User login
Patient and Support Person Satisfaction Following a Whole Health-Informed Interdisciplinary Pain Team Meeting
Patient and Support Person Satisfaction Following a Whole Health-Informed Interdisciplinary Pain Team Meeting
Chronic pain is one of the most prevalent public health concerns in the United States, affecting > 51 million adults with about $500 billion in health care costs.1 Military veterans are among the most vulnerable subpopulations, with 65% of veterans reporting chronic pain in the last 3 months.2 Chronic pain is complex, affecting the biopsychosocial-spiritual levels of human health, and requires multimodal and comprehensive treatment approaches.3 Hence, chronic pain treatment can be best delivered via interdisciplinary teams (IDTs) that use a patient-centered approach.4,5
The Veterans Healthcare Administration (VHA) is a leader in developing and delivering interdisciplinary pain care.6,7 VHA Directive 2009-053 requires every US Department of Veterans Affairs (VA) medical center to offer an IDT for chronic pain. However, VHA and non-VHA IDT programs vary significantly.8-11 A recent systematic review found a median of 5 disciplines included on IDTs (range, 2-8), and program content often included exercise and education; only 11% of included IDTs met simultaneously with patients.11 The heterogeneity of IDT programs has made determining best practices challenging.8,11 The Initiative on Methods, Measurement, and Pain Assessment in Clinical Trials has denoted several core measures and measurement domains that were critical for determining the success of pain management interventions, including patient satisfaction.12,13 Nevertheless, the association of IDTs with high patient satisfaction and improvement in pain measures has been documented.5,11
The VHA has worked to implement the Whole Health System into health care, which considers well-being across physical, behavioral, spiritual, and socioeconomic domains. As such, the Whole Health System involves an interpersonal, team-based approach, “anchored in trusting longitudinal relationships to promote resilience, prevent disease, and restore health.”14 It aligns with the patient’s mission, aspiration, and purpose. Surgeon General VADM Vivek H. Murthy, MD, MBA, recently endorsed this approach.15,16 Other health care systems adopting whole health tend to have higher patient satisfaction, increased access to care, and improved patient-reported outcomes.15 Within the VHA, the Whole Health System has shifted the conversation between clinicians and patients from “What is the matter with you?” to “What is important to you?” while emphasizing a proactive and personalized approach to health care.17 Rather than emphasizing passive modalities such as medications and clinician-led services (eg, interventional pain service), the Whole Health System highlights self-care.3,17 Initial research findings within the VHA have been promising.18-21 Whole health peer coaching calls appear to be an effective approach for veterans diagnosed with PTSD, and the use of whole health services is associated with a decrease in opioid use.19,22 However, there are negligible data on patient experiences after meeting with a whole health-focused pain IDT, and studies to date have focused on urban populations.23 One approach to IDT that has shown promise for other health issues involves a patient meeting simultaneously with all members of the IDT.24-27 With the integration of the Whole Health System and the VHA priorities to provide veterans with the “soonest and best care,” more data are needed on the experiences of patients and support persons with various approaches to IDT pain care.28 This study aimed to evaluate patient and support person experiences with a whole health-focused pain IDT that met simultaneously with the patient and support person during an initial evaluation. This study was approved by the institutional review board at the Salem VA Health Care System (SVAHCS) in Virginia.
Methods
The PREVAIL IDT Track is a clinical program offered at SVAHCS with a whole health-focused approach that involves patients and their support persons meeting simultaneously with a pain IDT. PREVAIL IDT Track is designed to help veterans more effectively self-manage chronic pain (Table 1).6,29 Health care practitioners (HCPs) at SVAHCS recommended that veterans with pain persisting for > 3 months participate in PREVAIL IDT Track. After meeting with an advanced practice clinician for an intake, veterans elected to participate in the PREVAIL IDT Track program and completed the initial 6 weeks of pain education. Veterans were then invited to be evaluated by the pain IDT. A team including HCPs from interventional pain, psychology, pharmacy, nutrition, and physical therapy services met with the veteran for 60 minutes. Veterans were also invited to bring a support person to the IDT initial evaluation.
During the IDT initial evaluation, HCPs inquired about the patient’s mission, aspiration, and purpose (“If you were in less pain, what would you be doing more of?”) and about whole health self-care and wellness factors that may contribute to their chronic pain using the Personal Health Inventory.30,31 Veterans were then invited to select 3 whole health self-care areas to focus on during the 6-month program.3 The IDT HCPs worked with the veteran to establish the treatment plan for the first month in the areas of self-care selected by the patient and made recommendations for additional treatments. If the veteran brought a support person to the IDT initial evaluation, their feedback was elicited throughout and at the end to ensure the final treatment plan and first month’s goals were realistic. At the end of the appointment, the veteran and their support person were asked to complete a program-specific satisfaction survey. The HCPs on the team and the veteran executed the treatment plan developed during the appointment, except for medication prescribing. Recommendations for medication changes are included in clinical notes. Veterans then received 5 monthly coaching calls from a nurse navigator with training in whole health and a 6-month follow-up appointment with the IDT HCPs to discuss a plan for continuity of care.
Participant demographic information was not collected, and participants were not compensated for completing the survey. Veterans in PREVAIL IDT Track are predominantly residents of central Appalachia, White, male, unemployed, have ≥ 1 mental and physical health comorbidity, and have a history of mental health treatment.32 Veterans participating in PREVAIL IDT had a mean age of 57 years, and about 1 in 3 have opioid prescriptions.32
A program-specific 17-question satisfaction survey was developed, which included questions related to satisfaction with previous SVAHCS pain care and staff interactions. To assess the overall impression of the IDT initial evaluation, 3 yes/no questions and a 0 to 10-point scale were used. The 5 remaining open-ended questions allowed participants to give feedback about the IDT initial evaluation.
Data Analysis
A convergent mixed-methods approach was used to evaluate participant satisfaction with the initial IDT evaluation. The study team collected and analyzed quantitative and qualitative survey data and triangulated the findings.33 For quantitative responses, frequencies and means were calculated using Python. For qualitative responses, thematic data analysis was conducted by systematic coding, using inductive methods and allowing themes to emerge. Study team members performed a line-by-line analysis of responses using NVivo to identify important codes and reach a consensus. This study adhered to the Consolidated Criteria for Reporting Qualitative Research and followed the National Institute for Health Care Excellence checklist.34,35
Results
Quantitative Responses
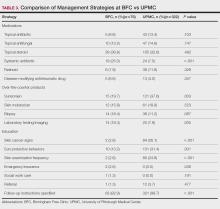
In 2022, 168 veterans completed the initial IDT evaluation, and 144 (85.2%) completed the satisfaction survey and were included in this study. Thirty-two support persons who attended the initial IDT evaluation and completed the survey also were included. Of the 12 quantitative questions, 4 had a 100% completion rate, while 8 had ≤ 3% missing responses. When describing care prior to participating in PREVAIL, participants indicated a mean (SD) response of 4.6 (1.4) with the health care they received at SVAHCS and 4.3 (1.4) with SVAHCS pain management services, both on 6-point scales. All but 2 participants (98.9%) reported always being treated with courtesy and respect by PREVAIL HCPs during the initial IDT evaluation, with a mean (SD) score of 4.0 (0.2) on a 4-point scale. Most respondents (96.6%) reported that PREVAIL HCPs always listened carefully during the initial IDT evaluation, with a mean (SD) 4.0 (0.3) on a 4-point scale. Similarly, 92.6% reported that PREVAIL HCPs explained things clearly during the initial IDT evaluation, with a mean (SD) 3.9 (0.3) on a 4-point scale.
All respondents agreed that PREVAIL HCPs considered veteran preferences and those of their support persons in deciding their health care needs during the initial IDT evaluation, with a mean (SD) 3.7 (0.5) on a 4-point scale. Most respondents left the appointment with a good understanding of their responsibilities for chronic pain management with 99.4% (n = 169) strongly agreeing or agreeing (mean [SD] 3.6 [0.5]). A total of 135 respondents (79.4%) reported they left appointments with written information on their treatment plan. All 170 respondents reported that they would recommend PREVAIL to a friend, and 169 respondents (98.8%) felt that the initial PREVAIL IDT evaluation was a valuable use of time. Eighty-seven respondents (50.9%) rated the initial IDT evaluation as the “best clinical experience possible” with a mean (SD) score of 9.2 (1.1) on a 10-point scale (Table 2).
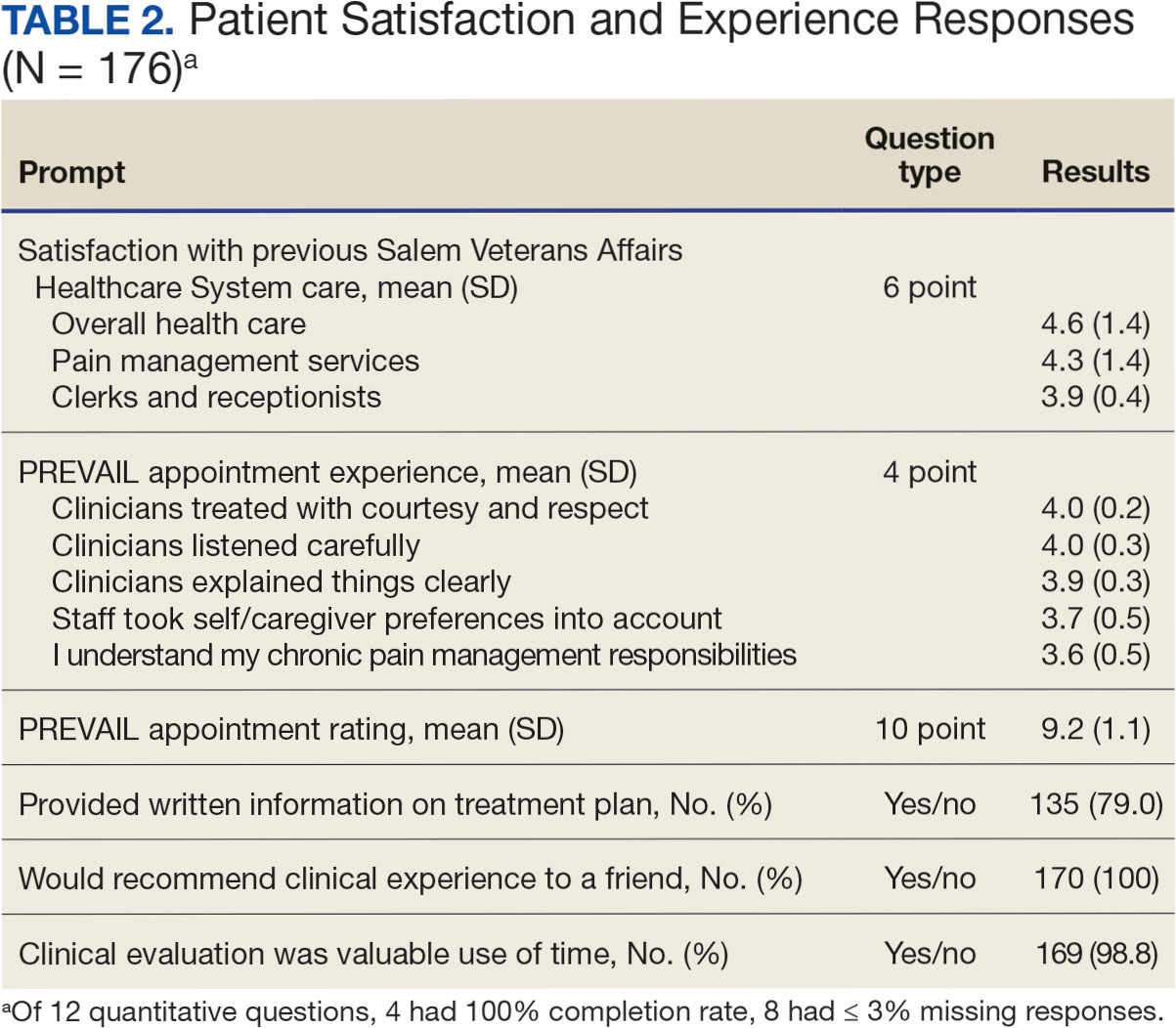
Qualitative Responses
Respondents provided complementary feedback on the program, with many participants stating that they enjoyed every aspect (eAppendix, available at doi:10.12788/fp.0503). In terms of positive aspects of the program, several themes emerged: participants appreciated meeting as an IDT, feeling cared for and listened to, learning more about their pain and ways to manage it, and specific services offered. Thirty-three of 144 respondents wanted longer appointment times. Twenty-two respondents suggested logistics improvements (eg, meeting in a larger room, having a written plan at the end, sending paperwork ahead of time, and later appointment times).
Discussion
Veterans and support persons were satisfied with the initial IDT evaluation for the PREVAIL whole health-focused pain clinical program for veterans predominantly residing in central Appalachia. These satisfaction findings are noteworthy since 20% of this same sample reported dissatisfaction with prior pain services, which could affect engagement and outcomes in pain care. In addition to high satisfaction levels, the PREVAIL IDT model may benefit veterans with limited resources. Rather than needing to attend several individual appointments, the PREVAIL IDT Track provides a 1-stop shop approach that decreases patient burden and barriers to care (eg, travel, transportation, and time) as well as health care system burden. For instance, schedulers need only to make 1 appointment for the veteran rather than several. This approach was highly acceptable to veterans served at SVAHCS and may increase the reach and impact of VHA IDT pain care.
The PREVAIL model may foster rapport with HCPs and encourage an active role in self-managing pain.36,37 Participants noted that their preferences were considered and that they had a good understanding of their responsibilities for managing their chronic pain. This patient-centered approach, emphasizing an active role for the patient, is a hallmark of the VHA Whole Health System and aligns with the overarching PREVAIL IDT Track goal to enhance self-management skills, thus improving functioning through decreased pain interference.14,38-41
Participants in PREVAIL provided substantial open-ended feedback that has contributed to the program’s improvement and may provide information into preferred components of pain IDT programs, particularly for rural veterans. When asked about their favorite component of the initial IDT evaluation, the most emergent theme was meeting simultaneously with HCPs on the IDT. This finding is significant, given that only 11% of IDTs involve direct patient interaction.
Furthermore, unlike most IDTs, PREVAIL IDT includes a dietitian.11,42 IDT programs may benefit from dietitian involvement given the importance of the anti-inflammatory diet on chronic pain.43-46 Participants recommended improvements, (eg, changes to the location and timing, adding a written treatment plan at the end of the appointment, and completing paperwork prior to the appointment) many of which have been addressed. The program now uses validated measures to track progress and comprehensive assessments of pain in response to calls for measurement-based care.13,47 These process improvement suggestions may be instructive for other VA medical centers with rural populations.
Limitations
This study used a program-specific satisfaction survey with open-ended questions to allow for rich responses; however, the survey has not been validated. It also sought to minimize bias by asking participants to give completed surveys to staff members who were not HCPs on the IDT. However, participants’ responses may still have been influenced by this process. Response rate and demographics for support persons were impossible to determine. The results analyzed the responses of veterans and support persons together, which may have skewed the data. Future studies of pain IDT programs should consider analyzing responses from veterans and their support persons separately and identifying factors (eg, demographics or clinical characteristics) that influence the patients’ experiences while participating.
Conclusions
The initial PREVAIL IDT evaluation at SVAHCS is associated with high-levels of satisfaction. These veterans living in rural Appalachia, similar to the 4.4 million rural US veterans, are more likely to encounter barriers to care (eg, drive time, or transportation concerns) and be prescribed opioids.48 These veterans are also at high risk of chronic physical and mental health comorbidities, drug misuse, overdose, and suicide.49,50 Providing veterans in rural communities the opportunity to attend a single appointment with a pain IDT instead of requiring several individual appointments could improve the reach of evidence-based pain care.
This model of meeting simultaneously with all HCPs on the pain IDT may connect all veterans to the most available and best care, something prioritized by the VHA.28 The initial PREVAIL IDT evaluation also utilizes the Personal Health Inventory and the VHA Whole Health System Circle of Health to design patient-centered treatment plans. Integration of the Whole Health System is currently a high priority within VHA. The PREVAIL IDT Track model warrants additional efficacy research.
- Rikard SM, Strahan AE, Schmit KM, Guy GP Jr. Chronic pain among adults - United States, 2019-2021. MMWR Morb Mortal Wkly Rep. 2023;72(15):379-385. doi:10.15585/mmwr.mm7215a1
- Nahin RL. Severe Pain in Veterans: The effect of age and sex, and comparisons with the general population. J Pain. 2017;18(3):247-254. doi:10.1016/j.jpain.2016.10.021
- Courtney RE, Schadegg MJ, Bolton R, Smith S, Harden SM. Using a whole health approach to build biopsychosocial-spiritual personal health plans for veterans with chronic pain. Pain Manag Nurs. 2024;25(1):69-74. doi:10.1016/j.pmn.2023.09.010
- Mackey SC, Pearl RG. Pain management: optimizing patient care through comprehensive, interdisciplinary models and continuous innovations. Anesthesiol Clin. 2023;41(2):xv-xvii. doi:10.1016/j.anclin.2023.03.011
- Gatchel RJ, McGeary DD, McGeary CA, Lippe B. Interdisciplinary chronic pain management: past, present, and future. Am Psychol. 2014;69(2):119-130. doi:10.1037/a0035514
- Courtney RE, Schadegg MJ. Chronic, noncancer pain care in the veterans administration: current trends and future directions. Anesthesiol Clin. 2023;41(2):519-529. doi:10.1016/j.anclin.2023.02.004
- Gallagher RM. Advancing the pain agenda in the veteran population. Anesthesiol Clin. 2016;34(2):357-378. doi:10.1016/j.anclin.2016.01.003
- Kamper SJ, Apeldoorn AT, Chiarotto A, et al. Multidisciplinary biopsychosocial rehabilitation for chronic low back pain: cochrane systematic review and meta-analysis. BMJ. 2015;350:h444. doi:10.1136/bmj.h444
- Waterschoot FPC, Dijkstra PU, Hollak N, De Vries HJ, Geertzen JHB, Reneman MF. Dose or content? Effectiveness of pain rehabilitation programs for patients with chronic low back pain: a systematic review. Pain. 2014;155(1):179-189. doi:10.1016/j.pain.2013.10.006
- Scascighini L, Toma V, Dober-Spielmann S, Sprott H. Multidisciplinary treatment for chronic pain: a systematic review of interventions and outcomes. Rheumatology (Oxford). 2008;47(5):670-678. doi:10.1093/rheumatology/ken021
- Elbers S, Wittink H, Konings S, et al. Longitudinal outcome evaluations of interdisciplinary multimodal pain Treatment programmes for patients with chronic primary musculoskeletal pain: a systematic review and meta-analysis. Eur J Pain. 2022;26(2):310-335. doi:10.1002/ejp.1875
- Turk DC, Dworkin RH, Allen RR, et al. Core outcome domains for chronic pain clinical trials: IMMPACT recommendations. Pain. 2003;106(3):337-345. doi:10.1016/j.pain.2003.08.001
- Dworkin RH, Turk DC, Farrar JT, et al. Core outcome measures for chronic pain clinical trials: IMMPACT recommendations. Pain. 2005;113(1-2):9-19. doi:10.1016/j.pain.2004.09.012
- Kligler B, Hyde J, Gantt C, Bokhour B. The whole health transformation at the Veterans Health Administration: moving from “what’s the matter with you?” to “what matters to you?”. Med Care. 2022;60(5):387-391. doi:10.1097/mlr.0000000000001706
- National Academies of Sciences, Engineering, and Medicine; Health and Medicine Division; Board on Health Care Services; Committee on Transforming Health Care to Create Whole Health: Strategies to Assess, Scale, and Spread the Whole Person Approach to Health, Meisnere M, SouthPaul J, Krist AH, eds. Achieving Whole Health: A New Approach for Veterans and the Nation. National Academies Press (US); February 15, 2023.
- The time Is now for a whole-person health approach to public health. Public Health Rep. 2023;138(4):561-564. doi:10.1177/00333549231154583
- Krejci LP, Carter K, Gaudet T. Whole health: the vision and implementation of personalized, proactive, patient-driven health care for veterans. Med Care. 2014;52(12 Suppl 5):S5-S8. doi:10.1097/mlr.0000000000000226
- Bokhour BG, Hyde J, Kligler B, et al. From patient outcomes to system change: evaluating the impact of VHA’s implementation of the whole health system of care. Health Serv Res. 2022;57 Suppl 1(Suppl 1):53-65. doi:10.1111/1475-6773.13938
- Zeliadt SB, Douglas JH, Gelman H, et al. Effectiveness of a whole health model of care emphasizing complementary and integrative health on reducing opioid use among patients with chronic pain. BMC Health Serv Res. 2022;22(1):1053. doi:10.1186/s12913-022-08388-2
- Reed DE 2nd, Bokhour BG, Gaj L, et al. Whole health use and interest across veterans with cooccurring chronic pain and PTSD: an examination of the 18 VA medical center flagship sites. Glob Adv Health Med. 2022;11:21649561211065374. doi:10.1177/21649561211065374
- Etingen B, Smith BM, Zeliadt SB, et al. VHA whole health services and complementary and integrative health therapies: a gateway to evidence-based mental health treatment. J Gen Intern Med. 2023;38(14):3144-3151. doi:10.1007/s11606-023-08296-z
- Johnson EM, Possemato K, Khan S, Chinman M, Maisto SA. Engagement, experience, and satisfaction with peerdelivered whole health coaching for veterans with PTSD: a mixed methods process evaluation. Psychol Serv. 2021;19(2):305-316. doi:10.1037/ser0000529
- Purcell N, Zamora K, Gibson C, et al. Patient experiences with integrated pain care: a qualitative evaluation of one VA’s biopsychosocial approach to chronic pain treatment and opioid safety. Glob Adv Health Med. 2019;8:2164956119838845. doi:10.1177/2164956119838845
- Will KK, Johnson ML, Lamb G. Team-based care and patient satisfaction in the hospital setting: a systematic review. J Patient Cent Res Rev. 2019;6(2):158-171. doi:10.17294/2330-0698.1695
- van Dongen JJJ, Habets IGJ, Beurskens A, van Bokhoven MA. Successful participation of patients in interprofessional team meetings: a qualitative study. Health Expect. 2017;20(4):724-733. doi:10.1111/hex.12511
- Oliver DP, Albright DL, Kruse RL, Wittenberg-Lyles E, Washington K, Demiris G. Caregiver evaluation of the ACTIVE intervention: “it was like we were sitting at the table with everyone.” Am J Hosp Palliat Care. 2014;31(4):444-453. doi:10.1177/1049909113490823
- Ansmann L, Heuser C, Diekmann A, et al. Patient participation in multidisciplinary tumor conferences: how is it implemented? What is the patients’ role? What are patients’ experiences? Cancer Med. 2021;10(19):6714-6724. doi:10.1002/cam4.4213
- US Department of Veterans Affairs, Veterans Health Administration. Updated March 20, 2023. Accessed June 11, 2024. https://www.va.gov/health/priorities/index.asp
- Darnall BD, Edwards KA, Courtney RE, Ziadni MS, Simons LE, Harrison LE. Innovative treatment formats, technologies, and clinician trainings that improve access to behavioral pain treatment for youth and adults. Front Pain Res (Lausanne). 2023;4:1223172. doi:10.3389/fpain.2023.1223172
- Kligler B. Whole health in the Veterans Health Administration. Glob Adv Health Med. 2022;11:2164957X221077214.
- Howe RJ, Poulin LM, Federman DG. The personal health inventory: current use, perceived barriers, and benefits. Fed Pract. 2017;34(5):23-26. doi:10.1177/2164957X221077214
- Hicks N, Harden S, Oursler KA, Courtney RE. Determining the representativeness of participants in a whole health interdisciplinary chronic pain program (PREVAIL) in a VA medical center: who did we reach? Presented at: PAINWeek 2022; September 6-9, 2022; Las Vegas, Nevada. Accessed September 10, 2024. https://www.tandfonline.com/doi/full/10.1080/00325481.2022.2116839
- Creswell JW, Creswell JD. Research Design: Qualitative, Quantitative, and Mixed Methods Approaches. SAGE Publications; 2018.
- Tong A, Sainsbury P, Craig J. Consolidated criteria for reporting qualitative research (COREQ): a 32-item checklist for interviews and focus groups. Int J Qual in Health Care. 2007;19(6):349-357. doi:10.1093/intqhc/mzm042
- National Institute for Health and Care Excellence. Methods for the development of NICE public health guidance, 3rd edition. Published September 26, 2012. Accessed June 11, 2024. https://www.nice.org.uk/process/pmg4/chapter/introduction
- Alexander JA, Hearld LR, Mittler JN, Harvey J. Patient-physician role relationships and patient activation among individuals with chronic illness. Health Serv Res. 2012;47(3 PART 1):1201-1223. doi:10.1111/j.1475-6773.2011.01354.x
- Fu Y, Yu G, McNichol E, Marczewski K, Closs SJ. The association between patient-professional partnerships and self-management of chronic back pain: a mixed methods study. Eur J Pain. 2018;22(7):1229-1244. doi:10.1002/ejp.1210
- Nicholas MK, Asghari A, Blyth FM, et al. Self-management intervention for chronic pain in older adults: a randomised controlled trial. Pain. 2013;154(6):824-835. doi:10.1016/j.pain.2013.02.009
- Nøst TH, Steinsbekk A, Bratås O, Grønning K. Twelvemonth effect of chronic pain self-management intervention delivered in an easily accessible primary healthcare service - a randomised controlled trial. BMC Health Serv Res. 2018;18(1):1012. doi:10.1186/s12913-018-3843-x
- Blyth FM, March LM, Nicholas MK, Cousins MJ. Selfmanagement of chronic pain: a population-based study. Pain. 2005;113(3):285-292. doi:10.1016/j.pain.2004.12.004
- Damush TM, Kroenke K, Bair MJ, et al. Pain self-management training increases self-efficacy, self-management behaviours and pain and depression outcomes. Eur J Pain. 2016;20(7):1070-1078. doi:10.1002/ejp.830
- Murphy JL, Palyo SA, Schmidt ZS, et al. The resurrection of interdisciplinary pain rehabilitation: outcomes across a veterans affairs collaborative. Pain Med. 2021;22(2):430- 443. doi:10.1093/pm/pnaa417
- Brain K, Burrows TL, Bruggink L, et al. Diet and chronic non-cancer pain: the state of the art and future directions. J Clin Med. 2021;10(21):5203. doi:10.3390/jcm10215203
- Field R, Pourkazemi F, Turton J, Rooney K. Dietary interventions are beneficial for patients with chronic pain: a systematic review with meta-analysis. Pain Med). 2021;22(3):694-714. doi:10.1093/pm/pnaa378
- Bjørklund G, Aaseth J, Do§a MD, et al. Does diet play a role in reducing nociception related to inflammation and chronic pain? Nutrition. 2019;66:153-165. doi:10.1016/j.nut.2019.04.007
- Kaushik AS, Strath LJ, Sorge RE. Dietary interventions for treatment of chronic pain: oxidative stress and inflammation. Pain Ther. 2020;9(2):487-498. doi:10.1007/s40122-020-00200-5
- Boswell JF, Hepner KA, Lysell K, et al. The need for a measurement-based care professional practice guideline. Psychotherapy (Chic). 2023;60(1):1-16. doi:10.1037/pst0000439
- Lund BC, Ohl ME, Hadlandsmyth K, Mosher HJ. Regional and rural-urban variation in opioid prescribing in the Veterans Health Administration. Mil Med. 2019;184(11-12):894- 900. doi:10.1093/milmed/usz104
- US Department of Veterans Affairs, Office of Rural Health. Rural veterans. Updated May 14, 2024. Accessed June 11, 2024. https://www.ruralhealth.va.gov/aboutus/ruralvets.asp
- McCarthy JF, Blow FC, Ignacio R V., Ilgen MA, Austin KL, Valenstein M. Suicide among patients in the Veterans Affairs health system: rural-urban differences in rates, risks, and methods. Am J Public Health. 2012;102 Suppl 1(suppl 1):S111-S117. doi:10.2105/AJPH.2011.300463
Chronic pain is one of the most prevalent public health concerns in the United States, affecting > 51 million adults with about $500 billion in health care costs.1 Military veterans are among the most vulnerable subpopulations, with 65% of veterans reporting chronic pain in the last 3 months.2 Chronic pain is complex, affecting the biopsychosocial-spiritual levels of human health, and requires multimodal and comprehensive treatment approaches.3 Hence, chronic pain treatment can be best delivered via interdisciplinary teams (IDTs) that use a patient-centered approach.4,5
The Veterans Healthcare Administration (VHA) is a leader in developing and delivering interdisciplinary pain care.6,7 VHA Directive 2009-053 requires every US Department of Veterans Affairs (VA) medical center to offer an IDT for chronic pain. However, VHA and non-VHA IDT programs vary significantly.8-11 A recent systematic review found a median of 5 disciplines included on IDTs (range, 2-8), and program content often included exercise and education; only 11% of included IDTs met simultaneously with patients.11 The heterogeneity of IDT programs has made determining best practices challenging.8,11 The Initiative on Methods, Measurement, and Pain Assessment in Clinical Trials has denoted several core measures and measurement domains that were critical for determining the success of pain management interventions, including patient satisfaction.12,13 Nevertheless, the association of IDTs with high patient satisfaction and improvement in pain measures has been documented.5,11
The VHA has worked to implement the Whole Health System into health care, which considers well-being across physical, behavioral, spiritual, and socioeconomic domains. As such, the Whole Health System involves an interpersonal, team-based approach, “anchored in trusting longitudinal relationships to promote resilience, prevent disease, and restore health.”14 It aligns with the patient’s mission, aspiration, and purpose. Surgeon General VADM Vivek H. Murthy, MD, MBA, recently endorsed this approach.15,16 Other health care systems adopting whole health tend to have higher patient satisfaction, increased access to care, and improved patient-reported outcomes.15 Within the VHA, the Whole Health System has shifted the conversation between clinicians and patients from “What is the matter with you?” to “What is important to you?” while emphasizing a proactive and personalized approach to health care.17 Rather than emphasizing passive modalities such as medications and clinician-led services (eg, interventional pain service), the Whole Health System highlights self-care.3,17 Initial research findings within the VHA have been promising.18-21 Whole health peer coaching calls appear to be an effective approach for veterans diagnosed with PTSD, and the use of whole health services is associated with a decrease in opioid use.19,22 However, there are negligible data on patient experiences after meeting with a whole health-focused pain IDT, and studies to date have focused on urban populations.23 One approach to IDT that has shown promise for other health issues involves a patient meeting simultaneously with all members of the IDT.24-27 With the integration of the Whole Health System and the VHA priorities to provide veterans with the “soonest and best care,” more data are needed on the experiences of patients and support persons with various approaches to IDT pain care.28 This study aimed to evaluate patient and support person experiences with a whole health-focused pain IDT that met simultaneously with the patient and support person during an initial evaluation. This study was approved by the institutional review board at the Salem VA Health Care System (SVAHCS) in Virginia.
Methods
The PREVAIL IDT Track is a clinical program offered at SVAHCS with a whole health-focused approach that involves patients and their support persons meeting simultaneously with a pain IDT. PREVAIL IDT Track is designed to help veterans more effectively self-manage chronic pain (Table 1).6,29 Health care practitioners (HCPs) at SVAHCS recommended that veterans with pain persisting for > 3 months participate in PREVAIL IDT Track. After meeting with an advanced practice clinician for an intake, veterans elected to participate in the PREVAIL IDT Track program and completed the initial 6 weeks of pain education. Veterans were then invited to be evaluated by the pain IDT. A team including HCPs from interventional pain, psychology, pharmacy, nutrition, and physical therapy services met with the veteran for 60 minutes. Veterans were also invited to bring a support person to the IDT initial evaluation.

During the IDT initial evaluation, HCPs inquired about the patient’s mission, aspiration, and purpose (“If you were in less pain, what would you be doing more of?”) and about whole health self-care and wellness factors that may contribute to their chronic pain using the Personal Health Inventory.30,31 Veterans were then invited to select 3 whole health self-care areas to focus on during the 6-month program.3 The IDT HCPs worked with the veteran to establish the treatment plan for the first month in the areas of self-care selected by the patient and made recommendations for additional treatments. If the veteran brought a support person to the IDT initial evaluation, their feedback was elicited throughout and at the end to ensure the final treatment plan and first month’s goals were realistic. At the end of the appointment, the veteran and their support person were asked to complete a program-specific satisfaction survey. The HCPs on the team and the veteran executed the treatment plan developed during the appointment, except for medication prescribing. Recommendations for medication changes are included in clinical notes. Veterans then received 5 monthly coaching calls from a nurse navigator with training in whole health and a 6-month follow-up appointment with the IDT HCPs to discuss a plan for continuity of care.
Participant demographic information was not collected, and participants were not compensated for completing the survey. Veterans in PREVAIL IDT Track are predominantly residents of central Appalachia, White, male, unemployed, have ≥ 1 mental and physical health comorbidity, and have a history of mental health treatment.32 Veterans participating in PREVAIL IDT had a mean age of 57 years, and about 1 in 3 have opioid prescriptions.32
A program-specific 17-question satisfaction survey was developed, which included questions related to satisfaction with previous SVAHCS pain care and staff interactions. To assess the overall impression of the IDT initial evaluation, 3 yes/no questions and a 0 to 10-point scale were used. The 5 remaining open-ended questions allowed participants to give feedback about the IDT initial evaluation.
Data Analysis
A convergent mixed-methods approach was used to evaluate participant satisfaction with the initial IDT evaluation. The study team collected and analyzed quantitative and qualitative survey data and triangulated the findings.33 For quantitative responses, frequencies and means were calculated using Python. For qualitative responses, thematic data analysis was conducted by systematic coding, using inductive methods and allowing themes to emerge. Study team members performed a line-by-line analysis of responses using NVivo to identify important codes and reach a consensus. This study adhered to the Consolidated Criteria for Reporting Qualitative Research and followed the National Institute for Health Care Excellence checklist.34,35
Results
Quantitative Responses
In 2022, 168 veterans completed the initial IDT evaluation, and 144 (85.2%) completed the satisfaction survey and were included in this study. Thirty-two support persons who attended the initial IDT evaluation and completed the survey also were included. Of the 12 quantitative questions, 4 had a 100% completion rate, while 8 had ≤ 3% missing responses. When describing care prior to participating in PREVAIL, participants indicated a mean (SD) response of 4.6 (1.4) with the health care they received at SVAHCS and 4.3 (1.4) with SVAHCS pain management services, both on 6-point scales. All but 2 participants (98.9%) reported always being treated with courtesy and respect by PREVAIL HCPs during the initial IDT evaluation, with a mean (SD) score of 4.0 (0.2) on a 4-point scale. Most respondents (96.6%) reported that PREVAIL HCPs always listened carefully during the initial IDT evaluation, with a mean (SD) 4.0 (0.3) on a 4-point scale. Similarly, 92.6% reported that PREVAIL HCPs explained things clearly during the initial IDT evaluation, with a mean (SD) 3.9 (0.3) on a 4-point scale.
All respondents agreed that PREVAIL HCPs considered veteran preferences and those of their support persons in deciding their health care needs during the initial IDT evaluation, with a mean (SD) 3.7 (0.5) on a 4-point scale. Most respondents left the appointment with a good understanding of their responsibilities for chronic pain management with 99.4% (n = 169) strongly agreeing or agreeing (mean [SD] 3.6 [0.5]). A total of 135 respondents (79.4%) reported they left appointments with written information on their treatment plan. All 170 respondents reported that they would recommend PREVAIL to a friend, and 169 respondents (98.8%) felt that the initial PREVAIL IDT evaluation was a valuable use of time. Eighty-seven respondents (50.9%) rated the initial IDT evaluation as the “best clinical experience possible” with a mean (SD) score of 9.2 (1.1) on a 10-point scale (Table 2).

Qualitative Responses
Respondents provided complementary feedback on the program, with many participants stating that they enjoyed every aspect (eAppendix, available at doi:10.12788/fp.0503). In terms of positive aspects of the program, several themes emerged: participants appreciated meeting as an IDT, feeling cared for and listened to, learning more about their pain and ways to manage it, and specific services offered. Thirty-three of 144 respondents wanted longer appointment times. Twenty-two respondents suggested logistics improvements (eg, meeting in a larger room, having a written plan at the end, sending paperwork ahead of time, and later appointment times).
Discussion
Veterans and support persons were satisfied with the initial IDT evaluation for the PREVAIL whole health-focused pain clinical program for veterans predominantly residing in central Appalachia. These satisfaction findings are noteworthy since 20% of this same sample reported dissatisfaction with prior pain services, which could affect engagement and outcomes in pain care. In addition to high satisfaction levels, the PREVAIL IDT model may benefit veterans with limited resources. Rather than needing to attend several individual appointments, the PREVAIL IDT Track provides a 1-stop shop approach that decreases patient burden and barriers to care (eg, travel, transportation, and time) as well as health care system burden. For instance, schedulers need only to make 1 appointment for the veteran rather than several. This approach was highly acceptable to veterans served at SVAHCS and may increase the reach and impact of VHA IDT pain care.
The PREVAIL model may foster rapport with HCPs and encourage an active role in self-managing pain.36,37 Participants noted that their preferences were considered and that they had a good understanding of their responsibilities for managing their chronic pain. This patient-centered approach, emphasizing an active role for the patient, is a hallmark of the VHA Whole Health System and aligns with the overarching PREVAIL IDT Track goal to enhance self-management skills, thus improving functioning through decreased pain interference.14,38-41
Participants in PREVAIL provided substantial open-ended feedback that has contributed to the program’s improvement and may provide information into preferred components of pain IDT programs, particularly for rural veterans. When asked about their favorite component of the initial IDT evaluation, the most emergent theme was meeting simultaneously with HCPs on the IDT. This finding is significant, given that only 11% of IDTs involve direct patient interaction.
Furthermore, unlike most IDTs, PREVAIL IDT includes a dietitian.11,42 IDT programs may benefit from dietitian involvement given the importance of the anti-inflammatory diet on chronic pain.43-46 Participants recommended improvements, (eg, changes to the location and timing, adding a written treatment plan at the end of the appointment, and completing paperwork prior to the appointment) many of which have been addressed. The program now uses validated measures to track progress and comprehensive assessments of pain in response to calls for measurement-based care.13,47 These process improvement suggestions may be instructive for other VA medical centers with rural populations.
Limitations
This study used a program-specific satisfaction survey with open-ended questions to allow for rich responses; however, the survey has not been validated. It also sought to minimize bias by asking participants to give completed surveys to staff members who were not HCPs on the IDT. However, participants’ responses may still have been influenced by this process. Response rate and demographics for support persons were impossible to determine. The results analyzed the responses of veterans and support persons together, which may have skewed the data. Future studies of pain IDT programs should consider analyzing responses from veterans and their support persons separately and identifying factors (eg, demographics or clinical characteristics) that influence the patients’ experiences while participating.
Conclusions
The initial PREVAIL IDT evaluation at SVAHCS is associated with high-levels of satisfaction. These veterans living in rural Appalachia, similar to the 4.4 million rural US veterans, are more likely to encounter barriers to care (eg, drive time, or transportation concerns) and be prescribed opioids.48 These veterans are also at high risk of chronic physical and mental health comorbidities, drug misuse, overdose, and suicide.49,50 Providing veterans in rural communities the opportunity to attend a single appointment with a pain IDT instead of requiring several individual appointments could improve the reach of evidence-based pain care.
This model of meeting simultaneously with all HCPs on the pain IDT may connect all veterans to the most available and best care, something prioritized by the VHA.28 The initial PREVAIL IDT evaluation also utilizes the Personal Health Inventory and the VHA Whole Health System Circle of Health to design patient-centered treatment plans. Integration of the Whole Health System is currently a high priority within VHA. The PREVAIL IDT Track model warrants additional efficacy research.
Chronic pain is one of the most prevalent public health concerns in the United States, affecting > 51 million adults with about $500 billion in health care costs.1 Military veterans are among the most vulnerable subpopulations, with 65% of veterans reporting chronic pain in the last 3 months.2 Chronic pain is complex, affecting the biopsychosocial-spiritual levels of human health, and requires multimodal and comprehensive treatment approaches.3 Hence, chronic pain treatment can be best delivered via interdisciplinary teams (IDTs) that use a patient-centered approach.4,5
The Veterans Healthcare Administration (VHA) is a leader in developing and delivering interdisciplinary pain care.6,7 VHA Directive 2009-053 requires every US Department of Veterans Affairs (VA) medical center to offer an IDT for chronic pain. However, VHA and non-VHA IDT programs vary significantly.8-11 A recent systematic review found a median of 5 disciplines included on IDTs (range, 2-8), and program content often included exercise and education; only 11% of included IDTs met simultaneously with patients.11 The heterogeneity of IDT programs has made determining best practices challenging.8,11 The Initiative on Methods, Measurement, and Pain Assessment in Clinical Trials has denoted several core measures and measurement domains that were critical for determining the success of pain management interventions, including patient satisfaction.12,13 Nevertheless, the association of IDTs with high patient satisfaction and improvement in pain measures has been documented.5,11
The VHA has worked to implement the Whole Health System into health care, which considers well-being across physical, behavioral, spiritual, and socioeconomic domains. As such, the Whole Health System involves an interpersonal, team-based approach, “anchored in trusting longitudinal relationships to promote resilience, prevent disease, and restore health.”14 It aligns with the patient’s mission, aspiration, and purpose. Surgeon General VADM Vivek H. Murthy, MD, MBA, recently endorsed this approach.15,16 Other health care systems adopting whole health tend to have higher patient satisfaction, increased access to care, and improved patient-reported outcomes.15 Within the VHA, the Whole Health System has shifted the conversation between clinicians and patients from “What is the matter with you?” to “What is important to you?” while emphasizing a proactive and personalized approach to health care.17 Rather than emphasizing passive modalities such as medications and clinician-led services (eg, interventional pain service), the Whole Health System highlights self-care.3,17 Initial research findings within the VHA have been promising.18-21 Whole health peer coaching calls appear to be an effective approach for veterans diagnosed with PTSD, and the use of whole health services is associated with a decrease in opioid use.19,22 However, there are negligible data on patient experiences after meeting with a whole health-focused pain IDT, and studies to date have focused on urban populations.23 One approach to IDT that has shown promise for other health issues involves a patient meeting simultaneously with all members of the IDT.24-27 With the integration of the Whole Health System and the VHA priorities to provide veterans with the “soonest and best care,” more data are needed on the experiences of patients and support persons with various approaches to IDT pain care.28 This study aimed to evaluate patient and support person experiences with a whole health-focused pain IDT that met simultaneously with the patient and support person during an initial evaluation. This study was approved by the institutional review board at the Salem VA Health Care System (SVAHCS) in Virginia.
Methods
The PREVAIL IDT Track is a clinical program offered at SVAHCS with a whole health-focused approach that involves patients and their support persons meeting simultaneously with a pain IDT. PREVAIL IDT Track is designed to help veterans more effectively self-manage chronic pain (Table 1).6,29 Health care practitioners (HCPs) at SVAHCS recommended that veterans with pain persisting for > 3 months participate in PREVAIL IDT Track. After meeting with an advanced practice clinician for an intake, veterans elected to participate in the PREVAIL IDT Track program and completed the initial 6 weeks of pain education. Veterans were then invited to be evaluated by the pain IDT. A team including HCPs from interventional pain, psychology, pharmacy, nutrition, and physical therapy services met with the veteran for 60 minutes. Veterans were also invited to bring a support person to the IDT initial evaluation.

During the IDT initial evaluation, HCPs inquired about the patient’s mission, aspiration, and purpose (“If you were in less pain, what would you be doing more of?”) and about whole health self-care and wellness factors that may contribute to their chronic pain using the Personal Health Inventory.30,31 Veterans were then invited to select 3 whole health self-care areas to focus on during the 6-month program.3 The IDT HCPs worked with the veteran to establish the treatment plan for the first month in the areas of self-care selected by the patient and made recommendations for additional treatments. If the veteran brought a support person to the IDT initial evaluation, their feedback was elicited throughout and at the end to ensure the final treatment plan and first month’s goals were realistic. At the end of the appointment, the veteran and their support person were asked to complete a program-specific satisfaction survey. The HCPs on the team and the veteran executed the treatment plan developed during the appointment, except for medication prescribing. Recommendations for medication changes are included in clinical notes. Veterans then received 5 monthly coaching calls from a nurse navigator with training in whole health and a 6-month follow-up appointment with the IDT HCPs to discuss a plan for continuity of care.
Participant demographic information was not collected, and participants were not compensated for completing the survey. Veterans in PREVAIL IDT Track are predominantly residents of central Appalachia, White, male, unemployed, have ≥ 1 mental and physical health comorbidity, and have a history of mental health treatment.32 Veterans participating in PREVAIL IDT had a mean age of 57 years, and about 1 in 3 have opioid prescriptions.32
A program-specific 17-question satisfaction survey was developed, which included questions related to satisfaction with previous SVAHCS pain care and staff interactions. To assess the overall impression of the IDT initial evaluation, 3 yes/no questions and a 0 to 10-point scale were used. The 5 remaining open-ended questions allowed participants to give feedback about the IDT initial evaluation.
Data Analysis
A convergent mixed-methods approach was used to evaluate participant satisfaction with the initial IDT evaluation. The study team collected and analyzed quantitative and qualitative survey data and triangulated the findings.33 For quantitative responses, frequencies and means were calculated using Python. For qualitative responses, thematic data analysis was conducted by systematic coding, using inductive methods and allowing themes to emerge. Study team members performed a line-by-line analysis of responses using NVivo to identify important codes and reach a consensus. This study adhered to the Consolidated Criteria for Reporting Qualitative Research and followed the National Institute for Health Care Excellence checklist.34,35
Results
Quantitative Responses
In 2022, 168 veterans completed the initial IDT evaluation, and 144 (85.2%) completed the satisfaction survey and were included in this study. Thirty-two support persons who attended the initial IDT evaluation and completed the survey also were included. Of the 12 quantitative questions, 4 had a 100% completion rate, while 8 had ≤ 3% missing responses. When describing care prior to participating in PREVAIL, participants indicated a mean (SD) response of 4.6 (1.4) with the health care they received at SVAHCS and 4.3 (1.4) with SVAHCS pain management services, both on 6-point scales. All but 2 participants (98.9%) reported always being treated with courtesy and respect by PREVAIL HCPs during the initial IDT evaluation, with a mean (SD) score of 4.0 (0.2) on a 4-point scale. Most respondents (96.6%) reported that PREVAIL HCPs always listened carefully during the initial IDT evaluation, with a mean (SD) 4.0 (0.3) on a 4-point scale. Similarly, 92.6% reported that PREVAIL HCPs explained things clearly during the initial IDT evaluation, with a mean (SD) 3.9 (0.3) on a 4-point scale.
All respondents agreed that PREVAIL HCPs considered veteran preferences and those of their support persons in deciding their health care needs during the initial IDT evaluation, with a mean (SD) 3.7 (0.5) on a 4-point scale. Most respondents left the appointment with a good understanding of their responsibilities for chronic pain management with 99.4% (n = 169) strongly agreeing or agreeing (mean [SD] 3.6 [0.5]). A total of 135 respondents (79.4%) reported they left appointments with written information on their treatment plan. All 170 respondents reported that they would recommend PREVAIL to a friend, and 169 respondents (98.8%) felt that the initial PREVAIL IDT evaluation was a valuable use of time. Eighty-seven respondents (50.9%) rated the initial IDT evaluation as the “best clinical experience possible” with a mean (SD) score of 9.2 (1.1) on a 10-point scale (Table 2).

Qualitative Responses
Respondents provided complementary feedback on the program, with many participants stating that they enjoyed every aspect (eAppendix, available at doi:10.12788/fp.0503). In terms of positive aspects of the program, several themes emerged: participants appreciated meeting as an IDT, feeling cared for and listened to, learning more about their pain and ways to manage it, and specific services offered. Thirty-three of 144 respondents wanted longer appointment times. Twenty-two respondents suggested logistics improvements (eg, meeting in a larger room, having a written plan at the end, sending paperwork ahead of time, and later appointment times).
Discussion
Veterans and support persons were satisfied with the initial IDT evaluation for the PREVAIL whole health-focused pain clinical program for veterans predominantly residing in central Appalachia. These satisfaction findings are noteworthy since 20% of this same sample reported dissatisfaction with prior pain services, which could affect engagement and outcomes in pain care. In addition to high satisfaction levels, the PREVAIL IDT model may benefit veterans with limited resources. Rather than needing to attend several individual appointments, the PREVAIL IDT Track provides a 1-stop shop approach that decreases patient burden and barriers to care (eg, travel, transportation, and time) as well as health care system burden. For instance, schedulers need only to make 1 appointment for the veteran rather than several. This approach was highly acceptable to veterans served at SVAHCS and may increase the reach and impact of VHA IDT pain care.
The PREVAIL model may foster rapport with HCPs and encourage an active role in self-managing pain.36,37 Participants noted that their preferences were considered and that they had a good understanding of their responsibilities for managing their chronic pain. This patient-centered approach, emphasizing an active role for the patient, is a hallmark of the VHA Whole Health System and aligns with the overarching PREVAIL IDT Track goal to enhance self-management skills, thus improving functioning through decreased pain interference.14,38-41
Participants in PREVAIL provided substantial open-ended feedback that has contributed to the program’s improvement and may provide information into preferred components of pain IDT programs, particularly for rural veterans. When asked about their favorite component of the initial IDT evaluation, the most emergent theme was meeting simultaneously with HCPs on the IDT. This finding is significant, given that only 11% of IDTs involve direct patient interaction.
Furthermore, unlike most IDTs, PREVAIL IDT includes a dietitian.11,42 IDT programs may benefit from dietitian involvement given the importance of the anti-inflammatory diet on chronic pain.43-46 Participants recommended improvements, (eg, changes to the location and timing, adding a written treatment plan at the end of the appointment, and completing paperwork prior to the appointment) many of which have been addressed. The program now uses validated measures to track progress and comprehensive assessments of pain in response to calls for measurement-based care.13,47 These process improvement suggestions may be instructive for other VA medical centers with rural populations.
Limitations
This study used a program-specific satisfaction survey with open-ended questions to allow for rich responses; however, the survey has not been validated. It also sought to minimize bias by asking participants to give completed surveys to staff members who were not HCPs on the IDT. However, participants’ responses may still have been influenced by this process. Response rate and demographics for support persons were impossible to determine. The results analyzed the responses of veterans and support persons together, which may have skewed the data. Future studies of pain IDT programs should consider analyzing responses from veterans and their support persons separately and identifying factors (eg, demographics or clinical characteristics) that influence the patients’ experiences while participating.
Conclusions
The initial PREVAIL IDT evaluation at SVAHCS is associated with high-levels of satisfaction. These veterans living in rural Appalachia, similar to the 4.4 million rural US veterans, are more likely to encounter barriers to care (eg, drive time, or transportation concerns) and be prescribed opioids.48 These veterans are also at high risk of chronic physical and mental health comorbidities, drug misuse, overdose, and suicide.49,50 Providing veterans in rural communities the opportunity to attend a single appointment with a pain IDT instead of requiring several individual appointments could improve the reach of evidence-based pain care.
This model of meeting simultaneously with all HCPs on the pain IDT may connect all veterans to the most available and best care, something prioritized by the VHA.28 The initial PREVAIL IDT evaluation also utilizes the Personal Health Inventory and the VHA Whole Health System Circle of Health to design patient-centered treatment plans. Integration of the Whole Health System is currently a high priority within VHA. The PREVAIL IDT Track model warrants additional efficacy research.
- Rikard SM, Strahan AE, Schmit KM, Guy GP Jr. Chronic pain among adults - United States, 2019-2021. MMWR Morb Mortal Wkly Rep. 2023;72(15):379-385. doi:10.15585/mmwr.mm7215a1
- Nahin RL. Severe Pain in Veterans: The effect of age and sex, and comparisons with the general population. J Pain. 2017;18(3):247-254. doi:10.1016/j.jpain.2016.10.021
- Courtney RE, Schadegg MJ, Bolton R, Smith S, Harden SM. Using a whole health approach to build biopsychosocial-spiritual personal health plans for veterans with chronic pain. Pain Manag Nurs. 2024;25(1):69-74. doi:10.1016/j.pmn.2023.09.010
- Mackey SC, Pearl RG. Pain management: optimizing patient care through comprehensive, interdisciplinary models and continuous innovations. Anesthesiol Clin. 2023;41(2):xv-xvii. doi:10.1016/j.anclin.2023.03.011
- Gatchel RJ, McGeary DD, McGeary CA, Lippe B. Interdisciplinary chronic pain management: past, present, and future. Am Psychol. 2014;69(2):119-130. doi:10.1037/a0035514
- Courtney RE, Schadegg MJ. Chronic, noncancer pain care in the veterans administration: current trends and future directions. Anesthesiol Clin. 2023;41(2):519-529. doi:10.1016/j.anclin.2023.02.004
- Gallagher RM. Advancing the pain agenda in the veteran population. Anesthesiol Clin. 2016;34(2):357-378. doi:10.1016/j.anclin.2016.01.003
- Kamper SJ, Apeldoorn AT, Chiarotto A, et al. Multidisciplinary biopsychosocial rehabilitation for chronic low back pain: cochrane systematic review and meta-analysis. BMJ. 2015;350:h444. doi:10.1136/bmj.h444
- Waterschoot FPC, Dijkstra PU, Hollak N, De Vries HJ, Geertzen JHB, Reneman MF. Dose or content? Effectiveness of pain rehabilitation programs for patients with chronic low back pain: a systematic review. Pain. 2014;155(1):179-189. doi:10.1016/j.pain.2013.10.006
- Scascighini L, Toma V, Dober-Spielmann S, Sprott H. Multidisciplinary treatment for chronic pain: a systematic review of interventions and outcomes. Rheumatology (Oxford). 2008;47(5):670-678. doi:10.1093/rheumatology/ken021
- Elbers S, Wittink H, Konings S, et al. Longitudinal outcome evaluations of interdisciplinary multimodal pain Treatment programmes for patients with chronic primary musculoskeletal pain: a systematic review and meta-analysis. Eur J Pain. 2022;26(2):310-335. doi:10.1002/ejp.1875
- Turk DC, Dworkin RH, Allen RR, et al. Core outcome domains for chronic pain clinical trials: IMMPACT recommendations. Pain. 2003;106(3):337-345. doi:10.1016/j.pain.2003.08.001
- Dworkin RH, Turk DC, Farrar JT, et al. Core outcome measures for chronic pain clinical trials: IMMPACT recommendations. Pain. 2005;113(1-2):9-19. doi:10.1016/j.pain.2004.09.012
- Kligler B, Hyde J, Gantt C, Bokhour B. The whole health transformation at the Veterans Health Administration: moving from “what’s the matter with you?” to “what matters to you?”. Med Care. 2022;60(5):387-391. doi:10.1097/mlr.0000000000001706
- National Academies of Sciences, Engineering, and Medicine; Health and Medicine Division; Board on Health Care Services; Committee on Transforming Health Care to Create Whole Health: Strategies to Assess, Scale, and Spread the Whole Person Approach to Health, Meisnere M, SouthPaul J, Krist AH, eds. Achieving Whole Health: A New Approach for Veterans and the Nation. National Academies Press (US); February 15, 2023.
- The time Is now for a whole-person health approach to public health. Public Health Rep. 2023;138(4):561-564. doi:10.1177/00333549231154583
- Krejci LP, Carter K, Gaudet T. Whole health: the vision and implementation of personalized, proactive, patient-driven health care for veterans. Med Care. 2014;52(12 Suppl 5):S5-S8. doi:10.1097/mlr.0000000000000226
- Bokhour BG, Hyde J, Kligler B, et al. From patient outcomes to system change: evaluating the impact of VHA’s implementation of the whole health system of care. Health Serv Res. 2022;57 Suppl 1(Suppl 1):53-65. doi:10.1111/1475-6773.13938
- Zeliadt SB, Douglas JH, Gelman H, et al. Effectiveness of a whole health model of care emphasizing complementary and integrative health on reducing opioid use among patients with chronic pain. BMC Health Serv Res. 2022;22(1):1053. doi:10.1186/s12913-022-08388-2
- Reed DE 2nd, Bokhour BG, Gaj L, et al. Whole health use and interest across veterans with cooccurring chronic pain and PTSD: an examination of the 18 VA medical center flagship sites. Glob Adv Health Med. 2022;11:21649561211065374. doi:10.1177/21649561211065374
- Etingen B, Smith BM, Zeliadt SB, et al. VHA whole health services and complementary and integrative health therapies: a gateway to evidence-based mental health treatment. J Gen Intern Med. 2023;38(14):3144-3151. doi:10.1007/s11606-023-08296-z
- Johnson EM, Possemato K, Khan S, Chinman M, Maisto SA. Engagement, experience, and satisfaction with peerdelivered whole health coaching for veterans with PTSD: a mixed methods process evaluation. Psychol Serv. 2021;19(2):305-316. doi:10.1037/ser0000529
- Purcell N, Zamora K, Gibson C, et al. Patient experiences with integrated pain care: a qualitative evaluation of one VA’s biopsychosocial approach to chronic pain treatment and opioid safety. Glob Adv Health Med. 2019;8:2164956119838845. doi:10.1177/2164956119838845
- Will KK, Johnson ML, Lamb G. Team-based care and patient satisfaction in the hospital setting: a systematic review. J Patient Cent Res Rev. 2019;6(2):158-171. doi:10.17294/2330-0698.1695
- van Dongen JJJ, Habets IGJ, Beurskens A, van Bokhoven MA. Successful participation of patients in interprofessional team meetings: a qualitative study. Health Expect. 2017;20(4):724-733. doi:10.1111/hex.12511
- Oliver DP, Albright DL, Kruse RL, Wittenberg-Lyles E, Washington K, Demiris G. Caregiver evaluation of the ACTIVE intervention: “it was like we were sitting at the table with everyone.” Am J Hosp Palliat Care. 2014;31(4):444-453. doi:10.1177/1049909113490823
- Ansmann L, Heuser C, Diekmann A, et al. Patient participation in multidisciplinary tumor conferences: how is it implemented? What is the patients’ role? What are patients’ experiences? Cancer Med. 2021;10(19):6714-6724. doi:10.1002/cam4.4213
- US Department of Veterans Affairs, Veterans Health Administration. Updated March 20, 2023. Accessed June 11, 2024. https://www.va.gov/health/priorities/index.asp
- Darnall BD, Edwards KA, Courtney RE, Ziadni MS, Simons LE, Harrison LE. Innovative treatment formats, technologies, and clinician trainings that improve access to behavioral pain treatment for youth and adults. Front Pain Res (Lausanne). 2023;4:1223172. doi:10.3389/fpain.2023.1223172
- Kligler B. Whole health in the Veterans Health Administration. Glob Adv Health Med. 2022;11:2164957X221077214.
- Howe RJ, Poulin LM, Federman DG. The personal health inventory: current use, perceived barriers, and benefits. Fed Pract. 2017;34(5):23-26. doi:10.1177/2164957X221077214
- Hicks N, Harden S, Oursler KA, Courtney RE. Determining the representativeness of participants in a whole health interdisciplinary chronic pain program (PREVAIL) in a VA medical center: who did we reach? Presented at: PAINWeek 2022; September 6-9, 2022; Las Vegas, Nevada. Accessed September 10, 2024. https://www.tandfonline.com/doi/full/10.1080/00325481.2022.2116839
- Creswell JW, Creswell JD. Research Design: Qualitative, Quantitative, and Mixed Methods Approaches. SAGE Publications; 2018.
- Tong A, Sainsbury P, Craig J. Consolidated criteria for reporting qualitative research (COREQ): a 32-item checklist for interviews and focus groups. Int J Qual in Health Care. 2007;19(6):349-357. doi:10.1093/intqhc/mzm042
- National Institute for Health and Care Excellence. Methods for the development of NICE public health guidance, 3rd edition. Published September 26, 2012. Accessed June 11, 2024. https://www.nice.org.uk/process/pmg4/chapter/introduction
- Alexander JA, Hearld LR, Mittler JN, Harvey J. Patient-physician role relationships and patient activation among individuals with chronic illness. Health Serv Res. 2012;47(3 PART 1):1201-1223. doi:10.1111/j.1475-6773.2011.01354.x
- Fu Y, Yu G, McNichol E, Marczewski K, Closs SJ. The association between patient-professional partnerships and self-management of chronic back pain: a mixed methods study. Eur J Pain. 2018;22(7):1229-1244. doi:10.1002/ejp.1210
- Nicholas MK, Asghari A, Blyth FM, et al. Self-management intervention for chronic pain in older adults: a randomised controlled trial. Pain. 2013;154(6):824-835. doi:10.1016/j.pain.2013.02.009
- Nøst TH, Steinsbekk A, Bratås O, Grønning K. Twelvemonth effect of chronic pain self-management intervention delivered in an easily accessible primary healthcare service - a randomised controlled trial. BMC Health Serv Res. 2018;18(1):1012. doi:10.1186/s12913-018-3843-x
- Blyth FM, March LM, Nicholas MK, Cousins MJ. Selfmanagement of chronic pain: a population-based study. Pain. 2005;113(3):285-292. doi:10.1016/j.pain.2004.12.004
- Damush TM, Kroenke K, Bair MJ, et al. Pain self-management training increases self-efficacy, self-management behaviours and pain and depression outcomes. Eur J Pain. 2016;20(7):1070-1078. doi:10.1002/ejp.830
- Murphy JL, Palyo SA, Schmidt ZS, et al. The resurrection of interdisciplinary pain rehabilitation: outcomes across a veterans affairs collaborative. Pain Med. 2021;22(2):430- 443. doi:10.1093/pm/pnaa417
- Brain K, Burrows TL, Bruggink L, et al. Diet and chronic non-cancer pain: the state of the art and future directions. J Clin Med. 2021;10(21):5203. doi:10.3390/jcm10215203
- Field R, Pourkazemi F, Turton J, Rooney K. Dietary interventions are beneficial for patients with chronic pain: a systematic review with meta-analysis. Pain Med). 2021;22(3):694-714. doi:10.1093/pm/pnaa378
- Bjørklund G, Aaseth J, Do§a MD, et al. Does diet play a role in reducing nociception related to inflammation and chronic pain? Nutrition. 2019;66:153-165. doi:10.1016/j.nut.2019.04.007
- Kaushik AS, Strath LJ, Sorge RE. Dietary interventions for treatment of chronic pain: oxidative stress and inflammation. Pain Ther. 2020;9(2):487-498. doi:10.1007/s40122-020-00200-5
- Boswell JF, Hepner KA, Lysell K, et al. The need for a measurement-based care professional practice guideline. Psychotherapy (Chic). 2023;60(1):1-16. doi:10.1037/pst0000439
- Lund BC, Ohl ME, Hadlandsmyth K, Mosher HJ. Regional and rural-urban variation in opioid prescribing in the Veterans Health Administration. Mil Med. 2019;184(11-12):894- 900. doi:10.1093/milmed/usz104
- US Department of Veterans Affairs, Office of Rural Health. Rural veterans. Updated May 14, 2024. Accessed June 11, 2024. https://www.ruralhealth.va.gov/aboutus/ruralvets.asp
- McCarthy JF, Blow FC, Ignacio R V., Ilgen MA, Austin KL, Valenstein M. Suicide among patients in the Veterans Affairs health system: rural-urban differences in rates, risks, and methods. Am J Public Health. 2012;102 Suppl 1(suppl 1):S111-S117. doi:10.2105/AJPH.2011.300463
- Rikard SM, Strahan AE, Schmit KM, Guy GP Jr. Chronic pain among adults - United States, 2019-2021. MMWR Morb Mortal Wkly Rep. 2023;72(15):379-385. doi:10.15585/mmwr.mm7215a1
- Nahin RL. Severe Pain in Veterans: The effect of age and sex, and comparisons with the general population. J Pain. 2017;18(3):247-254. doi:10.1016/j.jpain.2016.10.021
- Courtney RE, Schadegg MJ, Bolton R, Smith S, Harden SM. Using a whole health approach to build biopsychosocial-spiritual personal health plans for veterans with chronic pain. Pain Manag Nurs. 2024;25(1):69-74. doi:10.1016/j.pmn.2023.09.010
- Mackey SC, Pearl RG. Pain management: optimizing patient care through comprehensive, interdisciplinary models and continuous innovations. Anesthesiol Clin. 2023;41(2):xv-xvii. doi:10.1016/j.anclin.2023.03.011
- Gatchel RJ, McGeary DD, McGeary CA, Lippe B. Interdisciplinary chronic pain management: past, present, and future. Am Psychol. 2014;69(2):119-130. doi:10.1037/a0035514
- Courtney RE, Schadegg MJ. Chronic, noncancer pain care in the veterans administration: current trends and future directions. Anesthesiol Clin. 2023;41(2):519-529. doi:10.1016/j.anclin.2023.02.004
- Gallagher RM. Advancing the pain agenda in the veteran population. Anesthesiol Clin. 2016;34(2):357-378. doi:10.1016/j.anclin.2016.01.003
- Kamper SJ, Apeldoorn AT, Chiarotto A, et al. Multidisciplinary biopsychosocial rehabilitation for chronic low back pain: cochrane systematic review and meta-analysis. BMJ. 2015;350:h444. doi:10.1136/bmj.h444
- Waterschoot FPC, Dijkstra PU, Hollak N, De Vries HJ, Geertzen JHB, Reneman MF. Dose or content? Effectiveness of pain rehabilitation programs for patients with chronic low back pain: a systematic review. Pain. 2014;155(1):179-189. doi:10.1016/j.pain.2013.10.006
- Scascighini L, Toma V, Dober-Spielmann S, Sprott H. Multidisciplinary treatment for chronic pain: a systematic review of interventions and outcomes. Rheumatology (Oxford). 2008;47(5):670-678. doi:10.1093/rheumatology/ken021
- Elbers S, Wittink H, Konings S, et al. Longitudinal outcome evaluations of interdisciplinary multimodal pain Treatment programmes for patients with chronic primary musculoskeletal pain: a systematic review and meta-analysis. Eur J Pain. 2022;26(2):310-335. doi:10.1002/ejp.1875
- Turk DC, Dworkin RH, Allen RR, et al. Core outcome domains for chronic pain clinical trials: IMMPACT recommendations. Pain. 2003;106(3):337-345. doi:10.1016/j.pain.2003.08.001
- Dworkin RH, Turk DC, Farrar JT, et al. Core outcome measures for chronic pain clinical trials: IMMPACT recommendations. Pain. 2005;113(1-2):9-19. doi:10.1016/j.pain.2004.09.012
- Kligler B, Hyde J, Gantt C, Bokhour B. The whole health transformation at the Veterans Health Administration: moving from “what’s the matter with you?” to “what matters to you?”. Med Care. 2022;60(5):387-391. doi:10.1097/mlr.0000000000001706
- National Academies of Sciences, Engineering, and Medicine; Health and Medicine Division; Board on Health Care Services; Committee on Transforming Health Care to Create Whole Health: Strategies to Assess, Scale, and Spread the Whole Person Approach to Health, Meisnere M, SouthPaul J, Krist AH, eds. Achieving Whole Health: A New Approach for Veterans and the Nation. National Academies Press (US); February 15, 2023.
- The time Is now for a whole-person health approach to public health. Public Health Rep. 2023;138(4):561-564. doi:10.1177/00333549231154583
- Krejci LP, Carter K, Gaudet T. Whole health: the vision and implementation of personalized, proactive, patient-driven health care for veterans. Med Care. 2014;52(12 Suppl 5):S5-S8. doi:10.1097/mlr.0000000000000226
- Bokhour BG, Hyde J, Kligler B, et al. From patient outcomes to system change: evaluating the impact of VHA’s implementation of the whole health system of care. Health Serv Res. 2022;57 Suppl 1(Suppl 1):53-65. doi:10.1111/1475-6773.13938
- Zeliadt SB, Douglas JH, Gelman H, et al. Effectiveness of a whole health model of care emphasizing complementary and integrative health on reducing opioid use among patients with chronic pain. BMC Health Serv Res. 2022;22(1):1053. doi:10.1186/s12913-022-08388-2
- Reed DE 2nd, Bokhour BG, Gaj L, et al. Whole health use and interest across veterans with cooccurring chronic pain and PTSD: an examination of the 18 VA medical center flagship sites. Glob Adv Health Med. 2022;11:21649561211065374. doi:10.1177/21649561211065374
- Etingen B, Smith BM, Zeliadt SB, et al. VHA whole health services and complementary and integrative health therapies: a gateway to evidence-based mental health treatment. J Gen Intern Med. 2023;38(14):3144-3151. doi:10.1007/s11606-023-08296-z
- Johnson EM, Possemato K, Khan S, Chinman M, Maisto SA. Engagement, experience, and satisfaction with peerdelivered whole health coaching for veterans with PTSD: a mixed methods process evaluation. Psychol Serv. 2021;19(2):305-316. doi:10.1037/ser0000529
- Purcell N, Zamora K, Gibson C, et al. Patient experiences with integrated pain care: a qualitative evaluation of one VA’s biopsychosocial approach to chronic pain treatment and opioid safety. Glob Adv Health Med. 2019;8:2164956119838845. doi:10.1177/2164956119838845
- Will KK, Johnson ML, Lamb G. Team-based care and patient satisfaction in the hospital setting: a systematic review. J Patient Cent Res Rev. 2019;6(2):158-171. doi:10.17294/2330-0698.1695
- van Dongen JJJ, Habets IGJ, Beurskens A, van Bokhoven MA. Successful participation of patients in interprofessional team meetings: a qualitative study. Health Expect. 2017;20(4):724-733. doi:10.1111/hex.12511
- Oliver DP, Albright DL, Kruse RL, Wittenberg-Lyles E, Washington K, Demiris G. Caregiver evaluation of the ACTIVE intervention: “it was like we were sitting at the table with everyone.” Am J Hosp Palliat Care. 2014;31(4):444-453. doi:10.1177/1049909113490823
- Ansmann L, Heuser C, Diekmann A, et al. Patient participation in multidisciplinary tumor conferences: how is it implemented? What is the patients’ role? What are patients’ experiences? Cancer Med. 2021;10(19):6714-6724. doi:10.1002/cam4.4213
- US Department of Veterans Affairs, Veterans Health Administration. Updated March 20, 2023. Accessed June 11, 2024. https://www.va.gov/health/priorities/index.asp
- Darnall BD, Edwards KA, Courtney RE, Ziadni MS, Simons LE, Harrison LE. Innovative treatment formats, technologies, and clinician trainings that improve access to behavioral pain treatment for youth and adults. Front Pain Res (Lausanne). 2023;4:1223172. doi:10.3389/fpain.2023.1223172
- Kligler B. Whole health in the Veterans Health Administration. Glob Adv Health Med. 2022;11:2164957X221077214.
- Howe RJ, Poulin LM, Federman DG. The personal health inventory: current use, perceived barriers, and benefits. Fed Pract. 2017;34(5):23-26. doi:10.1177/2164957X221077214
- Hicks N, Harden S, Oursler KA, Courtney RE. Determining the representativeness of participants in a whole health interdisciplinary chronic pain program (PREVAIL) in a VA medical center: who did we reach? Presented at: PAINWeek 2022; September 6-9, 2022; Las Vegas, Nevada. Accessed September 10, 2024. https://www.tandfonline.com/doi/full/10.1080/00325481.2022.2116839
- Creswell JW, Creswell JD. Research Design: Qualitative, Quantitative, and Mixed Methods Approaches. SAGE Publications; 2018.
- Tong A, Sainsbury P, Craig J. Consolidated criteria for reporting qualitative research (COREQ): a 32-item checklist for interviews and focus groups. Int J Qual in Health Care. 2007;19(6):349-357. doi:10.1093/intqhc/mzm042
- National Institute for Health and Care Excellence. Methods for the development of NICE public health guidance, 3rd edition. Published September 26, 2012. Accessed June 11, 2024. https://www.nice.org.uk/process/pmg4/chapter/introduction
- Alexander JA, Hearld LR, Mittler JN, Harvey J. Patient-physician role relationships and patient activation among individuals with chronic illness. Health Serv Res. 2012;47(3 PART 1):1201-1223. doi:10.1111/j.1475-6773.2011.01354.x
- Fu Y, Yu G, McNichol E, Marczewski K, Closs SJ. The association between patient-professional partnerships and self-management of chronic back pain: a mixed methods study. Eur J Pain. 2018;22(7):1229-1244. doi:10.1002/ejp.1210
- Nicholas MK, Asghari A, Blyth FM, et al. Self-management intervention for chronic pain in older adults: a randomised controlled trial. Pain. 2013;154(6):824-835. doi:10.1016/j.pain.2013.02.009
- Nøst TH, Steinsbekk A, Bratås O, Grønning K. Twelvemonth effect of chronic pain self-management intervention delivered in an easily accessible primary healthcare service - a randomised controlled trial. BMC Health Serv Res. 2018;18(1):1012. doi:10.1186/s12913-018-3843-x
- Blyth FM, March LM, Nicholas MK, Cousins MJ. Selfmanagement of chronic pain: a population-based study. Pain. 2005;113(3):285-292. doi:10.1016/j.pain.2004.12.004
- Damush TM, Kroenke K, Bair MJ, et al. Pain self-management training increases self-efficacy, self-management behaviours and pain and depression outcomes. Eur J Pain. 2016;20(7):1070-1078. doi:10.1002/ejp.830
- Murphy JL, Palyo SA, Schmidt ZS, et al. The resurrection of interdisciplinary pain rehabilitation: outcomes across a veterans affairs collaborative. Pain Med. 2021;22(2):430- 443. doi:10.1093/pm/pnaa417
- Brain K, Burrows TL, Bruggink L, et al. Diet and chronic non-cancer pain: the state of the art and future directions. J Clin Med. 2021;10(21):5203. doi:10.3390/jcm10215203
- Field R, Pourkazemi F, Turton J, Rooney K. Dietary interventions are beneficial for patients with chronic pain: a systematic review with meta-analysis. Pain Med). 2021;22(3):694-714. doi:10.1093/pm/pnaa378
- Bjørklund G, Aaseth J, Do§a MD, et al. Does diet play a role in reducing nociception related to inflammation and chronic pain? Nutrition. 2019;66:153-165. doi:10.1016/j.nut.2019.04.007
- Kaushik AS, Strath LJ, Sorge RE. Dietary interventions for treatment of chronic pain: oxidative stress and inflammation. Pain Ther. 2020;9(2):487-498. doi:10.1007/s40122-020-00200-5
- Boswell JF, Hepner KA, Lysell K, et al. The need for a measurement-based care professional practice guideline. Psychotherapy (Chic). 2023;60(1):1-16. doi:10.1037/pst0000439
- Lund BC, Ohl ME, Hadlandsmyth K, Mosher HJ. Regional and rural-urban variation in opioid prescribing in the Veterans Health Administration. Mil Med. 2019;184(11-12):894- 900. doi:10.1093/milmed/usz104
- US Department of Veterans Affairs, Office of Rural Health. Rural veterans. Updated May 14, 2024. Accessed June 11, 2024. https://www.ruralhealth.va.gov/aboutus/ruralvets.asp
- McCarthy JF, Blow FC, Ignacio R V., Ilgen MA, Austin KL, Valenstein M. Suicide among patients in the Veterans Affairs health system: rural-urban differences in rates, risks, and methods. Am J Public Health. 2012;102 Suppl 1(suppl 1):S111-S117. doi:10.2105/AJPH.2011.300463
Patient and Support Person Satisfaction Following a Whole Health-Informed Interdisciplinary Pain Team Meeting
Patient and Support Person Satisfaction Following a Whole Health-Informed Interdisciplinary Pain Team Meeting
Clinical, Laboratory, and Trichoscopic Features of Pediatric Androgenetic Alopecia
Clinical, Laboratory, and Trichoscopic Features of Pediatric Androgenetic Alopecia
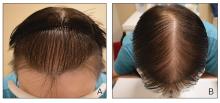
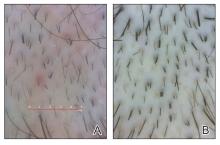
Androgenetic alopecia (AGA) is the most common type of hair loss after adolescence, with a high prevalence of 21.3% among males and 6.0% among females in China.1 In men, AGA manifests as diffuse hair loss in the frontal and temporal areas of the scalp; in women, it is characterized by thinning of the hair on the top of the head with a wide part and less recession of the frontal line. Although the specific pathogenesis of AGA still is unclear, it is believed to be related mainly to genetics and androgen levels.1 Androgenetic alopecia is not considered a life-threatening medical condition, but it can have a major impact on patients’ self-esteem and quality of life.
The prevalence of pediatric AGA has been steadily rising over the past few decades and is thought to be correlated to a hyperinsulinemic diet and elevated circulating androgens at younger ages, resulting in early onset in genetically susceptible children and adolescents.2,3 Additionally, studies have shown that early-onset AGA is associated with metabolic syndrome,4-6 which includes conditions such as obesity, insulin resistance, hyperglycemia, and dyslipidemia.7,8 Furthermore, polycystic ovary syndrome (PCOS) is commonly observed in adolescent girls with early-onset AGA. The condition is associated with hormonal imbalances, particularly elevated androgens, which can contribute to the early onset of AGA. In girls, these hormonal changes may accelerate hair thinning and hair loss, making AGA a potential early indicator of underlying PCOS.9,10
Available research on early-onset AGA in pediatric patients is limited, with most studies having a relatively small sample size and generalized findings. Data on pediatric AGA in China is scarce; therefore, the objective of this retrospective study was to analyze the clinical, laboratory, and trichoscopic features of AGA in 133 pediatric patients with AGA who visited the hair disease clinic of the Department of Dermatology at the First Affiliated Hospital of Nanjing Medical University (Nanjing, China), from January 2010 to December 2023.
Methods
Study Population—Pediatric patients with early-onset AGA who were registered for outpatient consultations at the hair disease clinic of the Department of Dermatology at The First Affiliated Hospital of Nanjing Medical University from January 2010 to December 2023 were included. Patients aged 18 years and younger with a definitive diagnosis of AGA were selected for data collection and analysis. Any uncertain information was confirmed through telephone follow-up with patients.
Collection of Demographic Information and Laboratory Tests—Patient demographics and medical history including age, sex, age at disease onset, and duration of AGA were collected from the electronic medical record. Height and weight also were collected to calculate patients’ body mass index (BMI). Detailed laboratory test results were recorded, including assessments of sex hormone—binding globulin (SHBG), vitamin D, testosterone, and ferritin.
Analysis of Comorbidities—Due to the influence of genetic factors on body composition, there are differences in how obesity is defined across racial populations. The World Health Organization international standard defines the term overweight as a BMI greater than 25 and obese as a BMI greater than 30; however, the World Health Organization recommends a lower definition standard for these classifications in the Chinese population. China established specific BMI standards for classification of patients as overweight (24.0.27.9 kg/m2) and obese (≥28 kg/m2).11 During outpatient consultations, a comprehensive medical history was obtained from each patient, including the presence of PCOS, acne, seborrheic dermatitis, hirsutism, and sleeping disorders. During routine outpatient assessments, experienced dermatologists (including W.F.) determined the presence of symptoms and confirmed the diagnosis.
Hair Loss Classification and Trichoscopy—Hair loss patterns for male patients were assessed using the basic and specific classification system, while the Ludwig scale was utilized for female patients.12,13 Trichoscopy was utilized with high-resolution imaging systems and advanced software for image analysis, enabling precise assessment of hair in different scalp regions. Parameters such as hair density, hair diameter, percentage of terminal hairs, and percentage of vellus hair were recorded to monitor changes in hair growth for the patients.
Statistical Analysis—Categorical data were analyzed using the x2 test. A P value less than .05 was considered statistically significant. All statistical analyses were conducted using SPSS software version 26 (IBM).
Results
Patient Characteristics and Hair Loss Patterns—A sample of 133 pediatric patients (60 males, 73 females) who were diagnosed with AGA at the hair disease clinic of the Department of Dermatology at the First Affiliated Hospital of Nanjing Medical University from January 2010 to December 2023 were selected. The mean age of the patients was 15.5 years (range, 10–18 years). The mean age was slightly lower in females compared with males (15.05 vs 16.19 years, respectively). Additionally, females showed earlier onset of the disease, with a mean age at onset of 13.41 years compared to 14.44 years in males. The time between onset of AGA symptoms and first seeking medical care ranged from 4 months to 3 years, with a mean disease duration of 1.72 years. There was no significant difference in the duration of disease between males and females (1.76 and 1.70 years, respectively). Patient characteristics by age group are summarized in eTable 1.
The pediatric patients in our study exhibited hair loss patterns similar to those typically observed in adults. Male patients typically showed diffuse thinning on the crown and varying degrees of temporal thinning, while female patients demonstrated diffuse thinning on the crown with a preserved frontal hairline; however, 5 (8.3%) male patients presented with Christmas tree– like pattern of hair loss with a preserved hairline and a thinning crown (Figures 1 and 2).
Diffuse thinning of the hair on the crown demonstrated a Christmas tree-like pattern with a preserved frontal hairline.
BMI and Comorbidities—Among our study sample, 27.1% (36/133) of patients were identified as overweight or obese. It came to our attention that the prevalence of patients who were overweight and obese was notably higher in patients aged older than 14 but younger than 18 years compared with those aged 14 years or younger (24.1% vs 3.0% [32/133 vs 4/133]). A more detailed analysis of patients who were overweight and obese is outlined in eTable 2.
Seborrheic dermatitis was identified as the most prevalent comorbidity associated with pediatric AGA (51.9% [69/133]), followed by acne (42.8% [57/133]), hirsutism (33.1% [44/133]), and sleep disturbances/insomnia (28.6% [38/133]). The prevalence of these comorbidities varied by age group, with a higher incidence observed among patients aged older than 14 years as compared to those aged 14 years or younger.
Family History of AGA—Our study results indicated that most (78.2% [104/133]) patients had a family history of AGA. Among males and females, 81.7% and 75.3% (49/60 and 55/73) had a positive family history, respectively. Further analysis showed that 43.3% (26/60) of males and 21.9% (16/73) of females reported AGA in their father, while 16.7% (10/60) of males and 35.6% insert (26/73) of females reported AGA in their mother. Both parents were affected in 21.7% (13/60) of male patients and 17.8% (13/73) of female patients (eTable 3).
Related Laboratory Tests of Pediatric Patients With AGA—The results of laboratory testing for vitamin D deficiency, low SHBG, high testosterone, and low ferritin levels in the study sample are outlined in eTable 4. Among the study participants, 15.9% (10/63) of females exhibited increased levels of both free and total testosterone. Low SHBG was observed in 47.1% (56/119) of patients, with a slightly higher proportion in males (48.2% [27/56] than females (46.0% [29/63]). Vitamin D deficiency was prevalent in 60.5% (72/119) of the study population, with a higher incidence rate in females (71.4%[45/63]) compared to males (48.2%[27/56]). Moreover, 21.8% (26/119) of pediatric patients had low ferritin levels, with a higher incidence rate in females (33.3%[21/63]) compared to males (8.9%[5/56]).
Female Patients With PCOS—In our study, 6 (8.2%) female patients with AGA had been diagnosed with PCOS prior to their referral to the First Affiliated Hospital of Nanjing Medical University. Information regarding their age at treatment, hair loss grade, comorbidities, and laboratory test results is provided in eTable 5.
Degree of Hair Loss at First Visit—In male pediatric patients with AGA, the majority were classified as M type according to the basic and specific classification. Specifically, the main hairloss level in males was concentrated in M1 and M2 (80.0% [48/60]), while specific type F was mainly distributed in F1 and F2 (81.7% [49/60]), and specific type V was mainly distributed in V1 and V2 (80.0% [48/60]). On the other hand, female patients were mainly (87.7% [64/73]) classified as type I or II in the Ludwig scale.
Clinical Features of Trichoscopy Examinations at First Visit—We present the trichoscopic findings of our study regarding hair characteristics, including hair density, hair diameter, terminal hair ratio, and vellus hair ratio, among male and female pediatric participants stratified into 2 age groups: 14 years or younger, and older than 14 but younger than 18 years. In males, those aged 14 years or younger had a lower average hair density than those older than 14 years but thicker hair diameter. Conversely, males aged 14 years and older were more likely to seek treatment of hair loss than those aged 14 years or younger. Among females, those older than 14 years had higher hair density, hair diameter, and terminal hair ratio than those younger than 14 years. Hair trichoscopy characteristics among pediatric patients with AGA in our study population were similar to those of adults with AGA (Figure 3).
Efficacy and Adverse Effects of Topical Minoxidil—There were 56 (42.1%) patients who had used topical minoxidil for more than 6 months: 33 (58.9%) males and 23 (41.1%) females. In terms of efficacy, 51 (91.1%) patients responded positively, demonstrating improved scalp coverage, increased hair density, or greater hair diameter. There were 2 (3.6%) cases of minor adverse reactions: 1 case of scalp itching with increased dandruff that improved with local symptomatic treatment, and 1 case of hirsutism, which improved after discontinuing the drug. Among the 28 (50.0%) pediatric patients who used topical minoxidil for more than 12 months, there were no reported adverse reactions. Overall, topical minoxidil was effective and well tolerated in pediatric patients, with mild adverse reactions.
Comment
In our study, the youngest AGA patient was 10 years old, which is slightly older than a 6-year-old patient reported in the literature.14 Females showed a higher incidence of AGA compared to males, which is consistent with some previous studies14,15 but contradicts the findings of Gonzalez et al16 and Kim et al.17 We speculate that the differences in AGA incidence could be attributed to the diverse genetic background and racial disparities between the populations included in the study by Gonzalez et al16—primarily White patients from Europe and the United States—and our study, which included individuals from East Asia. Furthermore, variations in lifestyle and environment in Europe and the United States vs Asia (eg, dietary habits, stress, environmental pollution) may contribute to the differing sexspecific incidence rates. Additionally, our study showed that female patients tended to experience AGA at a younger age than male patients, as indicated by younger age of disease onset and at the initial visit. These findings are consistent with other studies reporting a slightly younger age of disease onset in female patients.14,16,17 The importance lies in raising awareness among both patients and physicians about early-onset AGA, facilitating earlier detection, diagnosis, and treatment. Furthermore, our study revealed a higher prevalence of a positive family history of AGA in our study population (78.2%) compared to other studies.14 Paternal family history was more commonly observed than maternal history (81.7% and 75.3%, respectively); moreover, 19.5% of patients reported a positive family history of AGA in both parents. Therefore, it is essential to raise awareness among pediatric patients with a positive family history of AGA, as they may experience hair loss at a younger age.
Patients with AGA commonly present with concurrent skin conditions, most notably acne, seborrheic dermatitis, and hirsutism. Therefore, it is important to monitor these associated diseases and adopt appropriate treatments. Moreover, it is worth mentioning that a considerable number of pediatric patients reported experiencing sleep difficulties. It is well known that sleep disturbances can lead to hormonal abnormalities, which are also a risk factor for AGA.18-20 Therefore, further research is needed to investigate whether treating sleep disturbances can delay onset or progression of pediatric AGA. A previous retrospective study reported a PCOS prevalence of 47.4% (9/19) in adolescent females with AGA,16 but our study observed a much lower incidence of 4.5%. This discrepancy may be due to the fact that diagnostic imaging was not required for all female patients suspected of having PCOS in our study, which may have resulted in the exclusion of some undiagnosed PCOS cases from the data analysis.
In our study, a considerable proportion of patients exhibited moderate hair loss at their first visit, and there were differences in hair density and diameter among different age groups, with female patients having finer hair than male patients. Therefore, it is necessary to raise awareness of and perform early diagnosis and treatment of AGA in pediatric patients presenting with hair loss. Upon evaluation of laboratory results, we observed a notable proportion of pediatric patients with AGA who had low levels of vitamin D, SHBG, and ferritin. Notably, female patients were more susceptible to low vitamin D levels compared with males. Screening for these indicators, particularly in female patients, could aid in the diagnosis and treatment of pediatric AGA. Surprisingly, testosterone levels did not show a significant increase in male patients with AGA. Furthermore, only a small percentage of female patients exhibited elevated testosterone levels, indicating that androgens may not play a dominant role in the pathogenesis of male pediatric AGA and that other factors and mechanisms may be involved. Although AGA has been extensively studied in adults, there is limited knowledge about its occurrence and characteristics in children and adolescents. Our study represents one of the few investigations into AGA in this population and is among the largest to explore the clinical features, laboratory testing and results, trichoscopic characteristics, and comorbidities in Chinese pediatric patients with AGA. Our findings offer valuable insights into early clinical characteristics of pediatric AGA in this specific demographic population to inform future research directions and clinical practice guidelines.
Given that we conducted a retrospective study with a relatively small sample size from a single clinic site, the generalizability of our research findings may be limited. In addition, the patients included in our study did not have frequent routine testing for metabolic and hormonal indicators to analyze further correlations between hormonal changes with severity of pediatric AGA. Future research with prospective multicenter designs and larger sample sizes are needed to increase representativeness and generalizability, and comprehensive testing is needed to validate and extend our findings. Furthermore, the psychological impact among pediatric patients with AGA warrants further investigation on early intervention to reduce psychological stress.
Besides enhancing the understanding of AGA in children and adolescents among dermatologists and pediatricians, there is a need for individualized, step-by-step, and comprehensive treatment. Initial assessment generally includes addressing hormonal disorders such as seborrheic dermatitis, folliculitis, PCOS, and acne. Some adult treatments may be effective in pediatric cases. In one study of 15 pediatric patients using minoxidil 5% daily (6 females, 4 males), 4 (66.7%) females had stable alopecia (follow-up, >6 months); 4 (44.4%) males using minoxidil 5% daily and 1 mg finasteride and 5 (55.6%) taking 1 mg of finasteride alone showed hair density gains.16 In another study,21 373 adolescents with AGA (286 boys, 87 girls; age range, 10–17 years) were treated with topical minoxidil solution over an 18-month period, with 95.0% responding positively: 54.0% showed improved scalp coverage, and 41.0% experienced slower hair thinning. Topical minoxidil generally is well tolerated in pediatric patients with no significant impact on blood pressure, pulse rate, or other vital signs.21 The primary adverse reactions to topical minoxidil observed in clinical practice are mild scalp irritation and increased facial hair, which usually resolve upon discontinuation.22 In China, topical minoxidil (available in 2% or 5% concentrations) commonly is used in children and adolescents, with adjustments made based on treatment response and adverse effects. Despite its proven efficacy and tolerability, it is essential that adverse effects be promptly communicated to health care providers for appropriate dosage adjustments, and that concurrent conditions, such as vitamin D and iron deficiencies, be adequately managed. Encouraging patients to adhere to prescribed medications and undergo long-term follow-up typically results in favorable outcomes.
- Jiang W, Yan Q, Tu P, et al. Chinese expert consensus on diagnosis and management of androgenic alopecia in both males and females. Int J Dermatol Venereol. 2019;3:195-202.
- Griggs J, Burroway B, Tosti A. Pediatric androgenetic alopecia: a review. J Am Acad Dermatol. 2021;85:1267-1273.
- Alfredo R, Andrea D, Flavia P. The diagnosis of androgenetic alopecia in children: considerations of pathophysiological plausibility. Australas J Dermatol. 2019;60:279-283.
- Sarkar P, Chakraborti K, Mondal S. Association of metabolic syndrome with early-onset androgenetic alopecia: a case-control study.
Iran J Dermatol. 2022;25:106-110. - Qiu Y, Zhou X, Fu S, et al. Systematic review and meta-analysis of the association between metabolic syndrome and androgenetic alopecia. Acta Derm Venereol. 2022;102:adv000645.
- Memon FH, Rahimoon AG. Androgenetic alopecia as a marker of metabolic syndrome. J Pharm Res Int. 2021;33:146-153.
- Rodríguez-Gutiérrez R, Salcido-Montenegro A, González-González JG. Early clinical expressions of insulin resistance: the real enemy to look for. Diabetes Ther. 2018;9:435-438.
- Wang YX, Chen XW, Wang SB, et al. Association between androgenic alopecia and coronary artery disease: a cross-sectional study of Han Chinese male population. Int J Gen Med. 2021;14:4809-4818.
- Tu YA, Lin SJ, Chen PL, et al. HSD3B1 gene polymorphism and female pattern hair loss in women with polycystic ovary syndrome. J Formos Med Assoc. 2019;118:1225-1231.
- Sanke S, Chander R, Jain A, et al. A comparison of the hormonal profile of early androgenetic alopecia in men with the phenotypic equivalent of polycystic ovarian syndrome in women. JAMA Dermatol. 2016;152:986-991.
- National Health Commission of the People’s Republic of China. (2021). Chinese Guidelines for the Prevention and Control of Overweight and Obesity in Adults.
- Lee WS, Ro BI, Hong SP. A new classification of pattern hair loss that is universal for men and women: basic and specific (BASP) classification. J Am Acad Dermatol. 2007;57:37-46.
- Ludwig, E. Classification of the types of androgenetic alopecia (common baldness) occurring in the female sex. Br J Dermatol. 1977;97:247-254.
- Tosti A, Iorizzo M, Piraccini BM. Androgenetic alopecia in children: report of 20 cases. Br J Dermatol. 2005;152:556-559.
- Özcan D. Pediatric androgenetic alopecia: a retrospective review of clinical characteristics, hormonal assays and metabolic syndrome risk factors in 23 patients. An Bras Dermatol. 2022;97:166-172.
- Gonzalez ME, Cantatore-Francis J, Orlow SJ. Androgenetic alopecia in the paediatric population: a retrospective review of 57 patients. Br J Dermatol. 2010;163:378-385.
- Kim BJ, Kim JY, Eun HC. Androgenetic alopecia in adolescents: a report of 43 cases. J Dermatol. 2006;33:696-699.
- B Liamsombut S, Pomsoong C, Kositkuljorn C. Sleep quality in men with androgenetic alopecia. Sleep Breath. 2023;27:371-378.
- Baik I, Lee S, Thomas RJ. Obstructive sleep apnea, low transferrin saturation levels, and male-pattern baldness. Int J Dermatol. 2019;58:67-74.
- Yi Y, Qiu J, Jia J. Severity of androgenetic alopecia associated with poor sleeping habits and carnivorous eating and junk food consumption—a web-based investigation of male pattern hair loss in China. Dermatol Ther. 2020;33:E13273.
- Price VH. Androgenetic alopecia in adolescents. Cutis. 2003;71:115-121.
- Gomes TF, Soares RO. Pediatric androgenetic alopecia: an updated review. J Dtsch Dermatol Ges. 2023;21:19-25.
Androgenetic alopecia (AGA) is the most common type of hair loss after adolescence, with a high prevalence of 21.3% among males and 6.0% among females in China.1 In men, AGA manifests as diffuse hair loss in the frontal and temporal areas of the scalp; in women, it is characterized by thinning of the hair on the top of the head with a wide part and less recession of the frontal line. Although the specific pathogenesis of AGA still is unclear, it is believed to be related mainly to genetics and androgen levels.1 Androgenetic alopecia is not considered a life-threatening medical condition, but it can have a major impact on patients’ self-esteem and quality of life.
The prevalence of pediatric AGA has been steadily rising over the past few decades and is thought to be correlated to a hyperinsulinemic diet and elevated circulating androgens at younger ages, resulting in early onset in genetically susceptible children and adolescents.2,3 Additionally, studies have shown that early-onset AGA is associated with metabolic syndrome,4-6 which includes conditions such as obesity, insulin resistance, hyperglycemia, and dyslipidemia.7,8 Furthermore, polycystic ovary syndrome (PCOS) is commonly observed in adolescent girls with early-onset AGA. The condition is associated with hormonal imbalances, particularly elevated androgens, which can contribute to the early onset of AGA. In girls, these hormonal changes may accelerate hair thinning and hair loss, making AGA a potential early indicator of underlying PCOS.9,10
Available research on early-onset AGA in pediatric patients is limited, with most studies having a relatively small sample size and generalized findings. Data on pediatric AGA in China is scarce; therefore, the objective of this retrospective study was to analyze the clinical, laboratory, and trichoscopic features of AGA in 133 pediatric patients with AGA who visited the hair disease clinic of the Department of Dermatology at the First Affiliated Hospital of Nanjing Medical University (Nanjing, China), from January 2010 to December 2023.
Methods
Study Population—Pediatric patients with early-onset AGA who were registered for outpatient consultations at the hair disease clinic of the Department of Dermatology at The First Affiliated Hospital of Nanjing Medical University from January 2010 to December 2023 were included. Patients aged 18 years and younger with a definitive diagnosis of AGA were selected for data collection and analysis. Any uncertain information was confirmed through telephone follow-up with patients.
Collection of Demographic Information and Laboratory Tests—Patient demographics and medical history including age, sex, age at disease onset, and duration of AGA were collected from the electronic medical record. Height and weight also were collected to calculate patients’ body mass index (BMI). Detailed laboratory test results were recorded, including assessments of sex hormone—binding globulin (SHBG), vitamin D, testosterone, and ferritin.
Analysis of Comorbidities—Due to the influence of genetic factors on body composition, there are differences in how obesity is defined across racial populations. The World Health Organization international standard defines the term overweight as a BMI greater than 25 and obese as a BMI greater than 30; however, the World Health Organization recommends a lower definition standard for these classifications in the Chinese population. China established specific BMI standards for classification of patients as overweight (24.0.27.9 kg/m2) and obese (≥28 kg/m2).11 During outpatient consultations, a comprehensive medical history was obtained from each patient, including the presence of PCOS, acne, seborrheic dermatitis, hirsutism, and sleeping disorders. During routine outpatient assessments, experienced dermatologists (including W.F.) determined the presence of symptoms and confirmed the diagnosis.
Hair Loss Classification and Trichoscopy—Hair loss patterns for male patients were assessed using the basic and specific classification system, while the Ludwig scale was utilized for female patients.12,13 Trichoscopy was utilized with high-resolution imaging systems and advanced software for image analysis, enabling precise assessment of hair in different scalp regions. Parameters such as hair density, hair diameter, percentage of terminal hairs, and percentage of vellus hair were recorded to monitor changes in hair growth for the patients.
Statistical Analysis—Categorical data were analyzed using the x2 test. A P value less than .05 was considered statistically significant. All statistical analyses were conducted using SPSS software version 26 (IBM).
Results
Patient Characteristics and Hair Loss Patterns—A sample of 133 pediatric patients (60 males, 73 females) who were diagnosed with AGA at the hair disease clinic of the Department of Dermatology at the First Affiliated Hospital of Nanjing Medical University from January 2010 to December 2023 were selected. The mean age of the patients was 15.5 years (range, 10–18 years). The mean age was slightly lower in females compared with males (15.05 vs 16.19 years, respectively). Additionally, females showed earlier onset of the disease, with a mean age at onset of 13.41 years compared to 14.44 years in males. The time between onset of AGA symptoms and first seeking medical care ranged from 4 months to 3 years, with a mean disease duration of 1.72 years. There was no significant difference in the duration of disease between males and females (1.76 and 1.70 years, respectively). Patient characteristics by age group are summarized in eTable 1.
The pediatric patients in our study exhibited hair loss patterns similar to those typically observed in adults. Male patients typically showed diffuse thinning on the crown and varying degrees of temporal thinning, while female patients demonstrated diffuse thinning on the crown with a preserved frontal hairline; however, 5 (8.3%) male patients presented with Christmas tree– like pattern of hair loss with a preserved hairline and a thinning crown (Figures 1 and 2).
Diffuse thinning of the hair on the crown demonstrated a Christmas tree-like pattern with a preserved frontal hairline.
BMI and Comorbidities—Among our study sample, 27.1% (36/133) of patients were identified as overweight or obese. It came to our attention that the prevalence of patients who were overweight and obese was notably higher in patients aged older than 14 but younger than 18 years compared with those aged 14 years or younger (24.1% vs 3.0% [32/133 vs 4/133]). A more detailed analysis of patients who were overweight and obese is outlined in eTable 2.
Seborrheic dermatitis was identified as the most prevalent comorbidity associated with pediatric AGA (51.9% [69/133]), followed by acne (42.8% [57/133]), hirsutism (33.1% [44/133]), and sleep disturbances/insomnia (28.6% [38/133]). The prevalence of these comorbidities varied by age group, with a higher incidence observed among patients aged older than 14 years as compared to those aged 14 years or younger.
Family History of AGA—Our study results indicated that most (78.2% [104/133]) patients had a family history of AGA. Among males and females, 81.7% and 75.3% (49/60 and 55/73) had a positive family history, respectively. Further analysis showed that 43.3% (26/60) of males and 21.9% (16/73) of females reported AGA in their father, while 16.7% (10/60) of males and 35.6% insert (26/73) of females reported AGA in their mother. Both parents were affected in 21.7% (13/60) of male patients and 17.8% (13/73) of female patients (eTable 3).
Related Laboratory Tests of Pediatric Patients With AGA—The results of laboratory testing for vitamin D deficiency, low SHBG, high testosterone, and low ferritin levels in the study sample are outlined in eTable 4. Among the study participants, 15.9% (10/63) of females exhibited increased levels of both free and total testosterone. Low SHBG was observed in 47.1% (56/119) of patients, with a slightly higher proportion in males (48.2% [27/56] than females (46.0% [29/63]). Vitamin D deficiency was prevalent in 60.5% (72/119) of the study population, with a higher incidence rate in females (71.4%[45/63]) compared to males (48.2%[27/56]). Moreover, 21.8% (26/119) of pediatric patients had low ferritin levels, with a higher incidence rate in females (33.3%[21/63]) compared to males (8.9%[5/56]).
Female Patients With PCOS—In our study, 6 (8.2%) female patients with AGA had been diagnosed with PCOS prior to their referral to the First Affiliated Hospital of Nanjing Medical University. Information regarding their age at treatment, hair loss grade, comorbidities, and laboratory test results is provided in eTable 5.
Degree of Hair Loss at First Visit—In male pediatric patients with AGA, the majority were classified as M type according to the basic and specific classification. Specifically, the main hairloss level in males was concentrated in M1 and M2 (80.0% [48/60]), while specific type F was mainly distributed in F1 and F2 (81.7% [49/60]), and specific type V was mainly distributed in V1 and V2 (80.0% [48/60]). On the other hand, female patients were mainly (87.7% [64/73]) classified as type I or II in the Ludwig scale.
Clinical Features of Trichoscopy Examinations at First Visit—We present the trichoscopic findings of our study regarding hair characteristics, including hair density, hair diameter, terminal hair ratio, and vellus hair ratio, among male and female pediatric participants stratified into 2 age groups: 14 years or younger, and older than 14 but younger than 18 years. In males, those aged 14 years or younger had a lower average hair density than those older than 14 years but thicker hair diameter. Conversely, males aged 14 years and older were more likely to seek treatment of hair loss than those aged 14 years or younger. Among females, those older than 14 years had higher hair density, hair diameter, and terminal hair ratio than those younger than 14 years. Hair trichoscopy characteristics among pediatric patients with AGA in our study population were similar to those of adults with AGA (Figure 3).
Efficacy and Adverse Effects of Topical Minoxidil—There were 56 (42.1%) patients who had used topical minoxidil for more than 6 months: 33 (58.9%) males and 23 (41.1%) females. In terms of efficacy, 51 (91.1%) patients responded positively, demonstrating improved scalp coverage, increased hair density, or greater hair diameter. There were 2 (3.6%) cases of minor adverse reactions: 1 case of scalp itching with increased dandruff that improved with local symptomatic treatment, and 1 case of hirsutism, which improved after discontinuing the drug. Among the 28 (50.0%) pediatric patients who used topical minoxidil for more than 12 months, there were no reported adverse reactions. Overall, topical minoxidil was effective and well tolerated in pediatric patients, with mild adverse reactions.
Comment
In our study, the youngest AGA patient was 10 years old, which is slightly older than a 6-year-old patient reported in the literature.14 Females showed a higher incidence of AGA compared to males, which is consistent with some previous studies14,15 but contradicts the findings of Gonzalez et al16 and Kim et al.17 We speculate that the differences in AGA incidence could be attributed to the diverse genetic background and racial disparities between the populations included in the study by Gonzalez et al16—primarily White patients from Europe and the United States—and our study, which included individuals from East Asia. Furthermore, variations in lifestyle and environment in Europe and the United States vs Asia (eg, dietary habits, stress, environmental pollution) may contribute to the differing sexspecific incidence rates. Additionally, our study showed that female patients tended to experience AGA at a younger age than male patients, as indicated by younger age of disease onset and at the initial visit. These findings are consistent with other studies reporting a slightly younger age of disease onset in female patients.14,16,17 The importance lies in raising awareness among both patients and physicians about early-onset AGA, facilitating earlier detection, diagnosis, and treatment. Furthermore, our study revealed a higher prevalence of a positive family history of AGA in our study population (78.2%) compared to other studies.14 Paternal family history was more commonly observed than maternal history (81.7% and 75.3%, respectively); moreover, 19.5% of patients reported a positive family history of AGA in both parents. Therefore, it is essential to raise awareness among pediatric patients with a positive family history of AGA, as they may experience hair loss at a younger age.
Patients with AGA commonly present with concurrent skin conditions, most notably acne, seborrheic dermatitis, and hirsutism. Therefore, it is important to monitor these associated diseases and adopt appropriate treatments. Moreover, it is worth mentioning that a considerable number of pediatric patients reported experiencing sleep difficulties. It is well known that sleep disturbances can lead to hormonal abnormalities, which are also a risk factor for AGA.18-20 Therefore, further research is needed to investigate whether treating sleep disturbances can delay onset or progression of pediatric AGA. A previous retrospective study reported a PCOS prevalence of 47.4% (9/19) in adolescent females with AGA,16 but our study observed a much lower incidence of 4.5%. This discrepancy may be due to the fact that diagnostic imaging was not required for all female patients suspected of having PCOS in our study, which may have resulted in the exclusion of some undiagnosed PCOS cases from the data analysis.
In our study, a considerable proportion of patients exhibited moderate hair loss at their first visit, and there were differences in hair density and diameter among different age groups, with female patients having finer hair than male patients. Therefore, it is necessary to raise awareness of and perform early diagnosis and treatment of AGA in pediatric patients presenting with hair loss. Upon evaluation of laboratory results, we observed a notable proportion of pediatric patients with AGA who had low levels of vitamin D, SHBG, and ferritin. Notably, female patients were more susceptible to low vitamin D levels compared with males. Screening for these indicators, particularly in female patients, could aid in the diagnosis and treatment of pediatric AGA. Surprisingly, testosterone levels did not show a significant increase in male patients with AGA. Furthermore, only a small percentage of female patients exhibited elevated testosterone levels, indicating that androgens may not play a dominant role in the pathogenesis of male pediatric AGA and that other factors and mechanisms may be involved. Although AGA has been extensively studied in adults, there is limited knowledge about its occurrence and characteristics in children and adolescents. Our study represents one of the few investigations into AGA in this population and is among the largest to explore the clinical features, laboratory testing and results, trichoscopic characteristics, and comorbidities in Chinese pediatric patients with AGA. Our findings offer valuable insights into early clinical characteristics of pediatric AGA in this specific demographic population to inform future research directions and clinical practice guidelines.
Given that we conducted a retrospective study with a relatively small sample size from a single clinic site, the generalizability of our research findings may be limited. In addition, the patients included in our study did not have frequent routine testing for metabolic and hormonal indicators to analyze further correlations between hormonal changes with severity of pediatric AGA. Future research with prospective multicenter designs and larger sample sizes are needed to increase representativeness and generalizability, and comprehensive testing is needed to validate and extend our findings. Furthermore, the psychological impact among pediatric patients with AGA warrants further investigation on early intervention to reduce psychological stress.
Besides enhancing the understanding of AGA in children and adolescents among dermatologists and pediatricians, there is a need for individualized, step-by-step, and comprehensive treatment. Initial assessment generally includes addressing hormonal disorders such as seborrheic dermatitis, folliculitis, PCOS, and acne. Some adult treatments may be effective in pediatric cases. In one study of 15 pediatric patients using minoxidil 5% daily (6 females, 4 males), 4 (66.7%) females had stable alopecia (follow-up, >6 months); 4 (44.4%) males using minoxidil 5% daily and 1 mg finasteride and 5 (55.6%) taking 1 mg of finasteride alone showed hair density gains.16 In another study,21 373 adolescents with AGA (286 boys, 87 girls; age range, 10–17 years) were treated with topical minoxidil solution over an 18-month period, with 95.0% responding positively: 54.0% showed improved scalp coverage, and 41.0% experienced slower hair thinning. Topical minoxidil generally is well tolerated in pediatric patients with no significant impact on blood pressure, pulse rate, or other vital signs.21 The primary adverse reactions to topical minoxidil observed in clinical practice are mild scalp irritation and increased facial hair, which usually resolve upon discontinuation.22 In China, topical minoxidil (available in 2% or 5% concentrations) commonly is used in children and adolescents, with adjustments made based on treatment response and adverse effects. Despite its proven efficacy and tolerability, it is essential that adverse effects be promptly communicated to health care providers for appropriate dosage adjustments, and that concurrent conditions, such as vitamin D and iron deficiencies, be adequately managed. Encouraging patients to adhere to prescribed medications and undergo long-term follow-up typically results in favorable outcomes.
Androgenetic alopecia (AGA) is the most common type of hair loss after adolescence, with a high prevalence of 21.3% among males and 6.0% among females in China.1 In men, AGA manifests as diffuse hair loss in the frontal and temporal areas of the scalp; in women, it is characterized by thinning of the hair on the top of the head with a wide part and less recession of the frontal line. Although the specific pathogenesis of AGA still is unclear, it is believed to be related mainly to genetics and androgen levels.1 Androgenetic alopecia is not considered a life-threatening medical condition, but it can have a major impact on patients’ self-esteem and quality of life.
The prevalence of pediatric AGA has been steadily rising over the past few decades and is thought to be correlated to a hyperinsulinemic diet and elevated circulating androgens at younger ages, resulting in early onset in genetically susceptible children and adolescents.2,3 Additionally, studies have shown that early-onset AGA is associated with metabolic syndrome,4-6 which includes conditions such as obesity, insulin resistance, hyperglycemia, and dyslipidemia.7,8 Furthermore, polycystic ovary syndrome (PCOS) is commonly observed in adolescent girls with early-onset AGA. The condition is associated with hormonal imbalances, particularly elevated androgens, which can contribute to the early onset of AGA. In girls, these hormonal changes may accelerate hair thinning and hair loss, making AGA a potential early indicator of underlying PCOS.9,10
Available research on early-onset AGA in pediatric patients is limited, with most studies having a relatively small sample size and generalized findings. Data on pediatric AGA in China is scarce; therefore, the objective of this retrospective study was to analyze the clinical, laboratory, and trichoscopic features of AGA in 133 pediatric patients with AGA who visited the hair disease clinic of the Department of Dermatology at the First Affiliated Hospital of Nanjing Medical University (Nanjing, China), from January 2010 to December 2023.
Methods
Study Population—Pediatric patients with early-onset AGA who were registered for outpatient consultations at the hair disease clinic of the Department of Dermatology at The First Affiliated Hospital of Nanjing Medical University from January 2010 to December 2023 were included. Patients aged 18 years and younger with a definitive diagnosis of AGA were selected for data collection and analysis. Any uncertain information was confirmed through telephone follow-up with patients.
Collection of Demographic Information and Laboratory Tests—Patient demographics and medical history including age, sex, age at disease onset, and duration of AGA were collected from the electronic medical record. Height and weight also were collected to calculate patients’ body mass index (BMI). Detailed laboratory test results were recorded, including assessments of sex hormone—binding globulin (SHBG), vitamin D, testosterone, and ferritin.
Analysis of Comorbidities—Due to the influence of genetic factors on body composition, there are differences in how obesity is defined across racial populations. The World Health Organization international standard defines the term overweight as a BMI greater than 25 and obese as a BMI greater than 30; however, the World Health Organization recommends a lower definition standard for these classifications in the Chinese population. China established specific BMI standards for classification of patients as overweight (24.0.27.9 kg/m2) and obese (≥28 kg/m2).11 During outpatient consultations, a comprehensive medical history was obtained from each patient, including the presence of PCOS, acne, seborrheic dermatitis, hirsutism, and sleeping disorders. During routine outpatient assessments, experienced dermatologists (including W.F.) determined the presence of symptoms and confirmed the diagnosis.
Hair Loss Classification and Trichoscopy—Hair loss patterns for male patients were assessed using the basic and specific classification system, while the Ludwig scale was utilized for female patients.12,13 Trichoscopy was utilized with high-resolution imaging systems and advanced software for image analysis, enabling precise assessment of hair in different scalp regions. Parameters such as hair density, hair diameter, percentage of terminal hairs, and percentage of vellus hair were recorded to monitor changes in hair growth for the patients.
Statistical Analysis—Categorical data were analyzed using the x2 test. A P value less than .05 was considered statistically significant. All statistical analyses were conducted using SPSS software version 26 (IBM).
Results
Patient Characteristics and Hair Loss Patterns—A sample of 133 pediatric patients (60 males, 73 females) who were diagnosed with AGA at the hair disease clinic of the Department of Dermatology at the First Affiliated Hospital of Nanjing Medical University from January 2010 to December 2023 were selected. The mean age of the patients was 15.5 years (range, 10–18 years). The mean age was slightly lower in females compared with males (15.05 vs 16.19 years, respectively). Additionally, females showed earlier onset of the disease, with a mean age at onset of 13.41 years compared to 14.44 years in males. The time between onset of AGA symptoms and first seeking medical care ranged from 4 months to 3 years, with a mean disease duration of 1.72 years. There was no significant difference in the duration of disease between males and females (1.76 and 1.70 years, respectively). Patient characteristics by age group are summarized in eTable 1.
The pediatric patients in our study exhibited hair loss patterns similar to those typically observed in adults. Male patients typically showed diffuse thinning on the crown and varying degrees of temporal thinning, while female patients demonstrated diffuse thinning on the crown with a preserved frontal hairline; however, 5 (8.3%) male patients presented with Christmas tree– like pattern of hair loss with a preserved hairline and a thinning crown (Figures 1 and 2).
Diffuse thinning of the hair on the crown demonstrated a Christmas tree-like pattern with a preserved frontal hairline.
BMI and Comorbidities—Among our study sample, 27.1% (36/133) of patients were identified as overweight or obese. It came to our attention that the prevalence of patients who were overweight and obese was notably higher in patients aged older than 14 but younger than 18 years compared with those aged 14 years or younger (24.1% vs 3.0% [32/133 vs 4/133]). A more detailed analysis of patients who were overweight and obese is outlined in eTable 2.
Seborrheic dermatitis was identified as the most prevalent comorbidity associated with pediatric AGA (51.9% [69/133]), followed by acne (42.8% [57/133]), hirsutism (33.1% [44/133]), and sleep disturbances/insomnia (28.6% [38/133]). The prevalence of these comorbidities varied by age group, with a higher incidence observed among patients aged older than 14 years as compared to those aged 14 years or younger.
Family History of AGA—Our study results indicated that most (78.2% [104/133]) patients had a family history of AGA. Among males and females, 81.7% and 75.3% (49/60 and 55/73) had a positive family history, respectively. Further analysis showed that 43.3% (26/60) of males and 21.9% (16/73) of females reported AGA in their father, while 16.7% (10/60) of males and 35.6% insert (26/73) of females reported AGA in their mother. Both parents were affected in 21.7% (13/60) of male patients and 17.8% (13/73) of female patients (eTable 3).
Related Laboratory Tests of Pediatric Patients With AGA—The results of laboratory testing for vitamin D deficiency, low SHBG, high testosterone, and low ferritin levels in the study sample are outlined in eTable 4. Among the study participants, 15.9% (10/63) of females exhibited increased levels of both free and total testosterone. Low SHBG was observed in 47.1% (56/119) of patients, with a slightly higher proportion in males (48.2% [27/56] than females (46.0% [29/63]). Vitamin D deficiency was prevalent in 60.5% (72/119) of the study population, with a higher incidence rate in females (71.4%[45/63]) compared to males (48.2%[27/56]). Moreover, 21.8% (26/119) of pediatric patients had low ferritin levels, with a higher incidence rate in females (33.3%[21/63]) compared to males (8.9%[5/56]).
Female Patients With PCOS—In our study, 6 (8.2%) female patients with AGA had been diagnosed with PCOS prior to their referral to the First Affiliated Hospital of Nanjing Medical University. Information regarding their age at treatment, hair loss grade, comorbidities, and laboratory test results is provided in eTable 5.
Degree of Hair Loss at First Visit—In male pediatric patients with AGA, the majority were classified as M type according to the basic and specific classification. Specifically, the main hairloss level in males was concentrated in M1 and M2 (80.0% [48/60]), while specific type F was mainly distributed in F1 and F2 (81.7% [49/60]), and specific type V was mainly distributed in V1 and V2 (80.0% [48/60]). On the other hand, female patients were mainly (87.7% [64/73]) classified as type I or II in the Ludwig scale.
Clinical Features of Trichoscopy Examinations at First Visit—We present the trichoscopic findings of our study regarding hair characteristics, including hair density, hair diameter, terminal hair ratio, and vellus hair ratio, among male and female pediatric participants stratified into 2 age groups: 14 years or younger, and older than 14 but younger than 18 years. In males, those aged 14 years or younger had a lower average hair density than those older than 14 years but thicker hair diameter. Conversely, males aged 14 years and older were more likely to seek treatment of hair loss than those aged 14 years or younger. Among females, those older than 14 years had higher hair density, hair diameter, and terminal hair ratio than those younger than 14 years. Hair trichoscopy characteristics among pediatric patients with AGA in our study population were similar to those of adults with AGA (Figure 3).
Efficacy and Adverse Effects of Topical Minoxidil—There were 56 (42.1%) patients who had used topical minoxidil for more than 6 months: 33 (58.9%) males and 23 (41.1%) females. In terms of efficacy, 51 (91.1%) patients responded positively, demonstrating improved scalp coverage, increased hair density, or greater hair diameter. There were 2 (3.6%) cases of minor adverse reactions: 1 case of scalp itching with increased dandruff that improved with local symptomatic treatment, and 1 case of hirsutism, which improved after discontinuing the drug. Among the 28 (50.0%) pediatric patients who used topical minoxidil for more than 12 months, there were no reported adverse reactions. Overall, topical minoxidil was effective and well tolerated in pediatric patients, with mild adverse reactions.
Comment
In our study, the youngest AGA patient was 10 years old, which is slightly older than a 6-year-old patient reported in the literature.14 Females showed a higher incidence of AGA compared to males, which is consistent with some previous studies14,15 but contradicts the findings of Gonzalez et al16 and Kim et al.17 We speculate that the differences in AGA incidence could be attributed to the diverse genetic background and racial disparities between the populations included in the study by Gonzalez et al16—primarily White patients from Europe and the United States—and our study, which included individuals from East Asia. Furthermore, variations in lifestyle and environment in Europe and the United States vs Asia (eg, dietary habits, stress, environmental pollution) may contribute to the differing sexspecific incidence rates. Additionally, our study showed that female patients tended to experience AGA at a younger age than male patients, as indicated by younger age of disease onset and at the initial visit. These findings are consistent with other studies reporting a slightly younger age of disease onset in female patients.14,16,17 The importance lies in raising awareness among both patients and physicians about early-onset AGA, facilitating earlier detection, diagnosis, and treatment. Furthermore, our study revealed a higher prevalence of a positive family history of AGA in our study population (78.2%) compared to other studies.14 Paternal family history was more commonly observed than maternal history (81.7% and 75.3%, respectively); moreover, 19.5% of patients reported a positive family history of AGA in both parents. Therefore, it is essential to raise awareness among pediatric patients with a positive family history of AGA, as they may experience hair loss at a younger age.
Patients with AGA commonly present with concurrent skin conditions, most notably acne, seborrheic dermatitis, and hirsutism. Therefore, it is important to monitor these associated diseases and adopt appropriate treatments. Moreover, it is worth mentioning that a considerable number of pediatric patients reported experiencing sleep difficulties. It is well known that sleep disturbances can lead to hormonal abnormalities, which are also a risk factor for AGA.18-20 Therefore, further research is needed to investigate whether treating sleep disturbances can delay onset or progression of pediatric AGA. A previous retrospective study reported a PCOS prevalence of 47.4% (9/19) in adolescent females with AGA,16 but our study observed a much lower incidence of 4.5%. This discrepancy may be due to the fact that diagnostic imaging was not required for all female patients suspected of having PCOS in our study, which may have resulted in the exclusion of some undiagnosed PCOS cases from the data analysis.
In our study, a considerable proportion of patients exhibited moderate hair loss at their first visit, and there were differences in hair density and diameter among different age groups, with female patients having finer hair than male patients. Therefore, it is necessary to raise awareness of and perform early diagnosis and treatment of AGA in pediatric patients presenting with hair loss. Upon evaluation of laboratory results, we observed a notable proportion of pediatric patients with AGA who had low levels of vitamin D, SHBG, and ferritin. Notably, female patients were more susceptible to low vitamin D levels compared with males. Screening for these indicators, particularly in female patients, could aid in the diagnosis and treatment of pediatric AGA. Surprisingly, testosterone levels did not show a significant increase in male patients with AGA. Furthermore, only a small percentage of female patients exhibited elevated testosterone levels, indicating that androgens may not play a dominant role in the pathogenesis of male pediatric AGA and that other factors and mechanisms may be involved. Although AGA has been extensively studied in adults, there is limited knowledge about its occurrence and characteristics in children and adolescents. Our study represents one of the few investigations into AGA in this population and is among the largest to explore the clinical features, laboratory testing and results, trichoscopic characteristics, and comorbidities in Chinese pediatric patients with AGA. Our findings offer valuable insights into early clinical characteristics of pediatric AGA in this specific demographic population to inform future research directions and clinical practice guidelines.
Given that we conducted a retrospective study with a relatively small sample size from a single clinic site, the generalizability of our research findings may be limited. In addition, the patients included in our study did not have frequent routine testing for metabolic and hormonal indicators to analyze further correlations between hormonal changes with severity of pediatric AGA. Future research with prospective multicenter designs and larger sample sizes are needed to increase representativeness and generalizability, and comprehensive testing is needed to validate and extend our findings. Furthermore, the psychological impact among pediatric patients with AGA warrants further investigation on early intervention to reduce psychological stress.
Besides enhancing the understanding of AGA in children and adolescents among dermatologists and pediatricians, there is a need for individualized, step-by-step, and comprehensive treatment. Initial assessment generally includes addressing hormonal disorders such as seborrheic dermatitis, folliculitis, PCOS, and acne. Some adult treatments may be effective in pediatric cases. In one study of 15 pediatric patients using minoxidil 5% daily (6 females, 4 males), 4 (66.7%) females had stable alopecia (follow-up, >6 months); 4 (44.4%) males using minoxidil 5% daily and 1 mg finasteride and 5 (55.6%) taking 1 mg of finasteride alone showed hair density gains.16 In another study,21 373 adolescents with AGA (286 boys, 87 girls; age range, 10–17 years) were treated with topical minoxidil solution over an 18-month period, with 95.0% responding positively: 54.0% showed improved scalp coverage, and 41.0% experienced slower hair thinning. Topical minoxidil generally is well tolerated in pediatric patients with no significant impact on blood pressure, pulse rate, or other vital signs.21 The primary adverse reactions to topical minoxidil observed in clinical practice are mild scalp irritation and increased facial hair, which usually resolve upon discontinuation.22 In China, topical minoxidil (available in 2% or 5% concentrations) commonly is used in children and adolescents, with adjustments made based on treatment response and adverse effects. Despite its proven efficacy and tolerability, it is essential that adverse effects be promptly communicated to health care providers for appropriate dosage adjustments, and that concurrent conditions, such as vitamin D and iron deficiencies, be adequately managed. Encouraging patients to adhere to prescribed medications and undergo long-term follow-up typically results in favorable outcomes.
- Jiang W, Yan Q, Tu P, et al. Chinese expert consensus on diagnosis and management of androgenic alopecia in both males and females. Int J Dermatol Venereol. 2019;3:195-202.
- Griggs J, Burroway B, Tosti A. Pediatric androgenetic alopecia: a review. J Am Acad Dermatol. 2021;85:1267-1273.
- Alfredo R, Andrea D, Flavia P. The diagnosis of androgenetic alopecia in children: considerations of pathophysiological plausibility. Australas J Dermatol. 2019;60:279-283.
- Sarkar P, Chakraborti K, Mondal S. Association of metabolic syndrome with early-onset androgenetic alopecia: a case-control study.
Iran J Dermatol. 2022;25:106-110. - Qiu Y, Zhou X, Fu S, et al. Systematic review and meta-analysis of the association between metabolic syndrome and androgenetic alopecia. Acta Derm Venereol. 2022;102:adv000645.
- Memon FH, Rahimoon AG. Androgenetic alopecia as a marker of metabolic syndrome. J Pharm Res Int. 2021;33:146-153.
- Rodríguez-Gutiérrez R, Salcido-Montenegro A, González-González JG. Early clinical expressions of insulin resistance: the real enemy to look for. Diabetes Ther. 2018;9:435-438.
- Wang YX, Chen XW, Wang SB, et al. Association between androgenic alopecia and coronary artery disease: a cross-sectional study of Han Chinese male population. Int J Gen Med. 2021;14:4809-4818.
- Tu YA, Lin SJ, Chen PL, et al. HSD3B1 gene polymorphism and female pattern hair loss in women with polycystic ovary syndrome. J Formos Med Assoc. 2019;118:1225-1231.
- Sanke S, Chander R, Jain A, et al. A comparison of the hormonal profile of early androgenetic alopecia in men with the phenotypic equivalent of polycystic ovarian syndrome in women. JAMA Dermatol. 2016;152:986-991.
- National Health Commission of the People’s Republic of China. (2021). Chinese Guidelines for the Prevention and Control of Overweight and Obesity in Adults.
- Lee WS, Ro BI, Hong SP. A new classification of pattern hair loss that is universal for men and women: basic and specific (BASP) classification. J Am Acad Dermatol. 2007;57:37-46.
- Ludwig, E. Classification of the types of androgenetic alopecia (common baldness) occurring in the female sex. Br J Dermatol. 1977;97:247-254.
- Tosti A, Iorizzo M, Piraccini BM. Androgenetic alopecia in children: report of 20 cases. Br J Dermatol. 2005;152:556-559.
- Özcan D. Pediatric androgenetic alopecia: a retrospective review of clinical characteristics, hormonal assays and metabolic syndrome risk factors in 23 patients. An Bras Dermatol. 2022;97:166-172.
- Gonzalez ME, Cantatore-Francis J, Orlow SJ. Androgenetic alopecia in the paediatric population: a retrospective review of 57 patients. Br J Dermatol. 2010;163:378-385.
- Kim BJ, Kim JY, Eun HC. Androgenetic alopecia in adolescents: a report of 43 cases. J Dermatol. 2006;33:696-699.
- B Liamsombut S, Pomsoong C, Kositkuljorn C. Sleep quality in men with androgenetic alopecia. Sleep Breath. 2023;27:371-378.
- Baik I, Lee S, Thomas RJ. Obstructive sleep apnea, low transferrin saturation levels, and male-pattern baldness. Int J Dermatol. 2019;58:67-74.
- Yi Y, Qiu J, Jia J. Severity of androgenetic alopecia associated with poor sleeping habits and carnivorous eating and junk food consumption—a web-based investigation of male pattern hair loss in China. Dermatol Ther. 2020;33:E13273.
- Price VH. Androgenetic alopecia in adolescents. Cutis. 2003;71:115-121.
- Gomes TF, Soares RO. Pediatric androgenetic alopecia: an updated review. J Dtsch Dermatol Ges. 2023;21:19-25.
- Jiang W, Yan Q, Tu P, et al. Chinese expert consensus on diagnosis and management of androgenic alopecia in both males and females. Int J Dermatol Venereol. 2019;3:195-202.
- Griggs J, Burroway B, Tosti A. Pediatric androgenetic alopecia: a review. J Am Acad Dermatol. 2021;85:1267-1273.
- Alfredo R, Andrea D, Flavia P. The diagnosis of androgenetic alopecia in children: considerations of pathophysiological plausibility. Australas J Dermatol. 2019;60:279-283.
- Sarkar P, Chakraborti K, Mondal S. Association of metabolic syndrome with early-onset androgenetic alopecia: a case-control study.
Iran J Dermatol. 2022;25:106-110. - Qiu Y, Zhou X, Fu S, et al. Systematic review and meta-analysis of the association between metabolic syndrome and androgenetic alopecia. Acta Derm Venereol. 2022;102:adv000645.
- Memon FH, Rahimoon AG. Androgenetic alopecia as a marker of metabolic syndrome. J Pharm Res Int. 2021;33:146-153.
- Rodríguez-Gutiérrez R, Salcido-Montenegro A, González-González JG. Early clinical expressions of insulin resistance: the real enemy to look for. Diabetes Ther. 2018;9:435-438.
- Wang YX, Chen XW, Wang SB, et al. Association between androgenic alopecia and coronary artery disease: a cross-sectional study of Han Chinese male population. Int J Gen Med. 2021;14:4809-4818.
- Tu YA, Lin SJ, Chen PL, et al. HSD3B1 gene polymorphism and female pattern hair loss in women with polycystic ovary syndrome. J Formos Med Assoc. 2019;118:1225-1231.
- Sanke S, Chander R, Jain A, et al. A comparison of the hormonal profile of early androgenetic alopecia in men with the phenotypic equivalent of polycystic ovarian syndrome in women. JAMA Dermatol. 2016;152:986-991.
- National Health Commission of the People’s Republic of China. (2021). Chinese Guidelines for the Prevention and Control of Overweight and Obesity in Adults.
- Lee WS, Ro BI, Hong SP. A new classification of pattern hair loss that is universal for men and women: basic and specific (BASP) classification. J Am Acad Dermatol. 2007;57:37-46.
- Ludwig, E. Classification of the types of androgenetic alopecia (common baldness) occurring in the female sex. Br J Dermatol. 1977;97:247-254.
- Tosti A, Iorizzo M, Piraccini BM. Androgenetic alopecia in children: report of 20 cases. Br J Dermatol. 2005;152:556-559.
- Özcan D. Pediatric androgenetic alopecia: a retrospective review of clinical characteristics, hormonal assays and metabolic syndrome risk factors in 23 patients. An Bras Dermatol. 2022;97:166-172.
- Gonzalez ME, Cantatore-Francis J, Orlow SJ. Androgenetic alopecia in the paediatric population: a retrospective review of 57 patients. Br J Dermatol. 2010;163:378-385.
- Kim BJ, Kim JY, Eun HC. Androgenetic alopecia in adolescents: a report of 43 cases. J Dermatol. 2006;33:696-699.
- B Liamsombut S, Pomsoong C, Kositkuljorn C. Sleep quality in men with androgenetic alopecia. Sleep Breath. 2023;27:371-378.
- Baik I, Lee S, Thomas RJ. Obstructive sleep apnea, low transferrin saturation levels, and male-pattern baldness. Int J Dermatol. 2019;58:67-74.
- Yi Y, Qiu J, Jia J. Severity of androgenetic alopecia associated with poor sleeping habits and carnivorous eating and junk food consumption—a web-based investigation of male pattern hair loss in China. Dermatol Ther. 2020;33:E13273.
- Price VH. Androgenetic alopecia in adolescents. Cutis. 2003;71:115-121.
- Gomes TF, Soares RO. Pediatric androgenetic alopecia: an updated review. J Dtsch Dermatol Ges. 2023;21:19-25.
Clinical, Laboratory, and Trichoscopic Features of Pediatric Androgenetic Alopecia
Clinical, Laboratory, and Trichoscopic Features of Pediatric Androgenetic Alopecia
PRACTICE POINTS
- Early identification of androgenetic alopecia (AGA) is key in pediatric patients, especially in those with a family history of AGA and comorbidities such as seborrheic dermatitis, acne, or sleep disturbances.
- It is important to evaluate pediatric patients with AGA for hormonal imbalances and deficiencies in vitamin D and iron to guide treatment.
- Use targeted therapies such as topical minoxidil to treat pediatric AGA while also monitoring for adverse effects. For optimal outcomes, encourage consistent medication use and regular follow-up.
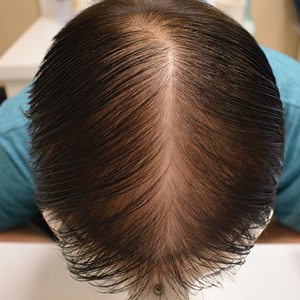
Satisfaction With Department of Veterans Affairs Prosthetics and Support Services as Reported by Women and Men Veterans
Satisfaction With Department of Veterans Affairs Prosthetics and Support Services as Reported by Women and Men Veterans
Limb loss is a significant and growing concern in the United States. Nearly 2 million Americans are living with limb loss, and up to 185,000 people undergo amputations annually.1-4 Of these patients, about 35% are women.5 The Veterans Health Administration (VHA) provides about 10% of US amputations.6-8 Between 2015 and 2019, the number of prosthetic devices provided to female veterans increased from 3.3 million to 4.6 million.5,9,10
Previous research identified disparities in prosthetic care between men and women, both within and outside the VHA. These disparities include slower prosthesis prescription and receipt among women, in addition to differences in self-reported mobility, satisfaction, rates of prosthesis rejection, and challenges related to prosthesis appearance and fit.5,10,11 Recent studies suggest women tend to have worse outcomes following amputation, and are underrepresented in amputation research.12,13 However, these disparities are poorly described in a large, national sample. Because women represent a growing portion of patients with limb loss in the VHA, understanding their needs is critical.14
The Johnny Isakson and David P. Roe, MD Veterans Health Care and Benefits Improvement Act of 2020 was enacted, in part, to improve the care provided to women veterans.15 The law required the VHA to conduct a survey of ≥ 50,000 veterans to assess the satisfaction of women veterans with prostheses provided by the VHA. To comply with this legislation and understand how women veterans rate their prostheses and related care in the VHA, the US Department of Veterans Affairs (VA) Center for Collaborative Evaluation (VACE) conducted a large national survey of veterans with limb loss that oversampled women veterans. This article describes the survey results, including characteristics of female veterans with limb loss receiving care from the VHA, assesses their satisfaction with prostheses and prosthetic care, and highlights where their responses differ from those of male veterans.
Methods
We conducted a cross-sectional, mixedmode survey of eligible amputees in the VHA Support Service Capital Assets Amputee Data Cube. We identified a cohort of veterans with any major amputation (above the ankle or wrist) or partial hand or foot amputation who received VHA care between October 1, 2019, and September 30, 2020. The final cohort yielded 46,646 potentially eligible veterans. Thirty-three had invalid contact information, leaving 46,613 veterans who were asked to participate, including 1356 women.
Survey
We created a survey instrument de novo that included questions from validated instruments, including the Trinity Amputation Prosthesis and Experience Scales to assess prosthetic device satisfaction, the Prosthesis Evaluation Questionnaire to assess quality of life (QOL) satisfaction, and the Orthotics Prosthetics Users Survey to assess prosthesis-related care satisfaction. 16-18 Additional questions were incorporated from a survey of veterans with upper limb amputation to assess the importance of cosmetic considerations related to the prosthesis and comfort with prosthesis use in intimate relationships.19 Questions were also included to assess amputation type, year of amputation, if a prosthesis was currently used, reasons for ceasing use of a prosthesis, reasons for never using a prosthesis, the types of prostheses used, intensity of prosthesis use, satisfaction with time required to receive a prosthetic limb, and if the prosthesis reflected the veteran’s selfidentified gender. Veterans were asked to answer questions based on their most recent amputation.
We tested the survey using cognitive interviews with 6 veterans to refine the survey and better understand how veterans interpreted the questions. Pilot testers completed the survey and participated in individual interviews with experienced interviewers (CL and RRK) to describe how they selected their responses.20 This feedback was used to refine the survey. The online survey was programmed using Qualtrics Software and manually translated into Spanish.
Given the multimodal design, surveys were distributed by email, text message, and US Postal Service (USPS). Surveys were emailed to all veterans for whom a valid email address was available. If emails were undeliverable, veterans were contacted via text message or the USPS. Surveys were distributed by text message to all veterans without an email address but with a cellphone number. We were unable to consistently identify invalid numbers among all text message recipients. Invitations with a survey URL and QR code were sent via USPS to veterans who had no valid email address or cellphone number. Targeted efforts were made to increase the response rate for women. A random sample of 200 women who had not completed the survey 2 weeks prior to the closing date (15% of women in sample) was selected to receive personal phone calls. Another random sample of 400 women was selected to receive personalized outreach emails. The survey data were confidential, and responses could not be traced to identifying information.
Data Analyses
We conducted a descriptive analysis, including percentages and means for responses to variables focused on describing amputation characteristics, prosthesis characteristics, and QOL. All data, including missing values, were used to document the percentage of respondents for each question. Removing missing data from the denominator when calculating percentages could introduce bias to the analysis because we cannot be certain data are missing at random. Missing variables were removed to avoid underinflation of mean scores.
We compared responses across 2 groups: individuals who self-identified as men and individuals who self-identified as women. For each question, we assessed whether each of these groups differed significantly from the remaining sample. For example, we examined whether the percentage of men who answered affirmatively to a question was significantly higher or lower than that of individuals not identifying as male, and whether the percentage of women who answered affirmatively was significantly higher or lower than that of individuals not identifying as female. We utilized x2 tests to determine significant differences for percentage calculations and t tests to determine significant differences in means across gender.
Since conducting multiple comparisons within a dataset may result in inflating statistical significance (type 1 errors), we used a more conservative estimate of statistical significance (α = 0.01) and high significance (α = 0.001). This study was deemed quality improvement by the VHA Rehabilitation and Prosthetic Services (12RPS) and acknowledged by the VA Research Office at Eastern Colorado Health Care System and was not subject to institutional review board review.
Results
Surveys were distributed to 46,613 veterans and were completed by 4981 respondents for a 10.7% overall response rate. Survey respondents were generally similar to the eligible population invited to participate, but the proportion of women who completed the survey was higher than the proportion of women eligible to participate (2.0% of eligible population vs 16.7% of respondents), likely due to specific efforts to target women. Survey respondents were slightly younger than the general population (67.3 years vs 68.7 years), less likely to be male (97.1% vs 83.3%), showed similar representation of Operation Enduring Freedom/Operation Iraqi Freedom (OEF/OIF) veterans (4.4% vs 4.1%), and were less likely to have diabetes (58.0% vs 52.7% had diabetes) (Table 1).
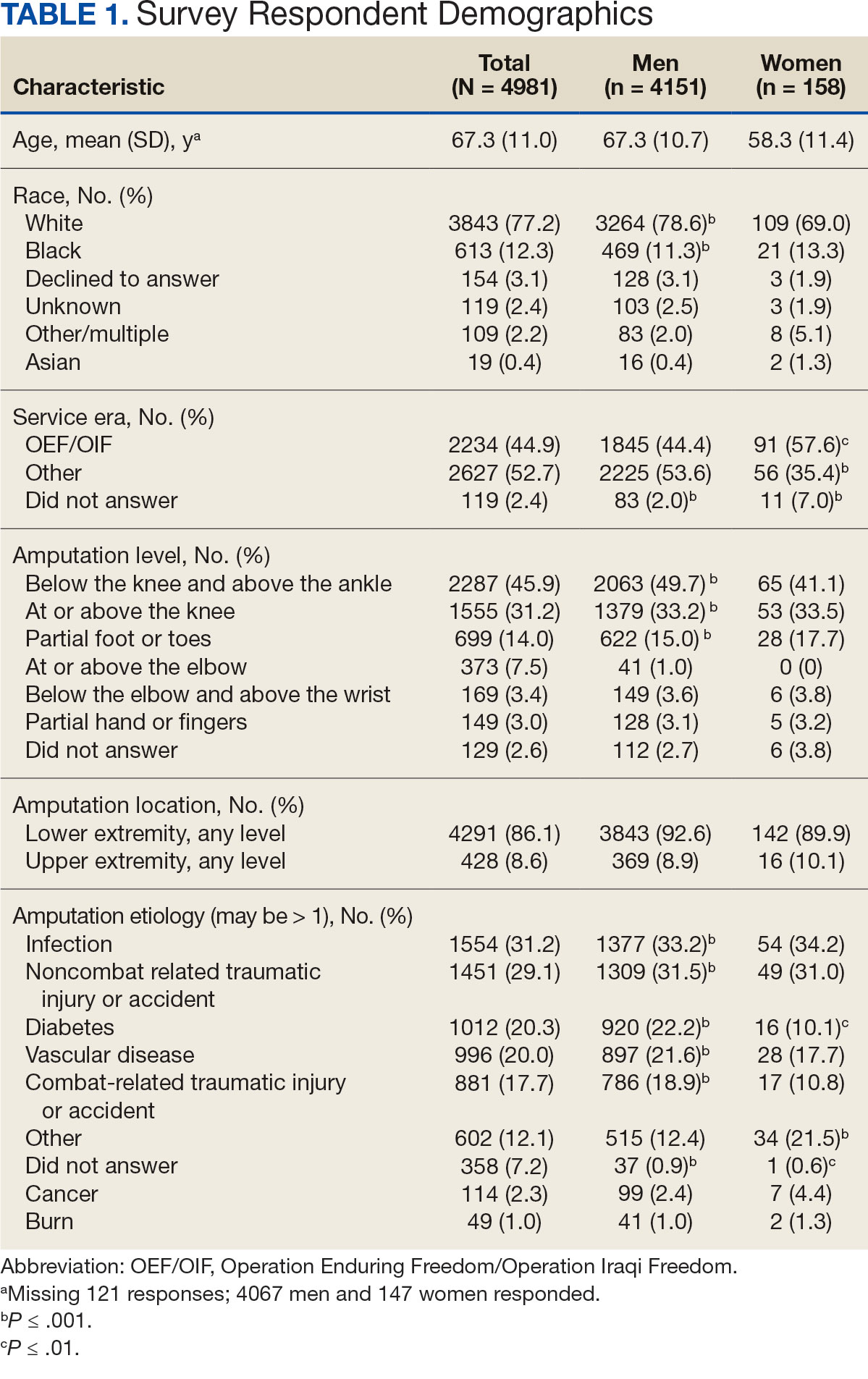
The mean age of male respondents was 67.3 years, while the mean age of female respondents was 58.3 years. The majority of respondents were male (83.3%) and White (77.2%). Female respondents were less likely to have diabetes (35.4% of women vs 53.5% of men) and less likely to report that their most recent amputation resulted from diabetes (10.1% of women vs 22.2% of men). Women respondents were more likely to report an amputation due to other causes, such as adverse results of surgery, neurologic disease, suicide attempt, blood clots, tumors, rheumatoid arthritis, and revisions of previous amputations. Most women respondents did not serve during the OEF or OIF eras. The most common amputation site for women respondents was lower limb, either below the knee and above the ankle or above the knee.
Most participants use an everyday prosthesis, but women were more likely to report using a sports-specific prosthesis (Table 2). Overall, most respondents report using a prosthesis (87.7%); however, women were more likely to report not using a prosthesis (19.4% of women vs 11.1% of men; P ≤ .01). Additionally, a lower proportion of women report using a prosthesis for < 12 hours per day (30.6% of women vs 46.4% of men; P ≤ .01) or using a prosthesis every day (54.8% of women vs 74.6% of men; P ≤ .001).
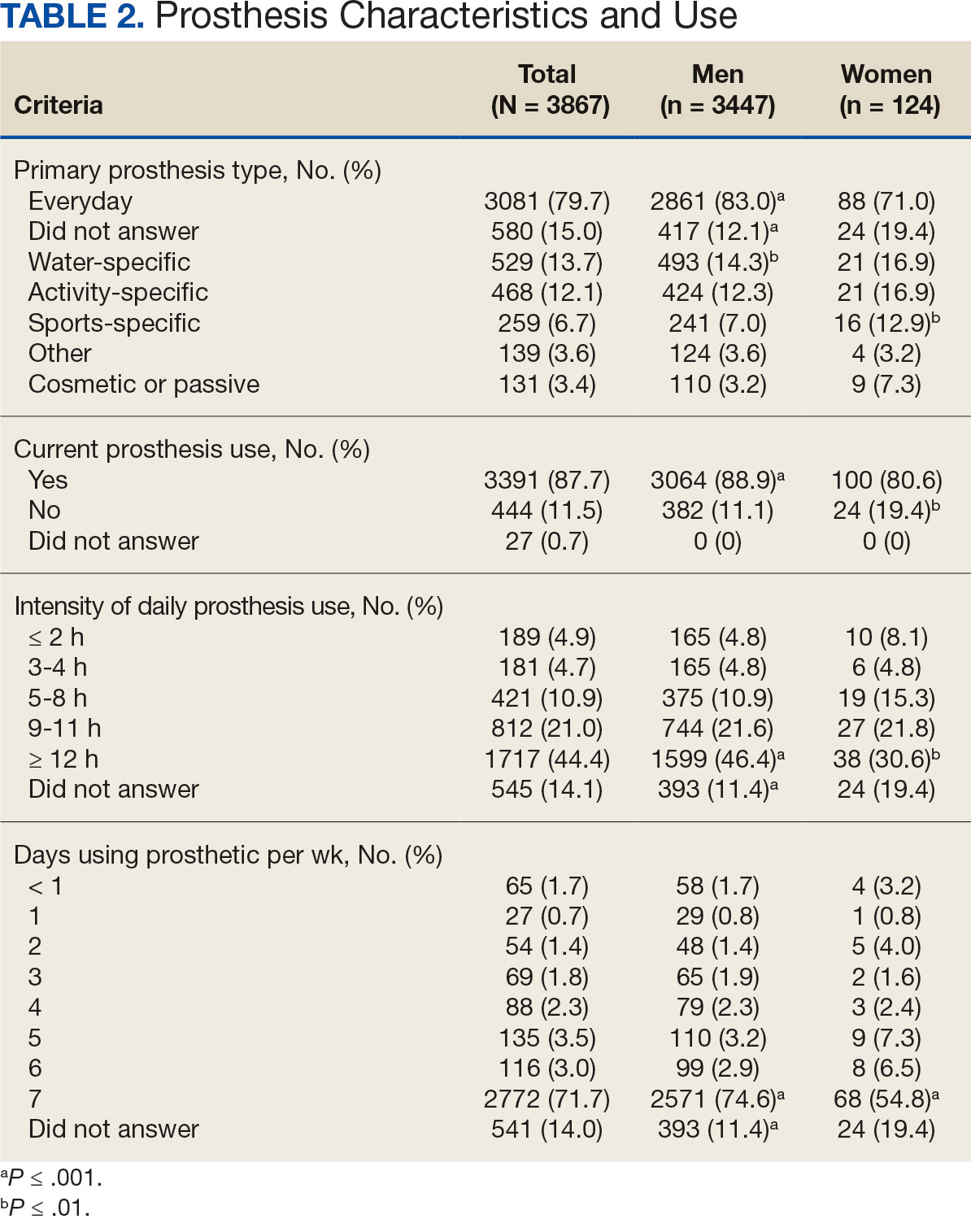
In the overall sample, the mean satisfaction score with a prosthesis was 2.7 on a 5-point scale, and women had slightly lower overall satisfaction scores (2.6 for women vs 2.7 for men; P ≤ .001) (Table 3). Women also had lower satisfaction scores related to appearance, usefulness, reliability, and comfort. Women were more likely to indicate that it was very important to be able to wear jewelry and accessories (20.2% of women vs 11.6% of men; P ≤ .01), while men were less likely to indicate that it was somewhat or very important that the prosthesis not restrict clothing or shoes (95.2% of women vs 82.9% of men; P ≤ .001). Men were more likely than women to report being comfortable or very comfortable using their prosthesis in intimate contact: 40.5% vs 29.0%, respectively (P ≤ .001).
Overall, participants reported high satisfaction with appointment times, wait times, courteous treatment, opportunities to express concerns, and staff responsiveness. Men were slightly more likely than women to be satisfied with training (P ≤ 0.001) and problem discussion (P ≤ 0.01) (Table 4). There were no statistically significant differences in satisfaction or QOL ratings between women and men. The overall sample rated both QOL and satisfaction with QOL 6.7 on a 10-point scale.
Discussion
The goal of this study was to characterize the experience of veterans with limb loss receiving care in the VHA and assess their satisfaction with prostheses and prosthetic care. We received responses from nearly 5000 veterans, 158 of whom were women. Women veteran respondents were slightly younger and less likely to have an amputation due to diabetes. We did not observe significant differences in amputation level between men and women but women were less likely to use a prosthesis, reported lower intensity of prosthesis use, and were less satisfied with certain aspects of their prostheses. Women may also be less satisfied with prosthesis training and problem discussion. However, we found no differences in QOL ratings between men and women.
Findings indicating women were more likely to report not using a prosthesis and that a lower proportion of women report using a prosthesis for > 12 hours a day or every day are consistent with previous research. 21,22 Interestingly, women were more likely to report using a sports-specific prosthesis. This is notable because prior research suggests that individuals with amputations may avoid participating in sports and exercise, and a lack of access to sports-specific prostheses may inhibit physical activity.23,24 Women in this sample were slightly less satisfied with their prostheses overall and reported lower satisfaction scores regarding appearance, usefulness, reliability, and comfort, consistent with previous findings.25
A lower percentage of women in this sample reported being comfortable or very comfortable using their prosthesis during intimate contact. Previous research on prosthesis satisfaction suggests individuals who rate prosthesis satisfaction lower also report lower body image across genders. 26 While women in this sample did not rate their prosthesis satisfaction lower than men, they did report lower intensity of prosthesis use, suggesting potential issues with their prostheses this survey did not evaluate. Women indicated the importance of prostheses not restricting jewelry, accessories, clothing, or shoes. These results have significant clinical and social implications. A recent qualitative study emphasizes that women veterans feel prostheses are primarily designed for men and may not work well with their physiological needs.9 Research focused on limbs better suited to women’s bodies could result in better fitting sockets, lightweight limbs, or less bulky designs. Additional research has also explored the difficulties in accommodating a range of footwear for patients with lower limb amputation. One study found that varying footwear heights affect the function of adjustable prosthetic feet in ways that may not be optimal.27
Ratings of satisfaction with prosthesisrelated services between men and women in this sample are consistent with a recent study showing that women veterans do not have significant differences in satisfaction with prosthesis-related services.28 However, this study focused specifically on lower limb amputations, while the respondents of this study include those with both upper and lower limb amputations. Importantly, our findings that women are less likely to be satisfied with prosthesis training and problem discussions support recent qualitative findings in which women expressed a desire to work with prosthetists who listen to them, take their concerns seriously, and seek solutions that fit their needs. We did not observe a difference in QOL ratings between men and women in the sample despite lower satisfaction among women with some elements of prosthesis-related services. Previous research suggests many factors impact QOL after amputation, most notably time since amputation.16,29
Limitations
This survey was deployed in a short timeline that did not allow for careful sample selection or implementing strategies to increase response rate. Additionally, the study was conducted among veterans receiving care in the VHA, and findings may not be generalizable to limb loss in other settings. Finally, the discrepancy in number of respondents who identified as men vs women made it difficult to compare differences between the 2 groups.
Conclusions
This is the largest sample of survey respondents of veterans with limb loss to date. While the findings suggest veterans are generally satisfied with prosthetic-related services overall, they also highlight several areas for improvement with services or prostheses. Given that most veterans with limb loss are men, there is a significant discrepancy between the number of women and men respondents. Additional studies with more comparable numbers of men and women have found similar ratings of satisfaction with prostheses and services.28 Further research specifically focused on improving the experiences of women should focus on better characterizing their experiences and identifying how they differ from those of male veterans. For example, understanding how to engage female veterans with limb loss in prosthesis training and problem discussions may improve their experience with their care teams and improve their use of prostheses. Understanding experiences and needs that are specific to women could lead to the development of processes, resources, or devices that are tailored to the unique requirements of women with limb loss.
- Ziegler-Graham K, MacKenzie EJ, Ephraim PL, Travison TG, Brookmeyer R. Estimating the prevalence of limb loss in the United States: 2005 to 2050. Arch Phys Med Rehabil. 2008;89(3):422-429. doi:10.1016/j.apmr.2007.11.005
- Dillingham TR, Pezzin LE, MacKenzie EJ. Limb amputation and limb deficiency: epidemiology and recent trends in the united states. South Med J. 2002;95(8):875-883. doi:10.1097/00007611-200208000-00018
- Dillingham TR, Pezzin LE, Shore AD. Reamputation, mortality, and health care costs among persons with dysvascular lower-limb amputations. Arch Phys Med Rehabil. 2005;86(3):480-486. doi:10.1016/j.apmr.2004.06.072
- Centers for Disease Control and Prevention. Ambulatory and inpatient procedures in the United States. Accessed September 30, 2024. https://www.cdc.gov/nchs/pressroom/98facts/ambulat.htm
- Ljung J, Iacangelo A. Identifying and acknowledging a sex gap in lower-limb prosthetics. JPO. 2024;36(1):e18-e24. doi:10.1097/JPO.0000000000000470
- Feinglass J, Brown JL, LoSasso A, et al. Rates of lower-extremity amputation and arterial reconstruction in the united states, 1979 to 1996. Am J Public Health. 1999;89(8):1222- 1227. doi:10.2105/ajph.89.8.1222
- Mayfield JA, Reiber GE, Maynard C, Czerniecki JM, Caps MT, Sangeorzan BJ. Trends in lower limb amputation in the Veterans Health Administration, 1989-1998. J Rehabil Res Dev. 2000;37(1):23-30.
- Feinglass J, Pearce WH, Martin GJ, et al. Postoperative and late survival outcomes after major amputation: findings from the department of veterans affairs national surgical quality improvement program. Surgery. 2001;130(1):21-29. doi:10.1067/msy.2001.115359
- Lehavot K, Young JP, Thomas RM, et al. Voices of women veterans with lower limb prostheses: a qualitative study. J Gen Intern Med. 2022;37(3):799-805. doi:10.1007/s11606-022-07572-8
- US Government Accountability Office. COVID-19: Opportunities to improve federal response. GAO-21-60. Published November 12, 2020. Accessed September 30, 2024. https://www.gao.gov/products/gao-21-60
- Littman AJ, Peterson AC, Korpak A, et al. Differences in prosthetic prescription between men and women veterans after transtibial or transfemoral lowerextremity amputation: a longitudinal cohort study. Arch Phys Med Rehabil. 2023;104(8)1274-1281. doi:10.1016/j.amjsurg.2023.02.011
- Cimino SR, Vijayakumar A, MacKay C, Mayo AL, Hitzig SL, Guilcher SJT. Sex and gender differences in quality of life and related domains for individuals with adult acquired lower-limb amputation: a scoping review. Disabil Rehabil. 2022 Oct 23;44(22):6899-6925. doi:10.1080/09638288.2021.1974106
- DadeMatthews OO, Roper JA, Vazquez A, Shannon DM, Sefton JM. Prosthetic device and service satisfaction, quality of life, and functional performance in lower limb prosthesis clients. Prosthet Orthot Int. 2024;48(4):422-430. doi:10.1097/PXR.0000000000000285
- Hamilton AB, Schwarz EB, Thomas HN, Goldstein KM. Moving women veterans’ health research forward: a special supplement. J Gen Intern Med. 2022;37(Suppl3):665– 667. doi:10.1007/s11606-022-07606-1
- US Congress. Public Law 116-315: An Act to Improve the Lives of Veterans, S 5108 (2) (F). 116th Congress; 2021. Accessed September 30, 2024. https://www.congress.gov/116/plaws/publ315/PLAW-116publ315.pdf
- Gallagher P, MacLachlan M. The Trinity amputation and prosthesis experience scales and quality of life in people with lower-limb amputation. Arch Phys Med Rehabil. 2004;85(5):730-736. doi:10.1016/j.apmr.2003.07.009
- Legro MW, Reiber GD, Smith DG, del Aguila M, Larsen J, Boone D. Prosthesis evaluation questionnaire for persons with lower limb amputations: assessing prosthesis-related quality of life. Arch Phys Med Rehabil. 1998;79(8):931-938. doi:10.1016/s0003-9993(98)90090-9
- Legro MW, Reiber GD, Smith DG, del Aguila M, Larsen J, Boone D. Prosthesis evaluation questionnaire for persons with lower limb amputations: assessing prosthesis-related quality of life. Arch Phys Med Rehabil. 1998;79(8):931-938. doi:10.1016/s0003-9993(98)90090-9
- Heinemann AW, Bode RK, O’Reilly C. Development and measurement properties of the orthotics and prosthetics users’ survey (OPUS): a comprehensive set of clinical outcome instruments. Prosthet Orthot Int. 2003;27(3):191-206. doi:10.1080/03093640308726682
- Resnik LJ, Borgia ML, Clark MA. A national survey of prosthesis use in veterans with major upper limb amputation: comparisons by gender. PM R. 2020;12(11):1086-1098. doi:10.1002/pmrj.12351
- Collins D. Pretesting survey instruments: an overview of cognitive methods. Qual Life Res. 2003;12(3):229-238. doi:10.1023/a:1023254226592
- Østlie K, Lesjø IM, Franklin RJ, Garfelt B, Skjeldal OH, Magnus P. Prosthesis rejection in acquired major upper-limb amputees: a population-based survey. Disabil Rehabil Assist Technol. 2012;7(4):294-303. doi:10.3109/17483107.2011.635405
- Pezzin LE, Dillingham TR, MacKenzie EJ, Ephraim P, Rossbach P. Use and satisfaction with prosthetic limb devices and related services. Arch Phys Med Rehabil. 2004;85(5):723-729. doi:10.1016/j.apmr.2003.06.002
- Deans S, Burns D, McGarry A, Murray K, Mutrie N. Motivations and barriers to prosthesis users participation in physical activity, exercise and sport: a review of the literature. Prosthet Orthot Int. 2012;36(3):260-269. doi:10.1177/0309364612437905
- McDonald CL, Kahn A, Hafner BJ, Morgan SJ. Prevalence of secondary prosthesis use in lower limb prosthesis users. Disabil Rehabil. 2023;46(5):1016-1022. doi:10.1080/09638288.2023.2182919
- Baars EC, Schrier E, Dijkstra PU, Geertzen JHB. Prosthesis satisfaction in lower limb amputees: a systematic review of associated factors and questionnaires. Medicine (Baltimore). 2018;97(39):e12296. doi:10.1097/MD.0000000000012296
- Murray CD, Fox J. Body image and prosthesis satisfaction in the lower limb amputee. Disabil Rehabil. 2002;24(17):925–931. doi:10.1080/09638280210150014
- Major MJ, Quinlan J, Hansen AH, Esposito ER. Effects of women’s footwear on the mechanical function of heel-height accommodating prosthetic feet. PLoS One. 2022;17(1). doi:10.1371/journal.pone.0262910.
- Kuo PB, Lehavot K, Thomas RM, et al. Gender differences in prosthesis-related outcomes among veterans: results of a national survey of U.S. veterans. PM R. 2024;16(3):239- 249. doi:10.1002/pmrj.13028
- Asano M, Rushton P, Miller WC, Deathe BA. Predictors of quality of life among individuals who have a lower limb amputation. Prosthet Orthot Int. 2008;32(2):231-243. doi:10.1080/03093640802024955
Limb loss is a significant and growing concern in the United States. Nearly 2 million Americans are living with limb loss, and up to 185,000 people undergo amputations annually.1-4 Of these patients, about 35% are women.5 The Veterans Health Administration (VHA) provides about 10% of US amputations.6-8 Between 2015 and 2019, the number of prosthetic devices provided to female veterans increased from 3.3 million to 4.6 million.5,9,10
Previous research identified disparities in prosthetic care between men and women, both within and outside the VHA. These disparities include slower prosthesis prescription and receipt among women, in addition to differences in self-reported mobility, satisfaction, rates of prosthesis rejection, and challenges related to prosthesis appearance and fit.5,10,11 Recent studies suggest women tend to have worse outcomes following amputation, and are underrepresented in amputation research.12,13 However, these disparities are poorly described in a large, national sample. Because women represent a growing portion of patients with limb loss in the VHA, understanding their needs is critical.14
The Johnny Isakson and David P. Roe, MD Veterans Health Care and Benefits Improvement Act of 2020 was enacted, in part, to improve the care provided to women veterans.15 The law required the VHA to conduct a survey of ≥ 50,000 veterans to assess the satisfaction of women veterans with prostheses provided by the VHA. To comply with this legislation and understand how women veterans rate their prostheses and related care in the VHA, the US Department of Veterans Affairs (VA) Center for Collaborative Evaluation (VACE) conducted a large national survey of veterans with limb loss that oversampled women veterans. This article describes the survey results, including characteristics of female veterans with limb loss receiving care from the VHA, assesses their satisfaction with prostheses and prosthetic care, and highlights where their responses differ from those of male veterans.
Methods
We conducted a cross-sectional, mixedmode survey of eligible amputees in the VHA Support Service Capital Assets Amputee Data Cube. We identified a cohort of veterans with any major amputation (above the ankle or wrist) or partial hand or foot amputation who received VHA care between October 1, 2019, and September 30, 2020. The final cohort yielded 46,646 potentially eligible veterans. Thirty-three had invalid contact information, leaving 46,613 veterans who were asked to participate, including 1356 women.
Survey
We created a survey instrument de novo that included questions from validated instruments, including the Trinity Amputation Prosthesis and Experience Scales to assess prosthetic device satisfaction, the Prosthesis Evaluation Questionnaire to assess quality of life (QOL) satisfaction, and the Orthotics Prosthetics Users Survey to assess prosthesis-related care satisfaction. 16-18 Additional questions were incorporated from a survey of veterans with upper limb amputation to assess the importance of cosmetic considerations related to the prosthesis and comfort with prosthesis use in intimate relationships.19 Questions were also included to assess amputation type, year of amputation, if a prosthesis was currently used, reasons for ceasing use of a prosthesis, reasons for never using a prosthesis, the types of prostheses used, intensity of prosthesis use, satisfaction with time required to receive a prosthetic limb, and if the prosthesis reflected the veteran’s selfidentified gender. Veterans were asked to answer questions based on their most recent amputation.
We tested the survey using cognitive interviews with 6 veterans to refine the survey and better understand how veterans interpreted the questions. Pilot testers completed the survey and participated in individual interviews with experienced interviewers (CL and RRK) to describe how they selected their responses.20 This feedback was used to refine the survey. The online survey was programmed using Qualtrics Software and manually translated into Spanish.
Given the multimodal design, surveys were distributed by email, text message, and US Postal Service (USPS). Surveys were emailed to all veterans for whom a valid email address was available. If emails were undeliverable, veterans were contacted via text message or the USPS. Surveys were distributed by text message to all veterans without an email address but with a cellphone number. We were unable to consistently identify invalid numbers among all text message recipients. Invitations with a survey URL and QR code were sent via USPS to veterans who had no valid email address or cellphone number. Targeted efforts were made to increase the response rate for women. A random sample of 200 women who had not completed the survey 2 weeks prior to the closing date (15% of women in sample) was selected to receive personal phone calls. Another random sample of 400 women was selected to receive personalized outreach emails. The survey data were confidential, and responses could not be traced to identifying information.
Data Analyses
We conducted a descriptive analysis, including percentages and means for responses to variables focused on describing amputation characteristics, prosthesis characteristics, and QOL. All data, including missing values, were used to document the percentage of respondents for each question. Removing missing data from the denominator when calculating percentages could introduce bias to the analysis because we cannot be certain data are missing at random. Missing variables were removed to avoid underinflation of mean scores.
We compared responses across 2 groups: individuals who self-identified as men and individuals who self-identified as women. For each question, we assessed whether each of these groups differed significantly from the remaining sample. For example, we examined whether the percentage of men who answered affirmatively to a question was significantly higher or lower than that of individuals not identifying as male, and whether the percentage of women who answered affirmatively was significantly higher or lower than that of individuals not identifying as female. We utilized x2 tests to determine significant differences for percentage calculations and t tests to determine significant differences in means across gender.
Since conducting multiple comparisons within a dataset may result in inflating statistical significance (type 1 errors), we used a more conservative estimate of statistical significance (α = 0.01) and high significance (α = 0.001). This study was deemed quality improvement by the VHA Rehabilitation and Prosthetic Services (12RPS) and acknowledged by the VA Research Office at Eastern Colorado Health Care System and was not subject to institutional review board review.
Results
Surveys were distributed to 46,613 veterans and were completed by 4981 respondents for a 10.7% overall response rate. Survey respondents were generally similar to the eligible population invited to participate, but the proportion of women who completed the survey was higher than the proportion of women eligible to participate (2.0% of eligible population vs 16.7% of respondents), likely due to specific efforts to target women. Survey respondents were slightly younger than the general population (67.3 years vs 68.7 years), less likely to be male (97.1% vs 83.3%), showed similar representation of Operation Enduring Freedom/Operation Iraqi Freedom (OEF/OIF) veterans (4.4% vs 4.1%), and were less likely to have diabetes (58.0% vs 52.7% had diabetes) (Table 1).

The mean age of male respondents was 67.3 years, while the mean age of female respondents was 58.3 years. The majority of respondents were male (83.3%) and White (77.2%). Female respondents were less likely to have diabetes (35.4% of women vs 53.5% of men) and less likely to report that their most recent amputation resulted from diabetes (10.1% of women vs 22.2% of men). Women respondents were more likely to report an amputation due to other causes, such as adverse results of surgery, neurologic disease, suicide attempt, blood clots, tumors, rheumatoid arthritis, and revisions of previous amputations. Most women respondents did not serve during the OEF or OIF eras. The most common amputation site for women respondents was lower limb, either below the knee and above the ankle or above the knee.
Most participants use an everyday prosthesis, but women were more likely to report using a sports-specific prosthesis (Table 2). Overall, most respondents report using a prosthesis (87.7%); however, women were more likely to report not using a prosthesis (19.4% of women vs 11.1% of men; P ≤ .01). Additionally, a lower proportion of women report using a prosthesis for < 12 hours per day (30.6% of women vs 46.4% of men; P ≤ .01) or using a prosthesis every day (54.8% of women vs 74.6% of men; P ≤ .001).

In the overall sample, the mean satisfaction score with a prosthesis was 2.7 on a 5-point scale, and women had slightly lower overall satisfaction scores (2.6 for women vs 2.7 for men; P ≤ .001) (Table 3). Women also had lower satisfaction scores related to appearance, usefulness, reliability, and comfort. Women were more likely to indicate that it was very important to be able to wear jewelry and accessories (20.2% of women vs 11.6% of men; P ≤ .01), while men were less likely to indicate that it was somewhat or very important that the prosthesis not restrict clothing or shoes (95.2% of women vs 82.9% of men; P ≤ .001). Men were more likely than women to report being comfortable or very comfortable using their prosthesis in intimate contact: 40.5% vs 29.0%, respectively (P ≤ .001).

Overall, participants reported high satisfaction with appointment times, wait times, courteous treatment, opportunities to express concerns, and staff responsiveness. Men were slightly more likely than women to be satisfied with training (P ≤ 0.001) and problem discussion (P ≤ 0.01) (Table 4). There were no statistically significant differences in satisfaction or QOL ratings between women and men. The overall sample rated both QOL and satisfaction with QOL 6.7 on a 10-point scale.

Discussion
The goal of this study was to characterize the experience of veterans with limb loss receiving care in the VHA and assess their satisfaction with prostheses and prosthetic care. We received responses from nearly 5000 veterans, 158 of whom were women. Women veteran respondents were slightly younger and less likely to have an amputation due to diabetes. We did not observe significant differences in amputation level between men and women but women were less likely to use a prosthesis, reported lower intensity of prosthesis use, and were less satisfied with certain aspects of their prostheses. Women may also be less satisfied with prosthesis training and problem discussion. However, we found no differences in QOL ratings between men and women.
Findings indicating women were more likely to report not using a prosthesis and that a lower proportion of women report using a prosthesis for > 12 hours a day or every day are consistent with previous research. 21,22 Interestingly, women were more likely to report using a sports-specific prosthesis. This is notable because prior research suggests that individuals with amputations may avoid participating in sports and exercise, and a lack of access to sports-specific prostheses may inhibit physical activity.23,24 Women in this sample were slightly less satisfied with their prostheses overall and reported lower satisfaction scores regarding appearance, usefulness, reliability, and comfort, consistent with previous findings.25
A lower percentage of women in this sample reported being comfortable or very comfortable using their prosthesis during intimate contact. Previous research on prosthesis satisfaction suggests individuals who rate prosthesis satisfaction lower also report lower body image across genders. 26 While women in this sample did not rate their prosthesis satisfaction lower than men, they did report lower intensity of prosthesis use, suggesting potential issues with their prostheses this survey did not evaluate. Women indicated the importance of prostheses not restricting jewelry, accessories, clothing, or shoes. These results have significant clinical and social implications. A recent qualitative study emphasizes that women veterans feel prostheses are primarily designed for men and may not work well with their physiological needs.9 Research focused on limbs better suited to women’s bodies could result in better fitting sockets, lightweight limbs, or less bulky designs. Additional research has also explored the difficulties in accommodating a range of footwear for patients with lower limb amputation. One study found that varying footwear heights affect the function of adjustable prosthetic feet in ways that may not be optimal.27
Ratings of satisfaction with prosthesisrelated services between men and women in this sample are consistent with a recent study showing that women veterans do not have significant differences in satisfaction with prosthesis-related services.28 However, this study focused specifically on lower limb amputations, while the respondents of this study include those with both upper and lower limb amputations. Importantly, our findings that women are less likely to be satisfied with prosthesis training and problem discussions support recent qualitative findings in which women expressed a desire to work with prosthetists who listen to them, take their concerns seriously, and seek solutions that fit their needs. We did not observe a difference in QOL ratings between men and women in the sample despite lower satisfaction among women with some elements of prosthesis-related services. Previous research suggests many factors impact QOL after amputation, most notably time since amputation.16,29
Limitations
This survey was deployed in a short timeline that did not allow for careful sample selection or implementing strategies to increase response rate. Additionally, the study was conducted among veterans receiving care in the VHA, and findings may not be generalizable to limb loss in other settings. Finally, the discrepancy in number of respondents who identified as men vs women made it difficult to compare differences between the 2 groups.
Conclusions
This is the largest sample of survey respondents of veterans with limb loss to date. While the findings suggest veterans are generally satisfied with prosthetic-related services overall, they also highlight several areas for improvement with services or prostheses. Given that most veterans with limb loss are men, there is a significant discrepancy between the number of women and men respondents. Additional studies with more comparable numbers of men and women have found similar ratings of satisfaction with prostheses and services.28 Further research specifically focused on improving the experiences of women should focus on better characterizing their experiences and identifying how they differ from those of male veterans. For example, understanding how to engage female veterans with limb loss in prosthesis training and problem discussions may improve their experience with their care teams and improve their use of prostheses. Understanding experiences and needs that are specific to women could lead to the development of processes, resources, or devices that are tailored to the unique requirements of women with limb loss.
Limb loss is a significant and growing concern in the United States. Nearly 2 million Americans are living with limb loss, and up to 185,000 people undergo amputations annually.1-4 Of these patients, about 35% are women.5 The Veterans Health Administration (VHA) provides about 10% of US amputations.6-8 Between 2015 and 2019, the number of prosthetic devices provided to female veterans increased from 3.3 million to 4.6 million.5,9,10
Previous research identified disparities in prosthetic care between men and women, both within and outside the VHA. These disparities include slower prosthesis prescription and receipt among women, in addition to differences in self-reported mobility, satisfaction, rates of prosthesis rejection, and challenges related to prosthesis appearance and fit.5,10,11 Recent studies suggest women tend to have worse outcomes following amputation, and are underrepresented in amputation research.12,13 However, these disparities are poorly described in a large, national sample. Because women represent a growing portion of patients with limb loss in the VHA, understanding their needs is critical.14
The Johnny Isakson and David P. Roe, MD Veterans Health Care and Benefits Improvement Act of 2020 was enacted, in part, to improve the care provided to women veterans.15 The law required the VHA to conduct a survey of ≥ 50,000 veterans to assess the satisfaction of women veterans with prostheses provided by the VHA. To comply with this legislation and understand how women veterans rate their prostheses and related care in the VHA, the US Department of Veterans Affairs (VA) Center for Collaborative Evaluation (VACE) conducted a large national survey of veterans with limb loss that oversampled women veterans. This article describes the survey results, including characteristics of female veterans with limb loss receiving care from the VHA, assesses their satisfaction with prostheses and prosthetic care, and highlights where their responses differ from those of male veterans.
Methods
We conducted a cross-sectional, mixedmode survey of eligible amputees in the VHA Support Service Capital Assets Amputee Data Cube. We identified a cohort of veterans with any major amputation (above the ankle or wrist) or partial hand or foot amputation who received VHA care between October 1, 2019, and September 30, 2020. The final cohort yielded 46,646 potentially eligible veterans. Thirty-three had invalid contact information, leaving 46,613 veterans who were asked to participate, including 1356 women.
Survey
We created a survey instrument de novo that included questions from validated instruments, including the Trinity Amputation Prosthesis and Experience Scales to assess prosthetic device satisfaction, the Prosthesis Evaluation Questionnaire to assess quality of life (QOL) satisfaction, and the Orthotics Prosthetics Users Survey to assess prosthesis-related care satisfaction. 16-18 Additional questions were incorporated from a survey of veterans with upper limb amputation to assess the importance of cosmetic considerations related to the prosthesis and comfort with prosthesis use in intimate relationships.19 Questions were also included to assess amputation type, year of amputation, if a prosthesis was currently used, reasons for ceasing use of a prosthesis, reasons for never using a prosthesis, the types of prostheses used, intensity of prosthesis use, satisfaction with time required to receive a prosthetic limb, and if the prosthesis reflected the veteran’s selfidentified gender. Veterans were asked to answer questions based on their most recent amputation.
We tested the survey using cognitive interviews with 6 veterans to refine the survey and better understand how veterans interpreted the questions. Pilot testers completed the survey and participated in individual interviews with experienced interviewers (CL and RRK) to describe how they selected their responses.20 This feedback was used to refine the survey. The online survey was programmed using Qualtrics Software and manually translated into Spanish.
Given the multimodal design, surveys were distributed by email, text message, and US Postal Service (USPS). Surveys were emailed to all veterans for whom a valid email address was available. If emails were undeliverable, veterans were contacted via text message or the USPS. Surveys were distributed by text message to all veterans without an email address but with a cellphone number. We were unable to consistently identify invalid numbers among all text message recipients. Invitations with a survey URL and QR code were sent via USPS to veterans who had no valid email address or cellphone number. Targeted efforts were made to increase the response rate for women. A random sample of 200 women who had not completed the survey 2 weeks prior to the closing date (15% of women in sample) was selected to receive personal phone calls. Another random sample of 400 women was selected to receive personalized outreach emails. The survey data were confidential, and responses could not be traced to identifying information.
Data Analyses
We conducted a descriptive analysis, including percentages and means for responses to variables focused on describing amputation characteristics, prosthesis characteristics, and QOL. All data, including missing values, were used to document the percentage of respondents for each question. Removing missing data from the denominator when calculating percentages could introduce bias to the analysis because we cannot be certain data are missing at random. Missing variables were removed to avoid underinflation of mean scores.
We compared responses across 2 groups: individuals who self-identified as men and individuals who self-identified as women. For each question, we assessed whether each of these groups differed significantly from the remaining sample. For example, we examined whether the percentage of men who answered affirmatively to a question was significantly higher or lower than that of individuals not identifying as male, and whether the percentage of women who answered affirmatively was significantly higher or lower than that of individuals not identifying as female. We utilized x2 tests to determine significant differences for percentage calculations and t tests to determine significant differences in means across gender.
Since conducting multiple comparisons within a dataset may result in inflating statistical significance (type 1 errors), we used a more conservative estimate of statistical significance (α = 0.01) and high significance (α = 0.001). This study was deemed quality improvement by the VHA Rehabilitation and Prosthetic Services (12RPS) and acknowledged by the VA Research Office at Eastern Colorado Health Care System and was not subject to institutional review board review.
Results
Surveys were distributed to 46,613 veterans and were completed by 4981 respondents for a 10.7% overall response rate. Survey respondents were generally similar to the eligible population invited to participate, but the proportion of women who completed the survey was higher than the proportion of women eligible to participate (2.0% of eligible population vs 16.7% of respondents), likely due to specific efforts to target women. Survey respondents were slightly younger than the general population (67.3 years vs 68.7 years), less likely to be male (97.1% vs 83.3%), showed similar representation of Operation Enduring Freedom/Operation Iraqi Freedom (OEF/OIF) veterans (4.4% vs 4.1%), and were less likely to have diabetes (58.0% vs 52.7% had diabetes) (Table 1).

The mean age of male respondents was 67.3 years, while the mean age of female respondents was 58.3 years. The majority of respondents were male (83.3%) and White (77.2%). Female respondents were less likely to have diabetes (35.4% of women vs 53.5% of men) and less likely to report that their most recent amputation resulted from diabetes (10.1% of women vs 22.2% of men). Women respondents were more likely to report an amputation due to other causes, such as adverse results of surgery, neurologic disease, suicide attempt, blood clots, tumors, rheumatoid arthritis, and revisions of previous amputations. Most women respondents did not serve during the OEF or OIF eras. The most common amputation site for women respondents was lower limb, either below the knee and above the ankle or above the knee.
Most participants use an everyday prosthesis, but women were more likely to report using a sports-specific prosthesis (Table 2). Overall, most respondents report using a prosthesis (87.7%); however, women were more likely to report not using a prosthesis (19.4% of women vs 11.1% of men; P ≤ .01). Additionally, a lower proportion of women report using a prosthesis for < 12 hours per day (30.6% of women vs 46.4% of men; P ≤ .01) or using a prosthesis every day (54.8% of women vs 74.6% of men; P ≤ .001).

In the overall sample, the mean satisfaction score with a prosthesis was 2.7 on a 5-point scale, and women had slightly lower overall satisfaction scores (2.6 for women vs 2.7 for men; P ≤ .001) (Table 3). Women also had lower satisfaction scores related to appearance, usefulness, reliability, and comfort. Women were more likely to indicate that it was very important to be able to wear jewelry and accessories (20.2% of women vs 11.6% of men; P ≤ .01), while men were less likely to indicate that it was somewhat or very important that the prosthesis not restrict clothing or shoes (95.2% of women vs 82.9% of men; P ≤ .001). Men were more likely than women to report being comfortable or very comfortable using their prosthesis in intimate contact: 40.5% vs 29.0%, respectively (P ≤ .001).

Overall, participants reported high satisfaction with appointment times, wait times, courteous treatment, opportunities to express concerns, and staff responsiveness. Men were slightly more likely than women to be satisfied with training (P ≤ 0.001) and problem discussion (P ≤ 0.01) (Table 4). There were no statistically significant differences in satisfaction or QOL ratings between women and men. The overall sample rated both QOL and satisfaction with QOL 6.7 on a 10-point scale.

Discussion
The goal of this study was to characterize the experience of veterans with limb loss receiving care in the VHA and assess their satisfaction with prostheses and prosthetic care. We received responses from nearly 5000 veterans, 158 of whom were women. Women veteran respondents were slightly younger and less likely to have an amputation due to diabetes. We did not observe significant differences in amputation level between men and women but women were less likely to use a prosthesis, reported lower intensity of prosthesis use, and were less satisfied with certain aspects of their prostheses. Women may also be less satisfied with prosthesis training and problem discussion. However, we found no differences in QOL ratings between men and women.
Findings indicating women were more likely to report not using a prosthesis and that a lower proportion of women report using a prosthesis for > 12 hours a day or every day are consistent with previous research. 21,22 Interestingly, women were more likely to report using a sports-specific prosthesis. This is notable because prior research suggests that individuals with amputations may avoid participating in sports and exercise, and a lack of access to sports-specific prostheses may inhibit physical activity.23,24 Women in this sample were slightly less satisfied with their prostheses overall and reported lower satisfaction scores regarding appearance, usefulness, reliability, and comfort, consistent with previous findings.25
A lower percentage of women in this sample reported being comfortable or very comfortable using their prosthesis during intimate contact. Previous research on prosthesis satisfaction suggests individuals who rate prosthesis satisfaction lower also report lower body image across genders. 26 While women in this sample did not rate their prosthesis satisfaction lower than men, they did report lower intensity of prosthesis use, suggesting potential issues with their prostheses this survey did not evaluate. Women indicated the importance of prostheses not restricting jewelry, accessories, clothing, or shoes. These results have significant clinical and social implications. A recent qualitative study emphasizes that women veterans feel prostheses are primarily designed for men and may not work well with their physiological needs.9 Research focused on limbs better suited to women’s bodies could result in better fitting sockets, lightweight limbs, or less bulky designs. Additional research has also explored the difficulties in accommodating a range of footwear for patients with lower limb amputation. One study found that varying footwear heights affect the function of adjustable prosthetic feet in ways that may not be optimal.27
Ratings of satisfaction with prosthesisrelated services between men and women in this sample are consistent with a recent study showing that women veterans do not have significant differences in satisfaction with prosthesis-related services.28 However, this study focused specifically on lower limb amputations, while the respondents of this study include those with both upper and lower limb amputations. Importantly, our findings that women are less likely to be satisfied with prosthesis training and problem discussions support recent qualitative findings in which women expressed a desire to work with prosthetists who listen to them, take their concerns seriously, and seek solutions that fit their needs. We did not observe a difference in QOL ratings between men and women in the sample despite lower satisfaction among women with some elements of prosthesis-related services. Previous research suggests many factors impact QOL after amputation, most notably time since amputation.16,29
Limitations
This survey was deployed in a short timeline that did not allow for careful sample selection or implementing strategies to increase response rate. Additionally, the study was conducted among veterans receiving care in the VHA, and findings may not be generalizable to limb loss in other settings. Finally, the discrepancy in number of respondents who identified as men vs women made it difficult to compare differences between the 2 groups.
Conclusions
This is the largest sample of survey respondents of veterans with limb loss to date. While the findings suggest veterans are generally satisfied with prosthetic-related services overall, they also highlight several areas for improvement with services or prostheses. Given that most veterans with limb loss are men, there is a significant discrepancy between the number of women and men respondents. Additional studies with more comparable numbers of men and women have found similar ratings of satisfaction with prostheses and services.28 Further research specifically focused on improving the experiences of women should focus on better characterizing their experiences and identifying how they differ from those of male veterans. For example, understanding how to engage female veterans with limb loss in prosthesis training and problem discussions may improve their experience with their care teams and improve their use of prostheses. Understanding experiences and needs that are specific to women could lead to the development of processes, resources, or devices that are tailored to the unique requirements of women with limb loss.
- Ziegler-Graham K, MacKenzie EJ, Ephraim PL, Travison TG, Brookmeyer R. Estimating the prevalence of limb loss in the United States: 2005 to 2050. Arch Phys Med Rehabil. 2008;89(3):422-429. doi:10.1016/j.apmr.2007.11.005
- Dillingham TR, Pezzin LE, MacKenzie EJ. Limb amputation and limb deficiency: epidemiology and recent trends in the united states. South Med J. 2002;95(8):875-883. doi:10.1097/00007611-200208000-00018
- Dillingham TR, Pezzin LE, Shore AD. Reamputation, mortality, and health care costs among persons with dysvascular lower-limb amputations. Arch Phys Med Rehabil. 2005;86(3):480-486. doi:10.1016/j.apmr.2004.06.072
- Centers for Disease Control and Prevention. Ambulatory and inpatient procedures in the United States. Accessed September 30, 2024. https://www.cdc.gov/nchs/pressroom/98facts/ambulat.htm
- Ljung J, Iacangelo A. Identifying and acknowledging a sex gap in lower-limb prosthetics. JPO. 2024;36(1):e18-e24. doi:10.1097/JPO.0000000000000470
- Feinglass J, Brown JL, LoSasso A, et al. Rates of lower-extremity amputation and arterial reconstruction in the united states, 1979 to 1996. Am J Public Health. 1999;89(8):1222- 1227. doi:10.2105/ajph.89.8.1222
- Mayfield JA, Reiber GE, Maynard C, Czerniecki JM, Caps MT, Sangeorzan BJ. Trends in lower limb amputation in the Veterans Health Administration, 1989-1998. J Rehabil Res Dev. 2000;37(1):23-30.
- Feinglass J, Pearce WH, Martin GJ, et al. Postoperative and late survival outcomes after major amputation: findings from the department of veterans affairs national surgical quality improvement program. Surgery. 2001;130(1):21-29. doi:10.1067/msy.2001.115359
- Lehavot K, Young JP, Thomas RM, et al. Voices of women veterans with lower limb prostheses: a qualitative study. J Gen Intern Med. 2022;37(3):799-805. doi:10.1007/s11606-022-07572-8
- US Government Accountability Office. COVID-19: Opportunities to improve federal response. GAO-21-60. Published November 12, 2020. Accessed September 30, 2024. https://www.gao.gov/products/gao-21-60
- Littman AJ, Peterson AC, Korpak A, et al. Differences in prosthetic prescription between men and women veterans after transtibial or transfemoral lowerextremity amputation: a longitudinal cohort study. Arch Phys Med Rehabil. 2023;104(8)1274-1281. doi:10.1016/j.amjsurg.2023.02.011
- Cimino SR, Vijayakumar A, MacKay C, Mayo AL, Hitzig SL, Guilcher SJT. Sex and gender differences in quality of life and related domains for individuals with adult acquired lower-limb amputation: a scoping review. Disabil Rehabil. 2022 Oct 23;44(22):6899-6925. doi:10.1080/09638288.2021.1974106
- DadeMatthews OO, Roper JA, Vazquez A, Shannon DM, Sefton JM. Prosthetic device and service satisfaction, quality of life, and functional performance in lower limb prosthesis clients. Prosthet Orthot Int. 2024;48(4):422-430. doi:10.1097/PXR.0000000000000285
- Hamilton AB, Schwarz EB, Thomas HN, Goldstein KM. Moving women veterans’ health research forward: a special supplement. J Gen Intern Med. 2022;37(Suppl3):665– 667. doi:10.1007/s11606-022-07606-1
- US Congress. Public Law 116-315: An Act to Improve the Lives of Veterans, S 5108 (2) (F). 116th Congress; 2021. Accessed September 30, 2024. https://www.congress.gov/116/plaws/publ315/PLAW-116publ315.pdf
- Gallagher P, MacLachlan M. The Trinity amputation and prosthesis experience scales and quality of life in people with lower-limb amputation. Arch Phys Med Rehabil. 2004;85(5):730-736. doi:10.1016/j.apmr.2003.07.009
- Legro MW, Reiber GD, Smith DG, del Aguila M, Larsen J, Boone D. Prosthesis evaluation questionnaire for persons with lower limb amputations: assessing prosthesis-related quality of life. Arch Phys Med Rehabil. 1998;79(8):931-938. doi:10.1016/s0003-9993(98)90090-9
- Legro MW, Reiber GD, Smith DG, del Aguila M, Larsen J, Boone D. Prosthesis evaluation questionnaire for persons with lower limb amputations: assessing prosthesis-related quality of life. Arch Phys Med Rehabil. 1998;79(8):931-938. doi:10.1016/s0003-9993(98)90090-9
- Heinemann AW, Bode RK, O’Reilly C. Development and measurement properties of the orthotics and prosthetics users’ survey (OPUS): a comprehensive set of clinical outcome instruments. Prosthet Orthot Int. 2003;27(3):191-206. doi:10.1080/03093640308726682
- Resnik LJ, Borgia ML, Clark MA. A national survey of prosthesis use in veterans with major upper limb amputation: comparisons by gender. PM R. 2020;12(11):1086-1098. doi:10.1002/pmrj.12351
- Collins D. Pretesting survey instruments: an overview of cognitive methods. Qual Life Res. 2003;12(3):229-238. doi:10.1023/a:1023254226592
- Østlie K, Lesjø IM, Franklin RJ, Garfelt B, Skjeldal OH, Magnus P. Prosthesis rejection in acquired major upper-limb amputees: a population-based survey. Disabil Rehabil Assist Technol. 2012;7(4):294-303. doi:10.3109/17483107.2011.635405
- Pezzin LE, Dillingham TR, MacKenzie EJ, Ephraim P, Rossbach P. Use and satisfaction with prosthetic limb devices and related services. Arch Phys Med Rehabil. 2004;85(5):723-729. doi:10.1016/j.apmr.2003.06.002
- Deans S, Burns D, McGarry A, Murray K, Mutrie N. Motivations and barriers to prosthesis users participation in physical activity, exercise and sport: a review of the literature. Prosthet Orthot Int. 2012;36(3):260-269. doi:10.1177/0309364612437905
- McDonald CL, Kahn A, Hafner BJ, Morgan SJ. Prevalence of secondary prosthesis use in lower limb prosthesis users. Disabil Rehabil. 2023;46(5):1016-1022. doi:10.1080/09638288.2023.2182919
- Baars EC, Schrier E, Dijkstra PU, Geertzen JHB. Prosthesis satisfaction in lower limb amputees: a systematic review of associated factors and questionnaires. Medicine (Baltimore). 2018;97(39):e12296. doi:10.1097/MD.0000000000012296
- Murray CD, Fox J. Body image and prosthesis satisfaction in the lower limb amputee. Disabil Rehabil. 2002;24(17):925–931. doi:10.1080/09638280210150014
- Major MJ, Quinlan J, Hansen AH, Esposito ER. Effects of women’s footwear on the mechanical function of heel-height accommodating prosthetic feet. PLoS One. 2022;17(1). doi:10.1371/journal.pone.0262910.
- Kuo PB, Lehavot K, Thomas RM, et al. Gender differences in prosthesis-related outcomes among veterans: results of a national survey of U.S. veterans. PM R. 2024;16(3):239- 249. doi:10.1002/pmrj.13028
- Asano M, Rushton P, Miller WC, Deathe BA. Predictors of quality of life among individuals who have a lower limb amputation. Prosthet Orthot Int. 2008;32(2):231-243. doi:10.1080/03093640802024955
- Ziegler-Graham K, MacKenzie EJ, Ephraim PL, Travison TG, Brookmeyer R. Estimating the prevalence of limb loss in the United States: 2005 to 2050. Arch Phys Med Rehabil. 2008;89(3):422-429. doi:10.1016/j.apmr.2007.11.005
- Dillingham TR, Pezzin LE, MacKenzie EJ. Limb amputation and limb deficiency: epidemiology and recent trends in the united states. South Med J. 2002;95(8):875-883. doi:10.1097/00007611-200208000-00018
- Dillingham TR, Pezzin LE, Shore AD. Reamputation, mortality, and health care costs among persons with dysvascular lower-limb amputations. Arch Phys Med Rehabil. 2005;86(3):480-486. doi:10.1016/j.apmr.2004.06.072
- Centers for Disease Control and Prevention. Ambulatory and inpatient procedures in the United States. Accessed September 30, 2024. https://www.cdc.gov/nchs/pressroom/98facts/ambulat.htm
- Ljung J, Iacangelo A. Identifying and acknowledging a sex gap in lower-limb prosthetics. JPO. 2024;36(1):e18-e24. doi:10.1097/JPO.0000000000000470
- Feinglass J, Brown JL, LoSasso A, et al. Rates of lower-extremity amputation and arterial reconstruction in the united states, 1979 to 1996. Am J Public Health. 1999;89(8):1222- 1227. doi:10.2105/ajph.89.8.1222
- Mayfield JA, Reiber GE, Maynard C, Czerniecki JM, Caps MT, Sangeorzan BJ. Trends in lower limb amputation in the Veterans Health Administration, 1989-1998. J Rehabil Res Dev. 2000;37(1):23-30.
- Feinglass J, Pearce WH, Martin GJ, et al. Postoperative and late survival outcomes after major amputation: findings from the department of veterans affairs national surgical quality improvement program. Surgery. 2001;130(1):21-29. doi:10.1067/msy.2001.115359
- Lehavot K, Young JP, Thomas RM, et al. Voices of women veterans with lower limb prostheses: a qualitative study. J Gen Intern Med. 2022;37(3):799-805. doi:10.1007/s11606-022-07572-8
- US Government Accountability Office. COVID-19: Opportunities to improve federal response. GAO-21-60. Published November 12, 2020. Accessed September 30, 2024. https://www.gao.gov/products/gao-21-60
- Littman AJ, Peterson AC, Korpak A, et al. Differences in prosthetic prescription between men and women veterans after transtibial or transfemoral lowerextremity amputation: a longitudinal cohort study. Arch Phys Med Rehabil. 2023;104(8)1274-1281. doi:10.1016/j.amjsurg.2023.02.011
- Cimino SR, Vijayakumar A, MacKay C, Mayo AL, Hitzig SL, Guilcher SJT. Sex and gender differences in quality of life and related domains for individuals with adult acquired lower-limb amputation: a scoping review. Disabil Rehabil. 2022 Oct 23;44(22):6899-6925. doi:10.1080/09638288.2021.1974106
- DadeMatthews OO, Roper JA, Vazquez A, Shannon DM, Sefton JM. Prosthetic device and service satisfaction, quality of life, and functional performance in lower limb prosthesis clients. Prosthet Orthot Int. 2024;48(4):422-430. doi:10.1097/PXR.0000000000000285
- Hamilton AB, Schwarz EB, Thomas HN, Goldstein KM. Moving women veterans’ health research forward: a special supplement. J Gen Intern Med. 2022;37(Suppl3):665– 667. doi:10.1007/s11606-022-07606-1
- US Congress. Public Law 116-315: An Act to Improve the Lives of Veterans, S 5108 (2) (F). 116th Congress; 2021. Accessed September 30, 2024. https://www.congress.gov/116/plaws/publ315/PLAW-116publ315.pdf
- Gallagher P, MacLachlan M. The Trinity amputation and prosthesis experience scales and quality of life in people with lower-limb amputation. Arch Phys Med Rehabil. 2004;85(5):730-736. doi:10.1016/j.apmr.2003.07.009
- Legro MW, Reiber GD, Smith DG, del Aguila M, Larsen J, Boone D. Prosthesis evaluation questionnaire for persons with lower limb amputations: assessing prosthesis-related quality of life. Arch Phys Med Rehabil. 1998;79(8):931-938. doi:10.1016/s0003-9993(98)90090-9
- Legro MW, Reiber GD, Smith DG, del Aguila M, Larsen J, Boone D. Prosthesis evaluation questionnaire for persons with lower limb amputations: assessing prosthesis-related quality of life. Arch Phys Med Rehabil. 1998;79(8):931-938. doi:10.1016/s0003-9993(98)90090-9
- Heinemann AW, Bode RK, O’Reilly C. Development and measurement properties of the orthotics and prosthetics users’ survey (OPUS): a comprehensive set of clinical outcome instruments. Prosthet Orthot Int. 2003;27(3):191-206. doi:10.1080/03093640308726682
- Resnik LJ, Borgia ML, Clark MA. A national survey of prosthesis use in veterans with major upper limb amputation: comparisons by gender. PM R. 2020;12(11):1086-1098. doi:10.1002/pmrj.12351
- Collins D. Pretesting survey instruments: an overview of cognitive methods. Qual Life Res. 2003;12(3):229-238. doi:10.1023/a:1023254226592
- Østlie K, Lesjø IM, Franklin RJ, Garfelt B, Skjeldal OH, Magnus P. Prosthesis rejection in acquired major upper-limb amputees: a population-based survey. Disabil Rehabil Assist Technol. 2012;7(4):294-303. doi:10.3109/17483107.2011.635405
- Pezzin LE, Dillingham TR, MacKenzie EJ, Ephraim P, Rossbach P. Use and satisfaction with prosthetic limb devices and related services. Arch Phys Med Rehabil. 2004;85(5):723-729. doi:10.1016/j.apmr.2003.06.002
- Deans S, Burns D, McGarry A, Murray K, Mutrie N. Motivations and barriers to prosthesis users participation in physical activity, exercise and sport: a review of the literature. Prosthet Orthot Int. 2012;36(3):260-269. doi:10.1177/0309364612437905
- McDonald CL, Kahn A, Hafner BJ, Morgan SJ. Prevalence of secondary prosthesis use in lower limb prosthesis users. Disabil Rehabil. 2023;46(5):1016-1022. doi:10.1080/09638288.2023.2182919
- Baars EC, Schrier E, Dijkstra PU, Geertzen JHB. Prosthesis satisfaction in lower limb amputees: a systematic review of associated factors and questionnaires. Medicine (Baltimore). 2018;97(39):e12296. doi:10.1097/MD.0000000000012296
- Murray CD, Fox J. Body image and prosthesis satisfaction in the lower limb amputee. Disabil Rehabil. 2002;24(17):925–931. doi:10.1080/09638280210150014
- Major MJ, Quinlan J, Hansen AH, Esposito ER. Effects of women’s footwear on the mechanical function of heel-height accommodating prosthetic feet. PLoS One. 2022;17(1). doi:10.1371/journal.pone.0262910.
- Kuo PB, Lehavot K, Thomas RM, et al. Gender differences in prosthesis-related outcomes among veterans: results of a national survey of U.S. veterans. PM R. 2024;16(3):239- 249. doi:10.1002/pmrj.13028
- Asano M, Rushton P, Miller WC, Deathe BA. Predictors of quality of life among individuals who have a lower limb amputation. Prosthet Orthot Int. 2008;32(2):231-243. doi:10.1080/03093640802024955
Satisfaction With Department of Veterans Affairs Prosthetics and Support Services as Reported by Women and Men Veterans
Satisfaction With Department of Veterans Affairs Prosthetics and Support Services as Reported by Women and Men Veterans
Effect of Alirocumab Monotherapy vs Ezetimibe Plus Statin Therapy on LDL-C Lowering in Veterans With History of ASCVD
Atherosclerotic cardiovascular disease (ASCVD) is a significant cause of morbidity and mortality in the United States. ASCVD involves the buildup of cholesterol plaque in arteries and includes acute coronary syndrome, peripheral arterial disease, and events such as myocardial infarction and stroke.1 Cardiovascular disease (CVD) risk factors include high cholesterol levels, elevated blood pressure, insulin resistance, elevated blood glucose levels, smoking, poor dietary habits, and a sedentary lifestyle.2
According to the Centers for Disease Control and Prevention, about 86 million adults aged ≥ 20 years have total cholesterol levels > 200 mg/dL. More than half (54.5%) who could benefit are currently taking cholesterol-lowering medications.3 Controlling high cholesterol in American adults, especially veterans, is essential for reducing CVD morbidity and mortality.
The 2018 American College of Cardiology/American Heart Association (ACC/AHA) guideline recommends a low-density lipoprotein cholesterol (LDL-C) target goal of < 70 mg/dL for patients at high risk for ASCVD. Very high-risk ASCVD includes a history of multiple major ASCVD events or 1 major ASCVD event and multiple high-risk conditions (eg, age ≥ 65 years, smoking, or diabetes).4 Major ASCVD events include recent acute coronary syndrome (within the past 12 months), a history of myocardial infarction or ischemic stroke, and symptomatic peripheral artery disease.
The ACC/AHA guideline suggests that if the LDL-C level remains ≥ 70 mg/dL, adding ezetimibe (a dietary cholesterol absorption inhibitor) to maximally tolerated statin therapy is reasonable. If LDL-C levels remain ≥ 70 mg/dL, adding a proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitor, such as alirocumab, is reasonable.4 The US Departments of Veterans Affairs/US Department of Defense guidelines recommend using maximally tolerated statins and ezetimibe before PCSK9 inhibitors due to established long-term safety and reduction in CVD events.
Generic statins and ezetimibe are administered orally and widely available. In contrast, PCSK9 inhibitors have unknown long-term safety profiles, require subcutaneous injection once or twice monthly, and are significantly more expensive. They also require patient education on proper use while providing comparable or lesser relative risk reductions.2
These 3 classes of medication vary in their mechanisms of action to reduce LDL.5,6 Ezetimibe and several statin medications are included on the Veterans Affairs Sioux Falls Health Care System (VASFHCS) formulary and do not require review prior to prescribing. Alirocumab is available at VASFHCS but is restricted to patients with a history of ASCVD or a diagnosis of familial hypercholesterolemia, and who are receiving maximally tolerated statin and ezetimibe therapy but require further LDL-C lowering to reduce their ASCVD risk.
Studies have found ezetimibe monotherapy reduces LDL-C in patients with dyslipidemia by 18% after 12 weeks.7 One found that the percentage reduction in LDL-C was significantly greater (P < .001) with all doses of ezetimibe plus simvastatin (46% to 59%) compared with either atorvastatin 10 mg (37%) or simvastatin 20 mg (38%) monotherapy after 6 weeks.8
Although alirocumab can be added to other lipid therapies, most VASFHCS patients are prescribed alirocumab monotherapy. In the ODYSSEY CHOICE II study, patients were randomly assigned to receive either a placebo or alirocumab 150 mg every 4 weeks or alirocumab 75 mg every 2 weeks. The primary efficacy endpoint was LDL-C percentage change from baseline to week 24. In the alirocumab 150 mg every 4 weeks and 75 mg every 2 weeks groups, the least-squares mean LDL-C changes from baseline to week 24 were 51.7% and 53.5%, respectively, compared to a 4.7% increase in the placebo group (both groups P < .001 vs placebo). The authors also reported that alirocumab 150 mg every 4 weeks as monotherapy demonstrated a 47.4% reduction in LDL-C levels from baseline in a phase 1 study.9Although alirocumab monotherapy and ezetimibe plus statin therapy have been shown to effectively decrease LDL-C independently, a direct comparison of alirocumab monotherapy vs ezetimibe plus statin therapy has not been assessed, to our knowledge. Understanding the differences in effectiveness and safety between these 2 regimens will be valuable for clinicians when selecting a medication regimen for veterans with a history of ASCVD.
METHODS
This retrospective, single-center chart review used VASFHCS Computerized Patient Record System (CPRS) and Joint Longitudinal Viewer (JLV) records to compare patients with a history of ASCVD events who were treated with alirocumab monotherapy or ezetimibe plus statin. The 2 groups were randomized in a 1:3 ratio. The primary endpoint was achieving LDL-C < 70 mg/dL after 4 to 12 weeks, 13 to 24 weeks, and 25 to 52 weeks. Secondary endpoints included the mean percentage change from baseline in total cholesterol (TC), high-density lipoprotein cholesterol (HDL-C), LDL-C, and triglycerides (TG) over 52 weeks. The incidence of ASCVD events during this period was also assessed. If LDL-C < 70 mg/dL was achieved > 1 time during each time frame, only 1 incident was counted for analysis. Safety was assessed based on the incidence of any adverse event (AE) that led to treatment discontinuation.
Patients were identified by screening the prescription fill history between October 1, 2019, and December 31, 2022. The 52-week data collection period was counted from the first available fill date. Additionally, the prior authorization drug request file from January 1, 2017, to December 31, 2022, was used to obtain a list of patients prescribed alirocumab. Patients were included if they were veterans aged ≥ 18 years and had a history of an ASCVD event, had a alirocumab monotherapy or ezetimibe plus statin prescription between October 1, 2019, and December 31, 2022, or had an approved prior authorization drug request for alirocumab between January 1, 2017, and December 31, 2022. Patients missing a baseline or follow-up lipid panel and those with concurrent use of alirocumab and ezetimibe and/or statin were excluded.
Baseline characteristics collected for patients included age, sex, race, weight, body mass index, lipid parameters (LDL-C, TC, HDL-C, and TG), dosing of each type of statin before adding ezetimibe, and use of any other antihyperlipidemic medication. We also collected histories of hypertension, hyperlipidemia, diabetes, chronic kidney disease, congestive heart failure, and smoking or tobacco use status. The baseline lipid panel was the most recent lipid panel documented before starting alirocumab or ezetimibe plus statin therapy. Follow-up lipid panel values were gathered at 4 to 12 weeks, 13 to 24 weeks, and 25 to 52 weeks following initiation of either therapy.
High-, moderate-, and low-intensity dosing of statin therapy and alirocumab dosing (75 mg every 2 weeks, 150 mg every 2 weeks, or 300 mg every 4 weeks) were recorded at the specified intervals. However, no patients in this study received the latter dosing regimen. ASCVD events and safety endpoints were recorded based on a review of clinical notes over the 52 weeks following the first available start date.
Statistical Analysis
The primary endpoint of achieving the LDL-C < 70 mg/dL goal from baseline to 4 to 12 weeks, 13 to 24 weeks, and 25 to 52 weeks after initiation was compared between alirocumab monotherapy and ezetimibe plus statin therapy using the χ² test. Mean percentage change from baseline in TC, HDL-C, LDL-C, and TG were compared using the independent t test. P < .05 was considered statistically significant. Incidence of ASCVD events and the safety endpoint (incidence of AEs leading to treatment discontinuation) were also compared using the χ² test. Continuous baseline characteristics were reported mean (SD) and nominal baseline characteristics were reported as a percentage.
RESULTS
There were 80 participants in this study: 20 in the alirocumab monotherapy group and 60 in the ezetimibe plus statin therapy group. More than 100 patients did not meet the prespecified inclusion criteria and were excluded. Mean (SD) age was 75 (8) years in the alirocumab group and 74 (8) years in the ezetimibe plus statin group. There was no significant differences in mean (SD) weight or mean (SD) body mass index. All study participants identified as White and male except for 2 patients in the ezetimibe plus statin therapy group whose race was not documented. Differences in lipid parameters were observed between groups, with mean baseline LDL-C, HDL-C, and TC higher in the alirocumab monotherapy group than in the ezetimibe plus statin therapy group, with significant differences in LDL-C and TC (Table 1).
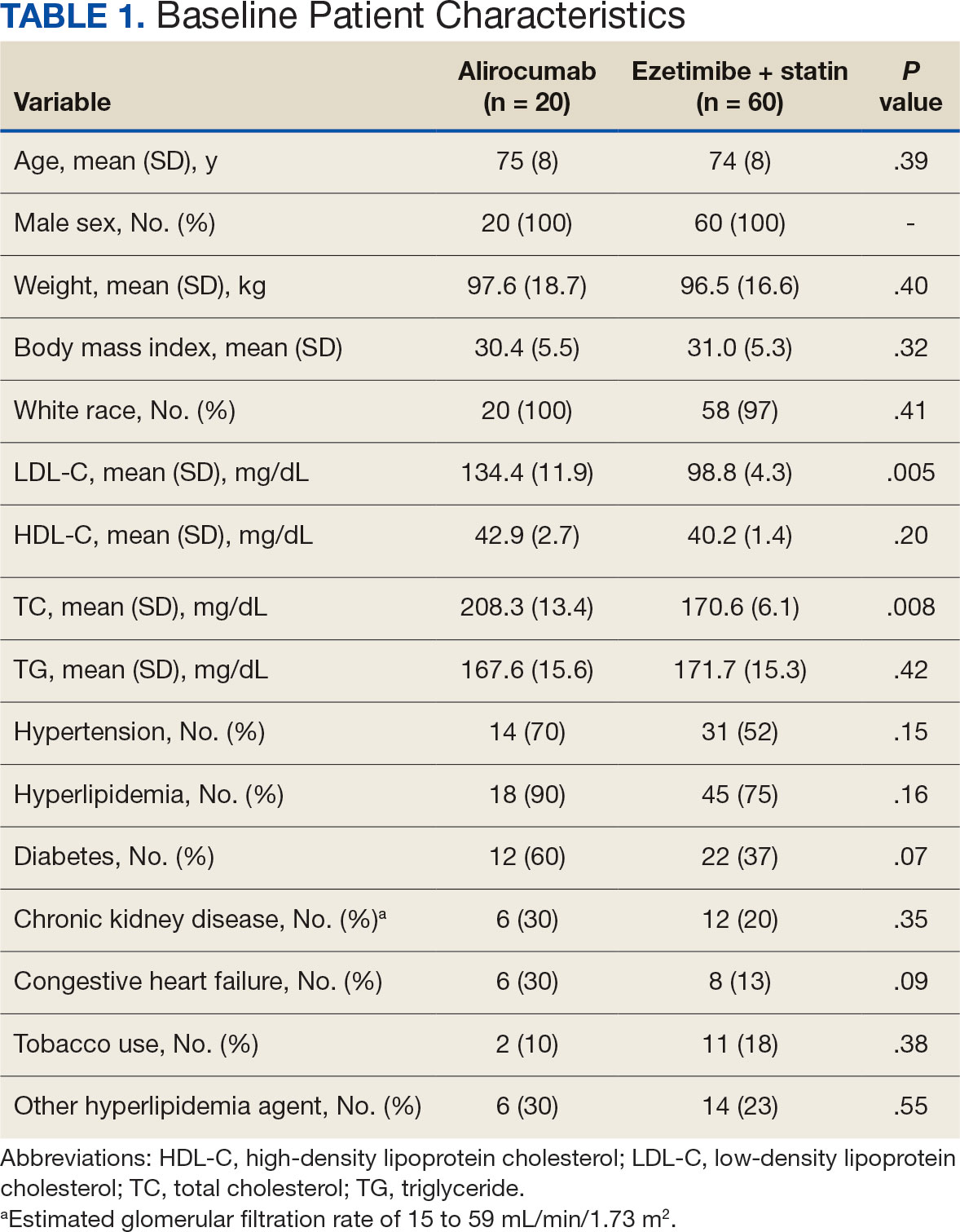
Fourteen patients (70%) in the alirocumab monotherapy group had hypertension, compared with 31 (52%) in the ezetimibe plus statin therapy group. In both groups, most patients had previously been diagnosed with hyperlipidemia. More patients (60%) in the alirocumab group had diabetes than in the ezetimibe plus statin therapy group (37%). The alirocumab monotherapy group also had a higher percentage of patients with diagnoses of congestive heart failure and used other antihyperlipidemic medications than in the ezetimibe plus statin therapy group. Five patients (25%) in the alirocumab monotherapy group and 12 patients (20%) in the ezetimibe plus statin therapy group took fish oil. In the ezetimibe plus statin therapy group, 2 patients (3%) took gemfibrozil, and 2 patients (3%) took fenofibrate. Six (30%) patients in the alirocumab monotherapy group and 12 (20%) patients in the ezetimibe plus statin therapy group had chronic kidney disease. Although the majority of patients in each group did not use tobacco products, there were more tobacco users in the ezetimibe plus statin therapy group.
In the alirocumab monotherapy group, 15 patients (75%) were prescribed 75 mg every 2 weeks and 5 patients (25%) were prescribed 150 mg every 2 weeks. In the ezetimibe plus statin therapy group, 59 patients (98%) were prescribed ezetimibe 10 mg/d (Table 2). Forty-three patients (72%) were prescribed a high-intensity statin 10 received moderate-intensity (17%) and 7 received low-intensity statin (12%). Most patients were prescribed rosuvastatin (45%), followed by atorvastatin (42%), pravastatin (10%), and simvastatin (3%).
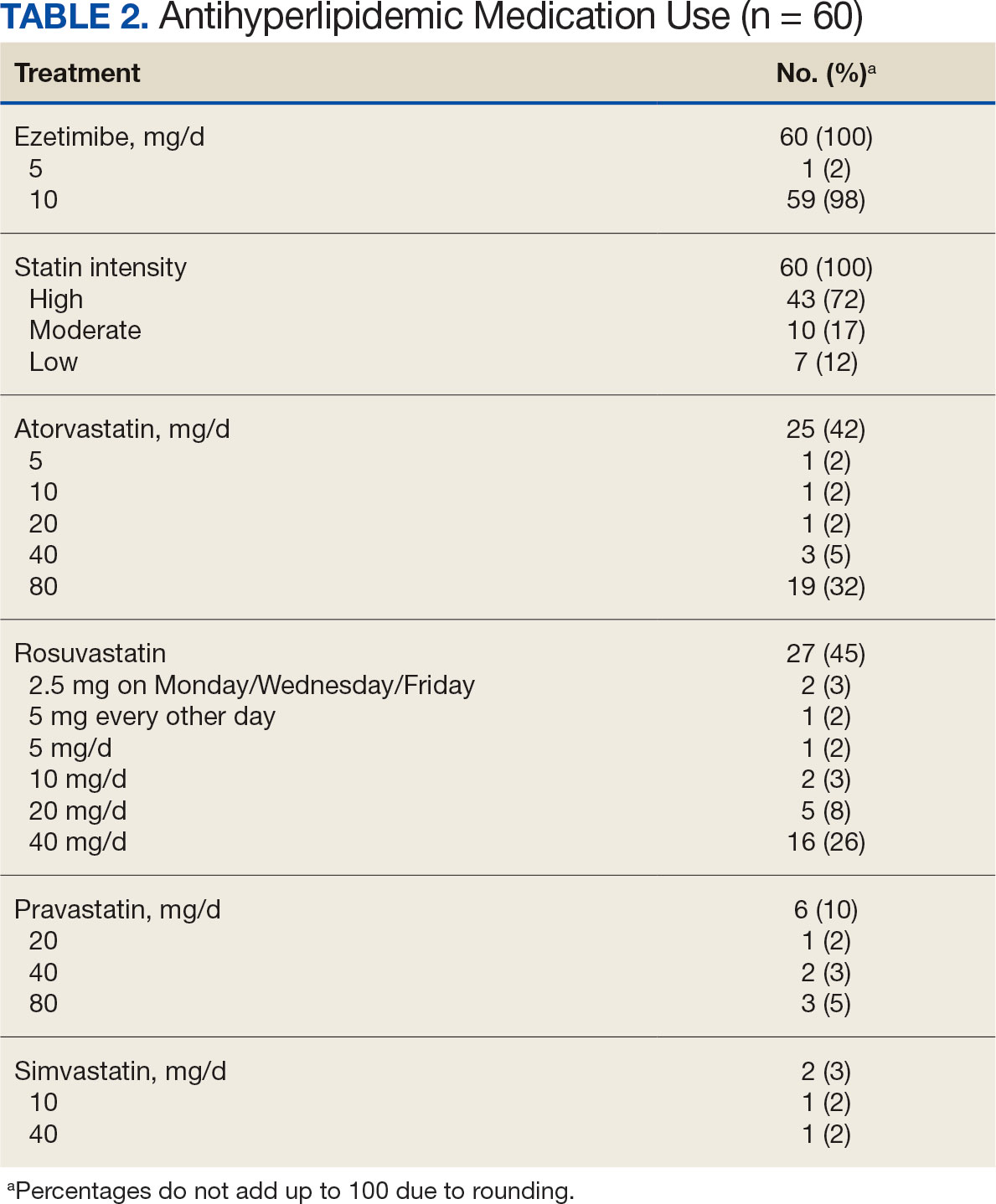
Primary Endpoint
During the 52-week study, more patients met the LDL-C goal of < 70 mg/dL in the alirocumab monotherapy group (70%) than in the ezetimibe plus statin therapy group (57%); however, the difference was not significant (P = .29). Of the patients prescribed alirocumab monotherapy who achieved LDL-C < 70 mg/dL, 15% achieved this goal in 4 to 12 weeks, 40% in 13 to 24 weeks, and 45% in 25 to 52 weeks. In the ezetimibe plus statin therapy group, 28% of patients achieved LDL-C < 70 mg/dL in 4 to 12 weeks, 31% in 13 to 24 weeks, and 41% in 25 to 52 weeks (Table 3).
Secondary Endpoints
During weeks 4 to 52 of treatment, the mean percentage change decreased in LDL-C (37.7% vs 21.4%; P = .01), TC (24.7% vs 12.5%; P = .01), and TG (0.9% vs 7.0%; P = .28) in the alirocumab monotherapy group and the ezetimibe plus statin therapy group, respectively (Table 4). The mean percentage change increased in HDL-C by 3.6% in the alirocumab monotherapy group and 1.8% in the ezetimibe plus statin therapy group (P = .36). During the study, ASCVD events occurred in 1 patient (5%) in the alirocumab monotherapy group and 3 patients (5%) in the ezetimibe plus statin therapy group (P = .99). The patient in the alirocumab monotherapy group had unstable angina 1 month after taking alirocumab. One patient in the ezetimibe plus statin therapy group had coronary artery disease and 2 patients had coronary heart disease that required stents during the 52-week period. There was 1 patient in each group who reported an AE that led to treatment discontinuation (P = .41). One patient stopped alirocumab after a trial of 2 months due to intolerance, but no specific AE was reported in the CPRS. In the ezetimibe plus statin therapy group, 1 patient requested to discontinue ezetimibe after a trial of 3 months without a specific reason noted in the medical record.
DISCUSSION
This study found no statistically significant difference in the incidence of reaching an LDL-C goal of < 70 mg/dL after alirocumab monotherapy initiation compared with ezetimibe plus statin therapy. This occurred despite baseline LDL-C being lower in the ezetimibe plus statin therapy group, which required a smaller reduction in LDL-C to reach the primary goal. Most patients on alirocumab monotherapy were prescribed a lower initial dose of 75 mg every 2 weeks. Of those patients, 30% did not achieve the LDL-C goal < 70 mg/dL. Thus, a higher dose may have led to more patients achieving the LDL-C goal.
Secondary endpoints, including mean percentage change in HDL-C and TG and incidence of ASCVD events during 52 weeks of treatment, were not statistically significant. The mean percentage increase in HDL-C was negligible in both groups, while the mean percentage reduction in TG favored the ezetimibe plus statin therapy group. In the ezetimibe plus statin therapy group, patients who also took fenofibrate experienced a significant reduction in TG while none of the patients in the alirocumab group were prescribed fenofibrate. Although the alirocumab monotherapy group had a statistically significant greater reduction in LDL-C and TC compared with those prescribed ezetimibe plus statin, the mean baseline LDL-C and TC were significantly greater in the alirocumab monotherapy group, which could contribute to higher reductions in LDL-C and TC after alirocumab monotherapy.Based on the available literature, we expected greater reductions in LDL-C in both study groups compared with statin therapy alone.8,9 However, it was unclear whether the LDL-C and TC reductions were clinically significant.
Limitations
The study design did not permit randomization prior to the treatments, restricting our ability to account for some confounding factors, such as diet, exercise, other antihyperlipidemic medication, and medication adherence, which may have affected LDL-C, HDL-C, TG, and TC levels. Differences in baseline characteristics—particularly major risk factors, such as hypertension, diabetes, and tobacco use—also could have confounding affect on lipid levels and ASCVD events. Additionally, patients prescribed alirocumab monotherapy may have switched from statin or ezetimibe therapy, and the washout period was not reviewed or recorded, which could have affected the lipid panel results.
The small sample size of this study also may have limited the ability to detect significant differences between groups. A direct comparison of alirocumab monotherapy vs ezetimibe plus statin therapy has not been performed, making it difficult to prospectively evaluate what sample size would be needed to power this study. A posthoc analysis was used to calculate power, which was found to be only 17%. Many patients were excluded due to a lack of laboratory results within the study period, contributing to the small sample size.
Another limitation was the reliance on documentation in CPRS and JLV. For example, having documentation of the specific AEs for the 2 patients who discontinued alirocumab or ezetimibe could have helped determine the severity of the AEs. Several patients were followed by non-VA clinicians, which could have contributed to limited documentation in the CPRS and JLV. It is difficult to draw any conclusions regarding ASCVD events and AEs that led to treatment discontinuation between alirocumab monotherapy and ezetimibe plus statin therapy based on the results of this retrospective study due to the limited number of events within the 52-week period.
CONCLUSIONS
This study found that there was no statistically significant difference in LDL-C reduction to < 70 mg/dL between alirocumab monotherapy and ezetimibe plus statin therapy in a small population of veterans with ASCVD, with a higher percentage of participants in both groups achieving that goal in 25 to 52 weeks. There also was no significant difference in percentage change in HDL-C or TG or in incidence of ASCVD events and AEs leading to treatment discontinuation. However, there was a statistically significant difference in percentage reduction for LDL-C and TC during 52 weeks of alirocumab monotherapy vs ezetimibe plus statin therapy.
Although there was no significant difference in LDL-C reduction to < 70 mg/dL, targeting this goal in patients with ASCVD is still clinically warranted. This study does not support a change in current VA criteria for use of alirocumab or a change in current guidelines for secondary prevention of ASCVD. Still, this study does indicate that the efficacy of alirocumab monotherapy is similar to that of ezetimibe plus statin therapy in patients with a history of ASCVD and may be useful in clinical settings when an alternative to ezetimibe plus statin therapy is needed. Alirocumab also may be more effective in lowering LDL-C and TC than ezetimibe plus statin therapy in veterans with ASCVD and could be added to statin therapy or ezetimibe when additional LDL-C or TC reduction is needed.
Lucchi T. Dyslipidemia and prevention of atherosclerotic cardiovascular disease in the elderly. Minerva Med. 2021;112:804-816. doi:10.23736/S0026-4806.21.07347-X
The Management of Dyslipidemia for Cardiovascular Risk Reduction Work Group. VA/DoD Clinical Practice Guideline for the Management of Dyslipidemia for Cardiovascular Risk Reduction. Version 4.0. June 2020. Accessed September 5, 2024. https://www.healthquality.va.gov/guidelines/CD/lipids/VADoDDyslipidemiaCPG5087212020.pdf
Centers for Disease Control and Prevention. High Cholesterol Facts. May 15, 2024. Accessed October 3, 2024. https://www.cdc.gov/cholesterol/data-research/facts-stats/index.html
Grundy SM, Stone NJ, Bailey AL, et al. 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA guideline on the management of blood cholesterol: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation. 2019;139:e1082-e1143. doi:10.1161/CIR.0000000000000625
Vavlukis M, Vavlukis A. Statins alone or in combination with ezetimibe or PCSK9 inhibitors in atherosclerotic cardiovascular disease protection. IntechOpen. January 24, 2019. doi:10.5772/intechopen.82520
Alirocumab. Prescribing information. Regeneron Pharmaceuticals, Inc.; 2024. Accessed September 5, 2024. https://www.regeneron.com/downloads/praluent_pi.pdf
Pandor A, Ara RM, Tumur I, et al. Ezetimibe monotherapy for cholesterol lowering in 2,722 people: systematic review and meta-analysis of randomized controlled trials. J Intern Med. 2009;265(5):568-580. doi:10.1111/j.1365-2796.2008.02062.x
McKenney J, Ballantyne CM, Feldman TA, et al. LDL-C goal attainment with ezetimibe plus simvastatin coadministration vs atorvastatin or simvastatin monotherapy in patients at high risk of CHD. MedGenMed. 2005;7(3):3.
Stroes E, Guyton JR, Lepor N, et al. Efficacy and safety of alirocumab 150 mg every 4 weeks in patients with hypercholesterolemia not on statin therapy: the ODYSSEY CHOICE II study. J Am Heart Assoc. 2016;5(9):e003421. doi:10.1161/JAHA.116.003421
Atherosclerotic cardiovascular disease (ASCVD) is a significant cause of morbidity and mortality in the United States. ASCVD involves the buildup of cholesterol plaque in arteries and includes acute coronary syndrome, peripheral arterial disease, and events such as myocardial infarction and stroke.1 Cardiovascular disease (CVD) risk factors include high cholesterol levels, elevated blood pressure, insulin resistance, elevated blood glucose levels, smoking, poor dietary habits, and a sedentary lifestyle.2
According to the Centers for Disease Control and Prevention, about 86 million adults aged ≥ 20 years have total cholesterol levels > 200 mg/dL. More than half (54.5%) who could benefit are currently taking cholesterol-lowering medications.3 Controlling high cholesterol in American adults, especially veterans, is essential for reducing CVD morbidity and mortality.
The 2018 American College of Cardiology/American Heart Association (ACC/AHA) guideline recommends a low-density lipoprotein cholesterol (LDL-C) target goal of < 70 mg/dL for patients at high risk for ASCVD. Very high-risk ASCVD includes a history of multiple major ASCVD events or 1 major ASCVD event and multiple high-risk conditions (eg, age ≥ 65 years, smoking, or diabetes).4 Major ASCVD events include recent acute coronary syndrome (within the past 12 months), a history of myocardial infarction or ischemic stroke, and symptomatic peripheral artery disease.
The ACC/AHA guideline suggests that if the LDL-C level remains ≥ 70 mg/dL, adding ezetimibe (a dietary cholesterol absorption inhibitor) to maximally tolerated statin therapy is reasonable. If LDL-C levels remain ≥ 70 mg/dL, adding a proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitor, such as alirocumab, is reasonable.4 The US Departments of Veterans Affairs/US Department of Defense guidelines recommend using maximally tolerated statins and ezetimibe before PCSK9 inhibitors due to established long-term safety and reduction in CVD events.
Generic statins and ezetimibe are administered orally and widely available. In contrast, PCSK9 inhibitors have unknown long-term safety profiles, require subcutaneous injection once or twice monthly, and are significantly more expensive. They also require patient education on proper use while providing comparable or lesser relative risk reductions.2
These 3 classes of medication vary in their mechanisms of action to reduce LDL.5,6 Ezetimibe and several statin medications are included on the Veterans Affairs Sioux Falls Health Care System (VASFHCS) formulary and do not require review prior to prescribing. Alirocumab is available at VASFHCS but is restricted to patients with a history of ASCVD or a diagnosis of familial hypercholesterolemia, and who are receiving maximally tolerated statin and ezetimibe therapy but require further LDL-C lowering to reduce their ASCVD risk.
Studies have found ezetimibe monotherapy reduces LDL-C in patients with dyslipidemia by 18% after 12 weeks.7 One found that the percentage reduction in LDL-C was significantly greater (P < .001) with all doses of ezetimibe plus simvastatin (46% to 59%) compared with either atorvastatin 10 mg (37%) or simvastatin 20 mg (38%) monotherapy after 6 weeks.8
Although alirocumab can be added to other lipid therapies, most VASFHCS patients are prescribed alirocumab monotherapy. In the ODYSSEY CHOICE II study, patients were randomly assigned to receive either a placebo or alirocumab 150 mg every 4 weeks or alirocumab 75 mg every 2 weeks. The primary efficacy endpoint was LDL-C percentage change from baseline to week 24. In the alirocumab 150 mg every 4 weeks and 75 mg every 2 weeks groups, the least-squares mean LDL-C changes from baseline to week 24 were 51.7% and 53.5%, respectively, compared to a 4.7% increase in the placebo group (both groups P < .001 vs placebo). The authors also reported that alirocumab 150 mg every 4 weeks as monotherapy demonstrated a 47.4% reduction in LDL-C levels from baseline in a phase 1 study.9Although alirocumab monotherapy and ezetimibe plus statin therapy have been shown to effectively decrease LDL-C independently, a direct comparison of alirocumab monotherapy vs ezetimibe plus statin therapy has not been assessed, to our knowledge. Understanding the differences in effectiveness and safety between these 2 regimens will be valuable for clinicians when selecting a medication regimen for veterans with a history of ASCVD.
METHODS
This retrospective, single-center chart review used VASFHCS Computerized Patient Record System (CPRS) and Joint Longitudinal Viewer (JLV) records to compare patients with a history of ASCVD events who were treated with alirocumab monotherapy or ezetimibe plus statin. The 2 groups were randomized in a 1:3 ratio. The primary endpoint was achieving LDL-C < 70 mg/dL after 4 to 12 weeks, 13 to 24 weeks, and 25 to 52 weeks. Secondary endpoints included the mean percentage change from baseline in total cholesterol (TC), high-density lipoprotein cholesterol (HDL-C), LDL-C, and triglycerides (TG) over 52 weeks. The incidence of ASCVD events during this period was also assessed. If LDL-C < 70 mg/dL was achieved > 1 time during each time frame, only 1 incident was counted for analysis. Safety was assessed based on the incidence of any adverse event (AE) that led to treatment discontinuation.
Patients were identified by screening the prescription fill history between October 1, 2019, and December 31, 2022. The 52-week data collection period was counted from the first available fill date. Additionally, the prior authorization drug request file from January 1, 2017, to December 31, 2022, was used to obtain a list of patients prescribed alirocumab. Patients were included if they were veterans aged ≥ 18 years and had a history of an ASCVD event, had a alirocumab monotherapy or ezetimibe plus statin prescription between October 1, 2019, and December 31, 2022, or had an approved prior authorization drug request for alirocumab between January 1, 2017, and December 31, 2022. Patients missing a baseline or follow-up lipid panel and those with concurrent use of alirocumab and ezetimibe and/or statin were excluded.
Baseline characteristics collected for patients included age, sex, race, weight, body mass index, lipid parameters (LDL-C, TC, HDL-C, and TG), dosing of each type of statin before adding ezetimibe, and use of any other antihyperlipidemic medication. We also collected histories of hypertension, hyperlipidemia, diabetes, chronic kidney disease, congestive heart failure, and smoking or tobacco use status. The baseline lipid panel was the most recent lipid panel documented before starting alirocumab or ezetimibe plus statin therapy. Follow-up lipid panel values were gathered at 4 to 12 weeks, 13 to 24 weeks, and 25 to 52 weeks following initiation of either therapy.
High-, moderate-, and low-intensity dosing of statin therapy and alirocumab dosing (75 mg every 2 weeks, 150 mg every 2 weeks, or 300 mg every 4 weeks) were recorded at the specified intervals. However, no patients in this study received the latter dosing regimen. ASCVD events and safety endpoints were recorded based on a review of clinical notes over the 52 weeks following the first available start date.
Statistical Analysis
The primary endpoint of achieving the LDL-C < 70 mg/dL goal from baseline to 4 to 12 weeks, 13 to 24 weeks, and 25 to 52 weeks after initiation was compared between alirocumab monotherapy and ezetimibe plus statin therapy using the χ² test. Mean percentage change from baseline in TC, HDL-C, LDL-C, and TG were compared using the independent t test. P < .05 was considered statistically significant. Incidence of ASCVD events and the safety endpoint (incidence of AEs leading to treatment discontinuation) were also compared using the χ² test. Continuous baseline characteristics were reported mean (SD) and nominal baseline characteristics were reported as a percentage.
RESULTS
There were 80 participants in this study: 20 in the alirocumab monotherapy group and 60 in the ezetimibe plus statin therapy group. More than 100 patients did not meet the prespecified inclusion criteria and were excluded. Mean (SD) age was 75 (8) years in the alirocumab group and 74 (8) years in the ezetimibe plus statin group. There was no significant differences in mean (SD) weight or mean (SD) body mass index. All study participants identified as White and male except for 2 patients in the ezetimibe plus statin therapy group whose race was not documented. Differences in lipid parameters were observed between groups, with mean baseline LDL-C, HDL-C, and TC higher in the alirocumab monotherapy group than in the ezetimibe plus statin therapy group, with significant differences in LDL-C and TC (Table 1).

Fourteen patients (70%) in the alirocumab monotherapy group had hypertension, compared with 31 (52%) in the ezetimibe plus statin therapy group. In both groups, most patients had previously been diagnosed with hyperlipidemia. More patients (60%) in the alirocumab group had diabetes than in the ezetimibe plus statin therapy group (37%). The alirocumab monotherapy group also had a higher percentage of patients with diagnoses of congestive heart failure and used other antihyperlipidemic medications than in the ezetimibe plus statin therapy group. Five patients (25%) in the alirocumab monotherapy group and 12 patients (20%) in the ezetimibe plus statin therapy group took fish oil. In the ezetimibe plus statin therapy group, 2 patients (3%) took gemfibrozil, and 2 patients (3%) took fenofibrate. Six (30%) patients in the alirocumab monotherapy group and 12 (20%) patients in the ezetimibe plus statin therapy group had chronic kidney disease. Although the majority of patients in each group did not use tobacco products, there were more tobacco users in the ezetimibe plus statin therapy group.
In the alirocumab monotherapy group, 15 patients (75%) were prescribed 75 mg every 2 weeks and 5 patients (25%) were prescribed 150 mg every 2 weeks. In the ezetimibe plus statin therapy group, 59 patients (98%) were prescribed ezetimibe 10 mg/d (Table 2). Forty-three patients (72%) were prescribed a high-intensity statin 10 received moderate-intensity (17%) and 7 received low-intensity statin (12%). Most patients were prescribed rosuvastatin (45%), followed by atorvastatin (42%), pravastatin (10%), and simvastatin (3%).

Primary Endpoint
During the 52-week study, more patients met the LDL-C goal of < 70 mg/dL in the alirocumab monotherapy group (70%) than in the ezetimibe plus statin therapy group (57%); however, the difference was not significant (P = .29). Of the patients prescribed alirocumab monotherapy who achieved LDL-C < 70 mg/dL, 15% achieved this goal in 4 to 12 weeks, 40% in 13 to 24 weeks, and 45% in 25 to 52 weeks. In the ezetimibe plus statin therapy group, 28% of patients achieved LDL-C < 70 mg/dL in 4 to 12 weeks, 31% in 13 to 24 weeks, and 41% in 25 to 52 weeks (Table 3).

Secondary Endpoints
During weeks 4 to 52 of treatment, the mean percentage change decreased in LDL-C (37.7% vs 21.4%; P = .01), TC (24.7% vs 12.5%; P = .01), and TG (0.9% vs 7.0%; P = .28) in the alirocumab monotherapy group and the ezetimibe plus statin therapy group, respectively (Table 4). The mean percentage change increased in HDL-C by 3.6% in the alirocumab monotherapy group and 1.8% in the ezetimibe plus statin therapy group (P = .36). During the study, ASCVD events occurred in 1 patient (5%) in the alirocumab monotherapy group and 3 patients (5%) in the ezetimibe plus statin therapy group (P = .99). The patient in the alirocumab monotherapy group had unstable angina 1 month after taking alirocumab. One patient in the ezetimibe plus statin therapy group had coronary artery disease and 2 patients had coronary heart disease that required stents during the 52-week period. There was 1 patient in each group who reported an AE that led to treatment discontinuation (P = .41). One patient stopped alirocumab after a trial of 2 months due to intolerance, but no specific AE was reported in the CPRS. In the ezetimibe plus statin therapy group, 1 patient requested to discontinue ezetimibe after a trial of 3 months without a specific reason noted in the medical record.

DISCUSSION
This study found no statistically significant difference in the incidence of reaching an LDL-C goal of < 70 mg/dL after alirocumab monotherapy initiation compared with ezetimibe plus statin therapy. This occurred despite baseline LDL-C being lower in the ezetimibe plus statin therapy group, which required a smaller reduction in LDL-C to reach the primary goal. Most patients on alirocumab monotherapy were prescribed a lower initial dose of 75 mg every 2 weeks. Of those patients, 30% did not achieve the LDL-C goal < 70 mg/dL. Thus, a higher dose may have led to more patients achieving the LDL-C goal.
Secondary endpoints, including mean percentage change in HDL-C and TG and incidence of ASCVD events during 52 weeks of treatment, were not statistically significant. The mean percentage increase in HDL-C was negligible in both groups, while the mean percentage reduction in TG favored the ezetimibe plus statin therapy group. In the ezetimibe plus statin therapy group, patients who also took fenofibrate experienced a significant reduction in TG while none of the patients in the alirocumab group were prescribed fenofibrate. Although the alirocumab monotherapy group had a statistically significant greater reduction in LDL-C and TC compared with those prescribed ezetimibe plus statin, the mean baseline LDL-C and TC were significantly greater in the alirocumab monotherapy group, which could contribute to higher reductions in LDL-C and TC after alirocumab monotherapy.Based on the available literature, we expected greater reductions in LDL-C in both study groups compared with statin therapy alone.8,9 However, it was unclear whether the LDL-C and TC reductions were clinically significant.
Limitations
The study design did not permit randomization prior to the treatments, restricting our ability to account for some confounding factors, such as diet, exercise, other antihyperlipidemic medication, and medication adherence, which may have affected LDL-C, HDL-C, TG, and TC levels. Differences in baseline characteristics—particularly major risk factors, such as hypertension, diabetes, and tobacco use—also could have confounding affect on lipid levels and ASCVD events. Additionally, patients prescribed alirocumab monotherapy may have switched from statin or ezetimibe therapy, and the washout period was not reviewed or recorded, which could have affected the lipid panel results.
The small sample size of this study also may have limited the ability to detect significant differences between groups. A direct comparison of alirocumab monotherapy vs ezetimibe plus statin therapy has not been performed, making it difficult to prospectively evaluate what sample size would be needed to power this study. A posthoc analysis was used to calculate power, which was found to be only 17%. Many patients were excluded due to a lack of laboratory results within the study period, contributing to the small sample size.
Another limitation was the reliance on documentation in CPRS and JLV. For example, having documentation of the specific AEs for the 2 patients who discontinued alirocumab or ezetimibe could have helped determine the severity of the AEs. Several patients were followed by non-VA clinicians, which could have contributed to limited documentation in the CPRS and JLV. It is difficult to draw any conclusions regarding ASCVD events and AEs that led to treatment discontinuation between alirocumab monotherapy and ezetimibe plus statin therapy based on the results of this retrospective study due to the limited number of events within the 52-week period.
CONCLUSIONS
This study found that there was no statistically significant difference in LDL-C reduction to < 70 mg/dL between alirocumab monotherapy and ezetimibe plus statin therapy in a small population of veterans with ASCVD, with a higher percentage of participants in both groups achieving that goal in 25 to 52 weeks. There also was no significant difference in percentage change in HDL-C or TG or in incidence of ASCVD events and AEs leading to treatment discontinuation. However, there was a statistically significant difference in percentage reduction for LDL-C and TC during 52 weeks of alirocumab monotherapy vs ezetimibe plus statin therapy.
Although there was no significant difference in LDL-C reduction to < 70 mg/dL, targeting this goal in patients with ASCVD is still clinically warranted. This study does not support a change in current VA criteria for use of alirocumab or a change in current guidelines for secondary prevention of ASCVD. Still, this study does indicate that the efficacy of alirocumab monotherapy is similar to that of ezetimibe plus statin therapy in patients with a history of ASCVD and may be useful in clinical settings when an alternative to ezetimibe plus statin therapy is needed. Alirocumab also may be more effective in lowering LDL-C and TC than ezetimibe plus statin therapy in veterans with ASCVD and could be added to statin therapy or ezetimibe when additional LDL-C or TC reduction is needed.
Atherosclerotic cardiovascular disease (ASCVD) is a significant cause of morbidity and mortality in the United States. ASCVD involves the buildup of cholesterol plaque in arteries and includes acute coronary syndrome, peripheral arterial disease, and events such as myocardial infarction and stroke.1 Cardiovascular disease (CVD) risk factors include high cholesterol levels, elevated blood pressure, insulin resistance, elevated blood glucose levels, smoking, poor dietary habits, and a sedentary lifestyle.2
According to the Centers for Disease Control and Prevention, about 86 million adults aged ≥ 20 years have total cholesterol levels > 200 mg/dL. More than half (54.5%) who could benefit are currently taking cholesterol-lowering medications.3 Controlling high cholesterol in American adults, especially veterans, is essential for reducing CVD morbidity and mortality.
The 2018 American College of Cardiology/American Heart Association (ACC/AHA) guideline recommends a low-density lipoprotein cholesterol (LDL-C) target goal of < 70 mg/dL for patients at high risk for ASCVD. Very high-risk ASCVD includes a history of multiple major ASCVD events or 1 major ASCVD event and multiple high-risk conditions (eg, age ≥ 65 years, smoking, or diabetes).4 Major ASCVD events include recent acute coronary syndrome (within the past 12 months), a history of myocardial infarction or ischemic stroke, and symptomatic peripheral artery disease.
The ACC/AHA guideline suggests that if the LDL-C level remains ≥ 70 mg/dL, adding ezetimibe (a dietary cholesterol absorption inhibitor) to maximally tolerated statin therapy is reasonable. If LDL-C levels remain ≥ 70 mg/dL, adding a proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitor, such as alirocumab, is reasonable.4 The US Departments of Veterans Affairs/US Department of Defense guidelines recommend using maximally tolerated statins and ezetimibe before PCSK9 inhibitors due to established long-term safety and reduction in CVD events.
Generic statins and ezetimibe are administered orally and widely available. In contrast, PCSK9 inhibitors have unknown long-term safety profiles, require subcutaneous injection once or twice monthly, and are significantly more expensive. They also require patient education on proper use while providing comparable or lesser relative risk reductions.2
These 3 classes of medication vary in their mechanisms of action to reduce LDL.5,6 Ezetimibe and several statin medications are included on the Veterans Affairs Sioux Falls Health Care System (VASFHCS) formulary and do not require review prior to prescribing. Alirocumab is available at VASFHCS but is restricted to patients with a history of ASCVD or a diagnosis of familial hypercholesterolemia, and who are receiving maximally tolerated statin and ezetimibe therapy but require further LDL-C lowering to reduce their ASCVD risk.
Studies have found ezetimibe monotherapy reduces LDL-C in patients with dyslipidemia by 18% after 12 weeks.7 One found that the percentage reduction in LDL-C was significantly greater (P < .001) with all doses of ezetimibe plus simvastatin (46% to 59%) compared with either atorvastatin 10 mg (37%) or simvastatin 20 mg (38%) monotherapy after 6 weeks.8
Although alirocumab can be added to other lipid therapies, most VASFHCS patients are prescribed alirocumab monotherapy. In the ODYSSEY CHOICE II study, patients were randomly assigned to receive either a placebo or alirocumab 150 mg every 4 weeks or alirocumab 75 mg every 2 weeks. The primary efficacy endpoint was LDL-C percentage change from baseline to week 24. In the alirocumab 150 mg every 4 weeks and 75 mg every 2 weeks groups, the least-squares mean LDL-C changes from baseline to week 24 were 51.7% and 53.5%, respectively, compared to a 4.7% increase in the placebo group (both groups P < .001 vs placebo). The authors also reported that alirocumab 150 mg every 4 weeks as monotherapy demonstrated a 47.4% reduction in LDL-C levels from baseline in a phase 1 study.9Although alirocumab monotherapy and ezetimibe plus statin therapy have been shown to effectively decrease LDL-C independently, a direct comparison of alirocumab monotherapy vs ezetimibe plus statin therapy has not been assessed, to our knowledge. Understanding the differences in effectiveness and safety between these 2 regimens will be valuable for clinicians when selecting a medication regimen for veterans with a history of ASCVD.
METHODS
This retrospective, single-center chart review used VASFHCS Computerized Patient Record System (CPRS) and Joint Longitudinal Viewer (JLV) records to compare patients with a history of ASCVD events who were treated with alirocumab monotherapy or ezetimibe plus statin. The 2 groups were randomized in a 1:3 ratio. The primary endpoint was achieving LDL-C < 70 mg/dL after 4 to 12 weeks, 13 to 24 weeks, and 25 to 52 weeks. Secondary endpoints included the mean percentage change from baseline in total cholesterol (TC), high-density lipoprotein cholesterol (HDL-C), LDL-C, and triglycerides (TG) over 52 weeks. The incidence of ASCVD events during this period was also assessed. If LDL-C < 70 mg/dL was achieved > 1 time during each time frame, only 1 incident was counted for analysis. Safety was assessed based on the incidence of any adverse event (AE) that led to treatment discontinuation.
Patients were identified by screening the prescription fill history between October 1, 2019, and December 31, 2022. The 52-week data collection period was counted from the first available fill date. Additionally, the prior authorization drug request file from January 1, 2017, to December 31, 2022, was used to obtain a list of patients prescribed alirocumab. Patients were included if they were veterans aged ≥ 18 years and had a history of an ASCVD event, had a alirocumab monotherapy or ezetimibe plus statin prescription between October 1, 2019, and December 31, 2022, or had an approved prior authorization drug request for alirocumab between January 1, 2017, and December 31, 2022. Patients missing a baseline or follow-up lipid panel and those with concurrent use of alirocumab and ezetimibe and/or statin were excluded.
Baseline characteristics collected for patients included age, sex, race, weight, body mass index, lipid parameters (LDL-C, TC, HDL-C, and TG), dosing of each type of statin before adding ezetimibe, and use of any other antihyperlipidemic medication. We also collected histories of hypertension, hyperlipidemia, diabetes, chronic kidney disease, congestive heart failure, and smoking or tobacco use status. The baseline lipid panel was the most recent lipid panel documented before starting alirocumab or ezetimibe plus statin therapy. Follow-up lipid panel values were gathered at 4 to 12 weeks, 13 to 24 weeks, and 25 to 52 weeks following initiation of either therapy.
High-, moderate-, and low-intensity dosing of statin therapy and alirocumab dosing (75 mg every 2 weeks, 150 mg every 2 weeks, or 300 mg every 4 weeks) were recorded at the specified intervals. However, no patients in this study received the latter dosing regimen. ASCVD events and safety endpoints were recorded based on a review of clinical notes over the 52 weeks following the first available start date.
Statistical Analysis
The primary endpoint of achieving the LDL-C < 70 mg/dL goal from baseline to 4 to 12 weeks, 13 to 24 weeks, and 25 to 52 weeks after initiation was compared between alirocumab monotherapy and ezetimibe plus statin therapy using the χ² test. Mean percentage change from baseline in TC, HDL-C, LDL-C, and TG were compared using the independent t test. P < .05 was considered statistically significant. Incidence of ASCVD events and the safety endpoint (incidence of AEs leading to treatment discontinuation) were also compared using the χ² test. Continuous baseline characteristics were reported mean (SD) and nominal baseline characteristics were reported as a percentage.
RESULTS
There were 80 participants in this study: 20 in the alirocumab monotherapy group and 60 in the ezetimibe plus statin therapy group. More than 100 patients did not meet the prespecified inclusion criteria and were excluded. Mean (SD) age was 75 (8) years in the alirocumab group and 74 (8) years in the ezetimibe plus statin group. There was no significant differences in mean (SD) weight or mean (SD) body mass index. All study participants identified as White and male except for 2 patients in the ezetimibe plus statin therapy group whose race was not documented. Differences in lipid parameters were observed between groups, with mean baseline LDL-C, HDL-C, and TC higher in the alirocumab monotherapy group than in the ezetimibe plus statin therapy group, with significant differences in LDL-C and TC (Table 1).

Fourteen patients (70%) in the alirocumab monotherapy group had hypertension, compared with 31 (52%) in the ezetimibe plus statin therapy group. In both groups, most patients had previously been diagnosed with hyperlipidemia. More patients (60%) in the alirocumab group had diabetes than in the ezetimibe plus statin therapy group (37%). The alirocumab monotherapy group also had a higher percentage of patients with diagnoses of congestive heart failure and used other antihyperlipidemic medications than in the ezetimibe plus statin therapy group. Five patients (25%) in the alirocumab monotherapy group and 12 patients (20%) in the ezetimibe plus statin therapy group took fish oil. In the ezetimibe plus statin therapy group, 2 patients (3%) took gemfibrozil, and 2 patients (3%) took fenofibrate. Six (30%) patients in the alirocumab monotherapy group and 12 (20%) patients in the ezetimibe plus statin therapy group had chronic kidney disease. Although the majority of patients in each group did not use tobacco products, there were more tobacco users in the ezetimibe plus statin therapy group.
In the alirocumab monotherapy group, 15 patients (75%) were prescribed 75 mg every 2 weeks and 5 patients (25%) were prescribed 150 mg every 2 weeks. In the ezetimibe plus statin therapy group, 59 patients (98%) were prescribed ezetimibe 10 mg/d (Table 2). Forty-three patients (72%) were prescribed a high-intensity statin 10 received moderate-intensity (17%) and 7 received low-intensity statin (12%). Most patients were prescribed rosuvastatin (45%), followed by atorvastatin (42%), pravastatin (10%), and simvastatin (3%).

Primary Endpoint
During the 52-week study, more patients met the LDL-C goal of < 70 mg/dL in the alirocumab monotherapy group (70%) than in the ezetimibe plus statin therapy group (57%); however, the difference was not significant (P = .29). Of the patients prescribed alirocumab monotherapy who achieved LDL-C < 70 mg/dL, 15% achieved this goal in 4 to 12 weeks, 40% in 13 to 24 weeks, and 45% in 25 to 52 weeks. In the ezetimibe plus statin therapy group, 28% of patients achieved LDL-C < 70 mg/dL in 4 to 12 weeks, 31% in 13 to 24 weeks, and 41% in 25 to 52 weeks (Table 3).

Secondary Endpoints
During weeks 4 to 52 of treatment, the mean percentage change decreased in LDL-C (37.7% vs 21.4%; P = .01), TC (24.7% vs 12.5%; P = .01), and TG (0.9% vs 7.0%; P = .28) in the alirocumab monotherapy group and the ezetimibe plus statin therapy group, respectively (Table 4). The mean percentage change increased in HDL-C by 3.6% in the alirocumab monotherapy group and 1.8% in the ezetimibe plus statin therapy group (P = .36). During the study, ASCVD events occurred in 1 patient (5%) in the alirocumab monotherapy group and 3 patients (5%) in the ezetimibe plus statin therapy group (P = .99). The patient in the alirocumab monotherapy group had unstable angina 1 month after taking alirocumab. One patient in the ezetimibe plus statin therapy group had coronary artery disease and 2 patients had coronary heart disease that required stents during the 52-week period. There was 1 patient in each group who reported an AE that led to treatment discontinuation (P = .41). One patient stopped alirocumab after a trial of 2 months due to intolerance, but no specific AE was reported in the CPRS. In the ezetimibe plus statin therapy group, 1 patient requested to discontinue ezetimibe after a trial of 3 months without a specific reason noted in the medical record.

DISCUSSION
This study found no statistically significant difference in the incidence of reaching an LDL-C goal of < 70 mg/dL after alirocumab monotherapy initiation compared with ezetimibe plus statin therapy. This occurred despite baseline LDL-C being lower in the ezetimibe plus statin therapy group, which required a smaller reduction in LDL-C to reach the primary goal. Most patients on alirocumab monotherapy were prescribed a lower initial dose of 75 mg every 2 weeks. Of those patients, 30% did not achieve the LDL-C goal < 70 mg/dL. Thus, a higher dose may have led to more patients achieving the LDL-C goal.
Secondary endpoints, including mean percentage change in HDL-C and TG and incidence of ASCVD events during 52 weeks of treatment, were not statistically significant. The mean percentage increase in HDL-C was negligible in both groups, while the mean percentage reduction in TG favored the ezetimibe plus statin therapy group. In the ezetimibe plus statin therapy group, patients who also took fenofibrate experienced a significant reduction in TG while none of the patients in the alirocumab group were prescribed fenofibrate. Although the alirocumab monotherapy group had a statistically significant greater reduction in LDL-C and TC compared with those prescribed ezetimibe plus statin, the mean baseline LDL-C and TC were significantly greater in the alirocumab monotherapy group, which could contribute to higher reductions in LDL-C and TC after alirocumab monotherapy.Based on the available literature, we expected greater reductions in LDL-C in both study groups compared with statin therapy alone.8,9 However, it was unclear whether the LDL-C and TC reductions were clinically significant.
Limitations
The study design did not permit randomization prior to the treatments, restricting our ability to account for some confounding factors, such as diet, exercise, other antihyperlipidemic medication, and medication adherence, which may have affected LDL-C, HDL-C, TG, and TC levels. Differences in baseline characteristics—particularly major risk factors, such as hypertension, diabetes, and tobacco use—also could have confounding affect on lipid levels and ASCVD events. Additionally, patients prescribed alirocumab monotherapy may have switched from statin or ezetimibe therapy, and the washout period was not reviewed or recorded, which could have affected the lipid panel results.
The small sample size of this study also may have limited the ability to detect significant differences between groups. A direct comparison of alirocumab monotherapy vs ezetimibe plus statin therapy has not been performed, making it difficult to prospectively evaluate what sample size would be needed to power this study. A posthoc analysis was used to calculate power, which was found to be only 17%. Many patients were excluded due to a lack of laboratory results within the study period, contributing to the small sample size.
Another limitation was the reliance on documentation in CPRS and JLV. For example, having documentation of the specific AEs for the 2 patients who discontinued alirocumab or ezetimibe could have helped determine the severity of the AEs. Several patients were followed by non-VA clinicians, which could have contributed to limited documentation in the CPRS and JLV. It is difficult to draw any conclusions regarding ASCVD events and AEs that led to treatment discontinuation between alirocumab monotherapy and ezetimibe plus statin therapy based on the results of this retrospective study due to the limited number of events within the 52-week period.
CONCLUSIONS
This study found that there was no statistically significant difference in LDL-C reduction to < 70 mg/dL between alirocumab monotherapy and ezetimibe plus statin therapy in a small population of veterans with ASCVD, with a higher percentage of participants in both groups achieving that goal in 25 to 52 weeks. There also was no significant difference in percentage change in HDL-C or TG or in incidence of ASCVD events and AEs leading to treatment discontinuation. However, there was a statistically significant difference in percentage reduction for LDL-C and TC during 52 weeks of alirocumab monotherapy vs ezetimibe plus statin therapy.
Although there was no significant difference in LDL-C reduction to < 70 mg/dL, targeting this goal in patients with ASCVD is still clinically warranted. This study does not support a change in current VA criteria for use of alirocumab or a change in current guidelines for secondary prevention of ASCVD. Still, this study does indicate that the efficacy of alirocumab monotherapy is similar to that of ezetimibe plus statin therapy in patients with a history of ASCVD and may be useful in clinical settings when an alternative to ezetimibe plus statin therapy is needed. Alirocumab also may be more effective in lowering LDL-C and TC than ezetimibe plus statin therapy in veterans with ASCVD and could be added to statin therapy or ezetimibe when additional LDL-C or TC reduction is needed.
Lucchi T. Dyslipidemia and prevention of atherosclerotic cardiovascular disease in the elderly. Minerva Med. 2021;112:804-816. doi:10.23736/S0026-4806.21.07347-X
The Management of Dyslipidemia for Cardiovascular Risk Reduction Work Group. VA/DoD Clinical Practice Guideline for the Management of Dyslipidemia for Cardiovascular Risk Reduction. Version 4.0. June 2020. Accessed September 5, 2024. https://www.healthquality.va.gov/guidelines/CD/lipids/VADoDDyslipidemiaCPG5087212020.pdf
Centers for Disease Control and Prevention. High Cholesterol Facts. May 15, 2024. Accessed October 3, 2024. https://www.cdc.gov/cholesterol/data-research/facts-stats/index.html
Grundy SM, Stone NJ, Bailey AL, et al. 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA guideline on the management of blood cholesterol: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation. 2019;139:e1082-e1143. doi:10.1161/CIR.0000000000000625
Vavlukis M, Vavlukis A. Statins alone or in combination with ezetimibe or PCSK9 inhibitors in atherosclerotic cardiovascular disease protection. IntechOpen. January 24, 2019. doi:10.5772/intechopen.82520
Alirocumab. Prescribing information. Regeneron Pharmaceuticals, Inc.; 2024. Accessed September 5, 2024. https://www.regeneron.com/downloads/praluent_pi.pdf
Pandor A, Ara RM, Tumur I, et al. Ezetimibe monotherapy for cholesterol lowering in 2,722 people: systematic review and meta-analysis of randomized controlled trials. J Intern Med. 2009;265(5):568-580. doi:10.1111/j.1365-2796.2008.02062.x
McKenney J, Ballantyne CM, Feldman TA, et al. LDL-C goal attainment with ezetimibe plus simvastatin coadministration vs atorvastatin or simvastatin monotherapy in patients at high risk of CHD. MedGenMed. 2005;7(3):3.
Stroes E, Guyton JR, Lepor N, et al. Efficacy and safety of alirocumab 150 mg every 4 weeks in patients with hypercholesterolemia not on statin therapy: the ODYSSEY CHOICE II study. J Am Heart Assoc. 2016;5(9):e003421. doi:10.1161/JAHA.116.003421
Lucchi T. Dyslipidemia and prevention of atherosclerotic cardiovascular disease in the elderly. Minerva Med. 2021;112:804-816. doi:10.23736/S0026-4806.21.07347-X
The Management of Dyslipidemia for Cardiovascular Risk Reduction Work Group. VA/DoD Clinical Practice Guideline for the Management of Dyslipidemia for Cardiovascular Risk Reduction. Version 4.0. June 2020. Accessed September 5, 2024. https://www.healthquality.va.gov/guidelines/CD/lipids/VADoDDyslipidemiaCPG5087212020.pdf
Centers for Disease Control and Prevention. High Cholesterol Facts. May 15, 2024. Accessed October 3, 2024. https://www.cdc.gov/cholesterol/data-research/facts-stats/index.html
Grundy SM, Stone NJ, Bailey AL, et al. 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA guideline on the management of blood cholesterol: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation. 2019;139:e1082-e1143. doi:10.1161/CIR.0000000000000625
Vavlukis M, Vavlukis A. Statins alone or in combination with ezetimibe or PCSK9 inhibitors in atherosclerotic cardiovascular disease protection. IntechOpen. January 24, 2019. doi:10.5772/intechopen.82520
Alirocumab. Prescribing information. Regeneron Pharmaceuticals, Inc.; 2024. Accessed September 5, 2024. https://www.regeneron.com/downloads/praluent_pi.pdf
Pandor A, Ara RM, Tumur I, et al. Ezetimibe monotherapy for cholesterol lowering in 2,722 people: systematic review and meta-analysis of randomized controlled trials. J Intern Med. 2009;265(5):568-580. doi:10.1111/j.1365-2796.2008.02062.x
McKenney J, Ballantyne CM, Feldman TA, et al. LDL-C goal attainment with ezetimibe plus simvastatin coadministration vs atorvastatin or simvastatin monotherapy in patients at high risk of CHD. MedGenMed. 2005;7(3):3.
Stroes E, Guyton JR, Lepor N, et al. Efficacy and safety of alirocumab 150 mg every 4 weeks in patients with hypercholesterolemia not on statin therapy: the ODYSSEY CHOICE II study. J Am Heart Assoc. 2016;5(9):e003421. doi:10.1161/JAHA.116.003421
Impact and Recovery of VHA Epilepsy Care Services During the COVID-19 Pandemic
The COVID-19 pandemic affected diverse workplaces globally, leading to temporary and permanent changes across the health care landscape. Included among the impacted areas of care were epilepsy and electroencephalogram (EEG) clinicians and services. Surveys among epilepsy specialists and neurophysiologists conducted at the onset of the pandemic to evaluate working conditions include analyses from the American Epilepsy Society (AES), the National Association of Epilepsy Centers (NAEC), the International League Against Epilepsy, and an Italian national survey.1-4 These investigations revealed reductions in epilepsy monitoring unit (EMU) admissions (23% decline), epilepsy surgery (6% decline), inpatient EEG (22% of respondents reported decline), and patients having difficulty accessing epilepsy professionals (28% of respondents reported decline) or obtaining medications (20% of respondents reported decline).1-3
While such research provided evidence for changes to epilepsy care in 2020, there are limited data on subsequent adaptations during the pandemic. These studies did not incorporate data on the spread of COVID-19 or administrative workload numbers to analyze service delivery beyond self reports. This study aimed to address this gap in the literature by highlighting results from longitudinal national surveys conducted at the Epilepsy Centers of Excellence (ECoE), a specialty care service within the Veterans Health Administration (VHA), which annually serves > 9 million veterans.5 The ECoE represents epileptologists and neurophysiologists across the United States at the 17 primary facilities that were established at the time of this survey (2 ECoEs have been added since survey completion) in 4 geographical regions and for which other regional facilities refer patients for diagnostic services or specialty care.6
National surveys were conducted among the ECoE directors regarding adaptations made from May 2020 to June 2022 to provide a comprehensive account of limitations they experienced and how adjustments have been made to improve patient care. Survey responses were compared to administrative workload numbers and COVID-19 spread data from the Centers for Disease Control and Prevention (CDC) to provide a comprehensive analysis of performance during the pandemic.
METHODS
Data were collected as part of a quality improvement initiative by the VHA ECoE; institutional review board approval was not required. An 18-item survey covering 5 broad domains was sent to ECoE directors 4 separate times to accumulate data from 4 time periods: May to June 2020 (T1); December 2020 to February 2021 (T2); July to August 2021 (T3); and June to July 2022 (T4). These periods correspond to the following phases of the pandemic: T1, onset of pandemic; T2, vaccine availability; T3, Delta variant predominant; T4, Omicron variant predominant.
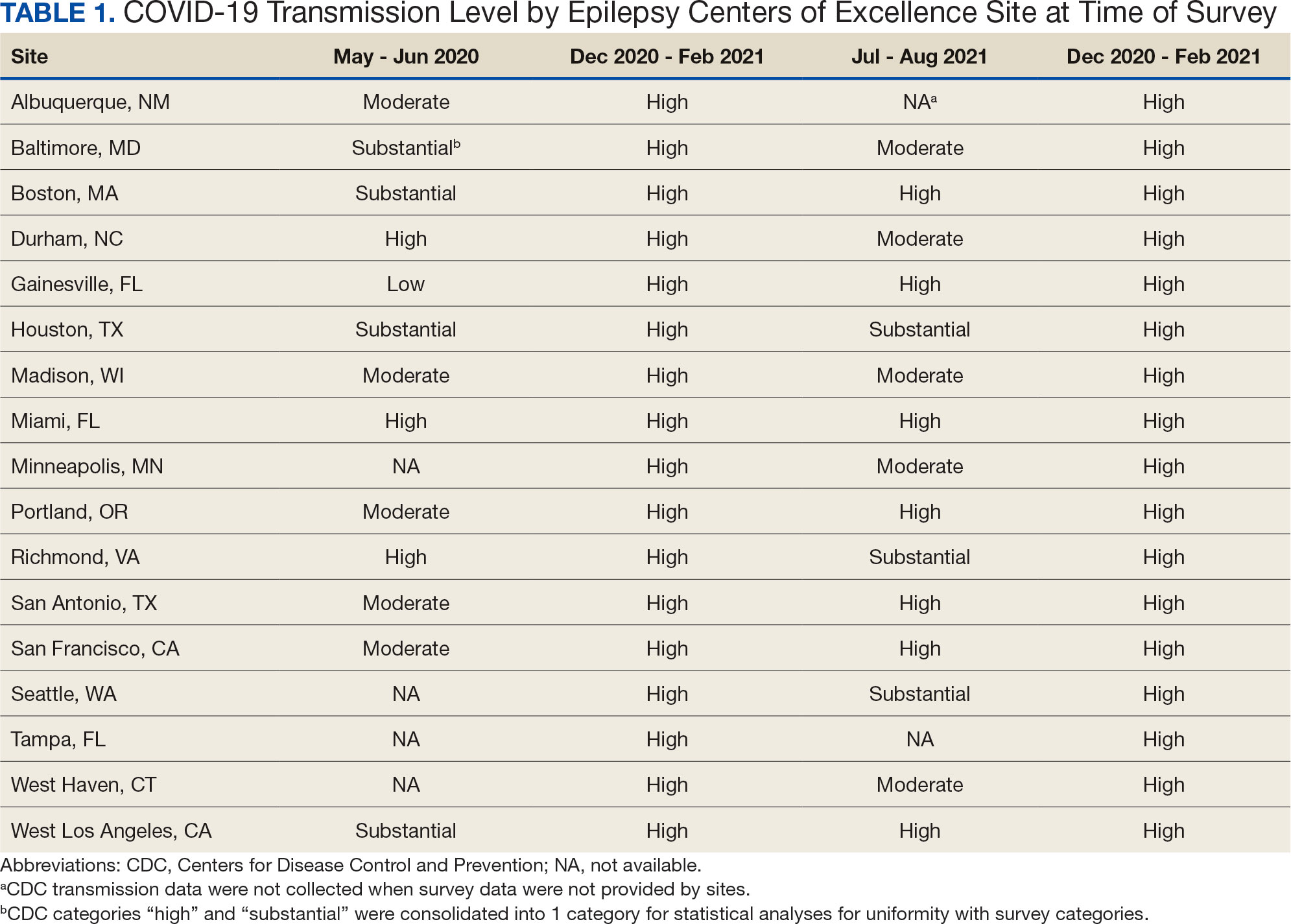
Data on the spread of COVID-19 were collected from the CDC archived dataset, US COVID-19 County Level of Community Transmission Historical Changes (Table 1).7 Administrative workload (patient counts) for EEG, EMU, and outpatient clinics were extracted from VHA administrative databases for the participating sites for the months prior to each survey: T1, April 2020; T2, November 2020; T3, June 2021; and T4, May 2022 (Table 2).
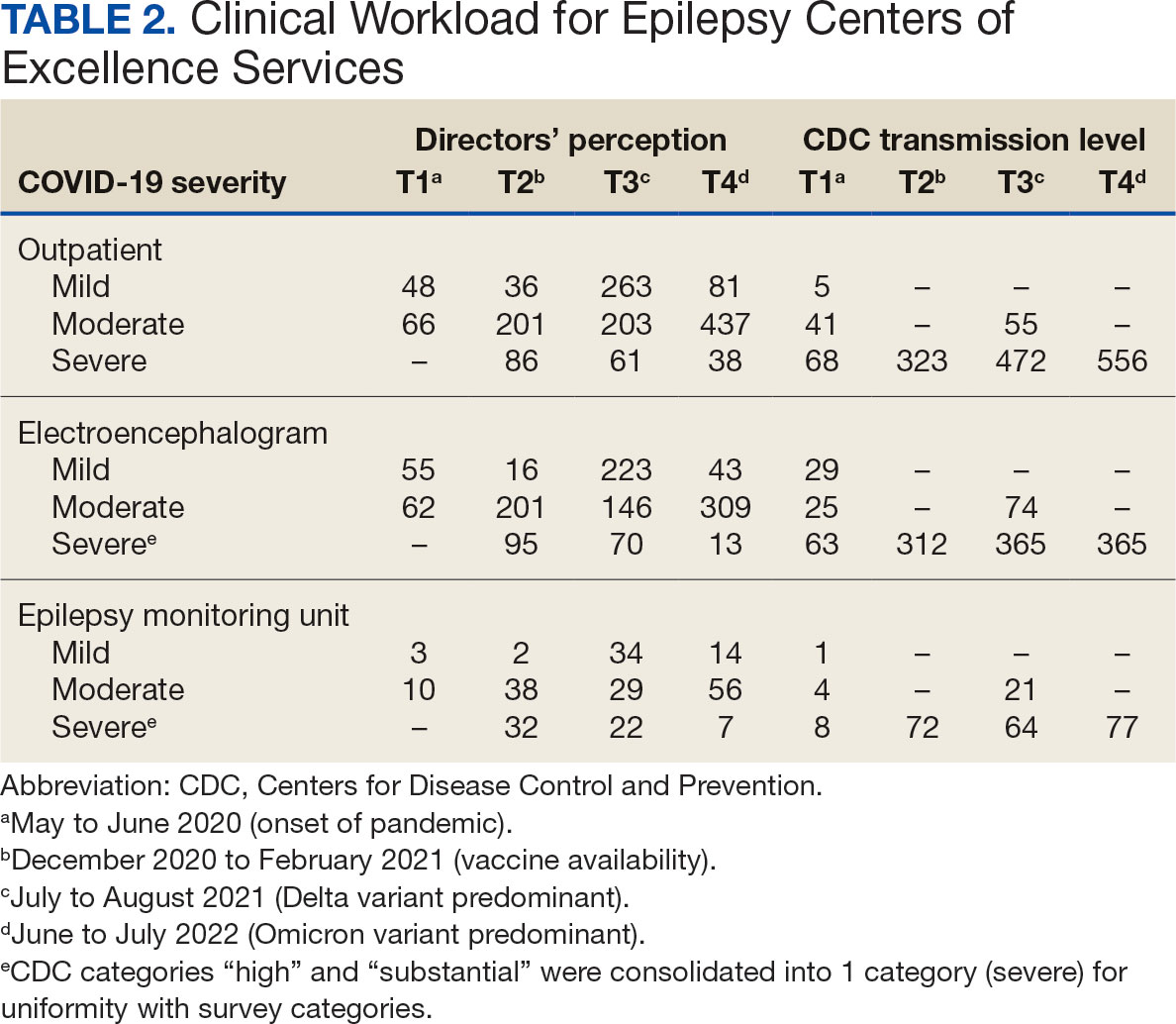
Survey Structure and Content
The survey was developed by the ECoE and was not validated prior to its use due to the time-sensitive nature of gathering information during the pandemic. The first survey (T1) was an emailed spreadsheet with open-ended questions to gauge availability of services (ie, outpatient clinic, EEG, EMU), assess whether safety precautions were being introduced, and understand whether national or local guidelines were thought to be helpful. Responses from this and subsequent surveys were standardized into yes/no and multiple choice formats. Subsequent surveys were administered online using a Research Electronic Data Capture tool.8,9
Availability of outpatient epilepsy services across the 4 time periods were categorized as unlimited (in-person with no restrictions), limited (in-person with restrictions), planned (not currently performed but scheduled for the near future), and unavailable (no in-person services offered) (eAppendices 1-6, available in article PDF).
Statistical Analyses
Analyses were performed to compare survey responses to workload and CDC data on COVID-19 community spread. The following associations were examined: (1) CDC COVID-19 spread vs respondents’ perception of spread; (2) respondents’ perception of spread vs availability of services; (3) CDC COVID-19 spread vs availability of services; (4) respondents’ perception of spread vs workload; and (5) CDC COVID-19 spread vs workload. Availability of services was dichotomized for analyses, with limited or fully available services classified as available. As services were mostly open at T3 regardless of the spread of the virus, and the CDC COVID-19 spread classification for all sites was severe or high at T2 and T4, corresponding associations were not tested at these time points. For associations 1 through 3, Fisher exact tests were used; for associations 4 and 5, Mann-Whitney U tests (where the COVID-19 spread fell into 2 categories) and Kruskal-Wallis tests (for 3 categories of COVID-19 spread) were performed. All tests were 2-tailed and performed at 0.05 error rate. Bonferroni corrections were applied to adjust P values for multiple hypotheses tests.
RESULTS
From the 17 sites invited, responses at each time point were obtained from 13 (T1),17 (T2), 15 (T3), and 16 (T4) centers. There was no significant association between self-reported COVID-19 spread and CDC classification of COVID spread. There were no associations between COVID-19 community spread (respondent reported or CDC severity level) and outpatient clinic availability (self-reported or workload captured). At T3, a positive association was found between the CDC spread level and workload (P = .008), but this was not significant after Bonferroni correction (P = .06).
EEG availability surpassed EMU availability at all time points, although EMU services made some recovery at T3 and T4. No associations were found between COVID-19 community spread (self-reported or CDC severity level) and outpatient EEG or EMU availability (self-reported or workload captured). At T3, there was a positive association between EEG workload and CDC COVID-19 severity level (P = .04), but this was not significant after Bonferroni correction (P = .30).
For outpatient EEG, staff and patient mask use were universally implemented by T2, while the use of full personal protective equipment (PPE) occurred at a subset of sites (T2, 6/17 [35%]; T3, 3/15 [20%]; T4: 4/16 [25%]). COVID-19 testing was rarely implemented prior to outpatient EEG (T1, 0 sites; T2, 1 site; T3, 1 site; T4, 0 sites). Within the EMU, safety precautions including COVID-19 testing, patient mask usage, staff mask usage, and aerosolization demonstrated a sustained majority usage across the 4 surveys.
National and Local Guidelines
The open-ended survey at T1 asked site directors, “Should there be national recommendations on how EEGs and related procedures should be done during the pandemic or should this be left to local conditions?” Responses were mixed, with 5 respondents desiring a national standard, 4 respondents favoring a local response, and 4 respondents believing a national standard should be in place but with modifications based on local outbreak levels and needs.
Surveys performed at T2 through T4 asked, “Which of the following do you feel was/will be helpful in adapting to COVID-19–related changes?” Overall, there was substantial agreement that guidelines were helpful. Most sites anticipated permanent changes in enhanced safety precautions and telehealth.
DISCUSSION
This longitudinal study across 4 time points describes how epilepsy services within the VHA and ECoE adapted to the COVID-19 pandemic. The first survey, conducted 2 months after COVID-19 was declared a pandemic, allowed a comparison with other concurrent US national surveys.1,2,10 The subsequent surveys describe longitudinal adaptations to balance patient and staff safety with service availability and is a unique feature of the current report. Results demonstrate flexibility and adaptability by the ECoEs surveyed, which surprisingly did not show significant associations between CDC COVID-19 spread data and administrative workload data.
Trends in Availability of Services
The most significant impact of COVID-19 restrictions was during T1. There were no significant relationships between service availability/workload and objective CDC COVID-19 spread levels or subjective self-reported COVID-19 spread. Respondents’ perceptions of local COVID-19 spread showed no association with CDC COVID-19 spread data. It appears that subjective perception of spread may be unreliable and factors other than actual or perceived COVID-19 spread were likely driving patterns for service availability.
In-person outpatient visits were most impacted at T1, similar to other civilian surveys, with only 1 site reporting in-person outpatient visits without limitations.1,2 These numbers significantly changed by T2, with all sites offering either limited or unlimited in-person visits. While the surveys did not evaluate factors leading to this rapid recovery, it may be related to the availability of COVID-19 vaccinations within the VHA during this time.11 The US Department of Veterans Affairs was the first federal agency to mandate employee vaccination.12 By the most recent time point (T4), all responding sites offered outpatient visits. Outpatient EEGs followed a similar trend, with T1 being the most restrictive and full, unrestricted outpatient EEGs available by T3.
Fiscal year (FY) trends from ECoE annual reports suggest that encounters slowly recovered over the course of the pandemic. In FY 2019 there were 13,143 outpatient encounters and 6394 EEGs, which dropped to 8097 outpatient encounters and 4432 EEGs in FY 2020 before rising to 8489 outpatient encounters and 5604 EEGs in FY 2021 and 9772 outpatient encounters and 5062 EEGs in FY 2022. Thus, while clinicians described availability of services, patients may have remained hesitant or were otherwise unable to fulfill in-person appointments. The increased availability of home EEG (145 encounter days in 2021 and 436 encounter days in 2022) may be filling this gap.
In contrast to outpatient clinics and EEG, EMU availability showed relatively slower reimplementation. In the last survey, about 30% of sites were still not offering EMU or had limited services. Early trends regarding reduced staffing and patient reluctance for elective admission cited in other surveys may have also affected EMU availability within the VHA.2,13 Consistent with trends in availability, ECoE annual report data suggest EMU patient participation was about one-half of prepandemic rates: 3069 encounters in FY 2019 dropped to 1614 encounters in 2020. By 2021, rates were about two-thirds of prepandemic rates with 2058 encounters in 2021 and 2101 encounters in 2022.
Early survey results (T1) from this study echo trends from other surveys. In the AES survey (April to June 2020), about a quarter of respondents (22%) reported doing fewer EEG studies than usual. The Italian national survey (April 2020) revealed reduced presurgical evaluations (81%), ambulatory EEG (78%), standard EEG (5%) and long-term EEG (32%).4 In the NAEC survey (end of 2020)—which roughly corresponded to T2—outpatient EEGs were still < 75% of pre-COVID levels in one-half of the centers.
National and Local Guidelines
Both national and local guidelines were perceived as useful by most respondents, with national guidelines being more beneficial. This aligns with the NAEC survey, where there was a perceived need for detailed recommendations for PPE and COVID-19 testing of patients, visitors, and staff. Based on national and local guidelines, ECoE implemented safety procedures, as reflected in responses. Staff masking procedures appeared to be the most widely adopted for all services, while the use of full PPE waned as the pandemic progressed. COVID-19 testing was rarely used for routine outpatient visits but common in EMU admissions. This is similar to a survey conducted by the American Academy of Neurology which found full PPE implementation intermittently in outpatient settings and more frequently in inpatient settings.14
Telehealth Attitudes
While most sites anticipated permanent implementation of safety precautions and telehealth, the latter was consistently reported as more likely to be sustained. The VHA had a large and well-developed system of telehealth services that considerably predated the pandemic.15,16 Through this established infrastructure, remote services were quickly increased across the VHA.17-19 This telehealth structure was supplemented by the ability of VHA clinicians to practice across state lines, following a 2018 federal rule.20 The AES survey noted the VHA ECoE's longstanding experience with telehealth as a model for telemedicine use in providing direct patient care, remote EEG analysis, and clinician-to-clinician consultation.1
Trends in the number of telehealth patients seen, observed through patterns in ECoE annual reports are consistent with positive views toward this method of service provision. Specifically, these annual reports capture trends in Video Telehealth Clinic (local station), Video Telehealth Clinic (different station), Home Video Telehealth, Telephone Clinic, and eConsults. Though video telehealth at in-person stations had a precipitous drop in 2020 that continued to wane in subsequent years (898 encounters in 2019; 455 encounters in 2020; 90 encounters in 2021; 88 encounters in 2022), use of home video telehealth rose over time (143 encounters in 2019; 1003 encounters in 2020; 3206 encounters in 2021; 3315 encounters in 2022). Use of telephone services rose drastically in 2020 but has since become a less frequently used service method (2636 in 2019; 5923 in 2020; 5319 in 2021; 3704 in 2022).
Limitations
While the survey encouraged a high response rate, this limited its scope and interpretability. While the availability of services was evaluated, the underlying reasons were not queried. Follow-up questions about barriers to reopening may have allowed for a better understanding of why some services, such as EMU, continued to operate suboptimally later in the pandemic. Similarly, asking about unique strategies or barriers for telehealth would have allowed for a better understanding of its current and future use. We hypothesize that staffing changes during the pandemic may have influenced the availability of services, but changes to staffing were not assessed via the survey and were not readily available via other sources (eg, ECoE annual reports) at the time of publication. An additional limitation is the lack of comparable surveys in the literature for time points T2 to T4, as most analogous surveys were performed early in 2020.
Conclusions
This longitudinal study performed at 4 time points during the COVID-19 pandemic is the first to offer a comprehensive picture of changes to epilepsy and EEG services over time, given that other similar surveys lacked follow-up. Results reveal a significant limitation of services at VHA ECoE shortly after the onset of the pandemic, with return to near-complete operational status 2 years later. While safety precautions and telehealth are predicted to continue, telehealth is perceived as a more permanent change in services.
Albert DVF, Das RR, Acharya JN, et al. The impact of COVID-19 on epilepsy care: a survey of the American Epilepsy Society membership. Epilepsy Curr. 2020;20(5):316-324. doi:10.1177/1535759720956994
Ahrens SM, Ostendorf AP, Lado FA, et al. Impact of the COVID-19 pandemic on epilepsy center practice in the United States. Neurology. 2022;98(19):e1893-e1901. doi:10.1212/WNL.0000000000200285
Cross JH, Kwon CS, Asadi-Pooya AA, et al. Epilepsy care during the COVID-19 pandemic. Epilepsia. 2021;62(10):2322-2332. doi:10.1111/epi.17045
Assenza G, Lanzone J, Ricci L, et al. Electroencephalography at the time of Covid-19 pandemic in Italy. Neurol Sci. 2020;41(8):1999-2004. doi:10.1007/s10072-020-04546-8
US Department of Veterans Affairs. National Center for Veterans Analysis and Statistics. Veteran population. Updated September 7, 2022. Accessed October 25, 2024. https://www.va.gov/vetdata/veteran_population.asp
US Department of Veterans Affairs, Veterans Health Administration. Epilepsy Centers of Excellence (ECoE). Annual report fiscal year 2019. Accessed October 25, 2024. https://www.epilepsy.va.gov/docs/FY19AnnualReport-VHAEpilepsyCentersofExcellence.pdf
Centers for Disease Control and Prevention. United States COVID-19 county level of community transmission historical changes – ARCHIVED. Updated February 20, 2024. Accessed October 25, 2024. https://data.cdc.gov/Public-Health-Surveillance/United-States-COVID-19-County-Level-of-Community-T/nra9-vzzn
Harris PA, Taylor R, Thielke R, Payne J, Gonzalez N, Conde JG. Research electronic data capture (REDCap)—a metadata-driven methodology and workflow process for providing translational research informatics support. J Biomed Inform. 2009;42(2):377-381. doi:10.1016/j.jbi.2008.08.010
Harris PA, Taylor R, Minor BL, et al. The REDCap consortium: building an international community of software platform partners. J Biomed Inform. 2019;95:103208. doi:10.1016/j.jbi.2019.103208
World Health Organization. Rolling updates on coronavirus disease (COVID-19). Updated July 31, 2020. Accessed October 25, 2024. https://www.who.int/emergencies/diseases/novel-coronavirus-2019/events-as-they-happen
US Department of Veterans Affairs. VA announces initial plans for COVID-19 vaccine distribution. News release. December 10, 2020. Accessed October 25, 2024. https://www.va.gov/opa/pressrel/pressrelease.cfm?id=5580
Steinhauer J. V.A. Issues Vaccine Mandate for Health Care Workers, a First for a Federal Agency. The New York Times. August 16, 2021. Accessed October 25, 2024. https://www.nytimes.com/2021/07/26/us/politics/veterans-affairs-coronavirus-covid-19.html
Zafar SF, Khozein RJ, LaRoche SM, Westover MB, Gilmore EJ. Impact of the COVID-19 pandemic on continuous EEG utilization. J Clin Neurophysiol. 2022;39(7):567-574. doi:10.1097/WNP.0000000000000802
Qureshi AI, Rheaume C, Huang W, et al. COVID-19 exposure during neurology practice. Neurologist. 2021;26(6):225-230. doi:10.1097/NRL.0000000000000346
Darkins A, Cruise C, Armstrong M, Peters J, Finn M. Enhancing access of combat-wounded veterans to specialist rehabilitation services: the VA Polytrauma Telehealth Network. Arch Phys Med Rehabil. 2008;89(1):182-187. doi:10.1016/j.apmr.2007.07.027
Darkins A, Ryan P, Kobb R, et al. Care coordination/home telehealth: the systematic implementation of health informatics, home telehealth, and disease management to support the care of veteran patients with chronic conditions. Telemed J E Health. 2008;14(10):1118-1126. doi:10.1089/tmj.2008.0021
Gentry MT, Puspitasari AJ, McKean AJ, et al. Clinician satisfaction with rapid adoption and implementation of telehealth services during the COVID-19 pandemic. Telemed J E Health. 2021;27(12):1385-1392. doi:10.1089/tmj.2020.0575
Connolly SL, Stolzmann KL, Heyworth L, et al. Patient and provider predictors of telemental health use prior to and during the COVID-19 pandemic within the Department of Veterans Affairs. Am Psychol. 2022;77(2):249-261. doi:10.1037/amp0000895
Shelton CJ, Kim A, Hassan AM, Bhat A, Barnello J, Castro CA. System-wide implementation of telehealth to support military veterans and their families in response to COVID-19: a paradigm shift. J Mil Veteran Fam Health. 2020;6(S2):50-57. doi:10.3138/jmvfh-CO19-0003
VA expands telehealth by allowing health care providers to treat patients across state lines. News release. US Dept of Veterans Affairs. May 11, 2018. Accessed October 25, 2024. https://news.va.gov/press-room/va-expands-telehealth-by-allowing-health-care-providers-to-treat-patients-across-state-lines/
The COVID-19 pandemic affected diverse workplaces globally, leading to temporary and permanent changes across the health care landscape. Included among the impacted areas of care were epilepsy and electroencephalogram (EEG) clinicians and services. Surveys among epilepsy specialists and neurophysiologists conducted at the onset of the pandemic to evaluate working conditions include analyses from the American Epilepsy Society (AES), the National Association of Epilepsy Centers (NAEC), the International League Against Epilepsy, and an Italian national survey.1-4 These investigations revealed reductions in epilepsy monitoring unit (EMU) admissions (23% decline), epilepsy surgery (6% decline), inpatient EEG (22% of respondents reported decline), and patients having difficulty accessing epilepsy professionals (28% of respondents reported decline) or obtaining medications (20% of respondents reported decline).1-3
While such research provided evidence for changes to epilepsy care in 2020, there are limited data on subsequent adaptations during the pandemic. These studies did not incorporate data on the spread of COVID-19 or administrative workload numbers to analyze service delivery beyond self reports. This study aimed to address this gap in the literature by highlighting results from longitudinal national surveys conducted at the Epilepsy Centers of Excellence (ECoE), a specialty care service within the Veterans Health Administration (VHA), which annually serves > 9 million veterans.5 The ECoE represents epileptologists and neurophysiologists across the United States at the 17 primary facilities that were established at the time of this survey (2 ECoEs have been added since survey completion) in 4 geographical regions and for which other regional facilities refer patients for diagnostic services or specialty care.6
National surveys were conducted among the ECoE directors regarding adaptations made from May 2020 to June 2022 to provide a comprehensive account of limitations they experienced and how adjustments have been made to improve patient care. Survey responses were compared to administrative workload numbers and COVID-19 spread data from the Centers for Disease Control and Prevention (CDC) to provide a comprehensive analysis of performance during the pandemic.
METHODS
Data were collected as part of a quality improvement initiative by the VHA ECoE; institutional review board approval was not required. An 18-item survey covering 5 broad domains was sent to ECoE directors 4 separate times to accumulate data from 4 time periods: May to June 2020 (T1); December 2020 to February 2021 (T2); July to August 2021 (T3); and June to July 2022 (T4). These periods correspond to the following phases of the pandemic: T1, onset of pandemic; T2, vaccine availability; T3, Delta variant predominant; T4, Omicron variant predominant.

Data on the spread of COVID-19 were collected from the CDC archived dataset, US COVID-19 County Level of Community Transmission Historical Changes (Table 1).7 Administrative workload (patient counts) for EEG, EMU, and outpatient clinics were extracted from VHA administrative databases for the participating sites for the months prior to each survey: T1, April 2020; T2, November 2020; T3, June 2021; and T4, May 2022 (Table 2).

Survey Structure and Content
The survey was developed by the ECoE and was not validated prior to its use due to the time-sensitive nature of gathering information during the pandemic. The first survey (T1) was an emailed spreadsheet with open-ended questions to gauge availability of services (ie, outpatient clinic, EEG, EMU), assess whether safety precautions were being introduced, and understand whether national or local guidelines were thought to be helpful. Responses from this and subsequent surveys were standardized into yes/no and multiple choice formats. Subsequent surveys were administered online using a Research Electronic Data Capture tool.8,9
Availability of outpatient epilepsy services across the 4 time periods were categorized as unlimited (in-person with no restrictions), limited (in-person with restrictions), planned (not currently performed but scheduled for the near future), and unavailable (no in-person services offered) (eAppendices 1-6, available in article PDF).
Statistical Analyses
Analyses were performed to compare survey responses to workload and CDC data on COVID-19 community spread. The following associations were examined: (1) CDC COVID-19 spread vs respondents’ perception of spread; (2) respondents’ perception of spread vs availability of services; (3) CDC COVID-19 spread vs availability of services; (4) respondents’ perception of spread vs workload; and (5) CDC COVID-19 spread vs workload. Availability of services was dichotomized for analyses, with limited or fully available services classified as available. As services were mostly open at T3 regardless of the spread of the virus, and the CDC COVID-19 spread classification for all sites was severe or high at T2 and T4, corresponding associations were not tested at these time points. For associations 1 through 3, Fisher exact tests were used; for associations 4 and 5, Mann-Whitney U tests (where the COVID-19 spread fell into 2 categories) and Kruskal-Wallis tests (for 3 categories of COVID-19 spread) were performed. All tests were 2-tailed and performed at 0.05 error rate. Bonferroni corrections were applied to adjust P values for multiple hypotheses tests.
RESULTS
From the 17 sites invited, responses at each time point were obtained from 13 (T1),17 (T2), 15 (T3), and 16 (T4) centers. There was no significant association between self-reported COVID-19 spread and CDC classification of COVID spread. There were no associations between COVID-19 community spread (respondent reported or CDC severity level) and outpatient clinic availability (self-reported or workload captured). At T3, a positive association was found between the CDC spread level and workload (P = .008), but this was not significant after Bonferroni correction (P = .06).
EEG availability surpassed EMU availability at all time points, although EMU services made some recovery at T3 and T4. No associations were found between COVID-19 community spread (self-reported or CDC severity level) and outpatient EEG or EMU availability (self-reported or workload captured). At T3, there was a positive association between EEG workload and CDC COVID-19 severity level (P = .04), but this was not significant after Bonferroni correction (P = .30).
For outpatient EEG, staff and patient mask use were universally implemented by T2, while the use of full personal protective equipment (PPE) occurred at a subset of sites (T2, 6/17 [35%]; T3, 3/15 [20%]; T4: 4/16 [25%]). COVID-19 testing was rarely implemented prior to outpatient EEG (T1, 0 sites; T2, 1 site; T3, 1 site; T4, 0 sites). Within the EMU, safety precautions including COVID-19 testing, patient mask usage, staff mask usage, and aerosolization demonstrated a sustained majority usage across the 4 surveys.
National and Local Guidelines
The open-ended survey at T1 asked site directors, “Should there be national recommendations on how EEGs and related procedures should be done during the pandemic or should this be left to local conditions?” Responses were mixed, with 5 respondents desiring a national standard, 4 respondents favoring a local response, and 4 respondents believing a national standard should be in place but with modifications based on local outbreak levels and needs.
Surveys performed at T2 through T4 asked, “Which of the following do you feel was/will be helpful in adapting to COVID-19–related changes?” Overall, there was substantial agreement that guidelines were helpful. Most sites anticipated permanent changes in enhanced safety precautions and telehealth.
DISCUSSION
This longitudinal study across 4 time points describes how epilepsy services within the VHA and ECoE adapted to the COVID-19 pandemic. The first survey, conducted 2 months after COVID-19 was declared a pandemic, allowed a comparison with other concurrent US national surveys.1,2,10 The subsequent surveys describe longitudinal adaptations to balance patient and staff safety with service availability and is a unique feature of the current report. Results demonstrate flexibility and adaptability by the ECoEs surveyed, which surprisingly did not show significant associations between CDC COVID-19 spread data and administrative workload data.
Trends in Availability of Services
The most significant impact of COVID-19 restrictions was during T1. There were no significant relationships between service availability/workload and objective CDC COVID-19 spread levels or subjective self-reported COVID-19 spread. Respondents’ perceptions of local COVID-19 spread showed no association with CDC COVID-19 spread data. It appears that subjective perception of spread may be unreliable and factors other than actual or perceived COVID-19 spread were likely driving patterns for service availability.
In-person outpatient visits were most impacted at T1, similar to other civilian surveys, with only 1 site reporting in-person outpatient visits without limitations.1,2 These numbers significantly changed by T2, with all sites offering either limited or unlimited in-person visits. While the surveys did not evaluate factors leading to this rapid recovery, it may be related to the availability of COVID-19 vaccinations within the VHA during this time.11 The US Department of Veterans Affairs was the first federal agency to mandate employee vaccination.12 By the most recent time point (T4), all responding sites offered outpatient visits. Outpatient EEGs followed a similar trend, with T1 being the most restrictive and full, unrestricted outpatient EEGs available by T3.
Fiscal year (FY) trends from ECoE annual reports suggest that encounters slowly recovered over the course of the pandemic. In FY 2019 there were 13,143 outpatient encounters and 6394 EEGs, which dropped to 8097 outpatient encounters and 4432 EEGs in FY 2020 before rising to 8489 outpatient encounters and 5604 EEGs in FY 2021 and 9772 outpatient encounters and 5062 EEGs in FY 2022. Thus, while clinicians described availability of services, patients may have remained hesitant or were otherwise unable to fulfill in-person appointments. The increased availability of home EEG (145 encounter days in 2021 and 436 encounter days in 2022) may be filling this gap.
In contrast to outpatient clinics and EEG, EMU availability showed relatively slower reimplementation. In the last survey, about 30% of sites were still not offering EMU or had limited services. Early trends regarding reduced staffing and patient reluctance for elective admission cited in other surveys may have also affected EMU availability within the VHA.2,13 Consistent with trends in availability, ECoE annual report data suggest EMU patient participation was about one-half of prepandemic rates: 3069 encounters in FY 2019 dropped to 1614 encounters in 2020. By 2021, rates were about two-thirds of prepandemic rates with 2058 encounters in 2021 and 2101 encounters in 2022.
Early survey results (T1) from this study echo trends from other surveys. In the AES survey (April to June 2020), about a quarter of respondents (22%) reported doing fewer EEG studies than usual. The Italian national survey (April 2020) revealed reduced presurgical evaluations (81%), ambulatory EEG (78%), standard EEG (5%) and long-term EEG (32%).4 In the NAEC survey (end of 2020)—which roughly corresponded to T2—outpatient EEGs were still < 75% of pre-COVID levels in one-half of the centers.
National and Local Guidelines
Both national and local guidelines were perceived as useful by most respondents, with national guidelines being more beneficial. This aligns with the NAEC survey, where there was a perceived need for detailed recommendations for PPE and COVID-19 testing of patients, visitors, and staff. Based on national and local guidelines, ECoE implemented safety procedures, as reflected in responses. Staff masking procedures appeared to be the most widely adopted for all services, while the use of full PPE waned as the pandemic progressed. COVID-19 testing was rarely used for routine outpatient visits but common in EMU admissions. This is similar to a survey conducted by the American Academy of Neurology which found full PPE implementation intermittently in outpatient settings and more frequently in inpatient settings.14
Telehealth Attitudes
While most sites anticipated permanent implementation of safety precautions and telehealth, the latter was consistently reported as more likely to be sustained. The VHA had a large and well-developed system of telehealth services that considerably predated the pandemic.15,16 Through this established infrastructure, remote services were quickly increased across the VHA.17-19 This telehealth structure was supplemented by the ability of VHA clinicians to practice across state lines, following a 2018 federal rule.20 The AES survey noted the VHA ECoE's longstanding experience with telehealth as a model for telemedicine use in providing direct patient care, remote EEG analysis, and clinician-to-clinician consultation.1
Trends in the number of telehealth patients seen, observed through patterns in ECoE annual reports are consistent with positive views toward this method of service provision. Specifically, these annual reports capture trends in Video Telehealth Clinic (local station), Video Telehealth Clinic (different station), Home Video Telehealth, Telephone Clinic, and eConsults. Though video telehealth at in-person stations had a precipitous drop in 2020 that continued to wane in subsequent years (898 encounters in 2019; 455 encounters in 2020; 90 encounters in 2021; 88 encounters in 2022), use of home video telehealth rose over time (143 encounters in 2019; 1003 encounters in 2020; 3206 encounters in 2021; 3315 encounters in 2022). Use of telephone services rose drastically in 2020 but has since become a less frequently used service method (2636 in 2019; 5923 in 2020; 5319 in 2021; 3704 in 2022).
Limitations
While the survey encouraged a high response rate, this limited its scope and interpretability. While the availability of services was evaluated, the underlying reasons were not queried. Follow-up questions about barriers to reopening may have allowed for a better understanding of why some services, such as EMU, continued to operate suboptimally later in the pandemic. Similarly, asking about unique strategies or barriers for telehealth would have allowed for a better understanding of its current and future use. We hypothesize that staffing changes during the pandemic may have influenced the availability of services, but changes to staffing were not assessed via the survey and were not readily available via other sources (eg, ECoE annual reports) at the time of publication. An additional limitation is the lack of comparable surveys in the literature for time points T2 to T4, as most analogous surveys were performed early in 2020.
Conclusions
This longitudinal study performed at 4 time points during the COVID-19 pandemic is the first to offer a comprehensive picture of changes to epilepsy and EEG services over time, given that other similar surveys lacked follow-up. Results reveal a significant limitation of services at VHA ECoE shortly after the onset of the pandemic, with return to near-complete operational status 2 years later. While safety precautions and telehealth are predicted to continue, telehealth is perceived as a more permanent change in services.
The COVID-19 pandemic affected diverse workplaces globally, leading to temporary and permanent changes across the health care landscape. Included among the impacted areas of care were epilepsy and electroencephalogram (EEG) clinicians and services. Surveys among epilepsy specialists and neurophysiologists conducted at the onset of the pandemic to evaluate working conditions include analyses from the American Epilepsy Society (AES), the National Association of Epilepsy Centers (NAEC), the International League Against Epilepsy, and an Italian national survey.1-4 These investigations revealed reductions in epilepsy monitoring unit (EMU) admissions (23% decline), epilepsy surgery (6% decline), inpatient EEG (22% of respondents reported decline), and patients having difficulty accessing epilepsy professionals (28% of respondents reported decline) or obtaining medications (20% of respondents reported decline).1-3
While such research provided evidence for changes to epilepsy care in 2020, there are limited data on subsequent adaptations during the pandemic. These studies did not incorporate data on the spread of COVID-19 or administrative workload numbers to analyze service delivery beyond self reports. This study aimed to address this gap in the literature by highlighting results from longitudinal national surveys conducted at the Epilepsy Centers of Excellence (ECoE), a specialty care service within the Veterans Health Administration (VHA), which annually serves > 9 million veterans.5 The ECoE represents epileptologists and neurophysiologists across the United States at the 17 primary facilities that were established at the time of this survey (2 ECoEs have been added since survey completion) in 4 geographical regions and for which other regional facilities refer patients for diagnostic services or specialty care.6
National surveys were conducted among the ECoE directors regarding adaptations made from May 2020 to June 2022 to provide a comprehensive account of limitations they experienced and how adjustments have been made to improve patient care. Survey responses were compared to administrative workload numbers and COVID-19 spread data from the Centers for Disease Control and Prevention (CDC) to provide a comprehensive analysis of performance during the pandemic.
METHODS
Data were collected as part of a quality improvement initiative by the VHA ECoE; institutional review board approval was not required. An 18-item survey covering 5 broad domains was sent to ECoE directors 4 separate times to accumulate data from 4 time periods: May to June 2020 (T1); December 2020 to February 2021 (T2); July to August 2021 (T3); and June to July 2022 (T4). These periods correspond to the following phases of the pandemic: T1, onset of pandemic; T2, vaccine availability; T3, Delta variant predominant; T4, Omicron variant predominant.

Data on the spread of COVID-19 were collected from the CDC archived dataset, US COVID-19 County Level of Community Transmission Historical Changes (Table 1).7 Administrative workload (patient counts) for EEG, EMU, and outpatient clinics were extracted from VHA administrative databases for the participating sites for the months prior to each survey: T1, April 2020; T2, November 2020; T3, June 2021; and T4, May 2022 (Table 2).

Survey Structure and Content
The survey was developed by the ECoE and was not validated prior to its use due to the time-sensitive nature of gathering information during the pandemic. The first survey (T1) was an emailed spreadsheet with open-ended questions to gauge availability of services (ie, outpatient clinic, EEG, EMU), assess whether safety precautions were being introduced, and understand whether national or local guidelines were thought to be helpful. Responses from this and subsequent surveys were standardized into yes/no and multiple choice formats. Subsequent surveys were administered online using a Research Electronic Data Capture tool.8,9
Availability of outpatient epilepsy services across the 4 time periods were categorized as unlimited (in-person with no restrictions), limited (in-person with restrictions), planned (not currently performed but scheduled for the near future), and unavailable (no in-person services offered) (eAppendices 1-6, available in article PDF).
Statistical Analyses
Analyses were performed to compare survey responses to workload and CDC data on COVID-19 community spread. The following associations were examined: (1) CDC COVID-19 spread vs respondents’ perception of spread; (2) respondents’ perception of spread vs availability of services; (3) CDC COVID-19 spread vs availability of services; (4) respondents’ perception of spread vs workload; and (5) CDC COVID-19 spread vs workload. Availability of services was dichotomized for analyses, with limited or fully available services classified as available. As services were mostly open at T3 regardless of the spread of the virus, and the CDC COVID-19 spread classification for all sites was severe or high at T2 and T4, corresponding associations were not tested at these time points. For associations 1 through 3, Fisher exact tests were used; for associations 4 and 5, Mann-Whitney U tests (where the COVID-19 spread fell into 2 categories) and Kruskal-Wallis tests (for 3 categories of COVID-19 spread) were performed. All tests were 2-tailed and performed at 0.05 error rate. Bonferroni corrections were applied to adjust P values for multiple hypotheses tests.
RESULTS
From the 17 sites invited, responses at each time point were obtained from 13 (T1),17 (T2), 15 (T3), and 16 (T4) centers. There was no significant association between self-reported COVID-19 spread and CDC classification of COVID spread. There were no associations between COVID-19 community spread (respondent reported or CDC severity level) and outpatient clinic availability (self-reported or workload captured). At T3, a positive association was found between the CDC spread level and workload (P = .008), but this was not significant after Bonferroni correction (P = .06).
EEG availability surpassed EMU availability at all time points, although EMU services made some recovery at T3 and T4. No associations were found between COVID-19 community spread (self-reported or CDC severity level) and outpatient EEG or EMU availability (self-reported or workload captured). At T3, there was a positive association between EEG workload and CDC COVID-19 severity level (P = .04), but this was not significant after Bonferroni correction (P = .30).
For outpatient EEG, staff and patient mask use were universally implemented by T2, while the use of full personal protective equipment (PPE) occurred at a subset of sites (T2, 6/17 [35%]; T3, 3/15 [20%]; T4: 4/16 [25%]). COVID-19 testing was rarely implemented prior to outpatient EEG (T1, 0 sites; T2, 1 site; T3, 1 site; T4, 0 sites). Within the EMU, safety precautions including COVID-19 testing, patient mask usage, staff mask usage, and aerosolization demonstrated a sustained majority usage across the 4 surveys.
National and Local Guidelines
The open-ended survey at T1 asked site directors, “Should there be national recommendations on how EEGs and related procedures should be done during the pandemic or should this be left to local conditions?” Responses were mixed, with 5 respondents desiring a national standard, 4 respondents favoring a local response, and 4 respondents believing a national standard should be in place but with modifications based on local outbreak levels and needs.
Surveys performed at T2 through T4 asked, “Which of the following do you feel was/will be helpful in adapting to COVID-19–related changes?” Overall, there was substantial agreement that guidelines were helpful. Most sites anticipated permanent changes in enhanced safety precautions and telehealth.
DISCUSSION
This longitudinal study across 4 time points describes how epilepsy services within the VHA and ECoE adapted to the COVID-19 pandemic. The first survey, conducted 2 months after COVID-19 was declared a pandemic, allowed a comparison with other concurrent US national surveys.1,2,10 The subsequent surveys describe longitudinal adaptations to balance patient and staff safety with service availability and is a unique feature of the current report. Results demonstrate flexibility and adaptability by the ECoEs surveyed, which surprisingly did not show significant associations between CDC COVID-19 spread data and administrative workload data.
Trends in Availability of Services
The most significant impact of COVID-19 restrictions was during T1. There were no significant relationships between service availability/workload and objective CDC COVID-19 spread levels or subjective self-reported COVID-19 spread. Respondents’ perceptions of local COVID-19 spread showed no association with CDC COVID-19 spread data. It appears that subjective perception of spread may be unreliable and factors other than actual or perceived COVID-19 spread were likely driving patterns for service availability.
In-person outpatient visits were most impacted at T1, similar to other civilian surveys, with only 1 site reporting in-person outpatient visits without limitations.1,2 These numbers significantly changed by T2, with all sites offering either limited or unlimited in-person visits. While the surveys did not evaluate factors leading to this rapid recovery, it may be related to the availability of COVID-19 vaccinations within the VHA during this time.11 The US Department of Veterans Affairs was the first federal agency to mandate employee vaccination.12 By the most recent time point (T4), all responding sites offered outpatient visits. Outpatient EEGs followed a similar trend, with T1 being the most restrictive and full, unrestricted outpatient EEGs available by T3.
Fiscal year (FY) trends from ECoE annual reports suggest that encounters slowly recovered over the course of the pandemic. In FY 2019 there were 13,143 outpatient encounters and 6394 EEGs, which dropped to 8097 outpatient encounters and 4432 EEGs in FY 2020 before rising to 8489 outpatient encounters and 5604 EEGs in FY 2021 and 9772 outpatient encounters and 5062 EEGs in FY 2022. Thus, while clinicians described availability of services, patients may have remained hesitant or were otherwise unable to fulfill in-person appointments. The increased availability of home EEG (145 encounter days in 2021 and 436 encounter days in 2022) may be filling this gap.
In contrast to outpatient clinics and EEG, EMU availability showed relatively slower reimplementation. In the last survey, about 30% of sites were still not offering EMU or had limited services. Early trends regarding reduced staffing and patient reluctance for elective admission cited in other surveys may have also affected EMU availability within the VHA.2,13 Consistent with trends in availability, ECoE annual report data suggest EMU patient participation was about one-half of prepandemic rates: 3069 encounters in FY 2019 dropped to 1614 encounters in 2020. By 2021, rates were about two-thirds of prepandemic rates with 2058 encounters in 2021 and 2101 encounters in 2022.
Early survey results (T1) from this study echo trends from other surveys. In the AES survey (April to June 2020), about a quarter of respondents (22%) reported doing fewer EEG studies than usual. The Italian national survey (April 2020) revealed reduced presurgical evaluations (81%), ambulatory EEG (78%), standard EEG (5%) and long-term EEG (32%).4 In the NAEC survey (end of 2020)—which roughly corresponded to T2—outpatient EEGs were still < 75% of pre-COVID levels in one-half of the centers.
National and Local Guidelines
Both national and local guidelines were perceived as useful by most respondents, with national guidelines being more beneficial. This aligns with the NAEC survey, where there was a perceived need for detailed recommendations for PPE and COVID-19 testing of patients, visitors, and staff. Based on national and local guidelines, ECoE implemented safety procedures, as reflected in responses. Staff masking procedures appeared to be the most widely adopted for all services, while the use of full PPE waned as the pandemic progressed. COVID-19 testing was rarely used for routine outpatient visits but common in EMU admissions. This is similar to a survey conducted by the American Academy of Neurology which found full PPE implementation intermittently in outpatient settings and more frequently in inpatient settings.14
Telehealth Attitudes
While most sites anticipated permanent implementation of safety precautions and telehealth, the latter was consistently reported as more likely to be sustained. The VHA had a large and well-developed system of telehealth services that considerably predated the pandemic.15,16 Through this established infrastructure, remote services were quickly increased across the VHA.17-19 This telehealth structure was supplemented by the ability of VHA clinicians to practice across state lines, following a 2018 federal rule.20 The AES survey noted the VHA ECoE's longstanding experience with telehealth as a model for telemedicine use in providing direct patient care, remote EEG analysis, and clinician-to-clinician consultation.1
Trends in the number of telehealth patients seen, observed through patterns in ECoE annual reports are consistent with positive views toward this method of service provision. Specifically, these annual reports capture trends in Video Telehealth Clinic (local station), Video Telehealth Clinic (different station), Home Video Telehealth, Telephone Clinic, and eConsults. Though video telehealth at in-person stations had a precipitous drop in 2020 that continued to wane in subsequent years (898 encounters in 2019; 455 encounters in 2020; 90 encounters in 2021; 88 encounters in 2022), use of home video telehealth rose over time (143 encounters in 2019; 1003 encounters in 2020; 3206 encounters in 2021; 3315 encounters in 2022). Use of telephone services rose drastically in 2020 but has since become a less frequently used service method (2636 in 2019; 5923 in 2020; 5319 in 2021; 3704 in 2022).
Limitations
While the survey encouraged a high response rate, this limited its scope and interpretability. While the availability of services was evaluated, the underlying reasons were not queried. Follow-up questions about barriers to reopening may have allowed for a better understanding of why some services, such as EMU, continued to operate suboptimally later in the pandemic. Similarly, asking about unique strategies or barriers for telehealth would have allowed for a better understanding of its current and future use. We hypothesize that staffing changes during the pandemic may have influenced the availability of services, but changes to staffing were not assessed via the survey and were not readily available via other sources (eg, ECoE annual reports) at the time of publication. An additional limitation is the lack of comparable surveys in the literature for time points T2 to T4, as most analogous surveys were performed early in 2020.
Conclusions
This longitudinal study performed at 4 time points during the COVID-19 pandemic is the first to offer a comprehensive picture of changes to epilepsy and EEG services over time, given that other similar surveys lacked follow-up. Results reveal a significant limitation of services at VHA ECoE shortly after the onset of the pandemic, with return to near-complete operational status 2 years later. While safety precautions and telehealth are predicted to continue, telehealth is perceived as a more permanent change in services.
Albert DVF, Das RR, Acharya JN, et al. The impact of COVID-19 on epilepsy care: a survey of the American Epilepsy Society membership. Epilepsy Curr. 2020;20(5):316-324. doi:10.1177/1535759720956994
Ahrens SM, Ostendorf AP, Lado FA, et al. Impact of the COVID-19 pandemic on epilepsy center practice in the United States. Neurology. 2022;98(19):e1893-e1901. doi:10.1212/WNL.0000000000200285
Cross JH, Kwon CS, Asadi-Pooya AA, et al. Epilepsy care during the COVID-19 pandemic. Epilepsia. 2021;62(10):2322-2332. doi:10.1111/epi.17045
Assenza G, Lanzone J, Ricci L, et al. Electroencephalography at the time of Covid-19 pandemic in Italy. Neurol Sci. 2020;41(8):1999-2004. doi:10.1007/s10072-020-04546-8
US Department of Veterans Affairs. National Center for Veterans Analysis and Statistics. Veteran population. Updated September 7, 2022. Accessed October 25, 2024. https://www.va.gov/vetdata/veteran_population.asp
US Department of Veterans Affairs, Veterans Health Administration. Epilepsy Centers of Excellence (ECoE). Annual report fiscal year 2019. Accessed October 25, 2024. https://www.epilepsy.va.gov/docs/FY19AnnualReport-VHAEpilepsyCentersofExcellence.pdf
Centers for Disease Control and Prevention. United States COVID-19 county level of community transmission historical changes – ARCHIVED. Updated February 20, 2024. Accessed October 25, 2024. https://data.cdc.gov/Public-Health-Surveillance/United-States-COVID-19-County-Level-of-Community-T/nra9-vzzn
Harris PA, Taylor R, Thielke R, Payne J, Gonzalez N, Conde JG. Research electronic data capture (REDCap)—a metadata-driven methodology and workflow process for providing translational research informatics support. J Biomed Inform. 2009;42(2):377-381. doi:10.1016/j.jbi.2008.08.010
Harris PA, Taylor R, Minor BL, et al. The REDCap consortium: building an international community of software platform partners. J Biomed Inform. 2019;95:103208. doi:10.1016/j.jbi.2019.103208
World Health Organization. Rolling updates on coronavirus disease (COVID-19). Updated July 31, 2020. Accessed October 25, 2024. https://www.who.int/emergencies/diseases/novel-coronavirus-2019/events-as-they-happen
US Department of Veterans Affairs. VA announces initial plans for COVID-19 vaccine distribution. News release. December 10, 2020. Accessed October 25, 2024. https://www.va.gov/opa/pressrel/pressrelease.cfm?id=5580
Steinhauer J. V.A. Issues Vaccine Mandate for Health Care Workers, a First for a Federal Agency. The New York Times. August 16, 2021. Accessed October 25, 2024. https://www.nytimes.com/2021/07/26/us/politics/veterans-affairs-coronavirus-covid-19.html
Zafar SF, Khozein RJ, LaRoche SM, Westover MB, Gilmore EJ. Impact of the COVID-19 pandemic on continuous EEG utilization. J Clin Neurophysiol. 2022;39(7):567-574. doi:10.1097/WNP.0000000000000802
Qureshi AI, Rheaume C, Huang W, et al. COVID-19 exposure during neurology practice. Neurologist. 2021;26(6):225-230. doi:10.1097/NRL.0000000000000346
Darkins A, Cruise C, Armstrong M, Peters J, Finn M. Enhancing access of combat-wounded veterans to specialist rehabilitation services: the VA Polytrauma Telehealth Network. Arch Phys Med Rehabil. 2008;89(1):182-187. doi:10.1016/j.apmr.2007.07.027
Darkins A, Ryan P, Kobb R, et al. Care coordination/home telehealth: the systematic implementation of health informatics, home telehealth, and disease management to support the care of veteran patients with chronic conditions. Telemed J E Health. 2008;14(10):1118-1126. doi:10.1089/tmj.2008.0021
Gentry MT, Puspitasari AJ, McKean AJ, et al. Clinician satisfaction with rapid adoption and implementation of telehealth services during the COVID-19 pandemic. Telemed J E Health. 2021;27(12):1385-1392. doi:10.1089/tmj.2020.0575
Connolly SL, Stolzmann KL, Heyworth L, et al. Patient and provider predictors of telemental health use prior to and during the COVID-19 pandemic within the Department of Veterans Affairs. Am Psychol. 2022;77(2):249-261. doi:10.1037/amp0000895
Shelton CJ, Kim A, Hassan AM, Bhat A, Barnello J, Castro CA. System-wide implementation of telehealth to support military veterans and their families in response to COVID-19: a paradigm shift. J Mil Veteran Fam Health. 2020;6(S2):50-57. doi:10.3138/jmvfh-CO19-0003
VA expands telehealth by allowing health care providers to treat patients across state lines. News release. US Dept of Veterans Affairs. May 11, 2018. Accessed October 25, 2024. https://news.va.gov/press-room/va-expands-telehealth-by-allowing-health-care-providers-to-treat-patients-across-state-lines/
Albert DVF, Das RR, Acharya JN, et al. The impact of COVID-19 on epilepsy care: a survey of the American Epilepsy Society membership. Epilepsy Curr. 2020;20(5):316-324. doi:10.1177/1535759720956994
Ahrens SM, Ostendorf AP, Lado FA, et al. Impact of the COVID-19 pandemic on epilepsy center practice in the United States. Neurology. 2022;98(19):e1893-e1901. doi:10.1212/WNL.0000000000200285
Cross JH, Kwon CS, Asadi-Pooya AA, et al. Epilepsy care during the COVID-19 pandemic. Epilepsia. 2021;62(10):2322-2332. doi:10.1111/epi.17045
Assenza G, Lanzone J, Ricci L, et al. Electroencephalography at the time of Covid-19 pandemic in Italy. Neurol Sci. 2020;41(8):1999-2004. doi:10.1007/s10072-020-04546-8
US Department of Veterans Affairs. National Center for Veterans Analysis and Statistics. Veteran population. Updated September 7, 2022. Accessed October 25, 2024. https://www.va.gov/vetdata/veteran_population.asp
US Department of Veterans Affairs, Veterans Health Administration. Epilepsy Centers of Excellence (ECoE). Annual report fiscal year 2019. Accessed October 25, 2024. https://www.epilepsy.va.gov/docs/FY19AnnualReport-VHAEpilepsyCentersofExcellence.pdf
Centers for Disease Control and Prevention. United States COVID-19 county level of community transmission historical changes – ARCHIVED. Updated February 20, 2024. Accessed October 25, 2024. https://data.cdc.gov/Public-Health-Surveillance/United-States-COVID-19-County-Level-of-Community-T/nra9-vzzn
Harris PA, Taylor R, Thielke R, Payne J, Gonzalez N, Conde JG. Research electronic data capture (REDCap)—a metadata-driven methodology and workflow process for providing translational research informatics support. J Biomed Inform. 2009;42(2):377-381. doi:10.1016/j.jbi.2008.08.010
Harris PA, Taylor R, Minor BL, et al. The REDCap consortium: building an international community of software platform partners. J Biomed Inform. 2019;95:103208. doi:10.1016/j.jbi.2019.103208
World Health Organization. Rolling updates on coronavirus disease (COVID-19). Updated July 31, 2020. Accessed October 25, 2024. https://www.who.int/emergencies/diseases/novel-coronavirus-2019/events-as-they-happen
US Department of Veterans Affairs. VA announces initial plans for COVID-19 vaccine distribution. News release. December 10, 2020. Accessed October 25, 2024. https://www.va.gov/opa/pressrel/pressrelease.cfm?id=5580
Steinhauer J. V.A. Issues Vaccine Mandate for Health Care Workers, a First for a Federal Agency. The New York Times. August 16, 2021. Accessed October 25, 2024. https://www.nytimes.com/2021/07/26/us/politics/veterans-affairs-coronavirus-covid-19.html
Zafar SF, Khozein RJ, LaRoche SM, Westover MB, Gilmore EJ. Impact of the COVID-19 pandemic on continuous EEG utilization. J Clin Neurophysiol. 2022;39(7):567-574. doi:10.1097/WNP.0000000000000802
Qureshi AI, Rheaume C, Huang W, et al. COVID-19 exposure during neurology practice. Neurologist. 2021;26(6):225-230. doi:10.1097/NRL.0000000000000346
Darkins A, Cruise C, Armstrong M, Peters J, Finn M. Enhancing access of combat-wounded veterans to specialist rehabilitation services: the VA Polytrauma Telehealth Network. Arch Phys Med Rehabil. 2008;89(1):182-187. doi:10.1016/j.apmr.2007.07.027
Darkins A, Ryan P, Kobb R, et al. Care coordination/home telehealth: the systematic implementation of health informatics, home telehealth, and disease management to support the care of veteran patients with chronic conditions. Telemed J E Health. 2008;14(10):1118-1126. doi:10.1089/tmj.2008.0021
Gentry MT, Puspitasari AJ, McKean AJ, et al. Clinician satisfaction with rapid adoption and implementation of telehealth services during the COVID-19 pandemic. Telemed J E Health. 2021;27(12):1385-1392. doi:10.1089/tmj.2020.0575
Connolly SL, Stolzmann KL, Heyworth L, et al. Patient and provider predictors of telemental health use prior to and during the COVID-19 pandemic within the Department of Veterans Affairs. Am Psychol. 2022;77(2):249-261. doi:10.1037/amp0000895
Shelton CJ, Kim A, Hassan AM, Bhat A, Barnello J, Castro CA. System-wide implementation of telehealth to support military veterans and their families in response to COVID-19: a paradigm shift. J Mil Veteran Fam Health. 2020;6(S2):50-57. doi:10.3138/jmvfh-CO19-0003
VA expands telehealth by allowing health care providers to treat patients across state lines. News release. US Dept of Veterans Affairs. May 11, 2018. Accessed October 25, 2024. https://news.va.gov/press-room/va-expands-telehealth-by-allowing-health-care-providers-to-treat-patients-across-state-lines/
Comparing Patient Care Models at a Local Free Clinic vs an Insurance- Based University Medical Center
Comparing Patient Care Models at a Local Free Clinic vs an Insurance- Based University Medical Center
Approximately 25% of Americans have at least one skin condition, and 20% are estimated to develop skin cancer during their lifetime.1,2 However, 40% of the US population lives in areas underserved by dermatologists. 3 The severity and mortality of skin cancers such as melanoma and mycosis fungoides have been positively associated with minoritized race, lack of health insurance, and unstable housing status.4-6 Patients who receive health care at free clinics often are of a racial or ethnic minoritized social group, are uninsured, and/or lack stable housing; this underserved group also includes recent immigrants to the United States who have limited English proficiency (LEP).7 Only 25% of free clinics offer specialty care services such as dermatology.7,8
Of the 42 free clinics and Federally Qualified Health Centers in Pittsburgh, Pennsylvania, the Birmingham Free Clinic (BFC) is one of the few that offers specialty care services including dermatology.9 Founded in 1994, the BFC serves as a safety net for Pittsburgh’s medically underserved population, offering primary and acute care, medication access, and social services. From January 2020 to May 2022, the BFC offered 27 dermatology clinics that provided approximately 100 people with comprehensive care including full-body skin examinations, dermatologic diagnoses and treatments, minor procedures, and dermatopathology services.
In this study, we compared the BFC dermatology patient care model with that of the dermatology department at the University of Pittsburgh Medical Center (UPMC), an insurance-based tertiary referral health care system in western Pennsylvania. By analyzing the demographics, dermatologic diagnoses, and management strategies of both the BFC and UPMC, we gained an understanding of how these patient care models differ and how they can be improved to care for diverse patient populations.
Methods
A retrospective chart review of dermatology patients seen in person at the BFC and UPMC during the period from January 2020 to May 2022 was performed. The UPMC group included patients seen by 3 general dermatologists (including A.J.J.) at matched time points. Data were collected from patients’ first in-person visit during the study period. Variables of interest included patient age, sex, race, ethnicity, primary language, zip code, health insurance status, distance to clinic (estimated using Google Maps to calculate the shortest driving distance from the patient’s zip code to the clinic), history of skin cancer, dermatologic diagnoses, and management strategies. These variables were not collected for patients who cancelled or noshowed their first in-person appointments. All patient charts and notes corresponding to the date and visit of interest were accessed through the electronic medical record (EMR). Patient data were de-identified and stored in a password-protected spreadsheet. Comparisons between the BFC and UPMC patient populations were performed using X2 tests of independence, Fisher exact tests, and Mann-Whitney U tests via SPSS software (IBM). Statistical significance was set at P<.05.
Results
Patient Characteristics—Our analysis included 76 initial appointments at the BFC and 322 at UPMC (Table 1). The mean age for patients at the BFC and UPMC was 39.6 years and 47.8 years, respectively (P=.001). Males accounted for 39 (51.3%) and 112 (34.8%) of BFC and UPMC patients, respectively (P=.008); 2 (0.6%) patients from UPMC were transgender. Of the BFC and UPMC patients, 44.7% (34/76) and 0.9% (3/322) were Hispanic, respectively (P<.001). With regard to race, 52.6% (40/76) of BFC patients were White, 19.7% (15/76) were Black, 6.6% (5/76) were Asian/Pacific Islander (Chinese, 1.3% [1/76]; other Asian, 5.3% [4/76]), and 21.1% (16/76) were American Indian/other/unspecified (American Indian, 1.3% [1/76]; other, 13.2% [10/76]; unspecified, 6.6% [5/76]). At UPMC, 61.2% (197/322) of patients were White, 28.0% (90/322) were Black, 5.3% (17/322) were Asian/Pacific Islander (Chinese, 1.2% [4/322]; Indian [Asian], 1.9% [6/322]; Japanese, 0.3% [1/322]; other Asian, 1.6% [5/322]; other Asian/American Indian, 0.3% [1/322]), and 5.6% (18/322) were American Indian/other/ unspecified (American Indian, 0.3% [1/322]; other, 0.3% [1/322]; unspecified, 5.0% [16/322]). Overall, the BFC patient population was more ethnically and racially diverse than that of UPMC (P<.001).
Forty-six percent (35/76) of BFC patients and 4.3% (14/322) of UPMC patients had LEP (P<.001). Primary languages among BFC patients were 53.9% (41/76) English, 40.8% (31/76) Spanish, and 5.2% (4/76) other/ unspecified (Chinese, 1.3% [1/76]; Indonesian, 2.6% [2/76]; unspecified, 1.3% [1/76]). Primary languages among UPMC patients were 95.7% (308/322) English and 4.3% (14/322) other/unspecified (Chinese, 0.6% [2/322]; Nepali, 0.6% [2/322]; Pali, 0.3% [1/322]; Russian, 0.3% [1/322]; unspecified, 2.5% [8/322]). There were notable differences in insurance status at the BFC vs UPMC (P<.001), with more UPMC patients having private insurance (52.8% [170/322] vs 11.8% [9/76]) and more BFC patients being uninsured (52.8% [51/76] vs 1.9% [6/322]). There was no significant difference in distance to clinic between the 2 groups (P=.183). More UPMC patients had a history of skin cancer (P=.003). More patients at the BFC were no-shows for their appointments (P<.001), and UPMC patients more frequently canceled their appointments (P<.001).
Dermatologic Diagnoses—The most commonly diagnosed dermatologic conditions at the BFC were dermatitis (23.7% [18/76]), neoplasm of uncertain behavior (15.8% [12/76]), alopecia (11.8% [9/76]), and acne (10.5% [8/76]) (Table 2). The most commonly diagnosed conditions at UPMC were nevi (26.4% [85/322]), dermatitis (22.7% [73/322]), seborrheic keratosis (21.7% [70/322]), and skin cancer screening (21.4% [70/322]). Neoplasm of uncertain behavior was more common in BFC vs UPMC patients (P=.040), while UPMC patients were more frequently diagnosed with nevi (P<.001), seborrheic keratosis (P<.001), and skin cancer screening (P<.001). There was no significant difference between the incidence of skin cancer diagnoses in the BFC (1.3% [1/76]) and UPMC (0.6% [2/76]) patient populations (P=.471). Among the biopsied neoplasms, there was also no significant difference in malignant (BFC, 50.0% [5/10]; UPMC, 32.0% [8/25]) and benign (BFC, 50.0% [5/10]; UPMC, 36.0% [9/25]) neoplasms diagnosed at each clinic (P=.444).
Management Strategies—Systemic antibiotics were more frequently prescribed (P<.001) and laboratory testing/ imaging were more frequently ordered (P=.005) at the BFC vs UPMC (Table 3). Patients at the BFC also more frequently required emergency insurance (P=.036). Patients at UPMC were more frequently recommended sunscreen (P=.003) and received education about skin cancer signs by review of the ABCDEs of melanoma (P<.001), sun-protective behaviors (P=.001), and skin examination frequency (P<.001). Notes in the EMR for UPMC patients more frequently specified patient followup instructions (P<.001).
Comment
As of 2020, the city of Pittsburgh had an estimated population of nearly 303,000 based on US Census data.10 Its population is predominantly White (62.7%) followed by Black/African American (22.8%) and Asian (6.5%); 5.9% identify as 2 or more races. Approximately 3.8% identify as Hispanic or Latino. More than 11% of the Pittsburgh population aged 5 years and older speaks a language other than English as their primary language, including Spanish (2.3%), other Indo-European languages (3.9%), and Asian and Pacific Island languages (3.5%).11 More than 5% of the Pittsburgh population does not have health insurance.12
The BFC is located in Pittsburgh’s South Side area, while one of UPMC’s primary dermatology clinics is located in the Oakland district; however, most patients who seek care at these clinics live outside these areas. Our study results indicated that the BFC and UPMC serve distinct groups of people within the Pittsburgh population. The BFC patient population was younger with a higher percentage of patients who were male, Hispanic, racially diverse, and with LEP compared with the UPMC patient population. In this clinical setting, the BFC health care team engages with people from diverse backgrounds and requires greater interpreter and medical support services.
The BFC largely is supported by volunteers, UPMC, grants, and philanthropy. Dermatology clinics are staffed by paid and volunteer team members. Paid team members include 1 nurse and 1 access lead who operates the front desk and registration. Volunteer team members include 1 board-certified dermatologist from UPMC (A.J.J.), or an affiliate clinic and 1 or 2 of each of the following: UPMC dermatology residents, medical or undergraduate students from the University of Pittsburgh, AmeriCorps national service members, and student or community medical interpreters. The onsite pharmacy is run by volunteer faculty, resident, and student pharmacists from the University of Pittsburgh. Dermatology clinics are half-day clinics that occur monthly. Volunteers for each clinic are recruited approximately 1 month in advance.
Dermatology patients at the BFC are referred from the BFC general medicine clinic and nearby Federally Qualified Health Center s for simple to complex medical and surgical dermatologic skin conditions. Each BFC dermatology clinic schedules an average of 7 patients per clinic and places other patients on a wait-list unless more urgent triage is needed. Patients are notified when they are scheduled via phone or text message, and they receive a reminder call or text 1 or 2 days prior to their appointment that also asks them to confirm attendance. Patients with LEP are called with an interpreter and also may receive text reminders that can be translated using Google Translate. Patients are instructed to notify the BFC if they need to cancel or reschedule their appointment. At the end of each visit, patients are given an after-visit summary that lists follow-up instructions, medications prescribed during the visit, and upcoming appointments. The BFC offers bus tickets to help patients get to their appointments. In rare cases, the BFC may pay for a car service to drive patients to and from the clinic.
Dermatology clinics at UPMC use scheduling and self-scheduling systems through which patients can make appointments at a location of their choice with any available board-certified dermatologist or physician assistant. Patients receive a reminder phone call 3 days prior to their appointment instructing them to call the office if they are unable to keep their appointment. Patients signed up for the online portal also receive a reminder message and an option to confirm or cancel their appointment. Patients with cell phone numbers in the UPMC system receive a text message approximately 2 days prior to their appointment that allows them to preregister and pay their copayment in advance. They receive another text 20 minutes prior to their appointment with an option for contactless check-in. At the conclusion of their visit, patients can schedule a follow-up appointment and receive a printed copy of their after-visit summary that provides information about follow-up instructions, prescribed medications, and upcoming visits. They may alternatively access this summary via the online patient portal. Patients are not provided transportation to UPMC clinics, but they are offered parking validation.
Among the most common dermatologic diagnoses for each group, BFC patients presented for treatment of more acute dermatologic conditions, while UPMC patients presented for more benign and preventive-care conditions. This difference may be attributable to the BFC’s referral and triage system, wherein patients with more urgent problems are given scheduling priority. This patient care model contrasts with UPMC’s scheduling process in which no known formal triage system is utilized. Interestingly, there was no difference in skin cancer incidence despite a higher percentage of preventive skin cancer screenings at UPMC.
Patients at the BFC more often required emergency insurance for surgical interventions, which is consistent with the higher percentage of uninsured individuals in this population. Patients at UPMC more frequently were recommended sunscreen and were educated about skin cancer, sun protection, and skin examination, in part due to this group’s more extensive history of skin cancer and frequent presentation for skin cancer screenings. At the same time, educational materials for skin care at both the BFC and UPMC are populated into the EMR in English, whereas materials in other languages are less readily available.
Our retrospective study had several limitations. Demographic information that relied on clinic-dependent intake questionnaires may be limited due to variable intake processes and patients opting out of self-reporting. By comparing patient populations between 2 clinics, confounding variables such as location and hours of operation may impact the patient demographics recorded at the BFC vs UPMC. Resources and staff availability may affect the management strategies and follow-up care offered by each clinic. Our study period also was unique in that COVID-19 may have affected resources, staffing, scheduling, and logistics at both clinics.
Based on the aforementioned differences between the BFC and UPMC patient characteristics, care models should be strategically designed to support the needs of diverse populations. The BFC patient care model appropriately focuses on communication skills with patients with LEP by using interpreter services. Providing more skin care education and follow-up instructions in patients’ primary languages will help them develop a better understanding of their skin conditions. Another key asset of the BFC patient care model is its provision of social services such as transportation and insurance assistance.
To improve the UPMC patient care model, providing patients with bus tickets and car services may potentially reduce appointment cancellations. Using interpreter services to call and text appointment reminders, as well as interpreter resources to facilitate patient visits and patient instructions, also can mitigate language barriers for patients with LEP. Implementing a triage system into the UPMC scheduling system may help patients with more urgent skin conditions to be seen in a timely manner.
Other investigators have analyzed costs of care and proven the value of dermatologic services at free clinics to guide allocation of supplies and resources, demonstrating an area for future investigation at the BFC.13 A cost analysis of care provided at the BFC compared to UPMC could inform us about the value of the BFC’s services.
Conclusion
The dermatology clinics at the BFC and UPMC have distinct demographics, diagnoses, and management strategies to provide an inclusive patient care model. The services provided by both clinics are necessary to ensure that people in Pittsburgh have access to dermatologic care regardless of social barriers (eg, lack of health insurance, LEP). To achieve greater accessibility and health equity, dermatologic care at the BFC and UPMC can be improved by strengthening communication with people with LEP, providing skin care education, and offering social and scheduling services.
- Lim HW, Collins SAB, Resneck JS, et al. The burden of skin disease in the United States. J Am Acad Dermatol. 2017;76:958-972.e2. doi:10.1016/j.jaad.2016.12.043
- American Academy of Dermatology. Skin cancer. Accessed October 7, 2024. https://www.aad.org/media/stats-skin-cancer
- Suneja T, Smith ED, Chen GJ, et al. Waiting times to see a dermatologist are perceived as too long by dermatologists: implications for the dermatology workforce. Arch Dermatol. 2001;137:1303-1307. doi:10.1001/archderm.137.10.1303
- Grossberg AL, Carranza D, Lamp K, et al. Dermatologic care in the homeless and underserved populations: observations from the Venice Family Clinic. Cutis. 2012;89:25-32.
- Amini A, Rusthoven CG, Waxweiler TV, et al. Association of health insurance with outcomes in adults ages 18 to 64 years with melanoma in the United States. J Am Acad Dermatol. 2016;74:309-316. doi:10.1016/j.jaad.2015.09.054
- Su C, Nguyen KA, Bai HX, et al. Racial disparity in mycosis fungoides: an analysis of 4495 cases from the US National Cancer Database. J Am Acad Dermatol. 2017;77:497-502.e2. doi:10.1016/j.jaad .2017.04.1137
- Darnell JS. Free clinics in the United States: a nationwide survey. Arch Intern Med. 2010;170:946-953. doi:10.1001/archinternmed .2010.107
- Madray V, Ginjupalli S, Hashmi O, et al. Access to dermatology services at free medical clinics: a nationwide cross-sectional survey. J Am Acad Dermatol. 2019;81:245-246. doi:10.1016/j.jaad.2018.12.011
- Pennsylvania free and income-based clinics. Accessed October 7, 2024. https://www.freeclinics.com/sta/pennsylvania
- United States Census Bureau. Decennial census. P1: race. Accessed October 7, 2024. https://data.census.gov/table/DECENNIALPL2020.P1?g=160XX00US4261000
- United States Census Bureau. American community survey. S1601: language spoken at home. Accessed October 7, 2024. https://data.census.gov/table/ACSST5Y2020S1601?g=160XX00US4261000
- United States Census Bureau. S2701: selected characteristics of health insurance coverage in the United States. Accessed October 7, 2024. https://data.census.gov/table/ACSST5Y2020.S2701?g=160XX00US4261000
- Lin CP, Loy S, Boothe WD, et al. Value of Dermatology Nights at a student-run free clinic. Proc (Bayl Univ Med Cent). 2020;34:260-261. doi:10.1080/08998280.2020.1834771
Approximately 25% of Americans have at least one skin condition, and 20% are estimated to develop skin cancer during their lifetime.1,2 However, 40% of the US population lives in areas underserved by dermatologists. 3 The severity and mortality of skin cancers such as melanoma and mycosis fungoides have been positively associated with minoritized race, lack of health insurance, and unstable housing status.4-6 Patients who receive health care at free clinics often are of a racial or ethnic minoritized social group, are uninsured, and/or lack stable housing; this underserved group also includes recent immigrants to the United States who have limited English proficiency (LEP).7 Only 25% of free clinics offer specialty care services such as dermatology.7,8
Of the 42 free clinics and Federally Qualified Health Centers in Pittsburgh, Pennsylvania, the Birmingham Free Clinic (BFC) is one of the few that offers specialty care services including dermatology.9 Founded in 1994, the BFC serves as a safety net for Pittsburgh’s medically underserved population, offering primary and acute care, medication access, and social services. From January 2020 to May 2022, the BFC offered 27 dermatology clinics that provided approximately 100 people with comprehensive care including full-body skin examinations, dermatologic diagnoses and treatments, minor procedures, and dermatopathology services.
In this study, we compared the BFC dermatology patient care model with that of the dermatology department at the University of Pittsburgh Medical Center (UPMC), an insurance-based tertiary referral health care system in western Pennsylvania. By analyzing the demographics, dermatologic diagnoses, and management strategies of both the BFC and UPMC, we gained an understanding of how these patient care models differ and how they can be improved to care for diverse patient populations.
Methods
A retrospective chart review of dermatology patients seen in person at the BFC and UPMC during the period from January 2020 to May 2022 was performed. The UPMC group included patients seen by 3 general dermatologists (including A.J.J.) at matched time points. Data were collected from patients’ first in-person visit during the study period. Variables of interest included patient age, sex, race, ethnicity, primary language, zip code, health insurance status, distance to clinic (estimated using Google Maps to calculate the shortest driving distance from the patient’s zip code to the clinic), history of skin cancer, dermatologic diagnoses, and management strategies. These variables were not collected for patients who cancelled or noshowed their first in-person appointments. All patient charts and notes corresponding to the date and visit of interest were accessed through the electronic medical record (EMR). Patient data were de-identified and stored in a password-protected spreadsheet. Comparisons between the BFC and UPMC patient populations were performed using X2 tests of independence, Fisher exact tests, and Mann-Whitney U tests via SPSS software (IBM). Statistical significance was set at P<.05.
Results
Patient Characteristics—Our analysis included 76 initial appointments at the BFC and 322 at UPMC (Table 1). The mean age for patients at the BFC and UPMC was 39.6 years and 47.8 years, respectively (P=.001). Males accounted for 39 (51.3%) and 112 (34.8%) of BFC and UPMC patients, respectively (P=.008); 2 (0.6%) patients from UPMC were transgender. Of the BFC and UPMC patients, 44.7% (34/76) and 0.9% (3/322) were Hispanic, respectively (P<.001). With regard to race, 52.6% (40/76) of BFC patients were White, 19.7% (15/76) were Black, 6.6% (5/76) were Asian/Pacific Islander (Chinese, 1.3% [1/76]; other Asian, 5.3% [4/76]), and 21.1% (16/76) were American Indian/other/unspecified (American Indian, 1.3% [1/76]; other, 13.2% [10/76]; unspecified, 6.6% [5/76]). At UPMC, 61.2% (197/322) of patients were White, 28.0% (90/322) were Black, 5.3% (17/322) were Asian/Pacific Islander (Chinese, 1.2% [4/322]; Indian [Asian], 1.9% [6/322]; Japanese, 0.3% [1/322]; other Asian, 1.6% [5/322]; other Asian/American Indian, 0.3% [1/322]), and 5.6% (18/322) were American Indian/other/ unspecified (American Indian, 0.3% [1/322]; other, 0.3% [1/322]; unspecified, 5.0% [16/322]). Overall, the BFC patient population was more ethnically and racially diverse than that of UPMC (P<.001).
Forty-six percent (35/76) of BFC patients and 4.3% (14/322) of UPMC patients had LEP (P<.001). Primary languages among BFC patients were 53.9% (41/76) English, 40.8% (31/76) Spanish, and 5.2% (4/76) other/ unspecified (Chinese, 1.3% [1/76]; Indonesian, 2.6% [2/76]; unspecified, 1.3% [1/76]). Primary languages among UPMC patients were 95.7% (308/322) English and 4.3% (14/322) other/unspecified (Chinese, 0.6% [2/322]; Nepali, 0.6% [2/322]; Pali, 0.3% [1/322]; Russian, 0.3% [1/322]; unspecified, 2.5% [8/322]). There were notable differences in insurance status at the BFC vs UPMC (P<.001), with more UPMC patients having private insurance (52.8% [170/322] vs 11.8% [9/76]) and more BFC patients being uninsured (52.8% [51/76] vs 1.9% [6/322]). There was no significant difference in distance to clinic between the 2 groups (P=.183). More UPMC patients had a history of skin cancer (P=.003). More patients at the BFC were no-shows for their appointments (P<.001), and UPMC patients more frequently canceled their appointments (P<.001).
Dermatologic Diagnoses—The most commonly diagnosed dermatologic conditions at the BFC were dermatitis (23.7% [18/76]), neoplasm of uncertain behavior (15.8% [12/76]), alopecia (11.8% [9/76]), and acne (10.5% [8/76]) (Table 2). The most commonly diagnosed conditions at UPMC were nevi (26.4% [85/322]), dermatitis (22.7% [73/322]), seborrheic keratosis (21.7% [70/322]), and skin cancer screening (21.4% [70/322]). Neoplasm of uncertain behavior was more common in BFC vs UPMC patients (P=.040), while UPMC patients were more frequently diagnosed with nevi (P<.001), seborrheic keratosis (P<.001), and skin cancer screening (P<.001). There was no significant difference between the incidence of skin cancer diagnoses in the BFC (1.3% [1/76]) and UPMC (0.6% [2/76]) patient populations (P=.471). Among the biopsied neoplasms, there was also no significant difference in malignant (BFC, 50.0% [5/10]; UPMC, 32.0% [8/25]) and benign (BFC, 50.0% [5/10]; UPMC, 36.0% [9/25]) neoplasms diagnosed at each clinic (P=.444).
Management Strategies—Systemic antibiotics were more frequently prescribed (P<.001) and laboratory testing/ imaging were more frequently ordered (P=.005) at the BFC vs UPMC (Table 3). Patients at the BFC also more frequently required emergency insurance (P=.036). Patients at UPMC were more frequently recommended sunscreen (P=.003) and received education about skin cancer signs by review of the ABCDEs of melanoma (P<.001), sun-protective behaviors (P=.001), and skin examination frequency (P<.001). Notes in the EMR for UPMC patients more frequently specified patient followup instructions (P<.001).
Comment
As of 2020, the city of Pittsburgh had an estimated population of nearly 303,000 based on US Census data.10 Its population is predominantly White (62.7%) followed by Black/African American (22.8%) and Asian (6.5%); 5.9% identify as 2 or more races. Approximately 3.8% identify as Hispanic or Latino. More than 11% of the Pittsburgh population aged 5 years and older speaks a language other than English as their primary language, including Spanish (2.3%), other Indo-European languages (3.9%), and Asian and Pacific Island languages (3.5%).11 More than 5% of the Pittsburgh population does not have health insurance.12
The BFC is located in Pittsburgh’s South Side area, while one of UPMC’s primary dermatology clinics is located in the Oakland district; however, most patients who seek care at these clinics live outside these areas. Our study results indicated that the BFC and UPMC serve distinct groups of people within the Pittsburgh population. The BFC patient population was younger with a higher percentage of patients who were male, Hispanic, racially diverse, and with LEP compared with the UPMC patient population. In this clinical setting, the BFC health care team engages with people from diverse backgrounds and requires greater interpreter and medical support services.
The BFC largely is supported by volunteers, UPMC, grants, and philanthropy. Dermatology clinics are staffed by paid and volunteer team members. Paid team members include 1 nurse and 1 access lead who operates the front desk and registration. Volunteer team members include 1 board-certified dermatologist from UPMC (A.J.J.), or an affiliate clinic and 1 or 2 of each of the following: UPMC dermatology residents, medical or undergraduate students from the University of Pittsburgh, AmeriCorps national service members, and student or community medical interpreters. The onsite pharmacy is run by volunteer faculty, resident, and student pharmacists from the University of Pittsburgh. Dermatology clinics are half-day clinics that occur monthly. Volunteers for each clinic are recruited approximately 1 month in advance.
Dermatology patients at the BFC are referred from the BFC general medicine clinic and nearby Federally Qualified Health Center s for simple to complex medical and surgical dermatologic skin conditions. Each BFC dermatology clinic schedules an average of 7 patients per clinic and places other patients on a wait-list unless more urgent triage is needed. Patients are notified when they are scheduled via phone or text message, and they receive a reminder call or text 1 or 2 days prior to their appointment that also asks them to confirm attendance. Patients with LEP are called with an interpreter and also may receive text reminders that can be translated using Google Translate. Patients are instructed to notify the BFC if they need to cancel or reschedule their appointment. At the end of each visit, patients are given an after-visit summary that lists follow-up instructions, medications prescribed during the visit, and upcoming appointments. The BFC offers bus tickets to help patients get to their appointments. In rare cases, the BFC may pay for a car service to drive patients to and from the clinic.
Dermatology clinics at UPMC use scheduling and self-scheduling systems through which patients can make appointments at a location of their choice with any available board-certified dermatologist or physician assistant. Patients receive a reminder phone call 3 days prior to their appointment instructing them to call the office if they are unable to keep their appointment. Patients signed up for the online portal also receive a reminder message and an option to confirm or cancel their appointment. Patients with cell phone numbers in the UPMC system receive a text message approximately 2 days prior to their appointment that allows them to preregister and pay their copayment in advance. They receive another text 20 minutes prior to their appointment with an option for contactless check-in. At the conclusion of their visit, patients can schedule a follow-up appointment and receive a printed copy of their after-visit summary that provides information about follow-up instructions, prescribed medications, and upcoming visits. They may alternatively access this summary via the online patient portal. Patients are not provided transportation to UPMC clinics, but they are offered parking validation.
Among the most common dermatologic diagnoses for each group, BFC patients presented for treatment of more acute dermatologic conditions, while UPMC patients presented for more benign and preventive-care conditions. This difference may be attributable to the BFC’s referral and triage system, wherein patients with more urgent problems are given scheduling priority. This patient care model contrasts with UPMC’s scheduling process in which no known formal triage system is utilized. Interestingly, there was no difference in skin cancer incidence despite a higher percentage of preventive skin cancer screenings at UPMC.
Patients at the BFC more often required emergency insurance for surgical interventions, which is consistent with the higher percentage of uninsured individuals in this population. Patients at UPMC more frequently were recommended sunscreen and were educated about skin cancer, sun protection, and skin examination, in part due to this group’s more extensive history of skin cancer and frequent presentation for skin cancer screenings. At the same time, educational materials for skin care at both the BFC and UPMC are populated into the EMR in English, whereas materials in other languages are less readily available.
Our retrospective study had several limitations. Demographic information that relied on clinic-dependent intake questionnaires may be limited due to variable intake processes and patients opting out of self-reporting. By comparing patient populations between 2 clinics, confounding variables such as location and hours of operation may impact the patient demographics recorded at the BFC vs UPMC. Resources and staff availability may affect the management strategies and follow-up care offered by each clinic. Our study period also was unique in that COVID-19 may have affected resources, staffing, scheduling, and logistics at both clinics.
Based on the aforementioned differences between the BFC and UPMC patient characteristics, care models should be strategically designed to support the needs of diverse populations. The BFC patient care model appropriately focuses on communication skills with patients with LEP by using interpreter services. Providing more skin care education and follow-up instructions in patients’ primary languages will help them develop a better understanding of their skin conditions. Another key asset of the BFC patient care model is its provision of social services such as transportation and insurance assistance.
To improve the UPMC patient care model, providing patients with bus tickets and car services may potentially reduce appointment cancellations. Using interpreter services to call and text appointment reminders, as well as interpreter resources to facilitate patient visits and patient instructions, also can mitigate language barriers for patients with LEP. Implementing a triage system into the UPMC scheduling system may help patients with more urgent skin conditions to be seen in a timely manner.
Other investigators have analyzed costs of care and proven the value of dermatologic services at free clinics to guide allocation of supplies and resources, demonstrating an area for future investigation at the BFC.13 A cost analysis of care provided at the BFC compared to UPMC could inform us about the value of the BFC’s services.
Conclusion
The dermatology clinics at the BFC and UPMC have distinct demographics, diagnoses, and management strategies to provide an inclusive patient care model. The services provided by both clinics are necessary to ensure that people in Pittsburgh have access to dermatologic care regardless of social barriers (eg, lack of health insurance, LEP). To achieve greater accessibility and health equity, dermatologic care at the BFC and UPMC can be improved by strengthening communication with people with LEP, providing skin care education, and offering social and scheduling services.
Approximately 25% of Americans have at least one skin condition, and 20% are estimated to develop skin cancer during their lifetime.1,2 However, 40% of the US population lives in areas underserved by dermatologists. 3 The severity and mortality of skin cancers such as melanoma and mycosis fungoides have been positively associated with minoritized race, lack of health insurance, and unstable housing status.4-6 Patients who receive health care at free clinics often are of a racial or ethnic minoritized social group, are uninsured, and/or lack stable housing; this underserved group also includes recent immigrants to the United States who have limited English proficiency (LEP).7 Only 25% of free clinics offer specialty care services such as dermatology.7,8
Of the 42 free clinics and Federally Qualified Health Centers in Pittsburgh, Pennsylvania, the Birmingham Free Clinic (BFC) is one of the few that offers specialty care services including dermatology.9 Founded in 1994, the BFC serves as a safety net for Pittsburgh’s medically underserved population, offering primary and acute care, medication access, and social services. From January 2020 to May 2022, the BFC offered 27 dermatology clinics that provided approximately 100 people with comprehensive care including full-body skin examinations, dermatologic diagnoses and treatments, minor procedures, and dermatopathology services.
In this study, we compared the BFC dermatology patient care model with that of the dermatology department at the University of Pittsburgh Medical Center (UPMC), an insurance-based tertiary referral health care system in western Pennsylvania. By analyzing the demographics, dermatologic diagnoses, and management strategies of both the BFC and UPMC, we gained an understanding of how these patient care models differ and how they can be improved to care for diverse patient populations.
Methods
A retrospective chart review of dermatology patients seen in person at the BFC and UPMC during the period from January 2020 to May 2022 was performed. The UPMC group included patients seen by 3 general dermatologists (including A.J.J.) at matched time points. Data were collected from patients’ first in-person visit during the study period. Variables of interest included patient age, sex, race, ethnicity, primary language, zip code, health insurance status, distance to clinic (estimated using Google Maps to calculate the shortest driving distance from the patient’s zip code to the clinic), history of skin cancer, dermatologic diagnoses, and management strategies. These variables were not collected for patients who cancelled or noshowed their first in-person appointments. All patient charts and notes corresponding to the date and visit of interest were accessed through the electronic medical record (EMR). Patient data were de-identified and stored in a password-protected spreadsheet. Comparisons between the BFC and UPMC patient populations were performed using X2 tests of independence, Fisher exact tests, and Mann-Whitney U tests via SPSS software (IBM). Statistical significance was set at P<.05.
Results
Patient Characteristics—Our analysis included 76 initial appointments at the BFC and 322 at UPMC (Table 1). The mean age for patients at the BFC and UPMC was 39.6 years and 47.8 years, respectively (P=.001). Males accounted for 39 (51.3%) and 112 (34.8%) of BFC and UPMC patients, respectively (P=.008); 2 (0.6%) patients from UPMC were transgender. Of the BFC and UPMC patients, 44.7% (34/76) and 0.9% (3/322) were Hispanic, respectively (P<.001). With regard to race, 52.6% (40/76) of BFC patients were White, 19.7% (15/76) were Black, 6.6% (5/76) were Asian/Pacific Islander (Chinese, 1.3% [1/76]; other Asian, 5.3% [4/76]), and 21.1% (16/76) were American Indian/other/unspecified (American Indian, 1.3% [1/76]; other, 13.2% [10/76]; unspecified, 6.6% [5/76]). At UPMC, 61.2% (197/322) of patients were White, 28.0% (90/322) were Black, 5.3% (17/322) were Asian/Pacific Islander (Chinese, 1.2% [4/322]; Indian [Asian], 1.9% [6/322]; Japanese, 0.3% [1/322]; other Asian, 1.6% [5/322]; other Asian/American Indian, 0.3% [1/322]), and 5.6% (18/322) were American Indian/other/ unspecified (American Indian, 0.3% [1/322]; other, 0.3% [1/322]; unspecified, 5.0% [16/322]). Overall, the BFC patient population was more ethnically and racially diverse than that of UPMC (P<.001).
Forty-six percent (35/76) of BFC patients and 4.3% (14/322) of UPMC patients had LEP (P<.001). Primary languages among BFC patients were 53.9% (41/76) English, 40.8% (31/76) Spanish, and 5.2% (4/76) other/ unspecified (Chinese, 1.3% [1/76]; Indonesian, 2.6% [2/76]; unspecified, 1.3% [1/76]). Primary languages among UPMC patients were 95.7% (308/322) English and 4.3% (14/322) other/unspecified (Chinese, 0.6% [2/322]; Nepali, 0.6% [2/322]; Pali, 0.3% [1/322]; Russian, 0.3% [1/322]; unspecified, 2.5% [8/322]). There were notable differences in insurance status at the BFC vs UPMC (P<.001), with more UPMC patients having private insurance (52.8% [170/322] vs 11.8% [9/76]) and more BFC patients being uninsured (52.8% [51/76] vs 1.9% [6/322]). There was no significant difference in distance to clinic between the 2 groups (P=.183). More UPMC patients had a history of skin cancer (P=.003). More patients at the BFC were no-shows for their appointments (P<.001), and UPMC patients more frequently canceled their appointments (P<.001).
Dermatologic Diagnoses—The most commonly diagnosed dermatologic conditions at the BFC were dermatitis (23.7% [18/76]), neoplasm of uncertain behavior (15.8% [12/76]), alopecia (11.8% [9/76]), and acne (10.5% [8/76]) (Table 2). The most commonly diagnosed conditions at UPMC were nevi (26.4% [85/322]), dermatitis (22.7% [73/322]), seborrheic keratosis (21.7% [70/322]), and skin cancer screening (21.4% [70/322]). Neoplasm of uncertain behavior was more common in BFC vs UPMC patients (P=.040), while UPMC patients were more frequently diagnosed with nevi (P<.001), seborrheic keratosis (P<.001), and skin cancer screening (P<.001). There was no significant difference between the incidence of skin cancer diagnoses in the BFC (1.3% [1/76]) and UPMC (0.6% [2/76]) patient populations (P=.471). Among the biopsied neoplasms, there was also no significant difference in malignant (BFC, 50.0% [5/10]; UPMC, 32.0% [8/25]) and benign (BFC, 50.0% [5/10]; UPMC, 36.0% [9/25]) neoplasms diagnosed at each clinic (P=.444).
Management Strategies—Systemic antibiotics were more frequently prescribed (P<.001) and laboratory testing/ imaging were more frequently ordered (P=.005) at the BFC vs UPMC (Table 3). Patients at the BFC also more frequently required emergency insurance (P=.036). Patients at UPMC were more frequently recommended sunscreen (P=.003) and received education about skin cancer signs by review of the ABCDEs of melanoma (P<.001), sun-protective behaviors (P=.001), and skin examination frequency (P<.001). Notes in the EMR for UPMC patients more frequently specified patient followup instructions (P<.001).
Comment
As of 2020, the city of Pittsburgh had an estimated population of nearly 303,000 based on US Census data.10 Its population is predominantly White (62.7%) followed by Black/African American (22.8%) and Asian (6.5%); 5.9% identify as 2 or more races. Approximately 3.8% identify as Hispanic or Latino. More than 11% of the Pittsburgh population aged 5 years and older speaks a language other than English as their primary language, including Spanish (2.3%), other Indo-European languages (3.9%), and Asian and Pacific Island languages (3.5%).11 More than 5% of the Pittsburgh population does not have health insurance.12
The BFC is located in Pittsburgh’s South Side area, while one of UPMC’s primary dermatology clinics is located in the Oakland district; however, most patients who seek care at these clinics live outside these areas. Our study results indicated that the BFC and UPMC serve distinct groups of people within the Pittsburgh population. The BFC patient population was younger with a higher percentage of patients who were male, Hispanic, racially diverse, and with LEP compared with the UPMC patient population. In this clinical setting, the BFC health care team engages with people from diverse backgrounds and requires greater interpreter and medical support services.
The BFC largely is supported by volunteers, UPMC, grants, and philanthropy. Dermatology clinics are staffed by paid and volunteer team members. Paid team members include 1 nurse and 1 access lead who operates the front desk and registration. Volunteer team members include 1 board-certified dermatologist from UPMC (A.J.J.), or an affiliate clinic and 1 or 2 of each of the following: UPMC dermatology residents, medical or undergraduate students from the University of Pittsburgh, AmeriCorps national service members, and student or community medical interpreters. The onsite pharmacy is run by volunteer faculty, resident, and student pharmacists from the University of Pittsburgh. Dermatology clinics are half-day clinics that occur monthly. Volunteers for each clinic are recruited approximately 1 month in advance.
Dermatology patients at the BFC are referred from the BFC general medicine clinic and nearby Federally Qualified Health Center s for simple to complex medical and surgical dermatologic skin conditions. Each BFC dermatology clinic schedules an average of 7 patients per clinic and places other patients on a wait-list unless more urgent triage is needed. Patients are notified when they are scheduled via phone or text message, and they receive a reminder call or text 1 or 2 days prior to their appointment that also asks them to confirm attendance. Patients with LEP are called with an interpreter and also may receive text reminders that can be translated using Google Translate. Patients are instructed to notify the BFC if they need to cancel or reschedule their appointment. At the end of each visit, patients are given an after-visit summary that lists follow-up instructions, medications prescribed during the visit, and upcoming appointments. The BFC offers bus tickets to help patients get to their appointments. In rare cases, the BFC may pay for a car service to drive patients to and from the clinic.
Dermatology clinics at UPMC use scheduling and self-scheduling systems through which patients can make appointments at a location of their choice with any available board-certified dermatologist or physician assistant. Patients receive a reminder phone call 3 days prior to their appointment instructing them to call the office if they are unable to keep their appointment. Patients signed up for the online portal also receive a reminder message and an option to confirm or cancel their appointment. Patients with cell phone numbers in the UPMC system receive a text message approximately 2 days prior to their appointment that allows them to preregister and pay their copayment in advance. They receive another text 20 minutes prior to their appointment with an option for contactless check-in. At the conclusion of their visit, patients can schedule a follow-up appointment and receive a printed copy of their after-visit summary that provides information about follow-up instructions, prescribed medications, and upcoming visits. They may alternatively access this summary via the online patient portal. Patients are not provided transportation to UPMC clinics, but they are offered parking validation.
Among the most common dermatologic diagnoses for each group, BFC patients presented for treatment of more acute dermatologic conditions, while UPMC patients presented for more benign and preventive-care conditions. This difference may be attributable to the BFC’s referral and triage system, wherein patients with more urgent problems are given scheduling priority. This patient care model contrasts with UPMC’s scheduling process in which no known formal triage system is utilized. Interestingly, there was no difference in skin cancer incidence despite a higher percentage of preventive skin cancer screenings at UPMC.
Patients at the BFC more often required emergency insurance for surgical interventions, which is consistent with the higher percentage of uninsured individuals in this population. Patients at UPMC more frequently were recommended sunscreen and were educated about skin cancer, sun protection, and skin examination, in part due to this group’s more extensive history of skin cancer and frequent presentation for skin cancer screenings. At the same time, educational materials for skin care at both the BFC and UPMC are populated into the EMR in English, whereas materials in other languages are less readily available.
Our retrospective study had several limitations. Demographic information that relied on clinic-dependent intake questionnaires may be limited due to variable intake processes and patients opting out of self-reporting. By comparing patient populations between 2 clinics, confounding variables such as location and hours of operation may impact the patient demographics recorded at the BFC vs UPMC. Resources and staff availability may affect the management strategies and follow-up care offered by each clinic. Our study period also was unique in that COVID-19 may have affected resources, staffing, scheduling, and logistics at both clinics.
Based on the aforementioned differences between the BFC and UPMC patient characteristics, care models should be strategically designed to support the needs of diverse populations. The BFC patient care model appropriately focuses on communication skills with patients with LEP by using interpreter services. Providing more skin care education and follow-up instructions in patients’ primary languages will help them develop a better understanding of their skin conditions. Another key asset of the BFC patient care model is its provision of social services such as transportation and insurance assistance.
To improve the UPMC patient care model, providing patients with bus tickets and car services may potentially reduce appointment cancellations. Using interpreter services to call and text appointment reminders, as well as interpreter resources to facilitate patient visits and patient instructions, also can mitigate language barriers for patients with LEP. Implementing a triage system into the UPMC scheduling system may help patients with more urgent skin conditions to be seen in a timely manner.
Other investigators have analyzed costs of care and proven the value of dermatologic services at free clinics to guide allocation of supplies and resources, demonstrating an area for future investigation at the BFC.13 A cost analysis of care provided at the BFC compared to UPMC could inform us about the value of the BFC’s services.
Conclusion
The dermatology clinics at the BFC and UPMC have distinct demographics, diagnoses, and management strategies to provide an inclusive patient care model. The services provided by both clinics are necessary to ensure that people in Pittsburgh have access to dermatologic care regardless of social barriers (eg, lack of health insurance, LEP). To achieve greater accessibility and health equity, dermatologic care at the BFC and UPMC can be improved by strengthening communication with people with LEP, providing skin care education, and offering social and scheduling services.
- Lim HW, Collins SAB, Resneck JS, et al. The burden of skin disease in the United States. J Am Acad Dermatol. 2017;76:958-972.e2. doi:10.1016/j.jaad.2016.12.043
- American Academy of Dermatology. Skin cancer. Accessed October 7, 2024. https://www.aad.org/media/stats-skin-cancer
- Suneja T, Smith ED, Chen GJ, et al. Waiting times to see a dermatologist are perceived as too long by dermatologists: implications for the dermatology workforce. Arch Dermatol. 2001;137:1303-1307. doi:10.1001/archderm.137.10.1303
- Grossberg AL, Carranza D, Lamp K, et al. Dermatologic care in the homeless and underserved populations: observations from the Venice Family Clinic. Cutis. 2012;89:25-32.
- Amini A, Rusthoven CG, Waxweiler TV, et al. Association of health insurance with outcomes in adults ages 18 to 64 years with melanoma in the United States. J Am Acad Dermatol. 2016;74:309-316. doi:10.1016/j.jaad.2015.09.054
- Su C, Nguyen KA, Bai HX, et al. Racial disparity in mycosis fungoides: an analysis of 4495 cases from the US National Cancer Database. J Am Acad Dermatol. 2017;77:497-502.e2. doi:10.1016/j.jaad .2017.04.1137
- Darnell JS. Free clinics in the United States: a nationwide survey. Arch Intern Med. 2010;170:946-953. doi:10.1001/archinternmed .2010.107
- Madray V, Ginjupalli S, Hashmi O, et al. Access to dermatology services at free medical clinics: a nationwide cross-sectional survey. J Am Acad Dermatol. 2019;81:245-246. doi:10.1016/j.jaad.2018.12.011
- Pennsylvania free and income-based clinics. Accessed October 7, 2024. https://www.freeclinics.com/sta/pennsylvania
- United States Census Bureau. Decennial census. P1: race. Accessed October 7, 2024. https://data.census.gov/table/DECENNIALPL2020.P1?g=160XX00US4261000
- United States Census Bureau. American community survey. S1601: language spoken at home. Accessed October 7, 2024. https://data.census.gov/table/ACSST5Y2020S1601?g=160XX00US4261000
- United States Census Bureau. S2701: selected characteristics of health insurance coverage in the United States. Accessed October 7, 2024. https://data.census.gov/table/ACSST5Y2020.S2701?g=160XX00US4261000
- Lin CP, Loy S, Boothe WD, et al. Value of Dermatology Nights at a student-run free clinic. Proc (Bayl Univ Med Cent). 2020;34:260-261. doi:10.1080/08998280.2020.1834771
- Lim HW, Collins SAB, Resneck JS, et al. The burden of skin disease in the United States. J Am Acad Dermatol. 2017;76:958-972.e2. doi:10.1016/j.jaad.2016.12.043
- American Academy of Dermatology. Skin cancer. Accessed October 7, 2024. https://www.aad.org/media/stats-skin-cancer
- Suneja T, Smith ED, Chen GJ, et al. Waiting times to see a dermatologist are perceived as too long by dermatologists: implications for the dermatology workforce. Arch Dermatol. 2001;137:1303-1307. doi:10.1001/archderm.137.10.1303
- Grossberg AL, Carranza D, Lamp K, et al. Dermatologic care in the homeless and underserved populations: observations from the Venice Family Clinic. Cutis. 2012;89:25-32.
- Amini A, Rusthoven CG, Waxweiler TV, et al. Association of health insurance with outcomes in adults ages 18 to 64 years with melanoma in the United States. J Am Acad Dermatol. 2016;74:309-316. doi:10.1016/j.jaad.2015.09.054
- Su C, Nguyen KA, Bai HX, et al. Racial disparity in mycosis fungoides: an analysis of 4495 cases from the US National Cancer Database. J Am Acad Dermatol. 2017;77:497-502.e2. doi:10.1016/j.jaad .2017.04.1137
- Darnell JS. Free clinics in the United States: a nationwide survey. Arch Intern Med. 2010;170:946-953. doi:10.1001/archinternmed .2010.107
- Madray V, Ginjupalli S, Hashmi O, et al. Access to dermatology services at free medical clinics: a nationwide cross-sectional survey. J Am Acad Dermatol. 2019;81:245-246. doi:10.1016/j.jaad.2018.12.011
- Pennsylvania free and income-based clinics. Accessed October 7, 2024. https://www.freeclinics.com/sta/pennsylvania
- United States Census Bureau. Decennial census. P1: race. Accessed October 7, 2024. https://data.census.gov/table/DECENNIALPL2020.P1?g=160XX00US4261000
- United States Census Bureau. American community survey. S1601: language spoken at home. Accessed October 7, 2024. https://data.census.gov/table/ACSST5Y2020S1601?g=160XX00US4261000
- United States Census Bureau. S2701: selected characteristics of health insurance coverage in the United States. Accessed October 7, 2024. https://data.census.gov/table/ACSST5Y2020.S2701?g=160XX00US4261000
- Lin CP, Loy S, Boothe WD, et al. Value of Dermatology Nights at a student-run free clinic. Proc (Bayl Univ Med Cent). 2020;34:260-261. doi:10.1080/08998280.2020.1834771
Comparing Patient Care Models at a Local Free Clinic vs an Insurance- Based University Medical Center
Comparing Patient Care Models at a Local Free Clinic vs an Insurance- Based University Medical Center
PRACTICE POINTS
- Both free clinics and insurance-based health care systems serve dermatology patients with diverse characteristics, necessitating inclusive health care models.
- Dermatologic care can be improved at both free and insurance-based clinics by strengthening communication with individuals with limited English proficiency, providing skin care education, and offering social and scheduling services such as transportation, insurance assistance, and triage.
Multidisciplinary Amputation Prevention at the DeBakey VA Hospital: Our First Decade
Individuals with diabetes are at risk for developing foot ulcers or full-thickness defects in the epithelium of the foot. These defects can lead to bacterial invasion and foot infection, potentially resulting in leg amputation (Figure 1). Effective treatment to prevent leg amputation, known as limb salvage, requires management across multiple medical specialties including podiatry, vascular surgery, and infectious diseases. The multidisciplinary team approach to limb salvage was introduced in Boston in 1928 and has been the prevailing approach to this cross-specialty medical problem for at least a decade.1,2
The Michael E. DeBakey Veterans Affairs Medical Center (MEDVAMC) has established an inpatient limb salvage program—a group of dedicated clinicians working collaboratively to provide evidence-guided management of patients hospitalized with foot ulcers, foot gangrene or any superimposed infection with the goal of avoiding leg amputations. We have seen a significant and durable reduction in the incidence of leg amputations among veterans at MEDVAMC.
This article describes the evolution and outcomes of the MEDVAMC limb salvage program over more than a decade. It includes changes to team structure and workflow, as well as past and present successes and challenges. The eAppendix provides a narrative summary with examples of how our clinical practice and research efforts have informed one another and how these findings are applied to clinical management. This process is part of the larger efforts of the Veterans Health Administration (VHA) to create a learning health system in which “internal data and experience are systematically integrated with external evidence, and that knowledge is put into practice.”3
Methods
Data from the VHA Support Service Center were used to obtain monthly major (leg) and minor (toe and partial foot) amputation records at MEDVAMC from October 2000 through May 2023. Yearly totals for the number of persons with diabetes and foot ulcers at MEDVAMC were also obtained from the support service center. Annual patient population sizes and number of persons with foot ulcers were converted to monthly estimates using cubic spline interpolation. Rates were calculated as 12-month rolling averages. Trend lines were created with locally weighted running line smoothing that used a span α of 0.1.
We characterized the patient population using data from cohorts of veterans treated for foot ulcers and foot infections at MEDVAMC. To compare the contemporary veteran population with nonveteran inpatients treated for foot ulcers and foot infections at other hospitals, we created a 2:1 nonveteran to veteran cohort matched by sex and zip code, using publicly available hospital admission data from the Texas Department of Health and State Health Services. Veterans used for this cohort comparison are consistent with the 100 consecutive patients who underwent angiography for limb salvage in 2022.
This research was approved by the Baylor College of Medicine Institutional Review Board (protocol H-34858) and the MEDVAMC Research Committee (IRBNet protocol 15A12. HB). All analyses used deidentified data in the R programming language version 4.2.2 using RStudio version 2022.06.0 Build 421.
Program Description
MEDVAMC is a 350-bed teaching hospital located in central Houston. Its hospital system includes 11 outpatient clinics, ranging from 28 to 126 miles (eAppendix, Supplemental Figure A) from MEDVAMC. MEDVAMC provides vascular, orthopedic, and podiatric surgery services, as well as many other highly specialized services such as liver and heart transplants. The hospital’s risk-adjusted rates of operative morbidity and mortality (observed-to-expected ratios) are significantly lower than expected.
Despite this, the incidence rate of leg amputations at MEDVAMC in early 2011 was nearly 3-times higher than the VHA average. The inpatient management of veterans with infected foot ulcers was fragmented, with the general, orthopedic, and vascular surgery teams separately providing siloed care. Delays in treatment were common. There was much service- and practitioner-level practice heterogeneity. No diagnostic or treatment protocols were used, and standard treatment components were sporadically provided.
Patient Population
Compared to the matched non-VHA patient cohort (Supplemental Table 1), veterans treated at MEDVAMC for limb salvage are older. Nearly half (46%) identify as Black, which is associated with a 2-fold higher riskadjusted rate of leg amputations.4 MEDVAMC patients also have significantly higher rates of diabetes, chronic kidney disease, and systolic heart failure. About 22% travel > 40 miles for treatment at MEDVAMC, double that of the matched cohort (10.7%). Additionally, 35% currently smoke and 37% have moderate to severe peripheral artery disease (PAD).5
Program Design
In late 2011, the MEDVAMC vascular surgery team led limb salvage efforts by implementing a single team model, which involved assuming the primary role of managing foot ulcers for all veterans, both infected and uninfected (eAppendix, Supplemental Figure B). Consultations were directed to a dedicated limb salvage pager. The vascular team provided interdisciplinary limb salvage management across the spectrum of disease, including the surgical treatment of infection, assessment for PAD, open surgical operations and endovascular interventions to treat PAD, and foot reconstruction (debridement, minor or partial foot amputations, and skin grafting). This care was complemented by frequent consultation with the infectious disease, vascular medicine, podiatry, and geriatric wound care teams. This approach streamlined the delivery of consistent multidisciplinary care.
This collaborative effort aimed to develop ideal multidisciplinary care plans through research spanning the spectrum of the diabetic foot infection disease process (eAppendix, Supplemental Table 1). Some of the most impactful practices were: (1) a proclivity towards surgical treatment of foot infections, especially osteomyelitis5; (2) improved identification of PAD6,7; (3) early surgical closure of foot wounds following revascularization8,9; and (4) palliative wound care as an alternative to leg amputation in veterans who are not candidates for revascularization and limb salvage.10 Initally, the vascular surgery team held monthly multidisciplinary limb salvage meetings to coordinate patient management, identify ways to streamline care and avoid waste, discuss research findings, and review the 12-month rolling average of the MEDVAMC leg amputation incidence rate.
During the study period, the MEDVAMC vascular surgery team consisted of 2 to 5 board certified vascular or general surgeons, 2 or 3 nurse practitioners, and 3 vascular ultrasound technologists. Associated specialists included 2 podiatrists, 3 geriatricians with wound care certification, as well as additional infectious diseases, vascular medicine, orthopedics, and general surgery specialists.
Program Assessment
We noted a significant and sustained decrease in the MEDVAMC leg amputation rate after implementing multidisciplinary meetings and a single- team model from early 2012 through 2017 (Figure 2). The amputation incidence rate decreased steadily over the period from a maximum of 160 per 100,000 per year in February 2012 to a nadir of 66 per 100,000 per year in April 2017, an overall 60% decrease. Increases were noted in early 2018 after ceasing the single- team model, and in the summer of 2022, following periods of bed shortages after the onset of the COVID-19 pandemic. Tracking this metric allowed clinicians to make course corrections.
The decreased leg amputation rate at MEDVAMC does not seem to be mirroring national or regional trends. During this 10-year period, the VHA annualized amputation rate decreased minimally, from 58 to 54 per 100,000 (eAppendix Supplemental Figure C). Leg amputation incidence at non-VHA hospitals in Texas slightly increased over the same period.11
Value was also reflected in other metrics. MEDVAMC improved safety through a bundled strategy that reduced the risk-adjusted rate of surgical wound infections by 95%.12 MEDVAMC prioritized limb salvage when selecting patients for angiography and nearly eliminated using stent-grafts, cryopreserved allogeneic saphenous vein grafts, and expensive surgical and endovascular implants, which were identified as more expensive and less effective than other options (Figure 3).13-15 The MEDVAMC team achieved a > 90% patient trust rating on the Veterans Signals survey in fiscal years 2021 and 2022.
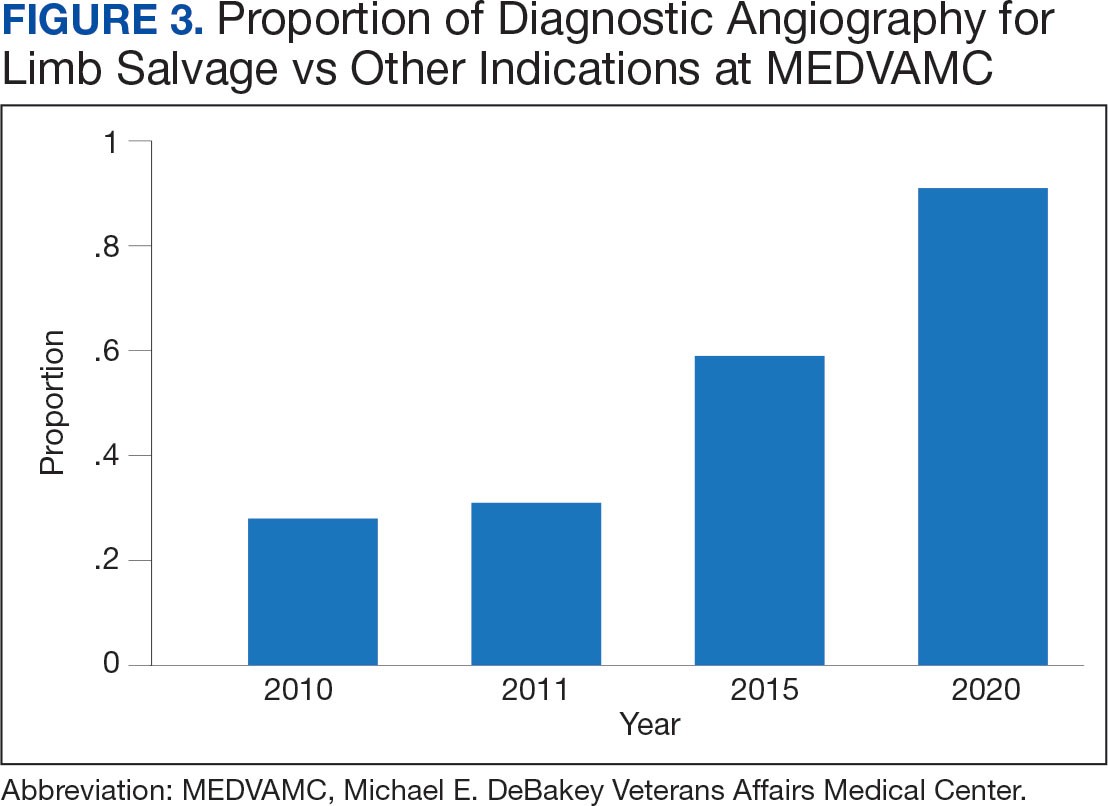
Challenges
A significant increase in the patient-physician ratio occurred 5 years into the program. In 2016, 2 vascular surgeons left MEDVAMC and a planned renovation of 1 of the 2 vascular surgery-assigned hybrid working facilities began even as the number of MEDVAMC patients with diabetes grew 120% (from 89,400 to 107,746 between 2010 and 2016), and the incidence rate of foot ulcers grew 300% (from 392 in 2010 to 1183 in 2016 per 100,000). The net result was a higher clinical workload among the remaining vascular surgeons with less operating room availability.
To stabilize surgeon retention, MEDVAMC reverted from the single team model back to inpatient care being distributed among general surgery, orthopedic surgery, and vascular surgery. After noting an increase in the leg amputation incidence rate, we adjusted the focus from multidisciplinary to interdisciplinary care (ie, majority of limb salvage clinical care can be provided by practitioners of any involved specialties). We worked to establish a local, written, interdisciplinary consensus on evaluating and managing veterans with nonhealing foot ulcers to mitigate the loss of a consolidated inpatient approach. Despite frequent staff turnover, ≥ 1 physician or surgeon from the core specialties of vascular surgery, podiatry, and infectious diseases remained throughout the study period.
The COVID-19 pandemic caused a shortage of hospital beds. This was followed by more bed shortages due to decreased nursing staff. Our health care system also had a period of restricted outpatient encounters early in the pandemic. During this time, we noted a delayed presentation of veterans with advanced infections and another increase in leg amputation incidence rate.
Like many health systems, MEDVAMC pivoted to telephone- and video-based outpatient encounters. Our team also used publicly available Texas hospitalization data to identify zip codes with particularly high leg amputation incidence rates, and > 3500 educational mailings to veterans categorized as moderate and high risk for leg amputation in these zip codes. These mailings provided information on recognizing foot ulcers and infections, emphasized timely evaluation, and named the MEDVAMC vascular surgery team as a point-of-contact. More recently, we have seen a further decrease in the MEDVAMC incidences of leg amputation to its lowest rate in > 20 years.
Discussion
A learning organization that directs its research based on clinical observations and informs its clinical care with research findings can produce palpable improvements in outcomes. Understanding the disease process and trying to better understand management across the entire range of this disease process has allowed our team to make consistent and systematic changes in care (Table). Consolidating inpatient care in a single team model seems to have been effective in reducing amputation rates among veterans with diabetes. The role the MEDVAMC vascular surgery team served for limb salvage patients may have been particularly beneficial because of the large impact untreated or unidentified PAD can have and because of the high prevalence of PAD among the limb salvage population seen at MEDVAMC. To be sustainable, though, a single-team model needs resources. A multiteam model can also be effective if the degree of multidisciplinary involvement for any given veteran is appropriate to the individual's clinical needs, teams are engaged and willing to contribute in a defined role within their specialty, and lines of communication remain open.
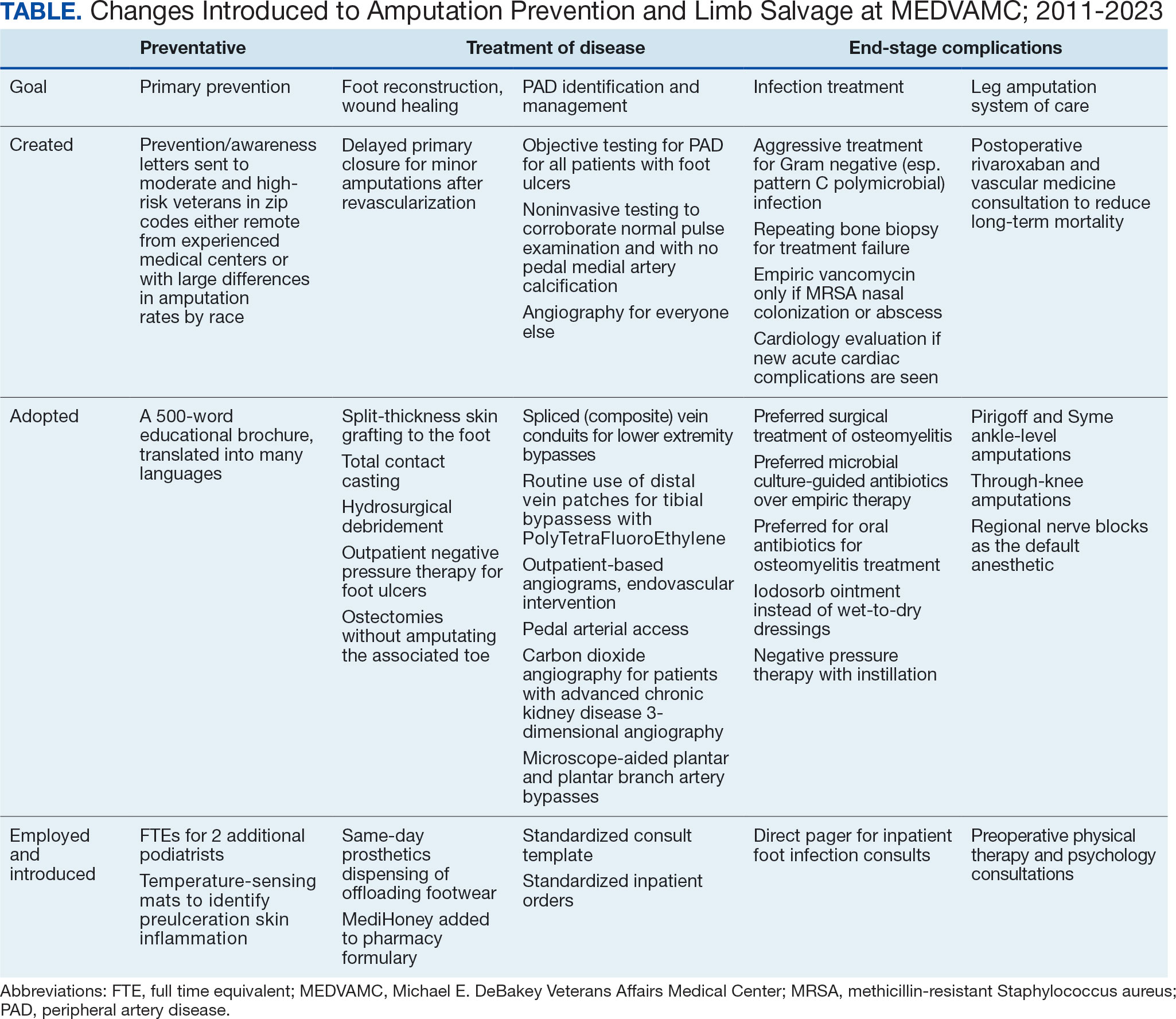
The primary challenge at MEDVAMC has been, and will continue to be, the retention of physicians and surgeons. MEDVAMC has excellent leadership and a collegial working environment, but better access to operating rooms for elective and time-sensitive operations, additional clinical staff support, and higher salary at non-VA positions have been the basis for many of physicians— especially surgeons—leaving MEDVAMC. Despite high staff turnover and a constant flow of resident and fellow trainees, MEDVAMC has been able to keep the clinical approach relatively consistent due to the use of written protocols and continuity of care as ≥ 1 physician or surgeon from each of the 4 main teams remained engaged with limb salvage throughout the entire period.
Going forward, we will work to ensure that all requirements of the 2022 Prevention of Amputation in Veterans Everywhere directive are incorporated into care.8 We plan to standardize MEDVAMC management algorithms further, both to streamline care and reduce the opportunity for disparities in treatment. More prophylactic podiatric procedures, surgical forms of offloading, and a shared multidisciplinary clinic space may also further help patients.
Conclusions
The introduction of multidisciplinary limb salvage at MEDVAMC has led to significant and sustained reductions in leg amputation incidence. These reductions do not seem dependent upon a specific team structure for inpatient care. To improve patient outcomes, efforts should focus on making improvements across the entire disease spectrum. For limb salvage, this includes primary prevention of foot ulcers, the treatment of foot infections, identification and management of PAD, surgical reconstruction/optimal wound healing, and care for patients who undergo leg amputation.
- Sanders LJ, Robbins JM, Edmonds ME. History of the team approach to amputation prevention: pioneers and milestones. J Am Podiatr Med Assoc. 2010;100(5):317- 334. doi:10.7547/1000317
- Sumpio BE, Armstrong DG, Lavery LA, Andros G. The role of interdisciplinary team approach in the management of the diabetic foot: a joint statement from the society for vascular surgery and the American podiatric medical association. J Am Podiatr Med Assoc. 2010;100(4):309-311. doi:10.7547/1000309
- About learning health systems. Agency for Healthcare Research and Quality. Published March 2019. Updated May 2019. Accessed October 9, 2024. https://www.ahrq.gov/learning-health-systems/about.html
- Barshes NR, Minc SD. Healthcare disparities in vascular surgery: a critical review. J Vasc Surg. 2021;74(2S):6S-14S.
- Barshes NR, Mindru C, Ashong C, Rodriguez-Barradas M, Trautner BW. Treatment failure and leg amputation among patients with foot osteomyelitis. Int J Low Extrem Wounds. 2016;15(4):303-312. doi:10.1177/1534734616661058
- Barshes NR, Flores E, Belkin M, Kougias P, Armstrong DG, Mills JL Sr. The accuracy and cost-effectiveness of strategies used to identify peripheral artery disease among patients with diabetic foot ulcers. J Vasc Surg. 2016;64(6):1682-1690.e3. doi:10.1016/j.jvs.2016.04.056 e1. doi:10.1016/j.jvs.2021.03.055
- Choi JC, Miranda J, Greenleaf E, et al. Lower-extremity pressure, staging, and grading thresholds to identify chronic limb-threatening ischemia. Vasc Med. 2023;28(1):45-53. doi:10.1177/1358863X221147945
- Barshes NR, Chambers JD, Cohen J, Belkin M; Model To Optimize Healthcare Value in Ischemic Extremities 1 (MOVIE) Study Collaborators. Cost-effectiveness in the contemporary management of critical limb ischemia with tissue loss. J Vasc Surg. 2012;56(4):1015-24.e1. doi:10.1016/j.jvs.2012.02.069
- Barshes NR, Bechara CF, Pisimisis G, Kougias P. Preliminary experiences with early primary closure of foot wounds after lower extremity revascularization. Ann Vasc Surg. 2014;28(1):48-52. doi:10.1016/j.avsg.2013.06.012
- Barshes NR, Gold B, Garcia A, Bechara CF, Pisimisis G, Kougias P. Minor amputation and palliative wound care as a strategy to avoid major amputation in patients with foot infections and severe peripheral arterial disease. Int J Low Extrem Wounds. 2014;13(3):211-219. doi:10.1177/1534734614543663
- Garcia M, Hernandez B, Ellington TG, et al. A lack of decline in major nontraumatic amputations in Texas: contemporary trends, risk factor associations, and impact of revascularization. Diabetes Care. 2019;42(6):1061-1066. doi:10.2337/dc19-0078
- Zamani N, Sharath SE, Vo E, Awad SS, Kougias P, Barshes NR. A multi-component strategy to decrease wound complications after open infra-inguinal re-vascularization. Surg Infect (Larchmt). 2018;19(1):87-94. doi:10.1089/sur.2017.193
- Barshes NR, Ozaki CK, Kougias P, Belkin M. A costeffectiveness analysis of infrainguinal bypass in the absence of great saphenous vein conduit. J Vasc Surg. 2013;57(6):1466-1470. doi:10.1016/j.jvs.2012.11.115
- Zamani N, Sharath S, Browder R, et al. PC158 longterm outcomes after endovascular stent placement for symptomatic, long-segment superficial femoral artery lesions. J Vasc Surg. 2017;65(6):182S-183S. doi:10.1016/j.jvs.2017.03.344
- Zamani N, Sharath SE, Browder RC, et al. Outcomes after endovascular stent placement for long-segment superficial femoral artery lesions. Ann Vasc Surg. 2021;71:298-307. doi:10.1016/j.avsg.2020.08.124
Individuals with diabetes are at risk for developing foot ulcers or full-thickness defects in the epithelium of the foot. These defects can lead to bacterial invasion and foot infection, potentially resulting in leg amputation (Figure 1). Effective treatment to prevent leg amputation, known as limb salvage, requires management across multiple medical specialties including podiatry, vascular surgery, and infectious diseases. The multidisciplinary team approach to limb salvage was introduced in Boston in 1928 and has been the prevailing approach to this cross-specialty medical problem for at least a decade.1,2

The Michael E. DeBakey Veterans Affairs Medical Center (MEDVAMC) has established an inpatient limb salvage program—a group of dedicated clinicians working collaboratively to provide evidence-guided management of patients hospitalized with foot ulcers, foot gangrene or any superimposed infection with the goal of avoiding leg amputations. We have seen a significant and durable reduction in the incidence of leg amputations among veterans at MEDVAMC.
This article describes the evolution and outcomes of the MEDVAMC limb salvage program over more than a decade. It includes changes to team structure and workflow, as well as past and present successes and challenges. The eAppendix provides a narrative summary with examples of how our clinical practice and research efforts have informed one another and how these findings are applied to clinical management. This process is part of the larger efforts of the Veterans Health Administration (VHA) to create a learning health system in which “internal data and experience are systematically integrated with external evidence, and that knowledge is put into practice.”3
Methods
Data from the VHA Support Service Center were used to obtain monthly major (leg) and minor (toe and partial foot) amputation records at MEDVAMC from October 2000 through May 2023. Yearly totals for the number of persons with diabetes and foot ulcers at MEDVAMC were also obtained from the support service center. Annual patient population sizes and number of persons with foot ulcers were converted to monthly estimates using cubic spline interpolation. Rates were calculated as 12-month rolling averages. Trend lines were created with locally weighted running line smoothing that used a span α of 0.1.
We characterized the patient population using data from cohorts of veterans treated for foot ulcers and foot infections at MEDVAMC. To compare the contemporary veteran population with nonveteran inpatients treated for foot ulcers and foot infections at other hospitals, we created a 2:1 nonveteran to veteran cohort matched by sex and zip code, using publicly available hospital admission data from the Texas Department of Health and State Health Services. Veterans used for this cohort comparison are consistent with the 100 consecutive patients who underwent angiography for limb salvage in 2022.
This research was approved by the Baylor College of Medicine Institutional Review Board (protocol H-34858) and the MEDVAMC Research Committee (IRBNet protocol 15A12. HB). All analyses used deidentified data in the R programming language version 4.2.2 using RStudio version 2022.06.0 Build 421.
Program Description
MEDVAMC is a 350-bed teaching hospital located in central Houston. Its hospital system includes 11 outpatient clinics, ranging from 28 to 126 miles (eAppendix, Supplemental Figure A) from MEDVAMC. MEDVAMC provides vascular, orthopedic, and podiatric surgery services, as well as many other highly specialized services such as liver and heart transplants. The hospital’s risk-adjusted rates of operative morbidity and mortality (observed-to-expected ratios) are significantly lower than expected.
Despite this, the incidence rate of leg amputations at MEDVAMC in early 2011 was nearly 3-times higher than the VHA average. The inpatient management of veterans with infected foot ulcers was fragmented, with the general, orthopedic, and vascular surgery teams separately providing siloed care. Delays in treatment were common. There was much service- and practitioner-level practice heterogeneity. No diagnostic or treatment protocols were used, and standard treatment components were sporadically provided.
Patient Population
Compared to the matched non-VHA patient cohort (Supplemental Table 1), veterans treated at MEDVAMC for limb salvage are older. Nearly half (46%) identify as Black, which is associated with a 2-fold higher riskadjusted rate of leg amputations.4 MEDVAMC patients also have significantly higher rates of diabetes, chronic kidney disease, and systolic heart failure. About 22% travel > 40 miles for treatment at MEDVAMC, double that of the matched cohort (10.7%). Additionally, 35% currently smoke and 37% have moderate to severe peripheral artery disease (PAD).5
Program Design
In late 2011, the MEDVAMC vascular surgery team led limb salvage efforts by implementing a single team model, which involved assuming the primary role of managing foot ulcers for all veterans, both infected and uninfected (eAppendix, Supplemental Figure B). Consultations were directed to a dedicated limb salvage pager. The vascular team provided interdisciplinary limb salvage management across the spectrum of disease, including the surgical treatment of infection, assessment for PAD, open surgical operations and endovascular interventions to treat PAD, and foot reconstruction (debridement, minor or partial foot amputations, and skin grafting). This care was complemented by frequent consultation with the infectious disease, vascular medicine, podiatry, and geriatric wound care teams. This approach streamlined the delivery of consistent multidisciplinary care.
This collaborative effort aimed to develop ideal multidisciplinary care plans through research spanning the spectrum of the diabetic foot infection disease process (eAppendix, Supplemental Table 1). Some of the most impactful practices were: (1) a proclivity towards surgical treatment of foot infections, especially osteomyelitis5; (2) improved identification of PAD6,7; (3) early surgical closure of foot wounds following revascularization8,9; and (4) palliative wound care as an alternative to leg amputation in veterans who are not candidates for revascularization and limb salvage.10 Initally, the vascular surgery team held monthly multidisciplinary limb salvage meetings to coordinate patient management, identify ways to streamline care and avoid waste, discuss research findings, and review the 12-month rolling average of the MEDVAMC leg amputation incidence rate.
During the study period, the MEDVAMC vascular surgery team consisted of 2 to 5 board certified vascular or general surgeons, 2 or 3 nurse practitioners, and 3 vascular ultrasound technologists. Associated specialists included 2 podiatrists, 3 geriatricians with wound care certification, as well as additional infectious diseases, vascular medicine, orthopedics, and general surgery specialists.
Program Assessment
We noted a significant and sustained decrease in the MEDVAMC leg amputation rate after implementing multidisciplinary meetings and a single- team model from early 2012 through 2017 (Figure 2). The amputation incidence rate decreased steadily over the period from a maximum of 160 per 100,000 per year in February 2012 to a nadir of 66 per 100,000 per year in April 2017, an overall 60% decrease. Increases were noted in early 2018 after ceasing the single- team model, and in the summer of 2022, following periods of bed shortages after the onset of the COVID-19 pandemic. Tracking this metric allowed clinicians to make course corrections.

The decreased leg amputation rate at MEDVAMC does not seem to be mirroring national or regional trends. During this 10-year period, the VHA annualized amputation rate decreased minimally, from 58 to 54 per 100,000 (eAppendix Supplemental Figure C). Leg amputation incidence at non-VHA hospitals in Texas slightly increased over the same period.11
Value was also reflected in other metrics. MEDVAMC improved safety through a bundled strategy that reduced the risk-adjusted rate of surgical wound infections by 95%.12 MEDVAMC prioritized limb salvage when selecting patients for angiography and nearly eliminated using stent-grafts, cryopreserved allogeneic saphenous vein grafts, and expensive surgical and endovascular implants, which were identified as more expensive and less effective than other options (Figure 3).13-15 The MEDVAMC team achieved a > 90% patient trust rating on the Veterans Signals survey in fiscal years 2021 and 2022.

Challenges
A significant increase in the patient-physician ratio occurred 5 years into the program. In 2016, 2 vascular surgeons left MEDVAMC and a planned renovation of 1 of the 2 vascular surgery-assigned hybrid working facilities began even as the number of MEDVAMC patients with diabetes grew 120% (from 89,400 to 107,746 between 2010 and 2016), and the incidence rate of foot ulcers grew 300% (from 392 in 2010 to 1183 in 2016 per 100,000). The net result was a higher clinical workload among the remaining vascular surgeons with less operating room availability.
To stabilize surgeon retention, MEDVAMC reverted from the single team model back to inpatient care being distributed among general surgery, orthopedic surgery, and vascular surgery. After noting an increase in the leg amputation incidence rate, we adjusted the focus from multidisciplinary to interdisciplinary care (ie, majority of limb salvage clinical care can be provided by practitioners of any involved specialties). We worked to establish a local, written, interdisciplinary consensus on evaluating and managing veterans with nonhealing foot ulcers to mitigate the loss of a consolidated inpatient approach. Despite frequent staff turnover, ≥ 1 physician or surgeon from the core specialties of vascular surgery, podiatry, and infectious diseases remained throughout the study period.
The COVID-19 pandemic caused a shortage of hospital beds. This was followed by more bed shortages due to decreased nursing staff. Our health care system also had a period of restricted outpatient encounters early in the pandemic. During this time, we noted a delayed presentation of veterans with advanced infections and another increase in leg amputation incidence rate.
Like many health systems, MEDVAMC pivoted to telephone- and video-based outpatient encounters. Our team also used publicly available Texas hospitalization data to identify zip codes with particularly high leg amputation incidence rates, and > 3500 educational mailings to veterans categorized as moderate and high risk for leg amputation in these zip codes. These mailings provided information on recognizing foot ulcers and infections, emphasized timely evaluation, and named the MEDVAMC vascular surgery team as a point-of-contact. More recently, we have seen a further decrease in the MEDVAMC incidences of leg amputation to its lowest rate in > 20 years.
Discussion
A learning organization that directs its research based on clinical observations and informs its clinical care with research findings can produce palpable improvements in outcomes. Understanding the disease process and trying to better understand management across the entire range of this disease process has allowed our team to make consistent and systematic changes in care (Table). Consolidating inpatient care in a single team model seems to have been effective in reducing amputation rates among veterans with diabetes. The role the MEDVAMC vascular surgery team served for limb salvage patients may have been particularly beneficial because of the large impact untreated or unidentified PAD can have and because of the high prevalence of PAD among the limb salvage population seen at MEDVAMC. To be sustainable, though, a single-team model needs resources. A multiteam model can also be effective if the degree of multidisciplinary involvement for any given veteran is appropriate to the individual's clinical needs, teams are engaged and willing to contribute in a defined role within their specialty, and lines of communication remain open.

The primary challenge at MEDVAMC has been, and will continue to be, the retention of physicians and surgeons. MEDVAMC has excellent leadership and a collegial working environment, but better access to operating rooms for elective and time-sensitive operations, additional clinical staff support, and higher salary at non-VA positions have been the basis for many of physicians— especially surgeons—leaving MEDVAMC. Despite high staff turnover and a constant flow of resident and fellow trainees, MEDVAMC has been able to keep the clinical approach relatively consistent due to the use of written protocols and continuity of care as ≥ 1 physician or surgeon from each of the 4 main teams remained engaged with limb salvage throughout the entire period.
Going forward, we will work to ensure that all requirements of the 2022 Prevention of Amputation in Veterans Everywhere directive are incorporated into care.8 We plan to standardize MEDVAMC management algorithms further, both to streamline care and reduce the opportunity for disparities in treatment. More prophylactic podiatric procedures, surgical forms of offloading, and a shared multidisciplinary clinic space may also further help patients.
Conclusions
The introduction of multidisciplinary limb salvage at MEDVAMC has led to significant and sustained reductions in leg amputation incidence. These reductions do not seem dependent upon a specific team structure for inpatient care. To improve patient outcomes, efforts should focus on making improvements across the entire disease spectrum. For limb salvage, this includes primary prevention of foot ulcers, the treatment of foot infections, identification and management of PAD, surgical reconstruction/optimal wound healing, and care for patients who undergo leg amputation.
Individuals with diabetes are at risk for developing foot ulcers or full-thickness defects in the epithelium of the foot. These defects can lead to bacterial invasion and foot infection, potentially resulting in leg amputation (Figure 1). Effective treatment to prevent leg amputation, known as limb salvage, requires management across multiple medical specialties including podiatry, vascular surgery, and infectious diseases. The multidisciplinary team approach to limb salvage was introduced in Boston in 1928 and has been the prevailing approach to this cross-specialty medical problem for at least a decade.1,2

The Michael E. DeBakey Veterans Affairs Medical Center (MEDVAMC) has established an inpatient limb salvage program—a group of dedicated clinicians working collaboratively to provide evidence-guided management of patients hospitalized with foot ulcers, foot gangrene or any superimposed infection with the goal of avoiding leg amputations. We have seen a significant and durable reduction in the incidence of leg amputations among veterans at MEDVAMC.
This article describes the evolution and outcomes of the MEDVAMC limb salvage program over more than a decade. It includes changes to team structure and workflow, as well as past and present successes and challenges. The eAppendix provides a narrative summary with examples of how our clinical practice and research efforts have informed one another and how these findings are applied to clinical management. This process is part of the larger efforts of the Veterans Health Administration (VHA) to create a learning health system in which “internal data and experience are systematically integrated with external evidence, and that knowledge is put into practice.”3
Methods
Data from the VHA Support Service Center were used to obtain monthly major (leg) and minor (toe and partial foot) amputation records at MEDVAMC from October 2000 through May 2023. Yearly totals for the number of persons with diabetes and foot ulcers at MEDVAMC were also obtained from the support service center. Annual patient population sizes and number of persons with foot ulcers were converted to monthly estimates using cubic spline interpolation. Rates were calculated as 12-month rolling averages. Trend lines were created with locally weighted running line smoothing that used a span α of 0.1.
We characterized the patient population using data from cohorts of veterans treated for foot ulcers and foot infections at MEDVAMC. To compare the contemporary veteran population with nonveteran inpatients treated for foot ulcers and foot infections at other hospitals, we created a 2:1 nonveteran to veteran cohort matched by sex and zip code, using publicly available hospital admission data from the Texas Department of Health and State Health Services. Veterans used for this cohort comparison are consistent with the 100 consecutive patients who underwent angiography for limb salvage in 2022.
This research was approved by the Baylor College of Medicine Institutional Review Board (protocol H-34858) and the MEDVAMC Research Committee (IRBNet protocol 15A12. HB). All analyses used deidentified data in the R programming language version 4.2.2 using RStudio version 2022.06.0 Build 421.
Program Description
MEDVAMC is a 350-bed teaching hospital located in central Houston. Its hospital system includes 11 outpatient clinics, ranging from 28 to 126 miles (eAppendix, Supplemental Figure A) from MEDVAMC. MEDVAMC provides vascular, orthopedic, and podiatric surgery services, as well as many other highly specialized services such as liver and heart transplants. The hospital’s risk-adjusted rates of operative morbidity and mortality (observed-to-expected ratios) are significantly lower than expected.
Despite this, the incidence rate of leg amputations at MEDVAMC in early 2011 was nearly 3-times higher than the VHA average. The inpatient management of veterans with infected foot ulcers was fragmented, with the general, orthopedic, and vascular surgery teams separately providing siloed care. Delays in treatment were common. There was much service- and practitioner-level practice heterogeneity. No diagnostic or treatment protocols were used, and standard treatment components were sporadically provided.
Patient Population
Compared to the matched non-VHA patient cohort (Supplemental Table 1), veterans treated at MEDVAMC for limb salvage are older. Nearly half (46%) identify as Black, which is associated with a 2-fold higher riskadjusted rate of leg amputations.4 MEDVAMC patients also have significantly higher rates of diabetes, chronic kidney disease, and systolic heart failure. About 22% travel > 40 miles for treatment at MEDVAMC, double that of the matched cohort (10.7%). Additionally, 35% currently smoke and 37% have moderate to severe peripheral artery disease (PAD).5
Program Design
In late 2011, the MEDVAMC vascular surgery team led limb salvage efforts by implementing a single team model, which involved assuming the primary role of managing foot ulcers for all veterans, both infected and uninfected (eAppendix, Supplemental Figure B). Consultations were directed to a dedicated limb salvage pager. The vascular team provided interdisciplinary limb salvage management across the spectrum of disease, including the surgical treatment of infection, assessment for PAD, open surgical operations and endovascular interventions to treat PAD, and foot reconstruction (debridement, minor or partial foot amputations, and skin grafting). This care was complemented by frequent consultation with the infectious disease, vascular medicine, podiatry, and geriatric wound care teams. This approach streamlined the delivery of consistent multidisciplinary care.
This collaborative effort aimed to develop ideal multidisciplinary care plans through research spanning the spectrum of the diabetic foot infection disease process (eAppendix, Supplemental Table 1). Some of the most impactful practices were: (1) a proclivity towards surgical treatment of foot infections, especially osteomyelitis5; (2) improved identification of PAD6,7; (3) early surgical closure of foot wounds following revascularization8,9; and (4) palliative wound care as an alternative to leg amputation in veterans who are not candidates for revascularization and limb salvage.10 Initally, the vascular surgery team held monthly multidisciplinary limb salvage meetings to coordinate patient management, identify ways to streamline care and avoid waste, discuss research findings, and review the 12-month rolling average of the MEDVAMC leg amputation incidence rate.
During the study period, the MEDVAMC vascular surgery team consisted of 2 to 5 board certified vascular or general surgeons, 2 or 3 nurse practitioners, and 3 vascular ultrasound technologists. Associated specialists included 2 podiatrists, 3 geriatricians with wound care certification, as well as additional infectious diseases, vascular medicine, orthopedics, and general surgery specialists.
Program Assessment
We noted a significant and sustained decrease in the MEDVAMC leg amputation rate after implementing multidisciplinary meetings and a single- team model from early 2012 through 2017 (Figure 2). The amputation incidence rate decreased steadily over the period from a maximum of 160 per 100,000 per year in February 2012 to a nadir of 66 per 100,000 per year in April 2017, an overall 60% decrease. Increases were noted in early 2018 after ceasing the single- team model, and in the summer of 2022, following periods of bed shortages after the onset of the COVID-19 pandemic. Tracking this metric allowed clinicians to make course corrections.

The decreased leg amputation rate at MEDVAMC does not seem to be mirroring national or regional trends. During this 10-year period, the VHA annualized amputation rate decreased minimally, from 58 to 54 per 100,000 (eAppendix Supplemental Figure C). Leg amputation incidence at non-VHA hospitals in Texas slightly increased over the same period.11
Value was also reflected in other metrics. MEDVAMC improved safety through a bundled strategy that reduced the risk-adjusted rate of surgical wound infections by 95%.12 MEDVAMC prioritized limb salvage when selecting patients for angiography and nearly eliminated using stent-grafts, cryopreserved allogeneic saphenous vein grafts, and expensive surgical and endovascular implants, which were identified as more expensive and less effective than other options (Figure 3).13-15 The MEDVAMC team achieved a > 90% patient trust rating on the Veterans Signals survey in fiscal years 2021 and 2022.

Challenges
A significant increase in the patient-physician ratio occurred 5 years into the program. In 2016, 2 vascular surgeons left MEDVAMC and a planned renovation of 1 of the 2 vascular surgery-assigned hybrid working facilities began even as the number of MEDVAMC patients with diabetes grew 120% (from 89,400 to 107,746 between 2010 and 2016), and the incidence rate of foot ulcers grew 300% (from 392 in 2010 to 1183 in 2016 per 100,000). The net result was a higher clinical workload among the remaining vascular surgeons with less operating room availability.
To stabilize surgeon retention, MEDVAMC reverted from the single team model back to inpatient care being distributed among general surgery, orthopedic surgery, and vascular surgery. After noting an increase in the leg amputation incidence rate, we adjusted the focus from multidisciplinary to interdisciplinary care (ie, majority of limb salvage clinical care can be provided by practitioners of any involved specialties). We worked to establish a local, written, interdisciplinary consensus on evaluating and managing veterans with nonhealing foot ulcers to mitigate the loss of a consolidated inpatient approach. Despite frequent staff turnover, ≥ 1 physician or surgeon from the core specialties of vascular surgery, podiatry, and infectious diseases remained throughout the study period.
The COVID-19 pandemic caused a shortage of hospital beds. This was followed by more bed shortages due to decreased nursing staff. Our health care system also had a period of restricted outpatient encounters early in the pandemic. During this time, we noted a delayed presentation of veterans with advanced infections and another increase in leg amputation incidence rate.
Like many health systems, MEDVAMC pivoted to telephone- and video-based outpatient encounters. Our team also used publicly available Texas hospitalization data to identify zip codes with particularly high leg amputation incidence rates, and > 3500 educational mailings to veterans categorized as moderate and high risk for leg amputation in these zip codes. These mailings provided information on recognizing foot ulcers and infections, emphasized timely evaluation, and named the MEDVAMC vascular surgery team as a point-of-contact. More recently, we have seen a further decrease in the MEDVAMC incidences of leg amputation to its lowest rate in > 20 years.
Discussion
A learning organization that directs its research based on clinical observations and informs its clinical care with research findings can produce palpable improvements in outcomes. Understanding the disease process and trying to better understand management across the entire range of this disease process has allowed our team to make consistent and systematic changes in care (Table). Consolidating inpatient care in a single team model seems to have been effective in reducing amputation rates among veterans with diabetes. The role the MEDVAMC vascular surgery team served for limb salvage patients may have been particularly beneficial because of the large impact untreated or unidentified PAD can have and because of the high prevalence of PAD among the limb salvage population seen at MEDVAMC. To be sustainable, though, a single-team model needs resources. A multiteam model can also be effective if the degree of multidisciplinary involvement for any given veteran is appropriate to the individual's clinical needs, teams are engaged and willing to contribute in a defined role within their specialty, and lines of communication remain open.

The primary challenge at MEDVAMC has been, and will continue to be, the retention of physicians and surgeons. MEDVAMC has excellent leadership and a collegial working environment, but better access to operating rooms for elective and time-sensitive operations, additional clinical staff support, and higher salary at non-VA positions have been the basis for many of physicians— especially surgeons—leaving MEDVAMC. Despite high staff turnover and a constant flow of resident and fellow trainees, MEDVAMC has been able to keep the clinical approach relatively consistent due to the use of written protocols and continuity of care as ≥ 1 physician or surgeon from each of the 4 main teams remained engaged with limb salvage throughout the entire period.
Going forward, we will work to ensure that all requirements of the 2022 Prevention of Amputation in Veterans Everywhere directive are incorporated into care.8 We plan to standardize MEDVAMC management algorithms further, both to streamline care and reduce the opportunity for disparities in treatment. More prophylactic podiatric procedures, surgical forms of offloading, and a shared multidisciplinary clinic space may also further help patients.
Conclusions
The introduction of multidisciplinary limb salvage at MEDVAMC has led to significant and sustained reductions in leg amputation incidence. These reductions do not seem dependent upon a specific team structure for inpatient care. To improve patient outcomes, efforts should focus on making improvements across the entire disease spectrum. For limb salvage, this includes primary prevention of foot ulcers, the treatment of foot infections, identification and management of PAD, surgical reconstruction/optimal wound healing, and care for patients who undergo leg amputation.
- Sanders LJ, Robbins JM, Edmonds ME. History of the team approach to amputation prevention: pioneers and milestones. J Am Podiatr Med Assoc. 2010;100(5):317- 334. doi:10.7547/1000317
- Sumpio BE, Armstrong DG, Lavery LA, Andros G. The role of interdisciplinary team approach in the management of the diabetic foot: a joint statement from the society for vascular surgery and the American podiatric medical association. J Am Podiatr Med Assoc. 2010;100(4):309-311. doi:10.7547/1000309
- About learning health systems. Agency for Healthcare Research and Quality. Published March 2019. Updated May 2019. Accessed October 9, 2024. https://www.ahrq.gov/learning-health-systems/about.html
- Barshes NR, Minc SD. Healthcare disparities in vascular surgery: a critical review. J Vasc Surg. 2021;74(2S):6S-14S.
- Barshes NR, Mindru C, Ashong C, Rodriguez-Barradas M, Trautner BW. Treatment failure and leg amputation among patients with foot osteomyelitis. Int J Low Extrem Wounds. 2016;15(4):303-312. doi:10.1177/1534734616661058
- Barshes NR, Flores E, Belkin M, Kougias P, Armstrong DG, Mills JL Sr. The accuracy and cost-effectiveness of strategies used to identify peripheral artery disease among patients with diabetic foot ulcers. J Vasc Surg. 2016;64(6):1682-1690.e3. doi:10.1016/j.jvs.2016.04.056 e1. doi:10.1016/j.jvs.2021.03.055
- Choi JC, Miranda J, Greenleaf E, et al. Lower-extremity pressure, staging, and grading thresholds to identify chronic limb-threatening ischemia. Vasc Med. 2023;28(1):45-53. doi:10.1177/1358863X221147945
- Barshes NR, Chambers JD, Cohen J, Belkin M; Model To Optimize Healthcare Value in Ischemic Extremities 1 (MOVIE) Study Collaborators. Cost-effectiveness in the contemporary management of critical limb ischemia with tissue loss. J Vasc Surg. 2012;56(4):1015-24.e1. doi:10.1016/j.jvs.2012.02.069
- Barshes NR, Bechara CF, Pisimisis G, Kougias P. Preliminary experiences with early primary closure of foot wounds after lower extremity revascularization. Ann Vasc Surg. 2014;28(1):48-52. doi:10.1016/j.avsg.2013.06.012
- Barshes NR, Gold B, Garcia A, Bechara CF, Pisimisis G, Kougias P. Minor amputation and palliative wound care as a strategy to avoid major amputation in patients with foot infections and severe peripheral arterial disease. Int J Low Extrem Wounds. 2014;13(3):211-219. doi:10.1177/1534734614543663
- Garcia M, Hernandez B, Ellington TG, et al. A lack of decline in major nontraumatic amputations in Texas: contemporary trends, risk factor associations, and impact of revascularization. Diabetes Care. 2019;42(6):1061-1066. doi:10.2337/dc19-0078
- Zamani N, Sharath SE, Vo E, Awad SS, Kougias P, Barshes NR. A multi-component strategy to decrease wound complications after open infra-inguinal re-vascularization. Surg Infect (Larchmt). 2018;19(1):87-94. doi:10.1089/sur.2017.193
- Barshes NR, Ozaki CK, Kougias P, Belkin M. A costeffectiveness analysis of infrainguinal bypass in the absence of great saphenous vein conduit. J Vasc Surg. 2013;57(6):1466-1470. doi:10.1016/j.jvs.2012.11.115
- Zamani N, Sharath S, Browder R, et al. PC158 longterm outcomes after endovascular stent placement for symptomatic, long-segment superficial femoral artery lesions. J Vasc Surg. 2017;65(6):182S-183S. doi:10.1016/j.jvs.2017.03.344
- Zamani N, Sharath SE, Browder RC, et al. Outcomes after endovascular stent placement for long-segment superficial femoral artery lesions. Ann Vasc Surg. 2021;71:298-307. doi:10.1016/j.avsg.2020.08.124
- Sanders LJ, Robbins JM, Edmonds ME. History of the team approach to amputation prevention: pioneers and milestones. J Am Podiatr Med Assoc. 2010;100(5):317- 334. doi:10.7547/1000317
- Sumpio BE, Armstrong DG, Lavery LA, Andros G. The role of interdisciplinary team approach in the management of the diabetic foot: a joint statement from the society for vascular surgery and the American podiatric medical association. J Am Podiatr Med Assoc. 2010;100(4):309-311. doi:10.7547/1000309
- About learning health systems. Agency for Healthcare Research and Quality. Published March 2019. Updated May 2019. Accessed October 9, 2024. https://www.ahrq.gov/learning-health-systems/about.html
- Barshes NR, Minc SD. Healthcare disparities in vascular surgery: a critical review. J Vasc Surg. 2021;74(2S):6S-14S.
- Barshes NR, Mindru C, Ashong C, Rodriguez-Barradas M, Trautner BW. Treatment failure and leg amputation among patients with foot osteomyelitis. Int J Low Extrem Wounds. 2016;15(4):303-312. doi:10.1177/1534734616661058
- Barshes NR, Flores E, Belkin M, Kougias P, Armstrong DG, Mills JL Sr. The accuracy and cost-effectiveness of strategies used to identify peripheral artery disease among patients with diabetic foot ulcers. J Vasc Surg. 2016;64(6):1682-1690.e3. doi:10.1016/j.jvs.2016.04.056 e1. doi:10.1016/j.jvs.2021.03.055
- Choi JC, Miranda J, Greenleaf E, et al. Lower-extremity pressure, staging, and grading thresholds to identify chronic limb-threatening ischemia. Vasc Med. 2023;28(1):45-53. doi:10.1177/1358863X221147945
- Barshes NR, Chambers JD, Cohen J, Belkin M; Model To Optimize Healthcare Value in Ischemic Extremities 1 (MOVIE) Study Collaborators. Cost-effectiveness in the contemporary management of critical limb ischemia with tissue loss. J Vasc Surg. 2012;56(4):1015-24.e1. doi:10.1016/j.jvs.2012.02.069
- Barshes NR, Bechara CF, Pisimisis G, Kougias P. Preliminary experiences with early primary closure of foot wounds after lower extremity revascularization. Ann Vasc Surg. 2014;28(1):48-52. doi:10.1016/j.avsg.2013.06.012
- Barshes NR, Gold B, Garcia A, Bechara CF, Pisimisis G, Kougias P. Minor amputation and palliative wound care as a strategy to avoid major amputation in patients with foot infections and severe peripheral arterial disease. Int J Low Extrem Wounds. 2014;13(3):211-219. doi:10.1177/1534734614543663
- Garcia M, Hernandez B, Ellington TG, et al. A lack of decline in major nontraumatic amputations in Texas: contemporary trends, risk factor associations, and impact of revascularization. Diabetes Care. 2019;42(6):1061-1066. doi:10.2337/dc19-0078
- Zamani N, Sharath SE, Vo E, Awad SS, Kougias P, Barshes NR. A multi-component strategy to decrease wound complications after open infra-inguinal re-vascularization. Surg Infect (Larchmt). 2018;19(1):87-94. doi:10.1089/sur.2017.193
- Barshes NR, Ozaki CK, Kougias P, Belkin M. A costeffectiveness analysis of infrainguinal bypass in the absence of great saphenous vein conduit. J Vasc Surg. 2013;57(6):1466-1470. doi:10.1016/j.jvs.2012.11.115
- Zamani N, Sharath S, Browder R, et al. PC158 longterm outcomes after endovascular stent placement for symptomatic, long-segment superficial femoral artery lesions. J Vasc Surg. 2017;65(6):182S-183S. doi:10.1016/j.jvs.2017.03.344
- Zamani N, Sharath SE, Browder RC, et al. Outcomes after endovascular stent placement for long-segment superficial femoral artery lesions. Ann Vasc Surg. 2021;71:298-307. doi:10.1016/j.avsg.2020.08.124
Eating Disorder Risk Factors and the Impact of Obesity in Patients With Psoriasis
Psoriasis is a chronic multisystemic inflammatory skin disease with a worldwide prevalence of 2% to 3%.1 Psoriasis can be accompanied by other conditions such as psoriatic arthritis, obesity, metabolic syndrome, diabetes mellitus, hypertension, dyslipidemia, atherosclerotic disease, inflammatory bowel disease, and anxiety/depression. It is important to manage comorbidities of psoriasis in addition to treating the cutaneous manifestations of the disease.1
Obesity is a major public health concern worldwide. Numerous observational and epidemiologic studies have reported a high prevalence of obesity among patients with psoriasis.2 Current evidence indicates that obesity may initiate or worsen psoriasis; furthermore, it is important to note that obesity may negatively impact the effectiveness of psoriasis-specific treatments or increase the incidence of adverse effects. Therefore, managing obesity is crucial in the treatment of psoriasis.3 Numerous studies have investigated the association between psoriasis and obesity, and they commonly conclude that both conditions share the same genetic metabolic pathways.2-4 However, it is important to consider environmental factors such as dietary habits, smoking, alcohol consumption, and a sedentary lifestyle—all of which are associated with psoriasis and also can contribute to the development of obesity.5 Because of the effects of obesity in psoriasis patients, factors that impact the development of obesity have become a popular research topic.
Eating disorders (EDs) are a crucial risk factor for both developing and maintaining obesity. In particular, two EDs that are associated with obesity include binge eating disorder and bulimia nervosa.6 According to the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition,7 binge eating disorder can be diagnosed when a patient has at least 1 episode of binge eating per week over a 3-month period. Bulimia nervosa can be diagnosed when a patient is excessively concerned with their body weight and shape and engages in behaviors to prevent weight gain (eg, forced vomiting, excessive use of laxatives).7 Psychiatrists who specialize in EDs make diagnoses based on these criteria. In daily practice, there are several quick and simple questionnaires available to screen for EDs that can be used by nonpsychiatrist physicians, including the commonly used 26-item Eating Attitudes Test (EAT-26).8 The EAT-26 has been used to screen for EDs in patients with inflammatory disorders.9
The aim of this study was to screen for EDs in patients with psoriasis to identify potential risk factors for development of obesity.
Materials and Methods
This study included patients with psoriasis who were screened for EDs at a tertiary dermatology clinic in Turkey between January 2021 and December 2023. This study was approved by the local ethics committee and was in accordance with the Declaration of Helsinki (decision number E-93471371-514.99-225000079).
Study Design and Patient Inclusion Criteria—This quantitative cross-sectional study utilized EAT-26, Dermatology Life Quality Index (DLQI), Attitude Scale for Healthy Nutrition (ASHN), and Depression Anxiety Stress Scale-21 (DASS-21) scores. All the questionnaire scales used in the study were adapted and validated in Turkey.8,10-12 The inclusion criteria consisted of being older than 18 years of age, being literate, having psoriasis for at least 1 year that was not treated topically or systemically, and having no psychiatric diseases outside an ED. The questionnaires were presented in written format following the clinical examination. Literacy was an inclusion criterion in this study due to the absence of auxiliary health personnel.
Study Variables—The study variables included age, sex, marital status (single/divorced or married), education status (primary/secondary school or high school/university), employment status (employed or unemployed/retired), body mass index (BMI), smoking status, alcohol-consumption status, Psoriasis Area Severity Index score, presence of nail psoriasis and psoriatic arthritis, duration of psoriasis, family history of psoriasis, EAT-26 score, ASHN score, DLQI score, and DASS-21 score. Body mass index was calculated by taking a participant’s weight in kilograms and dividing it by their height in meters squared. The BMI values were classified into 3 categories: normal (18.5–24.9 kg/m2), overweight (25.0–29.9 kg/m2), and obese (≥30 kg/m2).13
Questionnaires—The EAT-26 questionnaire includes 26 questions that are used to detect EDs. Responses to each question include Likert-type answer options (ie, “always,” “usually,” “often,” “sometimes,” “rarely,” and “never.”) Patients with scores of 20 points or higher (range, 0–78) are classified as high risk for EDs.8 In our study, EAT-26 scores were grouped into 2 categories: patients scoring less than 20 points and those scoring 20 points or higher.
The DLQI questionnaire includes 10 questions to measure dermatologic symptoms and qualiy of life. Responses to each question include Likert-type answer options (ie, “not at all,” “a little,” “a lot,” or “very much.”) On the DLQI scale, the higher the score, the lower the quality of life (score range, 0–30).10
The ASHN questionnaire includes 21 questions that measure attitudes toward healthy nutrition with 5 possible answer options (“strongly disagree,” “disagree,” “undecided,” “agree,” and “strongly agree”). On this scale, higher scores indicate the participant is more knowledgeable about healthy nutrition (score range, 0–78).11
The DASS-21 questionnaire includes 21 questions that measure the severity of a range of symptoms common to depression, anxiety, and stress. Responses include Likert-type answer options (eg, “never,” “sometimes,” “often,” and “almost always.”) On this scale, a higher score (range of 0–21 for each) indicates higher levels of depression, anxiety, and stress.12
Statistical Analysis—Descriptive statistics were analyzed using SPSS software version 22.0 (IBM). The Shapiro-Wilk test was applied to determine whether the data were normally distributed. For categorical variables, frequency differences among groups were compared using the Pearson χ2 test. A t test was used to compare the means of 2 independent groups with a normal distribution. One-way analysis of variance and Tukey Honest Significant Difference post hoc analysis were used to test whether there was a statistically significant difference among the normally distributed means of independent groups. Pearson correlation analysis was used to determine whether there was a linear relationship between 2 numeric measurements and, if so, to determine the direction and severity of this relationship. P<.05 indicated statistical significance in this study.
Results
Study Participant Demographics—This study included 82 participants with a mean age of 44.3 years; 52.4% (43/82) were female, and 85.4% (70/82) were married. The questionnaire took an average of 4.2 minutes for participants to complete. A total of 57.3% (47/82) of patients had completed primary/secondary education and 59.8% (49/82) were employed. The mean BMI was 28.1 kg/m2. According to the BMI classification, 26.8% (22/82) participants had a normal weight, 36.6% (30/82) were overweight, and 43.9% (36/82) were obese. A total of 48.8% (40/82) of participants smoked, and 4.9% (4/82) consumed alcohol. The mean Psoriasis Area and Severity Index score was 5.4. A total of 54.9% (45/82) of participants had nail psoriasis, and 24.4% (20/82) had psoriatic arthritis. The mean duration of psoriasis was 153 months. A total of 29.3% (24/82) of participants had a positive family history of psoriasis. The mean EAT-26 score was 11.1. A total of 12.2% (10/82) of participants had an EAT-26 score of 20 points or higher and were considered at high risk for an ED. The mean ASHN score was 72.9; the mean DLQI score was 5.5; and on the DASS-21 scale, mean scores for depression, anxiety, and stress were 6.3, 8.7, and 10.0, respectively (Table).
Comparative Evaluation of the BMI Groups—The only statistically significant differences among the 3 BMI groups were related to marital status, EAT-26 score, and anxiety and stress scores (P=.02, <.01, <.01, and <.01, respectively)(eTable 1). The number of single/divorced participants in the overweight group was significantly (P=.02) greater than in the normal weight group. The mean EAT-26 score for the normal weight group was significantly (P<.01) lower than for the overweight and obese groups; there was no significant difference in mean EAT-26 scores between the overweight and obese groups. The mean anxiety score was significantly (P<.01) lower in the normal weight group compared with the overweight and obese groups. There was no significant difference between the overweight and obese groups according to the mean depression score. The mean stress and anxiety scores were significantly (P<.01) lower in the normal weight group than in the overweight and obese groups. There was no significant difference between the overweight and obese groups according to the mean anxiety score.
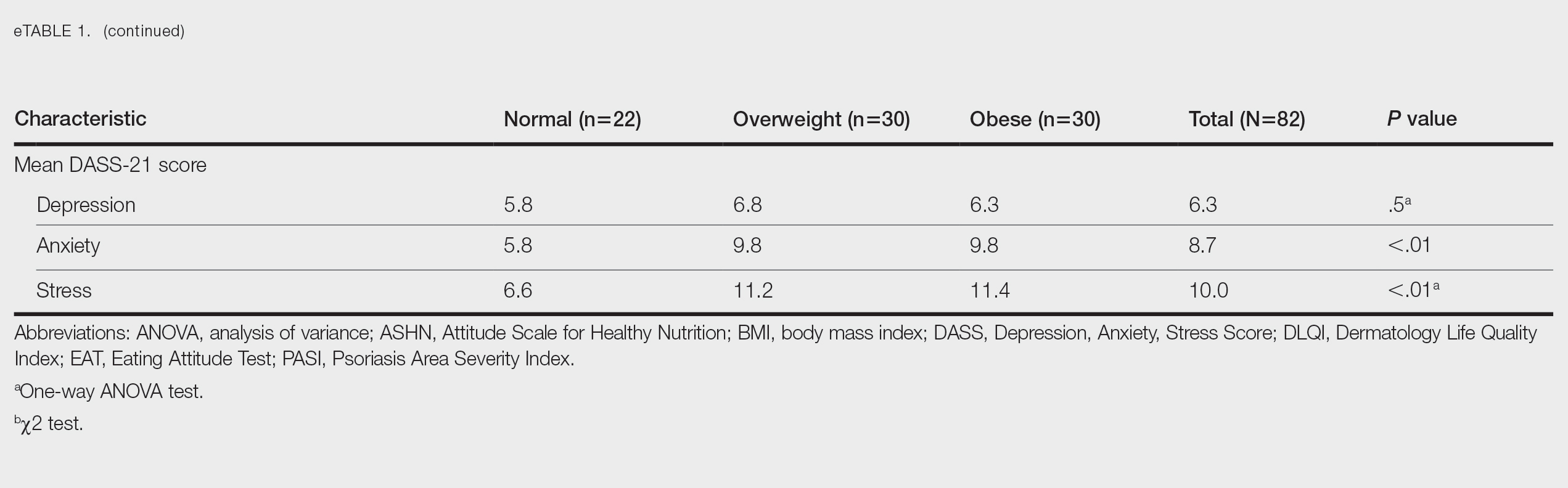
Comparative Evaluation of the EAT-26 Scores—There were statistically significant differences among the EAT-26 scores related to sex; BMI; and depression, anxiety, and stress scores (P=.04, .02, <.01, <.01, and <.01, respectively). The number of females in the group with a score of 20 points or higher was significantly (P=.04) less than that in the group scoring less than 20 points. The mean BMI in the group with a score of 20 points or higher was significantly (P=.02) greater than in group scoring less than 20 points. The mean depression, anxiety, and stress scores of the group scoring 20 points or higher were significantly (P<.01 for all) greater than in the group scoring less than 20 points (eTable 2).
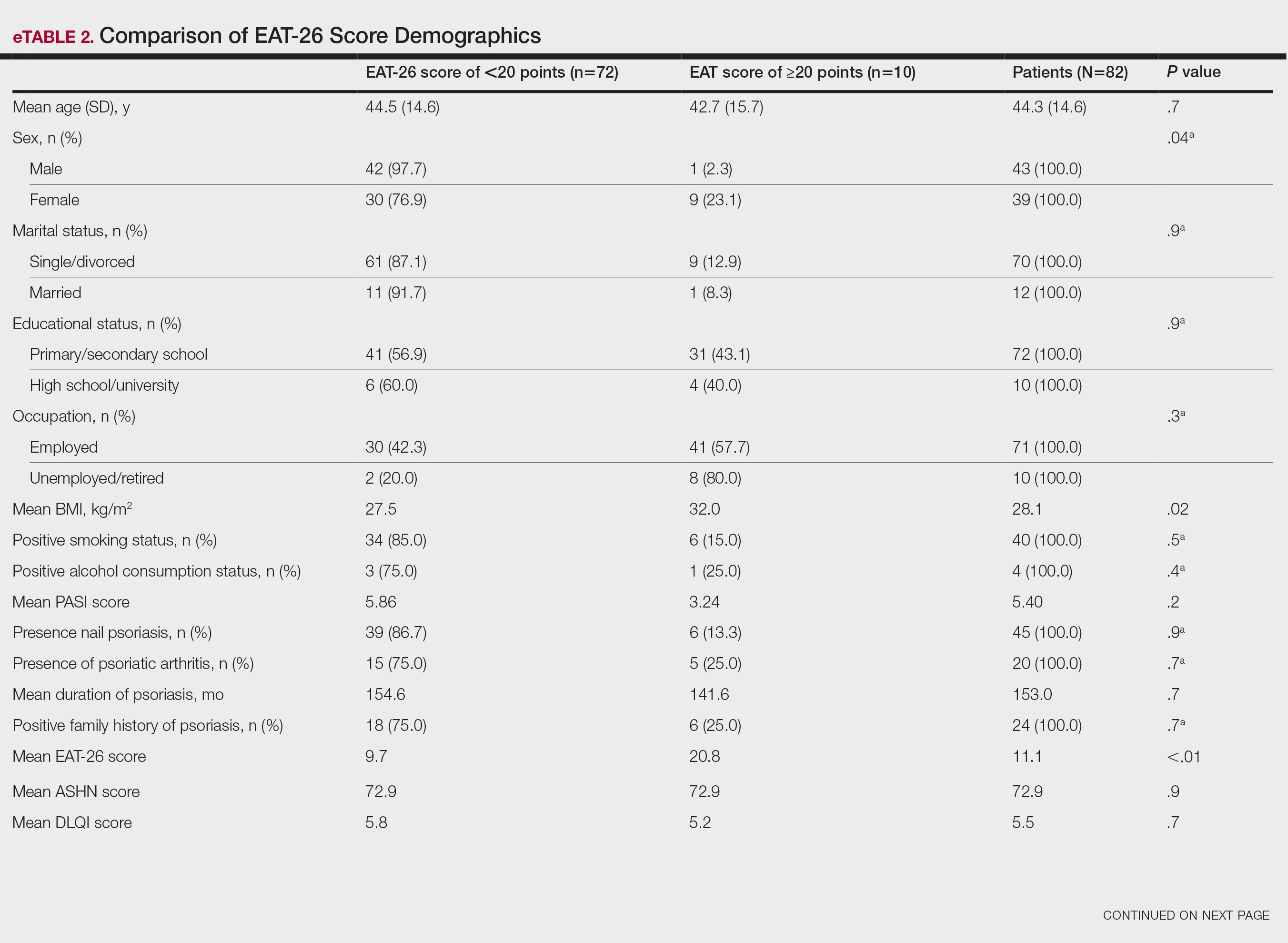
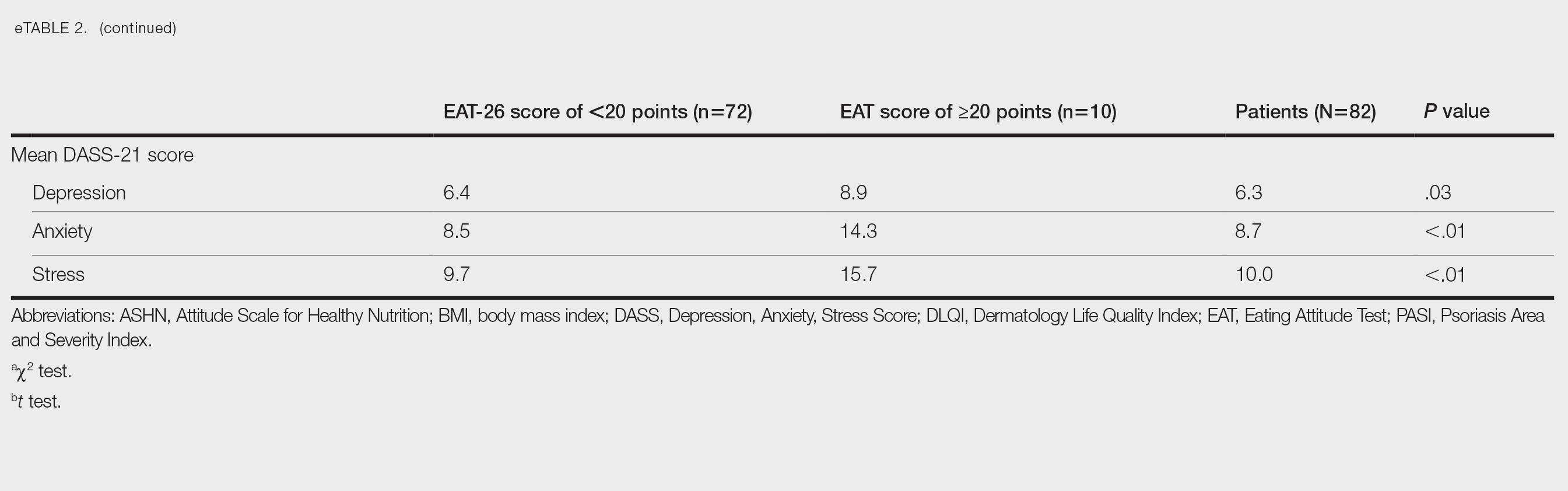
Correlation Analysis of the Study Variables—The EAT-26 scores were positively correlated with BMI, anxiety, depression, and stress (P<.01 for all)(eTable 3).
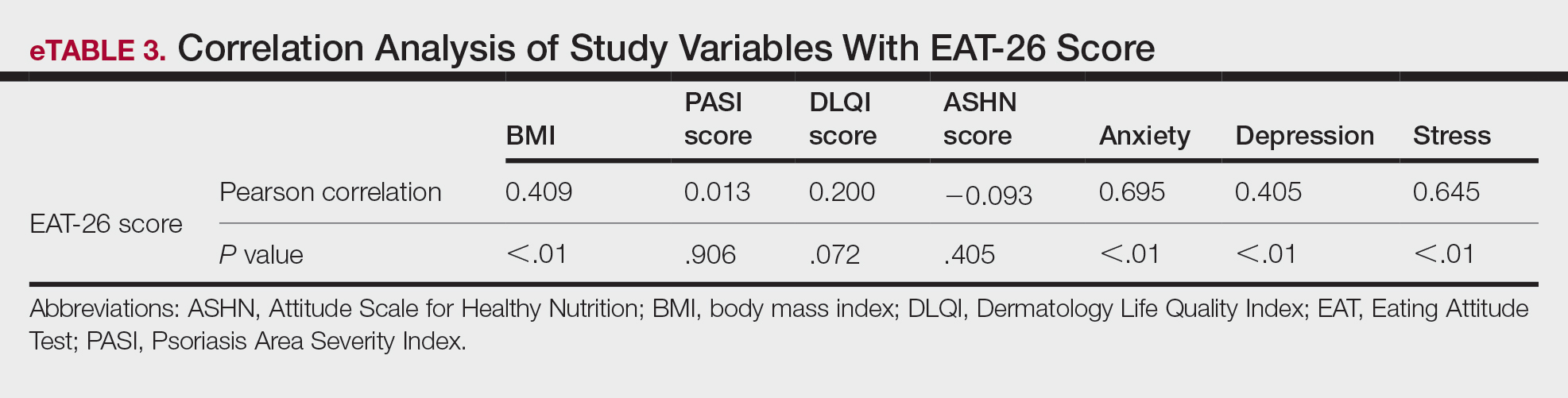
Comment
Eating disorders are psychiatric conditions that require a multidisciplinary approach. Nonpsychiatric medical departments may be involved due to the severe consequences (eg, various skin changes14) of these disorders. Psoriasis is not known to be directly affected by the presence of an ED; however, it is possible that EDs could indirectly affect patients with psoriasis by influencing obesity. Therefore, this study aimed to examine the relationship between ED risk factors and obesity in this population.
The relationship between psoriasis and obesity has been a popular research topic in dermatology since the 1990s.15 Epidemiologic and observational studies have reported that patients with psoriasis are more likely to be overweight or have obesity, which is an independent risk factor for psoriasis.3,16 However, the causal relationship between psoriasis and obesity remains unclear. In a comprehensive review, Barros et al17 emphasized the causal relationship between obesity and psoriasis under several headings. Firstly, a higher BMI increases the risk for psoriasis by promoting cytokine release and immune system dysregulation. Secondly, a Western diet (eg, processed foods and fast food) triggers obesity and psoriasis by increasing adipose tissue. Thirdly, the alteration of the skin and gut microbiota triggers chronic inflammation as a result of bacterial translocation in patients with obesity. Fourthly, a high-fat diet and palmitic acid disrupt the intestinal integrity of the gut and increase the risk for psoriasis and obesity by triggering chronic inflammation of bacterial fragments that pass into the blood. Finally, the decrease in the amount of adiponectin and the increase in the amount of leptin in patients with obesity may cause psoriasis by increasing proinflammatory cytokines, which are similar to those involved in the pathogenesis of psoriasis.17 Additionally, psoriatic inflammation can cause insulin resistance and metabolic dysfunction, leading to obesity.18 The relationship between psoriasis and obesity cannot be solely explained by metabolic pathways. Smoking, alcohol consumption, and a sedentary lifestyle all are associated with psoriasis and also can contribute to obesity.5 Our study revealed no significant difference in smoking or alcohol consumption between the normal weight and overweight/obesity groups. Based on our data, we determined that smoking and alcohol consumption did not affect obesity in our patients with psoriasis.
Observational and epidemiologic studies have shown that patients with psoriasis experience increased rates of depression, anxiety, and stress.19 In studies of pathogenesis, a connection between depression and psoriatic inflammation has been established.20 It is known that inflammatory cytokines similar to those in psoriasis are involved in the development of obesity.18 In addition, depression and anxiety can lead to binge eating, unhealthy food choices, and a more sedentary lifestyle.5 All of these variables may contribute to the associations between depression and anxiety with psoriasis and obesity. Zafiriou et al21 conducted a study to investigate the relationship between psoriasis, obesity, and depression through inflammatory pathways with a focus on the importance of IL-17. Data showing that IL-17–producing Th17-cell subgroups play a considerable role in the development of obesity and depression prompted the authors to suggest that psoriasis, obesity, and anxiety/depression may be interconnected manifestations of immune dysregulation, potentially linked to IL-17 and its associated cells.21 Mrowietz et al22 also suggested that metabolic inflammation may contribute to obesity and depression in patients with psoriasis and highlighted the importance of several cytokines, including tumor necrosis factor α, IL-6, IL-8, IL-17, and IL-23. Our study revealed no significant differences in depression scores between BMI groups. Another meta-analysis reported conflicting findings on the incidence of depression in obese patients with psoriasis.23 Some of the studies had a small number of participants. Compared to depression, anxiety has received less attention in studies of patients with obesity with psoriasis. However, these studies have shown a positive correlation between anxiety scores and BMI in patients with psoriasis.24,25 In our study, similar to the findings of previous studies, overweight patients and those with obesitywho have psoriasis had significantly (P<.01) greater anxiety and stress scores than did normal weight patients with psoriasis.
Obesity should be assessed in patients with psoriasis via a biopsychosocial approach that takes into account genetic, behavioral, and environmental factors.26 Eating disorders are considered to be one of the factors contributing to obesity. Numerous studies in the literature have demonstrated a greater incidence of EDs in patients with obesity vs those without obesity.5,6,27 Obesity and EDs have a bidirectional relationship: individuals with obesity are at risk for EDs due to body dissatisfaction, dieting habits, and depressive states. Conversely, poor eating behaviors in individuals with a normal weight can lead to obesity.28
There are few studies in the literature exploring the relationship between psoriasis and EDs. Crosta et al29 demonstrated that patients with psoriasis had impaired results on ED screening tests and that these scores deteriorated further as BMI increased. Moreover, Altunay et al30 demonstrated that patients with psoriasis and metabolic syndrome had higher scores on the ED screening test. In this study, patients with higher scores also exhibited high levels of anxiety.30 In our study, similar to the findings of previous studies, patients with psoriasis who were overweight or had obesity had significantly (P<.01) greater EAT-26 scores than those in the normal weight group. Patients with high EAT-26 scores also exhibited elevated levels of depression, anxiety, and stress. Additionally, EAT-26 scores were positively correlated with BMI, anxiety, depression, and stress scores. Our study as well as other studies in the literature indicate that additional research is needed to determine the associations between EDs and obesity in psoriasis.
Conclusion
Managing obesity is crucial for patients with psoriasis. This study showed that EAT-26 scores were higher in patients with psoriasis who were overweight or had obesity than in those who were normal weight. Participants with high EAT-26 scores (≥20 points) were more likely to be female and have higher anxiety and stress scores. In addition, EAT-26 scores were positively correlated with BMI as well as depression, anxiety, and stress scores. Eating disorders may contribute to the development of obesity in patients with psoriasis. Although our study was limited by a small sample size, the results suggest that there is a need for large-scale multicenter studies to investigate the relationship between psoriasis and EDs.
- Kalkan G. Comorbidities in psoriasis: the recognition of psoriasis as a systemic disease and current management. Turkderm-Turk Arch Dermatol Venereol. 2017;51:71-77.
- Armstrong AW, Harskamp CT, Armstrong EJ. The association between psoriasis and obesity: a systematic review and meta-analysis of observational studies. Nutr Diabetes. 2012;2:E54.
- Jensen P, Skov L. Psoriasis and obesity. Dermatology. 2016;232:633-639.
- Mirghani H, Altemani AT, Altemani ST, et al. The cross talk between psoriasis, obesity, and dyslipidemia: a meta-analysis. Cureus. 2023;15:e49253.
- Roehring M, Mashep MR, White MA, et al. The metabolic syndrome and behavioral correlates in obese patients with binge disorders. Obesity. 2009;17:481-486.
- da Luz FQ, Hay P, Touyz S, et al. Obesity with comorbid eating disorders: associated health risks and treatment approaches. Nutrients. 2018;10:829.
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition. American Psychiatric Association; 2013.
- Ergüney Okumus¸ FE, Sertel Berk HÖ. The psychometric properties of the Eating Attitudes Test short form (EAT-26) in a college sample. Stud Psychol. 2020;40:57-78.
- Stoleru G, Leopold A, Auerbach A, et al. Female gender, dissatisfaction with weight, and number of IBD related surgeries as independent risk factors for eating disorders among patients with inflammatory bowel diseases. BMC Gastroenterol. 2022;22:438.
- Öztürkcan S, Ermertcan AT, Eser E, et al. Cross validation of the Turkish version of dermatology life quality index. Int J Dermatol. 2006;45:1300-1307.
- Demir GT, Ciciog˘lu HI˙. Attitude scale for healthy nutrition (ASHN): validity and reliability study. Gaziantep Univ J Sport Sci. 2019;4:256-274.
- Yılmaz O, Boz H, Arslan A. The validity and reliability of depression stress and anxiety scale (DASS 21) Turkish short form. Res Financial Econ Soc Stud. 2017;2:78-91.
- Nuttall FQ. Body mass index: obesity, BMI, and health: a critical review. Nutr Today. 2015;50:117-128.
- Strumia R, Manzata E, Gualandi M. Is there a role for dermatologists in eating disorders? Expert Rev Dermatol. 2017; 2:109-112.
- Henseler T, Christophers E. Disease concomitance in psoriasis. J Am Acad Dermatol. 1995;32:982-986.
- Naldi L, Addis A, Chimenti S, et al. Impact of body mass index and obesity on clinical response to systemic treatment for psoriasis. evidence from the Psocare project. Dermatology. 2008;217:365-373.
- Barros G, Duran P, Vera I, et al. Exploring the links between obesity and psoriasis: a comprehensive review. Int J Mol Sci. 2022;23:7499.
- Hao Y, Zhu YJ, Zou S, et al. Metabolic syndrome and psoriasis: mechanisms and future directions. Front Immunol. 2021;12:711060.
- Jing D, Xiao H, Shen M, et al. Association of psoriasis with anxiety and depression: a case–control study in Chinese patients. Front Med (Lausanne). 2021;8:771645.
- Sahi FM, Masood A, Danawar NA, et al. Association between psoriasis and depression: a traditional review. Cureus. 2020;12:E9708.
- Zafiriou E, Daponte AI, Siokas V, et al. Depression and obesity in patients with psoriasis and psoriatic arthritis: is IL-17–mediated immune dysregulation the connecting link? Front Immunol. 2021;12:699848.
- Mrowietz U, Sümbül M, Gerdes S. Depression, a major comorbidity of psoriatic disease, is caused by metabolic inflammation. J Eur Acad Dermatol Venereol. 2023;37:1731-1738.
- Pavlova NT, Kioskli K, Smith C, et al. Psychosocial aspects of obesity in adults with psoriasis: a systematic review. Skin Health Dis. 2021;1:E33.
- Innamorati M, Quinto RM, Imperatori C, et al. Health-related quality of life and its association with alexithymia and difficulties in emotion regulation in patients with psoriasis. Compr Psychiatry. 2016;70:200-208.
- Tabolli S, Naldi L, Pagliarello C, et al. Evaluation of the impact of writing exercises interventions on quality of life in patients with psoriasis undergoing systemic treatments. Br J Dermatol. 2012;167:1254‐1264.
- Albuquerque D, Nóbrega C, Manco L, et al. The contribution of genetics and environment to obesity. Br Med Bull. 2017;123:159‐173.
- Balantekin KN, Grammer AC, Fitzsimmons-Craft EE, et al. Overweight and obesity are associated with increased eating disorder correlates and general psychopathology in university women with eating disorders. Eat Behav. 2021;41:101482.
- Jebeile H, Lister NB, Baur LA, et al. Eating disorder risk in adolescents with obesity. Obes Rev. 2021;22:E13173.
- Crosta ML, Caldarola G, Fraietta S, et al. Psychopathology and eating disorders in patients with psoriasis. G Ital Dermatol Venereol. 2014;149:355-361.
- Altunay I, Demirci GT, Ates B, et al. Do eating disorders accompany metabolic syndrome in psoriasis patients? results of a preliminary study. Clin Cosmet Investig Dermatol. 2011;4:139-143.
Psoriasis is a chronic multisystemic inflammatory skin disease with a worldwide prevalence of 2% to 3%.1 Psoriasis can be accompanied by other conditions such as psoriatic arthritis, obesity, metabolic syndrome, diabetes mellitus, hypertension, dyslipidemia, atherosclerotic disease, inflammatory bowel disease, and anxiety/depression. It is important to manage comorbidities of psoriasis in addition to treating the cutaneous manifestations of the disease.1
Obesity is a major public health concern worldwide. Numerous observational and epidemiologic studies have reported a high prevalence of obesity among patients with psoriasis.2 Current evidence indicates that obesity may initiate or worsen psoriasis; furthermore, it is important to note that obesity may negatively impact the effectiveness of psoriasis-specific treatments or increase the incidence of adverse effects. Therefore, managing obesity is crucial in the treatment of psoriasis.3 Numerous studies have investigated the association between psoriasis and obesity, and they commonly conclude that both conditions share the same genetic metabolic pathways.2-4 However, it is important to consider environmental factors such as dietary habits, smoking, alcohol consumption, and a sedentary lifestyle—all of which are associated with psoriasis and also can contribute to the development of obesity.5 Because of the effects of obesity in psoriasis patients, factors that impact the development of obesity have become a popular research topic.
Eating disorders (EDs) are a crucial risk factor for both developing and maintaining obesity. In particular, two EDs that are associated with obesity include binge eating disorder and bulimia nervosa.6 According to the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition,7 binge eating disorder can be diagnosed when a patient has at least 1 episode of binge eating per week over a 3-month period. Bulimia nervosa can be diagnosed when a patient is excessively concerned with their body weight and shape and engages in behaviors to prevent weight gain (eg, forced vomiting, excessive use of laxatives).7 Psychiatrists who specialize in EDs make diagnoses based on these criteria. In daily practice, there are several quick and simple questionnaires available to screen for EDs that can be used by nonpsychiatrist physicians, including the commonly used 26-item Eating Attitudes Test (EAT-26).8 The EAT-26 has been used to screen for EDs in patients with inflammatory disorders.9
The aim of this study was to screen for EDs in patients with psoriasis to identify potential risk factors for development of obesity.
Materials and Methods
This study included patients with psoriasis who were screened for EDs at a tertiary dermatology clinic in Turkey between January 2021 and December 2023. This study was approved by the local ethics committee and was in accordance with the Declaration of Helsinki (decision number E-93471371-514.99-225000079).
Study Design and Patient Inclusion Criteria—This quantitative cross-sectional study utilized EAT-26, Dermatology Life Quality Index (DLQI), Attitude Scale for Healthy Nutrition (ASHN), and Depression Anxiety Stress Scale-21 (DASS-21) scores. All the questionnaire scales used in the study were adapted and validated in Turkey.8,10-12 The inclusion criteria consisted of being older than 18 years of age, being literate, having psoriasis for at least 1 year that was not treated topically or systemically, and having no psychiatric diseases outside an ED. The questionnaires were presented in written format following the clinical examination. Literacy was an inclusion criterion in this study due to the absence of auxiliary health personnel.
Study Variables—The study variables included age, sex, marital status (single/divorced or married), education status (primary/secondary school or high school/university), employment status (employed or unemployed/retired), body mass index (BMI), smoking status, alcohol-consumption status, Psoriasis Area Severity Index score, presence of nail psoriasis and psoriatic arthritis, duration of psoriasis, family history of psoriasis, EAT-26 score, ASHN score, DLQI score, and DASS-21 score. Body mass index was calculated by taking a participant’s weight in kilograms and dividing it by their height in meters squared. The BMI values were classified into 3 categories: normal (18.5–24.9 kg/m2), overweight (25.0–29.9 kg/m2), and obese (≥30 kg/m2).13
Questionnaires—The EAT-26 questionnaire includes 26 questions that are used to detect EDs. Responses to each question include Likert-type answer options (ie, “always,” “usually,” “often,” “sometimes,” “rarely,” and “never.”) Patients with scores of 20 points or higher (range, 0–78) are classified as high risk for EDs.8 In our study, EAT-26 scores were grouped into 2 categories: patients scoring less than 20 points and those scoring 20 points or higher.
The DLQI questionnaire includes 10 questions to measure dermatologic symptoms and qualiy of life. Responses to each question include Likert-type answer options (ie, “not at all,” “a little,” “a lot,” or “very much.”) On the DLQI scale, the higher the score, the lower the quality of life (score range, 0–30).10
The ASHN questionnaire includes 21 questions that measure attitudes toward healthy nutrition with 5 possible answer options (“strongly disagree,” “disagree,” “undecided,” “agree,” and “strongly agree”). On this scale, higher scores indicate the participant is more knowledgeable about healthy nutrition (score range, 0–78).11
The DASS-21 questionnaire includes 21 questions that measure the severity of a range of symptoms common to depression, anxiety, and stress. Responses include Likert-type answer options (eg, “never,” “sometimes,” “often,” and “almost always.”) On this scale, a higher score (range of 0–21 for each) indicates higher levels of depression, anxiety, and stress.12
Statistical Analysis—Descriptive statistics were analyzed using SPSS software version 22.0 (IBM). The Shapiro-Wilk test was applied to determine whether the data were normally distributed. For categorical variables, frequency differences among groups were compared using the Pearson χ2 test. A t test was used to compare the means of 2 independent groups with a normal distribution. One-way analysis of variance and Tukey Honest Significant Difference post hoc analysis were used to test whether there was a statistically significant difference among the normally distributed means of independent groups. Pearson correlation analysis was used to determine whether there was a linear relationship between 2 numeric measurements and, if so, to determine the direction and severity of this relationship. P<.05 indicated statistical significance in this study.
Results
Study Participant Demographics—This study included 82 participants with a mean age of 44.3 years; 52.4% (43/82) were female, and 85.4% (70/82) were married. The questionnaire took an average of 4.2 minutes for participants to complete. A total of 57.3% (47/82) of patients had completed primary/secondary education and 59.8% (49/82) were employed. The mean BMI was 28.1 kg/m2. According to the BMI classification, 26.8% (22/82) participants had a normal weight, 36.6% (30/82) were overweight, and 43.9% (36/82) were obese. A total of 48.8% (40/82) of participants smoked, and 4.9% (4/82) consumed alcohol. The mean Psoriasis Area and Severity Index score was 5.4. A total of 54.9% (45/82) of participants had nail psoriasis, and 24.4% (20/82) had psoriatic arthritis. The mean duration of psoriasis was 153 months. A total of 29.3% (24/82) of participants had a positive family history of psoriasis. The mean EAT-26 score was 11.1. A total of 12.2% (10/82) of participants had an EAT-26 score of 20 points or higher and were considered at high risk for an ED. The mean ASHN score was 72.9; the mean DLQI score was 5.5; and on the DASS-21 scale, mean scores for depression, anxiety, and stress were 6.3, 8.7, and 10.0, respectively (Table).

Comparative Evaluation of the BMI Groups—The only statistically significant differences among the 3 BMI groups were related to marital status, EAT-26 score, and anxiety and stress scores (P=.02, <.01, <.01, and <.01, respectively)(eTable 1). The number of single/divorced participants in the overweight group was significantly (P=.02) greater than in the normal weight group. The mean EAT-26 score for the normal weight group was significantly (P<.01) lower than for the overweight and obese groups; there was no significant difference in mean EAT-26 scores between the overweight and obese groups. The mean anxiety score was significantly (P<.01) lower in the normal weight group compared with the overweight and obese groups. There was no significant difference between the overweight and obese groups according to the mean depression score. The mean stress and anxiety scores were significantly (P<.01) lower in the normal weight group than in the overweight and obese groups. There was no significant difference between the overweight and obese groups according to the mean anxiety score.


Comparative Evaluation of the EAT-26 Scores—There were statistically significant differences among the EAT-26 scores related to sex; BMI; and depression, anxiety, and stress scores (P=.04, .02, <.01, <.01, and <.01, respectively). The number of females in the group with a score of 20 points or higher was significantly (P=.04) less than that in the group scoring less than 20 points. The mean BMI in the group with a score of 20 points or higher was significantly (P=.02) greater than in group scoring less than 20 points. The mean depression, anxiety, and stress scores of the group scoring 20 points or higher were significantly (P<.01 for all) greater than in the group scoring less than 20 points (eTable 2).


Correlation Analysis of the Study Variables—The EAT-26 scores were positively correlated with BMI, anxiety, depression, and stress (P<.01 for all)(eTable 3).

Comment
Eating disorders are psychiatric conditions that require a multidisciplinary approach. Nonpsychiatric medical departments may be involved due to the severe consequences (eg, various skin changes14) of these disorders. Psoriasis is not known to be directly affected by the presence of an ED; however, it is possible that EDs could indirectly affect patients with psoriasis by influencing obesity. Therefore, this study aimed to examine the relationship between ED risk factors and obesity in this population.
The relationship between psoriasis and obesity has been a popular research topic in dermatology since the 1990s.15 Epidemiologic and observational studies have reported that patients with psoriasis are more likely to be overweight or have obesity, which is an independent risk factor for psoriasis.3,16 However, the causal relationship between psoriasis and obesity remains unclear. In a comprehensive review, Barros et al17 emphasized the causal relationship between obesity and psoriasis under several headings. Firstly, a higher BMI increases the risk for psoriasis by promoting cytokine release and immune system dysregulation. Secondly, a Western diet (eg, processed foods and fast food) triggers obesity and psoriasis by increasing adipose tissue. Thirdly, the alteration of the skin and gut microbiota triggers chronic inflammation as a result of bacterial translocation in patients with obesity. Fourthly, a high-fat diet and palmitic acid disrupt the intestinal integrity of the gut and increase the risk for psoriasis and obesity by triggering chronic inflammation of bacterial fragments that pass into the blood. Finally, the decrease in the amount of adiponectin and the increase in the amount of leptin in patients with obesity may cause psoriasis by increasing proinflammatory cytokines, which are similar to those involved in the pathogenesis of psoriasis.17 Additionally, psoriatic inflammation can cause insulin resistance and metabolic dysfunction, leading to obesity.18 The relationship between psoriasis and obesity cannot be solely explained by metabolic pathways. Smoking, alcohol consumption, and a sedentary lifestyle all are associated with psoriasis and also can contribute to obesity.5 Our study revealed no significant difference in smoking or alcohol consumption between the normal weight and overweight/obesity groups. Based on our data, we determined that smoking and alcohol consumption did not affect obesity in our patients with psoriasis.
Observational and epidemiologic studies have shown that patients with psoriasis experience increased rates of depression, anxiety, and stress.19 In studies of pathogenesis, a connection between depression and psoriatic inflammation has been established.20 It is known that inflammatory cytokines similar to those in psoriasis are involved in the development of obesity.18 In addition, depression and anxiety can lead to binge eating, unhealthy food choices, and a more sedentary lifestyle.5 All of these variables may contribute to the associations between depression and anxiety with psoriasis and obesity. Zafiriou et al21 conducted a study to investigate the relationship between psoriasis, obesity, and depression through inflammatory pathways with a focus on the importance of IL-17. Data showing that IL-17–producing Th17-cell subgroups play a considerable role in the development of obesity and depression prompted the authors to suggest that psoriasis, obesity, and anxiety/depression may be interconnected manifestations of immune dysregulation, potentially linked to IL-17 and its associated cells.21 Mrowietz et al22 also suggested that metabolic inflammation may contribute to obesity and depression in patients with psoriasis and highlighted the importance of several cytokines, including tumor necrosis factor α, IL-6, IL-8, IL-17, and IL-23. Our study revealed no significant differences in depression scores between BMI groups. Another meta-analysis reported conflicting findings on the incidence of depression in obese patients with psoriasis.23 Some of the studies had a small number of participants. Compared to depression, anxiety has received less attention in studies of patients with obesity with psoriasis. However, these studies have shown a positive correlation between anxiety scores and BMI in patients with psoriasis.24,25 In our study, similar to the findings of previous studies, overweight patients and those with obesitywho have psoriasis had significantly (P<.01) greater anxiety and stress scores than did normal weight patients with psoriasis.
Obesity should be assessed in patients with psoriasis via a biopsychosocial approach that takes into account genetic, behavioral, and environmental factors.26 Eating disorders are considered to be one of the factors contributing to obesity. Numerous studies in the literature have demonstrated a greater incidence of EDs in patients with obesity vs those without obesity.5,6,27 Obesity and EDs have a bidirectional relationship: individuals with obesity are at risk for EDs due to body dissatisfaction, dieting habits, and depressive states. Conversely, poor eating behaviors in individuals with a normal weight can lead to obesity.28
There are few studies in the literature exploring the relationship between psoriasis and EDs. Crosta et al29 demonstrated that patients with psoriasis had impaired results on ED screening tests and that these scores deteriorated further as BMI increased. Moreover, Altunay et al30 demonstrated that patients with psoriasis and metabolic syndrome had higher scores on the ED screening test. In this study, patients with higher scores also exhibited high levels of anxiety.30 In our study, similar to the findings of previous studies, patients with psoriasis who were overweight or had obesity had significantly (P<.01) greater EAT-26 scores than those in the normal weight group. Patients with high EAT-26 scores also exhibited elevated levels of depression, anxiety, and stress. Additionally, EAT-26 scores were positively correlated with BMI, anxiety, depression, and stress scores. Our study as well as other studies in the literature indicate that additional research is needed to determine the associations between EDs and obesity in psoriasis.
Conclusion
Managing obesity is crucial for patients with psoriasis. This study showed that EAT-26 scores were higher in patients with psoriasis who were overweight or had obesity than in those who were normal weight. Participants with high EAT-26 scores (≥20 points) were more likely to be female and have higher anxiety and stress scores. In addition, EAT-26 scores were positively correlated with BMI as well as depression, anxiety, and stress scores. Eating disorders may contribute to the development of obesity in patients with psoriasis. Although our study was limited by a small sample size, the results suggest that there is a need for large-scale multicenter studies to investigate the relationship between psoriasis and EDs.
Psoriasis is a chronic multisystemic inflammatory skin disease with a worldwide prevalence of 2% to 3%.1 Psoriasis can be accompanied by other conditions such as psoriatic arthritis, obesity, metabolic syndrome, diabetes mellitus, hypertension, dyslipidemia, atherosclerotic disease, inflammatory bowel disease, and anxiety/depression. It is important to manage comorbidities of psoriasis in addition to treating the cutaneous manifestations of the disease.1
Obesity is a major public health concern worldwide. Numerous observational and epidemiologic studies have reported a high prevalence of obesity among patients with psoriasis.2 Current evidence indicates that obesity may initiate or worsen psoriasis; furthermore, it is important to note that obesity may negatively impact the effectiveness of psoriasis-specific treatments or increase the incidence of adverse effects. Therefore, managing obesity is crucial in the treatment of psoriasis.3 Numerous studies have investigated the association between psoriasis and obesity, and they commonly conclude that both conditions share the same genetic metabolic pathways.2-4 However, it is important to consider environmental factors such as dietary habits, smoking, alcohol consumption, and a sedentary lifestyle—all of which are associated with psoriasis and also can contribute to the development of obesity.5 Because of the effects of obesity in psoriasis patients, factors that impact the development of obesity have become a popular research topic.
Eating disorders (EDs) are a crucial risk factor for both developing and maintaining obesity. In particular, two EDs that are associated with obesity include binge eating disorder and bulimia nervosa.6 According to the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition,7 binge eating disorder can be diagnosed when a patient has at least 1 episode of binge eating per week over a 3-month period. Bulimia nervosa can be diagnosed when a patient is excessively concerned with their body weight and shape and engages in behaviors to prevent weight gain (eg, forced vomiting, excessive use of laxatives).7 Psychiatrists who specialize in EDs make diagnoses based on these criteria. In daily practice, there are several quick and simple questionnaires available to screen for EDs that can be used by nonpsychiatrist physicians, including the commonly used 26-item Eating Attitudes Test (EAT-26).8 The EAT-26 has been used to screen for EDs in patients with inflammatory disorders.9
The aim of this study was to screen for EDs in patients with psoriasis to identify potential risk factors for development of obesity.
Materials and Methods
This study included patients with psoriasis who were screened for EDs at a tertiary dermatology clinic in Turkey between January 2021 and December 2023. This study was approved by the local ethics committee and was in accordance with the Declaration of Helsinki (decision number E-93471371-514.99-225000079).
Study Design and Patient Inclusion Criteria—This quantitative cross-sectional study utilized EAT-26, Dermatology Life Quality Index (DLQI), Attitude Scale for Healthy Nutrition (ASHN), and Depression Anxiety Stress Scale-21 (DASS-21) scores. All the questionnaire scales used in the study were adapted and validated in Turkey.8,10-12 The inclusion criteria consisted of being older than 18 years of age, being literate, having psoriasis for at least 1 year that was not treated topically or systemically, and having no psychiatric diseases outside an ED. The questionnaires were presented in written format following the clinical examination. Literacy was an inclusion criterion in this study due to the absence of auxiliary health personnel.
Study Variables—The study variables included age, sex, marital status (single/divorced or married), education status (primary/secondary school or high school/university), employment status (employed or unemployed/retired), body mass index (BMI), smoking status, alcohol-consumption status, Psoriasis Area Severity Index score, presence of nail psoriasis and psoriatic arthritis, duration of psoriasis, family history of psoriasis, EAT-26 score, ASHN score, DLQI score, and DASS-21 score. Body mass index was calculated by taking a participant’s weight in kilograms and dividing it by their height in meters squared. The BMI values were classified into 3 categories: normal (18.5–24.9 kg/m2), overweight (25.0–29.9 kg/m2), and obese (≥30 kg/m2).13
Questionnaires—The EAT-26 questionnaire includes 26 questions that are used to detect EDs. Responses to each question include Likert-type answer options (ie, “always,” “usually,” “often,” “sometimes,” “rarely,” and “never.”) Patients with scores of 20 points or higher (range, 0–78) are classified as high risk for EDs.8 In our study, EAT-26 scores were grouped into 2 categories: patients scoring less than 20 points and those scoring 20 points or higher.
The DLQI questionnaire includes 10 questions to measure dermatologic symptoms and qualiy of life. Responses to each question include Likert-type answer options (ie, “not at all,” “a little,” “a lot,” or “very much.”) On the DLQI scale, the higher the score, the lower the quality of life (score range, 0–30).10
The ASHN questionnaire includes 21 questions that measure attitudes toward healthy nutrition with 5 possible answer options (“strongly disagree,” “disagree,” “undecided,” “agree,” and “strongly agree”). On this scale, higher scores indicate the participant is more knowledgeable about healthy nutrition (score range, 0–78).11
The DASS-21 questionnaire includes 21 questions that measure the severity of a range of symptoms common to depression, anxiety, and stress. Responses include Likert-type answer options (eg, “never,” “sometimes,” “often,” and “almost always.”) On this scale, a higher score (range of 0–21 for each) indicates higher levels of depression, anxiety, and stress.12
Statistical Analysis—Descriptive statistics were analyzed using SPSS software version 22.0 (IBM). The Shapiro-Wilk test was applied to determine whether the data were normally distributed. For categorical variables, frequency differences among groups were compared using the Pearson χ2 test. A t test was used to compare the means of 2 independent groups with a normal distribution. One-way analysis of variance and Tukey Honest Significant Difference post hoc analysis were used to test whether there was a statistically significant difference among the normally distributed means of independent groups. Pearson correlation analysis was used to determine whether there was a linear relationship between 2 numeric measurements and, if so, to determine the direction and severity of this relationship. P<.05 indicated statistical significance in this study.
Results
Study Participant Demographics—This study included 82 participants with a mean age of 44.3 years; 52.4% (43/82) were female, and 85.4% (70/82) were married. The questionnaire took an average of 4.2 minutes for participants to complete. A total of 57.3% (47/82) of patients had completed primary/secondary education and 59.8% (49/82) were employed. The mean BMI was 28.1 kg/m2. According to the BMI classification, 26.8% (22/82) participants had a normal weight, 36.6% (30/82) were overweight, and 43.9% (36/82) were obese. A total of 48.8% (40/82) of participants smoked, and 4.9% (4/82) consumed alcohol. The mean Psoriasis Area and Severity Index score was 5.4. A total of 54.9% (45/82) of participants had nail psoriasis, and 24.4% (20/82) had psoriatic arthritis. The mean duration of psoriasis was 153 months. A total of 29.3% (24/82) of participants had a positive family history of psoriasis. The mean EAT-26 score was 11.1. A total of 12.2% (10/82) of participants had an EAT-26 score of 20 points or higher and were considered at high risk for an ED. The mean ASHN score was 72.9; the mean DLQI score was 5.5; and on the DASS-21 scale, mean scores for depression, anxiety, and stress were 6.3, 8.7, and 10.0, respectively (Table).

Comparative Evaluation of the BMI Groups—The only statistically significant differences among the 3 BMI groups were related to marital status, EAT-26 score, and anxiety and stress scores (P=.02, <.01, <.01, and <.01, respectively)(eTable 1). The number of single/divorced participants in the overweight group was significantly (P=.02) greater than in the normal weight group. The mean EAT-26 score for the normal weight group was significantly (P<.01) lower than for the overweight and obese groups; there was no significant difference in mean EAT-26 scores between the overweight and obese groups. The mean anxiety score was significantly (P<.01) lower in the normal weight group compared with the overweight and obese groups. There was no significant difference between the overweight and obese groups according to the mean depression score. The mean stress and anxiety scores were significantly (P<.01) lower in the normal weight group than in the overweight and obese groups. There was no significant difference between the overweight and obese groups according to the mean anxiety score.


Comparative Evaluation of the EAT-26 Scores—There were statistically significant differences among the EAT-26 scores related to sex; BMI; and depression, anxiety, and stress scores (P=.04, .02, <.01, <.01, and <.01, respectively). The number of females in the group with a score of 20 points or higher was significantly (P=.04) less than that in the group scoring less than 20 points. The mean BMI in the group with a score of 20 points or higher was significantly (P=.02) greater than in group scoring less than 20 points. The mean depression, anxiety, and stress scores of the group scoring 20 points or higher were significantly (P<.01 for all) greater than in the group scoring less than 20 points (eTable 2).


Correlation Analysis of the Study Variables—The EAT-26 scores were positively correlated with BMI, anxiety, depression, and stress (P<.01 for all)(eTable 3).

Comment
Eating disorders are psychiatric conditions that require a multidisciplinary approach. Nonpsychiatric medical departments may be involved due to the severe consequences (eg, various skin changes14) of these disorders. Psoriasis is not known to be directly affected by the presence of an ED; however, it is possible that EDs could indirectly affect patients with psoriasis by influencing obesity. Therefore, this study aimed to examine the relationship between ED risk factors and obesity in this population.
The relationship between psoriasis and obesity has been a popular research topic in dermatology since the 1990s.15 Epidemiologic and observational studies have reported that patients with psoriasis are more likely to be overweight or have obesity, which is an independent risk factor for psoriasis.3,16 However, the causal relationship between psoriasis and obesity remains unclear. In a comprehensive review, Barros et al17 emphasized the causal relationship between obesity and psoriasis under several headings. Firstly, a higher BMI increases the risk for psoriasis by promoting cytokine release and immune system dysregulation. Secondly, a Western diet (eg, processed foods and fast food) triggers obesity and psoriasis by increasing adipose tissue. Thirdly, the alteration of the skin and gut microbiota triggers chronic inflammation as a result of bacterial translocation in patients with obesity. Fourthly, a high-fat diet and palmitic acid disrupt the intestinal integrity of the gut and increase the risk for psoriasis and obesity by triggering chronic inflammation of bacterial fragments that pass into the blood. Finally, the decrease in the amount of adiponectin and the increase in the amount of leptin in patients with obesity may cause psoriasis by increasing proinflammatory cytokines, which are similar to those involved in the pathogenesis of psoriasis.17 Additionally, psoriatic inflammation can cause insulin resistance and metabolic dysfunction, leading to obesity.18 The relationship between psoriasis and obesity cannot be solely explained by metabolic pathways. Smoking, alcohol consumption, and a sedentary lifestyle all are associated with psoriasis and also can contribute to obesity.5 Our study revealed no significant difference in smoking or alcohol consumption between the normal weight and overweight/obesity groups. Based on our data, we determined that smoking and alcohol consumption did not affect obesity in our patients with psoriasis.
Observational and epidemiologic studies have shown that patients with psoriasis experience increased rates of depression, anxiety, and stress.19 In studies of pathogenesis, a connection between depression and psoriatic inflammation has been established.20 It is known that inflammatory cytokines similar to those in psoriasis are involved in the development of obesity.18 In addition, depression and anxiety can lead to binge eating, unhealthy food choices, and a more sedentary lifestyle.5 All of these variables may contribute to the associations between depression and anxiety with psoriasis and obesity. Zafiriou et al21 conducted a study to investigate the relationship between psoriasis, obesity, and depression through inflammatory pathways with a focus on the importance of IL-17. Data showing that IL-17–producing Th17-cell subgroups play a considerable role in the development of obesity and depression prompted the authors to suggest that psoriasis, obesity, and anxiety/depression may be interconnected manifestations of immune dysregulation, potentially linked to IL-17 and its associated cells.21 Mrowietz et al22 also suggested that metabolic inflammation may contribute to obesity and depression in patients with psoriasis and highlighted the importance of several cytokines, including tumor necrosis factor α, IL-6, IL-8, IL-17, and IL-23. Our study revealed no significant differences in depression scores between BMI groups. Another meta-analysis reported conflicting findings on the incidence of depression in obese patients with psoriasis.23 Some of the studies had a small number of participants. Compared to depression, anxiety has received less attention in studies of patients with obesity with psoriasis. However, these studies have shown a positive correlation between anxiety scores and BMI in patients with psoriasis.24,25 In our study, similar to the findings of previous studies, overweight patients and those with obesitywho have psoriasis had significantly (P<.01) greater anxiety and stress scores than did normal weight patients with psoriasis.
Obesity should be assessed in patients with psoriasis via a biopsychosocial approach that takes into account genetic, behavioral, and environmental factors.26 Eating disorders are considered to be one of the factors contributing to obesity. Numerous studies in the literature have demonstrated a greater incidence of EDs in patients with obesity vs those without obesity.5,6,27 Obesity and EDs have a bidirectional relationship: individuals with obesity are at risk for EDs due to body dissatisfaction, dieting habits, and depressive states. Conversely, poor eating behaviors in individuals with a normal weight can lead to obesity.28
There are few studies in the literature exploring the relationship between psoriasis and EDs. Crosta et al29 demonstrated that patients with psoriasis had impaired results on ED screening tests and that these scores deteriorated further as BMI increased. Moreover, Altunay et al30 demonstrated that patients with psoriasis and metabolic syndrome had higher scores on the ED screening test. In this study, patients with higher scores also exhibited high levels of anxiety.30 In our study, similar to the findings of previous studies, patients with psoriasis who were overweight or had obesity had significantly (P<.01) greater EAT-26 scores than those in the normal weight group. Patients with high EAT-26 scores also exhibited elevated levels of depression, anxiety, and stress. Additionally, EAT-26 scores were positively correlated with BMI, anxiety, depression, and stress scores. Our study as well as other studies in the literature indicate that additional research is needed to determine the associations between EDs and obesity in psoriasis.
Conclusion
Managing obesity is crucial for patients with psoriasis. This study showed that EAT-26 scores were higher in patients with psoriasis who were overweight or had obesity than in those who were normal weight. Participants with high EAT-26 scores (≥20 points) were more likely to be female and have higher anxiety and stress scores. In addition, EAT-26 scores were positively correlated with BMI as well as depression, anxiety, and stress scores. Eating disorders may contribute to the development of obesity in patients with psoriasis. Although our study was limited by a small sample size, the results suggest that there is a need for large-scale multicenter studies to investigate the relationship between psoriasis and EDs.
- Kalkan G. Comorbidities in psoriasis: the recognition of psoriasis as a systemic disease and current management. Turkderm-Turk Arch Dermatol Venereol. 2017;51:71-77.
- Armstrong AW, Harskamp CT, Armstrong EJ. The association between psoriasis and obesity: a systematic review and meta-analysis of observational studies. Nutr Diabetes. 2012;2:E54.
- Jensen P, Skov L. Psoriasis and obesity. Dermatology. 2016;232:633-639.
- Mirghani H, Altemani AT, Altemani ST, et al. The cross talk between psoriasis, obesity, and dyslipidemia: a meta-analysis. Cureus. 2023;15:e49253.
- Roehring M, Mashep MR, White MA, et al. The metabolic syndrome and behavioral correlates in obese patients with binge disorders. Obesity. 2009;17:481-486.
- da Luz FQ, Hay P, Touyz S, et al. Obesity with comorbid eating disorders: associated health risks and treatment approaches. Nutrients. 2018;10:829.
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition. American Psychiatric Association; 2013.
- Ergüney Okumus¸ FE, Sertel Berk HÖ. The psychometric properties of the Eating Attitudes Test short form (EAT-26) in a college sample. Stud Psychol. 2020;40:57-78.
- Stoleru G, Leopold A, Auerbach A, et al. Female gender, dissatisfaction with weight, and number of IBD related surgeries as independent risk factors for eating disorders among patients with inflammatory bowel diseases. BMC Gastroenterol. 2022;22:438.
- Öztürkcan S, Ermertcan AT, Eser E, et al. Cross validation of the Turkish version of dermatology life quality index. Int J Dermatol. 2006;45:1300-1307.
- Demir GT, Ciciog˘lu HI˙. Attitude scale for healthy nutrition (ASHN): validity and reliability study. Gaziantep Univ J Sport Sci. 2019;4:256-274.
- Yılmaz O, Boz H, Arslan A. The validity and reliability of depression stress and anxiety scale (DASS 21) Turkish short form. Res Financial Econ Soc Stud. 2017;2:78-91.
- Nuttall FQ. Body mass index: obesity, BMI, and health: a critical review. Nutr Today. 2015;50:117-128.
- Strumia R, Manzata E, Gualandi M. Is there a role for dermatologists in eating disorders? Expert Rev Dermatol. 2017; 2:109-112.
- Henseler T, Christophers E. Disease concomitance in psoriasis. J Am Acad Dermatol. 1995;32:982-986.
- Naldi L, Addis A, Chimenti S, et al. Impact of body mass index and obesity on clinical response to systemic treatment for psoriasis. evidence from the Psocare project. Dermatology. 2008;217:365-373.
- Barros G, Duran P, Vera I, et al. Exploring the links between obesity and psoriasis: a comprehensive review. Int J Mol Sci. 2022;23:7499.
- Hao Y, Zhu YJ, Zou S, et al. Metabolic syndrome and psoriasis: mechanisms and future directions. Front Immunol. 2021;12:711060.
- Jing D, Xiao H, Shen M, et al. Association of psoriasis with anxiety and depression: a case–control study in Chinese patients. Front Med (Lausanne). 2021;8:771645.
- Sahi FM, Masood A, Danawar NA, et al. Association between psoriasis and depression: a traditional review. Cureus. 2020;12:E9708.
- Zafiriou E, Daponte AI, Siokas V, et al. Depression and obesity in patients with psoriasis and psoriatic arthritis: is IL-17–mediated immune dysregulation the connecting link? Front Immunol. 2021;12:699848.
- Mrowietz U, Sümbül M, Gerdes S. Depression, a major comorbidity of psoriatic disease, is caused by metabolic inflammation. J Eur Acad Dermatol Venereol. 2023;37:1731-1738.
- Pavlova NT, Kioskli K, Smith C, et al. Psychosocial aspects of obesity in adults with psoriasis: a systematic review. Skin Health Dis. 2021;1:E33.
- Innamorati M, Quinto RM, Imperatori C, et al. Health-related quality of life and its association with alexithymia and difficulties in emotion regulation in patients with psoriasis. Compr Psychiatry. 2016;70:200-208.
- Tabolli S, Naldi L, Pagliarello C, et al. Evaluation of the impact of writing exercises interventions on quality of life in patients with psoriasis undergoing systemic treatments. Br J Dermatol. 2012;167:1254‐1264.
- Albuquerque D, Nóbrega C, Manco L, et al. The contribution of genetics and environment to obesity. Br Med Bull. 2017;123:159‐173.
- Balantekin KN, Grammer AC, Fitzsimmons-Craft EE, et al. Overweight and obesity are associated with increased eating disorder correlates and general psychopathology in university women with eating disorders. Eat Behav. 2021;41:101482.
- Jebeile H, Lister NB, Baur LA, et al. Eating disorder risk in adolescents with obesity. Obes Rev. 2021;22:E13173.
- Crosta ML, Caldarola G, Fraietta S, et al. Psychopathology and eating disorders in patients with psoriasis. G Ital Dermatol Venereol. 2014;149:355-361.
- Altunay I, Demirci GT, Ates B, et al. Do eating disorders accompany metabolic syndrome in psoriasis patients? results of a preliminary study. Clin Cosmet Investig Dermatol. 2011;4:139-143.
- Kalkan G. Comorbidities in psoriasis: the recognition of psoriasis as a systemic disease and current management. Turkderm-Turk Arch Dermatol Venereol. 2017;51:71-77.
- Armstrong AW, Harskamp CT, Armstrong EJ. The association between psoriasis and obesity: a systematic review and meta-analysis of observational studies. Nutr Diabetes. 2012;2:E54.
- Jensen P, Skov L. Psoriasis and obesity. Dermatology. 2016;232:633-639.
- Mirghani H, Altemani AT, Altemani ST, et al. The cross talk between psoriasis, obesity, and dyslipidemia: a meta-analysis. Cureus. 2023;15:e49253.
- Roehring M, Mashep MR, White MA, et al. The metabolic syndrome and behavioral correlates in obese patients with binge disorders. Obesity. 2009;17:481-486.
- da Luz FQ, Hay P, Touyz S, et al. Obesity with comorbid eating disorders: associated health risks and treatment approaches. Nutrients. 2018;10:829.
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition. American Psychiatric Association; 2013.
- Ergüney Okumus¸ FE, Sertel Berk HÖ. The psychometric properties of the Eating Attitudes Test short form (EAT-26) in a college sample. Stud Psychol. 2020;40:57-78.
- Stoleru G, Leopold A, Auerbach A, et al. Female gender, dissatisfaction with weight, and number of IBD related surgeries as independent risk factors for eating disorders among patients with inflammatory bowel diseases. BMC Gastroenterol. 2022;22:438.
- Öztürkcan S, Ermertcan AT, Eser E, et al. Cross validation of the Turkish version of dermatology life quality index. Int J Dermatol. 2006;45:1300-1307.
- Demir GT, Ciciog˘lu HI˙. Attitude scale for healthy nutrition (ASHN): validity and reliability study. Gaziantep Univ J Sport Sci. 2019;4:256-274.
- Yılmaz O, Boz H, Arslan A. The validity and reliability of depression stress and anxiety scale (DASS 21) Turkish short form. Res Financial Econ Soc Stud. 2017;2:78-91.
- Nuttall FQ. Body mass index: obesity, BMI, and health: a critical review. Nutr Today. 2015;50:117-128.
- Strumia R, Manzata E, Gualandi M. Is there a role for dermatologists in eating disorders? Expert Rev Dermatol. 2017; 2:109-112.
- Henseler T, Christophers E. Disease concomitance in psoriasis. J Am Acad Dermatol. 1995;32:982-986.
- Naldi L, Addis A, Chimenti S, et al. Impact of body mass index and obesity on clinical response to systemic treatment for psoriasis. evidence from the Psocare project. Dermatology. 2008;217:365-373.
- Barros G, Duran P, Vera I, et al. Exploring the links between obesity and psoriasis: a comprehensive review. Int J Mol Sci. 2022;23:7499.
- Hao Y, Zhu YJ, Zou S, et al. Metabolic syndrome and psoriasis: mechanisms and future directions. Front Immunol. 2021;12:711060.
- Jing D, Xiao H, Shen M, et al. Association of psoriasis with anxiety and depression: a case–control study in Chinese patients. Front Med (Lausanne). 2021;8:771645.
- Sahi FM, Masood A, Danawar NA, et al. Association between psoriasis and depression: a traditional review. Cureus. 2020;12:E9708.
- Zafiriou E, Daponte AI, Siokas V, et al. Depression and obesity in patients with psoriasis and psoriatic arthritis: is IL-17–mediated immune dysregulation the connecting link? Front Immunol. 2021;12:699848.
- Mrowietz U, Sümbül M, Gerdes S. Depression, a major comorbidity of psoriatic disease, is caused by metabolic inflammation. J Eur Acad Dermatol Venereol. 2023;37:1731-1738.
- Pavlova NT, Kioskli K, Smith C, et al. Psychosocial aspects of obesity in adults with psoriasis: a systematic review. Skin Health Dis. 2021;1:E33.
- Innamorati M, Quinto RM, Imperatori C, et al. Health-related quality of life and its association with alexithymia and difficulties in emotion regulation in patients with psoriasis. Compr Psychiatry. 2016;70:200-208.
- Tabolli S, Naldi L, Pagliarello C, et al. Evaluation of the impact of writing exercises interventions on quality of life in patients with psoriasis undergoing systemic treatments. Br J Dermatol. 2012;167:1254‐1264.
- Albuquerque D, Nóbrega C, Manco L, et al. The contribution of genetics and environment to obesity. Br Med Bull. 2017;123:159‐173.
- Balantekin KN, Grammer AC, Fitzsimmons-Craft EE, et al. Overweight and obesity are associated with increased eating disorder correlates and general psychopathology in university women with eating disorders. Eat Behav. 2021;41:101482.
- Jebeile H, Lister NB, Baur LA, et al. Eating disorder risk in adolescents with obesity. Obes Rev. 2021;22:E13173.
- Crosta ML, Caldarola G, Fraietta S, et al. Psychopathology and eating disorders in patients with psoriasis. G Ital Dermatol Venereol. 2014;149:355-361.
- Altunay I, Demirci GT, Ates B, et al. Do eating disorders accompany metabolic syndrome in psoriasis patients? results of a preliminary study. Clin Cosmet Investig Dermatol. 2011;4:139-143.
Practice Points
- Eating disorders are considered a contributing factor in obesity.
- Obesity is prevalent in patients with psoriasis, and current evidence indicates that obesity may initiate psoriasis or worsen existing disease.
- Obesity should be considered as contributory to the development of psoriasis via a biopsychosocial approach that accounts for genetic, behavioral, and environmental factors.
Evaluating Use of Empagliflozin for Diabetes Management in Veterans With Chronic Kidney Disease
More than 37 million Americans have diabetes mellitus (DM), and approximately 90% have type 2 DM (T2DM), including about 25% of veterans.1,2 The current guidelines suggest that therapy depends on a patient's comorbidities, management needs, and patient-centered treatment factors.3 About 1 in 3 adults with DM have chronic kidney disease (CKD), defined as the presence of kidney damage or an estimated glomerular filtration rate (eGFR) < 60 mL/min per 1.73 m2, persisting for ≥ 3 months.4
Sodium-glucose cotransporter-2 (SGLT-2) inhibitors are a class of antihyperglycemic agents acting on the SGLT-2 proteins expressed in the renal proximal convoluted tubules. They exert their effects by preventing the reabsorption of filtered glucose from the tubular lumen. There are 4 SGLT-2 inhibitors approved by the US Food and Drug Administration: canagliflozin, dapagliflozin, empagliflozin, and ertugliflozin. Empagliflozin is currently the preferred SGLT-2 inhibitor on the US Department of Veterans Affairs (VA) formulary.
According to the American Diabetes Association guidelines, empagliflozin is considered when an individual has or is at risk for atherosclerotic cardiovascular disease, heart failure, and CKD.3 SGLT-2 inhibitors are a favorable option due to their low risk for hypoglycemia while also promoting weight loss. The EMPEROR-Reduced trial demonstrated that, in addition to benefits for patients with heart failure, empagliflozin also slowed the progressive decline in kidney function in those with and without DM.5 The purpose of this study was to evaluate the effectiveness of empagliflozin on hemoglobin A1c (HbA1c) levels in patients with CKD at the Hershel “Woody” Williams VA Medical Center (HWWVAMC) in Huntington, West Virginia, along with other laboratory test markers.
Methods
The Marshall University Institutional Review Board #1 (Medical) and the HWWVAMC institutional review board and research and development committee each reviewed and approved this study. A retrospective chart review was conducted on patients diagnosed with T2DM and stage 3 CKD who were prescribed empagliflozin for DM management between January 1, 2015, and October 1, 2022, yielding 1771 patients. Data were obtained through the VHA Corporate Data Warehouse (CDW) and stored on the VA Informatics and Computing Infrastructure (VINCI) research server.
Patients were included if they were aged 18 to 89 years, prescribed empagliflozin by a VA clinician for the treatment of T2DM, had an eGFR between 30 and 59 mL/min/1.73 m2, and had an initial HbA1c between 7% and 10%. Using further random sampling, patients were either excluded or divided into, those with stage 3a CKD and those with stage 3b CKD. The primary endpoint of this study was the change in HbA1c levels in patients with stage 3b CKD (eGFR 30-44 mL/min/1.73 m2) compared with stage 3a (eGFR 45-59 mL/min/1.73 m2) after 12 months. The secondary endpoints included effects on renal function, weight, blood pressure, incidence of adverse drug events, and cardiovascular events. Of the excluded, 38 had HbA1c < 7%, 30 had HbA1c ≥ 10%, 21 did not have data at 1-year mark, 15 had the medication discontinued due to decline in renal function, 14 discontinued their medication without documented reason, 10 discontinued their medication due to adverse drug reactions (ADRs), 12 had eGFR > 60 mL/ min/1.73 m2, 9 died within 1 year of initiation, 4 had eGFR < 30 mL/min/1.73 m2, 1 had no baseline eGFR, and 1 was the spouse of a veteran.
Statistical Analysis
All statistical analyses were performed using STATA v.15. We used t tests to examine changes within each group, along with paired t tests to compare the 2 groups. Two-sample t tests were used to analyze the continuous data at both the primary and secondary endpoints.
Results
Of the 1771 patients included in the initial data set, a randomized sample of 255 charts were reviewed, 155 were excluded, and 100 were included. Fifty patients, had stage 3a CKD and 50 had stage 3b CKD. Baseline demographics were similar between the stage 3a and 3b groups (Table 1). Both groups were predominantly White and male, with mean age > 70 years.
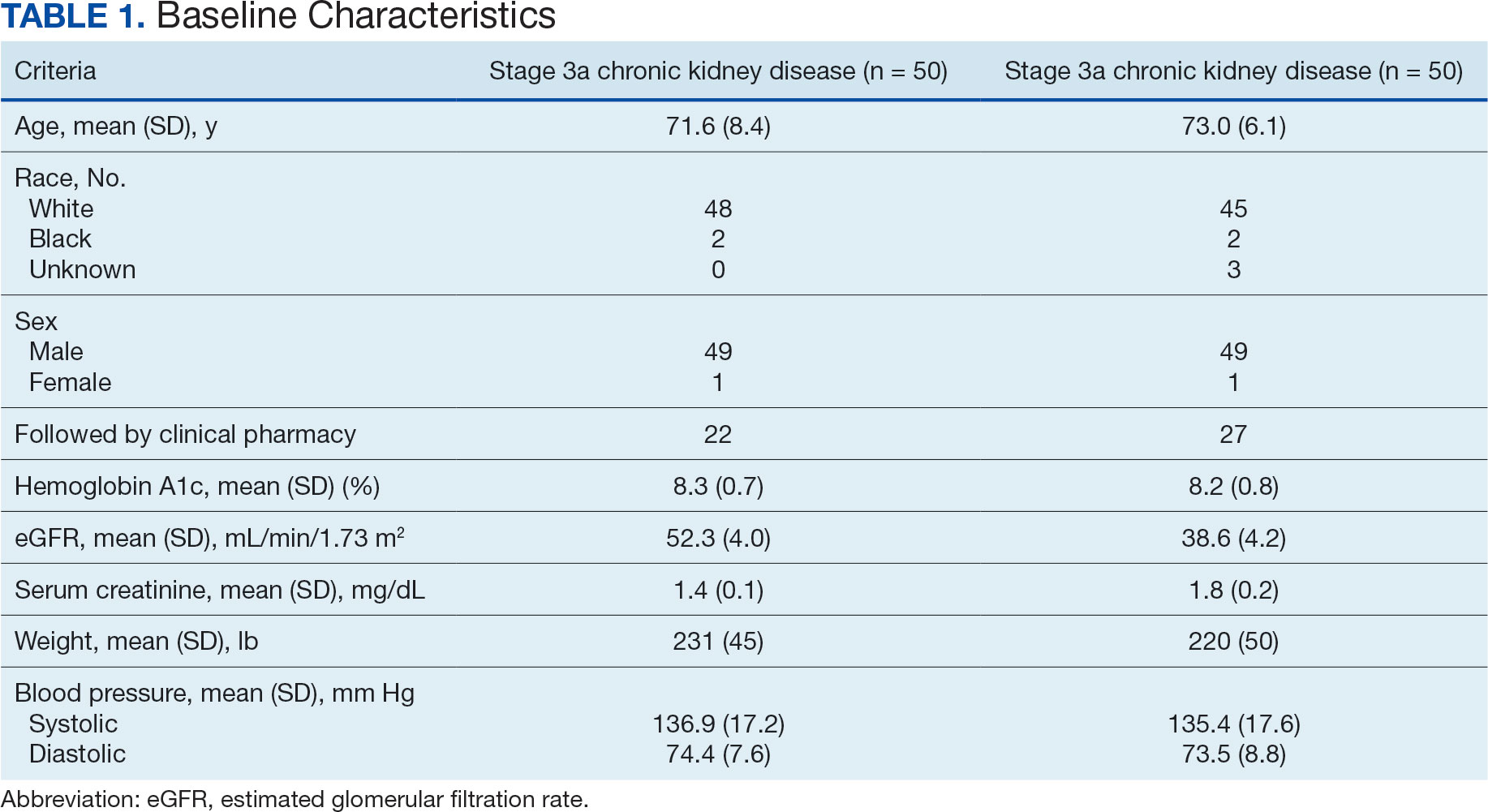
The primary endpoint was the differences in HbA1c levels over time and between groups for patients with stage 3a and stage 3b CKD 1 year after initiation of empagliflozin. The starting doses of empagliflozin were either 12.5 mg or 25.0 mg. For both groups, the changes in HbA1c levels were statistically significant (Table 2). HbA1c levels dropped 0.65% for the stage 3a group and 0.48% for the 3b group. When compared to one another, the results were not statistically significant (P = .51).
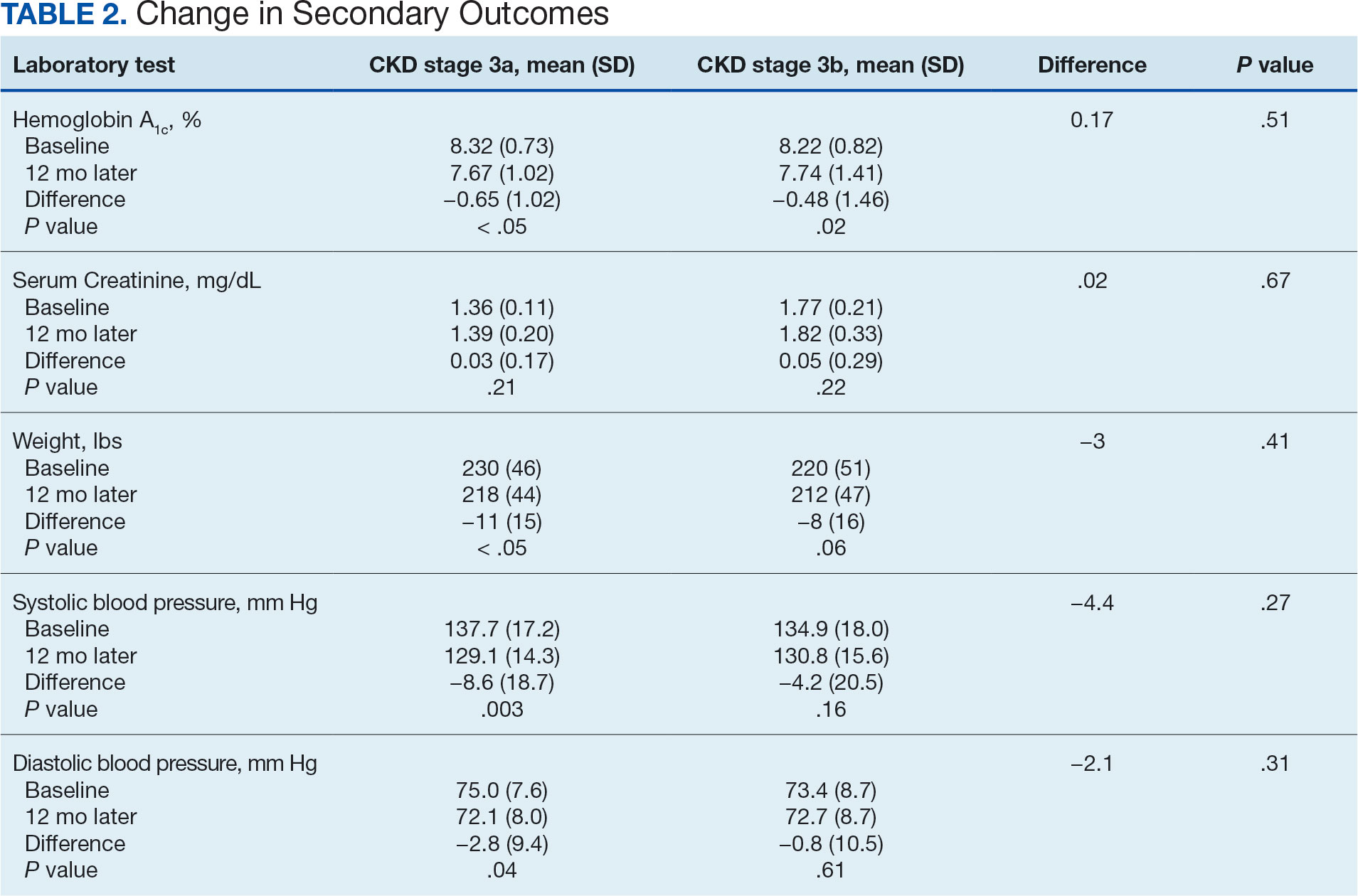
Secondary Endpoint
There was no statistically significant difference in serum creatinine levels within each group between baselines and 1 year later for the stage 3a (P = .21) and stage 3b (P = .22) groups, or when compared to each other (P = .67). There were statistically significant changes in weight for patients in the stage 3a group (P < .05), but not for stage 3b group (P = .06) or when compared to each other (P = .41). A statistically significant change in systolic blood pressure was observed for the stage 3a group (P = .003), but not the stage 3b group (P = .16) or when compared to each other (P = .27). There were statistically significant changes in diastolic blood pressure within the stage 3a group (P = .04), but not within the stage 3b group (P = .61) or when compared to each other (P = .31).
Ten patients discontinued empagliflozin before the 1-year mark due to ADRs, including dizziness, increased incidence of urinary tract infections, rash, and tachycardia (Table 3). Additionally, 3 ADRs resulted in the empagliflozin discontinuation after 1 year (Table 3).
Discussion
This study showed a statistically significant change in HbA1c levels for patients with stage 3a and stage 3b CKD. With eGFR levels in these 2 groups > 30 mL/min/1.73 m2, patients were able to achieve glycemic benefits. There were no significant changes to the serum creatinine levels. Both groups saw statistically significant changes in weight loss within their own group; however, there were no statistically significant changes when compared to each other. With both systolic and diastolic blood pressure, the stage 3a group had statistically significant changes.
The EMPA-REG BP study demonstrated that empagliflozin was associated with significant and clinically meaningful reductions in blood pressure and HbA1c levels compared with placebo and was well tolerated in patients with T2DM and hypertension.6,7,8
Limitations
This study had a retrospective study design, which resulted in missing information for many patients and higher rates of exclusion. The population was predominantly older, White, and male and may not reflect other populations. The starting doses of empagliflozin varied between the groups. The VA employs tablet splitting for some patients, and the available doses were either 10.0 mg, 12.5 mg, or 25.0 mg. Some prescribers start veterans at lower doses and gradually increase to the higher dose of 25.0 mg, adding to the variability in starting doses.
Patients with eGFR < 30 mL/min/1.73 m2 make it difficult to determine any potential benefit in this population. The EMPA-KIDNEY trial demonstrated that the benefits of empagliflozin treatment were consistent among patients with or without DM and regardless of eGFR at randomization.9 Furthermore, many veterans had an initial HbA1c levels outside the inclusion criteria range, which was a factor in the smaller sample size.
Conclusions
While the reduction in HbA1c levels was less in patients with stage 3b CKD compared to patients stage 3a CKD, all patients experienced a benefit. The overall incidence of ADRs was low in the study population, showing empagliflozin as a favorable choice for those with T2DM and CKD. Based on the findings of this study, empagliflozin is a potentially beneficial option for reducing HbA1c levels in patients with CKD.
- Centers for Disease Control and Prevention. Type 2 diabetes. Updated May 25, 2024. Accessed September 27, 2024. https://www.cdc.gov/diabetes/about/about-type-2-diabetes.html?CDC_AAref_Val
- US Department of Veterans Affairs, VA research on diabetes. Updated September 2019. Accessed September 27, 2024. https://www.research.va.gov/pubs/docs/va_factsheets/Diabetes.pdf
- American Diabetes Association. Standards of Medical Care in Diabetes-2022 Abridged for Primary Care Providers. Clin Diabetes. 2022;40(1):10-38. doi:10.2337/cd22-as01
- Centers for Disease Control and Prevention. Diabetes, chronic kidney disease. Updated May 15, 2024. Accessed September 27, 2024. https://www.cdc.gov/diabetes/diabetes-complications/diabetes-and-chronic-kidney-disease.html
- Packer M, Anker SD, Butler J, et al. Cardiovascular and Renal Outcomes with Empagliflozin in Heart Failure. N Engl J Med. 2020;383(15):1413-1424. doi:10.1056/NEJMoa2022190
- Tikkanen I, Narko K, Zeller C, et al. Empagliflozin reduces blood pressure in patients with type 2 diabetes and hypertension. Diabetes Care. 2015;38(3):420-428. doi:10.2337/dc14-1096
- Zinman B, Wanner C, Lachin JM, et al. Empagliflozin, cardiovascular outcomes, and mortality in type 2 diabetes. N Engl J Med. 2015;373(22):2117-2128. doi:10.1056/NEJMoa1504720
- Chilton R, Tikkanen I, Cannon CP, et al. Effects of empagliflozin on blood pressure and markers of arterial stiffness and vascular resistance in patients with type 2 diabetes. Diabetes Obes Metab. 2015;17(12):1180-1193. doi:10.1111/dom.12572
- The EMPA-KIDNEY Collaborative Group, Herrington WG, Staplin N, et al. Empagliflozin in Patients with Chronic Kidney Disease. N Engl J Med. 2023;388(2):117-127. doi:10.1056/NEJMoa2204233
More than 37 million Americans have diabetes mellitus (DM), and approximately 90% have type 2 DM (T2DM), including about 25% of veterans.1,2 The current guidelines suggest that therapy depends on a patient's comorbidities, management needs, and patient-centered treatment factors.3 About 1 in 3 adults with DM have chronic kidney disease (CKD), defined as the presence of kidney damage or an estimated glomerular filtration rate (eGFR) < 60 mL/min per 1.73 m2, persisting for ≥ 3 months.4
Sodium-glucose cotransporter-2 (SGLT-2) inhibitors are a class of antihyperglycemic agents acting on the SGLT-2 proteins expressed in the renal proximal convoluted tubules. They exert their effects by preventing the reabsorption of filtered glucose from the tubular lumen. There are 4 SGLT-2 inhibitors approved by the US Food and Drug Administration: canagliflozin, dapagliflozin, empagliflozin, and ertugliflozin. Empagliflozin is currently the preferred SGLT-2 inhibitor on the US Department of Veterans Affairs (VA) formulary.
According to the American Diabetes Association guidelines, empagliflozin is considered when an individual has or is at risk for atherosclerotic cardiovascular disease, heart failure, and CKD.3 SGLT-2 inhibitors are a favorable option due to their low risk for hypoglycemia while also promoting weight loss. The EMPEROR-Reduced trial demonstrated that, in addition to benefits for patients with heart failure, empagliflozin also slowed the progressive decline in kidney function in those with and without DM.5 The purpose of this study was to evaluate the effectiveness of empagliflozin on hemoglobin A1c (HbA1c) levels in patients with CKD at the Hershel “Woody” Williams VA Medical Center (HWWVAMC) in Huntington, West Virginia, along with other laboratory test markers.
Methods
The Marshall University Institutional Review Board #1 (Medical) and the HWWVAMC institutional review board and research and development committee each reviewed and approved this study. A retrospective chart review was conducted on patients diagnosed with T2DM and stage 3 CKD who were prescribed empagliflozin for DM management between January 1, 2015, and October 1, 2022, yielding 1771 patients. Data were obtained through the VHA Corporate Data Warehouse (CDW) and stored on the VA Informatics and Computing Infrastructure (VINCI) research server.
Patients were included if they were aged 18 to 89 years, prescribed empagliflozin by a VA clinician for the treatment of T2DM, had an eGFR between 30 and 59 mL/min/1.73 m2, and had an initial HbA1c between 7% and 10%. Using further random sampling, patients were either excluded or divided into, those with stage 3a CKD and those with stage 3b CKD. The primary endpoint of this study was the change in HbA1c levels in patients with stage 3b CKD (eGFR 30-44 mL/min/1.73 m2) compared with stage 3a (eGFR 45-59 mL/min/1.73 m2) after 12 months. The secondary endpoints included effects on renal function, weight, blood pressure, incidence of adverse drug events, and cardiovascular events. Of the excluded, 38 had HbA1c < 7%, 30 had HbA1c ≥ 10%, 21 did not have data at 1-year mark, 15 had the medication discontinued due to decline in renal function, 14 discontinued their medication without documented reason, 10 discontinued their medication due to adverse drug reactions (ADRs), 12 had eGFR > 60 mL/ min/1.73 m2, 9 died within 1 year of initiation, 4 had eGFR < 30 mL/min/1.73 m2, 1 had no baseline eGFR, and 1 was the spouse of a veteran.
Statistical Analysis
All statistical analyses were performed using STATA v.15. We used t tests to examine changes within each group, along with paired t tests to compare the 2 groups. Two-sample t tests were used to analyze the continuous data at both the primary and secondary endpoints.
Results
Of the 1771 patients included in the initial data set, a randomized sample of 255 charts were reviewed, 155 were excluded, and 100 were included. Fifty patients, had stage 3a CKD and 50 had stage 3b CKD. Baseline demographics were similar between the stage 3a and 3b groups (Table 1). Both groups were predominantly White and male, with mean age > 70 years.

The primary endpoint was the differences in HbA1c levels over time and between groups for patients with stage 3a and stage 3b CKD 1 year after initiation of empagliflozin. The starting doses of empagliflozin were either 12.5 mg or 25.0 mg. For both groups, the changes in HbA1c levels were statistically significant (Table 2). HbA1c levels dropped 0.65% for the stage 3a group and 0.48% for the 3b group. When compared to one another, the results were not statistically significant (P = .51).

Secondary Endpoint
There was no statistically significant difference in serum creatinine levels within each group between baselines and 1 year later for the stage 3a (P = .21) and stage 3b (P = .22) groups, or when compared to each other (P = .67). There were statistically significant changes in weight for patients in the stage 3a group (P < .05), but not for stage 3b group (P = .06) or when compared to each other (P = .41). A statistically significant change in systolic blood pressure was observed for the stage 3a group (P = .003), but not the stage 3b group (P = .16) or when compared to each other (P = .27). There were statistically significant changes in diastolic blood pressure within the stage 3a group (P = .04), but not within the stage 3b group (P = .61) or when compared to each other (P = .31).
Ten patients discontinued empagliflozin before the 1-year mark due to ADRs, including dizziness, increased incidence of urinary tract infections, rash, and tachycardia (Table 3). Additionally, 3 ADRs resulted in the empagliflozin discontinuation after 1 year (Table 3).

Discussion
This study showed a statistically significant change in HbA1c levels for patients with stage 3a and stage 3b CKD. With eGFR levels in these 2 groups > 30 mL/min/1.73 m2, patients were able to achieve glycemic benefits. There were no significant changes to the serum creatinine levels. Both groups saw statistically significant changes in weight loss within their own group; however, there were no statistically significant changes when compared to each other. With both systolic and diastolic blood pressure, the stage 3a group had statistically significant changes.
The EMPA-REG BP study demonstrated that empagliflozin was associated with significant and clinically meaningful reductions in blood pressure and HbA1c levels compared with placebo and was well tolerated in patients with T2DM and hypertension.6,7,8
Limitations
This study had a retrospective study design, which resulted in missing information for many patients and higher rates of exclusion. The population was predominantly older, White, and male and may not reflect other populations. The starting doses of empagliflozin varied between the groups. The VA employs tablet splitting for some patients, and the available doses were either 10.0 mg, 12.5 mg, or 25.0 mg. Some prescribers start veterans at lower doses and gradually increase to the higher dose of 25.0 mg, adding to the variability in starting doses.
Patients with eGFR < 30 mL/min/1.73 m2 make it difficult to determine any potential benefit in this population. The EMPA-KIDNEY trial demonstrated that the benefits of empagliflozin treatment were consistent among patients with or without DM and regardless of eGFR at randomization.9 Furthermore, many veterans had an initial HbA1c levels outside the inclusion criteria range, which was a factor in the smaller sample size.
Conclusions
While the reduction in HbA1c levels was less in patients with stage 3b CKD compared to patients stage 3a CKD, all patients experienced a benefit. The overall incidence of ADRs was low in the study population, showing empagliflozin as a favorable choice for those with T2DM and CKD. Based on the findings of this study, empagliflozin is a potentially beneficial option for reducing HbA1c levels in patients with CKD.
More than 37 million Americans have diabetes mellitus (DM), and approximately 90% have type 2 DM (T2DM), including about 25% of veterans.1,2 The current guidelines suggest that therapy depends on a patient's comorbidities, management needs, and patient-centered treatment factors.3 About 1 in 3 adults with DM have chronic kidney disease (CKD), defined as the presence of kidney damage or an estimated glomerular filtration rate (eGFR) < 60 mL/min per 1.73 m2, persisting for ≥ 3 months.4
Sodium-glucose cotransporter-2 (SGLT-2) inhibitors are a class of antihyperglycemic agents acting on the SGLT-2 proteins expressed in the renal proximal convoluted tubules. They exert their effects by preventing the reabsorption of filtered glucose from the tubular lumen. There are 4 SGLT-2 inhibitors approved by the US Food and Drug Administration: canagliflozin, dapagliflozin, empagliflozin, and ertugliflozin. Empagliflozin is currently the preferred SGLT-2 inhibitor on the US Department of Veterans Affairs (VA) formulary.
According to the American Diabetes Association guidelines, empagliflozin is considered when an individual has or is at risk for atherosclerotic cardiovascular disease, heart failure, and CKD.3 SGLT-2 inhibitors are a favorable option due to their low risk for hypoglycemia while also promoting weight loss. The EMPEROR-Reduced trial demonstrated that, in addition to benefits for patients with heart failure, empagliflozin also slowed the progressive decline in kidney function in those with and without DM.5 The purpose of this study was to evaluate the effectiveness of empagliflozin on hemoglobin A1c (HbA1c) levels in patients with CKD at the Hershel “Woody” Williams VA Medical Center (HWWVAMC) in Huntington, West Virginia, along with other laboratory test markers.
Methods
The Marshall University Institutional Review Board #1 (Medical) and the HWWVAMC institutional review board and research and development committee each reviewed and approved this study. A retrospective chart review was conducted on patients diagnosed with T2DM and stage 3 CKD who were prescribed empagliflozin for DM management between January 1, 2015, and October 1, 2022, yielding 1771 patients. Data were obtained through the VHA Corporate Data Warehouse (CDW) and stored on the VA Informatics and Computing Infrastructure (VINCI) research server.
Patients were included if they were aged 18 to 89 years, prescribed empagliflozin by a VA clinician for the treatment of T2DM, had an eGFR between 30 and 59 mL/min/1.73 m2, and had an initial HbA1c between 7% and 10%. Using further random sampling, patients were either excluded or divided into, those with stage 3a CKD and those with stage 3b CKD. The primary endpoint of this study was the change in HbA1c levels in patients with stage 3b CKD (eGFR 30-44 mL/min/1.73 m2) compared with stage 3a (eGFR 45-59 mL/min/1.73 m2) after 12 months. The secondary endpoints included effects on renal function, weight, blood pressure, incidence of adverse drug events, and cardiovascular events. Of the excluded, 38 had HbA1c < 7%, 30 had HbA1c ≥ 10%, 21 did not have data at 1-year mark, 15 had the medication discontinued due to decline in renal function, 14 discontinued their medication without documented reason, 10 discontinued their medication due to adverse drug reactions (ADRs), 12 had eGFR > 60 mL/ min/1.73 m2, 9 died within 1 year of initiation, 4 had eGFR < 30 mL/min/1.73 m2, 1 had no baseline eGFR, and 1 was the spouse of a veteran.
Statistical Analysis
All statistical analyses were performed using STATA v.15. We used t tests to examine changes within each group, along with paired t tests to compare the 2 groups. Two-sample t tests were used to analyze the continuous data at both the primary and secondary endpoints.
Results
Of the 1771 patients included in the initial data set, a randomized sample of 255 charts were reviewed, 155 were excluded, and 100 were included. Fifty patients, had stage 3a CKD and 50 had stage 3b CKD. Baseline demographics were similar between the stage 3a and 3b groups (Table 1). Both groups were predominantly White and male, with mean age > 70 years.

The primary endpoint was the differences in HbA1c levels over time and between groups for patients with stage 3a and stage 3b CKD 1 year after initiation of empagliflozin. The starting doses of empagliflozin were either 12.5 mg or 25.0 mg. For both groups, the changes in HbA1c levels were statistically significant (Table 2). HbA1c levels dropped 0.65% for the stage 3a group and 0.48% for the 3b group. When compared to one another, the results were not statistically significant (P = .51).

Secondary Endpoint
There was no statistically significant difference in serum creatinine levels within each group between baselines and 1 year later for the stage 3a (P = .21) and stage 3b (P = .22) groups, or when compared to each other (P = .67). There were statistically significant changes in weight for patients in the stage 3a group (P < .05), but not for stage 3b group (P = .06) or when compared to each other (P = .41). A statistically significant change in systolic blood pressure was observed for the stage 3a group (P = .003), but not the stage 3b group (P = .16) or when compared to each other (P = .27). There were statistically significant changes in diastolic blood pressure within the stage 3a group (P = .04), but not within the stage 3b group (P = .61) or when compared to each other (P = .31).
Ten patients discontinued empagliflozin before the 1-year mark due to ADRs, including dizziness, increased incidence of urinary tract infections, rash, and tachycardia (Table 3). Additionally, 3 ADRs resulted in the empagliflozin discontinuation after 1 year (Table 3).

Discussion
This study showed a statistically significant change in HbA1c levels for patients with stage 3a and stage 3b CKD. With eGFR levels in these 2 groups > 30 mL/min/1.73 m2, patients were able to achieve glycemic benefits. There were no significant changes to the serum creatinine levels. Both groups saw statistically significant changes in weight loss within their own group; however, there were no statistically significant changes when compared to each other. With both systolic and diastolic blood pressure, the stage 3a group had statistically significant changes.
The EMPA-REG BP study demonstrated that empagliflozin was associated with significant and clinically meaningful reductions in blood pressure and HbA1c levels compared with placebo and was well tolerated in patients with T2DM and hypertension.6,7,8
Limitations
This study had a retrospective study design, which resulted in missing information for many patients and higher rates of exclusion. The population was predominantly older, White, and male and may not reflect other populations. The starting doses of empagliflozin varied between the groups. The VA employs tablet splitting for some patients, and the available doses were either 10.0 mg, 12.5 mg, or 25.0 mg. Some prescribers start veterans at lower doses and gradually increase to the higher dose of 25.0 mg, adding to the variability in starting doses.
Patients with eGFR < 30 mL/min/1.73 m2 make it difficult to determine any potential benefit in this population. The EMPA-KIDNEY trial demonstrated that the benefits of empagliflozin treatment were consistent among patients with or without DM and regardless of eGFR at randomization.9 Furthermore, many veterans had an initial HbA1c levels outside the inclusion criteria range, which was a factor in the smaller sample size.
Conclusions
While the reduction in HbA1c levels was less in patients with stage 3b CKD compared to patients stage 3a CKD, all patients experienced a benefit. The overall incidence of ADRs was low in the study population, showing empagliflozin as a favorable choice for those with T2DM and CKD. Based on the findings of this study, empagliflozin is a potentially beneficial option for reducing HbA1c levels in patients with CKD.
- Centers for Disease Control and Prevention. Type 2 diabetes. Updated May 25, 2024. Accessed September 27, 2024. https://www.cdc.gov/diabetes/about/about-type-2-diabetes.html?CDC_AAref_Val
- US Department of Veterans Affairs, VA research on diabetes. Updated September 2019. Accessed September 27, 2024. https://www.research.va.gov/pubs/docs/va_factsheets/Diabetes.pdf
- American Diabetes Association. Standards of Medical Care in Diabetes-2022 Abridged for Primary Care Providers. Clin Diabetes. 2022;40(1):10-38. doi:10.2337/cd22-as01
- Centers for Disease Control and Prevention. Diabetes, chronic kidney disease. Updated May 15, 2024. Accessed September 27, 2024. https://www.cdc.gov/diabetes/diabetes-complications/diabetes-and-chronic-kidney-disease.html
- Packer M, Anker SD, Butler J, et al. Cardiovascular and Renal Outcomes with Empagliflozin in Heart Failure. N Engl J Med. 2020;383(15):1413-1424. doi:10.1056/NEJMoa2022190
- Tikkanen I, Narko K, Zeller C, et al. Empagliflozin reduces blood pressure in patients with type 2 diabetes and hypertension. Diabetes Care. 2015;38(3):420-428. doi:10.2337/dc14-1096
- Zinman B, Wanner C, Lachin JM, et al. Empagliflozin, cardiovascular outcomes, and mortality in type 2 diabetes. N Engl J Med. 2015;373(22):2117-2128. doi:10.1056/NEJMoa1504720
- Chilton R, Tikkanen I, Cannon CP, et al. Effects of empagliflozin on blood pressure and markers of arterial stiffness and vascular resistance in patients with type 2 diabetes. Diabetes Obes Metab. 2015;17(12):1180-1193. doi:10.1111/dom.12572
- The EMPA-KIDNEY Collaborative Group, Herrington WG, Staplin N, et al. Empagliflozin in Patients with Chronic Kidney Disease. N Engl J Med. 2023;388(2):117-127. doi:10.1056/NEJMoa2204233
- Centers for Disease Control and Prevention. Type 2 diabetes. Updated May 25, 2024. Accessed September 27, 2024. https://www.cdc.gov/diabetes/about/about-type-2-diabetes.html?CDC_AAref_Val
- US Department of Veterans Affairs, VA research on diabetes. Updated September 2019. Accessed September 27, 2024. https://www.research.va.gov/pubs/docs/va_factsheets/Diabetes.pdf
- American Diabetes Association. Standards of Medical Care in Diabetes-2022 Abridged for Primary Care Providers. Clin Diabetes. 2022;40(1):10-38. doi:10.2337/cd22-as01
- Centers for Disease Control and Prevention. Diabetes, chronic kidney disease. Updated May 15, 2024. Accessed September 27, 2024. https://www.cdc.gov/diabetes/diabetes-complications/diabetes-and-chronic-kidney-disease.html
- Packer M, Anker SD, Butler J, et al. Cardiovascular and Renal Outcomes with Empagliflozin in Heart Failure. N Engl J Med. 2020;383(15):1413-1424. doi:10.1056/NEJMoa2022190
- Tikkanen I, Narko K, Zeller C, et al. Empagliflozin reduces blood pressure in patients with type 2 diabetes and hypertension. Diabetes Care. 2015;38(3):420-428. doi:10.2337/dc14-1096
- Zinman B, Wanner C, Lachin JM, et al. Empagliflozin, cardiovascular outcomes, and mortality in type 2 diabetes. N Engl J Med. 2015;373(22):2117-2128. doi:10.1056/NEJMoa1504720
- Chilton R, Tikkanen I, Cannon CP, et al. Effects of empagliflozin on blood pressure and markers of arterial stiffness and vascular resistance in patients with type 2 diabetes. Diabetes Obes Metab. 2015;17(12):1180-1193. doi:10.1111/dom.12572
- The EMPA-KIDNEY Collaborative Group, Herrington WG, Staplin N, et al. Empagliflozin in Patients with Chronic Kidney Disease. N Engl J Med. 2023;388(2):117-127. doi:10.1056/NEJMoa2204233
VHA Support for Home Health Agency Staff and Patients During Natural Disasters
As large-scale natural disasters become more common, health care coalitions and the engagement of health systems with local, state, and federal public health departments have effectively bolstered communities’ resilience via collective sharing and distribution of resources.1 These resources may include supplies and the dissemination of emergency information, education, and training.2 The COVID-19 pandemic demonstrated that larger health care systems including hospital networks and nursing homes are better connected to health care coalition resources than smaller, independent systems, such as community home health agencies.3 This leaves some organizations on their own to meet requirements that maintain continuity of care and support their patients and staff throughout a natural disaster.
Home health care workers play important roles in the care of older adults.4 Older adults experience high levels of disability and comorbidities that put them at risk during emergencies; they often require support from paid, family, and neighborhood caregivers to live independently.5 More than 9.3 million US adults receive paid care from 2.6 million home health care workers (eg, home health aides and personal care assistants).6 Many of these individuals are hired through small independent home health agencies (HHAs), while others may work directly for an individual. When neighborhood resources and family caregiving are disrupted during emergencies, the critical services these workers administer become even more essential to ensuring continued access to medical care and social services.
The importance of these services was underscored by the Centers for Medicare and Medicaid Services 2017 inclusion of HHAs in federal emergency preparedness guidelines.7,8 The fractured and decentralized nature of the home health care industry means many HHAs struggle to maintain continuous care during emergencies and protect their staff. HHAs, and health care workers in the home, are often isolated, under-resourced, and disconnected from broader emergency planning efforts. Additionally, home care jobs are largely part-time, unstable, and low paying, making the workers themselves vulnerable during emergencies.3,9-13
This is a significant issue for the Veterans Health Administration (VHA), which annually purchases 10.5 million home health care worker visits for 150,000 veterans from community-based HHAs to enable those individuals to live independently. Figure 1 illustrates the existing structure of directly provided and contracted VHA services for community-dwelling veterans, highlighting the circle of care around the veteran.8,9 Home health care workers anchored health care teams during the COVID-19 pandemic, observing and reporting on patients’ well-being to family caregivers, primary care practitioners, and HHAs. They also provided critical emotional support and companionship to patients isolated from family and friends.9 These workers also exposed themselves and their families to considerable risk and often lacked the protection afforded by personal protective equipment (PPE) in accordance with infection prevention guidance.3,12

Abbreviations: HBPC, home based primary care; HHA, home health agency; VHA, Veterans Health Administration.
aAdapted with permission from Wyte-Lake and Franzosa.8,9
Through a combination of its national and local health care networks, the VHA has a robust and well-positioned emergency infrastructure to supportcommunity-dwelling older adults during disasters.14 This network is supported by the VHA Office of Emergency Management, which shares resources and guidance with local emergency managers at each facility as well as individual programs such as the VHA Home Based Primary Care (HBPC) program, which provides 38,000 seriously ill veterans with home medical visits.15 Working closely with their local and national hospital networks and emergency managers, individual VHA HBPC programs were able to maintain the safety of staff and continuity of care for patients enrolled in HBPC by rapidly administering COVID-19 vaccines to patients, caregivers, and staff, and providing emergency assistance during the 2017 hurricane season.16,17 These efforts were successful because HBPC practitioners and their patients, had access to a level of emergency-related information, resources, and technology that are often out of reach for individual community-based health care practitioners (HCPs). The US Department of Veterans Affairs (VA) also supports local communities through its Fourth Mission, which provides emergency resources to non-VHA health care facilities (ie, hospitals and nursing homes) during national emergencies and natural disasters.17 Although there has been an expansion in the definition of shared resources, such as extending behavioral health support to local communities, the VHA has not historically provided these resources to HHAs.14
This study examines opportunities to leverage VHA emergency management resources to support contracted HHAs and inform other large health system emergency planning efforts. The findings from the exploratory phase are described in this article. We interviewed VHA emergency managers, HBPC and VA staff who coordinate home health care worker services, as well as administrators at contracted HHAs within a Veterans Integrated Services Network (VISN). These findings will inform the second (single-site pilot study) and third (feasibility study) phases. Our intent was to (1) better understand the relationships between VA medical centers (VAMCs) and their contracted HHAs; (2) identify existing VHA emergency protocols to support community-dwelling older adults; and (3) determine opportunities to build on existing infrastructure and relationships to better support contracted HHAs and their staff in emergencies.
Methods
The 18 VISNs act as regional systems of care that are loosely connected to better meet local health needs and maximize access to care. This study was conducted at 6 of 9 VAMCs within VISN 2, the New York/New Jersey VHA Health Care Network.18 VAMCs that serve urban, rural, and mixed urban/rural catchment areas were included.
Each VAMC has an emergency management program led by an emergency manager, an HBPC program led by a program director and medical director, and a community care or purchased care office that has a liaison who manages contracted home health care worker services. The studyfocused on HBPC programs because they are most likely to interact with veterans’ home health care workers in the home and care for community-dwelling veterans during emergencies. Each VHA also contracts with a series of local HHAs that generally have a dedicated staff member who interfaces with the VHA liaison. Our goal was to interview ≥ 1 emergency manager, ≥ 1 HBPC team member, ≥ 1 community care staff person, and ≥ 1 contracted home health agency administrator at each site to gain multiple perspectives from the range of HCPs serving veterans in the community.
Recruitment and Data Collection
The 6 sites were selected in consultation with VISN 2 leadership for their strong HBPC and emergency management programs. To recruit respondents, we contacted VISN and VAMC leads and used our professional networks to identify a sample of multidisciplinary individuals who represent both community care and HBPC programs who were contacted via email.
Since each VAMC is organized differently, we utilized a snowball sampling approach to identify the appropriate contacts.19 At the completion of each interview, we asked the participant to suggest additional contacts and introduce us to any remaining stakeholders (eg, the emergency manager) at that site or colleagues at other VISN facilities. Because roles vary among VAMCs, we contacted the person who most closely resembled the identified role and asked them to direct us to a more appropriate contact, if necessary. We asked community care managers to identify 1 to 2 agencies serving the highest volume of patients who are veterans at their site and requested interviews with those liaisons. This resulted in the recruitment of key stakeholders from 4 teams across the 6 sites (Table).
A semistructured interview guide was jointly developed based on constructs of interest, including relationships within VAMCs and between VAMCs and HHAs; existing emergency protocols and experience during disasters; and suggestions and opportunities for supporting agencies during emergencies and potential barriers. Two researchers (TWL and EF) who were trained in qualitative methods jointly conducted interviews using the interview guide, with 1 researcher leading and another taking notes and asking clarifying questions.
Interviews were conducted virtually via Microsoft Teams with respondents at their work locations between September 2022 and January 2023. Interviews were audio recorded and transcribed and 2 authors (TWL and ESO) reviewed transcripts for accuracy. Interviews averaged 47 minutes in length (range, 20-59).
The study was reviewed and determined to be exempt by institutional review boards at the James J. Peters VAMC and Greater Los Angeles VAMC. We asked participants for verbal consent to participate and preserved their confidentiality.
Analysis
Data were analyzed via an inductive approach, which involves drawing salient themes rather than imposing preconceived theories.20 Three researchers (TWL, EF, and ES) listened to and discussed 2 staff interviews and tagged text with specific codes (eg, communication between the VHA and HHA, internal communication, and barriers to case fulfillment) so the team could selectively return to the interview text for deeper analysis, allowing for the development of a final codebook. The project team synthesized the findings to identify higher-level themes, drawing comparisons across and within the respondent groups, including within and between health care systems. Throughout the analysis, we maintained analytic memos, documented discussions, and engaged in analyst triangulation to ensure trustworthiness.21,22 To ensure the analysis accurately reflected the participants’ understanding, we held 2 virtual member-checking sessions with participants to share preliminary findings and conclusions and solicit feedback. Analysis was conducted using ATLAS.ti version 20.
Results
VHA-based participants described internal emergency management systems that are deployed during a disaster to support patients and staff. Agency participants described their own internal emergency management protocols. Respondents discussed how and when the 2 intersected, as well as opportunities for future mutual support. The analysis identified several themes: (1) relationships between VAMC teams; (2) relationships between VHA and HHAs; (3) VHA and agencies responses during emergencies; (4) receptivity and opportunities for extending VHA resources into the community; and (5) barriers and facilitators to deeper engagement.
Relationships Within VHA (n = 17)
Staff at all VHA sites described close relationships between the internal emergency management and HBPC teams. HBPC teams identified patients who were most at risk during emergencies to triage those with the highest medical needs (eg, patients dependent on home infusion, oxygen, or electronic medical devices) and worked alongside emergency managers to develop plans to continue care during an emergency. HBPC representatives were part of their facilities’ local emergency response committees. Due to this close collaboration, VHA emergency managers were familiar with the needs of homebound veterans and caregivers. “I invite our [HBPC] program manager to attend [committee] meetings and … they’re part of the EOC [emergency operations center]," an emergency manager said. “We work together and I’m constantly in contact with that individual, especially during natural disasters and so forth, to ensure that everybody’s prepared in the community.”
On the other hand, community caremanagers—who described frequent interactions with HBPC teams, largely around coordinating and managing non-VHA home care services—were less likely to have direct relationships with their facility emergency managers. For example, when asked if they had a relationship with their emergency manager, a community care manager admitted, “I [only] know who he is.” They also did not report having structured protocols for veteran outreach during emergencies, “because all those veterans who are receiving [home health care worker] services also belong to a primary care team,” and considered the outreach to be the responsibility of the primary care team and HHA.
Relationships Between the VHA and HHAs (n = 17)
Communication between VAMCs and contracted agencies primarily went through community care managers, who described established long-term relationships with agency administrators. Communication was commonly restricted to operational activities, such as processing referrals and occasional troubleshooting. According to a community care manager most communication is “why haven’t you signed my orders?” There was a general sense from participants that communication was promptly answered, problems were addressed, and professional collegiality existed between the agencies as patients were referred and placed for services. One community care manager reported meeting with agencies regularly, noting, “I talk to them pretty much daily.”
If problems arose, community care managers described themselves as “the liaison” between agencies and VHA HCPs who ordered the referrals. This is particularly the case if the agency needed help finding a VHA clinician or addressing differences in care delivery protocols.
Responding During Emergencies (n = 19)
During emergencies, VHA and agency staff described following their own organization’s protocols and communicating with each other only on a case-by-case basis rather than through formal or systematic channels and had little knowledge of their counterpart’s emergency protocols. Beyond patient care, there was no evidence of information sharing between VHA and agency staff. Regarding sharing information with their local community, an HBPC Program Director said, “it’s almost like the VHA had become siloed” and operated on its own without engaging with community health systems or emergency managers.
Beyond the guidance provided by state departments of public health, HHAs described collaborating with other agencies in their network and relying on their informal professional network to manage the volume of information and updates they followed during emergencies like the COVID-19 pandemic. One agency administrator did not frequently communicate with VHA partners during the pandemic but explained that the local public health department helped work through challenges. However, “we realized pretty quickly they were overloaded and there was only so much they could do.” The agency administrator turned to a “sister agency” and local hospitals, noting, “Wherever you have connections in the field or in the industry, you know you’re going to reach out to people for guidance on policies and… protocol.”
Opportunities for Extending VHA Resources to the Community (n = 16)
All VHA emergency managers were receptive to extending support to community-based HCPS and, in some cases, felt strongly that they were an essential part of veterans’ care networks. Emergency managers offered examples for how they supportedcommunity-based HCPs, such as helping those in the VAMC medical foster home program develop and evaluate emergency plans. Many said they had not explicitly considered HHAs before (Appendix).
Emergency managers also described how supporting community-based HCPs could be considered within the scope of the VHA role and mission, specifically the Fourth Mission. “I think that we should be making our best effort to make sure that we’re also providing that same level [of protection] to the people taking care of the veteran [as our VHA staff],” an emergency manager said. “It’s our responsibility to provide the best for the staff that are going into those homes to take care of that patient.”
In many cases, emergency managers had already developed practical tools that could be easily shared outside the VHA, including weather alerts, trainings, emergency plan templates, and lists of community resources and shelters (Figure 2). A number of these examples built on existing communication channels. One emergency manager said that the extension of resources could be an opportunity to decrease the perceived isolation of home health care workers through regular

Abbreviations: PPE, personal protective equipment; VA, US Department of Veterans Affairs.
On the agency side, participants noted that some HHAs could benefit more from support than others. While some agencies are well staffed and have good protocols and keep up to date, “There are smaller agencies, agencies that are starting up that may not have the resources to just disseminate all the information. Those are the agencies [that] could well benefit from the VHA,” an HBPC medical director explained. Agency administrators suggested several areas where they would welcome support, including a deeper understanding of available community resources and access to PPE for staff. Regarding informational resources, an administrator said, “Anytime we can get information, it’s good to have it come to you and not always have to go out searching for it.”
Barriers and Facilitators to Partnering With Community Agencies (n = 16)
A primary barrier regarding resource sharing was potential misalignment between each organization’s policies. HHAs followed state and federal public health guidelines, which sometimes differed from VHA policies. Given that agencies care for both VHA and non-VHA clients, questions also arose around how agencies would prioritize information from the VHA, if they were already receiving information from other sources. When asked about information sharing, both VHA staff and agencies agreed staff time to support any additional activities should be weighed against the value of the information gained.
Six participants also shared that education around emergency preparedness could be an opportunity to bridge gaps between VAMCs and their surrounding communities.
Two emergency managers noted the need to be sensitive in the way they engaged with partners, respecting and building on the work that agencies were already doing in this area to ensure VHA was seen as a trusted partner and resource rather than trying to impose new policies or rules on community-based HCPs. “I know that like all leadership in various organizations, there’s a little bit of bristling going on when other people try and tell them what to do,” an HBPC medical director said. “However, if it is established that as a sort of greater level like a state level or a federal level, that VHA can be a resource. I think that as long as that’s recognized by their own professional organizations within each state, then I think that that would be a tremendous advantage to many agencies.”
In terms of sharing physical resources, emergency managers raised concerns around potential liability, although they also acknowledged this issue was important enough to think about potential workarounds. As one emergency manager said, “I want to know that my PPE is not compromised in any way shape or form and that I am in charge of that PPE, so to rely upon going to a home and hoping that [the PPE] wasn’t compromised … would kind of make me a little uneasy.” This emergency manager suggested possible solutions, such as creating a sealed PPE package to give directly to an aide.
Discussion
As the prevalence of climate-related disasters increases, the need to ensure the safety and independence of older adults during emergencies grows more urgent. Health systems must think beyond the direct services they provide and consider the community resources upon which their patients rely. While relationships did not formally exist between VHA emergency managers and community home health HCPs in the sample analyzed in this article, there is precedent and interest in supporting contracted home health agencies caring for veterans in the community. Although not historically part of the VA Fourth Mission, creating a pipeline of support for contracted HHAs by leveraging existing relationships and resources can potentially strengthen its mission to protect older veterans in emergencies, help them age safely in place, and provide a model for health systems to collaborate with community-based HCPs around emergency planning and response (Figure 3).23

Existing research on the value of health care coalitions highlights the need for established and growing partnerships with a focus on ensuring they are value-added, which echoes concerns we heard in interviews.24 Investment in community partnerships not only includes sharing supplies but also relying on bidirectional support that can be a trusted form of timely information.1,25 The findings in this study exhibit strong communication practices within the VHA during periods of nonemergency and underscore the untapped value of the pre-existing relationship between VAMCs and their contracted HHAs as an area of potential growth for health care coalitions.
Sharing resources in a way that does not put new demands on partners contributes to the sustainability and value-added nature of coalitions. Examples include establishing new low-investment practices (ie, information sharing) that support capacity and compliance with existing requirements rather than create new responsibilities for either member of the coalition. The relationship between the VHA emergency managers and the VHA HBPC program can act as a guide. The emergency managers interviewed for this study are currently engaged with HBPC programs and therefore understand the needs of homebound older adults and their caregivers. Extending the information already available to the HBPC teams via existing channels strengthens workforce practices and increased security for the shared patient, even without direct relationships between emergency managers and agencies. It is important to understand the limitations of these practices, including concerns around conflicting federal and state mandates, legal concerns around the liability of sharing physical resources (such as PPE), and awareness that the objective is not for the VHA to increase burdens (eg, increasing compliance requirements) but rather to serve as a resource for a mutual population in a shared community.
Offering training and practical resources to HHA home health care workers can help them meet disaster preparedness requirements. This is particularly important considering the growing home care workforce shortages, a topic mentioned by all HBPC and community care participants interviewed for this study.26,27 Home health care workers report feeling underprepared and isolated while on the job in normal conditions, a sentiment exacerbated by the COVID-19 pandemic.3,10 Supporting these individuals may help them feel more prepared and connected to their work, improving stability and quality of care.
While these issues are priorities within the VHA, there is growing recognition at the state and federal level of the importance of including older adults and their HCPs in disaster preparedness and response.5,28 The US Department of Health and Human Services, for example, includes older adults and organizations that serve them on its National Advisory Committee on Seniors and Disasters. The Senate version of the 2023 reauthorization of the Pandemic and All-Hazards Preparedness and Response Act included specific provisions to support community-dwelling older adults and people with disabilities, incorporating funding for community organizations to support continuity of services and avoid institutionalization in an emergency.29 Other proposed legislation includes the Real Emergency Access for Aging and Disability Inclusion for Disasters Act, which would ensure the needs of older adults and people with disabilities are explicitly included in all phases of emergency planning and response.30
The VHA expansion of the its VEText program to include disaster response is an effort to more efficiently extend outreach to older and vulnerable patients who are veterans.31 Given these growing efforts, the VHA and other health systems have an opportunity to expand internal emergency preparedness efforts to ensure the health and safety of individuals living in the community.
Limitations
VISN 2 has been a target of terrorism and other disasters. In addition to the sites being initially recruited for their strong emergency management protocols, this context may have biased respondents who are favorable to extending their resources into the community. At the time of recruitment, contracted HHAs were still experiencing staff shortages due to the COVID-19 pandemic, which limited the ability of agency staff to participate in interviews. Additionally, while the comprehensive exploration of VISN 2 facilities allows for confidence of the organizational structures described, the qualitative research design and small study sample, the study findings cannot be immediately generalized to all VISNs.
Conclusions
Many older veterans increasingly rely on home health care workers to age safely. The VHA, as a large national health care system and leader in emergency preparedness, could play an important role in supporting home health care workers and ameliorating their sense of isolation during emergencies and natural disasters. Leveraging existing resources and relationships may be a low-cost, low-effort opportunity to build higher-level interventions that support the needs of patients. Future research and work in this field, including the authors’ ongoing work, will expand agency participation and engage agency staff in conceptualizing pilot projects to ensure they are viable and feasible for the field.

- Barnett DJ, Knieser L, Errett NA, Rosenblum AJ, Seshamani M, Kirsch TD. Reexamining health-care coalitions in light of COVID-19. Disaster Med public Health Prep. 2022;16(3):859-863. doi:10.1017/dmp.2020.431
- Wulff K, Donato D, Lurie N. What is health resilience and how can we build it? Annu Rev Public Health. 2015;36:361-374. doi:10.1146/annurev-publhealth-031914-122829
- Franzosa E, Wyte-Lake T, Tsui EK, Reckrey JM, Sterling MR. Essential but excluded: building disaster preparedness capacity for home health care workers and home care agencies. J Am Med Dir Assoc. 2022;23(12):1990-1996. doi:10.1016/j.jamda.2022.09.012
- Miner S, Masci L, Chimenti C, Rin N, Mann A, Noonan B. An outreach phone call project: using home health to reach isolated community dwelling adults during the COVID 19 lockdown. J Community Health. 2022;47(2):266-272. doi:10.1007/s10900-021-01044-6
- National Institute on Aging. Protecting older adults from the effects of natural disasters and extreme weather. October 18, 2022. Accessed August 19, 2024. https://www.nia.nih.gov/news/protecting-older-adults-effects-natural-disasters-and-extreme-weather
- PHI. Direct Care Workers in the United States: Key Facts. September 7, 2021. Accessed August 19, 2024. https://www.phinational.org/resource/direct-care-workers-in-the-united-states-key-facts-2/
- Centers for Medicare & Medicaid Services. Emergency Preparedness Rule. September 8, 2016. Updated September 6, 2023. Accessed August 19, 2024. https://www.cms.gov/medicare/health-safety-standards/quality-safety-oversight-emergency-preparedness/emergency-preparedness-rule
- Wyte-Lake T, Claver M, Tubbesing S, Davis D, Dobalian A. Development of a home health patient assessment tool for disaster planning. Gerontology. 2019;65(4):353-361. doi:10.1159/000494971
- Franzosa E, Judon KM, Gottesman EM, et al. Home health aides’ increased role in supporting older veterans and primary healthcare teams during COVID-19: a qualitative analysis. J Gen Intern Med. 2022;37(8):1830-1837. doi:10.1007/s11606-021-07271-w
- Franzosa E, Tsui EK, Baron S. “Who’s caring for us?”: understanding and addressing the effects of emotional labor on home health aides’ well-being. Gerontologist. 2019;59(6):1055-1064. doi:10.1093/geront/gny099
- Osakwe ZT, Osborne JC, Samuel T, et al. All alone: a qualitative study of home health aides’ experiences during the COVID-19 pandemic in New York. Am J Infect Control. 2021;49(11):1362-1368. doi:10.1016/j.ajic.2021.08.004
- Feldman PH, Russell D, Onorato N, et al. Ensuring the safety of the home health aide workforce and the continuation of essential patient care through sustainable pandemic preparedness. July 2022. Accessed August 19, 2024. https://www.vnshealth.org/wp-content/uploads/2022/08/Pandemic_Preparedness_IB_07_21_22.pdf
- Sterling MR, Tseng E, Poon A, et al. Experiences of home health care workers in New York City during the coronavirus disease 2019 pandemic: a qualitative analysis. JAMA Internal Med. 2020;180(11):1453-1459. doi:10.1001/jamainternmed.2020.3930
- Wyte-Lake T, Schmitz S, Kornegay RJ, Acevedo F, Dobalian A. Three case studies of community behavioral health support from the US Department of Veterans Affairs after disasters. BMC Public Health. 2021;21(1):639. doi:10.1186/s12889-021-10650-x
- Beales JL, Edes T. Veteran’s affairs home based primary care. Clin Geriatr Med. 2009;25(1):149-ix. doi:10.1016/j.cger.2008.11.002
- Wyte-Lake T, Manheim C, Gillespie SM, Dobalian A, Haverhals LM. COVID-19 vaccination in VA home based primary care: experience of interdisciplinary team members. J Am Med Dir Assoc. 2022;23(6):917-922. doi:10.1016/j.jamda.2022.03.014
- Wyte-Lake T, Schmitz S, Cosme Torres-Sabater R, Dobalian A. Case study of VA Caribbean Healthcare System’s community response to Hurricane Maria. J Emerg Manag. 2022;19(8):189-199. doi:10.5055/jem.0536
- US Department of Veterans Affairs. New York/New Jersey VA Health Care Network, VISN 2 Locations. Updated January 3, 2024. Accessed August 19, 2024. https://www.visn2.va.gov/visn2/facilities.asp
- Noy C. Sampling knowledge: the hermeneutics of snowball sampling in qualitative research. Int J Soc Res Methodol. 2008;11(4):327-344. doi:10.1080/13645570701401305
- Ritchie J, Lewis J, Nicholls CM, Ormston R, eds. Qualitative Research Practice: A Guide for Social Science Students and Researchers. 2nd ed. Sage; 2013.
- Morrow SL. Quality and trustworthiness in qualitative research in counseling psychology. J Couns Psychol. 2005;52(2):250-260. doi:10.1037/0022-0167.52.2.250
- Rolfe G. Validity, trustworthiness and rigour: quality and the idea of qualitative research. J Adv Nurs. 2006;53(3):304-310. doi:10.1111/j.1365-2648.2006.03727.x
- Schmitz S, Wyte-Lake T, Dobalian A. Facilitators and barriers to preparedness partnerships: a veterans affairs medical center perspective. Disaster Med Public Health Prep. 2018;12(4):431-436. doi:10.1017/dmp.2017.92
- Koch AE, Bohn J, Corvin JA, Seaberg J. Maturing into high-functioning health-care coalitions: a qualitative Nationwide study of emergency preparedness and response leadership. Disaster Med Public Health Prep. 2022;17:e111. doi:10.1017/dmp.2022.13
- Lin JS, Webber EM, Bean SI, Martin AM, Davies MC. Rapid evidence review: policy actions for the integration of public health and health care in the United States. Front Public Health. 2023;11:1098431. doi:10.3389/fpubh.2023.1098431
- Watts MOM, Burns A, Ammula M. Ongoing impacts of the pandemic on medicaid home & community-based services (HCBS) programs: findings from a 50-state survey. November 28, 2022. Accessed August 19, 2024. https://www.kff.org/medicaid/issue-brief/ongoing-impacts-of-the-pandemic-on-medicaid-home-community-based-services-hcbs-programs-findings-from-a-50-state-survey/
- Kreider AR, Werner RM. The home care workforce has not kept pace with growth in home and community-based services. Health Aff (Millwood). 2023;42(5):650-657. doi:10.1377/hlthaff.2022.01351
- FEMA introduces disaster preparedness guide for older adults. News release. FEMA. September 20, 2023. Accessed August 19, 2024. https://www.fema.gov/press-release/20230920/fema-introduces-disaster-preparedness-guide-older-adults
- Pandemic and All-Hazards Preparedness and Response Act, S 2333, 118th Cong, 1st Sess (2023). https://www.congress.gov/bill/118th-congress/senate-bill/2333/text
- REAADI for Disasters Act, HR 2371, 118th Cong, 1st Sess (2023). https://www.congress.gov/bill/118th-congress/house-bill/2371
- Wyte-Lake T, Brewster P, Hubert T, Gin J, Davis D, Dobalian A. VA’s experience building capability to conduct outreach to vulnerable patients during emergencies. Innov Aging. 2023;7(suppl 1):209. doi:10.1093/geroni/igad104.0690
As large-scale natural disasters become more common, health care coalitions and the engagement of health systems with local, state, and federal public health departments have effectively bolstered communities’ resilience via collective sharing and distribution of resources.1 These resources may include supplies and the dissemination of emergency information, education, and training.2 The COVID-19 pandemic demonstrated that larger health care systems including hospital networks and nursing homes are better connected to health care coalition resources than smaller, independent systems, such as community home health agencies.3 This leaves some organizations on their own to meet requirements that maintain continuity of care and support their patients and staff throughout a natural disaster.
Home health care workers play important roles in the care of older adults.4 Older adults experience high levels of disability and comorbidities that put them at risk during emergencies; they often require support from paid, family, and neighborhood caregivers to live independently.5 More than 9.3 million US adults receive paid care from 2.6 million home health care workers (eg, home health aides and personal care assistants).6 Many of these individuals are hired through small independent home health agencies (HHAs), while others may work directly for an individual. When neighborhood resources and family caregiving are disrupted during emergencies, the critical services these workers administer become even more essential to ensuring continued access to medical care and social services.
The importance of these services was underscored by the Centers for Medicare and Medicaid Services 2017 inclusion of HHAs in federal emergency preparedness guidelines.7,8 The fractured and decentralized nature of the home health care industry means many HHAs struggle to maintain continuous care during emergencies and protect their staff. HHAs, and health care workers in the home, are often isolated, under-resourced, and disconnected from broader emergency planning efforts. Additionally, home care jobs are largely part-time, unstable, and low paying, making the workers themselves vulnerable during emergencies.3,9-13
This is a significant issue for the Veterans Health Administration (VHA), which annually purchases 10.5 million home health care worker visits for 150,000 veterans from community-based HHAs to enable those individuals to live independently. Figure 1 illustrates the existing structure of directly provided and contracted VHA services for community-dwelling veterans, highlighting the circle of care around the veteran.8,9 Home health care workers anchored health care teams during the COVID-19 pandemic, observing and reporting on patients’ well-being to family caregivers, primary care practitioners, and HHAs. They also provided critical emotional support and companionship to patients isolated from family and friends.9 These workers also exposed themselves and their families to considerable risk and often lacked the protection afforded by personal protective equipment (PPE) in accordance with infection prevention guidance.3,12

Abbreviations: HBPC, home based primary care; HHA, home health agency; VHA, Veterans Health Administration.
aAdapted with permission from Wyte-Lake and Franzosa.8,9
Through a combination of its national and local health care networks, the VHA has a robust and well-positioned emergency infrastructure to supportcommunity-dwelling older adults during disasters.14 This network is supported by the VHA Office of Emergency Management, which shares resources and guidance with local emergency managers at each facility as well as individual programs such as the VHA Home Based Primary Care (HBPC) program, which provides 38,000 seriously ill veterans with home medical visits.15 Working closely with their local and national hospital networks and emergency managers, individual VHA HBPC programs were able to maintain the safety of staff and continuity of care for patients enrolled in HBPC by rapidly administering COVID-19 vaccines to patients, caregivers, and staff, and providing emergency assistance during the 2017 hurricane season.16,17 These efforts were successful because HBPC practitioners and their patients, had access to a level of emergency-related information, resources, and technology that are often out of reach for individual community-based health care practitioners (HCPs). The US Department of Veterans Affairs (VA) also supports local communities through its Fourth Mission, which provides emergency resources to non-VHA health care facilities (ie, hospitals and nursing homes) during national emergencies and natural disasters.17 Although there has been an expansion in the definition of shared resources, such as extending behavioral health support to local communities, the VHA has not historically provided these resources to HHAs.14
This study examines opportunities to leverage VHA emergency management resources to support contracted HHAs and inform other large health system emergency planning efforts. The findings from the exploratory phase are described in this article. We interviewed VHA emergency managers, HBPC and VA staff who coordinate home health care worker services, as well as administrators at contracted HHAs within a Veterans Integrated Services Network (VISN). These findings will inform the second (single-site pilot study) and third (feasibility study) phases. Our intent was to (1) better understand the relationships between VA medical centers (VAMCs) and their contracted HHAs; (2) identify existing VHA emergency protocols to support community-dwelling older adults; and (3) determine opportunities to build on existing infrastructure and relationships to better support contracted HHAs and their staff in emergencies.
Methods
The 18 VISNs act as regional systems of care that are loosely connected to better meet local health needs and maximize access to care. This study was conducted at 6 of 9 VAMCs within VISN 2, the New York/New Jersey VHA Health Care Network.18 VAMCs that serve urban, rural, and mixed urban/rural catchment areas were included.
Each VAMC has an emergency management program led by an emergency manager, an HBPC program led by a program director and medical director, and a community care or purchased care office that has a liaison who manages contracted home health care worker services. The studyfocused on HBPC programs because they are most likely to interact with veterans’ home health care workers in the home and care for community-dwelling veterans during emergencies. Each VHA also contracts with a series of local HHAs that generally have a dedicated staff member who interfaces with the VHA liaison. Our goal was to interview ≥ 1 emergency manager, ≥ 1 HBPC team member, ≥ 1 community care staff person, and ≥ 1 contracted home health agency administrator at each site to gain multiple perspectives from the range of HCPs serving veterans in the community.
Recruitment and Data Collection
The 6 sites were selected in consultation with VISN 2 leadership for their strong HBPC and emergency management programs. To recruit respondents, we contacted VISN and VAMC leads and used our professional networks to identify a sample of multidisciplinary individuals who represent both community care and HBPC programs who were contacted via email.
Since each VAMC is organized differently, we utilized a snowball sampling approach to identify the appropriate contacts.19 At the completion of each interview, we asked the participant to suggest additional contacts and introduce us to any remaining stakeholders (eg, the emergency manager) at that site or colleagues at other VISN facilities. Because roles vary among VAMCs, we contacted the person who most closely resembled the identified role and asked them to direct us to a more appropriate contact, if necessary. We asked community care managers to identify 1 to 2 agencies serving the highest volume of patients who are veterans at their site and requested interviews with those liaisons. This resulted in the recruitment of key stakeholders from 4 teams across the 6 sites (Table).
A semistructured interview guide was jointly developed based on constructs of interest, including relationships within VAMCs and between VAMCs and HHAs; existing emergency protocols and experience during disasters; and suggestions and opportunities for supporting agencies during emergencies and potential barriers. Two researchers (TWL and EF) who were trained in qualitative methods jointly conducted interviews using the interview guide, with 1 researcher leading and another taking notes and asking clarifying questions.
Interviews were conducted virtually via Microsoft Teams with respondents at their work locations between September 2022 and January 2023. Interviews were audio recorded and transcribed and 2 authors (TWL and ESO) reviewed transcripts for accuracy. Interviews averaged 47 minutes in length (range, 20-59).
The study was reviewed and determined to be exempt by institutional review boards at the James J. Peters VAMC and Greater Los Angeles VAMC. We asked participants for verbal consent to participate and preserved their confidentiality.
Analysis
Data were analyzed via an inductive approach, which involves drawing salient themes rather than imposing preconceived theories.20 Three researchers (TWL, EF, and ES) listened to and discussed 2 staff interviews and tagged text with specific codes (eg, communication between the VHA and HHA, internal communication, and barriers to case fulfillment) so the team could selectively return to the interview text for deeper analysis, allowing for the development of a final codebook. The project team synthesized the findings to identify higher-level themes, drawing comparisons across and within the respondent groups, including within and between health care systems. Throughout the analysis, we maintained analytic memos, documented discussions, and engaged in analyst triangulation to ensure trustworthiness.21,22 To ensure the analysis accurately reflected the participants’ understanding, we held 2 virtual member-checking sessions with participants to share preliminary findings and conclusions and solicit feedback. Analysis was conducted using ATLAS.ti version 20.
Results
VHA-based participants described internal emergency management systems that are deployed during a disaster to support patients and staff. Agency participants described their own internal emergency management protocols. Respondents discussed how and when the 2 intersected, as well as opportunities for future mutual support. The analysis identified several themes: (1) relationships between VAMC teams; (2) relationships between VHA and HHAs; (3) VHA and agencies responses during emergencies; (4) receptivity and opportunities for extending VHA resources into the community; and (5) barriers and facilitators to deeper engagement.
Relationships Within VHA (n = 17)
Staff at all VHA sites described close relationships between the internal emergency management and HBPC teams. HBPC teams identified patients who were most at risk during emergencies to triage those with the highest medical needs (eg, patients dependent on home infusion, oxygen, or electronic medical devices) and worked alongside emergency managers to develop plans to continue care during an emergency. HBPC representatives were part of their facilities’ local emergency response committees. Due to this close collaboration, VHA emergency managers were familiar with the needs of homebound veterans and caregivers. “I invite our [HBPC] program manager to attend [committee] meetings and … they’re part of the EOC [emergency operations center]," an emergency manager said. “We work together and I’m constantly in contact with that individual, especially during natural disasters and so forth, to ensure that everybody’s prepared in the community.”
On the other hand, community caremanagers—who described frequent interactions with HBPC teams, largely around coordinating and managing non-VHA home care services—were less likely to have direct relationships with their facility emergency managers. For example, when asked if they had a relationship with their emergency manager, a community care manager admitted, “I [only] know who he is.” They also did not report having structured protocols for veteran outreach during emergencies, “because all those veterans who are receiving [home health care worker] services also belong to a primary care team,” and considered the outreach to be the responsibility of the primary care team and HHA.
Relationships Between the VHA and HHAs (n = 17)
Communication between VAMCs and contracted agencies primarily went through community care managers, who described established long-term relationships with agency administrators. Communication was commonly restricted to operational activities, such as processing referrals and occasional troubleshooting. According to a community care manager most communication is “why haven’t you signed my orders?” There was a general sense from participants that communication was promptly answered, problems were addressed, and professional collegiality existed between the agencies as patients were referred and placed for services. One community care manager reported meeting with agencies regularly, noting, “I talk to them pretty much daily.”
If problems arose, community care managers described themselves as “the liaison” between agencies and VHA HCPs who ordered the referrals. This is particularly the case if the agency needed help finding a VHA clinician or addressing differences in care delivery protocols.
Responding During Emergencies (n = 19)
During emergencies, VHA and agency staff described following their own organization’s protocols and communicating with each other only on a case-by-case basis rather than through formal or systematic channels and had little knowledge of their counterpart’s emergency protocols. Beyond patient care, there was no evidence of information sharing between VHA and agency staff. Regarding sharing information with their local community, an HBPC Program Director said, “it’s almost like the VHA had become siloed” and operated on its own without engaging with community health systems or emergency managers.
Beyond the guidance provided by state departments of public health, HHAs described collaborating with other agencies in their network and relying on their informal professional network to manage the volume of information and updates they followed during emergencies like the COVID-19 pandemic. One agency administrator did not frequently communicate with VHA partners during the pandemic but explained that the local public health department helped work through challenges. However, “we realized pretty quickly they were overloaded and there was only so much they could do.” The agency administrator turned to a “sister agency” and local hospitals, noting, “Wherever you have connections in the field or in the industry, you know you’re going to reach out to people for guidance on policies and… protocol.”
Opportunities for Extending VHA Resources to the Community (n = 16)
All VHA emergency managers were receptive to extending support to community-based HCPS and, in some cases, felt strongly that they were an essential part of veterans’ care networks. Emergency managers offered examples for how they supportedcommunity-based HCPs, such as helping those in the VAMC medical foster home program develop and evaluate emergency plans. Many said they had not explicitly considered HHAs before (Appendix).
Emergency managers also described how supporting community-based HCPs could be considered within the scope of the VHA role and mission, specifically the Fourth Mission. “I think that we should be making our best effort to make sure that we’re also providing that same level [of protection] to the people taking care of the veteran [as our VHA staff],” an emergency manager said. “It’s our responsibility to provide the best for the staff that are going into those homes to take care of that patient.”
In many cases, emergency managers had already developed practical tools that could be easily shared outside the VHA, including weather alerts, trainings, emergency plan templates, and lists of community resources and shelters (Figure 2). A number of these examples built on existing communication channels. One emergency manager said that the extension of resources could be an opportunity to decrease the perceived isolation of home health care workers through regular

Abbreviations: PPE, personal protective equipment; VA, US Department of Veterans Affairs.
On the agency side, participants noted that some HHAs could benefit more from support than others. While some agencies are well staffed and have good protocols and keep up to date, “There are smaller agencies, agencies that are starting up that may not have the resources to just disseminate all the information. Those are the agencies [that] could well benefit from the VHA,” an HBPC medical director explained. Agency administrators suggested several areas where they would welcome support, including a deeper understanding of available community resources and access to PPE for staff. Regarding informational resources, an administrator said, “Anytime we can get information, it’s good to have it come to you and not always have to go out searching for it.”
Barriers and Facilitators to Partnering With Community Agencies (n = 16)
A primary barrier regarding resource sharing was potential misalignment between each organization’s policies. HHAs followed state and federal public health guidelines, which sometimes differed from VHA policies. Given that agencies care for both VHA and non-VHA clients, questions also arose around how agencies would prioritize information from the VHA, if they were already receiving information from other sources. When asked about information sharing, both VHA staff and agencies agreed staff time to support any additional activities should be weighed against the value of the information gained.
Six participants also shared that education around emergency preparedness could be an opportunity to bridge gaps between VAMCs and their surrounding communities.
Two emergency managers noted the need to be sensitive in the way they engaged with partners, respecting and building on the work that agencies were already doing in this area to ensure VHA was seen as a trusted partner and resource rather than trying to impose new policies or rules on community-based HCPs. “I know that like all leadership in various organizations, there’s a little bit of bristling going on when other people try and tell them what to do,” an HBPC medical director said. “However, if it is established that as a sort of greater level like a state level or a federal level, that VHA can be a resource. I think that as long as that’s recognized by their own professional organizations within each state, then I think that that would be a tremendous advantage to many agencies.”
In terms of sharing physical resources, emergency managers raised concerns around potential liability, although they also acknowledged this issue was important enough to think about potential workarounds. As one emergency manager said, “I want to know that my PPE is not compromised in any way shape or form and that I am in charge of that PPE, so to rely upon going to a home and hoping that [the PPE] wasn’t compromised … would kind of make me a little uneasy.” This emergency manager suggested possible solutions, such as creating a sealed PPE package to give directly to an aide.
Discussion
As the prevalence of climate-related disasters increases, the need to ensure the safety and independence of older adults during emergencies grows more urgent. Health systems must think beyond the direct services they provide and consider the community resources upon which their patients rely. While relationships did not formally exist between VHA emergency managers and community home health HCPs in the sample analyzed in this article, there is precedent and interest in supporting contracted home health agencies caring for veterans in the community. Although not historically part of the VA Fourth Mission, creating a pipeline of support for contracted HHAs by leveraging existing relationships and resources can potentially strengthen its mission to protect older veterans in emergencies, help them age safely in place, and provide a model for health systems to collaborate with community-based HCPs around emergency planning and response (Figure 3).23

Existing research on the value of health care coalitions highlights the need for established and growing partnerships with a focus on ensuring they are value-added, which echoes concerns we heard in interviews.24 Investment in community partnerships not only includes sharing supplies but also relying on bidirectional support that can be a trusted form of timely information.1,25 The findings in this study exhibit strong communication practices within the VHA during periods of nonemergency and underscore the untapped value of the pre-existing relationship between VAMCs and their contracted HHAs as an area of potential growth for health care coalitions.
Sharing resources in a way that does not put new demands on partners contributes to the sustainability and value-added nature of coalitions. Examples include establishing new low-investment practices (ie, information sharing) that support capacity and compliance with existing requirements rather than create new responsibilities for either member of the coalition. The relationship between the VHA emergency managers and the VHA HBPC program can act as a guide. The emergency managers interviewed for this study are currently engaged with HBPC programs and therefore understand the needs of homebound older adults and their caregivers. Extending the information already available to the HBPC teams via existing channels strengthens workforce practices and increased security for the shared patient, even without direct relationships between emergency managers and agencies. It is important to understand the limitations of these practices, including concerns around conflicting federal and state mandates, legal concerns around the liability of sharing physical resources (such as PPE), and awareness that the objective is not for the VHA to increase burdens (eg, increasing compliance requirements) but rather to serve as a resource for a mutual population in a shared community.
Offering training and practical resources to HHA home health care workers can help them meet disaster preparedness requirements. This is particularly important considering the growing home care workforce shortages, a topic mentioned by all HBPC and community care participants interviewed for this study.26,27 Home health care workers report feeling underprepared and isolated while on the job in normal conditions, a sentiment exacerbated by the COVID-19 pandemic.3,10 Supporting these individuals may help them feel more prepared and connected to their work, improving stability and quality of care.
While these issues are priorities within the VHA, there is growing recognition at the state and federal level of the importance of including older adults and their HCPs in disaster preparedness and response.5,28 The US Department of Health and Human Services, for example, includes older adults and organizations that serve them on its National Advisory Committee on Seniors and Disasters. The Senate version of the 2023 reauthorization of the Pandemic and All-Hazards Preparedness and Response Act included specific provisions to support community-dwelling older adults and people with disabilities, incorporating funding for community organizations to support continuity of services and avoid institutionalization in an emergency.29 Other proposed legislation includes the Real Emergency Access for Aging and Disability Inclusion for Disasters Act, which would ensure the needs of older adults and people with disabilities are explicitly included in all phases of emergency planning and response.30
The VHA expansion of the its VEText program to include disaster response is an effort to more efficiently extend outreach to older and vulnerable patients who are veterans.31 Given these growing efforts, the VHA and other health systems have an opportunity to expand internal emergency preparedness efforts to ensure the health and safety of individuals living in the community.
Limitations
VISN 2 has been a target of terrorism and other disasters. In addition to the sites being initially recruited for their strong emergency management protocols, this context may have biased respondents who are favorable to extending their resources into the community. At the time of recruitment, contracted HHAs were still experiencing staff shortages due to the COVID-19 pandemic, which limited the ability of agency staff to participate in interviews. Additionally, while the comprehensive exploration of VISN 2 facilities allows for confidence of the organizational structures described, the qualitative research design and small study sample, the study findings cannot be immediately generalized to all VISNs.
Conclusions
Many older veterans increasingly rely on home health care workers to age safely. The VHA, as a large national health care system and leader in emergency preparedness, could play an important role in supporting home health care workers and ameliorating their sense of isolation during emergencies and natural disasters. Leveraging existing resources and relationships may be a low-cost, low-effort opportunity to build higher-level interventions that support the needs of patients. Future research and work in this field, including the authors’ ongoing work, will expand agency participation and engage agency staff in conceptualizing pilot projects to ensure they are viable and feasible for the field.

As large-scale natural disasters become more common, health care coalitions and the engagement of health systems with local, state, and federal public health departments have effectively bolstered communities’ resilience via collective sharing and distribution of resources.1 These resources may include supplies and the dissemination of emergency information, education, and training.2 The COVID-19 pandemic demonstrated that larger health care systems including hospital networks and nursing homes are better connected to health care coalition resources than smaller, independent systems, such as community home health agencies.3 This leaves some organizations on their own to meet requirements that maintain continuity of care and support their patients and staff throughout a natural disaster.
Home health care workers play important roles in the care of older adults.4 Older adults experience high levels of disability and comorbidities that put them at risk during emergencies; they often require support from paid, family, and neighborhood caregivers to live independently.5 More than 9.3 million US adults receive paid care from 2.6 million home health care workers (eg, home health aides and personal care assistants).6 Many of these individuals are hired through small independent home health agencies (HHAs), while others may work directly for an individual. When neighborhood resources and family caregiving are disrupted during emergencies, the critical services these workers administer become even more essential to ensuring continued access to medical care and social services.
The importance of these services was underscored by the Centers for Medicare and Medicaid Services 2017 inclusion of HHAs in federal emergency preparedness guidelines.7,8 The fractured and decentralized nature of the home health care industry means many HHAs struggle to maintain continuous care during emergencies and protect their staff. HHAs, and health care workers in the home, are often isolated, under-resourced, and disconnected from broader emergency planning efforts. Additionally, home care jobs are largely part-time, unstable, and low paying, making the workers themselves vulnerable during emergencies.3,9-13
This is a significant issue for the Veterans Health Administration (VHA), which annually purchases 10.5 million home health care worker visits for 150,000 veterans from community-based HHAs to enable those individuals to live independently. Figure 1 illustrates the existing structure of directly provided and contracted VHA services for community-dwelling veterans, highlighting the circle of care around the veteran.8,9 Home health care workers anchored health care teams during the COVID-19 pandemic, observing and reporting on patients’ well-being to family caregivers, primary care practitioners, and HHAs. They also provided critical emotional support and companionship to patients isolated from family and friends.9 These workers also exposed themselves and their families to considerable risk and often lacked the protection afforded by personal protective equipment (PPE) in accordance with infection prevention guidance.3,12

Abbreviations: HBPC, home based primary care; HHA, home health agency; VHA, Veterans Health Administration.
aAdapted with permission from Wyte-Lake and Franzosa.8,9
Through a combination of its national and local health care networks, the VHA has a robust and well-positioned emergency infrastructure to supportcommunity-dwelling older adults during disasters.14 This network is supported by the VHA Office of Emergency Management, which shares resources and guidance with local emergency managers at each facility as well as individual programs such as the VHA Home Based Primary Care (HBPC) program, which provides 38,000 seriously ill veterans with home medical visits.15 Working closely with their local and national hospital networks and emergency managers, individual VHA HBPC programs were able to maintain the safety of staff and continuity of care for patients enrolled in HBPC by rapidly administering COVID-19 vaccines to patients, caregivers, and staff, and providing emergency assistance during the 2017 hurricane season.16,17 These efforts were successful because HBPC practitioners and their patients, had access to a level of emergency-related information, resources, and technology that are often out of reach for individual community-based health care practitioners (HCPs). The US Department of Veterans Affairs (VA) also supports local communities through its Fourth Mission, which provides emergency resources to non-VHA health care facilities (ie, hospitals and nursing homes) during national emergencies and natural disasters.17 Although there has been an expansion in the definition of shared resources, such as extending behavioral health support to local communities, the VHA has not historically provided these resources to HHAs.14
This study examines opportunities to leverage VHA emergency management resources to support contracted HHAs and inform other large health system emergency planning efforts. The findings from the exploratory phase are described in this article. We interviewed VHA emergency managers, HBPC and VA staff who coordinate home health care worker services, as well as administrators at contracted HHAs within a Veterans Integrated Services Network (VISN). These findings will inform the second (single-site pilot study) and third (feasibility study) phases. Our intent was to (1) better understand the relationships between VA medical centers (VAMCs) and their contracted HHAs; (2) identify existing VHA emergency protocols to support community-dwelling older adults; and (3) determine opportunities to build on existing infrastructure and relationships to better support contracted HHAs and their staff in emergencies.
Methods
The 18 VISNs act as regional systems of care that are loosely connected to better meet local health needs and maximize access to care. This study was conducted at 6 of 9 VAMCs within VISN 2, the New York/New Jersey VHA Health Care Network.18 VAMCs that serve urban, rural, and mixed urban/rural catchment areas were included.
Each VAMC has an emergency management program led by an emergency manager, an HBPC program led by a program director and medical director, and a community care or purchased care office that has a liaison who manages contracted home health care worker services. The studyfocused on HBPC programs because they are most likely to interact with veterans’ home health care workers in the home and care for community-dwelling veterans during emergencies. Each VHA also contracts with a series of local HHAs that generally have a dedicated staff member who interfaces with the VHA liaison. Our goal was to interview ≥ 1 emergency manager, ≥ 1 HBPC team member, ≥ 1 community care staff person, and ≥ 1 contracted home health agency administrator at each site to gain multiple perspectives from the range of HCPs serving veterans in the community.
Recruitment and Data Collection
The 6 sites were selected in consultation with VISN 2 leadership for their strong HBPC and emergency management programs. To recruit respondents, we contacted VISN and VAMC leads and used our professional networks to identify a sample of multidisciplinary individuals who represent both community care and HBPC programs who were contacted via email.
Since each VAMC is organized differently, we utilized a snowball sampling approach to identify the appropriate contacts.19 At the completion of each interview, we asked the participant to suggest additional contacts and introduce us to any remaining stakeholders (eg, the emergency manager) at that site or colleagues at other VISN facilities. Because roles vary among VAMCs, we contacted the person who most closely resembled the identified role and asked them to direct us to a more appropriate contact, if necessary. We asked community care managers to identify 1 to 2 agencies serving the highest volume of patients who are veterans at their site and requested interviews with those liaisons. This resulted in the recruitment of key stakeholders from 4 teams across the 6 sites (Table).
A semistructured interview guide was jointly developed based on constructs of interest, including relationships within VAMCs and between VAMCs and HHAs; existing emergency protocols and experience during disasters; and suggestions and opportunities for supporting agencies during emergencies and potential barriers. Two researchers (TWL and EF) who were trained in qualitative methods jointly conducted interviews using the interview guide, with 1 researcher leading and another taking notes and asking clarifying questions.
Interviews were conducted virtually via Microsoft Teams with respondents at their work locations between September 2022 and January 2023. Interviews were audio recorded and transcribed and 2 authors (TWL and ESO) reviewed transcripts for accuracy. Interviews averaged 47 minutes in length (range, 20-59).
The study was reviewed and determined to be exempt by institutional review boards at the James J. Peters VAMC and Greater Los Angeles VAMC. We asked participants for verbal consent to participate and preserved their confidentiality.
Analysis
Data were analyzed via an inductive approach, which involves drawing salient themes rather than imposing preconceived theories.20 Three researchers (TWL, EF, and ES) listened to and discussed 2 staff interviews and tagged text with specific codes (eg, communication between the VHA and HHA, internal communication, and barriers to case fulfillment) so the team could selectively return to the interview text for deeper analysis, allowing for the development of a final codebook. The project team synthesized the findings to identify higher-level themes, drawing comparisons across and within the respondent groups, including within and between health care systems. Throughout the analysis, we maintained analytic memos, documented discussions, and engaged in analyst triangulation to ensure trustworthiness.21,22 To ensure the analysis accurately reflected the participants’ understanding, we held 2 virtual member-checking sessions with participants to share preliminary findings and conclusions and solicit feedback. Analysis was conducted using ATLAS.ti version 20.
Results
VHA-based participants described internal emergency management systems that are deployed during a disaster to support patients and staff. Agency participants described their own internal emergency management protocols. Respondents discussed how and when the 2 intersected, as well as opportunities for future mutual support. The analysis identified several themes: (1) relationships between VAMC teams; (2) relationships between VHA and HHAs; (3) VHA and agencies responses during emergencies; (4) receptivity and opportunities for extending VHA resources into the community; and (5) barriers and facilitators to deeper engagement.
Relationships Within VHA (n = 17)
Staff at all VHA sites described close relationships between the internal emergency management and HBPC teams. HBPC teams identified patients who were most at risk during emergencies to triage those with the highest medical needs (eg, patients dependent on home infusion, oxygen, or electronic medical devices) and worked alongside emergency managers to develop plans to continue care during an emergency. HBPC representatives were part of their facilities’ local emergency response committees. Due to this close collaboration, VHA emergency managers were familiar with the needs of homebound veterans and caregivers. “I invite our [HBPC] program manager to attend [committee] meetings and … they’re part of the EOC [emergency operations center]," an emergency manager said. “We work together and I’m constantly in contact with that individual, especially during natural disasters and so forth, to ensure that everybody’s prepared in the community.”
On the other hand, community caremanagers—who described frequent interactions with HBPC teams, largely around coordinating and managing non-VHA home care services—were less likely to have direct relationships with their facility emergency managers. For example, when asked if they had a relationship with their emergency manager, a community care manager admitted, “I [only] know who he is.” They also did not report having structured protocols for veteran outreach during emergencies, “because all those veterans who are receiving [home health care worker] services also belong to a primary care team,” and considered the outreach to be the responsibility of the primary care team and HHA.
Relationships Between the VHA and HHAs (n = 17)
Communication between VAMCs and contracted agencies primarily went through community care managers, who described established long-term relationships with agency administrators. Communication was commonly restricted to operational activities, such as processing referrals and occasional troubleshooting. According to a community care manager most communication is “why haven’t you signed my orders?” There was a general sense from participants that communication was promptly answered, problems were addressed, and professional collegiality existed between the agencies as patients were referred and placed for services. One community care manager reported meeting with agencies regularly, noting, “I talk to them pretty much daily.”
If problems arose, community care managers described themselves as “the liaison” between agencies and VHA HCPs who ordered the referrals. This is particularly the case if the agency needed help finding a VHA clinician or addressing differences in care delivery protocols.
Responding During Emergencies (n = 19)
During emergencies, VHA and agency staff described following their own organization’s protocols and communicating with each other only on a case-by-case basis rather than through formal or systematic channels and had little knowledge of their counterpart’s emergency protocols. Beyond patient care, there was no evidence of information sharing between VHA and agency staff. Regarding sharing information with their local community, an HBPC Program Director said, “it’s almost like the VHA had become siloed” and operated on its own without engaging with community health systems or emergency managers.
Beyond the guidance provided by state departments of public health, HHAs described collaborating with other agencies in their network and relying on their informal professional network to manage the volume of information and updates they followed during emergencies like the COVID-19 pandemic. One agency administrator did not frequently communicate with VHA partners during the pandemic but explained that the local public health department helped work through challenges. However, “we realized pretty quickly they were overloaded and there was only so much they could do.” The agency administrator turned to a “sister agency” and local hospitals, noting, “Wherever you have connections in the field or in the industry, you know you’re going to reach out to people for guidance on policies and… protocol.”
Opportunities for Extending VHA Resources to the Community (n = 16)
All VHA emergency managers were receptive to extending support to community-based HCPS and, in some cases, felt strongly that they were an essential part of veterans’ care networks. Emergency managers offered examples for how they supportedcommunity-based HCPs, such as helping those in the VAMC medical foster home program develop and evaluate emergency plans. Many said they had not explicitly considered HHAs before (Appendix).
Emergency managers also described how supporting community-based HCPs could be considered within the scope of the VHA role and mission, specifically the Fourth Mission. “I think that we should be making our best effort to make sure that we’re also providing that same level [of protection] to the people taking care of the veteran [as our VHA staff],” an emergency manager said. “It’s our responsibility to provide the best for the staff that are going into those homes to take care of that patient.”
In many cases, emergency managers had already developed practical tools that could be easily shared outside the VHA, including weather alerts, trainings, emergency plan templates, and lists of community resources and shelters (Figure 2). A number of these examples built on existing communication channels. One emergency manager said that the extension of resources could be an opportunity to decrease the perceived isolation of home health care workers through regular

Abbreviations: PPE, personal protective equipment; VA, US Department of Veterans Affairs.
On the agency side, participants noted that some HHAs could benefit more from support than others. While some agencies are well staffed and have good protocols and keep up to date, “There are smaller agencies, agencies that are starting up that may not have the resources to just disseminate all the information. Those are the agencies [that] could well benefit from the VHA,” an HBPC medical director explained. Agency administrators suggested several areas where they would welcome support, including a deeper understanding of available community resources and access to PPE for staff. Regarding informational resources, an administrator said, “Anytime we can get information, it’s good to have it come to you and not always have to go out searching for it.”
Barriers and Facilitators to Partnering With Community Agencies (n = 16)
A primary barrier regarding resource sharing was potential misalignment between each organization’s policies. HHAs followed state and federal public health guidelines, which sometimes differed from VHA policies. Given that agencies care for both VHA and non-VHA clients, questions also arose around how agencies would prioritize information from the VHA, if they were already receiving information from other sources. When asked about information sharing, both VHA staff and agencies agreed staff time to support any additional activities should be weighed against the value of the information gained.
Six participants also shared that education around emergency preparedness could be an opportunity to bridge gaps between VAMCs and their surrounding communities.
Two emergency managers noted the need to be sensitive in the way they engaged with partners, respecting and building on the work that agencies were already doing in this area to ensure VHA was seen as a trusted partner and resource rather than trying to impose new policies or rules on community-based HCPs. “I know that like all leadership in various organizations, there’s a little bit of bristling going on when other people try and tell them what to do,” an HBPC medical director said. “However, if it is established that as a sort of greater level like a state level or a federal level, that VHA can be a resource. I think that as long as that’s recognized by their own professional organizations within each state, then I think that that would be a tremendous advantage to many agencies.”
In terms of sharing physical resources, emergency managers raised concerns around potential liability, although they also acknowledged this issue was important enough to think about potential workarounds. As one emergency manager said, “I want to know that my PPE is not compromised in any way shape or form and that I am in charge of that PPE, so to rely upon going to a home and hoping that [the PPE] wasn’t compromised … would kind of make me a little uneasy.” This emergency manager suggested possible solutions, such as creating a sealed PPE package to give directly to an aide.
Discussion
As the prevalence of climate-related disasters increases, the need to ensure the safety and independence of older adults during emergencies grows more urgent. Health systems must think beyond the direct services they provide and consider the community resources upon which their patients rely. While relationships did not formally exist between VHA emergency managers and community home health HCPs in the sample analyzed in this article, there is precedent and interest in supporting contracted home health agencies caring for veterans in the community. Although not historically part of the VA Fourth Mission, creating a pipeline of support for contracted HHAs by leveraging existing relationships and resources can potentially strengthen its mission to protect older veterans in emergencies, help them age safely in place, and provide a model for health systems to collaborate with community-based HCPs around emergency planning and response (Figure 3).23

Existing research on the value of health care coalitions highlights the need for established and growing partnerships with a focus on ensuring they are value-added, which echoes concerns we heard in interviews.24 Investment in community partnerships not only includes sharing supplies but also relying on bidirectional support that can be a trusted form of timely information.1,25 The findings in this study exhibit strong communication practices within the VHA during periods of nonemergency and underscore the untapped value of the pre-existing relationship between VAMCs and their contracted HHAs as an area of potential growth for health care coalitions.
Sharing resources in a way that does not put new demands on partners contributes to the sustainability and value-added nature of coalitions. Examples include establishing new low-investment practices (ie, information sharing) that support capacity and compliance with existing requirements rather than create new responsibilities for either member of the coalition. The relationship between the VHA emergency managers and the VHA HBPC program can act as a guide. The emergency managers interviewed for this study are currently engaged with HBPC programs and therefore understand the needs of homebound older adults and their caregivers. Extending the information already available to the HBPC teams via existing channels strengthens workforce practices and increased security for the shared patient, even without direct relationships between emergency managers and agencies. It is important to understand the limitations of these practices, including concerns around conflicting federal and state mandates, legal concerns around the liability of sharing physical resources (such as PPE), and awareness that the objective is not for the VHA to increase burdens (eg, increasing compliance requirements) but rather to serve as a resource for a mutual population in a shared community.
Offering training and practical resources to HHA home health care workers can help them meet disaster preparedness requirements. This is particularly important considering the growing home care workforce shortages, a topic mentioned by all HBPC and community care participants interviewed for this study.26,27 Home health care workers report feeling underprepared and isolated while on the job in normal conditions, a sentiment exacerbated by the COVID-19 pandemic.3,10 Supporting these individuals may help them feel more prepared and connected to their work, improving stability and quality of care.
While these issues are priorities within the VHA, there is growing recognition at the state and federal level of the importance of including older adults and their HCPs in disaster preparedness and response.5,28 The US Department of Health and Human Services, for example, includes older adults and organizations that serve them on its National Advisory Committee on Seniors and Disasters. The Senate version of the 2023 reauthorization of the Pandemic and All-Hazards Preparedness and Response Act included specific provisions to support community-dwelling older adults and people with disabilities, incorporating funding for community organizations to support continuity of services and avoid institutionalization in an emergency.29 Other proposed legislation includes the Real Emergency Access for Aging and Disability Inclusion for Disasters Act, which would ensure the needs of older adults and people with disabilities are explicitly included in all phases of emergency planning and response.30
The VHA expansion of the its VEText program to include disaster response is an effort to more efficiently extend outreach to older and vulnerable patients who are veterans.31 Given these growing efforts, the VHA and other health systems have an opportunity to expand internal emergency preparedness efforts to ensure the health and safety of individuals living in the community.
Limitations
VISN 2 has been a target of terrorism and other disasters. In addition to the sites being initially recruited for their strong emergency management protocols, this context may have biased respondents who are favorable to extending their resources into the community. At the time of recruitment, contracted HHAs were still experiencing staff shortages due to the COVID-19 pandemic, which limited the ability of agency staff to participate in interviews. Additionally, while the comprehensive exploration of VISN 2 facilities allows for confidence of the organizational structures described, the qualitative research design and small study sample, the study findings cannot be immediately generalized to all VISNs.
Conclusions
Many older veterans increasingly rely on home health care workers to age safely. The VHA, as a large national health care system and leader in emergency preparedness, could play an important role in supporting home health care workers and ameliorating their sense of isolation during emergencies and natural disasters. Leveraging existing resources and relationships may be a low-cost, low-effort opportunity to build higher-level interventions that support the needs of patients. Future research and work in this field, including the authors’ ongoing work, will expand agency participation and engage agency staff in conceptualizing pilot projects to ensure they are viable and feasible for the field.

- Barnett DJ, Knieser L, Errett NA, Rosenblum AJ, Seshamani M, Kirsch TD. Reexamining health-care coalitions in light of COVID-19. Disaster Med public Health Prep. 2022;16(3):859-863. doi:10.1017/dmp.2020.431
- Wulff K, Donato D, Lurie N. What is health resilience and how can we build it? Annu Rev Public Health. 2015;36:361-374. doi:10.1146/annurev-publhealth-031914-122829
- Franzosa E, Wyte-Lake T, Tsui EK, Reckrey JM, Sterling MR. Essential but excluded: building disaster preparedness capacity for home health care workers and home care agencies. J Am Med Dir Assoc. 2022;23(12):1990-1996. doi:10.1016/j.jamda.2022.09.012
- Miner S, Masci L, Chimenti C, Rin N, Mann A, Noonan B. An outreach phone call project: using home health to reach isolated community dwelling adults during the COVID 19 lockdown. J Community Health. 2022;47(2):266-272. doi:10.1007/s10900-021-01044-6
- National Institute on Aging. Protecting older adults from the effects of natural disasters and extreme weather. October 18, 2022. Accessed August 19, 2024. https://www.nia.nih.gov/news/protecting-older-adults-effects-natural-disasters-and-extreme-weather
- PHI. Direct Care Workers in the United States: Key Facts. September 7, 2021. Accessed August 19, 2024. https://www.phinational.org/resource/direct-care-workers-in-the-united-states-key-facts-2/
- Centers for Medicare & Medicaid Services. Emergency Preparedness Rule. September 8, 2016. Updated September 6, 2023. Accessed August 19, 2024. https://www.cms.gov/medicare/health-safety-standards/quality-safety-oversight-emergency-preparedness/emergency-preparedness-rule
- Wyte-Lake T, Claver M, Tubbesing S, Davis D, Dobalian A. Development of a home health patient assessment tool for disaster planning. Gerontology. 2019;65(4):353-361. doi:10.1159/000494971
- Franzosa E, Judon KM, Gottesman EM, et al. Home health aides’ increased role in supporting older veterans and primary healthcare teams during COVID-19: a qualitative analysis. J Gen Intern Med. 2022;37(8):1830-1837. doi:10.1007/s11606-021-07271-w
- Franzosa E, Tsui EK, Baron S. “Who’s caring for us?”: understanding and addressing the effects of emotional labor on home health aides’ well-being. Gerontologist. 2019;59(6):1055-1064. doi:10.1093/geront/gny099
- Osakwe ZT, Osborne JC, Samuel T, et al. All alone: a qualitative study of home health aides’ experiences during the COVID-19 pandemic in New York. Am J Infect Control. 2021;49(11):1362-1368. doi:10.1016/j.ajic.2021.08.004
- Feldman PH, Russell D, Onorato N, et al. Ensuring the safety of the home health aide workforce and the continuation of essential patient care through sustainable pandemic preparedness. July 2022. Accessed August 19, 2024. https://www.vnshealth.org/wp-content/uploads/2022/08/Pandemic_Preparedness_IB_07_21_22.pdf
- Sterling MR, Tseng E, Poon A, et al. Experiences of home health care workers in New York City during the coronavirus disease 2019 pandemic: a qualitative analysis. JAMA Internal Med. 2020;180(11):1453-1459. doi:10.1001/jamainternmed.2020.3930
- Wyte-Lake T, Schmitz S, Kornegay RJ, Acevedo F, Dobalian A. Three case studies of community behavioral health support from the US Department of Veterans Affairs after disasters. BMC Public Health. 2021;21(1):639. doi:10.1186/s12889-021-10650-x
- Beales JL, Edes T. Veteran’s affairs home based primary care. Clin Geriatr Med. 2009;25(1):149-ix. doi:10.1016/j.cger.2008.11.002
- Wyte-Lake T, Manheim C, Gillespie SM, Dobalian A, Haverhals LM. COVID-19 vaccination in VA home based primary care: experience of interdisciplinary team members. J Am Med Dir Assoc. 2022;23(6):917-922. doi:10.1016/j.jamda.2022.03.014
- Wyte-Lake T, Schmitz S, Cosme Torres-Sabater R, Dobalian A. Case study of VA Caribbean Healthcare System’s community response to Hurricane Maria. J Emerg Manag. 2022;19(8):189-199. doi:10.5055/jem.0536
- US Department of Veterans Affairs. New York/New Jersey VA Health Care Network, VISN 2 Locations. Updated January 3, 2024. Accessed August 19, 2024. https://www.visn2.va.gov/visn2/facilities.asp
- Noy C. Sampling knowledge: the hermeneutics of snowball sampling in qualitative research. Int J Soc Res Methodol. 2008;11(4):327-344. doi:10.1080/13645570701401305
- Ritchie J, Lewis J, Nicholls CM, Ormston R, eds. Qualitative Research Practice: A Guide for Social Science Students and Researchers. 2nd ed. Sage; 2013.
- Morrow SL. Quality and trustworthiness in qualitative research in counseling psychology. J Couns Psychol. 2005;52(2):250-260. doi:10.1037/0022-0167.52.2.250
- Rolfe G. Validity, trustworthiness and rigour: quality and the idea of qualitative research. J Adv Nurs. 2006;53(3):304-310. doi:10.1111/j.1365-2648.2006.03727.x
- Schmitz S, Wyte-Lake T, Dobalian A. Facilitators and barriers to preparedness partnerships: a veterans affairs medical center perspective. Disaster Med Public Health Prep. 2018;12(4):431-436. doi:10.1017/dmp.2017.92
- Koch AE, Bohn J, Corvin JA, Seaberg J. Maturing into high-functioning health-care coalitions: a qualitative Nationwide study of emergency preparedness and response leadership. Disaster Med Public Health Prep. 2022;17:e111. doi:10.1017/dmp.2022.13
- Lin JS, Webber EM, Bean SI, Martin AM, Davies MC. Rapid evidence review: policy actions for the integration of public health and health care in the United States. Front Public Health. 2023;11:1098431. doi:10.3389/fpubh.2023.1098431
- Watts MOM, Burns A, Ammula M. Ongoing impacts of the pandemic on medicaid home & community-based services (HCBS) programs: findings from a 50-state survey. November 28, 2022. Accessed August 19, 2024. https://www.kff.org/medicaid/issue-brief/ongoing-impacts-of-the-pandemic-on-medicaid-home-community-based-services-hcbs-programs-findings-from-a-50-state-survey/
- Kreider AR, Werner RM. The home care workforce has not kept pace with growth in home and community-based services. Health Aff (Millwood). 2023;42(5):650-657. doi:10.1377/hlthaff.2022.01351
- FEMA introduces disaster preparedness guide for older adults. News release. FEMA. September 20, 2023. Accessed August 19, 2024. https://www.fema.gov/press-release/20230920/fema-introduces-disaster-preparedness-guide-older-adults
- Pandemic and All-Hazards Preparedness and Response Act, S 2333, 118th Cong, 1st Sess (2023). https://www.congress.gov/bill/118th-congress/senate-bill/2333/text
- REAADI for Disasters Act, HR 2371, 118th Cong, 1st Sess (2023). https://www.congress.gov/bill/118th-congress/house-bill/2371
- Wyte-Lake T, Brewster P, Hubert T, Gin J, Davis D, Dobalian A. VA’s experience building capability to conduct outreach to vulnerable patients during emergencies. Innov Aging. 2023;7(suppl 1):209. doi:10.1093/geroni/igad104.0690
- Barnett DJ, Knieser L, Errett NA, Rosenblum AJ, Seshamani M, Kirsch TD. Reexamining health-care coalitions in light of COVID-19. Disaster Med public Health Prep. 2022;16(3):859-863. doi:10.1017/dmp.2020.431
- Wulff K, Donato D, Lurie N. What is health resilience and how can we build it? Annu Rev Public Health. 2015;36:361-374. doi:10.1146/annurev-publhealth-031914-122829
- Franzosa E, Wyte-Lake T, Tsui EK, Reckrey JM, Sterling MR. Essential but excluded: building disaster preparedness capacity for home health care workers and home care agencies. J Am Med Dir Assoc. 2022;23(12):1990-1996. doi:10.1016/j.jamda.2022.09.012
- Miner S, Masci L, Chimenti C, Rin N, Mann A, Noonan B. An outreach phone call project: using home health to reach isolated community dwelling adults during the COVID 19 lockdown. J Community Health. 2022;47(2):266-272. doi:10.1007/s10900-021-01044-6
- National Institute on Aging. Protecting older adults from the effects of natural disasters and extreme weather. October 18, 2022. Accessed August 19, 2024. https://www.nia.nih.gov/news/protecting-older-adults-effects-natural-disasters-and-extreme-weather
- PHI. Direct Care Workers in the United States: Key Facts. September 7, 2021. Accessed August 19, 2024. https://www.phinational.org/resource/direct-care-workers-in-the-united-states-key-facts-2/
- Centers for Medicare & Medicaid Services. Emergency Preparedness Rule. September 8, 2016. Updated September 6, 2023. Accessed August 19, 2024. https://www.cms.gov/medicare/health-safety-standards/quality-safety-oversight-emergency-preparedness/emergency-preparedness-rule
- Wyte-Lake T, Claver M, Tubbesing S, Davis D, Dobalian A. Development of a home health patient assessment tool for disaster planning. Gerontology. 2019;65(4):353-361. doi:10.1159/000494971
- Franzosa E, Judon KM, Gottesman EM, et al. Home health aides’ increased role in supporting older veterans and primary healthcare teams during COVID-19: a qualitative analysis. J Gen Intern Med. 2022;37(8):1830-1837. doi:10.1007/s11606-021-07271-w
- Franzosa E, Tsui EK, Baron S. “Who’s caring for us?”: understanding and addressing the effects of emotional labor on home health aides’ well-being. Gerontologist. 2019;59(6):1055-1064. doi:10.1093/geront/gny099
- Osakwe ZT, Osborne JC, Samuel T, et al. All alone: a qualitative study of home health aides’ experiences during the COVID-19 pandemic in New York. Am J Infect Control. 2021;49(11):1362-1368. doi:10.1016/j.ajic.2021.08.004
- Feldman PH, Russell D, Onorato N, et al. Ensuring the safety of the home health aide workforce and the continuation of essential patient care through sustainable pandemic preparedness. July 2022. Accessed August 19, 2024. https://www.vnshealth.org/wp-content/uploads/2022/08/Pandemic_Preparedness_IB_07_21_22.pdf
- Sterling MR, Tseng E, Poon A, et al. Experiences of home health care workers in New York City during the coronavirus disease 2019 pandemic: a qualitative analysis. JAMA Internal Med. 2020;180(11):1453-1459. doi:10.1001/jamainternmed.2020.3930
- Wyte-Lake T, Schmitz S, Kornegay RJ, Acevedo F, Dobalian A. Three case studies of community behavioral health support from the US Department of Veterans Affairs after disasters. BMC Public Health. 2021;21(1):639. doi:10.1186/s12889-021-10650-x
- Beales JL, Edes T. Veteran’s affairs home based primary care. Clin Geriatr Med. 2009;25(1):149-ix. doi:10.1016/j.cger.2008.11.002
- Wyte-Lake T, Manheim C, Gillespie SM, Dobalian A, Haverhals LM. COVID-19 vaccination in VA home based primary care: experience of interdisciplinary team members. J Am Med Dir Assoc. 2022;23(6):917-922. doi:10.1016/j.jamda.2022.03.014
- Wyte-Lake T, Schmitz S, Cosme Torres-Sabater R, Dobalian A. Case study of VA Caribbean Healthcare System’s community response to Hurricane Maria. J Emerg Manag. 2022;19(8):189-199. doi:10.5055/jem.0536
- US Department of Veterans Affairs. New York/New Jersey VA Health Care Network, VISN 2 Locations. Updated January 3, 2024. Accessed August 19, 2024. https://www.visn2.va.gov/visn2/facilities.asp
- Noy C. Sampling knowledge: the hermeneutics of snowball sampling in qualitative research. Int J Soc Res Methodol. 2008;11(4):327-344. doi:10.1080/13645570701401305
- Ritchie J, Lewis J, Nicholls CM, Ormston R, eds. Qualitative Research Practice: A Guide for Social Science Students and Researchers. 2nd ed. Sage; 2013.
- Morrow SL. Quality and trustworthiness in qualitative research in counseling psychology. J Couns Psychol. 2005;52(2):250-260. doi:10.1037/0022-0167.52.2.250
- Rolfe G. Validity, trustworthiness and rigour: quality and the idea of qualitative research. J Adv Nurs. 2006;53(3):304-310. doi:10.1111/j.1365-2648.2006.03727.x
- Schmitz S, Wyte-Lake T, Dobalian A. Facilitators and barriers to preparedness partnerships: a veterans affairs medical center perspective. Disaster Med Public Health Prep. 2018;12(4):431-436. doi:10.1017/dmp.2017.92
- Koch AE, Bohn J, Corvin JA, Seaberg J. Maturing into high-functioning health-care coalitions: a qualitative Nationwide study of emergency preparedness and response leadership. Disaster Med Public Health Prep. 2022;17:e111. doi:10.1017/dmp.2022.13
- Lin JS, Webber EM, Bean SI, Martin AM, Davies MC. Rapid evidence review: policy actions for the integration of public health and health care in the United States. Front Public Health. 2023;11:1098431. doi:10.3389/fpubh.2023.1098431
- Watts MOM, Burns A, Ammula M. Ongoing impacts of the pandemic on medicaid home & community-based services (HCBS) programs: findings from a 50-state survey. November 28, 2022. Accessed August 19, 2024. https://www.kff.org/medicaid/issue-brief/ongoing-impacts-of-the-pandemic-on-medicaid-home-community-based-services-hcbs-programs-findings-from-a-50-state-survey/
- Kreider AR, Werner RM. The home care workforce has not kept pace with growth in home and community-based services. Health Aff (Millwood). 2023;42(5):650-657. doi:10.1377/hlthaff.2022.01351
- FEMA introduces disaster preparedness guide for older adults. News release. FEMA. September 20, 2023. Accessed August 19, 2024. https://www.fema.gov/press-release/20230920/fema-introduces-disaster-preparedness-guide-older-adults
- Pandemic and All-Hazards Preparedness and Response Act, S 2333, 118th Cong, 1st Sess (2023). https://www.congress.gov/bill/118th-congress/senate-bill/2333/text
- REAADI for Disasters Act, HR 2371, 118th Cong, 1st Sess (2023). https://www.congress.gov/bill/118th-congress/house-bill/2371
- Wyte-Lake T, Brewster P, Hubert T, Gin J, Davis D, Dobalian A. VA’s experience building capability to conduct outreach to vulnerable patients during emergencies. Innov Aging. 2023;7(suppl 1):209. doi:10.1093/geroni/igad104.0690