User login
AHRQ's Director Looks to Hospitalists to Help Reduce Readmissions
Although a recently released study of Medicare data uncovers little progress in reducing hospital readmissions, and the Oct. 1 deadline to implement CMS’ Hospital Readmissions Reduction Program looms, Carolyn Clancy, MD, director of the Agency for Healthcare Research and Quality (AHRQ), says she's not worried about the ability of America’s hospitalists to rise to the occasion and get a handle on the problem.
Dr. Clancy recently wrote a commentary outlining the government's approach to controlling readmissions, stating that taking aim at readmissions is 1) an integral component of its value-based purchasing program and 2) is an opportunity for improving hospital quality and patient safety.
"Hospitalists are often on the receiving end of hospitalizations resulting from poor coordination of care. I think it would be very exciting to be part of the solution," Dr. Clancy says. She says she observed firsthand during a recent hospital stay how hospitalists helped her to think about how she should care for herself after returning home. But her father suffered a needless rehospitalization when important information (how much Coumadin to take) was miscommunicated in a post-discharge follow-up phone call, causing him to start bleeding.
"Hospitalists who want to embrace the challenge will find a phenomenal amount of information on Innovations Exchange, where people from all over America are sharing their clinical innovations."
Dr. Clancy says she hopes AHRQ-supported tools and studies "will make it easier for hospitals to do the right thing."
Although a recently released study of Medicare data uncovers little progress in reducing hospital readmissions, and the Oct. 1 deadline to implement CMS’ Hospital Readmissions Reduction Program looms, Carolyn Clancy, MD, director of the Agency for Healthcare Research and Quality (AHRQ), says she's not worried about the ability of America’s hospitalists to rise to the occasion and get a handle on the problem.
Dr. Clancy recently wrote a commentary outlining the government's approach to controlling readmissions, stating that taking aim at readmissions is 1) an integral component of its value-based purchasing program and 2) is an opportunity for improving hospital quality and patient safety.
"Hospitalists are often on the receiving end of hospitalizations resulting from poor coordination of care. I think it would be very exciting to be part of the solution," Dr. Clancy says. She says she observed firsthand during a recent hospital stay how hospitalists helped her to think about how she should care for herself after returning home. But her father suffered a needless rehospitalization when important information (how much Coumadin to take) was miscommunicated in a post-discharge follow-up phone call, causing him to start bleeding.
"Hospitalists who want to embrace the challenge will find a phenomenal amount of information on Innovations Exchange, where people from all over America are sharing their clinical innovations."
Dr. Clancy says she hopes AHRQ-supported tools and studies "will make it easier for hospitals to do the right thing."
Although a recently released study of Medicare data uncovers little progress in reducing hospital readmissions, and the Oct. 1 deadline to implement CMS’ Hospital Readmissions Reduction Program looms, Carolyn Clancy, MD, director of the Agency for Healthcare Research and Quality (AHRQ), says she's not worried about the ability of America’s hospitalists to rise to the occasion and get a handle on the problem.
Dr. Clancy recently wrote a commentary outlining the government's approach to controlling readmissions, stating that taking aim at readmissions is 1) an integral component of its value-based purchasing program and 2) is an opportunity for improving hospital quality and patient safety.
"Hospitalists are often on the receiving end of hospitalizations resulting from poor coordination of care. I think it would be very exciting to be part of the solution," Dr. Clancy says. She says she observed firsthand during a recent hospital stay how hospitalists helped her to think about how she should care for herself after returning home. But her father suffered a needless rehospitalization when important information (how much Coumadin to take) was miscommunicated in a post-discharge follow-up phone call, causing him to start bleeding.
"Hospitalists who want to embrace the challenge will find a phenomenal amount of information on Innovations Exchange, where people from all over America are sharing their clinical innovations."
Dr. Clancy says she hopes AHRQ-supported tools and studies "will make it easier for hospitals to do the right thing."
ONLINE EXCLUSIVE: Elbert Huang discusses primary care's role in providing access and value
Click here to listen to Dr. Huang
Click here to listen to Dr. Huang
Click here to listen to Dr. Huang
Nutrition and exercise in cancer survivors
Obesity has reached epidemic proportions in the United States in the past 2 decades. According to a recent report, 36% of the adult population currently has a body mass index of more than 30 kg/m2, which is the diagnostic for obesity.1 If we focus only on the US adult cancer survivor population, then the magnitude of being overweight or obese is notably higher, ranging from 52% to 68%.2 In adult survivors of childhood cancer, several factors are associated with increased risk for obesity, such as hypothalamic or pituitary radiation, the use of certain antidepressants, and lifestyle factors.3
*For a PDF of the full article, click on the link to the left of this introduction.
Obesity has reached epidemic proportions in the United States in the past 2 decades. According to a recent report, 36% of the adult population currently has a body mass index of more than 30 kg/m2, which is the diagnostic for obesity.1 If we focus only on the US adult cancer survivor population, then the magnitude of being overweight or obese is notably higher, ranging from 52% to 68%.2 In adult survivors of childhood cancer, several factors are associated with increased risk for obesity, such as hypothalamic or pituitary radiation, the use of certain antidepressants, and lifestyle factors.3
*For a PDF of the full article, click on the link to the left of this introduction.
Obesity has reached epidemic proportions in the United States in the past 2 decades. According to a recent report, 36% of the adult population currently has a body mass index of more than 30 kg/m2, which is the diagnostic for obesity.1 If we focus only on the US adult cancer survivor population, then the magnitude of being overweight or obese is notably higher, ranging from 52% to 68%.2 In adult survivors of childhood cancer, several factors are associated with increased risk for obesity, such as hypothalamic or pituitary radiation, the use of certain antidepressants, and lifestyle factors.3
*For a PDF of the full article, click on the link to the left of this introduction.
Workforce Shortages, Increased Patient Populations, and Funding Woes Pressure U.S. Primary-Care System
It’s been about 15 years since the last surge of interest in primary care as a career, when U.S. medical graduates temporarily reversed a long decline by flocking to family medicine, general internal medicine, and pediatrics. Newly minted doctors responded enthusiastically to a widely held perception in the mid-1990s that primary care would be central to a brave new paradigm of managed healthcare delivery.
That profound change never materialized, and the primary-care workforce has since resumed a downward slide that is sounding alarm bells throughout the country. Even more distressing, the medical profession’s recent misfortunes have spread far beyond the doctor’s office.
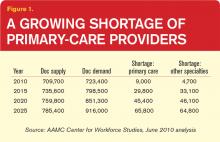
“What we’re looking at now is that there’s a shortage of somewhere around 90,000 physicians in the next 10 years, increasing in the five years beyond that to 125,000 or more,” says Atul Grover, MD, PhD, chief public policy officer of the Association of American Medical Colleges. The association’s estimates suggest that the 10- and 15-year shortfalls will be split nearly evenly between primary care and other specialties.
Hospitalists could feel that widening gap as well. With increasing numbers of aging baby boomers reaching Medicare eligibility and 32 million Americans set to join the ranks of the insured by 2019 through the Affordable Care Act, primary care’s difficulties arguably are the closest to a full-blown crisis. “Primary care in the United States needs a lifeline,” began a 2009 editorial in the New England Journal of Medicine.1 And that was before an estimate suggesting that new insurance mandates will require an additional 4,307 to 6,940 primary-care physicians to meet demand before the end of the decade contributing about 15% to the expected shortfall.2
Why should hospitalists care about the fate of their counterparts? For starters, what’s good for outpatient providers is good for a sound healthcare system. Researchers have linked strong
primary care to lower overall spending, fewer health disparities, and higher quality of care.3
Hospitalists and primary-care physicians (PCPs) also are inexorably linked. They follow similar training and education pathways, and need each other to ensure safe transitions of care. And despite the evidence pointing to a slew of contributing factors, HM regularly is blamed for many of primary care’s mounting woes.
Based on well-functioning healthcare systems around the world, analysts say the ideal primary-care-to-specialty-care-provider ratio should be roughly 40:60 or 50:50. According to Kathleen Klink, MD, director of the Division of Medicine and Dentistry in the Health Resources and Services Administration (HRSA), only about 32% of physicians in the U.S. are practicing primary care. Unless something changes, that percentage will erode even further. “We’re going in the wrong direction,” Dr. Klink says.
Opinions differ on the extent of the current PCP shortage. Nevertheless, there is clearly a “huge maldistribution problem,” says Robert Phillips, MD, MSPH, director of the Washington, D.C.-based Robert Graham Center, a primary-care research center. Rural and underserved areas already are being shortchanged as more doctors locate in more affluent and desirable areas, he says.
That phenomenon is hardly unique to primary care, but Dr. Phillips says the deficit in frontline doctors could cause disproportionately more hardships in rural and underserved communities given the shrinking pipeline for medical trainees. A decade ago, almost a third of all medical graduates were placed into primary-care residency training slots. Now, he says, that figure is a bit less than 22%. “We can’t even replace the primary-care workforce we have now with that kind of output,” Dr. Phillips says.
Already, many doctors are no longer accepting new Medicaid or Medicare patients because their practices are losing money from low reimbursement rates. The Affordable Care Act’s significant expansion of insurance benefits, Dr. Grover says, will effectively accelerate the timetable of growing imbalances between supply and demand. “I think the challenge you face is, Will the ACA efforts to expand access fail because you’re giving people an insurance card but you have nobody there to take care of them?”
Reasons Aplenty
Some medical students simply aren’t interested in primary care. For the rest, however, interviews with doctors, analysts, and federal officials suggest that the pipeline has been battered throughout its length. Of all the contributing factors, Dr. Phillips says, the main one might be income disparity. In a 2009 study, the center found that the growing gulf in salaries between primary care and subspecialty medicine “cuts in half the likelihood that a student will choose to go into primary care,” he says. Over a career, that gap translates into a difference of $3.5 million. “It dissuades them strongly,” Dr. Phillips says.
At the same time, medical school tuitions have increased at a rate far outstripping the consumer price index. “What we found is that when you hit somewhere between $200,000 and $250,000 in debt, that’s where you see the dropoff really happen,” he says. “Because it becomes almost unfathomable that you can, on a primary-care income, pay off your debts without it severely cutting into your lifestyle.”
Lori Heim, MD, former president of the American Academy of Family Physicians and a hospitalist at Scotland Memorial Hospital in Laurinburg, N.C., says the prevailing fee-for-service payment model has failed primary-care providers, requiring them to work more to meet soaring outpatient demand but reimbursing them less. “People talk about the hamster wheel,” she says. “And that has created more workplace dissatisfaction. Not only does it impact students, but it also impacts the number of primary-care physicians who want to stay in the community, practicing.”
Frederick Chen, MD, MPH, associate professor of family medicine at the University of Washington in Seattle, can relate. “I came from community practice, where you’re seeing 30 to 35 patients a day, and the pressure was entirely on your productivity, and that’s not fun,” he says. “So we’re burning out a lot of primary-care physicians, and students are seeing that very easily.”
The larger theme, several doctors say, is one of perceived worth. Leora Horwitz, MD, assistant professor of internal medicine at Yale School of Medicine in New Haven, Conn., says she has to think holistically about her patients’ symptoms, medication lists, family history, home situation, and other factors during her limited time with them. She bristles at the notion that specialists might spend their time considering only one aspect of her patients’ care yet bill twice as much.
“Realistically, I am providing better value to the healthcare system than a specialist does, and yet we pay specialists much more,” she says. “And until that’s different, people go where the money is and they also go where the respect is, and I think it’s going to be very hard to recruit more people to primary care.”
Despite research pointing to financial concerns, lifestyle perceptions, and training inadequacies as key factors in the decline of primary care, perceptions that HM is poaching young talent have been hard to shake. A recent article in The Atlantic asserts that HM might be a “rational choice” given the profession’s more favorable training, lifestyle, and financial considerations.4 The author, a general internist, contrasts those enticements “to the realities of office practice: Fifteen-minute visits with patients on multiple medications, oodles of paperwork that cause office docs to run a gauntlet just to get through their day, and more documentation and regulatory burdens than ever before.”
Nevertheless, the article describes PCPs who resist hospitals’ calls to move to a hospitalist system as honorable “holdouts” who are committed to being directly involved in their patients’ care.
In her blog post at KevinMD.com, “Hospitalists are Killing Primary Care, and other Myths Debunked,” Vineet Arora, MD, MPP, FHM, a hospitalist at the University of Chicago, addresses those perceptions head-on. “If hospitalists did not exist, there would still be declining interest in primary care among medical students and residents,” she writes.
In a subsequent interview, Dr. Arora contends that both primary care and HM instead might be losing out to higher-paying subspecialties, especially the “ROAD” quartet of radiology, ophthalmology, anesthesiology, and dermatology. She also questions the notion that the professions draw from the same talent pool. “Anecdotally, I can tell you that I don’t see a lot of people choosing between primary care and hospital medicine,” she says. “They’re thinking, ‘Do I want to do critical care, hospital medicine, or cardiology?’ Because the type of person who does hospital medicine is more attracted to that inpatient, acute environment.”
Dr. Horwitz agrees that the choice between a career in primary care and HM might not be as clear-cut as some detractors have suggested. Even so, she describes hospitalists as a “double-edged sword” for PCPs. “On the one hand, primary-care docs get paid so little for their outpatient visits that most need to see a high volume of patients in a day just to break even. So they have less and less time to go to the hospital to see hospitalizations,” Dr. Horwitz says. “The hospitalist movement was really a godsend in that respect, because it allowed primary-care docs to focus on their outpatient practice and not spend all that travel time going to the hospital.”
Other PCPs have lamented the erosion of their inpatient roles while recognizing that current economic realities are gradually pushing them out of the hospital. In fact, Dr. Horwitz says, PCPs often don’t know when their patients have been hospitalized, leading to a breakdown in the continuity of care. A weak primary-care infrastructure in a community, hospitalists say, can likewise imperil safe transitions. With the partitioning of inpatient and outpatient responsibilities, the potential for such miscommunications and lapses has clearly grown.
“We’re all in the same workforce; we’re all trying to take care of patients,” Dr. Heim says. “The discussion needs to be on how do we coordinate, not over turf wars.”
Signs of Life
Experts are focusing more on team-based approaches among the few potential short-term solutions, a common theme in HM circles. Advanced-practice registered nurses, physician assistants, and other providers can be trained more quickly than doctors, potentially extending the reach of primary care. In turn, the concept of team-based care could be beefed up during medical residencies.
Primary-care advocates say more equitable reimbursements also could help to ease the crisis, as would more federal support of residency training. But with many politicians focused on deficit reduction, new government incentives are debuting even as existing programs are being threatened or dismantled.
The Affordable Care Act, for example, more than doubled the capacity of the well-regarded National Health Service Corps, which provides scholarships and loan repayments to doctors who agree to practice in underserved communities. The law also created primary-care incentive payments that added $500 million to physician incomes in 2011. “So that’s a pretty strong message of value, and it’s some real value, too,” Dr. Phillips says.
—Atul Grover, MD, PhD, chief public policy officer, Association of American Medical Colleges
The Affordable Care Act, however, cuts $155 billion to hospital payments over 10 years, adding to the downward pressure on reimbursements. And President Obama’s fiscal-year 2013 budget proposal trims an additional $1 billion, or 10%, from Medicare’s annual payments for patient care, which could impact graduate medical education as hospitals seek to balance out the cuts.
Amid the challenges, primary care is showing some encouraging signs of life. Medical school enrollments are on pace to increase by 30% over their 2002 levels within the next three to five years. In both 2010 and 2011, the number of U.S. medical graduates going into family medicine increased by roughly 10% (followed by a more modest increase of 1% this year). Residency matches in general internal medicine also have been climbing. Dr. Heim and others say it’s no coincidence that students’ interest in primary care began rising again amid public discussions on healthcare reform that focused on the value of primary care.
In the end, the profession’s fate could depend in large part on whether the affirmations continue this time around. “There are some rock stars and heroes of primary care that are not as well-known to medical students as they should be,” says Elbert Huang, MD, associate professor of medicine at the University of Chicago. Highlighting some of those individual leaders, Dr. Huang believes, might significantly improve the profession’s standing among students.
“We need a Michael Jordan of primary care,” he says.
Bryn Nelson is a freelance medical writer in Seattle.
References
- Bodenheimer T, Grumbach K, Berenson RA. A lifeline for primary care. New Engl J Med. 2009;360(26):2693-2696.
- Hofer AN, Abraham JM, Moscovice I. Expansion of coverage under the Patient Protection and Affordable Care Act and primary care utilization. Milbank Q. 2011;89(1):69-89.
- Starfield B, Shi L, Macinko J. Contribution of primary care to health systems and health. Milbank Q. 2005;83(3):457-502.
- Henning Schumann, J. The doctor is out: young talent is turning away from primary care. The Atlantic; March 12, 2012.
It’s been about 15 years since the last surge of interest in primary care as a career, when U.S. medical graduates temporarily reversed a long decline by flocking to family medicine, general internal medicine, and pediatrics. Newly minted doctors responded enthusiastically to a widely held perception in the mid-1990s that primary care would be central to a brave new paradigm of managed healthcare delivery.
That profound change never materialized, and the primary-care workforce has since resumed a downward slide that is sounding alarm bells throughout the country. Even more distressing, the medical profession’s recent misfortunes have spread far beyond the doctor’s office.
“What we’re looking at now is that there’s a shortage of somewhere around 90,000 physicians in the next 10 years, increasing in the five years beyond that to 125,000 or more,” says Atul Grover, MD, PhD, chief public policy officer of the Association of American Medical Colleges. The association’s estimates suggest that the 10- and 15-year shortfalls will be split nearly evenly between primary care and other specialties.
Hospitalists could feel that widening gap as well. With increasing numbers of aging baby boomers reaching Medicare eligibility and 32 million Americans set to join the ranks of the insured by 2019 through the Affordable Care Act, primary care’s difficulties arguably are the closest to a full-blown crisis. “Primary care in the United States needs a lifeline,” began a 2009 editorial in the New England Journal of Medicine.1 And that was before an estimate suggesting that new insurance mandates will require an additional 4,307 to 6,940 primary-care physicians to meet demand before the end of the decade contributing about 15% to the expected shortfall.2
Why should hospitalists care about the fate of their counterparts? For starters, what’s good for outpatient providers is good for a sound healthcare system. Researchers have linked strong
primary care to lower overall spending, fewer health disparities, and higher quality of care.3
Hospitalists and primary-care physicians (PCPs) also are inexorably linked. They follow similar training and education pathways, and need each other to ensure safe transitions of care. And despite the evidence pointing to a slew of contributing factors, HM regularly is blamed for many of primary care’s mounting woes.
Based on well-functioning healthcare systems around the world, analysts say the ideal primary-care-to-specialty-care-provider ratio should be roughly 40:60 or 50:50. According to Kathleen Klink, MD, director of the Division of Medicine and Dentistry in the Health Resources and Services Administration (HRSA), only about 32% of physicians in the U.S. are practicing primary care. Unless something changes, that percentage will erode even further. “We’re going in the wrong direction,” Dr. Klink says.
Opinions differ on the extent of the current PCP shortage. Nevertheless, there is clearly a “huge maldistribution problem,” says Robert Phillips, MD, MSPH, director of the Washington, D.C.-based Robert Graham Center, a primary-care research center. Rural and underserved areas already are being shortchanged as more doctors locate in more affluent and desirable areas, he says.
That phenomenon is hardly unique to primary care, but Dr. Phillips says the deficit in frontline doctors could cause disproportionately more hardships in rural and underserved communities given the shrinking pipeline for medical trainees. A decade ago, almost a third of all medical graduates were placed into primary-care residency training slots. Now, he says, that figure is a bit less than 22%. “We can’t even replace the primary-care workforce we have now with that kind of output,” Dr. Phillips says.
Already, many doctors are no longer accepting new Medicaid or Medicare patients because their practices are losing money from low reimbursement rates. The Affordable Care Act’s significant expansion of insurance benefits, Dr. Grover says, will effectively accelerate the timetable of growing imbalances between supply and demand. “I think the challenge you face is, Will the ACA efforts to expand access fail because you’re giving people an insurance card but you have nobody there to take care of them?”
Reasons Aplenty
Some medical students simply aren’t interested in primary care. For the rest, however, interviews with doctors, analysts, and federal officials suggest that the pipeline has been battered throughout its length. Of all the contributing factors, Dr. Phillips says, the main one might be income disparity. In a 2009 study, the center found that the growing gulf in salaries between primary care and subspecialty medicine “cuts in half the likelihood that a student will choose to go into primary care,” he says. Over a career, that gap translates into a difference of $3.5 million. “It dissuades them strongly,” Dr. Phillips says.
At the same time, medical school tuitions have increased at a rate far outstripping the consumer price index. “What we found is that when you hit somewhere between $200,000 and $250,000 in debt, that’s where you see the dropoff really happen,” he says. “Because it becomes almost unfathomable that you can, on a primary-care income, pay off your debts without it severely cutting into your lifestyle.”
Lori Heim, MD, former president of the American Academy of Family Physicians and a hospitalist at Scotland Memorial Hospital in Laurinburg, N.C., says the prevailing fee-for-service payment model has failed primary-care providers, requiring them to work more to meet soaring outpatient demand but reimbursing them less. “People talk about the hamster wheel,” she says. “And that has created more workplace dissatisfaction. Not only does it impact students, but it also impacts the number of primary-care physicians who want to stay in the community, practicing.”
Frederick Chen, MD, MPH, associate professor of family medicine at the University of Washington in Seattle, can relate. “I came from community practice, where you’re seeing 30 to 35 patients a day, and the pressure was entirely on your productivity, and that’s not fun,” he says. “So we’re burning out a lot of primary-care physicians, and students are seeing that very easily.”
The larger theme, several doctors say, is one of perceived worth. Leora Horwitz, MD, assistant professor of internal medicine at Yale School of Medicine in New Haven, Conn., says she has to think holistically about her patients’ symptoms, medication lists, family history, home situation, and other factors during her limited time with them. She bristles at the notion that specialists might spend their time considering only one aspect of her patients’ care yet bill twice as much.
“Realistically, I am providing better value to the healthcare system than a specialist does, and yet we pay specialists much more,” she says. “And until that’s different, people go where the money is and they also go where the respect is, and I think it’s going to be very hard to recruit more people to primary care.”
Despite research pointing to financial concerns, lifestyle perceptions, and training inadequacies as key factors in the decline of primary care, perceptions that HM is poaching young talent have been hard to shake. A recent article in The Atlantic asserts that HM might be a “rational choice” given the profession’s more favorable training, lifestyle, and financial considerations.4 The author, a general internist, contrasts those enticements “to the realities of office practice: Fifteen-minute visits with patients on multiple medications, oodles of paperwork that cause office docs to run a gauntlet just to get through their day, and more documentation and regulatory burdens than ever before.”
Nevertheless, the article describes PCPs who resist hospitals’ calls to move to a hospitalist system as honorable “holdouts” who are committed to being directly involved in their patients’ care.
In her blog post at KevinMD.com, “Hospitalists are Killing Primary Care, and other Myths Debunked,” Vineet Arora, MD, MPP, FHM, a hospitalist at the University of Chicago, addresses those perceptions head-on. “If hospitalists did not exist, there would still be declining interest in primary care among medical students and residents,” she writes.
In a subsequent interview, Dr. Arora contends that both primary care and HM instead might be losing out to higher-paying subspecialties, especially the “ROAD” quartet of radiology, ophthalmology, anesthesiology, and dermatology. She also questions the notion that the professions draw from the same talent pool. “Anecdotally, I can tell you that I don’t see a lot of people choosing between primary care and hospital medicine,” she says. “They’re thinking, ‘Do I want to do critical care, hospital medicine, or cardiology?’ Because the type of person who does hospital medicine is more attracted to that inpatient, acute environment.”
Dr. Horwitz agrees that the choice between a career in primary care and HM might not be as clear-cut as some detractors have suggested. Even so, she describes hospitalists as a “double-edged sword” for PCPs. “On the one hand, primary-care docs get paid so little for their outpatient visits that most need to see a high volume of patients in a day just to break even. So they have less and less time to go to the hospital to see hospitalizations,” Dr. Horwitz says. “The hospitalist movement was really a godsend in that respect, because it allowed primary-care docs to focus on their outpatient practice and not spend all that travel time going to the hospital.”
Other PCPs have lamented the erosion of their inpatient roles while recognizing that current economic realities are gradually pushing them out of the hospital. In fact, Dr. Horwitz says, PCPs often don’t know when their patients have been hospitalized, leading to a breakdown in the continuity of care. A weak primary-care infrastructure in a community, hospitalists say, can likewise imperil safe transitions. With the partitioning of inpatient and outpatient responsibilities, the potential for such miscommunications and lapses has clearly grown.
“We’re all in the same workforce; we’re all trying to take care of patients,” Dr. Heim says. “The discussion needs to be on how do we coordinate, not over turf wars.”
Signs of Life
Experts are focusing more on team-based approaches among the few potential short-term solutions, a common theme in HM circles. Advanced-practice registered nurses, physician assistants, and other providers can be trained more quickly than doctors, potentially extending the reach of primary care. In turn, the concept of team-based care could be beefed up during medical residencies.
Primary-care advocates say more equitable reimbursements also could help to ease the crisis, as would more federal support of residency training. But with many politicians focused on deficit reduction, new government incentives are debuting even as existing programs are being threatened or dismantled.
The Affordable Care Act, for example, more than doubled the capacity of the well-regarded National Health Service Corps, which provides scholarships and loan repayments to doctors who agree to practice in underserved communities. The law also created primary-care incentive payments that added $500 million to physician incomes in 2011. “So that’s a pretty strong message of value, and it’s some real value, too,” Dr. Phillips says.
—Atul Grover, MD, PhD, chief public policy officer, Association of American Medical Colleges
The Affordable Care Act, however, cuts $155 billion to hospital payments over 10 years, adding to the downward pressure on reimbursements. And President Obama’s fiscal-year 2013 budget proposal trims an additional $1 billion, or 10%, from Medicare’s annual payments for patient care, which could impact graduate medical education as hospitals seek to balance out the cuts.
Amid the challenges, primary care is showing some encouraging signs of life. Medical school enrollments are on pace to increase by 30% over their 2002 levels within the next three to five years. In both 2010 and 2011, the number of U.S. medical graduates going into family medicine increased by roughly 10% (followed by a more modest increase of 1% this year). Residency matches in general internal medicine also have been climbing. Dr. Heim and others say it’s no coincidence that students’ interest in primary care began rising again amid public discussions on healthcare reform that focused on the value of primary care.
In the end, the profession’s fate could depend in large part on whether the affirmations continue this time around. “There are some rock stars and heroes of primary care that are not as well-known to medical students as they should be,” says Elbert Huang, MD, associate professor of medicine at the University of Chicago. Highlighting some of those individual leaders, Dr. Huang believes, might significantly improve the profession’s standing among students.
“We need a Michael Jordan of primary care,” he says.
Bryn Nelson is a freelance medical writer in Seattle.
References
- Bodenheimer T, Grumbach K, Berenson RA. A lifeline for primary care. New Engl J Med. 2009;360(26):2693-2696.
- Hofer AN, Abraham JM, Moscovice I. Expansion of coverage under the Patient Protection and Affordable Care Act and primary care utilization. Milbank Q. 2011;89(1):69-89.
- Starfield B, Shi L, Macinko J. Contribution of primary care to health systems and health. Milbank Q. 2005;83(3):457-502.
- Henning Schumann, J. The doctor is out: young talent is turning away from primary care. The Atlantic; March 12, 2012.
It’s been about 15 years since the last surge of interest in primary care as a career, when U.S. medical graduates temporarily reversed a long decline by flocking to family medicine, general internal medicine, and pediatrics. Newly minted doctors responded enthusiastically to a widely held perception in the mid-1990s that primary care would be central to a brave new paradigm of managed healthcare delivery.
That profound change never materialized, and the primary-care workforce has since resumed a downward slide that is sounding alarm bells throughout the country. Even more distressing, the medical profession’s recent misfortunes have spread far beyond the doctor’s office.
“What we’re looking at now is that there’s a shortage of somewhere around 90,000 physicians in the next 10 years, increasing in the five years beyond that to 125,000 or more,” says Atul Grover, MD, PhD, chief public policy officer of the Association of American Medical Colleges. The association’s estimates suggest that the 10- and 15-year shortfalls will be split nearly evenly between primary care and other specialties.
Hospitalists could feel that widening gap as well. With increasing numbers of aging baby boomers reaching Medicare eligibility and 32 million Americans set to join the ranks of the insured by 2019 through the Affordable Care Act, primary care’s difficulties arguably are the closest to a full-blown crisis. “Primary care in the United States needs a lifeline,” began a 2009 editorial in the New England Journal of Medicine.1 And that was before an estimate suggesting that new insurance mandates will require an additional 4,307 to 6,940 primary-care physicians to meet demand before the end of the decade contributing about 15% to the expected shortfall.2
Why should hospitalists care about the fate of their counterparts? For starters, what’s good for outpatient providers is good for a sound healthcare system. Researchers have linked strong
primary care to lower overall spending, fewer health disparities, and higher quality of care.3
Hospitalists and primary-care physicians (PCPs) also are inexorably linked. They follow similar training and education pathways, and need each other to ensure safe transitions of care. And despite the evidence pointing to a slew of contributing factors, HM regularly is blamed for many of primary care’s mounting woes.
Based on well-functioning healthcare systems around the world, analysts say the ideal primary-care-to-specialty-care-provider ratio should be roughly 40:60 or 50:50. According to Kathleen Klink, MD, director of the Division of Medicine and Dentistry in the Health Resources and Services Administration (HRSA), only about 32% of physicians in the U.S. are practicing primary care. Unless something changes, that percentage will erode even further. “We’re going in the wrong direction,” Dr. Klink says.
Opinions differ on the extent of the current PCP shortage. Nevertheless, there is clearly a “huge maldistribution problem,” says Robert Phillips, MD, MSPH, director of the Washington, D.C.-based Robert Graham Center, a primary-care research center. Rural and underserved areas already are being shortchanged as more doctors locate in more affluent and desirable areas, he says.
That phenomenon is hardly unique to primary care, but Dr. Phillips says the deficit in frontline doctors could cause disproportionately more hardships in rural and underserved communities given the shrinking pipeline for medical trainees. A decade ago, almost a third of all medical graduates were placed into primary-care residency training slots. Now, he says, that figure is a bit less than 22%. “We can’t even replace the primary-care workforce we have now with that kind of output,” Dr. Phillips says.
Already, many doctors are no longer accepting new Medicaid or Medicare patients because their practices are losing money from low reimbursement rates. The Affordable Care Act’s significant expansion of insurance benefits, Dr. Grover says, will effectively accelerate the timetable of growing imbalances between supply and demand. “I think the challenge you face is, Will the ACA efforts to expand access fail because you’re giving people an insurance card but you have nobody there to take care of them?”
Reasons Aplenty
Some medical students simply aren’t interested in primary care. For the rest, however, interviews with doctors, analysts, and federal officials suggest that the pipeline has been battered throughout its length. Of all the contributing factors, Dr. Phillips says, the main one might be income disparity. In a 2009 study, the center found that the growing gulf in salaries between primary care and subspecialty medicine “cuts in half the likelihood that a student will choose to go into primary care,” he says. Over a career, that gap translates into a difference of $3.5 million. “It dissuades them strongly,” Dr. Phillips says.
At the same time, medical school tuitions have increased at a rate far outstripping the consumer price index. “What we found is that when you hit somewhere between $200,000 and $250,000 in debt, that’s where you see the dropoff really happen,” he says. “Because it becomes almost unfathomable that you can, on a primary-care income, pay off your debts without it severely cutting into your lifestyle.”
Lori Heim, MD, former president of the American Academy of Family Physicians and a hospitalist at Scotland Memorial Hospital in Laurinburg, N.C., says the prevailing fee-for-service payment model has failed primary-care providers, requiring them to work more to meet soaring outpatient demand but reimbursing them less. “People talk about the hamster wheel,” she says. “And that has created more workplace dissatisfaction. Not only does it impact students, but it also impacts the number of primary-care physicians who want to stay in the community, practicing.”
Frederick Chen, MD, MPH, associate professor of family medicine at the University of Washington in Seattle, can relate. “I came from community practice, where you’re seeing 30 to 35 patients a day, and the pressure was entirely on your productivity, and that’s not fun,” he says. “So we’re burning out a lot of primary-care physicians, and students are seeing that very easily.”
The larger theme, several doctors say, is one of perceived worth. Leora Horwitz, MD, assistant professor of internal medicine at Yale School of Medicine in New Haven, Conn., says she has to think holistically about her patients’ symptoms, medication lists, family history, home situation, and other factors during her limited time with them. She bristles at the notion that specialists might spend their time considering only one aspect of her patients’ care yet bill twice as much.
“Realistically, I am providing better value to the healthcare system than a specialist does, and yet we pay specialists much more,” she says. “And until that’s different, people go where the money is and they also go where the respect is, and I think it’s going to be very hard to recruit more people to primary care.”
Despite research pointing to financial concerns, lifestyle perceptions, and training inadequacies as key factors in the decline of primary care, perceptions that HM is poaching young talent have been hard to shake. A recent article in The Atlantic asserts that HM might be a “rational choice” given the profession’s more favorable training, lifestyle, and financial considerations.4 The author, a general internist, contrasts those enticements “to the realities of office practice: Fifteen-minute visits with patients on multiple medications, oodles of paperwork that cause office docs to run a gauntlet just to get through their day, and more documentation and regulatory burdens than ever before.”
Nevertheless, the article describes PCPs who resist hospitals’ calls to move to a hospitalist system as honorable “holdouts” who are committed to being directly involved in their patients’ care.
In her blog post at KevinMD.com, “Hospitalists are Killing Primary Care, and other Myths Debunked,” Vineet Arora, MD, MPP, FHM, a hospitalist at the University of Chicago, addresses those perceptions head-on. “If hospitalists did not exist, there would still be declining interest in primary care among medical students and residents,” she writes.
In a subsequent interview, Dr. Arora contends that both primary care and HM instead might be losing out to higher-paying subspecialties, especially the “ROAD” quartet of radiology, ophthalmology, anesthesiology, and dermatology. She also questions the notion that the professions draw from the same talent pool. “Anecdotally, I can tell you that I don’t see a lot of people choosing between primary care and hospital medicine,” she says. “They’re thinking, ‘Do I want to do critical care, hospital medicine, or cardiology?’ Because the type of person who does hospital medicine is more attracted to that inpatient, acute environment.”
Dr. Horwitz agrees that the choice between a career in primary care and HM might not be as clear-cut as some detractors have suggested. Even so, she describes hospitalists as a “double-edged sword” for PCPs. “On the one hand, primary-care docs get paid so little for their outpatient visits that most need to see a high volume of patients in a day just to break even. So they have less and less time to go to the hospital to see hospitalizations,” Dr. Horwitz says. “The hospitalist movement was really a godsend in that respect, because it allowed primary-care docs to focus on their outpatient practice and not spend all that travel time going to the hospital.”
Other PCPs have lamented the erosion of their inpatient roles while recognizing that current economic realities are gradually pushing them out of the hospital. In fact, Dr. Horwitz says, PCPs often don’t know when their patients have been hospitalized, leading to a breakdown in the continuity of care. A weak primary-care infrastructure in a community, hospitalists say, can likewise imperil safe transitions. With the partitioning of inpatient and outpatient responsibilities, the potential for such miscommunications and lapses has clearly grown.
“We’re all in the same workforce; we’re all trying to take care of patients,” Dr. Heim says. “The discussion needs to be on how do we coordinate, not over turf wars.”
Signs of Life
Experts are focusing more on team-based approaches among the few potential short-term solutions, a common theme in HM circles. Advanced-practice registered nurses, physician assistants, and other providers can be trained more quickly than doctors, potentially extending the reach of primary care. In turn, the concept of team-based care could be beefed up during medical residencies.
Primary-care advocates say more equitable reimbursements also could help to ease the crisis, as would more federal support of residency training. But with many politicians focused on deficit reduction, new government incentives are debuting even as existing programs are being threatened or dismantled.
The Affordable Care Act, for example, more than doubled the capacity of the well-regarded National Health Service Corps, which provides scholarships and loan repayments to doctors who agree to practice in underserved communities. The law also created primary-care incentive payments that added $500 million to physician incomes in 2011. “So that’s a pretty strong message of value, and it’s some real value, too,” Dr. Phillips says.
—Atul Grover, MD, PhD, chief public policy officer, Association of American Medical Colleges
The Affordable Care Act, however, cuts $155 billion to hospital payments over 10 years, adding to the downward pressure on reimbursements. And President Obama’s fiscal-year 2013 budget proposal trims an additional $1 billion, or 10%, from Medicare’s annual payments for patient care, which could impact graduate medical education as hospitals seek to balance out the cuts.
Amid the challenges, primary care is showing some encouraging signs of life. Medical school enrollments are on pace to increase by 30% over their 2002 levels within the next three to five years. In both 2010 and 2011, the number of U.S. medical graduates going into family medicine increased by roughly 10% (followed by a more modest increase of 1% this year). Residency matches in general internal medicine also have been climbing. Dr. Heim and others say it’s no coincidence that students’ interest in primary care began rising again amid public discussions on healthcare reform that focused on the value of primary care.
In the end, the profession’s fate could depend in large part on whether the affirmations continue this time around. “There are some rock stars and heroes of primary care that are not as well-known to medical students as they should be,” says Elbert Huang, MD, associate professor of medicine at the University of Chicago. Highlighting some of those individual leaders, Dr. Huang believes, might significantly improve the profession’s standing among students.
“We need a Michael Jordan of primary care,” he says.
Bryn Nelson is a freelance medical writer in Seattle.
References
- Bodenheimer T, Grumbach K, Berenson RA. A lifeline for primary care. New Engl J Med. 2009;360(26):2693-2696.
- Hofer AN, Abraham JM, Moscovice I. Expansion of coverage under the Patient Protection and Affordable Care Act and primary care utilization. Milbank Q. 2011;89(1):69-89.
- Starfield B, Shi L, Macinko J. Contribution of primary care to health systems and health. Milbank Q. 2005;83(3):457-502.
- Henning Schumann, J. The doctor is out: young talent is turning away from primary care. The Atlantic; March 12, 2012.
Most Adverse Events at Hospitals Still Go Unreported
Nearly 13 years after the release of the Institute of Medicine’s landmark report To Err Is Human, which called national attention to the rate of preventable errors in U.S. hospitals and galvanized the patient-safety movement, 6 out of every 7 hospital-based errors, accidents, and other adverse events still go unreported.
That’s the troubling conclusion of a report released in January by the Department of Health and Human Services’ Office of the Inspector General (OIG). According to the OIG, the report the National Incident Study on Adverse Events in Hospitals Among Medicare Beneficiaries, is the first truly national adverse-event study in the nation documenting hospital-based adverse events among Medicare beneficiaries.1
Previous studies assessing errors and adverse events had tracked the number of events reported, but they had no denominator; in other words, one hospital might have 1,000 reports of adverse events and errors in a given month, while another might have 50, but there was no way to know how many adverse events had been missed and how much harm had actually occurred.
The OIG study dug deeper, conducting in-depth, yearlong reviews led by independent physicians to first identify the rates of harm, then returning to the hospitals in the study where they knew harm had occurred to find out if it had been reported or not.
Overall, the inspector general estimated that more than 130,000 Medicare beneficiaries experienced one or more adverse events in hospitals in a single month. Perhaps one of the most surprising findings was that even some of the most serious, egregious adverse events were not reported. Of the 12 deaths in the sample that were iatrogenic, only two were reported as errors. More serious errors were no more likely to be captured as errors than mild reactions, such as breaking out in hives as the result of a medication.
Why aren’t more hospital-based errors and adverse events being reported, and how can this situation be changed? The OIG’s report provides some insight into the underlying problem: The dominant reason that events weren’t reported was that they weren’t perceived as adverse events. (Not all adverse events are medical errors: A medical error is a preventable adverse event.) A urinary catheter infection, while not necessarily a medical error, is always an adverse event—and yet, only one of the 17 urinary catheter infections identified by doctors in medical review was reported.
Lack of recognition—seeing adverse events and errors as just part of routine care—is only one part of the problem, says Rosemary Gibson, MSc, a former senior program officer at the Robert Wood Johnson Foundation and author of “The Treatment Trap” and “Wall of Silence.” Other issues include:
- Fear of retribution. “You have to feel safe to report someone else’s error, or your own,” Gibson says. “Most hospitals have yet to create a safety culture.”
- A sense of disbelief. “I remember talking to a hospital CEO once right after the IOM report,” Gibson recalls. “He said that after a significant error, he would get up in the morning and look in the mirror and think, ‘Did this really happen here?’ There’s almost a sense that if you don’t see it, it didn’t exist.”
- Competing pressures. “The requirements of healthcare reform have taken up so much time and energy that I fear safety has moved to the back burner,” Gibson says. “Someone in a quality and safety leadership role at one hospital said to me, ‘Safety was just a fad. We’re not doing that anymore.’”
- Productivity demands. “Healthcare’s mantra today has become volume, volume, volume. If you already have an environment that’s not as safe as we would like, and you ramp up the volume so people have to do more in the name of productivity, what’s going to happen?” asks Gibson.
The Hospitalist Perspective
Greg Maynard, MD, SFHM, director of the University of California San Diego Center for Innovation and Improvement Science (CIIS) and senior vice president of SHM’s Center for Healthcare Improvement and Innovation, says hospitalists face multiple barriers to regular reporting. Some errors, he says, are so pervasive that they are “considered routine” and “happen all the time.”
“Hypoglycemic events, for example, don’t always get entered into adverse-event reporting systems because they’re so common,” he explains, “even though you should be entering and examining them all.”
—Dan Budnitz, MD, MPH, director, Medication Safety Program, Centers for Disease Control and Prevention
Improper coding is another hospitalist-centric issue. Since Oct. 1, 2008, the Centers for Medicare & Medicaid Services (CMS) has reduced payment for acute-care inpatient cases when designated hospital-acquired conditions are not present on admission and the condition would have increased the reimbursement.
“CMS went to a lot of trouble to implement codes that could help them identify, in the coding process, patients who had an iatrogenic event,” Dr. Maynard says. “But those codes simply aren’t being used properly, and administrative coding is always a problem with regard to accuracy.”
Hospitalists can be particularly vulnerable to errors due to the nature of their work—multiple tasks, multiple team members, multiple interruptions. “[Hospitalists] are prone to the type of errors that are related to the hectic nature of being a doctor and an air traffic controller at the same time,” Dr. Maynard says. “You’re in the middle of admitting someone and get two urgent calls on two other patients, so you have to switch out of what you’re doing on that patient, issue orders on the other two, and immediately switch back. It’s hard to keep your focus when dealing with many complex patients, many of whom you’re seeing for the first time, and try to remember all the things you should not omit.”
One area that has, in recent years, become more important to hospitalists in terms of error prevention—and reporting—is discharge planning.
“When you’re sending your patient out of the hospital, they may have started on new medications or changed medications while an inpatient,” says Dan Budnitz, MD, MPH, director of the Medication Safety Program at the Centers for Disease Control and Prevention. “A recent report in the New England Journal of Medicine looked at drug-only causes of emergency hospitalizations, and the most common drugs involved are blood thinners, insulin, and some diabetes pills. The implication for the hospitalist is clear: If you start a patient on these medications in the hospital, or change them, you need to pay particular attention, because they’re two of the top drugs that may put your patient back in the hospital.”
National Models
One institution that is far ahead of the curve in error reporting is the University of Michigan Health System. In 2001, just two years after To Err is Human was published, the health system adopted a process of full disclosure of medical errors. The process involves multiple components, including an online incident reporting system, a multidisciplinary claims review committee, open and honest communication with patients and families, and quality-improvement (QI) initiatives guided by reported errors.
“They’ll get 7,000 or 8,000 reports a year from staff,” Gibson says. “That’s what you want. Most of them are minor or near-misses, and you want to know about those so you can learn from them and prevent the larger errors. But you have to create a space where it’s safe for people to report.”
She says that hospitals must adhere to the principle of “just culture” in error reporting, a philosophy that recognizes that even competent professionals make mistakes. Quality-improvement leader and Harvard School of Public Health professor Lucian Leape, MD, has said that the biggest impediment to error reporting and prevention in medicine is “that we punish people for making mistakes.”2
“If I made a mistake and anybody else in my position could have made that same mistake, the chances are the system contributed to my making that mistake, and just culture would suggest I shouldn’t bear all the blame for it,” Gibson says. “That’s different from a case in which I did something that was truly negligent. Hospitals often do not make this distinction, and [they] lack a just culture.”
—Greg Maynard, MD, SFHM, director, University of California San Diego Center for Innovation and Improvement Science, senior vice president, SHM’s Center for Healthcare Improvement and Innovation
Traditional reporting systems in healthcare, however, often do not support a just-culture model or a true patient-safety-focused approach, says Brian Nussenbaum, MD, associate professor of otolaryngology and head and neck surgery at Washington University School of Medicine in Seattle. “Paper or online reports are sent to hospital risk management departments, whose concerns are primarily to limit the potential legal risk,” he says. “There is little emphasis on systems improvement or prevention. Dissemination of incidents to others in the organization is unusual, and the impact on clinical care is often not felt by providers.”
Who should be reporting errors? The responsibility ideally lies with everyone involved in patient care, Dr. Nussenbaum says. In practice, physicians are more likely to report events that caused permanent harm or the death or near-death of a patient, but overall, they report only 1% of adverse events.3 Nurses, on the other hand, are more likely to report events that cause no harm or harm that’s only temporary, and report about 45% of events. Residents, in the same study, reported only a little more than half of adverse events to attending physicians.
“Nurses and pharmacists have a lot more training in how to fill out the voluntary reporting,” Dr. Maynard says. “Nurses and pharmacists report the overwhelming majority of adverse events and errors. I think physicians need to take some responsibility for ensuring that errors are reported into the voluntary reporting system. When I get called about something that happens on one of my patients, or detect it, I don’t necessarily enter it myself, but I will specify with whoever is calling me that it should be reported.”
Society Initiatives
SHM has taken an aggressive stance that is more focused on improving quality and safety than that of most professional organizations, according to Dr. Maynard. “We feel that the best way to build hospitalists in their careers is to increase their value by helping them take care of patients and being part of the solution to these problems,” he says. “We have quality and safety modules for learning about error and medical harm and ways to prevent it.”
SHM’s website (www.hospitalmedicine.org)
features resources and tools for DVT prevention, glycemic control, and anticoagulation—all well-known areas for medical errors. “We’re about ready to embark on putting together an adverse-drug-events module, and Project BOOST [Better Outcomes for Older Adults through Safe Transitions] provides materials to help optimize transitions of care,” Dr. Maynard says. “We want to build this into the daily fabric of the hospitalist’s career.”
At the CDC’s medication safety program, Dr. Budnitz says, his focus is less on error reporting and more on tracking backward to see what could have been the cause of harm to a patient—error or not.
“Sometimes, medication-induced harms are caused by things that we classically think of as errors, and sometimes they are not,” he says. “Error reporting is important, but sometimes things don’t fit neatly into the ‘healthcare error’ box—like when a patient misses a follow-up appointment to get their blood thinner level checked, and [the] patient has an unintended bleeding event.”
—Rosemary Gibson, MSc, author, former senior program officer, Robert Wood Johnson Foundation
Gibson believes that what’s needed is a public system for the reporting of adverse events in hospitals—like a Consumer Product Safety Commission for healthcare.
“If your child gets injured by a defective crib, there’s a system for you to report that,” she says. “We need that for hospitals. There should be an open capability for reporting events online, in person, any way you want, whether you’re on the hospital staff, a patient, or a family member. Such a system should acknowledge the report, provide for an immediate action step in urgent situations, and offer assistance and support to the person doing the reporting.”
But overall, the response in the healthcare industry to the OIG’s report has been “we need to do more research,” Gibson notes. “We don’t need more research; we need to apply what is known. There are institutions that have made pockets of progress, and patients are alive today and home with their families because we’ve made care safer, but safety still hasn’t become a system property in healthcare.”
Gina Shaw is a freelance writer in New York City.
References
- Levinson DR. Adverse events in hospitals: national incidence among Medicare beneficiaries. U.S. Department of Health and Human Services’ Office of Inspector General website. Available at: http://oig.hhs.gov/oei/reports/oei-06-09-00090.pdf. Accessed May 31, 2012.
- Leape LL. Testimony, United States Congress, House Committee on Veterans’ Affairs; Oct. 12, 1997.
- Rowin EJ. Does error and adverse event reporting by physicians and nurses differ? Jt Comm J Qual Patient Saf. 2008;34(9):537-545.
Nearly 13 years after the release of the Institute of Medicine’s landmark report To Err Is Human, which called national attention to the rate of preventable errors in U.S. hospitals and galvanized the patient-safety movement, 6 out of every 7 hospital-based errors, accidents, and other adverse events still go unreported.
That’s the troubling conclusion of a report released in January by the Department of Health and Human Services’ Office of the Inspector General (OIG). According to the OIG, the report the National Incident Study on Adverse Events in Hospitals Among Medicare Beneficiaries, is the first truly national adverse-event study in the nation documenting hospital-based adverse events among Medicare beneficiaries.1
Previous studies assessing errors and adverse events had tracked the number of events reported, but they had no denominator; in other words, one hospital might have 1,000 reports of adverse events and errors in a given month, while another might have 50, but there was no way to know how many adverse events had been missed and how much harm had actually occurred.
The OIG study dug deeper, conducting in-depth, yearlong reviews led by independent physicians to first identify the rates of harm, then returning to the hospitals in the study where they knew harm had occurred to find out if it had been reported or not.
Overall, the inspector general estimated that more than 130,000 Medicare beneficiaries experienced one or more adverse events in hospitals in a single month. Perhaps one of the most surprising findings was that even some of the most serious, egregious adverse events were not reported. Of the 12 deaths in the sample that were iatrogenic, only two were reported as errors. More serious errors were no more likely to be captured as errors than mild reactions, such as breaking out in hives as the result of a medication.
Why aren’t more hospital-based errors and adverse events being reported, and how can this situation be changed? The OIG’s report provides some insight into the underlying problem: The dominant reason that events weren’t reported was that they weren’t perceived as adverse events. (Not all adverse events are medical errors: A medical error is a preventable adverse event.) A urinary catheter infection, while not necessarily a medical error, is always an adverse event—and yet, only one of the 17 urinary catheter infections identified by doctors in medical review was reported.
Lack of recognition—seeing adverse events and errors as just part of routine care—is only one part of the problem, says Rosemary Gibson, MSc, a former senior program officer at the Robert Wood Johnson Foundation and author of “The Treatment Trap” and “Wall of Silence.” Other issues include:
- Fear of retribution. “You have to feel safe to report someone else’s error, or your own,” Gibson says. “Most hospitals have yet to create a safety culture.”
- A sense of disbelief. “I remember talking to a hospital CEO once right after the IOM report,” Gibson recalls. “He said that after a significant error, he would get up in the morning and look in the mirror and think, ‘Did this really happen here?’ There’s almost a sense that if you don’t see it, it didn’t exist.”
- Competing pressures. “The requirements of healthcare reform have taken up so much time and energy that I fear safety has moved to the back burner,” Gibson says. “Someone in a quality and safety leadership role at one hospital said to me, ‘Safety was just a fad. We’re not doing that anymore.’”
- Productivity demands. “Healthcare’s mantra today has become volume, volume, volume. If you already have an environment that’s not as safe as we would like, and you ramp up the volume so people have to do more in the name of productivity, what’s going to happen?” asks Gibson.
The Hospitalist Perspective
Greg Maynard, MD, SFHM, director of the University of California San Diego Center for Innovation and Improvement Science (CIIS) and senior vice president of SHM’s Center for Healthcare Improvement and Innovation, says hospitalists face multiple barriers to regular reporting. Some errors, he says, are so pervasive that they are “considered routine” and “happen all the time.”
“Hypoglycemic events, for example, don’t always get entered into adverse-event reporting systems because they’re so common,” he explains, “even though you should be entering and examining them all.”
—Dan Budnitz, MD, MPH, director, Medication Safety Program, Centers for Disease Control and Prevention
Improper coding is another hospitalist-centric issue. Since Oct. 1, 2008, the Centers for Medicare & Medicaid Services (CMS) has reduced payment for acute-care inpatient cases when designated hospital-acquired conditions are not present on admission and the condition would have increased the reimbursement.
“CMS went to a lot of trouble to implement codes that could help them identify, in the coding process, patients who had an iatrogenic event,” Dr. Maynard says. “But those codes simply aren’t being used properly, and administrative coding is always a problem with regard to accuracy.”
Hospitalists can be particularly vulnerable to errors due to the nature of their work—multiple tasks, multiple team members, multiple interruptions. “[Hospitalists] are prone to the type of errors that are related to the hectic nature of being a doctor and an air traffic controller at the same time,” Dr. Maynard says. “You’re in the middle of admitting someone and get two urgent calls on two other patients, so you have to switch out of what you’re doing on that patient, issue orders on the other two, and immediately switch back. It’s hard to keep your focus when dealing with many complex patients, many of whom you’re seeing for the first time, and try to remember all the things you should not omit.”
One area that has, in recent years, become more important to hospitalists in terms of error prevention—and reporting—is discharge planning.
“When you’re sending your patient out of the hospital, they may have started on new medications or changed medications while an inpatient,” says Dan Budnitz, MD, MPH, director of the Medication Safety Program at the Centers for Disease Control and Prevention. “A recent report in the New England Journal of Medicine looked at drug-only causes of emergency hospitalizations, and the most common drugs involved are blood thinners, insulin, and some diabetes pills. The implication for the hospitalist is clear: If you start a patient on these medications in the hospital, or change them, you need to pay particular attention, because they’re two of the top drugs that may put your patient back in the hospital.”
National Models
One institution that is far ahead of the curve in error reporting is the University of Michigan Health System. In 2001, just two years after To Err is Human was published, the health system adopted a process of full disclosure of medical errors. The process involves multiple components, including an online incident reporting system, a multidisciplinary claims review committee, open and honest communication with patients and families, and quality-improvement (QI) initiatives guided by reported errors.
“They’ll get 7,000 or 8,000 reports a year from staff,” Gibson says. “That’s what you want. Most of them are minor or near-misses, and you want to know about those so you can learn from them and prevent the larger errors. But you have to create a space where it’s safe for people to report.”
She says that hospitals must adhere to the principle of “just culture” in error reporting, a philosophy that recognizes that even competent professionals make mistakes. Quality-improvement leader and Harvard School of Public Health professor Lucian Leape, MD, has said that the biggest impediment to error reporting and prevention in medicine is “that we punish people for making mistakes.”2
“If I made a mistake and anybody else in my position could have made that same mistake, the chances are the system contributed to my making that mistake, and just culture would suggest I shouldn’t bear all the blame for it,” Gibson says. “That’s different from a case in which I did something that was truly negligent. Hospitals often do not make this distinction, and [they] lack a just culture.”
—Greg Maynard, MD, SFHM, director, University of California San Diego Center for Innovation and Improvement Science, senior vice president, SHM’s Center for Healthcare Improvement and Innovation
Traditional reporting systems in healthcare, however, often do not support a just-culture model or a true patient-safety-focused approach, says Brian Nussenbaum, MD, associate professor of otolaryngology and head and neck surgery at Washington University School of Medicine in Seattle. “Paper or online reports are sent to hospital risk management departments, whose concerns are primarily to limit the potential legal risk,” he says. “There is little emphasis on systems improvement or prevention. Dissemination of incidents to others in the organization is unusual, and the impact on clinical care is often not felt by providers.”
Who should be reporting errors? The responsibility ideally lies with everyone involved in patient care, Dr. Nussenbaum says. In practice, physicians are more likely to report events that caused permanent harm or the death or near-death of a patient, but overall, they report only 1% of adverse events.3 Nurses, on the other hand, are more likely to report events that cause no harm or harm that’s only temporary, and report about 45% of events. Residents, in the same study, reported only a little more than half of adverse events to attending physicians.
“Nurses and pharmacists have a lot more training in how to fill out the voluntary reporting,” Dr. Maynard says. “Nurses and pharmacists report the overwhelming majority of adverse events and errors. I think physicians need to take some responsibility for ensuring that errors are reported into the voluntary reporting system. When I get called about something that happens on one of my patients, or detect it, I don’t necessarily enter it myself, but I will specify with whoever is calling me that it should be reported.”
Society Initiatives
SHM has taken an aggressive stance that is more focused on improving quality and safety than that of most professional organizations, according to Dr. Maynard. “We feel that the best way to build hospitalists in their careers is to increase their value by helping them take care of patients and being part of the solution to these problems,” he says. “We have quality and safety modules for learning about error and medical harm and ways to prevent it.”
SHM’s website (www.hospitalmedicine.org)
features resources and tools for DVT prevention, glycemic control, and anticoagulation—all well-known areas for medical errors. “We’re about ready to embark on putting together an adverse-drug-events module, and Project BOOST [Better Outcomes for Older Adults through Safe Transitions] provides materials to help optimize transitions of care,” Dr. Maynard says. “We want to build this into the daily fabric of the hospitalist’s career.”
At the CDC’s medication safety program, Dr. Budnitz says, his focus is less on error reporting and more on tracking backward to see what could have been the cause of harm to a patient—error or not.
“Sometimes, medication-induced harms are caused by things that we classically think of as errors, and sometimes they are not,” he says. “Error reporting is important, but sometimes things don’t fit neatly into the ‘healthcare error’ box—like when a patient misses a follow-up appointment to get their blood thinner level checked, and [the] patient has an unintended bleeding event.”
—Rosemary Gibson, MSc, author, former senior program officer, Robert Wood Johnson Foundation
Gibson believes that what’s needed is a public system for the reporting of adverse events in hospitals—like a Consumer Product Safety Commission for healthcare.
“If your child gets injured by a defective crib, there’s a system for you to report that,” she says. “We need that for hospitals. There should be an open capability for reporting events online, in person, any way you want, whether you’re on the hospital staff, a patient, or a family member. Such a system should acknowledge the report, provide for an immediate action step in urgent situations, and offer assistance and support to the person doing the reporting.”
But overall, the response in the healthcare industry to the OIG’s report has been “we need to do more research,” Gibson notes. “We don’t need more research; we need to apply what is known. There are institutions that have made pockets of progress, and patients are alive today and home with their families because we’ve made care safer, but safety still hasn’t become a system property in healthcare.”
Gina Shaw is a freelance writer in New York City.
References
- Levinson DR. Adverse events in hospitals: national incidence among Medicare beneficiaries. U.S. Department of Health and Human Services’ Office of Inspector General website. Available at: http://oig.hhs.gov/oei/reports/oei-06-09-00090.pdf. Accessed May 31, 2012.
- Leape LL. Testimony, United States Congress, House Committee on Veterans’ Affairs; Oct. 12, 1997.
- Rowin EJ. Does error and adverse event reporting by physicians and nurses differ? Jt Comm J Qual Patient Saf. 2008;34(9):537-545.
Nearly 13 years after the release of the Institute of Medicine’s landmark report To Err Is Human, which called national attention to the rate of preventable errors in U.S. hospitals and galvanized the patient-safety movement, 6 out of every 7 hospital-based errors, accidents, and other adverse events still go unreported.
That’s the troubling conclusion of a report released in January by the Department of Health and Human Services’ Office of the Inspector General (OIG). According to the OIG, the report the National Incident Study on Adverse Events in Hospitals Among Medicare Beneficiaries, is the first truly national adverse-event study in the nation documenting hospital-based adverse events among Medicare beneficiaries.1
Previous studies assessing errors and adverse events had tracked the number of events reported, but they had no denominator; in other words, one hospital might have 1,000 reports of adverse events and errors in a given month, while another might have 50, but there was no way to know how many adverse events had been missed and how much harm had actually occurred.
The OIG study dug deeper, conducting in-depth, yearlong reviews led by independent physicians to first identify the rates of harm, then returning to the hospitals in the study where they knew harm had occurred to find out if it had been reported or not.
Overall, the inspector general estimated that more than 130,000 Medicare beneficiaries experienced one or more adverse events in hospitals in a single month. Perhaps one of the most surprising findings was that even some of the most serious, egregious adverse events were not reported. Of the 12 deaths in the sample that were iatrogenic, only two were reported as errors. More serious errors were no more likely to be captured as errors than mild reactions, such as breaking out in hives as the result of a medication.
Why aren’t more hospital-based errors and adverse events being reported, and how can this situation be changed? The OIG’s report provides some insight into the underlying problem: The dominant reason that events weren’t reported was that they weren’t perceived as adverse events. (Not all adverse events are medical errors: A medical error is a preventable adverse event.) A urinary catheter infection, while not necessarily a medical error, is always an adverse event—and yet, only one of the 17 urinary catheter infections identified by doctors in medical review was reported.
Lack of recognition—seeing adverse events and errors as just part of routine care—is only one part of the problem, says Rosemary Gibson, MSc, a former senior program officer at the Robert Wood Johnson Foundation and author of “The Treatment Trap” and “Wall of Silence.” Other issues include:
- Fear of retribution. “You have to feel safe to report someone else’s error, or your own,” Gibson says. “Most hospitals have yet to create a safety culture.”
- A sense of disbelief. “I remember talking to a hospital CEO once right after the IOM report,” Gibson recalls. “He said that after a significant error, he would get up in the morning and look in the mirror and think, ‘Did this really happen here?’ There’s almost a sense that if you don’t see it, it didn’t exist.”
- Competing pressures. “The requirements of healthcare reform have taken up so much time and energy that I fear safety has moved to the back burner,” Gibson says. “Someone in a quality and safety leadership role at one hospital said to me, ‘Safety was just a fad. We’re not doing that anymore.’”
- Productivity demands. “Healthcare’s mantra today has become volume, volume, volume. If you already have an environment that’s not as safe as we would like, and you ramp up the volume so people have to do more in the name of productivity, what’s going to happen?” asks Gibson.
The Hospitalist Perspective
Greg Maynard, MD, SFHM, director of the University of California San Diego Center for Innovation and Improvement Science (CIIS) and senior vice president of SHM’s Center for Healthcare Improvement and Innovation, says hospitalists face multiple barriers to regular reporting. Some errors, he says, are so pervasive that they are “considered routine” and “happen all the time.”
“Hypoglycemic events, for example, don’t always get entered into adverse-event reporting systems because they’re so common,” he explains, “even though you should be entering and examining them all.”
—Dan Budnitz, MD, MPH, director, Medication Safety Program, Centers for Disease Control and Prevention
Improper coding is another hospitalist-centric issue. Since Oct. 1, 2008, the Centers for Medicare & Medicaid Services (CMS) has reduced payment for acute-care inpatient cases when designated hospital-acquired conditions are not present on admission and the condition would have increased the reimbursement.
“CMS went to a lot of trouble to implement codes that could help them identify, in the coding process, patients who had an iatrogenic event,” Dr. Maynard says. “But those codes simply aren’t being used properly, and administrative coding is always a problem with regard to accuracy.”
Hospitalists can be particularly vulnerable to errors due to the nature of their work—multiple tasks, multiple team members, multiple interruptions. “[Hospitalists] are prone to the type of errors that are related to the hectic nature of being a doctor and an air traffic controller at the same time,” Dr. Maynard says. “You’re in the middle of admitting someone and get two urgent calls on two other patients, so you have to switch out of what you’re doing on that patient, issue orders on the other two, and immediately switch back. It’s hard to keep your focus when dealing with many complex patients, many of whom you’re seeing for the first time, and try to remember all the things you should not omit.”
One area that has, in recent years, become more important to hospitalists in terms of error prevention—and reporting—is discharge planning.
“When you’re sending your patient out of the hospital, they may have started on new medications or changed medications while an inpatient,” says Dan Budnitz, MD, MPH, director of the Medication Safety Program at the Centers for Disease Control and Prevention. “A recent report in the New England Journal of Medicine looked at drug-only causes of emergency hospitalizations, and the most common drugs involved are blood thinners, insulin, and some diabetes pills. The implication for the hospitalist is clear: If you start a patient on these medications in the hospital, or change them, you need to pay particular attention, because they’re two of the top drugs that may put your patient back in the hospital.”
National Models
One institution that is far ahead of the curve in error reporting is the University of Michigan Health System. In 2001, just two years after To Err is Human was published, the health system adopted a process of full disclosure of medical errors. The process involves multiple components, including an online incident reporting system, a multidisciplinary claims review committee, open and honest communication with patients and families, and quality-improvement (QI) initiatives guided by reported errors.
“They’ll get 7,000 or 8,000 reports a year from staff,” Gibson says. “That’s what you want. Most of them are minor or near-misses, and you want to know about those so you can learn from them and prevent the larger errors. But you have to create a space where it’s safe for people to report.”
She says that hospitals must adhere to the principle of “just culture” in error reporting, a philosophy that recognizes that even competent professionals make mistakes. Quality-improvement leader and Harvard School of Public Health professor Lucian Leape, MD, has said that the biggest impediment to error reporting and prevention in medicine is “that we punish people for making mistakes.”2
“If I made a mistake and anybody else in my position could have made that same mistake, the chances are the system contributed to my making that mistake, and just culture would suggest I shouldn’t bear all the blame for it,” Gibson says. “That’s different from a case in which I did something that was truly negligent. Hospitals often do not make this distinction, and [they] lack a just culture.”
—Greg Maynard, MD, SFHM, director, University of California San Diego Center for Innovation and Improvement Science, senior vice president, SHM’s Center for Healthcare Improvement and Innovation
Traditional reporting systems in healthcare, however, often do not support a just-culture model or a true patient-safety-focused approach, says Brian Nussenbaum, MD, associate professor of otolaryngology and head and neck surgery at Washington University School of Medicine in Seattle. “Paper or online reports are sent to hospital risk management departments, whose concerns are primarily to limit the potential legal risk,” he says. “There is little emphasis on systems improvement or prevention. Dissemination of incidents to others in the organization is unusual, and the impact on clinical care is often not felt by providers.”
Who should be reporting errors? The responsibility ideally lies with everyone involved in patient care, Dr. Nussenbaum says. In practice, physicians are more likely to report events that caused permanent harm or the death or near-death of a patient, but overall, they report only 1% of adverse events.3 Nurses, on the other hand, are more likely to report events that cause no harm or harm that’s only temporary, and report about 45% of events. Residents, in the same study, reported only a little more than half of adverse events to attending physicians.
“Nurses and pharmacists have a lot more training in how to fill out the voluntary reporting,” Dr. Maynard says. “Nurses and pharmacists report the overwhelming majority of adverse events and errors. I think physicians need to take some responsibility for ensuring that errors are reported into the voluntary reporting system. When I get called about something that happens on one of my patients, or detect it, I don’t necessarily enter it myself, but I will specify with whoever is calling me that it should be reported.”
Society Initiatives
SHM has taken an aggressive stance that is more focused on improving quality and safety than that of most professional organizations, according to Dr. Maynard. “We feel that the best way to build hospitalists in their careers is to increase their value by helping them take care of patients and being part of the solution to these problems,” he says. “We have quality and safety modules for learning about error and medical harm and ways to prevent it.”
SHM’s website (www.hospitalmedicine.org)
features resources and tools for DVT prevention, glycemic control, and anticoagulation—all well-known areas for medical errors. “We’re about ready to embark on putting together an adverse-drug-events module, and Project BOOST [Better Outcomes for Older Adults through Safe Transitions] provides materials to help optimize transitions of care,” Dr. Maynard says. “We want to build this into the daily fabric of the hospitalist’s career.”
At the CDC’s medication safety program, Dr. Budnitz says, his focus is less on error reporting and more on tracking backward to see what could have been the cause of harm to a patient—error or not.
“Sometimes, medication-induced harms are caused by things that we classically think of as errors, and sometimes they are not,” he says. “Error reporting is important, but sometimes things don’t fit neatly into the ‘healthcare error’ box—like when a patient misses a follow-up appointment to get their blood thinner level checked, and [the] patient has an unintended bleeding event.”
—Rosemary Gibson, MSc, author, former senior program officer, Robert Wood Johnson Foundation
Gibson believes that what’s needed is a public system for the reporting of adverse events in hospitals—like a Consumer Product Safety Commission for healthcare.
“If your child gets injured by a defective crib, there’s a system for you to report that,” she says. “We need that for hospitals. There should be an open capability for reporting events online, in person, any way you want, whether you’re on the hospital staff, a patient, or a family member. Such a system should acknowledge the report, provide for an immediate action step in urgent situations, and offer assistance and support to the person doing the reporting.”
But overall, the response in the healthcare industry to the OIG’s report has been “we need to do more research,” Gibson notes. “We don’t need more research; we need to apply what is known. There are institutions that have made pockets of progress, and patients are alive today and home with their families because we’ve made care safer, but safety still hasn’t become a system property in healthcare.”
Gina Shaw is a freelance writer in New York City.
References
- Levinson DR. Adverse events in hospitals: national incidence among Medicare beneficiaries. U.S. Department of Health and Human Services’ Office of Inspector General website. Available at: http://oig.hhs.gov/oei/reports/oei-06-09-00090.pdf. Accessed May 31, 2012.
- Leape LL. Testimony, United States Congress, House Committee on Veterans’ Affairs; Oct. 12, 1997.
- Rowin EJ. Does error and adverse event reporting by physicians and nurses differ? Jt Comm J Qual Patient Saf. 2008;34(9):537-545.
What Is the Optimal Therapy for Acute DVT?
The Case
A 55-year-old female undergoes cholecystectomy. On post-operative Day 2, she develops right-lower-extremity swelling and pain; venous ultrasound detects a proximal deep venous thrombosis (DVT). The patient denies smoking or use of hormonal medications. She has no history of venous thromboembolism (VTE), although her brother had a DVT at age 60. The hospitalist team is consulted for management of acute DVT.
Overview
VTE, including lower- and upper-extremity DVT and pulmonary embolism (PE), is one of the most common and preventable hospital diseases. DVT with PE is associated with a 10% mortality rate, and DVT with post-thrombotic syndrome can be associated with significant morbidity, including pain, edema, skin/pigment change, venous dilation, and ulcer development.1,2 Recognition of clinical symptoms and risk factors for DVT (see Table 1) in conjunction with validated clinical scoring predictors (such as the Wells Prediction Rule) and a high-sensitivity D-dimer assay can help diagnose the condition and determine the need for ultrasound.3-7
Pharmacologic Treatment
Anticoagulation should be initiated in all patients with VTE, regardless of patient symptoms. Anticoagulant options include:
- Intravenous (IV) or subcutaneous (SC) unfractionated heparin (UFH);
- SC low-molecular-weight heparins (LMWH), such as enoxaparin and dalteparin; and
- Fondaparinux (as effective as LMWH for acute treatment of VTE).8
These agents can be used while transitioning to oral vitamin K antagonists (VKA), such as warfarin.3
The 2012 American College of Chest Physicians (ACCP) guidelines on antithrombotic therapy for VTE recommend initial therapy with LMWH or fondaparinux (rather than IV or SC UFH). The guidelines suggest that LMWH once-daily dosing is favored over twice-daily dosing, based mainly on patient convenience, although this is a weak recommendation (2C) based on the overall quality of the data. The recommendation applies only if the daily dosing of the LMWH, including tinzaparin, dalteparin, and nadroparin, is equivalent to the twice-daily dosing (i.e., dalteparin may be dosed at 100 units/kg BID vs. 200 units/kg daily). Of importance, enoxaparin has not been studied at a once-daily dose (2 mg/kg), which is equivalent to the twice-daily dosing regimen (1 mg/kg twice daily). Additionally, one study suggests that once-daily dosing of enoxaparin 1.5mg/kg might be inferior to 1 mg/kg twice-daily dosing; therefore, caution must be exercised in applying this recommendation to the LMWH enoxaparin at this time.3,27,28 (updated Aug. 28, 2012)
Warfarin should be started simultaneously at a usual daily dose of 5 mg for the first two days, with subsequent doses adjusted to achieve a goal international normalized ratio (INR) of 2.0 to 3.0. Parenteral agents should be given for a minimum of five days and until the INR has been >2.0 for at least 24 hours.3
The new factor-Xa inhibitor rivaroxaban and the direct thrombin inhibitor dabigatran are promising oral alternatives to warfarin.9-11 However, neither drug is currently FDA-approved for the treatment of VTE, nor are they recommended by current guidelines (given limited data for DVT treatment and concerns of bleeding risk).3,12,13 See Table 2 (above) for comparisons of common anticoagulants.3,14-17
Duration of anticoagulation. Anticoagulant treatment of acute DVT should continue for at least three months, as shorter durations are associated with higher recurrence rates. Longer treatment may be indicated depending on the patient’s risk of recurrence.3
The ACCP guidelines estimate risk of recurrence using primary, secondary, and additional factors (see Table 3, p. 19) and recommend the following durations:
- First episode provoked: three months (proximal or distal, provoked by surgery or a nonsurgical transient risk factor);
- First episode unprovoked distal: three months (see “Considerations for isolated distal DVT,” below);
- First episode unprovoked proximal: Indefinite if low to moderate bleeding risk, three months if high bleeding risk;
- Recurrent unprovoked: Indefinite if low to moderate bleeding risk, three months if high bleeding risk; and
- With active cancer: Indefinite with LMWH due to higher risk of recurrence.3,18
These treatment duration guidelines might need to be individualized based on other factors including patient preference, ability to obtain accurate INR monitoring (for those on warfarin), treatment cost, and comorbidities.3
Considerations for isolated distal DVT. Patients with an initial episode of distal DVT, without significant symptoms or risk factors for extension (e.g. positive D-dimer, extensive clot near proximal veins, absence of a reversible provoking factor, active cancer, inpatient status, or previous VTE) might not need anticoagulation.
The DVT can be followed with serial ultrasounds for the first two weeks; anticoagulation is recommended only if the thrombus extends during that time period. The development of significant symptoms or risk factors of extension might indicate the need for anticoagulation.3
Considerations for upper-extremity DVT (UEDVT). Anticoagulation for an UEDVT is generally consistent with the above guidelines for lower-extremity DVT, with a few caveats. If an UEDVT is associated with a central venous catheter (CVC), the CVC should be removed if possible; there are no recommendations to determine whether CVC removal should be preceded by a period of anticoagulation.
A catheter-associated UEDVT requires a minimum of three months of anticoagulation; if the CVC remains in place beyond three months, anticoagulation should be continued until the catheter is removed. Unprovoked UEDVT has a lower risk of recurrence than lower-extremity DVT and three months of anticoagulation, rather than indefinite therapy, is recommended.3
Mechanical Treatment
Non-pharmacologic therapies, such as knee-high graduated compression stockings with pressure of 30 mmHg to 40 mmHg at the ankle, can help reduce the morbidity of post-thrombotic syndrome (PTS) when combined with anticoagulation. Symptomatic patients who use compression stockings as soon as feasible and for a minimum of two years can reduce their incidence of PTS by 50%.3,19,20
Thigh-length stockings are not more effective than knee-high, and while multilayer compression bandages might relieve symptoms during the first-week post-DVT, they do not reduce the one-year incidence of PTS.21,22 Early mobilization is not associated with an increased risk of PE, extension of DVT, or death; patients should ambulate as soon as physically able.23,24
Pharmacomechanical Thrombolysis
For acute DVT, ACCP guidelines recommend anticoagulation alone over pharmacomechanical thrombolysis (either systemic or catheter-directed thrombolysis and mechanical thrombus fragmentation). The rare patient with impending venous gangrene despite anticoagulation is the only clinical scenario in which thrombolysis is clearly indicated. Patients who undergo pharmacomechanical thrombolysis still need a standard course of anticoagulation.3
Role for Inferior Vena Cava Filters
The optimal role of inferior vena cava (IVC) filters remains uncertain. Only one randomized trial found that IVC filters, in conjunction with systemic anticoagulation versus systemic anticoagulation alone, were associated with short-term reductions in the incidence of PE but long-term increases in recurrent DVT, with no differences in mortality or major bleeding. However, no trials have compared anticoagulation plus IVC filter placement with IVC filter placement alone.25,26
ACCP guidelines recommend IVC filter placement only in patients with acute, proximal DVT of the lower extremity, and a contraindication to anticoagulant therapy. If the contraindication resolves, a conventional course of anticoagulation can commence. Combining an IVC filter with an anticoagulant is not recommended. The risks and benefits of retrievable filters require further investigation.3
Back to the Case
Our patient has a provoked DVT secondary to a reversible risk factor (surgery) without additional clinical risk factors. Her family history of DVT is not significant (her brother was >age 50 when it occurred). This patient should be treated with LMWH or fondaparinux with initiation of warfarin with goal INR of 2.0 to 3.0 for at least three months. She does not need an IVC filter, and she should use compression stockings to reduce the risk of PTS.
Bottom Line
In hospitalized patients, treatment of DVT should include immediate anticoagulation with LMWH, fondaparinux, or IV heparin (in patients with renal failure) with transition to warfarin and a goal INR of 2.0 to 3.0. New oral anticoagulants could prove beneficial in acute treatment of DVT but require further testing. Duration of treatment is patient-specific, but most should be anticoagulated for at least three months; some warrant indefinite therapy based on risk factors.
Dr. Sebasky is an assistant professor and Dr. DeKorte is assistant professor of medicine in the division of hospital medicine at the University of California at San Diego.
References
- Agency for Healthcare Research and Quality. Talking Points to Attract Administration Support for Venous Thromboembolism Prevention Programs. U.S. Department of Health & Human Services website. Available at: http://www.ahrq.gov/qual/vtguide/vtguideapa.htm. Accessed Feb. 4, 2012.
- Kahn SR, Shbaklo H, Lamping DL, et al. Determinants of health-related quality of life during the 2 years following deep vein thrombosis. J Thromb Haemost. 2008;6:1105-1112.
- Kearon C, Akl E, Comerota AJ, et al. Antithrombotic Therapy for VTE Disease. Antithrombotic Therapy and Prevention of Thrombosis, 9th ed.: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(2 Suppl):e419S-e494S.
- Hirsh J, Hull RD, Raskob GE. Clinical features and diagnosis of venous thrombosis. J Am Coll Cardiol. 1986;8(6 Suppl B):114B-127B.
- Qaseem A, Snow V, Barry P, et al. Current diagnosis of venous thromboembolism in primary care: a clinical practice guideline from the American Academy of Family Physicians and the American College of Physicians. Ann Int Med. 2007;146:454-458.
- Tapson VF, Carroll BA, Davidson BL, et al. The diagnostic approach to acute venous thromboembolism. Clinical practice guideline. American Thoracic Society. Am J Respir Crit Care Med. 1999;160:1043-1066.
- Wells PS, Owen C, Doucette S, Fergusson D, Tran H. Does this patient have deep vein thrombosis? JAMA. 2006;295:199-207.
- Büller HR, Davidson BL, Decousus H, et al. Fondaparinux or enoxaparin for the initial treatment of symptomatic deep venous thrombosis: a randomized trial. Ann Intern Med. 2004;140:867-873.
- EINSTEIN Investigators, Bauersachs R, Berkowitz SD, et al. Oral rivaroxaban for symptomatic venous thromboembolism. N Engl J Med. 2010;363:2499-25
- Garcia, D, Libby E, Crowther M. The new oral anticoagulants. Blood. 2010;115:15-20.
- Douketis JD. Pharmacologic properties of the new oral anticoagulants: a clinician-oriented review with a focus on perioperative management. Curr Pharm Des. 2010;16:3436-3441.
- U.S. Food and Drug Administration. Pradaxa (dabigatran etexilate mesylate): Drug Safety Communication—Safety Review of Post-Market Reports of Serious Bleeding Events. U.S. Food and Drug Administration website. Available at: http://www.fda.gov/Safety/MedWatch/SafetyInformation/
SafetyAlertsforHumanMedicalProducts/ucm282820.htm. Accessed March 12, 2012.
- Levi M, Erenberg E, Kamphuisen PW. Bleeding risk and reversal strategies for old and new anticoagulants and antiplatelet agents. J Thromb Haemost. 2011;9:1705.
- Erkens PM, Prins MH. Fixed dose subcutaneous low molecular weight heparins versus adjusted dose unfractionated heparin for venous thromboembolism. Cochrane Database Syst Rev. 2010;8(9);CD001100.Vardi M, Zittan E, Bitterman H. Subcutaneous unfractionated heparin for the initial treatment of venous thromboembolism. Cochrane Database Syst Rev. 2009;(4):CD006771.
- Hirsh J, Levine MN. Low molecular weight heparin. Blood. 1992;79:1-17.
- Schulman S, Kearon C, Kakkar AK, et al. Dabigatran versus warfarin in the treatment of acute venous thromboembolism. N Engl J Med. 2009;361;2342-2352.
- Bauer KA. Long-term management of venous thromboembolism. JAMA. 2011;305:1336-1345.
- Prandoni P, Lensing AW, Prins MH, et al. Below-knee elastic compression stockings to prevent the postthrombotic syndrome: a randomized, controlled trial. Ann Intern Med. 2004;141:249-256.
- Brandjes DP, Büller HR, Heijboer H, et al. Randomised trial of effect of compression stockings in patients with symptomatic proximal-vein thrombosis. Lancet. 1997;349:759-762.
- Prandoni P, Noventa F, Quintavalla R, et al. Thigh-length versus below-knee compression elastic stockings for prevention of the post-thrombotic syndrome in patients with proximal-venous thrombosis: a randomized trial. Blood. 2012;119:1561-1565.
- Roumen-Klappe EM, den Heijer M, van Rossum J, et al. Multilayer compression bandaging in the acute phase of deep-vein thrombosis has no effect on the development of the post-thrombotic syndrome. J Thromb Thrombolysis. 2009;27:400-405.
- Aissaoui N, Martins E, Mouly S, Weber S, Meune C. A meta-analysis of bed rest versus early ambulation in the management of pulmonary embolism, deep venous thrombosis, or both. Int J Cardiol. 2009;137:37-41.
- Anderson CM, Overend TJ, Godwin J, Sealy C, Sunderji A. Ambulation after deep vein thrombosis: a systematic review. Physiother Can. 2009;61:133-140.
- Hann CL, Streiff MB. The role of vena caval filters in the management of venous thromboembolism. Blood Rev. 2005;19:179-202.
- Decousus H, Leizorovicz A, Page Y, et al. A clinical trial of vena caval filters in the prevention of pulmonary embolism in patients with proximal deep-vein thrombosis. N Engl J Med. 1998;338:409-415.
The Case
A 55-year-old female undergoes cholecystectomy. On post-operative Day 2, she develops right-lower-extremity swelling and pain; venous ultrasound detects a proximal deep venous thrombosis (DVT). The patient denies smoking or use of hormonal medications. She has no history of venous thromboembolism (VTE), although her brother had a DVT at age 60. The hospitalist team is consulted for management of acute DVT.
Overview
VTE, including lower- and upper-extremity DVT and pulmonary embolism (PE), is one of the most common and preventable hospital diseases. DVT with PE is associated with a 10% mortality rate, and DVT with post-thrombotic syndrome can be associated with significant morbidity, including pain, edema, skin/pigment change, venous dilation, and ulcer development.1,2 Recognition of clinical symptoms and risk factors for DVT (see Table 1) in conjunction with validated clinical scoring predictors (such as the Wells Prediction Rule) and a high-sensitivity D-dimer assay can help diagnose the condition and determine the need for ultrasound.3-7
Pharmacologic Treatment
Anticoagulation should be initiated in all patients with VTE, regardless of patient symptoms. Anticoagulant options include:
- Intravenous (IV) or subcutaneous (SC) unfractionated heparin (UFH);
- SC low-molecular-weight heparins (LMWH), such as enoxaparin and dalteparin; and
- Fondaparinux (as effective as LMWH for acute treatment of VTE).8
These agents can be used while transitioning to oral vitamin K antagonists (VKA), such as warfarin.3
The 2012 American College of Chest Physicians (ACCP) guidelines on antithrombotic therapy for VTE recommend initial therapy with LMWH or fondaparinux (rather than IV or SC UFH). The guidelines suggest that LMWH once-daily dosing is favored over twice-daily dosing, based mainly on patient convenience, although this is a weak recommendation (2C) based on the overall quality of the data. The recommendation applies only if the daily dosing of the LMWH, including tinzaparin, dalteparin, and nadroparin, is equivalent to the twice-daily dosing (i.e., dalteparin may be dosed at 100 units/kg BID vs. 200 units/kg daily). Of importance, enoxaparin has not been studied at a once-daily dose (2 mg/kg), which is equivalent to the twice-daily dosing regimen (1 mg/kg twice daily). Additionally, one study suggests that once-daily dosing of enoxaparin 1.5mg/kg might be inferior to 1 mg/kg twice-daily dosing; therefore, caution must be exercised in applying this recommendation to the LMWH enoxaparin at this time.3,27,28 (updated Aug. 28, 2012)
Warfarin should be started simultaneously at a usual daily dose of 5 mg for the first two days, with subsequent doses adjusted to achieve a goal international normalized ratio (INR) of 2.0 to 3.0. Parenteral agents should be given for a minimum of five days and until the INR has been >2.0 for at least 24 hours.3
The new factor-Xa inhibitor rivaroxaban and the direct thrombin inhibitor dabigatran are promising oral alternatives to warfarin.9-11 However, neither drug is currently FDA-approved for the treatment of VTE, nor are they recommended by current guidelines (given limited data for DVT treatment and concerns of bleeding risk).3,12,13 See Table 2 (above) for comparisons of common anticoagulants.3,14-17
Duration of anticoagulation. Anticoagulant treatment of acute DVT should continue for at least three months, as shorter durations are associated with higher recurrence rates. Longer treatment may be indicated depending on the patient’s risk of recurrence.3
The ACCP guidelines estimate risk of recurrence using primary, secondary, and additional factors (see Table 3, p. 19) and recommend the following durations:
- First episode provoked: three months (proximal or distal, provoked by surgery or a nonsurgical transient risk factor);
- First episode unprovoked distal: three months (see “Considerations for isolated distal DVT,” below);
- First episode unprovoked proximal: Indefinite if low to moderate bleeding risk, three months if high bleeding risk;
- Recurrent unprovoked: Indefinite if low to moderate bleeding risk, three months if high bleeding risk; and
- With active cancer: Indefinite with LMWH due to higher risk of recurrence.3,18
These treatment duration guidelines might need to be individualized based on other factors including patient preference, ability to obtain accurate INR monitoring (for those on warfarin), treatment cost, and comorbidities.3
Considerations for isolated distal DVT. Patients with an initial episode of distal DVT, without significant symptoms or risk factors for extension (e.g. positive D-dimer, extensive clot near proximal veins, absence of a reversible provoking factor, active cancer, inpatient status, or previous VTE) might not need anticoagulation.
The DVT can be followed with serial ultrasounds for the first two weeks; anticoagulation is recommended only if the thrombus extends during that time period. The development of significant symptoms or risk factors of extension might indicate the need for anticoagulation.3
Considerations for upper-extremity DVT (UEDVT). Anticoagulation for an UEDVT is generally consistent with the above guidelines for lower-extremity DVT, with a few caveats. If an UEDVT is associated with a central venous catheter (CVC), the CVC should be removed if possible; there are no recommendations to determine whether CVC removal should be preceded by a period of anticoagulation.
A catheter-associated UEDVT requires a minimum of three months of anticoagulation; if the CVC remains in place beyond three months, anticoagulation should be continued until the catheter is removed. Unprovoked UEDVT has a lower risk of recurrence than lower-extremity DVT and three months of anticoagulation, rather than indefinite therapy, is recommended.3
Mechanical Treatment
Non-pharmacologic therapies, such as knee-high graduated compression stockings with pressure of 30 mmHg to 40 mmHg at the ankle, can help reduce the morbidity of post-thrombotic syndrome (PTS) when combined with anticoagulation. Symptomatic patients who use compression stockings as soon as feasible and for a minimum of two years can reduce their incidence of PTS by 50%.3,19,20
Thigh-length stockings are not more effective than knee-high, and while multilayer compression bandages might relieve symptoms during the first-week post-DVT, they do not reduce the one-year incidence of PTS.21,22 Early mobilization is not associated with an increased risk of PE, extension of DVT, or death; patients should ambulate as soon as physically able.23,24
Pharmacomechanical Thrombolysis
For acute DVT, ACCP guidelines recommend anticoagulation alone over pharmacomechanical thrombolysis (either systemic or catheter-directed thrombolysis and mechanical thrombus fragmentation). The rare patient with impending venous gangrene despite anticoagulation is the only clinical scenario in which thrombolysis is clearly indicated. Patients who undergo pharmacomechanical thrombolysis still need a standard course of anticoagulation.3
Role for Inferior Vena Cava Filters
The optimal role of inferior vena cava (IVC) filters remains uncertain. Only one randomized trial found that IVC filters, in conjunction with systemic anticoagulation versus systemic anticoagulation alone, were associated with short-term reductions in the incidence of PE but long-term increases in recurrent DVT, with no differences in mortality or major bleeding. However, no trials have compared anticoagulation plus IVC filter placement with IVC filter placement alone.25,26
ACCP guidelines recommend IVC filter placement only in patients with acute, proximal DVT of the lower extremity, and a contraindication to anticoagulant therapy. If the contraindication resolves, a conventional course of anticoagulation can commence. Combining an IVC filter with an anticoagulant is not recommended. The risks and benefits of retrievable filters require further investigation.3
Back to the Case
Our patient has a provoked DVT secondary to a reversible risk factor (surgery) without additional clinical risk factors. Her family history of DVT is not significant (her brother was >age 50 when it occurred). This patient should be treated with LMWH or fondaparinux with initiation of warfarin with goal INR of 2.0 to 3.0 for at least three months. She does not need an IVC filter, and she should use compression stockings to reduce the risk of PTS.
Bottom Line
In hospitalized patients, treatment of DVT should include immediate anticoagulation with LMWH, fondaparinux, or IV heparin (in patients with renal failure) with transition to warfarin and a goal INR of 2.0 to 3.0. New oral anticoagulants could prove beneficial in acute treatment of DVT but require further testing. Duration of treatment is patient-specific, but most should be anticoagulated for at least three months; some warrant indefinite therapy based on risk factors.
Dr. Sebasky is an assistant professor and Dr. DeKorte is assistant professor of medicine in the division of hospital medicine at the University of California at San Diego.
References
- Agency for Healthcare Research and Quality. Talking Points to Attract Administration Support for Venous Thromboembolism Prevention Programs. U.S. Department of Health & Human Services website. Available at: http://www.ahrq.gov/qual/vtguide/vtguideapa.htm. Accessed Feb. 4, 2012.
- Kahn SR, Shbaklo H, Lamping DL, et al. Determinants of health-related quality of life during the 2 years following deep vein thrombosis. J Thromb Haemost. 2008;6:1105-1112.
- Kearon C, Akl E, Comerota AJ, et al. Antithrombotic Therapy for VTE Disease. Antithrombotic Therapy and Prevention of Thrombosis, 9th ed.: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(2 Suppl):e419S-e494S.
- Hirsh J, Hull RD, Raskob GE. Clinical features and diagnosis of venous thrombosis. J Am Coll Cardiol. 1986;8(6 Suppl B):114B-127B.
- Qaseem A, Snow V, Barry P, et al. Current diagnosis of venous thromboembolism in primary care: a clinical practice guideline from the American Academy of Family Physicians and the American College of Physicians. Ann Int Med. 2007;146:454-458.
- Tapson VF, Carroll BA, Davidson BL, et al. The diagnostic approach to acute venous thromboembolism. Clinical practice guideline. American Thoracic Society. Am J Respir Crit Care Med. 1999;160:1043-1066.
- Wells PS, Owen C, Doucette S, Fergusson D, Tran H. Does this patient have deep vein thrombosis? JAMA. 2006;295:199-207.
- Büller HR, Davidson BL, Decousus H, et al. Fondaparinux or enoxaparin for the initial treatment of symptomatic deep venous thrombosis: a randomized trial. Ann Intern Med. 2004;140:867-873.
- EINSTEIN Investigators, Bauersachs R, Berkowitz SD, et al. Oral rivaroxaban for symptomatic venous thromboembolism. N Engl J Med. 2010;363:2499-25
- Garcia, D, Libby E, Crowther M. The new oral anticoagulants. Blood. 2010;115:15-20.
- Douketis JD. Pharmacologic properties of the new oral anticoagulants: a clinician-oriented review with a focus on perioperative management. Curr Pharm Des. 2010;16:3436-3441.
- U.S. Food and Drug Administration. Pradaxa (dabigatran etexilate mesylate): Drug Safety Communication—Safety Review of Post-Market Reports of Serious Bleeding Events. U.S. Food and Drug Administration website. Available at: http://www.fda.gov/Safety/MedWatch/SafetyInformation/
SafetyAlertsforHumanMedicalProducts/ucm282820.htm. Accessed March 12, 2012.
- Levi M, Erenberg E, Kamphuisen PW. Bleeding risk and reversal strategies for old and new anticoagulants and antiplatelet agents. J Thromb Haemost. 2011;9:1705.
- Erkens PM, Prins MH. Fixed dose subcutaneous low molecular weight heparins versus adjusted dose unfractionated heparin for venous thromboembolism. Cochrane Database Syst Rev. 2010;8(9);CD001100.Vardi M, Zittan E, Bitterman H. Subcutaneous unfractionated heparin for the initial treatment of venous thromboembolism. Cochrane Database Syst Rev. 2009;(4):CD006771.
- Hirsh J, Levine MN. Low molecular weight heparin. Blood. 1992;79:1-17.
- Schulman S, Kearon C, Kakkar AK, et al. Dabigatran versus warfarin in the treatment of acute venous thromboembolism. N Engl J Med. 2009;361;2342-2352.
- Bauer KA. Long-term management of venous thromboembolism. JAMA. 2011;305:1336-1345.
- Prandoni P, Lensing AW, Prins MH, et al. Below-knee elastic compression stockings to prevent the postthrombotic syndrome: a randomized, controlled trial. Ann Intern Med. 2004;141:249-256.
- Brandjes DP, Büller HR, Heijboer H, et al. Randomised trial of effect of compression stockings in patients with symptomatic proximal-vein thrombosis. Lancet. 1997;349:759-762.
- Prandoni P, Noventa F, Quintavalla R, et al. Thigh-length versus below-knee compression elastic stockings for prevention of the post-thrombotic syndrome in patients with proximal-venous thrombosis: a randomized trial. Blood. 2012;119:1561-1565.
- Roumen-Klappe EM, den Heijer M, van Rossum J, et al. Multilayer compression bandaging in the acute phase of deep-vein thrombosis has no effect on the development of the post-thrombotic syndrome. J Thromb Thrombolysis. 2009;27:400-405.
- Aissaoui N, Martins E, Mouly S, Weber S, Meune C. A meta-analysis of bed rest versus early ambulation in the management of pulmonary embolism, deep venous thrombosis, or both. Int J Cardiol. 2009;137:37-41.
- Anderson CM, Overend TJ, Godwin J, Sealy C, Sunderji A. Ambulation after deep vein thrombosis: a systematic review. Physiother Can. 2009;61:133-140.
- Hann CL, Streiff MB. The role of vena caval filters in the management of venous thromboembolism. Blood Rev. 2005;19:179-202.
- Decousus H, Leizorovicz A, Page Y, et al. A clinical trial of vena caval filters in the prevention of pulmonary embolism in patients with proximal deep-vein thrombosis. N Engl J Med. 1998;338:409-415.
The Case
A 55-year-old female undergoes cholecystectomy. On post-operative Day 2, she develops right-lower-extremity swelling and pain; venous ultrasound detects a proximal deep venous thrombosis (DVT). The patient denies smoking or use of hormonal medications. She has no history of venous thromboembolism (VTE), although her brother had a DVT at age 60. The hospitalist team is consulted for management of acute DVT.
Overview
VTE, including lower- and upper-extremity DVT and pulmonary embolism (PE), is one of the most common and preventable hospital diseases. DVT with PE is associated with a 10% mortality rate, and DVT with post-thrombotic syndrome can be associated with significant morbidity, including pain, edema, skin/pigment change, venous dilation, and ulcer development.1,2 Recognition of clinical symptoms and risk factors for DVT (see Table 1) in conjunction with validated clinical scoring predictors (such as the Wells Prediction Rule) and a high-sensitivity D-dimer assay can help diagnose the condition and determine the need for ultrasound.3-7
Pharmacologic Treatment
Anticoagulation should be initiated in all patients with VTE, regardless of patient symptoms. Anticoagulant options include:
- Intravenous (IV) or subcutaneous (SC) unfractionated heparin (UFH);
- SC low-molecular-weight heparins (LMWH), such as enoxaparin and dalteparin; and
- Fondaparinux (as effective as LMWH for acute treatment of VTE).8
These agents can be used while transitioning to oral vitamin K antagonists (VKA), such as warfarin.3
The 2012 American College of Chest Physicians (ACCP) guidelines on antithrombotic therapy for VTE recommend initial therapy with LMWH or fondaparinux (rather than IV or SC UFH). The guidelines suggest that LMWH once-daily dosing is favored over twice-daily dosing, based mainly on patient convenience, although this is a weak recommendation (2C) based on the overall quality of the data. The recommendation applies only if the daily dosing of the LMWH, including tinzaparin, dalteparin, and nadroparin, is equivalent to the twice-daily dosing (i.e., dalteparin may be dosed at 100 units/kg BID vs. 200 units/kg daily). Of importance, enoxaparin has not been studied at a once-daily dose (2 mg/kg), which is equivalent to the twice-daily dosing regimen (1 mg/kg twice daily). Additionally, one study suggests that once-daily dosing of enoxaparin 1.5mg/kg might be inferior to 1 mg/kg twice-daily dosing; therefore, caution must be exercised in applying this recommendation to the LMWH enoxaparin at this time.3,27,28 (updated Aug. 28, 2012)
Warfarin should be started simultaneously at a usual daily dose of 5 mg for the first two days, with subsequent doses adjusted to achieve a goal international normalized ratio (INR) of 2.0 to 3.0. Parenteral agents should be given for a minimum of five days and until the INR has been >2.0 for at least 24 hours.3
The new factor-Xa inhibitor rivaroxaban and the direct thrombin inhibitor dabigatran are promising oral alternatives to warfarin.9-11 However, neither drug is currently FDA-approved for the treatment of VTE, nor are they recommended by current guidelines (given limited data for DVT treatment and concerns of bleeding risk).3,12,13 See Table 2 (above) for comparisons of common anticoagulants.3,14-17
Duration of anticoagulation. Anticoagulant treatment of acute DVT should continue for at least three months, as shorter durations are associated with higher recurrence rates. Longer treatment may be indicated depending on the patient’s risk of recurrence.3
The ACCP guidelines estimate risk of recurrence using primary, secondary, and additional factors (see Table 3, p. 19) and recommend the following durations:
- First episode provoked: three months (proximal or distal, provoked by surgery or a nonsurgical transient risk factor);
- First episode unprovoked distal: three months (see “Considerations for isolated distal DVT,” below);
- First episode unprovoked proximal: Indefinite if low to moderate bleeding risk, three months if high bleeding risk;
- Recurrent unprovoked: Indefinite if low to moderate bleeding risk, three months if high bleeding risk; and
- With active cancer: Indefinite with LMWH due to higher risk of recurrence.3,18
These treatment duration guidelines might need to be individualized based on other factors including patient preference, ability to obtain accurate INR monitoring (for those on warfarin), treatment cost, and comorbidities.3
Considerations for isolated distal DVT. Patients with an initial episode of distal DVT, without significant symptoms or risk factors for extension (e.g. positive D-dimer, extensive clot near proximal veins, absence of a reversible provoking factor, active cancer, inpatient status, or previous VTE) might not need anticoagulation.
The DVT can be followed with serial ultrasounds for the first two weeks; anticoagulation is recommended only if the thrombus extends during that time period. The development of significant symptoms or risk factors of extension might indicate the need for anticoagulation.3
Considerations for upper-extremity DVT (UEDVT). Anticoagulation for an UEDVT is generally consistent with the above guidelines for lower-extremity DVT, with a few caveats. If an UEDVT is associated with a central venous catheter (CVC), the CVC should be removed if possible; there are no recommendations to determine whether CVC removal should be preceded by a period of anticoagulation.
A catheter-associated UEDVT requires a minimum of three months of anticoagulation; if the CVC remains in place beyond three months, anticoagulation should be continued until the catheter is removed. Unprovoked UEDVT has a lower risk of recurrence than lower-extremity DVT and three months of anticoagulation, rather than indefinite therapy, is recommended.3
Mechanical Treatment
Non-pharmacologic therapies, such as knee-high graduated compression stockings with pressure of 30 mmHg to 40 mmHg at the ankle, can help reduce the morbidity of post-thrombotic syndrome (PTS) when combined with anticoagulation. Symptomatic patients who use compression stockings as soon as feasible and for a minimum of two years can reduce their incidence of PTS by 50%.3,19,20
Thigh-length stockings are not more effective than knee-high, and while multilayer compression bandages might relieve symptoms during the first-week post-DVT, they do not reduce the one-year incidence of PTS.21,22 Early mobilization is not associated with an increased risk of PE, extension of DVT, or death; patients should ambulate as soon as physically able.23,24
Pharmacomechanical Thrombolysis
For acute DVT, ACCP guidelines recommend anticoagulation alone over pharmacomechanical thrombolysis (either systemic or catheter-directed thrombolysis and mechanical thrombus fragmentation). The rare patient with impending venous gangrene despite anticoagulation is the only clinical scenario in which thrombolysis is clearly indicated. Patients who undergo pharmacomechanical thrombolysis still need a standard course of anticoagulation.3
Role for Inferior Vena Cava Filters
The optimal role of inferior vena cava (IVC) filters remains uncertain. Only one randomized trial found that IVC filters, in conjunction with systemic anticoagulation versus systemic anticoagulation alone, were associated with short-term reductions in the incidence of PE but long-term increases in recurrent DVT, with no differences in mortality or major bleeding. However, no trials have compared anticoagulation plus IVC filter placement with IVC filter placement alone.25,26
ACCP guidelines recommend IVC filter placement only in patients with acute, proximal DVT of the lower extremity, and a contraindication to anticoagulant therapy. If the contraindication resolves, a conventional course of anticoagulation can commence. Combining an IVC filter with an anticoagulant is not recommended. The risks and benefits of retrievable filters require further investigation.3
Back to the Case
Our patient has a provoked DVT secondary to a reversible risk factor (surgery) without additional clinical risk factors. Her family history of DVT is not significant (her brother was >age 50 when it occurred). This patient should be treated with LMWH or fondaparinux with initiation of warfarin with goal INR of 2.0 to 3.0 for at least three months. She does not need an IVC filter, and she should use compression stockings to reduce the risk of PTS.
Bottom Line
In hospitalized patients, treatment of DVT should include immediate anticoagulation with LMWH, fondaparinux, or IV heparin (in patients with renal failure) with transition to warfarin and a goal INR of 2.0 to 3.0. New oral anticoagulants could prove beneficial in acute treatment of DVT but require further testing. Duration of treatment is patient-specific, but most should be anticoagulated for at least three months; some warrant indefinite therapy based on risk factors.
Dr. Sebasky is an assistant professor and Dr. DeKorte is assistant professor of medicine in the division of hospital medicine at the University of California at San Diego.
References
- Agency for Healthcare Research and Quality. Talking Points to Attract Administration Support for Venous Thromboembolism Prevention Programs. U.S. Department of Health & Human Services website. Available at: http://www.ahrq.gov/qual/vtguide/vtguideapa.htm. Accessed Feb. 4, 2012.
- Kahn SR, Shbaklo H, Lamping DL, et al. Determinants of health-related quality of life during the 2 years following deep vein thrombosis. J Thromb Haemost. 2008;6:1105-1112.
- Kearon C, Akl E, Comerota AJ, et al. Antithrombotic Therapy for VTE Disease. Antithrombotic Therapy and Prevention of Thrombosis, 9th ed.: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(2 Suppl):e419S-e494S.
- Hirsh J, Hull RD, Raskob GE. Clinical features and diagnosis of venous thrombosis. J Am Coll Cardiol. 1986;8(6 Suppl B):114B-127B.
- Qaseem A, Snow V, Barry P, et al. Current diagnosis of venous thromboembolism in primary care: a clinical practice guideline from the American Academy of Family Physicians and the American College of Physicians. Ann Int Med. 2007;146:454-458.
- Tapson VF, Carroll BA, Davidson BL, et al. The diagnostic approach to acute venous thromboembolism. Clinical practice guideline. American Thoracic Society. Am J Respir Crit Care Med. 1999;160:1043-1066.
- Wells PS, Owen C, Doucette S, Fergusson D, Tran H. Does this patient have deep vein thrombosis? JAMA. 2006;295:199-207.
- Büller HR, Davidson BL, Decousus H, et al. Fondaparinux or enoxaparin for the initial treatment of symptomatic deep venous thrombosis: a randomized trial. Ann Intern Med. 2004;140:867-873.
- EINSTEIN Investigators, Bauersachs R, Berkowitz SD, et al. Oral rivaroxaban for symptomatic venous thromboembolism. N Engl J Med. 2010;363:2499-25
- Garcia, D, Libby E, Crowther M. The new oral anticoagulants. Blood. 2010;115:15-20.
- Douketis JD. Pharmacologic properties of the new oral anticoagulants: a clinician-oriented review with a focus on perioperative management. Curr Pharm Des. 2010;16:3436-3441.
- U.S. Food and Drug Administration. Pradaxa (dabigatran etexilate mesylate): Drug Safety Communication—Safety Review of Post-Market Reports of Serious Bleeding Events. U.S. Food and Drug Administration website. Available at: http://www.fda.gov/Safety/MedWatch/SafetyInformation/
SafetyAlertsforHumanMedicalProducts/ucm282820.htm. Accessed March 12, 2012.
- Levi M, Erenberg E, Kamphuisen PW. Bleeding risk and reversal strategies for old and new anticoagulants and antiplatelet agents. J Thromb Haemost. 2011;9:1705.
- Erkens PM, Prins MH. Fixed dose subcutaneous low molecular weight heparins versus adjusted dose unfractionated heparin for venous thromboembolism. Cochrane Database Syst Rev. 2010;8(9);CD001100.Vardi M, Zittan E, Bitterman H. Subcutaneous unfractionated heparin for the initial treatment of venous thromboembolism. Cochrane Database Syst Rev. 2009;(4):CD006771.
- Hirsh J, Levine MN. Low molecular weight heparin. Blood. 1992;79:1-17.
- Schulman S, Kearon C, Kakkar AK, et al. Dabigatran versus warfarin in the treatment of acute venous thromboembolism. N Engl J Med. 2009;361;2342-2352.
- Bauer KA. Long-term management of venous thromboembolism. JAMA. 2011;305:1336-1345.
- Prandoni P, Lensing AW, Prins MH, et al. Below-knee elastic compression stockings to prevent the postthrombotic syndrome: a randomized, controlled trial. Ann Intern Med. 2004;141:249-256.
- Brandjes DP, Büller HR, Heijboer H, et al. Randomised trial of effect of compression stockings in patients with symptomatic proximal-vein thrombosis. Lancet. 1997;349:759-762.
- Prandoni P, Noventa F, Quintavalla R, et al. Thigh-length versus below-knee compression elastic stockings for prevention of the post-thrombotic syndrome in patients with proximal-venous thrombosis: a randomized trial. Blood. 2012;119:1561-1565.
- Roumen-Klappe EM, den Heijer M, van Rossum J, et al. Multilayer compression bandaging in the acute phase of deep-vein thrombosis has no effect on the development of the post-thrombotic syndrome. J Thromb Thrombolysis. 2009;27:400-405.
- Aissaoui N, Martins E, Mouly S, Weber S, Meune C. A meta-analysis of bed rest versus early ambulation in the management of pulmonary embolism, deep venous thrombosis, or both. Int J Cardiol. 2009;137:37-41.
- Anderson CM, Overend TJ, Godwin J, Sealy C, Sunderji A. Ambulation after deep vein thrombosis: a systematic review. Physiother Can. 2009;61:133-140.
- Hann CL, Streiff MB. The role of vena caval filters in the management of venous thromboembolism. Blood Rev. 2005;19:179-202.
- Decousus H, Leizorovicz A, Page Y, et al. A clinical trial of vena caval filters in the prevention of pulmonary embolism in patients with proximal deep-vein thrombosis. N Engl J Med. 1998;338:409-415.
Ready to Reduce Your Hospital's Readmissions?
More than 100 hospitals across the country have used Project BOOST to reduce readmissions and improve their discharge processes. You and your hospital can be next by applying for Project BOOST. The deadline for the next national cohort of Project BOOST is Sept. 1.
To improve your chances of acceptance, start soon. In addition to an online form, the application requires a letter of support from an executive sponsor from each institution.
In October, accepted Project BOOST sites will receive:
- A comprehensive intervention developed by a panel of nationally recognized experts based on the best available evidence;
- A comprehensive implementation guide that provides step-by-step instructions and project management tools, such as the “teachback” training curriculum, to help interdisciplinary teams redesign work flow and plan, implement, and evaluate the intervention;
- Longitudinal technical assistance that provides face-to-face training and a year of expert mentoring and coaching to implement BOOST interventions that build a culture that supports safe and complete transitions. The mentoring program provides a “train-the-trainer” DVD and curriculum for nurses and case managers on using the teachback process, and webinars targeting the educational needs of other team members, including administrators, data analysts, physicians, nurses; and others;
- A collaboration that allows sites to communicate with and learn from each other via the BOOST listserv, BOOST community site, and quarterly all-site teleconferences and webinars; and
- Access to the BOOST data center, an online resource center that allows sites to store and benchmark data against control units and other sites and generate reports.
To start the application process, visit www.hospitalmedicine.org/boost.
Brendon Shank is SHM’s associate vice president for communications.
More than 100 hospitals across the country have used Project BOOST to reduce readmissions and improve their discharge processes. You and your hospital can be next by applying for Project BOOST. The deadline for the next national cohort of Project BOOST is Sept. 1.
To improve your chances of acceptance, start soon. In addition to an online form, the application requires a letter of support from an executive sponsor from each institution.
In October, accepted Project BOOST sites will receive:
- A comprehensive intervention developed by a panel of nationally recognized experts based on the best available evidence;
- A comprehensive implementation guide that provides step-by-step instructions and project management tools, such as the “teachback” training curriculum, to help interdisciplinary teams redesign work flow and plan, implement, and evaluate the intervention;
- Longitudinal technical assistance that provides face-to-face training and a year of expert mentoring and coaching to implement BOOST interventions that build a culture that supports safe and complete transitions. The mentoring program provides a “train-the-trainer” DVD and curriculum for nurses and case managers on using the teachback process, and webinars targeting the educational needs of other team members, including administrators, data analysts, physicians, nurses; and others;
- A collaboration that allows sites to communicate with and learn from each other via the BOOST listserv, BOOST community site, and quarterly all-site teleconferences and webinars; and
- Access to the BOOST data center, an online resource center that allows sites to store and benchmark data against control units and other sites and generate reports.
To start the application process, visit www.hospitalmedicine.org/boost.
Brendon Shank is SHM’s associate vice president for communications.
More than 100 hospitals across the country have used Project BOOST to reduce readmissions and improve their discharge processes. You and your hospital can be next by applying for Project BOOST. The deadline for the next national cohort of Project BOOST is Sept. 1.
To improve your chances of acceptance, start soon. In addition to an online form, the application requires a letter of support from an executive sponsor from each institution.
In October, accepted Project BOOST sites will receive:
- A comprehensive intervention developed by a panel of nationally recognized experts based on the best available evidence;
- A comprehensive implementation guide that provides step-by-step instructions and project management tools, such as the “teachback” training curriculum, to help interdisciplinary teams redesign work flow and plan, implement, and evaluate the intervention;
- Longitudinal technical assistance that provides face-to-face training and a year of expert mentoring and coaching to implement BOOST interventions that build a culture that supports safe and complete transitions. The mentoring program provides a “train-the-trainer” DVD and curriculum for nurses and case managers on using the teachback process, and webinars targeting the educational needs of other team members, including administrators, data analysts, physicians, nurses; and others;
- A collaboration that allows sites to communicate with and learn from each other via the BOOST listserv, BOOST community site, and quarterly all-site teleconferences and webinars; and
- Access to the BOOST data center, an online resource center that allows sites to store and benchmark data against control units and other sites and generate reports.
To start the application process, visit www.hospitalmedicine.org/boost.
Brendon Shank is SHM’s associate vice president for communications.
Adult Hospital Medicine Boot Camp Offers PAs, NPs Advanced Training
The Adult Hospital Medicine Boot Camp is a great way for physician assistants (PAs) and nurse practitioners (NPs) who practice as hospitalists to stay up to date on the latest in HM practice, and is the perfect opportunity to introduce new caregivers to the hospital setting.
Hosted by the American Academy of Physician Assistants (AAPA) and SHM, Adult Hospital Medicine Boot Camp will be Oct. 17-21 at the JW Marriott in New Orleans. Registration is available at www.aapa.org/bootcamp.
The specialized, education-focused “boot camp” immerses clinicians who are already practicing in HM, or who are interested in it, an intensive internal-medicine review of commonly encountered diagnoses and diseases in hospitalized adult patients. The boot camp has been approved for a maximum of 29.5 AAPA Category I CME credits.
The Adult Hospital Medicine Boot Camp is a great way for physician assistants (PAs) and nurse practitioners (NPs) who practice as hospitalists to stay up to date on the latest in HM practice, and is the perfect opportunity to introduce new caregivers to the hospital setting.
Hosted by the American Academy of Physician Assistants (AAPA) and SHM, Adult Hospital Medicine Boot Camp will be Oct. 17-21 at the JW Marriott in New Orleans. Registration is available at www.aapa.org/bootcamp.
The specialized, education-focused “boot camp” immerses clinicians who are already practicing in HM, or who are interested in it, an intensive internal-medicine review of commonly encountered diagnoses and diseases in hospitalized adult patients. The boot camp has been approved for a maximum of 29.5 AAPA Category I CME credits.
The Adult Hospital Medicine Boot Camp is a great way for physician assistants (PAs) and nurse practitioners (NPs) who practice as hospitalists to stay up to date on the latest in HM practice, and is the perfect opportunity to introduce new caregivers to the hospital setting.
Hosted by the American Academy of Physician Assistants (AAPA) and SHM, Adult Hospital Medicine Boot Camp will be Oct. 17-21 at the JW Marriott in New Orleans. Registration is available at www.aapa.org/bootcamp.
The specialized, education-focused “boot camp” immerses clinicians who are already practicing in HM, or who are interested in it, an intensive internal-medicine review of commonly encountered diagnoses and diseases in hospitalized adult patients. The boot camp has been approved for a maximum of 29.5 AAPA Category I CME credits.
Established Performance Metrics Help CMS Expand Its Value-Based Purchasing Program

—Thomas B. Valuck, MD, JD, senior vice president of strategic partnerships, National Quality Forum, former CMS adviser
No longer content to be a passive purchaser of healthcare services, the Centers for Medicare & Medicaid Services (CMS) is becoming a savvier shopper, holding providers increasingly accountable for the quality and efficiency of the care they deliver. With its value-based purchasing (VPB) program for hospitals already in place, now it’s the physicians’ turn.
CMS is marching toward a value-based payment modifier program that will adjust physician reimbursement based on the relative quality and efficiency of care that physicians provide to Medicare fee-for-service patients. The program will begin January 2015 and will extend to all physicians in 2017. Like the hospital VBP program, it will be budget-neutral—meaning that payment will increase for some physicians but decrease for others.
The coming months mark a pivotal period for physicians as CMS tweaks its accountability apparatus in ways that will determine how reimbursement will rise and fall, for whom, and for what.
Menu of Metrics
In crafting the payment modifier program, CMS can tap performance metrics from several of its existing programs, including the Physician Quality Reporting System (PQRS), the soon-to-be-expanded Physician Compare website (www.medicare.gov/find-a-doctor/provider-search.aspx), and the Physician Feedback Program.
“These agendas are part of a continuum, and of equal importance, in the evolution toward physician value-based purchasing,” says Patrick J. Torcson, MD, MMM, FACP, SFHM, chair of SHM’s Performance Measurement and Reporting Committee, and director of hospital medicine at St. Tammany Parish Hospital in Covington, La.
PQRS began as a voluntary “pay for reporting” system that gave physicians a modest financial bonus (currently 0.5% of allowable Medicare charges) for submitting quality data (left). The Affordable Care Act (ACA) has since authorized CMS to penalize physicians who do not participate—1.5% of allowable Medicare charges beginning in 2015, and 2% in 2016.
The Physician Compare website, launched at the end of 2010, currently contains such rudimentary information as education, gender, and whether a physician is enrolled in Medicare and satisfactorily reports data to the PQRS. But as of January, the site will begin reporting some PQRS data, as well as other metrics.
CMS’ Physician Feedback Program provides quality and cost information to physicians in an effort to encourage them to improve the care they provide and its efficiency. CMS recently combined the program with its value-based payment modifier program as it moves toward physician reimbursement that it says will reward “value rather than volume.” The program, currently being piloted in Iowa, Nebraska, Kansas, and Missouri, issues to physicians confidential quality and resource use reports (QRURs) that compare their performance to peer groups in similar specialties by tracking PQRS results, Healthcare Effectiveness Data and Information Set (HEDIS) measures, and per-capita cost data and preventable hospital admission rates for various medical conditions. CMS will roll out the program nationwide next year.
Metrics Lack Relevance
Developing performance measures that capture the most relevant activities of physicians across many different specialties with equal validity is notoriously difficult—something that CMS acknowledges.1
Assigning the right patient to the right physician (i.e. figuring out who contributed what care, in what proportion, to which patient) also is fraught with complications, especially in the inpatient care setting, where a patient is likely to see many different physicians during a hospitalization.
SHM president Shaun Frost, MD, SFHM, highlighted these challenges in a letter sent in May to acting CMS administrator Marilyn B. Tavenner in which he pointed to dramatic data deficiencies in the initial round of QRURs sent to Physician Feedback Program participants that included hospitalists in Iowa, Nebraska, Kansas and Missouri. Because hospitalists were categorized as general internal-medicine physicians in the reports, their per-capita cost of care was dramatically higher (73% higher, in one case study) than the average cost of all internal-medicine physicians. No allowance was made for distinguishing the outpatient-oriented practice of a general internist from the inherently more expensive inpatient-focused hospitalist practice.
In the case study reviewed by SHM, the hospitalist’s patients saw, on average, 28 different physicians over the course of a year, during which the hospitalist contributed to the care of many patients but did not direct the care of any one of them—facts that clearly highlight the difficulty of assigning responsibility and accountability for a patient’s care when comparing physician performance.
“Based on the measurement used in the QRUR, it seems likely that a hospitalist would be severely disadvantaged with the introduction of a value-based modifier based on the present QRUR methodology,” Dr. Frost wrote.
SHM is similarly critical of the PQRS measures, which Dr. Torcson says lack relevance to hospitalist practices. “We want to be defined as HM physicians with our own unique measures of quality and cost,” he says. “Our results will look very different from those of an internist with a primarily outpatient practice.”
Dr. Torcson notes that SHM is an active participant in providing feedback during CMS rule proposals and has offered to work with the CMS on further refining the measures. For example, SHM proposed adding additional measures related to care transitions, given their particular relevance to hospitalist practices.
Rule-Changing Reform
The disruptive innovation of CMS’ healthcare reform agenda might wind up being a game-changer that dramatically affects the contours of all provider performance reporting and incentive systems, redefining the issues of physician accountability and patient assignment.
“We’re going to need to figure out how to restructure our measurement systems to match our evolving healthcare delivery and payment systems,” says Thomas B. Valuck, MD, JD, senior vice president of strategic partnerships for the National Quality Forum and former CMS adviser to the VBP program. Healthcare quality reporting should focus more on measures that cut across care contexts and assess whether the care provided truly made a difference for patients—metrics such as health improvement, return to functional status, level of patient involvement in the management of their care, provider team coordination, and other patient needs and preferences, Dr. Valuck believes.
“We need to be focused more on measures that encourage joint responsibility and cooperation among providers, and are important to patients across hospital, post-acute, and ambulatory settings, rather than those that are compartmentalized to one setting or relevant only to specific diseases or subspecialties,” Dr. Valuck says.
Such measure sets, while still retaining some disease- and physician-specific metrics, ideally would be complementary with families of related measures at the community, state, and national levels, Dr. Valuck says. “Such a multidimensional framework can begin to tell a meaningful story about what’s happening to the patient, and how well our system is delivering the right care,” he adds.
Dr. Torcson says HM has a pioneering role to play in this evolution, and he notes that SHM has proposed that CMS harmonize measures that align hospital-based physician activities (e.g. hospital medicine, emergency medicine, anesthesia, radiology) with hospital-level performance agendas so that physicians practicing together in the hospital setting can report on measures that are relevant to both.
Christopher Guadagnino is a freelance medical writer in Philadelphia.
Reference
- Centers for Medicare & Medicaid Services. Physician Quality Reporting System Town Hall Meeting. Available at: http://www.usqualitymeasures.org/shared/content/C4M_PQRS_transcript.pdf. Accessed July 3, 2012.

—Thomas B. Valuck, MD, JD, senior vice president of strategic partnerships, National Quality Forum, former CMS adviser
No longer content to be a passive purchaser of healthcare services, the Centers for Medicare & Medicaid Services (CMS) is becoming a savvier shopper, holding providers increasingly accountable for the quality and efficiency of the care they deliver. With its value-based purchasing (VPB) program for hospitals already in place, now it’s the physicians’ turn.
CMS is marching toward a value-based payment modifier program that will adjust physician reimbursement based on the relative quality and efficiency of care that physicians provide to Medicare fee-for-service patients. The program will begin January 2015 and will extend to all physicians in 2017. Like the hospital VBP program, it will be budget-neutral—meaning that payment will increase for some physicians but decrease for others.
The coming months mark a pivotal period for physicians as CMS tweaks its accountability apparatus in ways that will determine how reimbursement will rise and fall, for whom, and for what.
Menu of Metrics
In crafting the payment modifier program, CMS can tap performance metrics from several of its existing programs, including the Physician Quality Reporting System (PQRS), the soon-to-be-expanded Physician Compare website (www.medicare.gov/find-a-doctor/provider-search.aspx), and the Physician Feedback Program.
“These agendas are part of a continuum, and of equal importance, in the evolution toward physician value-based purchasing,” says Patrick J. Torcson, MD, MMM, FACP, SFHM, chair of SHM’s Performance Measurement and Reporting Committee, and director of hospital medicine at St. Tammany Parish Hospital in Covington, La.
PQRS began as a voluntary “pay for reporting” system that gave physicians a modest financial bonus (currently 0.5% of allowable Medicare charges) for submitting quality data (left). The Affordable Care Act (ACA) has since authorized CMS to penalize physicians who do not participate—1.5% of allowable Medicare charges beginning in 2015, and 2% in 2016.
The Physician Compare website, launched at the end of 2010, currently contains such rudimentary information as education, gender, and whether a physician is enrolled in Medicare and satisfactorily reports data to the PQRS. But as of January, the site will begin reporting some PQRS data, as well as other metrics.
CMS’ Physician Feedback Program provides quality and cost information to physicians in an effort to encourage them to improve the care they provide and its efficiency. CMS recently combined the program with its value-based payment modifier program as it moves toward physician reimbursement that it says will reward “value rather than volume.” The program, currently being piloted in Iowa, Nebraska, Kansas, and Missouri, issues to physicians confidential quality and resource use reports (QRURs) that compare their performance to peer groups in similar specialties by tracking PQRS results, Healthcare Effectiveness Data and Information Set (HEDIS) measures, and per-capita cost data and preventable hospital admission rates for various medical conditions. CMS will roll out the program nationwide next year.
Metrics Lack Relevance
Developing performance measures that capture the most relevant activities of physicians across many different specialties with equal validity is notoriously difficult—something that CMS acknowledges.1
Assigning the right patient to the right physician (i.e. figuring out who contributed what care, in what proportion, to which patient) also is fraught with complications, especially in the inpatient care setting, where a patient is likely to see many different physicians during a hospitalization.
SHM president Shaun Frost, MD, SFHM, highlighted these challenges in a letter sent in May to acting CMS administrator Marilyn B. Tavenner in which he pointed to dramatic data deficiencies in the initial round of QRURs sent to Physician Feedback Program participants that included hospitalists in Iowa, Nebraska, Kansas and Missouri. Because hospitalists were categorized as general internal-medicine physicians in the reports, their per-capita cost of care was dramatically higher (73% higher, in one case study) than the average cost of all internal-medicine physicians. No allowance was made for distinguishing the outpatient-oriented practice of a general internist from the inherently more expensive inpatient-focused hospitalist practice.
In the case study reviewed by SHM, the hospitalist’s patients saw, on average, 28 different physicians over the course of a year, during which the hospitalist contributed to the care of many patients but did not direct the care of any one of them—facts that clearly highlight the difficulty of assigning responsibility and accountability for a patient’s care when comparing physician performance.
“Based on the measurement used in the QRUR, it seems likely that a hospitalist would be severely disadvantaged with the introduction of a value-based modifier based on the present QRUR methodology,” Dr. Frost wrote.
SHM is similarly critical of the PQRS measures, which Dr. Torcson says lack relevance to hospitalist practices. “We want to be defined as HM physicians with our own unique measures of quality and cost,” he says. “Our results will look very different from those of an internist with a primarily outpatient practice.”
Dr. Torcson notes that SHM is an active participant in providing feedback during CMS rule proposals and has offered to work with the CMS on further refining the measures. For example, SHM proposed adding additional measures related to care transitions, given their particular relevance to hospitalist practices.
Rule-Changing Reform
The disruptive innovation of CMS’ healthcare reform agenda might wind up being a game-changer that dramatically affects the contours of all provider performance reporting and incentive systems, redefining the issues of physician accountability and patient assignment.
“We’re going to need to figure out how to restructure our measurement systems to match our evolving healthcare delivery and payment systems,” says Thomas B. Valuck, MD, JD, senior vice president of strategic partnerships for the National Quality Forum and former CMS adviser to the VBP program. Healthcare quality reporting should focus more on measures that cut across care contexts and assess whether the care provided truly made a difference for patients—metrics such as health improvement, return to functional status, level of patient involvement in the management of their care, provider team coordination, and other patient needs and preferences, Dr. Valuck believes.
“We need to be focused more on measures that encourage joint responsibility and cooperation among providers, and are important to patients across hospital, post-acute, and ambulatory settings, rather than those that are compartmentalized to one setting or relevant only to specific diseases or subspecialties,” Dr. Valuck says.
Such measure sets, while still retaining some disease- and physician-specific metrics, ideally would be complementary with families of related measures at the community, state, and national levels, Dr. Valuck says. “Such a multidimensional framework can begin to tell a meaningful story about what’s happening to the patient, and how well our system is delivering the right care,” he adds.
Dr. Torcson says HM has a pioneering role to play in this evolution, and he notes that SHM has proposed that CMS harmonize measures that align hospital-based physician activities (e.g. hospital medicine, emergency medicine, anesthesia, radiology) with hospital-level performance agendas so that physicians practicing together in the hospital setting can report on measures that are relevant to both.
Christopher Guadagnino is a freelance medical writer in Philadelphia.
Reference
- Centers for Medicare & Medicaid Services. Physician Quality Reporting System Town Hall Meeting. Available at: http://www.usqualitymeasures.org/shared/content/C4M_PQRS_transcript.pdf. Accessed July 3, 2012.

—Thomas B. Valuck, MD, JD, senior vice president of strategic partnerships, National Quality Forum, former CMS adviser
No longer content to be a passive purchaser of healthcare services, the Centers for Medicare & Medicaid Services (CMS) is becoming a savvier shopper, holding providers increasingly accountable for the quality and efficiency of the care they deliver. With its value-based purchasing (VPB) program for hospitals already in place, now it’s the physicians’ turn.
CMS is marching toward a value-based payment modifier program that will adjust physician reimbursement based on the relative quality and efficiency of care that physicians provide to Medicare fee-for-service patients. The program will begin January 2015 and will extend to all physicians in 2017. Like the hospital VBP program, it will be budget-neutral—meaning that payment will increase for some physicians but decrease for others.
The coming months mark a pivotal period for physicians as CMS tweaks its accountability apparatus in ways that will determine how reimbursement will rise and fall, for whom, and for what.
Menu of Metrics
In crafting the payment modifier program, CMS can tap performance metrics from several of its existing programs, including the Physician Quality Reporting System (PQRS), the soon-to-be-expanded Physician Compare website (www.medicare.gov/find-a-doctor/provider-search.aspx), and the Physician Feedback Program.
“These agendas are part of a continuum, and of equal importance, in the evolution toward physician value-based purchasing,” says Patrick J. Torcson, MD, MMM, FACP, SFHM, chair of SHM’s Performance Measurement and Reporting Committee, and director of hospital medicine at St. Tammany Parish Hospital in Covington, La.
PQRS began as a voluntary “pay for reporting” system that gave physicians a modest financial bonus (currently 0.5% of allowable Medicare charges) for submitting quality data (left). The Affordable Care Act (ACA) has since authorized CMS to penalize physicians who do not participate—1.5% of allowable Medicare charges beginning in 2015, and 2% in 2016.
The Physician Compare website, launched at the end of 2010, currently contains such rudimentary information as education, gender, and whether a physician is enrolled in Medicare and satisfactorily reports data to the PQRS. But as of January, the site will begin reporting some PQRS data, as well as other metrics.
CMS’ Physician Feedback Program provides quality and cost information to physicians in an effort to encourage them to improve the care they provide and its efficiency. CMS recently combined the program with its value-based payment modifier program as it moves toward physician reimbursement that it says will reward “value rather than volume.” The program, currently being piloted in Iowa, Nebraska, Kansas, and Missouri, issues to physicians confidential quality and resource use reports (QRURs) that compare their performance to peer groups in similar specialties by tracking PQRS results, Healthcare Effectiveness Data and Information Set (HEDIS) measures, and per-capita cost data and preventable hospital admission rates for various medical conditions. CMS will roll out the program nationwide next year.
Metrics Lack Relevance
Developing performance measures that capture the most relevant activities of physicians across many different specialties with equal validity is notoriously difficult—something that CMS acknowledges.1
Assigning the right patient to the right physician (i.e. figuring out who contributed what care, in what proportion, to which patient) also is fraught with complications, especially in the inpatient care setting, where a patient is likely to see many different physicians during a hospitalization.
SHM president Shaun Frost, MD, SFHM, highlighted these challenges in a letter sent in May to acting CMS administrator Marilyn B. Tavenner in which he pointed to dramatic data deficiencies in the initial round of QRURs sent to Physician Feedback Program participants that included hospitalists in Iowa, Nebraska, Kansas and Missouri. Because hospitalists were categorized as general internal-medicine physicians in the reports, their per-capita cost of care was dramatically higher (73% higher, in one case study) than the average cost of all internal-medicine physicians. No allowance was made for distinguishing the outpatient-oriented practice of a general internist from the inherently more expensive inpatient-focused hospitalist practice.
In the case study reviewed by SHM, the hospitalist’s patients saw, on average, 28 different physicians over the course of a year, during which the hospitalist contributed to the care of many patients but did not direct the care of any one of them—facts that clearly highlight the difficulty of assigning responsibility and accountability for a patient’s care when comparing physician performance.
“Based on the measurement used in the QRUR, it seems likely that a hospitalist would be severely disadvantaged with the introduction of a value-based modifier based on the present QRUR methodology,” Dr. Frost wrote.
SHM is similarly critical of the PQRS measures, which Dr. Torcson says lack relevance to hospitalist practices. “We want to be defined as HM physicians with our own unique measures of quality and cost,” he says. “Our results will look very different from those of an internist with a primarily outpatient practice.”
Dr. Torcson notes that SHM is an active participant in providing feedback during CMS rule proposals and has offered to work with the CMS on further refining the measures. For example, SHM proposed adding additional measures related to care transitions, given their particular relevance to hospitalist practices.
Rule-Changing Reform
The disruptive innovation of CMS’ healthcare reform agenda might wind up being a game-changer that dramatically affects the contours of all provider performance reporting and incentive systems, redefining the issues of physician accountability and patient assignment.
“We’re going to need to figure out how to restructure our measurement systems to match our evolving healthcare delivery and payment systems,” says Thomas B. Valuck, MD, JD, senior vice president of strategic partnerships for the National Quality Forum and former CMS adviser to the VBP program. Healthcare quality reporting should focus more on measures that cut across care contexts and assess whether the care provided truly made a difference for patients—metrics such as health improvement, return to functional status, level of patient involvement in the management of their care, provider team coordination, and other patient needs and preferences, Dr. Valuck believes.
“We need to be focused more on measures that encourage joint responsibility and cooperation among providers, and are important to patients across hospital, post-acute, and ambulatory settings, rather than those that are compartmentalized to one setting or relevant only to specific diseases or subspecialties,” Dr. Valuck says.
Such measure sets, while still retaining some disease- and physician-specific metrics, ideally would be complementary with families of related measures at the community, state, and national levels, Dr. Valuck says. “Such a multidimensional framework can begin to tell a meaningful story about what’s happening to the patient, and how well our system is delivering the right care,” he adds.
Dr. Torcson says HM has a pioneering role to play in this evolution, and he notes that SHM has proposed that CMS harmonize measures that align hospital-based physician activities (e.g. hospital medicine, emergency medicine, anesthesia, radiology) with hospital-level performance agendas so that physicians practicing together in the hospital setting can report on measures that are relevant to both.
Christopher Guadagnino is a freelance medical writer in Philadelphia.
Reference
- Centers for Medicare & Medicaid Services. Physician Quality Reporting System Town Hall Meeting. Available at: http://www.usqualitymeasures.org/shared/content/C4M_PQRS_transcript.pdf. Accessed July 3, 2012.
Know Surgical Package Requirements before Billing Postoperative Care
Hospitalists often are involved in the postoperative care of the surgical patient. However, HM is emerging in the admitting/attending role for procedural patients. Confusion can arise as to the nature of the hospitalist service, and whether it is deemed billable. Knowing the surgical package requirements can help hospitalists consider the issues.
Global Surgical Package Period1
Surgical procedures, categorized as major or minor surgery, are reimbursed for pre-, intra-, and postoperative care. Postoperative care varies according to the procedure’s assigned global period, which designates zero, 10, or 90 postoperative days. (Physicians can review the global period for any given CPT code in the Medicare Physician Fee Schedule, available at www.cms.gov/apps/physician-fee-schedule/search/search-criteria.aspx.)
Services classified with “XXX” do not have the global period concept. “ZZZ” services denote an “add-on” procedure code that must always be reported with a primary procedure code and assumes the global period assigned to the primary procedure performed.
Major surgery allocates a 90-day global period in which the surgeon is responsible for all related surgical care one day before surgery through 90 postoperative days with no additional charge. Minor surgery, including endoscopy, appoints a zero-day or 10-day postoperative period. The zero-day global period encompasses only services provided on the surgical day, whereas 10-day global periods include services on the surgical day through 10 postoperative days.
Global Surgical Package Components2
The global surgical package comprises a host of responsibilities that include standard facility requirements of filling out all necessary paperwork involved in surgical cases (e.g. preoperative H&P, operative consent forms, preoperative orders). Additionally, the surgeon’s packaged payment includes (at no extra charge):
- Preoperative visits after making the decision for surgery beginning one day prior to surgery;
- All additional postoperative medical or surgical services provided by the surgeon related to complications but not requiring additional trips to the operating room;
- Postoperative visits by the surgeon related to recovery from surgery, including but not limited to dressing changes; local incisional care; removal of cutaneous sutures and staples; line removals; changes and removal of tracheostomy tubes; and discharge services; and
- Postoperative pain management provided by the surgeon.
- Examples of services that are not included in the global surgical package, (i.e. are separately billable and may require an appropriate modifier) are:
- The initial consultation or evaluation of the problem by the surgeon to determine the need for surgery;
- Services of other physicians except where the other physicians are providing coverage for the surgeon or agree on a transfer of care (i.e. a formal agreement in the form of a letter or an annotation in the discharge summary, hospital record, or ASC record);
- Postoperative visits by the surgeon unrelated to the diagnosis for which the surgical procedure is performed, unless the visits occur due to complications of the surgery;
- Diagnostic tests and procedures, including diagnostic radiological procedures;
- Clearly distinct surgical procedures during the postoperative period that do not result in repeat operations or treatment for complications;
- Treatment for postoperative complications that requires a return trip to the operating room (OR), catheterization lab or endoscopy suite;
- Immunosuppressive therapy for organ transplants; and
- Critical-care services (CPT codes 99291 and 99292) unrelated to the surgery where a seriously injured or burned patient is critically ill and requires constant attendance of the surgeon.
Classification of “Surgeon”
For billing purposes, the “surgeon” is a qualified physician who can perform “surgical” services within their scope of practice. All physicians with the same specialty designation in the same group practice as the “surgeon” (i.e. reporting services under the same tax identification number) are considered a single entity and must adhere to the global period billing rules initiated by the “surgeon.”
Alternately, physicians with different specialty designations in the same group practice (e.g. a hospitalist and a cardiologist in a multispecialty group who report services under the same tax identification number) or different group practices can perform and separately report medically necessary services during the surgeon’s global period, as long as a formal (mutually agreed-upon) transfer of care did not occur.
Medical Necessity
With the growth of HM programs and the admission/attending role expansion, involvement in surgical cases comes under scrutiny for medical necessity. Admitting a patient who has active medical conditions (e.g. hypertension, diabetes, emphysema) is reasonable and necessary because the patient has a well-defined need for medical management by the hospitalist. Participation in the care of these patients is separately billable from the surgeon’s global period package.
Alternatively, a hospitalist might be required to admit and follow surgical patients who have no other identifiable chronic or acute conditions aside from the surgical problem. In these cases, hospitalist involvement may satisfy facility policy (quality of care, risk reduction, etc.) and administrative functions (discharge services or coordination of care) rather than active clinical management. This “medical management” will not be considered “medically necessary” by the payor, and may be denied as incidental to the surgeon’s perioperative services. Erroneous payment can occur, which will result in refund requests, as payors do not want to pay twice for duplicate services. Hospitalists can attempt to negotiate other terms with facilities to account for the unpaid time and effort directed toward these types of cases.
Consider the Case
A patient with numerous medical comorbidities is admitted to the hospitalist service for stabilization prior to surgery, which will occur the next day. The hospitalist can report the appropriate admission code (99221-99223) without need for modifiers because the hospitalist is the attending of record and in a different specialty group. If a private insurer denies the claim as inclusive to the surgical service, the hospitalist can appeal with notes and a cover letter, along with the Medicare guidelines for global surgical package. The hospitalist may continue to provide postoperative daily care, as needed, to manage the patient’s chronic conditions, and report each service as subsequent hospital care (99231-99233) without modifier until the day of discharge (99238-99239). Again, if a payor issues a denial (inclusive to surgery), appealing with notes might be necessary.
Carol Pohlig is a billing and coding expert with the University of Pennsylvania Medical Center, Philadelphia. She is faculty for SHM’s inpatient coding course.
References
- Centers for Medicare & Medicaid Services. Medicare Claims Processing Manual: Chapter 12, Section 40. Centers for Medicare & Medicaid Services website. Available at: http://www.cms.gov/Regulations-and-Guidance/Guidance/Manuals/Downloads/clm104c12.pdf. Accessed May 5, 2012.
- Centers for Medicare & Medicaid Services. ICD-10: HHS proposes one-year delay of ICD-10 compliance date. Centers for Medicare & Medicaid Services website. Available at: http://www.cms.gov/Medicare/Coding/ICD10/index.html?redirect=/ICD10. Accessed May 5, 2012.
- Abraham M, Ahlman J, Anderson C, Boudreau A, Connelly J. Current Procedural Terminology 2012 Professional Edition. Chicago: American Medical Association Press; 2011.
Hospitalists often are involved in the postoperative care of the surgical patient. However, HM is emerging in the admitting/attending role for procedural patients. Confusion can arise as to the nature of the hospitalist service, and whether it is deemed billable. Knowing the surgical package requirements can help hospitalists consider the issues.
Global Surgical Package Period1
Surgical procedures, categorized as major or minor surgery, are reimbursed for pre-, intra-, and postoperative care. Postoperative care varies according to the procedure’s assigned global period, which designates zero, 10, or 90 postoperative days. (Physicians can review the global period for any given CPT code in the Medicare Physician Fee Schedule, available at www.cms.gov/apps/physician-fee-schedule/search/search-criteria.aspx.)
Services classified with “XXX” do not have the global period concept. “ZZZ” services denote an “add-on” procedure code that must always be reported with a primary procedure code and assumes the global period assigned to the primary procedure performed.
Major surgery allocates a 90-day global period in which the surgeon is responsible for all related surgical care one day before surgery through 90 postoperative days with no additional charge. Minor surgery, including endoscopy, appoints a zero-day or 10-day postoperative period. The zero-day global period encompasses only services provided on the surgical day, whereas 10-day global periods include services on the surgical day through 10 postoperative days.
Global Surgical Package Components2
The global surgical package comprises a host of responsibilities that include standard facility requirements of filling out all necessary paperwork involved in surgical cases (e.g. preoperative H&P, operative consent forms, preoperative orders). Additionally, the surgeon’s packaged payment includes (at no extra charge):
- Preoperative visits after making the decision for surgery beginning one day prior to surgery;
- All additional postoperative medical or surgical services provided by the surgeon related to complications but not requiring additional trips to the operating room;
- Postoperative visits by the surgeon related to recovery from surgery, including but not limited to dressing changes; local incisional care; removal of cutaneous sutures and staples; line removals; changes and removal of tracheostomy tubes; and discharge services; and
- Postoperative pain management provided by the surgeon.
- Examples of services that are not included in the global surgical package, (i.e. are separately billable and may require an appropriate modifier) are:
- The initial consultation or evaluation of the problem by the surgeon to determine the need for surgery;
- Services of other physicians except where the other physicians are providing coverage for the surgeon or agree on a transfer of care (i.e. a formal agreement in the form of a letter or an annotation in the discharge summary, hospital record, or ASC record);
- Postoperative visits by the surgeon unrelated to the diagnosis for which the surgical procedure is performed, unless the visits occur due to complications of the surgery;
- Diagnostic tests and procedures, including diagnostic radiological procedures;
- Clearly distinct surgical procedures during the postoperative period that do not result in repeat operations or treatment for complications;
- Treatment for postoperative complications that requires a return trip to the operating room (OR), catheterization lab or endoscopy suite;
- Immunosuppressive therapy for organ transplants; and
- Critical-care services (CPT codes 99291 and 99292) unrelated to the surgery where a seriously injured or burned patient is critically ill and requires constant attendance of the surgeon.
Classification of “Surgeon”
For billing purposes, the “surgeon” is a qualified physician who can perform “surgical” services within their scope of practice. All physicians with the same specialty designation in the same group practice as the “surgeon” (i.e. reporting services under the same tax identification number) are considered a single entity and must adhere to the global period billing rules initiated by the “surgeon.”
Alternately, physicians with different specialty designations in the same group practice (e.g. a hospitalist and a cardiologist in a multispecialty group who report services under the same tax identification number) or different group practices can perform and separately report medically necessary services during the surgeon’s global period, as long as a formal (mutually agreed-upon) transfer of care did not occur.
Medical Necessity
With the growth of HM programs and the admission/attending role expansion, involvement in surgical cases comes under scrutiny for medical necessity. Admitting a patient who has active medical conditions (e.g. hypertension, diabetes, emphysema) is reasonable and necessary because the patient has a well-defined need for medical management by the hospitalist. Participation in the care of these patients is separately billable from the surgeon’s global period package.
Alternatively, a hospitalist might be required to admit and follow surgical patients who have no other identifiable chronic or acute conditions aside from the surgical problem. In these cases, hospitalist involvement may satisfy facility policy (quality of care, risk reduction, etc.) and administrative functions (discharge services or coordination of care) rather than active clinical management. This “medical management” will not be considered “medically necessary” by the payor, and may be denied as incidental to the surgeon’s perioperative services. Erroneous payment can occur, which will result in refund requests, as payors do not want to pay twice for duplicate services. Hospitalists can attempt to negotiate other terms with facilities to account for the unpaid time and effort directed toward these types of cases.
Consider the Case
A patient with numerous medical comorbidities is admitted to the hospitalist service for stabilization prior to surgery, which will occur the next day. The hospitalist can report the appropriate admission code (99221-99223) without need for modifiers because the hospitalist is the attending of record and in a different specialty group. If a private insurer denies the claim as inclusive to the surgical service, the hospitalist can appeal with notes and a cover letter, along with the Medicare guidelines for global surgical package. The hospitalist may continue to provide postoperative daily care, as needed, to manage the patient’s chronic conditions, and report each service as subsequent hospital care (99231-99233) without modifier until the day of discharge (99238-99239). Again, if a payor issues a denial (inclusive to surgery), appealing with notes might be necessary.
Carol Pohlig is a billing and coding expert with the University of Pennsylvania Medical Center, Philadelphia. She is faculty for SHM’s inpatient coding course.
References
- Centers for Medicare & Medicaid Services. Medicare Claims Processing Manual: Chapter 12, Section 40. Centers for Medicare & Medicaid Services website. Available at: http://www.cms.gov/Regulations-and-Guidance/Guidance/Manuals/Downloads/clm104c12.pdf. Accessed May 5, 2012.
- Centers for Medicare & Medicaid Services. ICD-10: HHS proposes one-year delay of ICD-10 compliance date. Centers for Medicare & Medicaid Services website. Available at: http://www.cms.gov/Medicare/Coding/ICD10/index.html?redirect=/ICD10. Accessed May 5, 2012.
- Abraham M, Ahlman J, Anderson C, Boudreau A, Connelly J. Current Procedural Terminology 2012 Professional Edition. Chicago: American Medical Association Press; 2011.
Hospitalists often are involved in the postoperative care of the surgical patient. However, HM is emerging in the admitting/attending role for procedural patients. Confusion can arise as to the nature of the hospitalist service, and whether it is deemed billable. Knowing the surgical package requirements can help hospitalists consider the issues.
Global Surgical Package Period1
Surgical procedures, categorized as major or minor surgery, are reimbursed for pre-, intra-, and postoperative care. Postoperative care varies according to the procedure’s assigned global period, which designates zero, 10, or 90 postoperative days. (Physicians can review the global period for any given CPT code in the Medicare Physician Fee Schedule, available at www.cms.gov/apps/physician-fee-schedule/search/search-criteria.aspx.)
Services classified with “XXX” do not have the global period concept. “ZZZ” services denote an “add-on” procedure code that must always be reported with a primary procedure code and assumes the global period assigned to the primary procedure performed.
Major surgery allocates a 90-day global period in which the surgeon is responsible for all related surgical care one day before surgery through 90 postoperative days with no additional charge. Minor surgery, including endoscopy, appoints a zero-day or 10-day postoperative period. The zero-day global period encompasses only services provided on the surgical day, whereas 10-day global periods include services on the surgical day through 10 postoperative days.
Global Surgical Package Components2
The global surgical package comprises a host of responsibilities that include standard facility requirements of filling out all necessary paperwork involved in surgical cases (e.g. preoperative H&P, operative consent forms, preoperative orders). Additionally, the surgeon’s packaged payment includes (at no extra charge):
- Preoperative visits after making the decision for surgery beginning one day prior to surgery;
- All additional postoperative medical or surgical services provided by the surgeon related to complications but not requiring additional trips to the operating room;
- Postoperative visits by the surgeon related to recovery from surgery, including but not limited to dressing changes; local incisional care; removal of cutaneous sutures and staples; line removals; changes and removal of tracheostomy tubes; and discharge services; and
- Postoperative pain management provided by the surgeon.
- Examples of services that are not included in the global surgical package, (i.e. are separately billable and may require an appropriate modifier) are:
- The initial consultation or evaluation of the problem by the surgeon to determine the need for surgery;
- Services of other physicians except where the other physicians are providing coverage for the surgeon or agree on a transfer of care (i.e. a formal agreement in the form of a letter or an annotation in the discharge summary, hospital record, or ASC record);
- Postoperative visits by the surgeon unrelated to the diagnosis for which the surgical procedure is performed, unless the visits occur due to complications of the surgery;
- Diagnostic tests and procedures, including diagnostic radiological procedures;
- Clearly distinct surgical procedures during the postoperative period that do not result in repeat operations or treatment for complications;
- Treatment for postoperative complications that requires a return trip to the operating room (OR), catheterization lab or endoscopy suite;
- Immunosuppressive therapy for organ transplants; and
- Critical-care services (CPT codes 99291 and 99292) unrelated to the surgery where a seriously injured or burned patient is critically ill and requires constant attendance of the surgeon.
Classification of “Surgeon”
For billing purposes, the “surgeon” is a qualified physician who can perform “surgical” services within their scope of practice. All physicians with the same specialty designation in the same group practice as the “surgeon” (i.e. reporting services under the same tax identification number) are considered a single entity and must adhere to the global period billing rules initiated by the “surgeon.”
Alternately, physicians with different specialty designations in the same group practice (e.g. a hospitalist and a cardiologist in a multispecialty group who report services under the same tax identification number) or different group practices can perform and separately report medically necessary services during the surgeon’s global period, as long as a formal (mutually agreed-upon) transfer of care did not occur.
Medical Necessity
With the growth of HM programs and the admission/attending role expansion, involvement in surgical cases comes under scrutiny for medical necessity. Admitting a patient who has active medical conditions (e.g. hypertension, diabetes, emphysema) is reasonable and necessary because the patient has a well-defined need for medical management by the hospitalist. Participation in the care of these patients is separately billable from the surgeon’s global period package.
Alternatively, a hospitalist might be required to admit and follow surgical patients who have no other identifiable chronic or acute conditions aside from the surgical problem. In these cases, hospitalist involvement may satisfy facility policy (quality of care, risk reduction, etc.) and administrative functions (discharge services or coordination of care) rather than active clinical management. This “medical management” will not be considered “medically necessary” by the payor, and may be denied as incidental to the surgeon’s perioperative services. Erroneous payment can occur, which will result in refund requests, as payors do not want to pay twice for duplicate services. Hospitalists can attempt to negotiate other terms with facilities to account for the unpaid time and effort directed toward these types of cases.
Consider the Case
A patient with numerous medical comorbidities is admitted to the hospitalist service for stabilization prior to surgery, which will occur the next day. The hospitalist can report the appropriate admission code (99221-99223) without need for modifiers because the hospitalist is the attending of record and in a different specialty group. If a private insurer denies the claim as inclusive to the surgical service, the hospitalist can appeal with notes and a cover letter, along with the Medicare guidelines for global surgical package. The hospitalist may continue to provide postoperative daily care, as needed, to manage the patient’s chronic conditions, and report each service as subsequent hospital care (99231-99233) without modifier until the day of discharge (99238-99239). Again, if a payor issues a denial (inclusive to surgery), appealing with notes might be necessary.
Carol Pohlig is a billing and coding expert with the University of Pennsylvania Medical Center, Philadelphia. She is faculty for SHM’s inpatient coding course.
References
- Centers for Medicare & Medicaid Services. Medicare Claims Processing Manual: Chapter 12, Section 40. Centers for Medicare & Medicaid Services website. Available at: http://www.cms.gov/Regulations-and-Guidance/Guidance/Manuals/Downloads/clm104c12.pdf. Accessed May 5, 2012.
- Centers for Medicare & Medicaid Services. ICD-10: HHS proposes one-year delay of ICD-10 compliance date. Centers for Medicare & Medicaid Services website. Available at: http://www.cms.gov/Medicare/Coding/ICD10/index.html?redirect=/ICD10. Accessed May 5, 2012.
- Abraham M, Ahlman J, Anderson C, Boudreau A, Connelly J. Current Procedural Terminology 2012 Professional Edition. Chicago: American Medical Association Press; 2011.