective practices, and between-session learning assignments, to facilitate the piloting, implementation, and sustainment of system changes. As part of the CONCERT continuous QI process, the CONCERT team closely monitors the impact of the program and continues to pilot, adapt, and change practices as it learns more about how best to help CLCs improve.
Conclusion
A key CONCERT principle is that health care systems create health care outcomes. The CONCERT team uses the theory of relational coordination to support implementation of the LOCK bundle of practices to help CLCs change their systems to achieve high performance. Through implementation of the LOCK bundle of practices, CLC staff develop, pilot, and spread new systems for communication, teamwork, and collaborative problem solving, as well as developing skills to participate effectively in these systems. CONCERT represents just 1 way VA supports CLCs in their continual journeys toward ever-improved quality of veteran care.
Acknowledgments
The authors thank Barbara Frank and Cathie Brady for their contributions to the development of the CONCERT program.
US Department of Veterans Affairs (VA) Community Living Centers (CLCs) provide a dynamic array of long- and short-term health and rehabilitative services in a person-centered environment designed to meet the individual needs of veteran residents. The VA Office of Geriatrics and Extended Care (GEC) manages CLCs as part of its commitment to “optimizing the health and well-being of veterans with multiple chronic conditions, life-limiting illness, frailty or disability associated with chronic disease, aging or injury.”1
CLCs are home to veterans who require short stays before going home, as well as those who require longer or permanent domicile. CLCs also are home to several special populations of veterans, including those with spinal cord injury and those who choose palliative or hospice care. CLCs have embraced cultural transformation, creating therapeutic environments that function as real homes, with the kitchen at the center, and daily activities scheduled around the veterans’ preferences. Data about CLC quality are now available to the public, highlighting the important role of support for and continual refinement to quality improvement (QI) processes in the CLC system. 2,3
CONCERT Program
High-functioning teams are critical to achieving improvement in such processes.4 In fiscal year (FY) 2017, GEC launched a national center to engage and support CLC staff in creating high-functioning, relationship-based teams through specific QI practices, thereby aiming to improve veteran experience and quality of care. The center, known as the CLCs’ Ongoing National Center for Enhancing Resources and Training (CONCERT), is based on extensive VA-funded research in CLCs5-7 and builds on existing, evidence-based literature emphasizing the importance of strengths-based learning, collaborative problem solving, and structured observation.8-13 The CONCERT mission is to support CLCs in ongoing QI efforts, providing guidance, training, and resources. This article summarizes the previous research on which CONCERT is based and describes its current activities, which focus on implementing a national team-based quality improvement initiative.
Earlier VA-funded CLC research included a VA Office of Patient Centered Care and Cultural Transformation local innovation project and 2 VA Office of Research and Development-funded research studies. The local innovation project focused on strengthening staff leadership and relational skills in 1 CLC by engaging leaders and staff in collaborative work to reduce stress. The goal was to build high-functioning team skills through shared projects that created positive work experiences and reduced job-related stress while also improving veteran experience and quality of care.14,15 Over the course of a year, 2 national consultants in nursing home quality improvement worked with CLC leadership and staff, including conducting nine 4-day site visits. Using an approach designed to foster development of high-functioning teams, individual CLC neighborhoods (ie, units) developed and implemented neighborhood-initiated, neighborhood-based pilot projects, such as an individualized finger foods dining option for residents with dementia who became distressed when sitting at a table during a meal. Outcomes of these projects included improved staff communication and staff satisfaction, particularly psychological safety.
In the concurrently conducted pilot research study, a research team comprehensively assessed the person-centered care efforts of 3 CLCs prior to their construction of Green House-type (small house) homes. This mixed-methods study included more than 50 qualitative interviews conducted with VA medical center leadership and CLC staff and residents. Researchers also administered online employee surveys and conducted site visits, including more than 60 hours of direct observation of CLC life and team functioning. The local institutional review boards approved all study procedures, and researchers notified local unions.
Analyses highlighted 2 important aspects of person-centered care not captured by then-existing measurement instruments: the type, quality, and number of staff/resident interactions and the type, quality, and level of resident engagement. The team therefore developed a structured, systematic, observation-based instrument to measure these concepts.5 But while researchers found this instrument useful, it was too complex to be used by CLC staff for QI.
LOCK Quality Improvement
A later and larger research study addressed this issue. In the study, researchers worked with CLC staff to convert the complex observation-based research instrument into several structured tools that were easier for CLC staff to use.6 The researchers then incorporated their experience with the prior local innovation project and designed and implemented a QI program, which operationalized an evidence-based bundle of practices to implement the new tools in 6 CLCs. Researchers called the bundle of practices “LOCK”: (1) Learn from the bright spots; (2) Observe; (3) Collaborate in huddles; and (4) Keep it bite-sized.
Learn from the bright spots. Studies on strengths-based learning indicate that recognizing and sharing positive instances of ideal practice helps provide clear direction regarding what needs to be done differently to achieve success. Identifying and learning from outlying instances of successful practice encourages staff to continue those behaviors and gives staff tangible examples of how they may improve.16-19 That is, concentrating on instances where a negative outcome was at risk of occurring but did not occur (ie, a positive outlier or “bright spot”) enables staff to analyze what facilitated the success and design and pilot strategies to replicate it.
Observe. Human factors engineering is built on the principle that integrated approaches for studying work systems can identify areas for improvement.8 Observation is a key tool in this approach. A recent review of 69 studies that used observation to assess clinical performance found it useful in identifying factors affecting quality and safety.9
Collaborate in huddles. A necessary component to overcoming barriers to successful QI is having high-functioning teams effectively coordinate work. In the theory of relational coordination, this is operationalized as high-quality interactions (frequent, timely, and accurate communication) and high-quality relationships (share knowledge, shared goals, and mutual respect).10,11 Improved relational coordination can lead to higher quality of care outcomes and job satisfaction by enabling individuals to manage their tasks with less delay, more rapid and effective responses, fewer errors, and less wasted effort.12
Keep it bite-sized. Regular practice of a new behavior is one of the keys to making that new behavior part of an automatic routine (ie, a habit). To be successfully integrated into staff work routines, QI initiatives must be perceived as congruent with and easily integrated into care goals and workplace practices. Quick, focused, team-building and solution-oriented QI initiatives, therefore, have the greatest chance of success, particularly if staff feel they have little time for participating in new initiatives.13
Researchers designed the 4 LOCK practices to be interrelated and build on one another, creating a bundle to be used together to help facilitate positive change in resident/staff interactions and resident engagement.7 For 6 months, researchers studied the 6 CLCs’ use of the new structured observation tools as part of the LOCK-based QI program. The participating CLCs had such success in improving staff interactions with residents and residents’ engagement in CLC life that GEC, under the CONCERT umbrella, rolled out the LOCK bundle of practices to CLCs nationwide.20
CONCERT’s current activities focus on helping CLCs implement the LOCK bundle nationwide as a relational coordination-based national QI initiative designed to improve quality of care and staff satisfaction. The CONCERT team began this implementation in FY 2017 using a train-the-trainer approach through a staggered veterans integrated service network (VISN) rollout. Each CLC sent 2 leaders to a VISN-wide training program at a host CLC site (the host site was able to have more participants attend). Afterward, the CONCERT team provided individualized phone support to help CLCs implement the program. A VA Pulse (intranet-based social media portal) site hosts all training materials, program videos, an active blog, community discussions, etc.
In FY 2018, the program shifted to a VISN-based support system, with a CONCERT team member assigned to each VISN and VISN-based webinars to facilitate information exchange, collaboration, and group learning. In FY 2018, the CONCERT team also conducted site visits to selected CLCs with strong implementation success records to learn about program facilitators and to disseminate the lessons learned. Spanning FYs 2018 and 2019, the CONCERT team also supports historically low-performing CLCs through a series of rapid-cycle learning intensives based on the Institute for Healthcare Improvement breakthrough collaborative series model for accelerated and sustained QI.21 These incorporate in-person or virtual learning sessions, in which participants learn about and share effective practices, and between-session learning assignments, to facilitate the piloting, implementation, and sustainment of system changes. As part of the CONCERT continuous QI process, the CONCERT team closely monitors the impact of the program and continues to pilot, adapt, and change practices as it learns more about how best to help CLCs improve.
Conclusion
A key CONCERT principle is that health care systems create health care outcomes. The CONCERT team uses the theory of relational coordination to support implementation of the LOCK bundle of practices to help CLCs change their systems to achieve high performance. Through implementation of the LOCK bundle of practices, CLC staff develop, pilot, and spread new systems for communication, teamwork, and collaborative problem solving, as well as developing skills to participate effectively in these systems. CONCERT represents just 1 way VA supports CLCs in their continual journeys toward ever-improved quality of veteran care.
Acknowledgments
The authors thank Barbara Frank and Cathie Brady for their contributions to the development of the CONCERT program.
1. US Department of Veterans Affairs, Geriatrics and Extended Care Services (GEC). https://www.va.gov/GERIATRICS/index.asp. Updated February 25, 2019. Accessed April 9, 2019.
2. US Department of Veterans Affairs. https://www.accesstocare.va.gov/CNH/Statemap. Accessed April 10, 2019.
3. US Department of Veterans Affairs. https://www.va.gov/QUALITYOFCARE/apps/aspire/clcsurvey.aspx/. U
4. Gittell JH, Weinberg D, Pfefferle S, Bishop C. Impact of relational coordination on job satisfaction and quality outcomes: a study of nursing homes. Hum Resour Manag. 2008;18(2):154-170
5. Snow AL, Dodson, ML, Palmer JA, et al. Development of a new systematic observation tool of nursing home resident and staff engagement and relationship. Gerontologist. 2018;58(2):e15-e24.
6. Hartmann CW, Palmer JA, Mills WL, et al. Adaptation of a nursing home culture change research instrument for frontline staff quality improvement use. Psychol Serv. 2017;14(3):337-346.
7. Mills WL, Pimentel CB, Palmer JA, et al. Applying a theory-driven framework to guide quality improvement efforts in nursing homes: the LOCK model. Gerontologist. 2018;58(3):598-605.
8. Caravon P, Hundt AS, Karsh B, et al. Work system design for patient safety: the SEIPS model. Quality & Safety in Health Care. 2006;15(suppl 1), i50-i58.
9. Yanes AF, McElroy LM, Abecassis ZA, Holl J, Woods D, Ladner DP. Observation for assessment of clinician performance: a narrative review. BMJ Qual Saf. 2016;25(1):46-55.
10. Gittell JH. Supervisory span, relational coordination and flight departure performance: a reassessment of postbureaucracy theory. Organ Sci. 2011;12(4):468-483.
11. Gittell JH. New Directions for Relational Coordination Theory. In Spreitzer GM, Cameron KS, eds. The Oxford Handbook of Positive Organizational Scholarship. Oxford University Press: New York; 2012:400-411.
12. Weinberg DB, Lusenhop RW, Gittell JH, Kautz CM. Coordination between formal providers and informal caregivers. Health Care Manage Rev. 2007;32(2):140-149.
13. Phillips J, Hebish LJ, Mann S, Ching JM, Blackmore CC. Engaging frontline leaders and staff in real-time improvement. Jt Comm J Qual Patient Saf. 2016;42(4):170-183.
14. Farrell D, Brady C, Frank B. Meeting the Leadership Challenge in Long-Term Care: What You Do Matters. Health Professions Press: Baltimore, MD; 2011.
15. Brady C, Farrell D, Frank B. A Long-Term Leaders’ Guide to High Performance: Doing Better Together. Health Professions Press: Baltimore, MD; 2018.
16. Bradley EH, Curry LA, Ramanadhan S, Rowe L, Nembhard IM, Krumholz HM. Research in action: using positive deviance to improve quality of health care. Implement Sci. 2009;4:25.
17. Marsh DR, Schroeder DG, Dearden KA, Sternin J, Sternin M. The power of positive deviance. BMJ. 2004; 329(7475):1177-1179.
18. Vogt K, Johnson F, Fraser V, et al. An innovative, strengths-based, peer mentoring approach to professional development for registered dietitians. Can J Diet Pract Res. 2015;76(4):185-189.
19. Beckett P, Field J, Molloy L, Yu N, Holmes D, Pile E. Practice what you preach: developing person-centered culture in inpatient mental health settings through strengths-based, transformational leadership. Issues Ment Health Nurs. 2013;34(8):595-601.
20. Hartmann CW, Mills WL, Pimentel CB, et al. Impact of intervention to improve nursing home resident-staff interactions and engagement. Gerontologist. 2018;58(4):e291-e301.
21. Institute for Healthcare Improvement. The breakthrough series: IHI’s collaborative model for achieving breakthrough improvement. http://www.ihi.org/resources/Pages/IHIWhitePapers/TheBreakthroughSeriesIHIsCollaborativeModelforAchievingBreakthroughImprovement.aspx. Published 2003. Accessed April 9, 2019.
1. US Department of Veterans Affairs, Geriatrics and Extended Care Services (GEC). https://www.va.gov/GERIATRICS/index.asp. Updated February 25, 2019. Accessed April 9, 2019.
2. US Department of Veterans Affairs. https://www.accesstocare.va.gov/CNH/Statemap. Accessed April 10, 2019.
3. US Department of Veterans Affairs. https://www.va.gov/QUALITYOFCARE/apps/aspire/clcsurvey.aspx/. U
4. Gittell JH, Weinberg D, Pfefferle S, Bishop C. Impact of relational coordination on job satisfaction and quality outcomes: a study of nursing homes. Hum Resour Manag. 2008;18(2):154-170
5. Snow AL, Dodson, ML, Palmer JA, et al. Development of a new systematic observation tool of nursing home resident and staff engagement and relationship. Gerontologist. 2018;58(2):e15-e24.
6. Hartmann CW, Palmer JA, Mills WL, et al. Adaptation of a nursing home culture change research instrument for frontline staff quality improvement use. Psychol Serv. 2017;14(3):337-346.
7. Mills WL, Pimentel CB, Palmer JA, et al. Applying a theory-driven framework to guide quality improvement efforts in nursing homes: the LOCK model. Gerontologist. 2018;58(3):598-605.
8. Caravon P, Hundt AS, Karsh B, et al. Work system design for patient safety: the SEIPS model. Quality & Safety in Health Care. 2006;15(suppl 1), i50-i58.
9. Yanes AF, McElroy LM, Abecassis ZA, Holl J, Woods D, Ladner DP. Observation for assessment of clinician performance: a narrative review. BMJ Qual Saf. 2016;25(1):46-55.
10. Gittell JH. Supervisory span, relational coordination and flight departure performance: a reassessment of postbureaucracy theory. Organ Sci. 2011;12(4):468-483.
11. Gittell JH. New Directions for Relational Coordination Theory. In Spreitzer GM, Cameron KS, eds. The Oxford Handbook of Positive Organizational Scholarship. Oxford University Press: New York; 2012:400-411.
12. Weinberg DB, Lusenhop RW, Gittell JH, Kautz CM. Coordination between formal providers and informal caregivers. Health Care Manage Rev. 2007;32(2):140-149.
13. Phillips J, Hebish LJ, Mann S, Ching JM, Blackmore CC. Engaging frontline leaders and staff in real-time improvement. Jt Comm J Qual Patient Saf. 2016;42(4):170-183.
14. Farrell D, Brady C, Frank B. Meeting the Leadership Challenge in Long-Term Care: What You Do Matters. Health Professions Press: Baltimore, MD; 2011.
15. Brady C, Farrell D, Frank B. A Long-Term Leaders’ Guide to High Performance: Doing Better Together. Health Professions Press: Baltimore, MD; 2018.
16. Bradley EH, Curry LA, Ramanadhan S, Rowe L, Nembhard IM, Krumholz HM. Research in action: using positive deviance to improve quality of health care. Implement Sci. 2009;4:25.
17. Marsh DR, Schroeder DG, Dearden KA, Sternin J, Sternin M. The power of positive deviance. BMJ. 2004; 329(7475):1177-1179.
18. Vogt K, Johnson F, Fraser V, et al. An innovative, strengths-based, peer mentoring approach to professional development for registered dietitians. Can J Diet Pract Res. 2015;76(4):185-189.
19. Beckett P, Field J, Molloy L, Yu N, Holmes D, Pile E. Practice what you preach: developing person-centered culture in inpatient mental health settings through strengths-based, transformational leadership. Issues Ment Health Nurs. 2013;34(8):595-601.
20. Hartmann CW, Mills WL, Pimentel CB, et al. Impact of intervention to improve nursing home resident-staff interactions and engagement. Gerontologist. 2018;58(4):e291-e301.
21. Institute for Healthcare Improvement. The breakthrough series: IHI’s collaborative model for achieving breakthrough improvement. http://www.ihi.org/resources/Pages/IHIWhitePapers/TheBreakthroughSeriesIHIsCollaborativeModelforAchievingBreakthroughImprovement.aspx. Published 2003. Accessed April 9, 2019.
Occupational Hazard: Disruptive Behavior in Patients
While private or other public health care organizations can refuse to care for patients who have displayed disruptive behavior (DB), the VA Response to Disruptive Behavior of Patients law (38 CFR §17.107) prohibits the Veterans Health Administration (VHA) of the Department of Veterans Affairs (VA) from refusing care to veterans who display DB.1 The VHA defines DB as any behavior that is intimidating, threatening, or dangerous or that has, or could, jeopardize the health or safety of patients, VHA staff, or others.2
VA Response to DB Law
The VA Response to Disruptive Behavior of Patients requires the VHA to provide alternative care options that minimize risk while ensuring services; for example, providing care at a different location and/or time when additional staff are available to assist and monitor the patient. This can provide a unique opportunity to capture data on DB and the results of alternative forms of caring for this population.
The reason public health care organizations refuse care to persons who display DB is clear: DBs hinder business operations, are financially taxing, and put health care workers at risk.3-10 “In 2009, the VHA spent close to $5.5 million on workers’ compensation and medical expenditures for 425 incidents–or about $130,000 per DB incident (Hodgson M, Drummond D, Van Male L. Unpublished data, 2010).” In another study, 106 of 762 nurses in 1 hospital system reported an assault by a patient, and 30 required medical attention, which resulted in a total cost of $94,156.8 From 2002 to 2013, incidents of serious workplace violence requiring days off for an injured worker to recover on average were 4 times more common in health care than in other industries.6-11 Incidents of patient violence and aggression toward staff transcend specialization; however, hospital nurses and staff from the emergency, rehabilitation and gerontology departments, psychiatric unit, and home-based services are more susceptible and vulnerable to DB incidents than are other types of employees.8,10-19
Data reported by health care staff suggest that patients rather than staff members or visitors initiate > 70% of serious physical attacks against health care workers.9,13,20-23 A 2015 study of VHA health care providers (HCPs) found that > 60% had experienced some form of DB, verbal abuse being the most prevalent, followed by sexual abuse and physical abuse.20 Of 72,000 VHA staff responding to a nationwide survey, 13% experienced, on average, ≥ 1 assault by a veteran (eg, something was thrown at them; they were pushed, kicked, slapped; or were threatened or injured by a weapon).8,21
To meet its legal obligations and deliver empathetic care, the VHA documents and analyzes data on all patients who exhibit DB. A local DB Committee (DBC) reviews the data, whether it occurs in an inpatient or outpatient setting, such as community-based outpatient clinics. Once a DB incident is reported, the DBC begins an evidence-based risk evaluation, including the option of contacting the persons who displayed or experienced the DB. Goals are to (1) prevent future DB incidents; (2) detect vulnerabilities in the environment; and (3) collaborate with HCPs and patients to provide optimal care while improving the patient/provider interactions.
Effects of Disruptive Behavior
DB has negative consequences for both patients and health care workers and results in poor evaluations of care from both groups.27-32 Aside from interfering with safe medical care, DB also impacts care for other patients by delaying access to care and increasing appointment wait times due to employee absenteeism and staff shortages.3,4,20,32,33 For HCPs, patient violence is associated with unwillingness to provide care, briefer treatment periods, and decreases in occupational satisfaction, performance, and commitment
Harmful health effects experienced by HCPs who have been victims of DB include fear, mood disorders, anxiety, all symptoms of psychological distress and posttraumatic stress disorder (PTSD).10,22,30,34-36 In a study of the impact on productivity of PTSD triggered by job-related DB, PTSD symptoms were associated with withdrawal from or minimizing encounters with patients, job turnover, and troubles with thinking
Reporting Disruptive Behavior
The literature suggests that consistent and effective DB reporting is pivotal to improving the outcome and quality of care for those displaying DB.37-39 To provide high-quality health services to veterans who display DB, the VHA must promote the management and reporting of DB. Without knowledge of the full spectrum of DB events at VHA facilities, efforts to prevent or manage DB and ensure safety may have limited impact.7,37 Reports can be used for clinical decision making to optimize staff training in delivery of quality care while assuring staff safety. More than 80% of DB incidents occur during interactions with patients, thus this is a clinical issue that can affect the outcome of patient care.8,21
Documented DB reports are used to analyze the degree, frequency, and nature of incidents, which might reveal risk factors and develop preventive efforts and training for specific hazards.8,39 Some have argued that implementing a standardized DB reporting system is a crucial first step toward minimizing hazards and improving health care.38,40,41
When DB incidents were recorded through a hospital electronic reporting system and discussed in meetings, staff reported: (1) increased awareness of DB; (2) improved ability to manage DB incidents; and (3) amplified reporting of incidents.38,41,42 These findings support similar results from studies of an intervention implemented at VA Community Living Centers (CLCs) from 2013 to 2017: Staff Training in Assisted Living Residences (STAR-VA).4,12,19 The aim of STAR-VA was to minimize challenging dementia-related DB in CLCs. The intervention initially was established to train direct-care, assisted-living staff to provide better care to older patients displaying DB. Data revealed that documentation of DBs was, the first step to ensuring staff and patient safety.18,40
VHA Reporting System
In 2013, the VA Office of Inspector General (OIG) found no standardized documentation of DB events across the VA health care system.42 Instead, DB events were documented in multiple records in various locations, including administrative and progress notes in the electronic health record (EHR), police reports, e-mails, or letters submitted to DBC chairs.42 This situation reduced administrators’ ability to consider all relevant information and render appropriate decisions in DB cases.42 In 2015, based on OIG recommendations, the VHA implemented the Disruptive Behavior Reporting System (DBRS) nationwide, which allowed all VHA staff to report DB events. The DBRS was designed to address factors likely to impede reporting and management of DB, namely, complexity of and lack of access to a central reporting system.43,44 The DBRS is currently the primary VHA tool to document DB events.
The DBRS consists of 32 questions in 5 sections relating to the (1) location and time of DB event; (2) reporter; (3) disrupter; (4) DB event details; and (5) the person who experienced (experiencer) the event. The system also provides a list of the types of DB, such as inappropriate communication, bullying and/or intimidation, verbal or written threat of physical harm, physical violence, sexual harassment, sexual assault, and property damage. The DBRS has the potential to provide useful data on DB and DB reporting, such as the typical staff entering data and the number and/or types of DB occurring.
The DBRS complements the preexisting VHA policies and committees for care of veterans who display DB.1-3,14,21,24,25 The VHA Workplace Violence Prevention Program (WVPP) required facilities to submit data on DB events through a Workplace Behavioral Risk report. Data for the report were obtained from police reports, patient safety reports, DBC records, and notes in the EHR. Following implementations of DBRS, the number of DB events per year became a part of facility performance standards.
VHA is creating novel approaches to handling DB that allow health care workers to render care in a safe and effective manner guided by documented information. For example, DBCs can recommend the use of Category I Patient Record Flags (PRFs) following documented DB, which informs staff of the potential risk of DB and provides guidance on protective methods to use when meeting with the patient.2,21,24 A survey of 140 VA hospital chiefs of staff indicated that DBC procedures were related to a decrease in the rates of assaults.1 Additionally, VA provides training for staff in techniques to promote personal safety, such as identifying signs that precede DB, using verbal deescalation, and practicing therapeutic containment.
Resistance to Reporting
Many health care employees and employers are reticent to report DBs.22,31,43,45-48 Studies suggest health care organizations can cultivate a culture that is resistant to reporting DB.49,50 This complicates the ability of the health care system to design and maintain safety protocols and safer treatment plans.3,41,51 Worldwide, < 30% of DBs are reported.47 One barrier may be that supervisors may not wish to acknowledge DBs on their units or may not provide sufficient staff time for training or reporting.31,46,47 HCPs may worry that a DB report will stigmatize patients, especially those who are elderly or have cognitive impairment, brain injury, psychological illness, or developmental disability. Patients with cognitive conditions are reportedly 20% more likely to be violent toward caregivers and providers.31 A dementia diagnosis, for example, is associated with a high likelihood for DB.30,52 More than 80% of DB events displayed by patients with dementia may go unreported.26,31,50,52
Some clinicians may attribute DB to physiologic conditions that need to be treated, not reported. However, employers can face various legal liabilities if steps are not taken to protect employees.47,51 Federal and state statutes require that organizations provide a healthy and safe employment environment for workers. This requires that employers institute reasonable protective measures, such as procedures to intervene, policies on addressing DB incidents, and/or training to minimize or deescalate DB.51,53 Also, employees may sue employers if security measures are inadequate or deficient in properly investigating current and past evidence of DB or identifying vulnerabilities in the workplace. Unwillingness to investigate DB and safety-related workplace concerns have contributed to increased workplace violence and legal liability.52,53 The mission of caring and trust is consistent with assuring a safe environment.
Training and Empathetic Care
To combat cultural resistance to reporting DBs, more and perhaps different contextual approaches to education and training may be needed that address ethical dilemmas and concerns of providers. The success of training relies on administrators supporting staff in reporting DB. Training must address providers’ conflicting beliefs and assist with identifying strategies to provide the best possible care for patients who display DB.1,38 HCPs are less likely to document a DB if they feel that administrators are creating documentation that will have negative consequences for a patient. Thus, leadership is responsible for ensuring that misconceptions are dispelled through training and other efforts and information on how reported DB data will be used is communicated through strategic channels.
Education and training must consider empathic care that attempts to understand why patients behave as they do through the information gathered.55 Empathy in health care is multifaceted: It involves comprehending a patient’s viewpoint, circumstances, and feelings and the capacity to analyze whether one is comprehending these accurately in order to demonstrate supportive care.54,55
Improving patient and staff interaction once a problematic behavior is identified is the aim of empathic care. Increasing empathic care can improve compassionate, patient-centered interactions that begin once the patient seeks care. This approach has proven to decrease DB by patients with dementia and improve their care, lessen staff problems during interactions, and increase staff morale.20 Experts call for the adoption of an interpersonal approach to patient encounters, and there is evidence that creating organizational change by moving toward compassionate care can lead to a positive impact for patients.54,55
Future Studies
There are growth opportunities in utilization of the DBRS. Analysis of the DBRS database by the VA Central Office (VACO) showed that the system is underutilized by facilities across the VA system.56 In response to this current underutilization, VACO is taking steps to close these gaps through increasing training to staff and promotion of the use of the DBRS. A 2015 pilot study of VHA providers showed that > 70% of providers had experienced a DB as defined by VHA, but only 34% of them reported their most recently experienced DB within the past 12 months.20 Thus, DBRS use must be studied within the context that patient-perpetrated DB is underreported in health care organizations.5,9,29,41,43,57,58 Studies addressing national DBRS utilization patterns and the cost associated with implementing the DBRS also are needed. One study suggests that there is an association between measures of facility complexity and staff perceptions of safety, which should be considered in analyzing DBRS usage.57 Studies addressing the role of the DBRS and misconceptions that the tool may represent a punitive tool also are needed. VHA should consider how the attribution “disruptive behavior” assigns a negative connotation and leads HCPs to avoid using the DBRS. Additionally, DB reporting may increase when HCPs understand that DB reporting is part of the comprehensive, consultative strategy to provide the best care to patients.
Conclusion
Accurate reporting of DB events enables the development of strategies for multidisciplinary teams to work together to minimize hazards and to provide interventions that provide for the safe delivery of health care to all patients. Improving reporting ensures there is an accurate representation of how disruptive events impact care provided within a facility—and what types of variables may be associated with increased risk for these types of events.
Additionally, ensuring that reporting is maximized also provides the VHA with opportunities for DBCs to offer evidence-based risk assessment of violence and consultation to staff members who may benefit from improved competencies in working with patients who display DB. These potential improvements are consistent with the VHA I CARE values and will provide data that can inform recommendations for health care in other agencies/health care organizations.
Acknowledgments
This work was supported by the Center of Innovation on Disability and Rehabilitation Research (CINDRR) of the Health Services Research and Development Service, Office of Research and Development, Department of Veterans Affairs.
1. Hodgson MJ, Mohr DC, Drummond DJ, Bell M, Van Male L. Managing disruptive patients in health care: necessary solutions to a difficult problem. Am J Ind Med. 2012;55(11):1009-1017.
2. US Department of Veterans Affairs, Veterans Health Administration. VHA Directive 2010-053. Patient Record Flags. https://www.va.gov/vhapublications/ViewPublication.asp?pub_ID=2341 Published December 3, 2010. Accessed March 29, 2019.
3. US Department of Veterans Affairs, Veterans Health Administration. VHA Directive 2012-026. Sexual Assaults and Other Defined Public Safety Incidents in VHA Facilities. https://www.va.gov/vhapublications/ViewPublication.asp?pub_ID=2797. Published September 27, 2012. Accessed March 29, 2019.
4. Curyto KJ, McCurry SM, Luci K, Karlin BE, Teri L, Karel MJ. Managing challenging behaviors of dementia in veterans: identifying and changing activators and consequences using STAR-VA. J Gerontol Nurs. 2017;43(2):33-43.
5. Speroni KG, Fitch T, Dawson E, Dugan L, Atherton M. Incidence and cost of nurse workplace violence perpetrated by hospital patients or patient visitors. J Emerg Nurs. 2014;40(3):218-228.
6. Phillips JP. Workplace violence against health care workers in the United States. NEJM. 2016;374(17):1661-1669.
7. Janocha JA, Smith RT. Workplace safety and health in the health care and social assistance industry, 2003–07. https://www.bls.gov/opub/mlr/cwc/workplace-safety-and-health-in-the-health-care-and-social-assistance-industry-2003-07.pdf. Published August 30, 2010. Accessed February 19, 2019.
8. US Department of Labor, Occupational Safety and Health Administration. Workplace violence in healthcare: understanding the challenge. https://www.osha.gov/Publications/OSHA3826.pdf. Published December 2015. Accessed February 19, 2019.
9. US Department of Labor, Occupational Safety and Health Administration. Prevention of Workplace Violence in Healthcare and Social Assistance. Occupational Safety and Health Administration, https://www.govinfo.gov/content/pkg/FR-2016-12-07/pdf/2016-29197.pdf. Accessed January 20, 2017.
10. Gerberich SG, Church TR, McGovern PM, et al. An epidemiological study of the magnitude and consequences of work related violence: the Minnesota Nurses’ Study. Occup Environ Med. 2004;61(6):495-503.
11. Sherman MF, Gershon RRM, Samar SM, Pearson JM, Canton AN, Damsky MR. Safety factors predictive of job satisfaction and job retention among home healthcare aides. J Occup Environ Med. 2008;50(12):1430-1441.
12. Karel MJ, Teri L, McConnell E, Visnic S, Karlin BE. Effectiveness of expanded implementation of STAR-VA for managing dementia-related behaviors among veterans. Gerontologist. 2016;56(1):126-134.
13. US Department of Labor, Bureau of Labor Statistics. Nonfatal occupational injuries and illnesses requiring days away from work. https://www.bls.gov/news.release/archives/osh2_11192015.htm. Published November 19, 2015.
14. Beech B, Leather P. Workplace violence in the health care sector: A review of staff training and integration of training evaluation models. Aggression Violent Behav. 2006;11(1):27-43.
15. Campbell CL, McCoy S, Burg MA, Hoffman N. Enhancing home care staff safety through reducing client aggression and violence in noninstitutional care settings: a systematic review. Home Health Care Manage Pract. 2014;26(1):3-10.
16. Gallant-Roman MA. Strategies and tools to reduce workplace violence. AAOHNJ. 2008;56(11):449-454.
17. Weinberger LE, Sreenivasan S, Smee DE, McGuire J, Garrick T. Balancing safety against obstruction to health care access: an examination of behavioral flags in the VA health care system. J Threat Assess Manage. 2018;5(1):35-41.
18. Elbogen EB, Johnson SC, Wagner HR, et al. Protective factors and risk modification of violence in Iraq and Afghanistan war veterans. J Clin Psychiatry. 2012;73(6):e767-e773.
19. Karlin BE, Visnic S, McGee JS, Teri L. Results from the multisite implementation of STAR-VA: a multicomponent psychosocial intervention for managing challenging dementia-related behaviors of veterans. Psychol Serv. 2014;11(2):200-208.
20. Semeah LM, Campbell CL, Cowper DC, Peet AC. Serving our homeless veterans: patient perpetrated violence as a barrier to health care access. J Pub Nonprofit Aff. 2017;3(2):223-234.
21. Hodgson MJ, Reed R, Craig T, et al. Violence in healthcare facilities: lessons from the Veterans Health Administration. J Occup Environ Med. 2004;46(11):1158-1165.
22. Farrell GA, Bobrowski C, Bobrowski P. Scoping workplace aggression in nursing: findings from an Australian study. J Adv Nurs. 2006;55(6):778-787.
23. Barling J, Rogers AG, Kelloway EK. Behind closed doors: in-home workers’ experience of sexual harassment and workplace violence. J Occup Health Psychol. 2001;6(3):255-269.
24. Pompeii LA, Schoenfisch AL, Lipscomb HJ, Dement JM, Smith CD, Upadhyaya M. Physical assault, physical threat, and verbal abuse perpetrated against hospital workers by patients or visitors in six U.S. hospitals. Am J Ind Med. 2015;58(11):1194-1204.
25. Sippel LM, Mota NP, Kachadourian LK, et al. The burden of hostility in U.S. veterans: results from the National Health and Resilience in Veterans Study. Psychiatry Res. 2016;243(suppl C):421-430.
26. Campbell C. Patient Violence and Aggression in Non-Institutional Health Care Settings: Predictors of Reporting By Healthcare Providers [doctoral dissertation]. Orlando: University of Central Florida; 2016.
27. Galinsky T, Feng HA, Streit J, et al. Risk factors associated with patient assaults of home healthcare workers. Rehabil Nurs. 2010;35(5):206-215.
28. Campbell CL. Incident reporting by health-care workers in noninstitutional care settings. Trauma, Violence Abuse. 2017;18(4):445-456.
29. Arnetz JE, Arnetz BB. Violence towards health care staff and possible effects on the quality of patient care. Soc Sci Med. 2001;52(3):417-427.
30. Gates D, Fitzwater E, Succop P. Relationships of stressors, strain, and anger to caregiver assaults. Issues Ment Health Nurs. 2003;24(8):775-793.
31. Brillhart B, Kruse B, Heard L. Safety concerns for rehabilitation nurses in home care. Rehabil Nurs. 2004;29(6):227-229.
32. Taylor H. Patient violence against clinicians: managing the risk. Innov Clin Neurosci. 2013;10(3):40-42.
33. US Department of Veterans Affairs, Office of Public and Intergovernmental Affairs. The Joint Commission releases results of surveys of the VA health care system. https://www.va.gov/opa/pressrel/pressrelease.cfm?id=2808. Updated August 5, 2014. Accessed February 19, 2019.
34. Büssing A, Höge T. Aggression and violence against home care workers. J Occup Health Psychol. 2004;9(3):206-219.
35. Geiger-Brown J, Muntaner C, McPhaul K, Lipscomb J, Trinkoff A. Abuse and violence during home care work as predictor of worker depression. Home Health Care Serv Q. 2007;26(1):59-77.
36. Gates DM, Gillespie GL, Succop P. Violence against nurses and its impact on stress and productivity. Nurs Econ. 2011;29(2):59-66.
37. Petterson IL, Arnetz BB. Psychosocial stressors and well-being in health care workers: the impact of an intervention program. Soc Sci Med. 1998;47(11):1763-1772.
38. Arnetz JE, Arnetz BB. Implementation and evaluation of a practical intervention programme for dealing with violence towards health care workers. J Adv Nurs. 2000;31(3):668-680.
39. Arnetz JE, Hamblin L, Russell J, et al. Preventing patient-to-worker violence in hospitals: outcome of a randomized controlled intervention. J Occup Environ Med. 2017;59(1):18-27.
40. Elbogen EB, Tomkins AJ, Pothuloori AP, Scalora MJ. Documentation of violence risk information in psychiatric hospital patient charts: an empirical examination. J Am Acad Psychiatry Law. 2003;31(1):58-64.
41. Winsvold Prang I, Jelson-Jorgensen LP. Should I report? A qualitative study of barriers to incident reporting among nurses working in nursing homes. Geriatr Nurs. 2014;35(6):441-447.
42. US Department of Veterans Affairs, Office of Inspector General. Healthcare inspection: management of disruptive patient behavior at VA medical facilities. Report No. 11-02585-129. https://www.va.gov/oig/pubs/VAOIG-11-02585-129.pdf. Published Mrach 7, 2013. Accessed February 21, 2019.
43. Lipscomb J, London M. Not Part of the Job: How to Take a Stand Against Violence in the Work Setting. Silver Spring, MD: American Nurses Association; 2015.
44. May DD, Grubbs LM. The extent, nature, and precipitating factors of nurse assault among three groups of registered nurses in a regional medical center. J Emerg Nurs. 2002;28(1):11-17.
45. Wharton TC, Ford BK. What is known about dementia care recipient violence and aggression against caregivers? J Gerontol Soc Work. 2014;57(5):460-477.
46. Brennan C, Worrall-Davies A, McMillan D, Gilbody S, House A. The hospital anxiety and depression scale: a diagnostic meta-analysis of case-finding ability. J Psychosom Res. 2010;69(4):371-378.
47. McPhaul K, Lipscomb J, Johnson J. Assessing risk for violence on home health visits. Home Healthc Nurse. 2010;28(5):278-289.
48. McPhaul KM, London M, Murrett K, Flannery K, Rosen J, Lipscomb J. Environmental evaluation for workplace violence in healthcare and social services. J Safety Res. 2008;39(2):237-250.
49. Kelly JA, Somlai AM, DiFranceisco WJ, et al. Bridging the gap between the science and service of HIV prevention: transferring effective research-based HIV prevention interventions to community AIDS service providers. Am J Public Health. 2000;90(7):1082-1088.
50. Pawlin S. Reporting violence. Emerg Nurse. 2008;16(4):16-21.
51. Brakel SJ. Legal liability and workplace violence. J Am Acad Psychiatry Law. 1998;26(4):553-562.
52. Neuman JH, Baron RA. Workplace violence and workplace aggression: evidence concerning specific forms, potential causes, and preferred targets. J Manage. 1998;24(3):391-419.53. Ferns T, Chojnacka I. Angels and swingers, matrons and sinners: nursing stereotypes. Br J Nurs. 2005;14(19):1028-1032.
54. Mercer SW, Reynolds WJ. Empathy and quality of care. Br J Gen Pract 2002;52(suppl):S9-S12.
55. Lee TH. An Epidemic of Empathy in Healthcare: How to Deliver Compassionate, Connected Patient Care That Creates a Competitive Advantage. Columbus, OH: McGraw-Hill Education; 2015.
56. US Department of Veterans Affairs, Veterans Health Administrastion. Veterans Health Administration workplace violence prevention program (WVPP): disruptive behavior reporting system utilization report. Published 2017. https://vaww.portal2.va.gov/sites/wvpp/Shared%20Documents/DBRS%20Utilization%20Reports/FY2017%20DBRS%20Quarterly%20Utilization%20Report%20(Quarter%201).pdf. [Source not verified.]
57. Campbell CL, Burg, MA, Gammonley D. Measures for incident reporting of patient violence and aggression towards healthcare providers: a systematic review. Aggression Violent Behav. 2015;25(part B):314-322.
58. Carney PT, West P, Neily J, Mills PD, Bagian JP. The effect of facility complexity on perceptions of safety climate in the operating room: size matters. Am J Med Qual. 2010;25(6):457-461.
While private or other public health care organizations can refuse to care for patients who have displayed disruptive behavior (DB), the VA Response to Disruptive Behavior of Patients law (38 CFR §17.107) prohibits the Veterans Health Administration (VHA) of the Department of Veterans Affairs (VA) from refusing care to veterans who display DB.1 The VHA defines DB as any behavior that is intimidating, threatening, or dangerous or that has, or could, jeopardize the health or safety of patients, VHA staff, or others.2
VA Response to DB Law
The VA Response to Disruptive Behavior of Patients requires the VHA to provide alternative care options that minimize risk while ensuring services; for example, providing care at a different location and/or time when additional staff are available to assist and monitor the patient. This can provide a unique opportunity to capture data on DB and the results of alternative forms of caring for this population.
The reason public health care organizations refuse care to persons who display DB is clear: DBs hinder business operations, are financially taxing, and put health care workers at risk.3-10 “In 2009, the VHA spent close to $5.5 million on workers’ compensation and medical expenditures for 425 incidents–or about $130,000 per DB incident (Hodgson M, Drummond D, Van Male L. Unpublished data, 2010).” In another study, 106 of 762 nurses in 1 hospital system reported an assault by a patient, and 30 required medical attention, which resulted in a total cost of $94,156.8 From 2002 to 2013, incidents of serious workplace violence requiring days off for an injured worker to recover on average were 4 times more common in health care than in other industries.6-11 Incidents of patient violence and aggression toward staff transcend specialization; however, hospital nurses and staff from the emergency, rehabilitation and gerontology departments, psychiatric unit, and home-based services are more susceptible and vulnerable to DB incidents than are other types of employees.8,10-19
Data reported by health care staff suggest that patients rather than staff members or visitors initiate > 70% of serious physical attacks against health care workers.9,13,20-23 A 2015 study of VHA health care providers (HCPs) found that > 60% had experienced some form of DB, verbal abuse being the most prevalent, followed by sexual abuse and physical abuse.20 Of 72,000 VHA staff responding to a nationwide survey, 13% experienced, on average, ≥ 1 assault by a veteran (eg, something was thrown at them; they were pushed, kicked, slapped; or were threatened or injured by a weapon).8,21
To meet its legal obligations and deliver empathetic care, the VHA documents and analyzes data on all patients who exhibit DB. A local DB Committee (DBC) reviews the data, whether it occurs in an inpatient or outpatient setting, such as community-based outpatient clinics. Once a DB incident is reported, the DBC begins an evidence-based risk evaluation, including the option of contacting the persons who displayed or experienced the DB. Goals are to (1) prevent future DB incidents; (2) detect vulnerabilities in the environment; and (3) collaborate with HCPs and patients to provide optimal care while improving the patient/provider interactions.
Effects of Disruptive Behavior
DB has negative consequences for both patients and health care workers and results in poor evaluations of care from both groups.27-32 Aside from interfering with safe medical care, DB also impacts care for other patients by delaying access to care and increasing appointment wait times due to employee absenteeism and staff shortages.3,4,20,32,33 For HCPs, patient violence is associated with unwillingness to provide care, briefer treatment periods, and decreases in occupational satisfaction, performance, and commitment
Harmful health effects experienced by HCPs who have been victims of DB include fear, mood disorders, anxiety, all symptoms of psychological distress and posttraumatic stress disorder (PTSD).10,22,30,34-36 In a study of the impact on productivity of PTSD triggered by job-related DB, PTSD symptoms were associated with withdrawal from or minimizing encounters with patients, job turnover, and troubles with thinking
Reporting Disruptive Behavior
The literature suggests that consistent and effective DB reporting is pivotal to improving the outcome and quality of care for those displaying DB.37-39 To provide high-quality health services to veterans who display DB, the VHA must promote the management and reporting of DB. Without knowledge of the full spectrum of DB events at VHA facilities, efforts to prevent or manage DB and ensure safety may have limited impact.7,37 Reports can be used for clinical decision making to optimize staff training in delivery of quality care while assuring staff safety. More than 80% of DB incidents occur during interactions with patients, thus this is a clinical issue that can affect the outcome of patient care.8,21
Documented DB reports are used to analyze the degree, frequency, and nature of incidents, which might reveal risk factors and develop preventive efforts and training for specific hazards.8,39 Some have argued that implementing a standardized DB reporting system is a crucial first step toward minimizing hazards and improving health care.38,40,41
When DB incidents were recorded through a hospital electronic reporting system and discussed in meetings, staff reported: (1) increased awareness of DB; (2) improved ability to manage DB incidents; and (3) amplified reporting of incidents.38,41,42 These findings support similar results from studies of an intervention implemented at VA Community Living Centers (CLCs) from 2013 to 2017: Staff Training in Assisted Living Residences (STAR-VA).4,12,19 The aim of STAR-VA was to minimize challenging dementia-related DB in CLCs. The intervention initially was established to train direct-care, assisted-living staff to provide better care to older patients displaying DB. Data revealed that documentation of DBs was, the first step to ensuring staff and patient safety.18,40
VHA Reporting System
In 2013, the VA Office of Inspector General (OIG) found no standardized documentation of DB events across the VA health care system.42 Instead, DB events were documented in multiple records in various locations, including administrative and progress notes in the electronic health record (EHR), police reports, e-mails, or letters submitted to DBC chairs.42 This situation reduced administrators’ ability to consider all relevant information and render appropriate decisions in DB cases.42 In 2015, based on OIG recommendations, the VHA implemented the Disruptive Behavior Reporting System (DBRS) nationwide, which allowed all VHA staff to report DB events. The DBRS was designed to address factors likely to impede reporting and management of DB, namely, complexity of and lack of access to a central reporting system.43,44 The DBRS is currently the primary VHA tool to document DB events.
The DBRS consists of 32 questions in 5 sections relating to the (1) location and time of DB event; (2) reporter; (3) disrupter; (4) DB event details; and (5) the person who experienced (experiencer) the event. The system also provides a list of the types of DB, such as inappropriate communication, bullying and/or intimidation, verbal or written threat of physical harm, physical violence, sexual harassment, sexual assault, and property damage. The DBRS has the potential to provide useful data on DB and DB reporting, such as the typical staff entering data and the number and/or types of DB occurring.
The DBRS complements the preexisting VHA policies and committees for care of veterans who display DB.1-3,14,21,24,25 The VHA Workplace Violence Prevention Program (WVPP) required facilities to submit data on DB events through a Workplace Behavioral Risk report. Data for the report were obtained from police reports, patient safety reports, DBC records, and notes in the EHR. Following implementations of DBRS, the number of DB events per year became a part of facility performance standards.
VHA is creating novel approaches to handling DB that allow health care workers to render care in a safe and effective manner guided by documented information. For example, DBCs can recommend the use of Category I Patient Record Flags (PRFs) following documented DB, which informs staff of the potential risk of DB and provides guidance on protective methods to use when meeting with the patient.2,21,24 A survey of 140 VA hospital chiefs of staff indicated that DBC procedures were related to a decrease in the rates of assaults.1 Additionally, VA provides training for staff in techniques to promote personal safety, such as identifying signs that precede DB, using verbal deescalation, and practicing therapeutic containment.
Resistance to Reporting
Many health care employees and employers are reticent to report DBs.22,31,43,45-48 Studies suggest health care organizations can cultivate a culture that is resistant to reporting DB.49,50 This complicates the ability of the health care system to design and maintain safety protocols and safer treatment plans.3,41,51 Worldwide, < 30% of DBs are reported.47 One barrier may be that supervisors may not wish to acknowledge DBs on their units or may not provide sufficient staff time for training or reporting.31,46,47 HCPs may worry that a DB report will stigmatize patients, especially those who are elderly or have cognitive impairment, brain injury, psychological illness, or developmental disability. Patients with cognitive conditions are reportedly 20% more likely to be violent toward caregivers and providers.31 A dementia diagnosis, for example, is associated with a high likelihood for DB.30,52 More than 80% of DB events displayed by patients with dementia may go unreported.26,31,50,52
Some clinicians may attribute DB to physiologic conditions that need to be treated, not reported. However, employers can face various legal liabilities if steps are not taken to protect employees.47,51 Federal and state statutes require that organizations provide a healthy and safe employment environment for workers. This requires that employers institute reasonable protective measures, such as procedures to intervene, policies on addressing DB incidents, and/or training to minimize or deescalate DB.51,53 Also, employees may sue employers if security measures are inadequate or deficient in properly investigating current and past evidence of DB or identifying vulnerabilities in the workplace. Unwillingness to investigate DB and safety-related workplace concerns have contributed to increased workplace violence and legal liability.52,53 The mission of caring and trust is consistent with assuring a safe environment.
Training and Empathetic Care
To combat cultural resistance to reporting DBs, more and perhaps different contextual approaches to education and training may be needed that address ethical dilemmas and concerns of providers. The success of training relies on administrators supporting staff in reporting DB. Training must address providers’ conflicting beliefs and assist with identifying strategies to provide the best possible care for patients who display DB.1,38 HCPs are less likely to document a DB if they feel that administrators are creating documentation that will have negative consequences for a patient. Thus, leadership is responsible for ensuring that misconceptions are dispelled through training and other efforts and information on how reported DB data will be used is communicated through strategic channels.
Education and training must consider empathic care that attempts to understand why patients behave as they do through the information gathered.55 Empathy in health care is multifaceted: It involves comprehending a patient’s viewpoint, circumstances, and feelings and the capacity to analyze whether one is comprehending these accurately in order to demonstrate supportive care.54,55
Improving patient and staff interaction once a problematic behavior is identified is the aim of empathic care. Increasing empathic care can improve compassionate, patient-centered interactions that begin once the patient seeks care. This approach has proven to decrease DB by patients with dementia and improve their care, lessen staff problems during interactions, and increase staff morale.20 Experts call for the adoption of an interpersonal approach to patient encounters, and there is evidence that creating organizational change by moving toward compassionate care can lead to a positive impact for patients.54,55
Future Studies
There are growth opportunities in utilization of the DBRS. Analysis of the DBRS database by the VA Central Office (VACO) showed that the system is underutilized by facilities across the VA system.56 In response to this current underutilization, VACO is taking steps to close these gaps through increasing training to staff and promotion of the use of the DBRS. A 2015 pilot study of VHA providers showed that > 70% of providers had experienced a DB as defined by VHA, but only 34% of them reported their most recently experienced DB within the past 12 months.20 Thus, DBRS use must be studied within the context that patient-perpetrated DB is underreported in health care organizations.5,9,29,41,43,57,58 Studies addressing national DBRS utilization patterns and the cost associated with implementing the DBRS also are needed. One study suggests that there is an association between measures of facility complexity and staff perceptions of safety, which should be considered in analyzing DBRS usage.57 Studies addressing the role of the DBRS and misconceptions that the tool may represent a punitive tool also are needed. VHA should consider how the attribution “disruptive behavior” assigns a negative connotation and leads HCPs to avoid using the DBRS. Additionally, DB reporting may increase when HCPs understand that DB reporting is part of the comprehensive, consultative strategy to provide the best care to patients.
Conclusion
Accurate reporting of DB events enables the development of strategies for multidisciplinary teams to work together to minimize hazards and to provide interventions that provide for the safe delivery of health care to all patients. Improving reporting ensures there is an accurate representation of how disruptive events impact care provided within a facility—and what types of variables may be associated with increased risk for these types of events.
Additionally, ensuring that reporting is maximized also provides the VHA with opportunities for DBCs to offer evidence-based risk assessment of violence and consultation to staff members who may benefit from improved competencies in working with patients who display DB. These potential improvements are consistent with the VHA I CARE values and will provide data that can inform recommendations for health care in other agencies/health care organizations.
Acknowledgments
This work was supported by the Center of Innovation on Disability and Rehabilitation Research (CINDRR) of the Health Services Research and Development Service, Office of Research and Development, Department of Veterans Affairs.
While private or other public health care organizations can refuse to care for patients who have displayed disruptive behavior (DB), the VA Response to Disruptive Behavior of Patients law (38 CFR §17.107) prohibits the Veterans Health Administration (VHA) of the Department of Veterans Affairs (VA) from refusing care to veterans who display DB.1 The VHA defines DB as any behavior that is intimidating, threatening, or dangerous or that has, or could, jeopardize the health or safety of patients, VHA staff, or others.2
VA Response to DB Law
The VA Response to Disruptive Behavior of Patients requires the VHA to provide alternative care options that minimize risk while ensuring services; for example, providing care at a different location and/or time when additional staff are available to assist and monitor the patient. This can provide a unique opportunity to capture data on DB and the results of alternative forms of caring for this population.
The reason public health care organizations refuse care to persons who display DB is clear: DBs hinder business operations, are financially taxing, and put health care workers at risk.3-10 “In 2009, the VHA spent close to $5.5 million on workers’ compensation and medical expenditures for 425 incidents–or about $130,000 per DB incident (Hodgson M, Drummond D, Van Male L. Unpublished data, 2010).” In another study, 106 of 762 nurses in 1 hospital system reported an assault by a patient, and 30 required medical attention, which resulted in a total cost of $94,156.8 From 2002 to 2013, incidents of serious workplace violence requiring days off for an injured worker to recover on average were 4 times more common in health care than in other industries.6-11 Incidents of patient violence and aggression toward staff transcend specialization; however, hospital nurses and staff from the emergency, rehabilitation and gerontology departments, psychiatric unit, and home-based services are more susceptible and vulnerable to DB incidents than are other types of employees.8,10-19
Data reported by health care staff suggest that patients rather than staff members or visitors initiate > 70% of serious physical attacks against health care workers.9,13,20-23 A 2015 study of VHA health care providers (HCPs) found that > 60% had experienced some form of DB, verbal abuse being the most prevalent, followed by sexual abuse and physical abuse.20 Of 72,000 VHA staff responding to a nationwide survey, 13% experienced, on average, ≥ 1 assault by a veteran (eg, something was thrown at them; they were pushed, kicked, slapped; or were threatened or injured by a weapon).8,21
To meet its legal obligations and deliver empathetic care, the VHA documents and analyzes data on all patients who exhibit DB. A local DB Committee (DBC) reviews the data, whether it occurs in an inpatient or outpatient setting, such as community-based outpatient clinics. Once a DB incident is reported, the DBC begins an evidence-based risk evaluation, including the option of contacting the persons who displayed or experienced the DB. Goals are to (1) prevent future DB incidents; (2) detect vulnerabilities in the environment; and (3) collaborate with HCPs and patients to provide optimal care while improving the patient/provider interactions.
Effects of Disruptive Behavior
DB has negative consequences for both patients and health care workers and results in poor evaluations of care from both groups.27-32 Aside from interfering with safe medical care, DB also impacts care for other patients by delaying access to care and increasing appointment wait times due to employee absenteeism and staff shortages.3,4,20,32,33 For HCPs, patient violence is associated with unwillingness to provide care, briefer treatment periods, and decreases in occupational satisfaction, performance, and commitment
Harmful health effects experienced by HCPs who have been victims of DB include fear, mood disorders, anxiety, all symptoms of psychological distress and posttraumatic stress disorder (PTSD).10,22,30,34-36 In a study of the impact on productivity of PTSD triggered by job-related DB, PTSD symptoms were associated with withdrawal from or minimizing encounters with patients, job turnover, and troubles with thinking
Reporting Disruptive Behavior
The literature suggests that consistent and effective DB reporting is pivotal to improving the outcome and quality of care for those displaying DB.37-39 To provide high-quality health services to veterans who display DB, the VHA must promote the management and reporting of DB. Without knowledge of the full spectrum of DB events at VHA facilities, efforts to prevent or manage DB and ensure safety may have limited impact.7,37 Reports can be used for clinical decision making to optimize staff training in delivery of quality care while assuring staff safety. More than 80% of DB incidents occur during interactions with patients, thus this is a clinical issue that can affect the outcome of patient care.8,21
Documented DB reports are used to analyze the degree, frequency, and nature of incidents, which might reveal risk factors and develop preventive efforts and training for specific hazards.8,39 Some have argued that implementing a standardized DB reporting system is a crucial first step toward minimizing hazards and improving health care.38,40,41
When DB incidents were recorded through a hospital electronic reporting system and discussed in meetings, staff reported: (1) increased awareness of DB; (2) improved ability to manage DB incidents; and (3) amplified reporting of incidents.38,41,42 These findings support similar results from studies of an intervention implemented at VA Community Living Centers (CLCs) from 2013 to 2017: Staff Training in Assisted Living Residences (STAR-VA).4,12,19 The aim of STAR-VA was to minimize challenging dementia-related DB in CLCs. The intervention initially was established to train direct-care, assisted-living staff to provide better care to older patients displaying DB. Data revealed that documentation of DBs was, the first step to ensuring staff and patient safety.18,40
VHA Reporting System
In 2013, the VA Office of Inspector General (OIG) found no standardized documentation of DB events across the VA health care system.42 Instead, DB events were documented in multiple records in various locations, including administrative and progress notes in the electronic health record (EHR), police reports, e-mails, or letters submitted to DBC chairs.42 This situation reduced administrators’ ability to consider all relevant information and render appropriate decisions in DB cases.42 In 2015, based on OIG recommendations, the VHA implemented the Disruptive Behavior Reporting System (DBRS) nationwide, which allowed all VHA staff to report DB events. The DBRS was designed to address factors likely to impede reporting and management of DB, namely, complexity of and lack of access to a central reporting system.43,44 The DBRS is currently the primary VHA tool to document DB events.
The DBRS consists of 32 questions in 5 sections relating to the (1) location and time of DB event; (2) reporter; (3) disrupter; (4) DB event details; and (5) the person who experienced (experiencer) the event. The system also provides a list of the types of DB, such as inappropriate communication, bullying and/or intimidation, verbal or written threat of physical harm, physical violence, sexual harassment, sexual assault, and property damage. The DBRS has the potential to provide useful data on DB and DB reporting, such as the typical staff entering data and the number and/or types of DB occurring.
The DBRS complements the preexisting VHA policies and committees for care of veterans who display DB.1-3,14,21,24,25 The VHA Workplace Violence Prevention Program (WVPP) required facilities to submit data on DB events through a Workplace Behavioral Risk report. Data for the report were obtained from police reports, patient safety reports, DBC records, and notes in the EHR. Following implementations of DBRS, the number of DB events per year became a part of facility performance standards.
VHA is creating novel approaches to handling DB that allow health care workers to render care in a safe and effective manner guided by documented information. For example, DBCs can recommend the use of Category I Patient Record Flags (PRFs) following documented DB, which informs staff of the potential risk of DB and provides guidance on protective methods to use when meeting with the patient.2,21,24 A survey of 140 VA hospital chiefs of staff indicated that DBC procedures were related to a decrease in the rates of assaults.1 Additionally, VA provides training for staff in techniques to promote personal safety, such as identifying signs that precede DB, using verbal deescalation, and practicing therapeutic containment.
Resistance to Reporting
Many health care employees and employers are reticent to report DBs.22,31,43,45-48 Studies suggest health care organizations can cultivate a culture that is resistant to reporting DB.49,50 This complicates the ability of the health care system to design and maintain safety protocols and safer treatment plans.3,41,51 Worldwide, < 30% of DBs are reported.47 One barrier may be that supervisors may not wish to acknowledge DBs on their units or may not provide sufficient staff time for training or reporting.31,46,47 HCPs may worry that a DB report will stigmatize patients, especially those who are elderly or have cognitive impairment, brain injury, psychological illness, or developmental disability. Patients with cognitive conditions are reportedly 20% more likely to be violent toward caregivers and providers.31 A dementia diagnosis, for example, is associated with a high likelihood for DB.30,52 More than 80% of DB events displayed by patients with dementia may go unreported.26,31,50,52
Some clinicians may attribute DB to physiologic conditions that need to be treated, not reported. However, employers can face various legal liabilities if steps are not taken to protect employees.47,51 Federal and state statutes require that organizations provide a healthy and safe employment environment for workers. This requires that employers institute reasonable protective measures, such as procedures to intervene, policies on addressing DB incidents, and/or training to minimize or deescalate DB.51,53 Also, employees may sue employers if security measures are inadequate or deficient in properly investigating current and past evidence of DB or identifying vulnerabilities in the workplace. Unwillingness to investigate DB and safety-related workplace concerns have contributed to increased workplace violence and legal liability.52,53 The mission of caring and trust is consistent with assuring a safe environment.
Training and Empathetic Care
To combat cultural resistance to reporting DBs, more and perhaps different contextual approaches to education and training may be needed that address ethical dilemmas and concerns of providers. The success of training relies on administrators supporting staff in reporting DB. Training must address providers’ conflicting beliefs and assist with identifying strategies to provide the best possible care for patients who display DB.1,38 HCPs are less likely to document a DB if they feel that administrators are creating documentation that will have negative consequences for a patient. Thus, leadership is responsible for ensuring that misconceptions are dispelled through training and other efforts and information on how reported DB data will be used is communicated through strategic channels.
Education and training must consider empathic care that attempts to understand why patients behave as they do through the information gathered.55 Empathy in health care is multifaceted: It involves comprehending a patient’s viewpoint, circumstances, and feelings and the capacity to analyze whether one is comprehending these accurately in order to demonstrate supportive care.54,55
Improving patient and staff interaction once a problematic behavior is identified is the aim of empathic care. Increasing empathic care can improve compassionate, patient-centered interactions that begin once the patient seeks care. This approach has proven to decrease DB by patients with dementia and improve their care, lessen staff problems during interactions, and increase staff morale.20 Experts call for the adoption of an interpersonal approach to patient encounters, and there is evidence that creating organizational change by moving toward compassionate care can lead to a positive impact for patients.54,55
Future Studies
There are growth opportunities in utilization of the DBRS. Analysis of the DBRS database by the VA Central Office (VACO) showed that the system is underutilized by facilities across the VA system.56 In response to this current underutilization, VACO is taking steps to close these gaps through increasing training to staff and promotion of the use of the DBRS. A 2015 pilot study of VHA providers showed that > 70% of providers had experienced a DB as defined by VHA, but only 34% of them reported their most recently experienced DB within the past 12 months.20 Thus, DBRS use must be studied within the context that patient-perpetrated DB is underreported in health care organizations.5,9,29,41,43,57,58 Studies addressing national DBRS utilization patterns and the cost associated with implementing the DBRS also are needed. One study suggests that there is an association between measures of facility complexity and staff perceptions of safety, which should be considered in analyzing DBRS usage.57 Studies addressing the role of the DBRS and misconceptions that the tool may represent a punitive tool also are needed. VHA should consider how the attribution “disruptive behavior” assigns a negative connotation and leads HCPs to avoid using the DBRS. Additionally, DB reporting may increase when HCPs understand that DB reporting is part of the comprehensive, consultative strategy to provide the best care to patients.
Conclusion
Accurate reporting of DB events enables the development of strategies for multidisciplinary teams to work together to minimize hazards and to provide interventions that provide for the safe delivery of health care to all patients. Improving reporting ensures there is an accurate representation of how disruptive events impact care provided within a facility—and what types of variables may be associated with increased risk for these types of events.
Additionally, ensuring that reporting is maximized also provides the VHA with opportunities for DBCs to offer evidence-based risk assessment of violence and consultation to staff members who may benefit from improved competencies in working with patients who display DB. These potential improvements are consistent with the VHA I CARE values and will provide data that can inform recommendations for health care in other agencies/health care organizations.
Acknowledgments
This work was supported by the Center of Innovation on Disability and Rehabilitation Research (CINDRR) of the Health Services Research and Development Service, Office of Research and Development, Department of Veterans Affairs.
1. Hodgson MJ, Mohr DC, Drummond DJ, Bell M, Van Male L. Managing disruptive patients in health care: necessary solutions to a difficult problem. Am J Ind Med. 2012;55(11):1009-1017.
2. US Department of Veterans Affairs, Veterans Health Administration. VHA Directive 2010-053. Patient Record Flags. https://www.va.gov/vhapublications/ViewPublication.asp?pub_ID=2341 Published December 3, 2010. Accessed March 29, 2019.
3. US Department of Veterans Affairs, Veterans Health Administration. VHA Directive 2012-026. Sexual Assaults and Other Defined Public Safety Incidents in VHA Facilities. https://www.va.gov/vhapublications/ViewPublication.asp?pub_ID=2797. Published September 27, 2012. Accessed March 29, 2019.
4. Curyto KJ, McCurry SM, Luci K, Karlin BE, Teri L, Karel MJ. Managing challenging behaviors of dementia in veterans: identifying and changing activators and consequences using STAR-VA. J Gerontol Nurs. 2017;43(2):33-43.
5. Speroni KG, Fitch T, Dawson E, Dugan L, Atherton M. Incidence and cost of nurse workplace violence perpetrated by hospital patients or patient visitors. J Emerg Nurs. 2014;40(3):218-228.
6. Phillips JP. Workplace violence against health care workers in the United States. NEJM. 2016;374(17):1661-1669.
7. Janocha JA, Smith RT. Workplace safety and health in the health care and social assistance industry, 2003–07. https://www.bls.gov/opub/mlr/cwc/workplace-safety-and-health-in-the-health-care-and-social-assistance-industry-2003-07.pdf. Published August 30, 2010. Accessed February 19, 2019.
8. US Department of Labor, Occupational Safety and Health Administration. Workplace violence in healthcare: understanding the challenge. https://www.osha.gov/Publications/OSHA3826.pdf. Published December 2015. Accessed February 19, 2019.
9. US Department of Labor, Occupational Safety and Health Administration. Prevention of Workplace Violence in Healthcare and Social Assistance. Occupational Safety and Health Administration, https://www.govinfo.gov/content/pkg/FR-2016-12-07/pdf/2016-29197.pdf. Accessed January 20, 2017.
10. Gerberich SG, Church TR, McGovern PM, et al. An epidemiological study of the magnitude and consequences of work related violence: the Minnesota Nurses’ Study. Occup Environ Med. 2004;61(6):495-503.
11. Sherman MF, Gershon RRM, Samar SM, Pearson JM, Canton AN, Damsky MR. Safety factors predictive of job satisfaction and job retention among home healthcare aides. J Occup Environ Med. 2008;50(12):1430-1441.
12. Karel MJ, Teri L, McConnell E, Visnic S, Karlin BE. Effectiveness of expanded implementation of STAR-VA for managing dementia-related behaviors among veterans. Gerontologist. 2016;56(1):126-134.
13. US Department of Labor, Bureau of Labor Statistics. Nonfatal occupational injuries and illnesses requiring days away from work. https://www.bls.gov/news.release/archives/osh2_11192015.htm. Published November 19, 2015.
14. Beech B, Leather P. Workplace violence in the health care sector: A review of staff training and integration of training evaluation models. Aggression Violent Behav. 2006;11(1):27-43.
15. Campbell CL, McCoy S, Burg MA, Hoffman N. Enhancing home care staff safety through reducing client aggression and violence in noninstitutional care settings: a systematic review. Home Health Care Manage Pract. 2014;26(1):3-10.
16. Gallant-Roman MA. Strategies and tools to reduce workplace violence. AAOHNJ. 2008;56(11):449-454.
17. Weinberger LE, Sreenivasan S, Smee DE, McGuire J, Garrick T. Balancing safety against obstruction to health care access: an examination of behavioral flags in the VA health care system. J Threat Assess Manage. 2018;5(1):35-41.
18. Elbogen EB, Johnson SC, Wagner HR, et al. Protective factors and risk modification of violence in Iraq and Afghanistan war veterans. J Clin Psychiatry. 2012;73(6):e767-e773.
19. Karlin BE, Visnic S, McGee JS, Teri L. Results from the multisite implementation of STAR-VA: a multicomponent psychosocial intervention for managing challenging dementia-related behaviors of veterans. Psychol Serv. 2014;11(2):200-208.
20. Semeah LM, Campbell CL, Cowper DC, Peet AC. Serving our homeless veterans: patient perpetrated violence as a barrier to health care access. J Pub Nonprofit Aff. 2017;3(2):223-234.
21. Hodgson MJ, Reed R, Craig T, et al. Violence in healthcare facilities: lessons from the Veterans Health Administration. J Occup Environ Med. 2004;46(11):1158-1165.
22. Farrell GA, Bobrowski C, Bobrowski P. Scoping workplace aggression in nursing: findings from an Australian study. J Adv Nurs. 2006;55(6):778-787.
23. Barling J, Rogers AG, Kelloway EK. Behind closed doors: in-home workers’ experience of sexual harassment and workplace violence. J Occup Health Psychol. 2001;6(3):255-269.
24. Pompeii LA, Schoenfisch AL, Lipscomb HJ, Dement JM, Smith CD, Upadhyaya M. Physical assault, physical threat, and verbal abuse perpetrated against hospital workers by patients or visitors in six U.S. hospitals. Am J Ind Med. 2015;58(11):1194-1204.
25. Sippel LM, Mota NP, Kachadourian LK, et al. The burden of hostility in U.S. veterans: results from the National Health and Resilience in Veterans Study. Psychiatry Res. 2016;243(suppl C):421-430.
26. Campbell C. Patient Violence and Aggression in Non-Institutional Health Care Settings: Predictors of Reporting By Healthcare Providers [doctoral dissertation]. Orlando: University of Central Florida; 2016.
27. Galinsky T, Feng HA, Streit J, et al. Risk factors associated with patient assaults of home healthcare workers. Rehabil Nurs. 2010;35(5):206-215.
28. Campbell CL. Incident reporting by health-care workers in noninstitutional care settings. Trauma, Violence Abuse. 2017;18(4):445-456.
29. Arnetz JE, Arnetz BB. Violence towards health care staff and possible effects on the quality of patient care. Soc Sci Med. 2001;52(3):417-427.
30. Gates D, Fitzwater E, Succop P. Relationships of stressors, strain, and anger to caregiver assaults. Issues Ment Health Nurs. 2003;24(8):775-793.
31. Brillhart B, Kruse B, Heard L. Safety concerns for rehabilitation nurses in home care. Rehabil Nurs. 2004;29(6):227-229.
32. Taylor H. Patient violence against clinicians: managing the risk. Innov Clin Neurosci. 2013;10(3):40-42.
33. US Department of Veterans Affairs, Office of Public and Intergovernmental Affairs. The Joint Commission releases results of surveys of the VA health care system. https://www.va.gov/opa/pressrel/pressrelease.cfm?id=2808. Updated August 5, 2014. Accessed February 19, 2019.
34. Büssing A, Höge T. Aggression and violence against home care workers. J Occup Health Psychol. 2004;9(3):206-219.
35. Geiger-Brown J, Muntaner C, McPhaul K, Lipscomb J, Trinkoff A. Abuse and violence during home care work as predictor of worker depression. Home Health Care Serv Q. 2007;26(1):59-77.
36. Gates DM, Gillespie GL, Succop P. Violence against nurses and its impact on stress and productivity. Nurs Econ. 2011;29(2):59-66.
37. Petterson IL, Arnetz BB. Psychosocial stressors and well-being in health care workers: the impact of an intervention program. Soc Sci Med. 1998;47(11):1763-1772.
38. Arnetz JE, Arnetz BB. Implementation and evaluation of a practical intervention programme for dealing with violence towards health care workers. J Adv Nurs. 2000;31(3):668-680.
39. Arnetz JE, Hamblin L, Russell J, et al. Preventing patient-to-worker violence in hospitals: outcome of a randomized controlled intervention. J Occup Environ Med. 2017;59(1):18-27.
40. Elbogen EB, Tomkins AJ, Pothuloori AP, Scalora MJ. Documentation of violence risk information in psychiatric hospital patient charts: an empirical examination. J Am Acad Psychiatry Law. 2003;31(1):58-64.
41. Winsvold Prang I, Jelson-Jorgensen LP. Should I report? A qualitative study of barriers to incident reporting among nurses working in nursing homes. Geriatr Nurs. 2014;35(6):441-447.
42. US Department of Veterans Affairs, Office of Inspector General. Healthcare inspection: management of disruptive patient behavior at VA medical facilities. Report No. 11-02585-129. https://www.va.gov/oig/pubs/VAOIG-11-02585-129.pdf. Published Mrach 7, 2013. Accessed February 21, 2019.
43. Lipscomb J, London M. Not Part of the Job: How to Take a Stand Against Violence in the Work Setting. Silver Spring, MD: American Nurses Association; 2015.
44. May DD, Grubbs LM. The extent, nature, and precipitating factors of nurse assault among three groups of registered nurses in a regional medical center. J Emerg Nurs. 2002;28(1):11-17.
45. Wharton TC, Ford BK. What is known about dementia care recipient violence and aggression against caregivers? J Gerontol Soc Work. 2014;57(5):460-477.
46. Brennan C, Worrall-Davies A, McMillan D, Gilbody S, House A. The hospital anxiety and depression scale: a diagnostic meta-analysis of case-finding ability. J Psychosom Res. 2010;69(4):371-378.
47. McPhaul K, Lipscomb J, Johnson J. Assessing risk for violence on home health visits. Home Healthc Nurse. 2010;28(5):278-289.
48. McPhaul KM, London M, Murrett K, Flannery K, Rosen J, Lipscomb J. Environmental evaluation for workplace violence in healthcare and social services. J Safety Res. 2008;39(2):237-250.
49. Kelly JA, Somlai AM, DiFranceisco WJ, et al. Bridging the gap between the science and service of HIV prevention: transferring effective research-based HIV prevention interventions to community AIDS service providers. Am J Public Health. 2000;90(7):1082-1088.
50. Pawlin S. Reporting violence. Emerg Nurse. 2008;16(4):16-21.
51. Brakel SJ. Legal liability and workplace violence. J Am Acad Psychiatry Law. 1998;26(4):553-562.
52. Neuman JH, Baron RA. Workplace violence and workplace aggression: evidence concerning specific forms, potential causes, and preferred targets. J Manage. 1998;24(3):391-419.53. Ferns T, Chojnacka I. Angels and swingers, matrons and sinners: nursing stereotypes. Br J Nurs. 2005;14(19):1028-1032.
54. Mercer SW, Reynolds WJ. Empathy and quality of care. Br J Gen Pract 2002;52(suppl):S9-S12.
55. Lee TH. An Epidemic of Empathy in Healthcare: How to Deliver Compassionate, Connected Patient Care That Creates a Competitive Advantage. Columbus, OH: McGraw-Hill Education; 2015.
56. US Department of Veterans Affairs, Veterans Health Administrastion. Veterans Health Administration workplace violence prevention program (WVPP): disruptive behavior reporting system utilization report. Published 2017. https://vaww.portal2.va.gov/sites/wvpp/Shared%20Documents/DBRS%20Utilization%20Reports/FY2017%20DBRS%20Quarterly%20Utilization%20Report%20(Quarter%201).pdf. [Source not verified.]
57. Campbell CL, Burg, MA, Gammonley D. Measures for incident reporting of patient violence and aggression towards healthcare providers: a systematic review. Aggression Violent Behav. 2015;25(part B):314-322.
58. Carney PT, West P, Neily J, Mills PD, Bagian JP. The effect of facility complexity on perceptions of safety climate in the operating room: size matters. Am J Med Qual. 2010;25(6):457-461.
1. Hodgson MJ, Mohr DC, Drummond DJ, Bell M, Van Male L. Managing disruptive patients in health care: necessary solutions to a difficult problem. Am J Ind Med. 2012;55(11):1009-1017.
2. US Department of Veterans Affairs, Veterans Health Administration. VHA Directive 2010-053. Patient Record Flags. https://www.va.gov/vhapublications/ViewPublication.asp?pub_ID=2341 Published December 3, 2010. Accessed March 29, 2019.
3. US Department of Veterans Affairs, Veterans Health Administration. VHA Directive 2012-026. Sexual Assaults and Other Defined Public Safety Incidents in VHA Facilities. https://www.va.gov/vhapublications/ViewPublication.asp?pub_ID=2797. Published September 27, 2012. Accessed March 29, 2019.
4. Curyto KJ, McCurry SM, Luci K, Karlin BE, Teri L, Karel MJ. Managing challenging behaviors of dementia in veterans: identifying and changing activators and consequences using STAR-VA. J Gerontol Nurs. 2017;43(2):33-43.
5. Speroni KG, Fitch T, Dawson E, Dugan L, Atherton M. Incidence and cost of nurse workplace violence perpetrated by hospital patients or patient visitors. J Emerg Nurs. 2014;40(3):218-228.
6. Phillips JP. Workplace violence against health care workers in the United States. NEJM. 2016;374(17):1661-1669.
7. Janocha JA, Smith RT. Workplace safety and health in the health care and social assistance industry, 2003–07. https://www.bls.gov/opub/mlr/cwc/workplace-safety-and-health-in-the-health-care-and-social-assistance-industry-2003-07.pdf. Published August 30, 2010. Accessed February 19, 2019.
8. US Department of Labor, Occupational Safety and Health Administration. Workplace violence in healthcare: understanding the challenge. https://www.osha.gov/Publications/OSHA3826.pdf. Published December 2015. Accessed February 19, 2019.
9. US Department of Labor, Occupational Safety and Health Administration. Prevention of Workplace Violence in Healthcare and Social Assistance. Occupational Safety and Health Administration, https://www.govinfo.gov/content/pkg/FR-2016-12-07/pdf/2016-29197.pdf. Accessed January 20, 2017.
10. Gerberich SG, Church TR, McGovern PM, et al. An epidemiological study of the magnitude and consequences of work related violence: the Minnesota Nurses’ Study. Occup Environ Med. 2004;61(6):495-503.
11. Sherman MF, Gershon RRM, Samar SM, Pearson JM, Canton AN, Damsky MR. Safety factors predictive of job satisfaction and job retention among home healthcare aides. J Occup Environ Med. 2008;50(12):1430-1441.
12. Karel MJ, Teri L, McConnell E, Visnic S, Karlin BE. Effectiveness of expanded implementation of STAR-VA for managing dementia-related behaviors among veterans. Gerontologist. 2016;56(1):126-134.
13. US Department of Labor, Bureau of Labor Statistics. Nonfatal occupational injuries and illnesses requiring days away from work. https://www.bls.gov/news.release/archives/osh2_11192015.htm. Published November 19, 2015.
14. Beech B, Leather P. Workplace violence in the health care sector: A review of staff training and integration of training evaluation models. Aggression Violent Behav. 2006;11(1):27-43.
15. Campbell CL, McCoy S, Burg MA, Hoffman N. Enhancing home care staff safety through reducing client aggression and violence in noninstitutional care settings: a systematic review. Home Health Care Manage Pract. 2014;26(1):3-10.
16. Gallant-Roman MA. Strategies and tools to reduce workplace violence. AAOHNJ. 2008;56(11):449-454.
17. Weinberger LE, Sreenivasan S, Smee DE, McGuire J, Garrick T. Balancing safety against obstruction to health care access: an examination of behavioral flags in the VA health care system. J Threat Assess Manage. 2018;5(1):35-41.
18. Elbogen EB, Johnson SC, Wagner HR, et al. Protective factors and risk modification of violence in Iraq and Afghanistan war veterans. J Clin Psychiatry. 2012;73(6):e767-e773.
19. Karlin BE, Visnic S, McGee JS, Teri L. Results from the multisite implementation of STAR-VA: a multicomponent psychosocial intervention for managing challenging dementia-related behaviors of veterans. Psychol Serv. 2014;11(2):200-208.
20. Semeah LM, Campbell CL, Cowper DC, Peet AC. Serving our homeless veterans: patient perpetrated violence as a barrier to health care access. J Pub Nonprofit Aff. 2017;3(2):223-234.
21. Hodgson MJ, Reed R, Craig T, et al. Violence in healthcare facilities: lessons from the Veterans Health Administration. J Occup Environ Med. 2004;46(11):1158-1165.
22. Farrell GA, Bobrowski C, Bobrowski P. Scoping workplace aggression in nursing: findings from an Australian study. J Adv Nurs. 2006;55(6):778-787.
23. Barling J, Rogers AG, Kelloway EK. Behind closed doors: in-home workers’ experience of sexual harassment and workplace violence. J Occup Health Psychol. 2001;6(3):255-269.
24. Pompeii LA, Schoenfisch AL, Lipscomb HJ, Dement JM, Smith CD, Upadhyaya M. Physical assault, physical threat, and verbal abuse perpetrated against hospital workers by patients or visitors in six U.S. hospitals. Am J Ind Med. 2015;58(11):1194-1204.
25. Sippel LM, Mota NP, Kachadourian LK, et al. The burden of hostility in U.S. veterans: results from the National Health and Resilience in Veterans Study. Psychiatry Res. 2016;243(suppl C):421-430.
26. Campbell C. Patient Violence and Aggression in Non-Institutional Health Care Settings: Predictors of Reporting By Healthcare Providers [doctoral dissertation]. Orlando: University of Central Florida; 2016.
27. Galinsky T, Feng HA, Streit J, et al. Risk factors associated with patient assaults of home healthcare workers. Rehabil Nurs. 2010;35(5):206-215.
28. Campbell CL. Incident reporting by health-care workers in noninstitutional care settings. Trauma, Violence Abuse. 2017;18(4):445-456.
29. Arnetz JE, Arnetz BB. Violence towards health care staff and possible effects on the quality of patient care. Soc Sci Med. 2001;52(3):417-427.
30. Gates D, Fitzwater E, Succop P. Relationships of stressors, strain, and anger to caregiver assaults. Issues Ment Health Nurs. 2003;24(8):775-793.
31. Brillhart B, Kruse B, Heard L. Safety concerns for rehabilitation nurses in home care. Rehabil Nurs. 2004;29(6):227-229.
32. Taylor H. Patient violence against clinicians: managing the risk. Innov Clin Neurosci. 2013;10(3):40-42.
33. US Department of Veterans Affairs, Office of Public and Intergovernmental Affairs. The Joint Commission releases results of surveys of the VA health care system. https://www.va.gov/opa/pressrel/pressrelease.cfm?id=2808. Updated August 5, 2014. Accessed February 19, 2019.
34. Büssing A, Höge T. Aggression and violence against home care workers. J Occup Health Psychol. 2004;9(3):206-219.
35. Geiger-Brown J, Muntaner C, McPhaul K, Lipscomb J, Trinkoff A. Abuse and violence during home care work as predictor of worker depression. Home Health Care Serv Q. 2007;26(1):59-77.
36. Gates DM, Gillespie GL, Succop P. Violence against nurses and its impact on stress and productivity. Nurs Econ. 2011;29(2):59-66.
37. Petterson IL, Arnetz BB. Psychosocial stressors and well-being in health care workers: the impact of an intervention program. Soc Sci Med. 1998;47(11):1763-1772.
38. Arnetz JE, Arnetz BB. Implementation and evaluation of a practical intervention programme for dealing with violence towards health care workers. J Adv Nurs. 2000;31(3):668-680.
39. Arnetz JE, Hamblin L, Russell J, et al. Preventing patient-to-worker violence in hospitals: outcome of a randomized controlled intervention. J Occup Environ Med. 2017;59(1):18-27.
40. Elbogen EB, Tomkins AJ, Pothuloori AP, Scalora MJ. Documentation of violence risk information in psychiatric hospital patient charts: an empirical examination. J Am Acad Psychiatry Law. 2003;31(1):58-64.
41. Winsvold Prang I, Jelson-Jorgensen LP. Should I report? A qualitative study of barriers to incident reporting among nurses working in nursing homes. Geriatr Nurs. 2014;35(6):441-447.
42. US Department of Veterans Affairs, Office of Inspector General. Healthcare inspection: management of disruptive patient behavior at VA medical facilities. Report No. 11-02585-129. https://www.va.gov/oig/pubs/VAOIG-11-02585-129.pdf. Published Mrach 7, 2013. Accessed February 21, 2019.
43. Lipscomb J, London M. Not Part of the Job: How to Take a Stand Against Violence in the Work Setting. Silver Spring, MD: American Nurses Association; 2015.
44. May DD, Grubbs LM. The extent, nature, and precipitating factors of nurse assault among three groups of registered nurses in a regional medical center. J Emerg Nurs. 2002;28(1):11-17.
45. Wharton TC, Ford BK. What is known about dementia care recipient violence and aggression against caregivers? J Gerontol Soc Work. 2014;57(5):460-477.
46. Brennan C, Worrall-Davies A, McMillan D, Gilbody S, House A. The hospital anxiety and depression scale: a diagnostic meta-analysis of case-finding ability. J Psychosom Res. 2010;69(4):371-378.
47. McPhaul K, Lipscomb J, Johnson J. Assessing risk for violence on home health visits. Home Healthc Nurse. 2010;28(5):278-289.
48. McPhaul KM, London M, Murrett K, Flannery K, Rosen J, Lipscomb J. Environmental evaluation for workplace violence in healthcare and social services. J Safety Res. 2008;39(2):237-250.
49. Kelly JA, Somlai AM, DiFranceisco WJ, et al. Bridging the gap between the science and service of HIV prevention: transferring effective research-based HIV prevention interventions to community AIDS service providers. Am J Public Health. 2000;90(7):1082-1088.
50. Pawlin S. Reporting violence. Emerg Nurse. 2008;16(4):16-21.
51. Brakel SJ. Legal liability and workplace violence. J Am Acad Psychiatry Law. 1998;26(4):553-562.
52. Neuman JH, Baron RA. Workplace violence and workplace aggression: evidence concerning specific forms, potential causes, and preferred targets. J Manage. 1998;24(3):391-419.53. Ferns T, Chojnacka I. Angels and swingers, matrons and sinners: nursing stereotypes. Br J Nurs. 2005;14(19):1028-1032.
54. Mercer SW, Reynolds WJ. Empathy and quality of care. Br J Gen Pract 2002;52(suppl):S9-S12.
55. Lee TH. An Epidemic of Empathy in Healthcare: How to Deliver Compassionate, Connected Patient Care That Creates a Competitive Advantage. Columbus, OH: McGraw-Hill Education; 2015.
56. US Department of Veterans Affairs, Veterans Health Administrastion. Veterans Health Administration workplace violence prevention program (WVPP): disruptive behavior reporting system utilization report. Published 2017. https://vaww.portal2.va.gov/sites/wvpp/Shared%20Documents/DBRS%20Utilization%20Reports/FY2017%20DBRS%20Quarterly%20Utilization%20Report%20(Quarter%201).pdf. [Source not verified.]
57. Campbell CL, Burg, MA, Gammonley D. Measures for incident reporting of patient violence and aggression towards healthcare providers: a systematic review. Aggression Violent Behav. 2015;25(part B):314-322.
58. Carney PT, West P, Neily J, Mills PD, Bagian JP. The effect of facility complexity on perceptions of safety climate in the operating room: size matters. Am J Med Qual. 2010;25(6):457-461.
Nonopioid Alternatives to Addressing Pain Intensity: A Retrospective Look at 2 Noninvasive Pain Treatment Devices
Chronic pain is common among veterans treated in Veterans Health Administration (VHA) facilities, and optimal management remains challenging in the context of the national opioid misuse epidemic. The Eastern Oklahoma VA Health Care System (EOVAHCS) Pain Program offers a range of services that allow clinicians to tailor multimodal treatment strategies to a veteran’s needs. In 2014, a Modality Clinic was established to assess the utility of adding noninvasive treatment devices to the pain program’s armamentarium. This article addresses the context for introducing these devices and describes the EOVAHCS Pain Program and Modality Clinic. Also discussed are procedures and findings from an initial quality improvement evaluation designed to inform decision making regarding retention, expansion, or elimination of the EOVAHCS noninvasive, pain treatment device program.
Opioid prescriptions increased from 76 million in 1991 to 219 million in 2011. In 2011, the annual cost of chronic pain in the US was estimated at $635 billion.1-6 The confluence of an increasing concern about undertreatment of pain and overconfidence for the safety of opioids led to what former US Surgeon General Vivek H. Murthy, MD, called the opioid crisis.7 As awareness of its unintended consequences of opioid prescribing increased, the VHA began looking for nonopioid treatments that would decrease pain intensity. The 1993 article by Kehlet and Dahl was one of the first discussions of a multimodal nonpharmacologic strategy for addressing acute postoperative pain.8 Their pivotal literature review concluded that nonpharmacologic modalities, such as acupuncture, cranial manipulation, cranial electrostimulation treatment (CES), and low-level light technologies (LLLT), carried less risk and produced equal or greater clinical effects than those of drug therapies.8
Multimodal treatment approaches increasingly are encouraged, and nonopioid pain control has become more common across medical disciplines from physical therapy to anesthesiology.8-10 Innovative, noninvasive devices designed for self-use have appeared on the market. Many of these devices incorporate microcurrent electrical therapy (MET), CES, and/or LLLT (also known as cold laser).11-16 LLLT is a light modality that seems to lead to increased ATP production, resulting in improved healing and decreased inflammation.13-16 Although CES has been studied in a variety of patient populations, its effectiveness is not well understood.16 Research on the effects of CES on neurotransmitter levels as well as activation of parts of the brain involved in pain reception and transmission should clarify these mechanisms. Research has shown improvements in sleep and mood as well as overall pain reduction.11,16 Research has focused primarily on individual modalities rather than on combination devices and has been conducted on populations unlike the veteran population (eg, women with fibromyalgia).
Most of the devices that use electrical or LLLT cannot be used safely by patients who have implantable electrical devices or have medical conditions such as unstable seizures, pregnancy, and active malignancies.
The most common adverse effects (AEs) of CES—dizziness and headaches—are minimal compared with the AEs of pain medications. MET and LLLT AEs generally are limited to skin irritation and muscle soreness.11 Most devices require a prescription, and manufacturers provide training for purchase.
The Pain Program
EOVAHCS initially established its consultative pain program in 2013 to provide support, recommendations, and education about managing pain in veterans to primary care providers (PCPs). Veterans are referred to the pain program for a face-to-face assessment and set of recommendations to assist in developing a comprehensive pain treatment plan. Consistent with its multimodal, biopsychosocial rehabilitation model approach, the program also offers several chronic pain treatment services, including patient education courses, cognitive behavioral therapy (CBT) for chronic pain, chiropractic care, biofeedback, relaxation training, steroid injections, pain coaching, and a pain modality (noninvasive device) clinic. During their assessment, veterans are evaluated for the appropriateness of these programs, including treatment through the Pain Modality Clinic.
Pain Modality Clinic
The EOVAHCS Pain Modality Clinic was created in 2014 as a treatment and device-trial program to provide veterans access to newer noninvasive, patient-driven treatment devices as part of an active chronic pain self-management plan. A crucial innovation is that these devices are designed to be used by patients in their homes. These devices can be expensive, and not every patient will benefit from their use; therefore, clinic leaders recommended a trial before a device is issued to a veteran for home use.
The Pain Modality Clinic coordinator trains clinic facilitators on the device according to manufacturer’s guidelines. Each participating veteran takes part in a device trial to confirm that he or she is able to use the recommended device independently and is likely to benefit from its use. When appropriate, veterans who do not respond to the initial device trial could test the potential benefit of another device. Although data from these device trials are collected primarily to inform clinical decision making, this information also is useful in guiding local policy regarding continued support for each of the modalities.
Veterans who have chronic or persistent pain (≥ 3 months) that interferes with function or quality of life are considered good candidates for a device trial if they are actively involved in pain self-care, logistically able to participate, able to use a device long-term, and have no contraindications. “Active involvement” could be met by participation in any pain management effort, whether a specific exercise program, CBT, or other treatment.
The Modality Clinic currently offers device trials for persistent pain with Alpha-Stim-M (AS-M; Electromedical Products International, Mineral Wells, TX), Laser Touch One (LTO; Renewal Technologies, LLC, Phoenix, AZ), and Neurolumen (Oklahoma City, OK). Neurolumen devices were not available in the clinic initially and will not be discussed further in this article.
The first Alpha-Stim machine using MET and CES technology was created in 1981 for in-office pain management. In 2012, the currently used AS-M became available.11 AS-M is FDA approved for treating pain, anxiety, depression, and sleep problems and is the device used in the EOVAHCS Modality Clinic. AS-M uses probes or electrodes to send a MET waveform through the body area in pain. The device uses ear clips to provide CES, which is thought to increase alpha waves in the brain.11 The LTO is a device that combines LLLT and MET technologies in a home-use design.14 LTO is FDA approved for treating painand is a portable personal pain-relief device applied to the area of pain using electroconductive gel.
Both devices are designed for long-term, self-use, making them viable parts of a multimodal, chronic pain treatment plan. Contraindications for AS-M and LTO include having a pacemaker or an implantable defibrillator, pregnancy, current malignancy, or seizures. Eligible veterans with persistent pain and high levels of depression, anxiety, and/or sleep problems generally are triaged to AS-M, whereas those who have only pain intensity issues usually are assigned to LTO. Referral to the Modality Clinic is not limited to a specific type of pain; common pain conditions seen in the clinic are spine and joint pain, arthritis pain, myofascial pain, headaches, and neuropathy.
Training and Device Trials
Eligible veterans are educated about the device and complete clinical informed consent, which is documented in the electronic health record. The veterans’ primary care and/or specialist providers are contacted for concurrence regarding veterans’ participation in the treatment.
Protocols for the device trials are based on the manufacturers’ recommendations, adjusted to what is feasible in the clinic (manufacturers approved the changes). The number of treatments per trial varies by device. For AS-M, veterans come to the clinic 5 days a week for 2 weeks. For LTO, veterans attend the clinic 5 days a week for 1 week.
At the beginning of a device trial, a trained facilitator teaches each veteran and caregiver to use the device, sets functional goals for the trial, and provides education on the trial questionnaires and daily pain logs. The veteran then follows the device protocol in the clinic where the facilitator can respond to questions and address any issues. With support from their caregivers, veterans are expected to become independent on their device use by the end of the trial. Clinic staff or the veteran can stop the device trial at any point, without affecting the veteran’s participation in or eligibility for other EOVAHCS pain programs.
This project was submitted to the University of Oklahoma Health Sciences Center Institutional Review Board and was exempted from institutional review board oversight as a retrospective, quality improvement effort. Before data analysis, the EOVAHCS Coordinator for Research and Development reviewed the procedures to ensure that all policies were being followed.
Methods
Data for veterans who completed valid treatments of AS-M or LTO from May 9, 2014 to August 20, 2016, were included in the analyses. For an AS-M treatment to be considered valid, the veteran must have attended at least 8 sessions and completed assessment instruments at baseline (preintervention) and following completion (postintervention). For an LTO treatment to be considered valid, the veteran must have attended at least 4 sessions and completed assessment measures at baseline and after completion.
Measures
Veterans completed the following measures at baseline and after trial completion:
The Beck Depression Inventory (BDI-II) is a 21-item measure designed to assess depressive symptoms. Each item assesses intensity on a 0-to-3 scale. Scores from 0 to 13 indicate minimum depression; 14 to 19, mild depression; 20 to 28, moderate depression, and 29 to 63, severe depression.17
The Beck Anxiety Inventory (BAI) is a 21-item measure of anxiety symptoms that uses a 0-to-3 scale to assess severity of subjective, somatic, or panic-related symptoms of anxiety. Scores ranging from 0 to 9 indicate minimal anxiety; 10 to 16, mild anxiety; 17 to 29, moderate anxiety, and 30 to 63, severe anxiety.18
The Pain Catastrophizing Scale (PCS) is a 13-item measure of pain catastrophizing, a crucial marker of how individuals experience pain. Items are scored on a 0-to-4 scale; scores of ≥ 30 indicate a clinically relevant level of catastrophizing.19
The Subjective Units of Distress Scale (SUD) is a single-item measure of the subjective intensity of disturbance or distress currently being experienced. It is scored from 0 to 10; 1 to 4 is mild, 5 to 6 is moderate, and 7 to 10 is severe.20
The Brief Pain Inventory (BPI) measures pain intensity and the impact of pain on functioning. Four items assess pain intensity at its worst, least, and average over the previous 24 hours and at the time of assessment; responses are on a 0-to-10 scale with 10 being most severe. The pain intensity measure is the average of scores on these 4 items. Pain interference is measured with respect to 7 daily activities; general activity, walking, work, mood, enjoyment of life, relations with others, and sleep. Each of these items is scored on a 0-to-10 scale with 10 being the most severe. The pain interference measure is the average of scores on these 7 items.21
Participants completed a daily pain log and recorded self-ratings (0-to-10 scale) of pain and relaxation levels before and after using the device. These scores were primarily used to assist in determining whether goals, set collaboratively by the clinician and the veteran at the first session, had been met.
Analysis
Descriptive statistics were used to characterize the sample overall and by modality. Paired t tests were used to assess changes on each assessment measure over time and for each device separately. The significance of change was assessed for 8 outcomes for each device. In this context, using a conservative Bonferroni correction, significance was set at P < .006. Because AS-M is designed to address depression, anxiety, and sleep as well as pain, whereas LTO is not, device assignments were based on clinical considerations rather than randomization. Therefore, no comparisons were made between devices, and outcomes were assessed independently for the 2 devices. Analyses were performed using SAS 9.4 (Cary, NC).
Results
Device trials were initiated for 161 veterans (LTO, 70; AS-M, 91). Distribution of devices was unequal because veterans are assigned to 1 device or the other based on clinical presentation. Failure to complete a trial (n = 46; 28.6%) typically was because of travel barriers, lack of interest in continuing, and for 3 veterans, reports of headaches that they attributed to the AS-M treatment. Of the 115 participants who completed valid trials, 88 (76.5%) also completed assessment measures at pre- and postintervention (LTO = 38; AS-M = 50). None of the participants in this study completed trials with both the AS-M and LTO devices.
Most participants were male (84.1%) and rural residents (85.5%) (Table 1).
Pain Reduction
Treatment with AS-M or LTO was associated with statistically significant reductions in pain severity (BPI), pain interference (BPI), daily pain intensity scores (daily pain log), and pain catastrophizing (PCS) (Tables 2 and 3).
Impact on Mood
Use of AS-M was associated with statistically significant improvements in depression (BDI-II), anxiety (BAI), and distress (SUD) scores. In addition, veterans completing AS-M treatment showed a statistically significant improvement in self-reported relaxation scores. Interestingly, use of LTO also resulted in a statistically significant decrease in anxiety (BAI) and a nonstatistically significant decrease in depression (BDI-II).
Figure 1 and 2 illustrates the clinical impact of each device in shifting participants from 1 level of symptom severity to another.
Discussion
Use of both AS-M and LTO at EOVAHC was associated with reduced pain intensity. The devices also had positive effects beyond pain in areas such as depression, anxiety, and distress. Remission of depression and anxiety symptoms has been associated with significant decline in pain symptoms, suggesting that pain is best treated through multimodal approaches.22
In the context of the opioid crisis, the availability of effective nonopioid, nonpharmacologic, noninvasive treatments for chronic pain is needed. The Joint Commission recently expanded its pain management guidelines to support hospitals offering nonpharmacologic pain treatments.23 Integrating AS-M, LTO, or similar products into standard pain management practices allows for other treatment pathways with positive outcomes for providers and patients. The Joint Commission also recommends an interdisciplinary approach, defined as a process whereby health care professionals from different disciplines collaborate to diagnose and treat patients experiencing difficult pain conditions. This approach facilitates multimodal management because these disciplines contribute knowledge about a variety of treatment options. Devices such AS-M and LTO are well suited to interdisciplinary pain management because they are not seen as being under the purview of a specific health care specialty.
Limitations
Our findings are limited because they are derived from a retrospective, quality improvement evaluation of outcomes from a single clinic. Findings must be considered in the context of the relatively small samples of veterans. Because analyses were conducted as part of a quality improvement effort, veterans were offered a specific device based on clinical indications, there were no comparisons between devices, and there was no comparison group. Although most participants were using medication and other treatments as part of their pain treatment plan, all reported continued pain intensity before use of a device. Analyses did not control for variation in treatments received concurrently. Last, the logs used to collect self-report data on daily pain and relaxation levels were not validated.
The data highlight a clear need for research to better understand the long-term effects of these devices as well as the characteristics of patients who respond best to each device. Noninvasive treatments for pain often are dismissed as placebos. Rigorously designed, controlled studies will help demonstrate that these devices offer a statistically significant benefit beyond any placebo effect.
Conclusion
Understanding of chronic pain and its treatment will continue to evolve. It is clear that each person dealing with chronic pain requires a tailored combination of treatments and multimodal approaches, which is more effective than any single treatment. Nonpharmacologic, noninvasive devices pose fewer risks and seem to be more effective in reducing pain intensity than traditional treatments, including medications or surgical intervention. In light of the current emphasis on evidence-based health care and as the evidence for the effectiveness of noninvasive pain devices modalities grows, it is likely that treatments incorporating modalities such as MET, CES, and LLLT will become common options for managing chronic pain.
1. US Department of Veterans Affairs. Pain as the 5th Vital Sign Toolkit. https://www.va.gov/PAINMANAGEMENT/docs/Pain_As_the_5th_Vital_Sign_Toolkit.pdf. Published October 2000. Accessed February 11, 2019.
2. Institute of Medicine (US) Committee on Advancing Pain Research, Care, and Education. Relieving Pain in America: A Blueprint for Transforming Prevention, Care, Education, and Research. Washington, DC: National Academies Press; 2011.
3. Rosenblum A, Marsch LA, Joseph H, Portenoy RK. Opioids and the treatment of chronic pain: Controversies, current status, and future directions. Exp Clin Psychopharmacol. 2008;16(5):405-416.
4. Moayedi M, Davis KD. Theories of pain: from specificity to gate control. J Neurophysiol. 2013;109(1):5-12.
5. Mosher HJ, Krebs EE, Carrel M, Kaboli PJ, Weg MW, Lund BC. Trends in prevalent and incident opioid receipt: an observational study in Veterans Health Administration 2004-2012. J Gen Intern Med. 2015;30(5):597-604.
6. Reuben DB, Alvanzo AAH, Ashikaga T, et al. National Institutes of Health Pathways to Prevention Workshop: The role of opioids in the treatment of chronic pain. Ann Intern Med. 2015;162(4):295-300.
7. Murthy VH. Opioid epidemic: we all have a role in turning the tide. https://obamawhitehouse.archives.gov/blog/2016/10/05/opioid-epidemic-we-all-have-role-turning-tide. Published October 5, 2016. Accessed February 12, 2019.
8. Kehlet H, Dahl JB. The value of “multimodal” or “balanced analgesia” in postoperative pain treatment. Anesth Analg. 1993;77(5):1048-1056.
9. Crane P, Feinberg L, Morris J. A multimodal physical therapy approach to the management of a patient with temporomandibular dysfunction and head and neck lymphedema: a case report. J Man Manip Ther. 2015;23(1): 37-42.
10. Arnstein P. Multimodal approaches to pain management. Nurs. 2011;41(3): 60-61.
11. Alpha-Stim. http://www.alpha-stim.com. Accessed March 22, 2019
12. Shekelle PG, Cook IA, Miake-Lye IM, Booth MS, Beroes JM, Mak S. Benefits and harms of cranial electrical stimulation for chronic painful conditions, depression, anxiety, and insomnia. Ann Intern Med. 2018;168(6):414-421.
13. Chow RT, Heller GZ, Barnsley L. The effect of 300 mW, 830 nm laser on chronic neck pain: a double-blind, randomized, placebo-controlled study. Pain. 2006;124(1):201-210.
14. Kulkarni AD, Smith RB. The use of microcurrent electrical therapy and cranial electrotherapy stimulation in pain control. Clin Pract Alternative Med. 2001;2(2):99-102.
15. Chow RT, Johnson MI, Lopes-Martins RA, Bjordal JM. Efficacy of low-level laser therapy in the management of neck pain: a systematic review and meta-analysis of randomised placebo or active-treatment controlled trials. Lancet. 2009;374(9705):1897-1908.
16. Taylor AG, Anderson JG, Riedel SL, et al. Cranial electrical stimulation improves symptoms and functional status in individuals with fibromyalgia. Pain Manag Nurs. 2013;14(4):327-335.
17. Beck, AT, Steer, RA, Brown, GK. Manual for the Beck Depression Inventory-II. San Antonio, TX: Psychological Corporation; 1996.
18. Beck AT, Steer RA. Beck Anxiety Inventory: Manual. San Antonio, TX: Psychological Corporation; 1993.
19. Sullivan MJL, Bishop SR, Pivik J. The pain catastrophizing scale: development and validation. Psychol Assess. 1995;7(4):524-532.
20. Wolpe J. The Practice of Behavior Therapy. 4th ed. Elmsford, NY: Pergamon; 1990.
21. Cleeland CS. The Brief Pain Inventory User Manual. https://www.mdanderson.org/research/departments-labs-institutes/departments-divisions/symptom-research/symptom-assessment-tools/brief-pain-inventory.html. Published 2009. Accessed February 12, 2019.
22. Gerrits MM, van Marwijk HW, van Oppen P, Horst HVD, Penninx BW. Longitudinal association between pain, and depression and anxiety over four years. J Psychosom Res. 2015;78(1):64-70.
23. The Joint Commission. Joint Commission enhances pain assessment and management requirements for accredited hospitals. The Joint Commission Perspectives. https://www.jointcommission.org/assets/1/18/Joint_Commission_Enhances_Pain_Assessment_and_Management_Requirements_for_Accredited_Hospitals1.PDF. Published July 2017. Accessed March 21, 2019.
Chronic pain is common among veterans treated in Veterans Health Administration (VHA) facilities, and optimal management remains challenging in the context of the national opioid misuse epidemic. The Eastern Oklahoma VA Health Care System (EOVAHCS) Pain Program offers a range of services that allow clinicians to tailor multimodal treatment strategies to a veteran’s needs. In 2014, a Modality Clinic was established to assess the utility of adding noninvasive treatment devices to the pain program’s armamentarium. This article addresses the context for introducing these devices and describes the EOVAHCS Pain Program and Modality Clinic. Also discussed are procedures and findings from an initial quality improvement evaluation designed to inform decision making regarding retention, expansion, or elimination of the EOVAHCS noninvasive, pain treatment device program.
Opioid prescriptions increased from 76 million in 1991 to 219 million in 2011. In 2011, the annual cost of chronic pain in the US was estimated at $635 billion.1-6 The confluence of an increasing concern about undertreatment of pain and overconfidence for the safety of opioids led to what former US Surgeon General Vivek H. Murthy, MD, called the opioid crisis.7 As awareness of its unintended consequences of opioid prescribing increased, the VHA began looking for nonopioid treatments that would decrease pain intensity. The 1993 article by Kehlet and Dahl was one of the first discussions of a multimodal nonpharmacologic strategy for addressing acute postoperative pain.8 Their pivotal literature review concluded that nonpharmacologic modalities, such as acupuncture, cranial manipulation, cranial electrostimulation treatment (CES), and low-level light technologies (LLLT), carried less risk and produced equal or greater clinical effects than those of drug therapies.8
Multimodal treatment approaches increasingly are encouraged, and nonopioid pain control has become more common across medical disciplines from physical therapy to anesthesiology.8-10 Innovative, noninvasive devices designed for self-use have appeared on the market. Many of these devices incorporate microcurrent electrical therapy (MET), CES, and/or LLLT (also known as cold laser).11-16 LLLT is a light modality that seems to lead to increased ATP production, resulting in improved healing and decreased inflammation.13-16 Although CES has been studied in a variety of patient populations, its effectiveness is not well understood.16 Research on the effects of CES on neurotransmitter levels as well as activation of parts of the brain involved in pain reception and transmission should clarify these mechanisms. Research has shown improvements in sleep and mood as well as overall pain reduction.11,16 Research has focused primarily on individual modalities rather than on combination devices and has been conducted on populations unlike the veteran population (eg, women with fibromyalgia).
Most of the devices that use electrical or LLLT cannot be used safely by patients who have implantable electrical devices or have medical conditions such as unstable seizures, pregnancy, and active malignancies.
The most common adverse effects (AEs) of CES—dizziness and headaches—are minimal compared with the AEs of pain medications. MET and LLLT AEs generally are limited to skin irritation and muscle soreness.11 Most devices require a prescription, and manufacturers provide training for purchase.
The Pain Program
EOVAHCS initially established its consultative pain program in 2013 to provide support, recommendations, and education about managing pain in veterans to primary care providers (PCPs). Veterans are referred to the pain program for a face-to-face assessment and set of recommendations to assist in developing a comprehensive pain treatment plan. Consistent with its multimodal, biopsychosocial rehabilitation model approach, the program also offers several chronic pain treatment services, including patient education courses, cognitive behavioral therapy (CBT) for chronic pain, chiropractic care, biofeedback, relaxation training, steroid injections, pain coaching, and a pain modality (noninvasive device) clinic. During their assessment, veterans are evaluated for the appropriateness of these programs, including treatment through the Pain Modality Clinic.
Pain Modality Clinic
The EOVAHCS Pain Modality Clinic was created in 2014 as a treatment and device-trial program to provide veterans access to newer noninvasive, patient-driven treatment devices as part of an active chronic pain self-management plan. A crucial innovation is that these devices are designed to be used by patients in their homes. These devices can be expensive, and not every patient will benefit from their use; therefore, clinic leaders recommended a trial before a device is issued to a veteran for home use.
The Pain Modality Clinic coordinator trains clinic facilitators on the device according to manufacturer’s guidelines. Each participating veteran takes part in a device trial to confirm that he or she is able to use the recommended device independently and is likely to benefit from its use. When appropriate, veterans who do not respond to the initial device trial could test the potential benefit of another device. Although data from these device trials are collected primarily to inform clinical decision making, this information also is useful in guiding local policy regarding continued support for each of the modalities.
Veterans who have chronic or persistent pain (≥ 3 months) that interferes with function or quality of life are considered good candidates for a device trial if they are actively involved in pain self-care, logistically able to participate, able to use a device long-term, and have no contraindications. “Active involvement” could be met by participation in any pain management effort, whether a specific exercise program, CBT, or other treatment.
The Modality Clinic currently offers device trials for persistent pain with Alpha-Stim-M (AS-M; Electromedical Products International, Mineral Wells, TX), Laser Touch One (LTO; Renewal Technologies, LLC, Phoenix, AZ), and Neurolumen (Oklahoma City, OK). Neurolumen devices were not available in the clinic initially and will not be discussed further in this article.
The first Alpha-Stim machine using MET and CES technology was created in 1981 for in-office pain management. In 2012, the currently used AS-M became available.11 AS-M is FDA approved for treating pain, anxiety, depression, and sleep problems and is the device used in the EOVAHCS Modality Clinic. AS-M uses probes or electrodes to send a MET waveform through the body area in pain. The device uses ear clips to provide CES, which is thought to increase alpha waves in the brain.11 The LTO is a device that combines LLLT and MET technologies in a home-use design.14 LTO is FDA approved for treating painand is a portable personal pain-relief device applied to the area of pain using electroconductive gel.
Both devices are designed for long-term, self-use, making them viable parts of a multimodal, chronic pain treatment plan. Contraindications for AS-M and LTO include having a pacemaker or an implantable defibrillator, pregnancy, current malignancy, or seizures. Eligible veterans with persistent pain and high levels of depression, anxiety, and/or sleep problems generally are triaged to AS-M, whereas those who have only pain intensity issues usually are assigned to LTO. Referral to the Modality Clinic is not limited to a specific type of pain; common pain conditions seen in the clinic are spine and joint pain, arthritis pain, myofascial pain, headaches, and neuropathy.
Training and Device Trials
Eligible veterans are educated about the device and complete clinical informed consent, which is documented in the electronic health record. The veterans’ primary care and/or specialist providers are contacted for concurrence regarding veterans’ participation in the treatment.
Protocols for the device trials are based on the manufacturers’ recommendations, adjusted to what is feasible in the clinic (manufacturers approved the changes). The number of treatments per trial varies by device. For AS-M, veterans come to the clinic 5 days a week for 2 weeks. For LTO, veterans attend the clinic 5 days a week for 1 week.
At the beginning of a device trial, a trained facilitator teaches each veteran and caregiver to use the device, sets functional goals for the trial, and provides education on the trial questionnaires and daily pain logs. The veteran then follows the device protocol in the clinic where the facilitator can respond to questions and address any issues. With support from their caregivers, veterans are expected to become independent on their device use by the end of the trial. Clinic staff or the veteran can stop the device trial at any point, without affecting the veteran’s participation in or eligibility for other EOVAHCS pain programs.
This project was submitted to the University of Oklahoma Health Sciences Center Institutional Review Board and was exempted from institutional review board oversight as a retrospective, quality improvement effort. Before data analysis, the EOVAHCS Coordinator for Research and Development reviewed the procedures to ensure that all policies were being followed.
Methods
Data for veterans who completed valid treatments of AS-M or LTO from May 9, 2014 to August 20, 2016, were included in the analyses. For an AS-M treatment to be considered valid, the veteran must have attended at least 8 sessions and completed assessment instruments at baseline (preintervention) and following completion (postintervention). For an LTO treatment to be considered valid, the veteran must have attended at least 4 sessions and completed assessment measures at baseline and after completion.
Measures
Veterans completed the following measures at baseline and after trial completion:
The Beck Depression Inventory (BDI-II) is a 21-item measure designed to assess depressive symptoms. Each item assesses intensity on a 0-to-3 scale. Scores from 0 to 13 indicate minimum depression; 14 to 19, mild depression; 20 to 28, moderate depression, and 29 to 63, severe depression.17
The Beck Anxiety Inventory (BAI) is a 21-item measure of anxiety symptoms that uses a 0-to-3 scale to assess severity of subjective, somatic, or panic-related symptoms of anxiety. Scores ranging from 0 to 9 indicate minimal anxiety; 10 to 16, mild anxiety; 17 to 29, moderate anxiety, and 30 to 63, severe anxiety.18
The Pain Catastrophizing Scale (PCS) is a 13-item measure of pain catastrophizing, a crucial marker of how individuals experience pain. Items are scored on a 0-to-4 scale; scores of ≥ 30 indicate a clinically relevant level of catastrophizing.19
The Subjective Units of Distress Scale (SUD) is a single-item measure of the subjective intensity of disturbance or distress currently being experienced. It is scored from 0 to 10; 1 to 4 is mild, 5 to 6 is moderate, and 7 to 10 is severe.20
The Brief Pain Inventory (BPI) measures pain intensity and the impact of pain on functioning. Four items assess pain intensity at its worst, least, and average over the previous 24 hours and at the time of assessment; responses are on a 0-to-10 scale with 10 being most severe. The pain intensity measure is the average of scores on these 4 items. Pain interference is measured with respect to 7 daily activities; general activity, walking, work, mood, enjoyment of life, relations with others, and sleep. Each of these items is scored on a 0-to-10 scale with 10 being the most severe. The pain interference measure is the average of scores on these 7 items.21
Participants completed a daily pain log and recorded self-ratings (0-to-10 scale) of pain and relaxation levels before and after using the device. These scores were primarily used to assist in determining whether goals, set collaboratively by the clinician and the veteran at the first session, had been met.
Analysis
Descriptive statistics were used to characterize the sample overall and by modality. Paired t tests were used to assess changes on each assessment measure over time and for each device separately. The significance of change was assessed for 8 outcomes for each device. In this context, using a conservative Bonferroni correction, significance was set at P < .006. Because AS-M is designed to address depression, anxiety, and sleep as well as pain, whereas LTO is not, device assignments were based on clinical considerations rather than randomization. Therefore, no comparisons were made between devices, and outcomes were assessed independently for the 2 devices. Analyses were performed using SAS 9.4 (Cary, NC).
Results
Device trials were initiated for 161 veterans (LTO, 70; AS-M, 91). Distribution of devices was unequal because veterans are assigned to 1 device or the other based on clinical presentation. Failure to complete a trial (n = 46; 28.6%) typically was because of travel barriers, lack of interest in continuing, and for 3 veterans, reports of headaches that they attributed to the AS-M treatment. Of the 115 participants who completed valid trials, 88 (76.5%) also completed assessment measures at pre- and postintervention (LTO = 38; AS-M = 50). None of the participants in this study completed trials with both the AS-M and LTO devices.
Most participants were male (84.1%) and rural residents (85.5%) (Table 1).
Pain Reduction
Treatment with AS-M or LTO was associated with statistically significant reductions in pain severity (BPI), pain interference (BPI), daily pain intensity scores (daily pain log), and pain catastrophizing (PCS) (Tables 2 and 3).
Impact on Mood
Use of AS-M was associated with statistically significant improvements in depression (BDI-II), anxiety (BAI), and distress (SUD) scores. In addition, veterans completing AS-M treatment showed a statistically significant improvement in self-reported relaxation scores. Interestingly, use of LTO also resulted in a statistically significant decrease in anxiety (BAI) and a nonstatistically significant decrease in depression (BDI-II).
Figure 1 and 2 illustrates the clinical impact of each device in shifting participants from 1 level of symptom severity to another.
Discussion
Use of both AS-M and LTO at EOVAHC was associated with reduced pain intensity. The devices also had positive effects beyond pain in areas such as depression, anxiety, and distress. Remission of depression and anxiety symptoms has been associated with significant decline in pain symptoms, suggesting that pain is best treated through multimodal approaches.22
In the context of the opioid crisis, the availability of effective nonopioid, nonpharmacologic, noninvasive treatments for chronic pain is needed. The Joint Commission recently expanded its pain management guidelines to support hospitals offering nonpharmacologic pain treatments.23 Integrating AS-M, LTO, or similar products into standard pain management practices allows for other treatment pathways with positive outcomes for providers and patients. The Joint Commission also recommends an interdisciplinary approach, defined as a process whereby health care professionals from different disciplines collaborate to diagnose and treat patients experiencing difficult pain conditions. This approach facilitates multimodal management because these disciplines contribute knowledge about a variety of treatment options. Devices such AS-M and LTO are well suited to interdisciplinary pain management because they are not seen as being under the purview of a specific health care specialty.
Limitations
Our findings are limited because they are derived from a retrospective, quality improvement evaluation of outcomes from a single clinic. Findings must be considered in the context of the relatively small samples of veterans. Because analyses were conducted as part of a quality improvement effort, veterans were offered a specific device based on clinical indications, there were no comparisons between devices, and there was no comparison group. Although most participants were using medication and other treatments as part of their pain treatment plan, all reported continued pain intensity before use of a device. Analyses did not control for variation in treatments received concurrently. Last, the logs used to collect self-report data on daily pain and relaxation levels were not validated.
The data highlight a clear need for research to better understand the long-term effects of these devices as well as the characteristics of patients who respond best to each device. Noninvasive treatments for pain often are dismissed as placebos. Rigorously designed, controlled studies will help demonstrate that these devices offer a statistically significant benefit beyond any placebo effect.
Conclusion
Understanding of chronic pain and its treatment will continue to evolve. It is clear that each person dealing with chronic pain requires a tailored combination of treatments and multimodal approaches, which is more effective than any single treatment. Nonpharmacologic, noninvasive devices pose fewer risks and seem to be more effective in reducing pain intensity than traditional treatments, including medications or surgical intervention. In light of the current emphasis on evidence-based health care and as the evidence for the effectiveness of noninvasive pain devices modalities grows, it is likely that treatments incorporating modalities such as MET, CES, and LLLT will become common options for managing chronic pain.
Chronic pain is common among veterans treated in Veterans Health Administration (VHA) facilities, and optimal management remains challenging in the context of the national opioid misuse epidemic. The Eastern Oklahoma VA Health Care System (EOVAHCS) Pain Program offers a range of services that allow clinicians to tailor multimodal treatment strategies to a veteran’s needs. In 2014, a Modality Clinic was established to assess the utility of adding noninvasive treatment devices to the pain program’s armamentarium. This article addresses the context for introducing these devices and describes the EOVAHCS Pain Program and Modality Clinic. Also discussed are procedures and findings from an initial quality improvement evaluation designed to inform decision making regarding retention, expansion, or elimination of the EOVAHCS noninvasive, pain treatment device program.
Opioid prescriptions increased from 76 million in 1991 to 219 million in 2011. In 2011, the annual cost of chronic pain in the US was estimated at $635 billion.1-6 The confluence of an increasing concern about undertreatment of pain and overconfidence for the safety of opioids led to what former US Surgeon General Vivek H. Murthy, MD, called the opioid crisis.7 As awareness of its unintended consequences of opioid prescribing increased, the VHA began looking for nonopioid treatments that would decrease pain intensity. The 1993 article by Kehlet and Dahl was one of the first discussions of a multimodal nonpharmacologic strategy for addressing acute postoperative pain.8 Their pivotal literature review concluded that nonpharmacologic modalities, such as acupuncture, cranial manipulation, cranial electrostimulation treatment (CES), and low-level light technologies (LLLT), carried less risk and produced equal or greater clinical effects than those of drug therapies.8
Multimodal treatment approaches increasingly are encouraged, and nonopioid pain control has become more common across medical disciplines from physical therapy to anesthesiology.8-10 Innovative, noninvasive devices designed for self-use have appeared on the market. Many of these devices incorporate microcurrent electrical therapy (MET), CES, and/or LLLT (also known as cold laser).11-16 LLLT is a light modality that seems to lead to increased ATP production, resulting in improved healing and decreased inflammation.13-16 Although CES has been studied in a variety of patient populations, its effectiveness is not well understood.16 Research on the effects of CES on neurotransmitter levels as well as activation of parts of the brain involved in pain reception and transmission should clarify these mechanisms. Research has shown improvements in sleep and mood as well as overall pain reduction.11,16 Research has focused primarily on individual modalities rather than on combination devices and has been conducted on populations unlike the veteran population (eg, women with fibromyalgia).
Most of the devices that use electrical or LLLT cannot be used safely by patients who have implantable electrical devices or have medical conditions such as unstable seizures, pregnancy, and active malignancies.
The most common adverse effects (AEs) of CES—dizziness and headaches—are minimal compared with the AEs of pain medications. MET and LLLT AEs generally are limited to skin irritation and muscle soreness.11 Most devices require a prescription, and manufacturers provide training for purchase.
The Pain Program
EOVAHCS initially established its consultative pain program in 2013 to provide support, recommendations, and education about managing pain in veterans to primary care providers (PCPs). Veterans are referred to the pain program for a face-to-face assessment and set of recommendations to assist in developing a comprehensive pain treatment plan. Consistent with its multimodal, biopsychosocial rehabilitation model approach, the program also offers several chronic pain treatment services, including patient education courses, cognitive behavioral therapy (CBT) for chronic pain, chiropractic care, biofeedback, relaxation training, steroid injections, pain coaching, and a pain modality (noninvasive device) clinic. During their assessment, veterans are evaluated for the appropriateness of these programs, including treatment through the Pain Modality Clinic.
Pain Modality Clinic
The EOVAHCS Pain Modality Clinic was created in 2014 as a treatment and device-trial program to provide veterans access to newer noninvasive, patient-driven treatment devices as part of an active chronic pain self-management plan. A crucial innovation is that these devices are designed to be used by patients in their homes. These devices can be expensive, and not every patient will benefit from their use; therefore, clinic leaders recommended a trial before a device is issued to a veteran for home use.
The Pain Modality Clinic coordinator trains clinic facilitators on the device according to manufacturer’s guidelines. Each participating veteran takes part in a device trial to confirm that he or she is able to use the recommended device independently and is likely to benefit from its use. When appropriate, veterans who do not respond to the initial device trial could test the potential benefit of another device. Although data from these device trials are collected primarily to inform clinical decision making, this information also is useful in guiding local policy regarding continued support for each of the modalities.
Veterans who have chronic or persistent pain (≥ 3 months) that interferes with function or quality of life are considered good candidates for a device trial if they are actively involved in pain self-care, logistically able to participate, able to use a device long-term, and have no contraindications. “Active involvement” could be met by participation in any pain management effort, whether a specific exercise program, CBT, or other treatment.
The Modality Clinic currently offers device trials for persistent pain with Alpha-Stim-M (AS-M; Electromedical Products International, Mineral Wells, TX), Laser Touch One (LTO; Renewal Technologies, LLC, Phoenix, AZ), and Neurolumen (Oklahoma City, OK). Neurolumen devices were not available in the clinic initially and will not be discussed further in this article.
The first Alpha-Stim machine using MET and CES technology was created in 1981 for in-office pain management. In 2012, the currently used AS-M became available.11 AS-M is FDA approved for treating pain, anxiety, depression, and sleep problems and is the device used in the EOVAHCS Modality Clinic. AS-M uses probes or electrodes to send a MET waveform through the body area in pain. The device uses ear clips to provide CES, which is thought to increase alpha waves in the brain.11 The LTO is a device that combines LLLT and MET technologies in a home-use design.14 LTO is FDA approved for treating painand is a portable personal pain-relief device applied to the area of pain using electroconductive gel.
Both devices are designed for long-term, self-use, making them viable parts of a multimodal, chronic pain treatment plan. Contraindications for AS-M and LTO include having a pacemaker or an implantable defibrillator, pregnancy, current malignancy, or seizures. Eligible veterans with persistent pain and high levels of depression, anxiety, and/or sleep problems generally are triaged to AS-M, whereas those who have only pain intensity issues usually are assigned to LTO. Referral to the Modality Clinic is not limited to a specific type of pain; common pain conditions seen in the clinic are spine and joint pain, arthritis pain, myofascial pain, headaches, and neuropathy.
Training and Device Trials
Eligible veterans are educated about the device and complete clinical informed consent, which is documented in the electronic health record. The veterans’ primary care and/or specialist providers are contacted for concurrence regarding veterans’ participation in the treatment.
Protocols for the device trials are based on the manufacturers’ recommendations, adjusted to what is feasible in the clinic (manufacturers approved the changes). The number of treatments per trial varies by device. For AS-M, veterans come to the clinic 5 days a week for 2 weeks. For LTO, veterans attend the clinic 5 days a week for 1 week.
At the beginning of a device trial, a trained facilitator teaches each veteran and caregiver to use the device, sets functional goals for the trial, and provides education on the trial questionnaires and daily pain logs. The veteran then follows the device protocol in the clinic where the facilitator can respond to questions and address any issues. With support from their caregivers, veterans are expected to become independent on their device use by the end of the trial. Clinic staff or the veteran can stop the device trial at any point, without affecting the veteran’s participation in or eligibility for other EOVAHCS pain programs.
This project was submitted to the University of Oklahoma Health Sciences Center Institutional Review Board and was exempted from institutional review board oversight as a retrospective, quality improvement effort. Before data analysis, the EOVAHCS Coordinator for Research and Development reviewed the procedures to ensure that all policies were being followed.
Methods
Data for veterans who completed valid treatments of AS-M or LTO from May 9, 2014 to August 20, 2016, were included in the analyses. For an AS-M treatment to be considered valid, the veteran must have attended at least 8 sessions and completed assessment instruments at baseline (preintervention) and following completion (postintervention). For an LTO treatment to be considered valid, the veteran must have attended at least 4 sessions and completed assessment measures at baseline and after completion.
Measures
Veterans completed the following measures at baseline and after trial completion:
The Beck Depression Inventory (BDI-II) is a 21-item measure designed to assess depressive symptoms. Each item assesses intensity on a 0-to-3 scale. Scores from 0 to 13 indicate minimum depression; 14 to 19, mild depression; 20 to 28, moderate depression, and 29 to 63, severe depression.17
The Beck Anxiety Inventory (BAI) is a 21-item measure of anxiety symptoms that uses a 0-to-3 scale to assess severity of subjective, somatic, or panic-related symptoms of anxiety. Scores ranging from 0 to 9 indicate minimal anxiety; 10 to 16, mild anxiety; 17 to 29, moderate anxiety, and 30 to 63, severe anxiety.18
The Pain Catastrophizing Scale (PCS) is a 13-item measure of pain catastrophizing, a crucial marker of how individuals experience pain. Items are scored on a 0-to-4 scale; scores of ≥ 30 indicate a clinically relevant level of catastrophizing.19
The Subjective Units of Distress Scale (SUD) is a single-item measure of the subjective intensity of disturbance or distress currently being experienced. It is scored from 0 to 10; 1 to 4 is mild, 5 to 6 is moderate, and 7 to 10 is severe.20
The Brief Pain Inventory (BPI) measures pain intensity and the impact of pain on functioning. Four items assess pain intensity at its worst, least, and average over the previous 24 hours and at the time of assessment; responses are on a 0-to-10 scale with 10 being most severe. The pain intensity measure is the average of scores on these 4 items. Pain interference is measured with respect to 7 daily activities; general activity, walking, work, mood, enjoyment of life, relations with others, and sleep. Each of these items is scored on a 0-to-10 scale with 10 being the most severe. The pain interference measure is the average of scores on these 7 items.21
Participants completed a daily pain log and recorded self-ratings (0-to-10 scale) of pain and relaxation levels before and after using the device. These scores were primarily used to assist in determining whether goals, set collaboratively by the clinician and the veteran at the first session, had been met.
Analysis
Descriptive statistics were used to characterize the sample overall and by modality. Paired t tests were used to assess changes on each assessment measure over time and for each device separately. The significance of change was assessed for 8 outcomes for each device. In this context, using a conservative Bonferroni correction, significance was set at P < .006. Because AS-M is designed to address depression, anxiety, and sleep as well as pain, whereas LTO is not, device assignments were based on clinical considerations rather than randomization. Therefore, no comparisons were made between devices, and outcomes were assessed independently for the 2 devices. Analyses were performed using SAS 9.4 (Cary, NC).
Results
Device trials were initiated for 161 veterans (LTO, 70; AS-M, 91). Distribution of devices was unequal because veterans are assigned to 1 device or the other based on clinical presentation. Failure to complete a trial (n = 46; 28.6%) typically was because of travel barriers, lack of interest in continuing, and for 3 veterans, reports of headaches that they attributed to the AS-M treatment. Of the 115 participants who completed valid trials, 88 (76.5%) also completed assessment measures at pre- and postintervention (LTO = 38; AS-M = 50). None of the participants in this study completed trials with both the AS-M and LTO devices.
Most participants were male (84.1%) and rural residents (85.5%) (Table 1).
Pain Reduction
Treatment with AS-M or LTO was associated with statistically significant reductions in pain severity (BPI), pain interference (BPI), daily pain intensity scores (daily pain log), and pain catastrophizing (PCS) (Tables 2 and 3).
Impact on Mood
Use of AS-M was associated with statistically significant improvements in depression (BDI-II), anxiety (BAI), and distress (SUD) scores. In addition, veterans completing AS-M treatment showed a statistically significant improvement in self-reported relaxation scores. Interestingly, use of LTO also resulted in a statistically significant decrease in anxiety (BAI) and a nonstatistically significant decrease in depression (BDI-II).
Figure 1 and 2 illustrates the clinical impact of each device in shifting participants from 1 level of symptom severity to another.
Discussion
Use of both AS-M and LTO at EOVAHC was associated with reduced pain intensity. The devices also had positive effects beyond pain in areas such as depression, anxiety, and distress. Remission of depression and anxiety symptoms has been associated with significant decline in pain symptoms, suggesting that pain is best treated through multimodal approaches.22
In the context of the opioid crisis, the availability of effective nonopioid, nonpharmacologic, noninvasive treatments for chronic pain is needed. The Joint Commission recently expanded its pain management guidelines to support hospitals offering nonpharmacologic pain treatments.23 Integrating AS-M, LTO, or similar products into standard pain management practices allows for other treatment pathways with positive outcomes for providers and patients. The Joint Commission also recommends an interdisciplinary approach, defined as a process whereby health care professionals from different disciplines collaborate to diagnose and treat patients experiencing difficult pain conditions. This approach facilitates multimodal management because these disciplines contribute knowledge about a variety of treatment options. Devices such AS-M and LTO are well suited to interdisciplinary pain management because they are not seen as being under the purview of a specific health care specialty.
Limitations
Our findings are limited because they are derived from a retrospective, quality improvement evaluation of outcomes from a single clinic. Findings must be considered in the context of the relatively small samples of veterans. Because analyses were conducted as part of a quality improvement effort, veterans were offered a specific device based on clinical indications, there were no comparisons between devices, and there was no comparison group. Although most participants were using medication and other treatments as part of their pain treatment plan, all reported continued pain intensity before use of a device. Analyses did not control for variation in treatments received concurrently. Last, the logs used to collect self-report data on daily pain and relaxation levels were not validated.
The data highlight a clear need for research to better understand the long-term effects of these devices as well as the characteristics of patients who respond best to each device. Noninvasive treatments for pain often are dismissed as placebos. Rigorously designed, controlled studies will help demonstrate that these devices offer a statistically significant benefit beyond any placebo effect.
Conclusion
Understanding of chronic pain and its treatment will continue to evolve. It is clear that each person dealing with chronic pain requires a tailored combination of treatments and multimodal approaches, which is more effective than any single treatment. Nonpharmacologic, noninvasive devices pose fewer risks and seem to be more effective in reducing pain intensity than traditional treatments, including medications or surgical intervention. In light of the current emphasis on evidence-based health care and as the evidence for the effectiveness of noninvasive pain devices modalities grows, it is likely that treatments incorporating modalities such as MET, CES, and LLLT will become common options for managing chronic pain.
1. US Department of Veterans Affairs. Pain as the 5th Vital Sign Toolkit. https://www.va.gov/PAINMANAGEMENT/docs/Pain_As_the_5th_Vital_Sign_Toolkit.pdf. Published October 2000. Accessed February 11, 2019.
2. Institute of Medicine (US) Committee on Advancing Pain Research, Care, and Education. Relieving Pain in America: A Blueprint for Transforming Prevention, Care, Education, and Research. Washington, DC: National Academies Press; 2011.
3. Rosenblum A, Marsch LA, Joseph H, Portenoy RK. Opioids and the treatment of chronic pain: Controversies, current status, and future directions. Exp Clin Psychopharmacol. 2008;16(5):405-416.
4. Moayedi M, Davis KD. Theories of pain: from specificity to gate control. J Neurophysiol. 2013;109(1):5-12.
5. Mosher HJ, Krebs EE, Carrel M, Kaboli PJ, Weg MW, Lund BC. Trends in prevalent and incident opioid receipt: an observational study in Veterans Health Administration 2004-2012. J Gen Intern Med. 2015;30(5):597-604.
6. Reuben DB, Alvanzo AAH, Ashikaga T, et al. National Institutes of Health Pathways to Prevention Workshop: The role of opioids in the treatment of chronic pain. Ann Intern Med. 2015;162(4):295-300.
7. Murthy VH. Opioid epidemic: we all have a role in turning the tide. https://obamawhitehouse.archives.gov/blog/2016/10/05/opioid-epidemic-we-all-have-role-turning-tide. Published October 5, 2016. Accessed February 12, 2019.
8. Kehlet H, Dahl JB. The value of “multimodal” or “balanced analgesia” in postoperative pain treatment. Anesth Analg. 1993;77(5):1048-1056.
9. Crane P, Feinberg L, Morris J. A multimodal physical therapy approach to the management of a patient with temporomandibular dysfunction and head and neck lymphedema: a case report. J Man Manip Ther. 2015;23(1): 37-42.
10. Arnstein P. Multimodal approaches to pain management. Nurs. 2011;41(3): 60-61.
11. Alpha-Stim. http://www.alpha-stim.com. Accessed March 22, 2019
12. Shekelle PG, Cook IA, Miake-Lye IM, Booth MS, Beroes JM, Mak S. Benefits and harms of cranial electrical stimulation for chronic painful conditions, depression, anxiety, and insomnia. Ann Intern Med. 2018;168(6):414-421.
13. Chow RT, Heller GZ, Barnsley L. The effect of 300 mW, 830 nm laser on chronic neck pain: a double-blind, randomized, placebo-controlled study. Pain. 2006;124(1):201-210.
14. Kulkarni AD, Smith RB. The use of microcurrent electrical therapy and cranial electrotherapy stimulation in pain control. Clin Pract Alternative Med. 2001;2(2):99-102.
15. Chow RT, Johnson MI, Lopes-Martins RA, Bjordal JM. Efficacy of low-level laser therapy in the management of neck pain: a systematic review and meta-analysis of randomised placebo or active-treatment controlled trials. Lancet. 2009;374(9705):1897-1908.
16. Taylor AG, Anderson JG, Riedel SL, et al. Cranial electrical stimulation improves symptoms and functional status in individuals with fibromyalgia. Pain Manag Nurs. 2013;14(4):327-335.
17. Beck, AT, Steer, RA, Brown, GK. Manual for the Beck Depression Inventory-II. San Antonio, TX: Psychological Corporation; 1996.
18. Beck AT, Steer RA. Beck Anxiety Inventory: Manual. San Antonio, TX: Psychological Corporation; 1993.
19. Sullivan MJL, Bishop SR, Pivik J. The pain catastrophizing scale: development and validation. Psychol Assess. 1995;7(4):524-532.
20. Wolpe J. The Practice of Behavior Therapy. 4th ed. Elmsford, NY: Pergamon; 1990.
21. Cleeland CS. The Brief Pain Inventory User Manual. https://www.mdanderson.org/research/departments-labs-institutes/departments-divisions/symptom-research/symptom-assessment-tools/brief-pain-inventory.html. Published 2009. Accessed February 12, 2019.
22. Gerrits MM, van Marwijk HW, van Oppen P, Horst HVD, Penninx BW. Longitudinal association between pain, and depression and anxiety over four years. J Psychosom Res. 2015;78(1):64-70.
23. The Joint Commission. Joint Commission enhances pain assessment and management requirements for accredited hospitals. The Joint Commission Perspectives. https://www.jointcommission.org/assets/1/18/Joint_Commission_Enhances_Pain_Assessment_and_Management_Requirements_for_Accredited_Hospitals1.PDF. Published July 2017. Accessed March 21, 2019.
1. US Department of Veterans Affairs. Pain as the 5th Vital Sign Toolkit. https://www.va.gov/PAINMANAGEMENT/docs/Pain_As_the_5th_Vital_Sign_Toolkit.pdf. Published October 2000. Accessed February 11, 2019.
2. Institute of Medicine (US) Committee on Advancing Pain Research, Care, and Education. Relieving Pain in America: A Blueprint for Transforming Prevention, Care, Education, and Research. Washington, DC: National Academies Press; 2011.
3. Rosenblum A, Marsch LA, Joseph H, Portenoy RK. Opioids and the treatment of chronic pain: Controversies, current status, and future directions. Exp Clin Psychopharmacol. 2008;16(5):405-416.
4. Moayedi M, Davis KD. Theories of pain: from specificity to gate control. J Neurophysiol. 2013;109(1):5-12.
5. Mosher HJ, Krebs EE, Carrel M, Kaboli PJ, Weg MW, Lund BC. Trends in prevalent and incident opioid receipt: an observational study in Veterans Health Administration 2004-2012. J Gen Intern Med. 2015;30(5):597-604.
6. Reuben DB, Alvanzo AAH, Ashikaga T, et al. National Institutes of Health Pathways to Prevention Workshop: The role of opioids in the treatment of chronic pain. Ann Intern Med. 2015;162(4):295-300.
7. Murthy VH. Opioid epidemic: we all have a role in turning the tide. https://obamawhitehouse.archives.gov/blog/2016/10/05/opioid-epidemic-we-all-have-role-turning-tide. Published October 5, 2016. Accessed February 12, 2019.
8. Kehlet H, Dahl JB. The value of “multimodal” or “balanced analgesia” in postoperative pain treatment. Anesth Analg. 1993;77(5):1048-1056.
9. Crane P, Feinberg L, Morris J. A multimodal physical therapy approach to the management of a patient with temporomandibular dysfunction and head and neck lymphedema: a case report. J Man Manip Ther. 2015;23(1): 37-42.
10. Arnstein P. Multimodal approaches to pain management. Nurs. 2011;41(3): 60-61.
11. Alpha-Stim. http://www.alpha-stim.com. Accessed March 22, 2019
12. Shekelle PG, Cook IA, Miake-Lye IM, Booth MS, Beroes JM, Mak S. Benefits and harms of cranial electrical stimulation for chronic painful conditions, depression, anxiety, and insomnia. Ann Intern Med. 2018;168(6):414-421.
13. Chow RT, Heller GZ, Barnsley L. The effect of 300 mW, 830 nm laser on chronic neck pain: a double-blind, randomized, placebo-controlled study. Pain. 2006;124(1):201-210.
14. Kulkarni AD, Smith RB. The use of microcurrent electrical therapy and cranial electrotherapy stimulation in pain control. Clin Pract Alternative Med. 2001;2(2):99-102.
15. Chow RT, Johnson MI, Lopes-Martins RA, Bjordal JM. Efficacy of low-level laser therapy in the management of neck pain: a systematic review and meta-analysis of randomised placebo or active-treatment controlled trials. Lancet. 2009;374(9705):1897-1908.
16. Taylor AG, Anderson JG, Riedel SL, et al. Cranial electrical stimulation improves symptoms and functional status in individuals with fibromyalgia. Pain Manag Nurs. 2013;14(4):327-335.
17. Beck, AT, Steer, RA, Brown, GK. Manual for the Beck Depression Inventory-II. San Antonio, TX: Psychological Corporation; 1996.
18. Beck AT, Steer RA. Beck Anxiety Inventory: Manual. San Antonio, TX: Psychological Corporation; 1993.
19. Sullivan MJL, Bishop SR, Pivik J. The pain catastrophizing scale: development and validation. Psychol Assess. 1995;7(4):524-532.
20. Wolpe J. The Practice of Behavior Therapy. 4th ed. Elmsford, NY: Pergamon; 1990.
21. Cleeland CS. The Brief Pain Inventory User Manual. https://www.mdanderson.org/research/departments-labs-institutes/departments-divisions/symptom-research/symptom-assessment-tools/brief-pain-inventory.html. Published 2009. Accessed February 12, 2019.
22. Gerrits MM, van Marwijk HW, van Oppen P, Horst HVD, Penninx BW. Longitudinal association between pain, and depression and anxiety over four years. J Psychosom Res. 2015;78(1):64-70.
23. The Joint Commission. Joint Commission enhances pain assessment and management requirements for accredited hospitals. The Joint Commission Perspectives. https://www.jointcommission.org/assets/1/18/Joint_Commission_Enhances_Pain_Assessment_and_Management_Requirements_for_Accredited_Hospitals1.PDF. Published July 2017. Accessed March 21, 2019.
Providing Rural Veterans With Access to Exercise Through Gerofit
Clinical video telehealth can be used to deliver functional circuit exercise training to older veterans in remote locations.
Exercise increases endurance, muscle strength, and functional performance with corresponding gains in mobility, survival, and quality of life.1 However, even with these benefits and improvements in clinical outcomes, only 15% of adults aged ≥ 65 years follow current guidelines for exercise.2 Despite their prior military training, the majority of veterans do not meet physical activity recommendations.3 Time, travel, and support are common barriers to exercise participation and adherence—barriers that are further amplified among older adults.
The Veterans Health Administration (VHA) is recognized as a world leader in telehealth service development. Currently, 677,000 veterans have received telehealth services, which represents 12% of the 5.6 million veterans under VHA care.4 Clinical video telehealth (CVT) is widely used within the VHA system to deliver health care that otherwise would not be available to veterans. Veterans who have difficulty traveling to the nearest US Department of Veteran Affairs (VA) medical center (VAMC) can access CVT programs at a participating VHA community-based outpatient clinic (CBOC). The VA has more than 45 CVT programs, including programs for mental health, weight management, cardiology, and dermatology. Outside the VA, cardiac exercise rehabilitation provided by CVT has been shown to be as effective as center-based programs in improving cardiovascular risk factors and functional capacity.5 A VHA exercise program that leveraged CVT resources and was dedicated to older adults with a wide range of comorbid conditions would have a high impact on the health and well-being of older veterans.
Gerofit is a VHA clinical demonstration program of supervised center-based exercise for veterans aged ≥ 65 years. Developed at the Durham VAMC Geriatric Research, Education, and Clinical Center (GRECC) in North Carolina, it has demonstrated improved clinical outcomes, including physical function, mobility, quality of life, and survival.6-10 The program offers veterans individualized exercise in a group setting that focuses on improving endurance, strength, and balance. The exercise prescription is based on the patient’s physical limitations as identified in a physical performance assessment.
With support from VHA Geriatric Extended Care (GEC) and the Office of Rural Health (ORH), Gerofit was implemented in 10 VAMCs across 8 VISNs. However, barriers such as travel time, distance, and transportation limit participation. Previously, we found that rural veterans lack access to exercise programs.11,12 Although some do aerobic exercise (AEX), most do not do resistance training (RT), though they are willing to learn. Access to Gerofit for rural veterans is expanding with recent support from the ORH Enterprise Wide Initiative. Rural program expansion includes several different Gerofit initiatives, many involving CBOCs.
The Salem VAMC Gerofit program sought to adapt the facility-based assessment and exercise procedures into a self-reliant CVT class for its CBOCs. This article describes the development of the Salem VAMC Gerofit CVT program, hereafter referred to as Tele-Gerofit.
Related: Expanding the Scope of Telemedicine in Gastroenterology
Program Design
Gerofit was established in 1986 at the Durham GRECC as an exercise and health promotion program for veterans aged ≥ 65 years.13 Its goal is to prevent or improve functional decline from physical inactivity and age-related conditions. Gerofit targets the geriatric patient population and thus extends beyond cardiac and pulmonary rehabilitation or weight loss programs. The primary exclusion criteria are based on safety issues in the context of a group exercise setting of older adults and include oxygen dependency, unstable cardiac disease, and moderate-to-severe cognitive impairment.
To participate in Gerofit, veterans must be able to perform activities of daily living and self-manage an exercise prescription developed by the exercise instructor based on physical performance testing. These physical performance tests include measures that are independent predictors of disability, loss of independent living, and death, as well as surrogate measures of exercise capacity (eg, strength, endurance, balance).14,15 A novel aspect of Gerofit is that the physical performance assessment is used not only to determine physical limitations, but also to individualize the exercise prescription based on the observed deficits in strength, endurance, or balance. These assessments are performed at initial enrollment; 3 months, 6 months, and 1 year later; and annually after that. Currently, the center-based Gerofit programs administer 5 items of the Senior Fitness Test: 6-minute walk, 10-meter walk (10-MWT), 30-second 1-arm curl, 30-second chair-stand test, and 8-foot up-and-go.15 The side-by-side, semitandem, and tandem standing balance tests from the short physical performance battery also are performed.16 In addition, participants complete a questionnaire that includes items from the physical functioning scale of the 12-Item Short Form Health Survey (SF-12).
After each assessment, the Gerofit exercise instructor reviews the results with the veteran and formulates an individualized exercise prescription along with goals for improvement. Veterans are encouraged to attend supervised center-based exercise sessions 3 times weekly. Classes are offered in a gym or fitness center at the VAMC or in leased space. Each patient uses a cue card that lists an exercise plan personalized for intensity and duration for aerobic exercise (AEX; eg, treadmill walking, stationary bicycling, arm ergometry), RT using dumbbells and weight equipment, and functional exercises for flexibility and balance. Some medical centers also offer yoga, tai chi, or dancing Gerofit classes.
For participants in the Durham Gerofit program, mortality decreased 25% over a decade (hazard ratio, 0.75; 95% CI, 0.61-0.91).9 A substudy that included the Psychological General Well-Being Index found that 81% of participants significantly increased their score after 1 year.7 Observed initial improvement in physical performance has been sustained over 5 years.10,17 One-year results from the recent Gerofit expansion to 6 other VAMCs showed clinically and statistically significantly improved physical performance from baseline to 3-, 6-, and 12-month follow-up.18
Adaptation of Gerofit to CVT Delivery
Initial work. The Greater Los Angeles VAMC Gerofit program conducted a pilot CVT exercise class of 6 veterans at the rural Bakersfield CBOC in California.19 Each week, an exercise instructor broadcast a 60-minute exercise class that included warm-up, RT with bands, progressive balance training, and flexibility. Trained student volunteers from California State University in Bakersfield kinesiology program were on site at the Bakersfield CBOC to perform the assessments and aid in exercises during the CVT sessions. Despite the lack of AEX per se, veterans showed significant improvement in endurance as measured by an increase in the number of steps completed in 2 minutes at the 3-month assessment (P = .049). Although exercises were not delivered in a circuit format, the improved endurance supported the potential for cardiovascular benefit from RT in older adults.
This pilot project also demonstrated that key components of the Gerofit program could be delivered safely by telehealth with onsite supervision. The Miami VA Healthcare System also offers CVT Gerofit exercise classes broadcast to the rural Florida CBOCs of Key Largo and Homestead.11 The exercise activities offered for the Miami CVT participants incorporate components of AEX (calisthenics) and RT (resistance bands). Veterans enjoyed the classes, and adherence was good. However, availability of staff and space are an ongoing challenge.
In Key Largo, 5 veterans participated before the CVT classes were placed on hold owing to the demands of other CVT programs and limited availability of the telehealth clinical technician (TCT). The Homestead CBOC continues to offer CVT Gerofit exercise classes and has 6 regular participants. Notably, the physical space at the Homestead CBOC is smaller than that at the Key Largo CBOC; the Homestead CBOC has adjusted by shifting to exercises performed while standing or sitting, ensuring participants’ safety and satisfaction.
The Baltimore, Maryland VAMC Gerofit program offers other innovative CVT exercise classes, including a tai chi class, and a class with exercise performed while sitting in a chair. Although the Baltimore VAMC CVT exercise classes do not have the scope of the center-based exercise prescriptions, they are unique in that they are broadcast not only to their affiliated CBOCs, but also other Gerofit programs in different VISNs.
Related: Telehealth for Rural Veterans With Neurologic Disorders
Salem VAMC Gerofit Program. The center-based Salem VAMC Gerofit program was established in July 2015. In fiscal year 2017, its dedicated exercise facility had more than 5,000 patient visits. Despite the program’s success, we prioritized establishing Tele-Gerofit because of the medical center’s rural location in southwest Virginia and the large number of veterans who receive care at CBOCs. Therefore, much as with the pilot CVT Gerofit classes in Los Angeles and Miami, the target setting was rural CBOCs. The goal for Salem VAMC Tele-Gerofit was to modify Gerofit delivery to the CVT format and a CBOC setting with minimal modification of the content and personnel requirements of both physical performance testing and exercise training procedures.
Adjustments for CBOC Setting. The enrollment process for Tele-Gerofit is the same as that for the center-based program. To start, a veteran’s primary care provider reviews the list of eligibility criteria and, if the veteran qualifies, places a consult. A Gerofit team member then contacts the veteran by phone to describe the program and schedule an assessment. At the baseline physical performance assessment, American College of Sports Medicine guidelines on exercise participation, health screening, and exercise intensity are used to evaluate veterans and rank them by their cardiovascular risk.20 All new program participants start with low-intensity exercise and gradually progress to recommended levels of exercise. Before starting an exercise class, participants are instructed on use of the 10-point rating of perceived exertion (RPE).
Each CBOC site is supplied with an RPE poster that is displayed for participants’ use. During a Tele-Gerofit class, the exercise instructor asks participants to periodically report their RPE. This class differs slightly from the center-based exercise sessions in which RPE is primarily assessed when a different exercise is introduced or the duration or intensity of an exercise is increased. The Gerofit instructor monitors exercise and treatment fidelity, but the onsite TCT observes for safety during class. The TCT also takes initial vital signs and sets up the room for the class. Emergency contacts and procedures are posted in each CBOC CVT room and are available to the center-based exercise instructor. Because the CBOCs are not inside medical facilities, some CBOC directors have asked that heart rate monitors be used as an extra safety precaution to ensure that high-risk participants do not exceed a heart rate limit that may be set by their cardiologists.
Modifications to Physical Performance Assessment. Physical performance testing had to be adapted to the small rooms available at the CBOCs. For measuring normal gait speed, the 10-MWT was replaced with the 4-meter walk test (4-MWT). The 4-MWT has excellent test–retest reliability with an intraclass correlation coefficient (ICC) of 0.93, but the discrepancy in gait speed between the 4-MWT and the 10-MWT is such that the tests cannot be used interchangeably.21 For measuring endurance, the 6-minute walk test was replaced with the 2-minute step test (2-MST). In older adults, the 2-MST has a moderate correlation with 6-minute walk distance (r = 0.36; P = .04) and high reliability (ICC, 0.90).15,22 The 30-second 1-arm curl, the 30-second chair-stand test, and the 8-foot up-and-go test are performed without modification and require only dumbbells, a chair without wheels, and a stopwatch.
The exercise instructor at the Salem VAMC conducts physical performance testing by 2-way videoconferencing with the veteran in a room at the CBOC. The TCT at the CBOC assists by measuring and demarcating 4 meters on the floor and a designated height on the wall for knee elevation for 4-MWT and 2-MST, respectively. The TCT remains in the room during the assessment visit. Except for taking vital signs before and after the physical performance assessment, the TCT does not participate in the testing. To date, more than 20 physical performance assessments have been conducted without difficulty at Salem-affiliated CBOCs. The primary challenge has been scheduling the room with CVT equipment (ie, camera and screen) for the 30-minute individual assessment session, which occurs on a rolling basis as individuals are enrolled and followed.
After the assessment is completed, the exercise instructor reviews the results with the participant and provides feedback on areas in need of improvement. However, these education sessions can be lengthy and are best supported by giving the patient a personalized handout.
Functional Circuit Exercise. In Tele-Gerofit, exercise training is delivered by CVT broadcast from the Salem VAMC to veterans in a room (equipped with steps, dumbbells, chairs, and bands) at the CBOC. This type of exercise training, which uses only mobile equipment and plyometric (weight-bearing) exercises, is referred to as functional exercise. The AEX includes marching in place, moving on and off a raised step, and body-weight exercises, while RT uses dumbbells, resistance bands, and plyometric exercises (Table 2).
Progression of intensity is achieved by increasing the rate of stepping and the size of the steps (AEX) or the number of repetitions and the weight of the dumbbells or bands (RT). Each veteran exercises at an intensity level that is appropriate for his or her baseline limitations and medical conditions. The exercise instructor uses different forms of the same equipment (eg, heavier dumbbells, higher steps) to vary intensity among individuals while having them perform the same exercises as a group. The challenge is to adjust the pace of the AEX or the timing of the RT repetitions for individuals new to the class.
Delivery of exercise training in the form of circuits allows for a diverse exercise program in a setting with limited space. Circuit training is an exercise modality that consists of a series of different exercises, each usually completed in 30 to 60 seconds, with minimal rest between each type of exercise. Each Tele-Gerofit circuit has a mix of AEX and RT exercises performed for 3 minutes consecutively (Figure).
The design of the circuit training can be adjusted based on the number of individuals in the class. Larger classes can be split into 2 groups that alternate between exercise sets, while smaller classes have 1 group performing the same exercise set and then rotating to either the AEX or RT set. Total exercise time to complete the circuit depends on the number of different exercises, number of repetitions, and the rest between repetitions and the different exercises. In this way, total exercise time can be made shorter or longer depending on the veteran’s capacity.
Frequency. Tele-Gerofit exercise classes are currently offered twice weekly and last about 1 hour, which includes warm-up (8-10 minutes), functional circuit training (40 minutes), and cooldown/stretching (8-10 minutes). A challenge for the exercise instructor is the need to provide ongoing clear instructions both to the class and to individuals as needed. As the exercise prescription for each patient is based on physical performance testing, the exercise instructor for the training must be familiar with the test results. Derivation of the exercise prescription in Tele-Gerofit follows the same process as center-based Gerofit.
Each patient is given an exercise prescription written to address any impairments noted in the different domains of the physical performance assessment, scored using age and sex percentiles. For instance, individuals scoring poorly on lower body strength are given specific lower body strengthening exercises. Participants are given an exercise program that guides them toward achieving recommended physical activity guidelines using their RPE to modulate each exercise. Duration and intensity of each type of planned exercise are formally discussed after initial and follow-up assessments. In addition, exercise training is informally progressed throughout the program. For Tele-Gerofit, instructors must design each class with the group in mind while being prepared for modifications and specific changes for individuals.
Discussion
Tele-Gerofit adapts the well-established center-based Gerofit program to be executed without an exercise facility while maintaining the content of the evidence-based procedures. Physical performance testing and exercise training were modified, adding elements necessary for CVT assessments and classes to be broadcast from the Salem VAMC to its affiliated CBOCs. Tele-Gerofit exercises are performed in a circuit style that allows a veteran or small structured groups of veterans to move among exercises and requires less space than traditional group exercise does. Safety and monitoring concerns are addressed with a safety procedure that includes emergency plans for each site, prescreening of enrolled participants, and monitoring of exercise intensity in accordance with national guidelines.1 Similar to the center-based Gerofit program, the exercise prescription is tailored to each veteran’s physical limitations based on initial and ongoing assessment of physical performance. Tele-Gerofit physical performance testing fulfills the same need with only a few modifications using validated measures. Tele-Gerofit assessments are administered by CVT without the need for additional staff on site.
Adaptation of center-based Gerofit exercise classes to Tele-Gerofit is a major innovation. Use of a circuit exercise design was supported by findings in older adults that RT alone, when performed quickly with minimal rest between each set and exercise station, increases both aerobic capacity and strength.23,24 Older adult RT trials that compared circuit RT with traditional RT found that strength gains are comparable between circuit and traditional RT.24-26 Working with adults aged > 60 years, Takeshima and colleagues conducted a trial of circuit exercise with added callisthenic exercises performed in place between RT on exercise machines.27 This dual-modality (AEX+RT) circuit approach was well tolerated and effective, increasing aerobic capacity and strength. Unfortunately, the resistance exercise machines used in those circuit exercise studies and in the center-based Gerofit program are not an option for Tele-Gerofit.
The requirement for an exercise facility was removed by designing Tele-Gerofit exercise to include only functional exercises that rely on body weight or small mobile exercise equipment. Although popular among young adults, functional circuit exercise is understudied in older adults. Recently, a 12-week functional circuit exercise intervention in frail elderly adults demonstrated significant improvements in gait speed and the timed chair-stand test.28 A pilot observational study of Gerofit participants at the Canandaigua VAMC offered 27 veterans functional circuit exercise instead of their traditional exercise facility class and found larger increases in the timed chair-stand test and 6-minute walk distance compared with 11 Gerofit participants in the traditional program.29
This Tele-Gerofit exercise training combines functional and circuit exercise strategies into telehealth delivery. However, its effect on physical performance remains to be demonstrated. To address this question, we are conducting a single-arm pilot study of Tele-Gerofit with CVT broadcast to 3 Salem CBOC affiliates (Wytheville, Staunton, and Danville, Virginia). The goal is to determine the effect on physical performance and collect feasibility data, including attendance rate and patient satisfaction with the video broadcast. In addition, we are planning an effectiveness trial to compare the impact of functional circuit exercise delivered in person (center based, not CVT) with the parent Gerofit exercise program on direct measures of endurance and strength, in addition to physical performance.
Related: Setting and Method of Measurement Affect Blood Pressure Readings in Older Veterans
Implementation research is needed to determine how Tele-Gerofit can be disseminated to other VAMCs and community-based centers beyond CBOCs. Although the cost of the equipment used to implement Tele-Gerofit is minimal, the program requires dedicated and experienced exercise instructors, and the sharing of telehealth resources with other clinical programs. The authors expect that a diverse group of stakeholders is needed across service lines of primary care, geriatrics and extended care, physical medicine and rehabilitation, and telehealth. Of note, this multidisciplinary collaboration is a hallmark of the Gerofit program. The recent success of the implementation of center-based Gerofit in VAMCs across the US demonstrates the program’s flexibility and robust results.18
Plans also include refining strategies for physical performance testing and exercise monitoring. For instance, we would like to adapt telehealth technology for heart rate monitors that can be worn by high-risk veterans at the CBOC and viewed in real time by the exercise instructor.
Conclusion
Gerofit, which is designed to help older veterans maintain independent living and prevent disability, has been demonstrated to improve quality of life and survival. Our goal has been to adapt Gerofit to CVT and provide a supervised, individualized exercise program in a group setting—a program that can be widely disseminated. Salem VAMC Tele-Gerofit is an innovative and prescriptive program that delivers CVT functional circuit exercise training to remote locations without the need for stationary exercise equipment. This approach has the potential to become an effective and feasible exercise strategy for preventing and minimizing disability in the increasing population of older veterans. Work is needed to determine whether Tele-Gerofit provides a rapid translation of Gerofit to clinical practice and improved outcomes with substantial cost savings from reduced hospitalization and institutionalization.
Acknowledgments
Gerofit has been funded by the Veterans Health Affairs Office of Geriatrics and Extended Care Non-Institutional Long-Term Care Funding and Mentored Partnership Program, and the Veterans Health Affairs Office of Rural Health Rural Enterprise-Wide Initiative.
The authors thank Kim Birkett, MPH, for assistance in editing, references, and graphics and the staff at the Wytheville, Staunton, and Danville community-based outpatient clinics for their support.
1. American College of Sports Medicine, Chodzko-Zajko WJ, Proctor DN, et al. American College of Sports Medicine position stand. Exercise and physical activity for older adults. Med Sci Sports Exerc. 2009;41(7):1510-1530.
2. Centers for Disease Control and Prevention. Adult participation in aerobic and muscle-strengthening physical activities—United States, 2011. MMWR Morb Mortal Wkly Rep. 2013;62(17):326-330.
3. Littman AJ, Forsberg CW, Koepsell TD. Physical activity in a national sample of veterans. Med Sci Sports Exerc. 2009;41(5):1006-1013.
4. US Department of Veterans Affairs, Office of Rural Health. Annual Report: Thrive 2015. https://www.ruralhealth.va.gov/docs/ORH_Annual_Report_2015_FINAL.pdf. Published 2015. Accessed July 16, 2018.
5. Rawstorn JC, Gant N, Direito A, Beckmann C, Maddison R. Telehealth exercise-based cardiac rehabilitation: a systematic review and meta-analysis. Heart. 2016;102(15):1183-1192.
6. Morey MC. Celebrating 20 years of excellence in exercise for the older veteran. Fed Pract. 2007;24(10):49-65.
7. Cowper PA, Morey MC, Bearon LB, et al. The impact of supervised exercise on the psychological well-being and health status of older veterans. J Appl Gerontol. 1991;10(4):469-485.
8. Morey MC, Cowper PA, Feussner JR, et al. Evaluation of a supervised exercise program in a geriatric population. J Am Geriatr Soc. 1989;37(4):348-354.
9. Morey MC, Pieper CF, Crowley GM, Sullivan RJ, Puglisi CM. Exercise adherence and 10-year mortality in chronically ill older adults. J Am Geriatr Soc. 2002;50(12):1929-1933.
10. Morey MC, Pieper CF, Sullivan RJ Jr, Crowley GM, Cowper PA, Robbins MS. Five-year performance trends for older exercisers: a hierarchical model of endurance, strength, and flexibility. J Am Geriatr Soc. 1996;44(10):1226-1231.
11. Valencia WM, Botros D, Pendlebury D, et al. Proactive reach and telehealth monitoring (Gerofit) enhance resistance exercise at rural setting. Innovat Aging. 2017;1(suppl 1):225.12. Pendlebury D, Botros D VW. Proactive Reach: an innovative access approach to identify & deliver GEROFIT exercise telehealth counseling to rural veterans & enhance CBOC services. J Am Geriatr Soc. 2017(suppl 1):S208. Poster presented at: Annual Scientific Meeting of the American Geriatrics Society; May 18, 2017; San Antonio, TX.
13. Morey MC, Crowley GM, Robbins MS, Cowper PA, Sullivan RJ Jr. The Gerofit program: a VA innovation. South Med J. 1994;87(5):S83-S87.
14. Cooper R, Kuh D, Hardy R; Mortality Review Group; FALCon and HALCyon Study Teams. Objectively measured physical capability levels and mortality: systematic review and meta-analysis. BMJ. 2010;341:c4467.
15. Rikli RE, Jones CJ. Development and validation of a functional fitness test for community-residing older adults. J Aging Phys Act. 1999;7(2):129-161.
16. Guralnik JM, Simonsick EM, Ferrucci L, et al. A short physical performance battery assessing lower extremity function: association with self-reported disability and prediction of mortality and nursing home admission. J Gerontol. 1994;49(2):M85-M94.
17. Morey MC, Cowper PA, Feussner JR, et al. Two-year trends in physical performance following supervised exercise among community-dwelling older veterans. J Am Geriatr Soc. 1991;39(10):986-992.
18. Morey MC, Lee CC, Castle S, et al. Should structured exercise be promoted as a model of care? Dissemination of the Department of Veterans Affairs Gerofit program. J Am Geriatr Soc. 2018;66(5):1009-1016.
19. Blanchard E, Castle S, Ines E, et al. Delivering a clinical exercise program to rural veterans via video telehealth. Poster C167 presented at: Annual Scientific Meeting of the American Geriatrics Society; May 19-21, 2016; Long Beach, CA.
20. Riebe D, Ehrman JK, Liguori G, Magal M, eds; American College of Sports Medicine. ACSM’s Guidelines for Exercise Testing and Prescription. 10th ed. Philadelphia, PA: Wolters Kluwer Health; 2018.
21. Peters DM, Fritz SL, Krotish DE. Assessing the reliability and validity of a shorter walk test compared with the 10-meter walk test for measurements of gait speed in healthy, older adults. J Geriatr Phys Ther. 2013;36(1):24-30.
22. Pedrosa R, Holanda G. Correlation between the walk, 2-minute step and TUG tests among hypertensive older women. Rev Bras Fisioter. 2009;13(3):252-256.
23. Romero-Arenas S, Blazevich AJ, Martinez-Pascual M, et al. Effects of high-resistance circuit training in an elderly population. Exp Gerontol. 2013;48(3):334-340.
24. Brentano MA, Cadore EL, Da Silva EM, et al. Physiological adaptations to strength and circuit training in postmenopausal women with bone loss. J Strength Cond Res. 2008;22(6):1816-1825.
25. Romero-Arenas S, Martinez-Pascual M, Alcaraz PE. Impact of resistance circuit training on neuromuscular, cardiorespiratory and body composition adaptations in the elderly. Aging Dis. 2013;4(5):256-263.
26. Paoli A, Pacelli F, Bargossi AM, et al. Effects of three distinct protocols of fitness training on body composition, strength and blood lactate. J Sports Med Phys Fitness. 2010;50(1):43-51.
27. Takeshima N, Rogers ME, Islam MM, Yamauchi T, Watanabe E, Okada A. Effect of concurrent aerobic and resistance circuit exercise training on fitness in older adults. Eur J Appl Physiol. 2004;93(1-2):173-182.
28. Giné-Garriga M, Guerra M, Pagés E, Manini TM, Jiménez R, Unnithan VB. The effect of functional circuit training on physical frailty in frail older adults: a randomized controlled trial. J Aging Phys Act. 2010;18(4):401-424.
29. Biddle ED, Reynolds P, Kopp T, Cammarata H, Conway P. Implementation of functional training tools elicits improvements in aerobic fitness and lower body strength in older veterans. Poster C169 presented at: Annual Scientific Meeting of the American Geriatrics Society; May 19-21, 2016; Long Beach, CA.
Clinical video telehealth can be used to deliver functional circuit exercise training to older veterans in remote locations.
Clinical video telehealth can be used to deliver functional circuit exercise training to older veterans in remote locations.
Exercise increases endurance, muscle strength, and functional performance with corresponding gains in mobility, survival, and quality of life.1 However, even with these benefits and improvements in clinical outcomes, only 15% of adults aged ≥ 65 years follow current guidelines for exercise.2 Despite their prior military training, the majority of veterans do not meet physical activity recommendations.3 Time, travel, and support are common barriers to exercise participation and adherence—barriers that are further amplified among older adults.
The Veterans Health Administration (VHA) is recognized as a world leader in telehealth service development. Currently, 677,000 veterans have received telehealth services, which represents 12% of the 5.6 million veterans under VHA care.4 Clinical video telehealth (CVT) is widely used within the VHA system to deliver health care that otherwise would not be available to veterans. Veterans who have difficulty traveling to the nearest US Department of Veteran Affairs (VA) medical center (VAMC) can access CVT programs at a participating VHA community-based outpatient clinic (CBOC). The VA has more than 45 CVT programs, including programs for mental health, weight management, cardiology, and dermatology. Outside the VA, cardiac exercise rehabilitation provided by CVT has been shown to be as effective as center-based programs in improving cardiovascular risk factors and functional capacity.5 A VHA exercise program that leveraged CVT resources and was dedicated to older adults with a wide range of comorbid conditions would have a high impact on the health and well-being of older veterans.
Gerofit is a VHA clinical demonstration program of supervised center-based exercise for veterans aged ≥ 65 years. Developed at the Durham VAMC Geriatric Research, Education, and Clinical Center (GRECC) in North Carolina, it has demonstrated improved clinical outcomes, including physical function, mobility, quality of life, and survival.6-10 The program offers veterans individualized exercise in a group setting that focuses on improving endurance, strength, and balance. The exercise prescription is based on the patient’s physical limitations as identified in a physical performance assessment.
With support from VHA Geriatric Extended Care (GEC) and the Office of Rural Health (ORH), Gerofit was implemented in 10 VAMCs across 8 VISNs. However, barriers such as travel time, distance, and transportation limit participation. Previously, we found that rural veterans lack access to exercise programs.11,12 Although some do aerobic exercise (AEX), most do not do resistance training (RT), though they are willing to learn. Access to Gerofit for rural veterans is expanding with recent support from the ORH Enterprise Wide Initiative. Rural program expansion includes several different Gerofit initiatives, many involving CBOCs.
The Salem VAMC Gerofit program sought to adapt the facility-based assessment and exercise procedures into a self-reliant CVT class for its CBOCs. This article describes the development of the Salem VAMC Gerofit CVT program, hereafter referred to as Tele-Gerofit.
Related: Expanding the Scope of Telemedicine in Gastroenterology
Program Design
Gerofit was established in 1986 at the Durham GRECC as an exercise and health promotion program for veterans aged ≥ 65 years.13 Its goal is to prevent or improve functional decline from physical inactivity and age-related conditions. Gerofit targets the geriatric patient population and thus extends beyond cardiac and pulmonary rehabilitation or weight loss programs. The primary exclusion criteria are based on safety issues in the context of a group exercise setting of older adults and include oxygen dependency, unstable cardiac disease, and moderate-to-severe cognitive impairment.
To participate in Gerofit, veterans must be able to perform activities of daily living and self-manage an exercise prescription developed by the exercise instructor based on physical performance testing. These physical performance tests include measures that are independent predictors of disability, loss of independent living, and death, as well as surrogate measures of exercise capacity (eg, strength, endurance, balance).14,15 A novel aspect of Gerofit is that the physical performance assessment is used not only to determine physical limitations, but also to individualize the exercise prescription based on the observed deficits in strength, endurance, or balance. These assessments are performed at initial enrollment; 3 months, 6 months, and 1 year later; and annually after that. Currently, the center-based Gerofit programs administer 5 items of the Senior Fitness Test: 6-minute walk, 10-meter walk (10-MWT), 30-second 1-arm curl, 30-second chair-stand test, and 8-foot up-and-go.15 The side-by-side, semitandem, and tandem standing balance tests from the short physical performance battery also are performed.16 In addition, participants complete a questionnaire that includes items from the physical functioning scale of the 12-Item Short Form Health Survey (SF-12).
After each assessment, the Gerofit exercise instructor reviews the results with the veteran and formulates an individualized exercise prescription along with goals for improvement. Veterans are encouraged to attend supervised center-based exercise sessions 3 times weekly. Classes are offered in a gym or fitness center at the VAMC or in leased space. Each patient uses a cue card that lists an exercise plan personalized for intensity and duration for aerobic exercise (AEX; eg, treadmill walking, stationary bicycling, arm ergometry), RT using dumbbells and weight equipment, and functional exercises for flexibility and balance. Some medical centers also offer yoga, tai chi, or dancing Gerofit classes.
For participants in the Durham Gerofit program, mortality decreased 25% over a decade (hazard ratio, 0.75; 95% CI, 0.61-0.91).9 A substudy that included the Psychological General Well-Being Index found that 81% of participants significantly increased their score after 1 year.7 Observed initial improvement in physical performance has been sustained over 5 years.10,17 One-year results from the recent Gerofit expansion to 6 other VAMCs showed clinically and statistically significantly improved physical performance from baseline to 3-, 6-, and 12-month follow-up.18
Adaptation of Gerofit to CVT Delivery
Initial work. The Greater Los Angeles VAMC Gerofit program conducted a pilot CVT exercise class of 6 veterans at the rural Bakersfield CBOC in California.19 Each week, an exercise instructor broadcast a 60-minute exercise class that included warm-up, RT with bands, progressive balance training, and flexibility. Trained student volunteers from California State University in Bakersfield kinesiology program were on site at the Bakersfield CBOC to perform the assessments and aid in exercises during the CVT sessions. Despite the lack of AEX per se, veterans showed significant improvement in endurance as measured by an increase in the number of steps completed in 2 minutes at the 3-month assessment (P = .049). Although exercises were not delivered in a circuit format, the improved endurance supported the potential for cardiovascular benefit from RT in older adults.
This pilot project also demonstrated that key components of the Gerofit program could be delivered safely by telehealth with onsite supervision. The Miami VA Healthcare System also offers CVT Gerofit exercise classes broadcast to the rural Florida CBOCs of Key Largo and Homestead.11 The exercise activities offered for the Miami CVT participants incorporate components of AEX (calisthenics) and RT (resistance bands). Veterans enjoyed the classes, and adherence was good. However, availability of staff and space are an ongoing challenge.
In Key Largo, 5 veterans participated before the CVT classes were placed on hold owing to the demands of other CVT programs and limited availability of the telehealth clinical technician (TCT). The Homestead CBOC continues to offer CVT Gerofit exercise classes and has 6 regular participants. Notably, the physical space at the Homestead CBOC is smaller than that at the Key Largo CBOC; the Homestead CBOC has adjusted by shifting to exercises performed while standing or sitting, ensuring participants’ safety and satisfaction.
The Baltimore, Maryland VAMC Gerofit program offers other innovative CVT exercise classes, including a tai chi class, and a class with exercise performed while sitting in a chair. Although the Baltimore VAMC CVT exercise classes do not have the scope of the center-based exercise prescriptions, they are unique in that they are broadcast not only to their affiliated CBOCs, but also other Gerofit programs in different VISNs.
Related: Telehealth for Rural Veterans With Neurologic Disorders
Salem VAMC Gerofit Program. The center-based Salem VAMC Gerofit program was established in July 2015. In fiscal year 2017, its dedicated exercise facility had more than 5,000 patient visits. Despite the program’s success, we prioritized establishing Tele-Gerofit because of the medical center’s rural location in southwest Virginia and the large number of veterans who receive care at CBOCs. Therefore, much as with the pilot CVT Gerofit classes in Los Angeles and Miami, the target setting was rural CBOCs. The goal for Salem VAMC Tele-Gerofit was to modify Gerofit delivery to the CVT format and a CBOC setting with minimal modification of the content and personnel requirements of both physical performance testing and exercise training procedures.
Adjustments for CBOC Setting. The enrollment process for Tele-Gerofit is the same as that for the center-based program. To start, a veteran’s primary care provider reviews the list of eligibility criteria and, if the veteran qualifies, places a consult. A Gerofit team member then contacts the veteran by phone to describe the program and schedule an assessment. At the baseline physical performance assessment, American College of Sports Medicine guidelines on exercise participation, health screening, and exercise intensity are used to evaluate veterans and rank them by their cardiovascular risk.20 All new program participants start with low-intensity exercise and gradually progress to recommended levels of exercise. Before starting an exercise class, participants are instructed on use of the 10-point rating of perceived exertion (RPE).
Each CBOC site is supplied with an RPE poster that is displayed for participants’ use. During a Tele-Gerofit class, the exercise instructor asks participants to periodically report their RPE. This class differs slightly from the center-based exercise sessions in which RPE is primarily assessed when a different exercise is introduced or the duration or intensity of an exercise is increased. The Gerofit instructor monitors exercise and treatment fidelity, but the onsite TCT observes for safety during class. The TCT also takes initial vital signs and sets up the room for the class. Emergency contacts and procedures are posted in each CBOC CVT room and are available to the center-based exercise instructor. Because the CBOCs are not inside medical facilities, some CBOC directors have asked that heart rate monitors be used as an extra safety precaution to ensure that high-risk participants do not exceed a heart rate limit that may be set by their cardiologists.
Modifications to Physical Performance Assessment. Physical performance testing had to be adapted to the small rooms available at the CBOCs. For measuring normal gait speed, the 10-MWT was replaced with the 4-meter walk test (4-MWT). The 4-MWT has excellent test–retest reliability with an intraclass correlation coefficient (ICC) of 0.93, but the discrepancy in gait speed between the 4-MWT and the 10-MWT is such that the tests cannot be used interchangeably.21 For measuring endurance, the 6-minute walk test was replaced with the 2-minute step test (2-MST). In older adults, the 2-MST has a moderate correlation with 6-minute walk distance (r = 0.36; P = .04) and high reliability (ICC, 0.90).15,22 The 30-second 1-arm curl, the 30-second chair-stand test, and the 8-foot up-and-go test are performed without modification and require only dumbbells, a chair without wheels, and a stopwatch.
The exercise instructor at the Salem VAMC conducts physical performance testing by 2-way videoconferencing with the veteran in a room at the CBOC. The TCT at the CBOC assists by measuring and demarcating 4 meters on the floor and a designated height on the wall for knee elevation for 4-MWT and 2-MST, respectively. The TCT remains in the room during the assessment visit. Except for taking vital signs before and after the physical performance assessment, the TCT does not participate in the testing. To date, more than 20 physical performance assessments have been conducted without difficulty at Salem-affiliated CBOCs. The primary challenge has been scheduling the room with CVT equipment (ie, camera and screen) for the 30-minute individual assessment session, which occurs on a rolling basis as individuals are enrolled and followed.
After the assessment is completed, the exercise instructor reviews the results with the participant and provides feedback on areas in need of improvement. However, these education sessions can be lengthy and are best supported by giving the patient a personalized handout.
Functional Circuit Exercise. In Tele-Gerofit, exercise training is delivered by CVT broadcast from the Salem VAMC to veterans in a room (equipped with steps, dumbbells, chairs, and bands) at the CBOC. This type of exercise training, which uses only mobile equipment and plyometric (weight-bearing) exercises, is referred to as functional exercise. The AEX includes marching in place, moving on and off a raised step, and body-weight exercises, while RT uses dumbbells, resistance bands, and plyometric exercises (Table 2).
Progression of intensity is achieved by increasing the rate of stepping and the size of the steps (AEX) or the number of repetitions and the weight of the dumbbells or bands (RT). Each veteran exercises at an intensity level that is appropriate for his or her baseline limitations and medical conditions. The exercise instructor uses different forms of the same equipment (eg, heavier dumbbells, higher steps) to vary intensity among individuals while having them perform the same exercises as a group. The challenge is to adjust the pace of the AEX or the timing of the RT repetitions for individuals new to the class.
Delivery of exercise training in the form of circuits allows for a diverse exercise program in a setting with limited space. Circuit training is an exercise modality that consists of a series of different exercises, each usually completed in 30 to 60 seconds, with minimal rest between each type of exercise. Each Tele-Gerofit circuit has a mix of AEX and RT exercises performed for 3 minutes consecutively (Figure).
The design of the circuit training can be adjusted based on the number of individuals in the class. Larger classes can be split into 2 groups that alternate between exercise sets, while smaller classes have 1 group performing the same exercise set and then rotating to either the AEX or RT set. Total exercise time to complete the circuit depends on the number of different exercises, number of repetitions, and the rest between repetitions and the different exercises. In this way, total exercise time can be made shorter or longer depending on the veteran’s capacity.
Frequency. Tele-Gerofit exercise classes are currently offered twice weekly and last about 1 hour, which includes warm-up (8-10 minutes), functional circuit training (40 minutes), and cooldown/stretching (8-10 minutes). A challenge for the exercise instructor is the need to provide ongoing clear instructions both to the class and to individuals as needed. As the exercise prescription for each patient is based on physical performance testing, the exercise instructor for the training must be familiar with the test results. Derivation of the exercise prescription in Tele-Gerofit follows the same process as center-based Gerofit.
Each patient is given an exercise prescription written to address any impairments noted in the different domains of the physical performance assessment, scored using age and sex percentiles. For instance, individuals scoring poorly on lower body strength are given specific lower body strengthening exercises. Participants are given an exercise program that guides them toward achieving recommended physical activity guidelines using their RPE to modulate each exercise. Duration and intensity of each type of planned exercise are formally discussed after initial and follow-up assessments. In addition, exercise training is informally progressed throughout the program. For Tele-Gerofit, instructors must design each class with the group in mind while being prepared for modifications and specific changes for individuals.
Discussion
Tele-Gerofit adapts the well-established center-based Gerofit program to be executed without an exercise facility while maintaining the content of the evidence-based procedures. Physical performance testing and exercise training were modified, adding elements necessary for CVT assessments and classes to be broadcast from the Salem VAMC to its affiliated CBOCs. Tele-Gerofit exercises are performed in a circuit style that allows a veteran or small structured groups of veterans to move among exercises and requires less space than traditional group exercise does. Safety and monitoring concerns are addressed with a safety procedure that includes emergency plans for each site, prescreening of enrolled participants, and monitoring of exercise intensity in accordance with national guidelines.1 Similar to the center-based Gerofit program, the exercise prescription is tailored to each veteran’s physical limitations based on initial and ongoing assessment of physical performance. Tele-Gerofit physical performance testing fulfills the same need with only a few modifications using validated measures. Tele-Gerofit assessments are administered by CVT without the need for additional staff on site.
Adaptation of center-based Gerofit exercise classes to Tele-Gerofit is a major innovation. Use of a circuit exercise design was supported by findings in older adults that RT alone, when performed quickly with minimal rest between each set and exercise station, increases both aerobic capacity and strength.23,24 Older adult RT trials that compared circuit RT with traditional RT found that strength gains are comparable between circuit and traditional RT.24-26 Working with adults aged > 60 years, Takeshima and colleagues conducted a trial of circuit exercise with added callisthenic exercises performed in place between RT on exercise machines.27 This dual-modality (AEX+RT) circuit approach was well tolerated and effective, increasing aerobic capacity and strength. Unfortunately, the resistance exercise machines used in those circuit exercise studies and in the center-based Gerofit program are not an option for Tele-Gerofit.
The requirement for an exercise facility was removed by designing Tele-Gerofit exercise to include only functional exercises that rely on body weight or small mobile exercise equipment. Although popular among young adults, functional circuit exercise is understudied in older adults. Recently, a 12-week functional circuit exercise intervention in frail elderly adults demonstrated significant improvements in gait speed and the timed chair-stand test.28 A pilot observational study of Gerofit participants at the Canandaigua VAMC offered 27 veterans functional circuit exercise instead of their traditional exercise facility class and found larger increases in the timed chair-stand test and 6-minute walk distance compared with 11 Gerofit participants in the traditional program.29
This Tele-Gerofit exercise training combines functional and circuit exercise strategies into telehealth delivery. However, its effect on physical performance remains to be demonstrated. To address this question, we are conducting a single-arm pilot study of Tele-Gerofit with CVT broadcast to 3 Salem CBOC affiliates (Wytheville, Staunton, and Danville, Virginia). The goal is to determine the effect on physical performance and collect feasibility data, including attendance rate and patient satisfaction with the video broadcast. In addition, we are planning an effectiveness trial to compare the impact of functional circuit exercise delivered in person (center based, not CVT) with the parent Gerofit exercise program on direct measures of endurance and strength, in addition to physical performance.
Related: Setting and Method of Measurement Affect Blood Pressure Readings in Older Veterans
Implementation research is needed to determine how Tele-Gerofit can be disseminated to other VAMCs and community-based centers beyond CBOCs. Although the cost of the equipment used to implement Tele-Gerofit is minimal, the program requires dedicated and experienced exercise instructors, and the sharing of telehealth resources with other clinical programs. The authors expect that a diverse group of stakeholders is needed across service lines of primary care, geriatrics and extended care, physical medicine and rehabilitation, and telehealth. Of note, this multidisciplinary collaboration is a hallmark of the Gerofit program. The recent success of the implementation of center-based Gerofit in VAMCs across the US demonstrates the program’s flexibility and robust results.18
Plans also include refining strategies for physical performance testing and exercise monitoring. For instance, we would like to adapt telehealth technology for heart rate monitors that can be worn by high-risk veterans at the CBOC and viewed in real time by the exercise instructor.
Conclusion
Gerofit, which is designed to help older veterans maintain independent living and prevent disability, has been demonstrated to improve quality of life and survival. Our goal has been to adapt Gerofit to CVT and provide a supervised, individualized exercise program in a group setting—a program that can be widely disseminated. Salem VAMC Tele-Gerofit is an innovative and prescriptive program that delivers CVT functional circuit exercise training to remote locations without the need for stationary exercise equipment. This approach has the potential to become an effective and feasible exercise strategy for preventing and minimizing disability in the increasing population of older veterans. Work is needed to determine whether Tele-Gerofit provides a rapid translation of Gerofit to clinical practice and improved outcomes with substantial cost savings from reduced hospitalization and institutionalization.
Acknowledgments
Gerofit has been funded by the Veterans Health Affairs Office of Geriatrics and Extended Care Non-Institutional Long-Term Care Funding and Mentored Partnership Program, and the Veterans Health Affairs Office of Rural Health Rural Enterprise-Wide Initiative.
The authors thank Kim Birkett, MPH, for assistance in editing, references, and graphics and the staff at the Wytheville, Staunton, and Danville community-based outpatient clinics for their support.
Exercise increases endurance, muscle strength, and functional performance with corresponding gains in mobility, survival, and quality of life.1 However, even with these benefits and improvements in clinical outcomes, only 15% of adults aged ≥ 65 years follow current guidelines for exercise.2 Despite their prior military training, the majority of veterans do not meet physical activity recommendations.3 Time, travel, and support are common barriers to exercise participation and adherence—barriers that are further amplified among older adults.
The Veterans Health Administration (VHA) is recognized as a world leader in telehealth service development. Currently, 677,000 veterans have received telehealth services, which represents 12% of the 5.6 million veterans under VHA care.4 Clinical video telehealth (CVT) is widely used within the VHA system to deliver health care that otherwise would not be available to veterans. Veterans who have difficulty traveling to the nearest US Department of Veteran Affairs (VA) medical center (VAMC) can access CVT programs at a participating VHA community-based outpatient clinic (CBOC). The VA has more than 45 CVT programs, including programs for mental health, weight management, cardiology, and dermatology. Outside the VA, cardiac exercise rehabilitation provided by CVT has been shown to be as effective as center-based programs in improving cardiovascular risk factors and functional capacity.5 A VHA exercise program that leveraged CVT resources and was dedicated to older adults with a wide range of comorbid conditions would have a high impact on the health and well-being of older veterans.
Gerofit is a VHA clinical demonstration program of supervised center-based exercise for veterans aged ≥ 65 years. Developed at the Durham VAMC Geriatric Research, Education, and Clinical Center (GRECC) in North Carolina, it has demonstrated improved clinical outcomes, including physical function, mobility, quality of life, and survival.6-10 The program offers veterans individualized exercise in a group setting that focuses on improving endurance, strength, and balance. The exercise prescription is based on the patient’s physical limitations as identified in a physical performance assessment.
With support from VHA Geriatric Extended Care (GEC) and the Office of Rural Health (ORH), Gerofit was implemented in 10 VAMCs across 8 VISNs. However, barriers such as travel time, distance, and transportation limit participation. Previously, we found that rural veterans lack access to exercise programs.11,12 Although some do aerobic exercise (AEX), most do not do resistance training (RT), though they are willing to learn. Access to Gerofit for rural veterans is expanding with recent support from the ORH Enterprise Wide Initiative. Rural program expansion includes several different Gerofit initiatives, many involving CBOCs.
The Salem VAMC Gerofit program sought to adapt the facility-based assessment and exercise procedures into a self-reliant CVT class for its CBOCs. This article describes the development of the Salem VAMC Gerofit CVT program, hereafter referred to as Tele-Gerofit.
Related: Expanding the Scope of Telemedicine in Gastroenterology
Program Design
Gerofit was established in 1986 at the Durham GRECC as an exercise and health promotion program for veterans aged ≥ 65 years.13 Its goal is to prevent or improve functional decline from physical inactivity and age-related conditions. Gerofit targets the geriatric patient population and thus extends beyond cardiac and pulmonary rehabilitation or weight loss programs. The primary exclusion criteria are based on safety issues in the context of a group exercise setting of older adults and include oxygen dependency, unstable cardiac disease, and moderate-to-severe cognitive impairment.
To participate in Gerofit, veterans must be able to perform activities of daily living and self-manage an exercise prescription developed by the exercise instructor based on physical performance testing. These physical performance tests include measures that are independent predictors of disability, loss of independent living, and death, as well as surrogate measures of exercise capacity (eg, strength, endurance, balance).14,15 A novel aspect of Gerofit is that the physical performance assessment is used not only to determine physical limitations, but also to individualize the exercise prescription based on the observed deficits in strength, endurance, or balance. These assessments are performed at initial enrollment; 3 months, 6 months, and 1 year later; and annually after that. Currently, the center-based Gerofit programs administer 5 items of the Senior Fitness Test: 6-minute walk, 10-meter walk (10-MWT), 30-second 1-arm curl, 30-second chair-stand test, and 8-foot up-and-go.15 The side-by-side, semitandem, and tandem standing balance tests from the short physical performance battery also are performed.16 In addition, participants complete a questionnaire that includes items from the physical functioning scale of the 12-Item Short Form Health Survey (SF-12).
After each assessment, the Gerofit exercise instructor reviews the results with the veteran and formulates an individualized exercise prescription along with goals for improvement. Veterans are encouraged to attend supervised center-based exercise sessions 3 times weekly. Classes are offered in a gym or fitness center at the VAMC or in leased space. Each patient uses a cue card that lists an exercise plan personalized for intensity and duration for aerobic exercise (AEX; eg, treadmill walking, stationary bicycling, arm ergometry), RT using dumbbells and weight equipment, and functional exercises for flexibility and balance. Some medical centers also offer yoga, tai chi, or dancing Gerofit classes.
For participants in the Durham Gerofit program, mortality decreased 25% over a decade (hazard ratio, 0.75; 95% CI, 0.61-0.91).9 A substudy that included the Psychological General Well-Being Index found that 81% of participants significantly increased their score after 1 year.7 Observed initial improvement in physical performance has been sustained over 5 years.10,17 One-year results from the recent Gerofit expansion to 6 other VAMCs showed clinically and statistically significantly improved physical performance from baseline to 3-, 6-, and 12-month follow-up.18
Adaptation of Gerofit to CVT Delivery
Initial work. The Greater Los Angeles VAMC Gerofit program conducted a pilot CVT exercise class of 6 veterans at the rural Bakersfield CBOC in California.19 Each week, an exercise instructor broadcast a 60-minute exercise class that included warm-up, RT with bands, progressive balance training, and flexibility. Trained student volunteers from California State University in Bakersfield kinesiology program were on site at the Bakersfield CBOC to perform the assessments and aid in exercises during the CVT sessions. Despite the lack of AEX per se, veterans showed significant improvement in endurance as measured by an increase in the number of steps completed in 2 minutes at the 3-month assessment (P = .049). Although exercises were not delivered in a circuit format, the improved endurance supported the potential for cardiovascular benefit from RT in older adults.
This pilot project also demonstrated that key components of the Gerofit program could be delivered safely by telehealth with onsite supervision. The Miami VA Healthcare System also offers CVT Gerofit exercise classes broadcast to the rural Florida CBOCs of Key Largo and Homestead.11 The exercise activities offered for the Miami CVT participants incorporate components of AEX (calisthenics) and RT (resistance bands). Veterans enjoyed the classes, and adherence was good. However, availability of staff and space are an ongoing challenge.
In Key Largo, 5 veterans participated before the CVT classes were placed on hold owing to the demands of other CVT programs and limited availability of the telehealth clinical technician (TCT). The Homestead CBOC continues to offer CVT Gerofit exercise classes and has 6 regular participants. Notably, the physical space at the Homestead CBOC is smaller than that at the Key Largo CBOC; the Homestead CBOC has adjusted by shifting to exercises performed while standing or sitting, ensuring participants’ safety and satisfaction.
The Baltimore, Maryland VAMC Gerofit program offers other innovative CVT exercise classes, including a tai chi class, and a class with exercise performed while sitting in a chair. Although the Baltimore VAMC CVT exercise classes do not have the scope of the center-based exercise prescriptions, they are unique in that they are broadcast not only to their affiliated CBOCs, but also other Gerofit programs in different VISNs.
Related: Telehealth for Rural Veterans With Neurologic Disorders
Salem VAMC Gerofit Program. The center-based Salem VAMC Gerofit program was established in July 2015. In fiscal year 2017, its dedicated exercise facility had more than 5,000 patient visits. Despite the program’s success, we prioritized establishing Tele-Gerofit because of the medical center’s rural location in southwest Virginia and the large number of veterans who receive care at CBOCs. Therefore, much as with the pilot CVT Gerofit classes in Los Angeles and Miami, the target setting was rural CBOCs. The goal for Salem VAMC Tele-Gerofit was to modify Gerofit delivery to the CVT format and a CBOC setting with minimal modification of the content and personnel requirements of both physical performance testing and exercise training procedures.
Adjustments for CBOC Setting. The enrollment process for Tele-Gerofit is the same as that for the center-based program. To start, a veteran’s primary care provider reviews the list of eligibility criteria and, if the veteran qualifies, places a consult. A Gerofit team member then contacts the veteran by phone to describe the program and schedule an assessment. At the baseline physical performance assessment, American College of Sports Medicine guidelines on exercise participation, health screening, and exercise intensity are used to evaluate veterans and rank them by their cardiovascular risk.20 All new program participants start with low-intensity exercise and gradually progress to recommended levels of exercise. Before starting an exercise class, participants are instructed on use of the 10-point rating of perceived exertion (RPE).
Each CBOC site is supplied with an RPE poster that is displayed for participants’ use. During a Tele-Gerofit class, the exercise instructor asks participants to periodically report their RPE. This class differs slightly from the center-based exercise sessions in which RPE is primarily assessed when a different exercise is introduced or the duration or intensity of an exercise is increased. The Gerofit instructor monitors exercise and treatment fidelity, but the onsite TCT observes for safety during class. The TCT also takes initial vital signs and sets up the room for the class. Emergency contacts and procedures are posted in each CBOC CVT room and are available to the center-based exercise instructor. Because the CBOCs are not inside medical facilities, some CBOC directors have asked that heart rate monitors be used as an extra safety precaution to ensure that high-risk participants do not exceed a heart rate limit that may be set by their cardiologists.
Modifications to Physical Performance Assessment. Physical performance testing had to be adapted to the small rooms available at the CBOCs. For measuring normal gait speed, the 10-MWT was replaced with the 4-meter walk test (4-MWT). The 4-MWT has excellent test–retest reliability with an intraclass correlation coefficient (ICC) of 0.93, but the discrepancy in gait speed between the 4-MWT and the 10-MWT is such that the tests cannot be used interchangeably.21 For measuring endurance, the 6-minute walk test was replaced with the 2-minute step test (2-MST). In older adults, the 2-MST has a moderate correlation with 6-minute walk distance (r = 0.36; P = .04) and high reliability (ICC, 0.90).15,22 The 30-second 1-arm curl, the 30-second chair-stand test, and the 8-foot up-and-go test are performed without modification and require only dumbbells, a chair without wheels, and a stopwatch.
The exercise instructor at the Salem VAMC conducts physical performance testing by 2-way videoconferencing with the veteran in a room at the CBOC. The TCT at the CBOC assists by measuring and demarcating 4 meters on the floor and a designated height on the wall for knee elevation for 4-MWT and 2-MST, respectively. The TCT remains in the room during the assessment visit. Except for taking vital signs before and after the physical performance assessment, the TCT does not participate in the testing. To date, more than 20 physical performance assessments have been conducted without difficulty at Salem-affiliated CBOCs. The primary challenge has been scheduling the room with CVT equipment (ie, camera and screen) for the 30-minute individual assessment session, which occurs on a rolling basis as individuals are enrolled and followed.
After the assessment is completed, the exercise instructor reviews the results with the participant and provides feedback on areas in need of improvement. However, these education sessions can be lengthy and are best supported by giving the patient a personalized handout.
Functional Circuit Exercise. In Tele-Gerofit, exercise training is delivered by CVT broadcast from the Salem VAMC to veterans in a room (equipped with steps, dumbbells, chairs, and bands) at the CBOC. This type of exercise training, which uses only mobile equipment and plyometric (weight-bearing) exercises, is referred to as functional exercise. The AEX includes marching in place, moving on and off a raised step, and body-weight exercises, while RT uses dumbbells, resistance bands, and plyometric exercises (Table 2).
Progression of intensity is achieved by increasing the rate of stepping and the size of the steps (AEX) or the number of repetitions and the weight of the dumbbells or bands (RT). Each veteran exercises at an intensity level that is appropriate for his or her baseline limitations and medical conditions. The exercise instructor uses different forms of the same equipment (eg, heavier dumbbells, higher steps) to vary intensity among individuals while having them perform the same exercises as a group. The challenge is to adjust the pace of the AEX or the timing of the RT repetitions for individuals new to the class.
Delivery of exercise training in the form of circuits allows for a diverse exercise program in a setting with limited space. Circuit training is an exercise modality that consists of a series of different exercises, each usually completed in 30 to 60 seconds, with minimal rest between each type of exercise. Each Tele-Gerofit circuit has a mix of AEX and RT exercises performed for 3 minutes consecutively (Figure).
The design of the circuit training can be adjusted based on the number of individuals in the class. Larger classes can be split into 2 groups that alternate between exercise sets, while smaller classes have 1 group performing the same exercise set and then rotating to either the AEX or RT set. Total exercise time to complete the circuit depends on the number of different exercises, number of repetitions, and the rest between repetitions and the different exercises. In this way, total exercise time can be made shorter or longer depending on the veteran’s capacity.
Frequency. Tele-Gerofit exercise classes are currently offered twice weekly and last about 1 hour, which includes warm-up (8-10 minutes), functional circuit training (40 minutes), and cooldown/stretching (8-10 minutes). A challenge for the exercise instructor is the need to provide ongoing clear instructions both to the class and to individuals as needed. As the exercise prescription for each patient is based on physical performance testing, the exercise instructor for the training must be familiar with the test results. Derivation of the exercise prescription in Tele-Gerofit follows the same process as center-based Gerofit.
Each patient is given an exercise prescription written to address any impairments noted in the different domains of the physical performance assessment, scored using age and sex percentiles. For instance, individuals scoring poorly on lower body strength are given specific lower body strengthening exercises. Participants are given an exercise program that guides them toward achieving recommended physical activity guidelines using their RPE to modulate each exercise. Duration and intensity of each type of planned exercise are formally discussed after initial and follow-up assessments. In addition, exercise training is informally progressed throughout the program. For Tele-Gerofit, instructors must design each class with the group in mind while being prepared for modifications and specific changes for individuals.
Discussion
Tele-Gerofit adapts the well-established center-based Gerofit program to be executed without an exercise facility while maintaining the content of the evidence-based procedures. Physical performance testing and exercise training were modified, adding elements necessary for CVT assessments and classes to be broadcast from the Salem VAMC to its affiliated CBOCs. Tele-Gerofit exercises are performed in a circuit style that allows a veteran or small structured groups of veterans to move among exercises and requires less space than traditional group exercise does. Safety and monitoring concerns are addressed with a safety procedure that includes emergency plans for each site, prescreening of enrolled participants, and monitoring of exercise intensity in accordance with national guidelines.1 Similar to the center-based Gerofit program, the exercise prescription is tailored to each veteran’s physical limitations based on initial and ongoing assessment of physical performance. Tele-Gerofit physical performance testing fulfills the same need with only a few modifications using validated measures. Tele-Gerofit assessments are administered by CVT without the need for additional staff on site.
Adaptation of center-based Gerofit exercise classes to Tele-Gerofit is a major innovation. Use of a circuit exercise design was supported by findings in older adults that RT alone, when performed quickly with minimal rest between each set and exercise station, increases both aerobic capacity and strength.23,24 Older adult RT trials that compared circuit RT with traditional RT found that strength gains are comparable between circuit and traditional RT.24-26 Working with adults aged > 60 years, Takeshima and colleagues conducted a trial of circuit exercise with added callisthenic exercises performed in place between RT on exercise machines.27 This dual-modality (AEX+RT) circuit approach was well tolerated and effective, increasing aerobic capacity and strength. Unfortunately, the resistance exercise machines used in those circuit exercise studies and in the center-based Gerofit program are not an option for Tele-Gerofit.
The requirement for an exercise facility was removed by designing Tele-Gerofit exercise to include only functional exercises that rely on body weight or small mobile exercise equipment. Although popular among young adults, functional circuit exercise is understudied in older adults. Recently, a 12-week functional circuit exercise intervention in frail elderly adults demonstrated significant improvements in gait speed and the timed chair-stand test.28 A pilot observational study of Gerofit participants at the Canandaigua VAMC offered 27 veterans functional circuit exercise instead of their traditional exercise facility class and found larger increases in the timed chair-stand test and 6-minute walk distance compared with 11 Gerofit participants in the traditional program.29
This Tele-Gerofit exercise training combines functional and circuit exercise strategies into telehealth delivery. However, its effect on physical performance remains to be demonstrated. To address this question, we are conducting a single-arm pilot study of Tele-Gerofit with CVT broadcast to 3 Salem CBOC affiliates (Wytheville, Staunton, and Danville, Virginia). The goal is to determine the effect on physical performance and collect feasibility data, including attendance rate and patient satisfaction with the video broadcast. In addition, we are planning an effectiveness trial to compare the impact of functional circuit exercise delivered in person (center based, not CVT) with the parent Gerofit exercise program on direct measures of endurance and strength, in addition to physical performance.
Related: Setting and Method of Measurement Affect Blood Pressure Readings in Older Veterans
Implementation research is needed to determine how Tele-Gerofit can be disseminated to other VAMCs and community-based centers beyond CBOCs. Although the cost of the equipment used to implement Tele-Gerofit is minimal, the program requires dedicated and experienced exercise instructors, and the sharing of telehealth resources with other clinical programs. The authors expect that a diverse group of stakeholders is needed across service lines of primary care, geriatrics and extended care, physical medicine and rehabilitation, and telehealth. Of note, this multidisciplinary collaboration is a hallmark of the Gerofit program. The recent success of the implementation of center-based Gerofit in VAMCs across the US demonstrates the program’s flexibility and robust results.18
Plans also include refining strategies for physical performance testing and exercise monitoring. For instance, we would like to adapt telehealth technology for heart rate monitors that can be worn by high-risk veterans at the CBOC and viewed in real time by the exercise instructor.
Conclusion
Gerofit, which is designed to help older veterans maintain independent living and prevent disability, has been demonstrated to improve quality of life and survival. Our goal has been to adapt Gerofit to CVT and provide a supervised, individualized exercise program in a group setting—a program that can be widely disseminated. Salem VAMC Tele-Gerofit is an innovative and prescriptive program that delivers CVT functional circuit exercise training to remote locations without the need for stationary exercise equipment. This approach has the potential to become an effective and feasible exercise strategy for preventing and minimizing disability in the increasing population of older veterans. Work is needed to determine whether Tele-Gerofit provides a rapid translation of Gerofit to clinical practice and improved outcomes with substantial cost savings from reduced hospitalization and institutionalization.
Acknowledgments
Gerofit has been funded by the Veterans Health Affairs Office of Geriatrics and Extended Care Non-Institutional Long-Term Care Funding and Mentored Partnership Program, and the Veterans Health Affairs Office of Rural Health Rural Enterprise-Wide Initiative.
The authors thank Kim Birkett, MPH, for assistance in editing, references, and graphics and the staff at the Wytheville, Staunton, and Danville community-based outpatient clinics for their support.
1. American College of Sports Medicine, Chodzko-Zajko WJ, Proctor DN, et al. American College of Sports Medicine position stand. Exercise and physical activity for older adults. Med Sci Sports Exerc. 2009;41(7):1510-1530.
2. Centers for Disease Control and Prevention. Adult participation in aerobic and muscle-strengthening physical activities—United States, 2011. MMWR Morb Mortal Wkly Rep. 2013;62(17):326-330.
3. Littman AJ, Forsberg CW, Koepsell TD. Physical activity in a national sample of veterans. Med Sci Sports Exerc. 2009;41(5):1006-1013.
4. US Department of Veterans Affairs, Office of Rural Health. Annual Report: Thrive 2015. https://www.ruralhealth.va.gov/docs/ORH_Annual_Report_2015_FINAL.pdf. Published 2015. Accessed July 16, 2018.
5. Rawstorn JC, Gant N, Direito A, Beckmann C, Maddison R. Telehealth exercise-based cardiac rehabilitation: a systematic review and meta-analysis. Heart. 2016;102(15):1183-1192.
6. Morey MC. Celebrating 20 years of excellence in exercise for the older veteran. Fed Pract. 2007;24(10):49-65.
7. Cowper PA, Morey MC, Bearon LB, et al. The impact of supervised exercise on the psychological well-being and health status of older veterans. J Appl Gerontol. 1991;10(4):469-485.
8. Morey MC, Cowper PA, Feussner JR, et al. Evaluation of a supervised exercise program in a geriatric population. J Am Geriatr Soc. 1989;37(4):348-354.
9. Morey MC, Pieper CF, Crowley GM, Sullivan RJ, Puglisi CM. Exercise adherence and 10-year mortality in chronically ill older adults. J Am Geriatr Soc. 2002;50(12):1929-1933.
10. Morey MC, Pieper CF, Sullivan RJ Jr, Crowley GM, Cowper PA, Robbins MS. Five-year performance trends for older exercisers: a hierarchical model of endurance, strength, and flexibility. J Am Geriatr Soc. 1996;44(10):1226-1231.
11. Valencia WM, Botros D, Pendlebury D, et al. Proactive reach and telehealth monitoring (Gerofit) enhance resistance exercise at rural setting. Innovat Aging. 2017;1(suppl 1):225.12. Pendlebury D, Botros D VW. Proactive Reach: an innovative access approach to identify & deliver GEROFIT exercise telehealth counseling to rural veterans & enhance CBOC services. J Am Geriatr Soc. 2017(suppl 1):S208. Poster presented at: Annual Scientific Meeting of the American Geriatrics Society; May 18, 2017; San Antonio, TX.
13. Morey MC, Crowley GM, Robbins MS, Cowper PA, Sullivan RJ Jr. The Gerofit program: a VA innovation. South Med J. 1994;87(5):S83-S87.
14. Cooper R, Kuh D, Hardy R; Mortality Review Group; FALCon and HALCyon Study Teams. Objectively measured physical capability levels and mortality: systematic review and meta-analysis. BMJ. 2010;341:c4467.
15. Rikli RE, Jones CJ. Development and validation of a functional fitness test for community-residing older adults. J Aging Phys Act. 1999;7(2):129-161.
16. Guralnik JM, Simonsick EM, Ferrucci L, et al. A short physical performance battery assessing lower extremity function: association with self-reported disability and prediction of mortality and nursing home admission. J Gerontol. 1994;49(2):M85-M94.
17. Morey MC, Cowper PA, Feussner JR, et al. Two-year trends in physical performance following supervised exercise among community-dwelling older veterans. J Am Geriatr Soc. 1991;39(10):986-992.
18. Morey MC, Lee CC, Castle S, et al. Should structured exercise be promoted as a model of care? Dissemination of the Department of Veterans Affairs Gerofit program. J Am Geriatr Soc. 2018;66(5):1009-1016.
19. Blanchard E, Castle S, Ines E, et al. Delivering a clinical exercise program to rural veterans via video telehealth. Poster C167 presented at: Annual Scientific Meeting of the American Geriatrics Society; May 19-21, 2016; Long Beach, CA.
20. Riebe D, Ehrman JK, Liguori G, Magal M, eds; American College of Sports Medicine. ACSM’s Guidelines for Exercise Testing and Prescription. 10th ed. Philadelphia, PA: Wolters Kluwer Health; 2018.
21. Peters DM, Fritz SL, Krotish DE. Assessing the reliability and validity of a shorter walk test compared with the 10-meter walk test for measurements of gait speed in healthy, older adults. J Geriatr Phys Ther. 2013;36(1):24-30.
22. Pedrosa R, Holanda G. Correlation between the walk, 2-minute step and TUG tests among hypertensive older women. Rev Bras Fisioter. 2009;13(3):252-256.
23. Romero-Arenas S, Blazevich AJ, Martinez-Pascual M, et al. Effects of high-resistance circuit training in an elderly population. Exp Gerontol. 2013;48(3):334-340.
24. Brentano MA, Cadore EL, Da Silva EM, et al. Physiological adaptations to strength and circuit training in postmenopausal women with bone loss. J Strength Cond Res. 2008;22(6):1816-1825.
25. Romero-Arenas S, Martinez-Pascual M, Alcaraz PE. Impact of resistance circuit training on neuromuscular, cardiorespiratory and body composition adaptations in the elderly. Aging Dis. 2013;4(5):256-263.
26. Paoli A, Pacelli F, Bargossi AM, et al. Effects of three distinct protocols of fitness training on body composition, strength and blood lactate. J Sports Med Phys Fitness. 2010;50(1):43-51.
27. Takeshima N, Rogers ME, Islam MM, Yamauchi T, Watanabe E, Okada A. Effect of concurrent aerobic and resistance circuit exercise training on fitness in older adults. Eur J Appl Physiol. 2004;93(1-2):173-182.
28. Giné-Garriga M, Guerra M, Pagés E, Manini TM, Jiménez R, Unnithan VB. The effect of functional circuit training on physical frailty in frail older adults: a randomized controlled trial. J Aging Phys Act. 2010;18(4):401-424.
29. Biddle ED, Reynolds P, Kopp T, Cammarata H, Conway P. Implementation of functional training tools elicits improvements in aerobic fitness and lower body strength in older veterans. Poster C169 presented at: Annual Scientific Meeting of the American Geriatrics Society; May 19-21, 2016; Long Beach, CA.
1. American College of Sports Medicine, Chodzko-Zajko WJ, Proctor DN, et al. American College of Sports Medicine position stand. Exercise and physical activity for older adults. Med Sci Sports Exerc. 2009;41(7):1510-1530.
2. Centers for Disease Control and Prevention. Adult participation in aerobic and muscle-strengthening physical activities—United States, 2011. MMWR Morb Mortal Wkly Rep. 2013;62(17):326-330.
3. Littman AJ, Forsberg CW, Koepsell TD. Physical activity in a national sample of veterans. Med Sci Sports Exerc. 2009;41(5):1006-1013.
4. US Department of Veterans Affairs, Office of Rural Health. Annual Report: Thrive 2015. https://www.ruralhealth.va.gov/docs/ORH_Annual_Report_2015_FINAL.pdf. Published 2015. Accessed July 16, 2018.
5. Rawstorn JC, Gant N, Direito A, Beckmann C, Maddison R. Telehealth exercise-based cardiac rehabilitation: a systematic review and meta-analysis. Heart. 2016;102(15):1183-1192.
6. Morey MC. Celebrating 20 years of excellence in exercise for the older veteran. Fed Pract. 2007;24(10):49-65.
7. Cowper PA, Morey MC, Bearon LB, et al. The impact of supervised exercise on the psychological well-being and health status of older veterans. J Appl Gerontol. 1991;10(4):469-485.
8. Morey MC, Cowper PA, Feussner JR, et al. Evaluation of a supervised exercise program in a geriatric population. J Am Geriatr Soc. 1989;37(4):348-354.
9. Morey MC, Pieper CF, Crowley GM, Sullivan RJ, Puglisi CM. Exercise adherence and 10-year mortality in chronically ill older adults. J Am Geriatr Soc. 2002;50(12):1929-1933.
10. Morey MC, Pieper CF, Sullivan RJ Jr, Crowley GM, Cowper PA, Robbins MS. Five-year performance trends for older exercisers: a hierarchical model of endurance, strength, and flexibility. J Am Geriatr Soc. 1996;44(10):1226-1231.
11. Valencia WM, Botros D, Pendlebury D, et al. Proactive reach and telehealth monitoring (Gerofit) enhance resistance exercise at rural setting. Innovat Aging. 2017;1(suppl 1):225.12. Pendlebury D, Botros D VW. Proactive Reach: an innovative access approach to identify & deliver GEROFIT exercise telehealth counseling to rural veterans & enhance CBOC services. J Am Geriatr Soc. 2017(suppl 1):S208. Poster presented at: Annual Scientific Meeting of the American Geriatrics Society; May 18, 2017; San Antonio, TX.
13. Morey MC, Crowley GM, Robbins MS, Cowper PA, Sullivan RJ Jr. The Gerofit program: a VA innovation. South Med J. 1994;87(5):S83-S87.
14. Cooper R, Kuh D, Hardy R; Mortality Review Group; FALCon and HALCyon Study Teams. Objectively measured physical capability levels and mortality: systematic review and meta-analysis. BMJ. 2010;341:c4467.
15. Rikli RE, Jones CJ. Development and validation of a functional fitness test for community-residing older adults. J Aging Phys Act. 1999;7(2):129-161.
16. Guralnik JM, Simonsick EM, Ferrucci L, et al. A short physical performance battery assessing lower extremity function: association with self-reported disability and prediction of mortality and nursing home admission. J Gerontol. 1994;49(2):M85-M94.
17. Morey MC, Cowper PA, Feussner JR, et al. Two-year trends in physical performance following supervised exercise among community-dwelling older veterans. J Am Geriatr Soc. 1991;39(10):986-992.
18. Morey MC, Lee CC, Castle S, et al. Should structured exercise be promoted as a model of care? Dissemination of the Department of Veterans Affairs Gerofit program. J Am Geriatr Soc. 2018;66(5):1009-1016.
19. Blanchard E, Castle S, Ines E, et al. Delivering a clinical exercise program to rural veterans via video telehealth. Poster C167 presented at: Annual Scientific Meeting of the American Geriatrics Society; May 19-21, 2016; Long Beach, CA.
20. Riebe D, Ehrman JK, Liguori G, Magal M, eds; American College of Sports Medicine. ACSM’s Guidelines for Exercise Testing and Prescription. 10th ed. Philadelphia, PA: Wolters Kluwer Health; 2018.
21. Peters DM, Fritz SL, Krotish DE. Assessing the reliability and validity of a shorter walk test compared with the 10-meter walk test for measurements of gait speed in healthy, older adults. J Geriatr Phys Ther. 2013;36(1):24-30.
22. Pedrosa R, Holanda G. Correlation between the walk, 2-minute step and TUG tests among hypertensive older women. Rev Bras Fisioter. 2009;13(3):252-256.
23. Romero-Arenas S, Blazevich AJ, Martinez-Pascual M, et al. Effects of high-resistance circuit training in an elderly population. Exp Gerontol. 2013;48(3):334-340.
24. Brentano MA, Cadore EL, Da Silva EM, et al. Physiological adaptations to strength and circuit training in postmenopausal women with bone loss. J Strength Cond Res. 2008;22(6):1816-1825.
25. Romero-Arenas S, Martinez-Pascual M, Alcaraz PE. Impact of resistance circuit training on neuromuscular, cardiorespiratory and body composition adaptations in the elderly. Aging Dis. 2013;4(5):256-263.
26. Paoli A, Pacelli F, Bargossi AM, et al. Effects of three distinct protocols of fitness training on body composition, strength and blood lactate. J Sports Med Phys Fitness. 2010;50(1):43-51.
27. Takeshima N, Rogers ME, Islam MM, Yamauchi T, Watanabe E, Okada A. Effect of concurrent aerobic and resistance circuit exercise training on fitness in older adults. Eur J Appl Physiol. 2004;93(1-2):173-182.
28. Giné-Garriga M, Guerra M, Pagés E, Manini TM, Jiménez R, Unnithan VB. The effect of functional circuit training on physical frailty in frail older adults: a randomized controlled trial. J Aging Phys Act. 2010;18(4):401-424.
29. Biddle ED, Reynolds P, Kopp T, Cammarata H, Conway P. Implementation of functional training tools elicits improvements in aerobic fitness and lower body strength in older veterans. Poster C169 presented at: Annual Scientific Meeting of the American Geriatrics Society; May 19-21, 2016; Long Beach, CA.