User login
Should Hospitalists Who Fail to Provide a Standard of Care Be Paid for Subsequent Care?
A 72-year-old male with a history of CHF is admitted for elective total hip arthroplasty. On postoperative day one, he develops dyspnea and hypoxia, and is diagnosed with acute pulmonary edema by the hospitalist co-managing his care. Furosemide is prescribed, and he improves, and by day four is ready for discharge following another dose of diuretics. Overnight, he develops acute onset of shortness of breath and is diagnosed with a pulmonary embolism (PE). Under new regulations, the hospital will not be reimbursed for the extra cost associated with subsequent patient care. Should the hospitalist be paid for the subsequent care?
PRO
Nonpayment won’t improve quality or significantly decrease costs
The real essence of the question raised in the clinical case above is “Should doctors profit from errors?” The answer might be “It’s better than the alternative.” Allow me to explain. There essentially are two reasons to withhold payment in this scenario: one, as a mechanism for promoting quality; two, as a mechanism for decreasing costs to the payor.
The quality argument assumes the physician will deliver higher-quality care (i.e., prescribe chemical thromboprophylaxis) if a threat of nonpayment exists. This concept is simply hogwash. If expensive medical malpractice threats fail as quality-improvement (QI) mechanisms, it is absurd to think withholding a few subsequent-care charges will generate better results.
The key issue is the type of error involved. As defined by Lucien Leape, MD, in his celebrated 1994 article on medical errors, “mistakes” reflect failures during attentional behaviors, or incorrect choices.1 “Slips” reflect lapses in concentration. “Slips occur in the face of competing sensory or emotional distractions, fatigue, and stress,” and “reducing the risk of slips requires attention to the designs of protocols, devices, and work environments.”
Misjudging the type of error—in this case, a slip (find me a hospitalist who doesn’t know total hip arthroplasty requires thrombophrophylaxis)—and misapplying corrective actions will have little to no effect on outcomes. Thus, pay-withholding schemes can have a negative net effect by diverting resources from QI projects that truly improve patient outcomes.
Withholding payment in this case generates approximately $160 in direct savings to the payor (assuming Medicare payments for one 99233 and two 99232 subsequent care visits), yet the operational costs are not negligible and must be factored into the equation. The payor needs to first determine who is truly at fault: the hospitalist or the orthopedic surgeon. Answering that question requires the payor to review the co-management agreement, perhaps aided by an attorney. That’s a costly endeavor.
For the sake of argument, let’s assume in this case the hospitalist is at fault. The next step is determining if the hospitalist who failed to prescribe prophylaxis prior to the PE is the same hospitalist caring for the patient after the PE. It is inappropriate to withhold payment to hospitalist A if hospitalist B made the error. Again, significant manpower will be required to determine fault, as this is not information one finds on a UB-04 claim form submitted to Medicare.
Further eroding the $160 savings is the cost of determining whether a contraindication exists: Bleeding ulcer? Subdural hematoma? Heparinoid allergy? Let us not forget the additional costs in copying, shipping, warehousing, and eventual shredding of the records. One can readily see that the operational costs can quickly negate the $160 anticipated savings. In fact, it’s likely a negative return on investment.
Clearly, withholding payment in this scenario is an ineffective mechanism for improving quality or decreasing cost. I am not generally a proponent of rewarding failure, and perhaps as we usher in a new era of healthcare reform, the system will be redesigned in such a way that better aligns quality and cost-control measures. However, under the current system, payment denial as outlined above likely does more harm than good.
CON
Withhold payment when medical errors are easily identifiable
When I first learned of the proposal to withhold Medicare payment for hospital-acquired conditions (HACs), I had mixed emotions. On the one hand, I firmly believe that physicians should be accountable for their work; on the other hand, this policy seems to conflict sharply with the “no blame” mantra that has been prevalent in patient safety for more than a decade.2 More recently, though, many have argued for balancing the pursuit of system fixes for quality and patient-safety issues with the development of a culture of accountability.3
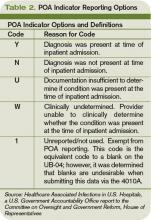
In theory, the HACs should meet the following criteria: They should be high-cost conditions, high-volume conditions, or both; they should be identifiable through ICD-9-CM coding as complicating conditions (CCs) or major complicating conditions (MCCs) that result in a higher-paying MS-DRG; and they should be reasonably preventable through the application of evidence-based guidelines. Some HACs are jaw-dropping lapses in care (e.g., leaving foreign bodies in during surgery). Other HACs seem to me to be much less preventable, especially fall injuries and catheter-associated urinary tract infections (UTIs). Several experts have written eloquently regarding the limitations of these new measures, particularly emphasizing the potential for increased administrative burden on hospitals and the potential for unintended consequences.4,5
However, in the case described above involving a hospitalist, I have no reservations in limiting payment to the provider. To me, failing to prescribe VTE prophylaxis in an elderly, immobilized, post-op hip replacement patient with a CHF exacerbation is the hospitalist’s equivalent to a surgeon leaving behind a sponge in an appendectomy. It also meets the elements outlined in the HAC withholding program:
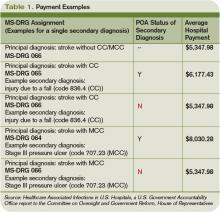
- It is high-cost. The 2007 MS-DRG payment for elective hip arthroplasty was $9,863, but adding an MCC increased that cost by one-third.6
- It is readily identifiable, though one concern might be that hospitals would perform unnecessary pre-operative testing to identify asymptomatic DVT, incurring increased testing and treatment costs and increasing the incidence of bleeding complications.
- It is very preventable. Without thromboprophylaxis, 40% to 60% of hip arthroplasty patients will develop an asymptomatic DVT, and 1 in 300 will die from a PE. However, such fatal events are exceedingly rare with appropriate prevention.7
Ultimately, I think a policy of nonpayment for this case keeps with the culture of accountability we need to foster in healthcare. The financial implications of nonpayment will drive hospital innovation and force the hospital to police provider behavior in more effective ways. This is likely to be a painful process, similar to the tribulations experienced with implementing pay-for-performance programs. The Centers for Medicare and Medicaid Services (CMS) needs to be flexible in adding—and removing—new HACs based on good evidence.
Regardless, the goal of achieving a safer, more effective healthcare system remains.
References
- Leape LL. Error in medicine. JAMA. 1994;272(23):1851-1857.
- Institute of Medicine. To Err Is Human: Building a Safer Healthcare System. Washington, D.C.: National Academies Press; 2000.
- Wachter RM, Pronovost PJ. Balancing “no blame” with accountability in patient safety. N Engl J Med. 2009;361:1401-1406.
- Saint S, Meddings JA, Calfee D, Kowalski CP, Krein SL. Catheter-associated urinary tract infection and the Medicare rule changes. Ann Intern Med. 2009;150(12):877-884.
- Inouye SK, Brown CJ, Tinetti ME. Medicare nonpayment, hospital falls, and unintended consequences. N Engl J Med. 2009;360(23):2390-2393.
- Wachter RM, Foster NE, Dudley RA. Medicare’s decision to withhold payment for hospital errors: the devil is in the det. Jt Comm J Qual Patient Saf. 2008;34(2):116-123.
- Geerts WH, Bergqvist D, Pineo GF, et al. Prevention of venous thromboembolism: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines (8th Edition). Chest. 2008;133(6 Suppl):381S-453S.
The opinions expressed herein are those of the authors and do not represent those of SHM or The Hospitalist.
A 72-year-old male with a history of CHF is admitted for elective total hip arthroplasty. On postoperative day one, he develops dyspnea and hypoxia, and is diagnosed with acute pulmonary edema by the hospitalist co-managing his care. Furosemide is prescribed, and he improves, and by day four is ready for discharge following another dose of diuretics. Overnight, he develops acute onset of shortness of breath and is diagnosed with a pulmonary embolism (PE). Under new regulations, the hospital will not be reimbursed for the extra cost associated with subsequent patient care. Should the hospitalist be paid for the subsequent care?
PRO
Nonpayment won’t improve quality or significantly decrease costs
The real essence of the question raised in the clinical case above is “Should doctors profit from errors?” The answer might be “It’s better than the alternative.” Allow me to explain. There essentially are two reasons to withhold payment in this scenario: one, as a mechanism for promoting quality; two, as a mechanism for decreasing costs to the payor.
The quality argument assumes the physician will deliver higher-quality care (i.e., prescribe chemical thromboprophylaxis) if a threat of nonpayment exists. This concept is simply hogwash. If expensive medical malpractice threats fail as quality-improvement (QI) mechanisms, it is absurd to think withholding a few subsequent-care charges will generate better results.
The key issue is the type of error involved. As defined by Lucien Leape, MD, in his celebrated 1994 article on medical errors, “mistakes” reflect failures during attentional behaviors, or incorrect choices.1 “Slips” reflect lapses in concentration. “Slips occur in the face of competing sensory or emotional distractions, fatigue, and stress,” and “reducing the risk of slips requires attention to the designs of protocols, devices, and work environments.”
Misjudging the type of error—in this case, a slip (find me a hospitalist who doesn’t know total hip arthroplasty requires thrombophrophylaxis)—and misapplying corrective actions will have little to no effect on outcomes. Thus, pay-withholding schemes can have a negative net effect by diverting resources from QI projects that truly improve patient outcomes.
Withholding payment in this case generates approximately $160 in direct savings to the payor (assuming Medicare payments for one 99233 and two 99232 subsequent care visits), yet the operational costs are not negligible and must be factored into the equation. The payor needs to first determine who is truly at fault: the hospitalist or the orthopedic surgeon. Answering that question requires the payor to review the co-management agreement, perhaps aided by an attorney. That’s a costly endeavor.
For the sake of argument, let’s assume in this case the hospitalist is at fault. The next step is determining if the hospitalist who failed to prescribe prophylaxis prior to the PE is the same hospitalist caring for the patient after the PE. It is inappropriate to withhold payment to hospitalist A if hospitalist B made the error. Again, significant manpower will be required to determine fault, as this is not information one finds on a UB-04 claim form submitted to Medicare.
Further eroding the $160 savings is the cost of determining whether a contraindication exists: Bleeding ulcer? Subdural hematoma? Heparinoid allergy? Let us not forget the additional costs in copying, shipping, warehousing, and eventual shredding of the records. One can readily see that the operational costs can quickly negate the $160 anticipated savings. In fact, it’s likely a negative return on investment.
Clearly, withholding payment in this scenario is an ineffective mechanism for improving quality or decreasing cost. I am not generally a proponent of rewarding failure, and perhaps as we usher in a new era of healthcare reform, the system will be redesigned in such a way that better aligns quality and cost-control measures. However, under the current system, payment denial as outlined above likely does more harm than good.
CON
Withhold payment when medical errors are easily identifiable
When I first learned of the proposal to withhold Medicare payment for hospital-acquired conditions (HACs), I had mixed emotions. On the one hand, I firmly believe that physicians should be accountable for their work; on the other hand, this policy seems to conflict sharply with the “no blame” mantra that has been prevalent in patient safety for more than a decade.2 More recently, though, many have argued for balancing the pursuit of system fixes for quality and patient-safety issues with the development of a culture of accountability.3
In theory, the HACs should meet the following criteria: They should be high-cost conditions, high-volume conditions, or both; they should be identifiable through ICD-9-CM coding as complicating conditions (CCs) or major complicating conditions (MCCs) that result in a higher-paying MS-DRG; and they should be reasonably preventable through the application of evidence-based guidelines. Some HACs are jaw-dropping lapses in care (e.g., leaving foreign bodies in during surgery). Other HACs seem to me to be much less preventable, especially fall injuries and catheter-associated urinary tract infections (UTIs). Several experts have written eloquently regarding the limitations of these new measures, particularly emphasizing the potential for increased administrative burden on hospitals and the potential for unintended consequences.4,5
However, in the case described above involving a hospitalist, I have no reservations in limiting payment to the provider. To me, failing to prescribe VTE prophylaxis in an elderly, immobilized, post-op hip replacement patient with a CHF exacerbation is the hospitalist’s equivalent to a surgeon leaving behind a sponge in an appendectomy. It also meets the elements outlined in the HAC withholding program:
- It is high-cost. The 2007 MS-DRG payment for elective hip arthroplasty was $9,863, but adding an MCC increased that cost by one-third.6
- It is readily identifiable, though one concern might be that hospitals would perform unnecessary pre-operative testing to identify asymptomatic DVT, incurring increased testing and treatment costs and increasing the incidence of bleeding complications.
- It is very preventable. Without thromboprophylaxis, 40% to 60% of hip arthroplasty patients will develop an asymptomatic DVT, and 1 in 300 will die from a PE. However, such fatal events are exceedingly rare with appropriate prevention.7
Ultimately, I think a policy of nonpayment for this case keeps with the culture of accountability we need to foster in healthcare. The financial implications of nonpayment will drive hospital innovation and force the hospital to police provider behavior in more effective ways. This is likely to be a painful process, similar to the tribulations experienced with implementing pay-for-performance programs. The Centers for Medicare and Medicaid Services (CMS) needs to be flexible in adding—and removing—new HACs based on good evidence.
Regardless, the goal of achieving a safer, more effective healthcare system remains.
References
- Leape LL. Error in medicine. JAMA. 1994;272(23):1851-1857.
- Institute of Medicine. To Err Is Human: Building a Safer Healthcare System. Washington, D.C.: National Academies Press; 2000.
- Wachter RM, Pronovost PJ. Balancing “no blame” with accountability in patient safety. N Engl J Med. 2009;361:1401-1406.
- Saint S, Meddings JA, Calfee D, Kowalski CP, Krein SL. Catheter-associated urinary tract infection and the Medicare rule changes. Ann Intern Med. 2009;150(12):877-884.
- Inouye SK, Brown CJ, Tinetti ME. Medicare nonpayment, hospital falls, and unintended consequences. N Engl J Med. 2009;360(23):2390-2393.
- Wachter RM, Foster NE, Dudley RA. Medicare’s decision to withhold payment for hospital errors: the devil is in the det. Jt Comm J Qual Patient Saf. 2008;34(2):116-123.
- Geerts WH, Bergqvist D, Pineo GF, et al. Prevention of venous thromboembolism: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines (8th Edition). Chest. 2008;133(6 Suppl):381S-453S.
The opinions expressed herein are those of the authors and do not represent those of SHM or The Hospitalist.
A 72-year-old male with a history of CHF is admitted for elective total hip arthroplasty. On postoperative day one, he develops dyspnea and hypoxia, and is diagnosed with acute pulmonary edema by the hospitalist co-managing his care. Furosemide is prescribed, and he improves, and by day four is ready for discharge following another dose of diuretics. Overnight, he develops acute onset of shortness of breath and is diagnosed with a pulmonary embolism (PE). Under new regulations, the hospital will not be reimbursed for the extra cost associated with subsequent patient care. Should the hospitalist be paid for the subsequent care?
PRO
Nonpayment won’t improve quality or significantly decrease costs
The real essence of the question raised in the clinical case above is “Should doctors profit from errors?” The answer might be “It’s better than the alternative.” Allow me to explain. There essentially are two reasons to withhold payment in this scenario: one, as a mechanism for promoting quality; two, as a mechanism for decreasing costs to the payor.
The quality argument assumes the physician will deliver higher-quality care (i.e., prescribe chemical thromboprophylaxis) if a threat of nonpayment exists. This concept is simply hogwash. If expensive medical malpractice threats fail as quality-improvement (QI) mechanisms, it is absurd to think withholding a few subsequent-care charges will generate better results.
The key issue is the type of error involved. As defined by Lucien Leape, MD, in his celebrated 1994 article on medical errors, “mistakes” reflect failures during attentional behaviors, or incorrect choices.1 “Slips” reflect lapses in concentration. “Slips occur in the face of competing sensory or emotional distractions, fatigue, and stress,” and “reducing the risk of slips requires attention to the designs of protocols, devices, and work environments.”
Misjudging the type of error—in this case, a slip (find me a hospitalist who doesn’t know total hip arthroplasty requires thrombophrophylaxis)—and misapplying corrective actions will have little to no effect on outcomes. Thus, pay-withholding schemes can have a negative net effect by diverting resources from QI projects that truly improve patient outcomes.
Withholding payment in this case generates approximately $160 in direct savings to the payor (assuming Medicare payments for one 99233 and two 99232 subsequent care visits), yet the operational costs are not negligible and must be factored into the equation. The payor needs to first determine who is truly at fault: the hospitalist or the orthopedic surgeon. Answering that question requires the payor to review the co-management agreement, perhaps aided by an attorney. That’s a costly endeavor.
For the sake of argument, let’s assume in this case the hospitalist is at fault. The next step is determining if the hospitalist who failed to prescribe prophylaxis prior to the PE is the same hospitalist caring for the patient after the PE. It is inappropriate to withhold payment to hospitalist A if hospitalist B made the error. Again, significant manpower will be required to determine fault, as this is not information one finds on a UB-04 claim form submitted to Medicare.
Further eroding the $160 savings is the cost of determining whether a contraindication exists: Bleeding ulcer? Subdural hematoma? Heparinoid allergy? Let us not forget the additional costs in copying, shipping, warehousing, and eventual shredding of the records. One can readily see that the operational costs can quickly negate the $160 anticipated savings. In fact, it’s likely a negative return on investment.
Clearly, withholding payment in this scenario is an ineffective mechanism for improving quality or decreasing cost. I am not generally a proponent of rewarding failure, and perhaps as we usher in a new era of healthcare reform, the system will be redesigned in such a way that better aligns quality and cost-control measures. However, under the current system, payment denial as outlined above likely does more harm than good.
CON
Withhold payment when medical errors are easily identifiable
When I first learned of the proposal to withhold Medicare payment for hospital-acquired conditions (HACs), I had mixed emotions. On the one hand, I firmly believe that physicians should be accountable for their work; on the other hand, this policy seems to conflict sharply with the “no blame” mantra that has been prevalent in patient safety for more than a decade.2 More recently, though, many have argued for balancing the pursuit of system fixes for quality and patient-safety issues with the development of a culture of accountability.3
In theory, the HACs should meet the following criteria: They should be high-cost conditions, high-volume conditions, or both; they should be identifiable through ICD-9-CM coding as complicating conditions (CCs) or major complicating conditions (MCCs) that result in a higher-paying MS-DRG; and they should be reasonably preventable through the application of evidence-based guidelines. Some HACs are jaw-dropping lapses in care (e.g., leaving foreign bodies in during surgery). Other HACs seem to me to be much less preventable, especially fall injuries and catheter-associated urinary tract infections (UTIs). Several experts have written eloquently regarding the limitations of these new measures, particularly emphasizing the potential for increased administrative burden on hospitals and the potential for unintended consequences.4,5
However, in the case described above involving a hospitalist, I have no reservations in limiting payment to the provider. To me, failing to prescribe VTE prophylaxis in an elderly, immobilized, post-op hip replacement patient with a CHF exacerbation is the hospitalist’s equivalent to a surgeon leaving behind a sponge in an appendectomy. It also meets the elements outlined in the HAC withholding program:
- It is high-cost. The 2007 MS-DRG payment for elective hip arthroplasty was $9,863, but adding an MCC increased that cost by one-third.6
- It is readily identifiable, though one concern might be that hospitals would perform unnecessary pre-operative testing to identify asymptomatic DVT, incurring increased testing and treatment costs and increasing the incidence of bleeding complications.
- It is very preventable. Without thromboprophylaxis, 40% to 60% of hip arthroplasty patients will develop an asymptomatic DVT, and 1 in 300 will die from a PE. However, such fatal events are exceedingly rare with appropriate prevention.7
Ultimately, I think a policy of nonpayment for this case keeps with the culture of accountability we need to foster in healthcare. The financial implications of nonpayment will drive hospital innovation and force the hospital to police provider behavior in more effective ways. This is likely to be a painful process, similar to the tribulations experienced with implementing pay-for-performance programs. The Centers for Medicare and Medicaid Services (CMS) needs to be flexible in adding—and removing—new HACs based on good evidence.
Regardless, the goal of achieving a safer, more effective healthcare system remains.
References
- Leape LL. Error in medicine. JAMA. 1994;272(23):1851-1857.
- Institute of Medicine. To Err Is Human: Building a Safer Healthcare System. Washington, D.C.: National Academies Press; 2000.
- Wachter RM, Pronovost PJ. Balancing “no blame” with accountability in patient safety. N Engl J Med. 2009;361:1401-1406.
- Saint S, Meddings JA, Calfee D, Kowalski CP, Krein SL. Catheter-associated urinary tract infection and the Medicare rule changes. Ann Intern Med. 2009;150(12):877-884.
- Inouye SK, Brown CJ, Tinetti ME. Medicare nonpayment, hospital falls, and unintended consequences. N Engl J Med. 2009;360(23):2390-2393.
- Wachter RM, Foster NE, Dudley RA. Medicare’s decision to withhold payment for hospital errors: the devil is in the det. Jt Comm J Qual Patient Saf. 2008;34(2):116-123.
- Geerts WH, Bergqvist D, Pineo GF, et al. Prevention of venous thromboembolism: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines (8th Edition). Chest. 2008;133(6 Suppl):381S-453S.
The opinions expressed herein are those of the authors and do not represent those of SHM or The Hospitalist.
Admit Documentation
In light of the recent elimination of consultation codes from the Medicare Physician Fee Schedule, physicians of all specialties are being asked to report initial hospital care services (99221-99223) for their first encounter with a patient.1 This leaves hospitalists with questions about the billing and financial implications of reporting admissions services.
Here’s a typical scenario: Dr. A admits a Medicare patient to the hospital from the ED for hyperglycemia and dehydration in the setting of uncontrolled diabetes. He performs and documents an initial hospital-care service on day one of the admission. On day two, another hospitalist, Dr. B, who works in the same HM group, sees the patient for the first time. What should each of the physicians report for their first encounter with the patient?
Each hospitalist should select the CPT code that best fits the service and their role in the case. Remember, only one physician is named “attending of record” or “admitting physician.”
When billing during the course of the hospitalization, consider all physicians of the same specialty in the same provider group as the “admitting physician/group.”
Admissions Service
On day one, Dr. A admits the patient. He performs and documents a comprehensive history, a comprehensive exam, and medical decision-making of high complexity. The documentation corresponds to the highest initial admission service, 99223. Given the recent Medicare billing changes, the attending of record is required to append modifier “AI” (principal physician of record) to the admission service (e.g., 99223-AI).
The purpose of this modifier is “to identify the physician who oversees the patient’s care from all other physicians who may be furnishing specialty care.”2 This modifier has no financial implications. It does not increase or decrease the payment associated with the reported visit level (i.e., 99223 is reimbursed at a national rate of approximately $190, with or without modifier AI).
Initial Encounter by Team Members
As previously stated, the elimination of consultation services requires physicians to report their initial hospital encounter with an initial hospital-care code (i.e., 99221-99223). However, Medicare states that “physicians in the same group practice who are in the same specialty must bill and be paid as though they were a single physician.”3 This means followup services performed on days subsequent to a group member’s initial admission service must be reported with subsequent hospital-care codes (99231-99233). Therefore, in the scenario above, Dr. B is obligated to report the appropriate subsequent hospital-care code for his patient encounter on day two.
Incomplete Documentation
Initial hospital-care services (99221-99223) require the physician to obtain, perform, and document the necessary elements of history, physical exam, and medical decision-making in support of the code reported on the claim. There are occasions when the physician’s documentation does not support the lowest code (i.e., 99221). A reasonable approach is to report the service with an unlisted E&M code (99499). “Unlisted” codes do not have a payor-recognized code description or fee. When reporting an unlisted code, the biller must manually enter a charge description (e.g., expanded problem-focused admissions service) and a fee. A payor-prompted request for documentation is likely before payment is made.
Some payors have more specific references to the situation and allow for options. Two options exist for coding services that do not meet the work and/or medical necessity requirements of 99221-99223: report an unlisted E&M service (99499); or report a subsequent hospital care code (99231-99233) that appropriately reflects physician work and medical necessity for the service, and avoids mandatory medical record submission and manual medical review.4
In fact, Medicare Administrator Contractor TrailBlazer Health’s Web site (www.trailblazerhealth.com) offers guidance to physicians who are unsure if subsequent hospital care is an appropriate choice for this dilemma: “TrailBlazer recognizes provider reluctance to miscode initial hospital care as subsequent hospital care. However, doing so is preferable in that it allows Medicare to process and pay the claims much more efficiently. For those concerned about miscoding these services, please understand that TrailBlazer will not find fault with providers who choose this option when records appropriately demonstrate the work and medical necessity of the subsequent code chosen.”4 TH
Carol Pohlig is a billing and coding expert with the University of Pennsylvania Medical Center, Philadelphia. She also is faculty for SHM’s inpatient coding course.
References
- CMS announces payment, policy changes for physicians services to Medicare beneficiaries in 2010. Centers for Medicare and Medicaid Services Web site. Available at: www.cms.hhs.gov/apps/media/ press/release.asp?Counter=3539&intNumPerPage=10&checkDate=&checkKey=&srchType=1&numDays=3500&srchOpt=0&srchData=&keywordType=All&chkNewsType=1%2C+2%2C+3%2C+4%2C+5&intPage=&showAll=&pYear=&year=&desc=&cboOrder=date. Accessed Nov. 12, 2009.
- Revisions to Consultation Services Payment Policy. Medicare Learning Network Web site. Available at: www.cms.hhs.gov/MLNMattersArticles/downloads/ MM6740.pdf. Accessed Jan. 16, 2010.
- Medicare Claims Processing Manual: Chapter 12, Section 30.6.5. CMS Web site. Available at: www.cms.hhs.gov/manuals/downloads/clm104c12.pdf. Accessed Jan. 16, 2010.
- Update-evaluation and management services formerly coded as consultations. Trailblazer Health Enterprises Web site. Available at: www.trailblazerhealth.com/Tools/Notices.aspx?DomainID=1. Accessed Jan. 17, 2010.
- Beebe M, Dalton J, Espronceda M, Evans D, Glenn R. Current Procedural Terminology Professional Edition. Chicago: American Medical Association Press; 2009;14-15.
In light of the recent elimination of consultation codes from the Medicare Physician Fee Schedule, physicians of all specialties are being asked to report initial hospital care services (99221-99223) for their first encounter with a patient.1 This leaves hospitalists with questions about the billing and financial implications of reporting admissions services.
Here’s a typical scenario: Dr. A admits a Medicare patient to the hospital from the ED for hyperglycemia and dehydration in the setting of uncontrolled diabetes. He performs and documents an initial hospital-care service on day one of the admission. On day two, another hospitalist, Dr. B, who works in the same HM group, sees the patient for the first time. What should each of the physicians report for their first encounter with the patient?
Each hospitalist should select the CPT code that best fits the service and their role in the case. Remember, only one physician is named “attending of record” or “admitting physician.”
When billing during the course of the hospitalization, consider all physicians of the same specialty in the same provider group as the “admitting physician/group.”
Admissions Service
On day one, Dr. A admits the patient. He performs and documents a comprehensive history, a comprehensive exam, and medical decision-making of high complexity. The documentation corresponds to the highest initial admission service, 99223. Given the recent Medicare billing changes, the attending of record is required to append modifier “AI” (principal physician of record) to the admission service (e.g., 99223-AI).
The purpose of this modifier is “to identify the physician who oversees the patient’s care from all other physicians who may be furnishing specialty care.”2 This modifier has no financial implications. It does not increase or decrease the payment associated with the reported visit level (i.e., 99223 is reimbursed at a national rate of approximately $190, with or without modifier AI).
Initial Encounter by Team Members
As previously stated, the elimination of consultation services requires physicians to report their initial hospital encounter with an initial hospital-care code (i.e., 99221-99223). However, Medicare states that “physicians in the same group practice who are in the same specialty must bill and be paid as though they were a single physician.”3 This means followup services performed on days subsequent to a group member’s initial admission service must be reported with subsequent hospital-care codes (99231-99233). Therefore, in the scenario above, Dr. B is obligated to report the appropriate subsequent hospital-care code for his patient encounter on day two.
Incomplete Documentation
Initial hospital-care services (99221-99223) require the physician to obtain, perform, and document the necessary elements of history, physical exam, and medical decision-making in support of the code reported on the claim. There are occasions when the physician’s documentation does not support the lowest code (i.e., 99221). A reasonable approach is to report the service with an unlisted E&M code (99499). “Unlisted” codes do not have a payor-recognized code description or fee. When reporting an unlisted code, the biller must manually enter a charge description (e.g., expanded problem-focused admissions service) and a fee. A payor-prompted request for documentation is likely before payment is made.
Some payors have more specific references to the situation and allow for options. Two options exist for coding services that do not meet the work and/or medical necessity requirements of 99221-99223: report an unlisted E&M service (99499); or report a subsequent hospital care code (99231-99233) that appropriately reflects physician work and medical necessity for the service, and avoids mandatory medical record submission and manual medical review.4
In fact, Medicare Administrator Contractor TrailBlazer Health’s Web site (www.trailblazerhealth.com) offers guidance to physicians who are unsure if subsequent hospital care is an appropriate choice for this dilemma: “TrailBlazer recognizes provider reluctance to miscode initial hospital care as subsequent hospital care. However, doing so is preferable in that it allows Medicare to process and pay the claims much more efficiently. For those concerned about miscoding these services, please understand that TrailBlazer will not find fault with providers who choose this option when records appropriately demonstrate the work and medical necessity of the subsequent code chosen.”4 TH
Carol Pohlig is a billing and coding expert with the University of Pennsylvania Medical Center, Philadelphia. She also is faculty for SHM’s inpatient coding course.
References
- CMS announces payment, policy changes for physicians services to Medicare beneficiaries in 2010. Centers for Medicare and Medicaid Services Web site. Available at: www.cms.hhs.gov/apps/media/ press/release.asp?Counter=3539&intNumPerPage=10&checkDate=&checkKey=&srchType=1&numDays=3500&srchOpt=0&srchData=&keywordType=All&chkNewsType=1%2C+2%2C+3%2C+4%2C+5&intPage=&showAll=&pYear=&year=&desc=&cboOrder=date. Accessed Nov. 12, 2009.
- Revisions to Consultation Services Payment Policy. Medicare Learning Network Web site. Available at: www.cms.hhs.gov/MLNMattersArticles/downloads/ MM6740.pdf. Accessed Jan. 16, 2010.
- Medicare Claims Processing Manual: Chapter 12, Section 30.6.5. CMS Web site. Available at: www.cms.hhs.gov/manuals/downloads/clm104c12.pdf. Accessed Jan. 16, 2010.
- Update-evaluation and management services formerly coded as consultations. Trailblazer Health Enterprises Web site. Available at: www.trailblazerhealth.com/Tools/Notices.aspx?DomainID=1. Accessed Jan. 17, 2010.
- Beebe M, Dalton J, Espronceda M, Evans D, Glenn R. Current Procedural Terminology Professional Edition. Chicago: American Medical Association Press; 2009;14-15.
In light of the recent elimination of consultation codes from the Medicare Physician Fee Schedule, physicians of all specialties are being asked to report initial hospital care services (99221-99223) for their first encounter with a patient.1 This leaves hospitalists with questions about the billing and financial implications of reporting admissions services.
Here’s a typical scenario: Dr. A admits a Medicare patient to the hospital from the ED for hyperglycemia and dehydration in the setting of uncontrolled diabetes. He performs and documents an initial hospital-care service on day one of the admission. On day two, another hospitalist, Dr. B, who works in the same HM group, sees the patient for the first time. What should each of the physicians report for their first encounter with the patient?
Each hospitalist should select the CPT code that best fits the service and their role in the case. Remember, only one physician is named “attending of record” or “admitting physician.”
When billing during the course of the hospitalization, consider all physicians of the same specialty in the same provider group as the “admitting physician/group.”
Admissions Service
On day one, Dr. A admits the patient. He performs and documents a comprehensive history, a comprehensive exam, and medical decision-making of high complexity. The documentation corresponds to the highest initial admission service, 99223. Given the recent Medicare billing changes, the attending of record is required to append modifier “AI” (principal physician of record) to the admission service (e.g., 99223-AI).
The purpose of this modifier is “to identify the physician who oversees the patient’s care from all other physicians who may be furnishing specialty care.”2 This modifier has no financial implications. It does not increase or decrease the payment associated with the reported visit level (i.e., 99223 is reimbursed at a national rate of approximately $190, with or without modifier AI).
Initial Encounter by Team Members
As previously stated, the elimination of consultation services requires physicians to report their initial hospital encounter with an initial hospital-care code (i.e., 99221-99223). However, Medicare states that “physicians in the same group practice who are in the same specialty must bill and be paid as though they were a single physician.”3 This means followup services performed on days subsequent to a group member’s initial admission service must be reported with subsequent hospital-care codes (99231-99233). Therefore, in the scenario above, Dr. B is obligated to report the appropriate subsequent hospital-care code for his patient encounter on day two.
Incomplete Documentation
Initial hospital-care services (99221-99223) require the physician to obtain, perform, and document the necessary elements of history, physical exam, and medical decision-making in support of the code reported on the claim. There are occasions when the physician’s documentation does not support the lowest code (i.e., 99221). A reasonable approach is to report the service with an unlisted E&M code (99499). “Unlisted” codes do not have a payor-recognized code description or fee. When reporting an unlisted code, the biller must manually enter a charge description (e.g., expanded problem-focused admissions service) and a fee. A payor-prompted request for documentation is likely before payment is made.
Some payors have more specific references to the situation and allow for options. Two options exist for coding services that do not meet the work and/or medical necessity requirements of 99221-99223: report an unlisted E&M service (99499); or report a subsequent hospital care code (99231-99233) that appropriately reflects physician work and medical necessity for the service, and avoids mandatory medical record submission and manual medical review.4
In fact, Medicare Administrator Contractor TrailBlazer Health’s Web site (www.trailblazerhealth.com) offers guidance to physicians who are unsure if subsequent hospital care is an appropriate choice for this dilemma: “TrailBlazer recognizes provider reluctance to miscode initial hospital care as subsequent hospital care. However, doing so is preferable in that it allows Medicare to process and pay the claims much more efficiently. For those concerned about miscoding these services, please understand that TrailBlazer will not find fault with providers who choose this option when records appropriately demonstrate the work and medical necessity of the subsequent code chosen.”4 TH
Carol Pohlig is a billing and coding expert with the University of Pennsylvania Medical Center, Philadelphia. She also is faculty for SHM’s inpatient coding course.
References
- CMS announces payment, policy changes for physicians services to Medicare beneficiaries in 2010. Centers for Medicare and Medicaid Services Web site. Available at: www.cms.hhs.gov/apps/media/ press/release.asp?Counter=3539&intNumPerPage=10&checkDate=&checkKey=&srchType=1&numDays=3500&srchOpt=0&srchData=&keywordType=All&chkNewsType=1%2C+2%2C+3%2C+4%2C+5&intPage=&showAll=&pYear=&year=&desc=&cboOrder=date. Accessed Nov. 12, 2009.
- Revisions to Consultation Services Payment Policy. Medicare Learning Network Web site. Available at: www.cms.hhs.gov/MLNMattersArticles/downloads/ MM6740.pdf. Accessed Jan. 16, 2010.
- Medicare Claims Processing Manual: Chapter 12, Section 30.6.5. CMS Web site. Available at: www.cms.hhs.gov/manuals/downloads/clm104c12.pdf. Accessed Jan. 16, 2010.
- Update-evaluation and management services formerly coded as consultations. Trailblazer Health Enterprises Web site. Available at: www.trailblazerhealth.com/Tools/Notices.aspx?DomainID=1. Accessed Jan. 17, 2010.
- Beebe M, Dalton J, Espronceda M, Evans D, Glenn R. Current Procedural Terminology Professional Edition. Chicago: American Medical Association Press; 2009;14-15.
In the Literature: March 2010
In This Edition
Literature at a Glance
A guide to this month’s studies
- Statins and postoperative cardiac outcomes
- Cardiac resynchronization therapy in patients with mild CHF symptoms
- Oral direct thrombin inhibitor versus warfarin for stroke prevention in atrial fibrillation
- Association of fatigue and medical error
- Effects of chronic inhaled steroid and beta-agonist use in COPD
- Dialysis and functional status in nursing home patients
- Outcomes with different insulin-dosing regimens
- Understanding of disease severity and outcomes in advanced dementia
Fluvastatin Improves Postoperative Cardiac Outcomes in Patients Undergoing Vascular Surgery
Clinical question: Does perioperative fluvastatin decrease adverse cardiac events after vascular surgery?
Background: Patients with atherosclerotic vascular disease who undergo vascular surgery are at high risk for postoperative cardiac events. Studies in nonsurgical populations have shown the beneficial effects of statin therapy on cardiac outcomes. However, no placebo-controlled trials have addressed the effect of statins on postoperative cardiac outcomes.
Study design: Randomized, double-blind, placebo-controlled trial.
Setting: Single large academic medical center in the Netherlands.
Synopsis: The study looked at 497 statin-naïve patients 40 years or older undergoing non-cardiac vascular surgery. The patients were randomized to 80 mg of extended-release fluvastatin versus placebo; all patients received a beta-blocker. Therapy began preoperatively (median of 37 days) and continued for at least 30 days after surgery. Outcomes were assessed at 30 days post-surgery.
Postoperative myocardial infarction (MI) was significantly less common in the fluvastatin group than with placebo (10.8% vs. 19%, hazard ratio (HR) 0.55, P=0.01). In addition, the treatment group had a lower frequency of death from cardiovascular causes (4.8% vs. 10.1%, HR 0.47, P=0.03). Statin therapy was not associated with an increased rate of adverse events.
Notably, all of the patients enrolled in this study were high-risk patients undergoing high-risk (vascular) surgery. Patients already on statins were excluded.
Further studies are needed to determine whether the findings can be extrapolated to other populations, including nonvascular surgery patients.
Bottom line: Perioperative statin therapy resulted in a significant decrease in postoperative MI and death within 30 days of vascular surgery.
Citation: Schouten O, Boersma E, Hoeks SE, et al. Fluvastatin and perioperative events in patients undergoing vascular surgery. N Engl J Med. 2009;361(10):980-989.
Cardiac Resynchronization Therapy with Implantable Cardioverter Defibrillator Placement Decreases Heart Failure
Clinical question: Does cardiac resynchronization therapy (CRT) with biventricular pacing decrease cardiac events in patients with reduced ejection fraction (EF) and wide QRS complex but only mild cardiac symptoms?
Background: In patients with severely reduced EF, implantable cardioverter defibrillators (ICDs) have been shown to improve survival. Meanwhile, CRT decreases heart-failure-related hospitalizations for patients with advanced heart-failure symptoms, EF less than 35%, and intraventricular conduction delay. It is not as clear whether patients with less-severe symptoms benefit from CRT.
Study design: Randomized, controlled trial.
Setting: 110 medical centers in the U.S., Canada, and Europe.
Synopsis: This Multicenter Automatic Defibrillator Implantation Trial with Cardiac Resynchronization Therapy (MADIT-CRT) study randomly assigned 1,820 adults with EF less than 30%, New York Health Association Class I or II congestive heart failure, and in sinus rhythm with QRS greater than 130 msec to receive ICD with CRT or ICD alone. The primary endpoint was all-cause mortality or nonfatal heart-failure events. Average followup was 2.4 years.
A 34% reduction in the primary endpoint was found in the ICD-CRT group when compared with the ICD-only group, primarily due to a 41% reduction in heart-failure events. In a subgroup analysis, women and patients with QRS greater than 150 msec experienced particular benefit. Echocardiography one year after device implantation demonstrated significant reductions in left ventricular end-systolic and end-diastolic volume, and a significant increase in EF with ICD-CRT versus ICD-only (P<0.001).
Bottom line: Compared with ICD alone, CRT in combination with ICD prevented heart-failure events in relatively asymptomatic heart-failure patients with low EF and prolonged QRS.
Citation: Moss AJ, Hall WJ, Cannom DS, et al. Cardiac-resynchronization therapy for the prevention of heart-failure events. N Engl J Med. 2009;361(14):1329-1338.
Dabigatran Is Not Inferior to Warfarin in Atrial Fibrillation
Clinical question: Is dabigatran, an oral thrombin inhibitor, an effective and safe alternative to warfarin in patients with atrial fibrillation?
Background: Warfarin reduces the risk of stroke among patients with atrial fibrillation (AF) but requires frequent laboratory monitoring. Dabigatran is an oral direct thrombin inhibitor given in fixed dosages without laboratory monitoring.
Study design: Randomized, multicenter, open-label, noninferiority trial.
Setting: 951 clinical centers in 44 countries.
Synopsis: More than 18,000 patients 65 and older with AF and at least one stroke risk factor were enrolled. The average CHADS2 score was 2.1. Patients were randomized to receive fixed doses of dabigatran (110 mg or 150 mg, twice daily) or warfarin adjusted to an INR of 2.0-3.0. The primary outcomes were a) stroke or systemic embolism and b) major hemorrhage. Median followup was two years.
The annual rates of stroke or systemic embolism for both doses of dabigatran were noninferior to warfarin (P<0.001); higher-dose dabigatran was statistically superior to warfarin (relative risk (RR)=0.66, P<0.001). The annual rate of major hemorrhage was lowest in the lower-dose dabigatran group (RR=0.80, P=0.003 compared with warfarin); the higher-dose dabigatran and warfarin groups had equivalent rates of major bleeding. No increased risk of liver function abnormalities was noted.
Bottom line: Dabigatran appears to be an effective and safe alternative to warfarin in AF patients. If the drug were to be FDA-approved, appropriate patient selection and cost will need to be established.
Citation: Connolly SJ, Ezekowitz MD, Yusuf S, et al. Dabigatran versus warfarin in patients with atrial fibrillation. N Engl J Med. 2009;361(12):1139-1151.
Resident Fatigue and Distress Contribute to Perceived Medical Errors
Clinical question: Do resident fatigue and distress contribute to medical errors?
Background: In recent years, such measures as work-hour limitations have been implemented to decrease resident fatigue and, it is presumed, medical errors. However, few studies address the relationship between residents’ well-being and self-reported medical errors.
Study design: Prospective six-year longitudinal cohort study.
Setting: Single academic medical center.
Synopsis: The authors had 380 internal-medicine residents complete quarterly surveys to assess fatigue, quality of life, burnout, symptoms of depression, and frequency of perceived medical errors. In a univariate analysis, fatigue/sleepiness, burnout, depression, and overall quality of life measures correlated significantly with self-reported major medical errors. Fatigue/sleepiness and measures of distress additively increased the risk of self-reported errors. Increases in one or both domains were estimated to increase the risk of self-reported errors by as much as 15% to 28%.
The authors studied only self-reported medical errors. It is difficult to know whether these errors directly affected patient outcomes. Additionally, results of this single-site study might not be able to be generalized.
Bottom line: Fatigue and distress contribute to self-perceived medical errors among residents.
Citation: West CP, Tan AD, Habermann TM, Sloan JA, Shanafelt TD. Association of resident fatigue and distress with perceived medical errors. JAMA. 2009;302(12):1294-1300.
Inhaled Corticosteroids Decrease Inflammation in Moderate to Severe COPD
Clinical question: Does long-term inhaled corticosteroid therapy, with and without long-acting beta-agonists, decrease airway inflammation and improve lung function in patients with moderate to severe chronic obstructive pulmonary disease (COPD)?
Background: Guideline-recommended treatment of COPD with inhaled corticosteroids and long-acting beta-agonists improves symptoms and exacerbation rates; little is known about the impact of these therapies on inflammation and long-term lung function.
Study design: Randomized, double-blind, placebo-controlled trial.
Setting: Two university medical centers in the Netherlands.
Synopsis: One hundred one steroid-naïve patients, ages 45 to 75 who were current or former smokers with moderate to severe COPD, were randomized to one of four regimens: 1) fluticasone for six months, then placebo for 24 months; 2) fluticasone for 30 months; 3) fluticasone and salmeterol for 30 months; or 4) placebo for 30 months. The primary outcome was inflammatory cell counts in bronchial biopsies/induced sputum. Secondary outcomes included postbronchodilator spirometry, methacholine hyperresponsiveness, and self-reported symptoms and health status. Patients with asthma were excluded.
Short-term fluticasone therapy decreased inflammation and improved forced expiratory volume in one second (FEV1). Long-term therapy also decreased the rate of FEV1 decline, reduced dyspnea, and improved health status. Discontinuation of therapy at six months led to inflammation relapse with worsened symptoms and increased rate of FEV1 decline. The addition of long-acting beta-agonists did not provide additional anti-inflammatory benefits, but it did improve FEV1 and dyspnea at six months.
Additional studies are needed to further define clinical outcomes and assess the cost benefit of these therapies.
Bottom line: Inhaled corticosteroids decrease inflammation in steroid-naïve patients with moderate to severe COPD and might decrease the rate of lung function decline. Long-acting beta-agonists do not offer additional anti-inflammatory benefit.
Citation: Lapperre TS, Snoeck-Stroband JB, Gosman MM, et al. Effect of fluticasone with and without salmeterol on pulmonary outcomes in chronic obstructive pulmonary disease: a randomized trial. Ann Intern Med. 2009;151(8):517-527.
Initiation of Dialysis Does Not Help Maintain Functional Status in Elderly
Clinical question: Is functional status in the elderly maintained over time after initiating long-term dialysis?
Background: Quality-of-life maintenance often is used as a goal when initiating long-term dialysis in elderly patients with end-stage renal disease. More elderly patients are being offered long-term dialysis treatment. Little is known about the functional status of elderly patients on long-term dialysis.
Study design: Retrospective cohort study.
Setting: U.S. nursing homes.
Synopsis: By cross-linking data from two population-based administrative datasets, this study identified 3,702 nursing home patients (mean 73.4 years) who had started long-term dialysis and whose functional status had been assessed. Activities of daily living assessments before and at three-month intervals after dialysis initiation were compared to see if functional status was maintained.
Within three months of starting dialysis, 61% of patients had a decline in functional status or had died. By one year, only 1 in 8 patients had maintained their pre-dialysis functional status.
Decline in functional status cannot be attributed solely to dialysis because study patients were not compared to patients with chronic kidney disease who were not dialyzed. In addition, these results might not apply to all elderly patients on dialysis, as the functional status of elderly nursing home patients might differ significantly from those living at home.
Bottom line: Functional status is not maintained in most elderly nursing home patients in the first 12 months after long-term dialysis is initiated. Elderly patients considering dialysis treatment should be aware that dialysis might not help maintain functional status and quality of life.
Citation: Kurella Tamura MK, Covinsky KE, Chertow GM, Yaffe C, Landefeld CS, McCulloch CE. Functional status of elderly adults before and after initiation of dialysis. N Engl J Med. 2009;361(16):1539-1547.
Adding Basal Insulin to Oral Agents in Type 2 Diabetes Might Offer Best Glycemic Control
Clinical question: When added to oral diabetic agents, which insulin regimen (biphasic, prandial or basal) best achieves glycemic control in patients with Type 2 diabetes?
Background: Most patients with Type 2 diabetes mellitus (DM2) require insulin when oral agents provide suboptimal glycemic control. Little is known about which insulin regimen is most effective.
Study design: Three-year, open-label, multicenter trial.
Setting: Fifty-eight clinical centers in the United Kingdom and Ireland.
Synopsis: The authors randomized 708 insulin-naïve DM2 patients (median age 62 years) with HgbA1c 7% to 10% on maximum-dose metformin or sulfonylurea to one of three regimens: biphasic insulin twice daily; prandial insulin three times daily; or basal insulin once daily. Outcomes were HgbA1c, hypoglycemia rates, and weight gain. Sulfonylureas were replaced by another insulin if glycemic control was unacceptable.
The patients were mostly Caucasian and overweight. At three years of followup, median HgbA1c was similar in all groups (7.1% biphasic, 6.8% prandial, 6.9% basal); however, more patients who received prandial or basal insulin achieved HgbA1c less than 6.5% (45% and 43%, respectively) than in the biphasic group (32%).
Hypoglycemia was significantly less frequent in the basal insulin group (1.7 per patient per year versus 3.0 and 5.5 with biphasic and prandial, respectively). Patients gained weight in all groups; the greatest gain was with prandial insulin. At three years, there were no significant between-group differences in blood pressure, cholesterol, albuminuria, or quality of life.
Bottom line: Adding insulin to oral diabetic regimens improves glycemic control. Basal or prandial insulin regimens achieve glycemic targets more frequently than biphasic dosing.
Citation: Holman RR, Farmer AJ, Davies MJ, et al. Three-year efficacy of complex insulin regimens in type 2 diabetes. N Engl J Med. 2009;361(18):1736-1747.
Advanced Dementia Is a Terminal Illness with High Morbidity and Mortality
Clinical question: Does understanding the expected clinical course of advanced dementia influence end-of-life decisions by proxy decision-makers?
Background: Advanced dementia is a leading cause of death in the United States, but the clinical course of advanced dementia has not been described in a rigorous, prospective manner. The lack of information might cause risk to be underestimated, and patients might receive suboptimal palliative care.
Study design: Multicenter prospective cohort study.
Setting: Twenty-two nursing homes in a single U.S. city.
Synopsis: The survey examined 323 nursing home residents with advanced dementia. The patients were clinically assessed at baseline and quarterly for 18 months through chart reviews, nursing interviews, and physical examinations. Additionally, their proxies were surveyed regarding their understanding of the subjects’ prognoses.
During the survey period, 41.1% of patients developed pneumonia, 52.6% of patients experienced a febrile episode, and 85.8% of patients developed an eating problem; cumulative all-cause mortality was 54.8%. Adjusted for age, sex, and disease duration, the six-month mortality rate for subjects who had pneumonia was 46.7%; a febrile episode, 44.5%; and an eating problem, 38.6%.
Distressing symptoms, including dyspnea (46.0%) and pain (39.1%), were common. In the last three months of life, 40.7% of subjects underwent at least one burdensome intervention (defined as hospitalization, ED visit, parenteral therapy, or tube feeding).
Subjects whose proxies reported an understanding of the poor prognosis and expected clinical complications of advanced dementia underwent significantly fewer burdensome interventions (adjusted odds ratio 0.12).
Bottom line: Advanced dementia is associated with frequent complications, including infections and eating problems, with high six-month mortality and significant associated morbidity. Patients whose healthcare proxies have a good understanding of the expected clinical course and prognosis receive less-aggressive end-of-life care.
Citation: Mitchell SL, Teno JM, Kiely DK, et al. The clinical course of advanced dementia. N Engl J Med. 2009;361(16):1529-1538. TH
In This Edition
Literature at a Glance
A guide to this month’s studies
- Statins and postoperative cardiac outcomes
- Cardiac resynchronization therapy in patients with mild CHF symptoms
- Oral direct thrombin inhibitor versus warfarin for stroke prevention in atrial fibrillation
- Association of fatigue and medical error
- Effects of chronic inhaled steroid and beta-agonist use in COPD
- Dialysis and functional status in nursing home patients
- Outcomes with different insulin-dosing regimens
- Understanding of disease severity and outcomes in advanced dementia
Fluvastatin Improves Postoperative Cardiac Outcomes in Patients Undergoing Vascular Surgery
Clinical question: Does perioperative fluvastatin decrease adverse cardiac events after vascular surgery?
Background: Patients with atherosclerotic vascular disease who undergo vascular surgery are at high risk for postoperative cardiac events. Studies in nonsurgical populations have shown the beneficial effects of statin therapy on cardiac outcomes. However, no placebo-controlled trials have addressed the effect of statins on postoperative cardiac outcomes.
Study design: Randomized, double-blind, placebo-controlled trial.
Setting: Single large academic medical center in the Netherlands.
Synopsis: The study looked at 497 statin-naïve patients 40 years or older undergoing non-cardiac vascular surgery. The patients were randomized to 80 mg of extended-release fluvastatin versus placebo; all patients received a beta-blocker. Therapy began preoperatively (median of 37 days) and continued for at least 30 days after surgery. Outcomes were assessed at 30 days post-surgery.
Postoperative myocardial infarction (MI) was significantly less common in the fluvastatin group than with placebo (10.8% vs. 19%, hazard ratio (HR) 0.55, P=0.01). In addition, the treatment group had a lower frequency of death from cardiovascular causes (4.8% vs. 10.1%, HR 0.47, P=0.03). Statin therapy was not associated with an increased rate of adverse events.
Notably, all of the patients enrolled in this study were high-risk patients undergoing high-risk (vascular) surgery. Patients already on statins were excluded.
Further studies are needed to determine whether the findings can be extrapolated to other populations, including nonvascular surgery patients.
Bottom line: Perioperative statin therapy resulted in a significant decrease in postoperative MI and death within 30 days of vascular surgery.
Citation: Schouten O, Boersma E, Hoeks SE, et al. Fluvastatin and perioperative events in patients undergoing vascular surgery. N Engl J Med. 2009;361(10):980-989.
Cardiac Resynchronization Therapy with Implantable Cardioverter Defibrillator Placement Decreases Heart Failure
Clinical question: Does cardiac resynchronization therapy (CRT) with biventricular pacing decrease cardiac events in patients with reduced ejection fraction (EF) and wide QRS complex but only mild cardiac symptoms?
Background: In patients with severely reduced EF, implantable cardioverter defibrillators (ICDs) have been shown to improve survival. Meanwhile, CRT decreases heart-failure-related hospitalizations for patients with advanced heart-failure symptoms, EF less than 35%, and intraventricular conduction delay. It is not as clear whether patients with less-severe symptoms benefit from CRT.
Study design: Randomized, controlled trial.
Setting: 110 medical centers in the U.S., Canada, and Europe.
Synopsis: This Multicenter Automatic Defibrillator Implantation Trial with Cardiac Resynchronization Therapy (MADIT-CRT) study randomly assigned 1,820 adults with EF less than 30%, New York Health Association Class I or II congestive heart failure, and in sinus rhythm with QRS greater than 130 msec to receive ICD with CRT or ICD alone. The primary endpoint was all-cause mortality or nonfatal heart-failure events. Average followup was 2.4 years.
A 34% reduction in the primary endpoint was found in the ICD-CRT group when compared with the ICD-only group, primarily due to a 41% reduction in heart-failure events. In a subgroup analysis, women and patients with QRS greater than 150 msec experienced particular benefit. Echocardiography one year after device implantation demonstrated significant reductions in left ventricular end-systolic and end-diastolic volume, and a significant increase in EF with ICD-CRT versus ICD-only (P<0.001).
Bottom line: Compared with ICD alone, CRT in combination with ICD prevented heart-failure events in relatively asymptomatic heart-failure patients with low EF and prolonged QRS.
Citation: Moss AJ, Hall WJ, Cannom DS, et al. Cardiac-resynchronization therapy for the prevention of heart-failure events. N Engl J Med. 2009;361(14):1329-1338.
Dabigatran Is Not Inferior to Warfarin in Atrial Fibrillation
Clinical question: Is dabigatran, an oral thrombin inhibitor, an effective and safe alternative to warfarin in patients with atrial fibrillation?
Background: Warfarin reduces the risk of stroke among patients with atrial fibrillation (AF) but requires frequent laboratory monitoring. Dabigatran is an oral direct thrombin inhibitor given in fixed dosages without laboratory monitoring.
Study design: Randomized, multicenter, open-label, noninferiority trial.
Setting: 951 clinical centers in 44 countries.
Synopsis: More than 18,000 patients 65 and older with AF and at least one stroke risk factor were enrolled. The average CHADS2 score was 2.1. Patients were randomized to receive fixed doses of dabigatran (110 mg or 150 mg, twice daily) or warfarin adjusted to an INR of 2.0-3.0. The primary outcomes were a) stroke or systemic embolism and b) major hemorrhage. Median followup was two years.
The annual rates of stroke or systemic embolism for both doses of dabigatran were noninferior to warfarin (P<0.001); higher-dose dabigatran was statistically superior to warfarin (relative risk (RR)=0.66, P<0.001). The annual rate of major hemorrhage was lowest in the lower-dose dabigatran group (RR=0.80, P=0.003 compared with warfarin); the higher-dose dabigatran and warfarin groups had equivalent rates of major bleeding. No increased risk of liver function abnormalities was noted.
Bottom line: Dabigatran appears to be an effective and safe alternative to warfarin in AF patients. If the drug were to be FDA-approved, appropriate patient selection and cost will need to be established.
Citation: Connolly SJ, Ezekowitz MD, Yusuf S, et al. Dabigatran versus warfarin in patients with atrial fibrillation. N Engl J Med. 2009;361(12):1139-1151.
Resident Fatigue and Distress Contribute to Perceived Medical Errors
Clinical question: Do resident fatigue and distress contribute to medical errors?
Background: In recent years, such measures as work-hour limitations have been implemented to decrease resident fatigue and, it is presumed, medical errors. However, few studies address the relationship between residents’ well-being and self-reported medical errors.
Study design: Prospective six-year longitudinal cohort study.
Setting: Single academic medical center.
Synopsis: The authors had 380 internal-medicine residents complete quarterly surveys to assess fatigue, quality of life, burnout, symptoms of depression, and frequency of perceived medical errors. In a univariate analysis, fatigue/sleepiness, burnout, depression, and overall quality of life measures correlated significantly with self-reported major medical errors. Fatigue/sleepiness and measures of distress additively increased the risk of self-reported errors. Increases in one or both domains were estimated to increase the risk of self-reported errors by as much as 15% to 28%.
The authors studied only self-reported medical errors. It is difficult to know whether these errors directly affected patient outcomes. Additionally, results of this single-site study might not be able to be generalized.
Bottom line: Fatigue and distress contribute to self-perceived medical errors among residents.
Citation: West CP, Tan AD, Habermann TM, Sloan JA, Shanafelt TD. Association of resident fatigue and distress with perceived medical errors. JAMA. 2009;302(12):1294-1300.
Inhaled Corticosteroids Decrease Inflammation in Moderate to Severe COPD
Clinical question: Does long-term inhaled corticosteroid therapy, with and without long-acting beta-agonists, decrease airway inflammation and improve lung function in patients with moderate to severe chronic obstructive pulmonary disease (COPD)?
Background: Guideline-recommended treatment of COPD with inhaled corticosteroids and long-acting beta-agonists improves symptoms and exacerbation rates; little is known about the impact of these therapies on inflammation and long-term lung function.
Study design: Randomized, double-blind, placebo-controlled trial.
Setting: Two university medical centers in the Netherlands.
Synopsis: One hundred one steroid-naïve patients, ages 45 to 75 who were current or former smokers with moderate to severe COPD, were randomized to one of four regimens: 1) fluticasone for six months, then placebo for 24 months; 2) fluticasone for 30 months; 3) fluticasone and salmeterol for 30 months; or 4) placebo for 30 months. The primary outcome was inflammatory cell counts in bronchial biopsies/induced sputum. Secondary outcomes included postbronchodilator spirometry, methacholine hyperresponsiveness, and self-reported symptoms and health status. Patients with asthma were excluded.
Short-term fluticasone therapy decreased inflammation and improved forced expiratory volume in one second (FEV1). Long-term therapy also decreased the rate of FEV1 decline, reduced dyspnea, and improved health status. Discontinuation of therapy at six months led to inflammation relapse with worsened symptoms and increased rate of FEV1 decline. The addition of long-acting beta-agonists did not provide additional anti-inflammatory benefits, but it did improve FEV1 and dyspnea at six months.
Additional studies are needed to further define clinical outcomes and assess the cost benefit of these therapies.
Bottom line: Inhaled corticosteroids decrease inflammation in steroid-naïve patients with moderate to severe COPD and might decrease the rate of lung function decline. Long-acting beta-agonists do not offer additional anti-inflammatory benefit.
Citation: Lapperre TS, Snoeck-Stroband JB, Gosman MM, et al. Effect of fluticasone with and without salmeterol on pulmonary outcomes in chronic obstructive pulmonary disease: a randomized trial. Ann Intern Med. 2009;151(8):517-527.
Initiation of Dialysis Does Not Help Maintain Functional Status in Elderly
Clinical question: Is functional status in the elderly maintained over time after initiating long-term dialysis?
Background: Quality-of-life maintenance often is used as a goal when initiating long-term dialysis in elderly patients with end-stage renal disease. More elderly patients are being offered long-term dialysis treatment. Little is known about the functional status of elderly patients on long-term dialysis.
Study design: Retrospective cohort study.
Setting: U.S. nursing homes.
Synopsis: By cross-linking data from two population-based administrative datasets, this study identified 3,702 nursing home patients (mean 73.4 years) who had started long-term dialysis and whose functional status had been assessed. Activities of daily living assessments before and at three-month intervals after dialysis initiation were compared to see if functional status was maintained.
Within three months of starting dialysis, 61% of patients had a decline in functional status or had died. By one year, only 1 in 8 patients had maintained their pre-dialysis functional status.
Decline in functional status cannot be attributed solely to dialysis because study patients were not compared to patients with chronic kidney disease who were not dialyzed. In addition, these results might not apply to all elderly patients on dialysis, as the functional status of elderly nursing home patients might differ significantly from those living at home.
Bottom line: Functional status is not maintained in most elderly nursing home patients in the first 12 months after long-term dialysis is initiated. Elderly patients considering dialysis treatment should be aware that dialysis might not help maintain functional status and quality of life.
Citation: Kurella Tamura MK, Covinsky KE, Chertow GM, Yaffe C, Landefeld CS, McCulloch CE. Functional status of elderly adults before and after initiation of dialysis. N Engl J Med. 2009;361(16):1539-1547.
Adding Basal Insulin to Oral Agents in Type 2 Diabetes Might Offer Best Glycemic Control
Clinical question: When added to oral diabetic agents, which insulin regimen (biphasic, prandial or basal) best achieves glycemic control in patients with Type 2 diabetes?
Background: Most patients with Type 2 diabetes mellitus (DM2) require insulin when oral agents provide suboptimal glycemic control. Little is known about which insulin regimen is most effective.
Study design: Three-year, open-label, multicenter trial.
Setting: Fifty-eight clinical centers in the United Kingdom and Ireland.
Synopsis: The authors randomized 708 insulin-naïve DM2 patients (median age 62 years) with HgbA1c 7% to 10% on maximum-dose metformin or sulfonylurea to one of three regimens: biphasic insulin twice daily; prandial insulin three times daily; or basal insulin once daily. Outcomes were HgbA1c, hypoglycemia rates, and weight gain. Sulfonylureas were replaced by another insulin if glycemic control was unacceptable.
The patients were mostly Caucasian and overweight. At three years of followup, median HgbA1c was similar in all groups (7.1% biphasic, 6.8% prandial, 6.9% basal); however, more patients who received prandial or basal insulin achieved HgbA1c less than 6.5% (45% and 43%, respectively) than in the biphasic group (32%).
Hypoglycemia was significantly less frequent in the basal insulin group (1.7 per patient per year versus 3.0 and 5.5 with biphasic and prandial, respectively). Patients gained weight in all groups; the greatest gain was with prandial insulin. At three years, there were no significant between-group differences in blood pressure, cholesterol, albuminuria, or quality of life.
Bottom line: Adding insulin to oral diabetic regimens improves glycemic control. Basal or prandial insulin regimens achieve glycemic targets more frequently than biphasic dosing.
Citation: Holman RR, Farmer AJ, Davies MJ, et al. Three-year efficacy of complex insulin regimens in type 2 diabetes. N Engl J Med. 2009;361(18):1736-1747.
Advanced Dementia Is a Terminal Illness with High Morbidity and Mortality
Clinical question: Does understanding the expected clinical course of advanced dementia influence end-of-life decisions by proxy decision-makers?
Background: Advanced dementia is a leading cause of death in the United States, but the clinical course of advanced dementia has not been described in a rigorous, prospective manner. The lack of information might cause risk to be underestimated, and patients might receive suboptimal palliative care.
Study design: Multicenter prospective cohort study.
Setting: Twenty-two nursing homes in a single U.S. city.
Synopsis: The survey examined 323 nursing home residents with advanced dementia. The patients were clinically assessed at baseline and quarterly for 18 months through chart reviews, nursing interviews, and physical examinations. Additionally, their proxies were surveyed regarding their understanding of the subjects’ prognoses.
During the survey period, 41.1% of patients developed pneumonia, 52.6% of patients experienced a febrile episode, and 85.8% of patients developed an eating problem; cumulative all-cause mortality was 54.8%. Adjusted for age, sex, and disease duration, the six-month mortality rate for subjects who had pneumonia was 46.7%; a febrile episode, 44.5%; and an eating problem, 38.6%.
Distressing symptoms, including dyspnea (46.0%) and pain (39.1%), were common. In the last three months of life, 40.7% of subjects underwent at least one burdensome intervention (defined as hospitalization, ED visit, parenteral therapy, or tube feeding).
Subjects whose proxies reported an understanding of the poor prognosis and expected clinical complications of advanced dementia underwent significantly fewer burdensome interventions (adjusted odds ratio 0.12).
Bottom line: Advanced dementia is associated with frequent complications, including infections and eating problems, with high six-month mortality and significant associated morbidity. Patients whose healthcare proxies have a good understanding of the expected clinical course and prognosis receive less-aggressive end-of-life care.
Citation: Mitchell SL, Teno JM, Kiely DK, et al. The clinical course of advanced dementia. N Engl J Med. 2009;361(16):1529-1538. TH
In This Edition
Literature at a Glance
A guide to this month’s studies
- Statins and postoperative cardiac outcomes
- Cardiac resynchronization therapy in patients with mild CHF symptoms
- Oral direct thrombin inhibitor versus warfarin for stroke prevention in atrial fibrillation
- Association of fatigue and medical error
- Effects of chronic inhaled steroid and beta-agonist use in COPD
- Dialysis and functional status in nursing home patients
- Outcomes with different insulin-dosing regimens
- Understanding of disease severity and outcomes in advanced dementia
Fluvastatin Improves Postoperative Cardiac Outcomes in Patients Undergoing Vascular Surgery
Clinical question: Does perioperative fluvastatin decrease adverse cardiac events after vascular surgery?
Background: Patients with atherosclerotic vascular disease who undergo vascular surgery are at high risk for postoperative cardiac events. Studies in nonsurgical populations have shown the beneficial effects of statin therapy on cardiac outcomes. However, no placebo-controlled trials have addressed the effect of statins on postoperative cardiac outcomes.
Study design: Randomized, double-blind, placebo-controlled trial.
Setting: Single large academic medical center in the Netherlands.
Synopsis: The study looked at 497 statin-naïve patients 40 years or older undergoing non-cardiac vascular surgery. The patients were randomized to 80 mg of extended-release fluvastatin versus placebo; all patients received a beta-blocker. Therapy began preoperatively (median of 37 days) and continued for at least 30 days after surgery. Outcomes were assessed at 30 days post-surgery.
Postoperative myocardial infarction (MI) was significantly less common in the fluvastatin group than with placebo (10.8% vs. 19%, hazard ratio (HR) 0.55, P=0.01). In addition, the treatment group had a lower frequency of death from cardiovascular causes (4.8% vs. 10.1%, HR 0.47, P=0.03). Statin therapy was not associated with an increased rate of adverse events.
Notably, all of the patients enrolled in this study were high-risk patients undergoing high-risk (vascular) surgery. Patients already on statins were excluded.
Further studies are needed to determine whether the findings can be extrapolated to other populations, including nonvascular surgery patients.
Bottom line: Perioperative statin therapy resulted in a significant decrease in postoperative MI and death within 30 days of vascular surgery.
Citation: Schouten O, Boersma E, Hoeks SE, et al. Fluvastatin and perioperative events in patients undergoing vascular surgery. N Engl J Med. 2009;361(10):980-989.
Cardiac Resynchronization Therapy with Implantable Cardioverter Defibrillator Placement Decreases Heart Failure
Clinical question: Does cardiac resynchronization therapy (CRT) with biventricular pacing decrease cardiac events in patients with reduced ejection fraction (EF) and wide QRS complex but only mild cardiac symptoms?
Background: In patients with severely reduced EF, implantable cardioverter defibrillators (ICDs) have been shown to improve survival. Meanwhile, CRT decreases heart-failure-related hospitalizations for patients with advanced heart-failure symptoms, EF less than 35%, and intraventricular conduction delay. It is not as clear whether patients with less-severe symptoms benefit from CRT.
Study design: Randomized, controlled trial.
Setting: 110 medical centers in the U.S., Canada, and Europe.
Synopsis: This Multicenter Automatic Defibrillator Implantation Trial with Cardiac Resynchronization Therapy (MADIT-CRT) study randomly assigned 1,820 adults with EF less than 30%, New York Health Association Class I or II congestive heart failure, and in sinus rhythm with QRS greater than 130 msec to receive ICD with CRT or ICD alone. The primary endpoint was all-cause mortality or nonfatal heart-failure events. Average followup was 2.4 years.
A 34% reduction in the primary endpoint was found in the ICD-CRT group when compared with the ICD-only group, primarily due to a 41% reduction in heart-failure events. In a subgroup analysis, women and patients with QRS greater than 150 msec experienced particular benefit. Echocardiography one year after device implantation demonstrated significant reductions in left ventricular end-systolic and end-diastolic volume, and a significant increase in EF with ICD-CRT versus ICD-only (P<0.001).
Bottom line: Compared with ICD alone, CRT in combination with ICD prevented heart-failure events in relatively asymptomatic heart-failure patients with low EF and prolonged QRS.
Citation: Moss AJ, Hall WJ, Cannom DS, et al. Cardiac-resynchronization therapy for the prevention of heart-failure events. N Engl J Med. 2009;361(14):1329-1338.
Dabigatran Is Not Inferior to Warfarin in Atrial Fibrillation
Clinical question: Is dabigatran, an oral thrombin inhibitor, an effective and safe alternative to warfarin in patients with atrial fibrillation?
Background: Warfarin reduces the risk of stroke among patients with atrial fibrillation (AF) but requires frequent laboratory monitoring. Dabigatran is an oral direct thrombin inhibitor given in fixed dosages without laboratory monitoring.
Study design: Randomized, multicenter, open-label, noninferiority trial.
Setting: 951 clinical centers in 44 countries.
Synopsis: More than 18,000 patients 65 and older with AF and at least one stroke risk factor were enrolled. The average CHADS2 score was 2.1. Patients were randomized to receive fixed doses of dabigatran (110 mg or 150 mg, twice daily) or warfarin adjusted to an INR of 2.0-3.0. The primary outcomes were a) stroke or systemic embolism and b) major hemorrhage. Median followup was two years.
The annual rates of stroke or systemic embolism for both doses of dabigatran were noninferior to warfarin (P<0.001); higher-dose dabigatran was statistically superior to warfarin (relative risk (RR)=0.66, P<0.001). The annual rate of major hemorrhage was lowest in the lower-dose dabigatran group (RR=0.80, P=0.003 compared with warfarin); the higher-dose dabigatran and warfarin groups had equivalent rates of major bleeding. No increased risk of liver function abnormalities was noted.
Bottom line: Dabigatran appears to be an effective and safe alternative to warfarin in AF patients. If the drug were to be FDA-approved, appropriate patient selection and cost will need to be established.
Citation: Connolly SJ, Ezekowitz MD, Yusuf S, et al. Dabigatran versus warfarin in patients with atrial fibrillation. N Engl J Med. 2009;361(12):1139-1151.
Resident Fatigue and Distress Contribute to Perceived Medical Errors
Clinical question: Do resident fatigue and distress contribute to medical errors?
Background: In recent years, such measures as work-hour limitations have been implemented to decrease resident fatigue and, it is presumed, medical errors. However, few studies address the relationship between residents’ well-being and self-reported medical errors.
Study design: Prospective six-year longitudinal cohort study.
Setting: Single academic medical center.
Synopsis: The authors had 380 internal-medicine residents complete quarterly surveys to assess fatigue, quality of life, burnout, symptoms of depression, and frequency of perceived medical errors. In a univariate analysis, fatigue/sleepiness, burnout, depression, and overall quality of life measures correlated significantly with self-reported major medical errors. Fatigue/sleepiness and measures of distress additively increased the risk of self-reported errors. Increases in one or both domains were estimated to increase the risk of self-reported errors by as much as 15% to 28%.
The authors studied only self-reported medical errors. It is difficult to know whether these errors directly affected patient outcomes. Additionally, results of this single-site study might not be able to be generalized.
Bottom line: Fatigue and distress contribute to self-perceived medical errors among residents.
Citation: West CP, Tan AD, Habermann TM, Sloan JA, Shanafelt TD. Association of resident fatigue and distress with perceived medical errors. JAMA. 2009;302(12):1294-1300.
Inhaled Corticosteroids Decrease Inflammation in Moderate to Severe COPD
Clinical question: Does long-term inhaled corticosteroid therapy, with and without long-acting beta-agonists, decrease airway inflammation and improve lung function in patients with moderate to severe chronic obstructive pulmonary disease (COPD)?
Background: Guideline-recommended treatment of COPD with inhaled corticosteroids and long-acting beta-agonists improves symptoms and exacerbation rates; little is known about the impact of these therapies on inflammation and long-term lung function.
Study design: Randomized, double-blind, placebo-controlled trial.
Setting: Two university medical centers in the Netherlands.
Synopsis: One hundred one steroid-naïve patients, ages 45 to 75 who were current or former smokers with moderate to severe COPD, were randomized to one of four regimens: 1) fluticasone for six months, then placebo for 24 months; 2) fluticasone for 30 months; 3) fluticasone and salmeterol for 30 months; or 4) placebo for 30 months. The primary outcome was inflammatory cell counts in bronchial biopsies/induced sputum. Secondary outcomes included postbronchodilator spirometry, methacholine hyperresponsiveness, and self-reported symptoms and health status. Patients with asthma were excluded.
Short-term fluticasone therapy decreased inflammation and improved forced expiratory volume in one second (FEV1). Long-term therapy also decreased the rate of FEV1 decline, reduced dyspnea, and improved health status. Discontinuation of therapy at six months led to inflammation relapse with worsened symptoms and increased rate of FEV1 decline. The addition of long-acting beta-agonists did not provide additional anti-inflammatory benefits, but it did improve FEV1 and dyspnea at six months.
Additional studies are needed to further define clinical outcomes and assess the cost benefit of these therapies.
Bottom line: Inhaled corticosteroids decrease inflammation in steroid-naïve patients with moderate to severe COPD and might decrease the rate of lung function decline. Long-acting beta-agonists do not offer additional anti-inflammatory benefit.
Citation: Lapperre TS, Snoeck-Stroband JB, Gosman MM, et al. Effect of fluticasone with and without salmeterol on pulmonary outcomes in chronic obstructive pulmonary disease: a randomized trial. Ann Intern Med. 2009;151(8):517-527.
Initiation of Dialysis Does Not Help Maintain Functional Status in Elderly
Clinical question: Is functional status in the elderly maintained over time after initiating long-term dialysis?
Background: Quality-of-life maintenance often is used as a goal when initiating long-term dialysis in elderly patients with end-stage renal disease. More elderly patients are being offered long-term dialysis treatment. Little is known about the functional status of elderly patients on long-term dialysis.
Study design: Retrospective cohort study.
Setting: U.S. nursing homes.
Synopsis: By cross-linking data from two population-based administrative datasets, this study identified 3,702 nursing home patients (mean 73.4 years) who had started long-term dialysis and whose functional status had been assessed. Activities of daily living assessments before and at three-month intervals after dialysis initiation were compared to see if functional status was maintained.
Within three months of starting dialysis, 61% of patients had a decline in functional status or had died. By one year, only 1 in 8 patients had maintained their pre-dialysis functional status.
Decline in functional status cannot be attributed solely to dialysis because study patients were not compared to patients with chronic kidney disease who were not dialyzed. In addition, these results might not apply to all elderly patients on dialysis, as the functional status of elderly nursing home patients might differ significantly from those living at home.
Bottom line: Functional status is not maintained in most elderly nursing home patients in the first 12 months after long-term dialysis is initiated. Elderly patients considering dialysis treatment should be aware that dialysis might not help maintain functional status and quality of life.
Citation: Kurella Tamura MK, Covinsky KE, Chertow GM, Yaffe C, Landefeld CS, McCulloch CE. Functional status of elderly adults before and after initiation of dialysis. N Engl J Med. 2009;361(16):1539-1547.
Adding Basal Insulin to Oral Agents in Type 2 Diabetes Might Offer Best Glycemic Control
Clinical question: When added to oral diabetic agents, which insulin regimen (biphasic, prandial or basal) best achieves glycemic control in patients with Type 2 diabetes?
Background: Most patients with Type 2 diabetes mellitus (DM2) require insulin when oral agents provide suboptimal glycemic control. Little is known about which insulin regimen is most effective.
Study design: Three-year, open-label, multicenter trial.
Setting: Fifty-eight clinical centers in the United Kingdom and Ireland.
Synopsis: The authors randomized 708 insulin-naïve DM2 patients (median age 62 years) with HgbA1c 7% to 10% on maximum-dose metformin or sulfonylurea to one of three regimens: biphasic insulin twice daily; prandial insulin three times daily; or basal insulin once daily. Outcomes were HgbA1c, hypoglycemia rates, and weight gain. Sulfonylureas were replaced by another insulin if glycemic control was unacceptable.
The patients were mostly Caucasian and overweight. At three years of followup, median HgbA1c was similar in all groups (7.1% biphasic, 6.8% prandial, 6.9% basal); however, more patients who received prandial or basal insulin achieved HgbA1c less than 6.5% (45% and 43%, respectively) than in the biphasic group (32%).
Hypoglycemia was significantly less frequent in the basal insulin group (1.7 per patient per year versus 3.0 and 5.5 with biphasic and prandial, respectively). Patients gained weight in all groups; the greatest gain was with prandial insulin. At three years, there were no significant between-group differences in blood pressure, cholesterol, albuminuria, or quality of life.
Bottom line: Adding insulin to oral diabetic regimens improves glycemic control. Basal or prandial insulin regimens achieve glycemic targets more frequently than biphasic dosing.
Citation: Holman RR, Farmer AJ, Davies MJ, et al. Three-year efficacy of complex insulin regimens in type 2 diabetes. N Engl J Med. 2009;361(18):1736-1747.
Advanced Dementia Is a Terminal Illness with High Morbidity and Mortality
Clinical question: Does understanding the expected clinical course of advanced dementia influence end-of-life decisions by proxy decision-makers?
Background: Advanced dementia is a leading cause of death in the United States, but the clinical course of advanced dementia has not been described in a rigorous, prospective manner. The lack of information might cause risk to be underestimated, and patients might receive suboptimal palliative care.
Study design: Multicenter prospective cohort study.
Setting: Twenty-two nursing homes in a single U.S. city.
Synopsis: The survey examined 323 nursing home residents with advanced dementia. The patients were clinically assessed at baseline and quarterly for 18 months through chart reviews, nursing interviews, and physical examinations. Additionally, their proxies were surveyed regarding their understanding of the subjects’ prognoses.
During the survey period, 41.1% of patients developed pneumonia, 52.6% of patients experienced a febrile episode, and 85.8% of patients developed an eating problem; cumulative all-cause mortality was 54.8%. Adjusted for age, sex, and disease duration, the six-month mortality rate for subjects who had pneumonia was 46.7%; a febrile episode, 44.5%; and an eating problem, 38.6%.
Distressing symptoms, including dyspnea (46.0%) and pain (39.1%), were common. In the last three months of life, 40.7% of subjects underwent at least one burdensome intervention (defined as hospitalization, ED visit, parenteral therapy, or tube feeding).
Subjects whose proxies reported an understanding of the poor prognosis and expected clinical complications of advanced dementia underwent significantly fewer burdensome interventions (adjusted odds ratio 0.12).
Bottom line: Advanced dementia is associated with frequent complications, including infections and eating problems, with high six-month mortality and significant associated morbidity. Patients whose healthcare proxies have a good understanding of the expected clinical course and prognosis receive less-aggressive end-of-life care.
Citation: Mitchell SL, Teno JM, Kiely DK, et al. The clinical course of advanced dementia. N Engl J Med. 2009;361(16):1529-1538. TH
Prevent Defense
Three U.S. medical centers have been recognized for innovative approaches to preventing DVT and its potentially fatal complications, which include pulmonary embolism (PE). Central to each of the DVT prevention strategies is a risk assessment tool that is easy to use, built directly into routine care, and linked directly to guideline-recommended choices for prophylaxis.
The University of California at San Diego (UCSD) Medical Center, Johns Hopkins Hospital in Baltimore, and the Veterans Affairs (VA) Medical Center in Washington, D.C., each received the first DVTeamCare Hospital Award. The North American Thrombosis Forum (NATF), in conjunction with pharmaceutical company Eisai Inc., recognized each center’s accomplishment based upon an evaluation by an independent panel of expert judges.

—Gregory A. Maynard, MD, FHM, hospital medicine division chief, University of California at San Diego
The award reflects NATF’s goal of enhancing thrombosis education, prevention, diagnosis, and treatment to improve patient outcomes, says NATF Executive Director Ilene Sussman, PhD. Dr. Sussman notes that DVT affects more than 600,000 Americans annually, kills more than 100,000, and is one of the leading causes of preventable deaths in hospitals. Preventable DVT-related complication is on Medicare’s list of “never events,” for which hospitals will no longer be reimbursed.
UCSD, representing medical centers with more than 200 beds, imbedded its VTE prevention protocol into admission, transfer, and perioperative order sets across all medical and surgical services, says Gregory A. Maynard, MD, FHM, hospital medicine division chief. The protocol flags three levels of DVT risk, notes possible contraindications for a particular kind of patient, and presents a set of options for guideline-recommended prophylaxis. The protocol can be paper- or computer-based. Prompting concurrent intervention is a central component of UCSD’s implementation strategy, “identifying in real-time patients who are not receiving the right DVT prophylaxis and having a front-line nurse or pharmacist intervene appropriately,” Dr. Maynard explains.
The percent of UCSD’s patients on adequate prophylaxis rose to more than 98% in the past two years, up from about 50% before the intervention, while preventable VTE dropped by 85%—about 50 fewer cases per year in a hospital with fewer than 300 beds. “Having DVT prevention protocols such as these in place allows hospitalists to provide better care with less effort by leaving hospitalists free to focus on more complicated patient-care issues,” Dr. Maynard says.
UCSD has partnered with SHM to develop DVT prevention toolkits and mentored collaboratives, with which hospitalists can take the lead on QI projects at their local institutions. SHM’s online VTE Implementation Guide is available at www.hospitalmedicine.org/ResourceRoomRedesign/RR_VTE/VTE_Home.cfm.
Johns Hopkins Hospital, representing medical centers with more than 200 beds, developed a mandatory computer-based decision-support system to facilitate specialty-specific risk-factor assessment and the application of risk-appropriate VTE prophylaxis, says Michael Streiff, MD, FACP, director of Johns Hopkins’ Anticoagulation Management Service and Outpatient Clinic, and a member of its Evidence-Based Practice Center. Before a physician can issue any orders—medications, lab tests, nursing instructions, etc.—using a physician transfer order set, the computerized order-entry system automatically guides them through a concise set of questions about a patient’s DVT risk factors, contraindications for blood thinners, and guideline-recommended prophylaxis choices, Dr. Streiff says.
Since implementing the system, the percent of patients being DVT-risk-stratified within 24 hours of hospital admission rose to more than 90%, and nearly 9 in 10 of the appropriate patients are now receiving risk-appropriate, American College of Chest Physicians-approved DVT prophylaxis, up from about 26% before the intervention, Dr. Streiff notes.
The VA Medical Center in Washington, D.C., representing medical centers with fewer than 200 beds, participated in a mentorship collaborative with UCSD’s Dr. Maynard and designed a seven-step process that walks providers through an evidence-based risk-factor assessment to determine appropriate thromboprophylactic therapy, says Divya Shroff, MD, associate chief of staff, Informatics. The guideline-driven steps are integrated into the VA’s computerized patient medical record system and take no more than 60 seconds to follow, says pharmacy practice resident Jovonne H. Jones, PharmD. The steps include:
- Assess patient DVT risk level;
- Educate patient about the order;
- Identify contraindications, if any;
- Choose prophylaxis drug or device;
- Accept order for drug or device;
- Check if additional prophylactic method is needed; and
- Accept the final order.
After the intervention, the rate at which patients receive appropriate prophylaxis upon admission more than doubled. Twenty VA medical centers around the country are in the process of implementing the system, Jones says.
The award-winning protocols will be presented at an NATF-hosted program April 9 at Harvard Medical School. The protocols and implementation plans will be made available at www.DVTeamCareAward.com to help other hospitals enhance their efforts to prevent DVT. TH
Chris Guadagnino is a freelance medical writer based in Philadelphia.
Three U.S. medical centers have been recognized for innovative approaches to preventing DVT and its potentially fatal complications, which include pulmonary embolism (PE). Central to each of the DVT prevention strategies is a risk assessment tool that is easy to use, built directly into routine care, and linked directly to guideline-recommended choices for prophylaxis.
The University of California at San Diego (UCSD) Medical Center, Johns Hopkins Hospital in Baltimore, and the Veterans Affairs (VA) Medical Center in Washington, D.C., each received the first DVTeamCare Hospital Award. The North American Thrombosis Forum (NATF), in conjunction with pharmaceutical company Eisai Inc., recognized each center’s accomplishment based upon an evaluation by an independent panel of expert judges.

—Gregory A. Maynard, MD, FHM, hospital medicine division chief, University of California at San Diego
The award reflects NATF’s goal of enhancing thrombosis education, prevention, diagnosis, and treatment to improve patient outcomes, says NATF Executive Director Ilene Sussman, PhD. Dr. Sussman notes that DVT affects more than 600,000 Americans annually, kills more than 100,000, and is one of the leading causes of preventable deaths in hospitals. Preventable DVT-related complication is on Medicare’s list of “never events,” for which hospitals will no longer be reimbursed.
UCSD, representing medical centers with more than 200 beds, imbedded its VTE prevention protocol into admission, transfer, and perioperative order sets across all medical and surgical services, says Gregory A. Maynard, MD, FHM, hospital medicine division chief. The protocol flags three levels of DVT risk, notes possible contraindications for a particular kind of patient, and presents a set of options for guideline-recommended prophylaxis. The protocol can be paper- or computer-based. Prompting concurrent intervention is a central component of UCSD’s implementation strategy, “identifying in real-time patients who are not receiving the right DVT prophylaxis and having a front-line nurse or pharmacist intervene appropriately,” Dr. Maynard explains.
The percent of UCSD’s patients on adequate prophylaxis rose to more than 98% in the past two years, up from about 50% before the intervention, while preventable VTE dropped by 85%—about 50 fewer cases per year in a hospital with fewer than 300 beds. “Having DVT prevention protocols such as these in place allows hospitalists to provide better care with less effort by leaving hospitalists free to focus on more complicated patient-care issues,” Dr. Maynard says.
UCSD has partnered with SHM to develop DVT prevention toolkits and mentored collaboratives, with which hospitalists can take the lead on QI projects at their local institutions. SHM’s online VTE Implementation Guide is available at www.hospitalmedicine.org/ResourceRoomRedesign/RR_VTE/VTE_Home.cfm.
Johns Hopkins Hospital, representing medical centers with more than 200 beds, developed a mandatory computer-based decision-support system to facilitate specialty-specific risk-factor assessment and the application of risk-appropriate VTE prophylaxis, says Michael Streiff, MD, FACP, director of Johns Hopkins’ Anticoagulation Management Service and Outpatient Clinic, and a member of its Evidence-Based Practice Center. Before a physician can issue any orders—medications, lab tests, nursing instructions, etc.—using a physician transfer order set, the computerized order-entry system automatically guides them through a concise set of questions about a patient’s DVT risk factors, contraindications for blood thinners, and guideline-recommended prophylaxis choices, Dr. Streiff says.
Since implementing the system, the percent of patients being DVT-risk-stratified within 24 hours of hospital admission rose to more than 90%, and nearly 9 in 10 of the appropriate patients are now receiving risk-appropriate, American College of Chest Physicians-approved DVT prophylaxis, up from about 26% before the intervention, Dr. Streiff notes.
The VA Medical Center in Washington, D.C., representing medical centers with fewer than 200 beds, participated in a mentorship collaborative with UCSD’s Dr. Maynard and designed a seven-step process that walks providers through an evidence-based risk-factor assessment to determine appropriate thromboprophylactic therapy, says Divya Shroff, MD, associate chief of staff, Informatics. The guideline-driven steps are integrated into the VA’s computerized patient medical record system and take no more than 60 seconds to follow, says pharmacy practice resident Jovonne H. Jones, PharmD. The steps include:
- Assess patient DVT risk level;
- Educate patient about the order;
- Identify contraindications, if any;
- Choose prophylaxis drug or device;
- Accept order for drug or device;
- Check if additional prophylactic method is needed; and
- Accept the final order.
After the intervention, the rate at which patients receive appropriate prophylaxis upon admission more than doubled. Twenty VA medical centers around the country are in the process of implementing the system, Jones says.
The award-winning protocols will be presented at an NATF-hosted program April 9 at Harvard Medical School. The protocols and implementation plans will be made available at www.DVTeamCareAward.com to help other hospitals enhance their efforts to prevent DVT. TH
Chris Guadagnino is a freelance medical writer based in Philadelphia.
Three U.S. medical centers have been recognized for innovative approaches to preventing DVT and its potentially fatal complications, which include pulmonary embolism (PE). Central to each of the DVT prevention strategies is a risk assessment tool that is easy to use, built directly into routine care, and linked directly to guideline-recommended choices for prophylaxis.
The University of California at San Diego (UCSD) Medical Center, Johns Hopkins Hospital in Baltimore, and the Veterans Affairs (VA) Medical Center in Washington, D.C., each received the first DVTeamCare Hospital Award. The North American Thrombosis Forum (NATF), in conjunction with pharmaceutical company Eisai Inc., recognized each center’s accomplishment based upon an evaluation by an independent panel of expert judges.

—Gregory A. Maynard, MD, FHM, hospital medicine division chief, University of California at San Diego
The award reflects NATF’s goal of enhancing thrombosis education, prevention, diagnosis, and treatment to improve patient outcomes, says NATF Executive Director Ilene Sussman, PhD. Dr. Sussman notes that DVT affects more than 600,000 Americans annually, kills more than 100,000, and is one of the leading causes of preventable deaths in hospitals. Preventable DVT-related complication is on Medicare’s list of “never events,” for which hospitals will no longer be reimbursed.
UCSD, representing medical centers with more than 200 beds, imbedded its VTE prevention protocol into admission, transfer, and perioperative order sets across all medical and surgical services, says Gregory A. Maynard, MD, FHM, hospital medicine division chief. The protocol flags three levels of DVT risk, notes possible contraindications for a particular kind of patient, and presents a set of options for guideline-recommended prophylaxis. The protocol can be paper- or computer-based. Prompting concurrent intervention is a central component of UCSD’s implementation strategy, “identifying in real-time patients who are not receiving the right DVT prophylaxis and having a front-line nurse or pharmacist intervene appropriately,” Dr. Maynard explains.
The percent of UCSD’s patients on adequate prophylaxis rose to more than 98% in the past two years, up from about 50% before the intervention, while preventable VTE dropped by 85%—about 50 fewer cases per year in a hospital with fewer than 300 beds. “Having DVT prevention protocols such as these in place allows hospitalists to provide better care with less effort by leaving hospitalists free to focus on more complicated patient-care issues,” Dr. Maynard says.
UCSD has partnered with SHM to develop DVT prevention toolkits and mentored collaboratives, with which hospitalists can take the lead on QI projects at their local institutions. SHM’s online VTE Implementation Guide is available at www.hospitalmedicine.org/ResourceRoomRedesign/RR_VTE/VTE_Home.cfm.
Johns Hopkins Hospital, representing medical centers with more than 200 beds, developed a mandatory computer-based decision-support system to facilitate specialty-specific risk-factor assessment and the application of risk-appropriate VTE prophylaxis, says Michael Streiff, MD, FACP, director of Johns Hopkins’ Anticoagulation Management Service and Outpatient Clinic, and a member of its Evidence-Based Practice Center. Before a physician can issue any orders—medications, lab tests, nursing instructions, etc.—using a physician transfer order set, the computerized order-entry system automatically guides them through a concise set of questions about a patient’s DVT risk factors, contraindications for blood thinners, and guideline-recommended prophylaxis choices, Dr. Streiff says.
Since implementing the system, the percent of patients being DVT-risk-stratified within 24 hours of hospital admission rose to more than 90%, and nearly 9 in 10 of the appropriate patients are now receiving risk-appropriate, American College of Chest Physicians-approved DVT prophylaxis, up from about 26% before the intervention, Dr. Streiff notes.
The VA Medical Center in Washington, D.C., representing medical centers with fewer than 200 beds, participated in a mentorship collaborative with UCSD’s Dr. Maynard and designed a seven-step process that walks providers through an evidence-based risk-factor assessment to determine appropriate thromboprophylactic therapy, says Divya Shroff, MD, associate chief of staff, Informatics. The guideline-driven steps are integrated into the VA’s computerized patient medical record system and take no more than 60 seconds to follow, says pharmacy practice resident Jovonne H. Jones, PharmD. The steps include:
- Assess patient DVT risk level;
- Educate patient about the order;
- Identify contraindications, if any;
- Choose prophylaxis drug or device;
- Accept order for drug or device;
- Check if additional prophylactic method is needed; and
- Accept the final order.
After the intervention, the rate at which patients receive appropriate prophylaxis upon admission more than doubled. Twenty VA medical centers around the country are in the process of implementing the system, Jones says.
The award-winning protocols will be presented at an NATF-hosted program April 9 at Harvard Medical School. The protocols and implementation plans will be made available at www.DVTeamCareAward.com to help other hospitals enhance their efforts to prevent DVT. TH
Chris Guadagnino is a freelance medical writer based in Philadelphia.
Hospital-Acquired Conditions & The Hospitalist
Hospitalist Neal Axon, MD, first became aware of an important change in his hospital’s policies last year while attending to an elderly patient the morning after admission to the community hospital where he works part time.
“This new form appeared in the chart requesting a urinalysis for my patient, who’d had a Foley catheter placed,” says Dr. Axon, an assistant professor of medicine at the Medical University of South Carolina in Charleston. “I didn’t know why, so I asked. I was told that it was now necessary to document that there was no UTI present on admission.” He asked the charge nurse, “So what do I do now that the catheter has been in place for 12 hours and has colonization without a true infection?”
The next thing he heard: silence.
The new form Dr. Axon encountered was an outgrowth of the requirements of the Deficit Reduction Act (DRA) of 2005, which ordered Medicare to withhold additional hospital payments for hospital-acquired complications (HAC) developed during a hospital stay. One result of the new rule is that much of a hospital’s response to these initiatives has been placed in the hands of the hospitalist. From accurate documentation of complications already present on admission (POA), to confirming that guidelines for treatment are being followed, to taking the lead on review of staff practices and education, hospitalists are in a position to have a wide-ranging impact on patient care and the financial health of their institutions.
Congress Pushes Reforms
In order for Medicare to not provide a reimbursement, an HAC has to be high-cost and/or high-volume, result in the assignment of the case to a higher payment when present as a secondary diagnosis, and “could reasonably be prevented through the application of evidence-based guidelines,” says Barry Straube, MD, chief medical officer and director of the Office of Clinical Standards and Quality at the Centers for Medicare and Medicaid Services (CMS). “CMS was to implement a process where we would not pay the hospitals additional money for these complications.”
The new rules mean Medicare pays hospitals on the basis of Medicare Severity Diagnostic-Related Groups (MS-DRG), which better reflect the complexity of a patient’s illness. The biggest change was a three-tiered payment schedule: a base level for the diagnosis, a second level adding money to reflect the presence of comorbidities and complications, and a third for major complications and comorbidities (see Table 1, p. 31).
“Instituting HACs means that hospitals would no longer receive the comorbidity and complication payments if the only reason a case qualified for higher payment was the HAC,” Dr. Straube explains. “We did carve out a POA exception for those conditions that were acquired outside of the hospital. HACs only impact additional payments; the hospitals are still paid for the diagnosis that resulted in the hospital admission.”
CMS also identifies three “never events” it won’t reimburse for (see “A Brief History of Never Events,” p. 35): performing the wrong procedure, performing a procedure on the wrong body part, and performing a procedure on the wrong patient. “Neither hospitals nor physicians that are involved in such egregious situations would be paid,” Dr. Straube says.
Preventability: Subject of Controversy
The big questions surrounding HACs: Could they reasonably be prevented through the application of evidence-based guidelines? How preventable are HACs? Who decides if a complication is preventable, and therefore payment for services is withheld?
They’re concerns that are widespread among physicians, hospital administrators, and regulators alike.
“The legislation required the conditions to be ‘reasonably preventable’ using established clinical guidelines,” Dr. Straube says. “We did not have to show 100% prevention. In an imperfect world, they might still take place occasionally, but with good medical care, almost all of these are preventable in this day and age.”
For CMS, the preventable conditions are an either/or situation: Either they existed prior to admission and are subject to payment, or they did not exist at admission and additional payment for the complication will not be made. “HACs do not currently consider a patient’s individual risk for complications,” says Jennifer Meddings, MD, MSc, clinical lecturer and health researcher in the Department of Internal Medicine at the University of Michigan Health System in Ann Arbor. “We know the best strategies to prevent complications in ideal patients, and these are reflected in the HACs. In real life, many of our patients just don’t fit into the guidelines for many reasons—and you have to individualize care.”
Dr. Meddings points to DVT as a prime example. For a certain number of inpatients, the guidelines can be followed to perfection. In other patients (e.g., those with kidney conditions), previous reactions to a medication or an individual’s predisposition to clotting might interfere with treatment. However, CMS doesn’t allow appeals of nonpayment decisions for HACs based on individual circumstances.
Some experts think the rigidness of the payment policy forces physicians to treat patients exactly to guidelines. Even then, payment could be declined if an HAC develops.
“One of the points of most discussion is how preventable some of these are, particularly when choosing those you are no longer going to pay for,” Dr. Meddings says. “Many of the complications currently under review have patients that are at higher risk than others. How much our prevention strategies can alleviate or reduce the risk varies widely among patients.”
Impact on HM Practice
Many of the preventable conditions outlined by CMS do not directly affect hospitalist payment. However, hospitalists often find themselves responsible for properly documenting admission and care.
“The rule changes regarding payment for HACs are only related to hospital payments, and to date, most physicians, including hospitalists, are not directly at financial risk,” says Heidi Wald, MD, MSPH, hospitalist and assistant professor of medicine in the divisions of Health Care Policy Research and General Internal Medicine at the University of Colorado Denver School of Medicine. “Although hospitalists have no financial skin in the game, there are plenty of reasons they would take an interest in addressing HACs in their hospital. In particular, they are often seen as the ‘go-to’ group for quality improvement in their hospitals.”
For example, some HM groups have been active in working with teams of physicians, nurses, and other healthcare providers to address local policies and procedures on prevention of catheter-associated urinary tract infections (UTIs) and DVT.
“This has certainly necessitated a team approach,” says Shaun Frost, MD, FHM, an SHM board member and regional medical director for Cogent Healthcare in St. Paul, Minn. “For many of the HACs that apply to our population of patients, the hospitalist alone cannot be expected to solely execute effective quality improvements. It takes a team effort in that regard, and one that includes many different disciplines.”
The Cogent-affiliated hospitalist group at Temple University Hospital in Philadelphia formed a task force to address issues with catheter-associated UTI. One initiative focused on educating all providers involved in the proper care of the catheters and similar interventions. A secondary focus of the project was an inventory of current practices and procedures.
“It was discovered that we did not have an automatic stop order for Foley catheters, so in some situations, they were likely being left in longer than needed while nursing [staff] tried to contact a physician,” Dr. Frost explains. “We created standardized order sets that include criteria for continuing the catheter. Once the criteria are no longer applicable, nursing will be able to discontinue it.”
Although CMS has only recently turned the spotlight on HACs and never events, hospitalists have been heavily involved in the patient-safety arena for years. “It is not a new phenomenon that hospitalists work for healthcare delivery and healthcare system improvement,” Dr. Meddings says.
Hospital administration at Temple University Hospital recognized the HM group’s quality-improvement (QI) work, and has “specifically charged us with spreading the work we have done in patient safety to the entire house,” Dr. Frost says. “That speaks to the administration’s opinion of the power of the HM program to assist with institution-wide QI initiatives.”
Documentation Is Key
Beyond applying proven methods to avoid HACs, hospitalists can make a difference through documentation. If the hospitalist notes all conditions when the patient first presents to the hospital, additional comorbidity and complication payments should be made.
“The part that probably has the greatest impact on the day-to-day practice of a hospitalist is the increased importance of documentation throughout the hospital episode,” Dr. Meddings says. “If complications are occurring and they are not present in the chart, the coders may not recognize that it has occurred and will not know to include it in the bill. This can have an adverse impact on the hospital and its finances.”
Documentation issues can impact hospital payment in several ways:
- Hospitals might receive additional payment by default if certain HACs are described incorrectly or without sufficient detail (e.g., receiving overpayment because the physician did not indicate a UTI was in fact a catheter-associated UTI);
- As more attention is invested in documenting all conditions POA, hospitals might be coding more comorbidities overall than previously, which also will generate additional payment for hospitals as any POA condition is eligible for increased payment; and
- Hospitals might lose payment when admitting providers fail to adequately document the condition as POA (e.g., a pre-existing decubitus ulcer not detected until the second day of the hospital stay).
The descriptions to be used in coding are very detailed. UTIs, for example, have one code to document the POA assessment, another code to show that a UTI occurred, and a third code to indicate it was catheter-associated. Each code requires appropriate documentation in the chart (see Table 1, above).
The impact hospitalists have on care and payment is not the same across the HAC spectrum. For instance, documenting the presence of pressure ulcers might be easier than distinguishing colonization from infection in those admitted with in-dwelling urinary catheters. Others, such as DVT or vascular catheter-associated infections, are rarely POA unless they are part of the admitting diagnosis.
“This new focus on hospital-acquired conditions may work to the patient’s benefit,” Dr. Meddings says. “The inclusion of pressure ulcers has led to increased attention to skin exams on admission and preventive measures during hospitalizations. In the past, skin exams upon admission may have been given a lower priority, but that has changed.”
Dr. Meddings is concerned that the new rules could force the shifting of resources to areas where the hospital could lose money. If, when, and how many changes will actually take place is still up in the air. “Resource shifting is a concern whenever there is any sort of pay-for-performance attention directed toward one particular complication,” she says. “To balance this, many of the strategies hospitals used to prevent complications are not specific to just the diagnosis that is covered by the HAC.”
Dr. Meddings also hopes the new focus on preventable conditions will have a “halo effect” in the healthcare community. For instance, CMS mandating DVT prevention following orthopedic operations will, hopefully, result in a greater awareness of the problem in other susceptible patients.
POA Indicators
Since hospitalists often perform the initial patient history, physical, and other admission work, they are in the best position to find and document POA indicators (see Table 2, p. below). Proper notes on such things as UTIs present and the state of skin integrity are an important part of making sure the hospital is paid correctly for the care it provides.
Education on the specific definition of each potential HAC is required to help physicians avoid overtreatment of certain conditions, especially UTIs. For example, the Centers for Disease Control and Prevention (CDC) defines all UTIs as symptomatic. Therefore, the screening of all admitted patients, regardless of symptoms, is wasteful and unlikely to help the hospital’s bottom line.
“If you start screening everyone that comes through the door so you don’t miss any pre-existing UTIs, you are going to find a lot of asymptomatic colonization,” Dr. Wald says. “You are also going to spend a lot of money and time on studies and possibly treatments that may not yield many true infections. It is important that physicians know the definition of these HACs to help avoid needless interventions.”
Minimal Loss
Many hospital administrators and physicians were worried when the HAC program was first announced. Much of the stress and concern, however, seems to have dissipated. CMS estimated the HAC program would save Medicare $21 million in fiscal year 2009. Others, such as Peter McNair and colleagues writing in Health Affairs, suggest the actual impact is closer to $1.1 million.1 The CMS-projected impact of the HAC provision in fiscal-year 2009 was $21 million, out of more than $100 billion in payments.
“I think the HACs will not have a major impact because of the way payments are made,” says internist Robert Berenson, MD, a fellow at the Urban Institute in Washington, D.C., who has studied Medicare policy issues extensively, and for two years was in charge of Medicare payment policies at the Health Care Finance Administration, the precursor to CMS. “Patients who have HACs often have another comorbidity that would kick them into a higher payment category regardless of the presence of a hospital-acquired complication. In the end, it is probably more symbolic and unlikely to make a major dent in hospital income—at least at this point.”
Another limitation to CMS nonpayment for HACs is the issue of deciding which conditions are truly preventable. Dr. Berenson questions the ability of the current system to identify many additional complications for which this approach will be feasible.
“CMS has laid out its strategy, suggesting that we should be able to continue increasing the number of conditions for which providers would be paid differently based on quality,” he says. “Many observers question whether there will ever be measurement tools that are robust enough, and there will be a wide agreement on the preventability of enough conditions that this initiative will go very far.”
Although hospitalists might not face a direct financial risk, they still have their hospitals’ best interest—and their reputations—on the line. “Hospitalists care about preventing complications,” Dr. Wald says. “We are very engaged in working with our hospitals to improve care, maximize quality, and minimize cost.” TH
Kurt Ullman is a freelance medical writer based in Indiana.
Reference
- McNair PD, Luft HS, Bindman AB. Medicare’s policy not to pay for treating hospital-acquired conditions: the impact. Health Aff (Millwood). 2009;28(5):1485-1493.
TOP IMAGE SOURCE: KAREEM RIZKHALLA/ISTOCKPHOTO.COM
Hospitalist Neal Axon, MD, first became aware of an important change in his hospital’s policies last year while attending to an elderly patient the morning after admission to the community hospital where he works part time.
“This new form appeared in the chart requesting a urinalysis for my patient, who’d had a Foley catheter placed,” says Dr. Axon, an assistant professor of medicine at the Medical University of South Carolina in Charleston. “I didn’t know why, so I asked. I was told that it was now necessary to document that there was no UTI present on admission.” He asked the charge nurse, “So what do I do now that the catheter has been in place for 12 hours and has colonization without a true infection?”
The next thing he heard: silence.
The new form Dr. Axon encountered was an outgrowth of the requirements of the Deficit Reduction Act (DRA) of 2005, which ordered Medicare to withhold additional hospital payments for hospital-acquired complications (HAC) developed during a hospital stay. One result of the new rule is that much of a hospital’s response to these initiatives has been placed in the hands of the hospitalist. From accurate documentation of complications already present on admission (POA), to confirming that guidelines for treatment are being followed, to taking the lead on review of staff practices and education, hospitalists are in a position to have a wide-ranging impact on patient care and the financial health of their institutions.
Congress Pushes Reforms
In order for Medicare to not provide a reimbursement, an HAC has to be high-cost and/or high-volume, result in the assignment of the case to a higher payment when present as a secondary diagnosis, and “could reasonably be prevented through the application of evidence-based guidelines,” says Barry Straube, MD, chief medical officer and director of the Office of Clinical Standards and Quality at the Centers for Medicare and Medicaid Services (CMS). “CMS was to implement a process where we would not pay the hospitals additional money for these complications.”
The new rules mean Medicare pays hospitals on the basis of Medicare Severity Diagnostic-Related Groups (MS-DRG), which better reflect the complexity of a patient’s illness. The biggest change was a three-tiered payment schedule: a base level for the diagnosis, a second level adding money to reflect the presence of comorbidities and complications, and a third for major complications and comorbidities (see Table 1, p. 31).
“Instituting HACs means that hospitals would no longer receive the comorbidity and complication payments if the only reason a case qualified for higher payment was the HAC,” Dr. Straube explains. “We did carve out a POA exception for those conditions that were acquired outside of the hospital. HACs only impact additional payments; the hospitals are still paid for the diagnosis that resulted in the hospital admission.”
CMS also identifies three “never events” it won’t reimburse for (see “A Brief History of Never Events,” p. 35): performing the wrong procedure, performing a procedure on the wrong body part, and performing a procedure on the wrong patient. “Neither hospitals nor physicians that are involved in such egregious situations would be paid,” Dr. Straube says.
Preventability: Subject of Controversy
The big questions surrounding HACs: Could they reasonably be prevented through the application of evidence-based guidelines? How preventable are HACs? Who decides if a complication is preventable, and therefore payment for services is withheld?
They’re concerns that are widespread among physicians, hospital administrators, and regulators alike.
“The legislation required the conditions to be ‘reasonably preventable’ using established clinical guidelines,” Dr. Straube says. “We did not have to show 100% prevention. In an imperfect world, they might still take place occasionally, but with good medical care, almost all of these are preventable in this day and age.”
For CMS, the preventable conditions are an either/or situation: Either they existed prior to admission and are subject to payment, or they did not exist at admission and additional payment for the complication will not be made. “HACs do not currently consider a patient’s individual risk for complications,” says Jennifer Meddings, MD, MSc, clinical lecturer and health researcher in the Department of Internal Medicine at the University of Michigan Health System in Ann Arbor. “We know the best strategies to prevent complications in ideal patients, and these are reflected in the HACs. In real life, many of our patients just don’t fit into the guidelines for many reasons—and you have to individualize care.”
Dr. Meddings points to DVT as a prime example. For a certain number of inpatients, the guidelines can be followed to perfection. In other patients (e.g., those with kidney conditions), previous reactions to a medication or an individual’s predisposition to clotting might interfere with treatment. However, CMS doesn’t allow appeals of nonpayment decisions for HACs based on individual circumstances.
Some experts think the rigidness of the payment policy forces physicians to treat patients exactly to guidelines. Even then, payment could be declined if an HAC develops.
“One of the points of most discussion is how preventable some of these are, particularly when choosing those you are no longer going to pay for,” Dr. Meddings says. “Many of the complications currently under review have patients that are at higher risk than others. How much our prevention strategies can alleviate or reduce the risk varies widely among patients.”
Impact on HM Practice
Many of the preventable conditions outlined by CMS do not directly affect hospitalist payment. However, hospitalists often find themselves responsible for properly documenting admission and care.
“The rule changes regarding payment for HACs are only related to hospital payments, and to date, most physicians, including hospitalists, are not directly at financial risk,” says Heidi Wald, MD, MSPH, hospitalist and assistant professor of medicine in the divisions of Health Care Policy Research and General Internal Medicine at the University of Colorado Denver School of Medicine. “Although hospitalists have no financial skin in the game, there are plenty of reasons they would take an interest in addressing HACs in their hospital. In particular, they are often seen as the ‘go-to’ group for quality improvement in their hospitals.”
For example, some HM groups have been active in working with teams of physicians, nurses, and other healthcare providers to address local policies and procedures on prevention of catheter-associated urinary tract infections (UTIs) and DVT.
“This has certainly necessitated a team approach,” says Shaun Frost, MD, FHM, an SHM board member and regional medical director for Cogent Healthcare in St. Paul, Minn. “For many of the HACs that apply to our population of patients, the hospitalist alone cannot be expected to solely execute effective quality improvements. It takes a team effort in that regard, and one that includes many different disciplines.”
The Cogent-affiliated hospitalist group at Temple University Hospital in Philadelphia formed a task force to address issues with catheter-associated UTI. One initiative focused on educating all providers involved in the proper care of the catheters and similar interventions. A secondary focus of the project was an inventory of current practices and procedures.
“It was discovered that we did not have an automatic stop order for Foley catheters, so in some situations, they were likely being left in longer than needed while nursing [staff] tried to contact a physician,” Dr. Frost explains. “We created standardized order sets that include criteria for continuing the catheter. Once the criteria are no longer applicable, nursing will be able to discontinue it.”
Although CMS has only recently turned the spotlight on HACs and never events, hospitalists have been heavily involved in the patient-safety arena for years. “It is not a new phenomenon that hospitalists work for healthcare delivery and healthcare system improvement,” Dr. Meddings says.
Hospital administration at Temple University Hospital recognized the HM group’s quality-improvement (QI) work, and has “specifically charged us with spreading the work we have done in patient safety to the entire house,” Dr. Frost says. “That speaks to the administration’s opinion of the power of the HM program to assist with institution-wide QI initiatives.”
Documentation Is Key
Beyond applying proven methods to avoid HACs, hospitalists can make a difference through documentation. If the hospitalist notes all conditions when the patient first presents to the hospital, additional comorbidity and complication payments should be made.
“The part that probably has the greatest impact on the day-to-day practice of a hospitalist is the increased importance of documentation throughout the hospital episode,” Dr. Meddings says. “If complications are occurring and they are not present in the chart, the coders may not recognize that it has occurred and will not know to include it in the bill. This can have an adverse impact on the hospital and its finances.”
Documentation issues can impact hospital payment in several ways:
- Hospitals might receive additional payment by default if certain HACs are described incorrectly or without sufficient detail (e.g., receiving overpayment because the physician did not indicate a UTI was in fact a catheter-associated UTI);
- As more attention is invested in documenting all conditions POA, hospitals might be coding more comorbidities overall than previously, which also will generate additional payment for hospitals as any POA condition is eligible for increased payment; and
- Hospitals might lose payment when admitting providers fail to adequately document the condition as POA (e.g., a pre-existing decubitus ulcer not detected until the second day of the hospital stay).
The descriptions to be used in coding are very detailed. UTIs, for example, have one code to document the POA assessment, another code to show that a UTI occurred, and a third code to indicate it was catheter-associated. Each code requires appropriate documentation in the chart (see Table 1, above).
The impact hospitalists have on care and payment is not the same across the HAC spectrum. For instance, documenting the presence of pressure ulcers might be easier than distinguishing colonization from infection in those admitted with in-dwelling urinary catheters. Others, such as DVT or vascular catheter-associated infections, are rarely POA unless they are part of the admitting diagnosis.
“This new focus on hospital-acquired conditions may work to the patient’s benefit,” Dr. Meddings says. “The inclusion of pressure ulcers has led to increased attention to skin exams on admission and preventive measures during hospitalizations. In the past, skin exams upon admission may have been given a lower priority, but that has changed.”
Dr. Meddings is concerned that the new rules could force the shifting of resources to areas where the hospital could lose money. If, when, and how many changes will actually take place is still up in the air. “Resource shifting is a concern whenever there is any sort of pay-for-performance attention directed toward one particular complication,” she says. “To balance this, many of the strategies hospitals used to prevent complications are not specific to just the diagnosis that is covered by the HAC.”
Dr. Meddings also hopes the new focus on preventable conditions will have a “halo effect” in the healthcare community. For instance, CMS mandating DVT prevention following orthopedic operations will, hopefully, result in a greater awareness of the problem in other susceptible patients.
POA Indicators
Since hospitalists often perform the initial patient history, physical, and other admission work, they are in the best position to find and document POA indicators (see Table 2, p. below). Proper notes on such things as UTIs present and the state of skin integrity are an important part of making sure the hospital is paid correctly for the care it provides.
Education on the specific definition of each potential HAC is required to help physicians avoid overtreatment of certain conditions, especially UTIs. For example, the Centers for Disease Control and Prevention (CDC) defines all UTIs as symptomatic. Therefore, the screening of all admitted patients, regardless of symptoms, is wasteful and unlikely to help the hospital’s bottom line.
“If you start screening everyone that comes through the door so you don’t miss any pre-existing UTIs, you are going to find a lot of asymptomatic colonization,” Dr. Wald says. “You are also going to spend a lot of money and time on studies and possibly treatments that may not yield many true infections. It is important that physicians know the definition of these HACs to help avoid needless interventions.”
Minimal Loss
Many hospital administrators and physicians were worried when the HAC program was first announced. Much of the stress and concern, however, seems to have dissipated. CMS estimated the HAC program would save Medicare $21 million in fiscal year 2009. Others, such as Peter McNair and colleagues writing in Health Affairs, suggest the actual impact is closer to $1.1 million.1 The CMS-projected impact of the HAC provision in fiscal-year 2009 was $21 million, out of more than $100 billion in payments.
“I think the HACs will not have a major impact because of the way payments are made,” says internist Robert Berenson, MD, a fellow at the Urban Institute in Washington, D.C., who has studied Medicare policy issues extensively, and for two years was in charge of Medicare payment policies at the Health Care Finance Administration, the precursor to CMS. “Patients who have HACs often have another comorbidity that would kick them into a higher payment category regardless of the presence of a hospital-acquired complication. In the end, it is probably more symbolic and unlikely to make a major dent in hospital income—at least at this point.”
Another limitation to CMS nonpayment for HACs is the issue of deciding which conditions are truly preventable. Dr. Berenson questions the ability of the current system to identify many additional complications for which this approach will be feasible.
“CMS has laid out its strategy, suggesting that we should be able to continue increasing the number of conditions for which providers would be paid differently based on quality,” he says. “Many observers question whether there will ever be measurement tools that are robust enough, and there will be a wide agreement on the preventability of enough conditions that this initiative will go very far.”
Although hospitalists might not face a direct financial risk, they still have their hospitals’ best interest—and their reputations—on the line. “Hospitalists care about preventing complications,” Dr. Wald says. “We are very engaged in working with our hospitals to improve care, maximize quality, and minimize cost.” TH
Kurt Ullman is a freelance medical writer based in Indiana.
Reference
- McNair PD, Luft HS, Bindman AB. Medicare’s policy not to pay for treating hospital-acquired conditions: the impact. Health Aff (Millwood). 2009;28(5):1485-1493.
TOP IMAGE SOURCE: KAREEM RIZKHALLA/ISTOCKPHOTO.COM
Hospitalist Neal Axon, MD, first became aware of an important change in his hospital’s policies last year while attending to an elderly patient the morning after admission to the community hospital where he works part time.
“This new form appeared in the chart requesting a urinalysis for my patient, who’d had a Foley catheter placed,” says Dr. Axon, an assistant professor of medicine at the Medical University of South Carolina in Charleston. “I didn’t know why, so I asked. I was told that it was now necessary to document that there was no UTI present on admission.” He asked the charge nurse, “So what do I do now that the catheter has been in place for 12 hours and has colonization without a true infection?”
The next thing he heard: silence.
The new form Dr. Axon encountered was an outgrowth of the requirements of the Deficit Reduction Act (DRA) of 2005, which ordered Medicare to withhold additional hospital payments for hospital-acquired complications (HAC) developed during a hospital stay. One result of the new rule is that much of a hospital’s response to these initiatives has been placed in the hands of the hospitalist. From accurate documentation of complications already present on admission (POA), to confirming that guidelines for treatment are being followed, to taking the lead on review of staff practices and education, hospitalists are in a position to have a wide-ranging impact on patient care and the financial health of their institutions.
Congress Pushes Reforms
In order for Medicare to not provide a reimbursement, an HAC has to be high-cost and/or high-volume, result in the assignment of the case to a higher payment when present as a secondary diagnosis, and “could reasonably be prevented through the application of evidence-based guidelines,” says Barry Straube, MD, chief medical officer and director of the Office of Clinical Standards and Quality at the Centers for Medicare and Medicaid Services (CMS). “CMS was to implement a process where we would not pay the hospitals additional money for these complications.”
The new rules mean Medicare pays hospitals on the basis of Medicare Severity Diagnostic-Related Groups (MS-DRG), which better reflect the complexity of a patient’s illness. The biggest change was a three-tiered payment schedule: a base level for the diagnosis, a second level adding money to reflect the presence of comorbidities and complications, and a third for major complications and comorbidities (see Table 1, p. 31).
“Instituting HACs means that hospitals would no longer receive the comorbidity and complication payments if the only reason a case qualified for higher payment was the HAC,” Dr. Straube explains. “We did carve out a POA exception for those conditions that were acquired outside of the hospital. HACs only impact additional payments; the hospitals are still paid for the diagnosis that resulted in the hospital admission.”
CMS also identifies three “never events” it won’t reimburse for (see “A Brief History of Never Events,” p. 35): performing the wrong procedure, performing a procedure on the wrong body part, and performing a procedure on the wrong patient. “Neither hospitals nor physicians that are involved in such egregious situations would be paid,” Dr. Straube says.
Preventability: Subject of Controversy
The big questions surrounding HACs: Could they reasonably be prevented through the application of evidence-based guidelines? How preventable are HACs? Who decides if a complication is preventable, and therefore payment for services is withheld?
They’re concerns that are widespread among physicians, hospital administrators, and regulators alike.
“The legislation required the conditions to be ‘reasonably preventable’ using established clinical guidelines,” Dr. Straube says. “We did not have to show 100% prevention. In an imperfect world, they might still take place occasionally, but with good medical care, almost all of these are preventable in this day and age.”
For CMS, the preventable conditions are an either/or situation: Either they existed prior to admission and are subject to payment, or they did not exist at admission and additional payment for the complication will not be made. “HACs do not currently consider a patient’s individual risk for complications,” says Jennifer Meddings, MD, MSc, clinical lecturer and health researcher in the Department of Internal Medicine at the University of Michigan Health System in Ann Arbor. “We know the best strategies to prevent complications in ideal patients, and these are reflected in the HACs. In real life, many of our patients just don’t fit into the guidelines for many reasons—and you have to individualize care.”
Dr. Meddings points to DVT as a prime example. For a certain number of inpatients, the guidelines can be followed to perfection. In other patients (e.g., those with kidney conditions), previous reactions to a medication or an individual’s predisposition to clotting might interfere with treatment. However, CMS doesn’t allow appeals of nonpayment decisions for HACs based on individual circumstances.
Some experts think the rigidness of the payment policy forces physicians to treat patients exactly to guidelines. Even then, payment could be declined if an HAC develops.
“One of the points of most discussion is how preventable some of these are, particularly when choosing those you are no longer going to pay for,” Dr. Meddings says. “Many of the complications currently under review have patients that are at higher risk than others. How much our prevention strategies can alleviate or reduce the risk varies widely among patients.”
Impact on HM Practice
Many of the preventable conditions outlined by CMS do not directly affect hospitalist payment. However, hospitalists often find themselves responsible for properly documenting admission and care.
“The rule changes regarding payment for HACs are only related to hospital payments, and to date, most physicians, including hospitalists, are not directly at financial risk,” says Heidi Wald, MD, MSPH, hospitalist and assistant professor of medicine in the divisions of Health Care Policy Research and General Internal Medicine at the University of Colorado Denver School of Medicine. “Although hospitalists have no financial skin in the game, there are plenty of reasons they would take an interest in addressing HACs in their hospital. In particular, they are often seen as the ‘go-to’ group for quality improvement in their hospitals.”
For example, some HM groups have been active in working with teams of physicians, nurses, and other healthcare providers to address local policies and procedures on prevention of catheter-associated urinary tract infections (UTIs) and DVT.
“This has certainly necessitated a team approach,” says Shaun Frost, MD, FHM, an SHM board member and regional medical director for Cogent Healthcare in St. Paul, Minn. “For many of the HACs that apply to our population of patients, the hospitalist alone cannot be expected to solely execute effective quality improvements. It takes a team effort in that regard, and one that includes many different disciplines.”
The Cogent-affiliated hospitalist group at Temple University Hospital in Philadelphia formed a task force to address issues with catheter-associated UTI. One initiative focused on educating all providers involved in the proper care of the catheters and similar interventions. A secondary focus of the project was an inventory of current practices and procedures.
“It was discovered that we did not have an automatic stop order for Foley catheters, so in some situations, they were likely being left in longer than needed while nursing [staff] tried to contact a physician,” Dr. Frost explains. “We created standardized order sets that include criteria for continuing the catheter. Once the criteria are no longer applicable, nursing will be able to discontinue it.”
Although CMS has only recently turned the spotlight on HACs and never events, hospitalists have been heavily involved in the patient-safety arena for years. “It is not a new phenomenon that hospitalists work for healthcare delivery and healthcare system improvement,” Dr. Meddings says.
Hospital administration at Temple University Hospital recognized the HM group’s quality-improvement (QI) work, and has “specifically charged us with spreading the work we have done in patient safety to the entire house,” Dr. Frost says. “That speaks to the administration’s opinion of the power of the HM program to assist with institution-wide QI initiatives.”
Documentation Is Key
Beyond applying proven methods to avoid HACs, hospitalists can make a difference through documentation. If the hospitalist notes all conditions when the patient first presents to the hospital, additional comorbidity and complication payments should be made.
“The part that probably has the greatest impact on the day-to-day practice of a hospitalist is the increased importance of documentation throughout the hospital episode,” Dr. Meddings says. “If complications are occurring and they are not present in the chart, the coders may not recognize that it has occurred and will not know to include it in the bill. This can have an adverse impact on the hospital and its finances.”
Documentation issues can impact hospital payment in several ways:
- Hospitals might receive additional payment by default if certain HACs are described incorrectly or without sufficient detail (e.g., receiving overpayment because the physician did not indicate a UTI was in fact a catheter-associated UTI);
- As more attention is invested in documenting all conditions POA, hospitals might be coding more comorbidities overall than previously, which also will generate additional payment for hospitals as any POA condition is eligible for increased payment; and
- Hospitals might lose payment when admitting providers fail to adequately document the condition as POA (e.g., a pre-existing decubitus ulcer not detected until the second day of the hospital stay).
The descriptions to be used in coding are very detailed. UTIs, for example, have one code to document the POA assessment, another code to show that a UTI occurred, and a third code to indicate it was catheter-associated. Each code requires appropriate documentation in the chart (see Table 1, above).
The impact hospitalists have on care and payment is not the same across the HAC spectrum. For instance, documenting the presence of pressure ulcers might be easier than distinguishing colonization from infection in those admitted with in-dwelling urinary catheters. Others, such as DVT or vascular catheter-associated infections, are rarely POA unless they are part of the admitting diagnosis.
“This new focus on hospital-acquired conditions may work to the patient’s benefit,” Dr. Meddings says. “The inclusion of pressure ulcers has led to increased attention to skin exams on admission and preventive measures during hospitalizations. In the past, skin exams upon admission may have been given a lower priority, but that has changed.”
Dr. Meddings is concerned that the new rules could force the shifting of resources to areas where the hospital could lose money. If, when, and how many changes will actually take place is still up in the air. “Resource shifting is a concern whenever there is any sort of pay-for-performance attention directed toward one particular complication,” she says. “To balance this, many of the strategies hospitals used to prevent complications are not specific to just the diagnosis that is covered by the HAC.”
Dr. Meddings also hopes the new focus on preventable conditions will have a “halo effect” in the healthcare community. For instance, CMS mandating DVT prevention following orthopedic operations will, hopefully, result in a greater awareness of the problem in other susceptible patients.
POA Indicators
Since hospitalists often perform the initial patient history, physical, and other admission work, they are in the best position to find and document POA indicators (see Table 2, p. below). Proper notes on such things as UTIs present and the state of skin integrity are an important part of making sure the hospital is paid correctly for the care it provides.
Education on the specific definition of each potential HAC is required to help physicians avoid overtreatment of certain conditions, especially UTIs. For example, the Centers for Disease Control and Prevention (CDC) defines all UTIs as symptomatic. Therefore, the screening of all admitted patients, regardless of symptoms, is wasteful and unlikely to help the hospital’s bottom line.
“If you start screening everyone that comes through the door so you don’t miss any pre-existing UTIs, you are going to find a lot of asymptomatic colonization,” Dr. Wald says. “You are also going to spend a lot of money and time on studies and possibly treatments that may not yield many true infections. It is important that physicians know the definition of these HACs to help avoid needless interventions.”
Minimal Loss
Many hospital administrators and physicians were worried when the HAC program was first announced. Much of the stress and concern, however, seems to have dissipated. CMS estimated the HAC program would save Medicare $21 million in fiscal year 2009. Others, such as Peter McNair and colleagues writing in Health Affairs, suggest the actual impact is closer to $1.1 million.1 The CMS-projected impact of the HAC provision in fiscal-year 2009 was $21 million, out of more than $100 billion in payments.
“I think the HACs will not have a major impact because of the way payments are made,” says internist Robert Berenson, MD, a fellow at the Urban Institute in Washington, D.C., who has studied Medicare policy issues extensively, and for two years was in charge of Medicare payment policies at the Health Care Finance Administration, the precursor to CMS. “Patients who have HACs often have another comorbidity that would kick them into a higher payment category regardless of the presence of a hospital-acquired complication. In the end, it is probably more symbolic and unlikely to make a major dent in hospital income—at least at this point.”
Another limitation to CMS nonpayment for HACs is the issue of deciding which conditions are truly preventable. Dr. Berenson questions the ability of the current system to identify many additional complications for which this approach will be feasible.
“CMS has laid out its strategy, suggesting that we should be able to continue increasing the number of conditions for which providers would be paid differently based on quality,” he says. “Many observers question whether there will ever be measurement tools that are robust enough, and there will be a wide agreement on the preventability of enough conditions that this initiative will go very far.”
Although hospitalists might not face a direct financial risk, they still have their hospitals’ best interest—and their reputations—on the line. “Hospitalists care about preventing complications,” Dr. Wald says. “We are very engaged in working with our hospitals to improve care, maximize quality, and minimize cost.” TH
Kurt Ullman is a freelance medical writer based in Indiana.
Reference
- McNair PD, Luft HS, Bindman AB. Medicare’s policy not to pay for treating hospital-acquired conditions: the impact. Health Aff (Millwood). 2009;28(5):1485-1493.
TOP IMAGE SOURCE: KAREEM RIZKHALLA/ISTOCKPHOTO.COM
Transitions of Care Integral to HM Patient Care
Transitions of Care Integral to HM Patient Care
I just finished my internal-medicine training and started a job as a hospitalist. We are a new hospitalist group, and I have been told that “transitions of care” is important to HM groups. I understand that getting information back to the patients’ primary-care physicians (PCPs) is important, but I am worried that I don’t have the whole picture. Is there something I am missing?
E. Parkhurst, MD
Tampa, Fla.
Dr. Hospitalist responds: Congrats on your new job. I am pleased to hear that you are motivated to learn more about transitions of care. It is important to hospitalist groups, but even more important to patients. I suspect your instincts are correct. You have an idea of what is meant by “transitions of care,” but probably do not appreciate all the nuances of the term. I certainly did not when I came out of training many years ago.
Transitions of care is a critical aspect of every patient’s care, and thus should be important to every healthcare provider. Our job is to care for the hospitalized patient and help them navigate through the complex systems of the hospital. How well we guide the patients through these transitions is reflected in their outcomes.
What is the definition of “transitions of care”? I find it useful to think about the patient’s journey when the decision is made to hospitalize the patient. When the patient is hospitalized, it is easy to recognize that the patient’s physical location is different; some, if not all, of the patient’s providers are different, too. The patient might have the same PCP caring for them in the hospital, but the nurses are different. The contrast is more evident if all of the patient’s providers are different. The ED is the point of entry for most patients. This is another location with another group of providers who do not have knowledge of all of the patient’s medical issues.
The hospital discharge is another inevitable transition. Most patients go home, but some will go to another healthcare facility (e.g., rehabilitation hospital) with another group of providers.
As you can see, the admission and discharge from the hospital involves multiple transitions. But multiple transitions also occur within the hospital. The patient could move from the general medical ward to the ICU and back; the patient might spend time in the surgical suite or operating room. Many patients go to radiology or other parts of the hospital for testing or procedures. At each location, the patient has a new group of providers.
But even if a physical location does not change, there could be a transition in care. During the day, one hospitalist or nurse might care for the patient. At night, another group of doctors and nurses are responsible for the patient’s care. Information must be transmitted and received between all of the parties at each transition in order for the appropriate care to proceed.
Effective transitions can improve provider efficiency. Think about how much easier it is to care for a patient whose care you assume when you have a clear understanding of the patient’s issues. Minimizing medical errors and increasing effective communication can reduce medical and legal risks. Effective transitions also minimize the length of hospital stay for the patient and minimize the risk of unnecessary readmission to the hospital. These can result in enhanced financial outcomes.
I think the key to effective and safe transitions of care is to create a mutually-agreed-upon process of communication and a level of expectation from all providers to carry out their role in the process. This is always easier said than done. In fact, the lack of an agreed-upon process often is a common barrier to effective transitions of care. Each participant’s role in the patient’s transitions might compete with another set of agendas.
As you can see, transitions of care is a complex topic, and I have only briefly reviewed it here. For more information, visit www.hospitalmedicine.org/boost. TH
Image Source: AMANE KANEKO
Transitions of Care Integral to HM Patient Care
I just finished my internal-medicine training and started a job as a hospitalist. We are a new hospitalist group, and I have been told that “transitions of care” is important to HM groups. I understand that getting information back to the patients’ primary-care physicians (PCPs) is important, but I am worried that I don’t have the whole picture. Is there something I am missing?
E. Parkhurst, MD
Tampa, Fla.
Dr. Hospitalist responds: Congrats on your new job. I am pleased to hear that you are motivated to learn more about transitions of care. It is important to hospitalist groups, but even more important to patients. I suspect your instincts are correct. You have an idea of what is meant by “transitions of care,” but probably do not appreciate all the nuances of the term. I certainly did not when I came out of training many years ago.
Transitions of care is a critical aspect of every patient’s care, and thus should be important to every healthcare provider. Our job is to care for the hospitalized patient and help them navigate through the complex systems of the hospital. How well we guide the patients through these transitions is reflected in their outcomes.
What is the definition of “transitions of care”? I find it useful to think about the patient’s journey when the decision is made to hospitalize the patient. When the patient is hospitalized, it is easy to recognize that the patient’s physical location is different; some, if not all, of the patient’s providers are different, too. The patient might have the same PCP caring for them in the hospital, but the nurses are different. The contrast is more evident if all of the patient’s providers are different. The ED is the point of entry for most patients. This is another location with another group of providers who do not have knowledge of all of the patient’s medical issues.
The hospital discharge is another inevitable transition. Most patients go home, but some will go to another healthcare facility (e.g., rehabilitation hospital) with another group of providers.
As you can see, the admission and discharge from the hospital involves multiple transitions. But multiple transitions also occur within the hospital. The patient could move from the general medical ward to the ICU and back; the patient might spend time in the surgical suite or operating room. Many patients go to radiology or other parts of the hospital for testing or procedures. At each location, the patient has a new group of providers.
But even if a physical location does not change, there could be a transition in care. During the day, one hospitalist or nurse might care for the patient. At night, another group of doctors and nurses are responsible for the patient’s care. Information must be transmitted and received between all of the parties at each transition in order for the appropriate care to proceed.
Effective transitions can improve provider efficiency. Think about how much easier it is to care for a patient whose care you assume when you have a clear understanding of the patient’s issues. Minimizing medical errors and increasing effective communication can reduce medical and legal risks. Effective transitions also minimize the length of hospital stay for the patient and minimize the risk of unnecessary readmission to the hospital. These can result in enhanced financial outcomes.
I think the key to effective and safe transitions of care is to create a mutually-agreed-upon process of communication and a level of expectation from all providers to carry out their role in the process. This is always easier said than done. In fact, the lack of an agreed-upon process often is a common barrier to effective transitions of care. Each participant’s role in the patient’s transitions might compete with another set of agendas.
As you can see, transitions of care is a complex topic, and I have only briefly reviewed it here. For more information, visit www.hospitalmedicine.org/boost. TH
Image Source: AMANE KANEKO
Transitions of Care Integral to HM Patient Care
I just finished my internal-medicine training and started a job as a hospitalist. We are a new hospitalist group, and I have been told that “transitions of care” is important to HM groups. I understand that getting information back to the patients’ primary-care physicians (PCPs) is important, but I am worried that I don’t have the whole picture. Is there something I am missing?
E. Parkhurst, MD
Tampa, Fla.
Dr. Hospitalist responds: Congrats on your new job. I am pleased to hear that you are motivated to learn more about transitions of care. It is important to hospitalist groups, but even more important to patients. I suspect your instincts are correct. You have an idea of what is meant by “transitions of care,” but probably do not appreciate all the nuances of the term. I certainly did not when I came out of training many years ago.
Transitions of care is a critical aspect of every patient’s care, and thus should be important to every healthcare provider. Our job is to care for the hospitalized patient and help them navigate through the complex systems of the hospital. How well we guide the patients through these transitions is reflected in their outcomes.
What is the definition of “transitions of care”? I find it useful to think about the patient’s journey when the decision is made to hospitalize the patient. When the patient is hospitalized, it is easy to recognize that the patient’s physical location is different; some, if not all, of the patient’s providers are different, too. The patient might have the same PCP caring for them in the hospital, but the nurses are different. The contrast is more evident if all of the patient’s providers are different. The ED is the point of entry for most patients. This is another location with another group of providers who do not have knowledge of all of the patient’s medical issues.
The hospital discharge is another inevitable transition. Most patients go home, but some will go to another healthcare facility (e.g., rehabilitation hospital) with another group of providers.
As you can see, the admission and discharge from the hospital involves multiple transitions. But multiple transitions also occur within the hospital. The patient could move from the general medical ward to the ICU and back; the patient might spend time in the surgical suite or operating room. Many patients go to radiology or other parts of the hospital for testing or procedures. At each location, the patient has a new group of providers.
But even if a physical location does not change, there could be a transition in care. During the day, one hospitalist or nurse might care for the patient. At night, another group of doctors and nurses are responsible for the patient’s care. Information must be transmitted and received between all of the parties at each transition in order for the appropriate care to proceed.
Effective transitions can improve provider efficiency. Think about how much easier it is to care for a patient whose care you assume when you have a clear understanding of the patient’s issues. Minimizing medical errors and increasing effective communication can reduce medical and legal risks. Effective transitions also minimize the length of hospital stay for the patient and minimize the risk of unnecessary readmission to the hospital. These can result in enhanced financial outcomes.
I think the key to effective and safe transitions of care is to create a mutually-agreed-upon process of communication and a level of expectation from all providers to carry out their role in the process. This is always easier said than done. In fact, the lack of an agreed-upon process often is a common barrier to effective transitions of care. Each participant’s role in the patient’s transitions might compete with another set of agendas.
As you can see, transitions of care is a complex topic, and I have only briefly reviewed it here. For more information, visit www.hospitalmedicine.org/boost. TH
Image Source: AMANE KANEKO
How Should Hospitalized Patients with Long QT Syndrome Be Managed?
Case
You are asked to admit a 63-year-old male with a history of hypertension and osteoarthritis. The patient, who fell at home, is scheduled for open repair of his femoral neck fracture the following day. The patient reports tripping over his granddaughter’s toys and denies any associated symptoms around the time of his fall. An electrocardiogram (ECG) reveals a QTc (QT) interval of 480 ms. How should this hospitalized patient’s prolonged QT interval be managed?
Overview
Patients with a prolonged QT interval on routine ECG present an interesting dilemma for clinicians. Although QT prolongation—either congenital or acquired—has been associated with dysrhythmias, the risk of torsades de pointes and sudden cardiac death varies considerably based on myriad underlying factors.1 Therefore, the principle job of the clinician who has recognized QT prolongation is to assess and minimize the risk of the development of clinically significant dysrhythmias, and to be prepared to manage them should they arise.
The QT interval encompasses ventricular depolarization and repolarization. This ventricular action potential proceeds through five phases. The initial upstroke (phase 0) of depolarization occurs with the opening of Na+ channels, triggering the inward Na+ current (INa), and causes the interior of the myocytes to become positively charged. This is followed by initial repolarization (phase 1) when the opening of K+ channels causes an outward K+ current (Ito). Next, the plateau phase (phase 2) of the action potential follows with a balance of inward current through Ca2+channels (Ica-L) and outward current through slow rectifier K+ channels (IKs), and then later through delayed, rapid K+ rectifier channels (IKr). Then, the inward current is deactivated, while the outward current increases through the rapid delayed rectifier (IKr) and opening of inward rectifier channels (IK1) to complete repolarization (phase 3). Finally, the action potential returns to baseline (phase 4) and Na+ begins to enter the cell again (see Figure 1, above).
The long QT syndrome (LQTS) is defined by a defect in these cardiac ion channels, which leads to abnormal repolarization, usually lengthening the QT interval and thus predisposing to ventricular dysrhythmias.2 It is estimated that as many as 85% of these syndromes are inherited, and up to 15% are acquired or sporadic.3 Depending on the underlying etiology of the LQTS, manifestations might first be appreciated at any time from in utero through adulthood.4 Symptoms including palpitations, syncope, seizures, or cardiac arrest bring these patients to medical attention.3 These symptoms frequently elicit physical or emotional stress, but they can occur without obvious inciting triggers.5 A 20% mortality risk exists in patients who are symptomatic and untreated in the first year following diagnosis, and up to 50% within 10 years following diagnosis.4
How is Long QT Syndrome Diagnosed?
The LQTS diagnosis is based on clinical history in combination with ECG abnormalities.6 Important historical elements include symptoms of palpitations, syncope, seizures, or cardiac arrest.3 In addition, a family history of unexplained syncope or sudden death, especially at a young age, should raise LQTS suspicion.5
A variety of ECG findings can be witnessed in LQTS patients.4,5 Although the majority of patients have a QTc >440 ms, approximately one-third have a QTc ≤460 ms, and about 10% have normal QTc intervals.5 Other ECG abnormalities include notched, biphasic, or prolonged T-waves, and the presence of U-waves.4,5 Schwartz et al used these elements to publish criteria (see Table 1, right) that physicians can use to assess the probability that a patient has LQTS.7
Types of Long QT Syndromes
Because the risk of developing significant dysrhythmias with LQTS is dependent on both the actual QT interval, with risk for sudden cardiac death increased two to three times with QT >440 ms compared with QT <440 ms and the specific underlying genotype, it is important to have an understanding of congenital and acquired LQTS and the associated triggers for torsades de pointes.
Congenital LQTS
Congenital LQTS is caused by mutations in cardiac ion channel proteins, primarily sodium, and potassium channels.5,6 These defects either slow depolarization or lengthen repolarization, leading to heterogeneity of repolarization of the membrane.5 This, in turn, predisposes to ventricular dysrhythmias, including torsades de pointes and subsequent ventricular fibrillation and death.2 Currently, 12 genetic defects have been identified in LQTS. Hundreds of mutations have been described to cause these defects (see Table 2, right).8 Approximately 70% of congenital LQTS are caused by mutations in three genes and are classified as LQTS 1, LQTS 2, and LQTS 3.8 The other seven mutation types account for about 5% of cases; a quarter of LQTS cases have no identified genetic mutations.8
LQTS usually can be distinguished by clinical features and some ECG characteristics, but diagnosis of the specific type requires genetic testing.8,9 The most common types of LQTS are discussed below.
- Long QT1 is the most common type, occurring in approximately 40% to 50% of patients diagnosed with LQTS. It is characterized by a defect in the potassium channel alpha subunit leading to IKs reduction.9 These patients typically present with syncope or adrenergic-induced torsades, might have wide, broad-based T-waves on ECG, and respond well to beta-blocker therapy.6 Triggers for these patients include physical exertion or emotional stressors, particularly exercise and swimming. These patients typically present in early childhood.1
- Long QT2 occurs in 35% to 40% of patients and is characterized by a different defect in the alpha subunit of the potassium channel, which leads to reduced IKr.9 ECGs in LQTS2 can demonstrate low-amplitude and notched T-waves. Sudden catecholamine surges related to emotional stress or loud noises and bradycardia can trigger dysrhythmias in Long QT2.6 Thus, beta blockers reduce overall cardiac events in LQTS2 but less effectively than in LQTS1.6 These patients also present in childhood but typically are older than patients with LQTS1.6
- Long QT3 is less common than LQTS1 or LQTS2, at 2% to 8% of LQTS patients, but carries a higher mortality and is not treated effectively with beta blockers. LQTS3 is characterized by a defect in a sodium channel, causing a gain-of-function in the INa.4,9 These patients are more likely to have a fatal dysrhythmia while sleeping, are less susceptible to exercise-induced events, and have increased morbidity and mortality associated with bradycardia.4,9 ECG frequently reveals a relatively long ST segment, followed by a peaked and tall T-wave. Beta-blocker therapy can predispose to dysrhythmias in these patients; therefore, many of these patients will have pacemakers implanted as first-line therapy.6
While less common, Jervell and Lange Nielson syndrome is an autosomal recessive form of LQTS in which affected patients have homozygous mutations in the KCNQ1 or KCNE1 genes. This syndrome occurs in approximately 1% to 7% of LQTS patients, displays a typical QTc >550 ms, can be triggered by exercise and emotional stress, is associated with deafness, and carries a high risk of cardiac events at a young age.6
Acquired Syndromes
In addition to congenital LQTS, certain patients can acquire LQTS after being treated with particular drugs or having metabolic abnormalities, namely hypomagnesemia, hypocalcemia, and hypokalemia. Most experts think patients who “acquire” LQTS that predisposes to torsades de pointes have underlying structural heart disease or LQTS genetic carrier mutations that combine with transient initiating events (e.g., drugs or metabolic abnormalities) to induce dysrhythmias.1 In addition to certain drugs, cardiac ischemia, and electrolyte abnormalities, cocaine abuse, HIV, and subarachnoid hemorrhage can induce dysrhythmias in susceptible patients.5
Many types of drugs can cause a prolonged QT interval, and others should be avoided in patients with pre-existing prolonged QT (see Table 3, p. 17). Potentially offending drugs that are frequently encountered by inpatient physicians include amiodarone, diltiazem, erythromycin, clarithromycin, ciprofloxacin, fluoxetine, paroxetine, sertraline, haloperidol, ritonavir, and methadone.1 Additionally, drugs that cause electrolyte abnormalities (e.g., diuretics and lithium) should be monitored closely.
Overall, the goals of therapy in LQTS are:
- Decrease the risk of dysrhythmic events;
- Minimize adrenergic response;
- Shorten the QTc;
- Decrease the dispersion of refractoriness; and
- Improve the function of the ion channels.3
Supportive measures should be taken for patients who are acutely symptomatic from LQTS and associated torsades de pointes. In addition to immediate cardioversion for ongoing and hemodynamically significant torsades, intravenous magnesium should be administered, electrolytes corrected, and offending drugs discontinued.5 Temporary transvenous pacing at rates of approximately 100 beats per minute is highly effective in preventing short-term recurrence of torsades in congenital and acquired LQTS, especially in bradycardic patients.5 Isoproterenol infusion increases the heart rate and effectively prevents acute recurrence of torsades in patients with acquired LQTS, but it should be used with caution in patients with structural heart disease.5
Long-term strategies to manage LQTS include:
- Minimizing the risk of triggering cardiac events via adrenergic stimulation;
- Preventing ongoing dysrhythmias;
- Avoiding medications known to prolong the QT interval; and
- Maintaining normal electrolytes and minerals.5
Most patients with congenital long QT should be discouraged from participating in competitive sports, and patients should attempt to eliminate exposures to stress or sudden awakening, though this is not practical in all cases.5 Beta blockers generally are the first-line therapy and are more effective for LQT1 than LQT2 or LQT3.4,5 If patients are still symptomatic despite adequate medical therapy, or have survived cardiac arrest, they should be considered for ICD therapy.4,5 In addition, patients with profound bradycardia benefit from pacemaker implantation.5 Patients who remain symptomatic despite both beta blockade and ICD placement might find cervicothoracic sympathectomy curative.4,5
Perioperative Considerations
Although little data is available to guide physicians in the prevention of torsades de pointes during the course of anesthesia, there are a number of considerations that may reduce the chances of symptomatic dysrhythmias.
First, care should be taken to avoid dysrhythmia triggers in LQTS by providing a calm, quiet environment during induction, monitoring, and treating metabolic abnormalities, and providing an appropriate level of anesthesia.10 Beta-blocker therapy should be continued and potentially measured preoperatively by assessing heart rate response during stress testing.5 An implantable cardioverter-defibrillator (AICD) should be interrogated prior to surgery and inactivated during the operation.5
Finally, Kies et al have recommended general anesthesia with propofol for induction (or throughout), isoflurane as the volatile agent, vecuronium for muscle relaxation, and intravenous fentanyl for analgesia when possible.10
Back to the Case
While the patient had no genetic testing for LQTS, evaluation of previous ECGs demonstrated a prolonged QT interval. The hip fracture repair was considered an urgent procedure, which precluded the ability to undertake genetic testing and consideration for device implantation. The only medication that was known to increase the risk for dysrhythmias in this patient was his diuretic, with the attendant risk of electrolyte abnormalities.
Thus, the patient’s hydrochlorothiazide was discontinued and his pre-existing atenolol continued. The patient’s electrolytes and minerals were monitored closely, and magnesium was administered on the day of surgery. Anesthesia was made aware of the prolonged QT interval, such that they were able to minimize the risk for and anticipate the treatment of dysrhythmias. The patient tolerated the surgery and post-operative period without complication and was scheduled for an outpatient workup and management for his prolonged QT interval.
Bottom Line
Long QT syndrome is frequently genetic in origin, but it can be caused by certain medications and perturbations of electrolytes. Beta blockers are the first-line therapy for the majority of LQTS cases, along with discontinuation of drugs that might induce or aggravate the QT prolongation.
Patients who have had cardiac arrest or who remain symptomatic despite beta-blocker therapy should have an ICD implanted.
In the perioperative period, patients’ electrolytes should be monitored and kept within normal limits. If the patient is on a beta blocker, it should be continued, and the anesthesiologist should be made aware of the diagnosis so that the anesthethic plan can be optimized to prevent arrhythmic complications. TH
Dr. Kamali is a medical resident at the University of Colorado Denver. Dr. Stickrath is a hospitalist at the Veterans Affairs Medical Center in Denver and an instructor of medicine at UC Denver. Dr. Prochazka is director of ambulatory care at the Denver VA and professor of medicine at UC Denver. Dr. Varosy is director of cardiac electrophysiology at the Denver VA and assistant professor of medicine at UC Denver.
References
- Kao LW, Furbee BR. Drug-induced q-T prolongation. Med Clin North Am. 2005;89(6):1125-1144.
- Marchlinski F. Chapter 226, The Tachyarrhythmias; Harrison's Principles of Internal Medicine, 17e. Available at: www.accessmedicine.com/resourceTOC .aspx?resourceID=4. Accessed Nov. 21, 2009.
- Zareba W, Cygankiewicz I. Long QT syndrome and short QT syndrome. Prog Cardiovasc Dis. 2008; 51(3):264-278.
- Booker PD, Whyte SD, Ladusans EJ. Long QT syndrome and anaesthesia. Br J Anaesth. 2003;90(3):349-366.
- Khan IA. Long QT syndrome: diagnosis and management. Am Heart J. 2002;143(1):7-14.
- Morita H, Wu J, Zipes DP. The QT syndromes: long and short. Lancet. 2008;372(9640):750-763.
- Schwartz PJ, Moss AJ, Vincent GM, Crampton RS. Diagnostic criteria for the long QT syndrome. An update. Circulation. 1993;88(2):782-784.
- Kapa S, Tester DJ, Salisbury BA, et al. Genetic testing for long-QT syndrome: distinguishing pathogenic mutations from benign variants. Circulation. 2009;120(18):1752-1760.
- Modell SM, Lehmann MH. The long QT syndrome family of cardiac ion channelopathies: a HuGE review. Genet Med. 2006;8(3):143-155.
- Kies SJ, Pabelick CM, Hurley HA, White RD, Ackerman MJ. Anesthesia for patients with congenital long QT syndrome. Anesthesiology. 2005;102(1):204-210.
- Wisely NA, Shipton EA. Long QT syndrome and anaesthesia. Eur J Anaesthesiol. 2002;19(12):853-859.
Case
You are asked to admit a 63-year-old male with a history of hypertension and osteoarthritis. The patient, who fell at home, is scheduled for open repair of his femoral neck fracture the following day. The patient reports tripping over his granddaughter’s toys and denies any associated symptoms around the time of his fall. An electrocardiogram (ECG) reveals a QTc (QT) interval of 480 ms. How should this hospitalized patient’s prolonged QT interval be managed?
Overview
Patients with a prolonged QT interval on routine ECG present an interesting dilemma for clinicians. Although QT prolongation—either congenital or acquired—has been associated with dysrhythmias, the risk of torsades de pointes and sudden cardiac death varies considerably based on myriad underlying factors.1 Therefore, the principle job of the clinician who has recognized QT prolongation is to assess and minimize the risk of the development of clinically significant dysrhythmias, and to be prepared to manage them should they arise.
The QT interval encompasses ventricular depolarization and repolarization. This ventricular action potential proceeds through five phases. The initial upstroke (phase 0) of depolarization occurs with the opening of Na+ channels, triggering the inward Na+ current (INa), and causes the interior of the myocytes to become positively charged. This is followed by initial repolarization (phase 1) when the opening of K+ channels causes an outward K+ current (Ito). Next, the plateau phase (phase 2) of the action potential follows with a balance of inward current through Ca2+channels (Ica-L) and outward current through slow rectifier K+ channels (IKs), and then later through delayed, rapid K+ rectifier channels (IKr). Then, the inward current is deactivated, while the outward current increases through the rapid delayed rectifier (IKr) and opening of inward rectifier channels (IK1) to complete repolarization (phase 3). Finally, the action potential returns to baseline (phase 4) and Na+ begins to enter the cell again (see Figure 1, above).
The long QT syndrome (LQTS) is defined by a defect in these cardiac ion channels, which leads to abnormal repolarization, usually lengthening the QT interval and thus predisposing to ventricular dysrhythmias.2 It is estimated that as many as 85% of these syndromes are inherited, and up to 15% are acquired or sporadic.3 Depending on the underlying etiology of the LQTS, manifestations might first be appreciated at any time from in utero through adulthood.4 Symptoms including palpitations, syncope, seizures, or cardiac arrest bring these patients to medical attention.3 These symptoms frequently elicit physical or emotional stress, but they can occur without obvious inciting triggers.5 A 20% mortality risk exists in patients who are symptomatic and untreated in the first year following diagnosis, and up to 50% within 10 years following diagnosis.4
How is Long QT Syndrome Diagnosed?
The LQTS diagnosis is based on clinical history in combination with ECG abnormalities.6 Important historical elements include symptoms of palpitations, syncope, seizures, or cardiac arrest.3 In addition, a family history of unexplained syncope or sudden death, especially at a young age, should raise LQTS suspicion.5
A variety of ECG findings can be witnessed in LQTS patients.4,5 Although the majority of patients have a QTc >440 ms, approximately one-third have a QTc ≤460 ms, and about 10% have normal QTc intervals.5 Other ECG abnormalities include notched, biphasic, or prolonged T-waves, and the presence of U-waves.4,5 Schwartz et al used these elements to publish criteria (see Table 1, right) that physicians can use to assess the probability that a patient has LQTS.7
Types of Long QT Syndromes
Because the risk of developing significant dysrhythmias with LQTS is dependent on both the actual QT interval, with risk for sudden cardiac death increased two to three times with QT >440 ms compared with QT <440 ms and the specific underlying genotype, it is important to have an understanding of congenital and acquired LQTS and the associated triggers for torsades de pointes.
Congenital LQTS
Congenital LQTS is caused by mutations in cardiac ion channel proteins, primarily sodium, and potassium channels.5,6 These defects either slow depolarization or lengthen repolarization, leading to heterogeneity of repolarization of the membrane.5 This, in turn, predisposes to ventricular dysrhythmias, including torsades de pointes and subsequent ventricular fibrillation and death.2 Currently, 12 genetic defects have been identified in LQTS. Hundreds of mutations have been described to cause these defects (see Table 2, right).8 Approximately 70% of congenital LQTS are caused by mutations in three genes and are classified as LQTS 1, LQTS 2, and LQTS 3.8 The other seven mutation types account for about 5% of cases; a quarter of LQTS cases have no identified genetic mutations.8
LQTS usually can be distinguished by clinical features and some ECG characteristics, but diagnosis of the specific type requires genetic testing.8,9 The most common types of LQTS are discussed below.
- Long QT1 is the most common type, occurring in approximately 40% to 50% of patients diagnosed with LQTS. It is characterized by a defect in the potassium channel alpha subunit leading to IKs reduction.9 These patients typically present with syncope or adrenergic-induced torsades, might have wide, broad-based T-waves on ECG, and respond well to beta-blocker therapy.6 Triggers for these patients include physical exertion or emotional stressors, particularly exercise and swimming. These patients typically present in early childhood.1
- Long QT2 occurs in 35% to 40% of patients and is characterized by a different defect in the alpha subunit of the potassium channel, which leads to reduced IKr.9 ECGs in LQTS2 can demonstrate low-amplitude and notched T-waves. Sudden catecholamine surges related to emotional stress or loud noises and bradycardia can trigger dysrhythmias in Long QT2.6 Thus, beta blockers reduce overall cardiac events in LQTS2 but less effectively than in LQTS1.6 These patients also present in childhood but typically are older than patients with LQTS1.6
- Long QT3 is less common than LQTS1 or LQTS2, at 2% to 8% of LQTS patients, but carries a higher mortality and is not treated effectively with beta blockers. LQTS3 is characterized by a defect in a sodium channel, causing a gain-of-function in the INa.4,9 These patients are more likely to have a fatal dysrhythmia while sleeping, are less susceptible to exercise-induced events, and have increased morbidity and mortality associated with bradycardia.4,9 ECG frequently reveals a relatively long ST segment, followed by a peaked and tall T-wave. Beta-blocker therapy can predispose to dysrhythmias in these patients; therefore, many of these patients will have pacemakers implanted as first-line therapy.6
While less common, Jervell and Lange Nielson syndrome is an autosomal recessive form of LQTS in which affected patients have homozygous mutations in the KCNQ1 or KCNE1 genes. This syndrome occurs in approximately 1% to 7% of LQTS patients, displays a typical QTc >550 ms, can be triggered by exercise and emotional stress, is associated with deafness, and carries a high risk of cardiac events at a young age.6
Acquired Syndromes
In addition to congenital LQTS, certain patients can acquire LQTS after being treated with particular drugs or having metabolic abnormalities, namely hypomagnesemia, hypocalcemia, and hypokalemia. Most experts think patients who “acquire” LQTS that predisposes to torsades de pointes have underlying structural heart disease or LQTS genetic carrier mutations that combine with transient initiating events (e.g., drugs or metabolic abnormalities) to induce dysrhythmias.1 In addition to certain drugs, cardiac ischemia, and electrolyte abnormalities, cocaine abuse, HIV, and subarachnoid hemorrhage can induce dysrhythmias in susceptible patients.5
Many types of drugs can cause a prolonged QT interval, and others should be avoided in patients with pre-existing prolonged QT (see Table 3, p. 17). Potentially offending drugs that are frequently encountered by inpatient physicians include amiodarone, diltiazem, erythromycin, clarithromycin, ciprofloxacin, fluoxetine, paroxetine, sertraline, haloperidol, ritonavir, and methadone.1 Additionally, drugs that cause electrolyte abnormalities (e.g., diuretics and lithium) should be monitored closely.
Overall, the goals of therapy in LQTS are:
- Decrease the risk of dysrhythmic events;
- Minimize adrenergic response;
- Shorten the QTc;
- Decrease the dispersion of refractoriness; and
- Improve the function of the ion channels.3
Supportive measures should be taken for patients who are acutely symptomatic from LQTS and associated torsades de pointes. In addition to immediate cardioversion for ongoing and hemodynamically significant torsades, intravenous magnesium should be administered, electrolytes corrected, and offending drugs discontinued.5 Temporary transvenous pacing at rates of approximately 100 beats per minute is highly effective in preventing short-term recurrence of torsades in congenital and acquired LQTS, especially in bradycardic patients.5 Isoproterenol infusion increases the heart rate and effectively prevents acute recurrence of torsades in patients with acquired LQTS, but it should be used with caution in patients with structural heart disease.5
Long-term strategies to manage LQTS include:
- Minimizing the risk of triggering cardiac events via adrenergic stimulation;
- Preventing ongoing dysrhythmias;
- Avoiding medications known to prolong the QT interval; and
- Maintaining normal electrolytes and minerals.5
Most patients with congenital long QT should be discouraged from participating in competitive sports, and patients should attempt to eliminate exposures to stress or sudden awakening, though this is not practical in all cases.5 Beta blockers generally are the first-line therapy and are more effective for LQT1 than LQT2 or LQT3.4,5 If patients are still symptomatic despite adequate medical therapy, or have survived cardiac arrest, they should be considered for ICD therapy.4,5 In addition, patients with profound bradycardia benefit from pacemaker implantation.5 Patients who remain symptomatic despite both beta blockade and ICD placement might find cervicothoracic sympathectomy curative.4,5
Perioperative Considerations
Although little data is available to guide physicians in the prevention of torsades de pointes during the course of anesthesia, there are a number of considerations that may reduce the chances of symptomatic dysrhythmias.
First, care should be taken to avoid dysrhythmia triggers in LQTS by providing a calm, quiet environment during induction, monitoring, and treating metabolic abnormalities, and providing an appropriate level of anesthesia.10 Beta-blocker therapy should be continued and potentially measured preoperatively by assessing heart rate response during stress testing.5 An implantable cardioverter-defibrillator (AICD) should be interrogated prior to surgery and inactivated during the operation.5
Finally, Kies et al have recommended general anesthesia with propofol for induction (or throughout), isoflurane as the volatile agent, vecuronium for muscle relaxation, and intravenous fentanyl for analgesia when possible.10
Back to the Case
While the patient had no genetic testing for LQTS, evaluation of previous ECGs demonstrated a prolonged QT interval. The hip fracture repair was considered an urgent procedure, which precluded the ability to undertake genetic testing and consideration for device implantation. The only medication that was known to increase the risk for dysrhythmias in this patient was his diuretic, with the attendant risk of electrolyte abnormalities.
Thus, the patient’s hydrochlorothiazide was discontinued and his pre-existing atenolol continued. The patient’s electrolytes and minerals were monitored closely, and magnesium was administered on the day of surgery. Anesthesia was made aware of the prolonged QT interval, such that they were able to minimize the risk for and anticipate the treatment of dysrhythmias. The patient tolerated the surgery and post-operative period without complication and was scheduled for an outpatient workup and management for his prolonged QT interval.
Bottom Line
Long QT syndrome is frequently genetic in origin, but it can be caused by certain medications and perturbations of electrolytes. Beta blockers are the first-line therapy for the majority of LQTS cases, along with discontinuation of drugs that might induce or aggravate the QT prolongation.
Patients who have had cardiac arrest or who remain symptomatic despite beta-blocker therapy should have an ICD implanted.
In the perioperative period, patients’ electrolytes should be monitored and kept within normal limits. If the patient is on a beta blocker, it should be continued, and the anesthesiologist should be made aware of the diagnosis so that the anesthethic plan can be optimized to prevent arrhythmic complications. TH
Dr. Kamali is a medical resident at the University of Colorado Denver. Dr. Stickrath is a hospitalist at the Veterans Affairs Medical Center in Denver and an instructor of medicine at UC Denver. Dr. Prochazka is director of ambulatory care at the Denver VA and professor of medicine at UC Denver. Dr. Varosy is director of cardiac electrophysiology at the Denver VA and assistant professor of medicine at UC Denver.
References
- Kao LW, Furbee BR. Drug-induced q-T prolongation. Med Clin North Am. 2005;89(6):1125-1144.
- Marchlinski F. Chapter 226, The Tachyarrhythmias; Harrison's Principles of Internal Medicine, 17e. Available at: www.accessmedicine.com/resourceTOC .aspx?resourceID=4. Accessed Nov. 21, 2009.
- Zareba W, Cygankiewicz I. Long QT syndrome and short QT syndrome. Prog Cardiovasc Dis. 2008; 51(3):264-278.
- Booker PD, Whyte SD, Ladusans EJ. Long QT syndrome and anaesthesia. Br J Anaesth. 2003;90(3):349-366.
- Khan IA. Long QT syndrome: diagnosis and management. Am Heart J. 2002;143(1):7-14.
- Morita H, Wu J, Zipes DP. The QT syndromes: long and short. Lancet. 2008;372(9640):750-763.
- Schwartz PJ, Moss AJ, Vincent GM, Crampton RS. Diagnostic criteria for the long QT syndrome. An update. Circulation. 1993;88(2):782-784.
- Kapa S, Tester DJ, Salisbury BA, et al. Genetic testing for long-QT syndrome: distinguishing pathogenic mutations from benign variants. Circulation. 2009;120(18):1752-1760.
- Modell SM, Lehmann MH. The long QT syndrome family of cardiac ion channelopathies: a HuGE review. Genet Med. 2006;8(3):143-155.
- Kies SJ, Pabelick CM, Hurley HA, White RD, Ackerman MJ. Anesthesia for patients with congenital long QT syndrome. Anesthesiology. 2005;102(1):204-210.
- Wisely NA, Shipton EA. Long QT syndrome and anaesthesia. Eur J Anaesthesiol. 2002;19(12):853-859.
Case
You are asked to admit a 63-year-old male with a history of hypertension and osteoarthritis. The patient, who fell at home, is scheduled for open repair of his femoral neck fracture the following day. The patient reports tripping over his granddaughter’s toys and denies any associated symptoms around the time of his fall. An electrocardiogram (ECG) reveals a QTc (QT) interval of 480 ms. How should this hospitalized patient’s prolonged QT interval be managed?
Overview
Patients with a prolonged QT interval on routine ECG present an interesting dilemma for clinicians. Although QT prolongation—either congenital or acquired—has been associated with dysrhythmias, the risk of torsades de pointes and sudden cardiac death varies considerably based on myriad underlying factors.1 Therefore, the principle job of the clinician who has recognized QT prolongation is to assess and minimize the risk of the development of clinically significant dysrhythmias, and to be prepared to manage them should they arise.
The QT interval encompasses ventricular depolarization and repolarization. This ventricular action potential proceeds through five phases. The initial upstroke (phase 0) of depolarization occurs with the opening of Na+ channels, triggering the inward Na+ current (INa), and causes the interior of the myocytes to become positively charged. This is followed by initial repolarization (phase 1) when the opening of K+ channels causes an outward K+ current (Ito). Next, the plateau phase (phase 2) of the action potential follows with a balance of inward current through Ca2+channels (Ica-L) and outward current through slow rectifier K+ channels (IKs), and then later through delayed, rapid K+ rectifier channels (IKr). Then, the inward current is deactivated, while the outward current increases through the rapid delayed rectifier (IKr) and opening of inward rectifier channels (IK1) to complete repolarization (phase 3). Finally, the action potential returns to baseline (phase 4) and Na+ begins to enter the cell again (see Figure 1, above).
The long QT syndrome (LQTS) is defined by a defect in these cardiac ion channels, which leads to abnormal repolarization, usually lengthening the QT interval and thus predisposing to ventricular dysrhythmias.2 It is estimated that as many as 85% of these syndromes are inherited, and up to 15% are acquired or sporadic.3 Depending on the underlying etiology of the LQTS, manifestations might first be appreciated at any time from in utero through adulthood.4 Symptoms including palpitations, syncope, seizures, or cardiac arrest bring these patients to medical attention.3 These symptoms frequently elicit physical or emotional stress, but they can occur without obvious inciting triggers.5 A 20% mortality risk exists in patients who are symptomatic and untreated in the first year following diagnosis, and up to 50% within 10 years following diagnosis.4
How is Long QT Syndrome Diagnosed?
The LQTS diagnosis is based on clinical history in combination with ECG abnormalities.6 Important historical elements include symptoms of palpitations, syncope, seizures, or cardiac arrest.3 In addition, a family history of unexplained syncope or sudden death, especially at a young age, should raise LQTS suspicion.5
A variety of ECG findings can be witnessed in LQTS patients.4,5 Although the majority of patients have a QTc >440 ms, approximately one-third have a QTc ≤460 ms, and about 10% have normal QTc intervals.5 Other ECG abnormalities include notched, biphasic, or prolonged T-waves, and the presence of U-waves.4,5 Schwartz et al used these elements to publish criteria (see Table 1, right) that physicians can use to assess the probability that a patient has LQTS.7
Types of Long QT Syndromes
Because the risk of developing significant dysrhythmias with LQTS is dependent on both the actual QT interval, with risk for sudden cardiac death increased two to three times with QT >440 ms compared with QT <440 ms and the specific underlying genotype, it is important to have an understanding of congenital and acquired LQTS and the associated triggers for torsades de pointes.
Congenital LQTS
Congenital LQTS is caused by mutations in cardiac ion channel proteins, primarily sodium, and potassium channels.5,6 These defects either slow depolarization or lengthen repolarization, leading to heterogeneity of repolarization of the membrane.5 This, in turn, predisposes to ventricular dysrhythmias, including torsades de pointes and subsequent ventricular fibrillation and death.2 Currently, 12 genetic defects have been identified in LQTS. Hundreds of mutations have been described to cause these defects (see Table 2, right).8 Approximately 70% of congenital LQTS are caused by mutations in three genes and are classified as LQTS 1, LQTS 2, and LQTS 3.8 The other seven mutation types account for about 5% of cases; a quarter of LQTS cases have no identified genetic mutations.8
LQTS usually can be distinguished by clinical features and some ECG characteristics, but diagnosis of the specific type requires genetic testing.8,9 The most common types of LQTS are discussed below.
- Long QT1 is the most common type, occurring in approximately 40% to 50% of patients diagnosed with LQTS. It is characterized by a defect in the potassium channel alpha subunit leading to IKs reduction.9 These patients typically present with syncope or adrenergic-induced torsades, might have wide, broad-based T-waves on ECG, and respond well to beta-blocker therapy.6 Triggers for these patients include physical exertion or emotional stressors, particularly exercise and swimming. These patients typically present in early childhood.1
- Long QT2 occurs in 35% to 40% of patients and is characterized by a different defect in the alpha subunit of the potassium channel, which leads to reduced IKr.9 ECGs in LQTS2 can demonstrate low-amplitude and notched T-waves. Sudden catecholamine surges related to emotional stress or loud noises and bradycardia can trigger dysrhythmias in Long QT2.6 Thus, beta blockers reduce overall cardiac events in LQTS2 but less effectively than in LQTS1.6 These patients also present in childhood but typically are older than patients with LQTS1.6
- Long QT3 is less common than LQTS1 or LQTS2, at 2% to 8% of LQTS patients, but carries a higher mortality and is not treated effectively with beta blockers. LQTS3 is characterized by a defect in a sodium channel, causing a gain-of-function in the INa.4,9 These patients are more likely to have a fatal dysrhythmia while sleeping, are less susceptible to exercise-induced events, and have increased morbidity and mortality associated with bradycardia.4,9 ECG frequently reveals a relatively long ST segment, followed by a peaked and tall T-wave. Beta-blocker therapy can predispose to dysrhythmias in these patients; therefore, many of these patients will have pacemakers implanted as first-line therapy.6
While less common, Jervell and Lange Nielson syndrome is an autosomal recessive form of LQTS in which affected patients have homozygous mutations in the KCNQ1 or KCNE1 genes. This syndrome occurs in approximately 1% to 7% of LQTS patients, displays a typical QTc >550 ms, can be triggered by exercise and emotional stress, is associated with deafness, and carries a high risk of cardiac events at a young age.6
Acquired Syndromes
In addition to congenital LQTS, certain patients can acquire LQTS after being treated with particular drugs or having metabolic abnormalities, namely hypomagnesemia, hypocalcemia, and hypokalemia. Most experts think patients who “acquire” LQTS that predisposes to torsades de pointes have underlying structural heart disease or LQTS genetic carrier mutations that combine with transient initiating events (e.g., drugs or metabolic abnormalities) to induce dysrhythmias.1 In addition to certain drugs, cardiac ischemia, and electrolyte abnormalities, cocaine abuse, HIV, and subarachnoid hemorrhage can induce dysrhythmias in susceptible patients.5
Many types of drugs can cause a prolonged QT interval, and others should be avoided in patients with pre-existing prolonged QT (see Table 3, p. 17). Potentially offending drugs that are frequently encountered by inpatient physicians include amiodarone, diltiazem, erythromycin, clarithromycin, ciprofloxacin, fluoxetine, paroxetine, sertraline, haloperidol, ritonavir, and methadone.1 Additionally, drugs that cause electrolyte abnormalities (e.g., diuretics and lithium) should be monitored closely.
Overall, the goals of therapy in LQTS are:
- Decrease the risk of dysrhythmic events;
- Minimize adrenergic response;
- Shorten the QTc;
- Decrease the dispersion of refractoriness; and
- Improve the function of the ion channels.3
Supportive measures should be taken for patients who are acutely symptomatic from LQTS and associated torsades de pointes. In addition to immediate cardioversion for ongoing and hemodynamically significant torsades, intravenous magnesium should be administered, electrolytes corrected, and offending drugs discontinued.5 Temporary transvenous pacing at rates of approximately 100 beats per minute is highly effective in preventing short-term recurrence of torsades in congenital and acquired LQTS, especially in bradycardic patients.5 Isoproterenol infusion increases the heart rate and effectively prevents acute recurrence of torsades in patients with acquired LQTS, but it should be used with caution in patients with structural heart disease.5
Long-term strategies to manage LQTS include:
- Minimizing the risk of triggering cardiac events via adrenergic stimulation;
- Preventing ongoing dysrhythmias;
- Avoiding medications known to prolong the QT interval; and
- Maintaining normal electrolytes and minerals.5
Most patients with congenital long QT should be discouraged from participating in competitive sports, and patients should attempt to eliminate exposures to stress or sudden awakening, though this is not practical in all cases.5 Beta blockers generally are the first-line therapy and are more effective for LQT1 than LQT2 or LQT3.4,5 If patients are still symptomatic despite adequate medical therapy, or have survived cardiac arrest, they should be considered for ICD therapy.4,5 In addition, patients with profound bradycardia benefit from pacemaker implantation.5 Patients who remain symptomatic despite both beta blockade and ICD placement might find cervicothoracic sympathectomy curative.4,5
Perioperative Considerations
Although little data is available to guide physicians in the prevention of torsades de pointes during the course of anesthesia, there are a number of considerations that may reduce the chances of symptomatic dysrhythmias.
First, care should be taken to avoid dysrhythmia triggers in LQTS by providing a calm, quiet environment during induction, monitoring, and treating metabolic abnormalities, and providing an appropriate level of anesthesia.10 Beta-blocker therapy should be continued and potentially measured preoperatively by assessing heart rate response during stress testing.5 An implantable cardioverter-defibrillator (AICD) should be interrogated prior to surgery and inactivated during the operation.5
Finally, Kies et al have recommended general anesthesia with propofol for induction (or throughout), isoflurane as the volatile agent, vecuronium for muscle relaxation, and intravenous fentanyl for analgesia when possible.10
Back to the Case
While the patient had no genetic testing for LQTS, evaluation of previous ECGs demonstrated a prolonged QT interval. The hip fracture repair was considered an urgent procedure, which precluded the ability to undertake genetic testing and consideration for device implantation. The only medication that was known to increase the risk for dysrhythmias in this patient was his diuretic, with the attendant risk of electrolyte abnormalities.
Thus, the patient’s hydrochlorothiazide was discontinued and his pre-existing atenolol continued. The patient’s electrolytes and minerals were monitored closely, and magnesium was administered on the day of surgery. Anesthesia was made aware of the prolonged QT interval, such that they were able to minimize the risk for and anticipate the treatment of dysrhythmias. The patient tolerated the surgery and post-operative period without complication and was scheduled for an outpatient workup and management for his prolonged QT interval.
Bottom Line
Long QT syndrome is frequently genetic in origin, but it can be caused by certain medications and perturbations of electrolytes. Beta blockers are the first-line therapy for the majority of LQTS cases, along with discontinuation of drugs that might induce or aggravate the QT prolongation.
Patients who have had cardiac arrest or who remain symptomatic despite beta-blocker therapy should have an ICD implanted.
In the perioperative period, patients’ electrolytes should be monitored and kept within normal limits. If the patient is on a beta blocker, it should be continued, and the anesthesiologist should be made aware of the diagnosis so that the anesthethic plan can be optimized to prevent arrhythmic complications. TH
Dr. Kamali is a medical resident at the University of Colorado Denver. Dr. Stickrath is a hospitalist at the Veterans Affairs Medical Center in Denver and an instructor of medicine at UC Denver. Dr. Prochazka is director of ambulatory care at the Denver VA and professor of medicine at UC Denver. Dr. Varosy is director of cardiac electrophysiology at the Denver VA and assistant professor of medicine at UC Denver.
References
- Kao LW, Furbee BR. Drug-induced q-T prolongation. Med Clin North Am. 2005;89(6):1125-1144.
- Marchlinski F. Chapter 226, The Tachyarrhythmias; Harrison's Principles of Internal Medicine, 17e. Available at: www.accessmedicine.com/resourceTOC .aspx?resourceID=4. Accessed Nov. 21, 2009.
- Zareba W, Cygankiewicz I. Long QT syndrome and short QT syndrome. Prog Cardiovasc Dis. 2008; 51(3):264-278.
- Booker PD, Whyte SD, Ladusans EJ. Long QT syndrome and anaesthesia. Br J Anaesth. 2003;90(3):349-366.
- Khan IA. Long QT syndrome: diagnosis and management. Am Heart J. 2002;143(1):7-14.
- Morita H, Wu J, Zipes DP. The QT syndromes: long and short. Lancet. 2008;372(9640):750-763.
- Schwartz PJ, Moss AJ, Vincent GM, Crampton RS. Diagnostic criteria for the long QT syndrome. An update. Circulation. 1993;88(2):782-784.
- Kapa S, Tester DJ, Salisbury BA, et al. Genetic testing for long-QT syndrome: distinguishing pathogenic mutations from benign variants. Circulation. 2009;120(18):1752-1760.
- Modell SM, Lehmann MH. The long QT syndrome family of cardiac ion channelopathies: a HuGE review. Genet Med. 2006;8(3):143-155.
- Kies SJ, Pabelick CM, Hurley HA, White RD, Ackerman MJ. Anesthesia for patients with congenital long QT syndrome. Anesthesiology. 2005;102(1):204-210.
- Wisely NA, Shipton EA. Long QT syndrome and anaesthesia. Eur J Anaesthesiol. 2002;19(12):853-859.
Step-by-Step Medicine
Evidence-based practice guidelines are key tools to help hospitalists practice high-quality medicine and demonstrate the value of their inpatient care model. Guidelines are designed to produce superior care outcomes and resource utilization efficiencies by encouraging proven medical practices and discouraging ineffective or unproven ones. Yet inefficiencies, variation, and quality gaps persist in medical care—much to the chagrin of policymakers.
Is the answer more guidelines, and better implementation of existing ones?
Research experts and many HM leaders say yes.
In fact, HM is leading the way in an important new area for which there is little uniform guidance: optimal care transitions during patient handoffs. Care transitions are a pivotal time in the patient care process and are replete with avoidable service duplication, poor communication among providers, gaps in care reconciliation, and patient-safety issues.
SHM has joined five other organizations in issuing a Transitions of Care Consensus Policy Statement, which promises more systematic, safe, and efficient patient handoffs.1 SHM also is targeting care-transition improvement in a variety of other venues, all of which can help hospitalists demonstrate more persuasively the value they bring to healthcare delivery.
Guidelines Work
Practice guidelines work, in the sense that they help providers practice in ways consistent with what the best aggregate knowledge and expert opinion says is most effective. The evidence allows physicians to avoid expending scarce resources on ineffective clinical services. Their importance is magnified by the current urgency given to value-based purchasing in healthcare reform. “The right care, for the right patient, at the right time” is the new mantra of payors and policymakers, many of whom are demanding the best and most efficient healthcare delivery at the lowest cost.

“When providers are not providing the right care at the right time to patients, we find that the patient often gets more services … that they didn’t need. That oftentimes exposes them to potential harm and (services) that are wasteful of resources,” says Janet M. Corrigan, PhD, MBA, president and CEO of the National Quality Forum (NQF), a standard-setting organization that convenes national experts to apply “gold standard” endorsement of guidelines developed by professional medical societies and other entities. “Guidelines are a way of synthesizing evidence and translating it into action steps that providers can follow so that they get the best results that we know how to get for their patients.”
Clinicians and healthcare organizations have several sources for guidelines. The Agency for Healthcare Research and Quality (AHRQ) systematically reviews and vets guidelines submitted for inclusion in its National Guideline Clearinghouse (www.guideline.gov), and makes them available for evidence-based clinical decision-making, says Jean Slutsky, director of AHRQ’s Center for Outcomes and Evidence. AHRQ also offers public access to the National Quality Measure Clearinghouse and the Health Care Innovations Exchange, repositories of searchable quality measures and tools relevant to an array of diseases and conditions.
The Institute for Healthcare Improvement (IHI), an independent nonprofit organization, helps frontline physicians implement guidelines, and also helps provider teams decide which guidelines are most appropriate to achieve their desired outcomes, according to Amy E. Boutwell, MD, MPP, IHI’s director of health policy strategy.
Hospitalists use an array of disease-specific practice guidelines from different specialty societies for diagnoses they frequently encounter, such as chest pain, stroke, pneumonia, myocardial infarction, gastrointestinal bleeding, asthma, and chronic obstructive pulmonary disease (COPD). “Most hospitalists want to keep up with the best available evidence,” says Patrick J. Torcson, MD, MMM, FACP, director of hospital medicine at St. Tammany Parish Hospital in Covington, La., and chair of SHM’s Performance and Standards Committee. “The recently updated American College of Cardiology (ACC) and American Heart Association (AHA) guidelines on heart failure are exceptional. The American College of Chest Physicians has an extremely comprehensive set of guidelines on thrombosis, which is the bible for handling anticoagulation.”
Studies are published every month demonstrating that physicians who implement national guidelines at the local level improve such patient outcomes as mortality, length of stay, and time to clinical stability. Dr. Corrigan notes that there are abundant examples of guideline adherence boosting quality outcomes, and cites as a prime example the AHA’s Get With the Guidelines program (www.american heart.org/presenter.jhtml?identifier=1165), which has documented quality gains in coronary artery disease, stroke, and heart failure patients.

—William T. Ford, MD, FHM, section chief of hospital medicine, Temple University Hospital, Philadelphia
Barriers to Acceptance and Adherence
But there are obstacles to guideline adherence, and widespread practice pattern variation remains a huge national problem. Providers in some regions of the country can use twice the resources as their counterparts in other regions and bring no additional benefit to patients (see “Medicare Fee Inspection,” p. 30). The Dartmouth Atlas of Health Care says unwarranted practice variation is responsible for as much as 30% of wasted healthcare spending in the U.S.—a cost that reformers are anxious to eliminate.
The traditional culture of autonomy in the medical profession is perhaps the most difficult and enduring barrier to reducing unjustified practice variation: clinicians don’t automatically follow guidelines, many treat them more as options than as true standards, and organizations do not sufficiently enforce or reward adherence to guidelines, wrote researchers in a special 2005 issue of Health Affairs focusing on guidelines.2
“In an age of mandated cost control and resource limitation under managed care,” the researchers wrote, some physicians still regard practice guidelines as “cookbook medicine” that threatens the use of clinical judgment and encourages treating patients as essentially interchangeable. In the face of that perceived threat, the researchers added, many physicians continue to uphold a traditional view of medicine as an art “in which individual expertise and technique are allowed to shine through and ultimately result in a higher standard of patient care.”
Dr. Corrigan acknowledges the significant obstacles to successful practice guideline implementation:
- Guidelines are developed by various sources, particularly specialty societies, who do not always coordinate their activities. Physicians are left with overlapping and sometimes contradictory guidelines for managing the same disease or condition.
- Guidelines must be maintained and kept current, or physicians will lose confidence and not follow them.
- Guidelines are of varying quality. Some provide clear clinical direction; others are not written in a way that physicians can clearly translate into clinical practice.
- There are significant gaps in the evidence basis for guideline development. Much more comparative effectiveness research needs to be conducted to develop more valid and meaningful guidelines.
- Guidelines must be communicated effectively to physicians, making them available and convenient at the point of clinical care. Electronic health records with user-friendly decision support functions show great promise in “making the right thing the easy thing to do.”
- The fee-for-service payment system encourages greater volume of services, irrespective of guideline recommendations.
Physicians also recognize inherent limitations of guidelines. “Guidelines typically apply across populations. Adding levels of clinical complexity gets further away from a guideline’s applicability. Many physicians will tell you that the patient in front of them is a special case requiring a modification of the protocol,” Dr. Boutwell explains. For example, diabetic management guidelines are based on what is best for a population of diabetics, versus what is best for said hospitalist’s patient who has eight co-morbidities, one of which is diabetes, Boutwell notes. “Guidelines come disease-specific. Patients don’t,” she adds.
Nevertheless, Dr. Boutwell notes, there are robust guidelines and the IHI tries to help front-line physicians and care teams to implement them reliably and effectively.
An obstacle that inhibits hospitalists from implementing guidelines in an optimal fashion “is that we’re not one specialty—we deal with it all—and that complexity can be overwhelming. There is no central repository where all of the guidelines can be found in one place,” according to William T. Ford, MD, FHM, program medical director for Cogent Healthcare and section chief of hospital medicine for Temple University Hospital in Philadelphia.
Make Guidelines Work
Researchers say guidelines are most successful when they are well-supported and uncomplicated, backed by strong leadership and sufficient resources, and are used as “rallying points” to stimulate interdependent and collaborative care among physicians, nurses, pharmacists, equipment suppliers, administrators, and patients.
“Guidelines are really the foundation for determining best practices,” Dr. Torcson says. “There is no shortage of excellent guidelines, or proof that specific interventions do improve outcomes. The key is achieving more uniform implementation. We need tools like pre-printed orders in electronic health records (EHR) to effectively integrate these guidelines into hospitalists’ practice.”
More widespread EHR adoption with user-friendly medical decision-support systems will play a huge role in boosting guideline adoption and effectiveness, says Mary Nix, MS, MT(ASCP)SBB, health science administrator at AHRQ and project officer for the agency’s Center for Outcomes and Evidence.
Dr. Ford says HM groups must evaluate the top 10 to 15 diagnosis-related groups (DRGs) that they see each day (e.g., congestive heart failure, acute kidney failure, pneumonia, cellulitis, or acute coronary syndrome) and come to consensus on which guidelines best address them.
HM groups must then secure buy-in to those guidelines from everyone in the group; from the subspecialists they work with; and from their hospital’s chief medical, financial, and utilization officers.
Care-Transition Guidelines: Opportunity for Hospitalists
A particularly important HM opportunity is improving care transitions. Deficits in communication and information transfer between hospital-based and primary-care physicians (PCPs) are “substantial and ubiquitous,” while delays and omissions are consistently large, and traditional methods of completing and delivering discharge summaries are “suboptimal for communicating timely, accurate, and medically important data to the physicians who will be responsible for follow-up care,” according to a hospitalist-authored Feb. 28, 2007, article in the Journal of the American Medical Association.3 PCPs routinely are not notified about patient admissions or complications during the hospital stay, and some PCPs fail to provide sufficient information to hospitalists at admission, fail to visit or call hospitalized patients, or fail to participate in discharge planning, the study’s researchers noted. For patients with chronic illnesses and frequent hospitalization, those deficits are multiplied, making completeness of information handoffs particularly important.
Because patient handoffs have notoriously been fraught with miscommunication and poor information exchange between providers, adopting a professional consensus on what constitutes the best, safest, and most effective activities during these handoffs is sorely needed.
“Care-transition guidelines can have tremendous power because they affect every hospital patient—each of whom experiences care transitions,” says Rusty Holman, MD, FHM, chief operating officer of Brentwood, Tenn.-based Cogent Healthcare and past president of SHM. “It is an area undergoing rapid development, evolution, and discovery, and hospitalists have positioned themselves as leaders and owners of this particular scenario.”
As care-transition guidelines emerge and mature, Holman thinks they eventually will be tied to value-based healthcare purchasing programs that affect hospitalists’ reimbursement equations and further boost incentives to follow those guidelines. A prime example: Medicare calculated it could save $12 billion annually by reducing preventable 30-day hospital readmissions and will soon stop paying for them. Perhaps 3% to 5% of a hospital’s DRG reimbursement will be at risk under Medicare’s proposal, Dr. Torcson notes.
“Hospitals are going to be much more motivated to build systems and engage physicians, especially hospitalists, to lower readmission rates. Hospitalists will be focusing more and more on how care-transition process improvements can lower those rates,” Dr. Holman says. “That’s a huge opportunity for hospitalists to make a business case for the value they bring to their institutions, and will further justify the financial support they already receive.”
Dr. Ford is more cautious in his appraisal of the financial rewards of better guideline implementation. “We do not capture that much revenue per patient, and even a length-of-stay reduction is difficult for a hospital’s CFO to extrapolate how much money hospitalists save,” he says. “I don’t think hospitalists will be paid more, even if they save the hospital money. You’re just doing your job, but you’re going to keep your job, and you’ll have an enormous bargaining chip when renegotiating contracts with hospitals.”
Still, a prevented readmission might mean a bed for a revenue-generating elective surgery, something that adds to the reward equation.

—Patrick Torcson, MD, MMM, FACP, director of hospital medicine, St. Tammany Parish Hospital, Covington, La., SHM Performance and Standards Committee chair
Transition Evolution
SHM and other sources offer physicians and hospitals expert assistance in implementing care-transition guidelines (see “Care-Transition Guidance,” p. 7). The transitions-of-care policy statement jointly issued by the SHM and five other specialty societies further demonstrates that hospitalists play a key leadership role on this front.1
The policy statement emerged from a multi-stakeholder consensus conference convened by SHM, the American College of Physicians (ACP), and the Society of General Internal Medicine, which was attended by more than 30 medical specialty societies, governmental agencies, and performance measure developers. Participants focused on what standard pieces of information should be exchanged among providers during inpatient to outpatient transitions, and they issued a set of standards for improving those transitions (see “Managing Transitions in Care Between the Inpatient and Outpatient Settings,” p. 7).
“This consensus statement has enormous significance,” Dr. Ford says. “We’re finally shedding light on how to tackle patient handoff and hospital readmission issues, and we as a specialty have to take on care-transition improvement as our mantra. If we were to solve just that one piece, we can more easily start implementing other clinical guidelines. Care-transition guidelines are a fundamental tool to build consensus within your own group and with other clinicians in a team approach.”
Dr. Corrigan applauds the physician groups for publishing the transitions-of-care statement and encourages the societies to work together to “take it to the next step, which is to develop the measures and get them endorsed through the NQF process.”
SHM members are participating in workgroups convened by the NQF to identify standardized performance measures and to develop action plans over the next few months for several national priority areas—one of which is care coordination. “We have a ways to go to achieve better patient handoffs and information exchange between hospitals and other settings in the community. Hospitalists can drive the development of those guidelines and protocols,” Dr. Corrigan says. TH
Christopher Guadagnino, PhD, is a freelance medical writer based in Pennsylvania.
References
- Kripalani S, LeFevre F, Phillips CO, Williams MV; Basaviah P, Baker DW. Deficits in communication and information transfer between hospital-based and primary care physicians: implications for patient safety and continuity of care. JAMA. 2007;297:831-841.
- Timmermans S, Mauck A. The promises and pitfalls of evidence-based medicine. Health Affairs. 2005; 24(1):18-28.
- Snow V, Beck D, Budnitz T, et al. Transitions of care consensus policy statement: American College of Physicians, Society of General Internal Medicine, Society of Hospital Medicine, American Geriatrics Society, American College of Emergency Physicians, Society of Academic Emergency Medicine. J Hosp Med. 2009: 4(6)364-370.
Top Image Source: GOLDEN PIXELS LLC/ALAMY
Evidence-based practice guidelines are key tools to help hospitalists practice high-quality medicine and demonstrate the value of their inpatient care model. Guidelines are designed to produce superior care outcomes and resource utilization efficiencies by encouraging proven medical practices and discouraging ineffective or unproven ones. Yet inefficiencies, variation, and quality gaps persist in medical care—much to the chagrin of policymakers.
Is the answer more guidelines, and better implementation of existing ones?
Research experts and many HM leaders say yes.
In fact, HM is leading the way in an important new area for which there is little uniform guidance: optimal care transitions during patient handoffs. Care transitions are a pivotal time in the patient care process and are replete with avoidable service duplication, poor communication among providers, gaps in care reconciliation, and patient-safety issues.
SHM has joined five other organizations in issuing a Transitions of Care Consensus Policy Statement, which promises more systematic, safe, and efficient patient handoffs.1 SHM also is targeting care-transition improvement in a variety of other venues, all of which can help hospitalists demonstrate more persuasively the value they bring to healthcare delivery.
Guidelines Work
Practice guidelines work, in the sense that they help providers practice in ways consistent with what the best aggregate knowledge and expert opinion says is most effective. The evidence allows physicians to avoid expending scarce resources on ineffective clinical services. Their importance is magnified by the current urgency given to value-based purchasing in healthcare reform. “The right care, for the right patient, at the right time” is the new mantra of payors and policymakers, many of whom are demanding the best and most efficient healthcare delivery at the lowest cost.

“When providers are not providing the right care at the right time to patients, we find that the patient often gets more services … that they didn’t need. That oftentimes exposes them to potential harm and (services) that are wasteful of resources,” says Janet M. Corrigan, PhD, MBA, president and CEO of the National Quality Forum (NQF), a standard-setting organization that convenes national experts to apply “gold standard” endorsement of guidelines developed by professional medical societies and other entities. “Guidelines are a way of synthesizing evidence and translating it into action steps that providers can follow so that they get the best results that we know how to get for their patients.”
Clinicians and healthcare organizations have several sources for guidelines. The Agency for Healthcare Research and Quality (AHRQ) systematically reviews and vets guidelines submitted for inclusion in its National Guideline Clearinghouse (www.guideline.gov), and makes them available for evidence-based clinical decision-making, says Jean Slutsky, director of AHRQ’s Center for Outcomes and Evidence. AHRQ also offers public access to the National Quality Measure Clearinghouse and the Health Care Innovations Exchange, repositories of searchable quality measures and tools relevant to an array of diseases and conditions.
The Institute for Healthcare Improvement (IHI), an independent nonprofit organization, helps frontline physicians implement guidelines, and also helps provider teams decide which guidelines are most appropriate to achieve their desired outcomes, according to Amy E. Boutwell, MD, MPP, IHI’s director of health policy strategy.
Hospitalists use an array of disease-specific practice guidelines from different specialty societies for diagnoses they frequently encounter, such as chest pain, stroke, pneumonia, myocardial infarction, gastrointestinal bleeding, asthma, and chronic obstructive pulmonary disease (COPD). “Most hospitalists want to keep up with the best available evidence,” says Patrick J. Torcson, MD, MMM, FACP, director of hospital medicine at St. Tammany Parish Hospital in Covington, La., and chair of SHM’s Performance and Standards Committee. “The recently updated American College of Cardiology (ACC) and American Heart Association (AHA) guidelines on heart failure are exceptional. The American College of Chest Physicians has an extremely comprehensive set of guidelines on thrombosis, which is the bible for handling anticoagulation.”
Studies are published every month demonstrating that physicians who implement national guidelines at the local level improve such patient outcomes as mortality, length of stay, and time to clinical stability. Dr. Corrigan notes that there are abundant examples of guideline adherence boosting quality outcomes, and cites as a prime example the AHA’s Get With the Guidelines program (www.american heart.org/presenter.jhtml?identifier=1165), which has documented quality gains in coronary artery disease, stroke, and heart failure patients.

—William T. Ford, MD, FHM, section chief of hospital medicine, Temple University Hospital, Philadelphia
Barriers to Acceptance and Adherence
But there are obstacles to guideline adherence, and widespread practice pattern variation remains a huge national problem. Providers in some regions of the country can use twice the resources as their counterparts in other regions and bring no additional benefit to patients (see “Medicare Fee Inspection,” p. 30). The Dartmouth Atlas of Health Care says unwarranted practice variation is responsible for as much as 30% of wasted healthcare spending in the U.S.—a cost that reformers are anxious to eliminate.
The traditional culture of autonomy in the medical profession is perhaps the most difficult and enduring barrier to reducing unjustified practice variation: clinicians don’t automatically follow guidelines, many treat them more as options than as true standards, and organizations do not sufficiently enforce or reward adherence to guidelines, wrote researchers in a special 2005 issue of Health Affairs focusing on guidelines.2
“In an age of mandated cost control and resource limitation under managed care,” the researchers wrote, some physicians still regard practice guidelines as “cookbook medicine” that threatens the use of clinical judgment and encourages treating patients as essentially interchangeable. In the face of that perceived threat, the researchers added, many physicians continue to uphold a traditional view of medicine as an art “in which individual expertise and technique are allowed to shine through and ultimately result in a higher standard of patient care.”
Dr. Corrigan acknowledges the significant obstacles to successful practice guideline implementation:
- Guidelines are developed by various sources, particularly specialty societies, who do not always coordinate their activities. Physicians are left with overlapping and sometimes contradictory guidelines for managing the same disease or condition.
- Guidelines must be maintained and kept current, or physicians will lose confidence and not follow them.
- Guidelines are of varying quality. Some provide clear clinical direction; others are not written in a way that physicians can clearly translate into clinical practice.
- There are significant gaps in the evidence basis for guideline development. Much more comparative effectiveness research needs to be conducted to develop more valid and meaningful guidelines.
- Guidelines must be communicated effectively to physicians, making them available and convenient at the point of clinical care. Electronic health records with user-friendly decision support functions show great promise in “making the right thing the easy thing to do.”
- The fee-for-service payment system encourages greater volume of services, irrespective of guideline recommendations.
Physicians also recognize inherent limitations of guidelines. “Guidelines typically apply across populations. Adding levels of clinical complexity gets further away from a guideline’s applicability. Many physicians will tell you that the patient in front of them is a special case requiring a modification of the protocol,” Dr. Boutwell explains. For example, diabetic management guidelines are based on what is best for a population of diabetics, versus what is best for said hospitalist’s patient who has eight co-morbidities, one of which is diabetes, Boutwell notes. “Guidelines come disease-specific. Patients don’t,” she adds.
Nevertheless, Dr. Boutwell notes, there are robust guidelines and the IHI tries to help front-line physicians and care teams to implement them reliably and effectively.
An obstacle that inhibits hospitalists from implementing guidelines in an optimal fashion “is that we’re not one specialty—we deal with it all—and that complexity can be overwhelming. There is no central repository where all of the guidelines can be found in one place,” according to William T. Ford, MD, FHM, program medical director for Cogent Healthcare and section chief of hospital medicine for Temple University Hospital in Philadelphia.
Make Guidelines Work
Researchers say guidelines are most successful when they are well-supported and uncomplicated, backed by strong leadership and sufficient resources, and are used as “rallying points” to stimulate interdependent and collaborative care among physicians, nurses, pharmacists, equipment suppliers, administrators, and patients.
“Guidelines are really the foundation for determining best practices,” Dr. Torcson says. “There is no shortage of excellent guidelines, or proof that specific interventions do improve outcomes. The key is achieving more uniform implementation. We need tools like pre-printed orders in electronic health records (EHR) to effectively integrate these guidelines into hospitalists’ practice.”
More widespread EHR adoption with user-friendly medical decision-support systems will play a huge role in boosting guideline adoption and effectiveness, says Mary Nix, MS, MT(ASCP)SBB, health science administrator at AHRQ and project officer for the agency’s Center for Outcomes and Evidence.
Dr. Ford says HM groups must evaluate the top 10 to 15 diagnosis-related groups (DRGs) that they see each day (e.g., congestive heart failure, acute kidney failure, pneumonia, cellulitis, or acute coronary syndrome) and come to consensus on which guidelines best address them.
HM groups must then secure buy-in to those guidelines from everyone in the group; from the subspecialists they work with; and from their hospital’s chief medical, financial, and utilization officers.
Care-Transition Guidelines: Opportunity for Hospitalists
A particularly important HM opportunity is improving care transitions. Deficits in communication and information transfer between hospital-based and primary-care physicians (PCPs) are “substantial and ubiquitous,” while delays and omissions are consistently large, and traditional methods of completing and delivering discharge summaries are “suboptimal for communicating timely, accurate, and medically important data to the physicians who will be responsible for follow-up care,” according to a hospitalist-authored Feb. 28, 2007, article in the Journal of the American Medical Association.3 PCPs routinely are not notified about patient admissions or complications during the hospital stay, and some PCPs fail to provide sufficient information to hospitalists at admission, fail to visit or call hospitalized patients, or fail to participate in discharge planning, the study’s researchers noted. For patients with chronic illnesses and frequent hospitalization, those deficits are multiplied, making completeness of information handoffs particularly important.
Because patient handoffs have notoriously been fraught with miscommunication and poor information exchange between providers, adopting a professional consensus on what constitutes the best, safest, and most effective activities during these handoffs is sorely needed.
“Care-transition guidelines can have tremendous power because they affect every hospital patient—each of whom experiences care transitions,” says Rusty Holman, MD, FHM, chief operating officer of Brentwood, Tenn.-based Cogent Healthcare and past president of SHM. “It is an area undergoing rapid development, evolution, and discovery, and hospitalists have positioned themselves as leaders and owners of this particular scenario.”
As care-transition guidelines emerge and mature, Holman thinks they eventually will be tied to value-based healthcare purchasing programs that affect hospitalists’ reimbursement equations and further boost incentives to follow those guidelines. A prime example: Medicare calculated it could save $12 billion annually by reducing preventable 30-day hospital readmissions and will soon stop paying for them. Perhaps 3% to 5% of a hospital’s DRG reimbursement will be at risk under Medicare’s proposal, Dr. Torcson notes.
“Hospitals are going to be much more motivated to build systems and engage physicians, especially hospitalists, to lower readmission rates. Hospitalists will be focusing more and more on how care-transition process improvements can lower those rates,” Dr. Holman says. “That’s a huge opportunity for hospitalists to make a business case for the value they bring to their institutions, and will further justify the financial support they already receive.”
Dr. Ford is more cautious in his appraisal of the financial rewards of better guideline implementation. “We do not capture that much revenue per patient, and even a length-of-stay reduction is difficult for a hospital’s CFO to extrapolate how much money hospitalists save,” he says. “I don’t think hospitalists will be paid more, even if they save the hospital money. You’re just doing your job, but you’re going to keep your job, and you’ll have an enormous bargaining chip when renegotiating contracts with hospitals.”
Still, a prevented readmission might mean a bed for a revenue-generating elective surgery, something that adds to the reward equation.

—Patrick Torcson, MD, MMM, FACP, director of hospital medicine, St. Tammany Parish Hospital, Covington, La., SHM Performance and Standards Committee chair
Transition Evolution
SHM and other sources offer physicians and hospitals expert assistance in implementing care-transition guidelines (see “Care-Transition Guidance,” p. 7). The transitions-of-care policy statement jointly issued by the SHM and five other specialty societies further demonstrates that hospitalists play a key leadership role on this front.1
The policy statement emerged from a multi-stakeholder consensus conference convened by SHM, the American College of Physicians (ACP), and the Society of General Internal Medicine, which was attended by more than 30 medical specialty societies, governmental agencies, and performance measure developers. Participants focused on what standard pieces of information should be exchanged among providers during inpatient to outpatient transitions, and they issued a set of standards for improving those transitions (see “Managing Transitions in Care Between the Inpatient and Outpatient Settings,” p. 7).
“This consensus statement has enormous significance,” Dr. Ford says. “We’re finally shedding light on how to tackle patient handoff and hospital readmission issues, and we as a specialty have to take on care-transition improvement as our mantra. If we were to solve just that one piece, we can more easily start implementing other clinical guidelines. Care-transition guidelines are a fundamental tool to build consensus within your own group and with other clinicians in a team approach.”
Dr. Corrigan applauds the physician groups for publishing the transitions-of-care statement and encourages the societies to work together to “take it to the next step, which is to develop the measures and get them endorsed through the NQF process.”
SHM members are participating in workgroups convened by the NQF to identify standardized performance measures and to develop action plans over the next few months for several national priority areas—one of which is care coordination. “We have a ways to go to achieve better patient handoffs and information exchange between hospitals and other settings in the community. Hospitalists can drive the development of those guidelines and protocols,” Dr. Corrigan says. TH
Christopher Guadagnino, PhD, is a freelance medical writer based in Pennsylvania.
References
- Kripalani S, LeFevre F, Phillips CO, Williams MV; Basaviah P, Baker DW. Deficits in communication and information transfer between hospital-based and primary care physicians: implications for patient safety and continuity of care. JAMA. 2007;297:831-841.
- Timmermans S, Mauck A. The promises and pitfalls of evidence-based medicine. Health Affairs. 2005; 24(1):18-28.
- Snow V, Beck D, Budnitz T, et al. Transitions of care consensus policy statement: American College of Physicians, Society of General Internal Medicine, Society of Hospital Medicine, American Geriatrics Society, American College of Emergency Physicians, Society of Academic Emergency Medicine. J Hosp Med. 2009: 4(6)364-370.
Top Image Source: GOLDEN PIXELS LLC/ALAMY
Evidence-based practice guidelines are key tools to help hospitalists practice high-quality medicine and demonstrate the value of their inpatient care model. Guidelines are designed to produce superior care outcomes and resource utilization efficiencies by encouraging proven medical practices and discouraging ineffective or unproven ones. Yet inefficiencies, variation, and quality gaps persist in medical care—much to the chagrin of policymakers.
Is the answer more guidelines, and better implementation of existing ones?
Research experts and many HM leaders say yes.
In fact, HM is leading the way in an important new area for which there is little uniform guidance: optimal care transitions during patient handoffs. Care transitions are a pivotal time in the patient care process and are replete with avoidable service duplication, poor communication among providers, gaps in care reconciliation, and patient-safety issues.
SHM has joined five other organizations in issuing a Transitions of Care Consensus Policy Statement, which promises more systematic, safe, and efficient patient handoffs.1 SHM also is targeting care-transition improvement in a variety of other venues, all of which can help hospitalists demonstrate more persuasively the value they bring to healthcare delivery.
Guidelines Work
Practice guidelines work, in the sense that they help providers practice in ways consistent with what the best aggregate knowledge and expert opinion says is most effective. The evidence allows physicians to avoid expending scarce resources on ineffective clinical services. Their importance is magnified by the current urgency given to value-based purchasing in healthcare reform. “The right care, for the right patient, at the right time” is the new mantra of payors and policymakers, many of whom are demanding the best and most efficient healthcare delivery at the lowest cost.

“When providers are not providing the right care at the right time to patients, we find that the patient often gets more services … that they didn’t need. That oftentimes exposes them to potential harm and (services) that are wasteful of resources,” says Janet M. Corrigan, PhD, MBA, president and CEO of the National Quality Forum (NQF), a standard-setting organization that convenes national experts to apply “gold standard” endorsement of guidelines developed by professional medical societies and other entities. “Guidelines are a way of synthesizing evidence and translating it into action steps that providers can follow so that they get the best results that we know how to get for their patients.”
Clinicians and healthcare organizations have several sources for guidelines. The Agency for Healthcare Research and Quality (AHRQ) systematically reviews and vets guidelines submitted for inclusion in its National Guideline Clearinghouse (www.guideline.gov), and makes them available for evidence-based clinical decision-making, says Jean Slutsky, director of AHRQ’s Center for Outcomes and Evidence. AHRQ also offers public access to the National Quality Measure Clearinghouse and the Health Care Innovations Exchange, repositories of searchable quality measures and tools relevant to an array of diseases and conditions.
The Institute for Healthcare Improvement (IHI), an independent nonprofit organization, helps frontline physicians implement guidelines, and also helps provider teams decide which guidelines are most appropriate to achieve their desired outcomes, according to Amy E. Boutwell, MD, MPP, IHI’s director of health policy strategy.
Hospitalists use an array of disease-specific practice guidelines from different specialty societies for diagnoses they frequently encounter, such as chest pain, stroke, pneumonia, myocardial infarction, gastrointestinal bleeding, asthma, and chronic obstructive pulmonary disease (COPD). “Most hospitalists want to keep up with the best available evidence,” says Patrick J. Torcson, MD, MMM, FACP, director of hospital medicine at St. Tammany Parish Hospital in Covington, La., and chair of SHM’s Performance and Standards Committee. “The recently updated American College of Cardiology (ACC) and American Heart Association (AHA) guidelines on heart failure are exceptional. The American College of Chest Physicians has an extremely comprehensive set of guidelines on thrombosis, which is the bible for handling anticoagulation.”
Studies are published every month demonstrating that physicians who implement national guidelines at the local level improve such patient outcomes as mortality, length of stay, and time to clinical stability. Dr. Corrigan notes that there are abundant examples of guideline adherence boosting quality outcomes, and cites as a prime example the AHA’s Get With the Guidelines program (www.american heart.org/presenter.jhtml?identifier=1165), which has documented quality gains in coronary artery disease, stroke, and heart failure patients.

—William T. Ford, MD, FHM, section chief of hospital medicine, Temple University Hospital, Philadelphia
Barriers to Acceptance and Adherence
But there are obstacles to guideline adherence, and widespread practice pattern variation remains a huge national problem. Providers in some regions of the country can use twice the resources as their counterparts in other regions and bring no additional benefit to patients (see “Medicare Fee Inspection,” p. 30). The Dartmouth Atlas of Health Care says unwarranted practice variation is responsible for as much as 30% of wasted healthcare spending in the U.S.—a cost that reformers are anxious to eliminate.
The traditional culture of autonomy in the medical profession is perhaps the most difficult and enduring barrier to reducing unjustified practice variation: clinicians don’t automatically follow guidelines, many treat them more as options than as true standards, and organizations do not sufficiently enforce or reward adherence to guidelines, wrote researchers in a special 2005 issue of Health Affairs focusing on guidelines.2
“In an age of mandated cost control and resource limitation under managed care,” the researchers wrote, some physicians still regard practice guidelines as “cookbook medicine” that threatens the use of clinical judgment and encourages treating patients as essentially interchangeable. In the face of that perceived threat, the researchers added, many physicians continue to uphold a traditional view of medicine as an art “in which individual expertise and technique are allowed to shine through and ultimately result in a higher standard of patient care.”
Dr. Corrigan acknowledges the significant obstacles to successful practice guideline implementation:
- Guidelines are developed by various sources, particularly specialty societies, who do not always coordinate their activities. Physicians are left with overlapping and sometimes contradictory guidelines for managing the same disease or condition.
- Guidelines must be maintained and kept current, or physicians will lose confidence and not follow them.
- Guidelines are of varying quality. Some provide clear clinical direction; others are not written in a way that physicians can clearly translate into clinical practice.
- There are significant gaps in the evidence basis for guideline development. Much more comparative effectiveness research needs to be conducted to develop more valid and meaningful guidelines.
- Guidelines must be communicated effectively to physicians, making them available and convenient at the point of clinical care. Electronic health records with user-friendly decision support functions show great promise in “making the right thing the easy thing to do.”
- The fee-for-service payment system encourages greater volume of services, irrespective of guideline recommendations.
Physicians also recognize inherent limitations of guidelines. “Guidelines typically apply across populations. Adding levels of clinical complexity gets further away from a guideline’s applicability. Many physicians will tell you that the patient in front of them is a special case requiring a modification of the protocol,” Dr. Boutwell explains. For example, diabetic management guidelines are based on what is best for a population of diabetics, versus what is best for said hospitalist’s patient who has eight co-morbidities, one of which is diabetes, Boutwell notes. “Guidelines come disease-specific. Patients don’t,” she adds.
Nevertheless, Dr. Boutwell notes, there are robust guidelines and the IHI tries to help front-line physicians and care teams to implement them reliably and effectively.
An obstacle that inhibits hospitalists from implementing guidelines in an optimal fashion “is that we’re not one specialty—we deal with it all—and that complexity can be overwhelming. There is no central repository where all of the guidelines can be found in one place,” according to William T. Ford, MD, FHM, program medical director for Cogent Healthcare and section chief of hospital medicine for Temple University Hospital in Philadelphia.
Make Guidelines Work
Researchers say guidelines are most successful when they are well-supported and uncomplicated, backed by strong leadership and sufficient resources, and are used as “rallying points” to stimulate interdependent and collaborative care among physicians, nurses, pharmacists, equipment suppliers, administrators, and patients.
“Guidelines are really the foundation for determining best practices,” Dr. Torcson says. “There is no shortage of excellent guidelines, or proof that specific interventions do improve outcomes. The key is achieving more uniform implementation. We need tools like pre-printed orders in electronic health records (EHR) to effectively integrate these guidelines into hospitalists’ practice.”
More widespread EHR adoption with user-friendly medical decision-support systems will play a huge role in boosting guideline adoption and effectiveness, says Mary Nix, MS, MT(ASCP)SBB, health science administrator at AHRQ and project officer for the agency’s Center for Outcomes and Evidence.
Dr. Ford says HM groups must evaluate the top 10 to 15 diagnosis-related groups (DRGs) that they see each day (e.g., congestive heart failure, acute kidney failure, pneumonia, cellulitis, or acute coronary syndrome) and come to consensus on which guidelines best address them.
HM groups must then secure buy-in to those guidelines from everyone in the group; from the subspecialists they work with; and from their hospital’s chief medical, financial, and utilization officers.
Care-Transition Guidelines: Opportunity for Hospitalists
A particularly important HM opportunity is improving care transitions. Deficits in communication and information transfer between hospital-based and primary-care physicians (PCPs) are “substantial and ubiquitous,” while delays and omissions are consistently large, and traditional methods of completing and delivering discharge summaries are “suboptimal for communicating timely, accurate, and medically important data to the physicians who will be responsible for follow-up care,” according to a hospitalist-authored Feb. 28, 2007, article in the Journal of the American Medical Association.3 PCPs routinely are not notified about patient admissions or complications during the hospital stay, and some PCPs fail to provide sufficient information to hospitalists at admission, fail to visit or call hospitalized patients, or fail to participate in discharge planning, the study’s researchers noted. For patients with chronic illnesses and frequent hospitalization, those deficits are multiplied, making completeness of information handoffs particularly important.
Because patient handoffs have notoriously been fraught with miscommunication and poor information exchange between providers, adopting a professional consensus on what constitutes the best, safest, and most effective activities during these handoffs is sorely needed.
“Care-transition guidelines can have tremendous power because they affect every hospital patient—each of whom experiences care transitions,” says Rusty Holman, MD, FHM, chief operating officer of Brentwood, Tenn.-based Cogent Healthcare and past president of SHM. “It is an area undergoing rapid development, evolution, and discovery, and hospitalists have positioned themselves as leaders and owners of this particular scenario.”
As care-transition guidelines emerge and mature, Holman thinks they eventually will be tied to value-based healthcare purchasing programs that affect hospitalists’ reimbursement equations and further boost incentives to follow those guidelines. A prime example: Medicare calculated it could save $12 billion annually by reducing preventable 30-day hospital readmissions and will soon stop paying for them. Perhaps 3% to 5% of a hospital’s DRG reimbursement will be at risk under Medicare’s proposal, Dr. Torcson notes.
“Hospitals are going to be much more motivated to build systems and engage physicians, especially hospitalists, to lower readmission rates. Hospitalists will be focusing more and more on how care-transition process improvements can lower those rates,” Dr. Holman says. “That’s a huge opportunity for hospitalists to make a business case for the value they bring to their institutions, and will further justify the financial support they already receive.”
Dr. Ford is more cautious in his appraisal of the financial rewards of better guideline implementation. “We do not capture that much revenue per patient, and even a length-of-stay reduction is difficult for a hospital’s CFO to extrapolate how much money hospitalists save,” he says. “I don’t think hospitalists will be paid more, even if they save the hospital money. You’re just doing your job, but you’re going to keep your job, and you’ll have an enormous bargaining chip when renegotiating contracts with hospitals.”
Still, a prevented readmission might mean a bed for a revenue-generating elective surgery, something that adds to the reward equation.

—Patrick Torcson, MD, MMM, FACP, director of hospital medicine, St. Tammany Parish Hospital, Covington, La., SHM Performance and Standards Committee chair
Transition Evolution
SHM and other sources offer physicians and hospitals expert assistance in implementing care-transition guidelines (see “Care-Transition Guidance,” p. 7). The transitions-of-care policy statement jointly issued by the SHM and five other specialty societies further demonstrates that hospitalists play a key leadership role on this front.1
The policy statement emerged from a multi-stakeholder consensus conference convened by SHM, the American College of Physicians (ACP), and the Society of General Internal Medicine, which was attended by more than 30 medical specialty societies, governmental agencies, and performance measure developers. Participants focused on what standard pieces of information should be exchanged among providers during inpatient to outpatient transitions, and they issued a set of standards for improving those transitions (see “Managing Transitions in Care Between the Inpatient and Outpatient Settings,” p. 7).
“This consensus statement has enormous significance,” Dr. Ford says. “We’re finally shedding light on how to tackle patient handoff and hospital readmission issues, and we as a specialty have to take on care-transition improvement as our mantra. If we were to solve just that one piece, we can more easily start implementing other clinical guidelines. Care-transition guidelines are a fundamental tool to build consensus within your own group and with other clinicians in a team approach.”
Dr. Corrigan applauds the physician groups for publishing the transitions-of-care statement and encourages the societies to work together to “take it to the next step, which is to develop the measures and get them endorsed through the NQF process.”
SHM members are participating in workgroups convened by the NQF to identify standardized performance measures and to develop action plans over the next few months for several national priority areas—one of which is care coordination. “We have a ways to go to achieve better patient handoffs and information exchange between hospitals and other settings in the community. Hospitalists can drive the development of those guidelines and protocols,” Dr. Corrigan says. TH
Christopher Guadagnino, PhD, is a freelance medical writer based in Pennsylvania.
References
- Kripalani S, LeFevre F, Phillips CO, Williams MV; Basaviah P, Baker DW. Deficits in communication and information transfer between hospital-based and primary care physicians: implications for patient safety and continuity of care. JAMA. 2007;297:831-841.
- Timmermans S, Mauck A. The promises and pitfalls of evidence-based medicine. Health Affairs. 2005; 24(1):18-28.
- Snow V, Beck D, Budnitz T, et al. Transitions of care consensus policy statement: American College of Physicians, Society of General Internal Medicine, Society of Hospital Medicine, American Geriatrics Society, American College of Emergency Physicians, Society of Academic Emergency Medicine. J Hosp Med. 2009: 4(6)364-370.
Top Image Source: GOLDEN PIXELS LLC/ALAMY