User login
Epidemiology of Bacteremia in Young Infants is Changing
Clinical question: What is the epidemiology of bacteremia in one-week to three-month-old infants?
Background: Large studies of bacteremia in infants <90 days of age were largely performed before the era of routine prenatal screening and prophylaxis for Group B Streptococcus (GBS). Additionally, these studies have focused on febrile infants, which might not allow for characterization of the incidence of bacteremia when nonfebrile infants are considered.
Study design: Retrospective review.
Setting: Large HMO database.
Synopsis: Of 160,818 full-term infants born at Kaiser Permanente Northern California from 2005 to 2009, 4,255 blood cultures were obtained from 4,122 infants in outpatient clinics, the ED, or in an inpatient setting within 24 hours of birth. Preterm infants <37 weeks, infants with underlying medical conditions, and infants with cultures drawn within three days of an original culture were excluded.
A total of 8% of the blood cultures were positive, with 2.2% deemed true positives and 5.8% due to contaminants. The incidence rate of true bacteremia was 0.57 per 1,000 full-term births, with gram-negative organisms (predominantly Escherichia coli) representing the majority (63%) of pathogens, followed by GBS (21%), Staphylococcus aureus (8%), and Streptococcus pneumoniae (3%). There were no cases of Listeria monocytogenes or Neisseria meningitidis bacteremia, and there was one case of enterococcal bacteremia. Fever was absent in 7% of cases.
The authors conclude that ampicillin may no longer be necessary for empiric antibiotic coverage in this age group given that 36% of pathogens were resistant to ampicillin, there were no cases of Listeria, and there was only one case of enterococcus. However, these recommendations should be considered in light of the specific study setting, and might not be applicable to all areas.
Bottom line: E. coli, GBS, and S. aureus, in that order, are the most common causes of bacteremia in infants aged one week to three months.
Citation: Greenhow TL, Hung YY, Herz AM. Changing epidemiology of bacteremia in infants aged 1 week to 3 months. Pediatrics. 2012;129(3):e590-e596.
Reviewed by Pediatric Editor Mark Shen, MD, SFHM, medical director of hospital medicine at Dell Children's Medical Center, Austin, Texas.
Clinical question: What is the epidemiology of bacteremia in one-week to three-month-old infants?
Background: Large studies of bacteremia in infants <90 days of age were largely performed before the era of routine prenatal screening and prophylaxis for Group B Streptococcus (GBS). Additionally, these studies have focused on febrile infants, which might not allow for characterization of the incidence of bacteremia when nonfebrile infants are considered.
Study design: Retrospective review.
Setting: Large HMO database.
Synopsis: Of 160,818 full-term infants born at Kaiser Permanente Northern California from 2005 to 2009, 4,255 blood cultures were obtained from 4,122 infants in outpatient clinics, the ED, or in an inpatient setting within 24 hours of birth. Preterm infants <37 weeks, infants with underlying medical conditions, and infants with cultures drawn within three days of an original culture were excluded.
A total of 8% of the blood cultures were positive, with 2.2% deemed true positives and 5.8% due to contaminants. The incidence rate of true bacteremia was 0.57 per 1,000 full-term births, with gram-negative organisms (predominantly Escherichia coli) representing the majority (63%) of pathogens, followed by GBS (21%), Staphylococcus aureus (8%), and Streptococcus pneumoniae (3%). There were no cases of Listeria monocytogenes or Neisseria meningitidis bacteremia, and there was one case of enterococcal bacteremia. Fever was absent in 7% of cases.
The authors conclude that ampicillin may no longer be necessary for empiric antibiotic coverage in this age group given that 36% of pathogens were resistant to ampicillin, there were no cases of Listeria, and there was only one case of enterococcus. However, these recommendations should be considered in light of the specific study setting, and might not be applicable to all areas.
Bottom line: E. coli, GBS, and S. aureus, in that order, are the most common causes of bacteremia in infants aged one week to three months.
Citation: Greenhow TL, Hung YY, Herz AM. Changing epidemiology of bacteremia in infants aged 1 week to 3 months. Pediatrics. 2012;129(3):e590-e596.
Reviewed by Pediatric Editor Mark Shen, MD, SFHM, medical director of hospital medicine at Dell Children's Medical Center, Austin, Texas.
Clinical question: What is the epidemiology of bacteremia in one-week to three-month-old infants?
Background: Large studies of bacteremia in infants <90 days of age were largely performed before the era of routine prenatal screening and prophylaxis for Group B Streptococcus (GBS). Additionally, these studies have focused on febrile infants, which might not allow for characterization of the incidence of bacteremia when nonfebrile infants are considered.
Study design: Retrospective review.
Setting: Large HMO database.
Synopsis: Of 160,818 full-term infants born at Kaiser Permanente Northern California from 2005 to 2009, 4,255 blood cultures were obtained from 4,122 infants in outpatient clinics, the ED, or in an inpatient setting within 24 hours of birth. Preterm infants <37 weeks, infants with underlying medical conditions, and infants with cultures drawn within three days of an original culture were excluded.
A total of 8% of the blood cultures were positive, with 2.2% deemed true positives and 5.8% due to contaminants. The incidence rate of true bacteremia was 0.57 per 1,000 full-term births, with gram-negative organisms (predominantly Escherichia coli) representing the majority (63%) of pathogens, followed by GBS (21%), Staphylococcus aureus (8%), and Streptococcus pneumoniae (3%). There were no cases of Listeria monocytogenes or Neisseria meningitidis bacteremia, and there was one case of enterococcal bacteremia. Fever was absent in 7% of cases.
The authors conclude that ampicillin may no longer be necessary for empiric antibiotic coverage in this age group given that 36% of pathogens were resistant to ampicillin, there were no cases of Listeria, and there was only one case of enterococcus. However, these recommendations should be considered in light of the specific study setting, and might not be applicable to all areas.
Bottom line: E. coli, GBS, and S. aureus, in that order, are the most common causes of bacteremia in infants aged one week to three months.
Citation: Greenhow TL, Hung YY, Herz AM. Changing epidemiology of bacteremia in infants aged 1 week to 3 months. Pediatrics. 2012;129(3):e590-e596.
Reviewed by Pediatric Editor Mark Shen, MD, SFHM, medical director of hospital medicine at Dell Children's Medical Center, Austin, Texas.
What Is the Best E&M of Fat Embolism Syndrome?
The Case
A 24-year-old white man with no past medical history is admitted after sustaining bilateral, closed femur fractures in a motor vehicle accident. Within hours of the trauma, he is taken to the operating room for open reduction and internal fixation. Of note, preoperatively, his hematocrit is 40%. After surgery, he is easily extubated and transferred to an unmonitored bed for further care. Approximately 30 hours after admission, he develops tachypnea with a respiratory rate of 35 breaths per minute and hypoxia with an oxygen saturation of 86% on room air. He is tachycardic (120 beats per minute) and febrile to 39.0oC. His blood pressure remains stable. He is somnolent, and when awake, he is confused. Notably, his hematocrit is now 22%. An electrocardiogram shows sinus tachycardia, an initial chest X-ray is normal, and a high-resolution CT scan is negative for a pulmonary embolism (PE).
Is this clinical picture consistent with fat embolism syndrome and, if so, how should he be managed?
Overview
“Fat embolism” refers to the presence of fat globules that obstruct the lung parenchyma and peripheral circulation. Fat embolism syndrome, on the other hand, is a more serious manifestation involving multiple organ systems. Specifically, it is a clinical diagnosis presenting with the classic triad of hypoxemia, neurologic abnormalities, and a petechial rash.
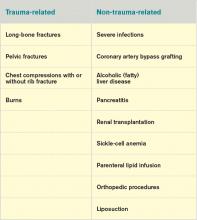
Fat embolism syndrome is usually associated with multiple traumas, including long-bone injuries and pelvic fractures. It is more frequently associated with closed fractures than open fractures, possibly due to the higher pressures associated with closed fractures. This syndrome has been less commonly associated with a variety of nontraumatic conditions (Table 1).
With an increased incidence of long-bone fractures in the younger demographic, fat embolism syndrome is most common in the second or third decade of life. While fat embolism occurs in up to 90% of patients with traumatic skeletal injuries, fat embolism syndrome occurs in 0.5% to 10% of patients following trauma, with a higher incidence in multiple fractures (5% to 10%) than in single long-bone fractures (0.5% to 2%).1-3
With the increasing role of hospitalists in assisting in the management of orthopedic patients, their knowledge of fat embolism syndrome is important so that it can be included in the differential diagnosis of acute respiratory failure in these orthopedic patients.
Review of the Data
Pathogenesis. Clinical manifestations of fat embolism syndrome have been acknowledged for more than 100 years. Since its first description in the 1860s, there has been speculation about the etiology of this condition. In the 1920s, two theories were proposed to explain the origin of the fat droplets: the mechanical and biochemical theories.2,4
Mechanical theory suggests that trauma to long bones disturbs fat cells within the bone marrow or adipose tissue, causing fat globules to mobilize.2,3 There is a rise in marrow pressure above venous pressure, which allows fat particles to enter the circulation through damaged venules surrounding the fracture site. Once lodged in the pulmonary microvasculature, embolized fat causes local ischemia and inflammation. Fat globules may pass into the arterial circulation either by paradoxical embolism through a patent foramen ovale, or by microemboli that pass through the lungs into the arterial circulation. This explains embolization to other organs, including the brain, retina, and skin.
Alternatively, biochemical theory hypothesizes that fat embolism syndrome is contingent on the production of toxic intermediaries from the breakdown of embolized fat.2,3 This theory suggests that the release of catecholamines after severe trauma can liberate free fatty acids from fat stores, or that acute-phase reactants at the trauma site affect fat solubility, causing agglutination and embolization. This theory helps to explain nontraumatic fat embolism syndrome, as well as the delay in development of the clinical syndrome after acute injury.
Clinical presentation. Most patients have a latent period after trauma of 12 to 72 hours before symptoms of fat embolism syndrome become apparent; however, clinical manifestations might occur immediately or up to one to two weeks following injury.2,4 As previously mentioned, the classic triad of symptoms includes respiratory compromise, neurological impairment, and a petechial rash.
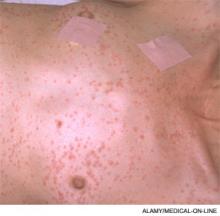
The most common and usually earliest manifestation is acute hypoxia, which must be distinguished from other treatable causes of hypoxia, including pneumothorax, hemothorax, PE, and pneumonia. Pulmonary changes might progress to respiratory failure similar to acute respiratory distress syndrome. Neurological manifestations are primarily nonspecific and include headache, irritability, delirium, seizures, and coma. Focal neurological deficits are rare but have been described.5 Almost all neurological symptoms are fully reversible. The petechial rash is distinctive and occurs on the chest, axilla, and subconjunctiva. Although the rash occurs in only 20% to 50% of patients and resolves fairly quickly, in the appropriate clinical setting, this rash is considered pathognomonic.1,2,4
A variety of other nonspecific signs and symptoms might also occur: pyrexia, tachycardia, fat in the urine or sputum, retinal changes, renal insufficiency, myocardial dysfunction, and an otherwise unexplained drop in hematocrit or platelet count.
Diagnosis. Fat embolism syndrome is a clinical diagnosis and a diagnosis of exclusion. There are no specific confirmatory tests. An arterial blood gas will usually reveal a PaO2 of <60 mmHg.3 Laboratory evaluation might also show fat globules in the urine or sputum on Sudan or Oil Red O staining, but these findings are nonspecific.3,4 Bronchoscopy with bronchial alveolar lavage (BAL) might similarly detect fat droplets in alveolar macrophages in the BAL fluid; however, the sensitivity and specificity for diagnosis of fat embolism syndrome are unknown.4 None of these tests can be used solely for the diagnosis of fat embolism syndrome.
Thrombocytopenia and anemia out of proportion to the expected drop from surgery are not uncommon in addition to other nonspecific laboratory findings, including hypocalcemia, elevated serum lipase level, and elevated erythrocyte sedimentation rate.4 Several radiological findings have been observed on lung and brain imaging, though the findings are nonspecific and none are diagnostic. A chest X-ray might be normal, but abnormalities are seen in 30% to 50% of cases.2 Typically, when abnormal, the chest X-ray shows diffuse interstitial and alveolar densities, as well as patchy perihilar and basilar infiltrates resembling pulmonary edema. These X-ray findings might not be seen for up to 12 to 24 hours following the onset of clinical symptoms.
The most commonly used diagnostic criteria for the diagnosis of fat embolism syndrome are published by Gurd et al.6 At least two major criteria or one major criterion and four minor criteria are required for the diagnosis of fat embolism syndrome. The major criteria are based on the three classic signs and symptoms of fat embolism syndrome; the minor criteria include the finding of fat globules in the urine and sputum as well as some of the previously mentioned nonspecific clinical signs and laboratory tests.
Other criteria for diagnosis have been suggested, including those published by Lindeque et al, which focuses primarily on the respiratory characteristics, and a more recent set of semiquantitative diagnostic criteria called the fat embolism index, published by Schonfeld et al.7,8 Schonfeld’s scoring index accounts for the major signs and symptoms of fat embolism syndrome and weighs them according to relative specificity. A score of 5 or more is required for diagnosis of fat embolism syndrome. Table 2 compares the three sets of criteria used for diagnosis of fat embolism syndrome.
Treatment. The treatment of fat embolism syndrome is supportive. Most often, this requires supplemental oxygen for hypoxia and, possibly, fluid resuscitation in the case of hypovolemia. Occasionally, though, these relatively minor supportive therapies need to be escalated to bipap or even full ventilatory support and vasopressors in the more severe cases.
Based on the premise that steroids will attenuate the inflammatory reaction to free fatty acids within the lung, steroids have been tried in the treatment of fat embolism syndrome. However, there are no studies that clearly show benefit with their use.
Prevention. Most of the methods of prevention involve surgical intervention rather than medical therapy. Because microscopic fat emboli are showered during manipulation of long-bone fragments, early immobilization of fractures is recommended, and operative correction rather than conservative management is the preferred method.2,3 One report estimates a 70% reduction in pulmonary complications from this intervention alone.9
Further, two surgical techniques are debated as possible means of preventing fat embolism syndrome. The first is “venting,” in which a hole is made distal to the site of intramedullary nail placement. This reduces intramedullary pressure elevation and, therefore, extravasation of fat into the circulation.10 The second technique is the use of a reamer, irrigator, aspirator (RIA) device. A reamer is a tool used to create an accurate-sized hole for an intramedullary nail. Reaming before intramedullary nail placement can release fat deposits into the circulation. The RIA device irrigates and aspirates resident fat deposits as it reams the canal, releasing fewer deposits into the circulation.11 At this time, these two techniques are considered but not used routinely by surgeons.
Corticosteroids remain a debated method of prevention of fat embolism syndrome. A number of smaller studies suggest steroid therapy might reduce the incidence of fat embolism syndrome and hypoxia; a 2009 meta-analysis pooling nearly 400 patients from these smaller studies found such results.12 Unfortunately, the included studies were noted to be of poor quality, and no change in mortality was found. These results, combined with the possibility of poor wound healing or infection as a complication of steroid use, keep steroids from being used routinely to prevent fat embolism syndrome.
Clinical course. The severity of fat embolism syndrome ranges from mild transient hypoxia with confusion to progressively worsening symptoms leading to acute respiratory distress syndrome and coma. Bulger et al found a 7% mortality rate in this population.1 Less commonly, patients have a fulminant presentation with symptom onset less than 12 hours after injury. With this presentation, patients have a higher rate of mortality—as high as 15%.13
Back to the Case
This young man with bilateral long-bone fractures was at high risk of developing fat embolism syndrome. As is recommended, he was quickly taken to the operating room for fracture stabilization with open reduction and internal fixation. In addition, a RIA device was used to decrease intramedullary pressure. Nonetheless, within the first two days of injury, he developed hypoxia and confusion. These clinical changes were associated with an unexpected drop in hematocrit.
Chest X-ray and high-resolution computed tomography did not reveal a cause of his hypoxia. Similarly, laboratory evaluation for a reversible cause of encephalopathy was negative. A Sudan stain of his urine revealed free fat globules. Though he did not develop axillary petechiae, this clinical picture is consistent with fat embolism syndrome based on Gurd’s criteria. He was supported with oxygen therapy, and he stabilized without further complications.
Drs. Smith and Rice are members of the Section of Hospital Medicine at Vanderbilt University in Nashville, Tenn.
References
- Bulger EM, Smith DG, Maier RV, Jurkovich GJ. Fat embolism syndrome. A 10-year review. Arch Surg. 1997;132:435-439.
- Levy D. The fat embolism syndrome. Clin Orthop. 1990;261:281-286.
- Akhtar S. Fat embolism. Anes Clin. 2009;27:533-550.
- Gupta A, Reilly C. Fat embolism. Anaesth Crit Care Pain. 2007;7:148-151.
- Thomas JE, Ayyar DR. Systemic fat embolism. Arch Neurol. 1972;26:517-523.
- Gurd AR, Wilson RI. The fat embolism syndrome. J Bone Joint Surg Br. 1974;56B:408-416.
- Lindeque BG, Schoeman HS, Dommisse GF, Boeyens MC, Vlok AL. Fat embolism and the fat embolism syndrome. A double-blind therapeutic study. J Bone Joint Surg Br. 1987;69:128-131.
- Schonfeld SA, Ploysongsang Y, DiLisio R, et al. Fat embolism prophylaxis with corticosteroids. A prospective study in high-risk patients. Ann Intern Med. 1983;99:438-443.
- Robinson CM. Current concepts of respiratory insufficiency syndromes after fracture. J Bone Joint Surg Br. 2001;83:781-791.
- Kim YH, Oh SW, Kim JS. Prevalence of fat embolism following bilateral simultaneous and unilateral total hip arthroplasty performed with or without cement: a prospective, randomized clinical study. J Bone Joint Surg Am. 2002;84A:1372-1379.
- Volgas DA, Burch T, Stannard JP, Ellis T, Bilotta J, Alonso JE. Fat embolus in femur fractures: a comparison of two reaming systems. Injury. 2010;41 Suppl 2:S90-S93.
- Bederman SS, Bhandari M, McKee MD, Schemitsch EH. Do corticosteroids reduce the risk of fat embolism syndrome in patients with long-bone fractures? A meta-analysis. Can J Surg. 2009;52:386-393.
- Bracco D, Favre JB, Joris F, Ravussin A. Fatal fat embolism syndrome: a case report. J Neurosurg Anesthesiol. 2000;12:221-224.
The Case
A 24-year-old white man with no past medical history is admitted after sustaining bilateral, closed femur fractures in a motor vehicle accident. Within hours of the trauma, he is taken to the operating room for open reduction and internal fixation. Of note, preoperatively, his hematocrit is 40%. After surgery, he is easily extubated and transferred to an unmonitored bed for further care. Approximately 30 hours after admission, he develops tachypnea with a respiratory rate of 35 breaths per minute and hypoxia with an oxygen saturation of 86% on room air. He is tachycardic (120 beats per minute) and febrile to 39.0oC. His blood pressure remains stable. He is somnolent, and when awake, he is confused. Notably, his hematocrit is now 22%. An electrocardiogram shows sinus tachycardia, an initial chest X-ray is normal, and a high-resolution CT scan is negative for a pulmonary embolism (PE).
Is this clinical picture consistent with fat embolism syndrome and, if so, how should he be managed?
Overview
“Fat embolism” refers to the presence of fat globules that obstruct the lung parenchyma and peripheral circulation. Fat embolism syndrome, on the other hand, is a more serious manifestation involving multiple organ systems. Specifically, it is a clinical diagnosis presenting with the classic triad of hypoxemia, neurologic abnormalities, and a petechial rash.
Fat embolism syndrome is usually associated with multiple traumas, including long-bone injuries and pelvic fractures. It is more frequently associated with closed fractures than open fractures, possibly due to the higher pressures associated with closed fractures. This syndrome has been less commonly associated with a variety of nontraumatic conditions (Table 1).
With an increased incidence of long-bone fractures in the younger demographic, fat embolism syndrome is most common in the second or third decade of life. While fat embolism occurs in up to 90% of patients with traumatic skeletal injuries, fat embolism syndrome occurs in 0.5% to 10% of patients following trauma, with a higher incidence in multiple fractures (5% to 10%) than in single long-bone fractures (0.5% to 2%).1-3
With the increasing role of hospitalists in assisting in the management of orthopedic patients, their knowledge of fat embolism syndrome is important so that it can be included in the differential diagnosis of acute respiratory failure in these orthopedic patients.
Review of the Data
Pathogenesis. Clinical manifestations of fat embolism syndrome have been acknowledged for more than 100 years. Since its first description in the 1860s, there has been speculation about the etiology of this condition. In the 1920s, two theories were proposed to explain the origin of the fat droplets: the mechanical and biochemical theories.2,4
Mechanical theory suggests that trauma to long bones disturbs fat cells within the bone marrow or adipose tissue, causing fat globules to mobilize.2,3 There is a rise in marrow pressure above venous pressure, which allows fat particles to enter the circulation through damaged venules surrounding the fracture site. Once lodged in the pulmonary microvasculature, embolized fat causes local ischemia and inflammation. Fat globules may pass into the arterial circulation either by paradoxical embolism through a patent foramen ovale, or by microemboli that pass through the lungs into the arterial circulation. This explains embolization to other organs, including the brain, retina, and skin.
Alternatively, biochemical theory hypothesizes that fat embolism syndrome is contingent on the production of toxic intermediaries from the breakdown of embolized fat.2,3 This theory suggests that the release of catecholamines after severe trauma can liberate free fatty acids from fat stores, or that acute-phase reactants at the trauma site affect fat solubility, causing agglutination and embolization. This theory helps to explain nontraumatic fat embolism syndrome, as well as the delay in development of the clinical syndrome after acute injury.
Clinical presentation. Most patients have a latent period after trauma of 12 to 72 hours before symptoms of fat embolism syndrome become apparent; however, clinical manifestations might occur immediately or up to one to two weeks following injury.2,4 As previously mentioned, the classic triad of symptoms includes respiratory compromise, neurological impairment, and a petechial rash.
The most common and usually earliest manifestation is acute hypoxia, which must be distinguished from other treatable causes of hypoxia, including pneumothorax, hemothorax, PE, and pneumonia. Pulmonary changes might progress to respiratory failure similar to acute respiratory distress syndrome. Neurological manifestations are primarily nonspecific and include headache, irritability, delirium, seizures, and coma. Focal neurological deficits are rare but have been described.5 Almost all neurological symptoms are fully reversible. The petechial rash is distinctive and occurs on the chest, axilla, and subconjunctiva. Although the rash occurs in only 20% to 50% of patients and resolves fairly quickly, in the appropriate clinical setting, this rash is considered pathognomonic.1,2,4
A variety of other nonspecific signs and symptoms might also occur: pyrexia, tachycardia, fat in the urine or sputum, retinal changes, renal insufficiency, myocardial dysfunction, and an otherwise unexplained drop in hematocrit or platelet count.
Diagnosis. Fat embolism syndrome is a clinical diagnosis and a diagnosis of exclusion. There are no specific confirmatory tests. An arterial blood gas will usually reveal a PaO2 of <60 mmHg.3 Laboratory evaluation might also show fat globules in the urine or sputum on Sudan or Oil Red O staining, but these findings are nonspecific.3,4 Bronchoscopy with bronchial alveolar lavage (BAL) might similarly detect fat droplets in alveolar macrophages in the BAL fluid; however, the sensitivity and specificity for diagnosis of fat embolism syndrome are unknown.4 None of these tests can be used solely for the diagnosis of fat embolism syndrome.
Thrombocytopenia and anemia out of proportion to the expected drop from surgery are not uncommon in addition to other nonspecific laboratory findings, including hypocalcemia, elevated serum lipase level, and elevated erythrocyte sedimentation rate.4 Several radiological findings have been observed on lung and brain imaging, though the findings are nonspecific and none are diagnostic. A chest X-ray might be normal, but abnormalities are seen in 30% to 50% of cases.2 Typically, when abnormal, the chest X-ray shows diffuse interstitial and alveolar densities, as well as patchy perihilar and basilar infiltrates resembling pulmonary edema. These X-ray findings might not be seen for up to 12 to 24 hours following the onset of clinical symptoms.
The most commonly used diagnostic criteria for the diagnosis of fat embolism syndrome are published by Gurd et al.6 At least two major criteria or one major criterion and four minor criteria are required for the diagnosis of fat embolism syndrome. The major criteria are based on the three classic signs and symptoms of fat embolism syndrome; the minor criteria include the finding of fat globules in the urine and sputum as well as some of the previously mentioned nonspecific clinical signs and laboratory tests.
Other criteria for diagnosis have been suggested, including those published by Lindeque et al, which focuses primarily on the respiratory characteristics, and a more recent set of semiquantitative diagnostic criteria called the fat embolism index, published by Schonfeld et al.7,8 Schonfeld’s scoring index accounts for the major signs and symptoms of fat embolism syndrome and weighs them according to relative specificity. A score of 5 or more is required for diagnosis of fat embolism syndrome. Table 2 compares the three sets of criteria used for diagnosis of fat embolism syndrome.
Treatment. The treatment of fat embolism syndrome is supportive. Most often, this requires supplemental oxygen for hypoxia and, possibly, fluid resuscitation in the case of hypovolemia. Occasionally, though, these relatively minor supportive therapies need to be escalated to bipap or even full ventilatory support and vasopressors in the more severe cases.
Based on the premise that steroids will attenuate the inflammatory reaction to free fatty acids within the lung, steroids have been tried in the treatment of fat embolism syndrome. However, there are no studies that clearly show benefit with their use.
Prevention. Most of the methods of prevention involve surgical intervention rather than medical therapy. Because microscopic fat emboli are showered during manipulation of long-bone fragments, early immobilization of fractures is recommended, and operative correction rather than conservative management is the preferred method.2,3 One report estimates a 70% reduction in pulmonary complications from this intervention alone.9
Further, two surgical techniques are debated as possible means of preventing fat embolism syndrome. The first is “venting,” in which a hole is made distal to the site of intramedullary nail placement. This reduces intramedullary pressure elevation and, therefore, extravasation of fat into the circulation.10 The second technique is the use of a reamer, irrigator, aspirator (RIA) device. A reamer is a tool used to create an accurate-sized hole for an intramedullary nail. Reaming before intramedullary nail placement can release fat deposits into the circulation. The RIA device irrigates and aspirates resident fat deposits as it reams the canal, releasing fewer deposits into the circulation.11 At this time, these two techniques are considered but not used routinely by surgeons.
Corticosteroids remain a debated method of prevention of fat embolism syndrome. A number of smaller studies suggest steroid therapy might reduce the incidence of fat embolism syndrome and hypoxia; a 2009 meta-analysis pooling nearly 400 patients from these smaller studies found such results.12 Unfortunately, the included studies were noted to be of poor quality, and no change in mortality was found. These results, combined with the possibility of poor wound healing or infection as a complication of steroid use, keep steroids from being used routinely to prevent fat embolism syndrome.
Clinical course. The severity of fat embolism syndrome ranges from mild transient hypoxia with confusion to progressively worsening symptoms leading to acute respiratory distress syndrome and coma. Bulger et al found a 7% mortality rate in this population.1 Less commonly, patients have a fulminant presentation with symptom onset less than 12 hours after injury. With this presentation, patients have a higher rate of mortality—as high as 15%.13
Back to the Case
This young man with bilateral long-bone fractures was at high risk of developing fat embolism syndrome. As is recommended, he was quickly taken to the operating room for fracture stabilization with open reduction and internal fixation. In addition, a RIA device was used to decrease intramedullary pressure. Nonetheless, within the first two days of injury, he developed hypoxia and confusion. These clinical changes were associated with an unexpected drop in hematocrit.
Chest X-ray and high-resolution computed tomography did not reveal a cause of his hypoxia. Similarly, laboratory evaluation for a reversible cause of encephalopathy was negative. A Sudan stain of his urine revealed free fat globules. Though he did not develop axillary petechiae, this clinical picture is consistent with fat embolism syndrome based on Gurd’s criteria. He was supported with oxygen therapy, and he stabilized without further complications.
Drs. Smith and Rice are members of the Section of Hospital Medicine at Vanderbilt University in Nashville, Tenn.
References
- Bulger EM, Smith DG, Maier RV, Jurkovich GJ. Fat embolism syndrome. A 10-year review. Arch Surg. 1997;132:435-439.
- Levy D. The fat embolism syndrome. Clin Orthop. 1990;261:281-286.
- Akhtar S. Fat embolism. Anes Clin. 2009;27:533-550.
- Gupta A, Reilly C. Fat embolism. Anaesth Crit Care Pain. 2007;7:148-151.
- Thomas JE, Ayyar DR. Systemic fat embolism. Arch Neurol. 1972;26:517-523.
- Gurd AR, Wilson RI. The fat embolism syndrome. J Bone Joint Surg Br. 1974;56B:408-416.
- Lindeque BG, Schoeman HS, Dommisse GF, Boeyens MC, Vlok AL. Fat embolism and the fat embolism syndrome. A double-blind therapeutic study. J Bone Joint Surg Br. 1987;69:128-131.
- Schonfeld SA, Ploysongsang Y, DiLisio R, et al. Fat embolism prophylaxis with corticosteroids. A prospective study in high-risk patients. Ann Intern Med. 1983;99:438-443.
- Robinson CM. Current concepts of respiratory insufficiency syndromes after fracture. J Bone Joint Surg Br. 2001;83:781-791.
- Kim YH, Oh SW, Kim JS. Prevalence of fat embolism following bilateral simultaneous and unilateral total hip arthroplasty performed with or without cement: a prospective, randomized clinical study. J Bone Joint Surg Am. 2002;84A:1372-1379.
- Volgas DA, Burch T, Stannard JP, Ellis T, Bilotta J, Alonso JE. Fat embolus in femur fractures: a comparison of two reaming systems. Injury. 2010;41 Suppl 2:S90-S93.
- Bederman SS, Bhandari M, McKee MD, Schemitsch EH. Do corticosteroids reduce the risk of fat embolism syndrome in patients with long-bone fractures? A meta-analysis. Can J Surg. 2009;52:386-393.
- Bracco D, Favre JB, Joris F, Ravussin A. Fatal fat embolism syndrome: a case report. J Neurosurg Anesthesiol. 2000;12:221-224.
The Case
A 24-year-old white man with no past medical history is admitted after sustaining bilateral, closed femur fractures in a motor vehicle accident. Within hours of the trauma, he is taken to the operating room for open reduction and internal fixation. Of note, preoperatively, his hematocrit is 40%. After surgery, he is easily extubated and transferred to an unmonitored bed for further care. Approximately 30 hours after admission, he develops tachypnea with a respiratory rate of 35 breaths per minute and hypoxia with an oxygen saturation of 86% on room air. He is tachycardic (120 beats per minute) and febrile to 39.0oC. His blood pressure remains stable. He is somnolent, and when awake, he is confused. Notably, his hematocrit is now 22%. An electrocardiogram shows sinus tachycardia, an initial chest X-ray is normal, and a high-resolution CT scan is negative for a pulmonary embolism (PE).
Is this clinical picture consistent with fat embolism syndrome and, if so, how should he be managed?
Overview
“Fat embolism” refers to the presence of fat globules that obstruct the lung parenchyma and peripheral circulation. Fat embolism syndrome, on the other hand, is a more serious manifestation involving multiple organ systems. Specifically, it is a clinical diagnosis presenting with the classic triad of hypoxemia, neurologic abnormalities, and a petechial rash.
Fat embolism syndrome is usually associated with multiple traumas, including long-bone injuries and pelvic fractures. It is more frequently associated with closed fractures than open fractures, possibly due to the higher pressures associated with closed fractures. This syndrome has been less commonly associated with a variety of nontraumatic conditions (Table 1).
With an increased incidence of long-bone fractures in the younger demographic, fat embolism syndrome is most common in the second or third decade of life. While fat embolism occurs in up to 90% of patients with traumatic skeletal injuries, fat embolism syndrome occurs in 0.5% to 10% of patients following trauma, with a higher incidence in multiple fractures (5% to 10%) than in single long-bone fractures (0.5% to 2%).1-3
With the increasing role of hospitalists in assisting in the management of orthopedic patients, their knowledge of fat embolism syndrome is important so that it can be included in the differential diagnosis of acute respiratory failure in these orthopedic patients.
Review of the Data
Pathogenesis. Clinical manifestations of fat embolism syndrome have been acknowledged for more than 100 years. Since its first description in the 1860s, there has been speculation about the etiology of this condition. In the 1920s, two theories were proposed to explain the origin of the fat droplets: the mechanical and biochemical theories.2,4
Mechanical theory suggests that trauma to long bones disturbs fat cells within the bone marrow or adipose tissue, causing fat globules to mobilize.2,3 There is a rise in marrow pressure above venous pressure, which allows fat particles to enter the circulation through damaged venules surrounding the fracture site. Once lodged in the pulmonary microvasculature, embolized fat causes local ischemia and inflammation. Fat globules may pass into the arterial circulation either by paradoxical embolism through a patent foramen ovale, or by microemboli that pass through the lungs into the arterial circulation. This explains embolization to other organs, including the brain, retina, and skin.
Alternatively, biochemical theory hypothesizes that fat embolism syndrome is contingent on the production of toxic intermediaries from the breakdown of embolized fat.2,3 This theory suggests that the release of catecholamines after severe trauma can liberate free fatty acids from fat stores, or that acute-phase reactants at the trauma site affect fat solubility, causing agglutination and embolization. This theory helps to explain nontraumatic fat embolism syndrome, as well as the delay in development of the clinical syndrome after acute injury.
Clinical presentation. Most patients have a latent period after trauma of 12 to 72 hours before symptoms of fat embolism syndrome become apparent; however, clinical manifestations might occur immediately or up to one to two weeks following injury.2,4 As previously mentioned, the classic triad of symptoms includes respiratory compromise, neurological impairment, and a petechial rash.
The most common and usually earliest manifestation is acute hypoxia, which must be distinguished from other treatable causes of hypoxia, including pneumothorax, hemothorax, PE, and pneumonia. Pulmonary changes might progress to respiratory failure similar to acute respiratory distress syndrome. Neurological manifestations are primarily nonspecific and include headache, irritability, delirium, seizures, and coma. Focal neurological deficits are rare but have been described.5 Almost all neurological symptoms are fully reversible. The petechial rash is distinctive and occurs on the chest, axilla, and subconjunctiva. Although the rash occurs in only 20% to 50% of patients and resolves fairly quickly, in the appropriate clinical setting, this rash is considered pathognomonic.1,2,4
A variety of other nonspecific signs and symptoms might also occur: pyrexia, tachycardia, fat in the urine or sputum, retinal changes, renal insufficiency, myocardial dysfunction, and an otherwise unexplained drop in hematocrit or platelet count.
Diagnosis. Fat embolism syndrome is a clinical diagnosis and a diagnosis of exclusion. There are no specific confirmatory tests. An arterial blood gas will usually reveal a PaO2 of <60 mmHg.3 Laboratory evaluation might also show fat globules in the urine or sputum on Sudan or Oil Red O staining, but these findings are nonspecific.3,4 Bronchoscopy with bronchial alveolar lavage (BAL) might similarly detect fat droplets in alveolar macrophages in the BAL fluid; however, the sensitivity and specificity for diagnosis of fat embolism syndrome are unknown.4 None of these tests can be used solely for the diagnosis of fat embolism syndrome.
Thrombocytopenia and anemia out of proportion to the expected drop from surgery are not uncommon in addition to other nonspecific laboratory findings, including hypocalcemia, elevated serum lipase level, and elevated erythrocyte sedimentation rate.4 Several radiological findings have been observed on lung and brain imaging, though the findings are nonspecific and none are diagnostic. A chest X-ray might be normal, but abnormalities are seen in 30% to 50% of cases.2 Typically, when abnormal, the chest X-ray shows diffuse interstitial and alveolar densities, as well as patchy perihilar and basilar infiltrates resembling pulmonary edema. These X-ray findings might not be seen for up to 12 to 24 hours following the onset of clinical symptoms.
The most commonly used diagnostic criteria for the diagnosis of fat embolism syndrome are published by Gurd et al.6 At least two major criteria or one major criterion and four minor criteria are required for the diagnosis of fat embolism syndrome. The major criteria are based on the three classic signs and symptoms of fat embolism syndrome; the minor criteria include the finding of fat globules in the urine and sputum as well as some of the previously mentioned nonspecific clinical signs and laboratory tests.
Other criteria for diagnosis have been suggested, including those published by Lindeque et al, which focuses primarily on the respiratory characteristics, and a more recent set of semiquantitative diagnostic criteria called the fat embolism index, published by Schonfeld et al.7,8 Schonfeld’s scoring index accounts for the major signs and symptoms of fat embolism syndrome and weighs them according to relative specificity. A score of 5 or more is required for diagnosis of fat embolism syndrome. Table 2 compares the three sets of criteria used for diagnosis of fat embolism syndrome.
Treatment. The treatment of fat embolism syndrome is supportive. Most often, this requires supplemental oxygen for hypoxia and, possibly, fluid resuscitation in the case of hypovolemia. Occasionally, though, these relatively minor supportive therapies need to be escalated to bipap or even full ventilatory support and vasopressors in the more severe cases.
Based on the premise that steroids will attenuate the inflammatory reaction to free fatty acids within the lung, steroids have been tried in the treatment of fat embolism syndrome. However, there are no studies that clearly show benefit with their use.
Prevention. Most of the methods of prevention involve surgical intervention rather than medical therapy. Because microscopic fat emboli are showered during manipulation of long-bone fragments, early immobilization of fractures is recommended, and operative correction rather than conservative management is the preferred method.2,3 One report estimates a 70% reduction in pulmonary complications from this intervention alone.9
Further, two surgical techniques are debated as possible means of preventing fat embolism syndrome. The first is “venting,” in which a hole is made distal to the site of intramedullary nail placement. This reduces intramedullary pressure elevation and, therefore, extravasation of fat into the circulation.10 The second technique is the use of a reamer, irrigator, aspirator (RIA) device. A reamer is a tool used to create an accurate-sized hole for an intramedullary nail. Reaming before intramedullary nail placement can release fat deposits into the circulation. The RIA device irrigates and aspirates resident fat deposits as it reams the canal, releasing fewer deposits into the circulation.11 At this time, these two techniques are considered but not used routinely by surgeons.
Corticosteroids remain a debated method of prevention of fat embolism syndrome. A number of smaller studies suggest steroid therapy might reduce the incidence of fat embolism syndrome and hypoxia; a 2009 meta-analysis pooling nearly 400 patients from these smaller studies found such results.12 Unfortunately, the included studies were noted to be of poor quality, and no change in mortality was found. These results, combined with the possibility of poor wound healing or infection as a complication of steroid use, keep steroids from being used routinely to prevent fat embolism syndrome.
Clinical course. The severity of fat embolism syndrome ranges from mild transient hypoxia with confusion to progressively worsening symptoms leading to acute respiratory distress syndrome and coma. Bulger et al found a 7% mortality rate in this population.1 Less commonly, patients have a fulminant presentation with symptom onset less than 12 hours after injury. With this presentation, patients have a higher rate of mortality—as high as 15%.13
Back to the Case
This young man with bilateral long-bone fractures was at high risk of developing fat embolism syndrome. As is recommended, he was quickly taken to the operating room for fracture stabilization with open reduction and internal fixation. In addition, a RIA device was used to decrease intramedullary pressure. Nonetheless, within the first two days of injury, he developed hypoxia and confusion. These clinical changes were associated with an unexpected drop in hematocrit.
Chest X-ray and high-resolution computed tomography did not reveal a cause of his hypoxia. Similarly, laboratory evaluation for a reversible cause of encephalopathy was negative. A Sudan stain of his urine revealed free fat globules. Though he did not develop axillary petechiae, this clinical picture is consistent with fat embolism syndrome based on Gurd’s criteria. He was supported with oxygen therapy, and he stabilized without further complications.
Drs. Smith and Rice are members of the Section of Hospital Medicine at Vanderbilt University in Nashville, Tenn.
References
- Bulger EM, Smith DG, Maier RV, Jurkovich GJ. Fat embolism syndrome. A 10-year review. Arch Surg. 1997;132:435-439.
- Levy D. The fat embolism syndrome. Clin Orthop. 1990;261:281-286.
- Akhtar S. Fat embolism. Anes Clin. 2009;27:533-550.
- Gupta A, Reilly C. Fat embolism. Anaesth Crit Care Pain. 2007;7:148-151.
- Thomas JE, Ayyar DR. Systemic fat embolism. Arch Neurol. 1972;26:517-523.
- Gurd AR, Wilson RI. The fat embolism syndrome. J Bone Joint Surg Br. 1974;56B:408-416.
- Lindeque BG, Schoeman HS, Dommisse GF, Boeyens MC, Vlok AL. Fat embolism and the fat embolism syndrome. A double-blind therapeutic study. J Bone Joint Surg Br. 1987;69:128-131.
- Schonfeld SA, Ploysongsang Y, DiLisio R, et al. Fat embolism prophylaxis with corticosteroids. A prospective study in high-risk patients. Ann Intern Med. 1983;99:438-443.
- Robinson CM. Current concepts of respiratory insufficiency syndromes after fracture. J Bone Joint Surg Br. 2001;83:781-791.
- Kim YH, Oh SW, Kim JS. Prevalence of fat embolism following bilateral simultaneous and unilateral total hip arthroplasty performed with or without cement: a prospective, randomized clinical study. J Bone Joint Surg Am. 2002;84A:1372-1379.
- Volgas DA, Burch T, Stannard JP, Ellis T, Bilotta J, Alonso JE. Fat embolus in femur fractures: a comparison of two reaming systems. Injury. 2010;41 Suppl 2:S90-S93.
- Bederman SS, Bhandari M, McKee MD, Schemitsch EH. Do corticosteroids reduce the risk of fat embolism syndrome in patients with long-bone fractures? A meta-analysis. Can J Surg. 2009;52:386-393.
- Bracco D, Favre JB, Joris F, Ravussin A. Fatal fat embolism syndrome: a case report. J Neurosurg Anesthesiol. 2000;12:221-224.
Palliative-Care-Focused Hospitalist Appreciates Training the Next Generation
Chithra Perumalswami, MD, knew early on what she wanted to do with her life. As a teenager, she volunteered in an ED and with a hospice group, volunteerism that continued throughout her education. When she graduated from high school, she was tapped for Brown University’s Program in Liberal Medical Education, which calls itself the only baccalaureate-MD program in the Ivy League. And though she eventually turned down the offer, she pursued dual majors in cellular and molecular biology and English at the University of Michigan, where she earned her medical degree in 2004 and completed her residency.
In 2009, she participated in the Palliative Care Education and Practice Program at Harvard Medical School in Boston, a two-week post-graduate course aimed at professional development for physicians dedicated to careers in palliative-care education. “I really found that there were just so many aspects to caring for a patient as a palliative-care specialist and as a hospitalist that really strike at the heart of what being a doctor is,” says Dr. Perumalswami, assistant professor of medicine in the Division of Hospital Medicine at Northwestern Memorial Hospital in Chicago and one of four new members of Team Hospitalist. “I think it’s been an interest I’ve always had. During my residency training, I definitely experienced quite a few patient cases where I felt that we really needed to help patients and their families, and I didn’t necessarily have the best skill set to do that until I had more experience and more training.”
Dr. Perumalswami now wants to get better at her craft.
“As an academic hospitalist, it’s not just about doing research and writing papers and seeing papers,” she says, “but it’s also developing those leadership skills and helping that become an integral part of the educational experience.”
Question: What drew you to a career in HM?
Answer: I chose a career in academic hospital medicine primarily because I enjoy taking care of acutely ill, hospitalized, adult patients. I also really enjoy teaching medical students, residents, and fellows, and I enjoy doing that in the hospital setting. I think that there’s great satisfaction from taking care of a patient from admission to discharge.… I enjoyed every aspect of internal medicine, and when I graduated, I thought I could choose a subspecialty, but I felt that my skills and my expertise was really in taking care of the hospitalized patient.
Q: You have sought out extra training in palliative care and pain management. How has that impacted your career?
A: It’s not something that I necessarily started out thinking that I would specialize in, but the more I took care of hospitalized patients, the more I realized that we actually take care of a fair number of patients who have really complex symptom needs, and also really have a lot of needs with regard to recognizing when their prognosis is poor and understanding what their options are, if they’re even amenable to a palliative approach. I really felt that that was a skill that I needed to fine-tune. So I ended up gaining enough clinical experience and participating with hospice patients to the point where that’s really supplemented my hospitalist career, because what I found is that it’s made me a better hospitalist, and being a hospitalist has made me a better palliative-care doc.
Q: Working in academia, there’s no way to escape talk of the duty-hour rules recently put in place. What’s your view on the issue?
A: My view is that the work hours are here to stay. I think that there are some definite benefits that we’ve gained from having work hours. I’d say first and foremost of those gains is public trust. I think most physicians will tell you that they don’t want a physician who’s in the 36th hour of their day taking care of them when we know that studies actually can demonstrate that when you’ve been awake that long, that your cognitive abilities decline.
Q: But?
A: I think we have a lot of challenges, though, because a lot of things require creative solutions. And I think the first on that list is education, because that’s the first thing that I think has the potential to drop to the bottom of the list.
Q: In terms of HM’s growth, as you see residents coming through your program, how popular do you think the model is going to be with them moving forward?
A: I do, actually, because as an academic hospitalist, I’ve had several medical students and residents tell me, “Watching you, I think that I want to go into this field.” Or they’ll say, “What do you think about doing this for a year or two?” Or, “What do you think about subspecializing, and then being a hospitalist?” And my answer to all of them is it’s a dynamic specialty, and if you’re up for creating change and being a leader, it’s a good field, because we need people in a lot of different buckets, so to speak. We need people who have done other things in their career to contribute to our field.
Q: How do you prepare trainees for all the challenges coming down the pike?
A: A lot of the people who are doing work in medical education are starting to look to other fields to see if there are other models that we can adapt, or that we can somehow absorb into our practice. I think that there are some parts of our education which are not really formalized early on, but I think we have a lot to learn from organizational behavior circles, and systems that actually look at teams and leadership.
Q: What do the next five to 10 years hold for you?
A: All physician leaders have to stay somewhat in the clinical world. I think if you lose sight of that, you can’t be a very effective leader, or a very effective agent for change. Because part of my work is with palliative care, and I really feel that it’s affected my work as a hospitalist in a positive way, I don’t think I ever see myself leaving the clinical world completely. But I do see myself becoming, ideally, more involved with leadership and more involved with helping to train the next set of leaders.
Q: What do you see as SHM’s role specific to academic HM?
A: HM is changing the way healthcare is delivered in the U.S., and I think having an organization to represent us is vital to our success in other arenas of change—including healthcare policy and innovative care models. I see SHM as a large umbrella group, of which academic HM is one part. Academic hospitalists are increasingly involved in the education of future generations of physicians, and are uniquely poised for facilitating cascading leadership. The traditional, hierarchical model of attending-fellow-resident-medical student is shifting, and academic hospitalists are well-suited to study and explore this leadership structure and how it affects patient care, feedback, and mentoring.
Richard Quinn is a freelance writer based in New Jersey.
Chithra Perumalswami, MD, knew early on what she wanted to do with her life. As a teenager, she volunteered in an ED and with a hospice group, volunteerism that continued throughout her education. When she graduated from high school, she was tapped for Brown University’s Program in Liberal Medical Education, which calls itself the only baccalaureate-MD program in the Ivy League. And though she eventually turned down the offer, she pursued dual majors in cellular and molecular biology and English at the University of Michigan, where she earned her medical degree in 2004 and completed her residency.
In 2009, she participated in the Palliative Care Education and Practice Program at Harvard Medical School in Boston, a two-week post-graduate course aimed at professional development for physicians dedicated to careers in palliative-care education. “I really found that there were just so many aspects to caring for a patient as a palliative-care specialist and as a hospitalist that really strike at the heart of what being a doctor is,” says Dr. Perumalswami, assistant professor of medicine in the Division of Hospital Medicine at Northwestern Memorial Hospital in Chicago and one of four new members of Team Hospitalist. “I think it’s been an interest I’ve always had. During my residency training, I definitely experienced quite a few patient cases where I felt that we really needed to help patients and their families, and I didn’t necessarily have the best skill set to do that until I had more experience and more training.”
Dr. Perumalswami now wants to get better at her craft.
“As an academic hospitalist, it’s not just about doing research and writing papers and seeing papers,” she says, “but it’s also developing those leadership skills and helping that become an integral part of the educational experience.”
Question: What drew you to a career in HM?
Answer: I chose a career in academic hospital medicine primarily because I enjoy taking care of acutely ill, hospitalized, adult patients. I also really enjoy teaching medical students, residents, and fellows, and I enjoy doing that in the hospital setting. I think that there’s great satisfaction from taking care of a patient from admission to discharge.… I enjoyed every aspect of internal medicine, and when I graduated, I thought I could choose a subspecialty, but I felt that my skills and my expertise was really in taking care of the hospitalized patient.
Q: You have sought out extra training in palliative care and pain management. How has that impacted your career?
A: It’s not something that I necessarily started out thinking that I would specialize in, but the more I took care of hospitalized patients, the more I realized that we actually take care of a fair number of patients who have really complex symptom needs, and also really have a lot of needs with regard to recognizing when their prognosis is poor and understanding what their options are, if they’re even amenable to a palliative approach. I really felt that that was a skill that I needed to fine-tune. So I ended up gaining enough clinical experience and participating with hospice patients to the point where that’s really supplemented my hospitalist career, because what I found is that it’s made me a better hospitalist, and being a hospitalist has made me a better palliative-care doc.
Q: Working in academia, there’s no way to escape talk of the duty-hour rules recently put in place. What’s your view on the issue?
A: My view is that the work hours are here to stay. I think that there are some definite benefits that we’ve gained from having work hours. I’d say first and foremost of those gains is public trust. I think most physicians will tell you that they don’t want a physician who’s in the 36th hour of their day taking care of them when we know that studies actually can demonstrate that when you’ve been awake that long, that your cognitive abilities decline.
Q: But?
A: I think we have a lot of challenges, though, because a lot of things require creative solutions. And I think the first on that list is education, because that’s the first thing that I think has the potential to drop to the bottom of the list.
Q: In terms of HM’s growth, as you see residents coming through your program, how popular do you think the model is going to be with them moving forward?
A: I do, actually, because as an academic hospitalist, I’ve had several medical students and residents tell me, “Watching you, I think that I want to go into this field.” Or they’ll say, “What do you think about doing this for a year or two?” Or, “What do you think about subspecializing, and then being a hospitalist?” And my answer to all of them is it’s a dynamic specialty, and if you’re up for creating change and being a leader, it’s a good field, because we need people in a lot of different buckets, so to speak. We need people who have done other things in their career to contribute to our field.
Q: How do you prepare trainees for all the challenges coming down the pike?
A: A lot of the people who are doing work in medical education are starting to look to other fields to see if there are other models that we can adapt, or that we can somehow absorb into our practice. I think that there are some parts of our education which are not really formalized early on, but I think we have a lot to learn from organizational behavior circles, and systems that actually look at teams and leadership.
Q: What do the next five to 10 years hold for you?
A: All physician leaders have to stay somewhat in the clinical world. I think if you lose sight of that, you can’t be a very effective leader, or a very effective agent for change. Because part of my work is with palliative care, and I really feel that it’s affected my work as a hospitalist in a positive way, I don’t think I ever see myself leaving the clinical world completely. But I do see myself becoming, ideally, more involved with leadership and more involved with helping to train the next set of leaders.
Q: What do you see as SHM’s role specific to academic HM?
A: HM is changing the way healthcare is delivered in the U.S., and I think having an organization to represent us is vital to our success in other arenas of change—including healthcare policy and innovative care models. I see SHM as a large umbrella group, of which academic HM is one part. Academic hospitalists are increasingly involved in the education of future generations of physicians, and are uniquely poised for facilitating cascading leadership. The traditional, hierarchical model of attending-fellow-resident-medical student is shifting, and academic hospitalists are well-suited to study and explore this leadership structure and how it affects patient care, feedback, and mentoring.
Richard Quinn is a freelance writer based in New Jersey.
Chithra Perumalswami, MD, knew early on what she wanted to do with her life. As a teenager, she volunteered in an ED and with a hospice group, volunteerism that continued throughout her education. When she graduated from high school, she was tapped for Brown University’s Program in Liberal Medical Education, which calls itself the only baccalaureate-MD program in the Ivy League. And though she eventually turned down the offer, she pursued dual majors in cellular and molecular biology and English at the University of Michigan, where she earned her medical degree in 2004 and completed her residency.
In 2009, she participated in the Palliative Care Education and Practice Program at Harvard Medical School in Boston, a two-week post-graduate course aimed at professional development for physicians dedicated to careers in palliative-care education. “I really found that there were just so many aspects to caring for a patient as a palliative-care specialist and as a hospitalist that really strike at the heart of what being a doctor is,” says Dr. Perumalswami, assistant professor of medicine in the Division of Hospital Medicine at Northwestern Memorial Hospital in Chicago and one of four new members of Team Hospitalist. “I think it’s been an interest I’ve always had. During my residency training, I definitely experienced quite a few patient cases where I felt that we really needed to help patients and their families, and I didn’t necessarily have the best skill set to do that until I had more experience and more training.”
Dr. Perumalswami now wants to get better at her craft.
“As an academic hospitalist, it’s not just about doing research and writing papers and seeing papers,” she says, “but it’s also developing those leadership skills and helping that become an integral part of the educational experience.”
Question: What drew you to a career in HM?
Answer: I chose a career in academic hospital medicine primarily because I enjoy taking care of acutely ill, hospitalized, adult patients. I also really enjoy teaching medical students, residents, and fellows, and I enjoy doing that in the hospital setting. I think that there’s great satisfaction from taking care of a patient from admission to discharge.… I enjoyed every aspect of internal medicine, and when I graduated, I thought I could choose a subspecialty, but I felt that my skills and my expertise was really in taking care of the hospitalized patient.
Q: You have sought out extra training in palliative care and pain management. How has that impacted your career?
A: It’s not something that I necessarily started out thinking that I would specialize in, but the more I took care of hospitalized patients, the more I realized that we actually take care of a fair number of patients who have really complex symptom needs, and also really have a lot of needs with regard to recognizing when their prognosis is poor and understanding what their options are, if they’re even amenable to a palliative approach. I really felt that that was a skill that I needed to fine-tune. So I ended up gaining enough clinical experience and participating with hospice patients to the point where that’s really supplemented my hospitalist career, because what I found is that it’s made me a better hospitalist, and being a hospitalist has made me a better palliative-care doc.
Q: Working in academia, there’s no way to escape talk of the duty-hour rules recently put in place. What’s your view on the issue?
A: My view is that the work hours are here to stay. I think that there are some definite benefits that we’ve gained from having work hours. I’d say first and foremost of those gains is public trust. I think most physicians will tell you that they don’t want a physician who’s in the 36th hour of their day taking care of them when we know that studies actually can demonstrate that when you’ve been awake that long, that your cognitive abilities decline.
Q: But?
A: I think we have a lot of challenges, though, because a lot of things require creative solutions. And I think the first on that list is education, because that’s the first thing that I think has the potential to drop to the bottom of the list.
Q: In terms of HM’s growth, as you see residents coming through your program, how popular do you think the model is going to be with them moving forward?
A: I do, actually, because as an academic hospitalist, I’ve had several medical students and residents tell me, “Watching you, I think that I want to go into this field.” Or they’ll say, “What do you think about doing this for a year or two?” Or, “What do you think about subspecializing, and then being a hospitalist?” And my answer to all of them is it’s a dynamic specialty, and if you’re up for creating change and being a leader, it’s a good field, because we need people in a lot of different buckets, so to speak. We need people who have done other things in their career to contribute to our field.
Q: How do you prepare trainees for all the challenges coming down the pike?
A: A lot of the people who are doing work in medical education are starting to look to other fields to see if there are other models that we can adapt, or that we can somehow absorb into our practice. I think that there are some parts of our education which are not really formalized early on, but I think we have a lot to learn from organizational behavior circles, and systems that actually look at teams and leadership.
Q: What do the next five to 10 years hold for you?
A: All physician leaders have to stay somewhat in the clinical world. I think if you lose sight of that, you can’t be a very effective leader, or a very effective agent for change. Because part of my work is with palliative care, and I really feel that it’s affected my work as a hospitalist in a positive way, I don’t think I ever see myself leaving the clinical world completely. But I do see myself becoming, ideally, more involved with leadership and more involved with helping to train the next set of leaders.
Q: What do you see as SHM’s role specific to academic HM?
A: HM is changing the way healthcare is delivered in the U.S., and I think having an organization to represent us is vital to our success in other arenas of change—including healthcare policy and innovative care models. I see SHM as a large umbrella group, of which academic HM is one part. Academic hospitalists are increasingly involved in the education of future generations of physicians, and are uniquely poised for facilitating cascading leadership. The traditional, hierarchical model of attending-fellow-resident-medical student is shifting, and academic hospitalists are well-suited to study and explore this leadership structure and how it affects patient care, feedback, and mentoring.
Richard Quinn is a freelance writer based in New Jersey.
OB/GYN Hospitalists Emerge as a Specialty
The OB/GYN hospitalist field is growing, with at least 164 identified programs and 1,500 to 2,500 practitioners who spend all or part of their workweek in hospital labor and delivery departments. SHM and the American College of Obstetricians and Gynecologists helped birth the 90-member Society of OB/GYN Hospitalists in 2011, but it is now independent, says founding president Rob Olson, MD, an OB/GYN hospitalist practicing in Bellingham, Wash. The fledgling society is planning its second annual conference, Sept. 27-29 in Denver, with obstetric emergency simulation training, clinical lectures, and pearls from the experience of general hospitalist practice by HM pioneer John Nelson, MD, MHM.
Also known as laborists, these board-certified OB/GYN docs’ dedicated presence affords rapid on-site response to changes in patients’ conditions, Dr. Olson says. Laborists might cover nights and weekends, pick up unassigned patients, or cover for private obstetricians who are fully engaged. Laborists do not supplant the private practitioner’s role in delivering babies in the hospital, Dr. Olson says, “unless the private physician asks them to,” which, he adds, is happening more often.
Laborists typically are limited to labor and delivery services, although some also address gynecological cases in the ED. Most of the programs provide coverage 24/7, and invariably they are in facilities with medical hospitalists who might consult on medical complications for expectant mothers. One to two new programs open every month, Dr. Olson says, and his website lists 120 job openings. For information, visit www.ObGynHospitalist.com.
The OB/GYN hospitalist field is growing, with at least 164 identified programs and 1,500 to 2,500 practitioners who spend all or part of their workweek in hospital labor and delivery departments. SHM and the American College of Obstetricians and Gynecologists helped birth the 90-member Society of OB/GYN Hospitalists in 2011, but it is now independent, says founding president Rob Olson, MD, an OB/GYN hospitalist practicing in Bellingham, Wash. The fledgling society is planning its second annual conference, Sept. 27-29 in Denver, with obstetric emergency simulation training, clinical lectures, and pearls from the experience of general hospitalist practice by HM pioneer John Nelson, MD, MHM.
Also known as laborists, these board-certified OB/GYN docs’ dedicated presence affords rapid on-site response to changes in patients’ conditions, Dr. Olson says. Laborists might cover nights and weekends, pick up unassigned patients, or cover for private obstetricians who are fully engaged. Laborists do not supplant the private practitioner’s role in delivering babies in the hospital, Dr. Olson says, “unless the private physician asks them to,” which, he adds, is happening more often.
Laborists typically are limited to labor and delivery services, although some also address gynecological cases in the ED. Most of the programs provide coverage 24/7, and invariably they are in facilities with medical hospitalists who might consult on medical complications for expectant mothers. One to two new programs open every month, Dr. Olson says, and his website lists 120 job openings. For information, visit www.ObGynHospitalist.com.
The OB/GYN hospitalist field is growing, with at least 164 identified programs and 1,500 to 2,500 practitioners who spend all or part of their workweek in hospital labor and delivery departments. SHM and the American College of Obstetricians and Gynecologists helped birth the 90-member Society of OB/GYN Hospitalists in 2011, but it is now independent, says founding president Rob Olson, MD, an OB/GYN hospitalist practicing in Bellingham, Wash. The fledgling society is planning its second annual conference, Sept. 27-29 in Denver, with obstetric emergency simulation training, clinical lectures, and pearls from the experience of general hospitalist practice by HM pioneer John Nelson, MD, MHM.
Also known as laborists, these board-certified OB/GYN docs’ dedicated presence affords rapid on-site response to changes in patients’ conditions, Dr. Olson says. Laborists might cover nights and weekends, pick up unassigned patients, or cover for private obstetricians who are fully engaged. Laborists do not supplant the private practitioner’s role in delivering babies in the hospital, Dr. Olson says, “unless the private physician asks them to,” which, he adds, is happening more often.
Laborists typically are limited to labor and delivery services, although some also address gynecological cases in the ED. Most of the programs provide coverage 24/7, and invariably they are in facilities with medical hospitalists who might consult on medical complications for expectant mothers. One to two new programs open every month, Dr. Olson says, and his website lists 120 job openings. For information, visit www.ObGynHospitalist.com.
Consumers Union Joins Hospital Safety Ratings Game
A new hospital patient-safety ranking system was released in July by Consumers Union (CU), an independent consumer advocacy organization in Yonkers, N.Y., and published in the August issue of its magazine Consumer Reports.1 CU rated 1,155 hospitals in six categories, including hospital-acquired infections, readmissions, and the quality of communication by physicians and nurses to patients. The highest-scoring hospital: Billings Clinic in Montana, which received a 72 on CU’s 100-point safety score.
“The new Consumer Reports hospital safety ratings add to a growing list of publicly reported performance rating schemes,” says SHM president Shaun Frost, MD, SFHM, FACP, associate medical director for care-delivery systems at HealthPartners in Minneapolis. Hospitalists should be aware of these ratings and review them “with an eye toward identifying improvement opportunities that are within their scope of influence....Hospitalists must embrace these issues as theirs to own and improve upon, as the ability to demonstrably improve the safety and care quality in the hospitals in which we practice is dependent on us.”
Reference
A new hospital patient-safety ranking system was released in July by Consumers Union (CU), an independent consumer advocacy organization in Yonkers, N.Y., and published in the August issue of its magazine Consumer Reports.1 CU rated 1,155 hospitals in six categories, including hospital-acquired infections, readmissions, and the quality of communication by physicians and nurses to patients. The highest-scoring hospital: Billings Clinic in Montana, which received a 72 on CU’s 100-point safety score.
“The new Consumer Reports hospital safety ratings add to a growing list of publicly reported performance rating schemes,” says SHM president Shaun Frost, MD, SFHM, FACP, associate medical director for care-delivery systems at HealthPartners in Minneapolis. Hospitalists should be aware of these ratings and review them “with an eye toward identifying improvement opportunities that are within their scope of influence....Hospitalists must embrace these issues as theirs to own and improve upon, as the ability to demonstrably improve the safety and care quality in the hospitals in which we practice is dependent on us.”
Reference
A new hospital patient-safety ranking system was released in July by Consumers Union (CU), an independent consumer advocacy organization in Yonkers, N.Y., and published in the August issue of its magazine Consumer Reports.1 CU rated 1,155 hospitals in six categories, including hospital-acquired infections, readmissions, and the quality of communication by physicians and nurses to patients. The highest-scoring hospital: Billings Clinic in Montana, which received a 72 on CU’s 100-point safety score.
“The new Consumer Reports hospital safety ratings add to a growing list of publicly reported performance rating schemes,” says SHM president Shaun Frost, MD, SFHM, FACP, associate medical director for care-delivery systems at HealthPartners in Minneapolis. Hospitalists should be aware of these ratings and review them “with an eye toward identifying improvement opportunities that are within their scope of influence....Hospitalists must embrace these issues as theirs to own and improve upon, as the ability to demonstrably improve the safety and care quality in the hospitals in which we practice is dependent on us.”
Reference
Teach Back Communication Strategy Helps Healthcare Providers Help Their Patients
“Do you understand what I just told you?”
Hospitalists who are experts on communicating with patients say that this kind of simple, yes/no question can engender all sorts of confusion. This kind of query often results in unrealistic discharge plans, failed post-discharge treatments, and unnecessary rehospitalizations. And they happen all the time in hospitals across the country.
“Yes/no questions, unless they are very concrete—such as ‘Do you have a headache?’—have little relation to assessing a patient’s understanding of essential healthcare information,” says Jeffrey Greenwald, MD, SFHM, hospitalist and educator at Massachusetts General Hospital in Boston. Many times, he adds, patients say “yes” just to please the doctor or nurse, or to avoid looking ignorant.
A better approach to patient-provider communication, one that successfully addresses communication gaps, is teach-back, a strategy in which providers ask patients to repeat (or “teach back”), in their own words, what they have just learned about their medical condition, treatments, and self-care. For example, a physician using the teach-back method could ask, “Mr. Smith, can you tell me why you are in the hospital?”
The approach is an integral part of SHM’s Project BOOST (Better Outcomes for Older Adults through Safer Transitions) care-transitions quality initiative, says Dr. Greenwald, a BOOST coinvestigator. But too often, hospitalists fail to embrace the mandate of effective communication, he says, leaving responsibility to clarify the patient’s understanding to nurses and other members of the care team.
“Having taught this skill all over the country, I continue to find members of my own profession who feel that it is not their job to assess if patients understand self-care and medication instructions. To me, there’s no question that physicians have a role to play as teachers. We educate patients, whether we think we do or not,” Dr. Greenwald says, adding that some hospitalists do so with greater effectiveness than others.
In an era of accountable-care organizations, patient-centered care, and shared decision-making, hospitalists need to provide their patients the same core messages as the rest of the care team does. According to a recent report by the University of California San Francisco (UCSF) Center for Vulnerable Populations at San Francisco General Hospital, 77 million Americans have difficulty understanding even basic healthcare information.1
In addition to widespread language barriers, the Institute of Medicine in a 2004 report concluded that 90 million U.S. adults have literacy skills that test below the high-school level.2 As most hospitalists know, many inpatients go to great lengths to mask these limitations—appearing knowledgeable while failing to grasp essential health concepts. Medical jargon, acronyms, and instructions can be confusing, even for patients with a high level of education.
“First and foremost, we have to be able to communicate with our patients—to make sure that we understand their goals and that they understand our approach to treatment and the therapeutic goals we are proposing,” Dr. Greenwald says. “This process also helps to achieve true informed consent. We want to ensure that their questions are being answered, and that the options we are discussing are in line with their goals of care so that they are more likely to actually undertake them when they go home. And if they can’t, we need to know that in advance, so that we can intervene in other ways.”
Embracing the Method
One health system that has embraced teach-back as a patient-education strategy is the Lehigh Valley Health Network (LVHN), a 988-bed, three-hospital system based in Allentown, Pa. In an HM12 workshop, the Lehigh Valley team explained how its teach-back initiative grew out of quality initiatives to target patient flow and readmissions. Lehigh joined an Institute for Healthcare Improvement quality collaborative, implemented “Lean” quality-improvement (QI) methodologies, and adopted a patient-coaching program.3
“This process has shined a stark spotlight on how poorly we as physicians communicate with patients overall,” says Michael Pistoria, DO, SFHM, former hospitalist and president of medical staff at LVHN who was a member of the teach-back team.
“I’d like to think I’m a little above averagein this regard, but it has made me realize how much better I could be in checking in with patients, gauging their health literacy, and engaging them with the care plan. As hospitalists, we all have to rethink how we talk to patients and families.”
A multidisciplinary team, originally created to analyze care transitions, was divided into four work groups. One group, which focused on patient/family understanding of the disease process, quickly learned that care-team members often failed to identify the patient’s “key learner”—the patient, a family member, or someone else. If the information is given to the wrong person, breakdowns can result. Such breakdowns usually lead to readmissions. One instrumental change that came as a result of the QI team’s efforts is that LVHN care-team members now recognize it is their responsibility to ask who the key learner is and to put that person’s name on a whiteboard in the patient’s room.
In the workshop in at SHM’s annual meeting in San Diego, Kim Jordan, MHA, BSN, RN, NE-BC, LVHN’s administrator of patient-care services, described teach-back as an effective, easy-to-use communication strategy that improves patient learning outcomes. “We created a standard work process using teach-back strategies across the healthcare system,” with training offered to all professionals who provide education to patients and families, she said.
Starting with heart failure, prompts were written into the electronic health record (EHR) to provide four scripted teach-back questions, each focused on the patient’s knowledge, attitudes, and behavior, to be asked consecutively over three days. Information was “chunked” into manageable pieces, emphasizing what was most important for the patient to learn on that day.
Results from one of the pilot units showed 30-day readmission rates for heart failure patients were cut in half, from 28% to 14%.4 Teach-back scripts also are being developed for the anticoagulant clinic and for patients with stroke, myocardial infarction, chronic obstructive pulmonary disease (COPD), community-acquired pneumonia, and diabetes.
“Continued analysis continues to show reduced rehospitalizations, and we even find that for those who are readmitted, their second admissions have been shorter,” Dr. Pistoria says, noting LVHN nurses have reported higher satisfaction. “They say, ‘This is wonderful. This is what I love about nursing—I get to teach the patients.’”
A Quality Mandate
The importance of effective communication with hospitalized patients is recognized in the federal Hospital Consumer Assessment of Healthcare Providers and Systems (HCAHPS)5 quality ratings and, more recently, by Consumers Union’s new hospital safety ratings,6 which include communication in its six categories of patient-safety measures. And a recent study from the University of Washington says patients place high value on verbal communication about their discharge care plans and how to improve their health, and personal communication between their inpatient and outpatient providers.7
Large volumes of important information often are “dumped” on hospitalized patients, and many times patients are provided insufficient time to assimilate the information or ask questions about it.8 Such situations are especially common at discharge. And although physicians and other care-team members might feel they can’t afford the time to assure themselves that the patient understands what they are saying, the alternative is a lot more time spent dealing with preventable crises, misunderstandings—and preventable readmissions.
“People say, ‘I don’t have time for this,’” says Laura Vento, MSN, RN, clinical nurse leader on an acute-care medical unit at the University of California at San Diego medical center, who spearheaded teach-back at her hospital. “I did some observations around discharge visits, and it took an average of six minutes. After we implemented teach-back, it took eight to nine minutes. Nurses and patients were both very satisfied with the results. I say to staff: ‘Give it a try, and see what a difference an extra two minutes can make.’”
Dr. Pistoria describes teach-back as “humble inquiry...the simple need and ability to ask patients, ‘I know I’ve been throwing a lot of information at you. Can you tell me what I just said?’ Then, shut up and listen.” The goal is to have patients confirm that they understand fully what the provider thinks they need to know. The technique is presented as a test not of the patient’s learning ability, but of the provider’s communication effectiveness and success in explaining the information (see Figure 1). If the message has not been transmitted successfully, the professional reteaches, corrects misconceptions, and again asks the patient to teach back.
The theory is that physicians will then avoid the closed-ended questions (“Do you understand what I just told you?”) that make patients uncomfortable or inhibit the communication that needs to happen between patient and provider.
“We didn’t invent teach-back, which long predated Project BOOST,” Dr. Greenwald says. “But we use it and endorse it strongly, and believe it is an important part of communication with patients, particularly around care transitions.”
Dr. Greenwald thinks teach-back “is not a big stretch for hospitalists.” But he says it requires meaningful training and practice, ideally in a multidisciplinary team context. Participating Project BOOST (BOOST@hospitalmedicine.org) and Electronic Quality Improvement Programs (eQUIPS) sites receive a two-year license to post the “train-the-trainer” curriculum on their intranet systems.8 An instructional webinar, and the trainer curriculum and video, are available in the SHM store (www.hospitalmedicine.org/store). SHM also provides on-site training sessions for health systems or learning collaboratives (contact tbudnitz@hospitalmedicine.org).
‘Teach-Back on Steroids’
Teach-back, while a useful approach for improving patients’ understanding about hospital discharges, post-discharge care plans, and patient self-care, is just one of many teaching models that hospitalist groups can use to improve provider-to-patient communication. HM groups should assess health literacy in their regions and physician communication skills before deciding on one or more improvement tools.
LVHN, for example, has incorporated brief motivational interviewing techniques to its teach-back system, and the results are now being studied, says Paula Robinson, MSN, RN, BC, LVHN’s manager of patient, family, and consumer education.
“A lot of research out there emphasizes how patient education and knowledge alone don’t make a difference in adherence or compliance to treatment plans, even if they are getting the knowledge right 100% of the time,” Robinson says. “You also need to give patients permission to explore their feelings.”
One of Robinson’s colleagues, patient-care specialist Debra Peters, MSN, RN, BC, CMSRN, remembers using teach-back with a heart failure patient with recurrent rehospitalizations, exploring why it was important to control his salt intake. “The patient said, ‘Honey, I salt my ham, and I have no intention of changing that.’ This issue would not have come up if we had just addressed the knowledge component and told him: ‘You need to reduce your salt intake.’”
There might not be easy solutions to that kind of patient attitude, although in this case Peters made a referral to a dietitian who worked with the patient on food substitutions and other tools for managing his salt intake. “I don’t know if we made a big difference, but I haven’t seen him back in the hospital,” she says.
Motivational interviewing is a directive, client-centered counseling style for eliciting behavioral changes by helping clients to explore and resolve their ambivalence about making changes.9 Robinson calls it “the next step in our journey, with teach-back as a jumping-off place. We’ve worked on open-ended questions, getting patients to tell their stories, and our own reflective listening skills. I look at teach-back as a great communication tool and strategy—and motivational interviewing as ‘teach-back on steroids.’”
A number of similar, comparable, or complementary techniques and systems, with names like “Teach-to-Goal” and often paired with such resources as written materials, can help advance the same ends (visit the-hospitalist.org to learn about a technique for communicating bad news to patients).
The Flinders Program, developed at Flinders University in Adelaide, Australia, is a four-part structured motivational interview that asks patients to identify what they see as the biggest problem they face.10 LVHN medical educator Krista Hirschmann, MA, PhD, says this method was taught at LVHN by Australian experts and is now being used by its home-care department. “It is truly a patient-centered approach, and could be used by anybody in the health system,” she says.
Project RED (Re-Engineered Discharge), a care-transitions strategy developed by Brian Jack, MD, and colleagues at Boston University School of Medicine, was used to create a “virtual discharge advocate,” a computer avatar that simulates the face-to-face interaction between a patient and a nurse at the bedside.11 Patients interact with the avatar, named Louise, through a touch-screen display to review their after-hospital care packet and to answer her questions, confirming their understanding without being rushed for time.
Ultimately, Dr. Greenwald says, it doesn’t matter if physicians use teach-back or some other system to improve health literacy. “What matters is whether your patients understand what they need to know in order to go home and take care of themselves,” he says.
Larry Beresford is a freelance writer based in Oakland, Calif.
References
- Brach C, Keller D, Hernandez LM, et al. Ten attributes of health literate health care organizations. Institute of Medicine of the National Academies website. Available at: http://iom.edu/Global/Perspectives/2012/HealthLitAttributes.aspx. Accessed Aug. 9, 2012.
- Nielsen-Bohlman L, Panzer AM, Kindig DA, eds., Committee on Health Literacy. Health Literacy: A Prescription to End Confusion. Washington: Institute of Medicine; 2004.
- Care Transitions Program; University of Colorado School of Medicine, Division of Health Care Policy and Research. Available at: www.caretransitions.org. Accessed July 28, 2012.
- Healthcare Benchmarks and Quality Improvement. Teach-back program reduces readmissions. HighBeam Research website. Available at: http://www.highbeam.com/doc/1G1-269890846.html. Accessed Aug. 9, 2012.
- Hospital Consumer Assessment of Healthcare Providers and Systems. Centers for Medicare & Medicaid Services website. Available at: www.hcahpsonline.org. Accessed July 23, 2012.
- Consumer Reports. How safe is your hospital? Our new ratings find that some are riskier than others. Consumer Reports website. Available at: http://www.consumerreports.org/cro/magazine/2012/08/how-safe-is-your-hospital/index.htm. Accessed Aug. 1, 2012.
- Shoeb M, Merel SE, Jackson MB, Anawalt BD. “Can’t we just stop and talk?” Patients value verbal communication about discharge care plans. J Hosp Med. 2012;7(6):504-7.
- Schillinger D, Piette J, Grumbach K, et al. Closing the loop: physician communication with diabetic patients who have low health literacy. Arch Intern Med. 2003;163:83-90.
- Borrelli B, Riekert KA, Weinstein W, Rathier L. Brief motivational interviewing as a clinical strategy to promote asthma medication adherence. J Allergy Clinical Immunol. 2007;120(5):1023-30.
- eQUIPS (Electronic Quality Improvement Programs). Society of Hospital Medicine website. Available at: http:// www.hospitalmedicine.org/equips. Accessed Aug. 1, 2012.
- Kelly J, Kubina N. Navigating self-management: a practical approach for Australian health agencies. Flinders University, Adelaide, Australia. Available at: www.flinders.edu.au/medicine/sites/fhbhru/self-management.cfm. Accessed Aug. 1, 2012.
- Project RED (Re-Engineered Discharge). Boston University Medical Center website. Available at: http:// www.bu.edu/fammed/projectred/publications.html. Accessed Aug. 1, 2012.
“Do you understand what I just told you?”
Hospitalists who are experts on communicating with patients say that this kind of simple, yes/no question can engender all sorts of confusion. This kind of query often results in unrealistic discharge plans, failed post-discharge treatments, and unnecessary rehospitalizations. And they happen all the time in hospitals across the country.
“Yes/no questions, unless they are very concrete—such as ‘Do you have a headache?’—have little relation to assessing a patient’s understanding of essential healthcare information,” says Jeffrey Greenwald, MD, SFHM, hospitalist and educator at Massachusetts General Hospital in Boston. Many times, he adds, patients say “yes” just to please the doctor or nurse, or to avoid looking ignorant.
A better approach to patient-provider communication, one that successfully addresses communication gaps, is teach-back, a strategy in which providers ask patients to repeat (or “teach back”), in their own words, what they have just learned about their medical condition, treatments, and self-care. For example, a physician using the teach-back method could ask, “Mr. Smith, can you tell me why you are in the hospital?”
The approach is an integral part of SHM’s Project BOOST (Better Outcomes for Older Adults through Safer Transitions) care-transitions quality initiative, says Dr. Greenwald, a BOOST coinvestigator. But too often, hospitalists fail to embrace the mandate of effective communication, he says, leaving responsibility to clarify the patient’s understanding to nurses and other members of the care team.
“Having taught this skill all over the country, I continue to find members of my own profession who feel that it is not their job to assess if patients understand self-care and medication instructions. To me, there’s no question that physicians have a role to play as teachers. We educate patients, whether we think we do or not,” Dr. Greenwald says, adding that some hospitalists do so with greater effectiveness than others.
In an era of accountable-care organizations, patient-centered care, and shared decision-making, hospitalists need to provide their patients the same core messages as the rest of the care team does. According to a recent report by the University of California San Francisco (UCSF) Center for Vulnerable Populations at San Francisco General Hospital, 77 million Americans have difficulty understanding even basic healthcare information.1
In addition to widespread language barriers, the Institute of Medicine in a 2004 report concluded that 90 million U.S. adults have literacy skills that test below the high-school level.2 As most hospitalists know, many inpatients go to great lengths to mask these limitations—appearing knowledgeable while failing to grasp essential health concepts. Medical jargon, acronyms, and instructions can be confusing, even for patients with a high level of education.
“First and foremost, we have to be able to communicate with our patients—to make sure that we understand their goals and that they understand our approach to treatment and the therapeutic goals we are proposing,” Dr. Greenwald says. “This process also helps to achieve true informed consent. We want to ensure that their questions are being answered, and that the options we are discussing are in line with their goals of care so that they are more likely to actually undertake them when they go home. And if they can’t, we need to know that in advance, so that we can intervene in other ways.”
Embracing the Method
One health system that has embraced teach-back as a patient-education strategy is the Lehigh Valley Health Network (LVHN), a 988-bed, three-hospital system based in Allentown, Pa. In an HM12 workshop, the Lehigh Valley team explained how its teach-back initiative grew out of quality initiatives to target patient flow and readmissions. Lehigh joined an Institute for Healthcare Improvement quality collaborative, implemented “Lean” quality-improvement (QI) methodologies, and adopted a patient-coaching program.3
“This process has shined a stark spotlight on how poorly we as physicians communicate with patients overall,” says Michael Pistoria, DO, SFHM, former hospitalist and president of medical staff at LVHN who was a member of the teach-back team.
“I’d like to think I’m a little above averagein this regard, but it has made me realize how much better I could be in checking in with patients, gauging their health literacy, and engaging them with the care plan. As hospitalists, we all have to rethink how we talk to patients and families.”
A multidisciplinary team, originally created to analyze care transitions, was divided into four work groups. One group, which focused on patient/family understanding of the disease process, quickly learned that care-team members often failed to identify the patient’s “key learner”—the patient, a family member, or someone else. If the information is given to the wrong person, breakdowns can result. Such breakdowns usually lead to readmissions. One instrumental change that came as a result of the QI team’s efforts is that LVHN care-team members now recognize it is their responsibility to ask who the key learner is and to put that person’s name on a whiteboard in the patient’s room.
In the workshop in at SHM’s annual meeting in San Diego, Kim Jordan, MHA, BSN, RN, NE-BC, LVHN’s administrator of patient-care services, described teach-back as an effective, easy-to-use communication strategy that improves patient learning outcomes. “We created a standard work process using teach-back strategies across the healthcare system,” with training offered to all professionals who provide education to patients and families, she said.
Starting with heart failure, prompts were written into the electronic health record (EHR) to provide four scripted teach-back questions, each focused on the patient’s knowledge, attitudes, and behavior, to be asked consecutively over three days. Information was “chunked” into manageable pieces, emphasizing what was most important for the patient to learn on that day.
Results from one of the pilot units showed 30-day readmission rates for heart failure patients were cut in half, from 28% to 14%.4 Teach-back scripts also are being developed for the anticoagulant clinic and for patients with stroke, myocardial infarction, chronic obstructive pulmonary disease (COPD), community-acquired pneumonia, and diabetes.
“Continued analysis continues to show reduced rehospitalizations, and we even find that for those who are readmitted, their second admissions have been shorter,” Dr. Pistoria says, noting LVHN nurses have reported higher satisfaction. “They say, ‘This is wonderful. This is what I love about nursing—I get to teach the patients.’”
A Quality Mandate
The importance of effective communication with hospitalized patients is recognized in the federal Hospital Consumer Assessment of Healthcare Providers and Systems (HCAHPS)5 quality ratings and, more recently, by Consumers Union’s new hospital safety ratings,6 which include communication in its six categories of patient-safety measures. And a recent study from the University of Washington says patients place high value on verbal communication about their discharge care plans and how to improve their health, and personal communication between their inpatient and outpatient providers.7
Large volumes of important information often are “dumped” on hospitalized patients, and many times patients are provided insufficient time to assimilate the information or ask questions about it.8 Such situations are especially common at discharge. And although physicians and other care-team members might feel they can’t afford the time to assure themselves that the patient understands what they are saying, the alternative is a lot more time spent dealing with preventable crises, misunderstandings—and preventable readmissions.
“People say, ‘I don’t have time for this,’” says Laura Vento, MSN, RN, clinical nurse leader on an acute-care medical unit at the University of California at San Diego medical center, who spearheaded teach-back at her hospital. “I did some observations around discharge visits, and it took an average of six minutes. After we implemented teach-back, it took eight to nine minutes. Nurses and patients were both very satisfied with the results. I say to staff: ‘Give it a try, and see what a difference an extra two minutes can make.’”
Dr. Pistoria describes teach-back as “humble inquiry...the simple need and ability to ask patients, ‘I know I’ve been throwing a lot of information at you. Can you tell me what I just said?’ Then, shut up and listen.” The goal is to have patients confirm that they understand fully what the provider thinks they need to know. The technique is presented as a test not of the patient’s learning ability, but of the provider’s communication effectiveness and success in explaining the information (see Figure 1). If the message has not been transmitted successfully, the professional reteaches, corrects misconceptions, and again asks the patient to teach back.
The theory is that physicians will then avoid the closed-ended questions (“Do you understand what I just told you?”) that make patients uncomfortable or inhibit the communication that needs to happen between patient and provider.
“We didn’t invent teach-back, which long predated Project BOOST,” Dr. Greenwald says. “But we use it and endorse it strongly, and believe it is an important part of communication with patients, particularly around care transitions.”
Dr. Greenwald thinks teach-back “is not a big stretch for hospitalists.” But he says it requires meaningful training and practice, ideally in a multidisciplinary team context. Participating Project BOOST (BOOST@hospitalmedicine.org) and Electronic Quality Improvement Programs (eQUIPS) sites receive a two-year license to post the “train-the-trainer” curriculum on their intranet systems.8 An instructional webinar, and the trainer curriculum and video, are available in the SHM store (www.hospitalmedicine.org/store). SHM also provides on-site training sessions for health systems or learning collaboratives (contact tbudnitz@hospitalmedicine.org).
‘Teach-Back on Steroids’
Teach-back, while a useful approach for improving patients’ understanding about hospital discharges, post-discharge care plans, and patient self-care, is just one of many teaching models that hospitalist groups can use to improve provider-to-patient communication. HM groups should assess health literacy in their regions and physician communication skills before deciding on one or more improvement tools.
LVHN, for example, has incorporated brief motivational interviewing techniques to its teach-back system, and the results are now being studied, says Paula Robinson, MSN, RN, BC, LVHN’s manager of patient, family, and consumer education.
“A lot of research out there emphasizes how patient education and knowledge alone don’t make a difference in adherence or compliance to treatment plans, even if they are getting the knowledge right 100% of the time,” Robinson says. “You also need to give patients permission to explore their feelings.”
One of Robinson’s colleagues, patient-care specialist Debra Peters, MSN, RN, BC, CMSRN, remembers using teach-back with a heart failure patient with recurrent rehospitalizations, exploring why it was important to control his salt intake. “The patient said, ‘Honey, I salt my ham, and I have no intention of changing that.’ This issue would not have come up if we had just addressed the knowledge component and told him: ‘You need to reduce your salt intake.’”
There might not be easy solutions to that kind of patient attitude, although in this case Peters made a referral to a dietitian who worked with the patient on food substitutions and other tools for managing his salt intake. “I don’t know if we made a big difference, but I haven’t seen him back in the hospital,” she says.
Motivational interviewing is a directive, client-centered counseling style for eliciting behavioral changes by helping clients to explore and resolve their ambivalence about making changes.9 Robinson calls it “the next step in our journey, with teach-back as a jumping-off place. We’ve worked on open-ended questions, getting patients to tell their stories, and our own reflective listening skills. I look at teach-back as a great communication tool and strategy—and motivational interviewing as ‘teach-back on steroids.’”
A number of similar, comparable, or complementary techniques and systems, with names like “Teach-to-Goal” and often paired with such resources as written materials, can help advance the same ends (visit the-hospitalist.org to learn about a technique for communicating bad news to patients).
The Flinders Program, developed at Flinders University in Adelaide, Australia, is a four-part structured motivational interview that asks patients to identify what they see as the biggest problem they face.10 LVHN medical educator Krista Hirschmann, MA, PhD, says this method was taught at LVHN by Australian experts and is now being used by its home-care department. “It is truly a patient-centered approach, and could be used by anybody in the health system,” she says.
Project RED (Re-Engineered Discharge), a care-transitions strategy developed by Brian Jack, MD, and colleagues at Boston University School of Medicine, was used to create a “virtual discharge advocate,” a computer avatar that simulates the face-to-face interaction between a patient and a nurse at the bedside.11 Patients interact with the avatar, named Louise, through a touch-screen display to review their after-hospital care packet and to answer her questions, confirming their understanding without being rushed for time.
Ultimately, Dr. Greenwald says, it doesn’t matter if physicians use teach-back or some other system to improve health literacy. “What matters is whether your patients understand what they need to know in order to go home and take care of themselves,” he says.
Larry Beresford is a freelance writer based in Oakland, Calif.
References
- Brach C, Keller D, Hernandez LM, et al. Ten attributes of health literate health care organizations. Institute of Medicine of the National Academies website. Available at: http://iom.edu/Global/Perspectives/2012/HealthLitAttributes.aspx. Accessed Aug. 9, 2012.
- Nielsen-Bohlman L, Panzer AM, Kindig DA, eds., Committee on Health Literacy. Health Literacy: A Prescription to End Confusion. Washington: Institute of Medicine; 2004.
- Care Transitions Program; University of Colorado School of Medicine, Division of Health Care Policy and Research. Available at: www.caretransitions.org. Accessed July 28, 2012.
- Healthcare Benchmarks and Quality Improvement. Teach-back program reduces readmissions. HighBeam Research website. Available at: http://www.highbeam.com/doc/1G1-269890846.html. Accessed Aug. 9, 2012.
- Hospital Consumer Assessment of Healthcare Providers and Systems. Centers for Medicare & Medicaid Services website. Available at: www.hcahpsonline.org. Accessed July 23, 2012.
- Consumer Reports. How safe is your hospital? Our new ratings find that some are riskier than others. Consumer Reports website. Available at: http://www.consumerreports.org/cro/magazine/2012/08/how-safe-is-your-hospital/index.htm. Accessed Aug. 1, 2012.
- Shoeb M, Merel SE, Jackson MB, Anawalt BD. “Can’t we just stop and talk?” Patients value verbal communication about discharge care plans. J Hosp Med. 2012;7(6):504-7.
- Schillinger D, Piette J, Grumbach K, et al. Closing the loop: physician communication with diabetic patients who have low health literacy. Arch Intern Med. 2003;163:83-90.
- Borrelli B, Riekert KA, Weinstein W, Rathier L. Brief motivational interviewing as a clinical strategy to promote asthma medication adherence. J Allergy Clinical Immunol. 2007;120(5):1023-30.
- eQUIPS (Electronic Quality Improvement Programs). Society of Hospital Medicine website. Available at: http:// www.hospitalmedicine.org/equips. Accessed Aug. 1, 2012.
- Kelly J, Kubina N. Navigating self-management: a practical approach for Australian health agencies. Flinders University, Adelaide, Australia. Available at: www.flinders.edu.au/medicine/sites/fhbhru/self-management.cfm. Accessed Aug. 1, 2012.
- Project RED (Re-Engineered Discharge). Boston University Medical Center website. Available at: http:// www.bu.edu/fammed/projectred/publications.html. Accessed Aug. 1, 2012.
“Do you understand what I just told you?”
Hospitalists who are experts on communicating with patients say that this kind of simple, yes/no question can engender all sorts of confusion. This kind of query often results in unrealistic discharge plans, failed post-discharge treatments, and unnecessary rehospitalizations. And they happen all the time in hospitals across the country.
“Yes/no questions, unless they are very concrete—such as ‘Do you have a headache?’—have little relation to assessing a patient’s understanding of essential healthcare information,” says Jeffrey Greenwald, MD, SFHM, hospitalist and educator at Massachusetts General Hospital in Boston. Many times, he adds, patients say “yes” just to please the doctor or nurse, or to avoid looking ignorant.
A better approach to patient-provider communication, one that successfully addresses communication gaps, is teach-back, a strategy in which providers ask patients to repeat (or “teach back”), in their own words, what they have just learned about their medical condition, treatments, and self-care. For example, a physician using the teach-back method could ask, “Mr. Smith, can you tell me why you are in the hospital?”
The approach is an integral part of SHM’s Project BOOST (Better Outcomes for Older Adults through Safer Transitions) care-transitions quality initiative, says Dr. Greenwald, a BOOST coinvestigator. But too often, hospitalists fail to embrace the mandate of effective communication, he says, leaving responsibility to clarify the patient’s understanding to nurses and other members of the care team.
“Having taught this skill all over the country, I continue to find members of my own profession who feel that it is not their job to assess if patients understand self-care and medication instructions. To me, there’s no question that physicians have a role to play as teachers. We educate patients, whether we think we do or not,” Dr. Greenwald says, adding that some hospitalists do so with greater effectiveness than others.
In an era of accountable-care organizations, patient-centered care, and shared decision-making, hospitalists need to provide their patients the same core messages as the rest of the care team does. According to a recent report by the University of California San Francisco (UCSF) Center for Vulnerable Populations at San Francisco General Hospital, 77 million Americans have difficulty understanding even basic healthcare information.1
In addition to widespread language barriers, the Institute of Medicine in a 2004 report concluded that 90 million U.S. adults have literacy skills that test below the high-school level.2 As most hospitalists know, many inpatients go to great lengths to mask these limitations—appearing knowledgeable while failing to grasp essential health concepts. Medical jargon, acronyms, and instructions can be confusing, even for patients with a high level of education.
“First and foremost, we have to be able to communicate with our patients—to make sure that we understand their goals and that they understand our approach to treatment and the therapeutic goals we are proposing,” Dr. Greenwald says. “This process also helps to achieve true informed consent. We want to ensure that their questions are being answered, and that the options we are discussing are in line with their goals of care so that they are more likely to actually undertake them when they go home. And if they can’t, we need to know that in advance, so that we can intervene in other ways.”
Embracing the Method
One health system that has embraced teach-back as a patient-education strategy is the Lehigh Valley Health Network (LVHN), a 988-bed, three-hospital system based in Allentown, Pa. In an HM12 workshop, the Lehigh Valley team explained how its teach-back initiative grew out of quality initiatives to target patient flow and readmissions. Lehigh joined an Institute for Healthcare Improvement quality collaborative, implemented “Lean” quality-improvement (QI) methodologies, and adopted a patient-coaching program.3
“This process has shined a stark spotlight on how poorly we as physicians communicate with patients overall,” says Michael Pistoria, DO, SFHM, former hospitalist and president of medical staff at LVHN who was a member of the teach-back team.
“I’d like to think I’m a little above averagein this regard, but it has made me realize how much better I could be in checking in with patients, gauging their health literacy, and engaging them with the care plan. As hospitalists, we all have to rethink how we talk to patients and families.”
A multidisciplinary team, originally created to analyze care transitions, was divided into four work groups. One group, which focused on patient/family understanding of the disease process, quickly learned that care-team members often failed to identify the patient’s “key learner”—the patient, a family member, or someone else. If the information is given to the wrong person, breakdowns can result. Such breakdowns usually lead to readmissions. One instrumental change that came as a result of the QI team’s efforts is that LVHN care-team members now recognize it is their responsibility to ask who the key learner is and to put that person’s name on a whiteboard in the patient’s room.
In the workshop in at SHM’s annual meeting in San Diego, Kim Jordan, MHA, BSN, RN, NE-BC, LVHN’s administrator of patient-care services, described teach-back as an effective, easy-to-use communication strategy that improves patient learning outcomes. “We created a standard work process using teach-back strategies across the healthcare system,” with training offered to all professionals who provide education to patients and families, she said.
Starting with heart failure, prompts were written into the electronic health record (EHR) to provide four scripted teach-back questions, each focused on the patient’s knowledge, attitudes, and behavior, to be asked consecutively over three days. Information was “chunked” into manageable pieces, emphasizing what was most important for the patient to learn on that day.
Results from one of the pilot units showed 30-day readmission rates for heart failure patients were cut in half, from 28% to 14%.4 Teach-back scripts also are being developed for the anticoagulant clinic and for patients with stroke, myocardial infarction, chronic obstructive pulmonary disease (COPD), community-acquired pneumonia, and diabetes.
“Continued analysis continues to show reduced rehospitalizations, and we even find that for those who are readmitted, their second admissions have been shorter,” Dr. Pistoria says, noting LVHN nurses have reported higher satisfaction. “They say, ‘This is wonderful. This is what I love about nursing—I get to teach the patients.’”
A Quality Mandate
The importance of effective communication with hospitalized patients is recognized in the federal Hospital Consumer Assessment of Healthcare Providers and Systems (HCAHPS)5 quality ratings and, more recently, by Consumers Union’s new hospital safety ratings,6 which include communication in its six categories of patient-safety measures. And a recent study from the University of Washington says patients place high value on verbal communication about their discharge care plans and how to improve their health, and personal communication between their inpatient and outpatient providers.7
Large volumes of important information often are “dumped” on hospitalized patients, and many times patients are provided insufficient time to assimilate the information or ask questions about it.8 Such situations are especially common at discharge. And although physicians and other care-team members might feel they can’t afford the time to assure themselves that the patient understands what they are saying, the alternative is a lot more time spent dealing with preventable crises, misunderstandings—and preventable readmissions.
“People say, ‘I don’t have time for this,’” says Laura Vento, MSN, RN, clinical nurse leader on an acute-care medical unit at the University of California at San Diego medical center, who spearheaded teach-back at her hospital. “I did some observations around discharge visits, and it took an average of six minutes. After we implemented teach-back, it took eight to nine minutes. Nurses and patients were both very satisfied with the results. I say to staff: ‘Give it a try, and see what a difference an extra two minutes can make.’”
Dr. Pistoria describes teach-back as “humble inquiry...the simple need and ability to ask patients, ‘I know I’ve been throwing a lot of information at you. Can you tell me what I just said?’ Then, shut up and listen.” The goal is to have patients confirm that they understand fully what the provider thinks they need to know. The technique is presented as a test not of the patient’s learning ability, but of the provider’s communication effectiveness and success in explaining the information (see Figure 1). If the message has not been transmitted successfully, the professional reteaches, corrects misconceptions, and again asks the patient to teach back.
The theory is that physicians will then avoid the closed-ended questions (“Do you understand what I just told you?”) that make patients uncomfortable or inhibit the communication that needs to happen between patient and provider.
“We didn’t invent teach-back, which long predated Project BOOST,” Dr. Greenwald says. “But we use it and endorse it strongly, and believe it is an important part of communication with patients, particularly around care transitions.”
Dr. Greenwald thinks teach-back “is not a big stretch for hospitalists.” But he says it requires meaningful training and practice, ideally in a multidisciplinary team context. Participating Project BOOST (BOOST@hospitalmedicine.org) and Electronic Quality Improvement Programs (eQUIPS) sites receive a two-year license to post the “train-the-trainer” curriculum on their intranet systems.8 An instructional webinar, and the trainer curriculum and video, are available in the SHM store (www.hospitalmedicine.org/store). SHM also provides on-site training sessions for health systems or learning collaboratives (contact tbudnitz@hospitalmedicine.org).
‘Teach-Back on Steroids’
Teach-back, while a useful approach for improving patients’ understanding about hospital discharges, post-discharge care plans, and patient self-care, is just one of many teaching models that hospitalist groups can use to improve provider-to-patient communication. HM groups should assess health literacy in their regions and physician communication skills before deciding on one or more improvement tools.
LVHN, for example, has incorporated brief motivational interviewing techniques to its teach-back system, and the results are now being studied, says Paula Robinson, MSN, RN, BC, LVHN’s manager of patient, family, and consumer education.
“A lot of research out there emphasizes how patient education and knowledge alone don’t make a difference in adherence or compliance to treatment plans, even if they are getting the knowledge right 100% of the time,” Robinson says. “You also need to give patients permission to explore their feelings.”
One of Robinson’s colleagues, patient-care specialist Debra Peters, MSN, RN, BC, CMSRN, remembers using teach-back with a heart failure patient with recurrent rehospitalizations, exploring why it was important to control his salt intake. “The patient said, ‘Honey, I salt my ham, and I have no intention of changing that.’ This issue would not have come up if we had just addressed the knowledge component and told him: ‘You need to reduce your salt intake.’”
There might not be easy solutions to that kind of patient attitude, although in this case Peters made a referral to a dietitian who worked with the patient on food substitutions and other tools for managing his salt intake. “I don’t know if we made a big difference, but I haven’t seen him back in the hospital,” she says.
Motivational interviewing is a directive, client-centered counseling style for eliciting behavioral changes by helping clients to explore and resolve their ambivalence about making changes.9 Robinson calls it “the next step in our journey, with teach-back as a jumping-off place. We’ve worked on open-ended questions, getting patients to tell their stories, and our own reflective listening skills. I look at teach-back as a great communication tool and strategy—and motivational interviewing as ‘teach-back on steroids.’”
A number of similar, comparable, or complementary techniques and systems, with names like “Teach-to-Goal” and often paired with such resources as written materials, can help advance the same ends (visit the-hospitalist.org to learn about a technique for communicating bad news to patients).
The Flinders Program, developed at Flinders University in Adelaide, Australia, is a four-part structured motivational interview that asks patients to identify what they see as the biggest problem they face.10 LVHN medical educator Krista Hirschmann, MA, PhD, says this method was taught at LVHN by Australian experts and is now being used by its home-care department. “It is truly a patient-centered approach, and could be used by anybody in the health system,” she says.
Project RED (Re-Engineered Discharge), a care-transitions strategy developed by Brian Jack, MD, and colleagues at Boston University School of Medicine, was used to create a “virtual discharge advocate,” a computer avatar that simulates the face-to-face interaction between a patient and a nurse at the bedside.11 Patients interact with the avatar, named Louise, through a touch-screen display to review their after-hospital care packet and to answer her questions, confirming their understanding without being rushed for time.
Ultimately, Dr. Greenwald says, it doesn’t matter if physicians use teach-back or some other system to improve health literacy. “What matters is whether your patients understand what they need to know in order to go home and take care of themselves,” he says.
Larry Beresford is a freelance writer based in Oakland, Calif.
References
- Brach C, Keller D, Hernandez LM, et al. Ten attributes of health literate health care organizations. Institute of Medicine of the National Academies website. Available at: http://iom.edu/Global/Perspectives/2012/HealthLitAttributes.aspx. Accessed Aug. 9, 2012.
- Nielsen-Bohlman L, Panzer AM, Kindig DA, eds., Committee on Health Literacy. Health Literacy: A Prescription to End Confusion. Washington: Institute of Medicine; 2004.
- Care Transitions Program; University of Colorado School of Medicine, Division of Health Care Policy and Research. Available at: www.caretransitions.org. Accessed July 28, 2012.
- Healthcare Benchmarks and Quality Improvement. Teach-back program reduces readmissions. HighBeam Research website. Available at: http://www.highbeam.com/doc/1G1-269890846.html. Accessed Aug. 9, 2012.
- Hospital Consumer Assessment of Healthcare Providers and Systems. Centers for Medicare & Medicaid Services website. Available at: www.hcahpsonline.org. Accessed July 23, 2012.
- Consumer Reports. How safe is your hospital? Our new ratings find that some are riskier than others. Consumer Reports website. Available at: http://www.consumerreports.org/cro/magazine/2012/08/how-safe-is-your-hospital/index.htm. Accessed Aug. 1, 2012.
- Shoeb M, Merel SE, Jackson MB, Anawalt BD. “Can’t we just stop and talk?” Patients value verbal communication about discharge care plans. J Hosp Med. 2012;7(6):504-7.
- Schillinger D, Piette J, Grumbach K, et al. Closing the loop: physician communication with diabetic patients who have low health literacy. Arch Intern Med. 2003;163:83-90.
- Borrelli B, Riekert KA, Weinstein W, Rathier L. Brief motivational interviewing as a clinical strategy to promote asthma medication adherence. J Allergy Clinical Immunol. 2007;120(5):1023-30.
- eQUIPS (Electronic Quality Improvement Programs). Society of Hospital Medicine website. Available at: http:// www.hospitalmedicine.org/equips. Accessed Aug. 1, 2012.
- Kelly J, Kubina N. Navigating self-management: a practical approach for Australian health agencies. Flinders University, Adelaide, Australia. Available at: www.flinders.edu.au/medicine/sites/fhbhru/self-management.cfm. Accessed Aug. 1, 2012.
- Project RED (Re-Engineered Discharge). Boston University Medical Center website. Available at: http:// www.bu.edu/fammed/projectred/publications.html. Accessed Aug. 1, 2012.
HM Leaders Highlight Benefits of Specialty Hospitalist Programs
Safer patients, improved quality of care, innovative uses of resources, increased job satisfaction: Those themes threaded their way through presentation after presentation at “Creating the Hospital of the Future: The Implications for Hospital-Focused Physician Practice,” a one-day meeting of hospitalist leaders and hospital administrators following the annual Health Forum/AHA Leadership Summit on July 21 in San Francisco.
The five-hour presentation to about 80 hospital CEOs, chief financial officers, and chief medical officers focused on the ever-expanding roles of subspecialty hospitalists and how subspecialty hospitalist programs can help administrators solve multiple challenges in an era of healthcare reform.
“When I started in 2007, I could only identify 15 hospitals in the United States with OB hospitalist programs. And now we know of 169, and the nation is adding one or two new programs a month,” says Rob Olson, MD, an OB/GYN hospitalistor “laborist”at PeaceHealth St. Joseph Medical Center in Bellingham, Wash., and editor of ObGynHospitalist.com. “If you think about it, if you can make it safer for women in labor, then duh! Why wouldn’t you want to do that?
“The finances work best at a hospital that delivers more than 2,000 babies a year. But many small hospitals, like those doing 800 or 1,000 deliveries a year, have programs because it makes it safer for women in labor,” he says. “Therefore, even though it might be more expensive at those lower numbers, it’s worth it. It’s the right thing to do.”
The July meeting was the second time SHM gathered stakeholders to discuss the growth of specialty hospitalists; a similar panel of experts convened last November in Las Vegas. Most at the San Francisco meeting recognized the upward trend in such HM-focused subspecialties as neurology, orthopedics, obstetrics, and general surgery, according to John Nelson, MD, MHM, organizer of the focused-practice meetings.
“Most people in healthcare feel like this is going to continue and intensify,” says Dr. Nelson, cofounder and past president of SHM, medical director of the hospitalist practice at Overlake Hospital Medical Center, Bellevue, Wash., and practice-management columnist for The Hospitalist. “There is consensus that each specialty can learn from the other about how they organize their practice and approach their work. There was consensus that we don’t have a lot of research data about what this means for things like cost of care, quality of care, patient experience, physician career longevity. So we need to encourage people to begin to study those things, and what this means for the stakeholders in healthcare.”
Four subspecialty hospitalists—neurohospitalist David Likosky, MD, SFHM, surgicalist John Maa, MD, orthopedic hospitalist Kurt Ehlert, MD, and Dr. Olson, the laborist—took part in a 90-minute panel discussion in which they explained their practice models and fielded questions from the audience.
“My message was that there are a large number of people doing this now—we estimate there are between 600 and 700 neurohospitalists nationally—and that the model holds a lot of promise. There’s not a huge amount of data right now on outcomes and other metrics, but we’re starting to see that data,” said Dr. Likosky, medical director of the Evergreen Neuroscience Institute in Kirkland, Wash., and co-founder of the Neurohospitalist Society (neurohospitalistsociety.org). “The neurohospitalist model is a good solution to many of the problems that hospitals are facing now.”
—Kurt Ehlert, MD, medical director, Orthopedic Hospitalists of New Bern, national director for orthopedic services, Delphi Healthcare Partners, Morrisville, N.C.
Dr. Ehlert, director of orthopaedic services, Orthopeadic Hospitalists of New Bern (N.C.), and national orthopaedic medical director of Delphi of TEAMHealth, says subspecialty HM programs offer hospitals a “great chance of improving quality and patient safety over what they have currently, even if they have their emergency room covered.”
“I think hospitalists who are subspecialists in trauma can provide a bump up in productivity, safety, reliability,” he says. “Results will be better. I think it will be less expensive. [I told them] that there is an option out there that can benefit them in all of the various key ways that groups are looking at right now.”
Dr. Ehlert’s ortho-hospitalist group formed when the 300-bed hospital in New Bern encountered a manpower issue not unfamiliar to hospitals across the country. The bylaws of the medical staff allowed subspecialists to stop taking call when they turned 55, and four of the seven orthopedists aged out.
“Three doctors taking all the call is not really sustainable for them in their private practices,” Dr. Ehlert said. “So they looked at various options, came to us, and we started in December of 2009. It has been very successful, according to the administration. They love us being there. The emergency room is very happy with our responsiveness. I think our results have been very good.”
The new arrangement is a win-win, Dr. Ehlert says. The orthopedists are focused on their elective practices, and “they’re very happy with that. So their life is much better; their elective practice has actually gotten busier because they’re not having to leave space open for all the trauma from the ER. So I think all around it’s been very successful.”
The hospital has added a general surgery hospitalist program, which is doing well, too. “They’re much busier,” he says. “They’re really taking a load off the general surgeons in town.”
Dr. Maa, assistant professor and director of the surgical hospitalist program at University of California San Francisco Medical Center, says growth in his field is fueled by the ever-growing crises in emergency departments.
“Most hospitals critically depend on a general surgical service,” he said. “If you can’t keep a panel of general surgeons to take call, you’re probably going to have to close your emergency room.”
Dr. Maa, who founded UCSF’s surgical hospitalist program in 2005, explained how the terms “surgicalist” and “acute-care surgeon” have come to represent the concept of a dedicated emergency surgeon, whether it be in trauma or in general surgery. “It really does parallel the medical hospitalist model,” he says, adding that his field has had to overcome doubts about scheduling and patient safety.
“The danger in each of these specialty programs is to become too much of the silo mentality, to focus on their own discipline,” he adds. “We need to work across specialties, we need to collaborate, we need to find ways of utilizing the precious existing resources for emergency care, and make certain that the needs of society are met. Society places trust in doctors, hospital leaders, to build a system that will care for them when they need it. It’s our ethical obligation to design the safest, best system, with the resources that we have.”
Jason Carris is editor of The Hospitalist.
Safer patients, improved quality of care, innovative uses of resources, increased job satisfaction: Those themes threaded their way through presentation after presentation at “Creating the Hospital of the Future: The Implications for Hospital-Focused Physician Practice,” a one-day meeting of hospitalist leaders and hospital administrators following the annual Health Forum/AHA Leadership Summit on July 21 in San Francisco.
The five-hour presentation to about 80 hospital CEOs, chief financial officers, and chief medical officers focused on the ever-expanding roles of subspecialty hospitalists and how subspecialty hospitalist programs can help administrators solve multiple challenges in an era of healthcare reform.
“When I started in 2007, I could only identify 15 hospitals in the United States with OB hospitalist programs. And now we know of 169, and the nation is adding one or two new programs a month,” says Rob Olson, MD, an OB/GYN hospitalistor “laborist”at PeaceHealth St. Joseph Medical Center in Bellingham, Wash., and editor of ObGynHospitalist.com. “If you think about it, if you can make it safer for women in labor, then duh! Why wouldn’t you want to do that?
“The finances work best at a hospital that delivers more than 2,000 babies a year. But many small hospitals, like those doing 800 or 1,000 deliveries a year, have programs because it makes it safer for women in labor,” he says. “Therefore, even though it might be more expensive at those lower numbers, it’s worth it. It’s the right thing to do.”
The July meeting was the second time SHM gathered stakeholders to discuss the growth of specialty hospitalists; a similar panel of experts convened last November in Las Vegas. Most at the San Francisco meeting recognized the upward trend in such HM-focused subspecialties as neurology, orthopedics, obstetrics, and general surgery, according to John Nelson, MD, MHM, organizer of the focused-practice meetings.
“Most people in healthcare feel like this is going to continue and intensify,” says Dr. Nelson, cofounder and past president of SHM, medical director of the hospitalist practice at Overlake Hospital Medical Center, Bellevue, Wash., and practice-management columnist for The Hospitalist. “There is consensus that each specialty can learn from the other about how they organize their practice and approach their work. There was consensus that we don’t have a lot of research data about what this means for things like cost of care, quality of care, patient experience, physician career longevity. So we need to encourage people to begin to study those things, and what this means for the stakeholders in healthcare.”
Four subspecialty hospitalists—neurohospitalist David Likosky, MD, SFHM, surgicalist John Maa, MD, orthopedic hospitalist Kurt Ehlert, MD, and Dr. Olson, the laborist—took part in a 90-minute panel discussion in which they explained their practice models and fielded questions from the audience.
“My message was that there are a large number of people doing this now—we estimate there are between 600 and 700 neurohospitalists nationally—and that the model holds a lot of promise. There’s not a huge amount of data right now on outcomes and other metrics, but we’re starting to see that data,” said Dr. Likosky, medical director of the Evergreen Neuroscience Institute in Kirkland, Wash., and co-founder of the Neurohospitalist Society (neurohospitalistsociety.org). “The neurohospitalist model is a good solution to many of the problems that hospitals are facing now.”
—Kurt Ehlert, MD, medical director, Orthopedic Hospitalists of New Bern, national director for orthopedic services, Delphi Healthcare Partners, Morrisville, N.C.
Dr. Ehlert, director of orthopaedic services, Orthopeadic Hospitalists of New Bern (N.C.), and national orthopaedic medical director of Delphi of TEAMHealth, says subspecialty HM programs offer hospitals a “great chance of improving quality and patient safety over what they have currently, even if they have their emergency room covered.”
“I think hospitalists who are subspecialists in trauma can provide a bump up in productivity, safety, reliability,” he says. “Results will be better. I think it will be less expensive. [I told them] that there is an option out there that can benefit them in all of the various key ways that groups are looking at right now.”
Dr. Ehlert’s ortho-hospitalist group formed when the 300-bed hospital in New Bern encountered a manpower issue not unfamiliar to hospitals across the country. The bylaws of the medical staff allowed subspecialists to stop taking call when they turned 55, and four of the seven orthopedists aged out.
“Three doctors taking all the call is not really sustainable for them in their private practices,” Dr. Ehlert said. “So they looked at various options, came to us, and we started in December of 2009. It has been very successful, according to the administration. They love us being there. The emergency room is very happy with our responsiveness. I think our results have been very good.”
The new arrangement is a win-win, Dr. Ehlert says. The orthopedists are focused on their elective practices, and “they’re very happy with that. So their life is much better; their elective practice has actually gotten busier because they’re not having to leave space open for all the trauma from the ER. So I think all around it’s been very successful.”
The hospital has added a general surgery hospitalist program, which is doing well, too. “They’re much busier,” he says. “They’re really taking a load off the general surgeons in town.”
Dr. Maa, assistant professor and director of the surgical hospitalist program at University of California San Francisco Medical Center, says growth in his field is fueled by the ever-growing crises in emergency departments.
“Most hospitals critically depend on a general surgical service,” he said. “If you can’t keep a panel of general surgeons to take call, you’re probably going to have to close your emergency room.”
Dr. Maa, who founded UCSF’s surgical hospitalist program in 2005, explained how the terms “surgicalist” and “acute-care surgeon” have come to represent the concept of a dedicated emergency surgeon, whether it be in trauma or in general surgery. “It really does parallel the medical hospitalist model,” he says, adding that his field has had to overcome doubts about scheduling and patient safety.
“The danger in each of these specialty programs is to become too much of the silo mentality, to focus on their own discipline,” he adds. “We need to work across specialties, we need to collaborate, we need to find ways of utilizing the precious existing resources for emergency care, and make certain that the needs of society are met. Society places trust in doctors, hospital leaders, to build a system that will care for them when they need it. It’s our ethical obligation to design the safest, best system, with the resources that we have.”
Jason Carris is editor of The Hospitalist.
Safer patients, improved quality of care, innovative uses of resources, increased job satisfaction: Those themes threaded their way through presentation after presentation at “Creating the Hospital of the Future: The Implications for Hospital-Focused Physician Practice,” a one-day meeting of hospitalist leaders and hospital administrators following the annual Health Forum/AHA Leadership Summit on July 21 in San Francisco.
The five-hour presentation to about 80 hospital CEOs, chief financial officers, and chief medical officers focused on the ever-expanding roles of subspecialty hospitalists and how subspecialty hospitalist programs can help administrators solve multiple challenges in an era of healthcare reform.
“When I started in 2007, I could only identify 15 hospitals in the United States with OB hospitalist programs. And now we know of 169, and the nation is adding one or two new programs a month,” says Rob Olson, MD, an OB/GYN hospitalistor “laborist”at PeaceHealth St. Joseph Medical Center in Bellingham, Wash., and editor of ObGynHospitalist.com. “If you think about it, if you can make it safer for women in labor, then duh! Why wouldn’t you want to do that?
“The finances work best at a hospital that delivers more than 2,000 babies a year. But many small hospitals, like those doing 800 or 1,000 deliveries a year, have programs because it makes it safer for women in labor,” he says. “Therefore, even though it might be more expensive at those lower numbers, it’s worth it. It’s the right thing to do.”
The July meeting was the second time SHM gathered stakeholders to discuss the growth of specialty hospitalists; a similar panel of experts convened last November in Las Vegas. Most at the San Francisco meeting recognized the upward trend in such HM-focused subspecialties as neurology, orthopedics, obstetrics, and general surgery, according to John Nelson, MD, MHM, organizer of the focused-practice meetings.
“Most people in healthcare feel like this is going to continue and intensify,” says Dr. Nelson, cofounder and past president of SHM, medical director of the hospitalist practice at Overlake Hospital Medical Center, Bellevue, Wash., and practice-management columnist for The Hospitalist. “There is consensus that each specialty can learn from the other about how they organize their practice and approach their work. There was consensus that we don’t have a lot of research data about what this means for things like cost of care, quality of care, patient experience, physician career longevity. So we need to encourage people to begin to study those things, and what this means for the stakeholders in healthcare.”
Four subspecialty hospitalists—neurohospitalist David Likosky, MD, SFHM, surgicalist John Maa, MD, orthopedic hospitalist Kurt Ehlert, MD, and Dr. Olson, the laborist—took part in a 90-minute panel discussion in which they explained their practice models and fielded questions from the audience.
“My message was that there are a large number of people doing this now—we estimate there are between 600 and 700 neurohospitalists nationally—and that the model holds a lot of promise. There’s not a huge amount of data right now on outcomes and other metrics, but we’re starting to see that data,” said Dr. Likosky, medical director of the Evergreen Neuroscience Institute in Kirkland, Wash., and co-founder of the Neurohospitalist Society (neurohospitalistsociety.org). “The neurohospitalist model is a good solution to many of the problems that hospitals are facing now.”
—Kurt Ehlert, MD, medical director, Orthopedic Hospitalists of New Bern, national director for orthopedic services, Delphi Healthcare Partners, Morrisville, N.C.
Dr. Ehlert, director of orthopaedic services, Orthopeadic Hospitalists of New Bern (N.C.), and national orthopaedic medical director of Delphi of TEAMHealth, says subspecialty HM programs offer hospitals a “great chance of improving quality and patient safety over what they have currently, even if they have their emergency room covered.”
“I think hospitalists who are subspecialists in trauma can provide a bump up in productivity, safety, reliability,” he says. “Results will be better. I think it will be less expensive. [I told them] that there is an option out there that can benefit them in all of the various key ways that groups are looking at right now.”
Dr. Ehlert’s ortho-hospitalist group formed when the 300-bed hospital in New Bern encountered a manpower issue not unfamiliar to hospitals across the country. The bylaws of the medical staff allowed subspecialists to stop taking call when they turned 55, and four of the seven orthopedists aged out.
“Three doctors taking all the call is not really sustainable for them in their private practices,” Dr. Ehlert said. “So they looked at various options, came to us, and we started in December of 2009. It has been very successful, according to the administration. They love us being there. The emergency room is very happy with our responsiveness. I think our results have been very good.”
The new arrangement is a win-win, Dr. Ehlert says. The orthopedists are focused on their elective practices, and “they’re very happy with that. So their life is much better; their elective practice has actually gotten busier because they’re not having to leave space open for all the trauma from the ER. So I think all around it’s been very successful.”
The hospital has added a general surgery hospitalist program, which is doing well, too. “They’re much busier,” he says. “They’re really taking a load off the general surgeons in town.”
Dr. Maa, assistant professor and director of the surgical hospitalist program at University of California San Francisco Medical Center, says growth in his field is fueled by the ever-growing crises in emergency departments.
“Most hospitals critically depend on a general surgical service,” he said. “If you can’t keep a panel of general surgeons to take call, you’re probably going to have to close your emergency room.”
Dr. Maa, who founded UCSF’s surgical hospitalist program in 2005, explained how the terms “surgicalist” and “acute-care surgeon” have come to represent the concept of a dedicated emergency surgeon, whether it be in trauma or in general surgery. “It really does parallel the medical hospitalist model,” he says, adding that his field has had to overcome doubts about scheduling and patient safety.
“The danger in each of these specialty programs is to become too much of the silo mentality, to focus on their own discipline,” he adds. “We need to work across specialties, we need to collaborate, we need to find ways of utilizing the precious existing resources for emergency care, and make certain that the needs of society are met. Society places trust in doctors, hospital leaders, to build a system that will care for them when they need it. It’s our ethical obligation to design the safest, best system, with the resources that we have.”
Jason Carris is editor of The Hospitalist.
John Nelson: Post-Discharge Calls
There are lots of places to learn methods to improve patient satisfaction, including my thoughts from the January 2009 issue. Run an Internet search on “improve patient satisfaction” to get a huge number of articles, many of which have useful information and inspiration.
If you’re in a high-functioning hospitalist group, you’ve already read a lot on the topic, listened to presentations by someone at your hospital and elsewhere, and reliably reported and analyzed satisfaction survey results including HCAHPS questions and others. Maybe you’ve even engaged a consultant to help.
You might already have in place a number of strategies, such as reliably providing a business card with your photo, always sitting down in the patient’s room, asking “Is there anything else I can do?” before ending your time with a patient, etc. You’re doing all these things and more, but perhaps you’ve barely moved the needle on your satisfaction scores.
Despite your efforts, I bet your hospitalist group’s aggregate score is among the lowest of any physician group at your hospital.
You’re not alone.
What can you do about this?
High-Value Strategy: Phoning Patients after Discharge
I’m lucky enough to practice with some of the smartest, most professional, and most personable hospitalists you could ever meet. Yet our satisfaction scores are among the lowest for physicians at our hospital. Despite all of the improvement strategies we put in place over the last few years, our scores have barely budged. But that all changed once we instituted a formal program of phoning patients after discharge. That produced the largest uptick in our scores we’ve ever seen.
I can’t guarantee that our results are generalizable. But I have all the anecdotal information I need to be willing to invest the resources to make the calls. They improve scores. Likely more than any other single strategy. And they seem to have a positive effect on all survey questions, from how well the doctor explained things (nearly always the lowest of the HCAHPS scores for hospitalists) to the patient’s opinion of the hospital food.
Though initially resistant to expending the time and energy to make the calls, most in our group have said that they regularly feel really gratified by the response they get from patients or families. I think it is much better if a hospitalist who cared for the patient makes the calls, and I suspect (I have no proof) that calls made by a nurse or clerk are much less effective at improving patient satisfaction. And the call can serve as a valuable clinical encounter to briefly troubleshoot a problem or review a test result that was pending at discharge.
Simple Strategies
- More than 80% of these calls should last less than three minutes. Most patients or family members will report things are going OK and thank you profusely for the call. “No doctor has ever called before,” many will say. “Can we get you the next time Mom is hospitalized?”
- You could reduce the number of calls needed if you limit them to patients eligible for a survey; this typically is only about half of a hospitalist’s patient census. For example, patients on observation status and those discharged somewhere other than to home (e.g. to a skilled-nursing facility) are not eligible for a survey.
- It’s usually best not to tell a patient or family to expect the call. Surprising them makes them more delighted when you do call, and a patient told to expect a call but doesn’t get one will be less satisfied than if never told to expect it. Best if no one at the hospital knows you’re making the calls, because someone might brag about you and tell the patient to expect the call.
- For patients seen by several hospitalists, decide ahead of time which doctor makes the call. The doctor who discharged the patient is probably the simplest protocol.
- Develop a system to track patients who have been discharged. Every morning, we get a printout of all patients discharged the prior day. We try to call all patients the day after discharge to ensure that we reach them before they’ve had a chance to complete a satisfaction survey and before the discharging doctor rotates off.
- Develop a protocol to document the calls. Calls that lead to any new advice or therapies (e.g. see your primary-care physician sooner than planned) must be documented in the medical record, e.g., by dictating an addendum to the discharge summary. Don’t let the system get too complicated or keep you from making the calls.
- Use your judgment about whether to call the patient or just call a family member directly; it’s often better to do the latter.
- If you reach a voicemail (about 50% of the calls I make), leave a message and don’t keep calling back to reach a person.
Sample Scripts
Here are some simple scripts to use for post-discharge calls. If you reach the patient or family member:
- “This is Dr. X from Superior Hospital. I was just thinking about you/your mother/your father and wanted to know how things have gone since you/she/he left the hospital.”
- Ask about something related to the reason for their stay. “How is your appetite?” or “You haven’t had any more fever, have you?” or “Have you made your appointment with Dr. PCP yet?”
- “I hope things go really well for you, but if you ever need the hospital again, we’d be happy to care for you at Superior Hospital.”
If you get a voicemail:
- “This is Dr. X from Superior Hospital. I’ve been thinking about you/your mother/father since you/she/he left the hospital, and I am calling just to check on how things are going.” (For HIPPA reasons, don’t use the patient’s name when leaving a voicemail.)
- Mention some medical concern specific to the patient, e.g., “Your culture test turned out OK and I hope you’ve been able to get the antibiotic I prescribed.”
- “You don’t need to call me back, but if you have questions or want to provide an update, I can be reached at 555-123-4567.” (It’s very important to include this last sentence and a number where you can be reached. If omitted, many patients/families will think you must have called to convey something really important and will be distressed until able to reach you.)
Dr. Nelson has been a practicing hospitalist since 1988. He is co-founder and past president of SHM, and principal in Nelson Flores Hospital Medicine Consultants. He is co-director for SHM’s “Best Practices in Managing a Hospital Medicine Program” course.
There are lots of places to learn methods to improve patient satisfaction, including my thoughts from the January 2009 issue. Run an Internet search on “improve patient satisfaction” to get a huge number of articles, many of which have useful information and inspiration.
If you’re in a high-functioning hospitalist group, you’ve already read a lot on the topic, listened to presentations by someone at your hospital and elsewhere, and reliably reported and analyzed satisfaction survey results including HCAHPS questions and others. Maybe you’ve even engaged a consultant to help.
You might already have in place a number of strategies, such as reliably providing a business card with your photo, always sitting down in the patient’s room, asking “Is there anything else I can do?” before ending your time with a patient, etc. You’re doing all these things and more, but perhaps you’ve barely moved the needle on your satisfaction scores.
Despite your efforts, I bet your hospitalist group’s aggregate score is among the lowest of any physician group at your hospital.
You’re not alone.
What can you do about this?
High-Value Strategy: Phoning Patients after Discharge
I’m lucky enough to practice with some of the smartest, most professional, and most personable hospitalists you could ever meet. Yet our satisfaction scores are among the lowest for physicians at our hospital. Despite all of the improvement strategies we put in place over the last few years, our scores have barely budged. But that all changed once we instituted a formal program of phoning patients after discharge. That produced the largest uptick in our scores we’ve ever seen.
I can’t guarantee that our results are generalizable. But I have all the anecdotal information I need to be willing to invest the resources to make the calls. They improve scores. Likely more than any other single strategy. And they seem to have a positive effect on all survey questions, from how well the doctor explained things (nearly always the lowest of the HCAHPS scores for hospitalists) to the patient’s opinion of the hospital food.
Though initially resistant to expending the time and energy to make the calls, most in our group have said that they regularly feel really gratified by the response they get from patients or families. I think it is much better if a hospitalist who cared for the patient makes the calls, and I suspect (I have no proof) that calls made by a nurse or clerk are much less effective at improving patient satisfaction. And the call can serve as a valuable clinical encounter to briefly troubleshoot a problem or review a test result that was pending at discharge.
Simple Strategies
- More than 80% of these calls should last less than three minutes. Most patients or family members will report things are going OK and thank you profusely for the call. “No doctor has ever called before,” many will say. “Can we get you the next time Mom is hospitalized?”
- You could reduce the number of calls needed if you limit them to patients eligible for a survey; this typically is only about half of a hospitalist’s patient census. For example, patients on observation status and those discharged somewhere other than to home (e.g. to a skilled-nursing facility) are not eligible for a survey.
- It’s usually best not to tell a patient or family to expect the call. Surprising them makes them more delighted when you do call, and a patient told to expect a call but doesn’t get one will be less satisfied than if never told to expect it. Best if no one at the hospital knows you’re making the calls, because someone might brag about you and tell the patient to expect the call.
- For patients seen by several hospitalists, decide ahead of time which doctor makes the call. The doctor who discharged the patient is probably the simplest protocol.
- Develop a system to track patients who have been discharged. Every morning, we get a printout of all patients discharged the prior day. We try to call all patients the day after discharge to ensure that we reach them before they’ve had a chance to complete a satisfaction survey and before the discharging doctor rotates off.
- Develop a protocol to document the calls. Calls that lead to any new advice or therapies (e.g. see your primary-care physician sooner than planned) must be documented in the medical record, e.g., by dictating an addendum to the discharge summary. Don’t let the system get too complicated or keep you from making the calls.
- Use your judgment about whether to call the patient or just call a family member directly; it’s often better to do the latter.
- If you reach a voicemail (about 50% of the calls I make), leave a message and don’t keep calling back to reach a person.
Sample Scripts
Here are some simple scripts to use for post-discharge calls. If you reach the patient or family member:
- “This is Dr. X from Superior Hospital. I was just thinking about you/your mother/your father and wanted to know how things have gone since you/she/he left the hospital.”
- Ask about something related to the reason for their stay. “How is your appetite?” or “You haven’t had any more fever, have you?” or “Have you made your appointment with Dr. PCP yet?”
- “I hope things go really well for you, but if you ever need the hospital again, we’d be happy to care for you at Superior Hospital.”
If you get a voicemail:
- “This is Dr. X from Superior Hospital. I’ve been thinking about you/your mother/father since you/she/he left the hospital, and I am calling just to check on how things are going.” (For HIPPA reasons, don’t use the patient’s name when leaving a voicemail.)
- Mention some medical concern specific to the patient, e.g., “Your culture test turned out OK and I hope you’ve been able to get the antibiotic I prescribed.”
- “You don’t need to call me back, but if you have questions or want to provide an update, I can be reached at 555-123-4567.” (It’s very important to include this last sentence and a number where you can be reached. If omitted, many patients/families will think you must have called to convey something really important and will be distressed until able to reach you.)
Dr. Nelson has been a practicing hospitalist since 1988. He is co-founder and past president of SHM, and principal in Nelson Flores Hospital Medicine Consultants. He is co-director for SHM’s “Best Practices in Managing a Hospital Medicine Program” course.
There are lots of places to learn methods to improve patient satisfaction, including my thoughts from the January 2009 issue. Run an Internet search on “improve patient satisfaction” to get a huge number of articles, many of which have useful information and inspiration.
If you’re in a high-functioning hospitalist group, you’ve already read a lot on the topic, listened to presentations by someone at your hospital and elsewhere, and reliably reported and analyzed satisfaction survey results including HCAHPS questions and others. Maybe you’ve even engaged a consultant to help.
You might already have in place a number of strategies, such as reliably providing a business card with your photo, always sitting down in the patient’s room, asking “Is there anything else I can do?” before ending your time with a patient, etc. You’re doing all these things and more, but perhaps you’ve barely moved the needle on your satisfaction scores.
Despite your efforts, I bet your hospitalist group’s aggregate score is among the lowest of any physician group at your hospital.
You’re not alone.
What can you do about this?
High-Value Strategy: Phoning Patients after Discharge
I’m lucky enough to practice with some of the smartest, most professional, and most personable hospitalists you could ever meet. Yet our satisfaction scores are among the lowest for physicians at our hospital. Despite all of the improvement strategies we put in place over the last few years, our scores have barely budged. But that all changed once we instituted a formal program of phoning patients after discharge. That produced the largest uptick in our scores we’ve ever seen.
I can’t guarantee that our results are generalizable. But I have all the anecdotal information I need to be willing to invest the resources to make the calls. They improve scores. Likely more than any other single strategy. And they seem to have a positive effect on all survey questions, from how well the doctor explained things (nearly always the lowest of the HCAHPS scores for hospitalists) to the patient’s opinion of the hospital food.
Though initially resistant to expending the time and energy to make the calls, most in our group have said that they regularly feel really gratified by the response they get from patients or families. I think it is much better if a hospitalist who cared for the patient makes the calls, and I suspect (I have no proof) that calls made by a nurse or clerk are much less effective at improving patient satisfaction. And the call can serve as a valuable clinical encounter to briefly troubleshoot a problem or review a test result that was pending at discharge.
Simple Strategies
- More than 80% of these calls should last less than three minutes. Most patients or family members will report things are going OK and thank you profusely for the call. “No doctor has ever called before,” many will say. “Can we get you the next time Mom is hospitalized?”
- You could reduce the number of calls needed if you limit them to patients eligible for a survey; this typically is only about half of a hospitalist’s patient census. For example, patients on observation status and those discharged somewhere other than to home (e.g. to a skilled-nursing facility) are not eligible for a survey.
- It’s usually best not to tell a patient or family to expect the call. Surprising them makes them more delighted when you do call, and a patient told to expect a call but doesn’t get one will be less satisfied than if never told to expect it. Best if no one at the hospital knows you’re making the calls, because someone might brag about you and tell the patient to expect the call.
- For patients seen by several hospitalists, decide ahead of time which doctor makes the call. The doctor who discharged the patient is probably the simplest protocol.
- Develop a system to track patients who have been discharged. Every morning, we get a printout of all patients discharged the prior day. We try to call all patients the day after discharge to ensure that we reach them before they’ve had a chance to complete a satisfaction survey and before the discharging doctor rotates off.
- Develop a protocol to document the calls. Calls that lead to any new advice or therapies (e.g. see your primary-care physician sooner than planned) must be documented in the medical record, e.g., by dictating an addendum to the discharge summary. Don’t let the system get too complicated or keep you from making the calls.
- Use your judgment about whether to call the patient or just call a family member directly; it’s often better to do the latter.
- If you reach a voicemail (about 50% of the calls I make), leave a message and don’t keep calling back to reach a person.
Sample Scripts
Here are some simple scripts to use for post-discharge calls. If you reach the patient or family member:
- “This is Dr. X from Superior Hospital. I was just thinking about you/your mother/your father and wanted to know how things have gone since you/she/he left the hospital.”
- Ask about something related to the reason for their stay. “How is your appetite?” or “You haven’t had any more fever, have you?” or “Have you made your appointment with Dr. PCP yet?”
- “I hope things go really well for you, but if you ever need the hospital again, we’d be happy to care for you at Superior Hospital.”
If you get a voicemail:
- “This is Dr. X from Superior Hospital. I’ve been thinking about you/your mother/father since you/she/he left the hospital, and I am calling just to check on how things are going.” (For HIPPA reasons, don’t use the patient’s name when leaving a voicemail.)
- Mention some medical concern specific to the patient, e.g., “Your culture test turned out OK and I hope you’ve been able to get the antibiotic I prescribed.”
- “You don’t need to call me back, but if you have questions or want to provide an update, I can be reached at 555-123-4567.” (It’s very important to include this last sentence and a number where you can be reached. If omitted, many patients/families will think you must have called to convey something really important and will be distressed until able to reach you.)
Dr. Nelson has been a practicing hospitalist since 1988. He is co-founder and past president of SHM, and principal in Nelson Flores Hospital Medicine Consultants. He is co-director for SHM’s “Best Practices in Managing a Hospital Medicine Program” course.
Establish Rules of Engagement before Covering Ortho Inpatients
One of our providers wants to use adult hospitalists for coverage of inpatient orthopedic surgery patients. Is this acceptable practice? Are there qualifiers?
—Libby Gardner
Dr. Hospitalist responds:
Let’s see how far we can tackle this open-ended question. There has been lots of discussion on the topic of comanagement in the past by people eminently more qualified than I am. Still, it never hurts to take a fresh look at things.
For one, on the subject of admissions, I am a firm believer that hospitalists should admit all adult hip fractures. The overwhelming majority of the time, these patients are elderly with comorbid conditions. Sure, they are going to get their hip fixed, because the alternative usually is unacceptable, but some thought needs to go into the process.
The orthopedic surgeon sees a hip that needs fixing and not much else. When such issues as renal failure, afib, congestive heart failure, prior DVT, dementia, and all the other common conditions are present, we as adult hospitalists should take charge of the case. That is the best way to ensure that the patient receives optimal medical care and the documentation that goes along with it. I love our orthopedic surgeons, but I don’t want them primarily admitting, managing, and discharging my elderly patients. Let the surgeon do what they do best—operate—and leave the rest to us as hospitalists.
On the subject of orthopedic trauma, I take the exact opposite approach—this is not something where we have daily expertise. A young, healthy patient with trauma should be admitted by the orthopedic service; that patient population’s complications are much more likely to be directly related to their trauma.
When it comes to elective surgery when the admitting surgeon (orthopedic or otherwise) wants the help of a hospitalist, then I think it is of paramount importance to establish clear “rules of engagement” (see “The Comanagement Conundrum,” April 2011, p. 1). I think with good expectations, you can have a fantastic relationship with your surgeons. Without them, it becomes a nightmare. As a real-life example, here are my HM group’s rules for elective orthopedic surgery:
- Orthopedics handles all pain medications and VTE prophylaxis, including discharge prescriptions;
- Medicine does the admission and medication reconciliation (“med rec”) at discharge;
- There is shared discussion on the need for transfusion; and
- There is shared discussion on the need for VTE prophylaxis when a patient already is on chronic anticoagulation.
We do not vary from this protocol. I never adjust a patient’s pain medications—even the floor nurses know this. Because I’m doing the admit and med rec, it also means that the patient doesn’t have their HCTZ continued after 600cc of EBL and spinal anesthesia. It works because the rules are clear and the communication is consistent.
This does not mean that we cover the orthopedic service at night; they are equally responsible for their patients under the items outlined above. In my view—and this might sound simplistic—the surgeon caused the post-op pain, so they should be responsible for managing it. With regard to VTE prophylaxis, I might take a more nuanced view, but for our surgeons, they own the wound and the post-op follow-up, so they get the choice on what agent to use.
Would I accept an arrangement in which I covered all the orthopedic issues out of regular hours? Nope—not when they have primary responsibility for the case; they should always be directly available to the nurse. I think that anything else would be a system ripe for abuse.
Our exact rules will not work for every situation, but I would strongly encourage the two basic tenets from above: No. 1, the hospitalist should primarily admit and manage elderly hip fractures, and No. 2, there should be clear rules of engagement with your orthopedic or surgery group. It’s a discussion worth having during daylight hours, because trying to figure out the rules at 3 in the morning rarely ends well.
One of our providers wants to use adult hospitalists for coverage of inpatient orthopedic surgery patients. Is this acceptable practice? Are there qualifiers?
—Libby Gardner
Dr. Hospitalist responds:
Let’s see how far we can tackle this open-ended question. There has been lots of discussion on the topic of comanagement in the past by people eminently more qualified than I am. Still, it never hurts to take a fresh look at things.
For one, on the subject of admissions, I am a firm believer that hospitalists should admit all adult hip fractures. The overwhelming majority of the time, these patients are elderly with comorbid conditions. Sure, they are going to get their hip fixed, because the alternative usually is unacceptable, but some thought needs to go into the process.
The orthopedic surgeon sees a hip that needs fixing and not much else. When such issues as renal failure, afib, congestive heart failure, prior DVT, dementia, and all the other common conditions are present, we as adult hospitalists should take charge of the case. That is the best way to ensure that the patient receives optimal medical care and the documentation that goes along with it. I love our orthopedic surgeons, but I don’t want them primarily admitting, managing, and discharging my elderly patients. Let the surgeon do what they do best—operate—and leave the rest to us as hospitalists.
On the subject of orthopedic trauma, I take the exact opposite approach—this is not something where we have daily expertise. A young, healthy patient with trauma should be admitted by the orthopedic service; that patient population’s complications are much more likely to be directly related to their trauma.
When it comes to elective surgery when the admitting surgeon (orthopedic or otherwise) wants the help of a hospitalist, then I think it is of paramount importance to establish clear “rules of engagement” (see “The Comanagement Conundrum,” April 2011, p. 1). I think with good expectations, you can have a fantastic relationship with your surgeons. Without them, it becomes a nightmare. As a real-life example, here are my HM group’s rules for elective orthopedic surgery:
- Orthopedics handles all pain medications and VTE prophylaxis, including discharge prescriptions;
- Medicine does the admission and medication reconciliation (“med rec”) at discharge;
- There is shared discussion on the need for transfusion; and
- There is shared discussion on the need for VTE prophylaxis when a patient already is on chronic anticoagulation.
We do not vary from this protocol. I never adjust a patient’s pain medications—even the floor nurses know this. Because I’m doing the admit and med rec, it also means that the patient doesn’t have their HCTZ continued after 600cc of EBL and spinal anesthesia. It works because the rules are clear and the communication is consistent.
This does not mean that we cover the orthopedic service at night; they are equally responsible for their patients under the items outlined above. In my view—and this might sound simplistic—the surgeon caused the post-op pain, so they should be responsible for managing it. With regard to VTE prophylaxis, I might take a more nuanced view, but for our surgeons, they own the wound and the post-op follow-up, so they get the choice on what agent to use.
Would I accept an arrangement in which I covered all the orthopedic issues out of regular hours? Nope—not when they have primary responsibility for the case; they should always be directly available to the nurse. I think that anything else would be a system ripe for abuse.
Our exact rules will not work for every situation, but I would strongly encourage the two basic tenets from above: No. 1, the hospitalist should primarily admit and manage elderly hip fractures, and No. 2, there should be clear rules of engagement with your orthopedic or surgery group. It’s a discussion worth having during daylight hours, because trying to figure out the rules at 3 in the morning rarely ends well.
One of our providers wants to use adult hospitalists for coverage of inpatient orthopedic surgery patients. Is this acceptable practice? Are there qualifiers?
—Libby Gardner
Dr. Hospitalist responds:
Let’s see how far we can tackle this open-ended question. There has been lots of discussion on the topic of comanagement in the past by people eminently more qualified than I am. Still, it never hurts to take a fresh look at things.
For one, on the subject of admissions, I am a firm believer that hospitalists should admit all adult hip fractures. The overwhelming majority of the time, these patients are elderly with comorbid conditions. Sure, they are going to get their hip fixed, because the alternative usually is unacceptable, but some thought needs to go into the process.
The orthopedic surgeon sees a hip that needs fixing and not much else. When such issues as renal failure, afib, congestive heart failure, prior DVT, dementia, and all the other common conditions are present, we as adult hospitalists should take charge of the case. That is the best way to ensure that the patient receives optimal medical care and the documentation that goes along with it. I love our orthopedic surgeons, but I don’t want them primarily admitting, managing, and discharging my elderly patients. Let the surgeon do what they do best—operate—and leave the rest to us as hospitalists.
On the subject of orthopedic trauma, I take the exact opposite approach—this is not something where we have daily expertise. A young, healthy patient with trauma should be admitted by the orthopedic service; that patient population’s complications are much more likely to be directly related to their trauma.
When it comes to elective surgery when the admitting surgeon (orthopedic or otherwise) wants the help of a hospitalist, then I think it is of paramount importance to establish clear “rules of engagement” (see “The Comanagement Conundrum,” April 2011, p. 1). I think with good expectations, you can have a fantastic relationship with your surgeons. Without them, it becomes a nightmare. As a real-life example, here are my HM group’s rules for elective orthopedic surgery:
- Orthopedics handles all pain medications and VTE prophylaxis, including discharge prescriptions;
- Medicine does the admission and medication reconciliation (“med rec”) at discharge;
- There is shared discussion on the need for transfusion; and
- There is shared discussion on the need for VTE prophylaxis when a patient already is on chronic anticoagulation.
We do not vary from this protocol. I never adjust a patient’s pain medications—even the floor nurses know this. Because I’m doing the admit and med rec, it also means that the patient doesn’t have their HCTZ continued after 600cc of EBL and spinal anesthesia. It works because the rules are clear and the communication is consistent.
This does not mean that we cover the orthopedic service at night; they are equally responsible for their patients under the items outlined above. In my view—and this might sound simplistic—the surgeon caused the post-op pain, so they should be responsible for managing it. With regard to VTE prophylaxis, I might take a more nuanced view, but for our surgeons, they own the wound and the post-op follow-up, so they get the choice on what agent to use.
Would I accept an arrangement in which I covered all the orthopedic issues out of regular hours? Nope—not when they have primary responsibility for the case; they should always be directly available to the nurse. I think that anything else would be a system ripe for abuse.
Our exact rules will not work for every situation, but I would strongly encourage the two basic tenets from above: No. 1, the hospitalist should primarily admit and manage elderly hip fractures, and No. 2, there should be clear rules of engagement with your orthopedic or surgery group. It’s a discussion worth having during daylight hours, because trying to figure out the rules at 3 in the morning rarely ends well.
TH.org Survey: VTE Prophylaxis a Gray Area for Some Hospitalists
Six out of 7 respondents to an unscientific survey at the-hospitalist.org correctly answered the question "Which patient being discharged DOES NOT require VTE prophylaxis?" while 14% of respondents answered incorrectly, according to veteran hospitalists who reviewed survey results.
Nearly 200 people responded to the survey, which provided the following answers:
• Hip/knee replacement patients;
• Hip-fracture surgery patients;
• Patients with advanced solid tumors;
• Patients with congestive heart failure; and
• Immobile or bed-bound patients.
The survey ran from June 29 to July 27 and was not limited to physicians. Sixty-six percent of respondents chose "patients with congestive heart failure" as their answer; 20% of respondents chose "patients with advanced solid tumors." Both types of discharged patients do not require VTE prophylaxis, according to the new American College of Chest Physicians (ACCP) guidelines, says Joseph Ming Wah Li, MD, SFHM, FACP, past president of SHM, associate professor of medicine at Harvard Medical School, and hospitalist at Beth Israel Deaconess Medical Center in Boston.
"Hospitalists are familiar with the fact that patients with advanced solid tumors without additional risk factors do not need extended prophylaxis, as per the guidelines put out by ACCP," Dr. Li says. "And patients with congestive heart failure being discharged from the hospital do not need extended thromboprophylaxis, either."
As for the 14% of respondents who chose incorrectly, Gregory Maynard, MD, MSc, SFHM, health sciences professor of medicine at the University of California at San Diego, says extended prophylaxis "may not be on their radar."
"Hospitalists are still struggling to get a consistent approach to inpatient VTE prophylaxis, especially since the ACCP 9 guidelines changed methodologies and provided weaker support for prophylaxis," says Dr. Maynard, senior vice president of SHM's Center for Hospital Innovation and Improvement.
For more information, check out our "Key Clinical Question" outlining VTE prophylaxis guidelines.
Six out of 7 respondents to an unscientific survey at the-hospitalist.org correctly answered the question "Which patient being discharged DOES NOT require VTE prophylaxis?" while 14% of respondents answered incorrectly, according to veteran hospitalists who reviewed survey results.
Nearly 200 people responded to the survey, which provided the following answers:
• Hip/knee replacement patients;
• Hip-fracture surgery patients;
• Patients with advanced solid tumors;
• Patients with congestive heart failure; and
• Immobile or bed-bound patients.
The survey ran from June 29 to July 27 and was not limited to physicians. Sixty-six percent of respondents chose "patients with congestive heart failure" as their answer; 20% of respondents chose "patients with advanced solid tumors." Both types of discharged patients do not require VTE prophylaxis, according to the new American College of Chest Physicians (ACCP) guidelines, says Joseph Ming Wah Li, MD, SFHM, FACP, past president of SHM, associate professor of medicine at Harvard Medical School, and hospitalist at Beth Israel Deaconess Medical Center in Boston.
"Hospitalists are familiar with the fact that patients with advanced solid tumors without additional risk factors do not need extended prophylaxis, as per the guidelines put out by ACCP," Dr. Li says. "And patients with congestive heart failure being discharged from the hospital do not need extended thromboprophylaxis, either."
As for the 14% of respondents who chose incorrectly, Gregory Maynard, MD, MSc, SFHM, health sciences professor of medicine at the University of California at San Diego, says extended prophylaxis "may not be on their radar."
"Hospitalists are still struggling to get a consistent approach to inpatient VTE prophylaxis, especially since the ACCP 9 guidelines changed methodologies and provided weaker support for prophylaxis," says Dr. Maynard, senior vice president of SHM's Center for Hospital Innovation and Improvement.
For more information, check out our "Key Clinical Question" outlining VTE prophylaxis guidelines.
Six out of 7 respondents to an unscientific survey at the-hospitalist.org correctly answered the question "Which patient being discharged DOES NOT require VTE prophylaxis?" while 14% of respondents answered incorrectly, according to veteran hospitalists who reviewed survey results.
Nearly 200 people responded to the survey, which provided the following answers:
• Hip/knee replacement patients;
• Hip-fracture surgery patients;
• Patients with advanced solid tumors;
• Patients with congestive heart failure; and
• Immobile or bed-bound patients.
The survey ran from June 29 to July 27 and was not limited to physicians. Sixty-six percent of respondents chose "patients with congestive heart failure" as their answer; 20% of respondents chose "patients with advanced solid tumors." Both types of discharged patients do not require VTE prophylaxis, according to the new American College of Chest Physicians (ACCP) guidelines, says Joseph Ming Wah Li, MD, SFHM, FACP, past president of SHM, associate professor of medicine at Harvard Medical School, and hospitalist at Beth Israel Deaconess Medical Center in Boston.
"Hospitalists are familiar with the fact that patients with advanced solid tumors without additional risk factors do not need extended prophylaxis, as per the guidelines put out by ACCP," Dr. Li says. "And patients with congestive heart failure being discharged from the hospital do not need extended thromboprophylaxis, either."
As for the 14% of respondents who chose incorrectly, Gregory Maynard, MD, MSc, SFHM, health sciences professor of medicine at the University of California at San Diego, says extended prophylaxis "may not be on their radar."
"Hospitalists are still struggling to get a consistent approach to inpatient VTE prophylaxis, especially since the ACCP 9 guidelines changed methodologies and provided weaker support for prophylaxis," says Dr. Maynard, senior vice president of SHM's Center for Hospital Innovation and Improvement.
For more information, check out our "Key Clinical Question" outlining VTE prophylaxis guidelines.